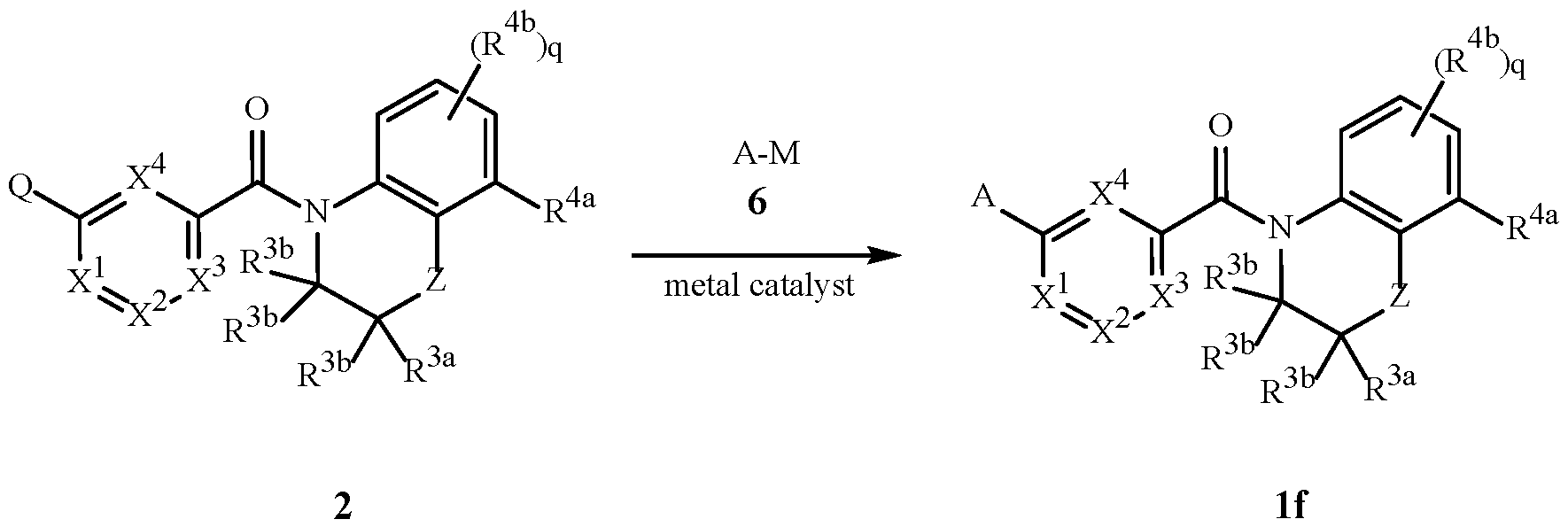
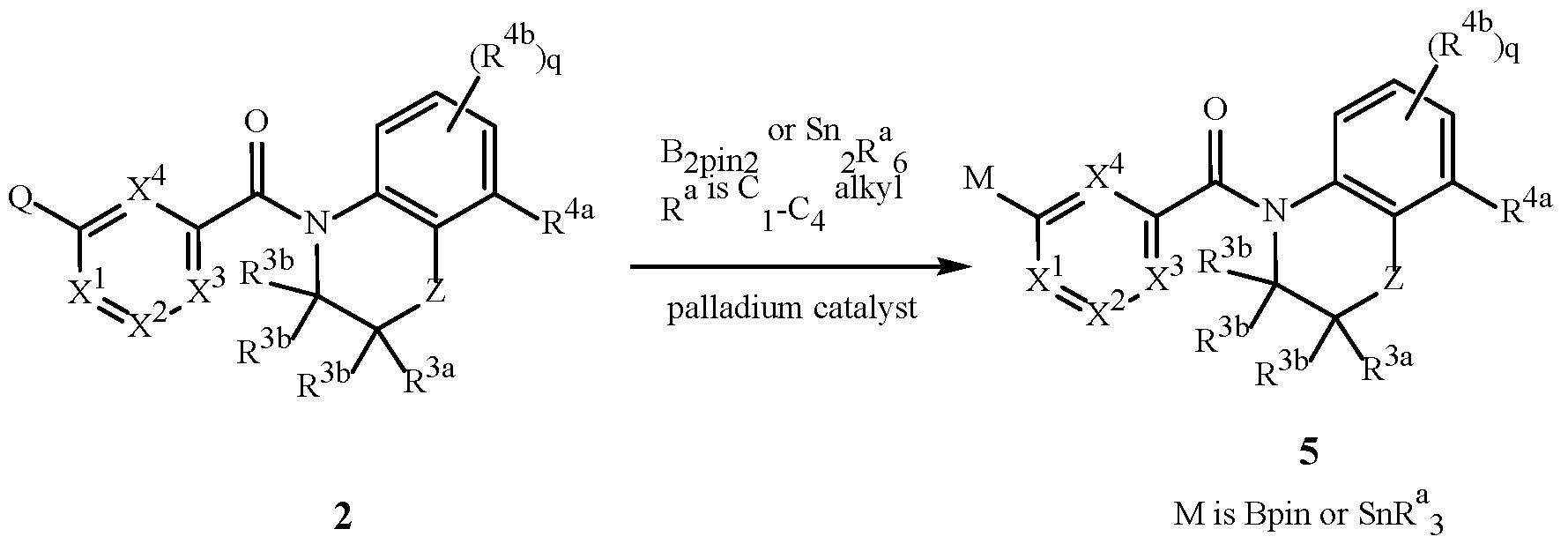
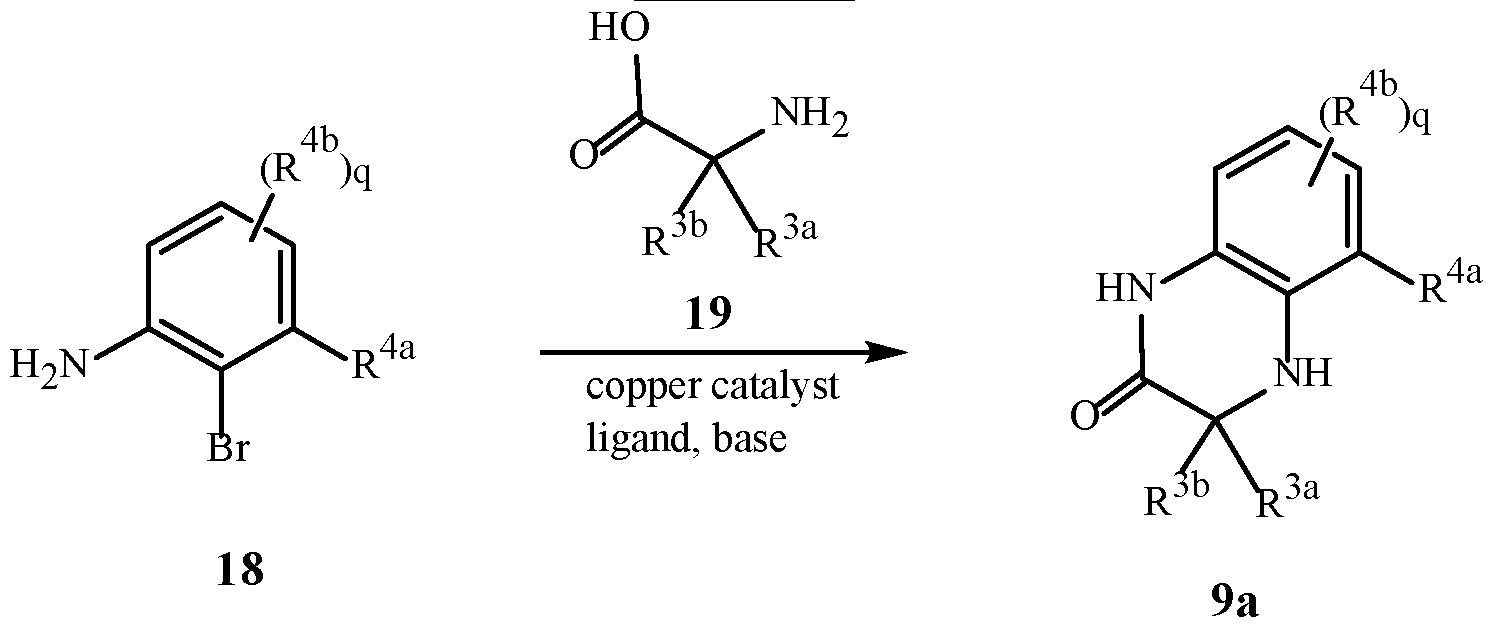
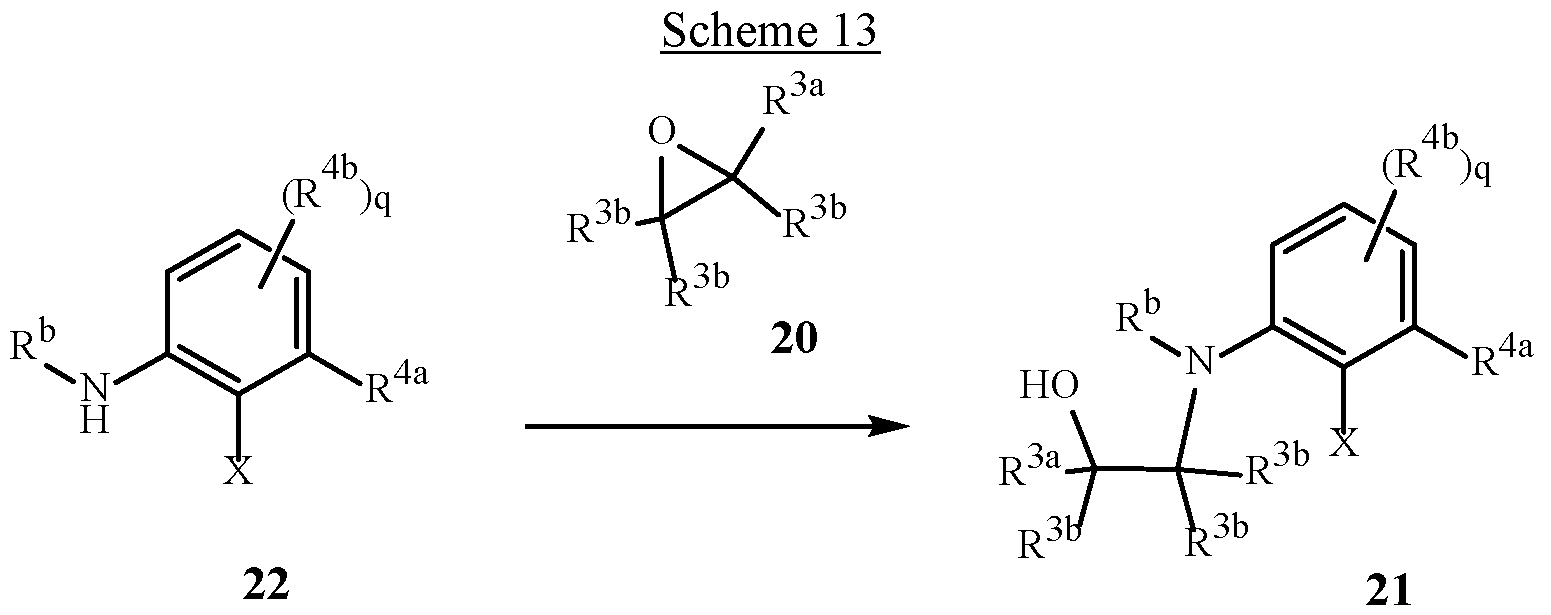
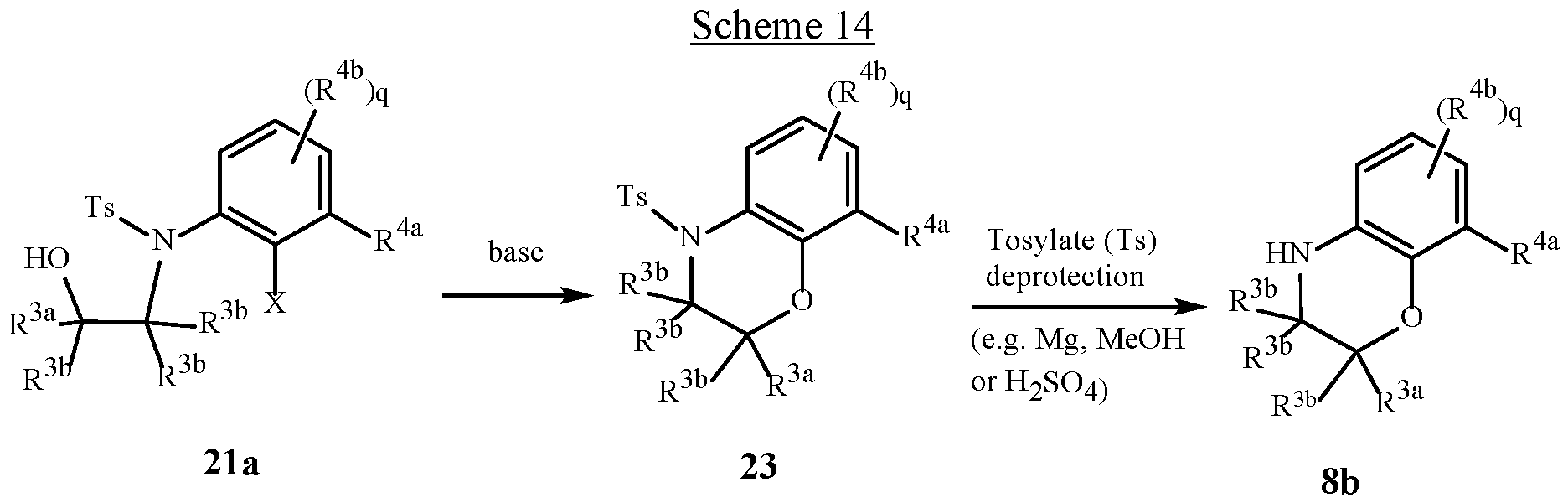
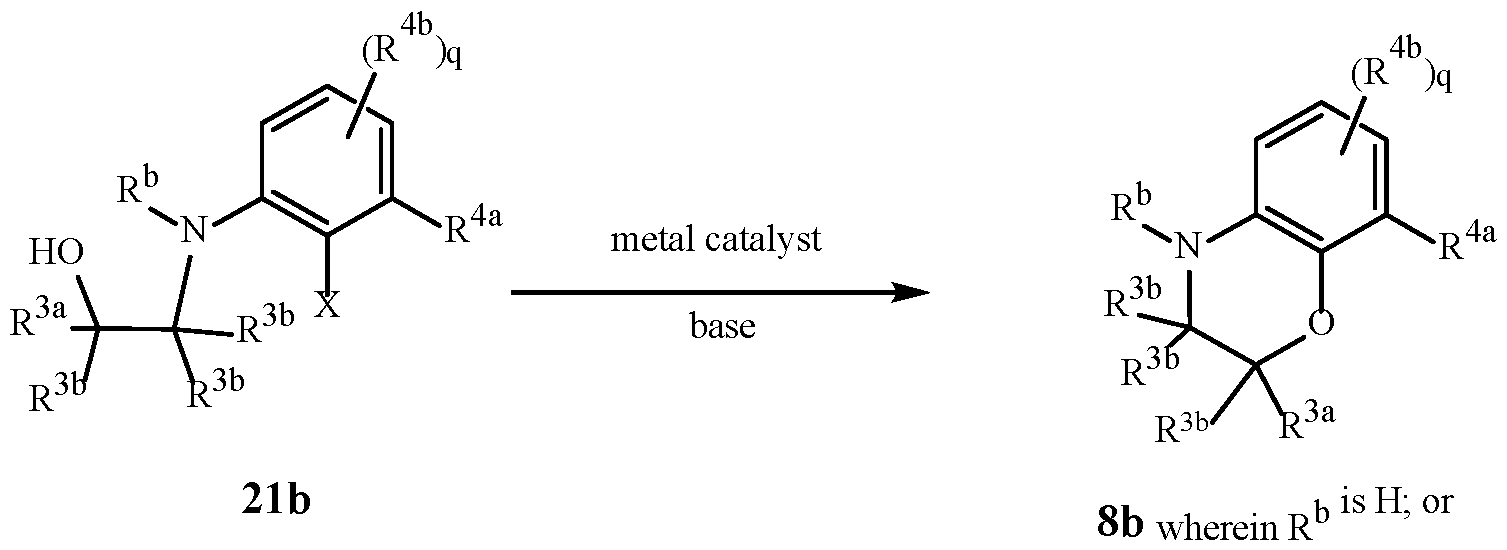
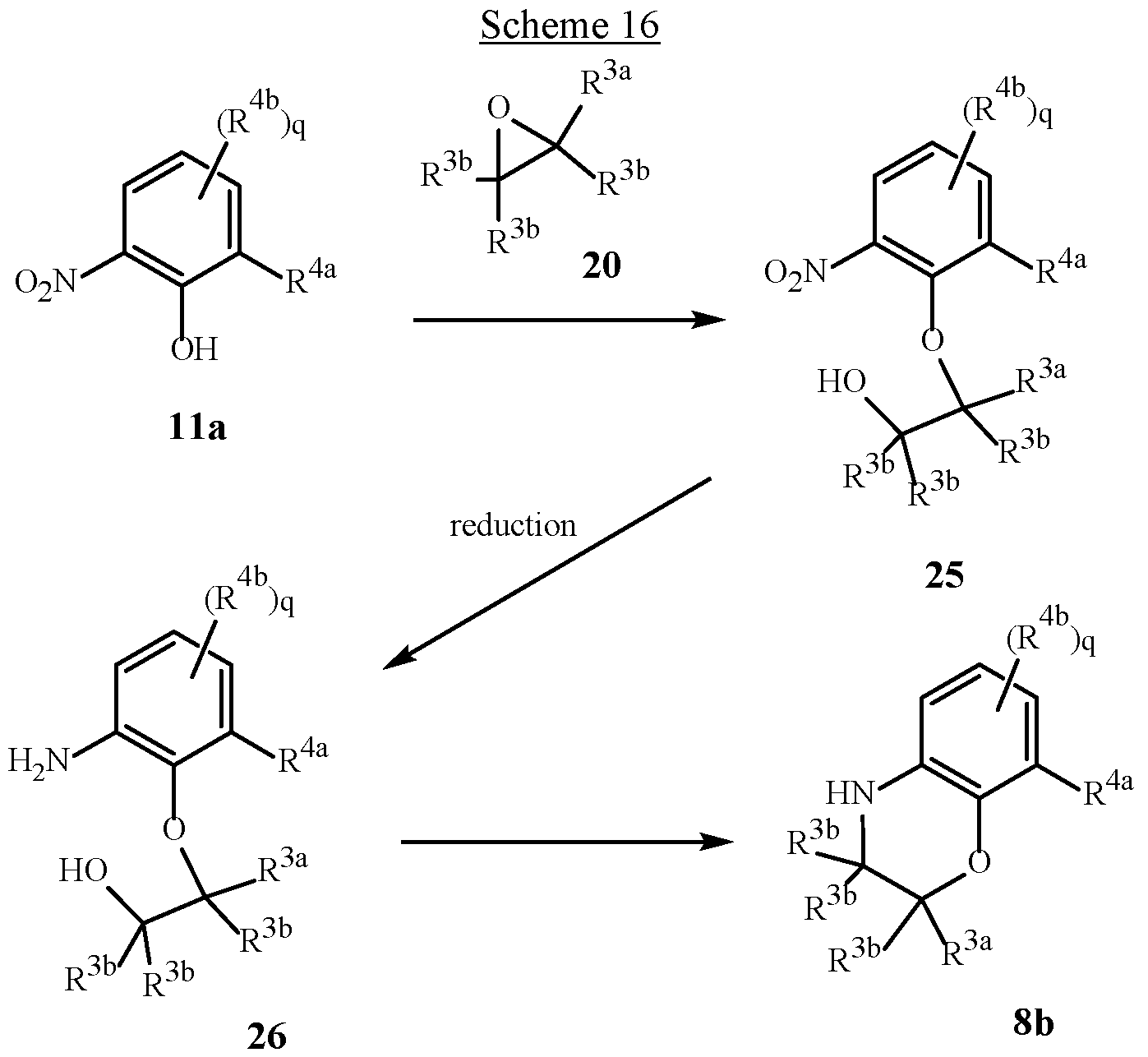
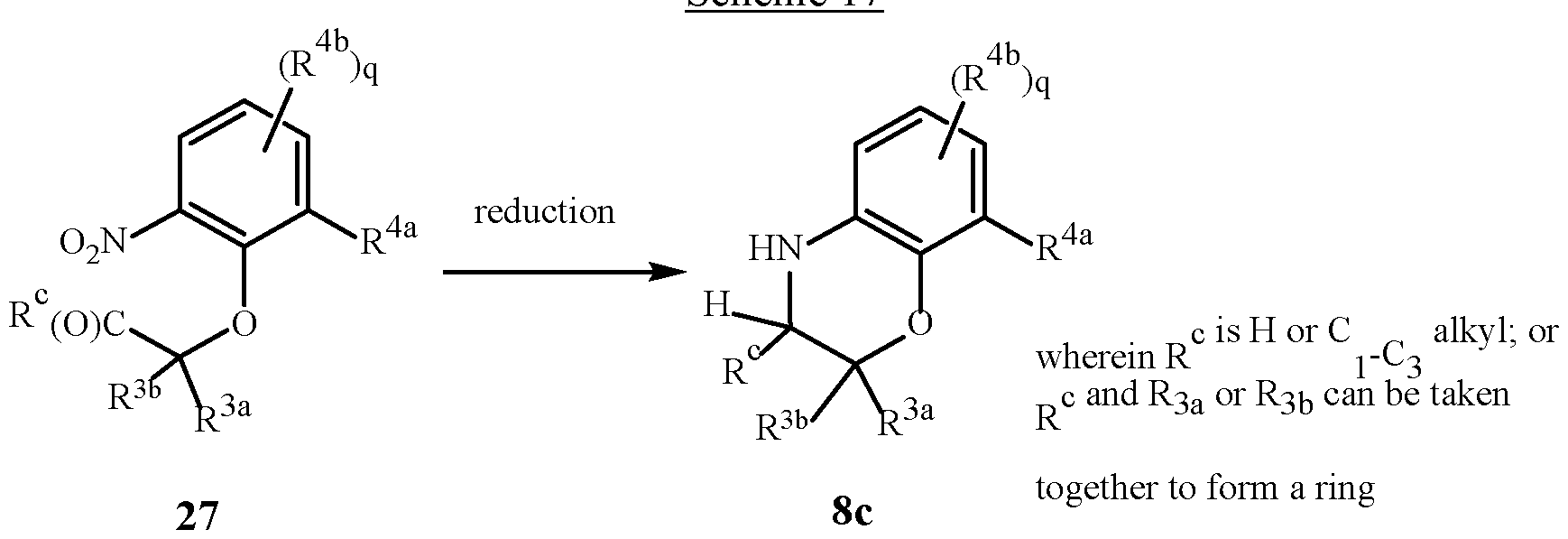
WO2025111184A1 - Substituted tetrahydroquinoline and tetrahydroquinoxaline herbicides - Google Patents
Substituted tetrahydroquinoline and tetrahydroquinoxaline herbicides Download PDFInfo
- Publication number
- WO2025111184A1 WO2025111184A1 PCT/US2024/055871 US2024055871W WO2025111184A1 WO 2025111184 A1 WO2025111184 A1 WO 2025111184A1 US 2024055871 W US2024055871 W US 2024055871W WO 2025111184 A1 WO2025111184 A1 WO 2025111184A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- alkyl
- cycloalkyl
- independently
- compound
- haloalkyl
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D413/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms
- C07D413/14—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms containing three or more hetero rings
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/64—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with three nitrogen atoms as the only ring hetero atoms
- A01N43/647—Triazoles; Hydrogenated triazoles
- A01N43/653—1,2,4-Triazoles; Hydrogenated 1,2,4-triazoles
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01P—BIOCIDAL, PEST REPELLANT, PEST ATTRACTANT OR PLANT GROWTH REGULATORY ACTIVITY OF CHEMICAL COMPOUNDS OR PREPARATIONS
- A01P13/00—Herbicides; Algicides
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D401/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom
- C07D401/02—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings
- C07D401/10—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings linked by a carbon chain containing aromatic rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D401/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom
- C07D401/14—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing three or more hetero rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D403/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00
- C07D403/02—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00 containing two hetero rings
- C07D403/10—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00 containing two hetero rings linked by a carbon chain containing aromatic rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D405/00—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom
- C07D405/14—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom containing three or more hetero rings
Definitions
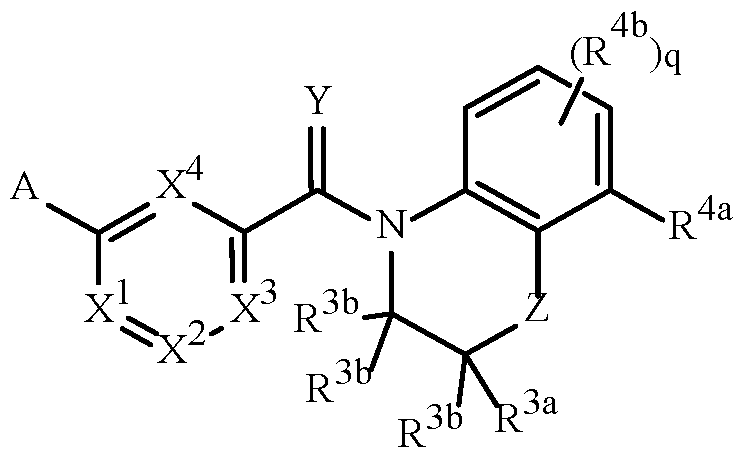
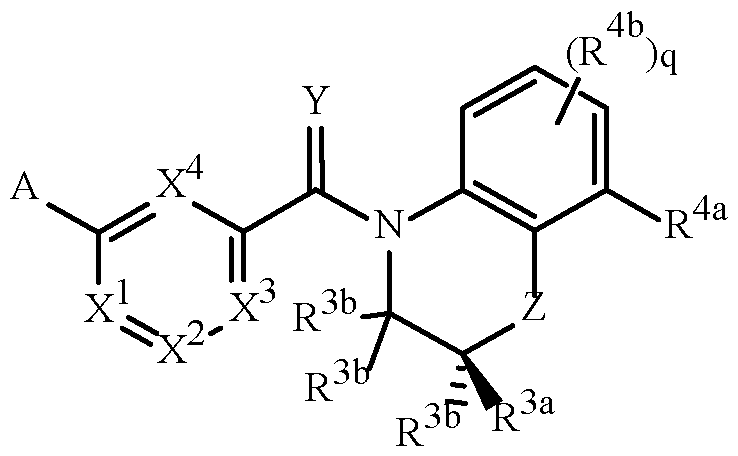
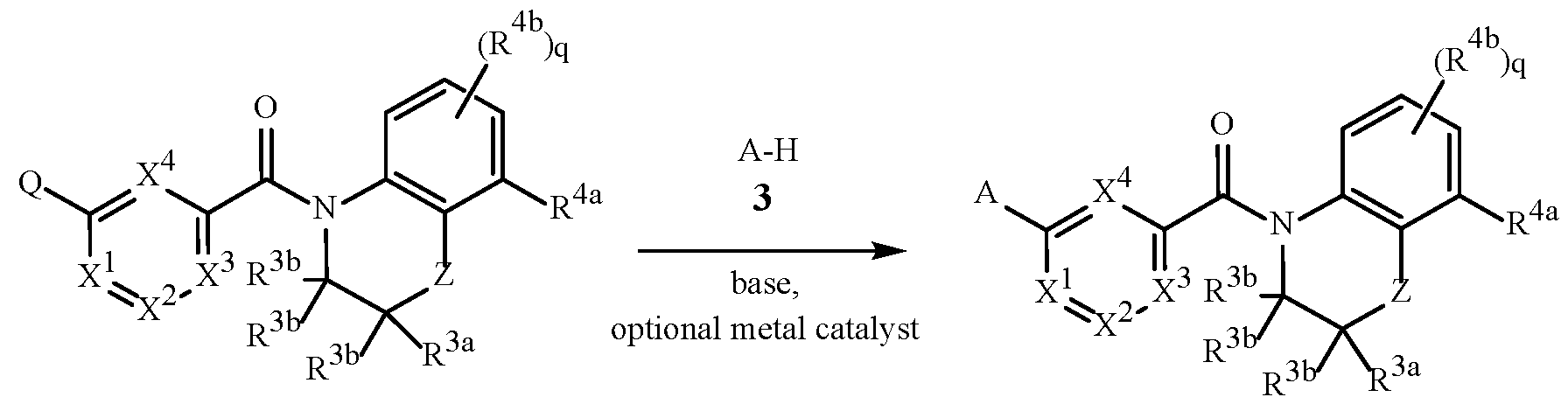
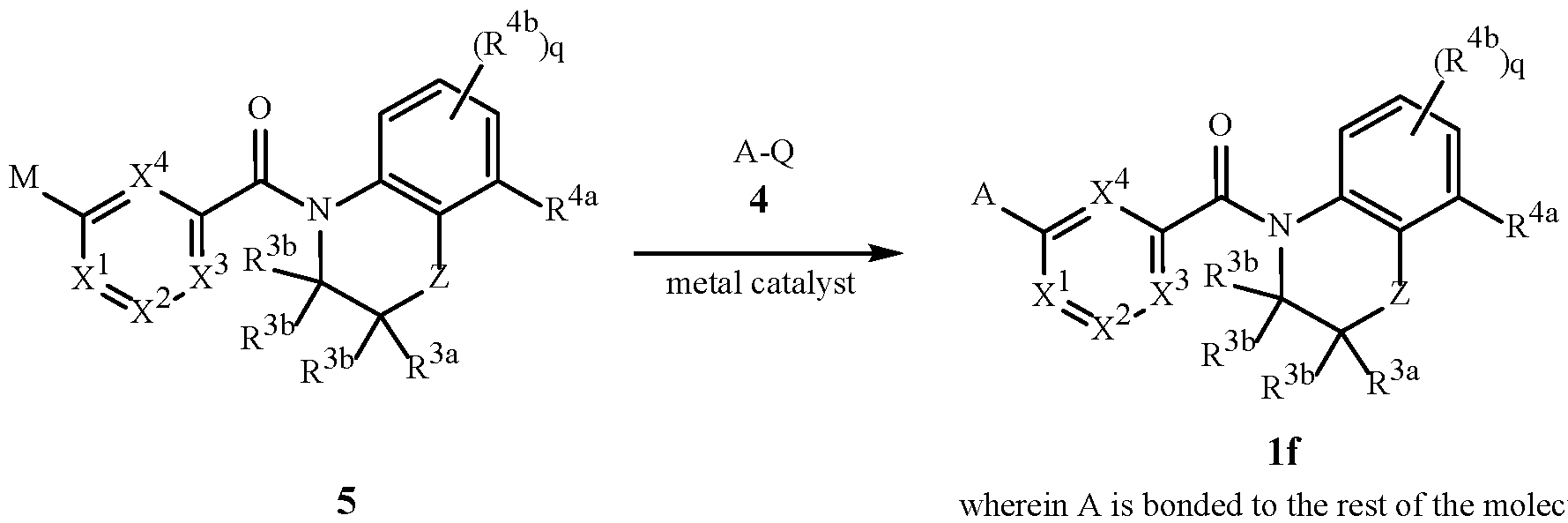
- A is a 5- or 6-membered heterocyclic ring, containing ring members selected from carbon atoms and up to 4 heteroatoms independently selected from up to 2 oxygen atoms, up to 2 sulfur atoms and up to 4 nitrogen atoms, one or two carbon or sulfur ring members of the heterocycle can optionally be in the oxidized form of a carbonyl, thiocarbonyl, sulfonyl, sulfinyl moiety, said ring bound to the remainder of Formula 1 through a carbon atom or a heteroatom, and optionally substituted with 1 to 4 R 1 ;
- R 1 is independently R 1a , (R 1b ) m , R 1c or any combination thereof;
- R 1a is H, halogen, cyano, C 1 –C 7 alkyl, C
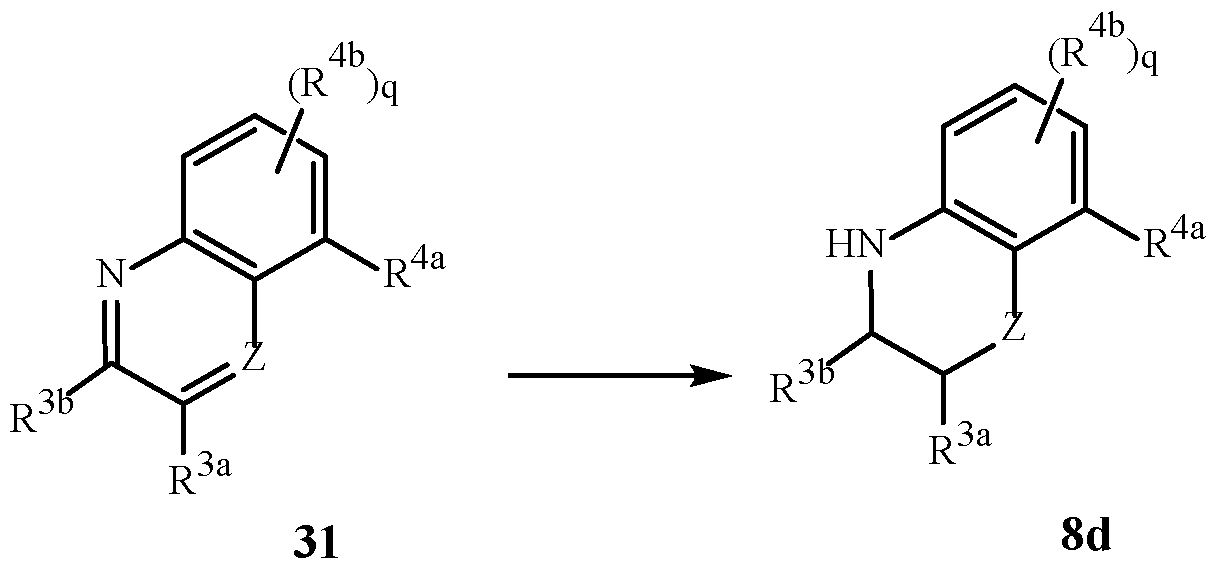
- this invention pertains to a compound of Formula 1 (including all stereoisomers), an N-oxide or a salt thereof.
- This invention also relates to a herbicidal composition comprising a compound of the invention (i.e., in a herbicidally effective amount) and at least one component selected from the group consisting of surfactants, solid diluents and liquid diluents.
- This invention further relates to a method for controlling the growth of undesired vegetation comprising contacting the vegetation or its environment with a herbicidally effective amount of a compound of the invention (e.g., as a composition described herein).
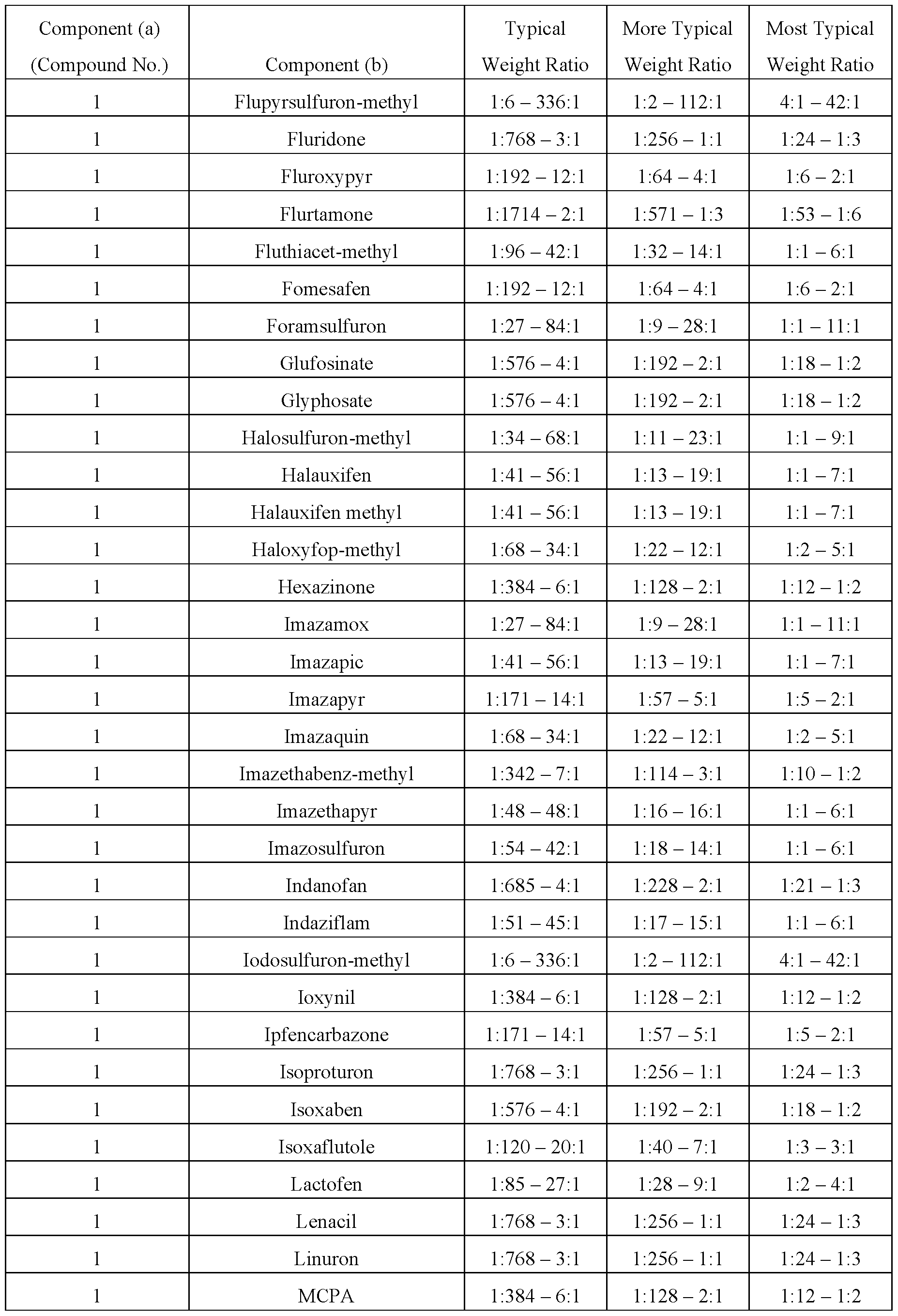
- This invention also includes a herbicidal mixture comprising (a) a compound selected from Formula 1, N-oxides, and salts thereof, and (b) at least one additional active ingredient selected from (b1) through (b18), and salts of compounds of (b1) through (b18), as described below.
- a herbicidal mixture comprising (a) a compound selected from Formula 1, N-oxides, and salts thereof, and (b) at least one additional active ingredient selected from (b1) through (b18), and salts of compounds of (b1) through (b18), as described below.
- compositions, mixture, process or method that comprises a list of elements is not necessarily limited to only those elements but may include other elements not expressly listed or inherent to such composition, mixture, process or method.
- the transitional phrase “consisting of” excludes any element, step, or ingredient not specified. If in the claim, such would close the claim to the inclusion of materials other than those recited except for impurities ordinarily associated therewith.
- the phrase “consisting of” appears in a clause of the body of a claim, rather than immediately following the preamble, it limits only the element set forth in that clause; other elements are not excluded from the claim as a whole.
- transitional phrase “consisting essentially of” is used to define a composition, mixture, method or process that includes materials, steps, features, components, or elements, in addition to those literally disclosed, provided that these additional materials, steps, features, components, or elements do not materially affect the basic and novel characteristic(s) of the claimed invention.
- the term “consisting essentially of” occupies a middle ground between “comprising” and “consisting of”.
- the indefinite articles “a” and “an” preceding an element or component of the invention are intended to be nonrestrictive regarding the number of instances (i.e., occurrences) of the element or component. Therefore “a” or “an” should be read to include one or at least one, and the singular word form of the element or component also includes the plural unless the number is obviously meant to be singular.
- seedling used either alone or in a combination of words means a young plant developing from the embryo of a seed.
- the term “broadleaf” used either alone or in words such as “broadleaf weed” means dicot or dicotyledon, a term used to describe a group of angiosperms characterized by embryos having two cotyledons.
- the term “alkylating” refers reaction in which nucleophile displaces a leaving group such as halide or sulfonate from a carbon-containing radical. Unless otherwise indicated, the term “alkylating” does not limit the carbon-containing radical to alkyl.
- alkyl used either alone or in compound words such as “alkylthio” or “haloalkyl” includes straight-chain or branched alkyl, such as, methyl, ethyl, n-propyl, i-propyl, or the different butyl, pentyl, hexyl and heptyl isomers.
- Alkenyl includes straight-chain or branched alkenes such as ethenyl, 1-propenyl, 2-propenyl, and the different butenyl, pentenyl and hexenyl isomers.
- Alkenyl also includes polyenes such as 1,2-propadienyl and 2,4-hexadienyl.
- Alkynyl includes straight-chain or branched alkynes such as ethynyl, 1-propynyl, 2-propynyl and the different butynyl, pentynyl and hexynyl isomers.
- Alkynyl can also include moieties comprised of multiple triple bonds such as 2,5-hexadiynyl.
- Alkoxy includes, for example, methoxy, ethoxy, n-propyloxy, isopropyloxy and the different butoxy, pentoxy, hexyloxy and heptyloxy isomers.
- Alkoxyalkyl denotes alkoxy substitution on alkyl. Examples of “alkoxyalkyl” include CH 3 OCH 2 , CH 3 OCH 2 CH 2 , CH 3 CH 2 OCH 2 , CH 3 CH 2 CH 2 CH 2 OCH 2 and CH 3 CH 2 OCH 2 CH 2 .
- Alkoxyalkoxy denotes alkoxy substitution on alkoxy.
- Alkoxycycloalkyl denotes an alkoxy substitution on cycloalkyl.
- Alkoxyhaloalkyl denotes an alkoxy substitution on haloalkyl.
- alkoxyhaloalkyl include CH 3 OCF 2 - and CH 3 CH 2 OCCl 2 CH 2 -.
- alkenyloxy denotes an alkenyl moiety bonded through oxygen.
- alkenyloxy may also contain more than one double bond.
- alkynyloxy denotes an alkynyl moiety bonded through oxygen.
- alkynyloxy include HC ⁇ CCH 2 O-, 1-propynyloxy or CH 3 C ⁇ CO-, 2-butynyloxy or CH 3 C ⁇ CCH 2 O-, and the different butynyloxy, pentynyloxy and hexynyloxy isomers.
- alkynyloxy may also contain more than one triple bond.
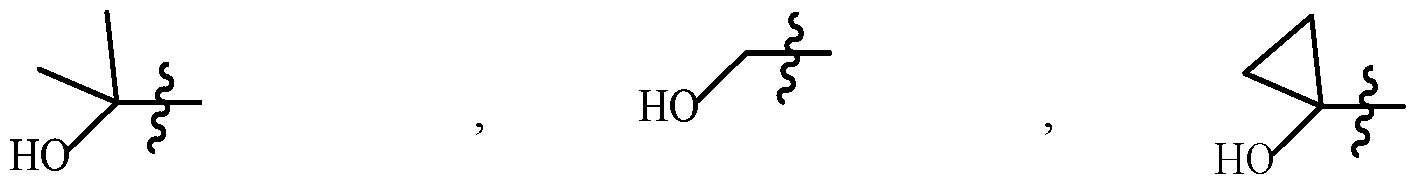
- “Hydroxyalkyl” denotes a hydroxy substitution on alkyl.
- Hydroxycycloalkyl denotes a hydroxy substitution on cycloalkyl.
- “Hydroxyhaloalkyl” denotes a hydroxy substitution on haloalkyl.
- Examples of “hydroxyalkyl”, “hydroxycycloalkyl”, “hydroxyhaloalkyl”, “alkoxycycloalkyl”, “alkoxyhaloalkyl” include the following structures: , 2-OH-propan-2-yl or HOC(Me) 2 hydroxymethyl or HOCH 2 1-OH-cyclopropyl (hydroxyalkyl) (hydroxyalkyl) (hydroxycycloalkyl) .
- Alkylthio includes branched or straight-chain alkylthio moieties such as methylthio, ethylthio, and the different propylthio, butylthio, pentylthio and hexylthio isomers. “Alkylthioalkyl” denotes alkylthio substitution on alkyl.
- alkylthioalkyl examples include CH 3 SCH 2 , CH 3 SCH 2 CH 2 , CH 3 CH 2 SCH 2 , CH 3 CH 2 CH 2 CH 2 SCH 2 , CH 3 CH 2 SCH 2 CH 2 and their different isomers.
- Alkylsulfinyl includes both enantiomers of an alkylsulfinyl group.
- alkylsulfinyl examples include CH 3 S(O)-, CH 3 CH 2 S(O)-, CH 3 CH 2 CH 2 S(O)-, (CH 3 ) 2 CHS(O)- and the different butylsulfinyl, pentylsulfinyl and hexylsulfinyl isomers.
- alkylsulfonyl examples include CH 3 S(O) 2 -, CH 3 CH 2 S(O) 2 -, CH 3 CH 2 CH 2 S(O) 2 -, (CH 3 ) 2 CHS(O) 2 -, and the different butylsulfonyl, pentylsulfonyl and hexylsulfonyl isomers.
- alkylsulfonate examples include CH 3 S(O) 2 O-, CH 3 CH 2 S(O) 2 O-, CH 3 CH 2 CH 2 S(O) 2 O-, (CH 3 ) 2 CHS(O) 2 O-, and the different butylsulfonate, pentylsulfonate and hexylsulfonate isomers.
- Cyanoalkyl denotes an alkyl group substituted with one cyano group.
- Examples of “cyanoalkyl” include NCCH 2 and NCCH 2 CH 2 (alternatively identified as CH 2 CH 2 CN).
- Niroalkyl denotes an alkyl group substituted with one nitro group.
- nitroalkyl examples include NO 2 CH 2 and NO 2 CH 2 CH 2 (alternatively identified as CH 2 CH 2 NO 2 ).
- Cyano means NC- or N ⁇ C-.
- Alkylamino denotes an NH radical substituted with straight-chain or branched alkyl. Examples of “alkylamino” include CH 3 CH 2 NH, CH 3 CH 2 CH 2 NH, and (CH 3 ) 2 CHCH 2 NH. Examples of “dialkylamino” include (CH 3 ) 2 N, (CH 3 CH 2 CH 2 ) 2 N and CH 3 CH 2 (CH 3 )N.
- Alkylsilyl includes a silyl radical substituted with straight-chain or branched alkyl.
- “trialkylsilyl” includes a silyl radical substituted with three straight-chain or branched alkyl. Examples of “trialkylsilyl” include (CH 3 ) 3 Si-, and (CH 3 CH 2 ) 3 Si-.
- “Trialkylsilylalkynyl” denotes trialkylsilyl substitution on alkynyl. Examples of “trialkylsilylalkynyl” include (CH 3 ) 3 SiC ⁇ C-, and (CH 3 CH 2 ) 3 SiC ⁇ C-.
- Cycloalkyl includes, for example, cyclopropyl, cyclobutyl, cyclopentyl and cyclohexyl.
- the term “cycloalkylalkyl” denotes cycloalkyl substitution on an alkyl moiety. Examples of “cycloalkylalkyl” include cyclopropylmethyl, cyclopentylethyl, and other cycloalkyl moieties bonded to straight-chain or branched alkyl groups.
- alkylcycloalkyl denotes an alkyl group bonded to a cycloalkyl moiety and includes, for example, ethylcyclopropyl, i-propylcyclobutyl, methylcyclopentyl and methylcyclohexyl.
- cycloalkoxy denotes cycloalkyl group bonded through oxygen. Examples of “cycloalkoxy” include cyclopropoxy, cyclobutoxy, and cyclopentoxy.
- cycloalkoxyalkyl denotes cycloalkoxy substitution on an alkyl moiety.
- cycloalkoxyalkyl examples include cyclopropoxymethyl, cyclobutoxyethyl, and cyclopentoxymethyl, and other cycloalkoxy moieties bonded to straight-chain or branched alkyl groups.
- alkoxycycloalkyl denotes alkoxy substitution on cycloalkyl.
- alkylthiocycloalkyl denotes alkylthio substitution on cycloalkyl.
- oxacycloalkyl denotes a cycloalkyl with one carbon ring member replaced with an oxygen atom.
- oxacycloalkyl examples include oxacyclopropyl, oxacyclobutyl and oxacyclopentyl.
- thiacycloalkyl denotes a cycloalkyl with one carbon ring member replaced with a sulfur atom.
- thiacycloalkyl examples include thiacyclopropyl, thiacyclobutyl and thiacyclopentyl.
- (O-thia)cycloalkyl denotes a cycloalkyl with one carbon ring member replaced with a -SO group.
- (O-thia)cycloalkyl examples include (O-thia)cyclopropy, (O-thia)cyclobutyl and (O-thia)cyclopentyl.
- the term “(O 2 thia)cycloalkyl” denotes a cycloalkyl with one carbon ring member replaced with a -SO 2 group.
- Examples of “(O 2 thia)cycloalkyl” include (O 2 thia)cyclopropy, (O 2 thia)cyclobutyl and (O 2 thia)cyclopentyl.
- halogen either alone or in compound words such as “haloalkyl”, or when used in descriptions such as “alkyl substituted with halogen” includes fluorine, chlorine, bromine or iodine. Further, when used in compound words such as “haloalkyl”, or when used in descriptions such as “alkyl substituted with halogen” said alkyl may be partially or fully substituted with halogen atoms which may be the same or different. Examples of “haloalkyl” or “alkyl substituted with halogen” include F 3 C, ClCH 2 , CF 3 CH 2 and CF 3 CCl 2 .
- haloalkoxy refers to any one of the following groups: “haloalkoxy” and “haloalkylthio”, “haloalkenyl”, “haloalkynyl”, “halocycloalkyl”, “haloalkylcycloalkyl”, “haloalkylsulfinyl”, “haloalkylsulfonyl” and the like, are as defined analogously to the term “haloalkyl”.
- haloalkoxy include CF 3 O-, CCl 3 CH 2 O-, HCF 2 CH 2 CH 2 O- and CF 3 CH 2 O-.
- haloalkoxyalkyl examples include CF 3 OCH 2 -, CCl 3 CH 2 OCH 2 -, HCF 2 CH 2 CH 2 OCH 2 - and CF 3 CH 2 OCH 2 -.
- haloalkylthio examples include CCl 3 S-, CF 3 S-, CCl 3 CH 2 S- and ClCH 2 CH 2 CH 2 S-.
- haloalkynyl examples include HC ⁇ CCHCl-, CF 3 C ⁇ C-, CCl 3 C ⁇ C- and FCH 2 C ⁇ CCH 2 -.
- halocycloalkyl examples include 1-chlorocyclopropyl, 2-chlorocyclopropyl, 2-fluorocyclopropyl, 1-chlorocyclobutyl, 1-fluorocyclobutyl and 2-fluorocyclobutyl.
- haloalkylcycloalkyl examples include 1-(chloromethyl)cyclopropyl, 2-(chloromethyl)cyclopropyl, 2- (fluoromethyl)cyclopropyl, 1-(chloromethyl)cyclobutyl, 2-(fluoroethyl)cyclobutyl and 2- (fluoromethyl)cyclobutyl.
- alkoxycarbonylalkyl denotes a straight-chain or branched alkoxycarbonyl moiety bonded through an alkyl moiety.
- alkylcarbonylalkyl denotes a straight or branched alkylcarbonyl moiety bonded through an alkyl moiety.
- alkanediyl or alkenediyl refers to a linear or branched alkane or alkene linking chain respectively.
- alkanediyl examples include —CH 2 –, –CH 2 CH(CH 3 )– or -CH 2 CH 2 CH 2 –.
- adjacent in the context of locating a substituent means “next to” or “immediately next to”.
- Alkylsulfoximinoalkyl denotes an alkylsulfoximine or cycloalkylsulfoximine substitution on alkyl or cycloalkyl.
- the total number of carbon atoms in a substituent group is indicated by the “C i –C j ” prefix where i and j are numbers from 1 to 8.
- C 1 –C 4 alkylsulfonyl designates methylsulfonyl through butylsulfonyl
- C 3 –C 6 alkylcarbonylalkyl can be, for example, CH 3 COCH 2 -, CH 3 COCH 2 CH 2 - or CH 3 CH 2 COCH 2 CH 2 CH 2 -
- C 4 –C 7 alkylcycloalkyl can be, for example, methylcyclopropyl, methylcyclobutyl, ethylcyclopropyl, or propylcyclobutyl
- C 2 alkoxyalkyl designates CH 3 OCH 2 -
- C 3 alkoxyalkyl designates, for example, CH 3 CH(OCH 3 )- , CH 3 OCH 2 CH 2 - or CH 3 CH 2 OCH 2 -
- C 4 alkoxyalkyl designates the various isomers of an alkyl group substituted with an alkoxy group containing a total of four carbon atom
- a wavy line in a structure fragment denotes the attachment point of the fragment to the remainder of the molecule.
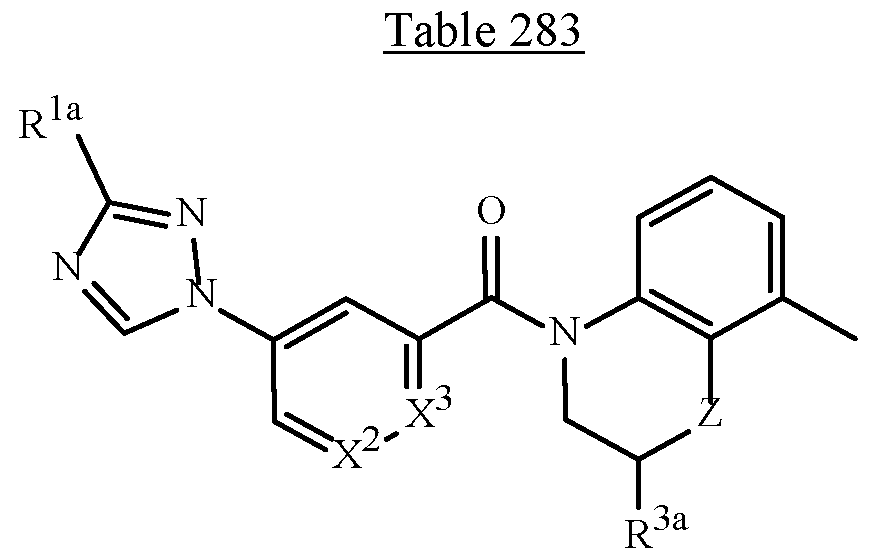
- the variables X 2 and X 3 in Formula 1 are defined as J-21 (i.e., X 1 and X 4 are C-H; X 2 and X 3 are C-R 2 ; and the two adjacent R 2 variables are taken together to form the ring)
- the wavy lines bisecting the bonds of J-21 in the position identified with asterisks (*) of the fused ring J-21 means that the fused ring J-21 is attached to the remainder of the structure of Formula 1 at said positions, as shown below.
- a "-" at the beginning of a fragment definition denotes the attachment point of said fragment to the remainder of the molecule; for example, "-CH 2 CH 2 OMe” denotes the fragment 2-methoxyethyl.
- a group with two short lines at the ends means the group is bonded to other moieties of the molecule through the two atoms right next to the lines. Some examples of these groups are -OCH 2 O- or -O(CH 2 ) 2 O- or -O(CH 2 ) 2 -.
- Cyclic fragments are represented by the use of two "-" within parentheses; for example, the fragment 1-cyanocyclopropyl is represented by "-C(CN)(-CH 2 CH 2 -)", wherein a carbon atom is bonded to both terminal carbon atoms of the two-carbon chain, as illustrated below.
- Naming of substituents in the present disclosure uses recognized terminology providing conciseness in precisely conveying to those skilled in the art the chemical structure. For conciseness, locant descriptors may be omitted.
- Compounds of this invention can exist as one or more stereoisomers.
- stereoisomers include enantiomers, diastereomers, cis-trans isomers or Z / E isomers (also known as geometric isomers) and atropisomers (also known as conformational isomers).
- Stereoisomers are isomers of identical constitution but differing in the arrangement of their atoms in space and include enantiomers, diastereomers, cis-trans isomers (also known as geometric isomers) and atropisomers.
- Atropisomers result from restricted rotation about single bonds where the rotational barrier is high enough to permit isolation of the isomeric species.
- one stereoisomer may be more active and/or may exhibit beneficial effects when enriched relative to the other stereoisomer(s) or when separated from the other stereoisomer(s). Additionally, the skilled artisan knows how to separate, enrich, and/or to selectively prepare said stereoisomers. For a comprehensive discussion of all aspects of stereoisomerism, see Ernest L. Eliel and Samuel H. Wilen, Stereochemistry of Organic Compounds, John Wiley & Sons, 1994. The compounds of the invention may be present as a mixture of stereoisomers, individual stereoisomers or as an optically active form.
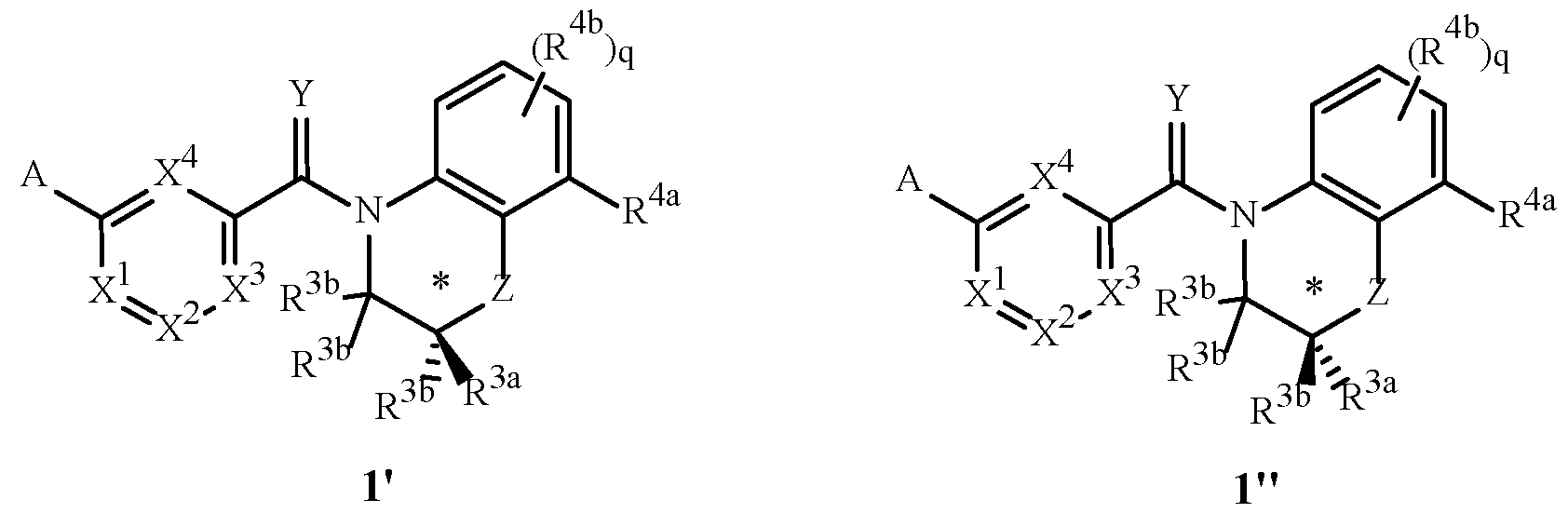
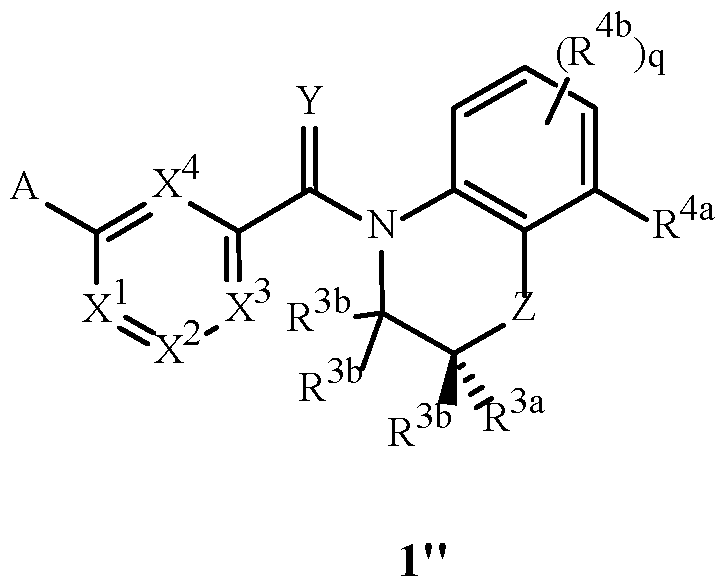
- two possible stereoisomers of Formula 1 are depicted as Formula 1' and 1'' involving the chiral center identified in the structures below with an asterisk (*).
- Compounds of Formula 1 can also comprise additional chiral centers.
- substituents and other molecular constituents such as R 2 may themselves contain chiral centers.
- This disclosure comprises racemic mixtures as well as enriched and essentially pure stereoconfigurations at these additional chiral centers.
- Molecular depictions drawn herein follow standard conventions for depicting stereochemistry. To indicate stereoconfiguration, bonds rising from the plane of the drawing and towards the viewer are denoted by solid wedges wherein the broad end of the wedge is attached to the atom rising from the plane of the drawing towards the viewer.
- Bonds going below the plane of the drawing and away from the viewer are denoted by dashed wedges wherein the broad end of the wedge is attached to the atom further away from the viewer.
- the compounds of the disclosure can exist as stereoisomers due to the possible chiral carbon atoms present in Formula 1.
- this disclosure comprises the individual stereoisomers of the compounds of Formula 1, as well as mixtures of stereoisomers of the compounds of Formula 1.
- This disclosure comprises racemic mixtures of equal amounts of the enantiomers of Formulae 1' and 1''.
- this disclosure includes mixtures that are enriched in the Formula 1' enantiomer compared to the racemic mixture of Formulae 1' and 1''.
- This disclosure also comprises the essentially pure enantiomer of Formula 1'.
- An embodiment of this disclosure comprises mixtures of stereoisomers of the compounds of Formula 1' and Formula 1'', wherein the ratio of 1' to 1'' is at least 75:25 (a 50% enantiomeric excess of 1').
- An embodiment of this disclosure comprises mixtures of stereoisomers of the compounds of Formula 1' and Formula 1'', wherein the ratio of 1' to 1'' is at least 90:10 (an 80% enantiomeric excess of 1').
- An embodiment of this disclosure comprises mixtures of stereoisomers of the compounds of Formula 1' and Formula 1'', wherein the ratio of 1' to 1' is at least 95:5 (a 90% enantiomeric excess of 1').
- An embodiment of this disclosure comprises mixtures of stereoisomers of the compounds of Formula 1' and Formula 1'', wherein the ratio of 1' to 1'' is at least 98:2 (a 96% enantiomeric excess of 1').
- An embodiment of this disclosure comprises mixtures of stereoisomers of the compounds of Formula 1' and Formula 1'', wherein the ratio of 1' to 1'' is at least 99:1 (a 98% enantiomeric excess of 1').
- An embodiment of this disclosure comprises mixtures of stereoisomers of the compounds of Formula 1' and Formula 1'', wherein the ratio of 1' to 1' is essentially 100:0.
- An embodiment of this disclosure comprises the compounds of Formula 1'.
- this disclosure includes mixtures that are enriched in the Formula 1'' enantiomer compared to the racemic mixture of Formulae 1'' and 1'.
- This disclosure also comprises the essentially pure enantiomer of Formula 1''.
- An embodiment of this disclosure comprises mixtures of stereoisomers of the compounds of Formula 1'' and Formula 1', wherein the ratio of 1'' to 1' is at least 75:25 (a 50% enantiomeric excess of 1'').
- An embodiment of this disclosure comprises mixtures of stereoisomers of the compounds of Formula 1'' and Formula 1', wherein the ratio of 1'' to 1' is at least 90:10 (an 80% enantiomeric excess of 1'').
- An embodiment of this disclosure comprises mixtures of stereoisomers of the compounds of Formula 1'' and Formula 1', wherein the ratio of 1'' to 1' is at least 95:5 (a 90% enantiomeric excess of 1'').
- An embodiment of this disclosure comprises mixtures of stereoisomers of the compounds of Formula 1'' and Formula 1', wherein the ratio of 1'' to 1' is at least 98:2 (a 96% enantiomeric excess of 1'').
- An embodiment of this disclosure comprises mixtures of stereoisomers of the compounds of Formula 1'' and Formula 1', wherein the ratio of 1'' to 1' is at least 99:1 (a 98% enantiomeric excess of 1'').
- An embodiment of this disclosure comprises mixtures of stereoisomers of the compounds of Formula 1'' and Formula 1', wherein the ratio of 1'' to 1' is essentially 100:0.
- An embodiment of this disclosure comprises the compounds of Formula 1''.
- Compounds of Formula 1 typically exist in more than one form, and Formula 1 thus include all crystalline and non-crystalline forms of the compounds they represent.
- Non- crystalline forms include embodiments which are solids such as waxes and gums as well as embodiments which are liquids such as solutions and melts.
- Crystalline forms include embodiments which represent essentially a single crystal type and embodiments which represent a mixture of polymorphs (i.e., different crystalline types).
- polymorph refers to a particular crystalline form of a chemical compound that can crystallize in different crystalline forms, these forms having different arrangements and/or conformations of the molecules in the crystal lattice. Although polymorphs can have the same chemical composition, they can also differ in composition due to the presence or absence of co- crystallized water or other molecules, which can be weakly or strongly bound in the lattice. Polymorphs can differ in such chemical, physical and biological properties as crystal shape, density, hardness, color, chemical stability, melting point, hygroscopicity, suspensibility, dissolution rate and biological availability.
- a polymorph of a compound of Formula 1 can exhibit beneficial effects (e.g., suitability for preparation of useful formulations, improved biological performance) relative to another polymorph or a mixture of polymorphs of the same compound of Formula 1.
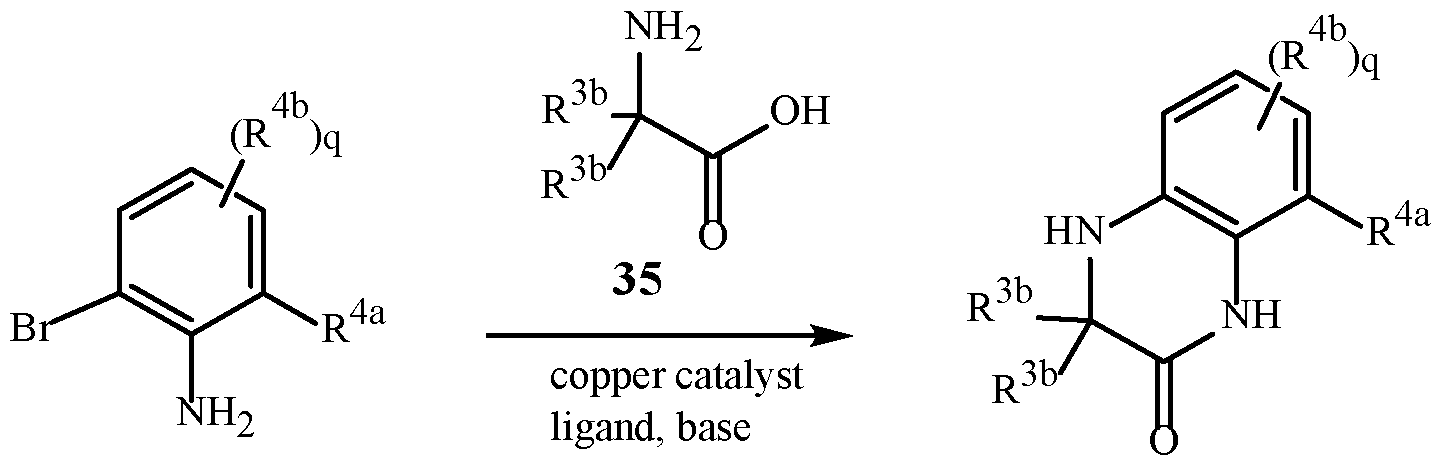
- Preparation and isolation of a particular polymorph of a compound of Formula 1 can be achieved by methods known to those skilled in the art including, for example, crystallization using selected solvents and temperatures.
- crystallization using selected solvents and temperatures.
- nitrogen-containing heterocycles can form N-oxides since the nitrogen requires an available lone pair for oxidation to the oxide; one skilled in the art will recognize those nitrogen-containing heterocycles which can form N-oxides.
- nitrogen-containing heterocycles which can form N-oxides.
- tertiary amines can form N-oxides.
- N-oxides of heterocycles and tertiary amines are very well known by one skilled in the art including the oxidation of heterocycles and tertiary amines with peroxy acids such as peracetic and m-chloroperbenzoic acid (MCPBA), hydrogen peroxide, alkyl hydroperoxides such as t-butyl hydroperoxide, sodium perborate, and dioxiranes such as dimethyldioxirane.
- MCPBA peroxy acids
- alkyl hydroperoxides such as t-butyl hydroperoxide
- sodium perborate sodium perborate
- dioxiranes such as dimethyldioxirane
- salts of chemical compounds are in equilibrium with their corresponding nonsalt forms, salts share the biological utility of the nonsalt forms.
- salts of a compound of Formula 1 are useful for control of undesired vegetation (i.e., are agriculturally suitable).
- the salts of a compound of Formula 1 include acid-addition salts with inorganic or organic acids such as hydrobromic, hydrochloric, nitric, phosphoric, sulfuric, acetic, butyric, fumaric, lactic, maleic, malonic, oxalic, propionic, salicylic, tartaric, 4-toluenesulfonic or valeric acids.
- salts also include those formed with organic or inorganic bases such as pyridine, triethylamine or ammonia, or amides, hydrides, hydroxides or carbonates of sodium, potassium, lithium, calcium, magnesium or barium.
- a “ring” as a component of Formula 1 is carbocyclic or heterocyclic.
- ring system denotes two or more fused rings.
- bicyclic ring denotes a ring system consisting of two fused rings.
- heterocyclic ring denotes a ring in which at least one atom forming the ring backbone is not carbon, e.g., nitrogen, oxygen or sulfur. Typically a heterocyclic ring contains no more than 4 nitrogens, no more than 2 oxygens and no more than 2 sulfurs. Unless otherwise indicated, a heterocyclic ring can be a saturated, partially unsaturated, or fully unsaturated ring. When a fully unsaturated heterocyclic ring satisfies Hückel’s rule, then said ring is also called a “heteroaromatic ring” or “aromatic heterocyclic ring”.
- heterocyclic rings can be attached through any available carbon or nitrogen by replacement of a hydrogen on said carbon or nitrogen.
- “Aromatic” indicates that each of the ring atoms is essentially in the same plane and has a p-orbital perpendicular to the ring plane, and that (4n + 2) ⁇ electrons, where n is a positive integer, are associated with the ring to comply with Hückel’s rule.
- the term “optionally substituted” in connection with a ring refers to groups which are unsubstituted or have at least one non-hydrogen substituent that does not extinguish the biological activity possessed by the unsubstituted analog. As used herein, the following definitions shall apply unless otherwise indicated.
- an optionally substituted group may have a substituent at each substitutable position of the group, and each substitution is independent of the other.
- a group contains a substituent which can be hydrogen, for example R 2 or R 4a , then when this substituent is taken as hydrogen, it is recognized that this is equivalent to said group being unsubstituted.
- a variable group is shown to be optionally attached to a position, for example (R 4b ) q in Formula 1 wherein q may be 0, then hydrogen may be at the remaining positions even if not recited in the definition of the variable group.
- substituents When a compound is substituted with a substituent bearing a subscript that indicates the number of said substituents can exceed 1, then said substituents (when they exceed 1) are independently selected from the group of defined substituents, (e.g., (R 4b ) q in Formula 1 wherein n is 0, 1 or 2), unless otherwise indicated. Further, when the subscript indicates a range, e.g., (R) i–j , then the number of substituents may be selected from the integers between i and j inclusive. The number of optional substituents may be restricted by an expressed limitation.
- a range specified for the number of substituents exceeds the number of positions available for substituents on a ring the actual higher end of the range is recognized to be the number of available positions.
- A can be a 5- or 6-membered heterocyclic ring, optionally substituted with one or more substituents selected from a group of substituents as defined in the Summary of the Invention.
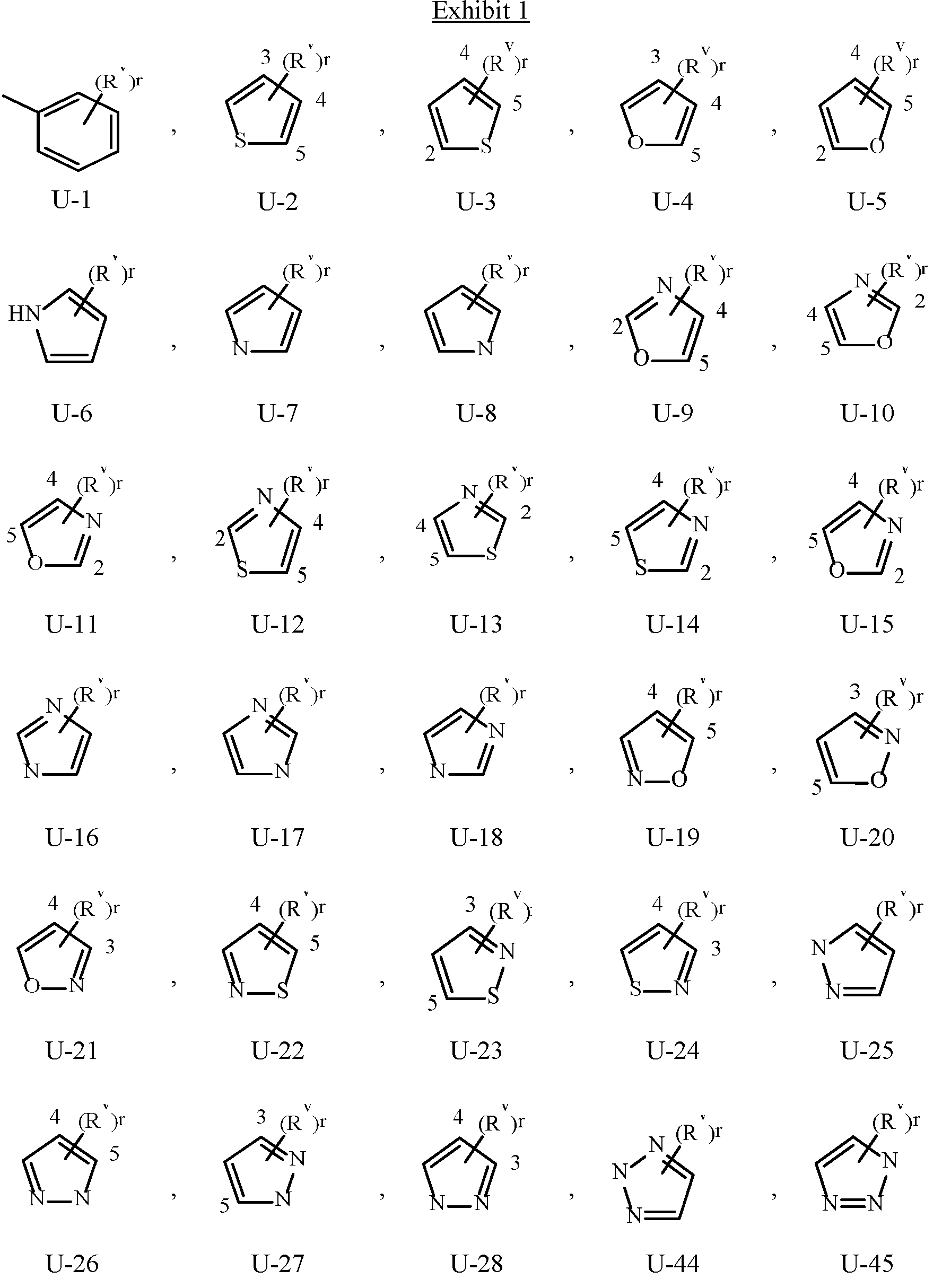
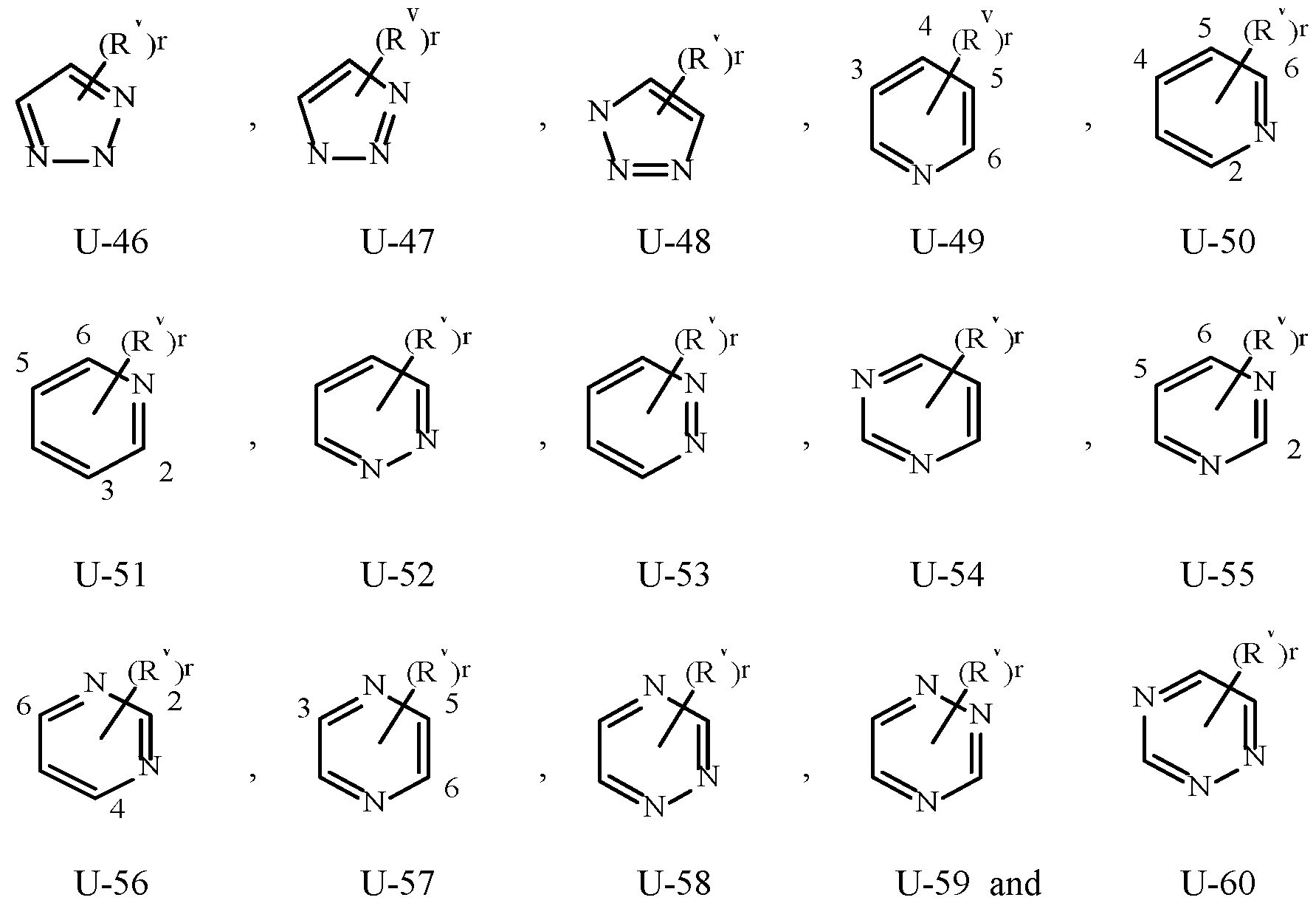
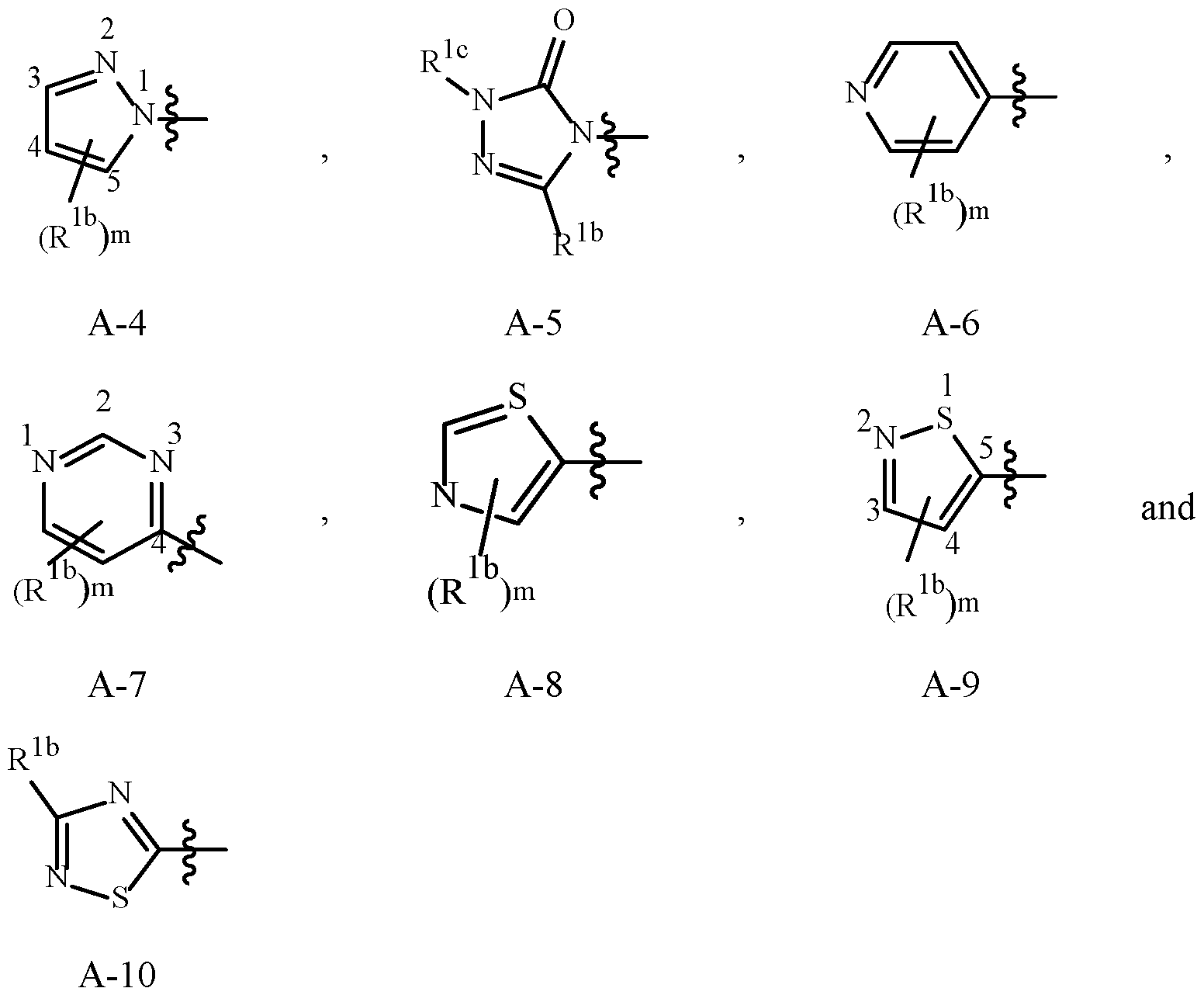
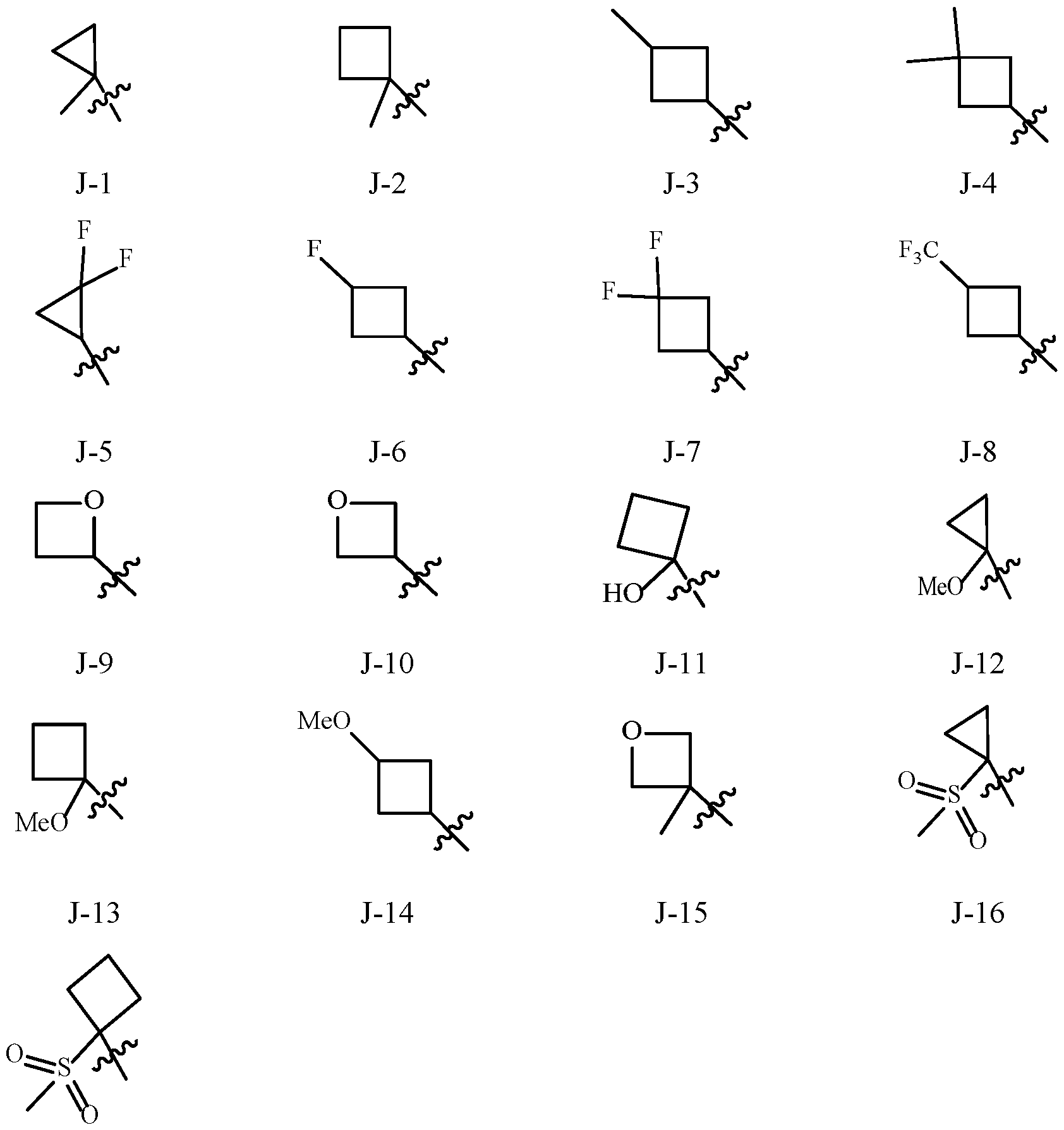
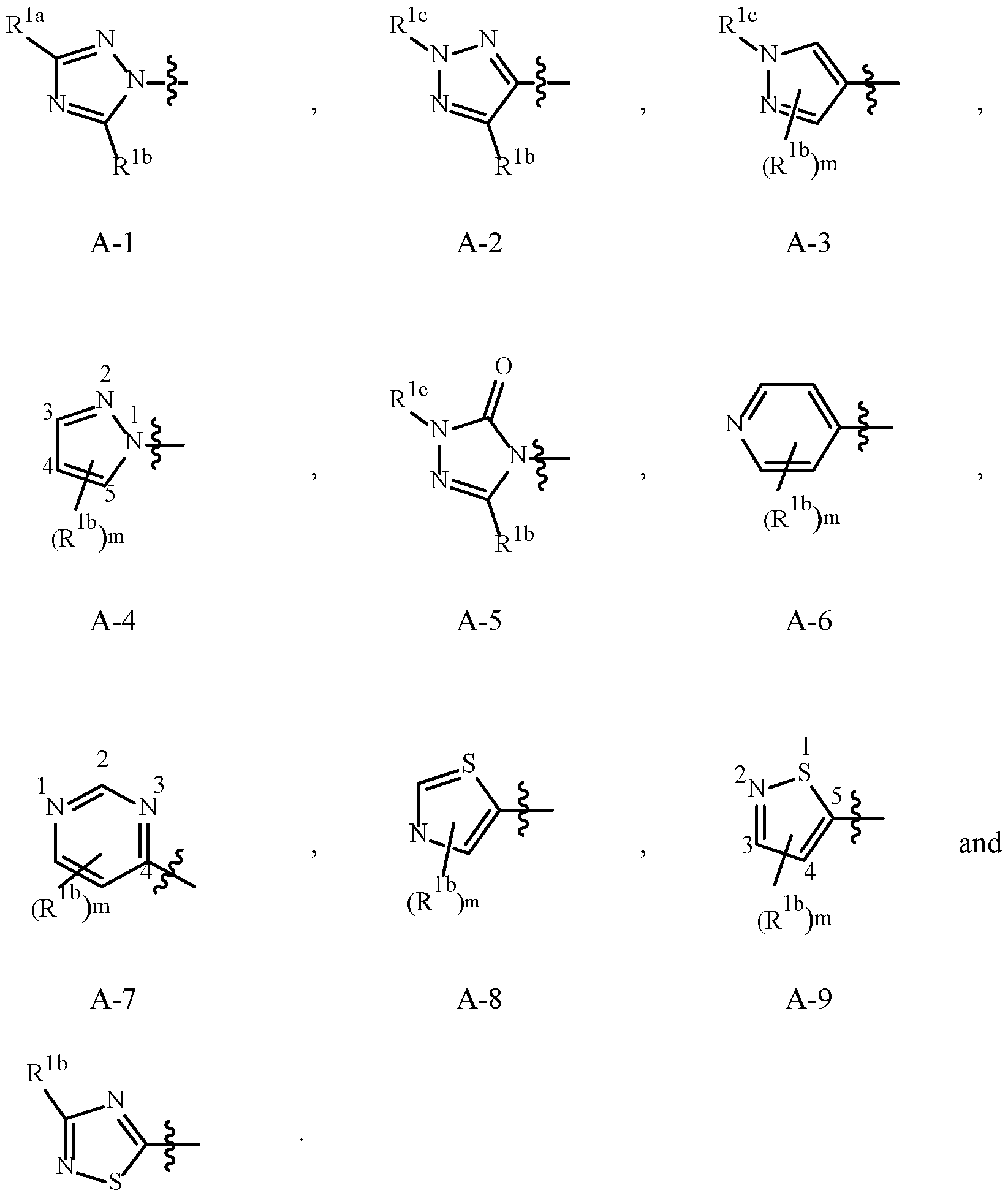
- Examples of a 5- or 6-membered unsaturated aromatic heterocyclic ring optionally substituted with from one or more substituents include the rings U-2 through U-60 illustrated in Exhibit 1 wherein R v is any substituent as defined in the Summary of the Invention on A (i.e., R 1 ) and r is an integer from 0 to 4, limited by the number of available positions on each U group.
