WO2025101478A1 - An automated and high-troughput analyzer method for a phosphorylated tau (p-tau) 217 immunoassay - Google Patents
An automated and high-troughput analyzer method for a phosphorylated tau (p-tau) 217 immunoassay Download PDFInfo
- Publication number
- WO2025101478A1 WO2025101478A1 PCT/US2024/054505 US2024054505W WO2025101478A1 WO 2025101478 A1 WO2025101478 A1 WO 2025101478A1 US 2024054505 W US2024054505 W US 2024054505W WO 2025101478 A1 WO2025101478 A1 WO 2025101478A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- reagent
- tau217
- alternatively
- sample
- vessel
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Classifications
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N35/00—Automatic analysis not limited to methods or materials provided for in any single one of groups G01N1/00 - G01N33/00; Handling materials therefor
- G01N35/10—Devices for transferring samples or any liquids to, in, or from, the analysis apparatus, e.g. suction devices, injection devices
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N33/00—Investigating or analysing materials by specific methods not covered by groups G01N1/00 - G01N31/00
- G01N33/48—Biological material, e.g. blood, urine; Haemocytometers
- G01N33/50—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing
- G01N33/68—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing involving proteins, peptides or amino acids
- G01N33/6893—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing involving proteins, peptides or amino acids related to diseases not provided for elsewhere
- G01N33/6896—Neurological disorders, e.g. Alzheimer's disease
Definitions
- Dementia is one of the costliest conditions to society. In 2022, the total national cost of caring for people living with Alzheimer’s Disease (AD) and other dementias was projected to reach $321 billion, and an estimated 6.5 million Americans aged 65 and older were estimated to be living with AD. Am JManag Care. 2022;28(suppl 10): S 188-S 196. By 2060, the number of people 65 and older with AD is projected to reach 13.8 million, barring the development of medical breakthroughs to prevent, slow or cure AD. Alzheimer ’s Association. 2022 Alzheimer’s Disease Facts and Figures. Alzheimers Dement 2022; 18.
- AD Dementia and AD are costly to society because affected individuals often require long-term care and support, and available treatments aim to manage symptoms and delay progression, rather than to reverse the underlying disease process. Accordingly, AD is impacting a growing population that is currently underserved with treatment options. Novel treatments for AD are emerging and driving an increased need for testing and diagnosis.
- p-tau217 Tau phosphorylated at threonine 217
- p-tau217 Tau phosphorylated at threonine 217
- a first aspect of the invention is a method for detecting phosphorylated tau (p-tau)217 in a biological sample using an immunoassay analyzer, the method comprising: aspirating, using a sample pipettor, a portion of the biological sample from a sample vessel and dispensing the aspirated biological sample into a reaction vessel of the immunoassay analyzer; wherein the immunoassay analyzer comprises: a reagent pack configured to hold a plurality of reagent vessels; a pipettor arrangement comprising at least one reagent pipettor and at least one sample pipettor; and a detector arrangement; aspirating, using the reagent pipettor, a portion of a first reagent from at least one reagent vessel and dispensing the aspirated reagent into the reaction vessel, generating a first reaction mixture; aspirating, using the reagent pipettor, a portion of a second reagent from at least one reagent vessel and dispensing
- a second aspect of the invention is a method for detecting phosphorylated tau (p-tau)217 in a biological sample using an immunoassay analyzer, the method comprising: aspirating, using a sample pipettor, a portion of the biological sample from a sample vessel and dispensing the aspirated sample into a reaction vessel of the immunoassay analyzer; wherein the immunoassay analyzer comprises: a reagent pack comprising a plurality of reagent vessels, wherein the reagent pack is configured to store a volume of reagent required for at least about 20 instances of a p-tau217 assay; a pipettor arrangement comprising at least one reagent pipettor and at a sample pipettor; and a detector arrangement; aspirating, using the reagent pipettor, a portion of a first reagent from at least one reagent vessel and dispensing the aspirated reagent into a reaction vessel of the immunoassay analyzer
- a third aspect of the invention is a method for detecting p-tau217 in a biological sample using an immunoassay analyzer, the method comprising: mixing the biological sample with a reagent comprising at least one affinity molecule that binds to at least one portion p-tau217, generating a first reaction mixture; mixing the first reaction mixture with a reagent comprising a detection molecule, generating a second reaction mixture; mixing the second reaction mixture with a substrate formulation, generating a detection mixture; wherein the immunoassay analyzer comprises an ultrasonic mixing module, and the first reaction mixture, second reaction mixture and/or detection mixture are agitated via the ultrasonic mixing module; detecting, using a detector arrangement, a reaction in the detection mixture; and determining a presence and/or concentration of p-tau217 in the biological sample based on the reaction detected in the detection mixture.
- the reaction in the detection mixture generates a chemiluminescent signal
- the method comprises correlating the chemiluminescent signal the presence and/or concentration of p-tau217 in the biological sample.
- the quantitation detection limit (LOQ) for measuring p-tau217 is equal to or less than about 3 pg/mL, alternatively equal to or less than about 2 pg/mL, equal to or less than about 1 pg/mL, equal to or less than about 0.6 pg/mL, and wherein the detected p-tau217 has a concentration above the LOQ.
- the detected p-tau217 has a concentration that is at least IX greater than the LOQ, alternatively at least 2X greater than the LOQ, or alternatively at least 3X greater than the LOQ.
- the detected p-tau217 exhibits a coefficient of variation (CV) of 20% or less, alternatively 15% or less, alternatively 10% or less, alternatively 5% or less, or alternatively 4% or less.
- CV coefficient of variation
- cycle time is about 45 seconds or less, alternatively about 40 seconds or less, alternatively about 35 seconds or less, alternatively about 30 seconds or less, alternatively about 25 seconds or less, alternatively about 20 seconds or less, or alternatively about 15 seconds.
- time to first result is about 60 minutes or less, alternatively about 55 minutes or less, alternatively about 50 minutes or less, alternatively about 45 minutes or less, alternatively about 40 minutes or less, alternatively about 35 minutes or less, alternatively about 30 minutes or less, alternatively about 25 minutes or less, alternatively about 20 minutes or less, alternatively about 15 minutes or less, or alternatively about 10 minutes or less.
- incubation time of the first reaction mixture is at least about 30 minutes, alternatively at least about 40 minutes, or alternatively at least about 50 minutes.
- incubation time of the second reaction mixture is at least about 5 minutes, alternatively at least about 8 minutes, or alternatively at least about 10 minutes.
- the first reagent comprises at least one affinity molecule configured to bind to at least one portion p-tau217
- the second reagent comprises at least one detection molecule
- the substrate formulation is configured to produce chemiluminescence.
- the substrate formulation is configured to produce chemiluminescence and comprises:
- A is Ci-ehaloalkyl, naphthyl, phenyl, substituted phenyl, or heteroaryl, wherein substituted phenyl comprises from 1 to 3 halo, Ci-6 alkyl, Ci-6 alkoxy, Ci-6 haloalkyl, C(O)Ri5, CN or NCh substituents;
- Ri is selected from the group consisting of Cs-uaryl, Ci-6 alkyl, Ci-6 haloalkyl, and C>. 14 aralkyl groups;
- R7-R14 are independently H, C1-6 alkoxy, halo, C1-4 alkyl, or R7 or R8-R9 or R9-R10 or R11-R12 or R12-R13 or R13-R14, can be joined together as a carbocyclic or heterocyclic ring system comprising at least one 5 or 6-membered ring;
- RI 5 is Ci-6 alkyl;
- each M is independently selected from the group consisting of H, an alkali metal, alkaline earth metal, transition metal, ammonium, phosphonium, organic amine salt, and an amino acid salt;
- Z is O or S
- n 0, 1, or 2;
- CAC cationic aromatic compound
- the affinity molecule is selected from the group consisting of an antibody, a monoclonal antibody, a polyclonal antibody, a synthetic antibody mimic, an aptamer, an affimer, DARPins, or oligonucleotides or peptides that bind to at least one epitope of p-tau217.
- the method further comprises exposing the biological sample to a first antibody which binds to a first p-tau217 epitope and a second antibody which binds to a second p-tau217 epitope.
- the detection molecule is an alkaline phosphatase (AP)-conjugated secondary antibody.
- the affinity molecule is a capture antibody and the detection molecule is a detection antibody wherein the method further comprises exposing the biological sample to the capture antibody which binds to a first p-tau217 epitope and the detection antibody which binds to a second p-tau217 epitope.
- the method further comprises exposing the biological sample to a second capture antibody which binds to a third p-tau217 epitope.
- the detection molecule binds to an epitope on p- Tau217 comprising amino acids 1 - 25, 1 - 20, 6 - 18, or 7 - 20 of SEQ ID NO: 1.
- the detection molecule binds to an epitope on p- Tau217 having at least 75% identical to the amino acid sequence set forth in SEQ ID NOs: 2, 3, or 4.
- the affinity molecule binds to an epitope on p- Tau217 comprising amino acids 217, 210 - 225, or 210 - 220 of SEQ ID NO: 1.
- the affinity molecule binds to an epitope on p- Tau217 having at least 75% identical to the amino acid sequence set forth in SEQ ID NO: 4.
- the affinity molecule and/or the detection molecule is conjugated to a magnetic bead or a magnetic particle.
- the first reaction mixture, second reaction mixture, and/or detection mixture is subjected to a magnetic field prior to detection.
- the detector arrangement comprises a light detector configured to sense photons emitted from assay reactions over a period of time, an analog circuit configured to provide an analog signal based on the photons emitted from the assay reactions over the period of time, and a counter circuit configured to provide a photon count based on the photons emitted from the assay reactions over the period of time.
- the immunoassay analyzer further comprises an ultrasonic mixing module.
- the first reaction mixture, second reaction mixture, and/or detection mixture is agitated via the ultrasonic mixing module.
- the immunoassay analyzer further comprises a washing arrangement, wherein the washing arrangement is configured to perform at least one wash action to wash away at least a portion of unreacted components, alternatively configured to perform at least two wash actions, alternatively configured to perform at least three wash actions, alternatively configured to perform at least four wash actions, or alternatively configured to perform at least five wash actions.
- the washing arrangement is configured to wash away at least a portion of the unreacted components in the first reaction mixture, second reaction mixture, and/or detection mixture.
- the immunoassay analyzer further comprises: a machine vision apparatus comprising an image capture device and an image interpretation device configured to monitor instrument and/or assay functionalities of the immunoassay analyzer.
- the instrument functionalities are selected from the group consisting of optical sensors, pressure sensors and thermistors.
- the assay functionalities are selected from the group consisting of sample volume monitoring, total reagent volume monitoring, residual volume monitoring, and particle retention monitoring.
- the biological sample is serum, whole blood, plasma, and/or cerebral spinal fluid.
- the method further comprises detecting at least one additional biomarker is selected from the group consisting of A
- the reagent vessels comprise an elastomeric selfsealing membrane.
- the reagent pack further comprises containment walls arranged between the reagent vessels.
- the immunoassay analyzer further comprises a reagent storage unit, wherein the reagent pack is housed in the reagent storage unit.
- a fourth aspect of the invention is a method for quantitatively determining an amount of phosphorylated tau (p-tau)217 in at least one plasma sample using a high-throughput analyzer, the method comprising: aspirating a portion of the at least one plasma sample from a sample vessel and dispensing the aspirated plasma sample into a first reaction vessel and second reaction vessel of an immunoassay analyzer, wherein the immunoassay analyzer further comprises: a pipettor arrangement comprising at least a first reagent pipettor, a second reagent pipettor, a third reagent pipettor and at least one sample pipettor; a plurality of reagent vessels; and a detector arrangement; aspirating, using the first reagent pipettor, a portion of a fluidic substance from a first reagent vessel and dispensing said fluidic substance into the first reaction vessel; aspirating, using the first reagent pipettor, a portion of a fluidic substance from
- the method is configured to analyze at least about 200 plasma samples/hr.
- the method is configured to analyze at least about 300 plasma samples/hr.
- the method is configured to analyze at least about 400 plasma samples/hr.
- the reaction in the detection mixture generates a chemiluminescent signal, wherein the chemiluminescent signal quantitatively corresponds to a concentration of p-tau217 in the plasma sample.
- the quantitation detection limit (LOQ) for measuring p-tau217 is equal to or less than about 3 pg/mL, alternatively equal to or less than about 2 pg/mL, equal to or less than about 1 pg/mL, equal to or less than about 0.6 pg/mL, and wherein the p-tau217 has a concentration above the LOQ.
- the quantitation detection limit (LOQ) for measuring p-tau217 is equal to or less than about 2 pg/mL.
- the quantitation detection limit (LOQ) for measuring p-tau217 is equal to or less than about 1 pg/mL.
- the quantitation detection limit (LOQ) for measuring p-tau217 is equal to or less than about 0.6 pg/mL, and wherein the p- tau217 has a concentration above the LOQ.
- the quantitation detection limit (LOQ) for measuring p-tau217 is equal to or less than about 50 fg/mL.
- the quantitation detection limit (LOQ) for measuring p-tau217 is equal to or less than about 15 fg/mL.
- the quantitation detection limit (LOQ) for measuring p-tau217 is equal to or less than about 10 fg/mL, and wherein the p-tau217 has a concentration above the LOQ.
- the detected p-tau217 has a concentration that is at least IX greater than the LOQ, alternatively at least 2X greater than the LOQ, or alternatively at least 3X greater than the LOQ.
- the p-tau217 exhibits a coefficient of variation (CV) of 20% or less.
- the p-tau217 exhibits a coefficient of variation (CV) of 4% or less.
- the immunoassay analyzer comprises a fourth reaction vessel and the pipettor arrangement comprises a fourth reagent pipettor, wherein the method further comprises aspirating a portion of the at least one plasma sample from a sample vessel and dispensing the aspirated plasma sample into the fourth reaction vessel; aspirating, using the fourth reagent pipettor, a portion of a fluidic substance from a first reagent vessel and dispensing said fluidic substance into the fourth reaction vessel; aspirating, using the fourth reagent pipettor, a portion of a fluidic substance from a second reagent vessel and dispensing said fluidic substance into the fourth reaction vessel; dispensing a substrate into the fourth reaction vessel, generating a detection mixture; detecting, using the detector arrangement, a presence of a reaction in the detection mixture in the fourth reaction vessel; and quantitatively determining an amount of p-tau217 in the plasma based on the presence of the reaction in the detection mixture.
- the first reagent pipettor, the second reagent pipettor, the third reagent pipettor and the fourth reagent pipettor are selectively and/or simultaneously operated.
- the fourth reagent pipettor is configured to engage a dispense tip prior to aspiration.
- cycle time is about 45 seconds or less, alternatively about 40 seconds or less, alternatively about 35 seconds or less, alternatively about 30 seconds or less, alternatively about 25 seconds or less, alternatively about 20 seconds or less, or alternatively about 15 seconds.
- time to first result is about 45 minutes or less, alternatively about 40 minutes or less, alternatively about 35 minutes or less, alternatively about 30 minutes or less, alternatively about 25 minutes or less, alternatively about 20 minutes or less, alternatively about 15 minutes or less, or alternatively about 10 minutes or less.
- dispensing the fluidic substance from the first reagent vessel into the first reaction vessel, second reaction vessel, third reaction vessel, and/or fourth reaction vessel generates a first reaction mixture and incubation time of the first reaction mixture is at least about 30 minutes, alternatively at least about 40 minutes, or alternatively at least about 50 minutes.
- the first reaction mixture in the first reaction vessel, second reaction vessel, third reaction vessel, and/or fourth reaction vessel are incubated simultaneously.
- dispensing the fluidic substance from the second reagent vessel into the first reaction vessel, second reaction vessel, third reaction vessel, and/or fourth reaction vessel generates a second reaction mixture and incubation time of the second reaction mixture is at least about 5 minutes, alternatively at least about 8 minutes, or alternatively at least about 10 minutes.
- the second reaction mixture in the first reaction vessel, second reaction vessel, third reaction vessel, and/or fourth reaction vessel are incubated simultaneously.
- the method is configured to simultaneously perform at least two assays for a plurality of plasma samples, alternatively at least three assays for a plurality of plasma samples, or at least four assays for a plurality of plasma samples.
- the plasma samples are from the same individual or from multiple individuals.
- the method comprises a plurality of sample vessels.
- At least one of the assays is p- tau217 assay, alternatively at least two of the assays are p-tau217 assays, alternatively at least three of the assays are p-tau217 assays, or alternatively at least four of the assays are p-tau217 assays.
- At least one of the assays is p- tau217 assay and at least one of the assays is a second analyte assay, wherein the second analyte is selected from the group consisting of A
- the second analyte is selected from the group consisting of A
- the fluidic substance from the second reagent vessel comprises at least one detection molecule, preferably the detection molecule is an alkaline phosphatase (AP)-conjugated secondary antibody.
- the detection molecule is an alkaline phosphatase (AP)-conjugated secondary antibody.
- the affinity molecule is selected from the group consisting of an antibody, a monoclonal antibody, a polyclonal antibody, a synthetic antibody mimic, an aptamer, an affimer, DARPins, or oligonucleotides or peptides that bind to at least one epitope of p-tau217.
- the affinity molecule is an antibody and the method further comprises exposing the plasma sample to a first antibody which binds to a first p-tau217 epitope and a second antibody which binds to a second p-tau217 epitope.
- the detection molecule is an alkaline phosphatase (AP)-conjugated secondary antibody.
- the affinity molecule and/or the detection molecule is conjugated to a magnetic bead or a magnetic particle.
- the first reagent vessel, second reagent vessel and/or third reagent vessel are housed in a reagent pack.
- the reagent pack further comprises containment walls arranged between the reagent vessels.
- the reagent vessels comprise an elastomeric self-sealing membrane.
- the immunoassay analyzer further comprises a reagent storage unit, wherein the reagent pack is housed in the reagent storage unit.
- each reagent vessel is configured to store a volume of reagent required for at least about 20 instances of a p-tau217 assay.
- the immunoassay analyzer further comprises: a machine vision apparatus comprising an image capture device and an image interpretation device configured to monitor instrument and/or assay functionalities of the immunoassay analyzer.
- the instrument functionalities are selected from the group consisting of optical sensors, pressure sensors and thermistors.
- the assay functionalities are selected from the group consisting of sample volume monitoring, total reagent volume monitoring, residual volume monitoring, and particle retention monitoring.
- the immunoassay analyzer further comprises an ultrasonic mixing module.
- the first reaction vessel, second reaction vessel, third reaction vessel, and/or fourth reaction vessel are agitated via the ultrasonic mixing module.
- the immunoassay analyzer further comprises a washing arrangement, wherein the washing arrangement is configured to perform at least one wash action to wash away at least a portion of unreacted components, alternatively configured to perform at least two wash actions, alternatively configured to perform at least three wash actions, alternatively configured to perform at least four wash actions, or alternatively configured to perform at least five wash actions.
- FIG. 1 is a table showing the performance characteristics an exemplary immunoassay analyzer disclosed herein.
- FIGs. 2A - 2C depict a method according to an aspect of the disclosure.
- FIG. 3 is a calibration curve plot constructed using calibrator antigens of varying concentrations using an exemplary p-tau217 assay and an exemplary immunoassay analyzer as disclosed herein.
- FIG. 4 is a box and whisker plot showing pTau217 concentrations associated with AD- positive versus normal samples analyzed using an exemplary p-tau217 assay and an exemplary immunoassay analyzer as disclosed herein.
- FIG. 5 is a plot depicting within-run CV was calculated from harmonized dose response and plotted relative to mean dose-response of samples analyzed using an exemplary p-tau217 assay and an exemplary immunoassay analyzer as disclosed herein.
- FIG. 6 is a graph depicting within-run precision profiles for samples analyzed using an exemplary p-tau217 assay, experimental p-tau217 antibody pairs, and an exemplary immunoassay analyzer as disclosed herein.
- FIG. 7 is a graph depicting a comparison of the measured concentrations for Alzheimer’s Disease samples and normal/healthy cohort for samples analyzed using an exemplary p-tau217 assay, experimental p-tau217 antibody pairs, and an exemplary immunoassay analyzer as disclosed herein.
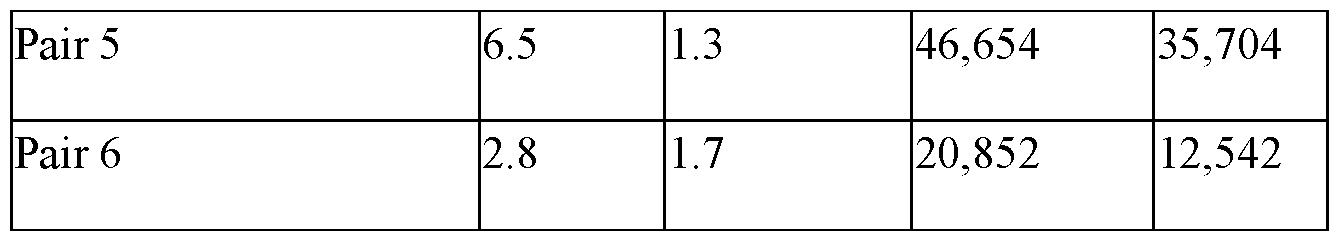
- FIGs. 8A-8C are graphs depicting precision profiles for an experimental p-tau217 antibody pair and conventional antibody pairs analyzed using an exemplary p-tau217 assay, and an exemplary immunoassay analyzer as disclosed herein.
- FIG. 8A depicts %CV for the analyzed antibody pairs.
- FIG. 8B depicts within-run precision profiles for the analyzed antibody pairs.
- FIG. 8C depicts the measured concentrations for the analyzed antibody pairs for AD Patients and Normal patients.
- FIG. 9 is a graph depicting a comparison of experimental p-tau217 antibody pairs and conventional p-tau217 antibody pairs using an exemplary p-tau217 assay and an exemplary immunoassay analyzer as disclosed herein.
- FIG. 10 is a graph depicting discrimination of amyloid positive patient samples and negative patient samples analyzed using an exemplary p-Tau217 assay as disclosed herein. DETAILED DESCRIPTION
- Tau is a microtubule-associated protein abundant in the axons of neurons and is thought to contribute to stabilization and formation of microtubules. Tau has six isoforms with different amino-terminal inserts and different numbers of tandem repeats near the carboxy terminus, ranging in length from 352 to 441 amino acids, and is thought to have over thirty different phosphorylation sites. The amino acid sequence of tau-441 is listed in Table 1.
- AD Alzheimer’s disease
- Phosphorylated tau has been proposed as providing a more accurate identification of AD due to the presence of hyperphosphorylated tau in neurofibrillary tangles.
- p-tau21-7 Tau phosphorylated at threonine 217 (p-tau217) has been shown to have diagnostic value in predicting Alzheimer’s Disease.
- pTau 217 is a variant of any tau isoform in which the threonine residue 217 is phosphorylated.
- the bolded Thr in Table 1 is the phosphorylation site in p-tau217 in tau-441.
- Plasma levels of p-tau217 have been found to be clinically significant in an early, specific and accurate diagnosis of AD, including the manifestation and progression of AD. Telser, J et al Clin Chim Acta. 2022 Jun 1 :531 : 100-111.
- Automated analyzers are commonly used in clinical chemistry, immunoassay, hematology, and other biological sampling and analyzing applications.
- Automated analytical equipment such as automated analytical chemistry instruments, automated analytical immunoassay instruments, automated analytical hematology instruments, etc., can efficiently perform clinical analysis on a large number of samples, with multiple tests being run concurrently or within short time intervals.
- Automated analytical instruments are particularly well-suited for high-volume and mid-volume testing environments.
- the present invention fulfills a need in the art by providing methods for detecting phosphorylated tau (p- tau)217 in a biological sample using an immunoassay analyzer.
- the steps may be conducted in any feasible order. And, as appropriate, any combination of two or more steps may be conducted simultaneously.
- x, y, and/or z means any element of the seven-element set ⁇ (x), (y), (z), (x, y), (x, z), (y, z), (x, y, z) ⁇ .
- x, y and/or z means "one or more of x, y and z”.
- endpoints include all numbers subsumed within that range (e.g., 1 to 5 includes 1, 1.5, 2, 2.75, 3, 3.80, 4, 5, etc.).
- values that are expressed as ranges can assume any specific value or subrange within the stated ranges in different embodiments of the disclosure, to the tenth of the unit of the lower limit of the range, unless the context clearly dictates otherwise.
- “up to” a number includes the number (for example, 50).
- the term “in the range” or “within a range” includes the endpoints of the stated range.
- exemplary means serving as a non-limiting example, instance, or illustration.
- terms "e.g.,” and “for example” set off lists of one or more non-limiting aspects, examples, instances, or illustrations.
- the term “substantially” refers to the qualitative condition of exhibiting total or near-total extent or degree of a characteristic or property of interest. Biological and chemical phenomena rarely, if ever, go to completion and/or proceed to completeness or achieve or avoid an absolute result. The term “substantially” is therefore used herein to capture the potential lack of completeness inherent in many biological and chemical phenomena. For example, “substantially” may refer to being within at least about 20%, alternatively at least about 10%, alternatively at least about 5% of a characteristic or property of interest.
- One aspect of the invention is a method for detecting phosphorylated tau (p-tau)217 in a biological sample using an immunoassay analyzer.
- Another aspect of the invention is a method for quantitatively determining the amount of phosphorylated tau (p-tau)217 in at least one plasma sample using an immunoassay analyzer
- immunoassay may be a laboratory method used to determine the amount of an analyte in a sample. It can be based on the interaction of antibodies with antigens, and because of the degree of selectivity for the analyte (either antigen or antibody), an immunoassay can be used to quantitatively determine very low concentrations of analyte in a test sample.
- An "immunoassay analyzer” can include an instrument on which immunoassays have been automated. Various immunoassay analyzers are commercially available including the Dxl® system (Beckman Coulter, CA).
- an immunoassay analyzer disclosed herein includes the following basic structural and functional modules: a sample presentation unit, an analytic unit, an incubator station, a washing station, a read station, and reagent storage.
- the immunoassay analyzer may include a pipettor arrangement with at least one sample pipettor and at least one reagent pipettor, and at least one transport device.
- the transport device includes mechanisms, such as pick-and-place grippers, which are used to transport sample and reaction vessels among the various modules of the immunoassay analyzer.
- the immunoassay analyzer may include a container carriage device which is configured to hold and carry the containers at various locations in the instrument so that the analytic unit, incubator station, wash station, and read station can use the containers in various manners.
- container carriage devices include vessel racks (e.g., a sample rack, a reagent rack, and a diluent rack), the sample presentation unit, vessel carriage units (e.g., a sample carriage unit, a reaction vessel carriage unit, and a reagent carriage unit), vessel transfer units (e.g., a sample transfer unit, a reagent transfer unit, an incubator transfer unit, and an reaction vessel transfer unit), and vessel holding plates or wheels (e.g., a sample wheel, an incubator, and a wash wheel) ), which are described herein.
- vessel racks e.g., a sample rack, a reagent rack, and a diluent rack
- the sample presentation unit e.g., a sample carriage unit, a reaction vessel carriage unit,
- the read station may include a detector arrangement.
- the detector arrangement may include a detector that is configured to detect light or luminescence, for example chemiluminescence.
- the detector may be a luminescence detector, a chemiluminescence detector, a luminometer, or a photomultiplier-based detection instrument.
- the detector includes a light detector configured to sense photons emitted from assay reactions over a period of time, an analog circuit configured to provide an analog signal based on the photons emitted from the assay reactions over the period of time, and a counter circuit configured to provide a photon count based on the photons emitted from the assay reactions over the period of time.
- U.S. Patent No. 11,604,146 which is incorporated by reference in its entirety herein, discloses non-limiting examples of a detector that may be used in an aspect of the invention.
- primary sample containers can be placed into an onload section (e.g., individually or on racks) of the sample presentation unit.
- the sample presentation unit has at least one sample rack, alternatively at least two sample racks, alternatively at least three sample racks, alternatively at least four sample racks, alternatively at least five sample racks, alternatively at least six sample racks, alternatively at least seven sample racks, alternatively at least eight sample racks, alternatively at least nine sample racks, alternatively at least ten sample racks, alternatively at least 11 sample racks, alternatively at least 12 sample racks, alternatively at least 13 sample racks, alternatively at least 14 sample racks, alternatively at least 15 sample racks, alternatively at least 16 sample racks, alternatively at least 17 sample racks, alternatively at least 18 sample racks, alternatively at least 19 sample racks, or alternatively at least 20 sample racks.
- each sample rack can hold at least one sample vessel, alternatively at least two sample vessels, alternatively at least three sample vessels, alternatively at least four sample vessels, alternatively at least five sample vessels, alternatively at least six sample vessels, or alternatively at least seven sample vessels.
- the sample presentation unit holds about 140 sample vessels. In a specific embodiment, the sample presentation unit has 20 sample racks with each rack holding seven sample vessels.
- the sample vessels include a barcode label that uniquely identifies the sample vessel in the immunoassay analyzer.
- the barcode label also may include alphanumeric characters that correspond to the barcode identification information.
- the immunoassay analyzer may include at least optical reader, such as a barcode scanner.
- the optical readers are area scan cameras that provide a two-dimensional image of the barcode and/or sample vessel. In another embodiment, optical readers are area scan cameras that provide a three-dimensional image of the barcode and/or sample vessel.
- the sample containers may be moved into a presentation section of the sample presentation unit.
- the sample presentation unit is maintained at between about 4.5°C to 14°C.
- the transfer station may receive primary sample containers from the sample presentation unit from a transport device.
- Primary sample containers delivered to the transfer station from the transport device may be initially processed in different ways. For example, the primary sample container may temporarily remain at the transfer station while the sample provided within is aspirated by the sample pipettor at the transfer station. Following such aspiration, the primary sample container may be expelled from the automated analyzer at the offload station of the sample presentation unit, or may be passed on to the transport device for further processing.
- the instrument may also contain a sample retention unit configured to receive sample retention vessels and store, analyze, or otherwise process samples retained within the sample retention vessels.
- a “primary sample container” loaded into the automated analyzer with a sample may also be considered a “sample retention vessel” when placed in a sample retention unit, such as a storage unit.
- sample retention vessel such as a storage unit.
- containers or “vessels” are analogous, and can be of various types, such as specimen tubes (also referred to herein as sample tubes) and pipettor tips, such as micro or disposable tips.
- the vessels are tubes with diameters between about 12mm to about 16mm and/or heights between about 75mm to about 100mm.
- the vessels are cups with volumes of about 0.5mL, alternatively about l.OmL, alternatively about 1.5mL, alternatively about 2.0mL, alternatively about 2.5mL, or alternatively about 3.0mL.
- the analytic unit is configured to receive and analyze samples.
- the analytic unit configured to perform an immunoassay.
- the analytic unit includes the pipettor arrangement.
- the pipettor arrangement may be configured to aliquot, aspirate, and dispense fluidic substances into various vessels, including, but not limited to sample vessels, diluent vessels, reagent vessels, and reaction vessels.
- Fluidic substances are substances that have fluidic characteristics.
- the fluidic substance is a single fluidic substance.
- the fluidic substance is a mixture of a plurality of substances.
- the pipettor arrangement may contain at least one, two, three, or four reagent pipettors used to mix reagents with sample aliquots for an assay.
- the reagent pipettors may be arranged as dual reagent pipetting stations and are independent to each other, each having its own fluid pumps and valves, wash towers, reaction vessel carriages, and pipettor.
- a sample aliquot may be transferred from a sample retention vessel into a reaction vessel using the sample pipettor in order to mix the sample aliquot with one or more reagents.
- the at least one reagent pipettor and at least one sample pipettor are configured to aspirate and/or dispense less than about 10 pL.
- the at least one reagent pipettor and at least one sample pipettor are configured to aspirate and/or dispense less than about 9.9 pL, alternatively less than about 9.5 pL, alternatively less than about 8.0 pL, alternatively less than about 7.0 pL, alternatively less than about 6.0 pL, alternatively less than about 5.0 pL, alternatively less than about 4.0 pL, alternatively less than about 3.0 pL, alternatively less than about 2.0 pL, alternatively between than about 9.9 pL and 2.0 pL.
- the immunoassay analyzer includes a reagent pack configured to hold a plurality of reagent vessels.
- a “reagent pack” may include any suitable container that can store a reagent.
- An example of a reagent pack can include a generally rectangular elongated body formed to include multiple reagent vessels including one or more large reagent vessels, and one or more relatively smaller reagent vessels, as well as features to facilitate handling and automation.
- US Patent No. 9,519,000 which is incorporated by reference in its entirety herein, discloses non-limiting examples of a reagent pack that may be used in an aspect of the invention.
- the reagent pack may be configured to accommodate sufficient volumes of reagents for multiple instances of an assay.
- each reagent pack includes reagents for about 20 to about 100 instances of an assay and in some cases about 50 instances.
- a reagent pack may supplied with empty or partially filled reagent vessels, to which reagents are subsequently transferred from bulk containers, such as bottles.
- Individual reagent vessels may differ in dimension to accommodate the requirements of an assay type. Factors that can determine the size of a reagent vessel include the number of uses desired for the reagent pack type, concentration-dependent stability issues with reagent components, and the need to minimize the volume of the final reaction mixture.
- each reagent pack can include a large reagent vessel and a plurality of small reagent vessels.
- Each reagent vessel may be large enough to accommodate a microtip or disposable tip (i.e., dispo-tip) of a reagent pipettor used to remove a volume of reagent for use in an assay.
- the reagent pack may be maintained at a temperature of between about 4°C to 10°C.
- a “reagent vessel” may refer to a vessel, unit, fluid container, or the like that is configured to store reagents.
- the reagent vessels include an elastomeric selfsealing membrane.
- An elastomeric self-sealing membrane may be a polymer, such as polypropylene, which is able to regain its original shape when pierced.
- the elastomeric membrane can be a thermoplastic elastomer with hardness of 30-40 durometer (Shore) A. In other embodiments, the hardness can be 20-50 (Shore) A, or about 30 (Shore) A. Elastomers deform sufficiently to form a tight seal with the vessel base. Thermoplastic elastomers are advantageous because of their compatibility with plastics injection molding processes.
- the elastomeric membrane can be large enough to provide adequate compression without bottoming on the sealing portion of the vessel.
- the hardness and dimensions can cooperate to allow the elastomeric membrane to the sealing portion with reasonable sealing force.
- the elastomeric membrane diameter is small enough so that, when compressed by engagement of the pipettor tip, it conforms to the sealing portion without contacting the wall of the pipettor tip. This advantageously concentrates sealing force to the sealing portion of vessel and distributes sealing force evenly to prevent leaks.
