WO2025088605A2 - Human growth hormone receptor antagonists and methods of use thereof - Google Patents
Human growth hormone receptor antagonists and methods of use thereof Download PDFInfo
- Publication number
- WO2025088605A2 WO2025088605A2 PCT/IL2024/051024 IL2024051024W WO2025088605A2 WO 2025088605 A2 WO2025088605 A2 WO 2025088605A2 IL 2024051024 W IL2024051024 W IL 2024051024W WO 2025088605 A2 WO2025088605 A2 WO 2025088605A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- formula
- antagonist
- otbu
- hgha
- lys
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K14/00—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- C07K14/435—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- C07K14/705—Receptors; Cell surface antigens; Cell surface determinants
- C07K14/72—Receptors; Cell surface antigens; Cell surface determinants for hormones
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/51—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent
- A61K47/54—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an organic compound
- A61K47/542—Carboxylic acids, e.g. a fatty acid or an amino acid
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/51—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent
- A61K47/62—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being a protein, peptide or polyamino acid
- A61K47/65—Peptidic linkers, binders or spacers, e.g. peptidic enzyme-labile linkers
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K14/00—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- C07K14/435—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- C07K14/575—Hormones
- C07K14/61—Growth hormone [GH], i.e. somatotropin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K38/00—Medicinal preparations containing peptides
Definitions
- growth hormone receptor antagonists and modified growth hormone receptor antagonists are useful in treating diseases or conditions, such as cancer and acromegaly, that are responsive to human growth hormone receptor antagonists.
- the described invention relates in general to compositions for use as receptor antagonists, and more specifically to human growth hormone antagonists that have the potential to be highly effective therapeutics.
- Human growth hormone also known as somatotropin or somatropin, is a peptide hormone that stimulates growth, cell reproduction, and regeneration in humans and other animals.
- Growth hormone is a type of mitogen that is specific only to certain kinds of cells and is a 191 -amino acid, single-chain polypeptide that is synthesized, stored, and secreted by somatotropic cells within the lateral wings of the anterior pituitary gland.
- a receptor is a protein molecule usually found embedded within the plasma membrane surface of a cell that receives diemical signals from outside the cell. When such diemical signals bind to a receptor, they cause some form of cellular/tissue response such as, for example, a change in the electrical activity of the cell. In this sense, a receptor is a protein molecule that recognizes and responds to endogenous chemical signals.
- An agonist such as human growth hormone, is a chemical composition that binds to a receptor and activates the receptor to produce a biological response.
- a receptor antagonist is a type of receptor ligand or drug that blocks or dampens agonist-mediated responses rather than provoking a biological response itself upon binding to a receptor.
- These compositions are sometimes called blockers and examples include alpha blockers, beta blockers, and calcium channel blockers.
- antagonists have affinity but no efficacy for their cognate receptors, and binding will disrupt the interaction and inhibit the function of an agonist or inverse agonist at receptors.
- Antagonists mediate their effects by binding to the active (orthosteric) site or to other (allosteric) sites on receptors, or they may interact at unique binding sites not normally involved in the biological regulation of the receptor’s activity. Antagonist activity may be reversible or irreversible depending on the longevity of the antagonist-receptor complex, which, in turn, depends on the nature of antagonist-receptor binding. The majority of drug antagonists achieve their potency by competing with endogenous ligands or substrates at structurally defined binding sites on receptors. By definition, antagonists display no efficacy to activate the receptors they bind and antagonists do not maintain the ability to activate a receptor. Once bound, however, antagonists inhibit the function of agonists, inverse agonists, and partial agonist
- Acromegaly is a syndrome that results when the anterior pituitary gland produces excess human growth hormone (hGH) after epiphyseal plate closure at puberty. If hGH is produced in excess prior to epiphyseal plate closure, the result is gigantism (or giantism).
- hGH human growth hormone
- gigantism or giantism
- a number of disorders may increase the pituitary’s hGH output, although most commonly it involves a tumor called pituitary adenoma, derived from a distinct type of cell (somatotrophs).
- somatotrophs somatotrophs
- Acromegaly most commonly affects adults in middle age and can result in severe disfigurement, complicating conditions, and premature death if untreated. Because of its pathogenesis and slow progression, the disease is hard to diagnose in the early stages and is frequently missed for years until changes in external features, especially of the face, become noticeable.
- Changes associated with acromegaly include coarsening of body hair, thickening and darkening of the skin, enlargement and overactivity of sebaceous and sweat glands such that patients frequently complain of excessive perspiration and offensive body odor, overgrowth of the mandible, cartilaginous proliferation of the larynx causing a deepening of the voice, and enlargement of the tongue.
- excess growth hormone (“GH”) in these patients is responsible for proliferation of articular cartilage which may undergo necrosis and erosion and endoneurial fibrous proliferation which causes peripheral neuropathies. Excess GH also increases tubular reabsorption of phosphate and leads to mild hyperphosphatemia. Many of these symptoms are also seen in patients with gigantism
- IGF-1 insulin-like growth factor- 1
- DM diabetes mellitus
- patients with poorly controlled DM have been found to have high levels of circulating GH
- hypophysectomy could reduce diabetic hyperglycemia, thus strongly implicating the role of GH as an active component of the metabolic derangements of diabetes (Houssay and Biasotti, Rev. Soc. Argent Biol. 6: 251-296 (1930)).
- hypersecretion of GH may be the cause as much as the consequence of poor diabetic control (Press etal, New England J. Med. 310: 810-814 (1984)).
- Diabetes-related nephropathy is another disorder that has the potential to be treated therapeutically using growth hormone receptor antagonists. It has been established that that in various experimental models of type 1 diabetes, GH and IGFs have measurable effects on both short- and long-term renal changes (Alex F. Muller and others, Growth Hormone Receptor Antagonists, The Journal of Clinical Endocrinology & Metabolism, Volume 89, Issue 4, 1 April 2004, Pages 1503- 1511). The hypothesis that GHR antagonists may be used in the treatment of diabetic renal changes was supported by long-term studies in streptozotocin-induced diabetic mice transgenic for a GHR antagonist Somatostatin analogs, but not GHR antagonists, have been studied in patients with type 1 diabetes. Human studies investigating the renal effects of GHR blockade in diabetes mellitus (type 1 or 2) have not been performed, and there remains a need for GHR antagonists with potential beneficial effects in diabetic nephropathy.
- Proliferative diabetic retinopathy is one of the leading causes of blindness in the United States and ranks second only to senile macular degeneration as a cause of permanent blindness.
- Benson et al. Diabetic Retinopathy, Duane, T., (eds.), Harper & Row, Philadelphia, Pa. pp. 1-24.
- In juveniles with insulin dependent diabetes there is no evidence of diabetic retinopathy up to 5 years. However, 27% of juveniles who have had diabetes for 5 to 10 years have diabetic retinopathy. Also 71% of juveniles who have had diabetes for longer than 10 years have diabetic retinopathy. Greater than 90% of jirveniles who have diabetes for 30 years will ultimately have diabetic retinopathy.
- the 5 year mortality rate for individuals blind from diabetic retinopathy is 36%, in which death generally is caused by cardiac or kidney complications.
- the kidneys are another organ that is affected by DM. Chen et al., Endocrinology 136:660- 667 (1995).
- One type of pathology seen in patients with diabetic nephropathy is glomerulosclerosis.
- Glomerulosclerosis is the sclerosis of mesangial cells which is preceded by mesangial cell proliferation Glomerular cells are responsible for filtering the blood and thus directly affect kidney function
- hGH antagonist (Somavert) is covalently bound to several polyethylene glycol (PEG) polymers. It acts by blocking the cell surface GH receptor and preventing its activation. In order to modify hGH to hGH antagonist a single mutation of Glyl20 is required. The hGH antagonist binds to the hGHR but there is no signal transduction. Since the efficacy of antagonist depends on its affinity to the receptor, additional point mutations were introduced and were shown to improve its affinity to GHR
- SOMAVERT® (pegvisomant) is an analog of human growth hormone (“hGH”) that has been structurally altered.
- the amino acid sequence of the protein component/intermediate (B-2036) of pegvisomant differs from the amino acid sequence of hGH at nine positions.
- the specific amino add substitutions are as follows: H18D, H21N, G120K, R167N, K168A, D171S, K172R, E174S, and I179T.
- the first letter i.e., H18D
- H18D represents the amino add in the sequence of hGH at the numbered position (i.e., 18th amino add position as indicated by H18D) which is substituted with the amino acid designated by the second letter (i. e. , Hl 8D). Therefore, Hl 8D designates a substitution of the amino acid his by the amino acid asp at the 1 Sth amino acid position of the wild-type hGH amino acid sequence.
- pegvisomant is a protein (containing 191 amino acid residues) to which predominantly 4 to 6 PEG units are covalently bound.
- SOMAVERT® is a genetically-engineered analogue of hGH that functions as a highly selective growth hormone receptor antagonist indicated for the treatment of acromegaly (van der Lely, et al., The Lancet 358: 1754-1759 (2001)). Such compositions are used if the tumor of the pituitary gland causing the acromegaly cannot be controlled with surgery or radiation and the use of somatostatin analogues is unsuccessful.
- HSA Human serum albumin
- albumin is the most abundant protein in plasma (-60% of all proteins, -40 [g/L]). It is a highly soluble and stable (pH, structural, and temp), nonglycosylated, negatively charged (and thus avoids filtration in the renal glomeruli) and IS considered to be highly hydrophilic. Albumin contains 17 disulfide bridges that contribute to its structural stability, thermal stability, and a single odd cysteine residue in position 34. Overall, the Cys34 residue makes up -80% of the free thiols in plasma. HSA is synthesized as a 585- residue single chain globular protein lacking prosthetic groups and glycosylation
- Albumin possess an extremely long half-life (-19 days). A central contributor to the latter is its ability to bind the FcRn receptor, be rescued, and be recycled into the blood stream (Larsen, Maja Thim, et al. "Albumin-based drug delivery: harnessing nature to cure disease.” Molecular and cellular therapies 4.1 (2016): 1-12). Almost every body fluid contains some amount of HSA In addition, HSA occurs within cells like ovarian cells, brain cells, peripheral nerve cells, lymphocytes, macrophages, and other cells. Tumor cells often take up HSA to a greater extent than non- tumorous cells of the same type. For example, albumin makes up 19% of the soluble protein of breast cancer cells.
- albumin long half-life, allowing it to take part in numerous important physiological activities. Due to its versatile and multiple binding domains it serves as a transporter and stabilizer to a variety of molecules such as: fatty acids, aromatics, ions, and peptides. The latter characteristics sparkled the imagination of scientist for designing Albuminbinding drugs that display an increased half-life.
- the Neonatal FC receptor (“FcRN”) was first found to the responsible for transporting antibodies of IgG class from the mother to the fetus. Since the early discovery, it was found that the FcRN receptor is broadly expressed in many other tissues, and despite the unrelated structure, can also bind Albumin (Zorzi, Alessandro, Sara Linciano, and Alessandro Angelini. "Non-covalent albumin-binding ligands for extending the circulating half-life of small biotherapeutics.” MedChemComm 10.7 (2019): 1068-1081).
- FIG. 1 An illustration of the Albumin binding sites with respect to the Fatty acids, bi-valent ions, and known drugs is presented in FIG. 1. It was established that the Din and DI loop are highly important for the FcRn binding, and that there are several fatty adds binding sites located at these regions as well. Therefore, upon designing an albumin-binding drug, must take into consideration the potential alternation of the Albumin conformation, and thus hampering its FcRn binding.
- LCFAs Long chain fatty adds
- carboxylic adds having a non-branched aliphatic chain having 16-20 carbon atoms in its backbone
- HSAs HSAs
- Fatty adds can be conjugated to therapeutic protdns to form longer-acting derivatives.
- This principle for prolongation of protdn or peptide half-life is based on the fad that FA can bind to human serum albumin (HSA; also referred to as albumin binding probes).
- HSA human serum albumin
- the association of a FA with human serum albumin in the blood stream can lead to a substantial prolongation of the half-life of the therapeutic protein as it will recycle together with albumin through the neonatal Fc receptor.
- FA and derivatives thereof e.g., corresponding methyl esters
- have shown similar albumin-binding properties Spector AA, J Lipid Res 1975; 16: 165-79.
- the present application has developed new hGH antagonists and harnessed the benefits of albumin binding to produce modified hGH antagonists with improved pharmacokinetic and therapeutic properties.
- GHR antagonist ’ or “hGHA”
- hGHA human growth hormone receptor antagonist
- G120K SEQ ID NO: 2
- Hl 8D H21N, F54P, R64K, R167N, D171S, E174S, I179T.
- any of said mutations increase the affinity of the GHR antagonist to the growth hormone receptor.
- the hGHA further comprises at least one amino acid mutation to cysteine.
- the amino acid mutated to cysteine is T3, E39, P48, Q69, N99, L129, T135, T142, H151 or combinations thereof.
- the amino add mutated to cysteine is L129 or T135.
- the hGHA comprises the amino acid sequence of SEQ ID NO: 5.
- the hGHA has the following amino add mutations: G120K, L129C, R167N, D171S, E174S, andI179T.
- the hGHA comprises the amino acid sequence of SEQ ID NO: 6.
- the hGHA has the following amino acid mutations: H18D, H21N, F54P, R64K, G120K, L129C, R.167N, D171S, E174S, andI179T.
- the hGHA comprises the amino acid sequence of SEQ ID NO: 7.
- the hGHA has the following amino acid mutations: H18D, H21N, G120K, L129C, R167N, D171S, E174S, andI179T.
- the hGHA comprises the amino acid sequence of SEQ ID NO: 8.
- the hGHA has the following amino acid mutations: H18D, H21N, G120K, T135C, R167N, D171S, E174S, andI179T.
- the hGHA comprises the amino acid sequence of any one of SEQ ID NOs: 5 to 8.
- modified GHR antagonist or “modified hGHA” having the structure of formula (I): W-X-Y-Z (I) wherein:
- W represents a binder
- X represents a spacer
- Y represents an optional linker
- Z represents a human growth hormone receptor antagonist (“GHR antagonist”); or a pharmaceutically acceptable salt thereof.
- the element Z of the modified hGHA comprises any of the hGHA disclosed herein.
- the element Z of formula I comprises any of the hGHA disclosed herein.
- the GHR antagonist of formula I is any of the hGHA disclosed herein.
- the element Z of the modified hGHA comprises the amino add sequence of any one of SEQ ID NOs: 5 to 8.
- the element Z of formula I comprises the amino acid sequence of any one of SEQ ID NOs: 5 to 8.
- the GHR antagonist of formula I comprises the amino add sequence of any one of SEQ ID NOs: 5 to 8.
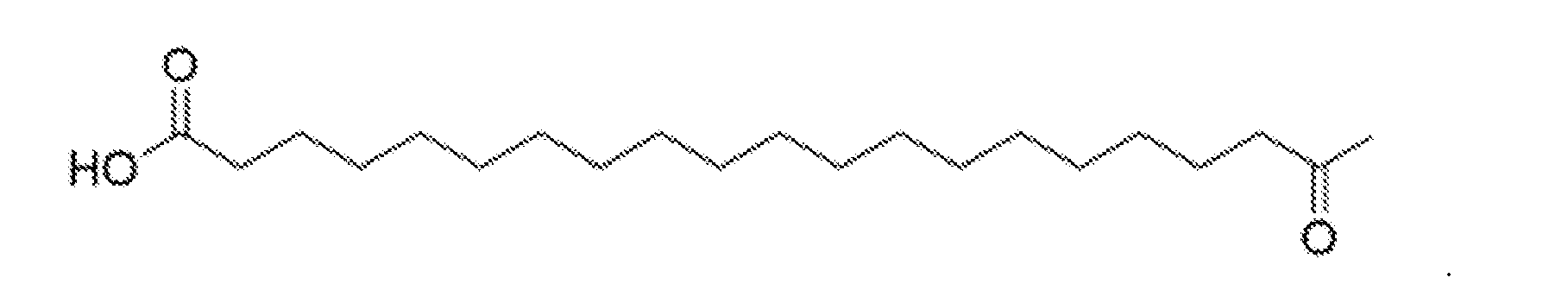
- W of formula I is a fatty add.
- W of formula I is octadecanedioic acid (Cl 8 diacid) or is eicosanedioic add (C20 diacid).
- W of formula I is represented by Formula II:
- W of formula I is represented by Formula II-A:
- W of formula I is represented by Formula HI: o
- W of formula I is represented by Formula HI -A: o
- the element X of formula I is gGlu-dun-(AEEA)m-Cys-Glyp, gGlu-Glun- (AEEA)m-Lys(AcBr)-Glyp, or gGlu-Glun-(AEEA)m-Lys-Glyp and n is 1, 2, or 3; m is 1, 2, or 3; and p is 1, 2, or 3.
- the element X of formula I is gGu-Gita-(AEEA)n-Cys-Oyn, n is 1, 2, or 3. In another aspect, the element X of formula I is gGlu-Glu-(AEEA)m-Cys-Gly, m is 1, 2, or 3.
- the element X of formula I is represented by Formula IV: n- 1, 2, 3
- the element X of formula I is gGlu-Glu-(AEEA)m-Cys-Gly, m is 1, 2, or 3 and is represented by Formula IV.
- the element X of formula I is represented by Formula IV-B: n» 1 ; 2, 3
- the element X of formula I is gGlu-Glu-(AEEA)m-Lys(AcBr)-Gly, m is 1, 2, or 3.
- the element X of formula I is gGlu-Glu-(AEEA)m-Lys-Gly, m is 1, 2, or 3.
- the element X of formula I is represented by Formula VI: n- 1, 2, 3
- the element X of formula I is gGlu-Glu-(AEEA)m-Lys-Gly, m is 1, 2, or 3.
- the element X of formula I is represented by Formula VI- A: n ⁇ 1,2, 3
- the element X of formula I is represented by Formula VI-B: n*1,2 t 3
- the element Y of formula I is Chloropropane-2-one-Fmoc-Mal.
- the element Y of formula I is 2-(3-(2,5-dioxo-2H-pyrrol-l(5H)-yl)propanamido)-9H-fluoren- 9-yl)methyl 3-chloro-2-oxopropylcarbamate.
- the element Y of formula I is Chloropropane-2-one-Fmoc-Mal and is represented by Formula VII:
- the element Y of formula I is Chloropropane-2-one-Fmoc-Mal and is represented by Formula VII- A: O o jy .
- the element Y of formula I is Chloropropane-2-one-Fmoc-Mal and is represented by Formula VII -B:
- the element Y of formula I is Chloropropane-2-one-Fmoc-Mal and is represented by Formula VH-C: O o jy .
- the element Y of formula I is Mal-NRFmoc-NHS.
- the element Y of formula I is Mal-NRFmoc-NHS and is represented by Formula X:
- the element Y of formula I is Mal-NRFmoc-NHS and is represented by Formula X-A: O o
- the element Y of formula I is Mal-NRFmoc-NHS and is represented by Formula X-B:
- the element Y of formula I is Mal-NRFmoc-NHS and is represented by Formula
- the bond between the GHR antagonist and the linker in formula I is a stable covalent bond.
- the bond between the GHR antagonist and the linker in formula I is a reversible covalent bond.
- the modified GHR antagonist comprises the following formula:
- the modified GHR antagonist comprises the following formula: O. Vss.
- the modified GHR antagonist comprises the following formula:
- the modified GHR antagonist comprises the following formula:
- the modified GHR antagonist comprises the following formula:
- the modified GHR antagonist comprises the following formula:
- the modified GHR antagonist comprises the following formula:
- the modified GHR antagonist comprises the following formula:
- the modified GHR antagonist comprises the following formula:
- the modified GHR antagonist comprises the following formula:
- the modified GHR antagonist comprises the following formula:
- the modified GHR antagonist comprises the following formula:
- a method of inhibiting human growth hormone (hGH) action in a subject comprising: administering to a subject in need thereof an effective amount of any GHR antagonist disclosed herein or any modified GHR antagonist disclosed herein, to inhibit hGH action in the subject
- the method disclosed comprises administering to any of the GHR antagonists or any of the modified GHR antagonists disclosed herein in an amount effective to treat, prevent, or ameliorate a H hGH gh related disorder in the subject
- the hGH related disorder is one or more of: acromegaly; age-related macular degeneration; cancer; diabetes; gigantism, vascular eye diseases, diabetic retinopathy, retinopathy of prematurity, and retinopathy of sickle-cell anemia; and, nephropathy.
- a method of treating, preventing, or ameliorating a hGH related disorder comprising: administering to a subject in need thereof an effective amount of any GHR antagonist disclosed herein or any modified GHR antagonist disclosed herein, to treat, prevent, or ameliorate a hGH related disorder in the subject
- the synthesizing of a binder backbone is done using solid-phase peptide synthesis (SPPS).
- SPPS solid-phase peptide synthesis
- the binder backbone is tBu-Fatty acid-gGlu(OtBu)-Glu(OtBu)-(AEEA)i- Lys(Mtt)-Gly-Wang resin, wherein i is 2 or 3 and the Fatty acid is eicosanedioic or octadecandioic.
- the removing in step (b) is done by applying 1%TFA in DCM wash cycles. In a related aspect, the removing in step (b) deprotects the Lys side chain.
- the intermediate composition following step (b) is the tBu-eiocosanedioc-yGiu(OtBu)-Glu(C)tBu)- (AEEA)3-Lys-Gly-Wang resin, tBu-eiocosanedioc-yGlu(C)tBu)-Glu(C)tBu)-(AEEA)2-Lys-Gly- Wang resin, tBu-octadecandioic-yGlu(OtBu)-Glu(C)tBu)-(AEEA)3-Lys-Gly-Wang resin, or tBu- octadecandioic-yGlu(OtBu)-Glu(C)tBu)-(AEEA)2-Lys-Gly-Wang resin.
- the linker moiety and deprotected binder backbone are added to a reactor and stirred.
- the linker moiety is coupled to the primary amine of the deprotected Lys side chain of the peptide backbone.
- the resin is Wang resin.
- the said cleaving in step (d) is done with a cocktail containing 95%TFA: 5% H2O, with a ratio of 10ml cleavage cocktail: lgr peptide-resin.
- the purifying in step (e) is done on RP-HPLC column.
- the linking moiety in step (c) is MAL-NRFmoc.
- around 2-3eq of the MAL-NRFmoc is added to the reactor.
- the pH in the reactor is around 5.0.
- the linking moiety in step (c) is Acetyl-Br.
- the Bromoacetic anhydride solution is added to the reactor and stirred.
- the stirring comprises 1 hour.
- the albumin binding complex is Formula XXXII: n « 15,17 i « 1,2,3
- the albumin binding complex is the albumin binding complex is tBu- eicosanedioic-yGlu(OtBu)-Glu(OtBu)-(AEEA)3-Lys(Ac-Br)-Gly-Wang resin, tBu-eicosanedioic- yGlu(OtBu)-Glu(OtBu)-(AEEA)2-Lys(Ac-Br)-Gly-Wang, resin tBu-octadecandioic-yGiu(OtBu)- Glu(OtBu)-(AEEA)3-Lys(Ac-Br)-Gly-Wang resin, or tBu-octadecandioic-yGiu(OtBu)-Glu(C)tBu)- (AEEA)2-Lys(Ac-Br)-Gly-Wang resin
- albumin binding complex is Formula XXXIII: n * 15,17 i » 1.2,3
- the albumin binding complex is tBu-eicosanedioic-yGlu(C)tBu)-Glu(C)tBu)-
- albumin binding complexes comprising Formula XXXII or Formula XXXIII.
- albumin binding complexes comprising tBu- eicosanedioic-yGlu(OtBu)-Glu(C)tBu)-(AEEA)3-Lys(Ac-Br)-Gly-Wang resin, tBu-eicosanedioic- ydu(OtBu)-Glu(C)tBu)-(AEEA)2-Lys(Ac-Br)-Gly-Wang resin, tBu-octadecandioic-yGlu(C)tBu)- Glu(OtBu)-(AEEA)3-Lys(Ac-Br)-Gly-Wang resin, or tBu-octadecandioic-yGlu(C)tBu)-Glu(C)tBu)- (AEEA)2-Lys(Ac-Br)-Gly-Wang resin.
- albumin binding complexes comprising tBu-eicosanedioic-yGlu(C)tBu)-du(C)tBu)-(AEEA)3-Lys(MAL-NRFmoc)- dy-Wang resin, tBu-eicosanedioic j ydu(OtBu)-du(OtBu)-(AEEA)2-Lys(MAL-NRFmoc)-Gly- Wang resin, tBu-octadecandioic j yGlu(OtBu)-Glu(OtBu)-(AEEA)3-Lys(MAL-NRFmoc)-dy-Wang resin, or tBu-octadecandioic-yGlu(C)tBu)-Glu(C)tBu)-(AEEA)2-Lys(MAL-NRFmoc)-Gly-Wang resin.
- BRIE tBu-e
- FIG. 1 is adapted from Ghuman, Jamie et al., “Structural basis of the drug-binding specificity of human serum albumin. ” Journal of molecular biology 353 1 (2005): 38-52, Figure 7, and shows the crystal structure of human albumin.
- FIG. 1 shows the crystal structure of human albumin solved in the presence of saturating amounts of palmitic acid.
- the a-helical structures of the three domains (DI, DII, and DM) are divided into subdomains (A and B) as indicated.
- Fatty acids binding sites are indicated via numbered circles (1-7) spread around the surface of the albumin three domains.
- FIG. 2 shows the generic formula for acylated Hgh antagonists with a binder complex containing a non-reversible Fmoc linker.
- the colored shading shows the different components of the acylated Hgh antagonist including: binder, spacer, Hnker, and Hgha Variant
- the value X corresponds to the Hgha Variants 1, 3, 4, and 5 described herein.
- FIG. 3 shows the generic formula for acylated Hgh antagonists with a binder complex containing a non-reversible Acetyl linker.
- the colored shading shows the different components of the acylated Hgh antagonist including: binder, spacer, Hnker, and Hgha Variant
- the value X corresponds to the Hgha Variants 1, 3, 4, and 5 described herein.
- FIGS. 4A-4D show representative binding assay results of different Hgha variants with two binder types (Binder Type A which is a Cl 8 albumin binding moiety and Binder Type B which is a C20 albumin binding) and without a binder (naked variants).
- FIG.4A shows the binding assay results of the reference standard (RS), Hgha Variant 1 (without any binder complex attached), acylated Hgha complex 1A, and acylated Hgha complex IB.
- FIG. 4B shows the binding assay results of the reference standard (RS), Hgha Variant 3 (without any binder complex attached), acylated Hgha complex 3A, and acylated Hgha complex 3B.
- FIG. 4C shows the binding assay results of the reference standard (RS), Hgha Variant 4 (without any binder complex attached), acylated Hgha complex 4A, acylated Hgha complex 4B.
- FIG. 4D shows the binding assay results of the reference standard (RS), Hgha Variant 5 (without any binder complex attached), acylated Hgha complex 5 A, and acylated Hgha complex 5B.
- FIGS. 5A and SB show representative binding assay results of acylated Hgh antagonists IB and ID compared to Somavert (FIG. SA) and acylated Hgh antagonists 5B and 5D compared to Somavert (FIG. SB).
- FIG. 6 shows the pharmacokinetic (PK) results of the acylated Hgh antagonists (1202-1A, 1202-1B, 1202-5 A, and 1202-5B) versus Somavert
- FIG. 7 shows the changes in IGF-1 serum levels (PD effects) in SD rats from study #12219, following single SC injection of the different acylated Hgha complexes and Somavert
- FIG. 8 shows the percentage changes in IGF-1 levels in SD rats from study #12219, following single SC injection of the different acylated Hgha complexes and Somavert
- FIG. 9 shows the PK results from Study #12220 which are based on non-compartmental pharmacokinetic analysis of the acylated Hgh antagonists (1202-1B and 1202-5B) at different dosages.
- FIG. 10 shows the changes in IGF-1 serum levels (PD effects) in SD rats from Study #12220, following single SC injection of the different acylated Hgha complexes and dosages.
- FIG. 11 shows the percentage changes in IGF-1 levels in SD rats from Study #12220, following single SC injection of the different acylated Hgha complexes and dosages.
- FIG. 12 shows the PK results based on non-compartmental pharmacokinetic analysis from Study #12222 of the acylated Hgh antagonists (1202-1D and 1202-5D) at different dosages.
- FIG. 13 shows the changes in IGF -1 serum levels (PD effects) in SD rats from Study #12222, following single SC injection of the different acylated Hgha complexes and dosages.
- FIG. 14 shows the percentage changes in IGF-1 levels in SD rats from Study #12222, following single SC injection of the different acylated Hgha complexes and dosages.
- FIGS. ISA and 15B show a process diagram for the manufacture of the acylated Hgha complexes disclosed herein.
- FIG. ISA shows the extraction and refolding phase.
- FIG. 15B shows the chromatographic phase.
- FIG. 16 shows a schematic illustration of the Albumin binder complex production routes.
- FIG. 17 shows RP-HPCD chromatograms.
- the top chromatograph shows the binder backbone before fatty add coupling (TFmoc), and the bottom chromatograph shows binder 1 backbone after fatty add coupling (8AA).
- FIG. 18 shows RP-HPCD chromatograms.
- the top chromatogram shows the binder before MAL-NRFmoc linker conjugation, and the bottom chromatogram shows the binder after linker conjugation
- FIG. 19 shows RP-HPCD chromatograms that represent the step of conjugating Ac-Br to the tBu- Eiocosanedioc-yGlu(OtBu)-Glu(OtBu)-(AEEA)3-Lys-Gly-Wang resin
- the top chromatogram shows the binder before Ac-Br linker conjugation
- the bottom chromatogram shows binder after linker conjugation
- FIG. 20 shows RP-HPCD chromatograms of purified Eicosanedioic-yGlu-Glu-(AEEA)3- Lys(Ac-Br)-Gly-OH in the top chromatogram, and Eicosanedioic-yGlu-Glu-(AEEA)3-Lys(MAL- NRFmoc)-Gly-OH in the bottom chromatogram.
- FIG. 21 shows mass spectrum of Eicosanedioic-yGlu-Glu-(AEEA)3-Lys(Ac-Br)-Gly-OH
- FIG. 22 shows mass spectrum of Eicosanedioic-yGlu-Glu-(AEEA)3-Lys(MAL-NRFmoc)- Gly-OH
- FIG.23 shows RP-HPLC chromatograms of Hgha conjugation reaction to an Albumin binder complex.
- the top chromatogram shows the Hgha solution; the middle chromatogram shows the Binder solution; and the bottom chromatogram shows the end of reaction (the acylated Hgha).
- FIG. 24 shows LC-MS analysis to the Hgha and the conjugated protein.
- the top chromatogram shows Hgha deconvoluted mass.
- the bottom chromatogram shows the conjugated protein deconvoluted mass.
- FIG.25 shows RP-HPLC chromatograms of Hgha conjugation reaction to an Albumin binder complex.
- the top chromatogram shows the Hgha solution; the middle chromatogram shows the Binder solution; and the bottom chromatogram shows the end of reaction (the acylated Hgha).
- FIG. 26 shows LC-MS analysis to the Hgha and the conjugated protein.
- the top chromatogram shows Hgha deconvoluted mass.
- the bottom chromatogram shows the conjugated protein deconvoluted mass.
- FIG. 27 shows the mean serum concentration of the MOD -1202-1 B and MOD-1202-1D acylated hGH antagonists in Study #OPK-009-PD.
- FIG. 28 shows the changes in IGF1 serum levels following repeated SC injection of the different compounds in Study &OPK-009-PD.
- FIG. 29 shows the changes in IGF1 relative to baseline following repeated SC injection of the different compounds in Study &OPK-009-PD.
- FIG. 30 shows the PK results of MOD-1202-1D at various dosages compared to Somavert in Study #OPK-OW-PD.
- FIG. 31 shows the changes in IGF1 serum levels following repeated SC injection of the different compounds in Study &OPK-010-PD.
- FIG. 32 shows the changes in IGF1 relative to baseline following repeated SC injection of the different compounds in Study &OPK-010-PD.
- a growth hormone antagonist refers to a compound that prevents, inhibits or reduces to any extent activation or expression of the growth hormone receptor.
- the compound that prevents or inhibits activation of the growth hormone receptor may act directly or indirectly on the growth hormone receptor.
- the compound may bind or interact directly with the growth hormone receptor in some embodiments.
- the compound may act indirectly by blocking access of the endogenous neuronal growth hormone to the growth hormone receptor.
- the compound may be able to block access of the endogenous neuronal growth hormone to the growth hormone receptor by interfering with the expression or activity of growth hormone or blocking the growth hormone receptor binding site on growth hormone or by preventing the release of endogenous neuronal growth hormone.
- human growth hormone receptor antagonist may be used interchangeably with the term “human growth hormone antagonist”, “growth hormone receptor antagonist’ ’, or “growth hormone antagonist”, having all the same meanings and qualities.
- growth hormone receptor antagonist may be used interchangeably with the term “growth hormone antagonist ’, “human growth hormone receptor antagonist”, or “human growth hormone antagonist”, having all the same meanings and qualities.
- Hgh The hormone-receptor complex between Hgh and the extracellular domain of its receptor (hGHbp) is known (Wells et al, Anna Rev. Biophys. Biomol. Struct, 22:329 (1993)).
- hGHbp The hormone-receptor complex between Hgh and the extracellular domain of its receptor
- GHR growth hormone receptor
- the present disclosure provides engineered Hgh antagonists resulted from introducing amino acid variations to Hgh.
- One of ordinary skill in the art would readily employ various standard techniques known in the art to introduce amino acid variations into Hgh antibody and then test the resulting modified Hgh for antagonist activity. While standard techniques may be used, the resultant antagonist activity of the newly created antibodies is not predictable and must be analyzed to determine functionality.
