WO2025106269A1 - Aqueous coating composition containing an aminosiloxane ester copolymer and a polyurethane binder, and methods for preparation and use thereof - Google Patents
Aqueous coating composition containing an aminosiloxane ester copolymer and a polyurethane binder, and methods for preparation and use thereof Download PDFInfo
- Publication number
- WO2025106269A1 WO2025106269A1 PCT/US2024/053778 US2024053778W WO2025106269A1 WO 2025106269 A1 WO2025106269 A1 WO 2025106269A1 US 2024053778 W US2024053778 W US 2024053778W WO 2025106269 A1 WO2025106269 A1 WO 2025106269A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- coating composition
- aqueous coating
- aqueous
- group
- water
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G18/00—Polymeric products of isocyanates or isothiocyanates
- C08G18/06—Polymeric products of isocyanates or isothiocyanates with compounds having active hydrogen
- C08G18/28—Polymeric products of isocyanates or isothiocyanates with compounds having active hydrogen characterised by the compounds used containing active hydrogen
- C08G18/40—High-molecular-weight compounds
- C08G18/42—Polycondensates having carboxylic or carbonic ester groups in the main chain
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G18/00—Polymeric products of isocyanates or isothiocyanates
- C08G18/06—Polymeric products of isocyanates or isothiocyanates with compounds having active hydrogen
- C08G18/08—Processes
- C08G18/0804—Manufacture of polymers containing ionic or ionogenic groups
- C08G18/0819—Manufacture of polymers containing ionic or ionogenic groups containing anionic or anionogenic groups
- C08G18/0823—Manufacture of polymers containing ionic or ionogenic groups containing anionic or anionogenic groups containing carboxylate salt groups or groups forming them
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G18/00—Polymeric products of isocyanates or isothiocyanates
- C08G18/06—Polymeric products of isocyanates or isothiocyanates with compounds having active hydrogen
- C08G18/28—Polymeric products of isocyanates or isothiocyanates with compounds having active hydrogen characterised by the compounds used containing active hydrogen
- C08G18/30—Low-molecular-weight compounds
- C08G18/34—Carboxylic acids; Esters thereof with monohydroxyl compounds
- C08G18/348—Hydroxycarboxylic acids
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G18/00—Polymeric products of isocyanates or isothiocyanates
- C08G18/06—Polymeric products of isocyanates or isothiocyanates with compounds having active hydrogen
- C08G18/28—Polymeric products of isocyanates or isothiocyanates with compounds having active hydrogen characterised by the compounds used containing active hydrogen
- C08G18/65—Low-molecular-weight compounds having active hydrogen with high-molecular-weight compounds having active hydrogen
- C08G18/66—Compounds of groups C08G18/42, C08G18/48, or C08G18/52
- C08G18/6633—Compounds of group C08G18/42
- C08G18/6659—Compounds of group C08G18/42 with compounds of group C08G18/34
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G18/00—Polymeric products of isocyanates or isothiocyanates
- C08G18/06—Polymeric products of isocyanates or isothiocyanates with compounds having active hydrogen
- C08G18/70—Polymeric products of isocyanates or isothiocyanates with compounds having active hydrogen characterised by the isocyanates or isothiocyanates used
- C08G18/72—Polyisocyanates or polyisothiocyanates
- C08G18/74—Polyisocyanates or polyisothiocyanates cyclic
- C08G18/75—Polyisocyanates or polyisothiocyanates cyclic cycloaliphatic
- C08G18/751—Polyisocyanates or polyisothiocyanates cyclic cycloaliphatic containing only one cycloaliphatic ring
- C08G18/752—Polyisocyanates or polyisothiocyanates cyclic cycloaliphatic containing only one cycloaliphatic ring containing at least one isocyanate or isothiocyanate group linked to the cycloaliphatic ring by means of an aliphatic group
- C08G18/753—Polyisocyanates or polyisothiocyanates cyclic cycloaliphatic containing only one cycloaliphatic ring containing at least one isocyanate or isothiocyanate group linked to the cycloaliphatic ring by means of an aliphatic group containing one isocyanate or isothiocyanate group linked to the cycloaliphatic ring by means of an aliphatic group having a primary carbon atom next to the isocyanate or isothiocyanate group
- C08G18/755—Polyisocyanates or polyisothiocyanates cyclic cycloaliphatic containing only one cycloaliphatic ring containing at least one isocyanate or isothiocyanate group linked to the cycloaliphatic ring by means of an aliphatic group containing one isocyanate or isothiocyanate group linked to the cycloaliphatic ring by means of an aliphatic group having a primary carbon atom next to the isocyanate or isothiocyanate group and at least one isocyanate or isothiocyanate group linked to a secondary carbon atom of the cycloaliphatic ring, e.g. isophorone diisocyanate
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D175/00—Coating compositions based on polyureas or polyurethanes; Coating compositions based on derivatives of such polymers
- C09D175/04—Polyurethanes
- C09D175/06—Polyurethanes from polyesters
-
- C—CHEMISTRY; METALLURGY
- C14—SKINS; HIDES; PELTS; LEATHER
- C14C—CHEMICAL TREATMENT OF HIDES, SKINS OR LEATHER, e.g. TANNING, IMPREGNATING, FINISHING; APPARATUS THEREFOR; COMPOSITIONS FOR TANNING
- C14C11/00—Surface finishing of leather
- C14C11/003—Surface finishing of leather using macromolecular compounds
-
- C—CHEMISTRY; METALLURGY
- C14—SKINS; HIDES; PELTS; LEATHER
- C14C—CHEMICAL TREATMENT OF HIDES, SKINS OR LEATHER, e.g. TANNING, IMPREGNATING, FINISHING; APPARATUS THEREFOR; COMPOSITIONS FOR TANNING
- C14C11/00—Surface finishing of leather
- C14C11/003—Surface finishing of leather using macromolecular compounds
- C14C11/006—Surface finishing of leather using macromolecular compounds using polymeric products of isocyanates (or isothiocyanates) with compounds having active hydrogen
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G77/00—Macromolecular compounds obtained by reactions forming a linkage containing silicon with or without sulfur, nitrogen, oxygen or carbon in the main chain of the macromolecule
- C08G77/42—Block-or graft-polymers containing polysiloxane sequences
- C08G77/452—Block-or graft-polymers containing polysiloxane sequences containing nitrogen-containing sequences
Definitions
- an aqueous coating composition and methods for its preparation and use for treating leather is provided. More particularly, the aqueous coating composition includes an aminosiloxane ester copolymer and a polyurethane binder.
- Representative silicone additives are commercially available as formulated blends, dispersions, suspensions, emulsions and fluids.
- Commercial examples include DOWSILTM FBL-3289 and DOWSILTM 5-7299 Dispersion (both high molecular weight silicone systems dispersed in water) and XIAMETERTM OFX-0531 fluid (aminomethoxy-functional polydimethyl siloxane), all available from The Dow Chemical Company of Midland, Michigan, USA.
- An aqueous coating composition comprises (A) an aminosiloxane ester copolymer, (B) a polyurethane binder, (C) a surfactant, and (D) water.
- the aqueous coating composition may be prepared by a method comprising mixing an aqueous copolymer emulsion and an aqueous polyurethane binder emulsion.
- the aqueous coating composition is useful for forming coatings on substrates where low coefficient of friction is desired.
- the aqueous coating composition introduced above comprises (A) an aminosiloxane ester copolymer, (B) a polymeric binder selected from the group consisting of a polyurethane and an acrylic polymer, (C) a surfactant, and (D) water.
- the aqueous coating composition may optionally further comprise an additional starting material, which may be selected from the group consisting of: (E) a biocide, (F) a pH modifier, (G) a pigment, (H) a thickener, a rheology modifier, a matting agent or duller (e.g., silica), an antifoam, a water repellent additive, an antiblocking additive, an abrasion resistance additive, an antioxidant, a UV absorber, a photostabilizer, an antistatic agent, a preservative (other than the biocide described above), a plasticizer, a flame retardant, a wetting agent (other than the surfactant described above), an opacifier, an extender, a plasticizer, and a combination of two or more thereof.
- an additional starting material which may be selected from the group consisting of: (E) a biocide, (F) a pH modifier, (G) a pigment, (H) a thickener, a r
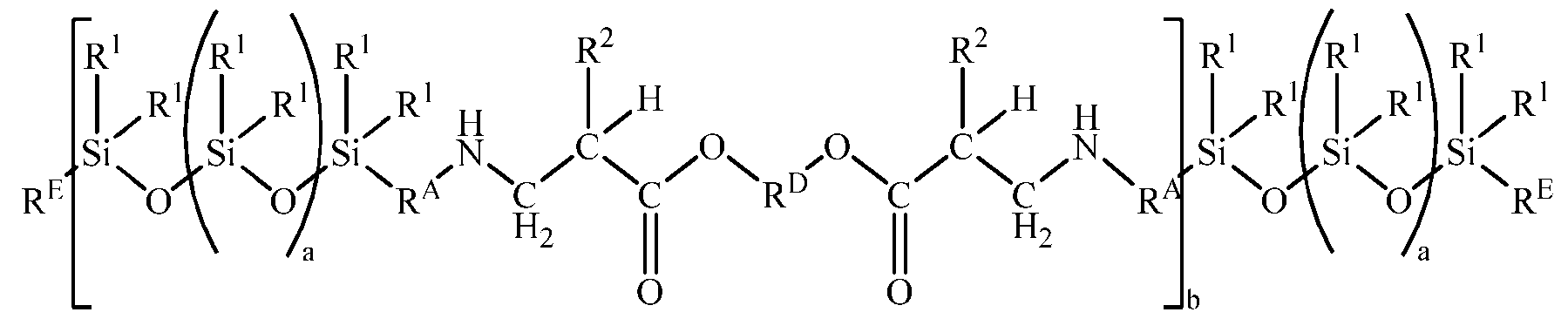
- Starting material (A) in the aqueous coating composition introduced above is an aminosiloxane ester copolymer.
- the aminosiloxane ester copolymer comprises formula (Al): where each R 1 is an independently selected monovalent hydrocarbon group of 1 to 12 carbon atoms, each R E is independently selected from the group consisting of hydroxyl and an aminofunctional group of formula H2N-R A -, each R A is an independently divalent hydrocarbon group of 1 to 12 carbon atoms, each R 2 is independently selected from the group consisting of hydrogen and methyl, each R D is an independently selected divalent hydrocarbon group of 2 to 20 carbon atoms, each subscript a independently has a value such that 0 ⁇ a ⁇ 150; and subscript has a value such that 1 ⁇ b ⁇ 100.
- Suitable monovalent hydrocarbon groups for R 1 include alkyl, alkenyl, aryl, and combinations thereof (e.g., aralkyl and aralkenyl).
- suitable alkyl groups include methyl, ethyl, propyl (including iso-propyl and n-propyl), butyl (including iso-butyl, n-butyl, sec-butyl, and tert-butyl), pentyl (including linear pentyl and/or cyclopentyl) and branched alkyl groups with 5 carbon atoms, hexyl (including linear hexyl and/or cyclohexyl) and branched alkyl groups with 6 carbon atoms), octyl (including linear octyl and/or cyclooctyl), branched alkyl groups with 8 carbon atoms), decyl (including linear decyl and/or cyclode
- the alkyl group for R 1 may be selected from the group consisting of methyl and ethyl, alternatively methyl.
- Suitable alkenyl groups for R 1 include vinyl, allyl and hexenyl; alternatively vinyl or allyl; and alternatively vinyl.
- Suitable aryl groups for R 1 may include cyclopentadienyl, phenyl, naphthyl, and anthracenyl.
- Suitable aralkyl groups for R 1 include tolyl, xylyl, benzyl, 1 -phenylethyl, and 2- phenylethyl.
- the aryl group for R 1 may be phenyl.
- Aralkyl groups such as benzyl, 1 -phenylethyl, and 2-phenylethyl, and aralkenyl groups such as styryl, may also be used for R 1 .
- each R 1 may be selected from the group consisting of methyl and phenyl.
- each R 1 may be methyl.
- Each R E is independently selected from the group consisting of a hydroxyl group and an amino-functional group of formula H2N-R A -, where R A is a divalent hydrocarbon group. Alternatively, each R E may be hydroxyl. Alternatively, each R E may be the amino-functional group of formula H2N-R A -.
- Each R A is an independently selected divalent hydrocarbon group of 1 to 12 carbon atoms, alternatively 2 to 12 carbon atoms, alternatively 2 to 5 carbon atoms, and alternatively 2 to 3 carbon atoms.
- the divalent hydrocarbon groups for R A may be linear, branched, or cyclic, or combinations thereof.
- Suitable divalent hydrocarbon groups for R A include alkylene groups, arylene groups, and combinations thereof (e.g., dialkylarylene groups).
- the alkylene group is exemplified by ethylene, propylene, or butylene.
- the arylene group for R A may be arylene group such as phenylene.
- R A may be a dialkylarylene group such as: each subscript u is independently 1 to 6, alternatively 1 to 2.
- each R A may be an alkylene group such as ethylene, propylene, or butylene; alternatively ethylene.
- Each R 2 is independently selected from the group consisting of hydrogen and methyl. Alternatively, each R 2 may be hydrogen.
- Each R D is an independently selected divalent hydrocarbon group of 2 to 20 carbon atoms, alternatively 2 to 12 carbon atoms, alternatively 3 to 12 carbon atoms, alternatively 4 to 12 carbon atoms, and alternatively 4 to 10 carbon atoms.
- the divalent hydrocarbon groups for R D may be linear, branched, cyclic, or combinations thereof. Suitable divalent hydrocarbon groups for R D include alkylene groups, arylene groups, and combinations thereof.
- each R D may be an alkylene group such as propylene, butylene, hexylene, octylene, decylene, or dodecylene; alternatively each R D may be butylene, hexylene, or decylene.
- R D may be a branched alkylene group.
- the arylene group for R D may be arylene group such as phenylene.
- R D may be a dialkylarylene group such as: each subscript u is independently 1 to 6, alternatively 1 to 2.
- Subscript a has a value such that 0 ⁇ a ⁇ 150.
- subscript a may have a value of 2 to 150, alternatively 14 to 145, alternatively 14 to 143, alternatively 14 to 84, alternatively 14 to 44, and alternatively 42 to 84.
- subscript a may be 14 to 16, alternatively 42 to 44, and alternatively 82 to 84.
- Subscript b has a value such that 2 ⁇ b ⁇ 100.
- subscript b may have a value of 2 to 20, alternatively 2 to 10, and alternatively 3 to 4.
- the copolymer described above may have a number average molecular weight (Mn) of > 1 ,000 g/mole to 250,000 g/mole measured by GPC according to the test method described hereinbelow.
