WO2025099280A1 - Improved anti-ox40l antibodies - Google Patents
Improved anti-ox40l antibodies Download PDFInfo
- Publication number
- WO2025099280A1 WO2025099280A1 PCT/EP2024/081745 EP2024081745W WO2025099280A1 WO 2025099280 A1 WO2025099280 A1 WO 2025099280A1 EP 2024081745 W EP2024081745 W EP 2024081745W WO 2025099280 A1 WO2025099280 A1 WO 2025099280A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- antibody
- cell
- seq
- fucose
- fucosylation
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P37/00—Drugs for immunological or allergic disorders
- A61P37/02—Immunomodulators
- A61P37/06—Immunosuppressants, e.g. drugs for graft rejection
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
- C07K16/18—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans
- C07K16/28—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants
- C07K16/2875—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants against the NGF/TNF superfamily, e.g. CD70, CD95L, CD153, CD154
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K2039/505—Medicinal preparations containing antigens or antibodies comprising antibodies
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/10—Immunoglobulins specific features characterized by their source of isolation or production
- C07K2317/14—Specific host cells or culture conditions, e.g. components, pH or temperature
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/40—Immunoglobulins specific features characterized by post-translational modification
- C07K2317/41—Glycosylation, sialylation, or fucosylation
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/50—Immunoglobulins specific features characterized by immunoglobulin fragments
- C07K2317/56—Immunoglobulins specific features characterized by immunoglobulin fragments variable (Fv) region, i.e. VH and/or VL
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/70—Immunoglobulins specific features characterized by effect upon binding to a cell or to an antigen
- C07K2317/72—Increased effector function due to an Fc-modification
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/70—Immunoglobulins specific features characterized by effect upon binding to a cell or to an antigen
- C07K2317/73—Inducing cell death, e.g. apoptosis, necrosis or inhibition of cell proliferation
- C07K2317/732—Antibody-dependent cellular cytotoxicity [ADCC]
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/70—Immunoglobulins specific features characterized by effect upon binding to a cell or to an antigen
- C07K2317/76—Antagonist effect on antigen, e.g. neutralization or inhibition of binding
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/90—Immunoglobulins specific features characterized by (pharmaco)kinetic aspects or by stability of the immunoglobulin
- C07K2317/92—Affinity (KD), association rate (Ka), dissociation rate (Kd) or EC50 value
Definitions
- the present invention relates to antibodies and antibody derivatives against OX40L with improved effects compared to prior art antibodies
- T-cells Activation of T-cells occurs through two cell-mediated signals, firstly the MHC (presenting the foreign antigen) and TOR interaction, and secondly through costimulatory molecules (OX40/OX40L, CD40L/CD40, etc.). The second signal is necessary for the activation of T-cells. Thirdly, soluble factors such as cytokines shape the final T-cell response.
- Human OX40L (gp34, CD252, SwissProt P23510), belongs to the tumour necrosis factor superfamily (TNFSF) group and can be expressed on B-cells, dendritic cells, macrophages, monocytes, endothelial cells, fibroblasts (Review: Weinberg, A. D., Trends Immunol. 23 (2002) 102-109). Its cognate receptor, 0X40, is expressed on T- cells. OX40/OX40L is a co-stimulatory pathway. It is not constitutively expressed but rather induced upon activation of T-cells after around 2-3 days.
- TNFSF tumour necrosis factor superfamily
- Administration of the antibody dramatically ameliorated the disease severity.
- This antibody showed similar activities in other related disease models, e.g., inflammatory skin disease, experimental autoimmune disease (EAE), GvHD, and inflammatory bowel disease.
- Adverse T-cell activation is a pathological driver in several diseases such as autoimmune disease ⁇ e.g., diabetes, multiple sclerosis, SLE, rheumatoid arthritis), inflammatory conditions (asthma, atopic dermatitis, chronic rhinosinusitis, inflammatory bowel disease), transplant-related conditions (graft-versus-host disease, allograft rejection).
- autoimmune disease e.g., diabetes, multiple sclerosis, SLE, rheumatoid arthritis
- inflammatory conditions as atopic dermatitis, chronic rhinosinusitis, inflammatory bowel disease
- transplant-related conditions graft-versus-host disease, allograft rejection
- Monoclonal antibodies can have a variety of post-translational modifications, including glycosylation.
- Monoclonal antibodies such as IgGs, have an N-linked glycosylation site at asparagine 297 (Asn297) of each heavy chain (two per intact antibody).
- the glycans attached to Asn297 on antibodies are typically complex biantennary structures with very low or no bisecting N-acetylglucosamine (bisecting GIcNAc) with low amounts of terminal sialic acid and variable amounts of galactose.
- the glycans also usually have high levels of core fucosylation. Reduction of core fucosylation in antibodies has been shown to alter Fc receptor affinity, altering the Fc-mediated effector functions.
- Antibodies against human OX40L are known.
- One example is Oxelumab (clone R4930), which previously underwent development for the treatment of asthma.
- the antibody is described in US 7501496 B1.
- Amlitelimab (also known as KY1005/SAR445229) is another anti-OX40L antibody for the treatment of atopic dermatitis. It is described in US9139653B1 and WO2015/132580 A1. Amlitelimab is an lgG4 isotype, non-depleting antibody. In WO 2018/083248, the effect of Amlitelimab in a primate GVHD model is shown.
- the invention provides antibodies and antibody derivatives and methods for producing anti-OX40L antibodies and antibody derivatives with reduced core fucosylation.
- the antibodies of the invention are Fc engineered such that the affinities for various Fey receptors are increased.
- the antibodies and antibody derivatives and methods are premised in part on the unexpected results presented in the examples showing that affinity maturation and optimisation of the Fc-region to reduce core fucosylation, produces antibodies and antibody derivatives with improved affinity to OX40L and Fc-receptors.
- Such antibodies and antibody derivatives exhibit increased effector function (ADCC), reduced T-cell activation, enriched regulatory T cell (T-Reg) populations, and maintenance of naive immune cell populations, as compared with antibodies or antibody derivatives present in the prior art. Said improved features are observed both in vitro and in vivo.
- the antibodies and antibody derivatives and methods of the invention exhibit elevated affinity for the human OX40L antigen as compared to a known anti-OX40L antibody; Oxelumab.
- the antibodies and antibody derivatives and methods of the invention exhibit an unexpectedly significant reduction in the proliferation of CD4+ and CD8+ T-cells, as compared to antibodies and other binding proteins of the prior art which are known to manipulate activation of T-cells via APC targeting (Belatacept, Oxelumab and Amlitelimab).
- the antibodies and antibody derivatives and methods of the invention induce a significant increase in the population of T-Regs and naive CD4+ T-cells among the proliferating cell population, as compared to antibodies and other binding proteins of the prior art which are known to manipulate activation of T-cells via APC targeting (Belatacept, Oxelumab and Amlitelimab). These data are unexpected as the level of T cell control and induction of a regulatory T cell phenotype, among proliferating cells, has not been described previously. Concomitant to this, the antibodies and antibody derivatives and methods of the invention reduce TNFa, and IL-4 levels compared with control and another OX40L- targeting antibody.
- the antibodies and antibody derivatives and methods of the invention exhibited a (3-4-fold) increase in the control of chimerism/graft proliferation in vivo, compared to previously published non-Fc region optimised anti- OX40L antibodies (Tripathi et al. 2019).
- the superior effects of the antibodies and antibody derivatives and methods of the invention were observed relative to both Fc-active (Oxelumab) and Fc-silent (Amlitelimab) anti-OX40L antibodies known in the prior art, thus highlighting the unexpected effect of reduced core fucosylation of the antibodies and antibody derivatives and methods of the invention results in the examples.
- the antibodies and antibody derivatives and methods of the invention exhibit superiority to anti-OX40L antibodies of the prior art.
- the present invention provides for an alternative strategy to control T cells in a non-depleting manner whilst the naive phenotype of the immune cell population is maintained (/.e., naive T-cells and Tregs).
- T cells are indirectly controlled by blockade of OX40-OX40L.
- This strategy is validated by the prominent contribution of effector T cells in the pathogenesis of many autoimmune diseases and inflammatory disorders, and the preferential location of co-stimulatory receptor-ligand pair OX40-OX40L at the inflamed sites.
- the complex downstream effects of targeting T-cells directly are prevented and the adaptive immune system's capability to regulate itself is preserved.
- the antibodies and antibody derivatives have new and inventive properties, thereby resulting in benefits for patients in need of an antibody-based therapy against OX40L, especially for patients suffering from inflammatory disorders and autoimmune diseases, including rheumatoid arthritis, allergic asthma, and GvHD in transplantation.
- the present invention provides for a polypeptide comprising an amino acid sequence of SEQ ID NO:1 (VL).
- the present invention provides for an anti-OX40L antibody having a variable light chain and a variable heavy chain, wherein the variable light chain comprises the following CDRs: a. SEQ ID NO: 2; b. SEQ ID NO: 3; and c. SEQ ID NO: 4; and wherein the variable heavy chain comprises the following CDRs: d. SEQ ID NO: 6; e. SEQ ID NO: 7; and f. SEQ ID NO: 8.
- the present invention provides for an anti-QX40L antibody comprising a heavy chain and a light chain, wherein the light chain comprises a VL comprising the following CDRs: a. SEQ ID NO: 2; b. SEQ ID NO: 3; and c. SEQ ID NO: 4; and wherein the heavy chain comprises a constant region and a VH comprising the following CDRs: d. SEQ ID NO: 6; e. SEQ ID NO: 7; and f. SEQ ID NO: 8, and, wherein core fucosylation of the constant region of the heavy chain is below 80% determined as % fucosylated glycans of total glycans.
- the core fucosylation of the constant region of the heavy chain is below 80%, as determined by capillary electrophoresis, LC-MS, High-Performance liquid chromatography with Fluorescence Detection (HPLC-FD), High-Performance Anion-Exchange chromatography with Pulsed Amperometric Detection (HPAE-PAD), mass spectrometry, hydrazinolysis and/or enzyme digestion.
- the present invention provides for an anti-QX40L antibody comprising an IgG 1 heavy chain and a light chain, wherein the light chain comprises a VL comprising the following CDRs: a. SEQ ID NO: 2; b. SEQ ID NO: 3; and c. SEQ ID NO: 4; and wherein the IgG 1 heavy chain comprises the following CDRs: d. SEQ ID NO: 6; e. SEQ ID NO: 7; and f. SEQ ID NO: 8, and, wherein core fucosylation of the lgG1 heavy chain is below 80%.
- the present invention provides for an antibody comprising a light chain comprising a VL sequence of SEQ ID NO: 1 , wherein core fucosylation of the antibody is below 80%.
- the present invention provides for an anti-OX40L antibody comprising a heavy chain and light chain, comprising a VL sequence of SEQ ID NO: 1, wherein core fucosylation of the heavy chain is below 80%.
- the antibody comprises an lgG1 heavy chain. In some embodiments, the antibody comprises an lgG1 heavy chain constant region. In some embodiments, the antibody comprises an IgG 1 heavy chain constant region, wherein the lgG1 heavy chain constant region is glycosylated.
- the present invention provides for an antibody comprising a heavy chain of SEQ ID NO: 14 and a light chain comprising a VL sequence of SEQ ID NO: 1, wherein core fucosylation of the heavy chain is below 80%.
- the present invention provides for an anti-QX40L antibody comprising a heavy chain of SEQ ID NO: 14 and a light chain comprising a VL sequence of SEQ ID NO: 1 , wherein core fucosylation of the heavy chain is below 80%.
- the antibody comprises a variable heavy chain of SEQ ID NO: 5, and a variable light chain of SEQ ID NO: 1. In another embodiment, the antibody comprises a heavy chain of SEQ ID NO: 14 and a light chain of SEQ ID NO: 13.
- the antibodies according to the invention are preferentially characterised as being affinity matured fully human anti-QX40L antibodies of subclass lgG1.
- the present invention provides for a method of producing an anti-QX40L antibody or antibody derivative, comprising expressing polynucleotides encoding polypeptide of SEQ ID NO: 13 and 14 in a cell, wherein the cell is exposed to culture medium comprising an inhibitor of glucosidases I and II, and subsequently purifying the antibody.
- the present invention provides for a method of producing an anti-OX40L antibody or antibody derivative, comprising expressing polynucleotides encoding polypeptides of SEQ ID NO: 13 and 14 in a cell, wherein the cell is exposed to culture medium comprising a fucose analog, and subsequently purifying the antibody.
- the present invention provides for a method of producing an anti-OX40L antibody or antibody derivative, comprising expressing polynucleotides encoding polypeptides of SEQ ID NO: 13 and 14 in a cell that is genetically modified to prevent core fucosylation.
- the present invention provides for a method of producing an anti-OX40L antibody or antibody derivative, comprising expressing polynucleotides encoding polypeptide of SEQ ID NO: 13 and 14 in a cell, and subsequently purifying and optionally folding the antibody.
- the present invention provides for a method of testing a defucosylated anti-QX40L antibody or antibody derivative, comprising testing the antibody in a T-cell activation assay and verifying that the T-cell activation is reduced compared to a fucosylated anti-QX40L antibody.
- FIG. 1 Inhibition of fucosylation by 2-fluorofucose (2-FF).
- X-axis denotes 2-FF concentration (pM).
- Y-axis denotes relative fucosylation (%). Reduction to less than 40% fucosylation was achieved following treatment with 200 pM 2-FF.
- Figure 2 Affinity of OX118 towards various Fc-receptors.
- X-axis denotes the degree of fucosylation.
- Y-axis denotes affinity towards Fc-receptors.
- Reduced degree of fucosylation resulted in increased affinity for CD16a (FcRgllla/CD16a 158V, FcRgllla /CD16a 158F) without influence on FcRn, C1q or CD32a binding.
- A CD4+ and (B) CD8+ T cells following treatment with different anti-OX40L antibodies; anti-OX40L (10 pg/mL) and CTLA-4 Ig (15 pg/mL).
- X-axis denotes treatment condition.
- Y-axis denotes the percentage of proliferating T cells (CFSE low) in a two-way MLR setting after 7 days of incubation.
- 0X118 (10 pg/mL) resulted in a lower degree of both proliferating CD4+ and CD8+ T cells compared to control and compared to other forms of OX40L targeting (10 pg/mL) and CTLA-4 Ig (15 pg/mL).
- 0X118 increases the proportion of T-regulatory cells (T-Regs) among proliferating CD4+ T-cells.
- T-Regs T-regulatory cells
- A The addition of 0X118 (10 pg/mL) in a two-way MLR resulted in an increased regulatory phenotype (Tregs, CD25hi, CD4+, FoxP3+) among proliferating CD4+ cells compared to control, other forms of anti-OX40L antibodies (10 pg/mL) and CTLA-4 Ig (15 pg/mL).
- B Concentration-dependent enrichment in T-reg population among proliferating CD4+ cells compared to control, other forms of anti- OX40L antibodies, and CTLA-4 Ig.
- X-axis concentration of test item ng/mL).
- 0X118 increases the proportion of naive CD4+ T-cells among proliferating CD4+ T-cells.
- X-axis denotes treatment condition.
- Y-axis denotes the percentage of naive (CD3+, CD4+, CD45RA+, CCR7+, CFSE low) CD4+ T cells among proliferating cells.
- Addition of 0X118 at a concentration (10 pg/mL) in a two- way MLR resulted in increased naive T cells among proliferating CD4+ cells compared to control and anti-OX40L lgG1 ref (10 pg/mL) and CTLA-4 Ig (15 pg/mL).
- 0X118 prevents induction of pro-inflammatory phenotype.
- X-axis denotes treatment condition.
- Y-axis denotes cytokine concentration (ng/mL);
- A IL-10
- B II-4
- C TNFa in the supernatant.
- Addition of 0X118 (10 pg/mL) in a two-way MLR resulted in decreased IL-4 and TNFa compared to control.
- IL-10 levels were comparable to control.
- Y-axis denotes the frequency of circulating human CD45+ leukocytes (hCD45+) in mice.
- X-axis denotes days (7, 14, and 21) post-induction of GvHD.
- Flow cytometry-assisted immune phenotyping was performed on Day 7, Day 14, and Day 21. hCD45+ cells were counted and compared between treatment and control groups.
- Figure 9. 0X118 reduces activation of T-cells in peripheral blood following induction of GvHD in mice.
- X-axis denotes treatment.
- Y-axis denotes the percentage of activated CD4+, CD8+ T cells, and CD14+ monocyte cells in peripheral blood.
- 0X118 increases proportion of T regulatory cells (T-Regs) among proliferating CD4+ T-cells following induction of GvHD in mice.
- T-Regs T regulatory cells
- X-axis denotes treatment.
- the Y-axis denotes the percentage of regulatory T cells among CD4+ cells.
- Regulatory T cells were defined as CD3+, CD4+, CD25, and FoxP3+ via FACS, data from 22 days post-GvHD induction.
- the X-axis denotes treatment concentration.
- the Y-axis denotes luminescence intensity (corresponding to ADCC signalling in report cell line).
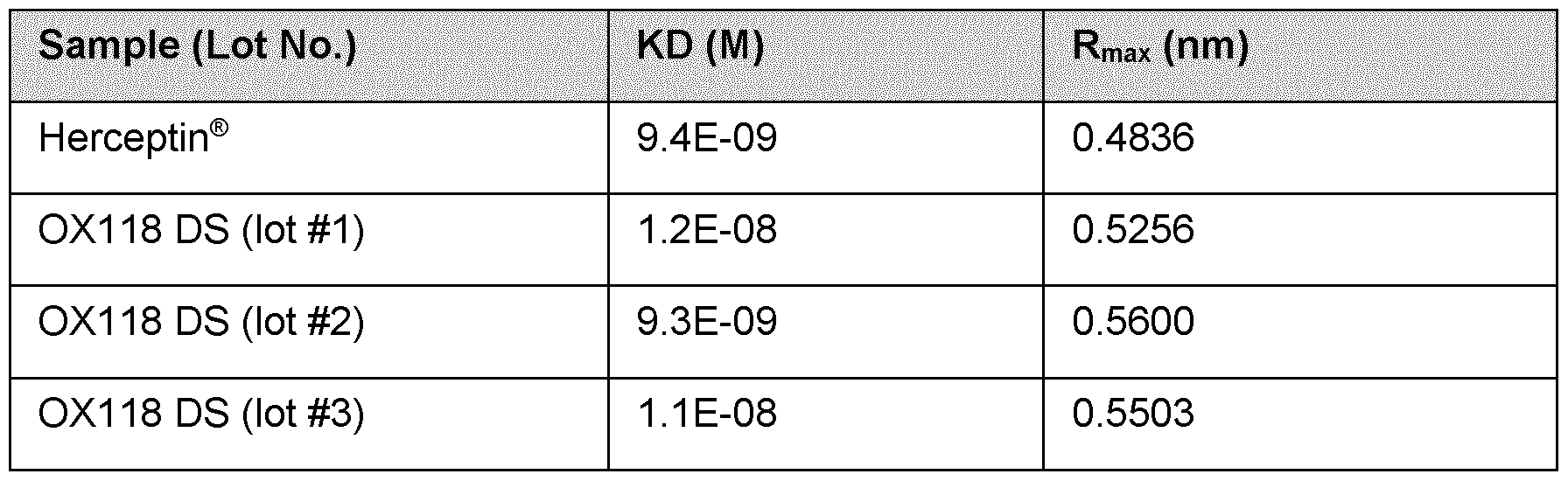
- Batches of 0X118 show reproducible results with high lot to lot consistency.
- the high affinity towards FcYRIIIa of 0X118 results in a low EC50 for ADCC signalling in the assay compared to a control antibody (trastuzumab).
- antibody and “antibodies” (immunoglobulins) may be an oligoclonal antibody, a polyclonal antibody, a monoclonal antibody (including full-length monoclonal antibodies), a camelised antibody, a chimeric antibody, a CDR-grafted antibody, a multi-specific antibody, a bi-specific antibody, a catalytic antibody, a chimeric antibody, a humanized antibody, a fully human antibody, an anti-idiotypic antibody and antibodies that can be labeled in soluble or bound form as well as fragments, variants or derivatives thereof, either alone or in combination with other amino acid sequences provided by known techniques.
- An antibody may be from any species.
- An antibody comprises a polypeptide or group of polypeptides that are comprised of at least one binding domain that is formed from the folding of polypeptide chains having three-dimensional binding spaces with internal surface shapes and charge distributions complementary to the features of an antigenic determinant of an antigen.
- An antibody typically has a tetrameric form, comprising two identical pairs of polypeptide chains, each pair having one "light” and one "heavy” chain. The variable regions of each light/heavy chain pair form an antibody binding site.
- Native antibodies are usually heterotetrameric glycoproteins of about 150,000 daltons, composed of two identical light (L) chains and two identical heavy (H) chains.
- Each light chain is linked to a heavy chain by one covalent disulfide bond, while the number of disulfide linkages varies between the heavy chains of different immunoglobulin isotypes.
- Each heavy and light chain also has regularly spaced intrachain disulfide bridges.
- Each heavy chain has at one end a variable domain (VH) followed by a number of constant domains.
- Each light chain has a variable domain at one end (VL) and a constant domain at its other end; the constant domain of the light chain is aligned with the first constant domain of the heavy chain, and the light chain variable domain is aligned with the variable domain of the heavy chain.
- Light chains are classified as either lambda chains or kappa chains based on the amino acid sequence of the light chain constant region.
- variable domain of a kappa light chain may also be denoted herein as VK.
- variable region may also be used to describe the variable domain of a heavy chain or light chain. Particular amino acid residues are believed to form an interface between the light and heavy chain variable domains.
- the variable regions of each light/heavy chain pair form an antibody binding site.
- antibody derivative means an antibody, as defined above (including an antibody fragment), or Fc domain or region of an antibody comprising a complex N- glycoside linked sugar chain, that is modified by covalent attachment of a heterologous molecule such as, e.g., by attachment of a heterologous polypeptide (e.g., a ligand binding domain of heterologous protein), or by glycosylation (other than core fucosylation), deglycosylation (other than non-core fucosylation), acetylation, phosphorylation or other modification not normally associated with the antibody or Fc domain or region.
- a heterologous polypeptide e.g., a ligand binding domain of heterologous protein
- ADCC antibody-dependent cellular cytotoxicity
- the term “monoclonal antibody” refers to an antibody that is derived from a single cell clone, including any eukaryotic or prokaryotic cell clone, or a phage clone, and not the method by which it is produced. Thus, the term “monoclonal antibody” is not limited to antibodies produced through hybridoma technology.
- Fc region refers to the constant region of an antibody, e.g., a Cnl-hinge- CH 2-CH 3 domain, optionally having a CH 4 domain, or a conservatively substituted derivative of such an Fc region.
- Glycan or "N-glycan” refers to an oligosaccharide comprising a core pentasaccharide Man3GlcNAc2.
- the N-glycan can be attached to a protein
- glycoprotein via the nitrogen of an asparagine (or occasionally arginine) residue, or free in solution.
- glycan refers to an
- N-glycan unless otherwise specified.
- oligosaccharide refers to a glycan that is not covalently bound to a protein.
- a useful reference for glycan, glycoprotein, and oligosaccharide nomenclature can be found at the website .html
- N-glycan variability is comprehensive but differences with functional importance are often limited to mannose content and degree of fucosylation (Reusch et al. 2015).
- glycosylation pattern or “glycosylation profile” refers to the observed glycosylation of a given glycoprotein or glycoproteins.
- a glycoprotein with a greater number of covalently linked sugar residues in the oligosaccharide chain is said to have an increased or more extensive glycosylation pattern and/or an increased total glycosylation.
- a glycoprotein with fewer covalently linked sugar residues in the oligosaccharide chain is said to have a decreased or less extensive glycosylation pattern and/or a decreased total glycosylation.
- the glycosylation pattern of a monoclonal antibody is variable depending on the method of synthesis or production.
- glycosylation pattern also refers to a characteristic distribution of several different glycosylation patterns on individual glycoproteins. In this sense, an increased glycosylation pattern means an increase in the characteristic distribution of glycosylation patterns of the expressed glycoproteins.
- total glycosylation or “total glycans” used herein refers to the total amount of individual glycan monosaccharides as quantified by known methods in the art, such as capillary electrophoresis, High-Performance liquid chromatography with Fluorescence Detection (HPLC-FD), High-Performance Anion-Exchange chromatography with Pulsed Amperometric Detection (HPAE-PAD), and mass spectrometry.
- HPLC-FD High-Performance liquid chromatography with Fluorescence Detection
- HPAE-PAD Pulsed Amperometric Detection
- Fc domain refers to the constant region domain of an antibody, e.g., a CH I, hinge, CH 2, CH 3, or CH 4 domain, or a conservatively substituted derivative of such an Fc domain.
- activated T cell refers to a T-cell that expresses antigens indicative of T-cell activation (that is, T cell activation markers).
- T cell activation markers include but are not limited to, CD25, CD26, CD30, CD38, CD69, CD70, CD71 , ICOS, OX-40, H LA-DR, and 4-1 BB.
- the expression of activation markers can be measured by techniques known to those of skill in the art, including, for example, western blot analysis, northern blot analysis, RT-PCR, immunofluorescence assays, and fluorescence-activated cell sorter (FACS) analysis.
- FACS fluorescence-activated cell sorter
- Tregs are a type of immune cells characterized by the expression of the biomarkers CD4, FOXP3, and CD25. Tregs are sometimes referred to as suppressor T cells and represent a subpopulation of T cells that modulate the immune system, maintain tolerance to self-antigens, and prevent autoimmune disease. Tregs are immunosuppressive and generally suppress or downregulate induction and proliferation of effector T (Teff) cells. Tregs can develop in the thymus (so-called CD4+ Foxp3+ “natural” Tregs) or differentiate from naive CD4+ T cells in the periphery, for example, following exposure to TGFp or retinoic acid. Tregs can express cell surface GARP-proTGFpi.
- OX40L refers to cognate ligand of the tumour necrosis factor receptor 0X40 (CD134). OX40L functions as a T cell co-stimulatory molecule. OX40-OX40L interactions have been proposed as a potential therapeutic target for treating autoimmunity.
- the present invention provides for a polypeptide comprising an amino acid sequence of SEQ ID NO:1 (VL).
- the present invention provides for an anti-OX40L antibody having a variable light chain and a variable heavy chain, wherein the variable light chain comprises the following CDRs: a. SEQ ID NO: 2; b. SEQ ID NO: 3; and c. SEQ ID NO: 4; and wherein the variable heavy chain comprises the following CDRs: d. SEQ ID NO: 6; e. SEQ ID NO: 7; and f. SEQ ID NO: 8.
- the present invention provides for an anti-QX40L antibody comprising a heavy chain and a light chain, wherein the light chain comprises a VL comprising the following CDRs: a. SEQ ID NO: 2; b. SEQ ID NO: 3; and c. SEQ ID NO: 4; and wherein the heavy chain comprises a constant region and a VH comprising the following CDRs: d. SEQ ID NO: 6; e. SEQ ID NO: 7; and f. SEQ ID NO: 8, and, wherein core fucosylation of the constant region of the heavy chain is below 80%.
- the present invention provides for an anti-QX40L antibody comprising an IgG 1 heavy chain and a light chain, wherein the light chain comprises a VL comprising the following CDRs: a. SEQ ID NO: 2; b. SEQ ID NO: 3; and c. SEQ ID NO: 4; and wherein the IgG 1 heavy chain comprises the following CDRs: d. SEQ ID NO: 6; e. SEQ ID NO: 7; and f. SEQ ID NO: 8, and, wherein core fucosylation of the lgG1 heavy chain is below 80%.
- the core fucosylation of the constant region of the heavy chain is below 80% , as determined by capillary electrophoresis, LC-MS, High-Performance liquid chromatography with Fluorescence Detection (HPLC-FD), High-Performance Anion-Exchange chromatography with Pulsed Amperometric Detection (HPAE-PAD), mass spectrometry, hydrazinolysis and/or enzyme digestion.
- the present invention provides for an antibody comprising a light chain comprising a VL sequence of SEQ ID NO: 1 , wherein core fucosylation of the antibody is below 80%.
- the present invention provides for an anti-QX40L antibody comprising a heavy chain and light chain, comprising a VL sequence of SEQ ID NO: 1 , wherein core fucosylation of the heavy chain is below 80%.
- the antibody comprises an lgG1 heavy chain.
- the antibody comprises an lgG1 heavy chain constant region.
- the antibody comprises an IgG 1 heavy chain constant region, wherein the IgG 1 heavy chain constant region is glycosylated.
- the present invention provides for an antibody comprising a heavy chain of SEQ ID NO: 14 and a light chain comprising a VL sequence of SEQ ID NO: 1, wherein core fucosylation of the heavy chain is below 80%.
- the present invention provides for an anti-OX40L antibody comprising a heavy chain of SEQ ID NO: 14 and a light chain comprising a VL sequence of SEQ ID NO: 1 , wherein core fucosylation of the heavy chain is below 80%.
- the antibody comprises a variable heavy chain of SEQ ID NO: 5, and a variable light chain of SEQ ID NO: 1. In another embodiment, the antibody comprises a heavy chain of SEQ ID NO: 14 and a light chain of SEQ ID NO: 13.
- Fc glycosylation is provides structural integrity to the antibody, and alterations in glycosylation patterns due to differences in production conditions have been reported to affect susceptibility to proteolytic degradation, clearance rate in vivo, Fey receptor binding and activation, antibody-dependent cellular cytotoxicity (ADCC) and C1 q component binding mediated complement activation. Alterations in glycosylation could therefore compromise effector functions including bioactivity, clinical efficacy, pharmacokinetics, safety, stability, and antigenicity.
- ADCC antibody-dependent cellular cytotoxicity
- glycosylation pattern can be influenced by cellular expression systems, culture conditions, and purification schemes. Determination of glycosylation pattern in glycoprotein-based drugs is recommended by the FDA and EMA, which suggest that the oligosaccharide content of glycoprotein products should be examined to ensure product consistency.
- the antibodies of the present invention may have variability in their glycosylation pattern and/or total glycosylation, such as +/- 1%, such as +/- 5%, such as +/-10%, such as +/- 15%, such as +/- 20%, such as +/- 25%, such as +/- 30%, such as at least 30%, such as at most 30%.
- glycosylation pattern of antibodies can be determined by known suitable methods, such as capillary electrophoresis (Wacker et a/. 2011), High-Performance liquid chromatography with Fluorescence Detection (HPLC-FD), High-Performance Anion-Exchange chromatography with Pulsed Amperometric Detection (HPAE-PAD), and mass spectrometry. All aforementioned methods of determination of glycosylation pattern are subject to interpretation of results, such as interpretation of chromatographs. Interpretation of results may lead to variability in glycosylation patterns, such as +/- 5% variability. This can vary depending on the method utilised, all methods include an error of between 2- 5%, such as between 2-4%.
- the present invention provides for anti-OX40L antibodies and antibody derivatives which have a standard N-Glycan (end-glycosylated) pattern of a human lgG1 expressed in CHO cells (for example CHO-K1 cells), for example as described in Reusch & Tomeda, 2015 (“Fc glycans of therapeutic antibodies as critical quality attributes”, Glycobiology, Volume 25, Issue 12, December 2015, Pages 1325-1334) and/or Luo & Zhang 2023 (“Benchmark glycan profile of therapeutic monoclonal antibodies produced by Mammalian cell expression systems”, Pharma. Res.) but with a reduced degree of fucosylation.
- N-Glycan end-glycosylated
- Luo & Zhang 2023 in Table 1 describes the structure of the most common N-Glycan structures and terminal epitopes in FDA-approved monoclonal antibodies.
