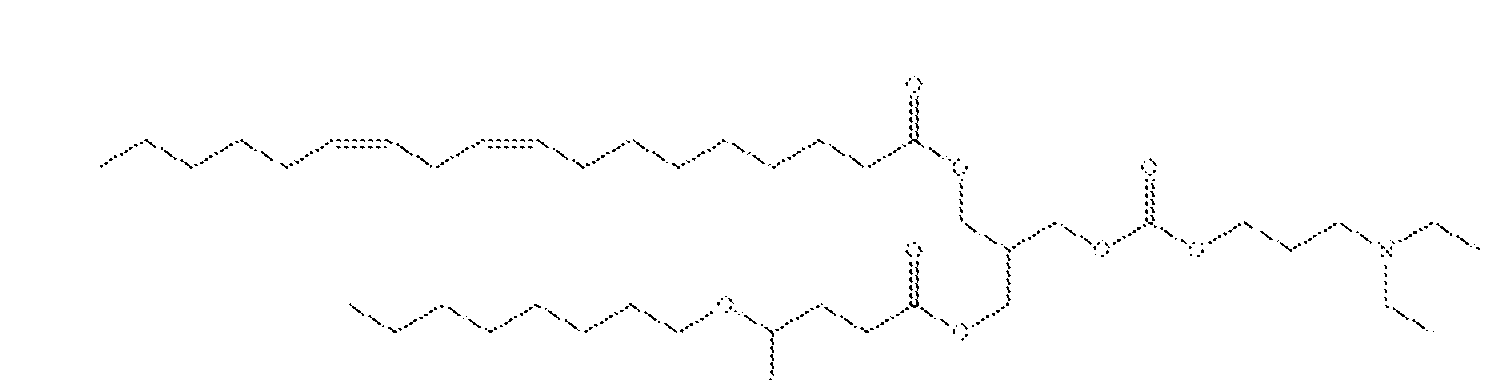
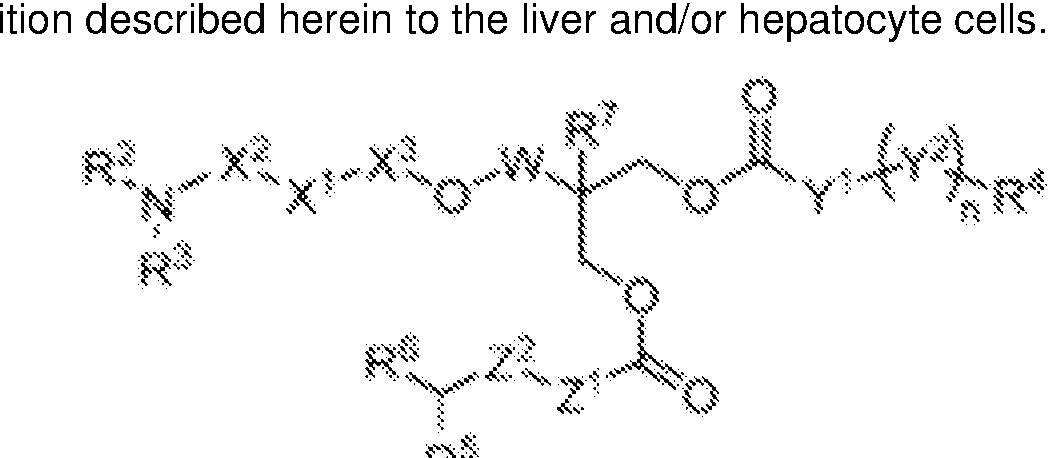
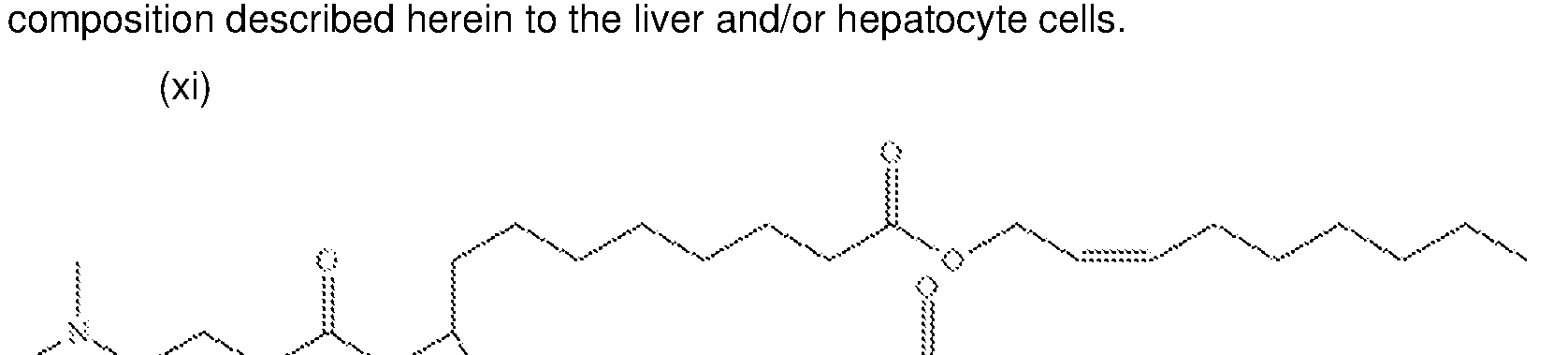
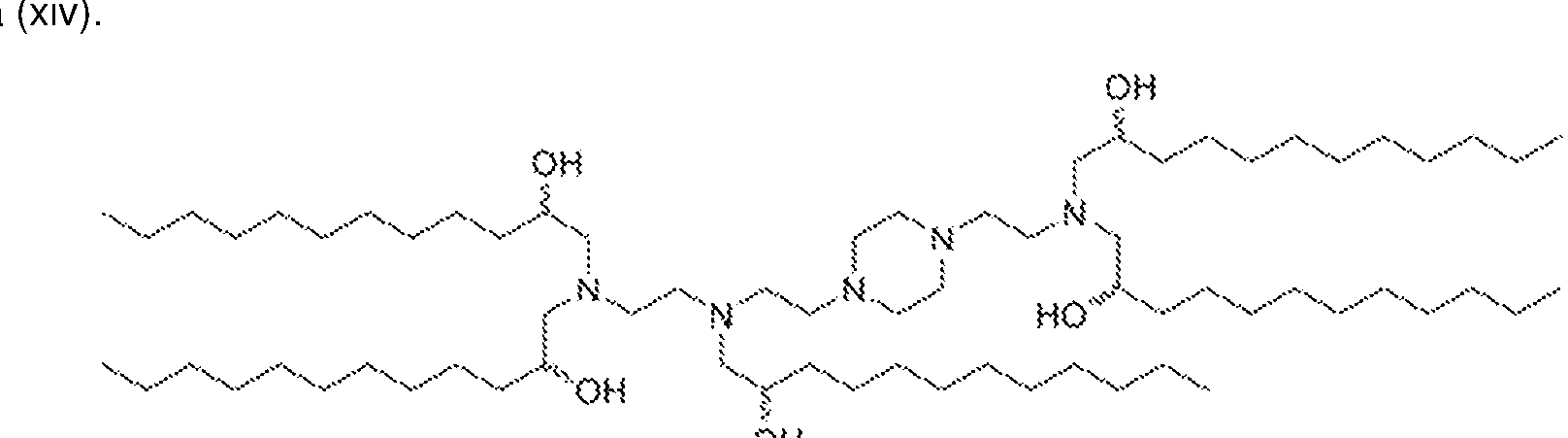
WO2025101842A1 - Compositions and methods for targeted delivery of therapeutic agents - Google Patents
Compositions and methods for targeted delivery of therapeutic agents Download PDFInfo
- Publication number
- WO2025101842A1 WO2025101842A1 PCT/US2024/055047 US2024055047W WO2025101842A1 WO 2025101842 A1 WO2025101842 A1 WO 2025101842A1 US 2024055047 W US2024055047 W US 2024055047W WO 2025101842 A1 WO2025101842 A1 WO 2025101842A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- fragment
- polypeptide
- variant
- human
- amino acid
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P37/00—Drugs for immunological or allergic disorders
- A61P37/02—Immunomodulators
- A61P37/06—Immunosuppressants, e.g. drugs for graft rejection
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K14/00—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- C07K14/435—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- C07K14/52—Cytokines; Lymphokines; Interferons
- C07K14/54—Interleukins [IL]
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K14/00—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- C07K14/435—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- C07K14/52—Cytokines; Lymphokines; Interferons
- C07K14/54—Interleukins [IL]
- C07K14/5428—IL-10
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
- C07K16/18—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans
- C07K16/28—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K2039/505—Medicinal preparations containing antigens or antibodies comprising antibodies
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K38/00—Medicinal preparations containing peptides
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K14/00—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- C07K14/435—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- C07K14/705—Receptors; Cell surface antigens; Cell surface determinants
- C07K14/715—Receptors; Cell surface antigens; Cell surface determinants for cytokines; for lymphokines; for interferons
- C07K14/7155—Receptors; Cell surface antigens; Cell surface determinants for cytokines; for lymphokines; for interferons for interleukins [IL]
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/20—Immunoglobulins specific features characterized by taxonomic origin
- C07K2317/24—Immunoglobulins specific features characterized by taxonomic origin containing regions, domains or residues from different species, e.g. chimeric, humanized or veneered
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/30—Immunoglobulins specific features characterized by aspects of specificity or valency
- C07K2317/35—Valency
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/50—Immunoglobulins specific features characterized by immunoglobulin fragments
- C07K2317/52—Constant or Fc region; Isotype
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/70—Immunoglobulins specific features characterized by effect upon binding to a cell or to an antigen
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/70—Immunoglobulins specific features characterized by effect upon binding to a cell or to an antigen
- C07K2317/76—Antagonist effect on antigen, e.g. neutralization or inhibition of binding
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/90—Immunoglobulins specific features characterized by (pharmaco)kinetic aspects or by stability of the immunoglobulin
- C07K2317/92—Affinity (KD), association rate (Ka), dissociation rate (Kd) or EC50 value
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2319/00—Fusion polypeptide
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N15/00—Mutation or genetic engineering; DNA or RNA concerning genetic engineering, vectors, e.g. plasmids, or their isolation, preparation or purification; Use of hosts therefor
- C12N15/09—Recombinant DNA-technology
- C12N15/11—DNA or RNA fragments; Modified forms thereof; Non-coding nucleic acids having a biological activity
- C12N15/62—DNA sequences coding for fusion proteins
Definitions
- compositions and related methods that effect targeted delivery of IL-22 polypeptides or fragments or variants thereof to effector targets in a desired cell, tissue and/or organ of interest while minimizing or avoiding undesirable delivery to other cells, tissues, or organs are provided.
- Compositions and methods related to macromolecules, such as an ANDbodyTM, that include an IL-22 polypeptide or a fragment or variant thereof and an address binding domain specific for an IL-10 family address target are described.
- IL-22 mutein polypeptides that have altered (e.g., decreased) affinity for the IL-22 receptor.
- Undesirable off-target effects are a problem for otherwise desirable therapeutic targets that are present in healthy as well as diseased tissues.
- compositions described herein comprise macromolecules, such as an ANDbodyTM, that include an effector target binding domain comprising an IL-22 polypeptide or fragment or variant thereof that binds an IL-22 receptor (e.g., a receptor comprising IL-22Ra, and/or IL-10R2, e.g., a receptor comprising IL-22Ra and IL-10R2), and an address binding domain specific for (e.g., that specifically binds to) an IL-10 family address target.
- an ANDbodyTM that include an effector target binding domain comprising an IL-22 polypeptide or fragment or variant thereof that binds an IL-22 receptor (e.g., a receptor comprising IL-22Ra, and/or IL-10R2, e.g., a receptor comprising IL-22Ra and IL-10R2), and an address binding domain specific for (e.g., that specifically binds to) an IL-10 family address target.
- the address target is generally sufficiently restricted in the subject to target the macromolecule to the desired cell, tissue or organ.
- the IL-22 polypeptide or fragment or variant thereof does not influence signaling by the IL-22 receptor in the absence of an address target binding domain.
- the address target binding domain may not influence signaling upon binding the IL-10 family address target.
- localization of the IL-22 polypeptide or fragment or variant thereof by the address target binding domain enables the IL-22 polypeptide or fragment or variant thereof to bind the IL-22 receptor sufficiently to elicit an influence on signaling by the IL-22 receptor in the target cell or tissue.
- the macromolecules described herein may be linked to one or more small molecules.
- compositions described herein can be used, e.g., to specifically deliver a therapeutic agent (for example, the IL-22 polypeptide or fragment or variant thereof, the small molecule, or both) to a desired location, e.g., a target cell, tissue or organ, in a subject, while avoiding undesirable off-target effects (e.g., undesirable off-target effects in the brain; skin; cardiovascular system, e.g., heart or vasculature) and/or avoiding certain toxicity (e.g., avoiding cardiovascular disease, such as stroke or myocardial infarction).
- a therapeutic agent for example, the IL-22 polypeptide or fragment or variant thereof, the small molecule, or both
- a desired location e.g., a target cell, tissue or organ
- undesirable off-target effects e.g., undesirable off-target effects in the brain; skin; cardiovascular system, e.g., heart or vasculature
- certain toxicity e.g., avoiding cardiovascular disease, such as stroke
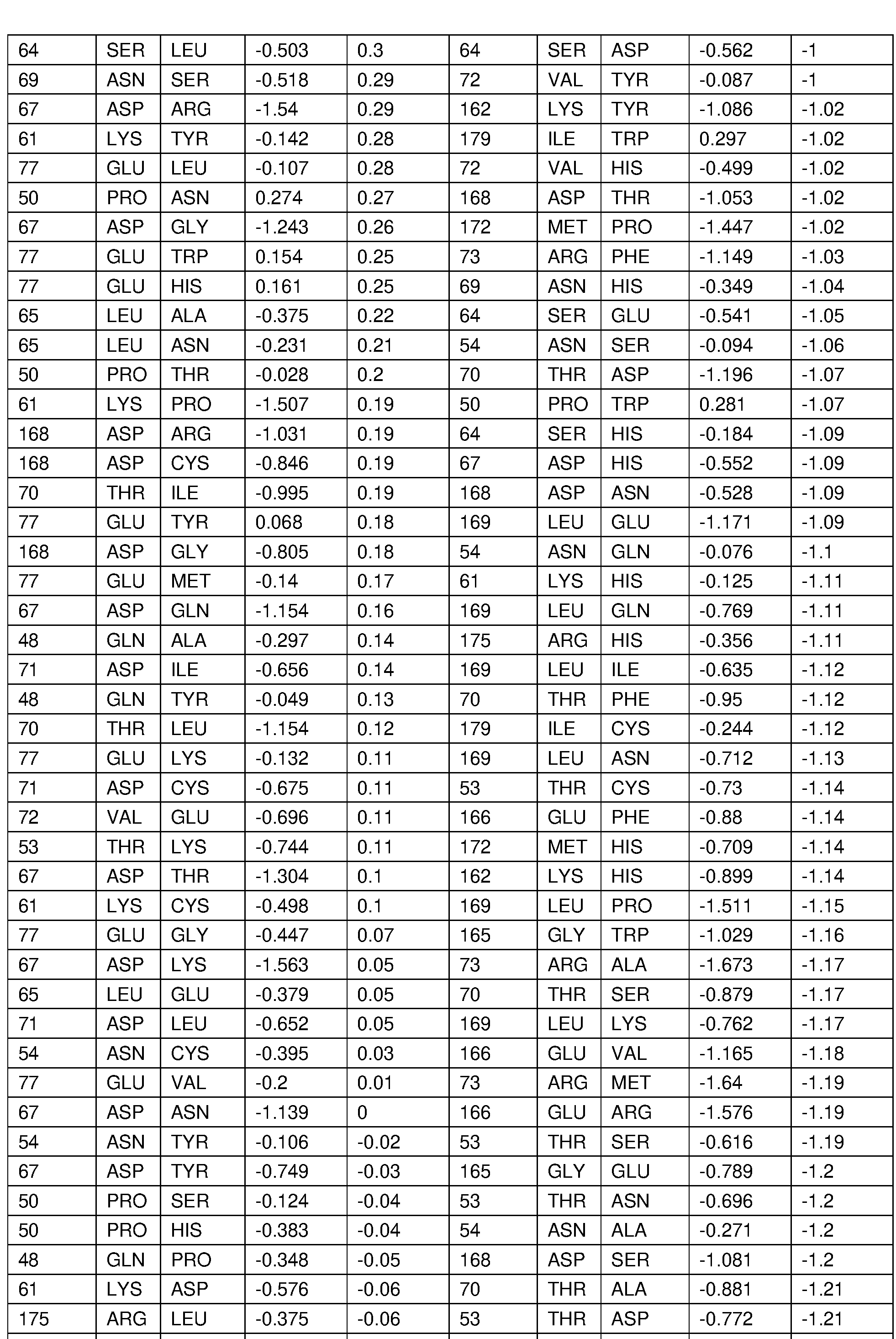
- the disclosure provides an IL-22 mutein polypeptide or a fragment or variant thereof that binds an IL-22 receptor on the surface of a target cell, wherein the IL-22 mutein polypeptide or fragment or variant thereof comprises one or more of the amino acid substitution mutations provided in Table 6, Table 7A, Table 7B, Table 8, Table 9A, Table 9B, or Table 9C, wherein the amino acid positions are numbered relative to SEQ ID NO: 7.
- the disclosure provides an IL-22 mutein polypeptide or a fragment or variant thereof that binds an IL-22 receptor on the surface of a target cell, wherein the IL-22 mutein polypeptide or fragment or variant thereof comprises an amino acid substitution mutation at one or more of positions K61 , D71 , R73, D168, and R175 relative to a human IL-22 polypeptide, wherein the amino acid positions are numbered relative to SEQ ID NO: 7.
- the IL-22 polypeptide or fragment or variant thereof comprises one or more of a K61 S, D71 N, D71 L, D71 G, R73Q, R73S, R73N, D168N, D168G, and R175S amino acid substitution mutation.
- the IL-22 mutein polypeptide or fragment or variant thereof comprises a K61 S amino acid substitution mutation, optionally wherein the IL-22 mutein polypeptide or fragment or variant thereof comprises SEQ ID NO: 17;
- the IL-22 mutein polypeptide or fragment or variant thereof comprises a D71 G amino acid substitution mutation, optionally wherein the IL-22 mutein polypeptide or fragment or variant thereof comprises SEQ ID NO: 15;
- the IL-22 mutein polypeptide or fragment or variant thereof comprises a D71 L amino acid substitution mutation, optionally wherein the IL-22 mutein polypeptide or fragment or variant thereof comprises SEQ ID NO: 11 ;
- the IL-22 mutein polypeptide or fragment or variant thereof comprises a D71 N amino acid substitution mutation, optionally wherein the IL- 22 mutein polypeptide or fragment or variant thereof comprises SEQ ID NO: 18;
- the IL-22 mutein polypeptide or fragment or variant thereof comprises an
- the IL-22 mutein polypeptide or fragment or variant thereof has a binding affinity for IL22RA1 (IL-22Ra) that is at least 1 .3-fold lower than the binding affinity of a wild-type IL-22 or fragment thereof.
- the IL-22 mutein polypeptide or fragment or variant thereof does not comprise a leader sequence.
- the IL-22 mutein polypeptide or fragment or variant thereof does not comprise the amino acid sequence of SEQ ID NO: 14.
- the disclosure provides a polypeptide comprising the IL-22 mutein polypeptide or fragment or variant thereof of any one of the above aspects.
- the disclosure provides an IL-22 mutein polypeptide that has at least 95% identity to human IL-22 (SEQ ID NO: 7), comprises D71 L and D168G amino acid substitution mutations relative to SEQ ID NO: 7, and has a significantly reduced binding affinity for IL-22Ra relative to a wildtype human IL-22 polypeptide.
- the disclosure provides an IL-22 mutein polypeptide that has at least 95% identity to human IL-22 (SEQ ID NO: 7), comprises an R73Q amino acid substitution mutation relative to SEQ ID NO: 7, and has a significantly reduced binding affinity for IL-22Ra relative to a wild-type human IL-22 polypeptide.
- the disclosure provides an IL-22 mutein polypeptide that has at least 95% identity to human IL-22 (SEQ ID NO: 7), comprises an R73S amino acid substitution mutation relative to SEQ ID NO: 7, and has a significantly reduced binding affinity for IL-22Ra relative to a wild-type human IL-22 polypeptide.
- the disclosure provides a macromolecule comprising a first binding site and a second binding site, wherein (a) the first binding site comprises an IL-22 polypeptide or a fragment or variant thereof that binds an IL-22 receptor on the surface of a target cell, and (b) the second binding site is specific for an IL-10 family address target provided in Table 1 that is expressed on the surface of the target cell; wherein: (i) the second binding site localizes the first binding site to the IL-10 family address target such that the IL-22 polypeptide or a fragment or variant thereof influences signaling by the IL-22 receptor in the target cell; (ii) the second binding site does not substantially influence signaling upon binding the IL-10 family address target; and (iii) the IL-22 polypeptide or fragment or variant thereof does not substantially influence signaling by the IL-22 receptor in the absence of localization by the second binding site.
- the IL-22 polypeptide or fragment or variant thereof has been engineered to have reduced binding affinity for the IL-22 receptor.
- the IL-22 polypeptide or fragment or variant thereof comprises one or more amino acid substitution mutations at one or more residues that are predicted to be interaction surfaces between IL-22 and IL-22Ra and/or IL-10R2.
- the IL-22 polypeptide or fragment or variant thereof is a variant of a human IL-22 or fragment thereof and comprises (a) one or more of the amino acid substitution mutations of Table 6, Table 7A, Table 7B, Table 8, Table 9A, Table 9B, or Table 9C; and/or (b) one or more of a K61 S, D71 N, D71 L, D71 G, R73Q, R73S, R73N, D168N, D168G, and R175S amino acid substitution mutation.
- the disclosure provides a method of modulating IL-22 receptor signaling in a target cell of a subject, the method comprising administering to the subject the macromolecule of any one of the above aspects.
- the disclosure provides a method of treating a disease or disorder selected from Crohn's disease, ulcerative colitis, graft-vs-host disease, atopic dermatitis, psoriasis, wounds, diabetic skin ulcers, acute pancreatitis, chronic pancreatitis, autoimmune pancreatitis, nephritis, acute lung injury, viral lung injury, fibrosis, fibrosis, non-alcoholic steatohepatitis, viral hepatitis, and autoimmune hepatitis in a subject, the method comprising administering to the subject the macromolecule of any one of the above aspects.
- a disease or disorder selected from Crohn's disease, ulcerative colitis, graft-vs-host disease, atopic dermatitis, psoriasis, wounds, diabetic skin ulcers, acute pancreatitis, chronic pancreatitis, autoimmune pancreatitis, nephritis, acute lung injury, viral lung injury,
- the disclosure provides a macromolecule comprising (a) a first binding site comprising an IL-22 polypeptide or a fragment or variant thereof, and (b) a second binding site comprising an anti-CDH17 antibody or antigen-binding fragment thereof, wherein the macromolecule is a fusion protein, and wherein the first binding site and the second binding site are connected by a linker.
- the disclosure provides a macromolecule comprising a first binding site and a second binding site, wherein (a) the first binding site comprises an IL-22 polypeptide or a fragment or variant thereof that binds an IL-22 receptor on the surface of a target cell, and (b) the second binding site is specific for an IL-10 family address target provided in Table 1 that is expressed on the surface of the target cell; wherein (i) the second binding site localizes the first binding site to the IL-10 family address target such that the IL-22 polypeptide or a fragment or variant thereof influences signaling by the IL-22 receptor in the target cell; (ii) the second binding site does not substantially influence signaling upon binding the IL-10 family address target; and (iii) the IL-22 polypeptide or fragment or variant thereof does not substantially influence signaling by the IL-22 receptor in the absence of localization by the second binding site.
- the IL-10 family address target is a protein, lipid, or sugar. In some embodiments, the IL-10 family address target is a protein.
- the target cell is a lower gastrointestinal (G I) tract cell
- the IL-10 family address target is MS4A18, SI, MS4A12, ATP4B, MUC17, TMIGD1 , MS4A10, MEP1A, CDH17, TM4SF20, SLC26A3, ATP4A, GPA33, BTNL3, MEP1 B, UGT2B17, KCNE2, or TMPRSS15.
- the target cell is a kidney cell
- the IL-10 family address target is TMEM207, OR2T10, SLC22A6, SLC22A8, SLC12A1 , AQP2, SLC22A12, SLC12A3, TMEM174, FXYD4, SLC34A1 , KCNJ1 , SLC22A11 , TMEM52B, SLC6A18, CTXN3, SLC4A1 , TMEM72, SLC22A2, SLC4A9, BSND, CDH16, RDH8, or AQP6.
- the target cell is a liver cell
- the IL-10 family address target is UGT2B10, C8A, SLCO1 B1 , C9, SLC25A47, SLC17A2, SLC10A1 , UGT2B4, SLC22A1 , or CYP8B1 .
- the target cell is a pancreas cell
- the IL-10 family address target is GP2, AQP12A, AQP12B, CUZD1 , LHFPL5, AQP8, KIRREL2, G6PC2, SLC17A6, or GPR119.
- the target cell is a skin cell
- the IL-10 family address target is CLEC2A, KCNJ18, KLRF2, DSC1 , TYR, AWAT2, TMEM271 , DSG1 , ACER1 , GSDMA, TRPM1 , GJB4, CD1A, ASPRV1 , or ABCA12.
- the IL-10 family address target is selected from CDH17, GP2, CDH16, DSG1 , and DSG3.
- the IL-10 family address target is CDH17.
- the IL-10 family address target is CDH16.
- the IL-10 family address target is GP2.
- the disclosure provides a macromolecule comprising a first binding site and a second binding site, wherein (a) the first binding site comprises an IL-22 polypeptide or a fragment or variant thereof that binds an IL-22 receptor on the surface of a target cell, and (b) the second binding site is specific for an IL-10 family address target selected from CDH17, CDH16, and GP2 that is expressed on the surface of the target cell; wherein (i) the second binding site localizes the first binding site to the IL-10 family address target such that the IL-22 polypeptide or fragment or variant thereof influences signaling by the IL-22 receptor in the target cell; (ii) the second binding site does not substantially influence signaling upon binding the IL-10 family address target; and (iii) the IL-22 polypeptide or fragment or variant thereof does not substantially influence signaling by the IL-22 receptor in the absence of localization by the second binding site.
- the first binding site comprises an IL-22 polypeptide or a fragment or variant thereof that binds an IL-22 receptor
- the second binding site comprises a polypeptide.
- the second binding site is an antibody or antigen binding fragment thereof.
- the antibody or antigen-binding fragment thereof comprises an scFv, BsIgG, a BsAb fragment, a BiTE, a dual-affinity re-targeting protein (DART), a tandem diabody (TandAb), a diabody, an Fab2, a di-scFv, chemically linked F(ab’)2, an Ig molecule with 2, 3 or 4 different antigen binding sites, a DVI-IgG four-in- one, an ImmTac, an HSAbody, an IgG-IgG, a Cov-X-Body, an scFv1 -PEG-scFv2, an appended IgG, an DVD-IgG, an affibody, an affilin, an affimer, an affitin, an alphabody, an anti
- the second binding site comprises a ligand of the address target.
- the IL-10 family address target is CDH17 and the second binding site is an anti-CDH17 antibody or antigen-binding fragment thereof.
- the IL-10 family address target is CDH16 and the second binding site is an anti-CDH16 antibody or antigen-binding fragment thereof.
- the IL-10 family address target is GP2 and the second binding site is an anti-GP2 antibody or antigen-binding fragment thereof.
- the disclosure provides an IL-22 polypeptide or a fragment or variant thereof that binds (e.g., specifically binds) an IL-22 receptor on the surface of a target cell.
- the IL-22 polypeptide or fragment or variant thereof binds the IL-22 receptor. In some embodiments, the IL-22 polypeptide or fragment or variant thereof binds the human IL-22 receptor. In some embodiments, the IL-22 polypeptide or fragment or variant thereof binds human IL-22Ra and/or IL-10R2. For example, binding can be evaluated using an assay described herein. In some embodiments, the IL-22 polypeptide or fragment or variant thereof binds IL-22BP. In some embodiments, the IL-22 polypeptide or fragment or variant thereof binds human IL-22BP.
- the IL-22 polypeptide or fragment or variant thereof agonizes the IL-22 receptor. In some embodiments, the IL-22 polypeptide or fragment or variant thereof agonizes the human IL-22 receptor. In some embodiments, the IL-22 polypeptide or fragment or variant thereof agonizes human IL-22Ra and/or IL-10R2, e.g., in an assay described herein.
- the macromolecule i.e., a macromolecule comprising the IL-22 polypeptide or fragment or variant thereof agonizes IL-22BP. In some embodiments, the macromolecule agonizes human IL-22BP.
- the IL-22 polypeptide or fragment or variant thereof has low (e.g., lower) binding affinity for IL-22Ra and/or IL-10R2, as compared to the binding affinity of wild type human IL-22 (e.g., of SEQ ID NO: 8) for the IL-22 receptor, e.g., in an assay described herein.
- the IL-22 polypeptide or fragment or variant thereof has low (e.g., lower) binding affinity for IL-22BP, as compared to the binding affinity of wild type human IL-22 (e.g., of SEQ ID NO: 8) for IL-22BP, e.g., in an assay described herein.
- the affinity of the IL-22 polypeptide or fragment or variant thereof for IL- 22Ra, IL-22BP, and/or IL-10R2 is lower than the affinity of the second binding site for the IL-10 family address target.
- the avidity of the IL-22 polypeptide or fragment or variant thereof for IL- 22Ra, IL-22BP, and/or IL-10R2 is lower than the avidity of the second binding site for the IL-10 family address target.
- the Kd of the IL-22 polypeptide or fragment or variant thereof for IL- 22Ra, IL-22BP, and/or IL-10R2 is higher than the Kd of the second binding site for the IL-10 family address target;
- the ECso of the IL-22 polypeptide or fragment or variant thereof for IL-22Ra and/or IL- 10R2 is higher than the ECso of the second binding site for the IL-10 family address target; or
- the IC50 of the IL-22 polypeptide or fragment or variant thereof for IL-22Ra and/or IL-10R2 is higher than the IC50 of the second binding site for the IL-10 family address target.
- the IL-22 polypeptide or fragment or variant thereof has an affinity to the IL-22 receptor of at least about 2 times, at least about 5 times, or at least about 10 times less than the affinity of the second binding site to the IL-10 family address target.
- the affinity of the second binding site to the IL-10 family address target has a Kd of greater than about 1 nM, greater than about 2 nM, or greater than about 50 nm.
- the IL-22 polypeptide or fragment or variant thereof has been engineered to have reduced binding affinity for the IL-22 receptor.
- the IL-22 polypeptide or fragment or variant thereof when part of the macromolecule, exhibits reduced agonist activity relative to the IL-22 polypeptide or fragment or variant thereof when not part of the macromolecule, on a mole-adjusted basis.
- the IL-22 polypeptide or fragment or variant thereof comprises one or more amino acid substitution mutations at one or more residues that are predicted to be interaction surfaces between IL-22 and IL-22Ra and/or IL-10R2. In some embodiments, the IL-22 polypeptide or fragment or variant thereof has reduced binding affinity for IL22RA1 relative to a wild-type IL-22 or fragment thereof. In some embodiments, the IL-22 polypeptide or fragment or variant thereof comprises one or more amino acid substitution mutations at one or more residues that are predicted to be interaction surfaces between IL-22 and IL-22Ra.
- the IL-22 polypeptide or fragment or variant thereof does not comprise a leader sequence, e.g., does not comprise a leader sequence of a pro-form of an IL-22 polypeptide. In some embodiments, the IL-22 polypeptide or fragment or variant thereof does not comprise the leader sequence comprised by the polypeptide of SEQ ID NO: 7, e.g., does not comprise residues 1 to 33 included in SEQ ID NO: 7.
- the IL-22 polypeptide or fragment or variant thereof does not comprise the sequence of SEQ ID NO: 14.
- the IL-22 polypeptide or fragment or variant thereof is a human IL-22 or fragment or variant thereof.
- the IL-22 polypeptide or fragment or variant thereof is a human IL-22 or fragment or variant thereof and comprises one or more of the amino acid substitution mutations of Table 6, Table 7A, Table 7B, Table 8, Table 9A, Table 9B, or Table 9C.
- the human IL-22 polypeptide or fragment or variant thereof comprises an amino acid substitution mutation of one or more of K61 , D71 , R73, D168, and R175. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a K61 amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D71 amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a R73 amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D168 amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a R175 amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D71 amino acid substitution mutation and a D168 amino acid substitution mutation. The amino acid position numbering is with reference to the IL-22 sequence of SEQ ID NO: 7.
- the human IL-22 polypeptide or fragment or variant thereof comprises one or more of a K61 S, D71 N, D71 L, D71 G, R73Q, R73S, R73N, D168N, D168G, and R175S amino acid substitution mutation.
- the human IL-22 polypeptide or fragment or variant thereof comprises a K61 S amino acid substitution mutation.
- the human IL-22 polypeptide or fragment or variant thereof comprises a D71 N amino acid substitution mutation.
- the human IL-22 polypeptide or fragment or variant thereof comprises a D71 L amino acid substitution mutation.
- the human IL-22 polypeptide or fragment or variant thereof comprises a D71 G amino acid substitution mutation.
- the human IL-22 polypeptide or fragment or variant thereof comprises a R73Q amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a R73S amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a R73N amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D168N amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D168G amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a R175S amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D71 L amino acid substitution mutation and a D168G amino acid substitution mutation. The amino acid position numbering is with reference to the IL- 22 sequence of SEQ ID NO: 7.
- the human IL-22 polypeptide or fragment or variant thereof comprises a K61 S amino acid substitution mutation.
- the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 17 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 17).
- the human IL-22 polypeptide or fragment or variant thereof comprises a K61 S amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the human IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 17.
- the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 17.
- the human IL-22 polypeptide or fragment or variant thereof comprises a D71 G amino acid substitution mutation.
- the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 15 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 15).
- the human IL-22 polypeptide or fragment or variant thereof comprises a D71 G amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the human IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 15.
- the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 15.
- the human IL-22 polypeptide or fragment or variant thereof comprises a D71 L amino acid substitution mutation.
- the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 11 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 11 ).
- the human IL-22 polypeptide or fragment or variant thereof comprises a D71 L amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the human IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 11 .
- the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 11. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D71 N amino acid substitution mutation.
- the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 18 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 18).
- the human IL-22 polypeptide or fragment or variant thereof comprises a D71 N amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the human IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 18.
- the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 18.
- the human IL-22 polypeptide or fragment or variant thereof comprises a R73S amino acid substitution mutation.
- the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 16 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 16).
- the human IL-22 polypeptide or fragment or variant thereof comprises a R73S amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the human IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 16.
- the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 16.
- the human IL-22 polypeptide or fragment or variant thereof comprises a R73N amino acid substitution mutation.
- the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 22 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 22).
- the human IL-22 polypeptide or fragment or variant thereof comprises a R73N amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the human IL-22 polypeptide or fragment or variant thereof to SEQ ID NO:22
- the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 22.
- the human IL-22 polypeptide or fragment or variant thereof comprises a R73Q amino acid substitution mutation.
- the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 12 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 12).
- the human IL-22 polypeptide or fragment or variant thereof comprises a R73Q amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the human IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 12.
- the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 12.
- the human IL-22 polypeptide or fragment or variant thereof comprises a D168N amino acid substitution mutation.
- the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 19 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 19).
- the human IL-22 polypeptide or fragment or variant thereof comprises a D168N amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the human IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 19.
- the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 19.
- the human IL-22 polypeptide or fragment or variant thereof comprises a D168G amino acid substitution mutation.
- the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 20 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 20).
- the human IL-22 polypeptide or fragment or variant thereof comprises a D168G amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the human IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 20.
- the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 20.
- the human IL-22 polypeptide or fragment or variant thereof comprises D71 L and D168G amino acid substitution mutations.
- the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 13 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 13).
- the human IL-22 polypeptide or fragment or variant thereof comprises D71 L and D168 amino acid substitution mutations and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the human IL- 22 polypeptide or fragment or variant thereof to SEQ ID NO: 13.
- the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 13.
- the human IL-22 polypeptide or fragment or variant thereof comprises a R175S amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 21 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 21 ).
- the human IL-22 polypeptide or fragment or variant thereof comprises a R175S amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the human IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 21 .
- the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 21.
- the macromolecule comprises two first binding sites comprising an IL-22 polypeptide or fragment or variant thereof that binds IL-22Ra, IL-22BP, and/or IL-10R2.
- the two first binding sites each bind to IL-22Ra, each bind to IL-22BP, or each bind to IL- 10R2.
- the macromolecule comprises two second binding sites specific for an IL- 10 family address target expressed on the surface of the target cell. In some embodiments, the two second binding sites are specific for the same IL-10 family address target.
- the macromolecule is a homodimer comprising two subunits, each subunit comprising the first binding site and the second binding site.
- the two subunits are fusion proteins.
- each of the fusion proteins comprises a linker conjugated to the first binding site and the second binding site.
- the linker is a GS linker.
- the two subunits are substantially identical.
- the disclosure provides a macromolecule comprising two subunits, each comprising a first binding site and a second binding site, wherein (a) each of the first binding sites comprises an IL-22 polypeptide or a fragment or variant thereof that binds an IL-22 receptor on the surface of a target cell, and (b) each of the second binding sites is specific for an IL-10 family address target provided in Table 1 that is expressed on the surface of the target cell; wherein: (i) the second binding sites localize the first binding sites to the IL-10 family address target such that the IL-22 polypeptides or fragments or variants thereof influence signaling by the IL-22 receptor in the target cell; (ii) the second binding sites do not substantially influence signaling upon binding the IL-10 family address targets; and (iii) the IL-22 polypeptides or fragments or variants thereof do not substantially influence signaling by the IL-22 receptor in the absence of localization by the second binding sites.
- the two subunits are substantially identical.
- the disclosure provides a macromolecule comprising two subunits, each comprising a first binding site and a second binding site, wherein (a) each of the first binding sites comprises an IL-22 polypeptide or a fragment or variant thereof that binds an IL-22 receptor on the surface of a target cell, and (b) each of the second binding sites is specific for an IL-10 family address target selected from CDH17, CDH16, and GP2 that is expressed on the surface of the target cell; wherein (i) the second binding sites localize the first binding sites to the IL-10 family address target such that the IL-22 polypeptides or fragments or variants thereof influence signaling by the IL-22 receptor in the target cell; (ii) the second binding sites do not substantially influence signaling upon binding the IL-10 family address targets; and (iii) the IL-22 polypeptides or fragments or variants thereof do not substantially influence signaling by the IL-22 receptor in the absence of localization by the second binding sites.
- the two subunits each comprising a first
- the macromolecule comprises a single first binding site. In some embodiments, the macromolecule comprises a single second binding site. In some embodiments, the macromolecule is a heterodimer comprising two subunits, wherein the first subunit comprises the first binding site and second subunit comprises the second binding site. In some embodiments, the macromolecule is a fusion protein. In some embodiments, the fusion protein comprises a linker conjugated to the first binding site and the second binding site. In some embodiments, the linker is a GS linker.
- binding of the second binding site to the IL-10 family address target increases accumulation of the macromolecule in tissues expressing the IL-10 family address target, (ii) reduces accumulation of the macromolecule in tissues that do not express the IL-10 family address target, or (iii) both (i) and (ii).
- the disclosure provides a method of modulating IL-22 receptor signaling in a target cell of a subject, the method comprising administering to the subject the macromolecule of any one of the above aspects.
- the disclosure provides a macromolecule comprising (a) a first binding site comprising an IL-22 polypeptide or a fragment or variant thereof, and (b) a second binding site comprising an anti-CDH17 antibody or antigen-binding fragment thereof, wherein the macromolecule is a fusion protein, and wherein the first binding site and the second binding site are connected by a linker.
- the anti-CDH17 antibody or antigen-binding fragment thereof localizes the first binding site to an IL-22 receptor such that the IL-22 polypeptide or fragment or variant thereof influences signaling by the IL-22 receptor in the target cell; (ii) the anti-CDH17 antibody or antigen-binding fragment thereof does not substantially influence signaling upon binding CDH17; and (iii) the IL-22 polypeptide or fragment or variant thereof does not substantially influence signaling by the IL-22 receptor in the absence of localization by the anti-CDH17 antibody or antigen-binding fragment thereof.
- the disclosure provides a macromolecule comprising two subunits, each comprising a first binding site and a second binding site, wherein (a) each first binding site comprises an IL-22 polypeptide or a fragment or variant thereof, and (b) each second binding site comprises an anti- CDH17 antibody or antigen-binding fragment thereof, wherein the macromolecule is a fusion protein, and wherein the first binding site and the second binding site of each subunit are connected by a linker.
- the anti-CDH17 antibodies or antigen-binding fragments thereof localize the first binding sites to an IL-22 receptor such that the IL-22 polypeptides or fragments or variants thereof influence signaling by the IL-22 receptor in the target cell; (ii) the anti-CDH17 antibodies or antigenbinding fragments thereof do not substantially influence signaling upon binding CDH17; and (iii) the IL-22 polypeptides or fragments or variants thereof do not substantially influence signaling by the IL-22 receptor in the absence of localization by the anti-CDH17 antibodies or antigen-binding fragments thereof.
- the disclosure provides a method of modulating IL-22 receptor signaling in the small intestine or colon of a subject, the method comprising administering to the subject the macromolecule of either of the above two aspects.
- the disclosure provides a macromolecule comprising (a) a first binding site comprising an IL-22 polypeptide or a fragment or variant thereof, and (b) a second binding site comprising an anti-CDH16 antibody or antigen-binding fragment thereof, wherein the macromolecule is a fusion protein, and wherein the first binding site and the second binding site are connected by a linker.
- the anti-CDH16 antibody or antigen-binding fragment thereof localizes the first binding site to an IL-22 receptor such that the IL-22 polypeptide or fragment or variant thereof influences signaling by the IL-22 receptor in the target cell; (ii) the anti-CDH16 antibody or antigen-binding fragment thereof does not substantially influence signaling upon binding CDH16; and (iii) the IL-22 polypeptide or fragment or variant thereof does not substantially influence signaling by the IL-22 receptor in the absence of localization by the anti-CDH16 antibody or antigen-binding fragment thereof.
- the disclosure provides a macromolecule comprising two subunits, each comprising a first binding site and a second binding site, wherein (a) each first binding site comprises an IL-22 polypeptide or a fragment or variant thereof, and (b) each second binding site comprises an anti- CDH16 antibody or antigen-binding fragment thereof, wherein the macromolecule is a fusion protein, and wherein the first binding site and the second binding site of each subunit are connected by a linker.
- the anti-CDH16 antibodies or antigen-binding fragments thereof localize the first binding sites to an IL-22 receptor such that the IL-22 polypeptides or fragments or variants thereof influence signaling by the IL-22 receptor in the target cell; (ii) the anti-CDH16 antibodies or antigenbinding fragments thereof do not substantially influence signaling upon binding CDH16; and (iii) the IL-22 polypeptides or fragments or variants thereof do not substantially influence signaling by the IL-22 receptor in the absence of localization by the anti-CDH16 antibodies or antigen-binding fragments thereof.
- the disclosure provides a method of modulating IL-22 receptor signaling in the kidney of a subject, the method comprising administering to the subject the macromolecule of either of the above two aspects.
- the disclosure provides a macromolecule comprising (a) a first binding site comprising an IL-22 polypeptide or a fragment or variant thereof, and (b) a second binding site comprising an anti-GP2 antibody or antigen-binding fragment thereof, wherein the macromolecule is a fusion protein, and wherein the first binding site and the second binding site are connected by a linker.
- the anti-GP2 antibody or antigen-binding fragment thereof localizes the first binding site to an IL-22 receptor such that the IL-22 polypeptide or fragment or variant thereof influences signaling by the IL-22 receptor in the target cell; (ii) the anti-GP2 antibody or antigen-binding fragment thereof does not substantially influence signaling upon binding GP2; and (iii) the IL-22 polypeptide or fragment or variant thereof does not substantially influence signaling by the IL-22 receptor in the absence of localization by the anti-GP2 antibody or antigen-binding fragment thereof.
- the disclosure provides a macromolecule comprising two subunits, each comprising a first binding site and a second binding site, wherein (a) each first binding site comprises an IL-22 polypeptide or a fragment or variant thereof, and (b) each second binding site comprises an anti- GP2 antibody or antigen-binding fragment thereof, wherein the macromolecule is a fusion protein, and wherein the first binding site and the second binding site of each subunit are connected by a linker.
- the anti-GP2 antibodies or antigen-binding fragments thereof localize the first binding sites to an IL-22 receptor such that the IL-22 polypeptides or fragments or variants thereof influence signaling by the IL-22 receptor in the target cell; (ii) the anti-GP2 antibodies or antigen-binding fragments thereof do not substantially influence signaling upon binding GP2; and (iii) the IL-22 polypeptides or fragments or variants thereof do not substantially influence signaling by the IL-22 receptor in the absence of localization by the second binding sites.
- the disclosure provides a method of modulating IL-22 receptor signaling in the pancreas of a subject, the method comprising administering to the subject the macromolecule of either one of the above aspects.
- the IL-22 polypeptide or fragment or variant thereof is a human IL-22 or fragment or variant thereof.
- the IL-22 polypeptide or fragment or variant thereof is a human IL-22 polypeptide or fragment or variant thereof and comprises one or more of the amino acid substitution mutations of Table 6, Table 7A, Table 7B, Table 8, Table 9A, Table 9B, or Table 9C.
- the human IL-22 polypeptide or fragment or variant thereof comprises an amino acid substitution mutation of one or more of K61 , D71 , R73, D168, and R175. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a K61 amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D71 amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a R73 amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D168 amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a R175 amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D71 amino acid substitution mutation and a D168 amino acid substitution mutation. The amino acid positions are with reference to the IL-22 sequence of SEQ ID NO: 7.
- the human IL-22 polypeptide or fragment or variant thereof comprises one or more of a K61 S, D71 N, D71 L, D71 G, R73Q, R73S, R73N, D168N, D168G, and R175S amino acid substitution mutation.
- the human IL-22 polypeptide or fragment or variant thereof comprises a K61 S amino acid substitution mutation.
- the human IL-22 polypeptide or fragment or variant thereof comprises a D71 N amino acid substitution mutation.
- the human IL-22 polypeptide or fragment or variant thereof comprises a D71 L amino acid substitution mutation.
- the human IL-22 polypeptide or fragment or variant thereof comprises a D71 G amino acid substitution mutation.
- the human IL-22 polypeptide or fragment or variant thereof comprises a R73Q amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a R73S amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a R73N amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D168N amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D168G amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a R175S amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D71 L amino acid substitution mutation and a D168G amino acid substitution mutation. The amino acid position numbering is with reference to the IL- 22 sequence of SEQ ID NO: 7.
- the human IL-22 polypeptide or fragment or variant thereof comprises a K61 S amino acid substitution mutation.
- the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 17 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 17).
- the human IL-22 polypeptide or fragment or variant thereof comprises a K61 S amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the human IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 17.
- the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 17.
- the human IL-22 polypeptide or fragment or variant thereof comprises a D71 G amino acid substitution mutation.
- the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 15 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 15).
- the human IL-22 polypeptide or fragment or variant thereof comprises a D71 G amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the human IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 15.
- the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 15.
- the human IL-22 polypeptide or fragment or variant thereof comprises a D71 L amino acid substitution mutation.
- the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 11 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 11 ).
- the human IL-22 polypeptide or fragment or variant thereof comprises a D71 L amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the human IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 11 .
- the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 11. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D71 N amino acid substitution mutation.
- the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 18 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 18).
- the human IL-22 polypeptide or fragment or variant thereof comprises a D71 N amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the human IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 18.
- the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 18.
- the human IL-22 polypeptide or fragment or variant thereof comprises a R73S amino acid substitution mutation.
- the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 16 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 16).
- the human IL-22 polypeptide or fragment or variant thereof comprises a R73S amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the human IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 16.
- the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 16.
- the human IL-22 polypeptide or fragment or variant thereof comprises a R73N amino acid substitution mutation.
- the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 22 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 22).
- the human IL-22 polypeptide or fragment or variant thereof comprises a R73N amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the human IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 22
- the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 22.
- the human IL-22 polypeptide or fragment or variant thereof comprises a R73Q amino acid substitution mutation.
- the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 12 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 12).
- the human IL-22 polypeptide or fragment or variant thereof comprises a R73Q amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the human IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 12.
- the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 12.
- the human IL-22 polypeptide or fragment or variant thereof comprises a D168N amino acid substitution mutation.
- the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 19 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 19).
- the human IL-22 polypeptide or fragment or variant thereof comprises a D168N amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the human IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 19.
- the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 19.
- the human IL-22 polypeptide or fragment or variant thereof comprises a D168G amino acid substitution mutation.
- the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 20 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 20).
- the human IL-22 polypeptide or fragment or variant thereof comprises a D168G amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the human IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 20.
- the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 20.
- the human IL-22 polypeptide or fragment or variant thereof comprises D71 L and D168G amino acid substitution mutations.
- the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 13 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 13).
- the human IL-22 polypeptide or fragment or variant thereof comprises D71 L and D168 amino acid substitution mutations and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the human IL- 22 polypeptide or fragment or variant thereof to SEQ ID NO: 13.
- the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 13.
- the human IL-22 polypeptide or fragment or variant thereof comprises a R175S amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 21 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 21 ).
- the human IL-22 polypeptide or fragment or variant thereof comprises a R175S amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the human IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 21 .
- the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 21.
- the disclosure provides a macromolecule comprising a first binding site and a second binding site, wherein (a) the first binding site comprises an IL-22 polypeptide or a fragment or variant thereof that binds an IL-22 receptor on the surface of a target cell, and (b) the second binding site is specific for CDH17, e.g., that is expressed on the surface of the target cell; wherein (i) the second binding site localizes the first binding site to the CDH17 such that the IL-22 polypeptide or fragment or variant thereof influences signaling by the IL-22 receptor in the target cell; (ii) the second binding site does not substantially influence signaling upon binding the CDH17; and (iii) the IL-22 polypeptide or fragment or variant thereof does not substantially influence signaling by the IL-22 receptor in the absence of localization by the second binding site.
- the IL-22 polypeptide or fragment or variant thereof is a human IL-22 or fragment or variant thereof.
- the IL-22 polypeptide or fragment or variant thereof is a human IL-22 polypeptide or fragment or variant thereof and comprises one or more of the amino acid substitution mutations of Table 6, Table 7A, Table 7B, Table 8, Table 9A, Table 9B, or Table 9C.
- the human IL-22 polypeptide or fragment or variant thereof comprises an amino acid substitution mutation of one or more of K61 , D71 , R73, D168, and R175. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a K61 amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D71 amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a R73 amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D168 amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a R175 amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D71 amino acid substitution mutation and a D168 amino acid substitution mutation. The amino acid positions are with reference to the IL-22 sequence of SEQ ID NO: 7.
- the human IL-22 polypeptide or fragment or variant thereof comprises one or more of a K61 S, D71 N, D71 L, D71 G, R73Q, R73S, R73N, D168N, D168G, and R175S amino acid substitution mutation.
- the human IL-22 polypeptide or fragment or variant thereof comprises a K61 S amino acid substitution mutation.
- the human IL-22 polypeptide or fragment or variant thereof comprises a D71 N amino acid substitution mutation.
- the human IL-22 polypeptide or fragment or variant thereof comprises a D71 L amino acid substitution mutation.
- the human IL-22 polypeptide or fragment or variant thereof comprises a D71 G amino acid substitution mutation.
- the human IL-22 polypeptide or fragment or variant thereof comprises a R73Q amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a R73S amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a R73N amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D168N amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D168G amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a R175S amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D71 L amino acid substitution mutation and a D168G amino acid substitution mutation. The amino acid position numbering is with reference to the IL- 22 sequence of SEQ ID NO: 7.
- the human IL-22 polypeptide or fragment or variant thereof comprises a K61 S amino acid substitution mutation.
- the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 17 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 17).
- the human IL-22 polypeptide or fragment or variant thereof comprises a K61 S amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the human IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 17.
- the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 17.
- the human IL-22 polypeptide or fragment or variant thereof comprises a D71 G amino acid substitution mutation.
- the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 15 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 15).
- the human IL-22 polypeptide or fragment or variant thereof comprises a D71 G amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the human IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 15.
- the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 15.
- the human IL-22 polypeptide or fragment or variant thereof comprises a D71 L amino acid substitution mutation.
- the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 11 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 11 ).
- the human IL-22 polypeptide or fragment or variant thereof comprises a D71 L amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the human IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 11 .
- the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 11.
- the human IL-22 polypeptide or fragment or variant thereof comprises a D71 N amino acid substitution mutation.
- the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 18 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 18).
- the human IL-22 polypeptide or fragment or variant thereof comprises a D71 N amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the human IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 18.
- the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 18.
- the human IL-22 polypeptide or fragment or variant thereof comprises a R73S amino acid substitution mutation.
- the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 16 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 16).
- the human IL-22 polypeptide or fragment or variant thereof comprises a R73S amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the human IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 16.
- the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 16.
- the human IL-22 polypeptide or fragment or variant thereof comprises a R73N amino acid substitution mutation.
- the human IL-22 polypeptide or fragment or variant thereof comprises a R73N amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the human IL-22 polypeptide or fragment or variant thereof to SEQ ID NO:22
- the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 22. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a R73Q amino acid substitution mutation.
- the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 12 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 12).
- the human IL-22 polypeptide or fragment or variant thereof comprises a R73Q amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the human IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 12.
- the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 12.
- the human IL-22 polypeptide or fragment or variant thereof comprises a D168N amino acid substitution mutation.
- the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 19 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 19).
- the human IL-22 polypeptide or fragment or variant thereof comprises a D168N amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the human IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 19.
- the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 19.
- the human IL-22 polypeptide or fragment or variant thereof comprises a D168G amino acid substitution mutation.
- the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 20 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 20).
- the human IL-22 polypeptide or fragment or variant thereof comprises a D168G amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the human IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 20.
- the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 20.
- the human IL-22 polypeptide or fragment or variant thereof comprises D71 L and D168G amino acid substitution mutations.
- the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 13 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 13).
- the human IL-22 polypeptide or fragment or variant thereof comprises D71 L and D168 amino acid substitution mutations and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the human IL- 22 polypeptide or fragment or variant thereof to SEQ ID NO: 13.
- the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 13.
- the human IL-22 polypeptide or fragment or variant thereof comprises a R175S amino acid substitution mutation.
- the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 21 (e.g., has at least 91 %, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 21 ).
- the human IL-22 polypeptide or fragment or variant thereof comprises a R175S amino acid substitution mutation and has at least 90% identity (e.g., has at least 91 %, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the human IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 21 .
- the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 21 .
- the IL-22 polypeptide e.g., human IL-22 polypeptide
- the IL-22 polypeptide or fragment or variant thereof has a melting onset temperature (Tonset) of about 36°C to about 60 °C, e.g., as determined by a method described herein, e.g., as determined by differential scanning calorimetry (DSC).
- Tonset melting onset temperature
- the IL-22 polypeptide e.g., human IL-22 polypeptide
- the IL-22 polypeptide or fragment or variant thereof has a melting onset temperature (Tonset) of about 38°C, e.g., as determined by a method described herein, e.g., as determined by differential scanning calorimetry (DSC).
- Tonset melting onset temperature
- the IL-22 polypeptide e.g., human IL-22 polypeptide
- the IL-22 polypeptide or fragment or variant thereof has a melting onset temperature (Tonset) of about 50°C, e.g., as determined by a method described herein, e.g., as determined by differential scanning calorimetry (DSC).
- Tonset melting onset temperature
- the IL-22 polypeptide e.g., human IL-22 polypeptide
- the IL-22 polypeptide or fragment or variant thereof has a melting onset temperature (Tonset) of about 58°C, e.g., as determined by a method described herein, e.g., as determined by differential scanning calorimetry (DSC).
- Tonset melting onset temperature
- the IL-22 polypeptide e.g., human IL-22 polypeptide
- the IL-22 polypeptide or fragment or variant thereof has a binding affinity to human IL-22Ra of between about 1 E-8 to about 10E-9, e.g., as determined by a method described herein.
- the IL-22 polypeptide e.g., human IL-22 polypeptide
- the IL-22 polypeptide or fragment or variant thereof has a binding affinity to human IL-22Ra of about 7.7E-9, e.g., as determined by a method described herein.
- the IL-22 polypeptide e.g., human IL-22 polypeptide
- the IL-22 polypeptide has a binding affinity to human IL-22Ra of about 2.3E-8, e.g., as determined by a method described herein.
- the IL-22 polypeptide e.g., human IL-22 polypeptide
- the IL-22 polypeptide or fragment or variant thereof has a binding affinity to human IL-22Ra of about 1 .5E - 8, e.g., as determined by a method described herein.
- the IL-22 polypeptide e.g., human IL-22 polypeptide
- the IL-22 polypeptide or fragment or variant thereof has a binding affinity to human IL-22Ra of about 7.7E-8, e.g., as determined by a method described herein.
- the IL-22 polypeptide e.g., human IL-22 polypeptide
- the IL-22 polypeptide or fragment or variant thereof has a binding affinity to human IL-22Ra of about 10E-9, e.g., as determined by a method described herein.
- the IL-22 polypeptide e.g., human IL-22 polypeptide
- the IL-22 polypeptide or fragment or variant thereof has a binding affinity to human IL-22Ra of about 3.1 E-8, e.g., as determined by a method described herein.
- the IL-22 polypeptide e.g., human IL-22 polypeptide
- the IL-22 polypeptide or fragment or variant thereof has about a 1 .3-fold to about a10-fold decreased binding affinity to human IL-22Ra as compared to the binding affinity of wild type human IL-22 polypeptide to human IL-22Ra, e.g., as determined by a method described herein.
- the IL-22 polypeptide e.g., human IL-22 polypeptide
- the IL-22 polypeptide or fragment or variant thereof has about a 3-fold decreased binding affinity to human IL-22Ra as compared to the binding affinity of wild type human IL-22 polypeptide to human IL-22Ra, e.g., as determined by a method described herein.
- the IL-22 polypeptide e.g., human IL-22 polypeptide
- the IL-22 polypeptide or fragment or variant thereof has about a 2-fold decreased binding affinity to human IL-22Ra as compared to the binding affinity of wild type human IL-22 polypeptide to human IL-22Ra, e.g., as determined by a method described herein.
- the IL-22 polypeptide e.g., human IL-22 polypeptide
- the IL-22 polypeptide or fragment or variant thereof has about a 10-fold decreased binding affinity to human IL-22Ra as compared to the binding affinity of wild type human IL-22 polypeptide to human IL-22Ra, e.g., as determined by a method described herein.
- the IL-22 polypeptide e.g., human IL-22 polypeptide
- the IL-22 polypeptide or fragment or variant thereof has about a 1 .3-fold decreased binding affinity to human IL-22Ra as compared to the binding affinity of wild type human IL-22 polypeptide to human IL-22Ra, e.g., as determined by a method described herein.
- the IL-22 polypeptide e.g., human IL-22 polypeptide
- the IL-22 polypeptide or fragment or variant thereof has about a 4-fold decreased binding affinity to human IL-22Ra as compared to the binding affinity of wild type human IL-22 polypeptide to human IL-22Ra, e.g., as determined by a method described herein.
- the disclosure provides an IL-22 polypeptide or a fragment or variant thereof (e.g., an IL-22 mutein polypeptide or fragment or variant thereof, e.g., an IL-22 mutein polypeptide or fragment or variant thereof that is derived from human IL-22) that binds an IL-22 receptor on the surface of a target cell.
- the IL-22 polypeptide or a fragment or variant thereof binds the IL-22 receptor.
- the IL-22 polypeptide or fragment or variant thereof binds the human IL-22 receptor.
- the IL-22 polypeptide or fragment or variant thereof binds human IL-22Ra and/or IL-10R2.
- the IL-22 polypeptide or fragment or variant thereof binds IL-22BP. In some embodiments, the IL-22 polypeptide or fragment or variant thereof binds human IL-22BP.
- the IL-22 polypeptide or fragment or variant thereof agonizes the IL-22 receptor. In some embodiments, the IL-22 polypeptide or fragment or variant thereof agonizes the human IL-22 receptor. In some embodiments, the IL-22 polypeptide or fragment or variant thereof agonizes human IL-22Ra and/or IL-10R2, e.g., in an assay described herein.
- the IL-22 polypeptide or fragment or variant thereof agonizes IL-22BP. In some embodiments, the IL-22 polypeptide or fragment or variant thereof agonizes human IL-22BP.
- the IL-22 polypeptide or fragment or variant thereof has low (e.g., lower) binding affinity for IL-22Ra and/or IL-10R2, as compared to the binding affinity of wild type human IL-22 (e.g., of SEQ ID NO: 8) for the IL-22 receptor, e.g., in an assay described herein.
- the IL-22 polypeptide or fragment or variant thereof has low (e.g., lower) binding affinity for IL-22BP, as compared to the binding affinity of wild type human IL-22 (e.g., of SEQ ID NO: 8) for IL-2BP, e.g., in an assay described herein.
- the IL-22 polypeptide or fragment or variant thereof has been engineered to have reduced binding affinity for the IL-22 receptor (e.g., for one or both subunits of the receptor, e.g., IL-22Ra and/or IL-10R2), e.g., as compared to the binding affinity of wild type human IL-22 (e.g., of SEQ ID NO: 8) for the IL-22 receptor, e.g., in an assay described herein.
- the IL-22 receptor e.g., for one or both subunits of the receptor, e.g., IL-22Ra and/or IL-10R2
- wild type human IL-22 e.g., of SEQ ID NO: 8
- the binding affinity for the IL-22 receptor of the engineered IL-22 polypeptide or fragment or variant thereof ranges from minimally denatured (e.g., ⁇ 10-fold decreased) to hundreds of fold weakened, e.g., at least 300x decreased, e.g., as measured by biolayer interferometry (BLI), e.g., on a GATOR® instrument. See also the examples provided herein.
- minimally denatured e.g., ⁇ 10-fold decreased
- hundreds of fold weakened e.g., at least 300x decreased, e.g., as measured by biolayer interferometry (BLI), e.g., on a GATOR® instrument.
- the IL-22 polypeptide or fragment or variant thereof comprises one or more amino acid substitution mutations at one or more residues that are predicted to be interaction surfaces between IL-22 and IL-22Ra and/or IL-10R2.
- the IL-22 polypeptide or fragment or variant thereof has reduced binding affinity for IL22RA1 (also referred to as IL-22RA or IL-22Ra) relative to a wild-type IL-22 or fragment thereof.
- the IL-22 polypeptide or fragment or variant thereof comprises one or more amino acid substitution mutations at one or more residues that are predicted to be interaction surfaces between IL-22 and IL-22Ra.
- the IL-22 polypeptide or fragment or variant thereof is a human IL-22 or fragment or variant thereof.
- the IL-22 polypeptide or fragment or variant thereof is a human IL-22 or fragment or variant thereof and comprises one or more of the amino acid substitution mutations of Table 6, Table 7A, Table 7B, Table 8, Table 9A, Table 9B, or Table 9C.
- the human IL-22 polypeptide or fragment or variant thereof comprises an amino acid substitution mutation of one or more of K61 , D71 , R73, D168, and R175.
- the human IL-22 polypeptide or fragment or variant thereof comprises a K61 amino acid substitution mutation.
- the human IL-22 polypeptide or fragment or variant thereof comprises a D71 amino acid substitution mutation.
- the human IL-22 polypeptide or fragment or variant thereof comprises a R73 amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D168 amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a R175 amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D71 amino acid substitution mutation and a D168 amino acid substitution mutation. The amino acid position numbering is with reference to the IL-22 sequence of SEQ ID NO: 7.
- the human IL-22 polypeptide or fragment or variant thereof comprises one or more of a K61 S, D71 N, D71 L, D71 G, R73Q, R73S, R73N, D168N, D168G, and R175S amino acid substitution mutation.
- the human IL-22 polypeptide or fragment or variant thereof comprises a K61 S amino acid substitution mutation.
- the human IL-22 polypeptide or fragment or variant thereof comprises a D71 N amino acid substitution mutation.
- the human IL-22 polypeptide or fragment or variant thereof comprises a D71 L amino acid substitution mutation.
- the human IL-22 polypeptide or fragment or variant thereof comprises a D71 G amino acid substitution mutation.
- the human IL-22 polypeptide or fragment or variant thereof comprises a R73Q amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a R73S amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a R73N amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D168N amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D168G amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a R175S amino acid substitution mutation.
- the human IL-22 polypeptide or fragment or variant thereof comprises a D71 L amino acid substitution mutation and a D168G amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D71 G amino acid substitution mutation and a D168G amino acid substitution mutation.
- the amino acid position numbering is with reference to the IL-22 sequence of SEQ ID NO: 7.
- the human IL-22 polypeptide or fragment or variant thereof comprises a K61 S amino acid substitution mutation.
- the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 17 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 17).
- the human IL-22 polypeptide or fragment or variant thereof comprises a K61 S amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the human IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 17.
- the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 17.
- the human IL-22 polypeptide or fragment or variant thereof comprises a D71 G amino acid substitution mutation.
- the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 15 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 15).
- the human IL-22 polypeptide or fragment or variant thereof comprises a D71 G amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the human IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 15.
- the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 15.
- the human IL-22 polypeptide or fragment or variant thereof comprises a D71 L amino acid substitution mutation.
- the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 11 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 11 ).
- the human IL-22 polypeptide or fragment or variant thereof comprises a D71 L amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the human IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 11 .
- the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 11.
- the human IL-22 polypeptide or fragment or variant thereof comprises a D71 N amino acid substitution mutation.
- the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 18 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 18).
- the human IL-22 polypeptide or fragment or variant thereof comprises a D71 N amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the human IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 18.
- the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 18.
- the human IL-22 polypeptide or fragment or variant thereof comprises a R73S amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 16 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 16).
- the human IL-22 polypeptide or fragment or variant thereof comprises a R73S amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the human IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 16.
- the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 16.
- the human IL-22 polypeptide or fragment or variant thereof comprises a R73N amino acid substitution mutation.
- the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 22 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 22).
- the human IL-22 polypeptide or fragment or variant thereof comprises a R73N amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the human IL-22 polypeptide or fragment or variant thereof to SEQ ID NO:22
- the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 22.
- the human IL-22 polypeptide or fragment or variant thereof comprises a R73Q amino acid substitution mutation.
- the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 12 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 12).
- the human IL-22 polypeptide or fragment or variant thereof comprises a R73Q amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the human IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 12.
- the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 12.
- the human IL-22 polypeptide or fragment or variant thereof comprises a D168N amino acid substitution mutation.
- the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 19 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 19).
- the human IL-22 polypeptide or fragment or variant thereof comprises a D168N amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the human IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 19.
- the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 19.
- the human IL-22 polypeptide or fragment or variant thereof comprises a D168G amino acid substitution mutation.
- the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 20 (e.g., has at least 91 %, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 20).
- the human IL-22 polypeptide or fragment or variant thereof comprises a D168G amino acid substitution mutation and has at least 90% identity (e.g., has at least 91 %, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the human IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 20.
- the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 20.
- the human IL-22 polypeptide or fragment or variant thereof comprises D71 L and D168G amino acid substitution mutations.
- the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 13 (e.g., has at least 91 %, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 13).
- the human IL-22 polypeptide or fragment or variant thereof comprises D71 L and D168 amino acid substitution mutations and has at least 90% identity (e.g., has at least 91 %, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the human IL- 22 polypeptide or fragment or variant thereof to SEQ ID NO: 13.
- the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 13.
- the human IL-22 polypeptide or fragment or variant thereof comprises a R175S amino acid substitution mutation.
- the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 21 (e.g., has at least 91 %, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 21 ).
- the human IL-22 polypeptide or fragment or variant thereof comprises a R175S amino acid substitution mutation and has at least 90% identity (e.g., has at least 91 %, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the human IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 21 .
- the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 21 .
- the IL-22 polypeptide or fragment or variant thereof has a melting onset temperature (Tonset) of about 36°C to about 60 °C, e.g., as determined by a method described herein, e.g., as determined by differential scanning calorimetry (DSC). In some embodiments of an IL-22 polypeptide or fragment or variant thereof provided herein, the IL-22 polypeptide or fragment or variant thereof has a melting onset temperature (Tonset) of about 38°C, e.g., as determined by a method described herein, e.g., as determined by differential scanning calorimetry (DSC).