- the copolymer may have a Mn of 4,000 g/mole to 250,000 g/mole, alternatively 4,000 g/mole to 100,000 g/mole, measured by GPC.
- the copolymer described above may have a weight average molecular weight (Mw) of 2,000 g/mol to 400,000 g/mol.
- Mw may be 10,000 g/mol to 390,000 g/mol; alternatively 12,000 g/mol to 200,000 g/mol; alternatively 15,000 g/mol to 185,000 g/mol; alternatively 19,000 g/mol to 175,000 g/mol; alternatively 20,000 g/mol to 100,000 g/mol; alternatively 21,000 g/mol to 80,000 g/mol; alternatively 22,000 g/mol to 75,000 g/mol; alternatively 25,000 g/mol to 65,000 g/mol; alternatively 30,000 g/mol to 60,000 g/mol; alternatively 35,000 g/mol to 55,000 g/mol; and alternatively 40,000 g/mol to 50,000 g/mol.
- the copolymer may be prepared by known methods, and the copolymer may be delivered in an emulsion.
- the copolymer
- the emulsion may comprise: (I) a liquid continuous phase comprising water, and (11) a discontinuous phase dispersed in the liquid continuous phase, where the discontinuous phase comprises the aminosiloxane ester copolymer described above.
- the amount of copolymer added to the emulsion can vary and is not limited. However, the amount typically may range from a weight ratio of copolymer/emulsion of 1 to 70%, alternatively 2 to 60%. Water (and additional starting materials, if present) may constitute the balance of the emulsion to 100%.
- the water is not generally limited, and may be utilized neat (i.e., absent any carrier vehicles/solvents), and/or pure (i.e., free from or substantially free from minerals and/or other impurities).
- the water may be processed or unprocessed prior to making the emulsion described herein.
- processes that may be used for purifying the water include distilling, filtering, deionizing, and combinations of two or more thereof, such that the water may be deionized, distilled, and/or filtered.
- the water may be unprocessed (e.g. may be tap water, i.e., provided by a municipal water system or well water, used without further purification).
- the water may be purified before use to make the emulsion.
- the emulsion described above may further comprise an additional starting material selected from the group consisting of a surfactant, an acid compound, an acid anhydride, a thickener, a stabilizer, a preservative, and a combination of two or more thereof.
- an additional starting material selected from the group consisting of a surfactant, an acid compound, an acid anhydride, a thickener, a stabilizer, a preservative, and a combination of two or more thereof.
- the copolymer described above may be self-emulsifying (i.e., a separate surfactant is optional).
- the surfactant may be anionic, cationic, nonionic, or amphoteric, or a combination of two or more thereof.
- the amount of surfactant may be 2 % to 25%, alternatively 2% to 20%, based on combined weights of all starting materials in the emulsion.
- the emulsion comprising (A) aminosiloxane ester copolymer may further comprise a surfactant.
- the surfactant may be anionic, cationic, nonionic, amphoteric, or a combination thereof.
- the anionic surfactant may selected from alkali metal sulfosuccinates, sulfonated glyceryl esters of fatty acids, salts of sulfonated monovalent alcohol esters, amides of amino sulfonic acids, sulfonated products of fatty acids nitriles, sulfonated aromatic hydrocarbons, condensation products of naphthalene sulfonic acids with formaldehyde, sodium octahydroanthracene sulfonate, sodium lauryl sulfate, alkali metal alkyl sulfates, alkyl ether sulfates having at least 8 carbon atoms, alkyl aryl ether sul
- Suitable anionic surfactants are commercially available from various sources including sodium lauryl sulfate, which is available from Pilot under the tradename CALIMULSETM SLS.
- Other anionic surfactants commercially available from The Dow Chemical Company include alkyldiphenyloxide disulfonate salts, which are available under the tradename DOWFAXTM; dioctyl sulfosuccinates, which are available under the tradename TRITONTM GR; phosphate esters, which are available under the tradename TRITONTM H-55, H-65, QS-44, OR XQS-20; sulfates and sulfonates, which are available under the tradename TRITONTM QS-15 and TRITONTM XN-45S.
- the cationic surfactant may be selected from dodecylamine acetate, octadecyl amine acetate, acetates of the amines of tallow fatty acids, homologues of aromatic amines having fatty acids, fatty amides derived from aliphatic diamines, fatty amides derived from aliphatic diamines, fatty amides derived from disubstituted amines, derivatives of ethylene diamine, quaternary ammonium compounds, salts of quaternary ammonium compounds, alkyl trimethylammonium hydroxides, dialkyldimethylammonium hydroxides, coconut oil, methylpolyoxyethylene cocoammonium chloride, dipalmitoylethyl hydroxyethylammonium methosulfate, amide derivatives of amino alcohols, amine salts of long chain fatty acids, and combinations thereof.
- Cationic surfactants are commercially available from various sources including dialkylmethyl quaternary ammonium compounds (e.g., cetrimonium chloride) under the tradename ARQUADTM from Akzo Nobel; ADOGENTM cationic surfactants from Evonik; TOMAHTM cationic surfactants from Tomah Products, Inc. of Milton, Wisconsin, USA; and VARIQUATTM cationic surfactants from Sea-Land Chemical Company of Westlake, Ohio, USA.
- dialkylmethyl quaternary ammonium compounds e.g., cetrimonium chloride
- ARQUADTM dialkylmethyl quaternary ammonium compounds
- ADOGENTM cationic surfactants from Evonik
- TOMAHTM cationic surfactants from Tomah Products, Inc. of Milton, Wisconsin, USA
- VARIQUATTM cationic surfactants from Sea-Land Chemical Company of Westlake, Ohio, USA.
- the nonionic surfactant may be selected from alkylphenol alkoxylates, ethoxylated and propoxylated fatty alcohols, alkyl polyglucosides and hydroxyalkyl polyglucosides, sorbitan derivatives, N- alkylglucamides, alkylene oxide block copolymers, such as block copolymers of ethylene oxide, propylene oxide and/or butylene oxide, fatty alcohol polyglycolethers, polyhydroxy and polyalkoxy fatty acid derivatives, amine oxides, silicone polyethers, various polymeric surfactants.
- Nonionic surfactants are commercially available, for example, alkylphenol alkoxylates are available under the tradename ECOSURFTM EH; secondary alcohol ethoxylates, nonylphenol ethoxylates, and ethylene oxide/propylene oxide copolymers are commercially available under the tradename TERGITOLTM; and specialty alkoxylates such as amine ethoxylates and octylphenol ethoxylates are available under the tradename TRITON TM, all from The Dow Chemical Company.
- alkylphenol alkoxylates are available under the tradename ECOSURFTM EH
- secondary alcohol ethoxylates, nonylphenol ethoxylates, and ethylene oxide/propylene oxide copolymers are commercially available under the tradename TERGITOLTM
- specialty alkoxylates such as amine ethoxylates and octylphenol ethoxylates are available under the tradename TRITON TM, all from
- the nonionic surfactant may be, e.g., trideceth-6 or trideceth-12, which are available under the tradename SYNPERONICTM from Croda or LUTENSOLTM from BASF.
- the nonionic surfactant may be e.g., a fatty alcohol polyglycol ether such as GENAPOLTM UD 050, and GENAPOLTM UDI 10, which are commercially available from Clariant of Frankfurt, Germany.
- the nonionic surfactant may comprise, or may be, a silicone polyether (SPE).
- SPE silicone polyether
- the silicone polyether as an emulsifier may have a rake type structure wherein the polyoxyethylene or polyoxy ethylene-polyoxypropylene copolymeric units are grafted onto the siloxane backbone, or the SPE can have an ABA block copolymeric structure wherein A represents the polyether portion and B the siloxane portion of an ABA structure.
- Suitable silicone polyethers include DOWSILTM OFX-5329 Fluid from The Dow Chemical Company.
- the nonionic surfactant may be selected from polyoxyalkylene-substituted silicones, silicone alkanolamides, silicone esters and silicone glycosides.
- Such silicone-based surfactants are known in the art, and have been described, for example, in U.S. Patent 4,122,029 to Gee et al., U.S. Patent 5,387,417 to Rentsch, and U.S. Patent 5,811,487 to Schulz et al.
- Suitable amphoteric surfactants include betaines such as alkyl(C12-14)betaine, cocoamidopropylbetaine, cocoamidopropyldimethyl-hydroxysulphobetaine, dodecylbetaine, hexadecylbetaine, and tetradecylbetaine; sultaines such as cocamidopropylhydroxysultaine; lecithin; hydrogenated lecithin; cocoamphodiacetates; cocoiminodipropionate; and dodecyliminodipropionate.
- betaines such as alkyl(C12-14)betaine, cocoamidopropylbetaine, cocoamidopropyldimethyl-hydroxysulphobetaine, dodecylbetaine, hexadecylbetaine, and tetradecylbetaine
- sultaines such as cocamidopropylhydroxysultaine
- lecithin hydrogenated lecithin
- the surfactant in the aqueous copolymer emulsion may be a nonionic surfactant.
- the surfactant may be an organic surfactant.
- the surfactant may be both organic and nonionic.
- the aqueous copolymer emulsion may be formed as a water-in-oil emulsion (w/o), which contains a water-in-oil surfactant, (which may subsequently inverted by addition of more water).
- the water-in-oil surfactant may be nonionic and may be selected from polyoxyalkylene- substituted silicones, silicone alkanolamides, silicone esters and silicone glycosides, as described above.
- the emulsion is an oil-in-water (o/w) emulsion, it may include nonionic surfactants known in the art to prepare o/w emulsions.
- Suitable nonionic surfactants for this embodiment are exemplified by the polyoxyethylene alkyl ethers, polyoxyethylene alkylphenol ethers, polyoxyethylene lauryl ethers, polyoxyethylene sorbitan monooleates, polyoxyethylene alkyl esters, polyoxyethylene sorbitan alkyl esters, polyethylene glycol, polypropylene glycol, diethylene glycol, ethoxylated trimethylnonanols, and polyoxyalkylene glycol modified poly siloxane surfactants, as described above.
- the acid compound may optionally be added to the aqueous copolymer emulsion for adjusting pH.
- Suitable acids include acetic acid, formic acid, propionic acid, and combinations thereof. Suitable acids for adjusting pH are disclosed, for example, in U.S. Patent 6180117.
- Emulsions may be prepared in a batch, semi-continuous, or continuous process using conventional equipment.
- mixing the starting materials to form the emulsion may occur, for example using, batch equipment with high-shear and high-speed dispersers include those made by Charles Ross & Sons (NY), Hockmeyer Equipment Corp. (NJ); batch mixing equipment such as those sold under the tradename SpeedmixerTM; batch equipment with high shear actions include Banbury-type (CW Brabender Instruments Inc., NJ) and Henschel type (Henschel mixers America, TX).
- Illustrative examples of continuous mixers/compounders include extruders, such as single-screw, twin-screw, and multi-screw extruders, co-rotating extruders, such as those manufactured by Krupp Werner & Pfleiderer Corp (Ramsey, NJ), and Leistritz (NJ); twin-screw counterrotating extruders, two-stage extruders, twin-rotor continuous mixers, dynamic or static mixers or combinations of these equipment.
- extruders such as single-screw, twin-screw, and multi-screw extruders
- co-rotating extruders such as those manufactured by Krupp Werner & Pfleiderer Corp (Ramsey, NJ), and Leistritz (NJ)
- twin-screw counterrotating extruders twin-stage extruders
- twin-rotor continuous mixers dynamic or static mixers or combinations of these equipment.
- any acid compound may be added at the end of the method, i.e., once the desired dilution level is reached.
- the emulsion of the aminosiloxane ester copolymer described above may be used in an amount sufficient to provide 0.1% to 50%, alternatively 0.1% to 10%, alternatively 0.5% to 10%, and alternatively 1% to 5%, of (A) the aminosiloxane ester copolymer based on combined weights of all the starting materials in the aqueous coating composition described herein.
- Starting material (B) in the aqueous coating composition described herein is a polymeric binder (dry polymer).
- the polymeric binder may be (Bl) a polyurethane or (B2) an acrylic polymer.
- the polyurethane may be delivered in an aqueous dispersion.
- the aqueous polyurethane dispersion used herein to prepare the aqueous coating composition can be an externally stabilized polyurethane dispersion or an internally stabilized polyurethane dispersion.
- Internally stabilized polyurethane dispersion herein refers to a polyurethane dispersion that is stabilized through the incorporation of ionically or nonionically hydrophilic pendant groups within the polyurethane particles dispersed in the liquid medium.
- nonionic internally stabilized polyurethane dispersions are described in U.S. Patents 3905929 and 3920598.
- Ionic internally stabilized polyurethane dispersions are known and are described in U.S. Patent 6231926.
- dihydroxyalkylcarboxylic acids such as described in U.S. Patent 3412054 are used to make anionic internally stabilized polyurethane dispersions.
- a common monomer used to make an anionic internally stabilized polyurethane dispersion is dimethylolpropionic acid (DMPA).
- the polyurethane may be prepared by polymerization of monomers selected from polyisocyanates having 2 or more isocyanate functionalities and having 4 to 40 carbon atoms, polyols such as diols, monomers bearing at least one isocyanate group or at least one isocyanate reactive group and which in addition bear at least one hydrophilic group or potentially hydrophlic group, and optionally one or more compounds having reactive groups comprising alcoholic hydroxyl groups, primary or secondary amino groups or isocyanate groups.
- monomers selected from polyisocyanates having 2 or more isocyanate functionalities and having 4 to 40 carbon atoms, polyols such as diols, monomers bearing at least one isocyanate group or at least one isocyanate reactive group and which in addition bear at least one hydrophilic group or potentially hydrophlic group, and optionally one or more compounds having reactive groups comprising alcoholic hydroxyl groups, primary or secondary amino groups or isocyanate groups.
- Suitable polyisocyanates include conventional aliphatic, cycloaliphatic, araliphatic and aromatic isocyanates.
- the polyisocyanate may be selected from the group of diphenylmethane diisocyanates (“MDI”), polymeric diphenylmethane diisocyanates (“pMDI”), toluene diisocyanates (“TDI”), hexamethylene diisocyanates (“HD I”), dicyclohexylmethane diisocyanates (“HMD I”), isophorone diisocyanates (“IPDI”), cyclohexyl diisocyanates (“CHDI”), naphthalene diisocyanate (“NDI’’), phenyl diisocyanate (“PDI”), tetramethylene diisocyanate (“TMDI”), and combinations thereof.
- MDI diphenylmethane diisocyanates
- pMDI polymeric diphenylmethane diis
- the polyisocyanate may have formula OCN — R — NCO, wherein R is an alkyl moiety, an aryl moiety, or an arylalkyl moiety.