- the sealing force is about 44 newtons (about 9.9 lbs) and produces a pressure on the sealing surface of about 300 (about 43.5 pounds per square inch) to about 1000 kPa (145.0 pounds per square inch).
- the elastomeric self-sealing membrane allows for aspiration of the reagents without concern of evaporation.
- the reagent pack further includes containment walls arranged between the reagent vessels.
- the reagent pack may include at least one reagent vessel, alternatively at least two reagent vessels, alternatively at least three reagent vessels, alternatively at least four reagent vessels, alternatively at least five reagent vessels, or alternatively at least ten reagent vessels.
- the immunoassay analyzer further includes a reagent storage unit, wherein the reagent pack is housed in the reagent storage unit.
- a reagent pipettor may be outfitted with a tip that allows it to perform ultrasonic mixing of a reagent in a reagent pack before aspirating it for transport to a reaction vessel, thereby ensuring that the aspirated reagent would not be impacted by any settling that may have taken place in the reagent pack.
- Sample pipettors may similarly be specialized.
- the transfer unit transfers the reaction vessels to and from the incubator station which includes an incubator. In some embodiments, the transfer unit transfers one or more of the pipetted reaction vessels from the reagent carriage unit to the incubator. Further, the transfer unit can transfer one or more reaction vessels from the incubator to the reagent carriage unit. The transfer unit can also remove from the reaction vessels that have been read or completed the incubator.
- the incubator is thermally controlled to maintain a predetermined temperature. In some embodiments, the incubator is maintained about 30 °C to 40 °C. In other embodiments, the incubator is maintained about 37 °C to ensure immunological reaction and enzyme reaction, for example. By way of example, the incubator performs assay incubation.
- the transfer unit transfers incubated reaction vessels from the incubator to the wash unit, transfers assay reaction vessels from the wash unit to the incubator, transfers reaction vessels containing substrate from the wash unit to the incubator for substrate incubation or enzyme reaction, transfers washed reaction vessels from the incubator to detector arrangement after substrate incubation, and transfers the reaction vessels that have been read or completed from the detector arrangement to the incubator.
- the used reaction vessels can be delivered to a waste location.
- a sample and a reagent are dispensed into a reaction vessel and mixed.
- the mixture is then transferred to the incubator.
- the sample and the reagent interact.
- the resulting “first reaction mixture” is a result of the incubation between the sample and the reagent.
- the reagent may include a specific-binding reagent, such as an affinity molecule specific to the analyte being analyzed by the immunoassay analyzer.
- the incubation time of the first reaction mixture is at least about 30 minutes, alternatively at least about 40 minutes, alternatively at least about 50 minutes, alternatively at least about 55 minutes, or alternatively at least about 60 minutes.
- a reagent which is different from the reagent used in the first reaction mixture, is added to the first reaction mixture and mixed. The mixture is then transferred to the incubator. During incubation, the first reaction mixture and the reagent interact. The resulting “second reaction mixture” is a result of an incubation between the first reaction mixture and the reagent.
- the reagent may include a detection molecule, such as an alkaline phosphatase (AP)- conjugated secondary antibody, or a labeled antibody.
- the incubation time of the second reaction mixture is at least about 5 minutes, alternatively at least about 8 minutes, or alternatively at least about 10 minutes.
- the wash station receives and supports reaction vessels thereon such that various aspects of diagnostic process are performed with the immunoassay analyzer.
- the wash station is configured to wash away at least some of the unreacted components. Unreacted components may include unreacted reagents (e.g., free antigens, antibodies, unbound reactants, particles, and/or fluid, etc.) and unreacted sample.
- the wash station may be configured to perform a set number of wash actions depending on the assay.
- the wash station may also be configured to perform a set number of washes within a predetermined sequence.
- the wash station is configured to perform at least one wash action to wash away at least a portion of unreacted components, alternatively configured to perform at least two wash actions, alternatively configured to perform at least three wash actions, alternatively configured to perform at least four wash actions, or alternatively configured to perform at least five wash actions.
- the wash station is a thermally controlled device to separate bound or free analytes from particles after incubation.
- the wash unit is maintained about 30 °C to 40 °C. In other embodiments, the wash unit is maintained about 37 °C to ensure enzyme reaction, for example.
- U.S. Patent Publication No. 2022/0357352 which is incorporated by reference in its entirety herein, discloses configurable wash processes according to an aspect of the invention.
- the wash station may comprise a washing arrangement which may be configured to provide a base number of wash series (or wash actions) for each reaction vessel and optionally provide an additional number(s) of wash actions.
- the additional number(s) of wash actions may include one, a plurality, or all of a potential number of wash actions.
- An additional number of wash actions beyond the base number of wash actions may be specified for certain assays in an assay protocol file.
- the washing arrangement may include cleaning dispense nozzle (or probe) which dispenses a rinsing fluid and a cleaning aspiration nozzle (or probe) which aspirates the unreacted components.
- a base number of wash actions performed may be three, and the additional number of wash action(s) may be one or two.
- the base number of wash actions may be performed if three probes dispense buffer solution once per vessel and the three probes aspirate the at least some of the unreacted components some of the buffer solution, and/or the at least some of the unreacted reagents once per vessel.
- certain probe(s) may be selectively used to dispense clean buffer solution into the vessel and aspirate the at least some of the unreacted components of the sample, some of the buffer solution, and/or the at least some of the unreacted reagents from the vessel to perform the additional wash action(s).
- the affinity molecule and/or the detection molecule is conjugated to a magnetic bead or a magnetic particle.
- Magnetic beads also known as magnetic particles, paramagnetic particles, or superparamagnetic particles
- Magnetic beads consist of a polystyrene core surrounded by a thin layer of small iron oxide particles (-20-30 nm), such as magnetite.
- the magnetic beads are encapsulated by, for example, a polymer, protein A, protein G, protein L, a secondary antibody, or an epoxy. Surface modification of the coating minimizes any non-specific protein binding.
- Antibodies targeting the analyte of interest can be covalently coupled to the surface of the magnetic bead. In some embodiments, there is about 2 pg or antibody per mg of magnetic bead. In some embodiments, there is about 3 pg or antibody per mg of magnetic bead.
- the first reaction mixture or second reaction mixture is subjected to a magnetic field.
- the magnetic beads do not exhibit bead-to-bead attraction, only migrating when a magnetic field is applies. Captured analytes or targets are separated from the mixture and magnetization may be used to retain desired components within a reaction vessel.
- the vessel containing the first reaction mixture is moved near one or more magnets.
- the one or more magnets attract the magnetic bead(s) to one or more sides of the reaction vessel.
- the reaction vessel is then subject to a wash process in which a cleaning dispense nozzle dispenses a rinsing fluid and a cleaning aspiration nozzle aspirates the unreacted components.
- the aspiration nozzle may be washed with a probe washer before and/or after the aspirating.
- the reaction vessel may undergo a series of wash process which may include at least two series of dispensing the rinsing fluid and aspirating the uncollected fluid components, alternatively at least three series, alternatively at least four series, alternatively at least five series.
- an unreacted substance or substances in the vessel is removed (e.g., rinsed away) by the bound-free cleaning aspiration nozzle.
- the vessel containing the second reaction mixture is moved near one or more magnets.
- the one or more magnets attract the magnetic bead(s) or magnetic particle(s) to one or more sides of the reaction vessel.
- the reaction vessel is then subject to a wash process in which a cleaning dispense nozzle dispenses a rinsing fluid and a cleaning aspiration nozzle aspirates the unreacted components.
- the aspiration nozzle may be washed with a probe washer before and/or after the aspirating.
- the reaction vessel may undergo a series of wash process which may include at least two series of dispensing the rinsing fluid and aspirating the uncollected fluid components, alternatively at least three series, alternatively at least four series, alternatively at least five series.
- a series of wash process which may include at least two series of dispensing the rinsing fluid and aspirating the uncollected fluid components, alternatively at least three series, alternatively at least four series, alternatively at least five series.
- a substrate is dispensed into the second reaction mixture and mixed. After a certain reaction time necessary for the substrate and the second reaction mixture to interact.
- the reaction vessel is then subject to a wash process in which a cleaning dispense nozzle dispenses a rinsing fluid and a cleaning aspiration nozzle aspirates the unreacted components.
- the aspiration nozzle may be washed with a probe washer before and/or after the aspirating.
- the reaction vessel may undergo a series of wash process which may include at least two series of dispensing the rinsing fluid and aspirating the uncollected fluid components, alternatively at least three series, alternatively at least four series, alternatively at least five series.
- an unreacted substance or substances in the vessel is removed (e.g., rinsed away) by the bound- free cleaning aspiration nozzle.
- the resulting detection mixture is transferred to the detector arrangement.
- Assays using substrates including features and/or characteristics described herein may benefit from one or more additional number(s) of wash actions.
- These “two-site” or “sandwich” immunoassays employ a first antibody or antibody fragment, which is described as the “capture” antibody, is bound to a solid support, such as magnetic beads or particles disclosed herein, using procedures known in the art.
- a second antibody or antibody fragment which is described as the “detection” antibody, is coupled or conjugated with a label, such as the enzymes disclosed herein, using procedures known in the art.
- the label produces a detectable signal when provided with substrate(s), so that the amount of signal measured corresponds to the amount of detection antibody that is bound to the analyte.
- the substrate formulation is configured to produce chemiluminescence. These substrates can produce light and thereby provide detection corresponding to a quantity of analytes captured.
- chemiluminescent compound refers to a compound that produces chemiluminescence in the presence of a phosphatase enzyme and oxygen under appropriate conditions as provided herein.
- the substrate formulation includes a chemiluminescent compound of formula I or a salt thereof:
- A is Ci-6haloalkyl, naphthyl, phenyl, substituted phenyl, or heteroaryl, wherein substituted phenyl comprises from 1 to 3 halo, Ci-6 alkyl, Ci-6 alkoxy, Ci-6 haloalkyl, C(0)Ri5, CN or NO2 substituents;
- Ri is selected from the group consisting of Cs-naryl, Ci-6 alkyl, Ci -6 haloalkyl, and C5- 14 aralkyl groups;
- R7-R14 are independently H, Ci-6 alkoxy, halo, Ci-4alkyl, or R7 -Rs or Rs -R9 or R9-
- R10 R11-R12 or R12-R13 or R13-R14 can be joined together as a carbocyclic or heterocyclic ring system comprising at least one 5 or 6-membered ring;
- Ri5 is Ci-6 alkyl
- each M is independently selected from H, or an alkali metal, alkaline earth metal, transition metal, ammonium, phosphonium, organic amine salt, amino acid salt or ;
- Z is O or S
- n 0, 1, or 2;
- CAC cationic aromatic compound
- Chemiluminescent compounds useful in the present formulations are capable of generating chemiluminescence when contacted with an alkaline phosphatase. Such compounds can be synthesized as described in U.S. Pat. Nos. 6,45,727, 6,90,571, 6,139,782, 6,218,137, 6,270,695, 6,296,787, and 10,703,971 each of which is incorporated by reference herein.
- the chemiluminescent substrate is added to the vessel with the second reaction mixture and light generated by the reaction is measured with a luminometer.
- the light production is inversely proportional to the concentration of free analyte in the sample.
- the amount of analyte in the sample is then determined from a stored, multi-point calibration curve.
- the detector can generate an output signal that can be processed to generate a relative light unit (“RLU”) value (i.e., an output response) indicating a result of the assay.
- RLU relative light unit
- a larger RLU value indicates more light, which indicates a larger amount of the analyte in the biological sample than a smaller RLU value indicates.
- the reaction is performed at a temperature between 5° C and 50° C, preferably between 20° C and 40° C in an aqueous buffer solution at a pH between 7 and 12, 8 and 11, or preferably between 8.5 and 10.
- the enzyme is preferably an alkaline phosphatase or an alkaline phosphatase conjugate.
- the substrate formulation comprises 0.01 mM-50 mM compound I, 0.01- 200 uM cationic aromatic compound, 1 uM-10 mM background reducing agent, 0.05-20 g/L ether-linked non-ionic surfactant or hydrophilic polymer, 0.01-10 g/L anionic surfactant, and an amine buffer at from 0.025M to 0.65M and at pH 7-12.
- the substrate formulation comprises 0.05 mM-10 mM compound I, 0.05- 50 pM cationic aromatic compound, 10 uM-1000 uM background reducing agent, 0.1 to 10 g/L ether-linked non-ionic surfactant or hydrophilic polymer, 0.1 to 5 g/L anionic surfactant, and an amine buffer at from 0.05M to 0.5M and at pH 8-11.
- the substrate formulation comprises 0.1 mM-5 mM compound I, 0.1-25 pM cationic aromatic compound, 50 to 500 uM background reducing agent, 0.2 to 5 g/L ether- linked non-ionic surfactant or hydrophilic polymer, 0.1 to 5 g/L anionic surfactant, and an amine buffer at from 0.1M-0.4M and at pH 8-11.
- the compound I has the formula
- the transport device includes three pick-and-place grippers, where a first pick-and-place gripper may be used to transport sample containers among the onload section, the transfer station, and reagent pipetting stations.
- a second pick-and-place gripper may be used to transport reaction vessels between the reagent pipetting stations and the incubator station or read station.
- a third pick-and-place gripper may be used to transport reaction vessels between the incubator station and the wash station or read station.
- the immunoassay analyzer includes a machine vision apparatus comprising an image capture device and an image interpretation device configured to monitor instrument and/or assay functionalities of the immunoassay analyzer.
- instrument functionalities may include optical sensors, pressure sensors and thermistors.
- the assay functionalities may include sample volume monitoring, total reagent volume monitoring, residual volume monitoring, and particle retention monitoring.
- the machine vision apparatus operates to evaluate the preparation of samples for subsequent analysis.
- the machine vision apparatus utilizes one or more image capture units to determine whether samples have been appropriately prepared for analysis.
- the machine vision apparatus provides direct and simple measurements of volume or integrity of a sample to determine whether the sample is appropriately prepared so that the analytic unit produces a reliable result.
- An exemplary machine vision apparatus is described in U.S. Patent No. 11,263,433, which is incorporated by reference herein.
- the machine vision apparatus operates to detect a volume of a fluidic substance in a container and determine whether the volume held in the container is appropriate as targeted. As described herein, this volume detection is configured to detect a volume at a dispense tip using the dispense tip image capture unit, and a volume at a vessel using a vessel image capture unit.
- the machine vision apparatus operates to detect any interferents, which can interfere with an analytic procedure and may generate incorrect results in the dispense tip.
- this dispense tip evaluation is configured to determine a quality of a fluidic substance at a dispense tip using a dispense tip image capture unit, and an alignment of the dispense tip with respect to the dispense tip image capture unit.
- the machine vision apparatus operates to determine a particle concentration in a fluidic substance contained in a vessel, such as a reaction vessel, a sample vessel, a dilution vessel, a cuvette, or any suitable type of vessel, which is used throughout the process in the immunoassay analyzer.
- a vessel such as a reaction vessel, a sample vessel, a dilution vessel, a cuvette, or any suitable type of vessel, which is used throughout the process in the immunoassay analyzer.
- this reaction vessel particle concentration check uses the vessel image capture unit.
- the dispense tip image capture unit operates to capture images of dispense tips in one or more locations.
- the dispense tip image capture unit is fixed at a particular location in the instrument.
- the dispense tip image capture unit is movably disposed in the instrument, which can move either independently from other components of the instrument or together with one or more components of the instrument.
- Some embodiments of the instrument include a plurality of dispense tip image capture units. As described herein, the dispense tip image capture unit can include a camera unit.
- the vessel image capture unit operates to capture images of vessels in one or more locations.
- the vessel image capture unit is fixed at a particular location in the immunoassay analyzer.
- the vessel image capture unit is movably disposed in the immunoassay analyzer, which can move either independently from other components of the immunoassay analyzer or together with one or more components of the immunoassay analyzer.
- Some embodiments of the immunoassay analyzer include a plurality of vessel image capture units.
- the vessel tip image capture unit includes a camera unit.
- the carriage image capture unit operates to capture images of container carriage devices with or without containers in one or more locations.
- the carriage image capture unit is fixed at a particular location in the instrument.
- the carriage image capture unit is movably disposed in the instrument, which can move either independently from other components of the instrument or together with one or more components of the instrument.
- Some embodiments of the instrument include a plurality of carriage image capture units.
- All of the units of the immunoassay analyzer are connected to a controller, which can perform block control of all of the analyzer functions by using, for example, a microcomputer.
- the controller may contain subunits such as a data processing unit, a communication interface, and others.
- a controller in accordance with an exemplary embodiment of the present technology may comprise a data processor, a non-transitory computer-readable medium, and a data storage coupled to the data processor.
- the non-transitory computer-readable medium may comprise code, executable by the data processor, to perform the functions described herein.
- the data processor may store, for example, data for processing samples, sample data, or data for analyzing sample data.
- the data processor may include any suitable data computation device or combination of such devices.
- An exemplary data processor may comprise one or more microprocessors working together to accomplish a desired function.
- the data processor may include a CPU that comprises at least one high-speed data processor adequate to execute program components for executing user and/or system-generated requests.
- the CPU may be a microprocessor such as AMD’s Athlon, Duron and/or Opteron; IBM and/or Motorola’s PowerPC; IBM’s and Sony’s Cell processor; Intel’s Celeron, Itanium, Pentium, Xeon, and/or XScale; Apple Ml, and/or the like processor(s).
- the computer-readable medium and the data storage may be any suitable device or devices that can store electronic data.
- Examples of memories may comprise, for example, one or more memory chips, disk drives, etc. Such memories may operate using any suitable electrical, optical, and/or magnetic mode of operation.
- the computer-readable medium may comprise code, executable by the data processor to perform any suitable method.
- the computer-readable medium may comprise code, executable by the processor, to cause the controller to operate on a pre-determined schedule.
- the pre-determined schedule is a constituent test.
- the cycle time is about 45 seconds or less.
- the “cycle time” is the time required for all modules and/or functions of an immunoassay analyzer to complete its tasks necessary for generating a result.
- the cycle time is about 40 seconds or less, alternatively about 35 seconds or less, alternatively about 30 seconds or less, alternatively about 25 seconds or less, alternatively about 20 seconds or less, or alternatively about 15 seconds.
- the sample pipettor can complete its tasks in 8 seconds and the reagent pipettor can complete its tasks in 32 seconds.
- the immunoassay analyzer has at least 30 incubation positions, alternatively at least 40 incubation positions, alternatively at least 50 incubation positions, alternatively at least 60 incubation positions, alternatively at least 70 incubation positions, alternatively at least 80 incubation positions, alternatively at least 90 incubation positions, alternatively at least 100 incubation positions, alternatively at least 125 incubation positions, alternatively at least 150 incubation positions, alternatively at least 175 incubation positions, or alternatively at least 200 incubation positions.
- the time to first result is about 60 minutes or less.
- the “TTFR” is a measure of time from when the sample is aspirated to when the presence and/or concentration of an analyte is determined.
- the TTFR is about 60 minutes or less, alternatively about 55 minutes or less, alternatively about 50 minutes or less, alternatively about 45 minutes or less, alternatively about 40 minutes or less, alternatively about 35 minutes or less, alternatively about 30 minutes or less, alternatively about 25 minutes or less, alternatively about 20 minutes or less, alternatively about 15 minutes or less, or alternatively about 10 minutes or less.
- a biological sample is analyzed for p- tau217 using an immunoassay analyzer disclosed herein.
- the method includes aspirating, using a sample pipettor, a portion of the biological sample from a sample vessel and dispensing the aspirated sample into a reaction vessel of an immunoassay analyzer.
- the biological sample is serum, whole blood, plasma, and/or cerebral spinal fluid.
- the method includes aspirating, using a reagent pipettor, a portion of a first reagent from at least one reagent vessel and dispensing the aspirated reagent into the reaction vessel.
- the reagent pack may include a plurality of reagent vessels, wherein the reagent pack is configured to store a volume of reagent required for at least about 20 instances of a p-tau217 assay.
- the reagent pack includes at least three reagent vessels, wherein each reagent vessel is independently configured to store a volume of reagent required for at least about 20 instances of a p-tau217 assay.
- the reagent pack includes at least four reagent vessels, wherein each reagent vessel is independently configured to store a volume of reagent required for at least about 20 instances of a p-tau217 assay. In a non-limiting example, the reagent pack includes at least five reagent vessels, wherein each reagent vessel is independently configured to store a volume of reagent required for at least about 20 instances of a p-tau217 assay.
- a first reagent includes at least one affinity molecule configured to bind to at least one portion p-tau217.
- the affinity molecule may be an antibody, a monoclonal antibody, a polyclonal antibody, a synthetic antibody mimic, an aptamer, an affimer, DARPins, or oligonucleotides or peptides that bind to at least one epitope of p-tau217.
- the affinity molecule of the first reagent binds with the p-tau217 forming a first reaction mixture.
- the incubation time of the first reaction mixture is at least about 30 minutes, alternatively at least about 40 minutes, or alternatively at least about 50 minutes, alternatively at least about 55 minutes, or alternatively at least about 60 minutes.
- the affinity molecule is an antibody and the method further comprises exposing the biological sample to a first antibody which binds to a first p-tau217 epitope and a second antibody which binds to a second p-tau217 epitope.
- the first antibody is a capture antibody and the second antibody is a detection antibody. In other embodiments, both antibodies are capture antibodies.
- a p-tau217 antibody is bound to magnetic bead.
- Magnetic beads comprising said antibody are added to the sample in a reaction vessel along. At least one buffer or other reagents may be added as required by the assay.
- the mixture is incubated for a period of time necessary for the antibody to bind with a p-tau217 epitope forming a first reaction mixture.
- a magnetic field is applied to the reaction vessel and the isolated, bead-bound proteins are washed using the washing arrangement.
- a base number (three) of wash actions is performed with optional one or two additional wash actions.
- the method includes, aspirating, using the reagent pipettor, a portion of a second reagent from at least one reagent vessel and dispensing the aspirated reagent into the reaction vessel.
- a second reagent includes a detection molecule.
- the detection molecule may be, for example, a detection antibody, such as alkaline phosphatase (AP)- conjugated secondary antibody.
- the detection molecule of the second reagent binds with the affinity molecule of the first reagent (which is bound with the p-tau217 in the biological sample) or binds to a different p-217 epitope.
- the incubation time of the second reaction mixture is at least about 5 minutes, alternatively at least about 8 minutes, or alternatively at least about 10 minutes.
- a p-tau217 antibody conjugated to alkaline phosphatase is added to the first reaction mixture in a reaction vessel.
- the mixture in incubated for a period of time necessary for the antibody to bind with a second p-tau217 epitope, forming a second reaction mixture.
- a magnetic field is applied to the reaction vessel and the isolated, bead-bound proteins are washed using the washing arrangement.
- a base number (three) of wash actions is performed with optional one or two additional wash actions.
- antibody refers to a protein which exhibits binding specificity to a specific antigen.
- An antibody often comprises a variable domain and a constant domain, each of which includes a heavy chain and a light chain. Accordingly, most antibodies have a heavy chain variable domain (VH) and a light chain variable domain (VL) that together form the portion of the antibody that binds to the antigen. Within each variable domain are three complementarity determining regions (CDR) which form loops in the heavy chain variable domain (VH) and light chain variable domain (VL) that contact the surface of the antigen.
- CDR complementarity determining regions
- Antibodies herein also include intact molecules as well as functional fragments thereof, which are also referred to as an “antigen binding portion” or fragments of the antibody that are capable of binding to the antigen.
- chimeric antibodies are antibodies having a portion of the heavy and/or light chain identical with or homologous to corresponding sequences in antibodies derived from a particular species or belonging to a particular antibody class or subclass, while the remainder of the chain(s) is identical with or homologous to corresponding sequences in antibodies derived from another species or belonging to another antibody class or subclass, as well as fragments of such antibodies, so long as they exhibit the desired biological activity (see e.g., Morrison et al., Proc. Natl. Acad. Sci. USA 81 :6851-6855 (1984)).
- p-tau217 antibodies comprise at least one heavy chain and at least one light chain. In some embodiments, p-tau217 antibodies comprise at least one heavy chain comprising a VH and at least one light chain comprising a VL, wherein each VH and VL comprises three CDRs. The amino acid sequences of the VH and VL and the CDRs determine the antigen binding specificity and antigen binding strength of the antibody. [0213] In some embodiments, the detection antibody binds to an epitope on p-tau217 comprising amino acids 1 - 25, 1 - 20, 6 - 18 or 7 - 20 of SEQ ID NO: 1.
- the detection antibody binds to an epitope on p-tau217 comprising 7 - 20 of SEQ ID NO: 1. In some embodiments the detection antibody binds to an epitope on p-tau217 comprising 6 - 18 of SEQ ID NO: 1. In some embodiments the capture antibody binds to an epitope on p-tau217 comprising amino acids 217, 210 - 225, or 210 - 220 of SEQ ID NO: 1. In some embodiments the capture antibody binds to an epitope on p-tau217 comprising amino acids 210 - 220 of SEQ ID NO: 1 of SEQ ID NO: 1.
- the detection antibody binds to an epitope on p-tau217 having the amino acid sequence set forth in SEQ ID NOS: 2, 3 or 4 herein. In some embodiments the detection antibody binds to an epitope on p-tau217 having an amino acid sequence with a sequence identity of at least about 75%, at least about 80%, at least about 81%, at least about 82%, at least about 83%, at least about 84%, at least about 85%, at least about 86%, at least about 87%, at least about 88%, at least about 89%, at least about 90%, at least about 91%, at least about 92%, at least about 93%, at least about 94%, at least about 95%, at least about 96%, at least about 97%, at least about 98%, or at least about 99% identical to the amino acid sequences set forth in SEQ ID NOS: 2, 3 or 4 herein.
- the detection antibody binds to an epitope on p-tau217 comprising amino acids 1 - 20 of SEQ ID NO: 1
- the capture antibody binds to an epitope on p-tau217 comprising amino acids 210 - 220 of SEQ ID NO: 1.
- the detection antibody binds to an epitope on p-tau217 having the amino acid sequence set forth in SEQ ID NO: 2
- the capture antibody binds to an epitope on p-tau217 having an epitope on p-tau217 having the amino acid sequence set forth in SEQ ID NO: 4.
- the detection antibody binds to an epitope on p-tau217 having an amino acid sequence with a sequence identity of at least about 75%, at least about 80%, at least about 81%, at least about 82%, at least about 83%, at least about 84%, at least about 85%, at least about 86%, at least about 87%, at least about 88%, at least about 89%, at least about 90%, at least about 91%, at least about 92%, at least about 93%, at least about 94%, at least about 95%, at least about 96%, at least about 97%, at least about 98%, or at least about 99% identical to the amino acid sequence set forth in SEQ ID NO: 2, and the capture antibody binds to an epitope on p-tau217 having an amino acid sequence with a sequence identity of at least about 75%, at least about 80%, at least about 81%, at least about 82%, at least about 83%, at least about 84%, at least about 85%, at least about 86%, at least
- the detection antibody that binds p-Tau217 comprises a heavy chain variable region (VH) of SEQ ID NO: 13. In some instances, the detection antibody that binds p-tau217 comprises an antigen-binding site of a light chain variable region (VL) of SEQ ID NO: 14.
- the detection antibody that binds p-Tau217 comprises a VH having a sequence identity of at least about 75%, at least about 80%, at least about 81%, at least about 82%, at least about 83%, at least about 84%, at least about 85%, at least about 86%, at least about 87%, at least about 88%, at least about 89%, at least about 90%, at least about 91%, at least about 92%, at least about 93%, at least about 94%, at least about 95%, at least about 96%, at least about 97%, at least about 98%, or at least about 99% identical to an amino acid sequence set forth as SEQ ID NO: 13.
- the capture antibody that binds p-Tau217 comprises a VL having a sequence identity of at least about 75%, at least about 80%, at least about 81%, at least about 82%, at least about 83%, at least about 84%, at least about 85%, at least about 86%, at least about 87%, at least about 88%, at least about 89%, at least about 90%, at least about 91%, at least about 92%, at least about 93%, at least about 94%, at least about 95%, at least about 96%, at least about 97%, at least about 98%, or at least about 99% identical to an amino acid sequence set forth as SEQ ID NO: 14.
- the capture antibody that binds p-tau217 comprises an antigenbinding site of a heavy chain variable region (VH) of SEQ ID NO: 13, and an antigen-binding site of a light chain variable region (VL) of SEQ ID NO: 14.
- the detection antibody comprises heavy chain CDRs - HCDR1, HCDR2 and HCDR3 - shown in SEQ ID NOs: 15, 16 and 17. In some instances, the detection antibody comprises light chain CDRs - LCDR1, LCDR2 and LCDR3 - shown in SEQ ID NOs: 18, 19 and 20.
- the detection antibody comprises heavy chain CDRs - HCDR1, HCDR2 and HCDR3- having a sequence identity of at least about 75%, at least about 80%, at least about 81%, at least about 82%, at least about 83%, at least about 84%, at least about 85%, at least about 86%, at least about 87%, at least about 88%, at least about 89%, at least about 90%, at least about 91%, at least about 92%, at least about 93%, at least about 94%, at least about 95%, at least about 96%, at least about 97%, at least about 98%, or at least about 99% identical to an amino acid sequence set forth as SEQ ID NOs: 15, 16 and 17.
- the detection antibody comprises light chain CDRs - LCDR1, LCDR2 and LCDR3- having a sequence identity of at least about 75%, at least about 80%, at least about 81%, at least about 82%, at least about 83%, at least about 84%, at least about 85%, at least about 86%, at least about 87%, at least about 88%, at least about 89%, at least about 90%, at least about 91%, at least about 92%, at least about 93%, at least about 94%, at least about 95%, at least about 96%, at least about 97%, at least about 98%, or at least about 99% identical to an amino acid sequence set forth as SEQ ID NOs: 18, 19 and 20.
- the detection antibody comprises heavy chain CDRs - HCDR1, HCDR2 and HCDR3 - having the amino acids sequence set forth in SEQ ID NOs: 15, 16 and 17 and light chain CDRs - LCDR1, LCDR2 and LCDR3 - having the amino acid sequence set forth in SEQ ID NOs: 18, 19 and 20.
- the detection antibody and/or capture antibody may be a polyclonal antibody or a monoclonal antibody.
- Monoclonal antibodies may be acquired using know techniques including by using an antibody-producing cells such as hybridomas.
- the capture antibody that binds p-tau217 comprises a heavy chain variable region (VH) of SEQ ID NO: 5. In some instances, the capture antibody that binds p- tau217 comprises a light chain variable region (VL) of SEQ ID NO: 6.
- the capture antibody that binds p-tau217 comprises a VH having a sequence identity of at least about 75%, at least about 80%, at least about 81%, at least about 82%, at least about 83%, at least about 84%, at least about 85%, at least about 86%, at least about 87%, at least about 88%, at least about 89%, at least about 90%, at least about 91%, at least about 92%, at least about 93%, at least about 94%, at least about 95%, at least about 96%, at least about 97%, at least about 98%, or at least about 99% identical to an amino acid sequence set forth as SEQ ID NO: 5.
- the capture antibody that binds p-tau217 comprises a VL having a sequence identity of at least about 75%, at least about 80%, at least about 81%, at least about 82%, at least about 83%, at least about 84%, at least about 85%, at least about 86%, at least about 87%, at least about 88%, at least about 89%, at least about 90%, at least about 91%, at least about 92%, at least about 93%, at least about 94%, at least about 95%, at least about 96%, at least about 97%, at least about 98%, or at least about 99% identical to an amino acid sequence set forth as SEQ ID NO: 6.
- the capture antibody that binds p-tau217 comprises an antigen-binding site of a heavy chain variable region (VH) of SEQ ID NO: 5, and an antigen-binding site of a light chain variable region (VL) of SEQ ID NO: 6.
- the capture antibody comprises heavy chain CDRs - HCDR1, HCDR2 and HCDR3 - having the amino acid sequence set forth in SEQ ID NOs: 7, 8 and 9. In some instances, the capture antibody comprises light chain CDRs - LCDR1, LCDR2 and LCDR3- having the amino acid sequence set forth in SEQ ID NOs: 10, 11 and 12.
- the capture antibody comprises heavy chain CDRs - HCDR1, HCDR2 and HCDR3 - having a sequence identity of at least about 75%, at least about 80%, at least about 81%, at least about 82%, at least about 83%, at least about 84%, at least about 85%, at least about 86%, at least about 87%, at least about 88%, at least about 89%, at least about 90%, at least about 91%, at least about 92%, at least about 93%, at least about 94%, at least about 95%, at least about 96%, at least about 97%, at least about 98%, or at least about 99% identical to an amino acid sequence set forth as SEQ ID NOs: 7, 8 and 9.
- the capture antibody comprises light chain CDRs - LCDR1, LCDR2 and LCDR3 - having a sequence identity of at least about 75%, at least about 80%, at least about 81%, at least about 82%, at least about 83%, at least about 84%, at least about 85%, at least about 86%, at least about 87%, at least about 88%, at least about 89%, at least about 90%, at least about 91%, at least about 92%, at least about 93%, at least about 94%, at least about 95%, at least about 96%, at least about 97%, at least about 98%, or at least about 99% identical to an amino acid sequence set forth as SEQ ID NOs: 10, 11 and 12.
- the capture antibody comprises heavy chain CDRs - HCDR1, HCDR2 and HCDR3 - having a sequence as set forth in SEQ ID NOs: 7, 8 and 9 and light chain CDRs - LCDR1, LCDR2 and LCDR3 - having a sequence as set forth in SEQ ID NOs: 10, 11 and 12.
- the method includes dispensing a substrate formulation into the reaction vessel, generating a detection mixture.
- the substrate formulation comprises compound I, a cationic aromatic compound, a background reducing agent, an ether-linked non-ionic surfactant or hydrophilic polymer, an anionic surfactant, and an amine buffer.
- a magnetic field is applied to the reaction vessel and the isolated, bead-bound proteins are washed using the washing arrangement.
- a base number (three) of wash actions is performed with optional one or two additional wash actions.
- the method further includes detecting, using a detector arrangement, a presence of a reaction in the detection mixture and determining a presence and/or concentration of p-tau217 in the biological sample based on the presence of the reaction in the detection mixture.
- the reaction in the detection mixture generates a chemiluminescent signal, wherein the chemiluminescent signal corresponds to the presence and/or concentration of p-tau217 in the biological sample.
