Aty. Docket. No.018716.208867 (WN3426) A POLYGONATUM POLYSACCHARIDE COMPOSITION FOR TREATING ULCERATIVE COLITIS AND METHOD OF PREPARING THE POLYSACCHARIDE COMPOSITION CROSS-REFERENCE TO RELATED APPLICATIONS [0001] This application claims the benefit of Chinese Application No.202311376930.2, filed on 23 October 2023, the disclosure of which is incorporated herein by reference in its entirety. FIELD OF THE INVENTION [0002] The present invention relates generaly to compositions and methods for treating ulcerative colitis and, more specificaly, to a method of preparing a Polygonatum polysaccharide composition for treating ulcerative colitis (UC) in a subject. The Polygonatum polysaccharide composition of the present invention comprises, consists essentialy of, or consists of, an isolated polysaccharide. BACKGROUND OF THE INVENTION [0003] Ulcerative colitis (UC) is a chronic, nonspecific, complex gastrointestinal inflammatory bowel disease (IBD) with unknown etiology and having the characteristics of chronic progression, long course of disease, repeated atack and the like, and is characterized by local inflammatory cel infiltration, goblet cel disappearance and soluble inflammatory medium aggregation of intestinal tract as pathological features. The incidence and prevalence rates of UC in China have continuously increased in the past 20 years and are closely related to the occurence of colon cancer, and the treatment of UC has become a clinicaly intractable problem. Also, historical, and epidemiological studies have demonstrated that the incidence of UC can be linked to a Western high-fat diet. [0004] Curently, an effective treatment method for UC is lacking. One treatment approach is to step-up from 5-aminosalicy-late folowed by thiopurines, steroids, and anti-tumor necrosis factor therapy, such as vedolizumab, ustekinumab, tofacitinib, tacrolimus and cyclosporine. However, most current drugs aim to suppress inflammatory mediators and cannot achieve long- term therapeutic effects. For example, while the application of glucocorticoid, salicylic acid preparations and immunosuppressants can relieve symptoms, the effect is not satisfactory, and the long-term application can cause serious adverse reactions. In addition to serious side effects, the expense of treatment limit efective and sustained treatment, and patients are often under remarkable physical, psychological, and financial stress. Due to the high cost of therapeutic drugs as wel as the side efects, developing new therapeutic strategies to prevent and/or treat UC is essential and the search for efective, low-toxicity drugs is imminent.
Aty. Docket. No.018716.208867 (WN3426) [0005] With the progress of UC basic research, the focus of treatment is on an important link to pathogenesis. The abnormal barrier function of the intestinal mucosa is an important link for the onset of UC, and the improvement of the barier function of the intestine is one of the targets pursued by the treatment of UC. The intestinal mucosa barier function is damaged due to the expression defect of the intestinal eflux transporter P-glycoprotein (P-gp), and colitis similar to UC is induced by the invasion of the disordered intestinal flora and the metabolites thereof. The single layer of epithelial cels constitutes the first intestine line of defense. Chronic inflammation of the gastrointestinal tract represented as UC show intestinal barier dysfunction, with a subsequent increase in permeability. [0006] Natural polysaccharides are of wide interest to researchers because of their high content as wel as low toxicity. In recent years, Chinese medicinal polysaccharide has atracted much atention as one of the main active ingredients of Chinese medicines. The traditional Chinese medicine polysaccharide can play a role in treating UC by resisting inflammation, reducing excessive immune response of intestinal tracts, regulating intestinal tract microecological disorder and the like. As a traditional medicine-food homologous plant, the Polygonatum is various in varieties and wide in distribution, has a medical and edible history of thousands of years, can resist tumors, and has antiviral, antibacterial, antiaging, blood sugar lowering, and blood lipid reducing efects. Polygonatum cyrtonema Hua (P. cyrtonema) has been used as a traditional tonic food and herb in China. The multiple bioactive compounds of P. cyrtonema made it widely used in therapeutic diseases, such as anti-osteoporotic, anti-fatigue, cough, and other respiratory problems. Further exploration was applied to faty liver disease, diabetes melitus, Alzheimer’s disease, and cancer. Such potential health benefits are closely atributed to the secondary metabolites of P. cyrtonema. Polysaccharides, saponins, flavonoids, and phenols are the major ingredients of the secondary metabolite of P. cyrtonema, and the most frequently studied. Previous studies have shown that the potential health benefits of P. cyrtonema may be atributed to its bioactive components such as polysaccharides, steroidal saponins, flavonoids, and phenols. Of these, polysaccharides appear to be one of the major biologicaly active components. The quality of P. cyrtonema has been evaluated in Chinese Pharmacopoeia based on the polysaccharide content, which has been defined as a minimal content of 7%. [0007] Modern pharmacological research proves that the Polygonatum polysaccharide has various pharmacological efects of resisting aging, resisting tumors, reducing blood sugar, reducing blood fat, preventing arteriosclerosis, resisting bacteria, improving the immunity of the
Aty. Docket. No.018716.208867 (WN3426) organism and the like. So far, however, the prevention and treatment advantages of Polygonatum polysaccharide in intestinal diseases are not fuly utilized. [0008] In view of the foregoing, there remains an opportunity to provide new and useful polysaccharide compositions and methods of obtaining Polygonatum polysaccharide compositions from Polygonatum. Such Polygonatum polysaccharide compositions can ofer potential preventive and therapeutic approaches for preventing and/or treating UC. BRIEF SUMMARY OF THE INVENTION [0009] A method of preparing a Polygonatum polysaccharide composition is disclosed. The Polygonatum polysaccharide composition may also be refered to as a composition comprising, optionaly consisting essentialy of, or optionaly consisting of, one or more polysaccharides obtained or isolated from Polygonatum. [0010] The method includes performing a first extraction of Polygonatum material with a first solvent composition to obtain a first residue and a first supernatant, performing a second extraction of the first residue with a second solvent composition to obtain a second residue and a second supernatant, performing a third extraction of the second residue to obtain a third residue and a third supernatant, folowed by performing precipitation of the third supernatant with a fourth solvent composition to obtain crude polysaccharides, performing elution of the crude polysaccharides with a first eluent composition to obtain a first eluate, and performing filtration of the first eluate. [0011] An isolated polysaccharide is also disclosed. The isolated polysaccharide is obtained from Polygonatum material. For example, the isolated polysaccharide can be obtained via the preparation method above. The isolated polysaccharide comprises a repeat unit having the folowing structure: β-Galp(1

6 →4)–β-Galp(1→4)–β-Galp(1→4)–[β-Galp(1→4)]x–β-Galp(1→ wherein each β-Galp is β-D-galactopyranose (or Beta-D-galactopyranose), and subscript x is an integer from 1 to 5. [0012] In various embodiments, the Polygonatum polysaccharide composition and the isolated polysaccharide are the same, or they are essentialy the same. Thus, reference to the Polygonatum polysaccharide composition herein may be interchangeable with reference to the isolated polysaccharide herein, and vice versa.
Aty. Docket. No.018716.208867 (WN3426) [0013] In various embodiments, the Polygonatum material is a rhizome of Polygonatum. Also, prior to performing the first extraction, the Polygonatum material may be steamed or scalded. In other embodiments, the Polygonatum material is not steamed or scalded. [0014] A Polygonatum polysaccharide composition obtained by the method, an oral composition for oral administration to a subject and including the Polygonatum polysaccharide composition, a use of the Polygonatum polysaccharide composition in preparing a medicine (or medicament) for treatment of ulcerative colitis (UC) in a subject, and a method of treating UC in a subject are also provided. The method includes administering an efective amount of the composition to the subject, the composition being the Polygonatum polysaccharide composition or the oral care composition. In various embodiments, the composition is administered oraly to the subject. BRIEF DESCRIPTION OF THE DRAWINGS [0015] Figure 1 is a flow chart showing a method of preparing a Polygonatum polysaccharide composition in accordance with embodiments of the disclosure; [0016] Figure 2 is a plot showing a high-performance liquid chromatogram (HPLC) (refractive index detector, super hydrogel linear chromatographic column KS-804 and KS-802 connected in series) of the Polygonatum polysaccharide composition; [0017] Figure 3 is a plot showing the Fourier-transform infrared spectroscopy (FT-IR) spectrum of the Polygonatum polysaccharide composition; [0018] Figure 4 are plots showing the gas chromatography (GC) spectrum of the Polygonatum polysaccharide composition ilustrating a monosaccharide analysis of the composition; [0019] Figure 5 are plots showing the GC spectrum of the Polygonatum polysaccharide composition after methylation; [0020] Figure 6a is plot showing the1H nuclear magnetic resonance (NMR) spectrum of the Polygonatum polysaccharide composition; [0021] Figure 6b is a plot showing the13C NMR spectrum of the Polygonatum polysaccharide composition; [0022] Figure 6c is a plot showing the13C distortion-less enhancement by polarization transfer (DEPT) NMR spectrum of the Polygonatum polysaccharide composition; [0023] Figure 6d is a plot showing the heteronuclear single quantum coherence (HSQC) NMR spectrum of the Polygonatum polysaccharide composition; [0024] Figure 6e is a plot showing the heteronuclear multiple bond corelation (HMBC) NMR spectrum of the Polygonatum polysaccharide composition;
Aty. Docket. No.018716.208867 (WN3426) [0025] Figure 6f is a plot showing the1H-1H corelation spectroscopy (COSY) NMR spectrum of the Polygonatum polysaccharide composition; [0026] Figure 6g is a schematic ilustration showing the chemical structure of the Polygonatum polysaccharide composition; [0027] Figure 6h is another schematic ilustration showing the chemical structure of the Polygonatum polysaccharide composition; [0028] Figure 7a is a chart showing the experimental design of a study of the efects of the Polygonatum polysaccharide composition for treating an induced ulcerative colitis (UC) condition; [0029] Figure 7b is a plot showing the body weight percent of the test subjects as a function of time; [0030] Figure 7c is a plot showing the disease activity index (DAI) of the test subjects as a function of time; [0031] Figure 7d is a plot showing the colon length of the test subjects after completion of the treatment period; [0032] Figure 7e is an image of the colon histomorphology of the test subjects after completion of the treatment period; [0033] Figure 7f is a plot showing the spleen index of the test subjects after completion of the treatment period; [0034] Figure 7g is an image of the spleen morphology of the test subjects after completion of the treatment period; [0035] Figure 8 are plots showing the gas chromatography (GC) spectrum of the Polygonatum polysaccharide composition (PSP-W-1) ilustrating a monosaccharide analysis of the composition (1. rhamnose; 2. fucose; 3. arabinose; 4. xylose; 5. mannose; 6. glucose; 7. galactose; 8. L-chiro-inositol); [0036] Figure 9 are plots showing the gas chromatography (GC) spectrum of a D-Galactose standard, an L-Galactose standard, absolute configurations standards, and the Polygonatum polysaccharide composition (PSP-W-1); [0037] Figure 10 is a plot showing mass spectra of partialy methylated alditol acetates (PMMAs) for T-Galp; [0038] Figure 11 is a plot showing mass spectra of partialy methylated alditol acetates (PMMAs) for 1,4-Galp;
Aty. Docket. No.018716.208867 (WN3426) [0039] Figure 12 is a plot showing mass spectra of partialy methylated alditol acetates (PMMAs) for 1,4,6-Galp; [0040] Figure 13 is a plot showing two-dimensional NMR spectra of the Polygonatum polysaccharide composition; [0041] Figure 14 is a plot showing the thymus index of the test subjects after completion of the treatment period; [0042] Figure 15 is a plot showing the histological score of the colon of the test subjects after completion of the treatment period; [0043] Figure 16 is are images of the histomorphology of the colon of the test subjects after completion of the treatment period; [0044] Figure 17 is a plot showing the inhibition of mRNA expression of the inflammatory mediator iNOS of the test subjects after completion of the treatment period (***p <0.001 compared with the DSS group, ###p <0.001 compared with the control group); [0045] Figure 18 is a plot showing the inhibition of mRNA expression of the inflammatory mediator COX-2 of the test subjects after completion of the treatment period (*p <0.05 and ***p <0.001 compared with the DSS group, ###p <0.001 compared with the control group); [0046] Figure 19 is a plot showing the inhibition of mRNA expression of the inflammatory factor IL-6 of the test subjects after completion of the treatment period (*p <0.05 and **p <0.001 compared with the DSS group, ###p <0.01 compared with the control group); [0047] Figure 20 is a plot showing 16S rRNA sequencing analysis of gut microbiota regulated by the Polygonatum polysaccharide composition of the test subjects after completion of the treatment period, using the Sobs index; [0048] Figure 21 is a plot showing 16S rRNA sequencing analysis of gut microbiota regulated by the Polygonatum polysaccharide composition of the test subjects after completion of the treatment period, using the Ace index; [0049] Figure 22 is a plot showing 16S rRNA sequencing analysis of gut microbiota regulated by the Polygonatum polysaccharide composition of the test subjects after completion of the treatment period, using the Chao index; [0050] Figure 23 is a plot showing 16S rRNA sequencing analysis of gut microbiota regulated by the Polygonatum polysaccharide composition of the test subjects after completion of the treatment period, using the Shannon index; [0051] Figure 24 is a plot showing principal coordinates analysis (PCoA) of gut microbiota of the test subjects after completion of the treatment period;
Aty. Docket. No.018716.208867 (WN3426) [0052] Figure 25 is a plot showing Nonmetric multidimensional scaling (NMDS) of gut microbiota of the test subjects after completion of the treatment period; [0053] Figure 26 is a plot showing a Venn diagram for 16S rRNA sequencing analysis of gut microbiota of the test subjects after completion of the treatment period; [0054] Figure 27 is a plot showing a community histogram of the microbial compositional profiling of the test subjects after completion of the treatment period at the phylum level; [0055] Figure 28 is a plot showing the relative abundance of microbial compositional profiling of the test subjects after completion of the treatment period at a family level; [0056] Figure 29 is a plot showing linear discriminant analysis (LDA) of gut microbiota of the test subjects after completion of the treatment period indicating the significantly abundant genus; [0057] Figure 30 is another plot showing linear discriminant analysis (LDA) of gut microbiota of the test subjects after completion of the treatment period indicating the significantly abundant genus; [0058] Figure 31 is a plot showing the relative abundance of gut microbiota of the test subjects that is altered at the phylum level after completion of the treatment period (*p < 0.05, **p < 0.01); and [0059] Figure 32 is a plot showing the relative abundance of gut microbiota of the test subjects that is altered at the genus level after completion of the treatment period (*p < 0.05, **p < 0.01). DETAILED DESCRIPTION OF THE CURRENT EMBODIMENTS [0060] A method of preparing a Polygonatum polysaccharide composition is disclosed (which may be refered to herein simply as “the composition”). An isolated polysaccharide is also disclosed. The composition comprises at least one botanical active component. The at least one botanical active component can be the isolated polysaccharide. The composition and its method of preparation is described below, folowed by description of the isolated polysaccharide, and associated uses and methods. The Polygonatum polysaccharide composition comprises, consists essentialy of, or consists of, the isolated polysaccharide. [0061] As wil be understood in view of this disclosure, the composition is not particularly limited aside from the botanical active component (e.g., the isolated polysaccharide) and, in particular, the extract(s) thereof, as wel as the related methods of obtaining the botanical active component. As such, the composition may be formulated, for example, as or in an oral composition or as or in a nutraceutical, pharmaceutical, or supplement, and may be utilized as a unique and stand-alone therapeutic or in combination with other therapeutics compatible therewith.