- the polyisocyanate can include any number of carbon atoms described above, alternatively from 4 to 20 carbon atoms.
- suitable polyisocyanates include: alkylene diisocyanates with 4 to 12 carbons in the alkylene radical such as 1,12-dodecane diisocyanate, 2-ethyl-l,4- tetramethylene diisocyanate, 2-methyl-l,5-pentamethylene diisocyanate, 1,4-tetramethylene diisocyanate and preferably 1,6-hexamethylene diisocyanate; cycloaliphatic diisocyanates such as 1,3- and 1 ,4-cyclohexane diisocyanate as well as any mixtures of these isomers, 1-isocyanato- 3,3,5-trimethyl-5-isocyanatomethylcyclohexane, 2,4- and 2,6-hexahydrotoluene diisocyanate as well as the corresponding isomeric mixtures, 4,4'- 2,2'-, and 2,4'-dicyclohexylmethane diisocyanate
- the polyisocyanate may comprise IPDI.
- the polyurethane may be made from one or more diisocyanates, such as IPDI or TMDI and one or more polyols, such as a polyether polyol, polycarbonate polyol, or polyester polyol, e.g., having a molecular weight (Mw) of 5,000 or less, or of 2,000 or less.
- polyols may be linear and may have have two hydroxyl groups, one at each end.
- Suitable polyols include polyester polyols, which are reactive with the isocyanate described above include, but are not limited to, hydroxyl-functional reaction products of polyhydric alcohols, such as ethylene glycol, propylene glycol, diethylene glycol, 1,4- butanediol, neopentylglycol, 1,6-hexanediol, cyclohexane dimethanol, glycerol, trimethylolpropane, pentaerythritol, sucrose, or polyether polyols or mixtures of such polyhydric alcohols, and polycarboxylic acids, particularly dicarboxylic acids or their ester-forming derivatives, for example succinic, glutaric and adipic acids or their dimethyl esters, sebacic acid, phthalic anhydride, tetrachlorophthalic anhydride, dimethyl terephthalate or mixtures thereof.
- polyhydric alcohols such as ethylene glycol, propylene glyco
- Polyester polyols obtained by the polymerization of lactones, e.g. caprolactone, in conjunction with a polyol, or of hydroxy carboxylic acids, e.g. hydroxy caproic acid, may also be used.
- the polyol comprises a mixture of polyester and polyether polyols.
- “Externally stabilized polyurethane dispersion” herein refers to a polyurethane dispersion that fails to have an ionic or nonionic hydrophilic pendant groups and thus requires the addition of a surfactant to stabilize the polyurethane dispersion.
- the surfactant can be those described in the copolymer emulaion above. Examples of externally stabilized polyurethane dispersions are described in U.S. Patents 2,968,575; 5,539,021; 5,688,842 and 5,959,027.
- the polyurethane dispersion may be an internally stabilized polyurethane dispersion.
- the polyurethane dispersion may comprise an aqueous polyurethane dispersion based on isophorone diisocyanate and polyester polyol, stabilized by carboxylate groups in the polyurethane backbone.
- the polyurethane may have glass transition temperature of -44 °C.
- Suitable polyurethane dispersions for use in preparing the aqueous coating composition are known in the art and are commercially available, for example, BAYDERMTM polyurethane dispersions such as BAYDERMTM 91UD, which is available from The Dow Chemical Company of Midland, Michigan, USA.
- the polyurethane dispersion may be as described in U.S. Patent 11,518,905 to Lenoble et al.
- polyurethane dispersions can be prepared by methods conventional in the art. See, for example, the methods described in P. Pieterich, Aqueous Emulsion, Dispersion and Solutions of Polyurethanes; Synthesis and Properties in Progress in Organic Coatings 9 (1981) 281-340. See also: US7232859, US2004/0167252 and US2011/0112245.
- Such polyurethanes are commonly prepared by reacting an organic polyisocyanate(s) with an organic compound(s) containing isocyanatereactive groups, particularly a polyol. The reaction may be carried out in the presence of a catalyst such as organic tin compounds and/or tertiary amines.
- the polyurethanes are made into aqueous dispersion by conventional means and may be anionic salt functional, non-ionic or anionic polyurethane dispersions.
- the polyurethane dispersion may be an anionic polyurethane dispersion prepared by reacting one or more polyol with an organic compound having at least one acid group and at least two active hydrogen functionalities and a polyisocyanate.
- Suitable organic compounds having at least one acid group and at least two active hydrogen functionalities include, for example, 2,2-dimethylolacetic acid and 2,2- dimethylolpropionic acid.
- acid groups suitable for the organic compound include, carboxylic acid, sulfonic acid, phosphoric, phosphonic acid and the like.
- the surfactant may be introduced into the aqueous coating composition with starting material (A) the aminosiloxane ester copolymer and (B) the polymeric binder, both of which may be delivered in aqueous emulsions or dispersions, as described above, and/or the surfactant may be added separately, or both.
- the surfactant in the aqueous coating composition may comprise an organic surfactant.
- the surfactant in the aqueous coating composition may be a nonionic surfactant.
- the surfactant in the aqueous coating composition may be an organic, nonionic surfactant. Said organic, nonionic surfactant may have a relatively high hydrophobic - lipophobic balance (HLB) value.
- organic, nonionic surfactants are as described above in the emulsion for starting material (A), the aminosiloxane ester copolymer, and include those which are commercially available such as (i) 2,6,8-trimethyl-4- nonyloxy polyethylene oxy ethanols (6EO) and (10EO) sold under the names TERGITOLTM TMN-6 and TERGITOLTM TMN-10; (ii) the Ci 1-15 secondary alkyl polyoxyethylene ethers (e.g., Ci 1-15 secondary alcohol ethoxylates 7EO, 9EO, and 15EO sold under the names TERGITOLTM 15-S-7, TERGITOLTM 15-S-9, and TERGITOLTM 15-S-15, which has HL value 15.4), other Cuis secondary alcohol ethoxylates sold under the tradenames ECOSURFTM EH-40 and TERGITOLTM 15-S-12, TERGITOLTM 15-S-30, and TERGITOLTM 15-S-40
- polyoxyethylene 23 lauryl ether (Laureth-23) sold commercially under the trademark BRUTM 23; and RENEXTM 30, a polyoxyethylene ether alcohol;
- alkyLoxo alcohol poly glycol ethers such as GENAPOLTM UD 050 (with HLB value of 11.4), and GENAPOLTM UDI 10 (with HLB value of 14.4),
- alkyl polyethylene glycol ether based on ClO-Guerbet alcohol and ethylene oxide such as LUTENSOLTM XP 79
- alkyl poly glycosides such as those sold under the trade name GlucoponTM by BASF, and alkyl glucosides such as decyl glucoside, lauryl glucoside, and coco-glucoside, which are sold under the trade name EcoSenseTM by The Dow Chemical Company of Midland, Michigan, USA.
- nonionic surfactants include TERGITOLTM 15-S-5, also from The Dow Chemical Company, which has an HLB value of 10.5; Lutensol XP 50 with an HLB value of 10, Lutensol XP 79 (an alcohol ethoxylate), Lutensol XP 100 (an alcohol ethoxylate), and Lutensol XP 140 with an HLB value of 16, each of which is available from BASF.
- the amount of surfactant in the aqueous coating composition may be, for example, 0.03% to 25%, alternatively 0.03% to 4% based on combined weights of all the starting materials in the aqueous coating composition described herein.
- Starting material (D) in the aqueous coating composition is water. All or a portion of the water may be introduced into the aqueous coating composition with starting material (A) the aminosiloxane ester copolymer and (B) the polyurethane binder, both of which are typically delivered in aqueous emulsions or dispersions, as described above.
- the water is not specifically restricted and may be as described above in the emulsion of starting material (A) the aminosiloxane ester copolymer.
- the amount of water in the aqueous coating composition is not specifically restricted. The amount of water may be > 0 to 70%, alternatively > 0 to 65%, alternatively 40% to 70%, based on combined weights of all the starting materials in the aqueous coating composition described herein.
- One or more other optional additional starting materials may be included in the aqueous coating composition, as long as properties of the aqueous coating composition and performance of the coating made from the composition are not compromised.
- Suitable other optional starting materials include (E) a biocide, (F) a pH modifier, (G) a pigment, (H) a thickener, a rheology modifier, a matting agent or duller (e.g., silica), an antifoam, a water repellent additive, an antiblocking additive, an abrasion resistance additive, an antioxidant, a UV absorber, a photo-stabilizer, an antistatic agent, a preservative (other than the biocide described above), a plasticizer, a flame retardant, a wetting agent (other than the surfactant described above), an opacifier, an extender, a plasticizer, and a combination of two or more thereof.
- Starting material (E) is a biocide.
- the biocide is optional and may be added to the aqueous coating composition to ensure that the coating prepared from the aqueous coating composition provides protection against microbial attack during storage and transportation.
- the biocide is exemplified by a fungicide, an herbicide, a pesticide, an antimicrobial agent, or a combination thereof.
- the aqueous coating composition may comprise a fungicide, an antimicrobial agent, or a combination thereof.
- the amount of biocide will vary depending on factors including the type of biocide selected and the benefit desired.
- the amount of biocide may range from greater than 0 % to 5 %, alternatively from 1 ppm to 1500 ppm, each based on the weight of all starting materials in the aqueous coating composition.
- Biocides are known in the art and are commercially available.
- PREVENTOLTM C40-L which is a preservative based on p-chloro-m-cresol (PCMC); BIOBANTM O 45 Antimicrobial;
- PreventolTM BIT 20 N; PreventolTM BM 5; PreventolTM CMK 40; and PreventolTM D 7 are biocides suitable for leather treatment that are commercially available from Lanxess of Pittsburgh, Pennsylvania, USA.
- Starting material (F) is a pH modifier.
- the pH modifier is optional and may be added to change pH of the aqueous coating composition. Without wishing to be bound by theory, it is thought that the pH modifier may be used to reduce particle size of the aqueous coating composition (which is in the form of an emulsion) as compared to a composition that does not contain the pH modifier.
- the pH modifier may be an acid as described above for the aqueous copolymer emulsion to deliver (A) the aminosiloxane ester copolymer.
- the pH modifier may be a buffer such as sodium carbonate, sodium bicarbonate, and a combination thereof.
- the amount of the pH modifier depends on various factors such as the type of rheology modifier present, if any, however the amount of pH modifier may be 0 to 5%, alternatively > 0 to 5%, based on combined weights of all starting materials in the aqueous coating composition.
- Starting material (G) is a pigment.
- the pigment is optional and may be added to the aqueous coating composition to impart a desired color to a coating prepared from the aqueous coating composition.
- Suitable pigments are known in the art and are commercially available. Pigments include carbon black and titanium dioxide. The amount of pigment may be > 0% based on based on combined weights of all the starting materials combined to prepare the aqueous coating composition described herein.
- starting materials to make the aqueous coating composition described above there may be overlap between types because certain starting materials described herein may have more than one function. For example, certain particulates may be useful as pigments and as flame retardants, e.g., carbon black.
- the additional starting materials are distinct from one another and from starting materials (A), (B), (C) and (D) described above. Examples of suitable optional additional starting materials and their amounts may be found for example, in US Patents 9200404, 10100377, and 11518905.
- the aqueous coating composition may be prepared by any convenient means using any convenient equipment.
- the aqueous coating composition may be prepared by a method comprising:
- step 1) a discontinuous phase dispersed in the liquid continuous phase
- the discontinuous phase comprises (A) the aminosiloxane ester copolymer, where the aqueous copolymer emulsion further comprises a surfactant, and water; b) an aqueous binder emulsion comprising (B) the polymeric binder and water.
- the method may optionally further comprise one or more additional steps before step 1), e.g., dispersing a starting material comprising a pigment in water before step 1), thereby preparing an aqueous pigment dispersion, and mixing the aqueous pigment dispersion with the starting materials in step 1).
- the method may optionally further comprise one or more additional steps after step 1), e.g.
- the method may further comprise removing agglomerated particles after step 1) and/or the method may further comprise 2) adding an additional starting material selected from the group consisting of (E) a biocide, (F) a pH modifier, (G) a pigment, (H) a thickener, a rheology modifier, a matting agent or duller (e.g., silica), an antifoam, a water repellent additive, an antiblocking additive, an abrasion resistance additive, an antioxidant, a UV absorber, a photostabilizer, an antistatic agent, a preservative (other than the biocide described above), a plasticizer, a flame retardant, a wetting agent (other than the surfactant described above), an opacifier, an extender, a plasticizer, and a combination of two or more thereof.
- an additional starting material selected from the group consisting of (E) a biocide, (F) a pH modifier, (G) a pigment, (H) a thickener
- the method may optionally further comprise adding additional water for dilution, for example, for use at remote site to reduce the total solids of the aqueous coating composition to a desired range. Accordingly, the aqueous coating composition may be shipped in any stable concentrated form.
- step 1) Mixing the starting materials in step 1) (and any optional additional steps) may be performed by any convenient means, such as mixing optionally under shear, using the equipment and methods described above for making the emulsion of (A) the aminosiloxane ester copolymer.
- simple mixing may be performed to mix the starting materials in step 1), e.g., shear is not required.
- the aqueous coating composition prepared described above may be combined with a crosslinker and then used to treat substrates comprising leather.
- a crosslinker and the aqueous coating composition are combined shortly before use (e.g., shortly before applying the resulting crosslinkable coating composition to a substrate).
- the aqueous coating composition described herein may be provided in a multiple-part system comprising a base part and a curing agent part.
- the base part comprises the aqueous coating composition comprising starting materials (A), (B), (C), and (D), and optionally one or more additional starting materials, as described above.
- the curing agent part comprises a crosslinker.
- Suitable crosslinkers for polyurethanes are known in the art and include melamine resins, polyaziridine resins, aminoplast resins, amide- and amine-formaldehyde resins, and polyisocyanates (which may be blocked or unblocked polyisocyanates).
- Polyisocyanates may contain free isocyanate groups based on aliphatic, cycloaliphatic, araliphatic and/or aromatic isocyanates.
- the crosslinker may be water dispersible.
- the crosslinker may comprise a polyisocyanate, alternatively an aliphatic polymeric isocyanate.
- the crosslinking agent may be used in an amount of 0.1% or more, and up to 20%, for example, from 1% to 10%, based on combined weights of base part and curing agent part.