- the sample pipettor can aspirate the sample from the sample vessel and then move to a sample dispense position, where the reaction vessels are provided.
- a nonlimiting example of an operation is depicted in FIGs. 2A-2C.
- the sample pipettor operates to dispense the aspirated samples into the reaction vessels 202.
- the sample pipettor operates to dispense a predetermined volume of sample into four different reaction vessels.
- the first reagent pipettor operates to aspirate a portion of a fluidic substance from a first reagent vessel and dispense a predetermined volume of reagent into a first reaction vessel 204.
- the second reagent pipettor operates to aspirate a portion of a fluidic substance from a first reagent vessel and dispense a predetermined volume of reagent into a second reaction vessel 206.
- the third reagent pipettor operating to aspirate a portion of a fluidic substance from a first reagent vessel and dispense a predetermined volume of reagent into a third reaction vessel 208
- the fourth reagent pipettor operating to aspirate a portion of a fluidic substance from a first reagent vessel and dispense a predetermined volume of reagent into a fourth reaction vessel 210.
- all of the reaction vessel may be incubated simultaneously to form a first reaction mixture.
- the first reagent pipettor operates to aspirate a portion of a fluidic substance from a second reagent vessel and dispense a predetermined volume of reagent into a first reaction vessel 212.
- the second reagent pipettor operates to aspirate a portion of a fluidic substance from a second reagent vessel and dispense a predetermined volume of reagent into a second reaction vessel 214.
- the third reagent pipettor operating to aspirate a portion of a fluidic substance from a second reagent vessel and dispense a predetermined volume of reagent into a third reaction vessel 216 and the fourth reagent pipettor operating to aspirate a portion of a fluidic substance from a second reagent vessel and dispense a predetermined volume of reagent into a fourth reaction vessel 218.
- all of the reaction vessel may be incubated simultaneously to form a second reaction mixture.
- a substrate formulation is dispensed into the first reaction vessel, generating a first reaction mixture 220.
- a substrate formulation is dispensed into the second reaction vessel, generating a second reaction mixture 222.
- a substrate formulation is dispensed into the second reaction vessel, generating a second reaction mixture 222.
- a substrate formulation is dispensed into the third reaction vessel, generating a third reaction mixture 224 and dispensing a substrate formulation into the fourth reaction vessel, generating a fourth reaction mixture 226.
- all of the reaction vessels may be incubated simultaneously to form the detection mixtures.
- the incubated detection mixtures are then individually and independently analyzed. This includes the use of the detector arrangement to detect a reaction in the detection mixture in the first reaction vessel, the second reaction vessel, the third reaction vessel, and the fourth reaction vessel 228. Based on the presence of a reaction in the detection mixture, the amount of p-tau217 in the plasma sample in each reaction vessel can be quantitatively determined 230.
- the claimed configuration allows for the simultaneous performance of at least two assays for a plurality of plasma samples, alternatively at least three assays for a plurality of plasma samples, or at least four assays for a plurality of plasma samples.
- the plasma samples may be from the same subject or from multiple subjects.
- the method further comprises a plurality of sample vessels.
- the method comprises at least one sample vessel, at least two sample vessels, at least three sample vessels, at least four sample vessels, at least five sample vessels, at least ten sample vessels, at least twenty sample vessels, at least fifty sample vessels, at least one hundred sample vessels, at least two hundred sample vessels, at least three hundred sample vessels, at least four hundred sample vessels, or at least five hundred sample vessels.
- the sample vessels are housed within the immunoassay analyzer.
- At least one of the assays is p-tau217 assay, alternatively at least two of the assays are p-tau217 assays, alternatively at least three of the assays are p-tau217 assays, or alternatively at least four of the assays are p-tau217 assays.
- the method may include detecting at least one additional biomarker related to Alzheimer’s Disease or another neurological disorder.
- at least one of the assays is p-tau217 assay and at least one of the assays is a second analyte assay, wherein the second analyte is selected from the group consisting of A
- the first pipettor may be configured to aspirate a reagent from a reagent vessel comprising a first affinity molecule (e.g., a p-tau217 antibody) and dispense into a first reaction vessel
- a second pipettor may be configured to simultaneously aspirate a reagent from a reagent vessel comprising a second affinity molecule (e.g., second analyte antibody) and dispense into a second reaction vessel.
- the third and fourth pipettor may also be configured to simultaneously aspirate a reagent from a reagent vessel comprising a third and fourth affinity molecule and dispense into a third and fourth reaction vessel.
- the disclosed simultaneous and/or selective operation of the pipettors allows for a high- throughput analysis.
- the method is configured to analyze at least about 200 plasma samples/hr., alternatively at least about 300 plasma samples/hr., or alternatively at least about 400 plasma samples/hr.
- the levels of p- tau217 in blood may be in the AttoMole, Milli-international Unit (mIU), pg, or fg range.
- the quantitation detection limit (LOQ) for measuring p-tau217 is equal to or less than about 3 pg/mL, alternatively equal to or less than about 2 pg/mL, equal to or less than about 1 pg/mL, equal to or less than about 0.6 pg/mL and wherein the detected p-tau217 has a concentration above the LOQ.
- the quantitation detection limit (LOQ) for measuring p-tau217 is equal to or less than about 100 pg/mL, less than about 90 pg/mL, less than about 80 pg/mL, less than about 70 pg/mL, less than about 60 pg/mL, less than about 50 pg/mL, less than about 40 pg/mL, less than about 30 pg/mL, less than about 20 pg/mL, or less than about 10 pg/mL.
- the quantitation detection limit (LOQ) for measuring p-tau217 is equal to or less than about less than about 100 pg/mL, less than about 90 pg/mL, less than about 80 pg/mL, less than about 70 pg/mL, less than about 60 pg/mL, less than about 50 pg/mL, less than about 40 pg/mL, less than about 30 pg/mL, less than about 20 pg/mL, or less than about 10 pg/mL.
- the quantitation detection limit (LOQ) for measuring p-tau217 is equal to or less than about 10 pg/mL, less than about 9 pg/mL, less than about 8 pg/mL, less than about 7 pg/mL, less than about 6 pg/mL, less than about 5 pg/mL, less than about 4 pg/mL, less than about 3 pg/mL, less than about 2 pg/mL, or less than about 1 pg/mL.
- the quantitation detection limit (LOQ) for measuring p-tau217 is equal to or less than about Ipg/mL, less than about 0.9pg/mL, less than about 0.8pg/mL, less than about 0.7pg/mL, less than about 0.6pg/mL, less than about 0.5pg/mL, less than about 0.4pg/mL, less than about 0.3pg/mL, less than about 0.2pg/mL, or less than about O. lpg/mL.
- the quantitation detection limit (LOQ) for measuring p- tau217 is equal to or less than about 100 fg/mL, less than about 90 fg/mL, less than about 80 fg/mL, less than about 70 fg/mL, less than about 60 fg/mL, less than about 50 fg/mL, less than about 40 fg/mL, less than about 30 fg/mL, less than about 20 fg/mL, or less than about 10 fg/mL.
- the quantitation detection limit (LOQ) for measuring p- tau217 is equal to or less than about 10 fg/mL, less than about 9 fg/mL, less than about 8 fg/mL, less than about 7 fg/mL, less than about 6 fg/mL, less than about 5 fg/mL, less than about 4 fg/mL, less than about 3 fg/mL, less than about 2 fg/mL, or less than about 1 fg/mL.
- the detected p-tau217 has a concentration that is at least IX greater than the LOQ, alternatively at least 2X greater than the LOQ, alternatively at least 3X greater than the LOQ, alternatively at least 4X greater than the LOQ, alternatively at least 5X greater than the LOQ, alternatively at least 10X greater than the LOQ, alternatively at least 100X greater than the LOQ, or alternatively at least 100X greater than the LOQ.
- the detected p-tau217 exhibits a coefficient of variation (CV) of 20% or less, 19% or less, 18% or less, 17% or less, 16% or less, 15% or less, 14% or less, 13% or less, 12% or less, 11% or less, 10% or less, 9% or less, 8% or less, 7% or less, 6% or less, 5% or less, 4% or less, 3% or less, 2% or less, or 1% or less.
- the detected p-tau217 exhibits a coefficient of variation (CV) of 20% or less.
- the detected p-tau217 exhibits a coefficient of variation (CV) of 4% or less.
- an exemplary immunoassay analyzer i.e., an immunoassay analyzer according to an aspect of this disclosure
- conventional immunoassay analyzers were evaluated to demonstrate the sensitivity for existing immunoassays.
- Thyroid-Stimulating Hormone (TSH) commercial assays were employed per manufacturers’ protocol.
- An exemplary assay protocol according to an aspect of the invention is listed in Example 1.
- Commercially available calibrators were used to create series of dilutions. The assay protocol was performed on each dilution to generate a measurement in RLUs, and corresponding coefficient of variation (CV), signal-noise-ratios, LOQs, and other performance characteristics were calculated.
- CV coefficient of variation
- the signal -to-noise ratio gives a measure of the degree of confidence that a difference in signal noise and background is real.
- S/N The signal -to-noise ratio
- a blank control is analyzed and the resulting values are used to calculate S/N.
- the error range corresponds to the CV of the specific reportable parameter at the concentration tested. It is noted that the CV varies depending on the concentrations and instruments.
- the 1st conventional immunoassay analyzer had low CVs, but there was little RLUs left at the lower concentrations.
- the 2nd conventional immunoassay analyzer had improved signal/noise ratios, but poor CVs at the end.
- the exemplary immunoassay analyzer was the only immunoassay analyzer that did not have technical limitations in achieving the sensitivity useful for detecting low p-tau217 concentrations.
- the LOQ of the exemplary immunoassay analyzer is at least around 0.013 pIU/mL (appx. 2 pg /mL) with a CV of around 14.2%.
- the disclosed immunoassay methods achieve a functional sensitivity of 0.01-0.02 pIU/mL (mIU/L) with an interassay %CV ⁇ 20%. This unpredictable magnitude of sensitivity is superior to conventional methods allowing for the sensitivity and precision needed to distinguish and quantify different low levels of p-tau217.
- a calibrator (SI, 0.0500 pIU/mL (micro-international units per milliliter) was used to create a series of dilutions with concentrations of 0.0050 pIU/mL (Sl/10), 0.0025 pIU/mL (Sl/20), 0.0013 pIU/mL (Sl/40), 0.0006 pIU/mL (Sl/80), and 0.0000 pIU/mL (SO).
- the TSH sensitivity comparison was performed using an exemplary immunoassay analyzer having (i) four reagent pipettors and one sample pipettor configured to aspirate and dispense less than about 10 pL; (ii) reagent packs configured to store volumes of reagents for at least 20 instances of assays; (iii) an ultrasonic mixer; and (iv) the capability to perform over 5 wash cycles per reaction vessel.
- Paramagnetic particle conjugated with TSH antibodies capable of binding to an epitope TSH were pipetted into a reaction vessel using one of four reagent pipettors. A sample aliquot was pipetted into the reaction vessel using a sample pipettor. Then the reaction vessel was mixed using the ultrasonic mixer and incubated, generating a first reaction mixture. A magnetic field was applied to the reaction vessel and the first reaction mixture was washed using a wash buffer to remove any unreacted components. A secondary antibody capable of binding a different epitope of TSH conjugated to alkaline phosphatase (“ALP”) was added to the reaction vessel containing the first reaction mixture. The reaction vessel was mixed ultrasonically and incubated, generating a second reaction mixture.
- ALP alkaline phosphatase
- a magnetic field was applied to the reaction vessel and the second reaction mixture was washed using a wash buffer to remove any unreacted components.
- a substrate according to the present disclosure was added to the reaction vessel, and the signal generated from the resulting reaction was read using a luminometer.
- Test results were determined automatically by the system software. Detection of analyte in the sample was determined from the measured light production by means of the stored calibration data. The RLU data collected for each assayed sample was summed for each individual run, averaged over the triplicates for each sample and presented in FIG. 1. A ratio of signal to noise (S/SO), RLU CVs, CVCF, and Dose CVs were calculated at each concentration value.
- the exemplary immunoassay analyzer was found to have both a higher signal to noise ratio (S/SO) and lower CVs at the lower concentrations. This is in contrast to 1 st conventional immunoassay analyzer which had lower CVs, but lower RLUs left at the lower concentrations. The 2nd conventional immunoassay analyzer had higher CVs at the lower concentrations.
- the LOQ of the exemplary immunoassay analyzer is at least around 0.013 pIU/mL (appx. 2 pg/mL) with a CV of around 14.2%.
- Example 2 Exemplary p-tau217 assay
- An exemplary assay protocol (Table 4) is used to detect the presence of p-tau217 in a plasma sample.
- the assay was performed using an exemplary immunoassay analyzer according to an aspect of the disclosure.
- a single calibration is generated (duplicate replicates of each level) and a calibration curve is fit to the corresponding signal output.
- the assay is then used to measure p-tau217 levels in the biological samples
- Example 3 Clinical Discrimination of pTau217 Assay on Exemplary Clinical Analyzer
- Samples tested included 26 normal patient samples (8 young ⁇ 55 years patients; 18 aged healthy patients >55 years) and 40 Alzheimer’s positive patient samples (confirmed by physician; MMSE scores ranging from 2 to 25).
- an exemplary immunoassay analyzer having (i) four reagent pipettors and one sample pipettor configured to aspirate and dispense less than about 10 pL; (ii) reagent packs configured to store volumes of reagents for at least 20 instances of assays; (iii) an ultrasonic mixer; and (iv) the capability to perform over 5 wash cycles per reaction vessel, the following steps were carried out:
- Step 1 Paramagnetic particle conjugated p-tau217 antibodies capable of binding to an epitope comprising amino acid 217 (SEQ ID NO: 4) of p-tau217 were pipetted into a reaction vessel using one of four reagent pipettors.
- the paramagnetic particle conjugated p-tau217 antibodies comprised CDR regions having the amino acid sequences of SEQ ID NOs: 7 - 12, a VL region having the amino sequence of SEQ ID NO: 6, and a VH region having the amino acid sequence of SEQ ID NO: 5.
- a sample aliquot was pipetted into the reaction vessel using a sample pipettor. Then the reaction vessel was mixed using the ultrasonic mixer and incubated, generating a first reaction mixture.
- Step 2 A magnetic field was applied to the reaction vessel and the first reaction mixture was washed using a wash buffer to remove any unreacted components.
- Step 3 A p-tau217 antibody capable of binding to the N-terminus of p-tau217 (SEQ ID NO: 3) conjugated to alkaline phosphatase (“ALP”) was added to the reaction vessel containing the first reaction mixture.
- the ALP conjugated p-tau217 antibodies comprised CDR regions having the amino acid sequences of SEQ ID NOs: 15 - 20, a VL region having the amino sequence of SEQ ID NO: 14, and a VH region having the amino acid sequence of SEQ ID NO: 13.
- the reaction vessel was mixed ultrasonically and incubated, generating a second reaction mixture.
- Step 4 A magnetic field was applied to the reaction vessel and the second reaction mixture was washed using a wash buffer to remove any unreacted components.
- Step 5 Substrate according to the present disclosure was added to the reaction vessel, and the signal generated from the resulting reaction was read using a luminometer.
- Alzheimer’s positive population were determined to have a median pTau217 concentration of 261 fg/mL, whereas Alzheimer’s normal healthy population were found to have a pTau217 concentration of 88 fg/mL, with a median based ratio of AD-positive to normal concentrations of 3.0.
- AD Alzheimer’s Disease
- Samples included 20 Alzheimer’s positive Patients and 20 Normal Patients. Each of the assays was performed using one of six antibody pairs.
- Each p-Tau217 assay as described herein was performed on a Beckman Coulter’s Dxl 9000 analyzer having (i) four reagent pipettors and one sample pipettor configured to aspirate and dispense less than about 10 pL; (ii) a reagent pack with 4 reagent vessels (the first vessel containing paramagnetic particle conjugated p-tau217 antibodies, the second vessel containing ALP conjugated p-tau217 antibodies and the third and fourth vessels containing ancillary buffers required for the assay), where each reagent vessel is configured to store volumes of reagents for at least 20 instances of assays; (iii) an ultrasonic mixer; and (iv) the capability to perform over 5 wash cycles per reaction vessel.
- Step 1 An aliquot from the first vessel containing paramagnetic particle conjugated p- tau217 antibodies was pipetted into a reaction vessel using one of four reagent pipettors. A sample aliquot was pipetted into the reaction vessel using a sample pipettor and an aliquot from the third reagent vessel containing ancillary buffers was pipetted into the reaction vessel using one of four reagent pipettors. Then the reaction vessel was incubated for 32 seconds. A second sample aliquot was pipetted into the reaction vessel using a sample pipettor and the reaction vessel was mixed using the ultrasonic mixer and incubated for about 20 minutes, generating a first reaction mixture.
- Step 2 A magnetic field was applied to the reaction vessel and the first reaction mixture was washed three times using a wash buffer to remove any unreacted components.
- Step 3 An aliquot from the second vessel containing ALP conjugated p-tau217 antibodies was pipetted into a reaction vessel using one of four reagent pipettors. The reaction vessel was mixed ultrasonically and incubated for about 8 minutes, generating a second reaction mixture.
- Step 4 A magnetic field was applied to the reaction vessel and the second reaction mixture was washed five times using a wash buffer to remove any unreacted components.
- Step 5 Substrate according to the present disclosure was added to the reaction vessel, and the signal generated from the resulting reaction was read using a luminometer.
- the assay was performed on the Normal (4 replicates each) and AD-confirmed (2 replicates each) patient samples and a calibrator matrix (20 replicates). Corresponding concentrations were calculated for each replicate based on calibration curve fit and the CV% calculated for each sample. Within-run precision was analyzed to generate a precision profile to estimate Limit of Blank, Limit of Detection, and the 20% Limit of Quantitation. The precision profile and calculated attributes of the assay are depicted in FIG. 6.
- AD Alzheimer’s Disease
- Samples included 32 Alzheimer’s + Patients (MMSE Range 10-24), 27 Normal Patients (Age Range 36-74), 4 CSF Spiked Plasma Samples, and 9 Antigen Spiked Samples (recombinant). Each of the assays was performed using antibody pair 1, antibody pair 4, and two conventional antibody pairs.
- Each p-Tau217 assay as described herein was performed on a Beckman Coulter’s Dxl 9000 analyzer having (i) four reagent pipettors and one sample pipettor configured to aspirate and dispense less than about 10 pL; (ii) a reagent pack with 4 reagent vessels (the first vessel containing paramagnetic particle conjugated p-tau217 antibodies, the second vessel containing ALP conjugated p-tau217 antibodies and the third and fourth vessels containing ancillary buffers required for the assay), where each reagent vessel is configured to store volumes of reagents for at least 20 instances of assays; (iii) an ultrasonic mixer; and (iv) the capability to perform over 5 wash cycles per reaction vessel.
- Step 1 An aliquot from the first vessel containing paramagnetic particle conjugated p- tau217 antibodies was pipetted into a reaction vessel using one of four reagent pipettors. A sample aliquot was pipetted into the reaction vessel using a sample pipettor and an aliquot from the third reagent vessel containing ancillary buffers was pipetted into the reaction vessel using one of four reagent pipettors. Then the reaction vessel was incubated for 32 seconds. A second sample aliquot was pipetted into the reaction vessel using a sample pipettor and the reaction vessel was mixed using the ultrasonic mixer and incubated for about 20 minutes, generating a first reaction mixture. [0288] Step 2: A magnetic field was applied to the reaction vessel and the first reaction mixture was washed three times using a wash buffer to remove any unreacted components.
- Step 3 An aliquot from the second vessel containing ALP conjugated p-tau217 antibodies was pipetted into a reaction vessel using one of four reagent pipettors. The reaction vessel was mixed ultrasonically and incubated for about 8 minutes, generating a second reaction mixture.
- Step 4 A magnetic field was applied to the reaction vessel and the second reaction mixture was washed five times using a wash buffer to remove any unreacted components.
- Step 5 Substrate according to the present disclosure was added to the reaction vessel, and the signal generated from the resulting reaction was read using a luminometer.
- FIGs. 8A-8C The precision profile and calculated attributes of antibody pair 1 and both conventional antibody pairs are depicted in FIGs. 8A-8C.
- FIG. 8A depicts %CV for the analyzed antibody pairs
- FIG. 8B depicts within-run precision profiles for the analyzed antibody pairs
- FIG. 8C depicts the measured concentrations for the analyzed antibody pairs for AD Patients and Normal patients.
- FIG. 9 is a graph of a comparison between the assay performed using pairs 1 and 4 to conventional assays. As shown in FIG. 9 the experimental antibody pairs demonstrate similar sensitivity and clinical discrimination between healthy and Alzheimer’s diseased patient EDTA plasma samples as the conventional antibody pairs.
- Both antibody pairs show superior specific signal production compared to predicate conventional antibody pairs and superior analytical sensitivity compared to predicate conventional antibody pairs.
- An exemplary p-Tau217 immunoassay using an exemplary antibody pair was assessed for clinical performance including the ability to distinguish between Alzheimer’s Disease (AD) confirmed patient samples, other neurodegenerative disease patient samples, and normal patient samples.
- AD Alzheimer’s Disease
- the samples included 45 samples from patients with an Alzheimer’s diagnosis, 20 samples from patients with other neurodegenerative diseases (including frontotemporal dementia and Lewy body disease), and 25 aged match control patient samples.
- the patients with an Alzheimer’s diagnosis were characterized for their amyloid status by CSF Ab42/Ab40 and/or amyloid PET.
- the p-Tau217 assay as described herein was performed on a Beckman Coulter’s Dxl 9000 analyzer having (i) four reagent pipettors and one sample pipettor configured to aspirate and dispense less than about 10 pL; (ii) a reagent pack with 4 reagent vessels (the first vessel containing paramagnetic particle conjugated p-tau217 antibodies, the second vessel containing ALP conjugated p-tau217 antibodies and the third and fourth vessels containing ancillary buffers required for the assay), where each reagent vessel is configured to store volumes of reagents for at least 20 instances of assays; (iii) an ultrasonic mixer; and (iv) the capability to perform over 5 wash cycles per reaction vessel.
- Step 1 An aliquot from the first vessel containing paramagnetic particle conjugated p- tau217 antibodies was pipetted into a reaction vessel using one of four reagent pipettors. A sample aliquot was pipetted into the reaction vessel using a sample pipettor and an aliquot from the third reagent vessel containing ancillary buffers was pipetted into the reaction vessel using one of four reagent pipettors. Then the reaction vessel was incubated. A second sample aliquot was pipetted into the reaction vessel using a sample pipettor and the reaction vessel was mixed using the ultrasonic mixer and incubated, generating a first reaction mixture.
- Step 2 A magnetic field was applied to the reaction vessel and the first reaction mixture was washed three times using a wash buffer to remove any unreacted components.
- Step 3 An aliquot from the second vessel containing ALP conjugated p-tau217 antibodies was pipetted into a reaction vessel using one of four reagent pipettors. The reaction vessel was mixed ultrasonically and incubated, generating a second reaction mixture.
- Step 4 A magnetic field was applied to the reaction vessel and the second reaction mixture was washed five times using a wash buffer to remove any unreacted components.
- Step 5 Substrate according to the present disclosure was added to the reaction vessel, and the signal generated from the resulting reaction was read using a luminometer.
- the sensitivity versus 1-Specifity for the resulting data was plotted, generating a receiver operating characteristic (ROC) curve.
- the area under the curve (AUC) which is an effective and combined measure of sensitivity and specificity that describes the inherent validity of diagnostic tests, was then calculated.
- a method for detecting phosphorylated tau (p-tau)217 in a biological sample using an immunoassay analyzer comprising: aspirating, using a sample pipettor, a portion of the biological sample from a sample vessel and dispensing the aspirated biological sample into a reaction vessel of the immunoassay analyzer; wherein the immunoassay analyzer comprises: a reagent pack configured to hold a plurality of reagent vessels; a pipettor arrangement comprising at least one reagent pipettor and at least one sample pipettor; and a detector arrangement; aspirating, using the reagent pipettor, a portion of a first reagent from at least one reagent vessel and dispensing the aspirated reagent into the reaction vessel, generating a first reaction mixture; aspirating, using the reagent pipettor, a portion of a second reagent from at least one reagent vessel and dispensing the aspirated reagent into the reaction vessel
- cycle time is about 45 seconds or less, alternatively about 40 seconds or less, alternatively about 35 seconds or less, alternatively about 30 seconds or less, alternatively about 25 seconds or less, alternatively about 20 seconds or less, or alternatively about 15 seconds.
- time to first result is about 60 minutes or less, alternatively about 55 minutes or less, alternatively about 50 minutes or less, alternatively about 45 minutes or less, alternatively about 40 minutes or less, alternatively about 35 minutes or less, alternatively about 30 minutes or less, alternatively about 25 minutes or less, alternatively about 20 minutes or less, alternatively about 15 minutes or less, or alternatively about 10 minutes or less.
- incubation time of the first reaction mixture is at least about 30 minutes, alternatively at least about 40 minutes, or alternatively at least about 50 minutes.
- the first reagent comprises at least one affinity molecule configured to bind to at least one portion p-tau217
- the second reagent comprises at least one detection molecule
- the substrate formulation is configured to produce chemiluminescence.
- the substrate formulation is configured to produce chemiluminescence and comprises: a chemiluminescent compound of formula I or a salt thereof: wherein
- A is Ci-ehaloalkyl, naphthyl, phenyl, substituted phenyl, or heteroaryl, wherein substituted phenyl comprises from 1 to 3 halo, Ci-6 alkyl, Ci-6 alkoxy, Ci-6 haloalkyl, C(O)Ris, CN or NO2 substituents;
- Ri is selected from the group consisting of Cs-uaryl, C1-6 alkyl, C1-6 haloalkyl, and C5-14 aralkyl groups;
- R7-R14 are independently H, C1-6 alkoxy, halo, C1-4 alkyl, or R7 or R8-R9 or R9-R10 or R11-R12 or R12-R13 or R13-R14, can be joined together as a carbocyclic or heterocyclic ring system comprising at least one 5 or 6-membered ring;
- R15 is C1-6 alkyl; ach M is independently selected from the group consisting of H, an alkali metal, alkaline earth metal, transition metal, ammonium, phosphonium, organic amine salt, and an amino acid salt;
- Z is O or S; and n is 0, 1, or 2; a cationic aromatic compound (CAC); a background reducing agent; and an ether-linked nonionic surfactant or a hydrophilic polymer.
- CAC cationic aromatic compound
- the affinity molecule is selected from the group consisting of an antibody, a monoclonal antibody, a polyclonal antibody, a synthetic antibody mimic, an aptamer, an affimer, DARPins, or oligonucleotides or peptides that bind to at least one epitope of p-tau217.
- the detection molecule is an alkaline phosphatase (AP)-conjugated secondary antibody.
- the affinity molecule is a capture antibody and the detection molecule is a detection antibody wherein the method further comprises exposing the biological sample to the capture antibody which binds to a first p-tau217 epitope and the detection antibody which binds to a second p-tau217 epitope.
- the detector arrangement comprises a light detector configured to sense photons emitted from assay reactions over a period of time, an analog circuit configured to provide an analog signal based on the photons emitted from the assay reactions over the period of time, and a counter circuit configured to provide a photon count based on the photons emitted from the assay reactions over the period of time.
- the immunoassay analyzer further comprises a washing arrangement, wherein the washing arrangement is configured to perform at least one wash action to wash away at least a portion of unreacted components, alternatively configured to perform at least two wash actions, alternatively configured to perform at least three wash actions, alternatively configured to perform at least four wash actions, or alternatively configured to perform at least five wash actions.
- the immunoassay analyzer further comprises: a machine vision apparatus comprising an image capture device and an image interpretation device configured to monitor instrument and/or assay functionalities of the immunoassay analyzer.
- any one of items 1 to 31, wherein the method further comprises detecting at least one additional biomarker is selected from the group consisting of Ap peptides, Neurofilament light chain (NfL), total tau (t-tau), p-tau231, p-taul81, and combinations thereof.
- reagent pack further comprises containment walls arranged between the reagent vessels.
- the immunoassay analyzer further comprises a reagent storage unit, wherein the reagent pack is housed in the reagent storage unit.
- a method for detecting phosphorylated tau (p-tau)217 in a biological sample using an immunoassay analyzer comprising: aspirating, using a sample pipettor, a portion of the biological sample from a sample vessel and dispensing the aspirated sample into a reaction vessel of the immunoassay analyzer; wherein the immunoassay analyzer comprises: a reagent pack comprising a plurality of reagent vessels, wherein the reagent pack is configured to store a volume of reagent required for at least about 20 instances of a p- tau217 assay; a pipettor arrangement comprising at least one reagent pipettor and at a sample pipettor; and a detector arrangement; aspirating, using the reagent pipettor, a portion
- cycle time is about 45 seconds or less, alternatively about 40 seconds or less, alternatively about 35 seconds or less, alternatively about 30 seconds or less, alternatively about 25 seconds or less, alternatively about 20 seconds or less, or alternatively about 15 seconds.
- time to first result is about 60 minutes or less, alternatively about 55 minutes or less, alternatively about 50 minutes or less, alternatively about 45 minutes or less, alternatively about 40 minutes or less, alternatively about 35 minutes or less, alternatively about 30 minutes or less, alternatively about 25 minutes or less, alternatively about 20 minutes or less, alternatively about 15 minutes or less, or alternatively about 10 minutes or less.
- the substrate formulation is configured to produce chemiluminescence and comprises: a chemiluminescent compound of formula I or a salt thereof: wherein
- A is Ci-ehaloalkyl, naphthyl, phenyl, substituted phenyl, or heteroaryl, wherein substituted phenyl comprises from 1 to 3 halo, Ci-6 alkyl, Ci-6 alkoxy, Ci-6 haloalkyl, C(O)Ris, CN or NO2 substituents;
- Ri is selected from the group consisting of Cs-uaryl, C1-6 alkyl, C1-6 haloalkyl, and C5-14 aralkyl groups;
- R7-R14 are independently H, C1-6 alkoxy, halo, C1-4 alkyl, or R7 or R8-R9 or R9-R10 or R11-R12 or R12-R13 or R13-R14, can be joined together as a carbocyclic or heterocyclic ring system comprising at least one 5 or 6-membered ring;
- R15 is C1-6 alkyl; each M is independently selected from the group consisting of H, an alkali metal, alkaline earth metal, transition metal, ammonium, phosphonium, organic amine salt, and an amino acid salt;
- Z is O or S; and n is 0, 1, or 2; a cationic aromatic compound (CAC); a background reducing agent; and an ether-linked nonionic surfactant or a hydrophilic polymer.
- CAC cationic aromatic compound
- the affinity molecule is selected from the group consisting of an antibody, a monoclonal antibody, a polyclonal antibody, a synthetic antibody mimic, an aptamer, an affimer, DARPins, or oligonucleotides or peptides that bind to at least one epitope of p-tau217.
- the affinity molecule is a capture antibody and the detection molecule is a detection antibody wherein the method further comprises exposing the biological sample to the capture antibody which binds to a first p-tau217 epitope and the detection antibody which binds to a second p-tau217 epitope.
- the detector arrangement comprises a light detector configured to sense photons emitted from assay reactions over a period of time, an analog circuit configured to provide an analog signal based on the photons emitted from the assay reactions over the period of time, and a counter circuit configured to provide a photon count based on the photons emitted from the assay reactions over the period of time.
- the immunoassay analyzer further comprises an ultrasonic mixing module.
- the first reaction mixture, second reaction mixture, and/or detection mixture is agitated via the ultrasonic mixing module.
- the immunoassay analyzer further comprises a washing arrangement, wherein the washing arrangement is configured to perform at least one wash action to wash away at least a portion of unreacted components, alternatively configured to perform at least two wash actions, alternatively configured to perform at least three wash actions, alternatively configured to perform at least four wash actions, or alternatively configured to perform at least five wash actions.
- washing arrangement is configured to wash away at least a portion of the unreacted components in the first reaction mixture, second reaction mixture, and/or detection mixture.
- the immunoassay analyzer further comprises: a machine vision apparatus comprising an image capture device and an image interpretation device configured to monitor instrument and/or assay functionalities of the immunoassay analyzer.
- instrument functionalities are selected from the group consisting of optical sensors, pressure sensors and thermistors.
- any one items 36 to 65 wherein the biological sample is serum, whole blood, plasma, and/or cerebral spinal fluid.
- the method further comprises detecting at least one additional biomarker is selected from the group consisting of Ap peptides, Neurofilament light chain (NfL), total tau (t-tau), p-tau231, p-taul81, and combinations thereof.
- a method for detecting p-tau217 in a biological sample using an immunoassay analyzer comprising: mixing the biological sample with a reagent comprising at least one affinity molecule that binds to at least one portion p-tau217, generating a first reaction mixture; mixing the first reaction mixture with a reagent comprising a detection molecule, generating a second reaction mixture; mixing the second reaction mixture with a substrate formulation, generating a detection mixture; wherein the immunoassay analyzer comprises an ultrasonic mixing module, and the first reaction mixture, second reaction mixture and/or detection mixture are agitated via the ultrasonic mixing module; detecting, using a detector arrangement, a reaction in the detection mixture; and determining a presence and/or concentration of p-tau217 in the biological sample based on the reaction detected in the detection mixture.
- the quantitation detection limit (LOQ) for measuring p-tau217 is equal to or less than about 3 pg/mL, alternatively equal to or less than about 2 pg/mL, equal to or less than about 1 pg/mL, equal to or less than about 0.6 pg/mL, and wherein the detected p-tau217 has a concentration above the LOQ.
- the detected p-tau217 has a concentration that is at least IX greater than the LOQ, alternatively at least 2X greater than the LOQ, or alternatively at least 3X greater than the LOQ.
- cycle time is about 45 seconds or less, alternatively about 40 seconds or less, alternatively about 35 seconds or less, alternatively about 30 seconds or less, alternatively about 25 seconds or less, alternatively about 20 seconds or less, or alternatively about 15 seconds.
- time to first result is about 60 minutes or less, alternatively about 55 minutes or less, alternatively about 50 minutes or less, alternatively about 45 minutes or less, alternatively about 40 minutes or less, alternatively about 35 minutes or less, alternatively about 30 minutes or less, alternatively about 25 minutes or less, alternatively about 20 minutes or less, alternatively about 15 minutes or less, or alternatively about 10 minutes or less.
- any one of items 69 to 76 wherein the first reagent comprises at least one affinity molecule configured to bind to at least one portion p-tau217, the second reagent comprises at least one detection molecule, and the substrate formulation is configured to produce chemiluminescence.
