WO2025117353A1 - Crosslinked and hydrophobically modified xanthan gum - Google Patents
Crosslinked and hydrophobically modified xanthan gum Download PDFInfo
- Publication number
- WO2025117353A1 WO2025117353A1 PCT/US2024/057020 US2024057020W WO2025117353A1 WO 2025117353 A1 WO2025117353 A1 WO 2025117353A1 US 2024057020 W US2024057020 W US 2024057020W WO 2025117353 A1 WO2025117353 A1 WO 2025117353A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- xanthan gum
- modified xanthan
- crosslinked
- ether
- emulsion
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08B—POLYSACCHARIDES; DERIVATIVES THEREOF
- C08B37/00—Preparation of polysaccharides not provided for in groups C08B1/00 - C08B35/00; Derivatives thereof
- C08B37/0006—Homoglycans, i.e. polysaccharides having a main chain consisting of one single sugar, e.g. colominic acid
- C08B37/0024—Homoglycans, i.e. polysaccharides having a main chain consisting of one single sugar, e.g. colominic acid beta-D-Glucans; (beta-1,3)-D-Glucans, e.g. paramylon, coriolan, sclerotan, pachyman, callose, scleroglucan, schizophyllan, laminaran, lentinan or curdlan; (beta-1,6)-D-Glucans, e.g. pustulan; (beta-1,4)-D-Glucans; (beta-1,3)(beta-1,4)-D-Glucans, e.g. lichenan; Derivatives thereof
- C08B37/0033—Xanthan, i.e. D-glucose, D-mannose and D-glucuronic acid units, saubstituted with acetate and pyruvate, with a main chain of (beta-1,4)-D-glucose units; Derivatives thereof
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61Q—SPECIFIC USE OF COSMETICS OR SIMILAR TOILETRY PREPARATIONS
- A61Q19/00—Preparations for care of the skin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61Q—SPECIFIC USE OF COSMETICS OR SIMILAR TOILETRY PREPARATIONS
- A61Q19/00—Preparations for care of the skin
- A61Q19/10—Washing or bathing preparations
Definitions
- the present invention relates to crosslinked and hydrophobically modified xanthan gum polymers and to a method for making the same. These compounds are useful for the preparation of personal care, home care, industrial, and health care applications, particularly as emulsifiers in the formulation of phase stable emulsions.
- Microbial polysaccharides are biocompatible, biodegradable, and usually nontoxic natural biopolymers that possess physicochemical properties suitable for use as cosmetic ingredients. These properties are also inherent to xanthan gum.
- Xanthan gum is a microbial polysaccharide produced commercially by a fermentation process using Xanthomonas campestris bacteria.
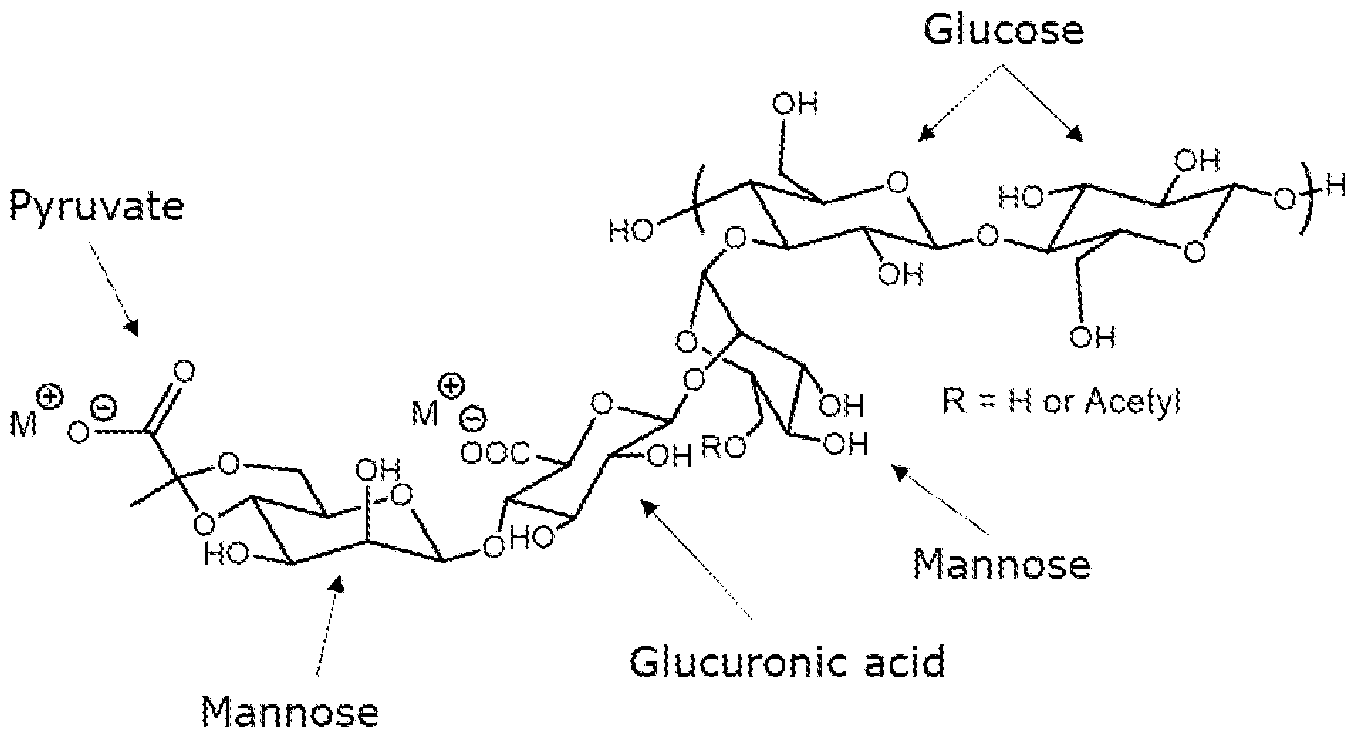
- the structure of xanthan gum is composed of a p-1 ,4-D-glucose backbone (identical to cellulose) with a regular trimeric sidechain on every second glucose to make a pentasaccharide repeat unit.
- Each side chain is connected to the backbone via an a-1 ,3 linkage and has a glucuronic acid between two mannoses.
- an aqueous solution of xanthan gum exhibits high intrinsic viscosity at low concentrations and behaves like a pseudoplastic fluid.
- the high viscous rheology, pH and salt-resistant properties of xanthan gum are satisfactory for use as thickening agent, suspending agent, emulsion stabilization for food, pharmaceuticals, oral care and cosmetics.
- native xanthan gum has its own limitations, including unstable viscosity, inadequate impact on yield values, insufficient performance stabilizing emulsions, unsatisfactory texture and being susceptible to microbial contamination.
- JP9003101A provides a method for improving the thickening effect and transparency of xanthan gum as well as improving water solubility, in order to provide useful xanthan derivatives in toiletry and cosmetic fields.
- the present invention sets out to meet some or all of the above-identified needs and solve some or all of the above-identified problems.
- the disclosed invention relates to a crosslinked and hydrophobically modified xanthan gum that offers a more natural and biodegradable alternative to traditional emulsifiers, while provides superior thickening and stabilization of emulsions comprising a variety of oils (polar to non-polar).
- the modified xanthan gum disclosed herein also allows to formulate emulsion compositions with suitable texture and superior electrolyte stability at low use levels.
- one object of the present invention is the provision of a crosslinked and hydrophobically modified xanthan gum wherein a portion of the hydrogen atoms of pendant hydroxyl groups of the backbone are substituted with at least one hydrophobic moiety according to formula (I):
- A is selected from a divalent linear or branched, substituted or unsubstituted C1- C6 alkylene radical;
- R is selected from a hydrocarbon group having from 8 to 22 carbon atoms; a is 0 or 1 , and b is 0 or 1 , with the proviso that when a is 0, b is 0; and, wherein the xanthan gum is crosslinked with a multifunctional crosslinking agent comprising epoxy and/or alkyl halide functional groups in an amount of from 0.1 to 1 wt.% based on the total weight the xanthan gum.
- Another object of the invention is the provision of oil-in-water emulsions comprising: a water phase; an oil phase dispersed in said water phase; and, the crosslinked and hydrophobically modified xanthan gum described herein.
- a further object of the invention is a method of stabilizing an oil-in-water emulsion comprising an oil phase and a water phase comprising the step of mixing a crosslinked and hydrophobically modified xanthan gum described herein with the oil phase and the water phase.
- the present invention also provides cleaning or fabric care compositions comprising the crosslinked and hydrophobically modified xanthan of the present invention.
- Another object of the invention is the provision of a method for preparing the crosslinked and hydrophobically modified xanthan gum of the present invention comprising reacting a xanthan gum with a) an alkylating agent and b) a multifunctional crosslinker comprising epoxide and/or alkyl halide functional groups.
- Figure 1 Amplitude sweeps for: Xanthan (native xanthan gum, not reacted), control xanthan gum that underwent same reaction conditions but without hydrophobe, and hydrophobically modified xanthan gum.
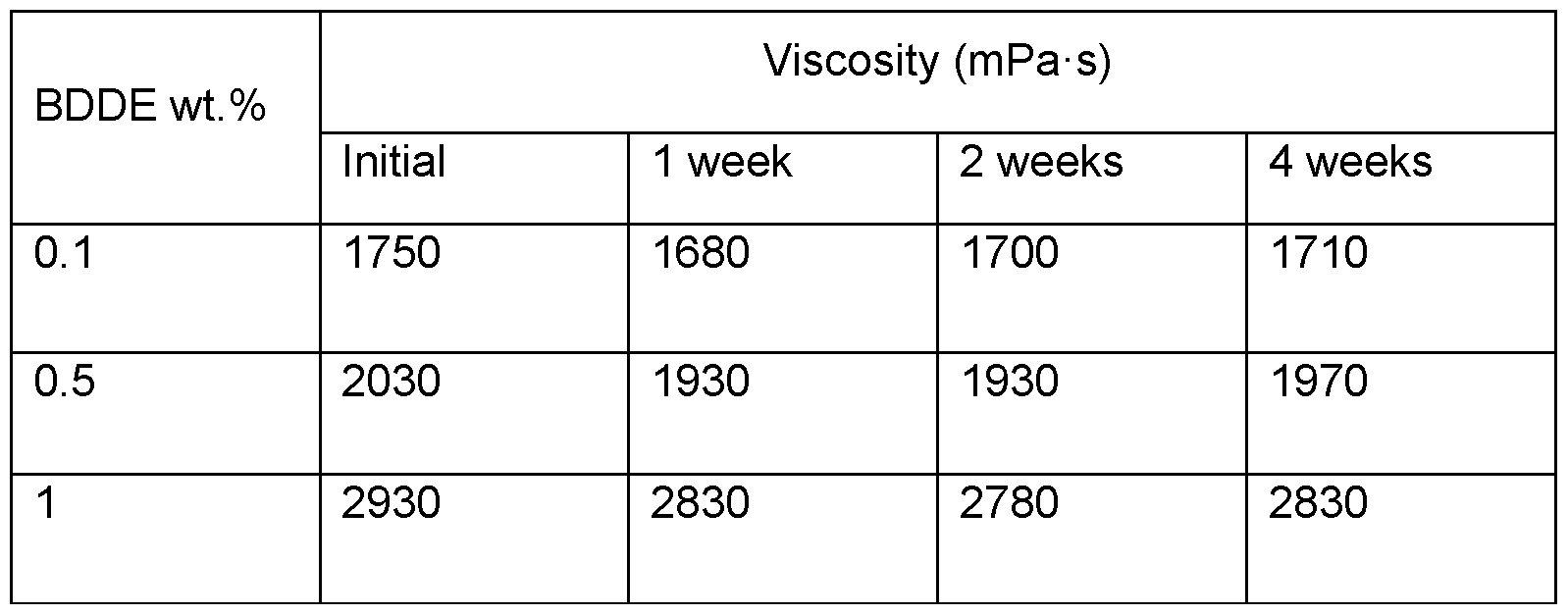
- Figure 2 Amplitude sweeps for (A) 0.5 wt.% dispersions and (B) 1.0 wt.% dispersions of Ci6-modified xanthan gum at various crosslink levels with 1 ,4-butanediol diglycidyl ether (BDDE). Weight of crosslinker expressed as wt.% based on the total weight of the unmodified xanthan gum.
- BDDE 1,4-butanediol diglycidyl ether
- Figure 3 Amplitude sweeps of 0.5 wt.% dispersions of: A) unmodified xanthan gum; B) crosslinked and hydrophobically modified xanthan gum; C) crosslinked xanthan gum; and D) xanthan gum that underwent the same reaction conditions without crosslinker or hydrophobe.
- Figure 4 Emulsions containing 0.5 wt.% of different samples of xanthan gum and 20 wt.% caprylic/capric triglyceride.
- FIG. 6 Amplitude sweeps for 0.5 wt.% dispersions containing crosslinked and hydrophobically modified xanthan gum at various molar substitution (MS) levels of Ci6. Xanthan gum was crosslinked with 0.5 wt.% 1 ,4-butanediol diglycidyl ether (BDDE).
- Figure 7. Microscope image showing oil drop size in cleansing formulations.
- Figure 8. Microscope image showing oil drop size in cleansing formulations.
- personal care includes, without being limited thereto, cosmetics, toiletries, cosmeceuticals, beauty aids, insect repellents, sunscreens, UV absorbers, hand sanitizers, personal hygiene and cleansing products (e.g., shampoos, conditioning shampoos, anti-dandruff shampoos, body washes, hand soaps, facial scrubs, and the like) applied to the body, including the skin, hair, scalp, and nails of humans and mammals
- the invention provides a crosslinked and hydrophobically modified xanthan gum, wherein a portion of the hydrogen atoms of pendant hydroxyl groups of the backbone are substituted with at least one hydrophobic moiety according to formula (I):
- A is selected from a divalent linear or branched, substituted or unsubstituted C1- C6 alkylene radical;
- R is selected from a hydrocarbon group having from 8 to 22 carbon atoms; a is 0 or 1 , and b is 0 or 1 , with the proviso that when a is 0, b is 0; and, wherein the xanthan gum is crosslinked with a multifunctional crosslinking agent comprising epoxy and/or alkyl halide functional groups in an amount of from 0.1 to 1 wt.% based on the total weight of the xanthan gum.
- the crosslinked and hydrophobically xanthan gum of the present invention can be produced from readily commercially available xanthan gum.
- Non-modified xanthan gum used as raw material is also herein referred as “native xanthan gum”, “unmodified xanthan gum” or generically as “xanthan gum”.
- Non-limiting examples of xanthan gum are commercially available from Jungbunzlauer, Fufeng, Jianlong or CP Kelco (under Keltrol® trademark).
- Xanthan gum is typically obtained by a fermentation process using Xanthomonas campestris bacteria. The molecular weight of native xanthan gum is typically in the range from (2 x 10 5 to 20 x 10 6 Da).
- the performance increasing coalescence and stability of emulsions, viscosity, and suitable texture of formulations provided by the crosslinked and hydrophobically modified xanthan gum of the present invention represents a balance between the hydrophobic modifications and crosslinking.
- A is selected from a divalent linear or branched, substituted or unsubstituted C1- C6 alkylene radical
- R is selected from a hydrocarbon group having from 8 to 22 carbon atoms
- a is 0 or 1
- b is 0 or 1 , with the proviso that when a is 0 b is 0.
- a is 1 and b is 1.
- the hydroxyl groups of each sugar unit of the xanthan gum can be hydrophobically derivatized with hydrophobic moieties according to formula (I).
- the modified xanthan can have a degree of molar substitution (MS) in a range from 0.001 to 0.5, particularly from 0.01 to 0.4, more particularly from 0.025 to 0.3 (mol I mol sug ar).
- the degree of molar substitution can be measured according to the techniques well known to a person skilled in art, such as, for example, performing hydrolysis following by gas chromatography.
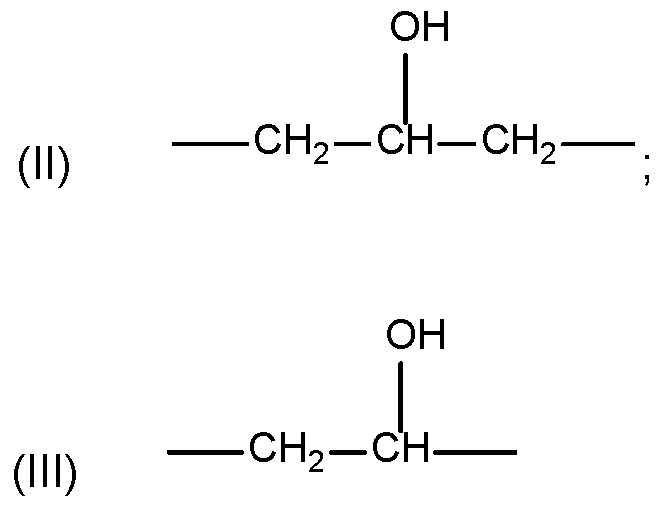
- the divalent alkylene radical A can particularly be represented by formula (II) or (III): [0040] More particularly, the divalent alkylene radical A can be an alkylene radical according to formula (II).
- the hydrocarbon group can be a saturated or unsaturated alicyclic group, a saturated or unsaturated aliphatic group, or an aromatic group having from 8 to 22 carbon atoms.
- the hydrocarbon group can be linear or branched.
- the hydrocarbon group can particularly be a linear hydrocarbon group, i.e., the hydrocarbon group is not branched.
- the hydrocarbon group can particularly be a saturated hydrocarbon group. In one embodiment, the hydrocarbon group is a linear saturated hydrocarbon group.
- hydrocarbon groups having from 12 to 16 carbon atoms, which provide better coalescence ratings when the modified xanthan gum is used in emulsions.
- the hydrocarbon group is a linear saturated hydrocarbon group having from 12 to 16 carbon atoms, particularly 16 carbon atoms.
- the hydrogen atoms of the pendent groups of the xanthan gum are substituted with at least one hydrophobic moiety according to the following formula (IV): wherein n is an integer from 7 to 21 , particularly from 11 to 15. More particularly, n is 15.
- the hydrophobic modification entails alkylation by reacting pendant hydroxyl groups present in the backbone of xanthan gum or derivative thereof with an alkylation agent. Without being bound to the theory it is believed that alkylation agent reacts with the hydrogen atoms of the C2, C3, C4, and/or the C6 hydroxyls of the xanthan gum.
- the alkylating agent can react with the hydroxyl groups on the C-2, C-3, and/or C-6 carbon atoms of the glucose units, and/or the C-2 and/or C-3 carbon atoms of the glucuronic acid units, and/or the C3 and C4 carbon atoms of the mannose units of the xanthan gum.
- the hydrogen atoms of the pendent hydroxyl groups of the xanthan gum that are substituted with at least one hydrophobic moiety can be the hydrogen atoms of the C-2, C-3, and/or C-6 hydroxyls of the glucose units, and/or the C-2 and/or C-3, hydroxyls of the glucuronic acid units, and/or the C2, C3, C4 and/or C6 hydroxyls of the mannose units of the xanthan gum. While not wishing to be bound by any theory, it is contemplated that probable reaction can occur at one or more positions.
- the hydrogen atoms of the hydroxyl groups of the xanthan gum that are substituted with at least one hydrophobic moiety are the hydrogen atoms of at least the C-6 hydroxyls of the D-glucose units.
- an “alkylating” or “alkylation agent” is a reactive compound containing a hydrocarbon group that can be reacted with a pendant hydroxyl group on the xanthan gum to form an ether linkage with the xanthan gum as the starting material.
- Typical alkylation agents reactive with the pendant xanthan gum hydroxyl groups include alkyl halides, epoxides, and glycidyl ethers that contain a hydrocarbon group.
- the alkylation agent can be an alkyl halide represented by formula (V) below: (V) X-R 1 wherein X is a halogen atom selected from the group consisting of bromine, chlorine, fluorine, and iodine; and R 1 is a hydrocarbon group selected from cycloalkyl, linear or branched alkyl, cycloalkenyl, linear or branched alkenyl, aryl, alkylaryl, alkenylaryl, and combinations thereof, wherein the hydrocarbon group contains from 8 to 22 carbon atoms.
- V alkyl halide represented by formula (V) below: (V) X-R 1 wherein X is a halogen atom selected from the group consisting of bromine, chlorine, fluorine, and iodine; and R 1 is a hydrocarbon group selected from cycloalkyl, linear or branched alkyl, cycloalkenyl, linear or branched alkenyl, aryl,
- alkyl halide alkylating agents include, but are not limited to, octyl, decyl, dodecyl, myristyl, hexadecyl, stearyl and behenyl bromides, fluorides, chlorides, and iodides.
- the alkylating agent can be an alkyl halide wherein the alkyl group is a linear or branched C8 to C22 alkyl group, particularly a C12 to C16 alkyl group, more particularly a C16 alkyl group.
- the alkyl halide is an alkyl halide wherein the alkyl group is a linear C8 to C22 alkyl group, particularly a linear C12 to C16 alkyl group, more particularly a linear C16 alkyl group.
- the halide can be chloride or bromide, more particularly can be a chloride.
- the alkyl halide is selected from the group consisting of dodecyl, myristyl and hexadecyl chloride.
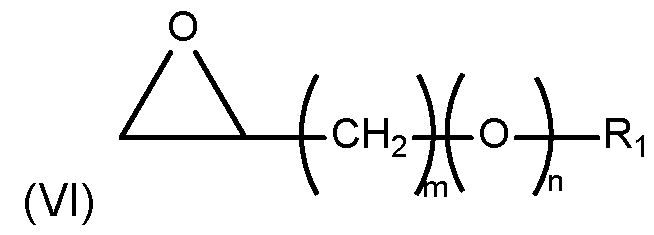
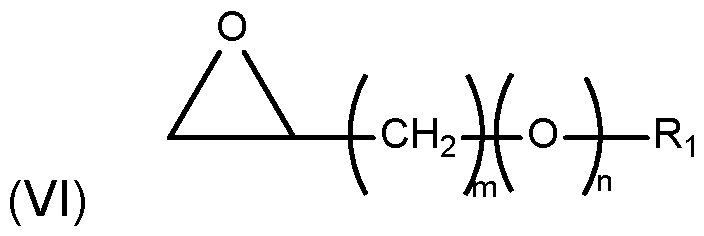
- alkylation agents Of particular interest in the context of this invention as alkylation agents are glycidyl ethers represented by formula (VI) as follows:
- n is 0 or 1
- Ri is selected from a hydrocarbon group having from 8 to 22 carbon atoms.
- said hydrocarbon group can have from 12 to 16 carbon atoms.
- the hydrocarbon group can particularly be a saturated hydrocarbon group, more particularly a saturated linear hydrocarbon group.
- Representative glycidyl ethers set forth under formula (VI) useful as alkylation agents include but are not limited to 2-(1 ,1-dimethylethyl)-2-(phenylmethyl)-oxirane, 2-[2- (4-chlorophenyl)ethyl]-2-(1 ,1-dimethylethyl)-oxirane, 1 ,2-epoxyoctane, 1 ,2-epoxydecane, 1 ,2-epoxy dodecane, 1 ,2-epoxytetradecane, 1 ,2-epoxy hexadecane, 1 ,2- epoxyoctadecane, 1 ,2-epoxyeicosane, 1 ,2-epoxyethylbenzene, 2-ethylhexyl glycidyl ether, dodecyl glycidyl ether, tetradecyl glycidyl ether, hex
- the alkylation agent is a glycidyl ether represented by formula (VI) wherein m is 1 and n is 1 and Ri is a hydrocarbon group having from 8 to 22 carbon atoms, particularly from 12 to 16 carbon atoms.
- the hydrocarbon group is a linear saturated hydrocarbon group.
- the glycidyl ether is selected from the group consisting of dodecyl glycidyl ether, tetradecyl glycidyl ether and hexadecyl glycidyl ether. More particularly, the alkylation agent is hexadecyl glycidyl ether.
- the xanthan gum according to the invention is crosslinked with a multifunctional crosslinking agent comprising epoxy and/or alkyl halide functional groups.
- the amount of crosslinking agent can be from 0.1 to 1 wt.% or from 0.2 to 1 wt. %, particularly from 0.2 to 0.9 wt.%, more particularly from 0.4 to 0.6 wt.% based on the total weight of the xanthan gum.
- the multifunctional crosslinking agent can be a homo-bifunctional crosslinking agent or hetero-bifunctional crosslinking agent.
- the multifunctional crosslinking agent can be selected from the group consisting of polyglycidyl ethers, multifunctional alkyl halides and epihalohydrin.
- Suitable polyglycidyl ethers as crosslinking agents in the context of this invention include, but are not limited to, glycerol diglycidyl ether, polyethylene glycol diglycidyl ether, polyoxyethylene glycol diglycidyl ether, neopentyl glycol diglycidyl ether, 1 ,4-butanediol diglycidyl ether (BDDE), 1 ,6-hexanediol diglycidyl ether, glycerol diglycidyl ether, trimethylolpropane diglycidyl ether, trimethylolpropane triglycidyl ether, glycerol triglycidyl ether, pentaerythritol diglycidyl ether, pentaerythritol triglycidyl ether, pentaerythritol tetraglycidyl ether, sorbitol dig
- the polyglycidyl ether can be a diglycidyl ether. More particularly, the polyglycidyl ether can be 1,4-butanediol diglycidyl ether (BDDE).
- BDDE 1,4-butanediol diglycidyl ether
- the crosslinking agent can be polypropylene glycol) diglycidyl ether.
- Suitable alkyl halides as multifunctional crosslinking agents in the context of the present invention include, but are not limited to, ethyl, propyl, isopropyl, n-butyl, t- butyl, pentyl, neopentyl, hexyl, octyl, decyl, dodecyl, myristyl, hexadecyl, stearyl and behenyl dibromides, dichlorides, and di-iodides.
- the crosslinking agent can be a multifunctional alkyl halide wherein the alkyl group is a linear or branched C2 to C22 alkyl group.
- the crosslinking agent can be 1 ,4-dichorobutane.
- the multifunctional crosslinking agent is an epihalohydrin, more particularly an epichlorohydrin.
- the alkylating agent and the crosslinker will be capable to react with the hydroxyl groups present in the saccharide units after deprotonating with an alkali agent.
- the alkylating agent and the crosslinker can react with the hydroxyl groups on the C-2, C-3, and/or C-6 carbon atoms of the glucose units, and/or the C-2 and/or C- 3, carbon atoms of the glucuronic acid chain units, and/or the C2, C3, 04, and/or 06 carbon atoms of the mannose chain units of the xanthan gum. While not wishing to be bound by any theory, it is contemplated that probable reaction can occur at one or more positions.
- the invention also provides a method for preparing a crosslinked and hydrophobically modified xanthan gum described herein comprising reacting a xanthan gum with a) an alkylating agent and b) a multifunctional crosslinker comprising epoxide and/or alkyl halide functional groups.
- the crosslinking and hydrophobic modification can be conducted in any order, or simultaneously, as well as repeated, to produce the desired crosslinked and hydrophobically modified xanthan gum.
- one of the advantages of the present invention is that the crosslinking and hydrophobic modifications are performed simultaneously, i.e., the reaction with the alkylating agent and the multifunctional crosslinker is conducted simultaneously.
- the method for preparing the crosslinked and hydrophobically modified xanthan gum can comprise the steps of: i) Mixing a xanthan gum with an organic solvent or a mixture of an organic solvent and water to form a suspension or slurry; ii) Increase the pH value of the suspension or slurry to be higher than 9 with an alkaline solution; iii) Adding an alkylating agent and a multifunctional crosslinker comprising epoxide and/or alkyl halide functional groups; iv) Reacting said reaction mixture for at least 2h at a temperature from 50 to 100°C to obtain a crosslinked and hydrophobically modified xanthan gum; v) Neutralizing the crosslinked and hydrophobically modified xanthan gum with an acid.
- the xanthan gum used as starting material is typically in powder form and is normally treated with an organic solvent or a mixture of organic solvent and water thereby obtaining a suspension or slurry.
- the amount of xanthan gum in the organic solvent or the mixture of organic solvent and water i.e., the suspension or slurry
- the organic solvent used can be acetone, methanol, ethanol, isopropanol, n-propyl alcohol, n-butyl alcohol, isobutyl alcohol, t-butyl alcohol, or the like, and mixtures thereof.
- the organic solvent is isopropanol.
- the amount of organic solvent when combined with water can be from 50 to 80 wt.%, more particularly from 55 to 75 wt.%.
- the organic solvent combined with water can comprise from 50 to 80 wt.%, particularly from 55 to 75 wt.%, more particularly 60 wt.% of isopropanol in water.
- concentrations in the range from 55 to 65 wt.% allow to simultaneously add the alkylating agent and the multifunctional crosslinker, while controlling particle swelling, which affect the extent and location of modifications.
- the pH in step ii) is increased to be 10 or higher, particularly from 10 to 13, particularly from 11 to 13.
- the pH is typically adjusted with an alkali such as sodium hydroxide, potassium hydroxide, calcium hydroxide, lithium hydroxide, and ammonium hydroxide, for example.
- the alkali is sodium hydroxide.
- the amount of alkali used is the amount necessary for increasing the pH to the desired values.
- the mixture can be allowed to react from 2h to 5h, or from 2h to 4h, or preferably for 3h.
- the reaction temperature can particularly be from 55 to 65 °C.
- the reaction temperature can be 60°C.
- the mixture is allowed to react for from 2h to 4h at 55 to 65°C.
- the reaction can be conducted in the presence of nitrogen atmosphere.
- the modified xanthan gum is neutralized.
- the neutralization is normally adjusted to a pH which is within a range of from about 4 to about 10, or from about 6 to about 8, or about 7.
- Any acid may be selected to neutralize the solution, including strong acids such as hydrochloric acid and sulfuric acid or weak acids such as acetic acid, citric acid, carbon dioxide (carbonic acid), trifluoroacetic acid, etc.
- strong acids such as hydrochloric acid and sulfuric acid or weak acids
- acetic acid citric acid, carbon dioxide (carbonic acid), trifluoroacetic acid, etc.
- carbon dioxide carbon dioxide
- trifluoroacetic acid etc.