- U-29, U-30, U-36, U-37, U-38, U-39, U-40, U-41, U-42 and U-43 have only one available position, for these U groups r is limited to the integers 0 or 1, and r being 0 means that the U group is unsubstituted and a hydrogen is present at the position indicated by (R v ) r .
- two adjacent R 2 may be taken together to form a 5- to 8-membered ring, the 5- to 8-membered ring can be (among others) saturated or unsaturated, optionally substituted with one or more substituents selected from a group of substituents as defined in the Summary of the Invention.
- Examples of a 5- to 8-membered unsaturated aromatic ring optionally substituted with from one or more substituents include the rings U-1 through U-60 illustrated in Exhibit 1 wherein R v is independently H, halogen, cyano, nitro, C 1 –C 4 alkyl, C 3 –C 6 cycloalkyl, C 1 –C 4 haloalkyl, C 1 –C 4 alkoxy and C 1 –C 4 haloalkoxy and r is an integer from 0 to 2, limited by the number of available positions on each U group.
- the U group can share any two available neighboring atoms with the connecting ring. , , , , , , , .
- one or two carbon ring members of the heterocycle can optionally be in the oxidized form of a carbonyl moiety.
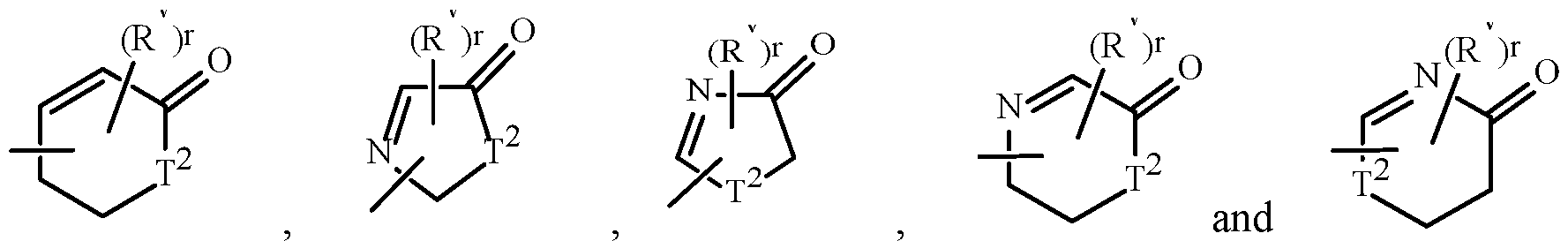
- Examples of a 5- to 8-membered heterocyclic ring that is saturated or non-aromatic unsaturated heterocyclic ring containing ring members selected from up to two O atoms and up to two S atoms, and optionally substituted on carbon atom ring members with up to four R v include the rings T-1 through T-35 as illustrated in Exhibit 2.
- the T group can share any two available neighboring atoms with the connecting ring.
- the optional substituents corresponding to R v can be attached to any available carbon or nitrogen by replacing a hydrogen atom.
- r is typically an integer from 0 to 4, limited by the number of available positions on each T group.
- the term “optionally substituted” means “substituted or unsubstituted”. Note that when T 2 is N, the nitrogen atom can complete its valence by substitution with either H or the substituents corresponding to R v as defined in the Summary of the Invention. , , , , , , , , .
- R v groups are shown in the structures U-1 through U-60, and T-1 through T-35, it is noted that they do not need to be present since they are optional substituents. Note that when R v is H when attached to an atom, this is the same as if said atom is unsubstituted. The nitrogen atoms that require substitution to fill their valence are substituted with H or R v . Note that when the attachment point between (R v ) r and the U (or T) group is illustrated as floating, (R v ) r can be attached to any available carbon atom or nitrogen atom of the U group.
- Formula 1 includes stereoisomers, N-oxides and salts thereof, and reference to “a compound of Formula 1” includes the definitions of substituents specified in the Summary of the Invention unless further defined in the Embodiments.
- Embodiment 1. A compound of Formula 1, stereoisomers, N-oxides, and salts thereof, agricultural compositions containing them and their use as herbicides as described in the Summary of the disclosure.
- Embodiment 2. A compound of Formula 1 or Embodiment 1 wherein A is selected from , Embodiment 2a.
- Embodiment 2b A compound of Embodiment 2 wherein A is A-1, A-2, A-3, A-5, A-7, A-8 or A-10.
- Embodiment 2a wherein A is A-1, A-2 or A-8.
- Embodiment 2c A compound of Embodiment 2b wherein A is A-1 or A-2.
- Embodiment 2d A compound of Embodiment 2c wherein A is A-1.
- Embodiment 3. A compound of Formula 1 or any one of the preceding Embodiments wherein X 1 , X 2 , X 3 and X 4 are each independently N or CR 2 wherein up to two X 1 , X 2 , X 3 and X 4 can be N; Embodiment 3a.
- Embodiment 3aa Embodiment 3a.
- Embodiment 3b. A compound of Embodiment 3a wherein X 1 , X 2 , X 3 and X 4 are each independently CR 2 , and Z is other than O; or X 1 is N, and X 2 , X 3 and X 4 are each independently N or CR 2 , and Z is other than O; or X 1 and X 2 are N, and X 3 and X 4 are each independently CR 2 , and Z is other than O; or X 2 and X 3 are N, and X 1 and X 4 are each independently CR 2 ; or X 1 and X 3 are N, and X 2 and X 4 are each independently CR 2 ; or X 2 is N, and X 1 , X 3 and X 4 are each independently CR 2 ; or X 2 is N, and X 1 ,
- Embodiment 3b A compound of Embodiment 3b wherein X 2 and X 3 are N, and X 1 and X 4 are each independently CR 2 , and Z is O; or X 1 and X 3 are N, and X 2 and X 4 are each independently CR 2 , and Z is O.
- a compound of Embodiment 3b1 wherein X 2 and X 3 are N, and X 1 and X 4 are each independently CR 2 , and Z is O.
- Embodiment 3c A compound of Embodiment 3c.
- a compound of Embodiment 3b wherein X 1 is N, and X 2 , X 3 and X 4 are each independently N or CR 2 , and Z is other than O.
- Embodiment 3g. A compound of Embodiment 3 or any one of the preceding Embodiments wherein X 1 is CR 2 and R 2 is H.
- Embodiment 3h. A compound of Embodiment 3 or any one of the preceding Embodiments wherein X 2 is CR 2 and R 2 is H, C 1 –C 3 alkoxy, F, C 1 –C 3 alkyl, C 3 –C 4 cycloalkyl or taken together with X 3 to form a ring.
- Embodiment 3h wherein the ring contains -OCH 2 O- or -O(CH 2 ) 2 O- or -O(CH 2 ) 2 -.
- Embodiment 3j A compound of Embodiment 3h wherein X 2 is CR 2 and R 2 is H, C 1 –C 3 alkoxy or is taken together with X 3 to form a ring, and said ring contains -OCH 2 O- or -O(CH 2 ) 2 O- or -O(CH 2 ) 2 -.
- Embodiment 3k Embodiment 3k.
- Embodiment 3 A compound of Embodiment 3 or any one of the preceding Embodiments wherein X 3 is CR 2 and R 2 is C 1 –C 4 alkyl, C 1 –C 3 alkoxy, F, Cl, C 3 –C 4 cycloalkyl, C 2 –C 4 alkynyloxy, C 2 –C 3 haloalkoxy, C 1 –C 3 alkylsulfonate or taken together with X 2 to form a ring.
- Embodiment 3l A compound of Embodiment 3k wherein the ring contains -OCH 2 O- or -O(CH 2 ) 2 O- or -O(CH 2 ) 2 -.
- Embodiment 3m Embodiment 3m.
- Embodiment 3k A compound of Embodiment 3k or any one of the preceding Embodiments wherein X 3 is CR 2 and R 2 is C 1 –C 4 alkyl, C 1 –C 3 alkoxy or taken together with X 2 to form a ring.
- Embodiment 3n A compound of Embodiment 3m wherein the ring contains -OCH 2 O- or -O(CH 2 ) 2 O- or -O(CH 2 ) 2 -.
- Embodiment 3o A compound of Embodiment 3k wherein R 2 is C 1 –C 4 alkyl or C 1 –C 3 alkoxy.
- Embodiment 3p A compound of Embodiment 3k or any one of the preceding Embodiments wherein X 3 is CR 2 and R 2 is C 1 –C 4 alkyl, C 1 –C 3 alkoxy or taken together with X 2 to form a ring.
- Embodiment 3n A compound of
- Embodiment 4. A compound of Formula 1 or any one of the preceding Embodiments wherein R 1a is H, halogen, cyano, C 1 –C 7 alkyl, C 2 –C 6 alkenyl, C 2 –C 6 alkynyl, C 3 –C 7 cycloalkyl, C 4 –C 7 cycloalkylalkyl, C 4 –C 7 alkylcycloalkyl, C 1 –C 7 haloalkyl, C 1 –C 6 hydroxyalkyl, C 3 –C 7 hydroxycycloalkyl, C 1 –C 6 hydroxyhaloalkyl, C 4 –C 8 alkoxycycloalkyl, C 2 –C 7 alkoxyhaloalkyl, C 2 –C 6 oxacycloalkyl, C 3 –C 7
- R 1a is H, C 1 –C 7 alkyl, C 2 –C 6 alkenyl, C 2 –C 6 alkynyl, C 3 –C 7 cycloalkyl, C 4 –C 7 cycloalkylalkyl, C 4 –C 7 alkylcycloalkyl, C 1 –C 7 haloalkyl, C 1 –C 6 hydroxyalkyl, C 3 –C 7 hydroxycycloalkyl, C 1 –C 6 hydroxyhaloalkyl, C 4 –C 8 alkoxycycloalkyl, C 2 –C 7 alkoxyhaloalkyl, C 2 –C 6 oxacycloalkyl, C 3 –C 7 oxacycloalkylalkyl, C 3 –C 7 alkyloxacycloalkyl, C 2 –C 6 thiacycloalkyl, C 3 –C 7 thiacycloalkyl
- R 1a is H, C 1 –C 7 alkyl, C 2 –C 6 alkenyl, C 2 –C 6 alkynyl, C 3 –C 7 cycloalkyl, C 4 –C 7 cycloalkylalkyl, C 4 –C 7 alkylcycloalkyl, C 1 –C 7 haloalkyl, C 1 –C 6 hydroxyalkyl, C 3 –C 7 hydroxycycloalkyl, C 1 –C 6 hydroxyhaloalkyl, C 4 –C 8 alkoxycycloalkyl, C 2 –C 7 alkoxyhaloalkyl, C 2 –C 6 oxacycloalkyl, C 3 –C 7 oxacycloalkylalkyl, C 3 –C 7 alkyloxacycloalkyl, C 2 –C 6 thiacycloalkyl, C 3 –C 7 thiacycloalkyl
- R 1a is H, C 1 –C 7 alkyl, C 2 –C 6 alkenyl, C 2 –C 6 alkynyl, C 3 –C 7 cycloalkyl, C 4 –C 7 cycloalkylalkyl, C 4 –C 7 alkylcycloalkyl, C 1 –C 7 haloalkyl, C 1 –C 6 hydroxyalkyl, C 4 –C 8 alkoxycycloalkyl, C 2 –C 7 alkoxyhaloalkyl, C 2 –C 6 oxacycloalkyl, C 3 –C 7 oxacycloalkylalkyl, C 3 –C 7 alkyloxacycloalkyl, C 2 –C 6 thiacycloalkyl, C 3 –C 7 thiacycloalkylalkyl, C 2 –C 6 haloalkenyl, C 2 –C 6 hal
- R 1a is H, C 1 –C 7 alkyl, C 2 –C 6 alkenyl, C 2 –C 6 alkynyl, C 3 –C 7 cycloalkyl, C 4 –C 7 cycloalkylalkyl, C 4 –C 7 alkylcycloalkyl, C 1 –C 7 haloalkyl, C 1 –C 6 hydroxyalkyl, C 4 –C 8 alkoxycycloalkyl, C 2 –C 7 alkoxyhaloalkyl, C 2 –C 6 oxacycloalkyl, C 3 –C 7 oxacycloalkylalkyl, C 3 –C 7 alkyloxacycloalkyl, C 2 –C 6 thiacycloalkyl, C 2 –C 7 alkoxyalkyl, C 1 –C 7 alkoxy, C 3 –C 7 cycloalkoxy, C 4 –
- each R 1b is independently H, halogen, cyano, nitro, C 1 –C 4 alkyl, C 3 –C 5 cycloalkyl, C 1 –C 4 haloalkyl, C 3 –C 5 halocycloalkyl, C 2 –C 4 alkoxyalkyl, C 1 –C 4 alkoxy, C 1 –C 4 alkylthio or C 2 –C 4 alkoxycarbonyl.
- each R 1b is independently H, halogen, cyano, nitro, C 1 –C 4 alkyl, C 3 –C 5 cycloalkyl, C 1 –C 4 haloalkyl, C 3 –C 5 halocycloalkyl, C 2 –C 4 alkoxyalkyl, C 1 –C 4 alkoxy, C 1 –C 4 alkylthio or C 2 –C 4 alkoxycarbonyl.
- Embodiment 5a is independently H, halogen,
- each R 1b is independently H, C 1 –C 4 alkyl, C 3 –C 5 cycloalkyl, C 1 –C 4 haloalkyl, C 2 –C 4 alkoxyalkyl, C 1 –C 4 alkoxy or C 1 –C 4 alkylthio.
- Embodiment 5b A compound of Embodiment 5a wherein each R 1b is independently H, C 1 –C 4 alkyl or C 3 –C 5 cycloalkyl.
- Embodiment 5c A compound of Embodiment 5b wherein each R 1b is independently H, C 1 –C 4 alkyl or C 3 –C 4 cycloalkyl.
- Embodiment 5d A compound of Embodiment 5c wherein R 1b is H when A is A-1, A-2, A-3 and A-5, and m is 1.
- Embodiment 5dd A compound of Embodiment 5d wherein R 1b is H when A is A-1.
- Embodiment 5e A compound of Embodiment 5b wherein R 1b is C 1 –C 4 alkyl or C 3 –C 5 cycloalkyl when A is A-4, A-7, A-8, A-9 or A-10 and m is 1.
- Embodiment 5f A compound of Embodiment 5c wherein R 1b is H.
- Embodiment 6. A compound of Formula 1 or Embodiment 1 wherein m is 0, 1, or 2.
- Embodiment 6a A compound of Embodiment 6 wherein m is 0.
- Embodiment 6b A compound of Embodiment 6 wherein m is 1.
- Embodiment 6c A compound of Embodiment 6 wherein m is 2.
- Embodiment 7. A compound of Formula 1 or Embodiment 1 wherein R 1c is H, C 1 –C 7 alkyl, C 3 –C 7 cycloalkyl, C 2 –C 6 oxacycloalkyl or C 1 –C 7 haloalkyl.
- Embodiment 7a A compound of Formula 1 or Embodiment 1 wherein R 1c is H, C 1 –C 7 alkyl, C 3 –C 7 cycloalkyl, C 2 –C 6 oxacycloalkyl or C 1 –C 7 haloalkyl.
- Embodiment 7 wherein R 1c is H, C 1 –C 4 alkyl, C 3 –C 5 cycloalkyl, C 2 –C 6 oxacycloalkyl or C 1 –C 7 haloalkyl.
- Embodiment 7b A compound of Embodiment 7a wherein R 1c is H, C 1 –C 4 alkyl or C 3 – C 5 cycloalkyl.
- Embodiment 7c A compound of Embodiment 7a wherein R 1c is H, Me, c-Pr, i-Pr, c-Bu and t-Bu.
- Embodiment 7d A compound of Embodiment 7b wherein R 1c is Me.
- Embodiment 7e A compound of Embodiment 7b wherein R 1c is i-Pr.
- Embodiment 7f A compound of Embodiment 7b wherein R 1c is c-Pr.
- Embodiment 7g A compound of Embodiment 7b wherein R 1c is H.
- Embodiment 7h A compound of Embodiment 7b wherein R 1c is c-Bu.
- Embodiment 7i A compound of Embodiment 7b wherein R 1c is t-Bu.
- each R 2 is independently H, halogen, cyano, nitro, hydroxy, C 1 –C 5 alkyl, C 2 –C 5 alkenyl, C 2 –C 5 alkynyl, C 2 –C 5 alkenyloxy, C 2 –C 5 alkynyloxy, C 3 –C 7 cycloalkoxy, C 4 – C 7 cycloalkoxyalkyl, C 3 –C 6 cycloalkyl, C 4 –C 7 cycloalkylalkyl, C 1 –C 5 haloalkyl, C 2 –C 5 haloalkenyl, C 2 –C 5 haloalkynyl, C 2 –C 5 alkoxyalkyl, C 2 –C 5 haloalkoxyalkyl, C 1 –C 5 alkoxy, C 1 –C 5 haloalkoxy, C 1 –C 5 alkylthio
- Embodiment 9a A compound of Embodiment 9 wherein each R 2 is independently H, halogen, cyano, nitro, hydroxy, C 1 –C 5 alkyl, C 2 –C 5 alkenyl, C 2 –C 5 alkynyl, C 2 – C 5 alkenyloxy, C 2 –C 5 alkynyloxy, C 3 –C 7 cycloalkoxy, C 4 –C 7 cycloalkoxyalkyl, C 3 –C 6 cycloalkyl, C 4 –C 7 cycloalkylalkyl, C 1 –C 5 haloalkyl, C 2 –C 5 haloalkenyl, C 2 –C 5 haloalkynyl, C 2 –C 5 alkoxyalkyl, C 2 –C 5 haloalkoxyalkyl, C 1 –C 5 alkoxy, C 1 –C 5 haloalkoxy, C 1 –C 5 al
- Embodiment 9b A compound of Embodiment 9a wherein each R 2 is independently H, halogen, cyano, OH, C 1 –C 5 alkyl, C 2 –C 5 alkenyl, C 2 –C 5 alkynyl, C 2 –C 5 alkenyloxy, C 2 –C 5 alkynyloxy, C 3 –C 7 cycloalkoxy, C 4 –C 7 cycloalkoxyalkyl, C 3 –C 6 cycloalkyl, C 4 –C 7 cycloalkylalkyl, C 1 –C 5 haloalkyl, C 2 –C 5 haloalkenyl, C 2 –C 5 haloalkynyl, C 2 –C 5 alkoxyalkyl, C 2 –C 5 haloalkoxyalkyl, C 1 –C 5 alkoxy, C 1 –C 5 haloalkoxy.
- Embodiment 9bb A compound of Embodiment 9a wherein each R 2 is independently H, halogen, cyano, C 1 –C 5 alkyl, C 2 –C 5 alkenyl, C 2 –C 5 alkynyl, C 2 –C 5 alkenyloxy, C 2 –C 5 alkynyloxy, C 3 –C 7 cycloalkoxy, C 4 –C 7 cycloalkoxyalkyl, C 3 –C 6 cycloalkyl, C 4 –C 7 cycloalkylalkyl, C 1 –C 5 haloalkyl, C 2 –C 5 haloalkenyl, C 2 –C 5 haloalkynyl, C 2 –C 5 alkoxyalkyl, C 2 –C 5 haloalkoxyalkyl, C 1 –C 5 alkoxy, C 1 –C 5 haloalkoxy.
- Embodiment 9c A compound of Embodiment 9b wherein each R 2 is independently H, halogen, cyano, OH, C 1 –C 5 alkyl, C 2 –C 5 alkenyl, C 2 –C 5 alkynyl, C 3 –C 6 cycloalkyl, C 4 –C 7 cycloalkylalkyl, C 1 –C 5 haloalkyl, C 2 –C 5 alkoxyalkyl, C 1 –C 5 alkoxy, C 1 –C 5 haloalkoxy.
- Embodiment 9cc A compound of Embodiment 9b wherein each R 2 is independently H, halogen, cyano, OH, C 1 –C 5 alkyl, C 2 –C 5 alkenyl, C 2 –C 5 alkynyl, C 3 –C 6 cycloalkyl, C 4 –C 7 cycloalkylalkyl, C 1 –C 5 hal
- each R 2 is independently H, halogen, cyano, C 1 –C 5 alkyl, C 2 –C 5 alkenyl, C 2 –C 5 alkynyl, C 3 –C 6 cycloalkyl, C 4 –C 7 cycloalkylalkyl, C 1 –C 5 haloalkyl, C 2 –C 5 alkoxyalkyl, C 1 –C 5 alkoxy, C 1 –C 5 haloalkoxy.
- Embodiment 9d Embodiment 9d.
- each R 2 is independently H, halogen, cyano, OH, C 1 –C 5 alkyl, C 2 –C 5 alkenyl, C 2 –C 5 alkynyl, C 3 –C 6 cycloalkyl, C 1 –C 5 haloalkyl, C 1 –C 5 alkoxy or C 1 –C 5 haloalkoxy.
- Embodiment 9dd is independently H, halogen, cyano, OH, C 1 –C 5 alkyl, C 2 –C 5 alkenyl, C 2 –C 5 alkynyl, C 3 –C 6 cycloalkyl, C 1 –C 5 haloalkyl, C 1 –C 5 alkoxy or C 1 –C 5 haloalkoxy.
- each R 2 is independently H, halogen, cyano, C 1 –C 5 alkyl, C 2 –C 5 alkenyl, C 2 –C 5 alkynyl, C 3 –C 6 cycloalkyl, C 1 –C 5 haloalkyl, C 1 –C 5 alkoxy or C 1 –C 5 haloalkoxy.
- Embodiment 9dd 1.
- each R 2 is independently H, OEt, propargyl, allyl, c-Pr, F, Cl, Br, CN, Me, Et, OMe, C F 3 , OCF 3 or CH 2 CF 3 .
- Embodiment 9c wherein each R 2 is independently H, C 1 –C 5 alkyl or C 1 –C 5 alkoxy.
- Embodiment 9e A compound of Embodiment 9d wherein each R 2 is independently H, Me, OMe or Et.
- Embodiment 9ee A compound of Embodiment 9d wherein each R 2 is independently H, Me or OMe.
- Embodiment 9f A compound of Embodiment 9e wherein R 2 is H.
- Embodiment 9ff A compound of Embodiment 9f wherein R 2 is H when X 4 is CR 2 .
- Embodiment 9g A compound of Embodiment 9e wherein R 2 is Me. Embodiment 9gg.
- Embodiment 9h. A compound of Embodiment 9e wherein R 2 is Et.
- Embodiment 9i. A compound of Embodiment 9 wherein two adjacent R 2 may be taken together to form a saturated or unsaturated 5- to 8-membered ring, containing carbon atoms and optionally 1 to 3 oxygen, sulfur or nitrogen atoms as ring members, one or two carbon or sulfur ring members of the heterocycle can optionally be in the oxidized form of a carbonyl, sulfonyl, sulfinyl moiety, said ring unsubstituted or substituted with at least one substituent independently selected from the group consisting of halogen, cyano, nitro, C 1 –C 4 alkyl, C 3 –C 6 cycloalkyl, C 1 –C 4 haloalkyl, C 1 –C 4 alkoxy and C 1 –C 4 haloal
- Embodiment 9j A compound of Embodiment 9i wherein two adjacent R 2 may be taken together to form a 5- or 6- membered ring, containing up to 2 oxygen atoms as the ring members.
- Embodiment 9k A compound of Embodiment 9j wherein two adjacent R 2 may be taken together to form a 5- or 6- membered ring and the said ring contains a group of - OCH 2 O- or -O(CH 2 ) 2 O- or -O(CH 2 ) 2 -.
- Embodiment 9kk A compound of Embodiment 9i wherein two adjacent R 2 may be taken together to form a 5- or 6- membered ring and the said ring contains a group of - OCH 2 O- or -O(CH 2 ) 2 O- or -O(CH 2 ) 2 -.
- a compound of Embodiment 9j wherein two adjacent R 2 may be taken together to form a 5- or 6- membered ring, wherein said ring, in addition to the two connecting carbon atoms, contains ring members selected from the group consisting of -OCH 2 O-, -O(CH 2 ) 2 O-, and -O(CH 2 ) 2 -.
- Embodiment 10. A compound of Formula 1 or Embodiment 1 wherein Y is O or S.
- Embodiment 10a. A compound of Embodiment 10 wherein Y is O.
- Embodiment 11 A compound of Embodiment 9j wherein two adjacent R 2 may be taken together to form a 5- or 6- membered ring, wherein said ring, in addition to the two connecting carbon atoms, contains ring members selected from the group consisting of -OCH 2 O-, -O(CH 2 ) 2 O-
- R 3a is halogen, cyano, nitro, C 1 –C 7 alkyl, C 2 –C 6 alkenyl, C 2 –C 6 alkynyl, C 3 –C 7 cycloalkyl, C 1 –C 7 alkoxy, C 1 –C 7 haloalkoxy, C 4 –C 7 cycloalkylalkyl, C 4 –C 7 alkylcycloalkyl, C 1 –C 7 haloalkyl, C 2 –C 6 haloalkenyl, C 2 –C 6 haloalkynyl, C 3 – C 7 halocycloalkyl, C 4 –C 7 haloalkylcycloalkyl, C 4 –C 7 cycloalkylhaloalkyl, C 2 – C 7 alkoxyalkyl, C 2 –C 7 haloalkoxyalkyl, C 2 –C
- Embodiment 11a A compound of Embodiment 11 wherein R 3a is halogen, C 1 –C 7 alkyl, C 2 –C 6 alkenyl, C 2 –C 6 alkynyl, C 3 –C 7 cycloalkyl, C 1 –C 7 alkoxy, C 4 –C 7 cycloalkylalkyl, C 4 –C 7 alkylcycloalkyl, C 1 –C 7 haloalkyl, C 2 –C 6 haloalkenyl, C 2 –C 6 haloalkynyl, C 3 –C 7 halocycloalkyl, C 4 –C 7 haloalkylcycloalkyl, C 2 –C 7 alkoxyalkyl or C 2 –C 7 haloalkoxyalkyl.
- R 3a is halogen, C 1 –C 7 alkyl, C 2 –C 6 alkenyl, C 2 –C 6 alky
- Embodiment 11b A compound of Embodiment 11a wherein R 3a is halogen, C 1 –C 7 alkyl, C 2 –C 6 alkenyl, C 2 –C 6 alkynyl, C 3 –C 7 cycloalkyl, C 1 –C 7 alkoxy, C 4 –C 7 cycloalkylalkyl, C 4 –C 7 alkylcycloalkyl, C 1 –C 7 haloalkyl, C 3 –C 7 halocycloalkyl, C 2 –C 7 alkoxyalkyl or C 2 –C 7 haloalkoxyalkyl.
- Embodiment 11c Embodiment 11c.
- Embodiment 11d wherein R 3a is halogen, Me, c-Pr, CF 3 , CH 2 OMe, CH 2 OEt.
- Embodiment 11f A compound of Embodiment 11e wherein R 3a is Me.
- Embodiment 11ff A compound of Embodiment 11e wherein R 3a is Et.
- Embodiment 11g A compound of Embodiment 11d wherein R 3a is halogen.
- Embodiment 11h A compound of Embodiment 11g wherein R 3a is F or Cl.
- Embodiment 11i A compound of Embodiment 11d wherein R 3a is CH 2 OMe. Embodiment 12.
- Embodiment 12a A compound of Embodiment 12 wherein R 3b is H.
- Embodiment 12b A compound of Embodiment 12 wherein R 3b is halogen.
- Embodiment 12c A compound of Formula 1 or Embodiment 1 wherein each R 3b is independently H or halogen.
- Embodiment 12d Embodiment 12d.
- Embodiment 13 wherein Z is O, C(R 3c ) p or NR 3d .
- Embodiment 13b A compound of Embodiment 13a wherein Z is C(R 3c ) p or NR 3d .
- Embodiment 13c A compound of Embodiment 13b wherein Z is C(R 3c ) p .
- Embodiment 13d A compound of Embodiment 13a wherein Z is NR 3d .
- Embodiment 13e A compound of Embodiment 13a wherein Z is O.
- Embodiment 13f A compound of Embodiment 13e wherein Z is O, and both X 1 and X 3 are N.
- Embodiment 13g A compound of Embodiment 13g.
- Embodiment 13e wherein Z is O, and both X 2 and X 3 are N.
- Embodiment 13h A compound of Embodiment 13e wherein Z is O, and both X 1 and X 4 are N.
- Embodiment 13i A compound of Embodiment 13e wherein Z is O, and both X 2 and X 4 are N.
- Embodiment 13j A compound of Embodiment 13e wherein Z is O, and both X 3 and X 4 are N.
- each R 3c is independently H, halogen, cyano, nitro, hydroxy, C 1 –C 5 alkyl, C 2 –C 5 alkenyl, C 2 –C 5 alkynyl, C 3 –C 5 cycloalkyl, C 1 –C 5 haloalkyl, C 2 –C 5 haloalkenyl, C 2 –C 5 haloalkynyl, C 2 –C 5 alkoxyalkyl, C 2 –C 5 haloalkoxyalkyl, C 1 –C 5 alkoxy, C 1 –C 5 haloalkoxy, C 1 –C 5 alkylthio, C 1 –C 4 alkylsulfinyl, C 1 –C 4 alkylsulfonyl, C 1 –C 5 haloalkylthio, C 1 –C 4 haloalkylsulfinyl, C 1
- Embodiment 14a A compound of Embodiment 14 wherein each R 3c is independently H, halogen, cyano, nitro, hydroxy, C 1 –C 5 alkyl, C 2 –C 5 alkenyl, C 2 –C 5 alkynyl, C 3 –C 5 cycloalkyl, C 1 –C 5 haloalkyl, C 2 –C 5 haloalkenyl, C 2 –C 5 haloalkynyl, C 2 – C 5 alkoxyalkyl, C 2 –C 5 haloalkoxyalkyl, C 1 –C 5 alkoxy, C 1 –C 5 haloalkoxy, C 1 – C 5 alkylthio, C 1 –C 4 alkylsulfinyl, C 1 –C 4 alkylsulfonyl, C 1 –C 5 haloalkylthio, C 1 –C 4 haloalkylsulfiny
- Embodiment 14b A compound of Embodiment 14a wherein each R 3c is independently H, halogen, hydroxy, C 1 –C 5 alkyl, C 3 –C 5 cycloalkyl, C 1 –C 5 haloalkyl, C 2 –C 5 alkoxyalkyl, C 2 –C 5 haloalkoxyalkyl, C 1 –C 5 alkoxy, C 1 –C 5 haloalkoxy, C 4 –C 7 cycloalkylalkyl, C 3 –C 7 cycloalkoxy, C 3 –C 6 alkylcarbonylalkyl, C 3 –C 6 alkoxycarbonylalkyl or C 2 –C 5 alkoxycarbonyl, C 2 –C 6 alkylcarbonyloxy.
- Embodiment 14c A compound of Embodiment 14b wherein each R 3c is independently H, halogen, hydroxy, C 1 –C 5 alkyl, C 3 –C 5 cycloalkyl, C 2 –C 5 alkoxyalkyl, C 1 –C 5 alkoxy, C 4 –C 7 cycloalkylalkyl, C 3 –C 7 cycloalkoxy, C 3 –C 6 alkylcarbonylalkyl, C 3 –C 6 alkoxycarbonylalkyl or C 2 –C 5 alkoxycarbonyl, C 2 –C 6 alkylcarbonyloxy.
- Embodiment 14d Embodiment 14d.
- Embodiment 14f A compound of Embodiment 14e wherein each R 3c is independently H.
- a compound of Formula 1 or Embodiment 1 wherein R 3d is H, C 1 –C 4 alkyl, C 3 –C 6 cycloalkyl, C 1 –C 4 haloalkyl, C 2 –C 5 alkylcarbonyl, C 2 –C 5 haloalkylcarbonyl, C 2 –C 5 alkoxycarbonyl, C 2 –C 5 alkylaminocarbonyl, C 2 –C 5 dialkylaminocarbonyl, C 1 –C 5 alkylsulfonyl or C 1 –C 5 haloalkylsulfonyl; Embodiment 15a.
- a compound of Embodiment 15 wherein R 3d is H, C 1 –C 4 alkyl, C 2 – C 5 alkylcarbonyl, C 2 –C 5 haloalkylcarbonyl, C 2 –C 5 alkoxycarbonyl, C 2 –C 5 alkylaminocarbonyl, C 2 –C 5 dialkylaminocarbonyl, C 1 –C 5 alkylsulfonyl or C 1 – C 5 haloalkylsulfonyl.
- Embodiment 15b A compound of Embodiment 15a wherein R 3d is H or C 1 –C 4 alkyl.
- Embodiment 15c is H, C 1 –C 4 alkyl.
- Embodiment 15b wherein R 3d is H or Me.
- Embodiment 15d A compound of Embodiment 15c wherein R 3d is H.
- Embodiment 15e A compound of Embodiment 15c wherein R 3d is Me.
- Embodiment 16 A compound of Formula 1 or Embodiment 1 wherein p is 0 or 1.
- Embodiment 16a A compound of Embodiment 16 wherein p is 0. Embodiment 16b.
- Embodiment 16 wherein p is 1.
- Embodiment 17 A compound of Formula 1 or Embodiment 1 wherein R 4a is H, halogen, cyano, nitro, C 1 –C 5 alkyl, C 2 –C 5 alkenyl, C 2 –C 5 alkynyl, C 3 –C 5 cycloalkyl, C 1 –C 5 haloalkyl, C 2 –C 5 haloalkenyl, C 2 –C 5 haloalkynyl, C 2 –C 5 alkoxyalkyl, C 2 –C 5 haloalkoxyalkyl, C 1 –C 5 alkoxy, C 1 –C 5 haloalkoxy, C 3 –C 6 trialkylsilyl, C 1 –C 5 alkylthio, C 1 –C 5 haloalkylthio or C 2 –C 5 alkoxycarbonyl.
- Embodiment 17b A compound of Embodiment 17 wherein R 4a is H, halogen, C 1 –C 5 alkyl, C 1 –C 5 haloalkyl.
- Embodiment 17c A compound of Embodiment 17b wherein R 4a is H, halogen, C 1 –C 4 alkyl, C 1 –C 4 haloalkyl.
- Embodiment 17d A compound of Embodiment 17c wherein R 4a is H, Cl, Me, F, CF 3 .
- Embodiment 17e A compound of Embodiment 17d wherein R 4a is H, Cl or Me.
- Embodiment 17f A compound of Embodiment 17d wherein R 4a is H.
- Embodiment 17g A compound of Embodiment 17d wherein R 4a is Cl.
- Embodiment 17h A compound of Embodiment 17d wherein R 4a is Me.
- Embodiment 17i A compound of Embodiment 17d wherein R 4a is CF 3 .
- Embodiment 17j A compound of Embodiment 17d wherein R 4a is F.
- Embodiment 18. A compound of Formula 1 or Embodiment 1 wherein q is 0 or 1.
- Embodiment 18a A compound of Embodiment 18 wherein q is 0.
- Embodiment 18b A compound of Embodiment 18 wherein q is 1.
- Embodiment 19 A compound of Embodiment 19
- Embodiments of this invention, including Embodiments 1–20 above as well as any other embodiments described herein, can be combined in any manner, and the descriptions of variables in the embodiments pertain not only to the compounds of Formula 1 but also to the starting compounds and intermediate compounds useful for preparing the compounds of Formula 1.
- Embodiment X A compound of Formula 1 as described in the Summary of the Disclosure wherein A is selected from , ,
- Embodiment A1 The compound of Embodiment X wherein Z is C(R 3c ) p or NR 3d .
- Embodiment A2. The compound of Embodiment A1 wherein A is A-1, A-2, A-3, A-5, A-7, A-8 or A-10; X 1 , X 2 , X 3 and X 4 are each independently CR 2 ; or X 1 is N, and X 2 , X 3 and X 4 are each independently N or CR 2 ; or X 1 and X 2 are N, and X 3 and X 4 are each independently CR 2 ; or X 2 and X 3 are N, and X 1 and X 4 are each independently CR 2 ; or X 1 and X 3 are N, and X 2 and X 4 are each independently CR 2 ; or X 1 and X 3 are N, and X 2 and X 4 are each independently CR 2 ; or X 2 is N, and X 1 ,
- R 1a is H, C 1 –C 7 alkyl, C 2 –C 6 alkenyl, C 2 –C 6 alkynyl, C 3 –C 7 cycloalkyl, C 4 –C 7 cycloalkylalkyl, C 4 –C 7 alkylcycloalkyl, C 1 –C 7 haloalkyl, C 1 –C 6 hydroxyalkyl, C 3 –C 7 hydroxycycloalkyl, C 1 –C 6 hydroxyhaloalkyl, C 4 –C 8 alkoxycycloalkyl, C 2 –C 7 alkoxyhaloalkyl, C 2 –C 6 oxacycloalkyl, C 3 –C 7 oxacycloalkylalkyl, C 3 –C 7 alkyloxacycloalkyl, C 2 –C 6 thiacycloalkyl, C 3 – C 7 thiacycloalkylalkyl, C 2 –
- R 3a is halogen, C 1 –C 7 alkyl, C 2 –C 6 alkenyl, C 2 –C 6 alkynyl, C 3 –C 7 cycloalkyl, C 1 –C 7 alkoxy, C 4 –C 7 cycloalkylalkyl, C 4 –C 7 alkylcycloalkyl, C 1 –C 7 haloalkyl, C 2 –C 6 haloalkenyl, C 2 –C 6 haloalkynyl, C 3 –C 7 halocycloalkyl, C 4 –C 7 haloalkylcycloalkyl, C 2 –C 7 alkoxyalkyl or C 2 –C 7 haloalkoxyalkyl; each R 3c is independently H, halogen, hydroxy, C 1 –C 5 alkyl, C 1 –C 5 alkoxy, C 3 – C 6 alkylcarbonylalkyl, C 3 –
- Embodiment A3 The compound of Embodiment A2 wherein A is A-1, A-2 or A-8; R 1a is H, C 1 –C 7 alkyl, C 3 –C 7 cycloalkyl, C 4 –C 7 cycloalkylalkyl, C 4 –C 7 alkylcycloalkyl, C 1 –C 7 haloalkyl, C 1 –C 6 hydroxyalkyl, C 4 –C 8 alkoxycycloalkyl, C 2 –C 6 oxacycloalkyl, C 3 –C 7 alkyloxacycloalkyl, C 2 –C 7 alkoxyalkyl, C 1 –C 7 alkoxy, C 1 –C 7 alkylthio, C 2 –C 7 alkylthioalkyl, C 2 –C 7 alkylsulfinylalkyl or C 2 –C 7 alkylsulfonylalkyl; each R 1b is independently
- Embodiment A4 The compound of Embodiment A3 wherein A is A-1; X 1 , X 2 , X 3 and X 4 are CR 2 ; R 1a is C 1 –C 5 alkyl, C 3 –C 5 cycloalkyl, C 1 –C 5 haloalkyl, C 1 –C 6 hydroxyalkyl, C 2 –C 5 oxacycloalkyl or C 2 –C 7 alkylsulfonylalkyl; each R 2 is independently H, halogen, cyano, OH, C 1 –C 5 alkyl, C 2 –C 5 alkenyl, C 2 –C 5 alkynyl, C 3 –C 6 cycloalkyl, C 1 –C 5 haloalkyl, C 1 –C 5 alkoxy or C 1 – C 5 haloalkoxy; or two adjacent R 2 may be taken together to form a 5- or 6- membered ring and
- Embodiment A5 The compound of Embodiment A4 wherein R 1a is Et, i-Pr, t-Bu, c-Bu, CFMe 2 , CF 2 Me, oxetan-3-yl, CH 2 SO 2 Me or CMe 2 OH; R 1b is H; R 1c is H, Me, c-Pr or i-Pr; each R 2 is independently H, OH, OEt, propargyl, allyl, c-Pr, F, Cl, Br, CN, Me, E t, OMe, CF 3 , OCF 3 or CH 2 CF 3 ; Y is O; R 3a is halogen, Me, c-Pr, CF 3 , CH 2 OMe, or CH 2 OEt; R 3b is H; R 3d is H or Me; and R 4a is H, Cl or Me.
- Embodiment A6 The compound of any one of the preceding Embodiments wherein Z is C(R 3c ) p .
- Embodiment B1. The compound of Embodiment X wherein Z is O.
- Embodiment B2. The compound of Embodiment B1 wherein X 2 and X 3 are N, and X 1 and X 4 are each independently CR 2 ; or X 1 and X 3 are N, and X 2 and X 4 are each independently CR 2 .
- A is A-1, A-2, A-3, A-5, A-7, A-8 or A-10;
- R 1a is H, C 1 –C 7 alkyl, C 2 –C 6 alkenyl, C 2 –C 6 alkynyl, C 3 –C 7 cycloalkyl, C 4 –C 7 cycloalkylalkyl, C 4 –C 7 alkylcycloalkyl, C 1 –C 7 haloalkyl, C 1 –C 6 hydroxyalkyl, C 3 –C 7 hydroxycycloalkyl, C 1 –C 6 hydroxyhaloalkyl, C 4 –C 8 alkoxycycloalkyl, C 2 –C 7 alkoxyhaloalkyl, C 2 –C 6 oxacycloalkyl, C 3 –C 7 oxacycloalkylalkyl, C 3 –C 7 alkyloxacycloalkyl, C 2 –
- Embodiment B4 The compound of Embodiment B3 wherein A is A-1, A-2 or A-8; R 1a is H, C 1 –C 7 alkyl, C 3 –C 7 cycloalkyl, C 4 –C 7 cycloalkylalkyl, C 4 –C 7 alkylcycloalkyl, C 1 –C 7 haloalkyl, C 1 –C 6 hydroxyalkyl, C 4 –C 8 alkoxycycloalkyl, C 2 –C 6 oxacycloalkyl, C 3 –C 7 alkyloxacycloalkyl, C 2 –C 7 alkoxyalkyl, C 1 –C 7 alkoxy, C 1 –C 7 alkylthio, C 2 –C 7 alkylthioalkyl, C 2 –C 7 alkylsulfinylalkyl or C 2 –C 7 alkylsulfonylalkyl; each R 1b is independently
- Embodiment B5. The compound of Embodiment B4 wherein A is A-1; X 1 and X 3 are N, and X 2 and X 4 are independently CR 2 ; R 1a is C 1 –C 5 alkyl, C 3 –C 5 cycloalkyl, C 1 –C 5 haloalkyl, C 1 –C 6 hydroxyalkyl, C 2 –C 5 oxacycloalkyl or C 2 –C 7 alkylsulfonylalkyl; each R 2 is independently H, halogen, cyano, OH, C 1 –C 5 alkyl, C 2 –C 5 alkenyl, C 2 –C 5 alkynyl, C 3 –C 6 cycloalkyl, C 1 –C 5 haloalkyl, C 1 –C 5 alkoxy or C 1 – C 5 haloalkoxy; or two adjacent R 2 may be taken together to form a 5- or 6- membered
- Embodiment B6 The compound of Embodiment B5 wherein R 1a is Et, i-Pr, t-Bu, c-Bu, CFMe 2 , CF 2 Me, oxetan-3-yl, CH 2 SO 2 Me or CMe 2 OH; R 1b is H; R 1c is H, Me, c-Pr or i-Pr; each R 2 is independently H, OH, OEt, propargyl, allyl, c-Pr, F, Cl, Br, CN, Me, E t, OMe, CF 3 , OCF 3 or CH 2 CF 3 ; Y is O; R 3a is halogen, Me, c-Pr, CF 3 , CH 2 OMe, or CH 2 OEt; R 3b is H; R 3d is H or Me; and R 4a is H or Me.
- R 1a is Et, i-Pr, t-Bu, c-Bu, CFMe 2
- Embodiment B7 The compound of Embodiment B4 wherein A is A-1; X 2 and X 3 are N, and X 1 and X 4 are independently CR 2 ; R 1a is C 1 –C 5 alkyl, C 3 –C 5 cycloalkyl, C 1 –C 5 haloalkyl, C 1 –C 6 hydroxyalkyl, C 2 –C 5 oxacycloalkyl or C 2 –C 7 alkylsulfonylalkyl; each R 2 is independently H, halogen, cyano, OH, C 1 –C 5 alkyl, C 2 –C 5 alkenyl, C 2 –C 5 alkynyl, C 3 –C 6 cycloalkyl, C 1 –C 5 haloalkyl, C 1 –C 5 alkoxy or C 1 – C 5 haloalkoxy; or two adjacent R 2 may be taken together to form a 5- or 6- membered
- Embodiment B8 The compound of Embodiment B7 wherein R 1a is Et, i-Pr, t-Bu, c-Bu, CFMe 2 , CF 2 Me, oxetan-3-yl, CH 2 SO 2 Me or CMe 2 OH; R 1b is H; R 1c is H, Me, c-Pr or i-Pr; each R 2 is independently H, OH, OEt, propargyl, allyl, c-Pr, F, Cl, Br, CN, Me, E t, OMe, CF 3 , OCF 3 or CH 2 CF 3 ; Y is O; R 3a is halogen, Me, c-Pr, CF 3 , CH 2 OMe, or CH 2 OEt; R 3b is H; R 3d is H or Me; and R 4a is H or Me.
- R 1a is Et, i-Pr, t-Bu, c-Bu, CFMe 2
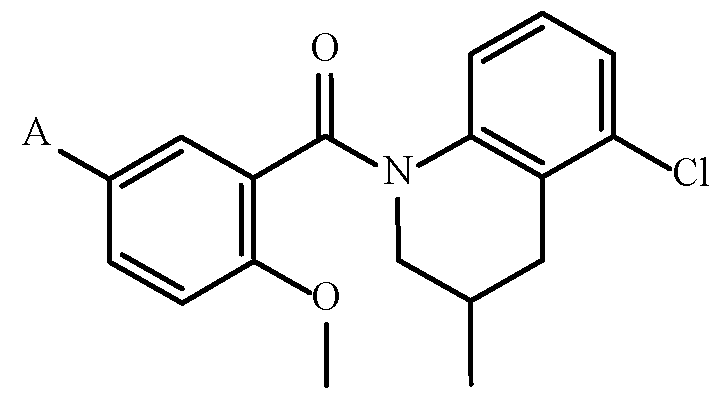
- Embodiment C Specific embodiment including a compound of Formula 1 wherein: A is A-1, R 1a is i-Pr, R 1b is H, X 1 , X 2 and X 4 are all CH, X 3 is COMe, Y is O, R 3a is Me, R 3b is H, Z is CH 2 and R 4a is Me; (Compound 3) Embodiment D.
- Specific embodiments include compounds of Formula 1 selected from the group consisting of: (3,4-dihydro-3,5-dimethyl-1(2H)-quinolinyl)-[2-methoxy-5-[3-(1-methylethyl)- 1H-1,2,4-triazol-1-yl]phenyl]methanone; [(3S)-3,4-dihydro-3,5-dimethyl-1(2H)-quinolinyl][2-methoxy-5-[3-(1- methylethyl)-1H-1,2,4-triazol-1-yl]phenyl]methanone; [(3R)-3,4-dihydro-3,5-dimethyl-1(2H)-quinolinyl][2-methoxy-5-[3-(1- methylethyl)-1H-1,2,4-triazol-1-yl]phenyl]methanone; [5-fluoro-3,4-dihydro-3-(
- This invention also relates to a method for controlling undesired vegetation comprising applying to the locus of the vegetation herbicidally effective amounts of the compounds of the invention (e.g., as a composition described herein).
- the compounds of the invention e.g., as a composition described herein.
- embodiments relating to methods of use are those involving the compounds of embodiments described above.
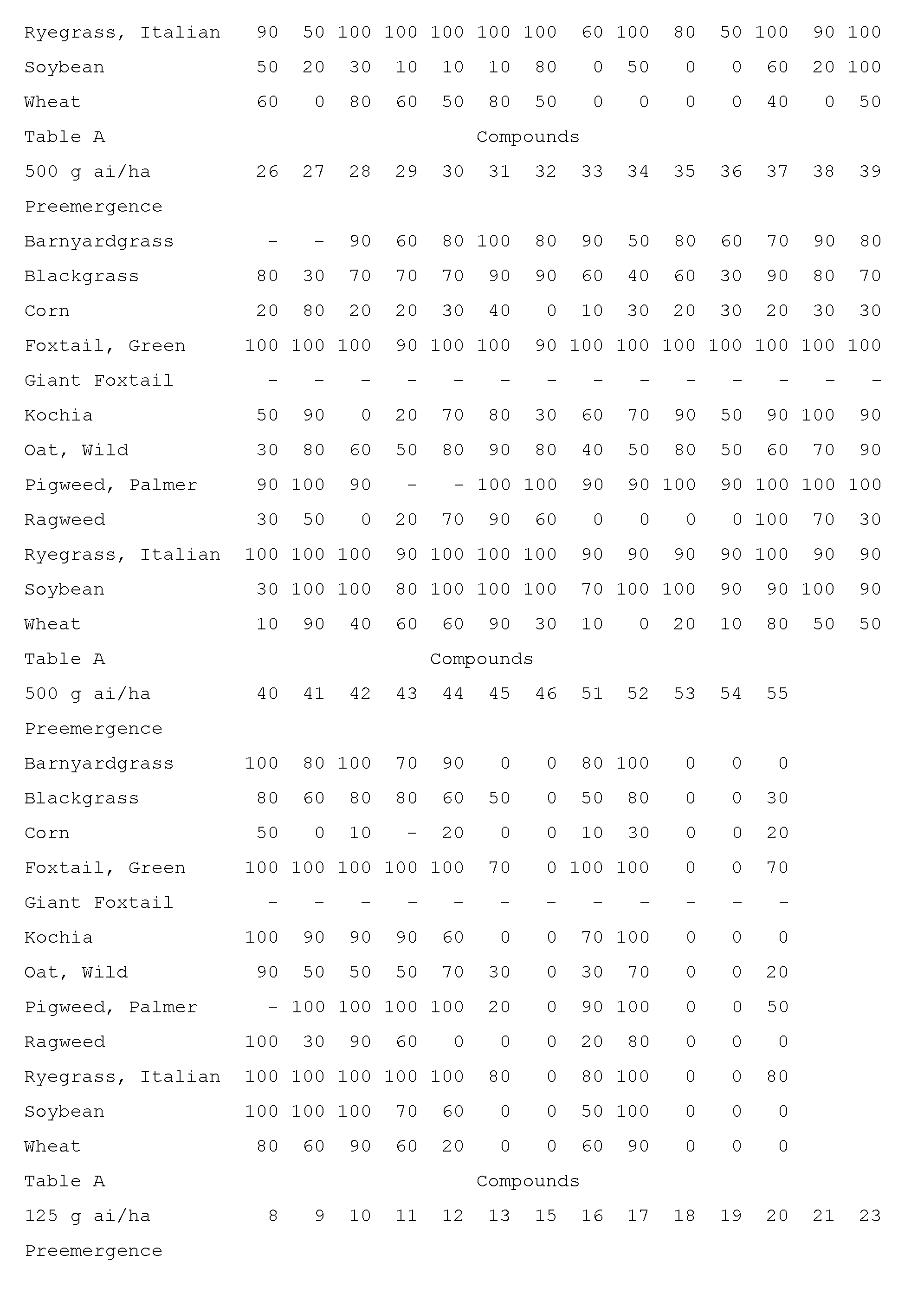
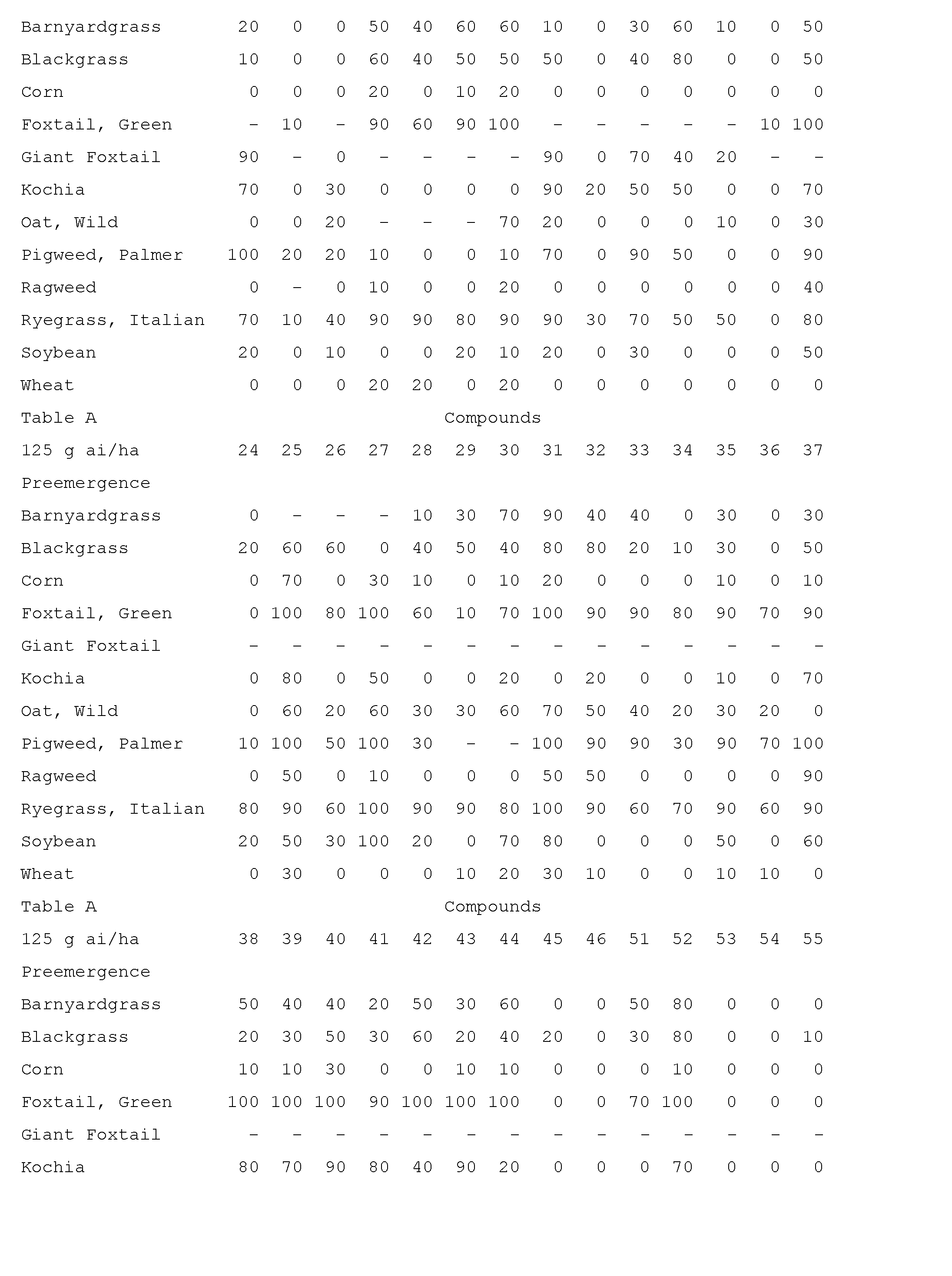
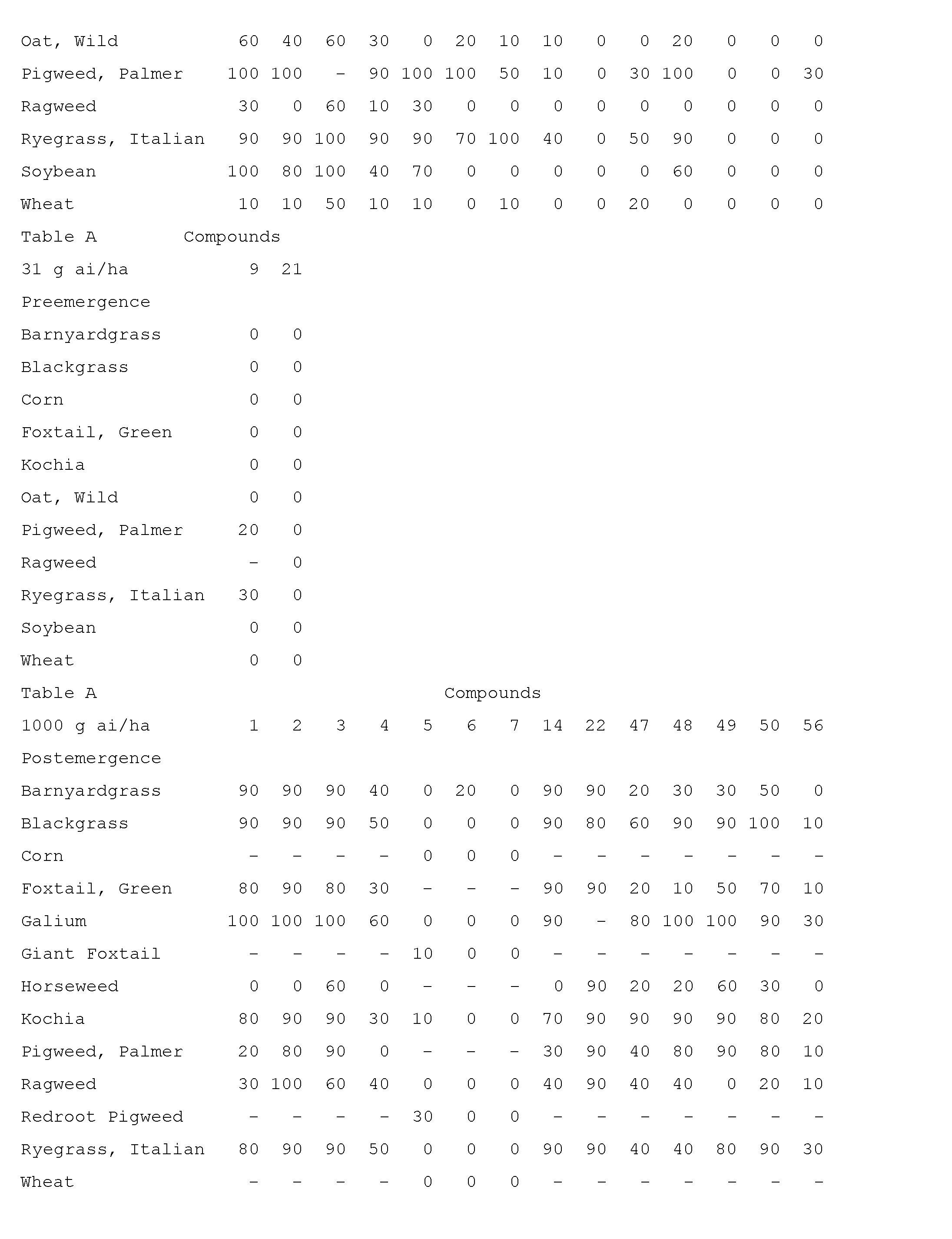
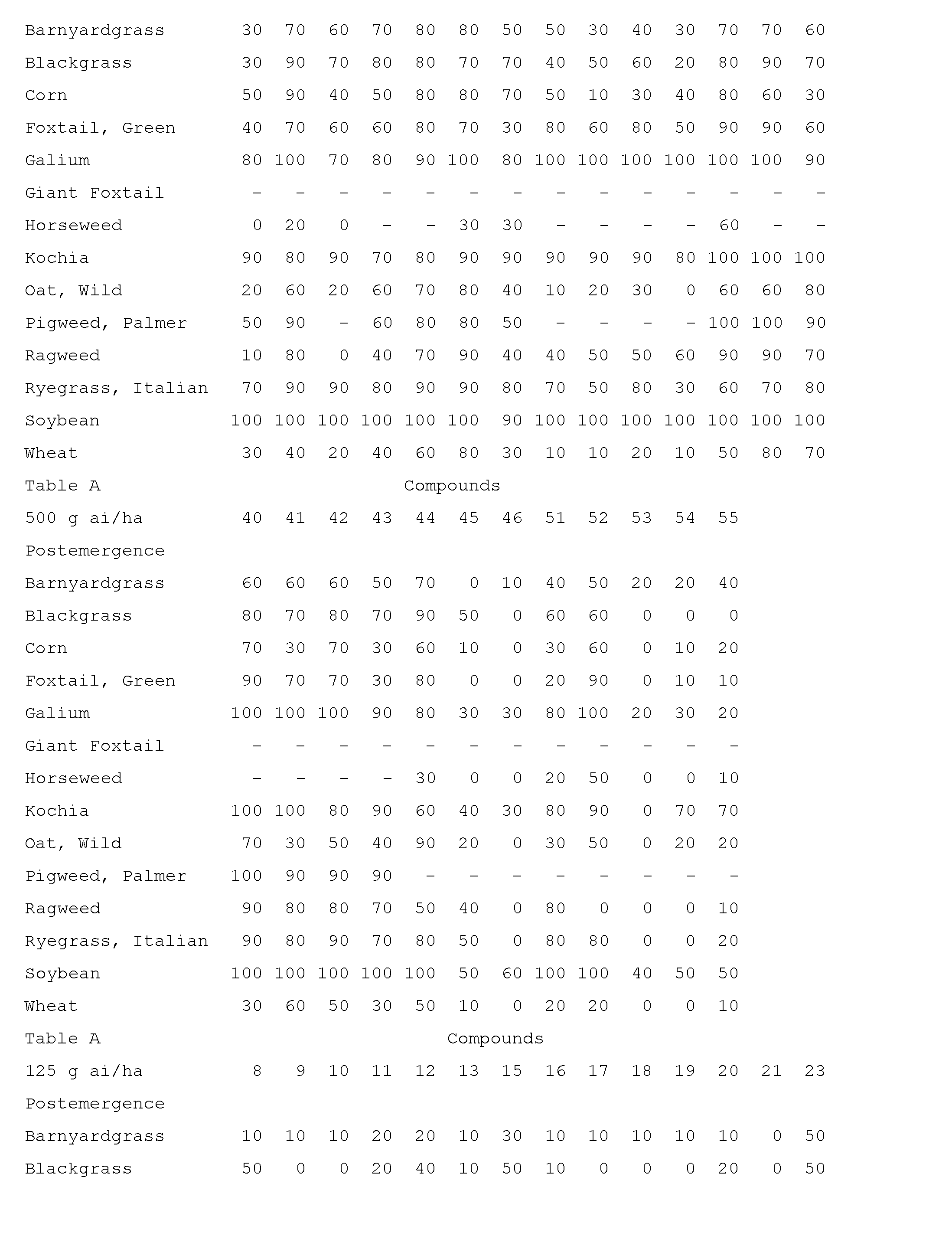
- Compounds of the invention are particularly useful for selective control of weeds in crops such as wheat, barley, maize, soybean, sunflower, cotton, oilseed rape and rice, and specialty crops such as sugarcane, citrus, fruit and nut crops.
- herbicidal compositions of the present invention comprising the compounds of embodiments described above.
- This invention also includes a herbicidal mixture comprising (a) a compound selected from Formula 1, N-oxides, and salts thereof, and (b) at least one additional active ingredient selected from (b1) photosystem II inhibitors, (b2) acetohydroxy acid synthase (AHAS) inhibitors, (b3) acetyl-CoA carboxylase (ACCase) inhibitors, (b4) auxin mimics, (b5) 5-enol- pyruvylshikimate-3-phosphate (EPSP) synthase inhibitors, (b6) photosystem I electron diverters, (b7) protoporphyrinogen oxidase (PPO) inhibitors, (b8) glutamine synthetase (GS) inhibitors, (b9) very long chain fatty acid (VLCFA) elongase inhibitors, (b10) auxin transport inhibitors, (b11) phytoene desaturase (PDS) inhibitors, (b12) 4-hydroxyphenyl-pyruvate dioxygena
- Photosystem II inhibitors are chemical compounds that bind to the D-1 protein at the Q B -binding niche and thus block electron transport from Q A to Q B in the chloroplast thylakoid membranes. The electrons blocked from passing through photosystem II are transferred through a series of reactions to form toxic compounds that disrupt cell membranes and cause chloroplast swelling, membrane leakage, and ultimately cellular destruction.
- the Q B -binding niche has three different binding sites: binding site A binds the triazines such as atrazine, triazinones such as hexazinone, and uracils such as bromacil, binding site B binds the phenylureas such as diuron, and binding site C binds benzothiadiazoles such as bentazon, nitriles such as bromoxynil and phenyl-pyridazines such as pyridate.
- triazines such as atrazine
- triazinones such as hexazinone
- uracils such as bromacil
- binding site B binds the phenylureas such as diuron
- binding site C binds benzothiadiazoles such as bentazon, nitriles such as bromoxynil and phenyl-pyridazines such as pyridate.
- photosystem II inhibitors include ametryn, amicarbazone, atrazine, bentazon, bromacil, bromofenoxim, bromoxynil, chlorbromuron, chloridazon, chlorotoluron, chloroxuron, cumyluron, cyanazine, daimuron, desmedipham, desmetryn, dimefuron, dimethametryn, diuron, ethidimuron, fenuron, fluometuron, hexazinone, ioxynil, isoproturon, isouron, lenacil, linuron, metamitron, methabenzthiazuron, metobromuron, metoxuron, metribuzin, monolinuron, neburon, pentanochlor, phenmedipham, prometon, prometryn, propanil, propazine, pyridafol, pyridate, siduron, simazine, simetryn,
- AHAS inhibitors are chemical compounds that inhibit acetohydroxy acid synthase (AHAS), also known as acetolactate synthase (ALS), and thus kill plants by inhibiting the production of the branched-chain aliphatic amino acids such as valine, leucine and isoleucine, which are required for protein synthesis and cell growth.
- AHAS acetohydroxy acid synthase
- ALS acetolactate synthase
- AHAS inhibitors include amidosulfuron, azimsulfuron, bensulfuron-methyl, bispyribac-sodium, cloransulam-methyl, chlorimuron-ethyl, chlorsulfuron, cinosulfuron, cyclosulfamuron, diclosulam, ethametsulfuron-methyl, ethoxysulfuron, flazasulfuron, florasulam, flucarbazone-sodium, flumetsulam, flupyrsulfuron-methyl, flupyrsulfuron-sodium, foramsulfuron, halosulfuron-methyl, imazamethabenz-methyl, imazamox, imazapic, imazapyr, imazaquin, imazethapyr, imazosulfuron, iodosulfuron-methyl (including sodium salt), iodosulfuron-sodium, iofensulfuron (2-i
- ACCase inhibitors are chemical compounds that inhibit the acetyl-CoA carboxylase enzyme, which is responsible for catalyzing an early step in lipid and fatty acid synthesis in plants. Lipids are essential components of cell membranes, and without them, new cells cannot be produced. The inhibition of acetyl CoA carboxylase and the subsequent lack of lipid production leads to losses in cell membrane integrity, especially in regions of active growth such as meristems. Eventually shoot and rhizome growth ceases, and shoot meristems and rhizome buds begin to die back.
- ACCase inhibitors include alloxydim, butroxydim, clethodim, clodinafop, cycloxydim, cyhalofop, diclofop, fenoxaprop, fluazifop, haloxyfop, metproxybicyclone, pinoxaden, profoxydim, propaquizafop, quizalofop, sethoxydim, tepraloxydim and tralkoxydim, including resolved forms such as fenoxaprop-P, fluazifop-P, haloxyfop-P and quizalofop-P and ester forms such as clodinafop-propargyl, cyhalofop-butyl, diclofop-methyl and fenoxaprop-P-ethyl.
- auxin is a plant hormone that regulates growth in many plant tissues.
- auxin mimics are chemical compounds mimicking the plant growth hormone auxin, thus causing uncontrolled and disorganized growth leading to plant death in susceptible species.
- auxin mimics include aminocyclopyrachlor (6-amino-5-chloro-2-cyclopropyl-4- pyrimidinecarboxylic acid) and its methyl and ethyl esters and its sodium and potassium salts, 4-amino-3-chloro-5-fluoro-6-(7-fluoro-1H-indol-6-yl)-2-pyridinecarboxylic 2-propyn-1-yl ester (CAS No.
- EPSP synthase inhibitors are chemical compounds that inhibit the enzyme, 5-enol-pyruvylshikimate-3-phosphate synthase, which is involved in the synthesis of aromatic amino acids such as tyrosine, tryptophan and phenylalanine.
- EPSP inhibitor herbicides are readily absorbed through plant foliage and translocated in the phloem to the growing points.
- Glyphosate is a relatively nonselective postemergence herbicide that belongs to this group. Glyphosate includes esters and salts such as ammonium, isopropylammonium, potassium, sodium (including sesquisodium) and trimesium (alternatively named sulfosate).
- Photosystem I electron diverters are chemical compounds that accept electrons from Photosystem I, and after several cycles, generate hydroxyl radicals. These radicals are extremely reactive and readily destroy unsaturated lipids, including membrane fatty acids and chlorophyll. This destroys cell membrane integrity, so that cells and organelles “leak”, leading to rapid leaf wilting and desiccation, and eventually to plant death. Examples of this second type of photosynthesis inhibitor include diquat, paraquat and 1-(2-carboxyethyl)-4-(2- pyrimidinyl)pyridazinium (CAS No.2285384-11-2) and salts and esters thereof. Of note is a photosystem I electron diverter selected from diquat and paraquat.
- PPO inhibitors are chemical compounds that inhibit the enzyme protoporphyrinogen oxidase, quickly resulting in formation of highly reactive compounds in plants that rupture cell membranes, causing cell fluids to leak out.
- PPO inhibitors include acifluorfen-sodium, azafenidin, benzfendizone, bifenox, butafenacil, carfentrazone, carfentrazone-ethyl, chlomethoxyfen, 3-[2-chloro-5-[3,6-dihydro-3-methyl-2,6-dioxo-4- (trifluoromethyl)-1(2H)-pyrimidinyl]-4-fluorophenyl]-4,5-dihydro-5-methyl-5-isoxazole- carboxylic ethyl ester (CAS No.
- GS inhibitors are chemical compounds that inhibit the activity of the glutamine synthetase enzyme, which plants use to convert ammonia into glutamine. Consequently, ammonia accumulates and glutamine levels decrease. Plant damage probably occurs due to the combined effects of ammonia toxicity and deficiency of amino acids required for other metabolic processes.
- the GS inhibitors include glufosinate and its esters and salts such as glufosinate-ammonium and other phosphinothricin derivatives, glufosinate-P ((2S)-2-amino- 4-(hydroxymethylphosphinyl)butanoic acid) and bilanaphos.
- VLCFA elongase inhibitors are herbicides having a wide variety of chemical structures, which inhibit the elongase.
- Elongase is one of the enzymes located in or near chloroplasts which are involved in biosynthesis of VLCFAs.
- very-long-chain fatty acids are the main constituents of hydrophobic polymers that prevent desiccation at the leaf surface and provide stability to pollen grains.
- Such herbicides include acetochlor, alachlor, anilofos, butachlor, cafenstrole, dimethachlor, dimethenamid, diphenamid, fenoxasulfone (3- [[(2,5-dichloro-4-ethoxyphenyl)methyl]sulfonyl]-4,5-dihydro-5,5-dimethylisoxazole), fentrazamide, flufenacet, indanofan, mefenacet, metazachlor, metolachlor, naproanilide, napropamide, napropamide-M ((2R)-N,N-diethyl-2-(1-naphthalenyloxy)propanamide), pethoxamid, piperophos, pretilachlor, propachlor, propisochlor, pyroxasulfone, and thenylchlor, including resolved forms such as S-metolachlor and chloroacetamides and oxyace
- auxin transport inhibitors are chemical substances that inhibit auxin transport in plants, such as by binding with an auxin-carrier protein.
- auxin transport inhibitors include diflufenzopyr, naptalam (also known as N-(1-naphthyl)phthalamic acid and 2-[(1-naphthalenylamino)carbonyl]benzoic acid).
- PDS inhibitors are chemical compounds that inhibit carotenoid biosynthesis pathway at the phytoene desaturase step. Examples of PDS inhibitors include beflubutamid, diflufenican, fluridone, flurochloridone, flurtamone norflurzon and picolinafen.
- HPPD inhibitors are chemical substances that inhibit the biosynthesis of synthesis of 4-hydroxyphenyl-pyruvate dioxygenase.
- HPPD inhibitors include benquinotrione, benzobicyclon, benzofenap, bicyclopyrone (4-hydroxy-3-[[2-[(2- methoxyethoxy)methyl]-6-(trifluoromethyl)-3-pyridinyl]carbonyl]bicyclo[3.2.1]oct-3-en-2- one), bipyrazone, cyprafluone, fenpyrazone, fenquinotrione (2-[[8-chloro-3,4-dihydro-4-(4- methoxyphenyl)-3-oxo-2-quinoxalinyl]carbonyl]-1,3-cyclohexanedione), flusulfinam, iptriazopyrid, isoxachlortole, isoxaflutole, lancotrione, meso
- DXP synthase inhibitors “Deoxy-D-Xylulose Phosphate Synthase (DXP synthase) inhibitors” (b13) isoxazolidinone class of herbicides is classified according to its inhibition of the deoxy-d- xylulose phosphate synthase (DXP synthase) which is a component of the carotenoid biosynthetic pathway.
- DXP synthase inhibitors include bixlozone, broclozone and clomazone. Of note is a DXP synthase inhibitor selected from bixlozone and clomazone.
- HST homogentisate solanesyltransferase inhibitors
- HST inhibitors include cyclopyrimorate (6-chloro-3-(2- cyclopropyl-6-methylphenoxy)-4-pyridazinyl 4-morpholinecarboxylate), haloxydine, pyriclor, 3-(2-chloro-3,6-difluorophenyl)-4-hydroxy-1-methyl-1,5-naphthyridin-2(1H)-one, 7-(3,5-dichloro-4-pyridinyl)-5-(2,2-difluoroethyl)-8-hydroxypyrido[2,3-b]pyrazin-6(5H)-one and 4-(2,6-diethyl-4-methylphenyl)-5-hydroxy-2,6-dimethyl-3(2H)-pyridazinone.
- cyclopyrimorate 6-chloro-3-(2- cyclopropyl-6-methylphenoxy)-4-pyridazinyl 4-morpholinecarboxylate
- HST inhibitor is 6-chloro-4-(2,7-dimethyl-1-naphthalenyl)-5-hydroxy-2- methyl-3(2H)-pyridazinone.
- HST inhibitors also include compounds of Formulae A and B.
- Rd1 is H, Cl or CF 3 ;
- Rd2 is H, Cl or Br;
- Rd3 is H or Cl;
- Rd4 is H, Cl or CF 3 ;
- Rd5 is CH 3 , CH 2 CH 3 or CH 2 CHF 2 ;
- Re1 is H, F, Cl, CH 3 o r CH 2 CH 3 ;
- Re2 is H or CF 3 ;
- Re3 is H, CH 3 or CH 2 CH 3 ;
- Re4 is H, F or Br;
- Re5 is Cl, CH 3 , CF 3 , OCF 3 or CH 2 CH 3 ;
- Re6 is H, CH 3 , CH 2 CHF 2 or C ⁇ CH;
- a e8 is
- Cellulose biosynthesis inhibitors inhibit the biosynthesis of cellulose in certain plants. They are most effective when applied preemergence or early postemergence on young or rapidly growing plants. Examples of cellulose biosynthesis inhibitors include chlorthiamid, dichlobenil, flupoxam, indaziflam (N 2 -[(1R,2S)-2,3-dihydro-2,6-dimethyl-1H-inden-1-yl]-6- (1-fluoroethyl)-1,3,5-triazine-2,4-diamine), isoxaben and triaziflam.
- “DHODH (dihydroorotate dehydrogenase) inhibitors” act through inhibiting catalysis of the fourth step (i.e., inhibition of dihydroorotate dehydrogenase) of the de-novo pyrimidine biosynthetic pathway. Inhibition of pyrimidine biosynthesis leads to the cessation of plant growth. The first two steps of de-novo pyrimidine biosynthesis occur in the chloroplast after which the product, N-carbamoyl aspartate, is shuttled to the cytoplasm where dihydroorotase resides, producing dihydroorotate, the DHODH substrate. Plant DHODH is a flavin-dependent enzyme residing on the outer surface of the inner mitochondrial membrane.
- Reducing equivalents pass from dihydroorotate via the tightly bound flavin cofactor to a ubiquinone acceptor molecule that subsequently exchanges with the ubiquinol pool of the membrane and ultimately links DHODH enzymatic activity with oxidative phosphorylation.
- the remaining steps of the pathway leading to pyrimidine nucleotides occur in the cytoplasm.
- DHODH inhibitors include a compound of Formula (b16A) ( b16A) wherein R 12 is H, C 1 –C 6 alkyl, C 1 –C 6 haloalkyl or C 4 –C 8 cycloalkyl; R 13 is H, C 1 –C 6 alkyl or C 1 –C 6 alkoxy; Q 1 is an optionally substituted ring system selected from the group consisting of phenyl, thienyl, pyridinyl, benzodioxolyl, naphthyl, naphthalenyl, benzofuranyl, furanyl, benzothiophenyl and pyrazolyl, wherein when substituted said ring system is substituted by 1 to 3 R 14 ; Q 2 is an optionally substituted ring system selected from the group consisting of phenyl, pyridinyl, benzodioxolyl, pyridinonyl, thiadiazolyl, thiazolyl
- R 12 is H or C 1 –C 6 alkyl; more preferably R 12 is H or methyl.
- R 13 is H.
- Q 1 is either a phenyl ring or a pyridinyl ring, each ring substituted by 1 to 3 R 14 ; more preferably Q 1 is a phenyl ring substituted by 1 to 2 R 14 .
- Q 2 is a phenyl ring substituted by 1 to 3 R 15 ; more preferably Q 2 is a phenyl ring substituted by 1 to 2 R 15 .
- each R 14 is independently halogen, C 1 –C 4 alkyl, C 1 –C 3 haloalkyl, C 1 –C 3 alkoxy or C 1 –C 3 haloalkoxy; more preferably each R 14 is independently chloro, fluoro, bromo, C 1 –C 2 haloalkyl, C 1 –C 2 haloalkoxy or C 1 –C 2 alkoxy.
- each R 15 is independently halogen, C 1 –C 4 alkyl, C 1 –C 3 haloalkoxy; more preferably each R 15 is independently chloro, fluoro, bromo, C 1 –C 2 haloalkyl, C 1 –C 2 haloalkoxy or C 1 –C 2 alkoxy.
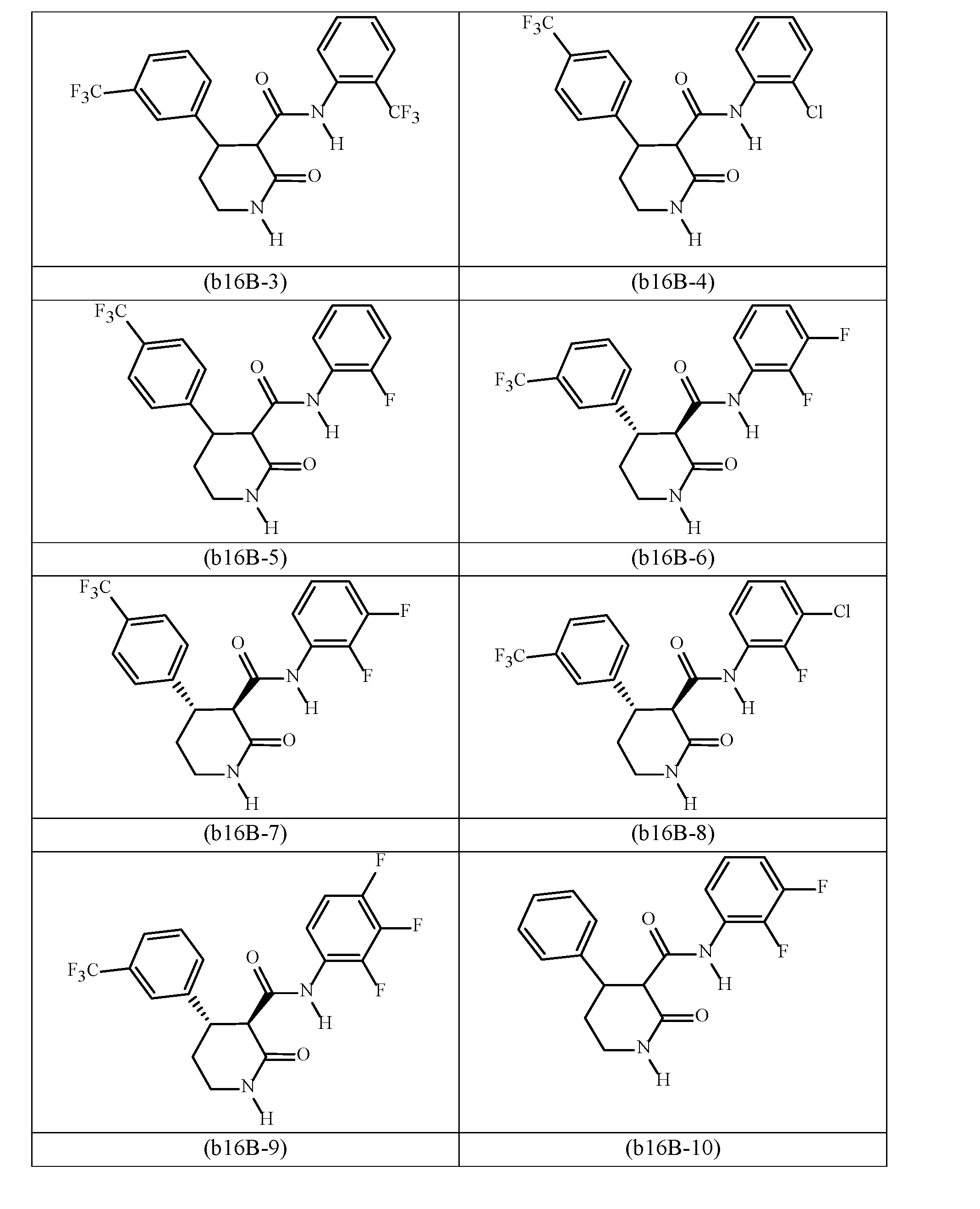
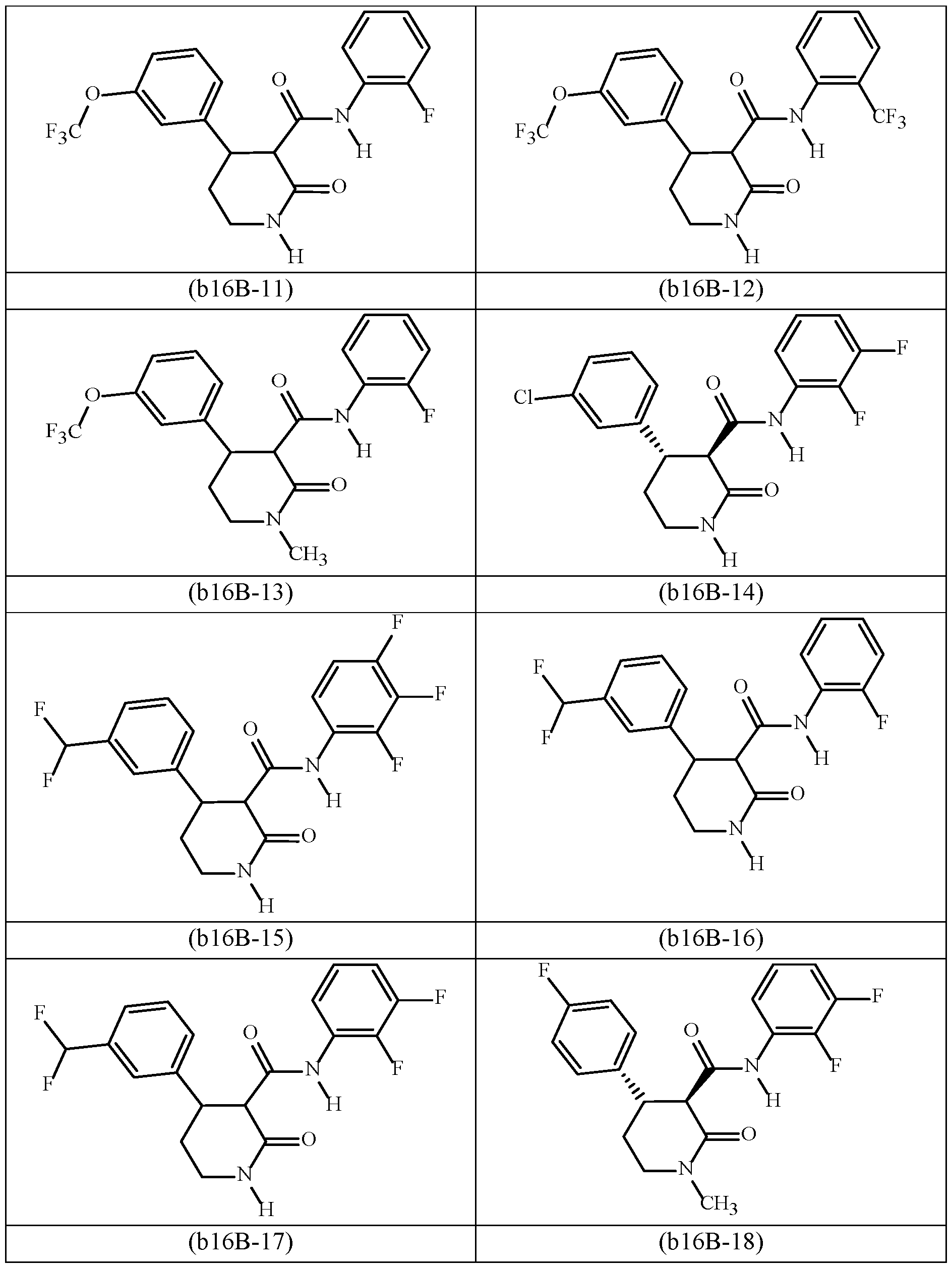
- DHODH inhibitors include any one of the following (b16A-1) through (b16A-17): N-(2-fluorophenyl)-2-oxo-4-[3-(trifluoromethyl)phenyl]-3-pyrrolidinecarboxamide (b-16A- 1); N-(2,3-difluorophenyl)-2-oxo-4-[3-(trifluoromethyl)phenyl]-3-pyrrolidinecarboxamide (b16A-2); 2-oxo-4-[3-(trifluoromethyl)phenyl]-N-(2,3,4-trifluorophenyl)-3-pyrrolidinecarboxamide (b16A-3); N-(2-fluorophenyl)-1-methyl-2-oxo-4-[3-(trifluoromethyl)phenyl]-3-pyrrolidinecarboxamide (b16A-4); N-(2-fluorophenyl)-2-oxo-4-[4-(trifluoromethyl)phenyl]-3
- DHODH inhibitor is tetflupyrolimet.
- “Other herbicides” include herbicides that act through a variety of different modes of action such as mitotic disruptors (e.g., icafolin, flamprop-M-methyl and flamprop-M-isopropyl), organic arsenicals (e.g., DSMA, and MSMA), 7,8-dihydropteroate synthase inhibitors, chloroplast isoprenoid synthesis inhibitors and cell-wall biosynthesis inhibitors.
- Other herbicides include those herbicides having unknown modes of action or do not fall into a specific category listed in (b1) through (b16) or act through a combination of modes of action listed above.
- herbicides examples include aclonifen, asulam, amitrole, bromobutide, cinmethylin, cumyluron, cyclopyrimorate (6-chloro-3-(2-cyclopropyl- 6-methylphenoxy)-4-pyridazinyl 4-morpholinecarboxylate), daimuron, difenzoquat, dimesulfazet, etobenzanid, fluometuron, flurenol, fosamine, fosamine-ammonium, dazomet, dymron, ipfencarbazone (1-(2,4-dichlorophenyl)-N-(2,4-difluorophenyl)-1,5-dihydro-N-(1- methylethyl)-5-oxo-4H-1,2,4-triazole-4-carboxamide), metam, methyldymron, oleic acid, oxaziclomefone, pel
- “Other herbicides” also include a compound of Formula (b16B) wherein R 18 is H, C 1 –C 6 alkyl, C 1 –C 6 haloalkyl or C 4 –C 8 cycloalkyl; each R 19 is independently halogen, C 1 –C 6 haloalkyl or C 1 –C 6 haloalkoxy; p is an integer of 0, 1, 2 or 3; each R 20 is independently halogen, C 1 –C 6 haloalkyl or C 1 –C 6 haloalkoxy; and q is an integer of 0, 1, 2 or 3.
- R 18 is H, methyl, ethyl or propyl; more preferably R 18 is H or methyl; most preferably R 18 is H.
- each R 19 is independently chloro, fluoro, C 1 – C 3 haloalkyl or C 1 –C 3 haloalkoxy; more preferably each R 19 is independently chloro, fluoro, C 1 fluoroalkyl (i.e., fluoromethyl, difluoromethyl or trifluoromethyl) or C 1 fluoroalkoxy (i.e., trifluoromethoxy, difluoromethoxy or fluoromethoxy).
- each R 20 is independently chloro, fluoro, C 1 haloalkyl or C 1 haloalkoxy; more preferably each R 20 is independently chloro, fluoro, C 1 fluoroalkyl (i.e., fluoromethyl, difluoromethyl or trifluromethyl) or C 1 fluoroalkoxy (i.e., trifluoromethoxy, difluoromethoxy or fluoromethoxy).
- R1 is Cl, Br or CN
- An example of an other herbicide is rimisoxafen.
