WO2025094144A1 - Controlling loss of kidney function - Google Patents
Controlling loss of kidney function Download PDFInfo
- Publication number
- WO2025094144A1 WO2025094144A1 PCT/IB2024/060834 IB2024060834W WO2025094144A1 WO 2025094144 A1 WO2025094144 A1 WO 2025094144A1 IB 2024060834 W IB2024060834 W IB 2024060834W WO 2025094144 A1 WO2025094144 A1 WO 2025094144A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- ipth
- patient
- egfr
- serum
- target
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/59—Compounds containing 9, 10- seco- cyclopenta[a]hydrophenanthrene ring systems
- A61K31/593—9,10-Secocholestane derivatives, e.g. cholecalciferol, i.e. vitamin D3
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/59—Compounds containing 9, 10- seco- cyclopenta[a]hydrophenanthrene ring systems
- A61K31/592—9,10-Secoergostane derivatives, e.g. ergocalciferol, i.e. vitamin D2
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P13/00—Drugs for disorders of the urinary system
- A61P13/12—Drugs for disorders of the urinary system of the kidneys
Definitions
- the disclosure relates generally to method of preventing or mitigating loss of kidney function.
- CKD chronic kidney disease
- SHPT secondary hyperparathyroidism
- iPTH intact parathyroid hormone
- FGF23 serum phosphorus and fibroblast growth factor 23
- VDI vitamin D insufficiency
- SHPT can lead to bone disease, increased fracture rates, vascular calcification, morbidity and mortality.
- Reduced serum levels of 1,25-dihydroxy vitamin D cause increased, and ultimately excessive, secretion of iPTH by direct and indirect mechanisms.
- the resulting hyperparathyroidism leads to markedly increased bone turnover and its sequela of mineral and bone disorder (MBD), which may include a variety of other diseases, such as, renal osteodystrophy, osteitis fibrosa cystica, osteomalacia, osteoporosis, extraskeletal calcification and related disorders, e.g., bone pain, periarticular inflammation and Mockerberg's sclerosis.
- Reduced serum levels of 1,25-dihydroxy vitamin D also can cause muscle weakness and growth retardation with skeletal deformities (most often seen in pediatric patients).
- Vitamin D compounds have traditionally been administered in immediate-release formulations. Formulations for delivery of active vitamin D, analogs thereof, and prohormones thereof have been disclosed, including some extended-release dosage forms. Some modified-release or extended-release dosage forms of vitamin D compounds have been described, e.g., in wax matrix form.
- One such formulation is marketed in the United States (U.S.) under the brand name RAYALDEE® (calcif ediol), a product which is approved to treat SHPT in patients with stage 3 or 4 CKD and VDI.
- RAYALDEE® a wax based extended-release formulation of 25-hydroxy vitamin D3 . See U.S. Patent Application Publication Nos.
- the disclosure provides a method of preventing or mitigating loss of kidney function in a CKD patient having SHPT, comprising reducing the patient’s iPTH level to or below a target threshold and maintaining the iPTH level at or below the target level for a duration of time, thereby mitigating loss of kidney function as measured by estimated glomerular filtration rate (eGFR).
- eGFR estimated glomerular filtration rate
- the disclosure also provides iPTH reducing compounds for use in a method of preventing or mitigating loss of kidney function in a CKD patient having SHPT, comprising reducing the patient’ s iPTH level to or below a target threshold and maintaining the iPTH level at or below the target level for a duration of time, and thereby mitigating loss of kidney function as measured by eGFR.
- the iPTH reducing compounds can be active vitamin hormones, 25-hydroxy vitamin D3, 25-hydroxy vitamin D3, 25-hydroxyvitaminD4, or metabolites of any one of the foregoing.
- the disclosure also provides an iPTH reducing composition for use in said method of preventing or mitigating loss of kidney function in a CKD patient having SHPT.
- the iPTH reducing composition comprises an active vitamin hormone, 25-hydroxy vitamin D3, 25-hydroxy vitamin D3, 25 -hydroxy vitamin D4,or metabolites of any one of the foregoing.
- the composition can be an extended-released composition, or it can be administered in a way to mimic extended-release of the compound.
- Figure 1 shows 26-wk Changes in Mean Serum Total 25-hydroxy vitamin D, Serum Total 1,25-dihydroxy vitamin D and Plasma iPTH by baseline (BL) eGFR Group for the patients of Example 1 who were treated with extended-release calcifediol (ERC) for 26 weeks.
- Figure 2 shows BL through End-of-Treatment (EOT) Mean iPTH and 1,25- dihydroxy vitamin D by BL eGFR Group for the patients of Example 1 who were treated with ERC for 26 weeks.
- EOT End-of-Treatment
- Figure 3 shows 52-wk Changes in Mean Serum Total 25-hydroxy vitamin D by BL eGFR for the patients of Example 1 who were treated with ERC for 52 weeks.
- Figure 4 shows 52-wk Changes in Mean Plasma iPTH by BL eGFR for the patients of Example 1 who were treated with ERC for 52 weeks.
- Figure 5 shows 52-wk Changes in Mean eGFR by BL eGFR for the patients of Example 1 who were treated with ERC for 52 weeks.
- Figure 6 shows Reductions in Mean PTH as a Function of Mean Serum 25- hydroxy vitamin D and BL eGFR group for the patients of Example 1 who were treated with ERC for 52 weeks.
- Figure 7 shows Increases in Mean Serum Total 1,25-dihydroxy vitamin D as a function of Serum Total 25 -hydroxy vitamin D and BL eGFR group for the patients of Example 1 who were treated with ERC for 52 weeks.
- Figure 8 shows Mean iPTH by Duration of iPTH Control for the patients of Example 1 who were treated with ERC for 52 weeks.
- Figure 9 shows Mean 25-hydroxy vitamin D by Duration of iPTH Control for the patients of Example 1 who were treated with ERC for 52 weeks.
- Figure 10 shows Mean 25-hydroxy vitamin D3 during the first 26 weeks of treatment by Duration of iPTH Control for the patients of Example 1 who were treated with ERC for 52 weeks.
- Figure 11 shows Mean 1,25-dihydroxy vitamin D by Duration of iPTH Control for the patients of Example 1 who were treated with ERC for 52 weeks.
- Figure 12 shows Mean 24,25-dihydroxyvitamin D3 by Duration of iPTH Control for the patients of Example 1 who were treated with ERC for 52 weeks.
- Figure 13 shows Mean 24,25-dihydroxyvitamin D3/25-hydroxy vitamin D3 Ratio during the first 26 weeks of treatment by Duration of iPTH Control for the patients of Example 1 who were treated with ERC for 52 weeks.
- Figure 14 shows Mean Change in eGFR from BL by Duration of iPTH Control for the patients of Example 1 who were treated with ERC for 52 weeks.
- Figure 15 shows Mean Percent Change in eGFR from BL.
- the duration of iPTH control periods are plotted left to right from 4 or 5 quarters to 0 quarters.
- Figure 17 shows the Prevalence of Increased eGFR at End of Treatment Compared to Duration of iPTH Control for the patients of Example 1 who were treated with ERC for 52 weeks. *Significantly different from 0 Quarters by Chi-test, p ⁇ 0.05.
- Figure 18 shows Mean Serum 24,25-dihydroxyvitamin D3 as a Function of Serum 25-hydroxy vitamin D3 and CKD Stage in patients treated with ERC.
- Figure 19 shows Mean Serum 1,25-dihydroxy vitamin D3 or Total 1,25- dihydroxyvitamin D as a Function of Serum 25 -hydroxy vitamin D3 or Total 25- hydroxyvitamin D by BL eGFR in patients treated with ERC.
- Figure 20 shows the Rate of Increase in Serum 24,25-dihydroxyvitamin D3 compared to Serum 25-hydroxy vitamin D3 by Baseline eGFR in patients treated with ERC. *Slope of regression line in Figure 18.
- Figure 21 A shows the time course of mean (+SE) serum total 25-hydroxy vitamin D for 126 study participants, 51 participants who achieved sustained iPTH reductions of > 30% during the last two quarters of the one-year treatment period, and 75 participants who did not achieve sustained iPTH reductions of > 30% during the last two quarters of the one-year treatment period.
- Figure 2 IB shows the time course mean of iPTH for the 126 study participates of Figure 21 A.
- Figure 22A shows an association between duration of plasma iPTH reduction and changes in eGFR. Changes in mean eGFR from BL are shown for all 126 study participants, for 51 participants who achieved sustained iPTH reductions of > 30% during the last two quarters of the one-year treatment period, and 75 participants who did not achieve sustained iPTH reductions of > 30% during the last two quarters of the one-year treatment period .
- Figure 22B shows an association between the degree of iPTH reduction at EOT and changes in eGFR.
- 25-hydroy vitamin D includes one or more hydroxy forms, such as of 25 -hydroxy vitamin D3, 25-hydroxyvitamin D2, and 25-hydroxyvitamin D4.
- 1, 25-dihydroy vitamin D includes 1,25-dihydroxy vitamin D3, 1,25 -hydroxy vitamin D2, and 1.25-hydroxyvitamin D4.
- Methods of mitigating loss of kidney function in accordance with the disclosure include administration of sufficient repeated dosages of 25-hydroxyvitamin D to a patient with SHPT in order to achieve a sustained reduction of iPTH compared to pre-treatment BL for a duration of at least about 6 months.
- rate of kidney function loss can be mitigated by maintaining reduced iPTH levels for a duration of at least about 6 months.
- mistation of the rate of kidney function loss is measured as a function of observed decreases in eGFR over time, with loss of kidney function being mitigated in a patient when the rate of decrease in eGFR is less than the rate observed over a one-year period preceding treatment initiation.
- the patient can have stage 3 or stage 4 CKD.
- the patients have SHPT.
- Sustained reduction of iPTH to or below a target threshold or target percent reduction threshold refers to maintained reduction of the average iPTH level to or below a target threshold over the target time duration or maintaining an average target percent reduction from pre-treatment BL over the target time duration. It should be understood herein that iPTH levels vary in patients and achievement of target thresholds or precent reductions is made herein with reference to average values over the target duration.
- the target threshold for iPTH can be, for example, 100 pg/mL.
- the target threshold for iPTH can further include a lower bound, optionally as low as 70 pg/mL, considered to be the upper limit of the laboratory reference range.
- the target precent reduction of iPTH from pre-treatment BL can be, for example, at least 5%, at least 10%, at least 20%, or at least 30%.
- Methods of the disclosure can include administering repeated doses of 25- hydroxyvitamin D for a duration and in an amount to achieve a target percent reduction of iPTH from pre-treatment BL and then further administration of repeated doses of 25- hydroxyvitamin D to maintain the target precent reduction of iPTH in the patient.
- Administration of repeated doses to maintain the target percent reduction can include dose amount increases where needed to maintain the target percent reduction over the target duration.
- the target time duration can be at least 6 months, or at least 9 months, or at least 10 months, or at least 11 months, or at least 12 months, or longer.
- the duration of sustained reduction of iPTH was directly correlated with mitigating loss of kidney function as measured by eGFR.
- the patient can have an initial eGFR value before the patient’s iPTH is reduced, and the method includes limiting the subsequent eGFR loss within 2.5 mL/min/1.73m 2 of the patient’s initial eGFR, or within 2.0 mL/min/1.73m 2 , or within 1.5 mL/min/1.73m 2 , or within 1.0 mE/min/1.73m 2 of the patient’s initial eGFR for at least 12 months.
- reducing the patient’ s iPTH level to at or below a target threshold or achieving a target percent reduction from pretreatment BL can include administering sufficient repeated doses of one or more of 25 -hydroxy vitamin D, an active vitamin D compound in extended-release dosing regimen or dosage form, or metabolite of any one of the foregoing to the patient.
- the compound is selected from 25-hydroxyvitamin D3.
- the 25-hdyroxvitamin D compound is formulated in an extended-release formulation.
- the amount of iPTH reducing compound can, in repeated doses, be sufficient to increase a patient’s serum total 25-hydroxyvitamin D level from pretreatment BL to at least 40 ng/mL, at least 50 ng/mL, at least 55 ng/mL, or at least 60 ng/mL, and/or an increase in serum total 1,25-dihydroxy vitamin D level of at least 5 pg/mL.
- serum total increases in 25-hydroxyvitamin D and 1,25-dihydroxyvitamin D levels can be achieved for at least 6 months, or at least 9 months, or at least 10 months, or at least 11 months, or at least 12 months, or longer.
- dose titration and increasing dosage amounts to achieve the intended increases in serum total 25-hydroxyvitamin D and/or 1, 25-dihydroxy vitamin D levels may differ in patient populations, for example, based on body mass index (BMI).
- BMI body mass index
- contemplated compounds to be administered include, but are not limited to, 25-hydroxyvitamin D3, 25-hydroxyvitamin D2, 25-hydroxyvitamin D4, la, 25- dihydroxy vitamin D3, l , 25-dihydroxy vitamin D2, la, 25-dihydroxy vitamin D4, and vitamin D analogs (including all hydroxylated, dihydroxylated and trihydroxylated forms), including
- active vitamin D and “activated vitamin D” refer to a vitamin D compound that is hydroxylated in at least the la position. Active vitamin D compounds include calcitriol, 1, 25-dihydroxy vitamin D2, alfacalcidol, doxercalciferol, 22- oxacalcitriol, and paricalcitol.
- reducing the patient’ s iPTH level to or below a target threshold can include administering sufficient repeated doses of 25-hydroxyvitamin D to the patient.
- the 25-hydroxyvitamin D can be 25-hydroxyvitamin D3.
- the repeated doses can be administered in extended-release compositions.
- the amount of administered 25- hydroxyvitamin D can, in repeated doses, be sufficient to increase the patient’s serum total
- 1.25-dihydroxy vitamin D level relative to pretreatment BL by least 5 pg/ml, at least 10 pg/ml, or at least 20 pg/ml.
- Such increase in serum total 1, 25-dihydroxy vitamin D level can be achieved for at least 6 months, or at least 9 months, or at least 10 months, or at least 11 months, or at least 12 months, or longer.
- the amount of 25-hydroxyvitamin D can, in repeated doses, be sufficient to achieve a serum total 25-hydroxyvitamin D level in a range of least 40 ng/mL to 60 ng/mL, e.g. at least 40 ng/mL, or at least 50 ng/mL, or at least 60 ng/mL or at least 80 ng/mL, or at least 100 ng/mL or at least 130 ng/mL, or at least 160 ng/mL or at least 170 ng/mL, or at least 200 ng/mL.