- Suitable crosslinkers are commercially available and include BINDER LS-3486-HS Crosslinker, which is supplied as a solution with viscosity ⁇ 50 cP (measured by a Brookfield LVT at 60 rpm) containing a reactive aliphatic polyisocyanate resin (50-52%); and BINDER LS-3492 Crosslinker, which is supplied as a solution with viscosity ⁇ 50 cP (measured by a Brookfield LVT at 60 rpm) containing a reactive aliphatic polyisocyanate resin (49%-51%) supplied in ethyl 3-ethoxypropionate (49%- 51%), both of which are commercially available from The Dow Chemical Company.
- kits further comprises instructions for combining the base part and the curing agent part and optionally for using the resulting crosslinkable coating composition for treating substrates comprising leather, as follows.
- the aqueous coating composition prepared as described above may be used to treat substrates comprising leather (i.e., natural or synthetic leather).
- a method for treating a substrate comprises: pre-i) combining the base part comprising the aqueous coating composition and the curing agent part comprising crosslinker, described above, thereby preparing a crosslinkable coating composition; i) applying the crosslinkable coating composition to a substrate comprising leather; and ii) drying the crosslinkable coating composition to remove water, thereby forming a coating on the substrate.
- the base part and the curing agent part may be combined by any convenient means, such as simple mixing, e.g., when the crosslinker is supplied in water.
- the crosslinkable coating composition may be applied to the substrate by any convenient method.
- the crosslinkable coating composition may be applied on the substrate by a method selected from the group consisting of spraying, knife coating, roll coating, casting, drum coating, dipping, gravure coating, bar coating, screen coating, curtain coating, brush coating, and combinations thereof.
- the amount of the crosslinkable coating composition applied on the substrate is not specifically restricted, and may have a wet coating thickness of 10 pm to 100 pm, which may correspond to a dry coating thickness of 2 pm to 70 pm. Drying may be performed by any convenient method, such as air drying or heat drying the coated substrate.
- the conditions for heat drying depend on various factors including the substrate selected. For example, when the substrate comprises natural leather, the heat drying temperature may be ⁇ 120 °C.
- the heat drying temperature may be ⁇ 180 °C, alternatively ⁇ 150 °C for a time sufficient to remove most or all of the water.
- the method may optionally further comprise iii) repeating steps i) and ii) one or more times to increase the thickness of the coating on the substrate.
- the thickness of the coating to be formed on the substrate is not specifically restricted.
- the aqueous coating composition and method for treating a substrate described herein may be used to provide coatings on leather, which includes both natural and synthetic leathers, such as for automobile and other motor vehicle applications (e.g., as coatings on armrests, dashboards, seating, and other interior components); clothing such as coats, pants, flight jackets, motorcycle clothing, shoes, and gloves; luggage or handbags; accessories such as belts, wallets, and datebooks; furniture; or saddles for bicycles or motorcycles.
- leather which includes both natural and synthetic leathers, such as for automobile and other motor vehicle applications (e.g., as coatings on armrests, dashboards, seating, and other interior components); clothing such as coats, pants, flight jackets, motorcycle clothing, shoes, and gloves; luggage or handbags; accessories such as belts, wallets, and datebooks; furniture; or saddles for bicycles or motorcycles.
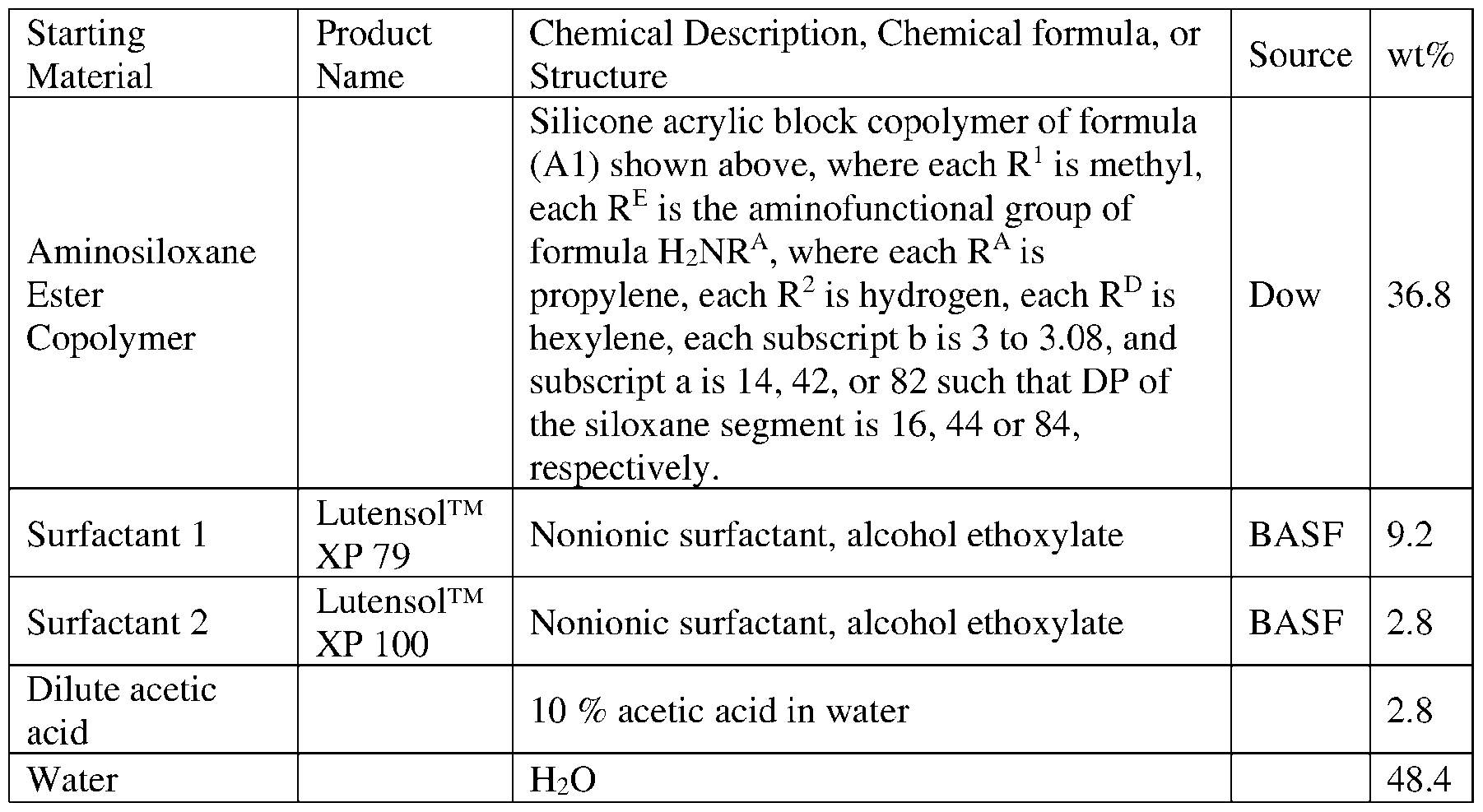
- aqueous copolymer emulsions with the starting materials in amounts shown below in Table 1 were prepared as described in PCT Patent Application Publication WO2023/278918.
- aminosiloxane ester copolymer, surfactants and inversion water were added and mixed by a high shear mixer at 3500 rpm for 30 sec (dental mixer), then the resulting thick phase was diluted by water, and the acetic acid solution added at the end of the process.
- aqueous coating compositions were prepared as follows. Aqueous emulsions and other starting materials except for thickener (if present) under low shear stirring. The resultant mixture was then filtered to remove agglomerated particles. Finally, thickener (if present) was added into the mixture.
- the starting materials in these aqueous coating compositions are described in Table 4 and the amounts of each starting material are shown below in Table 5 for compositions with a polyurethane binder and Table 7 for an acrylic polymer binder.
- the aqueous coating compositions were used to prepare coating on substrates and tested, as follows: To form the aqueous coating compositions of the present invention, the aqueous copolymer emulsions described above in Reference Example 3 and Binder 1 described below in Table 4 were mixed together using a laboratory stirrer under low shear (IKA lab mixer, 500 rpm, 5 min).
- the second step was to blend the emulsion described above with Crosslinker 1 under low shear stirring (IKA lab mixer, 500 rpm, 5 min).
- the resulting crosslinkable coating composition was applied using a 60, um cylindrical film applicator from BYK upon a sheet of a sealed opacity chart (form 2A obtained from Leneta Company).
- the top of the chart was black and the bottom was white with an overall dimensions of 5-1/2 x 10 in (140 x 254 mm).
- the coated chart was then dried in a conventional oven for 2 minutes at 80 °C.
- an aminosiloxane ester copolymer as additive at a low loading in an aqueous coating composition including a polyurethane water-based binder provided beneficial properties in the coatings prepared therewith, such as low CoF.
- beneficial properties in the coatings prepared therewith such as low CoF.
- using the aminosiloxane ester copolymer may further provide one or more of the following benefits to the coating: high contact angle with water, low stick slip performance, and low leaching.
- the aminosiloxane ester copolymer may be delivered as an emulsion using a nonionic surfactant.
- the aminosiloxane ester copolymer may provide one or more benefits over other silicone leather treatment composition active ingredients.
- the aminosiloxane ester copolymer has low cyclic polyorganosiloxane content ( ⁇ 0.05 % octamethylcyclotetrasiloxane), stability after aging as evidenced by low cyclics content after aging, little or no crosslinking, and potential for biodegradability.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Health & Medical Sciences (AREA)
- Medicinal Chemistry (AREA)
- Polymers & Plastics (AREA)
- Engineering & Computer Science (AREA)
- General Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Materials Engineering (AREA)
- Wood Science & Technology (AREA)
- Manufacturing & Machinery (AREA)
- Paints Or Removers (AREA)
Abstract
An aqueous coating composition includes an aminosiloxane ester copolymer, a polyurethane polymer, a surfactant, and water. The aqueous coating composition can be prepared by mixing an aqueous emulsion comprising the aminosiloxane copolymer, the surfactant and water with an aqueous polyurethane emulsion or dispersion. The aqueous coating composition is useful for leather treatment. The aqueous coating composition may be combined with a crosslinker, applied to a leather substrate and dried to remove water, thereby providing a coating with a low coefficient of friction on the leather substrate.
Description
AQUEOUS COATING COMPOSITION CONTAINING AN AMINOSILOXANE ESTER COPOLYMER AND A POLYURETHANE BINDER, AND METHODS FOR PREPARATION AND USE THEREOF
CROSS REFERENCE TO RELATED APPLICATIONS
[0001] This application claims the benefit of U.S. Provisional Patent Application Serial No. 63/599,579 filed on 16 November 2023 under 35 U.S.C. §119 (e). U.S. Provisional Patent Application Serial No. 63/599,579 is hereby incorporated by reference.
FIELD
[0002] An aqueous coating composition and methods for its preparation and use for treating leather is provided. More particularly, the aqueous coating composition includes an aminosiloxane ester copolymer and a polyurethane binder.
INTRODUCTION
[0003] Leather is often finished with one or more coatings in order to improve its overall performance, e.g. wear resistance, flexibility, etc. Such coatings are most commonly dispersions of polyurethane. Representative examples are described in: U.S. Patents 3930921, 6353051, 6794445, 8591999, 9200404, and 10100377; and U.S. Patent Application Publications 2005/0222368 and 2010/0310882. Silicones are often included in the aqueous coating composition to improve one or more of haptic properties (“hand”), appearance, water repellency, abrasion resistance and breathability as described in U.S. Patent 11518905. Representative silicone additives are commercially available as formulated blends, dispersions, suspensions, emulsions and fluids. Commercial examples include DOWSIL™ FBL-3289 and DOWSIL™ 5-7299 Dispersion (both high molecular weight silicone systems dispersed in water) and XIAMETER™ OFX-0531 fluid (aminomethoxy-functional polydimethyl siloxane), all available from The Dow Chemical Company of Midland, Michigan, USA.
[0004] There continues to be a need for improved aqueous leather coating compositions that offer improved performance attributes including low coefficient of friction.
SUMMARY
[0005] An aqueous coating composition comprises (A) an aminosiloxane ester copolymer, (B) a polyurethane binder, (C) a surfactant, and (D) water. The aqueous coating composition may be prepared by a method comprising mixing an aqueous copolymer emulsion and an aqueous polyurethane binder emulsion. The aqueous coating composition is useful for forming coatings on substrates where low coefficient of friction is desired.
DETAILED DESCRIPTION
[0006] The aqueous coating composition introduced above comprises (A) an aminosiloxane ester copolymer, (B) a polymeric binder selected from the group consisting of a polyurethane and an acrylic polymer, (C) a surfactant, and (D) water. The aqueous coating composition may optionally further comprise an additional starting material, which may be selected from the group consisting of: (E) a biocide, (F) a pH modifier, (G) a pigment, (H) a thickener, a rheology modifier, a matting agent or duller (e.g., silica), an antifoam, a water repellent additive, an antiblocking additive, an abrasion resistance additive, an antioxidant, a UV absorber, a photostabilizer, an antistatic agent, a preservative (other than the biocide described above), a plasticizer, a flame retardant, a wetting agent (other than the surfactant described above), an opacifier, an extender, a plasticizer, and a combination of two or more thereof.
( A) Aminosiloxane Ester Copolymer
[0007] Starting material (A) in the aqueous coating composition introduced above is an aminosiloxane ester copolymer. The aminosiloxane ester copolymer comprises formula (Al):
where each R1 is an independently selected monovalent hydrocarbon group of 1 to 12 carbon atoms, each RE is independently selected from the group consisting of hydroxyl and an aminofunctional group of formula H2N-RA-, each RA is an independently divalent hydrocarbon group of 1 to 12 carbon atoms, each R2 is independently selected from the group consisting of hydrogen and methyl, each RD is an independently selected divalent hydrocarbon group of 2 to 20 carbon atoms, each subscript a independently has a value such that 0 < a < 150; and subscript has a value such that 1 < b < 100.
[0008] Suitable monovalent hydrocarbon groups for R1 include alkyl, alkenyl, aryl, and combinations thereof (e.g., aralkyl and aralkenyl). For example, suitable alkyl groups include methyl, ethyl, propyl (including iso-propyl and n-propyl), butyl (including iso-butyl, n-butyl, sec-butyl, and tert-butyl), pentyl (including linear pentyl and/or cyclopentyl) and branched alkyl groups with 5 carbon atoms, hexyl (including linear hexyl and/or cyclohexyl) and branched alkyl groups with 6 carbon atoms), octyl (including linear octyl and/or cyclooctyl), branched alkyl
groups with 8 carbon atoms), decyl (including linear decyl and/or cyclodecyl) and branched alkyl groups with 10 carbon atoms, and dodecyl (including linear dodecyl and/or cyclododecyl) and branched alkyl groups with 12 carbon atoms. Alternatively, the alkyl group for R1 may be selected from the group consisting of methyl and ethyl, alternatively methyl. Suitable alkenyl groups for R1 include vinyl, allyl and hexenyl; alternatively vinyl or allyl; and alternatively vinyl. Suitable aryl groups for R1 may include cyclopentadienyl, phenyl, naphthyl, and anthracenyl. Suitable aralkyl groups for R1 include tolyl, xylyl, benzyl, 1 -phenylethyl, and 2- phenylethyl. Alternatively, the aryl group for R1 may be phenyl. Aralkyl groups such as benzyl, 1 -phenylethyl, and 2-phenylethyl, and aralkenyl groups such as styryl, may also be used for R1. Alternatively, each R1 may be selected from the group consisting of methyl and phenyl. Alternatively, each R1 may be methyl.