- DSC differential scanning calorimetry
- the IL-22 polypeptide or fragment or variant thereof has a melting onset temperature (Tonset) of about 50°C, e.g., as determined by a method described herein, e.g., as determined by differential scanning calorimetry (DSC).
- Tonset melting onset temperature
- the IL-22 polypeptide or fragment or variant thereof has a melting onset temperature (Tonset) of about 58°C, e.g., as determined by a method described herein, e.g., as determined by differential scanning calorimetry (DSC).
- Tonset melting onset temperature
- the IL-22 polypeptide or fragment or variant thereof has a binding affinity to human IL-22Ra of between about 1 E-8 to about 10E-9, e.g., as determined by a method described herein.
- the IL-22 polypeptide or fragment or variant thereof has a binding affinity to IL-22Ra of about 7.7E-9, e.g., as determined by a method described herein.
- the IL-22 polypeptide or fragment or variant thereof has a binding affinity to IL-22Ra of about 2.3E-8, e.g., as determined by a method described herein.
- the IL-22 polypeptide or fragment or variant thereof has a binding affinity to IL-22Ra of about 1 .5E -8, e.g., as determined by a method described herein.
- the IL-22 polypeptide or fragment or variant thereof has a binding affinity to human IL-22Ra of about 7.7E-8, e.g., as determined by a method described herein.
- the IL-22 polypeptide or fragment or variant thereof has a binding affinity to human IL-22Ra of about 10E-9, e.g., as determined by a method described herein.
- the IL-22 polypeptide or fragment or variant thereof has a binding affinity to human IL-22Ra of about 3.1 E-8, e.g., as determined by a method described herein.
- the IL-22 polypeptide or fragment or variant thereof has about a 1 .3-fold to about a10-fold decreased binding affinity to human IL-22Ra as compared to the binding affinity of wild type human IL-22 polypeptide to human IL-22Ra, e.g., as determined by a method described herein.
- the IL-22 polypeptide or fragment or variant thereof has about a 3-fold decreased binding affinity to human IL-22Ra as compared to the binding affinity of wild type human IL-22 polypeptide to human IL-22Ra, e.g., as determined by a method described herein. In some embodiments of an IL-22 polypeptide or fragment or variant thereof provided herein, the IL-22 polypeptide or fragment or variant thereof has about a 2-fold decreased binding affinity to human IL-22Ra as compared to the binding affinity of wild type human IL-22 polypeptide to human IL-22Ra, e.g., as determined by a method described herein.
- the IL-22 polypeptide or fragment or variant thereof has about a 10-fold decreased binding affinity to human IL-22Ra as compared to the binding affinity of wild type human IL-22 polypeptide to human IL-22Ra, e.g., as determined by a method described herein.
- the IL-22 polypeptide or fragment or variant thereof has about a 1 .3-fold decreased binding affinity to human IL-22Ra as compared to the binding affinity of wild type human IL-22 polypeptide to human IL-22Ra, e.g., as determined by a method described herein.
- the IL-22 polypeptide or fragment or variant thereof has about a 4-fold decreased binding affinity to human IL-22Ra as compared to the binding affinity of wild type human IL-22 polypeptide to human IL-22Ra, e.g., as determined by a method described herein.
- the disclosure provides a polypeptide comprising a human IL-22 polypeptide or fragment or variant thereof provided herein.
- the disclosure provides a composition comprising any one of the polypeptides provided herein.
- the composition comprises a pharmaceutically acceptable carrier or excipient.
- the composition is a unit dosage form suitable for administration to a human subject.
- the disclosure provides one or more nucleic acids encoding any one of the polypeptides provided herein.
- the nucleic acids are linear nucleic acids, circular nucleic acids, and/or modified nucleic acids.
- the disclosure provides one or more vectors comprising the one or more nucleic acids of the above aspect.
- the disclosure provides one or more host cells comprising the one or more vectors of the above aspect.
- the disclosure provides a method of producing any one of the polypeptides provided herein, the method comprising culturing the one or more host cells of the above aspect in a culture medium. In some embodiments, the method further comprises recovering the polypeptide from the one or more host cells or the culture medium.
- the disclosure provides a polypeptide comprising an IL-22 polypeptide or a fragment or variant thereof provided herein, and further comprises an Fc region.
- the Fc region is N terminal to the IL-22 polypeptide or fragment or variant thereof.
- the Fc region is C terminal to the IL-22 polypeptide or fragment or variant thereof.
- the disclosure provides a composition comprising any one of the macromolecules provided herein.
- the composition comprises a pharmaceutically acceptable carrier or excipient.
- the composition is a unit dosage form suitable for administration to a human subject.
- the disclosure provides one or more nucleic acids encoding any one of the macromolecules provided herein or one or more subunits thereof.
- the nucleic acids are linear nucleic acids, circular nucleic acids, and/or modified nucleic acids.
- the disclosure provides one or more vectors comprising the one or more nucleic acids of the above aspect.
- the disclosure provides one or more host cells comprising the one or more vectors of the above aspect.
- the disclosure provides a method of producing any one of the macromolecules provided herein, the method comprising culturing the one or more host cells of the above aspect in a culture medium. In some embodiments, the method further comprises recovering the macromolecule from the one or more host cells or the culture medium.
- the disclosure provides a method comprising administering a therapeutically effective amount of any one of the macromolecules, compositions, polypeptides, or nucleic acids provided herein to a subject in need thereof.
- the subject has, is suspected of having, or is at increased risk of developing a disease or disorder selected from: Crohn's disease, ulcerative colitis, graft-vs-host disease, atopic dermatitis, psoriasis, wounds, diabetic skin ulcers, acute pancreatitis, chronic pancreatitis, autoimmune pancreatitis, nephritis, acute lung injury, viral lung injury, fibrosis, non-alcoholic steatohepatitis, viral hepatitis, and autoimmune hepatitis.
- the disease or disorder comprises Crohn's disease.
- the disease or disorder comprises ulcerative colitis.
- the disclosure provides a pharmaceutical composition
- a pharmaceutical composition comprising a pharmaceutically acceptable excipient and a macromolecule, a composition, a polypeptide, or a nucleic acid of any one of the above aspects.
- pharmaceutically acceptable excipient means a pharmaceutically acceptable material, or vehicle, such as a liquid or solid filter, diluent, excipient, solvent or encapsulating material useful for formulating a macromolecule complex, macromolecule, nucleic acid, or pair of nucleic acids for medicinal or therapeutic use.
- the disclosure provides the use of one or more of the macromolecule complexes, macromolecules, compositions, pharmaceutical compositions, polypeptides, or nucleic acids of any one of the above aspects in the manufacture of a medicament for the treatment of a subject.
- the subject has, is suspected of having, or is at increased risk of developing a disease or disorder selected from: Crohn's disease, ulcerative colitis, graft-vs-host disease, atopic dermatitis, psoriasis, wounds, diabetic skin ulcers, acute pancreatitis, chronic pancreatitis, autoimmune pancreatitis, nephritis, acute lung injury, viral lung injury, fibrosis, non-alcoholic steatohepatitis, viral hepatitis, and autoimmune hepatitis.
- the disease or disorder comprises Crohn's disease.
- the disease or disorder comprises ulcerative colitis.
- the disclosure provides one or more of the macromolecule complexes, macromolecules, compositions, pharmaceutical compositions, polypeptides, or nucleic acids of any one of the above aspects for use in treating a subject.
- the subject has, is suspected of having, or is at increased risk of developing a disease or disorder selected from: Crohn's disease, ulcerative colitis, graft-vs-host disease, atopic dermatitis, psoriasis, wounds, diabetic skin ulcers, acute pancreatitis, chronic pancreatitis, autoimmune pancreatitis, nephritis, acute lung injury, viral lung injury, fibrosis, non-alcoholic steatohepatitis, viral hepatitis, and autoimmune hepatitis.
- the disease or disorder comprises Crohn's disease.
- the disease or disorder comprises ulcerative colitis.
- Fig. 1 A is a schematic diagram showing the design of a monovalent ANDbody that simultaneously binds to an IL-22 receptor and an antibody target (“monovalent IL-22/mAb”).
- Fab fragment antigen-binding region.
- Fig. 1B is a schematic diagram showing the design of a divalent ANDbody that simultaneously binds to an IL-22 receptor and an antibody target (“divalent IL-22/mAb”).
- Fig. 2 is a chart showing the concentration of the indicated ANDbodies and control monoclonal antibodies (mAbs) in serum from treated mice over time.
- Fig. 3A is a plot showing the level of a monovalent anti-mCDH17/anti-IL-22 ANDbody (ng/mg total protein) in the indicated tissue samples from treated mice at the indicated timepoints.
- Fig. 3B is a plot showing the level of a monovalent anti-RSV/anti-IL-22 ANDbody (ng/mg total protein) in the indicated tissue samples from treated mice at the indicated timepoints.
- Fig. 4A is a set of bar graphs showing the levels of the IL-22 target genes REG3B, REG3G and SOCS3 in small intestine tissue from mice treated with monovalent anti-mCDH17/IL-22 and monovalent anti-RSV/IL-22 ANDbodies at the indicated concentrations (fold change relative to phosphate-buffered saline (PBS) control).
- PBS phosphate-buffered saline
- Fig. 4B is a set of bar graphs showing the levels of the IL-22 target genes REG3B, REG3G and SOCS3 in colon tissue from mice treated with monovalent anti-mCDH17/IL-22 and monovalent anti- RSV/IL-22 ANDbodies at the indicated concentrations (fold change relative to PBS control).
- Fig. 4C is a set of bar graphs showing the levels of the IL-22 target genes REG3B, REG3G and SOCS3 in pancreas tissue from mice treated with monovalent anti-mCDH17/IL-22 and monovalent anti- RSV/IL-22 ANDbodies at the indicated concentrations (fold change relative to PBS control).
- Fig. 4D is a set of bar graphs showing the levels of the IL-22 target genes REG3B, REG3G and SOCS3 in liver tissue from mice treated with monovalent anti-mCDH17/IL-22 and monovalent anti- RSV/IL-22 ANDbodies at the indicated concentrations (fold change relative to PBS control).
- Fig. 5 is a chart showing the concentration of ANDbodies comprising the indicated amino acid substitution mutations in the IL-22 polypeptide in serum from treated mice over time.
- Fig. 6A is a set of bar graphs showing the levels of the IL-22 target genes REG3B, REG3G and SOCS3 in small intestine tissue from mice treated with divalent anti-mCDH17/IL-22 and divalent anti- RSV/IL-22 ANDbodies comprising the indicated amino acid substitution mutations in the IL-22 polypeptide at the indicated concentrations (fold change relative to PBS control).
- Fig. 6B is a set of bar graphs showing the levels of the IL-22 target genes REG3B, REG3G and
- Fig. 6C is a set of bar graphs showing the levels of the IL-22 target genes REG3B, REG3G and SOCS3 in pancreas tissue from mice treated with divalent anti-mCDH17/IL-22 and divalent anti-RSV/IL- 22 ANDbodies comprising the indicated amino acid substitution mutations in the IL-22 polypeptide at the indicated concentrations (fold change relative to PBS control).
- Fig. 6D is a set of bar graphs showing the levels of the IL-22 target genes REG3B, REG3G and SOCS3 in liver tissue from mice treated with divalent anti-mCDH17/IL-22 and divalent anti-RSV/IL-22 ANDbodies comprising the indicated amino acid substitution mutations in the IL-22 polypeptide at the indicated concentrations (fold change relative to PBS control).
- Fig. 7A is a set of plots showing the levels of the IL-22 target genes REG3G, REG3B, and SOCS3 in small intestine tissue from mice treated with divalent anti-mCDH17/IL-22 or divalent anti- RSV/IL-22 ANDbodies comprising native IL-22 or IL-22 comprising the indicated amino acid substitution mutations at the indicated concentrations (fold change relative to PBS control).
- Fig. 7B is a set of plots showing the levels of the IL-22 target genes REG3G, REG3B, and SOCS3 in colon tissue from mice treated with divalent anti-mCDH17/IL-22 or divalent anti-RSV/IL-22 ANDbodies comprising native IL-22 or IL-22 comprising the indicated amino acid substitution mutations at the indicated concentrations (fold change relative to PBS control).
- Fig. 7C is a set of plots showing the levels of the IL-22 target genes REG3G, REG3B, and SOCS3 in pancreas tissue from mice treated with divalent anti-mCDH17/IL-22 or divalent anti-RSV/IL-22 ANDbodies comprising native IL-22 or IL-22 comprising the indicated amino acid substitution mutations at the indicated concentrations (fold change relative to PBS control).
- Fig. 7D is a set of plots showing the levels of the IL-22 target genes MT1 and MT2 in liver tissue from mice treated with divalent anti-mCDH17/IL-22 or divalent anti-RSV/IL-22 ANDbodies comprising native IL-22 or IL-22 comprising the indicated amino acid substitution mutations at the indicated concentrations (fold change relative to PBS control).
- Fig. 8A is a plot showing receptor occupancy for a range of binding affinities of IL-22 and CDH17 in the disease compartment of a four-compartment model.
- Fig. 8B is a plot showing receptor occupancy for a range of binding affinities of IL-22 and CDH17 in the toxicity (tox) compartment of a four-compartment model.
- Fig. 9A is a plot showing the induction levels of the IL-22 target genes REG3G and REG3B in pancreas tissue from mice treated with divalent anti-mGP2/IL-22 or divalent anti-RSV/IL-22 ANDbody proteins comprising native IL-22 or IL-22 mutein (D71 L / D168G). Expression was quantified as foldchange relative to PBS control. ANDbodies were administered at 0.3, 0.1 , or 0.03 milligrams per kilogram of body weight (MPK), as illustrated by the triangular concentration ramps.
- MPK milligrams per kilogram of body weight
- Fig. 9B is a plot showing the induction levels of the IL-22 target genes REG3G and REG3B in small intestine tissue from mice treated with divalent anti-mGP2/IL-22 or divalent anti-RSV/IL-22 ANDbody proteins comprising native IL-22 or IL-22 mutein (D71 L / D168G). Expression was quantified as fold-change relative to PBS control. ANDbodies were administered at 0.3, 0.1 , or 0.03 MPK, as illustrated by the triangular concentration ramps. Fig.
- 9C is a plot showing the induction levels of the IL-22 target genes REG3G and REG3B in colon tissue from mice treated with divalent anti-mGP2/IL-22 or divalent anti-RSV/IL-22 ANDbody proteins comprising native IL-22 or IL-22 mutein (D71 L / D168G). Expression was quantified as foldchange relative to PBS control. ANDbodies were administered at 0.3, 0.1 , or 0.03 MPK, as illustrated by the triangular concentration ramps.
- Fig. 10A is a plot showing the change in body weight of mice during a dextran sulfate sodium (DSS)-induced acute colitis study. Mice were administered DSS in drinking water as indicated, and ANDbody molecules or murine IL-22-Fc (mulL22-Fc) were administered intraperitoneally (I.P.) at days -1 , 1 , 4, and 6. Body weight was recorded daily, and plotted as % change in body weight.
- DSS dextran sulfate sodium
- Fig. 10B is a plot showing the disease activity index (DAI) of mice during the DSS-induced acute colitis study described in Fig. 10A.
- DAI was composed as body weight change, diarrhea, and hematochezia.
- Fig. 11 A is a graph showing the thermal stability (as determined by differential scanning calorimetry (DSC)) of an IL-22 mutein polypeptide comprising D71 L and D168G amino acid substitution mutations.
- Fig. 11B is a graph showing the thermal stability (as determined by DSC) of an IL-22 mutein polypeptide comprising an R73Q amino acid substitution mutation.
- Fig. 11C is a graph showing the thermal stability (as determined by DSC) of an IL-22 mutein polypeptide comprising an R73S amino acid substitution mutation.
- Fig. 12 is a graph showing levels of phosphorylated STAT3 (pSTAT3) on HaCaT (human keratinocyte) cells that were treated with native IL-22 or IL-22 mutein polypeptides comprising D71 L / D168G, R73Q, or R73S amino acid substitution mutations.
- pSTAT3 phosphorylated STAT3
- ANDbodyTM molecules that include a therapeutic IL-22 polypeptide or fragment or variant thereof and an IL-10 family address target binding domain.
- the therapeutic IL-22 polypeptide or fragment or variant thereof on the ANDbody molecule productively engages its therapeutic effector target only if the IL-10 family address target binding domain also engages an IL-10 family address target (i.e., a cell surface moiety displayed on the surface of a cell that also expresses the IL-22 receptor) on a target tissue or cell to localize the IL-22 polypeptide or fragment or variant thereof to the targeted cell or tissue, e.g., to form an AND-gate type of logic gate.
- an IL-10 family address target i.e., a cell surface moiety displayed on the surface of a cell that also expresses the IL-22 receptor
- an ANDbody is a macromolecule comprising at least (a) a first binding site comprising an IL-22 polypeptide or fragment or variant thereof that binds an IL-22 receptor on the surface of a target cell (e.g., wherein the IL-10 family receptor is broadly expressed in a mammalian subject); and (b) a second binding site specific for an IL-10 family address target.
- expression of the address target is restricted in vivo in a subject.
- the binding of a first binding site to a therapeutic effector target is weaker than the binding of the second binding site to the address marker.
- the IL-22 receptor and the IL-10 family address target are on the same cell.
- IL-22 mutein polypeptides or fragments or variants thereof that have altered (e.g., decreased) affinity for the IL-22 receptor.
- an ANDbody can be any macromolecule, such as a polypeptide or protein, that contains both an effector target binding site or binding domain, and an address target binding site or binding domain.
- the binding sites may be present on the same polypeptide chain or different polypeptide chains that are linked together, e.g., through disulfide bonds.
- the disclosure provides a macromolecule (i.e., an ANDbody) comprising a first binding site and a second binding site, wherein (a) the first binding site comprises an IL-22 polypeptide or a fragment or variant thereof that binds an IL-22 receptor on the surface of a target cell, and (b) the second binding site is specific for an IL-10 family address target provided in Table 1 that is expressed on the surface of the target cell; wherein (i) the second binding site localizes the first binding site to the IL- 10 family address target such that the IL-22 polypeptide or fragment or variant thereof influences signaling by the IL-22 receptor in the target cell; (ii) the second binding site does not substantially influence signaling upon binding the IL-10 family address target; and (iii) the IL-22 polypeptide or fragment or variant thereof does not substantially influence signaling by the IL-22 receptor in the absence of localization by the second binding site.
- a macromolecule i.e., an ANDbody
- the disclosure provides a macromolecule (i.e., an ANDbody) comprising a first binding site and a second binding site, wherein (a) the first binding site comprises an IL-22 polypeptide or a fragment or variant thereof that binds an IL-22 receptor on the surface of a target cell, and (b) the second binding site is specific for an IL-10 family address target selected from CDH17, CDH16, and GP2 that is expressed on the surface of the target cell; wherein (i) the second binding site localizes the first binding site to the IL-10 family address target such that the IL-22 polypeptide or fragment or variant thereof influences signaling by the IL-22 receptor in the target cell; (ii) the second binding site does not substantially influence signaling upon binding the IL-10 family address target; and (iii) the IL-22 polypeptide or fragment or variant thereof does not substantially influence signaling by the IL-22 receptor in the absence of localization by the second binding site.
- a macromolecule i.e., an ANDbody
- An ANDbody as disclosed herein, can be linked to an additional moiety or moieties, e.g., an extracellular component, an intracellular component, a soluble factor (e.g., an enzyme, hormone, cytokine, growth factor, toxin, venom, pollutant, etc.), or a transmembrane protein (e.g., a cell surface receptor).
- a soluble factor e.g., an enzyme, hormone, cytokine, growth factor, toxin, venom, pollutant, etc.
- a transmembrane protein e.g., a cell surface receptor
- first binding site and second binding site in the ANDbody are directly joined to each other.
- directly joined is meant that the first binding site coding sequences abut the second binding site coding sequences and no sequences derived from other sequences (such as linkers) are present.
- the first binding site and second binding site in the ANDbody are not directly joined to each other.
- ANDbodies of the present technology include binding domains that bind address target or effector target proteins.
- binding domains of the present ANDbodies may bind protein sequences that include a signal peptide.
- binding domains of the present ANDbodies may bind proteins that lack a signal protein.
- binding domains of the present ANDbodies may bind full-length proteins.
- binding domains of the present ANDbodies may bind protein fusions, such as full-length protein sequences, or peptide fragments thereof, with or without signal peptide regions, fused to other proteins, such as, for example, Fc sequences. Binding domains of the present ANDbodies may bind proteins that comprise less than the full-length protein sequence, such as a peptide fragment of the address target or effector target.
- the macromolecule comprises two first binding sites comprising an IL-22 polypeptide or a fragment or variant thereof that binds an IL-22 receptor.
- the two first binding sites bind to the same subunit or subunits of an IL-22 family receptor (e.g., both bind to IL- 22Ra, both bind to IL-22BP, or both bind to IL-10R2).
- the two first binding sites comprise the same IL-22 polypeptide or fragment or variant thereof and/or are substantially identical in sequence.
- the two first binding sites comprise different fragments or variants of the same IL-22 polypeptide or fragment or variant thereof, e.g., comprise fragments or variants having different affinity for the IL-22 receptor.
- the two first binding sites may comprise different IL-22 polypeptides or fragments or variants thereof and/or bind to different subunits of an IL-22 receptor.
- the macromolecule comprises two second binding sites specific for an IL-10 family address target expressed on the surface of the target cell.
- the two second binding sites are specific for the same IL-10 family address target.
- the two second binding sites are substantially identical in sequence.
- the two second binding sites may bind to two different IL-10 family address targets.
- the macromolecule is a homodimer comprising two subunits, each subunit comprising the first binding site and the second binding site (e.g., wherein the two subunits of the homodimer are substantially identical (e.g., are substantially identical in sequence)).
- each subunit of the homodimer is a fusion protein (e.g., a fusion protein comprising a linker (e.g., a GS linker) conjugated to the first binding site and the second binding site).
- the disclosure provides a macromolecule comprising two subunits, each comprising a first binding site and a second binding site, wherein: (a) each of the first binding sites comprises an IL-22 polypeptide or a fragment or variant thereof that binds an IL-22 receptor on the surface of a target cell, and (b) each of the second binding sites is specific for an IL-10 family address target provided in Table 1 that is expressed on the surface of the target cell; wherein (i) the second binding sites localize the first binding sites to the IL-10 family address target such that the IL-22 polypeptides or fragments or variants thereof influence signaling by the IL-22 receptor in the target cell; (ii) the second binding sites do not substantially influence signaling upon binding the IL-10 family address targets; and (Hi) the IL-22 polypeptides or fragments or variants thereof do not substantially influence signaling by the IL-22 receptor in the absence of localization by the second binding sites.
- the two subunits are substantially identical.
- the disclosure provides a macromolecule comprising two subunits, each comprising a first binding site and a second binding site, wherein (a) each of the first binding sites comprises an IL-22 polypeptide or a fragment or variant thereof that binds an IL-22 receptor on the surface of a target cell, and (b) each of the second binding sites is specific for an IL-10 family address target selected from CDH17, CDH16, and GP2 that is expressed on the surface of the target cell; wherein (i) the second binding sites localize the first binding sites to the IL-10 family address target such that the IL-22 polypeptides or fragments or variants thereof influence signaling by the IL-22 receptor in the target cell; (ii) the second binding sites do not substantially influence signaling upon binding the IL-10 family address targets; and (iii) the IL-22 polypeptides or fragments or variants thereof do not substantially influence signaling by the IL-22 receptor in the absence of localization by the second binding sites.
- the two subunits are
- the disclosure features a macromolecule comprising two subunits, each comprising a first binding site and a second binding site, wherein (a) each first binding site comprises IL- 22 or a fragment or variant thereof, and (b) each second binding site comprises an anti-CDH17 antibody or antigen-binding fragment thereof, wherein the macromolecule is a fusion protein, and wherein the first binding site and the second binding site of each subunit are connected by a linker.
- the IL-22 or fragment or variant thereof is a detuned or attenuated IL-22 or fragment or variant thereof, e.g., as described in Section B(i)(b), below.
- the anti-CDH17 antibodies or antigen-binding fragments thereof localize the first binding sites to an IL-22 receptor such that the IL-22 or fragments or variants thereof influence signaling by the IL-22 receptor in the target cell; (ii) the anti-CDH17 antibodies or antigenbinding fragments thereof do not substantially influence signaling upon binding CDH17; and/or (iii) the IL- 22 or fragments or variants thereof do not substantially influence signaling by the IL-22 receptor in the absence of localization by the anti-CDH17 antibodies or antigen-binding fragments thereof.
- the disclosure also features a method of modulating IL-22 receptor signaling in the small intestine or colon of a subject, the method comprising administering the above-described macromolecule to the subject.
- the disclosure features a macromolecule comprising two subunits, each comprising a first binding site and a second binding site, wherein (a) each first binding site comprises IL- 22 or a fragment or variant thereof, and (b) each second binding site comprises an anti-CDH16 antibody or antigen-binding fragment thereof, wherein the macromolecule is a fusion protein, and wherein the first binding site and the second binding site of each subunit are connected by a linker.
- the IL-22 or fragment or variant thereof is a detuned or attenuated IL-22 or fragment or variant thereof, e.g., as described in Section B(i)(b), below.
- the anti-CDH16 antibodies or antigen-binding fragments thereof localize the first binding sites to an IL-22 receptor such that the IL-22 or fragments or variants thereof influence signaling by the IL-22 receptor in the target cell; (ii) the anti-CDH16 antibodies or antigenbinding fragments thereof do not substantially influence signaling upon binding CDH16; and/or (iii) the IL- 22 or fragments or variants thereof do not substantially influence signaling by the IL-22 receptor in the absence of localization by the anti-CDH16 antibodies or antigen-binding fragments thereof.
- the disclosure also features a method of modulating IL-22 receptor signaling in the kidney of a subject, the method comprising administering the above-described macromolecule to the subject.
- the disclosure features a macromolecule comprising two subunits, each comprising a first binding site and a second binding site, wherein (a) each first binding site comprises IL- 22 or a fragment or variant thereof, and (b) each second binding site comprises an anti-GP2 antibody or antigen-binding fragment thereof, wherein the macromolecule is a fusion protein, and wherein the first binding site and the second binding site of each subunit are connected by a linker.
- the IL-22 or fragment or variant thereof is a detuned or attenuated IL-22 or fragment or variant thereof, e.g., as described in Section B(i)(b), below.
- the anti-GP2 antibodies or antigen-binding fragments thereof localize the first binding sites to an IL-22 receptor such that the IL-22 or fragments or variants thereof influence signaling by the IL-22 receptor in the target cell; (ii) the anti-GP2 antibodies or antigen-binding fragments thereof do not substantially influence signaling upon binding GP2; and/or (iii) the IL-22 or fragments or variants thereof do not substantially influence signaling by the IL-22 receptor in the absence of localization by the second binding sites.
- the disclosure also features a method of modulating IL-22 receptor signaling in the pancreas of a subject, the method comprising administering the above-described macromolecule to the subject.
- the macromolecule comprises a single first binding site and/or comprises a single second binding site (e.g., comprises a single first binding site and a single second binding site).
- the macromolecule is a heterodimer comprising two subunits, wherein the first subunit comprises the first binding site and second subunit comprises the second binding site.
- the macromolecule is a fusion protein.
- the fusion protein comprises a linker (e.g., a GS linker) conjugated to the first binding site and the second binding site.
- the disclosure features a macromolecule comprising (a) a first binding site comprising IL-22 or a fragment or variant thereof, and (b) a second binding site comprising an anti- CDH17 antibody or antigen-binding fragment thereof, wherein the macromolecule is a fusion protein, and wherein the first binding site and the second binding site are connected by a linker.
- the IL-22 or fragment or variant thereof is a detuned or attenuated IL-22 or fragment or variant thereof, e.g., as described in Section B(i)(b), below.
- the anti-CDH17 antibody or antigen-binding fragment thereof localizes the first binding site to an IL-22 receptor such that the IL-22 or fragment or variant thereof influences signaling by the IL-22 receptor in the target cell; (ii) the anti-CDH17 antibody or antigen-binding fragment thereof does not substantially influence signaling upon binding CDH17; and/or (iii) the IL-22 or fragment or variant thereof does not substantially influence signaling by the IL-22 receptor in the absence of localization by the anti-CDH17 antibody or antigen-binding fragment thereof.
- the disclosure also features a method of modulating IL-22 receptor signaling in the small intestine or colon of a subject, the method comprising administering the above-described macromolecule to the subject.
- the disclosure features a macromolecule comprising (a) a first binding site comprising IL-22 or a fragment or variant thereof, and (b) a second binding site comprising an anti- CDH16 antibody or antigen-binding fragment thereof, wherein the macromolecule is a fusion protein, and wherein the first binding site and the second binding site are connected by a linker.
- the IL-22 or fragment or variant thereof is a detuned or attenuated IL-22 or fragment or variant thereof, e.g., as described in Section B(i)(b), below.
- the anti-CDH16 antibody or antigen-binding fragment thereof localizes the first binding site to an IL-22 receptor such that the IL-22 or fragment or variant thereof influences signaling by the IL-22 receptor in the target cell; (ii) the anti-CDH16 antibody or antigen-binding fragment thereof does not substantially influence signaling upon binding CDH16; and/or (iii) the IL-22 or fragment or variant thereof does not substantially influence signaling by the IL-22 receptor in the absence of localization by the anti-CDH16 antibody or antigen-binding fragment thereof.
- the disclosure also features a method of modulating IL-22 receptor signaling in the kidney of a subject, the method comprising administering the above-described macromolecule to the subject.
- a method of modulating IL-22 receptor signaling in the kidney of a subject comprising administering the above-described macromolecule to the subject.
- the disclosure features a macromolecule comprising (a) a first binding site comprising IL-22 or a fragment or variant thereof, and (b) a second binding site comprising an anti-GP2 antibody or antigen-binding fragment thereof, wherein the macromolecule is a fusion protein, and wherein the first binding site and the second binding site are connected by a linker.
- the IL-22 or fragment or variant thereof is a detuned or attenuated IL-22 or fragment or variant thereof, e.g., as described in Section B(i)(b), below.
- the anti-GP2 antibody or antigen-binding fragment thereof localizes the first binding site to an IL-22 receptor such that the IL-22 or fragment or variant thereof influences signaling by the IL-22 receptor in the target cell; (ii) the anti-GP2 antibody or antigen-binding fragment thereof does not substantially influence signaling upon binding GP2; and/or (iii) the IL-22 or fragment or variant thereof does not substantially influence signaling by the IL-22 receptor in the absence of localization by the anti-GP2 antibody or antigen-binding fragment thereof.
- the disclosure also features a method of modulating IL-22 receptor signaling in the pancreas of a subject, the method comprising administering the above-described macromolecule to the subject. iii. ANDbody Function
- the macromolecules (i.e ., ANDbodies) described herein comprise an effector binding domain (an IL-22 polypeptide or fragment or variant thereof) specific for an effector target (an IL-22 receptor).
- an effector binding domain an IL-22 polypeptide or fragment or variant thereof
- the effector Upon binding of the effector binding domain to the effector target, the effector modulates the target cell or tissue to produce a biological response, such as a therapeutic effect, on the subject.
- the effector target binding domain provided herein may not elicit a biological effect unless it is provided in conjunction with an address targeting domain (e.g., an IL-10 family address target) to localize the effector to the desired target address in a targeted cell or tissue.
- an address targeting domain e.g., an IL-10 family address target
- such therapeutic signaling may require the binding of multiple effector targets by multiple macromolecules according to the invention.
- an IL-22 polypeptide or fragment or variant thereof may produce a small/weak biological effect when provided alone and provide a larger/stronger biological effect when provided in conjunction with an address targeting domain that localizes and concentrates/focuses the effector to the desired target address in a targeted cell or tissue.
- an effector target binding domain may produce an acceptable biological effect when provided alone and provide an even larger/stronger biological effect when provided in conjunction with an address targeting domain to localize the effector target binding domain to a targeted cell or tissue.
- an effector target binding domain may produce a strong biological effect when provided alone and provide a strong, or stronger, targeted effect when provided in conjunction with an address targeting domain to localize the effector target binding domain to a targeted cell or tissue.
- an effector target binding domain may produce a biological effect with undesirable off target biological effects when provided alone, but can be targeted, concentrated, and focused to desired addresses in a targeted cell or tissue when provided in conjunction with an address targeting domain in order to decrease or eliminate undesirable off-target biological effects. Accordingly, effector target binding domains of the present technology provide superior therapeutic agents that provide stronger, targeted biological effects with less side effects, including less unintended off-target biological effects, when provided in conjunction with address target binding domains as described herein.
- binding of the address targeting domain to the IL-10 family address target increases accumulation of the macromolecule in tissues expressing the IL-10 family address target, (ii) reduces accumulation of the macromolecule in tissues that do not express the IL-10 family address target, or (iii) (i) and (ii).
- At least 25% of the macromolecule (e.g., ANDbody) detectable in the subject is detected at the target tissue or cell at a time point between 1 and 7 days (e.g., at 1 day, 2 days, 3 days, 4 days, 5, days, 6 days, and/or 7 days) following administration of the macromolecule (e.g., ANDbody) to the subject.
- 1 and 7 days e.g., at 1 day, 2 days, 3 days, 4 days, 5, days, 6 days, and/or 7 days
- therapeutic signaling effects that may be induced by a macromolecule provided herein include, but are not limited to:
- ADCC antibody-dependent cellular cytotoxicity
- Signaling effects that may be induced by a macromolecule provided herein further include modulation of cytokine signaling (e.g., modulation that affects immune response, response to infection, inflammation, allergy, autoimmunity, cancer, and/or tissue homeostasis).
- cytokine signaling pathways are described, e.g., in Ouyang and O’Garra, Immunity, 50: 871 -891 , 2019).
- the therapeutic effector target (i.e., IL-22 receptor) is more broadly expressed than the address target in the subject.
- the therapeutic effector target is expressed systemically, regionally, or locally in the organism.
- Systemic expression of a therapeutic effector target means that the therapeutic effector target is expressed at substantially the same levels in most parts of a subject organism body. Systemic expression involves a plurality of tissues.
- Regular expression of a therapeutic effector target means that the therapeutic target is expressed in an area less than systemic expression but more than local expression.
- Regional expression is not limited to a single tissue but can occur in a plurality of different tissues.
- “Local expression” of a therapeutic effector target means that the therapeutic target is expressed in single or few tissue areas. Local expression is not limited to a single tissue but can occur in a plurality of different tissues.
- the effector target binding domain has a low affinity for the effector target.
- a low affinity may be an affinity of greater than 10 nM (e.g., an affinity between 10 nM-1 pM, e.g., an affinity between 10 nM and 100 nM).
- the effector target binding domain has a low avidity for the effector target.
- An ANDbodyTM of the invention comprises an effector (i.e., an IL-22 polypeptide or fragment or variant thereof, e.g., an IL-22 mutein polypeptide) that modulates a therapeutic effector target (i.e., an IL- 22 receptor to which the IL-22 polypeptide or fragment or variant thereof binds) in a cell or in a subject, e.g., a mammalian subject such as a human, in need thereof.
- an effector i.e., an IL-22 polypeptide or fragment or variant thereof, e.g., an IL-22 mutein polypeptide
- a therapeutic effector target i.e., an IL- 22 receptor to which the IL-22 polypeptide or fragment or variant thereof binds
- the disclosure also provides IL-22 mutein polypeptides or fragments or variants thereof that have altered (e.g., decreased) affinity for the IL-22 receptor.
- the IL-22 mutein polypeptide or fragment or variant thereof has about a 1 .3-fold to about a 10-fold decreased binding affinity to human IL-22Ra as compared to the binding affinity of wild type human IL-22 polypeptide to human IL-22Ra, e.g., as determined by a method described herein.
- the IL-22 mutein polypeptide or fragment or variant thereof has at least about a 1 .3-fold decreased binding affinity to human IL-22Ra as compared to the binding affinity of wild type human IL-22 polypeptide to human IL- 22Ra. In some embodiments, the IL-22 mutein polypeptide or fragment or variant thereof has at least about a 2-fold, 4-fold, 6-fold, 8-fold, or 10-fold decreased binding affinity to human IL-22Ra as compared to the binding affinity of wild type human IL-22 polypeptide to human IL-22Ra, or has a more than 10-fold decreased binding affinity to human IL-22Ra as compared to the binding affinity of wild type human IL-22 polypeptide to human IL-22Ra.
- the effector is human Interleukin 22 (IL-22), e.g., as exemplified by UniProt Accession Number Q9GZX6; gene symbol IL22; and/or RefSeq NP_065386.1 or a fragment or variant thereof (e.g., a fully functional or partially functional fragment or variant thereof).
- IL-22 human Interleukin 22
- a fragment or variant thereof e.g., a fully functional or partially functional fragment or variant thereof.
- the IL-22 polypeptide or fragment or variant thereof comprises the mature form of the human IL-22 peptide.
- the IL-22 polypeptide or fragment or variant does not include a leader sequence, e.g., does not comprise the leader sequence of a pro-form of the IL-22 polypeptide.
- the IL-22 polypeptide or fragment or variant does not contain the leader sequence present in SEQ ID NO: 7, e.g., does not contain residues 1 to 33 of SEQ ID NO: 7.
- an IL-22 polypeptide or fragment or variant thereof comprises one or more amino acid substitution mutations (e.g., is an IL-22 mutein polypeptide comprising an amino acid substitution mutation at one or more of positions K61 , D71 , R73, D168, and R175, wherein the amino acid positions are numbered relative to SEQ ID NO: 7); and such IL-22 polypeptide or fragment or variant does not include the leader sequence of the pro-form of human IL-22 of SEQ ID NO: 7 (e.g., does not contain residues 1 to 33 of SEQ ID NO: 7).
- amino acid substitution mutations e.g., is an IL-22 mutein polypeptide comprising an amino acid substitution mutation at one or more of positions K61 , D71 , R73, D168, and R175, wherein the amino acid positions are numbered relative to SEQ ID NO: 7
- IL-22 polypeptide or fragment or variant does not include the leader sequence of the pro-form of human IL-22 of SEQ ID NO: 7 (e.
- numbering of amino acid residues in the human IL-22 polypeptide are with reference to SEQ ID NO: 7 (the pro-form of human IL-22, comprising a leader sequence consisting of residues 1 -33).
- SEQ ID NO: 7 the pro-form of human IL-22, comprising a leader sequence consisting of residues 1 -33.
- the numbering of these amino acid residues refers to the sequence of SEQ ID NO: 7.
- the IL-22 polypeptide or fragment or variant can comprise or consist of the mature form of the polypeptide that does not include a leader sequence, e.g., may not comprise the leader sequence of a pro-form of the IL-22 polypeptide (such as the leader sequence present in SEQ ID NO: 7, e.g., does not contain a sequence corresponding to residues 1 to 33 of SEQ ID NO: 7).
- the numbering of residues nevertheless refers to SEQ ID NO: 7 (e.g., such that the first residue of the IL-22 polypeptide, fragment, or variant is position 34).
- the IL-22 R73S polypeptide provided in SED IQ NO: 16 comprises an Arg to Ser amino acid substitution mutation at position 73, with reference to SEQ ID NO: 7 for position numbering.
- the IL-22 R73S polypeptide of SED IQ NO: 16 does not contain a leader sequence, e.g., the leader sequence of SEQ ID NO: 7, such that the R73S amino acid substitution mutation is located 40 residues from the N-terminus of the IL-22 polypeptide.
- the IL-22 polypeptide or fragment or variant thereof exhibits reduced IL- 22 receptor (e.g., IL-22a) binding relative to the binding of wild type IL-22 polypeptide, e.g., as described in an assay provided herein.
- the human IL-22 polypeptide or fragment or variant thereof has about a 1 .3-fold to about a10-fold decreased binding affinity to human IL-22Ra as compared to the binding affinity of wild type human IL-22 polypeptide to human IL-22Ra, e.g., as determined by a method described herein.
- the IL-22 polypeptide or fragment or variant thereof when part of the macromolecule, exhibits reduced IL-22 receptor binding relative to the IL-22 polypeptide or fragment or variant thereof when not part of the macromolecule, on a mole-adjusted basis, e.g., in an assay described herein.
- the IL-22 polypeptide or fragment or variant thereof exhibits reduced IL- 22 receptor binding relative to the IL-22 polypeptide or fragment or variant thereof on a mole-adjusted basis, e.g., in an assay described herein.
- the IL-22 polypeptide or fragment or variant thereof when part of the macromolecule, exhibits reduced agonist activity relative to the IL-22 polypeptide or fragment or variant thereof when not part of the macromolecule, on a mole-adjusted basis.
- the IL-22 polypeptide or fragment or variant thereof exhibits reduced agonist activity relative to the IL-22 polypeptide or fragment or variant thereof on a mole-adjusted basis.
- the disclosure provides an IL-22 polypeptide or fragment thereof that has lower binding affinity for the IL-22 receptor than a comparator IL-22 polypeptide or fragment or variant thereof (e.g., a wild-type IL-22 (e.g., of SEQ ID NO: 8) and/or an IL-22 that is a native ligand of the IL-22 receptor), e.g., is detuned or attenuated, e.g., as measured using an assay described herein.
- a comparator IL-22 polypeptide or fragment or variant thereof e.g., a wild-type IL-22 (e.g., of SEQ ID NO: 8) and/or an IL-22 that is a native ligand of the IL-22 receptor
- the IL-22 polypeptide or fragment or variant thereof may be engineered to have reduced affinity for the receptor or may be a fragment of a wild-type IL-22 that has reduced affinity for the receptor (e.g., to IL-22Ra), e.g., as compared to the binding affinity of wild type human IL-22 (e.g., of SEQ ID NO: 8) for the IL-22 receptor, e.g., as measured an assay described herein (i.e., is an IL-22 mutein polypeptide).
- the IL-22 mutein polypeptide or fragment thereof comprises one or more amino acid substitution mutations at one or more residues that are (or are predicted to be) at or near the interaction surface between IL-22 (and/or the IL-22 mutein polypeptide or fragment thereof) and the IL-22 receptor.
- the IL-22 mutein polypeptide or fragment thereof may have a binding affinity for the IL-22 receptor that is at least 5%, 10%, 15%, 20%, 25%, 30%, 35%, 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, or 95% (e.g., 5-10%, 10-20%, 20-30%, 30-40%, 40-50%, 50-60%, 60- 70%, 70-80%, 80-90%, or more than 90%) lower than a comparator IL-22 polypeptide or fragment or variant thereof, e.g., as compared to the binding affinity of wild type human IL-22 (e.g., of SEQ ID NO: 8) for the IL-22 receptor (e.g., to IL-22Ra), e.g., in an assay described herein.
- a macromolecule of the invention e.g., an ANDbody
- the IL-22 polypeptide or fragment or variant thereof of the macromolecule exhibits reduced agonist activity (e.g., reduced activation of signaling by the IL-22 receptor), e.g., relative to a comparator IL-22 polypeptide or fragment or variant thereof (e.g., a wild-type IL-22 polypeptide, e.g., on a mole-adjusted basis).
- the reduced agonist activity may be global, or may be measured at one or more target cells.
- the IL-22 mutein polypeptide or fragment thereof may exhibit agonist activity that is reduced by at least 5%, 10%, 15%, 20%, 25%, 30%, 35%, 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, or 95% (e.g., 5-10%, 10-20%, 20-30%, 30-40%, 40-50%, 50-60%, 60-70%, 70-80%, 80-90%, or more than 90%) lower than a comparator IL-22 polypeptide or fragment or variant thereof (e.g., at one or more target cells), or may exhibit no observable agonist activity.
- a comparator IL-22 polypeptide or fragment or variant thereof e.g., at one or more target cells
- the reduced agonist activity is observed when the IL-22 polypeptide or fragment or variant thereof is not in the context of a macromolecule comprising a binding domain or other moiety that compensates for the reduced affinity of the IL-22 mutein polypeptide (e.g., an ANDbody), e.g., by binding to a target on a target cell.
- a macromolecule comprising a binding domain or other moiety that compensates for the reduced affinity of the IL-22 mutein polypeptide (e.g., an ANDbody), e.g., by binding to a target on a target cell.
- the IL-22 mutein polypeptide exhibits reduced agonist activity or exhibits no observable agonist activity in the absence of a binding site targeting an IL-10 family address target, and the reduction or abrogation of agonist activity partially or entirely resolved (e.g., partially or entirely resolved at one or more target cells of the macromolecule) when the IL-22 mutein polypeptide or fragment thereof is in the context of a macromolecule targeting an IL-10 family address target (e.g., is fused to a domain that targets an IL-10 family address target).
- the IL-22 polypeptide or fragment or variant thereof comprised by a macromolecule (e.g., ANDbody) of the invention e.g., IL-22 polypeptide or fragment or variant thereof having reduced binding affinity for the IL-22 receptor, e.g., IL-22 mutein polypeptide
- a macromolecule e.g., ANDbody
- the invention does not substantially bind to the IL-22 receptor and/or does not exhibit substantial agonist activity at a target cell or tissue in the absence of a second binding site that specifically binds a target (e.g., an IL-10 family address target), thereby targeting the macromolecule to the target cell or tissue (e.g., signaling activity by the IL-22 polypeptide or fragment or variant thereof (e.g., IL-22 mutein polypeptide) at the target cell or tissue is conditional upon the presence of the second binding site in the macromolecule).
- a target e.g., an IL-10 family address target
- the affinity of the IL-22 polypeptide or fragment or variant thereof for the IL-22 receptor (and/or the affinity of the second binding site for the IL-10 family address target) is engineered such that substantial binding to the IL-22 receptor and substantial induction of signaling is conditional upon the presence of the second binding site in the macromolecule.
- the IL-22 or fragment or variant thereof has reduced binding affinity for IL-22Ra and/or IL-10R2.
- the IL-22 or fragment or variant thereof of the macromolecule may have a binding affinity for IL-22Ra and/or IL-10R2 that is at least 5%, 10%, 15%, 20%, 25%, 30%, 35%, 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, or 95% (e.g., 5-10%, 10-20%, 20-30%, 30-40%, 40-50%, 50-60%, 60-70%, 70-80%, 80-90%, or more than 90%) lower than a comparator IL-22, e.g., as compared to the binding affinity of wild type human IL-22 (e.g., of SEQ ID NO: 8) for the IL-22 receptor, e.g., as measured in an assay described herein.
- a comparator IL-22 e.g., as compared to the binding affinity of wild type human
- the IL-22 polypeptide or fragment or variant thereof has about a 1 .3-fold to about a10-fold decreased binding affinity to human IL-22Ra as compared to the binding affinity of wild type human IL-22 polypeptide to human IL-22Ra, e.g., as determined by a method described herein.
- the disclosure provides IL-22 polypeptides and fragments or variants thereof (e.g., IL-22 mutein polypeptides) that are capable of binding an IL-22 receptor on the surface of a target cell.
- the IL-22 polypeptide or fragment or variant thereof binds the IL- 22 receptor only when paired with another moiety (e.g., a targeting domain), e.g., conditionally binds the IL-22 receptor.
- the IL-22 polypeptide or fragment or variant thereof binds the IL-22 receptor at a threshold level (e.g., a level sufficient to induce downstream signaling and/or a therapeutically relevant level) only when paired with another moiety (e.g., a targeting domain).
- a threshold level e.g., a level sufficient to induce downstream signaling and/or a therapeutically relevant level
- another moiety e.g., a targeting domain
- the IL-22 polypeptide or fragment or variant thereof constitutively binds the IL- 22 receptor.
- the IL-22 polypeptide or fragment or variant thereof binds the IL-22 receptor (e.g., conditionally or constitutively binds the IL-22 receptor). In some embodiments, the IL-22 polypeptide or fragment or variant thereof binds the human IL-22 receptor (e.g., conditionally or constitutively binds the human IL-22 receptor). In some embodiments, the IL-22 polypeptide or fragment or variant thereof binds human IL-22Ra and/or IL-10R2 (e.g., conditionally or constitutively binds human IL-22Ra and/or IL-10R2).
- the IL-22 polypeptide or a fragment or variant thereof binds IL-22BP (e.g., conditionally or constitutively binds IL-22BP). In some embodiments, the IL-22 polypeptide or fragment or variant thereof binds human IL-22BP (e.g., conditionally or constitutively binds human IL-22BP).
- the IL-22 polypeptide or fragment or variant thereof agonizes the IL-22 receptor (e.g., conditionally or constitutively agonizes the IL-22 receptor). In some embodiments, the IL- 22 polypeptide or fragment or variant thereof agonizes the human IL-22 receptor (e.g., conditionally or constitutively agonizes the human IL-22 receptor). In some embodiments, the IL-22 polypeptide or fragment or variant thereof agonizes human IL-22Ra and/or IL-10R2 (e.g., conditionally or constitutively agonizes human IL-22Ra and/or IL-10R2).
- the macromolecule agonizes IL-22BP (e.g., conditionally or constitutively agonizes IL-22BP). In some embodiments, the macromolecule agonizes human IL-22BP (e.g., conditionally or constitutively agonizes human IL-22BP).
- the IL-22 polypeptide or fragment or variant thereof has low (e.g., lower) binding affinity for IL-22Ra and/or IL-10R2, as compared to the binding affinity of wild type human IL-22 (e.g., of SEQ ID NO: 8) for the IL-22 receptor, e.g., as measured in an assay described herein.
- the IL-22 polypeptide or fragment or variant thereof has low (e.g., lower) binding affinity for IL-22BP, as compared to the binding affinity of wild type human IL-22 (e.g., of SEQ ID NO: 8) for IL-2BP, e.g., as measured in an assay described herein, e.g., a BLI assay described herein.
- the IL-22 or fragment thereof (e.g., IL-22 mutein polypeptide) comprises one or more amino acid substitution mutations at one or more residues that are (or are predicted to be) at or near the interaction surface between IL-22 and IL-22Ra and/or IL-10R2.
- Residues at the interaction surface between IL-22 and its receptors are described, e.g., in Saxton et al., Immunity, 54(4): 660-672e.9, 2021 and in Wu et al., Journal of Molecular Biology, 382(5): 1168- 1183, 2008.
- the IL-22 or fragment or variant thereof has reduced binding affinity for IL22RA1 relative to a wild-type IL-22 or fragment thereof, e.g., as compared to the binding affinity of wild type human IL-22 (e.g., of SEQ ID NO: 8) for the IL-22 receptor, e.g., as measured in an assay described herein.
- the IL-22 polypeptide or fragment or variant thereof has about a 1 .3-fold to about a10-fold decreased binding affinity to human IL- 22Ra as compared to the binding affinity of wild type human IL-22 polypeptide to human IL-22Ra, e.g., as determined by a method described herein.
- the IL-22 polypeptide or fragment or variant thereof does not comprise a leader sequence, e.g., does not comprise a leader sequence of a proform of a IL-22 polypeptide.
- the IL-22 polypeptide or fragment or variant thereof does not comprise the leader sequence comprised in SEQ ID NO: 7, e.g., does not comprise residues 1 to 33 of SEQ ID NO: 7.
- the IL-22 polypeptide or fragment or variant thereof does not comprise the sequence of SEQ ID NO: 14.
- the IL-22 polypeptide or fragment or variant thereof is a human IL-22 or fragment or variant thereof.
- the IL-22 polypeptide or fragment or variant thereof is a modified human IL-22 or fragment or variant thereof and comprises one or more of the amino acid substitution mutations of Table 6, Table 7A, Table 7B, Table 8, Table 9A, Table 9B, or Table 9C (e.g., comprises one or more of said amino acid mutations and has at least 80%, 85%, 90%, 95%, 98%, or 99% identity to a human IL- 22, e.g., as set forth in SEQ ID NO: 7 or SEQ ID NO: 8).
- the IL-22 polypeptide or fragment or variant thereof comprises an amino acid substitution mutation at one or more of positions K61 , D71 , R73, D168, and R175, wherein the amino acid position numbering is with respect to the human IL-22 sequence provided in SEQ ID NO: 7.
- the IL-22 polypeptide or fragment or variant thereof comprises a K61 amino acid substitution mutation.
- the IL-22 polypeptide or fragment or variant thereof comprises a D71 amino acid substitution mutation.
- the IL-22 polypeptide or fragment or variant thereof comprises a R73 amino acid substitution mutation.
- the IL-22 polypeptide or fragment or variant thereof comprises a D168 amino acid substitution mutation. In some embodiments, the IL-22 polypeptide or fragment or variant thereof comprises a R175 amino acid substitution mutation. In some embodiments, the IL-22 polypeptide or fragment or variant thereof comprises a D71 amino acid substitution mutation and a D168 amino acid substitution mutation. In some embodiments, the IL-22 mutein polypeptide comprising the amino acid substitution mutation at position K61 , D71 , R73, D168, and/or R175 has at least 80%, 85%, 90%, 95%, 98%, or 99% identity to a human IL-22, e.g., as set forth in SEQ ID NO: 7 or SEQ ID NO: 8).
- the IL-22 polypeptide or fragment or variant thereof comprises one or more of a K61 S, D71 N, D71 L, D71 G, R73Q, R73S, R73N, D168N, D168G, and R175S amino acid substitution mutation, wherein the amino acid position numbering is with respect to the human IL-22 sequence provided in SEQ ID NO: 7.
- the IL-22 polypeptide or fragment or variant thereof comprises a K61 S amino acid substitution mutation.
- the IL-22 polypeptide or fragment or variant thereof comprises a D71 N amino acid substitution mutation.
- the IL-22 polypeptide or fragment or variant thereof comprises a D71 L amino acid substitution mutation.
- the IL-22 polypeptide or fragment or variant thereof comprises a D71 G amino acid substitution mutation. In some embodiments, the IL-22 polypeptide or fragment or variant thereof comprises a R73Q amino acid substitution mutation. In some embodiments, the IL-22 polypeptide or fragment or variant thereof comprises a R73S amino acid substitution mutation. In some embodiments, the IL-22 polypeptide or fragment or variant thereof comprises a R73N amino acid substitution mutation. In some embodiments, the IL-22 polypeptide or fragment or variant thereof comprises a D168N amino acid substitution mutation. In some embodiments, the IL-22 polypeptide or fragment or variant thereof comprises a D168G amino acid substitution mutation.
- the IL-22 polypeptide or fragment or variant thereof comprises a R175S amino acid substitution mutation. In some embodiments, the IL-22 polypeptide or fragment or variant thereof comprises a D71 L amino acid substitution mutation and a D168G amino acid substitution mutation. In some embodiments, the IL-22 mutein polypeptide comprising the one or more amino acid substitution mutations has at least 80%, 85%, 90%, 95%, 98%, or 99% identity to a human IL-22, e.g., as set forth in SEQ ID NO: 7.
- the IL-22 mutein polypeptide comprising the one or more amino acid substitution mutations has at least 80%, 85%, 90%, 95%, 98%, or 99% identity to a human IL-22, e.g., as set forth in SEQ ID NO: 8.
- the IL-22 mutein polypeptide has about a 1 .3-fold to about a 10- fold decreased binding affinity to human IL-22Ra as compared to the binding affinity of wild type human IL-22 polypeptide to human IL-22Ra, e.g., as determined by a method described herein.
- the IL-22 polypeptide or fragment or variant thereof comprises a K61 S amino acid substitution mutation.
- the IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 17 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 17).
- the IL-22 mutein polypeptide binds to IL-22Ra.
- the IL-22 mutein polypeptide has about a 1 .3-fold to about a10-fold decreased binding affinity to human IL-22Ra as compared to the binding affinity of wild type human IL-22 polypeptide to human IL-22Ra, e.g., as determined by a method described herein.
- the IL-22 polypeptide or fragment or variant thereof comprises a K61 S amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 17.
- the IL-22 mutein polypeptide binds to IL- 22Ra.
- the IL-22 mutein polypeptide has about a 1 .3-fold to about a10-fold decreased binding affinity to human IL-22Ra as compared to the binding affinity of wild type human IL-22 polypeptide to human IL-22Ra, e.g., as determined by a method described herein.
- the IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 17. In some embodiments, the IL-22 polypeptide or fragment or variant thereof consists of SEQ ID NO: 17.
- the IL-22 polypeptide or fragment or variant thereof comprises a D71 G amino acid substitution mutation.
- the IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 15 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 15).
- the IL-22 mutein polypeptide binds to IL-22Ra.
- the IL-22 mutein polypeptide has about a 1 .3-fold to about a10-fold decreased binding affinity to human IL-22Ra as compared to the binding affinity of wild type human IL-22 polypeptide to human IL-22Ra, e.g., as determined by a method described herein.
- the IL-22 polypeptide or fragment or variant thereof comprises a D71 G amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 15.
- the IL-22 mutein polypeptide binds to IL- 22Ra.
- the IL-22 mutein polypeptide has about a 1 .3-fold to about a10-fold decreased binding affinity to human IL-22Ra as compared to the binding affinity of wild type human IL-22 polypeptide to human IL-22Ra, e.g., as determined by a method described herein.
- the IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 15. In some embodiments, the IL-22 polypeptide or fragment or variant thereof consists of SEQ ID NO: 15.
- the IL-22 polypeptide or fragment or variant thereof comprises a D71 L amino acid substitution mutation.
- the IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 11 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 11 ).
- the IL-22 mutein polypeptide binds to IL-22Ra.
- the IL-22 mutein polypeptide has about a 1 .3-fold to about a10-fold decreased binding affinity to human IL-22Ra as compared to the binding affinity of wild type human IL-22 polypeptide to human IL-22Ra, e.g., as determined by a method described herein.
- the IL-22 polypeptide or fragment or variant thereof comprises a D71 L amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 11 .
- the IL-22 mutein polypeptide binds to IL- 22Ra.
- the IL-22 mutein polypeptide has about a 1 .3-fold to about a10-fold decreased binding affinity to human IL-22Ra as compared to the binding affinity of wild type human IL-22 polypeptide to human IL-22Ra, e.g., as determined by a method described herein.
- the IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 11. In some embodiments, the IL-22 polypeptide or fragment or variant thereof consists of SEQ ID NO: 11.
- the IL-22 polypeptide or fragment or variant thereof comprises a D71 N amino acid substitution mutation.
- the IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 18 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 18).
- the IL-22 mutein polypeptide binds to IL-22Ra.
- the IL-22 mutein polypeptide has about a 1 .3-fold to about a10-fold decreased binding affinity to human IL-22Ra as compared to the binding affinity of wild type human IL-22 polypeptide to human IL-22Ra, e.g., as determined by a method described herein.
- the IL-22 polypeptide or fragment or variant thereof comprises a D71 N amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 18.
- the IL-22 mutein polypeptide binds to IL- 22Ra.
- the IL-22 mutein polypeptide has about a 1 .3-fold to about a10-fold decreased binding affinity to human IL-22Ra as compared to the binding affinity of wild type human IL-22 polypeptide to human IL-22Ra, e.g., as determined by a method described herein.
- the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 18. In some embodiments, the IL-22 polypeptide or fragment or variant thereof consists of SEQ ID NO: 18.
- the IL-22 polypeptide or fragment or variant thereof comprises a R73S amino acid substitution mutation.
- the IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 16 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 16).
- the IL-22 mutein polypeptide binds to IL-22Ra.
- the IL-22 mutein polypeptide has about a 1 .3-fold to about a10-fold decreased binding affinity to human IL-22Ra as compared to the binding affinity of wild type human IL-22 polypeptide to human IL-22Ra, e.g., as determined by a method described herein.
- the IL-22 polypeptide or fragment or variant thereof comprises a R73S amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 16.
- the IL-22 mutein polypeptide binds to IL- 22Ra.
- the IL-22 mutein polypeptide has about a 1 .3-fold to about a10-fold decreased binding affinity to human IL-22Ra as compared to the binding affinity of wild type human IL-22 polypeptide to human IL-22Ra, e.g., as determined by a method described herein.
- the IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 16. In some embodiments, the IL-22 polypeptide or fragment or variant thereof consists of SEQ ID NO: 16.
- the IL-22 polypeptide or fragment or variant thereof comprises a R73N amino acid substitution mutation.
- the IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 22 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 22).
- the IL-22 mutein polypeptide binds to IL-22Ra.
- the IL-22 mutein polypeptide has about a 1 .3-fold to about a10-fold decreased binding affinity to human IL-22Ra as compared to the binding affinity of wild type human IL-22 polypeptide to human IL-22Ra, e.g., as determined by a method described herein.
- the IL-22 polypeptide or fragment or variant thereof comprises a R73N amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the IL-22 polypeptide or fragment or variant thereof to SEQ ID NO:22, In some embodiments, the IL-22 mutein polypeptide binds to IL- 22Ra.
- the IL-22 mutein polypeptide has about a 1 .3-fold to about a10-fold decreased binding affinity to human IL-22Ra as compared to the binding affinity of wild type human IL-22 polypeptide to human IL-22Ra, e.g., as determined by a method described herein.
- the IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 22. In some embodiments, the IL-22 polypeptide or fragment or variant thereof consists of SEQ ID NO: 22.
- the IL-22 polypeptide or fragment or variant thereof comprises a R73Q amino acid substitution mutation.
- the IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 12 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 12).
- the IL-22 mutein polypeptide binds to IL-22Ra.
- the IL-22 mutein polypeptide has about a 1 .3-fold to about a10-fold decreased binding affinity to human IL-22Ra as compared to the binding affinity of wild type human IL-22 polypeptide to human IL-22Ra, e.g., as determined by a method described herein.
- the IL-22 polypeptide or fragment or variant thereof comprises a R73Q amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 12.
- the IL-22 mutein polypeptide binds to IL- 22Ra.
- the IL-22 mutein polypeptide has about a 1 .3-fold to about a10-fold decreased binding affinity to human IL-22Ra as compared to the binding affinity of wild type human IL-22 polypeptide to human IL-22Ra, e.g., as determined by a method described herein.
- the IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 12. In some embodiments, the IL-22 polypeptide or fragment or variant thereof consists of SEQ ID NO: 12.
- the IL-22 polypeptide or fragment or variant thereof comprises a D168N amino acid substitution mutation.
- the IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 19 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 19).
- the IL-22 mutein polypeptide binds to IL-22Ra.
- the IL-22 mutein polypeptide has about a 1 .3-fold to about a10-fold decreased binding affinity to human IL-22Ra as compared to the binding affinity of wild type human IL-22 polypeptide to human IL-22Ra, e.g., as determined by a method described herein.
- the IL-22 polypeptide or fragment or variant thereof comprises a D168N amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 19.
- the IL-22 mutein polypeptide binds to IL- 22Ra.
- the IL-22 mutein polypeptide has about a 1 .3-fold to about a10-fold decreased binding affinity to human IL-22Ra as compared to the binding affinity of wild type human IL-22 polypeptide to human IL-22Ra, e.g., as determined by a method described herein.
- the IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 19.
- the IL-22 polypeptide or fragment or variant thereof consists of SEQ ID NO: 19.
- the IL-22 polypeptide or fragment or variant thereof comprises a D168G amino acid substitution mutation.
- the IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 20 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 20).
- the IL-22 mutein polypeptide binds to IL-22Ra.
- the IL-22 mutein polypeptide has about a 1 .3-fold to about a10-fold decreased binding affinity to human IL-22Ra as compared to the binding affinity of wild type human IL-22 polypeptide to human IL-22Ra, e.g., as determined by a method described herein.
- the IL-22 polypeptide or fragment or variant thereof comprises a D168G amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 20.
- the IL-22 mutein polypeptide binds to IL- 22Ra.
- the IL-22 mutein polypeptide has about a 1 .3-fold to about a10-fold decreased binding affinity to human IL-22Ra as compared to the binding affinity of wild type human IL-22 polypeptide to human IL-22Ra, e.g., as determined by a method described herein.
- the IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 20. In some embodiments, the IL-22 polypeptide or fragment or variant thereof consists of SEQ ID NO: 20.
- the IL-22 polypeptide or fragment or variant thereof comprises D71 L and D168G amino acid substitution mutations.
- the IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 13 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 13).
- the IL-22 mutein polypeptide binds to IL-22Ra.
- the IL-22 mutein polypeptide has about a 1 .3-fold to about a10-fold decreased binding affinity to human IL-22Ra as compared to the binding affinity of wild type human IL-22 polypeptide to human IL-22Ra, e.g., as determined by a method described herein.
- the IL-22 polypeptide or fragment or variant thereof comprises D71 L and D168 amino acid substitution mutations and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 13.
- the IL-22 mutein polypeptide binds to IL-22Ra.
- the IL-22 mutein polypeptide has about a 1 .3-fold to about a10-fold decreased binding affinity to human IL-22Ra as compared to the binding affinity of wild type human IL-22 polypeptide to human IL-22Ra, e.g., as determined by a method described herein.
- the IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 13. In some embodiments, the IL-22 polypeptide or fragment or variant thereof consists of SEQ ID NO: 13.
- the IL-22 polypeptide or fragment or variant thereof comprises a R175S amino acid substitution mutation.
- the IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 21 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 21 ).
- the IL-22 mutein polypeptide binds to IL-22Ra.
- the IL-22 mutein polypeptide has about a 1 .3-fold to about a10-fold decreased binding affinity to human IL-22Ra as compared to the binding affinity of wild type human IL-22 polypeptide to human IL-22Ra, e.g., as determined by a method described herein.
- the IL-22 polypeptide or fragment or variant thereof comprises a R175S amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 21 .
- the IL-22 mutein polypeptide binds to IL- 22Ra.
- the IL-22 mutein polypeptide has about a 1 .3-fold to about a10-fold decreased binding affinity to human IL-22Ra as compared to the binding affinity of wild type human IL-22 polypeptide to human IL-22Ra, e.g., as determined by a method described herein.
- the IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 21 . In some embodiments, the IL-22 polypeptide or fragment or variant thereof consists of SEQ ID NO: 21.
- an "effector target” is a discrete structure (e.g., a cell surface protein, a transmembrane protein, or a receptor) of a cell or tissue of a subject, to which a therapeutic effector binding domain of an ANDbody can bind and exert a modulating effect, such as a therapeutic effect on the subject.
- Effector targets as provided herein are molecules that are capable of binding to an IL-22 polypeptide or fragment or variant thereof as described above, wherein the binding affects (e.g., agonizes) an activity of the effector target (e.g., activates or enhances downstream signaling).
- IL-22 receptor subunits are known in the art, and include IL-1 ORB (IL-10R2) and IL-22Ra (IL22RA1 ) (see, e.g., Ouyang and O’Garra, Immunity, 50: 871 -891 , 2019). These receptor subunits generally form heterodimers and are agonized by IL-22.
- a macromolecule or polypeptide provided herein agonizes an IL-22 receptor (e.g., agonizes one or more IL-22 receptor subunits, e.g., agonizes IL-1 ORB and/or IL-22Ra, e.g., agonizes a heterodimeric receptor comprising IL-1 ORB and IL-22Ra).
- agonizes one or more IL-22 receptor subunits e.g., agonizes IL-1 ORB and/or IL-22Ra, e.g., agonizes a heterodimeric receptor comprising IL-1 ORB and IL-22Ra.
- a macromolecule or polypeptide provided herein agonizes one or more IL-22 receptor subunits, e.g., agonizes IL-1 ORB and/or IL-22Ra.
- a macromolecule or polypeptide provided herein binds one or more IL-22 receptor subunits, e.g., binds IL-1 ORB and/or IL-22Ra.
- the IL-22 receptor comprises IL-1 ORB (e.g., human IL-1 ORB, e.g., as exemplified by UniProt Accession Number Q08334).
- IL-1 ORB e.g., human IL-1 ORB, e.g., as exemplified by UniProt Accession Number Q08334.
- the IL-22 receptor comprises IL-22RA (IL-22Ra; IL-22RA1 ) (e.g., human IL-22RA, e.g., as exemplified by UniProt Accession Number Q8N6P7).
- IL-22RA IL-22Ra; IL-22RA1
- human IL-22RA e.g., as exemplified by UniProt Accession Number Q8N6P7.
- a macromolecule or polypeptide provided herein agonizes a decoy receptor, e.g., IL-22 BP. In some aspects, a macromolecule or polypeptide provided herein binds a decoy receptor, e.g., IL-22 BP.
- the IL-22 decoy receptor comprises IL-22BP (IL-22RA2) (e.g., human IL- 22BP, e.g., as exemplified by UniProt Accession Number Q969J5).
- IL-22BP IL-22RA2
- human IL- 22BP e.g., as exemplified by UniProt Accession Number Q969J5
- a macromolecule (i.e., ANDbody) of the invention also comprises an address target binder (second binding site) that binds to, e.g., specifically binds to or is specific for, an address target to provide targeted delivery of the effector.