- Such serum total 25-hydroxyvitamin D levels can be achieved for at least 6 months, 9 months, or at least 10 months, or at least 11 months, or at least 12 months, or longer.
- a patient’s pretreatment BL serum total 25- hydroxyvitamin D can be in a range of about 10 ng/mL to about 50 ng/mL and can be increased with repeated dosing of the iPTH reducing compound to a range of about 70 ng/mL to about 200 ng/mL.
- a patient can have a pretreatment BL serum total 25-hydroxyvitamin D greater than 50 ng/mL and can still benefit from methods of the disclosure and increasing serum total 25-hydroxy vitamin D to levels to reduce iPTH levels to the target threshold or to achieve the target percent reduction for a target duration.
- reducing and sustaining reduction of the patient’s iPTH level to or below a target threshold and/or achieve a target percent reduction as compared to pre-treatment BL can include administering sufficient repeated doses of 25- hydroxyvitamin D to the patient until the target threshold or target percent reduction of the iPTH is achieved and then maintaining the dosing or increasing where needed to maintain the target threshold or target precent reduction of iPTH. Dosing can be titrated up as needed to achieve the target serum level needed to reduce the patient’ s iPTH levels to the target threshold or by the target precent reduction relative to pre-treatment BL. Dose titration can account, in part, for BMI of the patient.
- the methods of the disclosure include continued administration of repeated doses to maintain, and optionally modify, when needed, the serum levels and the reduction of iPTH for a target duration.
- the target duration can be, for example, for at least 6 months, or at least 9 months, or at least 10 months, or at least 11 months, or at least 12 months, or longer.
- repeated doses can be administered to increase serum blood 25-hydroxy vitamin D levels from pre-treatment BL to at least about 60 ng/mL, or at least 70 ng/mL, or at least 80 ng/mL, or at least 90 ng/mL, or at least 100 ng/mL.
- Maintenance of serum levels at such levels can provide for a sustained decrease of mean levels of iPTH of about >20% or > 30% in said patient. It has been observed that increasing serum 25-hydroxyvitamin D levels as described herein and maintaining a sustained decrease of iPTH can limit subsequent eGFR decline in such patients.
- Vitamin D compounds and compositions are described in the following patent publications: WO 03/018545 Al, WO 03/031400 Al, WO 03/106411 Al, WO 2004/054968, WO 2006/113990, WO 00/59513, W02007/092755, US-2009-0209501-A1, US-2009- 0082317-A1, US-2009-0176748-A1, WO 2008/134518, WO 2008/134512, WO 2009/047644, WO 2008/134523, US-2009-0311316-Al, US-2009-0233889-A1, US-2011- 0052567-Al, US-2010-0144684-A1, US-2010-0120728-A1, US-2010-0204189-A1, US- 2012-0130133-A1, US-2013-0157987-A1, US-2013-0137663-A1, US-2014-0349979-A1, WO 2014/143941, WO 2016/020508, US
- This example demonstrates the effect of controlling SHPT with extended-release calcifediol (ERC) on the rate of CKD progression.
- Subjects were stratified by CKD stage and were randomized in a 2: 1 ratio to receive a once daily 30 pg oral dose of ERC (or matching placebo) for 12 weeks at bedtime followed by an additional 14 weeks of treatment with once daily bedtime doses of either 30 or 60 pg of ERC (or placebo).
- the daily dose was increased to 60 pg at the start of week 13 if plasma iPTH remained >70 pg/mL (the upper limit of the laboratory reference range), serum total 25- hydroxy vitamin D was ⁇ 65 ng/mL (to reduce the risk of driving values above 100 ng/mL) and serum calcium was ⁇ 9.8 mg/dL.
- the sole primary efficacy end point was the proportion of subjects in the intent-to-treat (ITT) population that attained a mean decrease of >30% in plasma iPTH from pre-treatment baseline in the efficacy assessment period (EAP), defined as treatment weeks 20 through 26.
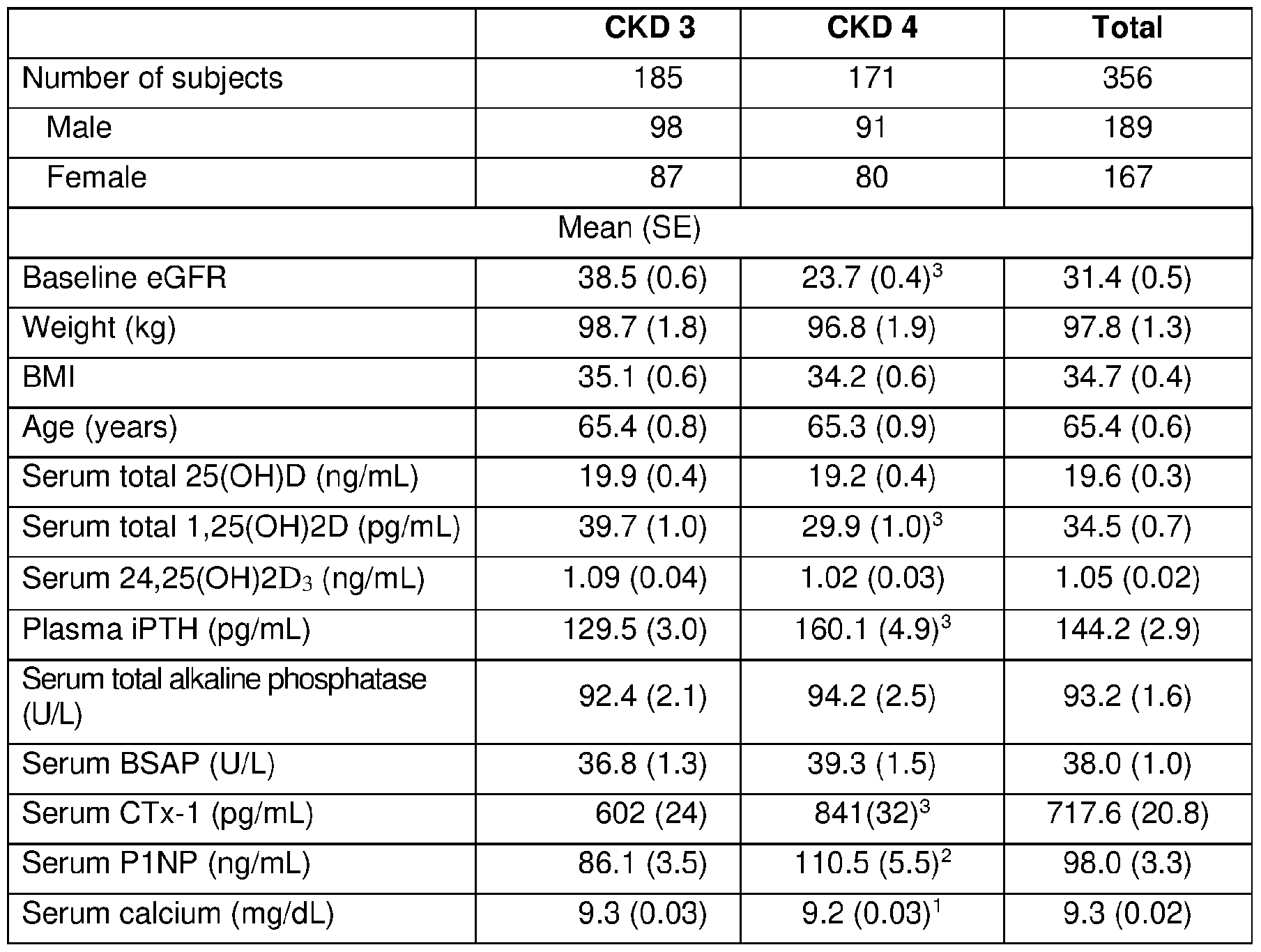
- Demographic and baseline data for the PP population are summarized in Table 1, grouped by CKD stage. Only analyses of the PP population are reported here as they yielded results that did not differ materially from those based on analyses of the ITT population, and because the number of subjects remained constant across the 26-week treatment period.
- Serum 25 -hydroxy vitamin D3 (lower limit of quantitation: 5.00 ng/mL; %CV of 0.82 to 1.84 within-run, 2.01 to 4.26% between-run) and 24,25-dihydroxyvitamin D3 (lower limit of quantitation: 0.52 ng/mL; %CV 2.18 to 4.60 within-run, 3.79 to 9.29 between-run) were determined by LC-MS (Syneos) for the purpose of calculating the vitamin D metabolite ratio (VMR), calculated as serum 24,25-dihydroxyvitamin Ds/scrum total 25-hydroxy vitamin D3*100. Other parameters were determined by standard procedures. Serum calcium values were corrected for low albumin. eGFR was measured at baseline and quarterly thereafter (5 assessments). Serum total 25-hydroxy vitamin D and plasma PTH were measured monthly. 166 patients participated in an extension of the study to 52 weeks.
- iPTH levels were considered: consistently controlled if ⁇ 100 pg/mL at 4 of the 5 assessments at pre-treatment BL and quarterly thereafter; partially controlled if ⁇ 100 pg/mL at 1-3 of the 5 assessments; uncontrolled if >100 pg/mL at all 5 assessments.
- Figure 1 shows that 26 weeks of ERC treatment effectively raised serum 25- hydroxyvitamin D and 1,25-dihydroxy vitamin D, and reduced elevated plasma iPTH in patients with stage 3 or 4 CKD, irrespective of declining BL eGFR.
- Figure 2 shows how plasma iPTH fell consistently across BL eGFR ranges during 26 weeks of treatment with ERC as a function of increasing serum 1,25-dihydroxy vitamin D.
- Figure 3 shows that increases in mean serum total 25-hydroxy vitamin D were unaffected by declining BL eGFR.
- Figure 4 shows that reductions in mean plasma iPTH were similar in all BL eGFR groups.
- Figure 5 shows that CKD progression was similar in all BL eGFR groups.
- Figure 6 shows that reductions in mean plasma iPTH were similar in all BL eGFR groups.
- Figure 7 shows that increases in mean serum 1,25-dihydroxy vitamin D were similar in all BL eGFR groups.
- Figure 8 shows that reductions in mean iPTH differed by duration of iPTH control.
- Figure 9 shows that increases in serum total 25-hydroxy vitamin D trended directly with duration of iPTH control.
- Figure 10 shows that increases in serum 25-hydroxyvitamin D3 trended directly with duration of iPTH control.
- Figure 11 shows that increases in serum total 1,25-dihydroxyvitamin D trended directly with duration of iPTH control.
- Figure 12 shows that increases in serum 24,25-dihydroxy vitamin D3 directly correlated with duration of iPTH control.
- Figure 13 shows that increases in serum 24,25-dihydroxy vitamin Ds:25- hydroxyvitamin D3 ratio directly correlated with duration of iPTH control.
- Figures 14 and 15 show that 52-wk decreases in eGFR from BL were inversely proportional to duration of iPTH control.
- Figure 16 shows that mean eGFR decline was inversely proportional to duration of iPTH control.
- Figure 17 shows that the number of subjects having an increased eGFR by EOT rose (p ⁇ 0.05) in proportion to the duration of iPTH control achieved, from 6 (no control) to 18 (consistent control).
- ERC oral efficacy endpoint
- Participants who completed either of these two RCTs could elect to join, without interruption, an open-label extension study during which ERC (but not placebo) was administered for an additional 26 weeks, with similar dose titration to 60 pg/day allowed at Week 38.
- the 126 participants who completed 52 weeks of ERC treatment were the focus of the present study.
- Measurements of 25 -hydroxy vitamin D (DiaSorin), calcium (corrected for low albumin), phosphorus and iPTH (Roche Elecsys) were obtained at BL and at biweekly or monthly intervals.
- Measurements of eGFR (MDRD equation), spot urine albumin-to- creatinine ratio (uACR), 1,25-dihydroxy vitamin D (IDS), FGF23 (Millipore), and bone turnover markers (BTM) were obtained at BL and the subsequent four quarterly intervals.
- BSALP bone-specific alkaline phosphatase
- Cx-1 collagen type 1 C-telopeptide
- P1NP procollagen type 1 N-terminal propeptide
- ALP total alkaline phosphatase
- the 126 participants were grouped into two categories depending on achievement of a sustained mean iPTH reduction of >30% (versus pre-treatment BL) at both Week 38 (mean of data obtained at Weeks 34 through 38) and Week 52 (mean of Weeks 48 through 52) of the one-year treatment period. Observed differences between these categories were evaluated by t-test or one-way analysis of variance (ANOVA) followed by Tukey’s test of the means.
- ACE angiotensin-converting enzyme
- ARBs angiotensin receptor blockers
- Serum 1,25- dihydroxyvitamin D rose with ERC treatment by 10.1 ⁇ 1.8 pg/mL (from 34.9 ⁇ 1.2; p ⁇ 0.001), serum calcium by 0.3+0.03 mg/dL (from 9.2+0.03; p ⁇ 0.001), serum phosphorus by 0.2+0.05 mg/dL (from 3.80+0.05; p ⁇ 0.01), serum FGF23 by 29.7+14.8 pg/mL (from 32.3+2.8; p ⁇ 0.05; one participant’s values rose from 57 to 1,488) and all BTMs improved (p ⁇ 0.01). Similar changes in these parameters were observed in both categories. No change during ERC treatment was observed in mean uACR for the full group of participants.
- Plasma iPTH decreased by 24.3% from 143.8 ⁇ 5.8 to 108.8 ⁇ 7.2 pg/mL, and reached concentrations that averaged 67.8 pg/mL lower in the category achieving sustained iPTH reductions of >30% (Figure 2 IB).
- Figure 2 IB sustained iPTH reductions of >30%
- the other category comprised of 75 participants, 45 achieved iPTH reductions of any magnitude that were sustained over the last 2 quarters, of which 15 and 18 achieved at least 20% and 10% reductions, respectively. Another 30 participants did not exhibit an iPTH reduction in both of the last 2 quarters.
- Example 3 - ERC overcomes impact of low eGFR on Vitamin D metabolism
- Baseline eGFR and serum total 25-hydroxy vitamin D levels of subjects included in the present analysis are listed in the table below, along with subject numbers, ERC doses, and treatment durations for each population.
- ERC reliably raised serum 25-hydroxy vitamin D3, 1,25-dihydroxy vitamin D3, and total 25-hydroxy vitamin D and 1,25-dihydroxy vitamin D irrespective of eGFR, making it an attractive alternative to calcitriol (or la-hydroxylated analogs) for treating persistently rising PTH in CKD 3-4.