- Hgh polypeptide also includes glycosylated Hgh, such as but not limited to, polypeptides glycosylated at any amino add position, N-linked or O-linked glycosylated forms of the polypeptide. Variants containing single nucleotide or amino acid changes are also considered as biologically active variants of Hgh polypeptide. In addition, splice variants are also included.
- Hgh polypeptide also includes Hgh polypeptide heterodimers, homodimers, heteromultimers, or homomultimers of any one or more Hgh polypeptides or any other polypeptide, protein, carbohydrate, polymer, small molecule, linker, ligand, or other biologically active molecule of any type, linked by chemical means or expressed as a fusion protein, as well as polypeptide analogues containing, for example, specific deletions or other modifications yet maintain biological activity.
- Hgh from a growth agonist to a growth antagonist requires only a single amino acid change at Hgh position 120 from the native glycine to any amino acid except alanine (Chen et al., 1994).
- This molecule cannot be used as a therapeutic for conditions of excess growth (e.g., acromegaly) due to its short in vivo half-life.
- researchers have addressed this problem by the addition of polyethylene glycol molecules to the Hgh antagonist Hgh G120K to decrease the clearance of the molecule through the kidneys.
- Somavert® an FDA approved treatment for acromegaly, contains 4-6 linear PEG molecules with molecular weights of 5000 Daltons each.
- the growth hormone comprises a human growth hormone.
- the wild-type Hgh polypeptide sequence is: FPTIPLSRLFDNAMLRAHRLHQLAFDTYQEFEEAYIPKEQKYSFLQNPQTSLCFSESIPTPSN REETQQKSNLELLRISLLLIQSWLEPVQFLRSVFANSLVYGASDSNVYDLLKDLEEGIQTL MGRLEDGSPRTGQIFKQTYSKFDTNSHNDDALLKNYGLLYCFRKDMDKVETFLRIVQCRS VEGSCGF (SEQ ID NO: 1).
- the Hgh receptor antagonist GG112200KK sequence is: FPTIPLSRLFDNAMLRAHRLHQLAFDTYQEFEEAYIPKEQKYSFLQNPQTSLCFSESIPTPSN REETQQKSNLELLRISLLLIQSWLEPVQFLRSVFANSLVYGASDSNVYDLLKDLEEKIQTL MGRLEDGSPRTGQIFKQTYSKFDTNSHNDDALLKNYGLLYCFRKDMDKVETFLRIVQCRS VEGSCGF (SEQ ID NO: 2).
- the Hgh receptor antagonist G120K (SEQ ID NO: 2) corresponds to the native hGH polypeptide sEQUence with a single amino acid mutation G120K which converts Hgh to Hgh antagonist
- amino acid sequence of the Hgh antagonist corresponding to pegvisomant is:
- the signal peptide of the Hgh antagonist comprises the following amino acid sequence: MATGSRTSLLLAFCHXCLPWLQEGSA (SEQ ID NO: 4).
- the growth hormone comprises a human growth hormone antagonist (“Hgh antagonist’ “GH antagonist’ or “Hgha”).
- Hgh antagonist is a recombinant protein.
- Hgh antagonist is a recombinant glycoprotein.
- the GH antagonist is a growth hormone receptor (GHR) antagonist, a small molecule growth hormone receptor antagonist, a protein growth hormone receptor antagonist, a GHR inverse agonist, or an inhibitory nucleic add.
- GHR growth hormone receptor
- Protein or peptide GHR antagonists are well known in the art and include, but are not limited to pegvisomant (SOMAVERT®), B2036, B2036- PEG, G120R, G120RhGH, Hgh receptor antagonist G120K or analogs thereof.
- Hgh polypeptide encompasses Hgh polypeptides comprising one or more amino acid substitutions, additions or deletions.
- Hgh polypeptides of the present invention may be comprised of modifications with one or more natural amino acids in conjunction with one or more non-natural amino add modification Substitutions in a wide variety of amino acid positions in naturally-occurring Hgh polypeptides have been described, including but not limited to substitutions that modulate one or more of the biological activities of the Hgh polypeptide, such as but not limited to, increase antagonist activity, increase solubility of the polypeptide, decrease protease susceptibility, convert the polypeptide into an antagonist, etc. and are encompassed by the term “Hgh polypeptide”, “Hgh”, “hGHa”, or “GHR antagonisf’.
- amino add substitution resulting in antagonist activity is at residue 120 and is lysine.
- the Hgh antagonist is mutated to improve its binding characteristics (See Lowman, Henry B., and James A. Wells. “ Affinity maturation of human growth hormone by monovalent phage display.” Journal of molecular biology 234.3 (1993): 564-578).
- the number of mutations of the Hgh antagonist are selected to improve its binding characteristics.
- the Hgh antagonist contains between 4 and 8 mutations to improve its binding characteristics.
- the Hgh antagonist contains 4 mutations to improve its binding characteristics.
- the Hgh antagonist contains 5 mutations to improve its binding diaraderistics.
- the Hgh antagonist contains 6 mutations to improve its binding diaraderistics.
- the Hgh antagonist contains 7 mutations to improve its binding characteristics.
- the Hgh antagonist contains 8 mutations to improve its binding characteristics.
- the Hgh antagonist contains between 8 and 15 mutations to improve its binding characteristics. In another embodiment, the Hgh antagonist contains 8 mutations to improve its binding characteristics. In another embodiment, the Hgh antagonist contains 9 mutations to improve its binding characteristics. In another embodiment, the Hgh antagonist contains 10 mutations to improve its binding characteristics. In another embodiment, the Hgh antagonist contains 12 mutations to improve its binding characteristics. In another embodiment, the Hgh antagonist contains 13 mutations to improve its binding characteristics. In another embodiment, the Hgh antagonist contains 14 mutations to improve its binding characteristics. In another embodiment, the Hgh antagonist contains 15 mutations to improve its binding characteristics.
- the Hgh antagonist disclosed herein has the following point mutations as shown in Table 1, when comparing the amino acid sequence of the mutated Hgh antagonist to the Hgh antagonist G120K
- a human growth hormone receptor antagonist wherein at least one amino acid of the human growth hormone receptor antagonist has been mutated to cysteine.
- a human growth hormone receptor antagonist wherein at least one amino add of the human growth hormone receptor antagonist has been mutated to cysteine in order to introduce a cysteine as a conjugation site for the binder complex.
- a human growth hormone receptor antagonist wherein at least one amino acid of the human growth hormone receptor antagonist has been mutated to cysteine in order to introduce a cysteine as a conjugation site for the binder complex and without interrupting the protein-receptor interaction
- a human growth hormone receptor antagonist G120K wherein at least one amino add of the human growth hormone receptor antagonist G120K has been mutated to cystdne.
- a human growth hormone receptor antagonist G120K wherdn at least one amino acid of the human growth hormone receptor antagonist G120K has been mutated to cysteine in order to introduce a cysteine as a conjugation site for the binder complex.
- a human growth hormone receptor antagonist G120K wherein at least one amino acid of the human growth hormone receptor antagonist G120K has been mutated to cysteine in order to introduce a cysteine as a conjugation site for the binder complex and without interrupting the protein-receptor interaction
- the amino acid of the Hgh receptor antagonist which is mutated to cysteine is T3, E39, P48, Q69, N99, G120, L129, T135, T142, H151 or combinations thereof.
- the amino add of the Hgh receptor antagonist which is mutated to cysteine is T3, E39, P48, Q69, N99, G120, T142, Hl 51 or combinations thereof.
- the amino add of the Hgh receptor antagonist which is mutated to cysteine is L129 or T135 or combinations thereof.
- the amino acid of the Hgh receptor antagonist G120K which is mutated to cysteine is T3, E39, P48, Q69, N99, L129, T135, T142, H151 or combinations thereof. In another embodiment, the amino acid of the Hgh receptor antagonist G120K which is mutated to cysteine is T3, E39, P48, Q69, N99, T142, H151 or combinations thereof. In another embodiment, the amino acid of the Hgh receptor antagonist G120K which is mutated to cysteine is L129 or T135 or combinations thereof.
- the Hgh receptor antagonist has the following mutations: G120K, L129C, R167N, D171S, E174S, and I179T (“1202-1” or “Variant 1”).
- the mutation of L129C in Variant 1 allows for a conjugation site for a binder complex.
- the mutation of G120K in Variant 1 imparts antagonist activity to the composition
- the mutations R167N, D171S, E174S, and I179T improve the binding characteristics or increase binding affinity of Variant 1.
- the GHR antagonist of Variant 1 has the following mutations compared to Hgh: L129C; G120K; R167N; D171S; E174S; I179T.
- Variant 1 comprises the following amino add sequence: MFPTIPLSRLFDNAMLRAHRLHQLAFDTYQEFEEAYIPKEQKYSFLQNPQTSLCFSESIPTPS NREETQQKSNLELLRISLLUQSWLEPVQFLRSVFANSLVYGASDSNVYDLLKDLEEGIQTL MGRLEDGSPRTGQIFKQTYSKFDTNSHNDDALLKNYGLLYCFRKDMDKVETFLRIVQCRS VEGSCGF (SEQ ID NO: 5).
- the Hgh receptor antagonist has the following mutations: H18D, H21N, F54P, R64K, G120K, L129C, R167N, D171S, E174S, and I179T (“1202-3” or “Variant 3”).
- the mutation of L129C in Variant 3 allows for a conjugation site for a binder complex.
- the mutation of G120K in Variant 3 imparts antagonist activity to the composition
- the mutations H18D, H21N, F54P, R64K, R167N, D171S, E174S, and I179T improve the binding characteristics or increase binding affinity of Variant 3.
- the GHR antagonist of Variant 3 has the following mutations compared to Hgh: L129C; H18D; H21N; F54P; R64K; G120K; R167N; D171S; E174S; I179T.
- Variant 3 comprises the following amino acid sequence: MFPTIPLSRLFDNAMLRADRLNQLAFDTYQEFEEAYIPKEQKYSFLQNPQTSLCPSESIPTPS NKEETQQKSNLEUJUSUJJQSWLEPVQFLRSVFANSLVYGASDSNVYDLLKDLEEKIQTL MGRLCDGSPRTGQIFKQTYSKFDTNSHNDDALLKNYGLLYCFNKDMSKVSTFLRTVQCR SVEGSCGF (SEQ ID NO: 6).
- the Hgh receptor antagonist has the following mutations: H18D, H21N, G120K, L129C, R167N, D171S, E174S, and I179T (“1202 ⁇ ” or “Variant 4”).
- the mutation of L129C in Variant 4 allows for a conjugation site for a binder complex.
- the mutation of G120K in Variant 4 imparts antagonist activity to the composition.
- the mutations H18D, H21N, R167N, D171S, E174S, and I179T improve the binding characteristics or increase binding affinity of Variant 4.
- the GHR antagonist of Variant 4 has the following mutations compared to Hgh: L129C; H18D; H21N; G120K; R167N; D171S; E174S; I179T.
- Variant 4 comprises the following amino add sequence: MFPTIPLSRLFDNAMLRADRLNQLAFDTYQEFEEAYIPKEQKYSFLQNPQTSLCFSESIPTPS NREETQQKSNLELLRISLLUQSWLEPVQFLRSVFANSLVYGASDSNVYDLLKDLEEKIQTL MGRIXTXjSPRTGQIFKQTYSKFDTNSHNDDALLKNYGLLYCFNKDMSKVSTFLRTVQCR SVEGSCGF (SEQ ID NO: 7).
- the Hgh receptor antagonist has the following mutations: H18D, H21N, G120K, T135C, R167N, D171S, E174S, and I179T (“1202-5” or “Variant 5”).
- the mutation of T135C in Variant 5 allows for a conjugation site for a binder complex.
- the mutation of G120K in Variant 5 imparts antagonist activity to the composition.
- the mutations H18D, H21N, R167N, D171S, E174S, and I179T improve the binding characteristics or increase binding affinity of Variant
- the GHR antagonist of Variant 5 has the following mutations compared to Hgh: T135C; H18D; H21N; G120K; R167N; D171S; E174S; I179T.
- Variant 5 comprises the following amino add sequence: MFPTIPLSRLFDNAMLRADRLNQLAFDTYQEFEEAYIPKEQKYSFLQNPQTSLCFSESIPTPS NREETQQKSNLELLRISLLUQSWLEPVQFLRSVFANSLVYGASDSNVYDLLKDLEEKIQTL MGRLEDGSPRCGQIFKQTVSKFDTNSHNDDALLKNYGLLYCFNKDMSKVSTFLRTVQCR SVEGSCGF (SEQ ID NO: 8).
- the hGH receptor antagonist corresponds to the amino sequence of any of SEQ ID Nos: 5 to 8. In another embodiment, the hGH receptor antagonist corresponds to the amino acid sequence of any of SEQ ID NO: 5 to SEQ ID NO: 8. In another embodiment, the Hgh receptor antagonist corresponds to the amino acid sequence of any of SEQ ID Nos: 5 through 8. In another embodiment, the hGH receptor antagonist corresponds to the amino acid sequence of any of SEQ ID NO: 5 through SEQ ID NO: 8.
- the hGH receptor antagonist corresponds to the amino sequence of SEQ ID NO: 5. In one embodiment, the hGH receptor antagonist corresponds to the amino sequence of SEQ ID NO: 6. In one embodiment, the hGH receptor antagonist corresponds to the amino sequence of SEQ ID NO: 7. In one embodiment, the hGH receptor antagonist corresponds to the amino sequence of SEQ ID NO: 8.
- the invention includes a homologue of a growth hormone antagonist
- the invention includes a homologue of a human growth hormone antagonist.
- homologues e.g., polypeptides which are at least 50%, at least 55%, at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 87%, at least 89%, at least 91%, at least 93%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% homologous to a growth hormone or human growth hormone antagonist as determined using BlastP software of the National Center of Biotechnology Information (NCBI) using default parameters.
- NCBI National Center of Biotechnology Information
- the enhanced biological activity stems from the longer half-life of the engineered hGHA or acylated hGHA while maintaining at least some biological activity.
- the enhanced biological activity stems from enhanced biological activity resulting from the conjugation of the binder complex.
- the enhanced biological activity stems from both a longer half-life and from enhanced functionality of the binder complex.
- the hGHA or acylated hGHA includes a signal peptide. In another embodiment, the hGHA or acylated hGHA does not comprise a signal peptide.
- the hGH antagonists disclosed herein are modified. In another embodiment, the hGH antagonists disclosed herein are modified to increase albumin binding. In another embodiment, the hGH antagonists disclosed herein are modified to increase their half-life.
- the hGH antagonists disclosed herein are acylated. In another embodiment, the hGH antagonists disclosed herein are acylated to increase albumin binding. In another embodiment, the hGH antagonists disclosed herein are acylated to increase their half-life.
- acylated human growth hormone receptor antagonist acylated hGH antagonist, acylated hGHA, acylated human growth hormone receptor antagonist complex, acylated hGH antagonist complex, acylated hGHA complex, modified human growth hormone receptor antagonist, modified hGH antagonist, modified hGHA, modified human growth hormone receptor antagonist complex, modified hGH antagonist complex, and modified hGHA complex are synonymous in the present application.
- an "acylated" amino add is an amino add comprising an acyl group which is non-native to a naturally occurring amino add, regardless of how it is produced.
- Methods of produdng acylated amino adds and acylated peptides are known in the art and include acylating an amino acid before inclusion in the peptide or peptide synthesis followed by chemical acylation of the peptide.
- the acyl group causes the peptide to have one or more of (i) a prolonged half- life in circulation, (ii) a delayed onset of action, (iii) an improved resistance to proteases, or (iv) an extended duration of action.
- At least one amino acid of the human growth hormone receptor antagonist or human growth hormone receptor antagonist G120K is mutated to cysteine.
- at least one amino add of the human growth hormone receptor antagonist or human growth hormone receptor antagonist G120K is mutated to cysteine as a conjugation site for the binder complex.
- at least one amino acid of the human growth hormone receptor antagonist or human growth hormone receptor antagonist G120K is mutated to cysteine and without interrupting the protein-receptor interaction.
- cysteine is L129 or T135 of the human growth hormone receptor antagonist or human growth hormone receptor antagonist G120K is mutated to cysteine. These cysteine mutations introduce a cysteine as a conjugation site for the binder complex and without interrupting the protein-receptor interaction.
- cysteine is L129 or T135 of the human growth hormone receptor antagonist or human growth hormone receptor antagonist G120K is mutated to cysteine as a conjugation site for the reversible binder complex and without interrupting the protein-receptor interaction
- cysteine of the hGH receptor antagonist or the hGH receptor antagonist G120K is attached to the reversible binder complexes 1 or 2 as described in Table 7.
- cysteine is L129 or T135 of the human growth hormone receptor antagonist or human growth hormone receptor antagonist G120K is mutated to cysteine as a conjugation site for the permanent binder complex and without interrupting the protein-receptor interaction
- cysteine of the hGH receptor antagonist or the hGH receptor antagonist G120K is attached to the permanent binder complexes A, B, C, or D as described in Table 8.
- the present application provides new compounds which are capable of binding albumin for use in extending the half-life of biologically active moieties to which they are attached.
- Specifically present application provides new compounds which are capable of binding albumin for use in extending the half-life of hGH antagonists to which they are attached.
- albumin-binding compounds consisting essentially of the following elements: a binder, a spacer, a linker, and a biologically active moiety.
- the present application provides new compounds which are capable of binding albumin for use in extending the half-life of biologically active moieties to which they are attached. Specifically present application provides new compounds which are capable of binding albumin for use in extending the half-life of hGH antagonists to which they are attached.
- albumin-binding compounds consisting essentially of the following elements: a binder, a spacer, and a biologically active moiety.
- the present application provides new compounds which are capable of binding albumin for use in extending the half-life of biologically active moieties to which they are attached.
- new compounds which are capable of binding albumin for use in extending the half-life of hGH antagonists to which they are attached provides albumin-binding compounds consisting essentially of the following elements: a binder, a spacer, an optional linker, and a biologically active moiety.
- albumin binding moiety means a residue which binds non- covalently to human serum albumin.
- acylation with fatty acids delays clearance of the peptide it is attached to.
- the hGHA is acylated with an acyl group of sufficient size to bind serum albumin.
- the modified GHR antagonists consist of the following elements: the present application provides albumin-binding compounds consisting essentially of the following elements: a binder, a spacer, a linker, and a GHR antagonist In another embodiment, the modified GHR antagonists consist of the following elements: the present application provides albumin-binding compounds consisting essentially of the following elements: a binder, a spacer, a reversible linker, and a GHR antagonist In another embodiment, the modified GHR antagonists consist of the following elements: the present application provides albumin-binding compounds consisting essentially of the following elements: a binder, a spacer, a non-reversible linker, and a GHR antagonist
- the modified GHR antagonists consist of the following elements: the present application provides albumin-binding compounds consisting essentially of the following elements: a fatty acid chain, a spacer, a linker, and a GHR antagonist In another embodiment, the modified GHR antagonists consist of the following elements: the present application provides albumin-binding compounds consisting essentially of the following elements: a fatty add chain, a spacer, a reversible linker, and a GHR antagonist In another embodiment, the modified GHR antagonists consist of the following elements: the present application provides albumin-binding compounds consisting essentially of the following elements: a fatty acid chain, a spacer, a non- reversible linker, and a GHR antagonist
- the modified GHR antagonists consist of the following elements: the present application provides albumin-binding compounds consisting essentially of the following elements: a binder, a spacer, and a GHR antagonist In another embodiment, the modified GHR antagonists consist of the following elements: the present application provides albumin-binding compounds consisting essentially of the following elements: a fatty acid chain, a spacer, and a GHR antagonist
- the modified GHR antagonists consist of the following elements: the present application provides albumin-binding compounds consisting essentially of the following elements: a binder, a spacer, an optional linker, and a GHR antagonist In another embodiment, the modified GHR antagonists consist of the following elements: the present application provides albumin-binding compounds consisting essentially of the following elements: a binder, a spacer, an optional reversible linker, and a GHR antagonist. In another embodiment, the modified GHR antagonists consist of the following elements: the present application provides albumin-binding compounds consisting essentially of the following elements: a binder, a spacer, an optional non- reversible linker, and a GHR antagonist
- the modified GHR antagonists consist of the following elements: the present application provides albumin-binding compounds consisting essentially of the following elements: a fatty acid chain, a spacer, an optional linker, and a GHR antagonist In another embodiment, the modified GHR antagonists consist of the following elements: the present application provides albumin-binding compounds consisting essentially of the following elements: a fatty add drain, a spacer, an optional reversible linker, and a GHR antagonist In another embodiment, the modified GHR antagonists consist of the following elements: the present application provides albumin-binding compounds consisting essentially of the following elements: a fatty add chain, a spacer, an optional non-reversible linker, and a GHR antagonist
- modified GHR antagonists consisting essentially of:
- the modified GHR antagonists have the structure of formula (I): W-X-Y-Z (Formula I) wherein:
- W represents a binder
- X represents a spacer
- Y represents an optional linker
- GHR antagonist human growth hormone receptor antagonist
- the term “binder complex” refers to the binder, spacer, and an optional linker.
- the term “binder complex” refers to W, X, and Y of Formula I.
- the term “binder complex” refers to W and X of Formula I.
- the term “binder complex” refers to a fatty add chain, a spacer, and an optional linker.
- the term “binder complex” refers to a fatty acid chain and a spacer.
- binding complex can be used interchangeably with any of the following terms: “probe”, “albumin binding probe”, or “albumin-binding probe”.
- the W of Formula I is a fatty add chain.
- the binder of Formula I is a fatty acid chain.
- fatty add chain refers to the hydrocarbon backbone of fatty adds (excluding the terminal acidic group) containing 2 to 40 carbon atoms.
- Fatty acid chain can refer to a ctadecanedioic add (Cl 8 diacid) or eicosanedioic acid (C20 diacid).
- the octadecanedioic acid (Cl 8 diacid) fatty add has the following structure of Formula II:
- the eicosanedioic add (C20 diadd) fatty acid has the following structure of Formula HI: or has the following structure of Formula O-A:
- the W of Formula I is a hydrocarbon backbone of fatty acids containing 2 to 40 carbon atoms.
- the binder of Formula I is a hydrocarbon backbone of fatty acids containing 2 to 40 carbon atoms.
- the W of Formula I is octadecanedioic add (Cl 8 diacid) or eicosanedioic acid (C20 diadd).
- the binder of Formula I is octadecanedioic add (Cl 8 diacid) or eicosanedioic add (C20 diacid).
- the W of Formula I is octadecanedioic acid (C18 diacid) and is represented by Formula II:
- the W of Formula I is octadecanedioic acid (Cl 8 diadd) and is represented by Formula II-A:
- the W of Formula I is dcosanedioic add (C20 diadd) and is represented by Formula HI: o
- the W of Formula I is eicosanedioic acid (C20 diacid) and is represented by Formula O-A:
- the X of Formula I is a spacer.
- the X of Formula I is any one of gGiu-Giu n -(AEEA)m-Cys-Glyp, gdu- Giu n -(AEEA)m-Lys(AcBr)-Glyp, or gdu-Giu n -(AEEA)m-Lys-Glyp and n is 1, 2, or 3; m is 1, 2, or 3; and p is 1, 2, or 3.
- the X of Formula I is any one of gGiu-Giun-(AEEA) m -Cys- dyp, gGiu-Giu n -(AEEA)m-Lys(AcBr)-Gly, or gGlu-Giu n -(AEEA)m-Lys-Gly and n is 1, 2, or 3; m is 1, 2, or 3; and p is 1, 2, or 3.
- the X of Formula I is any one of gGlu-Glu-(AEEA)m-Cys-Gly, gGlu- Glu-(AEEA)m-Lys(AcBr)-Gly, or gGlu-Glu-(AEEA)m-Lys-Gly and m is 1, 2, or 3.
- the X of Formula I is any one of gdu-du-(AEEA) m -Cys-Gly, gGlu-Glu-(AEEA)m- Lys(AcBr)-Gly, or gGlu-Glu-(AEEA)m-Lys-Gly and m is 1.
- the X of Formula I is any one of gGlu-Glu-(AEEA)m-Cys-Gly, gGlu-Glu-(AEEA)m-Lys(AcBr)-Gly, or gGlu- Glu-(AEEA)m-Lys-Gly and m is 2. In another embodiment, the X of Formula I is any one of gGlu- Glu-(AEEA)m-Cys-Gly, gGlu-Glu-(AEEA)m-Lys(AcBr)-Gly, or gGlu-Glu-(AEEA)m-Lys-Gly and m is 3.
- the X of Formula I is represented by Formula IV: n- 1, 2, 3
- the X of Formula I is gGlu-Glu-(AEEA)m-Cys-Gly, m is 1, 2, or 3, and is represented by Formula IV.
- the X of Formula I is represented by Formula IV- A: n ⁇ 1, 2, 3
- the X of Formula I is represented by Formula IV-B: n» 1, 2 t 3
- the X of Formula I is represented by Formula IV-C: n « 1, 2, 3
- the X of Formula I is represented by Formula IV-D:
- the X of Formula I is represented by Formula V:
- n 1, 2. 3
- the X of Formula I is gdu-Glu-(AEEA)m-Lys(AcBr)-Gly, m is 1, 2, or 3, and is represented by Formula V.
- the X of Formula I is represented by Formula V-A: n» 1.2.3
- the X of Formula I is represented by Formula V-B: H Br
- the X of Formula I is represented by Formula V-C: n-1.2. 3
- the X of formula I is X is gGlu-Glu-(AEEA)m-Lys-Gly, m is 1, 2, or 3, and is represented by Formula VI.
- the X of formula I is represented by Formula VI-B:
- the X of formula I is represented by Formula VI-C: n* 1, 2 t 3
- the Y of Formula I is a linker. In a related embodiment, the Y of Formula I is a linker and links W and Z of Formula I.
- the Y of Formula I is an optional linker.
- the Y of Formula I is not present In a related embodiment, the Y of Formula I is not present, and Formula I then comprises W-X-Z.
- the Y of Formula I is Chloropropane-2-one-Fmoc-Mal or Mal-NRFmoc- NHS. In another embodiment, the Y of Formula I is Chloropropane-2-one-Fmoc-Mal. In another embodiment, the Y of Formula I is Mal-NRFmoc-NHS.
- the Y of F ormula I is 2-(3 -(2, 5-dioxo-2H-pyrrol-l (5H)-yl)propanamido)- 9H-fluoren-9-yl)methyl 3-chloro-2-oxopropylcarbamate.
- the Y of Formula I is a linker and is a stable covalent bond. In another embodiment, the Y of Formula I is a non-reversible linker. In another embodiment, the bond between the GHR antagonist and the linker of Formula I is a stable covalent bond. In another embodiment, the bond between Z and Y of Formula I is a stable covalent bond.
- the Y of Formula I is Mal-NRFmoc-NHS and is a stable covalent bond. In another embodiment, the Y of Formula I is Mal-NRFmoc-NHS and is a non-reversible linker. In another embodiment, the bond between the GHR antagonist and Mal-NRFmoc-NHS as the linker of Formula I is a stable covalent bond. In another embodiment, the bond between Z and Mal-NRFmoc- NHS is a stable covalent bond.
- the Y of Formula I is a linker and is a reversible covalent bond. In another embodiment, the Y of Formula I is a reversible linker. In another embodiment, the bond between the GHR antagonist and the linker of Formula I is a reversible covalent bond. In another embodiment, the bond between Z and Y of Formula I is a reversible covalent bond.
- the Y of Formula I is Chloropropane-2-one-Fmoc-Mal and is a reversible covalent bond. In another embodiment, the Y of Formula I is Chloropropane-2-one-Fmoc-Mal and is a reversible linker. In another embodiment, the bond between the GHR antagonist and Chloropropane-2-one-Fmoc-Mal is a reversible covalent bond. In another embodiment, the bond between Z and Chloropropane-2-one-Fmoc-Mal is a reversible covalent bond.
- the Y of Formula I is Chloropropane-2-one-Fmoc-Mal and is represented by Formula VII (in another embodiment the Chloropropane can be substituted with Bromopropane or lodopropane):
- the Y of formula I is Chloropropane-2-one-Fmoc-Mal and is represented by Formula VII- A:
- the Y of formula I is Chloropropane-2-one-Fmoc-Mal and is represented by Formula VH-B: O o jy .
- the Y of formula I is Chloropropane-2-one-Fmoc-Mal and is represented by Formula VH-C:
- the Y of Formula I is Chloropropane-2-one-Fmoc-Mal and is represented by Formula VII, and the Chloropropane can be substituted with Bromopropane or lodopropane.
- the Y of Formula I is Chloropropane-2-one-Fmoc-Mal and the Chloropropane can be substituted with Bromopropane or lodopropane.
- the Y of Formula I is Mal-Fmoc-Propyl bromide.
- the Y of Formula I is (2-(3-(2,5-dioxo-2H-pyrrol-l(5H)- yl)propanamido)-9H-fluoren-9-yl) methyl 3-bromo-2-oxopropylcarbamate.
- the Y of formula I is represented by Formula XXXIV: o o
- the Y of formula I is Mal-Fmoc-Propyl bromide and is represented by Formula XXXIV.
- the Y of Formula I is Mal-Fmoc-Propyl Chloride.
- the Y of Formula I is (2-(3-(2,5-dioxo-2H-pyrrol-l(5H)- yl)propanamido)-9H-fluoren-9-yl) methyl 3-chloro-2-oxopropylcarbamate.
- the Y of Formula I is represented by Formula XXXV: o
- the Y of Formula I is Mal-Fmoc-Propyl Chloride and is represented by Formula XXXV. [0264] In one embodiment, the Y of Formula I is Mal-Fmoc-Propyl Iodide.
- the Y of Formula I is (2-(3-(2,5-dioxo-2H-pyrrol-l(5H)- yl)propanamido)-9H-fluoren-9-yl)methyl 3 -Iodo-2 -oxopropylcarbamate.
- the Y of Formula I is represented by Formula XXXVI: o o
- the Y of Formula I is Mal-Fmoc-Propyl Iodide and is represented by Formula XXXVI.
- the Y of Formula I is Bromopropane -2-one-Fmoc-Mal and is represented by Formula VO: o
- the Y of Formula I is Iodopropane-2-one-Fmoc-Mal and is represented by Formula IX:
- the Y of formula I is represented by Formula X-A: O o
- the Y of formula I is represented by Formula X-B:
- the Y of formula I is represented by Formula X-C:
- the modified GHR antagonists disclosed throughout the application include pharmaceutically acceptable salts thereof.
- the GHR antagonists in the modified GHR antagonists disclosed include any the GHR antagonists previously disclosed.
- the GHR antagonists in the modified GHR antagonists disclosed include any of SEQ ID NO: 5 through SEQ ID NO: 8.
- the GHR antagonists in the modified GHR antagonists have an amino acid mutation to cysteine at T3, E39, P48, Q69, N99, G120, L129, T135, T142, H151 or combinations thereof. In another embodiment, the GHR antagonists in the modified GHR antagonists have an amino acid mutation to cysteine is T3, E39, P48, Q69, N99, G120, T142, H151 or combinations thereof. In another embodiment, the GHR antagonists in the modified GHR antagonists have an amino acid mutation to cysteine is L129 or T135 or combinations thereof.
- the GHR antagonist in the modified GHR antagonists has the following mutations: G120K, L129C, R167N, D171S, E174S, and I179T (“1202-1” or “Variant 1”) or comprises the amino add sequence of SEQ ID NO: 5.
- the GHR antagonist in the modified GHR antagonists has the following mutations: H18D, H21N, F54P, R64K, G120K, L129C, R167N, D171S, E174S, and I179T (“1202- 3” or “Variant 3 or has the amino add sequence of SEQ ID NO: 6.
- the GHR antagonist in the modified GHR antagonists has the following mutations: H18D, H21N, G120K, L129C, R167N, D171S, E174S, andI179T (“1202-4” or “Variant 4”) or has the amino add sequence of SEQ ID NO: 7. [0280] In one embodiment, the GHR antagonist in the modified GHR antagonists has the following mutations: H18D, H21N, G120K, T135C, R167N, D171S, E174S, andI179T (“1202-5” or “Variant 5”) or has the amin acid sequence of SEQ ID NO: 8.
- the GHR antagonist in the modified GHR antagonist corresponds to the amino sequence of any of SEQ ID Nos: 5 to 8. In another embodiment, the GHR antagonist in the modified GHR antagonist corresponds to the amino acid sequence of any of SEQ ID NO: 5 to SEQ ID NO: 8. In another embodiment, the GHR antagonist in the modified GHR antagonist corresponds to the amino add sequence of any of SEQ ID Nos: 5 through 8. In another embodiment, the GHR antagonist in the modified GHR antagonist corresponds to the amino acid sequence of any of SEQ ID NO: 5 through SEQ ID NO: 8.
- the GHR antagonist in the modified GHR antagonist corresponds to the amino sequence of SEQ ID NO: 5. In one embodiment, the GHR antagonist in the modified GHR antagonist corresponds to the amino sequence of SEQ ID NO: 6. In one embodiment, the GHR antagonist in the modified GHR antagonist corresponds to the amino sequence of SEQ ID NO: 7. In one embodiment, the GHR antagonist in the modified GHR antagonist corresponds to the amino sequence of SEQ ID NO: 8.