[0009] Each RE is independently selected from the group consisting of a hydroxyl group and an amino-functional group of formula H2N-RA-, where RA is a divalent hydrocarbon group. Alternatively, each RE may be hydroxyl. Alternatively, each RE may be the amino-functional group of formula H2N-RA-. Each RA is an independently selected divalent hydrocarbon group of 1 to 12 carbon atoms, alternatively 2 to 12 carbon atoms, alternatively 2 to 5 carbon atoms, and alternatively 2 to 3 carbon atoms. The divalent hydrocarbon groups for RA may be linear, branched, or cyclic, or combinations thereof. Suitable divalent hydrocarbon groups for RA include alkylene groups, arylene groups, and combinations thereof (e.g., dialkylarylene groups). The alkylene group is exemplified by ethylene, propylene, or butylene. The arylene group for RA may be arylene group such as phenylene. Alternatively, RA may be a dialkylarylene group such as:
each subscript u is independently 1 to 6, alternatively 1 to 2. Alternatively, each RA may be an alkylene group such as ethylene, propylene, or butylene; alternatively ethylene.
[0010] Each R2 is independently selected from the group consisting of hydrogen and methyl. Alternatively, each R2 may be hydrogen.
[0011] Each RD is an independently selected divalent hydrocarbon group of 2 to 20 carbon atoms, alternatively 2 to 12 carbon atoms, alternatively 3 to 12 carbon atoms, alternatively 4 to 12 carbon atoms, and alternatively 4 to 10 carbon atoms. The divalent hydrocarbon groups for RD may be linear, branched, cyclic, or combinations thereof. Suitable divalent hydrocarbon
groups for RD include alkylene groups, arylene groups, and combinations thereof. Alternatively, each RD may be an alkylene group such as propylene, butylene, hexylene, octylene, decylene, or dodecylene; alternatively each RD may be butylene, hexylene, or decylene. Alternatively, RD may be a branched alkylene group. The arylene group for RD may be arylene group such as phenylene. Alternatively, RD may be a dialkylarylene group such as:
each subscript u is independently 1 to 6, alternatively 1 to 2.
[0012] Subscript a has a value such that 0 < a < 150. Alternatively, subscript a may have a value of 2 to 150, alternatively 14 to 145, alternatively 14 to 143, alternatively 14 to 84, alternatively 14 to 44, and alternatively 42 to 84. Alternatively, subscript a may be 14 to 16, alternatively 42 to 44, and alternatively 82 to 84.
[0013] Subscript b has a value such that 2 < b < 100. Alternatively, subscript b may have a value of 2 to 20, alternatively 2 to 10, and alternatively 3 to 4.
[0014] The copolymer described above may have a number average molecular weight (Mn) of > 1 ,000 g/mole to 250,000 g/mole measured by GPC according to the test method described hereinbelow. Alternatively, the copolymer may have a Mn of 4,000 g/mole to 250,000 g/mole, alternatively 4,000 g/mole to 100,000 g/mole, measured by GPC.
[0015] Alternatively, the copolymer described above may have a weight average molecular weight (Mw) of 2,000 g/mol to 400,000 g/mol. Alternatively, Mw may be 10,000 g/mol to 390,000 g/mol; alternatively 12,000 g/mol to 200,000 g/mol; alternatively 15,000 g/mol to 185,000 g/mol; alternatively 19,000 g/mol to 175,000 g/mol; alternatively 20,000 g/mol to 100,000 g/mol; alternatively 21,000 g/mol to 80,000 g/mol; alternatively 22,000 g/mol to 75,000 g/mol; alternatively 25,000 g/mol to 65,000 g/mol; alternatively 30,000 g/mol to 60,000 g/mol; alternatively 35,000 g/mol to 55,000 g/mol; and alternatively 40,000 g/mol to 50,000 g/mol. The copolymer may be prepared by known methods, and the copolymer may be delivered in an emulsion. The copolymer and the emulsion may be prepared as described in PCT Patent Application Publication WO2023/278918.
[0016] The emulsion may comprise: (I) a liquid continuous phase comprising water, and (11) a discontinuous phase dispersed in the liquid continuous phase, where the discontinuous phase comprises the aminosiloxane ester copolymer described above. The amount of copolymer added to the emulsion can vary and is not limited. However, the amount typically may range from a
weight ratio of copolymer/emulsion of 1 to 70%, alternatively 2 to 60%. Water (and additional starting materials, if present) may constitute the balance of the emulsion to 100%.
Waler
[0017] The water is not generally limited, and may be utilized neat (i.e., absent any carrier vehicles/solvents), and/or pure (i.e., free from or substantially free from minerals and/or other impurities). For example, the water may be processed or unprocessed prior to making the emulsion described herein. Examples of processes that may be used for purifying the water include distilling, filtering, deionizing, and combinations of two or more thereof, such that the water may be deionized, distilled, and/or filtered. Alternatively, the water may be unprocessed (e.g. may be tap water, i.e., provided by a municipal water system or well water, used without further purification). Alternatively, the water may be purified before use to make the emulsion.
Additional Starting Materials
[0018] The emulsion described above may further comprise an additional starting material selected from the group consisting of a surfactant, an acid compound, an acid anhydride, a thickener, a stabilizer, a preservative, and a combination of two or more thereof.
[0019] The copolymer described above may be self-emulsifying (i.e., a separate surfactant is optional). However, when used, the surfactant may be anionic, cationic, nonionic, or amphoteric, or a combination of two or more thereof. The amount of surfactant may be 2 % to 25%, alternatively 2% to 20%, based on combined weights of all starting materials in the emulsion.
Surfactant
[0020] The emulsion comprising (A) aminosiloxane ester copolymer may further comprise a surfactant. The surfactant may be anionic, cationic, nonionic, amphoteric, or a combination thereof. The anionic surfactant may selected from alkali metal sulfosuccinates, sulfonated glyceryl esters of fatty acids, salts of sulfonated monovalent alcohol esters, amides of amino sulfonic acids, sulfonated products of fatty acids nitriles, sulfonated aromatic hydrocarbons, condensation products of naphthalene sulfonic acids with formaldehyde, sodium octahydroanthracene sulfonate, sodium lauryl sulfate, alkali metal alkyl sulfates, alkyl ether sulfates having at least 8 carbon atoms, alkyl aryl ether sulfates, alkylarylsulfonates having at least 8 carbon atoms, alkylbenzenesulfonic acids, salts of alkylbenzenesulfonic acids, sulfuric esters of polyoxyethylene alkyl ether, amine salts or sodium salts or potassium salts of alkylnaphthylsulfonic acid, and combinations thereof. Suitable anionic surfactants are commercially available from various sources including sodium lauryl sulfate, which is available from Pilot under the tradename CALIMULSE™ SLS. Other anionic surfactants commercially
available from The Dow Chemical Company, include alkyldiphenyloxide disulfonate salts, which are available under the tradename DOWFAX™; dioctyl sulfosuccinates, which are available under the tradename TRITON™ GR; phosphate esters, which are available under the tradename TRITON™ H-55, H-65, QS-44, OR XQS-20; sulfates and sulfonates, which are available under the tradename TRITON™ QS-15 and TRITON™ XN-45S.
[0021] The cationic surfactant may be selected from dodecylamine acetate, octadecyl amine acetate, acetates of the amines of tallow fatty acids, homologues of aromatic amines having fatty acids, fatty amides derived from aliphatic diamines, fatty amides derived from aliphatic diamines, fatty amides derived from disubstituted amines, derivatives of ethylene diamine, quaternary ammonium compounds, salts of quaternary ammonium compounds, alkyl trimethylammonium hydroxides, dialkyldimethylammonium hydroxides, coconut oil, methylpolyoxyethylene cocoammonium chloride, dipalmitoylethyl hydroxyethylammonium methosulfate, amide derivatives of amino alcohols, amine salts of long chain fatty acids, and combinations thereof. Cationic surfactants are commercially available from various sources including dialkylmethyl quaternary ammonium compounds (e.g., cetrimonium chloride) under the tradename ARQUAD™ from Akzo Nobel; ADOGEN™ cationic surfactants from Evonik; TOMAH™ cationic surfactants from Tomah Products, Inc. of Milton, Wisconsin, USA; and VARIQUAT™ cationic surfactants from Sea-Land Chemical Company of Westlake, Ohio, USA.
[0022] The nonionic surfactant may be selected from alkylphenol alkoxylates, ethoxylated and propoxylated fatty alcohols, alkyl polyglucosides and hydroxyalkyl polyglucosides, sorbitan derivatives, N- alkylglucamides, alkylene oxide block copolymers, such as block copolymers of ethylene oxide, propylene oxide and/or butylene oxide, fatty alcohol polyglycolethers, polyhydroxy and polyalkoxy fatty acid derivatives, amine oxides, silicone polyethers, various polymeric surfactants. Nonionic surfactants are commercially available, for example, alkylphenol alkoxylates are available under the tradename ECOSURF™ EH; secondary alcohol ethoxylates, nonylphenol ethoxylates, and ethylene oxide/propylene oxide copolymers are commercially available under the tradename TERGITOL™; and specialty alkoxylates such as amine ethoxylates and octylphenol ethoxylates are available under the tradename TRITON ™, all from The Dow Chemical Company. Alternatively, the nonionic surfactant may be, e.g., trideceth-6 or trideceth-12, which are available under the tradename SYNPERONIC™ from Croda or LUTENSOL™ from BASF. Alternatively, the nonionic surfactant may be e.g., a fatty alcohol polyglycol ether such as GENAPOL™ UD 050, and GENAPOL™ UDI 10, which are commercially available from Clariant of Frankfurt, Germany.
[0023] Alternatively, the nonionic surfactant may comprise, or may be, a silicone polyether
(SPE). The silicone polyether as an emulsifier may have a rake type structure wherein the polyoxyethylene or polyoxy ethylene-polyoxypropylene copolymeric units are grafted onto the siloxane backbone, or the SPE can have an ABA block copolymeric structure wherein A represents the polyether portion and B the siloxane portion of an ABA structure. Suitable silicone polyethers include DOWSIL™ OFX-5329 Fluid from The Dow Chemical Company. Alternatively, the nonionic surfactant may be selected from polyoxyalkylene-substituted silicones, silicone alkanolamides, silicone esters and silicone glycosides. Such silicone-based surfactants are known in the art, and have been described, for example, in U.S. Patent 4,122,029 to Gee et al., U.S. Patent 5,387,417 to Rentsch, and U.S. Patent 5,811,487 to Schulz et al.
[0024] Suitable amphoteric surfactants include betaines such as alkyl(C12-14)betaine, cocoamidopropylbetaine, cocoamidopropyldimethyl-hydroxysulphobetaine, dodecylbetaine, hexadecylbetaine, and tetradecylbetaine; sultaines such as cocamidopropylhydroxysultaine; lecithin; hydrogenated lecithin; cocoamphodiacetates; cocoiminodipropionate; and dodecyliminodipropionate.
[0025] Alternatively, the surfactant in the aqueous copolymer emulsion may be a nonionic surfactant. Alternatively, the surfactant may be an organic surfactant. Alternatively, the surfactant may be both organic and nonionic.
[0026] The aqueous copolymer emulsion may be formed as a water-in-oil emulsion (w/o), which contains a water-in-oil surfactant, (which may subsequently inverted by addition of more water). The water-in-oil surfactant may be nonionic and may be selected from polyoxyalkylene- substituted silicones, silicone alkanolamides, silicone esters and silicone glycosides, as described above. Alternatively, when the emulsion is an oil-in-water (o/w) emulsion, it may include nonionic surfactants known in the art to prepare o/w emulsions. Suitable nonionic surfactants for this embodiment are exemplified by the polyoxyethylene alkyl ethers, polyoxyethylene alkylphenol ethers, polyoxyethylene lauryl ethers, polyoxyethylene sorbitan monooleates, polyoxyethylene alkyl esters, polyoxyethylene sorbitan alkyl esters, polyethylene glycol, polypropylene glycol, diethylene glycol, ethoxylated trimethylnonanols, and polyoxyalkylene glycol modified poly siloxane surfactants, as described above.
Acid Compound
[0027] The acid compound may optionally be added to the aqueous copolymer emulsion for adjusting pH. Suitable acids include acetic acid, formic acid, propionic acid, and combinations thereof. Suitable acids for adjusting pH are disclosed, for example, in U.S. Patent 6180117.
Method for Making the Aqueous Copolymer Emulsion
[0028] Emulsions may be prepared in a batch, semi-continuous, or continuous process using conventional equipment. For example, mixing the starting materials to form the emulsion may
occur, for example using, batch equipment with high-shear and high-speed dispersers include those made by Charles Ross & Sons (NY), Hockmeyer Equipment Corp. (NJ); batch mixing equipment such as those sold under the tradename Speedmixer™; batch equipment with high shear actions include Banbury-type (CW Brabender Instruments Inc., NJ) and Henschel type (Henschel mixers America, TX). Illustrative examples of continuous mixers/compounders include extruders, such as single-screw, twin-screw, and multi-screw extruders, co-rotating extruders, such as those manufactured by Krupp Werner & Pfleiderer Corp (Ramsey, NJ), and Leistritz (NJ); twin-screw counterrotating extruders, two-stage extruders, twin-rotor continuous mixers, dynamic or static mixers or combinations of these equipment.
[0029] The starting materials described above may be combined under any suitable conditions for forming an emulsion. For example, to simplify the mixing process and keep the emulsion viscosity low while handling, any acid compound may be added at the end of the method, i.e., once the desired dilution level is reached.
[0030] The emulsion of the aminosiloxane ester copolymer described above may be used in an amount sufficient to provide 0.1% to 50%, alternatively 0.1% to 10%, alternatively 0.5% to 10%, and alternatively 1% to 5%, of (A) the aminosiloxane ester copolymer based on combined weights of all the starting materials in the aqueous coating composition described herein.