- participant had eGFR 15 to ⁇ 60 mL/min/1.73 m 2 , plasma iPTH 85 to ⁇ 500 pg/mL, serum 25-hydroxyvitamin D 10 to ⁇ 30 ng/mL, corrected serum calcium (Ca) 8.4 to ⁇ 9.8, serum phosphorus (P) 2.0 to ⁇ 5.0, and absence of nephrotic range proteinuria ( ⁇ 3 mg/mg creatinine).
- Mean BL 25-hydroxyvitamin D was 67 n/gmL and rose 11-13 ng/mL in both groups (p ⁇ 0.001).
- mean BL iPTH was 143 pg/mL and fell by 35% (p ⁇ 0.01 ) with adjunctive therapy versus 4% without.
- Mean Ca and FGF23 increased with adjunctive therapy (p ⁇ 0.001) by o.5 mg/dL and 47 pg/mL respectively, versus 0.0 and 2.0 without.
- Mean P remained unchanged.
- Mean BL eGFR was 25.4 mL/min/1.73 m 2 and fell by 10% (p ⁇ 0.05) with adjunctive therapy versus 4% without.
- adjunctive active vitamin D enabled more iPTH reduction (35%) in CKD patients taking ERC for 52 weeks, it increased serum Ca by 0.5 mg/dL, FGF23 by 136%, and hastened eGFR decline by 10%. Higher doses of ERC alone are believed to be more suitable for improving iPTH control and eGFR stabilization in patients.
- compositions are described as including components or materials, it is contemplated that the compositions can also consist essentially of, or consist of, any combination of the recited components or materials, unless described otherwise.
- methods are described as including particular steps, it is contemplated that the methods can also consist essentially of, or consist of, any combination of the recited steps, unless described otherwise.
- the invention illustratively disclosed herein suitably may be practiced in the absence of any element or step which is not specifically disclosed herein.
Landscapes
- Health & Medical Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Epidemiology (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Organic Chemistry (AREA)
- Engineering & Computer Science (AREA)
- General Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Urology & Nephrology (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
Methods of preventing or mitigating loss of kidney function in a chronic kidney disease (CKD) patient having secondary hyperparathyroidism (SHPT) are disclosed.
Description
CONTROLLING LOSS OF KIDNEY FUNCTION
CROSS-REFERENCE TO RELATED APPLICATION
[0001] The benefit of priority to U.S. Provisional Patent Application No. 63/595,709 filed November 2, 2023, is hereby claimed and the disclosure is incorporated herein by reference in its entirety.
BACKGROUND
Field of the Disclosure
[0002] The disclosure relates generally to method of preventing or mitigating loss of kidney function.
Brief Description of Related Technology
[0003] Chronic kidney disease (CKD) increases the risk of secondary hyperparathyroidism (SHPT), a disorder characterized by overproduction of intact parathyroid hormone (iPTH) and hypertrophy of the parathyroid glands. SHPT is driven by low serum total 25- hydroxyvitamin D, elevation of serum phosphorus and fibroblast growth factor 23 (FGF23), and decreased serum 1,25-dihydroxy vitamin D and calcium. SHPT frequently develops as CKD progresses and can also develop in individuals with healthy kidneys due to environmental, cultural, or dietary factors, which prevent adequate vitamin D supply and promote vitamin D insufficiency (VDI).
[0004] Untreated, SHPT can lead to bone disease, increased fracture rates, vascular calcification, morbidity and mortality. Reduced serum levels of 1,25-dihydroxy vitamin D cause increased, and ultimately excessive, secretion of iPTH by direct and indirect mechanisms. The resulting hyperparathyroidism leads to markedly increased bone turnover and its sequela of mineral and bone disorder (MBD), which may include a variety of other diseases, such as, renal osteodystrophy, osteitis fibrosa cystica, osteomalacia, osteoporosis, extraskeletal calcification and related disorders, e.g., bone pain, periarticular inflammation and Mockerberg's sclerosis. Reduced serum levels of 1,25-dihydroxy vitamin D also can cause muscle weakness and growth retardation with skeletal deformities (most often seen in pediatric patients).
[0005] Vitamin D compounds have traditionally been administered in immediate-release formulations. Formulations for delivery of active vitamin D, analogs thereof, and prohormones thereof have been disclosed, including some extended-release dosage forms. Some modified-release or extended-release dosage forms of vitamin D compounds have been
described, e.g., in wax matrix form. One such formulation is marketed in the United States (U.S.) under the brand name RAYALDEE® (calcif ediol), a product which is approved to treat SHPT in patients with stage 3 or 4 CKD and VDI. The prescribing information for this drug provides that formulation for RAYALDEE® is a wax based extended-release formulation of 25-hydroxy vitamin D3 . See U.S. Patent Application Publication Nos. US 2009/311316 Al (Dec. 17, 2009), US 2009/0176748 Al (Jul. 9, 2009), US 2013/0137663 Al (May 30, 2013), US 2014/0349979 Al (Nov. 27, 2014), WO 2017/182237 Al (Oct. 26, 2017), and U.S. Patent Application No. 62/725,940 (filed Aug. 31, 2018), the disclosures of which are incorporated herein by reference in their entireties.
[0006] Clinical practice guidelines applicable to the management of CKD patients target vitamin D sufficiency. Consensus, however, is lacking on the definition of vitamin D sufficiency in CKD. In 2003, the National Kidney Foundation (NKF) defined vitamin D sufficiency as serum total 25-hydroxy vitamin D concentrations of at least 30 ng/mL and in 2011, the Endocrine Society defined it as concentrations between 30 and 100 ng/mL. The U.S. Institute of Medicine (IOM) disagreed, stating in 2011 that “practically all persons are sufficient at serum 25-hydroxy vitamin D levels of at least 20 ng/mL.”
SUMMARY
[0007] The disclosure provides a method of preventing or mitigating loss of kidney function in a CKD patient having SHPT, comprising reducing the patient’s iPTH level to or below a target threshold and maintaining the iPTH level at or below the target level for a duration of time, thereby mitigating loss of kidney function as measured by estimated glomerular filtration rate (eGFR).
[0008] The disclosure also provides iPTH reducing compounds for use in a method of preventing or mitigating loss of kidney function in a CKD patient having SHPT, comprising reducing the patient’ s iPTH level to or below a target threshold and maintaining the iPTH level at or below the target level for a duration of time, and thereby mitigating loss of kidney function as measured by eGFR. The iPTH reducing compounds can be active vitamin hormones, 25-hydroxy vitamin D3, 25-hydroxy vitamin D3, 25-hydroxyvitaminD4, or metabolites of any one of the foregoing.
[0009] The disclosure also provides an iPTH reducing composition for use in said method of preventing or mitigating loss of kidney function in a CKD patient having SHPT. The iPTH reducing composition comprises an active vitamin hormone, 25-hydroxy vitamin D3, 25-hydroxy vitamin D3, 25 -hydroxy vitamin D4,or metabolites of any one of the foregoing.
The composition can be an extended-released composition, or it can be administered in a way to mimic extended-release of the compound.
[0010] For the methods, related compounds and compositions described herein, optional features, including but not limited to components, compositional ranges thereof, substituents, conditions, and steps, are contemplated to be selected from the various aspects, embodiments, and examples provided herein.
[0011] Further aspects and advantages will be apparent to those of ordinary skill in the art from a review of the following detailed description, taken in conjunction with the drawings. While the methods, related compounds and compositions are susceptible of embodiments in various forms, the description hereafter includes specific embodiments with the understanding that the disclosure is illustrative, and is not intended to limit the invention to the specific embodiments described herein.
BRIEF DESCRIPTION OF THE DRAWINGS
[0012] For further facilitating the understanding of the present invention, twenty drawing figures are appended hereto.
Figure 1 shows 26-wk Changes in Mean Serum Total 25-hydroxy vitamin D, Serum Total 1,25-dihydroxy vitamin D and Plasma iPTH by baseline (BL) eGFR Group for the patients of Example 1 who were treated with extended-release calcifediol (ERC) for 26 weeks.
[0013] Figure 2 shows BL through End-of-Treatment (EOT) Mean iPTH and 1,25- dihydroxy vitamin D by BL eGFR Group for the patients of Example 1 who were treated with ERC for 26 weeks.
[0014] Figure 3 shows 52-wk Changes in Mean Serum Total 25-hydroxy vitamin D by BL eGFR for the patients of Example 1 who were treated with ERC for 52 weeks.
[0015] Figure 4 shows 52-wk Changes in Mean Plasma iPTH by BL eGFR for the patients of Example 1 who were treated with ERC for 52 weeks.
[0016] Figure 5 shows 52-wk Changes in Mean eGFR by BL eGFR for the patients of Example 1 who were treated with ERC for 52 weeks.
[0017] Figure 6 shows Reductions in Mean PTH as a Function of Mean Serum 25- hydroxy vitamin D and BL eGFR group for the patients of Example 1 who were treated with ERC for 52 weeks.
[0018] Figure 7 shows Increases in Mean Serum Total 1,25-dihydroxy vitamin D as a function of Serum Total 25 -hydroxy vitamin D and BL eGFR group for the patients of Example 1 who were treated with ERC for 52 weeks.
[0019] Figure 8 shows Mean iPTH by Duration of iPTH Control for the patients of Example 1 who were treated with ERC for 52 weeks.
[0020] Figure 9 shows Mean 25-hydroxy vitamin D by Duration of iPTH Control for the patients of Example 1 who were treated with ERC for 52 weeks.
[0021] Figure 10 shows Mean 25-hydroxy vitamin D3 during the first 26 weeks of treatment by Duration of iPTH Control for the patients of Example 1 who were treated with ERC for 52 weeks.
[0022] Figure 11 shows Mean 1,25-dihydroxy vitamin D by Duration of iPTH Control for the patients of Example 1 who were treated with ERC for 52 weeks.
[0023] Figure 12 shows Mean 24,25-dihydroxyvitamin D3 by Duration of iPTH Control for the patients of Example 1 who were treated with ERC for 52 weeks.
[0024] Figure 13 shows Mean 24,25-dihydroxyvitamin D3/25-hydroxy vitamin D3 Ratio during the first 26 weeks of treatment by Duration of iPTH Control for the patients of Example 1 who were treated with ERC for 52 weeks.
[0025] Figure 14 shows Mean Change in eGFR from BL by Duration of iPTH Control for the patients of Example 1 who were treated with ERC for 52 weeks.
[0026] Figure 15 shows Mean Percent Change in eGFR from BL. The duration of iPTH control periods are plotted left to right from 4 or 5 quarters to 0 quarters.
[0027] Figure 16 shows Mean eGFR change vs. Duration of iPTH <= 100 pg/mL for the patients of Example 1 who were treated with ERC for 52 weeks.
[0028] Figure 17 shows the Prevalence of Increased eGFR at End of Treatment Compared to Duration of iPTH Control for the patients of Example 1 who were treated with ERC for 52 weeks. *Significantly different from 0 Quarters by Chi-test, p < 0.05.
[0029] Figure 18 shows Mean Serum 24,25-dihydroxyvitamin D3 as a Function of Serum 25-hydroxy vitamin D3 and CKD Stage in patients treated with ERC.
[0030] Figure 19 shows Mean Serum 1,25-dihydroxy vitamin D3 or Total 1,25- dihydroxyvitamin D as a Function of Serum 25 -hydroxy vitamin D3 or Total 25- hydroxyvitamin D by BL eGFR in patients treated with ERC.
[0031] Figure 20 shows the Rate of Increase in Serum 24,25-dihydroxyvitamin D3 compared to Serum 25-hydroxy vitamin D3 by Baseline eGFR in patients treated with ERC. *Slope of regression line in Figure 18.
[0032] Figure 21 A shows the time course of mean (+SE) serum total 25-hydroxy vitamin D for 126 study participants, 51 participants who achieved sustained iPTH reductions of > 30% during the last two quarters of the one-year treatment period, and 75 participants who did not achieve sustained iPTH reductions of > 30% during the last two quarters of the one-year treatment period.
[0033] Figure 2 IB shows the time course mean of iPTH for the 126 study participates of Figure 21 A.
[0034] Figure 22A shows an association between duration of plasma iPTH reduction and changes in eGFR. Changes in mean eGFR from BL are shown for all 126 study participants, for 51 participants who achieved sustained iPTH reductions of > 30% during the last two quarters of the one-year treatment period, and 75 participants who did not achieve sustained iPTH reductions of > 30% during the last two quarters of the one-year treatment period .
[0035] Figure 22B shows an association between the degree of iPTH reduction at EOT and changes in eGFR.
DETAILED DESCRIPTION
[0036] The methods are contemplated to include embodiments including any combination of one or more of the additional optional elements, features, and steps further described below (including those shown in the figures), unless stated otherwise.
[0037] In jurisdictions that forbid the patenting of methods that are practiced on the human body, the meaning of “administering” of a composition to a human subject shall be restricted to prescribing a controlled substance that a human subject will self-administer by any technique (e.g., orally, inhalation, topical application, injection, insertion, etc.). The broadest reasonable interpretation that is consistent with laws or regulations defining patentable subject matter is intended. In jurisdictions that do not forbid the patenting of methods that are practiced on the human body, the “administering” of compositions includes both methods practiced on the human body and also the foregoing activities.
[0038] As used herein, the term “comprising” indicates the potential inclusion of other agents, elements, steps, or features, in addition to those specified.
[0039] As used herein, 25-hydroy vitamin D includes one or more hydroxy forms, such as of 25 -hydroxy vitamin D3, 25-hydroxyvitamin D2, and 25-hydroxyvitamin D4. In the same way, 1, 25-dihydroy vitamin D includes 1,25-dihydroxy vitamin D3, 1,25 -hydroxy vitamin D2, and 1.25-hydroxyvitamin D4.
[0040] Methods of mitigating loss of kidney function in accordance with the disclosure include administration of sufficient repeated dosages of 25-hydroxyvitamin D to a patient with SHPT in order to achieve a sustained reduction of iPTH compared to pre-treatment BL for a duration of at least about 6 months. Surprisingly, it has been found that the rate of kidney function loss can be mitigated by maintaining reduced iPTH levels for a duration of at least about 6 months. As used herein “mitigation of the rate of kidney function loss” is measured as a function of observed decreases in eGFR over time, with loss of kidney function being mitigated in a patient when the rate of decrease in eGFR is less than the rate observed over a one-year period preceding treatment initiation.