- an “address target” is a structure on a cell or tissue whose expression is sufficiently restricted in an organism to allow it to identify an organ, tissue, cell, or cell state of interest in an organism.
- the address target can be, e.g., a cell surface protein, or a structure localizing to the extracellular matrix.
- “restricted” expression of an address target means that the address target has a differential, e.g., less broad, in vivo expression, as opposed to systemic expression.
- the address target is expressed, for example, in a single cell type, tissue or cell state in a mammalian subject, such as a human subject.
- an “IL-10 family address target” is a cell surface moiety (e.g., a protein, a lipid, or a sugar) that is expressed on the surface of a cell that also expresses an IL-10 family receptor (e.g., expresses an IL-22 receptor to which the IL-22 polypeptide or fragment or variant thereof comprised by the ANDbody binds).
- an IL-10 family address target is a cell surface moiety (e.g., a protein, a lipid, or a sugar) that is expressed on the surface of a cell that also expresses an IL-10 family receptor (e.g., expresses an IL-22 receptor to which the IL-22 polypeptide or fragment or variant thereof comprised by the ANDbody binds).
- an address target binder e.g., a second binding site
- an IL- 10 family address target provided in Table 1 that is expressed on the surface of the target cell.
- the terms “specifically binds” and “is specific for” refer to a preferential interaction between a binding domain and its target or ligand (such as binding between an antibody and an antigen or epitope) that may be determinative of the presence of the target or ligand in the presence of a heterogeneous population of molecules including biological molecules.
- a binding domain that specifically binds to a ligand may be a binding domain that binds this ligand with greater affinity, avidity, more readily, and/or with greater duration than it binds to other ligands.
- the extent of binding of a binding domain to an unrelated molecule (non-ligand) is less than about 10% of the binding of the binding domain to the ligand as measured using an appropriate assay.
- a binding domain that specifically binds to a ligand has a dissociation constant (KD) of ⁇ 1 pM, ⁇ 100 nM, ⁇ 10 nM, ⁇ 1 nM, or ⁇ 0.1 nM.
- KD dissociation constant
- Specific binding can include, but does not require exclusive binding.
- “specific binding” refers to binding wherein a binding domain binds to a particular ligand (e.g., a polypeptide or antigen or epitope on a particular polypeptide) without substantially binding to any other ligand (e.g., polypeptide or polypeptide antigen or epitope).
- IL-10 family address targets are presented in Table 1 , below.
- the second binding site is specific for an IL-10 family address target provided in Table 1 (e.g., is specific for is an IL-10 family address target provided in Table 1 that is expressed on the surface of a cell that also expresses an IL-10 family receptor (e.g., expresses IL-22 family receptor to which the IL-22 polypeptide or fragment or variant thereof comprised by the ANDbody binds)).
- the cell on which the IL-10 family address target is expressed is a an epithelial cell or a cell type from one or more tissues selected from small intestine, large intestine, lower gastrointestinal (Gl) tract, rectum, skin, pancreas, kidney, lungs, and liver.
- the first binding site of the ANDbody comprises IL-22 or a fragment or variant thereof, and the second binding site is specific for an IL-10 family address target provided in Table 1 .
- the target cell is a lower gastrointestinal (Gl) tract cell
- the IL-10 family address target is MS4A18, SI, MS4A12, ATP4B, MUC17, TMIGD1 , MS4A10, MEP1 A, CDH17, TM4SF20, SLC26A3, ATP4A, GPA33, BTNL3, MEP1 B, UGT2B17, KCNE2, or TMPRSS15
- the second binding site is specific for MS4A18, SI, MS4A12, ATP4B, MUC17, TMIGD1 , MS4A10, MEP1 A, CDH17, TM4SF20, SLC26A3, ATP4A, GPA33, BTNL3, MEP1 B, UGT2B17, KCNE2, or TMPRSS15
- the first binding site of the ANDbody comprises IL-22 or a fragment or variant thereof.
- the target cell is a kidney cell
- the IL-10 family address target is TMEM207, OR2T10, SLC22A6, SLC22A8, SLC12A1 , AQP2, SLC22A12, SLC12A3, TMEM174, FXYD4, SLC34A1 , KCNJ1 , SLC22A1 1 , TMEM52B, SLC6A18, CTXN3, SLC4A1 , TMEM72, SLC22A2, SLC4A9, BSND, CDH16, RDH8, or AQP6 (i.e., the second binding site is specific for TMEM207, OR2T10, SLC22A6, SLC22A8, SLC12A1 , AQP2, SLC22A12, SLC12A3, TMEM174, FXYD4, SLC34A1 , KCNJ1 , SLC22A1 1 , TMEM52B, SLC6A18, CTXN3, SLC4
- the target cell is a liver cell
- the IL-10 family address target is UGT2B10, C8A, SLCO1 B1 , C9, SLC25A47, SLC17A2, SLC10A1 , UGT2B4, SLC22A1 , or CYP8B1
- the second binding site is specific for UGT2B10, C8A, SLCO1 B1 , C9, SLC25A47, SLC17A2, SLC10A1 , UGT2B4, SLC22A1 , or CYP8B1
- the first binding site of the ANDbody comprises IL-22 or a fragment or variant thereof.
- the target cell is a pancreas cell
- the IL-10 family address target is GP2, AQP12A, AQP12B, CUZD1 , LHFPL5, AQP8, KIRREL2, G6PC2, SLC17A6, or GPR1 19
- the second binding site is specific for GP2, AQP12A, AQP12B, CUZD1 , LHFPL5, AQP8, KIRREL2, G6PC2, SLC17A6, or GPR1 19
- the first binding site of the ANDbody comprises IL-22 or a fragment or variant thereof.
- the target cell is a skin cell
- the IL-10 family address target is CLEC2A, KCNJ18, KLRF2, DSC1 , TYR, AWAT2, TMEM271 , DSG1 , ACER1 , GSDMA, TRPM1 , GJB4, CD1 A, ASPRV1 , or ABCA12
- the second binding site is specific for CLEC2A, KCNJ18, KLRF2, DSC1 , TYR, AWAT2, TMEM271 , DSG1 , ACER1 , GSDMA, TRPM1 , GJB4, CD1 A, ASPRV1 , or ABCA12
- the first binding site of the ANDbody comprises IL-22 or a fragment or variant thereof.
- the IL-10 family address target is selected from CDH17, GP2, CDH16, DSG1 , and DSG3.
- the IL-10 family address target is CDH17.
- the second binding site is an anti-CDH17 antibody or antigen-binding fragment thereof.
- the cell is a gut epithelial cell and the macromolecule is targeted to small intestine and colon tissues.
- the IL-10 family address target is CDH16.
- the second binding site is an anti-CDH16 antibody or antigen-binding fragment thereof.
- the cell is a kidney cell (e.g., an epithelial cell in the kidney tubules) and the macromolecule is targeted to the kidney.
- the IL-10 family address target is GP2.
- the second binding site is an anti-GP2 antibody or antigen-binding fragment thereof.
- the cell is a kidney cell and the macromolecule is targeted to the pancreas.
- the second binding site comprises an antibody.
- antibody includes full-length antibodies and antigen binding antibody fragments (e.g., scFvs).
- an antibody molecule has specificity for more than one, e.g., 2, 3, or 4 antigens, e.g., the antibody molecule comprises a plurality of variable domain sequences, wherein a first variable domain sequence of the plurality has binding specificity for a first epitope (e.g., the effector target) and a second variable domain sequence of the plurality has binding specificity for a second epitope (e.g., the address target).
- the antibody molecule is monospecific.
- the second binding site has the structure of an scFv, BsIgG, a BsAb fragment, a BiTE, a dual-affinity re-targeting protein (DART), a tandem diabody (TandAb), a diabody, an Fab2, a di-scFv, chemically linked F(ab’)2, an Ig molecule with 2, 3 or 4 different antigen binding sites, a DVI-IgG four-in-one, an ImmTac, an HSAbody, an IgG-IgG, a Cov-X-Body, an SCFV1 -PEG-SCFV2, an appended IgG, an DVD-IgG, an affibody, an affilin, an affimer, an affitin, an alphabody, an anticalin, an avimer, a DARPin, a Fynomer, a monobody, a nanoCLAMP,
- the second binding site (address target binder) comprises a ligand of the address target.
- the currently provided address target binding domains do not substantially influence biological signaling upon binding to the address target, e.g., do not modulate a signal transduction pathway or other biological response in the target cell or tissue.
- the address target binder can be inert or inactive, in which it lacks any additional activity (other than binding), including lacking catalytic activity, after binding to the address target.
- the address target binder binds a non-signaling site or motif of the address target. “Signal” is used herein to indicate a conformational, enzymatic, and/or electrical consequence occurs as a result of target binding. Accordingly, as described herein, address target binding domains do not signal upon address target binding.
- a domain that does not “substantially” influence biological signaling is a domain that modulates a signal transduction pathway or other biological response in the target cell or tissue to which it binds by no more than 25% relative to a control condition, e.g., relative to signaling in the absence of the domain.
- the domain may modulate (e.g., increase or decrease) the signal transduction pathway or other biological response by less than 20%, less than 15%, less than 10%, less than 5%, less than 2%, or less than 1% (e.g., 20-25%, 15-20%, 10-15%, 5-10%, 2- 5%, or 1 -2%).
- the address target is used for organ-specific addressing, tissue-specific addressing, or cell-specific addressing. In some embodiments, the address target is used or cell-specific addressing.
- a Gini coefficient (GO) score which is a method for assessing the expression variation of a particular gene in a data set.
- GO Gini coefficient
- Address target binders can be identified using cell expression data generated for address target binders.
- address target markers exhibit Gini scores of greater than 0.4, such as between 0.74 and 1 .00.
- non-address markers that are expressed more systemically may exhibit Gini Scores of between 0.15 to 0.19.
- a Tau score which represents the expression variation of a particular gene in a data set. Calculating Tau uses the information of expression of a gene in each tissue and its maximal expression over all tissues while also taking into account the number of tissues where expression is measured (see Itai Yanai, et al., Genome-wide midrange transcription profiles reveal expression level relationships in human tissue specification, Bioinformatics, Volume 21 , Issue 5, 1 March 2005, Pages 650-659; Kryuchkova-Mostacci N, Robinson-Rechavi M. A benchmark of gene expression tissue-specificity metrics. Brief Bioinform. 2017 Mar 1 ;18(2):205-214. doi: 10.1093/bib/bbw008).
- address target markers exhibit Tau scores of greater than 0.6, such as between 0.74 and 1 .00.
- non-address markers that are expressed more systemically may exhibit Tau Scores of below 0.3, such as 0.15 to 0.19.
- specificity of address target binding domains for a particular cell or tissue is determined with a tissue based analysis that does not include tissues having a natural biological separation barrier (i.e., blood-brain barrier).
- Gini and/or Tau scores may be calculated without data from tissues such as (but not limited to): central nervous system, brain, eye, and/or testis tissues.
- an address target as provided herein identifies a cell state.
- a “cell state” refers to a given physiological condition of a cell.
- a cell state may be, e.g., a disease state (relative to a non-disease state or normal state of a cell or tissue); or an activated state (relative to a non-activated state of a cell).
- disease states include inflammation, infection (e.g., bacterial, viral, or fungal infection), and states relating to cancer (e.g., precancerous or cancerous cell states).
- cell state reflects the fact that cells of a particular type can exhibit variability with regard to one or more features and/or can exist in a variety of different conditions, while retaining the features of their particular cell type and not gain features that would cause them to be classified as a different cell type.
- the different states or conditions in which a cell can exist may be characteristic of a particular cell type (e.g., may involve properties or characteristics exhibited only by that cell type and/or involve functions performed only or primarily by that cell type) or may occur in multiple different cell types.
- a cell state reflects the capability of a cell to respond to a particular stimulus or environmental condition (e.g., whether or not the cell will respond, or the type of response that will be elicited) or is a condition of the cell brought about by a stimulus or environmental condition.
- Cells in different cell states may be distinguished from one another in a variety of ways.
- a cell state may be a condition of the cell in which the cell expresses, produces, or secretes one or more markers, exhibits particular protein modification(s), has a particular appearance, and/or will or will not exhibit one or more biological response(s) to a stimulus or environmental condition.
- an effector target binding domain e.g., an IL-22 polypeptide or fragment or variant thereof, e.g., a detuned or attenuated IL-22 or fragment or variant thereof (e.g., an IL-22 mutein polypeptide), e.g., as described in Section B(i)(a), above
- an effector target binding domain may not substantially signal, or may not signal at all, when it is not localized by an address target binding domain.
- an effector target binding domain signals with higher potency (e.g., has higher avidity) when it is localized by an address target binding domain compared to the signal when it is not localized by an address target binding domain.
- effector target signaling can be influenced as discussed above. /. Relative Binding Affinity of First and Second Binding Sites
- the affinity of the effector target binding site and address target binding site of an ANDbody for their respective binding partners may differ.
- the affinity of the first binding site to the therapeutic effector target it binds is weaker than the affinity of the second binding site to the address target.
- the affinity of the first binding site to the therapeutic effector target it binds is more than 2-fold, 3-fold, 4-fold, 5-fold, 10-fold, 15-fold, 20-fold, 50-fold, 100-fold, 200-fold, 500-fold, 1000-fold weaker than the affinity of the second binding site to the address target.
- binding affinity and “binding activity” refer to the tendency of a macromolecule, e.g., a polypeptide molecule, to bind or not to bind to a target.
- binding affinity and “binding activity” refer to the tendency of a macromolecule, e.g., a polypeptide molecule, to bind or not to bind to a target.
- the relative affinities of the two binding sites can be determined by, for example, measuring their respective affinities when each binding site is present on a common scaffold, such as in the form of a single chain antibody. Such a comparison allows a comparison of the affinities of two binding sites while eliminating any interference from other binding sites present on the macromolecule of the present invention.
- Binding affinity may be quantified by determining the dissociation constant (Kd; KD) for a polypeptide and its binder. A lower Kd is indicative of a higher affinity for a binding partner.
- Kd dissociation constant
- the specificity of binding of a polypeptide to its binding partner may be defined in terms of the comparative dissociation constants (Kd) of the polypeptide for its binding partner as compared to the dissociation constant with respect to the polypeptide and another, non-target molecule.
- this dissociation constant can be determined by known methods.
- the Kd may be established using a double-filter nitrocellulose filter binding assay such as that disclosed by Wong & Lohman (Proc. Natl. Acad. Sci. USA 90, 5428-5432, 1993).
- Other standard assays to evaluate the binding ability of ligands such as antibodies towards targets are known in the art, including for example, ELISAs, Western blots, RIAs, and flow cytometry analysis.
- the binding kinetics (e.g., binding affinity) of the antibody also can be assessed by standard assays known in the art, such as by BiacoreTM system analysis.
- ECso or IC50 may be used to determine relative affinities.
- EC50 indicates the concentration at which a polypeptide achieves 50% of its maximum binding to a fixed quantity of binding partner.
- IC50 indicates the concentration at which a polypeptide inhibits 50% of the maximum binding of a fixed quantity of competitor to a fixed quantity of binding partner.
- a lower level of EC50 or IC50 indicates a higher affinity for a target.
- the EC50 and IC50 values of an ANDbody binding site for its binding partner can both be determined by well-known methods, for example ELISA.
- the Kd of therapeutic effector target binder might be higher than about 1 pM, about 10pM, about 10OpM, about 1 nM, about 10nM, about 10OnM, about 500nM, or about 1 uM (e.g., may be between 1 pM and 10pM, between 10 pM and 100pM, between 100 pM and 1 nM, between 1 nM and 10 nM, between 10 nM and 100 nM, between 100 nM and 500nM, or between 500 nM and 1 uM).
- the Kd of the address target binder might be less than about 1 uM, about 500nM, about 10OnM, about 10nM, about 1 nM, about 10OpM, about 10pM, or about 1 pM (e.g., may be between 1 uM and 500nM, between 500 nM and 10OnM, between 100 nM and 10nM, between 10 nM and 1 nM, between 1 nM and 10OpM, between 100 pM and 10 pM, or between 10pM and 1 pM).
- the Kd for the therapeutic effector target binder may be about 6-fold, about 5-fold, about 4- fold, about 3-fold, or about 2-fold higher than the Kd for the address target binder.
- the ECso of therapeutic effector target binder might be higher than about 1 pM, about 10pM, about 10OpM, about 1 nM, about 10nM, about 10OnM, about 500nM, or about 1 uM (e.g., may be between 1 pM and 10pM, between 10 pM and 100pM, between 100 pM and 1 nM, between 1 nM and 10 nM, between 10 nM and 100 nM, between 100 nM and 500nM, or between 500 nM and 1 uM).
- the ECso of the address target binder might be less than about 1 uM, about 500nM, about 10OnM, about 10nM, about 1 nM, about 10OpM, about 10pM, or about 1 pM (e.g., may be between 1 uM and 500nM, between 500 nM and 10OnM, between 100 nM and 10nM, between 10 nM and 1 nM, between 1 nM and 10OpM, between 100 pM and 10 pM, or between 10pM and 1 pM).
- the ECso for the therapeutic effector target binder may be about 6-fold, about 5-fold, about 4-fold, about 3-fold, or about 2-fold higher than the ECso for the address target binder.
- the IC50 of therapeutic effector target binder might be higher than about 1 pM, about 10pM, about 10OpM, about 1 nM, about 10nM, about 10OnM, about 500nM, or about 1 uM (e.g., may be between 1 pM and 10pM, between 10 pM and 100pM, between 100 pM and 1 nM, between 1 nM and 10 nM, between 10 nM and 100 nM, between 100 nM and 500nM, or between 500 nM and 1 uM).
- the IC50 of the address target binder might be less than about 1 uM, about 500nM, about 10OnM, about 10nM, about 1 nM, about 10OpM, about 10pM, or about 1 pM (e.g., may be between 1 uM and 500nM, between 500 nM and 10OnM, between 100 nM and 10nM, between 10 nM and 1 nM, between 1 nM and 10OpM, between 100 pM and 10 pM, or between 10pM and 1 pM).
- the IC50 for the therapeutic effector target binder may be about 6-fold, about 5-fold, about 4-fold, about 3-fold, or about 2-fold higher than the IC50 for the address target binder.
- the cellular or tissue density of the effector target and address target bound by an ANDbody may differ.
- the density of the therapeutic effector target on a cell bound by the effector target binding site of an ANDbody is more than about 2-fold, about 3-fold, about 4-fold, about 5-fold, about 10-fold, about 15-fold, about 20-fold, about 50-fold, about 100-fold, about 200-fold, about 500-fold, about 1000-fold, about 10,000-fold, about 100,000-fold less than the density of the address target on a cell bound by the address target binding site.
- the affinity of the first binding site to the therapeutic effector target it binds is about one-half (1/2) X Kd less than the affinity of the second binding site to the address target it binds and the density of the therapeutic effector target on a cell bound by the first binding site is about one-half (I/2) X Kd less than the density of the address target on a cell bound by the second binding site.
- the ANDbody has both the affinity and density parameters as described hereinabove.
- ANDbodies with binding sites with altered affinities can be made using methods known in the art, e.g., an ANDbody can be engineered to have a target binding site that has decreased affinity for the effector target. See, e.g., US Patent No. 10,654,928.
- an ANDbody may be modified to alter the affinity of an effector target binding site to its effector target or to alter the affinity of an address target binding site to its address target. The modification can increase or decrease affinity for the binding site’s binding partner.
- a macromolecule of the invention may be linked to a small molecule.
- the macromolecule and the small molecule may be linked by a cleavable linker.
- the macromolecule and the small molecule may be linked by a non- cleavable linker. Any useful linker may be employed for this purpose.
- One or more (for example, one, two, three, four, five, or more) small molecules may be linked to the macromolecule. If multiple small molecules are linked to a macromolecule, the small molecules may be the same. Alternatively, one or more of the small molecules linked to the macromolecule may be different.
- a small molecule to be linked to the macromolecule may be any desired small molecule.
- the small molecule may be a therapeutic agent of interest that is to be localized or concentrated at a particular site by the macromolecule.
- the small molecule may be a therapeutic agent that acts together with or complements the effector target binding site domain.
- the small molecule may modulate the effector target binding site domain.
- the small molecule may modulate the address target binding site domain.
- Small molecules may be conjugated to a macromolecule of the invention using any conjugation technique known in the art.
- small molecule carboxy, hydroxyl, and amine residues may be joined to amine and sulfhydryl residues on proteins using linkage techniques.
- any complementary functional groups on the two components may be used to react with each other to form a covalent bond.
- complementary reactive functional groups include, but are not limited to, e.g., maleimide and cysteine, amine and activated carboxylic acid, thiol and maleimide, activated sulfonic acid and amine, isocyanate and amine, azide and alkyne, and alkene and tetrazine.
- any available linker may be utilized in the invention including heterobifunctional linkers that allow attachment of small molecules through, for example, disulfide bonds and amide bonds.
- Polypeptide components of the macromolecules (ANDbodies and/or IL-22 mutein polypeptides) of the invention may be produced by any suitable means.
- all or part of an ANDbody or an IL-22 mutein polypeptide may be expressed by a host cell comprising one or more nucleotides which encode the ANDbody or IL-22 mutein polypeptide.
- the invention provides one or more nucleic acids encoding any one of the macromolecules provided herein or one or more subunits thereof.
- the nucleic acids may be, e.g., linear nucleic acids, circular nucleic acids, and/or modified nucleic acids.
- the invention further provides one or more vectors comprising the one or more nucleic acids encoding any one of the macromolecules provided herein or one or more subunits thereof.
- the invention also provides one or more host cells (e.g., mammalian host cells) comprising one or more nucleic acids encoding any one of the macromolecules provided herein or one or more subunits thereof (e.g., comprising vectors comprising said one or more nucleic acids).
- the invention further provides a method of producing any one of the macromolecules provided herein, the method comprising culturing the one or more host cells in a culture medium.
- the method may further comprise recovering the macromolecule from the one or more host cells or the culture medium.
- Methods for producing an ANDbody and/or an IL-22 mutein polypeptide may involve expression in mammalian cells, although recombinant proteins can also be produced using insect cells, yeast, bacteria, or other cells under the control of appropriate promoters.
- Mammalian expression vectors may comprise nontranscribed elements such as an origin of replication, a suitable promoter and enhancer, and other 5' or 3' flanking nontranscribed sequences, and 5' or 3' nontranslated sequences such as necessary ribosome binding sites, a polyadenylation site, splice donor and acceptor sites, and termination sequences.
- DNA sequences derived from the SV40 viral genome for example, SV40 origin, early promoter, enhancer, splice, and polyadenylation sites may be used to provide the other genetic elements required for expression of a heterologous DNA sequence.
- Appropriate cloning and expression vectors for use with bacterial, fungal, yeast, and mammalian cellular hosts are described in Green & Sambrook, Molecular Cloning: A Laboratory Manual (Fourth Edition), Cold Spring Harbor Laboratory Press (2012).
- mammalian cell culture systems can be employed to express and manufacture an ANDbody and/or an IL-22 mutein polypeptide described herein.
- mammalian expression systems include CHO cells, COS cells, HeLA and BHK cell lines.
- Processes of host cell culture for production of protein therapeutics are described in, e.g., Zhou and Kantardjieff (Eds.), Mammalian Cell Cultures for Biologies Manufacturing (Advances in Biochemical Engineering/Biotechnology), Springer (2014). Purification of protein therapeutics is described in Franks, Protein Biotechnology: Isolation, Characterization, and Stabilization, Humana Press (2013); and in Cutler, Protein Purification Protocols (Methods in Molecular Biology), Humana Press (2010). Formulation of protein therapeutics is described in Meyer (Ed.), Therapeutic Protein Drug Products: Practical Approaches to formulation in the Laboratory, Manufacturing, and the Clinic, Woodhead Publishing Series (2012).
- Antibody production techniques are known. See, for example, Zhiqiang (Editor), Therapeutic Monoclonal Antibodies: From Bench to Clinic. 1st Edition. Wiley 2009; Greenfield (Ed.) Antibodies: A Laboratory Manual. (Second edition) Cold Spring Harbor Laboratory Press 2013; Ferrara et al. 2012. Using Phage and Yeast Display to Select Hundreds of Monoclonal Antibodies: Application to Antigen 85, a Tuberculosis Biomarker.
- ANDbody and/or IL-22 mutein polypeptide RNAs may be produced, e.g., for delivery to a subject.
- therapeutic mRNAs are made by in vitro transcription. Modification such as incorporation of modified bases, 5’cap analogues, and polyA tails can optimize activity and function. For example, translation and stability of mRNA can be accomplished, by cap and poly A tail modifications.
- cap analogs such as ARCA (anti-reverse cap analogs) and a poly(A) tail of 100-200 bp into in vitro transcribed (IVT) mRNAs improves expression and stability (Kaczmarek et al. Genome Medicine (2017) 9:60).
- New types of cap analogs such as 1 ,2-dithiodiphosphate-modified caps, can further improve efficiency of translation (Strenkowska et al. Nucleic Acids Res. 2016;44:9578-90). Codon optimization can also improve efficacy of protein synthesis and limit mRNA destabilization by rare codons (Presnyak et al. Cell. 2015;160:1111-24.
- UTRs 3' and 5' untranslated regions
- RBPs RNA-binding proteins
- miRNAs 3' and 5' untranslated regions
- RBPs RNA-binding proteins
- miRNAs 3' and 5' untranslated regions
- RBPs RNA-binding proteins
- miRNAs 3' and 5' untranslated regions
- Kaczmarek 3' and 5' untranslated regions
- UTRs can be modified to encode regulatory elements (e.g., K-turn motifs and miRNA binding sites), in order to control RNA expression in a cell-specific manner (Wroblewska et al. Nat Biotechnol. 2015;33:839-41 ).
- RNA base modifications e.g., pseudouridine incorporated mRNA, e.g., N1 -methyl-pseudouridine
- RNA base modifications contribute to masking mRNA immune-stimulatory activity and increase mRNA translation by enhancing translation initiation (Andries et al. J Control Release. 2015;217:337-44; Svitkin et al. Nucleic Acids Res.
- mRNA compositions and methods of their manufacture are known and are disclosed, e.g., in WO2016011306; WO2016014846; WO2016022914; WO2016077123; WO2016164762;
- RNA datasets include those a genotype-Tissue Expression (GTEx) dataset (see, e.g., https://www.genome.gov/Funded-Programs- Projects/Genotype-Tissue-Expression-Project) or a Human Protein Atlas (HPA) dataset (https //www . prote i n atl as . o rg/) .
- GTEx genotype-Tissue Expression
- HPA Human Protein Atlas
- tissues in which expression of the therapeutic target can be assessed includes, e.g., the pancreas, adrenal gland, liver, kidney (cortex), kidney (medulla), gastrointestinal (Gl) tract, lower Gl tract, small intestine colon, transverse colon, sigmoid colon, terminal ileum, skin, non-sun- exposed skin, sun-exposed skin, spleen, stomach,.
- Address markers can be assessed using methods well known in the art, e.g., gene expression can be assessed at the mRNA level using Northern blots, cDNA or oligonucleotide microarrays, or sequencing ⁇ e.g., RNA-Seq), or at the level of protein expression using protein microarrays, Western blots, flow cytometry, immunohistochemistry, etc. Modifications can be assessed, e.g., using antibodies that are specific for a particular modified form of a protein, e.g., phospho-specific antibodies, or mass spectrometry.
- ANDbody molecules e.g., ANDbodies
- IL-22 mutein polypeptides and their pharmaceutical compositions provided herein are suitable for administration to a subject in need thereof, wherein the subject is a human or a non-human animal, for example, suitable for human therapeutic or veterinary use.
- a method comprising administering a therapeutically effective amount of any one of the macromolecules (i.e., ANDbody molecules, ANDbodies, or IL-22 mutein polypeptides) provided herein, a composition comprising the same, or one or more nucleic acids encoding the same to a subject in need thereof.
- Veterinary use includes use for treatment of mammals, including commercially relevant mammals, e.g., pet and live-stock animals, such as cattle, pigs, horses, sheep, goats, cats, dogs, mice, and/or rats; and/or birds, including commercially relevant birds such as parrots, poultry, chickens, ducks, geese, hens or roosters and/or turkeys; zoo animals, e.g., a feline; non-mammal animals, e.g., reptiles, fish, amphibians, etc.
- mammals including commercially relevant mammals, e.g., pet and live-stock animals, such as cattle, pigs, horses, sheep, goats, cats, dogs, mice, and/or rats; and/or birds, including commercially relevant birds such as parrots, poultry, chickens, ducks, geese, hens or roosters and/or turkeys; zoo animals, e.g., a feline; non-mam
- the invention is further directed to a subject or subject cell comprising the ANDbody or IL-22 mutein polypeptide composition described herein.
- the subject or subject cell is a plant, insect, bacteria, fungus, vertebrate, mammal (e.g., human), or other organism or cell.
- a subject or a subject cell is contacted with (e.g., delivered to or administered to) the ANDbody composition or IL-22 mutein polypeptide.
- the subject is a mammal, such as a human.
- the amount of the ANDbody composition or IL-22 mutein polypeptide, expression product, or both in the subject can be measured at any time after administration.
- a subject to which an ANDbody, ANDbody composition, or IL-22 mutein polypeptide provided herein is administered has, is suspected of having, or is at increased risk of developing a disease or disorder selected from Crohn's disease, ulcerative colitis, graft-vs-host disease, atopic dermatitis, psoriasis, wounds, diabetic skin ulcers, acute pancreatitis, chronic pancreatitis, autoimmune pancreatitis, nephritis, acute lung injury, viral lung injury, fibrosis, fibrosis, non-alcoholic steatohepatitis, viral hepatitis, and autoimmune hepatitis.
- a disease or disorder selected from Crohn's disease, ulcerative colitis, graft-vs-host disease, atopic dermatitis, psoriasis, wounds, diabetic skin ulcers, acute pancreatitis, chronic pancreatitis, autoimmune pancreatitis, nephriti
- a method of potentiating activation of a signaling pathway in a tissue or cell, the method comprising contacting the tissue or cell with a macromolecule provided herein, wherein the tissue or cell expresses an IL-10 family receptor and an IL-10 family address target, and wherein neither the first binding site (IL-22 polypeptide or fragment or variant thereof) nor the second binding site (IL-10 family address target binder) substantially activates the signaling pathway in the absence of localization by the second binding site; wherein upon contacting the tissue or cell with the macromolecule, activation of the signaling pathway in the tissue or cell by the first binding site is substantially increased relative to a reference macromolecule lacking the second binding site.
- a signaling pathway e.g., a cytokine signaling pathway
- a method of modulating IL-22 receptor signaling in a target cell of a subject comprising administering to the subject any one of the macromolecules (e.g., ANDbodies and/or IL-22 mutein polypeptides) provided herein.
- the macromolecules e.g., ANDbodies and/or IL-22 mutein polypeptides
- the ANDbody and IL-22 mutein polypeptide compositions described herein ⁇ e.g., ANDbody and/or IL-22 mutein polypeptide, nucleic acid, or RNA compositions) and the polypeptide compositions described herein may be administered to a subject in need thereof.
- the invention includes pharmaceutical compositions that include a macromolecule provided herein (i.e. , an ANDbody or IL-22 mutein polypeptide), e.g., combination in with one or more pharmaceutically acceptable carriers or excipients.
- the invention includes pharmaceutical compositions that include a polypeptide provided herein, e.g., in combination with one or more pharmaceutically acceptable carriers or excipients.
- the composition is a unit dosage form suitable for administration to a human subject (e.g., intravenous, oral, or subcutaneous administration).
- Nucleic acids ⁇ e.g., RNA) encoding an ANDbody and/or an IL-22 mutein polypeptide can alternatively or additionally be administered to a subject.
- therapeutic mRNAs are made by in vitro transcription. Modification such as incorporation of modified bases, 5’cap analogues, and polyA tails can optimize activity and function.
- translation and stability of mRNA can be accomplished, by cap and poly A tail modifications.
- incorporation of cap analogs such as ARCA (anti-reverse cap analogs) and a poly(A) tail of 100-200 bp into in vitro transcribed (IVT) mRNAs improves expression and stability (Kaczmarek et al.
- UTRs can be modified to encode regulatory elements (e.g., K-turn motifs and miRNA binding sites), in order to control RNA expression in a cell-specific manner (Wroblewska et al. Nat Biotechnol. 2015;33:839-41 ).
- RNA base modifications e.g., pseudouridine incorporated mRNA, e.g., N1 - methyl-pseudouridine
- RNA base modifications contribute to masking mRNA immune-stimulatory activity and increase mRNA translation by enhancing translation initiation (Andries et al. J Control Release. 2015;217:337-44; Svitkin et al. Nucleic Acids Res. 2017;45:6023-36).
- mRNA compositions and methods of their manufacture are known and are disclosed, e.g., in WO2016011306; WO2016014846; WO2016022914; WO2016077123; WO2016164762; WO2016201377; WO2017049275; US9937233; US8710200; US10022425; US9878056; US9572897; Jemielity et al. RNA. 2003;9:1108-22. 90; Mockey et al. Biochem Biophys Res Commun. 2006;340:1062-8. 91 ; Strenkowska et al. Nucleic Acids Res. 2016;44:9578-90. 92; Presnyak et al. Cell.
- the RNA is a circular RNA. See, for example, WO2019118919, describing the expression of a therapeutic RNA, such as an antibody RNA, from a circular RNA.
- the invention includes a circular polyribonucleotide that comprises (a) an internal ribosome entry site (IRES), (b) an expression sequence encoding a ANDbody described herein and lacking a poly-A sequence, and (c) a termination element.
- IRS internal ribosome entry site
- a circular RNA encoding an ANDbody described herein may be delivered naked (i.e. , without formulation with a carrier) or with a carrier.
- an ANDbody or ANDbody composition provided herein is administered in combination with one or more additional therapeutic agents.
- an IL-22 mutein polypeptide or IL-22 mutein polypeptide composition provided herein is administered in combination with one or more additional therapeutic agents.
- Formulations of the compositions described herein for in vivo delivery with a carrier include lipid nanoparticle (LNP) formulations.
- LNP lipid nanoparticle
- LNPs in some embodiments, comprise one or more ionic lipids, such as non-cationic lipids (e.g., neutral or anionic, or zwitterionic lipids); one or more conjugated lipids (such as PEG-conjugated lipids or lipids conjugated to polymers described in Table 5 of WO2019217941 ; incorporated herein by reference in its entirety); one or more sterols (e.g., cholesterol); and, optionally, one or more targeting molecules (e.g., conjugated receptors, receptor ligands, antibodies); or combinations of the foregoing.
- ionic lipids such as non-cationic lipids (e.g., neutral or anionic, or zwitterionic lipids)
- conjugated lipids such as PEG-conjugated lipids or lipids conjugated to polymers described in Table 5 of WO2019217941 ; incorporated herein by reference in its entirety
- sterols e.g.,
- Lipids that can be used in nanoparticle formations include, for example those described in Table 4 of WO2019217941 , which is incorporated herein by reference — e.g., a lipid- containing nanoparticle can comprise one or more of the lipids in Table 4 of WO2019217941 .
- Lipid nanoparticles can include additional elements, such as polymers, such as the polymers described in Table 5 of WO2019217941 , incorporated by reference.
- conjugated lipids when present, can include one or more of PEG- diacylglycerol (DAG) (such as l-(monomethoxy-polyethyleneglycol)-2,3- dimyristoylglycerol (PEG-DMG)), PEG-dialkyloxypropyl (DAA), PEG-phospholipid, PEG- ceramide (Cer), a pegylated phosphatidylethanoloamine (PEG-PE), PEG succinate diacylglycerol (PEGS-DAG) (such as 4-0-(2',3'- di(tetradecanoyioxy)propyi-l-0-(w- methoxy(polyethoxy)ethyl) butanedioate (PEG-S-DMG)), PEG dialkoxypropylcarbam, N- (carbonyl-methoxypoly ethylene glycol 2000)- 1 ,2-distearoyl-sn-
- DAG P
- sterols that can be incorporated into lipid nanoparticles include one or more of cholesterol or cholesterol derivatives, such as those in W02009/127060 or US2010/0130588, which are incorporated by reference. Additional exemplary sterols include phytosterols, including those described in Eygeris et al (2020), dx.doi.org/10.1021/acs.nanolett.0c01386, incorporated herein by reference.
- the lipid particle comprises an ionizable lipid, a non-cationic lipid, a conjugated lipid that inhibits aggregation of particles, and a sterol. The amounts of these components can be varied independently and to achieve desired properties.
- the lipid nanoparticle comprises an ionizable lipid is in an amount from about 20 mol % to about 90 mol % of the total lipids (in other embodiments it may be 20-70% (mol), 30-60% (mol) or 40-50% (mol); about 50 mol % to about 90 mol % of the total lipid present in the lipid nanoparticle), a non-cationic lipid in an amount from about 5 mol % to about 30 mol % of the total lipids, a conjugated lipid in an amount from about 0.5 mol % to about 20 mol % of the total lipids, and a sterol in an amount from about 20 mol % to about 50 mol % of the total lipids.
- the ratio of total lipid to nucleic acid can be varied as desired.
- the total lipid to nucleic acid (mass or weight) ratio can be from about 10: 1 to about 30: 1 .
- the lipid to nucleic acid ratio (mass/mass ratio; w/w ratio) can be in the range of from about 1 : 1 to about 25: 1 , from about 10: 1 to about 14: 1 , from about 3 : 1 to about 15: 1 , from about 4: 1 to about 10: 1 , from about 5: 1 to about 9: 1 , or about 6: 1 to about 9: 1 .
- the amounts of lipids and nucleic acid can be adjusted to provide a desired N/P ratio, for example, N/P ratio of 3, 4, 5, 6, 7, 8, 9, 10 or higher.
- the lipid nanoparticle formulation’s overall lipid content can range from about 5 mg/ml to about 30 mg/mL.
- lipid compounds that may be used (e.g., in combination with other lipid components) to form lipid nanoparticles for the delivery of compositions described herein, e.g., nucleic acid (e.g., RNA) described herein includes,
- an LNP comprising Formula (i) is used to deliver an ANDbody RNA composition described herein to the liver and/or hepatocyte cells.
- an LNP comprising Formula (ii) is used to deliver an ANDbody RNA composition described herein to the liver and/or hepatocyte cells.
- an LNP comprising Formula (iii) is used to deliver an ANDbody RNA composition described herein to the liver and/or hepatocyte cells.
- an LNP comprising Formula (v) is used to deliver an ANDbody RNA composition described herein to the liver and/or hepatocyte cells.
- an LNP comprising Formula (vi) is used to deliver an ANDbody RNA composition described herein to the liver and/or hepatocyte cells.
- an LNP comprising Formula (viii) is used to deliver an ANDbody RNA composition described herein to the liver and/or hepatocyte cells.
- an LNP comprising Formula (ix) is used to deliver an ANDbody RNA composition described herein to the liver and/or hepatocyte cells.
- X1 is O, NR1 , or a direct bond
- X2 is C2-5 alkylene
- R1 is H or Me
- R3 is Ci-3 alkyl
- R2 is Ci-3 alkyl
- R2 taken together with the nitrogen atom to which it is attached and 1 -3 carbon atoms of X2 form a 4-, 5-, or 6-membered ring
- X1 is NR1 , R1 and R2 taken together with the nitrogen atoms to which they are attached form a 5- or 6-membered ring, or R2 taken together with R3 and the nitrogen atom to which they are attached form a 5-, 6-, or 7-membered ring
- Y1 is C2-12 alkylene
- Y2 is selected from , (in either orientation), , p at if Z1 is a direct bond, Z2 is absent;
- R5 is C5-9 alkyl or C6-10 alkoxy
- R6 is C5-9 alkyl or C6-10 alkoxy
- W is methylene or a direct bond
- an LNP comprising Formula (xii) is used to deliver an ANDbody RNA composition described herein to the liver and/or hepatocyte cells.
- an LNP comprising Formula (xi) is used to deliver an ANDbody RNA composition described herein to the liver and/or hepatocyte cells.
- an LNP comprises a compound of Formula (xiii) and a compound of Formula (xiv). In some embodiments an LNP comprising Formula (xv) is used to deliver an ANDbody RNA composition described herein to the liver and/or hepatocyte cells. In some embodiments an LNP comprising a formulation of Formula (xvi) is used to deliver an
- RNA composition described herein to the lung endothelial cells.
- a lipid compound used to form lipid nanoparticles for the delivery of compositions described herein, e.g., nucleic acid (e.g., RNA) described herein is made by one of the following reactions:
- a composition described herein e.g., a nucleic acid or a protein
- an LNP that comprises an ionizable lipid.
- the ionizable lipid is heptadecan-9-yl 8-((2-hydroxyethyl)(6-oxo-6-(undecyloxy)hexyl)amino)octanoate (SM-102); e.g., as described in Example 1 of US9,867,888 (incorporated by reference herein in its entirety).
- the ionizable lipid is 9Z,12Z)-3-((4,4-bis(octyloxy)butanoyl)oxy)-2-((((3- (diethylamino)propoxy)carbonyl)oxy)methyl)propyl octadeca-9,12-dienoate (LP01 ), e.g., as synthesized in Example 13 of WO2015/095340 (incorporated by reference herein in its entirety).
- the ionizable lipid is Di((Z)-non-2-en-1 -yl) 9-((4-dimethylamino)butanoyl)oxy)heptadecanedioate (L319), e.g. as synthesized in Example 7, 8, or 9 of US2012/0027803 (incorporated by reference herein in its entirety).
- the ionizable lipid is 1 ,1 '-((2-(4-(2-((2-(Bis(2- hydroxydodecyl)amino)ethyl)(2-hydroxydodecyl) amino)ethyl)piperazin-1 -yl)ethyl)azanediyl)bis(dodecan-2- ol) (C12-200), e.g., as synthesized in Examples 14 and 16 of WO2010/053572 (incorporated by reference herein in its entirety).
- the ionizable lipid is Imidazole cholesterol ester (ICE) lipid (3S, 10R, 13R, 17R)-10, 13-dimethyl-17- ((R)-6-methylheptan-2-yl)-2, 3, 4, 7, 8, 9, 10, 1 1 , 12, 13, 14, 15, 16, 17-tetradecahydro-IH- cyclopenta[a]phenanthren-3-yl 3-(1 H-imidazol-4-yl)propanoate, e.g., Structure (I) from W02020/106946 (incorporated by reference herein in its entirety).
- ICE Imidazole cholesterol ester
- an ionizable lipid may be a cationic lipid, an ionizable cationic lipid, e.g., a cationic lipid that can exist in a positively charged or neutral form depending on pH, or an amine-containing lipid that can be readily protonated.
- the cationic lipid is a lipid capable of being positively charged, e.g., under physiological conditions.
- Exemplary cationic lipids include one or more amine group(s) which bear the positive charge.
- the lipid particle comprises a cationic lipid in formulation with one or more of neutral lipids, ionizable amine-containing lipids, biodegradable alkyne lipids, steroids, phospholipids including polyunsaturated lipids, structural lipids (e.g., sterols), PEG, cholesterol and polymer conjugated lipids.
- the cationic lipid may be an ionizable cationic lipid.
- An exemplary cationic lipid as disclosed herein may have an effective pKa over 6.0.
- a lipid nanoparticle may comprise a second cationic lipid having a different effective pKa (e.g., greater than the first effective pKa), than the first cationic lipid.
- a lipid nanoparticle may comprise between 40 and 60 mol percent of a cationic lipid, a neutral lipid, a steroid, a polymer conjugated lipid, and a therapeutic agent, e.g., a nucleic acid (e.g., RNA) described herein, encapsulated within or associated with the lipid nanoparticle.
- the nucleic acid is co-formulated with the cationic lipid.
- the nucleic acid may be adsorbed to the surface of an LNP, e.g., an LNP comprising a cationic lipid.
- the nucleic acid may be encapsulated in an LNP, e.g., an LNP comprising a cationic lipid.
- the lipid nanoparticle may comprise a targeting moiety, e.g., coated with a targeting agent.
- the LNP formulation is biodegradable.
- a lipid nanoparticle comprising one or more lipid described herein, e.g., Formula (i), (ii), (ii), (vii) and/or (ix) encapsulates at least 1 %, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, at least 50%, at least 60%, at least 70%, at least 80%, at least 90%, at least 92%, at least 95%, at least 97%, at least 98% or 100% of an RNA molecule.
- Formula (i), (ii), (ii), (vii) and/or (ix) encapsulates at least 1 %, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, at least 50%, at least 60%, at least 70%, at least 80%, at least 90%, at least 92%, at least 95%, at least 97%, at least 98% or 100% of an RNA molecule.
- Exemplary ionizable lipids that can be used in lipid nanoparticle formulations include, without limitation, those listed in Table 1 of WO2019051289, incorporated herein by reference. Additional exemplary lipids include, without limitation, one or more of the following formulae: X of US2016/031 1759; I of US201503761 15 or in US2016/0376224; I, II or III of US20160151284; I, IA, II, or HA of US20170210967; l-c of US20150140070; A of US2013/0178541 ; I of US2013/0303587 or US2013/0123338; I of US2015/0141678; II, III, IV, or V of US2015/0239926; I of US2017/01 19904; I or II of WO2017/1 17528; A of US2012/0149894; A of US2015/0057373; A of WO2013/1 16126; A of US2013/0090372; A of US2013
- the ionizable lipid is MC3 (6Z,9Z,28Z,3 IZ)-heptatriaconta- 6,9,28,3 I- tetraen-l9-yl-4-(dimethylamino) butanoate (DLin-MC3-DMA or MC3), e.g., as described in Example 9 of WO2019051289A9 (incorporated by reference herein in its entirety).
- the ionizable lipid is the lipid ATX-002, e.g., as described in Example 10 of WO2019051289A9 (incorporated by reference herein in its entirety).
- the ionizable lipid is (l3Z,l6Z)-A,A-dimethyl-3- nonyldocosa-13, 16-dien-l-amine (Compound 32), e.g., as described in Example 1 1 of WO2019051289A9 (incorporated by reference herein in its entirety).
- the ionizable lipid is Compound 6 or Compound 22, e.g., as described in Example 12 of WO2019051289A9 (incorporated by reference herein in its entirety).
- non-cationic lipids include, but are not limited to, distearoyl-sn-glycero- phosphoethanolamine, distearoylphosphatidylcholine (DSPC), dioleoylphosphatidylcholine (DOPC), dipalmitoylphosphatidylcholine (DPPC), dioleoylphosphatidylglycerol (DOPG), dipalmitoylphosphatidylglycerol (DPPG), dioleoyl-phosphatidylethanolamine (DOPE), palmitoyloleoylphosphatidylcholine (POPC), palmitoyloleoylphosphatidylethanolamine (POPE), dioleoylphosphatidylethanolamine 4-(N-maleimidomethyl)-cyclohexane- 1 - carboxylate (DOPE-mal), dipalmitoyl phosphatidyl ethanolamine (DPPE), dimyristoylphosphoethanolamine (
- acyl groups in these lipids are preferably acyl groups derived from fatty acids having C10-C24 carbon chains, e.g., lauroyl, myristoyl, paimitoyl, stearoyl, or oleoyl.
- Additional exemplary lipids include, without limitation, those described in Kim et al. (2020) dx.doi.org/10.1021/acs.nanolett.0c01386, incorporated herein by reference.
- Such lipids include, in some embodiments, plant lipids found to improve liver transfection with mRNA (e.g., DGTS).
- non-cationic lipids suitable for use in the lipid nanoparticles include, without limitation, nonphosphorous lipids such as, e.g., stearylamine, dodeeylamine, hexadecylamine, acetyl palmitate, glycerol ricinoleate, hexadecyl stereate, isopropyl myristate, amphoteric acrylic polymers, triethanolamine-lauryl sulfate, alkyl-aryl sulfate polyethyloxylated fatty acid amides, dioctadecyl dimethyl ammonium bromide, ceramide, sphingomyelin, and the like.
- non-cationic lipids are described in WO2017/099823 or US patent publication US2018/0028664, the contents of which is incorporated herein by reference in their entirety.
- the non-cationic lipid is oleic acid or a compound of Formula I, II, or IV of US2018/0028664, incorporated herein by reference in its entirety.
- the non-cationic lipid can comprise, for example, 0-30% (mol) of the total lipid present in the lipid nanoparticle.
- the noncationic lipid content is 5-20% (mol) or 10-15% (mol) of the total lipid present in the lipid nanoparticle.
- the molar ratio of ionizable lipid to the neutral lipid ranges from about 2:1 to about 8:1 (e.g., about 2:1 , 3:1 , 4:1 , 5:1 , 6:1 , 7:1 , or 8:1 ).
- the lipid nanoparticles do not comprise any phospholipids.
- the lipid nanoparticle can further comprise a component, such as a sterol, to provide membrane integrity.
- a component such as a sterol
- One exemplary sterol that can be used in the lipid nanoparticle is cholesterol and derivatives thereof.
- Non-limiting examples of cholesterol derivatives include polar analogues such as 5a-choiestanol, 53-coprostanol, choiesteryl-(2,-hydroxy)-ethyl ether, choiesteryl-(4'- hydroxy)-butyl ether, and 6-ketocholestanol; non-polar analogues such as 5a-cholestane, cholestenone, 5a-cholestanone, 5p- cholestanone, and cholesteryl decanoate; and mixtures thereof.
- the cholesterol derivative is a polar analogue, e.g., choiesteryl-(4 '-hydroxy)-buty1 ether.
- Exemplary cholesterol derivatives are described in PCT publication W02009/127060 and US patent publication US2010/0130588, each of which is incorporated herein by reference in its entirety.
- the component providing membrane integrity such as a sterol
- such a component is 20-50% (mol) 30-40% (mol) of the total lipid content of the lipid nanoparticle.
- the lipid nanoparticle can comprise a polyethylene glycol (PEG) or a conjugated lipid molecule. Generally, these are used to inhibit aggregation of lipid nanoparticles and/or provide steric stabilization.
- PEG polyethylene glycol
- exemplary conjugated lipids include, but are not limited to, PEG-lipid conjugates, polyoxazoline (POZ)-lipid conjugates, polyamide-lipid conjugates (such as ATTA-lipid conjugates), cationic-polymer lipid (CPL) conjugates, and mixtures thereof.
- the conjugated lipid molecule is a PEG-lipid conjugate, for example, a (methoxy polyethylene glycol)- conjugated lipid.
- PEG-lipid conjugates include, but are not limited to, PEG-diacylglycerol (DAG) (such as l-(monomethoxy-polyethyleneglycol)-2,3-dimyristoylglycerol (PEG-DMG)), PEG-dialkyloxypropyl (DAA), PEG-phospholipid, PEG-ceramide (Cer), a pegylated phosphatidylethanoloamine (PEG-PE), PEG succinate diacylglycerol (PEGS-DAG) (such as 4-0-(2',3'-di(tetradecanoyloxy)propyl-l-0-(w- methoxy(polyethoxy)ethyl) butanedioate (PEG-S-DMG)), PEG dialkoxypropylcarbam, N-(carbonyl- methoxypolyethylene glycol 2000)-l,2-distearoyl-sn-glycero
- a PEG-lipid is a compound of Formula III, lll-a-l, lll-a-2, lll-b-1 , lll-b-2, or V of US2018/0028664, the content of which is incorporated herein by reference in its entirety.
- a PEG-lipid is of Formula II of US20150376115 or US2016/0376224, the content of both of which is incorporated herein by reference in its entirety.
- the PEG-DAA conjugate can be, for example, PEG-dilauryloxypropyl, PEG- dimyristyloxypropyl, PEG-dipalmityloxypropyl, or PEG-distearyloxypropyl.
- the PEG-lipid can be one or more of PEG-DMG, PEG-dilaurylglycerol, PEG-dipalmitoylglycerol, PEG- disterylglycerol, PEG- dilaurylglycamide, PEG-dimyristylglycamide, PEG- dipalmitoylglycamide, PEG-disterylglycamide, PEG- cholesterol (l-[8'-(Cholest-5-en-3[beta]- oxy)carboxamido-3',6'-dioxaoctanyl] carbamoyl-[omega]-methyl- poly(ethylene glycol), PEG- DMB (3,4-Ditetradecoxylbenzyl- [omega]-methyl-poly(ethylene glycol) ether), and 1 ,2- dimyristoyl-sn-glycero-3-phosphoethanolamine-N-[methoxy(polyethylene glycol)-
- the PEG-lipid comprises PEG-DMG, 1 ,2- dimyristoyl-sn-glycero-3-phosphoethanolamine-N- [methoxy(polyethylene glycol)-2000]. In some embodiments, the PEG-lipid comprises a structure selected from:
- lipids conjugated with a molecule other than a PEG can also be used in place of PEG-lipid.
- PEG-lipid conjugates polyoxazoline (POZ)-lipid conjugates, polyamide-lipid conjugates (such as ATTA-lipid conjugates), and cationic-polymer lipid (GPL) conjugates can be used in place of or in addition to the PEG-lipid.
- POZ polyoxazoline
- GPL cationic-polymer lipid
- conjugated lipids i.e. , PEG-lipids, (POZ)-lipid conjugates, ATTA-lipid conjugates and cationic polymer-lipids are described in the PCT and LIS patent applications listed in Table 2 of WO2019051289A9, the contents of all of which are incorporated herein by reference in their entirety.
- the PEG or the conjugated lipid can comprise 0-20% (mol) of the total lipid present in the lipid nanoparticle. In some embodiments, PEG or the conjugated lipid content is 0.5- 10% or 2-5% (mol) of the total lipid present in the lipid nanoparticle. Molar ratios of the ionizable lipid, non-cationic- lipid, sterol, and PEG/conjugated lipid can be varied as needed.
- the lipid particle can comprise 30-70% ionizable lipid by mole or by total weight of the composition, 0-60% cholesterol by mole or by total weight of the composition, 0-30% non-cationic-lipid by mole or by total weight of the composition and 1 -10% conjugated lipid by mole or by total weight of the composition.
- the composition comprises 30-40% ionizable lipid by mole or by total weight of the composition, 40-50% cholesterol by mole or by total weight of the composition, and 10- 20% non-cationic-lipid by mole or by total weight of the composition.
- the composition is 50-75% ionizable lipid by mole or by total weight of the composition, 20-40% cholesterol by mole or by total weight of the composition, and 5 to 10% non-cationic-lipid, by mole or by total weight of the composition and 1 -10% conjugated lipid by mole or by total weight of the composition.
- the composition may contain 60-70% ionizable lipid by mole or by total weight of the composition, 25-35% cholesterol by mole or by total weight of the composition, and 5-10% non-cationic-lipid by mole or by total weight of the composition.
- the composition may also contain up to 90% ionizable lipid by mole or by total weight of the composition and 2 to 15% non-cationic lipid by mole or by total weight of the composition.
- the formulation may also be a lipid nanoparticle formulation, for example comprising 8-30% ionizable lipid by mole or by total weight of the composition, 5-30% noncationic lipid by mole or by total weight of the composition, and 0-20% cholesterol by mole or by total weight of the composition; 4-25% ionizable lipid by mole or by total weight of the composition, 4-25% noncationic lipid by mole or by total weight of the composition, 2 to 25% cholesterol by mole or by total weight of the composition, 10 to 35% conjugate lipid by mole or by total weight of the composition, and 5% cholesterol by mole or by total weight of the composition; or 2-30% ionizable lipid by mole or by total weight of the composition, 2-30% non-cationic lipid by mole or by total weight of the composition, 1 to
- the lipid particle formulation comprises ionizable lipid, phospholipid, cholesterol and a PEG-ylated lipid in a molar ratio of 50: 10:38.5: 1 .5. In some other embodiments, the lipid particle formulation comprises ionizable lipid, cholesterol and a PEG-ylated lipid in a molar ratio of 60:38.5:1.5.
- the lipid particle comprises ionizable lipid, non-cationic lipid (e.g. phospholipid), a sterol (e.g., cholesterol) and a PEG-ylated lipid, where the molar ratio of lipids ranges from 20 to 70 mole percent for the ionizable lipid, with a target of 40-60, the mole percent of non-cationic lipid ranges from 0 to 30, with a target of 0 to 15, the mole percent of sterol ranges from 20 to 70, with a target of 30 to 50, and the mole percent of PEG-ylated lipid ranges from 1 to 6, with a target of 2 to 5.
- non-cationic lipid e.g. phospholipid
- a sterol e.g., cholesterol
- PEG-ylated lipid e.g., PEG-ylated lipid
- the lipid particle comprises ionizable lipid I non-cationic- lipid I sterol I conjugated lipid at a molar ratio of 50:10:38.5:1 .5.
- the disclosure provides a lipid nanoparticle formulation comprising phospholipids, lecithin, phosphatidylcholine and phosphatidylethanolamine.
- one or more additional compounds can also be included. Those compounds can be administered separately, or the additional compounds can be included in the lipid nanoparticles of the invention.
- the lipid nanoparticles can contain other compounds in addition to the nucleic acid or at least a second nucleic acid, different than the first.
- other additional compounds can be selected from the group consisting of small or large organic or inorganic molecules, monosaccharides, disaccharides, trisaccharides, oligosaccharides, polysaccharides, peptides, proteins, peptide analogs and derivatives thereof, peptidomimetics, nucleic acids, nucleic acid analogs and derivatives, an extract made from biological materials, or any combinations thereof.
- LNPs are directed to specific tissues by the addition of LNP targeting domains.
- biological ligands may be displayed on the surface of LNPs to enhance interaction with cells displaying cognate receptors, thus driving association with and cargo delivery to tissues wherein cells express the receptor.
- the biological ligand may be a ligand that drives delivery to the liver, e.g., LNPs that display GalNAc result in delivery of nucleic acid cargo to hepatocytes that display asialoglycoprotein receptor (ASGPR).
- ASGPR asialoglycoprotein receptor
- Mol Ther 18(7):1357-1364 (2010) teaches the conjugation of a trivalent GalNAc ligand to a PEG-lipid (GalNAc-PEG-DSG) to yield LNPs dependent on ASGPR for observable LNP cargo effect (see, e.g., FIG. 6 of Akinc et al. 2010, supra).
- Other ligand-displaying LNP formulations e.g., incorporating folate, transferrin, or antibodies, are discussed in WO2017223135, which is incorporated herein by reference in its entirety, in addition to the references used therein, namely Kolhatkar et al., Curr Drug Discov Technol. 2011 8:197-206; Musacchio and Torchilin, Front Biosci.
- LNPs are selected for tissue-specific activity by the addition of a Selective ORgan Targeting (SORT) molecule to a formulation comprising traditional components, such as ionizable cationic lipids, amphipathic phospholipids, cholesterol and polyethylene glycol) (PEG) lipids.
- SORT Selective ORgan Targeting
- traditional components such as ionizable cationic lipids, amphipathic phospholipids, cholesterol and polyethylene glycol) (PEG) lipids.
- PEG polyethylene glycol
- the LNPs comprise biodegradable, ionizable lipids.
- the LNPs comprise (9Z,l2Z)-3-((4,4-bis(octyloxy)butanoyl)oxy)-2-((((3- (diethylamino)propoxy)carbonyl)oxy)methyl)propyl octadeca-9,l2-dienoate, also called 3- ((4,4- bis(octyloxy)butanoyl)oxy)-2-(((3-(diethylamino)propoxy)carbonyl)oxy)methyl)propyl (9Z,l2Z)-octadeca- 9,12-dienoate) or another ionizable lipid.
- lipids of WO2019/067992, WO/2017/173054, WO2015/095340, and WO2014/136086 as well as references provided therein.
- the term cationic and ionizable in the context of LNP lipids is interchangeable, e.g., wherein ionizable lipids are cationic depending on the pH.
- the average LNP diameter of the LNP formulation may be between 10s of nm and 100s of nm, e.g., measured by dynamic light scattering (DLS). In some embodiments, the average LNP diameter of the LNP formulation may be from about 40 nm to about 150 nm, such as about 40 nm, 45 nm, 50 nm, 55 nm, 60 nm, 65 nm, 70 nm, 75 nm, 80 nm, 85 nm, 90 nm, 95 nm, 100 nm, 105 nm, 110 nm, 115 nm, 120 nm, 125 nm, 130 nm, 135 nm, 140 nm, 145 nm, or 150 nm.
- DLS dynamic light scattering
- the average LNP diameter of the LNP formulation may be from about 50 nm to about 100 nm, from about 50 nm to about 90 nm, from about 50 nm to about 80 nm, from about 50 nm to about 70 nm, from about 50 nm to about 60 nm, from about 60 nm to about 100 nm, from about 60 nm to about 90 nm, from about 60 nm to about 80 nm, from about 60 nm to about 70 nm, from about 70 nm to about 100 nm, from about 70 nm to about 90 nm, from about 70 nm to about 80 nm, from about 80 nm to about 100 nm, from about 80 nm to about 90 nm, or from about 90 nm to about 100 nm.
- the average LNP diameter of the LNP formulation may be from about 70 nm to about 100 nm. In a particular embodiment, the average LNP diameter of the LNP formulation may be about 80 nm. In some embodiments, the average LNP diameter of the LNP formulation may be about 100 nm. In some embodiments, the average LNP diameter of the LNP formulation ranges from about I mm to about 500 mm, from about 5 mm to about 200 mm, from about 10 mm to about 100 mm, from about 20 mm to about 80 mm, from about 25 mm to about 60 mm, from about 30 mm to about 55 mm, from about 35 mm to about 50 mm, or from about 38 mm to about 42 mm.
- a LNP may, in some instances, be relatively homogenous.
- a polydispersity index may be used to indicate the homogeneity of a LNP, e.g., the particle size distribution of the lipid nanoparticles.
- a small (e.g., less than 0.3) polydispersity index generally indicates a narrow particle size distribution.
- a LNP may have a polydispersity index from about 0 to about 0.25, such as 0.01 , 0.02, 0.03, 0.04, 0.05, 0.06, 0.07, 0.08, 0.09, 0.10, 0.11 , 0.12, 0.13, 0.14, 0.15, 0.16, 0.17, 0.18, 0.19, 0.20, 0.21 , 0.22, 0.23, 0.24, or 0.25.
- the polydispersity index of a LNP may be from about 0.10 to about 0.20.
- the zeta potential of a LNP may be used to indicate the electrokinetic potential of the composition.
- the zeta potential may describe the surface charge of an LNP. Lipid nanoparticles with relatively low charges, positive or negative, are generally desirable, as more highly charged species may interact undesirably with cells, tissues, and other elements in the body.
- the zeta potential of a LNP may be from about -10 mV to about +20 mV, from about -10 mV to about +15 mV, from about -10 mV to about +10 mV, from about -10 mV to about +5 mV, from about -10 mV to about 0 mV, from about -10 mV to about -5 mV, from about -5 mV to about +20 mV, from about -5 mV to about +15 mV, from about -5 mV to about +10 mV, from about -5 mV to about +5 mV, from about -5 mV to about 0 mV, from about 0 mV to about +20 mV, from about 0 mV to about +15 mV, from about 0 mV to about +10 mV, from about 0 mV to about +5 mV, from about 0 mV to about +20 mV, from
- the efficiency of encapsulation of a protein and/or nucleic acid describes the amount of protein and/or nucleic acid that is encapsulated or otherwise associated with a LNP after preparation, relative to the initial amount provided.
- the encapsulation efficiency is desirably high (e.g., close to 100%).
- the encapsulation efficiency may be measured, for example, by comparing the amount of protein or nucleic acid in a solution containing the lipid nanoparticle before and after breaking up the lipid nanoparticle with one or more organic solvents or detergents.
- An anion exchange resin may be used to measure the amount of free protein or nucleic acid (e.g., RNA) in a solution.
- Fluorescence may be used to measure the amount of free protein and/or nucleic acid (e.g., RNA) in a solution.
- the encapsulation efficiency of a protein and/or nucleic acid may be at least 50%, for example 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or 100%.
- the encapsulation efficiency may be at least 80%.
- the encapsulation efficiency may be at least 90%.
- the encapsulation efficiency may be at least 95%.
- a LNP may optionally comprise one or more coatings.
- a LNP may be formulated in a capsule, film, or table having a coating.
- a capsule, film, or tablet including a composition described herein may have any useful size, tensile strength, hardness or density.
- in vitro or ex vivo cell lipofections are performed using Lipofectamine MessengerMax (Thermo Fisher) or TransIT-mRNA Transfection Reagent (Mirus Bio).
- LNPs are formulated using the GenVoyJLM ionizable lipid mix (Precision NanoSystems).
- LNPs are formulated using 2,2-dilinoleyl-4-dimethylaminoethyl-[1 ,3]-dioxolane (DLin- KC2-DMA) or dilinoleylmethyl-4-dimethylaminobutyrate (DLin-MC3-DMA or MC3), the formulation and in vivo use of which are taught in Jayaraman et al. Angew Chem Int Ed Engl 51 (34):8529-8533 (2012), incorporated herein by reference in its entirety.
- DLin- KC2-DMA 2,2-dilinoleyl-4-dimethylaminoethyl-[1 ,3]-dioxolane
- DLin-MC3-DMA or MC3 dilinoleylmethyl-4-dimethylaminobutyrate
- LNP formulations optimized for the delivery of CRISPR-Cas systems e.g., Cas9-gRNA RNP, gRNA, Cas9 mRNA, are described in WO2019067992 and WO2019067910, both incorporated by reference.
- Exemplary dosing of LNPs comprising the RNA compositions described herein may include about 0.1 , 0.25, 0.3, 0.5, 1 , 2, 3, 4, 5, 6, 8, 10, or 100 mg/kg (RNA).
- Exemplary dosing of AAV comprising a nucleic acid encoding one or more components of the system may include an MOI of about 1011 , 1012, 1013, and 1014 vg/kg.
- the invention includes a lipid nanoparticle (LNP) comprising the ANDbody polypeptide (or RNA encoding the same), nucleic acid molecule, or DNA encoding an ANDbody described herein.
- LNP comprises a cationic lipid.
- the LNP further comprises one or more neutral lipid, e.g., DSPC, DPPC, DMPC, DOPC, POPC, DOPE, SM, a steroid, e.g., cholesterol, and/or one or more polymer conjugated lipid, e.g., a pegylated lipid, e.g., PEG-DAG, PEG-PE, PEG-S-DAG, PEG-cer or a PEG dialkyoxypropylcarbamate.
- the cationic lipid of the LNP has a structure according to:
- compositions described herein can be delivered by a viral vector (e.g., a viral vector expressing an RNA).
- a viral vector e.g., a viral vector expressing an RNA.
- a viral vector may be administered to a cell or to a subject (e.g., a human subject or non-human animal).
- a viral vector may be locally or systemically administered.
- viral vectors examples include a retrovirus (e.g., Retroviridae family viral vector), adenovirus (e.g., Ad5, Ad26, Ad34, Ad35, and Ad48), parvovirus (e.g., adeno-associated viruses), coronavirus, negative strand RNA viruses such as orthomyxovirus (e.g., influenza virus), rhabdovirus (e.g., rabies and vesicular stomatitis virus), paramyxovirus (e.g., measles and Sendai), positive strand RNA viruses, such as picornavirus and alphavirus, and double stranded DNA viruses including adenovirus, herpesvirus (e.g., Herpes Simplex virus types 1 and 2, Epstein-Barr virus, cytomegalovirus, replication deficient herpes virus), and poxvirus (e.g., vaccinia, modified vaccinia Ankara (MVA), fowlpox and canary
- viruses include Norwalk virus, togavirus, flavivirus, reoviruses, papovavirus, hepadnavirus, human papilloma virus, human foamy virus, and hepatitis virus, for example.
- retroviruses include: avian leukosis-sarcoma, avian C-type viruses, mammalian C-type, B-type viruses, D-type viruses, oncoretroviruses, HTLV-BLV group, lentivirus, alpharetrovirus, gammaretrovirus, spumavirus (Coffin, J. M., Retroviridae: The viruses and their replication, Virology (Third Edition) Lippincott-Raven, Philadelphia, 1996).
- murine leukemia viruses include murine leukemia viruses, murine sarcoma viruses, mouse mammary tumor virus, bovine leukemia virus, feline leukemia virus, feline sarcoma virus, avian leukemia virus, human T-cell leukemia virus, baboon endogenous virus, Gibbon ape leukemia virus, Mason Pfizer monkey virus, simian immunodeficiency virus, simian sarcoma virus, Rous sarcoma virus and lentiviruses.
- vectors are described, for example, in US Patent No. 5,801 ,030, the teachings of which are incorporated herein by reference.
- Anellovirus vectors can also be used for delivering an ANDbody composition described herein.