Aty. Docket. No.018716.208867 (WN3426) [0062] The composition is useful for treating, preventing, and/or ameliorating various conditions, such as those associated with ulcerative colitis (UC). Specificaly, as wil be appreciated in view of the description and examples below, the composition of the present embodiments is believed to be capable of preventing, inhibiting and/or treating ulcerative colitis. [0063] As such, the composition may be utilized to treat (i.e., slow, prevent, reverse, etc.) conditions generaly associated with ulcerative colitis, such as but not limited to reducing inflammatory UC factors, improve the repair of intestinal mucosa bariers, improve the intestinal barier function, regulate the metabolism of intestinal flora, and improve the expression of P- glycoprotein. [0064] As introduced above, the composition comprises a botanical active component. More specificaly, the botanical active component comprises, optionaly consists essentialy of, or optionaly consists of, at least one extract of a plant from the genus Polygonatum. In various embodiments, the botanical active component consists of a single isolated extract of a plant from the genus Polygonatum as described herein. As introduced above, the botanical active component can be the isolated polysaccharide as described herein. Polygonatum [0065] The botanical active component, and thus the composition, comprises the extract of Polygonatum rhizome, i.e., an extract comprising, optionaly consisting essentialy of, or optionaly consisting of, material from the plant genus Polygonatum. The Polygonatum rhizome extract is not particularly limited, and may comprise or be any rhizome extract or combination of rhizome extracts from a Polygonatum plant obtained by the methods disclosed herein and suitable for use in the embodiments herein. More specificaly, exemplary Polygonatum rhizome extracts include those capable of inhibiting UC or eliciting/exhibiting any other such activities described herein as part of the botanical active component. [0066] Polygonatum has been reported to contain various bioactive components, such as polysaccharides, steroidal saponins, flavonoids, and phenols. In various embodiments, the Polygonatum is Polygonatum cyrtonema Hua, which may simply be refered to as Polygonatum cyrtonema, P. cyrtonema, or via various other names such as Polygonatum Multiflorum, King Solomon’s seal, Solomon’s seal, Manyflower Solomonseal, Polygonati Rhizoma, “Duo Hua Huang

[0067] In certain embodiments, the extract of the plant of the genus Polygonatum or the species Polygonatum cyrtonema is obtained by extracting rhizome or rhizome-based plant material of Polygonatum. The rhizome may be fresh or dried, typicaly dried to prevent decay. The dried
Aty. Docket. No.018716.208867 (WN3426) rhizome may then be formed into a powder, which is further processed to form the extract as like described below. [0068] In various embodiments, the botanical active component consists of the extract of Polygonatum rhizome. In further or other embodiments, the composition is substantialy to completely free of components obtained from non-rhizome-based plant material of Polygonatum. In these embodiments, the non-rhizome-based plant material of Polygonatum can be, for example, the flower, stem, or leaf of a Polygonatum plant. Without being bound by any particular theory, it is believed that the rhizome of Polygonatum is most useful for anti-ulcerative colitis efect; whereas other parts of Polygonatum are not. [0069] Suitable rhizome extracts may be processed (e.g., defated, partialy defated, ground, dried, precipitated, washed, filtered, mesh-sorted, extracted, distiled, concentrated, etc.) to obtain the Polygonatum rhizome extract. Likewise, Polygonatum rhizome may be extracted in raw form, or processed prior to extraction of the Polygonatum rhizome extract (e.g., used in raw form, suspended form, dehydrated form, concentrated form, etc.). [0070] The amount of the Polygonatum extract utilized in the botanical active component may vary, and wil be selected based on the number and types of components being utilized in the botanical active component. In certain embodiments, the botanical active component comprises from 1 to 2000 mg of the Polygonatum extract, such as from 1 to 1000, optionaly of from 2 to 800, optionaly of from 20 to 750, or optionaly of from 50 to 500, mg. However, amounts outside these ranges may also be utilized. For example, in certain embodiments, the botanical active component includes the Polygonatum extract in an amount of at least 1, optionaly of at least about 20, optionaly of at least about 50, optionaly of at least 100, optionaly of at least 250, optionaly of at least 500, optionaly of at least 1000, or optionaly of at least 1500, mg. In these or other embodiments, an upper boundary may be selected such that the botanical active component comprises the Polygonatum extract in an amount of ≤100, ≤250, ≤500, ≤750, ≤1000, ≤2000, ≤5000 mg. In various embodiments, the botanical active component can include an amount of Polygonatum extract optionaly in an amount of greater than 1, optionaly greater than 5, optionaly greater than 10, optionaly greater than 25, optionaly greater than 50, optionaly greater than 75, optionaly greater than 80, or optionaly greater than 95, wt.%, based on the total weight of the botanical active component. In such embodiments, an upper boundary may be selected to be generaly ≤10, ≤20, ≤30, ≤40, ≤50, ≤60, ≤70, ≤80, ≤90, and ≤99 wt.%, respectively, based on the total weight of the botanical active component.
Aty. Docket. No.018716.208867 (WN3426) [0071] In certain embodiments, the botanical active component comprises more than one Polygonatum extract, such as 2, 3, 4, or more Polygonatum extracts. In such embodiments, each Polygonatum extract is independently selected, may be the same as or diferent from any other Polygonatum extract, and each utilized in an amount as described above. [0072] The Polygonatum extract may be utilized in any form, such as neat (i.e., absent solvents, carier vehicles, diluents, etc.), or disposed in a carier vehicle, such as a solvent or dispersant. The carrier vehicle, if present, may comprise an aqueous solvent (e.g., water), an organic solvent, fluid, or oil, or the like, or combinations thereof. When utilized, the carier vehicle wil be selected based on the particular components of the botanical active component and/or the composition, such as the particular Polygonatum extract(s) utilized. It wil be appreciated that the Polygonatum extract may be combined with the carrier vehicle, if utilized, prior to, during, or after being combined with any other components of the botanical active component and/or composition. Method of Preparing the Polygonatum Polysaccharide Composition [0073] With reference to Figure 1, the method of preparing a Polygonatum polysaccharide composition generaly includes extraction of Polygonatum material, folowed by separation of the obtained crude extract and subsequent purification. In specific embodiments of the method, the Polygonatum material is the rhizome of Polygonatum plants, and the Polygonatum rhizome material is first steamed prior to the extraction steps. For example, the fresh rhizome of Polygonatum plants can be steamed or scalded with hot water for a time period of minutes, then dried and subsequently cut into pieces or alternatively kept whole. However, in other embodiments the Polygonatum rhizome material is not steamed or scalded before beginning the extraction. In yet other embodiments, other species of Polygonatum such as Polygonatum kingianum Col. et Hemsl. (Solomon’s seal, Dian Huang Jing,滇黄精) or Polygonatum sibiricum Red. (Siberian Solomon’s seal, Huang Jing,黄精) may be used instead of or in addition to Polygonatum cyrtonema. In various embodiments, the Polygonatum material is obtained from a plant selected from: i) Polygonatum kingianum; i) Polygonatum sibiricum; ii) Polygonatum cyrtonema; or iv) a combination of two or more of i) to ii). [0074] The extraction of polysaccharides from the Polygonatum material includes a series of extraction steps to obtain an extract. The term “extract” is used herein in the conventional sense to refer to a composition that has been obtained via fluid extraction from a source material. As such, the term “extract” or “botanical extract” is to be understood as a composition obtained via fluid extraction (e.g., solvent extraction, gas extraction, CO2 extraction, etc.) from a botanical
Aty. Docket. No.018716.208867 (WN3426) source (i.e., a plant material). Extraction methods include water extractions, steam extractions, solvent extractions, etc. as known in the art. Exemplary extraction techniques include solvent extraction, e.g., via use of a polar solvent such as an alcohol (e.g., methanol, ethanol, butylene glycol, etc.), ether (e.g., diethyl ether, methyl tert butyl ether, etc.), ketone (e.g., acetone), ester (e.g., ethyl acetate), phenol, water, and the like, a nonpolar solvent such as benzene, xylenes, toluene, etc., as wel as derivatives, modifications, and combinations thereof (e.g., solvent-water blends, including alcohol-water, acetone-water, etc.). Additional and alternative extraction techniques include sequential fractionations, total hydro-ethanolic extractions, lump-sum extractions, supercritical fluid extractions (e.g., with CO2), and the like, as wel as those utilizing sequential or secondary extractions from a first extract (e.g., a non-polar solvent extract of a botanical extract obtained from a polar solvent extraction) or other processing techniques such as filtration, purification, distilation, dehydration, evaporation, concentration, drying, etc. Specific examples of suitable extraction methods are described in U.S. Patent No.7,897,184, which is incorporated herein by reference. [0075] In one example, the botanical extracts can be obtained using an organic solvent extraction technique. In another example, solvent sequential fractionation can be used to obtain the botanical extracts. Total hydro-ethanolic extraction techniques can also be used to obtain the botanical extracts. Generaly, this is refered to as a lump-sum extraction. The botanical extract generated in the process wil contain a broad variety of phytochemicals present in the extracted material including fat and water-soluble phytochemicals. Folowing colection of the botanical extract solution, the solvent wil be evaporated, resulting in the botanical extract. [0076] Total ethanol extraction may also be used. This technique uses ethanol as the solvent. This extraction technique generates a botanical extract that may include fat soluble and/or lipophilic compounds in addition to water-soluble compounds. Total methanol extraction may also be used in a similar manner with similar results. [0077] Another example of an extraction technique that can be used to obtain the botanical extracts is supercritical fluid carbon dioxide extraction (SFE). In this extraction procedure, the material to be extracted is not exposed to any organic solvents. Rather, the extraction solvent is carbon dioxide (CO2), with or without a modifier, in super-critical conditions (e.g., >31.3°C and >73.8 bar). Those of skil in the art wil appreciate that temperature and pressure conditions can be varied to obtain the best yield of botanical extract. This technique generates a botanical extract of fat soluble and/or lipophilic compounds, similar to total hexane and ethyl acetate extraction techniques, which may also be used.