- hydrochloric or acetic acid is used.
- the amount of acid used is the amount necessary for neutralization.
- the modified xanthan gum obtained after reaction in the presence of alkylating agent and a multifunctional crosslinker can be subsequently separated from the solvent, in particular by physical separation methods.
- separating techniques include filtering, centrifuging and the like.
- the solvent can be removed by filtration.
- the remaining solid can be further washed with an organic solvent or a mixture of organic solvent and water.
- the remaining solid is washed with a mixture of organic solvent and water comprising from 60 to 90 wt. %, or from 75 to 85 wt.%, or about 80 wt.% of organic solvent in water.
- Non-limiting organic solvents that can be used are acetone, methanol, ethanol, isopropanol, n-propyl alcohol, n-butyl alcohol, isobutyl alcohol, t-butyl alcohol, and the like. Mixtures of these alcohols can also be used. Preferably, isopropanol in water is used.
- the volume of the wash liquid is much greater than the amount of modified xanthan and can be performed in batch wise or multiple applications. Typically, 1 to 4 independent wash cycles are completed. However, additional wash cycles can be used if needed.
- the modified xanthan gum can be dried either directly after separating the xanthan gum from the reaction medium or subsequent washing to remove the remaining solvent.
- drying techniques include air drying, evaporative drying, vacuum drying, freeze drying, fluidized bed and the like.
- drying at temperatures from 50 to 115°C under ambient conditions or reduced pressure can be advantageous.
- the drying temperature can range from 60 to 80°C. It has been surprisingly found that drying at temperatures in the range from 60 to 80°C improves the viscosity, yield values and stability of formulations and compositions comprising the modified xanthan gum according to the invention. More particularly, the temperature can be 60, 70 or 80°C.
- the drying times can range from 1 to 4 hours.
- the drying step is conducted at 80°C for 4 hours, particularly under vacuum conditions.
- the crosslinked and hydrophobically modified xanthan gum of the present invention can be formulated in compositions for personal care products, topical health care products, household care products, institutional and industrial (l&l) products, and fabric care products.
- one object of the present invention is the provision of an oil-in- water emulsion comprising: a water phase; an oil phase dispersed in said water phase; and, a crosslinked and hydrophobically modified xanthan gum according to the present invention.
- the invention also provides a method of stabilizing an oil-in-water emulsion comprising an oil phase and a water phase comprising the step of mixing the crosslinked and hydrophobically modified xanthan gum according to the present invention with the oil phase and the water phase.
- the invention also provides for the use of a crosslinked and hydrophobically modified xanthan gum according to the invention to stabilize an emulsion comprising an oil phase and a water phase.
- oil means a compound or a mixture of compounds that is insoluble in water and has a liquid appearance at a temperature of 25°C.
- the oil phase is not restricted to a specific oil. Any oil suitable for cosmetic or personal care formulations can be used, including but not limited to, vegetable oils, oils derived from petroleum (e.g., mineral oils, liquid paraffin), fatty esters, etc.
- the oil phase may comprise at least one of an ester oil, vegetable oil, alcohol, paraffin oil or silicone.
- the oil phase can comprise one or more oils chosen from mineral oils such as paraffin oil, petroleum jelly, isoparaffins or white mineral oils; oils of animal origin, such as squalene or squalane; vegetable oils, such as phytosqualane, sweet almond oil, coconut oil, castor oil, jojoba oil, olive oil, rapeseed oil, peanut oil, sunflower oil, wheat germ oil, corn germ oil, soybean oil, cottonseed oil, alfalfa oil, poppy oil, pumpkin oil, evening primrose oil, millet oil, barley oil, rye oil, safflower oil,nadooulier oil, passionflower oil, hazelnut oil, palm oil, shea butter, apricot kernel oil, coriander seed oil, beech oil, calophyllum oil, sisymbrium oil, avocado oil, calendula oil, oils from flowers or vegetables ethoxylated vegetable oils, algae
- the oil phase comprises at least an oil selected from the group consisting of fatty acid esters, monoglycerides, diglycerides and triglycerides of fatty acids and mixtures thereof.
- the fatty acid ester can be particularly selected from the group consisting of butyl myristate, propyl myristate, isopropyl myristate, cetyl myristate, isopropyl palmitate, octyl palmitate, butyl stearate, hexadecyl stearate, isopropyl stearate, isopropyl isostearate, octyl stearate, isocetyl stearate, dodecyl oleate, hexyl laurate, propylene glycol dicaprylate, and esters derived from lanolic acid, such as isopropyl lanolate, isocetyl lanolate, and combinations thereof. More particularly, the fatty acid ester can be isopropyl stearate, isopropyl isostearate.
- the oil phase can comprise an oil selected from the group consisting of triglycerides of fatty acids, isostearyl isostearate, and isohexadecane, and combinations thereof. More particularly, the triglyceride of fatty acid can be caprylic/capric triglyceride or triolein.
- the oil phase may be present in an amount of from 1 to 50 wt.% based on the total weight of the emulsion.
- the oil phase can be present from 1 to 40 wt.%, particularly 5 to 40 wt.%, more particularly from 10 to 40 wt.%, even more particularly from 20 to 30 wt.% based on the total weight of the emulsion.
- the oil phase can also contain other components that are oil soluble.
- the oil phase can comprise at least 90 wt.%, at least 95 wt.%, or at least 98 wt.%, of one or more than one oils, based on the total weight of the oil phase.
- the emulsion of the invention comprises a water phase, which constitutes the continuous phase.
- the water phase comprises water and can contain other components that are water soluble.
- the water phase can comprise at least 90 wt.%, at least 95 wt.%, or at least 98 wt.% water, based on the total weight of the water phase.
- the modified xanthan gum of the present invention is in an amount ranging from 0.01 to 2 wt.% based on the total weight of the emulsion.
- the modified xanthan gum can be in a concentration from 0.05 to 2 wt.%, or from 0.1 to 1.5 wt.%, or from 0.2 to 1 .5 wt.%, or from 0.5 to 1 .25 wt.% of the based on the total weight of the emulsion.
- the emulsion can further comprise co-emulsifiers or other surfactants.
- HLB value Hydrophilic Lipophilic Balance
- the HLB value is equal or higher than 7, or is equal or higher than 8, or is equal or higher than 10.
- the co-emulsifier can be selected from the group consisting of amino acid-based surfactants and (poly)glyceryl fatty acids. More particularly, the coemulsifier is selected from the group consisting of sodium stearoyl glutamate, polyglyceryl-3 Laurate, and polyglyceryl- 10 Laurate.
- the concentration of the co-emulsifier is in the range from 0.1 to 1 wt.% based on the total weight of the emulsion.
- the viscosity of the emulsions according to the invention as determined as Brookfield viscosity at 20 rpm at room temperature is in the range from 1 ,000 to 9,000 mPa s.
- the invention also provides formulations or compositions for personal care and topical health care comprising the modified xanthan gum of the present invention or the oil-in-water emulsions comprising the modified xanthan gum of the present invention.
- the invention also provides cleaning compositions comprising the crosslinked and hydrophobically modified xanthan gum of the present invention.
- Typical cleaning compositions include household and industrial and institutional (l&l) cleaning compositions, formulations and products that can contain the modified xanthan gum of the present invention.
- Non-limiting examples include surface cleansers for kitchen and bathroom counter tops, tiled surfaces, and utilities, including appliances employed or located therein, toilet cleaners, including toilet bowl rim gels, floor cleansers, wall cleansers, polishes, air freshener gels, detergents, treatments and cleansers for dishes.
- toilet cleaners including toilet bowl rim gels, floor cleansers, wall cleansers, polishes, air freshener gels, detergents, treatments and cleansers for dishes.
- the invention also provides fabric care compositions comprising the crosslinked and hydrophobically modified xanthan gum of the present invention.
- fabric care compositions include compositions and formulations designed for treating fabric.
- Such compositions include, but are not limited to, laundry cleaning compositions and detergents, fabric softening compositions, fabric enhancing compositions, fabric freshening compositions, laundry prewash, laundry pretreat, laundry additives, dry cleaning agent or composition, laundry rinse additive, wash additive, post-rinse fabric treatment, and the like.
- Such compositions may be used as a pre-laundering treatment, a post-laundering treatment, or may be added during the rinse or wash cycle of the laundering operation.
- compositions for personal care and topical health care can comprise any cosmetic, toiletry, and topical pharmaceutical formulation.
- Typical personal care formulations that can include the modified xanthan gum according to the present invention include, without being limited thereto, shampoos, chemical and non-chemical hair curling and hair straightening products, hair style maintenance products (e.g., hair fixatives and hair color products), emulsion lotions and creams for the nails, hands, feet, face, scalp, and body, hair dyes, face and body makeup, nail care products, astringents, deodorants, antiperspirants, depilatories, skin protective creams and lotions, such as sunscreens, skin and body cleansers, skin conditioners, skin toners, skin firming compositions, liquid soaps, soap bars, bath products, shaving products, and the like.
- hair style maintenance products e.g., hair fixatives and hair color products
- emulsion lotions and creams for the nails, hands, feet, face, scalp, and body hair dyes, face and body
- compositions for personal care and topical health care can be in the form of, without being limited thereto, liquids, such as rinses, gels, hydroalcoholic gels (e.g., hand sanitizers), sprays, emulsions, such as lotions and creams, shampoos, pomades, foams, ointments, tablets, sticks, such as lip care products, makeup, and suppositories, and like products, which are applied to skin and hair and remain in contact therewith until removed as by rinsing with water or washing with shampoo or soap.
- Gels can be soft, stiff, or squeezable.
- the modified xanthan gum of the present invention is particularly suitable for body washes, antibacterial body washes, dual purpose body washes and shampoos, bath gels, shower gels, liquid hand soaps, body scrubs, bubble baths, facial scrubs, foot scrubs, and the like.
- the shampoo embodiments of the present invention can be formulated as 2-in-1 shampoos, baby shampoos, conditioning shampoos, bodyfying shampoos, moisturizing shampoos, temporary hair color shampoos, 3-in-1 shampoos, anti-dandruff shampoos, hair color maintenance shampoos, acid (neutralizing) shampoos, medicated shampoos, and salicylic acid shampoos, and the like.
- modified xanthan gum of the present invention that can be employed in the foregoing compositions can be determined by the person skilled in the formulation art.
- the modified xanthan gum is typically used in a concentration from 0.001 to 10 wt.%, or from 0.005 to 5 wt.%, or from 0.01 to 2 wt.%, 0.1 to 1.5 wt.%, or from 0.2 to 1 wt.%. or from 0.3 to 0.8 wt.% based on the total weight of the personal care composition.
- compositions, formulations and products of the invention can typically contain various additives and conventional adjuvants as are well known in the art, including, without being limited thereto, acidifying or alkalizing pH adjusting agents and buffering agents; fixatives and auxiliary film formers, such as gums, resins, polymers of synthetic or natural origin, and the like; auxiliary rheology modifiers, such as viscosity increasing polymeric thickeners or gellants, additives, such as emulsifiers, emulsion stabilizers, waxes, dispersants, and the like, and viscosity control agents, such as solvents, electrolytes, and the like; hair and skin conditioning agents, such as antistatic agents, synthetic oils, vegetable or animal oils, silicone oils, monomeric or polymeric quaternized ammonium salts, emollients, humectants, lubricants, sunscreen agents, disinfectants, antimicrobials, and the like; chemical hair waving or straightening agents; hair colorants,
- compositions comprising the modified xanthan gum according to the present invention can further include alkaline or acidifying compounds for adjusting the pH (i.e. , pH adjusting materials).
- Buffering agents can be used in the exemplary compositions. Suitable buffering agents include alkali or alkali earth metal carbonates, phosphates, bicarbonates, citrates, borates, acetates, acid anhydrides, succinates, and the like, such as sodium phosphate, sodium citrate, sodium acetate, sodium bicarbonate, and sodium carbonate. [00106] The amount of buffering agent utilized is dependent upon the desired pH range to be buffered or maintained. Such amounts can readily be determined by the skilled formulator.
- Surfactants can be used in exemplary compositions of the invention.
- Suitable anionic surfactants include but are not limited to alkyl sulfates, alkyl ether sulfates, alkyl sulfonates, alkaryl sulfonates, a-olefin-sulfonates, alkylamide sulfonates, alkarylpolyether sulphates, alkylamidoether sulfates, alkyl monoglyceryl ether sulfates, alkyl monoglyceride sulfates, alkyl monoglyceride sulfonates, alkyl succinates, alkyl sulfosuccinates, alkyl ether sulfosuccinates, alkyl sulfosuccinamates, alkyl amidosulfosuccinates; alkyl sulfoacetates, alkyl phosphates, alkyl ether phosphate
- anionic surfactants also include the sodium, potassium, lithium, magnesium, and ammonium salts of laureth sulfate, trideceth sulfate, myreth sulfate, C12-C13 pareth sulfate, C12-C14 pareth sulfate, and C12- C15 pareth sulfate, ethoxylated with 1 , 2, and 3 moles of ethylene oxide; the sodium, potassium, lithium, magnesium, ammonium, and triethanolammonium salts of lauryl sulfate, coco sulfate, tridecyl sulfate, myristyl sulfate, cetyl sulfate, cetearyl sulfate, stearyl sulfate, oleyl sulfate, and tallow sulfate, disodium lauryl sulfosuccinate, disodium laureth sulfosuccinate, sodium cocoyl
- compositions in the context of the present invention can include amino acid surfactants.
- Suitable amino acid surfactants include but are not limited to taurates, glutamates, alanine, alaninates, sacosinates, aspartates, glycinates, and mixtures thereof.
- Suitable cationic surfactants of the present technology include but are not limited to alkylamines, amidoamines, alkyl imidazolines, ethoxylated amines, quaternary compounds, and quaternized esters.
- alkylamine oxides can function as a cationic surfactant at a lower pH value.
- Suitable zwitterionic or amphoteric surfactants according to the invention include, but are not limited to, betaine-based surfactants, sultaine-based surfactants, amphocarboxylate-based surfactants, amine oxides and combinations thereof.
- Suitable nonionic surfactants in the context of the present invention include, but are not limited to, aliphatic (Ce-Cis) primary or secondary linear or branched chain acids, alcohols or phenols; alkyl ethoxylates; alkyl phenol alkoxylates (especially ethoxylates and mixed ethoxy/propoxy moieties); block alkylene oxide condensates of alkyl phenols; alkylene oxide condensates of alkanols; and ethylene oxide/propylene oxide block copolymers.
- nonionic surfactants include mono- or dialkyl alkanolamides; alkyl polyglucosides (APGs); sorbitan fatty acid esters; polyoxyethylene sorbitan fatty acid esters; polyoxyethylene sorbitol esters; polyoxyethylene acids, and polyoxyethylene alcohols.
- nonionic surfactants include coco mono- or diethanolamide, coco glucoside, decyl diglucoside, lauryl diglucoside, coco diglucoside, polysorbate 20, 40, 60, and 80, ethoxylated linear alcohols, cetearyl alcohol, lanolin alcohol, stearic acid, glyceryl stearate, PEG-100 stearate, laureth 7, and oleth 20.
- Non-ionic surfactants can include, but are not limited to, alkoxylated methyl glucosides such as, for example, methyl gluceth-10, methyl gluceth-20, PPG-10 methyl glucose ether, and PPG-20 methyl glucose ether, available from Lubrizol Advanced Materials, Inc., under the trade names, Glucam® E10, Glucam® E20, Glucam® P10, and Glucam® P20, respectively; and hydrophobically modified alkoxylated methyl glucosides, such as PEG 120 methyl glucose dioleate, PEG-120 methyl glucose trioleate, and PEG-20 methyl glucose sesquistearate, available from Lubrizol Advanced Materials, Inc., under the trade names, Glucamate® DOE- 120, GlucamateTM LT, and GlucamateTM SSE-20, respectively, are also suitable.
- Other exemplary hydrophobically modified alkoxylated methyl glucosides are disclosed in United States Patent Nos. 6,57
- surfactants may be in a concentration of from 0.1 to 50 wt.%, or from 0.1 to 30 wt.%, or from 0.5 to 26 wt.%, or from 1 to 22 wt.%, or from 5 to 18 wt.%, or from 7 to 12 wt.% based on the total weight of the personal care composition.
- compositions in the context of the present invention can also include cationic polymers.
- Cationic polymers are components that can enhance the delivery and deposition of conditioning agents and/or provide auxiliary conditioning benefits to the hair, scalp, or skin to improve and enhance the conditioning benefits delivered by the compositions of the present technology.
- Cationic polymer refers to polymers containing at least one cationic moiety or at least one moiety that can be ionized to form a cationic moiety.
- these cationic moieties are nitrogen containing groups such as quaternary ammonium or protonated amino groups.
- the cationic protonated amines can be primary, secondary, or tertiary amines.
- the cationic polymer typically has a cationic charge density ranging from about 0.2 to about 7 meq/g at the pH of the intended use of the composition.
- the average molecular weight of the cationic polymer ranges from about 5,000 daltons to about 10,000,000 daltons.
- Non-limiting examples of such polymers are described in the CTFA International Cosmetic Ingredient Dictionary/Handbook via the CTFA website.
- Suitable cationic polymers can be synthetically derived or natural polymers that are synthetically modified to contain cationic moieties. Particularly, the cationic polymer contains at least one repeating unit containing a quaternary ammonium salt moiety. Preparation of quaternary ammonium salt moiety containing polymers can be found, for example, in U.S. Patent. Nos. 3,288,770; 3,412,019; 4,772,462 and 5,275,809.
- Suitable cationic polymers include the chloride salts of the foregoing quaternized homopolymers and copolymers in which the alkyl group is methyl or ethyl, and are commercially available under the Merquat® series of trademarks from Lubrizol Advanced Materials, Inc.
- a copolymer prepared from DADMAC and acrylamide having the CTFA name, Polyquaternium-7, is sold under the MerquatTM 550 trademark.
- Another copolymer prepared from DADMAC and acrylic acid having the CTFA name, Polyquaternium-22 is sold under the Merquat280 trademark.
- ampholytic terpolymer prepared from a nonionic component derived from acrylamide or methyl acrylate, a cationic component derived from DADMAC or methacrylamidopropyl trimethyl ammonium chloride (MAPTAC), and an anionic component derived from acrylic acid or 2-acrylamido-2-methylpropane sulfonic acid or combinations of acrylic acid and 2-acrylamido-2-methylpropane sulfonic acid.
- An ampholytic terpolymer prepared from acrylic acid, DADMAC and acrylamide having the CTFA name, Polyquarternium-39, is available under the MerquatTM Plus 3330 and MequatTM 3330PR trademarks.
- MAPTAC methacrylamidopropyl trimethyl ammonium chloride
- acrylamide having the CTFA name, Polyquarternium-53, is available under the MerquatTM 2003PR trademark.
- Exemplary cationically modified natural polymers include polysaccharide polymers, such as cationically modified cellulose and cationically modified starch derivatives modified with a quaternary ammonium halide moiety.
- Exemplary cationically modified cellulose polymers are salts of hydroxyethyl cellulose reacted with trimethyl ammonium substituted epoxide (CTFA, Polyquaternium-10).
- CTFA trimethyl ammonium substituted epoxide
- Other suitable types of cationically modified cellulose include the polymeric quaternary ammonium salts of hydroxyethyl cellulose reacted with lauryl dimethyl ammonium substituted epoxide (CTFA, Polyquaternium-24).
- Cationically modified potato starch having the CTFA name, Starch Hydroxypropyltrimonium Chloride, is available under the SensomerTM CI-50 trademark, from Lubrizol Advanced Materials, Inc.
- Suitable cationically modified natural polymers include cationic polygalactomannan derivatives such as guar gum derivatives and cassia gum derivatives, e.g., CTFA: Guar Hydroxypropyltrimonium Chloride and Cassia Hydroxypropyltrimonium Chloride.
- Guar hydroxypropyltrimonium chloride is commercially available under the JaguarTM trade name series from Rhodia Inc. and the N-Hance trade name series from Ashland Inc.
- Cassia Hydroxypropyltrimonium Chloride is commercially available under the SensomerTM trademark from Lubrizol Advanced Materials, Inc.
- the cationic and ampholytic polymers can be present from about 0.05 to about 5 wt.% percent, or from about 0.1 to about 3 wt.%, or from about 0.5 to about 2.0 wt.%, based on the total weight of the composition.
- Silicone conditioning agents can be used in compositions employing the modified xanthan gum of the present invention.
- the optional silicone conditioning agent may comprise a volatile silicone, a nonvolatile silicone, or combinations thereof.
- Water soluble or water dispersible silicones can also be used in the disclosed technology. Such water-soluble silicones contain suitable anionic functionality, cationic functionality, and/or nonionic functionality to render the silicone water soluble or water dispersible.
- the water-soluble silicones can contain a polysiloxane main chain to which is grafted at least one anionic moiety.
- Watersoluble silicone copolyol are also useful in the practice of the disclosed technology.
- Dimethicone copolyols are disclosed in U.S. Patent Nos. 5, 136,063 and 5, 180,843, the disclosures of which are incorporated herein by reference.
- dimethicone copolyols are commercially available under the Silsoft® and Silwet® brand names from the General Electric Company (GE-OSi). Specific product designations include but are not limited to Silsoft 305, 430, 475, 810, 895, Silwet L 7604 (GE-OSi); Dow Corning® 5103 and 5329 from Dow Corning Corporation; and Abil® dimethicone copolyols, such as, for example WE 09, WS 08, EM 90 and EM 97 from Evonik Goldschmidt Corporation; and SilsenseTM dimethicone copolyols, such as SilsenseTM Copolyol-1 and SilsenseTM Copolyol-7, available from Lubrizol Advanced Materials, Inc.
- Silsoft® and Silwet® brand names from the General Electric Company (GE-OSi). Specific product designations include but are not limited to Silsoft 305, 430, 475, 810, 895, Silwet L 7604 (GE-OSi); Dow
- compositions of the present technology can also include nature derived emollients to be used as a silicone alternative to cyclopentasiloxane D5 such as C9-C12 alkanes available from Lubrizol Advanced Materials, Inc. under the trademark SilsenseTM Bio 5 Emollient.
- nature derived emollients to be used as a silicone alternative to cyclopentasiloxane D5 such as C9-C12 alkanes available from Lubrizol Advanced Materials, Inc. under the trademark SilsenseTM Bio 5 Emollient.
- the concentration of the silicone conditioning agent or silicone alternatives can range from about 0.01 % to about 20%, or about 0.05% to about 10%, from about 0.1 % to about 5%, or from about 0.2% to about 3% by wt. all based on the total weight of the composition.
- conditioning oils for use as conditioning agents include, but are not limited to, hydrocarbon oils having at least about 10 carbon atoms, such as cyclic hydrocarbons, straight chain aliphatic hydrocarbons (saturated or unsaturated), and branched chain aliphatic hydrocarbons (saturated or unsaturated), including polymers and mixtures thereof.
- Hydrocarbon oils typically contain about 12 to 19 carbon atoms.
- Branched chain hydrocarbon oils, including hydrocarbon polymers typically will contain more than 19 carbon atoms.
- Natural oil conditioners are also useful in the practice of the present invention technology, and include, but are not limited to, peanut, sesame, avocado, coconut, cocoa butter, almond, safflower, corn, cotton seed, sesame seed, walnut oil, castor, olive, jojoba, palm, palm kernel, soybean, wheat germ, linseed, sunflower seed; eucalyptus, lavender, vetiver, litsea, cubeba, lemon, sandalwood, rosemary, chamomile, savory, nutmeg, cinnamon, hyssop, caraway, orange, geranium, cade, and bergamot oils, fish oils, glycerol tricaprocaprylate; and mixtures thereof.
- the natural oils can also be utilized as emollients.
- Natural and synthetic wax conditioning agents can be employed in the compositions of the disclosed technology, including but are not limited to carnauba wax, carnauba acid wax, hydrolyzed carnauba wax, ethoxylated carnauba wax (e.g., PEG-12 carnauba wax), candelilla wax, hydrolyzed candelilla wax, clover wax, alfalfa wax, paraffin wax, ozokerite wax, olive wax, rice wax, hydrogenated castor wax, bayberry wax, hydrogenated jojoba wax, bees wax, modified bees wax, e.g., cera bellina wax, ethoxylated beeswax (e.g., PEG-6 beeswax, PEG-8 beeswax, PEG-12 beeswax, PEG- 20 beeswax), dimethicone copolyol beeswax esters and dimethiconol beeswax ester
- Liquid polyolefin conditioning oils can be used in the compositions of the present invention.
- the liquid polyolefin conditioning agents are typically poly-a-olefins that have been hydrogenated.
- Polyolefins for use herein can be prepared by the polymerization of C4 to about C14 olefinic monomers.
- Non-limiting examples of olefinic monomers for use in preparing the polyolefin liquids herein include ethylene, propylene, 1 -butene, 1 -pentene, 1 -hexene, 1 -octene, 1 -decene, 1 -dodecene, 1 -tetradecene, branched chain isomers such as 4-methyl-1 -pentene, and mixtures thereof.
- hydrogenated a-olefin monomers include but are not limited to 1-hexene to 1 -hexadecenes, 1-octene to 1 -tetradecene, and mixtures thereof.
- fatty esters having at least 10 carbon atoms include esters derived from fatty acids or alcohols (e.g., mono-esters, polyhydric alcohol esters, and di- and tri-carboxylic acid esters).
- the fatty esters hereof may include or have covalently bonded thereto other compatible functionalities, such as amides and alkoxy moieties (e.g., ethoxy or ether linkages, etc.).
- Exemplary fatty esters include, but are not limited to isopropyl isostearate, hexyl laurate, isohexyl laurate, isohexyl palmitate, isopropyl palmitate, decyl oleate, isodecyl oleate, hexadecyl stearate, decyl stearate, isopropyl isostearate, dihexyldecyl adipate, lauryl lactate, myristyl lactate, cetyl lactate, oleyl stearate, oleyl oleate, oleyl myristate, lauryl acetate, cetyl propionate, and oleyl adipate.
- fatty esters suitable for use in the compositions of the disclosed technology are mono-carboxylic acid esters of the general formula R 50 C(O)OR 51 , wherein R 50 and R 51 are alkyl or alkenyl radicals, and the sum of carbon atoms in R 50 and R 51 is at least 10 in one aspect, and at least 22 in another aspect of the disclosed technology.
- Still other fatty esters suitable for use in the compositions of the present invention are di- and tri-alkyl and alkenyl esters of carboxylic acids, such as esters of C4-C8 dicarboxylic acids (e.g., C1-C22 esters, preferably C1-C6, of succinic acid, glutaric acid, adipic acid).
- esters of C4-C8 dicarboxylic acids e.g., C1-C22 esters, preferably C1-C6, of succinic acid, glutaric acid, adipic acid.
- Specific non-limiting examples of di- and tri-alkyl and alkenyl esters of carboxylic acids include isocetyl stearyol stearate, diisopropyl adipate, and tristearyl citrate.
- Other fatty esters suitable for use in the compositions of the present invention are those known as polyhydric alcohol esters.
- the amount of conditioning oils, waxes and esters can range from about 0.05 to about 10 wt.%, or from about 0.5 to about 5 wt.%. or from about 1 to about 3 wt.%, based on the total weight of the composition.
- Fatty acids and fatty alcohols can be employed in the compositions of the present invention.
- Suitable fatty acids include saturated and unsaturated Cs to C30 fatty acids.
- Exemplary fatty acids include, but are not limited to, caprylic acid, capric acid, lauric acid, myristic acid, palmitic acid, palmitoleic acid, stearic acid, oleic acid, ricinoleic acid, vaccenic acid, linoleic acid, a-linolenic acid, y-linolenic acid, arachidic acid, gadoleic acid, arachidonic acid, EPA (5,8,11 ,14, 17-eicosapentaenoic acid), behenic acid, erucic acid, DHA (4,7,10,13,16,19-docosahexaenoic acid), lignoceric acid, and mixtures thereof.
- Alkoxylated fatty acids are also useful herein and can be formed by esterifying a fatty acid with an ethylene oxide and/or propylene oxide or with a pre-formed polymeric ether (e.g., polyethylene glycol or polypropylene glycol).
- a fatty acid with an ethylene oxide and/or propylene oxide or with a pre-formed polymeric ether (e.g., polyethylene glycol or polypropylene glycol).
- the fatty alcohols suitable for use in the compositions of the invention include, but are not limited to, the saturated and unsaturated C8-C30 fatty alcohols.
- Fatty alcohols are widely available and can be obtained through the hydrogenation of esterified vegetable and animal oils and fats.
- Suitable ethoxylated fatty alcohols are adducts of fatty alcohols and polyethylene oxide.
- the fatty acids and fatty alcohols be utilized in an amount ranging from about 0.1 to about 30 wt.%, or from about 0.5 to 25 wt.%, or from about 3 to 20 wt.%, or from 5 to about 10 wt.%, based on the total weight of the composition.
- Humectants are defined as materials that absorb or release water vapor, depending on the relative humidity of the environment, (Harry's Cosmeticology, Chemical Publishing Company Inc., 1982 p. 266). Suitable humectants that include, but are not limited to, allantoin; pyrrolidonecarboxylic acid and its salts; hyaluronic acid and salts thereof; sorbic acid and salts thereof; urea, lysine, cystine, and amino acids; polyhydroxy alcohols such as glycerin, propylene glycol, hexylene glycol, hexanetriol, ethoxydiglycol, dimethicone copolyol, and sorbitol, and the esters thereof; polyethylene glycol; glycolic acid and glycolate salts (e.g.
- ammonium and quaternary alkyl ammonium chitosan; aloe- vera extracts; algae extract; honey and derivatives thereof; inositol; lactic acid and lactate salts (e.g. ammonium and quaternary alkyl ammonium); sugars and starches (e.g., maltose, glucose, fructose); sugar and starch derivatives (e.g., glucose alkoxylated glucose, mannitol, xyliyol); DL-panthenol; magnesium ascorbyl phosphate, arbutin, kojic acid, lactamide monoethanolamine; acetamide monoethanolamine; and the like, and mixtures thereof.