- Anellovectors are known in the art and described, e.g., in W02020123773, WO2020123816, WO2018232017, and W02020123773.
- an anellovector composition comprises a genomic element that comprises a promoter operably linked to a nucleic acid sequence encoding an ANDbody described herein, the genetic element encapsulated by a proteinaceous exterior comprising an Anellovirus ORF1 , e.g., an anellovirus capsid protein.
- Cell and vesicle-based carriers e.g., an anellovirus capsid protein.
- compositions described herein can be administered to a cell in a cell, vesicle or other membrane-based carrier.
- the compositions and systems described herein can be formulated in liposomes or other similar vesicles.
- Liposomes are spherical vesicle structures composed of a uni- or multilamellar lipid bilayer surrounding internal aqueous compartments and a relatively impermeable outer lipophilic phospholipid bilayer. Liposomes may be anionic, neutral or cationic.
- Liposomes are biocompatible, nontoxic, can deliver both hydrophilic and lipophilic drug molecules, protect their cargo from degradation by plasma enzymes, and transport their load across biological membranes and the blood brain barrier (BBB) (see, e.g., Spuch and Navarro, Journal of Drug Delivery, vol. 2011 , Article ID 469679, 12 pages, 2011 . doi:10.1155/2011/469679 for review).
- Vesicles can be made from several different types of lipids; however, phospholipids are most commonly used to generate liposomes as drug carriers. Methods for preparation of multilamellar vesicle lipids are known in the art (see for example U.S. Pat. No.
- vesicle formation can be spontaneous when a lipid film is mixed with an aqueous solution, it can also be expedited by applying force in the form of shaking by using a homogenizer, sonicator, or an extrusion apparatus (see, e.g., Spuch and Navarro, Journal of Drug Delivery, vol. 2011 , Article ID 469679, 12 pages, 2011 . doi :10.1155/2011/469679 for review).
- Extruded lipids can be prepared by extruding through filters of decreasing size, as described in Templeton et al., Nature Biotech, 15:647-652, 1997, the teachings of which relating to extruded lipid preparation are incorporated herein by reference.
- Exosomes can also be used as drug delivery vehicles for the compositions and systems described herein.
- Exosomes can also be used as drug delivery vehicles for the compositions and systems described herein.
- Ex vivo differentiated red blood cells can also be used as a carrier for an agent (e.g., an inhibitor) described herein, e.g., an antibody or a nucleic acid described herein.
- an agent e.g., an inhibitor
- Fusosome compositions e.g., as described in WO2018208728, can also be used as carriers to deliver the agent or preparation described herein.
- Plant nanovesicles and plant messenger packs e.g., as described in WO2011097480, WO2013070324, WO2017004526, or W02020041784 can also be used as carriers to deliver the compositions described herein.
- RNA- sequencing data from various public and proprietary data sources were summarized to determine expression for each gene across each of 32 tissues using the mean expression as measured by Transcripts Per Million (TPM).
- TPM Transcripts Per Million
- Specificity scores were calculated using four different statistical methods that capture the specificity of expression for each gene in each tissue or tissue combination (e.g., grouped tissues from the lower gastrointestinal (Gl) tract). The average rank of a gene was calculated as the mean rank across the four different methods. The genes were ranked according to specificity score for each tissue.
- top addresses For each tissue, a list of the top (up to) 25 addresses that (i) met a set threshold for minimum specificity score and minimum expression level in the tissue and (ii) were annotated as containing a transmembrane domain or being otherwise membrane-associated was generated (“top addresses;” Table 1 ).
- Table 1 shows proteins with a specificity score greater than 0.7 in lower Gl, kidney, liver, pancreas, and skin.
- the model considered a biotherapeutic that binds to two target cell surface receptors on a single cell type acting as a receptor agonist for IL-22.
- the molecule can be monovalent or bivalent with avidity for each target.
- the percent of receptor occupied by the cytokine was used as a metric for signaling activity.
- the model parameterizes various drug properties (binding affinity, half-life, dosing mode and frequency, valency, avidity, etc.) and biophysical characteristics (cytokine concentration in various compartments, receptor numbers, receptor turnover rate, etc.) and predicts how drug agonism depends on these parameters.
- Pharmacokinetic/pharmacodynamic (PK/PD) modeling is performed to evaluate the therapeutic profile of localized IL-22 in IBD to identify and explain the relationships between drug exposure, safety, and efficacy and the differences among population subgroups.
- a nonlinear mixed effect model is used to characterize the relationships between dose, exposure, and biomarker and/or clinical endpoint response over time, variability between subjects (e.g., mice) and groups, residual variability and uncertainty, and predict minimum anticipated biological effect level (MABEL) dose for the selected biotherapeutic.
- MABEL minimum anticipated biological effect level
- Example 4 Design, expression, and binding activity of ANDbodies comprising IL-22/antibody fusion proteins
- the first format comprises a heterodimeric molecule comprising two arms.
- the first arm comprises a standard antibody heavy chain and light chain, where the Fc region of the antibody incorporates substitutions for heterodimeric pairing (e.g., as in SEQ ID NO: 4).
- the second arm comprises an Fc region with an IL-22 polypeptide fused in place of the antibody Fab region (e.g., as in SEQ ID NO: 9).
- the IL-22 polypeptide and Fc region are linked by a (G4S)2 linker (SEQ ID NO: 5).
- a schematic diagram of the monovalent IL- 22/mAb format is shown in Fig. 1 A.
- the second format referred to herein as “divalent IL-22/mAb,” comprises a standard antibody molecule with an IL-22 polypeptide fused to the C-terminus of both Fc domains. These polypeptides are connected by a (G4S)4 linker (SEQ ID NO: 6).
- a schematic diagram of the divalent IL-22/mAb format is shown in Fig. 1 B.
- any antibody variable domains of the light chain and heavy chain combination described herein can be used in the antibody (e.g., a heavy chain as presented in SEQ ID NO: 1 and/or a light chain as presented in SEQ ID NO: 2).
- This example provides antibodies in which the CHi and Fc region are human IgG 1 (SEQ ID NO: 3).
- the exemplary monovalent IL-22/mAb provided herein includes additional substitutions in the Fc region that favor heterodimeric pairing (Moore et al., Methods, 154: 38-50, 2019) and selective purification of the desired heterodimeric molecule over either homodimeric product (see, e.g., US Patent No. 8,586,713 B2), such as in SEQ ID NOs: 4 and 9. Alternative Fc sequences may also be utilized. Table 2. Protein Sequences B. Methods of production
- EXPI293FTM cells were transiently transfected with a 2:3 mass ratio of heavy chain: light chain plasmid using polyethylenimine (PEI) and maintained according to manufacturer’s instructions (37°C, 8% CO2 on a shaking platform). Cultures were fed 4-24 hours post-transfection to a final concentration of 5% v/v GIBCOTM EFFICIENTFEEDTM B, 1% v/v L-alanyl-glutamine, and 4 mM valproic acid. After 4-7 days, supernatants were harvested by centrifugation at 4000 x g followed by 0.2 pm filtration.
- PEI polyethylenimine
- Filtered supernatants were purified via Protein A affinity chromatography (Cytiva 5 mL MABSELECTTM SuRe column) in PBS 7.4 running buffer and 0.1 M glycine pH 2.7 elution buffer. Eluted protein was immediately neutralized with 10% v/v of 1 M sodium acetate, pH 6.0. Protein was buffer exchanged into 50 mM sodium acetate + 60 mM sucrose, pH 6.0 for storage.
- Monovalent IL-22 fusion proteins (e.g., monovalent IL-22/mAbs) were produced by transient PEI transfection of a 1 :1 :2 mass ratio of heavy chain : light chain : IL-22-Fc plasmid.
- the expression host, culture conditions, and purification protocol were identical to those described above for the divalent ANDbody.
- the IL-22-containing molecule comprised mutations in the Fc domain such that incorrectly homodimerized IL-22 molecules do not bind efficiently to Protein A.
- An alternative approach that can also be used is Fab arm exchange (Steinhardt et al, Pharmaceutics, 12(3), 2019). This uses different substitutions in the Fc region, but produces functionally identical proteins.
- Binding of each molecule to its antibody target and to human IL-22RA1 was evaluated using biolayer interferometry (BLI) on a GATOR® instrument at ambient temperature ( ⁇ 22°C).
- BLI biolayer interferometry
- the fusion protein was loaded on GATOR® biosensor probes coated with anti-human Fc antibody, and binding was evaluated by dipping into a solution containing the indicated target.
- Mixing speed was 1000 rpm.
- the antigens used in these Examples include ectodomains of hlL22RA1 (R&D Systems #2770-LR-050), mlL22RA1 (R&D Systems #4294-MR-050), mCDH17 (NCBI NP_062727, purified in-house), hlL22BP (Sino Biological US, 11025-H08H) and RSV glycoprotein F (Sino Biological US, 11049-V08B).
- Table 3 shows the binding affinity of each molecule for its two targets (murine targets are denoted with “m” before the gene name; human targets are denoted with “h”), as measured using BLI.
- Example 5 Signaling activity of both monovalent and divalent IL-22/antibody fusion proteins targeting mCDH17 is enhanced on cells that express mCDH17
- each IL-22 fusion protein provided in Example 4 was evaluated using a commercial reporter cell line (HEK-BLUETM IL-22 cells (InvivoGen)). These reporter cells produce secreted embryonic alkaline phosphatase (SEAP) in response to activation of IL-22 signaling.
- the reporter cells were modified to stably express murine CDH17 (NCBI NP_062727) (i.e., were transduced using lentiviral vectors followed by cell sorting to generate stable pools to produce cell strains with stable expression of murine CDH17).
- Table 4 shows the response to each fusion protein of parental reporter cells (“reporter cell”) and modified reporter cells expressing mCDH17 (“+mCDH17”).
- An antibody against respiratory syncytial virus (RSV) glycoprotein F was included as a negative control.
- Example 6 Monovalent and divalent IL-22/antibody fusion proteins targeting mCDH16 or mGP2 and signaling activity thereof
- Monovalent and divalent format IL-22 fusion proteins (monovalent IL-22/mAb and divalent IL- 22/mAb) as described in Example 4 are prepared using an antibody against the address target mCDH16 (NCBI NP_031689), which is expressed in kidney tubules.
- HEK-BLUETM IL-22 reporter cells expressing mCDH16 are prepared as described in Example 5.
- An assay as described in Example 5 is performed to evaluate address-dependent enhancement of signaling potency for the targeted IL-22 molecules (monovalent anti-mCDH16/IL-22 and divalent anti- mCDH16/IL-22) in reporter cells expressing the mCDH16 address target as compared to matched nontargeted molecules, differently targeted molecules, and reporter cells not comprising the address. Both formats are expected to show significant enhanced potency when address expression matches the antibody target, indicating that address-dependent potentiation is a general feature of the antibody/IL-22 fusion proteins.
- Monovalent and divalent format IL-22 fusion proteins (monovalent IL-22/mAb and divalent IL- 22/mAb) as described in Example 4 are prepared using an antibody against the address target mGP2 (NCBI NP_080265), which is expressed in pancreatic acinar cells.
- HEK-BLUETM IL-22 reporter cells expressing mGP2 are prepared as described in Example 5.
- An assay as described in Example 5 is performed to evaluate address-dependent enhancement of signaling potency for the targeted IL-22 molecules (monovalent anti-mGP2/IL-22 and divalent anti-mGP2/IL-22) in reporter cells expressing the mGP2 address target as compared to matched non-targeted molecules, differently targeted molecules, and reporter cells not comprising the address.
- the monovalent and the divalent IL-22 fusion protein formats are designed to elicit significantly enhanced potency in reporter cells that express an address that is targeted by the antibody. For example, increased potency is expected for the IL-22/mCDH16 antibody fusion proteins in reporter cells expressing mCDH16, and for the IL-22/mGP2 antibody fusion proteins in reporter cells expressing mGP2, thus showing that address-dependent potentiation is a general feature of the antibody/IL-22 fusion proteins.
- Example 7 Rationally engineered substitutions at the IL-22/IL-22RA1 interface reduce receptor binding and signaling activity of IL-22/antibody fusion proteins
- a structural modeling approach was used to predict residues and specific amino acid substitutions in IL-22 (SEQ ID NO: 8) that may reduce receptor binding affinity while maintaining structural stability of the molecule.
- the numbering of amino acid substitutions used herein is based on the sequence of the IL-22 precursor, including its signal peptide (SEQ ID NO: 7).
- the IL22 interface residues (Table 5) were identified as the residues located at most 5 angstrom away from a residue on the receptor (interface distance less than 5 A).
- T70A and E77Q did not express. Other results are summarized in Tables 7A and 7B, below. “Low” changes in binding affinity are less than 10-fold, “moderate” changes are 10-100-fold, and “high” changes are >1 OO-fold.
- Table 8 lists key IL22 interface residues that were identified and specific substitutions that were predicted to be useful for this purpose based on the previously described computational and initial screening results. Table 8. Key IL22 interface residues predicted to be useful for detuning
- IL-22/Fc fusion proteins in which each molecule contained two IL-22 moieties located at the N-terminal end of a human IgG 1 Fc. These were purified by protein A affinity, and the binding of each molecule to human and mouse IL22RA1 proteins was evaluated by biolayer interferometry. Results are shown in Table 9A.
- D71 L and a subset of additional substitutions of Table 8A were introduced into the IL-22 moiety at the C-terminus of the divalent IL-22/mAb provided in Example 2 (D71 L (SEQ ID NO: 11 ), R73Q (SEQ ID NO: 12), and D71 L/D168G (SEQ ID NO: 13)), wherein the mAb was an anti-mCDH17 antibody, and evaluated binding to human and murine IL22RA1 by BLI as in Example 4.
- Table 9B shows the measured affinity of the divalent IL-22/mAb as for human and murine IL22RA1 as measured by BLI. Table 9B. Effect of IL-22 substitutions on affinity for IL22RA1 in the divalent format measured by biolayer interferometry
- Table 9C shows the measured affinity of the divalent IL-22/mAb as for human IL22BP as measured by BLI.
- Example 8 Detuning substitutions that weaken binding affinity of IL-22 for IL-22RA1 increase the potentiation effect of targeting antibodies in in vitro signaling assays
- Example 7 The effect of detuning IL-22 as described in Example 7 was evaluated by comparing activation of IL-22 signaling by the divalent IL-22/mAb comprising the detuning substitutions in wild-type IL-22 reporter cells and IL-22 reporter cells expressing mCDH17, using assays as in Example 5.
- Table 10 shows the ECso for each molecule tested. In each case, weakening affinity for IL22RA1 increased the EC50 of the molecule compared to wild-type control in the absence of mCDH17. Thus, as hypothesized, molecules with weakened affinity demonstrated reduced signaling potency (e.g. higher EC50) on reporter cells that do not express mCDH17.
- Address-targeted IL-22 molecules are expected to bind to their target cells when injected into mice, and such binding may manifest as increased accumulation of the molecule in a target tissue compared to non-targeted control. It may also manifest as more rapid depletion from circulation in blood.
- a monovalent IL-22/mAb containing an anti-mCDH17 targeting arm (monovalent anti- mCDH17/IL-22) and a matched molecule containing an anti-RSV glycoprotein F antibody (monovalent anti-RSV/IL-22) were introduced into BALB/c mice by tail vein injection at 3 mg/kg. Three groups of three mice were used for each molecule. Blood samples were collected at 1 , 3, 6, 24, 48, and 96 hours. One group of mice for each molecule was euthanized at 24, 48 and 96 hours and tissues were collected to measure distribution of the molecule (small intestine, colon, liver, lung, skin).
- the concentration of each molecule in blood samples was measured by sandwich ELISA using anti-human Fc antibody to capture and anti-human light chain antibody for detection. Tissue samples were homogenized, total protein measured, and similar ELISA used to measure the amount of each fusion protein in 500 mg of extracted tissue lysate.
- Fig. 2 shows the level of each molecule in circulation over time.
- the monovalent anti- mCDH17/IL-22 molecule mostly cleared from circulation within 48 hours, while the non-targeted molecule persisted longer in circulation. Both molecules persist in circulation similarly to their respective parental monoclonal antibodies.
- Figs. 3A and 3B show the level of each molecule in small intestine, colon, lung, liver, and skin tissue at 24, 48 and 96 hours. There was a markedly higher level of the targeted molecule (monovalent anti-mCDH17/IL-22) than the non-targeted molecule (monovalent anti-RSV/IL-22) in small intestine, and the targeted molecule persisted in the colon despite clearing rapidly from circulation. The opposite was observed in off-target tissues, consistent with the higher level of the non-targeted molecule that persisted in circulation. These data show that the monovalent anti-mCDH17/IL-22 accumulates in the target tissues of small intestine and colon.
- Example 10 Monovalent IL-22/antibody fusion protein targeting gut epithelial cells has enhanced on-target dose response in mice compared to non-targeted molecule
- mice were treated with doses of mCDH17-targeted and non-targeted monovalent IL-22 molecules (monovalent anti-mCDH17/IL-22 and monovalent anti-RSV/IL-22, respectively) as described in Example 9 at doses of 1 mg/kg, 0.3 mg/kg, and 0.1 mg/kg.
- Groups contained 5 mice each, and data were collected at 24 and 48 hours after dosing.
- On-target tissues small intestine and colon
- off- target tissues pancreas and liver
- Figs. 4A-4D show the levels of REG3B, REG3G and SOCS3 in each treated group relative to mice that were treated with phosphate-buffered saline.
- Example 11 Detuning IL-22 binding to IL-22RA1 of gut-targeted monovalent IL-22/antibody fusion protein on pharmacodynamics to increase the tissue-specificity of their pharmacodynamic effect
- Monovalent IL-22/mAb molecules as described in Example 5 comprising (i) a detuned IL-22 moiety such as those described in Examples 7-10 and (ii) an antibody targeting murine CDH17 are generated.
- the detuned IL-22 moieties are selected to span a range of affinities up to about 100x reduction in binding to IL22RA1 .
- Matched molecules comprising an antibody that binds to RSV glycoprotein F (monovalent anti-RSV/IL-22) are produced as a negative control for address targeting.
- the molecules are introduced into mice by tail vein injection at doses ranging from 1 mg/kg to 0.1 mg/kg and on-target tissues (small intestine and colon tissues) and off-target comparator tissues (pancreas, skin, lung, and liver) are collected at timepoints over several days.
- the dose response of IL- 22 activity in each tissue is assayed by qPCR to compare levels of IL-22 target genes in treated versus untreated mice. These data are used to compare the dose level that generates a signaling response in on-target tissues as compared to each off-target tissue (thereby providing a specificity ratio).
- Molecules targeting mCDH17 and containing detuned IL-22 are expected to show a greater specificity ratio than molecules targeting mCDH17 and containing native IL-22.
- Non-targeted control molecules are expected to show less enhancement.
- Example 12 Monovalent IL-22/antibody fusion protein targeting pancreatic acinar cells that bind to acinar cells in mice and have enhanced on-target dose response compared to a non-targeted molecule
- Monovalent IL-22/mAb molecules as described in Example 4 comprising (i) a native IL-22 moiety or a detuned IL-22 moiety such as those described in Examples 7-10 and (ii) an antibody targeting murine GP2 are generated. GP2 is expressed predominantly on pancreatic acinar cells. Matched molecules comprising an antibody that binds to RSV glycoprotein F (monovalent anti-RSV/IL-22) are produced as a negative control for address targeting.
- the molecules are introduced into mice by tail vein injection at 3 mg/kg, and levels of each molecule in lysate of pancreatic tissue are assayed at timepoints over several days.
- the GP2-targeted molecules are expected to accumulate at a higher level in pancreatic tissue when compared to the nontargeted control that binds to RSV glycoprotein F.
- the molecules are then introduced into mice by tail vein injection at doses ranging from 1 mg/kg to 0.1 mg/kg and pancreatic tissue, along with off-target comparator tissues, are collected at timepoints over several days.
- the IL-22 activity in each tissue is assayed by using qPCR to compare levels of IL-22 target genes in treated as compared to untreated mice.
- the GP2-targeted molecules are expected to show an enhanced dose response in pancreas compared to the non-targeted control molecule, and a reduced dose response in the off-target tissues.
- Example 13 Monovalent IL-22/antibody fusion protein that target epithelial cells of the tubular epithelium, bind tubular epithelial cells in mice, and have enhanced on-target dose response compared to non-targeted molecule
- Monovalent IL-22/mAb molecules as described in Example 4 comprising (i) a native IL-22 moiety or a detuned IL-22 moiety such as those described in Examples 7-10 and (ii) an antibody targeting murine CDH16 are generated.
- CDH16 is expressed predominantly on epithelial cells in the kidney tubules.
- Matched molecules comprising an antibody that binds to RSV glycoprotein F (monovalent anti- RSV/IL-22) are produced as a negative control for address targeting.
- the molecules are introduced into mice by tail vein injection at 3 mg/kg, and levels of each molecule in lysate of pancreatic tissue are assayed at timepoints over several days.
- the CDH16- targeted molecules are expected to accumulate at a higher level in kidney tissue when compared to nontargeted control that binds to RSV glycoprotein F.
- the molecules are then introduced into mice by tail vein injection at doses ranging from 1 mg/kg to 0.1 mg/kg and kidney tissue, along with off-target comparator tissues, are collected at timepoints over several days.
- the IL-22 activity in each tissue is assayed by using qPCR to compare levels of IL-22 target genes in treated as compared to untreated mice.
- the CDH16-targeted molecules are expected to show an enhanced dose response in kidney compared to the non-targeted control molecule, and a reduced dose response in the off-target tissues.
- Example 14 Detuning IL-22 for IL-22RA1 enabled gut-targeting divalent IL-22/antibody fusions to have activity specifically in the small intestine and colon of mice
- the detuned divalent IL-22 molecules targeting mCDH17 provided in Examples 7 and 8 were introduced into BALB/c mice by tail vein injection at 3 mg/kg. Matched molecules targeting RSV glycoprotein F (Divalent IL-22/anti-RSV mAb) were used for comparison. Each group used nine mice. Blood samples were collected at timepoints from immediately after injection to 4 days after injection (0, 1 , 3, 6, 24, 48, and 96 hours after injection), and ELISA was used to measure the concentration of the IL-22 molecules in circulation. Three mice from each group were euthanized at 1 , 2 and 4 days, and tissues (small intestine, colon, pancreas, and liver) were collected to determine the pharmacodynamic effect of IL-22 on expression of target genes.
- Fig. 5 shows the levels of mCDH17-targeted IL-22 and RSV-targeted IL-22 fusion proteins in serum over time. All of the molecules showed a rapid distribution out of circulation in the first hour, followed by slower decline. The mCDH17-targeted molecules cleared more quickly than the RSV- targeted molecules, consistent with accumulation in the gut as observed for the monovalent molecules (Example 9). The various detuning substitutions in IL-22 had little effect on the rate of clearance.
- RT-PCR was used to determine expression of the IL-22 target genes REG3B, REG3G, and S0CS3 in small intestine, colon, pancreas, and liver at 1 , 2 and 4 days after treatment. Figs.
- Example 15 Detuned divalent IL-22 fusion protein targeting mCDH17 has normal dose response in mice without significant activity in other tissues
- mice were treated as in described in Example 10 with doses of mCDH17-targeted and nontargeted divalent detuned IL-22 molecules comprising native IL-22 or IL-22 comprising the D71 L/D168G amino acid substitution mutations described in Example 14 at dose levels of 1 mg/kg, 0.3 mg/kg, and 0.1 mg/kg. Groups contain 5 mice each, and time-points were 24 and 48 hours after dosing. On-target tissues (small intestine and colon) and off-target tissues (pancreas and liver) were processed to extract mRNA, and the levels of selected IL-22 target genes were measured by RT-PCR.
- the target genes REG3B, REG3G and SOCS3 were measured in each treated group relative to mice treated with phosphate-buffered saline.
- the genes MT1 and MT2 were used instead.
- Figs. 7A and 7B show that in the small intestine and colon, there was a strong response to native IL-22 in both targeted and non-targeted formats.
- the D71 L/D168G substitutions almost eliminated activity of the non-targeted molecules on both tissues, but the targeted molecule retained nearly native activity.
- Figs. 7C and 7D show that the native IL-22 molecules similarly have activity on pancreas and liver, with or without targeting. Activity on the pancreas is notably reduced by targeting, possibly because it localizes the molecule rapidly to gut tissues.
- activity of the IL-22 with D71 L/D168G substitutions is almost eliminated in pancreas and liver in both the targeted and non-targeted molecules.
- Example 16 Appropriately tuned divalent IL-22 fusion proteins that target pancreatic acinar cells, bind to target cells, and have enhanced on-target dose response in mice compared to nontargeted molecules
- IL-22 moieties were produced in the divalent format as fusions to an antibody targeting murine GP2 (mGP2).
- Matched molecules comprising an antibody that binds to RSV glycoprotein F were produced as a negative control for address targeting.
- the molecules were introduced into mice by tail vein injection at a range of dose levels from 0.03 to 0.3 MPK. Groups contained 5 mice each and timepoints were 24 and 96 hours after dosing. Pancreas tissue (on-target tissue) and off-target tissues (including small intestine and colon) were processed to extract mRNA, and the level of selected IL-22 target genes were measured. Key target genes REG3B and REG3G were measured in each treated group relative to mice treated with phosphate-buffered saline.
- Example 17 Appropriately tuned divalent IL-22 fusion proteins that target epithelial cells of kidney tubules, bind to target cells, and have enhanced on-target dose response in mice compared to a non-targeted molecule
- IL-22 moieties are produced in the divalent format as fusions to an antibody targeting murine CDH16 (mCDH16).
- Matched molecules comprising an antibody that binds to RSV glycoprotein F are produced as a negative control for address targeting.
- the molecules are introduced into mice by tail vein injection at a range of dose levels. Groups contain 5 mice each and time-points are 24 and 48 hours after dosing.
- On-target tissues small intestine and colon
- off-target tissues pancreas and liver
- Key target genes REG3B, REG3G and SOCS3 are measured in each treated group relative to mice treated with phosphate-buffered saline.
- Example 18 Detuning the affinity for anti-mCDH17 antibody to increase the circulating lifetime of gut-targeted monovalent IL-22/antibody fusion proteins in mice
- the native or detuned monovalent mCDH17-targeted IL-22 is modified with substitutions in the complementarity determining regions of the anti-mCDH17 antibody that change its binding affinity from ⁇ 1 nM to 1 -10 nM, 10-100 nM or >100 nM.
- the same modifications are made in detuned divalent IL-22 with anti-mCDH17 targeting.
- Anti-RSV glycoprotein F targeting is used as the negative control.
- Each molecule is introduced into mice at 3 mg/kg by tail vein injection. Each molecule has a group size of six mice. Levels in blood are measured at 0, 1 , 3, 6, 24 and 48 hours. Three mice from each group are euthanized at 24 hours, and key tissues are collected (small intestine, colon, liver, lung, kidney, pancreas, skin). The remaining three mice are euthanized at 48 hours for time same tissue collection. Levels of each IL-22 molecule are measured in blood and tissue by ELISA as above. Weakening the affinity of anti-mCDH17 is expected to increase the time in circulation for both the mono- and divalent IL-22 molecules. An intermediate affinity concentration that produces the highest local accumulation in gut tissues at 24 and 48 hours is expected. The data are expected to show that tuning anti-mCDH17 affinity to an intermediate level is necessary to provide optimal tissue-targeted distribution.
- Example 19 Detuning the affinity for anti-mCDH17 antibody to increase the duration of cellular response to IL-22/antibody fusion proteins in mice
- mice are introduced into mice as in Example 18 at a range of dose levels.
- Tissues are collected at time-points up to 1 week and the IL-22 response in on-target gut tissues is compared to off- target tissues. Consistent with the extended time in circulation, on-target tissues are expected to show a sustained increase in expression of IL-22 target genes.
- the on-target versus off-target enhancement of dose response is expected to be sustained.
- Example 20 Gut-targeted IL-22/antibody fusion proteins for treating mice following colon damage induced by dextran sulfate sodium
- Example 21 Other cytokines related to IL-22 are expected to show similar target-dependent potentiation when formatted into a tissue-targeted AND-body
- IL-22 and IL-10 are members in a family of cytokines with similar structural features and shared receptors (Ouyang et al., Immunity, 50: 871-891 , 2019).
- Specific cytokines in the family include IL-19, IL-20, IL-24, IL-26, IL-28A, IL-28B, and IL-29.
- a cytokine from this family which shares a receptor with IL-22 is fused to an antibody targeting a specific address expressed on one or more IL-22 target cells.
- a reporter cell assay similar to those described in the above examples is used to measure the activity of the cytokine/antibody fusion protein in (i) cells expressing or not expressing the address target, and (ii) cells expressing or not expressing the address target when the antibody is switched to a negative control. Potentiated signaling is expected to be observed only when the targeting antibody matches the address expressed on the reporter cells.
- Example 22. IL-22 muteins have decreased binding affinity to IL22Ra independent of ANDbody formatting
- a panel of nine point mutants of human IL-22 were generated as N-terminal fusions to human Fc. Constructs were expressed in Expi293 cells and purified using Protein A chromatography, as described in Example 4, and binding to human and mouse IL22Ra was measured using BLI. For these binding assays, the IL22-Fc protein variant was loaded onto an anti-human Fc biosensor probe (HFC, #160003, Gator Bio) for 120 seconds, and association was measured by dipping the probe into a solution of human IL22Ra extracellular domain for 150 seconds, followed by dissociation for 400 seconds. Data were experimentally fit to determine k O n and k o tf. KD was calculated as kott/kon. All experiments were performed with 1000 rpm shaking in Q Buffer (#120010, Gator Bio). Table 1 1 displays affinity data for binding of human IL-22-Fc muteins to human IL22Ra.
- Figs. 1 1 A-1 1 C show the thermal stability of a set of detuned IL-22 moieties (IL-22 muteins), as determined by differential scanning calorimetry (DSC) using a Nano DSC instrument (TA Instruments).
- the IL-22 muteins (D71 L / D168G (Fig. 1 1 A), R73Q (Fig. 1 1 B), and R73S (Fig. 1 1 C)) were fused to an anti-CDH17 humanized clone (AM93-015).
- Amino acid substitution mutations in IL-22 are numbered relative to the IL-22 sequence of SEQ ID NO: 7.
- Example 24 Activity of detuned IL-22 muteins on HaCaT cells
- Fig. 12 shows the potency of the IL-22 muteins D71 L / D168G, R73Q, and R73S on HaCaT (human keratinocyte) cells.
- these IL-22 muteins were designed to have attenuated binding to IL-22Ra, such that they would be expected to have reduced potency on cells expressing IL-22Ra unless otherwise recruited to these cells, e.g., via a connected (e.g., fused) “addressing” moiety (e.g., an antibody or fragment thereof that targets a moiety on the cell).
- HaCaT cells were stimulated with varying concentrations of native IL-22 or the IL-22 muteins fused to an anti-RSV antibody, then stained for phosphorylated STAT3 (pSTAT3) as a surrogate for IL-22 signaling activity. Fluorescence of the anti-pSTAT3 mAb was determined by flow cytometry.
- the R73S and R73Q muteins did not induce any measurable activity on HaCaT cells, even at the highest concentration tested (100 nM), indicating that these muteins have reduced potency on these cells relative to wild-type IL-22 and may be useful for avoiding unwanted IL-22 activity on skin.
- a macromolecule comprising a first binding site and a second binding site, wherein:
- the first binding site comprises an IL-22 polypeptide or a fragment or variant thereof that binds an IL-22 receptor on the surface of a target cell
- the second binding site is specific for an IL-10 family address target provided in Table 1 that is expressed on the surface of the target cell;
- the second binding site localizes the first binding site to the IL-10 family address target such that the IL-22 polypeptide or a fragment or variant thereof influences signaling by the IL-22 receptor in the target cell;
- the second binding site does not substantially influence signaling upon binding the IL-10 family address target; and (iii) the IL-22 polypeptide or fragment or variant thereof does not substantially influence signaling by the IL-22 receptor in the absence of localization by the second binding site.
- the target cell is a lower gastrointestinal (Gl) tract cell
- the IL-10 family address target is MS4A18, SI, MS4A12, ATP4B, MUC17, TMIGD1 , MS4A10, MEP1 A, CDH17, TM4SF20, SLC26A3, ATP4A, GPA33, BTNL3, MEP1 B, UGT2B17, KCNE2, or TMPRSS15.
- the target cell is a kidney cell
- the IL-10 family address target is TMEM207, OR2T10, SLC22A6, SLC22A8, SLC12A1 , AQP2, SLC22A12, SLC12A3, TMEM174, FXYD4, SLC34A1 , KCNJ1 , SLC22A1 1 , TMEM52B, SLC6A18, CTXN3, SLC4A1 , TMEM72, SLC22A2, SLC4A9, BSND, CDH16, RDH8, or AQP6.
- the target cell is a liver cell
- the IL-10 family address target is UGT2B10, C8A, SLCO1 B1 , C9, SLC25A47, SLC17A2, SLC10A1 , UGT2B4, SLC22A1 , or CYP8B1 .
- the target cell is a skin cell
- the IL-10 family address target is CLEC2A, KCNJ18, KLRF2, DSC1 , TYR, AWAT2, TMEM271 , DSG1 , ACER1 , GSDMA, TRPM1 , GJB4, CD1 A, ASPRV1 , or ABCA12.
- a macromolecule comprising a first binding site and a second binding site, wherein: (a) the first binding site comprises an IL-22 polypeptide or a fragment or variant thereof that binds an IL-22 receptor on the surface of a target cell, and
- the second binding site is specific for an IL-10 family address target selected from CDH17, CDH16, and GP2 that is expressed on the surface of the target cell;
- the second binding site localizes the first binding site to the IL-10 family address target such that the IL-22 polypeptide or fragment or variant thereof influences signaling by the IL-22 receptor in the target cell;
- the second binding site does not substantially influence signaling upon binding the IL-10 family address target
- the IL-22 polypeptide or fragment or variant thereof does not substantially influence signaling by the IL-22 receptor in the absence of localization by the second binding site.
- the antibody or antigen-binding fragment thereof comprises an scFv, BsIgG, a BsAb fragment, a BiTE, a dual-affinity re-targeting protein (DART), a tandem diabody (TandAb), a diabody, an Fab2, a di-scFv, chemically linked F(ab’)2, an Ig molecule with 2, 3 or 4 different antigen binding sites, a DVI-IgG four-in-one, an ImmTac, an HSAbody, an IgG-IgG, a Cov-X-Body, an scFv1 -PEG-scFv2, an appended IgG, an DVD-IgG, an affibody, an affilin, an affimer, an affitin, an alphabody, an anticalin, an avimer, a DARPin, a Fynomer, a monobody, a nanoCL
- the macromolecule of embodiment 15 or 16, wherein the IL-10 family address target is CDH16 and the second binding site is an anti-CDH16 antibody or antigen-binding fragment thereof.
- the IL-10 family address target is GP2 and the second binding site is an anti-GP2 antibody or antigen-binding fragment thereof.
- the Kd of the IL-22 polypeptide or fragment or variant thereof for IL-22Ra and/or IL-10R2 is higher than the Kd of the second binding site for the IL-10 family address target;
- the ECso of the IL-22 polypeptide or fragment or variant thereof for IL-22Ra and/or IL-10R2 is higher than the EC50 of the second binding site for the IL-10 family address target;
- the IC50 of the IL-22 polypeptide or fragment or variant thereof for IL-22Ra and/or IL-10R2 is higher than the IC50 of the second binding site for the IL-10 family address target.
- IL-22 polypeptide or fragment or variant thereof exhibits reduced agonist activity relative to the IL-22 polypeptide or fragment or variant thereof when not part of the macromolecule, on a mole-adjusted basis.
- 31 The macromolecule of any one of embodiments 1 -30, wherein the IL-22 polypeptide or fragment or variant thereof comprises one or more amino acid substitution mutations at one or more residues that are predicted to be interaction surfaces between IL-22 and IL-22Ra and/or IL-10R2.
- IL-22 polypeptide or fragment or variant thereof comprises one or more amino acid substitution mutations at one or more residues that are predicted to be interaction surfaces between IL-22 and IL-22Ra.
- IL-22 polypeptide or fragment or variant thereof is a human IL-22 or fragment thereof and comprises one or more of the amino acid substitution mutations of Table 6, Table 7A, Table 7B, Table 8, Table 9A, Table 9B, or Table 9C.
- the macromolecule of embodiment 40 wherein the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 13. 42.
- the macromolecule of any one of embodiments 1 -41 wherein the macromolecule comprises two first binding sites comprising an IL-22 polypeptide or fragment or variant thereof that binds IL-22Ra and/or IL-10R2.
- each of the fusion proteins comprises a linker conjugated to the first binding site and the second binding site.
- a macromolecule comprising two subunits, each comprising a first binding site and a second binding site, wherein:
- each of the first binding sites comprises an IL-22 polypeptide or a fragment or variant thereof that binds an IL-22 receptor on the surface of a target cell
- each of the second binding sites is specific for an IL-10 family address target provided in Table 1 that is expressed on the surface of the target cell;
- the second binding sites localize the first binding sites to the IL-10 family address target such that the IL-22 polypeptides or fragments or variants thereof influence signaling by the IL-22 receptor in the target cell;
- the second binding sites do not substantially influence signaling upon binding the IL-10 family address targets; and (iii) the IL-22 polypeptides or fragments or variants thereof do not substantially influence signaling by the IL-22 receptor in the absence of localization by the second binding sites.
- a macromolecule comprising two subunits, each comprising a first binding site and a second binding site, wherein:
- each of the first binding sites comprises an IL-22 polypeptide or a fragment or variant thereof that binds an IL-22 receptor on the surface of a target cell
- each of the second binding sites is specific for an IL-10 family address target selected from CDH17, CDH16, and GP2 that is expressed on the surface of the target cell;
- the second binding sites localize the first binding sites to the IL-10 family address target such that the IL-22 polypeptides or fragments or variants thereof influence signaling by the IL-22 receptor in the target cell;
- the IL-22 polypeptides or fragments or variants thereof do not substantially influence signaling by the IL-22 receptor in the absence of localization by the second binding sites.
- a method of modulating IL-22 receptor signaling in a target cell of a subject comprising administering to the subject the macromolecule of any one of embodiments 1 -61 .
- a macromolecule comprising:
- a second binding site comprising an anti-CDH17 antibody or antigen-binding fragment thereof, wherein the macromolecule is a fusion protein, and wherein the first binding site and the second binding site are connected by a linker.
- the anti-CDH17 antibody or antigen-binding fragment thereof localizes the first binding site to an IL-22 receptor such that the IL-22 polypeptide or fragment or variant thereof influences signaling by the IL-22 receptor in the target cell;
- the anti-CDH17 antibody or antigen-binding fragment thereof does not substantially influence signaling upon binding CDH17;
- the IL-22 polypeptide or fragment or variant thereof does not substantially influence signaling by the IL-22 receptor in the absence of localization by the anti-CDH17 antibody or antigen-binding fragment thereof.
- a macromolecule comprising two subunits, each comprising a first binding site and a second binding site, wherein:
- each first binding site comprises an IL-22 polypeptide or a fragment or variant thereof
- each second binding site comprises an anti-CDH17 antibody or antigen-binding fragment thereof, wherein the macromolecule is a fusion protein, and wherein the first binding site and the second binding site of each subunit are connected by a linker.
- the anti-CDH17 antibodies or antigen-binding fragments thereof localize the first binding sites to an IL-22 receptor such that the IL-22 polypeptides or fragments or variants thereof influence signaling by the IL-22 receptor in the target cell;
- the anti-CDH17 antibodies or antigen-binding fragments thereof do not substantially influence signaling upon binding CDH17;
- the IL-22 polypeptides or fragments or variants thereof do not substantially influence signaling by the IL-22 receptor in the absence of localization by the anti-CDH17 antibodies or antigen-binding fragments thereof.
- a method of modulating IL-22 receptor signaling in the small intestine or colon of a subject comprising administering to the subject the macromolecule of any one of embodiments 63-66.
- a macromolecule comprising:
- a second binding site comprising an anti-CDH16 antibody or antigen-binding fragment thereof, wherein the macromolecule is a fusion protein, and wherein the first binding site and the second binding site are connected by a linker.
- the anti-CDH16 antibody or antigen-binding fragment thereof localizes the first binding site to an IL-22 receptor such that the IL-22 polypeptide or fragment or variant thereof influences signaling by the IL-22 receptor in the target cell;
- the anti-CDH16 antibody or antigen-binding fragment thereof does not substantially influence signaling upon binding CDH16;
- the IL-22 polypeptide or fragment or variant thereof does not substantially influence signaling by the IL-22 receptor in the absence of localization by the anti-CDH16 antibody or antigen-binding fragment thereof.
- a macromolecule comprising two subunits, each comprising a first binding site and a second binding site, wherein:
- each first binding site comprises an IL-22 polypeptide or a fragment or variant thereof
- each second binding site comprises an anti-CDH16 antibody or antigen-binding fragment thereof, wherein the macromolecule is a fusion protein, and wherein the first binding site and the second binding site of each subunit are connected by a linker.
- the anti-CDH16 antibodies or antigen-binding fragments thereof localize the first binding sites to an IL-22 receptor such that the IL-22 polypeptides or fragments or variants thereof influence signaling by the IL-22 receptor in the target cell;
- the anti-CDH16 antibodies or antigen-binding fragments thereof do not substantially influence signaling upon binding CDH16;
- the IL-22 polypeptides or fragments or variants thereof do not substantially influence signaling by the IL-22 receptor in the absence of localization by the anti-CDH16 antibodies or antigen-binding fragments thereof.
- a method of modulating IL-22 receptor signaling in the kidney of a subject comprising administering to the subject the macromolecule of any one of embodiments 68-71 .
- a macromolecule comprising:
- a second binding site comprising an anti-GP2 antibody or antigen-binding fragment thereof, wherein the macromolecule is a fusion protein, and wherein the first binding site and the second binding site are connected by a linker.
- the anti-GP2 antibody or antigen-binding fragment thereof localizes the first binding site to an IL- 22 receptor such that the IL-22 polypeptide or fragment or variant thereof influences signaling by the IL- 22 receptor in the target cell;
- the anti-GP2 antibody or antigen-binding fragment thereof does not substantially influence signaling upon binding GP2;
- the IL-22 polypeptide or fragment or variant thereof does not substantially influence signaling by the IL-22 receptor in the absence of localization by the anti-GP2 antibody or antigen-binding fragment thereof.
- a macromolecule comprising two subunits, each comprising a first binding site and a second binding site, wherein:
- each first binding site comprises an IL-22 polypeptide or a fragment or variant thereof
- each second binding site comprises an anti-GP2 antibody or antigen-binding fragment thereof, wherein the macromolecule is a fusion protein, and wherein the first binding site and the second binding site of each subunit are connected by a linker.
- the anti-GP2 antibodies or antigen-binding fragments thereof localize the first binding sites to an IL-22 receptor such that the IL-22 polypeptides or fragments or variants thereof influence signaling by the IL-22 receptor in the target cell;
- the IL-22 polypeptides or fragments or variants thereof do not substantially influence signaling by the IL-22 receptor in the absence of localization by the second binding sites.
- a method of modulating IL-22 receptor signaling in the pancreas of a subject comprising administering to the subject the macromolecule of any one of embodiments 73-76.
- IL-22 polypeptide or fragment or variant thereof is a human IL-22 polypeptide or fragment or variant thereof and comprises one or more of the amino acid substitution mutations of Table 6, Table 7A, Table 7B, Table 8, Table 9A, Table 9B, or Table 9C.
- composition comprising the macromolecule of any one of embodiments 1 -61 , 63-66, 68-71 , 73- 76, and 78-85.
- composition of embodiment 86, wherein the composition comprises a pharmaceutically acceptable carrier or excipient.
- composition of embodiment 86 or 87, wherein the composition is a unit dosage form suitable for administration to a human subject.
- nucleic acids of embodiment 89 wherein the nucleic acids are linear nucleic acids, circular nucleic acids, and/or modified nucleic acids.
- One or more vectors comprising the one or more nucleic acids of embodiment 89 or 90.
- One or more host cells comprising the one or more vectors of embodiment 91 .
- the method further comprises recovering the macromolecule from the one or more host cells or the culture medium.
- a method comprising administering a therapeutically effective amount of the macromolecule, composition, or nucleic acid of any one of embodiments 1 -61 , 63-66, 68-71 , 73-76, and 78-85 to a subject in need thereof.
- a human IL-22 polypeptide or a fragment or variant thereof that binds an IL-22 receptor on the surface of a target cell wherein the IL-22 polypeptide or fragment or variant thereof comprises one or more of the amino acid substitution mutations of Table 6, Table 7A, Table 7B, Table 8, Table 9A, Table 9B, or Table 9C.
- a human IL-22 polypeptide or a fragment or variant thereof that binds an IL-22 receptor on the surface of a target cell wherein the human IL-22 polypeptide or fragment or variant thereof comprises an amino acid substitution mutation of one or more of K61 , D71 , R73, D168, and R175, wherein the amino acid position numbering is with reference to the sequence of SEQ ID NO: 7.
- human IL-22 polypeptide or a fragment or variant thereof of embodiment 98 wherein the human IL-22 polypeptide or fragment or variant thereof comprises one or more of a K61 S, D71 N, D71 L, D71 G, R73Q, R73S, R73N, D168N, D168G, and R175S amino acid substitution mutation.
- human IL-22 polypeptide or a fragment or variant thereof of embodiment 98 wherein the human IL-22 polypeptide or fragment or variant thereof comprises a K61 S amino acid substitution mutation.
- human IL-22 polypeptide or a fragment or variant thereof of embodiment 98 wherein the human IL-22 polypeptide or fragment or variant thereof comprises a R73Q amino acid substitution mutation.
- human IL-22 polypeptide or a fragment or variant thereof of embodiment 98 wherein the human IL-22 polypeptide or fragment or variant thereof comprises a D168N amino acid substitution mutation.
- human IL-22 polypeptide or a fragment or variant thereof of embodiment 98 wherein the human IL-22 polypeptide or fragment or variant thereof comprises D71 L and D168G amino acid substitution mutations.
- a polypeptide comprising the human IL-22 polypeptide or a fragment or variant thereof of any one of embodiments 98 to 129.
- composition comprising a polypeptide of any one of embodiments 98 to 130.
- composition of embodiment 131 wherein the composition comprises a pharmaceutically acceptable carrier or excipient.
- composition of embodiment 131 or 132, wherein the composition is a unit dosage form suitable for administration to a human subject.
- nucleic acid of embodiment 134 wherein the nucleic acid is a linear nucleic acid, a circular nucleic acid, and/or a modified nucleic acid.
- a vector comprising the nucleic acid of embodiment 134 or 135.
- a host cell comprising the vector of embodiment 136.
- An IL-22 mutein polypeptide or a fragment or variant thereof that binds an IL-22 receptor on the surface of a target cell wherein the IL-22 mutein polypeptide or fragment or variant thereof comprises one or more of the amino acid substitution mutations provided in Table 6, Table 7A, Table 7B, Table 8, Table 9A, Table 9B, or Table 9C, wherein the amino acid positions are numbered relative to SEQ ID NO: 7.
- An IL-22 mutein polypeptide or a fragment or variant thereof that binds an IL-22 receptor on the surface of a target cell wherein the IL-22 mutein polypeptide or fragment or variant thereof comprises an amino acid substitution mutation at one or more of positions K61 , D71 , R73, D168, and R175 relative to a human IL-22 polypeptide, wherein the amino acid positions are numbered relative to SEQ ID NO: 7.
- IL-22 mutein polypeptide or fragment or variant thereof of embodiment 141 wherein the IL- 22 polypeptide or fragment or variant thereof comprises one or more of a K61 S, D71 N, D71 L, D71 G, R73Q, R73S, R73N, D168N, D168G, and R175S amino acid substitution mutation.
- the IL-22 mutein polypeptide or fragment or variant thereof comprises a K61 S amino acid substitution mutation, optionally wherein the IL-22 mutein polypeptide or fragment or variant thereof comprises SEQ ID NO: 17;
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Life Sciences & Earth Sciences (AREA)
- Immunology (AREA)
- General Health & Medical Sciences (AREA)
- Medicinal Chemistry (AREA)
- Biochemistry (AREA)
- Biophysics (AREA)
- Genetics & Genomics (AREA)
- Molecular Biology (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Toxicology (AREA)
- Zoology (AREA)
- Gastroenterology & Hepatology (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Transplantation (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Pharmacology & Pharmacy (AREA)
- Animal Behavior & Ethology (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
- Peptides Or Proteins (AREA)
Abstract
Macromolecule compositions and related methods that effect targeted delivery of IL-22 polypeptides or fragments or variants thereof to effector targets in a desired cell, tissue and/or organ of interest while minimizing or avoiding undesirable delivery to other cells, tissues, or organs are provided. Compositions and methods related to macromolecules, such as an ANDbody™, that include an IL-22 polypeptide or fragment or variant thereof and an address binding domain specific for an IL-10 family address target are described. Also provided are IL-22 mutein polypeptides that have altered (e.g., decreased) affinity for the IL-22 receptor.
Description
COMPOSITIONS AND METHODS FOR TARGETED DELIVERY OF THERAPEUTIC AGENTS
SEQUENCE LISTING
The instant application contains a Sequence Listing which has been submitted electronically in XML format and is hereby incorporated by reference in its entirety. Said XML copy, created on November 7, 2024, is named 51666-005WO9_Sequence_Listing_11_7_24 and is 23,656 bytes in size.
FIELD OF THE INVENTION
Macromolecule compositions and related methods that effect targeted delivery of IL-22 polypeptides or fragments or variants thereof to effector targets in a desired cell, tissue and/or organ of interest while minimizing or avoiding undesirable delivery to other cells, tissues, or organs are provided. Compositions and methods related to macromolecules, such as an ANDbody™, that include an IL-22 polypeptide or a fragment or variant thereof and an address binding domain specific for an IL-10 family address target are described. Also provided are IL-22 mutein polypeptides that have altered (e.g., decreased) affinity for the IL-22 receptor.
BACKGROUND OF THE INVENTION
Undesirable off-target effects are a problem for otherwise desirable therapeutic targets that are present in healthy as well as diseased tissues.
SUMMARY OF THE INVENTION
The present disclosure describes, in part, macromolecule compositions and related methods that effect targeted delivery of therapeutic agents to effector targets in a targeted cell, tissue, or organ while minimizing or avoiding undesirable delivery to other cells, tissues or organs. Generally, compositions described herein comprise macromolecules, such as an ANDbody™, that include an effector target binding domain comprising an IL-22 polypeptide or fragment or variant thereof that binds an IL-22 receptor (e.g., a receptor comprising IL-22Ra, and/or IL-10R2, e.g., a receptor comprising IL-22Ra and IL-10R2), and an address binding domain specific for (e.g., that specifically binds to) an IL-10 family address target. The address target is generally sufficiently restricted in the subject to target the macromolecule to the desired cell, tissue or organ. In some embodiments, the IL-22 polypeptide or fragment or variant thereof does not influence signaling by the IL-22 receptor in the absence of an address target binding domain. Moreover, the address target binding domain may not influence signaling upon binding the IL-10 family address target. However, localization of the IL-22 polypeptide or fragment or variant thereof by the address target binding domain enables the IL-22 polypeptide or fragment or variant thereof to bind the IL-22 receptor sufficiently to elicit an influence on signaling by the IL-22 receptor in the target cell or tissue. In addition, the macromolecules described herein may be linked to one or more small molecules.
The compositions described herein can be used, e.g., to specifically deliver a therapeutic agent (for example, the IL-22 polypeptide or fragment or variant thereof, the small molecule, or both) to a desired location, e.g., a target cell, tissue or organ, in a subject, while avoiding undesirable off-target effects (e.g., undesirable off-target effects in the brain; skin; cardiovascular system, e.g., heart or
vasculature) and/or avoiding certain toxicity (e.g., avoiding cardiovascular disease, such as stroke or myocardial infarction).
In one aspect, the disclosure provides an IL-22 mutein polypeptide or a fragment or variant thereof that binds an IL-22 receptor on the surface of a target cell, wherein the IL-22 mutein polypeptide or fragment or variant thereof comprises one or more of the amino acid substitution mutations provided in Table 6, Table 7A, Table 7B, Table 8, Table 9A, Table 9B, or Table 9C, wherein the amino acid positions are numbered relative to SEQ ID NO: 7.
In another aspect, the disclosure provides an IL-22 mutein polypeptide or a fragment or variant thereof that binds an IL-22 receptor on the surface of a target cell, wherein the IL-22 mutein polypeptide or fragment or variant thereof comprises an amino acid substitution mutation at one or more of positions K61 , D71 , R73, D168, and R175 relative to a human IL-22 polypeptide, wherein the amino acid positions are numbered relative to SEQ ID NO: 7.
In some embodiments, the IL-22 polypeptide or fragment or variant thereof comprises one or more of a K61 S, D71 N, D71 L, D71 G, R73Q, R73S, R73N, D168N, D168G, and R175S amino acid substitution mutation.
In some embodiments, (i) the IL-22 mutein polypeptide or fragment or variant thereof comprises a K61 S amino acid substitution mutation, optionally wherein the IL-22 mutein polypeptide or fragment or variant thereof comprises SEQ ID NO: 17; (ii) the IL-22 mutein polypeptide or fragment or variant thereof comprises a D71 G amino acid substitution mutation, optionally wherein the IL-22 mutein polypeptide or fragment or variant thereof comprises SEQ ID NO: 15; (iii) the IL-22 mutein polypeptide or fragment or variant thereof comprises a D71 L amino acid substitution mutation, optionally wherein the IL-22 mutein polypeptide or fragment or variant thereof comprises SEQ ID NO: 11 ; (iv) the IL-22 mutein polypeptide or fragment or variant thereof comprises a D71 N amino acid substitution mutation, optionally wherein the IL- 22 mutein polypeptide or fragment or variant thereof comprises SEQ ID NO: 18; (v) the IL-22 mutein polypeptide or fragment or variant thereof comprises an R73S amino acid substitution mutation, optionally wherein the IL-22 mutein polypeptide or fragment or variant thereof comprises SEQ ID NO: 16; (vi) the IL-22 mutein polypeptide or fragment or variant thereof comprises an R73Q amino acid substitution mutation, optionally wherein the IL-22 mutein polypeptide or fragment or variant thereof comprises SEQ ID NO: 12; (vii) the IL-22 mutein polypeptide or fragment or variant thereof comprises a D168N amino acid substitution mutation, optionally wherein the IL-22 mutein polypeptide or fragment or variant thereof comprises SEQ ID NO: 19; (viii) the IL-22 mutein polypeptide or fragment or variant thereof comprises a D168G amino acid substitution mutation, optionally wherein the IL-22 mutein polypeptide or fragment or variant thereof comprises SEQ ID NO: 20; (ix) the IL-22 mutein polypeptide or fragment or variant thereof comprises D71 L and D168G amino acid substitution mutations, optionally wherein the IL-22 mutein polypeptide or fragment or variant thereof comprises SEQ ID NO: 13; (x) the IL- 22 mutein polypeptide or fragment or variant thereof comprises an R175S amino acid substitution mutation, optionally wherein the IL-22 mutein polypeptide or fragment or variant thereof comprises SEQ ID NO: 21 ; or (xi) the IL-22 mutein polypeptide or fragment or variant thereof comprises an R73N amino acid substitution mutation, optionally wherein the IL-22 mutein polypeptide or fragment or variant thereof comprises SEQ ID NO: 22.
In some embodiments, the IL-22 mutein polypeptide or fragment or variant thereof comprises one or more additional amino acid substitution mutations at one or more residues that are predicted to be interaction surfaces between IL-22 and IL-22Ra and/or IL-10R2.
In some embodiments, the IL-22 mutein polypeptide or fragment or variant thereof has a binding affinity for IL22RA1 (IL-22Ra) that is at least 1 .3-fold lower than the binding affinity of a wild-type IL-22 or fragment thereof.
In some embodiments, the IL-22 mutein polypeptide or fragment or variant thereof does not comprise a leader sequence.
In some embodiments, the IL-22 mutein polypeptide or fragment or variant thereof does not comprise the amino acid sequence of SEQ ID NO: 14.
In another aspect, the disclosure provides a polypeptide comprising the IL-22 mutein polypeptide or fragment or variant thereof of any one of the above aspects.
In another aspect, the disclosure provides an IL-22 mutein polypeptide that has at least 95% identity to human IL-22 (SEQ ID NO: 7), comprises D71 L and D168G amino acid substitution mutations relative to SEQ ID NO: 7, and has a significantly reduced binding affinity for IL-22Ra relative to a wildtype human IL-22 polypeptide.
In another aspect, the disclosure provides an IL-22 mutein polypeptide that has at least 95% identity to human IL-22 (SEQ ID NO: 7), comprises an R73Q amino acid substitution mutation relative to SEQ ID NO: 7, and has a significantly reduced binding affinity for IL-22Ra relative to a wild-type human IL-22 polypeptide.
In another aspect, the disclosure provides an IL-22 mutein polypeptide that has at least 95% identity to human IL-22 (SEQ ID NO: 7), comprises an R73S amino acid substitution mutation relative to SEQ ID NO: 7, and has a significantly reduced binding affinity for IL-22Ra relative to a wild-type human IL-22 polypeptide.
In another aspect, the disclosure provides a macromolecule comprising a first binding site and a second binding site, wherein (a) the first binding site comprises an IL-22 polypeptide or a fragment or variant thereof that binds an IL-22 receptor on the surface of a target cell, and (b) the second binding site is specific for an IL-10 family address target provided in Table 1 that is expressed on the surface of the target cell; wherein: (i) the second binding site localizes the first binding site to the IL-10 family address target such that the IL-22 polypeptide or a fragment or variant thereof influences signaling by the IL-22 receptor in the target cell; (ii) the second binding site does not substantially influence signaling upon binding the IL-10 family address target; and (iii) the IL-22 polypeptide or fragment or variant thereof does not substantially influence signaling by the IL-22 receptor in the absence of localization by the second binding site.
In some embodiments, the IL-22 polypeptide or fragment or variant thereof has been engineered to have reduced binding affinity for the IL-22 receptor. In some embodiments, the IL-22 polypeptide or fragment or variant thereof comprises one or more amino acid substitution mutations at one or more residues that are predicted to be interaction surfaces between IL-22 and IL-22Ra and/or IL-10R2. In some embodiments, the IL-22 polypeptide or fragment or variant thereof is a variant of a human IL-22 or fragment thereof and comprises (a) one or more of the amino acid substitution mutations of Table 6, Table 7A, Table 7B, Table 8, Table 9A, Table 9B, or Table 9C; and/or (b) one or more of a K61 S, D71 N, D71 L, D71 G, R73Q, R73S, R73N, D168N, D168G, and R175S amino acid substitution mutation.
In another aspect, the disclosure provides a method of modulating IL-22 receptor signaling in a target cell of a subject, the method comprising administering to the subject the macromolecule of any one of the above aspects.
In another aspect, the disclosure provides a method of treating a disease or disorder selected from Crohn's disease, ulcerative colitis, graft-vs-host disease, atopic dermatitis, psoriasis, wounds, diabetic skin ulcers, acute pancreatitis, chronic pancreatitis, autoimmune pancreatitis, nephritis, acute lung injury, viral lung injury, fibrosis, fibrosis, non-alcoholic steatohepatitis, viral hepatitis, and autoimmune hepatitis in a subject, the method comprising administering to the subject the macromolecule of any one of the above aspects.
In another aspect, the disclosure provides a macromolecule comprising (a) a first binding site comprising an IL-22 polypeptide or a fragment or variant thereof, and (b) a second binding site comprising an anti-CDH17 antibody or antigen-binding fragment thereof, wherein the macromolecule is a fusion protein, and wherein the first binding site and the second binding site are connected by a linker.
In another aspect, the disclosure provides a macromolecule comprising a first binding site and a second binding site, wherein (a) the first binding site comprises an IL-22 polypeptide or a fragment or variant thereof that binds an IL-22 receptor on the surface of a target cell, and (b) the second binding site is specific for an IL-10 family address target provided in Table 1 that is expressed on the surface of the target cell; wherein (i) the second binding site localizes the first binding site to the IL-10 family address target such that the IL-22 polypeptide or a fragment or variant thereof influences signaling by the IL-22 receptor in the target cell; (ii) the second binding site does not substantially influence signaling upon binding the IL-10 family address target; and (iii) the IL-22 polypeptide or fragment or variant thereof does not substantially influence signaling by the IL-22 receptor in the absence of localization by the second binding site.
In some embodiments, the IL-10 family address target is a protein, lipid, or sugar. In some embodiments, the IL-10 family address target is a protein.
In some embodiments, the target cell is a lower gastrointestinal (G I) tract cell, and the IL-10 family address target is MS4A18, SI, MS4A12, ATP4B, MUC17, TMIGD1 , MS4A10, MEP1A, CDH17, TM4SF20, SLC26A3, ATP4A, GPA33, BTNL3, MEP1 B, UGT2B17, KCNE2, or TMPRSS15.
In some embodiments, the target cell is a kidney cell, and the IL-10 family address target is TMEM207, OR2T10, SLC22A6, SLC22A8, SLC12A1 , AQP2, SLC22A12, SLC12A3, TMEM174, FXYD4, SLC34A1 , KCNJ1 , SLC22A11 , TMEM52B, SLC6A18, CTXN3, SLC4A1 , TMEM72, SLC22A2, SLC4A9, BSND, CDH16, RDH8, or AQP6.
In some embodiments, the target cell is a liver cell, and the IL-10 family address target is UGT2B10, C8A, SLCO1 B1 , C9, SLC25A47, SLC17A2, SLC10A1 , UGT2B4, SLC22A1 , or CYP8B1 .
In some embodiments, the target cell is a pancreas cell, and the IL-10 family address target is GP2, AQP12A, AQP12B, CUZD1 , LHFPL5, AQP8, KIRREL2, G6PC2, SLC17A6, or GPR119.
In some embodiments, the target cell is a skin cell, and the IL-10 family address target is CLEC2A, KCNJ18, KLRF2, DSC1 , TYR, AWAT2, TMEM271 , DSG1 , ACER1 , GSDMA, TRPM1 , GJB4, CD1A, ASPRV1 , or ABCA12.
In some embodiments, the IL-10 family address target is selected from CDH17, GP2, CDH16, DSG1 , and DSG3. In some embodiments, the IL-10 family address target is CDH17. In some embodiments, the IL-10 family address target is CDH16. In some embodiments, the IL-10 family address target is GP2.
In another aspect, the disclosure provides a macromolecule comprising a first binding site and a second binding site, wherein (a) the first binding site comprises an IL-22 polypeptide or a fragment or variant thereof that binds an IL-22 receptor on the surface of a target cell, and (b) the second binding site is specific for an IL-10 family address target selected from CDH17, CDH16, and GP2 that is expressed on the surface of the target cell; wherein (i) the second binding site localizes the first binding site to the IL-10 family address target such that the IL-22 polypeptide or fragment or variant thereof influences signaling by the IL-22 receptor in the target cell; (ii) the second binding site does not substantially influence signaling upon binding the IL-10 family address target; and (iii) the IL-22 polypeptide or fragment or variant thereof does not substantially influence signaling by the IL-22 receptor in the absence of localization by the second binding site.
In some embodiments, the second binding site comprises a polypeptide. In some embodiments, the second binding site is an antibody or antigen binding fragment thereof. In some embodiments, the antibody or antigen-binding fragment thereof comprises an scFv, BsIgG, a BsAb fragment, a BiTE, a dual-affinity re-targeting protein (DART), a tandem diabody (TandAb), a diabody, an Fab2, a di-scFv, chemically linked F(ab’)2, an Ig molecule with 2, 3 or 4 different antigen binding sites, a DVI-IgG four-in- one, an ImmTac, an HSAbody, an IgG-IgG, a Cov-X-Body, an scFv1 -PEG-scFv2, an appended IgG, an DVD-IgG, an affibody, an affilin, an affimer, an affitin, an alphabody, an anticalin, an avimer, a DARPin, a Fynomer, a monobody, a nanoCLAMP, a bis-Fab, an Fv, a Fab, a Fab’-SH, a linear antibody, an scFv, an antibody with only a heavy chain (Humabody), an ScFab, an IgG antibody fragment, a single-chain variable region antibody, a single-domain heavy chain antibody, a bispecific triplebody, a BiKE, a CrossMAb, a dsDb, an scDb, tandem a dAb I VHH, a triple dAb VHH, a tetravalent dAb I VHH, a Fab- scFv, a Fab-Fv, or a DART-Fc, an adnectin, a Kunitz-type inhibitor, or a receptor decoy.
In some embodiments, the second binding site comprises a ligand of the address target.
In some embodiments, the IL-10 family address target is CDH17 and the second binding site is an anti-CDH17 antibody or antigen-binding fragment thereof.
In some embodiments, the IL-10 family address target is CDH16 and the second binding site is an anti-CDH16 antibody or antigen-binding fragment thereof.
In some embodiments, the IL-10 family address target is GP2 and the second binding site is an anti-GP2 antibody or antigen-binding fragment thereof.
In one aspect, the disclosure provides an IL-22 polypeptide or a fragment or variant thereof that binds (e.g., specifically binds) an IL-22 receptor on the surface of a target cell.
In some embodiments, the IL-22 polypeptide or fragment or variant thereof binds the IL-22 receptor. In some embodiments, the IL-22 polypeptide or fragment or variant thereof binds the human IL-22 receptor. In some embodiments, the IL-22 polypeptide or fragment or variant thereof binds human IL-22Ra and/or IL-10R2. For example, binding can be evaluated using an assay described herein.
In some embodiments, the IL-22 polypeptide or fragment or variant thereof binds IL-22BP. In some embodiments, the IL-22 polypeptide or fragment or variant thereof binds human IL-22BP.
In some embodiments, the IL-22 polypeptide or fragment or variant thereof agonizes the IL-22 receptor. In some embodiments, the IL-22 polypeptide or fragment or variant thereof agonizes the human IL-22 receptor. In some embodiments, the IL-22 polypeptide or fragment or variant thereof agonizes human IL-22Ra and/or IL-10R2, e.g., in an assay described herein.
In some embodiments, the macromolecule (i.e., a macromolecule comprising the IL-22 polypeptide or fragment or variant thereof) agonizes IL-22BP. In some embodiments, the macromolecule agonizes human IL-22BP.
In some embodiments, the IL-22 polypeptide or fragment or variant thereof has low (e.g., lower) binding affinity for IL-22Ra and/or IL-10R2, as compared to the binding affinity of wild type human IL-22 (e.g., of SEQ ID NO: 8) for the IL-22 receptor, e.g., in an assay described herein.
In some embodiments, the IL-22 polypeptide or fragment or variant thereof has low (e.g., lower) binding affinity for IL-22BP, as compared to the binding affinity of wild type human IL-22 (e.g., of SEQ ID NO: 8) for IL-22BP, e.g., in an assay described herein.
In some embodiments, the affinity of the IL-22 polypeptide or fragment or variant thereof for IL- 22Ra, IL-22BP, and/or IL-10R2 is lower than the affinity of the second binding site for the IL-10 family address target.
In some embodiments, the avidity of the IL-22 polypeptide or fragment or variant thereof for IL- 22Ra, IL-22BP, and/or IL-10R2 is lower than the avidity of the second binding site for the IL-10 family address target.
In some embodiments, (a) the Kd of the IL-22 polypeptide or fragment or variant thereof for IL- 22Ra, IL-22BP, and/or IL-10R2 is higher than the Kd of the second binding site for the IL-10 family address target; (b) the ECso of the IL-22 polypeptide or fragment or variant thereof for IL-22Ra and/or IL- 10R2 is higher than the ECso of the second binding site for the IL-10 family address target; or (c) the IC50 of the IL-22 polypeptide or fragment or variant thereof for IL-22Ra and/or IL-10R2 is higher than the IC50 of the second binding site for the IL-10 family address target.
In some embodiments, the IL-22 polypeptide or fragment or variant thereof has an affinity to the IL-22 receptor of at least about 2 times, at least about 5 times, or at least about 10 times less than the affinity of the second binding site to the IL-10 family address target.
In some embodiments, the affinity of the second binding site to the IL-10 family address target has a Kd of greater than about 1 nM, greater than about 2 nM, or greater than about 50 nm.
In some embodiments, the IL-22 polypeptide or fragment or variant thereof has been engineered to have reduced binding affinity for the IL-22 receptor.