- the examples of the application also characterize the normal glycosylation pattern of a human lgG1 expressed in CHO-K1 cells. Accordingly, the antibodies of the present invention are inherently glycosylated.
- the glycosylation pattern of the antibodies of the present invention is determined by the aforementioned methods known in the art.
- the glycosylation pattern of the antibodies of the present invention will be determined relative to the expression system or method of synthesis or production utilised for production, for example, the methods of benchmark glycan profiling disclosed in Luo & Zhang (2023).
- the glycosylation pattern of the antibodies of the present invention will be determined relative to the United States Pharmacopeia (USP) developed monoclonal antibody reference standards, such as those disclosed in Guo et al. 2022.
- the glycosylation pattern of the antibodies of the present invention have a standard N-Glycan glycosylation pattern of a human I gG 1 , except for the reduction in fucosylation.
- the antibody comprises a light chain (LC) and IgG 1 heavy chain (HC) as herein described and is glycosylated with at the least 2 kDa of total glycans.
- the antibody is glycosylated with at the most 3 kDa of total glycans. In some embodiments, the antibody is glycosylated with at the least 2kDa of total glycans and at the most 3 kDa of total glycans.
- the heavy chain constant region of the antibody is glycosylated with at the least 2 kDa of total glycans. In some embodiments, the heavy chain constant region of the antibody is glycosylated with at the most 3 kDa of total glycans. In some embodiments, the heavy chain constant region of the antibody the heavy chain constant region is glycosylated with at the least 2kDa of total glycans and at the most 3 kDa of total glycans.
- the provided antibody is engineered to contain a high proportion of glycan GO, specifically within the range of 50% to 80% of total glycans, while achieving reduced levels of core fucosylation.
- the antibody glycans comprise between 50% and 80% of glycan GO.
- the antibody glycans comprise between 50% and 80% of glycan GO, such as between 55 and 80%, such as between 60% and 80%, such as between 65% and 80%, such as between 70% and 80%, such as between 75% and 80%, such as between 50% and 75%, such as between 50% and 70%, such as between 50% and 65%, such as between 50% and 60%, such as between 50% and 55%.
- total glycans of the antibody comprise between 50% and 80% of glycan GO. In some embodiments, total glycans of the antibody comprise between 50% and 80% of glycan GO, such as between 55 and 80%, such as between 60% and 80%, such as between 65% and 80%, such as between 70% and 80%, such as between 75% and 80%, such as between 50% and 75%, such as between 50% and 70%, such as between 50% and 65%, such as between 50% and 60%, such as between 50% and 55%. Control over GO levels, combined with reduced core fucosylation, allows for enhanced functional properties, including improved ADCC activity.
- core fucosylation refers to addition of fucose ("fucosylation") to N- acetylglucosamine (“GIcNAc”) at the reducing terminal of an N-linked glycan.
- core fucosylation may occur at the N-glycosylation site of asparagine at position 297 (Asn-297) within the Fc region.
- Reduction of core fucosylation in antibodies has been shown to alter Fc effector functions, in particular Fc-gamma receptor binding and ADCC activity. This means that antibodies lacking core fucose in their Fc-glycan exhibit high ADCC activity at lower concentrations compared to fucosylated counterparts (Yamane-Ohnuki and Satoh, 2009).
- low fucosylation refers to a single glycoprotein molecule having fewer fucose residues attached to it. Rather, reference is made to a glycoprotein preparation, and the glycoprotein preparation comprises a population of individual glycoprotein molecules, with members of the population having different glycosylation features.
- low fucosylation refers to a smaller number of individual glycoproteins having a fucose residue on an N-linked GIcNAc residue.
- Such "low fucosylation” or “reduced fucosylation” can be characterized in a variety of ways (see elsewhere herein), but reference is in each case to a relatively low (or reduced) number of the glycoproteins of the population having fucose residues on them as compared to a population of the same glycoprotein made in a cell line that lacks a modification, or to a population of the same glycoprotein made without exposure to inhibitors and/or analogs which reduce core fucosylation, or to a population of the same glycoprotein made without exposure to culturing conditions/additives which reduce fucosylation, in accordance with the invention.
- references to “relative fucosylation” denotes the number of fucosylated glycans compared to the total number of glycans contained on the antibody molecule or antibody derivative.
- the present invention provides for an anti-OX40L antibody, comprising a variable heavy chain, wherein core fucosylation of the heavy chain is below 80%.
- the anti-OX40L antibody is fucosylated only about at most 50%, such as at most 40%, such as at most 30%, such as at most 20%, such as at most 20%, such as at most 10% of the amount of fucosylation of the same glycoprotein made in a cell that does not contain the modification.
- a glycoprotein preparation according to the invention is fucosylated only about at most 50%, such as at most 40%, such as at most 30%, such as at most 20%, such as at most 20%, such as at most 10% of the amount of fucosylation of the same glycoprotein made in a cell line that lacks a modification, or of the same glycoprotein made without exposure to inhibitors and/or analogs which reduce core fucosylation, or of the same glycoprotein made without exposure to culturing conditions/additives which reduce fucosylation.
- a glycoprotein preparation made in a cell according to the invention is fucosylated only about at most 50%, such as at most 40%, such as at most 30%, such as at most 20%, such as at most 20%, such as at most 10% of the amount of fucosylation of the same glycoprotein made in a cell line that lacks a modification, or of the same glycoprotein made without exposure to inhibitors and/or analogs which reduce core fucosylation, or of the same glycoprotein made without exposure to culturing conditions/additives which reduce fucosylation.
- Another way to characterize a glycoprotein with decreased fucosylation is by the ratio of fucosylated to non-fucosylated glycans in the glycoprotein preparation made by the cell, or the glycoprotein preparation which has not been exposed to inhibitors and/or analogs which reduce core fucosylation, or the glycoprotein preparation which has not been exposed to culturing conditions/additives which reduce fucosylation.
- a glycoprotein preparation has a ratio of fucosylated glycans: nonfucosylated glycans that is about 1 :10 through 1 :15, 1 :15 through 1 :20, 1 :20 through 1 :40, 1 :40 through 1 :60, 1 :60 through 1 :80, 1 :80 through 1 :100, or 1 :100 through 1 :150.
- glycoprotein preparation made by a modified cell has a percent of non-fucosylated glycans that is at most 70%, such as at most 60%, such as at most 50%, such as at most 40%, such as at most 30% as compared with the same glycoprotein preparation made from a cell that lacks the modification.
- a glycoprotein preparation made via exposure to cell culture conditions and/or additives that decrease fucosylation of the glycoprotein has a percent of non-fucosylated glycans that is at most 70%, such as at most 60%, such as at most 50%, such as at most 40%, such as at most 30% as compared with the same glycoprotein preparation which has not been exposed to cell culture conditions and/or additives which decrease fucosylation of the glycoprotein.
- the glycans with core fucosylation are below 70%, such as below 60%, for example below 50%, such as below 40%, for example below 30%, such as below 20% of the total glycans.
- antibodies of the instant methods comprise at least 10%, at least 20%, at least 30%, at least 40%, or at least 50% non-core fucosylated glycans (e.g., lacking core fucosylation), as compared with control antibodies.
- antibodies or antibody derivatives produced by the instant methods comprise at least 60%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, or at least 95% non-core fucosylated antibody, as compared with a control antibody.
- a composition of antibodies or antibody derivatives produced by the instant methods comprises less than 100% non-core fucosylated antibodies and/or antibody derivatives.
- Another way to characterize a glycoprotein with decreased fucosylation is by the relative amount of fucose to glycan or the relative amount of fucose to glycan component of the glycoprotein preparation.
- the molar ratio of fucose to glycan moiety is no more than about 1:10, 1 :20, 1 :25, 1:33, 1 :50, 1 :100, or 1 :200.
- the amount of antibody protein made that is fucosylated is measured by deglycosylation of antibody protein with PNGase F followed by oligosaccharide analysis via HPLC wherein fucosyl-containing oligosaccharides are quantified by integration of glycan peak area, and, e.g., protein fucosylation is calculated based on glycan peak area.
- the identity (and composition) of the glycan can be determined (and/or quantified) by any suitable method, such as mass spectroscopy.
- the core fucosylation is determined using capillary electrophoresis or HLPC, such as hydrophilic interaction liquid chromatography HPLC (HILIC-HPLC), electrospray ionization mass spectrometry (ESIMS), hydrophilic interaction liquid chromatography equipped with fluorescence detection and tandem mass spectrometry (HILIC-FLD-MS/MS).
- HILIC-HPLC hydrophilic interaction liquid chromatography HPLC
- EIMS electrospray ionization mass spectrometry
- HILIC-FLD-MS/MS hydrophilic interaction liquid chromatography equipped with fluorescence detection and tandem mass spectrometry
- Methods for determining core fucosylation include hydrazinolysis or enzyme digestion (see, e.g., Biochemical Experimentation Methods 23: Method for Studying Glycoprotein Sugar Chain (Japan Scientific Societies Press), edited by Reiko Takahashi (1989)), fluorescence labelling or radioisotope labelling of the released sugar chain and then separating the labelled sugar chain by chromatography. Also, the compositions of the released sugar chains can be determined by analysing the chains by the HPAEC-PAD method (see, e.g., J Liq Chromatogr. 6:1557 (1983)). (See generally US 2004- 0110282.)
- the core fucosylation is determined as (Area with fucosylated glycan)/(total glycan area). In some embodiments of the invention, the core fucosylation is determined as (concentration of fucosylated glycan)/(concentration of total glycan). The fucosylation percentage is calculated taking into consideration peak areas of all the fucosylated species within the total area of glycan. The following equation (1) was used to calculate the fucosylation percentage:
- the core fucosylation is determined as (amount of fucosylated glycan)/(amount of total glycan).
- the results of the determination of fucosylation are dependent on the interpretation of results, such as of chromatographs. Interpretation of results may lead to variability in degrees of fucosylation, such as +/- 5% variability. This can vary depending on the method utilised, all methods of determining fucosylation disclosed herein include an error of between 2-5%, such as between 2-4%.
- the antibody is covalently linked to a ligand selected from the group consisting of chromophores, fluorophores, radiotracers, drugs, peptides, proteins, enzymes, single or double chained oligonucleotides and their analogs, biotin and, therapeutic moieties, such as cytotoxins, chemotherapeutic drugs, cytokines, and radioisotopes.
- a ligand selected from the group consisting of chromophores, fluorophores, radiotracers, drugs, peptides, proteins, enzymes, single or double chained oligonucleotides and their analogs, biotin and, therapeutic moieties, such as cytotoxins, chemotherapeutic drugs, cytokines, and radioisotopes.
- the antibody is humanized. In some embodiments of the invention, the antibody is of the human subclass lgG1.
- Methods for reducing core fucosylation include chemoenzymatic remodelling, media supplementation, and engineered cell lines (see Li et al. 2017).
- Small molecule inhibitors that act on enzymes in the glycosylation pathway can be utilised to reduce core fucosylation.
- Inhibitors such as castanospermine, deoxymannojirimycin, australine, act early in the glycosylation pathway via inhibiting glucosidases I and II, producing antibodies with immature glycans (e.g., high levels of mannose) and low fucosylation levels.
- One aspect of the present invention is to provide a method of producing an anti-OX40L antibody or antibody derivative, comprising expressing polynucleotides encoding polypeptides of SEQ ID NO: 13 and 14 in a cell, wherein the culture medium comprises an inhibitor of glucosidases I and II, and subsequently purifying the antibody.
- the cell is a mammalian cell.
- the inhibitor is selected from the group comprising: castanospermine, deoxynojrimycin, and australine.
- the inhibitor is a-L-fucosidase. Analogs
- Fucose analogs reduce the incorporation of fucose into complex N-glycoside-linked sugar chains of antibodies or antibody derivatives produced by host cells.
- Suitable fucose analogs are those that can be added to the host cell culture media and inhibit core fucosylation of complex N-glycoside-linked sugar chains of antibodies or antibody derivatives.
- the fucose analog is typically taken up by host cells (e.g., by active transport or passive diffusion) (see Almahayni et al. 2022).
- One aspect of the present invention is to provide an anti-OX40L antibody or antibody derivative, comprising expressing polynucleotides encoding polypeptides of SEQ ID NO: 13 and 14 in a cell, wherein the culture medium comprises a fucose analog, and subsequently purifying the antibody.
- the cell is a mammalian cell.
- the fucose analog is an inhibitor of fucokinase, GDP-fucose- pyrophosphorylase, fucosyltransferase (FUT), GDP-mannose 4,6-dehydratase, GDP- fucose synthetase, and/or a fucose transporter, such as GDP-fucose transporter.
- a fucose analog (or an intracellular metabolite or product of the fucose analog) inhibits an enzyme(s) in the fucose salvage pathway.
- an intracellular metabolite can be, for example, a GDP-modified analog a fully or partially de-esterified analog.
- a fucose analog (or an intracellular metabolite or product of the fucose analog) can inhibit the activity of fucokinase or GDP-fucose-pyrophosphorylase.
- a fucose analog (or an intracellular metabolite or product of the fucose analog) inhibits fucosyltransferase (preferably a 1,6-fucosyltransferase, e.g., the FLIT8 protein).
- the fucose analog is an inhibitor of a fucosyltransferase (FUT), such as 1 ,6- fucosyltransferase (FUT8).
- a fucose analog (or an intracellular metabolite or product of the fucose analog) can inhibit the activity of an enzyme in the de novo synthetic pathway for fucose.
- a fucose analog (or an intracellular metabolite or product of the fucose analog) can inhibit the activity of GDP-mannose 4,6-dehydratase and/or GDP-fucose synthetase.
- the fucose analog (or an intracellular metabolite or product of the fucose analog) can inhibit a fucose transporter (e.g., GDP-fucose transporter).
- the fucose analog is selected from the group comprising L- fucose, 2-fluro-fucose, 2- fluoro peracetylated fucose (2FF), 5-alkynyl-fucose, alkynyl fucose monoacetate, alkynyl fucose triacetate, alkynyl fucose diacetate and 5-alkynyl- fucose peracetate.
- the fucose analog is 2-fluoro peracetylated fucose (2FF).
- the fucose analog is alkynyl fucose peracetate.
- the fucose analog is alkynyl fucose triacetate. In some embodiments, the fucose analog is alkynyl fucose diacetate. In some embodiments, the fucose analog is a mixture of alkynyl fucose peracetate, alkynyl fucose triacetate, and alkynyl fucose diacetate. In some embodiments, the fucose analog is a mixture of alkynyl fucose peracetate, alkynyl fucose triacetate, alkynyl fucose diacetate, and alkynyl fucose monoacetate.
- the fucose analog is not fucose. In some embodiments, the fucose analog is not alkynyl fucose peracetate. In some embodiments, the fucose analog is not galactose or L-galactose.
- the fucosyltransferase (FUT) inhibitors are carbohydrate mimics.
- the carbohydrate mimics are selected from the group consisting of O- and C-glycosides, carbacycles, and nitrogen-containing heterocycles such as piperidines and pyrrolidines.
- the person skilled in the art will appreciate, that a variety of carbohydrates and structural analogs bearing a GDP unit in their structure can have inhibitory properties against fucosyltransferase.
- the fucosyltransferase inhibitor is 2-fluoro peracetylated fucose (2FF).
- 2FF is a fluorinated analog of fucose that can be taken up and metabolized to the desired donor substrate-based inhibitor of fucosyltransferases inside the cell. It also acts to prevent the de novo synthesis of the natural substrates, resulting in a global, family-wide shutdown of fucosyltransferases and remodelling of cell surface glycans.
- the amount of the fucose analog that is effective can be determined by standard cell culture methodologies. For example, cell culture assays may be employed to help identify optimal dosing ranges. The precise amount to be employed also depends on the time of administration, the host cell line, the cell density, and the like. Effective doses may be extrapolated from dose-response curves derived from in vitro model test systems. Accordingly, in one embodiment, the culture medium is supplemented with the fucose analog to maintain an effective concentration thereof.
- the fucose analog is present in the culture medium at a concentration of 10 nM to 50 mM. In some embodiments, the fucose analog is present in the culture medium at a concentration of 10 nM to 10 mM. In some embodiments, the fucose analog is present in the culture medium at a concentration of 100 nM to 5 mM. In some embodiments, the fucose analog is present in the culture medium at a concentration of 100 nM to 3 mM. In some embodiments, the fucose analog is present in the culture medium at a concentration of 100 nM to 2 mM. In some embodiments, the fucose analog is present in the culture medium at a concentration of 100 pM to 1 mM.
- the fucose analog is present in the culture medium at a concentration of 1 pM to 1 mM. In some embodiments, the fucose analog is present in the culture medium at a concentration of 10 pM to 1 mM. In some embodiments, the fucose analog is present in the culture medium at a concentration of 10 nM to 500 pM. In some embodiments, the fucose analog is present in the culture medium at a concentration of 1 pM to 500 pM. In some embodiments, the fucose analog is present in the culture medium at a concentration of 1 pM to 250 pM. In some embodiments, the fucose analog is present in the culture medium at a concentration of 10 pM to 100 pM.
- the fucose analog is present in the culture medium at a concentration of at least 150 pM, such as at least 200 pM, such as at least 250 pM. In some embodiments, the fucose analog is soluble in the culture medium (at the appropriate temperature for host cell maintenance/growth) at a concentration of at least 10 nM. In some embodiments, the fucose analog is soluble in the culture medium (at the appropriate temperature for host cell maintenance/growth) at a concentration of at least 100 nM.
- Antibodies and derivatives thereof that are useful in the present methods can be produced by recombinant expression techniques, from hybridomas, from myelomas, or from other antibody-expressing cells.
- Recombinant expression of an antibody or derivative thereof that binds to a target antigen typically involves construction of an expression vector containing a nucleic acid that encodes the antibody or derivative thereof. Once a nucleic acid encoding such a protein has been obtained, the vector for the production of the protein molecule may be produced by recombinant DNA technology using techniques well-known in the art.
- Methods for engineering cell lines which are unable to fucosylate proteins to reduce core fucosylation include gene knockouts, gene knock-ins, and RNA interference (RNAi), said methods are known and well referenced in Edwards et al. (2022).
- gene knock-outs the gene encoding FUT8 (alpha 1,6- fucosyltransferase enzyme) is inactivated.
- FUT8 catalyses the transfer of a fucosyl residue from GDP-fucose to position 6 of Asn-linked (N-linked) GIcNac of an N- glycan.
- FUT8 is reported to be the only enzyme responsible for adding fucose to the N-linked biantennary carbohydrate at Asn297.
- FUT8 fucosyl transferase gene
- CHO Chinese hamster ovary
- lectin-resistant CHO glycosylation mutants selected for resistance to certain lectins.
- Such cell lines are isolated by repeated selection for the inability to bind a particular lectin, in the presence of a mutagen.
- Other cell lines reportedly incapable of fucosylating proteins, e.g., antibodies, are known, see, e.g., US Patent No. 7,425,466 and US Patent No. 7,214,775 (a1,6-fucosyltransferase, i.e., FUT8 mutant).
- RNAi typically also targets FUT8 gene expression, leading to decreased mRNA transcript levels or knock out gene expression entirely.
- Another approach has been to disrupt the gene that encodes the enzyme GDP-mannose 4,6-dehydratase (GMD). GMD is involved in catalysing the conversion of d-glucose to GDP-fucose and is therefore crucial for the activity of o1,6-FucT in carrying out fucosylation downstream in this pathway (Kanda et al. 2007).
- Gene knock-ins add genes encoding enzymes such as p-1 ,4-N- acetylglucosaminyltransferase (GNTIII), GDP-6-deoxy-D-lyxo-4-hexulose reductase (RMD), or a golgi alpha-mannosidase II.
- GNTIII p-1 ,4-N- acetylglucosaminyltransferase
- RMD GDP-6-deoxy-D-lyxo-4-hexulose reductase
- golgi alpha-mannosidase II golgi alpha-mannosidase II.
- Another aspect of the present invention is to provide a method of producing an anti- OX40L antibody, comprising expressing polynucleotides encoding polypeptides of SEQ ID NO: 13 and 14 in a cell that is genetically modified to prevent core fucosylation, such as in a mammalian cell.
- Cell lines that are unable to fucosylate proteins are known in the art.
- the cell does not encode a functional fucosyltransferase.
- fucosylation- deficient cell lines can be generated in several ways. Ways to generate fucosylation- deficient cell lines include but are not limited to knockouts, such as homologous recombination, CRISPR-Cas9 and TALENs, and knockdowns, such as through antisense oligonucleotides, ribozymes and RNA interference.
- the cell encodes a functional p-1 ,4-N- acetylglucosaminyltransferase (GnTIII), GDP-6-deoxy-D-lyxo-4-hexulose reductase (RMD) and/or a Golgi a-mannosidase II (Manll).
- GnTIII functional p-1 ,4-N- acetylglucosaminyltransferase
- RMD GDP-6-deoxy-D-lyxo-4-hexulose reductase
- Manll Golgi a-mannosidase II
- the cell is a mammalian cell. In some embodiments, the cell is a recombinant cell. In some embodiments, the cell is selected from the following cells: CHO (e.g., CHO K1 , DXB-11 OHO, Veggie-CHO), COS (e.g., COS-7), Syrian hamster, rat myeloma, mouse myeloma (e.g., SP2/0, NSO), retinal cell, Vero, CV1, kidney (e.g., HEK293, 293 EBNA, MSR 293, MDCK, HaK, BHK, BHK21), HeLa, HepG2, WI38, MRC 5, Colo205, HB 8065, HL-60, Jurkat, Daudi, A431 (epidermal), CV-1, U937, 3T3, L cell, C127 cell, MMT 060562, Sertoli cell, BRL 3A cell, HT1080 cell, a human myeloma cell
- Monoclonal antibodies directed against the antigen can be obtained from the immunized, transgenic mice using conventional hybridoma technology.
- the host cells are from a hybridoma.
- the antibodies and antibody derivatives are produced, synthesised, or obtained by cell-free protein synthesis methods, such as those disclosed in Hunt et al. 2023, or Stech et al., 2017.
- the method of producing an anti-OX40L antibody or antibody derivative, comprising expressing polynucleotides encoding polypeptides of SEQ ID NO: 13 and 14, is cell- free protein synthesis.
- the present invention provides a method of producing an anti-OX40L antibody or antibody derivative, comprising expressing polynucleotides encoding polypeptides of SEQ ID NO: 13 and 14 in a cell that is genetically modified to prevent core fucosylation, wherein the antibody or antibody derivative is restored or titrated to a desired level via exposure to fucose, wherein the cell is exposure to culture medium comprising fucose.
- increasing concentrations of fucose are added to the culture media, such as up to 1mM.
- the method comprises an additional step of subsequently isolating the antibody.
- the method comprises an additional step of subsequently purifying the antibody.
- the method comprises an additional step of optionally folding the antibody.
- the antibodies of the present disclosure can be purified to homogeneity.
- the separation and purification of the antibodies can be performed by employing conventional protein separation and purification methods known by the person skilled in the art, for example, hydroxylapatite chromatography, gel electrophoresis, dialysis, and affinity chromatography such as Protein A purification.
- the cell is selected from the group consisting of mammalian cells, bacterial cells, yeast cells, and plant cells.
- the antibody is an lgG1. In some embodiments, the antibody is an intact antibody. In some embodiments, the antibody comprises a heavy and a light chain variable region and an Fc region. In some embodiments, the antibody derivative comprises an antibody Fc region and a ligand binding domain of a non-immunoglobulin protein.
- the present invention provides for compositions and methods to produce antibodies and antibody derivatives which inhibit the binding of 0X40 to OX40L, wherein the antibodies and antibody derivatives described in the present invention bind to human OX40L, thereby inhibiting the OX40/OX40L interaction, and thereby inhibiting the OX40L induced signal transduction.
- the antibodies and antibody derivatives of the present invention inhibit hOX40L/OX40 interaction.
- the antibody according to the invention inhibits the interaction of OX40L with 0X40.
- the quantification of the interaction of OX40L with 0X40 can be measured by techniques known to those of skill in the art, including, for example, an ELISA using immobilized OX40L, such as biotinylated OX40L immobilized on a streptavidine surface.
- ADCC antibody-dependent cell cytotoxicity
- the antibodies and antibody derivatives of the instant disclosure have higher effector function (e.g., ADCC activity) than the antibodies with normal levels of fucosylation, i.e., those produced in the absence of a fucose analog or inhibitor, or those produced from wild-type cell lines.
- the effector function activity may be modulated by altering the concentration of the fucose analog in the culture medium and/or the duration of exposure to the fucose analog.
- ADCC activity may be measured using assays known in the art and in exemplary embodiments increases ADCC by at least 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, 2-fold, 3-fold, 4-fold, 5-fold, 6- fold, 7-fold, 8-fold, 9-fold, 10-fold, 15-fold or 20-fold, as compared to the core fucosylated parent antibody.
- the antibodies of the invention are Fc engineered such that the affinities for various Fey receptors are increased.
- the antibodies and antibody derivatives of the instant disclosure have a higher affinity for Fc receptors, such as CD16A 158V and CD16a 158F, than the antibodies with normal levels of fucosylation, i.e., those produced in the absence of a fucose analog or inhibitor, or those produced from wild-type cell lines.
- the affinity for Fc receptors may be modulated by altering the concentration of the fucose analog in the culture medium and/or the duration of exposure to the fucose analog.
- Affinity for Fc receptors may be measured using assays known in the art and in exemplary embodiments increases affinity for Fc receptors by at least 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, 2-fold, 3-fold, 4-fold, 5-fold, 6-fold, 7-fold, 8-fold, 9- fold, 10-fold, 15-fold or 20-fold, as compared to the core fucosylated parent antibody.
- the invention provides for a method of testing a defucosylated anti- OX40L antibody or antibody derivative, comprising testing the antibody in a T-cell activation assay and verifying that the T-cell activation is reduced compared to a fucosylated anti-OX40L antibody.
- the T-cell activation is reduced by at least 5%, such as at least 10%, such as at least 15%, such as at least 25%, such as at least 30%.
- the present invention includes the use of the antibody or antibody derivatives in the prophylaxis and/or treatment of at least one disease or disorder that is associated with OX40L.
- the invention relates to a method for the prevention and/or treatment of at least one disease or disorder that can be treated by modulating OX40L, its biological or pharmacological activity, and/or the biological pathways or signalling in which OX40L is involved, said method comprising administering, to a subject in need thereof, a pharmaceutically active amount of the antibody or antibody derivates of the invention and/or of a pharmaceutical composition comprising the same.
- said pharmaceutically effective amount may be an amount that is sufficient to modulate OX40L, its biological or pharmacological activity, and/or the biological pathways or signalling in which OX40L is involved.
- the present invention provides for the use of the antibodies or antibody derivatives in the prophylaxis and/or treatment of inflammatory disorders and/or autoimmune disorders.
- the antibody or antibody derivative is for use in the prophylaxis and/or treatment and/or prevention of inflammatory disorders and/or autoimmune disorders.
- the antibody or antibody derivative is for use in the prophylaxis and/or treatment of inflammatory disorders.
- the antibody or antibody derivative is for use in the preparation of a medicament for the prophylaxis and treatment of inflammatory disorders.
- the antibody or antibody derivative is for use in the prophylaxis and/or treatment of autoimmune diseases.
- the antibody or antibody derivative is for use in the preparation of a medicament for the prophylaxis and treatment of autoimmune diseases.
- the disease is selected from the group consisting of Graft-vs-host-disease, allograft rejection, asthma, Systemic lupus, arthritis, inflammatory bowel disease, ulcerative colitis, Crohn’s disease, diabetes, atopic dermatitis, psoriasis, hidradenitis suppurativa, immunoglobulin A nephropathy, Hashimoto disease, Graves’ disease, chronic rhinosinusitis and multiple sclerosis.
- the invention provides for a kit comprising the aforementioned antibody or antibody derivative.
- the term "prophylaxis and/or treatment” not only comprises preventing and/or treating the disease, but also generally comprises preventing the onset of the disease, slowing or reversing the progress of disease, preventing or slowing the onset of one or more symptoms associated with the disease, reducing and/or alleviating one or more symptoms associated with the disease, reducing the severity and/or the duration of the disease and/or of any symptoms associated therewith and/or preventing a further increase in the severity of the disease and/or of any symptoms associated therewith, preventing, reducing or reversing any physiological damage caused by the disease, and generally any pharmacological action that is beneficial to the patient being treated.
- the subject to be treated may be any warm-blooded animal but is in particular a mammal, and more in particular a human being.
- the subject to be treated will in particular be a person suffering from, or at risk of, the diseases and disorders mentioned herein.
- the present invention includes antibodies and antibody derivatives, compositions and methods for use in the prophylaxis and/or treatment of inflammatory disorders and/or autoimmune disorders.
- Administration of the antibodies and antibody derivatives and/or compositions according to the present invention will typically be via any common route. This includes, but is not limited to parenteral, orthotopic, intradermal, subcutaneous, intramuscular, intraperitoneal, intranasal, or intravenous injection.
- Additional formulations which are suitable for other modes of administration include oral formulations.
- Oral formulations include such normally employed excipients as, for example, pharmaceutical grades of mannitol, lactose, starch, magnesium stearate, sodium saccharine, cellulose, magnesium carbonate, and the like.
- compositions take the form of solutions, suspensions, tablets, pills, capsules, sustained-release formulations, or powders and contain about 10% to about 95% of active ingredient, preferably about 25% to about 70%.
- compositions of the invention are administered in a manner compatible with the dosage formulation, and in such amount as will be therapeutically effective and immune modifying.
- the quantity to be administered depends on the subject to be treated. Precise amounts of active ingredient required to be administered depend on the judgment of the practitioner.
- the manner of application may be varied widely. Any of the conventional methods for administration of an antibody are applicable. These are believed to include oral application on a solid physiologically acceptable base or in a physiologically acceptable dispersion, parenterally, by injection, and the like.
- the dosage of the pharmaceutical composition will depend on the route of administration and will vary according to the size and health of the subject. The course of the administrations may be followed by assays for alloreactive immune responses and T-cell activity.
- phrases “pharmaceutically acceptable” or “pharmacologically acceptable” refer to molecular entities and compositions that do not produce an adverse, allergic, or other untoward reaction when administered to an animal, or human.
- pharmaceutically acceptable excipient includes any and all solvents, dispersion media, coatings, antibacterial and antifungal agents, isotonic and absorption delaying agents, and the like. The use of such media and agents for pharmaceutically active substances is well-known in the art. Except insofar as any conventional media or agent is incompatible with the active ingredients, its use in immunogenic and therapeutic compositions is contemplated.
- compositions may be formulated into a neutral or salt form.
- Pharmaceutically acceptable salts include the acid addition salts (formed with the free amino groups of the protein) which are formed with inorganic acids such as, for example, hydrochloric or phosphoric acids, or such organic acids as acetic, oxalic, tartaric, mandelic, and the like. Salts formed with the free carboxyl groups can also be derived from inorganic bases such as, for example, sodium, potassium, ammonium, calcium, or ferric hydroxides, and such organic bases as isopropylamine, trimethylamine, histidine, procaine, and the like.
- the carrier can also be a solvent or dispersion medium containing, for example, water, ethanol, polyol (for example, glycerol, propylene glycol, liquid polyethylene glycol, and the like), suitable mixtures thereof, and vegetable oils.
- polyol for example, glycerol, propylene glycol, liquid polyethylene glycol, and the like
- suitable mixtures thereof and vegetable oils.
- the prevention of the action of microorganisms can be brought about by various antibacterial and antifungal agents, for example, parabens, chlorobutanol, phenol, sorbic acid, thimerosal, and the like.