(B) Polyurethane Binder
[0031] Starting material (B) in the aqueous coating composition described herein is a polymeric binder (dry polymer). The polymeric binder may be (Bl) a polyurethane or (B2) an acrylic polymer. The polyurethane may be delivered in an aqueous dispersion. The aqueous polyurethane dispersion used herein to prepare the aqueous coating composition can be an externally stabilized polyurethane dispersion or an internally stabilized polyurethane dispersion. “Internally stabilized polyurethane dispersion” herein refers to a polyurethane dispersion that is stabilized through the incorporation of ionically or nonionically hydrophilic pendant groups within the polyurethane particles dispersed in the liquid medium. Examples of nonionic internally stabilized polyurethane dispersions are described in U.S. Patents 3905929 and 3920598. Ionic internally stabilized polyurethane dispersions are known and are described in U.S. Patent 6231926. Typically, dihydroxyalkylcarboxylic acids such as described in U.S. Patent 3412054 are used to make anionic internally stabilized polyurethane dispersions. A common monomer used to make an anionic internally stabilized polyurethane dispersion is dimethylolpropionic acid (DMPA).
[0032] The polyurethane may be prepared by polymerization of monomers selected from polyisocyanates having 2 or more isocyanate functionalities and having 4 to 40 carbon atoms, polyols such as diols, monomers bearing at least one isocyanate group or at least one isocyanate
reactive group and which in addition bear at least one hydrophilic group or potentially hydrophlic group, and optionally one or more compounds having reactive groups comprising alcoholic hydroxyl groups, primary or secondary amino groups or isocyanate groups.
[0033] Suitable polyisocyanates include conventional aliphatic, cycloaliphatic, araliphatic and aromatic isocyanates. The polyisocyanate may be selected from the group of diphenylmethane diisocyanates (“MDI”), polymeric diphenylmethane diisocyanates (“pMDI”), toluene diisocyanates (“TDI”), hexamethylene diisocyanates (“HD I”), dicyclohexylmethane diisocyanates (“HMD I”), isophorone diisocyanates (“IPDI”), cyclohexyl diisocyanates (“CHDI”), naphthalene diisocyanate (“NDI’’), phenyl diisocyanate (“PDI”), tetramethylene diisocyanate (“TMDI”), and combinations thereof. The polyisocyanate may have formula OCN — R — NCO, wherein R is an alkyl moiety, an aryl moiety, or an arylalkyl moiety. Alternatively, the polyisocyanate can include any number of carbon atoms described above, alternatively from 4 to 20 carbon atoms.
[0034] Specific examples of suitable polyisocyanates include: alkylene diisocyanates with 4 to 12 carbons in the alkylene radical such as 1,12-dodecane diisocyanate, 2-ethyl-l,4- tetramethylene diisocyanate, 2-methyl-l,5-pentamethylene diisocyanate, 1,4-tetramethylene diisocyanate and preferably 1,6-hexamethylene diisocyanate; cycloaliphatic diisocyanates such as 1,3- and 1 ,4-cyclohexane diisocyanate as well as any mixtures of these isomers, 1-isocyanato- 3,3,5-trimethyl-5-isocyanatomethylcyclohexane, 2,4- and 2,6-hexahydrotoluene diisocyanate as well as the corresponding isomeric mixtures, 4,4'- 2,2'-, and 2,4'-dicyclohexylmethane diisocyanate as well as the corresponding isomeric mixtures, and aromatic diisocyanates and polyisocyanates such as 2,4- and 2,6-toluene diisocyanate and the corresponding isomeric mixtures, 4,4'-, 2,4'-, and 2,2'-diphenylmethane diisocyanate and the corresponding isomeric mixtures, mixtures of 4,4'-, 2,4'-, and 2,2-diphenylmethane diisocyanates and polyphenylenepolymethylene polyisocyanates, as well as mixtures of MDI and toluene diisocyanate (TDI). Alternatively, the polyisocyanate may comprise IPDI. Alternatively, the polyurethane may be made from one or more diisocyanates, such as IPDI or TMDI and one or more polyols, such as a polyether polyol, polycarbonate polyol, or polyester polyol, e.g., having a molecular weight (Mw) of 5,000 or less, or of 2,000 or less. Such polyols may be linear and may have have two hydroxyl groups, one at each end.
[0035] Suitable polyols include polyester polyols, which are reactive with the isocyanate described above include, but are not limited to, hydroxyl-functional reaction products of polyhydric alcohols, such as ethylene glycol, propylene glycol, diethylene glycol, 1,4- butanediol, neopentylglycol, 1,6-hexanediol, cyclohexane dimethanol, glycerol, trimethylolpropane, pentaerythritol, sucrose, or polyether polyols or mixtures of such polyhydric
alcohols, and polycarboxylic acids, particularly dicarboxylic acids or their ester-forming derivatives, for example succinic, glutaric and adipic acids or their dimethyl esters, sebacic acid, phthalic anhydride, tetrachlorophthalic anhydride, dimethyl terephthalate or mixtures thereof. Polyester polyols obtained by the polymerization of lactones, e.g. caprolactone, in conjunction with a polyol, or of hydroxy carboxylic acids, e.g. hydroxy caproic acid, may also be used. In certain embodiments, the polyol comprises a mixture of polyester and polyether polyols. “Externally stabilized polyurethane dispersion” herein refers to a polyurethane dispersion that fails to have an ionic or nonionic hydrophilic pendant groups and thus requires the addition of a surfactant to stabilize the polyurethane dispersion. The surfactant can be those described in the copolymer emulaion above. Examples of externally stabilized polyurethane dispersions are described in U.S. Patents 2,968,575; 5,539,021; 5,688,842 and 5,959,027.
[0036] Alternatively, the polyurethane dispersion may be an internally stabilized polyurethane dispersion. Alternatively, the polyurethane dispersion may comprise an aqueous polyurethane dispersion based on isophorone diisocyanate and polyester polyol, stabilized by carboxylate groups in the polyurethane backbone. The polyurethane may have glass transition temperature of -44 °C. Suitable polyurethane dispersions for use in preparing the aqueous coating composition are known in the art and are commercially available, for example, BAYDERM™ polyurethane dispersions such as BAYDERM™ 91UD, which is available from The Dow Chemical Company of Midland, Michigan, USA. Alternatively, the polyurethane dispersion may be as described in U.S. Patent 11,518,905 to Lenoble et al.
[0037] Other representative examples of applicable commercial polyurethane dispersion products include: PERMUTEX™ from Stahl Polymer, HAUTHANE™ L-3121 from C. L. Hauthaway & Sons Corp, and PRIMAL™ BINDER available from The Dow Chemical Company and polyurethanes from Ableridingk Boley, Inc. Other polyurethane dispersions can be prepared by methods conventional in the art. See, for example, the methods described in P. Pieterich, Aqueous Emulsion, Dispersion and Solutions of Polyurethanes; Synthesis and Properties in Progress in Organic Coatings 9 (1981) 281-340. See also: US7232859, US2004/0167252 and US2011/0112245. Such polyurethanes are commonly prepared by reacting an organic polyisocyanate(s) with an organic compound(s) containing isocyanatereactive groups, particularly a polyol. The reaction may be carried out in the presence of a catalyst such as organic tin compounds and/or tertiary amines. The polyurethanes are made into aqueous dispersion by conventional means and may be anionic salt functional, non-ionic or anionic polyurethane dispersions. In one embodiment, the polyurethane dispersion may be an anionic polyurethane dispersion prepared by reacting one or more polyol with an organic compound having at least one acid group and at least two active hydrogen functionalities and a
polyisocyanate. Suitable organic compounds having at least one acid group and at least two active hydrogen functionalities include, for example, 2,2-dimethylolacetic acid and 2,2- dimethylolpropionic acid. Examples of acid groups suitable for the organic compound include, carboxylic acid, sulfonic acid, phosphoric, phosphonic acid and the like. (C) Surfactant [0038] Starting material (C) in the aqueous coating composition is a surfactant. The surfactant may be introduced into the aqueous coating composition with starting material (A) the aminosiloxane ester copolymer and (B) the polymeric binder, both of which may be delivered in aqueous emulsions or dispersions, as described above, and/or the surfactant may be added separately, or both. Alternatively, the surfactant in the aqueous coating composition may comprise an organic surfactant. Alternatively, the surfactant in the aqueous coating composition may be a nonionic surfactant. Alternatively, the surfactant in the aqueous coating composition may be an organic, nonionic surfactant. Said organic, nonionic surfactant may have a relatively high hydrophobic - lipophobic balance (HLB) value. For example, organic, nonionic surfactants are as described above in the emulsion for starting material (A), the aminosiloxane ester copolymer, and include those which are commercially available such as (i) 2,6,8-trimethyl-4- nonyloxy polyethylene oxy ethanols (6EO) and (10EO) sold under the names TERGITOL™ TMN-6 and TERGITOL™ TMN-10; (ii) the Ci 1-15 secondary alkyl polyoxyethylene ethers (e.g., Ci 1-15 secondary alcohol ethoxylates 7EO, 9EO, and 15EO sold under the names TERGITOL™ 15-S-7, TERGITOL™ 15-S-9, and TERGITOL™ 15-S-15, which has HL value 15.4), other Cuis secondary alcohol ethoxylates sold under the tradenames ECOSURF™ EH-40 and TERGITOL™ 15-S-12, TERGITOL™ 15-S-30, and TERGITOL™ 15-S-40, by the Dow Chemical Company, of Midland, Michigan, USA; octylphenyl polyoxyethylene (40) ether sold under the name TRITON™ X405 by the Dow Chemical Company; (iii) nonylphenyl polyoxyethylene (10) ether sold under the name MAKON™ 10 by the Stepan Company; (iv) ethoxylated alcohols sold under the name Trycol 5953 by Henkel Corp./Emery Group, of Cincinnati, Ohio, USA; (v) ethoxylated alcohols sold under the name BRIJ™ L23 (with HLB value of 16.9) and BRU™ L4 (with HLB value of 9.7) by Croda Inc. of Edison, New Jersey, USA, (vi) polyoxyethylene 23 lauryl ether (Laureth-23) sold commercially under the trademark BRU™ 23; and RENEX™ 30, a polyoxyethylene ether alcohol; (vii) alkyLoxo alcohol poly glycol ethers such as GENAPOL™ UD 050 (with HLB value of 11.4), and GENAPOL™ UDI 10 (with HLB value of 14.4), (viii) alkyl polyethylene glycol ether based on ClO-Guerbet alcohol and ethylene oxide such as LUTENSOL™ XP 79, and (ix) alkyl poly glycosides, such as those sold under the trade name Glucopon™ by BASF, and alkyl glucosides such as decyl glucoside, lauryl glucoside, and coco-glucoside, which are sold under the trade name EcoSense™ by The Dow Chemical Company of Midland, Michigan, USA. Other commercially
available nonionic surfactants include TERGITOL™ 15-S-5, also from The Dow Chemical Company, which has an HLB value of 10.5; Lutensol XP 50 with an HLB value of 10, Lutensol XP 79 (an alcohol ethoxylate), Lutensol XP 100 (an alcohol ethoxylate), and Lutensol XP 140 with an HLB value of 16, each of which is available from BASF. The amount of surfactant in the aqueous coating composition may be, for example, 0.03% to 25%, alternatively 0.03% to 4% based on combined weights of all the starting materials in the aqueous coating composition described herein.
(D) Water
[0039] Starting material (D) in the aqueous coating composition is water. All or a portion of the water may be introduced into the aqueous coating composition with starting material (A) the aminosiloxane ester copolymer and (B) the polyurethane binder, both of which are typically delivered in aqueous emulsions or dispersions, as described above. The water is not specifically restricted and may be as described above in the emulsion of starting material (A) the aminosiloxane ester copolymer. The amount of water in the aqueous coating composition is not specifically restricted. The amount of water may be > 0 to 70%, alternatively > 0 to 65%, alternatively 40% to 70%, based on combined weights of all the starting materials in the aqueous coating composition described herein.
[0040] One or more other optional additional starting materials may be included in the aqueous coating composition, as long as properties of the aqueous coating composition and performance of the coating made from the composition are not compromised. Suitable other optional starting materials include (E) a biocide, (F) a pH modifier, (G) a pigment, (H) a thickener, a rheology modifier, a matting agent or duller (e.g., silica), an antifoam, a water repellent additive, an antiblocking additive, an abrasion resistance additive, an antioxidant, a UV absorber, a photo-stabilizer, an antistatic agent, a preservative (other than the biocide described above), a plasticizer, a flame retardant, a wetting agent (other than the surfactant described above), an opacifier, an extender, a plasticizer, and a combination of two or more thereof.
(E) Biocide
[0041] Starting material (E) is a biocide. The biocide is optional and may be added to the aqueous coating composition to ensure that the coating prepared from the aqueous coating composition provides protection against microbial attack during storage and transportation. The biocide is exemplified by a fungicide, an herbicide, a pesticide, an antimicrobial agent, or a combination thereof. Alternatively, the aqueous coating composition may comprise a fungicide, an antimicrobial agent, or a combination thereof. The amount of biocide will vary depending on factors including the type of biocide selected and the benefit desired. However, the amount of biocide may range from greater than 0 % to 5 %, alternatively from 1 ppm to 1500 ppm, each
based on the weight of all starting materials in the aqueous coating composition. Biocides are known in the art and are commercially available. For example, PREVENTOL™ C40-L, which is a preservative based on p-chloro-m-cresol (PCMC); BIOBAN™ O 45 Antimicrobial;
Preventol™ BIT 20 N; Preventol™ BM 5; Preventol™ CMK 40; and Preventol™ D 7 are biocides suitable for leather treatment that are commercially available from Lanxess of Pittsburgh, Pennsylvania, USA.
(F) pH modifier
[0042] Starting material (F) is a pH modifier. The pH modifier is optional and may be added to change pH of the aqueous coating composition. Without wishing to be bound by theory, it is thought that the pH modifier may be used to reduce particle size of the aqueous coating composition (which is in the form of an emulsion) as compared to a composition that does not contain the pH modifier. The pH modifier may be an acid as described above for the aqueous copolymer emulsion to deliver (A) the aminosiloxane ester copolymer. Alternatively, the pH modifier may be a buffer such as sodium carbonate, sodium bicarbonate, and a combination thereof. The amount of the pH modifier depends on various factors such as the type of rheology modifier present, if any, however the amount of pH modifier may be 0 to 5%, alternatively > 0 to 5%, based on combined weights of all starting materials in the aqueous coating composition.
(G) Pigment
[0043] Starting material (G) is a pigment. The pigment is optional and may be added to the aqueous coating composition to impart a desired color to a coating prepared from the aqueous coating composition. Suitable pigments are known in the art and are commercially available. Pigments include carbon black and titanium dioxide. The amount of pigment may be > 0% based on based on combined weights of all the starting materials combined to prepare the aqueous coating composition described herein.