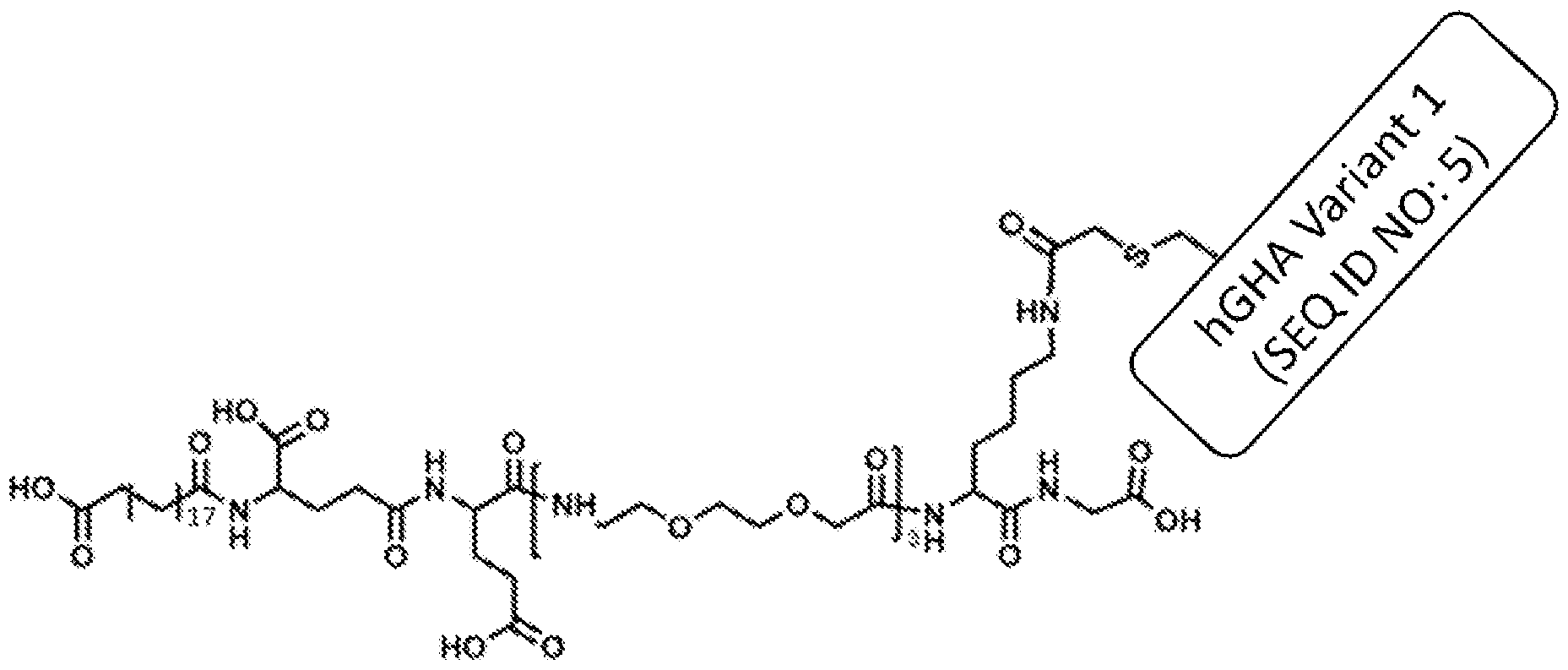
- the acylated hGHA complex is represented in FIG. 2 where n is 15 and X is 1 or hGHA Variant 1 (SEQ ID NO: 5). In another embodiment, the acylated hGHA complex is represented in FIG. 2 where n is 15 and X is 1. In another embodiment, the acylated hGHA complex is represented in FIG. 2 where n is 15 and X is 1 (hGHA Variant 1). In another embodiment, the acylated hGHA complex is represented in FIG. 2 where n is 15 and X is 1 (SEQ ID NO: 5).
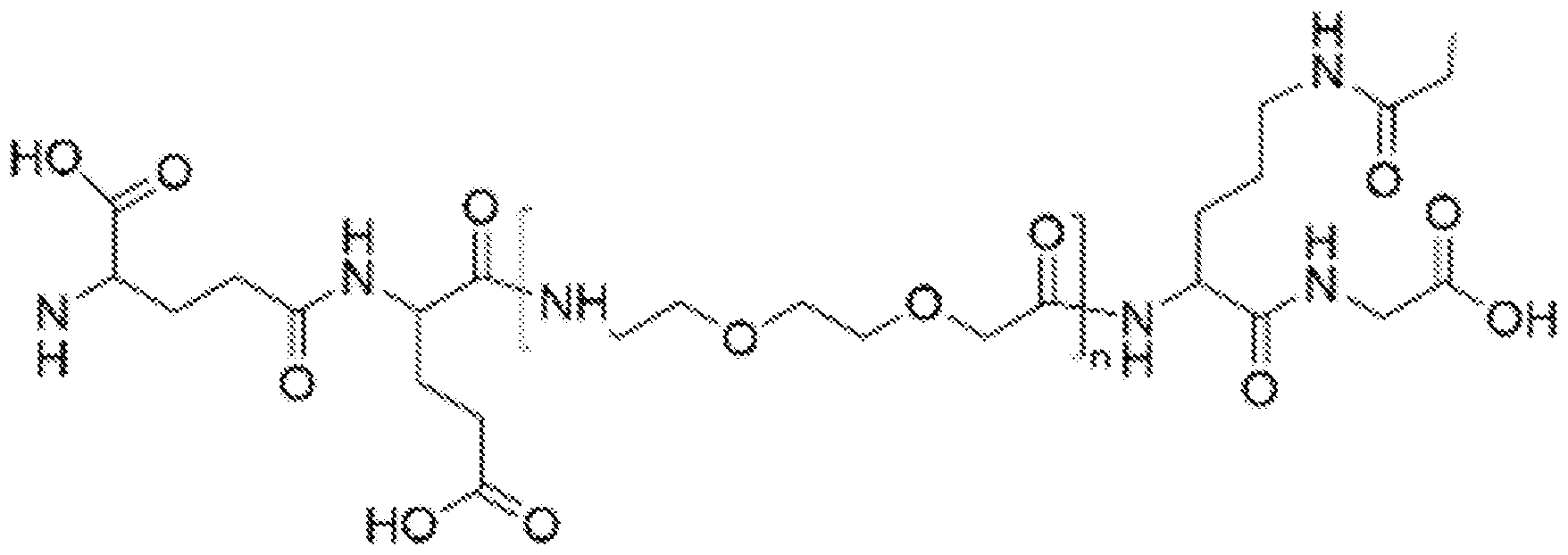
- the acylated hGHA complex is represented by the following chemical structure:
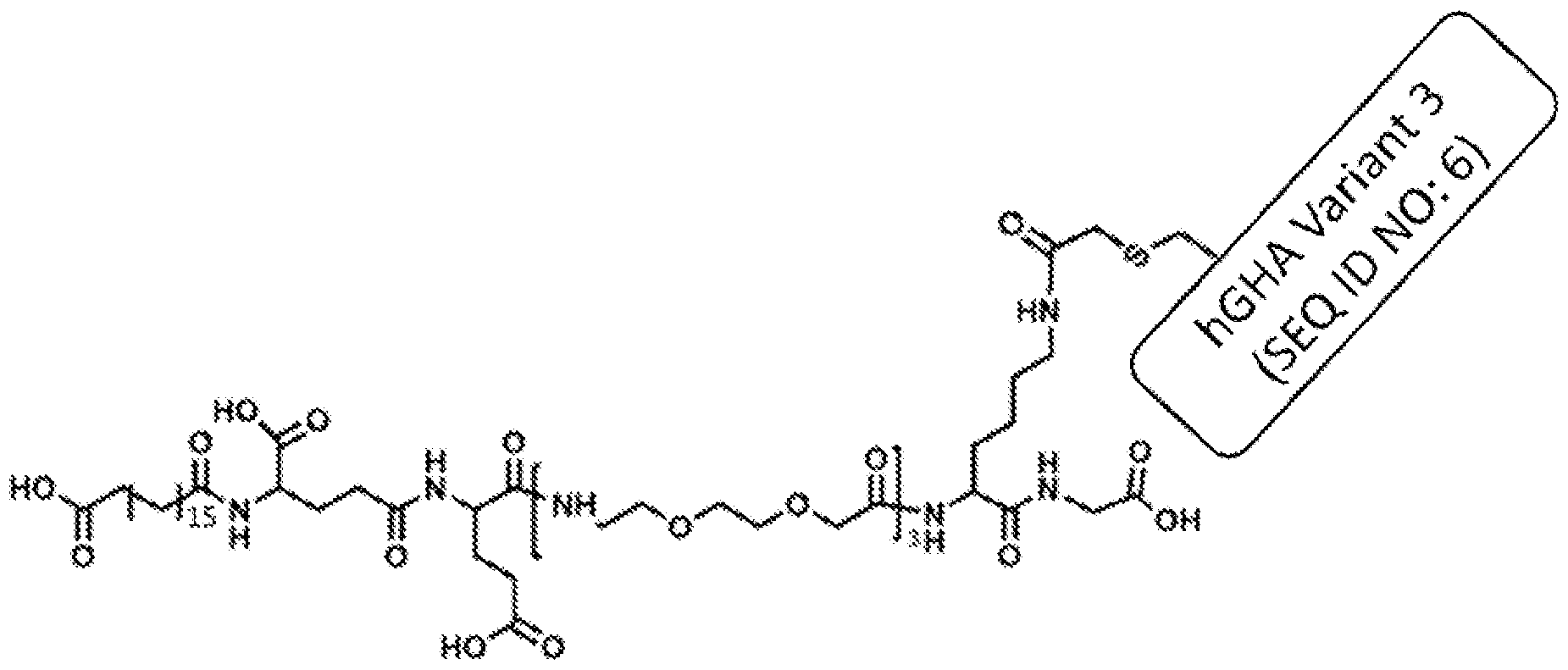
- the acylated hGHA complex is represented in FIG. 2 where n is 15 and X is 3 or hGHA Variant 3 (SEQ ID NO: 6). In another embodiment, the acylated hGHA complex is represented in FIG. 2 where n is 15 and X is 3. In another embodiment, the acylated hGHA complex is represented in FIG. 2 where n is 15 and X is 3 (hGHA Variant 3). In another embodiment, the acylated hGHA complex is represented in FIG. 2 where n is 15 and X is 3 (SEQ ID NO: 6).
- the acylated hGHA complex is represented by the following chemical structure:
- the acylated hGHA complex is represented in FIG. 2 where n is 15 and X is 4 or hGHA Variant 4 (SEQ ID NO: 7). In another embodiment, the acylated hGHA complex is represented in FIG. 2 where n is 15 and X is 4. In another embodiment, the acylated hGHA complex is represented in FIG. 2 where n is 15 and X is 4 (hGHA Variant 4). In another embodiment, the acylated hGHA complex is represented in FIG. 2 where n is 15 and X is 4 (SEQ ID NO: 7).
- the acylated hGHA complex is represented by the following chemical structure:
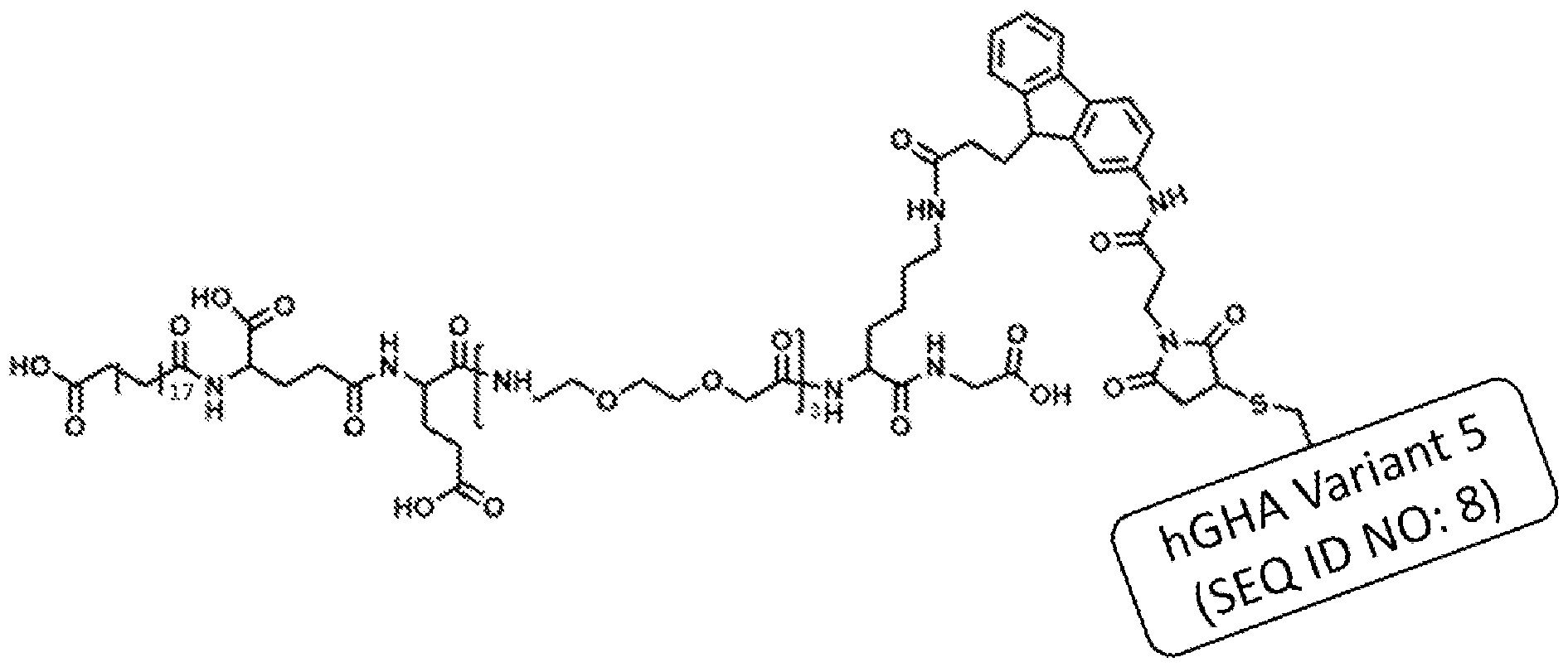
- the acylated hGHA complex is represented in FIG. 2 where n is 15 and X is 5 or hGHA Variant 5 (SEQ ID NO: 8). In another embodiment, the acylated hGHA complex is represented in FIG. 2 where n is 15 and X is 5. In another embodiment, the acylated hGHA complex is represented in FIG. 2 where n is 15 and X is 5 (hGHA Variant 5). In another embodiment, the acylated hGHA complex is represented in FIG. 2 where n is 15 and X is 5 (SEQ ID NO: 8).
- the acylated hGHA complex is represented by the following chemical structure:
- the acylated hGHA complex is represented in FIG. 2 where n is 17 and X is 1 or hGHA Variant 1 (SEQ ID NO: 5). In another embodiment, the acylated hGHA complex is represented in FIG. 2 where n is 17 and X is 1. In another embodiment, the acylated Hgha complex is represented in FIG. 2 where n is 17 and X is 1 (hGHA Variant 1). In another embodiment, the acylated hGHA complex is represented in FIG. 2 where n is 17 and X is 1 (SEQ ID NO: 5).
- the acylated hGHA complex is represented by the following chemical structure:
- the acylated hGHA complex is represented in FIG. 2 where n is 17 and X is 3 or hGHA Variant 3 (SEQ ID NO: 6). In another embodiment, the acylated hGHA complex is represented in FIG. 2 where n is 17 and X is 3. In another embodiment, the acylated hGHA complex is represented in FIG. 2 where n is 17 and X is 3 (hGHA Variant 3). In another embodiment, the acylated hGHA complex is represented in FIG. 2 where n is 17 and X is 3 (SEQ ID NO: 6).
- the acylated hGHA complex is represented by the following chemical structure:
- the acylated hGHA complex is represented in FIG. 2 where n is 17 and X is 4 or hGHA Variant 4 (SEQ ID NO: 7).
- the acylated hGHA complex is represented in FIG. 2 where n is 17 and X is 4.
- the acylated hGHA complex is represented in FIG. 2 where n is 17 and X is 4 (hGHA Variant 4).
- the acylated hGHA complex is represented in FIG. 2 where n is 17 and X is 4 (SEQ ID NO: 7).
- the acylated hGHA complex is represented by the following chemical structure:
- the acylated hGHA complex is represented in FIG. 2 where n is 17 and X is 5 or hGHA Variant 5 (SEQ ID NO: 8). In another embodiment, the acylated hGHA complex is represented in FIG. 2 where n is 17 and X is 5. In another embodiment, the acylated hGHA complex is represented in FIG. 2 where n is 17 and X is 5 (hGHA Variant 5). In another embodiment, the acylated hGHA complex is represented in FIG. 2 where n is 17 and X is 5 (SEQ ID NO: 8).
- the acylated hGHA complex is represented by the following chemical structure:
- the acylated hGHA complex is represented in FIG. 3 where n is 15 and X is 1 or hGHA Variant 1 (SEQ ID NO: 5). In another embodiment, the acylated hGHA complex is represented in FIG. 3 where n is 15 and X is 1. In another embodiment, the acylated hGHA complex is represented in FIG. 3 where n is 15 and X is 1 (hGHA Variant 1). In another embodiment, the acylated hGHA complex is represented in FIG. 3 where n is 15 and X is 1 (SEQ ID NO: 5).
- the acylated hGHA complex is represented by the following chemical structure:
- the acylated hGHA complex is represented in FIG. 3 where n is 15 and X is 3 or hGHA Variant 3 (SEQ ID NO: 6). In another embodiment, the acylated hGHA complex is represented in FIG. 3 where n is 15 and X is 3. In another embodiment, the acylated hGHA complex is represented in FIG. 3 where n is 15 and X is 3 (hGHA Variant 3). In another embodiment, the acylated hGHA complex is represented in FIG. 3 where n is 15 and X is 3 (SEQ ID NO: 6).
- the acylated hGHA complex is represented by the following chemical structure:
- the acylated hGHA complex is represented in FIG. 3 where n is 15 and X is 4 or hGHA Variant 4 (SEQ ID NO: 7). In another embodiment, the acylated hGHA complex is represented in FIG. 3 where n is 15 and X is 4. In another embodiment, the acylated hGHA complex is represented in FIG. 3 where n is 15 and X is 4 (hGHA Variant 4). In another embodiment, the acylated hGHA complex is represented in FIG. 3 where n is 15 and X is 4 (SEQ ID NO: 7).
- the acylated hGHA complex is represented by the following chemical structure:
- the acylated hGHA complex is represented in FIG. 3 where n is 15 and X is 5 or hGHA Variant 5 (SEQ ID NO: 8). In another embodiment, the acylated hGHA complex is represented in FIG. 3 where n is 15 and X is 5. In another embodiment, the acylated hGHA complex is represented in FIG. 3 where n is 15 and X is 5 (hGHA Variant 5). In another embodiment, the acylated hGHA complex is represented in FIG. 3 where n is 15 and X is 5 (SEQ ID NO: 8).
- the acylated hGHA complex is represented by the following chemical structure:
- the acylated hGHA complex is represented in FIG. 3 where n is 17 and X is 1 or hGHA Variant 1 (SEQ ID NO: 5). In another embodiment, the acylated hGHA complex is represented in FIG. 3 where n is 17 and X is 1. In another embodiment, the acylated hGHA complex is represented in FIG. 3 where n is 17 and X is 1 (hGHA Variant 1). In another embodiment, the acylated hGHA complex is represented in FIG. 3 where n is 17 and X is 1 (SEQ ID NO: 5).
- the acylated hGHA complex is represented by the following chemical structure:
- the acylated hGHA complex is represented in FIG. 3 where n is 17 and X is 3 or hGHA Variant 3 (SEQ ID NO: 6). In another embodiment, the acylated hGHA complex is represented in FIG. 3 where n is 17 and X is 3. In another embodiment, the acylated hGHA complex is represented in FIG. 3 where n is 17 and X is 3 (hGHA Variant 3). In another embodiment, the acylated hGHA complex is represented in FIG. 3 where n is 17 and X is 3 (SEQ ID NO: 6).
- the acylated hGHA complex is represented by the following chemical structure:
- the acylated hGHA complex is represented in FIG. 3 where n is 17 and X is 4 or hGHA Variant 4 (SEQ ID NO: 7). In another embodiment, the acylated hGHA complex is represented in FIG. 3 where n is 17 and X is 4. In another embodiment, the acylated hGHA complex is represented in FIG. 3 where n is 17 and X is 4 (hGHA Variant 4). In another embodiment, the acylated hGHA complex is represented in FIG. 3 where n is 17 and X is 4 (SEQ ID NO: 7).
- the acylated hGHA complex is represented by the following chemical structure:
- the acylated hGHA complex is represented in FIG. 3 where n is 17 and X is 5 or hGHA Variant 5 (SEQ ID NO: 8). In another embodiment, the acylated hGHA complex is represented in FIG. 3 where n is 17 and X is 5. In another embodiment, the acylated hGHA complex is represented in FIG. 3 where n is 17 and X is 5 (hGHA Variant 5). In another embodiment, the acylated hGHA complex is represented in FIG. 3 where n is 17 and X is 5 (SEQ ID NO: 8).
- the acylated hGHA complex is represented by the following chemical structure:
- the GHR antagonists in the modified GHR antagonists include a homologue of a growth hormone antagonist
- the GHR antagonists in the modified GHR antagonists includes a homologue of a human growth hormone antagonist
- homologues e.g., polypeptides which are at least 50%, at least 55%, at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 87%, at least 89%, at least 91%, at least 93%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% homologous to a growth hormone or human growth hormone antagonist as determined using BlastP software of the National Center of Biotechnology Information (NCBI) using default parameters.
- NCBI National Center of Biotechnology Information
- the disorders or diseases which can be treated by the method of the present invention include any disease that is exacerbated by the action of an agonist on GHRs can be treated by the present invention.
- the disorder is acromegaly or gigantism.
- the invention includes methods for treating acromegaly and/or giantism by administering to patients having such disorders a therapeutically effective amount of a growth hormone antagonist together with a pharmaceutically acceptable carrier to reduce a pathological effect or symptom of acromegaly and/or giantism and, in particular, to lower levels of IGF-1 in plasma and/or tissue.
- the disease is diabetes mellitus (DM). More specifically, the method of the present invention is used to prevent or reduce proliferative diseases associated with diabetes such as diabetic retinopathy and glomerulosclerosis in patients with DM.
- DM diabetes mellitus
- the method of the present invention is used to prevent or reduce proliferative diseases associated with diabetes such as diabetic retinopathy and glomerulosclerosis in patients with DM
- such methods include administering a growth hormone-antagonist in a therapeutically effective amount to reduce a pathological effect or symptom of diabetes, such as nephropathy or retinopathy, and/or to lower blood glucose levels.
- a method of treating, preventing, or ameliorating an hGH-related disorder comprising: administering to a subject in need thereof an effective amount of an hGH antagonist or modified GHR antagonist disclosed herein or a variant thereof to treat, prevent or ameliorate an hGH -associated disorder in the subject
- the hGH-related disorder is one or more of: acromegaly; age-related macular degeneration; cancer; diabetes mellitus; gigantism, vascular eye diseases including diabetic retinopathy, retinopathy of prematurity, and sickle cell anemia retinopathy; nephropathy, diabetes- induced nephropathy (glomerulosclerosis), neurodegeneration and cancer.
- a method of treating a variety of diseases involving the production of excess hGH comprising the step of administering an effective amount of an hGH antagonist or modified GHR antagonist disclosed herein.
- the methods include the treatment of acromegaly, gigantism, cancer, diabetes, vascular eye diseases (diabetic retinopathy, retinopathy of prematurity, age-related macular degeneration, sickle cell anemia retinopathy, and the tike), as well as nephropathy and neurodegeneration
- a method of inhibiting human growth hormone (hGH) protein action in a subject comprising: administering to a subject an effective amount of an hGH antagonist or modified GHR antagonist disclosed herein, or a variant thereof, to inhibit an hGH protein in the subject
- Diseases that may be treated by the methods of the present application are diseases characterized by one or more of the following criteria: elevated levels of hGH production, elevated levels of serum hGH, elevated levels of hGHR production, and elevated cellular response of hGHRs to hGH
- the term “elevated” as used herein is used with respect to the normal levels of hGH production, hGHR production, or hGH-mediated cellular response in a tissue (or tissues) of a diseased person (or animal) as compared to level in a normal individual.
- Diseases that may be treated by the methods of the invention include, but are not limited to, acromegaly, gigantism, cancer, diabetes, vascular eye diseases (diabetic retinopathy, retinopathy of prematurity, age-related macular degeneration, retinopathy of sickle-cell anemia, etc.) as well as nephropathy.
- method of treating cancers in a subject in another aspect, there is described herein method of treating cancers in a subject by administering an effective amount of an hGH antagonist or modified GHR antagonist disclosed herein.
- the methods include the treatment of cancers comprising tumor cells that express hGHRs.
- sarcoma angiosarcoma, fibrosarcoma, rhabdomyosarcoma, liposarcoma
- myxoma myxoma
- rhabdomyoma myxoma
- fibroma lipoma
- lipoma teratoma
- the methods include the treatment of bronchogenic carcinoma (squamous cell, undifferentiated small cell, undifferentiated large cell, adenocarcinoma), alveolar (bronchiolar) carcinoma, bronchial adenoma, sarcoma, lymphoma, chondromatous hamartoma, and mesothelioma.
- bronchogenic carcinoma squamous cell, undifferentiated small cell, undifferentiated large cell, adenocarcinoma
- alveolar (bronchiolar) carcinoma bronchial adenoma
- sarcoma sarcoma
- lymphoma chondromatous hamartoma
- mesothelioma mesothelioma
- the methods include the treatment of esophagus (squamous cell carcinoma, adenocarcinoma, leiomyosarcoma, lymphoma), stomach (carcinoma, lymphoma, leiomyosarcoma), pancreas (ductal adenocarcinoma, insulinoma, glucagonoma, gastrinoma, carcinoid tumors, vipoma), small bowel (adenocarcinoma, lymphoma, carcinoid tumors, Karposi's sarcoma, leiomyoma, hemangioma, lipoma, neurofibroma, fibroma), large bowel (adenocarcinoma, tubular adenoma, villous adenoma, hamartoma, and leiomyoma.
- esophagus squamous cell carcinoma, adenocarcinoma, leiomyosarcoma, lymphoma
- the methods include the treatment of kidney (adenocarcinoma, Wilm's tumor [nephroblastoma], lymphoma, leukemia), bladder and urethra (squamous cell carcinoma, transitional cell carcinoma, adenocarcinoma), prostate (adenocarcinoma, sarcoma), testis (seminoma, teratoma, embryonal carcinoma, teratocarcinoma, choriocarcinoma, sarcoma, interstitial cell carcinoma, fibroma, fibroadenoma, adenomatoid tumors, and lipoma.
- the methods include the treatment of hepatoma (hepatocellular carcinoma), cholangiocarcinoma, hepatoblastoma, angiosarcoma, hepatocellular adenoma, hemangioma.
- hepatoma hepatocellular carcinoma
- cholangiocarcinoma hepatoblastoma
- angiosarcoma hepatocellular adenoma
- hemangioma hemangioma
- the methods include the treatment of osteogenic sarcoma (osteosarcoma), fibrosarcoma, malignant fibrous histiocytoma, chondrosarcoma, Ewing's sarcoma, malignant lymphoma (reticulum cell sarcoma), multiple myeloma, malignant giant cell tumor, chordoma, osteochronfroma (osteocartilaginous exostoses), benign chondroma, chondroblastoma, chondromyxofibroma, osteoid osteoma and giant cell tumors.
- osteogenic sarcoma osteosarcoma
- fibrosarcoma malignant fibrous histiocytoma
- chondrosarcoma chondrosarcoma
- Ewing's sarcoma malignant lymphoma
- multiple myeloma malignant giant cell tumor
- chordoma chordoma
- osteochronfroma oste
- the methods include the treatment of skull (osteoma, hemangioma, granuloma, xanthoma, osteitis deformans), meninges (meningioma, meningiosarcoma, gliomatosis), brain (astrocytoma, medulloblastoma, glioma, ependymoma, germinoma [pinealoma], glioblastoma multiforme, oligodendroglioma, schwannoma, retinoblastoma, congenital tumors), spinal cord (neurofibroma, meningioma, glioma, sarcoma.
- skull osteoma, hemangioma, granuloma, xanthoma, osteitis deformans
- meninges meningioma, meningiosarcoma, gliomatosis
- brain
- the methods include the treatment of uterus (endometrial carcinoma), cervix (cervical carcinoma, pre-tumor cervical dysplasia), ovaries (ovarian carcinoma [serous cystadenocarcinoma, mucinous cystadenocarcinoma, endometrioid tumors, celioblastoma, clear cell carcinoma, unclassified carcinoma], granulosa-thecal cell tumors, Sertoli-Leydig cell tumors, dysgerminoma, malignant teratoma), vulva (squamous cell carcinoma, intraepithelial carcinoma, adenocarcinoma, fibrosarcoma, melanoma), vagina (clear cell carcinoma, squamous cell carcinoma, botryoid sarcoma [embra),
- the methods include the treatment of uterus (endometrial carcinoma), cervix (cervical carcinoma, pre-tumor cervical dysplasia), ovaries (ovarian carcinoma [serous cystadenocarcinoma, mucinous cystadenocarcinoma, endometrioid tumors, celioblastoma, clear cell carcinoma, unclassified carcinoma], granulosa-thecal cell tumors, Sertoli-Leydig cell tumors, dysgerminoma, malignant teratoma), vulva (squamous cell carcinoma, intraepithelial carcinoma, adenocarcinoma, fibrosarcoma, melanoma), vagina (clear cell carcinoma, squamous cell carcinoma, botryoid sarcoma [embra,
- a method of treating hematologic cancer in a subject by administering an effective amount of an hGH antagonist or modified GHR antagonist disclosed herein include the treatment of blood (myeloid leukemia [acute and chronic], acute lymphoblastic leukemia, chronic lymphocytic leukemia, myeloproliferative diseases, multiple myeloma, myelodysplastic syndrome), Hodgkin's disease, and non-Hodgkin's lymphoma [malignant lymphoma].
- blood myeloid leukemia [acute and chronic], acute lymphoblastic leukemia, chronic lymphocytic leukemia, myeloproliferative diseases, multiple myeloma, myelodysplastic syndrome
- Hodgkin's disease and non-Hodgkin's lymphoma [malignant lymphoma].
- a method of treating skin cancer in a subject by administering an effective amount of an hGH antagonist or modified GHR antagonist disclosed herein.
- the methods include the treatment of malignant melanoma, basal cell carcinoma, squamous cell carcinoma, Karposi's sarcoma, moles, dysplastic nevi, lipoma, angioma, dermatofibroma, keloids, and psoriasis.
- a method of treating cancer of the adrenal glands in a subject by administering an effective amount of an hGH antagonist or modified GHR antagonist disclosed herein.
- the methods include the treatment of neuroblastoma.
- the resin used in the methods of producing an albumin binding complex is a Wang resin.
- the synthesizing of a binder backbone is done using solid-phase peptide synthesis (SPPS).
- SPPS solid-phase peptide synthesis
- the binder backbone is tBu-Fatty acid-gGiu(OtBu)-Giu(OtBu)- (AEEA)i-Lys(Mtt)-Gly-Wang resin, wherein i is 2 or 3 and the Fatty acid is eicosanedioic or octadecandioic.
- step b. of the method of producing the albumin binding complex the removing is done by applying 1%TFA in DCM wash cycles. In a related embodiment, this removing step deprotects the Lys side chain.
- the intermediate composition following step (b) is the tBu-eiocosanedioc- yGlu(C)tBu)-Glu(C)tBu)-(AEEA)3-Lys-Gly-Wang resin, tBu-eiocosanedioc-yGlu(OtBu)-Glu(OtBu)- (AEEA)2-Lys-Gly-Wang resin, tBu-octadecandioic-yGlu(C)tBu)-Glu(C)tBu)-(AEEA)3-Lys-Gly- Wang resin, or tBu-octadecandioic-yGlu(C)tBu)-Glu(C)tBu)-(AEEA)2-Lys-Gly-Wang resin.
- step c. of the method of producing the albumin binding complex the linker moiety and deprotected binder backbone are added to a reactor and stirred.
- the linker moiety is coupled to the primary amine of the deprotected Lys side chain of the peptide backbone.
- the linking moiety in step c. of the method of producing the albumin binding complex, is MAL-NRFmoc. In another embodiment, in step c. of the method of producing the albumin binding complex, the linking moiety is Acetyl-Br.
- step c. of the method of producing the albumin binding complex about 2-3eq of the linking moiety is added to the reactor. In a related embodiment, in step c. of the method of producing the albumin binding complex, about 2-3eq of the MAL-NRFmoc is added to the reactor. [0349] In one embodiment, in step c. of the method of producing the albumin binding complex, 2- 3eq of the linking moiety is added to the reactor. In a related embodiment, in step c. of the method of producing the albumin binding complex, 2-3eq of the MAL-NRFmoc is added to the reactor.
- step c. of the method of producing the albumin binding complex about l-6eq of the linking moiety is added to the reactor. In a related embodiment, in step c. of the method of producing the albumin binding complex, about 1 -6eq of the MAL-NRFmoc is added to the reactor. [0351] In one embodiment, about 20eq of Bromoacetic anhydride is dissolved in DCM, and 20eq of DIPEA is added to the solution resulting in a Bromoacetic anhydride solution ready for Lys side chain conjugation.
- the Bromoacetic anhydride solution is added to the reactor and stirred.
- the stirring in the reactor comprises about 1 hour. In a related embodiment, the stirring in the reactor comprises between about 30 minutes to about 2 hours.
- pH in the reactor is about 5.0. In another embodiment, pH in the reactor is between about 4.0 to 6.0.