Aty. Docket. No.018716.208867 (WN3426) [0078] Each of the extraction methods above also may include and/or be utilized in combination with one or more additional processing steps understood in the art. For example, plant material may be comminuted, smashed, ground, etc. There also may be one or more filtration steps to remove, for example, celulosic/fibrous or other solid materials. There also may be one or more purification steps to remove, for example, certain constituents and/or contaminants. Such purification may be accomplished, for example, by distilation, evaporation, centrifugation, etc. There also may be one or more concentration and/or drying steps to remove water and/or other volatiles, e.g., alcohol, lighter compounds, VOCs, etc. Moreover, acids and/or bases may be added to adjust pH or neutralize. Depending on the desired form of the final/end botanical extract, one can also utilize various additional steps understood in the art, such as screening, pressing, miling, grinding, mixing, dispersing, etc. It is to be appreciated that combinations of these additional processing steps in duplicative and/or diferent orders is also contemplated. [0079] In various embodiments, the solvent used to obtain suitable botanical extracts for this disclosure is one in which the resulting botanical extract and/or a subsequent form thereof (e.g., botanical extract powder) is suitable for ingestion. For example, the solvent is water or ethanol. [0080] With further reference to Figure 1, in the present method a first extraction of the steamed or non-steamed Polygonatum material is performed with a first solvent composition to obtain a first residue (i.e., first extract) and a first supernatant. In specific embodiments, the first solvent composition includes an alcohol, preferably ethanol (EtOH). In certain embodiments, the first solvent composition is preferably 95% ethanol. Alternatively, the first solvent composition is between 85% and 97% ethanol, between 85% and 95% ethanol, between 87% and 95% ethanol, between 90% and 95% ethanol, or between 92% and 95% ethanol. Ethanol is used as a solvent to remove faty-soluble and/or semi-polar materials. However, use of other solvents is possible, such as water. In this case, water may be used as a solvent first, and then faty-soluble and/or semi-polar materials subsequently may be removed by ethyl acetate extraction or by chromatography with a microporous resin. In specific embodiments, the first extraction is performed for at least 2.0 hours, alternatively at least 2.5 hours, at least 3.0 hours, at least 3.5 hours, or at least 4.0 hours. Optionaly, the first extraction is performed for a time period in a range of 2.5 to 3.5 hours, alternatively between 2.6 and 3.4 hours, between 2.7 and 3.3 hours, between 2.8 and 3.3 hours, or between 2.9 and 3.1 hours. Preferably, the first extraction is performed for at least 3 hours. Using a high percentage (e.g., >80%) alcohol such as ethanol or methanol (or other organic solvent), the supernatant does not contain any useful
Aty. Docket. No.018716.208867 (WN3426) amount of polysaccharide, whereas the residue (i.e., first residue) of the extraction solution obtained by filtration or centrifugation includes polysaccharides and is kept for the next step in the method. The first supernatant therefore may be discarded. [0081] Folowing the first extraction, a second extraction of the first residue (obtained by the first extraction) is performed with a second solvent composition to obtain a second residue (i.e., second extract) and a second supernatant. In specific embodiments, the second solvent composition includes an alcohol, preferably ethanol (EtOH). In certain embodiments, the second solvent composition is preferably 70% ethanol. Alternatively, the second solvent composition is between 50% and 90% ethanol, between 55% and 85% ethanol, between 60% and 80% ethanol, between 65% and 75% ethanol, or between 67% and 73% ethanol. However, use of other solvents is possible, such as water. In this case, water may be used as a solvent first, and then faty-soluble and/or semi-polar materials subsequently may be removed by ethyl acetate extraction or by chromatography with a microporous resin. In specific embodiments, the first extraction is performed for at least 2.0 hours, alternatively at least 2.5 hours, at least 3.0 hours, at least 3.5 hours, or at least 4.0 hours. Optionaly, the first extraction is performed for a time period in a range of 2.5 to 3.5 hours, alternatively between 2.6 and 3.4 hours, between 2.7 and 3.3 hours, between 2.8 and 3.3 hours, or between 2.9 and 3.1 hours. Preferably, the first extraction is performed for at least 3 hours. Using a high percentage (e.g., >70%) alcohol such as ethanol or methanol (or other organic solvent), the supernatant does not contain any useful amount of polysaccharide, whereas the residue (i.e., second residue) of the extraction solution obtained by filtration or centrifugation includes polysaccharides and is kept for the next step in the method. The second supernatant therefore may be discarded. [0082] Folowing the second extraction, a third extraction of the second residue (obtained by the second extraction) is performed with a third solvent composition to obtain a third residue (i.e., second extract) and a third supernatant. In specific embodiments, the third solvent composition is water. However, use of other solvent compositions is possible, for example compositions including an alcohol such as ethanol, e.g., 0-40% alcohol in water, or another organic solvent such as acetone, ethyl acetate, or the like. In specific embodiments, the third extraction is performed for at least 1.0 hours, alternatively at least 1.5 hours, alternatively at least 2.0 hours, alternatively at least 2.5 hours, at least 3.0 hours, at least 3.5 hours, or at least 4.0 hours. Optionaly, the third extraction is performed for a time period in a range of 1.0 to 3.5 hours, alternatively between 1.5 and 3.5 hours, between 2.0 and 3.5 hours, between 2.5 and 3.5 hours, or between 2.8 and 3.2 hours. Preferably, the third extraction is performed for at least 1
Aty. Docket. No.018716.208867 (WN3426) hour. The length of the extraction time is dependent upon the size of the rhizome pieces of plant material, i.e., whether the rhizome is cut into smaler pieces and how smal the pieces are. [0083] Folowing the third extraction, precipitation of the third supernatant is performed with a fourth solvent composition to obtain crude polysaccharides. Before this precipitation, preferably the concentration of the third supernatant is increased to a higher Brix, i.e., a higher content of soluble solid polysaccharides. In specific embodiments, the fourth solvent composition includes an alcohol, preferably ethanol (EtOH). In certain embodiments, the fourth solvent composition is preferably 95% ethanol. Alternatively, the fourth solvent composition is between 60% and 97% ethanol, between 65% and 95% ethanol, between 70% and 95% ethanol, between 75% and 95% ethanol, between 80% and 95% ethanol, between 85% and 95% ethanol, between 90% and 95% ethanol, between 92% and 97% ethanol, or between 95% and 97% ethanol. The more amount of ethanol added to the concentrated third supernatant, the greater the amount of polysaccharide that is deposited/precipitated. Typicaly, polysaccharide deposits first appear when the mixture (third supernatant plus added fourth solvent composition (e.g., 95% ethanol) has an ethanol content in the range of 35% to 50% ethanol. The fourth solvent composition (e.g., 95% ethanol) is then further added to the mixture until the final ethanol concentration of the mixture is in the range of 60% to 85% ethanol. At this point, a suficient amount of polysaccharide wil have been precipitated from the third supernatant. It should be understood that use of other solvent compositions is also possible, such as those containing an alcohol other than ethanol. [0084] In certain embodiments, the third extraction is repeated at least one additional time such that the third extraction includes two or three or more similar or nearly identical sub-extraction steps. For example, the third extraction may be performed three consecutive times. In these particular embodiments, the third extraction includes performing a third extraction of the second residue with a third solvent composition to obtain a third residue and a third supernatant; performing a fourth extraction of the third residue with the third solvent composition to obtain a fourth residue and a fourth supernatant; and performing a fifth extraction of the fourth residue with the third solvent composition to obtain a fifth residue and a fifth supernatant. Preferably, the third solvent composition is water. However, the third solvent composition may be a solvent other than water, for example an alcohol such as ethanol at various concentrations in the range of 1% to 95% ethanol. In other alternative embodiments, the solvent composition used for the third extraction, the fourth extraction, and the fifth extraction may not be the same for each of these extractions steps, e.g., the solvent composition for at least one of the third extraction, the
Aty. Docket. No.018716.208867 (WN3426) fourth extraction, and the fifth extraction may be water, while the solvent composition for at least one other of the third extraction, the fourth extraction, and the fifth extraction may be an alcohol such as ethanol. Each of the third extraction, the fourth extraction, and the fifth extraction may be performed for the same or similar amount of time. Alternatively, the third extraction, the fourth extraction, and the fifth extraction are performed for diferent amounts of time. In specific embodiments, each of the third extraction, the fourth extraction, and the fifth extraction is independently performed for at least 1.0 hours, alternatively at least 1.5 hours, at least 2.0 hours, at least 2.5 hours, at least 3.0 hours, at least 3.5 hours, or at least 4.0 hours. Optionaly, each of the third extraction, the fourth extraction, and the fifth extraction is independently performed for a time period in a range of 1.0 to 3.5 hours, alternatively between 1.5 and 3.5 hours, between 2.0 and 3.5 hours, between 2.5 and 3.5 hours, or between 2.8 and 3.2 hours. Preferably, each of the third extraction, the fourth extraction, and the fifth extraction is performed for at least 1 hour. After the fifth extraction, precipitation of the fifth supernatant is performed with a fourth solvent composition (as described above) to obtain crude polysaccharides. More preferably, in other embodiments after the fifth extraction, the third supernatant, the fourth supernatant, and the fifth supernatant are combined, and precipitation of this mixture is performed with a fourth solvent composition as described above. Further, the mixture of the third supernatant, the fourth supernatant, and the fifth supernatant may be concentrated to a certain Brix prior to precipitation with the fourth solvent composition. [0085] Subsequent to the extraction steps above, the obtained crude polysaccharides are separated and/or purified to isolate and/or concentrate the desired polysaccharide(s) from the crude polysaccharides. At this stage of the method, in various embodiments the crude polysaccharides are subjected to a separation process. The separation process is not particularly limited, and may include, for example, use of membrane separation technology, gel filtration chromatography, molecular-exclusion chromatography, and/or microporous resin chromatography on a micro or macro scale. For example, on a macro scale, membrane separation technology or gel filtration chromatography may be used. In specific embodiments, subsequent to the extraction steps, elution of the crude polysaccharides is first performed with a first eluent composition to obtain a first eluate. The first eluent composition is preferably water, and the first eluate (“PSP-W” in Figure 1) is the polysaccharide composition obtained by water elution of the crude polysaccharides. In specific embodiments, the elution of the crude polysaccharides is performed via an anion exchange column. For example, the anion exchange column is a DEAE Sepharose Fast Flow column, i.e., the anion exchange column uses DEAE Sepharose or similar
Aty. Docket. No.018716.208867 (WN3426) equivalent as the exchange media. At this step, the elution may be performed using a salt gradient wherein the first eluent composition is stepwise varied from water to salt (e.g., sodium chloride (NaCl) solutions of diferent concentrations. For example, as shown in Figure 1, the elution first may be performed with water as the eluent to obtain the first eluate (“PSP-W”), folowed by 0.1M NaCl as the eluent to obtain a second eluate (“PSP-1”), folowed by 0.2M NaCl as the eluent to obtain a third eluate (“PSP-2”), folowed by 0.5M NaCl to obtain a fourth eluate (“PSP- 5”). In particular embodiments, the first eluate (“PSP-W”) contains the desired polysaccharide(s) while the others, i.e., the second eluate (“PSP-1”), the third eluate (“PSP-2”), and the fourth eluate (“PSP-5”), do not. Therefore, the second eluate (“PSP-1”), the third eluate (“PSP-2”), and the fourth eluate (“PSP-5”) may be discarded. [0086] Folowing the separation described above, the first eluate (“PSP-W”) may be concentrated/purified by a further separation process such as filtration. For example, filtration of the first eluate may be performed by at least one of gel filtration chromatography, membrane separation, and microporous resin chromatography, optionaly via gel filtration chromatography or molecular-exclusion chromatography. In specific embodiments, the first eluate is subjected to gel filtration chromatography. For example, the first eluate may be filtered with a SUPERDEX® 75 or SUPERDEX® 200 gel filtration column or similar equivalent. On a macro scale, gel chromatography may likewise be used. The product of the chromatography step is the desired Polygonatum polysaccharide composition (“PSP-W-1” in Figure 1). As introduced above, the Polygonatum polysaccharide composition can be the isolated polysaccharide of the present disclosure. Isolated Polysaccharide [0087] The botanical active component, and thus the Polygonatum polysaccharide composition, comprises an isolated polysaccharide. The composition can also consist essentialy of, or consist of, the isolated polysaccharide. The isolated polysaccharide is obtained or extracted from a plant of the genus Polygonatum. For example, the isolated polysaccharide can be obtained via the method described above. The isolated polysaccharide comprises a repeat unit having the folowing structure: β-Galp(1

6) [→4]–β-Galp(1→4)–β-Galp(1→4)–(β-Galp(1→4))x–β-Galp(1→)
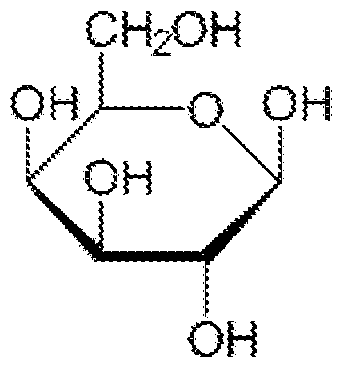
Aty. Docket. No.018716.208867 (WN3426) Each Galpisβ-D-galactopyranoseandeachsubscriptxisanintegerfrom1to5.Β-D- galactopyranose (Beta-D-galactopyranose) generaly has the folowing chemical structure:
[0088]The isolated polysaccharide is a galactan (galactosan) having 1,4-Galp and 1,4,6-Galp as the main chain. T-Galp is connected to the 6-position of 1,4,6-Galp. The 1,4,6-Galp therefore is bonded to three distinct monosaccharides in the polysaccharide chain. T-Galp can have molar ratio % relative to the total isolated polysaccharide of 10 to 20%, or optionaly 12 to 18%.1,4- Galp can have a molar ratio % relative to the total isolated polysaccharide of 60 to 80%, or optionaly 65 to 75%.1,4,6-Galp can have a molar ratio % relative to the total isolated polysaccharide of 10 to 20%, or optionaly 12 to 18%. Generaly, the isolated polysaccharide has from 5 to 20, optionaly from 8 to 18, optionaly from 10 to 16, optionaly from 12 to 14, or optionaly 13, repeat units. [0089]In some embodiments, the isolated polysaccharide has a molecular weight of from 10 to 20 kDa (kilodaltons), optionaly 12 to 18 kDa, or optionaly 14 to 16 kDa. In further or other embodiments, subscript x is from 2 to 4, or optionaly subscript x is 3. [0090]A specific embodiment of the isolated polysaccharide of this disclosure is depicted in Fig.6g*. In this embodiment, x is 3, and the number of repeat units is about 13 and/or the molecular weight is about 14.38 kDa. A further or alternate embodiment is depicted in Fig.24. Definitions [0091]In order to provide a clear and consistent understanding of the specification and claims, the folowing definitions are provided. [0092]The terms “composition” or “formulation” refer to a product that treats, improves, promotes, increases, manages, controls, maintains, optimizes, modifies, reduces, inhibits, or prevents a particular condition associated with a natural state, biological process or disease or disorder. For example, a composition or a formulation prevents, inhibits, and/or improves at least one condition of ulcerative colitis. The terms composition and formulation include, but are not limited to, pharmaceutical (i.e., drug), over-the counter (OTC), cosmetic, food, food ingredient or dietary supplement compositions that include an efective amount of an extract, at least one component thereof, or a mixture thereof. Exemplary compositions and/or formulations include