- unit dose refers to physically discrete units suitable for use in a subject, each unit containing a predetermined quantity of the composition calculated to produce the desired responses discussed above in association with its administration, i.e. , the appropriate route and regimen.
- the quantity to be administered depends on the result and/or protection desired. Precise amounts of the composition also depend on the judgment of the practitioner and are peculiar to each individual. Factors affecting dose include the physical and clinical state of the subject, route of administration, the intended goal of treatment (alleviation of symptoms versus cure), and potency, stability, and toxicity of the particular composition.
- solutions Upon formulation, solutions will be administered in a manner compatible with the dosage formulation and in such amount as is therapeutically or prophylactically effective. The formulations are easily administered in a variety of dosage forms.
- In vitro affinity maturation is based on the principles of mutation and selection. In vitro affinity maturation has successfully been used to optimize antibodies, antibody fragments, or other peptide molecules like antibody mimetics. Random mutations inside the CDRs are introduced using radiation, chemical mutagens, or error-prone PCR. In addition, the genetic diversity can be increased by chain shuffling. Two or three rounds of mutation and selection using display methods like phage display usually result in antibody fragments with affinities in the low nanomolar range.
- an anti-OX40L antibody with improved characteristics such as potency, binding affinity for OX40L, and antibody stickiness compared to existing anti-OX40L antibodies.
- Affinity maturation The affinity maturation was performed in the scFv format. A site- directed CDR mutagenesis was performed to introduce mutations into the parental antibody (Anti-OX40L, Clone R4930) sequence. An antibody-phage library was constructed, which was used for an in vitro selection under stringent conditions. Finally, antibodies were identified, characterized, and converted into the final antibody format. Antibody binding was validated, and the antibodies were further characterized to identify lead candidates.
- Antigen preparation The R4930 gene was cloned into a scFv phage display vector. After transformation of E. Coli, soluble scFv antibodies were expressed as well as antibody-phage particles packaged. Packaging and scFv production was tested by a Titration-ELISA (data not shown).
- CDR-mutation library A bioinformatic analysis of R4930 was performed to generate a site directed CDR-mutation library. After homology modelling of the antibody Fv regions and CDR grafting onto the template, CDR residues possibly involved into antigen binding were identified. For the heavy chain 18 positions and for the light chain 12 positions were identified. By analysing a NGS database commonly used amino acids for the specific germline were identified. Based on this, degenerated codons were designed, introducing mutations at the identified position possibly involved into antigen binding. Amino acids with unfavourable characteristics were avoided. The introduction of mutations can be described by a gaussian distribution with an average of four mutations for each antibody chain.
- Primers were designed based on the degenerated codons and used for introduction of mutations into the antibody sequence.
- the mutated antibody genes were cloned into a scFv phage display vector and three libraries were generated and packaged into antibody-phage particles.
- a library with a total functional diversity of ⁇ 7x10 8 CFUs was generated.
- Antibody clones with a functional open reading frame were determined by DNA Sanger sequence analysis. Packaging and purification of antibody-phage particles resulted in more than 3x10 12 CFU/mL for each library.
- Affinity maturation by in vitro selection The generated antibody-phage library was used for the affinity maturation by in vitro selection. The same overall excess of antibodyphage particles to functional size was used for each individual library. The specific amount was pooled into one library for in vitro selection. Two rounds of in vitro selection were performed with increasing stringency by antigen limitation and competition in panning round two. In both panning rounds a negative selection against several negative antigens were performed. Four different strategies were used for affinity maturation by in vitro selection. In panning round two the amount of antigen was reduced from strategy one to four by factor ten between each strategy. Eluted antibody-phage after panning round 1 were amplified and used for the second panning round, after panning round 2 eluted antibody-phage were used for antibody screening.
- Antibody screening Eluted antibody-phage particles after panning round two were used for infection of E. Coli. 384 clones from each strategy were selected randomly for antibody screening. In total, 1536 antibody clones were used for production of monoclonal scFv antibodies in the bacterial system. The produced antibody clones were tested for binding activity on the OX40L immobilized directly to the ELISA plate. Two negative antigens (Streptavidin and BSA) were immobilized directly to the ELISA plate. Soluble scFv antibodies were detected via the myc-tag using an HRP coupled secondary antibody. Antibody clones with the defined parameters as listed below were identified as an antigen specific clone:
- Antibody sequencing All 444 antigen specific antibodies were selected for DNA sanger sequence analysis. Sequence analysis revealed 271 uniquely mutated antibodies. These antibodies showed between one and seven mutations. Several hotspot mutations were identified indicating preferable mutations at different positions.
- Affinity ranking To narrow down the number of antibodies for antibody conversion into the final format, an affinity ranking using the BLI octet system was performed. First the assay was set up using the parental scFv antibody produced in the bacterial system and the biotinylated antigen. The antigen was immobilized to a streptavidin sensor, which was incubated in the bacterial scFv supernatant for association. The dissociation was measured by incubation in buffer, after modelling the association and dissociation profile, the dissociation rate was calculated. 271 clones were picked and used for scFv production in the bacterial system. The assay as described above was performed for each antibody. The antibodies off-rate was calculated, and the antibodies were ranked accordingly. From all measured antibodies 267 could be measured and fitted with high confidence, among these 267 clones, 55 clones showed an increased off-rate compared to the parental antibody. Within these 55 antibodies, 13 showed at least two times increased off-rate compared to the parental antibody.
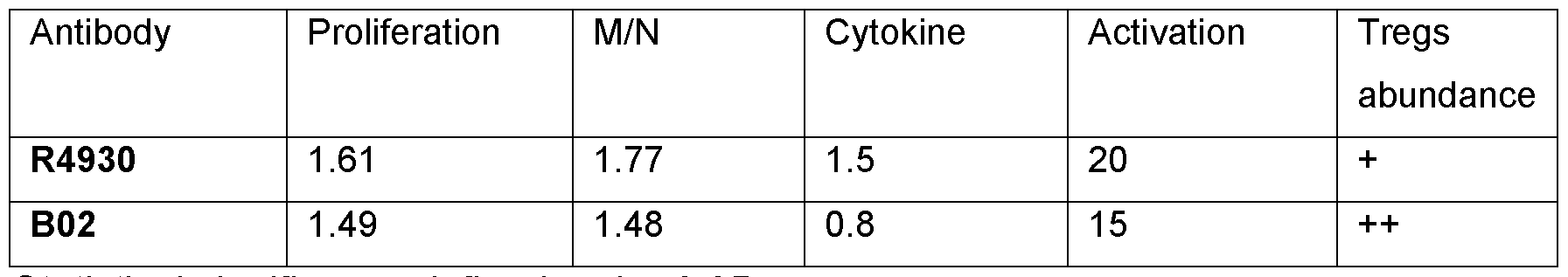
- Antibody affinity For further characterization of the antibodies an affinity determination using the BLI octet system was set up. The human IgG antibody was immobilized to protein A sensor tips and a titration of the human OX40L antigen was performed. The association and dissociation profile were modelled using either 50 nM or 15.8 nM concentrations, and the antibody affinity determined. The affinity of the parental antibody (R4930) and selected anti-OX40L antibody candidate (B02) was determined (see table 1). Allogeneic mixed lymphocyte reaction: Peripheral blood mononuclear cells (PBMC) were isolated via Ficoll Paque Plus (Cytiva) density gradient centrifugation from buffy coats (500g x 30 min).
- PBMC Peripheral blood mononuclear cells
- Buffy coats were obtained from anonymous healthy adult donors from the Akademiska University Hospital blood bank, and PBMC isolation was performed within 24 hours of blood collection. In line with Swedish legislation, an ethics approval was not needed since the blood bank provides buffy coats for research purposes from anonymous donors and biological material cannot be traced back to a specific individual. Isolated PBMC were mixed in PBS at a concentration of 2.0 x 10 7 cells per mL and stained with CFSE (Thermo Fisher Scientific Inc) according to the manufacturer’s instructions. PBMC from 2 different donors were mixed in a two-way allogeneic mixed lymphocyte reaction, i.e., PBMCs from neither donor were inactivated.
- CFSE Thermo Fisher Scientific Inc
- VPD450-stained PBMC were washed and resuspended in 10% heat- inactivated Ultralow IgG FBS (Gibco) in AIM V medium (Gibco, Thermo Fisher Scientific Inc). Resuspended PBMCs were dispensed into round-bottom 96-well cell culture plates, and pure medium or medium supplemented with test article was added to a final concentration of 2 x 10 6 cells per mL in a final volume of 200 pL. Mixed lymphocyte reactions were incubated at 37°C, 5% CO2 for 7 days.
- Flow cytometry After 7 days of mixed lymphocyte reaction, samples were washed and then stained with antibodies against cell surface antigens. For intracellular staining FoxP3 staining kit (BD Biosciences) was used according to the manufacturer’s instructions. Samples were stained in the dark at 4°C and washed twice in staining buffer (BD Biosciences), followed by analysis using a BC Cytoflex flow cytometer (Beckman Coulter). T cells were defined as CD3+ cells. The degree of proliferation calculated as a proliferation index (FlowJo software). Tregs were defined as CD3+, CD4+, CD25hi and FoxP3+. Naive T cells were defined as CD3+, CD45RA+, CCR7+. Memory T cells were defined as CD3+, CD45RA-. The amount of naive cells was expressed as a ratio memory cells/naive cells (M/N).
- M/N memory cells/naive cells
- B02 has an improved antibody affinity to OX40L, decreased cytokine production, activation and an increased T-Reg abundance and proliferation compared with prior art anti-OX40L antibody, R4930. These data demonstrate an independent effect of B02 on T-cell activation.
- Core fucosylation of an Fc N-linked glycan affects IgG antibody effector functions, as the absence of fucose increases the antibody dependent cell cytotoxicity (ADCC) response with increased binding to the Fey receptors.
- ADCC antibody dependent cell cytotoxicity
- One method utilised to reduce core fucosylation is to utilise a fucosyltransferase (FUT) inhibitor, 2- fluoro peracetylated fucose (2FF).
- 2FF is a competitive inhibitor of the fucosyltransferase enzyme (FLIT8) required in transferring fucose to the developing glycan in the Golgi (Rillahan et al., 2012).
- N-glycan composition was achieved via sample denaturation followed by enzymatic release via peptide N-glycosidase F (PNGase F, waters).
- PNGase F peptide N-glycosidase F
- the resulting N-glycans are fluorescently labeled with RapiFlour-MS (waters) for detection.
- the fluorescently labeled N-glycans are subjected to HILIC-FLD-MS, TripleTOF 4600 MS (AB SCIEX), HPLC system Nexera X2 (SHIMADZU), running a HILIC column (Waters).
- the N-glycans are then identified using the mass-to-charge ratio (m/z) detected by the mass spectrometry and the relative abundance of each N-glycan can be calculated from the peak area of the HILIC-FLD chromatogram. PeakView software was used to analyze the data.
- N-glycans may also be determined using the following method: The sample is first denatured by Rapigest-SF (Waters) and its N-glycans are then released by PNGase F (Waters or equivalent). The resulting N-glycans are further labeled with fluorescence tag RapiFlour-MS (Waters) for detection purpose and purified by solid phase extraction (SPE, BEH Amide, Waters). Such fluorescently labeled N-glycans are subjected to HILIC-FLD detection (e.g., 1290, Agilent). The N-glycans can be assigned by their retention time, and the percentage of each N-glycan can be calculated from the peak area of HILIC-FLD chromatogram.
- HILIC-FLD detection e.g., 1290, Agilent
- fucosylation)% (G0F-GN)% + (G0F)% + (G1 Fa)% + (G1 Fb)% + (G2F)%.
- fucosylation dropped significantly to 12.98%, while saturation of the fucosylation reduction effect was reached at approximately 150-200 pM, where fucosylation levels stabilised around 3.20%-2.83%.
- Increasing 2-FF to 400 pM resulted in a minimal further reduction to 2.29% fucosylation, indicating the effect plateaued at this concentration.
- Other components of the glycosylation profile such as Man5 and nonattributed peaks, remained consistent across increasing concentrations of 2-FF.
- the glycosylation profile with reduced fucosylation reflects an increase in G0%, with GO increasing from 3.54% at baseline to 75.62% at 400 pM 2-FF, demonstrating the anticipated shift in glycan structure as fucosylation is reduced. Similar results are shown in Table 3 for a different clone with a higher degree of fucosylation.
- CD32a CD16a and C1q 1% BSA in 0.02% PBST was used as a buffer and the temperature was set to 25°C. The Shake speed was set to 1000 rpm.
- FcRn binding buffer pH was set to pH 6.
- Association and dissociation time was 300 seconds for C1q.
- FcRn Association and dissociation time was 60 seconds.
- CD32a and CD16a association time was 60 seconds and dissociation time was 150 seconds.
- Herceptin was used as an internal reference antibody.
- the data acquisition rate is set to Standard kinetics (5.0Hz, averaging by 20). Data analysis was performed using Octet® System Data Analysis (Release 10.0).
- the binding of CD16a, CD32a and C1q to the Fc region of antibody is a weak affinity interaction (fast on and fast off) and is not suitable for complete kinetic analysis.
- the binding curves for each concentration of analyte have reached steady state and can be evaluated for affinity using the steady-state analytical SSG KD values.
- the CD64 and FcRn affinity were evaluated using the kinetics analytical KD values.
- 0X118 induces a concentration-dependent reduction of T cell activation both when fully fucosylated (97%) and when fucosylation is reduced (34%).
- 0X118 had reduced core fucosylation (34%) the reduction of T-cell activation was enhanced compared with fully fucosylated (97%) 0X118.
- CTLA-4 Ig (clone Belatacept, Bristol Myers Squibb, SEQ ID NO: 15) was purchased from Apoteket Hjartat AB.
- Anti-OX40L lgG1 reference antibody (clone R4930/Oxelumab; lgG1 , Fc-active) and Anti-OX40L lgG4 reference antibody (clone Amlitelimab; lgG4, Fc-silent, SEQ ID NO:16, SEQ ID NO:17) were produced via transient expression in CHO K1 by Proteogenix.
- PBMC Peripheral blood mononuclear cells
- Ficoll Paque Plus Chipa density gradient centrifugation from buffy coats (500g x 30 min). Buffy coats were obtained from anonymous healthy adult donors from the Akademiska University Hospital blood bank, and PBMC isolation was performed within 24 hours of blood collection. In line with Swedish legislation, an ethics approval was not needed since the blood bank provides buffy coats for research purposes from anonymous donors and biological material cannot be traced back to a specific individual. Isolated PBMC were mixed in PBS at a concentration of 2.0 x 10 7 cells per mL and stained with CFSE (Thermo Fisher Scientific Inc) according to the manufacturer’s instructions.
- CFSE Thermo Fisher Scientific Inc
- PBMC from 2 different donors were mixed in a two-way allogeneic mixed lymphocyte reaction, i.e., PBMCs from neither donor were inactivated.
- VPD450-stained PBMC were washed and resuspended in 10% heat- inactivated Ultralow IgG FBS (Gibco) in AIM V medium (Gibco, Thermo Fisher Scientific Inc).
- Resuspended PBMCs were dispensed into round-bottom 96-well cell culture plates, and pure medium or medium supplemented with test article was added to a final concentration of 2 x 10 6 cells per mL in a final volume of 200 pL.
- Mixed lymphocyte reactions were incubated at 37°C, 5% CO2 for 7 days. Supernatant postculture was kept and analysed for cytokine content (measuring levels of IL-10, IL-4, TNFa) utilizing Legendplex (BioLegend) cytokine assay according to the manufacture's instructions.
- T cells were defined as CD3+ cells.
- Proliferating T-cells were defined as CFSE low.
- Tregs were defined as CD3+, CD4+, CD25hi and FoxP3+.
- Naive T cells were defined as CD3+, CD45RA+, CCR7+.
- OX118 (10 pg/mL) decreased the proliferation of CD4+ and CD8+ cells compared to control, and other OX40L-targeting antibodies (lgG1 Ref and lgG4; 10 pg/mL) and CTLA-4 Ig (15 pg/mL) ( Figure 4).
- OX118 (10 pg/mL) increased the % of Tregs cells among all proliferating cells compared to control, and other OX40L-targeting antibodies (lgG1 Ref and lgG4; 10 mg/mL) and CTLA-4 Ig (15 pg/mL) (Figure 5A). This effect was observed to be concentration-dependent, with increases in the % of Tregs cells among all proliferating cells observed at OX118 concentrations from 10 2 -10 5 ng/mL ( Figure 5B).
- 0X118 (10 pg/mL) in a two-way mixed lymphocyte reaction resulted in a decrease in IL-4 compared to control.
- 0X118 (10 pg/mL) reduced TNFa levels compared with control and other OX40L-targeting antibody; lgG1 Ref (10 pg/mL), and CTLA-4 Ig (15 pg/mL).
- IL-10 levels remained comparable to control and other OX40L- targeting antibody; lgG1 Ref (10 pg/mL), and CTLA-4 Ig (15 pg/mL) ( Figure 7).
- 0X118 reduced proliferation of CD4+ and CD8+ cells in vitro, while it increased the amount of proliferating Tregs cells. Furthermore, 0X118 decreased levels of the pro- inflammatory cytokines; IL-4 and TNFa in vitro.
- mice Female NSG (NOD. Cg-Prkdc sdd H2rg tmlWl /Szi) mice 6-9 weeks of age were used in the study. Mice were grouped into treatment (control, 0X118 10mg/kg or 0X118 50mg/kg) groups based on body weight.
- PBMCs were obtained from one healthy, random, and uncharacterized donor. PBMCs were used freshly after isolation from leukoreduction system (LRS) chambers.
- LRS leukoreduction system
- the first dose of 0X118 was given intraperitoneally (i.p) 24 hours after PBMC injection, 0X118 was dosed once weekly for 3 consecutive weeks (4 i.p doses at days 1 , 8, 15 and 22). Group sizes were 5 mice per group. Human isotype IgG 1 was used as control vehicle (BioXCell, catalog #BE0297), 0X118 was given in doses of 10 mg/kg (low dose) or 50 mg/kg (high dose) once weekly until day 22. Activity scores were assessed daily (see Table 5). A score of 3 for more than two days or mice with a score ⁇ 3 for one day was euthanized.
- GvHD scores were assessed daily for monitoring progression of GvHD, by using whole number scoring in five different domains ranging from 0 (no symptoms) to 2 (severe symptoms).
- Flow cytometry Blood samples were collected at day 8, 15, 22 for flow cytometry. Cells were pelleted by centrifugation, at 400 x g for 5 min with subsequent removal of supernatant. Fc-block antibody (10 pg/well of a 1 :100 dilution in FC buffer). Then specific antibodies were added as recommended by the manufacturer in Zombie Aqua Fixable Viability stain (diluted 1 :100 in PBS buffer) and the plates were incubated at 4 °C protected from light for 30 min.
- Antibodies utilised were: hCD3 (clone LICHT1, Biolegend), hCD45 (clone H130, Biolegend), hCD4 (clone RPA-T4, Biolegend), hCD8 (clone RPA-T8, Biolegend), hCD25 (clone M-A251, Biolegend), H LA-DR (clone L243, Biolegend), hFoxp3 (clone PCH101, Thermo Scientific; for this antibody an intracellular staining protocol was used). HCD62L (clone DREG-56, Biolegend). Antibodies were used according to the manufacturer’s instructions.
- FC buffer 2% FBS in PBS; FBS (#A21-102 - PAA), PBS (#14190169 - Gibco/Fisher Scientific).
- Fc-block antibody CD16/32 purified (2.4G2), 0.5 mg/ml (#553142 - BD Biosciences). Zombie Aqua Fixable Viability Kit (#423102 - BioLegend).
- Doublet exclusion was performed according to forward scatter height versus forward scatter area to include only single cells followed by forward/sideward scatter to exclude debris and finally live/dead (live/dead, catalog no. 423102, Biolegend) discrimination. Gating was performed according to table 5.
- mice transplanted with human peripheral blood mononuclear cells Following induction of GvHD in mice, defined as rising GvHD score (mean GvHD score at day 30: control: 6,8;
- 0X118 reduces activation of T-cells in peripheral blood following induction of GvHD in mice: 21 days after induction of GvHD in mice, treatment with 0X118 reduced the relative populations of activated CD4+, CD8+ and CD14+ T-cells compared with control. This effect was observed at both low and high doses of 0X118; 10 mg/kg and 50 mg/kg, respectively (Figure 9).
- 0X118 increases proportion of T regulatory cells (T-Regs) among proliferating CD4+ T- cells following induction of GvHD in mice: 21 days after induction of GvHD in mice, treatment with 0X118 (pooled data from all 0X118 treated mice) increased the population of T-regs compared with control ( Figure 10).
- 0X118 treatment at both high and low doses, prevents induction and progression of GvHD in vivo via preventing peripheral chimerism, activation of peripheral T-cell populations and enriching the peripheral T-reg population.
- Example 5 Xenogeneic GvHD model - benchmarking 0X118 (Treatment A) vs antihuman OX40L lgG4 isotyope (Treatment B).
- Aim To evaluate and compare the efficacy of anti-OX40L antibodies with different isotypes in a xenogeneic Graft-versus-Host Disease (xeno-GvHD) model, focusing on chimerism and tissue infiltration by human CD45+ (hCD45+) cells.
- xeno-GvHD xenogeneic Graft-versus-Host Disease
- NOG mice were subjected to whole-body irradiation with a dose of 1.0 Gy. After a 24- hour rest period, 3 million human PBMCs (Peripheral Blood Mononuclear Cells) were injected into the tail vein, marking Day 0. Treatment began on Day 1 , 10 mg/kg doses of Treatment A (0X118), Treatment B (anti-hOX40L lgG4), and a control (human isotype IgG 1 ) were administered. GvHD scores (see Table 6) and body weight were assessed every other day.
- PBMCs Peripheral Blood Mononuclear Cells
- Flow cytometry Blood samples were collected and whole blood was centrifuged at 3000 rpm for 5 minutes at 4°C to isolate plasma (which was frozen at -80 for later cytokine analysis). The cells were resuspended in lysis buffer and incubated for 2 min at room temperature. The residual white blood cells were suspended in DPBS, Fc- block for mouse (rat anti-mouse CD16/CD32), human (Human TruStain FcX) and Live/Dead (Zombie NIR, BioLegend, 426106) was added, the mixture was incubated for 15 min at room temperature in the dark. Subsequently antibodies were added and incubated for 30 minutes at 4°C in the dark.
- treatment A resulted in a reduction of hCD45+ cell infiltration in the colon, skin, and lungs (Fig 11 D), compared with Treatment B.
- Aim To evaluate and compare the ADCC activity of 0X118 in relation to Herceptin® (Trastuzumab; Roche, SH0346) as a positive control.
- 0X118 is a humanized lgG1 antibody targeting OX40L.
- the ADCC Bioassay Effector Cell V variant (High Affinity) (BPS Bioscience, 60541) was used as the effector cells, while SKBR-3 cells (ATCC, HTB-30) were employed as target cells for system control.
- CHO-K1/OX40L cells (GenScript, M00563) were used as the test target cells.
- Herceptin® (Roche, SH0346) was utilized as the system positive control, and Human lgG1 (Sino Biological, MA17JU1250) served as the negative control.
- the assay was conducted using three different lots of 0X118 drug substance (DS).
- Target cells were plated in 96-well plates at a density of 10,000 cells per well. Controls and test samples were added to the wells, and the plates were incubated at 22-28°C for 30 minutes. Effector cells were then added at 60,000 cells per well, and the plates were further incubated in a cell incubator at 37°C with 5% CO 2 for 6 hours.
- the Bio-LiteTM Luciferase Assay System (Vazyme, DD1201) was added at 75 pL per well, and the luminescent signal was measured using a microplate reader. The signal was plotted against the logarithm of the antibody concentration (ng/mL), and dose-response curves were fitted using a four-parameter fit (4P-Fit) via GraphPad software to calculate the median effective concentration (ECso) values.
- 0X118 exhibited potent ADCC activity with significantly lower EC50 values compared to the system positive control, Herceptin®.
- the EC50 values for the three different 0X118 DS lots were consistent, demonstrating robust and reproducible ADCC activity.
- the negative control Human IgG 1
- Aim To evaluate the binding affinity of the Fc region of 0X118 to various Fey receptors, including FcyRla (CD64), FcyRlla (CD32a 131 H, CD32a 131 R), FcyRIHa (CD16a 158V, CD16a 158F), and the neonatal Fc receptor (FcRn), using Herceptin® as a positive control.
- Materials and Methods Biolayer Interferometry (BLI) was employed using the Sartorius RED96e system to assess the binding affinity of 0X118 drug substance (DS) to Fey receptors. The test was performed using three different lots of 0X118 DS: lot #1 toxicology batch), and lots #2 and #3 (GMP DS batches).
- Herceptin® (Roche, SH0346) was used as the system positive control, with Human lgG1 serving as the negative control.
- the FcyR proteins were immobilized, and gradient-diluted antibody samples were introduced for binding analysis after baseline equilibration, followed by dissociation in buffer.
- the assay was conducted at 25°C using 0.02% PBSTB as the running buffer. Data acquisition was performed at standard kinetics (5.0 Hz, averaging 20), and data analysis was conducted using the Octet® System Data Analysis software to calculate the binding kinetics parameters (KD, kon, koff).
- Aim To evaluate the binding affinity of 0X118 to C1q, a key component of the complement system, which is involved in initiating the classical complement pathway. Binding to 01 q is an important functional property for antibodies, as it can trigger immune responses leading to enhanced pathogen elimination. This study compares the binding of 0X118 to 01 q against Herceptin®, a known lgG1 isotype antibody with 01 q binding activity, as a positive control, to determine if 0X118 maintains this essential functional property.
- Biolayer Interferometry using the Sartorius Octet RED96e system was employed to measure the binding affinity of 0X118 drug substance (DS) to the C1q protein.
- Herceptin® (Roche, SH0346) was used as the positive control.
- Three different lots of 0X118 DS were tested: lot #1 (toxicology batch), and lots #2 and #3 (GMP DS batches).
- the assay was performed at 25°C, with 0.02% PBSTB as the running buffer.
- the C1q protein was immobilized on the sensor, and gradient-diluted antibodies were introduced for binding after baseline equilibration. Dissociation was conducted using buffer. Data were acquired at a standard kinetics rate (5.0 Hz, averaging 20), and analysis was performed using the Octet® System Data Analysis software to calculate binding parameters (KD, Rmax).
- 0X118 exhibits binding activity to C1q comparable to Herceptin®, with no significant difference in binding affinity across the tested lots of 0X118. This suggests that 0X118 has the ability to engage with the complement system. The consistency in C1q binding across different production batches supports the robustness of 0X118 manufacturing.
- the novel anti-OX40L monoclonal antibody (mAB; 0X118) is based on affinity maturation and Fc-optimisation of a previously clinically tested mAB; R4930 (US 7,501 ,496 B1). CDR sequences were determined via the Chothia Numbering Scheme. Underlined, bold amino acids are differing from corresponding amino acids of R4930. 0X118 VL sequence (SEQ NO:1)
- Amlitelimab Heavy chain (Anti-QX40L lgG4; SEQ ID NO: 16)
- Tripathi T Tripathi T, Yin W, Xue Y, Zurawski S, Fujita H, Hanabuchi S, Liu YJ, Oh S, Joo H.
- A1.An antibody comprising a heavy chain of SEQ ID NO: 14 and a light chain comprising a VL sequence of SEQ ID NO: 1 , wherein core fucosylation of the heavy chain is below 80%.
- the antibody of item 1 wherein the light chain has the amino acid sequence of SEQ ID NO: 13.
- the antibody of item 1 wherein the core fucosylation is below 70%, such as below 60%, for example below 50%, such as below 40%, for example below 30%, such as below 20% of the total glycans.
- a method of producing an anti-OX40L antibody or antibody derivative comprising expressing polynucleotides encoding polypeptide of SEQ ID NO: 13 and 14 in a cell, wherein the cell is exposed to culture medium comprising an inhibitor of glucosidases I and II, and subsequently purifying the antibody.
- a method of producing an anti-OX40L antibody or antibody derivative comprising expressing polynucleotides encoding polypeptide of SEQ ID NO: 13 and 14 in a cell, wherein the cell is exposed to culture medium comprising a fucose analog, and subsequently purifying the antibody.
- A7 The method of item 6, wherein the fucose analog is an inhibitor of fucokinase, GDP-fucose-pyrophosphorylase, fucosyltransferase (FUT), GDP-mannose 4,6- dehydratase, GDP-fucose synthetase, and/or a fucose transporter, such as GDP-fucose transporter.
- the fucose analog is an inhibitor of fucokinase, GDP-fucose-pyrophosphorylase, fucosyltransferase (FUT), GDP-mannose 4,6- dehydratase, GDP-fucose synthetase, and/or a fucose transporter, such as GDP-fucose transporter.
- the method of item 6, wherein the fucose analog is an inhibitor of a fucosyltransferase (FUT), such as 1,6-fucosyltransferase (FUT8).
- FUT fucosyltransferase
- a method of producing an anti-OX40L antibody or antibody derivative comprising expressing polynucleotides encoding polypeptide of SEQ ID NO: 13 and 14 in a cell that is genetically modified to prevent core fucosylation.
- the cell is selected from the following cells: CHO (e.g., CHO K1 , DXB-11 CHO, Veggie-CHO), COS (e.g., COS-7), Syrian hamster, rat myleloma, mouse myeloma (e.g., SP2/0, NSO), retinal cell, Vero, CV1 , kidney (e.g., HEK293, 293 EBNA, MSR 293, MDCK, HaK, BHK, BHK21), HeLa, HepG2, WI38, MRC 5, Colo205, HB 8065, HL-60, Jurkat, Daudi, A431 (epidermal), CV-1 , U937, 3T3, L cell, C127 cell, MMT 060562, Sertoli cell, BRL 3A cell, HT1080 cell, a human myeloma cell, tumor cell, a human lymphoma cell (e.g., a Namalwa cell
- CHO e.g.
- A1 1. The method according to any one of items 9 or 10, wherein fucosylation of the antibody or antibody derivative is restored and/or titrated to a desired level via exposure to fucose, wherein the cell is exposed to a culture medium comprising fucose.
- a method of testing a defucosylated anti-OX40L antibody or antibody derivative comprising testing the antibody in a T-cell activation assay and verifying that the T-cell activation is reduced compared to a fucosylated anti- OX40L antibody.
- A13 The antibody or antibody derivative according to any one of the preceding items, for use in the prophylaxis and/or treatment of inflammatory disorders.
- A14 The antibody or antibody derivative according to any one of the preceding items, for use in the prophylaxis and/or treatment of autoimmune diseases.
- A15 The antibody or antibody derivative according to items 13-15, wherein the disease is selected from the group consisting of: Graft-vs-host-disease, allograft rejection, asthma, Systemic lupus, arthritis, inflammatory bowel disease, ulcerative colitis, Crohn’s disease, diabetes, atopic dermatitis, psoriasis, hidradenitis suppurativa, immunoglobulin A nephropathy, Hashimoto disease, Graves’ disease, chronic rhinosinusitis and multiple sclerosis.
- a polypeptide comprising an amino acid sequence of SEQ ID NO:1 (VL).
- An anti-QX40L antibody comprising a heavy chain and a light chain, wherein the light chain comprises a VL comprising the following CDRs: g. SEQ ID NO: 2; h. SEQ ID NO: 3; and i. SEQ ID NO: 4; and wherein the heavy chain comprises a constant region and a VH comprising the following CDRs: j. SEQ ID NO: 6; k. SEQ ID NO: 7; and l. SEQ ID NO: 8, and, wherein core fucosylation of the constant region of the heavy chain is below%.