- Herbicide safeners are substances added to a herbicide formulation to eliminate or reduce phytotoxic effects of the herbicide to certain crops.
- herbicide safeners include but are not limited to benoxacor, cloquintocet-mexyl, cumyluron, cyometrinil, cyprosulfamide, daimuron, dichlormid, dicyclonon, dietholate, dimepiperate, fenchlorazole-ethyl, fenclorim, flurazole, fluxofenim, furilazole, isoxadifen-ethyl, mefenpyr-diethyl, mephenate, methoxyphenone, naphthalic anhydride, oxabetrinil, N-(aminocarbonyl)-2-methylbenzenesulfonamide, N- (aminocarbonyl)-2-fluorobenzenesulfonamide, 1-bromo-4-[(chloromethyl)sulfonyl]benzen
- Preferred for better control of undesired vegetation e.g., lower use rate such as from greater-than-additive effects, broader spectrum of weeds controlled, or enhanced crop safety
- a herbicide selected from the group consisting of 4-amino-3-chloro-5-fluoro- 6-(7-fluoro-1H-indol-6-yl)-2-pyridinecarboxylic 2-propyn-1-yl ester (CAS No.
- Compounds of Formulae 1a, 1b, 1c, 1d, 1f, 8a, 8b, 10a, 10b, 10c, 10d, 11a, 31a, 31b, 32a and 32b are various subsets of the compounds of Formulae 1, 8, 10, 11, 31 and 32; and all substituents for Formulae 1a, 1b, 1c, 1d, 1f, 8a, 8b, 10a, 10b, 10c, 10d, 11a, 31a, 31b, 32a and 32b are as defined above for Formula 1 unless otherwise noted in the disclosure including the schemes.
- Stereochemically enriched compounds of Formula 1' and 1'' can be obtained from mixtures containing compounds of Formula 1' and 1'' through the utilization of well-known chiral chromatography separation methods.
- compounds of Formula 1a i.e., a compound of Formula 1 wherein Y is S
- compounds of Formula 1b i.e., a compond of Formula 1 wherein Y is O
- a thionation reagent such as Lawesson’s reagent, tetraphosphorus decasulfide or diphosphorus pentasulfide
- an appropriate solvent e.g., tetrahydrofuran or toluene
- Scheme 1 Compounds of Formula 1c (i.e., a compound of Formula 1b wherein Z is NR 3d and R 3d is other than H) can be prepared using method depicted in Scheme 2 wherein a compound of Formula 1d (i.e., a compound of Formula 1b where Z is NH) reacts with an alkylating agent or other suitable electrophile (e.g., acyl chloride or anhydride, sulfonyl chloride or anhydride) in the presence of a base, such as sodium hydride in a solvent such as N,N-dimethylformamide.
- an alkylating agent or other suitable electrophile e.g., acyl chloride or anhydride, sulfonyl chloride or anhydride
- Alternative bases for Scheme 2 reactions include, but are not limited to, potassium carbonate, sodium hydroxide, pyridine and triethylamine, and alternative solvents include dimethyl sulfoxide, tetrahydrofuran, ethyl acetate and dichloromethane. Methods analogous to those described in J. Med. Chem.2018, 61, 4317 and Eur. J. Med. Chem.2020, 188, 111987 may be used.
- compounds of Formula 1e i.e., a compound of Formula 1b wherein A is bonded to the rest of the molecule through a nitrogen ring member of A
- compounds of Formula 2 wherein Q is a halogen or pseudohalogen such as Cl, Br, I or OTf
- Q is a halogen or pseudohalogen such as Cl, Br, I or OTf
- heterocycles of Formula 3 wherein H is connected to a nitrogen ring member of A
- Chem.2004, 69, 5578-5587 are often suitable and are typically catalyzed by a copper salt such as copper(I) iodide or copper(I) oxide and a ligand, such as trans-N,N′-dimethylcyclohexane-1,2-diamine, trans-1,2-diaminocyclohexane, N,N′-dimethylethylenediamine, 1,10-phenanthroline, 8-quinolinol, (S)-proline or 2-picolinic acid, in the presence of a base (e.g., potassium carbonate, cesium carbonate or potassium phosphate) in an appropriate solvent (e.g., N,N-dimethylformamide, dimethyl sulfoxide, N,N- dimethylacetamide, toluene, 1,4-dioxane or acetonitrile).
- a base e.g., potassium carbonate, cesium carbonate or potassium phosphate
- an appropriate solvent
- palladium-catalyzed coupling conditions may also be suitable for some heterocycles.
- Appropriate palladium catalysts include but are not limited to tetrakis(triphenylphosphine)palladium(0), [1,1′-bis(diphenylphosphino)ferrocene]dichloropalladium(II), bis(triphenylphosphine)- palladium(II) dichloride, palladium(II) acetate or tris(dibenzylideneacetone)dipalladium(0).
- a ligand is beneficial, including but not limited to, 2,2′- bis(diphenylphosphino)-1,1′-binaphthalene (BINAP), 2-dicyclohexylphosphino-2′,6′- dimethoxybiphenyl (SPhos), 2-dicyclohexylphosphino-2′,4′,6′-triisopropylbiphenyl (XPhos), 4,5-bis(diphenylphosphino)-9,9-dimethylxanthene (Xantphos), 2-(di-tert-butylphosphino)- biphenyl (JohnPhos), 2-dicyclohexylphosphino-2',6'-diisopropoxy-1,1'-biphenyl (RuPhos) or 2-di-tert-butylphosphino-3,4,5,6-tetramethyl-2′,4′,6′-triisopropyl
- BINAP 2,
- heterocycles of Formula 3 are generally commercially available or known in the literature.
- compounds of Formula 1e can generally be prepared without the addition of a metal catalyst.
- the metal catalysts used in these couplings include, but are not limited to, tetrakis(triphenylphosphine)palladium(0), [1,1′-bis(diphenylphosphino)ferrocene]dichloro- palladium(II), bis(triphenylphosphine)palladium(II) dichloride, palladium(II) acetate and tris(dibenzylideneacetone)dipalladium(0).
- a ligand is beneficial, including but not limited to, 2-dicyclohexylphosphino-2′,4′,6′-triisopropylbiphenyl (XPhos), 2-dicyclohexylphosphino-2′,6′-dimethoxybiphenyl (SPhos), tricyclohexylphosphine and tri(2-furyl)phosphine.
- XPhos 2-dicyclohexylphosphino-2′,4′,6′-triisopropylbiphenyl
- SPhos 2-dicyclohexylphosphino-2′,6′-dimethoxybiphenyl
- tricyclohexylphosphine tri(2-furyl)phosphine.
- these reactions are run in solvents such as dimethyl sulfoxide, N,N-dimethylformamide, N-methyl-2-pyrrolidone, toluene, 1,2-dimethoxyethane, 1,4-dioxane, tetrahydrofuran, acetonitrile or ethanol and temperatures generally range from ambient temperature to 150 °C.
- solvents such as dimethyl sulfoxide, N,N-dimethylformamide, N-methyl-2-pyrrolidone, toluene, 1,2-dimethoxyethane, 1,4-dioxane, tetrahydrofuran, acetonitrile or ethanol and temperatures generally range from ambient temperature to 150 °C.
- solvents such as dimethyl sulfoxide, N,N-dimethylformamide, N-methyl-2-pyrrolidone, toluene, 1,2-dimethoxyethane, 1,4-dioxane, tetrahydro
- compounds of Formula 2 are coupled with heterocycles of Formula 6 (wherein M is a transmetalating group such as, but not limited to, B(OH) 2 , B(–O(CMe 2 ) 2 O-), BF 3 K, Sn(n-Bu) 3 , SnMe 3 or ZnBr; and M is connected to a carbon ring member).
- M is a transmetalating group such as, but not limited to, B(OH) 2 , B(–O(CMe 2 ) 2 O-), BF 3 K, Sn(n-Bu) 3 , SnMe 3 or ZnBr; and M is connected to a carbon ring member.
- M is a transmetalating group such as, but not limited to, B(OH) 2 , B(–O(CMe 2 ) 2 O-), BF 3 K, Sn(n-Bu) 3 , SnMe 3 or ZnBr; and M is connected to a carbon ring member.
- Heterocycles of Formula 6 are generally commercially available or known in the literature. In some instances, compounds of Formula 1f may be more readily accessed using standard heterocyclic synthesis procedures known to those skilled in the art. For appropriate methods, see Science of Synthesis, Volumes 11-13 and 15, Schaumann, E., Neier, R., Storr, R. C.; Gilchrist, T. L. and Black, D. S. (volume editors), Thieme (2001-2004) and Comprehensive Heterocyclic Chemistry IV, Volumes 4-7, Black, D. S., Cossy, J. and Stevens, C. V.
- organometallic compounds of Formula 5 wherein M is a transmetalating group such as, but not limited to, B(–O(CMe 2 ) 2 O–), Sn(n-Bu) 3 or SnMe 3
- Q is a halogen or pseudohalogen such as Cl, Br, I or OTf
- pinacol boronic esters (wherein M is B(–O(CMe 2 ) 2 O—), can generally be prepared using palladium-catalyzed borylation conditions, such as those described in J. Org. Chem.1995, 60, 7508-7510 and J. Org. Chem. 2021, 86, 103-109; and references cited therein.
- a compound of Formula 2 is treated with bis(pinacolato)diboron in the presence of a palladium catalyst like [1,1′-bis(diphenylphosphino)ferrocene]dichloropalladium(II) and a base such as potassium acetate in a solvent such as dioxane or dimethyl sulfoxide, at temperatures ranging from ambient temperature to the reflux temperature of the solvent.
- a palladium catalyst like [1,1′-bis(diphenylphosphino)ferrocene]dichloropalladium(II) and a base such as potassium acetate
- a solvent such as dioxane or dimethyl sulfoxide
- trialkyl tin reagents can generally be prepared from a compound of Formula 2 by treatment with a stannane such as hexamethylditin or hexabutylditin in the presence of a palladium catalyst like tetrakis(triphenylphosphine)palladium(0) in a solvent like dioxane or toluene, at temperatures ranging from ambient temperature to the reflux temperature of the solvent.
- a stannane such as hexamethylditin or hexabutylditin
- a palladium catalyst like tetrakis(triphenylphosphine)palladium(0)
- solvent like dioxane or toluene
- Scheme 6 As shown in Scheme 7, compounds of Formulae 1 or 2 can be prepared by reacting a carboxylic acid of Formula 7 (where R a is H) with an amine of Formula 8. The reaction proceeds via activation of the carboxylic acid of Formula 7 followed by reaction with the amine of Formula 8.
- the carboxylic acid can be activated with a coupling reagent or by conversion of the carboxylic acid to an acid halide, such as an acid chloride.
- compounds of Formulae 7 and 8 can be reacted in the presence of a coupling reagent such as propylphosphonic anhydride (T3P), N-(3-dimethylaminopropyl)-N′-ethylcarbodiimide hydrochloride (EDC), 1,1′-carbonyldiimidazole (CDI), 2-chloro-1-methylpyridinium iodide (Mukaiyama’s reagent) or the combination of N,N,N′,N′-tetramethylchloroformamidinium hexafluorophosphate (TCFH) and N-methylimidazole (NMI).
- T3P propylphosphonic anhydride
- EDC N-(3-dimethylaminopropyl)-N′-ethylcarbodiimide hydrochloride
- CDI 1,1′-carbonyldiimidazole
- Mukaiyama’s reagent 2-chloro-1-
- Polymer supported reagents such as polymer-supported cyclohexylcarbodiimide, are also suitable. These reactions are typically run in an appropriate solvent (e.g., dichloromethane, 1,2-dichloroethane, ethyl acetate, acetonitrile or N,N-dimethylformamide) in the presence of a base (e.g., triethylamine, N,N-diisopropylethylamine or pyridine), optionally with a catalytic amount of 4-(dimethylamino)pyridine (DMAP), at temperatures ranging from 0 °C to the reflux temperature of the solvent.
- an appropriate solvent e.g., dichloromethane, 1,2-dichloroethane, ethyl acetate, acetonitrile or N,N-dimethylformamide
- a base e.g., triethylamine, N,N-diisopropylethyl
- a carboxylic acid of Formula 7 can be converted to an acid chloride by treatment with a reagent such as thionyl chloride, oxalyl chloride, phosphoryl chloride, phosphorus trichloride or phosphorus pentachloride, either neat or in an appropriate solvent (e.g., dichloromethane, 1,2-dichloroethane or toluene) and optionally with a catalytic amount of N,N-dimethylformamide, at temperatures ranging from 0 °C to the reflux temperature of the solvent.
- a reagent such as thionyl chloride, oxalyl chloride, phosphoryl chloride, phosphorus trichloride or phosphorus pentachloride, either neat or in an appropriate solvent (e.g., dichloromethane, 1,2-dichloroethane or toluene) and optionally with a catalytic amount of N,N-dimethylformamide, at temperatures ranging from
- Subsequent reaction of the acid chloride with the amine of Formula 8 generally occurs in the presence of a base (e.g., triethylamine, N,N- diisopropylethylamine or pyridine), optionally with a catalytic amount of 4- (dimethylamino)pyridine, in an appropriate solvent (e.g., tetrahydrofuran, dioxane, toluene, dichloromethane, chloroform, 1,2-dichloroethane or ethyl acetate) at temperatures ranging from 0 °C to the reflux temperature of the solvent.
- a base e.g., triethylamine, N,N- diisopropylethylamine or pyridine
- 4- (dimethylamino)pyridine e.g., tetrahydrofuran, dioxane, toluene, dichloromethane, chloroform, 1,2-dichloroethane or
- Carboxylic acids of Formula 7 are generally commercially available or known in the literature. In some cases, the corresponding acid chlorides are also commercially available. Where Q is A, such carboxylic acids of Formula 7 can be prepared using the reaction conditions described in Schemes 2, 3 and 4 starting from carboxylic acids or esters of Formula 7 where Q is a halogen or pseudohalogen.
- esters are used as starting materials (i.e., R a is C 1 -C 4 alkyl)
- a second step may be required to convert the ester to the carboxylic acid using standard conditions known to those skilled in the art (e.g., hydrolysis with sodium hydroxide).
- esters of Formula 7 (wherein R a is C1-C4 alkyl, typically methyl or ethyl) may undergo direct amidation with amines of Formula 8 by treatment with trimethylaluminum in a solvent like toluene, at temperatures ranging from 0 °C to the reflux temperature of the solvent.
- a solvent like toluene like toluene
- lactams of Formula 9 are treated with a reducing agent, such as borane (e.g., as a tetrahydrofuran or dimethylsulfide adduct), lithium aluminum hydride or diisobutylaluminum hydride in an appropriate solvent (e.g., tetrahydrofuran or toluene) at temperatures typically ranging from 0 °C to the reflux temperature of the solvent.
- a reducing agent such as borane (e.g., as a tetrahydrofuran or dimethylsulfide adduct)
- lithium aluminum hydride or diisobutylaluminum hydride e.g., tetrahydrofuran or toluene
- lactams of Formula 9 can be prepared from compounds of Formula 10 (wherein R a is C 1 -C 4 alkyl, typically methyl or ethyl) by a nitro reduction followed by cyclization which occurs during reduction or upon heating.
- This reaction is readily achieved under a range of conditions, such as iron metal in the presence of an acid like acetic acid, hydrochloric acid or aqueous ammonium chloride, optionally with a solvent such as methanol, ethanol, ethyl acetate or N,N-dimethylformamide at temperatures ranging from ambient temperature to the reflux temperature of the solvent.
- reaction can be achieved using a transition metal catalyst, such as palladium on carbon, platinum oxide or Raney nickel under an atmosphere of hydrogen, in an appropriate solvent (e.g., methanol, ethanol, ethyl acetate or tetrahydrofuran). Temperatures typically range from ambient temperature to 80 °C. This reaction can generally be done in a Parr hydrogenator. For relevant examples of this reaction in the literature, see: WO 2013/130660; WO 2015/095795; WO 2022/204336 and Angew. Chem. Int.
- Typical reaction conditions include triphenylphosphine and an azodicarboxylate such as diethyl azodicarboxylate or diisopropyl azodicarboxylate, in a solvent like tetrahydrofuran or dichloromethane at temperatures ranging from about –10 °C to the reflux temperature of the solvent.
- an azodicarboxylate such as diethyl azodicarboxylate or diisopropyl azodicarboxylate
- compounds of Formula 10b i.e., compounds of Formula 10 wherein Z is O or NR 3d
- compounds of Formula 11 can be prepared by reaction of compounds of Formula 11 (wherein Z is O or NR 3d ) with ⁇ -halo esters of Formula 13 (wherein LG is a leaving group such as Cl or Br and R a is C 1 -C 4 alkyl, typically methyl or ethyl) in the presence of a base, such as potassium carbonate or cesium carbonate, in a solvent like N,N-dimethylformamide, acetone or 1,4-dioxane, at temperatures typically ranging from 0 °C to the reflux temperature of the solvent.
- a base such as potassium carbonate or cesium carbonate
- compounds of Formula 10c i.e., a compound of Formula 10 wherein Z is CH 2
- compounds of Formula 10c can be prepared by the reaction of a compound of Formula 14 (wherein LG is a leaving group such as Cl or Br) with an ester of Formula 15 (wherein R a is C 1 -C 4 alkyl, typically methyl or ethyl) in the presence of a base, such as lithium diisopropylamide or lithium bis(trimethylsilyl)amide, in a solvent like tetrahydrofuran, at temperatures typically ranging from –78 °C to ambient temperature.
- a base such as lithium diisopropylamide or lithium bis(trimethylsilyl)amide
- reaction of the compound of Formula 14 (wherein LG is a leaving group such as Cl or Br) with a diester of Formula 16 can provide a compound of Formula 17 by treatment with a base, such as sodium hydride or cesium carbonate, in an appropriate solvent like tetrahydrofuran or N,N-dimethylformamide, at temperatures typically ranging from 0 °C to the reflux temperature of the solvent.
- a base such as sodium hydride or cesium carbonate
- a compound of Formula 10d (i.e., compounds of Formula 10 wherein Z is CH 2 and R 3b is H), which may be achieved by heating the compound of Formula 17 in a solvent system like N,N-dimethylformamide/water or dimethyl sulfoxide/water at temperatures ranging from 100 °C to 200 °C.
- R a is methyl
- Krapcho- type decarboxylation conditions can also be appropriate, by heating the compound of Formula 17 in the presence of a salt like sodium chloride or lithium chloride, in a solvent system like dimethyl sulfoxide/water or N,N-dimethylformamide/water at temperatures ranging from 100 °C to 200 °C.
- An alternative method to convert a compound of Formula 17 to a compound of Formula 10d involves ester hydrolysis (e.g., potassium hydroxide in ethanol) followed by thermal decarboxylation of the resulting carboxylic acid (e.g., reflux in a solvent like dioxane, toluene or xylene).
- ester hydrolysis e.g., potassium hydroxide in ethanol
- thermal decarboxylation of the resulting carboxylic acid e.g., reflux in a solvent like dioxane, toluene or xylene
- acidic conditions may be more appropriate (e.g., reflux in acetic acid or hydrochloric acid).
- hydrolysis of the remaining ester may occur; in these instances, re-esterification using standard conditions (e.g., thionyl chloride in methanol) may be necessary.
- Lactams of Formula 9a i.e., compounds of Formula 9 wherein Z is NH
- Scheme 12 Examples of procedures analogous to those depicted in Scheme 12 can be found in J. Med.
- ⁇ -aminoalcohols of Formula 21 (wherein R b is H or an appropriate protecting group such as tosyl (i.e., Ts) or benzyl (i.e., Bn); and X is a halogen or pseudohalogen such as F, Cl, Br, I or OTs) can be prepared by ring opening of epoxides of Formula 20 and subsequent nucleophilic attack by anilines of Formula 22. This reaction can generally be achieved by heating the aniline and epoxide either neat or in an appropriate solvent (e.g., ethanol or N,N- dimethylformamide) at temperatures generally ranging from 40 °C to 180 °C.
- an appropriate solvent e.g., ethanol or N,N- dimethylformamide
- a base e.g., potassium carbonate or sodium hydride
- Lewis acid e.g., ytterbium(III) trifluoromethanesulfonate or lithium bromide
- phase transfer catalyst e.g., benzyltriethylammonium chloride or tetrabutylammonium bromide
- acidic conditions may reverse the regioselectivity of epoxide ring-opening.
- N-tosyl protected amines of Formula 23 can be prepared from ⁇ -aminoalcohols of Formula 21a (i.e., compounds of Formula 21 wherein R b is Ts and X is typically F but in some instances is OTs) by treatment with a base such as, but not limited to, sodium hydroxide, potassium tert-butoxide or sodium hydride, optionally in the presence of a phase transfer catalyst like tetrabutylammonium bromide, typically in a solvent such as tetrahydrofuran at temperatures ranging from ambient temperature to the reflux temperature of the solvent.
- a base such as, but not limited to, sodium hydroxide, potassium tert-butoxide or sodium hydride
- a phase transfer catalyst like tetrabutylammonium bromide
- Amines of Formula 8b can be prepared by removal of the N-tosyl protecting group. This can be achieved under reducing conditions, for example, by treatment with magnesium metal in methanol at temperatures ranging from 0 °C to the reflux temperature of the solvent, optionally with sonication. Alternatively, the reaction can be achieved using acidic hydrolysis conditions, for example, treatment with sulfuric acid either neat or in a solvent like dichloromethane, at temperatures generally ranging from 0 °C to the reflux temperature of the solvent. For relevant examples of these reactions in the literature, see: Chem. Commun.
- amines of Formulae 8b (wherein R b is H) or protected amines of Formula 24 (wherein R b is a suitable protecting group like Ts or Bn) can be prepared from ⁇ -aminoalcohols of Formula 21b (i.e., compounds of Formula 21 wherein R b is H or a suitable protecting group and X is a halogen such as Cl, Br or I) by transition metal-catalyzed O-arylation.
- This reaction can be catalyzed by a copper salt such as copper(I) iodide and a ligand such as 1,10-phenanthroline, or by a palladium salt or complex such as palladium(II) acetate or tris(dibenzylideneacetone)dipalladium(0) and a phosphine ligand such as 2-di-tert-butylphosphino-2′-(N,N-dimethylamino)biphenyl (t-Bu-DavePhos) or rac-2-(Di- tert-butylphosphino)-1,1′-binaphthyl (TrixiePhos).
- a copper salt such as copper(I) iodide and a ligand such as 1,10-phenanthroline
- a palladium salt or complex such as palladium(II) acetate or tris(dibenzylideneacetone)dipalladium(0) and
- amines of Formula 8b may be more readily accessed according to the sequence shown in Scheme 16.
- Compounds of Formula 25 can be prepared by ring opening of epoxides of Formula 20 and subsequent nucleophilic attack by phenols of Formula 11a (i.e., compounds of Formula 11 wherein Z is O).
- This reaction can generally be achieved in the presence of a base (e.g., monosodium phosphate, sodium phosphate, potassium carbonate, sodium hydride, sodium hydroxide, cesium fluoride or 1,8-diazabicyclo[5.4.0]undec-7-ene) in an appropriate solvent (e.g., acetonitrile, N,N-dimethylformamide, dimethyl sulfoxide, dichloromethane, toluene, methanol, isopropanol and/or water), at temperatures generally ranging from ambient temperature to 180 °C.
- Lewis acids e.g., zinc chloride or boron trifluoride diethyl etherate
- compounds of Formula 26 can be prepared by nitro reduction of the compounds of Formula 25, which can be achieved using standard conditions as described previously in Scheme 9, such as for example palladium on carbon under an atmosphere of hydrogen in an appropriate solvent like methanol.
- an acid e.g., phosphoric acid or p-toluenesulfonic acid
- an appropriate solvent e.g., xylene or toluene
- an appropriate aniline protecting group e.g., tosyl
- Mitsunobu conditions as described previously in Scheme 10 (e.g., triphenyl phosphine and diethyl azodicarboxylate in an appropriate solvent like tetrahydrofuran).
- Mitsunobu conditions e.g., triphenyl phosphine and diethyl azodicarboxylate in an appropriate solvent like tetrahydrofuran.
- This reaction can be achieved using a transition metal catalyst, such as but not limited to palladium on carbon, platinum on carbon or Raney nickel under an atmosphere of hydrogen, in an appropriate solvent (e.g., methanol, ethanol, isopropanol, ethyl acetate, toluene or tetrahydrofuran). Temperatures typically range from ambient temperature to 80 °C.
- a transition metal catalyst such as but not limited to palladium on carbon, platinum on carbon or Raney nickel under an atmosphere of hydrogen
- an appropriate solvent e.g., methanol, ethanol, isopropanol, ethyl acetate, toluene or tetrahydrofuran.
- Temperatures typically range from ambient temperature to 80 °C.
- This reaction can generally be done using a Parr hydrogenator, optionally with reaction pressures above atmospheric pressure. Alternatively, the reaction can be achieved in a stepwise manner.
- Nitro reduction can be achieved under a range of conditions, such as iron metal in the presence of an acid such as acetic acid, hydrochloric acid or aqueous ammonium chloride, optionally with a solvent such as methanol, ethanol, ethyl acetate, tetrahydrofuran or N,N-dimethylformamide at temperatures ranging from ambient temperature to the reflux temperature of the solvent.
- an acid such as acetic acid, hydrochloric acid or aqueous ammonium chloride
- a solvent such as methanol, ethanol, ethyl acetate, tetrahydrofuran or N,N-dimethylformamide
- Other suitable conditions include zinc metal with acetic acid or aqueous ammonium chloride, and stannous chloride in aqueous hydrochloric acid or ethanol.
- the second step involves imine reduction which can be achieved using a reducing agent like sodium borohydride, sodium cyanoborohydride or lithium aluminum hydride in an appropriate solvent (e.g., ethanol, methanol, tetrahydrofuran or dichloromethane, optionally with a co-solvent or additive like water or acetic acid) at temperatures generally between -78 °C and the reflux temperature of the solvent.
- a reducing agent like sodium borohydride, sodium cyanoborohydride or lithium aluminum hydride in an appropriate solvent
- an appropriate solvent e.g., ethanol, methanol, tetrahydrofuran or dichloromethane, optionally with a co-solvent or additive like water or acetic acid
- suitable reducing conditions for this step include but are not limited to hydrogen and palladium on carbon in a solvent such as methanol, or triethylsilane and trifluoroacetic acid in a solvent such as dichloromethane.
- Scheme 17 compounds of Formula 27 can be prepared by reaction of nitrophenols of Formula 11a with compounds of Formula 30 (wherein LG is a leaving group such as Cl or Br and R c is H or C 1 -C 3 alkyl; or R c can be taken together with R 3a or R 3b to form a ring) in the presence of a base, such as potassium carbonate, cesium carbonate, sodium bicarbonate or sodium hydride, optionally with additives such as sodium iodide, in a solvent like N,N-dimethylformamide, acetone or 1,4-dioxane, at temperatures typically ranging from 0 °C to the reflux temperature of the solvent.
- a base such as potassium carbonate, cesium carbonate, sodium bicarbonate or sodium hydride
- Mitsunobu reaction conditions may be appropriate in some cases, for the reaction of nitrophenols of Formula 11a with compounds of Formula 29 (wherein R c is H or C 1 -C 3 alkyl; or R c can be taken together with R 3a or R 3b to form a ring).
- Typical reaction conditions include triphenylphosphine and an azodicarboxylate such as diethyl azodicarboxylate or diisopropyl azodicarboxylate, in a solvent like tetrahydrofuran or dichloromethane at temperatures ranging from about –10 °C to the reflux temperature of the solvent.
- Nitrophenols, ⁇ -hydroxy carbonyls and ⁇ -halo carbonyls of Formulae 11a, 29 and 30 are generally commercially available or known in the literature.
- compounds of Formula 27 may be more readily accessed using protecting groups to mask the carbonyl group, such as an acetal group to mask an aldehyde, or via standard functional group interconversions of one carbonyl functional group to another.
- Scheme 18 An alternative synthesis of amines of Formula 8d (i.e., compounds of Formula 8 wherein Z is CR 3c or NH) involves reduction of azaarenes of Formula 31, as shown in Scheme 19.
- This reaction can be achieved using a transition metal catalyst, such as palladium, palladium hydroxide, platinum, platinum dioxide or Raney nickel under an atmosphere of hydrogen, in an appropriate solvent (e.g., methanol, ethanol, ethyl acetate, tetrahydrofuran and/or acetic acid). Temperatures typically range from ambient temperature to 80 °C. This reaction can generally be done in a Parr hydrogenator.
- a transition metal catalyst such as palladium, palladium hydroxide, platinum, platinum dioxide or Raney nickel under an atmosphere of hydrogen
- an appropriate solvent e.g., methanol, ethanol, ethyl acetate, tetrahydrofuran and/or acetic acid.
- Suitable conditions include transfer hydrogenation using a Hantzsch dihydropyridine (e.g., Hantzsch ester) as the hydrogen source, in the presence of a catalyst (e.g., diphenyl phosphate, trifluoroacetic acid, boric acid, iron(II) triflate or silver(I) triflate) in an appropriate solvent (e.g., toluene, benzene, dichloromethane, 1,2-dichloroethane or chloroform) at temperatures generally ranging from 0 °C to the reflux temperature of the solvent.
- a catalyst e.g., diphenyl phosphate, trifluoroacetic acid, boric acid, iron(II) triflate or silver(I) triflate
- an appropriate solvent e.g., toluene, benzene, dichloromethane, 1,2-dichloroethane or chloroform
- Sodium cyanoborohydride is also an appropriate reducing agent, used in the presence of an acid (e.g., hydrochloric acid or acetic acid) in a solvent (e.g., ethanol, water, methanol, tetrahydrofuran, or combinations thereof) at temperatures generally ranging from 0 °C to the reflux temperature of the solvent.
- a solvent e.g., ethanol, water, methanol, tetrahydrofuran, or combinations thereof
- Borane e.g., borane tetrahydrofuran complex
- Many azaarenes of Formula 31 are commercially available or known in the literature.
- quinolines of Formula 31a i.e., compounds of Formula 31 wherein R 3b is other than halogen; Z is CR 3c ; and R 3c is other than a leaving group, such as halogen, cyano, nitro or alkoxy
- 2-aminobenzaldehydes or ketones of Formula 32a with aldehydes or ketones of Formula 33 (wherein R 3b is not halogen) in the presence of an acid (e.g., p-toluenesulfonic acid, trifluoroacetic acid, acetic acid or hydrochloric acid) or a base (e.g., sodium hydroxide, potassium hydroxide or sodium methoxide).
- an acid e.g., p-toluenesulfonic acid, trifluoroacetic acid, acetic acid or hydrochloric acid
- a base e.g., sodium hydroxide, potassium hydroxide or sodium methoxide
- a solvent is typically used (e.g., ethanol, methanol, butan-2-one or toluene, optionally with water as a co-solvent). Temperatures generally range from ambient temperature to 200 °C. Alternatively, the reaction may be promoted by organosilanes (e.g., trimethylsilyl chloride) or catalyzed by Lewis acids (e.g., ytterbium triflate). For relevant examples of these reactions in the literature, see: WO 2021/092525; WO 2022/053422 and Tetrahedron Lett. 2011, 52, 3474-3477.
- organosilanes e.g., trimethylsilyl chloride
- Lewis acids e.g., ytterbium triflate
- quinolines of Formula 31b can be prepared by reaction of 2-aminobenzoic acids, esters or acyl chlorides of Formula 32b with aldehydes or ketones of Formula 33 (where R 3b is not halogen).
- the reaction is often conducted in a solvent (e.g., diphenyl ether, xylene or toluene) at temperatures typically ranging from 80 °C to 230 °C.
- the reaction may be promoted by Br ⁇ nsted acids (e.g., methanesulfonic acid and/or polyphosphoric acid), where the acid may also be used as the solvent, or Lewis acids (e.g., zinc chloride).
- Lewis acids e.g., zinc chloride
- the hydroxy group (R 3c ) in quinolines of Formula 31b can be converted to other functional groups using methods known to those skilled in the art, for example treatment with phosphorus oxychloride may provide the corresponding chloroquinoline (i.e., R 3c is Cl).
- Compounds of Formulae 32 and 33 are generally commercially available or known in the literature.
- Scheme 20 other than halogen 3 2a is other than w herein R 3c 3 1a is oth w herein R 3c er than or alkoxy. or alkoxy. h alogen, CN, NO 2 halogen, CN, NO 2 32b wherein R 3c is OH. 3 1b is OH. w herein R 3c Dihydroquinoxalinones of Formula 8e (i.e., compounds of Formula 8 wherein Z is NH and the adjacent R 3a and R 3b are taken together with the carbon atom to which they are attached to form a carbonyl group) can be prepared by the reaction of 2-bromoanilines of Formula 34 with amino acids of Formula 35 as depicted in Scheme 21, using conditions as described previously in Scheme 12.
- amines of Formula 8f may be prepared according to the sequence shown in Scheme 22.
- compounds of Formula 36 can be prepared by reaction of 2-aminobenzaldehydes or ketones of Formula 32a with carboxylic acids or acid chlorides of Formula 38 (where R c is OH or Cl) using amidation conditions previously described in Scheme 7.
- compounds of Formula 37 can be prepared by a cyclization of compounds of Formula 36 in the presence of a base (e.g., potassium carbonate, sodium hydroxide, potassium tert-butoxide) in a solvent (e.g., N,N-dimethylformamide, ethanol, dimethyl sulfoxide, 1,4-dioxane) at temperatures generally ranging from ambient temperature to 120 °C.
- a base e.g., potassium carbonate, sodium hydroxide, potassium tert-butoxide
- a solvent e.g., N,N-dimethylformamide, ethanol, dimethyl sulfoxide, 1,4-dioxane
- amines of Formula 8f can be prepared from compounds of Formula 37 by treatment with a reducing agent like borane or lithium borohydride in a solvent like tetrahydrofuran at temperatures ranging from 0 °C to the reflux temperature of the solvent.
- a reducing agent like borane or lithium borohydride
- a solvent like tetrahydrofuran
- the same reduction can also be achieved using multi-step approaches, such as conversion of compounds of Formula 37 to 2-chloroquinolines by treatment with phosphorus oxychloride, followed by dechlorination (e.g., by hydrogenation in the presence of catalytic palladium) followed by the reduction of the resultant quinoline using the method previously described in Scheme 19.
- dechlorination e.g., by hydrogenation in the presence of catalytic palladium
- HPLC high performance liquid chromatography.
- MPLC medium pressure liquid chromatography.
- LCMS liquid chromatography coupled with mass spectrometry.
- a sealed tube was charged with 5-bromo-3-methyl-quinoline (i.e., the product of Step A) (200 mg, 0.9 mmol), trimethylboroxine (148 mg, 1.2 mmol), potassium carbonate (310 mg, 2.2 mmol) and 1,4-dioxane (9 mL). The mixture was sparged with argon for 5- 10 min. Tetrakis(triphenylphosphine)palladium(0) (520 mg, 0.45 mmol) was added to the mixture. The tube was sealed with a Teflon cap and stirred at 110 °C for 16 h. The mixture was cooled to room temperature, filtered through a pad of Celite ® with ethyl acetate.
- Step E Preparation of (3,4-dihydro-3,5-dimethyl-1(2H)-quinolinyl)[2-methoxy-5-[3- (1-methylethyl)-1H-1,2,4-triazol-1-yl]phenyl]methanone (alternatively named (3,5-dimethyl-3,4-dihydro-2H-quinolin-1-yl)-[5-(3-isopropyl-1,2,4-triazol-1- yl)-2-methoxy-phenyl]methanone).
- Step A Preparation of methyl (2S)-2-(2-methyl-6-nitro-phenoxy)propanoate.
- 2-methyl-6-nitro-phenol 10 g, 65 mmol
- triphenyl- phosphine 22 g, 85 mmol
- methyl (R)- lactate 9.4 mL, 98 mmol
- the reaction mixture was stirred at room temperature overnight and then concentrated under reduced pressure.
- Step D Preparation of 6-(3-isopropyl-1,2,4-triazol-1-yl)pyrimidine-4-carboxylic acid
- methyl 6-chloropyrimidine-4-carboxylate 250 mg, 1.45 mmol
- N,N-dimethylformamide 10 mL
- 3-isopropyl-1H-1,2,4-triazole 161 mg, 1.45 mmol
- potassium carbonate 401 mg, 2.90 mmol
- Step E Preparation of [(2S)-2,3-dihydro-2,8-dimethyl-4H-1,4-benzoxazin-4-yl][6-[3- (1-methylethyl)-1H-1,2,4-triazol-1-yl]-4-pyrimidinyl]methanone (alternatively named [(2S)-2,8-dimethyl-2,3-dihydro-1,4-benzoxazin-4-yl]-[6- (3-isopropyl-1,2,4-triazol-1-yl)pyrimidin-4-yl]methanone).
- Step A Preparation of 2-(3-isopropyl-1,2,4-triazol-1-yl)pyrimidine-4-carboxylic acid
- methyl 2-chloropyrimidine-4-carboxylate 250 mg, 1.45 mmol
- N,N-dimethylformamide 10 mL
- 3-isopropyl-1H-1,2,4-triazole 161 mg, 1.45 mmol
- potassium carbonate 401 mg, 2.90 mmol
- Step B Preparation of [(2S)-2,3-dihydro-2,8-dimethyl-4H-1,4-benzoxazin-4-yl][2-[3- (1-methylethyl)-1H-1,2,4-triazol-1-yl]-4-pyrimidinyl]methanone (alternatively named [(2S)-2,8-dimethyl-2,3-dihydro-1,4-benzoxazin-4-yl]-[2- (3-isopropyl-1,2,4-triazol-1-yl)pyrimidin-4-yl]methanone).
- Step A Preparation of 6-(3-isopropyl-1,2,4-triazol-1-yl)pyrazine-2-carboxylic acid
- methyl 6-chloropyrazine-2-carboxylate 250 mg, 1.45 mmol
- N,N-dimethylformamide 10 mL
- 3-isopropyl-1H-1,2,4-triazole 161 mg, 1.45 mmol
- potassium carbonate 401 mg, 2.90 mmol
- Step B Preparation of [(2S)-2,3-dihydro-2,8-dimethyl-4H-1,4-benzoxazin-4-yl][6-[3- (1-methylethyl)-1H-1,2,4-triazol-1-yl]-2-pyrazinyl]methanone (alternatively named [(2S)-2,8-dimethyl-2,3-dihydro-1,4-benzoxazin-4-yl]-[6-(3-isopropyl- 1,2,4-triazol-1-yl)pyrazin-2-yl]methanone).
- Step A Preparation of 4-(3-isopropyl-1,2,4-triazol-1-yl)-2-methylsulfanyl- pyrimidine.
- 3-isopropyl-1H-1,2,4-triazole 332 mg, 2.99 mmol
- sodium hydride 60% dispersion in mineral oil, 2.7 mmol
- a solution of 4-chloro-2-methylsulfanyl- pyrimidine 400 mg, 2.49 mmol
- N,N-dimethylformamide (2 mL) was added dropwise to the mixture.
- Step B Preparation of 4-(3-isopropyl-1,2,4-triazol-1-yl)-2-methylsulfonyl-pyrimidine
- 4-(3-isopropyl-1,2,4-triazol-1-yl)-2-methylsulfanyl-pyrimidine i.e., the product of Step A
- chloroform 20 mL
- meta- chloroperbenzoic acid 4.2 g, 24.3 mmol
- Step C Preparation of 4-(3-isopropyl-1,2,4-triazol-1-yl)pyrimidin-2-ol.
- 4-(3-isopropyl-1,2,4-triazol-1-yl)-2-methylsulfonyl-pyrimidine i.e., the product of Step B
- 1 N aqueous sodium hydroxide solution 1.3 mL
- Step E Preparation of ethyl 4-(3-isopropyl-1,2,4-triazol-1-yl)pyrimidine-2- carboxylate.
- Step F Preparation of 4-(3-isopropyl-1,2,4-triazol-1-yl)pyrimidine-2-carboxylic acid
- ethyl 4-(3-isopropyl-1,2,4-triazol-1-yl)pyrimidine-2- carboxylate i.e., the product of Step E
- lithium hydroxide monohydrate 115 mg, 2.7 mmol
- Step G Preparation of [(2S)-2,3-dihydro-2,8-dimethyl-4H-1,4-benzoxazin-4-yl][4-[3- (1-methylethyl)-1H-1,2,4-triazol-1-yl]-2-pyrimidinyl]methanone (alternatively named [(2S)-2,8-dimethyl-2,3-dihydro-1,4-benzoxazin-4-yl]-[4- (3-isopropyl-1,2,4-triazol-1-yl)pyrimidin-2-yl]methanone).
- Step A Preparation of 3-chloro-5-(3-isopropyl-1,2,4-triazol-1-yl)pyridazine
- 3-isopropyl-1H-1,2,4-triazole 1.0 g, 9.0 mmol
- N,N-dimethylformamide 15 mL
- potassium carbonate 8. g, 20.3 mmol
- 3,5-dichloropyridazine 1.5 g, 10.1 mmol
- Step B Preparation of ethyl 5-(3-isopropyl-1,2,4-triazol-1-yl)pyridazine-3- carboxylate
- a solution of 3-chloro-5-(3-isopropyl-1,2,4-triazol-1-yl)pyridazine i.e., the product of Step A
- sodium carbonate 71 mg, 0.67 mmol
- Step C Preparation of 5-(3-isopropyl-1,2,4-triazol-1-yl)pyridazine-3-carboxylic acid
- ethyl 5-(3-isopropyl-1,2,4-triazol-1-yl)pyridazine-3- carboxylate i.e., the product of Step B
- lithium hydroxide monohydrate 160 mg, 3.8 mmol
- the volatiles were evaporated and the mixture was diluted with water and washed with a 10% solution of ethyl acetate in hexanes.
- the aqueous phase was acidified with a 1 N aqueous hydrochloric acid solution and extracted with a solution of 20% methanol in dichloromethane.
- the combined organic extracts were washed with brine, dried over anhydrous sodium sulfate and concentrated under reduced pressure.
- the resultant crude material was washed with pentane to afford the title compound as a pale brown solid (320 mg).
- Step D Preparation of [(2S)-2,3-dihydro-2,8-dimethyl-4H-1,4-benzoxazin-4-yl][5-[3- (1-methylethyl)-1H-1,2,4-triazol-1-yl]-3-pyridazinyl]methanone (alternatively named [(2S)-2,8-dimethyl-2,3-dihydro-1,4-benzoxazin-4-yl]-[5- (3-isopropyl-1,2,4-triazol-1-yl)pyridazin-3-yl]methanone).
- Step A Preparation of 3-anilino-2-methyl-propanoic acid
- iodobenzene (4 g, 19.6 mmol) in dimethylformamide (40 mL) and water (4 mL) was added 3-amino-2-methyl-propanoic acid (2.01 g, 19.5 mmol), copper(I) iodide (0.745 g, 3.91 mmol) and potassium carbonate (6.76 g, 48.9 mmol).
- the reaction mixture was stirred at 110 °C for 4 h.
- the mixture was diluted with water (100 mL), acidified to pH 2, extracted with ethyl acetate (2x).
- Step C Preparation of methyl 5-(3-isopropyl-1,2,4-triazol-1-yl)-2-methyl-benzoate
- a solution of methyl 5-bromo-2-methyl-benzoate (10 g, 44 mmol) in N,N-dimethylformamide (100 mL) was sparged with nitrogen gas then potassium carbonate (18.2 g, 132 mmol), 3-isopropyl-1H-1,2,4-triazole (7.30 g, 65.7 mmol), copper(I) iodide (1.67 g, 8.77 mmol) and trans-N,N′-dimethylcyclohexane-1,2-diamine (3.5 ml, 22 mmol) were added.
- Step D Preparation of 5-(3-isopropyl-1,2,4-triazol-1-yl)-2-methyl-benzoic acid
- methyl 5-(3-isopropyl-1,2,4-triazol-1-yl)-2-methyl-benzoate i.e., the product of Step C
- lithium hydroxide monohydrate 2.23 g, 53 mmol
- the reaction mixture was stirred at room temperature for 16 h.
- the mixture was concentrated under reduced, diluted with water (150 mL) and acidified to pH ⁇ 2 with 1 N aqueous hydrochloric acid solution.
- Step E Preparation of 2,3-dihydro-3-methyl-1-[2-methyl-5-[3-(1-methylethyl)-1H- 1,2,4-triazol-1-yl]benzoyl]-4(1H)-quinolinone (alternatively named 1-[5-(3- isopropyl-1,2,4-triazol-1-yl)-2-methyl-benzoyl]-3-methyl-2,3- dihydroquinolin-4-one).
- Step A Preparation of 5-chloro-3-methyl-3,4-dihydro-1H-quinoxalin-2-one
- 2-bromo-3-chloro-aniline 200 mg, 0.97 mmol
- 2-aminopropanoic acid 174 mg, 1.95 mmol
- tripotassium phosphate 413 mg, 1.95 mmol
- N,N′-dimethylethylenediamine 17.1 mg, 0.194 mmol
- copper(I) chloride (0.96 mg, 0.0097 mmol
- Step B Preparation of 8-chloro-2-methyl-1,2,3,4-tetrahydroquinoxaline
- 5-chloro-3-methyl-3,4-dihydro-1H-quinoxalin-2-one i.e., the product of Step A
- lithium aluminum hydride 2M in tetrahydrofuran, 5 equiv.
- Step C Preparation of (5-chloro-3,4-dihydro-3-methyl-1(2H)-quinoxalinyl)[2- methyl-5-[3-(1-methylethyl)-1H-1,2,4-triazol-1-yl]phenyl]methanone (alternatively named (5-chloro-3-methyl-3,4-dihydro-2H-quinoxalin-1-yl)-[5- (3-isopropyl-1,2,4-triazol-1-yl)-2-methyl-phenyl]methanone.
- the mixture was stirred at 100 °C for 16 h, cooled to 0 °C, quenched with saturated aqueous sodium bicarbonate solution, and extracted with ethyl acetate. The combined organic extracts were washed with water and brine, dried over anhydrous sodium sulfate and concentrated under reduced pressure.
- the crude material was purified by normal phase MPLC (eluting with a gradient of 0 to 30% ethyl acetate in hexanes) to afford the title compound (250 mg), a compound of the present invention.
- Step A Preparation of (2-amino-6-methyl-phenyl)methanol.
- 2-amino-6-methylbenzoic acid 10 g, 66 mmol
- tetrahydrofuran 200 mL
- lithium aluminum hydride 1M in tetrahydrofuran, 99 mL, 99 mmol
- the reaction mixture was stirred for 30 minutes, warmed to room temperature and then stirred for 16 h.
- the reaction was quenched using the Fieser method, filtered through a pad of Celite ® with tetrahydrofuran.
- Step F Preparation of [(3S)-3,4-dihydro-5-methyl-3-(trifluoromethyl)-1(2H)- quinolinyl][2-methoxy-5-[3-(1-methylethyl)-1H-1,2,4-triazol-1-yl]- phenyl]methanone and [(3R)-3,4-dihydro-5-methyl-3-(trifluoro- methyl)-1(2H)-quinolinyl][2-methoxy-5-[3-(1-methylethyl)-1H-1,2,4- triazol-1-yl]phenyl]methanone.
- Step A Preparation of (2-amino-6-fluorophenyl)methanol.
- 2-amino-6-fluorobenzoic acid 10 g, 64 mmol
- anhydrous tetrahydrofuran 200 mL
- lithium aluminum hydride 2M solution in tetrahydrofuran, 39 mL, 78 mmol
- Step C Preparation of methyl 5-fluoroquinoline-3-carboxylate.
- 2-amino-6-fluorobenzaldehyde i.e., the product of Step B
- toluene 30 mL
- methyl 3,3-dimethoxypropanoate 5.33 g, 36 mmol
- p-toluenesulfonic acid 248 mg
- a Dean-Stark apparatus and condenser were attached to the reaction container, and the reaction mixture was heated at the reflux temperature of the mixture for 7 h. The reaction mixture was then cooled and concentrated under reduced pressure. To the resultant material was added a saturated aqueous solution of sodium bicarbonate, and extracted with ethyl acetate. The organic extract was dried over anhydrous sodium sulfate, filtered, concentrated, and purified by silica gel column chromatography (eluting with a 7-8% solution of ethyl acetate in hexanes) to afford the title compound as an orange solid (600 mg).
- reaction mixture was heated at 80 °C for 6 h, quenched with a cold saturated aqueous solution of ammonium chloride, and extracted with ethyl acetate.
- the organic extract was washed with a saturated aqueous solution of sodium chloride, dried over anhydrous sodium sulfate, filtered, concentrated, and purified by silica gel column chromatography to afford [5-fluoro-3,4- dihydro-3-(methoxymethyl)-1(2H)-quinolinyl][2-methoxy-5-[3-(1-methylethyl)-1H-1,2,4- triazol-1-yl]phenyl]methanone.
- Step B Preparation of 8-fluoro-3,4-dihydro-4-[2-methyl-5-[3-(1-methylethyl)-1H- 1,2,4-triazol-1-yl]benzoyl]-2(1H)-quinoxalinone (Compound 47).
- the reaction mixture was concentrated under reduced pressure to remove to provide an acid chloride intermediate (i.e., 5-(3-isopropyl-1,2,4-triazol-1-yl)-2-methyl-benzoyl chloride), which was used without further purification.
- an acid chloride intermediate i.e., 5-(3-isopropyl-1,2,4-triazol-1-yl)-2-methyl-benzoyl chloride
- the reaction mixture was stirred at 25 °C for 16 h, diluted with water (100 mL) and extracted with ethyl acetate (2 x 100 mL). The combined organic extract was washed with a saturated aqueous solution of sodium chloride (50 mL), dried over sodium sulfate, filtered, concentrated, and purified by silica gel column chromatography (eluting with 40 to 50% ethyl acetate in hexanes). The resultant material was further purified by preparative high-performance liquid chromatography, to afford the title compound, a compound of the present invention, as an off-white solid (110 mg).
- t means tertiary, s means secondary, n means normal, i means iso, c means cyclo, Me means methyl, Et means ethyl, Pr means propyl, Bu means butyl, i-Pr means isopropyl, c-Pr means cyclopropyl, t-Bu means tertiary butyl, Ph means phenyl, OMe means methoxy, OEt means ethoxy, SMe means methylthio, -CN means cyano, -NO 2 means nitro, TMS means trimethylsilyl, SOMe means methylsulfinyl, C 2 F 5 means CF 2 CF 3 and SO 2 Me means methylsulfonyl.
- J-1 through J-25 have the following meanings as defined below in Exhibit 3.
- the bond projecting to the lower right is connected at the position designated in the tables (e.g., R 1a and R 3a ).
- R 1b is H, X 1 is CH, X 2 is CH, X 3 is C-OMe, R 3b is H, R 3a is Me, R 4a is Cl and (R 4b )q is H.
- R 1a R 1a R 1a Table 2 is constructed in the same manner as Table 1 above except that the Row Heading “R 1b is H, X 1 is CH, X 2 is CH, X 3 is C-OMe, R 3b is H, R 3a is Me, R 4a is Cl and (R 4b ) q is H.” is replaced with the Row Heading listed for Table 2 below (i.e., “R 1b is H, X 1 is CH, X 2 is CH, X 3 is CH, R 3b is H, R 3a is Me, R 4a is Cl and (R 4b ) q is H.”).
- Table 2 is a compound of Formula 1 wherein R 1b is H, X 1 is CH, X 2 is CH, X 3 is CH, R 3b is H, R 3a is Me, R 4a is Cl, (R 4b ) q is H and R 1a is H.
- Tables 3 through 104 are constructed similarly, except that the row heading in Table 1 is replaced with the respective row headings shown below.
- Table Row Heading 2 R 1b is H, X 1 is CH, X 2 is CH, X 3 is CH, R 3b is H, R 3a is Me, R 4a is Cl and (R 4b )q is H.
- R 1b is H, X 1 is CH, X 2 is CH, X 3 is C-F, R 3b is H, R 3a is Me, R 4a is Cl and (R 4b )q is H.
- 4 R 1b is H, X 1 is CH, X 2 is CH, X 3 is C-Cl, R 3b is H, R 3a is Me, R 4a is Cl and (R 4b )q is H.
- R 1b is H, X 1 is CH, X 2 is CH, X 3 is C-Me, R 3b is H, R 3a is Me, R 4a is Cl and (R 4b )q is H.
- R 1b is H, X 1 is CH, X 2 is CH, X 3 is C-Et, R 3b is H, R 3a is Me, R 4a is Cl and (R 4b )q is H.
- R 1b is H, X 1 is CH, X 2 is CH, X 3 is C-CH2C ⁇ CH, R 3b is H, R 3a is Me, R 4a is Cl and (R 4b )q is H.
- R 1b is H, X 1 is CH, X 2 is CH, X 3 is C-OCH2C ⁇ CH, R 3b is H, R 3a is Me, R 4a is Cl and (R 4b )q is H.
- R 1b is H, X 1 is CH, X 2 is CH, X 3 is C-c-Pr, R 3b is H, R 3a is Me, R 4a is Cl and (R 4b )q is H.
- Table Row Heading 1 03 R 1b is H, X 1 is CH, X 2 is CH, X 3 is C-Me, R 3b is H, R 3a is CF3, R 4a is Me and (R 4b )q is H.
- R 1b is H, X 1 is CH, X 2 is CH, X 3 is C-Me, R 3b is H, R 3a is CF3, R 4a is F and (R 4b )q is H.
- Table 105 is constructed the same as Table 1 except that the structure in Table 1 is replaced with the structure above for Table 105 wherein the row heading is X 2 is CH, X 3 is C-OMe, R 3a is Me, Z is CH(Me); and R 1a is as defined in Table 1.
- Table 106 through Table 282 Table 106 is constructed in the same manner as Table 105 except that the Row Heading “X 2 is CH, X 3 is C-OMe, R 3a is Me and Z is CH(Me).” is replaced with the Row Heading listed for Table 106 below (i.e., “X 2 is CH, X 3 is C-OMe, R 3a is Me and Z is C(Me) 2 .”). Therefore the first entry in Table 106 is a compound with the structure shown immediately above wherein X 2 is CH, X 3 is C-OMe, R 3a is Me and Z is C(Me) 2 and R 1a is H.
- Tables 107 through 282 are constructed similarly, except that the row heading in Table 105 is replaced with the respective row headings shown below.
- Table Row Heading 1 06 X 2 is CH, X 3 is C-OMe, R 3a is Me and Z is C(Me)2.
- 107 X 2 is CH, X 3 is C-OMe, R 3a is Me and Z is CH(F).
- 108 X 2 is CH, X 3 is C-OMe, R 3a is Me and Z is CF2.
- 109 X 2 is CH, X 3 is C-OMe, R 3a is Me and Z is CH(OH).
- 110 X 2 is CH, X 3 is C-OMe, R 3a is Me and Z is CH(CF3).
- 111 X 2 is CH, X 3 is C-OMe, R 3a is Me and Z is CH(CH2OMe).
- 112 X 2 is CH, X 3 is C-OMe, R 3a is Me and Z is CH(OMe).
- 113 X 2 is CH, X 3 is C-OMe, R 3a is Me and Z is C(OMe)2.
- 114 X 2 is CH, X 3 is C-OMe, R 3a is Me and Z is CH(OEt).
- 115 X 2 is CH, X 3 is C-OMe, R 3a is Me and Z is CH(OCF3).
- Table Row Heading 1 16 X 2 is CH, X 3 is C-OMe, R 3a is Me and Z is CH(OCF2H).
- 117 X 2 is CH, X 3 is C-OMe, R 3a is Me and Z is CH(OCFH2).
- 118 X 2 is CH, X 3 is C-OMe, R 3a is Me and Z is CH(SMe).
- 119 X 2 is CH, X 3 is C-OMe, R 3a is Me and Z is C(SMe)2.
- 120 X 2 is CH, X 3 is C-OMe, R 3a is Me and Z is CH(SO2Me).
- 121 X 2 is CH, X 3 is C-OMe, R 3a is Me and Z is CH(CH2SMe).
- 122 X 2 is CH, X 3 is C-OMe, R 3a is Me and Z is CH(CH2SO2Me).
- 123 X 2 is CH, X 3 is C-OMe, R 3a is Me and Z is CH(CN).
- 124 X 2 is CH, X 3 is C-OMe, R 3a is Me and Z is CH(C ⁇ CH).
- 125 X 2 is CH, X 3 is C-OMe, R 3a is Me and Z is CH(SCF3).
- 126 X 2 is CH, X 3 is C-OMe, R 3a is Me and Z is CH(CH2SCF3).
- 127 X 2 is CH, X 3 is C-OMe, R 3a is Me and Z is CH(CH2CN).
- 128 X 2 is CH, X 3 is C-OMe, R 3a is Me and Z is CH(O-c-Pr).
- X 2 is CH, X 3 is C-OMe, R 3a is Me and Z is C(R 3c )2 wherein the two R 3c groups are taken together to form a 3-membered ring,.
- X 2 is CH, X 3 is C-OMe, R 3a is Me and Z is NH.
- 145 X 2 is CH, X 3 is C-OMe, R 3a is Me and Z is NSO2Me.
- 146 X 2 is CH, X 3 is C-OMe, R 3a is Me and Z is NSO2CF3.
- 147 X 2 is CH, X 3 is C-Me, R 3a is Me and Z is C(Me)2.
- 148 X 2 is CH, X 3 is C-Me, R 3a is Me and Z is CH(F).
- X 2 is CH, X 3 is C-Me, R 3a is Me and Z is CF2.
- Table Row Heading 1 50 X 2 is CH, X 3 is C-Me, R 3a is Me and Z is CH(OH).
- 151 X 2 is CH, X 3 is C-Me, R 3a is Me and Z is CH(CF3).
- 152 X 2 is CH, X 3 is C-Me, R 3a is Me and Z is CH(CH2OMe).
- 153 X 2 is CH, X 3 is C-Me, R 3a is Me and Z is CH(OMe).
- 154 X 2 is CH, X 3 is C-Me, R 3a is Me and Z is C(OMe)2.
- 155 X 2 is CH, X 3 is C-Me, R 3a is Me and Z is CH(OEt).
- 156 X 2 is CH, X 3 is C-Me, R 3a is Me and Z is CH(OCF3).
- 157 X 2 is CH, X 3 is C-Me, R 3a is Me and Z is CH(OCF2H).
- 158 X 2 is CH, X 3 is C-Me, R 3a is Me and Z is CH(OCFH2).
- 159 X 2 is CH, X 3 is C-Me, R 3a is Me and Z is CH(SMe).
- 160 X 2 is CH, X 3 is C-Me, R 3a is Me and Z is C(SMe)2.
- 161 X 2 is CH, X 3 is C-Me, R 3a is Me and Z is CH(SO2Me).
- 162 X 2 is CH, X 3 is C-Me, R 3a is Me and Z is CH(CH2SMe).
- 163 X 2 is CH, X 3 is C-Me, R 3a is Me and Z is CH(CH2SO2Me).
- 164 X 2 is CH, X 3 is C-Me, R 3a is Me and Z is CH(CN).
- 165 X 2 is CH, X 3 is C-Me, R 3a is Me and Z is CH(C ⁇ CH).
- 166 X 2 is CH, X 3 is C-Me, R 3a is Me and Z is CH(SCF3).
- 167 X 2 is CH, X 3 is C-Me, R 3a is Me and Z is CH(CH2SCF3).
- 168 X 2 is CH, X 3 is C-Me, R 3a is Me and Z is CH(CH2CN).
- 169 X 2 is CH, X 3 is C-Me, R 3a is Me and Z is CH(O-c-Pr).
- 179 X 2 and X 3 are CR 2 wherein the R 2 groups are taken together to form J-21, R 3a is M e and Z is C(Me) 2 .
- 1 80 X 2 and X 3 are CR 2 wherein the R 2 groups are taken together to form J-21, R 3a is Me and Z is CH(F).
- 1 81 X 2 and X 3 are CR 2 wherein the R 2 groups are taken together to form J-21, R 3a is M e and Z is CF 2 .
- Table Row Heading 1 82 X 2 and X 3 are CR 2 wherein the R 2 groups are taken together to form J-21, R 3a is Me and Z is CH(OH).
- 1 83 X 2 and X 3 are CR 2 wherein the R 2 groups are taken together to form J-21, R 3a is M e and Z is CH(CF 3 ).
- 1 84 X 2 and X 3 are CR 2 wherein the R 2 groups are taken together to form J-21, R 3a is M e and Z is CH(CH 2 OMe).
- 1 85 X 2 and X 3 are CR 2 wherein the R 2 groups are taken together to form J-21, R 3a is Me and Z is CH(OMe).
- 1 86 X 2 and X 3 are CR 2 wherein the R 2 groups are taken together to form J-21, R 3a is M e and Z is C(OMe) 2 .
- 1 87 X 2 and X 3 are CR 2 wherein the R 2 groups are taken together to form J-21, R 3a is Me and Z is CH(OEt).
- 1 88 X 2 and X 3 are CR 2 wherein the R 2 groups are taken together to form J-21, R 3a is M e and Z is CH(OCF 3 ).
- 1 89 X 2 and X 3 are CR 2 wherein the R 2 groups are taken together to form J-21, R 3a is M e and Z is CH(OCF 2 H).
- 1 90 X 2 and X 3 are CR 2 wherein the R 2 groups are taken together to form J-21, R 3a is M e and Z is CH(OCFH 2 ).
- 1 91 X 2 and X 3 are CR 2 wherein the R 2 groups are taken together to form J-21, R 3a is Me and Z is CH(SMe).
- 1 92 X 2 and X 3 are CR 2 wherein the R 2 groups are taken together to form J-21, R 3a is M e and Z is C(SMe) 2 .
- 1 93 X 2 and X 3 are CR 2 wherein the R 2 groups are taken together to form J-21, R 3a is M e and Z is CH(SO 2 Me).
- 1 94 X 2 and X 3 are CR 2 wherein the R 2 groups are taken together to form J-21, R 3a is M e and Z is CH(CH 2 SMe).
- 1 95 X 2 and X 3 are CR 2 wherein the R 2 groups are taken together to form J-21, R 3a is M e and Z is CH(CH 2 SO 2 Me).
- 1 96 X 2 and X 3 are CR 2 wherein the R 2 groups are taken together to form J-21, R 3a is Me and Z is CH(CN).
- 1 97 X 2 and X 3 are CR 2 wherein the R 2 groups are taken together to form J-21, R 3a is Me and Z is CH(C ⁇ CH).
- 1 98 X 2 and X 3 are CR 2 wherein the R 2 groups are taken together to form J-21, R 3a is M e and Z is CH(SCF 3 ).
- Table Row Heading 1 99 X 2 and X 3 are CR 2 wherein the R 2 groups are taken together to form J-21, R 3a is M e and Z is CH(CH 2 SCF 3 ).
- 2 00 X 2 and X 3 are CR 2 wherein the R 2 groups are taken together to form J-21, R 3a is M e and Z is CH(CH 2 CN).
- 2 01 X 2 and X 3 are CR 2 wherein the R 2 groups are taken together to form J-21, R 3a is Me and Z is CH(O-c-Pr).
- 2 11 X 2 is CH, X 3 is C-OMe, R 3a is CF3 and Z is C(Me)2.
- 212 X 2 is CH, X 3 is C-OMe, R 3a is CF3 and Z is CH(F).
- 213 X 2 is CH, X 3 is C-OMe, R 3a is CF3 and Z is CF2.
- 214 X 2 is CH, X 3 is C-OMe, R 3a is CF3 and Z is CH(OH).
- 215 X 2 is CH, X 3 is C-OMe, R 3a is CF3 and Z is CH(CF3).
- 216 X 2 is CH, X 3 is C-OMe, R 3a is CF3 and Z is CH(CH2OMe).
- 217 X 2 is CH, X 3 is C-OMe, R 3a is CF3 and Z is CH(OMe).
- Table 283 is constructed the same as Table 105 except that the structure in Table 105 is replaced with the structure above for Table 283.
- Table 284 through Table 460 Table 284 is constructed the same as Table 106 except that the structure in Table 284 is replaced with the structure above for Table 283.
- Tables 285 through 460 are constructed in the same fashion as Tables 107 through 282.
- Table 461 Table 461 is constructed the same as Table 105 except that the structure in Table 105 is replaced with the structure above for Table 461.
- Table 462 through Table 638 Table 462 is constructed the same as Table 106 except that the structure in Table 462 is replaced with the structure above for Table 461.
- Tables 463 through 638 are constructed in the same fashion as Tables 107 through 282.
- Table 639 is constructed the same as Table 105 except that the structure in Table 105 is eplaced with the structure above for Table 639.
- Table 640 through Table 816 Table 640 is constructed the same as Table 106 except that the structure in Table 640 is eplaced with the structure above for Table 639.
- Tables 642 through 816 are constructed in he same fashion as Tables 107 through 282.
- Table 817 A A Formulation/Utility A compound of this invention will generally be used as a herbicidal active ingredient in a composition, i.e., formulation, with at least one additional component selected from the group consisting of surfactants, solid diluents and liquid diluents, which serves as a carrier.
- the formulation or composition ingredients are selected to be consistent with the physical properties of the active ingredient, mode of application and environmental factors such as soil type, moisture and temperature.
- Useful formulations include both liquid and solid compositions.
- Liquid compositions include solutions (including emulsifiable concentrates), suspensions, emulsions (including microemulsions, oil-in -water emulsions, flowable concentrates and/or suspoemulsions) and the like, which optionally can be thickened into gels.
- the general types of aqueous liquid compositions are soluble concentrate, suspension concentrate, capsule suspension, concentrated emulsion, microemulsion, oil-in-water emulsion, flowable concentrate and suspo-emulsion.
- nonaqueous liquid compositions are emulsifiable concentrate, microemulsifiable concentrate, dispersible concentrate and oil dispersion.
- solid compositions are dusts, powders, granules, pellets, prills, pastilles, tablets, filled films (including seed coatings) and the like, which can be water-dispersible (“wettable”) or water-soluble. Films and coatings formed from film- forming solutions or flowable suspensions are particularly useful for seed treatment.
- Active ingredient can be (micro)encapsulated and further formed into a suspension or solid formulation; alternatively the entire formulation of active ingredient can be encapsulated (or “overcoated”). Encapsulation can control or delay release of the active ingredient.
- An emulsifiable granule combines the advantages of both an emulsifiable concentrate formulation and a dry granular formulation.
- High-strength compositions are primarily used as intermediates for further formulation.
- Sprayable formulations are typically extended in a suitable medium before spraying. Such liquid and solid formulations are formulated to be readily diluted in the spray medium, usually water, but occasionally another suitable medium like an aromatic or paraffinic hydrocarbon or vegetable oil.
- Spray volumes can range from about from about one to several thousand liters per hectare, but more typically are in the range from about ten to several hundred liters per hectare.
- Sprayable formulations can be tank mixed with water or another suitable medium for foliar treatment by aerial or ground application, or for application to the growing medium of the plant.
- Liquid and dry formulations can be metered directly into drip irrigation systems or metered into the furrow during planting.
- the formulations will typically contain effective amounts of active ingredient, diluent and surfactant within the following approximate ranges which add up to 100 percent by weight.
- Solid diluents include, for example, clays such as bentonite, montmorillonite, attapulgite and kaolin, gypsum, cellulose, titanium dioxide, zinc oxide, starch, dextrin, sugars (e.g., lactose, sucrose), silica, talc, mica, diatomaceous earth, urea, calcium carbonate, sodium carbonate and bicarbonate, and sodium sulfate.
- Liquid diluents include, for example, water, N,N-dimethylalkanamides (e.g., N,N-dimethylformamide), limonene, dimethyl sulfoxide, N-alkylpyrrolidones (e.g., N-methylpyrrolidinone), alkyl phosphates (e.g., triethyl phosphate), ethylene glycol, triethylene glycol, propylene glycol, dipropylene glycol, polypropylene glycol, propylene carbonate, butylene carbonate, paraffins (e.g., white mineral oils, normal paraffins, isoparaffins), alkylbenzenes, alkylnaphthalenes, glycerine, glycerol tria
- Liquid diluents also include glycerol esters of saturated and unsaturated fatty acids (typically C 6 –C 22 ), such as plant seed and fruit oils (e.g., oils of olive, castor, linseed, sesame, corn (maize), peanut, sunflower, grapeseed, safflower, cottonseed, soybean, rapeseed, coconut and palm kernel), animal-sourced fats (e.g., beef tallow, pork tallow, lard, cod liver oil, fish oil), and mixtures thereof.
- plant seed and fruit oils e.g., oils of olive, castor, linseed, sesame, corn (maize), peanut, sunflower, grapeseed, safflower, cottonseed, soybean, rapeseed, coconut and palm kernel
- animal-sourced fats e.g., beef tallow, pork tallow, lard, cod liver oil, fish oil
- Liquid diluents also include alkylated fatty acids (e.g., methylated, ethylated, butylated) wherein the fatty acids may be obtained by hydrolysis of glycerol esters from plant and animal sources, and can be purified by distillation. Typical liquid diluents are described in Marsden, Solvents Guide, 2nd Ed., Interscience, New York, 1950.
- the solid and liquid compositions of the present invention often include one or more surfactants. When added to a liquid, surfactants (also known as “surface-active agents”) generally modify, most often reduce, the surface tension of the liquid.
- surfactants can be useful as wetting agents, dispersants, emulsifiers or defoaming agents.
- surfactants can be classified as nonionic, anionic or cationic.
- Nonionic surfactants useful for the present compositions include, but are not limited to: alcohol alkoxylates such as alcohol alkoxylates based on natural and synthetic alcohols (which may be branched or linear) and prepared from the alcohols and ethylene oxide, propylene oxide, butylene oxide or mixtures thereof; amine ethoxylates, alkanolamides and ethoxylated alkanolamides; alkoxylated triglycerides such as ethoxylated soybean, castor and rapeseed oils; alkylphenol alkoxylates such as octylphenol ethoxylates, nonylphenol ethoxylates, dinonyl phenol ethoxylates and dodecyl phenol ethoxylates (prepared from the phenols and ethylene oxide, propylene oxide, butylene oxide or mixtures thereof); block polymers prepared from ethylene oxide or propylene oxide and reverse block polymers where the terminal blocks are prepared from propylene oxide
- Useful anionic surfactants include, but are not limited to: alkylaryl sulfonic acids and their salts; carboxylated alcohol or alkylphenol ethoxylates; diphenyl sulfonate derivatives; lignin and lignin derivatives such as lignosulfonates; maleic or succinic acids or their anhydrides; olefin sulfonates; phosphate esters such as phosphate esters of alcohol alkoxylates, phosphate esters of alkylphenol alkoxylates and phosphate esters of styryl phenol ethoxylates; protein-based surfactants; sarcosine derivatives; styryl phenol ether sulfate; sulfates and sulfonates of oils and fatty acids; sulfates and sulfonates of ethoxylated alkylphenols; sulfates of alcohols; sulfates of e
- Useful cationic surfactants include, but are not limited to: amides and ethoxylated amides; amines such as N-alkyl propanediamines, tripropylenetriamines and dipropylenetetramines, and ethoxylated amines, ethoxylated diamines and propoxylated amines (prepared from the amines and ethylene oxide, propylene oxide, butylene oxide or mixtures thereof); amine salts such as amine acetates and diamine salts; quaternary ammonium salts such as quaternary salts, ethoxylated quaternary salts and diquaternary salts; and amine oxides such as alkyldimethylamine oxides and bis-(2-hydroxyethyl)-alkylamine oxides.
- amines such as N-alkyl propanediamines, tripropylenetriamines and dipropylenetetramines, and ethoxylated amine
- Nonionic, anionic and cationic surfactants and their recommended uses are disclosed in a variety of published references including McCutcheon’s Emulsifiers and Detergents, annual American and International Editions published by McCutcheon’s Division, The Manufacturing Confectioner Publishing Co.; Sisely and Wood, Encyclopedia of Surface Active Agents, Chemical Publ. Co., Inc., New York, 1964; and A. S. Davidson and B. Milwidsky, Synthetic Detergents, Seventh Edition, John Wiley and Sons, New York, 1987.
- compositions of this invention may also contain formulation auxiliaries and additives, known to those skilled in the art as formulation aids (some of which may be considered to also function as solid diluents, liquid diluents or surfactants).
- formulation auxiliaries and additives may control: pH (buffers), foaming during processing (antifoams such polyorganosiloxanes), sedimentation of active ingredients (suspending agents), viscosity (thixotropic thickeners), in-container microbial growth (antimicrobials), product freezing (antifreezes), color (dyes/pigment dispersions), wash-off (film formers or stickers), evaporation (evaporation retardants), and other formulation attributes.
- Film formers include, for example, polyvinyl acetates, polyvinyl acetate copolymers, polyvinylpyrrolidone-vinyl acetate copolymer, polyvinyl alcohols, polyvinyl alcohol copolymers and waxes.
- formulation auxiliaries and additives include those listed in McCutcheon’s Volume 2: Functional Materials, annual International and North American editions published by McCutcheon’s Division, The Manufacturing Confectioner Publishing Co.; and PCT Publication WO 03/024222.
- the compound of Formula 1 and any other active ingredients are typically incorporated into the present compositions by dissolving the active ingredient in a solvent or by grinding in a liquid or dry diluent.
- Solutions including emulsifiable concentrates, can be prepared by simply mixing the ingredients. If the solvent of a liquid composition intended for use as an emulsifiable concentrate is water-immiscible, an emulsifier is typically added to emulsify the active-containing solvent upon dilution with water. Active ingredient slurries, with particle diameters of up to 2,000 ⁇ m can be wet milled using media mills to obtain particles with average diameters below 3 ⁇ m. Aqueous slurries can be made into finished suspension concentrates (see, for example, U.S.3,060,084) or further processed by spray drying to form water-dispersible granules.
- Dusts and powders can be prepared by blending and usually grinding (such as with a hammer mill or fluid-energy mill).
- Granules and pellets can be prepared by spraying the active material upon preformed granular carriers or by agglomeration techniques. See Browning, “Agglomeration”, Chemical Engineering, December 4, 1967, pp 147–48, Perry’s Chemical Engineer’s Handbook, 4th Ed., McGraw-Hill, New York, 1963, pages 8–57 and following, and WO 91/13546.
- Pellets can be prepared as described in U.S.4,172,714.
- Water-dispersible and water-soluble granules can be prepared as taught in U.S. 4,144,050, U.S. 3,920,442 and DE 3,246,493. Tablets can be prepared as taught in U.S. 5,180,587, U.S. 5,232,701 and U.S. 5,208,030. Films can be prepared as taught in GB 2,095,558 and U.S.3,299,566.
- T. S. Woods “The Formulator’s Toolbox–Product Forms for Modern Agriculture” in Pesticide Chemistry and Bioscience, The Food–Environment Challenge, T. Brooks and T. R.
- Example A High Strength Concentrate Compound 1 98.5% silica aerogel 0.5% synthetic amorphous fine silica 1.0%
- Example B Wettable Powder Compound 1 65.0% dodecylphenol polyethylene glycol ether 2.0% sodium ligninsulfonate 4.0% sodium silicoaluminate 6.0% montmorillonite (calcined) 23.0%
- Example C ranule Compound 1 10.0% attapulgite granules (low volatile matter, 0.71/0.30 mm; 90.0% U.S.S.
- Example D Extruded Pellet Compound 1 25.0% anhydrous sodium sulfate 10.0% crude calcium ligninsulfonate 5.0% sodium alkylnaphthalenesulfonate 1.0% calcium/magnesium bentonite 59.0%
- Example E Emulsifiable Concentrate Compound 1 10.0% polyoxyethylene sorbitol hexoleate 20.0% C 6 –C 10 fatty acid methyl ester 70.0%
- Example F Microemulsion Compound 1 5.0% polyvinylpyrrolidone-vinyl acetate copolymer 30.0% alkylpolyglycoside 30.0% glyceryl monooleate 15.0% water 20.0%
- Example G Suspension Concentrate Compound 1 35% butyl polyoxyethylene/polypropylene block copolymer 4.0% stearic acid/polyethylene glycol copolymer 1.0% styrene acrylic polymer 1.0% xanthan gum 0.1% propylene glycol
- the compounds of the inention generally show highest activity for postemergence weed control (i.e., applied after weed seedlings emerge from the soil) and preemergence weed control (i.e., applied before weed seedlings emerge from the soil).
- postemergence weed control i.e., applied after weed seedlings emerge from the soil
- preemergence weed control i.e., applied before weed seedlings emerge from the soil.
- Many of them have utility for broad-spectrum pre- and/or postemergence weed control in areas where complete control of all vegetation is desired such as around fuel storage tanks, industrial storage areas, parking lots, drive-in theaters, air fields, river banks, irrigation and other waterways, around billboards and highway and railroad structures.
- Compounds of this invention may show tolerance to important agronomic crops including, but is not limited to, alfalfa, barley, cotton, wheat, rape, sugar beets, corn (maize), sorghum, soybeans, rice, oats, peanuts, vegetables, tomato, potato, perennial plantation crops including coffee, cocoa, oil palm, rubber, sugarcane, citrus, grapes, fruit trees, nut trees, banana, plantain, pineapple, hops, tea and forests such as eucalyptus and conifers (e.g., loblolly pine), and turf species (e.g., Kentucky bluegrass, St. Augustine grass, Kentucky fescue and Bermuda grass).
- important agronomic crops including, but is not limited to, alfalfa, barley, cotton, wheat, rape, sugar beets, corn (maize), sorghum, soybeans, rice, oats, peanuts, vegetables, tomato, potato, perennial plantation crops including coffee, cocoa
- Compounds of this invention can be used in crops genetically transformed or bred to incorporate resistance to herbicides, express proteins toxic to invertebrate pests (such as Bacillus thuringiensis toxin), and/or express other useful traits. Those skilled in the art will appreciate that not all compounds are equally effective against all weeds. Alternatively, the subject compounds are useful to modify plant growth.
- the compounds of the invention have both preemergent and postemergent herbicidal activity, to control undesired vegetation by killing or injuring the vegetation or reducing its growth
- the compounds can be usefully applied by a variety of methods involving contacting a herbicidally effective amount of a compound of the invention, or a composition comprising said compound and at least one of a surfactant, a solid diluent or a liquid diluent, to the foliage or other part of the undesired vegetation or to the environment of the undesired vegetation such as the soil or water in which the undesired vegetation is growing or which surrounds the seed or other propagule of the undesired vegetation.
- Undesired vegetation includes at least one selected from the group consisting of grass weeds and broadleaf weeds.
- Undesired vegetation is selected from the group consisting of annual bluegrass, Benghal dayflower, blackgrass, black nightshade, broadleaf signalgrass, Canada thistle, cheat, common cocklebur (Xanthium pensylvanicum), common ragweed, corn poppies, field violet, giant foxtail, goosegrass, green foxtail, guinea grass, hairy beggarticks, herbicide-resistant black grass, horseweed, Italian rye grass, jimsonweed, Johnson grass (Sorghum halepense), large crabgrass, little seed canary grass, morning glory, Pennsylvania smartweed, pitted morning glory, prickly sida, quackgrass, redroot pigweed, shattercane, shepherd's purse, silky windgrass, sunflower (as weed in potato), wild buckwheat (Polygonum convolvulus), wild mustard (Brass
- a herbicidally effective amount of the compounds of this invention is determined by a number of factors. These factors include: formulation selected, method of application, amount and type of vegetation present, growing conditions, etc. In general, a herbicidally effective amount of compounds of this invention is about 0.001 to 20 kg/ha with a preferred range of about 0.004 to 1 kg/ha. One skilled in the art can easily determine the herbicidally effective amount necessary for the desired level of weed control.
- a compound of the invention is applied, typically in a formulated composition, to a locus comprising desired vegetation (e.g., crops) and undesired vegetation (i.e., weeds), both of which may be seeds, seedlings and/or larger plants, in contact with a growth medium (e.g., soil).
- desired vegetation e.g., crops
- undesired vegetation i.e., weeds
- a growth medium e.g., soil
- a composition comprising a compound of the invention can be directly applied to a plant or a part thereof, particularly of the undesired vegetation, and/or to the growth medium in contact with the plant.
- Plant varieties and cultivars of the desired vegetation in the locus treated with a compound of the invention can be obtained by conventional propagation and breeding methods or by genetic engineering methods.
- transgenic plants are those in which a heterologous gene (transgene) has been stably integrated into the plant's genome.
- a transgene that is defined by its particular location in the plant genome is called a transformation or transgenic event.
- Genetically modified plant cultivars in the locus which can be treated according to the invention include those that are resistant against one or more biotic stresses (pests such as nematodes, insects, mites, fungi, etc.) or abiotic stresses (drought, cold temperature, soil salinity, etc.), or that contain other desirable characteristics. Plants can be genetically modified to exhibit traits of, for example, herbicide tolerance, insect-resistance, modified oil profiles or drought tolerance.
- Exhibit C Useful genetically modified plants containing single gene transformation events or combinations of transformation events are listed in Exhibit C. Additional information for the genetic modifications listed in Exhibit C can be obtained from publicly available databases maintained, for example, by the U.S. Department of Agriculture. The following abbreviations, T1 through T37, are used in Exhibit C for traits. A “-“ means the entry is not available; “tol.” means “tolerance” and “res.” means resistance.
- compounds of the invention are used to control undesired vegetation
- contact of desired vegetation in the treated locus with compounds of the invention may result in super-additive or enhanced effects with genetic traits in the desired vegetation, including traits incorporated through genetic modification.
- traits incorporated through genetic modification For example, resistance to phytophagous insect pests or plant diseases, tolerance to biotic/abiotic stresses or storage stability may be greater than expected from the genetic traits in the desired vegetation.
- Compounds of this invention can also be mixed with one or more other biologically active compounds or agents including herbicides, herbicide safeners, fungicides, insecticides, nematocides, bactericides, acaricides, growth regulators such as insect molting inhibitors and rooting stimulants, chemosterilants, semiochemicals, repellents, attractants, pheromones, feeding stimulants, plant nutrients, other biologically active compounds or entomopathogenic bacteria, virus or fungi to form a multi-component pesticide giving an even broader spectrum of agricultural protection.
- Mixtures of the compounds of the invention with other herbicides can broaden the spectrum of activity against additional weed species, and suppress the proliferation of any resistant biotypes.
- the present invention also pertains to a composition
- a composition comprising a compound of Formula 1 (in a herbicidally effective amount) and at least one additional biologically active compound or agent (in a biologically effective amount) and can further comprise at least one of a surfactant, a solid diluent or a liquid diluent.
- the other biologically active compounds or agents can be formulated in compositions comprising at least one of a surfactant, solid or liquid diluent.
- one or more other biologically active compounds or agents can be formulated together with a compound of Formula 1, to form a premix, or one or more other biologically active compounds or agents can be formulated separately from the compound of Formula 1, and the formulations combined together before application (e.g., in a spray tank) or, alternatively, applied in succession.
- a mixture of one or more of the following herbicides with a compound of this invention may be particularly useful for weed control: allidochlor, acetochlor, acifluorfen and its sodium salt, aclonifen, acrolein (2-propenal), alachlor, alloxydim, ametryn, amicarbazone, amidosulfuron, aminocyclopyrachlor and its esters (e.g., methyl, ethyl) and salts (e.g., sodium, potassium), 4-amino-3-chloro-5-fluoro-6-(7-fluoro-1H-indol-6-yl)-2-pyridinecarboxylic 2- propyn-1-yl ester (CAS No.2251111-17-6), 4-amino-3-chloro-5-fluoro-6-(7-fluoro-1H-indol- 6-yl)-2-pyridinecarboxylic cyanomethyl ester (CAS No.
- aminopyralid aminopyralid, amitrole, ammonium sulfamate, 2,5-anhydro-3,4-dideoxy-4-[[[(5S)-3-(3,5-difluorophenyl)-5- ethenyl-4,5-dihydro-5-isoxazolyl]carbonyl]amino]-threo-pentonic methyl ester (CAS No.
- anilofos anisiflupurin, asulam, atrazine, azimsulfuron, beflubutamid, beflubutamid-M, benazolin, benazolin-ethyl, bencarbazone, benfluralin, benfuresate, benquitrione, bensulfuron-methyl, bensulide, bentazone, benzobicyclon, benzofenap, bicyclopyrone, bifenox, bilanafos, bipyrazone, bispyribac and its sodium salt, bixlozone, broclozone, bromacil, bromobutide, bromofenoxim, bromoxynil, bromoxynil octanoate, butachlor, butafenacil, butamifos, butralin, butroxydim, butylate, cafenstrole, carbetamide, 1- (2-carboxyethyl)-4-(
- the compound of Formula 1 can also be mixed with a compound selected from the group consisting of N-[2,4-dimethyl-5-(1-piperidinylcarbonyl)phenyl]-1,1,1-trifluoromethanesulfonamide; N-[2-chloro-4-methyl-5-(4-morpholinylcarbonyl)phenyl]-1,1,1- trifluoromethanesulfonamide; N-[2,4-dimethyl-5-(4-morpholinylcarbonyl)phenyl]-1,1,1- trifluoromethanesulfonamide; N-[2-chloro-4-methyl-5-(1-piperidinylcarbonyl)phenyl]-1,1,1- trifluoromethanesulfonamide; 3-fluoro-N,N,2,4-tetramethyl-5-[[(trifluoromethyl)sulfonyl]amino]benzamide; 1,1,1-trifluoro-N-[3-fluoro
- the compound of Formula 1 can also be mixed with a compound selected from the group consisting of [(2S)-8-chloro-2,3-dihydro-2-methyl-4H-1,4-benzoxazin-4-yl][2-methyl-5-[3-(1- methylethyl)-1H-1,2,4-triazol-1-yl]phenyl]methanone; [(2S)-8-chloro-2,3-dihydro-2-methyl-4H-1,4-benzoxazin-4-yl][5-[3-(1,1- dimethylethyl)-1H-1,2,4-triazol-1-yl]-2-methylphenyl]methanone; [(2S)-8-chloro-2,3-dihydro-2-methyl-4H-1,4-benzoxazin-4-yl][2-methoxy-5-[3- (1-methylethyl)-1H-1,2,4-triazol-1-yl]phenyl]methan
- the compound of Formula 1 can also be mixed with a compound selected from the group consisting of 2,2,2-trifluoroethyl (4S)-4-[[[(5S)-3-(3,5-difluorophenyl)-5-methyl-2-oxo-5- oxazolidinyl]carbonyl]amino]-1-cyclopentene-1-carboxylate; 2,2,2-trifluoroethyl (4S)-4-[[[(5R)-3-(3,5-difluorophenyl)-5-methyl-2-oxo-5- oxazolidinyl]carbonyl]amino]-1-cyclopentene-1-carboxylate; 2-(methylthio)ethyl (4S)-4-[[[(5S)-3-(3,5-difluorophenyl)-5-methyl-2-oxo-5- oxazolidinyl]carbonyl]amino]-1-cyclopentene-1-carboxylate;
- the compound of Formula 1 can also be mixed with a compound selected from the group consisting of 4-[(E)-(3-bromo-1-naphthalenyl)(methoxyimino)methyl]-5-hydroxy-2,6-dimethyl- 3(2H)-pyridazinone; 4-[(Z)-(3-bromo-1-naphthalenyl)(methoxyimino)methyl]-5-hydroxy-2,6-dimethyl- 3(2H)-pyridazinone; 4-[(E)-(3-bromo-1-naphthalenyl)[(2-propyn-1-yloxy)imino]methyl]-5-hydroxy-2,6- dimethyl-3(2H)-pyridazinone; 4-[(E)-(3-bromo-1-naphthalenyl)(ethoxyimino)methyl]-5-hydroxy-2,6-dimethyl-3(2H)- pyridazinone; 4-[(Z)-(4
- the compound of Formula 1 can also be mixed with a compound selected from the group consisting of 4-[[2-(4-fluorophenyl)-5-methyl-2H-1,2,3-triazol-4-yl]methyl]-2- (trifluoromethyl)pyridine; 4-[[2-(4-fluorophenyl)-5-methyl-2H-1,2,3-triazol-4-yl]oxy]-2- (trifluoromethyl)pyridine; 4-[[5-ethoxy-2-(4-fluorophenyl)-2H-1,2,3-triazol-4-yl]methyl]-2- (trifluoromethyl)pyridine; 4-[[5-methoxy-2-[4-(trifluoromethyl)phenyl]-2H-1,2,3-triazol-4-yl]methyl]-2- (trifluoromethyl)pyridine; 4-[[5-methyl-2-[4-(trifluoromethyl)phenyl]-2H-1,2,3-triazol-4-yl]methyl]-2
- herbicides also include bioherbicides such as Alternaria destruens Simmons, Colletotrichum gloeosporiodes (Penz.) Penz. & Sacc., Drechsiera monoceras (MTB-951), Myrothecium verrucaria (Albertini & Schweinitz) Ditmar: Fries, Phytophthora palmivora (Butl.) Butl. and Puccinia thlaspeos Schub.
- bioherbicides such as Alternaria destruens Simmons, Colletotrichum gloeosporiodes (Penz.) Penz. & Sacc., Drechsiera monoceras (MTB-951), Myrothecium verrucaria (Albertini & Schweinitz) Ditmar: Fries, Phytophthora palmivora (Butl.) Butl. and Puccinia thlaspeos Schub.
- Preferred for better control of undesired vegetation e.g., lower use rate such as from enhanced effects, broader spectrum of weeds controlled, or enhanced crop safety
- a herbicide selected from the group consisting of atrazine, azimsulfuron, S-beflubutamid, benzisothiazolinone, bixlozone, beflubutamid-M, carfentrazone, carfentrazone-ethyl, chlorimuron, chlorimuron-ethyl, clomazone, clopyralid potassium, cloransulam-methyl, 2- [(2,4-dichlorophenyl)methyl]-4,4-dimethyl-3-isoxazolidinone, 2-[(2,5-dichloro- phenyl)methyl]-4,4-dimethyl-3-isoxazolidinone, ethametsulfuron-methyl, florasul
- a herbicide selected from the group consisting of atrazine, azims
- Compounds of this invention can also be used in combination with plant growth regulators such as aviglycine, N-(phenylmethyl)-1H-purin-6-amine, epocholeone, gibberellic acid, gibberellin A 4 and A 7 , harpin protein, mepiquat chloride, prohexadione calcium, prohydrojasmon, sodium nitrophenolate and trinexapac-methyl, and plant growth modifying organisms such as Bacillus cereus strain BP01. Compounds of this invention can also be used in combination with pheromones.