[0041] Previous observational studies have identified a possible association between SHPT or elevated iPTH and the rate of kidney disease progression. In particular, it has been identified that patients with SHPT or elevated PTH experience higher rates of progression of kidney disease. While it has also been identified that appropriate administration of 25- hydroxyvitamin D to a patient with SHPT can be effective in reducing elevated iPTH, there remains nothing in the literature that would lead to the expectation that effective and sustained reduction of iPTH for an extended period of time is effective in mitigating a patient’s historical rate of kidney function loss.
[0042] In the method described herein, the patient can have stage 3 or stage 4 CKD. In methods described herein, the patients have SHPT.
[0043] Sustained reduction of iPTH to or below a target threshold or target percent reduction threshold as used herein refers to maintained reduction of the average iPTH level to or below a target threshold over the target time duration or maintaining an average target percent reduction from pre-treatment BL over the target time duration. It should be understood herein that iPTH levels vary in patients and achievement of target thresholds or precent reductions is made herein with reference to average values over the target duration. In the method described herein, the target threshold for iPTH can be, for example, 100 pg/mL. The target threshold for iPTH can further include a lower bound, optionally as low as 70 pg/mL, considered to be the upper limit of the laboratory reference range. The target
precent reduction of iPTH from pre-treatment BL can be, for example, at least 5%, at least 10%, at least 20%, or at least 30%.
[0044] Methods of the disclosure can include administering repeated doses of 25- hydroxyvitamin D for a duration and in an amount to achieve a target percent reduction of iPTH from pre-treatment BL and then further administration of repeated doses of 25- hydroxyvitamin D to maintain the target precent reduction of iPTH in the patient.
Administration of repeated doses to maintain the target percent reduction can include dose amount increases where needed to maintain the target percent reduction over the target duration.
[0045] In the method described herein, the target time duration can be at least 6 months, or at least 9 months, or at least 10 months, or at least 11 months, or at least 12 months, or longer. As shown in Example 1 herein, the duration of sustained reduction of iPTH was directly correlated with mitigating loss of kidney function as measured by eGFR.
[0046] In the method described herein, the patient can have an initial eGFR value before the patient’s iPTH is reduced, and the method includes limiting the subsequent eGFR loss within 2.5 mL/min/1.73m2 of the patient’s initial eGFR, or within 2.0 mL/min/1.73m2, or within 1.5 mL/min/1.73m2, or within 1.0 mE/min/1.73m2 of the patient’s initial eGFR for at least 12 months.
[0047] In the method described herein, reducing the patient’ s iPTH level to at or below a target threshold or achieving a target percent reduction from pretreatment BL can include administering sufficient repeated doses of one or more of 25 -hydroxy vitamin D, an active vitamin D compound in extended-release dosing regimen or dosage form, or metabolite of any one of the foregoing to the patient. In a preferred embodiment, the compound is selected from 25-hydroxyvitamin D3. In another preferred embodiment, the 25-hdyroxvitamin D compound is formulated in an extended-release formulation.
[0048] The amount of iPTH reducing compound can, in repeated doses, be sufficient to increase a patient’s serum total 25-hydroxyvitamin D level from pretreatment BL to at least 40 ng/mL, at least 50 ng/mL, at least 55 ng/mL, or at least 60 ng/mL, and/or an increase in serum total 1,25-dihydroxy vitamin D level of at least 5 pg/mL. Such serum total increases in 25-hydroxyvitamin D and 1,25-dihydroxyvitamin D levels can be achieved for at least 6 months, or at least 9 months, or at least 10 months, or at least 11 months, or at least 12 months, or longer.
[0049] It is contemplated that dose titration and increasing dosage amounts to achieve the intended increases in serum total 25-hydroxyvitamin D and/or 1, 25-dihydroxy vitamin D levels may differ in patient populations, for example, based on body mass index (BMI).
[0050] Specific examples of contemplated compounds to be administered include, but are not limited to, 25-hydroxyvitamin D3, 25-hydroxyvitamin D2, 25-hydroxyvitamin D4, la, 25- dihydroxy vitamin D3, l , 25-dihydroxy vitamin D2, la, 25-dihydroxy vitamin D4, and vitamin D analogs (including all hydroxylated, dihydroxylated and trihydroxylated forms), including
1.25-dihydroxy-19-nor- vitamin D2, 22-oxacalcitriol, 26,26,26,27,27,27-hexafluorocalcitriol (falecalcitriol), 24,25-dihydroxyvitamin D3 and 1,24,25-trihydroxyvitamin D3.
[0051] As used herein, the terms “active vitamin D” and “activated vitamin D” refer to a vitamin D compound that is hydroxylated in at least the la position. Active vitamin D compounds include calcitriol, 1, 25-dihydroxy vitamin D2, alfacalcidol, doxercalciferol, 22- oxacalcitriol, and paricalcitol.
[0052] In the method described herein, reducing the patient’ s iPTH level to or below a target threshold can include administering sufficient repeated doses of 25-hydroxyvitamin D to the patient. The 25-hydroxyvitamin D can be 25-hydroxyvitamin D3. The repeated doses can be administered in extended-release compositions. The amount of administered 25- hydroxyvitamin D can, in repeated doses, be sufficient to increase the patient’s serum total
1.25-dihydroxy vitamin D level relative to pretreatment BL by least 5 pg/ml, at least 10 pg/ml, or at least 20 pg/ml. Such increase in serum total 1, 25-dihydroxy vitamin D level can be achieved for at least 6 months, or at least 9 months, or at least 10 months, or at least 11 months, or at least 12 months, or longer.
[0053] In addition or in the alternative, the amount of 25-hydroxyvitamin D can, in repeated doses, be sufficient to achieve a serum total 25-hydroxyvitamin D level in a range of least 40 ng/mL to 60 ng/mL, e.g. at least 40 ng/mL, or at least 50 ng/mL, or at least 60 ng/mL or at least 80 ng/mL, or at least 100 ng/mL or at least 130 ng/mL, or at least 160 ng/mL or at least 170 ng/mL, or at least 200 ng/mL. Such serum total 25-hydroxyvitamin D levels can be achieved for at least 6 months, 9 months, or at least 10 months, or at least 11 months, or at least 12 months, or longer. For example, a patient’s pretreatment BL serum total 25- hydroxyvitamin D can be in a range of about 10 ng/mL to about 50 ng/mL and can be increased with repeated dosing of the iPTH reducing compound to a range of about 70 ng/mL to about 200 ng/mL. It is also contemplated herein that a patient can have a pretreatment BL serum total 25-hydroxyvitamin D greater than 50 ng/mL and can still benefit from methods of
the disclosure and increasing serum total 25-hydroxy vitamin D to levels to reduce iPTH levels to the target threshold or to achieve the target percent reduction for a target duration.
[0054] In methods of the disclosure, reducing and sustaining reduction of the patient’s iPTH level to or below a target threshold and/or achieve a target percent reduction as compared to pre-treatment BL can include administering sufficient repeated doses of 25- hydroxyvitamin D to the patient until the target threshold or target percent reduction of the iPTH is achieved and then maintaining the dosing or increasing where needed to maintain the target threshold or target precent reduction of iPTH. Dosing can be titrated up as needed to achieve the target serum level needed to reduce the patient’ s iPTH levels to the target threshold or by the target precent reduction relative to pre-treatment BL. Dose titration can account, in part, for BMI of the patient. Once a target serum level or target increase in serum level is achieved, the methods of the disclosure include continued administration of repeated doses to maintain, and optionally modify, when needed, the serum levels and the reduction of iPTH for a target duration. The target duration can be, for example, for at least 6 months, or at least 9 months, or at least 10 months, or at least 11 months, or at least 12 months, or longer.
[0055] Selection of a target serum level or increase in serum level from baseline can factor in, for example, a patient’s BMI. For example, for patients having a BMI of less than 30 kg/m2, the repeated doses can be administered to achieve an increase in target serum blood 25- hydroxyvitamin D from pre-treatment BL of about 5 to about 90 ng/mL. Such increases in serum level can be sustained with repeated dosing to achieve the sustained reduction of iPTH for the target duration. For example, in patients having a BMI of 30 kg/m2 or higher, repeated doses can be administered to increase serum blood 25-hydroxy vitamin D levels from pre-treatment BL to at least about 60 ng/mL, or at least 70 ng/mL, or at least 80 ng/mL, or at least 90 ng/mL, or at least 100 ng/mL. Maintenance of serum levels at such levels can provide for a sustained decrease of mean levels of iPTH of about >20% or > 30% in said patient. It has been observed that increasing serum 25-hydroxyvitamin D levels as described herein and maintaining a sustained decrease of iPTH can limit subsequent eGFR decline in such patients.
[0056] Vitamin D compounds and compositions are described in the following patent publications: WO 03/018545 Al, WO 03/031400 Al, WO 03/106411 Al, WO 2004/054968, WO 2006/113990, WO 00/59513, W02007/092755, US-2009-0209501-A1, US-2009- 0082317-A1, US-2009-0176748-A1, WO 2008/134518, WO 2008/134512, WO 2009/047644, WO 2008/134523, US-2009-0311316-Al, US-2009-0233889-A1, US-2011-
0052567-Al, US-2010-0144684-A1, US-2010-0120728-A1, US-2010-0204189-A1, US- 2012-0130133-A1, US-2013-0157987-A1, US-2013-0137663-A1, US-2014-0349979-A1, WO 2014/143941, WO 2016/020508, US-2016-0038514-Al, WO 2017/050438, US-2018- OO85381-A1, US-2019-0083513-A1, US-2019-0298744-A1, WO 2020/044314, US-2020- 0129545-A1, WO 2020/161543, US-2021-0308151-A1, US-2023-0293552-A1, and US- 2022-0226351 -A 1.
[0057] A contemplated extended -release oral formulation includes 25 -hydroxy vitamin D (e.g., about 30 pg, about 60 pg, or about 90 pg 25-hydroxy vitamin D3), about 2 wt% (e.g., 2.32 wt%) anhydrous ethanol, about 10 wt% (e.g., 9.75 wt%) GELUCIRE 44/14, about 20 wt% (e.g., 20.00 wt.%) hard paraffin, about 23 wt% (e.g., 22.55 wt%) GMS, about 35 wt% (e.g., 35.36 wt%) liquid paraffin or mineral oil, about 10 wt% HPMC, and optionally a small amount of preservative (e.g., 0.02 wt% BHT). The composition can be disposed in a capsule shell, for example a hard capsule shell (e.g., gelatin or non-gelatin) or a soft capsule shell (e.g., gelatin or non-gelatin).
[0058] Without intending to be bound by any particular theory, it is possible that excess PTH or iPTH operates as a uremic toxin, and by reducing the amount of PTH or iPTH in circulation, kidney function is better preserved, See, for example, the following publications which describe PTH as a uremic toxin: Vo, V., Sprague, S.M. (2020). Parathyroid Hormone as a Uremic Toxin. In: Covic, A., Goldsmith, D., Urena Torres, P. (eds) Parathyroid Glands in Chronic Kidney Disease. Springer, Cham; Duque et al. Toxins (Basel) 2020 Mar 17; 12(3): 189; and Rodriguez et al. Semin Dial. 2009 Jul-Aug;22(4):363-8.
EXAMPLES
[0059] The following examples are provided for illustration and are not intended to limit the scope of the invention.
Example 1 - Control Of SHPT Is Associated With Slower CKD Progression
[0060] This example demonstrates the effect of controlling SHPT with extended-release calcifediol (ERC) on the rate of CKD progression.
[0061] Adult subjects (n=429) with SHPT, VDI and stage 3 or 4 CKD were stratified by stage and treated daily with either ERC or placebo in two identical, parallel, randomized, double-blind studies. Specifically, two identical 26-week multicenter studies with randomized, double -blind, placebo-controlled designs enrolled a total of 429 subjects from 89
US sites with SHPT (plasma iPTH >85 and <500 pg/mL), stage 3 or 4 CKD (eGFR of >15 and <60 mL/min/1 73m2), and VDI (serum total 25-hydroxy vitamin D >10 and <30 ng/mL). Other eligibility criteria included serum calcium >8.4 and <9.8 mg/dL and serum phosphorus >2.0 and <5.0 mg/dL. Exclusion criteria included a spot urine calcium: creatinine (Ca:Cr) ratio of >0.2, nephrotic range proteinuria (>3 mg/mg Cr) and history of parathyroidectomy for SHPT or renal transplantation. Subjects were enrolled progressively at sites of many different latitudes in order to minimize seasonal variation in mean BL serum total 25- hydroxyvitamin D. Further details regarding these studies have been previously published in Sprague et ah, Use of extended-release calcif ediol to treat secondary hyperparathyroidism in stages 3 and 4 chronic kidney disease, Am J Nephrol 2016;44:316-325, and Sprague et al., Extended-release calcifediol for secondary hyperparathyroidism in stage 3-4 chronic kidney disease, Expert Review of Endocrinology & Metabolism 2017;12:289-301, the disclosures of which are incorporated herein by reference.
Subjects were stratified by CKD stage and were randomized in a 2: 1 ratio to receive a once daily 30 pg oral dose of ERC (or matching placebo) for 12 weeks at bedtime followed by an additional 14 weeks of treatment with once daily bedtime doses of either 30 or 60 pg of ERC (or placebo). The daily dose was increased to 60 pg at the start of week 13 if plasma iPTH remained >70 pg/mL (the upper limit of the laboratory reference range), serum total 25- hydroxy vitamin D was <65 ng/mL (to reduce the risk of driving values above 100 ng/mL) and serum calcium was <9.8 mg/dL. The sole primary efficacy end point was the proportion of subjects in the intent-to-treat (ITT) population that attained a mean decrease of >30% in plasma iPTH from pre-treatment baseline in the efficacy assessment period (EAP), defined as treatment weeks 20 through 26.
[0062] A total of 213 subjects participated in the first of these two RCTs (141 ERC and 72 placebo) and 216 subjects in the other (144 ERC and 72 placebo), and 354 subjects (83%) completed the studies. Data from both RCTs were pooled because: (a) the studies were governed by a common protocol; (b) they were conducted contemporaneously using multiple sites within the continental U.S.; (c) the subject populations were similar according to selection criteria and actual baseline demographic and biochemical characteristics; and, (d) the changes observed in serum total 25-hydroxy vitamin D, serum total 1,25- dihydroxy vitamin D and plasma iPTH were similar during ERC or placebo treatment. In aggregate, 222 subjects (51.7%) had stage 3 CKD (151 ERC and 71 placebo) and 207 subjects (48.3%) had stage 4 CKD (134 ERC and 73 placebo).