- An anti-QX40L antibody comprising a heavy chain and a light chain, wherein the light chain comprises a VL comprising the following CDRs: m. SEQ ID NO: 2; n. SEQ ID NO: 3; and o. SEQ ID NO: 4; and wherein the heavy chain comprises the following CDRs: p. SEQ ID NO: 6; q. SEQ ID NO: 7; and r. SEQ ID NO: 8, and, wherein core fucosylation of the heavy chain is below 80%.
- An anti-QX40L antibody comprising an IgG 1 heavy chain and a light chain, wherein the light chain comprises a VL comprising the following CDRs: s. SEQ ID NO: 2; t. SEQ ID NO: 3; and u. SEQ ID NO: 4; and wherein the IgG 1 heavy chain comprises the following CDRs: v. SEQ ID NO: 6; w. SEQ ID NO: 7; and x. SEQ ID NO: 8, and, wherein core fucosylation of the I gG 1 heavy chain is below 80%.
- An antibody comprising a light chain comprising a VL sequence of SEQ ID NO: 1 , wherein core fucosylation of the antibody is below 80%.
- An anti-QX40L antibody comprising a heavy chain and light chain, comprising a VL sequence of SEQ ID NO: 1, wherein core fucosylation of the heavy chain is below 80%.
- the antibody comprises an lgG1 heavy chain constant region, wherein the IgG 1 heavy chain constant region is glycosylated.
- An antibody comprising a heavy chain of SEQ ID NO: 14 and a light chain comprising a VL sequence of SEQ ID NO: 1 , wherein core fucosylation of the heavy chain is below 80%.
- An anti-QX40L antibody comprising a heavy chain of SEQ ID NO: 14 and a light chain comprising a VL sequence of SEQ ID NO: 1, wherein core fucosylation of the heavy chain is below 80%.
- the antibody of any of the preceding items comprising a heavy chain of SEQ ID NO: 14 and a light chain of SEQ ID NO: 13.
- the antibody glycans comprise between 50% and 80% of glycan GO, such as between 55 and 80%, such as between 60% and 80%, such as between 65% and 80%, such as between 70% and 80%, such as between 75% and 80%, such as between 50% and 75%, such as between 50% and 70%, such as between 50% and 65%, such as between 50% and 60%, such as between 50% and 55%.
- total glycans of the antibody comprise between 50% and 80% of glycan GO, such as between 55 and 80%, such as between 60% and 80%, such as between 65% and 80%, such as between 70% and 80%, such as between 75% and 80%, such as between 50% and 75%, such as between 50% and 70%, such as between 50% and 65%, such as between 50% and 60%, such as between 50% and 55%.
- HLPC hydrophilic interaction liquid chromatography HPLC
- ESI-MS electrospray ionization mass spectrometry
- HILIC-FLD-MS/MS hydrophilic interaction liquid chromatography equipped with fluorescence detection and tandem mass spectrometry
- a method of producing an anti-OX40L antibody or antibody derivative comprising expressing polynucleotides encoding polypeptide of SEQ ID NO: 13 and 14 in a cell, wherein the cell is exposed to culture medium comprising an inhibitor of glucosidases I and II, and subsequently purifying the antibody.
- inhibitor selected from the group comprising: castanospermine, deoxymannojirimycin and australine.
- a method of producing an anti-OX40L antibody or antibody derivative comprising expressing polynucleotides encoding polypeptide of SEQ ID NO: 13 and SEQ ID NO: 14 in a cell, wherein the cell is exposed to culture medium comprising a fucose analog, and subsequently purifying the antibody.
- the fucose analog is an inhibitor of fucokinase, GDP-fucose-pyrophosphorylase, fucosyltransferase (FUT), GDP-mannose 4,6-dehydratase, GDP-fucose synthetase, and/or a fucose transporter, such as GDP-fucose transporter.
- the fucose analog is an inhibitor of a fucosyltransferase (FUT), such as 1 ,6-fucosyltransferase (FUT8).
- FUT fucosyltransferase
- the fucose analog is selected from the group comprising of L-fucose, 2-fluro-fucose, 2- fluoro peracetylated fucose (2FF), 5-alkynyl-fucose, alkynyl fucose monoacetate, alkynyl fucose triacetate, alkynyl fucose diacetate and 5-alkynyl-fucose peracetate.
- any one of items 36 to 37, wherein the fucose analog is 2- fluoro peracetylated fucose (2FF).
- the method of any one of items 36 to 41 wherein the culture medium is supplemented with the fucose analog to maintain an effective concentration thereof.
- a method of producing an anti-OX40L antibody or antibody derivative comprising expressing polynucleotides encoding polypeptide of SEQ ID NO: 13 and SEQ ID NO: 14 in a cell that is genetically modified to prevent core fucosylation.
- the method according to item 43 wherein the cell encodes a functional (3-1,4- N-acetylglucosaminyltransferase (GnTIII), GDP-6-deoxy-D-lyxo-4-hexulose reductase (RMD) and/or a Golgi a-mannosidase II (Manll).
- GnTIII 3-1,4- N-acetylglucosaminyltransferase
- RMD GDP-6-deoxy-D-lyxo-4-hexulose reductase
- Manll Golgi a-mannosidase II
- the cell is selected from the following cells: CHO (e.g., CHO K1 , DXB-11 CHO, Veggie-CHO), COS (e.g., COS-7), Syrian hamster, rat myleloma, mouse myeloma (e.g., SP2/0, NSO), retinal cell, Vero, CV1 , kidney (e.g., HEK293, 293 EBNA, MSR 293, MDCK, HaK, BHK, BHK21), HeLa, HepG2, WI38, MRC 5, Colo205, HB 8065, HL-60, Jurkat, Daudi, A431 (epidermal), CV-1 , U937, 3T3, L cell, C127 cell, MMT 060562, Sertoli cell, BRL 3A cell, HT1080 cell, a human myeloma cell, tumor cell, a human lymphoma cell (e.g., a Nam
- a method of producing an anti-OX40L antibody or antibody derivative comprising expressing polynucleotides encoding polypeptide of SEQ ID NO: 13 and SEQ ID NO:14 in a cell, and subsequently purifying and optionally folding the antibody.
- cell is selected from the group consisting of mammalian cells, bacterial cells, yeast cells and plant cells.
- any one of items 33 to 55, wherein the antibody derivative comprises an antibody Fc region and a ligand binding domain of a nonimmunoglobulin protein.
- the method according to any one of the items 33 to 56, wherein the method comprises an additional step of isolating the antibody.
- the method according to any one of the items 33 to 57, wherein the method comprises an additional step of purifying the antibody.
- a method of testing the potency of a defucosylated anti-OX40L antibody or antibody derivative comprising testing the antibody in a T-cell activation assay and verifying that the T-cell activation is reduced compared to a fucosylated anti-OX40L antibody.
- a method of testing the potency of a defucosylated anti-OX40L antibody or antibody derivative according to any of the preceding items comprising testing the antibody in a T-cell activation assay and verifying that the T-cell activation is reduced compared to a fucosylated anti-OX40L antibody.
- a pharmaceutical composition comprising an antibody of any one of items 1 to 32 and at least one pharmaceutically acceptable excipient.
- the antibody or antibody derivative according to any one of the preceding items for use in the prophylaxis and/or treatment of at least one disease or disorder that is associated with OX40L.
- the antibody or antibody derivative according to any one of the preceding items for use in the prophylaxis and/or treatment of inflammatory disorders.
- 66. The antibody or antibody derivative according to any one of the preceding items, for use in the prophylaxis and/or treatment of autoimmune diseases.
- a kit comprising the antibody or antibody derivative of any one of items 1 to 32.
Landscapes
- Health & Medical Sciences (AREA)
- Immunology (AREA)
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Life Sciences & Earth Sciences (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Engineering & Computer Science (AREA)
- General Health & Medical Sciences (AREA)
- Medicinal Chemistry (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- General Chemical & Material Sciences (AREA)
- Genetics & Genomics (AREA)
- Biophysics (AREA)
- Biochemistry (AREA)
- Transplantation (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Molecular Biology (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Pharmacology & Pharmacy (AREA)
- Animal Behavior & Ethology (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Peptides Or Proteins (AREA)
Abstract
The present invention relates to antibodies and antibody derivatives against OX40L with improved effects compared to prior art antibodies.
Description
Improved anti-OX40L antibodies
Technical field
The present invention relates to antibodies and antibody derivatives against OX40L with improved effects compared to prior art antibodies
Background
The immune system consists of several different cell types and is divided into innate and adaptive immune systems. On a cellular level, the innate immune system consists of cells such as macrophages, dendritic cells, monocytes, and granulocytes whilst the adaptive immune system consists of T- and B-cells. Cells of the innate immune system play a significant role in bridging the innate-adaptive immune response.
Activation of T-cells occurs through two cell-mediated signals, firstly the MHC (presenting the foreign antigen) and TOR interaction, and secondly through costimulatory molecules (OX40/OX40L, CD40L/CD40, etc.). The second signal is necessary for the activation of T-cells. Thirdly, soluble factors such as cytokines shape the final T-cell response.
Human OX40L (gp34, CD252, SwissProt P23510), belongs to the tumour necrosis factor superfamily (TNFSF) group and can be expressed on B-cells, dendritic cells, macrophages, monocytes, endothelial cells, fibroblasts (Review: Weinberg, A. D., Trends Immunol. 23 (2002) 102-109). Its cognate receptor, 0X40, is expressed on T- cells. OX40/OX40L is a co-stimulatory pathway. It is not constitutively expressed but rather induced upon activation of T-cells after around 2-3 days. The interaction between 0X40 and OX40L has been studied and in its crystallizable form, the OX40L exists in a trimeric form and interacts with 0X40 as a trimer (Compaan et Hymowitz., 2006).
Yoshioka, A., et al. (2000) demonstrated the therapeutic potential of a neutralizing anti- mOX40L mAb in a mouse model for rheumatoid arthritis. Administration of the antibody dramatically ameliorated the disease severity. This antibody showed similar activities in other related disease models, e.g., inflammatory skin disease, experimental autoimmune disease (EAE), GvHD, and inflammatory bowel disease.
Adverse T-cell activation is a pathological driver in several diseases such as autoimmune disease {e.g., diabetes, multiple sclerosis, SLE, rheumatoid arthritis), inflammatory conditions (asthma, atopic dermatitis, chronic rhinosinusitis, inflammatory bowel disease), transplant-related conditions (graft-versus-host disease, allograft rejection). By hindering activation and proliferation of disease-causing T-cells diseases have been shown to be ameliorated in several examples (Chatenoud et al., 1994; Haile et a!., 2020).
Monoclonal antibodies can have a variety of post-translational modifications, including glycosylation. Monoclonal antibodies, such as IgGs, have an N-linked glycosylation site at asparagine 297 (Asn297) of each heavy chain (two per intact antibody). The glycans attached to Asn297 on antibodies are typically complex biantennary structures with very low or no bisecting N-acetylglucosamine (bisecting GIcNAc) with low amounts of terminal sialic acid and variable amounts of galactose. The glycans also usually have high levels of core fucosylation. Reduction of core fucosylation in antibodies has been shown to alter Fc receptor affinity, altering the Fc-mediated effector functions. In particular, the absence of core fucose on the Fc N-glycan has been shown to increase IgG 1 Fc binding affinity to the FcyRllla present on immune effector cells such as natural killer cells and lead to enhanced ADCC activity. This observation has led to interest in the production of antibodies with reduced core fucosylation.
Other anti-OX40L antibodies
Antibodies against human OX40L are known. One example is Oxelumab (clone R4930), which previously underwent development for the treatment of asthma. The antibody is described in US 7501496 B1.
Amlitelimab (also known as KY1005/SAR445229) is another anti-OX40L antibody for the treatment of atopic dermatitis. It is described in US9139653B1 and WO2015/132580 A1. Amlitelimab is an lgG4 isotype, non-depleting antibody. In WO 2018/083248, the effect of Amlitelimab in a primate GVHD model is shown.
Summary
The invention provides antibodies and antibody derivatives and methods for producing anti-OX40L antibodies and antibody derivatives with reduced core fucosylation. The
antibodies of the invention are Fc engineered such that the affinities for various Fey receptors are increased.
Current clinical agents for autoimmune diseases and inflammatory disorders treatment often cause substantial adverse effects and safety concerns, owing to non-specific immune modulation. Indeed, broad depletion of T-cells without distinguishing the pathological population is associated with several adverse outcomes such as increased risk of cancer, increased graft failure, disease relapse, drug toxicity, and compromised immunity to post-transplant infections, such as cytomegalovirus, which ultimately limits long-time use.
The antibodies and antibody derivatives and methods are premised in part on the unexpected results presented in the examples showing that affinity maturation and optimisation of the Fc-region to reduce core fucosylation, produces antibodies and antibody derivatives with improved affinity to OX40L and Fc-receptors. Such antibodies and antibody derivatives exhibit increased effector function (ADCC), reduced T-cell activation, enriched regulatory T cell (T-Reg) populations, and maintenance of naive immune cell populations, as compared with antibodies or antibody derivatives present in the prior art. Said improved features are observed both in vitro and in vivo.
Indeed, the antibodies and antibody derivatives and methods of the invention exhibit elevated affinity for the human OX40L antigen as compared to a known anti-OX40L antibody; Oxelumab. In addition, the antibodies and antibody derivatives and methods of the invention exhibit an unexpectedly significant reduction in the proliferation of CD4+ and CD8+ T-cells, as compared to antibodies and other binding proteins of the prior art which are known to manipulate activation of T-cells via APC targeting (Belatacept, Oxelumab and Amlitelimab). Further, the antibodies and antibody derivatives and methods of the invention induce a significant increase in the population of T-Regs and naive CD4+ T-cells among the proliferating cell population, as compared to antibodies and other binding proteins of the prior art which are known to manipulate activation of T-cells via APC targeting (Belatacept, Oxelumab and Amlitelimab). These data are unexpected as the level of T cell control and induction of a regulatory T cell phenotype, among proliferating cells, has not been described previously. Concomitant to this, the antibodies and antibody derivatives and methods of the invention reduce TNFa, and IL-4 levels compared with control and another OX40L-
targeting antibody. Surprisingly, the antibodies and antibody derivatives and methods of the invention exhibited a (3-4-fold) increase in the control of chimerism/graft proliferation in vivo, compared to previously published non-Fc region optimised anti- OX40L antibodies (Tripathi et al. 2019). Interestingly, the superior effects of the antibodies and antibody derivatives and methods of the invention were observed relative to both Fc-active (Oxelumab) and Fc-silent (Amlitelimab) anti-OX40L antibodies known in the prior art, thus highlighting the unexpected effect of reduced core fucosylation of the antibodies and antibody derivatives and methods of the invention results in the examples.
In view of the examples, the antibodies and antibody derivatives and methods of the invention exhibit superiority to anti-OX40L antibodies of the prior art. The present invention provides for an alternative strategy to control T cells in a non-depleting manner whilst the naive phenotype of the immune cell population is maintained (/.e., naive T-cells and Tregs). T cells are indirectly controlled by blockade of OX40-OX40L. This strategy is validated by the prominent contribution of effector T cells in the pathogenesis of many autoimmune diseases and inflammatory disorders, and the preferential location of co-stimulatory receptor-ligand pair OX40-OX40L at the inflamed sites. In turn, the complex downstream effects of targeting T-cells directly are prevented and the adaptive immune system's capability to regulate itself is preserved.
The antibodies and antibody derivatives have new and inventive properties, thereby resulting in benefits for patients in need of an antibody-based therapy against OX40L, especially for patients suffering from inflammatory disorders and autoimmune diseases, including rheumatoid arthritis, allergic asthma, and GvHD in transplantation.
In one aspect, the present invention provides for a polypeptide comprising an amino acid sequence of SEQ ID NO:1 (VL).
In another aspect, the present invention provides for an anti-OX40L antibody having a variable light chain and a variable heavy chain, wherein the variable light chain comprises the following CDRs: a. SEQ ID NO: 2; b. SEQ ID NO: 3; and c. SEQ ID NO: 4;
and wherein the variable heavy chain comprises the following CDRs: d. SEQ ID NO: 6; e. SEQ ID NO: 7; and f. SEQ ID NO: 8.
In another aspect, the present invention provides for an anti-QX40L antibody comprising a heavy chain and a light chain, wherein the light chain comprises a VL comprising the following CDRs: a. SEQ ID NO: 2; b. SEQ ID NO: 3; and c. SEQ ID NO: 4; and wherein the heavy chain comprises a constant region and a VH comprising the following CDRs: d. SEQ ID NO: 6; e. SEQ ID NO: 7; and f. SEQ ID NO: 8, and, wherein core fucosylation of the constant region of the heavy chain is below 80% determined as % fucosylated glycans of total glycans.
In some aspects, the core fucosylation of the constant region of the heavy chain is below 80%, as determined by capillary electrophoresis, LC-MS, High-Performance liquid chromatography with Fluorescence Detection (HPLC-FD), High-Performance Anion-Exchange chromatography with Pulsed Amperometric Detection (HPAE-PAD), mass spectrometry, hydrazinolysis and/or enzyme digestion.
In another aspect, the present invention provides for an anti-QX40L antibody comprising an IgG 1 heavy chain and a light chain, wherein the light chain comprises a VL comprising the following CDRs: a. SEQ ID NO: 2; b. SEQ ID NO: 3; and c. SEQ ID NO: 4; and wherein the IgG 1 heavy chain comprises the following CDRs: d. SEQ ID NO: 6; e. SEQ ID NO: 7; and f. SEQ ID NO: 8, and, wherein core fucosylation of the lgG1 heavy chain is below 80%.
In another aspect, the present invention provides for an antibody comprising a light chain comprising a VL sequence of SEQ ID NO: 1 , wherein core fucosylation of the antibody is below 80%.
In another aspect, the present invention provides for an anti-OX40L antibody comprising a heavy chain and light chain, comprising a VL sequence of SEQ ID NO: 1, wherein core fucosylation of the heavy chain is below 80%.
In some embodiments, the antibody comprises an lgG1 heavy chain. In some embodiments, the antibody comprises an lgG1 heavy chain constant region. In some embodiments, the antibody comprises an IgG 1 heavy chain constant region, wherein the lgG1 heavy chain constant region is glycosylated.
In another aspect, the present invention provides for an antibody comprising a heavy chain of SEQ ID NO: 14 and a light chain comprising a VL sequence of SEQ ID NO: 1, wherein core fucosylation of the heavy chain is below 80%.
In another aspect, the present invention provides for an anti-QX40L antibody comprising a heavy chain of SEQ ID NO: 14 and a light chain comprising a VL sequence of SEQ ID NO: 1 , wherein core fucosylation of the heavy chain is below 80%.
In some embodiments, the antibody comprises a variable heavy chain of SEQ ID NO: 5, and a variable light chain of SEQ ID NO: 1. In another embodiment, the antibody comprises a heavy chain of SEQ ID NO: 14 and a light chain of SEQ ID NO: 13.
The antibodies according to the invention are preferentially characterised as being affinity matured fully human anti-QX40L antibodies of subclass lgG1.
In one aspect, the present invention provides for a method of producing an anti-QX40L antibody or antibody derivative, comprising expressing polynucleotides encoding polypeptide of SEQ ID NO: 13 and 14 in a cell, wherein the cell is exposed to culture medium comprising an inhibitor of glucosidases I and II, and subsequently purifying the antibody.
In one aspect, the present invention provides for a method of producing an anti-OX40L antibody or antibody derivative, comprising expressing polynucleotides encoding polypeptides of SEQ ID NO: 13 and 14 in a cell, wherein the cell is exposed to culture medium comprising a fucose analog, and subsequently purifying the antibody.
In one aspect, the present invention provides for a method of producing an anti-OX40L antibody or antibody derivative, comprising expressing polynucleotides encoding polypeptides of SEQ ID NO: 13 and 14 in a cell that is genetically modified to prevent core fucosylation.
In one aspect, the present invention provides for a method of producing an anti-OX40L antibody or antibody derivative, comprising expressing polynucleotides encoding polypeptide of SEQ ID NO: 13 and 14 in a cell, and subsequently purifying and optionally folding the antibody.
In one aspect, the present invention provides for a method of testing a defucosylated anti-QX40L antibody or antibody derivative, comprising testing the antibody in a T-cell activation assay and verifying that the T-cell activation is reduced compared to a fucosylated anti-QX40L antibody.
Description of Drawings
Figure 1. Inhibition of fucosylation by 2-fluorofucose (2-FF). X-axis denotes 2-FF concentration (pM). Y-axis denotes relative fucosylation (%). Reduction to less than 40% fucosylation was achieved following treatment with 200 pM 2-FF.
Figure 2. Affinity of OX118 towards various Fc-receptors. X-axis denotes the degree of fucosylation. Y-axis denotes affinity towards Fc-receptors. Reduced degree of fucosylation resulted in increased affinity for CD16a (FcRgllla/CD16a 158V, FcRgllla /CD16a 158F) without influence on FcRn, C1q or CD32a binding.
Figure 3. Decreased CD3+ T cell activation following OX118 treatment.
X-axis concentrations of antibody in ng/mL. Y-axis degree of T cell activation (CD3+).
Two variants of 0X118 were compared, one with reduced fucosylation (34%) and one
with complete fucosylation (97%). Reduction in the degree of fucosylation resulted in a reduction in T cell activation.
Figure 4. 0X118 decreases proliferation of T cells compared with different anti- OX40L antibodies and CTLA-4 Ig. (A) CD4+ and (B) CD8+ T cells following treatment with different anti-OX40L antibodies; anti-OX40L (10 pg/mL) and CTLA-4 Ig (15 pg/mL). X-axis denotes treatment condition. Y-axis denotes the percentage of proliferating T cells (CFSE low) in a two-way MLR setting after 7 days of incubation. 0X118 (10 pg/mL) resulted in a lower degree of both proliferating CD4+ and CD8+ T cells compared to control and compared to other forms of OX40L targeting (10 pg/mL) and CTLA-4 Ig (15 pg/mL).
Figure 5. 0X118 increases the proportion of T-regulatory cells (T-Regs) among proliferating CD4+ T-cells. (A) The addition of 0X118 (10 pg/mL) in a two-way MLR resulted in an increased regulatory phenotype (Tregs, CD25hi, CD4+, FoxP3+) among proliferating CD4+ cells compared to control, other forms of anti-OX40L antibodies (10 pg/mL) and CTLA-4 Ig (15 pg/mL). (B) Concentration-dependent enrichment in T-reg population among proliferating CD4+ cells compared to control, other forms of anti- OX40L antibodies, and CTLA-4 Ig. X-axis concentration of test item (ng/mL).
Figure 6. 0X118 increases the proportion of naive CD4+ T-cells among proliferating CD4+ T-cells. X-axis denotes treatment condition. Y-axis denotes the percentage of naive (CD3+, CD4+, CD45RA+, CCR7+, CFSE low) CD4+ T cells among proliferating cells. Addition of 0X118 at a concentration (10 pg/mL) in a two- way MLR resulted in increased naive T cells among proliferating CD4+ cells compared to control and anti-OX40L lgG1 ref (10 pg/mL) and CTLA-4 Ig (15 pg/mL).
Figure 7. 0X118 prevents induction of pro-inflammatory phenotype. X-axis denotes treatment condition. Y-axis denotes cytokine concentration (ng/mL); (A) IL-10, (B) II-4, and (C) TNFa in the supernatant. Addition of 0X118 (10 pg/mL) in a two-way MLR resulted in decreased IL-4 and TNFa compared to control. IL-10 levels were comparable to control.
Figure 8. 0X118 treatment prevents GvHD development in mice transplanted with human peripheral blood mononuclear cells (PBMCs). Y-axis denotes the frequency
of circulating human CD45+ leukocytes (hCD45+) in mice. X-axis denotes days (7, 14, and 21) post-induction of GvHD. Peripheral blood chimerism or graft proliferation. Flow cytometry-assisted immune phenotyping was performed on Day 7, Day 14, and Day 21. hCD45+ cells were counted and compared between treatment and control groups.
Figure 9. 0X118 reduces activation of T-cells in peripheral blood following induction of GvHD in mice. X-axis denotes treatment. Y-axis denotes the percentage of activated CD4+, CD8+ T cells, and CD14+ monocyte cells in peripheral blood.
Figure 10. 0X118 increases proportion of T regulatory cells (T-Regs) among proliferating CD4+ T-cells following induction of GvHD in mice. X-axis denotes treatment. The Y-axis denotes the percentage of regulatory T cells among CD4+ cells. Regulatory T cells were defined as CD3+, CD4+, CD25, and FoxP3+ via FACS, data from 22 days post-GvHD induction.
Figure 11. Treatment A potently suppresses GvHD reaction in NSG mice. A) Showing clinical score, with lower score in Treatment A treated mice compared to control and Treatment B. B) Body weight changes throughout the study. C) Chimerism (as defined by percentage hCD45 positive cells) with markedly lower chimerism in Treatment A treated animals. D) Tissue-infiltrating hCD45+ cells expressed as positive rate ((Positive rate/Total Cells)*100 in skin, lung and colon.
Figure 12. ADCC assay comparing three lots of produced 0X118 vs trastuzumab.
The X-axis denotes treatment concentration. The Y-axis denotes luminescence intensity (corresponding to ADCC signalling in report cell line). Batches of 0X118 show reproducible results with high lot to lot consistency. The high affinity towards FcYRIIIa of 0X118 results in a low EC50 for ADCC signalling in the assay compared to a control antibody (trastuzumab).
Detailed description
Definitions
As used herein, the singular forms “a”, “an” and “the” include plural referents unless the context clearly states otherwise. Thus, for example, reference to “an antibody” includes a plurality of such constructs.
The term “some embodiments” can include one, or more than one embodiment.
As used herein "antibody" and "antibodies" (immunoglobulins) may be an oligoclonal antibody, a polyclonal antibody, a monoclonal antibody (including full-length monoclonal antibodies), a camelised antibody, a chimeric antibody, a CDR-grafted antibody, a multi-specific antibody, a bi-specific antibody, a catalytic antibody, a chimeric antibody, a humanized antibody, a fully human antibody, an anti-idiotypic antibody and antibodies that can be labeled in soluble or bound form as well as fragments, variants or derivatives thereof, either alone or in combination with other amino acid sequences provided by known techniques. An antibody may be from any species. An antibody comprises a polypeptide or group of polypeptides that are comprised of at least one binding domain that is formed from the folding of polypeptide chains having three-dimensional binding spaces with internal surface shapes and charge distributions complementary to the features of an antigenic determinant of an antigen. An antibody typically has a tetrameric form, comprising two identical pairs of polypeptide chains, each pair having one "light" and one "heavy" chain. The variable regions of each light/heavy chain pair form an antibody binding site. Native antibodies are usually heterotetrameric glycoproteins of about 150,000 daltons, composed of two identical light (L) chains and two identical heavy (H) chains. Each light chain is linked to a heavy chain by one covalent disulfide bond, while the number of disulfide linkages varies between the heavy chains of different immunoglobulin isotypes. Each heavy and light chain also has regularly spaced intrachain disulfide bridges. Each heavy chain has at one end a variable domain (VH) followed by a number of constant domains. Each light chain has a variable domain at one end (VL) and a constant domain at its other end; the constant domain of the light chain is aligned with the first constant domain of the heavy chain, and the light chain variable domain is aligned with the variable domain of the heavy chain. Light chains are classified as either lambda chains or kappa chains based on the amino acid sequence of the light chain constant region. The variable domain of a kappa light chain may also be denoted herein as VK. The term "variable
region" may also be used to describe the variable domain of a heavy chain or light chain. Particular amino acid residues are believed to form an interface between the light and heavy chain variable domains. The variable regions of each light/heavy chain pair form an antibody binding site.
The term "antibody derivative" means an antibody, as defined above (including an antibody fragment), or Fc domain or region of an antibody comprising a complex N- glycoside linked sugar chain, that is modified by covalent attachment of a heterologous molecule such as, e.g., by attachment of a heterologous polypeptide (e.g., a ligand binding domain of heterologous protein), or by glycosylation (other than core fucosylation), deglycosylation (other than non-core fucosylation), acetylation, phosphorylation or other modification not normally associated with the antibody or Fc domain or region.
The term “antibody-dependent cellular cytotoxicity (ADCC)” is a function mediated by Fc receptor binding and refers to the lysis of OX40L expressing target cells by an antibody according to the invention in the presence of effector cells.
The term "monoclonal antibody" refers to an antibody that is derived from a single cell clone, including any eukaryotic or prokaryotic cell clone, or a phage clone, and not the method by which it is produced. Thus, the term "monoclonal antibody" is not limited to antibodies produced through hybridoma technology.
The term "Fc region" refers to the constant region of an antibody, e.g., a Cnl-hinge- CH 2-CH 3 domain, optionally having a CH 4 domain, or a conservatively substituted derivative of such an Fc region.
The term “Glycan” or "N-glycan" refers to an oligosaccharide comprising a core pentasaccharide Man3GlcNAc2. The N-glycan can be attached to a protein
(glycoprotein) via the nitrogen of an asparagine (or occasionally arginine) residue, or free in solution. In the context of the present disclosure, the term "glycan" refers to an
N-glycan unless otherwise specified. The term "oligosaccharide" refers to a glycan that is not covalently bound to a protein. A useful reference for glycan, glycoprotein, and oligosaccharide nomenclature can be found at the website
.html N-glycan variability is comprehensive but
differences with functional importance are often limited to mannose content and degree of fucosylation (Reusch et al. 2015).
The term "glycosylation pattern" or “glycosylation profile” refers to the observed glycosylation of a given glycoprotein or glycoproteins. A glycoprotein with a greater number of covalently linked sugar residues in the oligosaccharide chain is said to have an increased or more extensive glycosylation pattern and/or an increased total glycosylation. Conversely, a glycoprotein with fewer covalently linked sugar residues in the oligosaccharide chain is said to have a decreased or less extensive glycosylation pattern and/or a decreased total glycosylation. The glycosylation pattern of a monoclonal antibody is variable depending on the method of synthesis or production. The term "glycosylation pattern" as used herein also refers to a characteristic distribution of several different glycosylation patterns on individual glycoproteins. In this sense, an increased glycosylation pattern means an increase in the characteristic distribution of glycosylation patterns of the expressed glycoproteins.
The term “total glycosylation” or “total glycans” used herein refers to the total amount of individual glycan monosaccharides as quantified by known methods in the art, such as capillary electrophoresis, High-Performance liquid chromatography with Fluorescence Detection (HPLC-FD), High-Performance Anion-Exchange chromatography with Pulsed Amperometric Detection (HPAE-PAD), and mass spectrometry.
The term "Fc domain" refers to the constant region domain of an antibody, e.g., a CH I, hinge, CH 2, CH 3, or CH 4 domain, or a conservatively substituted derivative of such an Fc domain.
The term “activated T cell," as used herein, refers to a T-cell that expresses antigens indicative of T-cell activation (that is, T cell activation markers). Examples of T cell activation markers include but are not limited to, CD25, CD26, CD30, CD38, CD69, CD70, CD71 , ICOS, OX-40, H LA-DR, and 4-1 BB. The expression of activation markers can be measured by techniques known to those of skill in the art, including, for example, western blot analysis, northern blot analysis, RT-PCR, immunofluorescence assays, and fluorescence-activated cell sorter (FACS) analysis.
"Regulatory T cells," or Tregs, are a type of immune cells characterized by the expression of the biomarkers CD4, FOXP3, and CD25. Tregs are sometimes referred to as suppressor T cells and represent a subpopulation of T cells that modulate the immune system, maintain tolerance to self-antigens, and prevent autoimmune disease. Tregs are immunosuppressive and generally suppress or downregulate induction and proliferation of effector T (Teff) cells. Tregs can develop in the thymus (so-called CD4+ Foxp3+ "natural" Tregs) or differentiate from naive CD4+ T cells in the periphery, for example, following exposure to TGFp or retinoic acid. Tregs can express cell surface GARP-proTGFpi.