- the substrate formulation is configured to produce chemiluminescence and comprises: a chemiluminescent compound of formula I or a salt thereof: wherein
- A is Ci-ehaloalkyl, naphthyl, phenyl, substituted phenyl, or heteroaryl, wherein substituted phenyl comprises from 1 to 3 halo, Ci-6 alkyl, Ci-6 alkoxy, Ci-6 haloalkyl, C(O)Ris, CN or NO2 substituents;
- Ri is selected from the group consisting of Cs-uaryl, C1-6 alkyl, C1-6 haloalkyl, and C5-14 aralkyl groups;
- R7-R14 are independently H, C1-6 alkoxy, halo, C1-4 alkyl, or R7 or R8-R9 or R9-R10 or R11-R12 or R12-R13 or R13-R14, can be joined together as a carbocyclic or heterocyclic ring system comprising at least one 5 or 6-membered ring;
- R15 is C1-6 alkyl; ach M is independently selected from the group consisting of H, an alkali metal, alkaline earth metal, transition metal, ammonium, phosphonium, organic amine salt, and an amino acid salt;
- Z is O or S; and n is 0, 1, or 2; a cationic aromatic compound (CAC); a background reducing agent; and an ether-linked nonionic surfactant or a hydrophilic polymer.
- CAC cationic aromatic compound
- the affinity molecule is selected from the group consisting of an antibody, a monoclonal antibody, a polyclonal antibody, a synthetic antibody mimic, an aptamer, an affimer, DARPins, or oligonucleotides or peptides that bind to at least one epitope of p-tau217.
- the affinity molecule is a capture antibody and the detection molecule is a detection antibody wherein the method further comprises exposing the biological sample to the capture antibody which binds to a first p-tau217 epitope and the detection antibody which binds to a second p-tau217 epitope.
- the detector arrangement comprises a light detector configured to sense photons emitted from assay reactions over a period of time, an analog circuit configured to provide an analog signal based on the photons emitted from the assay reactions over the period of time, and a counter circuit configured to provide a photon count based on the photons emitted from the assay reactions over the period of time.
- the immunoassay analyzer further comprises a washing arrangement, wherein the washing arrangement is configured to perform at least one wash action to wash away at least a portion of unreacted components, alternatively configured to perform at least two wash actions, alternatively configured to perform at least three wash actions, alternatively configured to perform at least four wash actions, or alternatively configured to perform at least five wash actions.
- washing arrangement is configured to wash away at least a portion of the unreacted components in the first reaction mixture, second reaction mixture, and/or detection mixture.
- the immunoassay analyzer further comprises: a machine vision apparatus comprising an image capture device and an image interpretation device configured to monitor instrument and/or assay functionalities of the immunoassay analyzer.
- any one of items 69 to 98 wherein the biological sample is serum, whole blood, plasma, and/or cerebral spinal fluid.
- the method further comprises detecting at least one additional biomarker is selected from the group consisting of Ap peptides, Neurofilament light chain (NfL), total tau (t-tau), p-tau231, p-taul81, and combinations thereof.
- a method for quantitatively determining an amount of phosphorylated tau (p-tau)217 in at least one plasma sample using a high-throughput analyzer comprising: aspirating a portion of the at least one plasma sample from a sample vessel and dispensing the aspirated plasma sample into a first reaction vessel and second reaction vessel of an immunoassay analyzer, wherein the immunoassay analyzer further comprises: a pipettor arrangement comprising at least a first reagent pipettor, a second reagent pipettor, a third reagent pipettor and at least one sample pipettor; a plurality of reagent vessels; and a detector arrangement; aspirating, using the first reagent pipettor, a portion of a fluidic substance from a first reagent vessel and dispensing said fluidic substance into the first reaction vessel; aspirating, using the first reagent pipettor, a portion of a fluidic substance from a second reagent vessel and dispens
- any one of items 100 to 107 wherein the quantitation detection limit (LOQ) for measuring p-tau217 is equal to or less than about 3 pg/mL, alternatively equal to or less than about 2 pg/mL, equal to or less than about 1 pg/mL, equal to or less than about 0.6 pg/mL, and wherein the p-tau217 has a concentration above the LOQ.
- LOQ quantitation detection limit
- time to first result is about 45 minutes or less, alternatively about 40 minutes or less, alternatively about 35 minutes or less, alternatively about 30 minutes or less, alternatively about 25 minutes or less, alternatively about 20 minutes or less, alternatively about 15 minutes or less, or alternatively about 10 minutes or less.
- the immunoassay analyzer comprises a fourth reaction vessel and the pipettor arrangement comprises a fourth reagent pipettor, wherein the method further includes aspirating a portion of the at least one plasma sample from a sample vessel and dispensing the aspirated plasma sample into the fourth reaction vessel; aspirating, using the fourth reagent pipettor, a portion of a fluidic substance from a first reagent vessel and dispensing said fluidic substance into the fourth reaction vessel; aspirating, using the fourth reagent pipettor, a portion of a fluidic substance from a second reagent vessel and dispensing said fluidic substance into the fourth reaction vessel; dispensing a substrate into the fourth reaction vessel, generating a detection mixture; detecting, using the detector arrangement, a presence of a reaction in the detection mixture in the fourth reaction vessel; and quantitatively determining an amount of p-tau217 in the plasma based on the presence of the reaction in the detection mixture.
- cycle time is about 45 seconds or less, alternatively about 40 seconds or less, alternatively about 35 seconds or less, alternatively about 30 seconds or less, alternatively about 25 seconds or less, alternatively about 20 seconds or less, or alternatively about 15 seconds.
- time to first result is about 45 minutes or less, alternatively about 40 minutes or less, alternatively about 35 minutes or less, alternatively about 30 minutes or less, alternatively about 25 minutes or less, alternatively about 20 minutes or less, alternatively about 15 minutes or less, or alternatively about 10 minutes or less.
- the fluidic substance from the first reagent vessel comprises at least one affinity molecule configured to bind to at least one portion p-tau217
- the fluidic substance from the second reagent vessel comprises at least one detection molecule
- the fluidic substance from the third reagent vessel comprises a substrate formulation configured to produce chemiluminescence.
- the affinity molecule is selected from the group consisting of an antibody, a monoclonal antibody, a polyclonal antibody, a synthetic antibody mimic, an aptamer, an affimer, DARPins, or oligonucleotides or peptides that bind to at least one epitope of p-tau217.
- the affinity molecule is an antibody and the method further comprises exposing the plasma sample to a first antibody which binds to a first p- tau217 epitope and a second antibody which binds to a second p-tau217 epitope.
- reagent pack further comprises containment walls arranged between the reagent vessels.
- reagent vessels comprise an elastomeric self-sealing membrane.
- immunoassay analyzer further comprises a reagent storage unit, wherein the reagent pack is housed in the reagent storage unit.
- each reagent vessel is configured to store a volume of reagent required for at least about 20 instances of a p-tau217 assay.
- the immunoassay analyzer further comprises: a machine vision apparatus comprising an image capture device and an image interpretation device configured to monitor instrument and/or assay functionalities of the immunoassay analyzer.
- the immunoassay analyzer further comprises a washing arrangement, wherein the washing arrangement is configured to perform at least one wash action to wash away at least a portion of unreacted components, alternatively configured to perform at least two wash actions, alternatively configured to perform at least three wash actions, alternatively configured to perform at least four wash actions, or alternatively configured to perform at least five wash actions.
- washing arrangement is configured to wash away at least a portion of the unreacted components in the first reaction mixture, second reaction mixture, and/or detection mixture.
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Engineering & Computer Science (AREA)
- Immunology (AREA)
- Biomedical Technology (AREA)
- Chemical & Material Sciences (AREA)
- Analytical Chemistry (AREA)
- Physics & Mathematics (AREA)
- Pathology (AREA)
- Hematology (AREA)
- General Physics & Mathematics (AREA)
- General Health & Medical Sciences (AREA)
- Molecular Biology (AREA)
- Urology & Nephrology (AREA)
- Biochemistry (AREA)
- Microbiology (AREA)
- Medicinal Chemistry (AREA)
- Food Science & Technology (AREA)
- Neurology (AREA)
- Neurosurgery (AREA)
- Biotechnology (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Cell Biology (AREA)
- Investigating Or Analysing Biological Materials (AREA)
Abstract
The presently claimed and described technology provides methods for detecting phosphorylated tau (p-tau)217 in a biological sample using an immunoassay analyzer.
Description
AN AUTOMATED AND HIGH-TROUGHPUT ANALYZER METHOD FOR A PHOSPHORYLATED TAU (P-TAU) 217 IMMUNOASSAY
RELATED APPLICATIONS
[0001] The present patent application claims the priority benefit of U.S. Provisional Patent Application Ser. Nos. 63/596,578, filed November 6, 2023; 63/596,581, filed November 6, 2023; 63/607,742, filed December 8, 2023; 63/607,747, filed December 8, 2023; 63/642,601, filed May 3, 2024; 63/642,605, filed May 3, 2024; 63/676,889, filed July 29, 2024; and 63/676,890, filed July 29, 2024 the content of each is hereby incorporated by reference in its entirety into this disclosure.
INCORPORATION BY REFERENCE OF AN ELECTRONIC SEQUENCE LISTING
[0002] This application contains a Sequence listing that has been submitted in a computer readable format and is hereby incorporated by reference in its entirety. The computer readable file, created on November 4, 2024, is named 67541WO01_SequenceListing.xml and is 21,658 bytes in size.
BACKGROUND
[0003] Dementia is one of the costliest conditions to society. In 2022, the total national cost of caring for people living with Alzheimer’s Disease (AD) and other dementias was projected to reach $321 billion, and an estimated 6.5 million Americans aged 65 and older were estimated to be living with AD. Am JManag Care. 2022;28(suppl 10): S 188-S 196. By 2060, the number of people 65 and older with AD is projected to reach 13.8 million, barring the development of medical breakthroughs to prevent, slow or cure AD. Alzheimer ’s Association. 2022 Alzheimer’s Disease Facts and Figures. Alzheimers Dement 2022; 18. Dementia and AD are costly to society because affected individuals often require long-term care and support, and available treatments aim to manage symptoms and delay progression, rather than to reverse the underlying disease process. Accordingly, AD is impacting a growing population that is currently underserved with treatment options. Novel treatments for AD are emerging and driving an increased need for testing and diagnosis.
[0004] Tau phosphorylated at threonine 217 (“p-tau217”) has been proposed as a biomarker to aid in identifying patients having, or at risk of having, AD or neurodegenerative diseases, and to select patients for disease modifying treatments. p-tau217 measurements have been shown
to be capable of distinguishing between AD and non- AD patients. A need exists for a p-tau217 assay that is sensitive and specific enough to distinguish between AD and non-AD patients, including in combination with other biomarkers or other clinical information, to aid, physicians and neurologists in diagnosing patients with AD and other dementias.
BRIEF SUMMARY
[0005] A first aspect of the invention is a method for detecting phosphorylated tau (p-tau)217 in a biological sample using an immunoassay analyzer, the method comprising: aspirating, using a sample pipettor, a portion of the biological sample from a sample vessel and dispensing the aspirated biological sample into a reaction vessel of the immunoassay analyzer; wherein the immunoassay analyzer comprises: a reagent pack configured to hold a plurality of reagent vessels; a pipettor arrangement comprising at least one reagent pipettor and at least one sample pipettor; and a detector arrangement; aspirating, using the reagent pipettor, a portion of a first reagent from at least one reagent vessel and dispensing the aspirated reagent into the reaction vessel, generating a first reaction mixture; aspirating, using the reagent pipettor, a portion of a second reagent from at least one reagent vessel and dispensing the aspirated reagent into the reaction vessel, generating a second reaction mixture; dispensing a substrate formulation into the reaction vessel, generating a detection mixture; wherein the at least one sample pipettor is configured to aspirate and/or dispense less than about 10 pL, alternatively about 2.0 pL to about 9.9 pL; and detecting, using the detector arrangement, a reaction in the detection mixture; and determining a presence and/or concentration of p-tau217 in the biological sample based on the reaction detected in the detection mixture.
[0006] A second aspect of the invention is a method for detecting phosphorylated tau (p-tau)217 in a biological sample using an immunoassay analyzer, the method comprising: aspirating, using a sample pipettor, a portion of the biological sample from a sample vessel and dispensing the aspirated sample into a reaction vessel of the immunoassay analyzer; wherein the immunoassay analyzer comprises: a reagent pack comprising a plurality of reagent vessels, wherein the reagent pack is configured to store a volume of reagent required for at least about 20 instances of a p-tau217 assay; a pipettor arrangement comprising at least one reagent pipettor and at a sample pipettor; and a detector arrangement; aspirating, using the reagent pipettor, a portion of a first reagent from at least one reagent vessel and dispensing the aspirated reagent into a reaction vessel of the immunoassay analyzer, generating a first reaction mixture; aspirating, using the reagent pipettor, a portion of a second reagent from at least one reagent vessel and dispensing the aspirated reagent into the reaction vessel, generating a second reaction
mixture; dispensing a substrate formulation into the reaction vessel, generating a detection mixture; detecting, using the detector arrangement, a reaction in the detection mixture; and determining a presence and/or concentration of p-tau217 in the biological sample based on the reaction detected in the detection mixture.
[0007] A third aspect of the invention is a method for detecting p-tau217 in a biological sample using an immunoassay analyzer, the method comprising: mixing the biological sample with a reagent comprising at least one affinity molecule that binds to at least one portion p-tau217, generating a first reaction mixture; mixing the first reaction mixture with a reagent comprising a detection molecule, generating a second reaction mixture; mixing the second reaction mixture with a substrate formulation, generating a detection mixture; wherein the immunoassay analyzer comprises an ultrasonic mixing module, and the first reaction mixture, second reaction mixture and/or detection mixture are agitated via the ultrasonic mixing module; detecting, using a detector arrangement, a reaction in the detection mixture; and determining a presence and/or concentration of p-tau217 in the biological sample based on the reaction detected in the detection mixture.
[0008] In an aspect, the reaction in the detection mixture generates a chemiluminescent signal, and the method comprises correlating the chemiluminescent signal the presence and/or concentration of p-tau217 in the biological sample.
[0009] In an aspect, the quantitation detection limit (LOQ) for measuring p-tau217 is equal to or less than about 3 pg/mL, alternatively equal to or less than about 2 pg/mL, equal to or less than about 1 pg/mL, equal to or less than about 0.6 pg/mL, and wherein the detected p-tau217 has a concentration above the LOQ.
[0010] In an aspect, the detected p-tau217 has a concentration that is at least IX greater than the LOQ, alternatively at least 2X greater than the LOQ, or alternatively at least 3X greater than the LOQ.
[0011] In one or more of the above aspects, the detected p-tau217 exhibits a coefficient of variation (CV) of 20% or less, alternatively 15% or less, alternatively 10% or less, alternatively 5% or less, or alternatively 4% or less.
[0012] In one or more of the above aspects, cycle time is about 45 seconds or less, alternatively about 40 seconds or less, alternatively about 35 seconds or less, alternatively about 30 seconds or less, alternatively about 25 seconds or less, alternatively about 20 seconds or less, or alternatively about 15 seconds.
[0013] In one or more of the above aspects, time to first result (TTFR) is about 60 minutes or less, alternatively about 55 minutes or less, alternatively about 50 minutes or less, alternatively
about 45 minutes or less, alternatively about 40 minutes or less, alternatively about 35 minutes or less, alternatively about 30 minutes or less, alternatively about 25 minutes or less, alternatively about 20 minutes or less, alternatively about 15 minutes or less, or alternatively about 10 minutes or less.
[0014] In one or more of the above aspects, incubation time of the first reaction mixture is at least about 30 minutes, alternatively at least about 40 minutes, or alternatively at least about 50 minutes.
[0015] In one or more of the above aspects, incubation time of the second reaction mixture is at least about 5 minutes, alternatively at least about 8 minutes, or alternatively at least about 10 minutes.
[0016] In one or more of the above aspects, the first reagent comprises at least one affinity molecule configured to bind to at least one portion p-tau217, the second reagent comprises at least one detection molecule, and the substrate formulation is configured to produce chemiluminescence.
[0017] In one or more of the above aspects, the substrate formulation is configured to produce chemiluminescence and comprises:
[0020] wherein
[0021] A is Ci-ehaloalkyl, naphthyl, phenyl, substituted phenyl, or heteroaryl, wherein substituted phenyl comprises from 1 to 3 halo, Ci-6 alkyl, Ci-6 alkoxy, Ci-6 haloalkyl, C(O)Ri5, CN or NCh substituents;
[0022] Ri is selected from the group consisting of Cs-uaryl, Ci-6 alkyl, Ci-6 haloalkyl, and C>. 14 aralkyl groups;
[0023] R7-R14 are independently H, C1-6 alkoxy, halo, C1-4 alkyl, or R7 or R8-R9 or R9-R10 or R11-R12 or R12-R13 or R13-R14, can be joined together as a carbocyclic or heterocyclic ring system comprising at least one 5 or 6-membered ring;
[0024] RI5 is Ci-6 alkyl;
[0025] each M is independently selected from the group consisting of H, an alkali metal, alkaline earth metal, transition metal, ammonium, phosphonium, organic amine salt, and an amino acid salt;
[0026] Z is O or S; and
[0027] n is 0, 1, or 2;
[0028] a cationic aromatic compound (CAC);
[0029] a background reducing agent; and
[0030] an ether-linked nonionic surfactant or a hydrophilic polymer.
[0031] In one or more of the above aspects, the affinity molecule is selected from the group consisting of an antibody, a monoclonal antibody, a polyclonal antibody, a synthetic antibody mimic, an aptamer, an affimer, DARPins, or oligonucleotides or peptides that bind to at least one epitope of p-tau217.
[0032] In one or more of the above aspects, the method further comprises exposing the biological sample to a first antibody which binds to a first p-tau217 epitope and a second antibody which binds to a second p-tau217 epitope.
[0033] In one or more of the above aspects, the detection molecule is an alkaline phosphatase (AP)-conjugated secondary antibody.
[0034] In one or more of the above aspects, the affinity molecule is a capture antibody and the detection molecule is a detection antibody wherein the method further comprises exposing the biological sample to the capture antibody which binds to a first p-tau217 epitope and the detection antibody which binds to a second p-tau217 epitope.
[0035] In one or more of the above aspects, the method further comprises exposing the biological sample to a second capture antibody which binds to a third p-tau217 epitope.
[0036] In one or more of the above aspects, the detection molecule binds to an epitope on p- Tau217 comprising amino acids 1 - 25, 1 - 20, 6 - 18, or 7 - 20 of SEQ ID NO: 1.
[0037] In one or more of the above aspects, the detection molecule binds to an epitope on p- Tau217 having at least 75% identical to the amino acid sequence set forth in SEQ ID NOs: 2, 3, or 4.
[0038] In one or more of the above aspects, the affinity molecule binds to an epitope on p- Tau217 comprising amino acids 217, 210 - 225, or 210 - 220 of SEQ ID NO: 1.
[0039] In one or more of the above aspects, the affinity molecule binds to an epitope on p- Tau217 having at least 75% identical to the amino acid sequence set forth in SEQ ID NO: 4.
[0040] In one or more of the above aspects, the affinity molecule and/or the detection molecule is conjugated to a magnetic bead or a magnetic particle.
[0041] In one or more of the above aspects, the first reaction mixture, second reaction mixture, and/or detection mixture is subjected to a magnetic field prior to detection.
[0042] In one or more of the above aspects, the detector arrangement comprises a light detector configured to sense photons emitted from assay reactions over a period of time, an analog circuit configured to provide an analog signal based on the photons emitted from the assay reactions over the period of time, and a counter circuit configured to provide a photon count based on the photons emitted from the assay reactions over the period of time.
[0043] In one or more of the above aspects, the immunoassay analyzer further comprises an ultrasonic mixing module.
[0044] In one or more of the above aspects, the first reaction mixture, second reaction mixture, and/or detection mixture is agitated via the ultrasonic mixing module.
[0045] In one or more of the above aspects, the immunoassay analyzer further comprises a washing arrangement, wherein the washing arrangement is configured to perform at least one wash action to wash away at least a portion of unreacted components, alternatively configured to perform at least two wash actions, alternatively configured to perform at least three wash actions, alternatively configured to perform at least four wash actions, or alternatively configured to perform at least five wash actions.
[0046] In one or more of the above aspects, the washing arrangement is configured to wash away at least a portion of the unreacted components in the first reaction mixture, second reaction mixture, and/or detection mixture.
[0047] In one or more of the above aspects, the immunoassay analyzer further comprises: a machine vision apparatus comprising an image capture device and an image interpretation device configured to monitor instrument and/or assay functionalities of the immunoassay analyzer.
[0048] In one or more of the above aspects, the instrument functionalities are selected from the group consisting of optical sensors, pressure sensors and thermistors.
[0049] In one or more of the above aspects, the assay functionalities are selected from the group consisting of sample volume monitoring, total reagent volume monitoring, residual volume monitoring, and particle retention monitoring.
[0050] In one or more of the above aspects, the biological sample is serum, whole blood, plasma, and/or cerebral spinal fluid.
[0051] In one or more of the above aspects, the method further comprises detecting at least one additional biomarker is selected from the group consisting of A|3 peptides, Neurofilament light chain (NfL), total tau (t-tau), p-tau231, p-taul81, and combinations thereof.
[0052] In one or more of the above aspects, the reagent vessels comprise an elastomeric selfsealing membrane.
[0053] In one or more of the above aspects, the reagent pack further comprises containment walls arranged between the reagent vessels.
[0054] In one or more of the above aspects, the immunoassay analyzer further comprises a reagent storage unit, wherein the reagent pack is housed in the reagent storage unit.
[0055] A fourth aspect of the invention is a method for quantitatively determining an amount of phosphorylated tau (p-tau)217 in at least one plasma sample using a high-throughput analyzer, the method comprising: aspirating a portion of the at least one plasma sample from a sample vessel and dispensing the aspirated plasma sample into a first reaction vessel and second reaction vessel of an immunoassay analyzer, wherein the immunoassay analyzer further comprises: a pipettor arrangement comprising at least a first reagent pipettor, a second reagent pipettor, a third reagent pipettor and at least one sample pipettor; a plurality of reagent vessels; and a detector arrangement; aspirating, using the first reagent pipettor, a portion of a fluidic substance from a first reagent vessel and dispensing said fluidic substance into the first reaction vessel; aspirating, using the first reagent pipettor, a portion of a fluidic substance from a second reagent vessel and dispensing said fluidic substance into the first reaction vessel; dispensing a substrate into the first reaction vessel, generating a detection mixture; detecting, using the detector arrangement, a presence of a reaction in the detection; and quantitatively determining an amount of p-tau217 in the plasma based on the presence of the reaction in the detection mixtures; wherein the second reagent pipettor and third reagent pipettor are configured to dispense reagents into second and third reaction vessels, simultaneously, as the first reagent pipettor dispenses fluid into the first reaction vessel.
[0056] In an embodiment of the fourth aspect of the invention, the method is configured to analyze at least about 200 plasma samples/hr.
[0057] In an embodiment of the fourth aspect of the invention, the method is configured to analyze at least about 300 plasma samples/hr.
[0058] In an embodiment of the fourth aspect of the invention, the method is configured to analyze at least about 400 plasma samples/hr.
[0059] In an embodiment of the fourth aspect of the invention, the reaction in the detection mixture generates a chemiluminescent signal, wherein the chemiluminescent signal quantitatively corresponds to a concentration of p-tau217 in the plasma sample.
[0060] In an embodiment of the fourth aspect of the invention, the quantitation detection limit (LOQ) for measuring p-tau217 is equal to or less than about 3 pg/mL, alternatively equal to or less than about 2 pg/mL, equal to or less than about 1 pg/mL, equal to or less than about 0.6 pg/mL, and wherein the p-tau217 has a concentration above the LOQ.
[0061] In an embodiment of the fourth aspect of the invention, the quantitation detection limit (LOQ) for measuring p-tau217 is equal to or less than about 2 pg/mL.
[0062] In an embodiment of the fourth aspect of the invention, the quantitation detection limit (LOQ) for measuring p-tau217 is equal to or less than about 1 pg/mL.
[0063] In an embodiment of the fourth aspect of the invention, the quantitation detection limit (LOQ) for measuring p-tau217 is equal to or less than about 0.6 pg/mL, and wherein the p- tau217 has a concentration above the LOQ.
[0064] In an embodiment of the fourth aspect of the invention, the quantitation detection limit (LOQ) for measuring p-tau217 is equal to or less than about 50 fg/mL.
[0065] In an embodiment of the fourth aspect of the invention, the quantitation detection limit (LOQ) for measuring p-tau217 is equal to or less than about 15 fg/mL.
[0066] In an embodiment of the fourth aspect of the invention, the quantitation detection limit (LOQ) for measuring p-tau217 is equal to or less than about 10 fg/mL, and wherein the p-tau217 has a concentration above the LOQ.
[0067] In an embodiment of the fourth aspect of the invention, the detected p-tau217 has a concentration that is at least IX greater than the LOQ, alternatively at least 2X greater than the LOQ, or alternatively at least 3X greater than the LOQ.
[0068] In an embodiment of the fourth aspect of the invention, the p-tau217 exhibits a coefficient of variation (CV) of 20% or less.
[0069] In an embodiment of the fourth aspect of the invention, the p-tau217 exhibits a coefficient of variation (CV) of 4% or less.
[0070] In an embodiment of the fourth aspect of the invention, the immunoassay analyzer comprises a fourth reaction vessel and the pipettor arrangement comprises a fourth reagent pipettor, wherein the method further comprises aspirating a portion of the at least one plasma sample from a sample vessel and dispensing the aspirated plasma sample into the fourth reaction vessel; aspirating, using the fourth reagent pipettor, a portion of a fluidic substance from a first reagent vessel and dispensing said fluidic substance into the fourth reaction vessel; aspirating,
using the fourth reagent pipettor, a portion of a fluidic substance from a second reagent vessel and dispensing said fluidic substance into the fourth reaction vessel; dispensing a substrate into the fourth reaction vessel, generating a detection mixture; detecting, using the detector arrangement, a presence of a reaction in the detection mixture in the fourth reaction vessel; and quantitatively determining an amount of p-tau217 in the plasma based on the presence of the reaction in the detection mixture.
[0071] In an embodiment of the fourth aspect of the invention, the first reagent pipettor, the second reagent pipettor, the third reagent pipettor and the fourth reagent pipettor are selectively and/or simultaneously operated.
[0072] In an embodiment of the fourth aspect of the invention, the fourth reagent pipettor is configured to engage a dispense tip prior to aspiration.
[0073] In an embodiment of the fourth aspect of the invention, cycle time is about 45 seconds or less, alternatively about 40 seconds or less, alternatively about 35 seconds or less, alternatively about 30 seconds or less, alternatively about 25 seconds or less, alternatively about 20 seconds or less, or alternatively about 15 seconds.
[0074] In an embodiment of the fourth aspect of the invention, time to first result (TFFR) is about 45 minutes or less, alternatively about 40 minutes or less, alternatively about 35 minutes or less, alternatively about 30 minutes or less, alternatively about 25 minutes or less, alternatively about 20 minutes or less, alternatively about 15 minutes or less, or alternatively about 10 minutes or less.
[0075] In an embodiment of the fourth aspect of the invention, dispensing the fluidic substance from the first reagent vessel into the first reaction vessel, second reaction vessel, third reaction vessel, and/or fourth reaction vessel generates a first reaction mixture and incubation time of the first reaction mixture is at least about 30 minutes, alternatively at least about 40 minutes, or alternatively at least about 50 minutes.
[0076] In an embodiment of the fourth aspect of the invention, the first reaction mixture in the first reaction vessel, second reaction vessel, third reaction vessel, and/or fourth reaction vessel are incubated simultaneously.
[0077] In an embodiment of the fourth aspect of the invention, dispensing the fluidic substance from the second reagent vessel into the first reaction vessel, second reaction vessel, third reaction vessel, and/or fourth reaction vessel generates a second reaction mixture and incubation time of the second reaction mixture is at least about 5 minutes, alternatively at least about 8 minutes, or alternatively at least about 10 minutes.
[0078] In an embodiment of the fourth aspect of the invention, the second reaction mixture in the first reaction vessel, second reaction vessel, third reaction vessel, and/or fourth reaction vessel are incubated simultaneously.
[0079] In an embodiment of the fourth aspect of the invention, the method is configured to simultaneously perform at least two assays for a plurality of plasma samples, alternatively at least three assays for a plurality of plasma samples, or at least four assays for a plurality of plasma samples.
[0080] In an embodiment of the fourth aspect of the invention, the plasma samples are from the same individual or from multiple individuals.
[0081] In an embodiment of the fourth aspect of the invention, the method comprises a plurality of sample vessels.
[0082] In an embodiment of the fourth aspect of the invention, at least one of the assays is p- tau217 assay, alternatively at least two of the assays are p-tau217 assays, alternatively at least three of the assays are p-tau217 assays, or alternatively at least four of the assays are p-tau217 assays.
[0083] In an embodiment of the fourth aspect of the invention, at least one of the assays is p- tau217 assay and at least one of the assays is a second analyte assay, wherein the second analyte is selected from the group consisting of A|3 peptides, Neurofilament light chain (NfL), total tau (t-tau), p-tau231, p-taul81, and combinations thereof.
[0084] In an embodiment of the fourth aspect of the invention, the fluidic substance from the second reagent vessel comprises at least one detection molecule, preferably the detection molecule is an alkaline phosphatase (AP)-conjugated secondary antibody.
[0085] In an embodiment of the fourth aspect of the invention, the affinity molecule is selected from the group consisting of an antibody, a monoclonal antibody, a polyclonal antibody, a synthetic antibody mimic, an aptamer, an affimer, DARPins, or oligonucleotides or peptides that bind to at least one epitope of p-tau217.
[0086] In an an embodiment of the fourth aspect of the invention, the affinity molecule is an antibody and the method further comprises exposing the plasma sample to a first antibody which binds to a first p-tau217 epitope and a second antibody which binds to a second p-tau217 epitope.
[0087] In an an embodiment of the fourth aspect of the invention, the detection molecule is an alkaline phosphatase (AP)-conjugated secondary antibody.
[0088] In an an embodiment of the fourth aspect of the invention, the affinity molecule and/or the detection molecule is conjugated to a magnetic bead or a magnetic particle.
[0089] In an embodiment of the fourth aspect of the invention, the first reagent vessel, second reagent vessel and/or third reagent vessel are housed in a reagent pack.
[0090] In an aspect, the reagent pack further comprises containment walls arranged between the reagent vessels.
[0091] In an embodiment of the fourth aspect of the invention, the reagent vessels comprise an elastomeric self-sealing membrane.
[0092] In an embodiment of the fourth aspect of the invention, the immunoassay analyzer further comprises a reagent storage unit, wherein the reagent pack is housed in the reagent storage unit.
[0093] In an embodiment of the fourth aspect of the invention, each reagent vessel is configured to store a volume of reagent required for at least about 20 instances of a p-tau217 assay.
[0094] In an embodiment of the fourth aspect of the invention, the immunoassay analyzer further comprises: a machine vision apparatus comprising an image capture device and an image interpretation device configured to monitor instrument and/or assay functionalities of the immunoassay analyzer.
[0095] In an embodiment of the fourth aspect of the invention, the instrument functionalities are selected from the group consisting of optical sensors, pressure sensors and thermistors.
[0096] In an embodiment of the fourth aspect of the invention, the assay functionalities are selected from the group consisting of sample volume monitoring, total reagent volume monitoring, residual volume monitoring, and particle retention monitoring.
[0097] In an embodiment of the fourth aspect of the invention, the immunoassay analyzer further comprises an ultrasonic mixing module.
[0098] In an embodiment of the fourth aspect of the invention, the first reaction vessel, second reaction vessel, third reaction vessel, and/or fourth reaction vessel are agitated via the ultrasonic mixing module.
[0099] In an embodiment of the fourth aspect of the invention, the immunoassay analyzer further comprises a washing arrangement, wherein the washing arrangement is configured to perform at least one wash action to wash away at least a portion of unreacted components, alternatively configured to perform at least two wash actions, alternatively configured to perform at least three wash actions, alternatively configured to perform at least four wash actions, or alternatively configured to perform at least five wash actions.
[0100] These and other advantages, aspects, and novel features of the present disclosure, as well as details of illustrated embodiments thereof, will be more fully understood from the following description and drawings.
BRIEF DESCRIPTION OF THE FIGURES
[0101] Embodiments of the present disclosure will now be described, by way of example only, with reference to the attached Figures, wherein:
[0102] FIG. 1 is a table showing the performance characteristics an exemplary immunoassay analyzer disclosed herein.
[0103] FIGs. 2A - 2C depict a method according to an aspect of the disclosure.
[0104] FIG. 3 is a calibration curve plot constructed using calibrator antigens of varying concentrations using an exemplary p-tau217 assay and an exemplary immunoassay analyzer as disclosed herein.
[0105] FIG. 4 is a box and whisker plot showing pTau217 concentrations associated with AD- positive versus normal samples analyzed using an exemplary p-tau217 assay and an exemplary immunoassay analyzer as disclosed herein.
[0106] FIG. 5 is a plot depicting within-run CV was calculated from harmonized dose response and plotted relative to mean dose-response of samples analyzed using an exemplary p-tau217 assay and an exemplary immunoassay analyzer as disclosed herein.
[0107] FIG. 6 is a graph depicting within-run precision profiles for samples analyzed using an exemplary p-tau217 assay, experimental p-tau217 antibody pairs, and an exemplary immunoassay analyzer as disclosed herein.
[0108] FIG. 7 is a graph depicting a comparison of the measured concentrations for Alzheimer’s Disease samples and normal/healthy cohort for samples analyzed using an exemplary p-tau217 assay, experimental p-tau217 antibody pairs, and an exemplary immunoassay analyzer as disclosed herein.
[0109] FIGs. 8A-8C are graphs depicting precision profiles for an experimental p-tau217 antibody pair and conventional antibody pairs analyzed using an exemplary p-tau217 assay, and an exemplary immunoassay analyzer as disclosed herein. FIG. 8A depicts %CV for the analyzed antibody pairs. FIG. 8B depicts within-run precision profiles for the analyzed antibody pairs. FIG. 8C depicts the measured concentrations for the analyzed antibody pairs for AD Patients and Normal patients.