[0063] The ITT population included all subjects (n=429) who were randomized to study drug. Subjects in the ITT population had a mean age of 66 years (range 25-85), 50% were male, 65% White, 32% African-American or Black, 21% Hispanic and 3% Other. The most common causes of CKD were diabetes and hypertension and the mean eGFR was 31 mL/min/1.73m2.
[0064] The per-protocol (PP) population included all subjects (n=356) who did not have a major protocol deviation and for whom at least two serum total 25 -hydroxy vitamin D and two plasma iPTH determinations were included in the calculated baseline value and in the EAP, defined as treatment weeks 20 through 26. Demographic and baseline data for the PP population are summarized in Table 1, grouped by CKD stage. Only analyses of the PP population are reported here as they yielded results that did not differ materially from those based on analyses of the ITT population, and because the number of subjects remained constant across the 26-week treatment period. Sixty-two ITT subjects were excluded because they discontinued treatment prior to the EAP, and 11 for major protocol violations: receipt of prohibited concomitant medication (n=4); failure to meet all selection criteria (n=3) ; dosing compliance <80% (n=3); and, premature unblinding (n=l).
[0065] Blood and spot urine samples were collected at weekly or biweekly intervals and analyzed during the applicable stability windows (documented in validation reports) at OOD Global Central Labs (Highland Heights, KY). Plasma iPTH levels were determined by two- site sandwich electrochemiluminescence (Roche Elecsys; reference range 15-65 pg/mL; % CV 2.7). Serum total 25-hydroxy vitamin D was determined by chemiluminescence (DiaSorin), and serum total 1,25-dihydroxy vitamin D was determined by radioimmunoassay (IDS). Serum 25 -hydroxy vitamin D3 (lower limit of quantitation: 5.00 ng/mL; %CV of 0.82 to 1.84 within-run, 2.01 to 4.26% between-run) and 24,25-dihydroxyvitamin D3 (lower limit of quantitation: 0.52 ng/mL; %CV 2.18 to 4.60 within-run, 3.79 to 9.29 between-run) were determined by LC-MS (Syneos) for the purpose of calculating the vitamin D metabolite ratio (VMR), calculated as serum 24,25-dihydroxyvitamin Ds/scrum total 25-hydroxy vitamin D3*100. Other parameters were determined by standard procedures. Serum calcium values were corrected for low albumin. eGFR was measured at baseline and quarterly thereafter (5 assessments). Serum total 25-hydroxy vitamin D and plasma PTH were measured monthly. 166 patients participated in an extension of the study to 52 weeks.
[0066] For each subject, iPTH levels were considered: consistently controlled if <100 pg/mL at 4 of the 5 assessments at pre-treatment BL and quarterly thereafter; partially controlled if <100 pg/mL at 1-3 of the 5 assessments; uncontrolled if >100 pg/mL at all 5 assessments.
[0067] Progressive changes in eGFR were examined post-hoc in 166 patients with vitamin D insufficiency, SHPT, and stage 3-4 CKD during one year of treatment with ERC in pivotal trials (Sprague 2016). ERC was administered daily at 30 mcg increasing, as needed, after 12 weeks to 60 mcg to achieve a targeted at least a 30% reduction in iPTH. Measurements of eGFR were obtained at BL and quarterly intervals, and 25-hydroxyvitamin D (DiaSorin), calcium (Ca; corrected for low albumin), phosphorus (P) and plasma iPTH (Roche Elecsys) at BL and monthly.
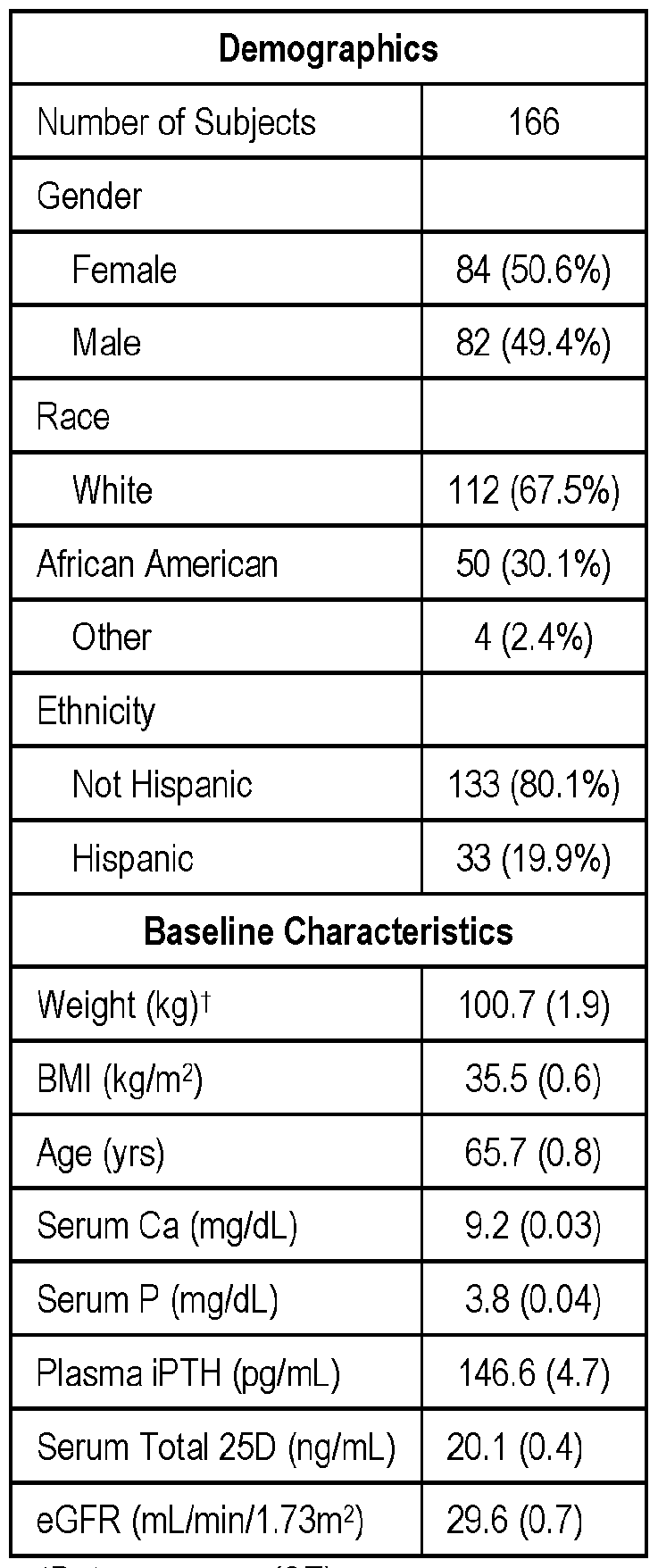
[0068] The demographic and BL characteristics of the 16 patent population are shown in Table 2 below. The population was appropriately balanced for gender and race, and the mean age, body weight, BMI and key biochemical parameters were representative of U.S. patients with stage 3-4 CKD.
[0069] The results demonstrate ERC treatment increased mean (± SE) serum 25- hydroxyvitamin D from 20.1 ± 0.4 ng/mL at BL to 77.8 + 2.0 at EOT (p<0.0001) and decreased mean iPTH from 146.6 + 4.7 pg/mL at BL to 104.4+6.5 (p<0.0001) without clinically meaningful changes in mean serum Ca or P. Moreover, decreases in mean iPTH were unaffected by BL eGFR.
[0070] Average eGFR decline was 2.3 ± 0.5 mL/min/1.73 m2 over the 1-year treatment period but differed significantly and proportionally with duration of iPTH control, being greatest (4.1 ± 0.7) in subjects who never achieved control (n=44) and least (0.61 ± 1.2) in subjects achieving consistent control (n=51; p<0.05).
[0071] The number of subjects having an increased eGFR by EOT rose (p < 0.05) in proportion to the duration of iPTH control achieved, from 6 (no control) to 18 (consistent control).
[0072] The results demonstrate that treatment of SHPT with ERC is associated with mitigation of eGFR decline in patients with insufficient 25-hydroxy vitamin D and stage 3-4 CKD.
[0073] In summary, these results demonstrate that early, sustained, and effective treatment of SHPT was associated with mitigation of eGFR decline in patients with insufficient 25- hydroxyvitamin D and stage 3-4 CKD.
[0074] Results are further described below in connection with the appended figures.
[0075] Figure 1 shows that 26 weeks of ERC treatment effectively raised serum 25- hydroxyvitamin D and 1,25-dihydroxy vitamin D, and reduced elevated plasma iPTH in patients with stage 3 or 4 CKD, irrespective of declining BL eGFR.
[0076] Figure 2 shows how plasma iPTH fell consistently across BL eGFR ranges during 26 weeks of treatment with ERC as a function of increasing serum 1,25-dihydroxy vitamin D.
[0077] Figure 3 shows that increases in mean serum total 25-hydroxy vitamin D were unaffected by declining BL eGFR.
[0078] Figure 4 shows that reductions in mean plasma iPTH were similar in all BL eGFR groups.
[0079] Figure 5 shows that CKD progression was similar in all BL eGFR groups.
[0080] Figure 6 shows that reductions in mean plasma iPTH were similar in all BL eGFR groups.
[0081] Figure 7 shows that increases in mean serum 1,25-dihydroxy vitamin D were similar in all BL eGFR groups.
[0082] Figure 8 shows that reductions in mean iPTH differed by duration of iPTH control.
[0083] Figure 9 shows that increases in serum total 25-hydroxy vitamin D trended directly with duration of iPTH control.
[0084] Figure 10 shows that increases in serum 25-hydroxyvitamin D3 trended directly with duration of iPTH control.
[0085] Figure 11 shows that increases in serum total 1,25-dihydroxyvitamin D trended directly with duration of iPTH control.
[0086] Figure 12 shows that increases in serum 24,25-dihydroxy vitamin D3 directly correlated with duration of iPTH control.
[0087] Figure 13 shows that increases in serum 24,25-dihydroxy vitamin Ds:25- hydroxyvitamin D3 ratio directly correlated with duration of iPTH control.
[0088] Figures 14 and 15 show that 52-wk decreases in eGFR from BL were inversely proportional to duration of iPTH control.
[0089] Figure 16 shows that mean eGFR decline was inversely proportional to duration of iPTH control.
[0090] Figure 17 shows that the number of subjects having an increased eGFR by EOT rose (p < 0.05) in proportion to the duration of iPTH control achieved, from 6 (no control) to 18 (consistent control).
Example 2 - Sustained iPTH Reduction with Extended-Release Calcifediol Slows CKD Progression
[0091] The study described in this example examined, for the first time, whether effective and sustained iPTH reduction provides a clinically meaningful benefit to non-dialysis CKD patients with SHPT. Specifically, it evaluated the hypothesis that sustained iPTH reduction can reduce the nephrotoxic impact of SHPT and forestall CKD progression using clinical trial data from non-dialysis CKD patients treated daily for one year with oral ERC.
[0092] Pooled data from two U.S. multicenter studies with ERC were evaluated post-hoc to assess the potential impact of sustained plasma iPTH reduction on CKD progression.
These two studies were conducted concurrently with identical randomized, double-blind, placebo-controlled designs. At enrollment, the 429 participants had plasma iPTH >85 to <500 pg/mL, eGFR >15 to <60 mL/min/1.73 m2, serum total 25-hydroxyvitamin D >10 to <30 ng/mL, and no macroalbuminuria (>3 mg/mg creatinine). They were stratified 1: 1 by CKD stage and randomized 2: 1 to receive a daily bedtime dose of oral ERC (30 pg) or matching placebo for 12 weeks, followed by 14 weeks of either 30 or 60 pg/day of ERC (or placebo) according to titration rules intended to produce a >30% reduction in iPTH (primary efficacy endpoint) while minimizing safety concerns. Participants who completed either of these two
RCTs could elect to join, without interruption, an open-label extension study during which ERC (but not placebo) was administered for an additional 26 weeks, with similar dose titration to 60 pg/day allowed at Week 38. The 126 participants who completed 52 weeks of ERC treatment were the focus of the present study.
[0093] Measurements of 25 -hydroxy vitamin D (DiaSorin), calcium (corrected for low albumin), phosphorus and iPTH (Roche Elecsys) were obtained at BL and at biweekly or monthly intervals. Measurements of eGFR (MDRD equation), spot urine albumin-to- creatinine ratio (uACR), 1,25-dihydroxy vitamin D (IDS), FGF23 (Millipore), and bone turnover markers (BTM) were obtained at BL and the subsequent four quarterly intervals. Monitored BTM were bone-specific alkaline phosphatase (BSALP; Quidel), collagen type 1 C-telopeptide (CTx-1; Roche Cobas), intact procollagen type 1 N-terminal propeptide (P1NP; Roche Cobas), and total alkaline phosphatase (ALP; Roche Cobas).
[0094] The 126 participants were grouped into two categories depending on achievement of a sustained mean iPTH reduction of >30% (versus pre-treatment BL) at both Week 38 (mean of data obtained at Weeks 34 through 38) and Week 52 (mean of Weeks 48 through 52) of the one-year treatment period. Observed differences between these categories were evaluated by t-test or one-way analysis of variance (ANOVA) followed by Tukey’s test of the means.
[0095] Demographic characteristics of the 126 participants who completed one year of treatment with ERC (30 or 60 pg/day) are summarized in Table 3, in aggregate and by iPTH reduction category.
[0096] A total of 51 participants (40.5%) achieved a mean iPTH reduction of >30% (versus pre-treatment BL) that was sustained over the last 2 quarters of the treatment period while 75 (59.5%) did not. Participants in both categories were similar in age, balanced for gender, race and Hispanic ethnicity, had similar utilization of angiotensin-converting enzyme (ACE) inhibitors and angiotensin receptor blockers (ARBs), and had mean BMI above 30 kg/m2, with those in the latter category having a greater mean BMI (p<0.05).
* p<0.05 compared to the category that failed to achieve iPTH reductions of > 30% in both
Q3 and Q4 (t-test)
[0098] Clinical characteristics for these participants at BL and EOT are summarized in Table 4, in aggregate and by iPTH reduction category. Participants in both categories had similar BL concentrations for all characteristics except for P1NP which was 31% higher in participants who did not achieve sustained iPTH reductions (p<0.01). Serum total 25- hydroxyvitamin D rose with ERC treatment from 20.5±0.5 (SE) to 79.0±2.3 ng/mL (p<0.001). Most participants (74%) titrated from 30 to 60 pg/day after 12 weeks, as did a few more after 38 weeks.