In some embodiments, when part of the macromolecule, the IL-22 polypeptide or fragment or variant thereof exhibits reduced agonist activity relative to the IL-22 polypeptide or fragment or variant thereof when not part of the macromolecule, on a mole-adjusted basis.
In some embodiments, the IL-22 polypeptide or fragment or variant thereof comprises one or more amino acid substitution mutations at one or more residues that are predicted to be interaction surfaces between IL-22 and IL-22Ra and/or IL-10R2.
In some embodiments, the IL-22 polypeptide or fragment or variant thereof has reduced binding affinity for IL22RA1 relative to a wild-type IL-22 or fragment thereof. In some embodiments, the IL-22 polypeptide or fragment or variant thereof comprises one or more amino acid substitution mutations at one or more residues that are predicted to be interaction surfaces between IL-22 and IL-22Ra.
In some embodiments, the IL-22 polypeptide or fragment or variant thereof does not comprise a leader sequence, e.g., does not comprise a leader sequence of a pro-form of an IL-22 polypeptide. In some embodiments, the IL-22 polypeptide or fragment or variant thereof does not comprise the leader sequence comprised by the polypeptide of SEQ ID NO: 7, e.g., does not comprise residues 1 to 33 included in SEQ ID NO: 7.
In some embodiments, the IL-22 polypeptide or fragment or variant thereof does not comprise the sequence of SEQ ID NO: 14.
In some embodiments, the IL-22 polypeptide or fragment or variant thereof is a human IL-22 or fragment or variant thereof.
In some embodiments, the IL-22 polypeptide or fragment or variant thereof is a human IL-22 or fragment or variant thereof and comprises one or more of the amino acid substitution mutations of Table 6, Table 7A, Table 7B, Table 8, Table 9A, Table 9B, or Table 9C.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises an amino acid substitution mutation of one or more of K61 , D71 , R73, D168, and R175. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a K61 amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D71 amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a R73 amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D168 amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a R175 amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D71 amino acid substitution mutation and a D168 amino acid substitution mutation. The amino acid position numbering is with reference to the IL-22 sequence of SEQ ID NO: 7.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises one or more of a K61 S, D71 N, D71 L, D71 G, R73Q, R73S, R73N, D168N, D168G, and R175S amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a K61 S amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D71 N amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D71 L amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D71 G amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a R73Q amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a R73S amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a R73N amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D168N amino acid substitution mutation. In some
embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D168G amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a R175S amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D71 L amino acid substitution mutation and a D168G amino acid substitution mutation. The amino acid position numbering is with reference to the IL- 22 sequence of SEQ ID NO: 7.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a K61 S amino acid substitution mutation.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 17 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 17).
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a K61 S amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the human IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 17.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 17.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D71 G amino acid substitution mutation.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 15 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 15).
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D71 G amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the human IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 15.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 15.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D71 L amino acid substitution mutation.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 11 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 11 ).
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D71 L amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the human IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 11 .
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 11.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D71 N amino acid substitution mutation.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 18 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 18).
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D71 N amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the human IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 18.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 18.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a R73S amino acid substitution mutation.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 16 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 16).
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a R73S amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the human IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 16.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 16.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a R73N amino acid substitution mutation.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 22 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 22).
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a R73N amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the human IL-22 polypeptide or fragment or variant thereof to SEQ ID NO:22
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 22.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a R73Q amino acid substitution mutation.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 12 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 12).
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a R73Q amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%,
94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the human IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 12.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 12.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D168N amino acid substitution mutation.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 19 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 19).
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D168N amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the human IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 19.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 19.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D168G amino acid substitution mutation.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 20 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 20).
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D168G amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the human IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 20.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 20.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises D71 L and D168G amino acid substitution mutations.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 13 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 13).
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises D71 L and D168 amino acid substitution mutations and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the human IL- 22 polypeptide or fragment or variant thereof to SEQ ID NO: 13.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 13.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a R175S amino acid substitution mutation.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 21 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 21 ).
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a R175S amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the human IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 21 .
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 21.
In some embodiments, the macromolecule comprises two first binding sites comprising an IL-22 polypeptide or fragment or variant thereof that binds IL-22Ra, IL-22BP, and/or IL-10R2. In some embodiments, the two first binding sites each bind to IL-22Ra, each bind to IL-22BP, or each bind to IL- 10R2.
In some embodiments, the macromolecule comprises two second binding sites specific for an IL- 10 family address target expressed on the surface of the target cell. In some embodiments, the two second binding sites are specific for the same IL-10 family address target.
In some embodiments, the macromolecule is a homodimer comprising two subunits, each subunit comprising the first binding site and the second binding site. In some embodiments, the two subunits are fusion proteins. In some embodiments, each of the fusion proteins comprises a linker conjugated to the first binding site and the second binding site. In some embodiments, the linker is a GS linker. In some embodiments, the two subunits are substantially identical.
In another aspect, the disclosure provides a macromolecule comprising two subunits, each comprising a first binding site and a second binding site, wherein (a) each of the first binding sites comprises an IL-22 polypeptide or a fragment or variant thereof that binds an IL-22 receptor on the surface of a target cell, and (b) each of the second binding sites is specific for an IL-10 family address target provided in Table 1 that is expressed on the surface of the target cell; wherein: (i) the second binding sites localize the first binding sites to the IL-10 family address target such that the IL-22 polypeptides or fragments or variants thereof influence signaling by the IL-22 receptor in the target cell; (ii) the second binding sites do not substantially influence signaling upon binding the IL-10 family address targets; and (iii) the IL-22 polypeptides or fragments or variants thereof do not substantially influence signaling by the IL-22 receptor in the absence of localization by the second binding sites. In some embodiments, the two subunits are substantially identical.
In another aspect, the disclosure provides a macromolecule comprising two subunits, each comprising a first binding site and a second binding site, wherein (a) each of the first binding sites comprises an IL-22 polypeptide or a fragment or variant thereof that binds an IL-22 receptor on the surface of a target cell, and (b) each of the second binding sites is specific for an IL-10 family address target selected from CDH17, CDH16, and GP2 that is expressed on the surface of the target cell; wherein (i) the second binding sites localize the first binding sites to the IL-10 family address target such that the IL-22 polypeptides or fragments or variants thereof influence signaling by the IL-22 receptor in the target cell; (ii) the second binding sites do not substantially influence signaling upon binding the IL-10
family address targets; and (iii) the IL-22 polypeptides or fragments or variants thereof do not substantially influence signaling by the IL-22 receptor in the absence of localization by the second binding sites. In some embodiments, the two subunits are substantially identical.
In some embodiments, the macromolecule comprises a single first binding site. In some embodiments, the macromolecule comprises a single second binding site. In some embodiments, the macromolecule is a heterodimer comprising two subunits, wherein the first subunit comprises the first binding site and second subunit comprises the second binding site. In some embodiments, the macromolecule is a fusion protein. In some embodiments, the fusion protein comprises a linker conjugated to the first binding site and the second binding site. In some embodiments, the linker is a GS linker.
In some embodiments, binding of the second binding site to the IL-10 family address target (i) increases accumulation of the macromolecule in tissues expressing the IL-10 family address target, (ii) reduces accumulation of the macromolecule in tissues that do not express the IL-10 family address target, or (iii) both (i) and (ii).
In another aspect, the disclosure provides a method of modulating IL-22 receptor signaling in a target cell of a subject, the method comprising administering to the subject the macromolecule of any one of the above aspects.
In another aspect, the disclosure provides a macromolecule comprising (a) a first binding site comprising an IL-22 polypeptide or a fragment or variant thereof, and (b) a second binding site comprising an anti-CDH17 antibody or antigen-binding fragment thereof, wherein the macromolecule is a fusion protein, and wherein the first binding site and the second binding site are connected by a linker. In some embodiments, (i) the anti-CDH17 antibody or antigen-binding fragment thereof localizes the first binding site to an IL-22 receptor such that the IL-22 polypeptide or fragment or variant thereof influences signaling by the IL-22 receptor in the target cell; (ii) the anti-CDH17 antibody or antigen-binding fragment thereof does not substantially influence signaling upon binding CDH17; and (iii) the IL-22 polypeptide or fragment or variant thereof does not substantially influence signaling by the IL-22 receptor in the absence of localization by the anti-CDH17 antibody or antigen-binding fragment thereof.
In another aspect, the disclosure provides a macromolecule comprising two subunits, each comprising a first binding site and a second binding site, wherein (a) each first binding site comprises an IL-22 polypeptide or a fragment or variant thereof, and (b) each second binding site comprises an anti- CDH17 antibody or antigen-binding fragment thereof, wherein the macromolecule is a fusion protein, and wherein the first binding site and the second binding site of each subunit are connected by a linker. In some embodiments, (i) the anti-CDH17 antibodies or antigen-binding fragments thereof localize the first binding sites to an IL-22 receptor such that the IL-22 polypeptides or fragments or variants thereof influence signaling by the IL-22 receptor in the target cell; (ii) the anti-CDH17 antibodies or antigenbinding fragments thereof do not substantially influence signaling upon binding CDH17; and (iii) the IL-22 polypeptides or fragments or variants thereof do not substantially influence signaling by the IL-22 receptor in the absence of localization by the anti-CDH17 antibodies or antigen-binding fragments thereof.
In another aspect, the disclosure provides a method of modulating IL-22 receptor signaling in the small intestine or colon of a subject, the method comprising administering to the subject the macromolecule of either of the above two aspects.
In another aspect, the disclosure provides a macromolecule comprising (a) a first binding site comprising an IL-22 polypeptide or a fragment or variant thereof, and (b) a second binding site comprising an anti-CDH16 antibody or antigen-binding fragment thereof, wherein the macromolecule is a fusion protein, and wherein the first binding site and the second binding site are connected by a linker. In some embodiments, (i) the anti-CDH16 antibody or antigen-binding fragment thereof localizes the first binding site to an IL-22 receptor such that the IL-22 polypeptide or fragment or variant thereof influences signaling by the IL-22 receptor in the target cell; (ii) the anti-CDH16 antibody or antigen-binding fragment thereof does not substantially influence signaling upon binding CDH16; and (iii) the IL-22 polypeptide or fragment or variant thereof does not substantially influence signaling by the IL-22 receptor in the absence of localization by the anti-CDH16 antibody or antigen-binding fragment thereof.
In another aspect, the disclosure provides a macromolecule comprising two subunits, each comprising a first binding site and a second binding site, wherein (a) each first binding site comprises an IL-22 polypeptide or a fragment or variant thereof, and (b) each second binding site comprises an anti- CDH16 antibody or antigen-binding fragment thereof, wherein the macromolecule is a fusion protein, and wherein the first binding site and the second binding site of each subunit are connected by a linker. In some embodiments, (i) the anti-CDH16 antibodies or antigen-binding fragments thereof localize the first binding sites to an IL-22 receptor such that the IL-22 polypeptides or fragments or variants thereof influence signaling by the IL-22 receptor in the target cell; (ii) the anti-CDH16 antibodies or antigenbinding fragments thereof do not substantially influence signaling upon binding CDH16; and (iii) the IL-22 polypeptides or fragments or variants thereof do not substantially influence signaling by the IL-22 receptor in the absence of localization by the anti-CDH16 antibodies or antigen-binding fragments thereof.
In another aspect, the disclosure provides a method of modulating IL-22 receptor signaling in the kidney of a subject, the method comprising administering to the subject the macromolecule of either of the above two aspects.
In another aspect, the disclosure provides a macromolecule comprising (a) a first binding site comprising an IL-22 polypeptide or a fragment or variant thereof, and (b) a second binding site comprising an anti-GP2 antibody or antigen-binding fragment thereof, wherein the macromolecule is a fusion protein, and wherein the first binding site and the second binding site are connected by a linker. In some embodiments, (i) the anti-GP2 antibody or antigen-binding fragment thereof localizes the first binding site to an IL-22 receptor such that the IL-22 polypeptide or fragment or variant thereof influences signaling by the IL-22 receptor in the target cell; (ii) the anti-GP2 antibody or antigen-binding fragment thereof does not substantially influence signaling upon binding GP2; and (iii) the IL-22 polypeptide or fragment or variant thereof does not substantially influence signaling by the IL-22 receptor in the absence of localization by the anti-GP2 antibody or antigen-binding fragment thereof.
In another aspect, the disclosure provides a macromolecule comprising two subunits, each comprising a first binding site and a second binding site, wherein (a) each first binding site comprises an
IL-22 polypeptide or a fragment or variant thereof, and (b) each second binding site comprises an anti- GP2 antibody or antigen-binding fragment thereof, wherein the macromolecule is a fusion protein, and wherein the first binding site and the second binding site of each subunit are connected by a linker. In some embodiments, (i) the anti-GP2 antibodies or antigen-binding fragments thereof localize the first binding sites to an IL-22 receptor such that the IL-22 polypeptides or fragments or variants thereof influence signaling by the IL-22 receptor in the target cell; (ii) the anti-GP2 antibodies or antigen-binding fragments thereof do not substantially influence signaling upon binding GP2; and (iii) the IL-22 polypeptides or fragments or variants thereof do not substantially influence signaling by the IL-22 receptor in the absence of localization by the second binding sites.
In another aspect, the disclosure provides a method of modulating IL-22 receptor signaling in the pancreas of a subject, the method comprising administering to the subject the macromolecule of either one of the above aspects.
In some embodiments, the IL-22 polypeptide or fragment or variant thereof is a human IL-22 or fragment or variant thereof.
In some embodiments, the IL-22 polypeptide or fragment or variant thereof is a human IL-22 polypeptide or fragment or variant thereof and comprises one or more of the amino acid substitution mutations of Table 6, Table 7A, Table 7B, Table 8, Table 9A, Table 9B, or Table 9C.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises an amino acid substitution mutation of one or more of K61 , D71 , R73, D168, and R175. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a K61 amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D71 amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a R73 amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D168 amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a R175 amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D71 amino acid substitution mutation and a D168 amino acid substitution mutation. The amino acid positions are with reference to the IL-22 sequence of SEQ ID NO: 7.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises one or more of a K61 S, D71 N, D71 L, D71 G, R73Q, R73S, R73N, D168N, D168G, and R175S amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a K61 S amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D71 N amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D71 L amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D71 G amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a R73Q amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a R73S amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a R73N amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide
or fragment or variant thereof comprises a D168N amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D168G amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a R175S amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D71 L amino acid substitution mutation and a D168G amino acid substitution mutation. The amino acid position numbering is with reference to the IL- 22 sequence of SEQ ID NO: 7.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a K61 S amino acid substitution mutation.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 17 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 17).
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a K61 S amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the human IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 17.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 17.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D71 G amino acid substitution mutation.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 15 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 15).
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D71 G amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the human IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 15.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 15.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D71 L amino acid substitution mutation.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 11 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 11 ).
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D71 L amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the human IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 11 .
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 11.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D71 N amino acid substitution mutation.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 18 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 18).
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D71 N amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the human IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 18.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 18.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a R73S amino acid substitution mutation.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 16 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 16).
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a R73S amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the human IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 16.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 16.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a R73N amino acid substitution mutation.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 22 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 22).
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a R73N amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the human IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 22
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 22.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a R73Q amino acid substitution mutation.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 12 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 12).
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a R73Q amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%,
94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the human IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 12.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 12.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D168N amino acid substitution mutation.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 19 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 19).
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D168N amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the human IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 19.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 19.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D168G amino acid substitution mutation.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 20 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 20).
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D168G amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the human IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 20.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 20.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises D71 L and D168G amino acid substitution mutations.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 13 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 13).
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises D71 L and D168 amino acid substitution mutations and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the human IL- 22 polypeptide or fragment or variant thereof to SEQ ID NO: 13.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 13.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a R175S amino acid substitution mutation.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 21 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 21 ).
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a R175S amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the human IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 21 .
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 21.
In another aspect, the disclosure provides a macromolecule comprising a first binding site and a second binding site, wherein (a) the first binding site comprises an IL-22 polypeptide or a fragment or variant thereof that binds an IL-22 receptor on the surface of a target cell, and (b) the second binding site is specific for CDH17, e.g., that is expressed on the surface of the target cell; wherein (i) the second binding site localizes the first binding site to the CDH17 such that the IL-22 polypeptide or fragment or variant thereof influences signaling by the IL-22 receptor in the target cell; (ii) the second binding site does not substantially influence signaling upon binding the CDH17; and (iii) the IL-22 polypeptide or fragment or variant thereof does not substantially influence signaling by the IL-22 receptor in the absence of localization by the second binding site.
In some embodiments, the IL-22 polypeptide or fragment or variant thereof is a human IL-22 or fragment or variant thereof.
In some embodiments, the IL-22 polypeptide or fragment or variant thereof is a human IL-22 polypeptide or fragment or variant thereof and comprises one or more of the amino acid substitution mutations of Table 6, Table 7A, Table 7B, Table 8, Table 9A, Table 9B, or Table 9C.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises an amino acid substitution mutation of one or more of K61 , D71 , R73, D168, and R175. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a K61 amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D71 amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a R73 amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D168 amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a R175 amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D71 amino acid substitution mutation and a D168 amino acid substitution mutation. The amino acid positions are with reference to the IL-22 sequence of SEQ ID NO: 7.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises one or more of a K61 S, D71 N, D71 L, D71 G, R73Q, R73S, R73N, D168N, D168G, and R175S amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a K61 S amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D71 N amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D71 L amino acid
substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D71 G amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a R73Q amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a R73S amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a R73N amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D168N amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D168G amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a R175S amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D71 L amino acid substitution mutation and a D168G amino acid substitution mutation. The amino acid position numbering is with reference to the IL- 22 sequence of SEQ ID NO: 7.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a K61 S amino acid substitution mutation.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 17 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 17).
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a K61 S amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the human IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 17.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 17.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D71 G amino acid substitution mutation.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 15 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 15).
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D71 G amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the human IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 15.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 15.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D71 L amino acid substitution mutation.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 11 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 11 ).
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D71 L amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the human IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 11 .
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 11.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D71 N amino acid substitution mutation.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 18 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 18).
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D71 N amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the human IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 18.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 18.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a R73S amino acid substitution mutation.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 16 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 16).
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a R73S amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the human IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 16.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 16.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a R73N amino acid substitution mutation.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 22 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 22).
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a R73N amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the human IL-22 polypeptide or fragment or variant thereof to SEQ ID NO:22
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 22.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a R73Q amino acid substitution mutation.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 12 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 12).
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a R73Q amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the human IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 12.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 12.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D168N amino acid substitution mutation.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 19 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 19).
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D168N amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the human IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 19.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 19.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D168G amino acid substitution mutation.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 20 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 20).
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D168G amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the human IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 20.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 20.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises D71 L and D168G amino acid substitution mutations.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 13 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 13).
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises D71 L and D168 amino acid substitution mutations and has at least 90% identity (e.g., has at least 91%,
92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the human IL- 22 polypeptide or fragment or variant thereof to SEQ ID NO: 13.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 13.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a R175S amino acid substitution mutation.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 21 (e.g., has at least 91 %, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 21 ).
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a R175S amino acid substitution mutation and has at least 90% identity (e.g., has at least 91 %, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the human IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 21 .
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 21 .
In some embodiments of a macromolecule provided herein, the IL-22 polypeptide (e.g., human IL-22 polypeptide) or fragment or variant thereof has a melting onset temperature (Tonset) of about 36°C to about 60 °C, e.g., as determined by a method described herein, e.g., as determined by differential scanning calorimetry (DSC).
In some embodiments of a macromolecule provided herein, the IL-22 polypeptide (e.g., human IL-22 polypeptide) or fragment or variant thereof has a melting onset temperature (Tonset) of about 38°C, e.g., as determined by a method described herein, e.g., as determined by differential scanning calorimetry (DSC).
In some embodiments of a macromolecule provided herein, the IL-22 polypeptide (e.g., human IL-22 polypeptide) or fragment or variant thereof has a melting onset temperature (Tonset) of about 50°C, e.g., as determined by a method described herein, e.g., as determined by differential scanning calorimetry (DSC).
In some embodiments of a macromolecule provided herein, the IL-22 polypeptide (e.g., human IL-22 polypeptide) or fragment or variant thereof has a melting onset temperature (Tonset) of about 58°C, e.g., as determined by a method described herein, e.g., as determined by differential scanning calorimetry (DSC).
In some embodiments of a macromolecule provided herein, the IL-22 polypeptide (e.g., human IL-22 polypeptide) or fragment or variant thereof has a binding affinity to human IL-22Ra of between about 1 E-8 to about 10E-9, e.g., as determined by a method described herein.
In some embodiments of a macromolecule provided herein, the IL-22 polypeptide (e.g., human IL-22 polypeptide) or fragment or variant thereof has a binding affinity to human IL-22Ra of about 7.7E-9, e.g., as determined by a method described herein.
In some embodiments of a macromolecule provided herein, the IL-22 polypeptide (e.g., human IL-22 polypeptide) or fragment or variant thereof has a binding affinity to human IL-22Ra of about 2.3E-8, e.g., as determined by a method described herein.
In some embodiments of a macromolecule provided herein, the IL-22 polypeptide (e.g., human IL-22 polypeptide) or fragment or variant thereof has a binding affinity to human IL-22Ra of about 1 .5E - 8, e.g., as determined by a method described herein.
In some embodiments of a macromolecule provided herein, the IL-22 polypeptide (e.g., human IL-22 polypeptide) or fragment or variant thereof has a binding affinity to human IL-22Ra of about 7.7E-8, e.g., as determined by a method described herein.
In some embodiments of a macromolecule provided herein, the IL-22 polypeptide (e.g., human IL-22 polypeptide) or fragment or variant thereof has a binding affinity to human IL-22Ra of about 10E-9, e.g., as determined by a method described herein.
In some embodiments of a macromolecule provided herein, the IL-22 polypeptide (e.g., human IL-22 polypeptide) or fragment or variant thereof has a binding affinity to human IL-22Ra of about 3.1 E-8, e.g., as determined by a method described herein.
In some embodiments of a macromolecule provided herein, the IL-22 polypeptide (e.g., human IL-22 polypeptide) or fragment or variant thereof has about a 1 .3-fold to about a10-fold decreased binding affinity to human IL-22Ra as compared to the binding affinity of wild type human IL-22 polypeptide to human IL-22Ra, e.g., as determined by a method described herein.
In some embodiments of a macromolecule provided herein, the IL-22 polypeptide (e.g., human IL-22 polypeptide) or fragment or variant thereof has about a 3-fold decreased binding affinity to human IL-22Ra as compared to the binding affinity of wild type human IL-22 polypeptide to human IL-22Ra, e.g., as determined by a method described herein.
In some embodiments of a macromolecule provided herein, the IL-22 polypeptide (e.g., human IL-22 polypeptide) or fragment or variant thereof has about a 2-fold decreased binding affinity to human IL-22Ra as compared to the binding affinity of wild type human IL-22 polypeptide to human IL-22Ra, e.g., as determined by a method described herein.
In some embodiments of a macromolecule provided herein, the IL-22 polypeptide (e.g., human IL-22 polypeptide) or fragment or variant thereof has about a 10-fold decreased binding affinity to human IL-22Ra as compared to the binding affinity of wild type human IL-22 polypeptide to human IL-22Ra, e.g., as determined by a method described herein.
In some embodiments of a macromolecule provided herein, the IL-22 polypeptide (e.g., human IL-22 polypeptide) or fragment or variant thereof has about a 1 .3-fold decreased binding affinity to human IL-22Ra as compared to the binding affinity of wild type human IL-22 polypeptide to human IL-22Ra, e.g., as determined by a method described herein.
In some embodiments of a macromolecule provided herein, the IL-22 polypeptide (e.g., human IL-22 polypeptide) or fragment or variant thereof has about a 4-fold decreased binding affinity to human IL-22Ra as compared to the binding affinity of wild type human IL-22 polypeptide to human IL-22Ra, e.g., as determined by a method described herein.
In one aspect, the disclosure provides an IL-22 polypeptide or a fragment or variant thereof (e.g., an IL-22 mutein polypeptide or fragment or variant thereof, e.g., an IL-22 mutein polypeptide or fragment or variant thereof that is derived from human IL-22) that binds an IL-22 receptor on the surface of a target cell.
In some embodiments, the IL-22 polypeptide or a fragment or variant thereof binds the IL-22 receptor. In some embodiments, the IL-22 polypeptide or fragment or variant thereof binds the human IL-22 receptor. In some embodiments, the IL-22 polypeptide or fragment or variant thereof binds human IL-22Ra and/or IL-10R2.
In some embodiments, the IL-22 polypeptide or fragment or variant thereof binds IL-22BP. In some embodiments, the IL-22 polypeptide or fragment or variant thereof binds human IL-22BP.
In some embodiments, the IL-22 polypeptide or fragment or variant thereof agonizes the IL-22 receptor. In some embodiments, the IL-22 polypeptide or fragment or variant thereof agonizes the human IL-22 receptor. In some embodiments, the IL-22 polypeptide or fragment or variant thereof agonizes human IL-22Ra and/or IL-10R2, e.g., in an assay described herein.
In some embodiments, the IL-22 polypeptide or fragment or variant thereof agonizes IL-22BP. In some embodiments, the IL-22 polypeptide or fragment or variant thereof agonizes human IL-22BP.
In some embodiments, the IL-22 polypeptide or fragment or variant thereof has low (e.g., lower) binding affinity for IL-22Ra and/or IL-10R2, as compared to the binding affinity of wild type human IL-22 (e.g., of SEQ ID NO: 8) for the IL-22 receptor, e.g., in an assay described herein.
In some embodiments, the IL-22 polypeptide or fragment or variant thereof has low (e.g., lower) binding affinity for IL-22BP, as compared to the binding affinity of wild type human IL-22 (e.g., of SEQ ID NO: 8) for IL-2BP, e.g., in an assay described herein.
In some embodiments, the IL-22 polypeptide or fragment or variant thereof has been engineered to have reduced binding affinity for the IL-22 receptor (e.g., for one or both subunits of the receptor, e.g., IL-22Ra and/or IL-10R2), e.g., as compared to the binding affinity of wild type human IL-22 (e.g., of SEQ ID NO: 8) for the IL-22 receptor, e.g., in an assay described herein. For example, in some embodiments, the binding affinity for the IL-22 receptor of the engineered IL-22 polypeptide or fragment or variant thereof ranges from minimally denatured (e.g., <10-fold decreased) to hundreds of fold weakened, e.g., at least 300x decreased, e.g., as measured by biolayer interferometry (BLI), e.g., on a GATOR® instrument. See also the examples provided herein.
In some embodiments, the IL-22 polypeptide or fragment or variant thereof comprises one or more amino acid substitution mutations at one or more residues that are predicted to be interaction surfaces between IL-22 and IL-22Ra and/or IL-10R2.
In some embodiments, the IL-22 polypeptide or fragment or variant thereof has reduced binding affinity for IL22RA1 (also referred to as IL-22RA or IL-22Ra) relative to a wild-type IL-22 or fragment thereof.
In some embodiments, the IL-22 polypeptide or fragment or variant thereof comprises one or more amino acid substitution mutations at one or more residues that are predicted to be interaction surfaces between IL-22 and IL-22Ra.
In some embodiments, the IL-22 polypeptide or fragment or variant thereof is a human IL-22 or fragment or variant thereof.
In some embodiments, the IL-22 polypeptide or fragment or variant thereof is a human IL-22 or fragment or variant thereof and comprises one or more of the amino acid substitution mutations of Table 6, Table 7A, Table 7B, Table 8, Table 9A, Table 9B, or Table 9C.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises an amino acid substitution mutation of one or more of K61 , D71 , R73, D168, and R175. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a K61 amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D71 amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a R73 amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D168 amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a R175 amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D71 amino acid substitution mutation and a D168 amino acid substitution mutation. The amino acid position numbering is with reference to the IL-22 sequence of SEQ ID NO: 7.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises one or more of a K61 S, D71 N, D71 L, D71 G, R73Q, R73S, R73N, D168N, D168G, and R175S amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a K61 S amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D71 N amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D71 L amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D71 G amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a R73Q amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a R73S amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a R73N amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D168N amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D168G amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a R175S amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D71 L amino acid substitution mutation and a D168G amino acid substitution mutation. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D71 G amino acid substitution mutation and a D168G amino acid substitution mutation. The amino acid position numbering is with reference to the IL-22 sequence of SEQ ID NO: 7.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a K61 S amino acid substitution mutation.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 17 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 17).
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a K61 S amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%,
94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the human IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 17.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 17.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D71 G amino acid substitution mutation.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 15 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 15).
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D71 G amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the human IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 15.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 15.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D71 L amino acid substitution mutation.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 11 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 11 ).
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D71 L amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the human IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 11 .
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 11.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D71 N amino acid substitution mutation.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 18 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 18).
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D71 N amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the human IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 18.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 18.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a R73S amino acid substitution mutation.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 16 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 16).
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a R73S amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the human IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 16.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 16.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a R73N amino acid substitution mutation.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 22 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 22).
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a R73N amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the human IL-22 polypeptide or fragment or variant thereof to SEQ ID NO:22
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 22.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a R73Q amino acid substitution mutation.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 12 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 12).
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a R73Q amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the human IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 12.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 12.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D168N amino acid substitution mutation.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 19 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 19).
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D168N amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the human IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 19.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 19.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D168G amino acid substitution mutation.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 20 (e.g., has at least 91 %, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 20).
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a D168G amino acid substitution mutation and has at least 90% identity (e.g., has at least 91 %, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the human IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 20.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 20.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises D71 L and D168G amino acid substitution mutations.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 13 (e.g., has at least 91 %, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 13).
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises D71 L and D168 amino acid substitution mutations and has at least 90% identity (e.g., has at least 91 %, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the human IL- 22 polypeptide or fragment or variant thereof to SEQ ID NO: 13.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 13.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a R175S amino acid substitution mutation.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 21 (e.g., has at least 91 %, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 21 ).
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises a R175S amino acid substitution mutation and has at least 90% identity (e.g., has at least 91 %, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the human IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 21 .
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 21 .
In some embodiments of an IL-22 polypeptide or fragment or variant thereof provided herein, the IL-22 polypeptide or fragment or variant thereof has a melting onset temperature (Tonset) of about 36°C to about 60 °C, e.g., as determined by a method described herein, e.g., as determined by differential scanning calorimetry (DSC).
In some embodiments of an IL-22 polypeptide or fragment or variant thereof provided herein, the IL-22 polypeptide or fragment or variant thereof has a melting onset temperature (Tonset) of about 38°C, e.g., as determined by a method described herein, e.g., as determined by differential scanning calorimetry (DSC).
In some embodiments of an IL-22 polypeptide or fragment or variant thereof provided herein, the IL-22 polypeptide or fragment or variant thereof has a melting onset temperature (Tonset) of about 50°C, e.g., as determined by a method described herein, e.g., as determined by differential scanning calorimetry (DSC).
In some embodiments of an IL-22 polypeptide or fragment or variant thereof provided herein, the IL-22 polypeptide or fragment or variant thereof has a melting onset temperature (Tonset) of about 58°C, e.g., as determined by a method described herein, e.g., as determined by differential scanning calorimetry (DSC).
In some embodiments of an IL-22 polypeptide or fragment or variant thereof provided herein, the IL-22 polypeptide or fragment or variant thereof has a binding affinity to human IL-22Ra of between about 1 E-8 to about 10E-9, e.g., as determined by a method described herein.
In some embodiments of a macromolecule provided herein, the IL-22 polypeptide or fragment or variant thereof has a binding affinity to IL-22Ra of about 7.7E-9, e.g., as determined by a method described herein.
In some embodiments of an IL-22 polypeptide or fragment or variant thereof provided herein, the IL-22 polypeptide or fragment or variant thereof has a binding affinity to IL-22Ra of about 2.3E-8, e.g., as determined by a method described herein.
In some embodiments of an IL-22 polypeptide or fragment or variant thereof provided herein, the IL-22 polypeptide or fragment or variant thereof has a binding affinity to IL-22Ra of about 1 .5E -8, e.g., as determined by a method described herein.
In some embodiments of an IL-22 polypeptide or fragment or variant thereof provided herein, the IL-22 polypeptide or fragment or variant thereof has a binding affinity to human IL-22Ra of about 7.7E-8, e.g., as determined by a method described herein.
In some embodiments of an IL-22 polypeptide or fragment or variant thereof provided herein, the IL-22 polypeptide or fragment or variant thereof has a binding affinity to human IL-22Ra of about 10E-9, e.g., as determined by a method described herein.
In some embodiments of an IL-22 polypeptide or fragment or variant thereof provided herein, the IL-22 polypeptide or fragment or variant thereof has a binding affinity to human IL-22Ra of about 3.1 E-8, e.g., as determined by a method described herein.
In some embodiments of an IL-22 polypeptide or fragment or variant thereof provided herein, the IL-22 polypeptide or fragment or variant thereof has about a 1 .3-fold to about a10-fold decreased binding affinity to human IL-22Ra as compared to the binding affinity of wild type human IL-22 polypeptide to human IL-22Ra, e.g., as determined by a method described herein.
In some embodiments of an IL-22 polypeptide or fragment or variant thereof provided herein, the IL-22 polypeptide or fragment or variant thereof has about a 3-fold decreased binding affinity to human IL-22Ra as compared to the binding affinity of wild type human IL-22 polypeptide to human IL-22Ra, e.g., as determined by a method described herein.
In some embodiments of an IL-22 polypeptide or fragment or variant thereof provided herein, the IL-22 polypeptide or fragment or variant thereof has about a 2-fold decreased binding affinity to human IL-22Ra as compared to the binding affinity of wild type human IL-22 polypeptide to human IL-22Ra, e.g., as determined by a method described herein.
In some embodiments of an IL-22 polypeptide or fragment or variant thereof provided herein, the IL-22 polypeptide or fragment or variant thereof has about a 10-fold decreased binding affinity to human IL-22Ra as compared to the binding affinity of wild type human IL-22 polypeptide to human IL-22Ra, e.g., as determined by a method described herein.
In some embodiments of an IL-22 polypeptide or fragment or variant thereof provided herein, the IL-22 polypeptide or fragment or variant thereof has about a 1 .3-fold decreased binding affinity to human IL-22Ra as compared to the binding affinity of wild type human IL-22 polypeptide to human IL-22Ra, e.g., as determined by a method described herein.
In some embodiments of an IL-22 polypeptide or fragment or variant thereof provided herein, the IL-22 polypeptide or fragment or variant thereof has about a 4-fold decreased binding affinity to human IL-22Ra as compared to the binding affinity of wild type human IL-22 polypeptide to human IL-22Ra, e.g., as determined by a method described herein.
In another aspect, the disclosure provides a polypeptide comprising a human IL-22 polypeptide or fragment or variant thereof provided herein.
In another aspect, the disclosure provides a composition comprising any one of the polypeptides provided herein. In some embodiments, the composition comprises a pharmaceutically acceptable carrier or excipient. In some embodiments, the composition is a unit dosage form suitable for administration to a human subject.
In another aspect, the disclosure provides one or more nucleic acids encoding any one of the polypeptides provided herein. In some embodiments, the nucleic acids are linear nucleic acids, circular nucleic acids, and/or modified nucleic acids.
In another aspect, the disclosure provides one or more vectors comprising the one or more nucleic acids of the above aspect.
In another aspect, the disclosure provides one or more host cells comprising the one or more vectors of the above aspect.
In another aspect, the disclosure provides a method of producing any one of the polypeptides provided herein, the method comprising culturing the one or more host cells of the above aspect in a culture medium. In some embodiments, the method further comprises recovering the polypeptide from the one or more host cells or the culture medium.
In another aspect, the disclosure provides a polypeptide comprising an IL-22 polypeptide or a fragment or variant thereof provided herein, and further comprises an Fc region. In some embodiments, the Fc region is N terminal to the IL-22 polypeptide or fragment or variant thereof. In some embodiments, the Fc region is C terminal to the IL-22 polypeptide or fragment or variant thereof.
In another aspect, the disclosure provides a composition comprising any one of the macromolecules provided herein. In some embodiments, the composition comprises a pharmaceutically acceptable carrier or excipient. In some embodiments, the composition is a unit dosage form suitable for administration to a human subject.
In another aspect, the disclosure provides one or more nucleic acids encoding any one of the macromolecules provided herein or one or more subunits thereof. In some embodiments, the nucleic acids are linear nucleic acids, circular nucleic acids, and/or modified nucleic acids.
In another aspect, the disclosure provides one or more vectors comprising the one or more nucleic acids of the above aspect.
In another aspect, the disclosure provides one or more host cells comprising the one or more vectors of the above aspect.
In another aspect, the disclosure provides a method of producing any one of the macromolecules provided herein, the method comprising culturing the one or more host cells of the above aspect in a culture medium. In some embodiments, the method further comprises recovering the macromolecule from the one or more host cells or the culture medium.
In another aspect, the disclosure provides a method comprising administering a therapeutically effective amount of any one of the macromolecules, compositions, polypeptides, or nucleic acids provided herein to a subject in need thereof. In some embodiments, the subject has, is suspected of having, or is at increased risk of developing a disease or disorder selected from: Crohn's disease, ulcerative colitis, graft-vs-host disease, atopic dermatitis, psoriasis, wounds, diabetic skin ulcers, acute pancreatitis, chronic pancreatitis, autoimmune pancreatitis, nephritis, acute lung injury, viral lung injury, fibrosis, non-alcoholic steatohepatitis, viral hepatitis, and autoimmune hepatitis. In some embodiments, the disease or disorder comprises Crohn's disease. In some embodiments, the disease or disorder comprises ulcerative colitis.
In another aspect, the disclosure provides a pharmaceutical composition comprising a pharmaceutically acceptable excipient and a macromolecule, a composition, a polypeptide, or a nucleic acid of any one of the above aspects. The term “pharmaceutically acceptable excipient” as used herein means a pharmaceutically acceptable material, or vehicle, such as a liquid or solid filter, diluent, excipient, solvent or encapsulating material useful for formulating a macromolecule complex, macromolecule, nucleic acid, or pair of nucleic acids for medicinal or therapeutic use.
In another aspect, the disclosure provides the use of one or more of the macromolecule complexes, macromolecules, compositions, pharmaceutical compositions, polypeptides, or nucleic acids of any one of the above aspects in the manufacture of a medicament for the treatment of a subject. In some embodiments, the subject has, is suspected of having, or is at increased risk of developing a disease or disorder selected from: Crohn's disease, ulcerative colitis, graft-vs-host disease, atopic dermatitis, psoriasis, wounds, diabetic skin ulcers, acute pancreatitis, chronic pancreatitis, autoimmune pancreatitis, nephritis, acute lung injury, viral lung injury, fibrosis, non-alcoholic steatohepatitis, viral hepatitis, and autoimmune hepatitis. In some embodiments, the disease or disorder comprises Crohn's disease. In some embodiments, the disease or disorder comprises ulcerative colitis.
In another aspect, the disclosure provides one or more of the macromolecule complexes, macromolecules, compositions, pharmaceutical compositions, polypeptides, or nucleic acids of any one of the above aspects for use in treating a subject. In some embodiments, the subject has, is suspected of having, or is at increased risk of developing a disease or disorder selected from: Crohn's disease, ulcerative colitis, graft-vs-host disease, atopic dermatitis, psoriasis, wounds, diabetic skin ulcers, acute
pancreatitis, chronic pancreatitis, autoimmune pancreatitis, nephritis, acute lung injury, viral lung injury, fibrosis, non-alcoholic steatohepatitis, viral hepatitis, and autoimmune hepatitis. In some embodiments, the disease or disorder comprises Crohn's disease. In some embodiments, the disease or disorder comprises ulcerative colitis.
The details of one or more embodiments of the invention are set forth in the description below. Other features or advantages of the present invention will be apparent from the following drawings and detailed description of several embodiments, and also from the appended claims.
BRIEF DESCRIPTION OF THE DRAWINGS
Fig. 1 A is a schematic diagram showing the design of a monovalent ANDbody that simultaneously binds to an IL-22 receptor and an antibody target (“monovalent IL-22/mAb”). Fab: fragment antigen-binding region.
Fig. 1B is a schematic diagram showing the design of a divalent ANDbody that simultaneously binds to an IL-22 receptor and an antibody target (“divalent IL-22/mAb”).
Fig. 2 is a chart showing the concentration of the indicated ANDbodies and control monoclonal antibodies (mAbs) in serum from treated mice over time.
Fig. 3A is a plot showing the level of a monovalent anti-mCDH17/anti-IL-22 ANDbody (ng/mg total protein) in the indicated tissue samples from treated mice at the indicated timepoints.
Fig. 3B is a plot showing the level of a monovalent anti-RSV/anti-IL-22 ANDbody (ng/mg total protein) in the indicated tissue samples from treated mice at the indicated timepoints.
Fig. 4A is a set of bar graphs showing the levels of the IL-22 target genes REG3B, REG3G and SOCS3 in small intestine tissue from mice treated with monovalent anti-mCDH17/IL-22 and monovalent anti-RSV/IL-22 ANDbodies at the indicated concentrations (fold change relative to phosphate-buffered saline (PBS) control).
Fig. 4B is a set of bar graphs showing the levels of the IL-22 target genes REG3B, REG3G and SOCS3 in colon tissue from mice treated with monovalent anti-mCDH17/IL-22 and monovalent anti- RSV/IL-22 ANDbodies at the indicated concentrations (fold change relative to PBS control).
Fig. 4C is a set of bar graphs showing the levels of the IL-22 target genes REG3B, REG3G and SOCS3 in pancreas tissue from mice treated with monovalent anti-mCDH17/IL-22 and monovalent anti- RSV/IL-22 ANDbodies at the indicated concentrations (fold change relative to PBS control).
Fig. 4D is a set of bar graphs showing the levels of the IL-22 target genes REG3B, REG3G and SOCS3 in liver tissue from mice treated with monovalent anti-mCDH17/IL-22 and monovalent anti- RSV/IL-22 ANDbodies at the indicated concentrations (fold change relative to PBS control).
Fig. 5 is a chart showing the concentration of ANDbodies comprising the indicated amino acid substitution mutations in the IL-22 polypeptide in serum from treated mice over time.
Fig. 6A is a set of bar graphs showing the levels of the IL-22 target genes REG3B, REG3G and SOCS3 in small intestine tissue from mice treated with divalent anti-mCDH17/IL-22 and divalent anti- RSV/IL-22 ANDbodies comprising the indicated amino acid substitution mutations in the IL-22 polypeptide at the indicated concentrations (fold change relative to PBS control).
Fig. 6B is a set of bar graphs showing the levels of the IL-22 target genes REG3B, REG3G and
SOCS3 in colon tissue from mice treated with divalent anti-mCDH17/IL-22 and divalent anti-RSV/IL-22
ANDbodies comprising the indicated amino acid substitution mutations in the IL-22 polypeptide at the indicated concentrations (fold change relative to PBS control).
Fig. 6C is a set of bar graphs showing the levels of the IL-22 target genes REG3B, REG3G and SOCS3 in pancreas tissue from mice treated with divalent anti-mCDH17/IL-22 and divalent anti-RSV/IL- 22 ANDbodies comprising the indicated amino acid substitution mutations in the IL-22 polypeptide at the indicated concentrations (fold change relative to PBS control).
Fig. 6D is a set of bar graphs showing the levels of the IL-22 target genes REG3B, REG3G and SOCS3 in liver tissue from mice treated with divalent anti-mCDH17/IL-22 and divalent anti-RSV/IL-22 ANDbodies comprising the indicated amino acid substitution mutations in the IL-22 polypeptide at the indicated concentrations (fold change relative to PBS control).
Fig. 7A is a set of plots showing the levels of the IL-22 target genes REG3G, REG3B, and SOCS3 in small intestine tissue from mice treated with divalent anti-mCDH17/IL-22 or divalent anti- RSV/IL-22 ANDbodies comprising native IL-22 or IL-22 comprising the indicated amino acid substitution mutations at the indicated concentrations (fold change relative to PBS control).
Fig. 7B is a set of plots showing the levels of the IL-22 target genes REG3G, REG3B, and SOCS3 in colon tissue from mice treated with divalent anti-mCDH17/IL-22 or divalent anti-RSV/IL-22 ANDbodies comprising native IL-22 or IL-22 comprising the indicated amino acid substitution mutations at the indicated concentrations (fold change relative to PBS control).
Fig. 7C is a set of plots showing the levels of the IL-22 target genes REG3G, REG3B, and SOCS3 in pancreas tissue from mice treated with divalent anti-mCDH17/IL-22 or divalent anti-RSV/IL-22 ANDbodies comprising native IL-22 or IL-22 comprising the indicated amino acid substitution mutations at the indicated concentrations (fold change relative to PBS control).
Fig. 7D is a set of plots showing the levels of the IL-22 target genes MT1 and MT2 in liver tissue from mice treated with divalent anti-mCDH17/IL-22 or divalent anti-RSV/IL-22 ANDbodies comprising native IL-22 or IL-22 comprising the indicated amino acid substitution mutations at the indicated concentrations (fold change relative to PBS control).
Fig. 8A is a plot showing receptor occupancy for a range of binding affinities of IL-22 and CDH17 in the disease compartment of a four-compartment model.
Fig. 8B is a plot showing receptor occupancy for a range of binding affinities of IL-22 and CDH17 in the toxicity (tox) compartment of a four-compartment model.
Fig. 9A is a plot showing the induction levels of the IL-22 target genes REG3G and REG3B in pancreas tissue from mice treated with divalent anti-mGP2/IL-22 or divalent anti-RSV/IL-22 ANDbody proteins comprising native IL-22 or IL-22 mutein (D71 L / D168G). Expression was quantified as foldchange relative to PBS control. ANDbodies were administered at 0.3, 0.1 , or 0.03 milligrams per kilogram of body weight (MPK), as illustrated by the triangular concentration ramps.
Fig. 9B is a plot showing the induction levels of the IL-22 target genes REG3G and REG3B in small intestine tissue from mice treated with divalent anti-mGP2/IL-22 or divalent anti-RSV/IL-22 ANDbody proteins comprising native IL-22 or IL-22 mutein (D71 L / D168G). Expression was quantified as fold-change relative to PBS control. ANDbodies were administered at 0.3, 0.1 , or 0.03 MPK, as illustrated by the triangular concentration ramps.
Fig. 9C is a plot showing the induction levels of the IL-22 target genes REG3G and REG3B in colon tissue from mice treated with divalent anti-mGP2/IL-22 or divalent anti-RSV/IL-22 ANDbody proteins comprising native IL-22 or IL-22 mutein (D71 L / D168G). Expression was quantified as foldchange relative to PBS control. ANDbodies were administered at 0.3, 0.1 , or 0.03 MPK, as illustrated by the triangular concentration ramps.
Fig. 10A is a plot showing the change in body weight of mice during a dextran sulfate sodium (DSS)-induced acute colitis study. Mice were administered DSS in drinking water as indicated, and ANDbody molecules or murine IL-22-Fc (mulL22-Fc) were administered intraperitoneally (I.P.) at days -1 , 1 , 4, and 6. Body weight was recorded daily, and plotted as % change in body weight.
Fig. 10B is a plot showing the disease activity index (DAI) of mice during the DSS-induced acute colitis study described in Fig. 10A. DAI was composed as body weight change, diarrhea, and hematochezia.
Fig. 11 A is a graph showing the thermal stability (as determined by differential scanning calorimetry (DSC)) of an IL-22 mutein polypeptide comprising D71 L and D168G amino acid substitution mutations.
Fig. 11B is a graph showing the thermal stability (as determined by DSC) of an IL-22 mutein polypeptide comprising an R73Q amino acid substitution mutation.
Fig. 11C is a graph showing the thermal stability (as determined by DSC) of an IL-22 mutein polypeptide comprising an R73S amino acid substitution mutation.
Fig. 12 is a graph showing levels of phosphorylated STAT3 (pSTAT3) on HaCaT (human keratinocyte) cells that were treated with native IL-22 or IL-22 mutein polypeptides comprising D71 L / D168G, R73Q, or R73S amino acid substitution mutations.
DETAILED DESCRIPTION OF THE INVENTION
Provided herein are ANDbody™ molecules that include a therapeutic IL-22 polypeptide or fragment or variant thereof and an IL-10 family address target binding domain. In some aspects, the therapeutic IL-22 polypeptide or fragment or variant thereof on the ANDbody molecule productively engages its therapeutic effector target only if the IL-10 family address target binding domain also engages an IL-10 family address target (i.e., a cell surface moiety displayed on the surface of a cell that also expresses the IL-22 receptor) on a target tissue or cell to localize the IL-22 polypeptide or fragment or variant thereof to the targeted cell or tissue, e.g., to form an AND-gate type of logic gate. For example, in some embodiments, an ANDbody is a macromolecule comprising at least (a) a first binding site comprising an IL-22 polypeptide or fragment or variant thereof that binds an IL-22 receptor on the surface of a target cell (e.g., wherein the IL-10 family receptor is broadly expressed in a mammalian subject); and (b) a second binding site specific for an IL-10 family address target. In embodiments, expression of the address target is restricted in vivo in a subject. In some embodiments, the binding of a first binding site to a therapeutic effector target is weaker than the binding of the second binding site to the address marker. In some embodiments, the IL-22 receptor and the IL-10 family address target are on the same cell.
Also provided herein are IL-22 mutein polypeptides or fragments or variants thereof that have altered (e.g., decreased) affinity for the IL-22 receptor.
A. ANDbody Structure and Function
In general, an ANDbody can be any macromolecule, such as a polypeptide or protein, that contains both an effector target binding site or binding domain, and an address target binding site or binding domain. The binding sites may be present on the same polypeptide chain or different polypeptide chains that are linked together, e.g., through disulfide bonds.
In another aspect, the disclosure provides a macromolecule (i.e., an ANDbody) comprising a first binding site and a second binding site, wherein (a) the first binding site comprises an IL-22 polypeptide or a fragment or variant thereof that binds an IL-22 receptor on the surface of a target cell, and (b) the second binding site is specific for an IL-10 family address target provided in Table 1 that is expressed on the surface of the target cell; wherein (i) the second binding site localizes the first binding site to the IL- 10 family address target such that the IL-22 polypeptide or fragment or variant thereof influences signaling by the IL-22 receptor in the target cell; (ii) the second binding site does not substantially influence signaling upon binding the IL-10 family address target; and (iii) the IL-22 polypeptide or fragment or variant thereof does not substantially influence signaling by the IL-22 receptor in the absence of localization by the second binding site.
In another aspect, the disclosure provides a macromolecule (i.e., an ANDbody) comprising a first binding site and a second binding site, wherein (a) the first binding site comprises an IL-22 polypeptide or a fragment or variant thereof that binds an IL-22 receptor on the surface of a target cell, and (b) the second binding site is specific for an IL-10 family address target selected from CDH17, CDH16, and GP2 that is expressed on the surface of the target cell; wherein (i) the second binding site localizes the first binding site to the IL-10 family address target such that the IL-22 polypeptide or fragment or variant thereof influences signaling by the IL-22 receptor in the target cell; (ii) the second binding site does not substantially influence signaling upon binding the IL-10 family address target; and (iii) the IL-22 polypeptide or fragment or variant thereof does not substantially influence signaling by the IL-22 receptor in the absence of localization by the second binding site.
An ANDbody, as disclosed herein, can be linked to an additional moiety or moieties, e.g., an extracellular component, an intracellular component, a soluble factor (e.g., an enzyme, hormone, cytokine, growth factor, toxin, venom, pollutant, etc.), or a transmembrane protein (e.g., a cell surface receptor).
In some embodiments, the first binding site and second binding site in the ANDbody are directly joined to each other. By directly joined is meant that the first binding site coding sequences abut the second binding site coding sequences and no sequences derived from other sequences (such as linkers) are present. In other embodiments, the first binding site and second binding site in the ANDbody are not directly joined to each other.
In some embodiments, ANDbodies of the present technology include binding domains that bind address target or effector target proteins. In embodiments, binding domains of the present ANDbodies may bind protein sequences that include a signal peptide. In other embodiments, binding domains of the present ANDbodies may bind proteins that lack a signal protein. In some embodiments, binding domains of the present ANDbodies may bind full-length proteins. In other embodiments, binding domains of the present ANDbodies may bind protein fusions, such as full-length protein sequences, or peptide
fragments thereof, with or without signal peptide regions, fused to other proteins, such as, for example, Fc sequences. Binding domains of the present ANDbodies may bind proteins that comprise less than the full-length protein sequence, such as a peptide fragment of the address target or effector target.
/. Divalent ANDbody format
In some aspects, the macromolecule comprises two first binding sites comprising an IL-22 polypeptide or a fragment or variant thereof that binds an IL-22 receptor. In some embodiments, the two first binding sites bind to the same subunit or subunits of an IL-22 family receptor (e.g., both bind to IL- 22Ra, both bind to IL-22BP, or both bind to IL-10R2). For example, in some embodiments, the two first binding sites comprise the same IL-22 polypeptide or fragment or variant thereof and/or are substantially identical in sequence. In other embodiments, the two first binding sites comprise different fragments or variants of the same IL-22 polypeptide or fragment or variant thereof, e.g., comprise fragments or variants having different affinity for the IL-22 receptor. Alternatively, the two first binding sites may comprise different IL-22 polypeptides or fragments or variants thereof and/or bind to different subunits of an IL-22 receptor.
In some aspects, the macromolecule comprises two second binding sites specific for an IL-10 family address target expressed on the surface of the target cell. In some embodiments, the two second binding sites are specific for the same IL-10 family address target. For example, in some embodiments, the two second binding sites are substantially identical in sequence. Alternatively, the two second binding sites may bind to two different IL-10 family address targets.
Accordingly, in some aspects, the macromolecule is a homodimer comprising two subunits, each subunit comprising the first binding site and the second binding site (e.g., wherein the two subunits of the homodimer are substantially identical (e.g., are substantially identical in sequence)). In some embodiments, each subunit of the homodimer is a fusion protein (e.g., a fusion protein comprising a linker (e.g., a GS linker) conjugated to the first binding site and the second binding site).
In some aspects, the disclosure provides a macromolecule comprising two subunits, each comprising a first binding site and a second binding site, wherein: (a) each of the first binding sites comprises an IL-22 polypeptide or a fragment or variant thereof that binds an IL-22 receptor on the surface of a target cell, and (b) each of the second binding sites is specific for an IL-10 family address target provided in Table 1 that is expressed on the surface of the target cell; wherein (i) the second binding sites localize the first binding sites to the IL-10 family address target such that the IL-22 polypeptides or fragments or variants thereof influence signaling by the IL-22 receptor in the target cell; (ii) the second binding sites do not substantially influence signaling upon binding the IL-10 family address targets; and (Hi) the IL-22 polypeptides or fragments or variants thereof do not substantially influence signaling by the IL-22 receptor in the absence of localization by the second binding sites. In some embodiments, the two subunits are substantially identical.
In some aspects, the disclosure provides a macromolecule comprising two subunits, each comprising a first binding site and a second binding site, wherein (a) each of the first binding sites comprises an IL-22 polypeptide or a fragment or variant thereof that binds an IL-22 receptor on the surface of a target cell, and (b) each of the second binding sites is specific for an IL-10 family address
target selected from CDH17, CDH16, and GP2 that is expressed on the surface of the target cell; wherein (i) the second binding sites localize the first binding sites to the IL-10 family address target such that the IL-22 polypeptides or fragments or variants thereof influence signaling by the IL-22 receptor in the target cell; (ii) the second binding sites do not substantially influence signaling upon binding the IL-10 family address targets; and (iii) the IL-22 polypeptides or fragments or variants thereof do not substantially influence signaling by the IL-22 receptor in the absence of localization by the second binding sites. In some aspects, the two subunits are substantially identical. a. Divalent anti-CDH17/IL-22 ANDbody
In another aspect, the disclosure features a macromolecule comprising two subunits, each comprising a first binding site and a second binding site, wherein (a) each first binding site comprises IL- 22 or a fragment or variant thereof, and (b) each second binding site comprises an anti-CDH17 antibody or antigen-binding fragment thereof, wherein the macromolecule is a fusion protein, and wherein the first binding site and the second binding site of each subunit are connected by a linker.
In some embodiments, the IL-22 or fragment or variant thereof is a detuned or attenuated IL-22 or fragment or variant thereof, e.g., as described in Section B(i)(b), below.
In some embodiments, (i) the anti-CDH17 antibodies or antigen-binding fragments thereof localize the first binding sites to an IL-22 receptor such that the IL-22 or fragments or variants thereof influence signaling by the IL-22 receptor in the target cell; (ii) the anti-CDH17 antibodies or antigenbinding fragments thereof do not substantially influence signaling upon binding CDH17; and/or (iii) the IL- 22 or fragments or variants thereof do not substantially influence signaling by the IL-22 receptor in the absence of localization by the anti-CDH17 antibodies or antigen-binding fragments thereof.
The disclosure also features a method of modulating IL-22 receptor signaling in the small intestine or colon of a subject, the method comprising administering the above-described macromolecule to the subject. b. Divalent anti-CDH16/IL-22 ANDbody
In another aspect, the disclosure features a macromolecule comprising two subunits, each comprising a first binding site and a second binding site, wherein (a) each first binding site comprises IL- 22 or a fragment or variant thereof, and (b) each second binding site comprises an anti-CDH16 antibody or antigen-binding fragment thereof, wherein the macromolecule is a fusion protein, and wherein the first binding site and the second binding site of each subunit are connected by a linker.
In some embodiments, the IL-22 or fragment or variant thereof is a detuned or attenuated IL-22 or fragment or variant thereof, e.g., as described in Section B(i)(b), below.
In some embodiments, (i) the anti-CDH16 antibodies or antigen-binding fragments thereof localize the first binding sites to an IL-22 receptor such that the IL-22 or fragments or variants thereof influence signaling by the IL-22 receptor in the target cell; (ii) the anti-CDH16 antibodies or antigenbinding fragments thereof do not substantially influence signaling upon binding CDH16; and/or (iii) the IL- 22 or fragments or variants thereof do not substantially influence signaling by the IL-22 receptor in the absence of localization by the anti-CDH16 antibodies or antigen-binding fragments thereof.
The disclosure also features a method of modulating IL-22 receptor signaling in the kidney of a subject, the method comprising administering the above-described macromolecule to the subject. c. Divalent anti-GP2/IL-22 ANDbody
In another aspect, the disclosure features a macromolecule comprising two subunits, each comprising a first binding site and a second binding site, wherein (a) each first binding site comprises IL- 22 or a fragment or variant thereof, and (b) each second binding site comprises an anti-GP2 antibody or antigen-binding fragment thereof, wherein the macromolecule is a fusion protein, and wherein the first binding site and the second binding site of each subunit are connected by a linker.
In some embodiments, the IL-22 or fragment or variant thereof is a detuned or attenuated IL-22 or fragment or variant thereof, e.g., as described in Section B(i)(b), below.
In some embodiments, (i) the anti-GP2 antibodies or antigen-binding fragments thereof localize the first binding sites to an IL-22 receptor such that the IL-22 or fragments or variants thereof influence signaling by the IL-22 receptor in the target cell; (ii) the anti-GP2 antibodies or antigen-binding fragments thereof do not substantially influence signaling upon binding GP2; and/or (iii) the IL-22 or fragments or variants thereof do not substantially influence signaling by the IL-22 receptor in the absence of localization by the second binding sites.
The disclosure also features a method of modulating IL-22 receptor signaling in the pancreas of a subject, the method comprising administering the above-described macromolecule to the subject. ii. Monovalent ANDbody format
In other aspects, the macromolecule comprises a single first binding site and/or comprises a single second binding site (e.g., comprises a single first binding site and a single second binding site).
In some embodiments, the macromolecule is a heterodimer comprising two subunits, wherein the first subunit comprises the first binding site and second subunit comprises the second binding site.
In some embodiments, the macromolecule is a fusion protein. In some embodiments, the fusion protein comprises a linker (e.g., a GS linker) conjugated to the first binding site and the second binding site. a. Monovalent anti-CDH17/IL-22 ANDbody
In another aspect, the disclosure features a macromolecule comprising (a) a first binding site comprising IL-22 or a fragment or variant thereof, and (b) a second binding site comprising an anti- CDH17 antibody or antigen-binding fragment thereof, wherein the macromolecule is a fusion protein, and wherein the first binding site and the second binding site are connected by a linker.
In some embodiments, the IL-22 or fragment or variant thereof is a detuned or attenuated IL-22 or fragment or variant thereof, e.g., as described in Section B(i)(b), below.
In some embodiments, (i) the anti-CDH17 antibody or antigen-binding fragment thereof localizes the first binding site to an IL-22 receptor such that the IL-22 or fragment or variant thereof influences signaling by the IL-22 receptor in the target cell; (ii) the anti-CDH17 antibody or antigen-binding fragment thereof does not substantially influence signaling upon binding CDH17; and/or (iii) the IL-22 or fragment
or variant thereof does not substantially influence signaling by the IL-22 receptor in the absence of localization by the anti-CDH17 antibody or antigen-binding fragment thereof.
The disclosure also features a method of modulating IL-22 receptor signaling in the small intestine or colon of a subject, the method comprising administering the above-described macromolecule to the subject. b. Monovalent anti-CDH16/IL-22 ANDbody
In another aspect, the disclosure features a macromolecule comprising (a) a first binding site comprising IL-22 or a fragment or variant thereof, and (b) a second binding site comprising an anti- CDH16 antibody or antigen-binding fragment thereof, wherein the macromolecule is a fusion protein, and wherein the first binding site and the second binding site are connected by a linker.
In some embodiments, the IL-22 or fragment or variant thereof is a detuned or attenuated IL-22 or fragment or variant thereof, e.g., as described in Section B(i)(b), below.
In some embodiments, (i) the anti-CDH16 antibody or antigen-binding fragment thereof localizes the first binding site to an IL-22 receptor such that the IL-22 or fragment or variant thereof influences signaling by the IL-22 receptor in the target cell; (ii) the anti-CDH16 antibody or antigen-binding fragment thereof does not substantially influence signaling upon binding CDH16; and/or (iii) the IL-22 or fragment or variant thereof does not substantially influence signaling by the IL-22 receptor in the absence of localization by the anti-CDH16 antibody or antigen-binding fragment thereof.
The disclosure also features a method of modulating IL-22 receptor signaling in the kidney of a subject, the method comprising administering the above-described macromolecule to the subject. c. Monovalent anti-GP2/IL-22 ANDbody
In another aspect, the disclosure features a macromolecule comprising (a) a first binding site comprising IL-22 or a fragment or variant thereof, and (b) a second binding site comprising an anti-GP2 antibody or antigen-binding fragment thereof, wherein the macromolecule is a fusion protein, and wherein the first binding site and the second binding site are connected by a linker.
In some embodiments, the IL-22 or fragment or variant thereof is a detuned or attenuated IL-22 or fragment or variant thereof, e.g., as described in Section B(i)(b), below.
In some embodiments, (i) the anti-GP2 antibody or antigen-binding fragment thereof localizes the first binding site to an IL-22 receptor such that the IL-22 or fragment or variant thereof influences signaling by the IL-22 receptor in the target cell; (ii) the anti-GP2 antibody or antigen-binding fragment thereof does not substantially influence signaling upon binding GP2; and/or (iii) the IL-22 or fragment or variant thereof does not substantially influence signaling by the IL-22 receptor in the absence of localization by the anti-GP2 antibody or antigen-binding fragment thereof.
The disclosure also features a method of modulating IL-22 receptor signaling in the pancreas of a subject, the method comprising administering the above-described macromolecule to the subject. iii. ANDbody Function
The macromolecules (i.e ., ANDbodies) described herein comprise an effector binding domain (an
IL-22 polypeptide or fragment or variant thereof) specific for an effector target (an IL-22 receptor). Upon binding of the effector binding domain to the effector target, the effector modulates the target cell or tissue to produce a biological response, such as a therapeutic effect, on the subject. However, in some embodiments, the effector target binding domain provided herein may not elicit a biological effect unless it is provided in conjunction with an address targeting domain (e.g., an IL-10 family address target) to localize the effector to the desired target address in a targeted cell or tissue. In some embodiments, such therapeutic signaling may require the binding of multiple effector targets by multiple macromolecules according to the invention.
In some embodiments, an IL-22 polypeptide or fragment or variant thereof (e.g., a detuned or attenuated IL-22, e.g., as described in Section B(i)(a), below) may produce a small/weak biological effect when provided alone and provide a larger/stronger biological effect when provided in conjunction with an address targeting domain that localizes and concentrates/focuses the effector to the desired target address in a targeted cell or tissue. In some embodiments, an effector target binding domain may produce an acceptable biological effect when provided alone and provide an even larger/stronger biological effect when provided in conjunction with an address targeting domain to localize the effector target binding domain to a targeted cell or tissue. In some embodiments, an effector target binding domain may produce a strong biological effect when provided alone and provide a strong, or stronger, targeted effect when provided in conjunction with an address targeting domain to localize the effector target binding domain to a targeted cell or tissue. In some embodiments, an effector target binding domain may produce a biological effect with undesirable off target biological effects when provided alone, but can be targeted, concentrated, and focused to desired addresses in a targeted cell or tissue when provided in conjunction with an address targeting domain in order to decrease or eliminate undesirable off-target biological effects. Accordingly, effector target binding domains of the present technology provide superior therapeutic agents that provide stronger, targeted biological effects with less side effects, including less unintended off-target biological effects, when provided in conjunction with address target binding domains as described herein.
In some embodiments, binding of the address targeting domain to the IL-10 family address target: (i) increases accumulation of the macromolecule in tissues expressing the IL-10 family address target, (ii) reduces accumulation of the macromolecule in tissues that do not express the IL-10 family address target, or (iii) (i) and (ii).
In some embodiments, at least 25% of the macromolecule (e.g., ANDbody) detectable in the subject is detected at the target tissue or cell at a time point between 1 and 7 days (e.g., at 1 day, 2 days, 3 days, 4 days, 5, days, 6 days, and/or 7 days) following administration of the macromolecule (e.g., ANDbody) to the subject. For example, in some embodiments, at least 30%, at least 35%, at least 40%, at least 45%, at least 50%, at least 55%, at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 98%, or 100% (e.g., 25-30%, 30-35%, 35-40%, 40-45%, 45-50%, 50-55%, 55-60%, 60-65%, 65-70%, 70-75%, 75-80%, 80-85%, 85-90%, 90-95%, or 95-100%) of the macromolecule detectable in the subject is detected at the target tissue or cell at a time point between 1 and 7 days following administration of the macromolecule the subject
Examples of therapeutic signaling effects that may be induced by a macromolecule provided
herein include, but are not limited to:
(i) blocking a signal transduction pathway that promotes or maintains a disease state;
(ii) activating a signal transduction pathway that reduces or prevents a disease state;
(iii) promoting antibody-dependent cellular cytotoxicity (ADCC);
(iv) inducing complement activation on the target cell or tissue;
(v) promoting phagocytosis;
(vi) blocking or activating a signal transduction pathway that promotes differentiation of a cell; and
(vii) inducing tissue remodeling to reduce or prevent fibrosis.
Signaling effects (e.g., therapeutic signaling effects) that may be induced by a macromolecule provided herein further include modulation of cytokine signaling (e.g., modulation that affects immune response, response to infection, inflammation, allergy, autoimmunity, cancer, and/or tissue homeostasis). Cytokine signaling pathways are described, e.g., in Ouyang and O’Garra, Immunity, 50: 871 -891 , 2019).
In some embodiments, the therapeutic effector target (i.e., IL-22 receptor) is more broadly expressed than the address target in the subject. In some embodiments, the therapeutic effector target is expressed systemically, regionally, or locally in the organism. “Systemic expression” of a therapeutic effector target means that the therapeutic effector target is expressed at substantially the same levels in most parts of a subject organism body. Systemic expression involves a plurality of tissues. “Regional expression” of a therapeutic effector target means that the therapeutic target is expressed in an area less than systemic expression but more than local expression. Regional expression is not limited to a single tissue but can occur in a plurality of different tissues. “Local expression” of a therapeutic effector target means that the therapeutic target is expressed in single or few tissue areas. Local expression is not limited to a single tissue but can occur in a plurality of different tissues.
In some embodiments, the effector target binding domain has a low affinity for the effector target. For example, a low affinity may be an affinity of greater than 10 nM (e.g., an affinity between 10 nM-1 pM, e.g., an affinity between 10 nM and 100 nM).
In some embodiments, the effector target binding domain has a low avidity for the effector target.
B. IL-22 Polypeptides, Receptors, and Address Targets
An ANDbody™ of the invention comprises an effector (i.e., an IL-22 polypeptide or fragment or variant thereof, e.g., an IL-22 mutein polypeptide) that modulates a therapeutic effector target (i.e., an IL- 22 receptor to which the IL-22 polypeptide or fragment or variant thereof binds) in a cell or in a subject, e.g., a mammalian subject such as a human, in need thereof.