[0110] FIG. 9 is a graph depicting a comparison of experimental p-tau217 antibody pairs and conventional p-tau217 antibody pairs using an exemplary p-tau217 assay and an exemplary immunoassay analyzer as disclosed herein.
[0111] FIG. 10 is a graph depicting discrimination of amyloid positive patient samples and negative patient samples analyzed using an exemplary p-Tau217 assay as disclosed herein.
DETAILED DESCRIPTION
[0112] I. Introduction
[0113] Tau is a microtubule-associated protein abundant in the axons of neurons and is thought to contribute to stabilization and formation of microtubules. Tau has six isoforms with different amino-terminal inserts and different numbers of tandem repeats near the carboxy terminus, ranging in length from 352 to 441 amino acids, and is thought to have over thirty different phosphorylation sites. The amino acid sequence of tau-441 is listed in Table 1.
0115] Alzheimer’s disease (AD) is a progressive neurodegenerative disorder that affects millions of individuals. Individuals with AD have amyloid accumulation in the brain which damages the synaptic function and forms neurofibrillary tangles. Hardy JA, et al Alzheimer's disease: the amyloid cascade hypothesis. Science. 1992;256: 184-5. Phosphorylated tau has been proposed as providing a more accurate identification of AD due to the presence of hyperphosphorylated tau in neurofibrillary tangles. Wattmo, C., et al. Cerebro-spinal fluid biomarker levels: phosphorylated tau (T) and total tau (N) as markers for rate of progression in Alzheimer’s disease. BMC Neurol 20, 10 (2020).
[0116] In particular, Tau phosphorylated at threonine 217 (p-tau217) has been shown to have diagnostic value in predicting Alzheimer’s Disease. In some instances, pTau 217 is a variant of any tau isoform in which the threonine residue 217 is phosphorylated. The bolded Thr in Table 1 is the phosphorylation site in p-tau217 in tau-441. Plasma levels of p-tau217 have been found to be clinically significant in an early, specific and accurate diagnosis of AD, including the manifestation and progression of AD. Telser, J et al Clin Chim Acta. 2022 Jun 1 :531 : 100-111.
[0117] Automated analyzers are commonly used in clinical chemistry, immunoassay, hematology, and other biological sampling and analyzing applications. Automated analytical equipment, such as automated analytical chemistry instruments, automated analytical immunoassay instruments, automated analytical hematology instruments, etc., can efficiently perform clinical analysis on a large number of samples, with multiple tests being run concurrently or within short time intervals. Automated analytical instruments are particularly well-suited for high-volume and mid-volume testing environments.
[0118] A need exists for an objective, standardized immunoassay method that employs an automated analyzer method that can screen a biological sample for p-tau217. The present invention fulfills a need in the art by providing methods for detecting phosphorylated tau (p- tau)217 in a biological sample using an immunoassay analyzer.
[0119] II. Definitions
[0120] Unless otherwise defined, all technical and scientific terms used herein have the same meaning as commonly understood by one of ordinary skill in the art to which the methods described herein belong. Any reference to standard methods (e.g., ASTM, TAPPI, AATCC, etc.) refers to the most recent available version of the method at the time of filing of this disclosure unless otherwise indicated.
[0121] For any method disclosed herein that includes discrete steps, the steps may be conducted in any feasible order. And, as appropriate, any combination of two or more steps may be conducted simultaneously.
[0122] All headings are for the convenience of the reader and should not be used to limit the meaning of the text that follows the heading, unless so specified.
[0123] The words "preferred" and "preferably" refer to embodiments of the invention that may afford certain benefits, under certain circumstances. However, other embodiments may also be preferred, under the same or other circumstances. Furthermore, the recitation of one or more preferred embodiments or aspects does not imply that other embodiments or aspects are not useful and is not intended to exclude other embodiments or aspects from the scope of the invention.
[0124] The term "comprises" and variations thereof do not have a limiting meaning where these terms appear in the description and claims. Such terms will be understood to imply the inclusion of a stated step or element or group of steps or elements but not the exclusion of any other step or element or group of steps or elements.
[0125] By "consisting of is meant including, and limited to, whatever follows the phrase "consisting of." Thus, the phrase "consisting of' indicates that the listed elements are required or mandatory, and that no other elements may be present. By "consisting essentially of is meant including any elements listed after the phrase, and limited to other elements that do not interfere with or contribute to the activity or action specified in the disclosure for the listed elements. Thus, the phrase "consisting essentially of indicates that the listed elements are required or mandatory, but that other elements are optional and may or may not be present depending upon whether or not they materially affect the activity or action of the listed elements.
[0126] The singular form "a", "an" and "the" include plural referents unless the context clearly dictates otherwise. These articles refer to one or to more than one (i.e., to at least one). As used herein, the term "or" is generally employed in its usual sense including "and/or" unless the content clearly dictates otherwise. The term "and/or" means any one or more of the items in the list joined by "and/or". As an example, "x and/or y" means any element of the three-element set {(x), (y), (x, y)}. In other words, "x and/or y" means "one or both of x and y". As another example, "x, y, and/or z" means any element of the seven-element set {(x), (y), (z), (x, y), (x, z), (y, z), (x, y, z)}. In other words, "x, y and/or z" means "one or more of x, y and z".
[0127] Where ranges are given, endpoints include all numbers subsumed within that range (e.g., 1 to 5 includes 1, 1.5, 2, 2.75, 3, 3.80, 4, 5, etc.). Furthermore, unless otherwise indicated or otherwise evident from the context and understanding of one of ordinary skill in the art, values that are expressed as ranges can assume any specific value or subrange within the stated ranges in different embodiments of the disclosure, to the tenth of the unit of the lower limit of the range, unless the context clearly dictates otherwise. Herein, "up to" a number (for example, up to 50) includes the number (for example, 50). The term "in the range" or "within a range" (and similar statements) includes the endpoints of the stated range.
[0128] Reference throughout this specification to "one aspect," "an aspect," "certain aspects," or "some aspects," "one embodiment," "an embodiment," "certain embodiment," or "some embodiment," etc., means that a particular feature, configuration, composition, or characteristic described in connection with the aspect is included in at least one aspect of the disclosure. Thus, the appearances of such phrases in various places throughout this specification are not necessarily referring to the same embodiment of the disclosure. Furthermore, the particular features, configurations, compositions, or characteristics may be combined in any suitable manner in one or more aspects.
[0129] Unless otherwise indicated, all numbers expressing quantities of components, molecular weights, and so forth used in the specification and claims are to be understood as being modified in all instances by the term "about." As used herein in connection with a measured quantity, the term "about" refers to that variation in the measured quantity as would be expected by the skilled artisan making the measurement and exercising a level of care commensurate with the objective of the measurement and the precision of the measuring equipment used. The term "about" as used in connection with a numerical value throughout the specification and the claims denotes an interval of accuracy, familiar and acceptable to a person skilled in the art. In general, such interval of accuracy is +/-10%. Accordingly, unless otherwise indicated to the contrary, the numerical parameters set forth in the specification and claims are approximations that may vary depending upon the desired properties sought to be obtained by the present invention. At the very least, and not as an attempt to limit the doctrine of equivalents to the scope of the claims, each numerical parameter should at least be construed in light of the number of reported significant digits and by applying ordinary rounding techniques.
[0130] Notwithstanding that the numerical ranges and parameters setting forth the broad scope of the invention are approximations, the numerical values set forth in the specific examples are reported as precisely as possible. All numerical values, however, inherently contain a range necessarily resulting from the standard deviation found in their respective testing measurements.
[0131] The term "exemplary" means serving as a non-limiting example, instance, or illustration. As utilized herein, the terms "e.g.," and "for example" set off lists of one or more non-limiting aspects, examples, instances, or illustrations.
[0132] As used herein, the term "substantially" refers to the qualitative condition of exhibiting total or near-total extent or degree of a characteristic or property of interest. Biological and chemical phenomena rarely, if ever, go to completion and/or proceed to completeness or achieve or avoid an absolute result. The term "substantially" is therefore used herein to capture the potential lack of completeness inherent in many biological and chemical phenomena. For example, "substantially" may refer to being within at least about 20%, alternatively at least about 10%, alternatively at least about 5% of a characteristic or property of interest.
[0133] The invention is defined in the claims. However, below is a non-exhaustive listing of non-limiting exemplary aspects. Any one or more of the features of these aspects may be combined with any one or more features of another example, embodiment, or aspect described herein.
[0134] III. Systems and Reagents for p-tau217 Assay
[0135] One aspect of the invention is a method for detecting phosphorylated tau (p-tau)217 in a biological sample using an immunoassay analyzer.
[0136] Another aspect of the invention is a method for quantitatively determining the amount of phosphorylated tau (p-tau)217 in at least one plasma sample using an immunoassay analyzer
[0137] The term "immunoassay" may be a laboratory method used to determine the amount of an analyte in a sample. It can be based on the interaction of antibodies with antigens, and because of the degree of selectivity for the analyte (either antigen or antibody), an immunoassay can be used to quantitatively determine very low concentrations of analyte in a test sample. An "immunoassay analyzer" can include an instrument on which immunoassays have been automated. Various immunoassay analyzers are commercially available including the Dxl® system (Beckman Coulter, CA).
[0138] In an aspect, an immunoassay analyzer disclosed herein includes the following basic structural and functional modules: a sample presentation unit, an analytic unit, an incubator station, a washing station, a read station, and reagent storage. In addition, the immunoassay analyzer may include a pipettor arrangement with at least one sample pipettor and at least one reagent pipettor, and at least one transport device. In an aspect, the transport device includes mechanisms, such as pick-and-place grippers, which are used to transport sample and reaction vessels among the various modules of the immunoassay analyzer.
[0139] The immunoassay analyzer may include a container carriage device which is configured to hold and carry the containers at various locations in the instrument so that the analytic unit, incubator station, wash station, and read station can use the containers in various manners. Examples of container carriage devices include vessel racks (e.g., a sample rack, a reagent rack, and a diluent rack), the sample presentation unit, vessel carriage units (e.g., a sample carriage unit, a reaction vessel carriage unit, and a reagent carriage unit), vessel transfer units (e.g., a sample transfer unit, a reagent transfer unit, an incubator transfer unit, and an reaction vessel transfer unit), and vessel holding plates or wheels (e.g., a sample wheel, an incubator, and a wash wheel) ), which are described herein.
[0140] The read station may include a detector arrangement. In an aspect, the detector arrangement may include a detector that is configured to detect light or luminescence, for example chemiluminescence. The detector may be a luminescence detector, a chemiluminescence detector, a luminometer, or a photomultiplier-based detection instrument. In an embodiment, the detector includes a light detector configured to sense photons emitted
from assay reactions over a period of time, an analog circuit configured to provide an analog signal based on the photons emitted from the assay reactions over the period of time, and a counter circuit configured to provide a photon count based on the photons emitted from the assay reactions over the period of time. U.S. Patent No. 11,604,146, which is incorporated by reference in its entirety herein, discloses non-limiting examples of a detector that may be used in an aspect of the invention.
[0141] In an aspect, primary sample containers can be placed into an onload section (e.g., individually or on racks) of the sample presentation unit. In certain embodiments, the sample presentation unit has at least one sample rack, alternatively at least two sample racks, alternatively at least three sample racks, alternatively at least four sample racks, alternatively at least five sample racks, alternatively at least six sample racks, alternatively at least seven sample racks, alternatively at least eight sample racks, alternatively at least nine sample racks, alternatively at least ten sample racks, alternatively at least 11 sample racks, alternatively at least 12 sample racks, alternatively at least 13 sample racks, alternatively at least 14 sample racks, alternatively at least 15 sample racks, alternatively at least 16 sample racks, alternatively at least 17 sample racks, alternatively at least 18 sample racks, alternatively at least 19 sample racks, or alternatively at least 20 sample racks. In certain embodiments, each sample rack can hold at least one sample vessel, alternatively at least two sample vessels, alternatively at least three sample vessels, alternatively at least four sample vessels, alternatively at least five sample vessels, alternatively at least six sample vessels, or alternatively at least seven sample vessels. In a specific embodiment, the sample presentation unit holds about 140 sample vessels. In a specific embodiment, the sample presentation unit has 20 sample racks with each rack holding seven sample vessels.
[0142] In certain embodiments, the sample vessels include a barcode label that uniquely identifies the sample vessel in the immunoassay analyzer. The barcode label also may include alphanumeric characters that correspond to the barcode identification information. The immunoassay analyzer may include at least optical reader, such as a barcode scanner. In one embodiment, the optical readers are area scan cameras that provide a two-dimensional image of the barcode and/or sample vessel. In another embodiment, optical readers are area scan cameras that provide a three-dimensional image of the barcode and/or sample vessel.
[0143] After being fed into the onload section, the sample containers may be moved into a presentation section of the sample presentation unit. In certain embodiments, the sample presentation unit is maintained at between about 4.5°C to 14°C. The transfer station may receive
primary sample containers from the sample presentation unit from a transport device. Primary sample containers delivered to the transfer station from the transport device may be initially processed in different ways. For example, the primary sample container may temporarily remain at the transfer station while the sample provided within is aspirated by the sample pipettor at the transfer station. Following such aspiration, the primary sample container may be expelled from the automated analyzer at the offload station of the sample presentation unit, or may be passed on to the transport device for further processing. The instrument may also contain a sample retention unit configured to receive sample retention vessels and store, analyze, or otherwise process samples retained within the sample retention vessels. A “primary sample container” loaded into the automated analyzer with a sample may also be considered a “sample retention vessel” when placed in a sample retention unit, such as a storage unit. As used herein, “containers” or “vessels” are analogous, and can be of various types, such as specimen tubes (also referred to herein as sample tubes) and pipettor tips, such as micro or disposable tips. In certain embodiments, the vessels are tubes with diameters between about 12mm to about 16mm and/or heights between about 75mm to about 100mm. In certain embodiments, the vessels are cups with volumes of about 0.5mL, alternatively about l.OmL, alternatively about 1.5mL, alternatively about 2.0mL, alternatively about 2.5mL, or alternatively about 3.0mL.
[0144] In an aspect, the analytic unit is configured to receive and analyze samples. In an aspect, the analytic unit configured to perform an immunoassay. In certain embodiments, the analytic unit includes the pipettor arrangement. The pipettor arrangement may be configured to aliquot, aspirate, and dispense fluidic substances into various vessels, including, but not limited to sample vessels, diluent vessels, reagent vessels, and reaction vessels. Fluidic substances are substances that have fluidic characteristics. In some embodiments, the fluidic substance is a single fluidic substance. In other embodiments, the fluidic substance is a mixture of a plurality of substances.
[0145] The pipettor arrangement may contain at least one, two, three, or four reagent pipettors used to mix reagents with sample aliquots for an assay. In an aspect, the reagent pipettors may be arranged as dual reagent pipetting stations and are independent to each other, each having its own fluid pumps and valves, wash towers, reaction vessel carriages, and pipettor. A sample aliquot may be transferred from a sample retention vessel into a reaction vessel using the sample pipettor in order to mix the sample aliquot with one or more reagents. In an aspect, the at least one reagent pipettor and at least one sample pipettor are configured to aspirate and/or dispense less than about 10 pL. In an embodiment, the at least one reagent pipettor and at least one
sample pipettor are configured to aspirate and/or dispense less than about 9.9 pL, alternatively less than about 9.5 pL, alternatively less than about 8.0 pL, alternatively less than about 7.0 pL, alternatively less than about 6.0 pL, alternatively less than about 5.0 pL, alternatively less than about 4.0 pL, alternatively less than about 3.0 pL, alternatively less than about 2.0 pL, alternatively between than about 9.9 pL and 2.0 pL.
[0146] In an aspect, the immunoassay analyzer includes a reagent pack configured to hold a plurality of reagent vessels. A “reagent pack” may include any suitable container that can store a reagent. An example of a reagent pack can include a generally rectangular elongated body formed to include multiple reagent vessels including one or more large reagent vessels, and one or more relatively smaller reagent vessels, as well as features to facilitate handling and automation. US Patent No. 9,519,000, which is incorporated by reference in its entirety herein, discloses non-limiting examples of a reagent pack that may be used in an aspect of the invention.
[0147] In an embodiment, the reagent pack may be configured to accommodate sufficient volumes of reagents for multiple instances of an assay. In some embodiments, each reagent pack includes reagents for about 20 to about 100 instances of an assay and in some cases about 50 instances.
[0148] In some embodiments a reagent pack may supplied with empty or partially filled reagent vessels, to which reagents are subsequently transferred from bulk containers, such as bottles. Individual reagent vessels may differ in dimension to accommodate the requirements of an assay type. Factors that can determine the size of a reagent vessel include the number of uses desired for the reagent pack type, concentration-dependent stability issues with reagent components, and the need to minimize the volume of the final reaction mixture. As noted above, in some embodiments, each reagent pack can include a large reagent vessel and a plurality of small reagent vessels. Each reagent vessel may be large enough to accommodate a microtip or disposable tip (i.e., dispo-tip) of a reagent pipettor used to remove a volume of reagent for use in an assay. In certain embodiments, the reagent pack may be maintained at a temperature of between about 4°C to 10°C.
[0149] A “reagent vessel” may refer to a vessel, unit, fluid container, or the like that is configured to store reagents. In an embodiment, the reagent vessels include an elastomeric selfsealing membrane. An elastomeric self-sealing membrane may be a polymer, such as polypropylene, which is able to regain its original shape when pierced. For some embodiments, the elastomeric membrane can be a thermoplastic elastomer with hardness of 30-40 durometer (Shore) A. In other embodiments, the hardness can be 20-50 (Shore) A, or about 30 (Shore) A.
Elastomers deform sufficiently to form a tight seal with the vessel base. Thermoplastic elastomers are advantageous because of their compatibility with plastics injection molding processes.
[0150] The elastomeric membrane can be large enough to provide adequate compression without bottoming on the sealing portion of the vessel. The hardness and dimensions can cooperate to allow the elastomeric membrane to the sealing portion with reasonable sealing force. In some embodiments, the elastomeric membrane diameter is small enough so that, when compressed by engagement of the pipettor tip, it conforms to the sealing portion without contacting the wall of the pipettor tip. This advantageously concentrates sealing force to the sealing portion of vessel and distributes sealing force evenly to prevent leaks. In some embodiments, the sealing force is about 44 newtons (about 9.9 lbs) and produces a pressure on the sealing surface of about 300 (about 43.5 pounds per square inch) to about 1000 kPa (145.0 pounds per square inch).
[0151] In methods where multiple samples are analyzed, the elastomeric self-sealing membrane allows for aspiration of the reagents without concern of evaporation. In some embodiments, the reagent pack further includes containment walls arranged between the reagent vessels. In an aspect, the reagent pack may include at least one reagent vessel, alternatively at least two reagent vessels, alternatively at least three reagent vessels, alternatively at least four reagent vessels, alternatively at least five reagent vessels, or alternatively at least ten reagent vessels. In an embodiment, the immunoassay analyzer further includes a reagent storage unit, wherein the reagent pack is housed in the reagent storage unit.
[0152] At operation, a fluidic substance is dispensed to a reaction vessel. Examples of the fluidic substance include a sample, diluent, reagent, substrate, or any combination thereof, as described herein. In some embodiments, the reaction vessel already contains other fluidic substances, such as a sample, and after a fluidic substance is dispensed to a reaction vessel, the fluidic substance is mixed with the other fluidic substances in the reaction vessel. The mixing can be performed with a stirrer in direct contact with the fluidic substances, an ultrasonic probe in direct or indirect contact with the fluidic substances, or any other suitable mixing apparatus. In some aspects, the immunoassay analyzer includes an ultrasonic mixing module. For instance, a reagent pipettor may be outfitted with a tip that allows it to perform ultrasonic mixing of a reagent in a reagent pack before aspirating it for transport to a reaction vessel, thereby ensuring that the aspirated reagent would not be impacted by any settling that may have taken place in the reagent pack. Sample pipettors may similarly be specialized.
[0153] The transfer unit transfers the reaction vessels to and from the incubator station which includes an incubator. In some embodiments, the transfer unit transfers one or more of the pipetted reaction vessels from the reagent carriage unit to the incubator. Further, the transfer unit can transfer one or more reaction vessels from the incubator to the reagent carriage unit. The transfer unit can also remove from the reaction vessels that have been read or completed the incubator.
[0154] The incubator is thermally controlled to maintain a predetermined temperature. In some embodiments, the incubator is maintained about 30 °C to 40 °C. In other embodiments, the incubator is maintained about 37 °C to ensure immunological reaction and enzyme reaction, for example. By way of example, the incubator performs assay incubation.
[0155] In some embodiments, the transfer unit transfers incubated reaction vessels from the incubator to the wash unit, transfers assay reaction vessels from the wash unit to the incubator, transfers reaction vessels containing substrate from the wash unit to the incubator for substrate incubation or enzyme reaction, transfers washed reaction vessels from the incubator to detector arrangement after substrate incubation, and transfers the reaction vessels that have been read or completed from the detector arrangement to the incubator. The used reaction vessels can be delivered to a waste location.
[0156] At operation, a sample and a reagent are dispensed into a reaction vessel and mixed. The mixture is then transferred to the incubator. During the incubation, the sample and the reagent interact. The resulting “first reaction mixture” is a result of the incubation between the sample and the reagent. The reagent may include a specific-binding reagent, such as an affinity molecule specific to the analyte being analyzed by the immunoassay analyzer. In a non-limiting example, the incubation time of the first reaction mixture is at least about 30 minutes, alternatively at least about 40 minutes, alternatively at least about 50 minutes, alternatively at least about 55 minutes, or alternatively at least about 60 minutes.
[0157] At operation, a reagent, which is different from the reagent used in the first reaction mixture, is added to the first reaction mixture and mixed. The mixture is then transferred to the incubator. During incubation, the first reaction mixture and the reagent interact. The resulting “second reaction mixture” is a result of an incubation between the first reaction mixture and the reagent. The reagent may include a detection molecule, such as an alkaline phosphatase (AP)- conjugated secondary antibody, or a labeled antibody. In a non-limiting example, the incubation time of the second reaction mixture is at least about 5 minutes, alternatively at least about 8 minutes, or alternatively at least about 10 minutes.
[0158] The wash station receives and supports reaction vessels thereon such that various aspects of diagnostic process are performed with the immunoassay analyzer. In an embodiment, the wash station is configured to wash away at least some of the unreacted components. Unreacted components may include unreacted reagents (e.g., free antigens, antibodies, unbound reactants, particles, and/or fluid, etc.) and unreacted sample. The wash station may be configured to perform a set number of wash actions depending on the assay. The wash station may also be configured to perform a set number of washes within a predetermined sequence. In certain embodiments, the wash station is configured to perform at least one wash action to wash away at least a portion of unreacted components, alternatively configured to perform at least two wash actions, alternatively configured to perform at least three wash actions, alternatively configured to perform at least four wash actions, or alternatively configured to perform at least five wash actions. In some embodiments, the wash station is a thermally controlled device to separate bound or free analytes from particles after incubation. In some embodiments, the wash unit is maintained about 30 °C to 40 °C. In other embodiments, the wash unit is maintained about 37 °C to ensure enzyme reaction, for example. U.S. Patent Publication No. 2022/0357352, which is incorporated by reference in its entirety herein, discloses configurable wash processes according to an aspect of the invention.
[0159] The wash station may comprise a washing arrangement which may be configured to provide a base number of wash series (or wash actions) for each reaction vessel and optionally provide an additional number(s) of wash actions. The additional number(s) of wash actions may include one, a plurality, or all of a potential number of wash actions. An additional number of wash actions beyond the base number of wash actions may be specified for certain assays in an assay protocol file. The washing arrangement may include cleaning dispense nozzle (or probe) which dispenses a rinsing fluid and a cleaning aspiration nozzle (or probe) which aspirates the unreacted components. At operation, a base number of wash actions performed may be three, and the additional number of wash action(s) may be one or two. In this embodiment, the base number of wash actions may be performed if three probes dispense buffer solution once per vessel and the three probes aspirate the at least some of the unreacted components some of the buffer solution, and/or the at least some of the unreacted reagents once per vessel. According to the principles of the present disclosure, certain probe(s) may be selectively used to dispense clean buffer solution into the vessel and aspirate the at least some of the unreacted components of the sample, some of the buffer solution, and/or the at least some of the unreacted reagents from the vessel to perform the additional wash action(s).
[0160] In an aspect, the affinity molecule and/or the detection molecule is conjugated to a magnetic bead or a magnetic particle. Magnetic beads (also known as magnetic particles, paramagnetic particles, or superparamagnetic particles) consist of a polystyrene core surrounded by a thin layer of small iron oxide particles (-20-30 nm), such as magnetite. On the surface, the magnetic beads are encapsulated by, for example, a polymer, protein A, protein G, protein L, a secondary antibody, or an epoxy. Surface modification of the coating minimizes any non-specific protein binding. Antibodies targeting the analyte of interest can be covalently coupled to the surface of the magnetic bead. In some embodiments, there is about 2 pg or antibody per mg of magnetic bead. In some embodiments, there is about 3 pg or antibody per mg of magnetic bead. In some embodiments, there is about 4 pg or antibody per mg of magnetic bead. In some embodiments, there is about 5 pg or antibody per mg of magnetic bead. In some embodiments, there is about 6 pg or antibody per mg of magnetic bead. In some embodiments, there is about 7 pg or antibody per mg of magnetic bead. In some embodiments, there is about 8 pg or antibody per mg of magnetic bead. In some embodiments, there is about 9 pg or antibody per mg of magnetic bead. In some embodiments, there is about 10 pg or antibody per mg of magnetic bead. In some embodiments, there is about 12 pg or antibody per mg of magnetic bead. In some embodiments, there is about 13 pg or antibody per mg of magnetic bead. In some embodiments, there is about 14 pg or antibody per mg of magnetic bead. In some embodiments, there is about 15 pg or antibody per mg of magnetic bead.
[0161] In an embodiment, the first reaction mixture or second reaction mixture is subjected to a magnetic field. The magnetic beads do not exhibit bead-to-bead attraction, only migrating when a magnetic field is applies. Captured analytes or targets are separated from the mixture and magnetization may be used to retain desired components within a reaction vessel.
[0162] At operation, the vessel containing the first reaction mixture is moved near one or more magnets. The one or more magnets attract the magnetic bead(s) to one or more sides of the reaction vessel. The reaction vessel is then subject to a wash process in which a cleaning dispense nozzle dispenses a rinsing fluid and a cleaning aspiration nozzle aspirates the unreacted components. The aspiration nozzle may be washed with a probe washer before and/or after the aspirating. The reaction vessel may undergo a series of wash process which may include at least two series of dispensing the rinsing fluid and aspirating the uncollected fluid components, alternatively at least three series, alternatively at least four series, alternatively at least five series. As a result, an unreacted substance or substances in the vessel is removed (e.g., rinsed away) by the bound-free cleaning aspiration nozzle.
[0163] At operation, the vessel containing the second reaction mixture is moved near one or more magnets. The one or more magnets attract the magnetic bead(s) or magnetic particle(s) to one or more sides of the reaction vessel. The reaction vessel is then subject to a wash process in which a cleaning dispense nozzle dispenses a rinsing fluid and a cleaning aspiration nozzle aspirates the unreacted components. The aspiration nozzle may be washed with a probe washer before and/or after the aspirating. The reaction vessel may undergo a series of wash process which may include at least two series of dispensing the rinsing fluid and aspirating the uncollected fluid components, alternatively at least three series, alternatively at least four series, alternatively at least five series. As a result, an unreacted substance or substances in the vessel is removed (e.g., rinsed away) by the bound-free cleaning aspiration nozzle.
[0164] At operation, a substrate is dispensed into the second reaction mixture and mixed. After a certain reaction time necessary for the substrate and the second reaction mixture to interact. The reaction vessel is then subject to a wash process in which a cleaning dispense nozzle dispenses a rinsing fluid and a cleaning aspiration nozzle aspirates the unreacted components. The aspiration nozzle may be washed with a probe washer before and/or after the aspirating. The reaction vessel may undergo a series of wash process which may include at least two series of dispensing the rinsing fluid and aspirating the uncollected fluid components, alternatively at least three series, alternatively at least four series, alternatively at least five series. As a result, an unreacted substance or substances in the vessel is removed (e.g., rinsed away) by the bound- free cleaning aspiration nozzle. The resulting detection mixture is transferred to the detector arrangement.
[0165] Assays using substrates including features and/or characteristics described herein may benefit from one or more additional number(s) of wash actions. These “two-site” or “sandwich” immunoassays employ a first antibody or antibody fragment, which is described as the “capture” antibody, is bound to a solid support, such as magnetic beads or particles disclosed herein, using procedures known in the art. Further, a second antibody or antibody fragment, which is described as the “detection” antibody, is coupled or conjugated with a label, such as the enzymes disclosed herein, using procedures known in the art. The label produces a detectable signal when provided with substrate(s), so that the amount of signal measured corresponds to the amount of detection antibody that is bound to the analyte.
[0166] In an aspect, the substrate formulation is configured to produce chemiluminescence. These substrates can produce light and thereby provide detection corresponding to a quantity of analytes captured. The term “chemiluminescent compound” refers to a compound that
produces chemiluminescence in the presence of a phosphatase enzyme and oxygen under appropriate conditions as provided herein. In a non-limiting example, the substrate formulation includes a chemiluminescent compound of formula I or a salt thereof:
[0168] wherein
[0169] A is Ci-6haloalkyl, naphthyl, phenyl, substituted phenyl, or heteroaryl, wherein substituted phenyl comprises from 1 to 3 halo, Ci-6 alkyl, Ci-6 alkoxy, Ci-6 haloalkyl, C(0)Ri5, CN or NO2 substituents;
[0170] Ri is selected from the group consisting of Cs-naryl, Ci-6 alkyl, Ci -6 haloalkyl, and C5- 14 aralkyl groups;
[0171] R7-R14 are independently H, Ci-6 alkoxy, halo, Ci-4alkyl, or R7 -Rs or Rs -R9 or R9-
[0172] R10 R11-R12 or R12-R13 or R13-R14, can be joined together as a carbocyclic or heterocyclic ring system comprising at least one 5 or 6-membered ring;
[0173] Ri5 is Ci-6 alkyl;
[0174] each M is independently selected from H, or an alkali metal, alkaline earth metal, transition metal, ammonium, phosphonium, organic amine salt, amino acid salt or ;
[0175] Z is O or S; and
[0176] n is 0, 1, or 2;
[0177] a cationic aromatic compound (CAC);
[0178] a background reducing agent; and
[0179] an ether-linked nonionic surfactant or a hydrophilic polymer
[0180] Chemiluminescent compounds useful in the present formulations are capable of generating chemiluminescence when contacted with an alkaline phosphatase. Such compounds
can be synthesized as described in U.S. Pat. Nos. 6,45,727, 6,90,571, 6,139,782, 6,218,137, 6,270,695, 6,296,787, and 10,703,971 each of which is incorporated by reference herein.
[0181] At operation, the chemiluminescent substrate is added to the vessel with the second reaction mixture and light generated by the reaction is measured with a luminometer. The light production is inversely proportional to the concentration of free analyte in the sample. The amount of analyte in the sample is then determined from a stored, multi-point calibration curve. In an embodiment, the detector can generate an output signal that can be processed to generate a relative light unit (“RLU”) value (i.e., an output response) indicating a result of the assay. For example, a larger RLU value indicates more light, which indicates a larger amount of the analyte in the biological sample than a smaller RLU value indicates.
[0182] In an exemplary method of producing light from the reaction of the chemiluminescent substrate with a phosphatase enzyme (e.g., detection antibody), the reaction is performed at a temperature between 5° C and 50° C, preferably between 20° C and 40° C in an aqueous buffer solution at a pH between 7 and 12, 8 and 11, or preferably between 8.5 and 10. The enzyme is preferably an alkaline phosphatase or an alkaline phosphatase conjugate.
[0183] In one aspect, the substrate formulation comprises 0.01 mM-50 mM compound I, 0.01- 200 uM cationic aromatic compound, 1 uM-10 mM background reducing agent, 0.05-20 g/L ether-linked non-ionic surfactant or hydrophilic polymer, 0.01-10 g/L anionic surfactant, and an amine buffer at from 0.025M to 0.65M and at pH 7-12.
[0184] In one aspect, the substrate formulation comprises 0.05 mM-10 mM compound I, 0.05- 50 pM cationic aromatic compound, 10 uM-1000 uM background reducing agent, 0.1 to 10 g/L ether-linked non-ionic surfactant or hydrophilic polymer, 0.1 to 5 g/L anionic surfactant, and an amine buffer at from 0.05M to 0.5M and at pH 8-11.
[0185] In one aspect, the substrate formulation comprises 0.1 mM-5 mM compound I, 0.1-25 pM cationic aromatic compound, 50 to 500 uM background reducing agent, 0.2 to 5 g/L ether- linked non-ionic surfactant or hydrophilic polymer, 0.1 to 5 g/L anionic surfactant, and an amine buffer at from 0.1M-0.4M and at pH 8-11.
[0187]
[0188] In certain embodiments, the transport device includes three pick-and-place grippers, where a first pick-and-place gripper may be used to transport sample containers among the onload section, the transfer station, and reagent pipetting stations. A second pick-and-place gripper may be used to transport reaction vessels between the reagent pipetting stations and the incubator station or read station. A third pick-and-place gripper may be used to transport reaction vessels between the incubator station and the wash station or read station. A detailed description of the configurations and functions of one embodiment of the vessel pick-and-place grippers is provided in U.S. Patent No. 7128874 and is incorporated herein in its entirety by reference. However, it should be understood that other pick-and-place mechanism that are capable of transporting sample and reaction vessels among the various modules of the immunoassay analyzer is also contemplated for the purpose of the present invention.
[0189] In an aspect, the immunoassay analyzer includes a machine vision apparatus comprising an image capture device and an image interpretation device configured to monitor instrument and/or assay functionalities of the immunoassay analyzer. In some embodiments, instrument functionalities may include optical sensors, pressure sensors and thermistors. In some embodiments, the assay functionalities may include sample volume monitoring, total reagent volume monitoring, residual volume monitoring, and particle retention monitoring. The machine vision apparatus operates to evaluate the preparation of samples for subsequent analysis. In some embodiments, the machine vision apparatus utilizes one or more image capture units to determine whether samples have been appropriately prepared for analysis. As described herein, the machine vision apparatus provides direct and simple measurements of volume or integrity of a sample to determine whether the sample is appropriately prepared so that the analytic unit produces a reliable result. An exemplary machine vision apparatus is described in U.S. Patent No. 11,263,433, which is incorporated by reference herein.