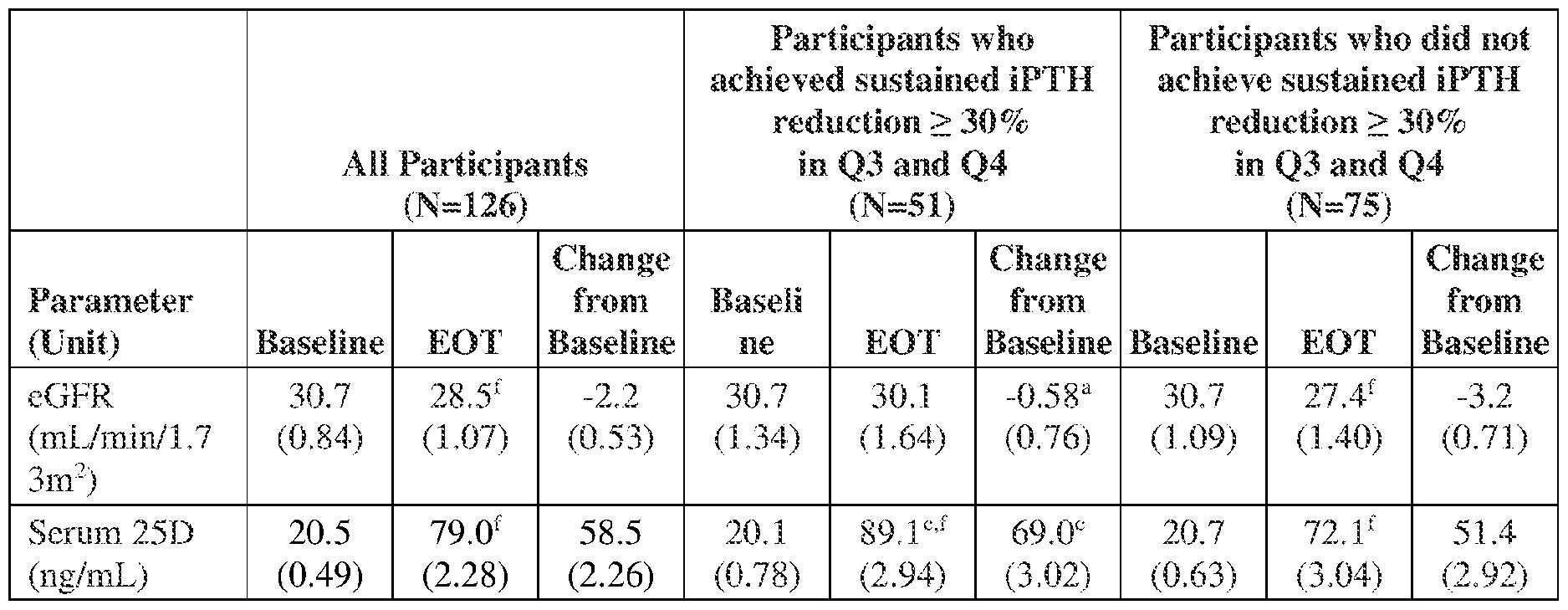
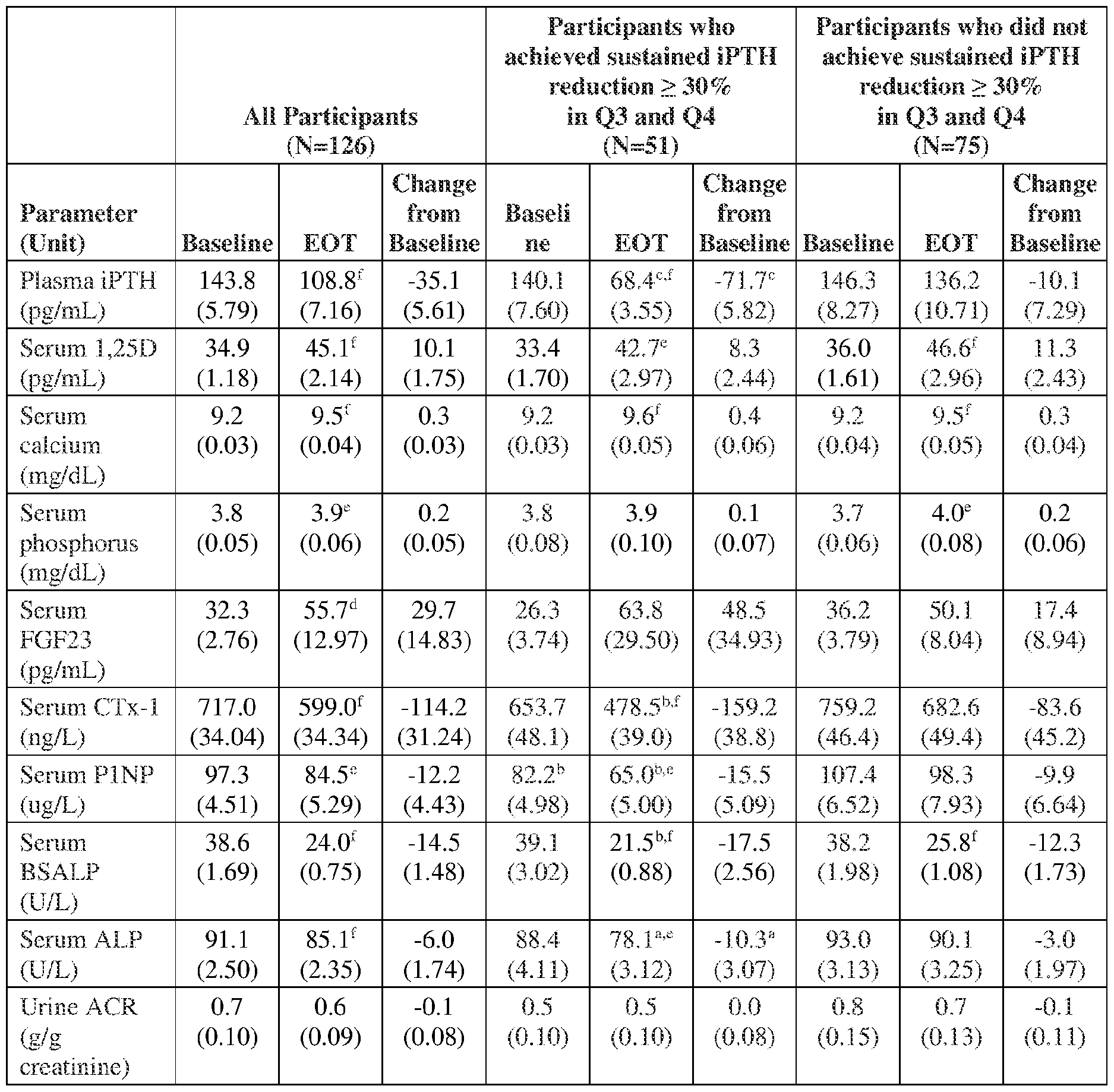
Table 4. Change in Clinical Parameters After 52 Weeks of ERC Treatment (versus BL)
ote: Values are n, mean (SE); EOT = end of treatment; eGFR = estimated glomerular filtration rate; 25D = total serum 25-hyroxy vitamin D; iPTH = intact parathyroid hormone; 1,25D = total serum 1, 25 -dihydroxy vitamin D; FGF23 = fibroblast growth factor-23; CTx-1 = type 1 collagen C-telopeptides; P1NP = procollagen 1 N-terminal propeptide; BSALP = bone- specific alkaline phosphatase ALP = alkaline phosphatase; ACR = albumin/creatinine ratio a p<0.05; b p<0.01; c p<0.001 between categories (t-test); d p<0.05; e p<0.01; f p<0.001 within category (paired t-test)
[0099] At EOT, serum 25 -hydroxy vitamin D averaged 17.0 ng/mL higher (p<0.001) in participants who achieved sustained iPTH reductions of >30% (Figure 21A). Serum 1,25-
dihydroxyvitamin D rose with ERC treatment by 10.1±1.8 pg/mL (from 34.9±1.2; p<0.001), serum calcium by 0.3+0.03 mg/dL (from 9.2+0.03; p<0.001), serum phosphorus by 0.2+0.05 mg/dL (from 3.80+0.05; p<0.01), serum FGF23 by 29.7+14.8 pg/mL (from 32.3+2.8; p<0.05; one participant’s values rose from 57 to 1,488) and all BTMs improved (p<0.01). Similar changes in these parameters were observed in both categories. No change during ERC treatment was observed in mean uACR for the full group of participants.
[00100] Plasma iPTH decreased by 24.3% from 143.8±5.8 to 108.8±7.2 pg/mL, and reached concentrations that averaged 67.8 pg/mL lower in the category achieving sustained iPTH reductions of >30% (Figure 2 IB). In the other category comprised of 75 participants, 45 achieved iPTH reductions of any magnitude that were sustained over the last 2 quarters, of which 15 and 18 achieved at least 20% and 10% reductions, respectively. Another 30 participants did not exhibit an iPTH reduction in both of the last 2 quarters.
[00101] The average decline in eGFR in the 126 participants was 2.2+0.5 mL/min/1.73m2 from 30.7+0.8 at BL to 28.5+1.1 at EOT (Figure 22A). The mean rate of eGFR decline was >5-fold higher (p=0.014) in participants who did not achieve sustained iPTH reductions of >30% (3.2+0.7; 12.7+2.2%) compared to those who did (0.6+0.8; 2.9+2.4%). It was highest in the 30 participants who did not exhibit an iPTH lowering response in both of the final 2 quarters of treatment (5.4±0.89; 20.9±3.4%). Analysis of mean decreases in eGFR by degree of iPTH reduction at EOT in all participants indicated that reductions of >30% were associated with near stabilization of CKD progression, while reductions of <30% were associated with the higher rates of eGFR decline (Figure 22B). The duration of iPTH reduction had no apparent impact on 52-week changes in mean serum calcium, phosphorus, FGF23, episodes of BTM oversuppression, hypercalcemia (confirmed serum calcium >10.3 mg/dL), hyperphosphatemia (confirmed serum phosphorus >5.5 mg/dL) or hypercalciuria (urine calcium to creatinine ratio of >200), or rates of adverse events.
[00102] This post-hoc analysis of clinical trial data showed that sustained reduction of elevated iPTH with ERC treatment was associated with slower rates of eGFR decline in nondialysis CKD patients diagnosed with SHPT without raising safety concerns.
Example 3 - ERC overcomes impact of low eGFR on Vitamin D metabolism
[00103] This example demonstrates the effect of declining eGFR on vitamin D metabolism in three different clinical populations treated with ERC: patients with normal kidney function, patients with stage 3-4 CKD, and patients with end-stage renal disease (ESRD).
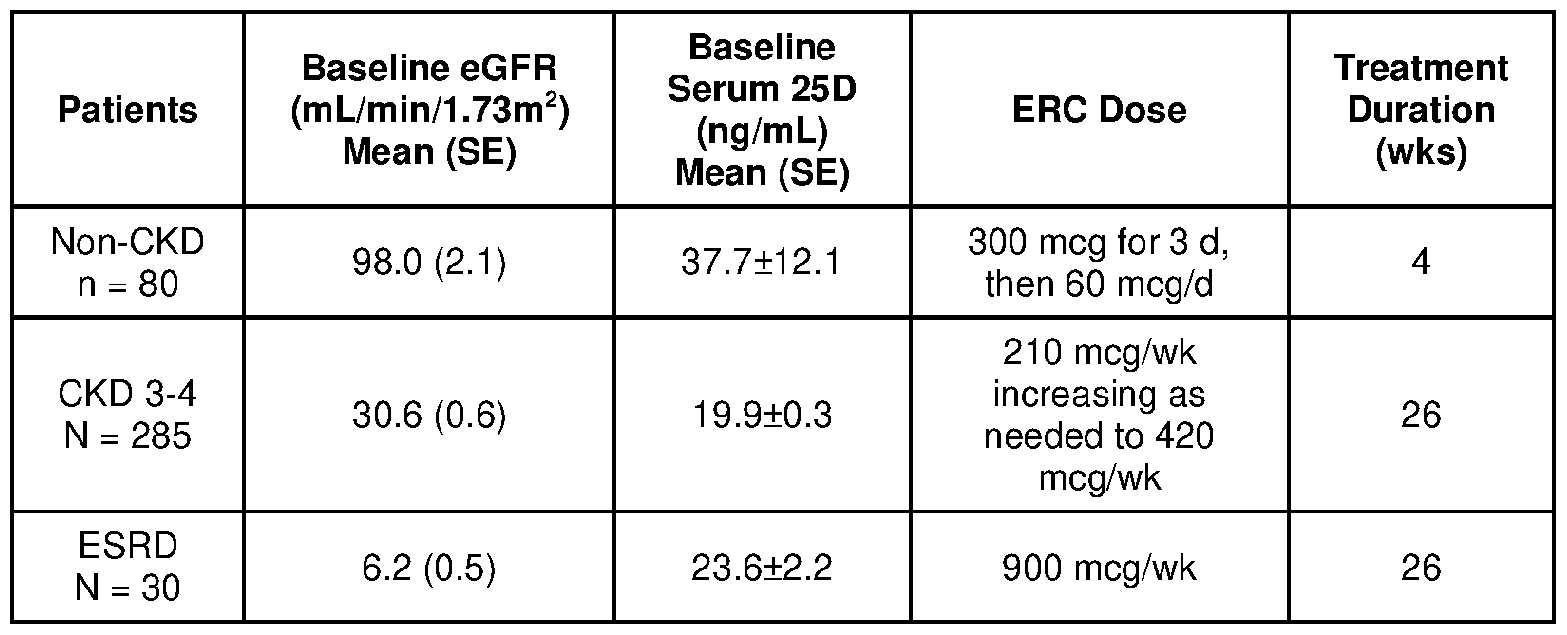
[00104] Changes in serum total 25 -hydroxy vitamin D and 1,25-dihydroxy vitamin D, calcifediol (25 -hydroxy vitamin D3), 24,25-dihydroxyvitamin D3 (24,25-dihydroxyvitamin D3) and calcitriol (1,25-dihydroxy vitamin D3) during ERC treatment in four randomized clinical trials were assessed compared as a function of eGFR. In one study, 80 non-CKD patients with COVID- 19 were treated for 4 weeks (wks) with 300 mcg/day (d) for the first three days and 60 mcg/d thereafter (Bishop CW, Ashfaq A, Melnick JZ et al. REsCue trial: Randomized controlled clinical trial with extended-release calcifediol in symptomatic COVID-19 outpatients. Nutrition 2023; 107: 111899). In two other studies (pooled), 285 nondialysis patients with eGFR of 30.6+0.6 (mean+SE) mL/min/1.73m2 were treated with 210 mcg/wk for 12 weeks, at which time 74% were increased to 420 mcg/wk (Sprague SM, et al. Am J Nephrol. 2016;44:316-25). In another, 33 hemodialysis (HD) patients were treated for 26 wks with 900 mcg/wk (Strugnell SA, Csomor P, Ashfaq A, Bishop CW. Initial evaluation of high-dose extended-release calcifediol (ERC) in patients with stage 5 CKD on hemodialysis. American Society of Nephrology 2021 Annual Meeting, Abstract TH-OR19.).