The disclosure also provides IL-22 mutein polypeptides or fragments or variants thereof that have altered (e.g., decreased) affinity for the IL-22 receptor. In some embodiments, the IL-22 mutein polypeptide or fragment or variant thereof has about a 1 .3-fold to about a 10-fold decreased binding affinity to human IL-22Ra as compared to the binding affinity of wild type human IL-22 polypeptide to human IL-22Ra, e.g., as determined by a method described herein. In some embodiments, the IL-22 mutein polypeptide or fragment or variant thereof has at least about a 1 .3-fold decreased binding affinity to human IL-22Ra as compared to the binding affinity of wild type human IL-22 polypeptide to human IL-
22Ra. In some embodiments, the IL-22 mutein polypeptide or fragment or variant thereof has at least about a 2-fold, 4-fold, 6-fold, 8-fold, or 10-fold decreased binding affinity to human IL-22Ra as compared to the binding affinity of wild type human IL-22 polypeptide to human IL-22Ra, or has a more than 10-fold decreased binding affinity to human IL-22Ra as compared to the binding affinity of wild type human IL-22 polypeptide to human IL-22Ra.
/. IL-22 Polypeptides and Fragments and Variants Thereof
In some embodiments, the effector is human Interleukin 22 (IL-22), e.g., as exemplified by UniProt Accession Number Q9GZX6; gene symbol IL22; and/or RefSeq NP_065386.1 or a fragment or variant thereof (e.g., a fully functional or partially functional fragment or variant thereof).
In some embodiments, the IL-22 polypeptide or fragment or variant thereof (e.g., IL-22 mutein polypeptide) comprises the mature form of the human IL-22 peptide. For example, in some embodiments, the IL-22 polypeptide or fragment or variant does not include a leader sequence, e.g., does not comprise the leader sequence of a pro-form of the IL-22 polypeptide. For example, in some embodiments, the IL-22 polypeptide or fragment or variant does not contain the leader sequence present in SEQ ID NO: 7, e.g., does not contain residues 1 to 33 of SEQ ID NO: 7. For example, in some embodiments, an IL-22 polypeptide or fragment or variant thereof comprises one or more amino acid substitution mutations (e.g., is an IL-22 mutein polypeptide comprising an amino acid substitution mutation at one or more of positions K61 , D71 , R73, D168, and R175, wherein the amino acid positions are numbered relative to SEQ ID NO: 7); and such IL-22 polypeptide or fragment or variant does not include the leader sequence of the pro-form of human IL-22 of SEQ ID NO: 7 (e.g., does not contain residues 1 to 33 of SEQ ID NO: 7).
As used herein, numbering of amino acid residues in the human IL-22 polypeptide are with reference to SEQ ID NO: 7 (the pro-form of human IL-22, comprising a leader sequence consisting of residues 1 -33). For example, for an IL-22 polypeptide or fragment or variant thereof that comprises an amino acid substitution mutation at one or more of positions K61 , D71 , R73, D168, and R175, the numbering of these amino acid residues refers to the sequence of SEQ ID NO: 7. However, as described above, the IL-22 polypeptide or fragment or variant (e.g., IL-22 mutein polypeptide) can comprise or consist of the mature form of the polypeptide that does not include a leader sequence, e.g., may not comprise the leader sequence of a pro-form of the IL-22 polypeptide (such as the leader sequence present in SEQ ID NO: 7, e.g., does not contain a sequence corresponding to residues 1 to 33 of SEQ ID NO: 7). In such embodiments, the numbering of residues nevertheless refers to SEQ ID NO: 7 (e.g., such that the first residue of the IL-22 polypeptide, fragment, or variant is position 34). For example, the IL-22 R73S polypeptide provided in SED IQ NO: 16 comprises an Arg to Ser amino acid substitution mutation at position 73, with reference to SEQ ID NO: 7 for position numbering. However, the IL-22 R73S polypeptide of SED IQ NO: 16 does not contain a leader sequence, e.g., the leader sequence of SEQ ID NO: 7, such that the R73S amino acid substitution mutation is located 40 residues from the N-terminus of the IL-22 polypeptide.
In some embodiments, the IL-22 polypeptide or fragment or variant thereof exhibits reduced IL- 22 receptor (e.g., IL-22a) binding relative to the binding of wild type IL-22 polypeptide, e.g., as described
in an assay provided herein. In some embodiments, the human IL-22 polypeptide or fragment or variant thereof has about a 1 .3-fold to about a10-fold decreased binding affinity to human IL-22Ra as compared to the binding affinity of wild type human IL-22 polypeptide to human IL-22Ra, e.g., as determined by a method described herein.
In some embodiments, when part of the macromolecule, the IL-22 polypeptide or fragment or variant thereof exhibits reduced IL-22 receptor binding relative to the IL-22 polypeptide or fragment or variant thereof when not part of the macromolecule, on a mole-adjusted basis, e.g., in an assay described herein.
In some embodiments, the IL-22 polypeptide or fragment or variant thereof exhibits reduced IL- 22 receptor binding relative to the IL-22 polypeptide or fragment or variant thereof on a mole-adjusted basis, e.g., in an assay described herein.
In some embodiments, when part of the macromolecule, the IL-22 polypeptide or fragment or variant thereof exhibits reduced agonist activity relative to the IL-22 polypeptide or fragment or variant thereof when not part of the macromolecule, on a mole-adjusted basis.
In some embodiments, the IL-22 polypeptide or fragment or variant thereof exhibits reduced agonist activity relative to the IL-22 polypeptide or fragment or variant thereof on a mole-adjusted basis.
/a. Attenuated IL-22 Polypeptides
In some embodiments, the disclosure provides an IL-22 polypeptide or fragment thereof that has lower binding affinity for the IL-22 receptor than a comparator IL-22 polypeptide or fragment or variant thereof (e.g., a wild-type IL-22 (e.g., of SEQ ID NO: 8) and/or an IL-22 that is a native ligand of the IL-22 receptor), e.g., is detuned or attenuated, e.g., as measured using an assay described herein. For example, the IL-22 polypeptide or fragment or variant thereof may be engineered to have reduced affinity for the receptor or may be a fragment of a wild-type IL-22 that has reduced affinity for the receptor (e.g., to IL-22Ra), e.g., as compared to the binding affinity of wild type human IL-22 (e.g., of SEQ ID NO: 8) for the IL-22 receptor, e.g., as measured an assay described herein (i.e., is an IL-22 mutein polypeptide). In some embodiments, the IL-22 mutein polypeptide or fragment thereof comprises one or more amino acid substitution mutations at one or more residues that are (or are predicted to be) at or near the interaction surface between IL-22 (and/or the IL-22 mutein polypeptide or fragment thereof) and the IL-22 receptor. In some embodiments, the IL-22 mutein polypeptide or fragment thereof may have a binding affinity for the IL-22 receptor that is at least 5%, 10%, 15%, 20%, 25%, 30%, 35%, 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, or 95% (e.g., 5-10%, 10-20%, 20-30%, 30-40%, 40-50%, 50-60%, 60- 70%, 70-80%, 80-90%, or more than 90%) lower than a comparator IL-22 polypeptide or fragment or variant thereof, e.g., as compared to the binding affinity of wild type human IL-22 (e.g., of SEQ ID NO: 8) for the IL-22 receptor (e.g., to IL-22Ra), e.g., in an assay described herein. A macromolecule of the invention (e.g., an ANDbody) may comprise such a detuned or attenuated IL-22 polypeptide.
In some embodiments, the IL-22 polypeptide or fragment or variant thereof of the macromolecule (e.g., a detuned or attenuated IL-22 (IL-22 mutein polypeptide)) exhibits reduced agonist activity (e.g., reduced activation of signaling by the IL-22 receptor), e.g., relative to a comparator IL-22 polypeptide or fragment or variant thereof (e.g., a wild-type IL-22 polypeptide, e.g., on a mole-adjusted basis). The
reduced agonist activity may be global, or may be measured at one or more target cells. For example, the IL-22 mutein polypeptide or fragment thereof may exhibit agonist activity that is reduced by at least 5%, 10%, 15%, 20%, 25%, 30%, 35%, 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, or 95% (e.g., 5-10%, 10-20%, 20-30%, 30-40%, 40-50%, 50-60%, 60-70%, 70-80%, 80-90%, or more than 90%) lower than a comparator IL-22 polypeptide or fragment or variant thereof (e.g., at one or more target cells), or may exhibit no observable agonist activity. In some instances, the reduced agonist activity is observed when the IL-22 polypeptide or fragment or variant thereof is not in the context of a macromolecule comprising a binding domain or other moiety that compensates for the reduced affinity of the IL-22 mutein polypeptide (e.g., an ANDbody), e.g., by binding to a target on a target cell. For example, in some aspects, the IL-22 mutein polypeptide exhibits reduced agonist activity or exhibits no observable agonist activity in the absence of a binding site targeting an IL-10 family address target, and the reduction or abrogation of agonist activity partially or entirely resolved (e.g., partially or entirely resolved at one or more target cells of the macromolecule) when the IL-22 mutein polypeptide or fragment thereof is in the context of a macromolecule targeting an IL-10 family address target (e.g., is fused to a domain that targets an IL-10 family address target).
In some embodiments, the IL-22 polypeptide or fragment or variant thereof comprised by a macromolecule (e.g., ANDbody) of the invention (e.g., IL-22 polypeptide or fragment or variant thereof having reduced binding affinity for the IL-22 receptor, e.g., IL-22 mutein polypeptide) does not substantially bind to the IL-22 receptor and/or does not exhibit substantial agonist activity at a target cell or tissue in the absence of a second binding site that specifically binds a target (e.g., an IL-10 family address target), thereby targeting the macromolecule to the target cell or tissue (e.g., signaling activity by the IL-22 polypeptide or fragment or variant thereof (e.g., IL-22 mutein polypeptide) at the target cell or tissue is conditional upon the presence of the second binding site in the macromolecule). In some embodiments, the affinity of the IL-22 polypeptide or fragment or variant thereof for the IL-22 receptor (and/or the affinity of the second binding site for the IL-10 family address target) is engineered such that substantial binding to the IL-22 receptor and substantial induction of signaling is conditional upon the presence of the second binding site in the macromolecule.
In some embodiments, the IL-22 or fragment or variant thereof (e.g., IL-22 mutein polypeptide) has reduced binding affinity for IL-22Ra and/or IL-10R2. For example, the IL-22 or fragment or variant thereof of the macromolecule may have a binding affinity for IL-22Ra and/or IL-10R2 that is at least 5%, 10%, 15%, 20%, 25%, 30%, 35%, 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, or 95% (e.g., 5-10%, 10-20%, 20-30%, 30-40%, 40-50%, 50-60%, 60-70%, 70-80%, 80-90%, or more than 90%) lower than a comparator IL-22, e.g., as compared to the binding affinity of wild type human IL-22 (e.g., of SEQ ID NO: 8) for the IL-22 receptor, e.g., as measured in an assay described herein. In some embodiments, the IL-22 polypeptide or fragment or variant thereof has about a 1 .3-fold to about a10-fold decreased binding affinity to human IL-22Ra as compared to the binding affinity of wild type human IL-22 polypeptide to human IL-22Ra, e.g., as determined by a method described herein.
The disclosure provides IL-22 polypeptides and fragments or variants thereof (e.g., IL-22 mutein polypeptides) that are capable of binding an IL-22 receptor on the surface of a target cell. In some aspects, the IL-22 polypeptide or fragment or variant thereof (e.g., IL-22 mutein polypeptide) binds the IL-
22 receptor only when paired with another moiety (e.g., a targeting domain), e.g., conditionally binds the IL-22 receptor. In some aspects, the IL-22 polypeptide or fragment or variant thereof (e.g., IL-22 mutein polypeptide) binds the IL-22 receptor at a threshold level (e.g., a level sufficient to induce downstream signaling and/or a therapeutically relevant level) only when paired with another moiety (e.g., a targeting domain). In other aspects, the IL-22 polypeptide or fragment or variant thereof constitutively binds the IL- 22 receptor.
In some embodiments, the IL-22 polypeptide or fragment or variant thereof binds the IL-22 receptor (e.g., conditionally or constitutively binds the IL-22 receptor). In some embodiments, the IL-22 polypeptide or fragment or variant thereof binds the human IL-22 receptor (e.g., conditionally or constitutively binds the human IL-22 receptor). In some embodiments, the IL-22 polypeptide or fragment or variant thereof binds human IL-22Ra and/or IL-10R2 (e.g., conditionally or constitutively binds human IL-22Ra and/or IL-10R2).
In some embodiments, the IL-22 polypeptide or a fragment or variant thereof binds IL-22BP (e.g., conditionally or constitutively binds IL-22BP). In some embodiments, the IL-22 polypeptide or fragment or variant thereof binds human IL-22BP (e.g., conditionally or constitutively binds human IL-22BP).
In some embodiments, the IL-22 polypeptide or fragment or variant thereof agonizes the IL-22 receptor (e.g., conditionally or constitutively agonizes the IL-22 receptor). In some embodiments, the IL- 22 polypeptide or fragment or variant thereof agonizes the human IL-22 receptor (e.g., conditionally or constitutively agonizes the human IL-22 receptor). In some embodiments, the IL-22 polypeptide or fragment or variant thereof agonizes human IL-22Ra and/or IL-10R2 (e.g., conditionally or constitutively agonizes human IL-22Ra and/or IL-10R2).
In some embodiments, the macromolecule agonizes IL-22BP (e.g., conditionally or constitutively agonizes IL-22BP). In some embodiments, the macromolecule agonizes human IL-22BP (e.g., conditionally or constitutively agonizes human IL-22BP).
In some embodiments, the IL-22 polypeptide or fragment or variant thereof (e.g., IL-22 mutein polypeptide) has low (e.g., lower) binding affinity for IL-22Ra and/or IL-10R2, as compared to the binding affinity of wild type human IL-22 (e.g., of SEQ ID NO: 8) for the IL-22 receptor, e.g., as measured in an assay described herein.
In some embodiments, the IL-22 polypeptide or fragment or variant thereof (e.g., IL-22 mutein polypeptide) has low (e.g., lower) binding affinity for IL-22BP, as compared to the binding affinity of wild type human IL-22 (e.g., of SEQ ID NO: 8) for IL-2BP, e.g., as measured in an assay described herein, e.g., a BLI assay described herein.
In some embodiments, the IL-22 or fragment thereof (e.g., IL-22 mutein polypeptide) comprises one or more amino acid substitution mutations at one or more residues that are (or are predicted to be) at or near the interaction surface between IL-22 and IL-22Ra and/or IL-10R2.
Residues at the interaction surface between IL-22 and its receptors are described, e.g., in Saxton et al., Immunity, 54(4): 660-672e.9, 2021 and in Wu et al., Journal of Molecular Biology, 382(5): 1168- 1183, 2008.
In some embodiments, the IL-22 or fragment or variant thereof (e.g., IL-22 mutein polypeptide) has reduced binding affinity for IL22RA1 relative to a wild-type IL-22 or fragment thereof, e.g., as
compared to the binding affinity of wild type human IL-22 (e.g., of SEQ ID NO: 8) for the IL-22 receptor, e.g., as measured in an assay described herein. In some embodiments, the IL-22 polypeptide or fragment or variant thereof has about a 1 .3-fold to about a10-fold decreased binding affinity to human IL- 22Ra as compared to the binding affinity of wild type human IL-22 polypeptide to human IL-22Ra, e.g., as determined by a method described herein.
In some embodiments, the IL-22 polypeptide or fragment or variant thereof (e.g., IL-22 mutein polypeptide) does not comprise a leader sequence, e.g., does not comprise a leader sequence of a proform of a IL-22 polypeptide. In some embodiments, the IL-22 polypeptide or fragment or variant thereof does not comprise the leader sequence comprised in SEQ ID NO: 7, e.g., does not comprise residues 1 to 33 of SEQ ID NO: 7.
In some embodiments, the IL-22 polypeptide or fragment or variant thereof does not comprise the sequence of SEQ ID NO: 14.
In some embodiments, the IL-22 polypeptide or fragment or variant thereof is a human IL-22 or fragment or variant thereof.
In some embodiments, the IL-22 polypeptide or fragment or variant thereof is a modified human IL-22 or fragment or variant thereof and comprises one or more of the amino acid substitution mutations of Table 6, Table 7A, Table 7B, Table 8, Table 9A, Table 9B, or Table 9C (e.g., comprises one or more of said amino acid mutations and has at least 80%, 85%, 90%, 95%, 98%, or 99% identity to a human IL- 22, e.g., as set forth in SEQ ID NO: 7 or SEQ ID NO: 8).
In some embodiments, the IL-22 polypeptide or fragment or variant thereof (e.g., IL-22 mutein polypeptide) comprises an amino acid substitution mutation at one or more of positions K61 , D71 , R73, D168, and R175, wherein the amino acid position numbering is with respect to the human IL-22 sequence provided in SEQ ID NO: 7. In some embodiments, the IL-22 polypeptide or fragment or variant thereof comprises a K61 amino acid substitution mutation. In some embodiments, the IL-22 polypeptide or fragment or variant thereof comprises a D71 amino acid substitution mutation. In some embodiments, the IL-22 polypeptide or fragment or variant thereof comprises a R73 amino acid substitution mutation. In some embodiments, the IL-22 polypeptide or fragment or variant thereof comprises a D168 amino acid substitution mutation. In some embodiments, the IL-22 polypeptide or fragment or variant thereof comprises a R175 amino acid substitution mutation. In some embodiments, the IL-22 polypeptide or fragment or variant thereof comprises a D71 amino acid substitution mutation and a D168 amino acid substitution mutation. In some embodiments, the IL-22 mutein polypeptide comprising the amino acid substitution mutation at position K61 , D71 , R73, D168, and/or R175 has at least 80%, 85%, 90%, 95%, 98%, or 99% identity to a human IL-22, e.g., as set forth in SEQ ID NO: 7 or SEQ ID NO: 8).
In some embodiments, the IL-22 polypeptide or fragment or variant thereof comprises one or more of a K61 S, D71 N, D71 L, D71 G, R73Q, R73S, R73N, D168N, D168G, and R175S amino acid substitution mutation, wherein the amino acid position numbering is with respect to the human IL-22 sequence provided in SEQ ID NO: 7. In some embodiments, the IL-22 polypeptide or fragment or variant thereof comprises a K61 S amino acid substitution mutation. In some embodiments, the IL-22 polypeptide or fragment or variant thereof comprises a D71 N amino acid substitution mutation. In some embodiments, the IL-22 polypeptide or fragment or variant thereof comprises a D71 L amino acid
substitution mutation. In some embodiments, the IL-22 polypeptide or fragment or variant thereof comprises a D71 G amino acid substitution mutation. In some embodiments, the IL-22 polypeptide or fragment or variant thereof comprises a R73Q amino acid substitution mutation. In some embodiments, the IL-22 polypeptide or fragment or variant thereof comprises a R73S amino acid substitution mutation. In some embodiments, the IL-22 polypeptide or fragment or variant thereof comprises a R73N amino acid substitution mutation. In some embodiments, the IL-22 polypeptide or fragment or variant thereof comprises a D168N amino acid substitution mutation. In some embodiments, the IL-22 polypeptide or fragment or variant thereof comprises a D168G amino acid substitution mutation. In some embodiments, the IL-22 polypeptide or fragment or variant thereof comprises a R175S amino acid substitution mutation. In some embodiments, the IL-22 polypeptide or fragment or variant thereof comprises a D71 L amino acid substitution mutation and a D168G amino acid substitution mutation. In some embodiments, the IL-22 mutein polypeptide comprising the one or more amino acid substitution mutations has at least 80%, 85%, 90%, 95%, 98%, or 99% identity to a human IL-22, e.g., as set forth in SEQ ID NO: 7. In some embodiments, the IL-22 mutein polypeptide comprising the one or more amino acid substitution mutations has at least 80%, 85%, 90%, 95%, 98%, or 99% identity to a human IL-22, e.g., as set forth in SEQ ID NO: 8. In some embodiments, the IL-22 mutein polypeptide has about a 1 .3-fold to about a 10- fold decreased binding affinity to human IL-22Ra as compared to the binding affinity of wild type human IL-22 polypeptide to human IL-22Ra, e.g., as determined by a method described herein.
In some embodiments, the IL-22 polypeptide or fragment or variant thereof comprises a K61 S amino acid substitution mutation.
In some embodiments, the IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 17 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 17). In some embodiments, the IL-22 mutein polypeptide binds to IL-22Ra. In some embodiments, the IL-22 mutein polypeptide has about a 1 .3-fold to about a10-fold decreased binding affinity to human IL-22Ra as compared to the binding affinity of wild type human IL-22 polypeptide to human IL-22Ra, e.g., as determined by a method described herein.
In some embodiments, the IL-22 polypeptide or fragment or variant thereof comprises a K61 S amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 17. In some embodiments, the IL-22 mutein polypeptide binds to IL- 22Ra. In some embodiments, the IL-22 mutein polypeptide has about a 1 .3-fold to about a10-fold decreased binding affinity to human IL-22Ra as compared to the binding affinity of wild type human IL-22 polypeptide to human IL-22Ra, e.g., as determined by a method described herein.
In some embodiments, the IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 17. In some embodiments, the IL-22 polypeptide or fragment or variant thereof consists of SEQ ID NO: 17.
In some embodiments, the IL-22 polypeptide or fragment or variant thereof comprises a D71 G amino acid substitution mutation.
In some embodiments, the IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 15 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99%
identity to SEQ ID NO: 15). In some embodiments, the IL-22 mutein polypeptide binds to IL-22Ra. In some embodiments, the IL-22 mutein polypeptide has about a 1 .3-fold to about a10-fold decreased binding affinity to human IL-22Ra as compared to the binding affinity of wild type human IL-22 polypeptide to human IL-22Ra, e.g., as determined by a method described herein.
In some embodiments, the IL-22 polypeptide or fragment or variant thereof comprises a D71 G amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 15. In some embodiments, the IL-22 mutein polypeptide binds to IL- 22Ra. In some embodiments, the IL-22 mutein polypeptide has about a 1 .3-fold to about a10-fold decreased binding affinity to human IL-22Ra as compared to the binding affinity of wild type human IL-22 polypeptide to human IL-22Ra, e.g., as determined by a method described herein.
In some embodiments, the IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 15. In some embodiments, the IL-22 polypeptide or fragment or variant thereof consists of SEQ ID NO: 15.
In some embodiments, the IL-22 polypeptide or fragment or variant thereof comprises a D71 L amino acid substitution mutation.
In some embodiments, the IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 11 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 11 ). In some embodiments, the IL-22 mutein polypeptide binds to IL-22Ra. In some embodiments, the IL-22 mutein polypeptide has about a 1 .3-fold to about a10-fold decreased binding affinity to human IL-22Ra as compared to the binding affinity of wild type human IL-22 polypeptide to human IL-22Ra, e.g., as determined by a method described herein.
In some embodiments, the IL-22 polypeptide or fragment or variant thereof comprises a D71 L amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 11 . In some embodiments, the IL-22 mutein polypeptide binds to IL- 22Ra. In some embodiments, the IL-22 mutein polypeptide has about a 1 .3-fold to about a10-fold decreased binding affinity to human IL-22Ra as compared to the binding affinity of wild type human IL-22 polypeptide to human IL-22Ra, e.g., as determined by a method described herein.
In some embodiments, the IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 11. In some embodiments, the IL-22 polypeptide or fragment or variant thereof consists of SEQ ID NO: 11.
In some embodiments, the IL-22 polypeptide or fragment or variant thereof comprises a D71 N amino acid substitution mutation.
In some embodiments, the IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 18 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 18). In some embodiments, the IL-22 mutein polypeptide binds to IL-22Ra. In some embodiments, the IL-22 mutein polypeptide has about a 1 .3-fold to about a10-fold decreased binding affinity to human IL-22Ra as compared to the binding affinity of wild type human IL-22 polypeptide to human IL-22Ra, e.g., as determined by a method described herein.
In some embodiments, the IL-22 polypeptide or fragment or variant thereof comprises a D71 N amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 18. In some embodiments, the IL-22 mutein polypeptide binds to IL- 22Ra. In some embodiments, the IL-22 mutein polypeptide has about a 1 .3-fold to about a10-fold decreased binding affinity to human IL-22Ra as compared to the binding affinity of wild type human IL-22 polypeptide to human IL-22Ra, e.g., as determined by a method described herein.
In some embodiments, the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 18. In some embodiments, the IL-22 polypeptide or fragment or variant thereof consists of SEQ ID NO: 18.
In some embodiments, the IL-22 polypeptide or fragment or variant thereof comprises a R73S amino acid substitution mutation.
In some embodiments, the IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 16 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 16). In some embodiments, the IL-22 mutein polypeptide binds to IL-22Ra. In some embodiments, the IL-22 mutein polypeptide has about a 1 .3-fold to about a10-fold decreased binding affinity to human IL-22Ra as compared to the binding affinity of wild type human IL-22 polypeptide to human IL-22Ra, e.g., as determined by a method described herein.
In some embodiments, the IL-22 polypeptide or fragment or variant thereof comprises a R73S amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 16. In some embodiments, the IL-22 mutein polypeptide binds to IL- 22Ra. In some embodiments, the IL-22 mutein polypeptide has about a 1 .3-fold to about a10-fold decreased binding affinity to human IL-22Ra as compared to the binding affinity of wild type human IL-22 polypeptide to human IL-22Ra, e.g., as determined by a method described herein.
In some embodiments, the IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 16. In some embodiments, the IL-22 polypeptide or fragment or variant thereof consists of SEQ ID NO: 16.
In some embodiments, the IL-22 polypeptide or fragment or variant thereof comprises a R73N amino acid substitution mutation.
In some embodiments, the IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 22 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 22). In some embodiments, the IL-22 mutein polypeptide binds to IL-22Ra. In some embodiments, the IL-22 mutein polypeptide has about a 1 .3-fold to about a10-fold decreased binding affinity to human IL-22Ra as compared to the binding affinity of wild type human IL-22 polypeptide to human IL-22Ra, e.g., as determined by a method described herein.
In some embodiments, the IL-22 polypeptide or fragment or variant thereof comprises a R73N amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the IL-22 polypeptide or fragment or variant thereof to SEQ ID NO:22, In some embodiments, the IL-22 mutein polypeptide binds to IL-
22Ra. In some embodiments, the IL-22 mutein polypeptide has about a 1 .3-fold to about a10-fold decreased binding affinity to human IL-22Ra as compared to the binding affinity of wild type human IL-22 polypeptide to human IL-22Ra, e.g., as determined by a method described herein.
In some embodiments, the IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 22. In some embodiments, the IL-22 polypeptide or fragment or variant thereof consists of SEQ ID NO: 22.
In some embodiments, the IL-22 polypeptide or fragment or variant thereof comprises a R73Q amino acid substitution mutation.
In some embodiments, the IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 12 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 12). In some embodiments, the IL-22 mutein polypeptide binds to IL-22Ra. In some embodiments, the IL-22 mutein polypeptide has about a 1 .3-fold to about a10-fold decreased binding affinity to human IL-22Ra as compared to the binding affinity of wild type human IL-22 polypeptide to human IL-22Ra, e.g., as determined by a method described herein.
In some embodiments, the IL-22 polypeptide or fragment or variant thereof comprises a R73Q amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 12. In some embodiments, the IL-22 mutein polypeptide binds to IL- 22Ra. In some embodiments, the IL-22 mutein polypeptide has about a 1 .3-fold to about a10-fold decreased binding affinity to human IL-22Ra as compared to the binding affinity of wild type human IL-22 polypeptide to human IL-22Ra, e.g., as determined by a method described herein.
In some embodiments, the IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 12. In some embodiments, the IL-22 polypeptide or fragment or variant thereof consists of SEQ ID NO: 12.
In some embodiments, the IL-22 polypeptide or fragment or variant thereof comprises a D168N amino acid substitution mutation.
In some embodiments, the IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 19 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 19). In some embodiments, the IL-22 mutein polypeptide binds to IL-22Ra. In some embodiments, the IL-22 mutein polypeptide has about a 1 .3-fold to about a10-fold decreased binding affinity to human IL-22Ra as compared to the binding affinity of wild type human IL-22 polypeptide to human IL-22Ra, e.g., as determined by a method described herein.
In some embodiments, the IL-22 polypeptide or fragment or variant thereof comprises a D168N amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 19. In some embodiments, the IL-22 mutein polypeptide binds to IL- 22Ra. In some embodiments, the IL-22 mutein polypeptide has about a 1 .3-fold to about a10-fold decreased binding affinity to human IL-22Ra as compared to the binding affinity of wild type human IL-22 polypeptide to human IL-22Ra, e.g., as determined by a method described herein.
In some embodiments, the IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 19. In some embodiments, the IL-22 polypeptide or fragment or variant thereof consists of SEQ ID NO: 19.
In some embodiments, the IL-22 polypeptide or fragment or variant thereof comprises a D168G amino acid substitution mutation.
In some embodiments, the IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 20 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 20). In some embodiments, the IL-22 mutein polypeptide binds to IL-22Ra. In some embodiments, the IL-22 mutein polypeptide has about a 1 .3-fold to about a10-fold decreased binding affinity to human IL-22Ra as compared to the binding affinity of wild type human IL-22 polypeptide to human IL-22Ra, e.g., as determined by a method described herein.
In some embodiments, the IL-22 polypeptide or fragment or variant thereof comprises a D168G amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 20. In some embodiments, the IL-22 mutein polypeptide binds to IL- 22Ra. In some embodiments, the IL-22 mutein polypeptide has about a 1 .3-fold to about a10-fold decreased binding affinity to human IL-22Ra as compared to the binding affinity of wild type human IL-22 polypeptide to human IL-22Ra, e.g., as determined by a method described herein.
In some embodiments, the IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 20. In some embodiments, the IL-22 polypeptide or fragment or variant thereof consists of SEQ ID NO: 20.
In some embodiments, the IL-22 polypeptide or fragment or variant thereof comprises D71 L and D168G amino acid substitution mutations.
In some embodiments, the IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 13 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 13). In some embodiments, the IL-22 mutein polypeptide binds to IL-22Ra. In some embodiments, the IL-22 mutein polypeptide has about a 1 .3-fold to about a10-fold decreased binding affinity to human IL-22Ra as compared to the binding affinity of wild type human IL-22 polypeptide to human IL-22Ra, e.g., as determined by a method described herein.
In some embodiments, the IL-22 polypeptide or fragment or variant thereof comprises D71 L and D168 amino acid substitution mutations and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 13. In some embodiments, the IL-22 mutein polypeptide binds to IL-22Ra. In some embodiments, the IL-22 mutein polypeptide has about a 1 .3-fold to about a10-fold decreased binding affinity to human IL-22Ra as compared to the binding affinity of wild type human IL-22 polypeptide to human IL-22Ra, e.g., as determined by a method described herein.
In some embodiments, the IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 13. In some embodiments, the IL-22 polypeptide or fragment or variant thereof consists of SEQ ID NO: 13.
In some embodiments, the IL-22 polypeptide or fragment or variant thereof comprises a R175S amino acid substitution mutation.
In some embodiments, the IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 21 (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 21 ). In some embodiments, the IL-22 mutein polypeptide binds to IL-22Ra. In some embodiments, the IL-22 mutein polypeptide has about a 1 .3-fold to about a10-fold decreased binding affinity to human IL-22Ra as compared to the binding affinity of wild type human IL-22 polypeptide to human IL-22Ra, e.g., as determined by a method described herein.
In some embodiments, the IL-22 polypeptide or fragment or variant thereof comprises a R175S amino acid substitution mutation and has at least 90% identity (e.g., has at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity) over the remaining sequence of the IL-22 polypeptide or fragment or variant thereof to SEQ ID NO: 21 . In some embodiments, the IL-22 mutein polypeptide binds to IL- 22Ra. In some embodiments, the IL-22 mutein polypeptide has about a 1 .3-fold to about a10-fold decreased binding affinity to human IL-22Ra as compared to the binding affinity of wild type human IL-22 polypeptide to human IL-22Ra, e.g., as determined by a method described herein.
In some embodiments, the IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 21 . In some embodiments, the IL-22 polypeptide or fragment or variant thereof consists of SEQ ID NO: 21.
As used herein, an "effector target" is a discrete structure (e.g., a cell surface protein, a transmembrane protein, or a receptor) of a cell or tissue of a subject, to which a therapeutic effector binding domain of an ANDbody can bind and exert a modulating effect, such as a therapeutic effect on the subject. Effector targets as provided herein are molecules that are capable of binding to an IL-22 polypeptide or fragment or variant thereof as described above, wherein the binding affects (e.g., agonizes) an activity of the effector target (e.g., activates or enhances downstream signaling).
An effector target provided herein is an IL-22 receptor. IL-22 receptor subunits are known in the art, and include IL-1 ORB (IL-10R2) and IL-22Ra (IL22RA1 ) (see, e.g., Ouyang and O’Garra, Immunity, 50: 871 -891 , 2019). These receptor subunits generally form heterodimers and are agonized by IL-22. Accordingly, in some aspects, a macromolecule or polypeptide provided herein agonizes an IL-22 receptor (e.g., agonizes one or more IL-22 receptor subunits, e.g., agonizes IL-1 ORB and/or IL-22Ra, e.g., agonizes a heterodimeric receptor comprising IL-1 ORB and IL-22Ra).
In some aspects, a macromolecule or polypeptide provided herein agonizes one or more IL-22 receptor subunits, e.g., agonizes IL-1 ORB and/or IL-22Ra.
In some aspects, a macromolecule or polypeptide provided herein binds one or more IL-22 receptor subunits, e.g., binds IL-1 ORB and/or IL-22Ra.
In some embodiments, the IL-22 receptor comprises IL-1 ORB (e.g., human IL-1 ORB, e.g., as exemplified by UniProt Accession Number Q08334).
In some embodiments, the IL-22 receptor comprises IL-22RA (IL-22Ra; IL-22RA1 ) (e.g., human IL-22RA, e.g., as exemplified by UniProt Accession Number Q8N6P7).
In some aspects, a macromolecule or polypeptide provided herein agonizes a decoy receptor, e.g., IL-22 BP.
In some aspects, a macromolecule or polypeptide provided herein binds a decoy receptor, e.g., IL-22 BP.
In some embodiments, the IL-22 decoy receptor comprises IL-22BP (IL-22RA2) (e.g., human IL- 22BP, e.g., as exemplified by UniProt Accession Number Q969J5).
Hi. IL-10 Family Address Targets and Binding Sites
A macromolecule (i.e., ANDbody) of the invention also comprises an address target binder (second binding site) that binds to, e.g., specifically binds to or is specific for, an address target to provide targeted delivery of the effector. As used herein, an “address target” is a structure on a cell or tissue whose expression is sufficiently restricted in an organism to allow it to identify an organ, tissue, cell, or cell state of interest in an organism. The address target can be, e.g., a cell surface protein, or a structure localizing to the extracellular matrix. As used herein, “restricted” expression of an address target means that the address target has a differential, e.g., less broad, in vivo expression, as opposed to systemic expression. In certain embodiments, the address target is expressed, for example, in a single cell type, tissue or cell state in a mammalian subject, such as a human subject.
Address targets as provided herein are IL-10 family address targets. As used herein, an “IL-10 family address target” is a cell surface moiety (e.g., a protein, a lipid, or a sugar) that is expressed on the surface of a cell that also expresses an IL-10 family receptor (e.g., expresses an IL-22 receptor to which the IL-22 polypeptide or fragment or variant thereof comprised by the ANDbody binds).
In some embodiments, an address target binder, e.g., a second binding site, is specific for an IL- 10 family address target provided in Table 1 that is expressed on the surface of the target cell. As used herein, the terms “specifically binds” and “is specific for” refer to a preferential interaction between a binding domain and its target or ligand (such as binding between an antibody and an antigen or epitope) that may be determinative of the presence of the target or ligand in the presence of a heterogeneous population of molecules including biological molecules. For example, a binding domain that specifically binds to a ligand (e.g., an antibody that specifically binds to an antigen or epitope) may be a binding domain that binds this ligand with greater affinity, avidity, more readily, and/or with greater duration than it binds to other ligands. In one aspect, the extent of binding of a binding domain to an unrelated molecule (non-ligand) is less than about 10% of the binding of the binding domain to the ligand as measured using an appropriate assay. In certain aspects, a binding domain that specifically binds to a ligand has a dissociation constant (KD) of < 1 pM, < 100 nM, < 10 nM, < 1 nM, or < 0.1 nM. Specific binding can include, but does not require exclusive binding. In one aspect, “specific binding” refers to binding wherein a binding domain binds to a particular ligand (e.g., a polypeptide or antigen or epitope on a particular polypeptide) without substantially binding to any other ligand (e.g., polypeptide or polypeptide antigen or epitope).
Exemplary IL-10 family address targets are presented in Table 1 , below. In some embodiments, the second binding site is specific for an IL-10 family address target provided in Table 1 (e.g., is specific for is an IL-10 family address target provided in Table 1 that is expressed on the surface of a cell that also expresses an IL-10 family receptor (e.g., expresses IL-22 family receptor to which the IL-22 polypeptide or fragment or variant thereof comprised by the ANDbody binds)). In some aspects, the cell
on which the IL-10 family address target is expressed (i.e., the cell targeted by the ANDbody) is a an epithelial cell or a cell type from one or more tissues selected from small intestine, large intestine, lower gastrointestinal (Gl) tract, rectum, skin, pancreas, kidney, lungs, and liver.
In some embodiments, the first binding site of the ANDbody comprises IL-22 or a fragment or variant thereof, and the second binding site is specific for an IL-10 family address target provided in Table 1 .
In some embodiments, the target cell is a lower gastrointestinal (Gl) tract cell, the IL-10 family address target is MS4A18, SI, MS4A12, ATP4B, MUC17, TMIGD1 , MS4A10, MEP1 A, CDH17, TM4SF20, SLC26A3, ATP4A, GPA33, BTNL3, MEP1 B, UGT2B17, KCNE2, or TMPRSS15 (i.e., the second binding site is specific for MS4A18, SI, MS4A12, ATP4B, MUC17, TMIGD1 , MS4A10, MEP1 A, CDH17, TM4SF20, SLC26A3, ATP4A, GPA33, BTNL3, MEP1 B, UGT2B17, KCNE2, or TMPRSS15), and the first binding site of the ANDbody comprises IL-22 or a fragment or variant thereof.
In some embodiments, the target cell is a kidney cell, the IL-10 family address target is TMEM207, OR2T10, SLC22A6, SLC22A8, SLC12A1 , AQP2, SLC22A12, SLC12A3, TMEM174, FXYD4, SLC34A1 , KCNJ1 , SLC22A1 1 , TMEM52B, SLC6A18, CTXN3, SLC4A1 , TMEM72, SLC22A2, SLC4A9, BSND, CDH16, RDH8, or AQP6 (i.e., the second binding site is specific for TMEM207, OR2T10, SLC22A6, SLC22A8, SLC12A1 , AQP2, SLC22A12, SLC12A3, TMEM174, FXYD4, SLC34A1 , KCNJ1 , SLC22A1 1 , TMEM52B, SLC6A18, CTXN3, SLC4A1 , TMEM72, SLC22A2, SLC4A9, BSND, CDH16, RDH8, or AQP6), and the first binding site of the ANDbody comprises IL-22 or a fragment or variant thereof.
In some embodiments, the target cell is a liver cell, the IL-10 family address target is UGT2B10, C8A, SLCO1 B1 , C9, SLC25A47, SLC17A2, SLC10A1 , UGT2B4, SLC22A1 , or CYP8B1 (i.e., the second binding site is specific for UGT2B10, C8A, SLCO1 B1 , C9, SLC25A47, SLC17A2, SLC10A1 , UGT2B4,
SLC22A1 , or CYP8B1 ), and the first binding site of the ANDbody comprises IL-22 or a fragment or variant thereof.
In some embodiments, the target cell is a pancreas cell, the IL-10 family address target is GP2, AQP12A, AQP12B, CUZD1 , LHFPL5, AQP8, KIRREL2, G6PC2, SLC17A6, or GPR1 19 (i.e., the second binding site is specific for GP2, AQP12A, AQP12B, CUZD1 , LHFPL5, AQP8, KIRREL2, G6PC2, SLC17A6, or GPR1 19), and the first binding site of the ANDbody comprises IL-22 or a fragment or variant thereof.
In some embodiments, the target cell is a skin cell, the IL-10 family address target is CLEC2A, KCNJ18, KLRF2, DSC1 , TYR, AWAT2, TMEM271 , DSG1 , ACER1 , GSDMA, TRPM1 , GJB4, CD1 A, ASPRV1 , or ABCA12 (i.e., the second binding site is specific for CLEC2A, KCNJ18, KLRF2, DSC1 , TYR, AWAT2, TMEM271 , DSG1 , ACER1 , GSDMA, TRPM1 , GJB4, CD1 A, ASPRV1 , or ABCA12), and the first binding site of the ANDbody comprises IL-22 or a fragment or variant thereof.
In some embodiments, the IL-10 family address target is selected from CDH17, GP2, CDH16, DSG1 , and DSG3.
In some embodiments, the IL-10 family address target is CDH17. In some embodiments, the second binding site is an anti-CDH17 antibody or antigen-binding fragment thereof. In some embodiments, the cell is a gut epithelial cell and the macromolecule is targeted to small intestine and colon tissues.
In some embodiments, the IL-10 family address target is CDH16. In some embodiments, the second binding site is an anti-CDH16 antibody or antigen-binding fragment thereof. In some embodiments, the cell is a kidney cell (e.g., an epithelial cell in the kidney tubules) and the macromolecule is targeted to the kidney.
In some embodiments, the IL-10 family address target is GP2. In some embodiments, the second binding site is an anti-GP2 antibody or antigen-binding fragment thereof. In some embodiments, the cell is a kidney cell and the macromolecule is targeted to the pancreas.
In some embodiments, the second binding site (address target binder) comprises an antibody. The term “antibody,” as used herein, includes full-length antibodies and antigen binding antibody fragments (e.g., scFvs). In some embodiments, an antibody molecule has specificity for more than one, e.g., 2, 3, or 4 antigens, e.g., the antibody molecule comprises a plurality of variable domain sequences, wherein a first variable domain sequence of the plurality has binding specificity for a first epitope (e.g., the effector target) and a second variable domain sequence of the plurality has binding specificity for a second epitope (e.g., the address target). In other embodiments, the antibody molecule is monospecific.
In some embodiments, the second binding site (address target binder) has the structure of an scFv, BsIgG, a BsAb fragment, a BiTE, a dual-affinity re-targeting protein (DART), a tandem diabody (TandAb), a diabody, an Fab2, a di-scFv, chemically linked F(ab’)2, an Ig molecule with 2, 3 or 4 different antigen binding sites, a DVI-IgG four-in-one, an ImmTac, an HSAbody, an IgG-IgG, a Cov-X-Body, an SCFV1 -PEG-SCFV2, an appended IgG, an DVD-IgG, an affibody, an affilin, an affimer, an affitin, an alphabody, an anticalin, an avimer, a DARPin, a Fynomer, a monobody, a nanoCLAMP, a bis-Fab, an Fv, a Fab, a Fab’-SH, a linear antibody, an scFv, an antibody with only a heavy chain (Humabody), an ScFab, an IgG antibody fragment, a single-chain variable region antibody, a single-domain heavy chain
antibody, a bispecific triplebody, a BiKE, a CrossMAb, a dsDb, an scDb, tandem a dAb I VHH, a triple dAb VHH, a tetravalent dAb I VHH, a Fab-scFv, a Fab-Fv, or a DART-Fc, an adnectin, a Kunitz-type inhibitor, or a receptor decoy.
In some embodiments, the second binding site (address target binder) comprises a ligand of the address target.
In some embodiments, the currently provided address target binding domains (IL-10 family address target binding domains) do not substantially influence biological signaling upon binding to the address target, e.g., do not modulate a signal transduction pathway or other biological response in the target cell or tissue. For example, the address target binder can be inert or inactive, in which it lacks any additional activity (other than binding), including lacking catalytic activity, after binding to the address target. For example, the address target binder binds a non-signaling site or motif of the address target. “Signal” is used herein to indicate a conformational, enzymatic, and/or electrical consequence occurs as a result of target binding. Accordingly, as described herein, address target binding domains do not signal upon address target binding. A domain that does not “substantially” influence biological signaling, as used herein, is a domain that modulates a signal transduction pathway or other biological response in the target cell or tissue to which it binds by no more than 25% relative to a control condition, e.g., relative to signaling in the absence of the domain. For example, the domain may modulate (e.g., increase or decrease) the signal transduction pathway or other biological response by less than 20%, less than 15%, less than 10%, less than 5%, less than 2%, or less than 1% (e.g., 20-25%, 15-20%, 10-15%, 5-10%, 2- 5%, or 1 -2%).
In some embodiments, the address target is used for organ-specific addressing, tissue-specific addressing, or cell-specific addressing. In some embodiments, the address target is used or cell-specific addressing.
The specificity of address target binding domains for a cell or tissue can be detected using methods known in the art. In one embodiment, a Gini coefficient (GO) score, which is a method for assessing the expression variation of a particular gene in a data set, is used. (See O’Hagan et al., GeneGini: assessment via the Gini coefficient of reference “housekeeping” genes and diverse human transporter expression profiles. Cell systems 6, 230-244, https://doi.Org/10.1016/j. cels.2018.01 .003 (2018); Wright Muelas et al., The role and robustness of the Gini coefficient as an unbiased tool for the selection of Gini genes for normalising expression profiling data. Sci Rep 9, 17960 (2019). https://doi.org/10.1038/s41598-019-54288-7). Address target binders can be identified using cell expression data generated for address target binders. In some embodiments, address target markers exhibit Gini scores of greater than 0.4, such as between 0.74 and 1 .00. Conversely, non-address markers that are expressed more systemically may exhibit Gini Scores of between 0.15 to 0.19.
In one embodiment, a Tau score, which represents the expression variation of a particular gene in a data set, is used. Calculating Tau uses the information of expression of a gene in each tissue and its maximal expression over all tissues while also taking into account the number of tissues where expression is measured (see Itai Yanai, et al., Genome-wide midrange transcription profiles reveal expression level relationships in human tissue specification, Bioinformatics, Volume 21 , Issue 5, 1 March 2005, Pages 650-659; Kryuchkova-Mostacci N, Robinson-Rechavi M. A benchmark of gene expression
tissue-specificity metrics. Brief Bioinform. 2017 Mar 1 ;18(2):205-214. doi: 10.1093/bib/bbw008). In some embodiments, address target markers exhibit Tau scores of greater than 0.6, such as between 0.74 and 1 .00. Conversely, non-address markers that are expressed more systemically may exhibit Tau Scores of below 0.3, such as 0.15 to 0.19.
In some embodiments, specificity of address target binding domains for a particular cell or tissue, such as that indicated by an appropriate Gini and/or Tau score, is determined with a tissue based analysis that does not include tissues having a natural biological separation barrier (i.e., blood-brain barrier). For example, in some embodiments, Gini and/or Tau scores may be calculated without data from tissues such as (but not limited to): central nervous system, brain, eye, and/or testis tissues. In some embodiments, an address target as provided herein identifies a cell state. As used herein a “cell state” refers to a given physiological condition of a cell. A cell state may be, e.g., a disease state (relative to a non-disease state or normal state of a cell or tissue); or an activated state (relative to a non-activated state of a cell). Exemplary disease states include inflammation, infection (e.g., bacterial, viral, or fungal infection), and states relating to cancer (e.g., precancerous or cancerous cell states). In some aspects, cell state reflects the fact that cells of a particular type can exhibit variability with regard to one or more features and/or can exist in a variety of different conditions, while retaining the features of their particular cell type and not gain features that would cause them to be classified as a different cell type. The different states or conditions in which a cell can exist may be characteristic of a particular cell type (e.g., may involve properties or characteristics exhibited only by that cell type and/or involve functions performed only or primarily by that cell type) or may occur in multiple different cell types. In some embodiments, a cell state reflects the capability of a cell to respond to a particular stimulus or environmental condition (e.g., whether or not the cell will respond, or the type of response that will be elicited) or is a condition of the cell brought about by a stimulus or environmental condition. Cells in different cell states may be distinguished from one another in a variety of ways. For example, they may express, produce, or secrete one or more different genes, proteins, or other molecules ("markers", such as the address targets provided herein), exhibit differences in protein modifications such as phosphorylation, acetylation, etc., or may exhibit differences in appearance. Thus a cell state may be a condition of the cell in which the cell expresses, produces, or secretes one or more markers, exhibits particular protein modification(s), has a particular appearance, and/or will or will not exhibit one or more biological response(s) to a stimulus or environmental condition.
C. Conditional Signaling
As a general principle, an effector target binding domain (e.g., an IL-22 polypeptide or fragment or variant thereof, e.g., a detuned or attenuated IL-22 or fragment or variant thereof (e.g., an IL-22 mutein polypeptide), e.g., as described in Section B(i)(a), above) may not substantially signal, or may not signal at all, when it is not localized by an address target binding domain. In embodiments, an effector target binding domain signals with higher potency (e.g., has higher avidity) when it is localized by an address target binding domain compared to the signal when it is not localized by an address target binding domain. When an effector target binding domain is localized to a targeted cell or tissue by an address target binding domain as part of the same macromolecule, effector target signaling can be influenced as discussed above.
/. Relative Binding Affinity of First and Second Binding Sites
The affinity of the effector target binding site and address target binding site of an ANDbody for their respective binding partners may differ. In some embodiments the affinity of the first binding site to the therapeutic effector target it binds is weaker than the affinity of the second binding site to the address target. In some embodiments the affinity of the first binding site to the therapeutic effector target it binds is more than 2-fold, 3-fold, 4-fold, 5-fold, 10-fold, 15-fold, 20-fold, 50-fold, 100-fold, 200-fold, 500-fold, 1000-fold weaker than the affinity of the second binding site to the address target.
The terms "binding affinity" and “binding activity" refer to the tendency of a macromolecule, e.g., a polypeptide molecule, to bind or not to bind to a target. For purposes of the present invention, which combines two binding sites, the relative affinities of the two binding sites can be determined by, for example, measuring their respective affinities when each binding site is present on a common scaffold, such as in the form of a single chain antibody. Such a comparison allows a comparison of the affinities of two binding sites while eliminating any interference from other binding sites present on the macromolecule of the present invention.
Binding affinity may be quantified by determining the dissociation constant (Kd; KD) for a polypeptide and its binder. A lower Kd is indicative of a higher affinity for a binding partner. Similarly, the specificity of binding of a polypeptide to its binding partner may be defined in terms of the comparative dissociation constants (Kd) of the polypeptide for its binding partner as compared to the dissociation constant with respect to the polypeptide and another, non-target molecule.
The value of this dissociation constant can be determined by known methods. For example, the Kd may be established using a double-filter nitrocellulose filter binding assay such as that disclosed by Wong & Lohman (Proc. Natl. Acad. Sci. USA 90, 5428-5432, 1993). Other standard assays to evaluate the binding ability of ligands such as antibodies towards targets are known in the art, including for example, ELISAs, Western blots, RIAs, and flow cytometry analysis. The binding kinetics (e.g., binding affinity) of the antibody also can be assessed by standard assays known in the art, such as by Biacore™ system analysis.
As an alternative to Kd, ECso or IC50 may be used to determine relative affinities. In this context EC50 indicates the concentration at which a polypeptide achieves 50% of its maximum binding to a fixed quantity of binding partner. IC50 indicates the concentration at which a polypeptide inhibits 50% of the maximum binding of a fixed quantity of competitor to a fixed quantity of binding partner. In both cases, a lower level of EC50 or IC50 indicates a higher affinity for a target. The EC50 and IC50 values of an ANDbody binding site for its binding partner can both be determined by well-known methods, for example ELISA.
In some embodiments the Kd of therapeutic effector target binder might be higher than about 1 pM, about 10pM, about 10OpM, about 1 nM, about 10nM, about 10OnM, about 500nM, or about 1 uM (e.g., may be between 1 pM and 10pM, between 10 pM and 100pM, between 100 pM and 1 nM, between 1 nM and 10 nM, between 10 nM and 100 nM, between 100 nM and 500nM, or between 500 nM and 1 uM). In some embodiments the Kd of the address target binder might be less than about 1 uM, about 500nM, about 10OnM, about 10nM, about 1 nM, about 10OpM, about 10pM, or about 1 pM (e.g., may be
between 1 uM and 500nM, between 500 nM and 10OnM, between 100 nM and 10nM, between 10 nM and 1 nM, between 1 nM and 10OpM, between 100 pM and 10 pM, or between 10pM and 1 pM). In some embodiments, the Kd for the therapeutic effector target binder may be about 6-fold, about 5-fold, about 4- fold, about 3-fold, or about 2-fold higher than the Kd for the address target binder.
In some embodiments the ECso of therapeutic effector target binder might be higher than about 1 pM, about 10pM, about 10OpM, about 1 nM, about 10nM, about 10OnM, about 500nM, or about 1 uM (e.g., may be between 1 pM and 10pM, between 10 pM and 100pM, between 100 pM and 1 nM, between 1 nM and 10 nM, between 10 nM and 100 nM, between 100 nM and 500nM, or between 500 nM and 1 uM). In some embodiments the ECso of the address target binder might be less than about 1 uM, about 500nM, about 10OnM, about 10nM, about 1 nM, about 10OpM, about 10pM, or about 1 pM (e.g., may be between 1 uM and 500nM, between 500 nM and 10OnM, between 100 nM and 10nM, between 10 nM and 1 nM, between 1 nM and 10OpM, between 100 pM and 10 pM, or between 10pM and 1 pM). In some embodiments, the ECso for the therapeutic effector target binder may be about 6-fold, about 5-fold, about 4-fold, about 3-fold, or about 2-fold higher than the ECso for the address target binder.
In some embodiments the IC50 of therapeutic effector target binder might be higher than about 1 pM, about 10pM, about 10OpM, about 1 nM, about 10nM, about 10OnM, about 500nM, or about 1 uM (e.g., may be between 1 pM and 10pM, between 10 pM and 100pM, between 100 pM and 1 nM, between 1 nM and 10 nM, between 10 nM and 100 nM, between 100 nM and 500nM, or between 500 nM and 1 uM). In some embodiments the IC50 of the address target binder might be less than about 1 uM, about 500nM, about 10OnM, about 10nM, about 1 nM, about 10OpM, about 10pM, or about 1 pM (e.g., may be between 1 uM and 500nM, between 500 nM and 10OnM, between 100 nM and 10nM, between 10 nM and 1 nM, between 1 nM and 10OpM, between 100 pM and 10 pM, or between 10pM and 1 pM). In some embodiments, the IC50 for the therapeutic effector target binder may be about 6-fold, about 5-fold, about 4-fold, about 3-fold, or about 2-fold higher than the IC50 for the address target binder.
The cellular or tissue density of the effector target and address target bound by an ANDbody may differ. In embodiments, the density of the therapeutic effector target on a cell bound by the effector target binding site of an ANDbody is more than about 2-fold, about 3-fold, about 4-fold, about 5-fold, about 10-fold, about 15-fold, about 20-fold, about 50-fold, about 100-fold, about 200-fold, about 500-fold, about 1000-fold, about 10,000-fold, about 100,000-fold less than the density of the address target on a cell bound by the address target binding site.
In some embodiments, the affinity of the first binding site to the therapeutic effector target it binds is about one-half (1/2) X Kd less than the affinity of the second binding site to the address target it binds and the density of the therapeutic effector target on a cell bound by the first binding site is about one-half (I/2) X Kd less than the density of the address target on a cell bound by the second binding site.
In some embodiments, the ANDbody has both the affinity and density parameters as described hereinabove.
ANDbodies with binding sites with altered affinities can be made using methods known in the art, e.g., an ANDbody can be engineered to have a target binding site that has decreased affinity for the effector target. See, e.g., US Patent No. 10,654,928. In general, an ANDbody may be modified to alter the affinity of an effector target binding site to its effector target or to alter the affinity of an address target
binding site to its address target. The modification can increase or decrease affinity for the binding site’s binding partner.
D. Small Molecules
A macromolecule of the invention (for example, an ANDbody and/or an IL-22 mutein polypeptide) may be linked to a small molecule. The macromolecule and the small molecule may be linked by a cleavable linker. Alternatively, the macromolecule and the small molecule may be linked by a non- cleavable linker. Any useful linker may be employed for this purpose.
One or more (for example, one, two, three, four, five, or more) small molecules may be linked to the macromolecule. If multiple small molecules are linked to a macromolecule, the small molecules may be the same. Alternatively, one or more of the small molecules linked to the macromolecule may be different.
A small molecule to be linked to the macromolecule may be any desired small molecule. For example, the small molecule may be a therapeutic agent of interest that is to be localized or concentrated at a particular site by the macromolecule. In one example, the small molecule may be a therapeutic agent that acts together with or complements the effector target binding site domain. Alternatively, the small molecule may modulate the effector target binding site domain. In another example, the small molecule may modulate the address target binding site domain.
Small molecules may be conjugated to a macromolecule of the invention using any conjugation technique known in the art. For example, small molecule carboxy, hydroxyl, and amine residues may be joined to amine and sulfhydryl residues on proteins using linkage techniques. Alternatively, any complementary functional groups on the two components may be used to react with each other to form a covalent bond. Examples of complementary reactive functional groups include, but are not limited to, e.g., maleimide and cysteine, amine and activated carboxylic acid, thiol and maleimide, activated sulfonic acid and amine, isocyanate and amine, azide and alkyne, and alkene and tetrazine. Further, any available linker may be utilized in the invention including heterobifunctional linkers that allow attachment of small molecules through, for example, disulfide bonds and amide bonds.
E. Production of Compositions
Production of polypeptides
Polypeptide components of the macromolecules (ANDbodies and/or IL-22 mutein polypeptides) of the invention may be produced by any suitable means. For example, all or part of an ANDbody or an IL-22 mutein polypeptide may be expressed by a host cell comprising one or more nucleotides which encode the ANDbody or IL-22 mutein polypeptide. Accordingly, in some embodiments, the invention provides one or more nucleic acids encoding any one of the macromolecules provided herein or one or more subunits thereof. The nucleic acids may be, e.g., linear nucleic acids, circular nucleic acids, and/or modified nucleic acids. The invention further provides one or more vectors comprising the one or more nucleic acids encoding any one of the macromolecules provided herein or one or more subunits thereof.
The invention also provides one or more host cells (e.g., mammalian host cells) comprising one or more nucleic acids encoding any one of the macromolecules provided herein or one or more subunits
thereof (e.g., comprising vectors comprising said one or more nucleic acids). The invention further provides a method of producing any one of the macromolecules provided herein, the method comprising culturing the one or more host cells in a culture medium. The method may further comprise recovering the macromolecule from the one or more host cells or the culture medium.
Such methods of making a therapeutic polypeptide are routine in the art. See, in general, Smales & James (Eds.), Therapeutic Proteins: Methods and Protocols (Methods in Molecular Biology), Humana Press (2005); and Crommelin, Sindelar & Meibohm (Eds.), Pharmaceutical Biotechnology: Fundamentals and Applications, Springer (2013).
Methods for producing an ANDbody and/or an IL-22 mutein polypeptide may involve expression in mammalian cells, although recombinant proteins can also be produced using insect cells, yeast, bacteria, or other cells under the control of appropriate promoters. Mammalian expression vectors may comprise nontranscribed elements such as an origin of replication, a suitable promoter and enhancer, and other 5' or 3' flanking nontranscribed sequences, and 5' or 3' nontranslated sequences such as necessary ribosome binding sites, a polyadenylation site, splice donor and acceptor sites, and termination sequences. DNA sequences derived from the SV40 viral genome, for example, SV40 origin, early promoter, enhancer, splice, and polyadenylation sites may be used to provide the other genetic elements required for expression of a heterologous DNA sequence. Appropriate cloning and expression vectors for use with bacterial, fungal, yeast, and mammalian cellular hosts are described in Green & Sambrook, Molecular Cloning: A Laboratory Manual (Fourth Edition), Cold Spring Harbor Laboratory Press (2012).
Various mammalian cell culture systems can be employed to express and manufacture an ANDbody and/or an IL-22 mutein polypeptide described herein. Examples of mammalian expression systems include CHO cells, COS cells, HeLA and BHK cell lines. Processes of host cell culture for production of protein therapeutics are described in, e.g., Zhou and Kantardjieff (Eds.), Mammalian Cell Cultures for Biologies Manufacturing (Advances in Biochemical Engineering/Biotechnology), Springer (2014). Purification of protein therapeutics is described in Franks, Protein Biotechnology: Isolation, Characterization, and Stabilization, Humana Press (2013); and in Cutler, Protein Purification Protocols (Methods in Molecular Biology), Humana Press (2010). Formulation of protein therapeutics is described in Meyer (Ed.), Therapeutic Protein Drug Products: Practical Approaches to formulation in the Laboratory, Manufacturing, and the Clinic, Woodhead Publishing Series (2012).
Antibody production techniques are known. See, for example, Zhiqiang (Editor), Therapeutic Monoclonal Antibodies: From Bench to Clinic. 1st Edition. Wiley 2009; Greenfield (Ed.) Antibodies: A Laboratory Manual. (Second edition) Cold Spring Harbor Laboratory Press 2013; Ferrara et al. 2012. Using Phage and Yeast Display to Select Hundreds of Monoclonal Antibodies: Application to Antigen 85, a Tuberculosis Biomarker. PLoS ONE 7(11 ): e49535, for methods of making recombinant antibodies, including antibody engineering, use of degenerate oligonucleotides, 5'-RACE, phage display, and mutagenesis; antibody testing and characterization; antibody pharmacokinetics and pharmacodynamics; antibody purification and storage; and screening and labeling techniques.
Production of ANDbody and IL-22 mutein polypeptide RNAs
In some embodiments, ANDbody and/or IL-22 mutein polypeptide RNAs (e.g., RNAs encoding one
or more components of an ANDbody and/or an IL-22 mutein polypeptide) may be produced, e.g., for delivery to a subject. Generally, therapeutic mRNAs are made by in vitro transcription. Modification such as incorporation of modified bases, 5’cap analogues, and polyA tails can optimize activity and function. For example, translation and stability of mRNA can be accomplished, by cap and poly A tail modifications. E.g., incorporation of cap analogs such as ARCA (anti-reverse cap analogs) and a poly(A) tail of 100-200 bp into in vitro transcribed (IVT) mRNAs improves expression and stability (Kaczmarek et al. Genome Medicine (2017) 9:60). New types of cap analogs, such as 1 ,2-dithiodiphosphate-modified caps, can further improve efficiency of translation (Strenkowska et al. Nucleic Acids Res. 2016;44:9578-90). Codon optimization can also improve efficacy of protein synthesis and limit mRNA destabilization by rare codons (Presnyak et al. Cell. 2015;160:1111-24. 93; Thess et al. Mol Ther. 2015;23: 1456-64). Modifying 3' and 5' untranslated regions (UTRs), which contain sequences responsible for recruiting RNA-binding proteins (RBPs) and miRNAs, can enhance the level of protein product (Kaczmarek). Further, UTRs can be modified to encode regulatory elements (e.g., K-turn motifs and miRNA binding sites), in order to control RNA expression in a cell-specific manner (Wroblewska et al. Nat Biotechnol. 2015;33:839-41 ). RNA base modifications (e.g., pseudouridine incorporated mRNA, e.g., N1 -methyl-pseudouridine) contribute to masking mRNA immune-stimulatory activity and increase mRNA translation by enhancing translation initiation (Andries et al. J Control Release. 2015;217:337-44; Svitkin et al. Nucleic Acids Res.
2017;45:6023-36). mRNA compositions and methods of their manufacture are known and are disclosed, e.g., in WO2016011306; WO2016014846; WO2016022914; WO2016077123; WO2016164762;
WO2016201377; WO2017049275; US9937233; US8710200; US10022425; US9878056; US9572897; Jemielity et al. RNA. 2003;9:1108-22. 90; Mockey et al. Biochem Biophys Res Commun. 2006;340:1062- 8. 91 ; Strenkowska et al. Nucleic Acids Res. 2016;44:9578-90. 92; Presnyak et al. Cell. 2015;160:1111 — 24. 93; Kaczmarek et al. Genome Medicine (2017) 9:60.
Assessment of Targets and Addresses
Expression of a therapeutic target (e.g., an IL-22 receptor) and/or an address target (e.g., an IL-10 family address target) can be assessed at either the RNA or protein level using methods known in the art. In embodiments, expression of the therapeutic target is assessed by measuring RNA expression, e.g., using an RNA sequence dataset as a proxy for protein expression levels. RNA datasets include those a genotype-Tissue Expression (GTEx) dataset (see, e.g., https://www.genome.gov/Funded-Programs- Projects/Genotype-Tissue-Expression-Project) or a Human Protein Atlas (HPA) dataset (https //www . prote i n atl as . o rg/) .
A non-limiting list of tissues in which expression of the therapeutic target can be assessed includes, e.g., the pancreas, adrenal gland, liver, kidney (cortex), kidney (medulla), gastrointestinal (Gl) tract, lower Gl tract, small intestine colon, transverse colon, sigmoid colon, terminal ileum, skin, non-sun- exposed skin, sun-exposed skin, spleen, stomach,.
Address markers can be assessed using methods well known in the art, e.g., gene expression can be assessed at the mRNA level using Northern blots, cDNA or oligonucleotide microarrays, or sequencing {e.g., RNA-Seq), or at the level of protein expression using protein microarrays, Western blots, flow cytometry, immunohistochemistry, etc. Modifications can be assessed, e.g., using antibodies that are specific for a particular modified form of a protein, e.g., phospho-specific antibodies, or mass spectrometry.
F. Uses of ANDbody molecules and IL-22 mutein polypeptides
ANDbody molecules (e.g., ANDbodies), IL-22 mutein polypeptides, and their pharmaceutical compositions provided herein are suitable for administration to a subject in need thereof, wherein the subject is a human or a non-human animal, for example, suitable for human therapeutic or veterinary use. Accordingly, in some aspects, provided herein is a method comprising administering a therapeutically effective amount of any one of the macromolecules (i.e., ANDbody molecules, ANDbodies, or IL-22 mutein polypeptides) provided herein, a composition comprising the same, or one or more nucleic acids encoding the same to a subject in need thereof.
Veterinary use includes use for treatment of mammals, including commercially relevant mammals, e.g., pet and live-stock animals, such as cattle, pigs, horses, sheep, goats, cats, dogs, mice, and/or rats; and/or birds, including commercially relevant birds such as parrots, poultry, chickens, ducks, geese, hens or roosters and/or turkeys; zoo animals, e.g., a feline; non-mammal animals, e.g., reptiles, fish, amphibians, etc.
The invention is further directed to a subject or subject cell comprising the ANDbody or IL-22 mutein polypeptide composition described herein. In some embodiments, the subject or subject cell is a plant, insect, bacteria, fungus, vertebrate, mammal (e.g., human), or other organism or cell.
In some embodiments, a subject or a subject cell is contacted with (e.g., delivered to or administered to) the ANDbody composition or IL-22 mutein polypeptide. In some embodiments, the subject is a mammal, such as a human. The amount of the ANDbody composition or IL-22 mutein polypeptide, expression product, or both in the subject can be measured at any time after administration.
In some embodiments, a subject to which an ANDbody, ANDbody composition, or IL-22 mutein polypeptide provided herein is administered has, is suspected of having, or is at increased risk of developing a disease or disorder selected from Crohn's disease, ulcerative colitis, graft-vs-host disease, atopic dermatitis, psoriasis, wounds, diabetic skin ulcers, acute pancreatitis, chronic pancreatitis, autoimmune pancreatitis, nephritis, acute lung injury, viral lung injury, fibrosis, fibrosis, non-alcoholic steatohepatitis, viral hepatitis, and autoimmune hepatitis.
In some aspects, provided herein is a method of potentiating activation of a signaling pathway (e.g., a cytokine signaling pathway) in a tissue or cell, the method comprising contacting the tissue or cell with a macromolecule provided herein, wherein the tissue or cell expresses an IL-10 family receptor and an IL-10 family address target, and wherein neither the first binding site (IL-22 polypeptide or fragment or variant thereof) nor the second binding site (IL-10 family address target binder) substantially activates the signaling pathway in the absence of localization by the second binding site; wherein upon contacting the tissue or cell with the macromolecule, activation of the signaling pathway in the tissue or cell by the first binding site is substantially increased relative to a reference macromolecule lacking the second binding site.