Aty. Docket. No.018716.208867 (WN3426) pils, tablets, capsules, powder, bar, beverage, or other liquid. Prefered compositions are formulated for oral administration/ingestion. [0093] As used herein, the term “efective amount” or “therapeuticaly effective amount” of a pure compound, composition, extract, extract mixture, component of the extract, and/or active agent or ingredient, or a combination thereof refers to an amount effective at dosages and for periods of time suficient to achieve a desired result. For example, the “efective amount” or “therapeuticaly efective amount” refers to that amount of a pure compound, composition, extract, botanical extract, extract mixture, botanical extract mixture, component of the extract, and/or active agent or ingredient, or a combination thereof of this invention which, when administered to a subject (e.g., mammal, such as a human), is suficient to effect treatment, such as preventing, inhibiting, or improving ulcerative colitis. The amount of a composition, extract, botanical extract, extract mixture, botanical extract mixture, component of the extract, and/or active agent or ingredient of this disclosure that constitutes an “efective amount” or “therapeuticaly efective treatment” wil vary depending on the active agent or the compound, the condition being treated and its severity, the manner of administration, the duration of treatment, or the age of the subject to be treated, but can be determined routinely by one of ordinary skil in the art having regard to his own knowledge and to this disclosure. [0094] The term “pharmaceuticaly acceptable” means those drugs, medicaments, extracts or inert ingredients, which are suitable for use in humans and lower animals without undue toxicity, incompatibility, instability, iritation, and the like, commensurate with a reasonable benefit/risk ratio. [0095] The terms “applying” and “administering” are defined as providing a composition to a subject via a route known in the art, including but not limited to intravenous, intra-arterial, oral, parenteral, buccal, transdermal, rectal, intramuscular, subcutaneous, intraosseous, transmucosal, or intraperitoneal routes of administration. In preferred embodiments, oral routes of administering the described composition are suitable. [0096] The terms “minimize,” “reduce,” “suppress,” “decrease” and/or “inhibit” refer to a decrease or reduction in ulcerative colitis and its conditions, in the presence of a botanical (or plant) ingredient or botanical extract as described herein, when compared to ulcerative colitis in the absence of a botanical ingredient or botanical extract as described herein, such as in a control sample. The degree of decrease or inhibition of ulcerative colitis wil vary with the nature and quantity of a botanical ingredient or botanical extract present, but wil be evident, e.g., as a detectable decrease in ulcerative colitis; desirably a degree of decrease greater than about 5%,
Aty. Docket. No.018716.208867 (WN3426) about 10%, about 15%, about 20%, about 25%, about 50%, about 75%, about 90%, about 95% or about 99% (or any degree of decrease in the range of from about 5% to about 99%) as compared to ulcerative colitis in the absence of the botanical ingredient or botanical extract. For example, a composition comprising a plant ingredient or plant extract of Polygonatum cyrtonema can minimize or reduce ulcerative colitis. [0097] As used herein, the term “subject” or “individual” includes mammals to which a composition may be administered. Non-limiting examples of mammals include humans, non- human primates, rodents (including transgenic and non-transgenic mice) or the like. In some embodiments, the subject is a mammal, and in some embodiments, the subject is human. Oral Compositions (or Oral Formulations) [0098] The oral composition may include any amount of the botanical active component (e.g., the isolated polysaccharide), which wil be selected based on the number and types of components being utilized in the oral composition as a whole. In general, the botanical active component is present in the oral composition in an amount efective to inhibit, prevent, or reduce UC in the subject. [0099] In certain embodiments, the oral composition comprises the botanical active component in an amount of from 1 to 5000, optionaly of from 2 to 2000 mg, optionaly of from 5 to 1750, optionaly of from 10 to 1500, optionaly of from 15 to 1250, optionaly of from 20 to 1000, optionaly of from 25 to 750, optionaly of from 30 to 500, optionaly of from 35 to 500, optionaly of from 40 to 500, optionaly of from 45 to 450, optionaly of from 50 to 450, or optionaly of from 50 to 400, mg. However, amounts outside and/or overlapping with these ranges may also be utilized. For example, it is to be appreciated that the ranges described above with respect to the amount of each botanical extract in the botanical active component may equaly apply to the amount of each botanical extract in the oral composition as a whole, such as when the botanical active component consists of but one of the botanical extracts. [00100] In general, the oral composition is not limited in terms of formulation, peripheral ingredients, form, number of functions, etc., aside from comprising the botanical active component and the botanical extract(s) thereof. Rather, the oral composition may be varied, and may be formulated in any fashion consistent with this disclosure. [00101] Typicaly, the oral composition is formulated or otherwise adapted for administration to a mammalian subject (e.g., a human). For example, in various embodiments, the oral composition is adapted to be consumed and/or oraly administered to a human subject. Thus, the oral composition is formulated for oral administration to the subject. In such embodiments, the
Aty. Docket. No.018716.208867 (WN3426) oral composition may also be refered to as an ingestible oral composition, and typicaly comprises at least one pharmaceuticaly acceptable additive in addition to the bioactive agent composition. In specific embodiments, the pharmaceuticaly acceptable additive is not naturaly occuring. In other words, the pharmaceuticaly acceptable additive is not a product of nature in these specific embodiments. In other embodiments, the pharmaceuticaly acceptable additive is selected from conventional additives understood in the art, and can be used in conventional amounts. [00102] As such, it should be appreciated that the particular additives, carriers, adjuvants, filers, etc. present in or combined with the oral composition may vary. Moreover, the physical form of the oral composition is not limited, and wil be selected based on the particular components of the oral composition, a desired use of the oral composition, etc. As such, as wil be understood in view of the description herein, the oral composition may be formulated as a liquid, dry powder, suspension, emulsion, gel, paste, etc., and combinations thereof. In certain embodiments, the oral composition is formulated as a sterile, non-pyrogenic liquid solution or suspension, a coated capsule, a suppository, a lyophilized powder, a transdermal patch, a softgel, or other forms are known. Other examples of suitable forms include solids, gels, liquids, powders, or other such preparations where the botanical active component is disposed in an appropriate carier vehicle, such as any of those described herein. [00103] The oral composition can be prepared using various methods. For example, actives of the oral composition (such as the botanical extract(s), and optionaly one or more inactives (such as one or more conventional components, additives, excipients, etc.), can be mixed or blended and compressed or compounded utilizing various techniques understood in the art. The oral composition of this disclosure is not limited to a particular order of manufacturing steps or method of manufacture. [00104] Excipients can be further classified as other components. Specificaly, excipients used in oral solid dosage forms have been classified based on their functionality into groups such as diluents, disintegrants, binders, compression aids, granulating agents, glidants, lubricants, release-controling polymers, stabilizers (such as antioxidants, chelators, and pH-modifiers), film-coating polymers, coating agents, vehicles, plasticizers, surfactants, colorants, sweeteners, and flavors. [00105] In various embodiments, the oral composition comprises at least one component selected from the group consisting of binders, lubricants, glidants, and combinations thereof. In certain embodiments, the oral composition includes one or more compounds including, but not
Aty. Docket. No.018716.208867 (WN3426) limited to, methylcelulose, hydroxypropyl methylcelulose, ethyl celulose, celulose acetate phthalate, acacia, gums, wax, glycerol monostearate, acrylic acid polymers and copolymers, methacrylic acid, methyl acrylate, ethyl acrylate, methyl methacrylate, ethyl methacrylate, lactose, calcium sulfate, calcium phosphate dibasic, sugar, microcrystaline celulose (MCC), starch, sodium starch glycolate, polyvinylpyrolidone, polyethylene glycol, and magnesium stearate. Combinations of such components can be utilized, and such components and other components used in conventional tablets are understood in the art. [00106] As used herein, “diluents” may be inert substances added to increase the bulk of the oral composition to make a tablet a practical size for compression. As such, they may also be refered to as bulking agents. Commonly used diluents include, but are not limited to, microcrystaline celulose (MCC), wood celulose, corn starch, modified corn starch, (tri)calcium phosphate, calcium sulfate, lactose, kaolin, mannitol, sodium chloride, dry starch, (powdered) sugar, dextrose, mannitol, sorbitol, and the like. The diluent/bulking agent may be used alone or in various mixtures, and utilized in any amount known in the art for oral compositions. [00107] As used herein, “flavoring agents” are compounds designed to give the oral composition a more palatable taste. Flavoring agents vary considerably in their chemical structure, ranging from simple esters, alcohols, and aldehydes to carbohydrates and complex volatile oils. Synthetic flavors of almost any desired type are now available and are wel known in the art. If hard taste, acid taste or biter taste derived from starting materials may be suppressed by seasoning or flavoring, the acidulant (e.g., citric acid, tartaric acid, malic acid, ascorbic acid, etc.), the sweetener (e.g., sodium saccharin, dipotassium glycyrhizinate, aspartame, stevia, thaumatin, etc.), or the perfume (e.g., various fruit perfumes containing lemon oil, orange oil or strawberry, and yoghurt, mint, menthol, etc.) may be included in the oral composition. The flavoring agent may be used alone or in various mixtures, and utilized in any amount known in the art for oral compositions. [00108] As used herein, “lubricants” are materials that perform a number of functions relating to oral compositions. In certain embodiments, like tablet manufacture, the lubricants perform one or more functions such as improving the rate of flow of the tablet granulation, preventing adhesion of the tablet material to the surface of dies and punches, reducing interparticle friction, and facilitating the ejection of the tablets from a die cavity. Examples of suitable lubricants include, but are not limited to, zinc stearate, gum arabic powder, cacao buter, carnauba wax, carmelosecalcium, carmelosesodium, caropeptide, aqueous silicon dioxide, dried aluminum
Aty. Docket. No.018716.208867 (WN3426) hydroxide gel, glycerin, magnesium silicate, light anhydrous silicic acid, light liquid parafin, crystaline celulose, hardened oil, synthetic aluminum silicate, sesame oil, flour starch, white beeswax, magnesium oxide, dimethyl polysiloxane, potassium sodium tartrate, sucrose faty acid ester, glycerin faty acid ester, silicon resin, aluminum hydroxide gel, stearyl alcohol, stearic acid, aluminum stearate, calcium stearate, polyoxyl stearate, magnesium stearate, cetanol, gelatin, talc, magnesium carbonate, precipitated calcium carbonate, cornstarch, lactose, hard fat, saccharose, potato starch, hydroxypropylcelulose, fumaric acid, sodium stearyl fumarate, polyethylene glycol, polyoxyethylene polyoxypropylene glycol, polysorbate, beeswax, magnesium aluminometasilicate, methylcelulose, Japan wax, glycerin monostearate, sodium lauryl sulfate, calcium sulfate, magnesium sulfate, liquid parafin, phosphoric acid, palmitic acid, and hydrogenated vegetable oils and fats. The lubricant may be used alone or in various mixtures, and utilized in any amount known in the art for oral compositions. [00109] As used herein, “binders” are agents used to impart cohesive qualities to powdered materials. Binders, or “granulators” as they are sometimes known, impart a cohesiveness to the tablet formulation, which ensures the tablet remaining intact after compression, as wel as improving the free-flowing qualities by the formulation of granules of desired hardness and size. Materials commonly used as binders include starch, such as corn starch and pregelatinized starch; gelatin; sugars, such as sucrose, glucose, dextrose, molasses, and lactose; natural and synthetic gums, such as gum acacia, sodium alginate, extract of Irish moss, panwar gum, ghati gum, mucilage of isapol husks, carboxymethylcelulose, methylcelulose, polyvinylpyrolidone (PVP), Veegum, microcrystaline celulose, microcrystaline dextrose, amylose, larch arabogalactan, ethyl celulose, celulose acetate, and the like. The binder may be used alone or in various mixtures, and utilized in any amount known in the art for oral compositions. [00110] As used herein, “colorants” are agents that give the oral composition a more pleasing appearance, and in addition help the manufacturer to control the product during its preparation and help the user to identify the product. Any of the approved certified water-soluble FD&C dyes, mixtures thereof, or their coresponding lakes may be used to color tablets. A color lake is the combination by adsorption of a water-soluble dye to a hydrous oxide of a heavy metal, resulting in an insoluble form of the dye. The colorant may be used alone or in various mixtures, and utilized in any amount known in the art for oral compositions. [00111] Other conventional ingredients that may optionaly be present in the oral composition include preservatives, stabilizers, anti-adherents or silica flow conditioners or glidants, such as
Aty. Docket. No.018716.208867 (WN3426) silicon dioxide. Such ingredients may be used alone or in various mixtures, and utilized in any amount known in the art for oral compositions. [00112] It is to be appreciated that certain components or additives may be classified under diferent terms of art and just because a component or additive is classified under such a term does not mean that they are limited to that function. If utilized, the additive or additives may be present in the oral composition in various amounts. Additional ingredients for optional use in the oral composition, e.g., when adapted for topical or oral administration, are described in U.S. Patent Nos.5,747,006; 5,980,904; 6,994,874; 7,060,304; 7,247,321; 7,348,034; 7,364,759; 7,700,110; 7,722,904; 8,202,556; 8,916,212; 9,445,975; 9,801,809; 10,307,366; 10,532,024; and 10,537,516; and in U.S. Publication Nos.2006/0257509; 2007/0224154; 2008/0081082; 2008/0124409; 2013/0302265; 2017/0252293; 2017/0281666; 2018/0200285; 2019/0083566; 2019/0160117; 2020/0171117; 2020/0383898; 2021/0017240; and 2021/0212926; the disclosures of which are hereby incorporated herein by reference in their entirety. Method of Administration [00113] The oral composition may be administered or applied as needed, daily, several times per day or in any suitable regimen such that the desired outcome is achieved. In the method of this disclosure, the frequency of administration (e.g., oral application) can depend on several factors, including the desired level of UC inhibition and/or treatment. The oral composition may comprise the isolated polysaccharide described herein. Generaly, a regimen includes administration of the oral composition once or twice daily to include an administration in the morning and/or an administration in the evening. The amount and/or frequency of administration of the oral composition may depend on several factors, including the level of desired results and the specific oral composition. [00114] As an example, the formulation of the present invention can be internaly administered with an acceptable carier in the form of a pil, tablet, powder, bar, beverage, etc. Thus, the formulations described herein are useful in a wide variety of finished products, including pharmaceutical products, food products, and beverage compositions. [00115] When the formulations of the present invention are oraly administered in the form of a liquid, the liquid may be water-based, milk-based, tea-based, fruit juice-based, or some combination thereof. Solid and liquid formulations for internal administration according to the present invention can further comprise thickeners, including xanthan gum, carboxymethyl- celulose, carboxyethylcelulose, hydroxypropylcelulose, methylcelulose, microcrystaline celulose, starches, dextrins, fermented whey, tofu, maltodextrins, polyols, including sugar
Aty. Docket. No.018716.208867 (WN3426) alcohols (e.g., sorbitol and mannitol), carbohydrates (e.g., lactose), propylene glycol alginate, gelan gum, guar, pectin, tragacanth gum, gum acacia, locust bean gum, gum arabic, gelatin, as wel as mixtures of these thickeners. These thickeners are typicaly included in the formulations of the present invention at levels up to about 0.1%, depending on the particular thickener involved and the viscosity efects desired. [00116] The solid and liquid (food and beverage) formulations of the present invention can, and typicaly wil, contain an efective amount of one or more sweeteners, including carbohydrate sweeteners and natural and/or artificial no/low calorie sweeteners. The amount of the sweetener used in the formulations of the present invention wil vary, but typicaly depends on the type of sweetener used and the sweetness intensity desired. [00117] Generaly, the formulations of the present invention are oraly administered at least on a daily basis for a period of time suficient to bring about the desired level of improvement in UC in the subject. Oral administration of the formulations of the invention may continue for any suitable period of time. More specificaly, within a few hours to within a few days of the initial application or ingestion, a user may notice the UC condition has improved. It should be appreciated that the frequency with which the formulations of the present invention should be ingested wil vary depending on the desired level improved UC condition. In particular, the degree of UC improvement wil vary directly with the total amount of oral composition used. [00118] Useful dosage forms can be prepared by methods and techniques that wil be wel understood by those of skil in the art and may include the use of additional ingredients in producing tablets, capsules, or liquid dosage forms. INDUSTRIAL APPLICABILITY [00119] This disclosure provides new and useful methods of preparing Polygonatum polysaccharide compositions including the botanical extracts described herein. The resulting Polygonatum polysaccharide compositions can ofer potential preventive and therapeutic approaches for lowering the risk of and symptoms associated with ulcerative colitis. As such, the compositions and methods described herein are useful for preventing and treating ulcerative colitis. [00120] General compositions and product lines provided by this disclosure relate to inflammatory bowel disorders including ulcerative colitis and its treatment and prevention via the disclosed Polygonatum polysaccharide compositions, and specific examples include pils, tablets, and liquids utilizing such Polygonatum polysaccharide compositions.