[0190] In some aspects, the machine vision apparatus operates to detect a volume of a fluidic substance in a container and determine whether the volume held in the container is appropriate as targeted. As described herein, this volume detection is configured to detect a volume at a
dispense tip using the dispense tip image capture unit, and a volume at a vessel using a vessel image capture unit.
[0191] In some aspects, the machine vision apparatus operates to detect any interferents, which can interfere with an analytic procedure and may generate incorrect results in the dispense tip. As described herein, this dispense tip evaluation is configured to determine a quality of a fluidic substance at a dispense tip using a dispense tip image capture unit, and an alignment of the dispense tip with respect to the dispense tip image capture unit.
[0192] In some aspects, the machine vision apparatus operates to determine a particle concentration in a fluidic substance contained in a vessel, such as a reaction vessel, a sample vessel, a dilution vessel, a cuvette, or any suitable type of vessel, which is used throughout the process in the immunoassay analyzer. In some embodiments, this reaction vessel particle concentration check uses the vessel image capture unit.
[0193] The dispense tip image capture unit operates to capture images of dispense tips in one or more locations. In some embodiments, the dispense tip image capture unit is fixed at a particular location in the instrument. In other embodiments, the dispense tip image capture unit is movably disposed in the instrument, which can move either independently from other components of the instrument or together with one or more components of the instrument. Some embodiments of the instrument include a plurality of dispense tip image capture units. As described herein, the dispense tip image capture unit can include a camera unit.
[0194] The vessel image capture unit operates to capture images of vessels in one or more locations. In some embodiments, the vessel image capture unit is fixed at a particular location in the immunoassay analyzer. In other embodiments, the vessel image capture unit is movably disposed in the immunoassay analyzer, which can move either independently from other components of the immunoassay analyzer or together with one or more components of the immunoassay analyzer. Some embodiments of the immunoassay analyzer include a plurality of vessel image capture units. As described herein, the vessel tip image capture unit includes a camera unit.
[0195] The carriage image capture unit operates to capture images of container carriage devices with or without containers in one or more locations. In some embodiments, the carriage image capture unit is fixed at a particular location in the instrument. In other embodiments, the carriage image capture unit is movably disposed in the instrument, which can move either independently from other components of the instrument or together with one or more components of the
instrument. Some embodiments of the instrument include a plurality of carriage image capture units.
[0196] All of the units of the immunoassay analyzer are connected to a controller, which can perform block control of all of the analyzer functions by using, for example, a microcomputer. The controller may contain subunits such as a data processing unit, a communication interface, and others. A controller in accordance with an exemplary embodiment of the present technology may comprise a data processor, a non-transitory computer-readable medium, and a data storage coupled to the data processor. The non-transitory computer-readable medium may comprise code, executable by the data processor, to perform the functions described herein. The data processor may store, for example, data for processing samples, sample data, or data for analyzing sample data.
[0197] The data processor may include any suitable data computation device or combination of such devices. An exemplary data processor may comprise one or more microprocessors working together to accomplish a desired function. The data processor may include a CPU that comprises at least one high-speed data processor adequate to execute program components for executing user and/or system-generated requests. The CPU may be a microprocessor such as AMD’s Athlon, Duron and/or Opteron; IBM and/or Motorola’s PowerPC; IBM’s and Sony’s Cell processor; Intel’s Celeron, Itanium, Pentium, Xeon, and/or XScale; Apple Ml, and/or the like processor(s).
[0198] The computer-readable medium and the data storage may be any suitable device or devices that can store electronic data. Examples of memories may comprise, for example, one or more memory chips, disk drives, etc. Such memories may operate using any suitable electrical, optical, and/or magnetic mode of operation.
[0199] The computer-readable medium may comprise code, executable by the data processor to perform any suitable method. For example, the computer-readable medium may comprise code, executable by the processor, to cause the controller to operate on a pre-determined schedule. In some embodiments of the presently claimed technology, the pre-determined schedule is a constituent test.
[0200] In an aspect the cycle time is about 45 seconds or less. The “cycle time” is the time required for all modules and/or functions of an immunoassay analyzer to complete its tasks necessary for generating a result. In certain embodiments, the cycle time is about 40 seconds or less, alternatively about 35 seconds or less, alternatively about 30 seconds or less, alternatively about 25 seconds or less, alternatively about 20 seconds or less, or alternatively about 15
seconds. At operation, the sample pipettor can complete its tasks in 8 seconds and the reagent pipettor can complete its tasks in 32 seconds. In this embodiment, to maintain the throughput, four reagent pipettors are present in the immunoassay analyzer (32 seconds/4 = 8 seconds). If the incubation of one of the reaction mixtures is 5 minutes, at least 38 incubation positions is needed to support this reaction time (300 seconds/8 seconds = 37.5 seconds). In certain embodiments, the immunoassay analyzer has at least 30 incubation positions, alternatively at least 40 incubation positions, alternatively at least 50 incubation positions, alternatively at least 60 incubation positions, alternatively at least 70 incubation positions, alternatively at least 80 incubation positions, alternatively at least 90 incubation positions, alternatively at least 100 incubation positions, alternatively at least 125 incubation positions, alternatively at least 150 incubation positions, alternatively at least 175 incubation positions, or alternatively at least 200 incubation positions.
[0201] In an aspect, the time to first result (TTFR) is about 60 minutes or less. The “TTFR” is a measure of time from when the sample is aspirated to when the presence and/or concentration of an analyte is determined. In certain embodiments the TTFR is about 60 minutes or less, alternatively about 55 minutes or less, alternatively about 50 minutes or less, alternatively about 45 minutes or less, alternatively about 40 minutes or less, alternatively about 35 minutes or less, alternatively about 30 minutes or less, alternatively about 25 minutes or less, alternatively about 20 minutes or less, alternatively about 15 minutes or less, or alternatively about 10 minutes or less.
[0202] In an aspect of the methods disclosed herein, a biological sample is analyzed for p- tau217 using an immunoassay analyzer disclosed herein. In an embodiment, the method includes aspirating, using a sample pipettor, a portion of the biological sample from a sample vessel and dispensing the aspirated sample into a reaction vessel of an immunoassay analyzer. In an aspect, the biological sample is serum, whole blood, plasma, and/or cerebral spinal fluid.
[0203] In an embodiment, the method includes aspirating, using a reagent pipettor, a portion of a first reagent from at least one reagent vessel and dispensing the aspirated reagent into the reaction vessel. In some embodiments, the reagent pack may include a plurality of reagent vessels, wherein the reagent pack is configured to store a volume of reagent required for at least about 20 instances of a p-tau217 assay. In a non-limiting example, the reagent pack includes at least three reagent vessels, wherein each reagent vessel is independently configured to store a volume of reagent required for at least about 20 instances of a p-tau217 assay. In a non-limiting example, the reagent pack includes at least four reagent vessels, wherein each reagent vessel is
independently configured to store a volume of reagent required for at least about 20 instances of a p-tau217 assay. In a non-limiting example, the reagent pack includes at least five reagent vessels, wherein each reagent vessel is independently configured to store a volume of reagent required for at least about 20 instances of a p-tau217 assay.
[0204] In an aspect, a first reagent includes at least one affinity molecule configured to bind to at least one portion p-tau217. Depending on the assay, the affinity molecule may be an antibody, a monoclonal antibody, a polyclonal antibody, a synthetic antibody mimic, an aptamer, an affimer, DARPins, or oligonucleotides or peptides that bind to at least one epitope of p-tau217. In an embodiment, the affinity molecule of the first reagent binds with the p-tau217 forming a first reaction mixture. In a non-limiting example, the incubation time of the first reaction mixture is at least about 30 minutes, alternatively at least about 40 minutes, or alternatively at least about 50 minutes, alternatively at least about 55 minutes, or alternatively at least about 60 minutes.
[0205] In a non-limiting example, the affinity molecule is an antibody and the method further comprises exposing the biological sample to a first antibody which binds to a first p-tau217 epitope and a second antibody which binds to a second p-tau217 epitope. In certain embodiments, the first antibody is a capture antibody and the second antibody is a detection antibody. In other embodiments, both antibodies are capture antibodies.
[0206] In an embodiment, a p-tau217 antibody is bound to magnetic bead. Magnetic beads comprising said antibody are added to the sample in a reaction vessel along. At least one buffer or other reagents may be added as required by the assay. The mixture is incubated for a period of time necessary for the antibody to bind with a p-tau217 epitope forming a first reaction mixture. A magnetic field is applied to the reaction vessel and the isolated, bead-bound proteins are washed using the washing arrangement. A base number (three) of wash actions is performed with optional one or two additional wash actions.
[0207] In an embodiment, the method includes, aspirating, using the reagent pipettor, a portion of a second reagent from at least one reagent vessel and dispensing the aspirated reagent into the reaction vessel. In an aspect, a second reagent includes a detection molecule. The detection molecule, may be, for example, a detection antibody, such as alkaline phosphatase (AP)- conjugated secondary antibody. In an embodiment, the detection molecule of the second reagent binds with the affinity molecule of the first reagent (which is bound with the p-tau217 in the biological sample) or binds to a different p-217 epitope. In an aspect, the incubation time of the
second reaction mixture is at least about 5 minutes, alternatively at least about 8 minutes, or alternatively at least about 10 minutes.
[0208] In an embodiment, a p-tau217 antibody conjugated to alkaline phosphatase is added to the first reaction mixture in a reaction vessel. The mixture in incubated for a period of time necessary for the antibody to bind with a second p-tau217 epitope, forming a second reaction mixture. A magnetic field is applied to the reaction vessel and the isolated, bead-bound proteins are washed using the washing arrangement. A base number (three) of wash actions is performed with optional one or two additional wash actions.
[0209] IV. p-tau217 Antibodies
[0210] Provided herein are antibodies that specifically bind to p-tau217. As used herein “antibody” refers to a protein which exhibits binding specificity to a specific antigen. An antibody often comprises a variable domain and a constant domain, each of which includes a heavy chain and a light chain. Accordingly, most antibodies have a heavy chain variable domain (VH) and a light chain variable domain (VL) that together form the portion of the antibody that binds to the antigen. Within each variable domain are three complementarity determining regions (CDR) which form loops in the heavy chain variable domain (VH) and light chain variable domain (VL) that contact the surface of the antigen. Antibodies herein also include intact molecules as well as functional fragments thereof, which are also referred to as an “antigen binding portion” or fragments of the antibody that are capable of binding to the antigen.
[0211] As used herein “chimeric” antibodies are antibodies having a portion of the heavy and/or light chain identical with or homologous to corresponding sequences in antibodies derived from a particular species or belonging to a particular antibody class or subclass, while the remainder of the chain(s) is identical with or homologous to corresponding sequences in antibodies derived from another species or belonging to another antibody class or subclass, as well as fragments of such antibodies, so long as they exhibit the desired biological activity (see e.g., Morrison et al., Proc. Natl. Acad. Sci. USA 81 :6851-6855 (1984)).
[0212] In some embodiments, p-tau217 antibodies comprise at least one heavy chain and at least one light chain. In some embodiments, p-tau217 antibodies comprise at least one heavy chain comprising a VH and at least one light chain comprising a VL, wherein each VH and VL comprises three CDRs. The amino acid sequences of the VH and VL and the CDRs determine the antigen binding specificity and antigen binding strength of the antibody.
[0213] In some embodiments, the detection antibody binds to an epitope on p-tau217 comprising amino acids 1 - 25, 1 - 20, 6 - 18 or 7 - 20 of SEQ ID NO: 1. In some embodiments the detection antibody binds to an epitope on p-tau217 comprising 7 - 20 of SEQ ID NO: 1. In some embodiments the detection antibody binds to an epitope on p-tau217 comprising 6 - 18 of SEQ ID NO: 1. In some embodiments the capture antibody binds to an epitope on p-tau217 comprising amino acids 217, 210 - 225, or 210 - 220 of SEQ ID NO: 1. In some embodiments the capture antibody binds to an epitope on p-tau217 comprising amino acids 210 - 220 of SEQ ID NO: 1 of SEQ ID NO: 1. In some embodiments the detection antibody binds to an epitope on p-tau217 having the amino acid sequence set forth in SEQ ID NOS: 2, 3 or 4 herein. In some embodiments the detection antibody binds to an epitope on p-tau217 having an amino acid sequence with a sequence identity of at least about 75%, at least about 80%, at least about 81%, at least about 82%, at least about 83%, at least about 84%, at least about 85%, at least about 86%, at least about 87%, at least about 88%, at least about 89%, at least about 90%, at least about 91%, at least about 92%, at least about 93%, at least about 94%, at least about 95%, at least about 96%, at least about 97%, at least about 98%, or at least about 99% identical to the amino acid sequences set forth in SEQ ID NOS: 2, 3 or 4 herein.
[0214] In some embodiments the detection antibody binds to an epitope on p-tau217 comprising amino acids 1 - 20 of SEQ ID NO: 1, and the capture antibody binds to an epitope on p-tau217 comprising amino acids 210 - 220 of SEQ ID NO: 1. In some embodiments the detection antibody binds to an epitope on p-tau217 having the amino acid sequence set forth in SEQ ID NO: 2 and the capture antibody binds to an epitope on p-tau217 having an epitope on p-tau217 having the amino acid sequence set forth in SEQ ID NO: 4. In some embodiments the detection antibody binds to an epitope on p-tau217 having an amino acid sequence with a sequence identity of at least about 75%, at least about 80%, at least about 81%, at least about 82%, at least about 83%, at least about 84%, at least about 85%, at least about 86%, at least about 87%, at least about 88%, at least about 89%, at least about 90%, at least about 91%, at least about 92%, at least about 93%, at least about 94%, at least about 95%, at least about 96%, at least about 97%, at least about 98%, or at least about 99% identical to the amino acid sequence set forth in SEQ ID NO: 2, and the capture antibody binds to an epitope on p-tau217 having an amino acid sequence with a sequence identity of at least about 75%, at least about 80%, at least about 81%, at least about 82%, at least about 83%, at least about 84%, at least about 85%, at least about 86%, at least about 87%, at least about 88%, at least about 89%, at least about 90%, at least about 91%, at least about 92%, at least about 93%, at least about 94%,
at least about 95%, at least about 96%, at least about 97%, at least about 98%, or at least about 99% % identical to the to the amino acid sequence set forth in SEQ ID NO: 4.
0216] In some instances, the detection antibody that binds p-Tau217 comprises a heavy chain variable region (VH) of SEQ ID NO: 13. In some instances, the detection antibody that binds p-tau217 comprises an antigen-binding site of a light chain variable region (VL) of SEQ ID NO: 14.
[0217] In some instances, the detection antibody that binds p-Tau217 comprises a VH having a sequence identity of at least about 75%, at least about 80%, at least about 81%, at least about 82%, at least about 83%, at least about 84%, at least about 85%, at least about 86%, at least about 87%, at least about 88%, at least about 89%, at least about 90%, at least about 91%, at least about 92%, at least about 93%, at least about 94%, at least about 95%, at least about 96%, at least about 97%, at least about 98%, or at least about 99% identical to an amino acid sequence set forth as SEQ ID NO: 13.
[0218] In some instances, the capture antibody that binds p-Tau217 comprises a VL having a sequence identity of at least about 75%, at least about 80%, at least about 81%, at least about 82%, at least about 83%, at least about 84%, at least about 85%, at least about 86%, at least about 87%, at least about 88%, at least about 89%, at least about 90%, at least about 91%, at least about 92%, at least about 93%, at least about 94%, at least about 95%, at least about 96%, at least about 97%, at least about 98%, or at least about 99% identical to an amino acid sequence set forth as SEQ ID NO: 14.
[0219] In some instances, the capture antibody that binds p-tau217 comprises an antigenbinding site of a heavy chain variable region (VH) of SEQ ID NO: 13, and an antigen-binding site of a light chain variable region (VL) of SEQ ID NO: 14.
[0220] In some instances, the detection antibody comprises heavy chain CDRs - HCDR1, HCDR2 and HCDR3 - shown in SEQ ID NOs: 15, 16 and 17. In some instances, the detection antibody comprises light chain CDRs - LCDR1, LCDR2 and LCDR3 - shown in SEQ ID NOs:
18, 19 and 20. In some instances, the detection antibody comprises heavy chain CDRs - HCDR1, HCDR2 and HCDR3- having a sequence identity of at least about 75%, at least about 80%, at least about 81%, at least about 82%, at least about 83%, at least about 84%, at least about 85%, at least about 86%, at least about 87%, at least about 88%, at least about 89%, at least about 90%, at least about 91%, at least about 92%, at least about 93%, at least about 94%, at least about 95%, at least about 96%, at least about 97%, at least about 98%, or at least about 99% identical to an amino acid sequence set forth as SEQ ID NOs: 15, 16 and 17.
[0221] In some instances, the detection antibody comprises light chain CDRs - LCDR1, LCDR2 and LCDR3- having a sequence identity of at least about 75%, at least about 80%, at least about 81%, at least about 82%, at least about 83%, at least about 84%, at least about 85%, at least about 86%, at least about 87%, at least about 88%, at least about 89%, at least about 90%, at least about 91%, at least about 92%, at least about 93%, at least about 94%, at least about 95%, at least about 96%, at least about 97%, at least about 98%, or at least about 99% identical to an amino acid sequence set forth as SEQ ID NOs: 18, 19 and 20. In some instances, the detection antibody comprises heavy chain CDRs - HCDR1, HCDR2 and HCDR3 - having the amino acids sequence set forth in SEQ ID NOs: 15, 16 and 17 and light chain CDRs - LCDR1, LCDR2 and LCDR3 - having the amino acid sequence set forth in SEQ ID NOs: 18, 19 and 20.
[0222] The detection antibody and/or capture antibody may be a polyclonal antibody or a monoclonal antibody. Monoclonal antibodies may be acquired using know techniques including by using an antibody-producing cells such as hybridomas.
[0223] In some instances, the capture antibody that binds p-tau217 comprises a heavy chain variable region (VH) of SEQ ID NO: 5. In some instances, the capture antibody that binds p- tau217 comprises a light chain variable region (VL) of SEQ ID NO: 6. In some instances, the capture antibody that binds p-tau217 comprises a VH having a sequence identity of at least about 75%, at least about 80%, at least about 81%, at least about 82%, at least about 83%, at least about 84%, at least about 85%, at least about 86%, at least about 87%, at least about 88%, at least about 89%, at least about 90%, at least about 91%, at least about 92%, at least about 93%, at least about 94%, at least about 95%, at least about 96%, at least about 97%, at least about 98%, or at least about 99% identical to an amino acid sequence set forth as SEQ ID NO: 5.
[0224] In some instances, the capture antibody that binds p-tau217 comprises a VL having a sequence identity of at least about 75%, at least about 80%, at least about 81%, at least about
82%, at least about 83%, at least about 84%, at least about 85%, at least about 86%, at least about 87%, at least about 88%, at least about 89%, at least about 90%, at least about 91%, at least about 92%, at least about 93%, at least about 94%, at least about 95%, at least about 96%, at least about 97%, at least about 98%, or at least about 99% identical to an amino acid sequence set forth as SEQ ID NO: 6. In some instances, the capture antibody that binds p-tau217 comprises an antigen-binding site of a heavy chain variable region (VH) of SEQ ID NO: 5, and an antigen-binding site of a light chain variable region (VL) of SEQ ID NO: 6.
[0225] In some instances, the capture antibody comprises heavy chain CDRs - HCDR1, HCDR2 and HCDR3 - having the amino acid sequence set forth in SEQ ID NOs: 7, 8 and 9. In some instances, the capture antibody comprises light chain CDRs - LCDR1, LCDR2 and LCDR3- having the amino acid sequence set forth in SEQ ID NOs: 10, 11 and 12. In some instances, the capture antibody comprises heavy chain CDRs - HCDR1, HCDR2 and HCDR3 - having a sequence identity of at least about 75%, at least about 80%, at least about 81%, at least about 82%, at least about 83%, at least about 84%, at least about 85%, at least about 86%, at least about 87%, at least about 88%, at least about 89%, at least about 90%, at least about 91%, at least about 92%, at least about 93%, at least about 94%, at least about 95%, at least about 96%, at least about 97%, at least about 98%, or at least about 99% identical to an amino acid sequence set forth as SEQ ID NOs: 7, 8 and 9.
[0226] In some instances, the capture antibody comprises light chain CDRs - LCDR1, LCDR2 and LCDR3 - having a sequence identity of at least about 75%, at least about 80%, at least about 81%, at least about 82%, at least about 83%, at least about 84%, at least about 85%, at least about 86%, at least about 87%, at least about 88%, at least about 89%, at least about 90%, at least about 91%, at least about 92%, at least about 93%, at least about 94%, at least about 95%, at least about 96%, at least about 97%, at least about 98%, or at least about 99% identical to an amino acid sequence set forth as SEQ ID NOs: 10, 11 and 12.
[0227] In some instances, the capture antibody comprises heavy chain CDRs - HCDR1, HCDR2 and HCDR3 - having a sequence as set forth in SEQ ID NOs: 7, 8 and 9 and light chain CDRs - LCDR1, LCDR2 and LCDR3 - having a sequence as set forth in SEQ ID NOs: 10, 11 and 12.
0229] In an embodiment, the method includes dispensing a substrate formulation into the reaction vessel, generating a detection mixture. In certain embodiments, the substrate formulation comprises compound I, a cationic aromatic compound, a background reducing agent, an ether-linked non-ionic surfactant or hydrophilic polymer, an anionic surfactant, and an amine buffer. A magnetic field is applied to the reaction vessel and the isolated, bead-bound proteins are washed using the washing arrangement. A base number (three) of wash actions is performed with optional one or two additional wash actions.
[0230] In an embodiment, the method further includes detecting, using a detector arrangement, a presence of a reaction in the detection mixture and determining a presence and/or concentration of p-tau217 in the biological sample based on the presence of the reaction in the detection mixture. In an aspect, the reaction in the detection mixture generates a chemiluminescent signal, wherein the chemiluminescent signal corresponds to the presence and/or concentration of p-tau217 in the biological sample.
[0231] At operation, the sample pipettor can aspirate the sample from the sample vessel and then move to a sample dispense position, where the reaction vessels are provided. A nonlimiting example of an operation is depicted in FIGs. 2A-2C. The sample pipettor operates to dispense the aspirated samples into the reaction vessels 202. In a non-limiting embodiment, the sample pipettor operates to dispense a predetermined volume of sample into four different reaction vessels. After the sample is dispensed into the reaction vessels, the first reagent pipettor operates to aspirate a portion of a fluidic substance from a first reagent vessel and dispense a predetermined volume of reagent into a first reaction vessel 204. Then the second reagent pipettor operates to aspirate a portion of a fluidic substance from a first reagent vessel and dispense a predetermined volume of reagent into a second reaction vessel 206. Followed by the third reagent pipettor operating to aspirate a portion of a fluidic substance from a first reagent vessel and dispense a predetermined volume of reagent into a third reaction vessel 208 and the fourth reagent pipettor operating to aspirate a portion of a fluidic substance from a first reagent vessel and dispense a predetermined volume of reagent into a fourth reaction vessel 210. After dispensing the first reagent into the reaction vessels, all of the reaction vessel may be incubated simultaneously to form a first reaction mixture.
[0232] After the required incubation time needed to form a first reaction mixture, the first reagent pipettor operates to aspirate a portion of a fluidic substance from a second reagent vessel and dispense a predetermined volume of reagent into a first reaction vessel 212. Then the second reagent pipettor operates to aspirate a portion of a fluidic substance from a second reagent vessel and dispense a predetermined volume of reagent into a second reaction vessel 214. Followed by the third reagent pipettor operating to aspirate a portion of a fluidic substance from a second reagent vessel and dispense a predetermined volume of reagent into a third reaction vessel 216 and the fourth reagent pipettor operating to aspirate a portion of a fluidic substance from a second reagent vessel and dispense a predetermined volume of reagent into a fourth reaction vessel 218. After dispensing the first reagent into the reaction vessels, all of the reaction vessel may be incubated simultaneously to form a second reaction mixture.
[0233] After the required incubation time needed for form a second reaction mixture, a substrate formulation is dispensed into the first reaction vessel, generating a first reaction mixture 220. Then a substrate formulation is dispensed into the second reaction vessel, generating a second reaction mixture 222. Followed by dispensing a substrate formulation into the third reaction vessel, generating a third reaction mixture 224 and dispensing a substrate formulation into the fourth reaction vessel, generating a fourth reaction mixture 226. After dispensing the substrate into the reaction vessels, all of the reaction vessels may be incubated simultaneously to form the detection mixtures. The incubated detection mixtures are then individually and independently analyzed. This includes the use of the detector arrangement to detect a reaction in the detection mixture in the first reaction vessel, the second reaction vessel, the third reaction vessel, and the fourth reaction vessel 228. Based on the presence of a reaction in the detection mixture, the amount of p-tau217 in the plasma sample in each reaction vessel can be quantitatively determined 230.
[0234] The claimed configuration allows for the simultaneous performance of at least two assays for a plurality of plasma samples, alternatively at least three assays for a plurality of plasma samples, or at least four assays for a plurality of plasma samples. Depending on the analysis desired, the plasma samples may be from the same subject or from multiple subjects. In some aspects, the method further comprises a plurality of sample vessels. In some embodiments, the method comprises at least one sample vessel, at least two sample vessels, at least three sample vessels, at least four sample vessels, at least five sample vessels, at least ten sample vessels, at least twenty sample vessels, at least fifty sample vessels, at least one hundred sample vessels, at least two hundred sample vessels, at least three hundred sample vessels, at least four hundred sample vessels, or at least five hundred sample vessels. In some aspects, the sample vessels are housed within the immunoassay analyzer.
[0235] In certain embodiments, at least one of the assays is p-tau217 assay, alternatively at least two of the assays are p-tau217 assays, alternatively at least three of the assays are p-tau217 assays, or alternatively at least four of the assays are p-tau217 assays.
[0236] In certain embodiments, the method may include detecting at least one additional biomarker related to Alzheimer’s Disease or another neurological disorder. In some embodiments, at least one of the assays is p-tau217 assay and at least one of the assays is a second analyte assay, wherein the second analyte is selected from the group consisting of A|3 peptides, Neurofilament light chain (NfL), total tau (t-tau), p-tau231, p-taul81, and combinations thereof.
[0237] In a non-limiting example, if multiple assays for multiple biomarkers are being performed, the first pipettor may be configured to aspirate a reagent from a reagent vessel comprising a first affinity molecule (e.g., a p-tau217 antibody) and dispense into a first reaction vessel, while a second pipettor may be configured to simultaneously aspirate a reagent from a reagent vessel comprising a second affinity molecule (e.g., second analyte antibody) and dispense into a second reaction vessel. Depending on the analysis desired, the third and fourth pipettor may also be configured to simultaneously aspirate a reagent from a reagent vessel comprising a third and fourth affinity molecule and dispense into a third and fourth reaction vessel.
[0238] The disclosed simultaneous and/or selective operation of the pipettors allows for a high- throughput analysis. In some embodiments, the method is configured to analyze at least about 200 plasma samples/hr., alternatively at least about 300 plasma samples/hr., or alternatively at least about 400 plasma samples/hr.
[0239] In some cases, for patients having AD, or at risk of progressing to AD, the levels of p- tau217 in blood may be in the AttoMole, Milli-international Unit (mIU), pg, or fg range. In a non-limiting example, the quantitation detection limit (LOQ) for measuring p-tau217 is equal to or less than about 3 pg/mL, alternatively equal to or less than about 2 pg/mL, equal to or less than about 1 pg/mL, equal to or less than about 0.6 pg/mL and wherein the detected p-tau217 has a concentration above the LOQ. In a non-limiting example, the quantitation detection limit (LOQ) for measuring p-tau217 is equal to or less than about 100 pg/mL, less than about 90 pg/mL, less than about 80 pg/mL, less than about 70 pg/mL, less than about 60 pg/mL, less than about 50 pg/mL, less than about 40 pg/mL, less than about 30 pg/mL, less than about 20 pg/mL, or less than about 10 pg/mL. In a non-limiting example, the quantitation detection limit (LOQ) for measuring p-tau217 is equal to or less than about less than about 100 pg/mL, less than about 90 pg/mL, less than about 80 pg/mL, less than about 70 pg/mL, less than about 60 pg/mL, less than about 50 pg/mL, less than about 40 pg/mL, less than about 30 pg/mL, less than about 20 pg/mL, or less than about 10 pg/mL. In a non-limiting example, the quantitation detection limit (LOQ) for measuring p-tau217 is equal to or less than about 10 pg/mL, less than about 9 pg/mL, less than about 8 pg/mL, less than about 7 pg/mL, less than about 6 pg/mL, less than about 5 pg/mL, less than about 4 pg/mL, less than about 3 pg/mL, less than about 2 pg/mL, or less than about 1 pg/mL. In a non-limiting example, the quantitation detection limit (LOQ) for measuring p-tau217 is equal to or less than about Ipg/mL, less than about 0.9pg/mL, less than about 0.8pg/mL, less than about 0.7pg/mL, less than about 0.6pg/mL, less than about 0.5pg/mL, less than about 0.4pg/mL, less than about 0.3pg/mL, less than about 0.2pg/mL, or less than about
O. lpg/mL. In a non-limiting example, the quantitation detection limit (LOQ) for measuring p- tau217 is equal to or less than about 100 fg/mL, less than about 90 fg/mL, less than about 80 fg/mL, less than about 70 fg/mL, less than about 60 fg/mL, less than about 50 fg/mL, less than about 40 fg/mL, less than about 30 fg/mL, less than about 20 fg/mL, or less than about 10 fg/mL. In a non-limiting example, the quantitation detection limit (LOQ) for measuring p- tau217 is equal to or less than about 10 fg/mL, less than about 9 fg/mL, less than about 8 fg/mL, less than about 7 fg/mL, less than about 6 fg/mL, less than about 5 fg/mL, less than about 4 fg/mL, less than about 3 fg/mL, less than about 2 fg/mL, or less than about 1 fg/mL. In another non-limiting example, the detected p-tau217 has a concentration that is at least IX greater than the LOQ, alternatively at least 2X greater than the LOQ, alternatively at least 3X greater than the LOQ, alternatively at least 4X greater than the LOQ, alternatively at least 5X greater than the LOQ, alternatively at least 10X greater than the LOQ, alternatively at least 100X greater than the LOQ, or alternatively at least 100X greater than the LOQ. In yet another non-limiting example, the detected p-tau217 exhibits a coefficient of variation (CV) of 20% or less, 19% or less, 18% or less, 17% or less, 16% or less, 15% or less, 14% or less, 13% or less, 12% or less, 11% or less, 10% or less, 9% or less, 8% or less, 7% or less, 6% or less, 5% or less, 4% or less, 3% or less, 2% or less, or 1% or less. In yet another non-limiting example, the detected p-tau217 exhibits a coefficient of variation (CV) of 20% or less. In yet another non-limiting example, the detected p-tau217 exhibits a coefficient of variation (CV) of 4% or less.
[0240] As shown in FIG. 1, an exemplary immunoassay analyzer (i.e., an immunoassay analyzer according to an aspect of this disclosure) and conventional immunoassay analyzers were evaluated to demonstrate the sensitivity for existing immunoassays. Thyroid-Stimulating Hormone (TSH), commercial assays were employed per manufacturers’ protocol. An exemplary assay protocol according to an aspect of the invention is listed in Example 1. Commercially available calibrators were used to create series of dilutions. The assay protocol was performed on each dilution to generate a measurement in RLUs, and corresponding coefficient of variation (CV), signal-noise-ratios, LOQs, and other performance characteristics were calculated. The signal -to-noise ratio (S/N) gives a measure of the degree of confidence that a difference in signal noise and background is real. In exemplary methods, a blank control is analyzed and the resulting values are used to calculate S/N. The error range corresponds to the CV of the specific reportable parameter at the concentration tested. It is noted that the CV varies depending on the concentrations and instruments. The 1st conventional immunoassay analyzer had low CVs, but there was little RLUs left at the lower concentrations. The 2nd conventional immunoassay analyzer had improved signal/noise ratios, but poor CVs at the end.
The exemplary immunoassay analyzer was the only immunoassay analyzer that did not have technical limitations in achieving the sensitivity useful for detecting low p-tau217 concentrations. As can be seen from FIG. 1, the LOQ of the exemplary immunoassay analyzer is at least around 0.013 pIU/mL (appx. 2 pg /mL) with a CV of around 14.2%.
[0241] The disclosed immunoassay methods achieve a functional sensitivity of 0.01-0.02 pIU/mL (mIU/L) with an interassay %CV < 20%. This unpredictable magnitude of sensitivity is superior to conventional methods allowing for the sensitivity and precision needed to distinguish and quantify different low levels of p-tau217.
[0242] Additional examples are provided below.
[0243] EXAMPLES
[0244] Example 1: TSH Sensitivity Comparison
[0245] A calibrator (SI, 0.0500 pIU/mL (micro-international units per milliliter) was used to create a series of dilutions with concentrations of 0.0050 pIU/mL (Sl/10), 0.0025 pIU/mL (Sl/20), 0.0013 pIU/mL (Sl/40), 0.0006 pIU/mL (Sl/80), and 0.0000 pIU/mL (SO). The TSH sensitivity comparison was performed using an exemplary immunoassay analyzer having (i) four reagent pipettors and one sample pipettor configured to aspirate and dispense less than about 10 pL; (ii) reagent packs configured to store volumes of reagents for at least 20 instances of assays; (iii) an ultrasonic mixer; and (iv) the capability to perform over 5 wash cycles per reaction vessel.
[0246] The following steps were carried out:
[0247] Paramagnetic particle conjugated with TSH antibodies capable of binding to an epitope TSH were pipetted into a reaction vessel using one of four reagent pipettors. A sample aliquot was pipetted into the reaction vessel using a sample pipettor. Then the reaction vessel was mixed using the ultrasonic mixer and incubated, generating a first reaction mixture. A magnetic field was applied to the reaction vessel and the first reaction mixture was washed using a wash buffer to remove any unreacted components. A secondary antibody capable of binding a different epitope of TSH conjugated to alkaline phosphatase (“ALP”) was added to the reaction vessel containing the first reaction mixture. The reaction vessel was mixed ultrasonically and incubated, generating a second reaction mixture. A magnetic field was applied to the reaction vessel and the second reaction mixture was washed using a wash buffer to remove any unreacted components. A substrate according to the present disclosure was added to the reaction vessel, and the signal generated from the resulting reaction was read using a luminometer.
