Attorney Docket No. ASET-043/001WO 325190-2258 FORMS OF QUINOXALINE DERIVATIVES AND RELATED USES THEREOF CROSS-REFERENCE TO RELATED APPLICATIONS [001] This application claims priority to and the benefit of U.S. Provisional Application No. 63/604,977, filed on December 1, 2023, which is incorporated by reference herein in its entirety for all purposes. BACKGROUND [002] Specific mutations in the human genes FGFR2 and FGFR3, which encode for the proteins FGFR2 and FGFR3 respectively, have been associated with several different types of cancers. A variety of different inhibitors for FGFR2 and/or FGFR3 have been developed for the treatment of cancer, including FDA-approved drugs such as erdafitinib and pemigatinib. However, existing inhibitors exhibit a variety of flaws that limit their effectiveness in the clinical. First, some of the existing inhibitors target only FGFR2 and fail to inhibit FGFR3, limiting their ability to treat certain types of cancers. Additionally, several of the existing inhibitors also target FGFR1, leading to dose-limiting toxicities such as hyperphosphatemia. Finally, following administration of some existing inhibitors, many patients develop additional mutations in FGFR2 and/or FGFR3, referred to as gatekeeper mutations, that results in resistance to the existing inhibitors. Thus, there is a long-felt need in the art for new therapies that specifically target FGFR2 and FGFR3. The present disclosure provides morphic forms of Compound No. 1 for preventing or treating cancer in patients with overexpression of and/or oncogenic mutations in FGFR2 and/or FGFR3, and the FGFR2 and/or FGFR3. SUMMARY [003] In some aspects, the present disclosure provides a form of Compound No.1:
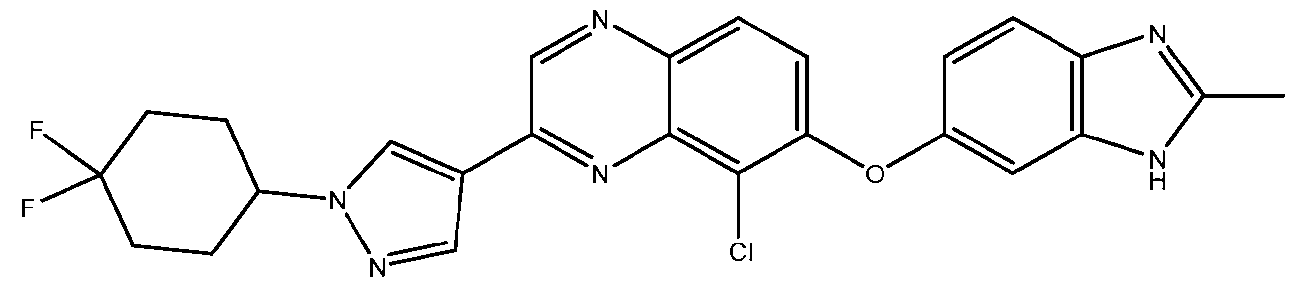
a solvate thereof, a hydrate thereof, or a pharmaceutically acceptable salt thereof. [004] In some aspects, the present disclosure provides a pharmaceutical composition comprising a form of Compound No. 1, a solvate thereof, a hydrate thereof, or a pharmaceutically acceptable salt thereof.
Attorney Docket No. ASET-043/001WO 325190-2258 [005] In some aspects, the present disclosure provides methods of treating or preventing cancer in a subject, the method comprising administering to the subject a form of Compound No.1, a solvate thereof, a hydrate thereof, or a pharmaceutically acceptable salt thereof. [006] In some aspects, the present disclosure provides a form of Compound No.1, a solvate thereof, a hydrate thereof, or a pharmaceutically acceptable salt thereof, for treating or preventing cancer in a subject. [007] In some aspects, the present disclosure provides a use of a form of Compound No. 1, a solvate thereof, a hydrate thereof, or a pharmaceutically acceptable salt thereof, in the manufacture of a medicament for treating or preventing cancer in a subject. [008] Unless otherwise defined, all technical and scientific terms used herein have the same meaning as commonly understood by one of ordinary skill in the art to which this disclosure belongs. In the specification, the singular forms also include the plural unless the context clearly dictates otherwise. Although methods and materials similar or equivalent to those described herein can be used in the practice or testing of the present disclosure, suitable methods and materials are described below. All publications, patent applications, patents and other references mentioned herein are incorporated by reference. The references cited herein are not admitted to be prior art to the claimed invention. In the case of conflict, the present specification, including definitions, will control. In addition, the materials, methods and examples are illustrative only and are not intended to be limiting. In the case of conflict between the chemical structures and names of the compounds disclosed herein, the chemical structures will control. [009] Other features and advantages of the disclosure will be apparent from the following detailed description and claims. BRIEF DESCRIPTIONS OF FIGURES [010] FIG.1 depicts the XRPD pattern of Form A of Compound No.1. [011] FIG.2 depicts the DSC thermogram of Form A of Compound No.1. [012] FIG.3 depicts the TGA thermogram of Form A of Compound No.1. [013] FIG.4 depicts the XRPD pattern of Form B of Compound No.1. [014] FIG.5 depicts the DSC thermogram of Form B of Compound No.1. [015] FIG.6 depicts the TGA thermogram of Form B of Compound No.1. [016] FIG.7 depicts the XRPD pattern of Form C of Compound No.1. [017] FIG.8 depicts the DSC thermogram of Form C of Compound No.1. [018] FIG.9 depicts the TGA thermogram of Form C of Compound No.1.
Attorney Docket No. ASET-043/001WO 325190-2258 [019] FIG.10 depicts the XRPD pattern of Form D of Compound No.1. [020] FIG.11 depicts the DSC thermogram of Form D of Compound No.1. [021] FIG.12 depicts the TGA thermogram of Form D of Compound No.1. [022] FIG. 13 depicts the TGA thermogram of a composition comprising an amorphous form of Compound No.1 and HPMC E3. [023] FIG. 14 depicts the TGA thermogram of a composition comprising an amorphous form of Compound No.1 and HPMC ASMG. [024] FIG. 15 depicts the TGA thermogram of a composition comprising an amorphous form of Compound No.1 and Eudragit
® E100. [025] FIG. 16 depicts the mDSC thermogram of a composition comprising an amorphous form of Compound No.1 and PVP K30. [026] FIG. 17 depicts the mDSC thermogram of a composition comprising an amorphous form of Compound No.1 and PVPVA64. [027] FIG. 18 depicts the mDSC thermogram of a composition comprising an amorphous form of Compound No.1 and Soluplus
®. [028] FIG. 19 depicts the mDSC thermogram of a composition comprising an amorphous form of Compound No.1 and HPMC E3. [029] FIG. 20 depicts the mDSC thermogram of a composition comprising an amorphous form of Compound No.1 and HPMC ASLG. [030] FIG. 21 depicts the mDSC thermogram of a composition comprising an amorphous form of Compound No.1 and HPMC ASMG. [031] FIG. 22 depicts the mDSC thermogram of a composition comprising an amorphous form of Compound No.1 and HPMC ASHG. [032] FIG. 23 depicts the mDSC thermogram of a composition comprising an amorphous form of Compound No.1 and HPC-SSL. [033] FIG. 24 depicts the mDSC thermogram of a composition comprising an amorphous form of Compound No.1 and Eudragit
® E100. [034] FIG. 25 depicts the mDSC thermogram of a composition comprising an amorphous form of Compound No.1 and Eudragit
® L100. DETAILED DESCRIPTION [035] It is understood that the term “Compound No.1,” as used herein, refers to a compound having the following structure:
Attorney Docket No. ASET-043/001WO 325190-2258 [036] In

a solvate thereof, a hydrate thereof, or a pharmaceutically acceptable salt thereof. [037] In some aspects, the present disclosure provides a morphic form of Compound No.1, a solvate thereof, a hydrate thereof, or a pharmaceutically acceptable salt thereof. [038] In some embodiments, the morphic form is a crystalline form. [039] In some embodiments, the morphic form is a morphic form (e.g., crystalline form) of Compound No.1, the solvate thereof, or the hydrate thereof. [040] In some embodiments, the morphic form is a morphic form (e.g., crystalline form) of Compound No.1. [041] In some embodiments, the morphic form is a morphic form (e.g., crystalline form) of a solvate (e.g., heterosolvate) of Compound No.1. [042] In some embodiments, the morphic form is a morphic form (e.g., crystalline form) of a hydrate of Compound No.1. [043] In some embodiments, the morphic form is a morphic form (e.g., crystalline form) of Compound No.1 or the pharmaceutically acceptable salt thereof. [044] In some embodiments, the morphic form is a morphic form (e.g., crystalline form) of a pharmaceutically acceptable salt of Compound No.1. [045] In some embodiments, the morphic form is Form A of Compound No. 1, the solvate thereof, the hydrate thereof, or the pharmaceutically acceptable salt thereof. [046] In some embodiments, the morphic form is Form B of Compound No. 1, the solvate thereof, the hydrate thereof, or the pharmaceutically acceptable salt thereof. [047] In some embodiments, the morphic form is Form C of Compound No. 1, the solvate thereof, the hydrate thereof, or the pharmaceutically acceptable salt thereof. [048] In some embodiments, the morphic form is Form D of Compound No. 1, the solvate thereof, the hydrate thereof, or the pharmaceutically acceptable salt thereof. [049] In some aspects, the present disclosure provides a method of preparing a morphic form of Compound No.1, the solvate thereof, the hydrate thereof, or the pharmaceutically acceptable salt thereof.
Attorney Docket No. ASET-043/001WO 325190-2258 Form A [050] In some embodiments, Form A is a crystalline form. [051] In some embodiments, Form A is a morphic form (e.g., crystalline form) of Compound No.1, the pharmaceutically acceptable salt thereof, the solvate thereof, or the hydrate thereof. [052] In some embodiments, the Form A is a morphic form (e.g., crystalline form) of Compound No.1. X-Ray Powder Diffraction (XRPD) Characterization [053] In some embodiments, Form A is characterized by an X-ray diffraction (“XRPD”) pattern comprising signals (e.g., peaks) at 9.8±0.2, 16.2±0.2, and 23.8±0.2 °2θ (e.g., 9.8±0.1, 16.2±0.1, and 23.8±0.1 °2θ (e.g., 9.8, 16.2, and 23.8 °2θ)) using Cu Kα radiation. [054] In some embodiments, the XRPD pattern of Form A further comprises at least one signal (e.g., peak) selected from 19.7±0.2, 25.6±0.2, and 28.1±0.2 °2θ (e.g., 19.7±0.1, 25.6±0.1, and 28.1±0.1 °2θ (e.g., 19.7, 25.6, and 28.1 °2θ)) using Cu Kα radiation. [055] In some embodiments, the XRPD pattern of Form A further comprises at least two signals (e.g., peaks) selected from 19.7±0.2, 25.6±0.2, and 28.1±0.2 °2θ (e.g., 19.7±0.1, 25.6±0.1, and 28.1±0.1 °2θ (e.g., 19.7, 25.6, and 28.1 °2θ)) using Cu Kα radiation. [056] In some embodiments, the XRPD pattern of Form A further comprises signals (e.g., peaks) at 19.7±0.2, 25.6±0.2, and 28.1±0.2 °2θ (e.g., 19.7±0.1, 25.6±0.1, and 28.1±0.1 °2θ (e.g., 19.7, 25.6, and 28.1 °2θ)) using Cu Kα radiation. [057] In some embodiments, Form A is characterized by an X-ray diffraction (“XRPD”) pattern comprising at least three signals (e.g., peaks) selected from 9.8±0.2, 16.2±0.2, 19.7±0.2, 23.8±0.2, 25.6±0.2, and 28.1±0.2 °2θ (e.g., 9.8±0.1, 16.2±0.1, 19.7±0.1, 23.8±0.1, 25.6±0.1, and 28.1±0.1 °2θ (e.g., 9.8, 16.2, 19.7, 23.8, 25.6, and 28.1 °2θ)) using Cu Kα radiation. [058] In some embodiments, the XRPD pattern of Form A comprises at least four signals (e.g., peaks) selected from 9.8±0.2, 16.2±0.2, 19.7±0.2, 23.8±0.2, 25.6±0.2, and 28.1±0.2 °2θ (e.g., 9.8±0.1, 16.2±0.1, 19.7±0.1, 23.8±0.1, 25.6±0.1, and 28.1±0.1 °2θ (e.g., 9.8, 16.2, 19.7, 23.8, 25.6, and 28.1 °2θ)) using Cu Kα radiation. [059] In some embodiments, the XRPD pattern of Form A comprises at least five signals (e.g., peaks) selected from 9.8±0.2, 16.2±0.2, 19.7±0.2, 23.8±0.2, 25.6±0.2, and 28.1±0.2 °2θ (e.g., 9.8±0.1, 16.2±0.1, 19.7±0.1, 23.8±0.1, 25.6±0.1, and 28.1±0.1 °2θ (e.g., 9.8, 16.2, 19.7, 23.8, 25.6, and 28.1 °2θ)) using Cu Kα radiation. [060] In some embodiments, the XRPD pattern of Form A comprises signals (e.g., peaks) at 9.8±0.2, 16.2±0.2, 19.7±0.2, 23.8±0.2, 25.6±0.2, and 28.1±0.2 °2θ (e.g., 9.8±0.1, 16.2±0.1,
Attorney Docket No. ASET-043/001WO 325190-2258 19.7±0.1, 23.8±0.1, 25.6±0.1, and 28.1±0.1 °2θ (e.g., 9.8, 16.2, 19.7, 23.8, 25.6, and 28.1 °2θ)) using Cu Kα radiation. [061] In some embodiments, the XRPD pattern of Form A further comprises at least one signal (e.g., peak) selected from 11.8±0.2, 18.7±0.2, 19.0±0.2, and 21.2±0.2 °2θ (e.g., 11.8±0.1, 18.7±0.1, 19.0±0.1, and 21.2±0.1 °2θ (e.g., 11.8, 18.7, 19.0, and 21.2 °2θ)) using Cu Kα radiation. [062] In some embodiments, the XRPD pattern of Form A further comprises at least two signals (e.g., peaks) selected from 11.8±0.2, 18.7±0.2, 19.0±0.2, and 21.2±0.2 °2θ (e.g., 11.8±0.1, 18.7±0.1, 19.0±0.1, and 21.2±0.1 °2θ (e.g.11.8, 18.7, 19.0, and 21.2 °2θ)) using Cu Kα radiation. [063] In some embodiments, the XRPD pattern of Form A further comprises at least three signals (e.g., peaks) selected from 11.8±0.2, 18.7±0.2, 19.0±0.2, and 21.2±0.2 °2θ (e.g., 11.8±0.1, 18.7±0.1, 19.0±0.1, and 21.2±0.1 °2θ (e.g.11.8, 18.7, 19.0, and 21.2 °2θ)) using Cu Kα radiation. [064] In some embodiments, the XRPD pattern of Form A further comprises signals (e.g., peaks) at 9.8±0.2, 11.8±0.2, 16.2±0.2, 18.7±0.2, 19.0±0.2, 19.7±0.2, 21.2±0.2, 23.8±0.2, 25.6±0.2, and 28.1±0.2 °2θ (e.g. 9.8±0.1, 11.8±0.1, 16.2±0.1, 18.7±0.1, 19.0±0.1, 19.7±0.1, 21.2±0.1, 23.8±0.1, 25.6±0.1, and 28.1±0.1 °2θ (e.g. 9.8, 11.8, 16.2, 18.7, 19.0, 19.7, 21.2, 23.8, 25.6, and 28.1 °2θ)) using Cu Kα radiation. [065] In some embodiments, Form A is characterized by an XRPD pattern substantially similar to that shown in FIG.1. [066] In some embodiments, Form A is characterized by an XRPD pattern comprising one or more of the signals (e.g., peaks) at approximately the positions shown in Table A. [067] In some embodiments, Form A is characterized by an XRPD pattern comprising two or more of the signals (e.g., peaks) at approximately the positions shown in Table A. [068] In some embodiments, Form A is characterized by an XRPD pattern comprising three or more of the signals (e.g., peaks) at approximately the positions shown in Table A. [069] In some embodiments, Form A is characterized by an XRPD pattern comprising four or more of the signals (e.g., peaks) at approximately the positions shown in Table A. [070] In some embodiments, Form A is characterized by an XRPD pattern comprising five or more of the signals (e.g., peaks) at approximately the positions shown in Table A. [071] In some embodiments, Form A is characterized by an XRPD pattern comprising six or more of the signals (e.g., peaks) at approximately the positions shown in Table A.