[0044] When selecting starting materials to make the aqueous coating composition described above, there may be overlap between types because certain starting materials described herein may have more than one function. For example, certain particulates may be useful as pigments and as flame retardants, e.g., carbon black. When adding additional starting materials to the aqueous coating composition, the additional starting materials are distinct from one another and from starting materials (A), (B), (C) and (D) described above. Examples of suitable optional additional starting materials and their amounts may be found for example, in US Patents 9200404, 10100377, and 11518905.
Method of Making the Aqueous Coating Composition
[0045] The aqueous coating composition may be prepared by any convenient means using any convenient equipment. The aqueous coating composition may be prepared by a method comprising:
1) mixing starting materials comprising a) an aqueous copolymer emulsion comprising
(I) a liquid continuous phase comprising water, and
(II) a discontinuous phase dispersed in the liquid continuous phase, where the discontinuous phase comprises (A) the aminosiloxane ester copolymer, where the aqueous copolymer emulsion further comprises a surfactant, and water; b) an aqueous binder emulsion comprising (B) the polymeric binder and water. The method may optionally further comprise one or more additional steps before step 1), e.g., dispersing a starting material comprising a pigment in water before step 1), thereby preparing an aqueous pigment dispersion, and mixing the aqueous pigment dispersion with the starting materials in step 1). The method may optionally further comprise one or more additional steps after step 1), e.g. the method may further comprise removing agglomerated particles after step 1) and/or the method may further comprise 2) adding an additional starting material selected from the group consisting of (E) a biocide, (F) a pH modifier, (G) a pigment, (H) a thickener, a rheology modifier, a matting agent or duller (e.g., silica), an antifoam, a water repellent additive, an antiblocking additive, an abrasion resistance additive, an antioxidant, a UV absorber, a photostabilizer, an antistatic agent, a preservative (other than the biocide described above), a plasticizer, a flame retardant, a wetting agent (other than the surfactant described above), an opacifier, an extender, a plasticizer, and a combination of two or more thereof. In addition, the method may optionally further comprise adding additional water for dilution, for example, for use at remote site to reduce the total solids of the aqueous coating composition to a desired range. Accordingly, the aqueous coating composition may be shipped in any stable concentrated form.
[0046] Mixing the starting materials in step 1) (and any optional additional steps) may be performed by any convenient means, such as mixing optionally under shear, using the equipment and methods described above for making the emulsion of (A) the aminosiloxane ester copolymer. Alternatively, simple mixing may be performed to mix the starting materials in step 1), e.g., shear is not required. The aqueous coating composition prepared described above may be combined with a crosslinker and then used to treat substrates comprising leather.
Crosslinker [0047] A crosslinker and the aqueous coating composition are combined shortly before use (e.g., shortly before applying the resulting crosslinkable coating composition to a substrate). For
example, the aqueous coating composition described herein may be provided in a multiple-part system comprising a base part and a curing agent part. The base part comprises the aqueous coating composition comprising starting materials (A), (B), (C), and (D), and optionally one or more additional starting materials, as described above. The curing agent part comprises a crosslinker. Suitable crosslinkers for polyurethanes are known in the art and include melamine resins, polyaziridine resins, aminoplast resins, amide- and amine-formaldehyde resins, and polyisocyanates (which may be blocked or unblocked polyisocyanates). Polyisocyanates may contain free isocyanate groups based on aliphatic, cycloaliphatic, araliphatic and/or aromatic isocyanates. The crosslinker may be water dispersible. Alternatively, the crosslinker may comprise a polyisocyanate, alternatively an aliphatic polymeric isocyanate. The crosslinking agent may be used in an amount of 0.1% or more, and up to 20%, for example, from 1% to 10%, based on combined weights of base part and curing agent part. Suitable crosslinkers are commercially available and include BINDER LS-3486-HS Crosslinker, which is supplied as a solution with viscosity < 50 cP (measured by a Brookfield LVT at 60 rpm) containing a reactive aliphatic polyisocyanate resin (50-52%); and BINDER LS-3492 Crosslinker, which is supplied as a solution with viscosity < 50 cP (measured by a Brookfield LVT at 60 rpm) containing a reactive aliphatic polyisocyanate resin (49%-51%) supplied in ethyl 3-ethoxypropionate (49%- 51%), both of which are commercially available from The Dow Chemical Company. Other commercially available crosslinkers include PZ-28 Polyfunctional Aziridine from Poly Aziridine LLC; polymeric carbodiimides from Angus Chemical Company such as Zoldine XL-29SE; such as Picassian brand or Permutex brand such as the Permutex XR 5508 from Stahl; and NeoAdd™ PAX from DSM. The system may be provided in a kit, where the kit further comprises instructions for combining the base part and the curing agent part and optionally for using the resulting crosslinkable coating composition for treating substrates comprising leather, as follows.
Method for Treating Leather
[0048] The aqueous coating composition prepared as described above may be used to treat substrates comprising leather (i.e., natural or synthetic leather). For example, a method for treating a substrate comprises: pre-i) combining the base part comprising the aqueous coating composition and the curing agent part comprising crosslinker, described above, thereby preparing a crosslinkable coating composition; i) applying the crosslinkable coating composition to a substrate comprising leather; and ii) drying the crosslinkable coating composition to remove water, thereby forming a coating on the substrate. The base part and the curing agent part may be combined by any
convenient means, such as simple mixing, e.g., when the crosslinker is supplied in water. The crosslinkable coating composition may be applied to the substrate by any convenient method. For example, the crosslinkable coating composition may be applied on the substrate by a method selected from the group consisting of spraying, knife coating, roll coating, casting, drum coating, dipping, gravure coating, bar coating, screen coating, curtain coating, brush coating, and combinations thereof. The amount of the crosslinkable coating composition applied on the substrate is not specifically restricted, and may have a wet coating thickness of 10 pm to 100 pm, which may correspond to a dry coating thickness of 2 pm to 70 pm. Drying may be performed by any convenient method, such as air drying or heat drying the coated substrate. The conditions for heat drying depend on various factors including the substrate selected. For example, when the substrate comprises natural leather, the heat drying temperature may be < 120 °C. Alternatively, for synthetic leather substrates, the heat drying temperature may be < 180 °C, alternatively < 150 °C for a time sufficient to remove most or all of the water. The method may optionally further comprise iii) repeating steps i) and ii) one or more times to increase the thickness of the coating on the substrate. The thickness of the coating to be formed on the substrate is not specifically restricted.
[0049] The aqueous coating composition and method for treating a substrate described herein may be used to provide coatings on leather, which includes both natural and synthetic leathers, such as for automobile and other motor vehicle applications (e.g., as coatings on armrests, dashboards, seating, and other interior components); clothing such as coats, pants, flight jackets, motorcycle clothing, shoes, and gloves; luggage or handbags; accessories such as belts, wallets, and datebooks; furniture; or saddles for bicycles or motorcycles.
EXAMPLES
[0050] The following examples are provided to illustrate the invention to one skilled in the art and are not to be interpreted to limit the scope of the invention set forth in the claims. Starting materials used in these examples are described below in Tables 1 and 2.
[0051] In this Reference Example 1 , aqueous copolymer emulsions with the starting materials in amounts shown below in Table 1 were prepared as described in PCT Patent Application Publication WO2023/278918. In a plastic dental cup, aminosiloxane ester copolymer, surfactants and inversion water were added and mixed by a high shear mixer at 3500 rpm for 30 sec (dental mixer), then the resulting thick phase was diluted by water, and the acetic acid solution added at the end of the process.
[0052] In this Reference Example 2, an aqueous emulsion of a commercially available aminofunctional polydiorganosiloxane was prepared using the starting materials in amounts shown below in Table 2.
[0053] In this Reference Example 3, aqueous coating compositions were prepared as follows. Aqueous emulsions and other starting materials except for thickener (if present) under low shear stirring. The resultant mixture was then filtered to remove agglomerated particles. Finally, thickener (if present) was added into the mixture. The starting materials in these aqueous coating compositions are described in Table 4 and the amounts of each starting material are
shown below in Table 5 for compositions with a polyurethane binder and Table 7 for an acrylic polymer binder.
[0054] In this Reference Example 4, the aqueous coating compositions were used to prepare coating on substrates and tested, as follows: To form the aqueous coating compositions of the present invention, the aqueous copolymer emulsions described above in Reference Example 3 and Binder 1 described below in Table 4 were mixed together using a laboratory stirrer under low shear (IKA lab mixer, 500 rpm, 5 min).
[0055] The second step was to blend the emulsion described above with Crosslinker 1 under low shear stirring (IKA lab mixer, 500 rpm, 5 min). The resulting crosslinkable coating composition was applied using a 60, um cylindrical film applicator from BYK upon a sheet of a sealed opacity chart (form 2A obtained from Leneta Company). The top of the chart was black and the bottom was white with an overall dimensions of 5-1/2 x 10 in (140 x 254 mm). The coated chart was then dried in a conventional oven for 2 minutes at 80 °C.
[0056] The coefficient of friction of each coating was measured between felt (spring) against coated sample (carriage) on a Ziegler Instruments SSP-04 stick-slip tester with a normal force of F = 2.0 N, a displacement of 40 mm and a velocity of v = 6.0 mm/s. The test was run over 10 cycles and data analysis was performed with the Ziegler Instruments software. The felt was sourced from Ideal felt, Brussels with reference: Type: Needle-Tex - IF-202-S1.
Table 4 - Starting Materials for Crosslinkable Coating Compositions Prepared According to
[0057] In the examples reported in the Table 5 (coating) the aqueous copolymer emulsion was first diluted to get 28% of active (copolymer) through the addition of water before use in making the aqueous coating composition in Table 5 (to 100g of emulsion 31.4 g of water was added).
[0058] The results in Table 6 show that in coatings prepared with polyurethane binders, addition of the aminosiloxane ester copolymer (in working examples Coat 4, Coat 5, and Coat 6) provided the benefits of decreasing static coefficient of friction and dynamic coefficient of friction for all conditions tested when compared to a coating that did not contain the aminosiloxane ester copolymer (i.e., Comparative Example 1, Coat 1). Furthermore, each
coating that contained the aminosiloxane ester copolymer outperformed a coating containing a commercially available aminofunctional siloxane instead of the aminosiloxane ester copolymer (as shown in Comparative Examples 2 and 3, Coat 2 and Coat 3, respectively).
INDUSTRIAL APPLICABILITY
[0059] Using an aminosiloxane ester copolymer as additive at a low loading in an aqueous coating composition including a polyurethane water-based binder provided beneficial properties in the coatings prepared therewith, such as low CoF. Without wishing to be bound by theory, it is thought that using the aminosiloxane ester copolymer may further provide one or more of the following benefits to the coating: high contact angle with water, low stick slip performance, and low leaching. To improve compatibility with the polyurethane binder, the aminosiloxane ester copolymer may be delivered as an emulsion using a nonionic surfactant. Furthermore, the aminosiloxane ester copolymer may provide one or more benefits over other silicone leather treatment composition active ingredients. For example, the aminosiloxane ester copolymer has low cyclic polyorganosiloxane content (< 0.05 % octamethylcyclotetrasiloxane), stability after aging as evidenced by low cyclics content after aging, little or no crosslinking, and potential for biodegradability.
[0060] Without wishing to be bound by theory, it is thought that using a copolymer in which subscript a is at least 14, alternatively at least 42, alternatively 42 to 150, and alternatively 42 to 84 may provide improved static and/or dynamic coefficient of friction to a coating prepared as described herein as compared to a coating prepared from a comparative composition excluding the aminosiloxane ester copolymer or containing a commercially available aminofunctional siloxane.
DEFINITIONS AND USAGE OF TERMS
[0061] All amounts, ratios, and percentages herein are by weight, unless otherwise indicated by the context of the specification. The articles ‘a’, ‘an’, and ‘the’ each refer to one or more, unless otherwise indicated by the context of specification. The singular includes the plural unless otherwise indicated by the context of the specification. The SUMMARY and ABSTRACT are hereby incorporated by reference. The amounts of all starting materials in a composition total 100%. The transitional phrases “comprising”, “consisting essentially of’, and “consisting of’ are used as described in the Manual of Patent Examining Procedure Ninth Edition, Revision 08.2017, Last Revised January 2018 at section §2111.03 I., IL, and III. Any feature or aspect of the invention may be used in combination with any other feature or aspect recited herein. The abbreviations used herein have the definitions in Table 7.
Table 7 - Abbreviations
Claims
1. An aqueous coating composition comprises:
(A) an aminosiloxane ester copolymer comprising formula
where each R1 is an independently selected monovalent hydrocarbon group of 1 to 12 carbon atoms, each RE is independently selected from the group consisting of hydroxyl and an amino-functional group of formula H2N-RA-, each RA is an independently divalent hydrocarbon group of 1 to 12 carbon atoms, each R2 is independently selected from the group consisting of hydrogen and methyl, each RD is an independently selected divalent hydrocarbon group of 2 to 20 carbon atoms, each subscript a independently has a value such that 0 < a < 150, and subscript has a value such that 1 < b < 100,
(B) a polyurethane binder;
(C) a surfactant; and
(D) water.
2. The aqueous coating composition of claim 1, where the polyurethane is based on isophorone diisocyanate and a polyester polyol, stabilized by a carboxylate group existing in the polyurethane backbone.
3. The aqueous coating composition of claim 1, further comprising an additional starting material selected from the group consisting of a biocide; a pH modifier; a pigment, a thickener, a rheology modifier, a matting agent or duller, an antifoam, a water repellent additive, an antiblocking additive, an abrasion resistance additive, an antioxidant, a UV absorber, a photostabilizer, an antistatic agent, a preservative, a plasticizer, a flame retardant, a wetting agent, an opacifier, an extender, a plasticizer, and a combination of two or more thereof.
4. The aqueous coating composition of claim 1 or claim 2, where the aqueous coating composition comprises:
0.1 weight% to 50 weight% of (A) the aminosiloxane ester copolymer,
30 weight% to 70 weight% of (B) the polymeric binder,
0.03 weight % to 25 weight % of (C) the surfactant, and
> 0 to 70 weight % of (D) the water.
5. The aqueous coating composition of any one of claims 1 to 4, further comprising:
1 ppm to 1500 ppm, by weight, based on combined weights of all starting materials int he composition, of (E) the biocide,
> 0 to 5 weight % of (F) the pH modifier from 0 to 5, and
> 0 to 1 weight % of a rheology modifier.