- sugars and starches e.g., maltose, glucose, fructose
- sugar and starch derivatives e.g., glucose alkoxylated glucose, mannitol, xyliyol
- DL-panthenol magnesium
- Humectants also include the C3 to Ce diols and triols, such as glycerin, propylene glycol, butane-1 ,2,3-triol, hexylene glycol, hexanetriol, and the like, and mixtures thereof.
- Such humectants may be present at from 0.01-20 wt. % of the composition, such as at least 0.1 wt.%, or at least 1 wt.%, e.g., up to 8 wt.%, or up to 5 wt.% based on the total weight of the composition.
- a skin sensate helps provide a sensory confirmation of the adequacy, activity, and evenness of the application thereof by a user.
- Some non-limiting examples of skin sensates are described in U.S. Pat. Nos. 4,230,688, 4,136,163, 6,183,766 and 7,001 ,594 each of which are incorporated herein by reference in their entireties.
- Non-limiting examples of suitable sensates include butanedioic acid monomenthyl ester, camphor, carvone, cineole, clove oil, ethyl carboxamide, ethyl menthane carboxamide, eucalyptus oil, eucolytol, ginger oil, l-isopulegol, menthol, menthone glycerin acetal, menthoxy-1 ,2- propanediol, menthyl lactate, methyl diisopropylpropioniamide, methyl salicylate, peppermint oil, rosemary oil, trimethyl butanamide, vanillyl butyl ether or combinations thereof.
- the sensate can be included in the composition in amounts ranging from about 0.01 wt.% to about 2 wt.% in one aspect, and from about 0.05 wt.% to about 1 wt.% in another aspect, based on the total weight of the composition.
- compositions of the present technology can also comprise fragrance or perfume components may range from about 0.000001 to about 2 wt.%, or from 0.00001 to about 1.5 wt.%, or from 0.0001 to about 1 wt.%, or from about 0.001 to about 0.8 wt.%, based on of the weight of the composition.
- Suitable preservatives include polymethoxy bicyclic oxazolidine, methyl paraben, propyl paraben, ethyl paraben, butyl paraben, benzyltriazole, DMDM hydantoin (also known as 1 ,3-dimethyl-5,5-dimethyl hydantoin), imidazolidinyl urea, phenoxyethanol, phenoxyethylparaben, methylisothiazolinone, methylchloroisothiazolinone, benzoisothiazolinone, triclosan, and suitable polyquaternium compounds as disclosed above (e.g., Polyquaternium- 1).
- Acid-based preservatives are also useful in the exemplary compositions.
- Exemplary acids are, but are not limited to, formic acid, acetic acid, propionic acid, sorbic acid, caprylic acid, and benzoic acid, oxalic acid, succinic acid, glutaric acid, adipic acid, azelaic acid, maleic acid, fumaric acid, lactic acid, glyceric acid, tartronic acid malic acid, tartaric acid, gluconic acid, citric acid, ascorbic acid, salicylic acid, phthalic acid, mandelic acid, benzilic acid, and mixtures thereof.
- Salts of the foregoing acids are also useful as long as they retain efficacy at low pH values.
- Suitable salts include the alkali metal (e.g., sodium, potassium, calcium) and ammonium salts of the acids enumerated above.
- the acid-based preservatives and/or their salts can be used alone or in combination with non-acidic preservatives typically employed in personal care, home care, health care, and institutional and industrial care products.
- Suitable antibacterial agents include, but are not limited to, 2-hydroxy-4,2',4'-trichlorodiphenylether (TCS), 2,6- dimethyl-4-hydroxychlorobenzene (PCMX),3,4,4'-trichlorocarbanilide (TCC), 3- trifluoromethyl-4,4'-dichlorocarbanilide (TFC), 2,2'-dihydroxy-3, 3', 5, 5', 6,6'- hexachlorodiphenylmethane, 2,2'-dihydroxy-3,3',5,5'-tetrachlorodiphenylmethane, 2,2'-dihydroxy-3,3',dibromo-5,5'-dichlorodiphenylmethane, 2-hydroxy4,4'- dichlorodiphenylether, 2-hydroxy-3,5',4-tribromodiphenylether, 1-hydroxyl-4-methyl- 6-(2,4,4-trimethylpenty
- compositions and products in the context of the present invention may be combined with antioxidant agents.
- antioxidant agents include EDTA and its salts, citric acid, tartaric acid, oxalic acid, BHA (butylhydroxyanisol), BHT (butylhydroxytoluene), tocopherol derivatives such as tocopherol acetate, and mixtures thereof.
- the preservatives, antibacterial agents and antioxidants may comprise from 0.01 to 3.0 wt.%, or from about 0.1 to about 1 wt.%, or from about 0.3 to about 1 wt.%, based on the weight of the total composition.
- the compositions of the present invention can be prepared as water-free or water-based formulations, and formulations containing water-miscible auxiliary solvents and/or diluents but are not limited thereto.
- the compositions of the present invention are particularly useful for water-based, solvent based, hydroalcoholic based, and mixed solvent formulations, and for formulations containing water-miscible auxiliary solvents.
- Useful solvents commonly employed are typically liquids, such as water (deionized, distilled or purified), alcohols, fatty alcohols, polyols, and the like, and mixtures thereof.
- Non-aqueous or hydrophobic auxiliary solvents are commonly employed in substantially water-free products, such as nail lacquers, aerosol propellant sprays, or for specific functions, such as removal of oily soils, sebum, make-up, or for dissolving dyes, fragrances, and the like, or are incorporated in the oily phase of an emulsion.
- auxiliary solvents other than water
- linear and branched alcohols such as ethanol, propanol, isopropanol, hexanol, and the like
- aromatic alcohols such as benzyl alcohol, cyclohexanol, and the like
- saturated C12 to C30 fatty alcohol such as lauryl alcohol, myristyl alcohol, cetyl alcohol, stearyl alcohol, behenyl alcohol, and the like.
- Nonlimiting examples of polyols include polyhydroxy alcohols, such as glycerin, propylene glycol, butylene glycol, hexylene glycol, C2 to C4 alkoxylated alcohols and C2 to C4 alkoxylated polyols, such as ethoxylated, propoxylated, and butoxylated ethers of alcohols, diols, and polyols having about 2 to about 30 carbon atoms and 1 to about 40 alkoxy units, polypropylene glycol, polybutylene glycol, and the like.
- polyhydroxy alcohols such as glycerin, propylene glycol, butylene glycol, hexylene glycol, C2 to C4 alkoxylated alcohols and C2 to C4 alkoxylated polyols, such as ethoxylated, propoxylated, and butoxylated ethers of alcohols, diols, and polyols having about 2 to about 30 carbon atoms and 1
- Non-limiting examples of non-aqueous auxiliary solvents or diluents include silicones, and silicone derivatives, such as cyclomethicone, and the like, ketones such as acetone and methylethyl ketone; natural and synthetic oils and waxes, such as vegetable oils, plant oils, animal oils, essential oils, mineral oils, C7 to C40 isoparaffins, alkyl carboxylic esters, such as ethyl acetate, amyl acetate, ethyl lactate, and the like, jojoba oil, shark liver oil, and the like.
- Some of the foregoing non-aqueous auxiliary solvents or diluents may also be conditioners and emulsifiers.
- Natural and/or synthetic agent (or combination thereof) to obtain enhanced thickening properties can be also used in the context of the present invention.
- the person skilled in the art will readily select a proper thickening agent(s) and amounts(s) thereof to obtain the desired rheology.
- Non-limiting examples of natural thickening agents are tree & shrub exudates (karaya gum, tragacanth gum, gum Arabic, gum ghatti), seed extracts (guar gum, cassia gum, locust been gum, tamarind seed), seaweed extracts (carrageenan, alginates, agar), fruit extracts (pectins, waxes), grains & roots (corn starch, potato starch, etc), microbial polysaccharides (Xanthan gum, dextran), modified natural products (cellulose derivatives such as hydropropyl cellulose, methylcellulose, hydroxypropyl methylcellulose, cellulose gum, etc.); and hydrophobically modified ethoxylated methyl glucosides, such as PEG 120 methyl glucose dioleate, PEG- 120 methyl glucose trioleate, and PEG-20 methyl glucose sesquistearate, available from Lubrizol Advanced Materials, Inc., under the trade names, GlucamateTM DOE-120, Glucamate
- Non-limiting examples of synthetic thickening agents include the polyethylene glycols (PEG) having 5 to 200 glycol units, such as, for example, those available under the INCI names PEG- 6, PEG-8, PEG-12, PEG-20, PEG-30, PEG-32, PEG-75, PEG-90, PEG-100 and PEG-200; acrylic/methacrylic acid homopolymers and copolymers; acrylamide homopolymers and copolymers; polymers prepared from 2- acrylamido-2-methylpropanesulfonic acid (AMPS® monomer).
- PEG polyethylene glycols having 5 to 200 glycol units
- PEG polyethylene glycols having 5 to 200 glycol units
- acrylic/methacrylic acid homopolymers and copolymers such as, for example, those available under the INCI names PEG
- HASE polymers include the hydrophobically modified alkali-swellable emulsion polymers, commonly referred to as (HASE) polymers.
- Typical HASE polymers are free radical addition polymers polymerized from pH sensitive or hydrophilic monomers (e.g., acrylic acid and/or methacrylic acid, 2- acrylamido-2-methylpropane sulfonic acid), hydrophobic monomers (e.g., C1-C30 alkyl esters of acrylic acid and/or methacrylic acid, acrylonitrile, styrene), an "associative monomer", and an optional crosslinking monomer.
- pH sensitive or hydrophilic monomers e.g., acrylic acid and/or methacrylic acid, 2- acrylamido-2-methylpropane sulfonic acid
- hydrophobic monomers e.g., C1-C30 alkyl esters of acrylic acid and/or methacrylic acid, acrylonitrile, styren
- HASE polymers are disclosed in U.S. Patent Nos. 3,657,175; 4,384,096; 4,464,524; 4,801 ,671 ; and 5,292,843, which are herein incorporated by reference.
- an extensive review of HASE polymers is found in Gregory D. Shay, Chapter 25, "Alkali-Swellable and Alkali-Soluble Thickener Technology A Review", Polymers in Aqueous Media - Performance Through Association, Advances in Chemistry Series 223, J. Edward Glass (ed.), ACS, pp. 457-494, Division Polymeric Materials, Washington, DC (1989), the relevant disclosures of which are incorporated herein by reference.
- the thickeners can comprise from about 0.01 wt.% to about 5 wt. % of the total weight of the personal care composition, in another aspect the amount ranges from about 0.1 wt. % to about 3 wt. %, and in a further aspect from about 0.1 wt. % to about 2.0 wt. % of the total weight of the personal care composition.
- Viscosity adjusting agents are used in cosmetics to enhance the fluidity of products without a significant lowering of the concentration of the active constituents.
- Suitable viscosity adjusting agents if present include organic and inorganic compounds, and combinations thereof.
- organic compounds include ethanol, isopropyl alcohol, sorbitol, propylene glycol, diethylene glycol, triethylene glycol, dimethyl ether, butylene glycol, and the like, and mixtures thereof.
- examples of inorganic compounds include sodium chloride, sodium sulfate, potassium chloride, potassium nitrate, and mixtures thereof. If utilized the viscosity adjusting agents typically comprise from about 0.1 wt. % to about 20 wt.% in one aspect, and from about 1 wt.% to about 5 wt.% of the total weight of the composition.
- compositions of the disclosed invention can contain active ingredients or pharmaceutically active agents.
- the ‘personal care active ingredients’ as used herein refers to ingredients that improve or enhance the physical appearance. They are also commonly known as ‘cosmetic active ingredients’.
- Non-limiting examples of ingredients commonly used in cosmetic or pharmaceutical compositions for example and, not restricted to, (i) anti-wrinkle agents, botox-like agents and/or anti-aging agents; (ii) firming agents, skin elasticity agents and/or restructuring agents; (iii) moisturizing agents; (iv) anti-photoaging agents, and/or bluelight protector agents; DNA protecting agents, DNA repair agents, and/or stem cell protecting agents; (v) free radical scavengers and/or anti-glycation agents, detoxifying agents, antioxidant and/or anti-pollution agents; (v) agents which increase the percutaneous absorption of any active compounds present therein; (vi) anti-perspirant agents; (vii) melanin synthesis stimulating or inhibiting agents; whitening or depigmenting agents; pro-pigmenting agents; self-tanning agents; (viii) lipolytic agents or agents stimulating lipolysis, adipogenic agents, etc.
- Suitable active ingredient in the context of the present invention include, but are not limited to, peptides, proteins, hydrolyzed proteins, enzymes, vitamins, mineral salts, sugars, nucleotides, nucleic acids, molecules and extracts of biological and biotechnological origin, vegetable extracts, cells extracts, essential oils, molecules of synthetic or natural origin, isoflavones, polyphenols, retinol, hyaluronic acid and/or mixtures thereof.
- the active ingredient can be typically used in a concentration from 0.001 to 10 wt. %, or from 0.1 to 5 wt. %, or from 0.2 to 3 wt.% based on the total weight of the composition.
- Brookfield viscosity measurement method [00159] Viscosity and yield values were determined with a Brookfield viscosimeter. Sample fluids were free of entrapped air as much as possible and maintained at constant temperature of 25°C (i.e., coming from a water bath or oven).
- the spindle of the viscosimeter was inserted into the sample at 25°C.
- the viscosimeter was set at different speeds namely 0.5, 1 , 2.5, 5, 10, and 20 rpm and the spindle allowed to rotate at the selected rpm for 2 minutes for 0.5 rpm and 1 minute for all other rpms.
- the spindle is selected to ensure that the % torque reading is between 10 and 90%.
- emulsion coalescence was determined by visual inspection and scored from 0 to 5 where: 0 represents visual formation of thick film layer of oil in the surface; 1 , a slight film layer formation in the surface; 2 oil pooling; 3, numerous small and large droplets; 4, few small droplets; 5, no visible oil droplets.
- the mixture was neutralized with glacial acetic acid (5.98 g) to a pH of 6.84.
- the solids were recovered by filtration using a Millipore pressure filtration device, and the filter cake was washed with 80 wt.% isopropanol (303.45 g) in a 1 L beaker using overhead stirring with a marine mixing blade for 30 min.
- the product was recovered by filtration, and the filter cake was dried in an aluminum pan in a vacuum oven set to 80°C for 4 h.
- the resulting tan powder had a total solids of 94.6 wt.%.
- a 0.5 wt.% dispersion of the product in water had a Brookfield viscosity of 1403 ⁇ 42.9 mPa s (20 rpm, average of 3 replicates).
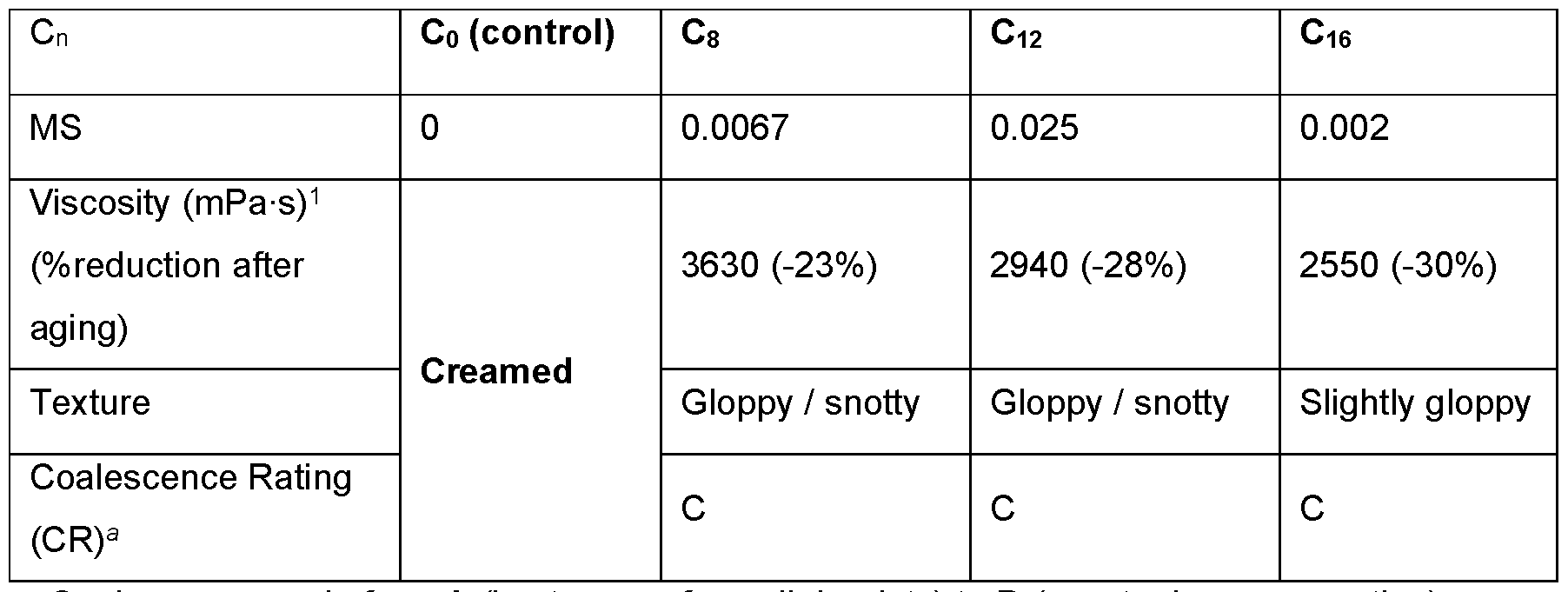
- compositions were generated by varying both the alkyl chain length of the hydrophobe and its molar substitution (MS) on the polymer, which was calculated as the molar ratio of hydrophobe per anhydroglycosidic unit (AGU).
- the alkyl chain length ranged from Cs to C22, and the MS ranged from 0.002 - 0.05.
- Xanthan gum demonstrated a general increase in dispersion viscosity and yield value after modification compared to the native xanthan gum ( Figure 1).
- Emulsion performance was monitored using an oil-in- water system containing 20 wt.% SchercemolTM 318 (INCI: Isopropyl Isostearate from Lubrizol Advanced Materials, INC), 0.5 wt.% xanthan polymer, and 0.5 wt.% phenoxyethanol. Homogenization with an Ultra-Turrax® homogeneizer was used at 10,000 rpm for one minute. Importantly, all emulsions containing hydrophobically modified xanthan polymers were stable against creaming during stability testing for four weeks at 50°C compared to a control polymer that underwent the same reaction conditions but without the hydrophobe. However, significant reduction in viscosity and yield values were observed during stability testing, as well as poor texture and coalescence ratings (Table 1).
- Emulsion results for polymers that were dried at low or high temperatures. All samples were Ci6-modified at MS 0.05. Emulsions contained 0.5 wt.% polymer, 20 wt.% Caprylic/Capric Triglyceride and 0.5 wt.% phenoxyethanol and were homogenized at 10,000 rpm for one minute. a Viscosity determined as Brookfield viscosity (20 rpm, at room temp.) after 24h rest.
- Emulsions contained 0.5 wt.% polymer, 20 wt. % caprylic/capric triglyceride, and 0.5 wt.% phenoxyethanol.
- Table 4 Coalescence rating of emulsions containing crosslinked and hydrophobic xanthan gum. The emulsions were made using 0.5 wt.% modified xanthan and 20 wt.% caprylic/capric triglyceride.
- Coalescence rating 5 is the best, 0 is the worst.
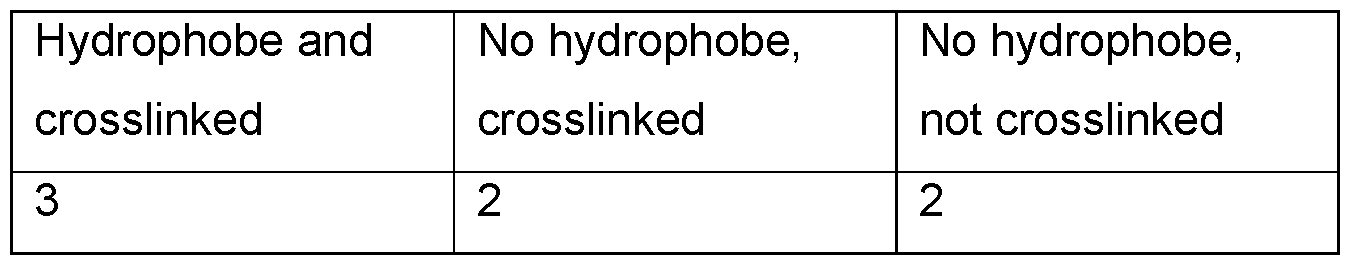
- the highest viscosity sample was the xanthan that was both hydrophobically modified and crosslinked (Table 7).
- Table 7 Viscosity and yield values for emulsions containing either hydrophobic and crosslinked xanthan, non-hydrophobic, but crosslinked xanthan, or non-hydrophobic and non-crosslinked xanthan.
- Emulsions contained 0.5 wt.% polymer and 20 wt.% Caprylic/capric triglyceride.
- Emulsion contains 0.5 wt.% polymer and 20 wt.% caprylic/capric trygliceride.
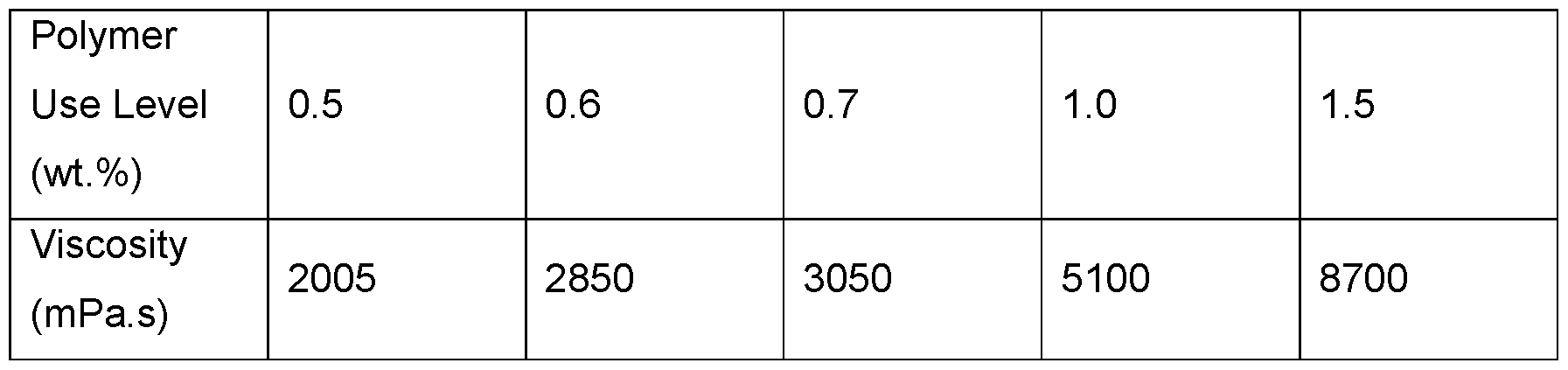
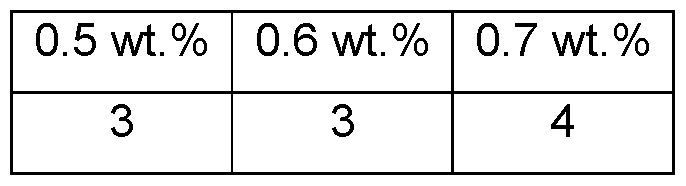
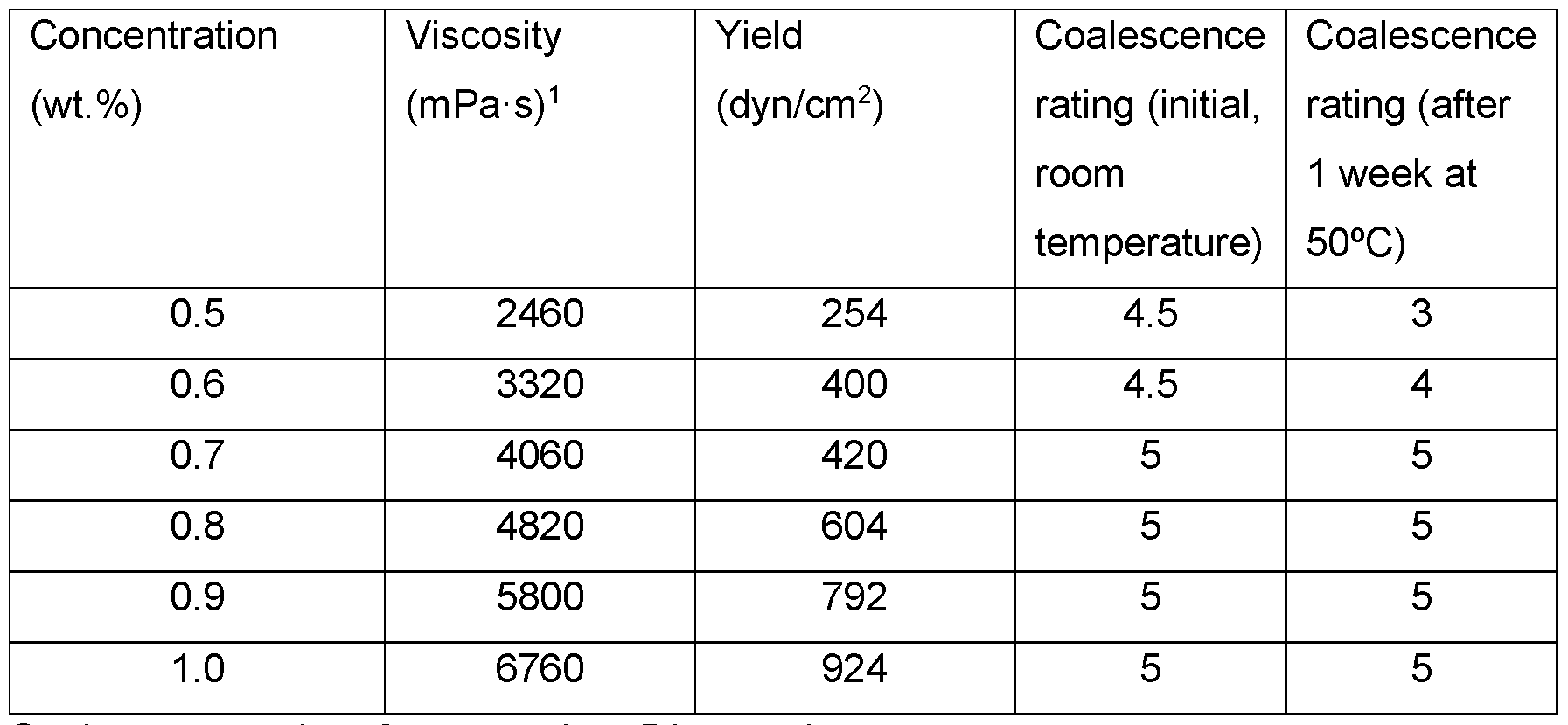
- Emulsion stability as a function of polymer use level was also evaluated using emulsions containing 20 wt.% caprylic/capric triglyceride at pH 5.5. Passing coalescence ratings were observed after aging at 50°C for one week for all emulsions with a polymer concentration >0.6 wt.% (Table 11). A range of viscosities was also observed.
- Emulsions containing modified xanthan of Example 1 at increased use levels contained 20 wt.% caprylic/capric triglyceride at pH 5.5.
- Coalescence rating 0 worst rating, 5 best rating.
- Viscosity determined as Brookfield viscosity at 20 rpm at room temperature.
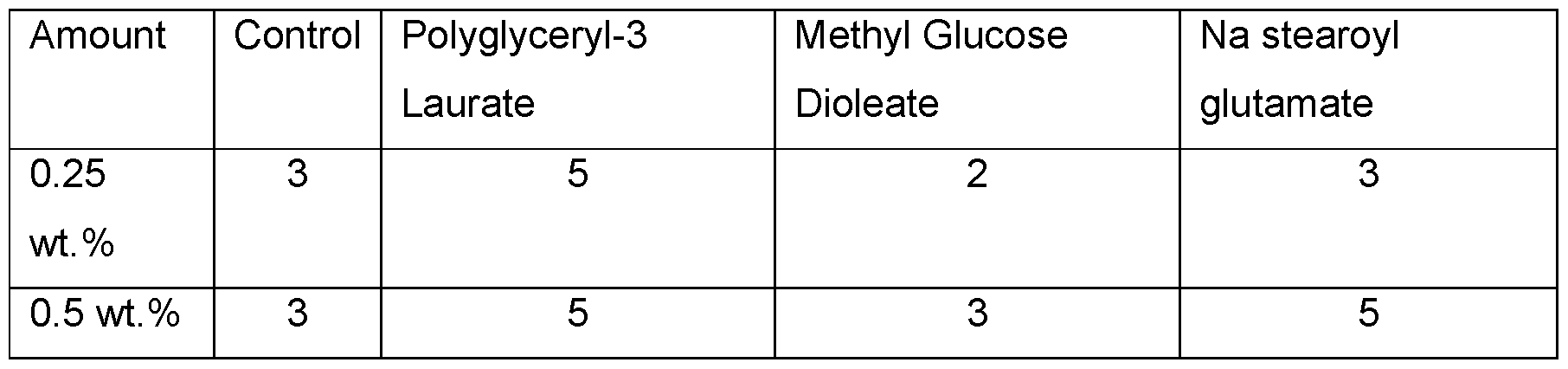
- GlucateTM DO (INCI: Methyl Glucose Dioleate from Lubrizol Advance Materials, Inc.) showed no improvement; therefore, mid- to high range HLB co-emulsifiers in combination with the modified xanthan gum according to the invention provide better results (Table 12).