The term “OX40L," as used herein, refers to cognate ligand of the tumour necrosis factor receptor 0X40 (CD134). OX40L functions as a T cell co-stimulatory molecule. OX40-OX40L interactions have been proposed as a potential therapeutic target for treating autoimmunity.
Anti-OX40L-Antibody
In one aspect, the present invention provides for a polypeptide comprising an amino acid sequence of SEQ ID NO:1 (VL).
In another aspect, the present invention provides for an anti-OX40L antibody having a variable light chain and a variable heavy chain, wherein the variable light chain comprises the following CDRs: a. SEQ ID NO: 2; b. SEQ ID NO: 3; and c. SEQ ID NO: 4; and wherein the variable heavy chain comprises the following CDRs: d. SEQ ID NO: 6; e. SEQ ID NO: 7; and f. SEQ ID NO: 8.
In another aspect, the present invention provides for an anti-QX40L antibody comprising a heavy chain and a light chain, wherein the light chain comprises a VL comprising the following CDRs: a. SEQ ID NO: 2; b. SEQ ID NO: 3; and
c. SEQ ID NO: 4; and wherein the heavy chain comprises a constant region and a VH comprising the following CDRs: d. SEQ ID NO: 6; e. SEQ ID NO: 7; and f. SEQ ID NO: 8, and, wherein core fucosylation of the constant region of the heavy chain is below 80%.
In another aspect, the present invention provides for an anti-QX40L antibody comprising an IgG 1 heavy chain and a light chain, wherein the light chain comprises a VL comprising the following CDRs: a. SEQ ID NO: 2; b. SEQ ID NO: 3; and c. SEQ ID NO: 4; and wherein the IgG 1 heavy chain comprises the following CDRs: d. SEQ ID NO: 6; e. SEQ ID NO: 7; and f. SEQ ID NO: 8, and, wherein core fucosylation of the lgG1 heavy chain is below 80%.
In some aspects, the core fucosylation of the constant region of the heavy chain is below 80% , as determined by capillary electrophoresis, LC-MS, High-Performance liquid chromatography with Fluorescence Detection (HPLC-FD), High-Performance Anion-Exchange chromatography with Pulsed Amperometric Detection (HPAE-PAD), mass spectrometry, hydrazinolysis and/or enzyme digestion.
In another aspect, the present invention provides for an antibody comprising a light chain comprising a VL sequence of SEQ ID NO: 1 , wherein core fucosylation of the antibody is below 80%.
In another aspect, the present invention provides for an anti-QX40L antibody comprising a heavy chain and light chain, comprising a VL sequence of SEQ ID NO: 1 , wherein core fucosylation of the heavy chain is below 80%.
In some embodiments, the antibody comprises an lgG1 heavy chain. In some embodiments, the antibody comprises an lgG1 heavy chain constant region. In some embodiments, the antibody comprises an IgG 1 heavy chain constant region, wherein the IgG 1 heavy chain constant region is glycosylated.
In another aspect, the present invention provides for an antibody comprising a heavy chain of SEQ ID NO: 14 and a light chain comprising a VL sequence of SEQ ID NO: 1, wherein core fucosylation of the heavy chain is below 80%.
In another aspect, the present invention provides for an anti-OX40L antibody comprising a heavy chain of SEQ ID NO: 14 and a light chain comprising a VL sequence of SEQ ID NO: 1 , wherein core fucosylation of the heavy chain is below 80%.
In some embodiments, the antibody comprises a variable heavy chain of SEQ ID NO: 5, and a variable light chain of SEQ ID NO: 1. In another embodiment, the antibody comprises a heavy chain of SEQ ID NO: 14 and a light chain of SEQ ID NO: 13.
Glycosylation pattern
Fc glycosylation is provides structural integrity to the antibody, and alterations in glycosylation patterns due to differences in production conditions have been reported to affect susceptibility to proteolytic degradation, clearance rate in vivo, Fey receptor binding and activation, antibody-dependent cellular cytotoxicity (ADCC) and C1 q component binding mediated complement activation. Alterations in glycosylation could therefore compromise effector functions including bioactivity, clinical efficacy, pharmacokinetics, safety, stability, and antigenicity.
The person skilled in the art will anticipate some heterogeneity in glycosylation pattern as a result of differing methods of production which lead to glycosylation patterns or relative abundance of each glycan species or total glycans, reduce reproducibility, and increase variability between batches, leading to variations in product quality. Indeed, heterogeneity in a glycosylation pattern can be influenced by cellular expression systems, culture conditions, and purification schemes. Determination of glycosylation pattern in glycoprotein-based drugs is recommended by the FDA and EMA, which suggest that the oligosaccharide content of glycoprotein products should be examined
to ensure product consistency. Accordingly, the antibodies of the present invention may have variability in their glycosylation pattern and/or total glycosylation, such as +/- 1%, such as +/- 5%, such as +/-10%, such as +/- 15%, such as +/- 20%, such as +/- 25%, such as +/- 30%, such as at least 30%, such as at most 30%.
The person skilled in the art will appreciate that the glycosylation pattern of antibodies can be determined by known suitable methods, such as capillary electrophoresis (Wacker et a/. 2011), High-Performance liquid chromatography with Fluorescence Detection (HPLC-FD), High-Performance Anion-Exchange chromatography with Pulsed Amperometric Detection (HPAE-PAD), and mass spectrometry. All aforementioned methods of determination of glycosylation pattern are subject to interpretation of results, such as interpretation of chromatographs. Interpretation of results may lead to variability in glycosylation patterns, such as +/- 5% variability. This can vary depending on the method utilised, all methods include an error of between 2- 5%, such as between 2-4%.
The present invention provides for anti-OX40L antibodies and antibody derivatives which have a standard N-Glycan (end-glycosylated) pattern of a human lgG1 expressed in CHO cells (for example CHO-K1 cells), for example as described in Reusch & Tajeda, 2015 (“Fc glycans of therapeutic antibodies as critical quality attributes”, Glycobiology, Volume 25, Issue 12, December 2015, Pages 1325-1334) and/or Luo & Zhang 2023 (“Benchmark glycan profile of therapeutic monoclonal antibodies produced by Mammalian cell expression systems”, Pharma. Res.) but with a reduced degree of fucosylation. Luo & Zhang 2023 in Table 1 describes the structure of the most common N-Glycan structures and terminal epitopes in FDA-approved monoclonal antibodies. The examples of the application also characterize the normal glycosylation pattern of a human lgG1 expressed in CHO-K1 cells. Accordingly, the antibodies of the present invention are inherently glycosylated. In one embodiment, the glycosylation pattern of the antibodies of the present invention is determined by the aforementioned methods known in the art. In one embodiment, the glycosylation pattern of the antibodies of the present invention will be determined relative to the expression system or method of synthesis or production utilised for production, for example, the methods of benchmark glycan profiling disclosed in Luo & Zhang (2023). In one embodiment, the glycosylation pattern of the antibodies of the present invention will be determined relative to the United States Pharmacopeia (USP) developed
monoclonal antibody reference standards, such as those disclosed in Guo et al. 2022. In one embodiment, the glycosylation pattern of the antibodies of the present invention have a standard N-Glycan glycosylation pattern of a human I gG 1 , except for the reduction in fucosylation. In a preferred embodiments, the antibody comprises a light chain (LC) and IgG 1 heavy chain (HC) as herein described and is glycosylated with at the least 2 kDa of total glycans. In a preferred embodiment, the antibody is glycosylated with at the most 3 kDa of total glycans. In some embodiments, the antibody is glycosylated with at the least 2kDa of total glycans and at the most 3 kDa of total glycans.
In some embodiments, the heavy chain constant region of the antibody is glycosylated with at the least 2 kDa of total glycans. In some embodiments, the heavy chain constant region of the antibody is glycosylated with at the most 3 kDa of total glycans. In some embodiments, the heavy chain constant region of the antibody the heavy chain constant region is glycosylated with at the least 2kDa of total glycans and at the most 3 kDa of total glycans.
In various embodiments of the invention, the provided antibody is engineered to contain a high proportion of glycan GO, specifically within the range of 50% to 80% of total glycans, while achieving reduced levels of core fucosylation. In some embodiments, the antibody glycans comprise between 50% and 80% of glycan GO. In some embodiments, the antibody glycans comprise between 50% and 80% of glycan GO, such as between 55 and 80%, such as between 60% and 80%, such as between 65% and 80%, such as between 70% and 80%, such as between 75% and 80%, such as between 50% and 75%, such as between 50% and 70%, such as between 50% and 65%, such as between 50% and 60%, such as between 50% and 55%.
In some embodiments, total glycans of the antibody comprise between 50% and 80% of glycan GO. In some embodiments, total glycans of the antibody comprise between 50% and 80% of glycan GO, such as between 55 and 80%, such as between 60% and 80%, such as between 65% and 80%, such as between 70% and 80%, such as between 75% and 80%, such as between 50% and 75%, such as between 50% and 70%, such as between 50% and 65%, such as between 50% and 60%, such as between 50% and 55%. Control over GO levels, combined with reduced core
fucosylation, allows for enhanced functional properties, including improved ADCC activity.
Fucosylation
As used herein, "core fucosylation" refers to addition of fucose ("fucosylation") to N- acetylglucosamine ("GIcNAc") at the reducing terminal of an N-linked glycan. In the context of IgG 1 antibodies, core fucosylation may occur at the N-glycosylation site of asparagine at position 297 (Asn-297) within the Fc region. Reduction of core fucosylation in antibodies has been shown to alter Fc effector functions, in particular Fc-gamma receptor binding and ADCC activity. This means that antibodies lacking core fucose in their Fc-glycan exhibit high ADCC activity at lower concentrations compared to fucosylated counterparts (Yamane-Ohnuki and Satoh, 2009).
In general, reference to "low fucosylation", “decreased fucosylation” or "reduced fucosylation" with respect to a glycoprotein does not refer to a single glycoprotein molecule having fewer fucose residues attached to it. Rather, reference is made to a glycoprotein preparation, and the glycoprotein preparation comprises a population of individual glycoprotein molecules, with members of the population having different glycosylation features. For purposes of illustration and not limitation, for an I gG 1 antibody expressed in a modified CHO cell according to the invention, "low fucosylation", “decreased fucosylation” or "reduced fucosylation" refers to a smaller number of individual glycoproteins having a fucose residue on an N-linked GIcNAc residue. Such "low fucosylation" or "reduced fucosylation" can be characterized in a variety of ways (see elsewhere herein), but reference is in each case to a relatively low (or reduced) number of the glycoproteins of the population having fucose residues on them as compared to a population of the same glycoprotein made in a cell line that lacks a modification, or to a population of the same glycoprotein made without exposure to inhibitors and/or analogs which reduce core fucosylation, or to a population of the same glycoprotein made without exposure to culturing conditions/additives which reduce fucosylation, in accordance with the invention.
Reference to “relative fucosylation” denotes the number of fucosylated glycans compared to the total number of glycans contained on the antibody molecule or antibody derivative.
In one aspect, the present invention provides for an anti-OX40L antibody, comprising a variable heavy chain, wherein core fucosylation of the heavy chain is below 80%. In one embodiment, the anti-OX40L antibody is fucosylated only about at most 50%, such as at most 40%, such as at most 30%, such as at most 20%, such as at most 20%, such as at most 10% of the amount of fucosylation of the same glycoprotein made in a cell that does not contain the modification.
A glycoprotein preparation according to the invention is fucosylated only about at most 50%, such as at most 40%, such as at most 30%, such as at most 20%, such as at most 20%, such as at most 10% of the amount of fucosylation of the same glycoprotein made in a cell line that lacks a modification, or of the same glycoprotein made without exposure to inhibitors and/or analogs which reduce core fucosylation, or of the same glycoprotein made without exposure to culturing conditions/additives which reduce fucosylation.
A glycoprotein preparation made in a cell according to the invention is fucosylated only about at most 50%, such as at most 40%, such as at most 30%, such as at most 20%, such as at most 20%, such as at most 10% of the amount of fucosylation of the same glycoprotein made in a cell line that lacks a modification, or of the same glycoprotein made without exposure to inhibitors and/or analogs which reduce core fucosylation, or of the same glycoprotein made without exposure to culturing conditions/additives which reduce fucosylation.
Another way to characterize a glycoprotein with decreased fucosylation is by the ratio of fucosylated to non-fucosylated glycans in the glycoprotein preparation made by the cell, or the glycoprotein preparation which has not been exposed to inhibitors and/or analogs which reduce core fucosylation, or the glycoprotein preparation which has not been exposed to culturing conditions/additives which reduce fucosylation. For example, a glycoprotein preparation has a ratio of fucosylated glycans: nonfucosylated glycans that is about 1 :10 through 1 :15, 1 :15 through 1 :20, 1 :20 through 1 :40, 1 :40 through 1 :60, 1 :60 through 1 :80, 1 :80 through 1 :100, or 1 :100 through 1 :150.
Another way to characterize a glycoprotein with decreased fucosylation is by the relative weight percent of nonfucosylated glycoprotein (as compared with total, i.e., fucosylated and nonfucosylated, glycoprotein). For example, a glycoprotein preparation
made by a modified cell has a percent of non-fucosylated glycans that is at most 70%, such as at most 60%, such as at most 50%, such as at most 40%, such as at most 30% as compared with the same glycoprotein preparation made from a cell that lacks the modification. For example, a glycoprotein preparation made via exposure to cell culture conditions and/or additives that decrease fucosylation of the glycoprotein has a percent of non-fucosylated glycans that is at most 70%, such as at most 60%, such as at most 50%, such as at most 40%, such as at most 30% as compared with the same glycoprotein preparation which has not been exposed to cell culture conditions and/or additives which decrease fucosylation of the glycoprotein. Thus, in some embodiments of this invention, the glycans with core fucosylation are below 70%, such as below 60%, for example below 50%, such as below 40%, for example below 30%, such as below 20% of the total glycans.
In some embodiments, antibodies of the instant methods comprise at least 10%, at least 20%, at least 30%, at least 40%, or at least 50% non-core fucosylated glycans (e.g., lacking core fucosylation), as compared with control antibodies. In some embodiments, antibodies or antibody derivatives produced by the instant methods comprise at least 60%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, or at least 95% non-core fucosylated antibody, as compared with a control antibody. In some embodiments, a composition of antibodies or antibody derivatives produced by the instant methods comprises less than 100% non-core fucosylated antibodies and/or antibody derivatives.
Another way to characterize a glycoprotein with decreased fucosylation is by the relative amount of fucose to glycan or the relative amount of fucose to glycan component of the glycoprotein preparation. In one embodiment, the molar ratio of fucose to glycan moiety is no more than about 1:10, 1 :20, 1 :25, 1:33, 1 :50, 1 :100, or 1 :200.
In one embodiment, the amount of antibody protein made that is fucosylated is measured by deglycosylation of antibody protein with PNGase F followed by oligosaccharide analysis via HPLC wherein fucosyl-containing oligosaccharides are quantified by integration of glycan peak area, and, e.g., protein fucosylation is calculated based on glycan peak area. The identity (and composition) of the glycan can be determined (and/or quantified) by any suitable method, such as mass spectroscopy.
Thus, in some embodiments of this invention, the core fucosylation is determined using capillary electrophoresis or HLPC, such as hydrophilic interaction liquid chromatography HPLC (HILIC-HPLC), electrospray ionization mass spectrometry (ESIMS), hydrophilic interaction liquid chromatography equipped with fluorescence detection and tandem mass spectrometry (HILIC-FLD-MS/MS).
Methods for determining core fucosylation include hydrazinolysis or enzyme digestion (see, e.g., Biochemical Experimentation Methods 23: Method for Studying Glycoprotein Sugar Chain (Japan Scientific Societies Press), edited by Reiko Takahashi (1989)), fluorescence labelling or radioisotope labelling of the released sugar chain and then separating the labelled sugar chain by chromatography. Also, the compositions of the released sugar chains can be determined by analysing the chains by the HPAEC-PAD method (see, e.g., J Liq Chromatogr. 6:1557 (1983)). (See generally US 2004- 0110282.)
In some embodiments of the invention, the core fucosylation is determined as (Area with fucosylated glycan)/(total glycan area). In some embodiments of the invention, the core fucosylation is determined as (concentration of fucosylated glycan)/(concentration of total glycan). The fucosylation percentage is calculated taking into consideration peak areas of all the fucosylated species within the total area of glycan. The following equation (1) was used to calculate the fucosylation percentage:
(1) Fucosylation percentage: Area with fucosylated glycan/total glycan area
In an antibody with 100% core fucosylation, all glycan species have core fucosylation. In an antibody with 50% core fucosylation, every second glycan species is not core fucosylated, and every second glycan species is fucosylated. Defucosylation only causes a minor change in the molecular weight of the complete antibody because only the incorporation of fucose moieties is reduced/prevented while all other saccharides are essentially unaffected.
In some embodiments of the invention, the core fucosylation is determined as (amount of fucosylated glycan)/(amount of total glycan).
The results of the determination of fucosylation are dependent on the interpretation of results, such as of chromatographs. Interpretation of results may lead to variability in degrees of fucosylation, such as +/- 5% variability. This can vary depending on the method utilised, all methods of determining fucosylation disclosed herein include an error of between 2-5%, such as between 2-4%.
In some embodiments of the invention, the antibody is covalently linked to a ligand selected from the group consisting of chromophores, fluorophores, radiotracers, drugs, peptides, proteins, enzymes, single or double chained oligonucleotides and their analogs, biotin and, therapeutic moieties, such as cytotoxins, chemotherapeutic drugs, cytokines, and radioisotopes.
In some embodiments of the invention, the antibody is humanized. In some embodiments of the invention, the antibody is of the human subclass lgG1.
Method of producing anti-OX40L-Antibodies with reduced fucosylation
Methods for reducing core fucosylation include chemoenzymatic remodelling, media supplementation, and engineered cell lines (see Li et al. 2017).
Inhibitors
Small molecule inhibitors that act on enzymes in the glycosylation pathway can be utilised to reduce core fucosylation. Inhibitors such as castanospermine, deoxymannojirimycin, australine, act early in the glycosylation pathway via inhibiting glucosidases I and II, producing antibodies with immature glycans (e.g., high levels of mannose) and low fucosylation levels.
One aspect of the present invention is to provide a method of producing an anti-OX40L antibody or antibody derivative, comprising expressing polynucleotides encoding polypeptides of SEQ ID NO: 13 and 14 in a cell, wherein the culture medium comprises an inhibitor of glucosidases I and II, and subsequently purifying the antibody. In some embodiments of the present invention, the cell is a mammalian cell. In some embodiments of the present invention, the inhibitor is selected from the group comprising: castanospermine, deoxynojrimycin, and australine. In another embodiment of the invention, the inhibitor is a-L-fucosidase.
Analogs
Fucose analogs reduce the incorporation of fucose into complex N-glycoside-linked sugar chains of antibodies or antibody derivatives produced by host cells. Suitable fucose analogs are those that can be added to the host cell culture media and inhibit core fucosylation of complex N-glycoside-linked sugar chains of antibodies or antibody derivatives. The fucose analog is typically taken up by host cells (e.g., by active transport or passive diffusion) (see Almahayni et al. 2022).
One aspect of the present invention is to provide an anti-OX40L antibody or antibody derivative, comprising expressing polynucleotides encoding polypeptides of SEQ ID NO: 13 and 14 in a cell, wherein the culture medium comprises a fucose analog, and subsequently purifying the antibody. In some embodiments of the present invention, the cell is a mammalian cell.
In some embodiments, the fucose analog is an inhibitor of fucokinase, GDP-fucose- pyrophosphorylase, fucosyltransferase (FUT), GDP-mannose 4,6-dehydratase, GDP- fucose synthetase, and/or a fucose transporter, such as GDP-fucose transporter. In some embodiments, a fucose analog (or an intracellular metabolite or product of the fucose analog) inhibits an enzyme(s) in the fucose salvage pathway. (As used herein, an intracellular metabolite can be, for example, a GDP-modified analog a fully or partially de-esterified analog. For example, a fucose analog (or an intracellular metabolite or product of the fucose analog) can inhibit the activity of fucokinase or GDP-fucose-pyrophosphorylase. In some embodiments, a fucose analog (or an intracellular metabolite or product of the fucose analog) inhibits fucosyltransferase (preferably a 1,6-fucosyltransferase, e.g., the FLIT8 protein). In some embodiments, the fucose analog is an inhibitor of a fucosyltransferase (FUT), such as 1 ,6- fucosyltransferase (FUT8). In some embodiments, a fucose analog (or an intracellular metabolite or product of the fucose analog) can inhibit the activity of an enzyme in the de novo synthetic pathway for fucose. For example, a fucose analog (or an intracellular metabolite or product of the fucose analog) can inhibit the activity of GDP-mannose 4,6-dehydratase and/or GDP-fucose synthetase. In some embodiments, the fucose analog (or an intracellular metabolite or product of the fucose analog) can inhibit a fucose transporter (e.g., GDP-fucose transporter).
In some embodiments, the fucose analog is selected from the group comprising L- fucose, 2-fluro-fucose, 2- fluoro peracetylated fucose (2FF), 5-alkynyl-fucose, alkynyl fucose monoacetate, alkynyl fucose triacetate, alkynyl fucose diacetate and 5-alkynyl- fucose peracetate. In some embodiments, the fucose analog is 2-fluoro peracetylated fucose (2FF). In some embodiments, the fucose analog is alkynyl fucose peracetate. In some embodiments, the fucose analog is alkynyl fucose triacetate. In some embodiments, the fucose analog is alkynyl fucose diacetate. In some embodiments, the fucose analog is a mixture of alkynyl fucose peracetate, alkynyl fucose triacetate, and alkynyl fucose diacetate. In some embodiments, the fucose analog is a mixture of alkynyl fucose peracetate, alkynyl fucose triacetate, alkynyl fucose diacetate, and alkynyl fucose monoacetate. In any of the various embodiments, the fucose analog is not fucose. In some embodiments, the fucose analog is not alkynyl fucose peracetate. In some embodiments, the fucose analog is not galactose or L-galactose.
In some embodiments of this invention, the fucosyltransferase (FUT) inhibitors are carbohydrate mimics. In some embodiments of this invention, the carbohydrate mimics are selected from the group consisting of O- and C-glycosides, carbacycles, and nitrogen-containing heterocycles such as piperidines and pyrrolidines. The person skilled in the art will appreciate, that a variety of carbohydrates and structural analogs bearing a GDP unit in their structure can have inhibitory properties against fucosyltransferase.
In a preferred embodiment, the fucosyltransferase inhibitor is 2-fluoro peracetylated fucose (2FF). 2FF is a fluorinated analog of fucose that can be taken up and metabolized to the desired donor substrate-based inhibitor of fucosyltransferases inside the cell. It also acts to prevent the de novo synthesis of the natural substrates, resulting in a global, family-wide shutdown of fucosyltransferases and remodelling of cell surface glycans.
The amount of the fucose analog that is effective can be determined by standard cell culture methodologies. For example, cell culture assays may be employed to help identify optimal dosing ranges. The precise amount to be employed also depends on the time of administration, the host cell line, the cell density, and the like. Effective doses may be extrapolated from dose-response curves derived from in vitro model test
systems. Accordingly, in one embodiment, the culture medium is supplemented with the fucose analog to maintain an effective concentration thereof.
In some embodiments, the fucose analog is present in the culture medium at a concentration of 10 nM to 50 mM. In some embodiments, the fucose analog is present in the culture medium at a concentration of 10 nM to 10 mM. In some embodiments, the fucose analog is present in the culture medium at a concentration of 100 nM to 5 mM. In some embodiments, the fucose analog is present in the culture medium at a concentration of 100 nM to 3 mM. In some embodiments, the fucose analog is present in the culture medium at a concentration of 100 nM to 2 mM. In some embodiments, the fucose analog is present in the culture medium at a concentration of 100 pM to 1 mM. In some embodiments, the fucose analog is present in the culture medium at a concentration of 1 pM to 1 mM. In some embodiments, the fucose analog is present in the culture medium at a concentration of 10 pM to 1 mM. In some embodiments, the fucose analog is present in the culture medium at a concentration of 10 nM to 500 pM. In some embodiments, the fucose analog is present in the culture medium at a concentration of 1 pM to 500 pM. In some embodiments, the fucose analog is present in the culture medium at a concentration of 1 pM to 250 pM. In some embodiments, the fucose analog is present in the culture medium at a concentration of 10 pM to 100 pM. In a preferred embodiment, the fucose analog is present in the culture medium at a concentration of at least 150 pM, such as at least 200 pM, such as at least 250 pM. In some embodiments, the fucose analog is soluble in the culture medium (at the appropriate temperature for host cell maintenance/growth) at a concentration of at least 10 nM. In some embodiments, the fucose analog is soluble in the culture medium (at the appropriate temperature for host cell maintenance/growth) at a concentration of at least 100 nM.
Cell engineering
Cell engineering has been successful in producing modified glycans by insertion or deletion of key genes. Antibodies and derivatives thereof that are useful in the present methods can be produced by recombinant expression techniques, from hybridomas, from myelomas, or from other antibody-expressing cells. Recombinant expression of an antibody or derivative thereof that binds to a target antigen typically involves construction of an expression vector containing a nucleic acid that encodes the antibody or derivative thereof. Once a nucleic acid encoding such a protein has been
obtained, the vector for the production of the protein molecule may be produced by recombinant DNA technology using techniques well-known in the art. Standard techniques such as those described in Sambrook and Russell, Molecular Cloning: A Laboratory Manual (Cold Spring Harbor Laboratory Press, Cold Spring Harbor, N.Y., 3rd ed., 2001) can be used for recombinant nucleic acid methods, nucleic acid synthesis, cell culture, transgene incorporation, and recombinant protein expression.
Methods for engineering cell lines which are unable to fucosylate proteins to reduce core fucosylation include gene knockouts, gene knock-ins, and RNA interference (RNAi), said methods are known and well referenced in Edwards et al. (2022). In gene knock-outs, the gene encoding FUT8 (alpha 1,6- fucosyltransferase enzyme) is inactivated. FUT8 catalyses the transfer of a fucosyl residue from GDP-fucose to position 6 of Asn-linked (N-linked) GIcNac of an N- glycan. FUT8 is reported to be the only enzyme responsible for adding fucose to the N-linked biantennary carbohydrate at Asn297. For example, deletion of the fucosyl transferase gene (FUT8) in Chinese hamster ovary (CHO) cells for producing completely defucosylated antibodies with enhanced antibody-dependent cellular cytotoxicity (Yamane-Ohnuki et al., 2004). Additionally, lectin-resistant CHO glycosylation mutants selected for resistance to certain lectins. Such cell lines are isolated by repeated selection for the inability to bind a particular lectin, in the presence of a mutagen. Other cell lines reportedly incapable of fucosylating proteins, e.g., antibodies, are known, see, e.g., US Patent No. 7,425,466 and US Patent No. 7,214,775 (a1,6-fucosyltransferase, i.e., FUT8 mutant).
RNAi typically also targets FUT8 gene expression, leading to decreased mRNA transcript levels or knock out gene expression entirely. Another approach has been to disrupt the gene that encodes the enzyme GDP-mannose 4,6-dehydratase (GMD). GMD is involved in catalysing the conversion of d-glucose to GDP-fucose and is therefore crucial for the activity of o1,6-FucT in carrying out fucosylation downstream in this pathway (Kanda et al. 2007).
Gene knock-ins add genes encoding enzymes such as p-1 ,4-N- acetylglucosaminyltransferase (GNTIII), GDP-6-deoxy-D-lyxo-4-hexulose reductase (RMD), or a golgi alpha-mannosidase II. An increase in the levels of such enzymes in cells diverts monoclonal antibodies from the fucosylation pathway (leading to
decreased core fucosylation) and having increased amount of bisecting N- acetylglucosamines.
Another aspect of the present invention is to provide a method of producing an anti- OX40L antibody, comprising expressing polynucleotides encoding polypeptides of SEQ ID NO: 13 and 14 in a cell that is genetically modified to prevent core fucosylation, such as in a mammalian cell. Cell lines that are unable to fucosylate proteins are known in the art.
In some embodiments of this invention, the cell does not encode a functional fucosyltransferase. The person skilled in the art will appreciate that fucosylation- deficient cell lines can be generated in several ways. Ways to generate fucosylation- deficient cell lines include but are not limited to knockouts, such as homologous recombination, CRISPR-Cas9 and TALENs, and knockdowns, such as through antisense oligonucleotides, ribozymes and RNA interference.
In some embodiments, the cell encodes a functional p-1 ,4-N- acetylglucosaminyltransferase (GnTIII), GDP-6-deoxy-D-lyxo-4-hexulose reductase (RMD) and/or a Golgi a-mannosidase II (Manll).
In some embodiments, the cell is a mammalian cell. In some embodiments, the cell is a recombinant cell. In some embodiments, the cell is selected from the following cells: CHO (e.g., CHO K1 , DXB-11 OHO, Veggie-CHO), COS (e.g., COS-7), Syrian hamster, rat myeloma, mouse myeloma (e.g., SP2/0, NSO), retinal cell, Vero, CV1, kidney (e.g., HEK293, 293 EBNA, MSR 293, MDCK, HaK, BHK, BHK21), HeLa, HepG2, WI38, MRC 5, Colo205, HB 8065, HL-60, Jurkat, Daudi, A431 (epidermal), CV-1, U937, 3T3, L cell, C127 cell, MMT 060562, Sertoli cell, BRL 3A cell, HT1080 cell, a human myeloma cell, tumor cell, a human lymphoma cell (e.g., a Namalwa cell) and a cell line derived from an aforementioned cell.
Monoclonal antibodies directed against the antigen can be obtained from the immunized, transgenic mice using conventional hybridoma technology. In some embodiments, the host cells are from a hybridoma.
Cell-free protein synthesis
Cell-free protein synthesis (CFPS) is the use of synthetic reaction systems comprising biological macromolecules in a reaction mix, such as biological extracts and/or defined reagents. The reaction mix will comprise a template for the production of the macromolecule, e.g. DNA, mRNA, etc.; monomers for the macromolecule to be synthesized, e.g. amino acids, etc., and such co-factors, enzymes, and other reagents that are necessary for the synthesis, e.g. ribosomes, tRNA, polymerases, transcriptional factors, etc., Such synthetic reaction systems are well-known in the art, and have been described in the literature. In some embodiments,
In one embodiment, the antibodies and antibody derivatives are produced, synthesised, or obtained by cell-free protein synthesis methods, such as those disclosed in Hunt et al. 2023, or Stech et al., 2017. In a further embodiment, the method of producing an anti-OX40L antibody or antibody derivative, comprising expressing polynucleotides encoding polypeptides of SEQ ID NO: 13 and 14, is cell- free protein synthesis.
Titration of desired levels of fucosylation
Reduction in fucosylation via cellular engineering/manipulation can be mitigated by the provision of fucose in the cell culture media to facilitate its incorporation into cells, and titration of the desired levels of fucosylation via the fucose salvage pathway. This provides the advantage of increasing the control of this bioprocess to a predetermined value, wherein the strategic use of methods for controlling fucosylation levels can produce a wide range of fucosylation levels in the bioproduction of monoclonal antibodies (Mishra et al. 2020).
Accordingly, in one embodiment of the present invention provides a method of producing an anti-OX40L antibody or antibody derivative, comprising expressing polynucleotides encoding polypeptides of SEQ ID NO: 13 and 14 in a cell that is genetically modified to prevent core fucosylation, wherein the antibody or antibody derivative is restored or titrated to a desired level via exposure to fucose, wherein the cell is exposure to culture medium comprising fucose. In some embodiments, increasing concentrations of fucose are added to the culture media, such as up to 1mM.