- step d. of the method of producing the albumin binding complex the cleaving is done with a cocktail containing 95%TFA: 5% H2O, with a ratio of 10ml cleavage cocktail: lgr peptide-resin.
- step d. of the method of producing the albumin binding complex the cleaving is done with a cocktail containing about 95%TFA: 5% H2O, with a ratio of about 10ml cleavage cocktail: lgr peptide-resin.
- the cleaving is done with a cocktail containing between about 85%TFA: 15% H2O to 99%TFA: 1% H2O.
- the cleaving is done with a cocktail with a ratio of between about 20ml cleavage cocktail: lgr peptide-resin to about lml cleavage cocktail: lgr peptide-resin.
- step e. of the method of producing the albumin binding complex the purifying is done on RP-HPLC column.
- the albumin binding complex is Formula XXXII. In another embodiment, the albumin binding complex is Formula XXXHI.
- the albumin binding complex is tBu-eicosanedioic-yGlu(C)tBu)- Glu(OtBu)-(AEEA)3-Lys(Ac-Br)-Gly-Wang resin, tBu-eicosanedioic-yGlu(OtBu)-Glu(OtBu)- (AEEA)2-Lys(Ac-Br)-Gly-Wang, resin tBu-octadecandioic-yGlu(C)tBu)-Glu(C)tBu)-(AEEA)3- Lys(Ac-Br)-Gly-Wang resin, or tBu-octadecandioic-yGlu(C)tBu)-Glu(C)tBu)-(AEEA)2-Lys(Ac-Br)- Gly-Wang resin.
- the albumin binding complex is tBu-eicosanedioic- yGlu(OtBu)-Glu(OtBu)-(AEEA)3-Lys(MAL-NRFmoc)-Gly-Wang resin, tBu-eicosanedioic- yGlu(OtBu)-Glu(OtBu)-(AEEA) 2 -Lys(MAL-NRFmoc)-Gly-Wang resin, tBu-octadecandioic- ydu(C)tBu)-Glu(OtBu)-(AEEA)3-Lys(MAL-NRFmoc)-Gly-Wang resin, or tBu-octadecandioic- yGlu(C)tBu)-Glu(OtBu)-(AEEA)2-Lys(MAL-NRFmoc)-Gly-Wang resin.
- albumin binding complexes including Formula xxxn andXXXm.
- a method of conjugating an albumin binding complex to an hGH antagonist by adding the binding complex to the hGHA in solution is added.
- the binder complex is added at about 1.25 eq.
- the binder complex is added at between .75 eq and 1.75 eq.
- the hGHA is added at about 1 eq.
- the hGHA is added at about .25 eq to about 1.75 eq.
- the solution conjugating an albumin binding complex is kept at room temperature.
- the solution is quenched.
- the quenching solution is lOOmM L-Cys at a pH of 7.
- the quenching solution is about lOOmM L-Cys.
- the quenching solution is between 75mM and 125 mM L-Cys.
- the quenching solution is at a pH of about 7.
- the quenching solution is at a pH of between 6 and 8.
- 1 volume unit of the quenching solution is added to 100 volume units of the reaction solution
- GHR antagonist 9 comprising growth hormone receptor antagonist G120K (SEQ ID NO: 2) and at least one of the following amino acid mutations: H18D, H21N, F54P, R64K, R167N, D171S, E174S, I179T.
- the GHR antagonist of claim 3, wherein the amino acid mutated to cysteine is T3, E39, P48, Q69, N99, L129, T135, T142, H151 or combinations thereof.
- GHR antagonist of claim 1 wherein the GHR antagonist has the following amino acid mutations: G120K, L129C, R167N, D171S, E174S, andI179T.
- GHR antagonist of claim 6, wherein the GHR antagonist comprises the amino acid sequence of SEQ ID NO: 5.
- GHR antagonist of claim 1 wherein the GHR antagonist has the following amino acid mutations: H18D, H21N, F54P, R64K, G120K, L129C, R167N, D171S, E174S, and I179T.
- GHR antagonist of claim 8 wherein the GHR antagonist comprises the amino acid sequence of SEQ ID NO: 6.
- GHR antagonist of claim 1 wherein the GHR antagonist has the following amino acid mutations: H18D, H21N, G120K, L129C, R167N, D171S, E174S, and I179T.
- GHR antagonist of claim 10 wherein the GHR antagonist comprises the amino acid sequence of SEQ ID NO: 7.
- the GHR antagonist of claim 12 wherein the GHR antagonist comprises the amino acid sequence of SEQ ID NO: 8.
- modified GHR antagonist or “modified hGHA” having the structure of formula (I):
- W represents a binder
- X represents a spacer
- Y represents an optional linker
- Z represents a human growth hormone receptor antagonist (“GHR antagonist”); or a pharmaceutically acceptable salt thereof.
- O N or is represented by Formula X-C: o o
- modified GHR antagonist of claim 15 wherein the modified GHR antagonist comprises the following formula:
- modified GHR antagonist of claim 15 wherein the modified GHR antagonist comprises the following formula: n
- modified GHR antagonist of claim 15 wherein the modified GHR antagonist comprises the following formula:
- modified GHR antagonist of claim 15 wherein the modified GHR antagonist comprises the following formula:
- modified GHR antagonist of claim 15 wherein the modified GHR antagonist comprises the following formula: o, *7 V
- modified GHR antagonist of claim 15 wherein the modified GHR antagonist comprises the following formula: hG (SEQ ID NO:
- modified GHR antagonist of claim 15 wherein the modified GHR antagonist comprises the following formula: o. toll o
- modified GHR antagonist of claim 15, wherein the modified GHR antagonist comprises the following formula:
- modified GHR antagonist of claim 15, wherein the modified GHR antagonist comprises the following formula:
- modified GHR antagonist of claim 15 wherein the modified GHR antagonist comprises the following formula:
- modified GHR antagonist of claim 15, wherein the modified GHR antagonist comprises the following formula:
- modified GHR antagonist of claim 15 wherein the modified GHR antagonist comprises the following formula:
- modified GHR antagonist of claim 15 wherein the modified GHR antagonist comprises the following formula:
- modified GHR antagonist of claim 15, wherein the modified GHR antagonist comprises the following formula:
- modified GHR antagonist of claim 15, wherein the modified GHR antagonist comprises the following formula:
- a method of inhibiting human growth hormone (hGH) action in a subject comprising: administering to a subject in need thereof an effective amount of the GHR antagonist of any one of claims 1 to 14 or the modified GHR antagonist of any one of claims 15 to 47, to inhibit hGH action in the subject.
- hGH human growth hormone
- the hGH related disorder is one or more of: acromegaly; age-related macular degeneration; cancer; diabetes; gigantism, vascular eye diseases, diabetic retinopathy, retinopathy of prematurity, and retinopathy of sickle-cell anemia; and, nephropathy.
- a method of treating, preventing, or ameliorating a hGH related disorder comprising: administering to a subject in need thereof an effective amount of the GHR antagonist of any one of claims 1 to 14 or the modified GHR antagonist of any one of claims 15 to 47, to treat, prevent, or ameliorate a hGH related disorder in the subject.
- a method of producing an albumin binding complex on a resin comprising:
- binder backbone is tBu-Fatty acid- gGhi(OtBu)-Ghi(OtBu)-(AEEA)i-Lys(Mtt)-Gly-Wang resin, wherein i is 2 or 3 and the Fatty acid is eicosanedioic or octadecandioic.
- step (b) is done by applying 1%TFA in DCM wash cycles.
- step (d) is done with a cocktail containing 95%TFA: 5% H2O, with a ratio of 10ml cleavage cocktail: lgr peptide-resin.
- step (e) is done on RP- HPLC column.
- step (c) is MAL- NRFmoc.
- albumin binding complex is tBu-eicosanedioic-yGlu(OtBu)-Glu(OtBu)-(AEEA)3-Lys(Ac-Br)-Gly-Wang resin, tBu- eicosanedioic-yGlu(OtBu)-Glu(OtBu)-(AEEA)2-Lys(Ac-Br)-Gly-Wang, resin tBu- octadecandioic-yGlu(OtBu)-Glu(OtBu)-(AEEA)3-Lys(Ac-Br)-Gly-Wang resin, or tBu- octadecandioic-yGlu(OtBu)-Glu(OtBu)-(AEEA)2-Lys(Ac-Br)-Gly-Wang resin.
- An albumin binding complex comprising tBu-eicosanedioic-yGlu(OtBu)-Glu(OtBu)- (AEEA)3-Lys(Ac-Br)-Gly-Wang resin, tBu-eicosanedioic-yGlu(OtBu)-Glu(OtBu)- (AEEA)2-Lys(Ac-Br)-Gly-Wang resin, tBu-octadecandioic-yGlu(OtBu)-Glu(OtBu)- (AEEA)3-Lys(Ac-Br)-Gly-Wang resin, or tBu-octadecandioic-yGlu(OtBu)-Glu(OtBu)- (AEEA)2-Lys(Ac-Br)-Gly-Wang resin.
- An albumin binding complex comprising tBu-eicosanedioic-yGlu(OtBu)-Glu(OtBu)- (AEEA)s-Lys(MAL-NRFmoc)-Gly-Wang resin, tBu-eicosanedioic-yGlu(OtBu)- Glu(OtBu)-(AEEA)2-Lys(MAL-NRFmoc)-Gly-Wang resin, tBu-octadecandioic- yGlu(OtBu)-Glu(OtBu)-(AEEA)3-Lys(MAL-NRFmoc)-Gly-Wang resin, or tBu- octadecandioic-yGlu(OtBu)-Glu(OtBu)-(AEEA)2-Lys(MAL-NRFmoc)-Gly-Wang resin.
- a “non-naturally encoded amino add” refers to an amino acid that is not one of the common amino acids or pyrrolysine or selenocysteine.
- Other terms that may be used synonymously with the term “non-naturally encoded amino add” are “non-natural amino acid,” “unnatural amino acid,” “non-naturally-occurring amino add,” and variously hyphenated and non-hyphenated versions thereof.
- the term “non-naturally encoded amino acid” also includes, but is not limited to, amino acids that occur by modification (e.g.
- a naturally encoded amino acid including but not limited to, the 20 common amino acids or pyrrolysine and selenocystdne
- non-naturally-occurring amino adds include, but are not limited to, N- acetylglucosaminyl-L-serine, N-acetylglucosaminyl-L-threonine, and O-phosphotyrosine.
- modified refers to any changes made to a given polypeptide, such as changes to the length of the polypeptide, the amino acid sequence, chemical structure, co- translational modification, or post-translational modification of a polypeptide.
- the form “(modified)” term means that the polypeptides being discussed are optionally modified, that is, the polypeptides under discussion can be modified or unmodified.
- the term “about”, may encompass a deviance of between 0.0001-5% from the indicated number or range of numbers. In some instances, the term “about”, may encompass a deviance of between 1 -10% from the indicated number or range of numbers. In some instances, the term “about”, encompasses a deviance of up to 25% from the indicated number or range of numbers.
- an “excipient” is a pharmaceutically acceptable substance formulated along with the active ingredients) of a medication, pharmaceutical composition, formulation, or drug delivery system. Excipients may be used, for example, to stabilize the composition, to bulk up the composition (thus often referred to as “bulking agents,” “fillers,” or “diluents” when used for this purpose), or to confer a therapeutic enhancement on the active ingredient in the final dosage form, such as facilitating drug absorption, reducing viscosity, or enhancing solubility. Excipients include pharmaceutically acceptable versions of antiadherents, binders, coatings, colors, disintegrants, flavors, glidants, lubricants, preservatives, sorbents, sweeteners, and vehicles.
- the main excipient that serves as a medium for conveying the active ingredient is usually called the vehicle.
- Excipients may also be used in the manufacturing process, for example, to aid in the handling of the active substance, such as by fadlitating powder flowability or non-stick properties, in addition to aiding in vitro stability such as prevention of denaturation or aggregation over the expected shelf life.
- the suitability of an excipient will typically vary depending on the route of administration, the dosage form, the active ingredient, as well as other factors.
- patienf refers to a living mammalian organism, such as a human, monkey, cow, horse, sheep, goat, dog, cat, mouse, rat, guinea pig, or transgenic species thereof.
- the patient or subject is a primate.
- Non-limiting examples of human patients are adults, juveniles, infants and fetuses.
- pharmaceutically acceptable refers to those compounds, materials, compositions, and/or dosage forms which are, within the scope of sound medical judgment, suitable for use in contact with the tissues, organs, and/or bodily fluids of human beings and animals without excessive toxicity, irritation, allergic response, or other problems or complications commensurate with a reasonable benefit/risk ratio.
- “Pharmaceutically acceptable salts” means salts of compounds of the present invention which are pharmaceutically acceptable, as defined above, and which possess the desired pharmacological activity.
- Non-limiting examples of such salts include add addition salts formed with inorganic acids such as hydrochloric acid, hydrobromic add, sulfuric acid, nitric add, and phosphoric add; or with organic adds such as 1,2-ethanedisulfonic acid, 2 -hydroxyethanesulfonic acid, 2-naphthalenesulfonic acid, 3-phenylpropionic acid, 4, 4'-methylenebis(3-hydroxy-2-ene-l -carboxylic acid), 4- methylbicyclo [2.2.2]oct-2-ene-l -carboxylic acid, acetic acid, aliphatic mono- and dicarboxylic adds, aliphatic sulfuric adds, aromatic sulfuric adds, benzenesulfonic acid, benzoic acid, camphorsulfonic acid,
- Pharmaceutically acceptable salts also include base addition salts which may be formed when addic protons present are capable of reacting with inorganic or organic bases.
- Acceptable inorganic bases include sodium hydroxide, sodium carbonate, potassium hydroxide, aluminum hydroxide and calcium hydroxide.
- Non-limiting examples of acceptable organic bases include ethanolamine, diethanolamine, triethanolamine, tromethamine, and N-methylglucamine. It should be recognized that the particular anion or cation forming a part of any salt of this invention is not critical, so long as the salt, as a whole, is pharmacologically acceptable. Additional examples of pharmaceutically acceptable salts and their methods of preparation and use are presented in Handbook of Pharmaceutical Salts: Properties, and Use (P. H Stahl & C. G. Wermuth eds., Verlag Helvetica Chimica Acta, 2002).
- a “pharmaceutically acceptable carrier,” “drug carrier,” or simply “carrier” is a pharmaceutically acceptable substance formulated along with the active ingredient medication that is involved in carrying, delivering and/or transporting a chemical agent
- Drug carriers may be used to improve the delivery and the effectiveness of drugs, including for example, controlled-release technology to modulate drug bioavailability, decrease drug metabolism, and/or reduce drug toxicity. Some drug carriers may increase the effectiveness of drug delivery to the specific target sites.
- carriers examples include: liposomes, microspheres (e.g., made of poly(lactic-co-glycolic) add), albumin microspheres, synthetic polymers, nanofibers, protein-DNA complexes, protein conjugates, erythrocytes, virosomes, and dendrimers.
- a “pharmaceutical drug” (also referred to as a pharmaceutical, pharmaceutical agent, pharmaceutical preparation, pharmaceutical composition, pharmaceutical formulation, pharmaceutical product, medicinal product, medicine, medication, medicament, or simply a drug) is a drug used to diagnose, cure, treat, or prevent disease.
- An active ingredient (Al) (defined above) is the ingredient in a pharmaceutical drug or a pesticide that is biologically adive.
- active pharmaceutical ingredient (API) and bulk adive are also used in medicine, and the term active substance may be used for pesticide formulations.
- Some medications and pesticide products may contain more than one active ingredient
- the inactive ingredients are usually called excipients (defined above) in pharmaceutical contexts.
- a “stereoisomer” or “optical isomer” is an isomer of a given compound in which the same atoms are bonded to the same other atoms, but where the configuration of those atoms in three dimensions differs.
- “Enantiomers” are stereoisomers of a given compound that are mirror images of each other, like left and right hands.
- “Diastereomers” are stereoisomers of a given compound that are not enantiomers.
- Chiral molecules contain a chiral center, also referred to as a stereocenter or stereogenic center, which is any point, though not necessarily an atom, in a molecule bearing groups such that an interchanging of any two groups leads to a stereoisomer.
- the chiral center is typically a carbon, phosphorus or sulfur atom, though it is also possible for other atoms to be stereocenters in organic and inorganic compounds.
- a molecule can have multiple stereocenters, giving it many stereoisomers.
- the total number of hypothetically possible stereoisomers will not exceed 2n, where n is the number of tetrahedral stereocenters.
- Molecules with symmetry frequently have fewer than the maximinn possible number of stereoisomers.
- a 50:50 mixture of enantiomers is referred to as a racemic mixture.
- a mixture of enantiomers can be enantiomerically enriched so that one enantiomer is present in an amount greater than 50%.
- enantiomers and/or diastereomers can be resolved or separated using techniques known in the art It is contemplated that that for any stereocenter or axis of chirality for which stereochemistry has not been defined, that stereocenter or axis of chirality can be present in its R form, S form, or as a mixture of the R and S forms, including racemic and non-racemic mixtures.
- the phrase “substantially free from other stereoisomers” means that the composition contains ⁇ 15%, more preferably ⁇ 10%, even more preferably ⁇ 5%, or most preferably ⁇ 1% of another stereoisomers).
- prevention includes: (1) inhibiting the onset of a disease in a subject or patient which may be at risk and/or predisposed to the disease but does not yet experience or display any or all of the pathology or symptomatology of the disease, and/or (2) slowing the onset of the pathology or symptomatology of a disease in a subject or patient which may be at risk and/or predisposed to the disease but does not yet experience or display any or all of the pathology or symptomatology of the disease.
- Treatment’ ’ or “treating” includes (1) inhibiting a disease in a subject or patient experiencing or displaying the pathology or symptomatology of the disease (e.g., arresting further development of the pathology and/or symptomatology), (2) ameliorating a disease in a subject or patient that is experiencing or displaying the pathology or symptomatology of the disease (e.g., reversing the pathology and/or symptomatology), and/or (3) effecting any measurable decrease in a disease in a subject or patient that is experiencing or displaying the pathology or symptomatology of the disease.
- inhibiting a disease in a subject or patient experiencing or displaying the pathology or symptomatology of the disease e.g., arresting further development of the pathology and/or symptomatology
- ameliorating a disease in a subject or patient that is experiencing or displaying the pathology or symptomatology of the disease e.g., reversing the pathology and/or symptomatology
- the goal of the present experiment is to design an albumin binding moiety of an hGH antagonist This is designed to increase the longevity of the hGH antagonist
- Three principles were prioritized when designing a long-acting prodrug employing an albumin-binding probe.
- the first principle is that following its conjugation, the probe introduced into the drug should have sufficient affinity to albumin to manifest prolonged action in vivo.
- the conjugate’ s activity with respect to binding and properly activating the drug target receptor should be assessed.
- probe conjugation There are two approaches for probe conjugation that can be applied: (I) Covalent binding to the Albumin probe results in an inactive product; The latter should undergo slow reactivation at physiological conditions by applying reversible Fmoc linkers resulting in probe hydrolysis and release of the drug; (II) Conjugate retains its activity (to appropriate extend); the probe is not interfering with the drug activity, and its hydrolysis is not mandatory.
- Binder Spacer (optional) Antagonist linker.
- acylated hGH antagonists discussed in this example are also referred to as MOD -1202, long-acting human growth hormone (hGH) antagonists, modified hGH antagonists or modified human growth hormone receptor (GHR) antagonists.
- the designed acylated GHR antagonists differ from each other in the following levels: protein (different mutations); linker (reversible, irreversible, or not present); spacer (differ in length); binders (different in fatty acid length).
- a human growth hormone antagonist was selected as the target protein. In order to enhance the potential albumin binding characteristics of the hGH antagonist, several point mutations were used. A single mutation of G120K compared to native hGH was selected to convert hGH to an hGH antagonist
- the mutation of either L129C or T135C was selected to introduce a cysteine as a conjugation site for the binder complex.
- the choice of these specific mutations was based on the hGH Receptor crystal structure (PDB code: 3HHR), and aimed to find an optional area for linker binding without interrupting the protein-receptor interaction, and while considering the amino acid conservation score. Outward-feeing amino acids were prioritized.
- the N-terminus and C-terminus hGH protein were evaluated as well for potential albumin linker binding sites, but in both cases, it seems that a large moiety such as albumin may cause structural interruptions to hGH antagonist protein and/or disrupt binding to the hGHR
- Variant 1 hGHL129C; G120K; R167N; D171S; E174S; I179T (SEQID NO: 5).
- TheL129C mutation introduced a cysteine as a conjugation site.
- the G120K mutation imparts antagonistic properties. 4 point mutations improve binding.
- Variant 3 hGH L129C; H18D; H21N; F54P; R64K; G120K; R167N; D171S; E174S; I179T (SEQ ID NO: 6).
- the L129C mutation introduced a cysteine as a conjugation site.
- the G120K mutation imparts antagonistic properties. 8 point mutations improve binding.
- Variant 4 hGHL129C; H18D; H21N; G120K; R167N; D171S; E174S; I179T (SEQ ID NO:
- the L129C mutation introduced a cysteine as a conjugation site.
- the G120K mutation imparts antagonistic properties. 6 point mutations improve binding.
- Variant 5 hGH T135C; H18D; H21N; G120K; R167N; D171S; E174S; I179T (SEQ ID NO:
- the T135C mutation introduced a cysteine as a conjugation site.
- the G120K mutation imparts antagonistic properties. 6 point mutations improve binding.
- the modified GHR antagonists should minimally contain a single Cys or equivalent nucleophilic amino acid side chain reactant located at an odd numbered amino add position on the polypeptide to serve as an albumin-binder conjugation site.
- the modified GHR antagonists have a Cys mutation located at either one of the following positions: 129 or 135. In these preferred positions, the Cys sulfur nucleophile is accessible to read with or couple to an albumin binding moiety. According to the structural analysis of the protein obtained via crystallography methodologies (data not shown), the proposed positions for albumin-binders coupling were the least probable to interfere with receptor binding.
- the binder part fatty acid drains of various lengths were considered, including short, medium, and long fatty add chains.
- C2 to C30 fatty adds were considered.
- the binder can be any of a C2 fatty acid, C4 fatty acid, C6 fatty add, C8 fatty acid, Cl 0 fatty acid, C12 fatty acid, C14 fatty add, C16 fatty acid, C18 fatty acid, C20 fatty add, C22 fatty acid, C24 fatty acid, C26 fatty acid, C28 fatty acid, or C30 fatty add.
- Three sizes of fatty add chains were recommended as candidates, C16, C18 and C20, as they were found to be the best analogues in different targets.
- reversible coupling also described as reversible acylation: the carbamate is hydrolyzed in the presence of water-based media at neutral-slight basic pHs (see formula XI & XII below).
- permanent coupling also described as permanent acylation: either MAL group or Br as a leaving group can serve for covalent reaction with free thiols (Michael addition, and Sn2 reactions, respectively).
- Formulas XHI and XIV exemplify the thioether bonds formed following the stable coupling using the MAL and Ac-Br functional groups.
- the site of attachment to the hGH antagonist is designed to be either mutated amino acids T135C or E129C. Based on the overall design principles discussed above, a modulated Albumin-binder/spacer complex was developed in order to obtain both Albumin binding and drug potency.
- the full modulated Albumin binder/spacer/linker matrix is detailed in Tables 2-6. Table 2: Full Matrix of hGH antagonist Binder-linker Complexes Designs Binding Type _ Binder Spacer Linker
- N/A (C20 diacid) Gly n l, 2, or 3
- Octadecanedioic Variant 1 (SEQ 1A gGlu-Glu-(AEEA)3-Lys Mal-NRFmoc-NHS acid (Cl 8 diacid) ID NO: 5) (or “1202-1 A) Octadecanedioic Variant 3 (SEQ 3A gGlu-Glu-(AEEA)3-Lys Mal-NRFmoc-NHS acid (Cl 8 diacid) ID NO: 6) (or 1202-3A) Octadecanedioic Variant 4 (SEQ 4A acid (Cl 8 diacid) ID NO: 7) (or 1202 -4 A) Octadecanedioic Variant 5 (SEQ 5A gGlu-Glu-(AEEA)3-Lys Mal-NRFmoc-NHS acid (Cl 8 diacid) ID NO: 8) (or 1202-5 A)
- Binding Type for all Complexes above is Permanent at Cys Position 129 or 135 of the hGHA Variant
- Eicosanedioic acid Variant 1 SEQ IB gGlu-Glu-(AEEA)3-Lys Mal-NRFmoc-NHS (C20 diacid) ID NO: 5) (or 1202-1B)
- Eicosanedioic acid Variant 3 (SEQ 3B gGlu-Glu-(AEEA)3-Lys Mal-NRFmoc-NHS (C20 diacid) ID NO: 6) (or 1202-3B)
- Eicosanedioic acid Variant 4 (SEQ 4B (C20 diacid) ID NO: 7) (or 1202-4B)
- Eicosanedioic acid Variant 5 (SEQ 5B gGlu-Glu-(AEEA)3-Lys Mal-NRFmoc-NHS (C20 diacid) ID NO: 8) (or 1202-5B) Binding Type for all Complexes above is Permanent at Cys Position 129 or 135 of the hGHA Variant
- Binding Type for all Complexes above is Permanent at Cys Position 129 or 135 of the hGHA Variant
- reversible albumin binding constructs having the designated binders, spacers and linkers or reactants thereof are shown in Table 7.
- the acylated hGHA complex 1A (also “1202-1 A”) is represented in FIG. 2 where n is 15 and X is 1 or hGHA Variant 1 (SEQ ID NO: 5).
- the acylated hGHA complex 1A (also “1202-1A”) is also represented by the following chemical structure:
- the acylated hGHA complex 3A (also “1202-3 A”) is represented in FIG. 2 where n is 15 and X is 3 or hGHA Variant 3 (SEQ ID NO: 6).
- the acylated hGHA complex 3A (also “1202-3A”) is also represented by the following chemical structure:
- the acylated hGHA complex 4A (also “1202-4A”) is represented in FIG. 2 where n is 15 and X is 4 or hGHA Variant 4 (SEQ ID NO: 7).
- the acylated hGHA complex 4A (also “1202-4A”) is also represented by the following chemical structure:
- the acylated hGHA complex 5A (also “1202-5 A”) is represented in FIG. 2 where n is 15 and X is 5 or hGHA Variant 5 (SEQ ID NO: 8).
- the acylated hGHA complex 5 A (also “1202-5 A”) is also represented by the following chemical structure:
- the acylated hGHA complex IB (also “1202-1B”) is represented in FIG. 2 where n is 17 and X is 1 or hGHA Variant 1 (SEQ ID NO: 5).
- the acylated hGHA complex IB (also “1202-1B”) is also represented by the following chemical structure:
- the acylated hGHA complex 3B (also “1202-3B”) is represented in FIG. 2 where n is 17 and X is 3 or hGHA Variant 3 (SEQ ID NO: 6).
- the acylated hGHA complex 3B (also “1202-3B”) is also represented by the following chemical structure:
- the acylated hGHA complex 4B (also “1202-4B”) is represented in FIG. 2 where n is 17 and X is 4 or hGHA Variant 4 (SEQ ID NO: 7).
- the acylated hGHA complex 4B (also “1202 ⁇ 8”) is also represented by the following chemical structure:
- the acylated hGHA complex 5B (also “1202-5B”) is represented in FIG. 2 where n is 17 and X is 5 or hGHA Variant 5 (SEQ ID NO: 8).
- the acylated hGHA complex 5B (also “1202-5B”) is also represented by the following chemical structure:
- the acylated hGHA complex 1C (also “1202-1 C”) is represented in FIG. 3 where n is 15 and X is 1 or hGHA Variant 1 (SEQ ID NO: 5).
- the acylated hGHA complex 1C (also “1202-1 C”) is also represented by the following chemical structure:
- the acylated hGHA complex 3C (also “1202-3C”) is represented in FIG. 3 where n is 15 and X is 3 or hGHA Variant 3 (SEQ ID NO: 6).
- the acylated hGHA complex 3C (also “1202-3C”) is also represented by the following chemical structure:
- the acylated hGHA complex 4C (also “1202-4C”) is represented in FIG. 3 where n is 15 and X is 4 or hGHA Variant 4 (SEQ ID NO: 7).
- the acylated hGHA complex 4C (also “1202-4C”) is also represented by the following chemical structure:
- the acylated hGHA complex 5C (also “1202-5C”) is represented in FIG. 3 where n is 15 and X is 5 or hGHA Variant 5 (SEQ ID NO: 8).
- the acylated hGHA complex 5C (also “1202-5C”) is also represented by the following chemical structure:
- the acylated hGHA complex ID (also “1202-1D”) is represented in FIG. 3 where n is 17 and X is 1 or hGHA Variant 1 (SEQ ID NO: 5).
- the acylated hGHA complex ID (also “1202-1D”) is also represented by the following chemical structure:
- the acylated hGHA complex 3D (also “1202-3D”) is represented in FIG. 3 where n is 17 and X is 3 or hGHA Variant 3 (SEQ ID NO: 6).
- the acylated hGHA complex 3D (also “1202-3D”) is also represented by the following chemical structure:
- the acylated hGHA complex 4D (also “1202-4D”) is represented in FIG. 3 where n is 17 and X is 4 or hGHA Variant 4 (SEQ ID NO: 7).
- the acylated hGHA complex 4D (also “1202 ⁇ 0”) is also represented by the following chemical structure:
- the acylated hGHA complex 5D (also “1202-5D”) is represented in FIG. 3 where n is 17 and X is 5 or hGHA Variant 5 (SEQ ID NO: 8).
- the acylated hGHA complex 5D (also “1202-5D”) is also represented by the following chemical structure:
- the Albumin binding complex is a three-components entity composed of (1) Fatty acid, (2) amino acid sequence (gGlu, Glu, AETA, Lys, and (Sy; For structure, please see Table 8), and (3) linking moiety enabling its coupling to the hGH antagonist polypeptide.
- the three-component binder complex was synthesized on resin, using a standard SPPS techniques.
- a TFA-containing cleavage cocktail was used for binder cleavage from the resin: 95% TFA, 5% HzO; ratio of 10ml cleavage cocktail to lgr resin. Cleavage was performed for 2hr at RT, followed by evaporation of -80% of the volume.
- An organic antisolvent was used for the crude binder precipitation: Diisopropyl ether, ratio of 10ml antisolvent to lml of evaporated cleavage cocktail.
- Binder purification was enabled via preparative HPLC purification cycles, and purified material was finally lyophilized in order to obtain a stable powder containing a TFA-salt purified Albumin binder complex
- a linking moiety is coupled to the primary amine via carbodiimide-initiated condensation reaction
- a general structure of the two types of the Albumin binding complexes, differ by their linking moiety are described as Albumin binder complex with either Acetyl-Br or MAL-NRFmoc as a linking moiety enabling the coupling to Hgh antagonist polypeptide:
- the binder backbone was synthesized with a CEM automatic synthesizer.
- FIG. 17 represents a coupling step of the fatty acid Eiocosanedioc to the Fmoc-ydu(OtBu)-du(C)tBu)-(AEEA)3-Lys(mtt)-Gly-Wang resin (peptide on resin).
- the fatty add-conjugated binder complex was eluted from the C- 18 RP-HPLC column using higher percentage of organic solvent (in this case ACN used in the mobile phase) compared to the non-conjugated peptide.
- FIG. 17 shows RP-HPCD chromatograms.
- the top chromatogram shows the binder backbone before fatty add coupling (TFmoc), and the bottom chromatogram shows binder 1 backbone after fatty add coupling (8AA).
- the Lys side chain was deprotected.
- the Mtt protecting group was removed from the Lys side chain by applying 1%TFA in DCM wash cycles. The washing cycles continued until the entire Mtt was removed. Reaction monitoring was performed by addition of TFA to a few beads. If the beads turn orange/yellow immediately, an additional cycle is required. If the beads do not change color, the cleavage has ended, and no further cycle is necessary.
- Step 3(A) outlines the Lys side chain conjugation method for the Mal- NRFmoc-NHS linker.
- Step 3(B) outlines the Lys side chain conjugation method for the Bromoacetic anhydride linker.
- MAL-NRFmoc-NHS linker was reconstituted in DMF to obtain a lOOmg/ml solution (85% of the total calculated volume was used). The concentration was determined by O.D. measurements at 280nm after dilution x500 & xlOOO in (20% 0.1%TFA/HaO:80% ACM). (EC: 21200 ml/(mmol*cm), MW: 501 mg/mmol). The concentration of the linker was adjusted with DMF according to the results obtained. DCM was added to the linker solution to obtain a 50mg/ml linker stock solution pH of the solution was verified to be around pH of 5.
- FIG. 18 represents a conjugation step of MAL-NRFmoc-NHS to the tBu-Eiocosanedioc- ydu(C)tBu)-Glu(C)tBu)-(AEEA)3-Lys-Gly-Wang resin.
- the MAL-NRFmoc-conjugated binder complex is eluted from the C-18 RP-HPLC column using higher percentage of organic solvent (in this case ACN used in the mobile phase) compared to the non-conjugated binding complex.
- FIG. 18 shows RP-HPCD chromatograms.
- the top chromatogram shows the binder before MAL-NRFmoc linker conjugation, and the bottom chromatogram shows the binder after linker conjugation.
- the Ac-Br -conjugated binder complex is eluted (as expected) from the C-18 RP-HPLC column using higher percentage of organic solvent (in this case ACN used in the mobile phase) compared to the non-conjugated binding complex.
- FIG. 19 shows RP-HPCD chromatograms that represent the step of conjugating Ac-Br to the tBu- Eiocosanedioc-yGlu(C)tBu)-Glu(C)tBu)-(AEEA)3-Lys-Gly-Wang resin.