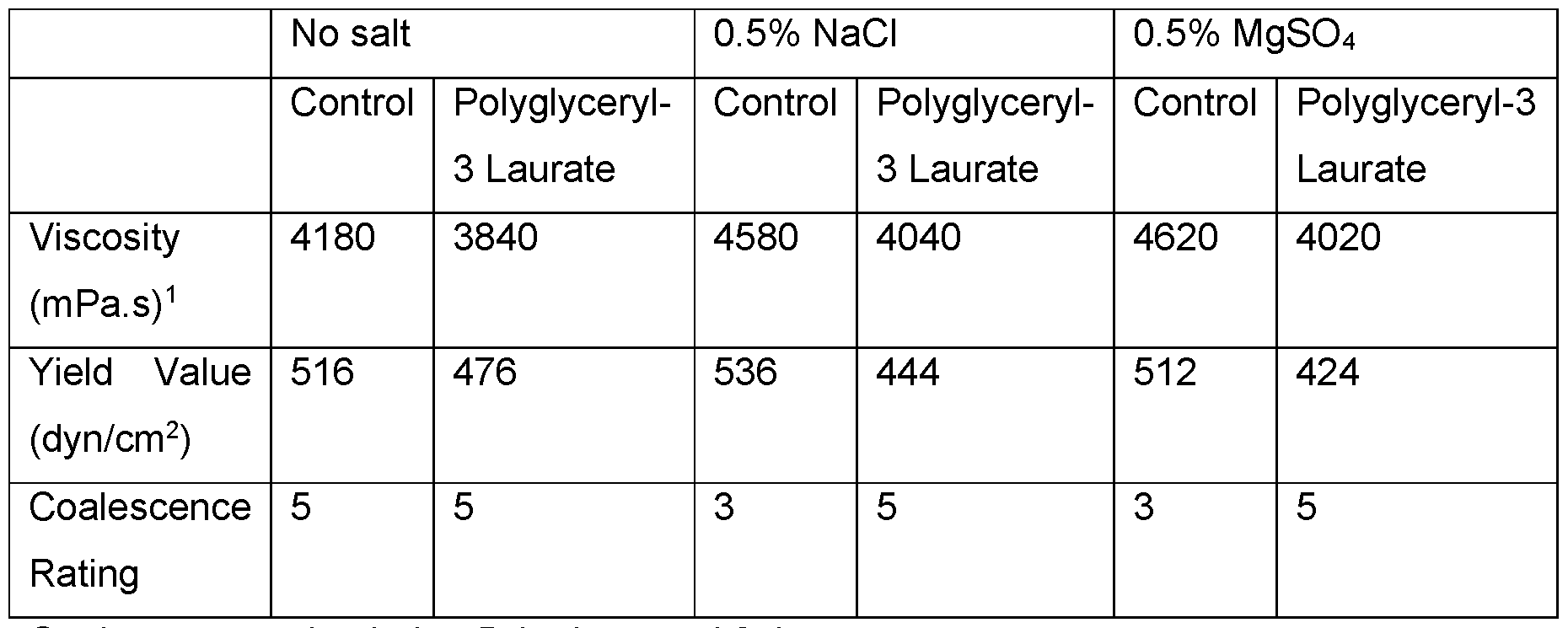
- Electrolyte tolerance was also evaluated by adding 0.5 wt.% NaCI or MgSC>4 to emulsions containing 0.7 wt.% of modified xanthan gum of Example 1 , and 20 wt.% caprylic/capric triglyceride. Emulsion viscosity and yield values were unaffected by salt addition, but the coalescence ratings decreased. Adding 0.1 wt.% Polyglyceryl-3 Laurate to the emulsion improved the initial coalescence rating (Table 13).
- Emulsion evaluation with the addition of 0.5 wt.% NaCI or MgSO 4 Emulsions contained 0.7 wt.% of modified xanthan of Example 1 and 20 wt.% caprylic/capric triglyceride oil. Data collected before aging.
- Coalescence rating being 5 the best and 0 the worst.
- Viscosity determined Brookfield viscosity at 20 rpm at room temperature.
- Modified xanthan of the invention was also found to be stable in formulations with pH ranging from 4 - 8, which allows for a wide range of skin care applications. Emulsions prepared at various processing conditions using higher shear rates demonstrated that there was no negative impact of shear on polymer performance. Additionally, the modified xanthan of the invention is compatible with glycols including GlucamTM E-20 humectant (from Lubrizol Advanced Materials, Inc), Zemea® propanediol (from CovationBio PDO LLC), and glycerin, which reduced hydration time without negatively affecting viscosity.
- glycols including GlucamTM E-20 humectant (from Lubrizol Advanced Materials, Inc), Zemea® propanediol (from CovationBio PDO LLC), and glycerin, which reduced hydration time without negatively affecting viscosity.
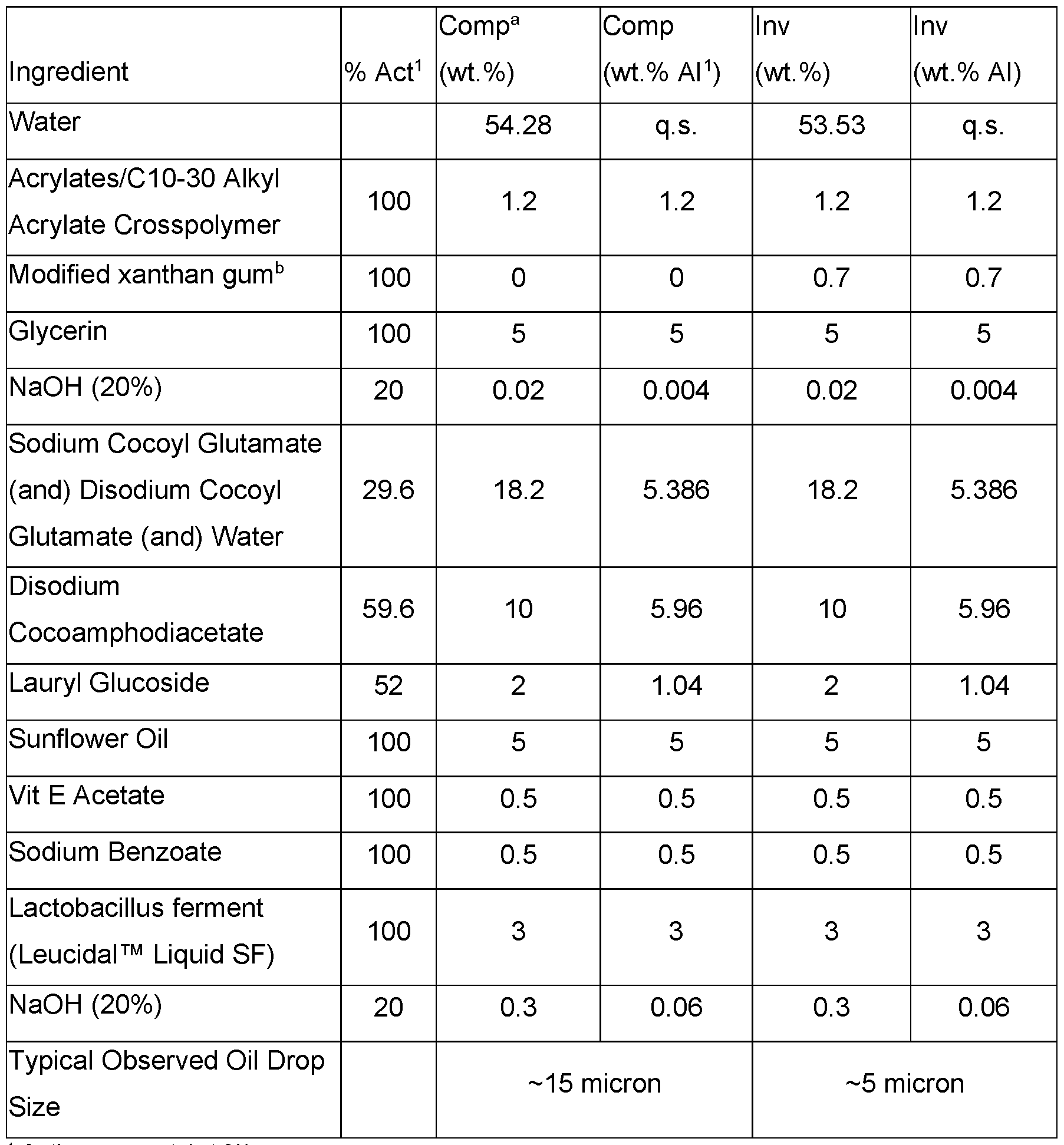
- a cleansing formulation was prepared as set out in Table 16.
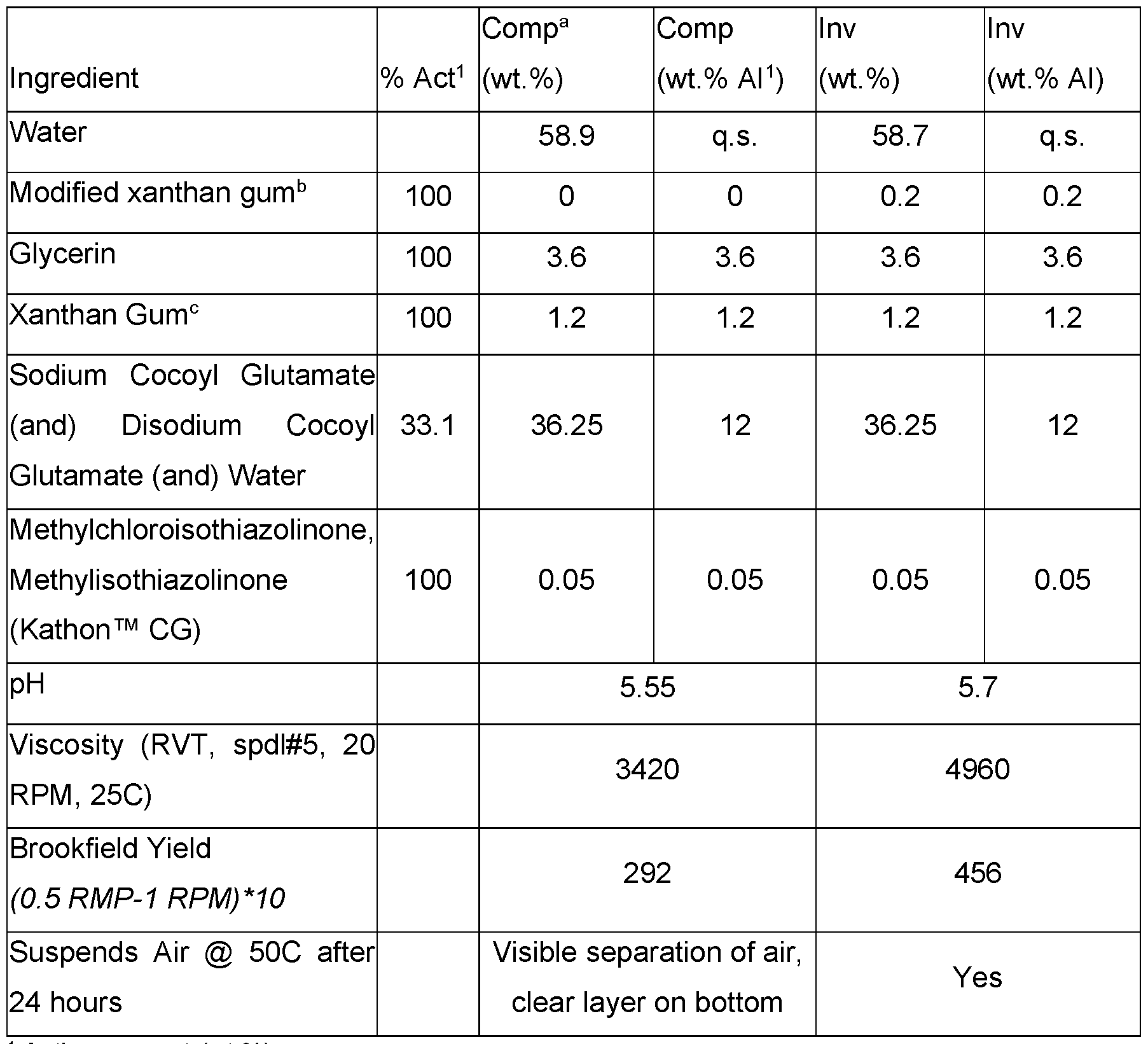
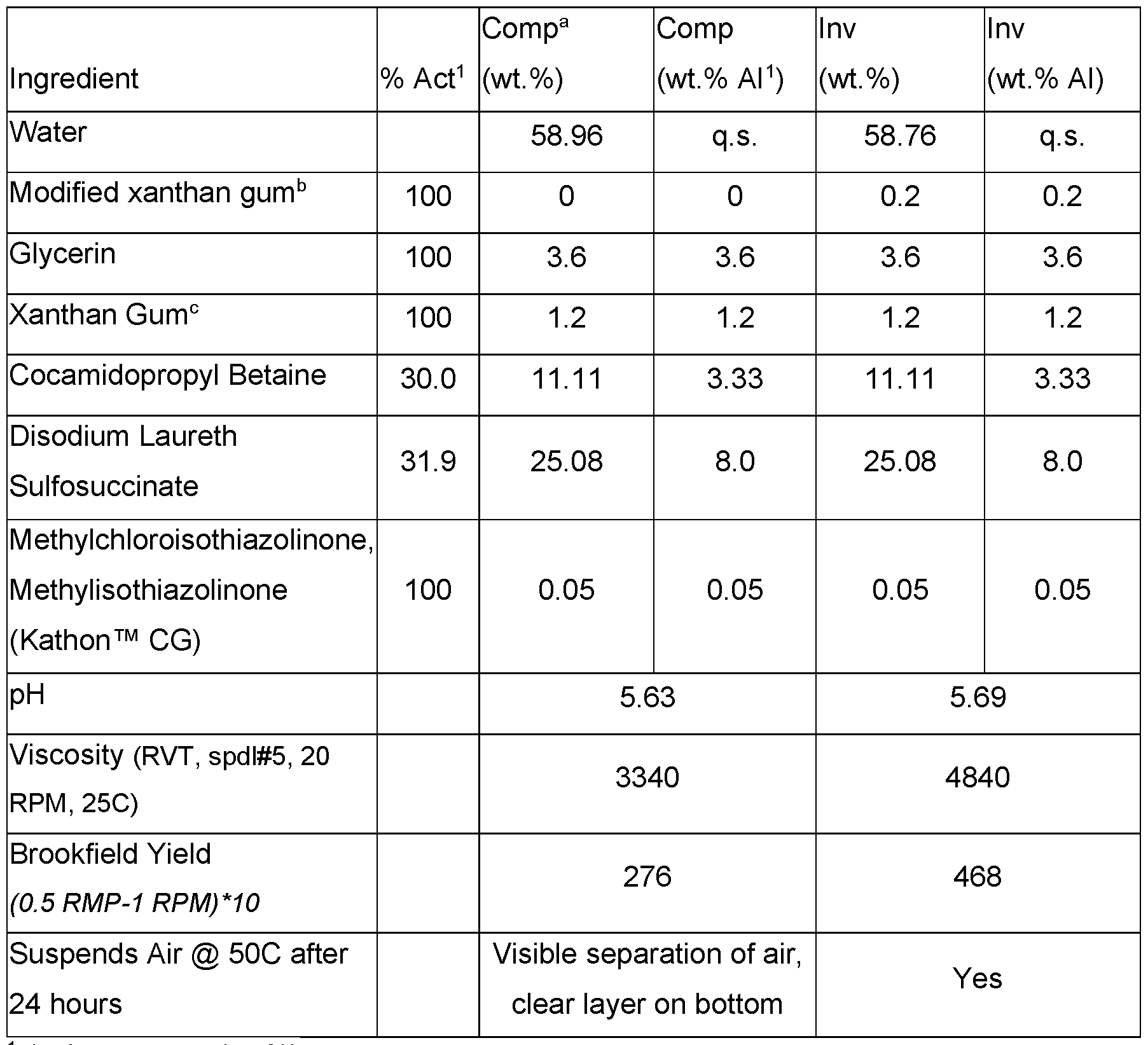
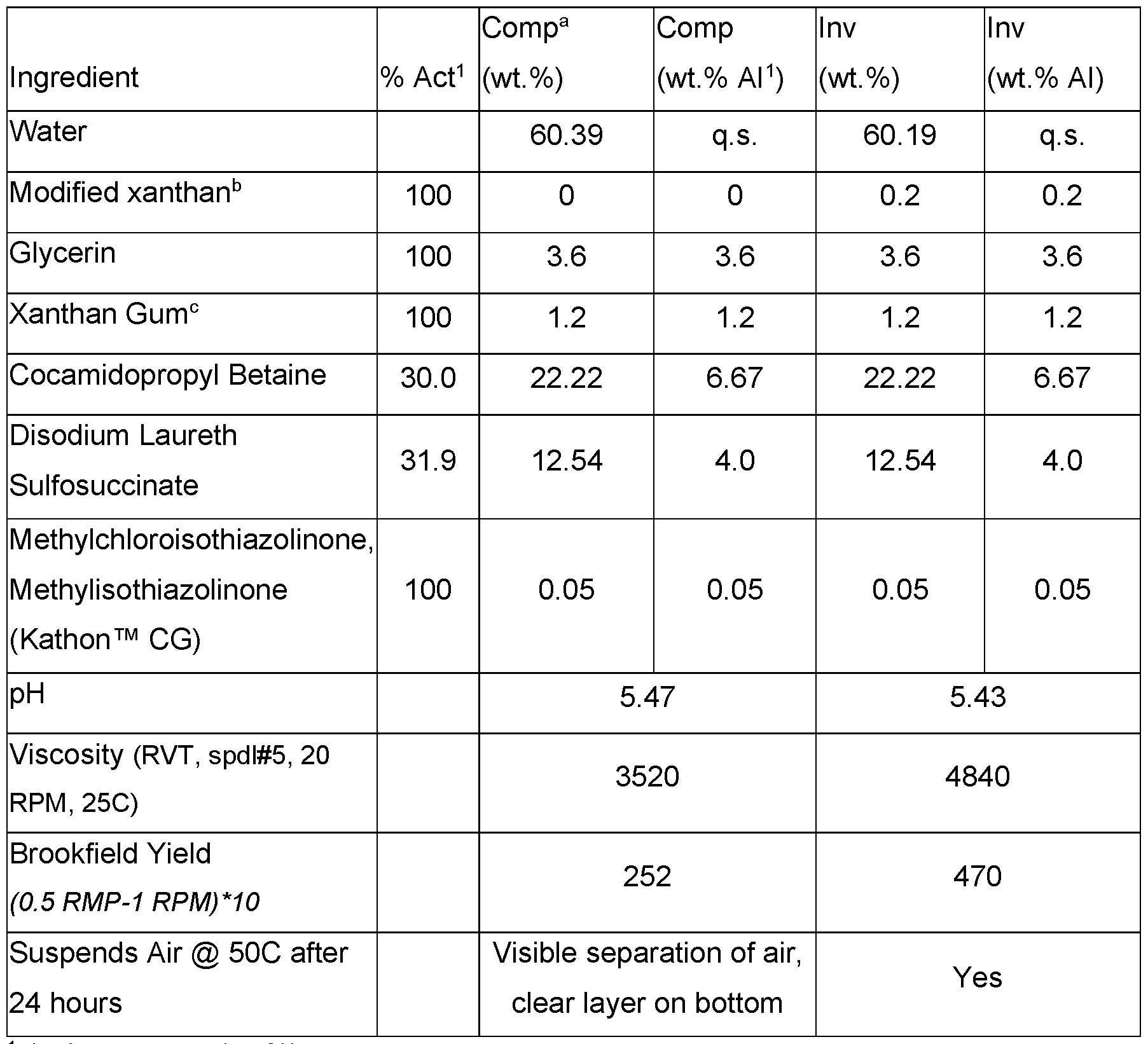
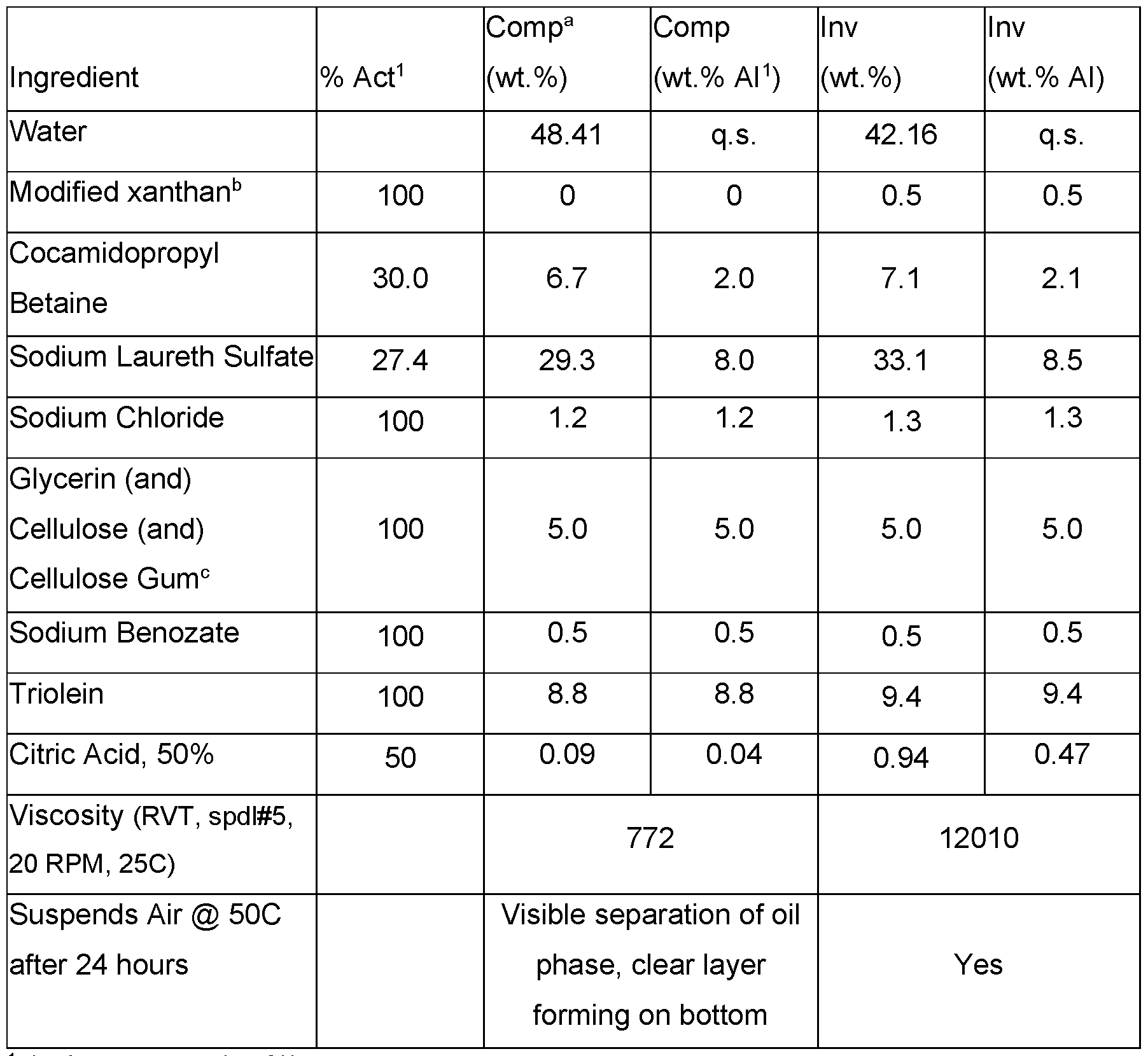
- the modified xanthan gum can significantly improve suspension of things other than oils, such as air, by improving the overall rheological properties (demonstrated through and increase in viscosity, yield, and the physical suspension of air at elevated temperatures).
- the ability to suspend visually pleasing ingredients such as air, beads, effect pigments, pearlizers, etc can be used to enhance the overall consumer perception and acceptance of personal care products.
- Cleansing formulations were prepared as set out in Table 18. Table 18. Cleansing formulations that suspend air with modified xanthan gum.
- Cleansing formulations were prepared as set out in Table 19. Table 19. Cleansing formulations that suspend oil with modified xanthan gum.
- Glycidyl ether HAGE-16 from Sachem, Inc (Texas, USA) (5.5 g) was melted in a 115°C oven for 10 min and dissolved in isopropanol (15.6 g).
- Epichlorohydrin (42 microliters) was added to 7.0 g of this solution which was then charged to the reactor at 60°C. The nitrogen flow rate was reduced to 0.185 L/min. After 3 h the reaction was cooled to 40 °C and had a pH of 12.3. The mixture was neutralized with glacial acetic acid (1 .7 g) to a pH of 6.7.
- the solids were recovered by vacuum filtration, and the filter cake was washed with 80 wt.% isopropanol (400 g) in a 1 L beaker using overhead stirring with a marine mixing blade for 30 min.
- the product was recovered by filtration, and the filter cake was dried in an aluminum pan in a vacuum oven set to 80°C for 4 h.
- the resulting tan powder had an amount of total solids of 97.2 wt.%.
- a 1 wt.% dispersion of the product in water had a Brookfield viscosity of 9520 mPa s (20 rpm).
- a Jacketed glass pressure/filter reactor and a 4-neck lid fitted with a thermocouple, glass stir shaft, nitrogen inlet, and condenser was charged with 60 wt.% isopropanol/water (351 .2 g).
- the solvent was stirred at 400 rpm while the headspace was purged with nitrogen at 0.4 L/min.
- Xanthan gum (81.4 g) was charged to the reactor slowly via powder funnel while stirring to provide a slurry.
- sodium hydroxide 50 wt.%, 12.0 g
- the pH of the reaction solution was 12.8.
- the temperature was set to 60°C.
- the solids were recovered by pressure filtration, and the filter cake was re-dispersed in 80 wt.% isopropanol (500 g) and washed for 30 min.
- the product was recovered by pressure filtration.
- the filter cake was removed from the reactor and dried in a vacuum oven set to 70°C for 4 h.
- the resulting tan powder had a total solids of 95.4 wt.%.
- a 1 wt.% dispersion of the product in water had a Brookfield viscosity of 5590 mPa s (20 rpm).
- the solids were recovered by pressure filtration, and the filter cake was re-dispersed in 80 wt.% isopropanol (500 g) and washed for 30 min.
- the product was recovered by pressure filtration.
- the filter cake was removed from the reactor and dried in a vacuum oven set to 70°C for 4 h.
- the resulting tan powder had a total solids of 95.6 wt.%.
- a 1 wt.% dispersion of the product in water had a Brookfield viscosity of 5540 mPa s (20 rpm).
- Glycidyl ether HAGE-16 from Sachem, Inc (Texas, USA) (6.9 g) was melted in a 115°C oven for 10 min and dissolved in isopropanol (21.1 g). 1 ,4-dichlorobutane (0.39 g) was added to the latter solution and charged to the reactor at 60°C. The nitrogen flow rate was reduced to 0.185 L/min. After 3 h the reaction was cooled to 40 °C and had a pH of 11.9. The mixture was neutralized with glacial acetic acid (6.5 g) to a pH of 6.1.
- the solids were recovered by pressure filtration, and the filter cake was re-dispersed in 80 wt.% isopropanol (500 g) and washed for 30 min.
- the product was recovered by pressure filtration.
- the filter cake was removed from the reactor and dried in a vacuum oven set to 70°C for 4 h.
- the resulting tan powder had a total solids of 96.2 wt.%.
- a 1 wt.% dispersion of the product in water had a Brookfield viscosity of 3400 mPa s (20 rpm).
- a Jacketed glass pressure/filter reactor and a 4-neck lid fitted with a thermocouple, glass stir shaft, nitrogen inlet, and condenser was charged with 60 wt.% isopropanol/water (355.7 g). The solvent was stirred at 400 rpm while the headspace was purged with nitrogen at 0.4 L/min. Xanthan gum (81 .4 g) was charged to the reactor slowly via powder funnel while stirring to provide a slurry. After 5 min, sodium hydroxide (50 wt.%, 12.0 g) was slowly added to the reactor while stirring and purging with nitrogen. The pH of the reaction solution was 12.7. The temperature was set to 60°C.
- Glycidyl ether HAGE-16 from Sachem, Inc (Texas, USA) (6.9 g) was melted in a 115°C oven for 10 min and dissolved in isopropanol (19.5 g).
- Polypropylene glycol) diglycidyl ether Mw 380 g mol’ 1 (1.1 g) was added to the latter solution and charged to the reactor at 60°C. The nitrogen flow rate was reduced to 0.185 L/min. After 3 h the reaction was cooled to 40 °C and had a pH of 11 .9. The mixture was neutralized with glacial acetic acid (6.2 g) to a pH of 6.1.
- the solids were recovered by pressure filtration, and the filter cake was redispersed in 80 wt.% isopropanol (500 g) and washed for 30 min.
- the product was recovered by pressure filtration.
- the filter cake was removed from the reactor and dried in a vacuum oven set to 70°C for 4 h.
- the resulting tan powder had a total solids of 97.4 wt.%.
- a 1 wt.% dispersion of the product in water had a Brookfield viscosity of 4080 mPa s (20 rpm).
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Dermatology (AREA)
- Animal Behavior & Ethology (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Molecular Biology (AREA)
- Emergency Medicine (AREA)
- Engineering & Computer Science (AREA)
- Biochemistry (AREA)
- Materials Engineering (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Medicinal Chemistry (AREA)
- Polymers & Plastics (AREA)
- Organic Chemistry (AREA)
- Cosmetics (AREA)
Abstract
The present invention relates to crosslinked and hydrophobically modified xanthan gums. The invention also extends to methods for preparing said modified xanthan gums. The disclosed technology is useful for the preparation of personal care, home care, industrial and health care applications, particularly as emulsifiers in the formulation of phase stable emulsions.
Description
CROSSLINKED AND HYDROPHOBICALLY MODIFIED XANTHAN GUM
FIELD OF THE INVENTION
[0001] The present invention relates to crosslinked and hydrophobically modified xanthan gum polymers and to a method for making the same. These compounds are useful for the preparation of personal care, home care, industrial, and health care applications, particularly as emulsifiers in the formulation of phase stable emulsions.
BACKGROUND OF THE INVENTION
[0002] Sustainability in the personal care industry has drawn considerable attention from media and society, and there is an increasing demand of ingredients with higher renewable carbon index and a lower environmental footprint.
[0003] Microbial polysaccharides are biocompatible, biodegradable, and usually nontoxic natural biopolymers that possess physicochemical properties suitable for use as cosmetic ingredients. These properties are also inherent to xanthan gum.