In some embodiments, the method comprises an additional step of subsequently isolating the antibody. In some embodiments, the method comprises an additional step of subsequently purifying the antibody. In some embodiments, the method comprises an additional step of optionally folding the antibody. The antibodies of the present disclosure can be purified to homogeneity. The separation and purification of the antibodies can be performed by employing conventional protein separation and purification methods known by the person skilled in the art, for example, hydroxylapatite chromatography, gel electrophoresis, dialysis, and affinity chromatography such as Protein A purification.
In some embodiments, the cell is selected from the group consisting of mammalian cells, bacterial cells, yeast cells, and plant cells.
In the aforementioned methods of the invention, in some embodiments, the antibody is an lgG1. In some embodiments, the antibody is an intact antibody. In some embodiments, the antibody comprises a heavy and a light chain variable region and an Fc region. In some embodiments, the antibody derivative comprises an antibody Fc region and a ligand binding domain of a non-immunoglobulin protein.
Method of testing anti-OX40L antibodies
Blocking of OX40L-OX40 interaction
The present invention provides for compositions and methods to produce antibodies and antibody derivatives which inhibit the binding of 0X40 to OX40L, wherein the antibodies and antibody derivatives described in the present invention bind to human OX40L, thereby inhibiting the OX40/OX40L interaction, and thereby inhibiting the OX40L induced signal transduction. Preferably, the antibodies and antibody derivatives of the present invention inhibit hOX40L/OX40 interaction. In one embodiment, the antibody according to the invention inhibits the interaction of OX40L with 0X40. The quantification of the interaction of OX40L with 0X40 can be measured by techniques known to those of skill in the art, including, for example, an ELISA using immobilized OX40L, such as biotinylated OX40L immobilized on a streptavidine surface.
Improved ADCC effects
Core fucosylation of an Fc N-linked glycan affects antibody effector functions, as the absence of fucose increases the antibody-dependent cell cytotoxicity (ADCC) response with increased binding to the Fc receptors. The cytotoxic activity against an antigenpositive cultured cell line can be evaluated by measuring effector function (e.g., as described in Cancer Immunol. Immunother. 36:373 (1993)).
In some embodiments, the antibodies and antibody derivatives of the instant disclosure have higher effector function (e.g., ADCC activity) than the antibodies with normal levels of fucosylation, i.e., those produced in the absence of a fucose analog or inhibitor, or those produced from wild-type cell lines. The effector function activity may be modulated by altering the concentration of the fucose analog in the culture medium and/or the duration of exposure to the fucose analog. ADCC activity may be measured using assays known in the art and in exemplary embodiments increases ADCC by at least 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, 2-fold, 3-fold, 4-fold, 5-fold, 6- fold, 7-fold, 8-fold, 9-fold, 10-fold, 15-fold or 20-fold, as compared to the core fucosylated parent antibody.
Fc-receptor affinity assay
The antibodies of the invention are Fc engineered such that the affinities for various Fey receptors are increased. In some embodiments, the antibodies and antibody derivatives of the instant disclosure have a higher affinity for Fc receptors, such as CD16A 158V and CD16a 158F, than the antibodies with normal levels of fucosylation, i.e., those produced in the absence of a fucose analog or inhibitor, or those produced from wild-type cell lines.
The affinity for Fc receptors may be modulated by altering the concentration of the fucose analog in the culture medium and/or the duration of exposure to the fucose analog. Affinity for Fc receptors may be measured using assays known in the art and in exemplary embodiments increases affinity for Fc receptors by at least 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, 2-fold, 3-fold, 4-fold, 5-fold, 6-fold, 7-fold, 8-fold, 9- fold, 10-fold, 15-fold or 20-fold, as compared to the core fucosylated parent antibody.
T-cell activation assay
In one aspect, the invention provides for a method of testing a defucosylated anti- OX40L antibody or antibody derivative, comprising testing the antibody in a T-cell
activation assay and verifying that the T-cell activation is reduced compared to a fucosylated anti-OX40L antibody. In some embodiments, the T-cell activation is reduced by at least 5%, such as at least 10%, such as at least 15%, such as at least 25%, such as at least 30%.
Indications
The present invention includes the use of the antibody or antibody derivatives in the prophylaxis and/or treatment of at least one disease or disorder that is associated with OX40L. In one embodiment, the invention relates to a method for the prevention and/or treatment of at least one disease or disorder that can be treated by modulating OX40L, its biological or pharmacological activity, and/or the biological pathways or signalling in which OX40L is involved, said method comprising administering, to a subject in need thereof, a pharmaceutically active amount of the antibody or antibody derivates of the invention and/or of a pharmaceutical composition comprising the same. In particular, said pharmaceutically effective amount may be an amount that is sufficient to modulate OX40L, its biological or pharmacological activity, and/or the biological pathways or signalling in which OX40L is involved.
In some embodiments, the present invention provides for the use of the antibodies or antibody derivatives in the prophylaxis and/or treatment of inflammatory disorders and/or autoimmune disorders. In some embodiments, the antibody or antibody derivative is for use in the prophylaxis and/or treatment and/or prevention of inflammatory disorders and/or autoimmune disorders. In one embodiment, the antibody or antibody derivative is for use in the prophylaxis and/or treatment of inflammatory disorders. In one embodiment, the antibody or antibody derivative is for use in the preparation of a medicament for the prophylaxis and treatment of inflammatory disorders. In one embodiment, the antibody or antibody derivative is for use in the prophylaxis and/or treatment of autoimmune diseases. In one embodiment, the antibody or antibody derivative is for use in the preparation of a medicament for the prophylaxis and treatment of autoimmune diseases. In one embodiment, the disease is selected from the group consisting of Graft-vs-host-disease, allograft rejection, asthma, Systemic lupus, arthritis, inflammatory bowel disease, ulcerative colitis, Crohn’s disease, diabetes, atopic dermatitis, psoriasis, hidradenitis suppurativa, immunoglobulin A nephropathy, Hashimoto disease, Graves’ disease, chronic
rhinosinusitis and multiple sclerosis. In one aspect, the invention provides for a kit comprising the aforementioned antibody or antibody derivative.
In the context of the present invention, the term "prophylaxis and/or treatment" not only comprises preventing and/or treating the disease, but also generally comprises preventing the onset of the disease, slowing or reversing the progress of disease, preventing or slowing the onset of one or more symptoms associated with the disease, reducing and/or alleviating one or more symptoms associated with the disease, reducing the severity and/or the duration of the disease and/or of any symptoms associated therewith and/or preventing a further increase in the severity of the disease and/or of any symptoms associated therewith, preventing, reducing or reversing any physiological damage caused by the disease, and generally any pharmacological action that is beneficial to the patient being treated.
The subject to be treated may be any warm-blooded animal but is in particular a mammal, and more in particular a human being. As will be clear to the skilled person, the subject to be treated will in particular be a person suffering from, or at risk of, the diseases and disorders mentioned herein.
Pharmaceutical compositions
The present invention includes antibodies and antibody derivatives, compositions and methods for use in the prophylaxis and/or treatment of inflammatory disorders and/or autoimmune disorders. Administration of the antibodies and antibody derivatives and/or compositions according to the present invention will typically be via any common route. This includes, but is not limited to parenteral, orthotopic, intradermal, subcutaneous, intramuscular, intraperitoneal, intranasal, or intravenous injection. Additional formulations which are suitable for other modes of administration include oral formulations. Oral formulations include such normally employed excipients as, for example, pharmaceutical grades of mannitol, lactose, starch, magnesium stearate, sodium saccharine, cellulose, magnesium carbonate, and the like. These compositions take the form of solutions, suspensions, tablets, pills, capsules, sustained-release formulations, or powders and contain about 10% to about 95% of active ingredient, preferably about 25% to about 70%.
Typically, compositions of the invention are administered in a manner compatible with the dosage formulation, and in such amount as will be therapeutically effective and immune modifying. The quantity to be administered depends on the subject to be treated. Precise amounts of active ingredient required to be administered depend on the judgment of the practitioner.
The manner of application may be varied widely. Any of the conventional methods for administration of an antibody are applicable. These are believed to include oral application on a solid physiologically acceptable base or in a physiologically acceptable dispersion, parenterally, by injection, and the like. The dosage of the pharmaceutical composition will depend on the route of administration and will vary according to the size and health of the subject. The course of the administrations may be followed by assays for alloreactive immune responses and T-cell activity.
The phrases "pharmaceutically acceptable" or "pharmacologically acceptable" refer to molecular entities and compositions that do not produce an adverse, allergic, or other untoward reaction when administered to an animal, or human. As used herein, "pharmaceutically acceptable excipient " includes any and all solvents, dispersion media, coatings, antibacterial and antifungal agents, isotonic and absorption delaying agents, and the like. The use of such media and agents for pharmaceutically active substances is well-known in the art. Except insofar as any conventional media or agent is incompatible with the active ingredients, its use in immunogenic and therapeutic compositions is contemplated.
The compositions may be formulated into a neutral or salt form. Pharmaceutically acceptable salts include the acid addition salts (formed with the free amino groups of the protein) which are formed with inorganic acids such as, for example, hydrochloric or phosphoric acids, or such organic acids as acetic, oxalic, tartaric, mandelic, and the like. Salts formed with the free carboxyl groups can also be derived from inorganic bases such as, for example, sodium, potassium, ammonium, calcium, or ferric hydroxides, and such organic bases as isopropylamine, trimethylamine, histidine, procaine, and the like.
The carrier can also be a solvent or dispersion medium containing, for example, water, ethanol, polyol (for example, glycerol, propylene glycol, liquid polyethylene glycol, and
the like), suitable mixtures thereof, and vegetable oils. The prevention of the action of microorganisms can be brought about by various antibacterial and antifungal agents, for example, parabens, chlorobutanol, phenol, sorbic acid, thimerosal, and the like.
An effective amount of therapeutic or prophylactic composition is determined based on the intended goal. The term "unit dose" or "dosage" refers to physically discrete units suitable for use in a subject, each unit containing a predetermined quantity of the composition calculated to produce the desired responses discussed above in association with its administration, i.e. , the appropriate route and regimen. The quantity to be administered, both according to number of treatments and unit dose, depends on the result and/or protection desired. Precise amounts of the composition also depend on the judgment of the practitioner and are peculiar to each individual. Factors affecting dose include the physical and clinical state of the subject, route of administration, the intended goal of treatment (alleviation of symptoms versus cure), and potency, stability, and toxicity of the particular composition. Upon formulation, solutions will be administered in a manner compatible with the dosage formulation and in such amount as is therapeutically or prophylactically effective. The formulations are easily administered in a variety of dosage forms.
Examples
Example 1: Affinity maturation
In vitro affinity maturation is based on the principles of mutation and selection. In vitro affinity maturation has successfully been used to optimize antibodies, antibody fragments, or other peptide molecules like antibody mimetics. Random mutations inside the CDRs are introduced using radiation, chemical mutagens, or error-prone PCR. In addition, the genetic diversity can be increased by chain shuffling. Two or three rounds of mutation and selection using display methods like phage display usually result in antibody fragments with affinities in the low nanomolar range.
Aim:
To generate an anti-OX40L antibody with improved characteristics such as potency, binding affinity for OX40L, and antibody stickiness compared to existing anti-OX40L antibodies.
Material and Methods:
Affinity maturation: The affinity maturation was performed in the scFv format. A site- directed CDR mutagenesis was performed to introduce mutations into the parental antibody (Anti-OX40L, Clone R4930) sequence. An antibody-phage library was constructed, which was used for an in vitro selection under stringent conditions. Finally, antibodies were identified, characterized, and converted into the final antibody format. Antibody binding was validated, and the antibodies were further characterized to identify lead candidates.
Antigen preparation: The R4930 gene was cloned into a scFv phage display vector. After transformation of E. Coli, soluble scFv antibodies were expressed as well as antibody-phage particles packaged. Packaging and scFv production was tested by a Titration-ELISA (data not shown).
Generation of a CDR-mutation library: A bioinformatic analysis of R4930 was performed to generate a site directed CDR-mutation library. After homology modelling of the antibody Fv regions and CDR grafting onto the template, CDR residues possibly involved into antigen binding were identified. For the heavy chain 18 positions and for the light chain 12 positions were identified. By analysing a NGS database commonly used amino acids for the specific germline were identified. Based on this, degenerated codons were designed, introducing mutations at the identified position possibly involved into antigen binding. Amino acids with unfavourable characteristics were avoided. The introduction of mutations can be described by a gaussian distribution with an average of four mutations for each antibody chain. Primers were designed based on the degenerated codons and used for introduction of mutations into the antibody sequence. The mutated antibody genes were cloned into a scFv phage display vector and three libraries were generated and packaged into antibody-phage particles. A library with a total functional diversity of ~7x108CFUs was generated. Antibody clones with a functional open reading frame were determined by DNA Sanger sequence analysis. Packaging and purification of antibody-phage particles resulted in more than 3x1012 CFU/mL for each library.
Affinity maturation by in vitro selection: The generated antibody-phage library was used for the affinity maturation by in vitro selection. The same overall excess of antibodyphage particles to functional size was used for each individual library. The specific amount was pooled into one library for in vitro selection. Two rounds of in vitro
selection were performed with increasing stringency by antigen limitation and competition in panning round two. In both panning rounds a negative selection against several negative antigens were performed. Four different strategies were used for affinity maturation by in vitro selection. In panning round two the amount of antigen was reduced from strategy one to four by factor ten between each strategy. Eluted antibody-phage after panning round 1 were amplified and used for the second panning round, after panning round 2 eluted antibody-phage were used for antibody screening.
Antibody screening: Eluted antibody-phage particles after panning round two were used for infection of E. Coli. 384 clones from each strategy were selected randomly for antibody screening. In total, 1536 antibody clones were used for production of monoclonal scFv antibodies in the bacterial system. The produced antibody clones were tested for binding activity on the OX40L immobilized directly to the ELISA plate. Two negative antigens (Streptavidin and BSA) were immobilized directly to the ELISA plate. Soluble scFv antibodies were detected via the myc-tag using an HRP coupled secondary antibody. Antibody clones with the defined parameters as listed below were identified as an antigen specific clone:
• ELISA binding signal to the positive antigen > 0.2
• ELISA binding signal to the negative antigen < 0.2
• S/N ratio between positive and negative antigen > 3
Around one third of all tested antibodies were identified as antigen specific clones. The parental scFv antibody were used for positive control. Several antibodies showed higher binding signals than the parental scFv antibody (data not shown). In parallel a negative control scFv (Trastuzumab) was produced and tested, this antibody didn't show any specific binding.
Antibody sequencing: All 444 antigen specific antibodies were selected for DNA sanger sequence analysis. Sequence analysis revealed 271 uniquely mutated antibodies. These antibodies showed between one and seven mutations. Several hotspot mutations were identified indicating preferable mutations at different positions.
Affinity ranking: To narrow down the number of antibodies for antibody conversion into the final format, an affinity ranking using the BLI octet system was performed. First the assay was set up using the parental scFv antibody produced in the bacterial system and the biotinylated antigen. The antigen was immobilized to a streptavidin sensor,
which was incubated in the bacterial scFv supernatant for association. The dissociation was measured by incubation in buffer, after modelling the association and dissociation profile, the dissociation rate was calculated. 271 clones were picked and used for scFv production in the bacterial system. The assay as described above was performed for each antibody. The antibodies off-rate was calculated, and the antibodies were ranked accordingly. From all measured antibodies 267 could be measured and fitted with high confidence, among these 267 clones, 55 clones showed an increased off-rate compared to the parental antibody. Within these 55 antibodies, 13 showed at least two times increased off-rate compared to the parental antibody.
Conversion into final format: 21 antibodies as well as the parental antibody sequence were cloned into a mammalian expression vector, containing either the constant domains of the heavy chain or light chain respectively. Transfection grade plasmid DNA was generated and verified by DNA sequence analysis. The plasmid DNA was used for transient transfection of HEK cells. After one week of antibody production in mammalian cell culture, the antibodies were purified using protein A affinity chromatography. The final buffer of the purified antibodies is phosphate buffered saline (no additive included). A quality control was performed by LIV/VIS spectrometry at 280 nM. The antibodies were tested in ELISA for antibody binding to the positive antigen human OX40L captured by streptavidin. For control of unspecific binding streptavidin was used. A titration series (1/square(10)) of each antibody starting at 10 pg/ml was performed and incubated on the coated and blocked ELISA plates. For detection a HRP coupled secondary antibody was used (Anti-Human IgG (Fc specific) - Peroxidase antibody; Sigma Aldrich #A0170). All antibodies showed potent and specific binding to the target antigen.
Antibody affinity: For further characterization of the antibodies an affinity determination using the BLI octet system was set up. The human IgG antibody was immobilized to protein A sensor tips and a titration of the human OX40L antigen was performed. The association and dissociation profile were modelled using either 50 nM or 15.8 nM concentrations, and the antibody affinity determined. The affinity of the parental antibody (R4930) and selected anti-OX40L antibody candidate (B02) was determined (see table 1).
Allogeneic mixed lymphocyte reaction: Peripheral blood mononuclear cells (PBMC) were isolated via Ficoll Paque Plus (Cytiva) density gradient centrifugation from buffy coats (500g x 30 min). Buffy coats were obtained from anonymous healthy adult donors from the Akademiska University Hospital blood bank, and PBMC isolation was performed within 24 hours of blood collection. In line with Swedish legislation, an ethics approval was not needed since the blood bank provides buffy coats for research purposes from anonymous donors and biological material cannot be traced back to a specific individual. Isolated PBMC were mixed in PBS at a concentration of 2.0 x 107 cells per mL and stained with CFSE (Thermo Fisher Scientific Inc) according to the manufacturer’s instructions. PBMC from 2 different donors were mixed in a two-way allogeneic mixed lymphocyte reaction, i.e., PBMCs from neither donor were inactivated. VPD450-stained PBMC were washed and resuspended in 10% heat- inactivated Ultralow IgG FBS (Gibco) in AIM V medium (Gibco, Thermo Fisher Scientific Inc). Resuspended PBMCs were dispensed into round-bottom 96-well cell culture plates, and pure medium or medium supplemented with test article was added to a final concentration of 2 x 106 cells per mL in a final volume of 200 pL. Mixed lymphocyte reactions were incubated at 37°C, 5% CO2 for 7 days. Supernatant postculture was kept and analysed for cytokine content (measuring levels of I N Fy, IL-6, TNFa) utilizing Legendplex (BioLegend) cytokine assay according to the manufacturers’ instructions. B02 or R4930 were added at the start of the culture at a concentration of 10 pg/mL.
Flow cytometry: After 7 days of mixed lymphocyte reaction, samples were washed and then stained with antibodies against cell surface antigens. For intracellular staining FoxP3 staining kit (BD Biosciences) was used according to the manufacturer’s instructions. Samples were stained in the dark at 4°C and washed twice in staining buffer (BD Biosciences), followed by analysis using a BC Cytoflex flow cytometer (Beckman Coulter). T cells were defined as CD3+ cells. The degree of proliferation calculated as a proliferation index (FlowJo software). Tregs were defined as CD3+, CD4+, CD25hi and FoxP3+. Naive T cells were defined as CD3+, CD45RA+, CCR7+. Memory T cells were defined as CD3+, CD45RA-. The amount of naive cells was expressed as a ratio memory cells/naive cells (M/N).
Results:
Statistical significance defined as *p<0.05.
The affinity of B02 to OX40L was increased compared with R4930, determined by the BLI octet system, as demonstrated by the decreased Kd value of B02; 1.9 nM, compared to R4930; 2.9 nM.
Table 2. Proliferation, M/N, cytokine, activation and T-Regs of 0X118 and R4930.
Statistical significance defined as *p<0.05.
B02 significantly decreased cytokine abundance compared to R4930, in addition T- Regs were more abundant. Further, R4930 displayed a favourable proliferation index, ratio between memory and naive cells (M/N) (Table 2).
Conclusion:
The inventors of the present disclosure have shown that B02 has an improved antibody affinity to OX40L, decreased cytokine production, activation and an increased T-Reg abundance and proliferation compared with prior art anti-OX40L antibody, R4930. These data demonstrate an independent effect of B02 on T-cell activation.
Example 2: Optimisation of Fc region of anti-OX40L antibody, 0X118
Core fucosylation of an Fc N-linked glycan affects IgG antibody effector functions, as the absence of fucose increases the antibody dependent cell cytotoxicity (ADCC) response with increased binding to the Fey receptors.
One method utilised to reduce core fucosylation is to utilise a fucosyltransferase (FUT) inhibitor, 2- fluoro peracetylated fucose (2FF). 2FF is a competitive inhibitor of the
fucosyltransferase enzyme (FLIT8) required in transferring fucose to the developing glycan in the Golgi (Rillahan et al., 2012).
Aim:
To optimise the Fc-region of B02 via reducing core fucosylation and obtain a novel anti- OX40L antibody (0X118) with an enhanced ADCC response, and in turn reduced T- cell activation comparative to prior art antibodies, thus has improved clinical efficacy. i) Reduction of fucosylation through treatment with 2-flurofucose (2-FF) First, the effect of 2-FF on the reduction of fucosylation levels was assessed.
Materials and Methods:
Determination of N-glycan composition was achieved via sample denaturation followed by enzymatic release via peptide N-glycosidase F (PNGase F, waters). The resulting N-glycans are fluorescently labeled with RapiFlour-MS (waters) for detection. The fluorescently labeled N-glycans are subjected to HILIC-FLD-MS, TripleTOF 4600 MS (AB SCIEX), HPLC system Nexera X2 (SHIMADZU), running a HILIC column (Waters). The N-glycans are then identified using the mass-to-charge ratio (m/z) detected by the mass spectrometry and the relative abundance of each N-glycan can be calculated from the peak area of the HILIC-FLD chromatogram. PeakView software was used to analyze the data.
N-glycans may also be determined using the following method: The sample is first denatured by Rapigest-SF (Waters) and its N-glycans are then released by PNGase F (Waters or equivalent). The resulting N-glycans are further labeled with fluorescence tag RapiFlour-MS (Waters) for detection purpose and purified by solid phase extraction (SPE, BEH Amide, Waters). Such fluorescently labeled N-glycans are subjected to HILIC-FLD detection (e.g., 1290, Agilent). The N-glycans can be assigned by their retention time, and the percentage of each N-glycan can be calculated from the peak area of HILIC-FLD chromatogram.
Results:
Baseline glycosylation profile of two clones of human I gG 1 B02 expressed in CHO K1 cells is presented in Table 3 and Table 4. Relative fucosylation (%) decreased following treatment with increasing concentrations of 2-FF up to a concentration of 400 pM. Concentrations of 2-FF greater than 200 pM results in a plateau of around 30%
fucosylation (Figure 1). Other parts of the glycosylation profile were ordinary for human lgG1 expressed in CHO K1 , man5 1.6-2.2% and did not vary in a dose dependent manner with 2-FF concentration. The glycosylation profile for the I gG 1 not exposed to 2-FF was ordinary for human lgG1 expressed in CHO K1, as demonstrated in Table 3. The glycosylation profile for the I gG 1 with reduced fucosylation to a degree of 30% was ordinary for human lgG1 expressed in CHO K1, as demonstrated in Table 3.
cn
Table 3. N-Glycan test results. Baseline glycosylation profile of human IgG 1 B02 expressed in CHO K1 cells (clone
%; abundance of each N-glycan. Calculated with the following formula: (fucosylation)% = 100% - (G0)% - (G0-GN)%.
Calculated with the following formula: fucosylation)% = (G0F-GN)% + (G0F)% + (G1 Fa)% + (G1 Fb)% + (G2F)%.
fucosylation dropped significantly to 12.98%, while saturation of the fucosylation reduction effect was reached at approximately 150-200 pM, where fucosylation levels stabilised around 3.20%-2.83%. Increasing 2-FF to 400 pM resulted in a minimal further reduction to 2.29% fucosylation, indicating the effect plateaued at this concentration. Other components of the glycosylation profile, such as Man5 and nonattributed peaks, remained consistent across increasing concentrations of 2-FF. The glycosylation profile with reduced fucosylation reflects an increase in G0%, with GO increasing from 3.54% at baseline to 75.62% at 400 pM 2-FF, demonstrating the anticipated shift in glycan structure as fucosylation is reduced. Similar results are shown in Table 3 for a different clone with a higher degree of fucosylation.
Table 4. N-Glycan test results. Baseline glycosylation profile of human I gG 1 (clone 8) expressed in CHO K1 cells and influence of 2-FF on fucosylation pattern.
bCalculated with the following formula: fucosylation) % = (G0F-GN)% + (G0F)% + (G1 Fa)% + (G1 Fb)% + (G2F)%.
Conclusion:
Saturation of the effect of 2-FF on the reduction of fucosylation is reached at a concentration of 200 pM 2-FF, wherein around 30% fucosylation (clone 125) and around 3% (clone 8) is achieved. ii) Fc-receptor affinity assay
Aim:
To investigate the relationship between levels of core fucosylation of 0X118 and affinity of 0X118 towards different Fc-receptors.
Material and Methods:
BLI experiments were performed on an Octet RED96e (ForteBio) instrument. Recombinant proteins used for testing were sourced from sino biologicals (FcRn, CD16a 158V, CD16a158F, CD32a 131Arg, CD32a 131 His), apart from C1q which was sourced from Sigma. The HIS1 K Sensor was used to capture his-tagged recombinant proteins such as FcRn, CD16a 158V, CD16a158F, CD32a 131Arg, CD32a 131 His and the FAB2G Sensor was used to capture Fab region of antibodies in C1q affinity evaluation. For CD32a, CD16a and C1q 1% BSA in 0.02% PBST was used as a buffer and the temperature was set to 25°C. The Shake speed was set to 1000 rpm. For FcRn binding buffer pH was set to pH 6. Association and dissociation time was 300 seconds for C1q. For FcRn Association and dissociation time was 60 seconds. For CD32a and CD16a association time was 60 seconds and dissociation time was 150 seconds. Herceptin was used as an internal reference antibody. The data acquisition rate is set to Standard kinetics (5.0Hz, averaging by 20). Data analysis was performed using Octet® System Data Analysis (Release 10.0). The binding of CD16a, CD32a and C1q to the Fc region of antibody is a weak affinity interaction (fast on and fast off) and is not suitable for complete kinetic analysis. The binding curves for each concentration of analyte have reached steady state and can be evaluated for affinity using the steady-state analytical SSG KD values. The CD64 and FcRn affinity were evaluated using the kinetics analytical KD values.
Results:
The affinity of 0X118 for CD16A, 158V and CD16a 158F increased with decreased fucosylation (%). Whereas the affinity of 0X118 for CD32a, 01 q and FcRn did not change with increased fucosylation (Figure 2). A saturation of the effect of reduced fucosylation was observed at -50% fucosylation, below which no further improvement in Fc receptor affinity was achieved.
Conclusion:
Reduced degree of fucosylation of 0X118 resulted in increased affinity for CD16a without influence on FcRn, C1q or CD32a binding. Saturation of this effect was achieved at -50% fucosylation of 0X118. iii) Reduced fucosylation and T-cell activation
Aim:
To investigate the relationship between levels of core fucosylation of 0X118 and capacity of 0X118 to induce T-cell activation.
Material and Methods:
Two variants of 0X118 with full (96%) and reduced (34%) levels of core fucosylation were produced via treatment with the fucosyltransferase inhibitor; 2- fluoro peracetylated fucose (2FF).
Results:
Addition of both 0X118 variants induced a concentration-dependent decrease in T-cell activation compared with controls (Figure 3). A reduction in levels of core fucosylation in 0X118 (34%) resulted in an enhanced concentration-dependent reduction in T-cell activation, wherein increasing doses results in decreased levels of T-cell activation, compared with the fully fucosylated 0X118 (96%) variant (Figure 3). These data reached significance at 103 and 104 ng/mL of 0X118 variants.
Conclusion:
0X118 induces a concentration-dependent reduction of T cell activation both when fully fucosylated (97%) and when fucosylation is reduced (34%). When 0X118 had reduced
core fucosylation (34%), the reduction of T-cell activation was enhanced compared with fully fucosylated (97%) 0X118.
Overall, Fc-optimisation of B02 to produce a novel anti-OX40L antibody (0X118) was achieved via exposure to 2-FF. Reduced levels of fucosylation and an increased affinity to Fc-receptors (CD16a) was observed following exposure to 2-FF. A saturation of the effects of which were observed at fucosylation levels of -50%, below which no improved Fc receptor affinity was achieved. Further, reduced fucosylation of 0X118 resulted in significantly decreased T-cell activation compared with control and fully fucosylated 0X118.
Example 3: Allogeneic mixed lymphocyte reaction
Aim:
To investigate the in vitro effect of on 0X118 on different cell types.
Material and Methods:
CTLA-4 Ig (clone Belatacept, Bristol Myers Squibb, SEQ ID NO: 15) was purchased from Apoteket Hjartat AB. Anti-OX40L lgG1 reference antibody (clone R4930/Oxelumab; lgG1 , Fc-active) and Anti-OX40L lgG4 reference antibody (clone Amlitelimab; lgG4, Fc-silent, SEQ ID NO:16, SEQ ID NO:17) were produced via transient expression in CHO K1 by Proteogenix. 0X118 with reduced core fucosylation (34%) was used in the experiments (other parts of the glycosylation profile were ordinary for human lgG1 expressed in CHO K1, man5 5.2%, GO 65.2%, GO-GN 1%, GOF 22%, G1Fa 1.43%, G1 Fb 0.54%, G2F 0.29%).
Allogeneic mixed lymphocyte reaction: Peripheral blood mononuclear cells (PBMC) were isolated via Ficoll Paque Plus (Cytiva) density gradient centrifugation from buffy coats (500g x 30 min). Buffy coats were obtained from anonymous healthy adult donors from the Akademiska University Hospital blood bank, and PBMC isolation was performed within 24 hours of blood collection. In line with Swedish legislation, an ethics approval was not needed since the blood bank provides buffy coats for research purposes from anonymous donors and biological material cannot be traced back to a specific individual. Isolated PBMC were mixed in PBS at a concentration of 2.0 x 107 cells per mL and stained with CFSE (Thermo Fisher Scientific Inc) according to the manufacturer’s instructions. PBMC from 2 different donors were mixed in a two-way
allogeneic mixed lymphocyte reaction, i.e., PBMCs from neither donor were inactivated. VPD450-stained PBMC were washed and resuspended in 10% heat- inactivated Ultralow IgG FBS (Gibco) in AIM V medium (Gibco, Thermo Fisher Scientific Inc). Resuspended PBMCs were dispensed into round-bottom 96-well cell culture plates, and pure medium or medium supplemented with test article was added to a final concentration of 2 x 106 cells per mL in a final volume of 200 pL. Mixed lymphocyte reactions were incubated at 37°C, 5% CO2 for 7 days. Supernatant postculture was kept and analysed for cytokine content (measuring levels of IL-10, IL-4, TNFa) utilizing Legendplex (BioLegend) cytokine assay according to the manufacture's instructions.
Flow cytometry: After 7 days of mixed lymphocyte reaction, samples were washed and then stained with antibodies against cell surface antigens. For intracellular staining FoxP3 staining kit (BD Biosciences) was used according to the manufacturer’s instructions. Samples were stained in the dark at 4 °C and washed twice in staining buffer (BD Biosciences), followed by analysis using a BC Cytoflex flow cytometer (Beckman Coulter). T cells were defined as CD3+ cells. Proliferating T-cells were defined as CFSE low. Tregs were defined as CD3+, CD4+, CD25hi and FoxP3+. Naive T cells were defined as CD3+, CD45RA+, CCR7+.