6. A crosslinkable coating composition comprising the aqueous coating composition of any one of claims 1 to 5 and a crosslinker.
7. A method for preparing the aqueous coating composition of any one of claims 1 to 5, where the method comprises:
1) mixing starting materials comprising a) an aqueous copolymer emulsion comprising
(I) a liquid continuous phase comprising water, and
(II) a discontinuous phase dispersed in the liquid continuous phase, where the discontinuous phase comprises (A) the aminosiloxane ester copolymer, where the aqueous emulsion further comprises the surfactant, the water, the biocide, and the pH modifier; b) an aqueous binder emulsion comprising the polymeric binder and water.
8. The method of claim 7, where the method further comprises dispersing a starting material comprising (G) a pigment in (D) water before step 1), thereby preparing an aqueous pigment dispersion, and mixing the aqueous pigment dispersion with the starting materials in step 1).
9. The method of claim 7 or claim 8, further comprising removing agglomerated particles after step 1).
10. The method of any one of claims 7 to 9, further comprising: 2) adding an additional starting material selected from the group consisting of a biocide; a pH modifier; a pigment, a thickener, a rheology modifier, a matting agent or duller, an antifoam, a water repellent additive, an antiblocking additive, an abrasion resistance additive, an antioxidant, a UV absorber, a photostabilizer, an antistatic agent, a preservative, a plasticizer, a flame retardant, a wetting agent, an opacifier, an extender, a plasticizer, and a combination of two or more thereof..
11. The method of any one of claims 7 to 10, further comprising: 3) combining the aqueous coating composition and a crosslinker.
12. A method for treating leather, wherein the method comprises: i) applying the crosslinkable coating composition of claim 6 to a substrate comprising leather; and ii) drying the crosslinkable coating composition, thereby forming a coating on the substrate.
13. The method of claim 12, further comprising: iii) repeating steps i) and ii) one or more times to increase thickness of the coating.
14. A system comprising:
I) an aqueous coating composition comprising
(A) an aminosiloxane ester copolymer comprising formula
where each R1 is an independently selected monovalent hydrocarbon group of 1 to 12 carbon atoms, each RE is independently selected from the group consisting of hydroxyl and an amino-functional group of formula H2N-RA-, each RA is an independently divalent hydrocarbon group of 1 to 12 carbon atoms, each R2 is independently selected from the group consisting of hydrogen and methyl,
each RD is an independently selected divalent hydrocarbon group of 2 to 20 carbon atoms, each subscript a independently has a value such that 0 < a < 150, and subscript has a value such that 1 < b < 100,
(B) a polyurethane binder,
(C) a surfactant; and
(D) water; and
II) a crosslinker.
15. A kit comprising: the system of claim 14, and
III) instructions for combining I) the aqueous coating composition and II) the crosslinker to form a crosslinkable coating composition.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US202363599579P | 2023-11-16 | 2023-11-16 | |
| US63/599,579 | 2023-11-16 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2025106269A1 true WO2025106269A1 (en) | 2025-05-22 |
Family
ID=93521731
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2024/053778 Pending WO2025106269A1 (en) | 2023-11-16 | 2024-10-31 | Aqueous coating composition containing an aminosiloxane ester copolymer and a polyurethane binder, and methods for preparation and use thereof |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2025106269A1 (en) |
Citations (26)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US2968575A (en) | 1953-06-30 | 1961-01-17 | Du Pont | Stable polyurethane latex and process of making same |
| US3412054A (en) | 1966-10-31 | 1968-11-19 | Union Carbide Corp | Water-dilutable polyurethanes |
| US3905929A (en) | 1973-03-23 | 1975-09-16 | Bayer Ag | Aqueous dispersions of polyurethane having side chain polyoxyethylene units |
| US3920598A (en) | 1973-03-23 | 1975-11-18 | Bayer Ag | Non-ionic polyurethane dispersions having side chains of polyoxyethylene |
| US3930921A (en) | 1971-07-19 | 1976-01-06 | Barrett & Company | Method of finishing leather |
| US4122029A (en) | 1977-07-27 | 1978-10-24 | Dow Corning Corporation | Emulsion compositions comprising a siloxane-oxyalkylene copolymer and an organic surfactant |
| US5387417A (en) | 1991-08-22 | 1995-02-07 | Dow Corning Corporation | Non-greasy petrolatum emulsion |
| US5539021A (en) | 1995-06-05 | 1996-07-23 | The Dow Chemical Company | Process for preparing high internal phase ratio emulsions and latexes derived thereof |
| US5811487A (en) | 1996-12-16 | 1998-09-22 | Dow Corning Corporation | Thickening silicones with elastomeric silicone polyethers |
| US5959027A (en) | 1997-03-17 | 1999-09-28 | The Dow Chemical Company | Continuous process for preparing a polyurethane latex |
| US6180117B1 (en) | 1994-05-27 | 2001-01-30 | General Electric Company | Method of preparing microemulsions of amino silicone fluids and MQ resin mixtures |
| US6231926B1 (en) | 1998-06-06 | 2001-05-15 | Basf Aktiengesellschaft | Poromeric synthetic leathers |
| US6353051B1 (en) | 1999-03-10 | 2002-03-05 | E. I. Du Pont De Nemours And Company | Top coating for synthetic leathers |
| US20040167252A1 (en) | 2003-02-26 | 2004-08-26 | Martin Melchiors | Aqueous coating compositions based on PU-PAC hybrid dispersions |
| US6794445B2 (en) | 2002-04-17 | 2004-09-21 | Goldschmidt Ag | Aqueous polysiloxane-polyurethane dispersion, its preparation and use in coating compositions |
| US20050222368A1 (en) | 2004-03-30 | 2005-10-06 | Juergen Reiners | Aqueous polyurethane dispersions |
| US7232859B2 (en) | 2002-10-31 | 2007-06-19 | Dow Global Technologies Inc. | Polyurethane dispersion and articles prepared therefrom |
| US20100310882A1 (en) | 2007-12-27 | 2010-12-09 | Midori Hokuyo Co., Ltd. | Topcoat |
| US20110112245A1 (en) | 2008-07-11 | 2011-05-12 | Taku Nakamura | Aqueous polyurethane resin dispersion, process for preparing the same and coating composition containing the same |
| US20130078474A1 (en) * | 2010-05-25 | 2013-03-28 | Clariant Finance (Bvi) Limited | Aqueous Polyurethane-Polyurea Dispersions |
| US8591999B2 (en) | 2008-05-28 | 2013-11-26 | Dow Corning Corporation | Coating compositions |
| US9200404B2 (en) | 2012-11-06 | 2015-12-01 | Dow Global Technologies Llc | Aqueous leather coating composition and method for coating leather |
| US10100377B2 (en) | 2015-08-31 | 2018-10-16 | Rohm And Haas Company | Aqueous multi-stage copolymer compositions for use in leather topcoats |
| US20220017771A1 (en) * | 2020-07-15 | 2022-01-20 | Nan Ya Plastics Corporation | Water-based surface treatment agent |
| US20220325134A1 (en) * | 2019-11-19 | 2022-10-13 | Dow Silicones Corporation | Aqueous leather coating composition |
| WO2023278918A1 (en) | 2021-06-28 | 2023-01-05 | Dow Silicones Corporation | Aminosiloxane ester copolymer and methods for the preparation and use of the copolymer |
-
2024
- 2024-10-31 WO PCT/US2024/053778 patent/WO2025106269A1/en active Pending
Patent Citations (28)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US2968575A (en) | 1953-06-30 | 1961-01-17 | Du Pont | Stable polyurethane latex and process of making same |
| US3412054A (en) | 1966-10-31 | 1968-11-19 | Union Carbide Corp | Water-dilutable polyurethanes |
| US3930921A (en) | 1971-07-19 | 1976-01-06 | Barrett & Company | Method of finishing leather |
| US3905929A (en) | 1973-03-23 | 1975-09-16 | Bayer Ag | Aqueous dispersions of polyurethane having side chain polyoxyethylene units |
| US3920598A (en) | 1973-03-23 | 1975-11-18 | Bayer Ag | Non-ionic polyurethane dispersions having side chains of polyoxyethylene |
| US4122029A (en) | 1977-07-27 | 1978-10-24 | Dow Corning Corporation | Emulsion compositions comprising a siloxane-oxyalkylene copolymer and an organic surfactant |
| US5387417A (en) | 1991-08-22 | 1995-02-07 | Dow Corning Corporation | Non-greasy petrolatum emulsion |
| US6180117B1 (en) | 1994-05-27 | 2001-01-30 | General Electric Company | Method of preparing microemulsions of amino silicone fluids and MQ resin mixtures |
| US5688842A (en) | 1995-06-05 | 1997-11-18 | The Dow Chemical Company | Process for preparing high internal phase ratio emulsions and latexes derived thereof |
| US5539021A (en) | 1995-06-05 | 1996-07-23 | The Dow Chemical Company | Process for preparing high internal phase ratio emulsions and latexes derived thereof |
| US5811487A (en) | 1996-12-16 | 1998-09-22 | Dow Corning Corporation | Thickening silicones with elastomeric silicone polyethers |
| US5959027A (en) | 1997-03-17 | 1999-09-28 | The Dow Chemical Company | Continuous process for preparing a polyurethane latex |
| US6231926B1 (en) | 1998-06-06 | 2001-05-15 | Basf Aktiengesellschaft | Poromeric synthetic leathers |
| US6353051B1 (en) | 1999-03-10 | 2002-03-05 | E. I. Du Pont De Nemours And Company | Top coating for synthetic leathers |
| US6794445B2 (en) | 2002-04-17 | 2004-09-21 | Goldschmidt Ag | Aqueous polysiloxane-polyurethane dispersion, its preparation and use in coating compositions |
| US7232859B2 (en) | 2002-10-31 | 2007-06-19 | Dow Global Technologies Inc. | Polyurethane dispersion and articles prepared therefrom |
| US20040167252A1 (en) | 2003-02-26 | 2004-08-26 | Martin Melchiors | Aqueous coating compositions based on PU-PAC hybrid dispersions |
| US20050222368A1 (en) | 2004-03-30 | 2005-10-06 | Juergen Reiners | Aqueous polyurethane dispersions |
| US20100310882A1 (en) | 2007-12-27 | 2010-12-09 | Midori Hokuyo Co., Ltd. | Topcoat |
| US8591999B2 (en) | 2008-05-28 | 2013-11-26 | Dow Corning Corporation | Coating compositions |
| US20110112245A1 (en) | 2008-07-11 | 2011-05-12 | Taku Nakamura | Aqueous polyurethane resin dispersion, process for preparing the same and coating composition containing the same |
| US20130078474A1 (en) * | 2010-05-25 | 2013-03-28 | Clariant Finance (Bvi) Limited | Aqueous Polyurethane-Polyurea Dispersions |
| US9200404B2 (en) | 2012-11-06 | 2015-12-01 | Dow Global Technologies Llc | Aqueous leather coating composition and method for coating leather |
| US10100377B2 (en) | 2015-08-31 | 2018-10-16 | Rohm And Haas Company | Aqueous multi-stage copolymer compositions for use in leather topcoats |
| US20220325134A1 (en) * | 2019-11-19 | 2022-10-13 | Dow Silicones Corporation | Aqueous leather coating composition |
| US11518905B2 (en) | 2019-11-19 | 2022-12-06 | Dow Silicones Corporation | Aqueous leather coating composition |
| US20220017771A1 (en) * | 2020-07-15 | 2022-01-20 | Nan Ya Plastics Corporation | Water-based surface treatment agent |
| WO2023278918A1 (en) | 2021-06-28 | 2023-01-05 | Dow Silicones Corporation | Aminosiloxane ester copolymer and methods for the preparation and use of the copolymer |
Non-Patent Citations (2)
| Title |
|---|
| "Manual of Patent Examining Procedure", January 2018 |
| P. PIETERICH: "Aqueous Emulsion, Dispersion and Solutions of Polyurethanes", SYNTHESIS AND PROPERTIES IN PROGRESS IN ORGANIC COATINGS, vol. 9, 1981, pages 281 - 340 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP1387859B1 (en) | Polyurethane-polyurea dispersions as coating agents | |
| US6228926B1 (en) | Water based plasticizer free polyurethane-wax coating and repair composition and method | |
| US5525427A (en) | Abrasion resistant silicone weatherstrip coating | |
| EP0317057B1 (en) | Process for preparing water-based silicone-organic polymer compositions | |
| CN106883370B (en) | Self-extinction waterborne polyurethane resin with ultralow glossiness and preparation method and application thereof | |
| US11066504B2 (en) | Aqueous polyurethane dispersions | |
| WO2019021794A1 (en) | Aqueous coating agent composition and aqueous coating composition comprising same for lubricating film | |
| WO2016098336A1 (en) | Water-based coating agent composition, water-based lubricating film paint composition comprising same, and member | |
| US11242458B2 (en) | Organopolysiloxane emulsion composition and resin composition | |
| JPH05222150A (en) | Water-dispersible polyisocyanate mixture, process for producing same, and method of using it in two-component water-based coating material | |
| US11932747B2 (en) | Use of long-chain citric acid esters in aqueous polyurethane dispersions | |
| EP0705884B1 (en) | Method for treating plastic, leather or rubber substrates | |
| EP2812376A1 (en) | Coating applications | |
| CN106414630B (en) | The water-based composition of elongation at break with raising | |
| CN108473771B (en) | Organopolysiloxane emulsified composition and resin composition | |
| CN114231155A (en) | Hydroxyalkyl aqueous organosilicon modified polyurethane aqueous dispersion composition and preparation method thereof | |
| CN110746812B (en) | Thixotropic mixture containing nonionic polyurethane compound and water-based leather surface treatment coating comprising the same | |
| CN111019087B (en) | Polyurethane-polyurea aqueous dispersion for synthetic leather, and preparation method and application thereof | |
| WO2025106269A1 (en) | Aqueous coating composition containing an aminosiloxane ester copolymer and a polyurethane binder, and methods for preparation and use thereof | |
| US20090076222A1 (en) | Clear functional coating for elastomers | |
| WO2025106268A1 (en) | Aqueous coating composition containing an aminosiloxane ester copolymer, method for preparation of the aqueous coating composition, and use for treating leather | |
| CN119013356A (en) | Coating composition and use thereof | |
| WO2025216964A1 (en) | Aqueous coating composition containing a hydroxyl – functional aminosiloxane ester copolymer, method for preparation thereof, and use for leather treatment | |
| JP4497755B2 (en) | Water-based polyurethane resin composition for condoms and method for producing condoms | |
| JP3197130B2 (en) | Thermocrosslinkable polyurethane emulsion composition |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 24805954 Country of ref document: EP Kind code of ref document: A1 |