[0004] Xanthan gum is a microbial polysaccharide produced commercially by a fermentation process using Xanthomonas campestris bacteria. The structure of xanthan gum is composed of a p-1 ,4-D-glucose backbone (identical to cellulose) with a regular trimeric sidechain on every second glucose to make a pentasaccharide repeat unit. Each side chain is connected to the backbone via an a-1 ,3 linkage and has a glucuronic acid between two mannoses. Additionally, there is a negatively charged pyruvate ketal on approximately 50% of the terminal mannose groups and a varying amount of acetylation at the O-6 position of the internal mannose unit.
[0005] Due to high molecular weight and hydrogen bonding interactions, an aqueous solution of xanthan gum exhibits high intrinsic viscosity at low concentrations and behaves like a pseudoplastic fluid. The high viscous rheology, pH and salt-resistant properties of xanthan gum are satisfactory for use as thickening agent, suspending agent, emulsion stabilization for food, pharmaceuticals, oral care and cosmetics.
[0006] However, native xanthan gum has its own limitations, including unstable viscosity, inadequate impact on yield values, insufficient performance stabilizing emulsions, unsatisfactory texture and being susceptible to microbial contamination.
[0007] JP9003101A provides a method for improving the thickening effect and transparency of xanthan gum as well as improving water solubility, in order to provide useful xanthan derivatives in toiletry and cosmetic fields.
[0008] There is the need to increase the performance properties of xanthan gum for personal care applications, such as emulsification of a variety of oils over a wide pH range, and improved sensory and textures with neutral odor and color. The present invention sets out to meet some or all of the above-identified needs and solve some or all of the above-identified problems.
SUMMARY OF THE INVENTION
[0009] The disclosed invention relates to a crosslinked and hydrophobically modified xanthan gum that offers a more natural and biodegradable alternative to traditional emulsifiers, while provides superior thickening and stabilization of emulsions comprising a variety of oils (polar to non-polar). The modified xanthan gum disclosed herein also allows to formulate emulsion compositions with suitable texture and superior electrolyte stability at low use levels.
[0010] Therefore, one object of the present invention is the provision of a crosslinked and hydrophobically modified xanthan gum wherein a portion of the hydrogen atoms of pendant hydroxyl groups of the backbone are substituted with at least one hydrophobic moiety according to formula (I):
(I) - (A)a-(O)b-R wherein A is selected from a divalent linear or branched, substituted or unsubstituted C1- C6 alkylene radical; R is selected from a hydrocarbon group having from 8 to 22 carbon atoms; a is 0 or 1 , and b is 0 or 1 , with the proviso that when a is 0, b is 0; and, wherein the xanthan gum is crosslinked with a multifunctional crosslinking agent comprising epoxy and/or alkyl halide functional groups in an amount of from 0.1 to 1 wt.% based on the total weight the xanthan gum.
[0011] Another object of the invention is the provision of oil-in-water emulsions comprising: a water phase; an oil phase dispersed in said water phase; and, the crosslinked and hydrophobically modified xanthan gum described herein.
[0012] A further object of the invention is a method of stabilizing an oil-in-water emulsion comprising an oil phase and a water phase comprising the step of mixing a crosslinked and hydrophobically modified xanthan gum described herein with the oil phase and the water phase.
[0013] The use of a crosslinked and hydrophobically modified xanthan gum described herein to stabilize an emulsion comprising an oil phase and a water phase, it is also an object of the present invention.
[0014] The present invention also provides personal care formulations comprising the crosslinked and hydrophobically modified xanthan of the present invention.
[0015] The present invention also provides cleaning or fabric care compositions comprising the crosslinked and hydrophobically modified xanthan of the present invention.
[0016] Another object of the invention is the provision of a method for preparing the crosslinked and hydrophobically modified xanthan gum of the present invention comprising reacting a xanthan gum with a) an alkylating agent and b) a multifunctional crosslinker comprising epoxide and/or alkyl halide functional groups.
BRIEF DESCRIPTION OF THE DRAWING
[0017] Figure 1. Amplitude sweeps for: Xanthan (native xanthan gum, not reacted), control xanthan gum that underwent same reaction conditions but without hydrophobe, and hydrophobically modified xanthan gum. CX indicates the alkyl chain length, where X = 22, 16 or 12, and the number in parentheses represents the wt.% of hydrophobe relative to the xanthan.
[0018] Figure 2. Amplitude sweeps for (A) 0.5 wt.% dispersions and (B) 1.0 wt.% dispersions of Ci6-modified xanthan gum at various crosslink levels with 1 ,4-butanediol diglycidyl ether (BDDE). Weight of crosslinker expressed as wt.% based on the total weight of the unmodified xanthan gum.
[0019] Figure 3. Amplitude sweeps of 0.5 wt.% dispersions of: A) unmodified xanthan gum; B) crosslinked and hydrophobically modified xanthan gum; C) crosslinked xanthan gum; and D) xanthan gum that underwent the same reaction conditions without crosslinker or hydrophobe.
[0020] Figure 4. Emulsions containing 0.5 wt.% of different samples of xanthan gum and 20 wt.% caprylic/capric triglyceride. A) Optical microscopy images 24 h after blending and B) photographs after aging each emulsion for four weeks at 50 °C. 1 , Hydrophobic and crosslinked xanthan gum; 2, Not hydrophobic; crosslinked xanthan gum; 3, not hydrophobic, not crosslinked xanthan gum.
[0021] Figure 5. Amplitude sweeps of 0.5 wt.% hydrophobically modified xanthan dispersions with various alkyl chain lengths (C16, C12 or C8). Each polymer has molar substitution (MS) = 0.025.
[0022] Figure 6. Amplitude sweeps for 0.5 wt.% dispersions containing crosslinked and hydrophobically modified xanthan gum at various molar substitution (MS) levels of Ci6. Xanthan gum was crosslinked with 0.5 wt.% 1 ,4-butanediol diglycidyl ether (BDDE).
[0023] Figure 7. Microscope image showing oil drop size in cleansing formulations. A. Comparative example; B. Formulation containing the modified xanthan gum of the present invention.
[0024] Figure 8. Microscope image showing oil drop size in cleansing formulations. A. Comparative example; B. Formulation containing the modified xanthan gum of the present invention.
DETAILED DESCRIPTION OF THE INVENTION
[0025] Various preferred features and embodiments will be described below by way of non-limiting illustration. Modifications, adaptations or variations of such exemplary embodiments described herein may become apparent to those skilled in the art as such are disclosed.
[0026] The articles "a" and "an" are used herein to refer to one or to more than one (i.e., to at least one) of the grammatical object of the article unless the context clearly indicates otherwise. By way of example, "an element" means one element or more than one element.
[0027] Except in the Examples, or where otherwise explicitly indicated, all numerical quantities in this description specifying amounts of materials, reaction conditions, molecular weights, number of carbon atoms, and the like, are to be understood as modified by the word "about". The term “about” as used herein, e.g. when referring to a measurable value (such as an amount or weight of a particular component or temperature), refers to variations of ±20%, ±10%, ±5%, ±1 %, ±0.5%, or, particularly, ±0.1% of the specified amount. Except where otherwise indicated, all numerical quantities in the description specifying amounts or ratios of materials are on a weight basis.
[0028] Unless otherwise stated, all percentages, parts, and ratios expressed herein are based upon weight of the total compositions of the present invention.
[0029] As used herein, the term “comprising”, which is inclusive or open-ended and does not exclude additional unrecited elements or method steps, is intended to encompass as alternative embodiments, the phrases “consisting essentially of” and “consisting of” where “consisting of’ excludes any element or step not specified and “consisting essentially of” permits the inclusion of additional unrecited elements or steps that do not materially affect the essential or basic and novel characteristics of the composition or method under consideration.
[0030] The term “personal care” as used herein includes, without being limited thereto, cosmetics, toiletries, cosmeceuticals, beauty aids, insect repellents, sunscreens,
UV absorbers, hand sanitizers, personal hygiene and cleansing products (e.g., shampoos, conditioning shampoos, anti-dandruff shampoos, body washes, hand soaps, facial scrubs, and the like) applied to the body, including the skin, hair, scalp, and nails of humans and mammals
[0031] In one aspect the invention provides a crosslinked and hydrophobically modified xanthan gum, wherein a portion of the hydrogen atoms of pendant hydroxyl groups of the backbone are substituted with at least one hydrophobic moiety according to formula (I):
(I) - (A)a-(O)b-R wherein A is selected from a divalent linear or branched, substituted or unsubstituted C1- C6 alkylene radical; R is selected from a hydrocarbon group having from 8 to 22 carbon atoms; a is 0 or 1 , and b is 0 or 1 , with the proviso that when a is 0, b is 0; and, wherein the xanthan gum is crosslinked with a multifunctional crosslinking agent comprising epoxy and/or alkyl halide functional groups in an amount of from 0.1 to 1 wt.% based on the total weight of the xanthan gum.
[0032] The crosslinked and hydrophobically xanthan gum of the present invention can be produced from readily commercially available xanthan gum. Non-modified xanthan gum used as raw material is also herein referred as “native xanthan gum”, “unmodified xanthan gum” or generically as “xanthan gum”. Non-limiting examples of xanthan gum are commercially available from Jungbunzlauer, Fufeng, Jianlong or CP Kelco (under Keltrol® trademark).
[0033] For illustrative purposes xanthan gum structure is schematically represented by the following structure:
wherein M+= Na, K+, 1 Ca2+.
[0034] Xanthan gum is typically obtained by a fermentation process using Xanthomonas campestris bacteria. The molecular weight of native xanthan gum is typically in the range from (2 x 105 to 20 x 106 Da).
[0035] The performance increasing coalescence and stability of emulsions, viscosity, and suitable texture of formulations provided by the crosslinked and hydrophobically modified xanthan gum of the present invention represents a balance between the hydrophobic modifications and crosslinking.
Hydrophobic modifications
[0036] A portion of the hydrogen atoms of the pendent hydroxyl groups of the xanthan gum according to the present invention are substituted with at least one hydrophobic moiety according to formula (I):
(I) - (A)a-(O)b-R wherein A is selected from a divalent linear or branched, substituted or unsubstituted C1- C6 alkylene radical, R is selected from a hydrocarbon group having from 8 to 22 carbon atoms, a is 0 or 1 , and b is 0 or 1 , with the proviso that when a is 0 b is 0.
[0037] In one embodiment, a is 1 and b is 1.
[0038] The hydroxyl groups of each sugar unit of the xanthan gum can be hydrophobically derivatized with hydrophobic moieties according to formula (I). The modified xanthan can have a degree of molar substitution (MS) in a range from 0.001 to 0.5, particularly from 0.01 to 0.4, more particularly from 0.025 to 0.3 (mol I molsugar). The degree of molar substitution can be measured according to the techniques well known to a person skilled in art, such as, for example, performing hydrolysis following by gas chromatography.
[0039] The divalent alkylene radical A can particularly be represented by formula (II) or (III):
[0040] More particularly, the divalent alkylene radical A can be an alkylene radical according to formula (II).
[0041] The hydrocarbon group can be a saturated or unsaturated alicyclic group, a saturated or unsaturated aliphatic group, or an aromatic group having from 8 to 22 carbon atoms. The hydrocarbon group can be linear or branched. The hydrocarbon group can particularly be a linear hydrocarbon group, i.e., the hydrocarbon group is not branched. The hydrocarbon group can particularly be a saturated hydrocarbon group. In one embodiment, the hydrocarbon group is a linear saturated hydrocarbon group.
[0042] Of particular interest are hydrocarbon groups having from 12 to 16 carbon atoms, which provide better coalescence ratings when the modified xanthan gum is used in emulsions. In one embodiment, the hydrocarbon group is a linear saturated hydrocarbon group having from 12 to 16 carbon atoms, particularly 16 carbon atoms.
[0043] In one embodiment, the hydrogen atoms of the pendent groups of the xanthan gum are substituted with at least one hydrophobic moiety according to the following formula (IV):
wherein n is an integer from 7 to 21 , particularly from 11 to 15. More particularly, n is 15. [0044] The hydrophobic modification entails alkylation by reacting pendant hydroxyl groups present in the backbone of xanthan gum or derivative thereof with an alkylation agent. Without being bound to the theory it is believed that alkylation agent reacts with the hydrogen atoms of the C2, C3, C4, and/or the C6 hydroxyls of the xanthan gum. For example, the alkylating agent can react with the hydroxyl groups on the C-2, C-3, and/or C-6 carbon atoms of the glucose units, and/or the C-2 and/or C-3 carbon atoms of the glucuronic acid units, and/or the C3 and C4 carbon atoms of the mannose units of the xanthan gum. Thus, the hydrogen atoms of the pendent hydroxyl groups of the xanthan gum that are substituted with at least one hydrophobic moiety can be the hydrogen atoms of the C-2, C-3, and/or C-6 hydroxyls of the glucose units, and/or the C-2 and/or C-3, hydroxyls of the glucuronic acid units, and/or the C2, C3, C4 and/or C6 hydroxyls of the mannose units of the xanthan gum. While not wishing to be bound by any theory, it is contemplated that probable reaction can occur at one or more positions. Typically, the hydrogen atoms of the hydroxyl groups of the xanthan gum that are substituted with at
least one hydrophobic moiety are the hydrogen atoms of at least the C-6 hydroxyls of the D-glucose units.
[0045] As defined here and throughout the specification an "alkylating" or "alkylation agent” is a reactive compound containing a hydrocarbon group that can be reacted with a pendant hydroxyl group on the xanthan gum to form an ether linkage with the xanthan gum as the starting material.
[0046] Typical alkylation agents reactive with the pendant xanthan gum hydroxyl groups include alkyl halides, epoxides, and glycidyl ethers that contain a hydrocarbon group.
[0047] The alkylation agent can be an alkyl halide represented by formula (V) below: (V) X-R1 wherein X is a halogen atom selected from the group consisting of bromine, chlorine, fluorine, and iodine; and R1 is a hydrocarbon group selected from cycloalkyl, linear or branched alkyl, cycloalkenyl, linear or branched alkenyl, aryl, alkylaryl, alkenylaryl, and combinations thereof, wherein the hydrocarbon group contains from 8 to 22 carbon atoms. Exemplary alkyl halide alkylating agents include, but are not limited to, octyl, decyl, dodecyl, myristyl, hexadecyl, stearyl and behenyl bromides, fluorides, chlorides, and iodides. Particularly, the alkylating agent can be an alkyl halide wherein the alkyl group is a linear or branched C8 to C22 alkyl group, particularly a C12 to C16 alkyl group, more particularly a C16 alkyl group. In one embodiment the alkyl halide is an alkyl halide wherein the alkyl group is a linear C8 to C22 alkyl group, particularly a linear C12 to C16 alkyl group, more particularly a linear C16 alkyl group. Particularly, the halide can be chloride or bromide, more particularly can be a chloride.
[0048] In one embodiment, the alkyl halide is selected from the group consisting of dodecyl, myristyl and hexadecyl chloride.
[0049] Of particular interest in the context of this invention as alkylation agents are glycidyl ethers represented by formula (VI) as follows:
[0050] Wherein m is an integer from 1 to 4, n is 0 or 1 ; and Ri is selected from a hydrocarbon group having from 8 to 22 carbon atoms. Particularly, said hydrocarbon group can have from 12 to 16 carbon atoms. The hydrocarbon group can particularly be a saturated hydrocarbon group, more particularly a saturated linear hydrocarbon group.
[0051] Representative glycidyl ethers set forth under formula (VI) useful as alkylation agents include but are not limited to 2-(1 ,1-dimethylethyl)-2-(phenylmethyl)-oxirane, 2-[2- (4-chlorophenyl)ethyl]-2-(1 ,1-dimethylethyl)-oxirane, 1 ,2-epoxyoctane, 1 ,2-epoxydecane, 1 ,2-epoxy dodecane, 1 ,2-epoxytetradecane, 1 ,2-epoxy hexadecane, 1 ,2- epoxyoctadecane, 1 ,2-epoxyeicosane, 1 ,2-epoxyethylbenzene, 2-ethylhexyl glycidyl ether, dodecyl glycidyl ether, tetradecyl glycidyl ether, hexadecyl glycidyl ether, behenyl glycidyl ether, glycidyl triphenylmethyl ether, nonylphenyl glycidyl ether. The hydrocarbon group can particularly be a linear hydrocarbon group, i.e. the hydrocarbon group is not branched.
[0052] In particular, the alkylation agent is a glycidyl ether represented by formula (VI) wherein m is 1 and n is 1 and Ri is a hydrocarbon group having from 8 to 22 carbon atoms, particularly from 12 to 16 carbon atoms. Preferably the hydrocarbon group is a linear saturated hydrocarbon group. In one embodiment, the glycidyl ether is selected from the group consisting of dodecyl glycidyl ether, tetradecyl glycidyl ether and hexadecyl glycidyl ether. More particularly, the alkylation agent is hexadecyl glycidyl ether.
Crosslinking agents
[0053] The xanthan gum according to the invention is crosslinked with a multifunctional crosslinking agent comprising epoxy and/or alkyl halide functional groups. The amount of crosslinking agent can be from 0.1 to 1 wt.% or from 0.2 to 1 wt. %, particularly from 0.2 to 0.9 wt.%, more particularly from 0.4 to 0.6 wt.% based on the total weight of the xanthan gum.
[0054] The multifunctional crosslinking agent can be a homo-bifunctional crosslinking agent or hetero-bifunctional crosslinking agent.
[0055] The multifunctional crosslinking agent can be selected from the group consisting of polyglycidyl ethers, multifunctional alkyl halides and epihalohydrin.
[0056] Suitable polyglycidyl ethers as crosslinking agents in the context of this invention include, but are not limited to, glycerol diglycidyl ether, polyethylene glycol diglycidyl ether, polyoxyethylene glycol diglycidyl ether, neopentyl glycol diglycidyl ether, 1 ,4-butanediol diglycidyl ether (BDDE), 1 ,6-hexanediol diglycidyl ether, glycerol diglycidyl ether, trimethylolpropane diglycidyl ether, trimethylolpropane triglycidyl ether, glycerol triglycidyl ether, pentaerythritol diglycidyl ether, pentaerythritol triglycidyl ether, pentaerythritol tetraglycidyl ether, sorbitol diglycidyl ether, sorbitol triglycidyl ether, sorbitol tetraglycidyl ether, sorbitol penta glycidyl ether, and sorbitol hexaglycidyl ether, resorcinol
diglycidyl ether, bisphenol A diglycidyl ether (BADGE), and bisphenol F diglycidyl ether (NOGE).
[0057] Particularly, the polyglycidyl ether can be a diglycidyl ether. More particularly, the polyglycidyl ether can be 1,4-butanediol diglycidyl ether (BDDE).
[0058] In some embodiments, the crosslinking agent can be polypropylene glycol) diglycidyl ether.
[0059] Suitable alkyl halides as multifunctional crosslinking agents in the context of the present invention include, but are not limited to, ethyl, propyl, isopropyl, n-butyl, t- butyl, pentyl, neopentyl, hexyl, octyl, decyl, dodecyl, myristyl, hexadecyl, stearyl and behenyl dibromides, dichlorides, and di-iodides. Particularly, the crosslinking agent can be a multifunctional alkyl halide wherein the alkyl group is a linear or branched C2 to C22 alkyl group.
[0060] In one embodiment, the crosslinking agent can be 1 ,4-dichorobutane.
[0061] In one embodiment, the multifunctional crosslinking agent is an epihalohydrin, more particularly an epichlorohydrin.
[0062] In synthesizing the crosslinked and hydrophobically modified xanthan gum described herein, the alkylating agent and the crosslinker will be capable to react with the hydroxyl groups present in the saccharide units after deprotonating with an alkali agent. For example, the alkylating agent and the crosslinker can react with the hydroxyl groups on the C-2, C-3, and/or C-6 carbon atoms of the glucose units, and/or the C-2 and/or C- 3, carbon atoms of the glucuronic acid chain units, and/or the C2, C3, 04, and/or 06 carbon atoms of the mannose chain units of the xanthan gum. While not wishing to be bound by any theory, it is contemplated that probable reaction can occur at one or more positions.
[0063] The invention also provides a method for preparing a crosslinked and hydrophobically modified xanthan gum described herein comprising reacting a xanthan gum with a) an alkylating agent and b) a multifunctional crosslinker comprising epoxide and/or alkyl halide functional groups.
[0064] The crosslinking and hydrophobic modification can be conducted in any order, or simultaneously, as well as repeated, to produce the desired crosslinked and hydrophobically modified xanthan gum. However, one of the advantages of the present invention is that the crosslinking and hydrophobic modifications are performed simultaneously, i.e., the reaction with the alkylating agent and the multifunctional crosslinker is conducted simultaneously.
[0065] In particular, the method for preparing the crosslinked and hydrophobically modified xanthan gum can comprise the steps of: i) Mixing a xanthan gum with an organic solvent or a mixture of an organic solvent and water to form a suspension or slurry; ii) Increase the pH value of the suspension or slurry to be higher than 9 with an alkaline solution; iii) Adding an alkylating agent and a multifunctional crosslinker comprising epoxide and/or alkyl halide functional groups; iv) Reacting said reaction mixture for at least 2h at a temperature from 50 to 100°C to obtain a crosslinked and hydrophobically modified xanthan gum; v) Neutralizing the crosslinked and hydrophobically modified xanthan gum with an acid.
[0066] The xanthan gum used as starting material is typically in powder form and is normally treated with an organic solvent or a mixture of organic solvent and water thereby obtaining a suspension or slurry. The amount of xanthan gum in the organic solvent or the mixture of organic solvent and water (i.e., the suspension or slurry) can be particularly in the range from 10 to 80 wt.%, more particularly from 15 to 30 wt.% based on the total weight of the suspension or slurry.
[0067] In practice, the organic solvent used can be acetone, methanol, ethanol, isopropanol, n-propyl alcohol, n-butyl alcohol, isobutyl alcohol, t-butyl alcohol, or the like, and mixtures thereof. Particularly, the organic solvent is isopropanol. In particular, the amount of organic solvent when combined with water can be from 50 to 80 wt.%, more particularly from 55 to 75 wt.%. In one embodiment, the organic solvent combined with water can comprise from 50 to 80 wt.%, particularly from 55 to 75 wt.%, more particularly 60 wt.% of isopropanol in water. Advantageously, concentrations in the range from 55 to 65 wt.% allow to simultaneously add the alkylating agent and the multifunctional crosslinker, while controlling particle swelling, which affect the extent and location of modifications.
[0068] In particular, the pH in step ii) is increased to be 10 or higher, particularly from 10 to 13, particularly from 11 to 13. The pH is typically adjusted with an alkali such as sodium hydroxide, potassium hydroxide, calcium hydroxide, lithium hydroxide, and ammonium hydroxide, for example. In one embodiment, the alkali is sodium hydroxide. The amount of alkali used is the amount necessary for increasing the pH to the desired values.
[0069] Once an alkylating agent and a multifunctional crosslinker are added to the suspension or slurry comprising the xanthan gum, the mixture is typically allowed to react for a period of time sufficient to ensure complete reaction of the reactants. The mixture can be stirred to improve the reaction. The reaction time and the temperature may be adjusted. In particular, the mixture can be allowed to react from 2h to 5h, or from 2h to 4h, or preferably for 3h. The reaction temperature can particularly be from 55 to 65 °C. Preferably, the reaction temperature can be 60°C. In one embodiment the mixture is allowed to react for from 2h to 4h at 55 to 65°C. Advantageously, the reaction can be conducted in the presence of nitrogen atmosphere.
[0070] After reaction, the modified xanthan gum is neutralized. The neutralization is normally adjusted to a pH which is within a range of from about 4 to about 10, or from about 6 to about 8, or about 7. Any acid may be selected to neutralize the solution, including strong acids such as hydrochloric acid and sulfuric acid or weak acids such as acetic acid, citric acid, carbon dioxide (carbonic acid), trifluoroacetic acid, etc. In one embodiment either hydrochloric or acetic acid is used. The amount of acid used is the amount necessary for neutralization.
[0071] The modified xanthan gum obtained after reaction in the presence of alkylating agent and a multifunctional crosslinker can be subsequently separated from the solvent, in particular by physical separation methods. Non-limiting examples of separating techniques include filtering, centrifuging and the like. In particular, the solvent can be removed by filtration. The remaining solid can be further washed with an organic solvent or a mixture of organic solvent and water. In particular, the remaining solid is washed with a mixture of organic solvent and water comprising from 60 to 90 wt. %, or from 75 to 85 wt.%, or about 80 wt.% of organic solvent in water. Non-limiting organic solvents that can be used are acetone, methanol, ethanol, isopropanol, n-propyl alcohol, n-butyl alcohol, isobutyl alcohol, t-butyl alcohol, and the like. Mixtures of these alcohols can also be used. Preferably, isopropanol in water is used.
[0072] The volume of the wash liquid is much greater than the amount of modified xanthan and can be performed in batch wise or multiple applications. Typically, 1 to 4 independent wash cycles are completed. However, additional wash cycles can be used if needed.
[0073] The modified xanthan gum can be dried either directly after separating the xanthan gum from the reaction medium or subsequent washing to remove the remaining solvent. Examples of drying techniques include air drying, evaporative drying, vacuum drying, freeze drying, fluidized bed and the like. In particular, drying at temperatures from
50 to 115°C under ambient conditions or reduced pressure can be advantageous. Advantageously, the drying temperature can range from 60 to 80°C. It has been surprisingly found that drying at temperatures in the range from 60 to 80°C improves the viscosity, yield values and stability of formulations and compositions comprising the modified xanthan gum according to the invention. More particularly, the temperature can be 60, 70 or 80°C. The drying times can range from 1 to 4 hours. Advantageously, the drying step is conducted at 80°C for 4 hours, particularly under vacuum conditions.
Applications, compositions and products
[0074] The crosslinked and hydrophobically modified xanthan gum of the present invention can be formulated in compositions for personal care products, topical health care products, household care products, institutional and industrial (l&l) products, and fabric care products.
[0075] Of particular interest in the context of the present invention are oil-in-water emulsions (O/W). Thus, one object of the present invention is the provision of an oil-in- water emulsion comprising: a water phase; an oil phase dispersed in said water phase; and, a crosslinked and hydrophobically modified xanthan gum according to the present invention.
[0076] The invention also provides a method of stabilizing an oil-in-water emulsion comprising an oil phase and a water phase comprising the step of mixing the crosslinked and hydrophobically modified xanthan gum according to the present invention with the oil phase and the water phase.
[0077] The invention also provides for the use of a crosslinked and hydrophobically modified xanthan gum according to the invention to stabilize an emulsion comprising an oil phase and a water phase.
[0078] As used herein, the term “oil” means a compound or a mixture of compounds that is insoluble in water and has a liquid appearance at a temperature of 25°C. The oil phase is not restricted to a specific oil. Any oil suitable for cosmetic or personal care formulations can be used, including but not limited to, vegetable oils, oils derived from petroleum (e.g., mineral oils, liquid paraffin), fatty esters, etc.
[0079] The oil phase may comprise at least one of an ester oil, vegetable oil, alcohol, paraffin oil or silicone. The oil phase can comprise one or more oils chosen from mineral oils such as paraffin oil, petroleum jelly, isoparaffins or white mineral oils; oils of animal
origin, such as squalene or squalane; vegetable oils, such as phytosqualane, sweet almond oil, coconut oil, castor oil, jojoba oil, olive oil, rapeseed oil, peanut oil, sunflower oil, wheat germ oil, corn germ oil, soybean oil, cottonseed oil, alfalfa oil, poppy oil, pumpkin oil, evening primrose oil, millet oil, barley oil, rye oil, safflower oil, bancoulier oil, passionflower oil, hazelnut oil, palm oil, shea butter, apricot kernel oil, coriander seed oil, beech oil, calophyllum oil, sisymbrium oil, avocado oil, calendula oil, oils from flowers or vegetables ethoxylated vegetable oils, algae oils; synthetic oils such as fatty acid esters such as butyl myristate, propyl myristate, isopropyl myristate, cetyl myristate, isopropyl palmitate, octyl palmitate, butyl stearate, hexadecyl stearate, isopropyl stearate, isopropyl isostearate, octyl stearate, isocetyl stearate, dodecyl oleate, hexyl laurate, propylene glycol dicaprylate, esters derived from lanolic acid, such as isopropyl lanolate, isocetyl lanolate, monoglycerides, diglycerides and triglycerides of fatty acids such as glycerol triheptanoate, alkylbenzoates, hydrogenated oils, poly (alpha-olefins), polyolefins such as poly (isobutane), synthetic isoalkanes such as isohexadecane, isododecane, perfluorinated oils; silicone oils such as dimethylpolysiloxanes, methylphenylpolysiloxanes, amino-modified silicones, fatty acid-modified silicones, alcohol-modified silicones, alcohol-modified silicones and fatty acids, silicone-modified silicones polyether groups, modified epoxy silicones, silicones modified by fluorinated groups, cyclic silicones and silicones modified by alkyl groups.
[0080] In one non-limiting embodiment, the oil phase comprises at least an oil selected from the group consisting of fatty acid esters, monoglycerides, diglycerides and triglycerides of fatty acids and mixtures thereof.
[0081] The fatty acid ester can be particularly selected from the group consisting of butyl myristate, propyl myristate, isopropyl myristate, cetyl myristate, isopropyl palmitate, octyl palmitate, butyl stearate, hexadecyl stearate, isopropyl stearate, isopropyl isostearate, octyl stearate, isocetyl stearate, dodecyl oleate, hexyl laurate, propylene glycol dicaprylate, and esters derived from lanolic acid, such as isopropyl lanolate, isocetyl lanolate, and combinations thereof. More particularly, the fatty acid ester can be isopropyl stearate, isopropyl isostearate.
[0082] The oil phase can comprise an oil selected from the group consisting of triglycerides of fatty acids, isostearyl isostearate, and isohexadecane, and combinations thereof. More particularly, the triglyceride of fatty acid can be caprylic/capric triglyceride or triolein.