Attorney Docket No. ASET-043/001WO 325190-2258 [072] In some embodiments, Form A is characterized by an XRPD pattern comprising seven or more of the signals (e.g., peaks) at approximately the positions shown in Table A. [073] In some embodiments, Form A is characterized by an XRPD pattern comprising eight or more of the signals (e.g., peaks) at approximately the positions shown in Table A. [074] In some embodiments, Form A is characterized by an XRPD pattern comprising nine or more of the signals (e.g., peaks) at approximately the positions shown in Table A. [075] In some embodiments, Form A is characterized by an XRPD pattern comprising 10 or more of the signals (e.g., peaks) at approximately the positions shown in Table A. [076] In some embodiments, Form A is characterized by an XRPD pattern comprising signals (e.g., peaks) at approximately the positions shown in Table A. [077] It is understood that the values in the table are approximate and subject to instrumental and experimental variations. Table A: Exemplary XRPD Signal List for Form A Angle d Value Net Intensity Gross Intensity Rel. Intensity 3.535 ° 24.97213 Å 16.0794 44.9325 1.7%
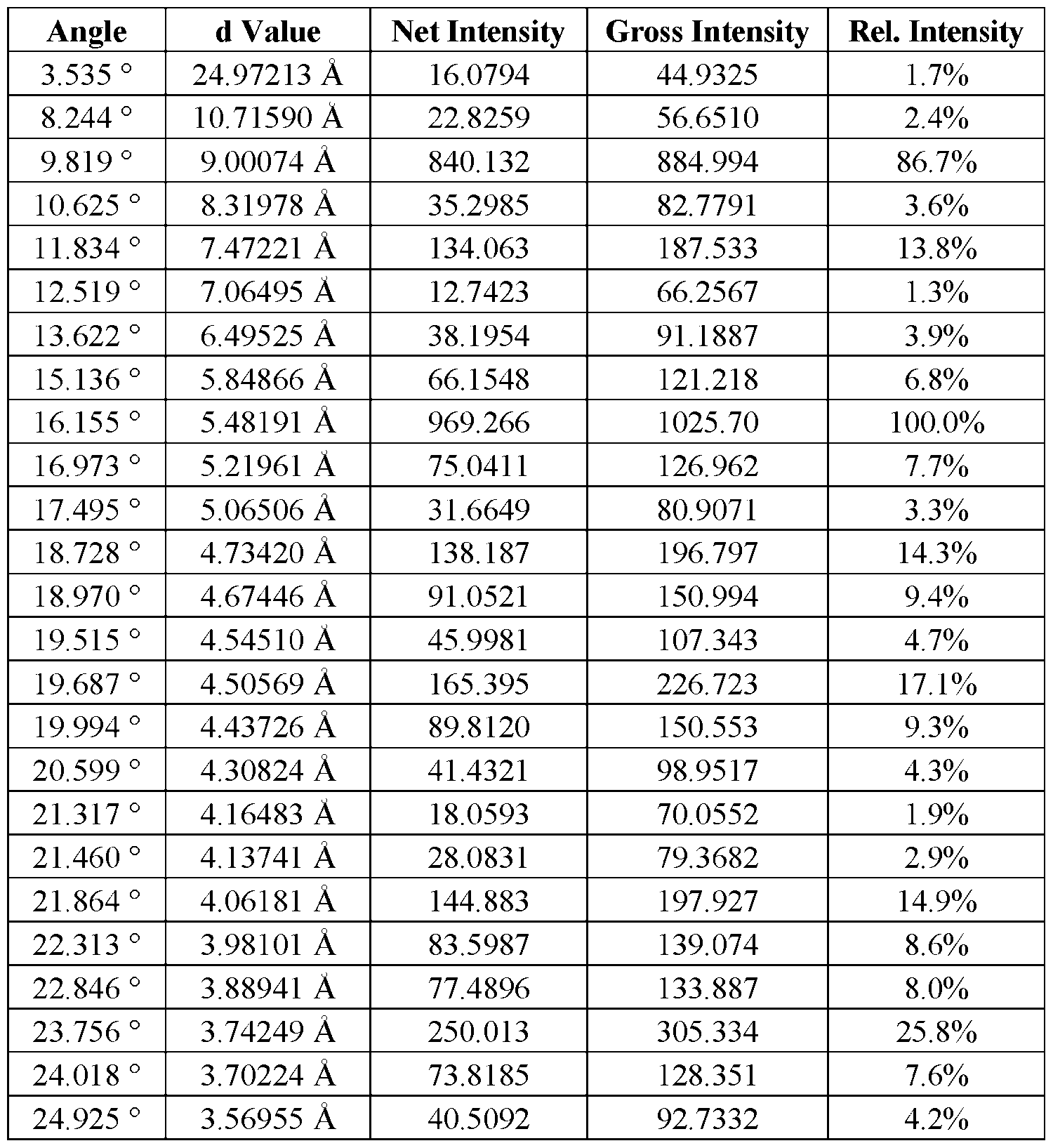
Attorney Docket No. ASET-043/001WO 325190-2258 25.598 ° 3.47715 Å 164.892 215.704 17.0% 27.009 ° 3.29865 Å 16.6736 63.6895 1.7% Differ

[078] In some embodiments, Form A is characterized by a DSC curve having an endothermic signal (e.g., peak) at 298 ± 20°C (e.g., 298 ± 10 °C (e.g., 298 ± 5 °C (e.g., 298 ± 4 °C (e.g., 298 ± 3 °C (e.g., 298 ± 2 °C (e.g., 298 ± 1 °C (e.g., 298 ± 0.5 °C))))))). [079] In some embodiments, Form A is characterized by a DSC curve having an endothermic signal (e.g., peak) at about 298 °C. [080] In some embodiments, Form A is characterized by a DSC curve having an endothermic signal (e.g., peak) associated with an enthalpy of about 105 ± 9 J/g (e.g., 105 ± 8 J/g (e.g., 105 ± 7 J/g (e.g., 105 ± 6 J/g (e.g., 105 ± 5 J/g (e.g., 105 ± 4 J/g (e.g., 105 ± 3 J/g (e.g., 105 ± 2 J/g))))))). [081] In some embodiments, Form A is characterized by a DSC curve having an endothermic signal (e.g., peak) associated with an enthalpy of about 105 J/g. [082] In some embodiments, Form A is characterized by a DSC curve substantially similar to that shown in FIG.2. Thermogravimetric Analysis (TGA) Characterizations [083] In some embodiments, Form A shows a weight loss of from about 0.5% to about 3%, at a temperature range from about 25 ± 20 °C (e.g., 25 ± 10 °C (e.g., 25 ± 5 °C (e.g., 25 ± 4 °C (e.g., 25 ± 3 °C (e.g., 25 ± 2 °C (e.g., 25 ± 1 °C (e.g., 25 ± 0.5 °C))))))) to about 125 ± 20 °C (e.g., 125 ± 10 °C (e.g., 125 ± 5 °C (e.g., 125 ± 4 °C (e.g., 125 ± 3 °C (e.g., 125 ± 2 °C (e.g., 125 ± 1 °C (e.g., 125 ± 0.5 °C))))))), as measured by TGA.
Attorney Docket No. ASET-043/001WO 325190-2258 [084] In some embodiments, Form A shows a weight loss of about 1.4%, at a temperature range from about 25 °C to about 125 °C, as measured by TGA. [085] In some embodiments, Form A is characterized by a TGA profile substantially similar to that shown in FIG.3. Properties [086] In some embodiments, the purity (e.g., chemical purity or morphic purity) of Form A is more than about 99.0 %, more than about 99.1 %, more than about 99.2 %, more than about 99.3 %, more than about 99.4 %, more than about 99.5 %, more than about 99.6 %, more than about 99.7 %, more than about 99.8 %, or more than about 99.9 %. [087] In some embodiments, the impurity (e.g., chemical impurity or morphic impurity) present in Form A is less than about 5%, less than about 4%, less than about 3%, less than about 2%, less than about 1%, less than about 0.5%, less than about 0.4%, less than about 0.3%, less than about 0.2%, or less than about 0.1%. [088] In some embodiments, the purity (e.g., chemical purity or morphic purity) of Form A is more than about 95 %, more than about 96 %, more than about 97 %, more than about 98 %, or more than about 99 %, in 10% HP-β-CD at a concentration of about 9 mg/mL, after being placed at room temperature (e.g., about 25 °C) for about two days, about six days, or about eight days. [089] In some embodiments, the impurity (e.g., chemical impurity or morphic impurity) present in Form A is less than about 5%, less than about 4%, less than about 3%, less than about 2%, less than about 1%, less than about 0.5%, less than about 0.4%, less than about 0.3%, less than about 0.2%, or less than about 0.1%, in 10% HP-β-CD at a concentration of about 9 mg/mL, after being placed at room temperature (e.g., about 25 °C) for about two days, about six days, or about eight days [090] In some embodiments, the purity (e.g., chemical purity or morphic purity) of Form A is more than about 95%, more than about 96%, more than about 97%, more than about 98%, or more than about 99%, in 10% HP-β-CD at a concentration of about 12 mg/mL, after being placed at room temperature (e.g., about 25 °C) for about two days, about six days, or about eight days. [091] In some embodiments, the impurity (e.g., chemical impurity or morphic impurity) present in Form A is less than about 5%, less than about 4%, less than about 3%, less than about 2%, less than about 1%, less than about 0.5%, less than about 0.4%, less than about 0.3%, less than about 0.2%, or less than about 0.1%, in 10% HP-β-CD at a concentration of about 12
Attorney Docket No. ASET-043/001WO 325190-2258 mg/mL, after being placed at room temperature (e.g., about 25 °C) for about two days, about six days, or about eight days. [092] It is understood that “10% HP-β-CD” is prepared by dissolving about 1.5g HP-β-CD in about 15 mL water. It is understood that 9 mg/ml of Form A in 10% HP-β-CD was prepared by mixing about 18 mg of Form A and about 2.0 mL of 10% HP-β-CD. It is understood that 12 mg/ml of Form A in 10% HP-β-CD was prepared by mixing about 24 mg of Form A and about 2.0 mL of 10% HP-β-CD. [093] In some embodiments, the purity (e.g., chemical purity or morphic purity) of Form A is more than about 95%, more than about 96%, more than about 97%, more than about 98%, or more than about 99%, in 0.2% Tween 80 + 0.5% CMC Na at a concentration of about 9 mg/mL, after being placed at room temperature (e.g., about 25 °C) for about two days, about six days, or about eight days. [094] In some embodiments, the impurity (e.g., chemical impurity or morphic impurity) present in Form A is less than about 5%, less than about 4%, less than about 3%, less than about 2%, less than about 1%, less than about 0.5%, less than about 0.4%, less than about 0.3%, less than about 0.2%, or less than about 0.1%, in 0.2% Tween 80 + 0.5% CMC Na at a concentration of about 9 mg/mL, after being placed at room temperature (e.g., about 25 °C) for about two days, about six days, or about eight days. [095] In some embodiments, the purity (e.g., chemical purity or morphic purity) of Form A is more than about 95%, more than about 96%, more than about 97%, more than about 98%, or more than about 99%, in 0.2% Tween 80 + 0.5% CMC Na at a concentration of about 12 mg/mL, after being placed at room temperature (e.g., about 25 °C) for about two days, about six days, or about eight days. [096] In some embodiments, the impurity (e.g., chemical impurity or morphic impurity) present in Form A is less than about 5%, less than about 4%, less than about 3%, less than about 2%, less than about 1%, less than about 0.5%, less than about 0.4%, less than about 0.3%, less than about 0.2%, or less than about 0.1%, in 0.2% Tween 80 + 0.5% CMC Na at a concentration of about 9 mg/mL, after being placed at room temperature (e.g., about 25 °C) for about two days, about six days, or about eight days. [097] It is understood that “0.2% Tween 80 + 0.5% CMC Na” is prepared by dissolving about 30 mg Tween 80 and about 75 mg CMC Na in about 15 mL water. It is understood that 9 mg/mL of Form A in 0.2% Tween 80 + 0.5% CMC Na was prepared by mixing about 18 mg of Form A and about 2.0 mL of 0.2% Tween 80 in 0.5% CMC Na. It is understood that 12
Attorney Docket No. ASET-043/001WO 325190-2258 mg/mL of Form A in 0.2% Tween 80 + 0.5% CMC Na was prepared by mixing about 24 mg of Form A and about 2.0 mL of 0.2% Tween 80 in 0.5% CMC Na. [098] In some embodiments, the purity (e.g., chemical purity or morphic purity) of Form A is more than about 95%, more than about 96%, more than about 97%, more than about 98%, or more than about 99%, after being placed at 40°C and 75% RH for about a week. [099] In some embodiments, the impurity (e.g., chemical impurity or morphic impurity) present in Form A is less than about 5%, less than about 4%, less than about 3%, less than about 2%, less than about 1%, less than about 0.5%, less than about 0.4%, less than about 0.3%, less than about 0.2%, or less than about 0.1%, after being placed at 40°C and 75% RH for about a week. [0100] In some embodiments, the purity (e.g., chemical purity or morphic purity) of Form A is more than about 95%, more than about 96%, more than about 97%, more than about 98%, or more than about 99%, after being placed at 60°C for a week. [0101] In some embodiments, the impurity (e.g., chemical impurity or morphic impurity) present in Form A is less than about 5%, less than about 4%, less than about 3%, less than about 2%, less than about 1%, less than about 0.5%, less than about 0.4%, less than about 0.3%, less than about 0.2%, or less than about 0.1%, after being placed at 60°C for a week. Form B [0102] In some embodiments, Form B is a crystalline form. [0103] In some embodiments, Form B is a morphic form (e.g., crystalline form) of Compound No.1, the solvate thereof, the hydrate thereof, or the pharmaceutically acceptable salt thereof. [0104] In some embodiments, Form B is a morphic form (e.g., crystalline form) of Compound No.1 or the pharmaceutically acceptable salt thereof. [0105] In some embodiments, Form B is a morphic form (e.g., crystalline form) of a pharmaceutically acceptable salt of Compound No.1. [0106] In some embodiments, Form B is a hydrochloride salt of Compound No.1. [0107] In some embodiments, Form B is a morphic form (e.g., crystalline form) of a hydrochloride salt of Compound No.1. [0108] In some embodiments, Form B is characterized by an X-ray diffraction (“XRPD”) pattern comprising signals (e.g., peaks) at 6.0±0.2, 17.1±0.2, and 19.6±0.2 °2θ (e.g., 6.0±0.1, 17.1±0.1, and 19.6±0.1 °2θ (e.g., 6.0, 17.1, and 19.6 °2θ)) using Cu Kα radiation.
Attorney Docket No. ASET-043/001WO 325190-2258 [0109] In some embodiments, the XRPD pattern of Form B further comprises at least one signal (e.g., peak) selected from 15.9±0.2, 18.1±0.2, and 33.4±0.2 °2θ (e.g., 15.9±0.1, 18.1±0.1, and 33.4±0.1 °2θ (e.g., 15.9, 18.1, and 33.4 °2θ)) using Cu Kα radiation. [0110] In some embodiments, the XRPD pattern of Form B further comprises at least two signals (e.g., peaks) selected from 15.9±0.2, 18.1±0.2, and 33.4±0.2 °2θ (e.g., 15.9±0.1, 18.1±0.1, and 33.4±0.1 °2θ (e.g., 15.9, 18.1, and 33.4 °2θ)) using Cu Kα radiation. [0111] In some embodiments, the XRPD pattern of Form B further comprises signals (e.g., peaks) at 15.9±0.2, 18.1±0.2, and 33.4±0.2 °2θ (e.g., 15.9±0.1, 18.1±0.1, and 33.4±0.1 °2θ (e.g., 15.9, 18.1, and 33.4 °2θ)) using Cu Kα radiation. [0112] In some embodiments, Form B is characterized by an X-ray diffraction (“XRPD”) pattern comprising at least three signals (e.g., peaks) selected from 6.0±0.2, 15.9±0.2, 17.1±0.2, 18.1±0.2, 19.6±0.2, and 33.4±0.2 °2θ (e.g., 6.0±0.1, 15.9±0.1, 17.1±0.1, 18.1±0.1, 19.6±0.1, and 33.4±0.1 °2θ (e.g., 6.0, 15.9, 17.1, 18.1, 19.6, and 33.4 °2θ)) using Cu Kα radiation. [0113] In some embodiments, the XRPD pattern of Form B comprises at least four signals (e.g., peaks) selected from 6.0±0.2, 15.9±0.2, 17.1±0.2, 18.1±0.2, 19.6±0.2, and 33.4±0.2 °2θ (e.g., 6.0±0.1, 15.9±0.1, 17.1±0.1, 18.1±0.1, 19.6±0.1, and 33.4±0.1 °2θ (e.g., 6.0, 15.9, 17.1, 18.1, 19.6, and 33.4 °2θ)) using Cu Kα radiation. [0114] In some embodiments, the XRPD pattern of Form B comprises at least five signals (e.g., peaks) selected from 6.0±0.2, 15.9±0.2, 17.1±0.2, 18.1±0.2, 19.6±0.2, and 33.4±0.2 °2θ (e.g., 6.0±0.1, 15.9±0.1, 17.1±0.1, 18.1±0.1, 19.6±0.1, and 33.4±0.1 °2θ (e.g., 6.0, 15.9, 17.1, 18.1, 19.6, and 33.4 °2θ)) using Cu Kα radiation. [0115] In some embodiments, the XRPD pattern of Form B comprises signals (e.g., peaks) at 6.0±0.2, 15.9±0.2, 17.1±0.2, 18.1±0.2, 19.6±0.2, and 33.4±0.2 °2θ (e.g., 6.0±0.1, 15.9±0.1, 17.1±0.1, 18.1±0.1, 19.6±0.1, and 33.4±0.1 °2θ (e.g., 6.0, 15.9, 17.1, 18.1, 19.6, and 33.4 °2θ)) using Cu Kα radiation. [0116] In some embodiments, the XRPD pattern of Form B further comprises at least one signal (e.g., peak) selected from 9.0±0.2, 11.3±0.2, 21.1±0.2, and 30.3±0.2 °2θ (e.g., 9.0±0.1, 11.3±0.1, 21.1±0.1, and 30.3±0.1 °2θ (e.g., 9.0, 11.3, 21.1, and 30.3 °2θ)) using Cu Kα radiation. [0117] In some embodiments, the XRPD pattern of Form B further comprises at least two signals (e.g., peaks) selected from 9.0±0.2, 11.3±0.2, 21.1±0.2, and 30.3±0.2 °2θ (e.g., 9.0±0.1, 11.3±0.1, 21.1±0.1, and 30.3±0.1 °2θ (e.g., 9.0, 11.3, 21.1, and 30.3 °2θ)) using Cu Kα radiation.
Attorney Docket No. ASET-043/001WO 325190-2258 [0118] In some embodiments, the XRPD pattern of Form B further comprises at least three signals (e.g., peaks) selected from 9.0±0.2, 11.3±0.2, 21.1±0.2, and 30.3±0.2 °2θ (e.g., 9.0±0.1, 11.3±0.1, 21.1±0.1, and 30.3±0.1 °2θ (e.g., 9.0, 11.3, 21.1, and 30.3 °2θ)) using Cu Kα radiation. [0119] In some embodiments, the XRPD pattern of Form B further comprises signals (e.g., peaks) at 6.0±0.2, 9.0±0.2, 11.3±0.2, 15.9±0.2, 17.1±0.2, 18.1±0.2, 19.6±0.2, 21.1±0.2, 30.3±0.2, and 33.4±0.2 °2θ (e.g. 6.0±0.1, 9.0±0.1, 11.3±0.1, 15.9±0.1, 17.1±0.1, 18.1±0.1, 19.6±0.1, 21.1±0.1, 30.3±0.1, and 33.4±0.1 °2θ (e.g.6.0, 9.0, 11.3, 15.9, 17.1, 18.1, 19.6, 21.1, 30.3, and 33.4 °2θ)) using Cu Kα radiation. [0120] In some embodiments, Form B is characterized by an XRPD pattern substantially similar to that shown in FIG.4. [0121] In some embodiments, Form B is characterized by an XRPD pattern comprising one or more signals (e.g., peaks) at approximately the positions shown in Table B. [0122] In some embodiments, Form B is characterized by an XRPD pattern comprising two or more of the signals (e.g., peaks) at approximately the positions shown in Table B. [0123] In some embodiments, Form B is characterized by an XRPD pattern comprising three or more of the signals (e.g., peaks) at approximately the positions shown in Table B. [0124] In some embodiments, Form B is characterized by an XRPD pattern comprising four or more of the signals (e.g., peaks) at approximately the positions shown in Table B. [0125] In some embodiments, Form B is characterized by an XRPD pattern comprising five or more of the signals (e.g., peaks) at approximately the positions shown in Table B. [0126] In some embodiments, Form B is characterized by an XRPD pattern comprising six or more of the signals (e.g., peaks) at approximately the positions shown in Table B. [0127] In some embodiments, Form B is characterized by an XRPD pattern comprising seven or more of the signals (e.g., peaks) at approximately the positions shown in Table B. [0128] In some embodiments, Form B is characterized by an XRPD pattern comprising eight or more of the signals (e.g., peaks) at approximately the positions shown in Table B. [0129] In some embodiments, Form B is characterized by an XRPD pattern comprising nine or more of the signals (e.g., peaks) at approximately the positions shown in Table B. [0130] In some embodiments, Form B is characterized by an XRPD pattern comprising 10 or more of the signals (e.g., peaks) at approximately the positions shown in Table B. [0131] In some embodiments, Form B is characterized by an XRPD pattern comprising signals (e.g., peaks) at approximately the positions shown in Table B.