Graphs and statistical analysis: Visualization of results and statistical analysis of underlying data were carried out using GraphPad Prism 10 software (GraphPad Software). Data were analysed using one-way analysis of variance (ANOVA) followed by Dunnett’s multiple comparison test.
Results:
OX118 (10 pg/mL) decreased the proliferation of CD4+ and CD8+ cells compared to control, and other OX40L-targeting antibodies (lgG1 Ref and lgG4; 10 pg/mL) and CTLA-4 Ig (15 pg/mL) (Figure 4).
OX118 (10 pg/mL) increased the % of Tregs cells among all proliferating cells compared to control, and other OX40L-targeting antibodies (lgG1 Ref and lgG4; 10 mg/mL) and CTLA-4 Ig (15 pg/mL) (Figure 5A). This effect was observed to be concentration-dependent, with increases in the % of Tregs cells among all proliferating cells observed at OX118 concentrations from 102-105 ng/mL (Figure 5B).
Addition of 0X118 (10 pg/mL) in a two-way mixed lymphocyte assay resulted in an increased population of naive T cells among proliferating CD4+ cells control, and other OX40L-targeting antibodies (lgG1 Ref and lgG4; 10 pg/mL) and CTLA-4 Ig (15 pg/mL) (Figure 6).
Addition of 0X118 (10 pg/mL) in a two-way mixed lymphocyte reaction resulted in a decrease in IL-4 compared to control. 0X118 (10 pg/mL) reduced TNFa levels compared with control and other OX40L-targeting antibody; lgG1 Ref (10 pg/mL), and CTLA-4 Ig (15 pg/mL). IL-10 levels remained comparable to control and other OX40L- targeting antibody; lgG1 Ref (10 pg/mL), and CTLA-4 Ig (15 pg/mL) (Figure 7).
Conclusions:
0X118 reduced proliferation of CD4+ and CD8+ cells in vitro, while it increased the amount of proliferating Tregs cells. Furthermore, 0X118 decreased levels of the pro- inflammatory cytokines; IL-4 and TNFa in vitro.
Example 4: Xeno-Graft-vs-host-disease model
Aim:
To assess the potential for 0X118 (anti-OX40L antibody with reduced fucosylation) to act preventively in the development of Graft-vs-host-disease in a xenogeneic setting where NSG mice are challenged with human PBMC.
Material and Methods:
Female NSG (NOD. Cg-Prkdcsdd H2rgtmlWl/Szi) mice 6-9 weeks of age were used in the study. Mice were grouped into treatment (control, 0X118 10mg/kg or 0X118 50mg/kg) groups based on body weight. PBMCs were obtained from one healthy, random, and uncharacterized donor. PBMCs were used freshly after isolation from leukoreduction system (LRS) chambers.
On day 0, 10 million freshly isolated human PBMCs were administered via tail vein.
The first dose of 0X118 was given intraperitoneally (i.p) 24 hours after PBMC injection, 0X118 was dosed once weekly for 3 consecutive weeks (4 i.p doses at days 1 , 8, 15 and 22). Group sizes were 5 mice per group. Human isotype IgG 1 was used as control
vehicle (BioXCell, catalog #BE0297), 0X118 was given in doses of 10 mg/kg (low dose) or 50 mg/kg (high dose) once weekly until day 22. Activity scores were assessed daily (see Table 5). A score of 3 for more than two days or mice with a score <3 for one day was euthanized.
GvHD scores (see Table 6) were assessed daily for monitoring progression of GvHD, by using whole number scoring in five different domains ranging from 0 (no symptoms) to 2 (severe symptoms).
Flow cytometry: Blood samples were collected at day 8, 15, 22 for flow cytometry. Cells were pelleted by centrifugation, at 400 x g for 5 min with subsequent removal of supernatant. Fc-block antibody (10 pg/well of a 1 :100 dilution in FC buffer). Then specific antibodies were added as recommended by the manufacturer in Zombie Aqua Fixable Viability stain (diluted 1 :100 in PBS buffer) and the plates were incubated at 4 °C protected from light for 30 min. Antibodies utilised were: hCD3 (clone LICHT1, Biolegend), hCD45 (clone H130, Biolegend), hCD4 (clone RPA-T4, Biolegend), hCD8 (clone RPA-T8, Biolegend), hCD25 (clone M-A251, Biolegend), H LA-DR (clone L243, Biolegend), hFoxp3 (clone PCH101, Thermo Scientific; for this antibody an intracellular staining protocol was used). HCD62L (clone DREG-56, Biolegend). Antibodies were used according to the manufacturer’s instructions. FC buffer: 2% FBS in PBS; FBS (#A21-102 - PAA), PBS (#14190169 - Gibco/Fisher Scientific). Fc-block antibody: CD16/32 purified (2.4G2), 0.5 mg/ml (#553142 - BD Biosciences). Zombie Aqua Fixable Viability Kit (#423102 - BioLegend).
Cells were washed by the addition of 200 pl FC buffer followed by centrifugation of the plates at 400 x g for 5 min, and the supernatant was removed. Samples were resuspended in 400 pl FC buffer for analysis with the Attune NXT flow cytometer (Thermo Fisher) (violet (405 nm)/blue (488 nm)/yellow (561 nm)/red (638 nm) laser configuration). Characterization of immune cellular phenotype of blood was performed using flow cytometry. Cell populations studied are detailed in Table 7.
Analysis: Doublet exclusion was performed according to forward scatter height versus forward scatter area to include only single cells followed by forward/sideward scatter to
exclude debris and finally live/dead (live/dead, catalog no. 423102, Biolegend) discrimination. Gating was performed according to table 5.
Results 0X118 treatment prevents GvHD development in mice transplanted with human peripheral blood mononuclear cells (PBMCs): Following induction of GvHD in mice, defined as rising GvHD score (mean GvHD score at day 30: control: 6,8;
10mg/kg 0X118 group; 0,8; 50mg/kg OX118 group; 1 ,2). Treatment with OX118 prevented the development of a peripheral population of hCD45+ leukocytes, compared with control. This effect was observed at both low and high doses of 0X118;
10 mg/kg and 50 mg/kg, respectively (Figure 8). Peripheral chimerism was observed to be between 1-3% of all cells in peripheral blood, compared with 63% in control groups at day 21. Comparative to previously published literature (Tripathi et al. 2019), this demonstrates a 3-4-fold increase in the control of chimerism/graft proliferation.
0X118 reduces activation of T-cells in peripheral blood following induction of GvHD in mice: 21 days after induction of GvHD in mice, treatment with 0X118 reduced the relative populations of activated CD4+, CD8+ and CD14+ T-cells compared with control. This effect was observed at both low and high doses of 0X118; 10 mg/kg and 50 mg/kg, respectively (Figure 9).
0X118 increases proportion of T regulatory cells (T-Regs) among proliferating CD4+ T- cells following induction of GvHD in mice: 21 days after induction of GvHD in mice, treatment with 0X118 (pooled data from all 0X118 treated mice) increased the population of T-regs compared with control (Figure 10).
Conclusion
0X118 treatment, at both high and low doses, prevents induction and progression of GvHD in vivo via preventing peripheral chimerism, activation of peripheral T-cell populations and enriching the peripheral T-reg population.
Example 5: Xenogeneic GvHD model - benchmarking 0X118 (Treatment A) vs antihuman OX40L lgG4 isotyope (Treatment B).
Aim: To evaluate and compare the efficacy of anti-OX40L antibodies with different isotypes in a xenogeneic Graft-versus-Host Disease (xeno-GvHD) model, focusing on chimerism and tissue infiltration by human CD45+ (hCD45+) cells.
Materials and Methods:
NOG mice were subjected to whole-body irradiation with a dose of 1.0 Gy. After a 24- hour rest period, 3 million human PBMCs (Peripheral Blood Mononuclear Cells) were injected into the tail vein, marking Day 0. Treatment began on Day 1 , 10 mg/kg doses of Treatment A (0X118), Treatment B (anti-hOX40L lgG4), and a control (human isotype IgG 1 ) were administered. GvHD scores (see Table 6) and body weight were assessed every other day.
Flow cytometry: Blood samples were collected and whole blood was centrifuged at 3000 rpm for 5 minutes at 4°C to isolate plasma (which was frozen at -80 for later cytokine analysis). The cells were resuspended in lysis buffer and incubated for 2 min at room temperature. The residual white blood cells were suspended in DPBS, Fc- block for mouse (rat anti-mouse CD16/CD32), human (Human TruStain FcX) and Live/Dead (Zombie NIR, BioLegend, 426106) was added, the mixture was incubated for 15 min at room temperature in the dark. Subsequently antibodies were added and incubated for 30 minutes at 4°C in the dark. For chimerism determination mCD45 (PerCP, BioLegend, 103130) and hCD45 (AF700, BioLegend, 368514) was added. All samples were detected by Cytek (Aurora-5L). If possible >5 000 hCD45+ events were recorded.
IHC: At the end of the study, organs (skin, colon, and lung) were harvested for immunohistochemical staining of human CD45+ cells in the tissue. Paraffin sectioning: wax block was onto Leica HistoCore Arcadia C for 15-30 min prior to section. Leica HistoCore BIOCUT machine sectioned the block to 4 pm thickness Slides were dried at 60°C for 60 min. Automatic IHC with BOND RX was used. hCD45 antibody used was CST, #13917S, dilution 1:200.
Results:
By Day 21 , the clinical score in the control group rapidly increased, while Treatment A and Treatment B maintained similar scores until around Day 40. At this point, the clinical score for Treatment A decreased, whereas the mice in Treatment B exhibited
increasing clinical severity (Fig 11 A). Similarly, body weight in Treatment A increased after Day 40, while it decreased in Treatment B (Figure 11B).
Chimerism, indicated by circulating hCD45+ cells, was significantly lower in Treatment A group compared to both Treatment B and the control group (Fig 11C).
Additionally, treatment A resulted in a reduction of hCD45+ cell infiltration in the colon, skin, and lungs (Fig 11 D), compared with Treatment B.
Conclusion:
0X118 (Treatment A) demonstrated superior control of the xenogeneic GvHD model compared to anti-OX40L lgG4 (Treatment B) and isotype lgG1 controls. 0X118 effectively reduced tissue infiltration by hCD45+ cells, supporting its potential in managing GvHD.
Example 6: Antibody-Dependent Cell-Mediated Cytotoxicity (ADCC) Assay
Aim: To evaluate and compare the ADCC activity of 0X118 in relation to Herceptin® (Trastuzumab; Roche, SH0346) as a positive control.
Materials and Methods:
0X118 is a humanized lgG1 antibody targeting OX40L. The ADCC Bioassay Effector Cell V variant (High Affinity) (BPS Bioscience, 60541) was used as the effector cells, while SKBR-3 cells (ATCC, HTB-30) were employed as target cells for system control. CHO-K1/OX40L cells (GenScript, M00563) were used as the test target cells. Herceptin® (Roche, SH0346) was utilized as the system positive control, and Human lgG1 (Sino Biological, MA17JU1250) served as the negative control. The assay was conducted using three different lots of 0X118 drug substance (DS).
Target cells were plated in 96-well plates at a density of 10,000 cells per well. Controls and test samples were added to the wells, and the plates were incubated at 22-28°C for
30 minutes. Effector cells were then added at 60,000 cells per well, and the plates were further incubated in a cell incubator at 37°C with 5% CO2 for 6 hours.
After incubation, the Bio-Lite™ Luciferase Assay System (Vazyme, DD1201) was added at 75 pL per well, and the luminescent signal was measured using a microplate reader. The signal was plotted against the logarithm of the antibody concentration (ng/mL), and dose-response curves were fitted using a four-parameter fit (4P-Fit) via GraphPad software to calculate the median effective concentration (ECso) values.
Results:
The dose-response curve for OX118 ADCC activity, as compared to Herceptin®, is illustrated in Figure 13. The EC50 values of the OX118 drug substance from different lots are summarized in Table 8_below:
Conclusion:
0X118 exhibited potent ADCC activity with significantly lower EC50 values compared to the system positive control, Herceptin®. The EC50 values for the three different 0X118 DS lots were consistent, demonstrating robust and reproducible ADCC activity. In contrast, the negative control (Human IgG 1 ) showed no ADCC activity, confirming the specificity of the response.
Example 7 0X118 binding to FcyR
Aim: To evaluate the binding affinity of the Fc region of 0X118 to various Fey receptors, including FcyRla (CD64), FcyRlla (CD32a 131 H, CD32a 131 R), FcyRIHa (CD16a 158V, CD16a 158F), and the neonatal Fc receptor (FcRn), using Herceptin® as a positive control.
Materials and Methods: Biolayer Interferometry (BLI) was employed using the Sartorius RED96e system to assess the binding affinity of 0X118 drug substance (DS) to Fey receptors. The test was performed using three different lots of 0X118 DS: lot #1 toxicology batch), and lots #2 and #3 (GMP DS batches). Herceptin® (Roche, SH0346) was used as the system positive control, with Human lgG1 serving as the negative control.
The FcyR proteins were immobilized, and gradient-diluted antibody samples were introduced for binding analysis after baseline equilibration, followed by dissociation in buffer. The assay was conducted at 25°C using 0.02% PBSTB as the running buffer. Data acquisition was performed at standard kinetics (5.0 Hz, averaging 20), and data analysis was conducted using the Octet® System Data Analysis software to calculate the binding kinetics parameters (KD, kon, koff).
Results:
The binding affinities of 0X118 and Herceptin® to various Fey receptors are presented in Tables 9, 10, 11 and 12.
Conclusion:OX118 demonstrated enhanced binding affinity toward FcyRIHa compared to Herceptin®. The consistency across the three different lots of 0X118 suggests robust manufacturing and reproducibility. Binding affinities to other Fey receptors, including FcyRla, FcyRlla, and FcRn, remained comparable to Herceptin®. 0X118 had an increased affinity for FcyRIHa, the main mediator of ADCC, while other FcRs remained similar to the reference antibody (Herceptin®).
Example 8: Binding Affinity of 0X118 to C1q
Aim: To evaluate the binding affinity of 0X118 to C1q, a key component of the complement system, which is involved in initiating the classical complement pathway. Binding to 01 q is an important functional property for antibodies, as it can trigger immune responses leading to enhanced pathogen elimination. This study compares the binding of 0X118 to 01 q against Herceptin®, a known lgG1 isotype antibody with 01 q binding activity, as a positive control, to determine if 0X118 maintains this essential functional property.
Materials and Methods:
Biolayer Interferometry (BLI) using the Sartorius Octet RED96e system was employed to measure the binding affinity of 0X118 drug substance (DS) to the C1q protein. Herceptin® (Roche, SH0346) was used as the positive control. Three different lots of 0X118 DS were tested: lot #1 (toxicology batch), and lots #2 and #3 (GMP DS batches).
The assay was performed at 25°C, with 0.02% PBSTB as the running buffer. The C1q protein was immobilized on the sensor, and gradient-diluted antibodies were introduced for binding after baseline equilibration. Dissociation was conducted using buffer. Data were acquired at a standard kinetics rate (5.0 Hz, averaging 20), and analysis was performed using the Octet® System Data Analysis software to calculate binding parameters (KD, Rmax).
Results:
The binding activity of 0X118 to 01 q, compared to Herceptin®, is summarized in Table 13 below:
Conclusion:
0X118 exhibits binding activity to C1q comparable to Herceptin®, with no significant difference in binding affinity across the tested lots of 0X118. This suggests that 0X118 has the ability to engage with the complement system. The consistency in C1q binding across different production batches supports the robustness of 0X118 manufacturing.
Sequence overview
The novel anti-OX40L monoclonal antibody (mAB; 0X118) is based on affinity maturation and Fc-optimisation of a previously clinically tested mAB; R4930 (US 7,501 ,496 B1). CDR sequences were determined via the Chothia Numbering Scheme. Underlined, bold amino acids are differing from corresponding amino acids of R4930.
0X118 VL sequence (SEQ NO:1)
CRASQGISSLLAWYQQKPEKAPKSLIFDRSRLQSGVPSRFSGSGSGTDFTLTISSLQP EDFATYYCQQYNSYPYTF
0X118 LC CDR1 (SEQ ID NO: 2)
RASQGISSLLA
0X118 LC CDR2 (SEQ ID NO: 3)
DRSRLQS
0X118 LC CDR3 (SEQ ID NO: 4)
QQYNSYPYT
0X118/R4930 VH sequence (SEQ N0:5)
EVQLLESGGGLVQPGGSLRLSCAASGFTFNSYAMSWVRQAPGKGLEWVSIISGSGG FTYYADSVKGRFTISRDNSRTTLYLQMNSLRAEDTAVYYCAKDRLVAPGTFDYWGQG ALVTVSS
OX118/R493Q HC CDR1 (SEQ ID NO: 6)
GFTFNSY
OX118/R493Q HC CDR2 (SEQ ID NO: 7)
SGSGGF
OX118/R493Q HC CDR3 (SEQ ID NO: 8)
DRLVAPGTFDY
R4930 VL sequence (SEQ N0:9)
CRASQGISSWLAWYQQKPEKAPKSLIYAASSLQSGVPSRFSGSGSGTDFTLTISSLQP EDFATYYCQQYNSYPYTF
R4930 LC CDR1 (SEQ ID NO: 10)
RASQGISSWLA
R4930 LC CDR2 (SEQ ID NO: 11)
AASSLQS
R4930 LC CDR3 (SEQ ID NO: 12)
QQYNSYPYT
0X118 Light Chain (SEQ NO: 13)
DIQMTQSPSSLSASVGDRVTITCRASQGISSLLAWYQQKPEKAPKSLIFDRSRLQSGV
PSRFSGSGSGTDFTLTISSLQPEDFATYYCQQYNSYPYTFGQGTKLEIKRTVAAPSVFI
FPPSDEQLKSGTASVVCLLNNFYPREAKVQWKVDNALQSGNSQESVTEQDSKDSTY
SLSSTLTLSKADYEKHKVYACEVTHQGLSSPVTKSFNRGEC
0X118 Heavy Chain (SEQ N0:14)
EVQLLESGGGLVQPGGSLRLSCAASGFTFNSYAMSWVRQAPGKGLEWVSIISGSGG
FTYYADSVKGRFTISRDNSRTTLYLQMNSLRAEDTAVYYCAKDRLVAPGTFDYWGQG
ALVTVSSASTKGPSVFPLAPSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGV
HTFPAVLQSSGLYSLSSVVTVPSSSLGTQTYICNVNHKPSNTKVDKKVEPKSCDKTHT
CPPCPAPELLGGPSVFLFPPKPKDTLMISRTPEVTCVVVDVSHEDPEVKFNWYVDGV
EVHNAKTKPREEQYNSTYRWSVLTVLHQDWLNGKEYKCKVSNKALPAPIEKTISKAK
GQPREPQVYTLPPSRDELTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKTTPPV
LDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPG
Belatacept (CTLA-4 Ig fusion protein; SEQ ID NO: 15)
MHVAQPAVVLASSRGIASFVCEYASPGKYTEVRVTVLRQADSQVTEVCAATYMMGN
ELTFLDDSICTGTSSGNQVNLTIQGLRAMDTGLYICKVELMYPPPYYEGIGNGTQIYVID
PEPCPDSDQEPKSSDKTHTSPPSPAPELLGGSSVFLFPPKPKDTLMISRTPEVTCVVV
DVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKEYK
CKVSNKALPAPIEKTISKAKGQPREPQVYTLPPSRDELTKNQVSLTCLVKGFYPSDIAV
EWESNGQPENNYKTTPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHN
HYTQKSLSLSPGK
Amlitelimab Heavy chain (Anti-QX40L lgG4; SEQ ID NO: 16)
ASTKGPSVFPLAPCSRSTSESTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVL
QSSGLYSLSSWTVPSSSLGTKTYTCNVDHKPSNTKVDKRVESKYGPPCPPCPAPEF
LGGPSVFLFPPKPKDTLMISRTPEVTCVWDVSQEDPEVQFNWYVDGVEVHNAKTKP
REEQFNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKGLPSSIEKTISKAKGQPREPQV YTLPPSQEEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKTTPPVLDSDGSFF LYSRLTVDKSRWQEGNVFSCSVMHEALHNHYTQKSLSLSLGK
Amlitelimab Light chain (Anti-QX40L lqG4; SEQ ID NO:17)
DIQMTQSPSSLSASVGDRVTITCRASQSISSYLNWYQQKPGKAPNLLIYAASSLQSGV PSRFSGSGSETDFTLTISSLQPEDFATYYCQQSHSVSFTFGPGTKVDIKTVAAPSVFIF PPSDEQLKSGTASVVCLLNNFYPREAKVQWKVDNALQSGNSQESVTEQDSKDSTYS LSSTLTLSKADYEKHKVYACEVTHQGLSSPVTKSFNRGEC
References
Almahayni K, Spiekermann M, Fiore A, Yu G, Pedram K, Mdckl L. Small molecule inhibitors of mammalian glycosylation. Matrix Biol Plus. 2022 Mar 16;16:100108. doi: 10.1016/j.mbplus.2022.100108. Erratum in: Matrix Biol Plus. 2022 Dec 13;16:100126. PMID: 36467541; PMCID: PMC9713294.
Chatenoud L, Thervet E, Primo J, Bach JF. Anti-CD3 antibody induces long-term remission of overt autoimmunity in nonobese diabetic mice. Proc Natl Acad Sci U S A. 1994 Jan 4;91(1):123-7. doi: 10.1073/pnas.91.1.123. PMID: 8278351; PMCID: PMC42898.
Compaan DM, Hymowitz SG. The crystal structure of the costimulatory OX40-OX40L complex. Structure. 2006 Aug;14(8):1321-30. doi: 10.1016/j.str.2006.06.015. PMID: 16905106.
Edwards E, Livanos M, Krueger A, Dell A, Haslam SM, Mark Smales C, Bracewell DG. Strategies to control therapeutic antibody glycosylation during bioprocessing: Synthesis and separation. Biotechnol Bioeng. 2022 Jun;119(6): 1343- 1358. doi: 10.1002/bit.28066. Epub 2022 Feb 28. PMID: 35182428; PMCID: PMC9310845.
Guo J, Tu H, Jing L, McCarthy D, Atouf F. USP Reference Standard Monoclonal Antibodies: Tools to Verify Glycan Structure. Pharmaceuticals (Basel). 2022 Mar 5;15(3):315. doi: 10.3390/ph15030315. PMID: 35337113; PMCID: PMC8951495.
Haile Y, Adegoke A, Laribi B, Lin J, Anderson CC. Anti-CD52 blocks EAE independent of PD-1 signals and promotes repopulation dominated by double-negative T cells and newly generated T and B cells. Eur J Immunol. 2020 Sep;50(9):1362-1373. doi: 10.1002/eji.201948288. Epub 2020 May 27. PMID: 32388861.
Kanda Y, Imai-Nishiya H, Kuni-Kamochi R, Mori K, Inoue M, Kitajima-Miyama K, Okazaki A, lida S, Shitara K, Satoh M. Establishment of a GDP-mannose 4,6- dehydratase (GMD) knockout host cell line: a new strategy for generating completely non-fucosylated recombinant therapeutics. J Biotechnol. 2007 Jun 30;130(3):300-10. doi: 10.1016/j.jbiotec.2007.04.025. Epub 2007 May 6. PMID: 17559959.
Li W, Zhu Z, Chen W, Feng Y, Dimitrov DS. Crystallizable Fragment Glycoengineering for Therapeutic Antibodies Development. Front Immunol. 2017 Nov 13;8:1554. doi: 10.3389/fimmu.2017.01554. PMID: 29181010; PMCID: PMC5693878.
Luo, S., Zhang, B. Benchmark Glycan Profile of Therapeutic Monoclonal Antibodies Produced by Mammalian Cell Expression Systems. Pharm Res (2023). https : //do i . org/10.1007/s 11095-023-03628-4
Mishra N, Spearman M, Donald L, Perreault H, Butler M. Comparison of two glycoengineering strategies to control the fucosylation of a monoclonal antibody. J Biotechnol. 2020;324S:100015. doi: 10.1016/j.btecx.2020.100015. Epub 2020 Feb 20. PMID: 34154738.
Reusch D, Tejada ML. Fc glycans of therapeutic antibodies as critical quality attributes. Glycobiology. 2015 Dec;25(12):1325-34. doi: 10.1093/glycob/cwv065. Epub 2015 Aug 11. PMID: 26263923; PMCID: PMC4634315.
Rillahan, C., Antonopoulos, A., Lefort, C. et al. Global metabolic inhibitors of sialyl- and fucosyltransferases remodel the glycome. Nat Chem Biol 8, 661-668 (2012). htps://doi.org/10.1038/nchembio.999
Tripathi T, Yin W, Xue Y, Zurawski S, Fujita H, Hanabuchi S, Liu YJ, Oh S, Joo H.
Central Roles of OX40L-OX40 Interaction in the Induction and Progression of Human T
Cell-Driven Acute Graft-versus-Host Disease. Immunohorizons. 2019 Mar;3(3):1 IQ-
120. doi: 10.4049/immunohorizons.1900001. PMID: 31240276; PMCID: PMC6592051 .
Wacker C, Berger CN, Girard P, Meier R. Glycosylation profiles of therapeutic antibody pharmaceuticals. Eur J Pharm Biopharm. 2011 Nov;79(3):503-7. doi: 10.1016/j.ejpb.2011.06.010. Epub 2011 Jul 2. PMID: 21745568.
Yamane-Ohnuki N, Satoh M. Production of therapeutic antibodies with controlled fucosylation. MAbs. 2009 May-Jun; 1(3):230-6. doi: 10.4161/mabs.1.3.8328. Epub 2009 May 28. PMID: 20065644; PMCID: PMC2726589.
Yamane-Ohnuki N, Kinoshita S, Inoue-Urakubo M, Kusunoki M, lida S, Nakano R, Wakitani M, Niwa R, Sakurada M, llchida K, Shitara K, Satoh M. Establishment of FLIT8 knockout Chinese hamster ovary cells: an ideal host cell line for producing completely defucosylated antibodies with enhanced antibody-dependent cellular cytotoxicity. Biotechnol Bioeng. 2004 Sep 5;87(5):614-22. doi: 10.1002/bit.20151. PMID: 15352059.
Yoshioka T, Nakajima A, Akiba H, Ishiwata T, Asano G, Yoshino S, Yagita H, Okumura K. Contribution of 0X40/0X40 ligand interaction to the pathogenesis of rheumatoid arthritis. Eur J Immunol. 2000 Oct;30(10):2815-23. doi: 10.1002/1521- 4141(200010)30:10<2815::AID-IMMU2815>3.0.CO;2-#. PMID: 11069062.
Items A
A1.An antibody comprising a heavy chain of SEQ ID NO: 14 and a light chain comprising a VL sequence of SEQ ID NO: 1 , wherein core fucosylation of the heavy chain is below 80%.
A2. The antibody of item 1 , wherein the light chain has the amino acid sequence of SEQ ID NO: 13.
A3. The antibody of item 1 , wherein the core fucosylation is below 70%, such as below 60%, for example below 50%, such as below 40%, for example below 30%, such as below 20% of the total glycans.
A4.The antibody of any one of items 1 - 3, wherein the core fucosylation is determined by quantitative analysis of glycans as (Area with fucosylated glycan)/(total glycan area).
A5. A method of producing an anti-OX40L antibody or antibody derivative, comprising expressing polynucleotides encoding polypeptide of SEQ ID NO: 13 and 14 in a cell, wherein the cell is exposed to culture medium comprising an inhibitor of glucosidases I and II, and subsequently purifying the antibody.
A6. A method of producing an anti-OX40L antibody or antibody derivative, comprising expressing polynucleotides encoding polypeptide of SEQ ID NO: 13 and 14 in a cell, wherein the cell is exposed to culture medium comprising a fucose analog, and subsequently purifying the antibody.
A7.The method of item 6, wherein the fucose analog is an inhibitor of fucokinase, GDP-fucose-pyrophosphorylase, fucosyltransferase (FUT), GDP-mannose 4,6- dehydratase, GDP-fucose synthetase, and/or a fucose transporter, such as GDP-fucose transporter.
A8.The method of item 6, wherein the fucose analog is an inhibitor of a fucosyltransferase (FUT), such as 1,6-fucosyltransferase (FUT8).
A9. A method of producing an anti-OX40L antibody or antibody derivative, comprising expressing polynucleotides encoding polypeptide of SEQ ID NO: 13 and 14 in a cell that is genetically modified to prevent core fucosylation.
A10. The method according to item 9, wherein the cell is selected from the following cells: CHO (e.g., CHO K1 , DXB-11 CHO, Veggie-CHO), COS (e.g., COS-7), Syrian hamster, rat myleloma, mouse myeloma (e.g., SP2/0, NSO), retinal cell, Vero, CV1 , kidney (e.g., HEK293, 293 EBNA, MSR 293, MDCK, HaK, BHK, BHK21), HeLa, HepG2, WI38, MRC 5, Colo205, HB 8065, HL-60, Jurkat, Daudi, A431 (epidermal), CV-1 , U937, 3T3, L cell, C127 cell, MMT 060562, Sertoli cell, BRL 3A cell, HT1080 cell, a human myeloma cell, tumor
cell, a human lymphoma cell (e.g., a Namalwa cell) and a cell line derived from an aforementioned cell.
A1 1. The method according to any one of items 9 or 10, wherein fucosylation of the antibody or antibody derivative is restored and/or titrated to a desired level via exposure to fucose, wherein the cell is exposed to a culture medium comprising fucose.
A12. A method of testing a defucosylated anti-OX40L antibody or antibody derivative, comprising testing the antibody in a T-cell activation assay and verifying that the T-cell activation is reduced compared to a fucosylated anti- OX40L antibody.
A13. The antibody or antibody derivative according to any one of the preceding items, for use in the prophylaxis and/or treatment of inflammatory disorders.
A14. The antibody or antibody derivative according to any one of the preceding items, for use in the prophylaxis and/or treatment of autoimmune diseases.
A15. The antibody or antibody derivative according to items 13-15, wherein the disease is selected from the group consisting of: Graft-vs-host-disease, allograft rejection, asthma, Systemic lupus, arthritis, inflammatory bowel disease, ulcerative colitis, Crohn’s disease, diabetes, atopic dermatitis, psoriasis, hidradenitis suppurativa, immunoglobulin A nephropathy, Hashimoto disease, Graves’ disease, chronic rhinosinusitis and multiple sclerosis.
Items
1. A polypeptide comprising an amino acid sequence of SEQ ID NO:1 (VL).
2. An anti-OX40L antibody having a variable light chain and a variable heavy chain, wherein the variable light chain comprises the following CDRs: a. SEQ ID NO: 2; b. SEQ ID NO: 3; and
c. SEQ ID NO: 4; and wherein the variable heavy chain comprises the following CDRs: d. SEQ ID NO: 6; e. SEQ ID NO: 7; and f. SEQ ID NO: 8.
3. An anti-QX40L antibody comprising a heavy chain and a light chain, wherein the light chain comprises a VL comprising the following CDRs: g. SEQ ID NO: 2; h. SEQ ID NO: 3; and i. SEQ ID NO: 4; and wherein the heavy chain comprises a constant region and a VH comprising the following CDRs: j. SEQ ID NO: 6; k. SEQ ID NO: 7; and l. SEQ ID NO: 8, and, wherein core fucosylation of the constant region of the heavy chain is below%.