[0083] The oil phase may be present in an amount of from 1 to 50 wt.% based on the total weight of the emulsion. The oil phase can be present from 1 to 40 wt.%, particularly
5 to 40 wt.%, more particularly from 10 to 40 wt.%, even more particularly from 20 to 30 wt.% based on the total weight of the emulsion.
[0084] The oil phase can also contain other components that are oil soluble. The oil phase can comprise at least 90 wt.%, at least 95 wt.%, or at least 98 wt.%, of one or more than one oils, based on the total weight of the oil phase.
[0085] The emulsion of the invention comprises a water phase, which constitutes the continuous phase. The water phase comprises water and can contain other components that are water soluble. The water phase can comprise at least 90 wt.%, at least 95 wt.%, or at least 98 wt.% water, based on the total weight of the water phase.
[0086] Typically, the modified xanthan gum of the present invention is in an amount ranging from 0.01 to 2 wt.% based on the total weight of the emulsion. In particular, the modified xanthan gum can be in a concentration from 0.05 to 2 wt.%, or from 0.1 to 1.5 wt.%, or from 0.2 to 1 .5 wt.%, or from 0.5 to 1 .25 wt.% of the based on the total weight of the emulsion.
[0087] The emulsion can further comprise co-emulsifiers or other surfactants. Exemplary co-emulsifier include, but are not limited to, Cetearyl Glucoside [HLB = 11 ± 1], Polysorbate 85 [HLB = 11 ± 1], Glyceryl Stearate (and) PEG-100 Stearate [HLB = 11 ± 1], Stearamide MEA [HLB = 11 ± 1], Oleth-2 [HLB = 4.9 ± 1], Oleth-10 I Polyoxyl 10 Oleyl Ether NF [HLB = 12.4 ± 1], Oleth-10 [HLB = 12.4 ± 1], Oleth-20 [HLB = 12.4 ± 1], Ceteth-10 [HLB = 12.9 ± 1], PEG-8 Laurate [HLB = 13 ± 1], Cocamide MEA [HLB = 13.5 ± 1], Polysorbate 60 [HLB = 14.9 ± 1], PEG-60 Almond Glycerides [HLB = 15 ± 1], lsosteareth-20 [HLB = 15 ± 1], Lauramide DEA [HLB = 15 ± 1], Polysorbate 80 [HLB = 15 ± 1], PEG-20 Methyl Glucose Sesquistearate [HLB = 15 ± 1], PEG-4 Dilaurate [HLB = 6 ± 1], Ceteareth-20 [HLB = 15.2 ± 1], Sorbitan Stearate (and) Sucrose Cocoate [HLB = 6 ± 1], Methyl Glucose Sesquistearate [HLB = 6.6 ± 1], Steareth-21 [HLB = 15.5 ± 1], Sodium Stearoyl Lactylate [HLB = 8.3 ± 1], Ceteth-20 [HLB = 15.7 ± 1], Sorbitan Laurate [HLB = 8.6 ± 1], lsoceteth-20 [HLB = 15.7 ± 1], PEG-40 Sorbitan Peroleate [HLB = 9 ± 1], Polysorbate 20 [HLB = 16.7 ± 1], Lecithin [HLB = 9.7 ± 1], Laureth-23 [HLB = 16.9 ± 1], Laureth-4 [HLB = 9.7 ± 1], PEG-100 Stearate [HLB = 18.8 ± 1], PEG-20 Almond Glycerides [HLB = 10 ± 1], PEG-80 Sorbitan Laurate [HLB = 19.1 ± 1], Linoleamide DEA [HLB = 10 ± 1], PEG-25 Hydrogenated Castor Oil [HLB = 10.8 ± 1], (Poly)glyceryl fatty acids and amino acid-based surfactants, Calcium Stearoyl Lactylate [HLB = 5.1 ± 1], Ceteth-2 [HLB = 5.3 ± 1], Glyceryl Laurate [HLB = 5.2 ± 1], Glyceryl Stearate [HLB = 3.8 ± 1], Glyceryl Stearate SE [HLB = 5.8 ± 1], Glycol Distearate [HLB = 1 ± 1], Glycol Stearate [HLB = 2.9 ± 1], PEG-30 Dipolyhydroxystearate [HLB = 5.5 ± 1], PEG-60 Almond
Glycerides [HLB = 15 ± 1], Polysorbate 60 [HLB = 14.9 ± 1], Sorbitan Isostearate [HLB = 4.7 ± 1], Sorbitan Oleate [HLB = 4.3 ± 1], Sorbitan Sesquioleate [HLB = 3.7 ± 1], Sorbitan Stearate [HLB = 4.7 ± 1], Sorbitan Trioleate [HLB = 1 .8 ± 1] and Steareth-2 [HLB = 4.9 ± 1].
[0088] Of particular interest are co-emulsifiers having a HLB value (Hydrophilic Lipophilic Balance) equal or higher than 6, which can improve the coalesce of emulsions and the tolerance to electrolytes. Particularly, the HLB value is equal or higher than 7, or is equal or higher than 8, or is equal or higher than 10.
[0089] In particular, the co-emulsifier can be selected from the group consisting of amino acid-based surfactants and (poly)glyceryl fatty acids. More particularly, the coemulsifier is selected from the group consisting of sodium stearoyl glutamate, polyglyceryl-3 Laurate, and polyglyceryl- 10 Laurate.
[0090] One of the beneficial effects of the present technology is that due to the enhanced properties of the modified xanthan gum of the present invention, low amounts of co-emulsifiers are needed to obtain stable emulsions. Thus, in one embodiment, the concentration of the co-emulsifier is in the range from 0.1 to 1 wt.% based on the total weight of the emulsion.
[0091] In particular, the viscosity of the emulsions according to the invention as determined as Brookfield viscosity at 20 rpm at room temperature is in the range from 1 ,000 to 9,000 mPa s.
[0092] The invention also provides formulations or compositions for personal care and topical health care comprising the modified xanthan gum of the present invention or the oil-in-water emulsions comprising the modified xanthan gum of the present invention. [0093] The invention also provides cleaning compositions comprising the crosslinked and hydrophobically modified xanthan gum of the present invention. Typical cleaning compositions include household and industrial and institutional (l&l) cleaning compositions, formulations and products that can contain the modified xanthan gum of the present invention. Non-limiting examples include surface cleansers for kitchen and bathroom counter tops, tiled surfaces, and utilities, including appliances employed or located therein, toilet cleaners, including toilet bowl rim gels, floor cleansers, wall cleansers, polishes, air freshener gels, detergents, treatments and cleansers for dishes.
[0094] The invention also provides fabric care compositions comprising the crosslinked and hydrophobically modified xanthan gum of the present invention.
[0095] As used herein, the phrase “fabric care compositions” include compositions and formulations designed for treating fabric. Such compositions include, but are not
limited to, laundry cleaning compositions and detergents, fabric softening compositions, fabric enhancing compositions, fabric freshening compositions, laundry prewash, laundry pretreat, laundry additives, dry cleaning agent or composition, laundry rinse additive, wash additive, post-rinse fabric treatment, and the like. Such compositions may be used as a pre-laundering treatment, a post-laundering treatment, or may be added during the rinse or wash cycle of the laundering operation.
[0096] Compositions for personal care and topical health care can comprise any cosmetic, toiletry, and topical pharmaceutical formulation. Typical personal care formulations that can include the modified xanthan gum according to the present invention include, without being limited thereto, shampoos, chemical and non-chemical hair curling and hair straightening products, hair style maintenance products (e.g., hair fixatives and hair color products), emulsion lotions and creams for the nails, hands, feet, face, scalp, and body, hair dyes, face and body makeup, nail care products, astringents, deodorants, antiperspirants, depilatories, skin protective creams and lotions, such as sunscreens, skin and body cleansers, skin conditioners, skin toners, skin firming compositions, liquid soaps, soap bars, bath products, shaving products, and the like.
[0097] Compositions for personal care and topical health care can be in the form of, without being limited thereto, liquids, such as rinses, gels, hydroalcoholic gels (e.g., hand sanitizers), sprays, emulsions, such as lotions and creams, shampoos, pomades, foams, ointments, tablets, sticks, such as lip care products, makeup, and suppositories, and like products, which are applied to skin and hair and remain in contact therewith until removed as by rinsing with water or washing with shampoo or soap. Gels can be soft, stiff, or squeezable.
[0098] The modified xanthan gum of the present invention is particularly suitable for body washes, antibacterial body washes, dual purpose body washes and shampoos, bath gels, shower gels, liquid hand soaps, body scrubs, bubble baths, facial scrubs, foot scrubs, and the like.
[0099] In particular, the shampoo embodiments of the present invention can be formulated as 2-in-1 shampoos, baby shampoos, conditioning shampoos, bodyfying shampoos, moisturizing shampoos, temporary hair color shampoos, 3-in-1 shampoos, anti-dandruff shampoos, hair color maintenance shampoos, acid (neutralizing) shampoos, medicated shampoos, and salicylic acid shampoos, and the like.
[00100] The amount of modified xanthan gum of the present invention that can be employed in the foregoing compositions can be determined by the person skilled in the formulation art.
[00101] When used in personal care compositions, the modified xanthan gum is typically used in a concentration from 0.001 to 10 wt.%, or from 0.005 to 5 wt.%, or from 0.01 to 2 wt.%, 0.1 to 1.5 wt.%, or from 0.2 to 1 wt.%. or from 0.3 to 0.8 wt.% based on the total weight of the personal care composition.
[00102] The exemplary compositions, formulations and products of the invention can typically contain various additives and conventional adjuvants as are well known in the art, including, without being limited thereto, acidifying or alkalizing pH adjusting agents and buffering agents; fixatives and auxiliary film formers, such as gums, resins, polymers of synthetic or natural origin, and the like; auxiliary rheology modifiers, such as viscosity increasing polymeric thickeners or gellants, additives, such as emulsifiers, emulsion stabilizers, waxes, dispersants, and the like, and viscosity control agents, such as solvents, electrolytes, and the like; hair and skin conditioning agents, such as antistatic agents, synthetic oils, vegetable or animal oils, silicone oils, monomeric or polymeric quaternized ammonium salts, emollients, humectants, lubricants, sunscreen agents, disinfectants, antimicrobials, and the like; chemical hair waving or straightening agents; hair colorants, such as pigments and dyes for temporary, semipermanent, or permanent hair dyeing; surfactants, such as anionic, cationic, nonionic, amphoteric and zwitterionic surfactants; polymer film modifying agents, such as plasticizers, humectants, tackifiers, detackifiers, wetting agents and the like, product finishing agents, such as chelating agents, opacifiers, pearlescing agents, preservatives, fragrances, solubilizers, colorants, such as pigments and dyes, UV absorbers, and the like; propellants (water-miscible or water-immiscible), such as fluorinated hydrocarbons, liquid volatile hydrocarbons, compressed gases, and the like; and mixtures thereof.
[00103] An extensive listing of personal care and cosmetic ingredients and their functions, for example, appears in the INCI Dictionary, generally, and in Vol. 2, Section 4 of the Seventh Edition. Those skilled in the art of formulating personal care and health care products recognize that some ingredients are multifunctional and, hence, can serve more than one purpose in the formulation.
[00104] The compositions comprising the modified xanthan gum according to the present invention can further include alkaline or acidifying compounds for adjusting the pH (i.e. , pH adjusting materials).
[00105] Buffering agents can be used in the exemplary compositions. Suitable buffering agents include alkali or alkali earth metal carbonates, phosphates, bicarbonates, citrates, borates, acetates, acid anhydrides, succinates, and the like, such as sodium phosphate, sodium citrate, sodium acetate, sodium bicarbonate, and sodium carbonate.
[00106] The amount of buffering agent utilized is dependent upon the desired pH range to be buffered or maintained. Such amounts can readily be determined by the skilled formulator.
[00107] Surfactants can be used in exemplary compositions of the invention. Suitable anionic surfactants include but are not limited to alkyl sulfates, alkyl ether sulfates, alkyl sulfonates, alkaryl sulfonates, a-olefin-sulfonates, alkylamide sulfonates, alkarylpolyether sulphates, alkylamidoether sulfates, alkyl monoglyceryl ether sulfates, alkyl monoglyceride sulfates, alkyl monoglyceride sulfonates, alkyl succinates, alkyl sulfosuccinates, alkyl ether sulfosuccinates, alkyl sulfosuccinamates, alkyl amidosulfosuccinates; alkyl sulfoacetates, alkyl phosphates, alkyl ether phosphates, alkyl ether carboxylates, alkyl amidoethercarboxylates, acyl lactylates, alkyl isethionates, acyl isethionates, carboxylate salts, fatty acid soaps, and amino acid derived surfactants such as N-alkyl amino acids, N-acyl amino acids, as well as alkyl peptides. Examples of anionic surfactants also include the sodium, potassium, lithium, magnesium, and ammonium salts of laureth sulfate, trideceth sulfate, myreth sulfate, C12-C13 pareth sulfate, C12-C14 pareth sulfate, and C12- C15 pareth sulfate, ethoxylated with 1 , 2, and 3 moles of ethylene oxide; the sodium, potassium, lithium, magnesium, ammonium, and triethanolammonium salts of lauryl sulfate, coco sulfate, tridecyl sulfate, myristyl sulfate, cetyl sulfate, cetearyl sulfate, stearyl sulfate, oleyl sulfate, and tallow sulfate, disodium lauryl sulfosuccinate, disodium laureth sulfosuccinate, sodium cocoyl isethionate, sodium lauroyl isethionate, sodium lauroyl methyl isethionate, sodium C12-C14 olefin sulfonate, sodium laureth-6 carboxylate, sodium dodecylbenzene sulfonate, triethanolamine monolauryl phosphate, and fatty acid salts (soaps), including the sodium, potassium, ammonium, and triethanolamine salts of a saturated and unsaturated fatty acids containing from about 8 to about 22 carbon atoms. [00108] Exemplary compositions in the context of the present invention can include amino acid surfactants. Suitable amino acid surfactants include but are not limited to taurates, glutamates, alanine, alaninates, sacosinates, aspartates, glycinates, and mixtures thereof.
[00109] Suitable cationic surfactants of the present technology include but are not limited to alkylamines, amidoamines, alkyl imidazolines, ethoxylated amines, quaternary compounds, and quaternized esters. In addition, alkylamine oxides can function as a cationic surfactant at a lower pH value.
[00110] Suitable zwitterionic or amphoteric surfactants according to the invention include, but are not limited to, betaine-based surfactants, sultaine-based surfactants, amphocarboxylate-based surfactants, amine oxides and combinations thereof.
[00111] Suitable nonionic surfactants in the context of the present invention include, but are not limited to, aliphatic (Ce-Cis) primary or secondary linear or branched chain acids, alcohols or phenols; alkyl ethoxylates; alkyl phenol alkoxylates (especially ethoxylates and mixed ethoxy/propoxy moieties); block alkylene oxide condensates of alkyl phenols; alkylene oxide condensates of alkanols; and ethylene oxide/propylene oxide block copolymers. Other suitable nonionic surfactants include mono- or dialkyl alkanolamides; alkyl polyglucosides (APGs); sorbitan fatty acid esters; polyoxyethylene sorbitan fatty acid esters; polyoxyethylene sorbitol esters; polyoxyethylene acids, and polyoxyethylene alcohols. Other examples of suitable nonionic surfactants include coco mono- or diethanolamide, coco glucoside, decyl diglucoside, lauryl diglucoside, coco diglucoside, polysorbate 20, 40, 60, and 80, ethoxylated linear alcohols, cetearyl alcohol, lanolin alcohol, stearic acid, glyceryl stearate, PEG-100 stearate, laureth 7, and oleth 20. Non-ionic surfactants can include, but are not limited to, alkoxylated methyl glucosides such as, for example, methyl gluceth-10, methyl gluceth-20, PPG-10 methyl glucose ether, and PPG-20 methyl glucose ether, available from Lubrizol Advanced Materials, Inc., under the trade names, Glucam® E10, Glucam® E20, Glucam® P10, and Glucam® P20, respectively; and hydrophobically modified alkoxylated methyl glucosides, such as PEG 120 methyl glucose dioleate, PEG-120 methyl glucose trioleate, and PEG-20 methyl glucose sesquistearate, available from Lubrizol Advanced Materials, Inc., under the trade names, Glucamate® DOE- 120, Glucamate™ LT, and Glucamate™ SSE-20, respectively, are also suitable. Other exemplary hydrophobically modified alkoxylated methyl glucosides are disclosed in United States Patent Nos. 6,573,375 and 6,727,357, the disclosures of which are hereby incorporated by reference in their entirety.
[00112] When present, surfactants may be in a concentration of from 0.1 to 50 wt.%, or from 0.1 to 30 wt.%, or from 0.5 to 26 wt.%, or from 1 to 22 wt.%, or from 5 to 18 wt.%, or from 7 to 12 wt.% based on the total weight of the personal care composition.
[00113] Exemplary compositions in the context of the present invention can also include cationic polymers. Cationic polymers are components that can enhance the delivery and deposition of conditioning agents and/or provide auxiliary conditioning benefits to the hair, scalp, or skin to improve and enhance the conditioning benefits delivered by the compositions of the present technology. Cationic polymer refers to polymers containing at least one cationic moiety or at least one moiety that can be ionized to form a cationic moiety. Typically, these cationic moieties are nitrogen containing groups such as quaternary ammonium or protonated amino groups. The cationic protonated amines can be primary, secondary, or tertiary amines. The cationic polymer typically has
a cationic charge density ranging from about 0.2 to about 7 meq/g at the pH of the intended use of the composition. The average molecular weight of the cationic polymer ranges from about 5,000 daltons to about 10,000,000 daltons. Non-limiting examples of such polymers are described in the CTFA International Cosmetic Ingredient Dictionary/Handbook via the CTFA website.
[00114] Suitable cationic polymers can be synthetically derived or natural polymers that are synthetically modified to contain cationic moieties. Particularly, the cationic polymer contains at least one repeating unit containing a quaternary ammonium salt moiety. Preparation of quaternary ammonium salt moiety containing polymers can be found, for example, in U.S. Patent. Nos. 3,288,770; 3,412,019; 4,772,462 and 5,275,809. [00115] Suitable cationic polymers include the chloride salts of the foregoing quaternized homopolymers and copolymers in which the alkyl group is methyl or ethyl, and are commercially available under the Merquat® series of trademarks from Lubrizol Advanced Materials, Inc. A homopolymer prepared from diallyl dimethyl ammonium chloride (DADMAC) having the CTFA name, Polyquaternium-6, is available under the Merquat™ 100 and Merquat™ 106 trademark. A copolymer prepared from DADMAC and acrylamide having the CTFA name, Polyquaternium-7, is sold under the Merquat™ 550 trademark. Another copolymer prepared from DADMAC and acrylic acid having the CTFA name, Polyquaternium-22, is sold under the Merquat280 trademark.
[00116] Also useful is an ampholytic terpolymer prepared from a nonionic component derived from acrylamide or methyl acrylate, a cationic component derived from DADMAC or methacrylamidopropyl trimethyl ammonium chloride (MAPTAC), and an anionic component derived from acrylic acid or 2-acrylamido-2-methylpropane sulfonic acid or combinations of acrylic acid and 2-acrylamido-2-methylpropane sulfonic acid. An ampholytic terpolymer prepared from acrylic acid, DADMAC and acrylamide having the CTFA name, Polyquarternium-39, is available under the Merquat™ Plus 3330 and Mequat™ 3330PR trademarks. Another ampholytic terpolymer prepared from acrylic acid, methacrylamidopropyl trimethyl ammonium chloride (MAPTAC) and methyl acrylate having the CTFA name, Polyquarternium-47, is available under the Merquat™ 2001 trademark. Still another ampholytic terpolymer prepared from acrylic acid, MAPTAC and acrylamide having the CTFA name, Polyquarternium-53, is available under the Merquat™ 2003PR trademark.
[00117] Exemplary cationically modified natural polymers include polysaccharide polymers, such as cationically modified cellulose and cationically modified starch derivatives modified with a quaternary ammonium halide moiety. Exemplary cationically
modified cellulose polymers are salts of hydroxyethyl cellulose reacted with trimethyl ammonium substituted epoxide (CTFA, Polyquaternium-10). Other suitable types of cationically modified cellulose include the polymeric quaternary ammonium salts of hydroxyethyl cellulose reacted with lauryl dimethyl ammonium substituted epoxide (CTFA, Polyquaternium-24). Cationically modified potato starch having the CTFA name, Starch Hydroxypropyltrimonium Chloride, is available under the Sensomer™ CI-50 trademark, from Lubrizol Advanced Materials, Inc.
[00118] Other suitable cationically modified natural polymers include cationic polygalactomannan derivatives such as guar gum derivatives and cassia gum derivatives, e.g., CTFA: Guar Hydroxypropyltrimonium Chloride and Cassia Hydroxypropyltrimonium Chloride. Guar hydroxypropyltrimonium chloride is commercially available under the Jaguar™ trade name series from Rhodia Inc. and the N-Hance trade name series from Ashland Inc. Cassia Hydroxypropyltrimonium Chloride is commercially available under the Sensomer™ trademark from Lubrizol Advanced Materials, Inc.
[00119] The cationic and ampholytic polymers can be present from about 0.05 to about 5 wt.% percent, or from about 0.1 to about 3 wt.%, or from about 0.5 to about 2.0 wt.%, based on the total weight of the composition.
Silicones
[00120] Silicone conditioning agents can be used in compositions employing the modified xanthan gum of the present invention. The optional silicone conditioning agent may comprise a volatile silicone, a nonvolatile silicone, or combinations thereof.
[00121] Water soluble or water dispersible silicones can also be used in the disclosed technology. Such water-soluble silicones contain suitable anionic functionality, cationic functionality, and/or nonionic functionality to render the silicone water soluble or water dispersible. The water-soluble silicones can contain a polysiloxane main chain to which is grafted at least one anionic moiety. Watersoluble silicone copolyol are also useful in the practice of the disclosed technology. [00122] Dimethicone copolyols are disclosed in U.S. Patent Nos. 5, 136,063 and 5, 180,843, the disclosures of which are incorporated herein by reference. In addition, dimethicone copolyols are commercially available under the Silsoft® and Silwet® brand names from the General Electric Company (GE-OSi). Specific product designations include but are not limited to Silsoft 305, 430, 475, 810, 895, Silwet L 7604 (GE-OSi); Dow Corning® 5103 and 5329 from Dow Corning Corporation; and Abil® dimethicone copolyols, such as, for example WE 09, WS 08, EM 90 and EM
97 from Evonik Goldschmidt Corporation; and Silsense™ dimethicone copolyols, such as Silsense™ Copolyol-1 and Silsense™ Copolyol-7, available from Lubrizol Advanced Materials, Inc.
[00123] The compositions of the present technology can also include nature derived emollients to be used as a silicone alternative to cyclopentasiloxane D5 such as C9-C12 alkanes available from Lubrizol Advanced Materials, Inc. under the trademark Silsense™ Bio 5 Emollient.
[00124] The concentration of the silicone conditioning agent or silicone alternatives can range from about 0.01 % to about 20%, or about 0.05% to about 10%, from about 0.1 % to about 5%, or from about 0.2% to about 3% by wt. all based on the total weight of the composition.
[00125] Suitable conditioning oils for use as conditioning agents (e.g., for the hair, scalp, skin, and nails) that can be also incorporated in compositions of the present invention include, but are not limited to, hydrocarbon oils having at least about 10 carbon atoms, such as cyclic hydrocarbons, straight chain aliphatic hydrocarbons (saturated or unsaturated), and branched chain aliphatic hydrocarbons (saturated or unsaturated), including polymers and mixtures thereof. Straight chain hydrocarbon oils typically contain about 12 to 19 carbon atoms. Branched chain hydrocarbon oils, including hydrocarbon polymers, typically will contain more than 19 carbon atoms.
[00126] Natural oil conditioners are also useful in the practice of the present invention technology, and include, but are not limited to, peanut, sesame, avocado, coconut, cocoa butter, almond, safflower, corn, cotton seed, sesame seed, walnut oil, castor, olive, jojoba, palm, palm kernel, soybean, wheat germ, linseed, sunflower seed; eucalyptus, lavender, vetiver, litsea, cubeba, lemon, sandalwood, rosemary, chamomile, savory, nutmeg, cinnamon, hyssop, caraway, orange, geranium, cade, and bergamot oils, fish oils, glycerol tricaprocaprylate; and mixtures thereof. The natural oils can also be utilized as emollients.
[00127] Natural and synthetic wax conditioning agents can be employed in the compositions of the disclosed technology, including but are not limited to carnauba wax, carnauba acid wax, hydrolyzed carnauba wax, ethoxylated carnauba wax (e.g., PEG-12 carnauba wax), candelilla wax, hydrolyzed candelilla wax, clover wax, alfalfa wax, paraffin wax, ozokerite wax, olive wax, rice wax, hydrogenated castor wax, bayberry wax, hydrogenated jojoba wax, bees wax, modified bees wax, e.g., cera bellina wax, ethoxylated beeswax (e.g., PEG-6 beeswax, PEG-8 beeswax, PEG-12 beeswax, PEG- 20 beeswax), dimethicone copolyol beeswax esters and dimethiconol beeswax ester (e.g.
Bis-Hydroxyethoxypropyl Dimethicone Beeswax Esters, Dimethicone PEG-8 Beeswax, and Dimethiconol Beeswax available from Lubrizol Advanced Materials, Inc. under the Ultrabee® trademark), marine waxes, polyolefin waxes, e.g., polyethylene wax; and mixtures thereof.
[00128] Liquid polyolefin conditioning oils can be used in the compositions of the present invention. The liquid polyolefin conditioning agents are typically poly-a-olefins that have been hydrogenated. Polyolefins for use herein can be prepared by the polymerization of C4 to about C14 olefinic monomers. Non-limiting examples of olefinic monomers for use in preparing the polyolefin liquids herein include ethylene, propylene, 1 -butene, 1 -pentene, 1 -hexene, 1 -octene, 1 -decene, 1 -dodecene, 1 -tetradecene, branched chain isomers such as 4-methyl-1 -pentene, and mixtures thereof. In one aspect of the disclosed technology hydrogenated a-olefin monomers include but are not limited to 1-hexene to 1 -hexadecenes, 1-octene to 1 -tetradecene, and mixtures thereof.
[00129] Other suitable ingredients include, but are not limited to, fatty esters having at least 10 carbon atoms. These fatty esters include esters derived from fatty acids or alcohols (e.g., mono-esters, polyhydric alcohol esters, and di- and tri-carboxylic acid esters). The fatty esters hereof may include or have covalently bonded thereto other compatible functionalities, such as amides and alkoxy moieties (e.g., ethoxy or ether linkages, etc.). Exemplary fatty esters include, but are not limited to isopropyl isostearate, hexyl laurate, isohexyl laurate, isohexyl palmitate, isopropyl palmitate, decyl oleate, isodecyl oleate, hexadecyl stearate, decyl stearate, isopropyl isostearate, dihexyldecyl adipate, lauryl lactate, myristyl lactate, cetyl lactate, oleyl stearate, oleyl oleate, oleyl myristate, lauryl acetate, cetyl propionate, and oleyl adipate.
[00130] Other fatty esters suitable for use in the compositions of the disclosed technology are mono-carboxylic acid esters of the general formula R50C(O)OR51, wherein R50 and R51 are alkyl or alkenyl radicals, and the sum of carbon atoms in R50 and R51 is at least 10 in one aspect, and at least 22 in another aspect of the disclosed technology.
[00131] Still other fatty esters suitable for use in the compositions of the present invention are di- and tri-alkyl and alkenyl esters of carboxylic acids, such as esters of C4-C8 dicarboxylic acids (e.g., C1-C22 esters, preferably C1-C6, of succinic acid, glutaric acid, adipic acid). Specific non-limiting examples of di- and tri-alkyl and alkenyl esters of carboxylic acids include isocetyl stearyol stearate, diisopropyl adipate, and tristearyl citrate. Other fatty esters suitable for use in the compositions of the present invention are those known as polyhydric alcohol esters. Guerbet esters are also suitable in the compositions of the present invention. Guerbet esters can be formed from the
esterification of a mono- or polyfunctional carboxylic acid by a Guerbet alcohol. Alternatively, the ester can be formed by reacting a Guerbet acid with a mono- or polyfunctional alcohol. For a review of Guerbet chemistry, see O'Lenick, A. J., Jr. 2001. Guerbet chemistry. Journal of Surfactants and Detergents 4: 311-315. Guerbet esters are commercially available from Lubrizol Advanced Materials, Inc. under product designations G-20, G-36, G-38, and G-66.
[00132] The amount of conditioning oils, waxes and esters can range from about 0.05 to about 10 wt.%, or from about 0.5 to about 5 wt.%. or from about 1 to about 3 wt.%, based on the total weight of the composition.
[00133] Fatty acids and fatty alcohols can be employed in the compositions of the present invention. Suitable fatty acids include saturated and unsaturated Cs to C30 fatty acids. Exemplary fatty acids include, but are not limited to, caprylic acid, capric acid, lauric acid, myristic acid, palmitic acid, palmitoleic acid, stearic acid, oleic acid, ricinoleic acid, vaccenic acid, linoleic acid, a-linolenic acid, y-linolenic acid, arachidic acid, gadoleic acid, arachidonic acid, EPA (5,8,11 ,14, 17-eicosapentaenoic acid), behenic acid, erucic acid, DHA (4,7,10,13,16,19-docosahexaenoic acid), lignoceric acid, and mixtures thereof. [00134] Alkoxylated fatty acids are also useful herein and can be formed by esterifying a fatty acid with an ethylene oxide and/or propylene oxide or with a pre-formed polymeric ether (e.g., polyethylene glycol or polypropylene glycol).
[00135] The fatty alcohols suitable for use in the compositions of the invention include, but are not limited to, the saturated and unsaturated C8-C30 fatty alcohols. Fatty alcohols are widely available and can be obtained through the hydrogenation of esterified vegetable and animal oils and fats. Suitable ethoxylated fatty alcohols are adducts of fatty alcohols and polyethylene oxide.
[00136] The fatty acids and fatty alcohols be utilized in an amount ranging from about 0.1 to about 30 wt.%, or from about 0.5 to 25 wt.%, or from about 3 to 20 wt.%, or from 5 to about 10 wt.%, based on the total weight of the composition.