- plant growth regulators such as aviglycine, N-(phenylmethyl)-1H-purin-6-amine, epocholeone, gibberellic acid, gibberellin A 4 and A 7 , harpin protein, mepiquat chloride, prohexadione calcium, prohydrojasmon, sodium nitrophenolate and trinexapac-methyl, and plant growth modifying organisms such as Bacill
- Examples of pheromones include Formaldehyde; 2,2-Dibromoacetaldehyde; Acetaldehyde; 2- Methyl-2-propenal; 2-Methylpropanal; 2-Propenal; 3,3-Dibromo-2-propenal; Propanal; 2- Butenal; 2-Methyl-2-butenal; 2-Methylbutanal; 2-Methylenebutanal; 3-Methyl-2-butenal; 3- Methyl-3-butenal; 3-Methylbutanal; Butanal; (E)-2-Pentenal; 2-Methylenepentanal; 2- Pentenal; 3-Methyl-1-(vinyloxy)-butane; 4-Methylpentanal; 4-Pentenal; 5-Methylfurfural; Furan-2-carbaldehyde; Pentanal; (E)-2-Hexenal; (E)-2-Methyl-2-hexenal
- pheromones include (Z)-5-decenyl acetate, dodecanyl acetate, (Z)-7-dodecenyl acetate, (E)-7-dodecenyl acetate, (Z)-8-dodecenyl acetate, (E)-8-dodecenyl acetate, (Z)-9-dodecenyl acetate, (E)-9-dodecenyl acetate, (E)-10-dodecenyl acetate, 11- dodecenyl acetate, (Z)-9,11-dodecadienyl acetate, (E)-9,11- dodecadienyl acetate, (Z)-11- tridecenyl acetate, (E)-11-tridecenyl acetate, tetradecanyl acetate, (E)-7-tetradecenyl acetate, (Z)-5-de
- pheromones include citral; geranial; neral; tetradecan-1-al; pentadecan-1-al; pentadecen-1-al; hexadecan-1-al; (Z)-9-hexadecen-1-al; (Z)-11-hexadecen- 1-al; (7E,9E)-undeca-7,9-dien-1-al; (11Z, 13Z)-hexadecadien-1-al; (9Z,12E)-tetradecadien-1- al; (8E,10E)-dodecadien-1-al; (11Z)-hexadecadien-1-al; (9Z)-tetradecen-1-al; 6,10-dimethyl- 5,9-undecadien-2-ol; (6E)-7,11-dimethyl-3-methylene-1,6,10-dodecatriene; [1S-(1a,2b,5 a
- active ingredients are often applied at an application rate between one-half and the full application rate specified on product labels for use of active ingredient alone. These amounts are listed in references such as The Pesticide Manual and The BioPesticide Manual.
- the weight ratio of these various mixing partners (in total) to the compound of Formula 1 is typically between about 1:3000 and about 3000:1. Of note are weight ratios between about 1:300 and about 300:1 (for example ratios between about 1:30 and about 30:1).
- One skilled in the art can easily determine through simple experimentation the biologically effective amounts of active ingredients necessary for the desired spectrum of biological activity. It will be evident that including these additional components may expand the spectrum of weeds controlled beyond the spectrum controlled by the compound of Formula 1 alone.
- combinations of a compound of this invention with other biologically active (particularly herbicidal) compounds or agents can result in a greater-than-additive (i.e., enhanced) effect on weeds and/or a less-than-additive effect (i.e., safening) on crops or other desirable plants.
- Reducing the quantity of active ingredients released in the environment while ensuring effective pest control is always desirable.
- Ability to use greater amounts of active ingredients to provide more effective weed control without excessive crop injury is also desirable.
- a combination of a compound of the invention with at least one other herbicidal active ingredient can be advantageous for increasing crop protection by reducing weed competition.
- a combination of a compound of the invention with at least one other herbicidal active ingredient can be advantageous for increasing crop protection by reducing weed competition.
- a combination of a compound of the invention with at least one other herbicidal active ingredient can be advantageous for increasing crop protection by reducing weed competition.
- a combination of a compound of the invention with at least one other herbicidal active ingredient has different site of action from the compound of the invention.
- a combination with at least one other herbicidal active ingredient having a similar spectrum of control but a different site of action will be particularly advantageous for resistance management.
- a composition of the present invention can further comprise (in a herbicidally effective amount) at least one additional herbicidal active ingredient having a similar spectrum of control but a different site of action.
- herbicide safeners such as allidochlor, benoxacor, cloquintocet-mexyl, cumyluron, cyometrinil, cyprosulfonamide, daimuron, dichlormid, dicyclonon, dietholate, dimepiperate, fenchlorazole-ethyl, fenclorim, flurazole, fluxofenim, furilazole, isoxadifen-ethyl, mefenpyr- diethyl, mephenate, methoxyphenone naphthalic anhydride (1,8-naphthalic anhydride), oxabetrinil, N-(aminocarbonyl)-2-methylbenzenesulfonamide, N-(aminocarbonyl)- 2-fluorobenzenesulfonamide, 1-bromo-4-[(chloromethyl)sulfonyl]benzene (BC
- Antidotally effective amounts of the herbicide safeners can be applied at the same time as the compounds of this invention, or applied as seed treatments. Therefore an aspect of the present invention relates to a herbicidal mixture comprising a compound of this invention and an antidotally effective amount of a herbicide safener. Seed treatment is particularly useful for selective weed control, because it physically restricts antidoting to the crop plants. Therefore a particularly useful embodiment of the present invention is a method for selectively controlling the growth of undesired vegetation in a crop comprising contacting the locus of the crop with a herbicidally effective amount of a compound of this invention wherein seed from which the crop is grown is treated with an antidotally effective amount of safener.
- Antidotally effective amounts of safeners can be easily determined by one skilled in the art through simple experimentation.
- Compounds of the invention can also be mixed with: (1) polynucleotides including but not limited to DNA, RNA, and/or chemically modified nucleotides influencing the amount of a particular target through down regulation, interference, suppression or silencing of the genetically derived transcript that render a herbicidal effect; or (2) polynucleotides including but not limited to DNA, RNA, and/or chemically modified nucleotides influencing the amount of a particular target through down regulation, interference, suppression or silencing of the genetically derived transcript that render a safening effect.
- compositions comprising a compound of the invention (in a herbicidally effective amount), at least one additional active ingredient selected from the group consisting of other herbicides and herbicide safeners (in an effective amount), and at least one component selected from the group consisting of surfactants, solid diluents and liquid diluents.
- Table A1 lists specific combinations of a Component (a) with Component (b) illustrative of the mixtures, compositions and methods of the present invention.
- Compound No. (Compound Number) i.e., Compound 1 in the Component (a) column is identified in Index Table A.
- the second column of Table A1 lists the specific Component (b) compound (e.g., “2,4-D” in the first line).
- Table A1 lists ranges of weight ratios for rates at which the Component (a) compound is typically applied to a field-grown crop relative to Component (b) (i.e., (a):(b)).
- the first line of Table A1 specifically discloses the combination of Component (a) (i.e., Compound 1 in Index Table A) with 2,4-D is typically applied in a weight ratio between 1:384–6:1.
- the remaining lines of Table A1 are to be construed similarly.
- TABLE A1 Table A2 is constructed the same as Table A1 above except that those entries below the “Component (a)” column heading are replaced with the respective Component (a) Column Entry shown below.
- Preferred for better control of undesired vegetation e.g., lower use rate such as from enhanced effects, broader spectrum of weeds controlled, or enhanced crop safety
- a herbicide selected from the group consisting of bixlozone, beflubutamid-M, carfentrazone, carfentrazone-ethyl, chlorimuron, chlorimuron-ethyl, clomazone, florasulam, flupyrsulfuron, flupyrsulfuron-methyl, fluthiacet, fluthiacet-methyl, mesotrione, metolachlor, S-metolachlor, nicosulfuron, pinoxaden, pyroxasulfone, pyroxsulam, rimisoxafen, sulfentrazone, tembotrione, tetflupyrolimet, thifensulfuron, thif
- Mass spectra are reported with an estimated precision within ⁇ 0.5 Da as the molecular weight of the highest isotopic abundance positively charged parent ion (M+1) formed by addition of H + (molecular weight of 1) to the molecule having the highest isotopic abundance by using atmospheric pressure chemical ionization (AP+).
- AP+ atmospheric pressure chemical ionization
- the presence of molecular ions containing one or more higher atomic weight isotopes of lower abundance e.g., 37 Cl, 81 Br
- the compounds in Index Tables A, B and C are mixtures containing compounds of Formula 1' and 1''.
- plants selected from these crops and weed species and galium (catchweed bedstraw, Galium aparine) and horseweed (Erigeron canadensis) were planted in pots containing the same blend of loam soil and sand and treated with postemergence applications of test chemicals formulated in the same manner. Plants ranged in height from 2 to 10 cm and were in the one- to two-leaf stage for the postemergence treatment. Treated plants and untreated controls were maintained in a greenhouse for 10 days, after which time all treated, plants were compared to untreated controls and visually evaluated for injury. Plant response ratings, summarized in Table A, are based on a 0 to 100 scale where 0 is no effect and 100 is complete control. A dash (–) response means no test result.
- TEST B Plant species in the flooded paddy test selected from barnyardgrass (Echinochloa crus- galli), ducksalad (Heteranthera limosa), rice (Oryza sativa), and sedge, umbrella (small- flower umbrella sedge, Cyperus difformis) were grown to the 2-leaf stage for testing.
- test pots were flooded to 3 cm above the soil surface, treated by application of test compounds directly to the paddy water, and then maintained at that water depth for the duration of the test.
- Treated plants and controls were maintained in a greenhouse for 13 days, after which time all species were compared to controls and visually evaluated.
- Plant response ratings, summarized in Table B are based on a scale of 0 to 100 where 0 is no effect and 100 is complete control. A dash (–) response means no test result.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Life Sciences & Earth Sciences (AREA)
- Zoology (AREA)
- Pest Control & Pesticides (AREA)
- Plant Pathology (AREA)
- Engineering & Computer Science (AREA)
- Environmental Sciences (AREA)
- Wood Science & Technology (AREA)
- Health & Medical Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Dentistry (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Agronomy & Crop Science (AREA)
- Agricultural Chemicals And Associated Chemicals (AREA)
- Plural Heterocyclic Compounds (AREA)
Abstract
Disclosed are compounds of Formula 1, including all stereoisomers, N‑oxides, and salts thereof, agricultural compositions containing them and their use as herbicides wherein A is a 5‑ or 6‑membered heterocyclic ring, containing ring members selected from carbon atoms and up to 4 heteroatoms independently selected from up to 2 O, up to 2 S and up to 4 N atoms, one or two carbon or sulfur ring members of the heterocycle can optionally be in the oxidized form of a carbonyl, sulfonyl, sulfinyl moiety, said ring bound to the remainder of Formula 1 through a carbon atom or a heteroatom, and optionally substituted with 1 to 4 R1; and R1, R3a, R3b, R4a, R4b, X1, X2, X3, X4, Z, Y and q are as defined in the disclosure.
Description
TITLE SUBSTITUTED TETRAHYDROQUINOLINE AND TETRAHYDROQUINOXALINE HERBICIDES CROSS-REFERENCE TO RELATED APPLICATION This application claims the benefit of U.S. Provisional Application No.63/601264, filed November 21, 2023, all of which is incorporated by reference herein in its entirety. FIELD OF THE INVENTION This invention relates to certain tetrahydroquinoline and tetrahydroquinoxaline herbicides, their N-oxides, salts and compositions, and methods of their use for controlling undesirable vegetation. BACKGROUND OF THE INVENTION The control of undesired vegetation is extremely important in achieving high crop efficiency. Achievement of selective control of the growth of weeds especially in such useful crops as rice, soybean, sugar beet, maize, potato, wheat, barley, tomato and plantation crops, among others, is very desirable. Unchecked weed growth in such useful crops can cause significant reduction in productivity and thereby result in increased costs to the consumer. The control of undesired vegetation in noncrop areas is also important. Many products are commercially available for these purposes, but the need continues for new compounds that are more effective, less costly, less toxic, environmentally safer or have different sites of action. SUMMARY OF THE INVENTION This invention is directed to compounds of Formula 1, all stereoisomers, N-oxides, and salts thereof, agricultural compositions containing them and their use as herbicides:
1 wherein A is a 5- or 6-membered heterocyclic ring, containing ring members selected from carbon atoms and up to 4 heteroatoms independently selected from up to 2 oxygen atoms, up to 2 sulfur atoms and up to 4 nitrogen atoms, one or two carbon or sulfur ring members of the heterocycle can optionally be in the oxidized form of a carbonyl, thiocarbonyl, sulfonyl, sulfinyl moiety, said ring
bound to the remainder of Formula 1 through a carbon atom or a heteroatom, and optionally substituted with 1 to 4 R1; R1 is independently R1a, (R1b)m, R1c or any combination thereof; R1a is H, halogen, cyano, C1–C7 alkyl, C2–C7 alkenyl, C2–C6 alkynyl, C3–C7 cycloalkyl, C4–C7 cycloalkylalkyl, C4–C7 alkylcycloalkyl, C
C7 haloalkyl, C1– C6 hydroxyalkyl, C3–C7 hydroxycycloalkyl, C1–C6 hydroxyhaloalkyl, C4–C8 alkoxycycloalkyl, C2–C7 alkoxyhaloalkyl, C2–C6 oxacycloalkyl, C3–C7 oxacycloalkylalkyl, C3–C7 alkyloxacycloalkyl, C2–C6 thiacycloalkyl, C3–C7 thiacycloalkylalkyl, C3–C7 alkylthiacycloalkyl, C2–C6 (O-thia)cycloalkyl, C3– C7(O-thia)cycloalkylalkyl, C3–C7 alkyl(O-thia)cycloalkyl, C2–C6 (O2thia)cycloalkyl, C3–C7 (O2thia)cycloalkylalkyl, C3–C7 alkyl(O2thia)cycloalkyl, C2–C6 haloalkenyl, C2–C6 haloalkynyl, C3–C7 halocycloalkyl, C4–C7 haloalkylcycloalkyl, C2–C7 alkoxyalkyl, C2–C7 haloalkoxyalkyl, C1–C7 alkoxy, C2–C5 alkenyloxy, C2–C5 alkynyloxy, C3–C7 cycloalkoxy, C4–C7 cycloalkoxyalkyl, C
haloalkoxy, C1
7 alkylthio, C2– C7 alkylthioalkyl, C1–C5 alkylsulfinyl, C1–C5 alkylsulfonyl, C1–C5 alkylsulfonate, C1–C5 haloalkylthio, C1–C5 haloalkylsulfinyl, C1–C5 haloalkylsulfonyl, C2–C7 alkylsulfinylalkyl, C2–C7 alkylsulfonylalkyl, C2–C7 haloalkylthioalkyl, C2–C7 haloalkylsulfinylalkyl, C2–C7 haloalkylsulfonylalkyl, C4–C7 alkylthiocycloalkyl, C4–C7 alkylsulfinylcycloalkyl, C4–C7 alkylsulfonylcycloalkyl, C4–C7 haloalkylthiocycloalkyl, C4–C7 haloalkylsulfinylcycloalkyl, C4–C7 haloalkylsulfonylcycloalkyl, C2-C7 (HN=S(alkyl)=O)alkyl, C4-C7 (HN=S(alkyl)=O)cycloalkyl, C3-C7 (alkyl- N=S(alkyl)=O)alkyl, C5-C8 (alkyl-N=S(alkyl)=O)cycloalkyl, C3-C7 (cyano- N=S(alkyl)=O)alkyl, C4-C7 (alkylcarbonyl-N=S(alkyl)=O)alkyl, C4-C7 (alkoxycarbonyl-N=S(alkyl)=O)alkyl, C2–C5 cyanoalkyl, C4–C7 cyanocycloalkyl, C1–C4 nitroalkyl, C2–C6 dialkylamino, C2–C6 alkylcarbonyl, C3–C6 alkylcarbonylalkyl, C2–C6 alkoxycarbonyl, C3–C6 alkoxycarbonylalkyl or C3–C6 trialkylsilyl; each R1b is independently H, halogen, cyano, nitro, C1–C4 alkyl, C3–C5 cycloalkyl, C1–C4 haloalkyl, C3–C5 halocycloalkyl, C2–C4 alkoxyalkyl, C1–C4 alkoxy, C1– C4 alkylthio or C2–C4 alkoxycarbonyl; m is 0, 1 or 2; R1c is H, C1–C7 alkyl, C3–C7 cycloalkyl, C2–C6 oxacycloalkyl or C1–C7 haloalkyl; X1, X2, X3 and X4 are each independently N or CR2 wherein up to two X1, X2, X3 and X4 can be N; each R2 is independently H, halogen, cyano, nitro, hydroxy, C1–C5 alkyl, C2–C5 alkenyl, C2–C5 alkynyl, C3–C6 cycloalkyl, C4–C7 cycloalkylalkyl, C4–C7
alkylcycloalkyl, C1–C5 haloalkyl, C2–C5 haloalkenyl, C2–C5 haloalkynyl, C3– C6 halocycloalkyl, C4–C7 haloalkylcycloalkyl, C2–C5 alkoxyalkyl, C2–C5 haloalkoxyalkyl, C1–C5 alkoxy, C2–C5 alkenyloxy, C2–C5 alkynyloxy, C3–C7 cycloalkoxy, C4–C7 cycloalkoxyalkyl, C2–C6 oxacycloalkyl, C3–C7 oxacycloalkylalkyl, C2–C6 oxacycloalkoxy, C1–C5 haloalkoxy, C1–C5 alkylthio, C1–C4 alkylsulfinyl, C1–C4 alkylsulfonyl, C1–C4 alkylsulfonate, C3–C5 cycloalkylsulfonate, C1–C4 haloalkylsulfonate, C1–C4 haloalkylthio, C1–C4 haloalkylsulfinyl, C1–C4 haloalkylsulfonyl, C2–C7 alkylthioalkyl, C2–C7 alkylsulfinylalkyl, C2–C7 alkylsulfonylalkyl, C2–C7 haloalkylthioalkyl, C2–C7 haloalkylsulfinylalkyl, C2–C7 haloalkylsulfonylalkyl, C2–C5 cyanoalkyl, C4–C6 cyanocycloalkyl or C2–C5 alkoxycarbonyl; or two adjacent R2 may be taken together to form a saturated or unsaturated 5- to 8- membered ring, containing carbon atoms and optionally 1 to 3 oxygen, sulfur or nitrogen atoms as ring members, one or two carbon or sulfur ring members of the heterocycle can optionally be in the oxidized form of a carbonyl, sulfonyl, sulfinyl moiety, said ring unsubstituted or substituted with at least one substituent independently selected from the group consisting of halogen, cyano, nitro, C1–C4 alkyl, C3–C6 cycloalkyl, C1–C4 haloalkyl, C1–C4 alkoxy and C1– C4 haloalkoxy; Y is O or S; Z is O, C(R3c)p, C(=O), C(=S), or NR3d; R3a is halogen, cyano, nitro, C1–C7 alkyl, C2–C6 alkenyl, C2–C6 alkynyl, C3–C7 cycloalkyl, C1–C7 alkoxy, C1–C7 haloalkoxy, C4–C7 cycloalkylalkyl, C4–C7 alkylcycloalkyl, C1–C7 haloalkyl, C2–C6 haloalkenyl, C2–C6 haloalkynyl, C3– C7 halocycloalkyl, C4–C7 haloalkylcycloalkyl, C4–C7 cycloalkylhaloalkyl, C2– C7 alkoxyalkyl, C2–C7 haloalkoxyalkyl, C2–C5 alkenyloxy, C2–C5 alkynyloxy, C2–C5 alkenyloxyalkyl, C2–C5 alkynyloxyalkyl, C3–C7 cycloalkoxy, C4–C7 cycloalkoxyalkyl, C2–C6 oxacycloalkyl, C3–C7 oxacycloalkylalkyl, C2–C6 oxacycloalkoxy, C3–C6 oxacycloalkoxyalkyl, C1–C5 alkylthio, C1–C4 alkylsulfinyl, C1–C4 alkylsulfonyl, C1–C4 haloalkylthio, C1–C4 haloalkylsulfinyl, C1–C4 haloalkylsulfonyl, C2–C7 alkoxyhaloalkyl, C2–C7 haloalkoxyhaloalkyl, C2–C7 alkylthioalkyl, C2–C7 alkylsulfinylalkyl, C2–C7 alkylsulfonylalkyl, C2–C7 haloalkylthioalkyl, C2–C7 haloalkylsulfinylalkyl, C2– C7 haloalkylsulfonylalkyl, C2–C5 cyanoalkyl, C4–C6 cyanocycloalkyl, C1–C4 nitroalkyl, C3–C6 alkylcarbonylalkyl or C3–C6 alkoxycarbonylalkyl; each R3b is independently H, halogen or C1–C3 alkyl; or R3a and R3b are taken together with the carbon atom to which they are attached to form a 3- to 7-membered ring containing ring members selected from carbon
atoms and up to 3 heteroatoms independently selected from 1 oxygen atom, 1 sulfur atom, and up to 3 nitrogen atoms, wherein up to 2 carbon atom ring members are independently selected from C(=O) and C(=S) and the sulfur atom ring member is selected from S, S(O) or S(O)2; or R3a and R3b are taken together with the carbon atom to which they are attached to form C(=O), provided Z is NR3d and R3d is H; or two R3b are taken together with the carbon atom to which they are attached to form a 3- to 7-membered ring containing ring members selected from carbon atoms and up to 3 heteroatoms independently selected from 1 oxygen atom, 1 sulfur atom, and up to 3 nitrogen atoms, wherein up to 2 carbon atom ring members are independently selected from C(=O) and C(=S) and the sulfur atom ring member is selected from S, S(O) or S(O)2; each R3c is independently H, halogen, cyano, nitro, hydroxy, C1–C5 alkyl, C2–C5 alkenyl, C2–C5 alkynyl, C3–C5 cycloalkyl, C1–C5 haloalkyl, C2–C5 haloalkenyl, C2–C5 haloalkynyl, C2–C5 alkoxyalkyl, C2–C5 haloalkoxyalkyl, C1–C5 alkoxy, C1–C5 haloalkoxy, C1–C5 alkylthio, C1–C4 alkylsulfinyl, C1–C4 alkylsulfonyl, C1–C5 haloalkylthio, C1–C4 haloalkylsulfinyl, C1–C4 haloalkylsulfonyl, C4–C7 cycloalkylalkyl, C4–C7 alkylcycloalkyl, C3–C7 halocycloalkyl, C4–C7 haloalkylcycloalkyl, C2–C7 alkylthioalkyl, C2–C7 alkylsulfinylalkyl, C2–C7 alkylsulfonylalkyl, C2–C7 haloalkylthioalkyl, C2–C7 haloalkylsulfinylalkyl, C2– C7 haloalkylsulfonylalkyl, C2–C5 cyanoalkyl, C4–C6 cyanocycloalkyl, C1–C4 nitroalkyl, C2–C6 oxacycloalkyl, C3–C6 oxacycloalkylalkyl, C3–C7 cycloalkoxy, C4–C7 cycloalkoxyalkyl, C3–C6 alkylcarbonylalkyl, C3–C6 alkoxycarbonylalkyl or C2–C5 alkoxycarbonyl, C2–C6 alkylcarbonyloxy, C2–C6 haloalkylcarbonyloxy, C4–C6 cycloalkylcarbonyloxy, C2–C6 alkoxycarbonyloxy, C2–C6 alkylaminocarbonyloxy, C3–C6 dialkylaminocarbonyloxy; or one R3c is taken together with the adjacent R3a or R3b and the carbon atom to which they are attached to form a 3- to 7-membered ring containing ring members selected from carbon atoms and up to 3 heteroatoms independently selected from 1 oxygen atom, 1 sulfur atom, and up to 3 nitrogen atoms, wherein up to 2 carbon atom ring members are independently selected from C(=O) and C(=S) and the sulfur atom ring member is selected from S, S(O) or S(O)2; or two R3c are taken together with the carbon atom to which they are attached to form a 3- to 7-membered ring containing ring members selected from carbon atoms and up to 3 heteroatoms independently selected from 1 oxygen atom, 1 sulfur atom, and up to 3 nitrogen atoms, wherein up to 2 carbon atom ring members are
independently selected from C(=O) and C(=S) and the sulfur atom ring member is selected from S, S(O) or S(O)2; p is 0, 1 or 2; R3d is H, C1–C4 alkyl, C3–C6 cycloalkyl, C1–C4 haloalkyl, C2–C5 alkylcarbonyl, C2– C5 haloalkylcarbonyl, C2–C5 alkoxycarbonyl, C2–C5 alkylaminocarbonyl, C2– C5 dialkylaminocarbonyl, C1–C5 alkylsulfonyl or C1–C5 haloalkylsulfonyl; R4a is H, halogen, cyano, nitro, C1–C5 alkyl, C2–C5 alkenyl, C2–C5 alkynyl, C3–C5 cycloalkyl, C1–C5 haloalkyl, C2–C5 haloalkenyl, C2–C5 haloalkynyl, C2–C5 alkoxyalkyl, C2–C5 haloalkoxyalkyl, C1–C5 alkoxy, C1–C5 haloalkoxy, C3–C6 trialkylsilyl, C1–C5 alkylthio, C1–C5 haloalkylthio or C2–C5 alkoxycarbonyl; q is 0, 1 or 2; and each R4b is independently H, halogen, cyano, nitro, C1–C4 alkyl, C1–C4 haloalkyl, C1–C4 alkoxy or C1–C4 alkylthio; provided that: (a) when Z is O, then at least one of X3 and X4 is N; and (b) the compound of Formula 1 is other than: (3,4-dihydro-3-methyl-1(2H)-quinolinyl)[3-(1-pyrrolidinyl)phenyl]methanone (CAS Reg. No.2733036-55-8), (3,4-dihydro-3-methyl-1(2H)-quinolinyl)[3-(1H-pyrrol-1-yl)phenyl]methanone (CAS Reg. No.2305349-83-9), 1-[3-[(3,4-dihydro-3-methyl-1(2H)-quinolinyl)carbonyl]phenyl]-2-pyrrolidinone (CAS Reg. No.1329539-14-1), (3,4-dihydro-3-methoxy-1(2H)-quinolinyl)[5-(4-morpholinyl)-3-pyridinyl]- methanone (CAS Reg. No.1951747-01-5), (3,4-dihydro-3-methyl-1(2H)-quinolinyl)[5-(1H-1,2,3-triazol-1-yl)-3-pyridinyl]- methanone (CAS Reg. No.2361850-50-0), [3,4-dihydro-3-(1-methylethyl)-1(2H)-quinoxalinyl][6-(1-pyrrolidinyl)pyrazin-2- yl]methanone (CAS Reg. No.2462832-39-7), (3,4-dihydro-3-methoxy-1(2H)-quinolinyl)[5-(1H-1,2,3-triazol-1-yl)-3-pyridinyl]- methanone (CAS Reg. No.2224059-86-1), (3,4-dihydro-3-methyl-1(2H)-quinolinyl)[3-(tetrahydro-1,1-dioxido-2H-1,2-thiazin- 2-yl)phenyl]methanone (CAS Reg. No.1328130-66-0), [3,4-dihydro-3-(1-methylethyl)-1(2H)-quinoxalinyl][3-(1H-1,2,4-triazol-1-yl)- phenyl]methanone (CAS Reg. No.1436256-93-7), (3,4-dihydro-3-methyl-1(2H)-quinolinyl)[4-methoxy-3-(1H-tetrazol-1-yl)phenyl]- methanone (CAS Reg. No.1797746-04-3), [3,4-dihydro-3-(1-methylethyl)-1(2H)-quinoxalinyl][4-(1H-pyrazol-1-yl)-2- pyridinyl]methanone (CAS Reg. No.1436352-84-9),
[3,4-dihydro-3-(1-methylethyl)-1(2H)-quinoxalinyl][6-(1H-imidazol-1-yl)-2- pyridinyl]methanone (CAS Reg. No.1607303-56-9), 2-[3-[(3,4-dihydro-3-methyl-1(2H)-quinolinyl)carbonyl]phenyl]-3-isothiazolidinone 1,1-dioxide (CAS Reg. No.1326771-60-1), 2-[3-[(3,4-dihydro-3-methyl-1(2H)-quinolinyl)carbonyl]phenyl]-4-methyl-3- isothiazolidinone 1,1-dioxide (CAS Reg. No.1329431-16-4), ethyl 2-ethyl-3,4-dihydro-6,7-dimethyl-4-[3-(5-methyl-1,2,4-oxadiazol-3- yl)benzoyl]-1(2H)-quinoxalinecarboxylate (CAS Reg. No.769129-25-1), or ethyl 4-[3-(3,5-dimethyl-1H-pyrazol-1-yl)benzoyl]-2-ethyl-3,4-dihydro-6,7- dimethyl-1(2H)-quinoxalinecarboxylate (CAS Reg. No.769129-36-4). More particularly, this invention pertains to a compound of Formula 1 (including all stereoisomers), an N-oxide or a salt thereof. This invention also relates to a herbicidal composition comprising a compound of the invention (i.e., in a herbicidally effective amount) and at least one component selected from the group consisting of surfactants, solid diluents and liquid diluents. This invention further relates to a method for controlling the growth of undesired vegetation comprising contacting the vegetation or its environment with a herbicidally effective amount of a compound of the invention (e.g., as a composition described herein). This invention also includes a herbicidal mixture comprising (a) a compound selected from Formula 1, N-oxides, and salts thereof, and (b) at least one additional active ingredient selected from (b1) through (b18), and salts of compounds of (b1) through (b18), as described below. DETAILS OF THE INVENTION As used herein, the terms “comprises,” “comprising,” “includes,” “including,” “has,” “having,” “contains”, “containing,” “characterized by” or any other variation thereof, are intended to cover a non-exclusive inclusion, subject to any limitation explicitly indicated. For example, a composition, mixture, process or method that comprises a list of elements is not necessarily limited to only those elements but may include other elements not expressly listed or inherent to such composition, mixture, process or method. The transitional phrase “consisting of” excludes any element, step, or ingredient not specified. If in the claim, such would close the claim to the inclusion of materials other than those recited except for impurities ordinarily associated therewith. When the phrase “consisting of” appears in a clause of the body of a claim, rather than immediately following the preamble, it limits only the element set forth in that clause; other elements are not excluded from the claim as a whole. The transitional phrase “consisting essentially of” is used to define a composition, mixture, method or process that includes materials, steps, features, components, or elements,
in addition to those literally disclosed, provided that these additional materials, steps, features, components, or elements do not materially affect the basic and novel characteristic(s) of the claimed invention. The term “consisting essentially of” occupies a middle ground between “comprising” and “consisting of”. Where applicants have defined an invention or a portion thereof with an open-ended term such as “comprising,” it should be readily understood that (unless otherwise stated) the description should be interpreted to also describe such an invention using the terms “consisting essentially of” or “consisting of.” Further, unless expressly stated to the contrary, “or” refers to an inclusive or and not to an exclusive or. For example, a condition A or B is satisfied by any one of the following: A is true (or present) and B is false (or not present), A is false (or not present) and B is true (or present), and both A and B are true (or present). Also, the indefinite articles “a” and “an” preceding an element or component of the invention are intended to be nonrestrictive regarding the number of instances (i.e., occurrences) of the element or component. Therefore “a” or “an” should be read to include one or at least one, and the singular word form of the element or component also includes the plural unless the number is obviously meant to be singular. As referred to herein, the term “seedling”, used either alone or in a combination of words means a young plant developing from the embryo of a seed. As referred to herein, the term “broadleaf” used either alone or in words such as “broadleaf weed” means dicot or dicotyledon, a term used to describe a group of angiosperms characterized by embryos having two cotyledons. As used herein, the term “alkylating” refers reaction in which nucleophile displaces a leaving group such as halide or sulfonate from a carbon-containing radical. Unless otherwise indicated, the term “alkylating” does not limit the carbon-containing radical to alkyl. In the above recitations, the term “alkyl”, used either alone or in compound words such as “alkylthio” or “haloalkyl” includes straight-chain or branched alkyl, such as, methyl, ethyl, n-propyl, i-propyl, or the different butyl, pentyl, hexyl and heptyl isomers. “Alkenyl” includes straight-chain or branched alkenes such as ethenyl, 1-propenyl, 2-propenyl, and the different butenyl, pentenyl and hexenyl isomers. “Alkenyl” also includes polyenes such as 1,2-propadienyl and 2,4-hexadienyl. “Alkynyl” includes straight-chain or branched alkynes such as ethynyl, 1-propynyl, 2-propynyl and the different butynyl, pentynyl and hexynyl isomers. “Alkynyl” can also include moieties comprised of multiple triple bonds such as 2,5-hexadiynyl. “Alkoxy” includes, for example, methoxy, ethoxy, n-propyloxy, isopropyloxy and the different butoxy, pentoxy, hexyloxy and heptyloxy isomers. “Alkoxyalkyl” denotes alkoxy substitution on alkyl. Examples of “alkoxyalkyl” include CH3OCH2, CH3OCH2CH2, CH3CH2OCH2, CH3CH2CH2CH2OCH2 and CH3CH2OCH2CH2. “Alkoxyalkoxy” denotes
alkoxy substitution on alkoxy. “Alkoxycycloalkyl” denotes an alkoxy substitution on cycloalkyl. “Alkoxyhaloalkyl” denotes an alkoxy substitution on haloalkyl. Examples of “alkoxyhaloalkyl” include CH3OCF2- and CH3CH2OCCl2CH2-. The term “alkenyloxy” denotes an alkenyl moiety bonded through oxygen. Examples of “alkenyloxy” include CH2=CHCH2O-, 1-propenyloxy or CH3CH=CHO-, 2-butenyloxy or CH3CH=CHCH2O-, and the different butenyloxy, pentenyloxy and hexenyloxy isomers. Examples of “alkenyloxy” may also contain more than one double bond. The term “alkynyloxy” denotes an alkynyl moiety bonded through oxygen. Examples of “alkynyloxy” include HC≡CCH2O-, 1-propynyloxy or CH3C≡CO-, 2-butynyloxy or CH3C≡CCH2O-, and the different butynyloxy, pentynyloxy and hexynyloxy isomers. Examples of “alkynyloxy” may also contain more than one triple bond. “Hydroxyalkyl” denotes a hydroxy substitution on alkyl. “Hydroxycycloalkyl” denotes a hydroxy substitution on cycloalkyl. “Hydroxyhaloalkyl” denotes a hydroxy substitution on haloalkyl. Examples of “hydroxyalkyl”, “hydroxycycloalkyl”, “hydroxyhaloalkyl”, “alkoxycycloalkyl”, “alkoxyhaloalkyl” include the following structures: ,
2-OH-propan-2-yl or HOC(Me)2 hydroxymethyl or HOCH2 1-OH-cyclopropyl (hydroxyalkyl) (hydroxyalkyl) (hydroxycycloalkyl) .
2-OH-trifluoro-propan-2-yl 1-OMe-cyclopropyl 2-OMe-trifluoropropan-2-yl (hydroxyhaloalkyl) (alkoxycycloalkyl) (alkoxyhaloalkyl) “Alkylthio” includes branched or straight-chain alkylthio moieties such as methylthio, ethylthio, and the different propylthio, butylthio, pentylthio and hexylthio isomers. “Alkylthioalkyl” denotes alkylthio substitution on alkyl. Examples of “alkylthioalkyl” include CH3SCH2, CH3SCH2CH2, CH3CH2SCH2, CH3CH2CH2CH2SCH2, CH3CH2SCH2CH2 and their different isomers. “Alkylsulfinyl” includes both enantiomers of an alkylsulfinyl group. Examples of “alkylsulfinyl” include CH3S(O)-, CH3CH2S(O)-, CH3CH2CH2S(O)-, (CH3)2CHS(O)- and the different butylsulfinyl, pentylsulfinyl and hexylsulfinyl isomers. Examples of “alkylsulfonyl” include CH3S(O)2-, CH3CH2S(O)2-, CH3CH2CH2S(O)2-, (CH3)2CHS(O)2-, and the different butylsulfonyl, pentylsulfonyl and hexylsulfonyl isomers. Examples of “alkylsulfonate” include CH3S(O)2O-, CH3CH2S(O)2O-, CH3CH2CH2S(O)2O-, (CH3)2CHS(O)2O-, and the different butylsulfonate, pentylsulfonate and hexylsulfonate isomers.
“Cyanoalkyl” denotes an alkyl group substituted with one cyano group. Examples of “cyanoalkyl” include NCCH2 and NCCH2CH2 (alternatively identified as CH2CH2CN). “Nitroalkyl” denotes an alkyl group substituted with one nitro group. Examples of “nitroalkyl” include NO2CH2 and NO2CH2CH2 (alternatively identified as CH2CH2NO2). “Cyano” means NC- or N≡C-. The term “formyl” (or “CHO”) means HC(=O)-. “Alkylamino” denotes an NH radical substituted with straight-chain or branched alkyl. Examples of “alkylamino” include CH3CH2NH, CH3CH2CH2NH, and (CH3)2CHCH2NH. Examples of “dialkylamino” include (CH3)2N, (CH3CH2CH2)2N and CH3CH2(CH3)N. “Alkylsilyl” includes a silyl radical substituted with straight-chain or branched alkyl. “trialkylsilyl” includes a silyl radical substituted with three straight-chain or branched alkyl. Examples of “trialkylsilyl” include (CH3)3Si-, and (CH3CH2)3Si-. “Trialkylsilylalkynyl” denotes trialkylsilyl substitution on alkynyl. Examples of “trialkylsilylalkynyl” include (CH3)3SiC≡C-, and (CH3CH2)3SiC≡C-. “Cycloalkyl” includes, for example, cyclopropyl, cyclobutyl, cyclopentyl and cyclohexyl. The term “cycloalkylalkyl” denotes cycloalkyl substitution on an alkyl moiety. Examples of “cycloalkylalkyl” include cyclopropylmethyl, cyclopentylethyl, and other cycloalkyl moieties bonded to straight-chain or branched alkyl groups. The term “alkylcycloalkyl” denotes an alkyl group bonded to a cycloalkyl moiety and includes, for example, ethylcyclopropyl, i-propylcyclobutyl, methylcyclopentyl and methylcyclohexyl. The term “cycloalkoxy” denotes cycloalkyl group bonded through oxygen. Examples of “cycloalkoxy” include cyclopropoxy, cyclobutoxy, and cyclopentoxy. The term “cycloalkoxyalkyl” denotes cycloalkoxy substitution on an alkyl moiety. Examples of “cycloalkoxyalkyl” include cyclopropoxymethyl, cyclobutoxyethyl, and cyclopentoxymethyl, and other cycloalkoxy moieties bonded to straight-chain or branched alkyl groups. The term “alkoxycycloalkyl” denotes alkoxy substitution on cycloalkyl. The term “alkylthiocycloalkyl” denotes alkylthio substitution on cycloalkyl. The term “oxacycloalkyl” denotes a cycloalkyl with one carbon ring member replaced with an oxygen atom. Examples of “oxacycloalkyl” include oxacyclopropyl, oxacyclobutyl and oxacyclopentyl. The term “thiacycloalkyl” denotes a cycloalkyl with one carbon ring member replaced with a sulfur atom. Examples of “thiacycloalkyl” include thiacyclopropyl, thiacyclobutyl and thiacyclopentyl. The term “(O-thia)cycloalkyl” denotes a cycloalkyl with one carbon ring member replaced with a -SO group. Examples of “(O-thia)cycloalkyl” include (O-thia)cyclopropy, (O-thia)cyclobutyl and (O-thia)cyclopentyl. The term “(O2thia)cycloalkyl” denotes a cycloalkyl with one carbon ring member replaced with a -SO2 group. Examples of “(O2thia)cycloalkyl” include (O2thia)cyclopropy, (O2thia)cyclobutyl and (O2thia)cyclopentyl. The term “halogen”, either alone or in compound words such as “haloalkyl”, or when used in descriptions such as “alkyl substituted with halogen” includes fluorine, chlorine,
bromine or iodine. Further, when used in compound words such as “haloalkyl”, or when used in descriptions such as “alkyl substituted with halogen” said alkyl may be partially or fully substituted with halogen atoms which may be the same or different. Examples of “haloalkyl” or “alkyl substituted with halogen” include F 3 C, ClCH 2 , CF 3 CH 2 and CF 3 CCl 2 . The terms “haloalkoxy”, “haloalkoxyalkyl”, “haloalkylthio”, “haloalkenyl”, “haloalkynyl”, “halocycloalkyl”, “haloalkylcycloalkyl”, “haloalkylsulfinyl”, “haloalkylsulfonyl” and the like, are as defined analogously to the term “haloalkyl”. Examples of “haloalkoxy” include CF 3 O-, CCl 3 CH 2 O-, HCF 2 CH 2 CH 2 O- and CF 3 CH 2 O-. Examples of “haloalkoxyalkyl” include CF 3 OCH 2 -, CCl 3 CH 2 OCH 2 -, HCF 2 CH 2 CH 2 OCH 2 - and CF 3 CH 2 OCH 2 -. Examples of “haloalkylthio” include CCl 3 S-, CF 3 S-, CCl 3 CH 2 S- and ClCH 2 CH 2 CH 2 S-. Examples of “haloalkenyl” include (Cl) 2 C=CH- (Cl) 2 C=CHCH 2 - and CF 3 CH 2 CH=CHCH 2 -. Examples of “haloalkynyl” include HC≡CCHCl-, CF 3 C≡C-, CCl 3 C≡C- and FCH 2 C≡CCH 2 -. Examples of “halocycloalkyl” include 1-chlorocyclopropyl, 2-chlorocyclopropyl, 2-fluorocyclopropyl, 1-chlorocyclobutyl, 1-fluorocyclobutyl and 2-fluorocyclobutyl. Examples of “haloalkylcycloalkyl” include 1-(chloromethyl)cyclopropyl, 2-(chloromethyl)cyclopropyl, 2- (fluoromethyl)cyclopropyl, 1-(chloromethyl)cyclobutyl, 2-(fluoroethyl)cyclobutyl and 2- (fluoromethyl)cyclobutyl. Examples of “haloalkylsulfinyl” include CF3S(=O), CCl3S(=O), CF3CH2S(=O) and CF3CF2S(=O). Examples of “haloalkylsulfonyl” include CF3S(=O)2, CCl3S(=O)2, CF3CH2S(=O)2 and CF3CF2S(=O)2. “Alkylcarbonyl” denotes a straight-chain or branched alkyl moiety bonded to a C(=O) moiety. Examples of “alkylcarbonyl” include CH3C(=O)-, CH3CH2C(=O)-, CH3CH2CH2C(=O)-, (CH3)2CHC(=O)- and the different butyl- or pentylcarbonyl isomers. “Alkoxycarbonyl” denotes a straight-chain or branched alkoxy moieties bonded to a C(=O) moiety. Examples of “alkoxycarbonyl” include CH3OC(=O)-, CH3CH2OC(=O)-, CH3CH2CH2OC(=O)-, (CH3)2CHOC(=O)- and the different butoxy- or pentoxycarbonyl isomers. C(=O) or C(O) designates carbonyl. The term “alkoxycarbonylalkyl” denotes a straight-chain or branched alkoxycarbonyl moiety bonded through an alkyl moiety. The term “alkylcarbonylalkyl” denotes a straight or branched alkylcarbonyl moiety bonded through an alkyl moiety. The term “alkylcarbonyloxy” denotes an alkylcarbony moiety bonded through oxygen. Examples of alkylcarbonyloxy include CH3C(=O)O-, CH3CH2C(=O)O-, CH3CH2CH2C(=O)O- and (CH3)2CHC(=O)-. The term alkanediyl or alkenediyl refers to a linear or branched alkane or alkene linking chain respectively. Examples of alkanediyl include –CH2–, –CH2CH(CH3)– or -CH2CH2CH2–. Examples of alkenediyl include –CH=CH–, –CH2C=CH– or -CH=C(CH3)-. The term “adjacent” in the context of locating a substituent means “next to” or “immediately next to”.
“Alkylsulfoximinoalkyl” denotes an alkylsulfoximine or cycloalkylsulfoximine substitution on alkyl or cycloalkyl. Examples of “alkylsulfoximinoalkyl” include the following structures: , ,
(methyloxycarbonyl- (methylcarbonyl- N=S(CH3)=O)CH2 N=S(CH3)=O)CH2 The chemical abbreviations S(O) and S(=O) as used herein represent a sulfinyl moiety. The chemical abbreviations SO2, S(O)2 and S(=O)2 as used herein represent a sulfonyl moiety. The chemical abbreviations C(O) and C(=O) as used herein represent a carbonyl moiety. The chemical abbreviations CO2, C(O)O and C(=O)O as used herein represent an oxycarbonyl moiety. The total number of carbon atoms in a substituent group is indicated by the “Ci–Cj” prefix where i and j are numbers from 1 to 8. For example, C1–C4 alkylsulfonyl designates methylsulfonyl through butylsulfonyl; C3–C6 alkylcarbonylalkyl can be, for example, CH3COCH2-, CH3COCH2CH2- or CH3CH2COCH2CH2CH2-; C4–C7 alkylcycloalkyl can be, for example, methylcyclopropyl, methylcyclobutyl, ethylcyclopropyl, or propylcyclobutyl; C2 alkoxyalkyl designates CH3OCH2-; C3 alkoxyalkyl designates, for example, CH3CH(OCH3)- , CH3OCH2CH2- or CH3CH2OCH2-; and C4 alkoxyalkyl designates the various isomers of an alkyl group substituted with an alkoxy group containing a total of four carbon atoms, examples including CH3CH2CH2OCH2- and CH3CH2OCH2CH2-. A wavy line in a structure fragment denotes the attachment point of the fragment to the remainder of the molecule. For example, when the variables X2 and X3 in Formula 1 are defined as J-21 (i.e., X1 and X4 are C-H; X2 and X3 are C-R2; and the two adjacent R2
variables are taken together to form the ring), the wavy lines bisecting the bonds of J-21 in the position identified with asterisks (*) of the fused ring J-21 means that the fused ring J-21 is attached to the remainder of the structure of Formula 1 at said positions, as shown below.
A "-" at the beginning of a fragment definition denotes the attachment point of said fragment to the remainder of the molecule; for example, "-CH2CH2OMe" denotes the fragment 2-methoxyethyl. A group with two short lines at the ends means the group is bonded to other moieties of the molecule through the two atoms right next to the lines. Some examples of these groups are -OCH2O- or -O(CH2)2O- or -O(CH2)2-. Cyclic fragments are represented by the use of two "-" within parentheses; for example, the fragment 1-cyanocyclopropyl is represented by "-C(CN)(-CH2CH2-)", wherein a carbon atom is bonded to both terminal carbon atoms of the two-carbon chain, as illustrated below.
Naming of substituents in the present disclosure uses recognized terminology providing conciseness in precisely conveying to those skilled in the art the chemical structure. For conciseness, locant descriptors may be omitted. Compounds of this invention can exist as one or more stereoisomers. The various stereoisomers include enantiomers, diastereomers, cis-trans isomers or Z / E isomers (also known as geometric isomers) and atropisomers (also known as conformational isomers). Stereoisomers are isomers of identical constitution but differing in the arrangement of their atoms in space and include enantiomers, diastereomers, cis-trans isomers (also known as geometric isomers) and atropisomers. Atropisomers result from restricted rotation about single bonds where the rotational barrier is high enough to permit isolation of the isomeric species. One skilled in the art will appreciate that one stereoisomer may be more active and/or may exhibit beneficial effects when enriched relative to the other stereoisomer(s) or when separated from the other stereoisomer(s). Additionally, the skilled artisan knows how to separate, enrich, and/or to selectively prepare said stereoisomers. For a comprehensive discussion of all aspects of stereoisomerism, see Ernest L. Eliel and Samuel H. Wilen, Stereochemistry of Organic Compounds, John Wiley & Sons, 1994.
The compounds of the invention may be present as a mixture of stereoisomers, individual stereoisomers or as an optically active form. For example, two possible stereoisomers of Formula 1 are depicted as Formula 1' and 1'' involving the chiral center identified in the structures below with an asterisk (*). Compounds of Formula 1 can also comprise additional chiral centers. For example, substituents and other molecular constituents such as R2 may themselves contain chiral centers. This disclosure comprises racemic mixtures as well as enriched and essentially pure stereoconfigurations at these additional chiral centers.
Molecular depictions drawn herein follow standard conventions for depicting stereochemistry. To indicate stereoconfiguration, bonds rising from the plane of the drawing and towards the viewer are denoted by solid wedges wherein the broad end of the wedge is attached to the atom rising from the plane of the drawing towards the viewer. Bonds going below the plane of the drawing and away from the viewer are denoted by dashed wedges wherein the broad end of the wedge is attached to the atom further away from the viewer. The compounds of the disclosure can exist as stereoisomers due to the possible chiral carbon atoms present in Formula 1. Thus, this disclosure comprises the individual stereoisomers of the compounds of Formula 1, as well as mixtures of stereoisomers of the compounds of Formula 1. This disclosure comprises racemic mixtures of equal amounts of the enantiomers of Formulae 1' and 1''. In addition, this disclosure includes mixtures that are enriched in the Formula 1' enantiomer compared to the racemic mixture of Formulae 1' and 1''. This disclosure also comprises the essentially pure enantiomer of Formula 1'. An embodiment of this disclosure comprises mixtures of stereoisomers of the compounds of Formula 1' and Formula 1'', wherein the ratio of 1' to 1'' is at least 75:25 (a 50% enantiomeric excess of 1'). An embodiment of this disclosure comprises mixtures of stereoisomers of the compounds of Formula 1' and Formula 1'', wherein the ratio of 1' to 1'' is at least 90:10 (an 80% enantiomeric excess of 1'). An embodiment of this disclosure comprises mixtures of stereoisomers of the compounds of Formula 1' and Formula 1'', wherein the ratio of 1' to 1'' is at least 95:5 (a 90% enantiomeric excess of 1').
An embodiment of this disclosure comprises mixtures of stereoisomers of the compounds of Formula 1' and Formula 1'', wherein the ratio of 1' to 1'' is at least 98:2 (a 96% enantiomeric excess of 1'). An embodiment of this disclosure comprises mixtures of stereoisomers of the compounds of Formula 1' and Formula 1'', wherein the ratio of 1' to 1'' is at least 99:1 (a 98% enantiomeric excess of 1'). An embodiment of this disclosure comprises mixtures of stereoisomers of the compounds of Formula 1' and Formula 1'', wherein the ratio of 1' to 1'' is essentially 100:0. An embodiment of this disclosure comprises the compounds of Formula 1'. In addition, this disclosure includes mixtures that are enriched in the Formula 1'' enantiomer compared to the racemic mixture of Formulae 1'' and 1'. This disclosure also comprises the essentially pure enantiomer of Formula 1''. An embodiment of this disclosure comprises mixtures of stereoisomers of the compounds of Formula 1'' and Formula 1', wherein the ratio of 1'' to 1' is at least 75:25 (a 50% enantiomeric excess of 1''). An embodiment of this disclosure comprises mixtures of stereoisomers of the compounds of Formula 1'' and Formula 1', wherein the ratio of 1'' to 1' is at least 90:10 (an 80% enantiomeric excess of 1''). An embodiment of this disclosure comprises mixtures of stereoisomers of the compounds of Formula 1'' and Formula 1', wherein the ratio of 1'' to 1' is at least 95:5 (a 90% enantiomeric excess of 1''). An embodiment of this disclosure comprises mixtures of stereoisomers of the compounds of Formula 1'' and Formula 1', wherein the ratio of 1'' to 1' is at least 98:2 (a 96% enantiomeric excess of 1''). An embodiment of this disclosure comprises mixtures of stereoisomers of the compounds of Formula 1'' and Formula 1', wherein the ratio of 1'' to 1' is at least 99:1 (a 98% enantiomeric excess of 1''). An embodiment of this disclosure comprises mixtures of stereoisomers of the compounds of Formula 1'' and Formula 1', wherein the ratio of 1'' to 1' is essentially 100:0. An embodiment of this disclosure comprises the compounds of Formula 1''. Compounds of Formula 1 typically exist in more than one form, and Formula 1 thus include all crystalline and non-crystalline forms of the compounds they represent. Non- crystalline forms include embodiments which are solids such as waxes and gums as well as embodiments which are liquids such as solutions and melts. Crystalline forms include embodiments which represent essentially a single crystal type and embodiments which represent a mixture of polymorphs (i.e., different crystalline types). The term “polymorph” refers to a particular crystalline form of a chemical compound that can crystallize in different
crystalline forms, these forms having different arrangements and/or conformations of the molecules in the crystal lattice. Although polymorphs can have the same chemical composition, they can also differ in composition due to the presence or absence of co- crystallized water or other molecules, which can be weakly or strongly bound in the lattice. Polymorphs can differ in such chemical, physical and biological properties as crystal shape, density, hardness, color, chemical stability, melting point, hygroscopicity, suspensibility, dissolution rate and biological availability. One skilled in the art will appreciate that a polymorph of a compound of Formula 1 can exhibit beneficial effects (e.g., suitability for preparation of useful formulations, improved biological performance) relative to another polymorph or a mixture of polymorphs of the same compound of Formula 1. Preparation and isolation of a particular polymorph of a compound of Formula 1 can be achieved by methods known to those skilled in the art including, for example, crystallization using selected solvents and temperatures. For a comprehensive discussion of polymorphism see R. Hilfiker, Ed., Polymorphism in the Pharmaceutical Industry, Wiley-VCH, Weinheim, 2006. One skilled in the art will appreciate that not all nitrogen-containing heterocycles can form N-oxides since the nitrogen requires an available lone pair for oxidation to the oxide; one skilled in the art will recognize those nitrogen-containing heterocycles which can form N-oxides. One skilled in the art will also recognize that tertiary amines can form N-oxides. Synthetic methods for the preparation of N-oxides of heterocycles and tertiary amines are very well known by one skilled in the art including the oxidation of heterocycles and tertiary amines with peroxy acids such as peracetic and m-chloroperbenzoic acid (MCPBA), hydrogen peroxide, alkyl hydroperoxides such as t-butyl hydroperoxide, sodium perborate, and dioxiranes such as dimethyldioxirane. These methods for the preparation of N-oxides have been extensively described and reviewed in the literature, see for example: T. L. Gilchrist in Comprehensive Organic Synthesis, vol. 7, pp 748–750, S. V. Ley, Ed., Pergamon Press; M. Tisler and B. Stanovnik in Comprehensive Heterocyclic Chemistry, vol. 3, pp 18–20, A. J. Boulton and A. McKillop, Eds., Pergamon Press; M. R. Grimmett and B. R. T. Keene in Advances in Heterocyclic Chemistry, vol. 43, pp 149–161, A. R. Katritzky, Ed., Academic Press; M. Tisler and B. Stanovnik in Advances in Heterocyclic Chemistry, vol.9, pp 285–291, A. R. Katritzky and A. J. Boulton, Eds., Academic Press; and G. W. H. Cheeseman and E. S. G. Werstiuk in Advances in Heterocyclic Chemistry, vol. 22, pp 390–392, A. R. Katritzky and A. J. Boulton, Eds., Academic Press. One skilled in the art recognizes that because in the environment and under physiological conditions salts of chemical compounds are in equilibrium with their corresponding nonsalt forms, salts share the biological utility of the nonsalt forms. Thus, a wide variety of salts of a compound of Formula 1 are useful for control of undesired vegetation (i.e., are agriculturally suitable). The salts of a compound of Formula 1 include acid-addition salts with inorganic or organic acids such as hydrobromic, hydrochloric, nitric, phosphoric, sulfuric, acetic, butyric,
fumaric, lactic, maleic, malonic, oxalic, propionic, salicylic, tartaric, 4-toluenesulfonic or valeric acids. When a compound of Formula 1 contains an acidic moiety, salts also include those formed with organic or inorganic bases such as pyridine, triethylamine or ammonia, or amides, hydrides, hydroxides or carbonates of sodium, potassium, lithium, calcium, magnesium or barium. Accordingly, the present invention comprises compounds selected from Formula 1, N-oxides and agriculturally suitable salts thereof. Unless otherwise indicated, a “ring” as a component of Formula 1 is carbocyclic or heterocyclic. The term “ring member” refers to an atom or other moiety (e.g., C(=O), C(=S), S(O) or S(O)2) forming the backbone of a ring or ring system. The term “ring system” denotes two or more fused rings. The term “bicyclic ring” denotes a ring system consisting of two fused rings. The term “heterocyclic ring” denotes a ring in which at least one atom forming the ring backbone is not carbon, e.g., nitrogen, oxygen or sulfur. Typically a heterocyclic ring contains no more than 4 nitrogens, no more than 2 oxygens and no more than 2 sulfurs. Unless otherwise indicated, a heterocyclic ring can be a saturated, partially unsaturated, or fully unsaturated ring. When a fully unsaturated heterocyclic ring satisfies Hückel’s rule, then said ring is also called a “heteroaromatic ring” or “aromatic heterocyclic ring”. Unless otherwise indicated, heterocyclic rings can be attached through any available carbon or nitrogen by replacement of a hydrogen on said carbon or nitrogen. “Aromatic” indicates that each of the ring atoms is essentially in the same plane and has a p-orbital perpendicular to the ring plane, and that (4n + 2) π electrons, where n is a positive integer, are associated with the ring to comply with Hückel’s rule. The term “optionally substituted” in connection with a ring refers to groups which are unsubstituted or have at least one non-hydrogen substituent that does not extinguish the biological activity possessed by the unsubstituted analog. As used herein, the following definitions shall apply unless otherwise indicated. The term "optionally substituted" is used interchangeably with the phrase “substituted or unsubstituted” or with the term “(un)substituted.” Unless otherwise indicated, an optionally substituted group may have a substituent at each substitutable position of the group, and each substitution is independent of the other. When a group contains a substituent which can be hydrogen, for example R2 or R4a, then when this substituent is taken as hydrogen, it is recognized that this is equivalent to said group being unsubstituted. When a variable group is shown to be optionally attached to a position, for example (R4b)q in Formula 1 wherein q may be 0, then hydrogen may be at the remaining positions even if not recited in the definition of the variable group. When one or more positions on a group are said to be “not substituted” or “unsubstituted”, then hydrogen atoms are attached to take up any free valency. Unless otherwise indicated as being optionally
substituted, the term “phenyl” means unsubstituted phenyl. Unless otherwise indicated as being optionally substituted, the term “benzyl” means unsubstituted benzyl. The attachment point of (R4b)q is illustrated as floating. Each R4b can be attached to any of the 3 available aromatic carbons by replacement of a hydrogen atom. When a compound is substituted with a substituent bearing a subscript that indicates the number of said substituents can exceed 1, then said substituents (when they exceed 1) are independently selected from the group of defined substituents, (e.g., (R4b)q in Formula 1 wherein n is 0, 1 or 2), unless otherwise indicated. Further, when the subscript indicates a range, e.g., (R)i–j, then the number of substituents may be selected from the integers between i and j inclusive. The number of optional substituents may be restricted by an expressed limitation. For example, the phrase “wherein up to 2 carbon atom ring members are independently selected from C(=O) and C(=S)” means that 0, 1 or 2 substituents can be present (if the number of potential connection points allows). When a range specified for the number of substituents exceeds the number of positions available for substituents on a ring, the actual higher end of the range is recognized to be the number of available positions. As noted above, A can be a 5- or 6-membered heterocyclic ring, optionally substituted with one or more substituents selected from a group of substituents as defined in the Summary of the Invention. Examples of a 5- or 6-membered unsaturated aromatic heterocyclic ring optionally substituted with from one or more substituents include the rings U-2 through U-60 illustrated in Exhibit 1 wherein Rv is any substituent as defined in the Summary of the Invention on A (i.e., R1) and r is an integer from 0 to 4, limited by the number of available positions on each U group. As U-29, U-30, U-36, U-37, U-38, U-39, U-40, U-41, U-42 and U-43 have only one available position, for these U groups r is limited to the integers 0 or 1, and r being 0 means that the U group is unsubstituted and a hydrogen is present at the position indicated by (Rv)r. As noted above, two adjacent R2 may be taken together to form a 5- to 8-membered ring, the 5- to 8-membered ring can be (among others) saturated or unsaturated, optionally substituted with one or more substituents selected from a group of substituents as defined in the Summary of the Invention. Examples of a 5- to 8-membered unsaturated aromatic ring optionally substituted with from one or more substituents include the rings U-1 through U-60 illustrated in Exhibit 1 wherein Rv is independently H, halogen, cyano, nitro, C1–C4 alkyl, C3–C6 cycloalkyl, C1–C4 haloalkyl, C1–C4 alkoxy and C1–C4 haloalkoxy and r is an integer from 0 to 2, limited by the number of available positions on each U group. The U group can share any two available neighboring atoms with the connecting ring.
, , , , ,
, ,
. Note that when the 5- to 8-membered saturated or unsaturated non-aromatic heterocyclic ring is optionally substituted with one or more substituents selected from the group of substituents as defined in the Summary of the Invention for R2, one or two carbon ring members of the heterocycle can optionally be in the oxidized form of a carbonyl moiety. Examples of a 5- to 8-membered heterocyclic ring that is saturated or non-aromatic unsaturated heterocyclic ring containing ring members selected from up to two O atoms and up to two S atoms, and optionally substituted on carbon atom ring members with up to four Rv include the rings T-1 through T-35 as illustrated in Exhibit 2. Note that the T group can share any two available neighboring atoms with the connecting ring. The optional substituents corresponding to Rv can be attached to any available carbon or nitrogen by replacing a hydrogen atom. For these T rings, r is typically an integer from 0 to 4, limited by the number of available positions on each T group. The term “optionally substituted” means “substituted or unsubstituted”. Note that when T2 is N, the nitrogen atom can complete its valence by substitution with either H or the substituents corresponding to Rv as defined in the Summary of the Invention.
, , , , , ,
, , , . T-31 T-32 T-33 T-34 T-35 Although Rv groups are shown in the structures U-1 through U-60, and T-1 through T-35, it is noted that they do not need to be present since they are optional substituents. Note that when Rv is H when attached to an atom, this is the same as if said atom is unsubstituted. The nitrogen atoms that require substitution to fill their valence are substituted with H or Rv. Note that when the attachment point between (Rv)r and the U (or T) group is illustrated as floating, (Rv)r can be attached to any available carbon atom or nitrogen atom of the U group. A wide variety of synthetic methods are known in the art to enable preparation of aromatic and nonaromatic heterocyclic rings and ring systems; for extensive reviews see the eight volume set of Comprehensive Heterocyclic Chemistry, A. R. Katritzky and C. W. Rees editors-in-chief, Pergamon Press, Oxford, 1984 and the twelve volume set of Comprehensive Heterocyclic Chemistry II, A. R. Katritzky, C. W. Rees and E. F. V. Scriven editors-in-chief, Pergamon Press, Oxford, 1996. Embodiments of the present invention as described in the Summary of the Invention include those described below. In the following Embodiments, Formula 1 includes stereoisomers, N-oxides and salts thereof, and reference to “a compound of Formula 1” includes the definitions of substituents specified in the Summary of the Invention unless further defined in the Embodiments. Embodiment 1. A compound of Formula 1, stereoisomers, N-oxides, and salts thereof, agricultural compositions containing them and their use as herbicides as described in the Summary of the disclosure. Embodiment 2. A compound of Formula 1 or Embodiment 1 wherein A is selected from ,
Embodiment 2a. A compound of Embodiment 2 wherein A is A-1, A-2, A-3, A-5, A-7, A-8 or A-10. Embodiment 2b. A compound of Embodiment 2a wherein A is A-1, A-2 or A-8. Embodiment 2c. A compound of Embodiment 2b wherein A is A-1 or A-2. Embodiment 2d. A compound of Embodiment 2c wherein A is A-1. Embodiment 3. A compound of Formula 1 or any one of the preceding Embodiments wherein X1, X2, X3 and X4 are each independently N or CR2 wherein up to two X1, X2, X3 and X4 can be N; Embodiment 3a. A compound of Embodiment 3 wherein X1, X2, X3 and X4 are each independently CR2; or X1 is N, and X2, X3 and X4 are each independently N or CR2; or X1 and X2 are N, and X3 and X4 are each independently CR2; or X2 and X3 are N, and X1 and X4 are each independently CR2; or X1 and X3 are N, and X2 and X4 are each independently CR2; or X2 is N, and X1, X3 and X4 are each independently CR2. Embodiment 3aa. A compound of Embodiment 3a wherein X1, X2, X3 and X4 are each independently CR2. Embodiment 3b. A compound of Embodiment 3a wherein X1, X2, X3 and X4 are each independently CR2, and Z is other than O; or X1 is N, and X2, X3 and X4 are each independently N or CR2, and Z is other than O; or X1 and X2 are N, and X3
and X4 are each independently CR2, and Z is other than O; or X2 and X3 are N, and X1 and X4 are each independently CR2; or X1 and X3 are N, and X2 and X4 are each independently CR2; or X2 is N, and X1, X3 and X4 are each independently CR2, and Z is other than O. Embodiment 3b1. A compound of Embodiment 3b wherein X2 and X3 are N, and X1 and X4 are each independently CR2, and Z is O; or X1 and X3 are N, and X2 and X4 are each independently CR2, and Z is O. Embodiment 3b2. A compound of Embodiment 3b1 wherein X2 and X3 are N, and X1 and X4 are each independently CR2, and Z is O. Embodiment 3b4. A compound of Embodiment 3b2 wherein X1 and X3 are N, and X2 and X4 are each independently CR2, and Z is O. Embodiment 3c. A compound of Embodiment 3b wherein X1, X2, X3 and X4 are each independently CR2 and Z is other than O. Embodiment 3cc. A compound of Embodiment 3b wherein X2 is N, and X1, X3 and X4 are each independently CR2, and Z is other than O. Embodiment 3d. A compound of Embodiment 3b wherein X1 is N, and X2, X3 and X4 are each independently N or CR2, and Z is other than O. Embodiment 3e. A compound of Embodiment 3b wherein X1 and X2 are N, and X3 and X4 are each independently CR2, and Z is other than O. Embodiment 3g. A compound of Embodiment 3 or any one of the preceding Embodiments wherein X1 is CR2 and R2 is H. Embodiment 3h. A compound of Embodiment 3 or any one of the preceding Embodiments wherein X2 is CR2 and R2 is H, C1–C3 alkoxy, F, C1–C3 alkyl, C3–C4 cycloalkyl or taken together with X3 to form a ring. Embodiment 3i. A compound of Embodiment 3h wherein the ring contains -OCH2O- or -O(CH2)2O- or -O(CH2)2-. Embodiment 3j. A compound of Embodiment 3h wherein X2 is CR2 and R2 is H, C1–C3 alkoxy or is taken together with X3 to form a ring, and said ring contains -OCH2O- or -O(CH2)2O- or -O(CH2)2-. Embodiment 3k. A compound of Embodiment 3 or any one of the preceding Embodiments wherein X3 is CR2 and R2 is C1–C4 alkyl, C1–C3 alkoxy, F, Cl, C3–C4 cycloalkyl, C2–C4 alkynyloxy, C2–C3 haloalkoxy, C1–C3 alkylsulfonate or taken together with X2 to form a ring. Embodiment 3l. A compound of Embodiment 3k wherein the ring contains -OCH2O- or -O(CH2)2O- or -O(CH2)2-.
Embodiment 3m. A compound of Embodiment 3k or any one of the preceding Embodiments wherein X3 is CR2 and R2 is C1–C4 alkyl, C1–C3 alkoxy or taken together with X2 to form a ring. Embodiment 3n. A compound of Embodiment 3m wherein the ring contains -OCH2O- or -O(CH2)2O- or -O(CH2)2-. Embodiment 3o. A compound of Embodiment 3k wherein R2 is C1–C4 alkyl or C1–C3 alkoxy. Embodiment 3p. A compound of Embodiment 3 or any one of the preceding Embodiments wherein X4 is CR2 and R2 is H. Embodiment 4. A compound of Formula 1 or any one of the preceding Embodiments wherein R1a is H, halogen, cyano, C1–C7 alkyl, C2–C6 alkenyl, C2–C6 alkynyl, C3–C7 cycloalkyl, C4–C7 cycloalkylalkyl, C4–C7 alkylcycloalkyl, C1–C7 haloalkyl, C1–C6 hydroxyalkyl, C3–C7 hydroxycycloalkyl, C1–C6 hydroxyhaloalkyl, C4–C8 alkoxycycloalkyl, C2–C7 alkoxyhaloalkyl, C2–C6 oxacycloalkyl, C3–C7 oxacycloalkylalkyl, C3–C7 alkyloxacycloalkyl, C2–C6 thiacycloalkyl, C3–C7 thiacycloalkylalkyl, C3–C7 alkylthiacycloalkyl, C2–C6 (O-thia)cycloalkyl, C3–C7(O-thia)cycloalkylalkyl, C3–C7 alkyl(O- thia)cycloalkyl, C2–C6 (O2thia)cycloalkyl, C3–C7 (O2thia)cycloalkylalkyl, C3– C7 alkyl(O2thia)cycloalkyl, C2–C6 haloalkenyl, C2–C6 haloalkynyl, C3–C7 halocycloalkyl, C4–C7 haloalkylcycloalkyl, C2–C7 alkoxyalkyl, C2–C7 haloalkoxyalkyl, C1–C7 alkoxy, C2–C5 alkenyloxy, C2–C5 alkynyloxy, C3–C7 cycloalkoxy, C4–C7 cycloalkoxyalkyl, C1–C7 haloalkoxy, C1–C7 alkylthio, C2– C7 alkylthioalkyl, C1–C5 alkylsulfinyl, C1–C5 alkylsulfonyl, C1–C5 haloalkylthio, C2–C7 alkylsulfinylalkyl, C2–C7 alkylsulfonylalkyl, C2–C7 haloalkylthioalkyl, C2–C7 haloalkylsulfinylalkyl, C2–C7 haloalkylsulfonylalkyl, C4–C7 alkylthiocycloalkyl, C4–C7 alkylsulfinylcycloalkyl, C4–C7 alkylsulfonylcycloalkyl, C4–C7 haloalkylthiocycloalkyl, C4–C7 haloalkylsulfinylcycloalkyl, C4–C7 haloalkylsulfonylcycloalkyl, C2-C7 (HN=S(alkyl)=O)alkyl, C4-C7 (HN=S(alkyl)=O)cycloalkyl, C3-C7 (alkyl- N=S(alkyl)=O)alkyl, C5-C8 (alkyl-N=S(alkyl)=O)cycloalkyl, C3-C7 (cyano- N=S(alkyl)=O)alkyl, C4-C7 (alkylcarbonyl-N=S(alkyl)=O)alkyl, C4-C7 (alkoxycarbonyl-N=S(alkyl)=O)alkyl, C2–C5 cyanoalkyl, C4–C7 cyanocycloalkyl, C1–C4 nitroalkyl, C2–C6 dialkylamino, C2–C6 alkylcarbonyl, C3–C6 alkylcarbonylalkyl, C2–C6 alkoxycarbonyl, C3–C6 alkoxycarbonylalkyl or C3–C6 trialkylsilyl; Embodiment 4a. A compound of Embodiment 4 wherein R1a is H, C1–C7 alkyl, C2–C6 alkenyl, C2–C6 alkynyl, C3–C7 cycloalkyl, C4–C7 cycloalkylalkyl, C4–C7
alkylcycloalkyl, C1–C7 haloalkyl, C1–C6 hydroxyalkyl, C3–C7 hydroxycycloalkyl, C1–C6 hydroxyhaloalkyl, C4–C8 alkoxycycloalkyl, C2–C7 alkoxyhaloalkyl, C2–C6 oxacycloalkyl, C3–C7 oxacycloalkylalkyl, C3–C7 alkyloxacycloalkyl, C2–C6 thiacycloalkyl, C3–C7 thiacycloalkylalkyl, C3–C7 alkylthiacycloalkyl, C2–C6 (O-thia)cycloalkyl, C3–C7(O-thia)cycloalkylalkyl, C3–C7 alkyl(O-thia)cycloalkyl, C2–C6 (O2thia)cycloalkyl, C3–C7 (O2thia)cycloalkylalkyl, C3–C7 alkyl(O2thia)cycloalkyl, C2–C6 haloalkenyl, C2– C6 haloalkynyl, C3–C7 halocycloalkyl, C4–C7 haloalkylcycloalkyl, C2–C7 alkoxyalkyl, C2–C7 haloalkoxyalkyl, C1–C7 alkoxy, C2–C5 alkenyloxy, C2–C5 alkynyloxy, C3–C7 cycloalkoxy, C4–C7 cycloalkoxyalkyl, C1–C7 haloalkoxy, C1–C7 alkylthio, C2–C7 alkylthioalkyl, C1–C5 alkylsulfinyl, C1–C5 alkylsulfonyl, C1–C5 haloalkylthio, C2–C7 alkylsulfinylalkyl, C2–C7 alkylsulfonylalkyl, C2–C7 haloalkylthioalkyl, C2–C7 haloalkylsulfinylalkyl, C2– C7 haloalkylsulfonylalkyl, C4–C7 alkylthiocycloalkyl, C4–C7 alkylsulfinylcycloalkyl, C4–C7 alkylsulfonylcycloalkyl, C4–C7 haloalkylthiocycloalkyl, C4–C7 haloalkylsulfinylcycloalkyl, C4–C7 haloalkylsulfonylcycloalkyl, C2-C7 (HN=S(alkyl)=O)alkyl, C4-C7 (HN=S(alkyl)=O)cycloalkyl, C3-C7 (alkyl-N=S(alkyl)=O)alkyl, C5-C8 (alkyl- N=S(alkyl)=O)cycloalkyl, C3-C7 (cyano-N=S(alkyl)=O)alkyl, C4-C7 (alkylcarbonyl-N=S(alkyl)=O)alkyl, C4-C7 (alkoxycarbonyl- N=S(alkyl)=O)alkyl, C2–C5 cyanoalkyl or C4–C7 cyanocycloalkyl; Embodiment 4b. A compound of Embodiment 4a wherein R1a is H, C1–C7 alkyl, C2–C6 alkenyl, C2–C6 alkynyl, C3–C7 cycloalkyl, C4–C7 cycloalkylalkyl, C4–C7 alkylcycloalkyl, C1–C7 haloalkyl, C1–C6 hydroxyalkyl, C3–C7 hydroxycycloalkyl, C1–C6 hydroxyhaloalkyl, C4–C8 alkoxycycloalkyl, C2–C7 alkoxyhaloalkyl, C2–C6 oxacycloalkyl, C3–C7 oxacycloalkylalkyl, C3–C7 alkyloxacycloalkyl, C2–C6 thiacycloalkyl, C3–C7 thiacycloalkylalkyl, C2–C6 haloalkenyl, C2–C6 haloalkynyl, C3–C7 halocycloalkyl, C4–C7 haloalkylcycloalkyl, C2–C7 alkoxyalkyl, C2–C7 haloalkoxyalkyl, C1–C7 alkoxy, C2–C5 alkenyloxy, C2–C5 alkynyloxy, C3–C7 cycloalkoxy, C4–C7 cycloalkoxyalkyl, C1–C7 haloalkoxy, C1–C7 alkylthio, C2–C7 alkylthioalkyl, C1–C5 alkylsulfinyl, C1–C5 alkylsulfonyl, C1–C5 haloalkylthio, C2–C7 alkylsulfinylalkyl, C2–C7 alkylsulfonylalkyl, C2–C7 haloalkylthioalkyl, C4–C7 haloalkylthiocycloalkyl, C4–C7 haloalkylsulfonylcycloalkyl or C2–C5 cyanoalkyl; Embodiment 4c. A compound of Embodiment 4b wherein R1a is H, C1–C7 alkyl, C2–C6 alkenyl, C2–C6 alkynyl, C3–C7 cycloalkyl, C4–C7 cycloalkylalkyl, C4–C7 alkylcycloalkyl, C1–C7 haloalkyl, C1–C6 hydroxyalkyl, C4–C8
alkoxycycloalkyl, C2–C7 alkoxyhaloalkyl, C2–C6 oxacycloalkyl, C3–C7 oxacycloalkylalkyl, C3–C7 alkyloxacycloalkyl, C2–C6 thiacycloalkyl, C3–C7 thiacycloalkylalkyl, C2–C6 haloalkenyl, C2–C6 haloalkynyl, C3–C7 halocycloalkyl, C4–C7 haloalkylcycloalkyl, C2–C7 alkoxyalkyl, C2–C7 haloalkoxyalkyl, C1–C7 alkoxy, C3–C7 cycloalkoxy, C4–C7 cycloalkoxyalkyl, C1–C7 haloalkoxy, C1–C7 alkylthio, C2–C7 alkylthioalkyl, C1–C5 alkylsulfinyl, C1–C5 alkylsulfonyl, C1–C5 haloalkylthio, C2–C7 alkylsulfinylalkyl, C2–C7 alkylsulfonylalkyl or C2–C5 cyanoalkyl; Embodiment 4d. A compound of Embodiment 4c wherein R1a is H, C1–C7 alkyl, C2–C6 alkenyl, C2–C6 alkynyl, C3–C7 cycloalkyl, C4–C7 cycloalkylalkyl, C4–C7 alkylcycloalkyl, C1–C7 haloalkyl, C1–C6 hydroxyalkyl, C4–C8 alkoxycycloalkyl, C2–C7 alkoxyhaloalkyl, C2–C6 oxacycloalkyl, C3–C7 oxacycloalkylalkyl, C3–C7 alkyloxacycloalkyl, C2–C6 thiacycloalkyl, C2–C7 alkoxyalkyl, C1–C7 alkoxy, C3–C7 cycloalkoxy, C4–C7 cycloalkoxyalkyl, C1– C7 alkylthio, C2–C7 alkylthioalkyl, C2–C7 alkylsulfinylalkyl or C2–C7 alkylsulfonylalkyl; Embodiment 4e. A compound of Embodiment 4d wherein R1a is H, C1–C7 alkyl, C3–C7 cycloalkyl, C4–C7 cycloalkylalkyl, C4–C7 alkylcycloalkyl, C1–C7 haloalkyl, C1– C6 hydroxyalkyl, C4–C8 alkoxycycloalkyl, C2–C6 oxacycloalkyl, C3–C7 alkyloxacycloalkyl, C2–C7 alkoxyalkyl, C1–C7 alkoxy, C1–C7 alkylthio, C2–C7 alkylthioalkyl, C2–C7 alkylsulfinylalkyl or C2–C7 alkylsulfonylalkyl; Embodiment 4f. A compound of Embodiment 4e wherein R1a is H, C1–C7 alkyl, C3–C7 cycloalkyl, C4–C7 cycloalkylalkyl, C4–C7 alkylcycloalkyl, C1–C7 haloalkyl, C1– C6 hydroxyalkyl, C2–C6 oxacycloalkyl, C2–C7 alkoxyalkyl, C1–C7 alkoxy or C2–C7 alkylsulfonylalkyl; Embodiment 4g. A compound of Embodiment 4f wherein R1a is H, C1–C5 alkyl, C3–C5 cycloalkyl, C4–C7 cycloalkylalkyl, C4–C7 alkylcycloalkyl, C1–C5 haloalkyl, C1– C6 hydroxyalkyl, C2–C5 oxacycloalkyl, C2–C7 alkoxyalkyl, C1–C5 alkoxy or C2–C7 alkylsulfonylalkyl; Embodiment 4h. A compound of Embodiment 4g wherein R1a is C1–C5 alkyl, C3–C5 cycloalkyl, C1–C5 haloalkyl, C1–C6 hydroxyalkyl, C2–C5 oxacycloalkyl or C2– C7 alkylsulfonylalkyl; Embodiment 4g. A compound of Embodiment 4f wherein R1a is Et, i-Pr, t-Bu, c-Bu, CFMe2, CF2Me, oxetan-3-yl, CH2SO2Me or CMe2OH. Embodiment 5. A compound of Formula 1 or any one of the preceding Embodiments wherein each R1b is independently H, halogen, cyano, nitro, C1–C4 alkyl, C3–C5
cycloalkyl, C1–C4 haloalkyl, C3–C5 halocycloalkyl, C2–C4 alkoxyalkyl, C1–C4 alkoxy, C1–C4 alkylthio or C2–C4 alkoxycarbonyl. Embodiment 5a. A compound of Embodiment 5 wherein each R1b is independently H, C1–C4 alkyl, C3–C5 cycloalkyl, C1–C4 haloalkyl, C2–C4 alkoxyalkyl, C1–C4 alkoxy or C1–C4 alkylthio. Embodiment 5b. A compound of Embodiment 5a wherein each R1b is independently H, C1–C4 alkyl or C3–C5 cycloalkyl. Embodiment 5c. A compound of Embodiment 5b wherein each R1b is independently H, C1–C4 alkyl or C3–C4 cycloalkyl. Embodiment 5d. A compound of Embodiment 5c wherein R1b is H when A is A-1, A-2, A-3 and A-5, and m is 1. Embodiment 5dd. A compound of Embodiment 5d wherein R1b is H when A is A-1. Embodiment 5e. A compound of Embodiment 5b wherein R1b is C1–C4 alkyl or C3–C5 cycloalkyl when A is A-4, A-7, A-8, A-9 or A-10 and m is 1. Embodiment 5f. A compound of Embodiment 5c wherein R1b is H. Embodiment 6. A compound of Formula 1 or Embodiment 1 wherein m is 0, 1, or 2. Embodiment 6a. A compound of Embodiment 6 wherein m is 0. Embodiment 6b. A compound of Embodiment 6 wherein m is 1. Embodiment 6c. A compound of Embodiment 6 wherein m is 2. Embodiment 7. A compound of Formula 1 or Embodiment 1 wherein R1c is H, C1–C7 alkyl, C3–C7 cycloalkyl, C2–C6 oxacycloalkyl or C1–C7 haloalkyl. Embodiment 7a. A compound of Embodiment 7 wherein R1c is H, C1–C4 alkyl, C3–C5 cycloalkyl, C2–C6 oxacycloalkyl or C1–C7 haloalkyl. Embodiment 7b. A compound of Embodiment 7a wherein R1c is H, C1–C4 alkyl or C3– C5 cycloalkyl. Embodiment 7c. A compound of Embodiment 7a wherein R1c is H, Me, c-Pr, i-Pr, c-Bu and t-Bu. Embodiment 7d. A compound of Embodiment 7b wherein R1c is Me. Embodiment 7e. A compound of Embodiment 7b wherein R1c is i-Pr. Embodiment 7f. A compound of Embodiment 7b wherein R1c is c-Pr. Embodiment 7g. A compound of Embodiment 7b wherein R1c is H. Embodiment 7h. A compound of Embodiment 7b wherein R1c is c-Bu. Embodiment 7i. A compound of Embodiment 7b wherein R1c is t-Bu. Embodiment 9. A compound of Formula 1 or Embodiment 1 wherein each R2 is independently H, halogen, cyano, nitro, hydroxy, C1–C5 alkyl, C2–C5 alkenyl, C2–C5 alkynyl, C2–C5 alkenyloxy, C2–C5 alkynyloxy, C3–C7 cycloalkoxy, C4– C7 cycloalkoxyalkyl, C3–C6 cycloalkyl, C4–C7 cycloalkylalkyl, C1–C5
haloalkyl, C2–C5 haloalkenyl, C2–C5 haloalkynyl, C2–C5 alkoxyalkyl, C2–C5 haloalkoxyalkyl, C1–C5 alkoxy, C1–C5 haloalkoxy, C1–C5 alkylthio, C1–C4 alkylsulfinyl, C1–C4 alkylsulfonyl, C1–C4 alkylsulfonate, C3–C5 cycloalkylsulfonate, C1–C4 haloalkylsulfonate, C1–C4 haloalkylthio, C1–C4 haloalkylsulfinyl, C1–C4 haloalkylsulfonyl, C2–C5 cyanoalkyl, C4–C6 cyanocycloalkyl or C2–C5 alkoxycarbonyl; or two adjacent R2 may be taken together to form a saturated or unsaturated 5- to 8- membered ring, containing carbon atoms and optionally 1 to 3 oxygen, sulfur or nitrogen atoms as ring members, one or two carbon or sulfur ring members of the heterocycle can optionally be in the oxidized form of a carbonyl, sulfonyl, sulfinyl moiety, said ring unsubstituted or substituted with at least one substituent independently selected from the group consisting of halogen, cyano, nitro, C1–C4 alkyl, C3–C6 cycloalkyl, C1–C4 haloalkyl, C1–C4 alkoxy and C1– C4 haloalkoxy. Embodiment 9a. A compound of Embodiment 9 wherein each R2 is independently H, halogen, cyano, nitro, hydroxy, C1–C5 alkyl, C2–C5 alkenyl, C2–C5 alkynyl, C2– C5 alkenyloxy, C2–C5 alkynyloxy, C3–C7 cycloalkoxy, C4–C7 cycloalkoxyalkyl, C3–C6 cycloalkyl, C4–C7 cycloalkylalkyl, C1–C5 haloalkyl, C2–C5 haloalkenyl, C2–C5 haloalkynyl, C2–C5 alkoxyalkyl, C2–C5 haloalkoxyalkyl, C1–C5 alkoxy, C1–C5 haloalkoxy, C1–C5 alkylthio, C1–C4 alkylsulfinyl, C1–C4 alkylsulfonyl, C1–C4 haloalkylthio, C1–C4 haloalkylsulfinyl, C1–C4 haloalkylsulfonyl, C1–C4 alkylsulfonate or C2–C5 alkoxycarbonyl. Embodiment 9b. A compound of Embodiment 9a wherein each R2 is independently H, halogen, cyano, OH, C1–C5 alkyl, C2–C5 alkenyl, C2–C5 alkynyl, C2–C5 alkenyloxy, C2–C5 alkynyloxy, C3–C7 cycloalkoxy, C4–C7 cycloalkoxyalkyl, C3–C6 cycloalkyl, C4–C7 cycloalkylalkyl, C1–C5 haloalkyl, C2–C5 haloalkenyl, C2–C5 haloalkynyl, C2–C5 alkoxyalkyl, C2–C5 haloalkoxyalkyl, C1–C5 alkoxy, C1–C5 haloalkoxy. Embodiment 9bb. A compound of Embodiment 9a wherein each R2 is independently H, halogen, cyano, C1–C5 alkyl, C2–C5 alkenyl, C2–C5 alkynyl, C2–C5 alkenyloxy, C2–C5 alkynyloxy, C3–C7 cycloalkoxy, C4–C7 cycloalkoxyalkyl, C3–C6 cycloalkyl, C4–C7 cycloalkylalkyl, C1–C5 haloalkyl, C2–C5 haloalkenyl, C2–C5 haloalkynyl, C2–C5 alkoxyalkyl, C2–C5 haloalkoxyalkyl, C1–C5 alkoxy, C1–C5 haloalkoxy. Embodiment 9c. A compound of Embodiment 9b wherein each R2 is independently H, halogen, cyano, OH, C1–C5 alkyl, C2–C5 alkenyl, C2–C5 alkynyl, C3–C6 cycloalkyl, C4–C7 cycloalkylalkyl, C1–C5 haloalkyl, C2–C5 alkoxyalkyl, C1–C5 alkoxy, C1–C5 haloalkoxy.
Embodiment 9cc. A compound of Embodiment 9bb wherein each R2 is independently H, halogen, cyano, C1–C5 alkyl, C2–C5 alkenyl, C2–C5 alkynyl, C3–C6 cycloalkyl, C4–C7 cycloalkylalkyl, C1–C5 haloalkyl, C2–C5 alkoxyalkyl, C1–C5 alkoxy, C1–C5 haloalkoxy. Embodiment 9d. A compound of Embodiment 9c wherein each R2 is independently H, halogen, cyano, OH, C1–C5 alkyl, C2–C5 alkenyl, C2–C5 alkynyl, C3–C6 cycloalkyl, C1–C5 haloalkyl, C1–C5 alkoxy or C1–C5 haloalkoxy. Embodiment 9dd. A compound of Embodiment 9cc wherein each R2 is independently H, halogen, cyano, C1–C5 alkyl, C2–C5 alkenyl, C2–C5 alkynyl, C3–C6 cycloalkyl, C1–C5 haloalkyl, C1–C5 alkoxy or C1–C5 haloalkoxy. Embodiment 9dd1. A compound of Embodiment 9d or Embodiment 9dd wherein each R2 is independently H, OEt, propargyl, allyl, c-Pr, F, Cl, Br, CN, Me, Et, OMe, CF 3 , OCF 3 or CH 2 CF 3 . Embodiment 9d1. A compound of Embodiment 9d wherein each R2 is independently H, OH, OEt, propargyl, allyl, c-Pr, F, Cl, Br, CN, Me, Et, OMe, CF 3 , OCF 3 or CH 2 CF 3 . Embodiment 9d2. A compound of Embodiment 9c wherein each R2 is independently H, C1–C5 alkyl or C1–C5 alkoxy. Embodiment 9e. A compound of Embodiment 9d wherein each R2 is independently H, Me, OMe or Et. Embodiment 9ee. A compound of Embodiment 9d wherein each R2 is independently H, Me or OMe. Embodiment 9f. A compound of Embodiment 9e wherein R2 is H. Embodiment 9ff. A compound of Embodiment 9f wherein R2 is H when X4 is CR2. Embodiment 9g. A compound of Embodiment 9e wherein R2 is Me. Embodiment 9gg. A compound of Embodiment 9e wherein R2 is OMe. Embodiment 9h. A compound of Embodiment 9e wherein R2 is Et. Embodiment 9i. A compound of Embodiment 9 wherein two adjacent R2 may be taken together to form a saturated or unsaturated 5- to 8-membered ring, containing carbon atoms and optionally 1 to 3 oxygen, sulfur or nitrogen atoms as ring members, one or two carbon or sulfur ring members of the heterocycle can optionally be in the oxidized form of a carbonyl, sulfonyl, sulfinyl moiety, said ring unsubstituted or substituted with at least one substituent independently selected from the group consisting of halogen, cyano, nitro, C1–C4 alkyl, C3–C6 cycloalkyl, C1–C4 haloalkyl, C1–C4 alkoxy and C1–C4 haloalkoxy. Embodiment 9j. A compound of Embodiment 9i wherein two adjacent R2 may be taken together to form a 5- or 6- membered ring, containing up to 2 oxygen atoms as the ring members.