Aty. Docket. No.018716.208867 (WN3426) [00121] The folowing examples, ilustrating the compositions and methods of this disclosure, are intended to ilustrate and not to limit the disclosure. EXAMPLES [00122] The protective efect and mechanism of Polygonatum polysaccharide compositions obtained from Polygonatum cyrtonema Hua via the disclosed preparation method was evaluated in mice subjected to dextran sodium sulfate (DSS)-induced ulcerative colitis (UC). Preparation of Polygonatum Polysaccharide Composition [00123] 9.8 kg of steamed Polygonatum cyrtonema Hua rhizome was subjected to stepwise extraction by ethanol and water precipitation in accordance with the method described above and as shown in Figure 1 to obtain 548 g of crude Polygonatum polysaccharides (“PSP”) in the form of brown powder at a yield of 5.6%. The PSP was passed through a DEAE Sepharose Fast Flow anion exchange column and eluted with deionized water and NaCl solutions of diferent concentrations (salt gradient) to obtain a partial polysaccharide (“PSP-W”) with a yield of 12.55%. The PSP-W was further separated and purified using a SUPERDEX® 75 (or SUPERDEX® 200) gel column to obtain the present Polygonatum polysaccharide composition (“PSP-W-1”) with a yield of 8%. The high-performance liquid chromatogram (HPLC) of the Polygonatum polysaccharide composition as shown in Figure 2 indicates that the polysaccharides contained in the composition are homogeneous. [00124] Structural characterization of the obtained Polygonatum polysaccharide composition (“PSP-W-1”) was performed by a variety of analytical methods to study its primary structure. The FT-IR spectrum of the Polygonatum polysaccharide composition is shown in Figure 3. Trifluoroacetic acid was used to completely acid hydrolyze a sample of the Polygonatum polysaccharide composition into monosaccharides. Sodium borohydride was used to reduce the monosaccharides to sugar alcohols, and then acetylated derivatives were prepared. Gas chromatography (GC) was used to determine the composition of monosaccharides as shown in Figure 4. In the graph, 1 coresponds to rhamnose, 2 coresponds to fucose, 3 coresponds to arabinose, 4 corresponds to xylose, 5 coresponds to mannose, 6 coresponds to glucose, and 7 coresponds to galactose. The molecular weight of the Polygonatum polysaccharide composition was determined by high-performance gel-permeation chromatography (HP-GPC) to be 14.38×103kDa. The analysis of the monosaccharide composition showed that the Polygonatum polysaccharide composition only contained galactose. Methyl iodide was used to completely methylate a sample of the Polygonatum polysaccharide composition, and then the fuly methylated polysaccharide was hydrolyzed and reduced. Gas chromatography–mass
Aty. Docket. No.018716.208867 (WN3426) spectrometry (GC-MS) detection and analysis was performed after this acetylation to determine the composition type, ratio, and position of the branched chains of the sugar chains (the characteristic peaks of the fragments of the acetylated derivatives of sugar methylation according to each connection method are different). The results are shown in Figure 5, wherein T-Galp is terminal galactopyranose, 1,4-Galp is 1,4 linked galactopyranose, and 1,4,6-Galp is 1,4,6 linked galactopyranose. The results of the methylation analysis showed that the Polygonatum polysaccharide composition contained Galp connected at the terminal group, Galp connected at the 1,4 positions, and Galp connected at the 1,4,6 position. Nuclear magnetic resonance (NMR) analysis was also performed on the Polygonatum polysaccharide composition to determine the types and connectivity of the glycosidic bonds, as shown in Figures 6a-h wherein A represents 1,4-β-D-Galp, B represents 1,4,6-β-D-Galp, and C represents T-β-D-Galp. From the NMR measurements it was deduced that the structure of the Polygonatum polysaccharide composition is a galactan (galactosan) having 1,4-Galp and 1,4,6-Galp as the main chain, and T-Galp is connected to the 6-position of 1,4,6-Galp. It is deemed that the obtained Polygonatum polysaccharide composition isolated from steamed Polygonatum cyrtonema Hua rhizome according to the method described herein is a newly discovered polysaccharide. Evaluation of the Prepared Polygonatum Polysaccharide Composition [00125] Gavage administration of various dosage levels of the present Polygonatum polysaccharide composition in mice was conducted for one week, along with the addition of 2.5% dextran sulfate sodium (DSS) to drinking water which induced mice to establish an ulcerative colitis model, in order to study the efect of the Polygonatum polysaccharide composition in reducing ulcerative colitis. As shown in the chart of Figure 7a, the control subject (“Normal”) received only water and no DSS, one subject (“Model”) received only DSS and no treatment, one subject (“Positive”) received DSS and 200 mg/kg salicylazosulfapyridine (SASP) as a comparative treatment, one subject (“L-PSP-W-1”)received DSS and 100 mg/kg of the Polygonatum polysaccharide composition as a treatment, one subject (“M-PSP-W-1”) received DSS and 200 mg/kg of the Polygonatum polysaccharide composition as a treatment, and one subject (“H-PSP-W-1”) received DSS and 400 mg/kg of the Polygonatum polysaccharide composition as a treatment. Disease activity index (DAI), colon length, spleen coeficient, and thymus coeficient were measured, and histopathological changes in the colon were detected using histological staining. The levels of inflammatory factors as wel as inflammatory mediators in colonic tissues were measured by q-PCR and the expression of intestinal epithelial proteins
Aty. Docket. No.018716.208867 (WN3426) was detected by immunofluorescence. The intestinal flora was analyzed by 16S rRNA sequencing. [00126] The present Polygonatum polysaccharide composition aleviated the symptoms of DSS-induced histopathological changes in UC, modulated intestinal inflammation by reducing the levels of inflammatory factors as wel as inflammatory mediators (iNOS, IL-6, COX-2), repaired the intestinal barier indicated by occluding ZO-1 and regulated the intestinal microbiota to aleviate UC symptoms. The results of the study are shown in Figures 7b-g. There were six total subjects. Each of the subjects having DSS-induced colitis were compared to the control, with * indicating p<0.05, ** indicating p<0.01, and *** indicating p<0.001 Figure 7b shows that other than the control, the subject that received the highest dosage of the Polygonatum polysaccharide composition best maintained body weight. Figure 7c shows that other than the control, the subject that received the highest dosage of the Polygonatum polysaccharide composition had the lowest DAI score. Figure 7d shows that other than the control, the subject that received the highest dosage of the Polygonatum polysaccharide composition had the longest colon length. Figure 7e shows that the histomorphology of the colons of the subjects receiving the Polygonatum polysaccharide composition (“L-PSP-W-1”; “M-PSP-W1”; “H-PSP-W-1”) was similar to that of the subject receiving SASP (“SASP”) as an alternative treatment and surpassed that of the subject that received no treatment (“DSS”). Figure 7f shows that the spleen weight index of the subjects receiving the Polygonatum polysaccharide composition (“L-PSP- W-1”; “M-PSP-W1”; “H-PSP-W-1”) was lower/beter than the subject that received no treatment (“DSS”) but not as low as the subject receiving SASP (“SASP”). Figure 7g shows the spleen morphology of the subjects receiving the Polygonatum polysaccharide composition (“L- PSP-W-1”; “M-PSP-W1”; “H-PSP-W-1”) was similar to that of the subject receiving SASP (“SASP”) as an alternative treatment and surpassed that of the subject that received no treatment (“DSS”). [00127] In conclusion, the present Polygonatum polysaccharide composition exerted a protective efect against UC by repairing the intestinal barier as wel as regulating the balance of intestinal flora. The results provide a basis for the development of the present Polygonatum polysaccharide composition as a functional food ingredient for the prevention and treatment of UC. [00128] Supplemental information, findings, and technical data regarding the inventions of this disclosure are provided below. The inventions are not necessarily limited by the folowing information, and the folowing information is intended only to support various embodiments of
Aty. Docket. No.018716.208867 (WN3426) the claimed invention. The information is included in the publication Gong et al., “Structural characteristics of steamed Polygonatum cyrtonema polysaccharide and its bioactivity on colitis via improving the intestinal barrier and modifying the gut microbiota,” Carbohydrate Polymers 327 (2024) 121669, the disclosure of which is incorporated herein by reference in its entirety. [00129] Inflammatory bowel disease (IBD), which includes ulcerative colitis (UC) and Crohn's disease (CD), arises largely from un-healthy lifestyle habits, such as a diet rich in sugar and fat, iregular work schedules, limited physical activity, and work-related stress. Among these factors, IBD, characterized by symptoms such as abdominal pain, diarrhea, and bloody stools, represents a chronic and intricate inflammatory condition marked by gastrointestinal tract lesions. Epidemiological investigations reveal that IBD's prevalence ranges from 6 to 25 cases per one hundred thousand individuals, with a particularly high incidence of 20 cases per one hundred thousand people in Western countries, and a relatively lower incidence of 2 cases per one hundred thousand individuals in China. Notably, IBD's incidence has been on the rise in China, projected to reach approximately 1.5 milion cases by 2025. Given these trends, comprehensive exploration of IBD's pathogenesis and treatment remains paramount and urgent due to its substantial prevalence. Yet, the etiological mechanisms remain unclear, and the eficacy of available treatments remains limited. Presently, mainstream therapeutic strategies comprise pharmaceutical interventions and surgical procedures. Prominent pharmaceutical agents include 5-aminosalicylate (5-ASA) and Sulfasalazine (SASP), which, regretably, may incur adverse efects or financial burdens. Thus, the pursuit of cost-efective, eficacious, and low-toxicity pharmaceutical agents or functional foods has emerged as a novel frontier in drug research and disease management. [00130] In recent decades, clinical investigation has indicated that natural polysaccharides have efective protection and relieving efects on IBD. Natural polysaccharides can relieve IBD symptoms by adjusting the structure and composition of intestinal microbiota, restoring the intestinal mucosal barier and the immune system. For example, compared with Astragalus, Honey-processed Astragalus exhibit beter anti-inflammatory efects in protecting the intestinal mucosa, adjusting the cel expression factor, and afecting microbial diversity; the Astragalus polysaccharides can not only improve the colonic pathological damages and reduce the myeloperoxidase (MPO) activity but also suppress the levels of pro-inflammatory cytokines. Polygonatum cyrtonema (PC) is the rhizome of Huangjing in the Liliaceae and genus of polygonatum, which is a kind of traditional Chinese medicine used in both medicine and food and has efects of tonifying Qi and nourishing Yin and strengthening the spleen, moistening the
Aty. Docket. No.018716.208867 (WN3426) lungs and benefiting the kidneys. According to the basic theory of traditional Chinese medicine, the pharmacodynamics of clinicaly used PC can be enhanced by multiple steaming, and one of its main activity components is polysaccharides. Modern pharmacological studies indicate that Polygonatum cyrtonema polysaccharides (PCP) have immunomodulatory, anti-inflammatory, hypoglycemic, cardiovascular protection, anti-tumor, and anti-oxidant activities. However, nearly none of any studies related to the pharmacological efects of the steamed Polygonatum cyrtonema polysaccharides (PSP) were reported, and their specific pharmacological characteristics are stil unclear. Although a study indicates that the PC oligosaccharide has therapeutic efects on dextran sodium sulfate (DSS)-induced colitis, the anti-inflammatory potential of PSP is not reported to the best of our knowledge, which is a main component of steamed Polygonatum cyrtonema (PS). Therefore, the mitigative efects and their mechanism for the colitis of PSP were studied. [00131] The structural characteristics of PSP were first studied by the combination of spectral and chromatographic techniques. Subsequently, the protective and aleviating efects of PSP were explored in the mice model of DSS-induced colitis. Furthermore, an in-depth analysis of its potential anti-inflammatory mechanism, with a particular focus on its impact on the gut microbiota, was conducted and discussed. Notably, this study marks the first thorough examination of the structural characteristics of PSP, coupled with experimental validation of its anti-inflammatory potential. The findings underscore the mitigating and therapeutic efects of polysaccharides on colitis, thereby establishing a foundational understanding of the utilization of PSP in the development of functional foods aimed at the prevention and treatment of colitis. [00132] The rhizomes of steamed Polygonatum cyrtonema Hua (Base fresh Polygonatum cyrtonema, washed, and put in the oven to dry (temperature 55–60 °C, four days). Then, they were placed in the Traditional Chinese medicine moisturizer sterilization cabinet to steam for 2 h (temperature 100–105 °C), removed and cooled). They were purchased from Guangdong Yifang Pharmaceutical Co. (China, Guangdong). Pululans with a range of molecular weights were purchased from Shodex Co. (Tokyo, Japan). DEAE Sepharose Fast Flow and SUPERDEX® columns were obtained from GE Healthcare (AB, Uppsala, Sweden). Monosaccharide standards (D-galactose, D-arabinose, L-rhamnose, L-fucose, D-mannose, D- fructose, D-xylose and D-glucose) and trifluoroacetic acid (TFA) were purchased from Sigma- Aldrich (St. Louis, MO, USA). L-chiro-inositol was obtained from Sigma (MO, USA). The reagents used were of analytical grade. Double-distiled water was used for chemical analysis and triple-distiled water was used for bioassays.