Attorney Docket No. ASET-043/001WO 325190-2258 [0132] It is understood that the values in the table are approximate and subject to instrumental and experimental variations. Table B: Exemplary XRPD Signal List for Form B Angle d Value Net Intensity Gross Intensity Rel. Intensity 5.681 ° 15.54349 Å 67.9346 97.1635 6.7%

Attorney Docket No. ASET-043/001WO 325190-2258 39.701 ° 2.26848 Å 58.7695 87.8328 5.8% Diffe

[0133] In some embodiments, Form B is characterized by a DSC curve having an endothermic signal (e.g., peak) at 185 ± 20°C (e.g., 185 ± 10 °C (e.g., 185 ± 5 °C (e.g., 185 ± 4 °C (e.g., 185 ± 3 °C (e.g., 185 ± 2 °C (e.g., 185 ± 1 °C (e.g., 185 ± 0.5 °C))))))). [0134] In some embodiments, Form B is characterized by a DSC curve having an endothermic signal (e.g., peak) at about 185 °C. [0135] In some embodiments, Form B is characterized by a DSC curve having an endothermic signal (e.g., peak) associated with an enthalpy of about 19 ± 9 J/g (e.g., 19 ± 8 J/g (e.g., 19 ± 7 J/g (e.g., 19 ± 6 J/g (e.g., 19 ± 5 J/g (e.g., 19 ± 4 J/g (e.g., 19 ± 3 J/g (e.g., 19 ± 2 J/g))))))). [0136] In some embodiments, Form B is characterized by a DSC curve having an endothermic signal (e.g., peak) associated with an enthalpy of about 19 J/g. [0137] In some embodiments, Form B is characterized by a DSC curve substantially similar to that shown in FIG.5. Thermogravimetric Analysis (TGA) Characterizations [0138] In some embodiments, Form B shows a weight loss of from about 0.5% to about 15%, at a temperature range from about 35 ± 20 °C (e.g., 35 ± 10 °C (e.g., 35 ± 5 °C (e.g., 35 ± 4 °C (e.g., 35 ± 3 °C (e.g., 35 ± 2 °C (e.g., 35 ± 1 °C (e.g., 35 ± 0.5 °C))))))) to about 130 ± 20 °C (e.g., 130 ± 10 °C (e.g., 130 ± 5 °C (e.g., 130 ± 4 °C (e.g., 130 ± 3 °C (e.g., 130 ± 2 °C (e.g., 130 ± 1 °C (e.g., 130 ± 0.5 °C))))))), as measured by TGA. [0139] In some embodiments, Form B shows a weight loss of about 6.7%, at a temperature range from about 35 °C to about 135 °C, as measured by TGA. [0140] In some embodiments, Form B is characterized by a TGA profile substantially similar to that shown in FIG.6. Form C [0141] In some embodiments, Form C is a crystalline form. [0142] In some embodiments, Form C is a morphic form (e.g., crystalline form) of Compound No.1, the solvate thereof, the hydrate thereof, or the pharmaceutically acceptable salt thereof. [0143] In some embodiments, Form C is a morphic form (e.g., crystalline form) of Compound No.1 or the pharmaceutically acceptable salt thereof.
Attorney Docket No. ASET-043/001WO 325190-2258 [0144] In some embodiments, the Form C is a morphic form (e.g., crystalline form) of a pharmaceutically acceptable salt of Compound No.1. [0145] In some embodiments, Form C is a sulfate salt of Compound No.1. [0146] In some embodiments, Form B is a morphic form (e.g., crystalline form) of a sulfate salt of Compound No.1. [0147] In some embodiments, Form C is characterized by an X-ray diffraction (“XRPD”) pattern comprising signals (e.g., peaks) at 12.8±0.2, 15.3±0.2, and 17.9±0.2 °2θ (e.g., 12.8±0.1, 15.3±0.1, and 17.9±0.1 °2θ (e.g., 12.8, 15.3, and 17.9 °2θ)) using Cu Kα radiation. [0148] In some embodiments, the XRPD pattern of Form C further comprises at least one signal (e.g., peak) selected from 13.1±0.2, 16.5±0.2, and 28.2±0.2 °2θ (e.g., 13.1±0.1, 16.5±0.1, and 28.2±0.1 °2θ (e.g., 13.1, 16.5, and 28.2 °2θ)) using Cu Kα radiation. [0149] In some embodiments, the XRPD pattern of Form C further comprises at least two signals (e.g., peaks) selected from 13.1±0.2, 16.5±0.2, and 28.2±0.2 °2θ (e.g., 13.1±0.1, 16.5±0.1, and 28.2±0.1 °2θ (e.g., 13.1, 16.5, and 28.2 °2θ)) using Cu Kα radiation. [0150] In some embodiments, the XRPD pattern of Form C further comprises signals (e.g., peaks) at 13.1±0.2, 16.5±0.2, and 28.2±0.2 °2θ (e.g., 13.1±0.1, 16.5±0.1, and 28.2±0.1 °2θ (e.g., 13.1, 16.5, and 28.2 °2θ)) using Cu Kα radiation. [0151] In some embodiments, Form C is characterized by an X-ray diffraction (“XRPD”) pattern comprising at least three signals (e.g., peaks) selected from 12.8±0.2, 13.1±0.2, 15.3±0.2, 16.5±0.2, 17.9±0.2, and 28.2±0.2 °2θ (e.g., 12.8±0.1, 13.1±0.1, 15.3±0.1, 16.5±0.1, 17.9±0.1, and 28.2±0.1 °2θ (e.g., 12.8, 13.1, 15.3, 16.5, 17.9, and 28.2 °2θ)) using Cu Kα radiation. [0152] In some embodiments, the XRPD pattern of Form C comprises at least four signals (e.g., peaks) selected from 12.8±0.2, 13.1±0.2, 15.3±0.2, 16.5±0.2, 17.9±0.2, and 28.2±0.2 °2θ (e.g., 12.8±0.1, 13.1±0.1, 15.3±0.1, 16.5±0.1, 17.9±0.1, and 28.2±0.1 °2θ (e.g., 12.8, 13.1, 15.3, 16.5, 17.9, and 28.2 °2θ)) using Cu Kα radiation. [0153] In some embodiments, the XRPD pattern of Form C comprises at least five signals (e.g., peaks) selected from 12.8±0.2, 13.1±0.2, 15.3±0.2, 16.5±0.2, 17.9±0.2, and 28.2±0.2 °2θ (e.g., 12.8±0.1, 13.1±0.1, 15.3±0.1, 16.5±0.1, 17.9±0.1, and 28.2±0.1 °2θ (e.g., 12.8, 13.1, 15.3, 16.5, 17.9, and 28.2 °2θ)) using Cu Kα radiation. [0154] In some embodiments, the XRPD pattern of Form C comprises signals (e.g., peaks) at 12.8±0.2, 13.1±0.2, 15.3±0.2, 16.5±0.2, 17.9±0.2, and 28.2±0.2 °2θ (e.g., 12.8±0.1, 13.1±0.1, 15.3±0.1, 16.5±0.1, 17.9±0.1, and 28.2±0.1 °2θ (e.g., 12.8, 13.1, 15.3, 16.5, 17.9, and 28.2 °2θ)) using Cu Kα radiation.
Attorney Docket No. ASET-043/001WO 325190-2258 [0155] In some embodiments, the XRPD pattern of Form C further comprises at least one signal (e.g., peak) selected from 5.1±0.2, 11.1±0.2, 22.1±0.2, and 23.2±0.2 °2θ (e.g., 5.1±0.1, 11.1±0.1, 22.1±0.1, and 23.2±0.1 °2θ (e.g., 5.1, 11.1, 22.1, and 23.2 °2θ)) using Cu Kα radiation. [0156] In some embodiments, the XRPD pattern of Form C further comprises at least two signals (e.g., peaks) selected from 5.1±0.2, 11.1±0.2, 22.1±0.2, and 23.2±0.2 °2θ (e.g., 5.1±0.1, 11.1±0.1, 22.1±0.1, and 23.2±0.1 °2θ (e.g., 5.1, 11.1, 22.1, and 23.2 °2θ)) using Cu Kα radiation. [0157] In some embodiments, the XRPD pattern of Form C further comprises at least three signals (e.g., peaks) selected from 5.1±0.2, 11.1±0.2, 22.1±0.2, and 23.2±0.2 °2θ (e.g., 5.1±0.1, 11.1±0.1, 22.1±0.1, and 23.2±0.1 °2θ (e.g., 5.1, 11.1, 22.1, and 23.2 °2θ)) using Cu Kα radiation. [0158] In some embodiments, the XRPD pattern of Form C further comprises signals (e.g., peaks) at 5.1±0.2, 11.1±0.2, 12.8±0.2, 13.1±0.2, 15.3±0.2, 16.5±0.2, 17.9±0.2, 22.1±0.2, 23.2±0.2, and 28.2±0.2 °2θ (e.g. 5.1±0.1, 11.1±0.1, 12.8±0.1, 13.1±0.1, 15.3±0.1, 16.5±0.1, 17.9±0.1, 22.1±0.1, 23.2±0.1, and 28.2±0.1 °2θ (e.g. 5.1, 11.1, 12.8, 13.1, 15.3, 16.5, 17.9, 22.1, 23.2, and 28.2 °2θ)) using Cu Kα radiation. [0159] In some embodiments, Form C is characterized by an XRPD pattern substantially similar to that shown in FIG.7. [0160] In some embodiments, Form C is characterized by an XRPD pattern comprising one or more signals (e.g., peaks) at approximately the positions shown in Table C. [0161] In some embodiments, Form C is characterized by an XRPD pattern comprising two or more of the signals (e.g., peaks) at approximately the positions shown in Table C. [0162] In some embodiments, Form C is characterized by an XRPD pattern comprising three or more of the signals (e.g., peaks) at approximately the positions shown in Table C. [0163] In some embodiments, Form C is characterized by an XRPD pattern comprising four or more of the signals (e.g., peaks) at approximately the positions shown in Table C. [0164] In some embodiments, Form C is characterized by an XRPD pattern comprising five or more of the signals (e.g., peaks) at approximately the positions shown in Table C. [0165] In some embodiments, Form C is characterized by an XRPD pattern comprising six or more of the signals (e.g., peaks) at approximately the positions shown in Table C. [0166] In some embodiments, Form C is characterized by an XRPD pattern comprising seven or more of the signals (e.g., peaks) at approximately the positions shown in Table C.
Attorney Docket No. ASET-043/001WO 325190-2258 [0167] In some embodiments, Form C is characterized by an XRPD pattern comprising eight or more of the signals (e.g., peaks) at approximately the positions shown in Table C. [0168] In some embodiments, Form C is characterized by an XRPD pattern comprising nine or more of the signals (e.g., peaks) at approximately the positions shown in Table C. [0169] In some embodiments, Form C is characterized by an XRPD pattern comprising 10 or more of the signals (e.g., peaks) at approximately the positions shown in Table C. [0170] In some embodiments, Form C is characterized by an XRPD pattern comprising signals (e.g., peaks) at approximately the positions shown in Table C. [0171] It is understood that the values in the table are approximate and subject to instrumental and experimental variations. Table C: Exemplary XRPD Signal List for Form C Angle d Value Net Intensity Gross Intensity Rel. Intensity 3327 ° 2653377 Å 176568 436160 16%

Attorney Docket No. ASET-043/001WO 325190-2258 24.692 ° 3.60272 Å 36.0633 108.774 3.4% 25.611 ° 3.47545 Å 28.7591 97.0884 2.7% Diffe
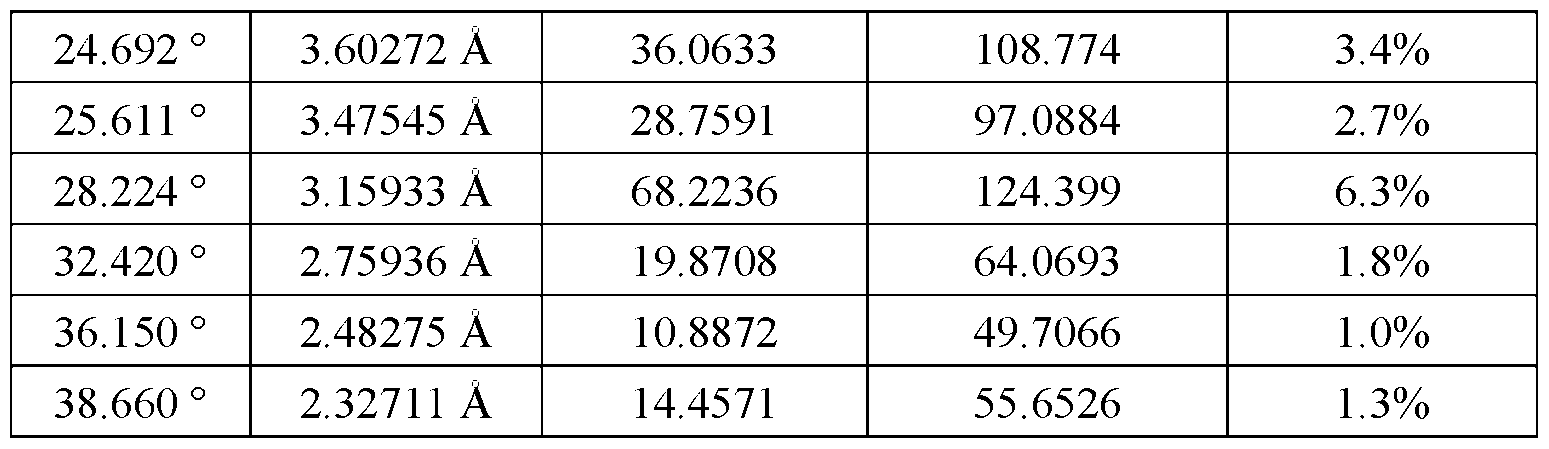
[0172] In some embodiments, Form C is characterized by a DSC curve having an endothermic signal (e.g., peak) at 246 ± 20°C (e.g., 246 ± 10 °C (e.g., 246 ± 5 °C (e.g., 246 ± 4 °C (e.g., 246 ± 3 °C (e.g., 246 ± 2 °C (e.g., 246 ± 1 °C (e.g., 246 ± 0.5 °C))))))). [0173] In some embodiments, Form C is characterized by a DSC curve having an endothermic signal (e.g., peak) at about 246 °C. [0174] In some embodiments, Form C is characterized by a DSC curve having an endothermic signal (e.g., peak) associated with an enthalpy of about 27 ± 9 J/g (e.g., 27 ± 8 J/g (e.g., 27 ± 7 J/g (e.g., 27 ± 6 J/g (e.g., 27 ± 5 J/g (e.g., 27 ± 4 J/g (e.g., 27 ± 3 J/g (e.g., 27 ± 2 J/g))))))). [0175] In some embodiments, Form C is characterized by a DSC curve having an endothermic signal (e.g., peak) associated with an enthalpy of about 27 J/g. [0176] In some embodiments, Form C is characterized by a DSC curve having an endothermic signal (e.g., peak) at 261 ± 20°C (e.g., 261 ± 10 °C (e.g., 261 ± 5 °C (e.g., 261 ± 4 °C (e.g., 261 ± 3 °C (e.g., 261 ± 2 °C (e.g., 261 ± 1 °C (e.g., 261 ± 0.5 °C))))))). [0177] In some embodiments, Form C is characterized by a DSC curve having an endothermic signal (e.g., peak) at about 261 °C. [0178] In some embodiments, Form C is characterized by a DSC curve having an endothermic signal (e.g., peak) associated with an enthalpy of about 8 ± 4 J/g (e.g., 8 ± 3 J/g (e.g., 8 ± 2 J/g (e.g., 8 ± 1 J/g (e.g., 8 ± 0.5 J/g)))). [0179] In some embodiments, Form C is characterized by a DSC curve having an endothermic signal (e.g., peak) associated with an enthalpy of about 8 J/g. [0180] In some embodiments, Form C is characterized by a DSC curve substantially similar to that shown in FIG.8. Thermogravimetric Analysis (TGA) Characterizations [0181] In some embodiments, Form C shows a weight loss of from about 0.5% to about 3%, at a temperature range from about 35 ± 20 °C (e.g., 35 ± 10 °C (e.g., 35 ± 5 °C (e.g., 35 ± 4 °C
Attorney Docket No. ASET-043/001WO 325190-2258 (e.g., 35 ± 3 °C (e.g., 35 ± 2 °C (e.g., 35 ± 1 °C (e.g., 35 ± 0.5 °C))))))) to about 130 ± 20 °C (e.g., 130 ± 10 °C (e.g., 130 ± 5 °C (e.g., 130 ± 4 °C (e.g., 130 ± 3 °C (e.g., 130 ± 2 °C (e.g., 130 ± 1 °C (e.g., 130 ± 0.5 °C))))))), as measured by TGA. [0182] In some embodiments, Form C shows a weight loss of about 1.6%, at a temperature range from about 35 °C to about 135 °C, as measured by TGA. [0183] In some embodiments, Form C is characterized by a TGA profile substantially similar to that shown in FIG.9. Form D [0184] In some embodiments, Form D is a crystalline form. [0185] In some embodiments, Form D of Compound No. 1, the solvate thereof, the hydrate thereof, or the pharmaceutically acceptable salt thereof. [0186] In some embodiments, Form D is a morphic form (e.g., crystalline form) of Compound No.1 or the pharmaceutically acceptable salt thereof. [0187] In some embodiments, Form D is a morphic form (e.g., crystalline form) of a pharmaceutically acceptable salt of Compound No.1. [0188] In some embodiments, Form D is a phosphate salt. [0189] In some embodiments, Form D is a morphic form (e.g., crystalline form) of a phosphate salt of Compound No.1. [0190] In some embodiments, Form D is characterized by an X-ray diffraction (“XRPD”) pattern comprising signals (e.g., peaks) at 10.6±0.2, 15.9±0.2, and 18.6±0.2 °2θ (e.g., 10.6±0.1, 15.9±0.1, and 18.6±0.1 °2θ (e.g., 10.6, 15.9, and 18.6 °2θ)) using Cu Kα radiation. [0191] In some embodiments, the XRPD pattern of Form D further comprises at least one signal (e.g., peak) selected from 5.3±0.2, 24.0±0.2, and 26.7±0.2 °2θ (e.g., 5.3±0.1, 24.0±0.1, and 26.7±0.1 °2θ (e.g., 5.3, 24.0, and 26.7 °2θ)) using Cu Kα radiation. [0192] In some embodiments, the XRPD pattern of Form D further comprises at least two signals (e.g., peaks) selected from 5.3±0.2, 24.0±0.2, and 26.7±0.2 °2θ (e.g., 5.3±0.1, 24.0±0.1, and 26.7±0.1 °2θ (e.g., 5.3, 24.0, and 26.7 °2θ)) using Cu Kα radiation. [0193] In some embodiments, the XRPD pattern of Form D further comprises signals (e.g., peaks) at 5.3±0.2, 24.0±0.2, and 26.7±0.2 °2θ (e.g., 5.3±0.1, 24.0±0.1, and 26.7±0.1 °2θ (e.g., 5.3, 24.0, and 26.7 °2θ)) using Cu Kα radiation. [0194] In some embodiments, Form D is characterized by an X-ray diffraction (“XRPD”) pattern comprising at least three signals (e.g., peaks) selected from 5.3±0.2, 10.6±0.2, 15.9±0.2,
Attorney Docket No. ASET-043/001WO 325190-2258 18.6±0.2, 24.0±0.2, and 26.7±0.2 °2θ (e.g., 5.3±0.1, 10.6±0.1, 15.9±0.1, 18.6±0.1, 24.0±0.1, and 26.7±0.1 °2θ (e.g., 5.3, 10.6, 15.9, 18.6, 24.0, and 26.7 °2θ)) using Cu Kα radiation. [0195] In some embodiments, the XRPD pattern of Form D comprises at least four signals (e.g., peaks) selected from 5.3±0.2, 10.6±0.2, 15.9±0.2, 18.6±0.2, 24.0±0.2, and 26.7±0.2 °2θ (e.g., 5.3±0.1, 10.6±0.1, 15.9±0.1, 18.6±0.1, 24.0±0.1, and 26.7±0.1 °2θ (e.g., 5.3, 10.6, 15.9, 18.6, 24.0, and 26.7 °2θ)) using Cu Kα radiation. [0196] In some embodiments, the XRPD pattern of Form D comprises at least five signals (e.g., peaks) selected from 5.3±0.2, 10.6±0.2, 15.9±0.2, 18.6±0.2, 24.0±0.2, and 26.7±0.2 °2θ (e.g., 5.3±0.1, 10.6±0.1, 15.9±0.1, 18.6±0.1, 24.0±0.1, and 26.7±0.1 °2θ (e.g., 5.3, 10.6, 15.9, 18.6, 24.0, and 26.7 °2θ)) using Cu Kα radiation. [0197] In some embodiments, the XRPD pattern of Form D comprises signals (e.g., peaks) at 5.3±0.2, 10.6±0.2, 15.9±0.2, 18.6±0.2, 24.0±0.2, and 26.7±0.2 °2θ (e.g., 5.3±0.1, 10.6±0.1, 15.9±0.1, 18.6±0.1, 24.0±0.1, and 26.7±0.1 °2θ (e.g., 5.3, 10.6, 15.9, 18.6, 24.0, and 26.7 °2θ)) using Cu Kα radiation. [0198] In some embodiments, the XRPD pattern of Form D further comprises at least one signal (e.g., peak) selected from 12.6±0.2, 14.9±0.2, 20.9±0.2, and 32.2±0.2 °2θ (e.g., 12.6±0.1, 14.9±0.1, 20.9±0.1, and 32.2±0.1 °2θ (e.g., 12.6, 14.9, 20.9, and 32.2 °2θ)) using Cu Kα radiation. [0199] In some embodiments, the XRPD pattern of Form D further comprises at least two signals (e.g., peaks) selected from 12.6±0.2, 14.9±0.2, 20.9±0.2, and 32.2±0.2 °2θ (e.g., 12.6±0.1, 14.9±0.1, 20.9±0.1, and 32.2±0.1 °2θ (e.g., 12.6, 14.9, 20.9, and 32.2 °2θ)) using Cu Kα radiation. [0200] In some embodiments, the XRPD pattern of Form D further comprises at least three signals (e.g., peaks) selected from 12.6±0.2, 14.9±0.2, 20.9±0.2, and 32.2±0.2 °2θ (e.g., 12.6±0.1, 14.9±0.1, 20.9±0.1, and 32.2±0.1 °2θ (e.g., 12.6, 14.9, 20.9, and 32.2 °2θ)) using Cu Kα radiation. [0201] In some embodiments, the XRPD pattern of Form D further comprises signals (e.g., peaks) at 5.3±0.2, 10.6±0.2, 12.6±0.2, 14.9±0.2, 15.9±0.2, 18.6±0.2, 20.9±0.2, 24.0±0.2, 26.7±0.2, and 32.2±0.2 °2θ (e.g. 5.3±0.1, 10.6±0.1, 12.6±0.1, 14.9±0.1, 15.9±0.1, 18.6±0.1, 20.9±0.1, 24.0±0.1, 26.7±0.1, and 32.2±0.1 °2θ (e.g. 5.3, 10.6, 12.6, 14.9, 15.9, 18.6, 20.9, 24.0, 26.7, and 32.2 °2θ)) using Cu Kα radiation. [0202] In some embodiments, Form D is characterized by an XRPD pattern substantially similar to that shown in FIG.10.