In some aspects, provided herein is a method of modulating IL-22 receptor signaling in a target cell of a subject, the method comprising administering to the subject any one of the macromolecules (e.g., ANDbodies and/or IL-22 mutein polypeptides) provided herein.
G. Pharmaceutical Compositions
Polypeptide Pharmaceutical Compositions
The ANDbody and IL-22 mutein polypeptide compositions described herein {e.g., ANDbody and/or IL-22 mutein polypeptide, nucleic acid, or RNA compositions) and the polypeptide compositions described herein may be administered to a subject in need thereof. The invention includes pharmaceutical compositions that include a macromolecule provided herein (i.e. , an ANDbody or IL-22 mutein polypeptide), e.g., combination in with one or more pharmaceutically acceptable carriers or excipients. The invention includes pharmaceutical compositions that include a polypeptide provided herein, e.g., in combination with one or more pharmaceutically acceptable carriers or excipients. In some embodiments, the composition is a unit dosage form suitable for administration to a human subject (e.g., intravenous, oral, or subcutaneous administration).
Formulation of protein therapeutics is routine. See, for example, Ribeiro et al., Insights on the Formulation of Recombinant Proteins. Adv Biochem Eng Biotechnol. 2020;171 :23-54. doi: 10.1007/10_2019_119. PMID: 31844925.
RNA pharmaceutical compositions
Nucleic acids {e.g., RNA) encoding an ANDbody and/or an IL-22 mutein polypeptide can alternatively or additionally be administered to a subject. Generally, therapeutic mRNAs are made by in vitro transcription. Modification such as incorporation of modified bases, 5’cap analogues, and polyA tails can optimize activity and function. For example, translation and stability of mRNA can be accomplished, by cap and poly A tail modifications. E.g., incorporation of cap analogs such as ARCA (anti-reverse cap analogs) and a poly(A) tail of 100-200 bp into in vitro transcribed (IVT) mRNAs improves expression and stability (Kaczmarek et al. Genome Medicine (2017) 9:60). New types of cap analogs, such as 1 ,2- dithiodiphosphate-modified caps, can further improve efficiency of translation (Strenkowska et al. Nucleic Acids Res. 2016;44:9578-90). Codon optimization can also improve efficacy of protein synthesis and limit mRNA destabilization by rare codons (Presnyak et al. Cell. 2015;160:1111-24. 93; Thess et al. Mol Ther. 2015;23: 1456-64). Modifying 3' and 5' untranslated regions (UTRs), which contain sequences responsible for recruiting RNA-binding proteins (RBPs) and miRNAs, can enhance the level of protein product (Kaczmarek). Further, UTRs can be modified to encode regulatory elements (e.g., K-turn motifs and miRNA binding sites), in order to control RNA expression in a cell-specific manner (Wroblewska et al. Nat Biotechnol. 2015;33:839-41 ). RNA base modifications (e.g., pseudouridine incorporated mRNA, e.g., N1 - methyl-pseudouridine) contribute to masking mRNA immune-stimulatory activity and increase mRNA translation by enhancing translation initiation (Andries et al. J Control Release. 2015;217:337-44; Svitkin et al. Nucleic Acids Res. 2017;45:6023-36). mRNA compositions and methods of their manufacture are known and are disclosed, e.g., in WO2016011306; WO2016014846; WO2016022914; WO2016077123; WO2016164762; WO2016201377; WO2017049275; US9937233; US8710200; US10022425; US9878056; US9572897; Jemielity et al. RNA. 2003;9:1108-22. 90; Mockey et al. Biochem Biophys Res Commun. 2006;340:1062-8. 91 ; Strenkowska et al. Nucleic Acids Res. 2016;44:9578-90. 92; Presnyak et al. Cell. 2015;160:1111 -24. 93; Kaczmarek et al. Genome Medicine (2017) 9:60.
In embodiments, the RNA is a circular RNA. See, for example, WO2019118919, describing the expression of a therapeutic RNA, such as an antibody RNA, from a circular RNA. In some embodiments, the invention includes a circular polyribonucleotide that comprises (a) an internal ribosome entry site (IRES), (b) an expression sequence encoding a ANDbody described herein and lacking a poly-A sequence, and (c) a termination element. A circular RNA encoding an ANDbody described herein may be delivered naked (i.e. , without formulation with a carrier) or with a carrier.
Combination Therapies
In some embodiments, an ANDbody or ANDbody composition provided herein is administered in combination with one or more additional therapeutic agents. In some embodiments, an IL-22 mutein polypeptide or IL-22 mutein polypeptide composition provided herein is administered in combination with one or more additional therapeutic agents.
Carriers
Lipid nanoparticles
Formulations of the compositions described herein (e.g., polypeptide or RNA ANDbody or IL-22 mutein polypeptide compositions or polypeptides) for in vivo delivery with a carrier include lipid nanoparticle (LNP) formulations. See, e.g., US Pat. 9,764,036; US Pat. 9,682,139; Kauffman et al. Nano Lett. 2015;15: 7300-6. 37; Fenton et al. Adv Mater. 2016;28:2939-43). LNPs, in some embodiments, comprise one or more ionic lipids, such as non-cationic lipids (e.g., neutral or anionic, or zwitterionic lipids); one or more conjugated lipids (such as PEG-conjugated lipids or lipids conjugated to polymers described in Table 5 of WO2019217941 ; incorporated herein by reference in its entirety); one or more sterols (e.g., cholesterol); and, optionally, one or more targeting molecules (e.g., conjugated receptors, receptor ligands, antibodies); or combinations of the foregoing.
Lipids that can be used in nanoparticle formations {e.g., lipid nanoparticles) include, for example those described in Table 4 of WO2019217941 , which is incorporated herein by reference — e.g., a lipid- containing nanoparticle can comprise one or more of the lipids in Table 4 of WO2019217941 . Lipid nanoparticles can include additional elements, such as polymers, such as the polymers described in Table 5 of WO2019217941 , incorporated by reference.
In some embodiments, conjugated lipids, when present, can include one or more of PEG- diacylglycerol (DAG) (such as l-(monomethoxy-polyethyleneglycol)-2,3- dimyristoylglycerol (PEG-DMG)), PEG-dialkyloxypropyl (DAA), PEG-phospholipid, PEG- ceramide (Cer), a pegylated phosphatidylethanoloamine (PEG-PE), PEG succinate diacylglycerol (PEGS-DAG) (such as 4-0-(2',3'- di(tetradecanoyioxy)propyi-l-0-(w- methoxy(polyethoxy)ethyl) butanedioate (PEG-S-DMG)), PEG dialkoxypropylcarbam, N- (carbonyl-methoxypoly ethylene glycol 2000)- 1 ,2-distearoyl-sn-glycero-3- phosphoethanolamine sodium salt, and those described in Table 2 of WO2019051289 (incorporated by reference), and combinations of the foregoing.
In some embodiments, sterols that can be incorporated into lipid nanoparticles include one or more of cholesterol or cholesterol derivatives, such as those in W02009/127060 or US2010/0130588, which are incorporated by reference. Additional exemplary sterols include phytosterols, including those described in Eygeris et al (2020), dx.doi.org/10.1021/acs.nanolett.0c01386, incorporated herein by reference.
In some embodiments, the lipid particle comprises an ionizable lipid, a non-cationic lipid, a conjugated lipid that inhibits aggregation of particles, and a sterol. The amounts of these components can be varied independently and to achieve desired properties. For example, in some embodiments, the lipid nanoparticle comprises an ionizable lipid is in an amount from about 20 mol % to about 90 mol % of the total lipids (in other embodiments it may be 20-70% (mol), 30-60% (mol) or 40-50% (mol); about 50 mol % to about 90 mol % of the total lipid present in the lipid nanoparticle), a non-cationic lipid in an amount from about 5 mol % to about 30 mol % of the total lipids, a conjugated lipid in an amount from about 0.5 mol % to about 20 mol % of the total lipids, and a sterol in an amount from about 20 mol % to about 50 mol % of the total lipids. The ratio of total lipid to nucleic acid can be varied as desired. For example, the total lipid to nucleic acid (mass or weight) ratio can be from about 10: 1 to about 30: 1 .
In some embodiments, the lipid to nucleic acid ratio (mass/mass ratio; w/w ratio) can be in the range of from about 1 : 1 to about 25: 1 , from about 10: 1 to about 14: 1 , from about 3 : 1 to about 15: 1 , from about 4: 1 to about 10: 1 , from about 5: 1 to about 9: 1 , or about 6: 1 to about 9: 1 . The amounts of lipids and nucleic acid can be adjusted to provide a desired N/P ratio, for example, N/P ratio of 3, 4, 5, 6, 7, 8, 9, 10 or higher. Generally, the lipid nanoparticle formulation’s overall lipid content can range from about 5 mg/ml to about 30 mg/mL.
Some non-limiting example of lipid compounds that may be used (e.g., in combination with other lipid components) to form lipid nanoparticles for the delivery of compositions described herein, e.g., nucleic acid (e.g., RNA) described herein includes,
(i)
In some embodiments an LNP comprising Formula (i) is used to deliver an ANDbody RNA composition described herein to the liver and/or hepatocyte cells.
In some embodiments an LNP comprising Formula (ii) is used to deliver an ANDbody RNA composition described herein to the liver and/or hepatocyte cells.
In some embodiments an LNP comprising Formula (iii) is used to deliver an ANDbody RNA composition described herein to the liver and/or hepatocyte cells.
(iv)
In some embodiments an LNP comprising Formula (v) is used to deliver an ANDbody RNA composition described herein to the liver and/or hepatocyte cells.
In some embodiments an LNP comprising Formula (vi) is used to deliver an ANDbody RNA composition described herein to the liver and/or hepatocyte cells.
In some embodiments an LNP comprising Formula (viii) is used to deliver an ANDbody RNA composition described herein to the liver and/or hepatocyte cells.
In some embodiments an LNP comprising Formula (ix) is used to deliver an ANDbody RNA composition described herein to the liver and/or hepatocyte cells.
wherein
X1 is O, NR1 , or a direct bond, X2 is C2-5 alkylene, X3 is C(=O) or a direct bond, R1 is H or Me, R3 is Ci-3 alkyl, R2 is Ci-3 alkyl, or R2 taken together with the nitrogen atom to which it is attached and 1 -3 carbon atoms of X2 form a 4-, 5-, or 6-membered ring, or X1 is NR1 , R1 and R2 taken together with the nitrogen atoms to which they are attached form a 5- or 6-membered ring, or R2 taken together with R3 and the nitrogen atom to which they are attached form a 5-, 6-, or 7-membered ring, Y1 is C2-12 alkylene, Y2 is selected from
, (in either orientation),
, p at if Z1 is a direct bond, Z2 is absent;
R5 is C5-9 alkyl or C6-10 alkoxy, R6 is C5-9 alkyl or C6-10 alkoxy, W is methylene or a direct bond, and R7 is H or Me, or a salt thereof, provided that if R3 and R2 are C2 alkyls, X1 is O, X2 is linear C3 alkylene, X3 is C(=0), Y1 is linear Ce alkylene, (Y2 )n-R4 is
R4 is linear C5 alkyl, Z1 is C2 alkylene, Z2 is absent, W is methylene, and R7 is H, then R5 and R6 are not Cx alkoxy.
In some embodiments an LNP comprising Formula (xii) is used to deliver an ANDbody RNA composition described herein to the liver and/or hepatocyte cells.
In some embodiments an LNP comprising Formula (xi) is used to deliver an ANDbody RNA composition described herein to the liver and/or hepatocyte cells.
(xiv)
In some embodiments an LNP comprises a compound of Formula (xiii) and a compound of Formula (xiv).
In some embodiments an LNP comprising Formula (xv) is used to deliver an ANDbody RNA composition described herein to the liver and/or hepatocyte cells.
In some embodiments an LNP comprising a formulation of Formula (xvi) is used to deliver an
ANDbody RNA composition described herein to the lung endothelial cells.
In some embodiments, a lipid compound used to form lipid nanoparticles for the delivery of compositions described herein, e.g., nucleic acid (e.g., RNA) described herein is made by one of the following reactions:
In some embodiments, a composition described herein (e.g., a nucleic acid or a protein) is provided in an LNP that comprises an ionizable lipid. In some embodiments, the ionizable lipid is heptadecan-9-yl 8-((2-hydroxyethyl)(6-oxo-6-(undecyloxy)hexyl)amino)octanoate (SM-102); e.g., as described in Example 1 of US9,867,888 (incorporated by reference herein in its entirety). In some embodiments, the ionizable lipid is 9Z,12Z)-3-((4,4-bis(octyloxy)butanoyl)oxy)-2-((((3- (diethylamino)propoxy)carbonyl)oxy)methyl)propyl octadeca-9,12-dienoate (LP01 ), e.g., as synthesized in Example 13 of WO2015/095340 (incorporated by reference herein in its entirety). In some embodiments, the ionizable lipid is Di((Z)-non-2-en-1 -yl) 9-((4-dimethylamino)butanoyl)oxy)heptadecanedioate (L319), e.g. as synthesized in Example 7, 8, or 9 of US2012/0027803 (incorporated by reference herein in its entirety). In some embodiments, the ionizable lipid is 1 ,1 '-((2-(4-(2-((2-(Bis(2- hydroxydodecyl)amino)ethyl)(2-hydroxydodecyl) amino)ethyl)piperazin-1 -yl)ethyl)azanediyl)bis(dodecan-2- ol) (C12-200), e.g., as synthesized in Examples 14 and 16 of WO2010/053572 (incorporated by reference herein in its entirety). In some embodiments, the ionizable lipid is Imidazole cholesterol ester (ICE) lipid (3S, 10R, 13R, 17R)-10, 13-dimethyl-17- ((R)-6-methylheptan-2-yl)-2, 3, 4, 7, 8, 9, 10, 1 1 , 12, 13, 14, 15, 16, 17-tetradecahydro-IH- cyclopenta[a]phenanthren-3-yl 3-(1 H-imidazol-4-yl)propanoate, e.g., Structure (I) from W02020/106946 (incorporated by reference herein in its entirety).
In some embodiments, an ionizable lipid may be a cationic lipid, an ionizable cationic lipid, e.g., a cationic lipid that can exist in a positively charged or neutral form depending on pH, or an amine-containing lipid that can be readily protonated. In some embodiments, the cationic lipid is a lipid capable of being positively charged, e.g., under physiological conditions. Exemplary cationic lipids include one or more amine group(s) which bear the positive charge. In some embodiments, the lipid particle comprises a cationic lipid in formulation with one or more of neutral lipids, ionizable amine-containing lipids, biodegradable alkyne lipids, steroids, phospholipids including polyunsaturated lipids, structural lipids (e.g., sterols), PEG, cholesterol and polymer conjugated lipids. In some embodiments, the cationic lipid may be an ionizable cationic lipid. An exemplary cationic lipid as disclosed herein may have an effective pKa over 6.0. In embodiments, a lipid nanoparticle may comprise a second cationic lipid having a different effective
pKa (e.g., greater than the first effective pKa), than the first cationic lipid. A lipid nanoparticle may comprise between 40 and 60 mol percent of a cationic lipid, a neutral lipid, a steroid, a polymer conjugated lipid, and a therapeutic agent, e.g., a nucleic acid (e.g., RNA) described herein, encapsulated within or associated with the lipid nanoparticle. In some embodiments, the nucleic acid is co-formulated with the cationic lipid. The nucleic acid may be adsorbed to the surface of an LNP, e.g., an LNP comprising a cationic lipid. In some embodiments, the nucleic acid may be encapsulated in an LNP, e.g., an LNP comprising a cationic lipid. In some embodiments, the lipid nanoparticle may comprise a targeting moiety, e.g., coated with a targeting agent. In embodiments, the LNP formulation is biodegradable. In some embodiments, a lipid nanoparticle comprising one or more lipid described herein, e.g., Formula (i), (ii), (ii), (vii) and/or (ix) encapsulates at least 1 %, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, at least 50%, at least 60%, at least 70%, at least 80%, at least 90%, at least 92%, at least 95%, at least 97%, at least 98% or 100% of an RNA molecule.
Exemplary ionizable lipids that can be used in lipid nanoparticle formulations include, without limitation, those listed in Table 1 of WO2019051289, incorporated herein by reference. Additional exemplary lipids include, without limitation, one or more of the following formulae: X of US2016/031 1759; I of US201503761 15 or in US2016/0376224; I, II or III of US20160151284; I, IA, II, or HA of US20170210967; l-c of US20150140070; A of US2013/0178541 ; I of US2013/0303587 or US2013/0123338; I of US2015/0141678; II, III, IV, or V of US2015/0239926; I of US2017/01 19904; I or II of WO2017/1 17528; A of US2012/0149894; A of US2015/0057373; A of WO2013/1 16126; A of US2013/0090372; A of US2013/0274523; A of US2013/0274504; A of US2013/0053572; A of W02013/016058; A of W02012/162210; I of US2008/042973; I, II, III, or IV of US2012/01287670; I or II of US2014/0200257; I, II, or III of US2015/0203446; I or III of US2015/0005363; I, IA, IB, IC, ID, II, HA, IIB, IIC, HD, or lll-XXIV of US2014/0308304; of US2013/0338210; I, II, III, or IV of W02009/132131 ; A of US2012/0101 1478; I or XXXV of US2012/0027796; XIV or XVII of US2012/0058144; of US2013/0323269;
I of US201 1/01 17125; I, II, or III of US201 1/0256175; I, II, III, IV, V, VI, VII, VIII, IX, X, XI, XII of US2012/0202871 ; I, II, III, IV, V, VI, VII, VIII, X, XII, XIII, XIV, XV, or XVI of US201 1/0076335; I or II of US2006/008378; I of US2013/0123338; I or X-A-Y-Z of US2015/0064242; XVI, XVII, or XVIII of US2013/0022649; I, II, or III of US2013/01 16307; I, II, or III of US2013/01 16307; I or II of US2010/0062967; l-X of US2013/0189351 ; I of US2014/0039032; V of US2018/0028664; I of US2016/0317458; I of US2013/0195920; 5, 6, or 10 of US10,221 , 127; HI-3 of WO2018/081480; I-5 or I-8 of W02020/081938; 18 or 25 of US9,867,888; A of US2019/0136231 ; II of W02020/219876; 1 of US2012/0027803; OF-02 of US2019/0240349; 23 of US10,086,013; cKK-E12/A6 of Miao et al (2020);
C12-200 of WO2010/053572; 7C1 of Dahlman et al (2017); 304-013 or 503-013 of Whitehead et al ; TS- P4C2 of US9,708,628; I of W02020/106946; I of W02020/106946.
In some embodiments, the ionizable lipid is MC3 (6Z,9Z,28Z,3 IZ)-heptatriaconta- 6,9,28,3 I- tetraen-l9-yl-4-(dimethylamino) butanoate (DLin-MC3-DMA or MC3), e.g., as described in Example 9 of WO2019051289A9 (incorporated by reference herein in its entirety). In some embodiments, the ionizable lipid is the lipid ATX-002, e.g., as described in Example 10 of WO2019051289A9 (incorporated by reference herein in its entirety). In some embodiments, the ionizable lipid is (l3Z,l6Z)-A,A-dimethyl-3- nonyldocosa-13, 16-dien-l-amine (Compound 32), e.g., as described in Example 1 1 of WO2019051289A9
(incorporated by reference herein in its entirety). In some embodiments, the ionizable lipid is Compound 6 or Compound 22, e.g., as described in Example 12 of WO2019051289A9 (incorporated by reference herein in its entirety).
Exemplary non-cationic lipids include, but are not limited to, distearoyl-sn-glycero- phosphoethanolamine, distearoylphosphatidylcholine (DSPC), dioleoylphosphatidylcholine (DOPC), dipalmitoylphosphatidylcholine (DPPC), dioleoylphosphatidylglycerol (DOPG), dipalmitoylphosphatidylglycerol (DPPG), dioleoyl-phosphatidylethanolamine (DOPE), palmitoyloleoylphosphatidylcholine (POPC), palmitoyloleoylphosphatidylethanolamine (POPE), dioleoylphosphatidylethanolamine 4-(N-maleimidomethyl)-cyclohexane- 1 - carboxylate (DOPE-mal), dipalmitoyl phosphatidyl ethanolamine (DPPE), dimyristoylphosphoethanolamine (DMPE), distearoyl-phosphatidyl- ethanolamine (DSPE), monomethyl-phosphatidylethanolamine (such as 16-O-monomethyl PE), dimethylphosphatidylethanolamine (such as 16-O-dimethyl PE), 18-l-trans PE, l-stearoyl-2-oleoyl- phosphatidyethanolamine (SOPE), hydrogenated soy phosphatidylcholine (HSPC), egg phosphatidylcholine (EPC), dioleoylphosphatidylserine (DOPS), sphingomyelin (SM), dimyristoyl phosphatidylcholine (DMPC), dimyristoyl phosphatidylglycerol (DMPG), distearoylphosphatidylglycerol (DSPG), dierucoylphosphatidylcholine (DEPC), palmitoyloleyolphosphatidylglycerol (POPG), dielaidoylphosphatidylethanolamine (DEPE), lecithin, phosphatidylethanolamine, lysolecithin, lysophosphatidylethanolamine, phosphatidylserine, phosphatidylinositol, sphingomyelin, egg sphingomyelin (ESM), cephalin, cardiolipin, phosphatidicacid, cerebrosides, dicetylphosphate, lysophosphatidylcholine, dilinoleoylphosphatidylcholine, or mixtures thereof. It is understood that other diacylphosphatidylcholine and diacylphosphatidylethanolamine phospholipids can also be used. The acyl groups in these lipids are preferably acyl groups derived from fatty acids having C10-C24 carbon chains, e.g., lauroyl, myristoyl, paimitoyl, stearoyl, or oleoyl. Additional exemplary lipids, in certain embodiments, include, without limitation, those described in Kim et al. (2020) dx.doi.org/10.1021/acs.nanolett.0c01386, incorporated herein by reference. Such lipids include, in some embodiments, plant lipids found to improve liver transfection with mRNA (e.g., DGTS).
Other examples of non-cationic lipids suitable for use in the lipid nanoparticles include, without limitation, nonphosphorous lipids such as, e.g., stearylamine, dodeeylamine, hexadecylamine, acetyl palmitate, glycerol ricinoleate, hexadecyl stereate, isopropyl myristate, amphoteric acrylic polymers, triethanolamine-lauryl sulfate, alkyl-aryl sulfate polyethyloxylated fatty acid amides, dioctadecyl dimethyl ammonium bromide, ceramide, sphingomyelin, and the like. Other non-cationic lipids are described in WO2017/099823 or US patent publication US2018/0028664, the contents of which is incorporated herein by reference in their entirety.
In some embodiments, the non-cationic lipid is oleic acid or a compound of Formula I, II, or IV of US2018/0028664, incorporated herein by reference in its entirety. The non-cationic lipid can comprise, for example, 0-30% (mol) of the total lipid present in the lipid nanoparticle. In some embodiments, the noncationic lipid content is 5-20% (mol) or 10-15% (mol) of the total lipid present in the lipid nanoparticle. In embodiments, the molar ratio of ionizable lipid to the neutral lipid ranges from about 2:1 to about 8:1 (e.g., about 2:1 , 3:1 , 4:1 , 5:1 , 6:1 , 7:1 , or 8:1 ).
In some embodiments, the lipid nanoparticles do not comprise any phospholipids.
In some aspects, the lipid nanoparticle can further comprise a component, such as a sterol, to provide membrane integrity. One exemplary sterol that can be used in the lipid nanoparticle is cholesterol and derivatives thereof. Non-limiting examples of cholesterol derivatives include polar analogues such as 5a-choiestanol, 53-coprostanol, choiesteryl-(2,-hydroxy)-ethyl ether, choiesteryl-(4'- hydroxy)-butyl ether, and 6-ketocholestanol; non-polar analogues such as 5a-cholestane, cholestenone, 5a-cholestanone, 5p- cholestanone, and cholesteryl decanoate; and mixtures thereof. In some embodiments, the cholesterol derivative is a polar analogue, e.g., choiesteryl-(4 '-hydroxy)-buty1 ether. Exemplary cholesterol derivatives are described in PCT publication W02009/127060 and US patent publication US2010/0130588, each of which is incorporated herein by reference in its entirety.
In some embodiments, the component providing membrane integrity, such as a sterol, can comprise 0-50% (mol) (e.g., 0-10%, 10-20%, 20-30%, 30-40%, or 40-50%) of the total lipid present in the lipid nanoparticle. In some embodiments, such a component is 20-50% (mol) 30-40% (mol) of the total lipid content of the lipid nanoparticle.
In some embodiments, the lipid nanoparticle can comprise a polyethylene glycol (PEG) or a conjugated lipid molecule. Generally, these are used to inhibit aggregation of lipid nanoparticles and/or provide steric stabilization. Exemplary conjugated lipids include, but are not limited to, PEG-lipid conjugates, polyoxazoline (POZ)-lipid conjugates, polyamide-lipid conjugates (such as ATTA-lipid conjugates), cationic-polymer lipid (CPL) conjugates, and mixtures thereof. In some embodiments, the conjugated lipid molecule is a PEG-lipid conjugate, for example, a (methoxy polyethylene glycol)- conjugated lipid.
Exemplary PEG-lipid conjugates include, but are not limited to, PEG-diacylglycerol (DAG) (such as l-(monomethoxy-polyethyleneglycol)-2,3-dimyristoylglycerol (PEG-DMG)), PEG-dialkyloxypropyl (DAA), PEG-phospholipid, PEG-ceramide (Cer), a pegylated phosphatidylethanoloamine (PEG-PE), PEG succinate diacylglycerol (PEGS-DAG) (such as 4-0-(2',3'-di(tetradecanoyloxy)propyl-l-0-(w- methoxy(polyethoxy)ethyl) butanedioate (PEG-S-DMG)), PEG dialkoxypropylcarbam, N-(carbonyl- methoxypolyethylene glycol 2000)-l,2-distearoyl-sn-glycero-3-phosphoethanolamine sodium salt, or a mixture thereof. Additional exemplary PEG-lipid conjugates are described, for example, in US5, 885,613, US6,287,59I,
US2003/0077829, US2003/0077829, US2005/0175682, US2008/0020058, US2011/0117125, US2010/0130588, US2016/0376224, US2017/0119904, and US/099823, the contents of all of which are incorporated herein by reference in their entirety. In some embodiments, a PEG-lipid is a compound of Formula III, lll-a-l, lll-a-2, lll-b-1 , lll-b-2, or V of US2018/0028664, the content of which is incorporated herein by reference in its entirety. In some embodiments, a PEG-lipid is of Formula II of US20150376115 or US2016/0376224, the content of both of which is incorporated herein by reference in its entirety. In some embodiments, the PEG-DAA conjugate can be, for example, PEG-dilauryloxypropyl, PEG- dimyristyloxypropyl, PEG-dipalmityloxypropyl, or PEG-distearyloxypropyl. The PEG-lipid can be one or more of PEG-DMG, PEG-dilaurylglycerol, PEG-dipalmitoylglycerol, PEG- disterylglycerol, PEG- dilaurylglycamide, PEG-dimyristylglycamide, PEG- dipalmitoylglycamide, PEG-disterylglycamide, PEG- cholesterol (l-[8'-(Cholest-5-en-3[beta]- oxy)carboxamido-3',6'-dioxaoctanyl] carbamoyl-[omega]-methyl- poly(ethylene glycol), PEG- DMB (3,4-Ditetradecoxylbenzyl- [omega]-methyl-poly(ethylene glycol) ether),
and 1 ,2- dimyristoyl-sn-glycero-3-phosphoethanolamine-N-[methoxy(polyethylene glycol)-2000]. In some embodiments, the PEG-lipid comprises PEG-DMG, 1 ,2- dimyristoyl-sn-glycero-3-phosphoethanolamine-N- [methoxy(polyethylene glycol)-2000]. In some embodiments, the PEG-lipid comprises a structure selected from:
In some embodiments, lipids conjugated with a molecule other than a PEG can also be used in place of PEG-lipid. For example, polyoxazoline (POZ)-lipid conjugates, polyamide-lipid conjugates (such as ATTA-lipid conjugates), and cationic-polymer lipid (GPL) conjugates can be used in place of or in addition to the PEG-lipid.
Exemplary conjugated lipids, i.e. , PEG-lipids, (POZ)-lipid conjugates, ATTA-lipid conjugates and cationic polymer-lipids are described in the PCT and LIS patent applications listed in Table 2 of WO2019051289A9, the contents of all of which are incorporated herein by reference in their entirety.
In some embodiments, the PEG or the conjugated lipid can comprise 0-20% (mol) of the total lipid present in the lipid nanoparticle. In some embodiments, PEG or the conjugated lipid content is 0.5- 10% or 2-5% (mol) of the total lipid present in the lipid nanoparticle. Molar ratios of the ionizable lipid, non-cationic- lipid, sterol, and PEG/conjugated lipid can be varied as needed. For example, the lipid particle can comprise 30-70% ionizable lipid by mole or by total weight of the composition, 0-60% cholesterol by mole or by total weight of the composition, 0-30% non-cationic-lipid by mole or by total weight of the composition and 1 -10% conjugated lipid by mole or by total weight of the composition. Preferably, the composition comprises 30-40% ionizable lipid by mole or by total weight of the composition, 40-50% cholesterol by mole or by total weight of the composition, and 10- 20% non-cationic-lipid by mole or by total weight of the composition. In some other embodiments, the composition is 50-75% ionizable lipid by mole or by total
weight of the composition, 20-40% cholesterol by mole or by total weight of the composition, and 5 to 10% non-cationic-lipid, by mole or by total weight of the composition and 1 -10% conjugated lipid by mole or by total weight of the composition. The composition may contain 60-70% ionizable lipid by mole or by total weight of the composition, 25-35% cholesterol by mole or by total weight of the composition, and 5-10% non-cationic-lipid by mole or by total weight of the composition. The composition may also contain up to 90% ionizable lipid by mole or by total weight of the composition and 2 to 15% non-cationic lipid by mole or by total weight of the composition. The formulation may also be a lipid nanoparticle formulation, for example comprising 8-30% ionizable lipid by mole or by total weight of the composition, 5-30% noncationic lipid by mole or by total weight of the composition, and 0-20% cholesterol by mole or by total weight of the composition; 4-25% ionizable lipid by mole or by total weight of the composition, 4-25% noncationic lipid by mole or by total weight of the composition, 2 to 25% cholesterol by mole or by total weight of the composition, 10 to 35% conjugate lipid by mole or by total weight of the composition, and 5% cholesterol by mole or by total weight of the composition; or 2-30% ionizable lipid by mole or by total weight of the composition, 2-30% non-cationic lipid by mole or by total weight of the composition, 1 to 15% cholesterol by mole or by total weight of the composition, 2 to 35% conjugate lipid by mole or by total weight of the composition, and 1 -20% cholesterol by mole or by total weight of the composition; or even up to 90% ionizable lipid by mole or by total weight of the composition and 2-10% non-cationic lipids by mole or by total weight of the composition, or even 100% cationic lipid by mole or by total weight of the composition. In some embodiments, the lipid particle formulation comprises ionizable lipid, phospholipid, cholesterol and a PEG-ylated lipid in a molar ratio of 50: 10:38.5: 1 .5. In some other embodiments, the lipid particle formulation comprises ionizable lipid, cholesterol and a PEG-ylated lipid in a molar ratio of 60:38.5:1.5.
In some embodiments, the lipid particle comprises ionizable lipid, non-cationic lipid (e.g. phospholipid), a sterol (e.g., cholesterol) and a PEG-ylated lipid, where the molar ratio of lipids ranges from 20 to 70 mole percent for the ionizable lipid, with a target of 40-60, the mole percent of non-cationic lipid ranges from 0 to 30, with a target of 0 to 15, the mole percent of sterol ranges from 20 to 70, with a target of 30 to 50, and the mole percent of PEG-ylated lipid ranges from 1 to 6, with a target of 2 to 5.
In some embodiments, the lipid particle comprises ionizable lipid I non-cationic- lipid I sterol I conjugated lipid at a molar ratio of 50:10:38.5:1 .5.
In an aspect, the disclosure provides a lipid nanoparticle formulation comprising phospholipids, lecithin, phosphatidylcholine and phosphatidylethanolamine.
In some embodiments, one or more additional compounds can also be included. Those compounds can be administered separately, or the additional compounds can be included in the lipid nanoparticles of the invention. In other words, the lipid nanoparticles can contain other compounds in addition to the nucleic acid or at least a second nucleic acid, different than the first. Without limitations, other additional compounds can be selected from the group consisting of small or large organic or inorganic molecules, monosaccharides, disaccharides, trisaccharides, oligosaccharides, polysaccharides, peptides, proteins, peptide analogs and derivatives thereof, peptidomimetics, nucleic acids, nucleic acid analogs and derivatives, an extract made from biological materials, or any combinations thereof.
In some embodiments, LNPs are directed to specific tissues by the addition of LNP targeting domains. For example, biological ligands may be displayed on the surface of LNPs to enhance interaction with cells displaying cognate receptors, thus driving association with and cargo delivery to tissues wherein cells express the receptor. In some embodiments, the biological ligand may be a ligand that drives delivery to the liver, e.g., LNPs that display GalNAc result in delivery of nucleic acid cargo to hepatocytes that display asialoglycoprotein receptor (ASGPR). The work of Akinc et al. Mol Ther 18(7):1357-1364 (2010) teaches the conjugation of a trivalent GalNAc ligand to a PEG-lipid (GalNAc-PEG-DSG) to yield LNPs dependent on ASGPR for observable LNP cargo effect (see, e.g., FIG. 6 of Akinc et al. 2010, supra). Other ligand-displaying LNP formulations, e.g., incorporating folate, transferrin, or antibodies, are discussed in WO2017223135, which is incorporated herein by reference in its entirety, in addition to the references used therein, namely Kolhatkar et al., Curr Drug Discov Technol. 2011 8:197-206; Musacchio and Torchilin, Front Biosci. 2011 16:1388-1412; Yu et al., Mol Membr Biol. 2010 27:286-298; Patil et al., Crit Rev Ther Drug Carrier Syst. 2008 25:1 -61 ; Benoit et al., Biomacromolecules. 2011 12:2708-2714; Zhao et al., Expert Opin Drug Deliv. 2008 5:309-319; Akinc et al., Mol Ther. 2010 18:1357-1364; Srinivasan et al., Methods Mol Biol. 2012 820:105-116; Ben-Arie et al., Methods Mol Biol. 2012 757:497-507; Peer 2010 J Control Release. 20:63-68; Peer et al., Proc Natl Acad Sci U S A. 2007 104:4095-4100; Kim et al., Methods Mol Biol. 2011 721 :339-353; Subramanya et al., Mol Ther. 2010 18:2028-2037; Song et al., Nat Biotechnol. 200523:709-717; Peer et al., Science. 2008319:627-630; and Peer and Lieberman, Gene Ther. 2011 18:1127-1133.
In some embodiments, LNPs are selected for tissue-specific activity by the addition of a Selective ORgan Targeting (SORT) molecule to a formulation comprising traditional components, such as ionizable cationic lipids, amphipathic phospholipids, cholesterol and polyethylene glycol) (PEG) lipids. The teachings of Cheng et al. Nat Nanotechnol 15(4):313-320 (2020) demonstrate that the addition of a supplemental “SORT” component precisely alters the in vivo RNA delivery profile and mediates tissuespecific (e.g., lungs, liver, spleen) gene delivery and editing as a function of the percentage and biophysical property of the SORT molecule.
In some embodiments, the LNPs comprise biodegradable, ionizable lipids. In some embodiments, the LNPs comprise (9Z,l2Z)-3-((4,4-bis(octyloxy)butanoyl)oxy)-2-((((3- (diethylamino)propoxy)carbonyl)oxy)methyl)propyl octadeca-9,l2-dienoate, also called 3- ((4,4- bis(octyloxy)butanoyl)oxy)-2-((((3-(diethylamino)propoxy)carbonyl)oxy)methyl)propyl (9Z,l2Z)-octadeca- 9,12-dienoate) or another ionizable lipid. See, e.g, lipids of WO2019/067992, WO/2017/173054, WO2015/095340, and WO2014/136086, as well as references provided therein. In some embodiments, the term cationic and ionizable in the context of LNP lipids is interchangeable, e.g., wherein ionizable lipids are cationic depending on the pH.
In some embodiments, the average LNP diameter of the LNP formulation may be between 10s of nm and 100s of nm, e.g., measured by dynamic light scattering (DLS). In some embodiments, the average LNP diameter of the LNP formulation may be from about 40 nm to about 150 nm, such as about 40 nm, 45 nm, 50 nm, 55 nm, 60 nm, 65 nm, 70 nm, 75 nm, 80 nm, 85 nm, 90 nm, 95 nm, 100 nm, 105 nm, 110 nm, 115 nm, 120 nm, 125 nm, 130 nm, 135 nm, 140 nm, 145 nm, or 150 nm. In some embodiments, the average LNP diameter of the LNP formulation may be from about 50 nm to about 100 nm, from about 50
nm to about 90 nm, from about 50 nm to about 80 nm, from about 50 nm to about 70 nm, from about 50 nm to about 60 nm, from about 60 nm to about 100 nm, from about 60 nm to about 90 nm, from about 60 nm to about 80 nm, from about 60 nm to about 70 nm, from about 70 nm to about 100 nm, from about 70 nm to about 90 nm, from about 70 nm to about 80 nm, from about 80 nm to about 100 nm, from about 80 nm to about 90 nm, or from about 90 nm to about 100 nm. In some embodiments, the average LNP diameter of the LNP formulation may be from about 70 nm to about 100 nm. In a particular embodiment, the average LNP diameter of the LNP formulation may be about 80 nm. In some embodiments, the average LNP diameter of the LNP formulation may be about 100 nm. In some embodiments, the average LNP diameter of the LNP formulation ranges from about I mm to about 500 mm, from about 5 mm to about 200 mm, from about 10 mm to about 100 mm, from about 20 mm to about 80 mm, from about 25 mm to about 60 mm, from about 30 mm to about 55 mm, from about 35 mm to about 50 mm, or from about 38 mm to about 42 mm.
A LNP may, in some instances, be relatively homogenous. A polydispersity index may be used to indicate the homogeneity of a LNP, e.g., the particle size distribution of the lipid nanoparticles. A small (e.g., less than 0.3) polydispersity index generally indicates a narrow particle size distribution. A LNP may have a polydispersity index from about 0 to about 0.25, such as 0.01 , 0.02, 0.03, 0.04, 0.05, 0.06, 0.07, 0.08, 0.09, 0.10, 0.11 , 0.12, 0.13, 0.14, 0.15, 0.16, 0.17, 0.18, 0.19, 0.20, 0.21 , 0.22, 0.23, 0.24, or 0.25. In some embodiments, the polydispersity index of a LNP may be from about 0.10 to about 0.20.
The zeta potential of a LNP may be used to indicate the electrokinetic potential of the composition. In some embodiments, the zeta potential may describe the surface charge of an LNP. Lipid nanoparticles with relatively low charges, positive or negative, are generally desirable, as more highly charged species may interact undesirably with cells, tissues, and other elements in the body. In some embodiments, the zeta potential of a LNP may be from about -10 mV to about +20 mV, from about -10 mV to about +15 mV, from about -10 mV to about +10 mV, from about -10 mV to about +5 mV, from about -10 mV to about 0 mV, from about -10 mV to about -5 mV, from about -5 mV to about +20 mV, from about -5 mV to about +15 mV, from about -5 mV to about +10 mV, from about -5 mV to about +5 mV, from about -5 mV to about 0 mV, from about 0 mV to about +20 mV, from about 0 mV to about +15 mV, from about 0 mV to about +10 mV, from about 0 mV to about +5 mV, from about +5 mV to about +20 mV, from about +5 mV to about +15 mV, or from about +5 mV to about +10 mV.
The efficiency of encapsulation of a protein and/or nucleic acid, describes the amount of protein and/or nucleic acid that is encapsulated or otherwise associated with a LNP after preparation, relative to the initial amount provided. The encapsulation efficiency is desirably high (e.g., close to 100%). The encapsulation efficiency may be measured, for example, by comparing the amount of protein or nucleic acid in a solution containing the lipid nanoparticle before and after breaking up the lipid nanoparticle with one or more organic solvents or detergents. An anion exchange resin may be used to measure the amount of free protein or nucleic acid (e.g., RNA) in a solution. Fluorescence may be used to measure the amount of free protein and/or nucleic acid (e.g., RNA) in a solution. For the lipid nanoparticles described herein, the encapsulation efficiency of a protein and/or nucleic acid may be at least 50%, for example 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or 100%. In some
embodiments, the encapsulation efficiency may be at least 80%. In some embodiments, the encapsulation efficiency may be at least 90%. In some embodiments, the encapsulation efficiency may be at least 95%.
A LNP may optionally comprise one or more coatings. In some embodiments, a LNP may be formulated in a capsule, film, or table having a coating. A capsule, film, or tablet including a composition described herein may have any useful size, tensile strength, hardness or density.
Additional exemplary lipids, formulations, methods, and characterization of LNPs are taught by W02020061457, which is incorporated herein by reference in its entirety.
In some embodiments, in vitro or ex vivo cell lipofections are performed using Lipofectamine MessengerMax (Thermo Fisher) or TransIT-mRNA Transfection Reagent (Mirus Bio). In certain embodiments, LNPs are formulated using the GenVoyJLM ionizable lipid mix (Precision NanoSystems). In certain embodiments, LNPs are formulated using 2,2-dilinoleyl-4-dimethylaminoethyl-[1 ,3]-dioxolane (DLin- KC2-DMA) or dilinoleylmethyl-4-dimethylaminobutyrate (DLin-MC3-DMA or MC3), the formulation and in vivo use of which are taught in Jayaraman et al. Angew Chem Int Ed Engl 51 (34):8529-8533 (2012), incorporated herein by reference in its entirety.
LNP formulations optimized for the delivery of CRISPR-Cas systems, e.g., Cas9-gRNA RNP, gRNA, Cas9 mRNA, are described in WO2019067992 and WO2019067910, both incorporated by reference.
Additional specific LNP formulations useful for delivery of nucleic acids are described in US8158601 and US8168775, both incorporated by reference, which include formulations used in patisiran, sold under the name ONPATTRO.
Exemplary dosing of LNPs comprising the RNA compositions described herein may include about 0.1 , 0.25, 0.3, 0.5, 1 , 2, 3, 4, 5, 6, 8, 10, or 100 mg/kg (RNA). Exemplary dosing of AAV comprising a nucleic acid encoding one or more components of the system may include an MOI of about 1011 , 1012, 1013, and 1014 vg/kg.
In some embodiments, the invention includes a lipid nanoparticle (LNP) comprising the ANDbody polypeptide (or RNA encoding the same), nucleic acid molecule, or DNA encoding an ANDbody described herein. In embodiments, the LNP comprises a cationic lipid. In some embodiments, the LNP further comprises one or more neutral lipid, e.g., DSPC, DPPC, DMPC, DOPC, POPC, DOPE, SM, a steroid, e.g., cholesterol, and/or one or more polymer conjugated lipid, e.g., a pegylated lipid, e.g., PEG-DAG, PEG-PE, PEG-S-DAG, PEG-cer or a PEG dialkyoxypropylcarbamate. In some embodiments, the cationic lipid of the LNP has a structure according to:
For a review of LNP, see also, e.g., Li et al. 2017, Nanomaterials 7, 122; doi:10.3390/nano7060122.
Other Carriers
Viral vectors
The compositions described herein (e.g., polypeptide (e.g., IL-22 mutein polypeptide) or RNA ANDbody compositions), can be delivered by a viral vector (e.g., a viral vector expressing an RNA). A viral vector may be administered to a cell or to a subject (e.g., a human subject or non-human animal). A viral vector may be locally or systemically administered.
Examples of viral vectors include a retrovirus (e.g., Retroviridae family viral vector), adenovirus (e.g., Ad5, Ad26, Ad34, Ad35, and Ad48), parvovirus (e.g., adeno-associated viruses), coronavirus, negative strand RNA viruses such as orthomyxovirus (e.g., influenza virus), rhabdovirus (e.g., rabies and vesicular stomatitis virus), paramyxovirus (e.g., measles and Sendai), positive strand RNA viruses, such as picornavirus and alphavirus, and double stranded DNA viruses including adenovirus, herpesvirus (e.g., Herpes Simplex virus types 1 and 2, Epstein-Barr virus, cytomegalovirus, replication deficient herpes virus), and poxvirus (e.g., vaccinia, modified vaccinia Ankara (MVA), fowlpox and canarypox). Other viruses include Norwalk virus, togavirus, flavivirus, reoviruses, papovavirus, hepadnavirus, human papilloma virus, human foamy virus, and hepatitis virus, for example. Examples of retroviruses include: avian leukosis-sarcoma, avian C-type viruses, mammalian C-type, B-type viruses, D-type viruses, oncoretroviruses, HTLV-BLV group, lentivirus, alpharetrovirus, gammaretrovirus, spumavirus (Coffin, J. M., Retroviridae: The viruses and their replication, Virology (Third Edition) Lippincott-Raven, Philadelphia, 1996). Other examples include murine leukemia viruses, murine sarcoma viruses, mouse mammary tumor virus, bovine leukemia virus, feline leukemia virus, feline sarcoma virus, avian leukemia virus, human T-cell leukemia virus, baboon endogenous virus, Gibbon ape leukemia virus, Mason Pfizer monkey virus, simian immunodeficiency virus, simian sarcoma virus, Rous sarcoma virus and lentiviruses. Other examples of vectors are described, for example, in US Patent No. 5,801 ,030, the teachings of which are incorporated herein by reference.
Anellovirus vectors can also be used for delivering an ANDbody composition described herein. Anellovectors are known in the art and described, e.g., in W02020123773, WO2020123816, WO2018232017, and W02020123773. In certain embodiments, an anellovector composition comprises a genomic element that comprises a promoter operably linked to a nucleic acid sequence encoding an ANDbody described herein, the genetic element encapsulated by a proteinaceous exterior comprising an Anellovirus ORF1 , e.g., an anellovirus capsid protein.
Cell and vesicle-based carriers
A composition described herein (e.g., polypeptide (e.g., IL-22 mutein polypeptide) or RNA ANDbody compositions), described herein can be administered to a cell in a cell, vesicle or other membrane-based carrier. In one embodiment, the compositions and systems described herein can be formulated in liposomes or other similar vesicles. Liposomes are spherical vesicle structures composed of a uni- or multilamellar lipid bilayer surrounding internal aqueous compartments and a relatively impermeable outer lipophilic phospholipid bilayer. Liposomes may be anionic, neutral or cationic. Liposomes are biocompatible, nontoxic, can deliver both hydrophilic and lipophilic drug molecules, protect their cargo from degradation by plasma enzymes, and transport their load across biological membranes and the blood brain barrier (BBB) (see, e.g., Spuch and Navarro, Journal of Drug Delivery, vol. 2011 , Article ID 469679, 12 pages, 2011 . doi:10.1155/2011/469679 for review). Vesicles can be made from several different types of lipids; however, phospholipids are most commonly used to generate liposomes as drug carriers. Methods for preparation of multilamellar vesicle lipids are known in the art (see for example U.S. Pat. No. 6,693,086, the teachings of which relating to multilamellar vesicle lipid preparation are incorporated herein by reference). Although vesicle formation can be spontaneous when a lipid film is mixed with an aqueous solution, it can also be expedited by applying force in the form of shaking by using a homogenizer, sonicator, or an extrusion apparatus (see, e.g., Spuch and Navarro, Journal of Drug Delivery, vol. 2011 , Article ID 469679, 12 pages, 2011 . doi :10.1155/2011/469679 for review). Extruded lipids can be prepared by extruding through filters of decreasing size, as described in Templeton et al., Nature Biotech, 15:647-652, 1997, the teachings of which relating to extruded lipid preparation are incorporated herein by reference.
Exosomes can also be used as drug delivery vehicles for the compositions and systems described herein. For a review, see Ha et al. July 2016. Acta Pharmaceutica Sinica B. Volume 6, Issue 4, Pages 287- 296; https://doi.Org/10.1016/j.apsb.2O16.02.001 .
Ex vivo differentiated red blood cells can also be used as a carrier for an agent (e.g., an inhibitor) described herein, e.g., an antibody or a nucleic acid described herein. See, e.g., WO2015073587; WO2017123646; WO2017123644; WO2018102740; wO2016183482; WO2015153102; WO2018151829; WO2018009838; Shi et al. 2014. Proc Natl Acad Sci USA. 111 (28): 10131-10136; US Patent 9,644,180; Huang et al. 2017. Nature Communications 8: 423; Shi et al. 2014. Proc Natl Acad Sci USA. 111 (28): 10131-10136.
Fusosome compositions, e.g., as described in WO2018208728, can also be used as carriers to deliver the agent or preparation described herein.
Plant nanovesicles and plant messenger packs (PMPs), e.g., as described in WO2011097480, WO2013070324, WO2017004526, or W02020041784 can also be used as carriers to deliver the compositions described herein.
Without further elaboration, it is believed that one skilled in the art can, based on the above description, utilize the present invention to its fullest extent. The following specific embodiments are, therefore, to be construed as merely illustrative, and not limitative of the remainder of the disclosure in any way whatsoever. All publications and sections thereof cited herein are herein incorporated by reference for the purposes or subject matter referenced herein.
EXAMPLES
The invention will be further illustrated in the following non-limiting examples.
Example 1. Computational prediction of protein “addresses” with specific expression in IL-22- responsive tissues
To identify potential address targets that could localize IL-22 activity to specific cells, RNA- sequencing data from various public and proprietary data sources were summarized to determine expression for each gene across each of 32 tissues using the mean expression as measured by Transcripts Per Million (TPM). Specificity scores were calculated using four different statistical methods that capture the specificity of expression for each gene in each tissue or tissue combination (e.g., grouped tissues from the lower gastrointestinal (Gl) tract). The average rank of a gene was calculated as the mean rank across the four different methods. The genes were ranked according to specificity score for each tissue.
For each tissue, a list of the top (up to) 25 addresses that (i) met a set threshold for minimum specificity score and minimum expression level in the tissue and (ii) were annotated as containing a transmembrane domain or being otherwise membrane-associated was generated (“top addresses;” Table 1 ). Table 1 (above) shows proteins with a specificity score greater than 0.7 in lower Gl, kidney, liver, pancreas, and skin.
Example 2. Early feasibility assessment using quantitative systems pharmacology modeling of IL- 22/CDH17 ANDbody predicts an improved therapeutic profile
A modeling assessment of the therapeutic profile of localized IL-22 in inflammatory bowel diseases (IBD) was performed. The model considered a biotherapeutic that binds to two target cell surface receptors on a single cell type acting as a receptor agonist for IL-22. The molecule can be monovalent or bivalent with avidity for each target. The percent of receptor occupied by the cytokine was used as a metric for signaling activity. The model parameterizes various drug properties (binding affinity, half-life, dosing mode and frequency, valency, avidity, etc.) and biophysical characteristics (cytokine concentration in various compartments, receptor numbers, receptor turnover rate, etc.) and predicts how drug agonism depends on these parameters. Parameter scans were performed around nominal values to determine the feasibility window with high receptor occupancy in the disease compartment and low
occupancy in the toxicity compartment. Receptor occupancy for a range of binding affinities for IL-22 and CDH17 in the disease and toxicity compartments is shown in Figs. 8A and 8B. These simulations show that an increased therapeutic index can be achieved at an appropriately low dissociation constant (KD) for the localizer (<100 nM in these simulations) and an intermediate KD for IL-22 (0.02 - 200nM). These results show qualitatively that it is feasible to design targeted IL-22 molecules with increased therapeutic index.
Example 3. PK/PD modeling of IL-22/CDH17 ANDbody to demonstrate improved therapeutic profile
Pharmacokinetic/pharmacodynamic (PK/PD) modeling is performed to evaluate the therapeutic profile of localized IL-22 in IBD to identify and explain the relationships between drug exposure, safety, and efficacy and the differences among population subgroups. A nonlinear mixed effect model is used to characterize the relationships between dose, exposure, and biomarker and/or clinical endpoint response over time, variability between subjects (e.g., mice) and groups, residual variability and uncertainty, and predict minimum anticipated biological effect level (MABEL) dose for the selected biotherapeutic.
Example 4. Design, expression, and binding activity of ANDbodies comprising IL-22/antibody fusion proteins
A. IL-22/antibody fusion protein formats
Two formats that allow IL-22 to be fused to functional antibodies were developed. All of the Fc domains used include LALAPG substitutions to abrogate Fc effector functions (Schlothauer et al., Protein Eng. Des. Sei. PEDS, 29: 457-466, 2016). The first format, referred to herein as “monovalent IL- 22/mAb,” comprises a heterodimeric molecule comprising two arms. The first arm comprises a standard antibody heavy chain and light chain, where the Fc region of the antibody incorporates substitutions for heterodimeric pairing (e.g., as in SEQ ID NO: 4). The second arm comprises an Fc region with an IL-22 polypeptide fused in place of the antibody Fab region (e.g., as in SEQ ID NO: 9). The IL-22 polypeptide and Fc region are linked by a (G4S)2 linker (SEQ ID NO: 5). A schematic diagram of the monovalent IL- 22/mAb format is shown in Fig. 1 A.
The second format, referred to herein as “divalent IL-22/mAb,” comprises a standard antibody molecule with an IL-22 polypeptide fused to the C-terminus of both Fc domains. These polypeptides are connected by a (G4S)4 linker (SEQ ID NO: 6). One representative divalent IL-22/mAb sequence pairs IL- 22 with an antibody that binds to glycoprotein F from respiratory syncytial virus (SEQ ID NO: 10). Another representative divalent IL-22/mAb sequence pairs IL-22 with an antibody that binds to CDH17 (e.g., human CDH17). A schematic diagram of the divalent IL-22/mAb format is shown in Fig. 1 B.
Any antibody variable domains of the light chain and heavy chain combination described herein can be used in the antibody (e.g., a heavy chain as presented in SEQ ID NO: 1 and/or a light chain as presented in SEQ ID NO: 2).
This example provides antibodies in which the CHi and Fc region are human IgG 1 (SEQ ID NO: 3). The exemplary monovalent IL-22/mAb provided herein includes additional substitutions in the Fc
region that favor heterodimeric pairing (Moore et al., Methods, 154: 38-50, 2019) and selective purification of the desired heterodimeric molecule over either homodimeric product (see, e.g., US Patent No. 8,586,713 B2), such as in SEQ ID NOs: 4 and 9. Alternative Fc sequences may also be utilized. Table 2. Protein Sequences
B. Methods of production
To produce antibodies and divalent IL-22 fusion proteins (e.g., divalent IL-22/mAbs), EXPI293F™ cells were transiently transfected with a 2:3 mass ratio of heavy chain: light chain plasmid using polyethylenimine (PEI) and maintained according to manufacturer’s instructions (37°C, 8% CO2 on a shaking platform). Cultures were fed 4-24 hours post-transfection to a final concentration of 5% v/v GIBCO™ EFFICIENTFEED™ B, 1% v/v L-alanyl-glutamine, and 4 mM valproic acid. After 4-7 days, supernatants were harvested by centrifugation at 4000 x g followed by 0.2 pm filtration. Filtered supernatants were purified via Protein A affinity chromatography (Cytiva 5 mL MABSELECT™ SuRe column) in PBS 7.4 running buffer and 0.1 M glycine pH 2.7 elution buffer. Eluted protein was immediately neutralized with 10% v/v of 1 M sodium acetate, pH 6.0. Protein was buffer exchanged into 50 mM sodium acetate + 60 mM sucrose, pH 6.0 for storage.
Monovalent IL-22 fusion proteins (e.g., monovalent IL-22/mAbs) were produced by transient PEI transfection of a 1 :1 :2 mass ratio of heavy chain : light chain : IL-22-Fc plasmid. The expression host, culture conditions, and purification protocol were identical to those described above for the divalent ANDbody. In these constructs, the IL-22-containing molecule comprised mutations in the Fc domain such that incorrectly homodimerized IL-22 molecules do not bind efficiently to Protein A. An alternative approach that can also be used is Fab arm exchange (Steinhardt et al, Pharmaceutics, 12(3), 2019). This uses different substitutions in the Fc region, but produces functionally identical proteins.
Binding of each molecule to its antibody target and to human IL-22RA1 was evaluated using biolayer interferometry (BLI) on a GATOR® instrument at ambient temperature (~22°C). In these assays, the fusion protein was loaded on GATOR® biosensor probes coated with anti-human Fc antibody, and binding was evaluated by dipping into a solution containing the indicated target. Mixing speed was 1000 rpm. The antigens used in these Examples include ectodomains of hlL22RA1 (R&D Systems #2770-LR-050), mlL22RA1 (R&D Systems #4294-MR-050), mCDH17 (NCBI NP_062727, purified in-house), hlL22BP (Sino Biological US, 11025-H08H) and RSV glycoprotein F (Sino Biological US, 11049-V08B).
Table 3 shows the binding affinity of each molecule for its two targets (murine targets are denoted with “m” before the gene name; human targets are denoted with “h”), as measured using BLI. These data show that both fusion protein formats achieved binding to both the antibody target and the IL- 22 receptor.
Table 3. BLI measurement of binding of IL-22/antibody fusion proteins to IL22RA1 and their antibody targets
Example 5. Signaling activity of both monovalent and divalent IL-22/antibody fusion proteins targeting mCDH17 is enhanced on cells that express mCDH17
The activity of each IL-22 fusion protein provided in Example 4 was evaluated using a commercial reporter cell line (HEK-BLUE™ IL-22 cells (InvivoGen)). These reporter cells produce secreted embryonic alkaline phosphatase (SEAP) in response to activation of IL-22 signaling. The reporter cells were modified to stably express murine CDH17 (NCBI NP_062727) (i.e., were transduced using lentiviral vectors followed by cell sorting to generate stable pools to produce cell strains with stable expression of murine CDH17).
Table 4 shows the response to each fusion protein of parental reporter cells (“reporter cell”) and modified reporter cells expressing mCDH17 (“+mCDH17”). An antibody against respiratory syncytial virus (RSV) glycoprotein F was included as a negative control. These data show that both monovalent and divalent formats of IL-22 targeted to mCDH17 have increased potency on reporter cells expressing mCDH17.
Table 4. Signaling response of IL-22/antibody fusion proteins targeting CDH17 on reporter cells with and without mCDH17 expression
Example 6. Monovalent and divalent IL-22/antibody fusion proteins targeting mCDH16 or mGP2 and signaling activity thereof
A. IL-22/mCDH16 antibody fusion proteins
Monovalent and divalent format IL-22 fusion proteins (monovalent IL-22/mAb and divalent IL- 22/mAb) as described in Example 4 are prepared using an antibody against the address target mCDH16 (NCBI NP_031689), which is expressed in kidney tubules.
HEK-BLUE™ IL-22 reporter cells expressing mCDH16 are prepared as described in Example 5. An assay as described in Example 5 is performed to evaluate address-dependent enhancement of signaling potency for the targeted IL-22 molecules (monovalent anti-mCDH16/IL-22 and divalent anti- mCDH16/IL-22) in reporter cells expressing the mCDH16 address target as compared to matched nontargeted molecules, differently targeted molecules, and reporter cells not comprising the address.
Both formats are expected to show significant enhanced potency when address expression matches the antibody target, indicating that address-dependent potentiation is a general feature of the antibody/IL-22 fusion proteins.
B. IL-22/mGP2 antibody fusion proteins
Monovalent and divalent format IL-22 fusion proteins (monovalent IL-22/mAb and divalent IL- 22/mAb) as described in Example 4 are prepared using an antibody against the address target mGP2 (NCBI NP_080265), which is expressed in pancreatic acinar cells.
HEK-BLUE™ IL-22 reporter cells expressing mGP2 are prepared as described in Example 5. An assay as described in Example 5 is performed to evaluate address-dependent enhancement of signaling potency for the targeted IL-22 molecules (monovalent anti-mGP2/IL-22 and divalent anti-mGP2/IL-22) in reporter cells expressing the mGP2 address target as compared to matched non-targeted molecules, differently targeted molecules, and reporter cells not comprising the address.
C. Fusion Protein Potentiation
The monovalent and the divalent IL-22 fusion protein formats are designed to elicit significantly enhanced potency in reporter cells that express an address that is targeted by the antibody. For example, increased potency is expected for the IL-22/mCDH16 antibody fusion proteins in reporter cells expressing mCDH16, and for the IL-22/mGP2 antibody fusion proteins in reporter cells expressing mGP2, thus showing that address-dependent potentiation is a general feature of the antibody/IL-22 fusion proteins.
Example 7. Rationally engineered substitutions at the IL-22/IL-22RA1 interface reduce receptor binding and signaling activity of IL-22/antibody fusion proteins
It was hypothesized that reducing the binding affinity of IL-22 for its receptor IL22RA1 (referred to herein as “detuning”) would increase the potentiation ratio of address-binding IL-22 proteins in cells expressing the address as compared to non-expressing cells.
A structural modeling approach was used to predict residues and specific amino acid substitutions in IL-22 (SEQ ID NO: 8) that may reduce receptor binding affinity while maintaining structural stability of the molecule. The numbering of amino acid substitutions used herein is based on the sequence of the IL-22 precursor, including its signal peptide (SEQ ID NO: 7).
First, the IL22 interface residues (Table 5) were identified as the residues located at most 5 angstrom away from a residue on the receptor (interface distance less than 5 A).
Next, the effect of various amino acid substitutions on binding affinity and stability were predicted using mCSM-PPI2 (Rodrigues et al., Nucleic Acids Research, 47(W1 ): W338-W344, 2019) and DynaMut2 (Rodrigues et al., Protein Science, 30(1 ): 60-69, 2021 ). Table 6 shows predicted changes in free energy for binding affinity and stability upon the indicated amino acid substitutions at the interface residues of Table 5.
Table 6. Predicted changes in free energy for binding affinity and stability upon various amino acid substitutions in the IL22 interface residues
The crystal structure of the IL-22/1 L-22R1 complex (PDB ID: 3DLQ) was visually inspected, and a few potentially important interface residues (K61 , T70, D71 , R73, E77, D168, R175) on a-helixes and loop were selected for initial screening. A small number of substitutions with low to moderate change in predicted binding affinity and stability and not involving any of proline, tryptophan, methionine, and cysteine were generated (“IL-22 muteins”) and screened in a non-purified form for binding affinity to the IL-22RA1 extracellular domain by GATOR® BLI. T70A and E77Q did not express. Other results are summarized in Tables 7A and 7B, below. “Low” changes in binding affinity are less than 10-fold, “moderate” changes are 10-100-fold, and “high” changes are >1 OO-fold.
Table 7A. Observed decrease in binding affinity for IL-22RA1 extracellular domain: IL-22 Muteins were expressed as N-terminal fusions to human lgG1 Fc (hlL22-Fc)
Table 7B. Observed decrease in binding affinity for IL-22RA1 extracellular domain: IL-22 Muteins were formatted as ANDbody proteins
Table 8 lists key IL22 interface residues that were identified and specific substitutions that were predicted to be useful for this purpose based on the previously described computational and initial screening results.
Table 8. Key IL22 interface residues predicted to be useful for detuning
A subset of these substitutions were expressed as IL-22/Fc fusion proteins, in which each molecule contained two IL-22 moieties located at the N-terminal end of a human IgG 1 Fc. These were purified by protein A affinity, and the binding of each molecule to human and mouse IL22RA1 proteins was evaluated by biolayer interferometry. Results are shown in Table 9A.
Based on these results, D71 L and a subset of additional substitutions of Table 8A were introduced into the IL-22 moiety at the C-terminus of the divalent IL-22/mAb provided in Example 2 (D71 L (SEQ ID NO: 11 ), R73Q (SEQ ID NO: 12), and D71 L/D168G (SEQ ID NO: 13)), wherein the mAb was an anti-mCDH17 antibody, and evaluated binding to human and murine IL22RA1 by BLI as in Example 4. Table 9B shows the measured affinity of the divalent IL-22/mAb as for human and murine IL22RA1 as measured by BLI.
Table 9B. Effect of IL-22 substitutions on affinity for IL22RA1 in the divalent format measured by biolayer interferometry
Table 9C shows the measured affinity of the divalent IL-22/mAb as for human IL22BP as measured by BLI.
Table 9C. Effect of IL-22 substitutions on affinity for IL22BP in the divalent format measured by biolayer interferometry
Example 8. Detuning substitutions that weaken binding affinity of IL-22 for IL-22RA1 increase the potentiation effect of targeting antibodies in in vitro signaling assays
The effect of detuning IL-22 as described in Example 7 was evaluated by comparing activation of IL-22 signaling by the divalent IL-22/mAb comprising the detuning substitutions in wild-type IL-22 reporter cells and IL-22 reporter cells expressing mCDH17, using assays as in Example 5. Table 10 shows the ECso for each molecule tested. In each case, weakening affinity for IL22RA1 increased the EC50 of the molecule compared to wild-type control in the absence of mCDH17. Thus, as hypothesized, molecules with weakened affinity demonstrated reduced signaling potency (e.g. higher EC50) on reporter cells that do not express mCDH17.
Table 10. Potency assay using IL-22 reporter cells with and without the mCDH17 address target
Remarkably, these data show that, in the divalent IL-22/mAb format, weakening the affinity of IL- 22 binding to IL22RA1 from < 1 nM to above 100 nM has no meaningful impact on signaling potency on cells expressing mCDH17. In this system, the relative potency in cells with and without the address target can achieve a ratio of at least 1 ,100. Across both formats and all substitutions, these data show that weakening affinity of IL-22 for IL22RA1 can increase the potentiation ratio.
Example 9. Gut-targeting monovalent IL-22/antibody fusion proteins accumulate preferentially in the small intestine and colon of mice compared to non-targeting molecules
Address-targeted IL-22 molecules are expected to bind to their target cells when injected into mice, and such binding may manifest as increased accumulation of the molecule in a target tissue compared to non-targeted control. It may also manifest as more rapid depletion from circulation in blood.
A monovalent IL-22/mAb containing an anti-mCDH17 targeting arm (monovalent anti- mCDH17/IL-22) and a matched molecule containing an anti-RSV glycoprotein F antibody (monovalent anti-RSV/IL-22) were introduced into BALB/c mice by tail vein injection at 3 mg/kg. Three groups of three mice were used for each molecule. Blood samples were collected at 1 , 3, 6, 24, 48, and 96 hours. One group of mice for each molecule was euthanized at 24, 48 and 96 hours and tissues were collected to measure distribution of the molecule (small intestine, colon, liver, lung, skin). The concentration of each molecule in blood samples was measured by sandwich ELISA using anti-human Fc antibody to capture and anti-human light chain antibody for detection. Tissue samples were homogenized, total protein measured, and similar ELISA used to measure the amount of each fusion protein in 500 mg of extracted tissue lysate.
Fig. 2 shows the level of each molecule in circulation over time. The monovalent anti- mCDH17/IL-22 molecule mostly cleared from circulation within 48 hours, while the non-targeted molecule persisted longer in circulation. Both molecules persist in circulation similarly to their respective parental monoclonal antibodies. Figs. 3A and 3B show the level of each molecule in small intestine, colon, lung, liver, and skin tissue at 24, 48 and 96 hours. There was a markedly higher level of the targeted molecule (monovalent anti-mCDH17/IL-22) than the non-targeted molecule (monovalent anti-RSV/IL-22) in small intestine, and the targeted molecule persisted in the colon despite clearing rapidly from circulation. The opposite was observed in off-target tissues, consistent with the higher level of the non-targeted molecule that persisted in circulation. These data show that the monovalent anti-mCDH17/IL-22 accumulates in the target tissues of small intestine and colon.
Example 10. Monovalent IL-22/antibody fusion protein targeting gut epithelial cells has enhanced on-target dose response in mice compared to non-targeted molecule
Mice were treated with doses of mCDH17-targeted and non-targeted monovalent IL-22 molecules (monovalent anti-mCDH17/IL-22 and monovalent anti-RSV/IL-22, respectively) as described in Example 9 at doses of 1 mg/kg, 0.3 mg/kg, and 0.1 mg/kg. Groups contained 5 mice each, and data were collected at 24 and 48 hours after dosing. On-target tissues (small intestine and colon) and off- target tissues (pancreas and liver) were processed to extract mRNA, and the expression levels of key IL-
22 target genes (REG3B, REG3G and S0CS3) were measured. Figs. 4A-4D show the levels of REG3B, REG3G and SOCS3 in each treated group relative to mice that were treated with phosphate-buffered saline.