Aty. Docket. No.018716.208867 (WN3426) [00133] The rhizomes of steamed Polygonatum cyrtonema were subjected to extraction with a 5-fold volume of 95% ethanol over a span of 2 h, then folowed by a similar extraction with 70% ethanol for 2 h. These sequential ethanol extractions aimed to eliminate pigments and smal, fat- soluble molecules. The defated rhizomes then underwent extraction with 10-fold the volume of boiling water, with the first extraction lasting 4 h and the subsequent second and third extractions lasting 3 and 2 h each. The extracts from aforementioned 3 rounds were mixed, concentrated and centrifuged (to remove impurities) to obtain the supernatant. The supernatant was then precipitated with three times the volume of 95% ethanol to yield crude polysaccharide (PSP). PSP (6 g) was dissolved in 80 mL of distiled water and centrifuged at 10,000 x g for 10 min. The supernatant was separated on a DEAE Sepharose Fast Flow column and eluted with water, and diferent concentrations of NaCl solutions (0.1 mol/L, 0.2 mol/L and 0.5 mol/L). The eluates from diferent parts were concentrated, dialyzed, and lyophilized to obtain diferent fractions denoted as PSP-W, PSP-1, PSP-2, and PSP-5. Subsequently, PSP-W (100 mg) was dissolved in 4 mL of 0.2 mol/L NaCl and centrifuged at 10,000 x g for 10 min. The supernatant was separated on a SUPERDEX® 200 column and eluted with 0.2 mol/L NaCl to yield the major polysaccharide PSP-W-1. [00134] Molecular weight and homogeneity of PSP-W-1 were measured using high- performance gel permeation chromatography (HPGPC) on an Agilent 1100 HPLC (Agilent, Santa Clara, CA, USA) units consisting of two 8 mm x 300 mm series KS-804 and KS-802 super hydrogel linear columns (Shodex Co., Tokyo, Japan), that was used for the determination. A standard curve was ploted using pululans (P-5 to P-800) with known molecular weights, and the column was eluted using 0.2 mol/L NaCl at a flow rate of 0.8 mL/min. PSP-W-1 was prepared at 2.0 mg/mL with 20 μL per injection. [00135] Using the method described in the literature, PSP-W-1 was completely hydrolyzed and analyzed for monosaccharide composition. PSP-W-1 (2 mg) was hydrolyzed at 120 °C after dissolving with 1 mL of 2 mol/L trifluoroacetic acid (TFA), and after completion of hydrolysis, 100 μL of the hydrolysis solution was neutralized with 100 μL of 2 mol/L sodium hydroxide (NaOH) folowed by the addition of 10 μL of 2 mg/mL L-chiro-inositol, and then reduced at 40 °C with NaBH4 or NaDH4 dissolved in DMSO for 90 min. After neutralization with acetic acid, acetic anhydride and 1-methylimidazole were reacted with the sample at 40 °C for 10 min to achieve acetylation. The resulting alditol acetates were analyzed by gas chromatography–mass spectrometry (GC–MS) using an Agilent GC 7890-5975A system (Agilent Technologies, USA) equipped with an HP-5 MS column (length: 60 m x 0.25 mm; liquid phase thickness: 0.25 μm,
Aty. Docket. No.018716.208867 (WN3426) Agilent). Monosaccharide standards and L-chiro-inositol (internal standard) were used to calculate the calibration factors (fi) for the diferent monosaccharides. The mono-saccharide content (mol%) was assayed as folows:
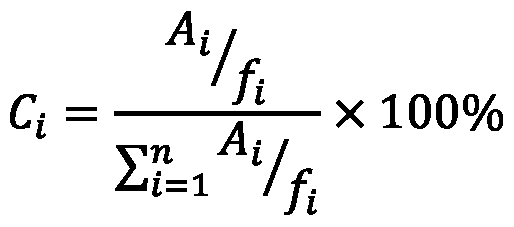
where Ci-calculated content of monosaccharides (molar %), Ai-area of each monosaccharide peak on GC–MS chromatogram, fi-molar response coeficient considering each monosaccharide. fi was detected as As (area of standard monosaccharides)/AIn (L-chiro-inositol), which were used in equal molar ratios. [00136] Methylation was assessed using the previous method as described in our previous report. The round botom flask containing 10 mg of PSP-W-1 was placed in a vacuum desiccator containing P2O5 overnight. On the next day, the round-botom flask was filed with nitrogen and dissolved in 2 mL of anhydrous DMSO.200 mg of NaOH powder was added and stired at room temperature for 1 h. Then, 0.3 mL of CH3I was added in five batches and stired at room temperature for 2 h. Finaly, the reaction was terminated by adding 2 mL of water.1.5 mL of chloroform was added for extraction, and the chloroform layer was washed three times with water. The polysaccharide was methylated three times to obtain the fuly methylated polysaccharide. Hydrolysis was continued for 2 h at 120 °C with 2 mL of 2 mol/L TFA. After the conversion of the fuly methylated PSP-W-1 to partialy methylated alditol acetate (PMAA), GC–MS analysis was performed using an HP-5 MS column. [00137] The absolute conformation of the monosaccharide of PSP-W-1 was assessed using previous methods as described in our previous reports. PSP-W-1 was dissolved in 1 mL of 2 mol/L TFA solution, and hydrolyzed in an oven at 120 °C for 2 h. After hydrolysis, the TFA was completely removed by vacuum evaporation, and 25 μL each of isopropanolamine-methanol solution (1:8 v/v), acetic acid-methanol solution (1:4 v/v) and 3 % sodium cyanoborohydride solution (w/v) were added and heated at 65 °C for 1.5 h. After vacuum evaporation, the sample was evaporated twice, and then the methanol-acetic acid solution (5:1 v/v) was added and repeatedly evaporated three times, then methanol was added and repeatedly evaporated three times, and pyridine and acetic anhydride were added and heated at 100 °C for 45 min. Finaly, the reaction was terminated by adding 2 mL of water. Added 1.5 mL of chloroform for extraction, and the chloroform layer was washed with water three times. The sample was dried with anhydrous sodium sulfate and sorted in a sample botle for GC–MS analysis.
Aty. Docket. No.018716.208867 (WN3426) [00138] FT-IR spectroscopy was performed using a PerkinElmer 2000 FTIR spectrometer. Dried PSP-W-1 (2 mg) was mixed with 200 mg KBr powder and pressed into pelets for FT-IR analysis in the wavelength range of 4000–400 cm-1. [00139] After drying under vacuum overnight, PSP-W-1 (50 mg) was dissolved in 0.6 mL D2O, 1H NMR,13C NMR, DEPT 135 NMR, 2D1H-1H COSY, HSQC and HMBC NMR spectra at 25 °C using a Bruker Avance II 600 spectrometer (Bruker, Germany) and a PABBO probe (5 mm, BB/ 19F-1H/D, Z-GRD). Acetone was used as an internal standard, and the shifts of13C and1H NMR relative to acetone were 31.45 and 2.225 ppm, respectively. Al data were processed using standard Bruker software and MestNova. [00140] Healthy female C57BL/6 mice, 6–8 weeks old, 20 ± 2 g, were provided by the Experimental Animal Center of Shanghai University of Traditional Chinese Medicine (a relative humidity of 55 ± 10 %, temperature of 23–25 °C in an SPF-grade rearing room with a 12 h daily light-dark cycle was provided, and freely accessed to food and water), with an ethics approval number of PZSHUTCM220725040. Before starting the experiment, the mice were fed at least one week in the aforementioned environment to acclimatize it. The protocols and operations involved in this experiment were performed under the guidance of the Experimental Animal Ethics Commitee of Shanghai University of Traditional Chinese Medicine. Meanwhile, animal experiments also complied with the National Research Council's Guide for the Care and Use of Laboratory Animals. [00141] Colitis was induced in the mice with 2.5 % DSS (w/v) dissolved in drinking water for 7 consecutive days. Pure water was given to the Control group mice. The mice were divided into 6 groups (n = 6/group). Mice in the low, medium and high-dose PSP-W-1 groups were administered 100, 200 and 400 mg/kg per day by gavage. The mesalazine (SASP) group was administered 200 mg/kg per day by gavage. Animals in each group received treatment from day 1 to day 7. On day 8, mice were executed by cervical dislocation. The spleen, thymus, cecum feces, and the entire colorectum of the mice were removed, and the distal colon specimens were frozen in liquid nitrogen or immediately fixed in 4 % (w/v) formalin solution for further analysis. [00142] Throughout the experimental period, daily observations and recordings were made regarding the mental state, diet, body weight, fecal consistency, and fecal blood of al mice. Based on the scores of weight loss, fecal consistency and fecal blood, DAI was calculated as the average score of these three parameters (refer to Table 1 for scoring criteria). In addition, rectums fixed in paraformaldehyde solution for at least 24 h were dehydrated in 100 % ethanol and isopropanol and embedded in paraffin. Then, the colonic parafin was cut into sections, dewaxed
Aty. Docket. No.018716.208867 (WN3426) with xylene, rehydrated with gradient concentrations of ethanol (100 %–75 %), stained with hematoxylin and eosin, and observed with a light microscope. The histopathological score of the rectum was calculated according to the tissue destruction and the degree of inflammatory cel infiltration (refer to Table 2 for scoring criteria). [00143] Total RNA of the colon tissue was extracted by using Trizol reagent (Life Technologies) according to the manufacturer's protocol. Then the RNA was transcribed into cDNA by Evo M-MLV Kit Reverse Transcriptase kit (Accurate Biology, China). Next, qRT- PCR was conducted by utilizing the SBYR Green Premix Pro Taq HS q-PCR kit (Accurate Biology) on Quant Studio 6 Flex System (Life technologies). The relative mRNA expression of the folowing genes was normalized by using β-actin as the endogenous control. The primer
Aty. Docket. No.018716.208867 (WN3426) sequences used were as folows: iNOS: forward, 5’-GGAGTGACGGCAAACATGACT-3’, reverse, 5’-TCGATGCACAACTGGGTGAAC-3’; interleukin 6 (IL-6): forward, 5’- TCTATACCACTTCACAAGTCGGA-3’, reverse, 5’-GAATTGCCATTGCA-CAACTCTTT- 3’; COX-2, forward, 5’-GCCTTCCCTACTTCACAA-3’, reverse, 5’- ACAACTCTTTTCTCATTTCCAC-3’; β-actin: forward, 5’-GGCTGTATTCCCCTCCATCG- 3’, reverse, 5’-CCAGTTGGTAA-CAATGCCATGT-3’. [00144] Immunofluorescence assay was applied to detect the expression levels of occludin and ZO-1 in colonic tissues. Mouse colon tissue was fixed with a paraformaldehyde fixation solution and washed with 100 % alcohol, then placed in a repair bufer containing EDTA (pH = 8.0). After incubation with BSA, ZO-1 antibody and occludin antibody were added to the slides overnight. After washing, the slides were incubated with HRP-labeled goat anti-rabbit IgG as wel as CY3-labeled goat anti-rabbit IgG at room temperature. Finaly, the slides were incubated with DAPI for 10 min at room temperature and protected from light, and placed under an immunofluorescence microscope (Olympus Corporation) for observation. [00145] On the eighth day of processing mice, the contents of the mouse cecum were colected and stored in a -80 °C refrigerator. Total DNA was obtained after total genomic DNA extraction from the microbial community according to the instructions of the E.Z.N.A. soil DNA kit (Omega Biotek, Norcross, GA, U.S.) Using the extracted total DNA as a template, total DNA was extracted using upstream primer 338F (5′-ACTCCTACGGGAGGCAGCAG-3′) and downstream primer 806R (5′-GGACTACHVGGGTWTCTAAT-3′) were used as templates for the analysis of the variable regions of the 16S ribosomal RNA (rRNA) gene V3-V4 on an ABI GeneAmp 9700 PCR instrument (ABI, USA) for amplification. The purified PCR products were library-built using NEXTFLEX Rapid DNA-Seq Kit and sequenced using Ilumina's Miseq PE300/NovaSeq PE250 platform. Operational taxonomic unit (OTU) clustering was performed on the quality control spliced sequences based on 97 % similarity using UPARSE software (htp:/ drive5.com/uparse/, version 7.1). Al data analyses were performed on the Megabio Cloud platform (htps:/cloud.majorbio.com). These analyses include alpha diversity analysis, principal coordinates analysis (PCoA), and non-metric multidimensional scaling analysis (NMDS). [00146] Al data were expressed as mean ± standard deviation (SD) and experiments were performed in triplicate. GraphPad Prism 8 software was used for al statistical analyses. Statistical significance was determined by one-way ANOVA. Results were considered significant at p < 0.05.