Embodiment 9k. A compound of Embodiment 9j wherein two adjacent R2 may be taken together to form a 5- or 6- membered ring and the said ring contains a group of - OCH2O- or -O(CH2)2O- or -O(CH2)2-. Embodiment 9kk. A compound of Embodiment 9j wherein two adjacent R2 may be taken together to form a 5- or 6- membered ring, wherein said ring, in addition to the two connecting carbon atoms, contains ring members selected from the group consisting of -OCH2O-, -O(CH2)2O-, and -O(CH2)2-. Embodiment 10. A compound of Formula 1 or Embodiment 1 wherein Y is O or S. Embodiment 10a. A compound of Embodiment 10 wherein Y is O. Embodiment 10b. A compound of Embodiment 10 wherein Y is S. Embodiment 11. A compound of Formula 1 or Embodiment 1 wherein R3a is halogen, cyano, nitro, C1–C7 alkyl, C2–C6 alkenyl, C2–C6 alkynyl, C3–C7 cycloalkyl, C1–C7 alkoxy, C1–C7 haloalkoxy, C4–C7 cycloalkylalkyl, C4–C7 alkylcycloalkyl, C1–C7 haloalkyl, C2–C6 haloalkenyl, C2–C6 haloalkynyl, C3– C7 halocycloalkyl, C4–C7 haloalkylcycloalkyl, C4–C7 cycloalkylhaloalkyl, C2– C7 alkoxyalkyl, C2–C7 haloalkoxyalkyl, C2–C5 alkenyloxy, C2–C5 alkynyloxy, C2–C5 alkenyloxyalkyl, C2–C5 alkynyloxyalkyl, C3–C7 cycloalkoxy, C4–C7 cycloalkoxyalkyl, C2–C6 oxacycloalkyl, C3–C7 oxacycloalkylalkyl, C2–C6 oxacycloalkoxy, C3–C6 oxacycloalkoxyalkyl, C1–C5 alkylthio, C1–C4 alkylsulfinyl, C1–C4 alkylsulfonyl, C1–C4 haloalkylthio, C1–C4 haloalkylsulfinyl, C1–C4 haloalkylsulfonyl, C2–C7 alkoxyhaloalkyl, C2–C7 haloalkoxyhaloalkyl, C2–C7 alkylthioalkyl, C2–C7 alkylsulfinylalkyl, C2–C7 alkylsulfonylalkyl, C2–C7 haloalkylthioalkyl, C2–C7 haloalkylsulfinylalkyl, C2– C7 haloalkylsulfonylalkyl, C2–C5 cyanoalkyl, C4–C6 cyanocycloalkyl, C1–C4 nitroalkyl, C3–C6 alkylcarbonylalkyl or C3–C6 alkoxycarbonylalkyl. Embodiment 11a. A compound of Embodiment 11 wherein R3a is halogen, C1–C7 alkyl, C2–C6 alkenyl, C2–C6 alkynyl, C3–C7 cycloalkyl, C1–C7 alkoxy, C4–C7 cycloalkylalkyl, C4–C7 alkylcycloalkyl, C1–C7 haloalkyl, C2–C6 haloalkenyl, C2–C6 haloalkynyl, C3–C7 halocycloalkyl, C4–C7 haloalkylcycloalkyl, C2–C7 alkoxyalkyl or C2–C7 haloalkoxyalkyl. Embodiment 11b. A compound of Embodiment 11a wherein R3a is halogen, C1–C7 alkyl, C2–C6 alkenyl, C2–C6 alkynyl, C3–C7 cycloalkyl, C1–C7 alkoxy, C4–C7 cycloalkylalkyl, C4–C7 alkylcycloalkyl, C1–C7 haloalkyl, C3–C7 halocycloalkyl, C2–C7 alkoxyalkyl or C2–C7 haloalkoxyalkyl. Embodiment 11c. A compound of Embodiment 11b wherein R3a is halogen, C1–C7 alkyl, C3–C7 cycloalkyl, C1–C7 alkoxy, C1–C7 haloalkyl or C2–C7 alkoxyalkyl.
Embodiment 11d. A compound of Embodiment 11c wherein R3a is halogen, C1–C4 alkyl, C3–C6 cycloalkyl, C1–C3 alkoxy, C1–C4 haloalkyl or C2–C5 alkoxyalkyl. Embodiment 11e. A compound of Embodiment 11d wherein R3a is halogen, Me, c-Pr, CF3, CH2OMe, CH2OEt. Embodiment 11f. A compound of Embodiment 11e wherein R3a is Me. Embodiment 11ff. A compound of Embodiment 11e wherein R3a is Et. Embodiment 11g. A compound of Embodiment 11d wherein R3a is halogen. Embodiment 11h. A compound of Embodiment 11g wherein R3a is F or Cl. Embodiment 11i. A compound of Embodiment 11d wherein R3a is CH2OMe. Embodiment 12. A compound of Formula 1 or Embodiment 1 wherein each R3b is independently H or halogen. Embodiment 12a. A compound of Embodiment 12 wherein R3b is H. Embodiment 12b. A compound of Embodiment 12 wherein R3b is halogen. Embodiment 12c. A compound of Formula 1 or Embodiment 1 wherein R3a and R3b are taken together with the carbon atom to which they are attached to form a 3- to 7- membered ring containing ring members selected from carbon atoms and up to 3 heteroatoms independently selected from 1 oxygen atom, 1 sulfur atom, and up to 3 nitrogen atoms, wherein up to 2 carbon atom ring members are independently selected from C(=O) and C(=S) and the sulfur atom ring member is selected from S, S(O) or S(O)2. Embodiment 12d. A compound of Formula 1 or Embodiment 1 wherein two R3b are taken together with the carbon atom to which they are attached to form a 3- to 7- membered ring containing ring members selected from carbon atoms and up to 3 heteroatoms independently selected from 1 oxygen atom, 1 sulfur atom, and up to 3 nitrogen atoms, wherein up to 2 carbon atom ring members are independently selected from C(=O) and C(=S) and the sulfur atom ring member is selected from S, S(O) or S(O)2. Embodiment 13. A compound of Formula 1 or Embodiment 1 wherein Z is O, C(R3c)p, C(=O), C(=S), or NR3d. Embodiment 13a. A compound of Embodiment 13 wherein Z is O, C(R3c)p or NR3d. Embodiment 13b. A compound of Embodiment 13a wherein Z is C(R3c)p or NR3d. Embodiment 13c. A compound of Embodiment 13b wherein Z is C(R3c)p. Embodiment 13d. A compound of Embodiment 13a wherein Z is NR3d. Embodiment 13e. A compound of Embodiment 13a wherein Z is O. Embodiment 13f. A compound of Embodiment 13e wherein Z is O, and both X1 and X3 are N.
Embodiment 13g. A compound of Embodiment 13e wherein Z is O, and both X2 and X3 are N. Embodiment 13h. A compound of Embodiment 13e wherein Z is O, and both X1 and X4 are N. Embodiment 13i. A compound of Embodiment 13e wherein Z is O, and both X2 and X4 are N. Embodiment 13j. A compound of Embodiment 13e wherein Z is O, and both X3 and X4 are N. Embodiment 14. A compound of Formula 1 or Embodiment 1 wherein each R3c is independently H, halogen, cyano, nitro, hydroxy, C1–C5 alkyl, C2–C5 alkenyl, C2–C5 alkynyl, C3–C5 cycloalkyl, C1–C5 haloalkyl, C2–C5 haloalkenyl, C2–C5 haloalkynyl, C2–C5 alkoxyalkyl, C2–C5 haloalkoxyalkyl, C1–C5 alkoxy, C1–C5 haloalkoxy, C1–C5 alkylthio, C1–C4 alkylsulfinyl, C1–C4 alkylsulfonyl, C1–C5 haloalkylthio, C1–C4 haloalkylsulfinyl, C1–C4 haloalkylsulfonyl, C4–C7 cycloalkylalkyl, C4–C7 alkylcycloalkyl, C3–C7 halocycloalkyl, C4–C7 haloalkylcycloalkyl, C2–C7 alkylthioalkyl, C2–C7 alkylsulfinylalkyl, C2–C7 alkylsulfonylalkyl, C2–C7 haloalkylthioalkyl, C2–C7 haloalkylsulfinylalkyl, C2– C7 haloalkylsulfonylalkyl, C2–C5 cyanoalkyl, C4–C6 cyanocycloalkyl, C1–C4 nitroalkyl, C2–C6 oxacycloalkyl, C3–C6 oxacycloalkylalkyl, C3–C7 cycloalkoxy, C4–C7 cycloalkoxyalkyl, C3–C6 alkylcarbonylalkyl, C3–C6 alkoxycarbonylalkyl or C2–C5 alkoxycarbonyl, C2–C6 alkylcarbonyloxy, C2–C6 haloalkylcarbonyloxy, C4–C6 cycloalkylcarbonyloxy, C2–C6 alkoxycarbonyloxy, C2–C6 alkylaminocarbonyloxy, C3–C6 dialkylaminocarbonyloxy; or one R3c is taken together with the adjacent R3a or R3b and the carbon atom to which they are attached to form a 3- to 7-membered ring containing ring members selected from carbon atoms and up to 3 heteroatoms independently selected from 1 oxygen atom, 1 sulfur atom, and up to 3 nitrogen atoms, wherein up to 2 carbon atom ring members are independently selected from C(=O) and C(=S) and the sulfur atom ring member is selected from S, S(O) or S(O)2; or two R3c are taken together with the carbon atom to which they are attached to form a 3- to 7-membered ring containing ring members selected from carbon atoms and up to 3 heteroatoms independently selected from 1 oxygen atom, 1 sulfur atom, and up to 3 nitrogen atoms, wherein up to 2 carbon atom ring members are independently selected from C(=O) and C(=S) and the sulfur atom ring member is selected from S, S(O) or S(O)2.
Embodiment 14a. A compound of Embodiment 14 wherein each R3c is independently H, halogen, cyano, nitro, hydroxy, C1–C5 alkyl, C2–C5 alkenyl, C2–C5 alkynyl, C3–C5 cycloalkyl, C1–C5 haloalkyl, C2–C5 haloalkenyl, C2–C5 haloalkynyl, C2– C5 alkoxyalkyl, C2–C5 haloalkoxyalkyl, C1–C5 alkoxy, C1–C5 haloalkoxy, C1– C5 alkylthio, C1–C4 alkylsulfinyl, C1–C4 alkylsulfonyl, C1–C5 haloalkylthio, C1–C4 haloalkylsulfinyl, C1–C4 haloalkylsulfonyl, C4–C7 cycloalkylalkyl, C4– C7 alkylcycloalkyl, C3–C7 halocycloalkyl, C4–C7 haloalkylcycloalkyl, C2–C7 alkylthioalkyl, C2–C7 alkylsulfinylalkyl, C2–C5 cyanoalkyl, C3–C7 cycloalkoxy, C3–C6 alkylcarbonylalkyl, C3–C6 alkoxycarbonylalkyl or C2–C5 alkoxycarbonyl, C2–C6 alkylcarbonyloxy. Embodiment 14b. A compound of Embodiment 14a wherein each R3c is independently H, halogen, hydroxy, C1–C5 alkyl, C3–C5 cycloalkyl, C1–C5 haloalkyl, C2–C5 alkoxyalkyl, C2–C5 haloalkoxyalkyl, C1–C5 alkoxy, C1–C5 haloalkoxy, C4–C7 cycloalkylalkyl, C3–C7 cycloalkoxy, C3–C6 alkylcarbonylalkyl, C3–C6 alkoxycarbonylalkyl or C2–C5 alkoxycarbonyl, C2–C6 alkylcarbonyloxy. Embodiment 14c. A compound of Embodiment 14b wherein each R3c is independently H, halogen, hydroxy, C1–C5 alkyl, C3–C5 cycloalkyl, C2–C5 alkoxyalkyl, C1–C5 alkoxy, C4–C7 cycloalkylalkyl, C3–C7 cycloalkoxy, C3–C6 alkylcarbonylalkyl, C3–C6 alkoxycarbonylalkyl or C2–C5 alkoxycarbonyl, C2–C6 alkylcarbonyloxy. Embodiment 14d. A compound of Embodiment 14c wherein R3c is independently H, halogen, hydroxy, C1–C5 alkyl, C1–C5 alkoxy, C3–C6 alkylcarbonylalkyl, C3– C6 alkoxycarbonylalkyl or C2–C6 alkylcarbonyloxy. Embodiment 14e. A compound of Embodiment 14d wherein each R3c is independently H, F, Cl, hydroxy, Me, OMe or -OC(=O)Me. Embodiment 14f. A compound of Embodiment 14e wherein each R3c is independently H. Embodiment 15. A compound of Formula 1 or Embodiment 1 wherein R3d is H, C1–C4 alkyl, C3–C6 cycloalkyl, C1–C4 haloalkyl, C2–C5 alkylcarbonyl, C2–C5 haloalkylcarbonyl, C2–C5 alkoxycarbonyl, C2–C5 alkylaminocarbonyl, C2–C5 dialkylaminocarbonyl, C1–C5 alkylsulfonyl or C1–C5 haloalkylsulfonyl; Embodiment 15a. A compound of Embodiment 15 wherein R3d is H, C1–C4 alkyl, C2– C5 alkylcarbonyl, C2–C5 haloalkylcarbonyl, C2–C5 alkoxycarbonyl, C2–C5 alkylaminocarbonyl, C2–C5 dialkylaminocarbonyl, C1–C5 alkylsulfonyl or C1– C5 haloalkylsulfonyl. Embodiment 15b. A compound of Embodiment 15a wherein R3d is H or C1–C4 alkyl. Embodiment 15c. A compound of Embodiment 15b wherein R3d is H or Me. Embodiment 15d. A compound of Embodiment 15c wherein R3d is H.
Embodiment 15d1. A compound of Formula 1 or Embodiment 13d or Embodiment 15d wherein R3d is H; and R3a and R3b are taken together with the carbon atom to which they are attached to form C(=O). Embodiment 15e. A compound of Embodiment 15c wherein R3d is Me. Embodiment 16. A compound of Formula 1 or Embodiment 1 wherein p is 0 or 1. Embodiment 16a. A compound of Embodiment 16 wherein p is 0. Embodiment 16b. A compound of Embodiment 16 wherein p is 1. Embodiment 17. A compound of Formula 1 or Embodiment 1 wherein R4a is H, halogen, cyano, nitro, C1–C5 alkyl, C2–C5 alkenyl, C2–C5 alkynyl, C3–C5 cycloalkyl, C1–C5 haloalkyl, C2–C5 haloalkenyl, C2–C5 haloalkynyl, C2–C5 alkoxyalkyl, C2–C5 haloalkoxyalkyl, C1–C5 alkoxy, C1–C5 haloalkoxy, C3–C6 trialkylsilyl, C1–C5 alkylthio, C1–C5 haloalkylthio or C2–C5 alkoxycarbonyl. Embodiment 17b. A compound of Embodiment 17 wherein R4a is H, halogen, C1–C5 alkyl, C1–C5 haloalkyl. Embodiment 17c. A compound of Embodiment 17b wherein R4a is H, halogen, C1–C4 alkyl, C1–C4 haloalkyl. Embodiment 17d. A compound of Embodiment 17c wherein R4a is H, Cl, Me, F, CF3. Embodiment 17e. A compound of Embodiment 17d wherein R4a is H, Cl or Me. Embodiment 17f. A compound of Embodiment 17d wherein R4a is H. Embodiment 17g. A compound of Embodiment 17d wherein R4a is Cl. Embodiment 17h. A compound of Embodiment 17d wherein R4a is Me. Embodiment 17i. A compound of Embodiment 17d wherein R4a is CF3. Embodiment 17j. A compound of Embodiment 17d wherein R4a is F. Embodiment 18. A compound of Formula 1 or Embodiment 1 wherein q is 0 or 1. Embodiment 18a. A compound of Embodiment 18 wherein q is 0. Embodiment 18b. A compound of Embodiment 18 wherein q is 1. Embodiment 19. A compound of Formula 1 or Embodiment 1 wherein the stereochemistry of the carbon atom with * is (1') depicted as Formula 1' below.
1'
Embodiment 20. A compound of Formula 1 or Embodiment 1 wherein the stereochemistry of the carbon atom with * is (1''), depicted as Formula 1'' below.
Embodiments of this invention, including Embodiments 1–20 above as well as any other embodiments described herein, can be combined in any manner, and the descriptions of variables in the embodiments pertain not only to the compounds of Formula 1 but also to the starting compounds and intermediate compounds useful for preparing the compounds of Formula 1. In addition, embodiments of this invention, including Embodiments 1–20 above as well as any other embodiments described herein, and any combination thereof, pertain to the compositions and methods of the present invention. Combinations of Embodiments 1–20 are illustrated by: Embodiment X. A compound of Formula 1 as described in the Summary of the Disclosure wherein A is selected from , ,
A-10 Embodiment A1. The compound of Embodiment X wherein Z is C(R3c)p or NR3d. Embodiment A2. The compound of Embodiment A1 wherein A is A-1, A-2, A-3, A-5, A-7, A-8 or A-10; X1, X2, X3 and X4 are each independently CR2; or X1 is N, and X2, X3 and X4 are each independently N or CR2; or X1 and X2 are N, and X3 and X4 are each independently CR2; or X2 and X3 are N, and X1 and X4 are each independently CR2; or X1 and X3 are N, and X2 and X4 are each independently CR2; or X2 is N, and X1, X3 and X4 are CR2. R1a is H, C1–C7 alkyl, C2–C6 alkenyl, C2–C6 alkynyl, C3–C7 cycloalkyl, C4–C7 cycloalkylalkyl, C4–C7 alkylcycloalkyl, C1–C7 haloalkyl, C1–C6 hydroxyalkyl, C3–C7 hydroxycycloalkyl, C1–C6 hydroxyhaloalkyl, C4–C8 alkoxycycloalkyl, C2–C7 alkoxyhaloalkyl, C2–C6 oxacycloalkyl, C3–C7 oxacycloalkylalkyl, C3–C7 alkyloxacycloalkyl, C2–C6 thiacycloalkyl, C3– C7 thiacycloalkylalkyl, C2–C6 haloalkenyl, C2–C6 haloalkynyl, C3–C7 halocycloalkyl, C4–C7 haloalkylcycloalkyl, C2–C7 alkoxyalkyl, C2–C7 haloalkoxyalkyl, C1–C7 alkoxy, C2–C5 alkenyloxy, C2–C5 alkynyloxy, C3–C7 cycloalkoxy, C4–C7 cycloalkoxyalkyl, C1–C7 haloalkoxy, C1–C7 alkylthio, C2–C7 alkylthioalkyl, C1–C5 alkylsulfinyl, C1–C5 alkylsulfonyl, C1–C5 haloalkylthio, C2–C7 alkylsulfinylalkyl, C2–C7 alkylsulfonylalkyl, C2–C7 haloalkylthioalkyl, C4–C7 haloalkylthiocycloalkyl, C4–C7 haloalkylsulfonylcycloalkyl or C2–C5 cyanoalkyl; each R1b is independently H, C1–C4 alkyl, C3–C5 cycloalkyl, C1–C4 haloalkyl, C2–C4 alkoxyalkyl, C1–C4 alkoxy or C1–C4 alkylthio; R1c is H, C1–C4 alkyl, C3–C5 cycloalkyl, C2–C6 oxacycloalkyl or C1–C7 haloalkyl;
each R2 is independently H, halogen, cyano, OH, C1–C5 alkyl, C2–C5 alkenyl, C2–C5 alkynyl, C2–C5 alkenyloxy, C2–C5 alkynyloxy, C3–C7 cycloalkoxy, C4–C7 cycloalkoxyalkyl, C3–C6 cycloalkyl, C4–C7 cycloalkylalkyl, C1–C5 haloalkyl, C2–C5 haloalkenyl, C2–C5 haloalkynyl, C2–C5 alkoxyalkyl, C2–C5 haloalkoxyalkyl, C1–C5 alkoxy, C1–C5 haloalkoxy; or two adjacent R2 may be taken together to form a 5- or 6- membered ring, containing up to 2 oxygen atoms as the ring members. R3a is halogen, C1–C7 alkyl, C2–C6 alkenyl, C2–C6 alkynyl, C3–C7 cycloalkyl, C1–C7 alkoxy, C4–C7 cycloalkylalkyl, C4–C7 alkylcycloalkyl, C1–C7 haloalkyl, C2–C6 haloalkenyl, C2–C6 haloalkynyl, C3–C7 halocycloalkyl, C4–C7 haloalkylcycloalkyl, C2–C7 alkoxyalkyl or C2–C7 haloalkoxyalkyl; each R3c is independently H, halogen, hydroxy, C1–C5 alkyl, C1–C5 alkoxy, C3– C6 alkylcarbonylalkyl, C3–C6 alkoxycarbonylalkyl or C2–C6 alkylcarbonyloxy; R3d is H, C1–C4 alkyl, C2–C5 alkylcarbonyl, C2–C5 haloalkylcarbonyl, C2–C5 alkoxycarbonyl, C2–C5 alkylaminocarbonyl, C2–C5 dialkylaminocarbonyl, C1–C5 alkylsulfonyl or C1–C5 haloalkylsulfonyl; and R4a is H, halogen, C1–C5 alkyl, C1–C5 haloalkyl. Embodiment A3. The compound of Embodiment A2 wherein A is A-1, A-2 or A-8; R1a is H, C1–C7 alkyl, C3–C7 cycloalkyl, C4–C7 cycloalkylalkyl, C4–C7 alkylcycloalkyl, C1–C7 haloalkyl, C1–C6 hydroxyalkyl, C4–C8 alkoxycycloalkyl, C2–C6 oxacycloalkyl, C3–C7 alkyloxacycloalkyl, C2–C7 alkoxyalkyl, C1–C7 alkoxy, C1–C7 alkylthio, C2–C7 alkylthioalkyl, C2–C7 alkylsulfinylalkyl or C2–C7 alkylsulfonylalkyl; each R1b is independently H, C1–C4 alkyl or C3–C4 cycloalkyl; R1c is H, C1–C4 alkyl or C3–C5 cycloalkyl; each R2 is independently H, halogen, cyano, OH, C1–C5 alkyl, C2–C5 alkenyl, C2–C5 alkynyl, C3–C6 cycloalkyl, C1–C5 haloalkyl, C1–C5 alkoxy or C1– C5 haloalkoxy; or two adjacent R2 may be taken together to form a 5- or 6- membered ring, containing up to 2 oxygen atoms as the ring members; R3a is halogen, C1–C7 alkyl, C3–C7 cycloalkyl, C1–C7 alkoxy, C1–C7 haloalkyl or C2–C7 alkoxyalkyl; R3c is independently H, F, Cl, hydroxy, Me, OMe or -OC(=O)Me; and R3d is H or C1–C4 alkyl. Embodiment A4. The compound of Embodiment A3 wherein A is A-1;
X1, X2, X3 and X4 are CR2; R1a is C1–C5 alkyl, C3–C5 cycloalkyl, C1–C5 haloalkyl, C1–C6 hydroxyalkyl, C2–C5 oxacycloalkyl or C2–C7 alkylsulfonylalkyl; each R2 is independently H, halogen, cyano, OH, C1–C5 alkyl, C2–C5 alkenyl, C2–C5 alkynyl, C3–C6 cycloalkyl, C1–C5 haloalkyl, C1–C5 alkoxy or C1– C5 haloalkoxy; or two adjacent R2 may be taken together to form a 5- or 6- membered ring and the said ring contains a group of -OCH2O- or - O(CH2)2O- or -O(CH2)2-; R3a is halogen, C1–C4 alkyl, C3–C6 cycloalkyl, C1–C3 alkoxy, C1–C4 haloalkyl or C2–C5 alkoxyalkyl; each R3b is independently H or halogen; and R3d is H or Me. Embodiment A5. The compound of Embodiment A4 wherein R1a is Et, i-Pr, t-Bu, c-Bu, CFMe2, CF2Me, oxetan-3-yl, CH2SO2Me or CMe2OH; R1b is H; R1c is H, Me, c-Pr or i-Pr; each R2 is independently H, OH, OEt, propargyl, allyl, c-Pr, F, Cl, Br, CN, Me, Et, OMe, CF 3 , OCF 3 or CH 2 CF 3 ; Y is O; R3a is halogen, Me, c-Pr, CF3, CH2OMe, or CH2OEt; R3b is H; R3d is H or Me; and R4a is H, Cl or Me. Embodiment A6. The compound of any one of the preceding Embodiments wherein Z is C(R3c)p. Embodiment B1. The compound of Embodiment X wherein Z is O. Embodiment B2. The compound of Embodiment B1 wherein X2 and X3 are N, and X1 and X4 are each independently CR2; or X1 and X3 are N, and X2 and X4 are each independently CR2. Embodiment B3. The compound of Embodiment B2 wherein A is A-1, A-2, A-3, A-5, A-7, A-8 or A-10; R1a is H, C1–C7 alkyl, C2–C6 alkenyl, C2–C6 alkynyl, C3–C7 cycloalkyl, C4–C7 cycloalkylalkyl, C4–C7 alkylcycloalkyl, C1–C7 haloalkyl, C1–C6 hydroxyalkyl, C3–C7 hydroxycycloalkyl, C1–C6 hydroxyhaloalkyl, C4–C8 alkoxycycloalkyl, C2–C7 alkoxyhaloalkyl, C2–C6 oxacycloalkyl, C3–C7
oxacycloalkylalkyl, C3–C7 alkyloxacycloalkyl, C2–C6 thiacycloalkyl, C3– C7 thiacycloalkylalkyl, C2–C6 haloalkenyl, C2–C6 haloalkynyl, C3–C7 halocycloalkyl, C4–C7 haloalkylcycloalkyl, C2–C7 alkoxyalkyl, C2–C7 haloalkoxyalkyl, C1–C7 alkoxy, C2–C5 alkenyloxy, C2–C5 alkynyloxy, C3– C7 cycloalkoxy, C4–C7 cycloalkoxyalkyl, C1–C7 haloalkoxy, C1–C7 alkylthio, C2–C7 alkylthioalkyl, C1–C5 alkylsulfinyl, C1–C5 alkylsulfonyl, C1–C5 haloalkylthio, C2–C7 alkylsulfinylalkyl, C2–C7 alkylsulfonylalkyl, C2–C7 haloalkylthioalkyl, C4–C7 haloalkylthiocycloalkyl, C4–C7 haloalkylsulfonylcycloalkyl or C2–C5 cyanoalkyl; each R1b is independently H, C1–C4 alkyl, C3–C5 cycloalkyl, C1–C4 haloalkyl, C2–C4 alkoxyalkyl, C1–C4 alkoxy or C1–C4 alkylthio; R1c is H, C1–C4 alkyl, C3–C5 cycloalkyl, C2–C6 oxacycloalkyl or C1–C7 haloalkyl; each R2 is independently H, halogen, cyano, OH, C1–C5 alkyl, C2–C5 alkenyl, C2–C5 alkynyl, C2–C5 alkenyloxy, C2–C5 alkynyloxy, C3–C7 cycloalkoxy, C4–C7 cycloalkoxyalkyl, C3–C6 cycloalkyl, C4–C7 cycloalkylalkyl, C1–C5 haloalkyl, C2–C5 haloalkenyl, C2–C5 haloalkynyl, C2–C5 alkoxyalkyl, C2– C5 haloalkoxyalkyl, C1–C5 alkoxy, C1–C5 haloalkoxy; or two adjacent R2 may be taken together to form a 5- or 6- membered ring, containing up to 2 oxygen atoms as the ring members; R3a is halogen, C1–C7 alkyl, C2–C6 alkenyl, C2–C6 alkynyl, C3–C7 cycloalkyl, C1–C7 alkoxy, C4–C7 cycloalkylalkyl, C4–C7 alkylcycloalkyl, C1–C7 haloalkyl, C2–C6 haloalkenyl, C2–C6 haloalkynyl, C3–C7 halocycloalkyl, C4–C7 haloalkylcycloalkyl, C2–C7 alkoxyalkyl or C2–C7 haloalkoxyalkyl; each R3c is independently H, halogen, hydroxy, C1–C5 alkyl, C1–C5 alkoxy, C3– C6 alkylcarbonylalkyl, C3–C6 alkoxycarbonylalkyl or C2–C6 alkylcarbonyloxy; R3d is H, C1–C4 alkyl, C2–C5 alkylcarbonyl, C2–C5 haloalkylcarbonyl, C2–C5 alkoxycarbonyl, C2–C5 alkylaminocarbonyl, C2–C5 dialkylaminocarbonyl, C1–C5 alkylsulfonyl or C1–C5 haloalkylsulfonyl; and R4a is H, halogen, C1–C5 alkyl, C1–C5 haloalkyl. Embodiment B4. The compound of Embodiment B3 wherein A is A-1, A-2 or A-8; R1a is H, C1–C7 alkyl, C3–C7 cycloalkyl, C4–C7 cycloalkylalkyl, C4–C7 alkylcycloalkyl, C1–C7 haloalkyl, C1–C6 hydroxyalkyl, C4–C8 alkoxycycloalkyl, C2–C6 oxacycloalkyl, C3–C7 alkyloxacycloalkyl, C2–C7
alkoxyalkyl, C1–C7 alkoxy, C1–C7 alkylthio, C2–C7 alkylthioalkyl, C2–C7 alkylsulfinylalkyl or C2–C7 alkylsulfonylalkyl; each R1b is independently H, C1–C4 alkyl or C3–C4 cycloalkyl; R1c is H, C1–C4 alkyl or C3–C5 cycloalkyl; each R2 is independently H, halogen, cyano, OH, C1–C5 alkyl, C2–C5 alkenyl, C2–C5 alkynyl, C3–C6 cycloalkyl, C1–C5 haloalkyl, C1–C5 alkoxy or C1– C5 haloalkoxy; or two adjacent R2 may be taken together to form a 5- or 6- membered ring, containing up to 2 oxygen atoms as the ring members; R3a is halogen, C1–C7 alkyl, C3–C7 cycloalkyl, C1–C7 alkoxy, C1–C7 haloalkyl or C2–C7 alkoxyalkyl; R3c is independently H, F, Cl, hydroxy, Me, OMe or -OC(=O)Me; and R3d is H or C1–C4 alkyl. Embodiment B5. The compound of Embodiment B4 wherein A is A-1; X1 and X3 are N, and X2 and X4 are independently CR2; R1a is C1–C5 alkyl, C3–C5 cycloalkyl, C1–C5 haloalkyl, C1–C6 hydroxyalkyl, C2–C5 oxacycloalkyl or C2–C7 alkylsulfonylalkyl; each R2 is independently H, halogen, cyano, OH, C1–C5 alkyl, C2–C5 alkenyl, C2–C5 alkynyl, C3–C6 cycloalkyl, C1–C5 haloalkyl, C1–C5 alkoxy or C1– C5 haloalkoxy; or two adjacent R2 may be taken together to form a 5- or 6- membered ring and the said ring contains a group of -OCH2O- or - O(CH2)2O- or -O(CH2)2-; R3a is halogen, C1–C4 alkyl, C3–C6 cycloalkyl, C1–C3 alkoxy, C1–C4 haloalkyl or C2–C5 alkoxyalkyl; each R3b is independently H or halogen; and R3d is H or Me. Embodiment B6. The compound of Embodiment B5 wherein R1a is Et, i-Pr, t-Bu, c-Bu, CFMe2, CF2Me, oxetan-3-yl, CH2SO2Me or CMe2OH; R1b is H; R1c is H, Me, c-Pr or i-Pr; each R2 is independently H, OH, OEt, propargyl, allyl, c-Pr, F, Cl, Br, CN, Me, Et, OMe, CF 3 , OCF 3 or CH 2 CF 3 ; Y is O; R3a is halogen, Me, c-Pr, CF3, CH2OMe, or CH2OEt; R3b is H; R3d is H or Me; and
R4a is H or Me. Embodiment B7. The compound of Embodiment B4 wherein A is A-1; X2 and X3 are N, and X1 and X4 are independently CR2; R1a is C1–C5 alkyl, C3–C5 cycloalkyl, C1–C5 haloalkyl, C1–C6 hydroxyalkyl, C2–C5 oxacycloalkyl or C2–C7 alkylsulfonylalkyl; each R2 is independently H, halogen, cyano, OH, C1–C5 alkyl, C2–C5 alkenyl, C2–C5 alkynyl, C3–C6 cycloalkyl, C1–C5 haloalkyl, C1–C5 alkoxy or C1– C5 haloalkoxy; or two adjacent R2 may be taken together to form a 5- or 6- membered ring and the said ring contains a group of -OCH2O- or - O(CH2)2O- or -O(CH2)2-; R3a is halogen, C1–C4 alkyl, C3–C6 cycloalkyl, C1–C3 alkoxy, C1–C4 haloalkyl or C2–C5 alkoxyalkyl; each R3b is independently H or halogen; and R3d is H or Me. Embodiment B8. The compound of Embodiment B7 wherein R1a is Et, i-Pr, t-Bu, c-Bu, CFMe2, CF2Me, oxetan-3-yl, CH2SO2Me or CMe2OH; R1b is H; R1c is H, Me, c-Pr or i-Pr; each R2 is independently H, OH, OEt, propargyl, allyl, c-Pr, F, Cl, Br, CN, Me, Et, OMe, CF 3 , OCF 3 or CH 2 CF 3 ; Y is O; R3a is halogen, Me, c-Pr, CF3, CH2OMe, or CH2OEt; R3b is H; R3d is H or Me; and R4a is H or Me. Embodiment C. Specific embodiment including a compound of Formula 1 wherein: A is A-1, R1a is i-Pr, R1b is H, X1, X2 and X4 are all CH, X3 is COMe, Y is O, R3a is Me, R3b is H, Z is CH2 and R4a is Me; (Compound 3) Embodiment D. Specific embodiments include compounds of Formula 1 selected from the group consisting of: (3,4-dihydro-3,5-dimethyl-1(2H)-quinolinyl)-[2-methoxy-5-[3-(1-methylethyl)- 1H-1,2,4-triazol-1-yl]phenyl]methanone; [(3S)-3,4-dihydro-3,5-dimethyl-1(2H)-quinolinyl][2-methoxy-5-[3-(1- methylethyl)-1H-1,2,4-triazol-1-yl]phenyl]methanone;
[(3R)-3,4-dihydro-3,5-dimethyl-1(2H)-quinolinyl][2-methoxy-5-[3-(1- methylethyl)-1H-1,2,4-triazol-1-yl]phenyl]methanone; [5-fluoro-3,4-dihydro-3-(trifluoromethyl)-1(2H)-quinolinyl][2-methoxy-5- [3-(1-methylethyl)-1H-1,2,4-triazol-1-yl]phenyl]methanone; [(3S)-5-fluoro-3,4-dihydro-3-(trifluoromethyl)-1(2H)-quinolinyl][2- methoxy-5-[3-(1-methylethyl)-1H-1,2,4-triazol-1- yl]phenyl]methanone; [(3R)-5-fluoro-3,4-dihydro-3-(trifluoromethyl)-1(2H)-quinolinyl][2- methoxy-5-[3-(1-methylethyl)-1H-1,2,4-triazol-1- yl]phenyl]methanone; [5-fluoro-3,4-dihydro-3-(trifluoromethyl)-1(2H)-quinolinyl][2-methyl-5-[3- (1-methylethyl)-1H-1,2,4-triazol-1-yl]phenyl]methanone; [(3S)-5-fluoro-3,4-dihydro-3-(trifluoromethyl)-1(2H)-quinolinyl][2-methyl- 5-[3-(1-methylethyl)-1H-1,2,4-triazol-1-yl]phenyl]methanone; [(3R)-5-fluoro-3,4-dihydro-3-(trifluoromethyl)-1(2H)-quinolinyl][2- methyl-5-[3-(1-methylethyl)-1H-1,2,4-triazol-1-yl]phenyl]methanone; [3,4-dihydro-5-methyl-3-(trifluoromethyl)-1(2H)-quinolinyl][2-methoxy-5- [3-(1-methylethyl)-1H-1,2,4-triazol-1-yl]phenyl]methanone; [(3S)-3,4-dihydro-5-methyl-3-(trifluoromethyl)-1(2H)-quinolinyl][2- methoxy-5-[3-(1-methylethyl)-1H-1,2,4-triazol-1- yl]phenyl]methanone; [(3R)-3,4-dihydro-5-methyl-3-(trifluoromethyl)-1(2H)-quinolinyl][2- methoxy-5-[3-(1-methylethyl)-1H-1,2,4-triazol-1- yl]phenyl]methanone; and [3,4-dihydro-3-methyl-5-(trifluoromethyl)-1(2H)-quinoxalinyl][2-methoxy- 5-[3-(1-methylethyl)-1H-1,2,4-triazol-1-yl]phenyl]methanone; and combinations thereof. This invention also relates to a method for controlling undesired vegetation comprising applying to the locus of the vegetation herbicidally effective amounts of the compounds of the invention (e.g., as a composition described herein). Of note as embodiments relating to methods of use are those involving the compounds of embodiments described above. Compounds of the invention are particularly useful for selective control of weeds in crops such as wheat, barley, maize, soybean, sunflower, cotton, oilseed rape and rice, and specialty crops such as sugarcane, citrus, fruit and nut crops. Also noteworthy as embodiments are herbicidal compositions of the present invention comprising the compounds of embodiments described above.
This invention also includes a herbicidal mixture comprising (a) a compound selected from Formula 1, N-oxides, and salts thereof, and (b) at least one additional active ingredient selected from (b1) photosystem II inhibitors, (b2) acetohydroxy acid synthase (AHAS) inhibitors, (b3) acetyl-CoA carboxylase (ACCase) inhibitors, (b4) auxin mimics, (b5) 5-enol- pyruvylshikimate-3-phosphate (EPSP) synthase inhibitors, (b6) photosystem I electron diverters, (b7) protoporphyrinogen oxidase (PPO) inhibitors, (b8) glutamine synthetase (GS) inhibitors, (b9) very long chain fatty acid (VLCFA) elongase inhibitors, (b10) auxin transport inhibitors, (b11) phytoene desaturase (PDS) inhibitors, (b12) 4-hydroxyphenyl-pyruvate dioxygenase (HPPD) inhibitors, (b13) DXP synthase inhibitors, (b14) homogentisate solanesyltransferase (HST) inhibitors, (b15) cellulose biosynthesis inhibitors, (b16) dehydrooritate dehydrogenase (DHODH) inhibitors, (b17) other herbicides including mitotic disruptors, organic arsenicals, asulam, bromobutide, cinflubrolin, cinmethylin, cumyluron, dazomet, difenzoquat, dymron, etobenzanid, flurenol, fosamine, fosamine-ammonium, hydantocidin, metam, methyldymron, oleic acid, oxaziclomefone, pelargonic acid and pyributicarb, (b18) herbicide safeners, and salts of compounds of (b1) through (b18). “Photosystem II inhibitors” (b1) are chemical compounds that bind to the D-1 protein at the QB-binding niche and thus block electron transport from QA to QB in the chloroplast thylakoid membranes. The electrons blocked from passing through photosystem II are transferred through a series of reactions to form toxic compounds that disrupt cell membranes and cause chloroplast swelling, membrane leakage, and ultimately cellular destruction. The QB-binding niche has three different binding sites: binding site A binds the triazines such as atrazine, triazinones such as hexazinone, and uracils such as bromacil, binding site B binds the phenylureas such as diuron, and binding site C binds benzothiadiazoles such as bentazon, nitriles such as bromoxynil and phenyl-pyridazines such as pyridate. Examples of photosystem II inhibitors include ametryn, amicarbazone, atrazine, bentazon, bromacil, bromofenoxim, bromoxynil, chlorbromuron, chloridazon, chlorotoluron, chloroxuron, cumyluron, cyanazine, daimuron, desmedipham, desmetryn, dimefuron, dimethametryn, diuron, ethidimuron, fenuron, fluometuron, hexazinone, ioxynil, isoproturon, isouron, lenacil, linuron, metamitron, methabenzthiazuron, metobromuron, metoxuron, metribuzin, monolinuron, neburon, pentanochlor, phenmedipham, prometon, prometryn, propanil, propazine, pyridafol, pyridate, siduron, simazine, simetryn, tebuthiuron, terbacil, terbumeton, terbuthylazine, terbutryn and trietazine. “AHAS inhibitors” (b2) are chemical compounds that inhibit acetohydroxy acid synthase (AHAS), also known as acetolactate synthase (ALS), and thus kill plants by inhibiting the production of the branched-chain aliphatic amino acids such as valine, leucine and isoleucine, which are required for protein synthesis and cell growth. Examples of AHAS inhibitors include amidosulfuron, azimsulfuron, bensulfuron-methyl, bispyribac-sodium, cloransulam-methyl, chlorimuron-ethyl, chlorsulfuron, cinosulfuron, cyclosulfamuron,
diclosulam, ethametsulfuron-methyl, ethoxysulfuron, flazasulfuron, florasulam, flucarbazone-sodium, flumetsulam, flupyrsulfuron-methyl, flupyrsulfuron-sodium, foramsulfuron, halosulfuron-methyl, imazamethabenz-methyl, imazamox, imazapic, imazapyr, imazaquin, imazethapyr, imazosulfuron, iodosulfuron-methyl (including sodium salt), iodosulfuron-sodium, iofensulfuron (2-iodo-N-[[(4-methoxy-6-methyl-1,3,5-triazin-2- yl)amino]carbonyl]benzenesulfonamide), iofensulfuron-sodium, mesosulfuron-methyl, metazosulfuron (3-chloro-4-(5,6-dihydro-5-methyl-1,4,2-dioxazin-3-yl)-N-[[(4,6-dimethoxy- 2-pyrimidinyl)amino]carbonyl]-1-methyl-1H-pyrazole-5-sulfonamide), metosulam, metsulfuron-methyl, nicosulfuron, oxasulfuron, penoxsulam, primisulfuron-methyl, propoxycarbazone-sodium, propyrisulfuron (2-chloro-N-[[(4,6-dimethoxy-2- pyrimidinyl)amino]carbonyl]-6-propylimidazo[1,2-b]pyridazine-3-sulfonamide), prosulfuron, pyrazosulfuron-ethyl, pyribenzoxim, pyriftalid, pyriminobac-methyl, pyrithiobac-sodium, rimsulfuron, sulfometuron-methyl, sulfosulfuron, thiencarbazone, thifensulfuron-methyl, triafamone (N-[2-[(4,6-dimethoxy-1,3,5-triazin-2-yl)carbonyl]-6- fluorophenyl]-1,1-difluoro-N-methylmethanesulfonamide), triasulfuron, tribenuron-methyl, trifloxysulfuron (including sodium salt), triflusulfuron-methyl and tritosulfuron. “ACCase inhibitors” (b3) are chemical compounds that inhibit the acetyl-CoA carboxylase enzyme, which is responsible for catalyzing an early step in lipid and fatty acid synthesis in plants. Lipids are essential components of cell membranes, and without them, new cells cannot be produced. The inhibition of acetyl CoA carboxylase and the subsequent lack of lipid production leads to losses in cell membrane integrity, especially in regions of active growth such as meristems. Eventually shoot and rhizome growth ceases, and shoot meristems and rhizome buds begin to die back. Examples of ACCase inhibitors include alloxydim, butroxydim, clethodim, clodinafop, cycloxydim, cyhalofop, diclofop, fenoxaprop, fluazifop, haloxyfop, metproxybicyclone, pinoxaden, profoxydim, propaquizafop, quizalofop, sethoxydim, tepraloxydim and tralkoxydim, including resolved forms such as fenoxaprop-P, fluazifop-P, haloxyfop-P and quizalofop-P and ester forms such as clodinafop-propargyl, cyhalofop-butyl, diclofop-methyl and fenoxaprop-P-ethyl. Auxin is a plant hormone that regulates growth in many plant tissues. “Auxin mimics” (b4) are chemical compounds mimicking the plant growth hormone auxin, thus causing uncontrolled and disorganized growth leading to plant death in susceptible species. Examples of auxin mimics include aminocyclopyrachlor (6-amino-5-chloro-2-cyclopropyl-4- pyrimidinecarboxylic acid) and its methyl and ethyl esters and its sodium and potassium salts, 4-amino-3-chloro-5-fluoro-6-(7-fluoro-1H-indol-6-yl)-2-pyridinecarboxylic 2-propyn-1-yl ester (CAS No. 2251111-17-6), 4-amino-3-chloro-5-fluoro-6-(7-fluoro-1H-indol-6-yl)-2- pyridinecarboxylic cyanomethyl ester (CAS No. 2251111-18-7), aminopyralid, benazolin-ethyl, chloramben, clacyfos, clomeprop, clopyralid, dicamba, 2,4-D, 2,4-DB, dichlorprop, fluchloraminopyr, fluchloraminopyr-tefuryl, fluroxypyr, halauxifen (4-amino-3-
chloro-6-(4-chloro-2-fluoro-3-methoxyphenyl)-2-pyridinecarboxylic acid), halauxifen- methyl (methyl 4-amino-3-chloro-6-(4-chloro-2-fluoro-3-methoxyphenyl)-2- pyridinecarboxylate), MCPA, MCPB, mecoprop, picloram, quinclorac, quinmerac, 2,3,6-TBA, triclopyr, and methyl 4-amino-3-chloro-6-(4-chloro-2-fluoro-3-methoxyphenyl)- 5-fluoro-2-pyridinecarboxylate. “EPSP synthase inhibitors” (b5) are chemical compounds that inhibit the enzyme, 5-enol-pyruvylshikimate-3-phosphate synthase, which is involved in the synthesis of aromatic amino acids such as tyrosine, tryptophan and phenylalanine. EPSP inhibitor herbicides are readily absorbed through plant foliage and translocated in the phloem to the growing points. Glyphosate is a relatively nonselective postemergence herbicide that belongs to this group. Glyphosate includes esters and salts such as ammonium, isopropylammonium, potassium, sodium (including sesquisodium) and trimesium (alternatively named sulfosate). “Photosystem I electron diverters” (b6) are chemical compounds that accept electrons from Photosystem I, and after several cycles, generate hydroxyl radicals. These radicals are extremely reactive and readily destroy unsaturated lipids, including membrane fatty acids and chlorophyll. This destroys cell membrane integrity, so that cells and organelles “leak”, leading to rapid leaf wilting and desiccation, and eventually to plant death. Examples of this second type of photosynthesis inhibitor include diquat, paraquat and 1-(2-carboxyethyl)-4-(2- pyrimidinyl)pyridazinium (CAS No.2285384-11-2) and salts and esters thereof. Of note is a photosystem I electron diverter selected from diquat and paraquat. “PPO inhibitors” (b7) are chemical compounds that inhibit the enzyme protoporphyrinogen oxidase, quickly resulting in formation of highly reactive compounds in plants that rupture cell membranes, causing cell fluids to leak out. Examples of PPO inhibitors include acifluorfen-sodium, azafenidin, benzfendizone, bifenox, butafenacil, carfentrazone, carfentrazone-ethyl, chlomethoxyfen, 3-[2-chloro-5-[3,6-dihydro-3-methyl-2,6-dioxo-4- (trifluoromethyl)-1(2H)-pyrimidinyl]-4-fluorophenyl]-4,5-dihydro-5-methyl-5-isoxazole- carboxylic ethyl ester (CAS No. 1949837-17-5), cinidon-ethyl, cyclopyranil, epyrifenacil, fluazolate, flufenoximacil, flufenpyr-ethyl, flumiclorac-pentyl, flumioxazin, fluoroglycofen-ethyl, fluthiacet-methyl, fomesafen, halosafen, lactofen, oxadiargyl, oxadiazon, oxyfluorfen, pentoxazone, profluazol, pyraclonil, pyraflufen-ethyl, saflufenacil, sulfentrazone, thidiazimin, trifludimoxazin (dihydro-1,5-dimehyl-6-thioxo-3-[2,2,7-trifluoro- 3,4-dihydro-3-oxo-4-(2-propyn-1-yl)-2H-1,4-benzoxazin-6-yl]-1,3,5-triazine-2,4(1H,3H)- dione), tiafenacil (methyl N-[2-[[2-chloro-5-[3,6-dihydro-3-methyl-2,6-dioxo-4-(trifluoro- methyl)-1(2H)-pyrimidinyl]-4-fluorophenyl]thio]-1-oxopropyl]-β-alaninate), and methyl 2-[2-[2-bromo-5-[3,6-dihydro-3-methyl-2,6-dioxo-4-(trifluoromethyl)-1(2H)-pyrimidinyl]-4- fluorophenoxy]phenoxy]-2-methoxyacetate, ethyl 3-[2-chloro-5-[3,6-dihydro-3-methyl-2,6- dioxo-4-(trifluoromethyl)-1(2H)-pyrimidinyl]-4-fluorophenyl]-4,5-dihydro-5-methyl-5- isoxazolecarboxylate.
“GS inhibitors” (b8) are chemical compounds that inhibit the activity of the glutamine synthetase enzyme, which plants use to convert ammonia into glutamine. Consequently, ammonia accumulates and glutamine levels decrease. Plant damage probably occurs due to the combined effects of ammonia toxicity and deficiency of amino acids required for other metabolic processes. The GS inhibitors include glufosinate and its esters and salts such as glufosinate-ammonium and other phosphinothricin derivatives, glufosinate-P ((2S)-2-amino- 4-(hydroxymethylphosphinyl)butanoic acid) and bilanaphos. “VLCFA elongase inhibitors” (b9) are herbicides having a wide variety of chemical structures, which inhibit the elongase. Elongase is one of the enzymes located in or near chloroplasts which are involved in biosynthesis of VLCFAs. In plants, very-long-chain fatty acids are the main constituents of hydrophobic polymers that prevent desiccation at the leaf surface and provide stability to pollen grains. Such herbicides include acetochlor, alachlor, anilofos, butachlor, cafenstrole, dimethachlor, dimethenamid, diphenamid, fenoxasulfone (3- [[(2,5-dichloro-4-ethoxyphenyl)methyl]sulfonyl]-4,5-dihydro-5,5-dimethylisoxazole), fentrazamide, flufenacet, indanofan, mefenacet, metazachlor, metolachlor, naproanilide, napropamide, napropamide-M ((2R)-N,N-diethyl-2-(1-naphthalenyloxy)propanamide), pethoxamid, piperophos, pretilachlor, propachlor, propisochlor, pyroxasulfone, and thenylchlor, including resolved forms such as S-metolachlor and chloroacetamides and oxyacetamides. “Auxin transport inhibitors” (b10) are chemical substances that inhibit auxin transport in plants, such as by binding with an auxin-carrier protein. Examples of auxin transport inhibitors include diflufenzopyr, naptalam (also known as N-(1-naphthyl)phthalamic acid and 2-[(1-naphthalenylamino)carbonyl]benzoic acid). “PDS inhibitors” (b11) are chemical compounds that inhibit carotenoid biosynthesis pathway at the phytoene desaturase step. Examples of PDS inhibitors include beflubutamid, diflufenican, fluridone, flurochloridone, flurtamone norflurzon and picolinafen. “HPPD inhibitors” (b12) are chemical substances that inhibit the biosynthesis of synthesis of 4-hydroxyphenyl-pyruvate dioxygenase. Examples of HPPD inhibitors include benquinotrione, benzobicyclon, benzofenap, bicyclopyrone (4-hydroxy-3-[[2-[(2- methoxyethoxy)methyl]-6-(trifluoromethyl)-3-pyridinyl]carbonyl]bicyclo[3.2.1]oct-3-en-2- one), bipyrazone, cyprafluone, fenpyrazone, fenquinotrione (2-[[8-chloro-3,4-dihydro-4-(4- methoxyphenyl)-3-oxo-2-quinoxalinyl]carbonyl]-1,3-cyclohexanedione), flusulfinam, iptriazopyrid, isoxachlortole, isoxaflutole, lancotrione, mesotrione, pyraquinate, pyrasulfotole, pyrazolynate, pyrazoxyfen, sulcotrione, tefuryltrione, tembotrione, tolpyralate (1-[[1-ethyl-4-[3-(2-methoxyethoxy)-2-methyl-4-(methylsulfonyl)benzoyl]-1H-pyrazol-5- yl]oxy]ethyl methyl carbonate), topramezone, 5-chloro-3-[(2-hydroxy-6-oxo-1-cyclohexen-1- yl)carbonyl]-1-(4-methoxyphenyl)-2(1H)-quinoxalinone, 4-(2,6-diethyl-4-methylphenyl)-5- hydroxy-2,6-dimethyl-3(2H)-pyridazinone, tripyrasulfone, 4-(4-fluorophenyl)-6-[(2-
hydroxy-6-oxo-1-cyclohexen-1-yl)carbonyl]-2-methyl-1,2,4-triazine-3,5(2H,4H)-dione, 5- [(2-hydroxy-6-oxo-1-cyclohexen-1-yl)carbonyl]-2-(3-methoxyphenyl)-3-(3-methoxypropyl)- 4(3H)-pyrimidinone, 2-methyl-N-(4-methyl-1,2,5-oxadiazol-3-yl)-3-(methylsulfinyl)-4- (trifluoromethyl)benzamide and 2-methyl-3-(methylsulfonyl)-N-(1-methyl-1H-tetrazol-5-yl)- 4-(trifluoromethyl)benzamide. “Deoxy-D-Xylulose Phosphate Synthase (DXP synthase) inhibitors” (b13) isoxazolidinone class of herbicides is classified according to its inhibition of the deoxy-d- xylulose phosphate synthase (DXP synthase) which is a component of the carotenoid biosynthetic pathway. Examples of DXP synthase inhibitors include bixlozone, broclozone and clomazone. Of note is a DXP synthase inhibitor selected from bixlozone and clomazone. “HST (homogentisate solanesyltransferase) inhibitors” (b14) disrupt a plant’s ability to convert homogentisate to 2-methyl-6-solanyl-1,4-benzoquinone, thereby disrupting carotenoid biosynthesis. Examples of HST inhibitors include cyclopyrimorate (6-chloro-3-(2- cyclopropyl-6-methylphenoxy)-4-pyridazinyl 4-morpholinecarboxylate), haloxydine, pyriclor, 3-(2-chloro-3,6-difluorophenyl)-4-hydroxy-1-methyl-1,5-naphthyridin-2(1H)-one, 7-(3,5-dichloro-4-pyridinyl)-5-(2,2-difluoroethyl)-8-hydroxypyrido[2,3-b]pyrazin-6(5H)-one and 4-(2,6-diethyl-4-methylphenyl)-5-hydroxy-2,6-dimethyl-3(2H)-pyridazinone. Another example of an HST inhibitor is 6-chloro-4-(2,7-dimethyl-1-naphthalenyl)-5-hydroxy-2- methyl-3(2H)-pyridazinone. HST inhibitors also include compounds of Formulae A and B.
wherein Rd1 is H, Cl or CF 3 ; Rd2 is H, Cl or Br; Rd3 is H or Cl; Rd4 is H, Cl or CF 3 ; Rd5 is CH 3 , CH 2 CH 3 or CH 2 CHF 2 ; and Rd6 is OH, or -OC(=O)-i-Pr; and Re1 is H, F, Cl, CH 3 or CH 2 CH 3 ; Re2 is H or CF 3 ; Re3 is H, CH 3 or CH 2 CH 3 ; Re4 is H, F or Br; Re5 is Cl, CH 3 , CF 3 , OCF 3 or CH 2 CH 3 ; Re6 is H, CH 3 , CH 2 CHF 2 or C≡CH; Re7 is OH, -OC(=O)Et, -OC(=O)-i-Pr or -OC(=O)-t-Bu; and Ae8 is N or CH. “Cellulose biosynthesis inhibitors” (b15) inhibit the biosynthesis of cellulose in certain plants. They are most effective when applied preemergence or early postemergence on young or rapidly growing plants. Examples of cellulose biosynthesis inhibitors include chlorthiamid,
dichlobenil, flupoxam, indaziflam (N2-[(1R,2S)-2,3-dihydro-2,6-dimethyl-1H-inden-1-yl]-6- (1-fluoroethyl)-1,3,5-triazine-2,4-diamine), isoxaben and triaziflam. “DHODH (dihydroorotate dehydrogenase) inhibitors” (b16) act through inhibiting catalysis of the fourth step (i.e., inhibition of dihydroorotate dehydrogenase) of the de-novo pyrimidine biosynthetic pathway. Inhibition of pyrimidine biosynthesis leads to the cessation of plant growth. The first two steps of de-novo pyrimidine biosynthesis occur in the chloroplast after which the product, N-carbamoyl aspartate, is shuttled to the cytoplasm where dihydroorotase resides, producing dihydroorotate, the DHODH substrate. Plant DHODH is a flavin-dependent enzyme residing on the outer surface of the inner mitochondrial membrane. Reducing equivalents pass from dihydroorotate via the tightly bound flavin cofactor to a ubiquinone acceptor molecule that subsequently exchanges with the ubiquinol pool of the membrane and ultimately links DHODH enzymatic activity with oxidative phosphorylation. The remaining steps of the pathway leading to pyrimidine nucleotides occur in the cytoplasm. DHODH inhibitors include a compound of Formula (b16A)
(b16A) wherein R12 is H, C1–C6 alkyl, C1–C6 haloalkyl or C4–C8 cycloalkyl; R13 is H, C1–C6 alkyl or C1–C6 alkoxy; Q1 is an optionally substituted ring system selected from the group consisting of phenyl, thienyl, pyridinyl, benzodioxolyl, naphthyl, naphthalenyl, benzofuranyl, furanyl, benzothiophenyl and pyrazolyl, wherein when substituted said ring system is substituted by 1 to 3 R14; Q2 is an optionally substituted ring system selected from the group consisting of phenyl, pyridinyl, benzodioxolyl, pyridinonyl, thiadiazolyl, thiazolyl, and oxazolyl, wherein when substituted said ring system is substituted by 1 to 3 R15; each R14 is independently halogen, C1–C6 alkyl, C1–C6 haloalkyl, C1–C6 alkoxy, C1–C6 haloalkoxy, C3–C8 cyaloalkyl, cyano, C1–C6 alkylthio, C1–C6 alkylsulfinyl, C1–C6 alkylsulfonyl, SF5, NHR17; or phenyl optionally substituted by 1 to 3 R16; or pyrazolyl optionally substituted by 1 to 3 R16;
each R15 is independently halogen, C1–C6 alkyl, C1–C6 haloalkyl, C1–C6 alkoxy, C1–C6 haloalkoxy, cyano, nitro, C1–C6 alkylthio, C1–C6 alkylsulfinyl, C1–C6 alkylsulfonyl; each R16 is independently halogen, C1–C6 alkyl or C1–C6 haloalkyl; R17 is C1–C4 alkoxycarbonyl. In one Embodiment wherein “DHODH inhbitors” (b16) also include a compound of Formula (b16A), it is preferred that R12 is H or C1–C6 alkyl; more preferably R12 is H or methyl. Preferrably R13 is H. Preferably Q1 is either a phenyl ring or a pyridinyl ring, each ring substituted by 1 to 3 R14; more preferably Q1 is a phenyl ring substituted by 1 to 2 R14. Preferably Q2 is a phenyl ring substituted by 1 to 3 R15; more preferably Q2 is a phenyl ring substituted by 1 to 2 R15. Preferably each R14 is independently halogen, C1–C4 alkyl, C1–C3 haloalkyl, C1–C3 alkoxy or C1–C3 haloalkoxy; more preferably each R14 is independently chloro, fluoro, bromo, C1–C2 haloalkyl, C1–C2 haloalkoxy or C1–C2 alkoxy. Preferrably each R15 is independently halogen, C1–C4 alkyl, C1–C3 haloalkoxy; more preferably each R15 is independently chloro, fluoro, bromo, C1–C2 haloalkyl, C1–C2 haloalkoxy or C1–C2 alkoxy. Specifically preferred as “DHODH inhibitors” (b16) include any one of the following (b16A-1) through (b16A-17): N-(2-fluorophenyl)-2-oxo-4-[3-(trifluoromethyl)phenyl]-3-pyrrolidinecarboxamide (b-16A- 1); N-(2,3-difluorophenyl)-2-oxo-4-[3-(trifluoromethyl)phenyl]-3-pyrrolidinecarboxamide (b16A-2); 2-oxo-4-[3-(trifluoromethyl)phenyl]-N-(2,3,4-trifluorophenyl)-3-pyrrolidinecarboxamide (b16A-3); N-(2-fluorophenyl)-1-methyl-2-oxo-4-[3-(trifluoromethyl)phenyl]-3-pyrrolidinecarboxamide (b16A-4); N-(2-fluorophenyl)-2-oxo-4-[4-(trifluoromethyl)phenyl]-3-pyrrolidinecarboxamide (b16A- 5); N-(2-fluorophenyl)-1-methyl-2-oxo-4-[4-(trifluoromethyl)phenyl]-3-pyrrolidinecarboxamide (b16A-6); N-(2,3-difluorophenyl)-2-oxo-4-[4-(trifluoromethyl)phenyl]-3-pyrrolidinecarboxamide (b16A-7); N-(2,3-difluorophenyl)-1-methyl-2-oxo-4-[4-(trifluoromethyl)phenyl]-3- pyrrolidinecarboxamide (b16A-8); 2-oxo-4-[4-(trifluoromethyl)phenyl]-N-(2,3,4-trifluorophenyl)-3-pyrrolidinecarboxamide (b16A-9); N-(2-fluorophenyl)-4-(4-fluorophenyl)-1-methyl-2-oxo-3-pyrrolidinecarboxamide (b16A- 10); N-(2,3-difluorophenyl)-4-(3,4-difluorophenyl)-2-oxo-3-pyrrolidinecarboxamide (b16A-11);
4-(3,4-difluorophenyl)-N-(2-fluorophenyl)-2-oxo-3-pyrrolidinecarboxamide (b16A-12); N-(2,4-difluorophenyl)-4-(3,5-difluorophenyl)-2-oxo-3-pyrrolidinecarboxamide (b16A-13); N-(2,3-difluorophenyl)-4-[3-(1-methylethyl)phenyl]-2-oxo-3-pyrrolidinecarboxamide (b16A-14); N-(2,3-difluorophenyl)-2-oxo-4-[6-(trifluoromethyl)-3-pyridinyl]-3-pyrrolidinecarboxamide (b16A-15); (3S,4S)-N-(2-fluorophenyl)-1-methyl-2-oxo-4-[3-(trifluoromethyl)phenyl]- 3- pyrrolidinecarboxamide (b16A-16) and (3S,4R)-N-(2,3-difluorophenyl)-1-methyl-4-[1-methyl-5-(trifluoromethyl)-1H-pyrazol-3-yl]- 2-oxo-3-pyrrolidinecaboxamide (b16A-17). An example of a DHODH inhibitor is tetflupyrolimet.
“Other herbicides” (b17) include herbicides that act through a variety of different modes of action such as mitotic disruptors (e.g., icafolin, flamprop-M-methyl and flamprop-M-isopropyl), organic arsenicals (e.g., DSMA, and MSMA), 7,8-dihydropteroate synthase inhibitors, chloroplast isoprenoid synthesis inhibitors and cell-wall biosynthesis inhibitors. Other herbicides include those herbicides having unknown modes of action or do not fall into a specific category listed in (b1) through (b16) or act through a combination of modes of action listed above. Examples of other herbicides include aclonifen, asulam, amitrole, bromobutide, cinmethylin, cumyluron, cyclopyrimorate (6-chloro-3-(2-cyclopropyl- 6-methylphenoxy)-4-pyridazinyl 4-morpholinecarboxylate), daimuron, difenzoquat, dimesulfazet, etobenzanid, fluometuron, flurenol, fosamine, fosamine-ammonium, dazomet, dymron, ipfencarbazone (1-(2,4-dichlorophenyl)-N-(2,4-difluorophenyl)-1,5-dihydro-N-(1- methylethyl)-5-oxo-4H-1,2,4-triazole-4-carboxamide), metam, methyldymron, oleic acid, oxaziclomefone, pelargonic acid, pyributicarb, 2,5-anhydro-3,4-dideoxy-4-[[[(5S)-3-(3,5- difluorophenyl)-5-ethenyl-4,5-dihydro-5-isoxazolyl]carbonyl]amino]-threo-pentonic methyl ester (CAS No. 27499989-21-6) and 5-[[(2,6-difluorophenyl)methoxy]methyl]-4,5-dihydro- 5-methyl-3-(3-methyl-2-thienyl)isoxazole. “Other herbicides” (b17) also include a compound of Formula (b16B)
wherein R18 is H, C1–C6 alkyl, C1–C6 haloalkyl or C4–C8 cycloalkyl; each R19 is independently halogen, C1–C6 haloalkyl or C1–C6 haloalkoxy; p is an integer of 0, 1, 2 or 3; each R20 is independently halogen, C1–C6 haloalkyl or C1–C6 haloalkoxy; and q is an integer of 0, 1, 2 or 3. In one Embodiment wherein “other herbicides” (b17) also include a compound of Formula (b16B), it is preferred that R18 is H, methyl, ethyl or propyl; more preferably R18 is H or methyl; most preferably R18 is H. Preferrably each R19 is independently chloro, fluoro, C1– C3 haloalkyl or C1–C3 haloalkoxy; more preferably each R19 is independently chloro, fluoro, C1 fluoroalkyl (i.e., fluoromethyl, difluoromethyl or trifluoromethyl) or C1 fluoroalkoxy (i.e., trifluoromethoxy, difluoromethoxy or fluoromethoxy). Preferably each R20 is independently chloro, fluoro, C1 haloalkyl or C1 haloalkoxy; more preferably each R20 is independently chloro, fluoro, C1 fluoroalkyl (i.e., fluoromethyl, difluoromethyl or trifluromethyl) or C1 fluoroalkoxy (i.e., trifluoromethoxy, difluoromethoxy or fluoromethoxy). Specifically preferred as “other herbicides” (b17) include any one of the following (b16B-1) through
Another Embodiment of “other herbicides” (b17) also includes a compound of Formula (b16C),
wherein R1 is Cl, Br or CN; and R2 is C(=O)CH 2 CH 2 CF 3 , CH 2 CH 2 CH 2 CH 2 CF 3 or 3-CHF 2 -isoxazol-5-yl. An example of an other herbicide is rimisoxafen. “Herbicide safeners” (b18) are substances added to a herbicide formulation to eliminate or reduce phytotoxic effects of the herbicide to certain crops. These compounds protect crops from injury by herbicides but typically do not prevent the herbicide from controlling undesired vegetation. Examples of herbicide safeners include but are not limited to benoxacor, cloquintocet-mexyl, cumyluron, cyometrinil, cyprosulfamide, daimuron, dichlormid, dicyclonon, dietholate, dimepiperate, fenchlorazole-ethyl, fenclorim, flurazole, fluxofenim, furilazole, isoxadifen-ethyl, mefenpyr-diethyl, mephenate, methoxyphenone, naphthalic anhydride, oxabetrinil, N-(aminocarbonyl)-2-methylbenzenesulfonamide, N- (aminocarbonyl)-2-fluorobenzenesulfonamide, 1-bromo-4-[(chloromethyl)sulfonyl]benzene (BCS), 4-(dichloroacetyl)-1-oxa-4-azospiro[4.5]decane (MON 4660), 2-(dichloromethyl)-2- methyl-1,3-dioxolane (MG 191), ethyl 1,6-dihydro-1-(2-methoxyphenyl)-6-oxo-2-phenyl-5- pyrimidinecarboxylate, 2-hydroxy-N,N-dimethyl-6-(trifluoromethyl)pyridine-3-carboxamide, 3-oxo-1-cyclohexen-l-yl 1-(3,4-dimethylphenyl)-l,6-dihydro-6-oxo-2-phenyl-5-pyrimidine- carboxylate, 2,2-dichloro-1-(2,2,5-trimethyl-3-oxazolidinyl)-ethanone and 2-methoxy-N-[[4- [[(methylamino)carbonyl]amino]phenyl]sulfonyl]-benzamide. Preferred for better control of undesired vegetation (e.g., lower use rate such as from greater-than-additive effects, broader spectrum of weeds controlled, or enhanced crop safety) or for preventing the development of resistant weeds are mixtures of a compound of this invention with a herbicide selected from the group consisting of 4-amino-3-chloro-5-fluoro- 6-(7-fluoro-1H-indol-6-yl)-2-pyridinecarboxylic 2-propyn-1-yl ester (CAS No. 2251111-17-
6), 4-amino-3-chloro-5-fluoro-6-(7-fluoro-1H-indol-6-yl)-2-pyridinecarboxylic cyanomethyl ester (CAS No.2251111-18-7), 2,5-anhydro-3,4-dideoxy-4-[[[(5S)-3-(3,5-difluorophenyl)-5- ethenyl-4,5-dihydro-5-isoxazolyl]carbonyl]amino]-threo-pentonic methyl ester (CAS No. 27499989-21-6), atrazine, azimsulfuron, beflubutamid, S-beflubutamid, beflubutamid-M, bixlozone, broclozone, benzisothiazolinone, 1-(2-carboxyethyl)-4-(2-pyrimidinyl)- pyridazinium (CAS No.2285384-11-2) and salts thereof, carfentrazone, carfentrazone-ethyl, chlorimuron, chlorimuron-ethyl, 3-[2-chloro-5-[3,6-dihydro-3-methyl-2,6-dioxo-4- (trifluoromethyl)-1(2H)-pyrimidinyl]-4-fluorophenyl]-4,5-dihydro-5-methyl-5-isoxazole- carboxylic ethyl ester (CAS No.1949837-17-5), chlorsulfuron-methyl, clomazone, clopyralid potassium, cloransulam-methyl, 2-[(2,4-dichlorophenyl)methyl]-4,4-dimethyl- isoxazolidinone, 2-[(2,5-dichlorophenyl)methyl]-4,4-dimethyl-isoxazolidinone, ethamet- sulfuron-methyl, florasulam, flumetsulam, 4-(4-fluorophenyl)-6-[(2-hydroxy-6-oxo-1- cyclohexen-1-yl)carbonyl]-2-methyl-1,2,4-triazine-3,5-(2H,4H)-dione, florasulam, flupyrsulfuron, flupyrsulfuron-methyl, fluthiacet, fluthiacet-methyl, fomesafen, imazethapyr, lenacil, mesotrione, metolachlor, S-metolachlor, metproxybicyclone, metribuzin, metsulfuron-methyl, nicosulfuron, pethoxamid, picloram, pinoxaden, pyroxasulfone, pyroxsulam, quinclorac, rimisoxafen, rimsulfuron, sulfentrazone, tembotrione, tetflupyrolimet, thifensulfuron, thifensulfuron-methyl, triflusulfuron-methyl, tribenuron and tribenuron-methyl. One or more of the following methods and variations as described in Schemes 1–22 can be used to prepare the compounds of Formula 1. The definitions of A, R1a, R1b, R1c, R2, R3a, R3b, R4a, R4b, X1, X2, n, p and q in the compounds of Formulae 1–38 below are as defined above in the Summary of the Invention unless otherwise noted. Compounds of Formulae 1a, 1b, 1c, 1d, 1f, 8a, 8b, 10a, 10b, 10c, 10d, 11a, 31a, 31b, 32a and 32b are various subsets of the compounds of Formulae 1, 8, 10, 11, 31 and 32; and all substituents for Formulae 1a, 1b, 1c, 1d, 1f, 8a, 8b, 10a, 10b, 10c, 10d, 11a, 31a, 31b, 32a and 32b are as defined above for Formula 1 unless otherwise noted in the disclosure including the schemes. Stereochemically enriched compounds of Formula 1' and 1'' can be obtained from mixtures containing compounds of Formula 1' and 1'' through the utilization of well-known chiral chromatography separation methods. For extensive reviews of chiral separation methods see the Chiral Separations: Methods and Protocols (Methods in Molecular Biology), 2nd ed., 2013 Edition, by Gerhard K. E. Scriba (Editor). As shown in Scheme 1, compounds of Formula 1a (i.e., a compound of Formula 1 wherein Y is S) can be prepared by treatment of compounds of Formula 1b (i.e., a compond of Formula 1 wherein Y is O) with a thionation reagent such as Lawesson’s reagent, tetraphosphorus decasulfide or diphosphorus pentasulfide in an appropriate solvent (e.g., tetrahydrofuran or toluene) at temperatures typically ranging from 0 °C to the reflux temperature of the solvent.
Scheme 1
Compounds of Formula 1c (i.e., a compound of Formula 1b wherein Z is NR3d and R3d is other than H) can be prepared using method depicted in Scheme 2 wherein a compound of Formula 1d (i.e., a compound of Formula 1b where Z is NH) reacts with an alkylating agent or other suitable electrophile (e.g., acyl chloride or anhydride, sulfonyl chloride or anhydride) in the presence of a base, such as sodium hydride in a solvent such as N,N-dimethylformamide. Alternative bases for Scheme 2 reactions include, but are not limited to, potassium carbonate, sodium hydroxide, pyridine and triethylamine, and alternative solvents include dimethyl sulfoxide, tetrahydrofuran, ethyl acetate and dichloromethane. Methods analogous to those described in J. Med. Chem.2018, 61, 4317 and Eur. J. Med. Chem.2020, 188, 111987 may be used. Scheme 2
wherein Z is NR3d; and wherein Z is NR3d; and is H R3d is other than H R3d Compounds of Formula 1b can be prepared using standard cross-coupling reactions, such as those described in Science of Synthesis: Cross Coupling and Heck-Type Reactions 1, Molander, G. A. (volume editor), Thieme (2013) and Science of Synthesis: Cross Coupling and Heck-Type Reactions 2, Wolfe, J. P. (volume editor), Thieme (2013); and references cited therein. For example, compounds of Formula 1e (i.e., a compound of Formula 1b wherein A is bonded to the rest of the molecule through a nitrogen ring member of A) can be prepared from compounds of Formula 2 (wherein Q is a halogen or pseudohalogen such as Cl, Br, I or OTf) via palladium- or copper-mediated coupling with heterocycles of Formula 3 (wherein H is connected to a nitrogen ring member of A), as shown in Scheme 3. Ullmann-type coupling
conditions, such as those described in J. Org. Chem.2004, 69, 5578-5587, are often suitable and are typically catalyzed by a copper salt such as copper(I) iodide or copper(I) oxide and a ligand, such as trans-N,N′-dimethylcyclohexane-1,2-diamine, trans-1,2-diaminocyclohexane, N,N′-dimethylethylenediamine, 1,10-phenanthroline, 8-quinolinol, (S)-proline or 2-picolinic acid, in the presence of a base (e.g., potassium carbonate, cesium carbonate or potassium phosphate) in an appropriate solvent (e.g., N,N-dimethylformamide, dimethyl sulfoxide, N,N- dimethylacetamide, toluene, 1,4-dioxane or acetonitrile). Temperatures between ambient temperature and 150 °C are generally appropriate for the reaction. Palladium-catalyzed coupling conditions may also be suitable for some heterocycles. Appropriate palladium catalysts include but are not limited to tetrakis(triphenylphosphine)palladium(0), [1,1′-bis(diphenylphosphino)ferrocene]dichloropalladium(II), bis(triphenylphosphine)- palladium(II) dichloride, palladium(II) acetate or tris(dibenzylideneacetone)dipalladium(0). In some cases, the addition of a ligand is beneficial, including but not limited to, 2,2′- bis(diphenylphosphino)-1,1′-binaphthalene (BINAP), 2-dicyclohexylphosphino-2′,6′- dimethoxybiphenyl (SPhos), 2-dicyclohexylphosphino-2′,4′,6′-triisopropylbiphenyl (XPhos), 4,5-bis(diphenylphosphino)-9,9-dimethylxanthene (Xantphos), 2-(di-tert-butylphosphino)- biphenyl (JohnPhos), 2-dicyclohexylphosphino-2',6'-diisopropoxy-1,1'-biphenyl (RuPhos) or 2-di-tert-butylphosphino-3,4,5,6-tetramethyl-2′,4′,6′-triisopropyl-1,1′-biphenyl (Me4-t-Butyl- Xphos). These reactions are generally run in the presence of a base (e.g., sodium tert-butoxide, lithium bis(trimethylsilyl)amide, cesium carbonate, potassium carbonate or potassium phosphate tribasic) and a solvent (e.g., tetrahydrofuran, 1,4-dioxane, toluene or tert-butanol) at temperatures generally ranging from ambient temperature to 150 °C. For reviews of these methods, see: Chem. Rev.2008, 108, 3054-3131; Chem. Sci.2010, 1, 13-31; Beilstein J. Org. Chem.2011, 7, 59-74; Chem. Rev.2016, 116, 12564-12649; Angew. Chem. Int. Ed.2017, 56, 16136-16179 and Tetrahedron 2019, 75, 4199-4211. One skilled in the art will recognize that when more than one nitrogen atom is present in the heterocycle of Formula 3, formation of regioisomers is possible and standard purification methods such as chromatography, can generally be used to separate the mixture. Heterocycles of Formula 3 are generally commercially available or known in the literature. Alternatively, when the aryl ring is sufficiently activated, for example when X1, X3 or X4 is N or when R2 is a suitably positioned electron-withdrawing group, compounds of Formula 1e can generally be prepared without the addition of a metal catalyst. This is achieved by reaction of compounds of Formula 2 (wherein Q is a halogen such as F or Cl) with heterocycles of Formula 3 in the presence of a base (e.g., potassium carbonate, sodium carbonate, cesium carbonate, potassium phosphate, sodium hydroxide, potassium tert- butoxide, sodium hydride or potassium hydride) in an appropriate solvent (e.g., dimethyl sulfoxide, N,N-dimethylformamide, N-methyl-2-pyrrolidone, tetrahydrofuran, 1,4-dioxane, 1,2-dimethoxyethane, diglyme, acetonitrile or toluene). Temperatures ranging from 0 °C to
200 °C are generally suitable for the reaction. For examples of this reaction in the literature, see: WO 2012/054510; WO 2020/207941 and J. Med. Chem.2014, 57, 10013-10030. In some instances, compounds of Formula 1e may be accessed using standard heterocyclic synthesis procedures known to those skilled in the art. For appropriate methods, see Science of Synthesis, Volumes 12-13, Neier, R., Storr, R. C. and Gilchrist, T. L. (volume editors), Thieme (2002-2003); Comprehensive Heterocyclic Chemistry IV, Volumes 4-6, Black, D. S., Cossy, J. and Stevens, C. V. (editors-in-chief), Elsevier (2022); WO 2015/160636 and WO 2017/205709; and references cited therein. Scheme 3
2
wherein A is bonded to the rest of the molecule through a nitrogen ring member of A As shown in Scheme 4, compounds of Formula 1f (i.e., a compound of Formula 1b wherein A is bonded to the rest of the molecule through a carbon ring member of A), can be prepared by well-known metal-catalyzed cross-coupling reactions between a heterocycle of Formula 4 (wherein Q is a halogen or pseudohalogen such as Cl, Br, I or OTf and is connected to A through a carbon ring member) and an organometallic compound of Formula 5 (wherein M is a transmetalating group), such as, but not limited to, a boronic acid (e.g., M is B(OH)2), boronate ester (e.g., M is B(–O(CMe2)2O–) or organotin reagent (e.g., M is Sn(n-Bu)3, SnMe3). The metal catalysts used in these couplings include, but are not limited to, tetrakis(triphenylphosphine)palladium(0), [1,1′-bis(diphenylphosphino)ferrocene]dichloro- palladium(II), bis(triphenylphosphine)palladium(II) dichloride, palladium(II) acetate and tris(dibenzylideneacetone)dipalladium(0). In some cases, the addition of a ligand is beneficial, including but not limited to, 2-dicyclohexylphosphino-2′,4′,6′-triisopropylbiphenyl (XPhos), 2-dicyclohexylphosphino-2′,6′-dimethoxybiphenyl (SPhos), tricyclohexylphosphine and tri(2-furyl)phosphine. Generally, these reactions are run in solvents such as dimethyl sulfoxide, N,N-dimethylformamide, N-methyl-2-pyrrolidone, toluene, 1,2-dimethoxyethane, 1,4-dioxane, tetrahydrofuran, acetonitrile or ethanol and temperatures generally range from ambient temperature to 150 °C. One skilled in the art will recognize that the reaction conditions will depend on the organometallic species used in the reaction, for example, when the compound of Formula 5 is a boron reagent, a base, often an aqueous base, is required.
Suitable bases include potassium carbonate, sodium carbonate, cesium carbonate, sodium bicarbonate or potassium phosphate. When the compound of Formula 5 is a tin reagent, a base is not required. For reviews of transition metal-catalyzed cross-coupling reactions, see: E. Negishi, Handbook of Organopalladium Chemistry for Organic Synthesis, John Wiley and Sons, Inc., New York, 2002; N. Miyaura, Cross-Coupling Reactions: A Practical Guide, Springer, New York, 2002; H. C. Brown et al., Organic Synthesis via Boranes, Vol.3, Aldrich Chemical Co., Milwaukee, WI, 2002; Suzuki et al., Chem. Rev. 1995, 95, 2457-2483; Molander et al., Acc. Chem. Res.2007, 40, 275-286 and Chem. Soc. Rev.2013, 42, 5270. For relevant examples of reactions in the literature using boron reagents, see: WO 2012/137982, WO 2015/017610, WO 2016/040223 and WO 2020/182990. For relevant examples of reactions in the literature using tin reagents, see: WO 2019/195810 and WO 2021/242677. Heterocycles of Formula 4 are generally commercially available or known in the literature. Scheme 4
cule through a carbon ring member One skilled in the art will recognize that in some instances, due to the availability or stability of the required reagents, it may be beneficial to reverse the polarity of the coupling partners, as shown in Scheme 5. In this scenario, compounds of Formula 2 (wherein Q is a halogen or pseudohalogen such as Cl, Br, I or OTf) are coupled with heterocycles of Formula 6 (wherein M is a transmetalating group such as, but not limited to, B(OH)2, B(–O(CMe2)2O-), BF3K, Sn(n-Bu)3, SnMe3 or ZnBr; and M is connected to a carbon ring member). For relevant examples in the literature, see: WO 2012/063207 and WO 2019/162323. For examples of cross-couplings of (hetero)aryl halides with alkyl coupling partners, see: Chem. Rev. 2011, 111, 1417-1492; ACS Med. Chem. Lett.2020, 11, 597-604 and J. Org. Chem.2021, 86, 10380- 10396; and references cited therein. Heterocycles of Formula 6 are generally commercially available or known in the literature. In some instances, compounds of Formula 1f may be more readily accessed using standard heterocyclic synthesis procedures known to those skilled in the art. For appropriate methods, see Science of Synthesis, Volumes 11-13 and 15, Schaumann, E., Neier, R., Storr, R. C.; Gilchrist, T. L. and Black, D. S. (volume editors), Thieme (2001-2004) and Comprehensive
Heterocyclic Chemistry IV, Volumes 4-7, Black, D. S., Cossy, J. and Stevens, C. V. (editors- in-chief), Elsevier (2022); and references cited therein. Scheme 5
wherein A is bonded to the rest of the molecule through a carbon ring member As shown in Scheme 6, organometallic compounds of Formula 5 (wherein M is a transmetalating group such as, but not limited to, B(–O(CMe2)2O–), Sn(n-Bu)3 or SnMe3) can be prepared from compounds of Formula 2 (wherein Q is a halogen or pseudohalogen such as Cl, Br, I or OTf) using well-known metal-catalyzed cross-coupling reactions. For example, pinacol boronic esters (wherein M is B(–O(CMe2)2O–), can generally be prepared using palladium-catalyzed borylation conditions, such as those described in J. Org. Chem.1995, 60, 7508-7510 and J. Org. Chem. 2021, 86, 103-109; and references cited therein. Typically, a compound of Formula 2 is treated with bis(pinacolato)diboron in the presence of a palladium catalyst like [1,1′-bis(diphenylphosphino)ferrocene]dichloropalladium(II) and a base such as potassium acetate in a solvent such as dioxane or dimethyl sulfoxide, at temperatures ranging from ambient temperature to the reflux temperature of the solvent. Similarly, trialkyl tin reagents (e.g., wherein M is SnMe3 or SnBu3) can generally be prepared from a compound of Formula 2 by treatment with a stannane such as hexamethylditin or hexabutylditin in the presence of a palladium catalyst like tetrakis(triphenylphosphine)palladium(0) in a solvent like dioxane or toluene, at temperatures ranging from ambient temperature to the reflux temperature of the solvent. For examples of this reaction in the literature, see: WO 03/077918 and WO 2021/050964. For additional details on preparation and use of organoboron and organotin reagents, see Science of Synthesis, Volumes 5-6, Moloney, M. G. and Kaufmann, D. E. (volume editors), Thieme (2002-2004); and references cited therein.
Scheme 6
As shown in Scheme 7, compounds of Formulae 1 or 2 can be prepared by reacting a carboxylic acid of Formula 7 (where R a is H) with an amine of Formula 8. The reaction proceeds via activation of the carboxylic acid of Formula 7 followed by reaction with the amine of Formula 8. The carboxylic acid can be activated with a coupling reagent or by conversion of the carboxylic acid to an acid halide, such as an acid chloride. For example, compounds of Formulae 7 and 8 can be reacted in the presence of a coupling reagent such as propylphosphonic anhydride (T3P), N-(3-dimethylaminopropyl)-N′-ethylcarbodiimide hydrochloride (EDC), 1,1′-carbonyldiimidazole (CDI), 2-chloro-1-methylpyridinium iodide (Mukaiyama’s reagent) or the combination of N,N,N′,N′-tetramethylchloroformamidinium hexafluorophosphate (TCFH) and N-methylimidazole (NMI). Polymer supported reagents, such as polymer-supported cyclohexylcarbodiimide, are also suitable. These reactions are typically run in an appropriate solvent (e.g., dichloromethane, 1,2-dichloroethane, ethyl acetate, acetonitrile or N,N-dimethylformamide) in the presence of a base (e.g., triethylamine, N,N-diisopropylethylamine or pyridine), optionally with a catalytic amount of 4-(dimethylamino)pyridine (DMAP), at temperatures ranging from 0 °C to the reflux temperature of the solvent. Alternatively, a carboxylic acid of Formula 7 can be converted to an acid chloride by treatment with a reagent such as thionyl chloride, oxalyl chloride, phosphoryl chloride, phosphorus trichloride or phosphorus pentachloride, either neat or in an appropriate solvent (e.g., dichloromethane, 1,2-dichloroethane or toluene) and optionally with a catalytic amount of N,N-dimethylformamide, at temperatures ranging from 0 °C to the reflux temperature of the solvent. Subsequent reaction of the acid chloride with the amine of Formula 8 generally occurs in the presence of a base (e.g., triethylamine, N,N- diisopropylethylamine or pyridine), optionally with a catalytic amount of 4- (dimethylamino)pyridine, in an appropriate solvent (e.g., tetrahydrofuran, dioxane, toluene, dichloromethane, chloroform, 1,2-dichloroethane or ethyl acetate) at temperatures ranging from 0 °C to the reflux temperature of the solvent. For relevant examples of this reaction in the literature, see: J. Med. Chem. 2012, 55, 10475-10489; WO2010/049302; WO 2016/001631 and Org. Lett.2018, 20, 4218-4222. Carboxylic acids of Formula 7 are generally
commercially available or known in the literature. In some cases, the corresponding acid chlorides are also commercially available. Where Q is A, such carboxylic acids of Formula 7 can be prepared using the reaction conditions described in Schemes 2, 3 and 4 starting from carboxylic acids or esters of Formula 7 where Q is a halogen or pseudohalogen. Where esters are used as starting materials (i.e., Ra is C1-C4 alkyl), a second step may be required to convert the ester to the carboxylic acid using standard conditions known to those skilled in the art (e.g., hydrolysis with sodium hydroxide). Alternatively, esters of Formula 7 (wherein R a is C1-C4 alkyl, typically methyl or ethyl) may undergo direct amidation with amines of Formula 8 by treatment with trimethylaluminum in a solvent like toluene, at temperatures ranging from 0 °C to the reflux temperature of the solvent. For an example of this reaction in the literature, see: WO 2002/074764.
wherein Q is A, F, Cl, Br, I or OTf; ( is H or C1-C4 a 1 wherein and R a lkyl 2 ( wherein
Several methods useful for preparing amines of Formula 8 are well-known in the literature. In addition, some amines of Formula 8 are commercially available. One method to prepare amines of Formula 8a (i.e., a compound of Formula 8 wherein the two R3b groups attached to the carbon at the α-position to the nitrogen are H) is illustrated in Scheme 8. In the method of Scheme 8, lactams of Formula 9 are treated with a reducing agent, such as borane (e.g., as a tetrahydrofuran or dimethylsulfide adduct), lithium aluminum hydride or diisobutylaluminum hydride in an appropriate solvent (e.g., tetrahydrofuran or toluene) at temperatures typically ranging from 0 °C to the reflux temperature of the solvent. For relevant examples of this reaction in the literature, see WO 2015/095795; WO 2015/136091; WO 2016/081692 and Org. Lett. 2019, 21, 4340. Some lactams of Formula 9 are commercially available or known in the literature.