- the top chromatogram shows the binder before Ac-Br linker conjugation, and the bottom chromatogram shows binder after linker conjugation
- Cleavage was performed with a cocktail containing 95%TFA: 5% H2O, with a ratio of 10ml cleavage cocktail: lgr peptide-resin. After 2hr of cleavage, about 80% of the cleavage cocktail was evaporated (using N2), and then remaining solution was precipitated in cold IPE (-20°C), with a ratio of 10ml IPE: 1 ml of evaporated cleavage cocktail. The binder precipitants were kept at -20°C overnight to achieve full precipitation After a full precipitation, the binders were washed with IPE 3 more times and dried under N2, and the pellet was weighed. Assuming about 50% residual solvents in the pellet, the pellet was reconstituted in DMF to obtain a concentration of ⁇ 100mg/ml of stable binder prior to purification and was kept at -80°C until purification started.
- FIG. 20 shows RP-HPCD chromatograms of purified Eicosanedioic-yGlu-Glu-(AEEA)3- Lys(Ac-Br)-Gly-OH in the top chromatogram, and Eicosanedioic-yGlu-Glu-(AEEA)3-Lys(MAL- NRFmoc)-Gly-OH in the bottom chromatogram.
- FIG. 21 shows mass spectrum of Eicosanedioic-yGlu-Glu-(AEEA)3-Lys(Ac-Br)-Gly-OH
- FIG. 22 shows mass spectrum of Eicosanedioic-yGlu-Glu-(AEEA)3-Lys(MAL-NRFmoc)- Gly-OH
- Binder Conjugation to a hGH antagonist polypeptides [0475] The binders presented in this Example were dissolved in a water containing buffer systems (e.g. Na-Pi) and were further conjugated to the hGH antagonist polypeptides thought their unpaired Cys side chains (thiol groups).
- the conjugation step was performed between the MAL group of the Albumin binder complex, and thiol group located on the Cys side chain of the hGH antagonist (See Scheme 1).
- the reaction was performed in a water-based media for 1-2 hours, and quenching of unreacted MAL groups was achieved via lOOmM L-Cys stock.
- Scheme 1 below is a schematic representation of hGH antagonist conjugation to an Albumin binder complex via a bi-functional Fmoc linker (MAL).
- MAL bi-functional Fmoc linker
- acylated hGH antagonists described throughout the application can be conjugated applying the same outlined above. Moreover, the length of the fatty add chain (C-20 or C-18 for example), the number of spacer units (for example 2-3 instances of AEEA) has no effect on the reaction yields.
- the hGH antagonist protein was thawed to room temperature on the bench, or in a 22 ⁇ 3°C water bath.
- the thawed protein was diluted to -1-1.5 mg/mL with a dilution buffer that contains: 20mM nA-Pi, 200Mm naCl, pH 7.
- the hGHA solution pH was adjusted to 7 using drop-wise of nAoh (IN).
- Diluted hGHA was filtered using a 0.22
- im PVDF/PES filter to remove any undesired precipitants. Filtered hGHA was analyzed for concentration using A280 via NanoDrop (EC 7.96).
- the binder solution WAs added to the hGHA solution (1.25eq binder: leq protein). Reaction was kept at RT for 2 hours and analyzed via RP-HPLC for reaction progress every 1 hour (See FIG. 23). At the end of the reaction, the residual binder was quenched by addition of 1 volume unit of quenching solution (lOOmM L-Cys, pH 7) to 100 volume units of the reaction solution.
- the conjugated hGHA sample was filtrated using 0.22
- FIG. 23 shows RP-HPLc Chromatograms of hGHA conjugation reaction to an Albumin binder complex
- the top chromatogram shows the hGHA solution; the middle chromatogram shows the Binder solution; and the bottom chromatogram shows the end of reaction (the acylated hGHA).
- the hGHA is 1202-1
- the binder is type B (C-20, MAL)
- the acylated hGHA is 1202-1D
- FIG. 24 shows LC-ms Analysis to the hGHA and the conjugated protein.
- the top chromatogram shows hGHA deconvoluted mass (SEQ ID No: 5) and the bottom chromatogram shows the conjugated protein (1202-1D) deconvoluted mass.
- the conjugation step was performed between the Acetyl Bromide group of the Albumin binder complex, and thiol group located on the Cys side chain of the hGH antagonist (See Scheme 2).
- the reaction was performed in a water-based media for 3-5 hours, and quenching was performed via lOOmML-Cys stock.
- Scheme 2 below is a schematic representation of hGH antagonist conjugation to Albumin binder complex (Acetyl Bromide).
- the hGH antagonist was thawed to RT on the bench or in a 22 ⁇ 3°C water bath.
- the thawed protein was diluted to ⁇ 1 -1.5 mg/mL with a dilution buffer that contains: 20mM na-Pi, 200Mm NaCl pH 7.
- Diluted MOD-1202x was filtered using a 0.22
- im PVDF/PES filter in order to remove any undesired precipitants. Filtered hGHA was analyzed for concentration using A.280 via NanoDrop (EC 7.96).
- reaction buffer containing: 0.05%HPcd, 20mM na-Pi, 200mM NaCl pH 7 were added to 9 volume units of the hGHA, and a sample was withdrawn for analysis using RP-HPLC (See FIG. 25). The protein solution was kept at RT until the reaction was started.
- Binder handling and preparation [0498] Binder handling and preparation:
- Lyophilized binder was thawed on the bench for 1 hour.
- the thawed binder was reconstituted in reaction buffer containing: 0.005%HPcd, 20mM na-Pi, 200mM NaCl pH 7 to reach a concentration of 2-4 mg/mL (wt/V).
- the binder concentration was analyzed using A.205 via NanoDrop, and a sample was taken for RP-HPLC analysis (See FIG. 26).
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Chemical & Material Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Organic Chemistry (AREA)
- Medicinal Chemistry (AREA)
- Endocrinology (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Molecular Biology (AREA)
- Pharmacology & Pharmacy (AREA)
- Genetics & Genomics (AREA)
- Biochemistry (AREA)
- Gastroenterology & Hepatology (AREA)
- Zoology (AREA)
- Toxicology (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Biophysics (AREA)
- Epidemiology (AREA)
- Animal Behavior & Ethology (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Cell Biology (AREA)
- Immunology (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
- Peptides Or Proteins (AREA)
Abstract
Described herein are growth hormone receptor antagonists and modified growth hormone receptor antagonists. The growth hormone receptor antagonists and modified growth hormone receptor antagonists are useful in treating diseases or conditions, such as cancer and acromegaly, that are responsive to human growth hormone receptor antagonists.
Description
HUMAN GROWTH HORMONE RECEPTOR ANTAGONISTS AND METHODS OF
USE THEREOF
CROSS REFERENCE TO RELATED APPLICATIONS
[0001] This application claims the benefit of U.S. Provisional Application Serial No. 63/592,760, filed on October 24, 2023, which is incorporated in its entirety herein by reference.
SEQUENCE LISTING
[0002] A Sequence Listing conforming to the rules of WIPO Standard ST.26 is hereby incorporated by reference. The instant application contains a Sequence Listing which has been submitted electronically in XML file format The electronic document, created on October 14, 2024, is entitled “P-625439-PC_ST26.xml”, and is 12,680 bytes in size.
FIELD OF THE INVENTION
[0003] Described herein are growth hormone receptor antagonists and modified growth hormone receptor antagonists. The growth hormone receptor antagonists and modified growth hormone receptor antagonists are useful in treating diseases or conditions, such as cancer and acromegaly, that are responsive to human growth hormone receptor antagonists.
BACKGROUND
[0004] The described invention relates in general to compositions for use as receptor antagonists, and more specifically to human growth hormone antagonists that have the potential to be highly effective therapeutics.
[0005] Human growth hormone, also known as somatotropin or somatropin, is a peptide hormone that stimulates growth, cell reproduction, and regeneration in humans and other animals. Growth hormone is a type of mitogen that is specific only to certain kinds of cells and is a 191 -amino acid, single-chain polypeptide that is synthesized, stored, and secreted by somatotropic cells within the lateral wings of the anterior pituitary gland.
[0006] A receptor is a protein molecule usually found embedded within the plasma membrane surface of a cell that receives diemical signals from outside the cell. When such diemical signals bind to a receptor, they cause some form of cellular/tissue response such as, for example, a change in the
electrical activity of the cell. In this sense, a receptor is a protein molecule that recognizes and responds to endogenous chemical signals. An agonist, such as human growth hormone, is a chemical composition that binds to a receptor and activates the receptor to produce a biological response. Whereas an agonist causes an action, an antagonist blocks the action of the agonist and an inverse agonist causes an action opposite to that of the agonist A receptor antagonist is a type of receptor ligand or drug that blocks or dampens agonist-mediated responses rather than provoking a biological response itself upon binding to a receptor. These compositions are sometimes called blockers and examples include alpha blockers, beta blockers, and calcium channel blockers. In pharmacology, antagonists have affinity but no efficacy for their cognate receptors, and binding will disrupt the interaction and inhibit the function of an agonist or inverse agonist at receptors. Antagonists mediate their effects by binding to the active (orthosteric) site or to other (allosteric) sites on receptors, or they may interact at unique binding sites not normally involved in the biological regulation of the receptor’s activity. Antagonist activity may be reversible or irreversible depending on the longevity of the antagonist-receptor complex, which, in turn, depends on the nature of antagonist-receptor binding. The majority of drug antagonists achieve their potency by competing with endogenous ligands or substrates at structurally defined binding sites on receptors. By definition, antagonists display no efficacy to activate the receptors they bind and antagonists do not maintain the ability to activate a receptor. Once bound, however, antagonists inhibit the function of agonists, inverse agonists, and partial agonist
[0007] Acromegaly is a syndrome that results when the anterior pituitary gland produces excess human growth hormone (hGH) after epiphyseal plate closure at puberty. If hGH is produced in excess prior to epiphyseal plate closure, the result is gigantism (or giantism). A number of disorders may increase the pituitary’s hGH output, although most commonly it involves a tumor called pituitary adenoma, derived from a distinct type of cell (somatotrophs). Acromegaly most commonly affects adults in middle age and can result in severe disfigurement, complicating conditions, and premature death if untreated. Because of its pathogenesis and slow progression, the disease is hard to diagnose in the early stages and is frequently missed for years until changes in external features, especially of the face, become noticeable.
[0008] Acromegaly is a rare endocrine condition caused by excessive secretion of growth hormone (GH). The resulting clinical effects are attributable to high serum concentrations of both GH and insulin-like growth factor-1 (IGF-1), which is GH dependent GH antagonist is indicated for the
treatment of acromegaly in patients who demonstrate an inadequate response to surgery, and/or radiation therapy, and other medical therapies, or for whom these therapies are not appropriate. Treatment with GH antagonist aims to restore normal serum IGF-1 levels and improve clinical signs and symptoms.
[0009] Changes associated with acromegaly include coarsening of body hair, thickening and darkening of the skin, enlargement and overactivity of sebaceous and sweat glands such that patients frequently complain of excessive perspiration and offensive body odor, overgrowth of the mandible, cartilaginous proliferation of the larynx causing a deepening of the voice, and enlargement of the tongue. In addition, excess growth hormone (“GH”) in these patients is responsible for proliferation of articular cartilage which may undergo necrosis and erosion and endoneurial fibrous proliferation which causes peripheral neuropathies. Excess GH also increases tubular reabsorption of phosphate and leads to mild hyperphosphatemia. Many of these symptoms are also seen in patients with gigantism
[0010] The hallmark of treatments for acromegaly and gigantism is their ability to lower insulin-like growth factor- 1 ("IGF-1") in plasma and/or tissue through either destruction of the pituitary or drug treatment The role of IGF-1 in GH-mediated disorders, such as acromegaly and gigantism is well recognized (Melmed et al., Amer. J. Med. 97: 468-473 (1994)).
[0011] Another disorder that has been associated with abnormal GH levels is diabetes mellitus (DM). Characteristically, patients with poorly controlled DM have been found to have high levels of circulating GH It has been shown that hypophysectomy could reduce diabetic hyperglycemia, thus strongly implicating the role of GH as an active component of the metabolic derangements of diabetes (Houssay and Biasotti, Rev. Soc. Argent Biol. 6: 251-296 (1930)). It has been suggested that hypersecretion of GH may be the cause as much as the consequence of poor diabetic control (Press etal, New England J. Med. 310: 810-814 (1984)).
[0012] Most diabetics do not die of acute hyperglycemia. The overwhelming majority of diabetics die from complications associated with diabetes such as end organ failure. While diabetes affects almost all organs, heart and kidney failure are the most common causes of death. Other organs or systems that are commonly affected by DM are the eyes, the blood vessels and the nervous system. Patients with long standing diabetes will commonly have diabetic retinopathy, angiopathy and peripheral neuropathy. It is possible that normal GH secretion has a permissive role in patients predisposed to severe diabetic retinopathy. In such patients and in others in whom attempts to
optimize glycemic control are unsuccessful, pharmacologic intervention may be beneficial not only in improving glycemic control but also in preventing severe proliferative diabetic retinopathy. Gerich etal, New England J. Med. 310:848-850 (1984).
[0013] Diabetes-related nephropathy is another disorder that has the potential to be treated therapeutically using growth hormone receptor antagonists. It has been established that that in various experimental models of type 1 diabetes, GH and IGFs have measurable effects on both short- and long-term renal changes (Alex F. Muller and others, Growth Hormone Receptor Antagonists, The Journal of Clinical Endocrinology & Metabolism, Volume 89, Issue 4, 1 April 2004, Pages 1503- 1511). The hypothesis that GHR antagonists may be used in the treatment of diabetic renal changes was supported by long-term studies in streptozotocin-induced diabetic mice transgenic for a GHR antagonist Somatostatin analogs, but not GHR antagonists, have been studied in patients with type 1 diabetes. Human studies investigating the renal effects of GHR blockade in diabetes mellitus (type 1 or 2) have not been performed, and there remains a need for GHR antagonists with potential beneficial effects in diabetic nephropathy.
[0014] Proliferative diabetic retinopathy is one of the leading causes of blindness in the United States and ranks second only to senile macular degeneration as a cause of permanent blindness. Benson et al., Diabetic Retinopathy, Duane, T., (eds.), Harper & Row, Philadelphia, Pa. pp. 1-24. In juveniles with insulin dependent diabetes, there is no evidence of diabetic retinopathy up to 5 years. However, 27% of juveniles who have had diabetes for 5 to 10 years have diabetic retinopathy. Also 71% of juveniles who have had diabetes for longer than 10 years have diabetic retinopathy. Greater than 90% of jirveniles who have diabetes for 30 years will ultimately have diabetic retinopathy. Also, the 5 year mortality rate for individuals blind from diabetic retinopathy is 36%, in which death generally is caused by cardiac or kidney complications.
[0015] The kidneys are another organ that is affected by DM. Chen et al., Endocrinology 136:660- 667 (1995). One type of pathology seen in patients with diabetic nephropathy is glomerulosclerosis. Glomerulosclerosis is the sclerosis of mesangial cells which is preceded by mesangial cell proliferation Glomerular cells are responsible for filtering the blood and thus directly affect kidney function
[0016] The pathogenesis of proliferative diabetic retinopathy is believed to be mediated by (Hl It has been shown that human GH stimulates proliferation of human retinal microvascular endothelial cells in the diabetic; proliferation of these cells is the primary cause of proliferative diabetic
retinopathy. Rymaszewski et al., Proc. Natl. Acad. Sci. USA 88:617-621 (1991). Thus, the involvement of GH in end organ damage in the diabetic is well established. Smith et al., Abstract of Presentation at ARVO meeting (May, 1995).
[0017] Commercially available, daily injected, hGH antagonist (Somavert) is covalently bound to several polyethylene glycol (PEG) polymers. It acts by blocking the cell surface GH receptor and preventing its activation. In order to modify hGH to hGH antagonist a single mutation of Glyl20 is required. The hGH antagonist binds to the hGHR but there is no signal transduction. Since the efficacy of antagonist depends on its affinity to the receptor, additional point mutations were introduced and were shown to improve its affinity to GHR
[0018] SOMAVERT® (pegvisomant) is an analog of human growth hormone (“hGH”) that has been structurally altered. The amino acid sequence of the protein component/intermediate (B-2036) of pegvisomant differs from the amino acid sequence of hGH at nine positions. The specific amino add substitutions are as follows: H18D, H21N, G120K, R167N, K168A, D171S, K172R, E174S, and I179T. As is well recognized in the art, the first letter (i.e., H18D) represents the amino add in the sequence of hGH at the numbered position (i.e., 18th amino add position as indicated by H18D) which is substituted with the amino acid designated by the second letter (i. e. , Hl 8D). Therefore, Hl 8D designates a substitution of the amino acid his by the amino acid asp at the 1 Sth amino acid position of the wild-type hGH amino acid sequence. Structurally, pegvisomant is a protein (containing 191 amino acid residues) to which predominantly 4 to 6 PEG units are covalently bound.
[0019] limitations thereby expanding the indications to the so-called peripherally acting agents [0020] SOMAVERT® is a genetically-engineered analogue of hGH that functions as a highly selective growth hormone receptor antagonist indicated for the treatment of acromegaly (van der Lely, et al., The Lancet 358: 1754-1759 (2001)). Such compositions are used if the tumor of the pituitary gland causing the acromegaly cannot be controlled with surgery or radiation and the use of somatostatin analogues is unsuccessful. Although several of the amino add side drain residues in SOMAVERT® are derivatized with polyethylene glycol (PEG) polymers, the produtt is still administered once-daily, indicating that the pharmaceutical properties are not optimal. SOMAVERT® (pegvisomant) is typically delivered as a powder that is mixed with water and injected under the skin.
[0021] Real life studies of SOMAVERT® reported an efficient long-term usage only for part of the acromegaly patients. Costs, compliance (daily injections) and side or undesired effects (Ever toxicity,
enlargement of the adenoma) currently limit its use. In this setting, the advent of other compounds targeting the GH receptor may overcome some of these.
[0022] Albumin (A “chaperone "protein mediating die increase of a drugs half-life)
[0023] Human serum albumin (“HSA” or “albumin”) is the most abundant protein in plasma (-60% of all proteins, -40 [g/L]). It is a highly soluble and stable (pH, structural, and temp), nonglycosylated, negatively charged (and thus avoids filtration in the renal glomeruli) and IS considered to be highly hydrophilic. Albumin contains 17 disulfide bridges that contribute to its structural stability, thermal stability, and a single odd cysteine residue in position 34. Overall, the Cys34 residue makes up -80% of the free thiols in plasma. HSA is synthesized as a 585- residue single chain globular protein lacking prosthetic groups and glycosylation
[0024] Albumin possess an extremely long half-life (-19 days). A central contributor to the latter is its ability to bind the FcRn receptor, be rescued, and be recycled into the blood stream (Larsen, Maja Thim, et al. "Albumin-based drug delivery: harnessing nature to cure disease." Molecular and cellular therapies 4.1 (2016): 1-12). Almost every body fluid contains some amount of HSA In addition, HSA occurs within cells like ovarian cells, brain cells, peripheral nerve cells, lymphocytes, macrophages, and other cells. Tumor cells often take up HSA to a greater extent than non- tumorous cells of the same type. For example, albumin makes up 19% of the soluble protein of breast cancer cells.
[0025] All of the above characteristics attribute to the albumin long half-life, allowing it to take part in numerous important physiological activities. Due to its versatile and multiple binding domains it serves as a transporter and stabilizer to a variety of molecules such as: fatty acids, aromatics, ions, and peptides. The latter characteristics sparkled the imagination of scientist for designing Albuminbinding drugs that display an increased half-life.
[0026] There are several key attributes gained when binding an Albumin to a drug, specifically to a relatively small drug (aromatic and aliphatic compounds, peptides, and small proteins) with a short half-life. The attributes are as follows: drug protection from enzymatical degradation; drug physical stability; reduce renal clearance of the drug (size matter); the recycling mechanism of the Albumin bounded drug via the FcRn receptor; and molecules capable of binding to the neonatal Fc receptor (“FcRn”), such as IgG molecules and albumin, can be rescued from lysosomal degradation and can be recycled into the blood stream.
[0027] The Neonatal FC receptor (“FcRN”) was first found to the responsible for transporting antibodies of IgG class from the mother to the fetus. Since the early discovery, it was found that the
FcRN receptor is broadly expressed in many other tissues, and despite the unrelated structure, can also bind Albumin (Zorzi, Alessandro, Sara Linciano, and Alessandro Angelini. "Non-covalent albumin-binding ligands for extending the circulating half-life of small biotherapeutics." MedChemComm 10.7 (2019): 1068-1081).
[0028] Several families of Albumin probes (or Albumin binders in other words) have already been shown to successfully prolong the half-life of drugs. For each of these families, one can find examples of market available products displaying improved drug longevity, reduced frequent of injection, and increased safety. Among such binders are aromatic compounds, peptides, and nano structures, with fatty acids being one of the most promising albumin binding moieties. Moreover, the ability of serum albumin to bind long fatty adds with a high affinity inspired the use of posttranslational acylation as a safe and natural platform for prolonging the half-life of peptides and small proteins.
[0029] There are seven different fatty acids binding domains spread around three Albumin binding (and sub binding) sites. An illustration of the Albumin binding sites with respect to the Fatty acids, bi-valent ions, and known drugs is presented in FIG. 1. It was established that the Din and DI loop are highly important for the FcRn binding, and that there are several fatty adds binding sites located at these regions as well. Therefore, upon designing an albumin-binding drug, must take into consideration the potential alternation of the Albumin conformation, and thus hampering its FcRn binding.
[0030] Long chain fatty adds (LCFAs, i.e., carboxylic adds having a non-branched aliphatic chain having 16-20 carbon atoms in its backbone) are essential for many cellular functions. LCFAs serve as an important energy resource and are also critical components of lipids, hormones, and proteins. LCFAs are known to be bound and transported by HSAs within the human body.
[0031] Fatty adds (FA) can be conjugated to therapeutic protdns to form longer-acting derivatives. This principle for prolongation of protdn or peptide half-life is based on the fad that FA can bind to human serum albumin (HSA; also referred to as albumin binding probes). The association of a FA with human serum albumin in the blood stream can lead to a substantial prolongation of the half-life of the therapeutic protein as it will recycle together with albumin through the neonatal Fc receptor. FA and derivatives thereof (e.g., corresponding methyl esters) have shown similar albumin-binding properties (Spector AA, J Lipid Res 1975; 16: 165-79).
[0032] Formation of conjugates between LCFAs and many small molecules is known to enhance the serum stability and delivery of the small molecules by a mechanism facilitated by binding of the small
molecule-LCFA conjugate with HAS.
[0033] The present application has developed new hGH antagonists and harnessed the benefits of albumin binding to produce modified hGH antagonists with improved pharmacokinetic and therapeutic properties.
SUMMARY
[0034] In one aspect, disclosed herein is a human growth hormone receptor antagonist (“GHR antagonist’ ’ or “hGHA"), comprising growth hormone receptor antagonist G120K (SEQ ID NO: 2) and at least one of the following amino acid mutations: Hl 8D, H21N, F54P, R64K, R167N, D171S, E174S, I179T.
[0035] In a related aspect, any of said mutations increase the affinity of the GHR antagonist to the growth hormone receptor.
[0036] In another aspect, the hGHA further comprises at least one amino acid mutation to cysteine. In a related aspect, the amino acid mutated to cysteine is T3, E39, P48, Q69, N99, L129, T135, T142, H151 or combinations thereof. In yet another related aspect, the amino add mutated to cysteine is L129 or T135.
[0037] In one aspect, the hGHA comprises the amino acid sequence of SEQ ID NO: 5. In a related aspect, the hGHA has the following amino add mutations: G120K, L129C, R167N, D171S, E174S, andI179T.
[0038] In one aspect, the hGHA comprises the amino acid sequence of SEQ ID NO: 6. In a related aspect, the hGHA has the following amino acid mutations: H18D, H21N, F54P, R64K, G120K, L129C, R.167N, D171S, E174S, andI179T.
[0039] In one aspect, the hGHA comprises the amino acid sequence of SEQ ID NO: 7. In a related aspect, the hGHA has the following amino acid mutations: H18D, H21N, G120K, L129C, R167N, D171S, E174S, andI179T.
[0040] In one aspect, the hGHA comprises the amino acid sequence of SEQ ID NO: 8. In a related aspect, the hGHA has the following amino acid mutations: H18D, H21N, G120K, T135C, R167N, D171S, E174S, andI179T.
[0041] In another aspect, the hGHA comprises the amino acid sequence of any one of SEQ ID NOs: 5 to 8.
[0042] In a one aspect, disclosed herein is a modified human growth hormone receptor antagonist
(“modified GHR antagonist” or “modified hGHA”) having the structure of formula (I): W-X-Y-Z (I) wherein:
W represents a binder;
X represents a spacer;
Y represents an optional linker;
Z represents a human growth hormone receptor antagonist (“GHR antagonist”); or a pharmaceutically acceptable salt thereof.
[0043] In a related aspect, the element Z of the modified hGHA comprises any of the hGHA disclosed herein. In another aspect, the element Z of formula I comprises any of the hGHA disclosed herein. In yet another related aspect, the GHR antagonist of formula I is any of the hGHA disclosed herein.
[0044] In a related aspect, the element Z of the modified hGHA comprises the amino add sequence of any one of SEQ ID NOs: 5 to 8. In another aspect, the element Z of formula I comprises the amino acid sequence of any one of SEQ ID NOs: 5 to 8. In yet another related aspect, the GHR antagonist of formula I comprises the amino add sequence of any one of SEQ ID NOs: 5 to 8.
[0045] In one aspect, W of formula I is a fatty add. In a related aspect, W of formula I is octadecanedioic acid (Cl 8 diacid) or is eicosanedioic add (C20 diacid).
[0046] In a related aspect, W of formula I is represented by Formula II:
O
OH
HO o
[0047] In a related aspect, W of formula I is represented by Formula II-A:
O
HO
O
[0048] In a related aspect, W of formula I is represented by Formula HI: o
OH
HO o
[0049] In a related aspect, W of formula I is represented by Formula HI -A: o
HO o
[0050] In a related aspect, the element X of formula I is gGlu-dun-(AEEA)m-Cys-Glyp, gGlu-Glun- (AEEA)m-Lys(AcBr)-Glyp, or gGlu-Glun-(AEEA)m-Lys-Glyp and n is 1, 2, or 3; m is 1, 2, or 3; and p is 1, 2, or 3.
[0051] In another aspect, the element X of formula I is gGu-Gita-(AEEA)n-Cys-Oyn, n is 1, 2, or 3. In another aspect, the element X of formula I is gGlu-Glu-(AEEA)m-Cys-Gly, m is 1, 2, or 3.
[0053] In one aspect, the element X of formula I is gGlu-Glu-(AEEA)m-Cys-Gly, m is 1, 2, or 3 and is represented by Formula IV.
[0054] In another aspect, the element X of formula I is represented by Formula IV- A:
[0056] In another aspect, the element X of formula I is represented by Formula IV-C:
[0058] In another aspect, the element X of formula I is represented by Formula IV-E:
[0059] In one aspect, the element X of formula I is gGlu-Glu-(AEEA)m-Lys(AcBr)-Gly, m is 1, 2, or 3.
[0061] In one aspect, the element X of Formula I is represented by Formula V-A:
H
HQ ,o o
H 9r o O H
H2N r-N OH o O
HO o n= 1.2, 3
1, 2, 3
[0063] In one aspect, the element X of Formula I is represented by Formula V-C:
[0064] In one aspect, the element X of formula I is gGlu-Glu-(AEEA)m-Lys-Gly, m is 1, 2, or 3.
[0066] In one aspect, the element X of formula I is gGlu-Glu-(AEEA)m-Lys-Gly, m is 1, 2, or 3.
[0069] In one aspect, the element X of formula I is represented by Formula VI-C:
[0070] In one aspect, the element Y of formula I is Chloropropane-2-one-Fmoc-Mal. In a related aspect, the element Y of formula I is 2-(3-(2,5-dioxo-2H-pyrrol-l(5H)-yl)propanamido)-9H-fluoren- 9-yl)methyl 3-chloro-2-oxopropylcarbamate. In a related aspect, the element Y of formula I is Chloropropane-2-one-Fmoc-Mal and is represented by Formula VII:
[0071] In one aspect, the element Y of formula I is Chloropropane-2-one-Fmoc-Mal and is represented by Formula VII- A:
O o jy .
N H
O o
HN o
Q
[0072] In one aspect, the element Y of formula I is Chloropropane-2-one-Fmoc-Mal and is represented by Formula VII -B:
O
O
N H o
O
HN o o
Cl
[0073] In one aspect, the element Y of formula I is Chloropropane-2-one-Fmoc-Mal and is represented by Formula VH-C:
O o jy .
N H o o
HN o
Q
[0074] In one aspect, the element Y of formula I is Mal-NRFmoc-NHS. In a related aspect, the element Y of formula I is Mal-NRFmoc-NHS and is represented by Formula X:
O
O
N H
O
O o
O N o
[0075] In one aspect, the element Y of formula I is Mal-NRFmoc-NHS and is represented by Formula X-A:
O o
N H o o O
[0076] In one aspect, the element Y of formula I is Mal-NRFmoc-NHS and is represented by Formula X-B:
[0077] In one aspect, the element Y of formula I is Mal-NRFmoc-NHS and is represented by Formula
[0078] In one aspect, the bond between the GHR antagonist and the linker in formula I is a stable covalent bond.
[0079] In one aspect, the bond between the GHR antagonist and the linker in formula I is a reversible covalent bond.
(Formula XV).
[0081] In one aspect, the modified GHR antagonist comprises the following formula:
O. Vss.
HN
,0
HO^O hGHA Variant (SEQ ID NO: 8)
(Formula XVIII).
[0084] In one aspect, the modified GHR antagonist comprises the following formula:
O;
HN
O=< o
3L. M. V
O' hGHA Variant
(Formula XIX).
[0085] In one aspect, the modified GHR antagonist comprises the following formula:
(Formula XXII).
(Formula XXIII)
[0089] In one aspect, the modified GHR antagonist comprises the following formula:
(Formula XXIV).
(Formula XXV).
[0091] In one aspect, the modified GHR antagonist comprises the following formula:
(Formula XXVI)
(Formula XXVII).
[0093] In one aspect, the modified GHR antagonist comprises the following formula:
(Formula XXVIII).
(F ormula XXIX).
[0095] In one aspect, the modified GHR antagonist comprises the following formula:
(Formula XXX).
[0096] In one aspect, disclosed herein is a method of inhibiting human growth hormone (hGH) action in a subject, the method comprising: administering to a subject in need thereof an effective amount of any GHR antagonist disclosed herein or any modified GHR antagonist disclosed herein, to inhibit hGH action in the subject In a related aspect, the method disclosed comprises administering to any of the GHR antagonists or any of the modified GHR antagonists disclosed herein in an amount effective to treat, prevent, or ameliorate a H hGH gh related disorder in the subject
[0097] In a related aspect, the hGH related disorder is one or more of: acromegaly; age-related macular degeneration; cancer; diabetes; gigantism, vascular eye diseases, diabetic retinopathy, retinopathy of prematurity, and retinopathy of sickle-cell anemia; and, nephropathy.
[0098] In one aspect, disclosed herein is a method of treating, preventing, or ameliorating a hGH related disorder, the method comprising: administering to a subject in need thereof an effective amount of any GHR antagonist disclosed herein or any modified GHR antagonist disclosed herein, to treat, prevent, or ameliorate a hGH related disorder in the subject
[0099] In one aspect, disclosed herein is a method of producing an albumin binding complex on a resin, the steps comprising: a. Synthesizing a protected binder backbone of Formula XXXI on a resin
n - 15,17 i = 1,2,3
(Formula XXXI); b. Removing the methyltrityl (Mtt) resulting in a deprotected binder backbone; c. Coupling a linking moiety to the deprotected binder backbone resulting in an albumin binding peptide complex; d. Cleaving the albumin binding peptide complex from the resin; e. Purifying the albumin binding peptide complex; and f. Lyophilizing the albumin binding peptide complex.
[0100] In one aspect, the synthesizing of a binder backbone is done using solid-phase peptide synthesis (SPPS).
[0101] In a related aspect, the binder backbone is tBu-Fatty acid-gGlu(OtBu)-Glu(OtBu)-(AEEA)i- Lys(Mtt)-Gly-Wang resin, wherein i is 2 or 3 and the Fatty acid is eicosanedioic or octadecandioic. [0102] In one aspect, the removing in step (b) is done by applying 1%TFA in DCM wash cycles. In a related aspect, the removing in step (b) deprotects the Lys side chain. In a further aspect, the intermediate composition following step (b) is the tBu-eiocosanedioc-yGiu(OtBu)-Glu(C)tBu)- (AEEA)3-Lys-Gly-Wang resin, tBu-eiocosanedioc-yGlu(C)tBu)-Glu(C)tBu)-(AEEA)2-Lys-Gly- Wang resin, tBu-octadecandioic-yGlu(OtBu)-Glu(C)tBu)-(AEEA)3-Lys-Gly-Wang resin, or tBu- octadecandioic-yGlu(OtBu)-Glu(C)tBu)-(AEEA)2-Lys-Gly-Wang resin.
[0103] In one aspect, the linker moiety and deprotected binder backbone are added to a reactor and stirred. In a related aspect, the linker moiety is coupled to the primary amine of the deprotected Lys side chain of the peptide backbone.
[0104] In one aspect, the resin is Wang resin.
[0105] In a further aspect, the said cleaving in step (d) is done with a cocktail containing 95%TFA: 5% H2O, with a ratio of 10ml cleavage cocktail: lgr peptide-resin.