4. An anti-QX40L antibody comprising a heavy chain and a light chain, wherein the light chain comprises a VL comprising the following CDRs: m. SEQ ID NO: 2; n. SEQ ID NO: 3; and o. SEQ ID NO: 4; and wherein the heavy chain comprises the following CDRs: p. SEQ ID NO: 6; q. SEQ ID NO: 7; and r. SEQ ID NO: 8, and, wherein core fucosylation of the heavy chain is below 80%.
5. An anti-QX40L antibody comprising an IgG 1 heavy chain and a light chain, wherein the light chain comprises a VL comprising the following CDRs: s. SEQ ID NO: 2; t. SEQ ID NO: 3; and
u. SEQ ID NO: 4; and wherein the IgG 1 heavy chain comprises the following CDRs: v. SEQ ID NO: 6; w. SEQ ID NO: 7; and x. SEQ ID NO: 8, and, wherein core fucosylation of the I gG 1 heavy chain is below 80%.
6. An antibody comprising a light chain comprising a VL sequence of SEQ ID NO: 1 , wherein core fucosylation of the antibody is below 80%.
7. An anti-QX40L antibody comprising a heavy chain and light chain, comprising a VL sequence of SEQ ID NO: 1, wherein core fucosylation of the heavy chain is below 80%.
8. The antibody according to any of the preceding items, wherein the antibody comprises an IgG 1 heavy chain.
9. The antibody according to any of the preceding items, wherein the antibody comprises an lgG1 heavy chain constant region.
10. The antibody according to any of the preceding items, wherein the antibody comprises an lgG1 heavy chain constant region, wherein the IgG 1 heavy chain constant region is glycosylated.
11. An antibody comprising a heavy chain of SEQ ID NO: 14 and a light chain comprising a VL sequence of SEQ ID NO: 1 , wherein core fucosylation of the heavy chain is below 80%.
12. An anti-QX40L antibody comprising a heavy chain of SEQ ID NO: 14 and a light chain comprising a VL sequence of SEQ ID NO: 1, wherein core fucosylation of the heavy chain is below 80%.
13. The antibody of any of the preceding items, wherein the core fucosylation of the constant region of the heavy chain is below 80%, as determined by capillary electrophoresis, LC-MS, High-Performance liquid chromatography with Fluorescence Detection (HPLC-FD), High-Performance Anion-Exchange
chromatography with Pulsed Amperometric Detection (HPAE-PAD), mass spectrometry, hydrazinolysis and/or enzyme digestion.
14. The antibody of any of the preceding items, comprising a variable heavy chain of SEQ ID NO: 5, and a variable light chain of SEQ ID NO: 1.
15. The antibody of any of the preceding items, comprising a heavy chain of SEQ ID NO: 14 and a light chain of SEQ ID NO: 13.
16. The antibody of any of the preceding items, wherein the antibody is glycosylated with at the least 2 kDa of total glycans.
17. The antibody of any of the preceding items, wherein the antibody is glycosylated with at the most 3 kDa of total glycans.
18. The antibody of any of the preceding items, wherein the antibody is glycosylated with at the least 2kDa of total glycans and at the most 3 kDa of total glycans.
19. The antibody of any of the preceding items, wherein the heavy chain constant region is glycosylated with at the least 2 kDa of total glycans.
20. The antibody of any of the preceding items, wherein the heavy chain constant region is glycosylated with at the most 3 kDa of total glycans.
21 . The antibody of any of the preceding items, wherein the heavy chain constant region is glycosylated with at the least 2kDa of total glycans and at the most 3 kDa of total glycans.
22. The antibody of any of the preceding items, wherein the antibody glycans comprise between 50% and 80% of glycan GO.
23. The antibody of any of the preceding items, wherein the antibody glycans comprise between 50% and 80% of glycan GO, such as between 55 and 80%, such as between 60% and 80%, such as between 65% and 80%, such as
between 70% and 80%, such as between 75% and 80%, such as between 50% and 75%, such as between 50% and 70%, such as between 50% and 65%, such as between 50% and 60%, such as between 50% and 55%.
24. The antibody of any of the preceding items, wherein total glycans of the antibody comprise between 50% and 80% of glycan GO.
25. The antibody of any of the preceding items, wherein total glycans of the antibody comprise between 50% and 80% of glycan GO, such as between 55 and 80%, such as between 60% and 80%, such as between 65% and 80%, such as between 70% and 80%, such as between 75% and 80%, such as between 50% and 75%, such as between 50% and 70%, such as between 50% and 65%, such as between 50% and 60%, such as between 50% and 55%.
26. The antibody of any of the preceding items, wherein the core fucosylation is below 70%, such as below 60%, for example below 50%, such as below 40%, for example below 30%, such as below 20% of total glycans of the antibody.
27. The antibody of any of the preceding items, wherein the core fucosylation is determined by quantitative analysis of glycans as (Area with fucosylated glycan)/(total glycan area).
28. The antibody of any one of any of items 1-26, wherein the core fucosylation is determined as (amount of fucosylated glycan)/(amount of total glycan).
29. The antibody of any one of any of the preceding items, wherein the core fucosylation is determined using capillary electrophoresis or HLPC, such as hydrophilic interaction liquid chromatography HPLC (HILIC-HPLC), electrospray ionization mass spectrometry (ESI-MS), hydrophilic interaction liquid chromatography equipped with fluorescence detection and tandem mass spectrometry (HILIC-FLD-MS/MS).
30. The antibody of any one of the preceding items, wherein the antibody is covalently linked to a ligand selected from the group consisting of chromophores, fluorophores, radiotracers, drugs, peptides, proteins, enzymes,
single or double chained oligonucleotides and their analogs, biotin and therapeutic moieties, such as cytotoxins, chemotherapeutic drugs, cytokines, and radioisotopes.
31 . The antibody of any one of the preceding items, wherein the antibody is humanized.
32. The antibody of any one of the preceding items, wherein the antibody is of human subclass lgG1 , preferably wherein the lgG1 has a standard N-Glycan glycosylation pattern of a human I gG 1 except for the reduction in fucosylation.
33. A method of producing an anti-OX40L antibody or antibody derivative, comprising expressing polynucleotides encoding polypeptide of SEQ ID NO: 13 and 14 in a cell, wherein the cell is exposed to culture medium comprising an inhibitor of glucosidases I and II, and subsequently purifying the antibody.
34. The method according to item 33, wherein the cell is a mammalian cell.
35. The method of any one of items 33-34, wherein the inhibitor is selected from the group comprising: castanospermine, deoxymannojirimycin and australine.
36. A method of producing an anti-OX40L antibody or antibody derivative, comprising expressing polynucleotides encoding polypeptide of SEQ ID NO: 13 and SEQ ID NO: 14 in a cell, wherein the cell is exposed to culture medium comprising a fucose analog, and subsequently purifying the antibody.
37. The method according to item 36, wherein the cell is a mammalian cell.
38. The method of any one of items 36 to 37, wherein the fucose analog is an inhibitor of fucokinase, GDP-fucose-pyrophosphorylase, fucosyltransferase (FUT), GDP-mannose 4,6-dehydratase, GDP-fucose synthetase, and/or a fucose transporter, such as GDP-fucose transporter.
39. The method of item 36 to 37, wherein the fucose analog is an inhibitor of a fucosyltransferase (FUT), such as 1 ,6-fucosyltransferase (FUT8).
The method of any one of items 36 to 37, wherein the fucose analog is selected from the group comprising of L-fucose, 2-fluro-fucose, 2- fluoro peracetylated fucose (2FF), 5-alkynyl-fucose, alkynyl fucose monoacetate, alkynyl fucose triacetate, alkynyl fucose diacetate and 5-alkynyl-fucose peracetate. The method of any one of items 36 to 37, wherein the fucose analog is 2- fluoro peracetylated fucose (2FF). The method of any one of items 36 to 41 , wherein the culture medium is supplemented with the fucose analog to maintain an effective concentration thereof. A method of producing an anti-OX40L antibody or antibody derivative, comprising expressing polynucleotides encoding polypeptide of SEQ ID NO: 13 and SEQ ID NO: 14 in a cell that is genetically modified to prevent core fucosylation. The method according to item 43, wherein the cell does not encode a functional fucosyltransferase. The method according to item 43, wherein the cell encodes a functional (3-1,4- N-acetylglucosaminyltransferase (GnTIII), GDP-6-deoxy-D-lyxo-4-hexulose reductase (RMD) and/or a Golgi a-mannosidase II (Manll). The method according to any one of items 43 to45, wherein the cell is a mammalian cell. The method according to any one of items 43 to 46, wherein the cell is a recombinant cell. The method according to any one of items 43 to47, wherein the cell is selected from the following cells: CHO (e.g., CHO K1 , DXB-11 CHO, Veggie-CHO), COS (e.g., COS-7), Syrian hamster, rat myleloma, mouse myeloma (e.g., SP2/0, NSO), retinal cell, Vero, CV1 , kidney (e.g., HEK293, 293 EBNA, MSR
293, MDCK, HaK, BHK, BHK21), HeLa, HepG2, WI38, MRC 5, Colo205, HB 8065, HL-60, Jurkat, Daudi, A431 (epidermal), CV-1 , U937, 3T3, L cell, C127 cell, MMT 060562, Sertoli cell, BRL 3A cell, HT1080 cell, a human myeloma cell, tumor cell, a human lymphoma cell (e.g., a Namalwa cell) and a cell line derived from an aforementioned cell.
49. The method according to any one of items 43 to 48, wherein fucosylation of the antibody or antibody derivative is restored and/or titrated to a desired level via exposure to fucose, wherein the cell is exposed to a culture medium comprising fucose.
50. The method according to item 49, wherein increasing concentrations of fucose are added to the culture media, such as up to 1mM.
51. A method of producing an anti-OX40L antibody or antibody derivative, comprising expressing polynucleotides encoding polypeptide of SEQ ID NO: 13 and SEQ ID NO:14 in a cell, and subsequently purifying and optionally folding the antibody.
52. The method according to item 51 , wherein the cell is selected from the group consisting of mammalian cells, bacterial cells, yeast cells and plant cells.
53. The method of any one of items 33 to 52, wherein the antibody is an IgG 1.
54. The method of any one of items 33 to 53, wherein the antibody is an intact antibody.
55. The method of any one of items 33 to 54, wherein the antibody comprises a heavy and a light chain variable region and an Fc region.
56. The method of any one of items 33 to 55, wherein the antibody derivative comprises an antibody Fc region and a ligand binding domain of a nonimmunoglobulin protein.
The method according to any one of the items 33 to 56, wherein the method comprises an additional step of isolating the antibody. The method according to any one of the items 33 to 57, wherein the method comprises an additional step of purifying the antibody. A method of testing the potency of a defucosylated anti-OX40L antibody or antibody derivative, comprising testing the antibody in a T-cell activation assay and verifying that the T-cell activation is reduced compared to a fucosylated anti-OX40L antibody. A method of testing the potency of a defucosylated anti-OX40L antibody or antibody derivative according to any of the preceding items, comprising testing the antibody in a T-cell activation assay and verifying that the T-cell activation is reduced compared to a fucosylated anti-OX40L antibody. The method according to any one of items 59-60, wherein the T-cell activation is reduced by at least 5%, such as at least 10%, such as at least 15%, such as at least 25%, such as at least 30%. A pharmaceutical composition comprising an antibody of any one of items 1 to 32 and at least one pharmaceutically acceptable excipient. The antibody or antibody derivative according to any one of the preceding items, for use in the prophylaxis and/or treatment of at least one disease or disorder that is associated with OX40L. The antibody or antibody derivative according to any one of the preceding items, for use in the prophylaxis and/or treatment of inflammatory disorders. The use of an antibody or antibody derivative as defined in any of items 1 to 32 for use in the preparation of a medicament for the prophylaxis and treatment of inflammatory disorders.
66. The antibody or antibody derivative according to any one of the preceding items, for use in the prophylaxis and/or treatment of autoimmune diseases.
67. The use of an antibody or antibody derivative as defined in any of items 1 to 32 for use in the preparation of a medicament for the prophylaxis and treatment of autoimmune diseases.
68. The antibody or antibody derivative according to items 63-67, wherein the disease is selected from the group consisting of: Graft-vs-host-disease, allograft rejection, asthma, Systemic lupus, arthritis, inflammatory bowel disease, ulcerative colitis, Crohn’s disease, diabetes, atopic dermatitis, psoriasis, hidradenitis suppurativa, immunoglobulin A nephropathy, Hashimoto disease, Graves’ disease, chronic rhinosinusitis and multiple sclerosis. 69. A kit comprising the antibody or antibody derivative of any one of items 1 to 32.
Claims
1. An antibody comprising a heavy chain of SEQ ID NO: 14 and a light chain comprising a VL sequence of SEQ ID NO: 1 , wherein core fucosylation of the heavy chain is below 80%.
2. The antibody of claim 1, wherein the light chain has the amino acid sequence of SEQ ID NO: 13.
3. The antibody of claim 1, wherein the core fucosylation is below 70%, such as below 60%, for example below 50%, such as below 40%, for example below 30%, such as below 20% of the total glycans.
4. The antibody of any one of the preceding claims, wherein the antibody is glycosylated with at the least 2 kDa of total glycans.
5. The antibody of any one of the preceding claims, wherein the antibody is glycosylated with at the most 3 kDa of total glycans.
6. The antibody of any one of the preceding claims, wherein the antibody is glycosylated with at the least 2kDa of total glycans and at the most 3 kDa of total glycans.
7. The antibody of any one of the preceding claims, wherein the antibody glycans comprise between 50% and 80% of glycan GO.
8. The antibody of any one of the preceding claims, wherein the antibody glycans comprise between 50% and 80% of glycan GO, such as between 55 and 80%, such as between 60% and 80%, such as between 65% and 80%, such as between 70% and 80%, such as between 75% and 80%, such as between 50% and 75%, such as between 50% and 70%, such as between 50% and 65%, such as between 50% and 60%, such as between 50% and 55%.
9. The antibody of any one of the preceding claims, wherein the core fucosylation is determined by quantitative analysis of glycans as (Area with fucosylated glycan)/(total glycan area).
10. The antibody of any one of the preceding claims, wherein the core fucosylation is determined as (amount of fucosylated glycan)/(amount of total glycan).
11. A method of producing an anti-OX40L antibody or antibody derivative, comprising expressing polynucleotides encoding polypeptide of SEQ ID NO: 13 and 14 in a cell, wherein the cell is exposed to culture medium comprising an inhibitor of glucosidases I and II, and subsequently purifying the antibody.
12. A method of producing an anti-OX40L antibody or antibody derivative, comprising expressing polynucleotides encoding polypeptide of SEQ ID NO: 13 and 14 in a cell, wherein the cell is exposed to culture medium comprising a fucose analog, and subsequently purifying the antibody.
13. The method of claim 12, wherein the fucose analog is an inhibitor of fucokinase, GDP-fucose-pyrophosphorylase, fucosyltransferase (FUT), GDP-mannose 4,6- dehydratase, GDP-fucose synthetase, and/or a fucose transporter, such as GDP-fucose transporter.
14. The method of claim 12, wherein the fucose analog is an inhibitor of a fucosyltransferase (FUT), such as 1,6-fucosyltransferase (FUT8).
15. A method of producing an anti-OX40L antibody or antibody derivative, comprising expressing polynucleotides encoding polypeptide of SEQ ID NO: 13 and SEQ ID NO: 14 in a cell that is genetically modified to prevent core fucosylation.
16. The method according to claim 15, wherein the cell is selected from the following cells: CHO (e.g., CHO K1 , DXB-11 CHO, Veggie-CHO), COS (e.g., COS-7), Syrian hamster, rat myleloma, mouse myeloma (e.g., SP2/0, NSO), retinal cell, Vero, CV1 , kidney (e.g., HEK293, 293 EBNA, MSR 293, MDCK, HaK, BHK, BHK21), HeLa, HepG2, WI38, MRC 5, Colo205, HB 8065, HL-60,
Jurkat, Daudi, A431 (epidermal), CV-1 , U937, 3T3, L cell, C127 cell, MMT 060562, Sertoli cell, BRL 3A cell, HT1080 cell, a human myeloma cell, tumor cell, a human lymphoma cell (e.g., a Namalwa cell) and a cell line derived from an aforementioned cell.
17. The method according to any one of claims 15 or 16, wherein fucosylation of the antibody or antibody derivative is restored and/or titrated to a desired level via exposure to fucose, wherein the cell is exposed to a culture medium comprising fucose.
18. A method of testing the potency of a defucosylated anti-OX40L antibody or antibody derivative, comprising testing the antibody in a T-cell activation assay and verifying that the T-cell activation is reduced compared to a fucosylated anti-OX40L antibody.
19. The antibody or antibody derivative according to any one of the preceding claims, for use in the prophylaxis and/or treatment of inflammatory disorders.
20. The antibody or antibody derivative according to any one of the preceding claims, for use in the prophylaxis and/or treatment of autoimmune diseases.
21. The antibody or antibody derivative according to claims 19-20, wherein the disease is selected from the group consisting of: Graft-vs-host-disease, allograft rejection, asthma, Systemic lupus, arthritis, inflammatory bowel disease, ulcerative colitis, Crohn’s disease, diabetes, atopic dermatitis, psoriasis, hidradenitis suppurativa, immunoglobulin A nephropathy, Hashimoto disease, Graves’ disease, chronic rhinosinusitis and multiple sclerosis.
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US202363597161P | 2023-11-08 | 2023-11-08 | |
| US63/597,161 | 2023-11-08 | ||
| EP23210318.4 | 2023-11-16 | ||
| EP23210318 | 2023-11-16 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2025099280A1 true WO2025099280A1 (en) | 2025-05-15 |
Family
ID=93378107
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2024/081745 Pending WO2025099280A1 (en) | 2023-11-08 | 2024-11-08 | Improved anti-ox40l antibodies |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2025099280A1 (en) |
Citations (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20040110282A1 (en) | 2002-04-09 | 2004-06-10 | Kyowa Hakko Kogyo Co., Ltd. | Cells in which activity of the protein involved in transportation of GDP-fucose is reduced or lost |
| US7214775B2 (en) | 1999-04-09 | 2007-05-08 | Kyowa Hakko Kogyo Co., Ltd. | Method of modulating the activity of functional immune molecules |
| US7425466B2 (en) | 2004-04-26 | 2008-09-16 | Samsung Electronics Co., Ltd | Wire bonding system and method of use |
| US7501496B1 (en) | 2004-09-17 | 2009-03-10 | Roche Palo Alto Llc | Anti-OX40L antibodies |
| WO2015132580A1 (en) | 2014-03-04 | 2015-09-11 | Kymab Limited | Antibodies, uses & methods |
| US9139653B1 (en) | 2015-04-30 | 2015-09-22 | Kymab Limited | Anti-human OX40L antibodies and methods of treatment |
| WO2018083248A1 (en) | 2016-11-03 | 2018-05-11 | Kymab Limited | Antibodies, combinations comprising antibodies, biomarkers, uses & methods |
| WO2020086776A1 (en) * | 2018-10-23 | 2020-04-30 | Magenta Therapeutics, Inc. | Fc silenced antibody drug conjugates (adcs) and uses thereof |
| US20220305038A1 (en) * | 2019-08-16 | 2022-09-29 | H. Lee Moffitt Cancer Center And Research Institute, Inc. | Fucosylation and immune modulation in cancer |
-
2024
- 2024-11-08 WO PCT/EP2024/081745 patent/WO2025099280A1/en active Pending
Patent Citations (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7214775B2 (en) | 1999-04-09 | 2007-05-08 | Kyowa Hakko Kogyo Co., Ltd. | Method of modulating the activity of functional immune molecules |
| US20040110282A1 (en) | 2002-04-09 | 2004-06-10 | Kyowa Hakko Kogyo Co., Ltd. | Cells in which activity of the protein involved in transportation of GDP-fucose is reduced or lost |
| US7425466B2 (en) | 2004-04-26 | 2008-09-16 | Samsung Electronics Co., Ltd | Wire bonding system and method of use |
| US7501496B1 (en) | 2004-09-17 | 2009-03-10 | Roche Palo Alto Llc | Anti-OX40L antibodies |
| WO2015132580A1 (en) | 2014-03-04 | 2015-09-11 | Kymab Limited | Antibodies, uses & methods |
| US9139653B1 (en) | 2015-04-30 | 2015-09-22 | Kymab Limited | Anti-human OX40L antibodies and methods of treatment |
| WO2018083248A1 (en) | 2016-11-03 | 2018-05-11 | Kymab Limited | Antibodies, combinations comprising antibodies, biomarkers, uses & methods |
| WO2020086776A1 (en) * | 2018-10-23 | 2020-04-30 | Magenta Therapeutics, Inc. | Fc silenced antibody drug conjugates (adcs) and uses thereof |
| US20220305038A1 (en) * | 2019-08-16 | 2022-09-29 | H. Lee Moffitt Cancer Center And Research Institute, Inc. | Fucosylation and immune modulation in cancer |
Non-Patent Citations (27)
| Title |
|---|
| "Antibody expression and production", 16 May 2011, SPRINGER, article MAUREEN SPEARMAN ET AL: "Chapter 12 : The role of glycosylation in therapeutic antibodies", pages: 251 - 292, XP055240498 * |
| "Biochemical Experimentation Methods", vol. 23, 1989, JAPAN SCIENTIFIC SOCIETIES PRESS |
| ALMAHAYNI KSPIEKERMANN MFIORE AYU GPEDRAM KMÖCKL L: "Small molecule inhibitors of mammalian glycosylation", MATRIX BIOL PLUS., vol. 16, 16 March 2022 (2022-03-16), pages 100108 |
| CHATENOUD LTHERVET EPRIMO JBACH JF: "Anti-CD3 antibody induces long-term remission of overt autoimmunity in non obese diabetic mice", PROC NATL ACAD SCI U S A., vol. 91, no. 1, 4 January 1994 (1994-01-04), pages 123 - 7 |
| COMPAAN DMHYMOWITZ SG: "The crystal structure of the costimulatory OX40-OX40L complex", STRUCTURE, vol. 14, no. 8, August 2006 (2006-08-01), pages 1321 - 30, XP025134045, DOI: 10.1016/j.str.2006.06.015 |
| EDWARDS ELIVANOS MKRUEGER ADELL AHASLAM SMMARK SMALES CBRACEWELL DG: "Strategies to control therapeutic antibody glycosylation during bioprocessing: Synthesis and separation", BIOTECHNOL BIOENG., vol. 119, no. 6, 28 February 2022 (2022-02-28), pages 1343 - 1358, XP093119873, DOI: 10.1002/bit.28066 |
| ERRATUM, MATRIX BIOL PLUS., vol. 16, 13 December 2022 (2022-12-13), pages 100126 |
| GUO JTU HJING LMCCARTHY DATOUF F: "USP Reference Standard Monoclonal Antibodies: Tools to Verify Glycan Structure", PHARMACEUTICALS (BASEL)., vol. 15, no. 3, 5 March 2022 (2022-03-05), pages 315 |
| HAILE YADEGOKE ALARIBI BLIN JANDERSON CC: "Anti-CD52 blocks EAE independent of PD-1 signals and promotes repopulation dominated by double-negative T cells and newly generated T and B cells", EUR J IMMUNOL., vol. 50, no. 9, 27 May 2020 (2020-05-27), pages 1362 - 1373 |
| J LIQ CHROMATOGR, vol. 6, 1983, pages 1557 |
| KANDA YIMAI-NISHIYA HKUNI-KAMOCHI RMORI KINOUE MKITAJIMA-MIYAMA KOKAZAKI ALIDA SSHITARA KSATOH M: "Establishment of a GDP-mannose 4,6-dehydratase (GMD) knockout host cell line: a new strategy for generating completely non-fucosylated recombinant therapeutics", J BIOTECHNOL., vol. 130, no. 3, 6 May 2007 (2007-05-06), pages 300 - 10, XP022119693, DOI: 10.1016/j.jbiotec.2007.04.025 |
| LI WZHU ZCHEN WFENG YDIMITROV DS: "Crystallizable Fragment Glycoengineering for Therapeutic Antibodies Development", FRONT IMMUNOL., vol. 8, 13 November 2017 (2017-11-13), pages 1554 |
| LUO, S.ZHANG, B.: "Benchmark Glycan Profile of Therapeutic Monoclonal Antibodies Produced by Mammalian Cell Expression Systems", PHARM RES, 2023, Retrieved from the Internet <URL:https://doi.org/10.1007/s11095-023-03628-4> |
| LUOZHANG: "Benchmark glycan profile of therapeutic monoclonal antibodies produced by Mammalian cell expression systems", PHARMA. RES., 2023 |
| MIMURA YUSUKE ET AL: "Enhanced Immunomodulatory Effect of Intravenous Immunoglobulin by Fc Galactosylation and Nonfucosylation", FRONTIERS IN IMMUNOLOGY, vol. 13, 28 January 2022 (2022-01-28), Lausanne, CH, XP093145776, ISSN: 1664-3224, DOI: 10.3389/fimmu.2022.818382 * |
| MISHRA NSPEARMAN MDONALD LPERREAULT HBUTLER M: "Comparison of two glycoengineering strategies to control the fucosylation of a monoclonal antibody", J BIOTECHNOL., vol. 324, 20 February 2020 (2020-02-20), pages 100015, XP086638446, DOI: 10.1016/j.btecx.2020.100015 |
| REUSCH DTEJADA ML: "Fc glycans of therapeutic antibodies as critical quality attributes", GLYCOBIOLOGY, vol. 25, no. 12, 11 August 2015 (2015-08-11), pages 1325 - 34, XP055425665, DOI: 10.1093/glycob/cwv065 |
| REUSCHTAJEDA: "Fc glycans of therapeutic antibodies as critical quality attributes", GLYCOBIOLOGY, vol. 25, December 2015 (2015-12-01), pages 1325 - 1334 |
| RILLAHAN, C.ANTONOPOULOS, A.LEFORT, C. ET AL.: "Global metabolic inhibitors of sialyl- and fucosyltransferases remodel the glycome", NAT CHEM BIOL, vol. 8, 2012, pages 661 - 668, XP055059726, Retrieved from the Internet <URL:https://doi.orq/10.1038/nchembio.999> DOI: 10.1038/nchembio.999 |
| SAMBROOKRUSSELL: "Molecular Cloning: A Laboratory Manual", 2001, COLD SPRING HARBOR LABORATORY PRESS |
| TRIPATHI TRIVENDRA ET AL: "Central Roles of OX40L-OX40 Interaction in the Induction and Progression of Human T Cell-Driven Acute Graft-versus-Host Disease", IMMUNOHORIZONS, vol. 3, no. 3, 1 March 2019 (2019-03-01), pages 110 - 120, XP093145341, ISSN: 2573-7732, Retrieved from the Internet <URL:https://watermark.silverchair.com/ih1900001.pdf?token=AQECAHi208BE49Ooan9kkhW_Ercy7Dm3ZL_9Cf3qfKAc485ysgAABegwggXkBgkqhkiG9w0BBwagggXVMIIF0QIBADCCBcoGCSqGSIb3DQEHATAeBglghkgBZQMEAS4wEQQMnN7q9A7Ca0476StBAgEQgIIFm1_CDFaKdsojcIrveV6X910iH6CsCRDVmEd81s-6oNw2Y8VO15C935v0O1tpmJA6ZD0_IBkXaG7isGCaTw8vkZVbFX> DOI: 10.4049/immunohorizons.1900001 * |
| TRIPATHI TYIN WXUE YZURAWSKI SFUJITA HHANABUCHI SLIU YJOH SJOO H: "Central Roles of OX40L-OX40 Interaction in the Induction and Progression of Human T Cell-Driven Acute Graft-versus-Host Disease", IMMUNOHORIZONS, vol. 3, no. 3, March 2019 (2019-03-01), pages 110 - 120, XP093145341, DOI: 10.4049/immunohorizons.1900001 |
| WACKER CBERGER CNGIRARD PMEIER R, EUR J PHARM BIOPHARM., vol. 79, no. 3, 2 July 2011 (2011-07-02), pages 503 - 7 |
| WEINBERG, A. D., TRENDS IMMUNOL., vol. 23, 2002, pages 102 - 109 |
| YAMANE-OHNUKI NKINOSHITA SINOUE-URAKUBO MKUSUNOKI MLIDA SNAKANO RWAKITANI MNIWA RSAKURADA MUCHIDA K: "Establishment of FUT8 knockout Chinese hamster ovary cells: an ideal host cell line for producing completely defucosylated antibodies with enhanced antibody-dependent cellular cytotoxicity", BIOTECHNOL BIOENG., vol. 87, no. 5, 5 September 2004 (2004-09-05), pages 614 - 22, XP002984450, DOI: 10.1002/bit.20151 |
| YAMANE-OHNUKI NSATOH M: "Production of therapeutic antibodies with controlled fucosylation", MABS, vol. 1, no. 3, 28 May 2009 (2009-05-28), pages 230 - 6, XP002731447, DOI: 10.4161/mabs.1.3.8328 |
| YOSHIOKA TNAKAJIMA AAKIBA HISHIWATA TASANO GYOSHINO SYAGITA HOKUMURA K: "Contribution of OX40/OX40 ligand interaction to the pathogenesis of rheumatoid arthritis", EUR J IMMUNOL., vol. 30, no. 10, October 2000 (2000-10-01), pages 2815 - 23, XP002349478, DOI: 10.1002/1521-4141(200010)30:10<2815::AID-IMMU2815>3.0.CO;2-# |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN113583131B (en) | Anti-TIGIT antibodies | |
| TWI777924B (en) | Anti-bcma antibodies, bispecific antigen binding molecules that bind bcma and cd3, and uses thereof | |
| DK1896071T3 (en) | Methods and compositions with increased therapeutic activity | |
| EP3558365B1 (en) | Anti-neuropilin antigen-binding proteins and methods of use thereof | |
| US20190270826A1 (en) | Anti-il1rap antibodies, bispecific antigen binding molecules that bind il1rap and cd3, and uses thereof | |
| EP2374883B1 (en) | Anti-cd4 antibody | |
| TW202302636A (en) | Trispecific antibody targeting bcma, gprc5d, and cd3 | |
| WO2020037154A1 (en) | Anti-il1rap antibodies and methods of use thereof | |
| US11970541B2 (en) | Anti-CD20 antibody | |
| AU2019218959B2 (en) | Bispecific antigen-binding molecules and methods of use | |
| EP3831849A1 (en) | Bispecific antibodies against ceacam5 and cd47 | |
| WO2018155692A1 (en) | Anti-hla-dq2.5/8 antibody and its use for the treatment of celiac disease | |
| AU2024209001A1 (en) | Multispecific antibodies and uses thereof | |
| JP7216806B2 (en) | Afucosylated antibody and method for producing same | |
| WO2025099280A1 (en) | Improved anti-ox40l antibodies | |
| EP4286414A1 (en) | Anti-cd20 antibody compositions | |
| EP4286413A1 (en) | Anti-cd20 antibody compositions | |
| EP4286010A1 (en) | Anti-cd20 antibody compositions | |
| EP3438261A1 (en) | Autoimmune disease therapeutic agent containing, as active ingredient, antibody that binds to haptoglobin in blood to form polyvalent immune complex | |
| WO2022090801A2 (en) | Pvrl2 and/or pvrig as biomarkers for treatment | |
| HK40103541A (en) | Anti-cd20 antibody compositions | |
| HK40103538A (en) | Anti-cd20 antibody compositions | |
| HK40104044A (en) | Anti-cd20 antibody compositions | |
| EP4590715A1 (en) | Anti-ccr8 monoclonal antibodies and their therapeutic use | |
| KR20250111754A (en) | Anti-CCR8 monoclonal antibodies and therapeutic uses thereof |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 24801309 Country of ref document: EP Kind code of ref document: A1 |