Scheme 8
As shown in Scheme 9, lactams of Formula 9 can be prepared from compounds of Formula 10 (wherein Ra is C1-C4 alkyl, typically methyl or ethyl) by a nitro reduction followed by cyclization which occurs during reduction or upon heating. This reaction is readily achieved under a range of conditions, such as iron metal in the presence of an acid like acetic acid, hydrochloric acid or aqueous ammonium chloride, optionally with a solvent such as methanol, ethanol, ethyl acetate or N,N-dimethylformamide at temperatures ranging from ambient temperature to the reflux temperature of the solvent. Other suitable conditions include zinc metal with acetic acid or aqueous ammonium chloride, and stannous chloride in aqueous hydrochloric acid or ethanol. Alternatively, the reaction can be achieved using a transition metal catalyst, such as palladium on carbon, platinum oxide or Raney nickel under an atmosphere of hydrogen, in an appropriate solvent (e.g., methanol, ethanol, ethyl acetate or tetrahydrofuran). Temperatures typically range from ambient temperature to 80 °C. This reaction can generally be done in a Parr hydrogenator. For relevant examples of this reaction in the literature, see: WO 2013/130660; WO 2015/095795; WO 2022/204336 and Angew. Chem. Int. Ed.2014, 53, 6126-6130. Scheme 9
As shown in Scheme 10, compounds of Formula 10a (i.e., compounds of Formula 10 wherein Z is O or NRb, and Rb is a suitable electron-withdrawing group like COCF3, SO2Me or SO2CF3) can be prepared by Mitsunobu reaction of compounds of Formula 11 (wherein Z is O or NRb, and where Rb is a suitable electron-withdrawing group like COCF3, SO2Me or SO2CF3) with α-hydroxy esters of Formula 12 (wherein Ra is C1-C4 alkyl, typically methyl
or ethyl). Typical reaction conditions include triphenylphosphine and an azodicarboxylate such as diethyl azodicarboxylate or diisopropyl azodicarboxylate, in a solvent like tetrahydrofuran or dichloromethane at temperatures ranging from about –10 °C to the reflux temperature of the solvent. Alternatively, compounds of Formula 10b (i.e., compounds of Formula 10 wherein Z is O or NR3d) can be prepared by reaction of compounds of Formula 11 (wherein Z is O or NR3d) with α-halo esters of Formula 13 (wherein LG is a leaving group such as Cl or Br and Ra is C1-C4 alkyl, typically methyl or ethyl) in the presence of a base, such as potassium carbonate or cesium carbonate, in a solvent like N,N-dimethylformamide, acetone or 1,4-dioxane, at temperatures typically ranging from 0 °C to the reflux temperature of the solvent. In some cases, the addition of sodium iodide is beneficial. For relevant examples of these reactions in the literature, see: WO 2008/146871, US 2015/0148342, WO 2017/205536, Bioorg. Med. Chem.2007, 15, 5912-5949; Bioorg. Med. Chem.2013, 23, 4501-4505; and Bioorg. Med. Chem.2023, 78, 117152. Nitrobenzenes, α-hydroxy esters and α-halo esters of Formulae 11, 12 and 13 are generally commercially available or known in the literature. Scheme 10
As shown in Scheme 11, compounds of Formula 10c (i.e., a compound of Formula 10 wherein Z is CH2) can be prepared by the reaction of a compound of Formula 14 (wherein LG is a leaving group such as Cl or Br) with an ester of Formula 15 (wherein Ra is C1-C4 alkyl, typically methyl or ethyl) in the presence of a base, such as lithium diisopropylamide or lithium bis(trimethylsilyl)amide, in a solvent like tetrahydrofuran, at temperatures typically ranging from –78 °C to ambient temperature. Alternatively, the reaction of the compound of Formula 14 (wherein LG is a leaving group such as Cl or Br) with a diester of Formula 16 can
provide a compound of Formula 17 by treatment with a base, such as sodium hydride or cesium carbonate, in an appropriate solvent like tetrahydrofuran or N,N-dimethylformamide, at temperatures typically ranging from 0 °C to the reflux temperature of the solvent. Subsequent dealkoxycarbonylation provides a compound of Formula 10d (i.e., compounds of Formula 10 wherein Z is CH2 and R3b is H), which may be achieved by heating the compound of Formula 17 in a solvent system like N,N-dimethylformamide/water or dimethyl sulfoxide/water at temperatures ranging from 100 °C to 200 °C. When Ra is methyl, Krapcho- type decarboxylation conditions can also be appropriate, by heating the compound of Formula 17 in the presence of a salt like sodium chloride or lithium chloride, in a solvent system like dimethyl sulfoxide/water or N,N-dimethylformamide/water at temperatures ranging from 100 °C to 200 °C. An alternative method to convert a compound of Formula 17 to a compound of Formula 10d involves ester hydrolysis (e.g., potassium hydroxide in ethanol) followed by thermal decarboxylation of the resulting carboxylic acid (e.g., reflux in a solvent like dioxane, toluene or xylene). In some instances, acidic conditions may be more appropriate (e.g., reflux in acetic acid or hydrochloric acid). One skilled in the art will recognize that under some conditions, hydrolysis of the remaining ester may occur; in these instances, re-esterification using standard conditions (e.g., thionyl chloride in methanol) may be necessary. For relevant examples of these reactions in the literature, see: WO 2015/086506; WO 2011/162267; J. Agric. Food Chem.1995, 43, 1929-1934 and Synthesis 2013, 45, 2699- 2705. Compounds of Formulae 14, 15 and 16 are generally commercially available or known in the literature. Scheme 11
Lactams of Formula 9a (i.e., compounds of Formula 9 wherein Z is NH) can be prepared by the reaction of 2-bromoanilines of Formula 18 with amino acids of Formula 19 as depicted in Scheme 12. Examples of procedures analogous to those depicted in Scheme 12 can be found in J. Med. Chem.2018, 61, 4317, where copper(I) chloride is used as a catalyst in the presence of a ligand (e.g., N,N′-dimethylethylenediamine) and a base (e.g., 1,8-diaza- bicyclo[5.4.0]undec-7-ene) in a solvent like dimethyl sulfoxide with heating. Alternative bases for the method of Scheme 12 include potassium phosphate and potassium carbonate. 2- Bromoanilines and amino acids of Formulae 18 and 19 are generally commercially available or known in the literature. Scheme 12
Amines of Formula 8b (i.e., compounds of Formula 8 wherein Z is O) can be prepared according to the sequence shown in Schemes 13 and 14. As shown in Scheme 13, β-aminoalcohols of Formula 21 (wherein Rb is H or an appropriate protecting group such as tosyl (i.e., Ts) or benzyl (i.e., Bn); and X is a halogen or pseudohalogen such as F, Cl, Br, I or OTs) can be prepared by ring opening of epoxides of Formula 20 and subsequent nucleophilic attack by anilines of Formula 22. This reaction can generally be achieved by heating the aniline and epoxide either neat or in an appropriate solvent (e.g., ethanol or N,N- dimethylformamide) at temperatures generally ranging from 40 °C to 180 °C. Optionally, a base (e.g., potassium carbonate or sodium hydride), Lewis acid (e.g., ytterbium(III) trifluoromethanesulfonate or lithium bromide) or phase transfer catalyst (e.g., benzyltriethylammonium chloride or tetrabutylammonium bromide) can be added to facilitate the reaction. One skilled in the art will recognize that the epoxide substituents and reaction conditions impact the regioselectivity of the reaction, for example acidic conditions may reverse the regioselectivity of epoxide ring-opening. For relevant examples of these reactions in the literature, see: Ind. Eng. Chem. Res.2003, 42, 680-686; Eur. J. Org. Chem.2004, 3597- 3600; J. Het. Chem.2010, 47, 1406-1410 and WO 2015/095792; and references cited therein. Anilines and epoxides of Formulae 22 and 20 are generally commercially available or known in the literature.
As shown in Scheme 14, N-tosyl protected amines of Formula 23 can be prepared from β-aminoalcohols of Formula 21a (i.e., compounds of Formula 21 wherein Rb is Ts and X is typically F but in some instances is OTs) by treatment with a base such as, but not limited to, sodium hydroxide, potassium tert-butoxide or sodium hydride, optionally in the presence of a phase transfer catalyst like tetrabutylammonium bromide, typically in a solvent such as tetrahydrofuran at temperatures ranging from ambient temperature to the reflux temperature of the solvent. For examples of this reaction in the literature, see: Ind. Eng. Chem. Res.2003, 42, 680-686; WO 2015/095792 and J. Mol. Catal. A Chem. 2008, 288, 28-32. Amines of Formula 8b can be prepared by removal of the N-tosyl protecting group. This can be achieved under reducing conditions, for example, by treatment with magnesium metal in methanol at temperatures ranging from 0 °C to the reflux temperature of the solvent, optionally with sonication. Alternatively, the reaction can be achieved using acidic hydrolysis conditions, for example, treatment with sulfuric acid either neat or in a solvent like dichloromethane, at temperatures generally ranging from 0 °C to the reflux temperature of the solvent. For relevant examples of these reactions in the literature, see: Chem. Commun. 1999, 2095-2096; WO 2015/095792 and J. Org. Chem.2021, 86, 16573-16581; and references cited therein.
Alternatively, as shown in Scheme 15, amines of Formulae 8b (wherein Rb is H) or protected amines of Formula 24 (wherein Rb is a suitable protecting group like Ts or Bn) can be prepared from β-aminoalcohols of Formula 21b (i.e., compounds of Formula 21 wherein Rb is H or a suitable protecting group and X is a halogen such as Cl, Br or I) by transition metal-catalyzed O-arylation. This reaction can be catalyzed by a copper salt such as copper(I)
iodide and a ligand such as 1,10-phenanthroline, or by a palladium salt or complex such as palladium(II) acetate or tris(dibenzylideneacetone)dipalladium(0) and a phosphine ligand such as 2-di-tert-butylphosphino-2′-(N,N-dimethylamino)biphenyl (t-Bu-DavePhos) or rac-2-(Di- tert-butylphosphino)-1,1′-binaphthyl (TrixiePhos). These reactions are typically run in a solvent such as dioxane or toluene, in the presence of a base such as sodium tert-butoxide, cesium carbonate or potassium phosphate, at temperatures generally ranging from ambient temperature to the reflux temperature of the solvent. For examples of this reaction in the literature, see: J. Am. Chem. Soc. 2000, 122, 12907-12908; J. Am. Chem. Soc. 2001, 123, 12202-12206; J. Mol. Catal. A Chem.2008, 288, 28-32 and Tetrahedron Lett.2009, 50, 3790- 3793. Where protecting groups are utilized, removal using standard procedures known to those skilled in the art provides amines of Formula 8b. Scheme 15
24 wherein R b is a protecting group In some instances, amines of Formula 8b may be more readily accessed according to the sequence shown in Scheme 16. Compounds of Formula 25 can be prepared by ring opening of epoxides of Formula 20 and subsequent nucleophilic attack by phenols of Formula 11a (i.e., compounds of Formula 11 wherein Z is O). This reaction can generally be achieved in the presence of a base (e.g., monosodium phosphate, sodium phosphate, potassium carbonate, sodium hydride, sodium hydroxide, cesium fluoride or 1,8-diazabicyclo[5.4.0]undec-7-ene) in an appropriate solvent (e.g., acetonitrile, N,N-dimethylformamide, dimethyl sulfoxide, dichloromethane, toluene, methanol, isopropanol and/or water), at temperatures generally ranging from ambient temperature to 180 °C. Lewis acids (e.g., zinc chloride or boron trifluoride diethyl etherate) may also be used to facilitate the reaction. One skilled in the art will recognize that the epoxide substituents and reaction conditions impact the regioselectivity of the reaction, for example acidic conditions may reverse the regioselectivity of epoxide ring- opening. For relevant examples of this reaction in the literature, see: WO 2000/010994; US 2007/0185097 Al; WO 2009/009501; J. Med. Chem. 2023, 66, 1583-1600 and Angew. Chem. Int. Ed.2023, 62, e202217064. In a subsequent step, compounds of Formula 26 can be prepared by nitro reduction of the compounds of Formula 25, which can be achieved using standard conditions as described previously in Scheme 9, such as for example palladium on
carbon under an atmosphere of hydrogen in an appropriate solvent like methanol. For additional relevant examples of this reaction in the literature, see: WO 2009/009501, WO 2010/047956 and WO 2019/162323. Amines of Formula 8b can be prepared from compounds of Formula 26 by treatment with an acid (e.g., phosphoric acid or p-toluenesulfonic acid) in an appropriate solvent (e.g., xylene or toluene) at temperatures generally ranging from ambient temperature to the reflux temperature of the solvent. For relevant examples of this reaction in the literature, see: WO 2017/108723 and Synth. Commun. 1998, 28, 4105-4121. In some instances, it may be beneficial to convert the alcohol to a leaving group (e.g., Cl or mesylate (OMs)) to facilitate the reaction, which may then proceed in the presence of a base (e.g., potassium carbonate) in an appropriate solvent (e.g., N,N-dimethylformamide) at temperatures ranging from ambient temperature to the reflux temperature of the solvent. For a relevant example of this reaction in the literature, see: WO 2002/070726. Alternatively, in some instances, use of an appropriate aniline protecting group (e.g., tosyl) enables ring closure using Mitsunobu conditions as described previously in Scheme 10 (e.g., triphenyl phosphine and diethyl azodicarboxylate in an appropriate solvent like tetrahydrofuran). For relevant examples of this reaction in the literature, see: Org. Biomol. Chem.2010, 8, 2823-2828 and J. Org. Chem.2015, 80, 3815-3824.
As shown in Scheme 17, amines of Formula 8c (i.e., compounds of Formula 8 wherein Z is O and one α-amino substituent R3b is H; the remaining α-amino substituent R3b is Rc; and Rc is H or C1-C3 alkyl, or Rc can be taken together with R3a or the α-oxo substituent R3b to
form a ring) can be prepared from compounds of Formula 27 (wherein Rc is H or C1-C3 alkyl; or Rc can be taken together with R3a or R3b to form a ring) by nitro reduction followed by reductive amination. This reaction can be achieved using a transition metal catalyst, such as but not limited to palladium on carbon, platinum on carbon or Raney nickel under an atmosphere of hydrogen, in an appropriate solvent (e.g., methanol, ethanol, isopropanol, ethyl acetate, toluene or tetrahydrofuran). Temperatures typically range from ambient temperature to 80 °C. This reaction can generally be done using a Parr hydrogenator, optionally with reaction pressures above atmospheric pressure. Alternatively, the reaction can be achieved in a stepwise manner. Nitro reduction can be achieved under a range of conditions, such as iron metal in the presence of an acid such as acetic acid, hydrochloric acid or aqueous ammonium chloride, optionally with a solvent such as methanol, ethanol, ethyl acetate, tetrahydrofuran or N,N-dimethylformamide at temperatures ranging from ambient temperature to the reflux temperature of the solvent. Other suitable conditions include zinc metal with acetic acid or aqueous ammonium chloride, and stannous chloride in aqueous hydrochloric acid or ethanol. The second step involves imine reduction which can be achieved using a reducing agent like sodium borohydride, sodium cyanoborohydride or lithium aluminum hydride in an appropriate solvent (e.g., ethanol, methanol, tetrahydrofuran or dichloromethane, optionally with a co-solvent or additive like water or acetic acid) at temperatures generally between -78 °C and the reflux temperature of the solvent. Other suitable reducing conditions for this step include but are not limited to hydrogen and palladium on carbon in a solvent such as methanol, or triethylsilane and trifluoroacetic acid in a solvent such as dichloromethane. For relevant examples of this reaction in the literature, see: J. Org. Chem.2002, 67, 6097-6103; WO2014/171527; J. Org. Chem.2015, 80, 3815-3824; WO2015/124868; WO 2017/172505 and WO 2022/035799. Scheme 17
As shown in Scheme 18, compounds of Formula 27 can be prepared by reaction of nitrophenols of Formula 11a with compounds of Formula 30 (wherein LG is a leaving group such as Cl or Br and Rc is H or C1-C3 alkyl; or Rc can be taken together with R3a or R3b to form a ring) in the presence of a base, such as potassium carbonate, cesium carbonate, sodium bicarbonate or sodium hydride, optionally with additives such as sodium iodide, in a solvent
like N,N-dimethylformamide, acetone or 1,4-dioxane, at temperatures typically ranging from 0 °C to the reflux temperature of the solvent. Alternatively, Mitsunobu reaction conditions may be appropriate in some cases, for the reaction of nitrophenols of Formula 11a with compounds of Formula 29 (wherein Rc is H or C1-C3 alkyl; or Rc can be taken together with R3a or R3b to form a ring). Typical reaction conditions include triphenylphosphine and an azodicarboxylate such as diethyl azodicarboxylate or diisopropyl azodicarboxylate, in a solvent like tetrahydrofuran or dichloromethane at temperatures ranging from about –10 °C to the reflux temperature of the solvent. For relevant examples of these reactions in the literature, see: J. Med. Chem.1988, 31, 1548-1558; WO 2001/090088; WO 2004/080973 and WO 2018/013770. Nitrophenols, α-hydroxy carbonyls and α-halo carbonyls of Formulae 11a, 29 and 30 are generally commercially available or known in the literature. In some instances, compounds of Formula 27 may be more readily accessed using protecting groups to mask the carbonyl group, such as an acetal group to mask an aldehyde, or via standard functional group interconversions of one carbonyl functional group to another. Scheme 18
An alternative synthesis of amines of Formula 8d (i.e., compounds of Formula 8 wherein Z is CR3c or NH) involves reduction of azaarenes of Formula 31, as shown in Scheme 19. This reaction can be achieved using a transition metal catalyst, such as palladium, palladium hydroxide, platinum, platinum dioxide or Raney nickel under an atmosphere of hydrogen, in an appropriate solvent (e.g., methanol, ethanol, ethyl acetate, tetrahydrofuran and/or acetic acid). Temperatures typically range from ambient temperature to 80 °C. This reaction can generally be done in a Parr hydrogenator. Other suitable conditions include transfer hydrogenation using a Hantzsch dihydropyridine (e.g., Hantzsch ester) as the hydrogen source, in the presence of a catalyst (e.g., diphenyl phosphate, trifluoroacetic acid, boric acid, iron(II)
triflate or silver(I) triflate) in an appropriate solvent (e.g., toluene, benzene, dichloromethane, 1,2-dichloroethane or chloroform) at temperatures generally ranging from 0 °C to the reflux temperature of the solvent. Sodium cyanoborohydride is also an appropriate reducing agent, used in the presence of an acid (e.g., hydrochloric acid or acetic acid) in a solvent (e.g., ethanol, water, methanol, tetrahydrofuran, or combinations thereof) at temperatures generally ranging from 0 °C to the reflux temperature of the solvent. Borane (e.g., borane tetrahydrofuran complex) may also be used for this reaction. For examples of related reactions in the literature, see: WO 2016/086200; Bioorg. Med. Chem. Lett.2001, 11, 1989-1992 and Org. Lett.2020, 22, 4038-4042. Many azaarenes of Formula 31 are commercially available or known in the literature. Scheme 19
As shown in Scheme 20, quinolines of Formula 31a (i.e., compounds of Formula 31 wherein R3b is other than halogen; Z is CR3c; and R3c is other than a leaving group, such as halogen, cyano, nitro or alkoxy) can be prepared by reaction of 2-aminobenzaldehydes or ketones of Formula 32a with aldehydes or ketones of Formula 33 (wherein R3b is not halogen) in the presence of an acid (e.g., p-toluenesulfonic acid, trifluoroacetic acid, acetic acid or hydrochloric acid) or a base (e.g., sodium hydroxide, potassium hydroxide or sodium methoxide). A solvent is typically used (e.g., ethanol, methanol, butan-2-one or toluene, optionally with water as a co-solvent). Temperatures generally range from ambient temperature to 200 °C. Alternatively, the reaction may be promoted by organosilanes (e.g., trimethylsilyl chloride) or catalyzed by Lewis acids (e.g., ytterbium triflate). For relevant examples of these reactions in the literature, see: WO 2021/092525; WO 2022/053422 and Tetrahedron Lett. 2011, 52, 3474-3477. Similarly, quinolines of Formula 31b (i.e., compounds of Formula 31 wherein R3b is not halogen; Z is CR3c; and R3c is hydroxy) can be prepared by reaction of 2-aminobenzoic acids, esters or acyl chlorides of Formula 32b with aldehydes or ketones of Formula 33 (where R3b is not halogen). The reaction is often conducted in a solvent (e.g., diphenyl ether, xylene or toluene) at temperatures typically ranging from 80 °C to 230 °C. The reaction may be promoted by Brønsted acids (e.g., methanesulfonic acid and/or polyphosphoric acid), where the acid may also be used as the solvent, or Lewis acids (e.g., zinc chloride). For examples of these reactions in the literature,
see: WO 2015/057205; WO 2019/210837 and J. Med. Chem. 2003, 46, 4895-4903. The hydroxy group (R3c) in quinolines of Formula 31b can be converted to other functional groups using methods known to those skilled in the art, for example treatment with phosphorus oxychloride may provide the corresponding chloroquinoline (i.e., R3c is Cl). Compounds of Formulae 32 and 33 are generally commercially available or known in the literature. Scheme 20
other than halogen
32a is other than wherein R 3c 31a is oth wherein R 3c er than or alkoxy. or alkoxy. halogen, CN, NO 2 halogen, CN, NO 2 32b wherein R 3c is OH. 31b is OH. wherein R 3c Dihydroquinoxalinones of Formula 8e (i.e., compounds of Formula 8 wherein Z is NH and the adjacent R3a and R3b are taken together with the carbon atom to which they are attached to form a carbonyl group) can be prepared by the reaction of 2-bromoanilines of Formula 34 with amino acids of Formula 35 as depicted in Scheme 21, using conditions as described previously in Scheme 12. For a relevant example of this reaction in the literature, see: ACS Omega 2017, 2, 1875. 2-Bromoanilines and amino acids of Formulae 34 and 35 are commercially available or known in the literature. Scheme 21
34 8e Alternatively, amines of Formula 8f (i.e., compounds of Formula 8 wherein Z is CR3c) may be prepared according to the sequence shown in Scheme 22. In the first step, compounds of Formula 36 can be prepared by reaction of 2-aminobenzaldehydes or ketones of Formula 32a with carboxylic acids or acid chlorides of Formula 38 (where Rc is OH or Cl) using amidation conditions previously described in Scheme 7. For relevant examples of this reaction in the literature, see: RSC Adv. 2017, 7, 28298; J. Nat. Prod. 2020, 83, 3181 and
WO 2011/025690. In the second step, compounds of Formula 37 can be prepared by a cyclization of compounds of Formula 36 in the presence of a base (e.g., potassium carbonate, sodium hydroxide, potassium tert-butoxide) in a solvent (e.g., N,N-dimethylformamide, ethanol, dimethyl sulfoxide, 1,4-dioxane) at temperatures generally ranging from ambient temperature to 120 °C. For a relevant example of this reaction in the literature, see: WO 2019/191667. In the third step, amines of Formula 8f can be prepared from compounds of Formula 37 by treatment with a reducing agent like borane or lithium borohydride in a solvent like tetrahydrofuran at temperatures ranging from 0 °C to the reflux temperature of the solvent. For a relevant example of this reaction in the literature, see: WO 2015/095795. The same reduction can also be achieved using multi-step approaches, such as conversion of compounds of Formula 37 to 2-chloroquinolines by treatment with phosphorus oxychloride, followed by dechlorination (e.g., by hydrogenation in the presence of catalytic palladium) followed by the reduction of the resultant quinoline using the method previously described in Scheme 19. For an example of this reaction in the literature, see: J. Med. Chem. 1989, 32, 1927. Compounds of Formulae 32a and 38 are generally commercially available or known in the literature. Scheme 22
It is recognized by one skilled in the art that various functional groups can be converted into others to provide different compounds of Formula 1. For a valuable resource that illustrates the interconversion of functional groups in a simple and straightforward fashion, see Larock, R. C., Comprehensive Organic Transformations: A Guide to Functional Group
Preparations, 2nd Ed., Wiley-VCH, New York, 1999. For example, intermediates for the preparation of compounds of Formula 1 may contain aromatic nitro groups, which can be reduced to amino groups, and then converted via reactions well known in the art such as the Sandmeyer reaction, to various halides, providing compounds of Formula 1. The above reactions can also in many cases be performed in alternate order. It is recognized that some reagents and reaction conditions described above for preparing compounds of Formula 1 may not be compatible with certain functionalities present in the intermediates. In these instances, the incorporation of protection/deprotection sequences or functional group interconversions into the synthesis will aid in obtaining the desired products. The use and choice of the protecting groups will be apparent to one skilled in chemical synthesis (see, for example, Greene, T. W.; Wuts, P. G. M. Protective Groups in Organic Synthesis, 2nd ed.; Wiley: New York, 1991). One skilled in the art will recognize that, in some cases, after the introduction of a given reagent as depicted in any individual scheme, it may be necessary to perform additional routine synthetic steps not described in detail to complete the synthesis of compounds of Formula 1. One skilled in the art will also recognize that it may be necessary to perform a combination of the steps illustrated in the above schemes in an order other than that implied by the particular presented to prepare the compounds of Formula 1. One skilled in the art will also recognize that compounds of Formula 1 and the intermediates described herein can be subjected to various electrophilic, nucleophilic, radical, organometallic, oxidation, and reduction reactions to add substituents or modify existing substituents. Without further elaboration, it is believed that one skilled in the art using the preceding description can utilize the present invention to its fullest extent. The following non-limiting Examples are illustrative of the invention. Steps in the following Examples illustrate a procedure for each step in an overall synthetic transformation, and the starting material for each step may not have necessarily been prepared by a particular preparative run whose procedure is described in other Examples or Steps. Percentages are by weight except for chromatographic solvent mixtures or where otherwise indicated. Parts and percentages for chromatographic solvent mixtures are by volume unless otherwise indicated. 1H NMR spectra are reported in ppm downfield from tetramethylsilane; “s” means singlet, “d” means doublet, “t” means triplet, “q” means quarte, “p” means pentet, “m” means multiplet, “br s” means broad singlet, “br d” means broad doublet and “dd” means doublet of doublets. The term HPLC means high performance liquid chromatography. The term MPLC means medium pressure liquid chromatography. The term “LCMS” means liquid chromatography coupled with mass spectrometry.
SYNTHESIS EXAMPLE 1 Preparation of (3,4-dihydro-3,5-dimethyl-1(2H)-quinolinyl)[2-methoxy-5-[3-(1-methyl- ethyl)-1H-1,2,4-triazol-1-yl]phenyl]methanone (alternatively named (3,5- dimethyl-3,4-dihydro-2H-quinolin-1-yl)-[5-(3-isopropyl-1,2,4-triazol-1-yl)-2- methoxy-phenyl]methanone) (Compound 3). Step A: Preparation of 5-bromo-3-methyl-quinoline. To a mixture of 2-amino-6-bromo-benzaldehyde (200 mg, 1 mmol), propionaldehyde (0.1 mL, 1.4 mmol) and ethanol (2 mL) in a microwave vial was added 1 N aqueous sodium hydroxide solution (2 mL). The mixture was stirred for 30 minutes at 110 °C in a microwave, then was cooled to room temperature and extracted with dichloromethane (3x). The combined organic extracts were washed with brine (1x), dried over anhydrous sodium sulfate and concentrated under reduced pressure. The resultant crude material was purified by column chromatography on silica gel (eluting with ethyl acetate in hexanes) to afford the title compound (80 mg). 1H NMR (CDCl3) δ 8.78 (m, 1H), 8.30 (m, 1H), 8.06-8.04 (m, 1H), 7.81-7.79 (m, 1H), 7.52- 7.48 (m, 1H), 2.58 (s, 3H). Step B: Preparation of 3,5-dimethylquinoline. A sealed tube was charged with 5-bromo-3-methyl-quinoline (i.e., the product of Step A) (200 mg, 0.9 mmol), trimethylboroxine (148 mg, 1.2 mmol), potassium carbonate (310 mg, 2.2 mmol) and 1,4-dioxane (9 mL). The mixture was sparged with argon for 5- 10 min. Tetrakis(triphenylphosphine)palladium(0) (520 mg, 0.45 mmol) was added to the mixture. The tube was sealed with a Teflon cap and stirred at 110 °C for 16 h. The mixture was cooled to room temperature, filtered through a pad of Celite® with ethyl acetate. The filtrate was concentrated under reduced pressure and the resultant crude material was purified by silica gel column chromatography (eluting with a solution of ethyl acetate in hexanes) to afford the title compound (140 mg). 1H NMR (CDCl3) δ 8.76 (m, 1H), 8.08 (m, 1H), 7.93-7.91 (m, 1H), 7.54-7.50 (m, 1H), 7.34- 7.32 (m, 1H), 2.66 (s, 3H), 2.54 (s, 3H). Step C: Preparation of 3,5-dimethyl-1,2,3,4-tetrahydroquinoline. To a stirred solution of 3,5-dimethylquinoline (i.e., the product of Step B) (60 mg, 0.38 mmol) in a mixture of ethanol and water (3:1, 4 mL) was added ammonium formate (598 mg, 9.5 mmol). The mixture was sparged with argon for 5 min. Palladium on carbon (10% Pd, 50% wet, 45 mg) was added and the mixture was stirred at 75 °C for 16 h. The mixture was cooled to room temperature, filtered through a pad of Celite® with ethanol. The filtrate was concentrated under reduced pressure and the resultant crude material was purified
by silica gel column chromatography (elutng with a solution of ethyl acetate in hexanes) to afford the title compound (25 mg). MS m/z Found: (M+H)+, C11H15N, 162.1, requires 162.1. Step D: Preparation of methyl 5-(3-isopropyl-1,2,4-triazol-1-yl)-2-methoxy-benzoate. To a solution of methyl 5-iodo-2-methoxy-benzoate (200 mg, 0.68 mmol) in toluene (2 mL) in a microwave vial was added 3-isopropyl-1H-1,2,4-triazole (456 mg) and potassium carbonate (208 mg, 1.5 mmol). The mixture was sparged with argon. Copper(I) iodide (6.5 mg, 0.034 mmol) and trans-N,N′-dimethylcyclohexane-1,2-diamine (19.5 mg, 0.14 mmol) were added and the mixture was stirred for 2 h at 140 °C in a microwave. The mixture was cooled to room temperature, filtered through a pad of Celite® with ethyl acetate. The filtrate was washed with water (1x), brine (1x), dried over anhydrous sodium sulfate and concentrated under reduced pressure. The resultant crude material was purified by silica gel column chromatography (eluting with a solution of ethyl acetate in hexanes) to afford the title compound (40 mg). 1H NMR (DMSO-d6) δ 9.09 (s, 1H), 8.05-8.04 (m, 1H), 8.00-7.96 (m, 1H), 7.33-7.31 (m, 1H), 3.88 (s, 3H), 3.83 (s, 3H), 3.08-3.01 (m, 1H), 1.29 (d, 6H). Step E: Preparation of (3,4-dihydro-3,5-dimethyl-1(2H)-quinolinyl)[2-methoxy-5-[3- (1-methylethyl)-1H-1,2,4-triazol-1-yl]phenyl]methanone (alternatively named (3,5-dimethyl-3,4-dihydro-2H-quinolin-1-yl)-[5-(3-isopropyl-1,2,4-triazol-1- yl)-2-methoxy-phenyl]methanone). A sealed flask was charged with methyl 5-(3-isopropyl-1,2,4-triazol-1-yl)-2-methoxy- benzoate (i.e., the product of Step D) (100 mg, 0.36 mmol), 3,5-dimethyl-1,2,3,4- tetrahydroquinoline (i.e., the product of Step C) (88 mg, 0.55 mmol) and toluene (3 mL) then trimethylaluminum (2M solution in toluene, 0.27 mL, 0.54 mmol) and triethylamine (0.1 mL, 0.72 mmol) were added. The mixture was stirred at 80 °C for 5 h, cooled to room temperature and a saturated aqueous sodium bicarbonate solution was added. The layers were separated, the aqueous phase was extracted with ethyl acetate (3x). The combined organic extracts were dried over anhydrous sodium sulfate and concentrated under reduced pressure. The crude material was purified by column chromatography on silica gel (eluting with a solution of ethyl acetate in hexanes) to afford the title compound (40 mg), a compound of the present invention. MS m/z: (M+H)+, C24H28N4O2, 405.2.
SYNTHESIS EXAMPLE 2 Preparation of [(2S)-2,3-dihydro-2,8-dimethyl-4H-1,4-benzoxazin-4-yl][6-[3-(1- methylethyl)-1H-1,2,4-triazol-1-yl]-4-pyrimidinyl]methanone (alternatively named [(2S)-2,8-dimethyl-2,3-dihydro-1,4-benzoxazin-4-yl]-[6-(3-isopropyl- 1,2,4-triazol-1-yl)pyrimidin-4-yl]methanone) (Compound 2). Step A: Preparation of methyl (2S)-2-(2-methyl-6-nitro-phenoxy)propanoate. To a stirred mixture of 2-methyl-6-nitro-phenol (10 g, 65 mmol) and triphenyl- phosphine (22 g, 85 mmol) in anhydrous tetrahydrofuran (100 mL) was added methyl (R)- lactate (9.4 mL, 98 mmol). The mixture was cooled to 0 °C then a solution of diisopropyl azodicarboxylate (17 mL, 85 mmol) in anhydrous tetrahydrofuran (20 mL) was added slowly. The reaction mixture was stirred at room temperature overnight and then concentrated under reduced pressure. A mixture of hexanes and diethyl ether (1:1, 350 mL) was added to the mixture and stirred for 1 h, resulting in formation of a precipitate. The mixture was filtered through a pad of Celite® with a solution of hexanes in diethyl ether (1:1, 150 mL). The resultant filtrate was concentrated under reduced pressure. The crude material was purified by column chromatography on silica gel (eluting with a gradient of 0-30% ethyl acetate in hexanes) to afford the title compound as a yellow oil (14.5 g). 1H NMR (CDCl3) δ 7.65-7.63 (m, 1H), 7.41-7.39 (m, 1H), 7.12-7.08 (m, 1H), 4.56 (q, 1H), 3.71 (s, 3H), 2.38 (s, 3H), 1.60 (d, 3H). Step B: Preparation of (2S)-2,8-dimethyl-4H-1,4-benzoxazin-3-one. To a stirred solution of methyl (2S)-2-(2-methyl-6-nitro-phenoxy)propanoate (i.e., the product of Step A) (13.9 g, 58.2 mmol) in ethanol (210 mL) at 50 °C was added a solution of ammonium chloride (6.2 g, 116 mmol) in water (23 mL). Iron powder (9.7 g, 175 mmol) was then added portionwise over 15 minutes as the reaction mixture was heated from 50 °C to 70 °C. After stirring at 70 °C for 23 h, the mixture was cooled to room temperature and filtered through a pad of Celite®. The filtrate was concentrated under reduced pressure followed by the addition of ethyl acetate and water. The layers were separated and the aqueous phase was extracted with ethyl acetate (1x). The combined organic extracts were washed with saturated aqueous ammonium chloride solution (1x), dried over anhydrous magnesium sulfate, filtered through a pad of Celite® and concentrated under reduced pressure to afford the title compound as a white solid (9.2 g), which was used in the next step without further purification. 1H NMR (CDCl3) δ 7.80 (br s, 1H), 6.87-6.83 (m, 2H), 6.64-6.60 (m, 1H), 4.66 (q, 1H), 2.25 (s, 3H), 1.58 (d, 3H). Step C: Preparation of (2S)-2,8-dimethyl-3,4-dihydro-2H-1,4-benzoxazine. To a stirred solution of (2S)-2,8-dimethyl-4H-1,4-benzoxazin-3-one (i.e., the product of Step B) (10.3 g, 58 mmol) in anhydrous tetrahydrofuran (100 mL) at 0 °C was added borane
tetrahydrofuran complex (1M in tetrahydrofuran, 116 mL, 116 mmol) over 30 minutes. The reaction mixture was stirred at room temperature overnight, cooled to 0 °C and methanol (70 mL) was slowly added. After stirring at room temperature for 1 h, the mixture was concentrated under reduced pressure and partitioned between ethyl acetate and water. The layers were separated and the organic phase was washed with water (1x), brine (1x), dried over anhydrous magnesium sulfate, filtered and concentrated under reduced pressure to afford the title compound as a light brown oil (9.4 g), which was used in the next step without further purification. 1H NMR (CDCl3) δ 6.68-6.65 (m, 1H), 6.57-6.55 (m, 1H), 6.48-6.46 (m, 1H), 4.28-4.22 (m, 1H), 3.68 (br s, 1H), 3.34 (dd, 1H), 3.10 (dd, 1H), 2.20 (s, 3H), 1.40 (d, 3H). Step D: Preparation of 6-(3-isopropyl-1,2,4-triazol-1-yl)pyrimidine-4-carboxylic acid To a stirred solution of methyl 6-chloropyrimidine-4-carboxylate (250 mg, 1.45 mmol) in N,N-dimethylformamide (10 mL) was added 3-isopropyl-1H-1,2,4-triazole (161 mg, 1.45 mmol) and potassium carbonate (401 mg, 2.90 mmol). The mixture was stirred at 90 °C for 12 h. Ice-water was added to the mixture. The mixture was then extracted with ethyl acetate. The combined organic extracts were washed with brine and concentrated under reduced pressure to give the title compound (220 mg), which was used without further purification. 1H NMR (DMSO-d6) δ 9.47 (s, 1H), 9.28 (s, 1H), 8.21 (s, 1H), 3.13-3.09 (m, 1H), 1.33 (d,
234.2. Step E: Preparation of [(2S)-2,3-dihydro-2,8-dimethyl-4H-1,4-benzoxazin-4-yl][6-[3- (1-methylethyl)-1H-1,2,4-triazol-1-yl]-4-pyrimidinyl]methanone (alternatively named [(2S)-2,8-dimethyl-2,3-dihydro-1,4-benzoxazin-4-yl]-[6- (3-isopropyl-1,2,4-triazol-1-yl)pyrimidin-4-yl]methanone). To a stirred solution of 6-(3-isopropyl-1,2,4-triazol-1-yl)pyrimidine-4-carboxylic acid (i.e., the product of Step D) (220 mg, 0.94 mmol) in 1,2-dichloroethane (10 mL) was added (2S)-2,8-dimethyl-3,4-dihydro-2H-1,4-benzoxazine (i.e., the product of Step C) (154 mg, 0.94 mmol), propylphosphonic anhydride (50 wt.% in ethyl acetate, 0.56 mL) and triethylamine (0.46 mL, 3.3 mmol). The mixture was stirred at 60 °C for 3 h. Ice-water was then added, the mixture was extracted with ethyl acetate, the combined organic extracts were washed with brine and concentrated under reduced pressure. The crude material was purified by preparative HPLC to afford the title compound, a compound of the present invention, as a yellow solid (78 mg). 1H NMR (DMSO-d6, 100 °C) δ 9.36 (m, 1H), 9.13 (m, 1H), 8.04 (m, 1H), 7.36-7.31 (m, 1H), 6.96-6.94 (m, 1H), 6.70-6.67 (m, 1H), 4.52 (m, 1H), 4.09-4.05 (m, 1H), 3.53-3.48 (m, 1H), 3.16-3.09 (m, 1H), 2.18 (m, 3H), 1.36-1.34 (m, 9H).
MS m/z: (M+H)+, C20H22N6O2, 379.35. SYNTHESIS EXAMPLE 3 Preparation of [(2S)-2,3-dihydro-2,8-dimethyl-4H-1,4-benzoxazin-4-yl][2-[3-(1- methylethyl)-1H-1,2,4-triazol-1-yl]-4-pyrimidinyl]methanone (alternatively named [(2S)-2,8-dimethyl-2,3-dihydro-1,4-benzoxazin-4-yl]-[2-(3-isopropyl- 1,2,4-triazol-1-yl)pyrimidin-4-yl]methanone) (Compound 1). Step A: Preparation of 2-(3-isopropyl-1,2,4-triazol-1-yl)pyrimidine-4-carboxylic acid To a stirred solution of methyl 2-chloropyrimidine-4-carboxylate (250 mg, 1.45 mmol) in N,N-dimethylformamide (10 mL) was added 3-isopropyl-1H-1,2,4-triazole (161 mg, 1.45 mmol) and potassium carbonate (401 mg, 2.90 mmol). The mixture was stirred at 90 °C for 15 h. Ice-water was added to the mixture, and the mixture was extracted with ethyl acetate. The combined organic extracts were washed with brine and concentrated under reduced pressure to give the title compound (250 mg), which was used in the next step without further purification. 1H), 9.14 (d, 1H), 8.00 (d, 1H), 3.15-3.07 (m, 1H), 1.32 (d,
234.2. Step B: Preparation of [(2S)-2,3-dihydro-2,8-dimethyl-4H-1,4-benzoxazin-4-yl][2-[3- (1-methylethyl)-1H-1,2,4-triazol-1-yl]-4-pyrimidinyl]methanone (alternatively named [(2S)-2,8-dimethyl-2,3-dihydro-1,4-benzoxazin-4-yl]-[2- (3-isopropyl-1,2,4-triazol-1-yl)pyrimidin-4-yl]methanone). To a stirred solution of 2-(3-isopropyl-1,2,4-triazol-1-yl)pyrimidine-4-carboxylic acid (i.e., the product of Step A) (150 mg, 0.64 mmol) in 1,2-dichloroethane (10 mL) was added (2S)-2,8-dimethyl-3,4-dihydro-2H-1,4-benzoxazine (i.e., the product of Step C in Synthesis Example 2) (105 mg, 0.64 mmol), propylphosphonic anhydride (50 wt.% in ethyl acetate, 0.38 mL) and triethylamine (0.36 mL, 2.6 mmol). The mixture was stirred at 60 °C for 6 h. Ice-water was added to the mixture, the mixture was extracted with ethyl acetate. The combined organic extracts were washed with brine and concentrated under reduced pressure. The resultant crude material was purified by preparative HPLC to afford the title compound, a compound of the present invention, as a yellow solid (75 mg). 1H NMR (DMSO-d6, 100 °C) δ 9.10-9.04 (m, 2H), 7.73-7.72 (m, 1H), 7.44 (m, 1H), 6.98- 6.96 (m, 1H), 6.72-6.70 (m, 1H), 4.54 (m, 1H), 4.13-4.10 (m, 1H), 3.55-3.50 (m, 1H), 3.14- 3.07 (m, 1H), 2.19 (m, 3H), 1.36-1.33 (m, 9H). MS m/z: (M+H)+, C20H22N6O2, 379.33.
SYNTHESIS EXAMPLE 4 Preparation of [(2S)-2,3-dihydro-2,8-dimethyl-4H-1,4-benzoxazin-4-yl][6-[3-(1- methylethyl)-1H-1,2,4-triazol-1-yl]-2-pyrazinyl]methanone (alternatively named [(2S)-2,8-dimethyl-2,3-dihydro-1,4-benzoxazin-4-yl]-[6-(3-isopropyl- 1,2,4-triazol-1-yl)pyrazin-2-yl]methanone) (Compound 4). Step A: Preparation of 6-(3-isopropyl-1,2,4-triazol-1-yl)pyrazine-2-carboxylic acid To a stirred solution of methyl 6-chloropyrazine-2-carboxylate (250 mg, 1.45 mmol) in N,N-dimethylformamide (10 mL) was added 3-isopropyl-1H-1,2,4-triazole (161 mg, 1.45 mmol) and potassium carbonate (401 mg, 2.90 mmol). The mixture was stirred at 90 °C for 15 h. Ice-water was added to the mixture and the mixture was extracted with ethyl acetate. The combined organic extracts were washed with brine and concentrated under reduced pressure to give the title compound (300 mg), which was used without further purification. MS m/z: (M+H)+, C10H11N5O2, 234.3. Step B: Preparation of [(2S)-2,3-dihydro-2,8-dimethyl-4H-1,4-benzoxazin-4-yl][6-[3- (1-methylethyl)-1H-1,2,4-triazol-1-yl]-2-pyrazinyl]methanone (alternatively named [(2S)-2,8-dimethyl-2,3-dihydro-1,4-benzoxazin-4-yl]-[6-(3-isopropyl- 1,2,4-triazol-1-yl)pyrazin-2-yl]methanone). To a stirred solution of 6-(3-isopropyl-1,2,4-triazol-1-yl)pyrazine-2-carboxylic acid (i.e., the product of Step A) (250 mg, 1.07 mmol) in 1,2-dichloroethane (20 mL) was added (2S)-2,8-dimethyl-3,4-dihydro-2H-1,4-benzoxazine (i.e., the product of Step C in Synthesis Example 2) (175 mg, 1.07 mmol), propylphosphonic anhydride (50 wt.% in ethyl acetate, 0.64 mL) and triethylamine (0.45 mL, 3.2 mmol). The mixture was stirred at 60 °C for 6 h. Ice- water was added, the mixture was extracted with ethyl acetate; the combined organic extracts were washed with brine and concentrated under reduced pressure. The crude material was purified by preparative HPLC to afford the title compound, a compound of the present invention, as a sticky yellow solid (72 mg). 1H NMR (DMSO-d6, 100 °C) δ 9.17 (m, 1H), 8.96-8.92 (m, 2H), 7.30 (m, 1H), 6.97-6.95 (m, 1H), 6.70-6.66 (m, 1H), 4.55-4.54 (m, 1H), 4.18-4.14 (m, 1H), 3.60-3.55 (m, 1H), 3.16-3.10 (m, 1H), 2.20 (m, 3H), 1.36 – 1.34 (m, 9H). MS m/z: (M+H)+, C20H22N6O2, 379.1.
SYNTHESIS EXAMPLE 5 Preparation of [(2S)-2,3-dihydro-2,8-dimethyl-4H-1,4-benzoxazin-4-yl][4-[3-(1- methylethyl)-1H-1,2,4-triazol-1-yl]-2-pyrimidinyl]methanone (alternatively named [(2S)-2,8-dimethyl-2,3-dihydro-1,4-benzoxazin-4-yl]-[4-(3-isopropyl- 1,2,4-triazol-1-yl)pyrimidin-2-yl]methanone) (Compound 21). Step A: Preparation of 4-(3-isopropyl-1,2,4-triazol-1-yl)-2-methylsulfanyl- pyrimidine. To a stirred solution of 3-isopropyl-1H-1,2,4-triazole (332 mg, 2.99 mmol) in N,N-dimethylformamide (3 mL) was added sodium hydride (60% dispersion in mineral oil, 2.7 mmol) portionwise at 0 °C. After 10 minutes, a solution of 4-chloro-2-methylsulfanyl- pyrimidine (400 mg, 2.49 mmol) in N,N-dimethylformamide (2 mL) was added dropwise to the mixture. The mixture was allowed to warm to room temperature, stirred for 4 h, quenched with cold water and extracted with dichloromethane. The combined organic extracts were washed with water, brine, dried over anhydrous sodium sulfate and concentrated under reduced pressure. The crude material was purified by column chromatography on silica gel to afford the title compound (300 mg). 1H NMR
δ 9.41 (s, 1H), 8.76 (d, 1H), 7.49 (d, 1H), 3.12-3.05 (m, 1H), 2.60 (s, 3H), 1.30 (d, 6H). Step B: Preparation of 4-(3-isopropyl-1,2,4-triazol-1-yl)-2-methylsulfonyl-pyrimidine To a stirred solution of 4-(3-isopropyl-1,2,4-triazol-1-yl)-2-methylsulfanyl-pyrimidine (i.e., the product of Step A) (2.3 g, 9.8 mmol) in chloroform (20 mL) was added meta- chloroperbenzoic acid (4.2 g, 24.3 mmol) at 0 °C. The mixture was stirred at room temperature for 16 h and then quenched with saturated aqueous sodium thiosulfate solution at 0 °C. After stirring at this temperature for 30 minutes, the mixture was filtered through a pad of Celite® with dichloromethane. The filtrate was mostly concentrated under reduced pressure, and the layers were separated. The organic phase was washed with saturated aqueous sodium bicarbonate solution, brine, dried over anhydrous sodium sulfate and concentrated under reduced pressure. The crude material was purified by column chromatography on silica gel to afford the title compound as a white solid (1.7 g). 1H NMR (DMSO-d6) δ 9.58 (s, 1H), 9.17 (d, 1H), 8.06 (d, 1H), 3.53 (s, 3H), 3.15-3.08 (m, 1H), 1.32 (d, 6H). MS m/z: (M+H)+, C10H13N5O2S, 268.0. Step C: Preparation of 4-(3-isopropyl-1,2,4-triazol-1-yl)pyrimidin-2-ol. To a stirred solution of 4-(3-isopropyl-1,2,4-triazol-1-yl)-2-methylsulfonyl-pyrimidine (i.e., the product of Step B) (200 mg, 0.75 mmol) in 1,4-dioxane (15 mL) was added 1 N aqueous sodium hydroxide solution (1.3 mL) at room temperature. The mixture was stirred
for 16 h. Water was added. The mixture was acidified to pH~2 with 2 N aqueous hydrochloric acid solution and extracted with a 20% solution of methanol in dichloromethane. The combined organic extracts were washed with brine, dried over anhydrous sodium sulfate and concentrated under reduced pressure. The crude material was washed with pentane to afford the title compound as an off white solid (140 mg). 9.26 (s, 1H), 8.14 (d, 1H), 6.78 (d, 1H), 3.09-3.03 (m, 1H), 1.29 (d,
1N5O, 206.18. Step D: Preparation of 2-chloro-4-(3-isopropyl-1,2,4-triazol-1-yl)pyrimidine. A stirred suspension of 4-(3-isopropyl-1,2,4-triazol-1-yl)pyrimidin-2-ol (i.e., the product of Step C) (1.2 g, 5.8 mmol) in phosphorus oxychloride (8 mL) was heated at 90 °C for 2 h. The volatiles were evaporated. The mixture was quenched with crushed ice at 0 °C and extracted with ethyl acetate. The combined organic extracts were washed with brine, dried over anhydrous sodium sulfate, filtered and concentrated under reduced pressure to give the title compound (900 mg crude), which was used without further purification. MS m/z: (M+H)+, C9H10ClN5, 224.2. Step E: Preparation of ethyl 4-(3-isopropyl-1,2,4-triazol-1-yl)pyrimidine-2- carboxylate. To a suspension of 2-chloro-4-(3-isopropyl-1,2,4-triazol-1-yl)pyrimidine (i.e., the product of Step D) (900 mg crude) in ethanol (35 mL) was added sodium carbonate (427 mg, 4.0 mmol), 1,1′-ferrocenediyl-bis(diphenylphosphine) (223 mg, 0.40 mmol) and palladium(II) acetate (90 mg, 0.40 mmol). The mixture was sparged with argon for 10 minutes. The mixture was stirred under an atmosphere of carbon monoxide (50 psi) at 50 °C for 16 h in an autoclave. The mixture was cooled to room temperature and filtered through a pad of Celite® with dichloromethane. The filtrate was concentrated under reduced pressure and the resulting crude material was purified by column chromatography on silica gel to afford the title compound as a pale brown solid (350 mg). MSO-d6) δ 9.35 (s, 1H), 9.09 (d, 1H), 8.01 (d, 1H), 4.44-4.39 (m, 2H), 3.14-3.07 8-1.31 (m, 9H).
+H)+, C12H15N5O2, 262.3. Step F: Preparation of 4-(3-isopropyl-1,2,4-triazol-1-yl)pyrimidine-2-carboxylic acid To a stirred solution of ethyl 4-(3-isopropyl-1,2,4-triazol-1-yl)pyrimidine-2- carboxylate (i.e., the product of Step E) (350 mg, 1.3 mmol) in tetrahydrofuran (4.5 mL) and water (1.5 mL) was added lithium hydroxide monohydrate (115 mg, 2.7 mmol). The reaction mixture was stirred at room temperature for 3 h. The volatiles were evaporated then the mixture was diluted with water and washed with a 10% solution of ethyl acetate in hexanes.
The aqueous phase was acidified with 2 N aqueous hydrochloric acid solution. The resulting precipitate was collected by filtration and washed with pentane to afford the title compound as a white solid (180 mg). 1H), 9.08 (d, 1H), 7.97 (d, 1H), 3.16-3.07 (m, 1H), 1.32 (d,
234.1. Step G: Preparation of [(2S)-2,3-dihydro-2,8-dimethyl-4H-1,4-benzoxazin-4-yl][4-[3- (1-methylethyl)-1H-1,2,4-triazol-1-yl]-2-pyrimidinyl]methanone (alternatively named [(2S)-2,8-dimethyl-2,3-dihydro-1,4-benzoxazin-4-yl]-[4- (3-isopropyl-1,2,4-triazol-1-yl)pyrimidin-2-yl]methanone). To a stirred solution of 4-(3-isopropyl-1,2,4-triazol-1-yl)pyrimidine-2-carboxylic acid (i.e., the product of Step F) (180 mg, 0.77 mmol) in 1,2-dichloroethane (5 mL) was added (2S)-2,8-dimethyl-3,4-dihydro-2H-1,4-benzoxazine (i.e., the product of Step C in Synthesis Example 2) (126 mg, 0.77 mmol), propylphosphonic anhydride (50 wt.% in ethyl acetate, 0.9 mL) and triethylamine (0.45 mL, 3.2 mmol). The mixture was stirred at 60 °C for 3 h. Cold water was added to the mixture. The mixture was extracted with dichloromethane. The combined organic extracts were washed with brine, dried over anhydrous sodium sulfate, filtered and concentrated under reduced pressure. The resultant crude material was purified by column chromatography on silica gel to afford the title compound, a compound of the present invention, as a white solid (200 mg). 1H NMR (DMSO-d6, 100 °C) δ 9.20 (m, 1H), 8.99-8.98 (m, 1H), 7.87-7.86 (m, 1H), 7.17 (m, 1H), 6.94-6.92 (m, 1H), 6.64 (m, 1H), 4.53-4.52 (m, 1H), 4.02-3.99 (m, 1H), 3.55-3.50 (m, 1H), 3.16-3.09 (m, 1H), 2.18 (m, 3H), 1.39-1.32 (m, 9H). MS m/z: (M+H)+, C20H22N6O2, 379.2. SYNTHESIS EXAMPLE 6 Preparation of [(2S)-2,3-dihydro-2,8-dimethyl-4H-1,4-benzoxazin-4-yl][5-[3-(1- methylethyl)-1H-1,2,4-triazol-1-yl]-3-pyridazinyl]methanone (alternatively named [(2S)-2,8-dimethyl-2,3-dihydro-1,4-benzoxazin-4-yl]-[5-(3-isopropyl- 1,2,4-triazol-1-yl)pyridazin-3-yl]methanone) (Compound 22). Step A: Preparation of 3-chloro-5-(3-isopropyl-1,2,4-triazol-1-yl)pyridazine To a stirred solution of 3-isopropyl-1H-1,2,4-triazole (1.0 g, 9.0 mmol) in N,N-dimethylformamide (15 mL) was added potassium carbonate (2.8 g, 20.3 mmol) followed by 3,5-dichloropyridazine (1.5 g, 10.1 mmol) portionwise at room temperature. The mixture was stirred at 50 °C for 2 h. Cold water was added to the mixture. The mixture was extracted with ethyl acetate. The combined organic extracts were washed with brine, dried over anhydrous sodium sulfate and concentrated under reduced pressure. The resultant crude
material was purified by column chromatography on silica gel to afford the title compound as a white solid (1.0 g). d6) δ 9.81 (m, 1H), 9.52 (s, 1H), 8.34-8.33 (m, 1H), 3.12-3.07 (m, 1H), 1.31
C9H10ClN5, 224.1. Step B: Preparation of ethyl 5-(3-isopropyl-1,2,4-triazol-1-yl)pyridazine-3- carboxylate To a solution of 3-chloro-5-(3-isopropyl-1,2,4-triazol-1-yl)pyridazine (i.e., the product of Step A) (150 mg, 0.67 mmol) in ethanol (10 mL) was added sodium carbonate (71 mg, 0.67 mmol). The mixture was sparged with argon for 10 minutes. Palladium(II) acetate (15 mg, 0.07 mmol) and 1,1′-ferrocenediyl-bis(diphenylphosphine) (38 mg, 0.07 mmol) were added and the mixture was stirred under an atmosphere of carbon monoxide (50 psi) at 50 °C for 16 h in an autoclave. The mixture was cooled to room temperature, filtered through a pad of Celite® with dichloromethane. The filtrate was concentrated under reduced pressure. The resulting crude material was purified by column chromatography on silica gel to afford the title compound (85 mg). 1H NMR
δ 10.00 (m, 1H), 9.63 (s, 1H), 8.52-8.51 (m, 1H), 4.51-4.45 (m, 2H), 3.15-3.08 (m, 1H), 1.41-1.38 (m, 3H), 1.33-1.31 (m, 6H). Step C: Preparation of 5-(3-isopropyl-1,2,4-triazol-1-yl)pyridazine-3-carboxylic acid To a stirred solution of ethyl 5-(3-isopropyl-1,2,4-triazol-1-yl)pyridazine-3- carboxylate (i.e., the product of Step B) (450 mg, 1.7 mmol) in tetrahydrofuran (4.5 mL) and water (1.5 mL) was added lithium hydroxide monohydrate (160 mg, 3.8 mmol). The reaction mixture was stirred at room temperature for 3 h. The volatiles were evaporated and the mixture was diluted with water and washed with a 10% solution of ethyl acetate in hexanes. The aqueous phase was acidified with a 1 N aqueous hydrochloric acid solution and extracted with a solution of 20% methanol in dichloromethane. The combined organic extracts were washed with brine, dried over anhydrous sodium sulfate and concentrated under reduced pressure. The resultant crude material was washed with pentane to afford the title compound as a pale brown solid (320 mg). 1H NMR (DMSO-d6) δ 9.97 (m, 1H), 9.62 (s, 1H), 8.52 (m, 1H), 3.14-3.07 (m, 1H), 1.32 (d, 6H). MS m/z: (M−H)−, C10H11N5O2, 232.1.
Step D: Preparation of [(2S)-2,3-dihydro-2,8-dimethyl-4H-1,4-benzoxazin-4-yl][5-[3- (1-methylethyl)-1H-1,2,4-triazol-1-yl]-3-pyridazinyl]methanone (alternatively named [(2S)-2,8-dimethyl-2,3-dihydro-1,4-benzoxazin-4-yl]-[5- (3-isopropyl-1,2,4-triazol-1-yl)pyridazin-3-yl]methanone). To a stirred solution of 5-(3-isopropyl-1,2,4-triazol-1-yl)pyridazine-3-carboxylic acid (i.e., the product of Step C) (210 mg, 0.90 mmol) in 1,2-dichloroethane (4 mL) was added (2S)-2,8-dimethyl-3,4-dihydro-2H-1,4-benzoxazine (i.e., the product of Step C in Synthesis Example 2) (147 mg, 0.90 mmol), propylphosphonic anhydride (50 wt.% in ethyl acetate, 0.54 mL) and triethylamine (0.44 mL, 3.2 mmol) at 0 °C. The mixture was stirred at 60 °C for 3 h. Cold water was added to the mixture. The mixture was extracted with dichloromethane. The combined organic extracts were washed with brine, dried over anhydrous sodium sulfate and concentrated under reduced pressure. The crude material was purified by column chromatography on silica gel to afford the title compound, a compound of the present invention, as a pale yellow solid (240 mg). 1H NMR (DMSO-d6, 100 °C) δ 9.84 (m, 1H), 9.45 (m, 1H), 8.33 (m, 1H), 7.25 (m, 1H), 6.95- 6.93 (m, 1H), 6.68-6.64 (m, 1H), 4.56-4.53 (m, 1H), 4.13-4.09 (m, 1H), 3.58-3.53 (m, 1H), 3.17-3.10 (m, 1H), 2.19 (m, 3H), 1.40-1.34 (m, 9H). MS m/z: (M+H)+, C20H22N6O2, 379.2. SYNTHESIS EXAMPLE 7 Preparation of 2,3-dihydro-3-methyl-1-[2-methyl-5-[3-(1-methylethyl)-1H-1,2,4-triazol-1- yl]benzoyl]-4(1H)-quinolinone (alternatively named 1-[5-(3-isopropyl-1,2,4- triazol-1-yl)-2-methyl-benzoyl]-3-methyl-2,3-dihydroquinolin-4-one) (Compound 15). Step A: Preparation of 3-anilino-2-methyl-propanoic acid To a stirred solution of iodobenzene (4 g, 19.6 mmol) in
dimethylformamide (40 mL) and water (4 mL) was added 3-amino-2-methyl-propanoic acid (2.01 g, 19.5 mmol), copper(I) iodide (0.745 g, 3.91 mmol) and potassium carbonate (6.76 g, 48.9 mmol). The reaction mixture was stirred at 110 °C for 4 h. The mixture was diluted with water (100 mL), acidified to pH 2, extracted with ethyl acetate (2x). The combined organic extracts were dried over anhydrous sodium sulfate and concentrated under reduced pressure. The resultant crude material was purified by column chromatography on silica gel (eluting with a 50% solution of ethyl acetate in hexanes) to afford the title compound as a yellow oil (3.6 g).
7.17 (m, 2H), 6.76-6.72 (m, 1H), 6.66-6.64 (m, 2H), 3.45-3.40 (m, 1H), 3.29-3.24 (m, 1H), 2.87-2.82 (m, 1H), 1.29-1.27 (m, 3H).
Step B: Preparation of 3-methyl-2,3-dihydro-1H-quinolin-4-one. A solution of 3-anilino-2-methyl-propanoic acid (i.e., the product of Step A) (3.6 g, 20 mmol) in polyphosphoric acid (30 g) was stirred at 120 °C for 5 h. The mixture was cooled to room temperature. Ice-cold water was added (50 mL) to the mixture and the mixture was extracted with diethyl ether (x2). The combined organic extracts were dried over anhydrous sodium sulfate and concentrated under reduced pressure. The crude material was purified by column chromatography on silica gel (eluting with a solution of 30% ethyl acetate in hexanes) to afford the title compound as a pale yellow solid (1.8 g). 1H NMR (CDCl3) δ 7.87-7.85 (m, 1H), 7.31-7.26 (m, 1H), 6.75-6.72 (m, 1H), 6.66-6.65 (m, 1H), 3.57-3.54 (m, 1H), 3.31-3.26 (m, 1H), 2.72-2.67 (m, 1H), 1.24-1.22 (m, 3H). MS m/z: (M+H)+, C10H11NO, 161.99. Step C: Preparation of methyl 5-(3-isopropyl-1,2,4-triazol-1-yl)-2-methyl-benzoate In a sealed tube, a solution of methyl 5-bromo-2-methyl-benzoate (10 g, 44 mmol) in N,N-dimethylformamide (100 mL) was sparged with nitrogen gas then potassium carbonate (18.2 g, 132 mmol), 3-isopropyl-1H-1,2,4-triazole (7.30 g, 65.7 mmol), copper(I) iodide (1.67 g, 8.77 mmol) and trans-N,N′-dimethylcyclohexane-1,2-diamine (3.5 ml, 22 mmol) were added. The mixture was stirred at 120 °C for 16 h, diluted with water (200 mL), and extracted with ethyl acetate (2x). The combined organic extracts were dried over anhydrous sodium sulfate and concentrated under reduced pressure. The resultant crude material was purified by column chromatography on silica gel (eluting with a solution of 35% ethyl acetate in hexanes) to afford the title compound as an off-white semisolid (9.2 g). MS m/z: (M+H)+, C14H17N3O2, 260.27. Step D: Preparation of 5-(3-isopropyl-1,2,4-triazol-1-yl)-2-methyl-benzoic acid To a stirred solution of methyl 5-(3-isopropyl-1,2,4-triazol-1-yl)-2-methyl-benzoate (i.e., the product of Step C) (9.2 g, 35 mmol) in tetrahydrofuran (70 mL) and water (30 mL) was added lithium hydroxide monohydrate (2.23 g, 53 mmol). The reaction mixture was stirred at room temperature for 16 h. The mixture was concentrated under reduced, diluted with water (150 mL) and acidified to pH~2 with 1 N aqueous hydrochloric acid solution. The resulting precipitate was collected by filtration, washed with water (20 mL) and dried to afford the title compound as an off-white solid (4.5 g). MS m/z: (M+H)+, C13H15N3O2, 246.26.
Step E: Preparation of 2,3-dihydro-3-methyl-1-[2-methyl-5-[3-(1-methylethyl)-1H- 1,2,4-triazol-1-yl]benzoyl]-4(1H)-quinolinone (alternatively named 1-[5-(3- isopropyl-1,2,4-triazol-1-yl)-2-methyl-benzoyl]-3-methyl-2,3- dihydroquinolin-4-one). To a stirred solution of 5-(3-isopropyl-1,2,4-triazol-1-yl)-2-methyl-benzoic acid (i.e., the product of Step D) (2.8 g, 11.4 mmol) and 3-methyl-2,3-dihydro-1H-quinolin-4-one (i.e., the product of Step B) (2.06 g, 12.8 mmol) in pyridine (30 mL) at 0 °C was added phosphorus oxychloride (3.2 ml, 34 mmol). The mixture was stirred at room temperature for 5 h. The mixture was concentrated under reduced pressure, quenched with ice-cold water (50 mL) and extracted with ethyl acetate (2x). The combined organic extracts were dried over anhydrous sodium sulfate and concentrated under reduced pressure. The resultant crude material was purified by column chromatography on silica gel (eluting with a solution of 40% ethyl acetate in hexanes) to afford the title compound, a compound of the present invention, as an off-white solid (2.5 g). 1H NMR (DMSO-d6, 90 °C) δ 8.95 (s, 1H), 7.93-7.91 (m, 1H), 7.79-7.77 (m, 2H), 7.50-7.41 (m, 3H), 7.27-7.23 (m, 1H), 4.30-4.25 (m, 1H), 3.73-3.67 (m, 1H), 3.07-2.94 (m, 2H), 2.30 (s, 3H), 1.29 (d, 6H), 1.12 (d, 3H). MS m/z: (M+H)+, C23H24N4O2, 389.29. SYNTHESIS EXAMPLE 8 Preparation of (3,4-dihydro-4-hydroxy-3-methyl-1(2H)-quinolinyl)[2-methyl-5-[3-(1- methylethyl)-1H-1,2,4-triazol-1-yl]phenyl]methanone (alternatively named (4-hydroxy-3-methyl-3,4-dihydro-2H-quinolin-1-yl)-[5-(3-isopropyl-1,2,4- triazol-1-yl)-2-methyl-phenyl]methanone) (Compound 11). To a stirred solution of 2,3-dihydro-3-methyl-1-[2-methyl-5-[3-(1-methylethyl)-1H- 1,2,4-triazol-1-yl]benzoyl]-4(1H)-quinolinone (i.e., the product of Synthesis Example 7) (350 mg, 0.9 mmol) in methanol (4 mL) at 0 °C was added sodium borohydride (34 mg, 0.9 mmol). The reaction mixture was stirred at room temperature for 1 h, quenched with ice- cold water (20 mL), and extracted with ethyl acetate (2x). The combined organic extracts were dried over anhydrous sodium sulfate and concentrated under reduced pressure. The crude material was purified by column chromatography on silica gel (eluting with a solution of 60% ethyl acetate in hexanes) to afford the title compound, a compound of the present invention, (as a ~1.2:1 mixture of diastereomers) as an off-white solid (200 mg). MS m/z: (M+H)+, C23H26N4O2, 391.38.
SYNTHESIS EXAMPLE 9 Preparation of [4-(acetyloxy)-3,4-dihydro-3-methyl-1(2H)-quinolinyl][2-methyl-5-[3-(1- methylethyl)-1H-1,2,4-triazol-1-yl]phenyl]methanone (alternatively named [1-[5-(3-isopropyl-1,2,4-triazol-1-yl)-2-methyl-benzoyl]-3-methyl-3,4- dihydro-2H-quinolin-4-yl] acetate) (Compound 14). To a stirred solution of (3,4-dihydro-4-hydroxy-3-methyl-1(2H)-quinolinyl)[2- methyl-5-[3-(1-methylethyl)-1H-1,2,4-triazol-1-yl]phenyl]methanone (i.e., the product of Synthesis Example 8) (300 mg, 0.77 mmol) in N
dimethylformamide (4 mL) at 0 °C was added sodium hydride (60% dispersion in mineral oil, 33 mg, 0.83 mmol) and acetyl chloride (120 mg, 1.53 mmol). The mixture was stirred at room temperature for 2 h. The reaction was quenched with ice-cold water (10 mL), and extracted with ethyl acetate (2x). The combined organic extracts were dried over anhydrous sodium sulfate and concentrated under reduced pressure. The resultant crude material was purified by column chromatography on silica gel (eluting with a solution of 60% ethyl acetate in hexanes) to afford the title compound, a compound of the present invention, (as a mixture of diastereomers) as a colorless semisolid (110 mg). MS m/z: (M+H)+, C25H28N4O3, 433.33. SYNTHESIS EXAMPLE 10 Preparation of (5-chloro-3,4-dihydro-3-methyl-1(2H)-quinoxalinyl)[2-methyl-5-[3-(1- methylethyl)-1H-1,2,4-triazol-1-yl]phenyl]methanone (alternatively named (5-chloro-3-methyl-3,4-dihydro-2H-quinoxalin-1-yl)-[5-(3-isopropyl-1,2,4- triazol-1-yl)-2-methyl-phenyl]methanone (Compound 16). Step A: Preparation of 5-chloro-3-methyl-3,4-dihydro-1H-quinoxalin-2-one In a 15 mL sealed tube under an atmosphere of argon, 2-bromo-3-chloro-aniline (200 mg, 0.97 mmol) was dissolved in anhydrous dimethyl sulfoxide (4 mL). 2-aminopropanoic acid (174 mg, 1.95 mmol) was added followed by addition of tripotassium phosphate (413 mg, 1.95 mmol), N,N′-dimethylethylenediamine (17.1 mg, 0.194 mmol) and copper(I) chloride (0.96 mg, 0.0097 mmol). The reaction vessel was sealed and heated at 110 °C for 24 hours. The mixture was cooled to room temperature, quenched with ice cold water, and extracted with ethyl acetate. The combined organic extracts were washed with water and brine, dried over anhydrous sodium sulfate, concentrated under reduced pressure and purified by normal phase MPLC (eluting with gradient of 0 to 30% solution of ethyl acetate in hexanes) to afford the title compound (100 mg). 1H NMR (DMSO-d6) δ 10.41 (br s, 1H), 6.91-6.89 (m, 1H), 6.71-6.69 (m, 1H), 6.63-6.59 (m, 1H), 5.76 (br s, 1H), 3.92-3.89 (m, 1H), 1.27 (d, 3H).
Step B: Preparation of 8-chloro-2-methyl-1,2,3,4-tetrahydroquinoxaline To a stirred solution of 5-chloro-3-methyl-3,4-dihydro-1H-quinoxalin-2-one (i.e., the product of Step A) (870 mg, 4.4 mmol) in tetrahydrofuran (10 mL) at 0 °C was added lithium aluminum hydride (2M in tetrahydrofuran, 5 equiv.). After the addition was complete, the temperature of the reaction was slowly raised to room temperature and stirring was continued for 16 h. The mixture was cooled to 0 °C, quenched with ice cold water and the resulting mixture was passed through a pad of Celite®. The filtrate was extracted with ethyl acetate and the combined organic extracts were washed with water and brine, dried over anhydrous sodium sulfate, concentrated under reduced pressure and the residue was purified by normal phase MPLC (eluting with a gradient of 0 to 30% ethyl acetate in hexanes) to afford the title compound (450 mg). 1H NMR (DMSO-d6) δ 6.47-6.44 (m, 1H), 6.38-6.33 (m, 2H), 5.69 (s, 1H), 4.88 (s, 1H), 3.39- 3.34 (m, 1H), 3.23-3.19 (m, 1H), 2.84-2.79 (m, 1H), 1.14 (d, 3H). Step C: Preparation of (5-chloro-3,4-dihydro-3-methyl-1(2H)-quinoxalinyl)[2- methyl-5-[3-(1-methylethyl)-1H-1,2,4-triazol-1-yl]phenyl]methanone (alternatively named (5-chloro-3-methyl-3,4-dihydro-2H-quinoxalin-1-yl)-[5- (3-isopropyl-1,2,4-triazol-1-yl)-2-methyl-phenyl]methanone. To a stirred solution of methyl 5-(3-isopropyl-1,2,4-triazol-1-yl)-2-methyl-benzoate (i.e., the product of Step C in Synthesis Example 7) (400 mg, 1.5 mmol) and 8-chloro-2- methyl-1,2,3,4-tetrahydroquinoxaline (i.e., the product of Step B) (417 mg, 2.3 mmol) in toluene (4 mL) was added triethylamine (0.43 ml, 3.1 mmol) and trimethylaluminum (3.1 mmol). The mixture was stirred at 100 °C for 16 h, cooled to 0 °C, quenched with saturated aqueous sodium bicarbonate solution, and extracted with ethyl acetate. The combined organic extracts were washed with water and brine, dried over anhydrous sodium sulfate and concentrated under reduced pressure. The crude material was purified by normal phase MPLC (eluting with a gradient of 0 to 30% ethyl acetate in hexanes) to afford the title compound (250 mg), a compound of the present invention. 1H NMR (acetone-d6) δ 8.86 (br s, 1H), 7.78 (m, 2H), 7.37 (br s, 1H), 7.01 (br s, 1H), 6.30 (br s, 2H), 5.39 (br s, 1H), 3.83 (br s, 1H), 3.27 (br s, 1H), 3.10-3.03 (m, 1H), 2.26 (br s, 3H), 1.33-1.28 (m, 9H). Note: most signals were broadened due to presence of rotameric forms of the amide. MS m/z: (M+H)+, C22H24ClN5O, 410.24.
SYNTHESIS EXAMPLE 11 Preparation of (5-chloro-3,4-dihydro-3,4-dimethyl-1(2H)-quinoxalinyl)[2-methyl-5-[3-(1- methylethyl)-1H-1,2,4-triazol-1-yl]phenyl]methanone (alternatively named (5-chloro-3,4-dimethyl-2,3-dihydroquinoxalin-1-yl)-[5-(3-isopropyl-1,2,4- triazol-1-yl)-2-methyl-phenyl]methanone) (Compound 19). To a stirred solution of (5-chloro-3,4-dihydro-3-methyl-1(2H)-quinoxalinyl)[2- methyl-5-[3-(1-methylethyl)-1H-1,2,4-triazol-1-yl]phenyl]methanone (i.e., the product of Synthesis Example 10) (150 mg, 0.37 mmol) and iodomethane (0.07 mL, 1.1 mmol) in tetrahydrofuran (5 mL) at 0 °C was added sodium hydride (60% dispersion in mineral oil, 22 mg, 0.55 mmol). The resulting mixture was allowed to warm to room temperature. After stirring for 16 h, the mixture was cooled in an ice-water bath, quenched with saturated aqueous ammonium chloride solution, and extracted with ethyl acetate. The combined organic extracts were washed with brine, dried over anhydrous sodium sulfate and concentrated under reduced pressure. The crude material was purified by normal phase MPLC (eluting with ethyl acetate in hexanes) to afford the title compound (30 mg), a compound of the present invention. 1H NMR (DMSO-d6, 100 °C) δ 8.98 (s, 1H), 7.76-7.72 (m, 2H), 7.38 (d, 1H), 7.17 (s, 1H), 7.11 (d, 1H), 6.70 (t, 1H), 4.18 (br d, 1H), 3.46-3.40 (m, 1H), 3.22-3.17 (m, 1H), 3.06 (septet, 1H), 3.00 (s, 3H), 2.26 (s, 3H), 1.31 (d, 6H), 1.11 (d, 3H). SYNTHESIS EXAMPLE 12 Preparation of [(3S)-3,4-dihydro-5-methyl-3-(trifluoromethyl)-1(2H)-quinolinyl][2- methoxy-5-[3-(1-methylethyl)-1H-1,2,4-triazol-1- yl]phenyl]methanone and [(3R)-3,4-dihydro-5-methyl-3- (trifluoromethyl)-1(2H)-quinolinyl][2-methoxy-5-[3-(1-methylethyl)- 1H-1,2,4-triazol-1-yl]phenyl]methanone. Step A: Preparation of (2-amino-6-methyl-phenyl)methanol. To a stirred mixture of 2-amino-6-methylbenzoic acid (10 g, 66 mmol) in tetrahydrofuran (200 mL) at 0 °C lithium aluminum hydride (1M in tetrahydrofuran, 99 mL, 99 mmol) was added over 30 minutes. The reaction mixture was stirred for 30 minutes, warmed to room temperature and then stirred for 16 h. The reaction was quenched using the Fieser method, filtered through a pad of Celite® with tetrahydrofuran. The filtrate was concentrated under reduced pressure and purified by trituration using n-pentane and diethyl ether to afford the title compound (8 g). 1H NMR (DMSO-d6) δ 6.85-6.81 (m, 1H), 6.50-6.48 (m, 1H), 6.37-6.35 (m, 1H), 4.88 (br s, 2H), 4.72-4.70 (m, 1H), 4.42-4.41 (m, 2H), 2.22 (s, 3H).
Step B: Preparation of 2-amino-6-methyl-benzaldehyde. To a stirred solution of (2-amino-6-methyl-phenyl)methanol (i.e., the product of Step A) (8 g, 58 mmol) in dichloromethane (200 mL) at room temperature was added activated manganese dioxide (25.3 g, 291 mmol) and heated at 40 °C for 5 h. The reaction mixture was then filtered through a pad of Celite® with dichloromethane. The filtrate was concentrated under reduced pressure and purified by column chromatography to afford the title compound (6 g). 1H NMR (DMSO-d6) δ 10.23 (s, 1H), 7.36 (br s, 2H), 7.16-7.12 (m, 1H), 6.60-6.58 (m, 1H), 6.35-6.33 (m, 1H), 2.49 (s, 3H). Step C: Preparation of 3,3,3-trifluoro-N-(2-formyl-3-methylphenyl)propanamide. To a stirred solution of 2-amino-6-methyl-benzaldehyde (i.e., the product of Step B) (4 g, 29.6 mmol) in ethyl acetate (50 mL) was added 3,3,3-trifluoropropionic acid (5.68 g, 44.4 mmol) and N,N′-dicyclohexylcarbodiimide (7.32 g, 35.5 mmol). The reaction mixture was heated at the reflux temperature of the reaction mixture for 16 h, diluted with hot water and extracted with ethyl acetate. The combined organic extract was dried over anhydrous sodium sulfate, filtered, concentrated under reduced pressure, and purified by column chromatography to afford the title compound as an off-white solid (4 g). 1H NMR (DMSO-d6) δ 11.11 (br s, 1H), 10.24 (s, 1H), 7.85-7.82 (m, 1H), 7.56-7.53 (m, 1H), 7.15-7.13 (m, 1H), 3.73-3.64 (m, 2H), 2.60 (s, 3H). Step D: Preparation of 5-methyl-3-(trifluoromethyl)-1H-quinolin-2-one. To a stirred solution of 3,3,3-trifluoro-N-(2-formyl-3-methyl-phenyl)propanamide (i.e., the product of Step C) (4 g, 16 mmol) in N,N-dimethylformamide (30 mL) was added potassium carbonate (4.51 g, 32.6 mmol). The reaction mixture was stirred at room temperature for 3 h. Cold water was added to the reaction mixture and extracted with ethyl acetate. The combined organic extract was dried over anhydrous sodium sulfate, filtered, concentrated under reduced pressure, and purified by trituration with diethyl ether to afford the title compound as an off-white solid (3 g). 1H NMR (DMSO-d6) δ 12.31 (br s, 1H), 8.43 (s, 1H), 7.55-7.51 (m, 1H), 7.23-7.21 (m, 1H), 7.12-7.10 (m, 1H), 2.58 (s, 3H). Step E: Preparation of 5-methyl-3-(trifluoromethyl)-1,2,3,4-tetrahydroquinoline. To 5-methyl-3-(trifluoromethyl)-1H-quinolin-2-one (i.e., the product of Step D) (1 g, 4.4 mmol) in tetrahydrofuran (20 mL) at 0 °C was added borane dimethyl sulfide complex (3.3 mL, 35 mmol). The reaction mixture was stirred at room temperature for 16 h. The reaction mixture was cooled with an ice bath, quenched with methanol, concentrated under reduced pressure, and extracted with ethyl acetate. The organic phase was washed with a saturated aqueous solution of sodium chloride, dried over anhydrous sodium sulfate, filtered,
concentrated under reduced pressure, and purified by column chromatography to afford the title compound (300 mg). 1H NMR (CDCl3) δ 6.95-6.91 (m, 1H), 6.58-6.56 (m, 1H), 6.43-6.41 (m, 1H), 3.85 (br s, 1H), 3.51-3.49 (m, 1H), 3.20-3.14 (m, 1H), 2.91-2.88 (m, 1H), 2.75-2.62 (m, 2H), 2.20 (s, 3H). Step F: Preparation of [(3S)-3,4-dihydro-5-methyl-3-(trifluoromethyl)-1(2H)- quinolinyl][2-methoxy-5-[3-(1-methylethyl)-1H-1,2,4-triazol-1-yl]- phenyl]methanone and [(3R)-3,4-dihydro-5-methyl-3-(trifluoro- methyl)-1(2H)-quinolinyl][2-methoxy-5-[3-(1-methylethyl)-1H-1,2,4- triazol-1-yl]phenyl]methanone. To a stirred solution of methyl 5-(3-isopropyl-1,2,4-triazol-1-yl)-2-methoxy-benzoate (0.92 g, 3.34 mmol) (i.e., the product of Step D in Synthesis Example 1) and 5-methyl-3- (trifluoromethyl)-1,2,3,4-tetrahydroquinoline (i.e., the product of Step E in Synthesis Example 12) (0.60 g, 2.79 mmol) in toluene (2 mL) at 0 °C was added trimethylaluminum (2M solution in toluene, 7 mL, 14 mmol) and triethylamine (3.9 mL, 28 mmol). The reaction mixture was heated at 70 °C for 8 h; quenched with an ice-cold, saturated, aqueous solution of ammonium chloride, and extracted with ethyl acetate. The organic phase was washed with a saturated aqueous solution of sodium chloride, dried over anhydrous sodium sulfate, filtered, concentrated under reduced pressure and purified by column chromatography to afford [3,4- dihydro-5-methyl-3-(trifluoromethyl)-1(2H)-quinolinyl][2-methoxy-5-[3-(1- methylethyl)-1H-1,2,4-triazol-1-yl]phenyl]methanone (530 mg), a compound of the present invention. 1H NMR (DMSO-d6, 100 °C) δ 8.91 (s, 1H), 7.79-7.76 (m, 2H), 7.06-7.03 (m, 1H), 6.95 (m, 1H), 6.86 (m, 2H), 4.24 (m, 1H), 3.63-3.54 (m, 4H), 3.21-3.11 (m, 2H), 2.78-2.72 (m, 1H), 2.29 (s, 3H), 1.33-1.31 (m, 6H). The enantiomers of [3,4-dihydro-5-methyl-3-(trifluoromethyl)-1(2H)-quinolin- yl][2-methoxy-5-[3-(1-methylethyl)-1H-1,2,4-triazol-1-yl]phenyl]methanone were separated by preparative high performance liquid chromatography (HPLC) to yield Compound 40, a compound of the present invention, with an optical rotation of [α] D 25 –91.5° (concentration = 0.513 in methanol); and Compound 41, also a compound of the present invention, with an optical rotation of [α] D 25 +87.8° (concentration = 0.515 in methanol).
SYNTHESIS EXAMPLE 13 Preparation of [(3S)-5-fluoro-3,4-dihydro-3-(methoxymethyl)-1(2H)-quinolinyl][2-methoxy- 5-[3-(1-methylethyl)-1H-1,2,4-triazol-1-yl]phenyl]methanone and [(3R)-5- fluoro-3,4-dihydro-3-(methoxymethyl)-1(2H)-quinolinyl][2-methoxy-5-[3-(1- methylethyl)-1H-1,2,4-triazol-1-yl]phenyl]methanone. Step A: Preparation of (2-amino-6-fluorophenyl)methanol. To a stirred solution of 2-amino-6-fluorobenzoic acid (10 g, 64 mmol) in anhydrous tetrahydrofuran (200 mL) at 0 °C was slowly added lithium aluminum hydride (2M solution in tetrahydrofuran, 39 mL, 78 mmol) and stirred at room temperature for 16 h. The reaction mixture was quenched using the Fieser method, filtered through a pad of Celite® with tetrahydrofuran. The filtrate was then concentrated and purified by silica gel column chromatography (eluting with a 50% solution of ethyl acetate in hexanes) to afford the title compound as an off-white solid (8 g).
δ 6.98-6.92 (m, 1H), 6.46-6.44 (m, 1H), 6.30-6.26 (m, 1H), 5.29 (br s, 2H), 4.96-4.94 (m, 1H), 4.45-4.44 (m, 2H). Step B: Preparation of 2-amino-6-fluorobenzaldehyde. To a stirred solution of (2-amino-6-fluorophenyl)methanol (i.e., the product of Step A) (8.2 g, 58 mmol) in dichloromethane (200 mL) at room temperature was added manganese dioxide (25.3 g, 291 mmol). After addition was complete, the temperature of the reaction mixture was slowly raised to the reflux temperature of the reaction mixture and maintained for 5 h. The reaction mixture was filtered through a pad of Celite® with dichloromethane. The filtrate was concentrated and purified by silica gel column chromatography (eluting with a 10% solution of ethyl acetate in hexanes) to afford the title compound as a yellow solid (6 g). 1H NMR (DMSO-d6) δ 10.15 (s, 1H), 7.51 (br s, 2H), 7.31-7.25 (m, 1H), 6.58-6.56 (m, 1H), 6.33-6.28 (m, 1H). Step C: Preparation of methyl 5-fluoroquinoline-3-carboxylate. To a stirred solution of 2-amino-6-fluorobenzaldehyde (i.e., the product of Step B) (2 g, 14 mmol) in toluene (30 mL) was added methyl 3,3-dimethoxypropanoate (5.33 g, 36 mmol) and p-toluenesulfonic acid (248 mg). A Dean-Stark apparatus and condenser were attached to the reaction container, and the reaction mixture was heated at the reflux temperature of the mixture for 7 h. The reaction mixture was then cooled and concentrated under reduced pressure. To the resultant material was added a saturated aqueous solution of sodium bicarbonate, and extracted with ethyl acetate. The organic extract was dried over anhydrous sodium sulfate, filtered, concentrated, and purified by silica gel column chromatography (eluting with a 7-8% solution of ethyl acetate in hexanes) to afford the title compound as an orange solid (600 mg).
1H NMR (CDCl3) δ 9.49 (s, 1H), 9.15 (s, 1H), 8.03-8.01 (m, 1H), 7.82-7.77 (m, 1H), 7.33- 7.29 (m, 1H), 4.03 (s, 3H). Step D: Preparation of (5-fluoroquinolin-3-yl)methanol. To a stirred solution of methyl 5-fluoroquinoline-3-carboxylate (i.e., the product of Step C) (1 g, 4.9 mmol) in dry tetrahydrofuran (25 mL) at −78 °C was slowly added lithium tri-tert-butoxyaluminum hydride (1M solution in tetrahydrofuran, 11 mL, 11 mmol). The reaction mixture was stirred at −78 °C for 2 h. The temperature of the reaction mixture was slowly raised to 0 °C, stirred for 1 h, quenched with an ice-cold, solution of saturated aqueous ammonium chloride, and extracted with ethyl acetate. The organic extract was dried over anhydrous sodium sulfate, filtered, concentrated, and purified by silica gel column chromatography (eluting with a 50 to 80% gradient solution of ethyl acetate in hexanes) to afford the title compound as a white solid (700 mg). 1H NMR (DMSO-d6) δ 8.94 (s, 1H), 8.36 (s, 1H), 7.89-7.87 (m, 1H), 7.75-7.69 (m, 1H), 7.47- 7.42 (m, 1H), 5.56-5.53 (m, 1H), 4.77-4.76 (m, 2H). Step E: Preparation of 5-fluoro-3-(methoxymethyl)quinoline. To a stirred solution of (5-fluoroquinolin-3-yl)methanol (i.e., the product of Step D) (650 mg, 3.67 mmol) in tetrahydrofuran (15 mL) at 0 °C was added sodium hydride (60% dispersion in mineral oil, 176 mg, 4.4 mmol), stirred cold for 10 minutes, warmed to room temperature and stirred for 15 minutes. The reaction mixture was cooled to 0 °C and iodomethane (0.27 mL, 4.3 mmol) was added. The progress of the reaction was monitored for completion by thin-layer chromatography (TLC) and LCMS. After completion, the reaction was quenched with cold water and extracted with ethyl acetate. The combined organic extract was washed with a saturated aqueous solution of sodium chloride, dried over anhydrous sodium sulfate, filtered, concentrated under reduced pressure, and purified by silica gel column chromatography on (eluting with a 10% solution of ethyl acetate in hexanes) to afford the title compound as a colorless liquid (510 mg). 1H NMR (CDCl3) δ 8.92 (s, 1H), 8.39 (s, 1H), 7.93-7.91 (m, 1H), 7.66-7.60 (m, 1H), 7.22- 7.20 (m, 1H), 4.67 (s, 2H), 3.47 (s, 3H). Step F: Preparation of 5-fluoro-1,2,3,4-tetrahydro-3-(methoxymethyl)quinoline. To a stirred solution of 5-fluoro-3-(methoxymethyl)quinoline (i.e., the product of Step E) (500 mg, 2.62 mmol) in a mixture of ethanol and water (3:1, 32 mL) was added ammonium formate (2.47 g, 39 mmol) and palladium (10% on carbon, 100 mg). The reaction mixture was heated at 80 °C in a sealed round bottom flask for 10 h. The mixture was filtered through a pad of Celite® with ethyl acetate. The filtrate was concentrated and the crude material was purified by silica gel column chromatography to afford the title compound as a colorless liquid (400 mg).