[0106] In one aspect, the purifying in step (e) is done on RP-HPLC column.
[0107] In yet a further aspect, the linking moiety in step (c) is MAL-NRFmoc. In a related aspect, around 2-3eq of the MAL-NRFmoc is added to the reactor.
[0108] In one aspect, the pH in the reactor is around 5.0.
[0109] In one aspect, the linking moiety in step (c) is Acetyl-Br. In a related aspect, around 20eq of Bromoacetic anhydride is dissolved in DCM, and 20eq of DIPEA is added to the solution resulting in a Bromoacetic anhydride solution reatty for Lys side chain conjugation In another aspect, the Bromoacetic anhydride solution is added to the reactor and stirred. In a related aspect, the stirring comprises 1 hour.
(Formula XXXII).
[0111] In one aspect, the albumin binding complex is the albumin binding complex is tBu- eicosanedioic-yGlu(OtBu)-Glu(OtBu)-(AEEA)3-Lys(Ac-Br)-Gly-Wang resin, tBu-eicosanedioic- yGlu(OtBu)-Glu(OtBu)-(AEEA)2-Lys(Ac-Br)-Gly-Wang, resin tBu-octadecandioic-yGiu(OtBu)- Glu(OtBu)-(AEEA)3-Lys(Ac-Br)-Gly-Wang resin, or tBu-octadecandioic-yGiu(OtBu)-Glu(C)tBu)- (AEEA)2-Lys(Ac-Br)-Gly-Wang resin
(Formula XXXIII).
[0113] In one aspect, the albumin binding complex is tBu-eicosanedioic-yGlu(C)tBu)-Glu(C)tBu)-
(AEEA)3-Lys(MAL-NRFmoc)-Gly-Wang rreessiinn,, tBu-eicosanedioic-yQu(OtBu)-Qu(OtBu)-
(AEEA)2-Lys(MAL-NRFmoc)-Gly-Wang rreessiinn,, tBu-octadecandioic-ydu(OtBu)-du(C)tBu)-
(AEEA)3-Lys(MAL-NRFmoc)-Gly-Wang resin, or tBu-octadecandioic-yGlu(C)tBu)-Glu(C)tBu)- (AEEA)2-Lys(MAL-NRFmoc)-Gly-Wang resin
[0114] In one aspect, disclosed herein are albumin binding complexes comprising Formula XXXII or Formula XXXIII.
[0115] In another aspect, disclosed herein are albumin binding complexes comprising tBu- eicosanedioic-yGlu(OtBu)-Glu(C)tBu)-(AEEA)3-Lys(Ac-Br)-Gly-Wang resin, tBu-eicosanedioic- ydu(OtBu)-Glu(C)tBu)-(AEEA)2-Lys(Ac-Br)-Gly-Wang resin, tBu-octadecandioic-yGlu(C)tBu)- Glu(OtBu)-(AEEA)3-Lys(Ac-Br)-Gly-Wang resin, or tBu-octadecandioic-yGlu(C)tBu)-Glu(C)tBu)- (AEEA)2-Lys(Ac-Br)-Gly-Wang resin. In another aspect, disclosed herein are albumin binding complexes comprising tBu-eicosanedioic-yGlu(C)tBu)-du(C)tBu)-(AEEA)3-Lys(MAL-NRFmoc)- dy-Wang resin, tBu-eicosanedioicjydu(OtBu)-du(OtBu)-(AEEA)2-Lys(MAL-NRFmoc)-Gly- Wang resin, tBu-octadecandioicjyGlu(OtBu)-Glu(OtBu)-(AEEA)3-Lys(MAL-NRFmoc)-dy-Wang resin, or tBu-octadecandioic-yGlu(C)tBu)-Glu(C)tBu)-(AEEA)2-Lys(MAL-NRFmoc)-Gly-Wang resin.
BRIEF DESCRIPTION OF THE DRAWINGS
[0116] The patent or patent application file contains at least one drawing executed in color. Copies of this patent or patent application publication with color drawing(s) will be provided by the Office upon request and payment of the necessary fee.
[0117] The present disclosure of engineered growth hormone receptor antagonists, both as to their generation and method of use, together with objects, features, and advantages thereof, may best be understood by reference to the following detailed description when read with the accompanying drawings in which:
[0118] FIG. 1 is adapted from Ghuman, Jamie et al., “Structural basis of the drug-binding specificity of human serum albumin. ” Journal of molecular biology 353 1 (2005): 38-52, Figure 7, and shows the crystal structure of human albumin. FIG. 1 shows the crystal structure of human albumin solved in the presence of saturating amounts of palmitic acid. The a-helical structures of the three domains (DI, DII, and DM) are divided into subdomains (A and B) as indicated. Fatty acids binding sites are indicated via numbered circles (1-7) spread around the surface of the albumin three domains.
[0119] FIG. 2 shows the generic formula for acylated Hgh antagonists with a binder complex containing a non-reversible Fmoc linker. The colored shading shows the different components of the acylated Hgh antagonist including: binder, spacer, Hnker, and Hgha Variant The value X corresponds to the Hgha Variants 1, 3, 4, and 5 described herein.
[0120] FIG. 3 shows the generic formula for acylated Hgh antagonists with a binder complex containing a non-reversible Acetyl linker. The colored shading shows the different components of the acylated Hgh antagonist including: binder, spacer, Hnker, and Hgha Variant The value X corresponds to the Hgha Variants 1, 3, 4, and 5 described herein.
[0121] FIGS. 4A-4D show representative binding assay results of different Hgha variants with two binder types (Binder Type A which is a Cl 8 albumin binding moiety and Binder Type B which is a C20 albumin binding) and without a binder (naked variants). FIG.4A shows the binding assay results of the reference standard (RS), Hgha Variant 1 (without any binder complex attached), acylated Hgha complex 1A, and acylated Hgha complex IB. FIG. 4B shows the binding assay results of the reference standard (RS), Hgha Variant 3 (without any binder complex attached), acylated Hgha complex 3A, and acylated Hgha complex 3B. FIG. 4C shows the binding assay results of the reference standard (RS), Hgha Variant 4 (without any binder complex attached), acylated Hgha
complex 4A, acylated Hgha complex 4B. FIG. 4D shows the binding assay results of the reference standard (RS), Hgha Variant 5 (without any binder complex attached), acylated Hgha complex 5 A, and acylated Hgha complex 5B. These results show that the addition of binders leads to a minor decrease in potency compared to the naked variant
[0122] FIGS. 5A and SB show representative binding assay results of acylated Hgh antagonists IB and ID compared to Somavert (FIG. SA) and acylated Hgh antagonists 5B and 5D compared to Somavert (FIG. SB).
[0123] FIG. 6 shows the pharmacokinetic (PK) results of the acylated Hgh antagonists (1202-1A, 1202-1B, 1202-5 A, and 1202-5B) versus Somavert
[0124] FIG. 7 shows the changes in IGF-1 serum levels (PD effects) in SD rats from study #12219, following single SC injection of the different acylated Hgha complexes and Somavert
[0125] FIG. 8 shows the percentage changes in IGF-1 levels in SD rats from study #12219, following single SC injection of the different acylated Hgha complexes and Somavert
[0126] FIG. 9 shows the PK results from Study #12220 which are based on non-compartmental pharmacokinetic analysis of the acylated Hgh antagonists (1202-1B and 1202-5B) at different dosages.
[0127] FIG. 10 shows the changes in IGF-1 serum levels (PD effects) in SD rats from Study #12220, following single SC injection of the different acylated Hgha complexes and dosages.
[0128] FIG. 11 shows the percentage changes in IGF-1 levels in SD rats from Study #12220, following single SC injection of the different acylated Hgha complexes and dosages.
[0129] FIG. 12 shows the PK results based on non-compartmental pharmacokinetic analysis from Study #12222 of the acylated Hgh antagonists (1202-1D and 1202-5D) at different dosages.
[0130] FIG. 13 shows the changes in IGF -1 serum levels (PD effects) in SD rats from Study #12222, following single SC injection of the different acylated Hgha complexes and dosages.
[0131] FIG. 14 shows the percentage changes in IGF-1 levels in SD rats from Study #12222, following single SC injection of the different acylated Hgha complexes and dosages.
[0132] FIGS. ISA and 15B show a process diagram for the manufacture of the acylated Hgha complexes disclosed herein. FIG. ISA shows the extraction and refolding phase. FIG. 15B shows the chromatographic phase.
[0133] FIG. 16 shows a schematic illustration of the Albumin binder complex production routes.
[0134] FIG. 17 shows RP-HPCD chromatograms. The top chromatograph shows the binder
backbone before fatty add coupling (TFmoc), and the bottom chromatograph shows binder 1 backbone after fatty add coupling (8AA).
[0135] FIG. 18 shows RP-HPCD chromatograms. The top chromatogram shows the binder before MAL-NRFmoc linker conjugation, and the bottom chromatogram shows the binder after linker conjugation
[0136] FIG. 19 shows RP-HPCD chromatograms that represent the step of conjugating Ac-Br to the tBu- Eiocosanedioc-yGlu(OtBu)-Glu(OtBu)-(AEEA)3-Lys-Gly-Wang resin The top chromatogram shows the binder before Ac-Br linker conjugation, and the bottom chromatogram shows binder after linker conjugation
[0137] FIG. 20 shows RP-HPCD chromatograms of purified Eicosanedioic-yGlu-Glu-(AEEA)3- Lys(Ac-Br)-Gly-OH in the top chromatogram, and Eicosanedioic-yGlu-Glu-(AEEA)3-Lys(MAL- NRFmoc)-Gly-OH in the bottom chromatogram.
[0138] FIG. 21 shows mass spectrum of Eicosanedioic-yGlu-Glu-(AEEA)3-Lys(Ac-Br)-Gly-OH
[0139] FIG. 22 shows mass spectrum of Eicosanedioic-yGlu-Glu-(AEEA)3-Lys(MAL-NRFmoc)- Gly-OH
[0140] FIG.23 shows RP-HPLC chromatograms of Hgha conjugation reaction to an Albumin binder complex. The top chromatogram shows the Hgha solution; the middle chromatogram shows the Binder solution; and the bottom chromatogram shows the end of reaction (the acylated Hgha).
[0141] FIG. 24 shows LC-MS analysis to the Hgha and the conjugated protein. The top chromatogram shows Hgha deconvoluted mass. The bottom chromatogram shows the conjugated protein deconvoluted mass.
[0142] FIG.25 shows RP-HPLC chromatograms of Hgha conjugation reaction to an Albumin binder complex. The top chromatogram shows the Hgha solution; the middle chromatogram shows the Binder solution; and the bottom chromatogram shows the end of reaction (the acylated Hgha).
[0143] FIG. 26 shows LC-MS analysis to the Hgha and the conjugated protein. The top chromatogram shows Hgha deconvoluted mass. The bottom chromatogram shows the conjugated protein deconvoluted mass.
[0144] FIG. 27 shows the mean serum concentration of the MOD -1202-1 B and MOD-1202-1D acylated hGH antagonists in Study #OPK-009-PD.
[0145] FIG. 28 shows the changes in IGF1 serum levels following repeated SC injection of the different compounds in Study &OPK-009-PD.
[0146] FIG. 29 shows the changes in IGF1 relative to baseline following repeated SC injection of the different compounds in Study &OPK-009-PD.
[0147] FIG. 30 shows the PK results of MOD-1202-1D at various dosages compared to Somavert in Study #OPK-OW-PD.
[0148] FIG. 31 shows the changes in IGF1 serum levels following repeated SC injection of the different compounds in Study &OPK-010-PD.
[0149] FIG. 32 shows the changes in IGF1 relative to baseline following repeated SC injection of the different compounds in Study &OPK-010-PD.
DETAILED DESCRIPTION
[0150] In the following detailed description, numerous specific details are set forth in order to provide a thorough understanding of the GHR antagonists disclosed herein. However, it will be understood by those skilled in the art that preparation and uses of GHR antagonists disclosed herein may in certain cases be practiced without these specific details. In other instances, well-known methods, procedures, and components have not been described in detail so as not to obscure the disclosure presented herein. [0151] Throughout this application, various references or publications are cited. Disclosures of these references or publications in their entireties are hereby incorporated by reference into this application in order to more fully describe the state of the art to which this invention pertains.
[0152] A growth hormone antagonist, as used herein, refers to a compound that prevents, inhibits or reduces to any extent activation or expression of the growth hormone receptor. The compound that prevents or inhibits activation of the growth hormone receptor may act directly or indirectly on the growth hormone receptor. For example, the compound may bind or interact directly with the growth hormone receptor in some embodiments. In other embodiments the compound may act indirectly by blocking access of the endogenous neuronal growth hormone to the growth hormone receptor. For instance, the compound may be able to block access of the endogenous neuronal growth hormone to the growth hormone receptor by interfering with the expression or activity of growth hormone or blocking the growth hormone receptor binding site on growth hormone or by preventing the release of endogenous neuronal growth hormone.
[0153] As used herein, the term “human growth hormone receptor antagonist” may be used interchangeably with the term “human growth hormone antagonist”, “growth hormone receptor antagonist’ ’, or “growth hormone antagonist”, having all the same meanings and qualities. As used
herein, the term “growth hormone receptor antagonist” may be used interchangeably with the term “growth hormone antagonist ’, “human growth hormone receptor antagonist”, or “human growth hormone antagonist”, having all the same meanings and qualities.
[0154] The hormone-receptor complex between Hgh and the extracellular domain of its receptor (hGHbp) is known (Wells et al, Anna Rev. Biophys. Biomol. Struct, 22:329 (1993)). High- resolution structural and mutational analysis and structural analysis has shown that one molecule of Hgh binds two receptor molecules sequentially using distinct sites on the hormone, called sites 1 and 2 (Cunningham et al, Science, 244: 1081 (1989); Cunningham et al, Science, 254:821 (1991); De Vos et al, Science, 255:306 (1991)). As such, a number of additional growth hormone receptor (GHR) antagonists may be designed.
[0155] In one embodiment, the present disclosure provides engineered Hgh antagonists resulted from introducing amino acid variations to Hgh. One of ordinary skill in the art would readily employ various standard techniques known in the art to introduce amino acid variations into Hgh antibody and then test the resulting modified Hgh for antagonist activity. While standard techniques may be used, the resultant antagonist activity of the newly created antibodies is not predictable and must be analyzed to determine functionality.
[0156] Human Growth Hormone Antagonists
[0157] The term “Hgh polypeptide” also includes glycosylated Hgh, such as but not limited to, polypeptides glycosylated at any amino add position, N-linked or O-linked glycosylated forms of the polypeptide. Variants containing single nucleotide or amino acid changes are also considered as biologically active variants of Hgh polypeptide. In addition, splice variants are also included. The term “Hgh polypeptide” also includes Hgh polypeptide heterodimers, homodimers, heteromultimers, or homomultimers of any one or more Hgh polypeptides or any other polypeptide, protein, carbohydrate, polymer, small molecule, linker, ligand, or other biologically active molecule of any type, linked by chemical means or expressed as a fusion protein, as well as polypeptide analogues containing, for example, specific deletions or other modifications yet maintain biological activity.
[0158] The conversion of Hgh from a growth agonist to a growth antagonist requires only a single amino acid change at Hgh position 120 from the native glycine to any amino acid except alanine (Chen et al., 1994). This molecule, however, cannot be used as a therapeutic for conditions of excess growth (e.g., acromegaly) due to its short in vivo half-life. Researchers have addressed this problem by the addition of polyethylene glycol molecules to the Hgh antagonist Hgh G120K to decrease the
clearance of the molecule through the kidneys. Somavert®, an FDA approved treatment for acromegaly, contains 4-6 linear PEG molecules with molecular weights of 5000 Daltons each. The addition of the PEGs, which are attached randomly to surface lysines (van der Lely and Kopchick, 2006), increases the in vivo half-life of the antagonist from less than an hour to approximately 72 hours (Finn, 2009). The affinity of the PEGylated antagonist for the membrane bound receptor, however, is reduced approximately 30 fold compared with the unpegylated molecule (Ross et al., 2001). Despite the decrease in receptor affinity, Somavert® is an effective treatment for acromegaly, although a large daily dose of 5-30 mg is typically prescribed.
[0159] In one embodiment, the growth hormone comprises a human growth hormone.
[0160] In oonnee embodiment, the wild-type Hgh polypeptide sequence is: FPTIPLSRLFDNAMLRAHRLHQLAFDTYQEFEEAYIPKEQKYSFLQNPQTSLCFSESIPTPSN REETQQKSNLELLRISLLLIQSWLEPVQFLRSVFANSLVYGASDSNVYDLLKDLEEGIQTL MGRLEDGSPRTGQIFKQTYSKFDTNSHNDDALLKNYGLLYCFRKDMDKVETFLRIVQCRS VEGSCGF (SEQ ID NO: 1).
[0161] In oonnee embodiment, the Hgh receptor antagonist GG112200KK sequence is: FPTIPLSRLFDNAMLRAHRLHQLAFDTYQEFEEAYIPKEQKYSFLQNPQTSLCFSESIPTPSN REETQQKSNLELLRISLLLIQSWLEPVQFLRSVFANSLVYGASDSNVYDLLKDLEEKIQTL MGRLEDGSPRTGQIFKQTYSKFDTNSHNDDALLKNYGLLYCFRKDMDKVETFLRIVQCRS VEGSCGF (SEQ ID NO: 2). The Hgh receptor antagonist G120K (SEQ ID NO: 2) corresponds to the native hGH polypeptide sEQUence with a single amino acid mutation G120K which converts Hgh to Hgh antagonist
[0162] In one embodiment, the amino acid sequence of the Hgh antagonist corresponding to pegvisomant (SOMAVERT®) is:
FPTIPLSRLFDNAMLRADRLNQLAFDTYQEFEEAYIPKEQKYSFLQNPQTSLCFSESIPTPSN REETQQKSNLELLRISLLLIQSWLEPVQFLRSVFANSLVYGASDSNVYDLLKDLEEKIQTL MGRLEDGSPRTGQIFKQTYSKFDTNSHNDDALLKNYGLLYCFNADMSRVSTFLRTVQCRS VEGSCGF (SEQ ID NO: 3).
[0163] In one embodiment, the signal peptide of the Hgh antagonist comprises the following amino acid sequence: MATGSRTSLLLAFCHXCLPWLQEGSA (SEQ ID NO: 4).
[0164] In one embodiment, the growth hormone comprises a human growth hormone antagonist (“Hgh antagonist’ “GH antagonist’ or “Hgha”).
[0165] In another embodiment, the Hgh antagonist is a recombinant protein. In another embodiment, the Hgh antagonist is a recombinant glycoprotein.
[0166] In some embodiments, the GH antagonist is a growth hormone receptor (GHR) antagonist, a small molecule growth hormone receptor antagonist, a protein growth hormone receptor antagonist, a GHR inverse agonist, or an inhibitory nucleic add. Protein or peptide GHR antagonists are well known in the art and include, but are not limited to pegvisomant (SOMAVERT®), B2036, B2036- PEG, G120R, G120RhGH, Hgh receptor antagonist G120K or analogs thereof.
[0167] The term “Hgh polypeptide”, “Hgh”, “hGHa”, or “GHR antagonisf’ encompasses Hgh polypeptides comprising one or more amino acid substitutions, additions or deletions. Hgh polypeptides of the present invention may be comprised of modifications with one or more natural amino acids in conjunction with one or more non-natural amino add modification Substitutions in a wide variety of amino acid positions in naturally-occurring Hgh polypeptides have been described, including but not limited to substitutions that modulate one or more of the biological activities of the Hgh polypeptide, such as but not limited to, increase antagonist activity, increase solubility of the polypeptide, decrease protease susceptibility, convert the polypeptide into an antagonist, etc. and are encompassed by the term “Hgh polypeptide”, “Hgh”, “hGHa”, or “GHR antagonisf’.
[0168] In another aspect, the amino add substitution resulting in antagonist activity is at residue 120 and is lysine.
[0169] In one embodiment, the Hgh antagonist is mutated to improve its binding characteristics (See Lowman, Henry B., and James A. Wells. “ Affinity maturation of human growth hormone by monovalent phage display.” Journal of molecular biology 234.3 (1993): 564-578). In another embodiment, the number of mutations of the Hgh antagonist are selected to improve its binding characteristics. In another embodiment, the Hgh antagonist contains between 4 and 8 mutations to improve its binding characteristics. In another embodiment, the Hgh antagonist contains 4 mutations to improve its binding characteristics. In another embodiment, the Hgh antagonist contains 5 mutations to improve its binding diaraderistics. In another embodiment, the Hgh antagonist contains 6 mutations to improve its binding diaraderistics. In another embodiment, the Hgh antagonist contains 7 mutations to improve its binding characteristics. In another embodiment, the Hgh antagonist contains 8 mutations to improve its binding characteristics.
[0170] In one embodiment, the Hgh antagonist contains between 8 and 15 mutations to improve its binding characteristics. In another embodiment, the Hgh antagonist contains 8 mutations to improve
its binding characteristics. In another embodiment, the Hgh antagonist contains 9 mutations to improve its binding characteristics. In another embodiment, the Hgh antagonist contains 10 mutations to improve its binding characteristics. In another embodiment, the Hgh antagonist contains 12 mutations to improve its binding characteristics. In another embodiment, the Hgh antagonist contains 13 mutations to improve its binding characteristics. In another embodiment, the Hgh antagonist contains 14 mutations to improve its binding characteristics. In another embodiment, the Hgh antagonist contains 15 mutations to improve its binding characteristics.
[0171 ] In another embodiment, the Hgh antagonist disclosed herein has the following point mutations as shown in Table 1, when comparing the amino acid sequence of the mutated Hgh antagonist to the Hgh antagonist G120K
Table 1: Point Mutations of Hgh Antagonist as Compared to Wild Type Hgh
Amino Acid AA of Wild-type Hgh AA of Hgh Antagonist
Position
18 His Asp
21 His Asn
54 Phe Pro
64 Arg Lys
167 Arg Asn
171 Asp Ser
174 Glu Ser
179 He Th¬
[0172] In another embodiment, disclosed is a human growth hormone receptor antagonist wherein at least one amino acid of the human growth hormone receptor antagonist has been mutated to cysteine. In another embodiment, disclosed is a human growth hormone receptor antagonist wherein at least one amino add of the human growth hormone receptor antagonist has been mutated to cysteine in order to introduce a cysteine as a conjugation site for the binder complex. In another embodiment, disclosed is a human growth hormone receptor antagonist wherein at least one amino acid of the human growth hormone receptor antagonist has been mutated to cysteine in order to introduce a cysteine as a conjugation site for the binder complex and without interrupting the protein-receptor interaction
[0173] In another embodiment, disclosed is a human growth hormone receptor antagonist G120K
wherein at least one amino add of the human growth hormone receptor antagonist G120K has been mutated to cystdne. In another embodiment, disclosed is a human growth hormone receptor antagonist G120K wherdn at least one amino acid of the human growth hormone receptor antagonist G120K has been mutated to cysteine in order to introduce a cysteine as a conjugation site for the binder complex. In another embodiment, disclosed is a human growth hormone receptor antagonist G120K wherein at least one amino acid of the human growth hormone receptor antagonist G120K has been mutated to cysteine in order to introduce a cysteine as a conjugation site for the binder complex and without interrupting the protein-receptor interaction
[0174] In another embodiment, the amino acid of the Hgh receptor antagonist which is mutated to cysteine is T3, E39, P48, Q69, N99, G120, L129, T135, T142, H151 or combinations thereof. In another embodiment, the amino add of the Hgh receptor antagonist which is mutated to cysteine is T3, E39, P48, Q69, N99, G120, T142, Hl 51 or combinations thereof. In another embodiment, the amino add of the Hgh receptor antagonist which is mutated to cysteine is L129 or T135 or combinations thereof.
[0175] In another embodiment, the amino acid of the Hgh receptor antagonist G120K which is mutated to cysteine is T3, E39, P48, Q69, N99, L129, T135, T142, H151 or combinations thereof. In another embodiment, the amino acid of the Hgh receptor antagonist G120K which is mutated to cysteine is T3, E39, P48, Q69, N99, T142, H151 or combinations thereof. In another embodiment, the amino acid of the Hgh receptor antagonist G120K which is mutated to cysteine is L129 or T135 or combinations thereof.
[0176] In one embodiment, the Hgh receptor antagonist has the following mutations: G120K, L129C, R167N, D171S, E174S, and I179T (“1202-1” or “Variant 1”). The mutation of L129C in Variant 1 allows for a conjugation site for a binder complex. The mutation of G120K in Variant 1 imparts antagonist activity to the composition The mutations R167N, D171S, E174S, and I179T improve the binding characteristics or increase binding affinity of Variant 1.
[0177] In a related embodiment, the GHR antagonist of Variant 1 has the following mutations compared to Hgh: L129C; G120K; R167N; D171S; E174S; I179T. In another embodiment, Variant 1 comprises the following amino add sequence: MFPTIPLSRLFDNAMLRAHRLHQLAFDTYQEFEEAYIPKEQKYSFLQNPQTSLCFSESIPTPS NREETQQKSNLELLRISLLUQSWLEPVQFLRSVFANSLVYGASDSNVYDLLKDLEEGIQTL
MGRLEDGSPRTGQIFKQTYSKFDTNSHNDDALLKNYGLLYCFRKDMDKVETFLRIVQCRS VEGSCGF (SEQ ID NO: 5).
[0178] In one embodiment, the Hgh receptor antagonist has the following mutations: H18D, H21N, F54P, R64K, G120K, L129C, R167N, D171S, E174S, and I179T (“1202-3” or “Variant 3”). The mutation of L129C in Variant 3 allows for a conjugation site for a binder complex. The mutation of G120K in Variant 3 imparts antagonist activity to the composition The mutations H18D, H21N, F54P, R64K, R167N, D171S, E174S, and I179T improve the binding characteristics or increase binding affinity of Variant 3.
[0179] In a related embodiment, the GHR antagonist of Variant 3 has the following mutations compared to Hgh: L129C; H18D; H21N; F54P; R64K; G120K; R167N; D171S; E174S; I179T. In another embodiment, Variant 3 comprises the following amino acid sequence: MFPTIPLSRLFDNAMLRADRLNQLAFDTYQEFEEAYIPKEQKYSFLQNPQTSLCPSESIPTPS NKEETQQKSNLEUJUSUJJQSWLEPVQFLRSVFANSLVYGASDSNVYDLLKDLEEKIQTL MGRLCDGSPRTGQIFKQTYSKFDTNSHNDDALLKNYGLLYCFNKDMSKVSTFLRTVQCR SVEGSCGF (SEQ ID NO: 6).
[0180] In one embodiment, the Hgh receptor antagonist has the following mutations: H18D, H21N, G120K, L129C, R167N, D171S, E174S, and I179T (“1202^” or “Variant 4”). The mutation of L129C in Variant 4 allows for a conjugation site for a binder complex. The mutation of G120K in Variant 4 imparts antagonist activity to the composition. The mutations H18D, H21N, R167N, D171S, E174S, and I179T improve the binding characteristics or increase binding affinity of Variant 4.
[0181] In a related embodiment, the GHR antagonist of Variant 4 has the following mutations compared to Hgh: L129C; H18D; H21N; G120K; R167N; D171S; E174S; I179T. In another embodiment, Variant 4 comprises the following amino add sequence: MFPTIPLSRLFDNAMLRADRLNQLAFDTYQEFEEAYIPKEQKYSFLQNPQTSLCFSESIPTPS NREETQQKSNLELLRISLLUQSWLEPVQFLRSVFANSLVYGASDSNVYDLLKDLEEKIQTL MGRIXTXjSPRTGQIFKQTYSKFDTNSHNDDALLKNYGLLYCFNKDMSKVSTFLRTVQCR SVEGSCGF (SEQ ID NO: 7).
[0182] In one embodiment, the Hgh receptor antagonist has the following mutations: H18D, H21N, G120K, T135C, R167N, D171S, E174S, and I179T (“1202-5” or “Variant 5”). The mutation of T135C in Variant 5 allows for a conjugation site for a binder complex. The mutation of G120K in
Variant 5 imparts antagonist activity to the composition. The mutations H18D, H21N, R167N, D171S, E174S, and I179T improve the binding characteristics or increase binding affinity of Variant
[0183] In a related embodiment, the GHR antagonist of Variant 5 has the following mutations compared to Hgh: T135C; H18D; H21N; G120K; R167N; D171S; E174S; I179T. In another embodiment, Variant 5 comprises the following amino add sequence: MFPTIPLSRLFDNAMLRADRLNQLAFDTYQEFEEAYIPKEQKYSFLQNPQTSLCFSESIPTPS NREETQQKSNLELLRISLLUQSWLEPVQFLRSVFANSLVYGASDSNVYDLLKDLEEKIQTL MGRLEDGSPRCGQIFKQTVSKFDTNSHNDDALLKNYGLLYCFNKDMSKVSTFLRTVQCR SVEGSCGF (SEQ ID NO: 8).
[0184] In one embodiment, the hGH receptor antagonist corresponds to the amino sequence of any of SEQ ID Nos: 5 to 8. In another embodiment, the hGH receptor antagonist corresponds to the amino acid sequence of any of SEQ ID NO: 5 to SEQ ID NO: 8. In another embodiment, the Hgh receptor antagonist corresponds to the amino acid sequence of any of SEQ ID Nos: 5 through 8. In another embodiment, the hGH receptor antagonist corresponds to the amino acid sequence of any of SEQ ID NO: 5 through SEQ ID NO: 8.
[0185] In one embodiment, the hGH receptor antagonist corresponds to the amino sequence of SEQ ID NO: 5. In one embodiment, the hGH receptor antagonist corresponds to the amino sequence of SEQ ID NO: 6. In one embodiment, the hGH receptor antagonist corresponds to the amino sequence of SEQ ID NO: 7. In one embodiment, the hGH receptor antagonist corresponds to the amino sequence of SEQ ID NO: 8.
[0186] In another embodiment, the invention includes a homologue of a growth hormone antagonist In another embodiment, the invention includes a homologue of a human growth hormone antagonist. In another embodiment, homologues e.g., polypeptides which are at least 50%, at least 55%, at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 87%, at least 89%, at least 91%, at least 93%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% homologous to a growth hormone or human growth hormone antagonist as determined using BlastP software of the National Center of Biotechnology Information (NCBI) using default parameters.
[0187] In another embodiment, “signal sequence” and “signal peptide” are used interchangeably herein having all the same qualities and meanings. In another embodiment, “sequence” when in reference to a polynucleotide molecule can refer to a coding portion. In one embodiment, the
enhanced biological activity stems from the longer half-life of the engineered hGHA or acylated hGHA while maintaining at least some biological activity. In another embodiment, the enhanced biological activity stems from enhanced biological activity resulting from the conjugation of the binder complex. In another embodiment, the enhanced biological activity stems from both a longer half-life and from enhanced functionality of the binder complex.
[0188] In one embodiment, the hGHA or acylated hGHA includes a signal peptide. In another embodiment, the hGHA or acylated hGHA does not comprise a signal peptide.
[0189] Acylated Human Growth Hormone Receptor Antagonists
[0190] In one embodiment, the hGH antagonists disclosed herein are modified. In another embodiment, the hGH antagonists disclosed herein are modified to increase albumin binding. In another embodiment, the hGH antagonists disclosed herein are modified to increase their half-life.
[0191] In another embodiment, the hGH antagonists disclosed herein are acylated. In another embodiment, the hGH antagonists disclosed herein are acylated to increase albumin binding. In another embodiment, the hGH antagonists disclosed herein are acylated to increase their half-life.
[0192] The terms acylated human growth hormone receptor antagonist, acylated hGH antagonist, acylated hGHA, acylated human growth hormone receptor antagonist complex, acylated hGH antagonist complex, acylated hGHA complex, modified human growth hormone receptor antagonist, modified hGH antagonist, modified hGHA, modified human growth hormone receptor antagonist complex, modified hGH antagonist complex, and modified hGHA complex are synonymous in the present application.
[0193] In one embodiment, an "acylated" amino add is an amino add comprising an acyl group which is non-native to a naturally occurring amino add, regardless of how it is produced. Methods of produdng acylated amino adds and acylated peptides are known in the art and include acylating an amino acid before inclusion in the peptide or peptide synthesis followed by chemical acylation of the peptide. In another embodiment, the acyl group causes the peptide to have one or more of (i) a prolonged half- life in circulation, (ii) a delayed onset of action, (iii) an improved resistance to proteases, or (iv) an extended duration of action.
[0194] As stated earlier in the disclosure, at least one amino acid of the human growth hormone receptor antagonist or human growth hormone receptor antagonist G120K is mutated to cysteine. In a related embodiment, at least one amino add of the human growth hormone receptor antagonist or
human growth hormone receptor antagonist G120K is mutated to cysteine as a conjugation site for the binder complex. Specifically, at least one amino acid of the human growth hormone receptor antagonist or human growth hormone receptor antagonist G120K is mutated to cysteine and without interrupting the protein-receptor interaction.
[0195] In another embodiment, cysteine is L129 or T135 of the human growth hormone receptor antagonist or human growth hormone receptor antagonist G120K is mutated to cysteine. These cysteine mutations introduce a cysteine as a conjugation site for the binder complex and without interrupting the protein-receptor interaction.
[0196] In another embodiment, cysteine is L129 or T135 of the human growth hormone receptor antagonist or human growth hormone receptor antagonist G120K is mutated to cysteine as a conjugation site for the reversible binder complex and without interrupting the protein-receptor interaction In a related embodiment, the cysteine of the hGH receptor antagonist or the hGH receptor antagonist G120K is attached to the reversible binder complexes 1 or 2 as described in Table 7.