[0248] Test results were determined automatically by the system software. Detection of analyte in the sample was determined from the measured light production by means of the stored calibration data. The RLU data collected for each assayed sample was summed for each individual run, averaged over the triplicates for each sample and presented in FIG. 1. A ratio of signal to noise (S/SO), RLU CVs, CVCF, and Dose CVs were calculated at each concentration value.
[0249] From the results shown in FIG. 1, the exemplary immunoassay analyzer was found to have both a higher signal to noise ratio (S/SO) and lower CVs at the lower concentrations. This is in contrast to 1 st conventional immunoassay analyzer which had lower CVs, but lower RLUs left at the lower concentrations. The 2nd conventional immunoassay analyzer had higher CVs at the lower concentrations. As can be seen from FIG. 1, the LOQ of the exemplary immunoassay analyzer is at least around 0.013 pIU/mL (appx. 2 pg/mL) with a CV of around 14.2%.
[0250] Example 2: Exemplary p-tau217 assay
[0251] An exemplary assay protocol (Table 4) is used to detect the presence of p-tau217 in a plasma sample. The assay was performed using an exemplary immunoassay analyzer according to an aspect of the disclosure.
[0253] A single calibration is generated (duplicate replicates of each level) and a calibration curve is fit to the corresponding signal output. The assay is then used to measure p-tau217 levels in the biological samples
[0254] Example 3: Clinical Discrimination of pTau217 Assay on Exemplary Clinical Analyzer
[0255] Assays according to the present disclosure were assessed for the ability to distinguish between Alzheimer’s Disease (AD) confirmed and normal patient samples.
[0256] Samples tested included 26 normal patient samples (8 young <55 years patients; 18 aged healthy patients >55 years) and 40 Alzheimer’s positive patient samples (confirmed by physician; MMSE scores ranging from 2 to 25).
[0257] A p-tau217 assay as described herein was performed on each sample using Beckman Coulter’s Dxl 9000 analyzer. Assay methodology is summarized below:
[0258] Using an exemplary immunoassay analyzer having (i) four reagent pipettors and one sample pipettor configured to aspirate and dispense less than about 10 pL; (ii) reagent packs configured to store volumes of reagents for at least 20 instances of assays; (iii) an ultrasonic mixer; and (iv) the capability to perform over 5 wash cycles per reaction vessel, the following steps were carried out:
[0259] Step 1 : Paramagnetic particle conjugated p-tau217 antibodies capable of binding to an epitope comprising amino acid 217 (SEQ ID NO: 4) of p-tau217 were pipetted into a reaction vessel using one of four reagent pipettors. The paramagnetic particle conjugated p-tau217 antibodies comprised CDR regions having the amino acid sequences of SEQ ID NOs: 7 - 12, a VL region having the amino sequence of SEQ ID NO: 6, and a VH region having the amino acid sequence of SEQ ID NO: 5. A sample aliquot was pipetted into the reaction vessel using a sample pipettor. Then the reaction vessel was mixed using the ultrasonic mixer and incubated, generating a first reaction mixture.
[0260] Step 2: A magnetic field was applied to the reaction vessel and the first reaction mixture was washed using a wash buffer to remove any unreacted components.
[0261] Step 3: A p-tau217 antibody capable of binding to the N-terminus of p-tau217 (SEQ ID NO: 3) conjugated to alkaline phosphatase (“ALP”) was added to the reaction vessel containing the first reaction mixture. The ALP conjugated p-tau217 antibodies comprised CDR regions having the amino acid sequences of SEQ ID NOs: 15 - 20, a VL region having the amino sequence of SEQ ID NO: 14, and a VH region having the amino acid sequence of SEQ ID NO: 13. The reaction vessel was mixed ultrasonically and incubated, generating a second reaction mixture.
[0262] Step 4: A magnetic field was applied to the reaction vessel and the second reaction mixture was washed using a wash buffer to remove any unreacted components.
[0263] Step 5: Substrate according to the present disclosure was added to the reaction vessel, and the signal generated from the resulting reaction was read using a luminometer.
[0264] The above steps were performed using calibrator antigens of varying concentrations (2 replicates each), and a calibration curve was constructed and reduced with adequate fit. The mean signal response for each of the calibrator levels is shown below:
[0265] The assay was performed on the Normal (4 replicates each) and AD-confirmed (2 replicates each) patient samples, and the calibration curves were used to correlating the resulting signal produced to concentrations. Resulting calibration curve plot is shown in FIG. 3.
[0266] All patient samples were run on the exemplary assay and were shown to be able to distinguish between Alzheimer’s positive versus normal sample. A box and whisker plot showing pTau217 concentrations associated with AD-positive versus normal samples is illustrated in FIG. 4. Alzheimer’s positive population were determined to have a median pTau217 concentration of 261 fg/mL, whereas Alzheimer’s normal healthy population were found to have a pTau217 concentration of 88 fg/mL, with a median based ratio of AD-positive to normal concentrations of 3.0.
[0267] An analysis of the analytical sensitivity of the exemplary pTau217 was also conducted. Within-run CV was calculated from harmonized dose response and plotted relative to mean dose-response, as shown in FIG. 5. A log-log quadratic regression was fit to model overall precision. The LOQs at 20% and 10% CV were than calculated and compared to published performance of other conventional analyzers, as shown below in Table 6.
Table 6: Within-Run LoQ Estimates
0268] Example 4: Quantitative Sandwich Immunoassay for pTau217
[0269] Six assays according to the present disclosure were assessed for the ability to distinguish between Alzheimer’s Disease (AD) confirmed and normal patient samples.
[0270] Samples included 20 Alzheimer’s positive Patients and 20 Normal Patients. Each of the assays was performed using one of six antibody pairs.
[0271] Each p-Tau217 assay as described herein was performed on a Beckman Coulter’s Dxl 9000 analyzer having (i) four reagent pipettors and one sample pipettor configured to aspirate and dispense less than about 10 pL; (ii) a reagent pack with 4 reagent vessels (the first vessel containing paramagnetic particle conjugated p-tau217 antibodies, the second vessel containing ALP conjugated p-tau217 antibodies and the third and fourth vessels containing ancillary buffers required for the assay), where each reagent vessel is configured to store volumes of reagents for at least 20 instances of assays; (iii) an ultrasonic mixer; and (iv) the capability to perform over 5 wash cycles per reaction vessel.
[0272] Assay methodology is summarized below:
[0273] Step 1 : An aliquot from the first vessel containing paramagnetic particle conjugated p- tau217 antibodies was pipetted into a reaction vessel using one of four reagent pipettors. A sample aliquot was pipetted into the reaction vessel using a sample pipettor and an aliquot from the third reagent vessel containing ancillary buffers was pipetted into the reaction vessel using one of four reagent pipettors. Then the reaction vessel was incubated for 32 seconds. A second sample aliquot was pipetted into the reaction vessel using a sample pipettor and the reaction
vessel was mixed using the ultrasonic mixer and incubated for about 20 minutes, generating a first reaction mixture.
[0274] Step 2: A magnetic field was applied to the reaction vessel and the first reaction mixture was washed three times using a wash buffer to remove any unreacted components.
[0275] Step 3: An aliquot from the second vessel containing ALP conjugated p-tau217 antibodies was pipetted into a reaction vessel using one of four reagent pipettors. The reaction vessel was mixed ultrasonically and incubated for about 8 minutes, generating a second reaction mixture.
[0276] Step 4: A magnetic field was applied to the reaction vessel and the second reaction mixture was washed five times using a wash buffer to remove any unreacted components.
[0277] Step 5: Substrate according to the present disclosure was added to the reaction vessel, and the signal generated from the resulting reaction was read using a luminometer.
[0278] The assay was performed on the Normal (4 replicates each) and AD-confirmed (2 replicates each) patient samples and a calibrator matrix (20 replicates). Corresponding concentrations were calculated for each replicate based on calibration curve fit and the CV% calculated for each sample. Within-run precision was analyzed to generate a precision profile to estimate Limit of Blank, Limit of Detection, and the 20% Limit of Quantitation. The precision profile and calculated attributes of the assay are depicted in FIG. 6.
[0279] The measured concentrations for the Alzheimer’s Disease population and the normal/healthy cohort were then compared, and the results are depicted in FIG. 7. Table 7 plots the signal in RLUs generated by each assay, along with the signal ratio of signal produced by AD Patients versus Normal patients and the dose ratio of concentration of pTau217 detected for AD Patients versus Normal patients.
[0281] As shown in FIG. 7 and Table 7, most samples showed consistent signal correlation across conditions.
[0282] Example 5: p-Tau217 Immunoassay Method Comparison
[0283] Assays performed using antibody pair 1 and pair 4 were compared with two conventional antibody pairs to assess the ability to distinguish between Alzheimer’s Disease (AD) confirmed and normal patient samples.
[0284] Samples included 32 Alzheimer’s + Patients (MMSE Range 10-24), 27 Normal Patients (Age Range 36-74), 4 CSF Spiked Plasma Samples, and 9 Antigen Spiked Samples (recombinant). Each of the assays was performed using antibody pair 1, antibody pair 4, and two conventional antibody pairs.
[0285] Each p-Tau217 assay as described herein was performed on a Beckman Coulter’s Dxl 9000 analyzer having (i) four reagent pipettors and one sample pipettor configured to aspirate and dispense less than about 10 pL; (ii) a reagent pack with 4 reagent vessels (the first vessel containing paramagnetic particle conjugated p-tau217 antibodies, the second vessel containing ALP conjugated p-tau217 antibodies and the third and fourth vessels containing ancillary buffers required for the assay), where each reagent vessel is configured to store volumes of reagents for at least 20 instances of assays; (iii) an ultrasonic mixer; and (iv) the capability to perform over 5 wash cycles per reaction vessel.
[0286] Assay methodology is summarized below:
[0287] Step 1 : An aliquot from the first vessel containing paramagnetic particle conjugated p- tau217 antibodies was pipetted into a reaction vessel using one of four reagent pipettors. A sample aliquot was pipetted into the reaction vessel using a sample pipettor and an aliquot from the third reagent vessel containing ancillary buffers was pipetted into the reaction vessel using one of four reagent pipettors. Then the reaction vessel was incubated for 32 seconds. A second sample aliquot was pipetted into the reaction vessel using a sample pipettor and the reaction vessel was mixed using the ultrasonic mixer and incubated for about 20 minutes, generating a first reaction mixture.
[0288] Step 2: A magnetic field was applied to the reaction vessel and the first reaction mixture was washed three times using a wash buffer to remove any unreacted components.
[0289] Step 3: An aliquot from the second vessel containing ALP conjugated p-tau217 antibodies was pipetted into a reaction vessel using one of four reagent pipettors. The reaction vessel was mixed ultrasonically and incubated for about 8 minutes, generating a second reaction mixture.
[0290] Step 4: A magnetic field was applied to the reaction vessel and the second reaction mixture was washed five times using a wash buffer to remove any unreacted components.
[0291] Step 5: Substrate according to the present disclosure was added to the reaction vessel, and the signal generated from the resulting reaction was read using a luminometer.
[0292] All samples were analyzed in duplicate. Corresponding concentrations were calculated for each replicate based on calibration curve fit and the CV% calculated for each sample. Within-run precision was analyzed to generate a precision profile to estimate the 20% Limit of Quantitation of antibody pair 1 (~ 1.2 attoM/mL). The precision profile and calculated attributes of antibody pair 1 and both conventional antibody pairs are depicted in FIGs. 8A-8C. FIG. 8A depicts %CV for the analyzed antibody pairs, FIG. 8B depicts within-run precision profiles for the analyzed antibody pairs, and FIG. 8C depicts the measured concentrations for the analyzed antibody pairs for AD Patients and Normal patients.
[0293] FIG. 9 is a graph of a comparison between the assay performed using pairs 1 and 4 to conventional assays. As shown in FIG. 9 the experimental antibody pairs demonstrate similar sensitivity and clinical discrimination between healthy and Alzheimer’s diseased patient EDTA plasma samples as the conventional antibody pairs.
[0294] Both antibody pairs show superior specific signal production compared to predicate conventional antibody pairs and superior analytical sensitivity compared to predicate conventional antibody pairs.
[0295] Example 6: p-Tau217 Immunoassay Clinical Performance
[0296] An exemplary p-Tau217 immunoassay using an exemplary antibody pair was assessed for clinical performance including the ability to distinguish between Alzheimer’s Disease (AD) confirmed patient samples, other neurodegenerative disease patient samples, and normal patient samples.
[0297] The samples included 45 samples from patients with an Alzheimer’s diagnosis, 20 samples from patients with other neurodegenerative diseases (including frontotemporal
dementia and Lewy body disease), and 25 aged match control patient samples. The patients with an Alzheimer’s diagnosis were characterized for their amyloid status by CSF Ab42/Ab40 and/or amyloid PET.
[0298] The p-Tau217 assay as described herein was performed on a Beckman Coulter’s Dxl 9000 analyzer having (i) four reagent pipettors and one sample pipettor configured to aspirate and dispense less than about 10 pL; (ii) a reagent pack with 4 reagent vessels (the first vessel containing paramagnetic particle conjugated p-tau217 antibodies, the second vessel containing ALP conjugated p-tau217 antibodies and the third and fourth vessels containing ancillary buffers required for the assay), where each reagent vessel is configured to store volumes of reagents for at least 20 instances of assays; (iii) an ultrasonic mixer; and (iv) the capability to perform over 5 wash cycles per reaction vessel.
[0299] Step 1 : An aliquot from the first vessel containing paramagnetic particle conjugated p- tau217 antibodies was pipetted into a reaction vessel using one of four reagent pipettors. A sample aliquot was pipetted into the reaction vessel using a sample pipettor and an aliquot from the third reagent vessel containing ancillary buffers was pipetted into the reaction vessel using one of four reagent pipettors. Then the reaction vessel was incubated. A second sample aliquot was pipetted into the reaction vessel using a sample pipettor and the reaction vessel was mixed using the ultrasonic mixer and incubated, generating a first reaction mixture.
[0300] Step 2: A magnetic field was applied to the reaction vessel and the first reaction mixture was washed three times using a wash buffer to remove any unreacted components.
[0301] Step 3: An aliquot from the second vessel containing ALP conjugated p-tau217 antibodies was pipetted into a reaction vessel using one of four reagent pipettors. The reaction vessel was mixed ultrasonically and incubated, generating a second reaction mixture.
[0302] Step 4: A magnetic field was applied to the reaction vessel and the second reaction mixture was washed five times using a wash buffer to remove any unreacted components.
[0303] Step 5: Substrate according to the present disclosure was added to the reaction vessel, and the signal generated from the resulting reaction was read using a luminometer.
[0304] All samples were analyzed in duplicate. As shown in FIG. 10, the exemplary p-Tau217 immunoassay was able discriminate between amyloid positive and negative patients.
[0305] The sensitivity versus 1-Specifity for the resulting data was plotted, generating a receiver operating characteristic (ROC) curve. The area under the curve (AUC), which is an effective and combined measure of sensitivity and specificity that describes the inherent validity
of diagnostic tests, was then calculated. The ROC curve shows an AUC = 0.93 with 95% confidence interval 0.858 to 0.994 for the detection of Alzheimer’s disease (43 AD patient samples and 48 Non-AD patient samples) and an AUC = 0.95 with 95% confidence interval 0.892 to 1.004 for the prediction of amyloid status (48 amyloid(+) patient samples and 40 amyloid(-) patient samples) indicating an excellent diagnostic performance of the exemplary p- Tau217 assay.
[0306] All features disclosed in the specification, including the claims, abstracts, and drawings, and all the steps in any method or process disclosed, may be combined in any combination, except combinations where at least some of such features and/or steps are mutually exclusive. Each feature disclosed in the specification, including the claims, abstract, and drawings, can be replaced by alternative features serving the same, equivalent or similar purpose, unless expressly stated otherwise. Thus, unless expressly stated otherwise, each feature disclosed is one example only of a generic series of equivalent or similar features.
Further embodiments of the invention are described herein:
1. A method for detecting phosphorylated tau (p-tau)217 in a biological sample using an immunoassay analyzer, the method comprising: aspirating, using a sample pipettor, a portion of the biological sample from a sample vessel and dispensing the aspirated biological sample into a reaction vessel of the immunoassay analyzer; wherein the immunoassay analyzer comprises: a reagent pack configured to hold a plurality of reagent vessels; a pipettor arrangement comprising at least one reagent pipettor and at least one sample pipettor; and a detector arrangement; aspirating, using the reagent pipettor, a portion of a first reagent from at least one reagent vessel and dispensing the aspirated reagent into the reaction vessel, generating a first reaction mixture; aspirating, using the reagent pipettor, a portion of a second reagent from at least one reagent vessel and dispensing the aspirated reagent into the reaction vessel, generating a second reaction mixture;
dispensing a substrate formulation into the reaction vessel, generating a detection mixture; wherein the at least one sample pipettor is configured to aspirate and/or dispense less than about 10 pL, alternatively about 2.0 pL to about 9.9 pL; and detecting, using the detector arrangement, a reaction in the detection mixture; and determining a presence and/or concentration of p-tau217 in the biological sample based on the reaction detected in the detection mixture.
2. The method of item 1, wherein the reaction in the detection mixture generates a chemiluminescent signal, and the method comprises correlating the chemiluminescent signal the presence and/or concentration of p-tau217 in the biological sample.
3. The method of item 1 or item 2, wherein the quantitation detection limit (LOQ) for measuring p-tau217 is equal to or less than about 3 pg/mL, alternatively equal to or less than about 2 pg/mL, equal to or less than about 1 pg/mL, equal to or less than about 0.6 pg/mL, and wherein the detected p-tau217 has a concentration above the LOQ.
4. The method of any one of items 1 to 3, wherein the detected p-tau217 has a concentration that is at least IX greater than the LOQ, alternatively at least 2X greater than the LOQ, or alternatively at least 3X greater than the LOQ.
5. The method of any one of items 1 to 4, wherein the detected p-tau217 exhibits a coefficient of variation (CV) of 20% or less, alternatively 15% or less, alternatively 10% or less, alternatively 5% or less, or alternatively 4% or less.
6. The method of any one of items 1 to 5, wherein cycle time is about 45 seconds or less, alternatively about 40 seconds or less, alternatively about 35 seconds or less, alternatively about 30 seconds or less, alternatively about 25 seconds or less, alternatively about 20 seconds or less, or alternatively about 15 seconds.
7. The method of any one of items 1 to 6, wherein time to first result (TTFR) is about 60 minutes or less, alternatively about 55 minutes or less, alternatively about 50 minutes or less, alternatively about 45 minutes or less, alternatively about 40 minutes or less, alternatively about 35 minutes or less, alternatively about 30 minutes or less, alternatively about 25 minutes or less, alternatively about 20 minutes or less, alternatively about 15 minutes or less, or alternatively about 10 minutes or less.
8. The method of any one of items 1 to 7, wherein incubation time of the first reaction mixture is at least about 30 minutes, alternatively at least about 40 minutes, or alternatively at least about 50 minutes.
9. The method of any one of item 1 to 8, wherein incubation time of the second reaction mixture is at least about 5 minutes, alternatively at least about 8 minutes, or alternatively at least about 10 minutes.
10. The method of any one of items 1 to 9, wherein the first reagent comprises at least one affinity molecule configured to bind to at least one portion p-tau217, the second reagent comprises at least one detection molecule, and the substrate formulation is configured to produce chemiluminescence.
11. The method of any one of items 1 to 10, wherein the substrate formulation is configured to produce chemiluminescence and comprises: a chemiluminescent compound of formula I or a salt thereof:
wherein
A is Ci-ehaloalkyl, naphthyl, phenyl, substituted phenyl, or heteroaryl, wherein substituted phenyl comprises from 1 to 3 halo, Ci-6 alkyl, Ci-6 alkoxy, Ci-6 haloalkyl, C(O)Ris, CN or NO2 substituents;
Ri is selected from the group consisting of Cs-uaryl, C1-6 alkyl, C1-6 haloalkyl, and C5-14 aralkyl groups;
R7-R14 are independently H, C1-6 alkoxy, halo, C1-4 alkyl, or R7 or R8-R9 or R9-R10 or R11-R12 or R12-R13 or R13-R14, can be joined together as a carbocyclic or heterocyclic ring system comprising at least one 5 or 6-membered ring;
R15 is C1-6 alkyl; ach M is independently selected from the group consisting of H, an alkali metal, alkaline earth metal, transition metal, ammonium, phosphonium, organic amine salt, and an amino acid salt;
Z is O or S; and n is 0, 1, or 2; a cationic aromatic compound (CAC); a background reducing agent; and an ether-linked nonionic surfactant or a hydrophilic polymer.
12. The method of item 10 or item 11, wherein the affinity molecule is selected from the group consisting of an antibody, a monoclonal antibody, a polyclonal antibody, a synthetic antibody mimic, an aptamer, an affimer, DARPins, or oligonucleotides or peptides that bind to at least one epitope of p-tau217.
13. The method of item 11, wherein the method further comprises exposing the biological sample to a first antibody which binds to a first p-tau217 epitope and a second antibody which binds to a second p-tau217 epitope.
14. The method of any one of items 10 to 12, wherein the detection molecule is an alkaline phosphatase (AP)-conjugated secondary antibody.
15. The method of item 14, wherein the affinity molecule is a capture antibody and the detection molecule is a detection antibody wherein the method further comprises exposing the biological sample to the capture antibody which binds to a first p-tau217 epitope and the detection antibody which binds to a second p-tau217 epitope.
16. The method of item 15, wherein the method further comprises exposing the biological sample to a second capture antibody which binds to a third p-tau217 epitope.
17. The method of any one of item 14 to 16, wherein the detection molecule binds to an epitope on p-Tau217 comprising amino acids 1 - 25, 1 - 20, 6 - 18, or 7 - 20 of SEQ ID NO: 1.
18. The method of any one of items 14 to 17, wherein the detection molecule binds to an epitope on p-Tau217 having at least 75% identical to the amino acid sequence set forth in SEQ ID NOs: 2, 3, or 4.
19. The method of any one of items 12 to 18, wherein the affinity molecule binds to an epitope on p-Tau217 comprising amino acids 217, 210 - 225, or 210 - 220 of SEQ ID NO: 1.
20. The method of any one of items 12 to 19, wherein the affinity molecule binds to an epitope on p-Tau217 having at least 75% identical to the amino acid sequence set forth in SEQ ID NO: 4.
21. The method of any one of items 10 to 20, wherein the affinity molecule and/or the detection molecule is conjugated to a magnetic bead or a magnetic particle.
22. The method of item 21, wherein the first reaction mixture, second reaction mixture, and/or detection mixture is subjected to a magnetic field prior to detection.
23. The method of any one of items 1 to 22, wherein the detector arrangement comprises a light detector configured to sense photons emitted from assay reactions over a period of time, an analog circuit configured to provide an analog signal based on the photons emitted from the assay reactions over the period of time, and a counter circuit configured to provide a photon count based on the photons emitted from the assay reactions over the period of time.
24. The method of any one of item 1 to 23, wherein the immunoassay analyzer further comprises an ultrasonic mixing module.
25. The method of item 24, wherein the first reaction mixture, second reaction mixture, and/or detection mixture is agitated via the ultrasonic mixing module.
26. The method of any one of items 1 to 25, wherein the immunoassay analyzer further comprises a washing arrangement, wherein the washing arrangement is configured to perform at least one wash action to wash away at least a portion of unreacted components, alternatively configured to perform at least two wash actions, alternatively configured to perform at least three wash actions, alternatively configured to perform at least four wash actions, or alternatively configured to perform at least five wash actions.
27. The method of item 26, wherein the washing arrangement is configured to wash away at least a portion of the unreacted components in the first reaction mixture, second reaction mixture, and/or detection mixture.
28. The method of any one of item 1 to 27, wherein the immunoassay analyzer further comprises:
a machine vision apparatus comprising an image capture device and an image interpretation device configured to monitor instrument and/or assay functionalities of the immunoassay analyzer.
29. The method of item 28, wherein the instrument functionalities are selected from the group consisting of optical sensors, pressure sensors and thermistors.
30. The method of item 29, wherein the assay functionalities are selected from the group consisting of sample volume monitoring, total reagent volume monitoring, residual volume monitoring, and particle retention monitoring.
31. The method of any one of items 1 to 30, wherein the biological sample is serum, whole blood, plasma, and/or cerebral spinal fluid.
32. The method of any one of items 1 to 31, wherein the method further comprises detecting at least one additional biomarker is selected from the group consisting of Ap peptides, Neurofilament light chain (NfL), total tau (t-tau), p-tau231, p-taul81, and combinations thereof.
33. The method of any one of items 1 to 32, wherein the reagent vessels comprise an elastomeric self-sealing membrane.
34. The method of any one of items 1 to 33, wherein the reagent pack further comprises containment walls arranged between the reagent vessels.
35. The method of any one of items 1 to 34, wherein the immunoassay analyzer further comprises a reagent storage unit, wherein the reagent pack is housed in the reagent storage unit.
36. A method for detecting phosphorylated tau (p-tau)217 in a biological sample using an immunoassay analyzer, the method comprising: aspirating, using a sample pipettor, a portion of the biological sample from a sample vessel and dispensing the aspirated sample into a reaction vessel of the immunoassay analyzer; wherein the immunoassay analyzer comprises: a reagent pack comprising a plurality of reagent vessels, wherein the reagent pack is configured to store a volume of reagent required for at least about 20 instances of a p- tau217 assay; a pipettor arrangement comprising at least one reagent pipettor and at a sample pipettor; and a detector arrangement; aspirating, using the reagent pipettor, a portion of a first reagent from at least one reagent vessel and dispensing the aspirated reagent into a reaction vessel of the immunoassay analyzer, generating a first reaction mixture; aspirating, using the reagent pipettor, a portion of a second reagent from at least one reagent vessel and dispensing the aspirated reagent into the reaction vessel, generating a second reaction mixture; dispensing a substrate formulation into the reaction vessel, generating a detection mixture; detecting, using the detector arrangement, a reaction in the detection mixture; and determining a presence and/or concentration of p-tau217 in the biological sample based on the reaction detected in the detection mixture.
37. The method of item 36, wherein the reaction in the detection mixture generates a chemiluminescent signal, wherein the chemiluminescent signal corresponds to the presence and/or concentration of p-tau217 in the biological sample.
38. The method of item 36 or item 37, wherein the quantitation detection limit (LOQ) for measuring p-tau217 is equal to or less than about 3 pg/mL, alternatively equal to or less than
about 2 pg/mL, equal to or less than about 1 pg/mL, equal to or less than about 0.6 pg/mL, and wherein the detected p-tau217 has a concentration above the LOQ.
39. The method of item 38, wherein the detected p-tau217 has a concentration that is at least IX greater than the LOQ, alternatively at least 2X greater than the LOQ, or alternatively at least 3X greater than the LOQ.
40. The method of any one of items 36 to 39, wherein the detected p-tau217 exhibits a coefficient of variation (CV) of 20% or less, alternatively 15% or less, alternatively 10% or less, alternatively 5% or less, or alternatively 4% or less.
41. The method of any one of items 36 to 40, wherein cycle time is about 45 seconds or less, alternatively about 40 seconds or less, alternatively about 35 seconds or less, alternatively about 30 seconds or less, alternatively about 25 seconds or less, alternatively about 20 seconds or less, or alternatively about 15 seconds.
42. The method of any one of items 36 to 41, wherein time to first result (TTFR) is about 60 minutes or less, alternatively about 55 minutes or less, alternatively about 50 minutes or less, alternatively about 45 minutes or less, alternatively about 40 minutes or less, alternatively about 35 minutes or less, alternatively about 30 minutes or less, alternatively about 25 minutes or less, alternatively about 20 minutes or less, alternatively about 15 minutes or less, or alternatively about 10 minutes or less.
43. The method of any one of items 36 to 42, wherein incubation time of the first reaction mixture is at least about 30 minutes, alternatively at least about 40 minutes, or alternatively at least about 50 minutes.
44. The method of any one of items 36 to 43, wherein incubation time of the second reaction mixture is at least about 5 minutes, alternatively at least about 8 minutes, or alternatively at least about 10 minutes.
45. The method of any one of items 36 to 44, wherein the first reagent comprises at least one affinity molecule configured to bind to at least one portion p-tau217, the second reagent comprises at least one detection molecule, and the substrate formulation is configured to produce chemiluminescence.
46. The method of any one of items 36 to 45, wherein the substrate formulation is configured to produce chemiluminescence and comprises: a chemiluminescent compound of formula I or a salt thereof:
wherein
A is Ci-ehaloalkyl, naphthyl, phenyl, substituted phenyl, or heteroaryl, wherein substituted phenyl comprises from 1 to 3 halo, Ci-6 alkyl, Ci-6 alkoxy, Ci-6 haloalkyl, C(O)Ris, CN or NO2 substituents;
Ri is selected from the group consisting of Cs-uaryl, C1-6 alkyl, C1-6 haloalkyl, and C5-14 aralkyl groups;
R7-R14 are independently H, C1-6 alkoxy, halo, C1-4 alkyl, or R7 or R8-R9 or R9-R10 or R11-R12 or R12-R13 or R13-R14, can be joined together as a carbocyclic or heterocyclic ring system comprising at least one 5 or 6-membered ring;
R15 is C1-6 alkyl; each M is independently selected from the group consisting of H, an alkali metal, alkaline earth metal, transition metal, ammonium, phosphonium, organic amine salt, and an amino acid salt;
Z is O or S; and n is 0, 1, or 2;
a cationic aromatic compound (CAC); a background reducing agent; and an ether-linked nonionic surfactant or a hydrophilic polymer.
47. The method of item 45 or item 46, wherein the affinity molecule is selected from the group consisting of an antibody, a monoclonal antibody, a polyclonal antibody, a synthetic antibody mimic, an aptamer, an affimer, DARPins, or oligonucleotides or peptides that bind to at least one epitope of p-tau217.
48. The method of item 47, wherein the method further comprises exposing the biological sample to a first antibody which binds to a first p-tau217 epitope and a second antibody which binds to a second p-tau217 epitope.
49. The method of any one of items 44 to 48, wherein the detection molecule is an alkaline phosphatase (AP)-conjugated secondary antibody.
50. The method of item 49, wherein the affinity molecule is a capture antibody and the detection molecule is a detection antibody wherein the method further comprises exposing the biological sample to the capture antibody which binds to a first p-tau217 epitope and the detection antibody which binds to a second p-tau217 epitope.
51. The method of item 50, wherein the method further comprises exposing the biological sample to a second capture antibody which binds to a third p-tau217 epitope.
52. The method of any one of items 45 to 51, wherein the detection molecule binds to an epitope on p-Tau217 comprising amino acids 1 - 25, 1 - 20, 6 - 18 or 7 - 20 of SEQ ID NO: 1.
53. The method of any one of items 45 to 52, wherein the detection molecule binds to an epitope on p-Tau217 having at least 75% identical to the amino acid sequence set forth in SEQ ID NOs: 2, 3, or 4.
54. The method of any one of items 45 to 53, wherein the affinity molecule binds to an epitope on p-Tau217 comprising amino acids 217, 210 - 225, or 210 - 220 of SEQ ID NO: 1.
55. The method of any one of items 45 to 54, wherein the affinity molecule binds to an epitope on p-Tau217 having at least 75% identical to the amino acid sequence set forth in SEQ ID NO: 4.
56. The method of any one of items 45 to 55, wherein the affinity molecule and/or the detection molecule is conjugated to a magnetic bead or a magnetic particle.
57. The method of item 56, wherein the first reaction mixture, second reaction mixture, and/or detection mixture is subjected to a magnetic field prior to detection.
58. The method of any one of items 36 to 57, wherein the detector arrangement comprises a light detector configured to sense photons emitted from assay reactions over a period of time, an analog circuit configured to provide an analog signal based on the photons emitted from the assay reactions over the period of time, and a counter circuit configured to provide a photon count based on the photons emitted from the assay reactions over the period of time.
59. The method of any one of items 36 to 58, wherein the immunoassay analyzer further comprises an ultrasonic mixing module.
60. The method of item 59, wherein the first reaction mixture, second reaction mixture, and/or detection mixture is agitated via the ultrasonic mixing module.
61. The method of any one of items 36 to 60, wherein the immunoassay analyzer further comprises a washing arrangement, wherein the washing arrangement is configured to perform at least one wash action to wash away at least a portion of unreacted components, alternatively configured to perform at least two wash actions, alternatively configured to perform at least three wash actions, alternatively configured to perform at least four wash actions, or alternatively configured to perform at least five wash actions.
62. The method of item 61, wherein the washing arrangement is configured to wash away at least a portion of the unreacted components in the first reaction mixture, second reaction mixture, and/or detection mixture.
63. The method of any one of items 36 to 62, wherein the immunoassay analyzer further comprises: a machine vision apparatus comprising an image capture device and an image interpretation device configured to monitor instrument and/or assay functionalities of the immunoassay analyzer.
64. The method of item 63, wherein the instrument functionalities are selected from the group consisting of optical sensors, pressure sensors and thermistors.
65. The method of item 64, wherein the assay functionalities are selected from the group consisting of sample volume monitoring, total reagent volume monitoring, residual volume monitoring, and particle retention monitoring.
66. The method of any one items 36 to 65, wherein the biological sample is serum, whole blood, plasma, and/or cerebral spinal fluid.
67. The method of any one items 36 to 66, wherein the method further comprises detecting at least one additional biomarker is selected from the group consisting of Ap peptides, Neurofilament light chain (NfL), total tau (t-tau), p-tau231, p-taul81, and combinations thereof.
68. A method for detecting p-tau217 in a biological sample using an immunoassay analyzer, the method comprising: mixing the biological sample with a reagent comprising at least one affinity molecule that binds to at least one portion p-tau217, generating a first reaction mixture; mixing the first reaction mixture with a reagent comprising a detection molecule, generating a second reaction mixture; mixing the second reaction mixture with a substrate formulation, generating a detection mixture; wherein the immunoassay analyzer comprises an ultrasonic mixing module, and the first reaction mixture, second reaction mixture and/or detection mixture are agitated via the ultrasonic mixing module; detecting, using a detector arrangement, a reaction in the detection mixture; and determining a presence and/or concentration of p-tau217 in the biological sample based on the reaction detected in the detection mixture.
69. The method of item 68, wherein the reaction in the detection mixture generates a chemiluminescent signal, wherein the chemiluminescent signal corresponds to the presence and/or concentration of p-tau217 in the biological sample.