Attorney Docket No. ASET-043/001WO 325190-2258 [0203] In some embodiments, Form D is characterized by an XRPD pattern comprising signals (e.g., peaks) at approximately the positions shown in the table below. It is understood that the values in the table are approximate and subject to instrumental and experimental variations. Table D: Exemplary XRPD Signal List for Form D Angle d Value Net Intensity Gross Intensity Rel. Intensity 5.314 ° 16.61590 Å 43.3652 81.9473 10.4% Di

fferential Scanning Calorimeter (DSC) Characterizations [0204] In some embodiments, Form D is characterized by a DSC curve having an endothermic signal (e.g., peak) at 251 ± 20°C (e.g., 251 ± 10 °C (e.g., 251 ± 5 °C (e.g., 251 ± 4 °C (e.g., 251 ± 3 °C (e.g., 251 ± 2 °C (e.g., 251 ± 1 °C (e.g., 251 ± 0.5 °C))))))). [0205] In some embodiments, Form D is characterized by a DSC curve having an endothermic signal (e.g., peak) at about 251 °C. [0206] In some embodiments, Form D is characterized by a DSC curve having an endothermic signal (e.g., peak) associated with an enthalpy of about 119 ± 9 J/g (e.g., 119 ± 8 J/g (e.g., 119 ± 7 J/g (e.g., 119 ± 6 J/g (e.g., 119 ± 5 J/g (e.g., 119 ± 4 J/g (e.g., 119 ± 3 J/g (e.g., 119 ± 2 J/g))))))). [0207] In some embodiments, Form D is characterized by a DSC curve having an endothermic signal (e.g., peak) associated with an enthalpy of about 119 J/g. [0208] In some embodiments, Form D is characterized by a DSC curve substantially similar to that shown in FIG.11. Thermogravimetric Analysis (TGA) Characterizations
Attorney Docket No. ASET-043/001WO 325190-2258 [0209] In some embodiments, Form D shows a weight loss of from about 0.5% to about 4%, at a temperature range from about 35 ± 20 °C (e.g., 35 ± 10 °C (e.g., 35 ± 5 °C (e.g., 35 ± 4 °C (e.g., 35 ± 3 °C (e.g., 35 ± 2 °C (e.g., 35 ± 1 °C (e.g., 35 ± 0.5 °C))))))) to about 130 ± 20 °C (e.g., 130 ± 10 °C (e.g., 130 ± 5 °C (e.g., 130 ± 4 °C (e.g., 130 ± 3 °C (e.g., 130 ± 2 °C (e.g., 130 ± 1 °C (e.g., 130 ± 0.5 °C))))))), as measured by TGA. [0210] In some embodiments, Form D shows a weight loss of about 1.9%, at a temperature range from about 35 °C to about 130 °C, as measured by TGA. [0211] In some embodiments, Form D is characterized by a TGA profile substantially similar to that shown in FIG.12. Amorphous Forms, Combinations Thereof, and Compositions Thereof [0212] In some aspects, the present disclosure provides an amorphous form of Compound No. 1, a salt thereof, a solvate thereof, a hydrate thereof, or a pharmaceutically acceptable salt thereof. [0213] In some aspects, the present disclosure provides an amorphous form of Compound No. 1. [0214] In some aspects, the present disclosure provides a combination comprising: an amorphous form of Compound No. 1, a salt thereof, a solvate thereof, a hydrate thereof, or a pharmaceutically acceptable salt thereof; and a polymer. [0215] In some aspects, the present disclosure provides a composition (e.g., a pharmaceutical composition) comprising: an amorphous form of Compound No. 1, a salt thereof, a solvate thereof, a hydrate thereof, or a pharmaceutically acceptable salt thereof; and a polymer. [0216] In some embodiments, the polymer is polyvinylpyrrolidone (PVP). [0217] In some embodiments, the polymer comprises a polyvinyl caprolactam-polyvinyl acetate-polyethylene glycol graft co-polymer. In some embodiments, the polymer is PVP K30, PVP VA64, Soluplus
®, HPMC E3, HPMC ASLG, HPMC ASMG, HPMC ASHG, HPC-SSL, Eudragit
® E100, or Eudragit
® L100. [0218] In some embodiments, the polymer is sold as Soluplus
®. [0219] In some embodiments, the polymer is hydroxypropyl methylcellulose (HPMC). [0220] In some embodiments, the polymer is hydroxypropyl cellulose (HPC). [0221] In some embodiments, the polymer comprises a polymethacrylate-based copolymer.
Attorney Docket No. ASET-043/001WO 325190-2258 [0222] In some embodiments, the polymer is sold as Eudragit
®. [0223] In some embodiments, the polymer is PVP K30. [0224] In some embodiments, the polymer is PVP VA64. [0225] In some embodiments, the polymer is HPMC E3. [0226] In some embodiments, the polymer is HPMC ASLG. [0227] In some embodiments, the polymer is HPMC ASMG. [0228] In some embodiments, the polymer is HPMC ASHG. [0229] In some embodiments, the polymer is HPC-SSL. [0230] In some embodiments, the polymer is Eudragit
® E100. [0231] In some embodiments, the polymer is Eudragit
® L100. [0232] In some embodiments, the amorphous form and the polymer are present in the combination (e.g., composition) at a ratio of about 1:0.5 (w/w), about 1:1 (w/w), about 1:2 (w/w), about 1:3 (w/w), about 1:4 (w/w), or about 1:5 (w/w). [0233] In some embodiments, the amorphous form and the polymer are present in the combination (e.g., composition) at a ratio of about 1:1 (w/w). [0234] In some embodiments, the amorphous form and the polymer are present in the combination (e.g., composition) at a ratio of about 1:4 (w/w). [0235] In some embodiments, the composition is a solid dispersion. [0236] In some aspects, the composition is a solid dispersion prepared by fast evaporation. [0237] In some aspects, the composition is a solid dispersion prepared by spray drying. Thermogravimetric Analysis (TGA) Characterizations [0238] In some embodiments, the combination (e.g., the composition comprising the amorphous form of Compound No.1 and HPMC E3) shows a weight loss of from about 0.5% to about 10%, at a temperature range from about 35 ± 20 °C (e.g., 35 ± 10 °C (e.g., 35 ± 5 °C (e.g., 35 ± 4 °C (e.g., 35 ± 3 °C (e.g., 35 ± 2 °C (e.g., 35 ± 1 °C (e.g., 35 ± 0.5 °C))))))) to about 100 ± 20 °C (e.g., 100 ± 10 °C (e.g., 100 ± 5 °C (e.g., 100 ± 4 °C (e.g., 100 ± 3 °C (e.g., 100 ± 2 °C (e.g., 100 ± 1 °C (e.g., 100 ± 0.5 °C))))))), as measured by TGA. [0239] In some embodiments, the combination (e.g., the composition comprising the amorphous form of Compound No.1 and HPMC E3) shows a weight loss of about 4.5%, at a temperature range from about 35 °C to about 100 °C, as measured by TGA. [0240] In some embodiments, the combination (e.g., the composition comprising the amorphous form of Compound No. 1 and HPMC E3) is characterized by a TGA profile substantially similar to that shown in FIG.13.
Attorney Docket No. ASET-043/001WO 325190-2258 [0241] In some embodiments, the combination (e.g., the composition comprising the amorphous form of Compound No. 1 and HPMC ASMG) shows a weight loss of from about 0.5% to about 5%, at a temperature range from about 35 ± 20 °C (e.g., 35 ± 10 °C (e.g., 35 ± 5 °C (e.g., 35 ± 4 °C (e.g., 35 ± 3 °C (e.g., 35 ± 2 °C (e.g., 35 ± 1 °C (e.g., 35 ± 0.5 °C))))))) to about 100 ± 20 °C (e.g., 100 ± 10 °C (e.g., 100 ± 5 °C (e.g., 100 ± 4 °C (e.g., 100 ± 3 °C (e.g., 100 ± 2 °C (e.g., 100 ± 1 °C (e.g., 100 ± 0.5 °C))))))), as measured by TGA. [0242] In some embodiments, the combination (e.g., the composition comprising the amorphous form of Compound No.1 and HPMC ASMG) shows a weight loss of about 2.6%, at a temperature range from about 35 °C to about 100 °C, as measured by TGA. [0243] In some embodiments, the combination (e.g., the composition comprising the amorphous form of Compound No. 1 and HPMC ASMG) is characterized by a TGA profile substantially similar to that shown in FIG.14. [0244] In some embodiments, the combination (e.g., the composition comprising the amorphous form of Compound No.1 and Eudragit
® E100) shows a weight loss of from about 0.05% to about 2%, at a temperature range from about 35 ± 20 °C (e.g., 35 ± 10 °C (e.g., 35 ± 5 °C (e.g., 35 ± 4 °C (e.g., 35 ± 3 °C (e.g., 35 ± 2 °C (e.g., 35 ± 1 °C (e.g., 35 ± 0.5 °C))))))) to about 100 ± 20 °C (e.g., 100 ± 10 °C (e.g., 100 ± 5 °C (e.g., 100 ± 4 °C (e.g., 100 ± 3 °C (e.g., 100 ± 2 °C (e.g., 100 ± 1 °C (e.g., 100 ± 0.5 °C))))))), as measured by TGA. [0245] In some embodiments, the combination (e.g., the composition comprising the amorphous form of Compound No.1 and Eudragit
® E100) shows a weight loss of about 0.8%, at a temperature range from about 35 °C to about 100 °C, as measured by TGA. [0246] In some embodiments, the combination (e.g., the composition comprising the amorphous form of Compound No. 1 and Eudragit
® E100) is characterized by a TGA profile substantially similar to that shown in FIG.15. Modulated Differential Scanning Calorimetry (mDSC) Characterizations [0247] In some embodiments, the combination (e.g., the composition comprising the amorphous form of Compound No. 1 and PVP K30) is characterized by a glass transition temperature (Tg) as measured by mDSC at 161 ± 20°C (e.g., 161 ± 10 °C (e.g., 161 ± 5 °C (e.g., 161 ± 4 °C (e.g., 161 ± 3 °C (e.g., 161 ± 2 °C (e.g., 161 ± 1 °C (e.g., 161 ± 0.5 °C))))))). [0248] In some embodiments, the combination (e.g., the composition comprising the amorphous form of Compound No. 1 and PVP K30) is characterized by a glass transition temperature (Tg) as measured by mDSC at about 161 °C.
Attorney Docket No. ASET-043/001WO 325190-2258 [0249] In some embodiments, the combination (e.g., the composition comprising the amorphous form of Compound No. 1 and PVP K30) is characterized by an mDSC profile substantially similar to that shown in FIG.16. [0250] In some embodiments, the combination (e.g., the composition comprising the amorphous form of Compound No. 1 and PVP VA64) is characterized by a glass transition temperature (Tg) as measured by mDSC at 111 ± 20°C (e.g., 111 ± 10 °C (e.g., 111 ± 5 °C (e.g., 111 ± 4 °C (e.g., 111 ± 3 °C (e.g., 111 ± 2 °C (e.g., 111 ± 1 °C (e.g., 111 ± 0.5 °C))))))). [0251] In some embodiments, the combination (e.g., the composition comprising the amorphous form of Compound No. 1 and PVP VA64) is characterized by a glass transition temperature (Tg) as measured by mDSC at about 111 °C. [0252] In some embodiments, the combination (e.g., the composition comprising the amorphous form of Compound No. 1 and PVP VA64) is characterized by an mDSC profile substantially similar to that shown in FIG.17. [0253] In some embodiments, the combination (e.g., the composition comprising the amorphous form of Compound No. 1 and Soluplus
®) is characterized by a glass transition temperature (Tg) as measured by mDSC at 145 ± 20°C (e.g., 145 ± 10 °C (e.g., 145 ± 5 °C (e.g., 145 ± 4 °C (e.g., 145 ± 3 °C (e.g., 145 ± 2 °C (e.g., 145 ± 1 °C (e.g., 145 ± 0.5 °C))))))). [0254] In some embodiments, the combination (e.g., the composition comprising the amorphous form of Compound No. 1 and Soluplus
®) is characterized by a glass transition temperature (Tg) as measured by mDSC at about 145 °C. [0255] In some embodiments, the combination (e.g., the composition comprising the amorphous form of Compound No. 1 and Soluplus
®) is characterized by an mDSC profile substantially similar to that shown in FIG.18. [0256] In some embodiments, the combination (e.g., the composition comprising the amorphous form of Compound No. 1 and HPMC E3) is characterized by a glass transition temperature (Tg) as measured by mDSC at 115 ± 20°C (e.g., 115 ± 10 °C (e.g., 115 ± 5 °C (e.g., 115 ± 4 °C (e.g., 115 ± 3 °C (e.g., 115 ± 2 °C (e.g., 115 ± 1 °C (e.g., 115 ± 0.5 °C))))))). [0257] In some embodiments, the combination (e.g., the composition comprising the amorphous form of Compound No. 1 and HPMC E3) is characterized by a glass transition temperature (Tg) as measured by mDSC at about 115 °C. [0258] In some embodiments, the combination (e.g., the composition comprising the amorphous form of Compound No. 1 and HPMC E3) is characterized by an mDSC profile substantially similar to that shown in FIG.19.
Attorney Docket No. ASET-043/001WO 325190-2258 [0259] In some embodiments, the combination (e.g., the composition comprising the amorphous form of Compound No.1 and HPMC ASLG) is characterized by a glass transition temperature (Tg) as measured by mDSC at 93 ± 20°C (e.g., 93 ± 10 °C (e.g., 93 ± 5 °C (e.g., 93 ± 4 °C (e.g., 93 ± 3 °C (e.g., 93 ± 2 °C (e.g., 93 ± 1 °C (e.g., 93 ± 0.5 °C))))))). [0260] In some embodiments, the combination (e.g., the composition comprising the amorphous form of Compound No.1 and HPMC ASLG) is characterized by a glass transition temperature (Tg) as measured by mDSC at about 93 °C. [0261] In some embodiments, the combination (e.g., the composition comprising the amorphous form of Compound No.1 and HPMC ASLG) is characterized by an mDSC profile substantially similar to that shown in FIG.20. [0262] In some embodiments, the combination (e.g., the composition comprising the amorphous form of Compound No.1 and HPMC ASMG) is characterized by a glass transition temperature (Tg) as measured by mDSC at 95 ± 20°C (e.g., 95 ± 10 °C (e.g., 95 ± 5 °C (e.g., 95 ± 4 °C (e.g., 95 ± 3 °C (e.g., 95 ± 2 °C (e.g., 95 ± 1 °C (e.g., 95 ± 0.5 °C))))))). [0263] In some embodiments, the combination (e.g., the composition comprising the amorphous form of Compound No.1 and HPMC ASMG) is characterized by a glass transition temperature (Tg) as measured by mDSC at about 95 °C. [0264] In some embodiments, the combination (e.g., the composition comprising the amorphous form of Compound No.1 and HPMC ASMG) is characterized by an mDSC profile substantially similar to that shown in FIG.21. [0265] In some embodiments, the combination (e.g., the composition comprising the amorphous form of Compound No.1 and HPMC ASHG) is characterized by a glass transition temperature (Tg) as measured by mDSC at 97 ± 20°C (e.g., 97 ± 10 °C (e.g., 97 ± 5 °C (e.g., 97 ± 4 °C (e.g., 97 ± 3 °C (e.g., 97 ± 2 °C (e.g., 97 ± 1 °C (e.g., 97 ± 0.5 °C))))))). [0266] In some embodiments, the combination (e.g., the composition comprising the amorphous form of Compound No.1 and HPMC ASHG) is characterized by a glass transition temperature (Tg) as measured by mDSC at about 97 °C. [0267] In some embodiments, the combination (e.g., the composition comprising the amorphous form of Compound No.1 and HPMC ASHG) is characterized by an mDSC profile substantially similar to that shown in FIG.22. [0268] In some embodiments, the combination (e.g., the composition comprising the amorphous form of Compound No. 1 and HPC-SSL) is characterized by a glass transition temperature (Tg) as measured by mDSC at 89 ± 20°C (e.g., 89 ± 10 °C (e.g., 89 ± 5 °C (e.g., 89 ± 4 °C (e.g., 89 ± 3 °C (e.g., 89 ± 2 °C (e.g., 89 ± 1 °C (e.g., 89 ± 0.5 °C))))))).