[0197] In another embodiment, cysteine is L129 or T135 of the human growth hormone receptor antagonist or human growth hormone receptor antagonist G120K is mutated to cysteine as a conjugation site for the permanent binder complex and without interrupting the protein-receptor interaction In a related embodiment, the cysteine of the hGH receptor antagonist or the hGH receptor antagonist G120K is attached to the permanent binder complexes A, B, C, or D as described in Table 8.
[0198] The present application provides new compounds which are capable of binding albumin for use in extending the half-life of biologically active moieties to which they are attached. Specifically present application provides new compounds which are capable of binding albumin for use in extending the half-life of hGH antagonists to which they are attached. Thus, the present application provides albumin-binding compounds consisting essentially of the following elements: a binder, a spacer, a linker, and a biologically active moiety.
[0199] The present application provides new compounds which are capable of binding albumin for use in extending the half-life of biologically active moieties to which they are attached. Specifically present application provides new compounds which are capable of binding albumin for use in extending the half-life of hGH antagonists to which they are attached. Thus, the present application provides albumin-binding compounds consisting essentially of the following elements: a binder, a spacer, and a biologically active moiety.
[0200] The present application provides new compounds which are capable of binding albumin for use in extending the half-life of biologically active moieties to which they are attached. Specifically present application provides new compounds which are capable of binding albumin for use in extending the half-life of hGH antagonists to which they are attached. Thus, the present application provides albumin-binding compounds consisting essentially of the following elements: a binder, a spacer, an optional linker, and a biologically active moiety.
[0201] The term “albumin binding moiety” as used herein means a residue which binds non- covalently to human serum albumin. In another embodiment, acylation with fatty acids delays clearance of the peptide it is attached to. In another embodiment, the hGHA is acylated with an acyl group of sufficient size to bind serum albumin.
[0202] In one embodiment, the modified GHR antagonists consist of the following elements: the present application provides albumin-binding compounds consisting essentially of the following elements: a binder, a spacer, a linker, and a GHR antagonist In another embodiment, the modified GHR antagonists consist of the following elements: the present application provides albumin-binding compounds consisting essentially of the following elements: a binder, a spacer, a reversible linker, and a GHR antagonist In another embodiment, the modified GHR antagonists consist of the following elements: the present application provides albumin-binding compounds consisting essentially of the following elements: a binder, a spacer, a non-reversible linker, and a GHR antagonist
[0203] In another embodiment, the modified GHR antagonists consist of the following elements: the present application provides albumin-binding compounds consisting essentially of the following elements: a fatty acid chain, a spacer, a linker, and a GHR antagonist In another embodiment, the modified GHR antagonists consist of the following elements: the present application provides albumin-binding compounds consisting essentially of the following elements: a fatty add chain, a spacer, a reversible linker, and a GHR antagonist In another embodiment, the modified GHR antagonists consist of the following elements: the present application provides albumin-binding compounds consisting essentially of the following elements: a fatty acid chain, a spacer, a non- reversible linker, and a GHR antagonist
[0204] In one embodiment, the modified GHR antagonists consist of the following elements: the present application provides albumin-binding compounds consisting essentially of the following elements: a binder, a spacer, and a GHR antagonist In another embodiment, the modified GHR
antagonists consist of the following elements: the present application provides albumin-binding compounds consisting essentially of the following elements: a fatty acid chain, a spacer, and a GHR antagonist
[0205] In one embodiment, the modified GHR antagonists consist of the following elements: the present application provides albumin-binding compounds consisting essentially of the following elements: a binder, a spacer, an optional linker, and a GHR antagonist In another embodiment, the modified GHR antagonists consist of the following elements: the present application provides albumin-binding compounds consisting essentially of the following elements: a binder, a spacer, an optional reversible linker, and a GHR antagonist. In another embodiment, the modified GHR antagonists consist of the following elements: the present application provides albumin-binding compounds consisting essentially of the following elements: a binder, a spacer, an optional non- reversible linker, and a GHR antagonist
[0206] In another embodiment, the modified GHR antagonists consist of the following elements: the present application provides albumin-binding compounds consisting essentially of the following elements: a fatty acid chain, a spacer, an optional linker, and a GHR antagonist In another embodiment, the modified GHR antagonists consist of the following elements: the present application provides albumin-binding compounds consisting essentially of the following elements: a fatty add drain, a spacer, an optional reversible linker, and a GHR antagonist In another embodiment, the modified GHR antagonists consist of the following elements: the present application provides albumin-binding compounds consisting essentially of the following elements: a fatty add chain, a spacer, an optional non-reversible linker, and a GHR antagonist
[0207] In one embodiment, the present application provides modified GHR antagonists consisting essentially of:
Linker GHR
Binder Spacer
(optional) Antagonist
[0208] In one embodiment, the modified GHR antagonists have the structure of formula (I): W-X-Y-Z (Formula I)
wherein:
W represents a binder;
X represents a spacer;
Y represents an optional linker;
Z represents a human growth hormone receptor antagonist (“GHR antagonist”).
[0209] In one embodiment, the term “binder complex” refers to the binder, spacer, and an optional linker. In a related embodiment, the term “binder complex” refers to W, X, and Y of Formula I. In a related embodiment, the term “binder complex” refers to W and X of Formula I. In another embodiment, the term "binder complex” refers to a fatty add chain, a spacer, and an optional linker. In another embodiment, the term "binder complex” refers to a fatty acid chain and a spacer.
[0210] In one embodiment, the term “binder complex” can be used interchangeably with any of the following terms: “probe”, “albumin binding probe”, or “albumin-binding probe”.
[0211] In one embodiment, the W of Formula I is a fatty add chain. In another embodiment, the binder of Formula I is a fatty acid chain.
[0212] As used herein, the term “fatty add chain” refers to the hydrocarbon backbone of fatty adds (excluding the terminal acidic group) containing 2 to 40 carbon atoms. Fatty acid chain can refer to a ctadecanedioic add (Cl 8 diacid) or eicosanedioic acid (C20 diacid).
[0213] As used herein, the octadecanedioic acid (Cl 8 diacid) fatty add has the following structure of Formula II:
O
OH
HO
O or has the following structure of Formula II-A:
O
HO
O
[0214] As used herein, the eicosanedioic add (C20 diadd) fatty acid has the following structure of Formula HI:
or has the following structure of Formula O-A:
[0215] In one embodiment, the W of Formula I is a hydrocarbon backbone of fatty acids containing 2 to 40 carbon atoms. In another embodiment, the binder of Formula I is a hydrocarbon backbone of fatty acids containing 2 to 40 carbon atoms.
[0216] In one embodiment, the W of Formula I is octadecanedioic add (Cl 8 diacid) or eicosanedioic acid (C20 diadd). In another embodiment, the binder of Formula I is octadecanedioic add (Cl 8 diacid) or eicosanedioic add (C20 diacid).
[0217] In one embodiment, the W of Formula I is octadecanedioic acid (C18 diacid) and is represented by Formula II:
[0218] In another embodiment, the W of Formula I is octadecanedioic acid (Cl 8 diadd) and is represented by Formula II-A:
[0219] In one embodiment, the W of Formula I is dcosanedioic add (C20 diadd) and is represented by Formula HI:
o
OH
HO
0
W « Eicosanedioic acid (C20 diacid).
[0220] In another embodiment, the W of Formula I is eicosanedioic acid (C20 diacid) and is represented by Formula O-A:
O
HO
O
[0221] In one embodiment, the X of Formula I is a spacer.
[0222] In one embodiment, the X of Formula I is any one of gGiu-Giun-(AEEA)m-Cys-Glyp, gdu- Giun-(AEEA)m-Lys(AcBr)-Glyp, or gdu-Giun-(AEEA)m-Lys-Glyp and n is 1, 2, or 3; m is 1, 2, or 3; and p is 1, 2, or 3. In one embodiment, the X of Formula I is any one of gGiu-Giun-(AEEA)m-Cys- dyp, gGiu-Giun-(AEEA)m-Lys(AcBr)-Gly, or gGlu-Giun-(AEEA)m-Lys-Gly and n is 1, 2, or 3; m is 1, 2, or 3; and p is 1, 2, or 3.
[0223] In one embodiment, the X of Formula I is any one of gGlu-Glu-(AEEA)m-Cys-Gly, gGlu- Glu-(AEEA)m-Lys(AcBr)-Gly, or gGlu-Glu-(AEEA)m-Lys-Gly and m is 1, 2, or 3. In another embodiment, the X of Formula I is any one of gdu-du-(AEEA)m-Cys-Gly, gGlu-Glu-(AEEA)m- Lys(AcBr)-Gly, or gGlu-Glu-(AEEA)m-Lys-Gly and m is 1. In another embodiment, the X of Formula I is any one of gGlu-Glu-(AEEA)m-Cys-Gly, gGlu-Glu-(AEEA)m-Lys(AcBr)-Gly, or gGlu- Glu-(AEEA)m-Lys-Gly and m is 2. In another embodiment, the X of Formula I is any one of gGlu- Glu-(AEEA)m-Cys-Gly, gGlu-Glu-(AEEA)m-Lys(AcBr)-Gly, or gGlu-Glu-(AEEA)m-Lys-Gly and m is 3.
[0225] In one embodiment, the X of Formula I is gGlu-Glu-(AEEA)m-Cys-Gly, m is 1, 2, or 3, and is represented by Formula IV.
[0229] In one embodiment, the X of Formula I is represented by Formula IV-D:
[0231] In one embodiment, the X of Formula I is represented by Formula V:
[0232] In one embodiment, the X of Formula I is gdu-Glu-(AEEA)m-Lys(AcBr)-Gly, m is 1, 2, or 3, and is represented by Formula V.
[0234] In one embodiment, the X of Formula I is represented by Formula V-B:
H Br
HQ ,o
H or O
O O H
N -N OH
H o O
HO O n=1»2, 3
[0236] In one embodiment, the X of Formula I is represented by Formula VI:
[0237] In one embodiment, the X of formula I is X is gGlu-Glu-(AEEA)m-Lys-Gly, m is 1, 2, or 3, and is represented by Formula VI.
[0239] In one embodiment, the X of formula I is represented by Formula VI-B:
[0241] In one embodiment, the Y of Formula I is a linker. In a related embodiment, the Y of Formula I is a linker and links W and Z of Formula I.
[0242] In one embodiment, the Y of Formula I is an optional linker.
[0243] In one embodiment, the Y of Formula I is not present In a related embodiment, the Y of Formula I is not present, and Formula I then comprises W-X-Z.
[0244] In one embodiment, the Y of Formula I is Chloropropane-2-one-Fmoc-Mal or Mal-NRFmoc- NHS. In another embodiment, the Y of Formula I is Chloropropane-2-one-Fmoc-Mal. In another embodiment, the Y of Formula I is Mal-NRFmoc-NHS.
[0245] In one embodiment, the Y of F ormula I is 2-(3 -(2, 5-dioxo-2H-pyrrol-l (5H)-yl)propanamido)-
9H-fluoren-9-yl)methyl 3-chloro-2-oxopropylcarbamate.
[0246] In one embodiment, the Y of Formula I is a linker and is a stable covalent bond. In another embodiment, the Y of Formula I is a non-reversible linker. In another embodiment, the bond between the GHR antagonist and the linker of Formula I is a stable covalent bond. In another embodiment, the bond between Z and Y of Formula I is a stable covalent bond.
[0247] In one embodiment, the Y of Formula I is Mal-NRFmoc-NHS and is a stable covalent bond. In another embodiment, the Y of Formula I is Mal-NRFmoc-NHS and is a non-reversible linker. In another embodiment, the bond between the GHR antagonist and Mal-NRFmoc-NHS as the linker of Formula I is a stable covalent bond. In another embodiment, the bond between Z and Mal-NRFmoc- NHS is a stable covalent bond.
[0248] In one embodiment, the Y of Formula I is a linker and is a reversible covalent bond. In another embodiment, the Y of Formula I is a reversible linker. In another embodiment, the bond between the GHR antagonist and the linker of Formula I is a reversible covalent bond. In another embodiment, the bond between Z and Y of Formula I is a reversible covalent bond.
[0249] In one embodiment, the Y of Formula I is Chloropropane-2-one-Fmoc-Mal and is a reversible covalent bond. In another embodiment, the Y of Formula I is Chloropropane-2-one-Fmoc-Mal and is a reversible linker. In another embodiment, the bond between the GHR antagonist and Chloropropane-2-one-Fmoc-Mal is a reversible covalent bond. In another embodiment, the bond between Z and Chloropropane-2-one-Fmoc-Mal is a reversible covalent bond.
[0250] In one embodiment, the Y of Formula I is Chloropropane-2-one-Fmoc-Mal and is represented by Formula VII (in another embodiment the Chloropropane can be substituted with Bromopropane or lodopropane):
[0251] In one embodiment, the Y of formula I is Chloropropane-2-one-Fmoc-Mal and is represented by Formula VII- A:
O o
N H
O o
HN o
O
[0252] In one embodiment, the Y of formula I is Chloropropane-2-one-Fmoc-Mal and is represented by Formula VH-B:
O o jy .
N H o
O
HN o
Q
Cl
[0253] In one embodiment, the Y of formula I is Chloropropane-2-one-Fmoc-Mal and is represented by Formula VH-C:
O o
N H
O
O
HN O
O
[0254] In another embodiment, the Y of Formula I is Chloropropane-2-one-Fmoc-Mal and is represented by Formula VII, and the Chloropropane can be substituted with Bromopropane or lodopropane.
[0255] In another embodiment, the Y of Formula I is Chloropropane-2-one-Fmoc-Mal and the Chloropropane can be substituted with Bromopropane or lodopropane.
[0256] In one embodiment, the Y of Formula I is Mal-Fmoc-Propyl bromide.
[0257] In another embodiment, the Y of Formula I is (2-(3-(2,5-dioxo-2H-pyrrol-l(5H)- yl)propanamido)-9H-fluoren-9-yl) methyl 3-bromo-2-oxopropylcarbamate.
[0258] In one embodiment, the Y of formula I is represented by Formula XXXIV: o o
N h H o o
HN o o.
Br
[0259] In one embodiment, the Y of formula I is Mal-Fmoc-Propyl bromide and is represented by Formula XXXIV.
[0260] In one embodiment, the Y of Formula I is Mal-Fmoc-Propyl Chloride.
[0261] In another embodiment, the Y of Formula I is (2-(3-(2,5-dioxo-2H-pyrrol-l(5H)- yl)propanamido)-9H-fluoren-9-yl) methyl 3-chloro-2-oxopropylcarbamate.
[0262] In one embodiment, the Y of Formula I is represented by Formula XXXV: o
N O H o N
N i o H
Cl
[0263] In one embodiment, the Y of Formula I is Mal-Fmoc-Propyl Chloride and is represented by Formula XXXV.
[0264] In one embodiment, the Y of Formula I is Mal-Fmoc-Propyl Iodide.
[0265] In another embodiment, the Y of Formula I is (2-(3-(2,5-dioxo-2H-pyrrol-l(5H)- yl)propanamido)-9H-fluoren-9-yl)methyl 3 -Iodo-2 -oxopropylcarbamate.
[0266] In one embodiment, the Y of Formula I is represented by Formula XXXVI: o o
N Il H o o
HN o
[0267] In another embodiment, the Y of Formula I is Mal-Fmoc-Propyl Iodide and is represented by Formula XXXVI.
[0268] In one embodiment, the Y of Formula I is Bromopropane -2-one-Fmoc-Mal and is represented by Formula VO: o
N h 4 H o o
HN o o.
Br
[0269] In one embodiment, the Y of Formula I is Iodopropane-2-one-Fmoc-Mal and is represented by Formula IX:
[0270] In one embodiment, the Y of Formula I is Mal-NRFmoc-NHS and is represented by Formula
[0271] In one embodiment, the Y of formula I is represented by Formula X-A:
O o
N H o o O
[0273] In one embodiment, the Y of formula I is represented by Formula X-C:
O o
N H o o O
[0274] In one embodiment, the modified GHR antagonists disclosed throughout the application include pharmaceutically acceptable salts thereof.
[0275] In one embodiment, the GHR antagonists in the modified GHR antagonists disclosed include any the GHR antagonists previously disclosed. In another embodiment, the GHR antagonists in the modified GHR antagonists disclosed include any of SEQ ID NO: 5 through SEQ ID NO: 8.
[0276] In another embodiment, the GHR antagonists in the modified GHR antagonists have an amino acid mutation to cysteine at T3, E39, P48, Q69, N99, G120, L129, T135, T142, H151 or combinations thereof. In another embodiment, the GHR antagonists in the modified GHR antagonists have an amino acid mutation to cysteine is T3, E39, P48, Q69, N99, G120, T142, H151 or combinations thereof. In another embodiment, the GHR antagonists in the modified GHR antagonists have an amino acid mutation to cysteine is L129 or T135 or combinations thereof.
[0277] In one embodiment, the GHR antagonist in the modified GHR antagonists has the following mutations: G120K, L129C, R167N, D171S, E174S, and I179T (“1202-1” or “Variant 1”) or comprises the amino add sequence of SEQ ID NO: 5.
[0278] In one embodiment, the GHR antagonist in the modified GHR antagonists has the following mutations: H18D, H21N, F54P, R64K, G120K, L129C, R167N, D171S, E174S, and I179T (“1202- 3” or “Variant 3 or has the amino add sequence of SEQ ID NO: 6.
[0279] In one embodiment, the GHR antagonist in the modified GHR antagonists has the following mutations: H18D, H21N, G120K, L129C, R167N, D171S, E174S, andI179T (“1202-4” or “Variant 4”) or has the amino add sequence of SEQ ID NO: 7.
[0280] In one embodiment, the GHR antagonist in the modified GHR antagonists has the following mutations: H18D, H21N, G120K, T135C, R167N, D171S, E174S, andI179T (“1202-5” or “Variant 5”) or has the amin acid sequence of SEQ ID NO: 8.
[0281] In one embodiment, the GHR antagonist in the modified GHR antagonist corresponds to the amino sequence of any of SEQ ID Nos: 5 to 8. In another embodiment, the GHR antagonist in the modified GHR antagonist corresponds to the amino acid sequence of any of SEQ ID NO: 5 to SEQ ID NO: 8. In another embodiment, the GHR antagonist in the modified GHR antagonist corresponds to the amino add sequence of any of SEQ ID Nos: 5 through 8. In another embodiment, the GHR antagonist in the modified GHR antagonist corresponds to the amino acid sequence of any of SEQ ID NO: 5 through SEQ ID NO: 8.
[0282] In one embodiment, the GHR antagonist in the modified GHR antagonist corresponds to the amino sequence of SEQ ID NO: 5. In one embodiment, the GHR antagonist in the modified GHR antagonist corresponds to the amino sequence of SEQ ID NO: 6. In one embodiment, the GHR antagonist in the modified GHR antagonist corresponds to the amino sequence of SEQ ID NO: 7. In one embodiment, the GHR antagonist in the modified GHR antagonist corresponds to the amino sequence of SEQ ID NO: 8.
[0283] In one embodiment, the acylated hGHA complex is represented in FIG. 2 where n is 15 and X is 1 or hGHA Variant 1 (SEQ ID NO: 5). In another embodiment, the acylated hGHA complex is represented in FIG. 2 where n is 15 and X is 1. In another embodiment, the acylated hGHA complex is represented in FIG. 2 where n is 15 and X is 1 (hGHA Variant 1). In another embodiment, the acylated hGHA complex is represented in FIG. 2 where n is 15 and X is 1 (SEQ ID NO: 5).
[0284] In one embodiment, the acylated hGHA complex is represented by the following chemical structure:
(Formula XV).
[0285] In one embodiment, the acylated hGHA complex is represented in FIG. 2 where n is 15 and X is 3 or hGHA Variant 3 (SEQ ID NO: 6). In another embodiment, the acylated hGHA complex is represented in FIG. 2 where n is 15 and X is 3. In another embodiment, the acylated hGHA complex is represented in FIG. 2 where n is 15 and X is 3 (hGHA Variant 3). In another embodiment, the acylated hGHA complex is represented in FIG. 2 where n is 15 and X is 3 (SEQ ID NO: 6).
[0286] In one embodiment, the acylated hGHA complex is represented by the following chemical structure:
(Formula XVI).
[0287] In one embodiment, the acylated hGHA complex is represented in FIG. 2 where n is 15 and X is 4 or hGHA Variant 4 (SEQ ID NO: 7). In another embodiment, the acylated hGHA complex is
represented in FIG. 2 where n is 15 and X is 4. In another embodiment, the acylated hGHA complex is represented in FIG. 2 where n is 15 and X is 4 (hGHA Variant 4). In another embodiment, the acylated hGHA complex is represented in FIG. 2 where n is 15 and X is 4 (SEQ ID NO: 7).
[0288] In one embodiment, the acylated hGHA complex is represented by the following chemical structure:
(Formula XVII).
[0289] In one embodiment, the acylated hGHA complex is represented in FIG. 2 where n is 15 and X is 5 or hGHA Variant 5 (SEQ ID NO: 8). In another embodiment, the acylated hGHA complex is represented in FIG. 2 where n is 15 and X is 5. In another embodiment, the acylated hGHA complex is represented in FIG. 2 where n is 15 and X is 5 (hGHA Variant 5). In another embodiment, the acylated hGHA complex is represented in FIG. 2 where n is 15 and X is 5 (SEQ ID NO: 8).
[0290] In one embodiment, the acylated hGHA complex is represented by the following chemical structure:
(Formula XVIII).
[0291] In one embodiment, the acylated hGHA complex is represented in FIG. 2 where n is 17 and X is 1 or hGHA Variant 1 (SEQ ID NO: 5). In another embodiment, the acylated hGHA complex is represented in FIG. 2 where n is 17 and X is 1. In another embodiment, the acylated Hgha complex is represented in FIG. 2 where n is 17 and X is 1 (hGHA Variant 1). In another embodiment, the acylated hGHA complex is represented in FIG. 2 where n is 17 and X is 1 (SEQ ID NO: 5).
[0292] In one embodiment, the acylated hGHA complex is represented by the following chemical structure:
(Formula XIX).
[0293] In one embodiment, the acylated hGHA complex is represented in FIG. 2 where n is 17 and X is 3 or hGHA Variant 3 (SEQ ID NO: 6). In another embodiment, the acylated hGHA complex is
represented in FIG. 2 where n is 17 and X is 3. In another embodiment, the acylated hGHA complex is represented in FIG. 2 where n is 17 and X is 3 (hGHA Variant 3). In another embodiment, the acylated hGHA complex is represented in FIG. 2 where n is 17 and X is 3 (SEQ ID NO: 6).
[0294] In one embodiment, the acylated hGHA complex is represented by the following chemical structure:
(Formula XX).
[0295] In one embodiment, the acylated hGHA complex is represented in FIG. 2 where n is 17 and X is 4 or hGHA Variant 4 (SEQ ID NO: 7). In another embodiment, the acylated hGHA complex is represented in FIG. 2 where n is 17 and X is 4. In another embodiment, the acylated hGHA complex is represented in FIG. 2 where n is 17 and X is 4 (hGHA Variant 4). In another embodiment, the acylated hGHA complex is represented in FIG. 2 where n is 17 and X is 4 (SEQ ID NO: 7).
[0296] In one embodiment, the acylated hGHA complex is represented by the following chemical structure:
(Formula XXI).
[0297] In one embodiment, the acylated hGHA complex is represented in FIG. 2 where n is 17 and X is 5 or hGHA Variant 5 (SEQ ID NO: 8). In another embodiment, the acylated hGHA complex is represented in FIG. 2 where n is 17 and X is 5. In another embodiment, the acylated hGHA complex is represented in FIG. 2 where n is 17 and X is 5 (hGHA Variant 5). In another embodiment, the acylated hGHA complex is represented in FIG. 2 where n is 17 and X is 5 (SEQ ID NO: 8).
[0298] In one embodiment, the acylated hGHA complex is represented by the following chemical structure:
(Formula XXII).
[0299] In one embodiment, the acylated hGHA complex is represented in FIG. 3 where n is 15 and X is 1 or hGHA Variant 1 (SEQ ID NO: 5). In another embodiment, the acylated hGHA complex is
represented in FIG. 3 where n is 15 and X is 1. In another embodiment, the acylated hGHA complex is represented in FIG. 3 where n is 15 and X is 1 (hGHA Variant 1). In another embodiment, the acylated hGHA complex is represented in FIG. 3 where n is 15 and X is 1 (SEQ ID NO: 5).
[0300] In one embodiment, the acylated hGHA complex is represented by the following chemical structure:
(Formula XXIII).
[0301] In one embodiment, the acylated hGHA complex is represented in FIG. 3 where n is 15 and X is 3 or hGHA Variant 3 (SEQ ID NO: 6). In another embodiment, the acylated hGHA complex is represented in FIG. 3 where n is 15 and X is 3. In another embodiment, the acylated hGHA complex is represented in FIG. 3 where n is 15 and X is 3 (hGHA Variant 3). In another embodiment, the acylated hGHA complex is represented in FIG. 3 where n is 15 and X is 3 (SEQ ID NO: 6).
[0302] In one embodiment, the acylated hGHA complex is represented by the following chemical structure:
Q
(Formula XXIV).
[0303] In one embodiment, the acylated hGHA complex is represented in FIG. 3 where n is 15 and X is 4 or hGHA Variant 4 (SEQ ID NO: 7). In another embodiment, the acylated hGHA complex is represented in FIG. 3 where n is 15 and X is 4. In another embodiment, the acylated hGHA complex is represented in FIG. 3 where n is 15 and X is 4 (hGHA Variant 4). In another embodiment, the acylated hGHA complex is represented in FIG. 3 where n is 15 and X is 4 (SEQ ID NO: 7).
[0304] In one embodiment, the acylated hGHA complex is represented by the following chemical structure:
(Formula XXV).
[0305] In one embodiment, the acylated hGHA complex is represented in FIG. 3 where n is 15 and
X is 5 or hGHA Variant 5 (SEQ ID NO: 8). In another embodiment, the acylated hGHA complex is represented in FIG. 3 where n is 15 and X is 5. In another embodiment, the acylated hGHA complex is represented in FIG. 3 where n is 15 and X is 5 (hGHA Variant 5). In another embodiment, the acylated hGHA complex is represented in FIG. 3 where n is 15 and X is 5 (SEQ ID NO: 8).
[0306] In one embodiment, the acylated hGHA complex is represented by the following chemical structure:
(Formula XXVI).
[0307] In one embodiment, the acylated hGHA complex is represented in FIG. 3 where n is 17 and X is 1 or hGHA Variant 1 (SEQ ID NO: 5). In another embodiment, the acylated hGHA complex is represented in FIG. 3 where n is 17 and X is 1. In another embodiment, the acylated hGHA complex is represented in FIG. 3 where n is 17 and X is 1 (hGHA Variant 1). In another embodiment, the acylated hGHA complex is represented in FIG. 3 where n is 17 and X is 1 (SEQ ID NO: 5).
[0308] In one embodiment, the acylated hGHA complex is represented by the following chemical structure:
(Formula XXVII).
[0309] In one embodiment, the acylated hGHA complex is represented in FIG. 3 where n is 17 and X is 3 or hGHA Variant 3 (SEQ ID NO: 6). In another embodiment, the acylated hGHA complex is represented in FIG. 3 where n is 17 and X is 3. In another embodiment, the acylated hGHA complex is represented in FIG. 3 where n is 17 and X is 3 (hGHA Variant 3). In another embodiment, the acylated hGHA complex is represented in FIG. 3 where n is 17 and X is 3 (SEQ ID NO: 6).
[0310] In one embodiment, the acylated hGHA complex is represented by the following chemical structure:
(Formula XXVIII).
[0311] In one embodiment, the acylated hGHA complex is represented in FIG. 3 where n is 17 and
X is 4 or hGHA Variant 4 (SEQ ID NO: 7). In another embodiment, the acylated hGHA complex is represented in FIG. 3 where n is 17 and X is 4. In another embodiment, the acylated hGHA complex is represented in FIG. 3 where n is 17 and X is 4 (hGHA Variant 4). In another embodiment, the acylated hGHA complex is represented in FIG. 3 where n is 17 and X is 4 (SEQ ID NO: 7).
[0312] In one embodiment, the acylated hGHA complex is represented by the following chemical structure:
(Formula XXIX).
[0313] In one embodiment, the acylated hGHA complex is represented in FIG. 3 where n is 17 and X is 5 or hGHA Variant 5 (SEQ ID NO: 8). In another embodiment, the acylated hGHA complex is represented in FIG. 3 where n is 17 and X is 5. In another embodiment, the acylated hGHA complex is represented in FIG. 3 where n is 17 and X is 5 (hGHA Variant 5). In another embodiment, the acylated hGHA complex is represented in FIG. 3 where n is 17 and X is 5 (SEQ ID NO: 8).
[0314] In one embodiment, the acylated hGHA complex is represented by the following chemical structure:
(Formula XXX).
[0315] In another embodiment, the GHR antagonists in the modified GHR antagonists include a homologue of a growth hormone antagonist In another embodiment, the GHR antagonists in the modified GHR antagonists includes a homologue of a human growth hormone antagonist In another embodiment, homologues e.g., polypeptides which are at least 50%, at least 55%, at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 87%, at least 89%, at least 91%, at least 93%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% homologous to a growth hormone or human growth hormone antagonist as determined using BlastP software of the National Center of Biotechnology Information (NCBI) using default parameters.
[0316] Methods of Use
[0317] The disorders or diseases which can be treated by the method of the present invention include any disease that is exacerbated by the action of an agonist on GHRs can be treated by the present invention. In one embodiment of the present invention, the disorder is acromegaly or gigantism. Specifically, the invention includes methods for treating acromegaly and/or giantism by administering to patients having such disorders a therapeutically effective amount of a growth hormone antagonist together with a pharmaceutically acceptable carrier to reduce a pathological effect or symptom of acromegaly and/or giantism and, in particular, to lower levels of IGF-1 in plasma and/or tissue.
[0318] In another embodiment, the disease is diabetes mellitus (DM). More specifically, the method of the present invention is used to prevent or reduce proliferative diseases associated with diabetes such as diabetic retinopathy and glomerulosclerosis in patients with DM In particular, such methods
include administering a growth hormone-antagonist in a therapeutically effective amount to reduce a pathological effect or symptom of diabetes, such as nephropathy or retinopathy, and/or to lower blood glucose levels.
[0319] In another aspect, described herein is a method of treating, preventing, or ameliorating an hGH-related disorder, the method comprising: administering to a subject in need thereof an effective amount of an hGH antagonist or modified GHR antagonist disclosed herein or a variant thereof to treat, prevent or ameliorate an hGH -associated disorder in the subject
[0320] In certain embodiments, the hGH-related disorder is one or more of: acromegaly; age-related macular degeneration; cancer; diabetes mellitus; gigantism, vascular eye diseases including diabetic retinopathy, retinopathy of prematurity, and sickle cell anemia retinopathy; nephropathy, diabetes- induced nephropathy (glomerulosclerosis), neurodegeneration and cancer.
[0321] In another aspect, there is provided a method of treating a variety of diseases involving the production of excess hGH, wherein the method comprises the step of administering an effective amount of an hGH antagonist or modified GHR antagonist disclosed herein. In particular, the methods include the treatment of acromegaly, gigantism, cancer, diabetes, vascular eye diseases (diabetic retinopathy, retinopathy of prematurity, age-related macular degeneration, sickle cell anemia retinopathy, and the tike), as well as nephropathy and neurodegeneration
[0322] In another aspect, there is described herein a method of inhibiting human growth hormone (hGH) protein action in a subject, the method comprising: administering to a subject an effective amount of an hGH antagonist or modified GHR antagonist disclosed herein, or a variant thereof, to inhibit an hGH protein in the subject
[0323] Diseases that may be treated by the methods of the present application are diseases characterized by one or more of the following criteria: elevated levels of hGH production, elevated levels of serum hGH, elevated levels of hGHR production, and elevated cellular response of hGHRs to hGH
[0324] The term “elevated” as used herein is used with respect to the normal levels of hGH production, hGHR production, or hGH-mediated cellular response in a tissue (or tissues) of a diseased person (or animal) as compared to level in a normal individual. Diseases that may be treated by the methods of the invention include, but are not limited to, acromegaly, gigantism, cancer, diabetes, vascular eye diseases (diabetic retinopathy, retinopathy of prematurity, age-related macular degeneration, retinopathy of sickle-cell anemia, etc.) as well as nephropathy.
[0325] In another aspect, there is described herein method of treating cancers in a subject In another aspect, there is described herein method of treating cancers in a subject by administering an effective amount of an hGH antagonist or modified GHR antagonist disclosed herein. In particular, the methods include the treatment of cancers comprising tumor cells that express hGHRs.
[0326] In another aspect, there is described herein method of treating cardiac cancers in a subject by administering an effective amount of an hGH antagonist or modified GHR antagonist disclosed herein. In particular, the methods include the treatment of sarcoma (angiosarcoma, fibrosarcoma, rhabdomyosarcoma, liposarcoma), myxoma, rhabdomyoma, fibroma, lipoma and teratoma.