[00105] Baseline eGFR and serum total 25-hydroxy vitamin D levels of subjects included in the present analysis are listed in the table below, along with subject numbers, ERC doses, and treatment durations for each population.
[00106] The results demonstrate that mean serum total 25 -hydroxy vitamin D values at baseline were lower in patients with reduced eGFR.
[00107] Moreover, the results demonstrate that the mean serum 1,25-dihydroxy vitamin D3 or total 1,25-dihydroxy vitamin D values at baseline (Figurel9) were proportional to eGFR. During ERC treatment, mean serum 25-hydroxyvitamin D3 or total 25 -hydroxy vitamin D rose to >70 ng/mL with peak levels proportional to the total administered dose (Figure 18).
Mean serum 1,25-dihydroxy vitamin D3 or total 1,25 -dihydroxy vitamin D rose linearly with serum 25-hydroxyvitamin D3 or total 25-hydroxyvitamin D at similar rates in all eGFR
groups (Figure 19). However, serum 24,25-dihydroxyvitamin D3 levels rose less quickly with rising serum 25-hydroxyvitamin D3 as eGFR decreased (Figures 18 and 20).
[00108] ERC reliably raised serum 25-hydroxy vitamin D3, 1,25-dihydroxy vitamin D3, and total 25-hydroxy vitamin D and 1,25-dihydroxy vitamin D irrespective of eGFR, making it an attractive alternative to calcitriol (or la-hydroxylated analogs) for treating persistently rising PTH in CKD 3-4.
[00109] Declining eGFR did not affect ERC’s ability to increase the rate of 1,25- dihydroxyvitamin D3 production, indicating that hormone generation occurred in extra-renal tissue.
[00110] Increases in serum 24,25-dihydroxyvitamin D3 were dependent on 25- hydroxyvitamin D3 elevation and limited by declining eGFR, suggesting that this metabolite is not disproportionately increased by ERC and derives solely from kidney.
Example 4 - Adjunctive Active Vitamin D Comparative Example
A prospective study examined whether adjunctive active vitamin D safely improved iPTH control and the related eGFR stabilizing effect of ERC in 78 non-dialysis chronic kidney disease (ND-CKD) adults unable to achieve iPTH normalization after 38 weeks of treatment, defined as BL. Participants had a mean age of 66 years, BMI of 35 kg/m2, 41% were female, 63% White, 36% Black, and 19% Hispanic. At ERC initiation, participants had eGFR 15 to <60 mL/min/1.73 m2, plasma iPTH 85 to<500 pg/mL, serum 25-hydroxyvitamin D 10 to <30 ng/mL, corrected serum calcium (Ca) 8.4 to <9.8, serum phosphorus (P) 2.0 to <5.0, and absence of nephrotic range proteinuria (<3 mg/mg creatinine). They were randomized to continuing daily ERC (60 mcg) with (n=40) or without (n=38) immediate-release adjunctive daily oral calcitriol (0.25 mcg; n=12), doxercalciferol (0.5 mcg; n=14), or paricalcitol (1.0 mcg; n=14) for 14 weeks. Measurement of eGFR, iPTH, 25-hydroxyvitamin D, Ca, P, FGF23 were obtained at BL and study end.
There were no significant BL inter-group differences. Mean BL 25-hydroxyvitamin D was 67 n/gmL and rose 11-13 ng/mL in both groups (p<0.001). mean BL iPTH was 143 pg/mL and fell by 35% (p <0.01 ) with adjunctive therapy versus 4% without. Mean Ca and FGF23 increased with adjunctive therapy (p<0.001) by o.5 mg/dL and 47 pg/mL respectively, versus 0.0 and 2.0 without. Mean P remained unchanged. Mean BL eGFR was 25.4 mL/min/1.73 m2 and fell by 10% (p<0.05) with adjunctive therapy versus 4% without.
Although adjunctive active vitamin D enabled more iPTH reduction (35%) in CKD patients taking ERC for 52 weeks, it increased serum Ca by 0.5 mg/dL, FGF23 by 136%, and hastened eGFR decline by 10%. Higher doses of ERC alone are believed to be more suitable for improving iPTH control and eGFR stabilization in patients.
[00111] The foregoing description is given for clearness of understanding only, and no unnecessary limitations should be understood therefrom, as modifications within the scope of the invention may be apparent to those having ordinary skill in the art.
[00112] Throughout this specification and the claims which follow, unless the context requires otherwise, the word “comprise” and variations such as “comprises” and “comprising” will be understood to imply the inclusion of a stated integer or step or group of integers or steps but not the exclusion of any other integer or step or group of integers or steps.
[00113] Throughout the specification, where compositions are described as including components or materials, it is contemplated that the compositions can also consist essentially of, or consist of, any combination of the recited components or materials, unless described otherwise. Eikewise, where methods are described as including particular steps, it is contemplated that the methods can also consist essentially of, or consist of, any combination of the recited steps, unless described otherwise. The invention illustratively disclosed herein suitably may be practiced in the absence of any element or step which is not specifically disclosed herein.
[00114] The practice of a method disclosed herein, and individual steps thereof, can be performed manually and/or with the aid of or automation provided by electronic equipment. Although processes have been described with reference to particular embodiments, a person of ordinary skill in the art will readily appreciate that other ways of performing the acts associated with the methods may be used. For example, the order of various of the steps may be changed without departing from the scope or spirit of the method, unless described otherwise. In addition, some of the individual steps can be combined, omitted, or further subdivided into additional steps.
[00115] All patents, publications and references cited herein are hereby fully incorporated by reference. In case of conflict between the present disclosure and incorporated patents, publications and references, the present disclosure should control.
Claims
1. A method of mitigating loss of kidney function in a chronic kidney disease (CKD) patient having secondary hyperparathyroidism (SHPT), comprising: administering repeated doses of 25 -hydroxy vitamin D in a sufficient dosage amount to increase a serum 25 -hydroxy vitamin D level of the patient to reduce iPTH of the patient by a target percent reduction from a pretreatment baseline; and continuing administering repeated doses of 25 -hydroxy vitamin D in a sufficient dosage amount to maintain and/or further increase, if needed, a serum 25 -hydroxy vitamin D level of the patient to maintain the target percent reduction of iPTH for a target duration.
2. A method of mitigating loss of kidney function in a CKD patient having SHPT, comprising administering repeated doses of 25-hydroxyvintamin D in a dosage amount and for a treatment time sufficient to achieve and sustain over a target duration an absolute or relative target reduction of iPTH from a pretreatment baseline, thereby mitigating loss of kidney function.
3. The method of any one of claims 1 or 2, wherein the target percent reduction is at least 5%, at least 10%, at least 20%, or at least 30% as compared to the pretreatment baseline.
4. A method of preventing decrease of estimated glomerular filtration rate (eGFR) in an amount of greater than 10% as compared to pretreatment baseline eGFR in a CKD patient having SHPT, comprising administering 25-hydroxy vitamin D in repeated doses and for a treatment time to achieve a sustained reduction of iPTH from a pretreatment baseline iPTH for at least 6 months, thereby preventing decrease of eGFR in an amount of greater than 2% as compared to the pretreatment baseline eGFR.
5. The method of claim 4, wherein the 25-hydroxy vitamin D is 25- hydroxvyvitamin D3 provided as an extended-release oral dosage.
6. A method of mitigating loss of kidney function in a CKD patient having SHPT, comprising reducing the patient’s iPTH level to or below an absolute or relative target threshold and maintaining the iPTH level at or below the target threshold for a target duration of time, and thereby mitigating loss of kidney function as measured by eGFR.
7. The method of claim 6, wherein the target threshold for serum iPTH is 100 pg/mL.
8. The method of claims 6 or 7, wherein the target threshold for serum iPTH further includes a lower bound, optionally as low as 70 pg/mL.
9. The method of any one of claims 6 to 8, wherein reducing the patient’s iPTH level to at or below a target threshold comprises administering sufficient repeat doses of one or more of an active vitamin compound, 25 -hydroxy vitamin D, or a metabolite of any one of the foregoing to the patient.
10. The method of claim 9, wherein reducing the patient’s intact iPTH level to at or below a target threshold comprises administering sufficient repeat doses of 25- hydroxyvitamin D to the patient.
11. The method of any one of the preceding claims, wherein the patient has stage 3 or stage 4 CKD.
12. The method of any one of the preceding claims, wherein the target duration is at least 6 months, or at least 9 months, or at least 12 months.
13. The method of any one of the preceding claims, wherein the target duration is continuous.
14. The method of any one of the preceding claims, wherein the patient has an initial eGFR value before the patient’ s iPTH is reduced, and comprising limiting the subsequent eGFR loss within 2.0 mL/min/1.73m2 of the patient’s initial eGFR, or within 2.0 mL/min/1.73m2, or within 1.5 mL/min/1.73m2, or within 1.0 mL/min/1.73m2 of the patient’s initial eGFR for at least 12 months.
15. The method of any one of the preceding claims, wherein the repeat doses are administered as extended-release oral doses.
16. A method of decreasing the rate of eGFR decline in a patient having a mean BMI in the range of about 30 to 40 kg/m2, comprising administering a pharmaceutically effective amount of a controlled release dosage form of 25-hydroxy vitamin D for a sustained period of time to achieve a sustained increase of total 25 -hydroxy vitamin D in serum to at least about 50 ng/mL and a sustained decrease in mean serum levels of iPTH of about > 30% in said patient.
17. A method of reducing the nephrotoxic impact of SHPT in a subject having a mean BMI in the range of about 30 kg/m2 to 40 kg/m2 comprising administering a pharmaceutically effective amount of a controlled release dosage form of 25- hydroxyvitamin D sufficient to raise serum blood levels by at least about 50 ng/mL while reducing the mean serum levels of iPTH in said patient by about >30% for a sustained period of time.