Humectants
[00137] Humectants are defined as materials that absorb or release water vapor, depending on the relative humidity of the environment, (Harry's Cosmeticology, Chemical Publishing Company Inc., 1982 p. 266). Suitable humectants that include, but are not limited to, allantoin; pyrrolidonecarboxylic acid and its salts; hyaluronic acid and salts thereof; sorbic acid and salts thereof; urea, lysine, cystine, and amino acids; polyhydroxy alcohols such as glycerin, propylene glycol, hexylene glycol, hexanetriol, ethoxydiglycol, dimethicone copolyol, and sorbitol, and the esters thereof; polyethylene glycol; glycolic
acid and glycolate salts (e.g. ammonium and quaternary alkyl ammonium); chitosan; aloe- vera extracts; algae extract; honey and derivatives thereof; inositol; lactic acid and lactate salts (e.g. ammonium and quaternary alkyl ammonium); sugars and starches (e.g., maltose, glucose, fructose); sugar and starch derivatives (e.g., glucose alkoxylated glucose, mannitol, xyliyol); DL-panthenol; magnesium ascorbyl phosphate, arbutin, kojic acid, lactamide monoethanolamine; acetamide monoethanolamine; and the like, and mixtures thereof. Humectants also include the C3 to Ce diols and triols, such as glycerin, propylene glycol, butane-1 ,2,3-triol, hexylene glycol, hexanetriol, and the like, and mixtures thereof. Ethoxylated methyl glucose ethers containing an average of 5 to 30 moles of ethoxylation, such as, for example, those available under the INCI names Lauryl Methyl Gluceth-10 Hydroxypropyldimonium chloride, Methyl Gluceth-10 and Methyl Gluceth-20, are suitable.
[00138] Such humectants may be present at from 0.01-20 wt. % of the composition, such as at least 0.1 wt.%, or at least 1 wt.%, e.g., up to 8 wt.%, or up to 5 wt.% based on the total weight of the composition.
[00139] A skin sensate helps provide a sensory confirmation of the adequacy, activity, and evenness of the application thereof by a user. Some non-limiting examples of skin sensates are described in U.S. Pat. Nos. 4,230,688, 4,136,163, 6,183,766 and 7,001 ,594 each of which are incorporated herein by reference in their entireties. Non-limiting examples of suitable sensates include butanedioic acid monomenthyl ester, camphor, carvone, cineole, clove oil, ethyl carboxamide, ethyl menthane carboxamide, eucalyptus oil, eucolytol, ginger oil, l-isopulegol, menthol, menthone glycerin acetal, menthoxy-1 ,2- propanediol, menthyl lactate, methyl diisopropylpropioniamide, methyl salicylate, peppermint oil, rosemary oil, trimethyl butanamide, vanillyl butyl ether or combinations thereof.
[00140] The sensate can be included in the composition in amounts ranging from about 0.01 wt.% to about 2 wt.% in one aspect, and from about 0.05 wt.% to about 1 wt.% in another aspect, based on the total weight of the composition.
[00141] The compositions of the present technology can also comprise fragrance or perfume components may range from about 0.000001 to about 2 wt.%, or from 0.00001 to about 1.5 wt.%, or from 0.0001 to about 1 wt.%, or from about 0.001 to about 0.8 wt.%, based on of the weight of the composition.
[00142] Any preservative suitable for use in personal care can be used in the compositions of the present invention. Suitable preservatives include polymethoxy bicyclic oxazolidine, methyl paraben, propyl paraben, ethyl paraben, butyl paraben,
benzyltriazole, DMDM hydantoin (also known as 1 ,3-dimethyl-5,5-dimethyl hydantoin), imidazolidinyl urea, phenoxyethanol, phenoxyethylparaben, methylisothiazolinone, methylchloroisothiazolinone, benzoisothiazolinone, triclosan, and suitable polyquaternium compounds as disclosed above (e.g., Polyquaternium- 1). Acid-based preservatives are also useful in the exemplary compositions. Exemplary acids are, but are not limited to, formic acid, acetic acid, propionic acid, sorbic acid, caprylic acid, and benzoic acid, oxalic acid, succinic acid, glutaric acid, adipic acid, azelaic acid, maleic acid, fumaric acid, lactic acid, glyceric acid, tartronic acid malic acid, tartaric acid, gluconic acid, citric acid, ascorbic acid, salicylic acid, phthalic acid, mandelic acid, benzilic acid, and mixtures thereof.
[00143] Salts of the foregoing acids are also useful as long as they retain efficacy at low pH values. Suitable salts include the alkali metal (e.g., sodium, potassium, calcium) and ammonium salts of the acids enumerated above.
[00144] The acid-based preservatives and/or their salts can be used alone or in combination with non-acidic preservatives typically employed in personal care, home care, health care, and institutional and industrial care products.
[00145] Examples of suitable antibacterial agents which can be used herein include, but are not limited to, 2-hydroxy-4,2',4'-trichlorodiphenylether (TCS), 2,6- dimethyl-4-hydroxychlorobenzene (PCMX),3,4,4'-trichlorocarbanilide (TCC), 3- trifluoromethyl-4,4'-dichlorocarbanilide (TFC), 2,2'-dihydroxy-3, 3', 5, 5', 6,6'- hexachlorodiphenylmethane, 2,2'-dihydroxy-3,3',5,5'-tetrachlorodiphenylmethane, 2,2'-dihydroxy-3,3',dibromo-5,5'-dichlorodiphenylmethane, 2-hydroxy4,4'- dichlorodiphenylether, 2-hydroxy-3,5',4-tribromodiphenylether, 1-hydroxyl-4-methyl- 6-(2,4,4-trimethylpentyl)-2(1 H)-pyridinone (Octopirox), salts of 2-pyridinethiol- 1 - oxide, salicylic acid, and other organic acids. Other suitable antibacterial agents are described in U.S. Patent. Nos. 3,835,057; 4,714,653; and 6,488,943.
[00146] The compositions and products in the context of the present invention may be combined with antioxidant agents. Non-limiting examples include EDTA and its salts, citric acid, tartaric acid, oxalic acid, BHA (butylhydroxyanisol), BHT (butylhydroxytoluene), tocopherol derivatives such as tocopherol acetate, and mixtures thereof.
[00147] The preservatives, antibacterial agents and antioxidants, may comprise from 0.01 to 3.0 wt.%, or from about 0.1 to about 1 wt.%, or from about 0.3 to about 1 wt.%, based on the weight of the total composition.
[00148] The compositions of the present invention can be prepared as water-free or water-based formulations, and formulations containing water-miscible auxiliary solvents and/or diluents but are not limited thereto. The compositions of the present invention are particularly useful for water-based, solvent based, hydroalcoholic based, and mixed solvent formulations, and for formulations containing water-miscible auxiliary solvents. Useful solvents commonly employed are typically liquids, such as water (deionized, distilled or purified), alcohols, fatty alcohols, polyols, and the like, and mixtures thereof. Non-aqueous or hydrophobic auxiliary solvents are commonly employed in substantially water-free products, such as nail lacquers, aerosol propellant sprays, or for specific functions, such as removal of oily soils, sebum, make-up, or for dissolving dyes, fragrances, and the like, or are incorporated in the oily phase of an emulsion. Non-limiting examples of auxiliary solvents, other than water, include linear and branched alcohols, such as ethanol, propanol, isopropanol, hexanol, and the like; aromatic alcohols, such as benzyl alcohol, cyclohexanol, and the like; saturated C12 to C30 fatty alcohol, such as lauryl alcohol, myristyl alcohol, cetyl alcohol, stearyl alcohol, behenyl alcohol, and the like. Nonlimiting examples of polyols include polyhydroxy alcohols, such as glycerin, propylene glycol, butylene glycol, hexylene glycol, C2 to C4 alkoxylated alcohols and C2 to C4 alkoxylated polyols, such as ethoxylated, propoxylated, and butoxylated ethers of alcohols, diols, and polyols having about 2 to about 30 carbon atoms and 1 to about 40 alkoxy units, polypropylene glycol, polybutylene glycol, and the like. Non-limiting examples of non-aqueous auxiliary solvents or diluents include silicones, and silicone derivatives, such as cyclomethicone, and the like, ketones such as acetone and methylethyl ketone; natural and synthetic oils and waxes, such as vegetable oils, plant oils, animal oils, essential oils, mineral oils, C7 to C40 isoparaffins, alkyl carboxylic esters, such as ethyl acetate, amyl acetate, ethyl lactate, and the like, jojoba oil, shark liver oil, and the like. Some of the foregoing non-aqueous auxiliary solvents or diluents may also be conditioners and emulsifiers.
[00149] Natural and/or synthetic agent (or combination thereof) to obtain enhanced thickening properties can be also used in the context of the present invention. The person skilled in the art will readily select a proper thickening agent(s) and amounts(s) thereof to obtain the desired rheology. Non-limiting examples of natural thickening agents are tree & shrub exudates (karaya gum, tragacanth gum, gum Arabic, gum ghatti), seed extracts (guar gum, cassia gum, locust been gum, tamarind seed), seaweed extracts (carrageenan, alginates, agar), fruit extracts (pectins, waxes), grains & roots (corn starch, potato starch, etc), microbial polysaccharides (Xanthan gum, dextran), modified natural
products (cellulose derivatives such as hydropropyl cellulose, methylcellulose, hydroxypropyl methylcellulose, cellulose gum, etc.); and hydrophobically modified ethoxylated methyl glucosides, such as PEG 120 methyl glucose dioleate, PEG- 120 methyl glucose trioleate, and PEG-20 methyl glucose sesquistearate, available from Lubrizol Advanced Materials, Inc., under the trade names, Glucamate™ DOE-120, Glucamate™ LT and VLT, and Glucamate™ SSE-20, respectively, are also suitable as thickening agents. Non-limiting examples of synthetic thickening agents include the polyethylene glycols (PEG) having 5 to 200 glycol units, such as, for example, those available under the INCI names PEG- 6, PEG-8, PEG-12, PEG-20, PEG-30, PEG-32, PEG-75, PEG-90, PEG-100 and PEG-200; acrylic/methacrylic acid homopolymers and copolymers; acrylamide homopolymers and copolymers; polymers prepared from 2- acrylamido-2-methylpropanesulfonic acid (AMPS® monomer).
[00150] Another class of synthetic thickeners suitable for use includes the hydrophobically modified alkali-swellable emulsion polymers, commonly referred to as (HASE) polymers. Typical HASE polymers are free radical addition polymers polymerized from pH sensitive or hydrophilic monomers (e.g., acrylic acid and/or methacrylic acid, 2- acrylamido-2-methylpropane sulfonic acid), hydrophobic monomers (e.g., C1-C30 alkyl esters of acrylic acid and/or methacrylic acid, acrylonitrile, styrene), an "associative monomer", and an optional crosslinking monomer.
[00151] Exemplary HASE polymers are disclosed in U.S. Patent Nos. 3,657,175; 4,384,096; 4,464,524; 4,801 ,671 ; and 5,292,843, which are herein incorporated by reference. In addition, an extensive review of HASE polymers is found in Gregory D. Shay, Chapter 25, "Alkali-Swellable and Alkali-Soluble Thickener Technology A Review", Polymers in Aqueous Media - Performance Through Association, Advances in Chemistry Series 223, J. Edward Glass (ed.), ACS, pp. 457-494, Division Polymeric Materials, Washington, DC (1989), the relevant disclosures of which are incorporated herein by reference.
[00152] If utilized, the thickeners can comprise from about 0.01 wt.% to about 5 wt. % of the total weight of the personal care composition, in another aspect the amount ranges from about 0.1 wt. % to about 3 wt. %, and in a further aspect from about 0.1 wt. % to about 2.0 wt. % of the total weight of the personal care composition.
[00153] Viscosity adjusting agents are used in cosmetics to enhance the fluidity of products without a significant lowering of the concentration of the active constituents. Suitable viscosity adjusting agents if present include organic and inorganic compounds, and combinations thereof. Examples of organic compounds include ethanol, isopropyl
alcohol, sorbitol, propylene glycol, diethylene glycol, triethylene glycol, dimethyl ether, butylene glycol, and the like, and mixtures thereof. Examples of inorganic compounds include sodium chloride, sodium sulfate, potassium chloride, potassium nitrate, and mixtures thereof. If utilized the viscosity adjusting agents typically comprise from about 0.1 wt. % to about 20 wt.% in one aspect, and from about 1 wt.% to about 5 wt.% of the total weight of the composition.
[00154] The compositions of the disclosed invention can contain active ingredients or pharmaceutically active agents. In the context of this invention, the ‘personal care active ingredients’ as used herein refers to ingredients that improve or enhance the physical appearance. They are also commonly known as ‘cosmetic active ingredients’.
[00155] Non-limiting examples of ingredients commonly used in cosmetic or pharmaceutical compositions, for example and, not restricted to, (i) anti-wrinkle agents, botox-like agents and/or anti-aging agents; (ii) firming agents, skin elasticity agents and/or restructuring agents; (iii) moisturizing agents; (iv) anti-photoaging agents, and/or bluelight protector agents; DNA protecting agents, DNA repair agents, and/or stem cell protecting agents; (v) free radical scavengers and/or anti-glycation agents, detoxifying agents, antioxidant and/or anti-pollution agents; (v) agents which increase the percutaneous absorption of any active compounds present therein; (vi) anti-perspirant agents; (vii) melanin synthesis stimulating or inhibiting agents; whitening or depigmenting agents; pro-pigmenting agents; self-tanning agents; (viii) lipolytic agents or agents stimulating lipolysis, adipogenic agents, etc.
[00156] Suitable active ingredient in the context of the present invention include, but are not limited to, peptides, proteins, hydrolyzed proteins, enzymes, vitamins, mineral salts, sugars, nucleotides, nucleic acids, molecules and extracts of biological and biotechnological origin, vegetable extracts, cells extracts, essential oils, molecules of synthetic or natural origin, isoflavones, polyphenols, retinol, hyaluronic acid and/or mixtures thereof.
[00157] The active ingredient can be typically used in a concentration from 0.001 to 10 wt. %, or from 0.1 to 5 wt. %, or from 0.2 to 3 wt.% based on the total weight of the composition.
[00158] The present invention will be better understood by reference to the following examples, which serve to illustrate the invention, but not to limit the same.
EXAMPLES
Experimental methods and determinations
Brookfield viscosity measurement method:
[00159] Viscosity and yield values were determined with a Brookfield viscosimeter. Sample fluids were free of entrapped air as much as possible and maintained at constant temperature of 25°C (i.e., coming from a water bath or oven).
[00160] The spindle of the viscosimeter was inserted into the sample at 25°C. The viscosimeter was set at different speeds namely 0.5, 1 , 2.5, 5, 10, and 20 rpm and the spindle allowed to rotate at the selected rpm for 2 minutes for 0.5 rpm and 1 minute for all other rpms. The spindle is selected to ensure that the % torque reading is between 10 and 90%. The viscosity values at 0.5 and 1 rpm are used to calculate the yield value, where yield value = (viscosity at 0.5 rpm - viscosity at 1 rpm)/100.
Coalescence determination
[00161] Unless otherwise indicated emulsion coalescence was determined by visual inspection and scored from 0 to 5 where: 0 represents visual formation of thick film layer of oil in the surface; 1 , a slight film layer formation in the surface; 2 oil pooling; 3, numerous small and large droplets; 4, few small droplets; 5, no visible oil droplets.
EXAMPLE 1
Synthesis of crosslinked and hydrophobically modified xanthan gum.
[00162] A four-neck round bottom flask fitted with a thermocouple, glass stir shaft with Teflon paddle, nitrogen adapter, and condenser was charged with 60 wt.% isopropanol/water (462.41 g). The solvent was stirred at 200 rpm while the headspace was purged with nitrogen at 0.4 L/min. Xanthan gum (108.58 g) was charged to the flask slowly via powder funnel while stirring to provide a slurry. After 5 min, sodium hydroxide (50 wt.%, 16.02 g) was slowly added to the reactor while stirring and purging with nitrogen. The pH of the reaction solution was 12.32. The temperature was set to 60°C. Glycidyl ether HAGE-16 from Sachem, Inc (Texas, USA) (9.20 g) was melted in a 115°C oven for 10 min and dissolved in isopropanol (7.87 g). This solution was charged to the reactor at 60°C and rinsed with isopropanol (6.07 g). Heloxy™ 67 (1 ,4-butane diol diglycidyl ether) from Hexion Inc. (Ohio, USA) (0.834 g) was weighed using a 1 mL syringe and charged to the reactor immediately after HAGE-16. The nitrogen flow rate was reduced to 0.185 L/min. After 3 h the reaction was cooled to 40 °C and had a pH of 12.18. The mixture was neutralized with glacial acetic acid (5.98 g) to a pH of 6.84. The solids were recovered by filtration using a Millipore pressure filtration device, and the filter cake was washed with 80 wt.% isopropanol (303.45 g) in a 1 L beaker using overhead stirring with a marine mixing blade for 30 min. The product was recovered by filtration, and the filter cake was
dried in an aluminum pan in a vacuum oven set to 80°C for 4 h. The resulting tan powder had a total solids of 94.6 wt.%. A 0.5 wt.% dispersion of the product in water had a Brookfield viscosity of 1403 ± 42.9 mPa s (20 rpm, average of 3 replicates).
EXAMPLE 2
[00163] Several compositions were generated by varying both the alkyl chain length of the hydrophobe and its molar substitution (MS) on the polymer, which was calculated as the molar ratio of hydrophobe per anhydroglycosidic unit (AGU). The alkyl chain length ranged from Cs to C22, and the MS ranged from 0.002 - 0.05. Xanthan gum demonstrated a general increase in dispersion viscosity and yield value after modification compared to the native xanthan gum (Figure 1). Emulsion performance was monitored using an oil-in- water system containing 20 wt.% Schercemol™ 318 (INCI: Isopropyl Isostearate from Lubrizol Advanced Materials, INC), 0.5 wt.% xanthan polymer, and 0.5 wt.% phenoxyethanol. Homogenization with an Ultra-Turrax® homogeneizer was used at 10,000 rpm for one minute. Importantly, all emulsions containing hydrophobically modified xanthan polymers were stable against creaming during stability testing for four weeks at 50°C compared to a control polymer that underwent the same reaction conditions but without the hydrophobe. However, significant reduction in viscosity and yield values were observed during stability testing, as well as poor texture and coalescence ratings (Table 1).
Table 1. Results for emulsions containing 20 wt.% Isopropyl Isostearate, 0.5 wt.% polymer, and 0.5 wt. % phenoxyethanol.
a Coalescence scale from A (best, no surface oil droplets) to D (worst, phase separation)
1 Viscosity determined as Brookfield viscosity (20 rpm, at room temp.) after 24h rest.
EXAMPLE 3
[00164] Xanthan gum was Ci6-modified at MS = 0.05. Samples were dried at either high (115°C) or low (60°C) temperature. Stability at 4 weeks at 50°C was monitored.
[00165] Heat treatment of hydrophobically modified xanthan gum on performance led to the discovery that the polymer crosslinks when dried at 115°C after the reaction. This led to increased viscosity and yield values, but on the other hand gave deleterious effects at long term on stability. When hydrophobically modified xanthan gum was dried at 60°C, the viscosity and yield of the emulsion were maintained during aging, and no creaming was observed (Table 2).
Table 2. Emulsion results for polymers that were dried at low or high temperatures. All samples were Ci6-modified at MS=0.05. Emulsions contained 0.5 wt.% polymer, 20 wt.% Caprylic/Capric Triglyceride and 0.5 wt.% phenoxyethanol and were homogenized at 10,000 rpm for one minute.
a Viscosity determined as Brookfield viscosity (20 rpm, at room temp.) after 24h rest.
EXAMPLE 4
[00166] The effect of using a crosslinker during the reaction was studied by adding 1 ,4-Butanediol diglycidyl ether (BDDE) to the reaction simultaneously with the alkyl glycidyl ether in varying amounts of 0.1 - 1.0 wt.% with respect to the xanthan gum and compared with the same process without BDDE. Figure 2 shows the dispersion rheology results of crosslinked xanthan gum containing a Ci6 hydrophobe at a MS=0.05. For 0.5 wt.% polymer dispersions, increased crosslinker resulted in an increase in viscosity, but at 1.0 wt.% crosslinker no linear viscoelastic region (LVER) was observed due to a decrease in particle swelling and an increase in polymer overlap concentration (c*). Increasing to 1.0 wt.% polymer dispersions resulted in the highest viscosity for polymer crosslinked at 1.0 wt.%, and no difference was observed for the 0.1 wt.% crosslinked polymer compared to the uncrosslinked polymer.
[00167] Emulsions made with this series followed the same trends as the dispersion rheology - increased crosslinker amount resulted in emulsions with higher viscosity. The coalescence ratings after aging at 50°C for four weeks also significantly improved with increasing crosslinker amount; however, undesirable applesauce texture was observed for the highest crosslink level studied. Importantly, unlike hydrophobic xanthan crosslinked via dry heating (i.e., 115°C), these diglycidyl ether crosslinked polymers maintained stable viscosity and yield values during aging at elevated temperature (Table 3).
Table 3. Viscosity of emulsions containing crosslinked and hydrophobic xanthan gum during aging studies at 50°C. Emulsions contained 0.5 wt.% polymer, 20 wt. % caprylic/capric triglyceride, and 0.5 wt.% phenoxyethanol.
Table 4. Coalescence rating of emulsions containing crosslinked and hydrophobic xanthan gum. The emulsions were made using 0.5 wt.% modified xanthan and 20 wt.% caprylic/capric triglyceride.
Coalescence rating: 5 is the best, 0 is the worst.
[00168] Xanthan modified with 0.5 wt.% crosslinker with respect to xanthan gum provided a better balance of increased efficiency and coalescence stability while still providing acceptable textures (Table 4).
EXAMPLE 5
[00169] The polymer use level in an emulsion containing 20 wt.% caprylic/capric triglyceride was incrementally increased from 0.5 to 1.5 wt.% (Tables 5 and 6).
Table 5. Viscosity depending on polymer use level in emulsions containing 20 wt.% Caprylic/capric triglyceride.
Table 6. Coalescence rating (being 5 is the best performance, 0 is the worst performance) of emulsions at varying polymer use levels. Emulsions contained 20 wt. % Caprylic/capric triglyceride.
[00170] At 0.7 wt.% polymer, good coalescence performance was observed (without co-emulsifier), and gel/cream textures were achieved with a viscosity >8000 mPa s at 1.5 wt.% polymer with minimal texture.
EXAMPLE 6
[00171] Samples were synthesized to validate the utility of hydrophobic and crosslinked xanthan; crosslinked xanthan without addition of hydrophobe and xanthan that underwent the same reaction conditions without crosslinker or hydrophobe. The dispersion rheology of 0.5 wt.% dispersions showed a decrease in viscosity for xanthan that does not have crosslinker or a hydrophobic group. When crosslinker was added to the reaction but no hydrophobe, the viscosity was still less than the native xanthan, but was higher than without crosslinker that underwent same reaction conditions, demonstrating the recovery of some efficiency by crosslinking. The highest viscosity sample was the xanthan that was both hydrophobically modified and crosslinked (Table 7).
Table 7. Viscosity and yield values for emulsions containing either hydrophobic and crosslinked xanthan, non-hydrophobic, but crosslinked xanthan, or non-hydrophobic and non-crosslinked xanthan. Emulsions contained 0.5 wt.% polymer and 20 wt.% Caprylic/capric triglyceride.
1 Determined as Brookfield viscosity at 20 rpm and room temperature.
[00172] Emulsions of these control reactions with 20 wt.% Caprylic/capric triglyceride at 0.5 wt.% xanthan polymer gave similar trends as the dispersion rheology (Figure 3). When only the hydrophobe was omitted from the reaction, but the crosslinker was present, initial viscosities and yield values were similar to the hydrophobic and crosslinked xanthan. However, the absence of both hydrophobe and crosslinker reduced both the viscosity and yield values of the emulsion. Optical microscopy images of each of these emulsions revealed the smallest particle size for the emulsion containing the hydrophobic and crosslinked xanthan (Figure 4A). After aging each emulsion for four weeks at 50 °C, the emulsion containing the hydrophobic and crosslinked xanthan had significantly better coalescence ratings than either of the control reactions (Figure 4B and Table 8). This demonstrates that while initial physical qualities of the emulsion can be achieved with crosslinked xanthan alone, the hydrophobic moiety is necessary to provide emulsification, and neither optimal viscosity nor stability could be achieved when no polymer modification occurred.
Table 8. Coalescence rating of 20 wt.% caprylic/capric triglyceride emulsions containing different types of polymer (wherein 5 is the best rating and 0 the worst).
EXAMPLE 7
[00173] The effect of hydrophobe chain length and MS on emulsion performance was studied. In comparison to Ci6-modified and crosslinked xanthan, the alkyl chain length was reduced to C12 (lauryl) and Cs (2-ethylhexyl) glycidyl ethers, each with a MS of 0.025. The dispersion efficiency for these three polymers was approximately equal, as shown in Figure 5. Similarly, the viscosity of emulsions containing 0.5 wt. % polymer was also quite similar (Table 9), although the coalescence stability was slightly better for both C16 and C12 polymers compared to the Cs polymer.
Table 9. Viscosity of emulsions containing modified xanthan polymers with varying alkyl chain lengths. Emulsion contains 0.5 wt.% polymer and 20 wt.% caprylic/capric trygliceride.
1 Determined as Brookfield viscosity at 20 rpm and room temperature.
[00174] The MS for a Ci6-modified xanthan polymer was increased from 0.05 to 0.3. Interestingly, no significant differences were observed for dispersion viscosities at 0.5 wt.% xanthan polymer, with complex viscosities from amplitude sweeps essentially overlaid (Figure 6).
[00175] Similar results were observed for emulsions made with these samples at 0.6 wt.% polymer in 20 wt.% caprylic/capric tryglyceride. The initial viscosity and yield values for the emulsions were similar (Table 10).
Table 10. Viscosity and yield values for emulsions containing 20 wt. % caprylic/capric triglyceride and 0.6 wt.% polymer. Polymers had various molar substitution of C16.
1 Determined as Brookfield viscosity at 20 rpm and room temperature.
EXAMPLE 8
[00176] Emulsion stability as a function of polymer use level was also evaluated using emulsions containing 20 wt.% caprylic/capric triglyceride at pH 5.5. Passing coalescence
ratings were observed after aging at 50°C for one week for all emulsions with a polymer concentration >0.6 wt.% (Table 11). A range of viscosities was also observed.
Table 11. O/W emulsions containing modified xanthan of Example 1 at increased use levels. Emulsion contained 20 wt.% caprylic/capric triglyceride at pH 5.5.
Coalescence rating: 0 worst rating, 5 best rating.
1 Viscosity determined as Brookfield viscosity at 20 rpm at room temperature.
Viscosity and yield values determined after 24h rest.
EXAMPLE 9
[00177] Because of the inherent stabilizing effect of modified xanthan gum of Example 1 , only low amounts of co-surfactants are required to boost emulsion performance. Hydramol™ TGL ester (INCI: Polyglyceryl-3 Laurate from Lubrizol Advance Materials, Inc) as low as 0.25 wt.% resulted in a passing coalescence rating at 0.5 wt.% of modified xanthan after aging at 50 °C for four weeks. Sodium stearoyl glutamate also improved coalescence, although at a slightly higher use level of 0.5 wt%. Glucate™ DO (INCI: Methyl Glucose Dioleate from Lubrizol Advance Materials, Inc.) showed no improvement; therefore, mid- to high range HLB co-emulsifiers in combination with the modified xanthan gum according to the invention provide better results (Table 12).
Table 12. Coalescence rating of emulsions containing 20 wt.% of caprylic/capric triglyceride and 0.5 wt.% of modified xanthan gum of Example 1 after aging at 50°C for four weeks containing various co-emulsifiers at 0.25 wt.% and 0.5 wt.%. Coalescence rating being 0 as the worst performance and 5 the best performance.
[00178] Electrolyte tolerance was also evaluated by adding 0.5 wt.% NaCI or MgSC>4 to emulsions containing 0.7 wt.% of modified xanthan gum of Example 1 , and 20 wt.% caprylic/capric triglyceride. Emulsion viscosity and yield values were unaffected by salt addition, but the coalescence ratings decreased. Adding 0.1 wt.% Polyglyceryl-3 Laurate to the emulsion improved the initial coalescence rating (Table 13).
Table 13. Emulsion evaluation with the addition of 0.5 wt.% NaCI or MgSO4. Emulsions contained 0.7 wt.% of modified xanthan of Example 1 and 20 wt.% caprylic/capric triglyceride oil. Data collected before aging.
Coalescence rating being 5 the best and 0 the worst.
1 Viscosity determined Brookfield viscosity at 20 rpm at room temperature.
[00179] Modified xanthan of the invention was also found to be stable in formulations with pH ranging from 4 - 8, which allows for a wide range of skin care applications. Emulsions prepared at various processing conditions using higher shear rates demonstrated that there was no negative impact of shear on polymer performance. Additionally, the modified xanthan of the invention is compatible with glycols including Glucam™ E-20 humectant (from Lubrizol Advanced Materials, Inc), Zemea® propanediol
(from CovationBio PDO LLC), and glycerin, which reduced hydration time without negatively affecting viscosity.
EXAMPLE 9
[00180] Cleansing formulations were prepared as set out in Table 14.
1 Active amount (wt.%) a Comparative Example b Modified xanthan gum according to EXAMPLE 1
[00181] As shown in Table 14 and illustrated in Figure 7. The oil drop size of formulations comprising modified xanthan according to the present invention was significantly reduced.
EXAMPLE 10
[00182] Cleansing formulations were prepared as set out in Table 15.
1 Active amount (wt.%) a Comparative Example b Modified xanthan gum according to EXAMPLE 1
[00183] As shown in Table 15 and illustrated in Figure 8, the xanthan gum significantly reduced the droplet size.
EXAMPLE 11
[00184] A cleansing formulation was prepared as set out in Table 16.