LCMS: m/z: 196.17 [M+H]+ Step G: Preparation of [(3S)-5-fluoro-3,4-dihydro-3-(methoxymethyl)-1(2H)- quinolinyl][2-methoxy-5-[3-(1-methylethyl)-1H-1,2,4-triazol-1- yl]phenyl]methanone and [(3R)-5-fluoro-3,4-dihydro-3-(methoxymethyl)- 1(2H)-quinolinyl][2-methoxy-5-[3-(1-methylethyl)-1H-1,2,4-triazol-1- yl]phenyl]methanone. To a stirred suspension of methyl 5-(3-isopropyl-1,2,4-triazol-1-yl)-2-methoxy- benzoate (i.e., the product of Step D in Synthesis Example 1) (677 mg, 2.46 mmol) and 5- fluoro-1,2,3,4-tetrahydro-3-(methoxymethyl)quinoline (i.e., the product of Step F in Example 13) (400 mg, 2.05 mmol) in toluene (2 mL) was added triethylamine (2.3 mL, 16.5 mmol) followed by trimethylaluminum (2M in toluene, 4.1 mL, 8.2 mmol) at 0 °C. The reaction mixture was heated at 80 °C for 6 h, quenched with a cold saturated aqueous solution of ammonium chloride, and extracted with ethyl acetate. The organic extract was washed with a saturated aqueous solution of sodium chloride, dried over anhydrous sodium sulfate, filtered, concentrated, and purified by silica gel column chromatography to afford [5-fluoro-3,4- dihydro-3-(methoxymethyl)-1(2H)-quinolinyl][2-methoxy-5-[3-(1-methylethyl)-1H-1,2,4- triazol-1-yl]phenyl]methanone. 1H NMR (DMSO-d6, 100 °C) δ 8.93 (s, 1H), 7.80-7.79 (m, 2H), 7.11-6.95 (m, 3H), 6.89-6.85 (m, 1H), 4.03 (m, 1H), 3.63 (s, 3H), 3.44-3.32 (m, 3H), 3.27 (s, 3H), 3.10-3.03 (m, 1H), 2.93- 2.91 (m, 1H), 2.45 (m, 1H), 2.43-2.33 (m, 1H), 1.32 (d, 6H). The enantiomers of [5-fluoro-3,4-dihydro-3-(methoxymethyl)-1(2H)-quinolinyl][2- methoxy-5-[3-(1-methylethyl)-1H-1,2,4-triazol-1-yl]phenyl]methanone were separated by chiral supercritical fluid chromatography (eluting with a 20:40:40:0.1 solution of hexane:dichloromethane:ethanol:isopropylamine on a 5 µm, 4.6 x 250 mm Chiralpak IG column to yield Compound 29 (99 mg), a compound of the present invention, with an optical rotation of [α] D 25 +74.6° (concentration = 0.516 in methanol); and Compound 30 (140 mg), also a compound of the present invention, with an optical rotation of [α] D 25 –68.5° (concentration = 0.518 in methanol). SYNTHESIS EXAMPLE 14 Preparation of 8-fluoro-3,4-dihydro-4-[2-methyl-5-[3-(1-methylethyl)-1H-1,2,4-triazol-1- yl]benzoyl]-2(1H)-quinoxalinone (Compound 47). Step A: Preparation of 8-fluoro-3,4-dihydro-2(1H)-quinoxalinone. To a solution of 2-bromo-6-fluoro-aniline (1.0 g, 5.3 mmol) in dimethyl sulfoxide (10 mL) was added tripotassium phosphate (2.24 g, 10.6 mmol) and degassed with argon for 15 minutes. To the degassed mixture was added copper(I) chloride (26 mg, 0.26 mmol) and glycine (0.79 g, 10.5 mmol). The mixture was stirred at 120 °C for 6 h, diluted with water
(100 mL) and extracted with ethyl acetate (2 x 100 mL). The combined organic extract was washed with a saturated aqueous solution of ammonium chloride (50 mL), dried over anhydrous sodium sulfate, filtered, concentrated under reduced pressure, and purified by silica gel column chromatography (eluting with 40 to 50% ethyl acetate in hexanes) to afford the title compound as an off-white solid (600 mg). 1H NMR
δ 10.24 (br s, 1H), 6.77-6.71 (m, 1H), 6.50-6.45 (m, 2H), 6.24 (br s, 1H), 3.73 (d, 2H). Step B: Preparation of 8-fluoro-3,4-dihydro-4-[2-methyl-5-[3-(1-methylethyl)-1H- 1,2,4-triazol-1-yl]benzoyl]-2(1H)-quinoxalinone (Compound 47). To a solution of 5-(3-isopropyl-1,2,4-triazol-1-yl)-2-methyl-benzoic acid (i.e., the product of Step D in Synthesis Example 7) (600 mg, 2.45 mmol) in chloroform (10 mL) at 0 °C was added thionyl chloride (0.9 mL, 12 mmol) followed by N,N-dimethylformamide (0.1 mL). The reaction mixture was stirred at 60 °C for 3 h. The reaction mixture was concentrated under reduced pressure to remove to provide an acid chloride intermediate (i.e., 5-(3-isopropyl-1,2,4-triazol-1-yl)-2-methyl-benzoyl chloride), which was used without further purification. To a stirred solution of 8-fluoro-3,4-dihydro-2(1H)-quinoxalinone (i.e., the product of Step A of Synthesis Example 14) (400 mg, 2.41 mmol) in dichloromethane (6 mL) at 0 °C was added triethylamine (0.4 mL, 2.9 mmol) followed by a solution of the acid chloride intermediate (i.e., 5-(3-isopropyl-1,2,4-triazol-1-yl)-2-methyl-benzoyl chloride)in dichloro- methane (6 mL). The reaction mixture was stirred at 25 °C for 16 h, diluted with water (100 mL) and extracted with ethyl acetate (2 x 100 mL). The combined organic extract was washed with a saturated aqueous solution of sodium chloride (50 mL), dried over sodium sulfate, filtered, concentrated, and purified by silica gel column chromatography (eluting with 40 to 50% ethyl acetate in hexanes). The resultant material was further purified by preparative high-performance liquid chromatography, to afford the title compound, a compound of the present invention, as an off-white solid (110 mg). 1H NMR (DMSO-d6, 90 °C) δ 10.58 (br s, 1H), 8.97 (s, 1H), 7.77-7.75 (m, 2H), 7.37-7.35 (m, 1H), 7.04-6.99 (m, 1H), 6.84-6.81 (m, 2H), 4.38 (br s, 2H), 3.02 (m, 1H), 2.15 (s, 3H), 1.30 (d, 6H). By the procedures described herein together with methods known in the art, the following compounds of Tables 1 to 817 can be prepared. The following abbreviations are used in the Tables which follow: t means tertiary, s means secondary, n means normal, i means iso, c means cyclo, Me means methyl, Et means ethyl, Pr means propyl, Bu means butyl, i-Pr means isopropyl, c-Pr means cyclopropyl, t-Bu means tertiary butyl, Ph means phenyl, OMe means methoxy, OEt means ethoxy, SMe means methylthio, -CN means
cyano, -NO2 means nitro, TMS means trimethylsilyl, SOMe means methylsulfinyl, C2F5 means CF 2 CF 3 and SO 2 Me means methylsulfonyl. In Tables 1 through 817, J-1 through J-25 have the following meanings as defined below in Exhibit 3. As illustrated below for J-1 to J-17, the bond projecting to the lower right is connected at the position designated in the tables (e.g., R1a and R3a). As illustrated below for J-18 to J-25, the bond projecting to the left represents the bond in Formula 1 connecting the aromatic ring containing X1-X4 to group “A” and the bond projecting to the right represents the bond of Formula 1 connecting the aromatic ring containing X1-X4 to the (C=Y) moiety. Exhibit 3
J-17
R 1b is H, X 1 is CH, X 2 is CH, X 3 is C-OMe, R 3b is H, R 3a is Me, R 4a is Cl and (R 4b )q is H. R 1a R 1a R 1a
Table 2 is constructed in the same manner as Table 1 above except that the Row Heading “R1b is H, X1 is CH, X2 is CH, X3 is C-OMe, R3b is H, R3a is Me, R4a is Cl and (R4b)q is H.” is replaced with the Row Heading listed for Table 2 below (i.e., “R1b is H, X1 is CH, X2 is CH, X3 is CH, R3b is H, R3a is Me, R4a is Cl and (R4b)q is H.”). Therefore the first entry in Table 2 is a compound of Formula 1 wherein R1b is H, X1 is CH, X2 is CH, X3 is CH, R3b is H, R3a is Me, R4a is Cl, (R4b)q is H and R1a is H. Tables 3 through 104 are constructed similarly, except that the row heading in Table 1 is replaced with the respective row headings shown below. Table Row Heading 2 R 1b is H, X 1 is CH, X 2 is CH, X 3 is CH, R 3b is H, R 3a is Me, R 4a is Cl and (R 4b )q is H. 3 R 1b is H, X 1 is CH, X 2 is CH, X 3 is C-F, R 3b is H, R 3a is Me, R 4a is Cl and (R 4b )q is H. 4 R 1b is H, X 1 is CH, X 2 is CH, X 3 is C-Cl, R 3b is H, R 3a is Me, R 4a is Cl and (R 4b )q is H. 5 R 1b is H, X 1 is CH, X 2 is CH, X 3 is C-Me, R 3b is H, R 3a is Me, R 4a is Cl and (R 4b )q is H. 6 R 1b is H, X 1 is CH, X 2 is CH, X 3 is C-Et, R 3b is H, R 3a is Me, R 4a is Cl and (R 4b )q is H. 7 R 1b is H, X 1 is CH, X 2 is CH, X 3 is C-CH=CH2, R 3b is H, R 3a is Me, R 4a is Cl and (R 4b )q is H. 8 R 1b is H, X 1 is CH, X 2 is CH, X 3 is C-CH2C≡CH, R 3b is H, R 3a is Me, R 4a is Cl and (R 4b )q is H. 9 R 1b is H, X 1 is CH, X 2 is CH, X 3 is C-OCH2C≡CH, R 3b is H, R 3a is Me, R 4a is Cl and (R 4b )q is H. 10 R 1b is H, X 1 is CH, X 2 is CH, X 3 is C-c-Pr, R 3b is H, R 3a is Me, R 4a is Cl and (R 4b )q is H.
Table Row Heading 103 R 1b is H, X 1 is CH, X 2 is CH, X 3 is C-Me, R 3b is H, R 3a is CF3, R 4a is Me and (R 4b )q is H. 104 R 1b is H, X 1 is CH, X 2 is CH, X 3 is C-Me, R 3b is H, R 3a is CF3, R 4a is F and (R 4b )q is H.
Table 105 is constructed the same as Table 1 except that the structure in Table 1 is replaced with the structure above for Table 105 wherein the row heading is X2 is CH, X3 is C-OMe, R3a is Me, Z is CH(Me); and R1a is as defined in Table 1. Table 106 through Table 282 Table 106 is constructed in the same manner as Table 105 except that the Row Heading “X2 is CH, X3 is C-OMe, R3a is Me and Z is CH(Me).” is replaced with the Row Heading listed for Table 106 below (i.e., “X2 is CH, X3 is C-OMe, R3a is Me and Z is C(Me)2.”). Therefore the first entry in Table 106 is a compound with the structure shown immediately above wherein X2 is CH, X3 is C-OMe, R3a is Me and Z is C(Me)2 and R1a is H. Tables 107 through 282 are constructed similarly, except that the row heading in Table 105 is replaced with the respective row headings shown below. Table Row Heading 106 X 2 is CH, X 3 is C-OMe, R 3a is Me and Z is C(Me)2. 107 X 2 is CH, X 3 is C-OMe, R 3a is Me and Z is CH(F). 108 X 2 is CH, X 3 is C-OMe, R 3a is Me and Z is CF2. 109 X 2 is CH, X 3 is C-OMe, R 3a is Me and Z is CH(OH). 110 X 2 is CH, X 3 is C-OMe, R 3a is Me and Z is CH(CF3). 111 X 2 is CH, X 3 is C-OMe, R 3a is Me and Z is CH(CH2OMe). 112 X 2 is CH, X 3 is C-OMe, R 3a is Me and Z is CH(OMe). 113 X 2 is CH, X 3 is C-OMe, R 3a is Me and Z is C(OMe)2. 114 X 2 is CH, X 3 is C-OMe, R 3a is Me and Z is CH(OEt). 115 X 2 is CH, X 3 is C-OMe, R 3a is Me and Z is CH(OCF3).
Table Row Heading 116 X 2 is CH, X 3 is C-OMe, R 3a is Me and Z is CH(OCF2H). 117 X 2 is CH, X 3 is C-OMe, R 3a is Me and Z is CH(OCFH2). 118 X 2 is CH, X 3 is C-OMe, R 3a is Me and Z is CH(SMe). 119 X 2 is CH, X 3 is C-OMe, R 3a is Me and Z is C(SMe)2. 120 X 2 is CH, X 3 is C-OMe, R 3a is Me and Z is CH(SO2Me). 121 X 2 is CH, X 3 is C-OMe, R 3a is Me and Z is CH(CH2SMe). 122 X 2 is CH, X 3 is C-OMe, R 3a is Me and Z is CH(CH2SO2Me). 123 X 2 is CH, X 3 is C-OMe, R 3a is Me and Z is CH(CN). 124 X 2 is CH, X 3 is C-OMe, R 3a is Me and Z is CH(C≡CH). 125 X 2 is CH, X 3 is C-OMe, R 3a is Me and Z is CH(SCF3). 126 X 2 is CH, X 3 is C-OMe, R 3a is Me and Z is CH(CH2SCF3). 127 X 2 is CH, X 3 is C-OMe, R 3a is Me and Z is CH(CH2CN). 128 X 2 is CH, X 3 is C-OMe, R 3a is Me and Z is CH(O-c-Pr). 129 X 2 is CH, X 3 is C-OMe, R 3a is Me and Z is CH(O(C=O)Me). 130 X 2 is CH, X 3 is C-OMe, R 3a is Me and Z is CH(O(C=O)Et). 131 X 2 is CH, X 3 is C-OMe, R 3a is Me and Z is CH(O(C=O)-t-Bu). 132 X 2 is CH, X 3 is C-OMe, R 3a is Me and Z is CH(O(C=O)CF3). 133 X 2 is CH, X 3 is C-OMe, R 3a is Me and Z is CH(O(C=O)-c-Pr). 134 X 2 is CH, X 3 is C-OMe, R 3a is Me and Z is CH(O(C=O)OMe). 135 X 2 is CH, X 3 is C-OMe, R 3a is Me and Z is CH(O(C=O)O-t-Bu). 136 X 2 is CH, X 3 is C-OMe, R 3a is Me and Z is CH(O(C=O)NHMe). 137 X 2 is CH, X 3 is C-OMe, R 3a is Me and Z is CH(O(C=O)NMe2). 138 X 2 is CH, X 3 is C-OMe, R 3a is Me and Z is C(R 3c )2 wherein the two R 3c groups are taken together to form a 3-membered ring,. 139 X 2 is CH, X 3 is C-OMe, R 3a is Me and Z is NH. 140 X 2 is CH, X 3 is C-OMe, R 3a is Me and Z is N(C=O)Me. 141 X 2 is CH, X 3 is C-OMe, R 3a is Me and Z is N(C=O)CF3. 142 X 2 is CH, X 3 is C-OMe, R 3a is Me and Z is N(C=O)OMe. 143 X 2 is CH, X 3 is C-OMe, R 3a is Me and Z is N(C=O)NHMe. 144 X 2 is CH, X 3 is C-OMe, R 3a is Me and Z is N(C=O)NMe2. 145 X 2 is CH, X 3 is C-OMe, R 3a is Me and Z is NSO2Me. 146 X 2 is CH, X 3 is C-OMe, R 3a is Me and Z is NSO2CF3. 147 X 2 is CH, X 3 is C-Me, R 3a is Me and Z is C(Me)2. 148 X 2 is CH, X 3 is C-Me, R 3a is Me and Z is CH(F). 149 X 2 is CH, X 3 is C-Me, R 3a is Me and Z is CF2.
Table Row Heading 150 X 2 is CH, X 3 is C-Me, R 3a is Me and Z is CH(OH). 151 X 2 is CH, X 3 is C-Me, R 3a is Me and Z is CH(CF3). 152 X 2 is CH, X 3 is C-Me, R 3a is Me and Z is CH(CH2OMe). 153 X 2 is CH, X 3 is C-Me, R 3a is Me and Z is CH(OMe). 154 X 2 is CH, X 3 is C-Me, R 3a is Me and Z is C(OMe)2. 155 X 2 is CH, X 3 is C-Me, R 3a is Me and Z is CH(OEt). 156 X 2 is CH, X 3 is C-Me, R 3a is Me and Z is CH(OCF3). 157 X 2 is CH, X 3 is C-Me, R 3a is Me and Z is CH(OCF2H). 158 X 2 is CH, X 3 is C-Me, R 3a is Me and Z is CH(OCFH2). 159 X 2 is CH, X 3 is C-Me, R 3a is Me and Z is CH(SMe). 160 X 2 is CH, X 3 is C-Me, R 3a is Me and Z is C(SMe)2. 161 X 2 is CH, X 3 is C-Me, R 3a is Me and Z is CH(SO2Me). 162 X 2 is CH, X 3 is C-Me, R 3a is Me and Z is CH(CH2SMe). 163 X 2 is CH, X 3 is C-Me, R 3a is Me and Z is CH(CH2SO2Me). 164 X 2 is CH, X 3 is C-Me, R 3a is Me and Z is CH(CN). 165 X 2 is CH, X 3 is C-Me, R 3a is Me and Z is CH(C≡CH). 166 X 2 is CH, X 3 is C-Me, R 3a is Me and Z is CH(SCF3). 167 X 2 is CH, X 3 is C-Me, R 3a is Me and Z is CH(CH2SCF3). 168 X 2 is CH, X 3 is C-Me, R 3a is Me and Z is CH(CH2CN). 169 X 2 is CH, X 3 is C-Me, R 3a is Me and Z is CH(O-c-Pr). 170 X 2 is CH, X 3 is C-Me, R 3a is Me and Z is CH(O(C=O)Me). 171 X 2 is CH, X 3 is C-Me, R 3a is Me and Z is CH(O(C=O)Et). 172 X 2 is CH, X 3 is C-Me, R 3a is Me and Z is CH(O(C=O)-t-Bu). 173 X 2 is CH, X 3 is C-Me, R 3a is Me and Z is CH(O(C=O)CF3). 174 X 2 is CH, X 3 is C-Me, R 3a is Me and Z is CH(O(C=O)-c-Pr). 175 X 2 is CH, X 3 is C-Me, R 3a is Me and Z is CH(O(C=O)OMe). 176 X 2 is CH, X 3 is C-Me, R 3a is Me and Z is CH(O(C=O)O-t-Bu). 177 X 2 is CH, X 3 is C-Me, R 3a is Me and Z is CH(O(C=O)NHMe). 178 X 2 is CH, X 3 is C-Me, R 3a is Me and Z is CH(O(C=O)NMe2). 179 X 2 and X 3 are CR 2 wherein the R 2 groups are taken together to form J-21, R 3a is Me and Z is C(Me) 2 . 180 X 2 and X 3 are CR 2 wherein the R 2 groups are taken together to form J-21, R 3a is Me and Z is CH(F). 181 X 2 and X 3 are CR 2 wherein the R 2 groups are taken together to form J-21, R 3a is Me and Z is CF 2 .
Table Row Heading 182 X 2 and X 3 are CR 2 wherein the R 2 groups are taken together to form J-21, R 3a is Me and Z is CH(OH). 183 X 2 and X 3 are CR 2 wherein the R 2 groups are taken together to form J-21, R 3a is Me and Z is CH(CF 3 ). 184 X 2 and X 3 are CR 2 wherein the R 2 groups are taken together to form J-21, R 3a is Me and Z is CH(CH 2 OMe). 185 X 2 and X 3 are CR 2 wherein the R 2 groups are taken together to form J-21, R 3a is Me and Z is CH(OMe). 186 X 2 and X 3 are CR 2 wherein the R 2 groups are taken together to form J-21, R 3a is Me and Z is C(OMe) 2 . 187 X 2 and X 3 are CR 2 wherein the R 2 groups are taken together to form J-21, R 3a is Me and Z is CH(OEt). 188 X 2 and X 3 are CR 2 wherein the R 2 groups are taken together to form J-21, R 3a is Me and Z is CH(OCF 3 ). 189 X 2 and X 3 are CR 2 wherein the R 2 groups are taken together to form J-21, R 3a is Me and Z is CH(OCF 2 H). 190 X 2 and X 3 are CR 2 wherein the R 2 groups are taken together to form J-21, R 3a is Me and Z is CH(OCFH 2 ). 191 X 2 and X 3 are CR 2 wherein the R 2 groups are taken together to form J-21, R 3a is Me and Z is CH(SMe). 192 X 2 and X 3 are CR 2 wherein the R 2 groups are taken together to form J-21, R 3a is Me and Z is C(SMe) 2 . 193 X 2 and X 3 are CR 2 wherein the R 2 groups are taken together to form J-21, R 3a is Me and Z is CH(SO 2 Me). 194 X 2 and X 3 are CR 2 wherein the R 2 groups are taken together to form J-21, R 3a is Me and Z is CH(CH 2 SMe). 195 X 2 and X 3 are CR 2 wherein the R 2 groups are taken together to form J-21, R 3a is Me and Z is CH(CH 2 SO 2 Me). 196 X 2 and X 3 are CR 2 wherein the R 2 groups are taken together to form J-21, R 3a is Me and Z is CH(CN). 197 X 2 and X 3 are CR 2 wherein the R 2 groups are taken together to form J-21, R 3a is Me and Z is CH(C≡CH). 198 X 2 and X 3 are CR 2 wherein the R 2 groups are taken together to form J-21, R 3a is Me and Z is CH(SCF 3 ).
Table Row Heading 199 X 2 and X 3 are CR 2 wherein the R 2 groups are taken together to form J-21, R 3a is Me and Z is CH(CH 2 SCF 3 ). 200 X 2 and X 3 are CR 2 wherein the R 2 groups are taken together to form J-21, R 3a is Me and Z is CH(CH 2 CN). 201 X 2 and X 3 are CR 2 wherein the R 2 groups are taken together to form J-21, R 3a is Me and Z is CH(O-c-Pr). 202 X 2 and X 3 are CR 2 wherein the R 2 groups are taken together to form J-21, R 3a is Me and Z is CH(O(C=O)Me). 203 X 2 and X 3 are CR 2 wherein the R 2 groups are taken together to form J-21, R 3a is Me and Z is CH(O(C=O)Et). 204 X 2 and X 3 are CR 2 wherein the R 2 groups are taken together to form J-21, R 3a is Me and Z is CH(O(C=O)-t-Bu). 205 X 2 and X 3 are CR 2 wherein the R 2 groups are taken together to form J-21, R 3a is Me and Z is CH(O(C=O)CF 3 ). 206 X 2 and X 3 are CR 2 wherein the R 2 groups are taken together to form J-21, R 3a is Me and Z is CH(O(C=O)-c-Pr). 207 X 2 and X 3 are CR 2 wherein the R 2 groups are taken together to form J-21, R 3a is Me and Z is CH(O(C=O)OMe). 208 X 2 and X 3 are CR 2 wherein the R 2 groups are taken together to form J-21, R 3a is Me and Z is CH(O(C=O)O-t-Bu). 209 X 2 and X 3 are CR 2 wherein the R 2 groups are taken together to form J-21, R 3a is Me and Z is CH(O(C=O)NHMe). 210 X 2 and X 3 are CR 2 wherein the R 2 groups are taken together to form J-21, R 3a is Me and Z is CH(O(C=O)NMe 2 ). 211 X 2 is CH, X 3 is C-OMe, R 3a is CF3 and Z is C(Me)2. 212 X 2 is CH, X 3 is C-OMe, R 3a is CF3 and Z is CH(F). 213 X 2 is CH, X 3 is C-OMe, R 3a is CF3 and Z is CF2. 214 X 2 is CH, X 3 is C-OMe, R 3a is CF3 and Z is CH(OH). 215 X 2 is CH, X 3 is C-OMe, R 3a is CF3 and Z is CH(CF3). 216 X 2 is CH, X 3 is C-OMe, R 3a is CF3 and Z is CH(CH2OMe). 217 X 2 is CH, X 3 is C-OMe, R 3a is CF3 and Z is CH(OMe). 218 X 2 is CH, X 3 is C-OMe, R 3a is CF3 and Z is C(OMe)2. 219 X 2 is CH, X 3 is C-OMe, R 3a is CF3 and Z is CH(OEt). 220 X 2 is CH, X 3 is C-OMe, R 3a is CF3 and Z is CH(OCF3). 221 X 2 is CH, X 3 is C-OMe, R 3a is CF3 and Z is CH(OCF2H).
Table Row Heading
Table Row Heading
Table 283 is constructed the same as Table 105 except that the structure in Table 105 is replaced with the structure above for Table 283. Table 284 through Table 460 Table 284 is constructed the same as Table 106 except that the structure in Table 284 is replaced with the structure above for Table 283. Tables 285 through 460 are constructed in the same fashion as Tables 107 through 282. Table 461
Table 461 is constructed the same as Table 105 except that the structure in Table 105 is replaced with the structure above for Table 461. Table 462 through Table 638 Table 462 is constructed the same as Table 106 except that the structure in Table 462 is replaced with the structure above for Table 461. Tables 463 through 638 are constructed in the same fashion as Tables 107 through 282.
Table 639 is constructed the same as Table 105 except that the structure in Table 105 is eplaced with the structure above for Table 639. Table 640 through Table 816 Table 640 is constructed the same as Table 106 except that the structure in Table 640 is eplaced with the structure above for Table 639. Tables 642 through 816 are constructed in he same fashion as Tables 107 through 282. Table 817
A A
Formulation/Utility A compound of this invention will generally be used as a herbicidal active ingredient in a composition, i.e., formulation, with at least one additional component selected from the group consisting of surfactants, solid diluents and liquid diluents, which serves as a carrier. The formulation or composition ingredients are selected to be consistent with the physical properties of the active ingredient, mode of application and environmental factors such as soil type, moisture and temperature. Useful formulations include both liquid and solid compositions. Liquid compositions include solutions (including emulsifiable concentrates), suspensions, emulsions (including microemulsions, oil-in -water emulsions, flowable concentrates and/or suspoemulsions) and the like, which optionally can be thickened into gels. The general types of aqueous liquid compositions are soluble concentrate, suspension concentrate, capsule suspension, concentrated emulsion, microemulsion, oil-in-water emulsion, flowable concentrate and suspo-emulsion. The general types of nonaqueous liquid compositions are emulsifiable concentrate, microemulsifiable concentrate, dispersible concentrate and oil dispersion. The general types of solid compositions are dusts, powders, granules, pellets, prills, pastilles, tablets, filled films (including seed coatings) and the like, which can be water-dispersible (“wettable”) or water-soluble. Films and coatings formed from film- forming solutions or flowable suspensions are particularly useful for seed treatment. Active ingredient can be (micro)encapsulated and further formed into a suspension or solid
formulation; alternatively the entire formulation of active ingredient can be encapsulated (or “overcoated”). Encapsulation can control or delay release of the active ingredient. An emulsifiable granule combines the advantages of both an emulsifiable concentrate formulation and a dry granular formulation. High-strength compositions are primarily used as intermediates for further formulation. Sprayable formulations are typically extended in a suitable medium before spraying. Such liquid and solid formulations are formulated to be readily diluted in the spray medium, usually water, but occasionally another suitable medium like an aromatic or paraffinic hydrocarbon or vegetable oil. Spray volumes can range from about from about one to several thousand liters per hectare, but more typically are in the range from about ten to several hundred liters per hectare. Sprayable formulations can be tank mixed with water or another suitable medium for foliar treatment by aerial or ground application, or for application to the growing medium of the plant. Liquid and dry formulations can be metered directly into drip irrigation systems or metered into the furrow during planting. The formulations will typically contain effective amounts of active ingredient, diluent and surfactant within the following approximate ranges which add up to 100 percent by weight.
Solid diluents include, for example, clays such as bentonite, montmorillonite, attapulgite and kaolin, gypsum, cellulose, titanium dioxide, zinc oxide, starch, dextrin, sugars (e.g., lactose, sucrose), silica, talc, mica, diatomaceous earth, urea, calcium carbonate, sodium carbonate and bicarbonate, and sodium sulfate. Typical solid diluents are described in Watkins et al., Handbook of Insecticide Dust Diluents and Carriers, 2nd Ed., Dorland Books, Caldwell, New Jersey.
Liquid diluents include, for example, water, N,N-dimethylalkanamides (e.g., N,N-dimethylformamide), limonene, dimethyl sulfoxide, N-alkylpyrrolidones (e.g., N-methylpyrrolidinone), alkyl phosphates (e.g., triethyl phosphate), ethylene glycol, triethylene glycol, propylene glycol, dipropylene glycol, polypropylene glycol, propylene carbonate, butylene carbonate, paraffins (e.g., white mineral oils, normal paraffins, isoparaffins), alkylbenzenes, alkylnaphthalenes, glycerine, glycerol triacetate, sorbitol, aromatic hydrocarbons, dearomatized aliphatics, alkylbenzenes, alkylnaphthalenes, ketones such as cyclohexanone, 2-heptanone, isophorone and 4-hydroxy-4-methyl-2-pentanone, acetates such as isoamyl acetate, hexyl acetate, heptyl acetate, octyl acetate, nonyl acetate, tridecyl acetate and isobornyl acetate, other esters such as alkylated lactate esters, dibasic esters, alkyl and aryl benzoates and γ-butyrolactone, and alcohols, which can be linear, branched, saturated or unsaturated, such as methanol, ethanol, n-propanol, isopropyl alcohol, n-butanol, isobutyl alcohol, n-hexanol, 2-ethylhexanol, n-octanol, decanol, isodecyl alcohol, isooctadecanol, cetyl alcohol, lauryl alcohol, tridecyl alcohol, oleyl alcohol, cyclohexanol, tetrahydrofurfuryl alcohol, diacetone alcohol, cresol and benzyl alcohol. Liquid diluents also include glycerol esters of saturated and unsaturated fatty acids (typically C6–C22), such as plant seed and fruit oils (e.g., oils of olive, castor, linseed, sesame, corn (maize), peanut, sunflower, grapeseed, safflower, cottonseed, soybean, rapeseed, coconut and palm kernel), animal-sourced fats (e.g., beef tallow, pork tallow, lard, cod liver oil, fish oil), and mixtures thereof. Liquid diluents also include alkylated fatty acids (e.g., methylated, ethylated, butylated) wherein the fatty acids may be obtained by hydrolysis of glycerol esters from plant and animal sources, and can be purified by distillation. Typical liquid diluents are described in Marsden, Solvents Guide, 2nd Ed., Interscience, New York, 1950. The solid and liquid compositions of the present invention often include one or more surfactants. When added to a liquid, surfactants (also known as “surface-active agents”) generally modify, most often reduce, the surface tension of the liquid. Depending on the nature of the hydrophilic and lipophilic groups in a surfactant molecule, surfactants can be useful as wetting agents, dispersants, emulsifiers or defoaming agents. Surfactants can be classified as nonionic, anionic or cationic. Nonionic surfactants useful for the present compositions include, but are not limited to: alcohol alkoxylates such as alcohol alkoxylates based on natural and synthetic alcohols (which may be branched or linear) and prepared from the alcohols and ethylene oxide, propylene oxide, butylene oxide or mixtures thereof; amine ethoxylates, alkanolamides and ethoxylated alkanolamides; alkoxylated triglycerides such as ethoxylated soybean, castor and rapeseed oils; alkylphenol alkoxylates such as octylphenol ethoxylates, nonylphenol ethoxylates, dinonyl phenol ethoxylates and dodecyl phenol ethoxylates (prepared from the phenols and ethylene oxide,
propylene oxide, butylene oxide or mixtures thereof); block polymers prepared from ethylene oxide or propylene oxide and reverse block polymers where the terminal blocks are prepared from propylene oxide; ethoxylated fatty acids; ethoxylated fatty esters and oils; ethoxylated methyl esters; ethoxylated tristyrylphenol (including those prepared from ethylene oxide, propylene oxide, butylene oxide or mixtures thereof); fatty acid esters, glycerol esters, lanolin- based derivatives, polyethoxylate esters such as polyethoxylated sorbitan fatty acid esters, polyethoxylated sorbitol fatty acid esters and polyethoxylated glycerol fatty acid esters; other sorbitan derivatives such as sorbitan esters; polymeric surfactants such as random copolymers, block copolymers, alkyd peg (polyethylene glycol) resins, graft or comb polymers and star polymers; polyethylene glycols (pegs); polyethylene glycol fatty acid esters; silicone-based surfactants; and sugar-derivatives such as sucrose esters, alkyl polyglycosides and alkyl polysaccharides. Useful anionic surfactants include, but are not limited to: alkylaryl sulfonic acids and their salts; carboxylated alcohol or alkylphenol ethoxylates; diphenyl sulfonate derivatives; lignin and lignin derivatives such as lignosulfonates; maleic or succinic acids or their anhydrides; olefin sulfonates; phosphate esters such as phosphate esters of alcohol alkoxylates, phosphate esters of alkylphenol alkoxylates and phosphate esters of styryl phenol ethoxylates; protein-based surfactants; sarcosine derivatives; styryl phenol ether sulfate; sulfates and sulfonates of oils and fatty acids; sulfates and sulfonates of ethoxylated alkylphenols; sulfates of alcohols; sulfates of ethoxylated alcohols; sulfonates of amines and amides such as N,N- alkyltaurates; sulfonates of benzene, cumene, toluene, xylene, and dodecyl and tridecylbenzenes; sulfonates of condensed naphthalenes; sulfonates of naphthalene and alkyl naphthalene; sulfonates of fractionated petroleum; sulfosuccinamates; and sulfosuccinates and their derivatives such as dialkyl sulfosuccinate salts. Useful cationic surfactants include, but are not limited to: amides and ethoxylated amides; amines such as N-alkyl propanediamines, tripropylenetriamines and dipropylenetetramines, and ethoxylated amines, ethoxylated diamines and propoxylated amines (prepared from the amines and ethylene oxide, propylene oxide, butylene oxide or mixtures thereof); amine salts such as amine acetates and diamine salts; quaternary ammonium salts such as quaternary salts, ethoxylated quaternary salts and diquaternary salts; and amine oxides such as alkyldimethylamine oxides and bis-(2-hydroxyethyl)-alkylamine oxides. Also useful for the present compositions are mixtures of nonionic and anionic surfactants or mixtures of nonionic and cationic surfactants. Nonionic, anionic and cationic surfactants and their recommended uses are disclosed in a variety of published references including McCutcheon’s Emulsifiers and Detergents, annual American and International Editions published by McCutcheon’s Division, The Manufacturing Confectioner Publishing Co.; Sisely
and Wood, Encyclopedia of Surface Active Agents, Chemical Publ. Co., Inc., New York, 1964; and A. S. Davidson and B. Milwidsky, Synthetic Detergents, Seventh Edition, John Wiley and Sons, New York, 1987. Compositions of this invention may also contain formulation auxiliaries and additives, known to those skilled in the art as formulation aids (some of which may be considered to also function as solid diluents, liquid diluents or surfactants). Such formulation auxiliaries and additives may control: pH (buffers), foaming during processing (antifoams such polyorganosiloxanes), sedimentation of active ingredients (suspending agents), viscosity (thixotropic thickeners), in-container microbial growth (antimicrobials), product freezing (antifreezes), color (dyes/pigment dispersions), wash-off (film formers or stickers), evaporation (evaporation retardants), and other formulation attributes. Film formers include, for example, polyvinyl acetates, polyvinyl acetate copolymers, polyvinylpyrrolidone-vinyl acetate copolymer, polyvinyl alcohols, polyvinyl alcohol copolymers and waxes. Examples of formulation auxiliaries and additives include those listed in McCutcheon’s Volume 2: Functional Materials, annual International and North American editions published by McCutcheon’s Division, The Manufacturing Confectioner Publishing Co.; and PCT Publication WO 03/024222. The compound of Formula 1 and any other active ingredients are typically incorporated into the present compositions by dissolving the active ingredient in a solvent or by grinding in a liquid or dry diluent. Solutions, including emulsifiable concentrates, can be prepared by simply mixing the ingredients. If the solvent of a liquid composition intended for use as an emulsifiable concentrate is water-immiscible, an emulsifier is typically added to emulsify the active-containing solvent upon dilution with water. Active ingredient slurries, with particle diameters of up to 2,000 μm can be wet milled using media mills to obtain particles with average diameters below 3 μm. Aqueous slurries can be made into finished suspension concentrates (see, for example, U.S.3,060,084) or further processed by spray drying to form water-dispersible granules. Dry formulations usually require dry milling processes, which produce average particle diameters in the 2 to 10 μm range. Dusts and powders can be prepared by blending and usually grinding (such as with a hammer mill or fluid-energy mill). Granules and pellets can be prepared by spraying the active material upon preformed granular carriers or by agglomeration techniques. See Browning, “Agglomeration”, Chemical Engineering, December 4, 1967, pp 147–48, Perry’s Chemical Engineer’s Handbook, 4th Ed., McGraw-Hill, New York, 1963, pages 8–57 and following, and WO 91/13546. Pellets can be prepared as described in U.S.4,172,714. Water-dispersible and water-soluble granules can be prepared as taught in U.S. 4,144,050, U.S. 3,920,442 and DE 3,246,493. Tablets can be
prepared as taught in U.S. 5,180,587, U.S. 5,232,701 and U.S. 5,208,030. Films can be prepared as taught in GB 2,095,558 and U.S.3,299,566. For further information regarding the art of formulation, see T. S. Woods, “The Formulator’s Toolbox–Product Forms for Modern Agriculture” in Pesticide Chemistry and Bioscience, The Food–Environment Challenge, T. Brooks and T. R. Roberts, Eds., Proceedings of the 9th International Congress on Pesticide Chemistry, The Royal Society of Chemistry, Cambridge, 1999, pp.120–133. See also U.S.3,235,361, Col.6, line 16 through Col.7, line 19 and Examples 10–41; U.S.3,309,192, Col.5, line 43 through Col.7, line 62 and Examples 8, 12, 15, 39, 41, 52, 53, 58, 132, 138–140, 162–164, 166, 167 and 169–182; U.S.2,891,855, Col.3, line 66 through Col.5, line 17 and Examples 1–4; Klingman, Weed Control as a Science, John Wiley and Sons, Inc., New York, 1961, pp 81–96; Hance et al., Weed Control Handbook, 8th Ed., Blackwell Scientific Publications, Oxford, 1989; and Developments in formulation technology, PJB Publications, Richmond, UK, 2000. In the following Examples, all percentages are by weight and all formulations are prepared in conventional ways. Compound numbers refer to compounds in Index Tables A- C. Without further elaboration, it is believed that one skilled in the art using the preceding description can utilize the present invention to its fullest extent. The following Examples are, therefore, to be construed as merely illustrative, and not limiting of the disclosure in any way whatsoever. Percentages are by weight except where otherwise indicated. Example A High Strength Concentrate Compound 1 98.5% silica aerogel 0.5% synthetic amorphous fine silica 1.0% Example B Wettable Powder Compound 1 65.0% dodecylphenol polyethylene glycol ether 2.0% sodium ligninsulfonate 4.0% sodium silicoaluminate 6.0% montmorillonite (calcined) 23.0% Example C Granule Compound 1 10.0% attapulgite granules (low volatile matter, 0.71/0.30 mm; 90.0% U.S.S. No.25–50 sieves)
Example D Extruded Pellet Compound 1 25.0% anhydrous sodium sulfate 10.0% crude calcium ligninsulfonate 5.0% sodium alkylnaphthalenesulfonate 1.0% calcium/magnesium bentonite 59.0% Example E Emulsifiable Concentrate Compound 1 10.0% polyoxyethylene sorbitol hexoleate 20.0% C6–C10 fatty acid methyl ester 70.0% Example F Microemulsion Compound 1 5.0% polyvinylpyrrolidone-vinyl acetate copolymer 30.0% alkylpolyglycoside 30.0% glyceryl monooleate 15.0% water 20.0% Example G Suspension Concentrate Compound 1 35% butyl polyoxyethylene/polypropylene block copolymer 4.0% stearic acid/polyethylene glycol copolymer 1.0% styrene acrylic polymer 1.0% xanthan gum 0.1% propylene glycol 5.0% silicone based defoamer 0.1% 1,2-benzisothiazolin-3-one 0.1% water 53.7% Example H Emulsion in Water Compound 1 10.0% butyl polyoxyethylene/polypropylene block copolymer 4.0% stearic acid/polyethylene glycol copolymer 1.0% styrene acrylic polymer 1.0%
xanthan gum 0.1% propylene glycol 5.0% silicone based defoamer 0.1% 1,2-benzisothiazolin-3-one 0.1% aromatic petroleum based hydrocarbon 20.0 water 58.7% Example I Oil Dispersion Compound 1 25% polyoxyethylene sorbitol hexaoleate 15% organically modified bentonite clay 2.5% fatty acid methyl ester 57.5% The present disclosure also includes Examples A through I above except that “Compound 1” is replaced with any one of “Compound 2” through “Compound 20”. Test results indicate that the compounds of the present invention are highly active preemergent and/or postemergent herbicides and/or plant growth regulants. The compounds of the inention generally show highest activity for postemergence weed control (i.e., applied after weed seedlings emerge from the soil) and preemergence weed control (i.e., applied before weed seedlings emerge from the soil). Many of them have utility for broad-spectrum pre- and/or postemergence weed control in areas where complete control of all vegetation is desired such as around fuel storage tanks, industrial storage areas, parking lots, drive-in theaters, air fields, river banks, irrigation and other waterways, around billboards and highway and railroad structures. Many of the compounds of this invention, by virtue of selective metabolism in crops versus weeds, or by selective activity at the locus of physiological inhibition in crops and weeds, or by selective placement on or within the environment of a mixture of crops and weeds, are useful for the selective control of grass and broadleaf weeds within a crop/weed mixture. One skilled in the art will recognize that the preferred combination of these selectivity factors within a compound or group of compounds can readily be determined by performing routine biological and/or biochemical assays. Compounds of this invention may show tolerance to important agronomic crops including, but is not limited to, alfalfa, barley, cotton, wheat, rape, sugar beets, corn (maize), sorghum, soybeans, rice, oats, peanuts, vegetables, tomato, potato, perennial plantation crops including coffee, cocoa, oil palm, rubber, sugarcane, citrus, grapes, fruit trees, nut trees, banana, plantain, pineapple, hops, tea and forests such as eucalyptus and conifers (e.g., loblolly pine), and turf species (e.g.,
Kentucky bluegrass, St. Augustine grass, Kentucky fescue and Bermuda grass). Compounds of this invention can be used in crops genetically transformed or bred to incorporate resistance to herbicides, express proteins toxic to invertebrate pests (such as Bacillus thuringiensis toxin), and/or express other useful traits. Those skilled in the art will appreciate that not all compounds are equally effective against all weeds. Alternatively, the subject compounds are useful to modify plant growth. As the compounds of the invention have both preemergent and postemergent herbicidal activity, to control undesired vegetation by killing or injuring the vegetation or reducing its growth, the compounds can be usefully applied by a variety of methods involving contacting a herbicidally effective amount of a compound of the invention, or a composition comprising said compound and at least one of a surfactant, a solid diluent or a liquid diluent, to the foliage or other part of the undesired vegetation or to the environment of the undesired vegetation such as the soil or water in which the undesired vegetation is growing or which surrounds the seed or other propagule of the undesired vegetation. Undesired vegetation includes at least one selected from the group consisting of grass weeds and broadleaf weeds. Undesired vegetation is selected from the group consisting of annual bluegrass, Benghal dayflower, blackgrass, black nightshade, broadleaf signalgrass, Canada thistle, cheat, common cocklebur (Xanthium pensylvanicum), common ragweed, corn poppies, field violet, giant foxtail, goosegrass, green foxtail, guinea grass, hairy beggarticks, herbicide-resistant black grass, horseweed, Italian rye grass, jimsonweed, Johnson grass (Sorghum halepense), large crabgrass, little seed canary grass, morning glory, Pennsylvania smartweed, pitted morning glory, prickly sida, quackgrass, redroot pigweed, shattercane, shepherd's purse, silky windgrass, sunflower (as weed in potato), wild buckwheat (Polygonum convolvulus), wild mustard (Brassica kaber), wild oat (Avena fatua), wild pointsettia, yellow foxtail, and yellow nutsedge (Cyperus esculentus). A herbicidally effective amount of the compounds of this invention is determined by a number of factors. These factors include: formulation selected, method of application, amount and type of vegetation present, growing conditions, etc. In general, a herbicidally effective amount of compounds of this invention is about 0.001 to 20 kg/ha with a preferred range of about 0.004 to 1 kg/ha. One skilled in the art can easily determine the herbicidally effective amount necessary for the desired level of weed control. In one common embodiment, a compound of the invention is applied, typically in a formulated composition, to a locus comprising desired vegetation (e.g., crops) and undesired vegetation (i.e., weeds), both of which may be seeds, seedlings and/or larger plants, in contact with a growth medium (e.g., soil). In this locus, a composition comprising a compound of the
invention can be directly applied to a plant or a part thereof, particularly of the undesired vegetation, and/or to the growth medium in contact with the plant. Plant varieties and cultivars of the desired vegetation in the locus treated with a compound of the invention can be obtained by conventional propagation and breeding methods or by genetic engineering methods. Genetically modified plants (transgenic plants) are those in which a heterologous gene (transgene) has been stably integrated into the plant's genome. A transgene that is defined by its particular location in the plant genome is called a transformation or transgenic event. Genetically modified plant cultivars in the locus which can be treated according to the invention include those that are resistant against one or more biotic stresses (pests such as nematodes, insects, mites, fungi, etc.) or abiotic stresses (drought, cold temperature, soil salinity, etc.), or that contain other desirable characteristics. Plants can be genetically modified to exhibit traits of, for example, herbicide tolerance, insect-resistance, modified oil profiles or drought tolerance. Useful genetically modified plants containing single gene transformation events or combinations of transformation events are listed in Exhibit C. Additional information for the genetic modifications listed in Exhibit C can be obtained from publicly available databases maintained, for example, by the U.S. Department of Agriculture. The following abbreviations, T1 through T37, are used in Exhibit C for traits. A “-“ means the entry is not available; “tol.” means “tolerance” and “res.” means resistance.
-
Although most typically, compounds of the invention are used to control undesired vegetation, contact of desired vegetation in the treated locus with compounds of the invention may result in super-additive or enhanced effects with genetic traits in the desired vegetation, including traits incorporated through genetic modification. For example, resistance to phytophagous insect pests or plant diseases, tolerance to biotic/abiotic stresses or storage stability may be greater than expected from the genetic traits in the desired vegetation. Compounds of this invention can also be mixed with one or more other biologically active compounds or agents including herbicides, herbicide safeners, fungicides, insecticides, nematocides, bactericides, acaricides, growth regulators such as insect molting inhibitors and rooting stimulants, chemosterilants, semiochemicals, repellents, attractants, pheromones, feeding stimulants, plant nutrients, other biologically active compounds or entomopathogenic bacteria, virus or fungi to form a multi-component pesticide giving an even broader spectrum of agricultural protection. Mixtures of the compounds of the invention with other herbicides can broaden the spectrum of activity against additional weed species, and suppress the proliferation of any resistant biotypes. Thus the present invention also pertains to a composition comprising a compound of Formula 1 (in a herbicidally effective amount) and at least one additional biologically active compound or agent (in a biologically effective amount) and can further comprise at least one of a surfactant, a solid diluent or a liquid diluent. The other biologically active compounds or agents can be formulated in compositions comprising at least one of a surfactant, solid or liquid diluent. For mixtures of the present invention, one or more other biologically active compounds or agents can be formulated together with a compound of Formula 1, to form a premix, or one or more other biologically active compounds or agents can be formulated separately from the compound of Formula 1, and the formulations combined together before application (e.g., in a spray tank) or, alternatively, applied in succession. A mixture of one or more of the following herbicides with a compound of this invention may be particularly useful for weed control: allidochlor, acetochlor, acifluorfen and its sodium salt, aclonifen, acrolein (2-propenal), alachlor, alloxydim, ametryn, amicarbazone, amidosulfuron, aminocyclopyrachlor and its esters (e.g., methyl, ethyl) and salts (e.g., sodium, potassium), 4-amino-3-chloro-5-fluoro-6-(7-fluoro-1H-indol-6-yl)-2-pyridinecarboxylic 2- propyn-1-yl ester (CAS No.2251111-17-6), 4-amino-3-chloro-5-fluoro-6-(7-fluoro-1H-indol- 6-yl)-2-pyridinecarboxylic cyanomethyl ester (CAS No. 2251111-18-7), aminopyralid, amitrole, ammonium sulfamate, 2,5-anhydro-3,4-dideoxy-4-[[[(5S)-3-(3,5-difluorophenyl)-5- ethenyl-4,5-dihydro-5-isoxazolyl]carbonyl]amino]-threo-pentonic methyl ester (CAS No. 27499989-21-6), anilofos, anisiflupurin, asulam, atrazine, azimsulfuron, beflubutamid, beflubutamid-M, benazolin, benazolin-ethyl, bencarbazone, benfluralin, benfuresate,
benquitrione, bensulfuron-methyl, bensulide, bentazone, benzobicyclon, benzofenap, bicyclopyrone, bifenox, bilanafos, bipyrazone, bispyribac and its sodium salt, bixlozone, broclozone, bromacil, bromobutide, bromofenoxim, bromoxynil, bromoxynil octanoate, butachlor, butafenacil, butamifos, butralin, butroxydim, butylate, cafenstrole, carbetamide, 1- (2-carboxyethyl)-4-(2-pyrimidinyl)pyridazinium (CAS No. 2285384-11-2) and salts thereof, carfentrazone-ethyl, catechin, chlomethoxyfen, chloramben, chlorbromuron, chlorflurenol- methyl, chloridazon, chlorimuron-ethyl, 3-[2-chloro-5-[3,6-dihydro-3-methyl-2,6-dioxo-4- (trifluoromethyl)-1(2H)-pyrimidinyl]-4-fluorophenyl]-4,5-dihydro-5-methyl-5-isoxazole- carboxylic ethyl ester (CAS No.1949837-17-5), chlorotoluron, chlorpropham, chlorsulfuron, chlorthal-dimethyl, chlorthiamid, cinidon-ethyl, cinflubrolin, cinmethylin, cinosulfuron, clacyfos, clefoxydim, clethodim, clodinafop-propargyl, clomazone, clomeprop, clopyralid, clopyralid-olamine, cloransulam-methyl, cumyluron, cyanazine, cycloate, cyclopyranil, cyclopyrimorate, cyclosulfamuron, cycloxydim, cyhalofop-butyl, cypyrafluone, 2,4-D and its butotyl, butyl, isoctyl and isopropyl esters and its dimethylammonium, diolamine and trolamine salts, daimuron, dalapon, dalapon-sodium, dazomet, 2,4-DB and its dimethylammonium, potassium and sodium salts, desmedipham, desmetryn, dicamba and its diglycolammonium, dimethylammonium, potassium and sodium salts, dichlobenil, dichlorprop, diclofop-methyl, diclosulam, difenzoquat metilsulfate, diflufenican, diflufenzopyr, dimefuron, dimepiperate, dimesulfazet, dimethachlor, dimethametryn, dimethenamid, dimethenamid-P, dimethipin, dimethylarsinic acid and its sodium salt, dinitramine, dinoterb, dioxopyritrione, diphenamid, diquat dibromide, dithiopyr, diuron, DNOC, endothal, EPTC, epyrifenacil, esprocarb, ethalfluralin, ethametsulfuron-methyl, ethiozin, ethofumesate, ethoxyfen, ethoxysulfuron, etobenzanid, fenoxaprop-ethyl, fenoxaprop-P-ethyl, fenoxasulfone, fenpyrazone, fenquinotrione, fentrazamide, fenuron, fenuron-TCA, feproxydim, flamprop-methyl, flamprop-M-isopropyl, flamprop-M-methyl, flazasulfuron, florasulam, fluazifop-butyl, fluazifop-P-butyl, fluazolate, flucarbazone, flucetosulfuron, fluchloralin, fluchloraminopyr, fluchloraminopyr-tefuryl, flufenacet, flufenoximacil, flufenpyr, flufenpyr-ethyl, flumetsulam, flumiclorac-pentyl, flumioxazin, fluometuron, fluoroglycofen-ethyl, flupoxam, flupyrsulfuron-methyl and its sodium salt, flurenol, flurenol-butyl, fluridone, flurochloridone, fluroxypyr, flurtamone, flusulfinam, fluthiacet-methyl, fomesafen, foramsulfuron, fosamine-ammonium, glufosinate, glufosinate-ammonium, L-glufosinate-ammonium, glufosinate-P, glyphosate and its salts such as ammonium, isopropylammonium, potassium, sodium (including sesquisodium) and trimesium (alternatively named sulfosate), halauxifen, halauxifen-methyl, halosulfuron-methyl, haloxyfop-etotyl, haloxyfop-methyl, hexazinone, hydantocidin, icafolin, icafolin-methyl, imazamethabenz-methyl, imazamox, imazapic, imazapyr, imazaquin,
imazaquin-ammonium, imazethapyr, imazethapyr-ammonium, imazosulfuron, indanofan, indaziflam, indolauxipyr, indolauxipyr-cyanomethyl, iofensulfuron, iofensulfuron-sodium, iodosulfuron-methyl, iodosulfuron-sodium, ioxynil, ioxynil octanoate, ioxynil-sodium, ipfencarbazone, iptriazopyrid, isoproturon, isouron, isoxaben, isoxaflutole, isoxachlortole, lactofen, lancotrione, lenacil, linuron, maleic hydrazide, MCPA and its salts (e.g., MCPA- dimethylammonium, MCPA-potassium and MCPA-sodium, esters (e.g., MCPA-2-ethylhexyl, MCPA-butotyl) and thioesters (e.g., MCPA-thioethyl), MCPB and its salts (e.g., MCPB- sodium) and esters (e.g., MCPB-ethyl), mecoprop, mecoprop-P, mefenacet, mefluidide, mesosulfuron-methyl, mesotrione, metam-sodium, metamifop, metamitron, metazachlor, metazosulfuron, methabenzthiazuron, methylarsonic acid and its calcium, monoammonium, monosodium and disodium salts, methyldymron, metobenzuron, metobromuron, metolachlor, S-metolachlor, metosulam, metoxuron, metproxybicyclone, metribuzin, metsulfuron-methyl, molinate, monolinuron, naproanilide, napropamide, napropamide-M, naptalam, neburon, nicosulfuron, norflurazon, orbencarb, orthosulfamuron, oryzalin, oxadiargyl, oxadiazon, oxasulfuron, oxaziclomefone, oxyfluorfen, paraquat dichloride, pebulate, pelargonic acid, pendimethalin, penoxsulam, pentanochlor, pentoxazone, perfluidone, pethoxamid, pethoxyamid, phenmedipham, picloram, picloram-potassium, picolinafen, pinoxaden, piperophos, pretilachlor, primisulfuron-methyl, prodiamine, profoxydim, prometon, prometryn, propachlor, propanil, propaquizafop, propazine, propham, propisochlor, propoxycarbazone, propyrisulfuron, propyzamide, prosulfocarb, prosulfuron, pyraclonil, pyraflufen-ethyl, pyraquinate, pyrasulfotole, pyrazogyl, pyrazolynate, pyrazoxyfen, pyrazosulfuron-ethyl, pyribenzoxim, pyributicarb, pyridate, pyriflubenzoxim, pyriftalid, pyriminobac-methyl, pyrimisulfan, pyrithiobac, pyrithiobac-sodium, pyroxasulfone, pyroxsulam, quinclorac, quinmerac, quinoclamine, quizalofop-ethyl, quizalofop-P-ethyl, quizalofop-P-tefuryl, rimisoxafen, rimsulfuron, saflufenacil, sethoxydim, siduron, simazine, simetryn, sulcotrione, sulfentrazone, sulfometuron-methyl, sulfosulfuron, 2,3,6-TBA, TCA, TCA-sodium, tebutam, tebuthiuron, tefuryltrione, tembotrione, tepraloxydim, terbacil, terbumeton, terbuthylazine, terbutryn, tetflupyrolimet, thenylchlor, thiazopyr, thiencarbazone, thifensulfuron-methyl, thiobencarb, tiafenacil, tiocarbazil, tolpyralate, topramezone, tralkoxydim, tri-allate, triafamone, triasulfuron, triaziflam, tribenuron-methyl, triclopyr, triclopyr-butotyl, triclopyr-triethylammonium, tridiphane, trietazine, trifloxysulfuron, trifludimoxazin, trifluralin, triflusulfuron-methyl, tripyrasulfone, tritosulfuron, vernolate, 3- (2-chloro-3,6-difluorophenyl)-4-hydroxy-1-methyl-1,5-naphthyridin-2(1H)-one, 6-chloro-4- (2,7-dimethyl-1-naphthalenyl)-5-hydroxy-2-methyl-3(2H)-pyridazinone, 5-chloro-3-[(2- hydroxy-6-oxo-1-cyclohexen-1-yl)carbonyl]-1-(4-methoxyphenyl)-2(1H)-quinoxalinone, 2- chloro-N-(1-methyl-1H-tetrazol-5-yl)-6-(trifluoromethyl)-3-pyridinecarboxamide, 7-(3,5-
dichloro-4-pyridinyl)-5-(2,2-difluoroethyl)-8-hydroxypyrido[2,3-b]pyrazin-6(5H)-one), 4- (2,6-diethyl-4-methylphenyl)-5-hydroxy-2,6-dimethyl-3(2H)-pyridazinone), 5-[[(2,6- difluorophenyl)methoxy]methyl]-4,5-dihydro-5-methyl-3-(3-methyl-2-thienyl)isoxazole (previously methioxolin), 4-(4-fluorophenyl)-6-[(2-hydroxy-6-oxo-1-cyclohexen-1- yl)carbonyl]-2-methyl-1,2,4-triazine-3,5(2H,4H)-dione, methyl 4-amino-3-chloro-6-(4- chloro-2-fluoro-3-methoxyphenyl)-5-fluoro-2-pyridinecarboxylate, 2-methyl-3- (methylsulfonyl)-N-(1-methyl-1H-tetrazol-5-yl)-4-(trifluoromethyl)benzamide, 2-methyl-N- (4-methyl-1,2,5-oxadiazol-3-yl)-3-(methylsulfinyl)-4-(trifluoromethyl)benzamide, methyl 2-[2-[2-bromo-5-[3,6-dihydro-3-methyl-2,6-dioxo-4-(trifluoromethyl)-1(2H)-pyrimidinyl]-4- fluorophenoxy]phenoxy]-2-methoxyacetate, ethyl 3-[2-chloro-5-[3,6-dihydro-3-methyl-2,6- dioxo-4-(trifluoromethyl)-1(2H)-pyrimidinyl]-4-fluorophenyl]-4,5-dihydro-5-methyl-5- isoxazolecarboxylate, 1-(2-carboxyethyl)-4-(2-pyrimidinyl)pyridazinium and its salts and esters. The compound of Formula 1 can also be mixed with a compound selected from the group consisting of N-[2,4-dimethyl-5-(1-piperidinylcarbonyl)phenyl]-1,1,1-trifluoromethanesulfonamide; N-[2-chloro-4-methyl-5-(4-morpholinylcarbonyl)phenyl]-1,1,1- trifluoromethanesulfonamide; N-[2,4-dimethyl-5-(4-morpholinylcarbonyl)phenyl]-1,1,1- trifluoromethanesulfonamide; N-[2-chloro-4-methyl-5-(1-piperidinylcarbonyl)phenyl]-1,1,1- trifluoromethanesulfonamide; 3-fluoro-N,N,2,4-tetramethyl-5-[[(trifluoromethyl)sulfonyl]amino]benzamide; 1,1,1-trifluoro-N-[3-fluoro-2,4-dimethyl-5-(4- morpholinylcarbonyl)phenyl]methanesulfonamide; N-[2,4-dimethyl-5-(1-oxa-2-azaspiro[4.4]non-2-en-3-yl)phenyl]-1,1,1- trifluoromethanesulfonamide; N-[2,4-dimethyl-5-[(3aR,6aR)-3a,5,6,6a-tetrahydro-4H-cyclopent[d]isoxazol-3- yl]phenyl]-1,1,1-trifluoromethanesulfonamide; N-[2,4-dimethyl-5-(1-oxa-2-azaspiro[4.5]dec-2-en-3-yl)phenyl]-1,1,1- trifluoromethanesulfonamide; N-[2,4-dimethyl-5-(3a,4,7,7a-tetrahydro-5H-pyrano[4,3-d]isoxazol-3-yl)phenyl]-1,1,1- trifluoromethanesulfonamide; N-[2,4-dimethyl-5-(3a,6,7,7a-tetrahydro-4H-pyrano[3,4-d]isoxazol-3-yl)phenyl]-1,1,1- trifluoromethanesulfonamide; N-[2,4-dimethyl-5-(1-oxo-2-azaspiro[4.5]dec-2-yl)phenyl]-1,1,1-trifluoro-N- [(trifluoromethyl)sulfonyl]methanesulfonamide;
[[2,4-dimethyl-5-(1-oxa-2-azaspiro[4.4]non-2-en-3- yl)phenyl][(trifluoromethyl)sulfonyl]amino]methyl 2,2-dimethylpropanoate; [[2,4-dimethyl-5-(1-oxa-2-azaspiro[4.5]dec-2-en-3- yl)phenyl][(trifluoromethyl)sulfonyl]amino]methyl 2,2-dimethylpropanoate; [[2,4-dimethyl-5-[(3aR,6aR)-3a,5,6,6a-tetrahydro-4H-cyclopent[d]isoxazol-3- yl]phenyl][(trifluoromethyl)sulfonyl]amino]methyl 2,2-dimethylpropanoate; [[2,4-dimethyl-5-(1-oxo-2-azaspiro[4.5]dec-2- yl)phenyl][(trifluoromethyl)sulfonyl]amino]methyl 2,2-dimethylpropanoate; [[(trifluoromethyl)sulfonyl][2,3,4-trimethyl-5-(4- morpholinylcarbonyl)phenyl]amino]methyl 2,2-dimethylpropanoate; ethyl N-[(trifluoromethyl)sulfonyl]-N-[2,3,4-trimethyl-5-(1- piperidinylcarbonyl)phenyl]carbamate; [[(trifluoromethyl)sulfonyl][2,3,4-trimethyl-5-(1- piperidinylcarbonyl)phenyl]amino]methyl 2,2-dimethylpropanoate; 1,1,1-trifluoro-N-[2,3,4-trimethyl-5-(4- morpholinylcarbonyl)phenyl]methanesulfonamide; and [[(trifluoromethyl)sulfonyl][2,3,4-trimethyl-5-[(3aR,6aR)-3a,5,6,6a-tetrahydro-4H- cyclopent[d]isoxazol-3-yl]phenyl]amino]methyl 2,2-dimethylpropanoate. The compound of Formula 1 can also be mixed with a compound selected from the group consisting of [(2S)-8-chloro-2,3-dihydro-2-methyl-4H-1,4-benzoxazin-4-yl][2-methyl-5-[3-(1- methylethyl)-1H-1,2,4-triazol-1-yl]phenyl]methanone; [(2S)-8-chloro-2,3-dihydro-2-methyl-4H-1,4-benzoxazin-4-yl][5-[3-(1,1- dimethylethyl)-1H-1,2,4-triazol-1-yl]-2-methylphenyl]methanone; [(2S)-8-chloro-2,3-dihydro-2-methyl-4H-1,4-benzoxazin-4-yl][2-methoxy-5-[3- (1-methylethyl)-1H-1,2,4-triazol-1-yl]phenyl]methanone; [(2S)-8-chloro-2,3-dihydro-2-methyl-4H-1,4-benzoxazin-4-yl][5-(3-ethyl-1H- 1,2,4-triazol-1-yl)-2-methoxyphenyl]methanone; [(2S)-8-chloro-2,3-dihydro-2-methyl-4H-1,4-benzoxazin-4-yl][2-ethyl-5-[3-(1- methylethyl)-1H-1,2,4-triazol-1-yl]phenyl]methanone; [(2S)-8-chloro-2,3-dihydro-2-methyl-4H-1,4-benzoxazin-4-yl][5-(3-cyclobutyl- 1H-1,2,4-triazol-1-yl)-2-methylphenyl]methanone; [(2S)-8-chloro-2,3-dihydro-2-methyl-4H-1,4-benzoxazin-4-yl][3-(3-cyclobutyl- 1H-1,2,4-triazol-1-yl)phenyl]methanone; [(2S)-2,3-dihydro-2,8-dimethyl-4H-1,4-benzoxazin-4-yl][2-methoxy-5-(1H-1,2,4- triazol-1-yl)phenyl]methanone;
. The compound of Formula 1 can also be mixed with a compound selected from the group consisting of
2,2,2-trifluoroethyl (4S)-4-[[[(5S)-3-(3,5-difluorophenyl)-5-methyl-2-oxo-5- oxazolidinyl]carbonyl]amino]-1-cyclopentene-1-carboxylate; 2,2,2-trifluoroethyl (4S)-4-[[[(5R)-3-(3,5-difluorophenyl)-5-methyl-2-oxo-5- oxazolidinyl]carbonyl]amino]-1-cyclopentene-1-carboxylate; 2-(methylthio)ethyl (4S)-4-[[[(5S)-3-(3,5-difluorophenyl)-5-methyl-2-oxo-5- oxazolidinyl]carbonyl]amino]-1-cyclopentene-1-carboxylate; 2-(methylthio)ethyl (4S)-4-[[[(5R)-3-(3,5-difluorophenyl)-5-methyl-2-oxo-5- oxazolidinyl]carbonyl]amino]-1-cyclopentene-1-carboxylate; methyl (4S)-4-[[[(5S)-3-(3,5-difluorophenyl)-5-methyl-2-oxo-5- oxazolidinyl]carbonyl]amino]-1-cyclopentene-1-carboxylate; methyl (4S)-4-[[[(5R)-3-(3,5-difluorophenyl)-5-methyl-2-oxo-5- oxazolidinyl]carbonyl]amino]-1-cyclopentene-1-carboxylate; methyl (4S)-4-[[[3-(3,5-difluorophenyl)-5-methyl-2-oxo-5- oxazolidinyl]carbonyl]amino]-1-cyclopentene-1-carboxylate; 1-methylethyl (1S,4R)-4-[[[3-(3-chloro-5-fluorophenyl)-5-methyl-2-oxo-5- oxazolidinyl]carbonyl]amino]-2-cyclopentene-1-carboxylate; methyl (1S,4R)-4-[[[3-(3-chloro-5-fluorophenyl)-5-methyl-2-oxo-5- oxazolidinyl]carbonyl]amino]-2-cyclopentene-1-carboxylate; 2-methylpropyl (4S)-4-[[[3-(3,5-difluorophenyl)-5-methyl-2-oxo-5- oxazolidinyl]carbonyl]amino]-1-cyclopentene-1-carboxylate; (1S,4R)-4-[[[3-(3,5-difluorophenyl)-5-methyl-2-oxo-5- oxazolidinyl]carbonyl]amino]-2-cyclopentene-1-carboxylic acid; (4S)-4-[[[3-(3,5-difluorophenyl)-5-methyl-2-oxo-5- oxazolidinyl]carbonyl]amino]-1-cyclopentene-1-carboxylic acid; (1S,4R)-4-[[[3-(3-chloro-5-fluorophenyl)-5-methyl-2-oxo-5- oxazolidinyl]carbonyl]amino]-2-cyclopentene-1-carboxylic acid; and ethyl (1S,4R)-4-[[[3-(3,5-difluorophenyl)-5-methyl-2-oxo-5- oxazolidinyl]carbonyl]amino]-2-cyclopentene-1-carboxylate. The compound of Formula 1 can also be mixed with a compound selected from the group consisting of 4-[(E)-(3-bromo-1-naphthalenyl)(methoxyimino)methyl]-5-hydroxy-2,6-dimethyl- 3(2H)-pyridazinone; 4-[(Z)-(3-bromo-1-naphthalenyl)(methoxyimino)methyl]-5-hydroxy-2,6-dimethyl- 3(2H)-pyridazinone;
4-[(E)-(3-bromo-1-naphthalenyl)[(2-propyn-1-yloxy)imino]methyl]-5-hydroxy-2,6- dimethyl-3(2H)-pyridazinone; 4-[(E)-(3-bromo-1-naphthalenyl)(ethoxyimino)methyl]-5-hydroxy-2,6-dimethyl-3(2H)- pyridazinone; 4-[(Z)-(4-fluoro-1-naphthalenyl)[(2-propyn-1-yloxy)imino]methyl]-5-hydroxy-2,6- dimethyl-3(2H)-pyridazinone; and 4-[(E)-(4-fluoro-1-naphthalenyl)[(2-propyn-1-yloxy)imino]methyl]-5-hydroxy-2,6- dimethyl-3(2H)-pyridazinone. The compound of Formula 1 can also be mixed with a compound selected from the group consisting of 4-[[2-(4-fluorophenyl)-5-methyl-2H-1,2,3-triazol-4-yl]methyl]-2- (trifluoromethyl)pyridine; 4-[[2-(4-fluorophenyl)-5-methyl-2H-1,2,3-triazol-4-yl]oxy]-2- (trifluoromethyl)pyridine; 4-[[5-ethoxy-2-(4-fluorophenyl)-2H-1,2,3-triazol-4-yl]methyl]-2- (trifluoromethyl)pyridine; 4-[[5-methoxy-2-[4-(trifluoromethyl)phenyl]-2H-1,2,3-triazol-4-yl]methyl]-2- (trifluoromethyl)pyridine; 4-[[5-methyl-2-[4-(trifluoromethyl)phenyl]-2H-1,2,3-triazol-4-yl]methyl]-2- (trifluoromethyl)pyridine; 4-[[5-ethoxy-2-[4-(trifluoromethyl)phenyl]-2H-1,2,3-triazol-4-yl]methyl]-2- (trifluoromethyl)pyridine; 4-[[5-(2,2,2-trifluoroethoxy)-2-[4-(trifluoromethyl)phenyl]-2H-1,2,3-triazol-4- yl]methyl]-2-(trifluoromethyl)pyridine; 4-[[5-ethyl-3-[4-(trifluoromethyl)phenyl]-1H-1,2,4-triazol-1-yl]methyl]-2- (trifluoromethyl)pyridine; 4-[[3-(4-fluorophenyl)-5-propyl-1H-1,2,4-triazol-1-yl]methyl]-2- (trifluoromethyl)pyridine; 4-[[5-ethoxy-3-(4-fluorophenyl)-1H-1,2,4-triazol-1-yl]methyl]-2- (trifluoromethyl)pyridine; 4-[[3-(4-fluorophenyl)-1-propyl-1H-1,2,4-triazol-5-yl]methyl]-2- (trifluoromethyl)pyridine; and 4-[[3-(4-fluorophenyl)-5-methoxy-1H-1,2,4-triazol-1-yl]methyl]-2- (trifluoromethyl)pyridine. Other herbicides also include bioherbicides such as Alternaria destruens Simmons, Colletotrichum gloeosporiodes (Penz.) Penz. & Sacc., Drechsiera monoceras (MTB-951),
Myrothecium verrucaria (Albertini & Schweinitz) Ditmar: Fries, Phytophthora palmivora (Butl.) Butl. and Puccinia thlaspeos Schub. Preferred for better control of undesired vegetation (e.g., lower use rate such as from enhanced effects, broader spectrum of weeds controlled, or enhanced crop safety) or for preventing the development of resistant weeds are mixtures of a compound of this invention with a herbicide selected from the group consisting of atrazine, azimsulfuron, S-beflubutamid, benzisothiazolinone, bixlozone, beflubutamid-M, carfentrazone, carfentrazone-ethyl, chlorimuron, chlorimuron-ethyl, clomazone, clopyralid potassium, cloransulam-methyl, 2- [(2,4-dichlorophenyl)methyl]-4,4-dimethyl-3-isoxazolidinone, 2-[(2,5-dichloro- phenyl)methyl]-4,4-dimethyl-3-isoxazolidinone, ethametsulfuron-methyl, florasulam, flumetsulam, 4-(4-fluorophenyl)-6-[(2-hydroxy-6-oxo-1-cyclohexen-1-yl)carbonyl]-2- methyl-1,2,4-triazine-3,5-(2H,4H)-dione, flupyrsulfuron, flupyrsulfuron-methyl, fluthiacet, fluthiacet-methyl, fomesafen, imazethapyr, lenacil, mesotrione, metolachlor, S-metolachlor, metribuzin, metsulfuron-methyl, nicosulfuron, pethoxamid, picloram, pinoxaden, pyroxasulfone, pyroxsulam, quinclorac, rimisoxafen, sulfentrazone, tembotrione, tetflupyrolimet, thifensulfuron, thifensulfuron-methyl, tribenuron and tribenuron-methyl. Compounds of this invention can also be used in combination with plant growth regulators such as aviglycine, N-(phenylmethyl)-1H-purin-6-amine, epocholeone, gibberellic acid, gibberellin A4 and A7, harpin protein, mepiquat chloride, prohexadione calcium, prohydrojasmon, sodium nitrophenolate and trinexapac-methyl, and plant growth modifying organisms such as Bacillus cereus strain BP01. Compounds of this invention can also be used in combination with pheromones. Examples of pheromones include Formaldehyde; 2,2-Dibromoacetaldehyde; Acetaldehyde; 2- Methyl-2-propenal; 2-Methylpropanal; 2-Propenal; 3,3-Dibromo-2-propenal; Propanal; 2- Butenal; 2-Methyl-2-butenal; 2-Methylbutanal; 2-Methylenebutanal; 3-Methyl-2-butenal; 3- Methyl-3-butenal; 3-Methylbutanal; Butanal; (E)-2-Pentenal; 2-Methylenepentanal; 2- Pentenal; 3-Methyl-1-(vinyloxy)-butane; 4-Methylpentanal; 4-Pentenal; 5-Methylfurfural; Furan-2-carbaldehyde; Pentanal; (E)-2-Hexenal; (E)-2-Methyl-2-hexenal; (E)-3-Hexenal; (E)-4-oxo-2-Hexenal; (E,E)-2,4-Dimethyl-2,4-hexadienal; (E,E)-2,4-Hexadienal; (Z)-2- Hexenal; (Z)-3-Hexenal; (Z)-4-oxo-2-Hexenal; 1-Hexenal; 2,3-Dihydroxybenzaldehyde; 2- Hexenal; 3-((E)-2-Hexenoxy)-hexanal; 3,5-Dimethylhexanal; 3-Ethoxyhexanal; 3- Hydroxybenzaldehyde; 3-Hydroxyhexanal; 4-Hydroxy-3,5-dimethoxybenzaldehyde; 4- Hydroxybenzaldehyde; 5-Methylhexanal; Hexanal; (1R,2S,5R)-2-Methyl-5-((R)-1- oxopropan-2-yl)-cyclopentanecarbaldehyde; (1R,2S,5S)-2-Methyl-5-((R)-1-oxopropan-2- yl)-cyclopentanecarbaldehyde; (1R,5S)-6,6-Dimethylbicyclo[3.1.1]hept-2-ene-2- carbaldehyde; (1S,2S,5R)-2-Methyl-5-((R)-1-oxopropan-2-yl)-cyclopentanecarbaldehyde;
(3S,8R)-2-Methyl-5-(1-formylethyl)-1-cyclopentene-1-carbaldehyde; (3S,8S)-2-Methyl-5- (1-formylethyl)-1-cyclopentene-1-carbaldehyde; (5S,8S)-2-Methyl-5-(1-formylethyl)-1- cyclopentene-1-carbaldehyde; (E)-2-(2-Hydroxyethyl)-6-methyl-2,5-heptadienal; (E)-2-(2- Hydroxyethylidene)-6-methyl-5-heptenal; (E)-2-Heptenal; (E)-2-Isopropyl-5-methyl-2- hexenal; (E)-2-Methyl-2-heptenal; (E,Z)-2,4-Heptadienal; (R)-2,6-Dimethyl-5-heptenal; (S)- 4-(Prop-1-en-2-yl)-cyclohex-1-enecarbaldehyde; (Z)-2-Isopropyl-5-methyl-2-hexenal; (Z,Z)- 2,4-Heptadienal; 2-(3-Methylcyclopentyl)-propanal; 2-(3-Methylcyclopentyl)-propanal; 2,3,6-Tribromo-4,5-dihydroxybenzaldehyde; 2,3-Dibromo-4,5-dihydroxybenzaldehyde; 2,6- Dimethyl-5-heptenal; 2-Methoxybenzaldehyde; 2-Methyl-1-cyclopentenecarboxaldehyde; 2- Methyl-2-heptenal; 2-Methyl-5-(1-oxopropan-2-yl)-cyclopentanecarbaldehyde; 2- Methylcyclopent-1-enecarbaldehyde; 3,3-Dimethyl-5-oxo-7-oxabicyclo[4.1.0]heptane-1- carbaldehyde; 3,4-Dimethylbenzaldehyde; 3,5-Dibromo-4,5-dihydroxybenzaldehyde; 3,5- Dibromo-4-hydroxybenzaldehyde; 3-Bromo-4,5-dihydroxybenzaldehyde; 3-Bromo-4- hydroxybenzaldehyde; 3-Bromo-5-hydroxy-4-methoxybenzaldehyde; 3-Hydroxybenzene- 1,2-dicarbaldehyde; 3-Methylbenzaldehyde; 4-(Heptyloxy)-butanal; 4- Methoxybenzaldehyde; 5-(1-Formylethyl)-2-methyl-2-cyclopentene-1-carbaldehyde; 6- Methyl-5-heptenal; 6-Methylheptanal; Benzaldehyde; Cartilagineal; Cyclohexanedial; Heptanal; Taxifolial D; (1R,2S)-cis-2-Isopropenyl-1-methylcyclobutaneethanal; (1S,2R,3S)- 2-(1-Formylvinyl)-5-methylcyclopentanecarbaldehyde; (1S,2S,3S)-2-(1-Formylvinyl)-5- methylcyclopentanecarbaldehyde; (2Z,6E)-8-Chloro-6-chloromethyl-2-methyl-2,6- octadienal; (4S)-(3-Oxoprop-1-en-2-yl)-cyclohex-1-enecarbaldehyde; (E)-(3,3-Dimethyl)- cyclohexylideneacetaldehyde; (E)-2-(3,3-Dimethylcyclohexylidene)-acetaldehyde; (E)-2-(4- Methyl-3-pentenyl)-butenedial; (E)-2-(4-Methyl-3-pentenylidene)-butanedial; (E)-2,7- Octadienal; (E)-2-Methyl-2-octenal; (E)-2-Methyl-5-(3-furyl)-2-pentenal; (E)-2-Octenal; (E)- 3,7-Dimethyl-2,6-octadienal; (E)-3,7-Dimethyl-2,6-octadienal; (E)-3-Octenal; (E)-4-oxo-2- Octenal; (E)-7-Methyl-2-octenal; (E,E)-2,4-Octadienal; (E,E)-2,6-Dimethyl-8-hydroxy-2,6- octadienal; (E,E)-2,6-Octadienal; (E,E)-2,6-Octadienedial; (E,Z)-2,4-Octadienal; (E,Z)-2,6- Octadienal; (R)-1,2-Dimethyl-3-methylenecyclopentyl-acetaldehyde; (R)-3,7-Dimethyl-6- octenal; (Z)-(3,3-Dimethyl)-cyclohexylideneacetaldehyde; (Z)-2-(3,3- Dimethylcyclohexylidene)-acetaldehyde; (Z)-3,7-Dimethyl-2,6-octadienal; (Z,E)-3,7- Dimethyl-2,6-octadienal; 1-Octenal; 2-(1-Formylvinyl)-5-methylcyclopentanecarbaldehyde; 2-(3,4-Dihydroxyphenyl)-2-oxoacetaldehyde; 2,6,6-Trimethyl-1-cyclohexene-1- carbaldehyde; 2-Ethyloctanal; 2-Hydroxy-6-methylbenzaldehyde; 2-Methyl benzaldehyde; 2- Methyl-5-(1-formylethyl)-1-cyclopentene-1-carbaldehyde; 2-Octenal; 2-Phenylacetaldehyde; 2-Phenylpropenal; 3,4-Dihydroxyphenylglyoxal; 3,7-Dimethyl-6-octenal; 3-Ethoxy-4- hydroxybenzaldehyde; 3-Ethyl benzaldehyde; 3-Isopropyl-6-methyl benzaldehyde; 3-
Octenal; 3-oxo-4-Isopropylidene-1-cyclohexene-1-carboxyaldehyde; 4-Ethylbenzaldehyde; 4-Hydroxy-2-methyl benzaldehyde; 4-Hydroxy-3-methoxybenzaldehyde; 4-Isopropenyl-1- cyclohexene-1-carbaldehyde; 4-Isopropenyl-3-oxo-1-cyclohexene-1-carboxyaldehyde; 4S-4- Isopropenyl-3-oxo-1-cyclohexene-1-carboxyaldehyde; 5-Ethylcyclopent-1-ene- carbaldehyde; 6,6-Dimethylbicyclo[3.1.1]hept-2-ene-2-carbaldehyde; 6-Methyloctanal; 7- Methyloctanal; Anisomorphal; cis-2-Isopropenyl-1-methylcyclobutaneethanal; Octanal; Peruphasmal; (1R,2S,6R)-2,6-Dimethyl-3-oxabicyclo[4.2.0]octane-2-carbaldehyde; (E)-2- Methyl-2-nonenal; (E)-2-Nonenal; (E)-3-Phenyl-2-propenal; (E)-4,8-Nonadienal; (E)-8- Methyl-2-nonenal; (E,E)-2,4-Nonadienal; (E,E,E)-2,4,6-Nonatrienal; (E,E,Z)-2,4,6- Nonatrienal; (E,Z)-2,6-Nonadienal; (E,Z,Z)-2,4,6-Nonatrienal; (Z)-2-Methyl-2-nonenal; (Z)- 3-Nonenal; (Z)-4,8-Nonadienal; (Z)-4-Nonenal; (Z)-8-Methyl-2-nonenal; 2,6-Nonadienal; 2- Formyl-3-methylcyclopenteneacetaldehyde; 2-Nonenal; 2-Phenyl-2-butenal; 3-(4- Methoxyphenyl)-2-propenal; 3,5-di-tert-Butyl-4-hydroxybenzaldehyde; 3-Phenyl-2- propenal; 3-Phenylpropanal; 6-Ethyl benzaldehyde; 7,7-Dimethylbicyclo[4.1.0]hept-3-ene-3- carbaldehyde; 7-Methylnonanal; 8-Methylnonanal; 9-Acetyloxynonanal; Gibepyrone C; Nonanal; (4R,8R)-4,8-Dimethyldecanal; (4R,8S)-4,8-Dimethyldecanal; (E)-17,18,19,20- Tetranorloba-8,10,13(15)-trien-16-al; (E)-2,9-Decadienal; (E)-2-Decenal; (E)-2-Methyl-2- decenal; (E)-2-Methyl-3-(2,3-dibromo-4,5-dihydroxyphenyl)-propenal; (E)-4-oxo-2- Decenal; (E)-8-Hydroxy-4,8-dimethyl-4,9-decadienal; (E)-9-Methyl-2-decenal; (E,E)-2,4- Decadienal; (E,Z)-2,4-Decadienal; (Z)-4-Decenal; (Z)-5-Decenal; (Z)-9-Methyl-2-decenal; (Z,Z)-2,4-Decadienal; 1-Decenal; 2-Decenal; 2-Ethyldecanal; 3-(2,3-Dibromo-4,5- dihydroxyphenyl)-2-methylpropanal; 4,5-Dimethyldecanal; 4,8-Dimethyldecanal; Caraibical; Decanal; Rogiolal; (2E,4E)-2,6,10-Trimethylundeca-2,4,9-trienal; (2E,4E,7Z)-2,6,10- Trimethylundeca-2,4,7,9-tetraenal; (5E)-2,6,10-Trimethylundeca-5,9-dienal; (E)-2- Undecenal; (E)-6-Ethyl-2,10-dimethyl-5,9-undecadienal; (Z)-4-Undecenal; 10-Undecenal; 2- Butyl-2-octenal; 2-Undecenal; 3-Isopropyl-6-methyl-10-oxoundeca-2,6-dienal; 5-Methyl-2- phenyl-2-hexenal; 8-Isopropyl-5-methyl-3,4,4a,5,6,7,8,8a-octahydronaphthalene-2- carbaldehyde; Austrodoral; Oxytoxin 1; syn-4,6-Dimethylundecanal; Taxifolial A; Taxifolial B; Taxifolial C; Undecanal; (1R,6R,7S,10R)-1-Hydroxy-4-cadinen-15-al; (2R,7S,11R)-7- Acetoxy-2-hydroxynardosin-1(10)-en-12-al; (3R,5R,9R)-3,5,9-Trimethyldodecanal; (3S,6E)- 7-Ethyl-3,11-dimethyldodeca-6,10-dienal; (9R)-3,5,9-Trimethyldodecanal; (E)-10- Dodecenal; (E)-2-Dodecenal; (E)-3,7,11-Trimethyl-6,10-dodecadienal; (E)-5-Dodecenal; (E)- 6-Dodecenal; (E)-7-Dodecenal; (E)-8-Dodecenal; (E)-9,11-Dodecadienal; (E)-9-Dodecenal; (E,E)-3,7,11-Trimethyl-2,6,10-dodecatrienal; (E,E)-7-Ethyl-3,11-dimethyl-2,6,10- dodecatrienal; (E,E)-8,10-Dodecadienal; (E,E,E)-3,7-Dimethyl-8,11-dioxo-2,6,9- dodecatrienal; (E,E,Z)-3,7-Dimethyl-8,11-dioxo-2,6,9-dodecatrienal; (E,Z)-2,6-
Dodecadienal; (E,Z)-5,7-Dodecadienal; (E,Z)-7,9-Dodecadienal; (E,Z)-8,10-Dodecadienal; (R)-10-Oxo-isodauc-3-en-15-al; (S,E)-3,7,11-Trimethyl-6,10-dodecadienal; (Z)-2-Methyl-5- ((1R,5R,6S)-2,6-dimethylbicyclo[3.1.1]hept-2-en-6-yl)-pent-2-enal; (Z)-5-Dodecenal; (Z)-7- Dodecenal; (Z)-9,11-Dodecadienal; (Z)-9-Dodecenal; (Z,E)-3,7,11-Trimethyl-2,6,10- dodecatrienal; (Z,E)-5,7-Dodecadienal; (Z,E)-7-Ethyl-3,11-dimethyl-2,6,10-dodecatrienal; (Z,E)-8,10-Dodecadienal; (Z,Z)-5,7-Dodecadienal; 10-Methyldodecanal; 2,10-Dibromo-3- chloro-7-chamigrene; 2-Dodecenal; 2-Ethyldodecanal; 2-Formylguaiazulene; 3,7,11- Trimethyl-(E)-6,10-dodecadienal; 5-Hydroxy-8-methoxycalamanen-15-al; 5-Hydroxy-8- methoxycalamenene-15-al; Aplysinal; Debromoaplysinal; Dodecanal; Parahigginol D; Polygodial; Sclerosporal; Sinuketal; syn-4,6-Dimethyldodecanal; trans-Calamenen-13-al; (3R,5S,9R,7E,11E)-3,5,9,11-Tetramethyl-7,11-tridecadienal; (3S,4R,6E,10Z)-3,4,7,11- Tetramethyl-6,10-tridecadienal; (E,E)-3,5,9,11-Tetramethyltrideca-7,11-dienal; (Z)-4- Tridecenal; 13,14,15,16-Tetranorclerod-3-en-12-al; 13-Acetyloxytridecanal; 4,6-bis(4- Methylpent-3-en-1-yl)-6-methylcyclo-1,3-hexadienecarbaldehyde; Acanthodoral; Ancistrodial; Cespitulin F; Isoacanthodoral; Tridecanal; (E)-11,13-Tetradecadienal; (E)-11- Tetradecenal; (E,E)-8,10-Tetradecadienal; (E,Z)-4,9-Tetradecadienal; (E,Z)-8,10- Tetradecadienal; (Z)-11,13-Tetradecadienal; (Z)-11-Tetradecenal; (Z)-5-Tetradecenal; (Z)-7- Tetradecenal; (Z)-8-Tetradecenal; (Z)-9,13-Tetradecadien-11-ynal; (Z)-9-Tetradecenal; (Z,E)-9,11,13-Tetradecatrienal; (Z,E)-9,11-Tetradecadienal; (Z,E)-9,12-Tetradecadienal; (Z,Z)-5,8-Tetradecadienal; (Z,Z)-8,10-Tetradecadienal; (Z,Z)-9,11-Tetradecadienal; 10,12- Tetradecadienal; 2,4-Tetradecadienal; 2-Ethyltetradecanal; 3-oxo-13-Tetradecenal; 3-oxo- Tetradecanal; 5,8-Tetradecadienal; 5-Tetradecenal; Norasperenal A; Norasperenal B; Norasperenal C; Norasperenal D; Sargasal I; Sargasal II; Tetradecanal; (6R)-6- Acetoxidichotoma-3,14-diene-1,17-dial; (6R)-6-Hydroxydichotoma-3,14-diene-1,17-dial; (E,E)-3,7,11,15-Tetramethyl-6,10,14-hexadecatrienal; (E,Z)-6,8-Pentadecadienal; (E,Z)- 9,11-Pentadecadienal; (E,Z,Z)-2,6,9-Pentadecatrienal; (Z)-10-Pentadecenal; (Z)-2- Chloropentadec-2-enal; (Z)-6,14-Pentadecadienal; (Z,Z)-6,9-Pentadecadienal; (Z,Z)-9,11- Pentadecadienal; 2-Hexyl-2-decenal; Azamial A; Azamial B; Isopachydictyolal; Pentadecanal; Sinularial A; Umbellacin A; Xeniafaraunol A; (1R)-Pimaral; (E)-10- Hexadecenal; (E)-11-Hexadecenal; (E)-14-Hexadecenal; (E)-14-Methyl-8-hexadecenal; (E)- 3,7,11,15-Tetramethyl-2-hexadecenal; (E,E)-10,12-Hexadecadienal; (E,E)-10,14- Hexadecadienal; (E,E)-11,13-Hexadecadienal; (E,E)-9,11-Hexadecadienal; (E,E,E)-10,12,14- Hexadecatrienal; (E,E,E)-3,7,11,15-tetramethyl-2,6,10,14-hexadecatetraenal; (E,E,Z)- 10,12,14-Hexadecatrienal; (E,E,Z)-4,6,11-Hexadecatrienal; (E,E,Z,Z)-4,6,11,13- Hexadecatetraenal; (E,Z)-10,12-Hexadecadienal; (E,Z)-11,13-Hexadecadienal; (E,Z)-4,6- Hexadecadienal; (E,Z)-6,11-Hexadecadienal; (E,Z)-8,11-Hexadecadienal; (E,Z)-9,11-
Hexadecadienal; (R)-(E)-14-Methyl-8-hexadecenal; (R)-(Z)-14-Methyl-8-hexadecenal; (S)- (E)-14-Methyl-8-hexadecenal; (S)-(Z)-14-Methyl-8-hexadecenal; (Z)-10-Hexadecenal; (Z)- 11-Hexadecenal; (Z)-12-Hexadecenal; (Z)-13-Hexadecen-11-ynal; (Z)-14-Methyl-8- hexadecenal; (Z)-3,7,11,15-Tetramethyl-2-hexadecenal; (Z)-3-oxo-9-Hexadecenal; (Z)-7- Hexadecenal; (Z)-9-Hexadecenal; (Z,E)-10,12-Hexadecadienal; (Z,E)-11,13-Hexadecadienal; (Z,E)-7,11-Hexadecadienal; (Z,E)-9,11-Hexadecadienal; (Z,Z)-10,12-Hexadecadienal; (Z,Z)- 11,13-Hexadecadienal; (Z,Z)-7,10-Hexadecadienal; (Z,Z)-7,11-Hexadecadienal; (Z,Z)-9,11- Hexadecadienal; (Z,Z,E)-7,11,13-Hexadecatrienal; 11-Hexadecenal; 11-Hexadecynal; 13(16),14-Spongiadien-19-al; 2-Methylhexadecanal; 7-Hexadecenal; 9-Hexadecenal; Deacetyl-dihydro-nor-thuridillonal; Dictyodial A; Dihydro-nor-thuridillonal; Hexadecanal; Keikipukalide A; Keikipukalide B; Keikipukalide C; Keikipukalide D; Keikipukalide E; Nor- thuridillonal; Pseudoplexaural; Pukalide aldehyde; Sanadaol; (E)-2-Tridecyl-2-heptadecenal; (Z)-9-Heptadecenal; 1-Heptadecenal; 2-Heptadecenal; Globostelletin C; Globostelletin D; Heptadecanal; (E)-11-Octadecenal; (E)-13-Octadecenal; (E)-14-Octadecenal; (E)-2- Octadecenal; (E)-6-Octadecenal; (E,E)-11,14-Octadecadienal; (E,Z)-2,13-Octadecadienal; (E,Z)-3,13-Octadecadienal; (E,Z)-6,11-Octadecadienal; (Z)-11-Octadecenal; (Z)-13- Octadecenal; (Z)-9-Octadecenal; (Z,E)-13,15-Octadecadienal; (Z,Z)-11,13-Octadecadienal; (Z,Z)-13,15-Octadecadienal; (Z,Z)-3,13-Octadecadienal; (Z,Z)-9,12-Octadecadienal; (Z,Z,Z)-9,12,15-Octadecatrienal; 11-Octadecenal; 13,15-Octadecadienal; 13-Octadecenal; 16-Methyloctadecanal; 1-Octadecenal; 3,6-Dihydroxy-24-nor-9-oxo-9,11-secocholesta-7,22- dien-11-al; 9-Octadecenal; Methyloctadecanal; Octadecanal; Panicein B2; Panicein B3; Panicein C; (Z)-10-Nonadecenal; (Z)-9-Nonadecenal; 9(11)-Pargueren-16-al; Hyrtiosal; Nonadecanal; (2E,6Z,9Z)-2-Methyl-2,6,9-eicosatrienal; (Z)-11-Eicosenal; 11-Eicosenal; 12,18-di-Episcalaradial; 12b-(3'b-Hydroxybutanoyloxy)-20,24-dimethyl-24-oxo-scalara-16- en-25-al; 12b-(3'b-Hydroxypentanoyloxy)-20,24-dimethyl-24-oxo-scalara-16-en-25-al; 12- Deacetoxy-12-oxo-scalaradial; 12-Episcalaradial; 15-Eicosenal; 1-Eicosenal; 3-Deacetyl- 22,23-dihydro-24,28-dehydroluffasterol B; 3-Deacetylluffasterol B; 9-Eicosenal; Anthogorgiene B; Deacetylscalaradial; Eicosadienal; Eicosanal; Mooloolabene A; Mooloolabene B; Scalaradial; and combinations thereof. Of note as an example of pheromones include (Z)-5-decenyl acetate, dodecanyl acetate, (Z)-7-dodecenyl acetate, (E)-7-dodecenyl acetate, (Z)-8-dodecenyl acetate, (E)-8-dodecenyl acetate, (Z)-9-dodecenyl acetate, (E)-9-dodecenyl acetate, (E)-10-dodecenyl acetate, 11- dodecenyl acetate, (Z)-9,11-dodecadienyl acetate, (E)-9,11- dodecadienyl acetate, (Z)-11- tridecenyl acetate, (E)-11-tridecenyl acetate, tetradecanyl acetate, (E)-7-tetradecenyl acetate, (Z)-8-tetradecenyl acetate, (E)-8-tetradecenyl acetate, (Z)-9-tetradecenyl acetate, (E)-9- tetradecenyl acetate, (Z)-10-tetradecenyl acetate, (E)-10-tetradecenyl acetate, (Z)-11-
tetradecenyl acetate, (E)-11-tetradecenyl acetate, (Z)-12-pentadecenyl acetate, (E)-12- pentadecenyl acetate, hexadecanyl acetate, (Z)-7-hexadecenyl acetate, (Z)-11-hexadecenyl acetate, (E)-11-hexadecenyl acetate, octadecanyl acetate, (E,Z)-7,9- dodecadienyl acetate, (Z,E)-7,9-dodecadienyl acetate, (E,E)-7,9-dodecadienyl acetate, (Z,Z)-7,9-dodecadienyl acetate, (E,E)-8,10-dodecadienyl acetate, (E,Z)-9,12-dodecadienyl acetate, (E,Z)-4,7- tridecadienyl acetate, (E,E)-9,11-tetradecadienyl acetate, (Z,Z)-9,12-tetradecadienyl acetate, (Z,Z)-7,11-hexadecadienyl acetate, (E,Z)-7,11-hexadecadienyl acetate, (Z,E)-7,11- hexadecadienyl acetate, (E,E)-7,11-hexadecadienyl acetate, (Z,E)-3,13-octadecadienyl acetate, (E,Z)-3,13-octadecadienyl acetate, (E,E)-3,13-octadecadienyl acetate, decanol, (Z)-6- nonenol, (E)-6-nonenol, dodecanol, (Z)-5-decenol, 11-dodecenol, (Z)-7-dodecenol, (E)-7- dodecenol, (Z)-8-dodecenol, (E)-8-dodecenol, (E)-9-dodecenol, (Z)-9-dodecenol, (E)-9,11- dodecadienol, (Z)-9,11-dodecadienol, (Z,E)-5,7-dodecadienol, (E,E)-5,7-dodecadienol, (E,E)-8,10-dodecadien-1-ol, (E,Z)-8,10-dodecadienol, (Z,Z)-8,10-dodecadienol, (Z,E)-8,10- dodecadienol, (E,Z)-7,9- dodecadienol, (Z,Z)-7,9-dodecadienol, (E)-5-tetradecenol, (Z)-8- tetradecenol, (Z)-9- tetradecenol, (E)-9-tetradecenol, (Z)-10-tetradecenol, (Z)-11- tetradecenol, (E)-11-tetradecenol, (Z)-11-hexadecenol, (Z,E)-9,11-tetradecadienol, (Z,E)- 9,12-tetradecadienol, (Z,Z)-9,12- tetradecadienol, (Z, Z)-10,12-tetradecadienol, (Z,Z)-7,11- hexadecadienol, (Z,E)-7,11-hexadecadienol, (E)-14-methyl-8- hexadecen-1-ol, (Z)-14- methyl-8-hexadecen-l-ol, (E,E)-10,12-hexadecadienol, (E,Z)-10,12-hexadecadienol, dodecanal, (Z)-9-dodecanal, tetradecanal, (Z)-7- tetradecenal, (Z)-9-tetradecenal, (Z)-11- tetradecenal, (E)-11-tetradecenal, (E)-11,13-tetradecadienal, (E,E)-8,10-tetradecadienal, (Z,E)-9,11-tetradecadienal, (Z,E)-9,12- tetradecadienal, hexadecanal, (Z)-8-hexadecenal, (Z)- 9-hexadecenal, (Z)-10-hexadecenal, (E)-10-hexadecenal, (Z)-11-hexadecenal, (E)- hexadecenal, (Z)-12-hexadecenal, (Z)-13-hexadecenal, (Z)-14-methyl-8-hexadecenal, (E)-14- methyl-8-hexadecenal, (Z,Z)-7,11-hexadecadienal, (Z,E)-7,11-hexadecadienal, (Z,E)-9,11- hexadecadienal, (E,E)-10,12-hexadecadienal, (E,Z)-10,12-hexadecadienal, (Z,E)-10,12- hexadecadienal, (Z,Z)-10,12-hexadecadienal, (Z,Z)-11,13-hexadecadienal, octadecenal, (Z)- 11-octadecenal, (E)-13-octadecenal, (Z)-13-octadecenal, (Z)-5-decenyl 3-methylbutanoate, (+) cis-7,8-epoxy-2-methyloctadecane. Also of note as examples of pheromones include citral; geranial; neral; tetradecan-1-al; pentadecan-1-al; pentadecen-1-al; hexadecan-1-al; (Z)-9-hexadecen-1-al; (Z)-11-hexadecen- 1-al; (7E,9E)-undeca-7,9-dien-1-al; (11Z, 13Z)-hexadecadien-1-al; (9Z,12E)-tetradecadien-1- al; (8E,10E)-dodecadien-1-al; (11Z)-hexadecadien-1-al; (9Z)-tetradecen-1-al; 6,10-dimethyl- 5,9-undecadien-2-ol; (6E)-7,11-dimethyl-3-methylene-1,6,10-dodecatriene; [1S-(1a,2b,5 a)]- 4,6,6-trimethyl-Bicyclo[3.1.1]hept-3-en-2-ol; 10-Hexadecenal; (Z)-10-hexadecenal; (E)-10- hexadecenal; and combinations thereof.