Aty. Docket. No.018716.208867 (WN3426) [00147] Crude polysaccharide PSP (548 g, yield 5.6 %) was obtained from 9.8 kg of PS by boiling water extraction and alcohol precipitation. After grading on a DEAE-Sepharose Fast Flow weak anion exchange column, PSP-W (12.55 % out of PSP) was obtained from the pure water eluate. PSP-W was purified using gel permeation chromatography on a SUPERDEX® 200 column to obtain a major polysaccharide, PSP-W-1 (29.5 % from PSP-W-1). The flow chart for the separation and purification of PSP-W-1 is shown in Figure 1. The HPGPC of PSP-W-1 yielded a single symmetric spike with an average molecular weight of 14.4 kDa, indicating that it is a homogeneous polysaccharide fraction (Figure 2). [00148] Figure 3 shows the infrared spectrum of PSP-W-1. The absorption bands at 3300 and 2890 cm-1 are typical for polysaccharides and are associated with O–H stretching vibrations and C–H stretching vibrations in polysaccharide molecules, respectively. The absorption peak at 1635 cm-1 is atributed to O–H bending vibrations. The strong absorption peak at 1035 cm-1 is atributed to C–O bending vibrations due to C-O-H or C-O-C, indicating the presence of pyranose rings. Additionaly, an absorption peak at 889 cm-1 suggested the presence of β- glycosidic bonds, coroborated in previous research findings. [00149] Gas-phase spectrum analysis (Figure 8) demonstrated the presence of a single peak, identified as galactose through comparison with standard monosaccharides. The results indicated that PSP-W-1 consisted of galactose and further suggested that PSP-W-1 was a galactan. At the same time, we examined this galactan by the m-hydroxybiphenyl method and found that it is basicaly a neutral polysaccharide without galacturonic acid. [00150] PSP-W-1 was hydrolyzed to monosaccharide by acid, it reacted with isopropanolamine under the acidic condition to form imine containing C––N double bond, and the imine was reduced to secylamine by sodium cyanoborohydride, and then reacted with acetic anhydride to form acetylated product, which was detected by GC–MS analysis, and the absolute configuration of monosaccharide in polysaccharide could be determined by comparing it with the peaks of monosaccharide derivatives of coresponding D/L configuration. The graphs show the GC chromatograms of PSP-W-1 and different configurations of galactose standards, and the D configuration of PSP-W-1 could be judged by comparison with the standards (Figure 9). [00151] To determine the linkage of the glycosidic bonds of PSP-W-1, it was methylated three times to obtain the fuly methylated polysaccharide, and the total ion chromatogram (TIC) (Figure 5) of the partialy methylated alditol acetate (PMAA) from PSP-W-1 was investigated. GC–MS analysis indicates that the PSP-W-1 derivative contained three coresponding glycosidic linkages of PSP-W-1, and the coresponding molar ratios are summarized in Table 3. With
Aty. Docket. No.018716.208867 (WN3426) reference to the information in the literature, for PSP-W-1, the three peaks in the TIC profile were identified as 1,5-di-O-acetyl-(1-diethyl)-2,3,4,6-tetra-O-methyl-galactitol (Figure 10), 1,4,5-tri-O-acetyl-(1-triethyl)-2,3,6-tri-O-methyl-galactitol (Figure 11),and 1,4,5,6-tetra-acetyl- (1-dibutyl)-2,3-di-O-methylgalactitol (Figure 12). PSP-W-1 is the main chain of 1,4-linked Galp and 1,4,6-linked Galp, with the main branching point at C-6 of 1,4,6-Galp. These structures were confirmed and further detailed by NMR analysis. [00152] To further obtain the detailed structure of PSP-W-1, NMR spectroscopy was performed. It mainly includes1H NMR spectra (Figure 6a) and 13C NMR spectra (Figure 6b) in 13C DEPT 135 NMR spectra (Figure 6c) as wel as two-dimensional spectra (Figure 13) and1H- 1H COSY (Figure 6f). [00153] As can be seen from Figure 6a, the proton signals on C-1 of the polysaccharide were usualy distributed at 4.0–5.5 ppm, which were easier to resolve, while most of the proton signals on C2-C6 were stacked at 3.0–4.0 ppm, which cross overlapped with each other and were dificult to resolve. Therefore, Figure 6a was mainly used to distinguish the conformations of glycosidic bonds in the polysaccharide structure and to further confirm the analytical results of Figure 6b. In general, the chemical shift value of α-glycosidic bond conformation was >5 ppm, and that of β-glycosidic bond conformation was <5 ppm. From the preliminary judgment in the figure, the β-glycosidic bond conformation was found in PSP-W-1. [00154] The13C NMR of PSP-W-1 is shown in Figure 6b.95–110 ppm is the hetero-capilary carbon signal of polysaccharide, 55–85 ppm is the signal of C2–C6 of PSP-W-1, and 103–106 ppm is the C-1 signal of galactose with diferent linkages. In Figure 6c, the inverted peak signal
Aty. Docket. No.018716.208867 (WN3426) at around 70 ppm indicates the presence of the 6-position linkage of hexose in PSP-W-1, which, combined with the methylation results, should be the C-6 signal of 1,4,6-Gal. The inverted peak signal at 61–63 ppm was the C signal of the unsubstituted hexose 6 or pentose 5 positions of the polysaccharide, which, combined with the methylation results, should be the C-6 signal of T- linked β-D-Galp and 1,4-linked β-D-Galp. [00155] The NMR spectra of PSP-W-1 were analyzed with reference to the literature data of similar structures. The 4.58 pm, 4.57 ppm, 4.55 ppm, 4.54 pm, 4.37 m, and 4.36 ppm at relatively high fields are the H-1 signals of β-Galp, and 104–106 ppm are the C-1 signals of β-Galp, so it is easy to assign 105.26/4.60 ppm (A), 105.18/4.55 ppm (B), 105.18/4.53 ppm (C) as C-1/H-1 signals of β-Galp. The C-4 (78.9/79.21 ppm) of residue A/B shows a shift to the low field, indicating a linkage at position 4; the C-6 (71.43 ppm) of residue B shows a shift to the low field, indicating a linkage at position 6; the C-6 (61.98/62.23 ppm) of residue A/C shows an inverted peak in the13C DEPT 135 NMR spectrum, indicating that its residue A is 1,4-β-Galp, B is 1,4,6- β-Galp, and C is T-β-Galp, based on the above information and methylation results. The remaining signals can be assigned according to Fig.4d (HSQC (blue), and the chemical shifts are listed in Table 4.

[00156] Figure 13 (HMBC (black), A1/A4 (105.6/4.09) shows the corelation between C-1 of 1,4-linked β-D-Galp and its adjacent H-4 of 1,4-linked β-D-Galp. B1/A4 and A1/A4 overlap with each other, showing the corelation between the C-1 of 1,4,6-linked β-D-Galp and the H-4 of 1,4-linked β-D-Galp. A4/A1 (78.9/4.57 (4.55) shows the corelation between the C-4 of 1,4- linked β-D-Galp and the H-1 of its adjacent 1,4-linked β-D-Galp. A4/B1 overlaps with A4/A1 and belongs to the C-4 of 1,4-linked β-D-Galp and the H-1 of 1,4,6-linked β-D-Galp. B6/C1
Aty. Docket. No.018716.208867 (WN3426) (71.43/ 4.37 (4.36)) shows the corelation between C-6 of 1,4,6-linked β-D-Galp and H-1 of T- linked β-D-Galp, indicating that T-linked β-D-Galp is atached to the C-6 position of 1,4-linked β-D-Galp. [00157] Based on these results, PSP-W-1 has the backbone of the 1,4-linked β-D-Galp repeat unit and is partialy substituted at C-6 of galactose with the branch connected by the T-linked β- D-Galp. Again, this is in accordance with the results of methylation analysis. Therefore, the possible structure of PSP-W-1 was deduced, as shown in Figure 6g. [00158] We further determined the efect of PSP-W-1 on DSS-induced colitis in vivo (The experimental design is shown in Figure 7a). DSS induces bloody stools, weight loss, diarhea, and other clinical manifestations in mice similar to IBD. In addition, inflammation and immunosuppression after DSS treatment lead to splenomegaly and transient thymic atrophy in mice. The colon, spleen, and thymus of the colected mice were used to calculate organ indices as typical indicators of colitis. Not surprisingly, DSS treatment significantly increased DAI levels (from day 4), decreased body weight (from day 3), increased spleen indices, and decreased thymus indices in mice compared to the control group. However, oral administration of L-PSP- W-1, M-PSP-W-1, H-PSP-W-1 and SASP restored body weight (Figure 7b), reduced DAI levels (Figure 7c), reduced spleen index (Figure 7d) and increased thymic index (Figure 14) compared to the DSS group. The morphology of the colon and the spleen are shown in Figures 7e and 7g. On the days 6 to 7 of the experiment, none of the colitis mice in the five treatment groups showed significant weight loss and the blood in the feces almost disappeared. In addition, although the five treatment groups were able to resist colitis-induced thymic suppression, they did not show significant diference. [00159] This is consistent with the results of previous pharmacodynamic studies by observing and studying the results of colorectal pathological sections (HE staining) in each group. The colorectal wal of the Control group mice was intact and continuous, the intestinal epithelial cels were clearly visible, the glandular structure was normal and wel-arranged, the cup cels were abundant, and the infiltration of acute inflammatory cels was rarely seen; the intestinal wal of DSS group mice was obviously thickened, the tissue structure was disturbed, the cup cels were missing, the crypt was destroyed and disappeared, and a large number of the DSS group, the intestinal wal was significantly thickened and disorganized, with loss of cupped cels, loss of crypt destruction, and infiltration of neutrophils and inflammatory cels into the mucosa and submucosa (Figure 16, with the organization scores shown in Figure 15). However, compared to the DSS group, the SASP and H-PSP-W-1 groups showed varying degrees of improvement
Aty. Docket. No.018716.208867 (WN3426) in histopathological damage, with the appearance of glandular structures, an increase in cupped cels, and a significant decrease in inflammatory infiltration. The most significant improvement was seen in the H-PSP-W-1 with the SASP group, with a largely ordered arangement of glands and visible basic histological structures. [00160] Studies have shown that inflammation is the main symptom of the colitis process. The levels of inflammatory cytokines and inflammatory mediators are associated with the severity of colitis. Abnormal secretion of severe inflammatory cytokines as wel as mediators promotes the progression of intestinal inflammation and leads to a disequilibrium in the intestinal anti- inflammatory system. The mRNA expression of inflammatory mediators (iNOS, COX-2) and inflammatory factors (IL-6) was low in the colonic tissues of mice in the Control group, and they were significantly up-regulated in the DSS group, while their expression was significantly down- regulated in the H-PSP-W-1 group relative to the DSS group (Figures 17-19). [00161] It is wel known that intestinal epithelial barier dysfunction and intestinal permeability are major factors contributing to susceptibility to inflammatory diseases. Typicaly, two components of the epithelial cytoskeleton, including ZO-1 and occludin, regulate the paracelular permeability and cel adhesion function. To investigate the effect of PSP-W-1 on the epithelial barier function of colitis, the protein expression of ZO-1 and occludin were measured using an immunofluorescence assay, which indicated that the tight junction proteins at the periphery of colonic epithelial cels in the Control group show continuous tight loops. The ZO-1 and occludin proteins in the DSS group were almost disrupted and the fluorescence staining is dark. Compared with the DSS group, the SASP as wel as the PSP-W-1 group were only partialy disrupted and the degree of damage is significantly reduced. [00162] It is wel known that a disequilibrium of intestinal flora is closely related to IBD. However, the lack of probiotics can afect or impair the health of the intestinal microenvironment. Therefore, to investigate whether and how PSP-W-1 afects the composition or abundance of the intestinal microbiota, we applied advanced 16S rRNA sequencing technology to perform an in-depth analysis of the intestinal bacterial composition in mice, mainly including alpha diversity analysis, beta diversity analysis, and intestinal flora abundance analysis. Alpha diversity refers to the diversity within an environment, and the diversity index can be analyzed to understand information such as species richness and diversity in the community. The indices commonly used to calculate the abundance of flora in research are mainly Sobs, Chao, and Ace, and the indices used to calculate the diversity of flora are Shannon. The higher Sobs, Chao and Ace indices indicate a higher richness of the flora; the higher
Aty. Docket. No.018716.208867 (WN3426) Shannon index indicates a higher diversity of the flora. The results of the Sobs (Figure 20), Ace (Figure 21) and Chao (Figure 22) indices showed that they are significantly lower in colitis model mice compared to the Control group, indicating that the richness of intestinal flora is afected by DSS modeling, while the lack of difference in Shannon (Figure 23) indices may be due to the short modeling time. However, PSP-W-1 administration increases Sobs, Chao, and Ace indices with significant diferences compared to DSS, infering that PSP-W-1 can increase the abundance of intestinal flora. [00163] To directly observe the diferences in the composition of the mice colonies in each group, we performed principal coordinates analysis (PCoA) as wel as non-metric multidimensional scaling (NMDS) at the genus level, where the more similar the sample species composition is, the closer the distance is reflected in the graph. From PCoA (Figure 24) and NMDS (Figure 25), it is shown that the DSS group is diferent from the Control group, indicating that the DSS model is successfuly established. Meanwhile, the distance between the Control and the PSP-W-1 treatment group was smaler than the spacing between the Control and the DSS model group, which indicates that PSP-W-1 could improve the gut microbiota of DSS-induced colitis mice. [00164] The number of OTUs in the feces reflects the diversity of the flora. As shown in Figure 26, we can find the overlapping or unique OTU data between the Control, DSS, and PSP-W-1 groups by the three-loop diagram. Also, the graph shows that the DSS group has unique 19 OTUs and the Control as wel as the PSP-W-1 group have >25 independent OTUs. The results indicated that DSS induction reduced the diversity of the intestinal flora, which could be reversed after PSP-W-1 administration while maintaining the balance of the intestinal flora. [00165] In addition. we identified five major bacterial phyla, including Firmicutes, Bacteroidota, Proteobacteria, Verrucomicrobiota, Actinobacteriota, at the phylum taxonomic level in the form of bar charts (Figure 27) for each group of samples. Among them, Firmicutes, Bacteroidota, Proteobacteria are the major components of the intestinal flora. The relative abundance of microbial compositional profiling at a family level is shown in Figure 28. We found that the relative abundance of the Firmicutes and Bacteroidota in the Control group accounted for >90 % of the total flora, and the relative abundance of the pathogenic Proteobacteria was lower. In the DSS group, the abundance of Firmicutes and Actinobacteriota in mouse feces decreased, while the abundance of pathogenic Proteobacteria increased significantly. This phenomenon was significantly reversed after the PSP-W-1 administration.