Attorney Docket No. ASET-043/001WO 325190-2258 [0269] In some embodiments, the combination (e.g., the composition comprising the amorphous form of Compound No. 1 and HPC-SSL) is characterized by a glass transition temperature (Tg) as measured by mDSC at about 89 °C. [0270] In some embodiments, the combination (e.g., the composition comprising the amorphous form of Compound No. 1 and HPC-SSL) is characterized by an mDSC profile substantially similar to that shown in FIG.23. [0271] In some embodiments, the combination (e.g., the composition comprising the amorphous form of Compound No.1 and Eudragit
® E100) is characterized by a glass transition temperature (Tg) as measured by mDSC at 93 ± 20°C (e.g., 93 ± 10 °C (e.g., 93 ± 5 °C (e.g., 93 ± 4 °C (e.g., 93 ± 3 °C (e.g., 93 ± 2 °C (e.g., 93 ± 1 °C (e.g., 93 ± 0.5 °C))))))). [0272] In some embodiments, the combination (e.g., the composition comprising the amorphous form of Compound No.1 and Eudragit
® E100) is characterized by a glass transition temperature (Tg) as measured by mDSC at about 93 °C. [0273] In some embodiments, the combination (e.g., the composition comprising the amorphous form of Compound No.1 and Eudragit
® E100) is characterized by an mDSC profile substantially similar to that shown in FIG.24. [0274] In some embodiments, the combination (e.g., the composition comprising the amorphous form of Compound No.1 and Eudragit
® L100) is characterized by a glass transition temperature (Tg) as measured by mDSC at 117 ± 20°C (e.g., 117 ± 10 °C (e.g., 117 ± 5 °C (e.g., 117 ± 4 °C (e.g., 117 ± 3 °C (e.g., 117 ± 2 °C (e.g., 117 ± 1 °C (e.g., 117 ± 0.5 °C))))))). [0275] In some embodiments, the combination (e.g., the composition comprising the amorphous form of Compound No.1 and Eudragit
® L100) is characterized by a glass transition temperature (Tg) as measured by mDSC at about 117 °C. [0276] In some embodiments, the combination (e.g., the composition comprising the amorphous form of Compound No.1 and Eudragit
® L100) is characterized by an mDSC profile substantially similar to that shown in FIG.25. Properties [0277] In some embodiments, the purity (e.g., chemical purity) of the combination (e.g., the composition comprising the amorphous form of Compound No.1 and HPMC E3) is more than about 95%, more than about 96%, more than about 97%, more than about 98%, or more than about 99%, after being placed at 40°C and 75% RH for about a week. [0278] In some embodiments, the impurity (e.g., chemical impurity) present the combination (e.g., the composition comprising the amorphous form of Compound No.1 and HPMC E3) is
Attorney Docket No. ASET-043/001WO 325190-2258 less than about 5%, less than about 4%, less than about 3%, less than about 2%, less than about 1%, less than about 0.5%, less than about 0.4%, less than about 0.3%, less than about 0.2%, or less than about 0.1%, after being placed at 40°C and 75% RH for about a week. [0279] In some embodiments, the purity (e.g., chemical purity) of the combination (e.g., the composition comprising the amorphous form of Compound No.1 and HPMC ASMG) is more than about 95%, more than about 96%, more than about 97%, more than about 98%, or more than about 99%, after being placed at 40°C and 75% RH for about a week. [0280] In some embodiments, the impurity (e.g., chemical impurity) present the combination (e.g., the composition comprising the amorphous form of Compound No.1 and HPMC ASMG) is less than about 5%, less than about 4%, less than about 3%, less than about 2%, less than about 1%, less than about 0.5%, less than about 0.4%, less than about 0.3%, less than about 0.2%, or less than about 0.1%, after being placed at 40°C and 75% RH for about a week. [0281] In some embodiments, the purity (e.g., chemical purity) of the combination (e.g., the composition comprising the amorphous form of Compound No.1 and Eudragit
® E100) is more than about 95%, more than about 96%, more than about 97%, more than about 98%, or more than about 99%, after being placed at 40°C and 75% RH for about a week. [0282] In some embodiments, the impurity (e.g., chemical impurity) present the combination (e.g., the composition comprising the amorphous form of Compound No. 1 and Eudragit
® E100) is less than about 5%, less than about 4%, less than about 3%, less than about 2%, less than about 1%, less than about 0.5%, less than about 0.4%, less than about 0.3%, less than about 0.2%, or less than about 0.1%, after being placed at 40°C and 75% RH for about a week. Methods of Preparing the Crystalline Forms [0283] In some aspects, the present disclosure features a method of preparing a form (e.g., a crystalline form) of Compound No.1, a solvate thereof, a hydrate thereof, or a pharmaceutically acceptable salt thereof. [0284] In some aspects, the present disclosure provides a method of preparing a form (e.g., a crystalline form) of Compound No.1, a solvate thereof, a hydrate thereof, or a pharmaceutically acceptable salt thereof, comprising one or more steps as described herein. [0285] In some aspects, the present disclosure provides a compound obtainable by, or obtained by, or directly obtained by a method for preparing a form (e.g., a crystalline form) of Compound No.1, a solvate thereof, a hydrate thereof, or a pharmaceutically acceptable salt thereof. [0286] The form of Compound No. 1, a solvate thereof, a hydrate thereof, or a pharmaceutically acceptable salt thereof can be prepared by any suitable technique known in
Attorney Docket No. ASET-043/001WO 325190-2258 the art, e.g., as described in PCT Application No. PCT/US2022/015353 (incorporated herein by reference). Particular processes for the preparation of these compounds are described further in the accompanying examples. Preparation of Form A [0287] In some embodiments, the method comprises: (i) preparing a mixture comprising Compound No.1 and a solvent, and (ii) removing the solvent from the mixture. [0288] In some embodiments, the solvent is DMSO. [0289] In some embodiments, the solvent is THF and H2O (e.g., at a ratio of about 4:1 v/v). [0290] In some embodiments, the method further comprises equilibrating the mixture, e.g., at about 25 °C or at about 50 °C. [0291] In some embodiments, the solvent is removed from the mixture at about 50±20 °C, about 50±15 °C, about 50±10 °C, about 50±9 °C, about 50±8 °C, about 50±7 °C, about 50±6 °C, about 50±5 °C, about 50±4 °C, about 50±3 °C, about 50±2 °C, or about 50±1 °C (e.g., about 50 °C). Preparation of Forms B, C, and D [0292] In some embodiments, the method comprises: (i) preparing a mixture comprising Compound No.1, an acid, and a solvent, and (ii) removing the solvent from the mixture. [0293] In some embodiments, the acid is hydrochloric acid, sulfuric acid, or phosphoric acid. [0294] In some embodiments, the acid is hydrochloric acid. [0295] In some embodiments, the acid is sulfuric acid. [0296] In some embodiments, the acid is phosphoric acid. [0297] In some embodiments, the solvent is THF and H2O (e.g., at a ratio of about 19:1 v/v). [0298] In some embodiments, the method further comprises equilibrating the mixture, e.g., at about 25 °C or at about 50 °C. [0299] In some embodiments, the solvent is removed from the mixture at about 50±20 °C, about 50±15 °C, about 50±10 °C, about 50±9 °C, about 50±8 °C, about 50±7 °C, about 50±6 °C, about 50±5 °C, about 50±4 °C, about 50±3 °C, about 50±2 °C, or about 50±1 °C (e.g., about 50 °C). Preparation of Amorphous Solid Dispersion
Attorney Docket No. ASET-043/001WO 325190-2258 [0300] In some aspects, the present disclosure features a method of preparing an amorphous solid dispersion comprising Compound No. 1, a solvate thereof, a hydrate thereof, or a pharmaceutically acceptable salt thereof. [0301] In some aspects, the present disclosure provides a method of preparing an amorphous solid dispersion comprising Compound No. 1, a solvate thereof, a hydrate thereof, or a pharmaceutically acceptable salt thereof, comprising one or more steps as described herein. [0302] In some aspects, the present disclosure provides an amorphous solid dispersion obtainable by, or obtained by, or directly obtained by a method described herein. [0303] In some embodiments, the method comprises: (i) preparing a mixture comprising Compound No.1, a polymer, and a solvent, and (ii) removing the solvent from the mixture. [0304] In some embodiments, the polymer is PVP K30, PVP VA64, Soluplus
®, HPMC E3, HPMC ASLG, HPMC ASMG, HPMC ASHG, HPC-SSL, Eudragit
® E100, or Eudragit
® L100. [0305] In some embodiments, the solvent is THF and H2O (e.g., at a ratio of about 4:1 v/v or about 9:1 v/v). [0306] In some embodiments, the solvent is removed from the mixture at about 80±20 °C, about 80±15 °C, about 80±10 °C, about 80±9 °C, about 80±8 °C, about 80±7 °C, about 80±6 °C, about 80±5 °C, about 80±4 °C, about 80±3 °C, about 80±2 °C, or about 80±1 °C (e.g., about 80 °C). [0307] In some embodiments, the solvent is removed from the mixture by spray drying. Biological Assays [0308] Compounds designed, selected and/or optimized by methods described above, once produced, can be characterized using a variety of assays known to those skilled in the art to determine whether the compounds have biological activity. For example, the molecules can be characterized by conventional assays, including but not limited to those assays described below, to determine whether they have a predicted activity, binding activity and/or binding specificity. [0309] Furthermore, high-throughput screening can be used to speed up analysis using such assays. As a result, it can be possible to rapidly screen the molecules described herein for activity, using techniques known in the art. General methodologies for performing high- throughput screening are described, for example, in Devlin (1998) High Throughput Screening, Marcel Dekker; and U.S. Patent No.5,763,263. High-throughput assays can use one or more different assay techniques including, but not limited to, those described below.
Attorney Docket No. ASET-043/001WO 325190-2258 [0310] Various in vitro or in vivo biological assays may be suitable for detecting the effect of the compounds of the present disclosure. These in vitro or in vivo biological assays can include, but are not limited to, enzymatic activity assays, electrophoretic mobility shift assays, reporter gene assays, in vitro cell viability assays, and the assays described herein. [0311] In some embodiments, the biological assays assess the ability of the compounds to inhibit cell proliferation. [0312] In some embodiments, cells (e.g., SNU-16 (FGFR2-amplification) cells) may be suspended and dispensed in plates. In some embodiments, cells (e.g., UM-UC-14 (FGFR3- S249C) cells) may be suspended and dispensed in plates. In some embodiments, cells (e.g., DMS-114 (FGFR1 overexpression) cells) may by suspended and dispensed in plates. In some embodiments, cells (e.g., RT-112 (FGFR3-Tacc3 fusion) cells) may be suspended and dispensed in plates. In some embodiments, to determine the effect of the compounds of the present disclosure on cell proliferation, cells (e.g., SNU-16, UM-UC-14, DMS-114 and RT112 cells) may be incubated in the presence of vehicle control (e.g., DMSO) or a compound of the present disclosure at varying concentrations and the inhibition of cell growth may be determined by luminescent quantification (e.g., of intracellular ATP content using CellTiterGlo), according to the manufacturers protocol. In some embodiments, to determine the IC50 values, the vehicle-treated cells were normalized as viable cells and the growth was analyzed using a software (e.g., the CDD Vault software (Collaborative Drug Discovery, Burlingame, CA)). Pharmaceutical Compositions [0313] In some aspects, the present disclosure features pharmaceutical compositions comprising a form (e.g., a morphic form) of Compound No.1, the solvate thereof, the hydrate thereof, or the pharmaceutically acceptable salt thereof described herein, and one or more pharmaceutically acceptable carriers or excipients. [0314] In some embodiments, the pharmaceutical composition comprises Form A of Compound No. 1, the solvate thereof, the hydrate thereof, or the pharmaceutically acceptable salt thereof. [0315] In some embodiments, the pharmaceutical composition comprises Form B of Compound No. 1, the solvate thereof, the hydrate thereof, or the pharmaceutically acceptable salt thereof.
Attorney Docket No. ASET-043/001WO 325190-2258 [0316] In some embodiments, the pharmaceutical composition comprises Form C of Compound No. 1, the solvate thereof, the hydrate thereof, or the pharmaceutically acceptable salt thereof. [0317] In some embodiments, the pharmaceutical composition comprises Form D of Compound No. 1, the solvate thereof, the hydrate thereof, or the pharmaceutically acceptable salt thereof. [0318] In some embodiments, the pharmaceutical composition comprises an amorphous form of Compound No.1, the solvate thereof, the hydrate thereof, or the pharmaceutically acceptable salt thereof. [0319] In some embodiments, the pharmaceutical composition further comprises a polymer. [0320] In some embodiments, the polymer is PVP K30, PVP VA64, Soluplus
®, HPMC E3, HPMC ASLG, HPMC ASMG, HPMC ASHG, HPC-SSL, Eudragit
® E100, or Eudragit
® L100. [0321] The pharmaceutical compositions containing active compounds of the present disclosure may be manufactured in a manner that is generally known, e.g., by means of conventional mixing, dissolving, granulating, dragee-making, levigating, emulsifying, encapsulating, entrapping, or lyophilizing processes. Pharmaceutical compositions may be formulated in a conventional manner using one or more pharmaceutically acceptable carriers comprising excipients and/or auxiliaries that facilitate processing of the active compounds into preparations that can be used pharmaceutically. Of course, the appropriate formulation is dependent upon the route of administration chosen. [0322] Pharmaceutical compositions suitable for injectable use include sterile aqueous solutions (where water soluble) or dispersions and sterile powders for the extemporaneous preparation of sterile injectable solutions or dispersion. For intravenous administration, suitable carriers include physiological saline, bacteriostatic water, Cremophor EL™ (BASF, Parsippany, N.J.) or phosphate buffered saline (PBS). In all cases, the composition must be sterile and should be fluid to the extent that easy syringeability exists. It must be stable under the conditions of manufacture and storage and must be preserved against the contaminating action of microorganisms such as bacteria and fungi. The carrier can be a solvent or dispersion medium containing, for example, water, ethanol, polyol (for example, glycerol, propylene glycol, and liquid polyethylene glycol, and the like), and suitable mixtures thereof. The proper fluidity can be maintained, for example, by the use of a coating such as lecithin, by the maintenance of the required particle size in the case of dispersion and by the use of surfactants. Prevention of the action of microorganisms can be achieved by various antibacterial and
Attorney Docket No. ASET-043/001WO 325190-2258 antifungal agents, for example, parabens, chlorobutanol, phenol, ascorbic acid, thimerosal, and the like. In many cases, it will be preferable to include isotonic agents, for example, sugars, polyalcohols such as mannitol and sorbitol, and sodium chloride in the composition. Prolonged absorption of the injectable compositions can be brought about by including in the composition an agent which delays absorption, for example, aluminum monostearate and gelatin. [0323] Sterile injectable solutions can be prepared by incorporating the active compound in the required amount in an appropriate solvent with one or a combination of ingredients enumerated above, as required, followed by filtered sterilization. Generally, dispersions are prepared by incorporating the active compound into a sterile vehicle that contains a basic dispersion medium and the required other ingredients from those enumerated above. In the case of sterile powders for the preparation of sterile injectable solutions, methods of preparation are vacuum drying and freeze-drying that yields a powder of the active ingredient plus any additional desired ingredient from a previously sterile-filtered solution thereof. [0324] Oral compositions generally include an inert diluent or an edible pharmaceutically acceptable carrier. They can be enclosed in gelatin capsules or compressed into tablets. For the purpose of oral therapeutic administration, the active compound can be incorporated with excipients and used in the form of tablets, troches, or capsules. Oral compositions can also be prepared using a fluid carrier for use as a mouthwash, wherein the compound in the fluid carrier is applied orally and swished and expectorated or swallowed. Pharmaceutically compatible binding agents, and/or adjuvant materials can be included as part of the composition. The tablets, pills, capsules, troches and the like can contain any of the following ingredients, or compounds of a similar nature: a binder such as microcrystalline cellulose, gum tragacanth or gelatin; an excipient such as starch or lactose, a disintegrating agent such as alginic acid, Primogel, or corn starch; a lubricant such as magnesium stearate or Sterotes; a glidant such as colloidal silicon dioxide; a sweetening agent such as sucrose or saccharin; or a flavoring agent such as peppermint, methyl salicylate, or orange flavoring. [0325] For administration by inhalation, the compounds are delivered in the form of an aerosol spray from a pressured container or dispenser, which contains a suitable propellant, e.g., a gas such as carbon dioxide, or a nebulizer. [0326] Systemic administration can also be by transmucosal or transdermal means. For transmucosal or transdermal administration, penetrants appropriate to the barrier to be permeated are used in the formulation. Such penetrants are generally known in the art, and include, for example, transmucosal administration, detergents, bile salts, and fusidic acid derivatives. Transmucosal administration can be accomplished through the use of nasal sprays
Attorney Docket No. ASET-043/001WO 325190-2258 or suppositories. For transdermal administration, the active compounds are formulated into ointments, salves, gels, or creams as generally known in the art. [0327] The active compounds can be prepared with pharmaceutically acceptable carriers that will protect the compound against rapid elimination from the body, such as a controlled release formulation, including implants and microencapsulated delivery systems. Biodegradable, biocompatible polymers can be used, such as ethylene vinyl acetate, polyanhydrides, polyglycolic acid, collagen, polyorthoesters, and polylactic acid. Methods for preparation of such formulations will be apparent to those skilled in the art. [0328] It may be especially advantageous to formulate oral or parenteral compositions in dosage unit form for ease of administration and uniformity of dosage. Dosage unit form as used herein refers to physically discrete units suited as unitary dosages for the subject to be treated; each unit containing a predetermined quantity of active compound calculated to produce the desired therapeutic effect in association with the required pharmaceutical carrier. The specification for the dosage unit forms of the disclosure are dictated by and directly dependent on the unique characteristics of the active compound and the particular therapeutic effect to be achieved. [0329] In therapeutic applications, the dosages of the pharmaceutical compositions used in accordance with the disclosure vary depending on the agent, the age, weight, and clinical condition of the recipient patient, and the experience and judgment of the clinician or practitioner administering the therapy, among other factors affecting the selected dosage. Generally, the dose should be sufficient to result in slowing, and preferably regressing, the symptoms of the disease and also preferably causing complete regression of the disease. [0330] It is understood that the pharmaceutical compositions can be included in a container, pack, or dispenser together with instructions for administration. Methods of Use [0331] In some aspects, the present disclosure provides a method of treating or preventing cancer in a subject, comprising administering to the subject a pharmaceutically effective amount of a form of Compound No. 1, the solvate thereof, the hydrate thereof, or the pharmaceutically acceptable salt thereof. [0332] In some aspects, the present disclosure provides a method of treating or preventing cancer in a subject, comprising administering to the subject a pharmaceutically effective amount of a form of Compound No.1 (e.g., Form A, Form, B, Form C, or Form D) , the solvate thereof, the hydrate thereof, or the pharmaceutically acceptable salt thereof.