18. A method of decreasing the rate of eGFR decline in a CKD patient having a mean BMI of greater than 30 kg/m2 comprising administering a pharmaceutically effective amount of a controlled release formulation of 25-hydroxy vitamin D sufficient to raise mean serum total 25 -hydroxy vitamin D levels to at least 150 ng/mL for a sustained period of time while correspondingly reducing the mean serum levels of iPTH by at least 20% for the sustained period of time.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US202363595709P | 2023-11-02 | 2023-11-02 | |
| US63/595,709 | 2023-11-02 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2025094144A1 true WO2025094144A1 (en) | 2025-05-08 |
Family
ID=93650458
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/IB2024/060834 Pending WO2025094144A1 (en) | 2023-11-02 | 2024-11-01 | Controlling loss of kidney function |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2025094144A1 (en) |
Citations (28)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US2011A (en) | 1841-03-18 | Appabatxts for piling saws | ||
| US52567A (en) | 1866-02-13 | Improved weather-strip | ||
| WO2000059513A1 (en) | 1999-04-01 | 2000-10-12 | Johns Hopkins University | NON-CALCEMIC, ANTIPROLIFERATIVE, TRANSCRIPTIONALLY ACTIVE SULFUR-CONTAINING ANALOGS OF 1α, 25-DIHYDROXY VITAMIN D¿3? |
| WO2003018545A1 (en) | 2001-08-22 | 2003-03-06 | Johns Hopkins University | 24-sulfur-substituted analogs of 1α,25-dihydroxy vitamin d¿3? |
| WO2003031400A1 (en) | 2001-10-12 | 2003-04-17 | Johns Hopkins University | Low-calcemic oxime analogs of 1alpha, 25-dihydroxy vitamin d3 |
| WO2003106411A1 (en) | 2002-06-13 | 2003-12-24 | Johns Hopkins University | 24-sulfoximine vitamin d3 compounds |
| WO2004054968A2 (en) | 2002-12-18 | 2004-07-01 | Johns Hopkins University | 25-so2-substituted analogs of 1alpha, 25-dihydroxyvitamin d3 (calcitriol) |
| WO2006113990A2 (en) | 2005-04-25 | 2006-11-02 | Cytochroma Inc. | LOW-CALCEMIC 16,23-DIENE 25-OXIME ANALOGS OF 1α ,25-DIHYDROXY VITAMIN D3 |
| WO2007092755A2 (en) | 2006-02-03 | 2007-08-16 | Proventiv Therapeutics, Llc | Treating vitamin d insufficiency and deficiency with 25-hydroxyvitamin d2 and 25-hydroxyvitamin d3 |
| WO2008134523A1 (en) | 2007-04-25 | 2008-11-06 | Proventiv Therapeutics, Llc | Method of safely and effectively treating and preventing secondary hyperparathyroidism in chronic kidney disease |
| WO2008134518A2 (en) | 2007-04-25 | 2008-11-06 | Cytochroma Inc. | Methods and compounds for vitamin d therapy |
| WO2008134512A1 (en) | 2007-04-25 | 2008-11-06 | Cytochroma Inc. | Oral controlled release compositions comprising vitamin d compound and waxy carrier |
| WO2009047644A2 (en) | 2007-04-25 | 2009-04-16 | Cytochroma Inc. | Method of treating vitamin d insufficiency and deficiency |
| US20090209501A1 (en) | 2006-06-21 | 2009-08-20 | Proventiv Therapeutics, Llc. | Method of treating and preventing secondary hyperparathyroidism |
| US20090233889A1 (en) | 2008-03-12 | 2009-09-17 | Uttam Saha | Stabilized 1,25-dihydroxyvitamin d2 and method of making same |
| US20120130133A1 (en) | 2008-11-26 | 2012-05-24 | Cytochroma Inc. | Method For Synthesizing Vitamin D Analogs |
| US20130137663A1 (en) | 2010-03-29 | 2013-05-30 | Eric J. Messner | Methods and compositions for reducing parathyroid levels |
| US20130157987A1 (en) | 2010-01-13 | 2013-06-20 | Johns Hopkins University | 1-deoxy analogs of 1,25-dihydroxyvitamin d3 compounds |
| WO2014143941A1 (en) | 2013-03-15 | 2014-09-18 | Opko IP Holdings II, Inc. | Stabilized modified release vitamin d formulation and method of administring same |
| US20160038514A1 (en) | 2014-08-07 | 2016-02-11 | Opko Ireland Global Holdings, Ltd. | Adjunctive Therapy With 25-Hydroxyvitamin D and Articles Therefor |
| WO2016020508A2 (en) | 2014-08-07 | 2016-02-11 | Opko Ireland Global Holdings Ltd. | Adjunctive therapy with 25-hydroxyvitamin d |
| WO2017182237A1 (en) | 2016-03-28 | 2017-10-26 | Opko Ireland Global Holdings, Limited | Methods of vitamin d treatment |
| US20190298744A1 (en) | 2018-04-03 | 2019-10-03 | Opko Ireland Global Holdings, Ltd. | Use of Calcifediol in Bariatric Surgery Patients |
| WO2020044314A1 (en) | 2018-08-31 | 2020-03-05 | Opko Ireland Global Holdings, Ltd. | Vitamin d pediatric dosage forms, methods of making and using |
| US20200129545A1 (en) | 2018-10-25 | 2020-04-30 | Opko Ireland Global Holdings, Ltd. | Methods of treatment with mixed metal compound |
| WO2020161543A1 (en) | 2019-02-06 | 2020-08-13 | Opko Ireland Global Holdings, Limited | Method of controlling progression of hyperparathyroidism with calcifediol, and compositions for use therein |
| US20210308151A1 (en) | 2020-04-06 | 2021-10-07 | Eirgen Pharma Ltd. | Activating endogenous antimicrobials to treat sars-cov-2 infection |
| US20230293552A1 (en) | 2020-05-31 | 2023-09-21 | Eirgen Pharma Ltd. | Hard capsule dosage form and uses thereof |
-
2024
- 2024-11-01 WO PCT/IB2024/060834 patent/WO2025094144A1/en active Pending
Patent Citations (39)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US52567A (en) | 1866-02-13 | Improved weather-strip | ||
| US2011A (en) | 1841-03-18 | Appabatxts for piling saws | ||
| WO2000059513A1 (en) | 1999-04-01 | 2000-10-12 | Johns Hopkins University | NON-CALCEMIC, ANTIPROLIFERATIVE, TRANSCRIPTIONALLY ACTIVE SULFUR-CONTAINING ANALOGS OF 1α, 25-DIHYDROXY VITAMIN D¿3? |
| WO2003018545A1 (en) | 2001-08-22 | 2003-03-06 | Johns Hopkins University | 24-sulfur-substituted analogs of 1α,25-dihydroxy vitamin d¿3? |
| WO2003031400A1 (en) | 2001-10-12 | 2003-04-17 | Johns Hopkins University | Low-calcemic oxime analogs of 1alpha, 25-dihydroxy vitamin d3 |
| WO2003106411A1 (en) | 2002-06-13 | 2003-12-24 | Johns Hopkins University | 24-sulfoximine vitamin d3 compounds |
| WO2004054968A2 (en) | 2002-12-18 | 2004-07-01 | Johns Hopkins University | 25-so2-substituted analogs of 1alpha, 25-dihydroxyvitamin d3 (calcitriol) |
| US20090082317A1 (en) | 2005-04-25 | 2009-03-26 | Cytochroma Inc. | Low-calcemic 16,23-diene 25-oxime analogs of 1alpha,25-dihydroxy vitamin d3 |
| WO2006113990A2 (en) | 2005-04-25 | 2006-11-02 | Cytochroma Inc. | LOW-CALCEMIC 16,23-DIENE 25-OXIME ANALOGS OF 1α ,25-DIHYDROXY VITAMIN D3 |
| WO2007092755A2 (en) | 2006-02-03 | 2007-08-16 | Proventiv Therapeutics, Llc | Treating vitamin d insufficiency and deficiency with 25-hydroxyvitamin d2 and 25-hydroxyvitamin d3 |
| US20090311316A1 (en) | 2006-02-03 | 2009-12-17 | Proventiv Thereapeutics, Llc | Treating vitamin d insufficiency and deficiency with 25-hydroxyvitamin d2 and 25-hydroxyvitamin d3 |
| US20090209501A1 (en) | 2006-06-21 | 2009-08-20 | Proventiv Therapeutics, Llc. | Method of treating and preventing secondary hyperparathyroidism |
| US20100120728A1 (en) | 2007-04-25 | 2010-05-13 | Cytochroma Inc. | Method of Treating Vitamin D Insufficiency and Deficiency |
| WO2009047644A2 (en) | 2007-04-25 | 2009-04-16 | Cytochroma Inc. | Method of treating vitamin d insufficiency and deficiency |
| US20090176748A1 (en) | 2007-04-25 | 2009-07-09 | Cytochroma Inc. | Methods and compositions for controlled release oral dosage of a vitamin d compound |
| WO2008134512A1 (en) | 2007-04-25 | 2008-11-06 | Cytochroma Inc. | Oral controlled release compositions comprising vitamin d compound and waxy carrier |
| WO2008134518A2 (en) | 2007-04-25 | 2008-11-06 | Cytochroma Inc. | Methods and compounds for vitamin d therapy |
| WO2008134523A1 (en) | 2007-04-25 | 2008-11-06 | Proventiv Therapeutics, Llc | Method of safely and effectively treating and preventing secondary hyperparathyroidism in chronic kidney disease |
| US20100144684A1 (en) | 2007-04-25 | 2010-06-10 | Proventiv Therapeutics, Inc. | Method of Safely and Effectively Treating and Preventing Secondary Hyperparathyroidism in Chronic Kidney Disease |
| US20100204189A1 (en) | 2007-04-25 | 2010-08-12 | Petkovich P Martin | Methods and Compounds for Vitamin D Therapy |
| US20090233889A1 (en) | 2008-03-12 | 2009-09-17 | Uttam Saha | Stabilized 1,25-dihydroxyvitamin d2 and method of making same |
| US20120130133A1 (en) | 2008-11-26 | 2012-05-24 | Cytochroma Inc. | Method For Synthesizing Vitamin D Analogs |
| US20130157987A1 (en) | 2010-01-13 | 2013-06-20 | Johns Hopkins University | 1-deoxy analogs of 1,25-dihydroxyvitamin d3 compounds |
| US20130137663A1 (en) | 2010-03-29 | 2013-05-30 | Eric J. Messner | Methods and compositions for reducing parathyroid levels |
| US20140349979A1 (en) | 2013-03-15 | 2014-11-27 | Opko IP Holdings II, Inc. | Stabilized modified release vitamin d formulation and method of administering same |
| WO2014143941A1 (en) | 2013-03-15 | 2014-09-18 | Opko IP Holdings II, Inc. | Stabilized modified release vitamin d formulation and method of administring same |
| US20180085381A1 (en) | 2014-08-07 | 2018-03-29 | Opko Ireland Global Holdings, Ltd. | Adjunctive Therapy With 25-Hydroxyvitamin D |
| US20160038514A1 (en) | 2014-08-07 | 2016-02-11 | Opko Ireland Global Holdings, Ltd. | Adjunctive Therapy With 25-Hydroxyvitamin D and Articles Therefor |
| WO2016020508A2 (en) | 2014-08-07 | 2016-02-11 | Opko Ireland Global Holdings Ltd. | Adjunctive therapy with 25-hydroxyvitamin d |
| WO2017050438A1 (en) | 2015-08-06 | 2017-03-30 | Opko Ireland Global Holdings Ltd. | Adjunctive therapy with 25-hydroxyvitamin d and articles therefor |
| US20190083513A1 (en) | 2016-03-28 | 2019-03-21 | Opko Ireland Global Holdings, Ltd. | Methods of treating vitamin d insufficiency in chronic kidney disease |
| WO2017182237A1 (en) | 2016-03-28 | 2017-10-26 | Opko Ireland Global Holdings, Limited | Methods of vitamin d treatment |
| US20190298744A1 (en) | 2018-04-03 | 2019-10-03 | Opko Ireland Global Holdings, Ltd. | Use of Calcifediol in Bariatric Surgery Patients |
| WO2020044314A1 (en) | 2018-08-31 | 2020-03-05 | Opko Ireland Global Holdings, Ltd. | Vitamin d pediatric dosage forms, methods of making and using |
| US20200129545A1 (en) | 2018-10-25 | 2020-04-30 | Opko Ireland Global Holdings, Ltd. | Methods of treatment with mixed metal compound |
| WO2020161543A1 (en) | 2019-02-06 | 2020-08-13 | Opko Ireland Global Holdings, Limited | Method of controlling progression of hyperparathyroidism with calcifediol, and compositions for use therein |
| US20220226351A1 (en) | 2019-02-06 | 2022-07-21 | Eirgen Pharma Ltd. | Method of Controlling Progression of Hyperparathyroidism, And Compositions for Use Therein |
| US20210308151A1 (en) | 2020-04-06 | 2021-10-07 | Eirgen Pharma Ltd. | Activating endogenous antimicrobials to treat sars-cov-2 infection |
| US20230293552A1 (en) | 2020-05-31 | 2023-09-21 | Eirgen Pharma Ltd. | Hard capsule dosage form and uses thereof |
Non-Patent Citations (12)
| Title |
|---|
| BISHOP CHARLES W. ET AL: "Sustained Reduction of Elevated Intact Parathyroid Hormone Concentrations with Extended-Release Calcifediol Slows Chronic Kidney Disease Progression in Secondary Hyperparathyroidism Patients", AMERICAN JOURNAL OF NEPHROLOGY, 27 August 2024 (2024-08-27), CH, pages 1 - 10, XP093247800, ISSN: 0250-8095, DOI: 10.1159/000541138 * |
| BISHOP CWASHFAQ AMELNICK JZ ET AL.: "REsCue trial: Randomized controlled clinical trial with extended-release calcifediol in symptomatic COVID-19 outpatients", NUTRITION, vol. 107, 2023, pages 111899 |
| DUQUE ET AL., TOXINS, vol. 12, no. 3, 17 March 2020 (2020-03-17), pages 189 |
| GERMAIN MICHAEL J. ET AL: "Real-world assessment: effectiveness and safety of extended-release calcifediol and other vitamin D therapies for secondary hyperparathyroidism in CKD patients", BMC NEPHROLOGY, vol. 23, no. 1, 11 November 2022 (2022-11-11), GB, XP093248179, ISSN: 1471-2369, Retrieved from the Internet <URL:https://link.springer.com/article/10.1186/s12882-022-02993-3/fulltext.html> DOI: 10.1186/s12882-022-02993-3 * |
| RODRIGUEZ ET AL., SEMIN DIAL, vol. 22, no. 4, July 2009 (2009-07-01), pages 363 - 8 |
| SPRAGUE ET AL.: "Extended-release calcifediol for secondary hyperparathyroidism in stage 3-4 chronic kidney disease", EXPERT REVIEW OF ENDOCRINOLOGY & METABOLISM, vol. 12, 2017, pages 289 - 301, XP055700728, DOI: 10.1080/17446651.2017.1347501 |
| SPRAGUE ET AL.: "Use of extended-release calcifediol to treat secondary hyperparathyroidism in stages 3 and 4 chronic kidney disease", AM J NEPHROL, vol. 44, 2016, pages 316 - 325, XP055700718, DOI: 10.1159/000450766 |
| SPRAGUE SM ET AL., AM J NEPHROL, vol. 44, 2016, pages 316 - 25 |
| STRUGNELL SACSOMOR PASHFAQ ABISHOP CW: "Initial evaluation of high-dose extended-release calcifediol (ERC) in patients with stage 5 CKD on hemodialysis", AMERICAN SOCIETY OF NEPHROLOGY 2021 ANNUAL MEETING |
| STUART M. SPRAGUE ET AL: "Extended-release calcifediol for secondary hyperparathyroidism in stage 3-4 chronic kidney disease", EXPERT REVIEW OF ENDOCRINOLOGY & METABOLISM, vol. 12, no. 5, 11 July 2017 (2017-07-11), GB, pages 289 - 301, XP055700728, ISSN: 1744-6651, DOI: 10.1080/17446651.2017.1347501 * |
| STUART M. SPRAGUE ET AL: "Modified-Release Calcifediol Effectively Controls Secondary Hyperparathyroidism Associated with Vitamin D Insufficiency in Chronic Kidney Disease", AMERICAN JOURNAL OF NEPHROLOGY, vol. 40, no. 6, 1 January 2014 (2014-01-01), CH, pages 535 - 545, XP055275509, ISSN: 0250-8095, DOI: 10.1159/000369939 * |
| VO, V.SPRAGUE, S.M: "Chronic Kidney Disease", 2020, SPRINGER, CHAM, article "Parathyroid Hormone as a Uremic Toxin" |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US11452734B2 (en) | Method of treating and preventing secondary hyperparathyroidism | |
| Sprague et al. | Paricalcitol versus calcitriol in the treatment of secondary hyperparathyroidism | |
| AU2025259887A1 (en) | Method of controlling progression of hyperparathyroidism with calcifediol, and compositions for use therein | |
| WO2025094144A1 (en) | Controlling loss of kidney function | |
| Arenas et al. | Consequences of the implementation of “K/DOQI Clinical Practice Guidelines for Bone Metabolism and Disease in Chronic Kidney Disease” in a population of patients on chronic hemodialysis | |
| de Paz et al. | Acute and long-term effects of zoledronate in adult patients with osteogenesis imperfecta. An observational Spanish study with five years of follow-up | |
| Chan et al. | Vitamin D therapy (supplementation) in early chronic kidney disease | |
| El-Reshaid et al. | The beneficial effect of paricalcitol (Zemplar) on secondary hyperparathyroidism refractory to calcitriol (Calcijex) and cinacalcet HCl (Mimpara) in patients on maintenance hemodialysis: a prospective and crossover study | |
| US20050239756A1 (en) | Methods of treating various vitamin D metabolism conditions with 1alpha-hydroxyvitamin D2 | |
| Fouda et al. | Successful Story of Conversion of Cinacalcet to Etelcalcetide During the COVID-19 Pandemic: TH-PO175 | |
| Kim et al. | A Review of Extended-Release Calcifediol in Patients with Chronic Kidney Disease Stage 3 and 4 and in Healthy Patients | |
| HK40063649A (en) | Method of controlling progression of hyperparathyroidism with calcifediol, and compositions for use therein | |
| WO2017205939A1 (en) | Vitamin d formulations and therapeutic uses thereof | |
| HK1193049A (en) | Therapy using vitamin d repletion agent and vitamin d hormone replacement agent |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 24812568 Country of ref document: EP Kind code of ref document: A1 |