General references for agricultural protectants (i.e., herbicides, herbicide safeners, insecticides, fungicides, nematocides, acaricides and biological agents) include The Pesticide Manual, 13th Edition, C. D. S. Tomlin, Ed., British Crop Protection Council, Farnham, Surrey, U.K., 2003 and The BioPesticide Manual, 2nd Edition, L. G. Copping, Ed., British Crop Protection Council, Farnham, Surrey, U.K., 2001. For embodiments where one or more of these various mixing partners are used, the mixing partners are typically used in the amounts similar to amounts customary when the mixture partners are used alone. More particularly in mixtures, active ingredients are often applied at an application rate between one-half and the full application rate specified on product labels for use of active ingredient alone. These amounts are listed in references such as The Pesticide Manual and The BioPesticide Manual. The weight ratio of these various mixing partners (in total) to the compound of Formula 1 is typically between about 1:3000 and about 3000:1. Of note are weight ratios between about 1:300 and about 300:1 (for example ratios between about 1:30 and about 30:1). One skilled in the art can easily determine through simple experimentation the biologically effective amounts of active ingredients necessary for the desired spectrum of biological activity. It will be evident that including these additional components may expand the spectrum of weeds controlled beyond the spectrum controlled by the compound of Formula 1 alone. In certain instances, combinations of a compound of this invention with other biologically active (particularly herbicidal) compounds or agents (i.e., active ingredients) can result in a greater-than-additive (i.e., enhanced) effect on weeds and/or a less-than-additive effect (i.e., safening) on crops or other desirable plants. Reducing the quantity of active ingredients released in the environment while ensuring effective pest control is always desirable. Ability to use greater amounts of active ingredients to provide more effective weed control without excessive crop injury is also desirable. When the enhanced effects of herbicidal mixtures of active ingredients occurs on weeds at application rates giving agronomically satisfactory levels of weed control, such combinations can be advantageous for reducing crop production cost and decreasing environmental load. When safening of herbicidal active ingredients occurs on crops, such combinations can be advantageous for increasing crop protection by reducing weed competition. Of note is a combination of a compound of the invention with at least one other herbicidal active ingredient. Of particular note is such a combination where the other herbicidal active ingredient has different site of action from the compound of the invention. In certain instances, a combination with at least one other herbicidal active ingredient having a similar spectrum of control but a different site of action will be particularly advantageous for resistance management. Thus, a composition of the present invention can further comprise
(in a herbicidally effective amount) at least one additional herbicidal active ingredient having a similar spectrum of control but a different site of action. Compounds of this invention can also be used in combination with herbicide safeners such as allidochlor, benoxacor, cloquintocet-mexyl, cumyluron, cyometrinil, cyprosulfonamide, daimuron, dichlormid, dicyclonon, dietholate, dimepiperate, fenchlorazole-ethyl, fenclorim, flurazole, fluxofenim, furilazole, isoxadifen-ethyl, mefenpyr- diethyl, mephenate, methoxyphenone naphthalic anhydride (1,8-naphthalic anhydride), oxabetrinil, N-(aminocarbonyl)-2-methylbenzenesulfonamide, N-(aminocarbonyl)- 2-fluorobenzenesulfonamide, 1-bromo-4-[(chloromethyl)sulfonyl]benzene (BCS), 4- (dichloroacetyl)-1-oxa-4-azospiro[4.5]decane (MON 4660), 2-(dichloromethyl)-2-methyl- 1,3-dioxolane (MG 191), ethyl 1,6-dihydro-1-(2-methoxyphenyl)-6-oxo-2-phenyl-5- pyrimidinecarboxylate, 2-hydroxy-N,N-dimethyl-6-(trifluoromethyl) p yridine-3-carboxamide, and 3-oxo-1-cyclohexen-l-yl 1-(3,4-dimethylphenyl)-l,6-dihydro-6-oxo-2-phenyl-5- pyrimidinecarboxylate, 2,2-dichloro-1-(2,2,5-trimethyl-3-oxazolidinyl)-ethanone and 2- methoxy-N-[[4-[[(methylamino)carbonyl]amino]phenyl]sulfonyl]-benzamide to increase safety to certain crops. Antidotally effective amounts of the herbicide safeners can be applied at the same time as the compounds of this invention, or applied as seed treatments. Therefore an aspect of the present invention relates to a herbicidal mixture comprising a compound of this invention and an antidotally effective amount of a herbicide safener. Seed treatment is particularly useful for selective weed control, because it physically restricts antidoting to the crop plants. Therefore a particularly useful embodiment of the present invention is a method for selectively controlling the growth of undesired vegetation in a crop comprising contacting the locus of the crop with a herbicidally effective amount of a compound of this invention wherein seed from which the crop is grown is treated with an antidotally effective amount of safener. Antidotally effective amounts of safeners can be easily determined by one skilled in the art through simple experimentation. Compounds of the invention can also be mixed with: (1) polynucleotides including but not limited to DNA, RNA, and/or chemically modified nucleotides influencing the amount of a particular target through down regulation, interference, suppression or silencing of the genetically derived transcript that render a herbicidal effect; or (2) polynucleotides including but not limited to DNA, RNA, and/or chemically modified nucleotides influencing the amount of a particular target through down regulation, interference, suppression or silencing of the genetically derived transcript that render a safening effect. Of note is a composition comprising a compound of the invention (in a herbicidally effective amount), at least one additional active ingredient selected from the group consisting
of other herbicides and herbicide safeners (in an effective amount), and at least one component selected from the group consisting of surfactants, solid diluents and liquid diluents. Table A1 lists specific combinations of a Component (a) with Component (b) illustrative of the mixtures, compositions and methods of the present invention. Compound No. (Compound Number) (i.e., Compound 1) in the Component (a) column is identified in Index Table A. The second column of Table A1 lists the specific Component (b) compound (e.g., “2,4-D” in the first line). The third, fourth and fifth columns of Table A1 lists ranges of weight ratios for rates at which the Component (a) compound is typically applied to a field-grown crop relative to Component (b) (i.e., (a):(b)). Thus, for example, the first line of Table A1 specifically discloses the combination of Component (a) (i.e., Compound 1 in Index Table A) with 2,4-D is typically applied in a weight ratio between 1:384–6:1. The remaining lines of Table A1 are to be construed similarly. TABLE A1
Table A2 is constructed the same as Table A1 above except that those entries below the “Component (a)” column heading are replaced with the respective Component (a) Column Entry shown below. Compound No. in the Component (a) column is identified in Index Table A. Thus, for example, in Table A2 the entries below the “Component (a)” column heading all recite “Compound 2” (i.e., Compound 2 identified in Index Table A), and the first line below the column headings in Table A2 specifically discloses a mixture of Compound 2 with 2,4-D. Tables A3 through A58 are constructed similarly.
Preferred for better control of undesired vegetation (e.g., lower use rate such as from enhanced effects, broader spectrum of weeds controlled, or enhanced crop safety) or for preventing the development of resistant weeds are mixtures of a compound of this invention with a herbicide selected from the group consisting of bixlozone, beflubutamid-M, carfentrazone, carfentrazone-ethyl, chlorimuron, chlorimuron-ethyl, clomazone, florasulam, flupyrsulfuron, flupyrsulfuron-methyl, fluthiacet, fluthiacet-methyl, mesotrione, metolachlor,
S-metolachlor, nicosulfuron, pinoxaden, pyroxasulfone, pyroxsulam, rimisoxafen, sulfentrazone, tembotrione, tetflupyrolimet, thifensulfuron, thifensulfuron-methyl, tribenuron and tribenuron-methyl. The following Tests demonstrate the control efficacy of the compounds of this invention against specific weeds. The weed control afforded by the compounds is not limited, however, to these species. See Index Tables A through D for compound descriptions. The following abbreviations are used in the Index Tables which follow: Me is methyl, i-Pr is isopropyl, OMe is methoxy, and OH is hydroxy. The abbreviation “Cmpd. No.” stands for “Compound Number”. The abbreviation “Ex.” stands for “Example” and is followed by a number indicating in which example the compound is prepared. The abbreviation “m.p.” stands for melting point. The abbreviation “M.S.” means mass spectra. Mass spectra are reported with an estimated precision within ±0.5 Da as the molecular weight of the highest isotopic abundance positively charged parent ion (M+1) formed by addition of H+ (molecular weight of 1) to the molecule having the highest isotopic abundance by using atmospheric pressure chemical ionization (AP+). The presence of molecular ions containing one or more higher atomic weight isotopes of lower abundance (e.g., 37Cl, 81Br) is not reported. Unless otherwise indicated, the compounds in Index Tables A, B and C are mixtures containing compounds of Formula 1' and 1''.
In Index Table A, the abbreviation “(1')-Me” in the R3a column denotes the absolute chirality of the asymmetric carbon center (*) is the configuration of the compound of Formula 1' as shown above. The abbreviation “(1'')-Me” in the R3a column denotes the configuration of the chiral compound of Formula 1''.
INDEX TABLE C
BIOLOGICAL EXAMPLES OF THE INVENTION TEST A Seeds of plant species selected from barnyardgrass (Echinochloa crus-galli), blackgrass (Alopecurus myosuroides), corn (Zea mays), green foxtail (Setaria viridis), giant foxtail (Setaria faberi), kochia (Bassia scoparia), wild oat (Avena fatua), Palmer amaranth (Palmer pigweed, Amaranthus palmeri), ragweed (common ragweed, Ambrosia artemisiifolia), redroot pigweed (Amaranthus retroflexus), Italian ryegrass (Lolium multiflorum), soybean (Glycine max) and wheat (Triticum aestivum) were planted into a blend of loam soil and sand and treated preemergence with a directed soil spray using test chemicals formulated in a non-phytotoxic solvent mixture which included a surfactant. At the same time, plants selected from these crops and weed species and galium (catchweed bedstraw, Galium aparine) and horseweed (Erigeron canadensis) were planted in pots containing the same blend of loam soil and sand and treated with postemergence applications of test chemicals formulated in the same manner. Plants ranged in height from 2 to 10 cm and were in the one- to two-leaf stage for the postemergence treatment. Treated plants and untreated controls were maintained in a greenhouse for 10 days, after which time all treated, plants were compared to untreated controls and visually evaluated for injury. Plant response ratings, summarized in Table A, are based on a 0 to 100 scale where 0 is no effect and 100 is complete control. A dash (–) response means no test result.
TEST B Plant species in the flooded paddy test selected from barnyardgrass (Echinochloa crus- galli), ducksalad (Heteranthera limosa), rice (Oryza sativa), and sedge, umbrella (small- flower umbrella sedge, Cyperus difformis) were grown to the 2-leaf stage for testing. At time of treatment, test pots were flooded to 3 cm above the soil surface, treated by application of test compounds directly to the paddy water, and then maintained at that water depth for the duration of the test. Treated plants and controls were maintained in a greenhouse for 13 days, after which time all species were compared to controls and visually evaluated. Plant response
ratings, summarized in Table B, are based on a scale of 0 to 100 where 0 is no effect and 100 is complete control. A dash (–) response means no test result.
Claims
CLAIMS What is claimed is: 1. A compound of Formula 1, all stereoisomers, N-oxides, and salts thereof wherein
A is a 5- or 6-membered heterocyclic ring, containing ring members selected from carbon atoms and up to 4 heteroatoms independently selected from up to 2 oxygen atoms, up to 2 sulfur atoms and up to 4 nitrogen atoms, one or two carbon or sulfur ring members of the heterocycle can optionally be in the oxidized form of a carbonyl, thiocarbonyl, sulfonyl, sulfinyl moiety, said ring bound to the remainder of Formula 1 through a carbon atom or a heteroatom, and optionally substituted with 1 to 4 R1; R1 is independently R1a, (R1b)m, R1c or any combination thereof; R1a is H, halogen, cyano, C1–C7 alkyl, C2–C7 alkenyl, C2–C6 alkynyl, C3–C7 cycloalkyl, C4–C7 cycloalkylalkyl, C4–C7 alkylcycloalkyl, C1–C7 haloalkyl, C1– C6 hydroxyalkyl, C3–C7 hydroxycycloalkyl, C1–C6 hydroxyhaloalkyl, C4–C8 alkoxycycloalkyl, C2–C7 alkoxyhaloalkyl, C2–C6 oxacycloalkyl, C3–C7 oxacycloalkylalkyl, C3–C7 alkyloxacycloalkyl, C2–C6 thiacycloalkyl, C3–C7 thiacycloalkylalkyl, C3–C7 alkylthiacycloalkyl, C2–C6 (O-thia)cycloalkyl, C3– C7(O-thia)cycloalkylalkyl, C3–C7 alkyl(O-thia)cycloalkyl, C2–C6 (O2thia)cycloalkyl, C3–C7 (O2thia)cycloalkylalkyl, C3–C7 alkyl(O2thia)cycloalkyl, C2–C6 haloalkenyl, C2–C6 haloalkynyl, C3–C7 halocycloalkyl, C4–C7 haloalkylcycloalkyl, C2–C7 alkoxyalkyl, C2–C7 haloalkoxyalkyl, C1–C7 alkoxy, C2–C5 alkenyloxy, C2–C5 alkynyloxy, C3–C7 cycloalkoxy, C4–C7 cycloalkoxyalkyl, C1–C7 haloalkoxy, C1–C7 alkylthio, C2– C7 alkylthioalkyl, C1–C5 alkylsulfinyl, C1–C5 alkylsulfonyl, C1–C5 alkylsulfonate, C1–C5 haloalkylthio, C1–C5 haloalkylsulfinyl, C1–C5
haloalkylsulfonyl, C2–C7 alkylsulfinylalkyl, C2–C7 alkylsulfonylalkyl, C2–C7 haloalkylthioalkyl, C2–C7 haloalkylsulfinylalkyl, C2–C7 haloalkylsulfonylalkyl, C4–C7 alkylthiocycloalkyl, C4–C7 alkylsulfinylcycloalkyl, C4–C7 alkylsulfonylcycloalkyl, C4–C7 haloalkylthiocycloalkyl, C4–C7 haloalkylsulfinylcycloalkyl, C4–C7 haloalkylsulfonylcycloalkyl, C2-C7 (HN=S(alkyl)=O)alkyl, C4-C7 (HN=S(alkyl)=O)cycloalkyl, C3-C7 (alkyl- N=S(alkyl)=O)alkyl, C5-C8 (alkyl-N=S(alkyl)=O)cycloalkyl, C3-C7 (cyano- N=S(alkyl)=O)alkyl, C4-C7 (alkylcarbonyl-N=S(alkyl)=O)alkyl, C4-C7 (alkoxycarbonyl-N=S(alkyl)=O)alkyl, C2–C5 cyanoalkyl, C4–C7 cyanocycloalkyl, C1–C4 nitroalkyl, C2–C6 dialkylamino, C2–C6 alkylcarbonyl, C3–C6 alkylcarbonylalkyl, C2–C6 alkoxycarbonyl, C3–C6 alkoxycarbonylalkyl or C3–C6 trialkylsilyl; each R1b is independently H, halogen, cyano, nitro, C1–C4 alkyl, C3–C5 cycloalkyl, C1–C4 haloalkyl, C3–C5 halocycloalkyl, C2–C4 alkoxyalkyl, C1–C4 alkoxy, C1– C4 alkylthio or C2–C4 alkoxycarbonyl; m is 0, 1 or 2; R1c is H, C1–C7 alkyl, C3–C7 cycloalkyl, C2–C6 oxacycloalkyl or C1–C7 haloalkyl; X1, X2, X3 and X4 are each independently N or CR2 wherein up to two X1, X2, X3 and X4 can be N; each R2 is independently H, halogen, cyano, nitro, hydroxy, C1–C5 alkyl, C2–C5 alkenyl, C2–C5 alkynyl, C3–C6 cycloalkyl, C4–C7 cycloalkylalkyl, C4–C7 alkylcycloalkyl, C1–C5 haloalkyl, C2–C5 haloalkenyl, C2–C5 haloalkynyl, C3– C6 halocycloalkyl, C4–C7 haloalkylcycloalkyl, C2–C5 alkoxyalkyl, C2–C5 haloalkoxyalkyl, C1–C5 alkoxy, C2–C5 alkenyloxy, C2–C5 alkynyloxy, C3–C7 cycloalkoxy, C4–C7 cycloalkoxyalkyl, C2–C6 oxacycloalkyl, C3–C7 oxacycloalkylalkyl, C2–C6 oxacycloalkoxy, C1–C5 haloalkoxy, C1–C5 alkylthio, C1–C4 alkylsulfinyl, C1–C4 alkylsulfonyl, C1–C4 alkylsulfonate, C3–C5 cycloalkylsulfonate, C1–C4 haloalkylsulfonate, C1–C4 haloalkylthio, C1–C4 haloalkylsulfinyl, C1–C4 haloalkylsulfonyl, C2–C7 alkylthioalkyl, C2–C7 alkylsulfinylalkyl, C2–C7 alkylsulfonylalkyl, C2–C7 haloalkylthioalkyl, C2–C7 haloalkylsulfinylalkyl, C2–C7 haloalkylsulfonylalkyl, C2–C5 cyanoalkyl, C4–C6 cyanocycloalkyl or C2–C5 alkoxycarbonyl; or two adjacent R2 may be taken together to form a saturated or unsaturated 5- to 8- membered ring, containing carbon atoms and optionally 1 to 3 oxygen, sulfur or nitrogen atoms as ring members, one or two carbon or sulfur ring members of the heterocycle can optionally be in the oxidized form of a carbonyl, sulfonyl,
sulfinyl moiety, said ring unsubstituted or substituted with at least one substituent independently selected from the group consisting of halogen, cyano, nitro, C1–C4 alkyl, C3–C6 cycloalkyl, C1–C4 haloalkyl, C1–C4 alkoxy and C1– C4 haloalkoxy; Y is O or S; Z is O, C(R3c)p, C(=O), C(=S), or NR3d; R3a is halogen, cyano, nitro, C1–C7 alkyl, C2–C6 alkenyl, C2–C6 alkynyl, C3–C7 cycloalkyl, C1–C7 alkoxy, C1–C7 haloalkoxy, C4–C7 cycloalkylalkyl, C4–C7 alkylcycloalkyl, C1–C7 haloalkyl, C2–C6 haloalkenyl, C2–C6 haloalkynyl, C3– C7 halocycloalkyl, C4–C7 haloalkylcycloalkyl, C4–C7 cycloalkylhaloalkyl, C2– C7 alkoxyalkyl, C2–C7 haloalkoxyalkyl, C2–C5 alkenyloxy, C2–C5 alkynyloxy, C2–C5 alkenyloxyalkyl, C2–C5 alkynyloxyalkyl, C3–C7 cycloalkoxy, C4–C7 cycloalkoxyalkyl, C2–C6 oxacycloalkyl, C3–C7 oxacycloalkylalkyl, C2–C6 oxacycloalkoxy, C3–C6 oxacycloalkoxyalkyl, C1–C5 alkylthio, C1–C4 alkylsulfinyl, C1–C4 alkylsulfonyl, C1–C4 haloalkylthio, C1–C4 haloalkylsulfinyl, C1–C4 haloalkylsulfonyl, C2–C7 alkoxyhaloalkyl, C2–C7 haloalkoxyhaloalkyl, C2–C7 alkylthioalkyl, C2–C7 alkylsulfinylalkyl, C2–C7 alkylsulfonylalkyl, C2–C7 haloalkylthioalkyl, C2–C7 haloalkylsulfinylalkyl, C2– C7 haloalkylsulfonylalkyl, C2–C5 cyanoalkyl, C4–C6 cyanocycloalkyl, C1–C4 nitroalkyl, C3–C6 alkylcarbonylalkyl or C3–C6 alkoxycarbonylalkyl; each R3b is independently H, halogen or C1–C3 alkyl; or R3a and R3b are taken together with the carbon atom to which they are attached to form a 3- to 7-membered ring containing ring members selected from carbon atoms and up to 3 heteroatoms independently selected from 1 oxygen atom, 1 sulfur atom, and up to 3 nitrogen atoms, wherein up to 2 carbon atom ring members are independently selected from C(=O) and C(=S) and the sulfur atom ring member is selected from S, S(O) or S(O)2; or R3a and R3b are taken together with the carbon atom to which they are attached to form C(=O), provided Z is NR3d and R3d is H; or two R3b are taken together with the carbon atom to which they are attached to form a 3- to 7-membered ring containing ring members selected from carbon atoms and up to 3 heteroatoms independently selected from 1 oxygen atom, 1 sulfur atom, and up to 3 nitrogen atoms, wherein up to 2 carbon atom ring members are independently selected from C(=O) and C(=S) and the sulfur atom ring member is selected from S, S(O) or S(O)2;
each R3c is independently H, halogen, cyano, nitro, hydroxy, C1–C5 alkyl, C2–C5 alkenyl, C2–C5 alkynyl, C3–C5 cycloalkyl, C1–C5 haloalkyl, C2–C5 haloalkenyl, C2–C5 haloalkynyl, C2–C5 alkoxyalkyl, C2–C5 haloalkoxyalkyl, C1–C5 alkoxy, C1–C5 haloalkoxy, C1–C5 alkylthio, C1–C4 alkylsulfinyl, C1–C4 alkylsulfonyl, C1–C5 haloalkylthio, C1–C4 haloalkylsulfinyl, C1–C4 haloalkylsulfonyl, C4–C7 cycloalkylalkyl, C4–C7 alkylcycloalkyl, C3–C7 halocycloalkyl, C4–C7 haloalkylcycloalkyl, C2–C7 alkylthioalkyl, C2–C7 alkylsulfinylalkyl, C2–C7 alkylsulfonylalkyl, C2–C7 haloalkylthioalkyl, C2–C7 haloalkylsulfinylalkyl, C2– C7 haloalkylsulfonylalkyl, C2–C5 cyanoalkyl, C4–C6 cyanocycloalkyl, C1–C4 nitroalkyl, C2–C6 oxacycloalkyl, C3–C6 oxacycloalkylalkyl, C3–C7 cycloalkoxy, C4–C7 cycloalkoxyalkyl, C3–C6 alkylcarbonylalkyl, C3–C6 alkoxycarbonylalkyl or C2–C5 alkoxycarbonyl, C2–C6 alkylcarbonyloxy, C2–C6 haloalkylcarbonyloxy, C4–C6 cycloalkylcarbonyloxy, C2–C6 alkoxycarbonyloxy, C2–C6 alkylaminocarbonyloxy, C3–C6 dialkylaminocarbonyloxy; or one R3c is taken together with the adjacent R3a or R3b and the carbon atom to which they are attached to form a 3- to 7-membered ring containing ring members selected from carbon atoms and up to 3 heteroatoms independently selected from 1 oxygen atom, 1 sulfur atom, and up to 3 nitrogen atoms, wherein up to 2 carbon atom ring members are independently selected from C(=O) and C(=S) and the sulfur atom ring member is selected from S, S(O) or S(O)2; or two R3c are taken together with the carbon atom to which they are attached to form a 3- to 7-membered ring containing ring members selected from carbon atoms and up to 3 heteroatoms independently selected from 1 oxygen atom, 1 sulfur atom, and up to 3 nitrogen atoms, wherein up to 2 carbon atom ring members are independently selected from C(=O) and C(=S) and the sulfur atom ring member is selected from S, S(O) or S(O)2; p is 0, 1 or 2; R3d is H, C1–C4 alkyl, C3–C6 cycloalkyl, C1–C4 haloalkyl, C2–C5 alkylcarbonyl, C2– C5 haloalkylcarbonyl, C2–C5 alkoxycarbonyl, C2–C5 alkylaminocarbonyl, C2– C5 dialkylaminocarbonyl, C1–C5 alkylsulfonyl or C1–C5 haloalkylsulfonyl; R4a is H, halogen, cyano, nitro, C1–C5 alkyl, C2–C5 alkenyl, C2–C5 alkynyl, C3–C5 cycloalkyl, C1–C5 haloalkyl, C2–C5 haloalkenyl, C2–C5 haloalkynyl, C2–C5 alkoxyalkyl, C2–C5 haloalkoxyalkyl, C1–C5 alkoxy, C1–C5 haloalkoxy, C3–C6 trialkylsilyl, C1–C5 alkylthio, C1–C5 haloalkylthio or C2–C5 alkoxycarbonyl; q is 0, 1 or 2; and
each R4b is independently H, halogen, cyano, nitro, C1–C4 alkyl, C1–C4 haloalkyl, C1–C4 alkoxy or C1–C4 alkylthio; provided that: (a) when Z is O, then at least one of X3 and X4 is N; and (b) the compound of Formula 1 is other than: (3,4-dihydro-3-methyl-1(2H)-quinolinyl)[3-(1-pyrrolidinyl)phenyl]methanone, (3,4-dihydro-3-methyl-1(2H)-quinolinyl)[3-(1H-pyrrol-1-yl)phenyl]methanone, 1-[3-[(3,4-dihydro-3-methyl-1(2H)-quinolinyl)carbonyl]phenyl]-2-pyrrolidinone, (3,4-dihydro-3-methoxy-1(2H)-quinolinyl)[5-(4-morpholinyl)-3-pyridinyl]- methanone, (3,4-dihydro-3-methyl-1(2H)-quinolinyl)[5-(1H-1,2,3-triazol-1-yl)-3-pyridinyl]- methanone, [3,4-dihydro-3-(1-methylethyl)-1(2H)-quinoxalinyl][6-(1-pyrrolidinyl)pyrazin-2- yl]methanone, (3,4-dihydro-3-methoxy-1(2H)-quinolinyl)[5-(1H-1,2,3-triazol-1-yl)-3-pyridinyl]- methanone, (3,4-dihydro-3-methyl-1(2H)-quinolinyl)[3-(tetrahydro-1,1-dioxido-2H-1,2-thiazin- 2-yl)phenyl]methanone, [3,4-dihydro-3-(1-methylethyl)-1(2H)-quinoxalinyl][3-(1H-1,2,4-triazol-1-yl)- phenyl]methanone, (3,4-dihydro-3-methyl-1(2H)-quinolinyl)[4-methoxy-3-(1H-tetrazol-1-yl)phenyl]- methanone, [3,4-dihydro-3-(1-methylethyl)-1(2H)-quinoxalinyl][4-(1H-pyrazol-1-yl)-2- pyridinyl]methanone, [3,4-dihydro-3-(1-methylethyl)-1(2H)-quinoxalinyl][6-(1H-imidazol-1-yl)-2- pyridinyl]methanone, 2-[3-[(3,4-dihydro-3-methyl-1(2H)-quinolinyl)carbonyl]phenyl]-3-isothiazolidinone 1,1-dioxide, 2-[3-[(3,4-dihydro-3-methyl-1(2H)-quinolinyl)carbonyl]phenyl]-4-methyl-3- isothiazolidinone 1,1-dioxide, ethyl 2-ethyl-3,4-dihydro-6,7-dimethyl-4-[3-(5-methyl-1,2,4-oxadiazol-3- yl)benzoyl]-1(2H)-quinoxalinecarboxylate, and ethyl 4-[3-(3,5-dimethyl-1H-pyrazol-1-yl)benzoyl]-2-ethyl-3,4-dihydro-6,7- dimethyl-1(2H)-quinoxalinecarboxylate.
3. The compound of Claim 2 wherein Z is C(R3c)p or NR3d.
4. The compound of Claim 3 wherein A is A-1, A-2, A-3, A-5, A-7, A-8 or A-10;
X1, X2, X3 and X4 are each independently CR2; or X1 is N, and X2, X3 and X4 are each independently N or CR2; or X1 and X2 are N, and X3 and X4 are each independently CR2; or X2 and X3 are N, and X1 and X4 are each independently CR2; or X1 and X3 are N, and X2 and X4 are each independently CR2; or X2 is N, and X1, X3 and X4 are CR2. R1a is H, C1–C7 alkyl, C2–C6 alkenyl, C2–C6 alkynyl, C3–C7 cycloalkyl, C4–C7 cycloalkylalkyl, C4–C7 alkylcycloalkyl, C1–C7 haloalkyl, C1–C6 hydroxyalkyl, C3–C7 hydroxycycloalkyl, C1–C6 hydroxyhaloalkyl, C4–C8 alkoxycycloalkyl, C2–C7 alkoxyhaloalkyl, C2–C6 oxacycloalkyl, C3–C7 oxacycloalkylalkyl, C3–C7 alkyloxacycloalkyl, C2–C6 thiacycloalkyl, C3– C7 thiacycloalkylalkyl, C2–C6 haloalkenyl, C2–C6 haloalkynyl, C3–C7 halocycloalkyl, C4–C7 haloalkylcycloalkyl, C2–C7 alkoxyalkyl, C2–C7 haloalkoxyalkyl, C1–C7 alkoxy, C2–C5 alkenyloxy, C2–C5 alkynyloxy, C3–C7 cycloalkoxy, C4–C7 cycloalkoxyalkyl, C1–C7 haloalkoxy, C1–C7 alkylthio, C2–C7 alkylthioalkyl, C1–C5 alkylsulfinyl, C1–C5 alkylsulfonyl, C1–C5 haloalkylthio, C2–C7 alkylsulfinylalkyl, C2–C7 alkylsulfonylalkyl, C2–C7 haloalkylthioalkyl, C4–C7 haloalkylthiocycloalkyl, C4–C7 haloalkylsulfonylcycloalkyl or C2–C5 cyanoalkyl; each R1b is independently H, C1–C4 alkyl, C3–C5 cycloalkyl, C1–C4 haloalkyl, C2–C4 alkoxyalkyl, C1–C4 alkoxy or C1–C4 alkylthio; R1c is H, C1–C4 alkyl, C3–C5 cycloalkyl, C2–C6 oxacycloalkyl or C1–C7 haloalkyl; each R2 is independently H, halogen, cyano, OH, C1–C5 alkyl, C2–C5 alkenyl, C2–C5 alkynyl, C2–C5 alkenyloxy, C2–C5 alkynyloxy, C3–C7 cycloalkoxy, C4–C7 cycloalkoxyalkyl, C3–C6 cycloalkyl, C4–C7 cycloalkylalkyl, C1–C5 haloalkyl, C2–C5 haloalkenyl, C2–C5 haloalkynyl, C2–C5 alkoxyalkyl, C2–C5 haloalkoxyalkyl, C1–C5 alkoxy, C1–C5 haloalkoxy; or two adjacent R2 may be taken together to form a 5- or 6- membered ring, containing up to 2 oxygen atoms as the ring members. R3a is halogen, C1–C7 alkyl, C2–C6 alkenyl, C2–C6 alkynyl, C3–C7 cycloalkyl, C1–C7 alkoxy, C4–C7 cycloalkylalkyl, C4–C7 alkylcycloalkyl, C1–C7 haloalkyl, C2–C6 haloalkenyl, C2–C6 haloalkynyl, C3–C7 halocycloalkyl, C4–C7 haloalkylcycloalkyl, C2–C7 alkoxyalkyl or C2–C7 haloalkoxyalkyl; each R3c is independently H, halogen, hydroxy, C1–C5 alkyl, C1–C5 alkoxy, C3– C6 alkylcarbonylalkyl, C3–C6 alkoxycarbonylalkyl or C2–C6 alkylcarbonyloxy;
R3d is H, C1–C4 alkyl, C2–C5 alkylcarbonyl, C2–C5 haloalkylcarbonyl, C2–C5 alkoxycarbonyl, C2–C5 alkylaminocarbonyl, C2–C5 dialkylaminocarbonyl, C1–C5 alkylsulfonyl or C1–C5 haloalkylsulfonyl; and R4a is H, halogen, C1–C5 alkyl, C1–C5 haloalkyl.
5. The compound of Claim 4 wherein A is A-1, A-2 or A-8; R1a is H, C1–C7 alkyl, C3–C7 cycloalkyl, C4–C7 cycloalkylalkyl, C4–C7 alkylcycloalkyl, C1–C7 haloalkyl, C1–C6 hydroxyalkyl, C4–C8 alkoxycycloalkyl, C2–C6 oxacycloalkyl, C3–C7 alkyloxacycloalkyl, C2–C7 alkoxyalkyl, C1–C7 alkoxy, C1–C7 alkylthio, C2–C7 alkylthioalkyl, C2–C7 alkylsulfinylalkyl or C2–C7 alkylsulfonylalkyl; each R1b is independently H, C1–C4 alkyl or C3–C4 cycloalkyl; R1c is H, C1–C4 alkyl or C3–C5 cycloalkyl; each R2 is independently H, halogen, cyano, OH, C1–C5 alkyl, C2–C5 alkenyl, C2–C5 alkynyl, C3–C6 cycloalkyl, C1–C5 haloalkyl, C1–C5 alkoxy or C1– C5 haloalkoxy; or two adjacent R2 may be taken together to form a 5- or 6- membered ring, containing up to 2 oxygen atoms as the ring members; R3a is halogen, C1–C7 alkyl, C3–C7 cycloalkyl, C1–C7 alkoxy, C1–C7 haloalkyl or C2–C7 alkoxyalkyl; R3c is independently H, F, Cl, hydroxy, Me, OMe or -OC(=O)Me; and R3d is H or C1–C4 alkyl.
6. The compound of Claim 5 wherein A is A-1; X1, X2, X3 and X4 are CR2; R1a is C1–C5 alkyl, C3–C5 cycloalkyl, C1–C5 haloalkyl, C1–C6 hydroxyalkyl, C2–C5 oxacycloalkyl or C2–C7 alkylsulfonylalkyl; each R2 is independently H, halogen, cyano, OH, C1–C5 alkyl, C2–C5 alkenyl, C2–C5 alkynyl, C3–C6 cycloalkyl, C1–C5 haloalkyl, C1–C5 alkoxy or C1– C5 haloalkoxy; or two adjacent R2 may be taken together to form a 5- or 6- membered ring and the said ring contains a group of -OCH2O- or - O(CH2)2O- or -O(CH2)2-; R3a is halogen, C1–C4 alkyl, C3–C6 cycloalkyl, C1–C3 alkoxy, C1–C4 haloalkyl or C2–C5 alkoxyalkyl; each R3b is independently H or halogen; and R3d is H or Me.
7. The compound of Claim 6 wherein R1a is Et, i-Pr, t-Bu, c-Bu, CFMe2, CF2Me, oxetan-3-yl, CH2SO2Me or CMe2OH; R1b is H; R1c is H, Me, c-Pr or i-Pr; each R2 is independently H, OH, OEt, propargyl, allyl, c-Pr, F, Cl, Br, CN, Me, Et, OMe, CF 3 , OCF 3 or CH 2 CF 3 ; Y is O; R3a is halogen, Me, c-Pr, CF3, CH2OMe, or CH2OEt; R3b is H; R3d is H or Me; and R4a is H, Cl or Me.
8. The compound of any one of the Claims 1 to 7 wherein Z is C(R3c)p.
9. The compound of Claim 2 wherein Z is O.
10. The compound of Claim 9 wherein X2 and X3 are N, and X1 and X4 are each independently CR2; or X1 and X3 are N, and X2 and X4 are each independently CR2.
11. The compound of Claim 10 wherein A is A-1, A-2, A-3, A-5, A-7, A-8 or A-10; R1a is H, C1–C7 alkyl, C2–C6 alkenyl, C2–C6 alkynyl, C3–C7 cycloalkyl, C4–C7 cycloalkylalkyl, C4–C7 alkylcycloalkyl, C1–C7 haloalkyl, C1–C6 hydroxyalkyl, C3–C7 hydroxycycloalkyl, C1–C6 hydroxyhaloalkyl, C4–C8 alkoxycycloalkyl, C2–C7 alkoxyhaloalkyl, C2–C6 oxacycloalkyl, C3–C7 oxacycloalkylalkyl, C3–C7 alkyloxacycloalkyl, C2–C6 thiacycloalkyl, C3– C7 thiacycloalkylalkyl, C2–C6 haloalkenyl, C2–C6 haloalkynyl, C3–C7 halocycloalkyl, C4–C7 haloalkylcycloalkyl, C2–C7 alkoxyalkyl, C2–C7 haloalkoxyalkyl, C1–C7 alkoxy, C2–C5 alkenyloxy, C2–C5 alkynyloxy, C3– C7 cycloalkoxy, C4–C7 cycloalkoxyalkyl, C1–C7 haloalkoxy, C1–C7 alkylthio, C2–C7 alkylthioalkyl, C1–C5 alkylsulfinyl, C1–C5 alkylsulfonyl, C1–C5 haloalkylthio, C2–C7 alkylsulfinylalkyl, C2–C7 alkylsulfonylalkyl, C2–C7 haloalkylthioalkyl, C4–C7 haloalkylthiocycloalkyl, C4–C7 haloalkylsulfonylcycloalkyl or C2–C5 cyanoalkyl;
each R1b is independently H, C1–C4 alkyl, C3–C5 cycloalkyl, C1–C4 haloalkyl, C2–C4 alkoxyalkyl, C1–C4 alkoxy or C1–C4 alkylthio; R1c is H, C1–C4 alkyl, C3–C5 cycloalkyl, C2–C6 oxacycloalkyl or C1–C7 haloalkyl; each R2 is independently H, halogen, cyano, OH, C1–C5 alkyl, C2–C5 alkenyl, C2–C5 alkynyl, C2–C5 alkenyloxy, C2–C5 alkynyloxy, C3–C7 cycloalkoxy, C4–C7 cycloalkoxyalkyl, C3–C6 cycloalkyl, C4–C7 cycloalkylalkyl, C1–C5 haloalkyl, C2–C5 haloalkenyl, C2–C5 haloalkynyl, C2–C5 alkoxyalkyl, C2– C5 haloalkoxyalkyl, C1–C5 alkoxy, C1–C5 haloalkoxy; or two adjacent R2 may be taken together to form a 5- or 6- membered ring, containing up to 2 oxygen atoms as the ring members; R3a is halogen, C1–C7 alkyl, C2–C6 alkenyl, C2–C6 alkynyl, C3–C7 cycloalkyl, C1–C7 alkoxy, C4–C7 cycloalkylalkyl, C4–C7 alkylcycloalkyl, C1–C7 haloalkyl, C2–C6 haloalkenyl, C2–C6 haloalkynyl, C3–C7 halocycloalkyl, C4–C7 haloalkylcycloalkyl, C2–C7 alkoxyalkyl or C2–C7 haloalkoxyalkyl; each R3c is independently H, halogen, hydroxy, C1–C5 alkyl, C1–C5 alkoxy, C3– C6 alkylcarbonylalkyl, C3–C6 alkoxycarbonylalkyl or C2–C6 alkylcarbonyloxy; R3d is H, C1–C4 alkyl, C2–C5 alkylcarbonyl, C2–C5 haloalkylcarbonyl, C2–C5 alkoxycarbonyl, C2–C5 alkylaminocarbonyl, C2–C5 dialkylaminocarbonyl, C1–C5 alkylsulfonyl or C1–C5 haloalkylsulfonyl; and R4a is H, halogen, C1–C5 alkyl, C1–C5 haloalkyl.
12. The compound of Claim 11 wherein A is A-1, A-2 or A-8; R1a is H, C1–C7 alkyl, C3–C7 cycloalkyl, C4–C7 cycloalkylalkyl, C4–C7 alkylcycloalkyl, C1–C7 haloalkyl, C1–C6 hydroxyalkyl, C4–C8 alkoxycycloalkyl, C2–C6 oxacycloalkyl, C3–C7 alkyloxacycloalkyl, C2–C7 alkoxyalkyl, C1–C7 alkoxy, C1–C7 alkylthio, C2–C7 alkylthioalkyl, C2–C7 alkylsulfinylalkyl or C2–C7 alkylsulfonylalkyl; each R1b is independently H, C1–C4 alkyl or C3–C4 cycloalkyl; R1c is H, C1–C4 alkyl or C3–C5 cycloalkyl; each R2 is independently H, halogen, cyano, OH, C1–C5 alkyl, C2–C5 alkenyl, C2–C5 alkynyl, C3–C6 cycloalkyl, C1–C5 haloalkyl, C1–C5 alkoxy or C1– C5 haloalkoxy; or two adjacent R2 may be taken together to form a 5- or 6- membered ring, containing up to 2 oxygen atoms as the ring members;
R3a is halogen, C1–C7 alkyl, C3–C7 cycloalkyl, C1–C7 alkoxy, C1–C7 haloalkyl or C2–C7 alkoxyalkyl; R3c is independently H, F, Cl, hydroxy, Me, OMe or -OC(=O)Me; and R3d is H or C1–C4 alkyl.
13. The compound of Claim 12 wherein A is A-1; X1 and X3 are N, and X2 and X4 are independently CR2; R1a is C1–C5 alkyl, C3–C5 cycloalkyl, C1–C5 haloalkyl, C1–C6 hydroxyalkyl, C2–C5 oxacycloalkyl or C2–C7 alkylsulfonylalkyl; each R2 is independently H, halogen, cyano, OH, C1–C5 alkyl, C2–C5 alkenyl, C2–C5 alkynyl, C3–C6 cycloalkyl, C1–C5 haloalkyl, C1–C5 alkoxy or C1– C5 haloalkoxy; or two adjacent R2 may be taken together to form a 5- or 6- membered ring and the said ring contains a group of -OCH2O- or - O(CH2)2O- or -O(CH2)2-; R3a is halogen, C1–C4 alkyl, C3–C6 cycloalkyl, C1–C3 alkoxy, C1–C4 haloalkyl or C2–C5 alkoxyalkyl; each R3b is independently H or halogen; and R3d is H or Me.
14. The compound of Claim 13 wherein R1a is Et, i-Pr, t-Bu, c-Bu, CFMe2, CF2Me, oxetan-3-yl, CH2SO2Me or CMe2OH; R1b is H; R1c is H, Me, c-Pr or i-Pr; each R2 is independently H, OH, OEt, propargyl, allyl, c-Pr, F, Cl, Br, CN, Me, Et, OMe, CF 3 , OCF 3 or CH 2 CF 3 ; Y is O; R3a is halogen, Me, c-Pr, CF3, CH2OMe, or CH2OEt; R3b is H; R3d is H or Me; and R4a is H or Me.
15. The compound of Claim 12 wherein A is A-1; X2 and X3 are N, and X1 and X4 are independently CR2; R1a is C1–C5 alkyl, C3–C5 cycloalkyl, C1–C5 haloalkyl, C1–C6 hydroxyalkyl, C2–C5 oxacycloalkyl or C2–C7 alkylsulfonylalkyl;
each R2 is independently H, halogen, cyano, OH, C1–C5 alkyl, C2–C5 alkenyl, C2–C5 alkynyl, C3–C6 cycloalkyl, C1–C5 haloalkyl, C1–C5 alkoxy or C1– C5 haloalkoxy; or two adjacent R2 may be taken together to form a 5- or 6- membered ring and the said ring contains a group of -OCH2O- or - O(CH2)2O- or -O(CH2)2-; R3a is halogen, C1–C4 alkyl, C3–C6 cycloalkyl, C1–C3 alkoxy, C1–C4 haloalkyl or C2–C5 alkoxyalkyl; each R3b is independently H or halogen; and R3d is H or Me.
16. The compound of Claim 15 wherein R1a is Et, i-Pr, t-Bu, c-Bu, CFMe2, CF2Me, oxetan-3-yl, CH2SO2Me or CMe2OH; R1b is H; R1c is H, Me, c-Pr or i-Pr; each R2 is independently H, OH, OEt, propargyl, allyl, c-Pr, F, Cl, Br, CN, Me, Et, OMe, CF 3 , OCF 3 or CH 2 CF 3 ; Y is O; R3a is halogen, Me, c-Pr, CF3, CH2OMe, or CH2OEt; R3b is H; R3d is H or Me; and R4a is H or Me.
17. The compound of Claim 1 selected from the group consisting of: (3,4-dihydro-3,5-dimethyl-1(2H)-quinolinyl)-[2-methoxy-5-[3-(1- methylethyl)-1H-1,2,4-triazol-1-yl]phenyl]methanone; [(3S)-3,4-dihydro-3,5-dimethyl-1(2H)-quinolinyl][2-methoxy-5-[3-(1- methylethyl)-1H-1,2,4-triazol-1-yl]phenyl]methanone; [(3R)-3,4-dihydro-3,5-dimethyl-1(2H)-quinolinyl][2-methoxy-5-[3-(1- methylethyl)-1H-1,2,4-triazol-1-yl]phenyl]methanone; [5-fluoro-3,4-dihydro-3-(trifluoromethyl)-1(2H)-quinolinyl][2- methoxy-5-[3-(1-methylethyl)-1H-1,2,4-triazol-1- yl]phenyl]methanone; [(3S)-5-fluoro-3,4-dihydro-3-(trifluoromethyl)-1(2H)- quinolinyl][2-methoxy-5-[3-(1-methylethyl)-1H-1,2,4-triazol- 1-yl]phenyl]methanone;
[(3R)-5-fluoro-3,4-dihydro-3-(trifluoromethyl)-1(2H)- quinolinyl][2-methoxy-5-[3-(1-methylethyl)-1H-1,2,4-triazol- 1-yl]phenyl]methanone; [5-fluoro-3,4-dihydro-3-(trifluoromethyl)-1(2H)-quinolinyl][2- methyl-5-[3-(1-methylethyl)-1H-1,2,4-triazol-1- yl]phenyl]methanone; [(3S)-5-fluoro-3,4-dihydro-3-(trifluoromethyl)-1(2H)- quinolinyl][2-methyl-5-[3-(1-methylethyl)-1H-1,2,4-triazol- 1-yl]phenyl]methanone; [(3R)-5-fluoro-3,4-dihydro-3-(trifluoromethyl)-1(2H)- quinolinyl][2-methyl-5-[3-(1-methylethyl)-1H-1,2,4-triazol- 1-yl]phenyl]methanone; [3,4-dihydro-5-methyl-3-(trifluoromethyl)-1(2H)-quinolinyl][2- methoxy-5-[3-(1-methylethyl)-1H-1,2,4-triazol-1- yl]phenyl]methanone; [(3S)-3,4-dihydro-5-methyl-3-(trifluoromethyl)-1(2H)- quinolinyl][2-methoxy-5-[3-(1-methylethyl)-1H-1,2,4-triazol- 1-yl]phenyl]methanone; [(3R)-3,4-dihydro-5-methyl-3-(trifluoromethyl)-1(2H)- quinolinyl][2-methoxy-5-[3-(1-methylethyl)-1H-1,2,4-triazol- 1-yl]phenyl]methanone; and [3,4-dihydro-3-methyl-5-(trifluoromethyl)-1(2H)-quinoxalinyl][2- methoxy-5-[3-(1-methylethyl)-1H-1,2,4-triazol-1- yl]phenyl]methanone; and combinations thereof.
18. A herbicidal composition comprising a compound of Claim 1 and at least one component selected from the group consisting of surfactants, solid diluents and liquid diluents.
19. A herbicidal composition comprising a compound of Claim 1, at least one additional active ingredient selected from the group consisting of other herbicides and herbicide safeners, and at least one component selected from the group consisting of surfactants, solid diluents and liquid diluents.
20. A herbicidal mixture comprising (a) a compound of any one of Claims 1 through 17, and (b) at least one additional active ingredient selected from photosystem II inhibitors (b1), AHAS inhibitors (b2), ACCase inhibitors (b3), auxin mimics (b4), EPSP synthase
inhibitors (b5), photosystem I electron diverters (b6), PPO inhibitors (b7), GS inhibitors (b8), VLCFA elongase inhibitors (b9), auxin transport inhibitors (b10), PDS inhibitors (b11), HPPD inhibitors (b12), DXP synthase inhibitors (b13), HST inhibitors (b14), cellulose biosynthesis inhibitors (b15), DHODH inhibitors (b16), other herbicides including mitotic disruptors, organic arsenicals, asulam, bromobutide, cinflubrolin, cinmethylin, cumyluron, dazomet, difenzoquat, dymron, etobenzanid, flurenol, fosamine, fosamine-ammonium, hydantocidin, metam, methyldymron, oleic acid, oxaziclomefone, pelargonic acid and pyributicarb (b17), and herbicide safeners (b18); and salts of compounds of (b1) through (b18).
21. A method for controlling the growth of undesired vegetation comprising contacting the vegetation or its environment with a herbicidally effective amount of a compound of Claim 1.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US202363601264P | 2023-11-21 | 2023-11-21 | |
| US63/601,264 | 2023-11-21 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2025111184A1 true WO2025111184A1 (en) | 2025-05-30 |
Family
ID=93799611
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2024/055871 Pending WO2025111184A1 (en) | 2023-11-21 | 2024-11-14 | Substituted tetrahydroquinoline and tetrahydroquinoxaline herbicides |
Country Status (2)
| Country | Link |
|---|---|
| AR (1) | AR134418A1 (en) |
| WO (1) | WO2025111184A1 (en) |
Citations (68)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US2891855A (en) | 1954-08-16 | 1959-06-23 | Geigy Ag J R | Compositions and methods for influencing the growth of plants |
| US3060084A (en) | 1961-06-09 | 1962-10-23 | Du Pont | Improved homogeneous, readily dispersed, pesticidal concentrate |
| US3235361A (en) | 1962-10-29 | 1966-02-15 | Du Pont | Method for the control of undesirable vegetation |
| US3299566A (en) | 1964-06-01 | 1967-01-24 | Olin Mathieson | Water soluble film containing agricultural chemicals |
| US3309192A (en) | 1964-12-02 | 1967-03-14 | Du Pont | Method of controlling seedling weed grasses |
| US3920442A (en) | 1972-09-18 | 1975-11-18 | Du Pont | Water-dispersible pesticide aggregates |
| US4144050A (en) | 1969-02-05 | 1979-03-13 | Hoechst Aktiengesellschaft | Micro granules for pesticides and process for their manufacture |
| US4172714A (en) | 1976-12-20 | 1979-10-30 | E. I. Du Pont De Nemours And Company | Dry compactible, swellable herbicidal compositions and pellets produced therefrom |
| GB2095558A (en) | 1981-03-30 | 1982-10-06 | Avon Packers Ltd | Formulation of agricultural chemicals |
| DE3246493A1 (en) | 1982-12-16 | 1984-06-20 | Bayer Ag, 5090 Leverkusen | METHOD FOR PRODUCING WATER-DISPERSIBLE GRANULES |
| WO1991013546A1 (en) | 1990-03-12 | 1991-09-19 | E.I. Du Pont De Nemours And Company | Water-dispersible or water-soluble pesticide granules from heat-activated binders |
| US5180587A (en) | 1988-06-28 | 1993-01-19 | E. I. Du Pont De Nemours And Company | Tablet formulations of pesticides |
| US5208030A (en) | 1989-08-30 | 1993-05-04 | Imperial Chemical Industries Plc | Active ingredient dosage device |
| US5232701A (en) | 1990-10-11 | 1993-08-03 | Sumitomo Chemical Company, Limited | Boron carbonate and solid acid pesticidal composition |
| EP0733625A1 (en) * | 1995-03-23 | 1996-09-25 | Nihon Bayer Agrochem K.K. | 1-Substituted-tetrazolinones and their use as herbicides |
| WO2000010994A1 (en) | 1998-08-19 | 2000-03-02 | Sankyo Company, Limited | Process for the preparation of 2-(aryloxymethyl)-2,5,7,8-tetramethylchroman-6-ols |
| WO2001090088A1 (en) | 2000-05-23 | 2001-11-29 | Syngenta Participations Ag | Method for making acylamides by synthesizing and acylating benzoxazines |
| WO2002070726A1 (en) | 2001-03-07 | 2002-09-12 | Daiichi Pharmaceutical Co., Ltd. | Process for preparation of optically active propoxyaniline derivatives |
| WO2002074764A2 (en) | 2001-03-15 | 2002-09-26 | Smithkline Beecham P.L.C. | Piperazine derivatives and their use as 5-ht1b ligands |
| WO2003024222A1 (en) | 2001-09-21 | 2003-03-27 | E. I. Du Pont De Nemours And Company | Anthranilamide arthropodicide treatment |
| WO2003077918A1 (en) | 2002-03-12 | 2003-09-25 | Merck & Co., Inc. | Di-aryl substituted tetrazole modulators of metabotropic glutamate receptor-5 |
| WO2004080973A1 (en) | 2003-03-07 | 2004-09-23 | Abbott Laboratories | Fused tri and tetra-cyclic pyrazole kinase inhibitors |
| WO2004085401A1 (en) * | 2003-03-28 | 2004-10-07 | Pfizer Products Inc. | 1,2,4-substituted 1,2,3,4-tetrahydro-and 1,2 dihydro-quinoline and 1,2,3,4-tetrahydro-quinoxaline derivatives as cetp inhibitors for the treatment of atherosclerosis and obesity |
| US20070185097A1 (en) | 2006-02-06 | 2007-08-09 | Bristol-Myers Squibb Company | Melanin concentrating hormone receptor-1 antagonists |
| WO2008146871A1 (en) | 2007-05-29 | 2008-12-04 | Santen Pharmaceutical Co., Ltd. | Novel 1,2,3,4-tetrahydroquinoxaline derivative which has, as substituent, phenyl group having sulfonic acid ester structure or sulfonic acid amide structure introduced therein and has glucocorticoid receptor-binding activity |
| WO2009009501A2 (en) | 2007-07-06 | 2009-01-15 | Bristol-Myers Squibb Company | Non-basic melanin concentrating hormone receptor-1 antagonists and methods |
| WO2010047956A1 (en) | 2008-10-08 | 2010-04-29 | Bristol-Myers Squibb Company | Azolopyrrolone melanin concentrating hormone receptor-1 antagonists |
| WO2010049302A1 (en) | 2008-10-29 | 2010-05-06 | F. Hoffmann-La Roche Ag | Novel phenyl amide or pyridil amide derivatives and their use as gpbar1 agonists |
| WO2011025690A1 (en) | 2009-08-27 | 2011-03-03 | Merck Sharp & Dohme Corp. | Novel pyrrolidine derived beta 3 adrenergic receptor agonists |
| WO2011162267A1 (en) | 2010-06-22 | 2011-12-29 | 株式会社 三和化学研究所 | Novel thiophenecarboxamide derivatives and use thereof as medicine |
| US20120065197A1 (en) * | 2010-05-27 | 2012-03-15 | Bayer Cropscience Ag | Pyridinylcarboxylic Acid Derivatives as Fungicides |
| WO2012041789A1 (en) * | 2010-10-01 | 2012-04-05 | Basf Se | Herbicidal benzoxazinones |
| WO2012054510A1 (en) | 2010-10-19 | 2012-04-26 | Comentis, Inc. | Oxadiazole compounds which inhibit beta-secretase activity and methods of use thereof |
| WO2012063207A1 (en) | 2010-11-10 | 2012-05-18 | Actelion Pharmaceuticals Ltd | Lactam derivatives useful as orexin receptor antagonists |
| WO2012137982A2 (en) | 2011-04-05 | 2012-10-11 | Takeda Pharmaceutical Company Limited | Sulfonamide derivative and use thereof |
| WO2013130660A1 (en) | 2012-02-28 | 2013-09-06 | Amgen Inc. | Amides as pim inhibitors |
| WO2014171527A1 (en) | 2013-04-19 | 2014-10-23 | 学校法人順天堂 | Novel compound with antibacterial activity |
| WO2015017610A1 (en) | 2013-07-31 | 2015-02-05 | Gilead Sciences, Inc. | Syk inhibitors |
| WO2015057205A1 (en) | 2013-10-15 | 2015-04-23 | Janssen Pharmaceutica Nv | Methylene linked quinolinyl modulators of ror-gamma-t |
| US20150148342A1 (en) | 2013-11-26 | 2015-05-28 | Incyte Corporation | Bicyclic heterocycles as bet protein inhibitors |
| WO2015086506A1 (en) | 2013-12-09 | 2015-06-18 | Ucb Biopharma Sprl | Fused imidazole and pyrazole derivatives as modulators of tnf activity |
| WO2015095792A1 (en) | 2013-12-20 | 2015-06-25 | Merck Sharp & Dohme Corp. | Carbamate benzoxaxine propionic acids and acid derivatives for modulation of rorgamma activity and the treatment of disease |
| WO2015095795A1 (en) | 2013-12-20 | 2015-06-25 | Merck Sharp & Dohme Corp. | TETRAHYDRONAPHTHYRIDINE, BENZOXAZINE, AZA-BENZOXAZINE, AND RELATED BICYCLIC COMPOUNDS FOR INHIBITION OF RORgamma ACTIVITY AND THE TREATMENT OF DISEASE |
| WO2015124868A1 (en) | 2014-02-21 | 2015-08-27 | Inventiva | New phenylazetidine, carboxylate or carboxamide compounds |
| WO2015136091A1 (en) | 2014-03-14 | 2015-09-17 | AbbVie Deutschland GmbH & Co. KG | Hexahydrodiazepinoquinolines carrying a substituted alkyl radical |
| WO2015160636A1 (en) | 2014-04-16 | 2015-10-22 | Merck Sharp & Dohme Corp. | Factor ixa inhibitors |
| WO2016001631A1 (en) | 2014-06-30 | 2016-01-07 | Astrazeneca Ab | Benzoxazinone amides as mineralocorticoid receptor modulators |
| WO2016040223A1 (en) | 2014-09-09 | 2016-03-17 | Bristol-Myers Squibb Company | Cyclobutane containing carboxylic acid gpr120 modulators |
| WO2016081692A2 (en) | 2014-11-19 | 2016-05-26 | Glaxosmithkline Llc | Substituted bridged urea analogs as sirtuin modulators |
| WO2016086200A1 (en) | 2014-11-27 | 2016-06-02 | Genentech, Inc. | 4,5,6,7-tetrahydro-1 h-pyrazolo[4,3-c]pyridin-3-amine compounds as cbp and/or ep300 inhibitors |
| WO2017108723A2 (en) | 2015-12-22 | 2017-06-29 | F. Hoffmann-La Roche Ag | PYRAZOLO[1,5a]PYRIMIDINE DERIVATIVES AS IRAK4 MODULATORS |
| WO2017172505A1 (en) | 2016-03-29 | 2017-10-05 | Merck Sharp & Dohme Corp. | Antidiabetic bicyclic compounds |
| WO2017205536A2 (en) | 2016-05-24 | 2017-11-30 | Genentech, Inc. | Therapeutic compounds and uses thereof |
| WO2017205709A1 (en) | 2016-05-27 | 2017-11-30 | Bristol-Myers Squibb Company | Triazolones and tetrazolones as inhibitors of rock |
| WO2018013770A1 (en) | 2016-07-14 | 2018-01-18 | Bristol-Myers Squibb Company | Bicyclic heteroaryl substituted compounds |
| WO2019162323A1 (en) | 2018-02-21 | 2019-08-29 | Boehringer Ingelheim International Gmbh | New benzimidazole compounds and derivatives as egfr inhibitors |
| WO2019191667A1 (en) | 2018-03-29 | 2019-10-03 | Board Of Regents, The University Of Texas System | Imidazopiperazine inhibitors of transcription activating proteins |
| WO2019195810A2 (en) | 2018-04-06 | 2019-10-10 | Praxis Biotech LLC | Atf6 inhibitors and uses thereof |
| WO2019210837A1 (en) | 2018-05-04 | 2019-11-07 | Dongguan HEC Pesticides R&D Co., Ltd. | Quinoline derivatives and preparation methods and uses thereof |
| WO2020182990A1 (en) | 2019-03-14 | 2020-09-17 | Janssen Sciences Ireland Unlimited Company | Fused ring pyrimidone derivatives for use in the treatment of hbv infection or of hbv-induced diseases |
| WO2020207941A1 (en) | 2019-04-09 | 2020-10-15 | F. Hoffmann-La Roche Ag | Heterocyclic compounds as inhibitors of monoacylglycerol lipase (magl) |
| WO2021050964A1 (en) | 2019-09-13 | 2021-03-18 | Nimbus Saturn, Inc. | Hpk1 antagonists and uses thereof |
| WO2021092525A1 (en) | 2019-11-08 | 2021-05-14 | Vanderbilt University | Wdr5 inhibitors and modulators |
| WO2021242677A1 (en) | 2020-05-28 | 2021-12-02 | Eli Lilly And Company | Trka inhibitor |
| WO2022035799A1 (en) | 2020-08-10 | 2022-02-17 | Prelude Therapeutics Incorporated | Heterocycle cdk inhibitors and their use thereof |
| WO2022053422A1 (en) | 2020-09-11 | 2022-03-17 | Syngenta Crop Protection Ag | Herbicidal compounds |
| WO2022204336A1 (en) | 2021-03-26 | 2022-09-29 | Novartis Ag | Novel cyclopental[c]pyrrol negative allosteric modulators of nr2b |
| WO2024015425A1 (en) * | 2022-07-14 | 2024-01-18 | Fmc Corporation | Herbicidal benzoxazines |
-
2024
- 2024-11-14 WO PCT/US2024/055871 patent/WO2025111184A1/en active Pending
- 2024-11-20 AR ARP240103186A patent/AR134418A1/en unknown
Patent Citations (68)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US2891855A (en) | 1954-08-16 | 1959-06-23 | Geigy Ag J R | Compositions and methods for influencing the growth of plants |
| US3060084A (en) | 1961-06-09 | 1962-10-23 | Du Pont | Improved homogeneous, readily dispersed, pesticidal concentrate |
| US3235361A (en) | 1962-10-29 | 1966-02-15 | Du Pont | Method for the control of undesirable vegetation |
| US3299566A (en) | 1964-06-01 | 1967-01-24 | Olin Mathieson | Water soluble film containing agricultural chemicals |
| US3309192A (en) | 1964-12-02 | 1967-03-14 | Du Pont | Method of controlling seedling weed grasses |
| US4144050A (en) | 1969-02-05 | 1979-03-13 | Hoechst Aktiengesellschaft | Micro granules for pesticides and process for their manufacture |
| US3920442A (en) | 1972-09-18 | 1975-11-18 | Du Pont | Water-dispersible pesticide aggregates |
| US4172714A (en) | 1976-12-20 | 1979-10-30 | E. I. Du Pont De Nemours And Company | Dry compactible, swellable herbicidal compositions and pellets produced therefrom |
| GB2095558A (en) | 1981-03-30 | 1982-10-06 | Avon Packers Ltd | Formulation of agricultural chemicals |
| DE3246493A1 (en) | 1982-12-16 | 1984-06-20 | Bayer Ag, 5090 Leverkusen | METHOD FOR PRODUCING WATER-DISPERSIBLE GRANULES |
| US5180587A (en) | 1988-06-28 | 1993-01-19 | E. I. Du Pont De Nemours And Company | Tablet formulations of pesticides |
| US5208030A (en) | 1989-08-30 | 1993-05-04 | Imperial Chemical Industries Plc | Active ingredient dosage device |
| WO1991013546A1 (en) | 1990-03-12 | 1991-09-19 | E.I. Du Pont De Nemours And Company | Water-dispersible or water-soluble pesticide granules from heat-activated binders |
| US5232701A (en) | 1990-10-11 | 1993-08-03 | Sumitomo Chemical Company, Limited | Boron carbonate and solid acid pesticidal composition |
| EP0733625A1 (en) * | 1995-03-23 | 1996-09-25 | Nihon Bayer Agrochem K.K. | 1-Substituted-tetrazolinones and their use as herbicides |
| WO2000010994A1 (en) | 1998-08-19 | 2000-03-02 | Sankyo Company, Limited | Process for the preparation of 2-(aryloxymethyl)-2,5,7,8-tetramethylchroman-6-ols |
| WO2001090088A1 (en) | 2000-05-23 | 2001-11-29 | Syngenta Participations Ag | Method for making acylamides by synthesizing and acylating benzoxazines |
| WO2002070726A1 (en) | 2001-03-07 | 2002-09-12 | Daiichi Pharmaceutical Co., Ltd. | Process for preparation of optically active propoxyaniline derivatives |
| WO2002074764A2 (en) | 2001-03-15 | 2002-09-26 | Smithkline Beecham P.L.C. | Piperazine derivatives and their use as 5-ht1b ligands |
| WO2003024222A1 (en) | 2001-09-21 | 2003-03-27 | E. I. Du Pont De Nemours And Company | Anthranilamide arthropodicide treatment |
| WO2003077918A1 (en) | 2002-03-12 | 2003-09-25 | Merck & Co., Inc. | Di-aryl substituted tetrazole modulators of metabotropic glutamate receptor-5 |
| WO2004080973A1 (en) | 2003-03-07 | 2004-09-23 | Abbott Laboratories | Fused tri and tetra-cyclic pyrazole kinase inhibitors |
| WO2004085401A1 (en) * | 2003-03-28 | 2004-10-07 | Pfizer Products Inc. | 1,2,4-substituted 1,2,3,4-tetrahydro-and 1,2 dihydro-quinoline and 1,2,3,4-tetrahydro-quinoxaline derivatives as cetp inhibitors for the treatment of atherosclerosis and obesity |
| US20070185097A1 (en) | 2006-02-06 | 2007-08-09 | Bristol-Myers Squibb Company | Melanin concentrating hormone receptor-1 antagonists |
| WO2008146871A1 (en) | 2007-05-29 | 2008-12-04 | Santen Pharmaceutical Co., Ltd. | Novel 1,2,3,4-tetrahydroquinoxaline derivative which has, as substituent, phenyl group having sulfonic acid ester structure or sulfonic acid amide structure introduced therein and has glucocorticoid receptor-binding activity |
| WO2009009501A2 (en) | 2007-07-06 | 2009-01-15 | Bristol-Myers Squibb Company | Non-basic melanin concentrating hormone receptor-1 antagonists and methods |
| WO2010047956A1 (en) | 2008-10-08 | 2010-04-29 | Bristol-Myers Squibb Company | Azolopyrrolone melanin concentrating hormone receptor-1 antagonists |
| WO2010049302A1 (en) | 2008-10-29 | 2010-05-06 | F. Hoffmann-La Roche Ag | Novel phenyl amide or pyridil amide derivatives and their use as gpbar1 agonists |
| WO2011025690A1 (en) | 2009-08-27 | 2011-03-03 | Merck Sharp & Dohme Corp. | Novel pyrrolidine derived beta 3 adrenergic receptor agonists |
| US20120065197A1 (en) * | 2010-05-27 | 2012-03-15 | Bayer Cropscience Ag | Pyridinylcarboxylic Acid Derivatives as Fungicides |
| WO2011162267A1 (en) | 2010-06-22 | 2011-12-29 | 株式会社 三和化学研究所 | Novel thiophenecarboxamide derivatives and use thereof as medicine |
| WO2012041789A1 (en) * | 2010-10-01 | 2012-04-05 | Basf Se | Herbicidal benzoxazinones |
| WO2012054510A1 (en) | 2010-10-19 | 2012-04-26 | Comentis, Inc. | Oxadiazole compounds which inhibit beta-secretase activity and methods of use thereof |
| WO2012063207A1 (en) | 2010-11-10 | 2012-05-18 | Actelion Pharmaceuticals Ltd | Lactam derivatives useful as orexin receptor antagonists |
| WO2012137982A2 (en) | 2011-04-05 | 2012-10-11 | Takeda Pharmaceutical Company Limited | Sulfonamide derivative and use thereof |
| WO2013130660A1 (en) | 2012-02-28 | 2013-09-06 | Amgen Inc. | Amides as pim inhibitors |
| WO2014171527A1 (en) | 2013-04-19 | 2014-10-23 | 学校法人順天堂 | Novel compound with antibacterial activity |
| WO2015017610A1 (en) | 2013-07-31 | 2015-02-05 | Gilead Sciences, Inc. | Syk inhibitors |
| WO2015057205A1 (en) | 2013-10-15 | 2015-04-23 | Janssen Pharmaceutica Nv | Methylene linked quinolinyl modulators of ror-gamma-t |
| US20150148342A1 (en) | 2013-11-26 | 2015-05-28 | Incyte Corporation | Bicyclic heterocycles as bet protein inhibitors |
| WO2015086506A1 (en) | 2013-12-09 | 2015-06-18 | Ucb Biopharma Sprl | Fused imidazole and pyrazole derivatives as modulators of tnf activity |
| WO2015095792A1 (en) | 2013-12-20 | 2015-06-25 | Merck Sharp & Dohme Corp. | Carbamate benzoxaxine propionic acids and acid derivatives for modulation of rorgamma activity and the treatment of disease |
| WO2015095795A1 (en) | 2013-12-20 | 2015-06-25 | Merck Sharp & Dohme Corp. | TETRAHYDRONAPHTHYRIDINE, BENZOXAZINE, AZA-BENZOXAZINE, AND RELATED BICYCLIC COMPOUNDS FOR INHIBITION OF RORgamma ACTIVITY AND THE TREATMENT OF DISEASE |
| WO2015124868A1 (en) | 2014-02-21 | 2015-08-27 | Inventiva | New phenylazetidine, carboxylate or carboxamide compounds |
| WO2015136091A1 (en) | 2014-03-14 | 2015-09-17 | AbbVie Deutschland GmbH & Co. KG | Hexahydrodiazepinoquinolines carrying a substituted alkyl radical |
| WO2015160636A1 (en) | 2014-04-16 | 2015-10-22 | Merck Sharp & Dohme Corp. | Factor ixa inhibitors |
| WO2016001631A1 (en) | 2014-06-30 | 2016-01-07 | Astrazeneca Ab | Benzoxazinone amides as mineralocorticoid receptor modulators |
| WO2016040223A1 (en) | 2014-09-09 | 2016-03-17 | Bristol-Myers Squibb Company | Cyclobutane containing carboxylic acid gpr120 modulators |
| WO2016081692A2 (en) | 2014-11-19 | 2016-05-26 | Glaxosmithkline Llc | Substituted bridged urea analogs as sirtuin modulators |
| WO2016086200A1 (en) | 2014-11-27 | 2016-06-02 | Genentech, Inc. | 4,5,6,7-tetrahydro-1 h-pyrazolo[4,3-c]pyridin-3-amine compounds as cbp and/or ep300 inhibitors |
| WO2017108723A2 (en) | 2015-12-22 | 2017-06-29 | F. Hoffmann-La Roche Ag | PYRAZOLO[1,5a]PYRIMIDINE DERIVATIVES AS IRAK4 MODULATORS |
| WO2017172505A1 (en) | 2016-03-29 | 2017-10-05 | Merck Sharp & Dohme Corp. | Antidiabetic bicyclic compounds |
| WO2017205536A2 (en) | 2016-05-24 | 2017-11-30 | Genentech, Inc. | Therapeutic compounds and uses thereof |
| WO2017205709A1 (en) | 2016-05-27 | 2017-11-30 | Bristol-Myers Squibb Company | Triazolones and tetrazolones as inhibitors of rock |
| WO2018013770A1 (en) | 2016-07-14 | 2018-01-18 | Bristol-Myers Squibb Company | Bicyclic heteroaryl substituted compounds |
| WO2019162323A1 (en) | 2018-02-21 | 2019-08-29 | Boehringer Ingelheim International Gmbh | New benzimidazole compounds and derivatives as egfr inhibitors |
| WO2019191667A1 (en) | 2018-03-29 | 2019-10-03 | Board Of Regents, The University Of Texas System | Imidazopiperazine inhibitors of transcription activating proteins |
| WO2019195810A2 (en) | 2018-04-06 | 2019-10-10 | Praxis Biotech LLC | Atf6 inhibitors and uses thereof |
| WO2019210837A1 (en) | 2018-05-04 | 2019-11-07 | Dongguan HEC Pesticides R&D Co., Ltd. | Quinoline derivatives and preparation methods and uses thereof |
| WO2020182990A1 (en) | 2019-03-14 | 2020-09-17 | Janssen Sciences Ireland Unlimited Company | Fused ring pyrimidone derivatives for use in the treatment of hbv infection or of hbv-induced diseases |
| WO2020207941A1 (en) | 2019-04-09 | 2020-10-15 | F. Hoffmann-La Roche Ag | Heterocyclic compounds as inhibitors of monoacylglycerol lipase (magl) |
| WO2021050964A1 (en) | 2019-09-13 | 2021-03-18 | Nimbus Saturn, Inc. | Hpk1 antagonists and uses thereof |
| WO2021092525A1 (en) | 2019-11-08 | 2021-05-14 | Vanderbilt University | Wdr5 inhibitors and modulators |
| WO2021242677A1 (en) | 2020-05-28 | 2021-12-02 | Eli Lilly And Company | Trka inhibitor |
| WO2022035799A1 (en) | 2020-08-10 | 2022-02-17 | Prelude Therapeutics Incorporated | Heterocycle cdk inhibitors and their use thereof |
| WO2022053422A1 (en) | 2020-09-11 | 2022-03-17 | Syngenta Crop Protection Ag | Herbicidal compounds |
| WO2022204336A1 (en) | 2021-03-26 | 2022-09-29 | Novartis Ag | Novel cyclopental[c]pyrrol negative allosteric modulators of nr2b |
| WO2024015425A1 (en) * | 2022-07-14 | 2024-01-18 | Fmc Corporation | Herbicidal benzoxazines |
Non-Patent Citations (69)
| Title |
|---|
| "Comprehensive Heterocyclic Chemistry II", 1996, PERGAMON PRESS |
| "Comprehensive Heterocyclic Chemistry IV", vol. 4-7, 2022, ELSEVIER |
| "Developments in formulation technology", 2000, PJB PUBLICATIONS |
| "McCutcheon's Emulsifiers and Detergents", MCCUTCHEON'S DIVISION, THE MANUFACTURING CONFECTIONER PUBLISHING CO., article "annual American and International Editions" |
| "McCutcheon's Volume 2: Functional Materials", MCCUTCHEON'S DIVISION, THE MANUFACTURING CONFECTIONER PUBLISHING CO., article "annual International and North American" |
| "Perry's Chemical Engineer's Handbook", 1963, MCGRAW-HILL, pages: 8 - 57 |
| "Polymorphism in the Pharmaceutical Industry", 2006, WILEY-VCH |
| "The BioPesticide Manual", 2001, BRITISH CROP PROTECTION COUNCIL |
| "The Pesticide Manual", 2003, BRITISH CROP PROTECTION COUNCIL |
| A. S. DAVIDSONB. MILWIDSKY: "Synthetic Detergents", 1987, JOHN WILEY AND SONS |
| ACS MED. CHEM. LETT., vol. 11, 2020, pages 597 - 604 |
| ACS OMEGA, vol. 2, 2017, pages 1875 |
| ANGEW. CHEM. INT. ED., vol. 53, 2014, pages 6126 - 6130 |
| ANGEW. CHEM. INT. ED., vol. 56, 2017, pages 16136 - 16179 |
| ANGEW. CHEM. INT. ED., vol. 62, 2023, pages e202217064 |
| BEILSTEIN J. ORG. CHEM., vol. 7, 2011, pages 59 - 74 |
| BIOORG. MED. CHEM. LETT., vol. 11, 2001, pages 1989 - 1992 |
| BIOORG. MED. CHEM., vol. 15, 2007, pages 5912 - 5949 |
| BIOORG. MED. CHEM., vol. 23, 2013, pages 4501 - 4505 |
| BIOORG. MED. CHEM., vol. 78, 2023, pages 117152 |
| BROWNING: "Agglomeration", CHEMICAL ENGINEERING, 4 December 1967 (1967-12-04), pages 147 - 48 |
| CHEM. COMMUN., 1999, pages 2095 - 2096 |
| CHEM. REV., vol. 108, 2008, pages 3054 - 3131 |
| CHEM. REV., vol. 111, 2011, pages 1417 - 1492 |
| CHEM. REV., vol. 116, 2016, pages 12564 - 12649 |
| CHEM. SCI., vol. 1, 2010, pages 13 - 31 |
| CHEM. SOC. REV., vol. 42, 2013, pages 5270 |
| ERNEST L. ELIELSAMUEL H. WILEN: "Stereochemistry of Organic Compounds", 1994, JOHN WILEY & SONS |
| EUR. J. MED. CHEM., vol. 188, 2020, pages 111987 |
| EUR. J. ORG. CHEM., 2004, pages 3597 - 3600 |
| G. W. H. CHEESEMANE. S. G. WERSTIUK: "Comprehensive Heterocyclic Chemistry", vol. 22, 1984, PERGAMON PRESS, pages: 390 - 392 |
| GREENE, T. W.WUTS, P. G. M.: "Protective Groups in Organic Synthesis", 1991, WILEY |
| HANCE ET AL.: "Weed Control Handbook", 1989, BLACKWELL SCIENTIFIC PUBLICATIONS |
| IND. ENG. CHEM. RES., vol. 42, 2003, pages 680 - 686 |
| J. AGRIC. FOOD CHEM., vol. 43, 1995, pages 1929 - 1934 |
| J. AM. CHEM. SOC., vol. 11-13, 2001, pages 12202 - 12206 |
| J. AM. CHEM. SOC., vol. 122, 2000, pages 12907 - 12908 |
| J. HET. CHEM., vol. 47, 2010, pages 1406 - 1410 |
| J. MED. CHEM., vol. 31, 1988, pages 1548 - 1558 |
| J. MED. CHEM., vol. 32, 1989, pages 1927 |
| J. MED. CHEM., vol. 46, 2003, pages 4895 - 4903 |
| J. MED. CHEM., vol. 55, 2012, pages 10475 - 10489 |
| J. MED. CHEM., vol. 57, 2014, pages 10013 - 10030 |
| J. MED. CHEM., vol. 61, 2018, pages 4317 |
| J. MED. CHEM., vol. 66, 2023, pages 1583 - 1600 |
| J. MOL. CATAL. A CHEM., vol. 288, 2008, pages 28 - 32 |
| J. NAT. PROD., vol. 83, 2020, pages 3181 |
| J. ORG. CHEM., vol. 12-13, 2002, pages 6097 - 6103 |
| J. ORG. CHEM., vol. 60, 1995, pages 7508 - 7510 |
| J. ORG. CHEM., vol. 69, 2004, pages 5578 - 5587 |
| J. ORG. CHEM., vol. 80, 2015, pages 3815 - 3824 |
| J. ORG. CHEM., vol. 86, 2021, pages 16573 - 16581 |
| KLINGMAN: "Weed Control as a Science", 1961, JOHN WILEY AND SONS, INC., pages: 81 - 96 |
| LAROCK, R. C.: "Proceedings of the 9th International Congress on Pesticide Chemistry", 1999, THE ROYAL SOCIETY OF CHEMISTRY, pages: 120 - 133 |
| MOLANDER ET AL., ACC. CHEM. RES., vol. 40, 2007, pages 275 - 286 |
| ORG. BIOMOL. CHEM., vol. 8, 2010, pages 2823 - 2828 |
| ORG. LETT., vol. 20, 2018, pages 4218 - 4222 |
| ORG. LETT., vol. 21, 2019, pages 4340 |
| ORG. LETT., vol. 22, 2020, pages 4038 - 4042 |
| RSC ADV., vol. 7, 2017, pages 28298 |
| SISELYWOOD: "Encyclopedia of Surface Active Agents", 1964, CHEMICAL PUBL. CO., INC. |
| SUZUKI ET AL., CHEM. REV., vol. 95, 1995, pages 2457 - 2483 |
| SYNTH. COMMUN., vol. 28, 1998, pages 4105 - 4121 |
| SYNTHESIS, vol. 45, 2013, pages 2699 - 2705 |
| T. S. WOODS: "The Formulator's Toolbox-Product Forms for Modern Agriculture", PESTICIDE CHEMISTRY AND BIOSCIENCE, THE FOOD ENVIRONMENT CHALLENGE |
| TETRAHEDRON LETT., vol. 50, 2009, pages 3790 - 3793 |
| TETRAHEDRON LETT., vol. 52, 2011, pages 3474 - 3477 |
| TETRAHEDRON, vol. 75, 2019, pages 4199 - 4211 |
| WATKINS ET AL.: "Handbook of Insecticide Dust Diluents and Carriers", 1950, DORLAND BOOKS |
Also Published As
| Publication number | Publication date |
|---|---|
| AR134418A1 (en) | 2026-01-14 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP3601263B1 (en) | Novel pyridazinone herbicides | |
| WO2016149315A1 (en) | Substituted pyrimidinyloxy pyridine derivatives as herbicides | |
| WO2015168010A1 (en) | Pyridazinone herbicides | |
| EP3303287A1 (en) | Substituted cyclic amides as herbicides | |
| EP3645515B1 (en) | 4-(3,4-dihydronaphth-1-yl or 2h-chromen-4-yl)-5-hydroxy-2h-pyradizin-3-ones as herbicides | |
| CA3064568A1 (en) | Herbicidal 3-substituted lactams | |
| WO2017074992A1 (en) | Novel pyrdazinone herbicides | |
| WO2018222646A1 (en) | Herbicidal amides | |
| WO2024072768A1 (en) | Substituted fluoropyridine as herbicides | |
| EP2912022B1 (en) | Substituted triazoles as herbicides | |
| WO2018204164A1 (en) | Pyrimidinyloxy benzo-fused compounds as herbicides | |
| WO2021183980A1 (en) | Substituted pyrimidines and triazines as herbicides | |
| WO2024015425A1 (en) | Herbicidal benzoxazines | |
| WO2015191377A1 (en) | Herbicidal substituted 3-arylpyrazoles | |
| WO2025111184A1 (en) | Substituted tetrahydroquinoline and tetrahydroquinoxaline herbicides | |
| EP4267556B1 (en) | Substituted pyridazinone herbicides | |
| WO2023129493A1 (en) | Substituted cyclopropylpyrimidne herbicides | |
| EP4294791A1 (en) | Herbicidal cyclic amides n-substituted with a haloalkylsulfonylanilide group | |
| AU2021315518A1 (en) | Substituted haloalkyl sulfonanilide herbicides | |
| IL269625A (en) | Novel pyridazinone herbicides |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 24818113 Country of ref document: EP Kind code of ref document: A1 |