Attorney Docket No. ASET-043/001WO 325190-2258 [0333] In some aspects, the present disclosure provides a form of Compound No.1, the solvate thereof, the hydrate thereof, or the pharmaceutically acceptable salt thereof for use in treating or preventing cancer in a subject. [0334] In some aspects, the present disclosure provides a form of Compound No.1 (e.g., Form A, Form, B, Form C, or Form D), the solvate thereof, the hydrate thereof, or the pharmaceutically acceptable salt thereof for use in treating or preventing cancer in a subject. [0335] In some aspects, the present disclosure provides use of a form of Compound No.1, the solvate thereof, the hydrate thereof, or the pharmaceutically acceptable salt thereof, in the manufacture of a medicament for treating or preventing cancer in a subject. [0336] In some aspects, the present disclosure provides use of a form of Compound No. 1 (e.g., Form A, Form, B, Form C, or Form D), the solvate thereof, the hydrate thereof, or the pharmaceutically acceptable salt thereof, in the manufacture of a medicament for treating or preventing cancer in a subject. [0337] In some embodiments, Form A of Compound No. 1, the solvate thereof, the hydrate thereof, or the pharmaceutically acceptable salt thereof is administered. [0338] In some embodiments, Form B of Compound No. 1, the solvate thereof, the hydrate thereof, or the pharmaceutically acceptable salt thereof is administered. [0339] In some embodiments, Form C of Compound No. 1, the solvate thereof, the hydrate thereof, or the pharmaceutically acceptable salt thereof is administered. [0340] In some embodiments, Form D of Compound No. 1, the solvate thereof, the hydrate thereof, or the pharmaceutically acceptable salt thereof is administered. [0341] In some embodiments, an amorphous form of Compound No. 1, the solvate thereof, the hydrate thereof, or the pharmaceutically acceptable salt thereof is administered. Subjects and Diseases [0342] FGFR2 is a human gene located on chromosome 10 that encodes for a protein known as FGFR2. FGFR3 is a human gene located on chromosome 4 that encodes for a protein known as FGFR3. Both FGFR2 and FGFR3 are produced in a variety of different isoforms through alternative splicing. The "b isoforms" are primarily expressed in epithelial tissues while the "c isoforms" are primarily expressed mesenchymal tissues. [0343] FGFR2 and FGFR3 are members of the fibroblast growth factor receptor family. Members of the fibroblast growth factor receptor family are proteins that are located on the cellular surface and bind to various members of the fibroblast growth factor (FGF) family of proteins. FGFR2 and FGFR3 have an extracellular ligand domain, composed of three
Attorney Docket No. ASET-043/001WO 325190-2258 immunoglobulin-like domains, a single transmembrane helix domain, and an intracellular tyrosine kinase domain. Upon binding of an FGF ligand to extracellular ligand domains, the receptors dimerize, and the intracellular tyrosine kinase domains within the dimer cross- phosphorylate each other, thereby activating the kinases domains, allowing them to subsequently bind to adaptor proteins and phosphorylate other intracellular signaling molecules. Members of the fibroblast growth factor receptor family are known to regulate various cellular process including, but not limited to, proliferation and differentiation, particularly within the context of development and tissue repair. [0344] Overexpression of FGFR2 has been implicated in a variety of different cancers, including, but not limited to, gastric cancer and triple negative breast cancer. [0345] Overexpression of FGFR3 has been implicated in a variety of different cancers, including, but not limited to, lung cancer and bladder cancer. [0346] Mutations in the FGFR2 gene, which produce mutant FGFR2 proteins, have been implicated in a variety of different cancers, including, but not limited to, endometrial carcinoma and lung cancer. [0347] Mutations in the FGFR3 gene, which produce mutant FGFR3 proteins, have been implicated in a variety of different cancers, including, but not limited to, bladder cancer. [0348] Fusions of the FGFR2 gene, which produce fusions of FGFR2 proteins, have been implicated in a variety of different cancers, including, but not limited to, bladder cancer and intrahepatic cholangiocarcinoma. [0349] Fusions of the FGFR3 gene, which produce fusions of FGFR3 proteins, have been implicated in a variety of different cancers, including, but not limited to, bladder cancer and glioblastoma. [0350] In some embodiments, the subject is a mammal. [0351] In some embodiments, the subject is a human. [0352] In some embodiments, the subject has previously undergone at least one round of anti- cancer therapy. In some embodiments, the subject has previously undergone at least one round of anti-cancer therapy and has acquired resistance to treatment with the anti-cancer therapy. In some embodiments, the anti-cancer therapy can comprise the administration of at least one of futibatinib, pemigatinib, erdafitinib, infigratinib, Debio-1347. [0353] In some embodiments, the cancer is characterized by at least one oncogenic mutation in the FGFR2 gene. [0354] In some embodiments, the cancer is characterized by at least one oncogenic mutation in the FGFR3 gene.
Attorney Docket No. ASET-043/001WO 325190-2258 [0355] It is understood that a cancer that is characterized by at least one oncogenic mutation in the FGFR2 gene and/or FGFR3 gene is a cancer that is typically associated with at least one oncogenic mutation in the FGFR2 gene and/or FGFR3 gene, including, but not limited to, cancers whose primary oncogenic activity is thought to be driven by the at least one oncogenic mutation in the FGFR2 gene and/or FGFR3 gene. [0356] In some embodiments, the cancer is characterized by overexpression of the FGFR2 gene. [0357] In some embodiments, the cancer is characterized by overexpression of the FGFR3 gene. [0358] It is understood that a cancer that is characterized by overexpression of the FGFR2 gene and/or FGFR3 gene is a cancer that is typically associated with the overexpression of the FGFR2 gene and/or FGFR3 gene, including, but not limited to, cancers whose primary oncogenic activity is thought to be driven by the overexpression of the FGFR2 gene and/or FGFR3 gene. [0359] In some embodiments, the cancer is characterized by at least one oncogenic variant of FGFR2. [0360] In some embodiments, the cancer is characterized by at least one oncogenic variant of FGFR3. [0361] It is understood that a cancer that is characterized by least one oncogenic variant of FGFR2 and/or FGFR3 is a cancer that is typically associated with at least one oncogenic variant of FGFR2 and/or FGFR3, including, but not limited to, cancers whose primary oncogenic activity is thought to be driven by the at least one oncogenic variant of FGFR2 and/or FGFR3. [0362] In some embodiments, the cancer is characterized by overexpression of FGFR2. [0363] In some embodiments, the cancer is characterized by overexpression of FGFR3. [0364] It is understood that a cancer that is characterized by overexpression of FGFR2 and/or FGFR3 is a cancer that is typically associated with overexpression of FGFR2 and/or FGFR3, including, but not limited to, cancers whose primary oncogenic activity is thought to be driven by overexpression of FGFR2 and/or FGFR3. [0365] It is understood that an oncogenic variant of FGFR2 is an FGFR2 protein that comprises at least one oncogenic mutation and that is produced as the result of the expression of a FGFR2 gene that comprises at least one oncogenic mutation. [0366] It is understood that an oncogenic variant of FGFR3 is an FGFR3 protein that comprises at least one oncogenic mutation and that is produced as the result of the expression of a FGFR3 gene that comprises at least one oncogenic mutation.
Attorney Docket No. ASET-043/001WO 325190-2258 [0367] In some embodiments, the subject has at least one oncogenic mutation in the FGFR2 gene. [0368] In some embodiments, the subject has at least one oncogenic mutation in the FGFR3 gene. [0369] In some embodiments, the subject has at least one tumor and/or cancerous cell that expresses an oncogenic variant of FGFR2. [0370] In some embodiments, the subject has at least one tumor and/or cancerous cell that expresses an oncogenic variant of FGFR3. [0371] In some embodiments, the subject has at least one tumor and/or cancerous cell that overexpresses FGFR2. [0372] In some embodiments, the subject has at least one tumor and/or cancerous cell that overexpresses FGFR3. [0373] As would be appreciated by the skilled artisan, in the context of a gene (e.g. FGFR2 and/or FGFR3), an oncogenic mutation can include, but is not limited to a mutation that results in the substitution of one amino acid for another at a specific position within FGFR2 and/or FGFR3, a mutation that results in the substitution of one or more amino acids for one or more amino acids between two specific positions within FGFR2 and/or FGFR3, a mutation that results in an insertion of one or more amino acids between two positions within FGFR2 and/or FGFR3, a mutation that results in the deletion of one more amino acids between two positions within FGFR2 and/or FGFR3, and mutation that results in a fusion of FGFR2 and/or FGFR3, or portion thereof, with another protein, or portion thereof, or any combination thereof. As would be appreciated by the skilled artisan, in the context of a gene, an oncogenic mutation can include, but is not limited to, a missense mutation, a nonsynonymous mutation, an insertion of one or more nucleotides, a deletion of one or more nucleotides, an inversion and a deletion- insertion. As would be appreciated by the skilled artisan, in the context of a gene (e.g. FGRF2 and/or FGFR3), the gene can have one or more of the aforementioned types of oncogenic mutations, including combinations of different types of oncogenic mutations. [0374] As would be appreciated by the skilled artisan, in the context of a protein (e.g. FGFR2 and/or FGFR3), an oncogenic mutation, but is not limited to, the substitution of one amino acid for another at a specific position within FGFR2 and/or FGFR3, the substitution of one or more amino acids for one or more amino acids between two specific positions within FGFR2 and/or FGFR3, an insertion of one or more amino acids between two positions within FGFR2 and/or FGFR3, a deletion of one more amino acids between two positions within FGFR2 and/or FGFR3, and a fusion of FGFR2 and/or FGFR3, or portion thereof, with another protein, or
Attorney Docket No. ASET-043/001WO 325190-2258 portion thereof, or any combination thereof. As would be appreciated by the skilled artisan, in the context of a protein (e.g. FGFR2 and/or FGFR3), the protein can have one or more of the aforementioned types of oncogenic mutations, including combinations of different types of oncogenic mutations. [0375] In some embodiments, an oncogenic mutation of FGFR2 can be any of the FGFR2 mutations put forth in Table 1a. In some embodiments, an oncogenic mutation of FGFR2 can be a gatekeeper mutation that results in resistance to the existing inhibitors, wherein the gatekeeper mutation is V565I or V565F. Table 1a. FGFR2 mutations S252W V565F V565I V565I N550K [0376] In some embodime

, FR3 can be any of the FGFR3 mutations put forth in Table 1b. In some embodiments, an oncogenic mutation of FGFR3 can be a gatekeeper mutation that results in resistance to the existing inhibitors, wherein the gatekeeper mutation is V555M, V555L, or V555F. Table 1b. FGFR3 mutations S249C Tacc3 fusion V555L [0377] In some embod
iments, an oncogenic variant of FGFR2 can be any of the FGFR2 variants put forth in Table 1c. Table 1c. FGFR2 oncogenic variants FGFR2-S252W FGFR2-V565F FGFR2-S252W+V565F
[0378] In some embodiments, an oncogenic variant of FGFR3 can be any of the FGFR3 variants put forth in Table 1d. Table 1d. FGFR3 oncogenic variants FGFR3-S249C FGFR3-Tacc3 fusion FGFR3-S249C+V555M
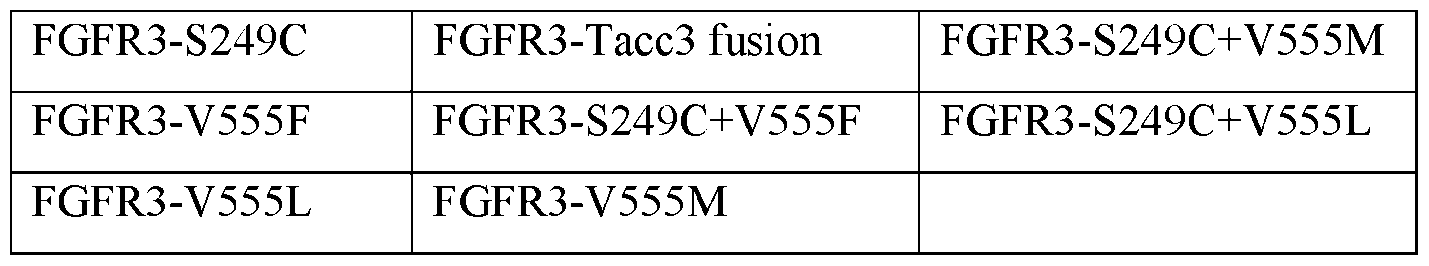
Attorney Docket No. ASET-043/001WO 325190-2258 [0379] In some embodiments, an oncogenic variant of FGFR2 can comprise a gatekeeper mutation that results in resistance to the existing inhibitors, wherein the gatekeeper mutation is V565I or V565F. [0380] In some embodiments, an oncogenic variant of FGFR3 can comprise a gatekeeper mutation that results in resistance to the existing inhibitors, wherein the gatekeeper mutation is V555M, V555L or V555F. [0381] As used herein, the term "activating mutation" refers to any oncogenic mutation that results in at least one of: a) increased ligand binding of FGFR2 and/or FGFR3; b) ligand- independent dimerization and activation of FGFR2 and/or FGFR3; and c) increased kinase activity of FGFR2 and/or FGFR3. [0382] In some embodiments, the cancer is a carcinoma, a lymphoma, a blastoma, a sarcoma, a leukemia, a brain cancer, a breast cancer, a blood cancer, a bone cancer, a lung cancer, a skin cancer, a liver cancer, an ovarian cancer, a bladder cancer, a renal cancer, a kidney cancer, a gastric cancer, a thyroid cancer, a pancreatic cancer, an esophageal cancer, a prostate cancer, a cervical cancer, a uterine cancer, a stomach cancer, a soft tissue cancer, a laryngeal cancer, a small intestine cancer, a testicular cancer, an anal cancer, a vulvar cancer, a joint cancer, an oral cancer, a pharynx cancer or a colorectal cancer. [0383] In some embodiments, the cancer is adrenocortical carcinoma, bladder urothelial carcinoma, breast invasive carcinoma, cervical squamous cell carcinoma, endocervical adenocarcinoma, cholangiocarcinoma, colon adenocarcinoma, lymphoid neoplasm diffuse large B-cell lymphoma, esophageal carcinoma, glioblastoma multiforme, head and neck squamous cell carcinoma, kidney chromophobe, kidney renal clear cell carcinoma, kidney renal papillary cell carcinoma, acute myeloid leukemia, brain lower grade glioma, liver hepatocellular carcinoma, lung adenocarcinoma, lung squamous cell carcinoma, mesothelioma, ovarian serous cystadenocarcinoma, pancreatic adenocarcinoma, pheochromocytoma, paraganglioma, prostate adenocarcinoma, rectum adenocarcinoma, sarcoma, skin cutaneous melanoma, stomach adenocarcinoma, testicular germ cell tumors, thyroid carcinoma, thymoma, uterine carcinosarcoma, uveal melanoma. Other examples include breast cancer, lung cancer, lymphoma, melanoma, liver cancer, colorectal cancer, ovarian cancer, bladder cancer, renal cancer or gastric cancer. Further examples of cancer include neuroendocrine cancer, non-small cell lung cancer (NSCLC), small cell lung cancer, thyroid cancer, endometrial cancer, biliary cancer, esophageal cancer, anal cancer, salivary, cancer, vulvar cancer, cervical cancer, Acute lymphoblastic leukemia (ALL), Acute myeloid
Attorney Docket No. ASET-043/001WO 325190-2258 leukemia (AML), Adrenal gland tumors, Anal cancer, Bile duct cancer, Bladder cancer, Bone cancer, Bowel cancer, Brain tumors, Breast cancer, Cancer of unknown primary (CUP), Cancer spread to bone, Cancer spread to brain, Cancer spread to liver, Cancer spread to lung, Carcinoid, Cervical cancer, Children's cancers, Chronic lymphocytic leukemia (CLL), Chrome myeloid leukemia (CML), Colorectal cancer, Ear cancer, Endometrial cancer, Eye cancer, Follicular dendritic cell sarcoma, Gallbladder cancer, Gastric cancer, Gastro esophageal junction cancers, Germ cell tumors, Gestational trophoblastic disease (GIT)), Hairy cell leukemia, Head and neck cancer, Hodgkin lymphoma, Kaposi’s sarcoma, Kidney cancer, Laryngeal cancer, Leukemia, Gastric linitis plastica, Liver cancer, Lung cancer, Lymphoma, Malignant schwannoma, Mediastinal germ cell tumors, Melanoma skin cancer, Men's cancer, Merkel cell skin cancer, Mesothelioma, Molar pregnancy, Mouth and oropharyngeal cancer, Myeloma, Nasal and paranasal sinus cancer, Nasopharyngeal cancer, Neuroblastoma, Neuroendocrine tumors, Non-Hodgkin lymphoma (NHL), Esophageal cancer, Ovarian cancer, Pancreatic cancer, Penile cancer, Persistent trophoblastic disease and choriocarcinoma, Pheochromocytoma, Prostate cancer, Pseudomyxoma peritonei, Rectal cancer. Retinoblastoma, Salivary gland cancer, Secondary' cancer, Signet cell cancer, Skin cancer, Small bowel cancer, Soft tissue sarcoma, Stomach cancer, T cell childhood non Hodgkin lymphoma (NHL), Testicular cancer, Thymus gland cancer, Thyroid cancer, Tongue cancer, Tonsil cancer, Tumors of the adrenal gland, Uterine cancer. Vaginal cancer, Vulval cancer, Wilms' tumor, Womb cancer and Gynaecological cancer. Examples of cancer also include, but are not limited to, Hematologic malignancies, Lymphoma, Cutaneous T-cell lymphoma, Peripheral T-cell lymphoma, Hodgkin’s lymphoma, Non-Hodgkin’s lymphoma, Multiple myeloma, Chrome lymphocytic leukemia, chronic myeloid leukemia, acute myeloid leukemia, Myelodysplastic syndromes, Myelofibrosis, Biliary tract cancer, Hepatocellular cancer, Colorectal cancer, Breast cancer, Lung cancer, Non-small cell lung cancer, Ovarian cancer, Thyroid Carcinoma, Renal Cell Carcinoma, Pancreatic cancer, Bladder cancer, skin cancer, malignant melanoma, merkel cell carcinoma, Uveal Melanoma or Glioblastoma multiforme. [0384] In some embodiments, the cancer is gastric cancer, triple negative breast cancer, melanoma, hepatobiliary cancer, cancer of unknown primary, esophagogastric cancer, cervical cancer, head and neck cancer, CNS cancer, brain cancer, NSCLC, ovarian cancer, breast cancer, soft tissue sarcoma, pancreatic cancer, prostate cancer, renal cell carcinoma, thyroid cancer, lung cancer, bladder cancer, endometrial carcinoma, intrahepatic cholangiocarcinoma or glioblastoma.
Attorney Docket No. ASET-043/001WO 325190-2258 Exemplary Embodiments [0385] Exemplary Embodiment No. 1. A morphic form of Compound No. 1, a solvate thereof, a hydrate thereof, or a pharmaceutically acceptable salt thereof. [0386] Exemplary Embodiment No. 2. The morphic form of embodiment 1, wherein the morphic form is a crystalline form. [0387] Exemplary Embodiment No. 3. The morphic form of any one of the preceding embodiments, wherein the morphic form is Form A of Compound No. 1, the solvate thereof, the hydrate thereof, or the pharmaceutically acceptable salt thereof. [0388] Exemplary Embodiment No. 4. The morphic form of any one of the preceding embodiments, wherein Form A is characterized by an X-ray diffraction (“XRPD”) pattern comprising signals at 9.8±0.2, 16.2±0.2, and 23.8±0.2 °2θ using Cu Kα radiation. [0389] Exemplary Embodiment No. 5. The morphic form of any one of the preceding embodiments, wherein the XRPD pattern of Form A further comprises at least one signal selected from 19.7±0.2, 25.6±0.2, and 28.1±0.2 °2θ using Cu Kα radiation. [0390] Exemplary Embodiment No. 6. The morphic form of any one of the preceding embodiments, wherein the XRPD pattern of Form A further comprises at least one signal selected from 11.8±0.2, 18.7±0.2, 19.0±0.2, and 21.2±0.2 °2θ using Cu Kα radiation. [0391] Exemplary Embodiment No. 7. The morphic form of any one of the preceding embodiments, wherein Form A is characterized by an XRPD pattern substantially similar to that shown in FIG.1. [0392] Exemplary Embodiment No. 8. The morphic form of any one of the preceding embodiments, wherein Form A is characterized by an XRPD pattern comprising one or more of the signals (e.g., peaks) at approximately the positions shown in Table A. [0393] Exemplary Embodiment No. 9. The morphic form of any one of the preceding embodiments, wherein Form A is characterized by a DSC profile having an endothermic signal at 298 ± 20°C. [0394] Exemplary Embodiment No. 10. The morphic form of any one of the preceding embodiments, wherein Form A shows a weight loss of from about 0.5% to about 3%, at a temperature range from about 25 ± 20 °C to about 125 ± 20 °C, as measured by TGA. [0395] Exemplary Embodiment No. 11. The morphic form of any one of the preceding embodiments, wherein the morphic form is Form B of Compound No. 1, the solvate thereof, the hydrate thereof, or the pharmaceutically acceptable salt thereof.