Aty. Docket. No.018716.208867 (WN3426) [00166] Linear discriminant analysis (LDA) effect sizes were also adopted to identify diferentialy enriched taxa in these groups, with LDA scores showing the dominant taxa and their effects at diferent taxonomic levels from phylum to genus. From Figures 29 and 30, the DSS group was enriched in the abundance of Proteobacteria, Gammaproteobacteria, Enterobacteriaceae, Bacteroidaceae, etc. The dominant bacteria in the control group were Lactobacilus, Lactobacilaceae, Lactobacilales, Bacili, Firmicutes, etc. The dominant bacteria in the PSP-W-1 group were g_norank_f__Muribaculaceae, Roseburia, and g_norank_f_norank_o_clostridia_UCG 014. [00167] Finaly, the marker bacteria were compared between diferent groups of mice at the phylum (Figure 31) as wel as genus level (Figure 32) by significant difference analysis. At the phylum level, the percentage of Proteobacteria was significantly higher in the DSS group and significantly lower after administration via PSP-W-1. The Firmicutes, as an additional species bacterium with large diferences, improved the expression disequilibrium after DSS modeling by treatment. At the genus level, administration by PSP-W-1 not only reversed the significant reduction of norank_f__Muribaculaceae in the DSS group but also increased the abundance of norank_f__Muribaculaceae. Moreover, we found that the occurence of Bacteroidota was disordered in the DSS group. [00168] The aforementioned results indicate that PSP-W-1 can reverse the disequilibrium of intestinal microbiota in response to inflammation in DSS-induced mice. Therefore, PSP-W-1 may be a good candidate for the treatment of colitis. [00169] In this study, we extracted and purified a novel polysaccharide (PSP-W-1) from the PS and focused on its structure as wel as its anti-inflammatory effects in vivo. The results indicate that PSP-W-1 can aleviate intestinal flora dysbiosis as wel as intestinal inflammation in mice. [00170] Numerous studies have shown that inflammatory cytokines are significantly elevated in colitis patients, among which inflammatory cytokines (IL-6) are characteristic of colitis and they are key pathophysiological regulators of the onset, progression and eventual resolution of inflammation. At the same time, excessive production of inflammatory mediators (COX-2, iNOS) accompanies the onset and progression of inflammation. Therefore, q-PCR was used to quantify the mRNA expression of IL-6, COX-2, and iNOS in colonic tissues. In the present study, induction of DSS resulted in the upregulation of IL-6, COX-2, and iNOS pro- inflammatory cytokines as wel as mediators. Treatment with PSP-W-1 significantly inhibited the overproduction of pro-inflammatory cytokines and mediators, achieving a beter anti- inflammatory efect. In addition, abnormal expression of tight junction proteins on the intestinal
Aty. Docket. No.018716.208867 (WN3426) epithelium, which mainly reduced expression of ZO-1 protein and Occludin protein, can seriously impair the health of the intestinal barier in colitis patients. In the present study, using a DSS-induced colitis mice model, it was found that the expression of intestinal epithelial tight junction proteins (ZO-1, Occludin) could be restored after administration of PSP-W-1. Similarly to a previous study, polysaccharides from Chinese yam and bamboo shoots also improved the integrity and permeability of the intestinal barier by restoring the expression of intestinal epithelial tight junction proteins. [00171] Oligogalactose, or “bifidus factor,” is used in the food industry as a prebiotic supplement to treat colitis, which promotes the growth of bifidobacteria and lactobacili, while reducing the growth of pathogenic bacteria. Studying the efect and mechanism of galactans in aleviating and treating colitis by regulating intestinal flora wil provide an additional basis for the application of the tonic herbal medicine PC as a functional ingredient in the food industry. Therefore, the efect of PSP-W-1 on intestinal microorganisms was also studied. In this study, PSP-W-1 could not only significantly increase the diversity of intestinal flora, but also significantly promote the growth of beneficial bacteria (e.g. norank_f__Muribaculaceae, Lactobacilus, norank_f__norank_o__Clostridia_UCG-014) as wel as inhibit the growth of harmful bacteria (Escherichia-Shigela). Thus, it achieves the treatment of treating intestinal inflammation. These findings are consistent with literature, which also reported a significant increase in the relative abundance of Escherichia-Shigela after DSS induction. In another study, bamboo shoot polysaccharides promoted the growth of Bifidobacterium as wel as Lactobacilus and inhibited the growth of Helicobacter and an unidentified bacterium from Enterobacteriaceae. A recent study shows that administration of Gastrodia elata polysaccharides results in a significant increase in the relative abundance of bacteria with potential anti-inflammatory efects (e.g., Ligilactobacilus and Aloprevotela) and a decrease in the levels of bacteria associated with pro-inflammatory responses, such as Bacteroides and Escherichia-Shigela. In conclusion, PSP-W-1 can aleviate DSS-induced colitis by modulating the intestinal flora. [00172] Polysaccharides can be categorized into glucan, galactan, fructan, mannoglucan, pectin, and arabinogalactan according to their structures. Previous studies have isolated and purified 1,4-β-D-galactan with a molecular weight of 42 kDa and a degree of polymerization of 6 from the steamed Polygonatum sibiricum; 1,4-β-D-galactan with a molecular weight of 7019 Da and a degree of polymerization of 8 from the PS. In this study, 1,4-β-D-galactan with a molecular weight of 14.4 kDa and a degree of polymerization of 5 was purified from the PS. Comparing
Aty. Docket. No.018716.208867 (WN3426) with their sources of Polygonatum, the molecular weight and the degree of polymerization of the isolated polysaccharide, we conclude that the galactan isolated in this study is a new polysaccharide. In addition, most studied polysaccharides that have clear structural characteristics focus on pectin, glucan, fructan, and mannoglucan due to their prevention and mitigation efects to the colitis. In order to qualitatively and quantitatively study the pharmacological mechanisms of galactans on the relief and treatment of colitis symptoms, the PC was steamed to ensure that the mostly extracted polysaccharide structures were al galactans, and meanwhile to reduce the influence of other polysaccharides. To the best of our knowledge, the aleviative, as wel as therapeutic effects of galactan on colitis, are investigated for the first time herein, providing new perspectives as wel as options for further research on functional foods of PSP. [00173] In conclusion, a homogeneous polysaccharide (PSP-W-1) extracted from steamed Polygonatum cyrtonema has an aleviating efect on colitis. Structural characterization showed that PSP-W-1 was composed of T-Galp, 1,4-Galp and 1,4,6-Galp. It is a polysaccharide containing only galactose components, whose molecular weight is 14.4 kDa. In vivo experiment results of the mice model have shown that PSP-W-1 can aleviate colitis by inhibiting inflammatory factors as wel as inflammatory mediators, enhancing the expression level of tight junction proteins and maintaining the balance of gut microbiota. Moreover, the Polygonatum cyrtonema is a medicinal and food plant which is beneficial to health. As a result, the Polygonatum cyrtonema has the potential to be developed as a functional food for the prevention and aleviation of colitis. [00174] The terms “comprising” or “comprise” are used herein in their broadest sense to mean and encompass the notions of “including,” “include,” “consist(ing) essentialy of,” and “consist(ing) of.” The use of “for example,” “e.g.,” “such as,” and “including” to list ilustrative examples does not limit to only the listed examples. Thus, “for example” or “such as” means “for example, but not limited to” or “such as, but not limited to” and encompasses other similar or equivalent examples. The term “about” as used herein serves to reasonably encompass or describe minor variations in numerical values measured by instrumental analysis or as a result of sample handling. Such minor variations may be in the order of ±0-10, ±0-5, or ±0-2.5, % of the numerical values. Further, the term “about” applies to both numerical values when associated with a range of values. Moreover, the term “about” may apply to numerical values even when not explicitly stated.
Aty. Docket. No.018716.208867 (WN3426) [00175] Generaly, as used herein a hyphen “-” or dash “–” in a range of values is “to” or “through”; a “>” is “above” or “greater-than”; a “≥” is “at least” or “greater-than or equal to”; a “<” is “below” or “less-than”; and a “≤” is “at most” or “less-than or equal to.” On an individual basis, each of the aforementioned applications for patent, patents, and/or patent application publications, is expressly incorporated herein by reference in its entirety in one or more non- limiting embodiments. [00176] It is to be understood that the appended claims are not limited to express and particular compounds, compositions, or methods described in the detailed description, which may vary between particular embodiments which fal within the scope of the appended claims. With respect to any Markush groups relied upon herein for describing particular features or aspects of various embodiments, it is to be appreciated that diferent, special, and/or unexpected results may be obtained from each member of the respective Markush group independent from al other Markush members. Each member of a Markush group may be relied upon individualy and or in combination and provides adequate support for specific embodiments within the scope of the appended claims. [00177] It is also to be understood that any ranges and subranges relied upon in describing various embodiments of the present invention independently and colectively fal within the scope of the appended claims, and are understood to describe and contemplate al ranges including whole and/or fractional values therein, even if such values are not expressly writen herein. One of skil in the art readily recognizes that the enumerated ranges and subranges suficiently describe and enable various embodiments of the present invention, and such ranges and subranges may be further delineated into relevant halves, thirds, quarters, fifths, and so on. As just one example, a range “of from 0.1 to 0.9” may be further delineated into a lower third, i.e., from 0.1 to 0.3, a middle third, i.e., from 0.4 to 0.6, and an upper third, i.e., from 0.7 to 0.9, which individualy and colectively are within the scope of the appended claims, and may be relied upon individualy and/or colectively and provide adequate support for specific embodiments within the scope of the appended claims. In addition, with respect to the language which defines or modifies a range, such as “at least,” “greater than,” “less than,” “no more than,” and the like, it is to be understood that such language includes subranges and/or an upper or lower limit. As another example, a range of “at least 10” inherently includes a subrange of from at least 10 to 35, a subrange of from at least 10 to 25, a subrange of from 25 to 35, and so on, and each subrange may be relied upon individualy and/or colectively and provides adequate support for specific embodiments within the scope of the appended claims. Finaly, an individual
Aty. Docket. No.018716.208867 (WN3426) number within a disclosed range may be relied upon and provides adequate support for specific embodiments within the scope of the appended claims. For example, a range “of from 1 to 9” includes various individual integers, such as 3, as wel as individual numbers including a decimal point (or fraction), such as 4.1, which may be relied upon and provide adequate support for specific embodiments within the scope of the appended claims. [00178] The present invention has been described herein in an ilustrative manner, and it is to be understood that the terminology which has been used is intended to be in the nature of words of description rather than of limitation. Many modifications and variations of the present invention are possible in light of the above teachings. The present invention may be practiced otherwise than as specificaly described within the scope of the appended claims. The subject mater of al combinations of independent and dependent claims, both single and multiple dependent, is herein expressly contemplated.