1 Active amount (wt.%) a Comparative Example b Modified xanthan gum according to EXAMPLE 1 c Native xanthan gum (Keltrol CG-SFT from CP Kelco)
[00185] As shown in Table 16, the modified xanthan gum can significantly improve suspension of things other than oils, such as air, by improving the overall rheological properties (demonstrated through and increase in viscosity, yield, and the physical suspension of air at elevated temperatures). The ability to suspend visually pleasing ingredients such as air, beads, effect pigments, pearlizers, etc can be used to enhance the overall consumer perception and acceptance of personal care products.
EXAMPLE 12
[00186] Cleansing compositions were prepared as set out in Table 17:
1 Active amount (wt.%) a Comparative Example b Modified xanthan gum according to EXAMPLE 1 c Native xanthan gum (Keltrol CG-SFT from CP Kelco)
EXAMPLE 13
[00187] Cleansing formulations were prepared as set out in Table 18.
Table 18. Cleansing formulations that suspend air with modified xanthan gum.
1 Active amount (wt.%) a Comparative Example b Modified xanthan gum according to EXAMPLE 1 c Native xanthan gum (Keltrol CG-SFT from CP Kelco)
EXAMPLE 14
[00188] Cleansing formulations were prepared as set out in Table 19.
Table 19. Cleansing formulations that suspend oil with modified xanthan gum.
1 Active amount (wt.%) a Comparative Example b Modified xanthan gum according to EXAMPLE 1 c Arbalon® R-50 Cellulose Liquid
[00189] A significant viscosity synergy is seen between the Modified Xanthan Gum and Cellulose (and) Cellulose Gum.
EXAMPLE 15
[00190] A four-neck round bottom flask fitted with a thermocouple, glass stir shaft with Teflon paddle, nitrogen adapter, and condenser was charged with 60 wt.% isopropanol/water (94.8 g). The solvent was stirred at 200 rpm while the headspace was purged with nitrogen at 0.4 L/min. Xanthan gum (21.7 g) was charged to the flask slowly
via powder funnel while stirring to provide a slurry. After 5 min, sodium hydroxide (50 wt.%, 3.2 g) was slowly added to the reactor while stirring and purging with nitrogen. The pH of the reaction solution was 12.2. The temperature was set to 60°C. Glycidyl ether HAGE-16 from Sachem, Inc (Texas, USA) (5.5 g) was melted in a 115°C oven for 10 min and dissolved in isopropanol (15.6 g). Epichlorohydrin (42 microliters) was added to 7.0 g of this solution which was then charged to the reactor at 60°C. The nitrogen flow rate was reduced to 0.185 L/min. After 3 h the reaction was cooled to 40 °C and had a pH of 12.3. The mixture was neutralized with glacial acetic acid (1 .7 g) to a pH of 6.7. The solids were recovered by vacuum filtration, and the filter cake was washed with 80 wt.% isopropanol (400 g) in a 1 L beaker using overhead stirring with a marine mixing blade for 30 min. The product was recovered by filtration, and the filter cake was dried in an aluminum pan in a vacuum oven set to 80°C for 4 h. The resulting tan powder had an amount of total solids of 97.2 wt.%. A 1 wt.% dispersion of the product in water had a Brookfield viscosity of 9520 mPa s (20 rpm).
EXAMPLE 16
[00191] A four-neck round bottom flask fitted with a thermocouple, glass stir shaft with Teflon paddle, nitrogen adapter, and condenser was charged with 60 wt.% isopropanol/water (94.8 g). The solvent was stirred at 200 rpm while the headspace was purged with nitrogen at 0.4 L/min. Xanthan gum (21.8 g) was charged to the flask slowly via powder funnel while stirring to provide a slurry. After 5 min, sodium hydroxide (50 wt.%, 3.2 g) was slowly added to the reactor while stirring and purging with nitrogen. The pH of the reaction solution was 12.2. The temperature was set to 60°C. Glycidyl ether HAGE-16 from Sachem, Inc (Texas, USA) (5.5 g) was melted in a 115°C oven for 10 min and dissolved in isopropanol (15.6 g). Epichlorohydrin (20 microliters) was added to 7.0 g of this solution which was then charged to the reactor at 60°C. The nitrogen flow rate was reduced to 0.185 L/min. After 3 h the reaction was cooled to 40 °C and had a pH of 12.3. The mixture was neutralized with glacial acetic acid (1.8 g) to a pH of 6.9. The solids were recovered by vacuum filtration, and the filter cake was washed with 80 wt.% isopropanol (400 g) in a 1 L beaker using overhead stirring with a marine mixing blade for 30 min. The product was recovered by filtration, and the filter cake was dried in an aluminum pan in a vacuum oven set to 80°C for 4 h. The resulting tan powder had a total solid of 95.6 wt.%. A 1 wt.% dispersion of the product in water had a Brookfield viscosity of 6830 mPa s (20 rpm).
EXAMPLE 17
[00192] A Jacketed glass pressure/filter reactor and a 4-neck lid fitted with a thermocouple, glass stir shaft, nitrogen inlet, and condenser was charged with 60 wt.% isopropanol/water (351 .2 g). The solvent was stirred at 400 rpm while the headspace was purged with nitrogen at 0.4 L/min. Xanthan gum (81.4 g) was charged to the reactor slowly via powder funnel while stirring to provide a slurry. After 5 min, sodium hydroxide (50 wt.%, 12.0 g) was slowly added to the reactor while stirring and purging with nitrogen. The pH of the reaction solution was 12.8. The temperature was set to 60°C. 1- chlorooctadecane (6.7 g) and Heloxy™ 67 (1 ,4-butane diol diglycidyl ether) from Hexion Inc. (Ohio, USA) (0.6 g) were dissolved in isopropanol (19.0 g). This solution was charged to the reactor at 60°C. The nitrogen flow rate was reduced to 0.185 L/min. After 3 h the reaction was cooled to 40 °C and had a pH of 11.9. The mixture was neutralized with glacial acetic acid (6.2 g) to a pH of 6.5. The solids were recovered by pressure filtration, and the filter cake was re-dispersed in 80 wt.% isopropanol (500 g) and washed for 30 min. The product was recovered by pressure filtration. The filter cake was removed from the reactor and dried in a vacuum oven set to 70°C for 4 h. The resulting tan powder had a total solids of 95.4 wt.%. A 1 wt.% dispersion of the product in water had a Brookfield viscosity of 5590 mPa s (20 rpm).
EXAMPLE 18
[00193] A Jacketed glass pressure/filter reactor and a 4-neck lid fitted with a thermocouple, glass stir shaft, nitrogen inlet, and condenser was charged with 60 wt.% isopropanol/water (354.0 g). The solvent was stirred at 400 rpm while the headspace was purged with nitrogen at 0.4 L/min. Xanthan gum (81 .0 g) was charged to the reactor slowly via powder funnel while stirring to provide a slurry. After 5 min, sodium hydroxide (50 wt.%, 12.0 g) was slowly added to the reactor while stirring and purging with nitrogen. The pH of the reaction solution was 12.8. The temperature was set to 60°C. 1 -chlorododecane (4.9 g) and Heloxy™ 67 (1 ,4-butane diol diglycidyl ether) (0.6 g) from Hexion Inc. (Ohio, USA) were dissolved in isopropanol (17.0 g). This solution was charged to the reactor at 60°C. The nitrogen flow rate was reduced to 0.185 L/min. After 3 h the reaction was cooled to 40 °C and had a pH of 11.9. The mixture was neutralized with glacial acetic acid (6.9 g) to a pH of 6.1. The solids were recovered by pressure filtration, and the filter cake was re-dispersed in 80 wt.% isopropanol (500 g) and washed for 30 min. The product was recovered by pressure filtration. The filter cake was removed from the reactor and dried in a vacuum oven set to 70°C for 4 h. The resulting tan powder had a total solids of 95.6
wt.%. A 1 wt.% dispersion of the product in water had a Brookfield viscosity of 5540 mPa s (20 rpm).
EXAMPLE 19
[00194] A Jacketed glass pressure/filter reactor and a 4-neck lid fitted with a thermocouple, glass stir shaft, nitrogen inlet, and condenser was charged with 60 wt.% isopropanol/water (355.0 g). The solvent was stirred at 400 rpm while the headspace was purged with nitrogen at 0.4 L/min. Xanthan gum (81 .0 g) was charged to the reactor slowly via powder funnel while stirring to provide a slurry. After 5 min, sodium hydroxide (50 wt.%, 12.0 g) was slowly added to the reactor while stirring and purging with nitrogen. The pH of the reaction solution was 12.7. The temperature was set to 60°C. Glycidyl ether HAGE-16 from Sachem, Inc (Texas, USA) (6.9 g) was melted in a 115°C oven for 10 min and dissolved in isopropanol (21.1 g). 1 ,4-dichlorobutane (0.39 g) was added to the latter solution and charged to the reactor at 60°C. The nitrogen flow rate was reduced to 0.185 L/min. After 3 h the reaction was cooled to 40 °C and had a pH of 11.9. The mixture was neutralized with glacial acetic acid (6.5 g) to a pH of 6.1. The solids were recovered by pressure filtration, and the filter cake was re-dispersed in 80 wt.% isopropanol (500 g) and washed for 30 min. The product was recovered by pressure filtration. The filter cake was removed from the reactor and dried in a vacuum oven set to 70°C for 4 h. The resulting tan powder had a total solids of 96.2 wt.%. A 1 wt.% dispersion of the product in water had a Brookfield viscosity of 3400 mPa s (20 rpm).
EXAMPLE 20
[00195] A Jacketed glass pressure/filter reactor and a 4-neck lid fitted with a thermocouple, glass stir shaft, nitrogen inlet, and condenser was charged with 60 wt.% isopropanol/water (355.7 g). The solvent was stirred at 400 rpm while the headspace was purged with nitrogen at 0.4 L/min. Xanthan gum (81 .4 g) was charged to the reactor slowly via powder funnel while stirring to provide a slurry. After 5 min, sodium hydroxide (50 wt.%, 12.0 g) was slowly added to the reactor while stirring and purging with nitrogen. The pH of the reaction solution was 12.7. The temperature was set to 60°C. Glycidyl ether HAGE-16 from Sachem, Inc (Texas, USA) (6.9 g) was melted in a 115°C oven for 10 min and dissolved in isopropanol (19.5 g). Polypropylene glycol) diglycidyl ether Mw 380 g mol’1 (1.1 g) was added to the latter solution and charged to the reactor at 60°C. The nitrogen flow rate was reduced to 0.185 L/min. After 3 h the reaction was cooled to 40 °C
and had a pH of 11 .9. The mixture was neutralized with glacial acetic acid (6.2 g) to a pH of 6.1. The solids were recovered by pressure filtration, and the filter cake was redispersed in 80 wt.% isopropanol (500 g) and washed for 30 min. The product was recovered by pressure filtration. The filter cake was removed from the reactor and dried in a vacuum oven set to 70°C for 4 h. The resulting tan powder had a total solids of 97.4 wt.%. A 1 wt.% dispersion of the product in water had a Brookfield viscosity of 4080 mPa s (20 rpm).
[00196] The invention has been explained in relation to its preferred embodiments, it is to be understood that various modifications thereof will become apparent to those skilled in the art upon reading the specification. Therefore, it is to be understood that the invention disclosed herein is intended to cover such modifications as fall within the scope of the appended claims.
Claims
1. A crosslinked and hydrophobically modified xanthan gum wherein a portion of the hydrogen atoms of pendant hydroxyl groups of the backbone are substituted with at least one hydrophobic moiety according to formula (I):
(I) - (A)a-(O)b-R wherein A is selected from a divalent linear or branched, substituted or unsubstituted C1-C6 alkylene radical; R is selected from a hydrocarbon group having from 8 to 22 carbon atoms; a is 0 or 1 , and b is 0 or 1 , with the proviso that when a is 0, b is 0; and, wherein the xanthan gum is crosslinked with a multifunctional crosslinking agent comprising epoxy and/or alkyl halide functional groups in an amount of from 0.1 to 1 wt.% based on the total weight of the xanthan gum.
2. A modified xanthan gum according to claim 1 , wherein a is 1 and b is 1.
3. A modified xanthan gum according to claims 1 or 2, wherein said divalent alkylene radical A is represented by formula (II) or (III):
4. A modified xanthan gum according to any one of the proceeding claims, wherein the R group is a saturated hydrocarbon group.
5. A modified xanthan gum according to any one of the preceding claims, wherein the R group is a linear hydrocarbon group.
6. A modified xanthan gum according to any one of the preceding claims wherein the hydrocarbon group has from 12 to 16 carbon atoms.
7. A modified xanthan gum according to any one of the preceding claims wherein the hydrocarbon group has 16 carbon atoms.
8. A modified xanthan gum according to any one of the preceding claims wherein the average degree of molar substitution of the xanthan gum by the hydrophobic substituent is in a range from 0.001 to 0.5, or from 0.01 to 0.4, or from 0.025 to 0.3.
9. A modified xanthan gum according to any one of the preceding claims wherein the crosslinking agent is a multifunctional alkyl halide.
10. A modified xanthan gum according to claim 9 wherein the multifunctional alkyl halide comprises an ethyl, propyl, isopropyl, n-butyl, t-butyl, pentyl, neopentyl, hexyl, octyl, decyl, dodecyl, myristyl, hexadecyl, stearyl or behenyl dibromide, dichloride or diiodide.
11 . A modified xanthan gum according to any one of claims 1 to 8 wherein the crosslinking agent is an epihalohydrin, preferably epichlorohydrin.
12. A modified xanthan gum according to any one of claims 1 to 8, wherein the crosslinking agent is a polyglycidyl ether.
13. A modified xanthan gum according to claim 12 wherein the polyglycidyl ether is selected from the group consisting of glycerol diglycidyl ether, polyethylene glycol diglycidyl ether, polyoxyethylene glycol diglycidyl ether, neopentyl glycol diglycidyl ether, 1 ,4-butanediol diglycidyl ether, 1 ,6-hexanediol diglycidyl ether, glycerol diglycidyl ether, trimethylolpropane diglycidyl ether, trimethylolpropane triglycidyl ether, glycerol triglycidyl ether, pentaerythritol diglycidyl ether, pentaerythritol triglycidyl ether, pentaerythritol tetraglycidyl ether, sorbitol diglycidyl ether, sorbitol triglycidyl ether, sorbitol tetraglycidyl ether, sorbitol penta glycidyl ether, and sorbitol hexaglycidyl ether, resorcinol diglycidyl ether, bisphenol A diglycidyl ether (BADGE), and bisphenol F diglycydyl ether (NOGE).
14. A modified xanthan gum according to claim any one of claims 12 or 13, wherein the crosslinking agent is 1 ,4-butanediol diglycidyl ether.
15. A modified xanthan gum according to any one of the preceding claims wherein the amount of crosslinking agent is of from 0.2 to 0.8 wt.% based on the total weight of the xanthan gum.
16. A modified xanthan gum according to any one of the preceding claims wherein the amount of crosslinking agent is of from 0.4 to 0.6 wt.% based on the total weight of the xanthan gum.
17. An oil-in-water emulsion comprising: a water phase; an oil phase dispersed in said water phase; and, a crosslinked and hydrophobically modified xanthan gum according to any one of claims 1 to 16.
18. An emulsion according to claim 17 wherein the modified xanthan gum is at a concentration of from 0.01 to 2 wt.% based on the total weight of the emulsion.
19. An emulsion according to claim 18 wherein the modified xanthan gum is at a concentration of from 0.2 to 1 .5 wt.%, or from 0.5 to 1.25 %.
20. An emulsion according to any one of claims 17 to 19 wherein the oil phase is at a concentration of from 5 to 40 wt.%, or from 10 to 30 wt.% based on the total weight of the emulsion.
21. An emulsion according to any one of claims 17 to 20 wherein the oil phase comprises an oil selected from the group consisting of fatty acid esters, monoglycerides, diglycerides and triglycerides of fatty acids and mixtures thereof.
22. An emulsion according to any one of claims 17 to 21 further comprising a co-emulsifier having a hydrophilic-lipophilic balance (HLB) value equal or higher than 6.
23. An emulsion according to claim 22 wherein the co-emulsifier has an HLB value of 10 or higher.
24. An emulsion according to claim 22 or claim 23 wherein the co-emulsifier is selected from the group consisting of amino acid-based surfactants and (poly)glyceryl fatty acids.
25. An emulsion according to any one of claims 22 to 24, wherein the co-emulsifier is at concentration of from 0.1 to 1 wt.%.
26. An emulsion according to any one of claims 17 to 25 where the viscosity determined as Brookfield viscosity at 20 rpm at room temperature is in the range from 1 ,000 to 9,000 mPa s.
27. A method of stabilizing an oil-in-water emulsion comprising an oil phase and a water phase comprising the step of mixing a crosslinked and hydrophobically modified xanthan gum according to any one of claims 1 to 16 with the oil phase and the water phase.
28. Use of a crosslinked and hydrophobically modified xanthan gum according to any one of claims 1 to 16 to stabilize an emulsion comprising an oil phase and a water phase.
29. A personal care formulation comprising a crosslinked and hydrophobically modified xanthan gum according to any one of claims 1 to 16 or an emulsion composition according to 17 to 26.
30. A cleaning or fabric care composition comprising a crosslinked and hydrophobically modified xanthan gum according to any one of claims 1 to 16.
31. A method for preparing a crosslinked and hydrophobically modified xanthan gum according to any one of claims 1 to 16 comprising reacting a xanthan gum with a) an alkylating agent and b) a multifunctional crosslinker comprising epoxide and/or alkyl halide functional groups.
32. A method according to claim 31 wherein the alkylating agent a) is an alkyl halide wherein the alkyl group is a linear or branched Csto C22 alkyl group.
34. A method according to claim any one of claims 31 to 33 wherein the multifunctional crosslinking agent is selected form the group consisting of epihalohydrin, a multifunctional alkyl halide and polyglycidyl ether.
35. A method according to any one of claims 31 to 34, wherein the reaction with the alkylating agent a) and the multifunctional crosslinker b) is conducted simultaneously.
36. A method according to any one of claims 31 to 35, comprising the steps of: i) Mixing a xanthan gum with an organic solvent or a mixture of an organic solvent and water to form a suspension or slurry; ii) Increase the pH value of the suspension or slurry to be higher than 9 with an alkaline solution; iii) Adding the alkylating agent and the multifunctional crosslinker comprising epoxide and/or alkyl halide functional groups; iv) Reacting said reaction mixture for at least 2h at a temperature from 50 to 100°C to obtain a crosslinked and hydrophobically modified xanthan gum; and, v) Neutralizing the crosslinked and hydrophobically modified xanthan gum with an acid.
37. A method according to claim 36 wherein the xanthan gum is mixed in step i) with a mixture of 50 to 80 wt.% of organic solvent in water
38. A method according to claim 37 wherein the amount of organic solvent in water is 70 wt.%.
39. A method according to any one of claims 36 to 38, wherein the organic solvent is isopropanol.
40. A method according to any one of claims 36 to 39 wherein the amount of xanthan gum is from 10 to 80 wt.%, or from 15 to 30 wt.% in the suspension or slurry.
41. A method according to any one of claims 36 to 40 wherein the pH is increased in step ii) to be from 10 to 13, or from 11 to 13.
42. A method according to any one of claims 36 to 41 wherein the reaction temperature in step iv) is from 50 to 80 °C.
43. A method according to any one of claims 36 to 42, wherein the reaction time in step iv) is from 2h to 5h, or from 2h to 4h, or preferably is 3h.
44. A method according to any one of claims 31 to 43, wherein the reaction is conducted in the presence of nitrogen atmosphere.
45. A method according to any one of claims 36 to 44 further comprising the step of vi) physically separating the crosslinked and hydrophobically modified xanthan gum from the solvent.
46. A method according to claim 45 wherein the polymer is separated by filtration.
47. A method according to any one of claims 45 or claim 46 further comprising the step of vii) washing the crosslinked and hydrophobically modified xanthan gum with an organic solvent or a mixture of an organic solvent and water after physical separation.
48. A method according to claim 47 wherein the crosslinked and hydrophobically modified xanthan gum is washed with a mixture of organic solvent and water comprising from 60 to 90 wt.%, or from 75 to 85 wt. % of organic solvent in water.
49. A method according to any one of claims 47 to 48 wherein the organic solvent in the washing step vii) is isopropanol.
50. A method according to any one of claims 45 to 49 further comprising the step of viii) drying at a temperature from 50 to 115 °C to obtain a dry powder.
51. A method according to claim 50 wherein the drying temperature is from 60 to 80°C.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US202363604379P | 2023-11-30 | 2023-11-30 | |
| US63/604,379 | 2023-11-30 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2025117353A1 true WO2025117353A1 (en) | 2025-06-05 |
Family
ID=93925088
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2024/057020 Pending WO2025117353A1 (en) | 2023-11-30 | 2024-11-22 | Crosslinked and hydrophobically modified xanthan gum |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2025117353A1 (en) |
Citations (20)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3288770A (en) | 1962-12-14 | 1966-11-29 | Peninsular Chem Res Inc | Water soluble quaternary ammonium polymers |
| US3412019A (en) | 1965-05-25 | 1968-11-19 | Calgon Corp | Method of flocculating suspended particulate matter from an aqueous medium |
| US3657175A (en) | 1969-06-26 | 1972-04-18 | Standard Brands Chem Ind Inc | Carboxylic acid latices providing unique thickening and dispersing agents |
| US3835057A (en) | 1968-07-15 | 1974-09-10 | Lever Brothers Ltd | Anti-bacterial detergent bar |
| US4136163A (en) | 1971-02-04 | 1979-01-23 | Wilkinson Sword Limited | P-menthane carboxamides having a physiological cooling effect |
| US4230688A (en) | 1972-04-18 | 1980-10-28 | Wilkinson Sword Limited | Acyclic carboxamides having a physiological cooling effect |
| US4384096A (en) | 1979-08-27 | 1983-05-17 | The Dow Chemical Company | Liquid emulsion polymers useful as pH responsive thickeners for aqueous systems |
| US4464524A (en) | 1983-07-26 | 1984-08-07 | The Sherwin-Williams Company | Polymeric thickeners and coatings containing same |
| US4714653A (en) | 1983-10-28 | 1987-12-22 | Rhone-Poulenc Recherches | Metallizable substrate composites and printed circuits produced therefrom |
| US4772462A (en) | 1986-10-27 | 1988-09-20 | Calgon Corporation | Hair products containing dimethyl diallyl ammonium chloride/acrylic acid-type polymers |
| US4801671A (en) | 1987-06-25 | 1989-01-31 | Desoto, Inc. | Production of alkali-soluble, carboxyl-functional aqueous emulsion thickeners |
| US5136063A (en) | 1990-06-18 | 1992-08-04 | Siltech Inc. | Silicone fatty esters |
| US5180843A (en) | 1990-06-18 | 1993-01-19 | Lenick Jr Anthony J O | Terminal substituted silicone fatty esters |
| US5275809A (en) | 1991-06-28 | 1994-01-04 | Calgon Corporation | Ampholyte terpolymers providing superior conditioning properties in shampoos and other hair care products |
| US5292843A (en) | 1992-05-29 | 1994-03-08 | Union Carbide Chemicals & Plastics Technology Corporation | Polymers containing macromonomers |
| US6183766B1 (en) | 1999-02-12 | 2001-02-06 | The Procter & Gamble Company | Skin sanitizing compositions |
| US6488943B1 (en) | 1999-04-13 | 2002-12-03 | The Procter & Gamble Company | Antimicrobial wipes which provide improved immediate germ reduction |
| US6573375B2 (en) | 2000-12-20 | 2003-06-03 | Union Carbide Chemicals & Plastics Technology Corporation | Liquid thickener for surfactant systems |
| US7001594B1 (en) | 2000-10-10 | 2006-02-21 | The Procter & Gamble Company | Scalp cosmetic compositions and corresponding methods of application to provide scalp moisturization and skin active benefits |
| CN108219766A (en) * | 2016-12-14 | 2018-06-29 | 中国石油天然气股份有限公司 | Polymer thickener and its preparation method and application |
-
2024
- 2024-11-22 WO PCT/US2024/057020 patent/WO2025117353A1/en active Pending
Patent Citations (21)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3288770A (en) | 1962-12-14 | 1966-11-29 | Peninsular Chem Res Inc | Water soluble quaternary ammonium polymers |
| US3412019A (en) | 1965-05-25 | 1968-11-19 | Calgon Corp | Method of flocculating suspended particulate matter from an aqueous medium |
| US3835057A (en) | 1968-07-15 | 1974-09-10 | Lever Brothers Ltd | Anti-bacterial detergent bar |
| US3657175A (en) | 1969-06-26 | 1972-04-18 | Standard Brands Chem Ind Inc | Carboxylic acid latices providing unique thickening and dispersing agents |
| US4136163A (en) | 1971-02-04 | 1979-01-23 | Wilkinson Sword Limited | P-menthane carboxamides having a physiological cooling effect |
| US4230688A (en) | 1972-04-18 | 1980-10-28 | Wilkinson Sword Limited | Acyclic carboxamides having a physiological cooling effect |
| US4384096A (en) | 1979-08-27 | 1983-05-17 | The Dow Chemical Company | Liquid emulsion polymers useful as pH responsive thickeners for aqueous systems |
| US4464524A (en) | 1983-07-26 | 1984-08-07 | The Sherwin-Williams Company | Polymeric thickeners and coatings containing same |
| US4714653A (en) | 1983-10-28 | 1987-12-22 | Rhone-Poulenc Recherches | Metallizable substrate composites and printed circuits produced therefrom |
| US4772462A (en) | 1986-10-27 | 1988-09-20 | Calgon Corporation | Hair products containing dimethyl diallyl ammonium chloride/acrylic acid-type polymers |
| US4801671A (en) | 1987-06-25 | 1989-01-31 | Desoto, Inc. | Production of alkali-soluble, carboxyl-functional aqueous emulsion thickeners |
| US5136063A (en) | 1990-06-18 | 1992-08-04 | Siltech Inc. | Silicone fatty esters |
| US5180843A (en) | 1990-06-18 | 1993-01-19 | Lenick Jr Anthony J O | Terminal substituted silicone fatty esters |
| US5275809A (en) | 1991-06-28 | 1994-01-04 | Calgon Corporation | Ampholyte terpolymers providing superior conditioning properties in shampoos and other hair care products |
| US5292843A (en) | 1992-05-29 | 1994-03-08 | Union Carbide Chemicals & Plastics Technology Corporation | Polymers containing macromonomers |
| US6183766B1 (en) | 1999-02-12 | 2001-02-06 | The Procter & Gamble Company | Skin sanitizing compositions |
| US6488943B1 (en) | 1999-04-13 | 2002-12-03 | The Procter & Gamble Company | Antimicrobial wipes which provide improved immediate germ reduction |
| US7001594B1 (en) | 2000-10-10 | 2006-02-21 | The Procter & Gamble Company | Scalp cosmetic compositions and corresponding methods of application to provide scalp moisturization and skin active benefits |
| US6573375B2 (en) | 2000-12-20 | 2003-06-03 | Union Carbide Chemicals & Plastics Technology Corporation | Liquid thickener for surfactant systems |
| US6727357B2 (en) | 2000-12-20 | 2004-04-27 | The Lubrizol Corporation | Liquid thickener for surfactant systems |
| CN108219766A (en) * | 2016-12-14 | 2018-06-29 | 中国石油天然气股份有限公司 | Polymer thickener and its preparation method and application |
Non-Patent Citations (6)
| Title |
|---|
| "Harry's Cosmeticology", 1982, CHEMICAL PUBLISHING COMPANY INC., pages: 266 |
| GREGORY D. SHAY: "Polymers in Aqueous Media - Performance Through Association, Advances in Chemistry Series", vol. 223, 1989, ACS, article "Alkali-Swellable and Alkali-Soluble Thickener Technology A Review", pages: 457 - 494 |
| O'LENICK, A. J., JR.: "Guerbet chemistry", JOURNAL OF SURFACTANTS AND DETERGENTS, vol. 4, 2001, pages 311 - 315, XP001078149, DOI: 10.1007/s11743-001-0185-1 |
| QUAN HONGPING ET AL: "Preparation and property evaluation of a hydrophobically modified xanthan gum XG-C16", JOURNAL OF DISPERSION SCIENCE AND TECHNOLOGY., vol. 41, no. 5, 14 May 2019 (2019-05-14), NEW YORK, NY, US, pages 656 - 666, XP093247242, ISSN: 0193-2691, DOI: 10.1080/01932691.2019.1610425 * |
| RASCHIP IRINA ELENA ET AL: "Ice-Templated and Cross-Linked Xanthan-Based Hydrogels: Towards Tailor-Made Properties", GELS, vol. 9, no. 7, 29 June 2023 (2023-06-29), pages 528, XP093247347, ISSN: 2310-2861, DOI: 10.3390/gels9070528 * |
| RIAZ TAHREEM ET AL: "A review of the enzymatic, physical, and chemical modification techniques of xanthan gum", INTERNATIONAL JOURNAL OF BIOLOGICAL MACROMOLECULES, vol. 186, 1 September 2021 (2021-09-01), NL, pages 472 - 489, XP093247247, ISSN: 0141-8130, DOI: 10.1016/j.ijbiomac.2021.06.196 * |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP2861204B1 (en) | Alkyl glycoside-based micellar thickeners for surfactant systems | |
| KR101947360B1 (en) | Acrylate copolymer thickeners | |
| EP2591010B1 (en) | Blends of acrylic copolymer thickeners | |
| EP2591027B1 (en) | Structured acrylate copolymer thickeners | |
| CN105793273B (en) | Dihydroxyalkyl-substituted polygalactomannans and methods for their preparation and use | |
| EP2558525B2 (en) | Thickened amino acid surfactant compositions and methods therefor | |
| US20130183361A1 (en) | Structured acrylate copolymer for use in multi-phase systems | |
| JP2014523456A (en) | Cassia derivative | |
| JP2013525579A (en) | Cassia derivative | |
| EP1739161A1 (en) | Foam-enhancing agent | |
| WO2025117353A1 (en) | Crosslinked and hydrophobically modified xanthan gum |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 24827356 Country of ref document: EP Kind code of ref document: A1 |