Attorney Docket No. ASET-043/001WO 325190-2258 [0396] Exemplary Embodiment No. 12. The morphic form of any one of the preceding embodiments, wherein Form B is characterized by an X-ray diffraction (“XRPD”) pattern comprising signals at 6.0±0.2, 17.1±0.2, and 19.6±0.2 °2θ using Cu Kα radiation. [0397] Exemplary Embodiment No. 13. The morphic form of any one of the preceding embodiments, wherein Form B is characterized by an X-ray diffraction (“XRPD”) pattern comprising signals at 15.9±0.2, 18.1±0.2, and 33.4±0.2 °2θ using Cu Kα radiation. [0398] Exemplary Embodiment No. 14. The morphic form of any one of the preceding embodiments, wherein the XRPD pattern of Form A further comprises at least one signal selected from 9.0±0.2, 11.3±0.2, 21.1±0.2, and 30.3±0.2 °2θ using Cu Kα radiation. [0399] Exemplary Embodiment No. 15. The morphic form of any one of the preceding embodiments, wherein Form B is characterized by an XRPD pattern substantially similar to that shown in FIG.4. [0400] Exemplary Embodiment No. 16. The morphic form of any one of the preceding embodiments, wherein Form B is characterized by an XRPD pattern comprising one or more of the signals (e.g., peaks) at approximately the positions shown in Table B. [0401] Exemplary Embodiment No. 17. The morphic form of any one of the preceding embodiments, wherein Form B is characterized by a DSC profile having an endothermic signal at 185 ± 20°C. [0402] Exemplary Embodiment No. 18. The morphic form of any one of the preceding embodiments, wherein Form B shows a weight loss of from about 0.5% to about 15%, at a temperature range from about 35 ± 20 °C to about 130 ± 20 °C, as measured by TGA. [0403] Exemplary Embodiment No. 19. The morphic form of any one of the preceding embodiments, wherein the morphic form is Form C of Compound No. 1, the solvate thereof, the hydrate thereof, or the pharmaceutically acceptable salt thereof. [0404] Exemplary Embodiment No. 20. The morphic form of any one of the preceding embodiments, wherein Form C is characterized by an X-ray diffraction (“XRPD”) pattern comprising signals at 12.8±0.2, 15.3±0.2, and 17.9±0.2 °2θ using Cu Kα radiation. [0405] Exemplary Embodiment No. 21. The morphic form of any one of the preceding embodiments, wherein Form C is characterized by an X-ray diffraction (“XRPD”) pattern comprising signals at 13.1±0.2, 16.5±0.2, and 28.2±0.2 °2θ using Cu Kα radiation. [0406] Exemplary Embodiment No. 22. The morphic form of any one of the preceding embodiments, wherein the XRPD pattern of Form C further comprises at least one signal selected from 5.1±0.2, 11.1±0.2, 22.1±0.2, and 23.2±0.2 °2θ using Cu Kα radiation.
Attorney Docket No. ASET-043/001WO 325190-2258 [0407] Exemplary Embodiment No. 23. The morphic form of any one of the preceding embodiments, wherein Form C is characterized by an XRPD pattern substantially similar to that shown in FIG.7. [0408] Exemplary Embodiment No. 24. The morphic form of any one of the preceding embodiments, wherein Form C is characterized by an XRPD pattern comprising one or more of the signals (e.g., peaks) at approximately the positions shown in Table C. [0409] Exemplary Embodiment No. 25. The morphic form of any one of the preceding embodiments, wherein Form C is characterized by a DSC profile having an endothermic signal at 246 ± 20°C. [0410] Exemplary Embodiment No. 26. The morphic form of any one of the preceding embodiments, wherein Form C shows a weight loss of from about 0.5% to about 3%, at a temperature range from about 35 ± 20 °C to about 130 ± 20 °C, as measured by TGA. [0411] Exemplary Embodiment No. 27. The morphic form of any one of the preceding embodiments, wherein the morphic form is Form D of Compound No. 1, the solvate thereof, the hydrate thereof, or the pharmaceutically acceptable salt thereof. [0412] Exemplary Embodiment No. 28. The morphic form of any one of the preceding embodiments, wherein Form D is characterized by an X-ray diffraction (“XRPD”) pattern comprising signals at 10.6±0.2, 15.9±0.2, and 18.6±0.2 °2θ using Cu Kα radiation. [0413] Exemplary Embodiment No. 29. The morphic form of any one of the preceding embodiments, wherein Form D is characterized by an X-ray diffraction (“XRPD”) pattern comprising signals at 5.3±0.2, 24.0±0.2, and 26.7±0.2 °2θ using Cu Kα radiation. [0414] Exemplary Embodiment No. 30. The morphic form of any one of the preceding embodiments, wherein the XRPD pattern of Form D further comprises at least one signal selected from 12.6±0.2, 14.9±0.2, 20.9±0.2, and 32.2±0.2 °2θ using Cu Kα radiation. [0415] Exemplary Embodiment No. 31. The morphic form of any one of the preceding embodiments, wherein Form D is characterized by an XRPD pattern substantially similar to that shown in FIG.10. [0416] Exemplary Embodiment No. 32. The morphic form of any one of the preceding embodiments, wherein Form D is characterized by an XRPD pattern comprising one or more of the signals (e.g., peaks) at approximately the positions shown in Table D. [0417] Exemplary Embodiment No. 33. The morphic form of any one of the preceding embodiments, wherein Form D is characterized by a DSC profile having an endothermic signal at 251 ± 20°C.
Attorney Docket No. ASET-043/001WO 325190-2258 [0418] Exemplary Embodiment No. 34. The morphic form of any one of the preceding embodiments, wherein Form D shows a weight loss of from about 0.5% to about 4%, at a temperature range from about 35 ± 20 °C to about 130 ± 20 °C, as measured by TGA. [0419] Exemplary Embodiment No.35. A combination comprising: an amorphous form of Compound No. 1, a salt thereof, a solvate thereof, a hydrate thereof, or a pharmaceutically acceptable salt thereof; and a polymer. [0420] Exemplary Embodiment No. 36. A composition comprising the combination of embodiment 35. [0421] Exemplary Embodiment No. 37. The composition of embodiment 36, wherein the polymer comprises a polyvinyl caprolactam-polyvinyl acetate-polyethylene glycol graft co- polymer. [0422] Exemplary Embodiment No. 38. The composition of any one of the preceding embodiments, wherein the polymer is sold as Soluplus
®. [0423] Exemplary Embodiment No. 39. The composition of any one of the preceding embodiments, wherein the composition is characterized by a Tg as measured by mDSC at about 145 °C. [0424] Exemplary Embodiment No. 40. The composition of any one of the preceding embodiments, wherein the composition is characterized by an mDSC profile substantially similar to that shown in FIG.18. [0425] Exemplary Embodiment No. 41. A composition comprising the combination of embodiment 36, wherein the polymer comprises polyvinyl pyrrolidone. [0426] Exemplary Embodiment No. 42. The composition of any one of the preceding embodiments, wherein the polymer is sold as PVK K30. [0427] Exemplary Embodiment No. 43. The composition of any one of the preceding embodiments, wherein the composition is characterized by a Tg as measured by mDSC at about 161 °C. [0428] Exemplary Embodiment No. 44. The composition of any one of the preceding embodiments, wherein the composition is characterized by an mDSC profile substantially similar to that shown in FIG.16. [0429] Exemplary Embodiment No. 45. The composition of embodiment 41, wherein the polymer is sold as PVP VA64.
Attorney Docket No. ASET-043/001WO 325190-2258 [0430] Exemplary Embodiment No. 46. The composition of any one of the preceding embodiments, wherein the composition is characterized by a Tg as measured by mDSC at about 111 °C. [0431] Exemplary Embodiment No. 47. The composition of any one of the preceding embodiments, wherein the composition is characterized by an mDSC profile substantially similar to that shown in FIG.17. [0432] Exemplary Embodiment No. 48. A composition comprising the combination of embodiment 36, wherein the polymer comprises hydroxypropyl methylcellulose. [0433] Exemplary Embodiment No. 49. The composition of any one of the preceding embodiments, wherein the polymer is sold as HPMC E3. [0434] Exemplary Embodiment No. 50. The composition of any one of the preceding embodiments, wherein the composition is characterized by a Tg as measured by mDSC at about 115 °C. [0435] Exemplary Embodiment No. 51. The composition of any one of the preceding embodiments, wherein the composition is characterized by an mDSC profile substantially similar to that shown in FIG.19. [0436] Exemplary Embodiment No. 52. The composition of any one of the preceding embodiments, wherein the composition shows a weight loss of from about 0.5% to about 10%, at a temperature range from about 35 ± 20 °C to about 100 ± 20 °C, as measured by TGA. [0437] Exemplary Embodiment No. 53. The composition of any one of the preceding embodiments, wherein the polymer is sold as HPMC ASLG. [0438] Exemplary Embodiment No. 54. The composition of any one of the preceding embodiments, wherein the composition is characterized by a Tg as measured by mDSC at about 93 °C. [0439] Exemplary Embodiment No. 55. The composition of any one of the preceding embodiments, wherein the composition is characterized by an mDSC profile substantially similar to that shown in FIG.20. [0440] Exemplary Embodiment No. 56. The composition of any one of the preceding embodiments, wherein the polymer is sold as HPMC ASMG. [0441] Exemplary Embodiment No. 57. The composition of any one of the preceding embodiments, wherein the composition is characterized by a Tg as measured by mDSC at about 95 °C.
Attorney Docket No. ASET-043/001WO 325190-2258 [0442] Exemplary Embodiment No. 58. The composition of any one of the preceding embodiments, wherein the composition is characterized by an mDSC profile substantially similar to that shown in FIG.21. [0443] Exemplary Embodiment No. 59. The composition of any one of the preceding embodiments, wherein the composition shows a weight loss of from about 0.5% to about 5%, at a temperature range from about 35 ± 20 °C to about 100 ± 20 °C, as measured by TGA. [0444] Exemplary Embodiment No. 60. The composition of any one of the preceding embodiments, wherein the polymer is sold as HPMC ASHG. [0445] Exemplary Embodiment No. 61. The composition of any one of the preceding embodiments, wherein the composition is characterized by a Tg as measured by mDSC at about 97 °C. [0446] Exemplary Embodiment No. 62. The composition of any one of the preceding embodiments, wherein the composition is characterized by an mDSC profile substantially similar to that shown in FIG.22. [0447] Exemplary Embodiment No. 63. A composition comprising the combination of embodiment 36, wherein the polymer comprises hydroxypropyl cellulose. [0448] Exemplary Embodiment No. 64. The composition of any one of the preceding embodiments, wherein the polymer is sold as HPC-SSL. [0449] Exemplary Embodiment No. 65. The composition of any one of the preceding embodiments, wherein the composition is characterized by a Tg as measured by mDSC at about 89 °C. [0450] Exemplary Embodiment No. 66. The composition of any one of the preceding embodiments, wherein the composition is characterized by an mDSC profile substantially similar to that shown in FIG.23. [0451] Exemplary Embodiment No. 67. A composition comprising the combination of embodiment 36, wherein the polymer comprises a polymethacrylate-based copolymer. [0452] Exemplary Embodiment No. 68. The composition of any one of the preceding embodiments, wherein the polymer is sold as Eudragit
® E100. [0453] Exemplary Embodiment No. 69. The composition of any one of the preceding embodiments, wherein the composition is characterized by a Tg as measured by mDSC at about 93 °C. [0454] Exemplary Embodiment No. 70. The composition of any one of the preceding embodiments, wherein the composition is characterized by an mDSC profile substantially similar to that shown in FIG.24.
Attorney Docket No. ASET-043/001WO 325190-2258 [0455] Exemplary Embodiment No. 71. The composition of any one of the preceding embodiments, wherein the composition shows a weight loss of from about 0.05% to about 2%, at a temperature range from about 35 ± 20 °C to about 100 ± 20 °C, as measured by TGA. [0456] Exemplary Embodiment No. 72. The composition of any one of the preceding embodiments, wherein the polymer is sold as Eudragit® L100. [0457] Exemplary Embodiment No. 73. The composition of any one of the preceding embodiments, wherein the composition is characterized by a Tg as measured by mDSC at about 117 °C. [0458] Exemplary Embodiment No. 74. The composition of any one of the preceding embodiments, wherein the composition is characterized by an mDSC profile substantially similar to that shown in FIG.25. [0459] Exemplary Embodiment No. 75. A pharmaceutical composition comprising the morphic form of any one of the preceding embodiments and one or more pharmaceutically acceptable carriers or excipients. [0460] Exemplary Embodiment No. 76. A method of treating or preventing cancer in a subject, the method comprising administering to the subject the morphic form of any one of the preceding embodiments. [0461] Exemplary Embodiment No. 77. A morphic form of any one of the preceding embodiments for treating or preventing cancer in a subject. [0462] Exemplary Embodiment No.78. Use of the morphic form of any one of the preceding embodiments in the manufacture of a medicament for treating or preventing cancer in a subject. [0463] Exemplary Embodiment No.79. Use of the morphic form of any one of the preceding embodiments for treating or preventing cancer in a subject. [0464] Exemplary Embodiment No.80. The method, morphic form, or use of any one of the preceding embodiments, wherein the subject has previously undergone at least one round of anti-cancer therapy. [0465] Exemplary Embodiment No.81. The method, morphic form, or use of any one of the preceding embodiments, wherein the cancer is characterized by at least one oncogenic mutation in the FGFR2 gene or the FGFR3 gene. [0466] Exemplary Embodiment No.82. The method, morphic form, or use of any one of the preceding embodiments, wherein the cancer is characterized by overexpression of the FGFR2 gene or the FGFR3 gene.
Attorney Docket No. ASET-043/001WO 325190-2258 [0467] Exemplary Embodiment No.83. The method, morphic form, or use of any one of the preceding embodiments, wherein the cancer is characterized by at least one oncogenic variant of FGFR2 or FGFR3. [0468] Exemplary Embodiment No.84. The method, morphic form, or use of any one of the preceding embodiments, wherein the cancer is a carcinoma, a lymphoma, a blastoma, a sarcoma, a leukemia, a brain cancer, a breast cancer, a blood cancer, a bone cancer, a lung cancer, a skin cancer, a liver cancer, an ovarian cancer, a bladder cancer, a renal cancer, a kidney cancer, a gastric cancer, a thyroid cancer, a pancreatic cancer, an esophageal cancer, a prostate cancer, a cervical cancer, a uterine cancer, a stomach cancer, a soft tissue cancer, a laryngeal cancer, a small intestine cancer, a testicular cancer, an anal cancer, a vulvar cancer, a joint cancer, an oral cancer, a pharynx cancer or a colorectal cancer. Definitions [0469] It is understood that the compounds described herein include the compounds themselves, as well as their salts, and their solvates, if applicable. A salt, for example, can be formed between an anion and a positively charged group (e.g., amino) on a substituted benzene compound. Suitable anions include chloride, bromide, iodide, sulfate, bisulfate, sulfamate, nitrate, phosphate, citrate, methanesulfonate, trifluoroacetate, glutamate, glucuronate, glutarate, malate, maleate, succinate, fumarate, tartrate, tosylate, salicylate, lactate, naphthalenesulfonate, and acetate (e.g., trifluoroacetate). [0470] Unless explicitly indicated otherwise, the terms “approximately” and “about” are synonymous. In some embodiments, “approximately” and “about” refer to the recited amount, value, dose, or duration ± 10%, ± 8%, ± 6%, ± 5%, ± 4%, ± 2%, ± 1%, or ± 0.5%. In some embodiments, “approximately” and “about” refer to the listed amount or duration ± 10%, ± 8%, ± 6%, ± 5%, ± 4%, or ± 2%. In some embodiments, “approximately” and “about” refer to the listed amount, value, dose, or duration ± 5%. In some embodiments, “approximately” and “about” refer to the listed amount, value, dose, or duration ± 2%. In some embodiments, “approximately” and “about” refer to the listed amount, value, dose, or duration ± 1%. In some embodiments, “approximately” and “about” refer to the listed value (e.g., for PXRD signal (e.g., peak) positions) ± 0.2, ± 0.15, ± 0.1, or ± 0.05. [0471] As used herein, the term “pharmaceutically acceptable anion” refers to an anion suitable for forming a pharmaceutically acceptable salt. Likewise, a salt can also be formed between a cation and a negatively charged group (e.g., carboxylate) on a substituted benzene compound. Suitable cations include sodium ion, potassium ion, magnesium ion, calcium ion,
Attorney Docket No. ASET-043/001WO 325190-2258 and an ammonium cation such as tetramethylammonium ion. The substituted benzene compounds also include those salts containing quaternary nitrogen atoms. [0472] It is understood that the compounds of the present disclosure, for example, the salts of the compounds, can exist in either hydrated or unhydrated (the anhydrous) form or as solvates with other solvent molecules. Nonlimiting examples of hydrates include monohydrates and dihydrates. Nonlimiting examples of solvates include ethanol solvates and acetone solvates. [0473] As used herein, the expressions “one or more of A, B, or C,” “one or more A, B, or C,” “one or more of A, B, and C,” “one or more A, B, and C,” “selected from the group consisting of A, B, and C”, “selected from A, B, and C”, and the like are used interchangeably and all refer to a selection from a group consisting of A, B, and/or C, i.e., one or more As, one or more Bs, one or more Cs, or any combination thereof, unless indicated otherwise. [0474] It is understood that, throughout the description, where compositions are described as having, including, or comprising specific components, it is contemplated that compositions also consist essentially of, or consist of, the recited components. Similarly, where methods or processes are described as having, including, or comprising specific process steps, the processes also consist essentially of, or consist of, the recited processing steps. Further, it should be understood that the order of steps or order for performing certain actions is immaterial so long as the invention remains operable. Moreover, two or more steps or actions can be conducted simultaneously. [0475] It is understood that compounds of the present disclosure can be prepared in a variety of ways using commercially available starting materials, compounds known in the literature, or from readily prepared intermediates, by employing standard synthetic methods and procedures either known to those skilled in the art, or which will be apparent to the skilled artisan in light of the teachings herein. Standard synthetic methods and procedures for the preparation of organic molecules and functional group transformations and manipulations can be obtained from the relevant scientific literature or from standard textbooks in the field. Although not limited to any one or several sources, classic texts such as Smith, M. B., March, J., March’s Advanced Organic Chemistry: Reactions, Mechanisms, and Structure, 5
th edition, John Wiley & Sons: New York, 2001; Greene, T.W., Wuts, P.G. M., Protective Groups in Organic Synthesis, 3
rd edition, John Wiley & Sons: New York, 1999; R. Larock, Comprehensive Organic Transformations, VCH Publishers (1989); L. Fieser and M. Fieser, Fieser and Fieser’s Reagents for Organic Synthesis, John Wiley and Sons (1994); and L. Paquette, ed., Encyclopedia of Reagents for Organic Synthesis, John Wiley and Sons (1995),
Attorney Docket No. ASET-043/001WO 325190-2258 incorporated by reference herein, are useful and recognized reference textbooks of organic synthesis known to those in the art. [0476] It is to be understood that, unless otherwise stated, any description of a method of treatment or prevention includes use of a crystalline form of Compound No. 1 or a pharmaceutically acceptable salt thereof to provide such treatment or prevention as is described herein. It is to be further understood, unless otherwise stated, any description of a method of treatment or prevention includes use of a crystalline form of Compound No. 1 or a pharmaceutically acceptable salt thereof to prepare a medicament to treat or prevent such condition. The treatment or prevention includes treatment or prevention of human or non- human animals including rodents and other disease models. [0477] It is to be understood that, unless otherwise stated, any description of a method of treatment includes use of a crystalline form of Compound No. 1 or a pharmaceutically acceptable salt thereof to provide such treatment as is described herein. It is to be further understood, unless otherwise stated, any description of a method of treatment includes use of a crystalline form of Compound No.1 or a pharmaceutically acceptable salt thereof to prepare a medicament to treat such condition. The treatment includes treatment of human or non-human animals including rodents and other disease models. [0478] As used herein, the term “subject” refers to a subject having a disease or having an increased risk of developing the disease. A “subject” includes a mammal. The mammal can be e.g., a human or appropriate non-human mammal, such as primate, mouse, rat, dog, cat, cow, horse, goat, camel, sheep or a pig. The subject can also be a bird or fowl. In one embodiment, the mammal is a human. [0479] In some embodiments, the term “subject in need thereof” can be one who has been previously diagnosed or identified as having a disease or disorder disclosed herein. A subject in need thereof can also be one who is suffering from a disease or disorder disclosed herein. Alternatively, a subject in need thereof can be one who has an increased risk of developing such disease or disorder relative to the population at large (i.e., a subject who is predisposed to developing such disorder relative to the population at large). A subject in need thereof can have a refractory or resistance to a disease or disorder disclosed herein (i.e., a disease or disorder disclosed herein that does not respond or has not yet responded to treatment). The subject in need thereof may be resistant at the start of treatment or may become resistant during treatment. In some embodiments, the subject in need thereof received and failed all known effective therapies for a disease or disorder disclosed herein. In some embodiments, the subject in need thereof received at least one prior therapy.