In the on-target tissues (small intestine and colon), a markedly stronger dose response was seen in all three target genes for the monovalent anti-mCDH17/IL-22 molecule compared to treatment with the non-targeted control. Similarly, a markedly reduced dose response was seen for the monovalent anti- mCDH17/IL-22 molecule in the pancreas (off-target tissue). Limited response was observed for either molecule in the liver, suggesting that these IL-22 molecules had limited activity in the liver at the doses tested.
These data show that targeting IL-22 to mCDH17 can substantially increase its potency in two gut tissues, while also reducing potency on an off-target tissue.
Example 11. Detuning IL-22 binding to IL-22RA1 of gut-targeted monovalent IL-22/antibody fusion protein on pharmacodynamics to increase the tissue-specificity of their pharmacodynamic effect
Monovalent IL-22/mAb molecules as described in Example 5 comprising (i) a detuned IL-22 moiety such as those described in Examples 7-10 and (ii) an antibody targeting murine CDH17 are generated. The detuned IL-22 moieties are selected to span a range of affinities up to about 100x reduction in binding to IL22RA1 . Matched molecules comprising an antibody that binds to RSV glycoprotein F (monovalent anti-RSV/IL-22) are produced as a negative control for address targeting.
The molecules are introduced into mice by tail vein injection at doses ranging from 1 mg/kg to 0.1 mg/kg and on-target tissues (small intestine and colon tissues) and off-target comparator tissues (pancreas, skin, lung, and liver) are collected at timepoints over several days. The dose response of IL- 22 activity in each tissue is assayed by qPCR to compare levels of IL-22 target genes in treated versus untreated mice. These data are used to compare the dose level that generates a signaling response in on-target tissues as compared to each off-target tissue (thereby providing a specificity ratio). Molecules targeting mCDH17 and containing detuned IL-22 are expected to show a greater specificity ratio than molecules targeting mCDH17 and containing native IL-22. Non-targeted control molecules are expected to show less enhancement.
Example 12. Monovalent IL-22/antibody fusion protein targeting pancreatic acinar cells that bind to acinar cells in mice and have enhanced on-target dose response compared to a non-targeted molecule
Monovalent IL-22/mAb molecules as described in Example 4 comprising (i) a native IL-22 moiety or a detuned IL-22 moiety such as those described in Examples 7-10 and (ii) an antibody targeting murine GP2 are generated. GP2 is expressed predominantly on pancreatic acinar cells. Matched molecules comprising an antibody that binds to RSV glycoprotein F (monovalent anti-RSV/IL-22) are produced as a negative control for address targeting.
The molecules are introduced into mice by tail vein injection at 3 mg/kg, and levels of each molecule in lysate of pancreatic tissue are assayed at timepoints over several days. The GP2-targeted molecules are expected to accumulate at a higher level in pancreatic tissue when compared to the nontargeted control that binds to RSV glycoprotein F.
The molecules are then introduced into mice by tail vein injection at doses ranging from 1 mg/kg to 0.1 mg/kg and pancreatic tissue, along with off-target comparator tissues, are collected at timepoints over several days. The IL-22 activity in each tissue is assayed by using qPCR to compare levels of IL-22 target genes in treated as compared to untreated mice. The GP2-targeted molecules are expected to show an enhanced dose response in pancreas compared to the non-targeted control molecule, and a reduced dose response in the off-target tissues.
Example 13. Monovalent IL-22/antibody fusion protein that target epithelial cells of the tubular epithelium, bind tubular epithelial cells in mice, and have enhanced on-target dose response compared to non-targeted molecule
Monovalent IL-22/mAb molecules as described in Example 4 comprising (i) a native IL-22 moiety or a detuned IL-22 moiety such as those described in Examples 7-10 and (ii) an antibody targeting murine CDH16 are generated. CDH16 is expressed predominantly on epithelial cells in the kidney tubules. Matched molecules comprising an antibody that binds to RSV glycoprotein F (monovalent anti- RSV/IL-22) are produced as a negative control for address targeting.
The molecules are introduced into mice by tail vein injection at 3 mg/kg, and levels of each molecule in lysate of pancreatic tissue are assayed at timepoints over several days. The CDH16- targeted molecules are expected to accumulate at a higher level in kidney tissue when compared to nontargeted control that binds to RSV glycoprotein F.
The molecules are then introduced into mice by tail vein injection at doses ranging from 1 mg/kg to 0.1 mg/kg and kidney tissue, along with off-target comparator tissues, are collected at timepoints over several days. The IL-22 activity in each tissue is assayed by using qPCR to compare levels of IL-22 target genes in treated as compared to untreated mice. The CDH16-targeted molecules are expected to show an enhanced dose response in kidney compared to the non-targeted control molecule, and a reduced dose response in the off-target tissues.
Example 14. Detuning IL-22 for IL-22RA1 enabled gut-targeting divalent IL-22/antibody fusions to have activity specifically in the small intestine and colon of mice
The detuned divalent IL-22 molecules targeting mCDH17 provided in Examples 7 and 8 were introduced into BALB/c mice by tail vein injection at 3 mg/kg. Matched molecules targeting RSV glycoprotein F (Divalent IL-22/anti-RSV mAb) were used for comparison. Each group used nine mice. Blood samples were collected at timepoints from immediately after injection to 4 days after injection (0, 1 , 3, 6, 24, 48, and 96 hours after injection), and ELISA was used to measure the concentration of the IL-22 molecules in circulation. Three mice from each group were euthanized at 1 , 2 and 4 days, and tissues (small intestine, colon, pancreas, and liver) were collected to determine the pharmacodynamic effect of IL-22 on expression of target genes.
Fig. 5 shows the levels of mCDH17-targeted IL-22 and RSV-targeted IL-22 fusion proteins in serum over time. All of the molecules showed a rapid distribution out of circulation in the first hour, followed by slower decline. The mCDH17-targeted molecules cleared more quickly than the RSV- targeted molecules, consistent with accumulation in the gut as observed for the monovalent molecules (Example 9). The various detuning substitutions in IL-22 had little effect on the rate of clearance.
RT-PCR was used to determine expression of the IL-22 target genes REG3B, REG3G, and S0CS3 in small intestine, colon, pancreas, and liver at 1 , 2 and 4 days after treatment. Figs. 6A-6D show that the mCDH17-targeted molecule (divalent IL-22/anti-mCDH17 mAb) containing the D71 L substitution mutation in IL-22 had modest specificity for small intestine and colon at day 1 , followed by somewhat better specificity at days 2 and 4, especially with reduced activity in pancreas at later timepoints. D71 L has the strongest binding to IL-22RA1 of the three detuned IL-2 muteins. The other two muteins (R73Q and D71 L/D168G) showed clearly increased on-target activity at all three timepoints, and lower activity in pancreas than the D71 L mutein. Only the two molecules containing D71 L mutein show a clear mRNA response in the liver; the two weaker binding muteins do not show a clear response in liver at any timepoint.
These data show that detuning the receptor binding affinity of IL-22 in the divalent format (divalent IL-22/mAb) targeting mCDH17 produces molecules that retain strong activity in the target tissues (small intestine and colon), while reducing the activity in non-target tissues (pancreas and liver).
Example 15. Detuned divalent IL-22 fusion protein targeting mCDH17 has normal dose response in mice without significant activity in other tissues
Mice were treated as in described in Example 10 with doses of mCDH17-targeted and nontargeted divalent detuned IL-22 molecules comprising native IL-22 or IL-22 comprising the D71 L/D168G amino acid substitution mutations described in Example 14 at dose levels of 1 mg/kg, 0.3 mg/kg, and 0.1 mg/kg. Groups contain 5 mice each, and time-points were 24 and 48 hours after dosing. On-target tissues (small intestine and colon) and off-target tissues (pancreas and liver) were processed to extract mRNA, and the levels of selected IL-22 target genes were measured by RT-PCR. For small intestine, colon, and pancreas tissues, the target genes REG3B, REG3G and SOCS3 were measured in each treated group relative to mice treated with phosphate-buffered saline. For liver tissue, the genes MT1 and MT2 were used instead.
Figs. 7A and 7B show that in the small intestine and colon, there was a strong response to native IL-22 in both targeted and non-targeted formats. The D71 L/D168G substitutions almost eliminated activity of the non-targeted molecules on both tissues, but the targeted molecule retained nearly native activity. Figs. 7C and 7D show that the native IL-22 molecules similarly have activity on pancreas and liver, with or without targeting. Activity on the pancreas is notably reduced by targeting, possibly because it localizes the molecule rapidly to gut tissues. Crucially, activity of the IL-22 with D71 L/D168G substitutions is almost eliminated in pancreas and liver in both the targeted and non-targeted molecules. These data show that detuning IL-22 in the divalent format and targeting the molecule to mCDH17 can retain IL-22 activity on target tissues while greatly reducing its effect on non-target tissues.
Example 16. Appropriately tuned divalent IL-22 fusion proteins that target pancreatic acinar cells, bind to target cells, and have enhanced on-target dose response in mice compared to nontargeted molecules
Detuned IL-22 moieties were produced in the divalent format as fusions to an antibody targeting murine GP2 (mGP2). Matched molecules comprising an antibody that binds to RSV glycoprotein F were
produced as a negative control for address targeting. The molecules were introduced into mice by tail vein injection at a range of dose levels from 0.03 to 0.3 MPK. Groups contained 5 mice each and timepoints were 24 and 96 hours after dosing. Pancreas tissue (on-target tissue) and off-target tissues (including small intestine and colon) were processed to extract mRNA, and the level of selected IL-22 target genes were measured. Key target genes REG3B and REG3G were measured in each treated group relative to mice treated with phosphate-buffered saline.
A markedly stronger dose response was seen in the pancreas with all three target genes compared to treatment with the non-targeted control (Fig. 9A). Similarly, a markedly reduced dose response was seen in the off-target tissues when compared with the non-targeted controls (Fig. 9B, 9C). These data are show that detuning IL-22 in the divalent format and targeting to mGP2 can substantially increase its potency in target tissue, while also reducing potency on off-target tissues.
Example 17. Appropriately tuned divalent IL-22 fusion proteins that target epithelial cells of kidney tubules, bind to target cells, and have enhanced on-target dose response in mice compared to a non-targeted molecule
Detuned IL-22 moieties are produced in the divalent format as fusions to an antibody targeting murine CDH16 (mCDH16). Matched molecules comprising an antibody that binds to RSV glycoprotein F are produced as a negative control for address targeting. The molecules are introduced into mice by tail vein injection at a range of dose levels. Groups contain 5 mice each and time-points are 24 and 48 hours after dosing. On-target tissues (small intestine and colon) and off-target tissues (pancreas and liver) are processed to extract mRNA and the level of selected IL-22 target genes are measured. Key target genes REG3B, REG3G and SOCS3 are measured in each treated group relative to mice treated with phosphate-buffered saline.
Markedly stronger dose response is expected to be seen in the kidney with all three target genes compared to treatment with the non-targeted control. Similarly, a markedly reduced dose response is expected to be seen in the off-target tissues when compared with the non-targeted controls. These data are expected to show that detuning IL-22 in the divalent format and targeting to mCDH16 can substantially increase its potency in target tissue, while also reducing potency on off-target tissues.
Example 18. Detuning the affinity for anti-mCDH17 antibody to increase the circulating lifetime of gut-targeted monovalent IL-22/antibody fusion proteins in mice
The native or detuned monovalent mCDH17-targeted IL-22 is modified with substitutions in the complementarity determining regions of the anti-mCDH17 antibody that change its binding affinity from <1 nM to 1 -10 nM, 10-100 nM or >100 nM. The same modifications are made in detuned divalent IL-22 with anti-mCDH17 targeting. Anti-RSV glycoprotein F targeting is used as the negative control.
Each molecule is introduced into mice at 3 mg/kg by tail vein injection. Each molecule has a group size of six mice. Levels in blood are measured at 0, 1 , 3, 6, 24 and 48 hours. Three mice from each group are euthanized at 24 hours, and key tissues are collected (small intestine, colon, liver, lung, kidney, pancreas, skin). The remaining three mice are euthanized at 48 hours for time same tissue collection. Levels of each IL-22 molecule are measured in blood and tissue by ELISA as above.
Weakening the affinity of anti-mCDH17 is expected to increase the time in circulation for both the mono- and divalent IL-22 molecules. An intermediate affinity concentration that produces the highest local accumulation in gut tissues at 24 and 48 hours is expected. The data are expected to show that tuning anti-mCDH17 affinity to an intermediate level is necessary to provide optimal tissue-targeted distribution.
Example 19. Detuning the affinity for anti-mCDH17 antibody to increase the duration of cellular response to IL-22/antibody fusion proteins in mice
The above-described monovalent and divalent molecules with detuned IL-22 and weakened binding to mCDH17 are introduced into mice as in Example 18 at a range of dose levels. Tissues are collected at time-points up to 1 week and the IL-22 response in on-target gut tissues is compared to off- target tissues. Consistent with the extended time in circulation, on-target tissues are expected to show a sustained increase in expression of IL-22 target genes. The on-target versus off-target enhancement of dose response is expected to be sustained.
Example 20. Gut-targeted IL-22/antibody fusion proteins for treating mice following colon damage induced by dextran sulfate sodium
Damage to the colonic epithelium of C57BL/6 mice was induced by addition of 3.5% dextran sulfate sodium (DSS) to their drinking water for 4 days. This induces a colitis-like inflammatory response characterized by weight loss and inflammation of the epithelium (Kim et al., JoVE Journal, Feb 1 , 60: 3678, 2012). Mice were then allowed to recover for 8 days to observe the healing process. Mice were treated with a therapeutic dose of optimized divalent IL-22 ANDbody or untargeted murine IL22-Fc comparator molecule at -1 , 1 , 4, and 6 days relative to start of DSS addition (day 0). Mice treated with mCDH17-targeted IL-22 showed protection from weight loss and faster recovery post DSS treatment (Fig. 10A), and reduced disease activity index (Fig. 10B).
Example 21. Other cytokines related to IL-22 are expected to show similar target-dependent potentiation when formatted into a tissue-targeted AND-body
IL-22 and IL-10 are members in a family of cytokines with similar structural features and shared receptors (Ouyang et al., Immunity, 50: 871-891 , 2019). Specific cytokines in the family include IL-19, IL-20, IL-24, IL-26, IL-28A, IL-28B, and IL-29. Using the fusion molecule formats described above, a cytokine from this family which shares a receptor with IL-22 is fused to an antibody targeting a specific address expressed on one or more IL-22 target cells. A reporter cell assay similar to those described in the above examples is used to measure the activity of the cytokine/antibody fusion protein in (i) cells expressing or not expressing the address target, and (ii) cells expressing or not expressing the address target when the antibody is switched to a negative control. Potentiated signaling is expected to be observed only when the targeting antibody matches the address expressed on the reporter cells.
Example 22. IL-22 muteins have decreased binding affinity to IL22Ra independent of ANDbody formatting
A panel of nine point mutants of human IL-22 were generated as N-terminal fusions to human Fc. Constructs were expressed in Expi293 cells and purified using Protein A chromatography, as described in Example 4, and binding to human and mouse IL22Ra was measured using BLI. For these binding assays, the IL22-Fc protein variant was loaded onto an anti-human Fc biosensor probe (HFC, #160003, Gator Bio) for 120 seconds, and association was measured by dipping the probe into a solution of human IL22Ra extracellular domain for 150 seconds, followed by dissociation for 400 seconds. Data were experimentally fit to determine kOn and kotf. KD was calculated as kott/kon. All experiments were performed with 1000 rpm shaking in Q Buffer (#120010, Gator Bio). Table 1 1 displays affinity data for binding of human IL-22-Fc muteins to human IL22Ra.
ND: not determined
NB: non-binding
Example 23. Thermal stability of IL-22 muteins
Figs. 1 1 A-1 1 C show the thermal stability of a set of detuned IL-22 moieties (IL-22 muteins), as determined by differential scanning calorimetry (DSC) using a Nano DSC instrument (TA Instruments). The IL-22 muteins (D71 L / D168G (Fig. 1 1 A), R73Q (Fig. 1 1 B), and R73S (Fig. 1 1 C)) were fused to an anti-CDH17 humanized clone (AM93-015). Amino acid substitution mutations in IL-22 are numbered relative to the IL-22 sequence of SEQ ID NO: 7.
Samples were dialyzed overnight in a buffer comprised of 50 mM sodium acetate, pH 6. Samples were then subjected to a temperature gradient from 20°C-70°C, and the signal was baseline subtracted using the dialysate as a reference. Baseline subtracted data and respective curve fits are shown in Figs. 1 1 A-1 1 C. Data were fit with four peaks using the Voight with Tonset model. The Tonset (onset of degradation) values are reported in Table 12. Tonset of the first peak was interpreted to be contributed by the IL-22 moiety.
As shown in Table 12, the R73Q and R73S IL-22 muteins had Tonset at or above 50°C. Tonset of a wild-type (WT) IL-22 fused to an Fc protein was 49.8°C; thus, the R73 muteins (R73Q and R73S) do not negatively impact thermal stability of the cytokine.
Example 24. Activity of detuned IL-22 muteins on HaCaT cells
Fig. 12 shows the potency of the IL-22 muteins D71 L / D168G, R73Q, and R73S on HaCaT (human keratinocyte) cells. As discussed in Example 22, these IL-22 muteins were designed to have attenuated binding to IL-22Ra, such that they would be expected to have reduced potency on cells expressing IL-22Ra unless otherwise recruited to these cells, e.g., via a connected (e.g., fused) “addressing” moiety (e.g., an antibody or fragment thereof that targets a moiety on the cell).
In these experiments, HaCaT cells were stimulated with varying concentrations of native IL-22 or the IL-22 muteins fused to an anti-RSV antibody, then stained for phosphorylated STAT3 (pSTAT3) as a surrogate for IL-22 signaling activity. Fluorescence of the anti-pSTAT3 mAb was determined by flow cytometry.
Importantly, the R73S and R73Q muteins did not induce any measurable activity on HaCaT cells, even at the highest concentration tested (100 nM), indicating that these muteins have reduced potency on these cells relative to wild-type IL-22 and may be useful for avoiding unwanted IL-22 activity on skin.
OTHER EMBODIMENTS
Some embodiments of the technology described herein can be defined according to any of the following numbered embodiments:
1 . A macromolecule comprising a first binding site and a second binding site, wherein:
(a) the first binding site comprises an IL-22 polypeptide or a fragment or variant thereof that binds an IL-22 receptor on the surface of a target cell, and
(b) the second binding site is specific for an IL-10 family address target provided in Table 1 that is expressed on the surface of the target cell; wherein:
(i) the second binding site localizes the first binding site to the IL-10 family address target such that the IL-22 polypeptide or a fragment or variant thereof influences signaling by the IL-22 receptor in the target cell;
(ii) the second binding site does not substantially influence signaling upon binding the IL-10 family address target; and
(iii) the IL-22 polypeptide or fragment or variant thereof does not substantially influence signaling by the IL-22 receptor in the absence of localization by the second binding site.
2. The macromolecule of embodiment 1 , wherein the IL-10 family address target is a protein, lipid, or sugar.
3. The macromolecule of embodiment 2, wherein the IL-10 family address target is a protein.
4. The macromolecule of any one of embodiments 1 -3, wherein the target cell is a lower gastrointestinal (Gl) tract cell, and the IL-10 family address target is MS4A18, SI, MS4A12, ATP4B, MUC17, TMIGD1 , MS4A10, MEP1 A, CDH17, TM4SF20, SLC26A3, ATP4A, GPA33, BTNL3, MEP1 B, UGT2B17, KCNE2, or TMPRSS15.
5. The macromolecule of any one of embodiments 1 -3, wherein the target cell is a kidney cell, and the IL-10 family address target is TMEM207, OR2T10, SLC22A6, SLC22A8, SLC12A1 , AQP2, SLC22A12, SLC12A3, TMEM174, FXYD4, SLC34A1 , KCNJ1 , SLC22A1 1 , TMEM52B, SLC6A18, CTXN3, SLC4A1 , TMEM72, SLC22A2, SLC4A9, BSND, CDH16, RDH8, or AQP6.
6. The macromolecule of any one of embodiments 1 -3, wherein the target cell is a liver cell, and the IL-10 family address target is UGT2B10, C8A, SLCO1 B1 , C9, SLC25A47, SLC17A2, SLC10A1 , UGT2B4, SLC22A1 , or CYP8B1 .
7. The macromolecule of any one of embodiments 1 -3, wherein the target cell is a pancreas cell, and the IL-10 family address target is GP2, AQP12A, AQP12B, CUZD1 , LHFPL5, AQP8, KIRREL2, G6PC2, SLC17A6, or GPR1 19.
8. The macromolecule of any one of embodiments 1 -3, wherein the target cell is a skin cell, and the IL-10 family address target is CLEC2A, KCNJ18, KLRF2, DSC1 , TYR, AWAT2, TMEM271 , DSG1 , ACER1 , GSDMA, TRPM1 , GJB4, CD1 A, ASPRV1 , or ABCA12.
9. The macromolecule of any one of embodiments 1 -3, wherein the IL-10 family address target is selected from CDH17, GP2, CDH16, DSG1 , and DSG3.
10. The macromolecule of embodiment 9, wherein the IL-10 family address target is CDH17.
1 1 . The macromolecule of embodiment 9, wherein the IL-10 family address target is CDH16.
12. The macromolecule of embodiment 9, wherein the IL-10 family address target is GP2.
13. A macromolecule comprising a first binding site and a second binding site, wherein:
(a) the first binding site comprises an IL-22 polypeptide or a fragment or variant thereof that binds an IL-22 receptor on the surface of a target cell, and
(b) the second binding site is specific for an IL-10 family address target selected from CDH17, CDH16, and GP2 that is expressed on the surface of the target cell; wherein:
(i) the second binding site localizes the first binding site to the IL-10 family address target such that the IL-22 polypeptide or fragment or variant thereof influences signaling by the IL-22 receptor in the target cell;
(ii) the second binding site does not substantially influence signaling upon binding the IL-10 family address target; and
(iii) the IL-22 polypeptide or fragment or variant thereof does not substantially influence signaling by the IL-22 receptor in the absence of localization by the second binding site.
14. The macromolecule of any one of embodiments 1 -13, wherein the second binding site comprises a polypeptide.
15. The macromolecule of any one of embodiments 1 -14, wherein the second binding site is an antibody or antigen binding fragment thereof.
16. The macromolecule of embodiment 15, wherein the antibody or antigen-binding fragment thereof comprises an scFv, BsIgG, a BsAb fragment, a BiTE, a dual-affinity re-targeting protein (DART), a tandem diabody (TandAb), a diabody, an Fab2, a di-scFv, chemically linked F(ab’)2, an Ig molecule with 2, 3 or 4 different antigen binding sites, a DVI-IgG four-in-one, an ImmTac, an HSAbody, an IgG-IgG, a Cov-X-Body, an scFv1 -PEG-scFv2, an appended IgG, an DVD-IgG, an affibody, an affilin, an affimer, an affitin, an alphabody, an anticalin, an avimer, a DARPin, a Fynomer, a monobody, a nanoCLAMP, a bis- Fab, an Fv, a Fab, a Fab’-SH, a linear antibody, an scFv, an antibody with only a heavy chain (Humabody), an ScFab, an IgG antibody fragment, a single-chain variable region antibody, a singledomain heavy chain antibody, a bispecific triplebody, a BiKE, a CrossMAb, a dsDb, an scDb, tandem a dAb I VHH, a triple dAb VHH, a tetravalent dAb I VHH, a Fab-scFv, a Fab-Fv, or a DART-Fc, an adnectin, a Kunitz-type inhibitor, or a receptor decoy.
17. The macromolecule of any one of embodiments 1 -14, wherein the second binding site comprises a ligand of the address target.
18. The macromolecule of embodiment 15 or 16, wherein the IL-10 family address target is CDH17 and the second binding site is an anti-CDH17 antibody or antigen-binding fragment thereof.
19. The macromolecule of embodiment 15 or 16, wherein the IL-10 family address target is CDH16 and the second binding site is an anti-CDH16 antibody or antigen-binding fragment thereof.
20. The macromolecule of embodiment 15 or 16, wherein the IL-10 family address target is GP2 and the second binding site is an anti-GP2 antibody or antigen-binding fragment thereof.
21 . The macromolecule of any one of embodiments 1 -20, wherein the macromolecule agonizes the IL-22 receptor.
22. The macromolecule of embodiment 21 , wherein the macromolecule agonizes human IL-22Ra and/or IL-10R2.
23. The macromolecule of any one of embodiments 1 -22, wherein the IL-22 polypeptide or fragment or variant thereof has low binding affinity for IL-22Ra and/or IL-10R2.
24. The macromolecule of any one of embodiments 1 -23, wherein the affinity of the IL-22 polypeptide or fragment or variant thereof for IL-22Ra and/or IL-10R2 is lower than the affinity of the second binding site for the IL-10 family address target.
25. The macromolecule of any one of embodiments 1 -24, wherein the avidity of the IL-22 polypeptide or fragment or variant thereof for IL-22Ra and/or IL-10R2 is lower than the avidity of the second binding site for the IL-10 family address target.
26. The macromolecule of any one of embodiments 1 -25, wherein:
(a) the Kd of the IL-22 polypeptide or fragment or variant thereof for IL-22Ra and/or IL-10R2 is higher than the Kd of the second binding site for the IL-10 family address target;
(b) the ECso of the IL-22 polypeptide or fragment or variant thereof for IL-22Ra and/or IL-10R2 is higher than the EC50 of the second binding site for the IL-10 family address target; or
(c) the IC50 of the IL-22 polypeptide or fragment or variant thereof for IL-22Ra and/or IL-10R2 is higher than the IC50 of the second binding site for the IL-10 family address target.
27. The macromolecule of any one of embodiments 1 -26, wherein the IL-22 polypeptide or fragment or variant thereof has an affinity to the IL-22 receptor of at least about 2 times, at least about 5 times, or at least about 10 times less than the affinity of the second binding site to the IL-10 family address target.
28. The macromolecule of any one of embodiments 1 -27, wherein the affinity of the second binding site to the IL-10 family address target has a Kd of greater than about 1 nM, greater than about 2 nM, or greater than about 50 nm.
29. The macromolecule of any one of embodiments 1 -28, wherein the IL-22 polypeptide or fragment or variant thereof has been engineered to have reduced binding affinity for the IL-22 receptor.
30. The macromolecule of any one of embodiments 1 -29, wherein, when part of the macromolecule, the IL-22 polypeptide or fragment or variant thereof exhibits reduced agonist activity relative to the IL-22 polypeptide or fragment or variant thereof when not part of the macromolecule, on a mole-adjusted basis.
31 . The macromolecule of any one of embodiments 1 -30, wherein the IL-22 polypeptide or fragment or variant thereof comprises one or more amino acid substitution mutations at one or more residues that are predicted to be interaction surfaces between IL-22 and IL-22Ra and/or IL-10R2.
32. The macromolecule of any one of embodiments 1 -31 , wherein the IL-22 polypeptide or fragment or variant thereof has reduced binding affinity for IL22RA1 relative to a wild-type IL-22 or fragment thereof.
33. The macromolecule of embodiment 32, wherein the IL-22 polypeptide or fragment or variant thereof comprises one or more amino acid substitution mutations at one or more residues that are predicted to be interaction surfaces between IL-22 and IL-22Ra.
34. The macromolecule of embodiment 32 or 33, wherein the IL-22 polypeptide or fragment or variant thereof is a human IL-22 or fragment thereof and comprises one or more of the amino acid substitution mutations of Table 6, Table 7A, Table 7B, Table 8, Table 9A, Table 9B, or Table 9C.
35. The macromolecule of embodiment 34, wherein the human IL-22 polypeptide or fragment or variant thereof comprises one or more of a K61 S, D71 N, D71 L, D71 G, R73Q, R73S, R73N, D168N, D168G, and R175S amino acid substitution mutation.
36. The macromolecule of embodiment 35, wherein the human IL-22 polypeptide or fragment or variant thereof comprises a D71 L amino acid substitution mutation.
37. The macromolecule of embodiment 36, wherein the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 1 1 .
38. The macromolecule of embodiment 37, wherein the human IL-22 polypeptide or fragment or variant thereof comprises a R73Q amino acid substitution mutation.
39. The macromolecule of embodiment 38, wherein the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 12.
40. The macromolecule of embodiment 35, wherein the human IL-22 polypeptide or fragment or variant thereof comprises D71 L and D168G amino acid substitution mutations.
41 . The macromolecule of embodiment 40, wherein the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 13.
42. The macromolecule of any one of embodiments 1 -41 , wherein the macromolecule comprises two first binding sites comprising an IL-22 polypeptide or fragment or variant thereof that binds IL-22Ra and/or IL-10R2.
43. The macromolecule of embodiment 42, wherein the two first binding sites each bind to IL-22Ra or each bind to IL-10R2.
44. The macromolecule of any one of embodiments 1 -43, wherein the macromolecule comprises two second binding sites specific for an IL-10 family address target expressed on the surface of the target cell.
45. The macromolecule of embodiment 44, wherein the two second binding sites are specific for the same IL-10 family address target.
46. The macromolecule of any one of embodiments 43-45, wherein the macromolecule is a homodimer comprising two subunits, each subunit comprising the first binding site and the second binding site.
47. The macromolecule of embodiment 46, wherein the two subunits are fusion proteins.
48. The macromolecule of embodiment 47, wherein each of the fusion proteins comprises a linker conjugated to the first binding site and the second binding site.
49. The macromolecule of embodiment 48, wherein the linker is a GS linker.
50. The macromolecule of any one of embodiments 46-49, wherein the two subunits are substantially identical.
51 . A macromolecule comprising two subunits, each comprising a first binding site and a second binding site, wherein:
(a) each of the first binding sites comprises an IL-22 polypeptide or a fragment or variant thereof that binds an IL-22 receptor on the surface of a target cell, and
(b) each of the second binding sites is specific for an IL-10 family address target provided in Table 1 that is expressed on the surface of the target cell; wherein:
(i) the second binding sites localize the first binding sites to the IL-10 family address target such that the IL-22 polypeptides or fragments or variants thereof influence signaling by the IL-22 receptor in the target cell;
(ii) the second binding sites do not substantially influence signaling upon binding the IL-10 family address targets; and
(iii) the IL-22 polypeptides or fragments or variants thereof do not substantially influence signaling by the IL-22 receptor in the absence of localization by the second binding sites.
52. The macromolecule of embodiment 51 , wherein the two subunits are substantially identical.
53. A macromolecule comprising two subunits, each comprising a first binding site and a second binding site, wherein:
(a) each of the first binding sites comprises an IL-22 polypeptide or a fragment or variant thereof that binds an IL-22 receptor on the surface of a target cell, and
(b) each of the second binding sites is specific for an IL-10 family address target selected from CDH17, CDH16, and GP2 that is expressed on the surface of the target cell; wherein:
(i) the second binding sites localize the first binding sites to the IL-10 family address target such that the IL-22 polypeptides or fragments or variants thereof influence signaling by the IL-22 receptor in the target cell;
(ii) the second binding sites do not substantially influence signaling upon binding the IL-10 family address targets; and
(iii) the IL-22 polypeptides or fragments or variants thereof do not substantially influence signaling by the IL-22 receptor in the absence of localization by the second binding sites.
54. The macromolecule of embodiment 53, wherein the two subunits are substantially identical.
55. The macromolecule of any one of embodiments 1 -41 , wherein the macromolecule comprises a single first binding site.
56. The macromolecule of any one of embodiments 1 -41 and 55, wherein the macromolecule comprises a single second binding site.
57. The macromolecule of embodiment 55 or 56, wherein the macromolecule is a heterodimer comprising two subunits, wherein the first subunit comprises the first binding site and second subunit comprises the second binding site.
58. The macromolecule of embodiment 57, wherein the macromolecule is a fusion protein.
59. The macromolecule of embodiment 58, wherein the fusion protein comprises a linker conjugated to the first binding site and the second binding site.
60. The macromolecule of embodiment 59, wherein the linker is a GS linker.
61 . The macromolecule of any one of embodiments 1 -60, wherein binding of the second binding site to the IL-10 family address target (i) increases accumulation of the macromolecule in tissues expressing
the IL-10 family address target, (ii) reduces accumulation of the macromolecule in tissues that do not express the IL-10 family address target, or (iii) both (i) and (ii).
62. A method of modulating IL-22 receptor signaling in a target cell of a subject, the method comprising administering to the subject the macromolecule of any one of embodiments 1 -61 .
63. A macromolecule comprising:
(a) a first binding site comprising an IL-22 polypeptide or a fragment or variant thereof, and
(b) a second binding site comprising an anti-CDH17 antibody or antigen-binding fragment thereof, wherein the macromolecule is a fusion protein, and wherein the first binding site and the second binding site are connected by a linker.
64. The macromolecule of embodiment 63, wherein:
(i) the anti-CDH17 antibody or antigen-binding fragment thereof localizes the first binding site to an IL-22 receptor such that the IL-22 polypeptide or fragment or variant thereof influences signaling by the IL-22 receptor in the target cell;
(ii) the anti-CDH17 antibody or antigen-binding fragment thereof does not substantially influence signaling upon binding CDH17; and
(iii) the IL-22 polypeptide or fragment or variant thereof does not substantially influence signaling by the IL-22 receptor in the absence of localization by the anti-CDH17 antibody or antigen-binding fragment thereof.
65. A macromolecule comprising two subunits, each comprising a first binding site and a second binding site, wherein:
(a) each first binding site comprises an IL-22 polypeptide or a fragment or variant thereof, and
(b) each second binding site comprises an anti-CDH17 antibody or antigen-binding fragment thereof, wherein the macromolecule is a fusion protein, and wherein the first binding site and the second binding site of each subunit are connected by a linker.
66. The macromolecule of embodiment 65, wherein:
(i) the anti-CDH17 antibodies or antigen-binding fragments thereof localize the first binding sites to an IL-22 receptor such that the IL-22 polypeptides or fragments or variants thereof influence signaling by the IL-22 receptor in the target cell;
(ii) the anti-CDH17 antibodies or antigen-binding fragments thereof do not substantially influence signaling upon binding CDH17; and
(iii) the IL-22 polypeptides or fragments or variants thereof do not substantially influence signaling by the IL-22 receptor in the absence of localization by the anti-CDH17 antibodies or antigen-binding fragments thereof.
67. A method of modulating IL-22 receptor signaling in the small intestine or colon of a subject, the method comprising administering to the subject the macromolecule of any one of embodiments 63-66.
68. A macromolecule comprising:
(a) a first binding site comprising an IL-22 polypeptide or a fragment or variant thereof, and
(b) a second binding site comprising an anti-CDH16 antibody or antigen-binding fragment thereof, wherein the macromolecule is a fusion protein, and wherein the first binding site and the second binding site are connected by a linker.
69. The macromolecule of embodiment 68, wherein:
(i) the anti-CDH16 antibody or antigen-binding fragment thereof localizes the first binding site to an IL-22 receptor such that the IL-22 polypeptide or fragment or variant thereof influences signaling by the IL-22 receptor in the target cell;
(ii) the anti-CDH16 antibody or antigen-binding fragment thereof does not substantially influence signaling upon binding CDH16; and
(iii) the IL-22 polypeptide or fragment or variant thereof does not substantially influence signaling by the IL-22 receptor in the absence of localization by the anti-CDH16 antibody or antigen-binding fragment thereof.
70. A macromolecule comprising two subunits, each comprising a first binding site and a second binding site, wherein:
(a) each first binding site comprises an IL-22 polypeptide or a fragment or variant thereof, and
(b) each second binding site comprises an anti-CDH16 antibody or antigen-binding fragment thereof, wherein the macromolecule is a fusion protein, and wherein the first binding site and the second binding site of each subunit are connected by a linker.
71 . The macromolecule of embodiment 70, wherein:
(i) the anti-CDH16 antibodies or antigen-binding fragments thereof localize the first binding sites to an IL-22 receptor such that the IL-22 polypeptides or fragments or variants thereof influence signaling by the IL-22 receptor in the target cell;
(ii) the anti-CDH16 antibodies or antigen-binding fragments thereof do not substantially influence signaling upon binding CDH16; and
(iii) the IL-22 polypeptides or fragments or variants thereof do not substantially influence signaling by the IL-22 receptor in the absence of localization by the anti-CDH16 antibodies or antigen-binding fragments thereof.
72. A method of modulating IL-22 receptor signaling in the kidney of a subject, the method comprising administering to the subject the macromolecule of any one of embodiments 68-71 .
73. A macromolecule comprising:
(a) a first binding site comprising an IL-22 polypeptide or a fragment or variant thereof, and
(b) a second binding site comprising an anti-GP2 antibody or antigen-binding fragment thereof,
wherein the macromolecule is a fusion protein, and wherein the first binding site and the second binding site are connected by a linker.
74. The macromolecule of embodiment 73, wherein:
(i) the anti-GP2 antibody or antigen-binding fragment thereof localizes the first binding site to an IL- 22 receptor such that the IL-22 polypeptide or fragment or variant thereof influences signaling by the IL- 22 receptor in the target cell;
(ii) the anti-GP2 antibody or antigen-binding fragment thereof does not substantially influence signaling upon binding GP2; and
(iii) the IL-22 polypeptide or fragment or variant thereof does not substantially influence signaling by the IL-22 receptor in the absence of localization by the anti-GP2 antibody or antigen-binding fragment thereof.
75. A macromolecule comprising two subunits, each comprising a first binding site and a second binding site, wherein:
(a) each first binding site comprises an IL-22 polypeptide or a fragment or variant thereof, and
(b) each second binding site comprises an anti-GP2 antibody or antigen-binding fragment thereof, wherein the macromolecule is a fusion protein, and wherein the first binding site and the second binding site of each subunit are connected by a linker.
76. The macromolecule of embodiment 75, wherein:
(i) the anti-GP2 antibodies or antigen-binding fragments thereof localize the first binding sites to an IL-22 receptor such that the IL-22 polypeptides or fragments or variants thereof influence signaling by the IL-22 receptor in the target cell;
(ii) the anti-GP2 antibodies or antigen-binding fragments thereof do not substantially influence signaling upon binding GP2; and
(iii) the IL-22 polypeptides or fragments or variants thereof do not substantially influence signaling by the IL-22 receptor in the absence of localization by the second binding sites.
77. A method of modulating IL-22 receptor signaling in the pancreas of a subject, the method comprising administering to the subject the macromolecule of any one of embodiments 73-76.
78. The macromolecule of any one of embodiments 63-66, 68-71 , and 73-76, wherein the IL-22 polypeptide or fragment or variant thereof is a human IL-22 polypeptide or fragment or variant thereof and comprises one or more of the amino acid substitution mutations of Table 6, Table 7A, Table 7B, Table 8, Table 9A, Table 9B, or Table 9C.
79. The macromolecule of embodiment 78, wherein the human IL-22 polypeptide or fragment or variant thereof comprises one or more of a K61 S, D71 N, D71 L, D71 G, R73Q, R73S, R73N, D168N, D168G, and R175S amino acid substitution mutation.
80. The macromolecule of embodiment 79, wherein the human IL-22 polypeptide or fragment or variant thereof comprises a D71 L amino acid substitution mutation.
81 . The macromolecule of embodiment 80, wherein the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 11 .
82. The macromolecule of embodiment 79, wherein the human IL-22 polypeptide or fragment or variant thereof comprises a R73Q amino acid substitution mutation.
83. The macromolecule of embodiment 82, wherein the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 12.
84. The macromolecule of embodiment 79, wherein the human IL-22 polypeptide or fragment or variant thereof comprises D71 L and D168G amino acid substitution mutations.
85. The macromolecule of embodiment 84, wherein the human IL-22 polypeptide or fragment or variant thereof has at least 90% identity to SEQ ID NO: 13.
86. A composition comprising the macromolecule of any one of embodiments 1 -61 , 63-66, 68-71 , 73- 76, and 78-85.
87. The composition of embodiment 86, wherein the composition comprises a pharmaceutically acceptable carrier or excipient.
88. The composition of embodiment 86 or 87, wherein the composition is a unit dosage form suitable for administration to a human subject.
89. One or more nucleic acids encoding the macromolecule of any one of embodiments 1 -61 , 63-66, 68-71 , 73-76, and 78-85 or one or more subunits thereof.
90. The one or more nucleic acids of embodiment 89, wherein the nucleic acids are linear nucleic acids, circular nucleic acids, and/or modified nucleic acids.
91 . One or more vectors comprising the one or more nucleic acids of embodiment 89 or 90.
92. One or more host cells comprising the one or more vectors of embodiment 91 .
93. A method of producing the macromolecule of any one of embodiments 1 -61 , 63-66, 68-71 , 73-76, and 78-85, the method comprising culturing the one or more host cells of embodiment 101 in a culture medium.
94. The method of embodiment 93, wherein the method further comprises recovering the macromolecule from the one or more host cells or the culture medium.
95. A method comprising administering a therapeutically effective amount of the macromolecule, composition, or nucleic acid of any one of embodiments 1 -61 , 63-66, 68-71 , 73-76, and 78-85 to a subject in need thereof.
96. The method of embodiment 95, wherein the subject has, is suspected of having, or is at increased risk of developing a disease or disorder selected from: Crohn's disease, ulcerative colitis, graft- vs-host disease, atopic dermatitis, psoriasis, wounds, diabetic skin ulcers, acute pancreatitis, chronic pancreatitis, autoimmune pancreatitis, nephritis, acute lung injury, viral lung injury, fibrosis, fibrosis, nonalcoholic steatohepatitis, viral hepatitis, and autoimmune hepatitis.
97. A human IL-22 polypeptide or a fragment or variant thereof that binds an IL-22 receptor on the surface of a target cell, wherein the IL-22 polypeptide or fragment or variant thereof comprises one or more of the amino acid substitution mutations of Table 6, Table 7A, Table 7B, Table 8, Table 9A, Table 9B, or Table 9C.
98. A human IL-22 polypeptide or a fragment or variant thereof that binds an IL-22 receptor on the surface of a target cell, wherein the human IL-22 polypeptide or fragment or variant thereof comprises an amino acid substitution mutation of one or more of K61 , D71 , R73, D168, and R175, wherein the amino acid position numbering is with reference to the sequence of SEQ ID NO: 7.
99. The human IL-22 polypeptide or a fragment or variant thereof of embodiment 98, wherein the human IL-22 polypeptide or fragment or variant thereof comprises one or more of a K61 S, D71 N, D71 L, D71 G, R73Q, R73S, R73N, D168N, D168G, and R175S amino acid substitution mutation.
100. The human IL-22 polypeptide or a fragment or variant thereof of embodiment 98, wherein the human IL-22 polypeptide or fragment or variant thereof comprises a K61 S amino acid substitution mutation.
101 . The human IL-22 polypeptide or a fragment or variant thereof of embodiment 98, wherein the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 17.
102. The human IL-22 polypeptide or a fragment or variant thereof of embodiment 98, wherein the human IL-22 polypeptide or fragment or variant thereof comprises a D71 G amino acid substitution mutation.
103. The human IL-22 polypeptide or a fragment or variant thereof of embodiment 98, wherein the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 15.
104. The human IL-22 polypeptide or a fragment or variant thereof of embodiment 98, wherein the human IL-22 polypeptide or fragment or variant thereof comprises a D71 L amino acid substitution mutation.
105. The human IL-22 polypeptide or a fragment or variant thereof of embodiment 98, wherein the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 11 .
106. The human IL-22 polypeptide or a fragment or variant thereof of embodiment 98, wherein the human IL-22 polypeptide or fragment or variant thereof comprises a D71 N amino acid substitution mutation.
107. The human IL-22 polypeptide or a fragment or variant thereof of embodiment 98, wherein the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 18.
108. The human IL-22 polypeptide or a fragment or variant thereof of embodiment 98, wherein the human IL-22 polypeptide or fragment or variant thereof comprises a R73S amino acid substitution mutation.
109. The human IL-22 polypeptide or a fragment or variant thereof of embodiment 98, wherein the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 16.
110. The human IL-22 polypeptide or a fragment or variant thereof of embodiment 98, wherein the human IL-22 polypeptide or fragment or variant thereof comprises a R73Q amino acid substitution mutation.
111. The human IL-22 polypeptide or a fragment or variant thereof of embodiment 98, wherein the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 12.
112. The human IL-22 polypeptide or a fragment or variant thereof of embodiment 98, wherein the human IL-22 polypeptide or fragment or variant thereof comprises a D168N amino acid substitution mutation.
113. The human IL-22 polypeptide or a fragment or variant thereof of embodiment 98, wherein the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 19.
114. The human IL-22 polypeptide or a fragment or variant thereof of embodiment 98, wherein the human IL-22 polypeptide or fragment or variant thereof comprises a D168G amino acid substitution mutation.
115. The human IL-22 polypeptide or a fragment or variant thereof of embodiment 98, wherein the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 20.
116. The human IL-22 polypeptide or a fragment or variant thereof of embodiment 98, wherein the human IL-22 polypeptide or fragment or variant thereof comprises D71 L and D168G amino acid substitution mutations.
117. The human IL-22 polypeptide or a fragment or variant thereof of embodiment 98, wherein the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 13.
118. The human IL-22 polypeptide or a fragment or variant thereof of embodiment 98, wherein the human IL-22 polypeptide or fragment or variant thereof comprises a R175S amino acid substitution mutation.
119. The human IL-22 polypeptide or a fragment or variant thereof of embodiment 98, wherein the human IL-22 polypeptide or fragment or variant thereof comprises SEQ ID NO: 21 .
120. The human IL-22 polypeptide or a fragment or variant thereof of any one of embodiments 98 to 119, wherein the human IL-22 polypeptide or a fragment or variant thereof agonizes the IL-22 receptor.
121 . The human IL-22 polypeptide or a fragment or variant thereof of any one of embodiments 98 to 119, wherein the human IL-22 polypeptide or a fragment or variant thereof agonizes human IL-22Ra and/or IL-10R2.
122. The human IL-22 polypeptide or a fragment or variant thereof of any one of embodiments 98 to 119, wherein the human IL-22 polypeptide or fragment or variant thereof has low binding affinity for IL- 22Ra and/or IL-10R2.
123. The human IL-22 polypeptide or a fragment or variant thereof of any one of embodiments 98 to 119, wherein the human IL-22 polypeptide or fragment or variant thereof has an affinity to the IL-22 receptor of at least about 2 times, at least about 5 times, or at least about 10 times less than the affinity of the second binding site to the IL-10 family address target.
124. The human IL-22 polypeptide or a fragment or variant thereof of any one of embodiments 98 to 119, wherein the human IL-22 polypeptide or fragment or variant thereof has been engineered to have reduced binding affinity for the IL-22 receptor.
125. The human IL-22 polypeptide or a fragment or variant thereof of any one of embodiments 98 to 119, wherein the human IL-22 polypeptide or fragment or variant thereof comprises one or more amino acid substitution mutations at one or more residues that are predicted to be interaction surfaces between IL-22 and IL-22Ra and/or IL-10R2.
126. The human IL-22 polypeptide or a fragment or variant thereof of any one of embodiments 98 to 119, wherein the human IL-22 polypeptide or fragment or variant thereof has reduced binding affinity for IL22RA1 (IL-22Ra) relative to a wild-type IL-22 or fragment thereof.
127. The human IL-22 polypeptide or a fragment or variant thereof of any one of embodiments 98 to 119, wherein the human IL-22 polypeptide or fragment or variant thereof comprises one or more amino acid substitution mutations at one or more residues that are predicted to be interaction surfaces between IL-22 and IL-22Ra.
128. The human IL-22 polypeptide or a fragment or variant thereof of any one of embodiments 98 to 127, wherein the human IL-22 polypeptide or fragment or variant thereof does not comprise a leader sequence.
129. The human IL-22 polypeptide or a fragment or variant thereof of any one of embodiments 98 to 127, wherein the human IL-22 polypeptide or fragment or variant thereof does not comprise the sequence of SEQ ID NO: 14.
130. A polypeptide comprising the human IL-22 polypeptide or a fragment or variant thereof of any one of embodiments 98 to 129.
131 . A composition comprising a polypeptide of any one of embodiments 98 to 130.
132. The composition of embodiment 131 , wherein the composition comprises a pharmaceutically acceptable carrier or excipient.
133. The composition of embodiment 131 or 132, wherein the composition is a unit dosage form suitable for administration to a human subject.
134. A nucleic acid encoding a polypeptide of any one of embodiments 98 to 130.
135. The nucleic acid of embodiment 134, wherein the nucleic acid is a linear nucleic acid, a circular nucleic acid, and/or a modified nucleic acid.
136. A vector comprising the nucleic acid of embodiment 134 or 135.
137. A host cell comprising the vector of embodiment 136.
138. A method of producing a polypeptide of any one of embodiments 98 to 130, the method comprising culturing the host cell of embodiment 137 in a culture medium.
139. The method of embodiment 138, wherein the method further comprises recovering the polypeptide from the host cell or the culture medium.
140. An IL-22 mutein polypeptide or a fragment or variant thereof that binds an IL-22 receptor on the surface of a target cell, wherein the IL-22 mutein polypeptide or fragment or variant thereof comprises one or more of the amino acid substitution mutations provided in Table 6, Table 7A, Table 7B, Table 8, Table 9A, Table 9B, or Table 9C, wherein the amino acid positions are numbered relative to SEQ ID NO: 7.
141 . An IL-22 mutein polypeptide or a fragment or variant thereof that binds an IL-22 receptor on the surface of a target cell, wherein the IL-22 mutein polypeptide or fragment or variant thereof comprises an amino acid substitution mutation at one or more of positions K61 , D71 , R73, D168, and R175 relative to a human IL-22 polypeptide, wherein the amino acid positions are numbered relative to SEQ ID NO: 7.
142. The IL-22 mutein polypeptide or fragment or variant thereof of embodiment 141 , wherein the IL- 22 polypeptide or fragment or variant thereof comprises one or more of a K61 S, D71 N, D71 L, D71 G, R73Q, R73S, R73N, D168N, D168G, and R175S amino acid substitution mutation.
143. The IL-22 mutein polypeptide or fragment or variant thereof of embodiment 141 , wherein:
(i) the IL-22 mutein polypeptide or fragment or variant thereof comprises a K61 S amino acid substitution mutation, optionally wherein the IL-22 mutein polypeptide or fragment or variant thereof comprises SEQ ID NO: 17;
(ii) the IL-22 mutein polypeptide or fragment or variant thereof comprises a D71 G amino acid substitution mutation, optionally wherein the IL-22 mutein polypeptide or fragment or variant thereof comprises SEQ ID NO: 15;
(Hi) the IL-22 mutein polypeptide or fragment or variant thereof comprises a D71 L amino acid substitution mutation, optionally wherein the IL-22 mutein polypeptide or fragment or variant thereof comprises SEQ ID NO: 11 ;
(iv) the IL-22 mutein polypeptide or fragment or variant thereof comprises a D71 N amino acid substitution mutation, optionally wherein the IL-22 mutein polypeptide or fragment or variant thereof comprises SEQ ID NO: 18;
(v) the IL-22 mutein polypeptide or fragment or variant thereof comprises an R73S amino acid substitution mutation, optionally wherein the IL-22 mutein polypeptide or fragment or variant thereof comprises SEQ ID NO: 16;
(vi) the IL-22 mutein polypeptide or fragment or variant thereof comprises an R73Q amino acid substitution mutation, optionally wherein the IL-22 mutein polypeptide or fragment or variant thereof comprises SEQ ID NO: 12;
(vii) the IL-22 mutein polypeptide or fragment or variant thereof comprises a D168N amino acid substitution mutation, optionally wherein the IL-22 mutein polypeptide or fragment or variant thereof comprises SEQ ID NO: 19;
(viii) the IL-22 mutein polypeptide or fragment or variant thereof comprises a D168G amino acid substitution mutation, optionally wherein the IL-22 mutein polypeptide or fragment or variant thereof comprises SEQ ID NO: 20;
(ix) the IL-22 mutein polypeptide or fragment or variant thereof comprises D71 L and D168G amino acid substitution mutations, optionally wherein the IL-22 mutein polypeptide or fragment or variant thereof comprises SEQ ID NO: 13;
(x) the IL-22 mutein polypeptide or fragment or variant thereof comprises an R175S amino acid substitution mutation, optionally wherein the IL-22 mutein polypeptide or fragment or variant thereof comprises SEQ ID NO: 21 ; or
(xi) the IL-22 mutein polypeptide or fragment or variant thereof comprises an R73N amino acid substitution mutation, optionally wherein the IL-22 mutein polypeptide or fragment or variant thereof comprises SEQ ID NO: 22.
144. The IL-22 mutein polypeptide or fragment or variant thereof of any one of embodiments 140- 144, wherein the IL-22 mutein polypeptide or fragment or variant thereof comprises one or more additional amino acid substitution mutations at one or more residues that are predicted to be interaction surfaces between IL-22 and IL-22Ra and/or IL-10R2.
145. The IL-22 mutein polypeptide or fragment or variant thereof of any one of embodiments 140-
144, wherein the IL-22 mutein polypeptide or fragment or variant thereof has a binding affinity for IL22RA1 (IL-22Ra) that is at least 1 .3-fold lower than the binding affinity of a wild-type IL-22 or fragment thereof.
146. The IL-22 mutein polypeptide or fragment or variant thereof of any one of embodiments 140-
145, wherein the IL-22 mutein polypeptide or fragment or variant thereof does not comprise a leader sequence.
147. The IL-22 mutein polypeptide or fragment or variant thereof of any one of embodiments 140-
146, wherein the IL-22 mutein polypeptide or fragment or variant thereof does not comprise the amino acid sequence of SEQ ID NO: 14.
148. A polypeptide comprising the IL-22 mutein polypeptide or fragment or variant thereof of any one of embodiments 140-147.
149. An IL-22 mutein polypeptide that has at least 95% identity to human IL-22 (SEQ ID NO: 7), comprises D71 L and D168G amino acid substitution mutations relative to SEQ ID NO: 7, and has a significantly reduced binding affinity for IL-22Ra relative to a wild-type human IL-22 polypeptide.
150. An IL-22 mutein polypeptide that has at least 95% identity to human IL-22 (SEQ ID NO: 7), comprises an R73Q amino acid substitution mutation relative to SEQ ID NO: 7, and has a significantly reduced binding affinity for IL-22Ra relative to a wild-type human IL-22 polypeptide.
151 . An IL-22 mutein polypeptide that has at least 95% identity to human IL-22 (SEQ ID NO: 7), comprises an R73S amino acid substitution mutation relative to SEQ ID NO: 7, and has a significantly reduced binding affinity for IL-22Ra relative to a wild-type human IL-22 polypeptide.
152. A macromolecule comprising a first binding site and a second binding site, wherein:
(a) the first binding site comprises an IL-22 polypeptide or a fragment or variant thereof that binds an IL-22 receptor on the surface of a target cell, and
(b) the second binding site is specific for an IL-10 family address target provided in Table 1 that is expressed on the surface of the target cell; wherein:
(i) the second binding site localizes the first binding site to the IL-10 family address target such that the IL-22 polypeptide or a fragment or variant thereof influences signaling by the IL-22 receptor in the target cell;
(ii) the second binding site does not substantially influence signaling upon binding the IL-10 family address target; and
(iii) the IL-22 polypeptide or fragment or variant thereof does not substantially influence signaling by the IL-22 receptor in the absence of localization by the second binding site.
153. The macromolecule of embodiment 152, wherein the IL-22 polypeptide or fragment or variant thereof has been engineered to have reduced binding affinity for the IL-22 receptor.
154. The macromolecule of embodiment 152 or 153, wherein the IL-22 polypeptide or fragment or variant thereof comprises one or more amino acid substitution mutations at one or more residues that are predicted to be interaction surfaces between IL-22 and IL-22Ra and/or IL-10R2.
155. The macromolecule of embodiment 154, wherein the IL-22 polypeptide or fragment or variant thereof is a variant of a human IL-22 or fragment thereof and comprises:
(a) one or more of the amino acid substitution mutations of Table 6, Table 7A, Table 7B, Table 8, Table 9A, Table 9B, or Table 9C; and/or
(b) one or more of a K61 S, D71 N, D71 L, D71 G, R73Q, R73S, R73N, D168N, D168G, and R175S amino acid substitution mutation.
156. A method of modulating IL-22 receptor signaling in a target cell of a subject, the method comprising administering to the subject the macromolecule of any one of embodiments 152-154.
157. A method of treating a disease or disorder selected from Crohn's disease, ulcerative colitis, graft-vs-host disease, atopic dermatitis, psoriasis, wounds, diabetic skin ulcers, acute pancreatitis, chronic pancreatitis, autoimmune pancreatitis, nephritis, acute lung injury, viral lung injury, fibrosis, fibrosis, non-alcoholic steatohepatitis, viral hepatitis, and autoimmune hepatitis in a subject, the method comprising administering to the subject the macromolecule of any one of embodiments 152-154.
158. A macromolecule comprising:
(a) a first binding site comprising an IL-22 polypeptide or a fragment or variant thereof, and
(b) a second binding site comprising an anti-CDH17 antibody or antigen-binding fragment thereof, wherein the macromolecule is a fusion protein, and wherein the first binding site and the second binding site are connected by a linker.
Although the foregoing invention has been described in some detail by way of illustration and example for purposes of clarity of understanding, the descriptions and examples should not be construed as limiting the scope of the invention.
Claims
1 . An IL-22 mutein polypeptide or a fragment or variant thereof that binds an IL-22 receptor on the surface of a target cell, wherein the IL-22 mutein polypeptide or fragment or variant thereof comprises one or more of the amino acid substitution mutations provided in Table 6, Table 7A, Table 7B, Table 8, Table 9A, Table 9B, or Table 9C, wherein the amino acid positions are numbered relative to SEQ ID NO: 7.
2. An IL-22 mutein polypeptide or a fragment or variant thereof that binds an IL-22 receptor on the surface of a target cell, wherein the IL-22 mutein polypeptide or fragment or variant thereof comprises an amino acid substitution mutation at one or more of positions K61 , D71 , R73, D168, and R175 relative to a human IL-22 polypeptide, wherein the amino acid positions are numbered relative to SEQ ID NO: 7.
3. The IL-22 mutein polypeptide or fragment or variant thereof of claim 2, wherein the IL-22 polypeptide or fragment or variant thereof comprises one or more of a K61 S, D71 N, D71 L, D71 G, R73Q, R73S, R73N, D168N, D168G, and R175S amino acid substitution mutation.
4. The IL-22 mutein polypeptide or fragment or variant thereof of claim 2, wherein:
(i) the IL-22 mutein polypeptide or fragment or variant thereof comprises a K61 S amino acid substitution mutation, optionally wherein the IL-22 mutein polypeptide or fragment or variant thereof comprises SEQ ID NO: 17;
(ii) the IL-22 mutein polypeptide or fragment or variant thereof comprises a D71 G amino acid substitution mutation, optionally wherein the IL-22 mutein polypeptide or fragment or variant thereof comprises SEQ ID NO: 15;
(iii) the IL-22 mutein polypeptide or fragment or variant thereof comprises a D71 L amino acid substitution mutation, optionally wherein the IL-22 mutein polypeptide or fragment or variant thereof comprises SEQ ID NO: 11 ;
(iv) the IL-22 mutein polypeptide or fragment or variant thereof comprises a D71 N amino acid substitution mutation, optionally wherein the IL-22 mutein polypeptide or fragment or variant thereof comprises SEQ ID NO: 18;
(v) the IL-22 mutein polypeptide or fragment or variant thereof comprises an R73S amino acid substitution mutation, optionally wherein the IL-22 mutein polypeptide or fragment or variant thereof comprises SEQ ID NO: 16;
(vi) the IL-22 mutein polypeptide or fragment or variant thereof comprises an R73Q amino acid substitution mutation, optionally wherein the IL-22 mutein polypeptide or fragment or variant thereof comprises SEQ ID NO: 12;
(vii) the IL-22 mutein polypeptide or fragment or variant thereof comprises a D168N amino acid substitution mutation, optionally wherein the IL-22 mutein polypeptide or fragment or variant thereof comprises SEQ ID NO: 19;
(viii) the IL-22 mutein polypeptide or fragment or variant thereof comprises a D168G amino acid
substitution mutation, optionally wherein the IL-22 mutein polypeptide or fragment or variant thereof comprises SEQ ID NO: 20;
(ix) the IL-22 mutein polypeptide or fragment or variant thereof comprises D71 L and D168G amino acid substitution mutations, optionally wherein the IL-22 mutein polypeptide or fragment or variant thereof comprises SEQ ID NO: 13;
(x) the IL-22 mutein polypeptide or fragment or variant thereof comprises an R175S amino acid substitution mutation, optionally wherein the IL-22 mutein polypeptide or fragment or variant thereof comprises SEQ ID NO: 21 ; or
(xi) the IL-22 mutein polypeptide or fragment or variant thereof comprises an R73N amino acid substitution mutation, optionally wherein the IL-22 mutein polypeptide or fragment or variant thereof comprises SEQ ID NO: 22.
5. The IL-22 mutein polypeptide or fragment or variant thereof of any one of claims 1 -4, wherein the IL-22 mutein polypeptide or fragment or variant thereof comprises one or more additional amino acid substitution mutations at one or more residues that are predicted to be interaction surfaces between IL- 22 and IL-22Ra and/or IL-10R2.
6. The IL-22 mutein polypeptide or fragment or variant thereof of any one of claims 1 -5, wherein the IL-22 mutein polypeptide or fragment or variant thereof has a binding affinity for IL22RA1 (IL-22Ra) that is at least 1 .3-fold lower than the binding affinity of a wild-type IL-22 or fragment thereof.
7. The IL-22 mutein polypeptide or fragment or variant thereof of any one of claims 1 -6, wherein the IL-22 mutein polypeptide or fragment or variant thereof does not comprise a leader sequence.
8. The IL-22 mutein polypeptide or fragment or variant thereof of any one of claims 1 -7, wherein the IL-22 mutein polypeptide or fragment or variant thereof does not comprise the amino acid sequence of SEQ ID NO: 14.
9. A polypeptide comprising the IL-22 mutein polypeptide or fragment or variant thereof of any one of claims 1 -8.
10. An IL-22 mutein polypeptide that has at least 95% identity to human IL-22 (SEQ ID NO: 7), comprises D71 L and D168G amino acid substitution mutations relative to SEQ ID NO: 7, and has a significantly reduced binding affinity for IL-22Ra relative to a wild-type human IL-22 polypeptide.
11 . An IL-22 mutein polypeptide that has at least 95% identity to human IL-22 (SEQ ID NO: 7), comprises an R73Q amino acid substitution mutation relative to SEQ ID NO: 7, and has a significantly reduced binding affinity for IL-22Ra relative to a wild-type human IL-22 polypeptide.
12. An IL-22 mutein polypeptide that has at least 95% identity to human IL-22 (SEQ ID NO: 7), comprises an R73S amino acid substitution mutation relative to SEQ ID NO: 7, and has a significantly reduced binding affinity for IL-22Ra relative to a wild-type human IL-22 polypeptide.
13. A macromolecule comprising a first binding site and a second binding site, wherein:
(a) the first binding site comprises an IL-22 polypeptide or a fragment or variant thereof that binds an IL-22 receptor on the surface of a target cell, and
(b) the second binding site is specific for an IL-10 family address target provided in Table 1 that is expressed on the surface of the target cell; wherein:
(i) the second binding site localizes the first binding site to the IL-10 family address target such that the IL-22 polypeptide or a fragment or variant thereof influences signaling by the IL-22 receptor in the target cell;
(ii) the second binding site does not substantially influence signaling upon binding the IL-10 family address target; and
(iii) the IL-22 polypeptide or fragment or variant thereof does not substantially influence signaling by the IL-22 receptor in the absence of localization by the second binding site.
14. The macromolecule of claim 13, wherein the IL-22 polypeptide or fragment or variant thereof has been engineered to have reduced binding affinity for the IL-22 receptor.
15. The macromolecule of claim 13 or 14, wherein the IL-22 polypeptide or fragment or variant thereof comprises one or more amino acid substitution mutations at one or more residues that are predicted to be interaction surfaces between IL-22 and IL-22Ra and/or IL-10R2.
16. The macromolecule of claim 15, wherein the IL-22 polypeptide or fragment or variant thereof is a variant of a human IL-22 or fragment thereof and comprises:
(a) one or more of the amino acid substitution mutations of Table 6, Table 7A, Table 7B, Table 8, Table 9A, Table 9B, or Table 9C; and/or
(b) one or more of a K61 S, D71 N, D71 L, D71 G, R73Q, R73S, R73N, D168N, D168G, and R175S amino acid substitution mutation.
17. A method of modulating IL-22 receptor signaling in a target cell of a subject, the method comprising administering to the subject the macromolecule of any one of claims 13-16.
18. A method of treating a disease or disorder selected from Crohn's disease, ulcerative colitis, graft- vs-host disease, atopic dermatitis, psoriasis, wounds, diabetic skin ulcers, acute pancreatitis, chronic pancreatitis, autoimmune pancreatitis, nephritis, acute lung injury, viral lung injury, fibrosis, fibrosis, nonalcoholic steatohepatitis, viral hepatitis, and autoimmune hepatitis in a subject, the method comprising administering to the subject the macromolecule of any one of claims 13-16.
19. A macromolecule comprising:
(a) a first binding site comprising an IL-22 polypeptide or a fragment or variant thereof, and
(b) a second binding site comprising an anti-CDH17 antibody or antigen-binding fragment thereof, wherein the macromolecule is a fusion protein, and wherein the first binding site and the second binding site are connected by a linker.
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US202363597531P | 2023-11-09 | 2023-11-09 | |
| US63/597,531 | 2023-11-09 | ||
| US202463680971P | 2024-08-08 | 2024-08-08 | |
| US63/680,971 | 2024-08-08 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2025101842A1 true WO2025101842A1 (en) | 2025-05-15 |
Family
ID=95696467
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2024/055047 Pending WO2025101842A1 (en) | 2023-11-09 | 2024-11-08 | Compositions and methods for targeted delivery of therapeutic agents |
Country Status (2)
| Country | Link |
|---|---|
| TW (1) | TW202535945A (en) |
| WO (1) | WO2025101842A1 (en) |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20110091417A1 (en) * | 1999-12-07 | 2011-04-21 | Gurney Austin L | Interleukin-22 Polypeptides, Nucleic Acids Encoding The Same And Methods For The Treatment Of Pancreatic Disorders |
| US20190169250A1 (en) * | 2015-07-16 | 2019-06-06 | Philogen S.P.A. | Il22 immunoconjugates |
| US20230192794A1 (en) * | 2020-04-17 | 2023-06-22 | The Board Of Trustees Of The Leland Stanford Junior University | Engineered interleukin-22 polypeptides and uses thereof |
-
2024
- 2024-11-08 TW TW113143076A patent/TW202535945A/en unknown
- 2024-11-08 WO PCT/US2024/055047 patent/WO2025101842A1/en active Pending
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20110091417A1 (en) * | 1999-12-07 | 2011-04-21 | Gurney Austin L | Interleukin-22 Polypeptides, Nucleic Acids Encoding The Same And Methods For The Treatment Of Pancreatic Disorders |
| US20190169250A1 (en) * | 2015-07-16 | 2019-06-06 | Philogen S.P.A. | Il22 immunoconjugates |
| US20230192794A1 (en) * | 2020-04-17 | 2023-06-22 | The Board Of Trustees Of The Leland Stanford Junior University | Engineered interleukin-22 polypeptides and uses thereof |
Also Published As
| Publication number | Publication date |
|---|---|
| TW202535945A (en) | 2025-09-16 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| AU2021202453B2 (en) | Methods and compositions for delivering mrna coded antibodies | |
| US20220354968A1 (en) | Mrna therapy for the treatment of ocular diseases | |
| KR20250017231A (en) | Compositions and methods for modulating immune responses | |
| WO2022261136A1 (en) | Compositions and methods for targeted delivery of therapeutic agents | |
| WO2025101842A1 (en) | Compositions and methods for targeted delivery of therapeutic agents | |
| WO2024081930A1 (en) | Compositions and methods for targeted delivery of therapeutic agents | |
| WO2024129988A1 (en) | Compositions and methods for delivery of therapeutic agents to bone | |
| AU2024249959A1 (en) | Trem compositions and methods of use | |
| WO2023201237A1 (en) | Compositions and methods | |
| WO2024215772A2 (en) | Compositions and methods | |
| HK40064900A (en) | Methods and compositions for delivering mrna coded antibodies |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 24889644 Country of ref document: EP Kind code of ref document: A1 |