70. The method of item 69, wherein the quantitation detection limit (LOQ) for measuring p-tau217 is equal to or less than about 3 pg/mL, alternatively equal to or less than about 2 pg/mL, equal to or less than about 1 pg/mL, equal to or less than about 0.6 pg/mL, and wherein the detected p-tau217 has a concentration above the LOQ.
71. The method of item 70, wherein the detected p-tau217 has a concentration that is at least IX greater than the LOQ, alternatively at least 2X greater than the LOQ, or alternatively at least 3X greater than the LOQ.
72. The method of any one of items 69 to 71, wherein the detected p-tau217 exhibits a coefficient of variation (CV) of 20% or less, alternatively 15% or less, alternatively 10% or less, alternatively 5% or less, or alternatively 4% or less.
73. The method of any one of items 69 to 72, wherein cycle time is about 45 seconds or less, alternatively about 40 seconds or less, alternatively about 35 seconds or less, alternatively about 30 seconds or less, alternatively about 25 seconds or less, alternatively about 20 seconds or less, or alternatively about 15 seconds.
74. The method of any one of items 69 to 73, wherein time to first result (TTFR) is about 60 minutes or less, alternatively about 55 minutes or less, alternatively about 50 minutes or less, alternatively about 45 minutes or less, alternatively about 40 minutes or less, alternatively about 35 minutes or less, alternatively about 30 minutes or less, alternatively about 25 minutes or less, alternatively about 20 minutes or less, alternatively about 15 minutes or less, or alternatively about 10 minutes or less.
75. The method of any one of items 69 to 74, wherein incubation time of the first reaction mixture is at least about 30 minutes, alternatively at least about 40 minutes, or alternatively at least about 50 minutes.
76. The method of any one of items 69 to 75, wherein incubation time of the second reaction mixture is at least about 5 minutes, alternatively at least about 8 minutes, or alternatively at least about 10 minutes.
77. The method of any one of items 69 to 76, wherein the first reagent comprises at least one affinity molecule configured to bind to at least one portion p-tau217, the second reagent
comprises at least one detection molecule, and the substrate formulation is configured to produce chemiluminescence.
78. The method of any one of items 69 to 77, wherein the substrate formulation is configured to produce chemiluminescence and comprises: a chemiluminescent compound of formula I or a salt thereof:
wherein
A is Ci-ehaloalkyl, naphthyl, phenyl, substituted phenyl, or heteroaryl, wherein substituted phenyl comprises from 1 to 3 halo, Ci-6 alkyl, Ci-6 alkoxy, Ci-6 haloalkyl, C(O)Ris, CN or NO2 substituents;
Ri is selected from the group consisting of Cs-uaryl, C1-6 alkyl, C1-6 haloalkyl, and C5-14 aralkyl groups;
R7-R14 are independently H, C1-6 alkoxy, halo, C1-4 alkyl, or R7 or R8-R9 or R9-R10 or R11-R12 or R12-R13 or R13-R14, can be joined together as a carbocyclic or heterocyclic ring system comprising at least one 5 or 6-membered ring;
R15 is C1-6 alkyl; ach M is independently selected from the group consisting of H, an alkali metal, alkaline earth metal, transition metal, ammonium, phosphonium, organic amine salt, and an amino acid salt;
Z is O or S; and n is 0, 1, or 2; a cationic aromatic compound (CAC); a background reducing agent; and
an ether-linked nonionic surfactant or a hydrophilic polymer.
79. The method of item 77 or item 78, wherein the affinity molecule is selected from the group consisting of an antibody, a monoclonal antibody, a polyclonal antibody, a synthetic antibody mimic, an aptamer, an affimer, DARPins, or oligonucleotides or peptides that bind to at least one epitope of p-tau217.
80. The method of item 79, wherein the method further comprises exposing the biological sample to a first antibody which binds to a first p-tau217 epitope and a second antibody which binds to a second p-tau217 epitope.
81. The method of any one of items 69 to 80, wherein the detection molecule is an alkaline phosphatase (AP)-conjugated secondary antibody.
82. The method of item 81, wherein the affinity molecule is a capture antibody and the detection molecule is a detection antibody wherein the method further comprises exposing the biological sample to the capture antibody which binds to a first p-tau217 epitope and the detection antibody which binds to a second p-tau217 epitope.
83. The method of item 82, wherein the method further comprises exposing the biological sample to a second capture antibody which binds to a third p-tau217 epitope.
84. The method of any one of items 76 to 83, wherein the detection molecule comprises a light chain variable region at least 75% identical to the amino acid sequence set forth as SEQ ID NO: 14.
85. The method of any one of items 74 to 84, wherein the detection molecule binds to an epitope on p-Tau217 comprising amino acids 1 - 25, 1 - 20, 6 - 18, or 7 - 20 of SEQ ID NO: 1.
86. The method of any one of items 74 to 85, wherein the detection molecule binds to an epitope on p-Tau217 having at least 75% identical to the amino acid sequence set forth in SEQ ID NOs: 2, 3, or 4.
87. The method of any one of items 74 to 86, wherein the affinity molecule binds to an epitope on p-Tau217 comprising amino acids 217, 210 - 225, or 210 - 220 of SEQ ID NO: 1.
88. The method of any one of items 74 to 87, wherein the affinity molecule binds to an epitope on p-Tau217 having at least 75% identical to the amino acid sequence set forth in SEQ ID NO: 4.
89. The method of any one of items 74 to 88, wherein the affinity molecule and/or the detection molecule is conjugated to a magnetic bead or a magnetic particle.
90. The method of item 89, wherein the first reaction mixture, second reaction mixture, and/or detection mixture is subjected to a magnetic field prior to detection.
91. The method of any one of item 69 to 90, wherein the detector arrangement comprises a light detector configured to sense photons emitted from assay reactions over a period of time, an analog circuit configured to provide an analog signal based on the photons emitted from the assay reactions over the period of time, and a counter circuit configured to provide a photon count based on the photons emitted from the assay reactions over the period of time.
92. The method of any one of items 69 to 91, wherein the immunoassay analyzer further comprises an ultrasonic mixing module.
93. The method of item 92, wherein the first reaction mixture, second reaction mixture, and/or detection mixture is agitated via the ultrasonic mixing module.
94. The method of any one of item 69 to 93, wherein the immunoassay analyzer further comprises a washing arrangement, wherein the washing arrangement is configured to perform at least one wash action to wash away at least a portion of unreacted components, alternatively configured to perform at least two wash actions, alternatively configured to perform at least three wash actions, alternatively configured to perform at least four wash actions, or alternatively configured to perform at least five wash actions.
95. The method of item 94, wherein the washing arrangement is configured to wash away at least a portion of the unreacted components in the first reaction mixture, second reaction mixture, and/or detection mixture.
96. The method of any one of items 69 to 95, wherein the immunoassay analyzer further comprises: a machine vision apparatus comprising an image capture device and an image interpretation device configured to monitor instrument and/or assay functionalities of the immunoassay analyzer.
97. The method of item 96, wherein the instrument functionalities are selected from the group consisting of optical sensors, pressure sensors and thermistors.
98. The method of item 97, wherein the assay functionalities are selected from the group consisting of sample volume monitoring, total reagent volume monitoring, residual volume monitoring, and particle retention monitoring.
99. The method of any one of items 69 to 98, wherein the biological sample is serum, whole blood, plasma, and/or cerebral spinal fluid.
100. The method of any one of items 69 to 99, wherein the method further comprises detecting at least one additional biomarker is selected from the group consisting of Ap peptides, Neurofilament light chain (NfL), total tau (t-tau), p-tau231, p-taul81, and combinations thereof.
101. A method for quantitatively determining an amount of phosphorylated tau (p-tau)217 in at least one plasma sample using a high-throughput analyzer, the method comprising: aspirating a portion of the at least one plasma sample from a sample vessel and dispensing the aspirated plasma sample into a first reaction vessel and second reaction vessel of an immunoassay analyzer, wherein the immunoassay analyzer further comprises: a pipettor arrangement comprising at least a first reagent pipettor, a second reagent pipettor, a third reagent pipettor and at least one sample pipettor; a plurality of reagent vessels; and a detector arrangement; aspirating, using the first reagent pipettor, a portion of a fluidic substance from a first reagent vessel and dispensing said fluidic substance into the first reaction vessel; aspirating, using the first reagent pipettor, a portion of a fluidic substance from a second reagent vessel and dispensing said fluidic substance into the first reaction vessel; dispensing a substrate into the first reaction vessel, generating a detection mixture; detecting, using the detector arrangement, a presence of a reaction in the detection; and quantitatively determining an amount of p-tau217 in the plasma based on the presence of the reaction in the detection mixtures; wherein the second reagent pipettor and third reagent pipettor are configured to dispense reagents into second and third reaction vessels, simultaneously, as the first reagent pipettor dispenses fluid into the first reaction vessel.
102. The method of item 100, wherein the first reagent pipettor, the second reagent pipettor, and/or the third reagent pipettor are selectively and/or simultaneously operated.
103. The method of item 100 or item 102, wherein the first reagent pipettor, second reagent pipettor, third reagent pipettor, and/or sample pipettor are configured to engage a dispense tip prior to aspiration.
104. The method of any one of items 100 to 103, wherein the method is configured to analyze at least about 200 plasma samples/hr.
105. The method of any one of items 100 to 103, wherein the method is configured to analyze at least about 300 plasma samples/hr.
106. The method of any one of items 100 to 103, wherein the method is configured to analyze at least about 400 plasma samples/hr.
107. The method of any one of items 100 to 106, wherein the reaction in the detection mixture generates a chemiluminescent signal, wherein the chemiluminescent signal quantitatively corresponds to a concentration of p-tau217 in the plasma sample.
108. The method of any one of items 100 to 107, wherein the quantitation detection limit (LOQ) for measuring p-tau217 is equal to or less than about 3 pg/mL, alternatively equal to or less than about 2 pg/mL, equal to or less than about 1 pg/mL, equal to or less than about 0.6 pg/mL, and wherein the p-tau217 has a concentration above the LOQ.
109. The method of any one of items 100 to 107, wherein the quantitation detection limit (LOQ) for measuring p-tau217 is equal to or less than about 2 pg/mL.
110. The method of any one of items 100 to 107, wherein the quantitation detection limit (LOQ) for measuring p-tau217 is equal to or less than about 1 pg/mL.
111. The method of any one of items 100 to 107, wherein the quantitation detection limit (LOQ) for measuring p-tau217 is equal to or less than about 0.6 pg/mL, and wherein the p- tau217 has a concentration above the LOQ.
112. The method of any one of items 100 to 107, wherein the quantitation detection limit (LOQ) for measuring p-tau217 is equal to or less than about 50 fg/mL.
113. The method of any one of items 100 to 107, wherein the quantitation detection limit (LOQ) for measuring p-tau217 is equal to or less than about 15 fg/mL.
114. The method of any one of items 100 to 107, wherein the quantitation detection limit (LOQ) for measuring p-tau217 is equal to or less than about 10 fg/mL, and wherein the p- tau217 has a concentration above the LOQ.
115. The method of any one of items 100 to 114, wherein the detected p-tau217 has a concentration that is at least IX greater than the LOQ, alternatively at least 2X greater than the LOQ, or alternatively at least 3X greater than the LOQ.
116. The method of any one of items 100 to 115, wherein the p-tau217 exhibits a coefficient of variation (CV) of 20% or less.
117. The method of any one of items 100 to 115, wherein the p-tau217 exhibits a coefficient of variation (CV) of 4% or less.
118. The method of any one of items 100 to 117, wherein cycle time is about 45 seconds or less, alternatively about 40 seconds or less, alternatively about 35 seconds or less, alternatively about 30 seconds or less, alternatively about 25 seconds or less, alternatively about 20 seconds or less, or alternatively about 15 seconds.
119. The method of any one of items 100 to 118, wherein time to first result (TFFR) is about 45 minutes or less, alternatively about 40 minutes or less, alternatively about 35 minutes or less, alternatively about 30 minutes or less, alternatively about 25 minutes or less, alternatively about 20 minutes or less, alternatively about 15 minutes or less, or alternatively about 10 minutes or less.
120. The method of any one of items 100 to 119, wherein the immunoassay analyzer comprises a fourth reaction vessel and the pipettor arrangement comprises a fourth reagent pipettor, wherein the method further includes aspirating a portion of the at least one plasma sample from a sample vessel and dispensing the aspirated plasma sample into the fourth reaction vessel; aspirating, using the fourth reagent pipettor, a portion of a fluidic substance from a first reagent vessel and dispensing said fluidic substance into the fourth reaction vessel; aspirating, using the fourth reagent pipettor, a portion of a fluidic substance from a second reagent vessel and dispensing said fluidic substance into the fourth reaction vessel; dispensing a substrate into the fourth reaction vessel, generating a detection mixture; detecting, using the detector arrangement, a presence of a reaction in the detection mixture in the fourth reaction vessel; and quantitatively determining an amount of p-tau217 in the plasma based on the presence of the reaction in the detection mixture.
121. The method of items 120, wherein the first reagent pipettor, the second reagent pipettor, the third reagent pipettor and the fourth reagent pipettor are selectively and/or simultaneously operated.
122. The method of item 190 and 120, wherein the fourth reagent pipettor is configured to engage a dispense tip prior to aspiration.
123. The method of any one of items 190 to 122, wherein cycle time is about 45 seconds or less, alternatively about 40 seconds or less, alternatively about 35 seconds or less, alternatively about 30 seconds or less, alternatively about 25 seconds or less, alternatively about 20 seconds or less, or alternatively about 15 seconds.
124. The method of any one of items 190 to 123, wherein time to first result (TFFR) is about 45 minutes or less, alternatively about 40 minutes or less, alternatively about 35 minutes or less, alternatively about 30 minutes or less, alternatively about 25 minutes or less, alternatively about 20 minutes or less, alternatively about 15 minutes or less, or alternatively about 10 minutes or less.
125. The method of any one of items 100 to 124, wherein dispensing the fluidic substance from the first reagent vessel into the first reaction vessel, second reaction vessel, third reaction vessel, and/or fourth reaction vessel generates a first reaction mixture and incubation time of the first reaction mixture is at least about 30 minutes, alternatively at least about 40 minutes, or alternatively at least about 50 minutes.
126. The method of items 125, wherein the first reaction mixture in the first reaction vessel, second reaction vessel, third reaction vessel, and/or fourth reaction vessel are incubated simultaneously.
127. The method of any one of items 100 to 126, wherein dispensing the fluidic substance from the second reagent vessel into the first reaction vessel, second reaction vessel, third reaction vessel, and/or fourth reaction vessel generates a second reaction mixture and incubation time of the second reaction mixture is at least about 5 minutes, alternatively at least about 8 minutes, or alternatively at least about 10 minutes.
128. The method of item 127, wherein the second reaction mixture in the first reaction vessel, second reaction vessel, third reaction vessel, and/or fourth reaction vessel are incubated simultaneously.
129. The method of any one of items 100 to 128, wherein the method is configured to simultaneously perform at least two assays for a plurality of plasma samples, alternatively at least three assays for a plurality of plasma samples, or at least four assays for a plurality of plasma samples.
130. The method of item 129, wherein the plasma samples are from the same individual or from multiple individuals.
131. The method of item 129 or item 130, wherein the method comprises a plurality of sample vessels.
132. The method of any one of items 129 to 131, wherein at least one of the assays is p- tau217 assay, alternatively at least two of the assays are p-tau217 assays, alternatively at least three of the assays are p-tau217 assays, or alternatively at least four of the assays are p-tau217 assays.
133. The method of any one of items 129 to 131, wherein at least one of the assays is p- tau217 assay and at least one of the assays is a second analyte assay, wherein the second analyte is selected from the group consisting of Ap peptides, Neurofilament light chain (NfL), total tau (t-tau), p-tau231, p-taul81, and combinations thereof.
134. The method of any one of item 100 to 133, wherein the fluidic substance from the first reagent vessel comprises at least one affinity molecule configured to bind to at least one portion p-tau217, the fluidic substance from the second reagent vessel comprises at least one
detection molecule, and the fluidic substance from the third reagent vessel comprises a substrate formulation configured to produce chemiluminescence.
135. The method of item 134, wherein the affinity molecule is selected from the group consisting of an antibody, a monoclonal antibody, a polyclonal antibody, a synthetic antibody mimic, an aptamer, an affimer, DARPins, or oligonucleotides or peptides that bind to at least one epitope of p-tau217.
136. The method of item 135, wherein the affinity molecule is an antibody and the method further comprises exposing the plasma sample to a first antibody which binds to a first p- tau217 epitope and a second antibody which binds to a second p-tau217 epitope.
137. The method of any one of items 134 to 136, wherein the detection molecule is an alkaline phosphatase (AP)-conjugated secondary antibody.
38. The method of any one of items 134 to 137, wherein the affinity molecule and/or the detection molecule is conjugated to a magnetic bead or a magnetic particle.
139. The method of any one of items 100 to 138, wherein the first reagent vessel, second reagent vessel and/or third reagent vessel are housed in a reagent pack.
140. The method of item 139, wherein the reagent pack further comprises containment walls arranged between the reagent vessels.
141. The method of item 139 or item 140, wherein the reagent vessels comprise an elastomeric self-sealing membrane.
142. The method of any one of items 139 to 141, wherein the immunoassay analyzer further comprises a reagent storage unit, wherein the reagent pack is housed in the reagent storage unit.
143. The method of any one of items 139 to 142, wherein each reagent vessel is configured to store a volume of reagent required for at least about 20 instances of a p-tau217 assay.
144. The method of any one of items 100 to 143, wherein the immunoassay analyzer further comprises: a machine vision apparatus comprising an image capture device and an image interpretation device configured to monitor instrument and/or assay functionalities of the immunoassay analyzer.
145. The method of item 144, wherein the instrument functionalities are selected from the group consisting of optical sensors, pressure sensors and thermistors.
146. The method of item 144, wherein the assay functionalities are selected from the group consisting of sample volume monitoring, total reagent volume monitoring, residual volume monitoring, and particle retention monitoring.
147. The method of any one of items 100 to 146, wherein the immunoassay analyzer further comprises an ultrasonic mixing module.
148. The method of item 47, wherein the first reaction vessel, second reaction vessel, third reaction vessel, and/or fourth reaction vessel are agitated via the ultrasonic mixing module.
149. The method of any one of items 100 to 148, wherein the immunoassay analyzer further comprises a washing arrangement,
wherein the washing arrangement is configured to perform at least one wash action to wash away at least a portion of unreacted components, alternatively configured to perform at least two wash actions, alternatively configured to perform at least three wash actions, alternatively configured to perform at least four wash actions, or alternatively configured to perform at least five wash actions.
150. The method of item 149, wherein the washing arrangement is configured to wash away at least a portion of the unreacted components in the first reaction mixture, second reaction mixture, and/or detection mixture.
[0307] It will be understood that while the invention has been described in conjunction with the detailed description thereof, the foregoing description is intended to illustrate and not limit the scope of the invention, which is defined by the scope of the appended claims. Other aspects, advantages, and modifications are within the scope of the following claims.
Claims
1. A method for quantitatively determining an amount of phosphorylated tau (p-tau) 217 in at least one plasma sample using a high-throughput immunoassay analyzer, the method comprising: aspirating a portion of the at least one plasma sample from a sample vessel and dispensing the aspirated plasma sample into a first reaction vessel of the immunoassay analyzer, wherein the immunoassay analyzer comprises: a pipettor arrangement comprising at least a first reagent pipettor and a second reagent pipettor; and a detector arrangement; detecting, using the detector arrangement, a presence of a reaction in the first reaction vessel; and quantitatively determining an amount of p-tau217 in the plasma based on the presence of the reaction; and wherein the second reagent pipettor is configured to dispense reagents into a second reaction vessel simultaneously as the first reagent pipettor dispenses fluid into the first reaction vessel.
2. The method of claim 1, wherein the method is configured to analyze at least about 200 plasma samples/hr.
3. The method of any one of claims 1 or 2, wherein the method is configured to analyze at least about 300 plasma samples/hr.
4. The method of any one of claims 1 to 3, wherein the method is configured to analyze at least about 400 plasma samples/hr.
5. The method of any one of claims 1 to 4, wherein the reaction in the first reaction vessel generates a chemiluminescent signal, wherein the chemiluminescent signal quantitatively corresponds to a concentration of p-tau217 in the plasma sample.
6. The method of any one of claims 1 to 5, wherein the quantitation detection limit (LOQ) for measuring p-tau217 is equal to or less than about 1 pg/mL.
7. The method of any one of claims 1 to 6, wherein the quantitation detection limit (LOQ) for measuring p-tau217 is equal to or less than about 50 fg/mL.
8. The method of any one of claims 1 to 7, wherein the quantitative determination of p- tau217 exhibits a coefficient of variation (CV) of 20% or less.
9. The method of any one of claims 1 to 8, wherein the immunoassay analyzer further comprises a third reagent pipettor, and wherein the second reagent pipettor and third reagent pipettor are configured to dispense reagents into the second reaction vessel and a third reaction vessel simultaneously.
10. The method of any one of claims 1 to 9, further comprising aspirating, using the first reagent pipettor, a portion of a fluidic substance from a first reagent vessel and dispensing said fluidic substance into the first reaction vessel, wherein the fluidic substance from the first reagent vessel comprises at least one affinity molecule configured to bind to at least one portion p-tau217; aspirating, using the first reagent pipettor, a portion of a fluidic substance from a second reagent vessel and dispensing said fluidic substance into the first reaction vessel, wherein the fluidic substance from the second reagent vessel comprises at least one detection molecule; and dispensing a substrate into the first reaction vessel.
11. The method of any one of claims 1 to 10, wherein the first reagent vessel and second reagent vessel are housed in a reagent pack.
12. The method of claim 11, wherein the reagent vessels comprise an elastomeric selfsealing membrane.
13. The method of any one of claims 11 or 12, wherein each reagent vessel is configured to store a volume of reagent required for at least about 20 instances of a p-tau217 assay.
14. The method of any one of claims 1 to 13, wherein the immunoassay analyzer further comprises: a machine vision apparatus comprising an image capture device and an image interpretation device configured to monitor instrument and/or assay functionalities of the immunoassay analyzer.
15. The method of any one of claims 1 to 14, wherein the immunoassay analyzer further comprises a washing arrangement, wherein the washing arrangement is configured to perform at least one wash action to wash away at least a portion of unreacted components, alternatively configured to perform at least two wash actions, alternatively configured to perform at least three wash actions, alternatively configured to perform at least four wash actions, or alternatively configured to perform at least five wash actions.
16. A method for detecting phosphorylated tau (p-tau) 217 in a biological sample using an immunoassay analyzer, the method comprising: aspirating, using a sample pipettor, a portion of the biological sample from a sample vessel and dispensing the aspirated biological sample into a reaction vessel of the immunoassay analyzer; wherein the immunoassay analyzer comprises: a pipettor arrangement comprising at least one reagent pipettor and at least one sample pipettor; and a detector arrangement; wherein the sample pipettor is configured to aspirate and/or dispense less than about 10 pL, alternatively about 2.0 pL to about 9.9 pL; detecting, using the detector arrangement, a presence of a reaction in a reaction vessel; and determining a presence and/or concentration of p-tau217 in the biological sample based on the reaction detected in the reaction vessel.
17. A method for detecting phosphorylated tau (p-tau)217 in a biological sample using an immunoassay analyzer, the method comprising: aspirating, using a sample pipettor, a portion of the biological sample from a sample vessel and dispensing the aspirated sample into a reaction vessel of the immunoassay analyzer; wherein the immunoassay analyzer comprises: a reagent pack comprising a plurality of reagent vessels, wherein the reagent pack is configured to store a volume of reagent required for at least about 20 instances of a p-tau217 assay; a pipettor arrangement comprising at least one reagent pipettor and at a sample pipettor; and a detector arrangement;
aspirating, using the reagent pipettor, a portion of a first reagent from at least one reagent vessel and dispensing the aspirated reagent into a reaction vessel of the immunoassay analyzer, generating a first reaction mixture; aspirating, using the reagent pipettor, a portion of a second reagent from at least one reagent vessel and dispensing the aspirated reagent into the reaction vessel, generating a second reaction mixture; detecting, using the detector arrangement, a reaction in the reaction vessel; and determining a presence and/or concentration of p-tau217 in the biological sample based on the reaction detected in the reaction vessel.
18. A method for detecting p-tau217 in a biological sample using an immunoassay analyzer, the method comprising: mixing the biological sample with a reagent comprising at least one affinity molecule that binds to at least one portion p-tau217, generating a first reaction mixture; mixing the first reaction mixture with a reagent comprising a detection molecule, generating a detection mixture; wherein the immunoassay analyzer comprises an ultrasonic mixing module, and the first reaction mixture and/or the detection mixture are agitated via the ultrasonic mixing module; detecting, using a detector arrangement, a reaction in the detection mixture; and determining a presence and/or concentration of p-tau217 in the biological sample based on the reaction detected in the detection mixture.
Applications Claiming Priority (16)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US202363596578P | 2023-11-06 | 2023-11-06 | |
| US202363596581P | 2023-11-06 | 2023-11-06 | |
| US63/596,581 | 2023-11-06 | ||
| US63/596,578 | 2023-11-06 | ||
| US202363607742P | 2023-12-08 | 2023-12-08 | |
| US202363607747P | 2023-12-08 | 2023-12-08 | |
| US63/607,747 | 2023-12-08 | ||
| US63/607,742 | 2023-12-08 | ||
| US202463642605P | 2024-05-03 | 2024-05-03 | |
| US202463642601P | 2024-05-03 | 2024-05-03 | |
| US63/642,601 | 2024-05-03 | ||
| US63/642,605 | 2024-05-03 | ||
| US202463676889P | 2024-07-29 | 2024-07-29 | |
| US202463676890P | 2024-07-29 | 2024-07-29 | |
| US63/676,889 | 2024-07-29 | ||
| US63/676,890 | 2024-07-29 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2025101478A1 true WO2025101478A1 (en) | 2025-05-15 |
Family
ID=93704630
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2024/054505 Pending WO2025101478A1 (en) | 2023-11-06 | 2024-11-05 | An automated and high-troughput analyzer method for a phosphorylated tau (p-tau) 217 immunoassay |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2025101478A1 (en) |
Citations (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6139782A (en) | 1996-01-16 | 2000-10-31 | Lumigen, Inc. | Compounds, compositions and methods for generating chemiluminescence with phosphatase enzymes |
| US6457270B1 (en) | 2000-10-27 | 2002-10-01 | Frederick W. Stark, III | Universal emergency sign |
| US6905710B2 (en) | 2001-08-31 | 2005-06-14 | Council Of Scientific And Industrial Research | Pharmaceutical composition useful for inhibition of osteoclast formation and a process for the extraction of mussel hudrolysate from indian green mussel |
| US20060180529A1 (en) * | 2005-02-11 | 2006-08-17 | Emilio Barbera-Guillem | Bioreactor for selectively controlling the molecular diffusion between fluids |
| US7128874B2 (en) | 2001-01-26 | 2006-10-31 | Beckman Coulter, Inc. | Method and system for picking and placing vessels |
| US9519000B2 (en) | 2010-07-23 | 2016-12-13 | Beckman Coulter, Inc. | Reagent cartridge |
| US10703971B2 (en) | 2016-06-30 | 2020-07-07 | Beckman Coulter, Inc. | Chemiluminescent substrates |
| CN113533746A (en) * | 2021-07-22 | 2021-10-22 | 深圳市天大生物医疗器械有限公司 | P-Tau protein chemiluminescence detection kit and preparation method thereof |
| US11263433B2 (en) | 2016-10-28 | 2022-03-01 | Beckman Coulter, Inc. | Substance preparation evaluation system |
| CN114778856A (en) * | 2022-05-30 | 2022-07-22 | 苏州宇测生物科技有限公司 | Phosphorylated tau protein detection kit |
| US20220357352A1 (en) | 2019-07-03 | 2022-11-10 | Beckman Coulter, Inc. | Configurable wash process for a sample analyzer |
| US11604146B2 (en) | 2017-09-19 | 2023-03-14 | Beckman Coulter, Inc. | Analog light measuring and photon counting with a luminometer system for assay reactions in chemiluminescence measurements |
-
2024
- 2024-11-05 WO PCT/US2024/054505 patent/WO2025101478A1/en active Pending
Patent Citations (15)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6218137B1 (en) | 1996-01-16 | 2001-04-17 | Lumigen, Inc. | Compounds, compositions and methods for generating chemiluminescene with phosphatase enzymes |
| US6270695B1 (en) | 1996-01-16 | 2001-08-07 | Lumigen, Inc. | Compounds, compositions, and methods for generating chemiluminescence with phosphatase enzymes |
| US6296787B1 (en) | 1996-01-16 | 2001-10-02 | Lumigen, Inc. | Compounds, compositions and methods for generating chemiluminescence with phosphatase enzymes |
| US6139782A (en) | 1996-01-16 | 2000-10-31 | Lumigen, Inc. | Compounds, compositions and methods for generating chemiluminescence with phosphatase enzymes |
| US6457270B1 (en) | 2000-10-27 | 2002-10-01 | Frederick W. Stark, III | Universal emergency sign |
| US7128874B2 (en) | 2001-01-26 | 2006-10-31 | Beckman Coulter, Inc. | Method and system for picking and placing vessels |
| US6905710B2 (en) | 2001-08-31 | 2005-06-14 | Council Of Scientific And Industrial Research | Pharmaceutical composition useful for inhibition of osteoclast formation and a process for the extraction of mussel hudrolysate from indian green mussel |
| US20060180529A1 (en) * | 2005-02-11 | 2006-08-17 | Emilio Barbera-Guillem | Bioreactor for selectively controlling the molecular diffusion between fluids |
| US9519000B2 (en) | 2010-07-23 | 2016-12-13 | Beckman Coulter, Inc. | Reagent cartridge |
| US10703971B2 (en) | 2016-06-30 | 2020-07-07 | Beckman Coulter, Inc. | Chemiluminescent substrates |
| US11263433B2 (en) | 2016-10-28 | 2022-03-01 | Beckman Coulter, Inc. | Substance preparation evaluation system |
| US11604146B2 (en) | 2017-09-19 | 2023-03-14 | Beckman Coulter, Inc. | Analog light measuring and photon counting with a luminometer system for assay reactions in chemiluminescence measurements |
| US20220357352A1 (en) | 2019-07-03 | 2022-11-10 | Beckman Coulter, Inc. | Configurable wash process for a sample analyzer |
| CN113533746A (en) * | 2021-07-22 | 2021-10-22 | 深圳市天大生物医疗器械有限公司 | P-Tau protein chemiluminescence detection kit and preparation method thereof |
| CN114778856A (en) * | 2022-05-30 | 2022-07-22 | 苏州宇测生物科技有限公司 | Phosphorylated tau protein detection kit |
Non-Patent Citations (5)
| Title |
|---|
| AD. TELSER, J ET AL., CLIN CHIM ACTA., vol. 531, 1 June 2022 (2022-06-01), pages 100 - 111 |
| AM J MANAG CARE., vol. 28, no. 10, 2022, pages S188 - S196 |
| HARDY JA ET AL.: "Alzheimer's disease: the amyloid cascade hypothesis", SCIENCE, vol. 256, 1992, pages 184 - 5 |
| MORRISON ET AL., PROC. NATL. ACAD. SCI. USA, vol. 81, 1984, pages 6851 - 6855 |
| WATTMO, C. ET AL.: "Cerebro-spinal fluid biomarker levels: phosphorylated tau (T) and total tau (N) as markers for rate of progression in Alzheimer's disease", BMC NEUROL, vol. 20, 2020, pages 10 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US10732111B2 (en) | Automated immunoanalyzer system for performing diagnostic assays for allergies and autoimmune diseases | |
| JP5005511B2 (en) | Immunodiagnostics with reduced non-specific reactions | |
| US20210190771A1 (en) | Automated liquid immunoassay device and method therefor | |
| EP3004882B1 (en) | Reagents, methods and devices to prevent aggregation in particle based tests for the detection of multimeric target molecules | |
| US20220397528A1 (en) | Systems and methods for rapid, sensitive multiplex immunoassays | |
| CN110646620A (en) | Method for simultaneously detecting four inflammation markers and kit thereof | |
| US9939448B2 (en) | Methods for detecting insulin autoantibody | |
| US20120258553A1 (en) | Analyte measurement apparatus and method | |
| TW202040132A (en) | Methods for suppressing abnormal detections in immunoassays conducted with an automatic analyzer, detection methods, immunoassay reagents, and immunoassay reagent kits | |
| WO2025101478A1 (en) | An automated and high-troughput analyzer method for a phosphorylated tau (p-tau) 217 immunoassay | |
| US20210341479A1 (en) | Quantitative antibody test | |
| US12222351B2 (en) | Immunoassay and measurement reagent for leucine-rich alpha2 glycoprotein | |
| JP7128196B2 (en) | Systems and methods for classification of biological samples for the presence of analytes | |
| WO2025160022A1 (en) | Detecting phosphorylated tau (p-tau) 217 using chemiluminescent dioxetane compounds | |
| WO2025160030A1 (en) | Chemiluminescent dioxetane compounds for detecting p24 antigen | |
| WO2025160017A1 (en) | Assay methods using chemiluminescent dioxetane compounds | |
| CN117470819A (en) | Autoimmune myositis disease detection kit and detection method thereof | |
| WO2025160104A1 (en) | DETECTING INTERFERON GAMMA ("IFN-γ") USING CHEMILUMINESCENT DIOXETANE COMPOUNDS | |
| US20220326271A1 (en) | Methods for onboard dilutions using automated analyzer | |
| CN101084441A (en) | Device for carrying out an individual immunoassay in a fully automatic manner | |
| CN115667928A (en) | Methods, systems, articles, kits and uses thereof for detection of biomolecules, bioorganelles, bioparticles, cells and microorganisms | |
| KR102893369B1 (en) | Method of Performing a Liquid Immunoassay Using an In Vitro Diagnostic Cartridge Equipped With a Magnetic Bead Well, a Reaction Well, a Washing Well, and a Measurement Well | |
| CN105445474A (en) | Anti-SS-A(Ro60) antibody kit and detection method thereof | |
| WO2025128956A1 (en) | High-throughput immunoassay methods for assessing the stages of ovarian aging | |
| JP6955261B2 (en) | Microarrays, microarray manufacturing methods, inspection methods, and inspection kits |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 24813531 Country of ref document: EP Kind code of ref document: A1 |