Attorney Docket No. ASET-043/001WO 325190-2258 [0480] As used herein, the term “treating” or “treat” describes the management and care of a patient for the purpose of combating a disease, condition, or disorder and includes the administration of a compound of the present disclosure, or a pharmaceutically acceptable salt, polymorph or solvate thereof, to alleviate the symptoms or complications of a disease, condition or disorder, or to eliminate the disease, condition or disorder. The term “treat” can also include treatment of a cell in vitro or an animal model. [0481] It is to be understood that a crystalline form of Compound No.1 or a pharmaceutically acceptable salt thereof, can or may also be used to prevent a relevant disease, condition or disorder, or used to identify suitable candidates for such purposes. [0482] As used herein, the term “preventing,” “prevent,” or “protecting against” describes reducing or eliminating the onset of the symptoms or complications of such disease, condition or disorder. [0483] It is to be understood that “solubility” or “solubility rating” refers to the property of a polymorph (e.g., Form A, B, C, or D) disclosed herein to dissolve in a liquid solvent and form a homogeneous solution. In some embodiments, solubility is expressed as a concentration, either by mass of solute per unit volume of solvent (e.g., g of solute per kg of solvent, g per dL (100 mL), mg/ml, etc.), molarity, molality, mole fraction, or other similar descriptions of concentration. A person of skill in the art may understand that the maximum equilibrium amount of solute that can dissolve per amount of solvent is the solubility of that solute in that solvent under the specified conditions, including temperature, pressure, pH, and the nature of the solvent. [0484] As used herein, “stable” refers to a polymorph that maintains purity, appearance, and/or analytical parameters over a defined time and temperature as compared to the polymorph as isolated. In some embodiments, the “stable” polymorph exhibits less than about 10%, about 9%, about 8%, about 7%, about 6%, about 5%, about 4%, about 3%, about 2%, about 1%, about 0.9%, about 0.8%, about 0.7%, about 0.6%, about 0.5%, about 0.4%, about 0.3%, about 0.2%, or about 0.1% impurity over a set period of time (e.g., 1 day, 2 days, 3 days, 4 days, 5 days, 6 days, one week, two weeks, three weeks, one month, two months, three months, or four months). [0485] As used herein, the term “pharmaceutical composition” is a formulation containing the compounds of the present disclosure in a form suitable for administration to a subject. In one embodiment, the pharmaceutical composition is in bulk or in unit dosage form. The unit dosage form is any of a variety of forms, including, for example, a capsule, an IV bag, a tablet, a single pump on an aerosol inhaler or a vial. The quantity of active ingredient (e.g., a formulation of
Attorney Docket No. ASET-043/001WO 325190-2258 the disclosed compound or salt, hydrate, solvate or isomer thereof) in a unit dose of composition is an effective amount and is varied according to the particular treatment involved. One skilled in the art will appreciate that it is sometimes necessary to make routine variations to the dosage depending on the age and condition of the patient. The dosage will also depend on the route of administration. A variety of routes are contemplated, including oral, pulmonary, rectal, parenteral, transdermal, subcutaneous, intravenous, intramuscular, intraperitoneal, inhalational, buccal, sublingual, intrapleural, intrathecal, intranasal, and the like. Dosage forms for the topical or transdermal administration of a compound of this disclosure include powders, sprays, ointments, pastes, creams, lotions, gels, solutions, patches and inhalants. In one embodiment, the active compound is mixed under sterile conditions with a pharmaceutically acceptable carrier, and with any preservatives, buffers, or propellants that are required. [0486] As used herein, the term “pharmaceutically acceptable” refers to those compounds, anions, cations, materials, compositions, carriers, and/or dosage forms which are, within the scope of sound medical judgment, suitable for use in contact with the tissues of human beings and animals without excessive toxicity, irritation, allergic response, or other problem or complication, commensurate with a reasonable benefit/risk ratio. [0487] As used herein, the term “pharmaceutically acceptable excipient” means an excipient that is useful in preparing a pharmaceutical composition that is generally safe, non-toxic and neither biologically nor otherwise undesirable, and includes excipient that is acceptable for veterinary use as well as human pharmaceutical use. A “pharmaceutically acceptable excipient” as used in the specification and claims includes both one and more than one such excipient. [0488] As used herein, the term “therapeutically effective amount”, refers to an amount of a pharmaceutical agent to treat, ameliorate, or prevent an identified disease or condition, or to exhibit a detectable therapeutic or inhibitory effect. The effect can be detected by any assay method known in the art. The precise effective amount for a subject will depend upon the subject’s body weight, size, and health; the nature and extent of the condition; and the therapeutic or combination of therapeutics selected for administration. Therapeutically effective amounts for a given situation can be determined by routine experimentation that is within the skill and judgment of the clinician. [0489] It is understood that, for the compounds of the present disclosure being capable of further forming salts, all of these forms are also contemplated within the scope of the claimed disclosure. [0490] As used herein, the term “pharmaceutically acceptable salts” refer to derivatives of the compounds of the present disclosure wherein the parent compound is modified by making acid
Attorney Docket No. ASET-043/001WO 325190-2258 or base salts thereof. In some embodiments, the pharmaceutically acceptable salt of a compound is also a prodrug of the compound. Examples of pharmaceutically acceptable salts include, but are not limited to, mineral or organic acid salts of basic residues such as amines, alkali or organic salts of acidic residues such as carboxylic acids, and the like. The pharmaceutically acceptable salts include the conventional non-toxic salts or the quaternary ammonium salts of the parent compound formed, for example, from non-toxic inorganic or organic acids. For example, such conventional non-toxic salts include, but are not limited to, those derived from inorganic and organic acids selected from 2-acetoxybenzoic, 2- hydroxyethane sulfonic, acetic, ascorbic, benzene sulfonic, benzoic, bicarbonic, carbonic, citric, edetic, ethane disulfonic, 1,2-ethane sulfonic, fumaric, glucoheptonic, gluconic, glutamic, glycolic, glycollyarsanilic, hexylresorcinic, hydrabamic, hydrobromic, hydrochloric, hydroiodic, hydroxymaleic, hydroxynaphthoic, isethionic, lactic, lactobionic, lauryl sulfonic, maleic, malic, mandelic, methane sulfonic, napsylic, nitric, oxalic, pamoic, pantothenic, phenylacetic, phosphoric, polygalacturonic, propionic, salicylic, stearic, subacetic, succinic, sulfamic, sulfanilic, sulfuric, tannic, tartaric, toluene sulfonic, and the commonly occurring amine acids, e.g., glycine, alanine, phenylalanine, arginine, etc. [0491] Other examples of pharmaceutically acceptable salts include hexanoic acid, cyclopentane propionic acid, pyruvic acid, malonic acid, 3-(4-hydroxybenzoyl)benzoic acid, cinnamic acid, 4-chlorobenzenesulfonic acid, 2-naphthalenesulfonic acid, 4-toluenesulfonic acid, camphorsulfonic acid, 4-methylbicyclo-[2.2.2]-oct-2-ene-1-carboxylic acid, 3- phenylpropionic acid, trimethylacetic acid, tertiary butylacetic acid, muconic acid, and the like. The present disclosure also encompasses salts formed when an acidic proton present in the parent compound either is replaced by a metal ion, e.g., an alkali metal ion, an alkaline earth ion, or an aluminum ion; or coordinates with an organic base such as ethanolamine, diethanolamine, triethanolamine, tromethamine, N-methylglucamine, and the like. In the salt form, it is understood that the ratio of the compound to the cation or anion of the salt can be 1:1, or any ration other than 1:1, e.g., 3:1, 2:1, 1:2, or 1:3. [0492] It is understood that all references to pharmaceutically acceptable salts include solvent addition forms (solvates) or crystal forms (polymorphs) as defined herein, of the same salt. [0493] The compounds, or pharmaceutically acceptable salts thereof, are administered orally, nasally, transdermally, pulmonary, inhalationally, buccally, sublingually, intraperitoneally, subcutaneously, intramuscularly, intravenously, rectally, intrapleurally, intrathecally and parenterally. In one embodiment, the compound is administered orally. One skilled in the art will recognize the advantages of certain routes of administration.
Attorney Docket No. ASET-043/001WO 325190-2258 [0494] The dosage regimen utilizing the compounds is selected in accordance with a variety of factors including type, species, age, weight, sex and medical condition of the patient; the severity of the condition to be treated; the route of administration; the renal and hepatic function of the patient; and the particular compound or salt thereof employed. An ordinarily skilled physician or veterinarian can readily determine and prescribe the effective amount of the drug required to prevent, counter, or arrest the progress of the condition. [0495] Techniques for formulation and administration of the disclosed compounds of the disclosure can be found in Remington: the Science and Practice of Pharmacy, 19
th edition, Mack Publishing Co., Easton, PA (1995). In an embodiment, the compounds described herein, and the pharmaceutically acceptable salts thereof, are used in pharmaceutical preparations in combination with a pharmaceutically acceptable carrier or diluent. Suitable pharmaceutically acceptable carriers include inert solid fillers or diluents and sterile aqueous or organic solutions. The compounds will be present in such pharmaceutical compositions in amounts sufficient to provide the desired dosage amount in the range described herein. [0496] All percentages and ratios used herein, unless otherwise indicated, are by weight. Other features and advantages of the present disclosure are apparent from the different examples. The provided examples illustrate different components and methodology useful in practicing the present disclosure. The examples do not limit the claimed disclosure. Based on the present disclosure the skilled artisan can identify and employ other components and methodology useful for practicing the present disclosure. [0497] All publications and patent documents cited herein are incorporated herein by reference as if each such publication or document was specifically and individually indicated to be incorporated herein by reference. Citation of publications and patent documents is not intended as an admission that any is pertinent prior art, nor does it constitute any admission as to the contents or date of the same. The invention having now been described by way of written description, those of skill in the art will recognize that the invention can be practiced in a variety of embodiments and that the foregoing description and examples below are for purposes of illustration and not limitation of the claims that follow. EXAMPLES [0498] It is understood that the values described in the examples are approximate and subject to experimental and instrumental variations.
Attorney Docket No. ASET-043/001WO 325190-2258 Instrumentation [0499] Polarized Light Microscopy (PLM). Details of polarized light microscope method used in the tests are mentioned below: • Nikon LV100POL equipped with 5 megapixel CCD • Physical Lens: 20X, 10X [0500] X-Ray Powder Diffractometer (XRPD). Samples were spread onto the Si-substrate for XRPD test using the parameters as below: • Tube: Cu: K- Alpha (λ=1.54179Ǻ). • Generator: Voltage: 40 kV; Current: 40 mA. • Scan Scope: 3 to 40 deg; • Scanning rate: 10 deg./min • Sample rotation speed: 15 rpm [0501] Thermal Gravimetric Analysis (TGA). Powders were placed into an open platinum pan from TA Instruments for TGA test (conventional TGA) using the parameters as below: • Ramp 10°C/min, RT to 300°C for weight % < 80% • Purge gas: 25 mL/min N
2 [0502] Differential Scanning Calorimetry (DSC). Powders were weighed into Tzero aluminum sample pan from TA Instruments covered by pinhole lid for DSC test (conventional DSC) using the parameters as below: • Equilibrate at 10°C • Modulate +/- 1°C every 60 seconds • Isothermal for 5.00 min • Ramp 10°C/min, RT to 300°C • Purge gas: 50 mL/min N
2 [0503] Modulated Differential Scanning Calorimetry (mDSC). ASD powders were weighed into Tzero aluminum sample pan from TA Instruments covered by pinhole lid for mDSC test (conventional mDSC) using the parameters as below: • Equilibrate at 10°C • Modulate +/- 1°C every 60 seconds • Isothermal for 5.00 min • Ramp 2°C/min, RT to 250°C • Purge gas: 50 mL/min N2
Attorney Docket No. ASET-043/001WO 325190-2258 Example 1. Exemplary Preparation and Study of Form A. [0504] Suspension of Compound No. 1 in different solvents were prepared. Obtained suspension was filtered through a 0.45 µm nylon membrane filter by centrifugation at 14,000 rpm after been stirred for 24 hours and 1 week. Solid parts (wet cakes) were vacuum dried at 50 °C for 2 hours and investigated by XRPD. Results are shown in Table I-a. Table I-a Solvent Concentration 24 h XRPD 1 Week XRPD D
MSO 35 mg/ml 25 °C Form A 25 °C Form A
n was set on polymorph screen plate at a heating-cooling rate of 0.1 °C/min from 5 °C to 50 °C for 10 cycles. Precipitates were collected by centrifugation filtration through a 0.45 µm nylon membrane filter at 14,000 rpm. Solid parts (wet cakes) were vacuum dried at 50 °C for 2h and investigated by XRPD. Results are shown in Table I-b. Table I-b S
olvent Preparation Cooling T
emperature Temperature Concentration XRPD
Example 2. Exemplary Preparation and Study of Forms B, C, and D. [0506] Various salt samples were prepared based on the conditions and procedures shown in Table II-a. Table II-a No. Counter-ions DMSO THF:H2O (95:5)

apo a o a
Attorney Docket No. ASET-043/001WO 325190-2258 [0507] For solid counter-ions, 5 mg of Compound No. 1 and 1.1 e.q. counter ions were weighed into 2 mL vials individually, and then 0.5 mL solvent was added into the vials. For liquid counter-ions, 5 mg of Compound No. 1 were weighed into 2 mL vials, and 0.5 mL solvent was added in the vials subsequently. Then 1.1 e.q. counter-ions solutions in solvent (22 μL, concentration: 0.5 mol/L) were added into the vials. The vials were placed on the magnetic stirrers and heated to 50°C. After keeping at 50°C under stirring at 600 rpm for 18 hrs, the vials were then cooled to 25°C. After keeping at 25°C till 3 days, the solids in suspensions were isolated by centrifugation and dried in the vacuum oven at 50°C for 2 hrs followed by characterization by XRPD. For those clear or hazy solution systems, the solvent was evaporated at 50°C for 2 hs and then the obtained solids were also characterized by XRPD, for those with different crystalline XRPD patterns, DSC and TGA were also performed to confirm the salt formation. The observation of results are summarized in Table II-b. Table II-b H
ydrochloric Sulfuric Phosphoric Fumaric Maleic (+)-L- Citric A
id A id A id A id A id Tartaric A id us u
s
xamp e . xemp ary repara on o morp ous orm an spers on. [0508] Amorphous Solid Dispersion (ASD) by Fast Evaporation. [0509] Solubility Test: Each solvent was added in increments of 100 μL into a 4-mL glass vial containing about 5 mg of Compound No.1, until the solids were dissolved or a total volume of 4 mL was reached. Results are summarized in Table III-a. According to solubility data, solvent properties and safety class, THF/Water=80%/20% (v,v) was appropriate as solvent system for ASD preparation. Table III-a Solvent Safety class Solubility (mg/mL) M H 2 12

Attorney Docket No. ASET-043/001WO 325190-2258 [0510] Polymer Screening: Fast evaporation was adopted to remove solvent to screen available polymers for ASD preparation. [0511] Preparation through Fast Evaporation: Samples for characterization: About 10 mg of Compound No.1 and 40 mg of corresponding polymers were weighed into 40-mL glass bottles, and 1 mL of solvents was added to obtain the clear solution with 20% drug load. Then, the solvent was removed fast through evaporation at 80°C. [0512] Kinetic Solubility Study: About 4 mg of Compound No.1 and 16 mg of corresponding polymers were weighed into 4-mL glass bottles, and 0.4 mL of solvents was added to obtain the clear solution with 20% load. The solvent was then removed through fast evaporation at 80°C. Compound No. 1 as control was removed the same way. Results are summarized in Table III-b. Table III-b SD Solvents and Tg by Formulation
Volume Appearance XRPD PLM mDSC
Attorney Docket No. ASET-043/001WO 325190-2258 [0513] Amorphous Solid Dispersion (ASD) by Spray Drying. [0514] Mixture of Compound No. 1 and polymer (1:4, w/w), with Compound No. 1 concentration of 10 mg/mL, were dissolved in THF/Water=90%/10% (v,v) and used as spray drying solution for solid dispersion preparation. About 0.2 g of Compound No. 1, 0.8 g of polymer, and 20 mL THF/Water=90%/10% were charged into 40 ml bottle and dissolved by magnetic stirring to obtain clear solution, followed by spray drying to form solid dispersions. Detail process parameters for solid dispersion preparation were listed in Table III-c. Obtained powders were dried in a vacuum drier at 40°C overnight, and the yield was ~75%. Further physical characterization and evaluation were performed. Table III-c Compound No.1 Compound No.1 Compound No. 1 + Eudragi
® + HPMC E3 + HPMC ASMG t
[0515] It is understood that the invention can be embodied in other specific forms without departing from the spirit or essential characteristics thereof. The foregoing embodiments are therefore to be considered in all respects illustrative rather than limiting on the invention described herein. Scope of the invention is thus indicated by the appended claims rather than by the foregoing description, and all changes that come within the meaning and range of equivalency of the claims are intended to be embraced therein.



