[0327] In another aspect, there is described herein method of treating lung cancers in a subject by administering an effective amount of an hGH antagonist or modified GHR antagonist disclosed herein. In particular, the methods include the treatment of bronchogenic carcinoma (squamous cell, undifferentiated small cell, undifferentiated large cell, adenocarcinoma), alveolar (bronchiolar) carcinoma, bronchial adenoma, sarcoma, lymphoma, chondromatous hamartoma, and mesothelioma. [0328] In another aspect, there is described herein method of treating gastrointestinal cancers in a subject by administering an effective amount of an hGH antagonist or modified GHR antagonist disclosed herein. In particular, the methods include the treatment of esophagus (squamous cell carcinoma, adenocarcinoma, leiomyosarcoma, lymphoma), stomach (carcinoma, lymphoma, leiomyosarcoma), pancreas (ductal adenocarcinoma, insulinoma, glucagonoma, gastrinoma, carcinoid tumors, vipoma), small bowel (adenocarcinoma, lymphoma, carcinoid tumors, Karposi's sarcoma, leiomyoma, hemangioma, lipoma, neurofibroma, fibroma), large bowel (adenocarcinoma, tubular adenoma, villous adenoma, hamartoma, and leiomyoma.
[0329] In another aspect, there is described herein method of treating genitourinary tract cancers in a subject by administering an effective amount of an Hgh antagonist or modified GHR antagonist disclosed herein In particular, the methods include the treatment of kidney (adenocarcinoma, Wilm's tumor [nephroblastoma], lymphoma, leukemia), bladder and urethra (squamous cell carcinoma, transitional cell carcinoma, adenocarcinoma), prostate (adenocarcinoma, sarcoma), testis (seminoma, teratoma, embryonal carcinoma, teratocarcinoma, choriocarcinoma, sarcoma, interstitial cell carcinoma, fibroma, fibroadenoma, adenomatoid tumors, and lipoma.
[0330] In another aspect, there is described herein method of treating Ever cancers in a subject by administering an effective amount of an hGH antagonist or modified GHR antagonist disclosed herein. In particular, the methods include the treatment of hepatoma (hepatocellular carcinoma),
cholangiocarcinoma, hepatoblastoma, angiosarcoma, hepatocellular adenoma, hemangioma.
[0331] In another aspect, there is described herein method of treating bone cancers in a subject by administering an effective amount of an hGH antagonist or modified GHR antagonist disclosed herein. In particular, the methods include the treatment of osteogenic sarcoma (osteosarcoma), fibrosarcoma, malignant fibrous histiocytoma, chondrosarcoma, Ewing's sarcoma, malignant lymphoma (reticulum cell sarcoma), multiple myeloma, malignant giant cell tumor, chordoma, osteochronfroma (osteocartilaginous exostoses), benign chondroma, chondroblastoma, chondromyxofibroma, osteoid osteoma and giant cell tumors.
[0332] In another aspect, there is described herein method of treating a cancer of the nervous system in a subject by administering an effective amount of an hGH antagonist or modified GHR antagonist disclosed herein. In particular, the methods include the treatment of skull (osteoma, hemangioma, granuloma, xanthoma, osteitis deformans), meninges (meningioma, meningiosarcoma, gliomatosis), brain (astrocytoma, medulloblastoma, glioma, ependymoma, germinoma [pinealoma], glioblastoma multiforme, oligodendroglioma, schwannoma, retinoblastoma, congenital tumors), spinal cord (neurofibroma, meningioma, glioma, sarcoma.
[0333] In another aspect, there is described herein method of treating gynecological cancer in a subject by administering an effective amount of an hGH antagonist or modified GHR antagonist disclosed herein. In particular, the methods include the treatment of uterus (endometrial carcinoma), cervix (cervical carcinoma, pre-tumor cervical dysplasia), ovaries (ovarian carcinoma [serous cystadenocarcinoma, mucinous cystadenocarcinoma, endometrioid tumors, celioblastoma, clear cell carcinoma, unclassified carcinoma], granulosa-thecal cell tumors, Sertoli-Leydig cell tumors, dysgerminoma, malignant teratoma), vulva (squamous cell carcinoma, intraepithelial carcinoma, adenocarcinoma, fibrosarcoma, melanoma), vagina (clear cell carcinoma, squamous cell carcinoma, botryoid sarcoma [embryonal rhabdomyosarcoma], and fallopian tubes (carcinoma).
[0334] In another aspect, there is described herein method of treating gynecological cancer in a subject by administering an effective amount of an hGH antagonist or modified GHR antagonist disclosed herein. In particular, the methods include the treatment of uterus (endometrial carcinoma), cervix (cervical carcinoma, pre-tumor cervical dysplasia), ovaries (ovarian carcinoma [serous cystadenocarcinoma, mucinous cystadenocarcinoma, endometrioid tumors, celioblastoma, clear cell carcinoma, unclassified carcinoma], granulosa-thecal cell tumors, Sertoli-Leydig cell tumors, dysgerminoma, malignant teratoma), vulva (squamous cell carcinoma, intraepithelial carcinoma,
adenocarcinoma, fibrosarcoma, melanoma), vagina (clear cell carcinoma, squamous cell carcinoma, botryoid sarcoma [embryonal rhabdomyosarcoma], and fallopian tubes (carcinoma).
[0335] In another aspect, there is described herein a method of treating hematologic cancer in a subject by administering an effective amount of an hGH antagonist or modified GHR antagonist disclosed herein. In particular, the methods include the treatment of blood (myeloid leukemia [acute and chronic], acute lymphoblastic leukemia, chronic lymphocytic leukemia, myeloproliferative diseases, multiple myeloma, myelodysplastic syndrome), Hodgkin's disease, and non-Hodgkin's lymphoma [malignant lymphoma].
[0336] In another aspect, there is described herein a method of treating skin cancer in a subject by administering an effective amount of an hGH antagonist or modified GHR antagonist disclosed herein. In particular, the methods include the treatment of malignant melanoma, basal cell carcinoma, squamous cell carcinoma, Karposi's sarcoma, moles, dysplastic nevi, lipoma, angioma, dermatofibroma, keloids, and psoriasis.
[0337] In another aspect, there is described herein a method of treating cancer of the adrenal glands in a subject by administering an effective amount of an hGH antagonist or modified GHR antagonist disclosed herein. In particular, the methods include the treatment of neuroblastoma.
[0338] In another aspect, there is described herein a method of treating breast cancers, colon cancers, colorectal cancers, or thyroid cancers in a subject by administering an effective amount of an hGH antagonist or modified GHR antagonist disclosed herein.
[0339] Methods of Producing
[0340] In one embodiment, there is described herein a method of producing an albumin binding complex on a resin, the steps comprising: a. Synthesizing a protected binder backbone of Formula XXXI on a resin
HNH
HO 0
Or
O O
H
NH N o H o IB OH o
HO “O n - 15,17 i « 1,2,3
(Formula XXXI); b. Removing the methyltrityl (Mtt) resulting in a deprotected binder backbone; c. Coupling a linking moiety to the deprotected binder backbone resulting in an albumin binding peptide complex; d. Cleaving the albumin binding peptide complex from the resin; e. Purifying the albumin binding peptide complex; and f. Lyophilizing the albumin binding peptide complex.
[0341] In one embodiment, the resin used in the methods of producing an albumin binding complex is a Wang resin.
[0342] In one embodiment, the synthesizing of a binder backbone is done using solid-phase peptide synthesis (SPPS).
[0343] In another embodiment, the binder backbone is tBu-Fatty acid-gGiu(OtBu)-Giu(OtBu)- (AEEA)i-Lys(Mtt)-Gly-Wang resin, wherein i is 2 or 3 and the Fatty acid is eicosanedioic or octadecandioic.
[0344] In another embodiment, in step b. of the method of producing the albumin binding complex, the removing is done by applying 1%TFA in DCM wash cycles. In a related embodiment, this removing step deprotects the Lys side chain.
[0345] In one embodiment, the intermediate composition following step (b) is the tBu-eiocosanedioc- yGlu(C)tBu)-Glu(C)tBu)-(AEEA)3-Lys-Gly-Wang resin, tBu-eiocosanedioc-yGlu(OtBu)-Glu(OtBu)- (AEEA)2-Lys-Gly-Wang resin, tBu-octadecandioic-yGlu(C)tBu)-Glu(C)tBu)-(AEEA)3-Lys-Gly- Wang resin, or tBu-octadecandioic-yGlu(C)tBu)-Glu(C)tBu)-(AEEA)2-Lys-Gly-Wang resin.
[0346] In one embodiment, in step c. of the method of producing the albumin binding complex, the linker moiety and deprotected binder backbone are added to a reactor and stirred. In a related
embodiment, the linker moiety is coupled to the primary amine of the deprotected Lys side chain of the peptide backbone.
[0347] In one embodiment, in step c. of the method of producing the albumin binding complex, the linking moiety is MAL-NRFmoc. In another embodiment, in step c. of the method of producing the albumin binding complex, the linking moiety is Acetyl-Br.
[0348] In one embodiment, in step c. of the method of producing the albumin binding complex, about 2-3eq of the linking moiety is added to the reactor. In a related embodiment, in step c. of the method of producing the albumin binding complex, about 2-3eq of the MAL-NRFmoc is added to the reactor. [0349] In one embodiment, in step c. of the method of producing the albumin binding complex, 2- 3eq of the linking moiety is added to the reactor. In a related embodiment, in step c. of the method of producing the albumin binding complex, 2-3eq of the MAL-NRFmoc is added to the reactor.
[0350] In one embodiment, in step c. of the method of producing the albumin binding complex, about l-6eq of the linking moiety is added to the reactor. In a related embodiment, in step c. of the method of producing the albumin binding complex, about 1 -6eq of the MAL-NRFmoc is added to the reactor. [0351] In one embodiment, about 20eq of Bromoacetic anhydride is dissolved in DCM, and 20eq of DIPEA is added to the solution resulting in a Bromoacetic anhydride solution ready for Lys side chain conjugation. In another embodiment, between about lOeq to 30 eq of Bromoacetic anhydride is dissolved in DCM, and about 10 eq to 30eq of DIPEA is added to the solution resulting in a Bromoacetic anhydride solution ready for Lys side chain conjugation.
[0352] In one embodiment, the Bromoacetic anhydride solution is added to the reactor and stirred.
[0353] In one embodiment, the stirring in the reactor comprises about 1 hour. In a related embodiment, the stirring in the reactor comprises between about 30 minutes to about 2 hours.
[0354] In one embodiment, pH in the reactor is about 5.0. In another embodiment, pH in the reactor is between about 4.0 to 6.0.
[0355] In another embodiment, in step d. of the method of producing the albumin binding complex, the cleaving is done with a cocktail containing 95%TFA: 5% H2O, with a ratio of 10ml cleavage cocktail: lgr peptide-resin. In a related embodiment, in step d. of the method of producing the albumin binding complex, the cleaving is done with a cocktail containing about 95%TFA: 5% H2O, with a ratio of about 10ml cleavage cocktail: lgr peptide-resin. In a related embodiment, in step d. of the method of producing the albumin binding complex, the cleaving is done with a cocktail containing between about 85%TFA: 15% H2O to 99%TFA: 1% H2O. In a related embodiment, in step d. of the
method of producing the albumin binding complex, the cleaving is done with a cocktail with a ratio of between about 20ml cleavage cocktail: lgr peptide-resin to about lml cleavage cocktail: lgr peptide-resin.
[0356] In another embodiment, in step e. of the method of producing the albumin binding complex, the purifying is done on RP-HPLC column.
[0357] In one embodiment, the albumin binding complex is Formula XXXII. In another embodiment, the albumin binding complex is Formula XXXHI.
[0358] In one embodiment, the albumin binding complex is tBu-eicosanedioic-yGlu(C)tBu)- Glu(OtBu)-(AEEA)3-Lys(Ac-Br)-Gly-Wang resin, tBu-eicosanedioic-yGlu(OtBu)-Glu(OtBu)- (AEEA)2-Lys(Ac-Br)-Gly-Wang, resin tBu-octadecandioic-yGlu(C)tBu)-Glu(C)tBu)-(AEEA)3- Lys(Ac-Br)-Gly-Wang resin, or tBu-octadecandioic-yGlu(C)tBu)-Glu(C)tBu)-(AEEA)2-Lys(Ac-Br)- Gly-Wang resin. In another embodiment, the albumin binding complex is tBu-eicosanedioic- yGlu(OtBu)-Glu(OtBu)-(AEEA)3-Lys(MAL-NRFmoc)-Gly-Wang resin, tBu-eicosanedioic- yGlu(OtBu)-Glu(OtBu)-(AEEA)2-Lys(MAL-NRFmoc)-Gly-Wang resin, tBu-octadecandioic- ydu(C)tBu)-Glu(OtBu)-(AEEA)3-Lys(MAL-NRFmoc)-Gly-Wang resin, or tBu-octadecandioic- yGlu(C)tBu)-Glu(OtBu)-(AEEA)2-Lys(MAL-NRFmoc)-Gly-Wang resin.
[0359] In one embodiment, disclosed herein are albumin binding complexes including Formula xxxn andXXXm.
[0360] In one embodiment, disclosed herein is a method of conjugating an albumin binding complex to an hGH antagonist by adding the binding complex to the hGHA in solution. In a related embodiment, the binder complex is added at about 1.25 eq. In a related embodiment, the binder complex is added at between .75 eq and 1.75 eq. In one embodiment, the hGHA is added at about 1 eq. In another embodiment, the hGHA is added at about .25 eq to about 1.75 eq.
[0361] In another embodiment, the solution conjugating an albumin binding complex is kept at room temperature.
[0362] In one embodiment, once the conjugation of the albumin binding complex is complete, the solution is quenched. In a related embodiment, the quenching solution is lOOmM L-Cys at a pH of 7. In another embodiment, the quenching solution is about lOOmM L-Cys. In another embodiment, the quenching solution is between 75mM and 125 mM L-Cys. In one embodiment, the quenching solution is at a pH of about 7. In another embodiment, the quenching solution is at a pH of between 6 and 8. In another embodiment, 1 volume unit of the quenching solution is added to 100 volume units
of the reaction solution
[0363] The embodiments may further be described using the following clauses:
1. A human growth hormone receptor antagonist (“GHR antagonist9 ’ or “hGHA"), comprising growth hormone receptor antagonist G120K (SEQ ID NO: 2) and at least one of the following amino acid mutations: H18D, H21N, F54P, R64K, R167N, D171S, E174S, I179T.
2. The GHR antagonist of claim 1, wherein any of said mutations increase the affinity of the GHR antagonist to the growth hormone receptor.
3. The GHR antagonist of any one of claims 1 to 2, further comprising at least one amino acid mutation to cysteine.
4. The GHR antagonist of claim 3, wherein the amino acid mutated to cysteine is T3, E39, P48, Q69, N99, L129, T135, T142, H151 or combinations thereof.
The GHR antagonist of claim 3, wherein the amino acid mutated to cysteine is LI 29 or T135.
6. The GHR antagonist of claim 1, wherein the GHR antagonist has the following amino acid mutations: G120K, L129C, R167N, D171S, E174S, andI179T.
7. The GHR antagonist of claim 6, wherein the GHR antagonist comprises the amino acid sequence of SEQ ID NO: 5.
8. The GHR antagonist of claim 1, wherein the GHR antagonist has the following amino acid mutations: H18D, H21N, F54P, R64K, G120K, L129C, R167N, D171S, E174S, and I179T.
9. The GHR antagonist of claim 8, wherein the GHR antagonist comprises the amino acid sequence of SEQ ID NO: 6.
10. The GHR antagonist of claim 1, wherein the GHR antagonist has the following amino acid mutations: H18D, H21N, G120K, L129C, R167N, D171S, E174S, and I179T.
11. The GHR antagonist of claim 10, wherein the GHR antagonist comprises the amino acid sequence of SEQ ID NO: 7.
12. The GHR antagonist of claim 1, wherein the GHR antagonist has the following amino acid mutations: H18D, H21N, G120K, T135C, R167N, D171S, E174S, andI179T.
13. The GHR antagonist of claim 12, wherein the GHR antagonist comprises the amino acid sequence of SEQ ID NO: 8.
14. The GHR antagonist of claim 1, wherein the GHR antagonist comprises the amino acid sequence of any one of SEQ ID NOs: 5 to 8.
15. A modified human growth hormone receptor antagonist (“modified GHR antagonist” or “modified hGHA”) having the structure of formula (I):
W-X-Y-Z (I) wherein:
W represents a binder;
X represents a spacer;
Y represents an optional linker;
Z represents a human growth hormone receptor antagonist (“GHR antagonist”); or a pharmaceutically acceptable salt thereof.
16. The modified GHR antagonist of claim 15, wherein the GHR antagonist comprises any of one of claims 1 to 14.
17. The modified GHR antagonist of claim 15, wherein said GHR antagonist comprises the amino acid sequence of any of SEQ ID NOs: 5 to 8.
18. The modified GHR antagonist of any one of claims 15 to 17, wherein W is a fatty acid.
19. The modified GHR antagonist of any one of claims 15 to 18, wherein W is octadecanedioic acid (Cl 8 diacid) or is eicosanedioic acid (C20 diacid).
20. The modified GHR antagonist of any one of claims 15 to 18, wherein W is octadecanedioic acid (Cl 8 diacid) and is represented by Formula II:
O
OH
HO
O or is represented by Formula II-A:
O
HO
O
21. The modified GHR antagonist of any one of claims 15 to 18, wherein W is eicosanedioic acid (C20 diacid) and is represented by Formula HI:
o
OH
HO
O or is represented by Formula IH-A: o
HO o
22. The modified GHR antagonist of any one of claims 15 to 21, wherein X is gGlu-Glun- (AEEA)m-Cys-Glyp, gGlu-Glun-(AEEA)m-Lys(AcBr)-Glyp, or gGlu-Glun-(AEEA)m-Lys- Glyp and n is 1, 2, or 3; m is 1, 2, or 3; and p is 1, 2, or 3.
23. The modified GHR antagonist of any one of claims 15 to 21, wherein X is gGlu-Glu- (AEEA)m-Cys-Gly, m is 1, 2, or 3, and is represented by Formula IV:
n» 1. 2, 3 or is represented by Formula IV-E;
n»1,2,3
24. The modified GHR antagonist of any one of claims 15 to 21, wherein X is gGlu-Glu- (AEEA)m-Lys(AcBr)-Gly, m is 1, 2, or 3, and is represented by Formula V:
n=1,2, 3 or is represented by Formula V-C:
25. The modified GHR antagonist of any one of claims 15 to 21, wherein X is gGlu-Glu- (AEEA)m-Lys-Gly, m is 1, 2, or 3, and is represented by Formula VI:
n« 1, 2, 3 or is represented by Formula VI-C:
26. The modified GHR antagonist of any one of claims 15 to 25, wherein ¥ is Chloropropane- 2-one-Fmoc-Mal.
27. The modified GHR antagonist of claim 26, wherein Chloropropane-2-one-Fmoc-Mal is represented by Formula VII:
or is represented by Formula VII-C:
O
N H o o
HN o o,
28. The modified GHR antagonist of any one of claims 15 to 25, wherein Y is Mal-NRFmoc- NHS.
29. The modified GHR antagonist of claim 28, wherein Mal-NRFmoc-NHS is represented by Formula X: o
O
N
H o
O o i
O N or is represented by Formula X-C:
o o
N H o o o
30. The modified GHR antagonist of any one of claims 28 to 29, wherein the bond between the GHR antagonist and the linker is a stable covalent bond.
31. The modified GHR antagonist of any one of claims 26 to 27, wherein the bond between the GHR antagonist and the linker is a reversible covalent bond.
32. The modified GHR antagonist of any one of claims 15-31, wherein the modified GHR antagonist comprises the following formula:
(Formula XV).
33. The modified GHR antagonist of claim 15, wherein the modified GHR antagonist comprises the following formula:
■o hGHA Variant 3 ^EQID NO: 6)
(Formula XVI).
34. The modified GHR antagonist of claim 15, wherein the modified GHR antagonist comprises the following formula: n
OJ
HN
O«=(
V .o
3 " 8 "1
■O hGHA Variant 4 (SEQ ID NO: 7)
(Formula XVII).
35. The modified GHR antagonist of claim 15, wherein the modified GHR antagonist comprises the following formula:
(Formula XVIII).
36. The modified GHR antagonist of claim 15, wherein the modified GHR antagonist comprises the following formula:
Oj </
HR
GSaZ
HO. .O o
M *«
O.H O’ ‘S-
HO^O hGHA Variant ^EQID NO: 5)
(Formula XIX).
37. The modified GHR antagonist of claim 15, wherein the modified GHR antagonist comprises the following formula:
o, *7 V
O$af *
9
OH O’ d
.. 1..
HO^O hGHA Variant -
'CEQ ID NO: 6)
(Formula XX).
38. The modified GHR antagonist of claim 15, wherein the modified GHR antagonist comprises the following formula:
hG (SEQ ID NO
(Formula XXI).
39. The modified GHR antagonist of claim 15, wherein the modified GHR antagonist comprises the following formula:
o. toll o
‘o'
■o QA
(Formula XXII).
40. The modified GHR antagonist of claim 15, wherein the modified GHR antagonist comprises the following formula:
(F ormula XXIII).
41. The modified GHR antagonist of claim 15, wherein the modified GHR antagonist comprises the following formula:
(Formula XXIV).
42. The modified GHR antagonist of claim 15, wherein the modified GHR antagonist comprises the following formula:
(Formula XXV).
43. The modified GHR antagonist of claim 15, wherein the modified GHR antagonist comprises the following formula:
(Formula XXVI)
44. The modified GHR antagonist of claim 15, wherein the modified GHR antagonist comprises the following formula:
(Formula XXVII).
45. The modified GHR antagonist of claim 15, wherein the modified GHR antagonist comprises the following formula:
(Formula XXVIII).
46. The modified GHR antagonist of claim 15, wherein the modified GHR antagonist comprises the following formula:
(F ormula XXIX).
47. The modified GHR antagonist of claim 15, wherein the modified GHR antagonist comprises the following formula:
(Formula XXX).
48. A method of inhibiting human growth hormone (hGH) action in a subject, the method comprising: administering to a subject in need thereof an effective amount of the GHR antagonist of any one of claims 1 to 14 or the modified GHR antagonist of any one of claims 15 to 47, to inhibit hGH action in the subject.
49. The method of claim 48, wherein the method comprises administering to the subject said GHR antagonist or the modified GHR antagonist in an amount effective to treat, prevent, or ameliorate a hGH related disorder in the subject
50. The method of claim 49, wherein the hGH related disorder is one or more of: acromegaly; age-related macular degeneration; cancer; diabetes; gigantism, vascular eye diseases, diabetic retinopathy, retinopathy of prematurity, and retinopathy of sickle-cell anemia; and, nephropathy.
51. A method of treating, preventing, or ameliorating a hGH related disorder, the method comprising: administering to a subject in need thereof an effective amount of the GHR antagonist of any one of claims 1 to 14 or the modified GHR antagonist of any one of claims 15 to 47, to treat, prevent, or ameliorate a hGH related disorder in the subject.
52. A method of producing an albumin binding complex on a resin, the steps comprising:
(a) Synthesizing a protected binder backbone of Formula XXXI on a resin
HNH
Or
O O
H
NH N o OH
O
HO o n - 15,17 i « 1,2,3
(Formula XXXI);
(b) Removing the methyltrityl (Mtt) resulting in a deprotected binder backbone;
(c) Coupling a linking moiety to the deprotected binder backbone resulting in an albumin binding peptide complex;
(d) Cleaving the albumin binding peptide complex from the resin;
(e) Purifying the albumin binding peptide complex; and
(f) Lyophilizing the albumin binding peptide complex.
53. The method of claim 52, wherein said synthesizing of a binder backbone is done using solid-phase peptide synthesis (SPPS).
54. The method of claim 52 or 53, wherein said binder backbone is tBu-Fatty acid- gGhi(OtBu)-Ghi(OtBu)-(AEEA)i-Lys(Mtt)-Gly-Wang resin, wherein i is 2 or 3 and the Fatty acid is eicosanedioic or octadecandioic.
55. The method of any one of claims 52-54, wherein said removing in step (b) is done by applying 1%TFA in DCM wash cycles.
56. The method of any one of claims 52-55, wherein said removing in step (b) deprotects the Lys side chain.
57. The method of any one of claims 52-56, wherein the intermediate composition following step (b) is the tBu-eiocosanedioc-yGlu(OtBu)-Glu(OtBu)-(AEEA)3-Lys-Gly-Wang resin, tBu-eiocosanedioc-yGlu(OtBu)-Glu(OtBu)-(AEEA)2-Lys-Gly-Wang resin, tBu- octadecandioic-yGlu(OtBu)-Glu(OtBu)-(AEEA)3-Lys-Gly-Wang resin, or tBu- octadecandioic-yGlu(OtBu)-Glu(OtBu)-(AEEA)2-Lys-Gly-Wang resin.
58. The method of any one of claims 52-57, wherein the linker moiety and deprotected binder backbone are added to a reactor and stirred.
59. The method of any one of claims 52-58, wherein the linker moiety is coupled to the primary amine of the deprotected Lys side chain of the peptide backbone.
60. The method of any one of claims 52-59, wherein said resin is Wang resin.
61. The method of any one of claims 52-60, wherein said cleaving in step (d) is done with a cocktail containing 95%TFA: 5% H2O, with a ratio of 10ml cleavage cocktail: lgr peptide-resin.
62. The method of any one of claims 52-61, wherein said purifying in step (e) is done on RP- HPLC column.
63. The method of any one of claims 52-62, wherein said linking moiety in step (c) is MAL- NRFmoc.
64. The method of claim 63, wherein around 2-3eq of the MAL-NRFmoc is added to the reactor.
65. The method of any one of claims 63-64, wherein the pH in the reactor is around 5.0.
66. The method of any one of claims 52-62, wherein said linking moiety in step (c) is Acetyl- Br.
67. The method of claim 66, wherein around 20eq of Bromoacetic anhydride is dissolved in DCM, and 20eq of DIPEA is added to the solution resulting in a Bromoacetic anhydride solution ready for Lys side chain conjugation.
68. The method of claim 67, wherein said Bromoacetic anhydride solution is added to the reactor and stirred.
69. The method of claim 68, wherein said stirring comprises 1 hour.
70. The method of any one of claims 52-62 or 66-69, wherein the albumin binding complex is Formula XXXII:
Q
HN Br
HO.
H Or
O O
HO N H
NH N
H 'N OH
O O
O
HO O n * 15,17 i ® 1.2,3
(Formula XXXII).
71. The method of any one of claims 52-62 or 66-69, wherein the albumin binding complex is tBu-eicosanedioic-yGlu(OtBu)-Glu(OtBu)-(AEEA)3-Lys(Ac-Br)-Gly-Wang resin, tBu- eicosanedioic-yGlu(OtBu)-Glu(OtBu)-(AEEA)2-Lys(Ac-Br)-Gly-Wang, resin tBu- octadecandioic-yGlu(OtBu)-Glu(OtBu)-(AEEA)3-Lys(Ac-Br)-Gly-Wang resin, or tBu- octadecandioic-yGlu(OtBu)-Glu(OtBu)-(AEEA)2-Lys(Ac-Br)-Gly-Wang resin.
72. The method of any one of claims 52-65, wherein the albumin binding complex is Formula XXXHI:
fi « 15.17 1,2,3
(Formula XXXIII).
73. The method of any of claims 52-65, wherein the albumin binding complex is tBu- eicosanedioic-yGlu(OtBu)-Glu(OtBu)-(AEEA)3-Lys(MAL-NRFmoc)-Gly-Wang resin, tBu-eicosanedioic-yGlu(OtBu)-Glu(OtBu)-(AEEA)2-Lys(MAL-NRFmoc)-Gly-Wang resin, tBu-octadecandioic-yGlu(OtBu)-Glu(OtBu)-(AEEA)3-Lys(MAL-NRFmoc)-Gly- Wang resin, or tBu-octadecandioic-yGlu(OtBu)-Glu(OtBu)-(AEEA)2-Lys(MAL- NRFmoc)-Gly-Wang resin.
74. An albumin binding complex comprising Formula XXXII:
Q
HN Br
O
O O
H
NH
N OH
O H
O
HO O n » 15,17 i ® 1,2,3
(Formula XXXII).
75. An albumin binding complex comprising tBu-eicosanedioic-yGlu(OtBu)-Glu(OtBu)- (AEEA)3-Lys(Ac-Br)-Gly-Wang resin, tBu-eicosanedioic-yGlu(OtBu)-Glu(OtBu)- (AEEA)2-Lys(Ac-Br)-Gly-Wang resin, tBu-octadecandioic-yGlu(OtBu)-Glu(OtBu)- (AEEA)3-Lys(Ac-Br)-Gly-Wang resin, or tBu-octadecandioic-yGlu(OtBu)-Glu(OtBu)- (AEEA)2-Lys(Ac-Br)-Gly-Wang resin.
76. A method of conjugating an albumin binding complex of any one of claims 74-75 to an hGH antagonist by adding said binding complex to said hGHA in solution
77. The method of claim 76, wherein said hGH antagonist is any one of claims 1-14.
78. The method of any one of claims 76-77, wherein said binder complex is 3eq.
79. The method of any one of claims 76-78, wherein said hGHA is leq.
80. The method of any one of claims 76-79, wherein the solution was kept at room temperature.
81. The method of any one of claims 76-80, wherein once the conjugation is completed the solution is quenched.
82. The method of claim 81, wherein said quenching solution is lOOmM L-Cys at a pH of 7.
83. The method of any one of claims 81-82, wherein 1 volume unit of the quenching solution is added to 20 volume units of the reaction solution.
1,2.3
(Formula XXXHI).
85. An albumin binding complex comprising tBu-eicosanedioic-yGlu(OtBu)-Glu(OtBu)- (AEEA)s-Lys(MAL-NRFmoc)-Gly-Wang resin, tBu-eicosanedioic-yGlu(OtBu)- Glu(OtBu)-(AEEA)2-Lys(MAL-NRFmoc)-Gly-Wang resin, tBu-octadecandioic- yGlu(OtBu)-Glu(OtBu)-(AEEA)3-Lys(MAL-NRFmoc)-Gly-Wang resin, or tBu- octadecandioic-yGlu(OtBu)-Glu(OtBu)-(AEEA)2-Lys(MAL-NRFmoc)-Gly-Wang resin.
86. A method of conjugating an albumin binding complex of any one of claims 84-85 to an hGH antagonist by adding said binding complex to said hGHA in solution
87. The method of claim 86, wherein said hGH antagonist is any one of claims 1-14.
88. The method of any one of claims 86-87, wherein said binder complex is 1.25eq.
89. The method of any one of claims 86-88, wherein said hGHA is leq.
90. The method of any one of claims 86-89, wherein the solution was kept at room temperature.
91. The method of any one of claims 86-90, wherein once the conjugation is completed the solution is quenched.
92. The method of claim 91, wherein said quenching solution is lOOmM L-Cys at a pH of 7.
93. The method of any one of claims 91-92, wherein 1 volume unit of the quenching solution is added to 100 volume units of the reaction solution.
[0364] Definitions
[0365] As used herein, the singular form "a", "an" and "the" include plural references unless the context clearly dictates otherwise. For example, the term "a compound" or "at least one compound" may include a plurality of compounds, including mixtures thereof.
[0366] A “non-naturally encoded amino add” refers to an amino acid that is not one of the common amino acids or pyrrolysine or selenocysteine. Other terms that may be used synonymously with the term “non-naturally encoded amino add” are “non-natural amino acid,” “unnatural amino acid,” “non-naturally-occurring amino add,” and variously hyphenated and non-hyphenated versions thereof. The term “non-naturally encoded amino acid” also includes, but is not limited to, amino acids that occur by modification (e.g. post-translational modifications) of a naturally encoded amino acid (including but not limited to, the 20 common amino acids or pyrrolysine and selenocystdne) but are not themselves naturally incorporated into a growing polypeptide chain by the translation complex. Examples of such non-naturally-occurring amino adds include, but are not limited to, N- acetylglucosaminyl-L-serine, N-acetylglucosaminyl-L-threonine, and O-phosphotyrosine.
[0367] The term “modified,” as used herein refers to any changes made to a given polypeptide, such as changes to the length of the polypeptide, the amino acid sequence, chemical structure, co- translational modification, or post-translational modification of a polypeptide. The form “(modified)” term means that the polypeptides being discussed are optionally modified, that is, the polypeptides under discussion can be modified or unmodified.
[0368] Throughout this application, various embodiments of this invention may be presented in a range format It should be understood that the description in range format is merely for convenience and brevity and should not be construed as an inflexible limitation on the scope of the invention Accordingly, the description of a range should be considered to have specifically disclosed all the possible sub ranges as well as individual numerical values within that range. For example, description of a range such as from 1 to 6 should be considered to have specifically disclosed sub ranges such as from 1 to 3, from 1 to 4, from 1 to 5, from 2 to 4, from 2 to 6, from 3 to 6 etc., as well as individual numbers within that range, for example, 1, 2, 3, 4, 5, and 6. This applies regardless of the breadth of the range.
[0369] Whenever a numerical range is indicated herein, it is meant to include any cited numeral (fractional or integral) within the indicated range. The phrases “ranging/ranges between” a first indicate number and a second indicate number and “ranging/ranges from” a first indicate number “to” a second indicate number are used herein interchangeably and are meant to include the first and second indicated numbers and all the fractional and integral numerals there between
[0370] A skilled artisan would appreciate that the term “about”, may encompass a deviance of between 0.0001-5% from the indicated number or range of numbers. In some instances, the term “about”, may encompass a deviance of between 1 -10% from the indicated number or range of numbers. In some instances, the term “about”, encompasses a deviance of up to 25% from the indicated number or range of numbers.
[0371] The terms “comprise,” “have” and “include” are open-ended linking verbs. Any forms or tenses of one or more of these verbs, such as “comprises,” “comprising,” “has,” “having,” “includes” and “including,” are also open-ended. For example, any method that “comprises,” “has” or “includes” one or more steps is not limited to possessing only those one or more steps and also covers other unlisted steps.
[0372] The term “effective,” as that term is used in the specification and/or claims, means adequate to accomplish a desired, expected, or intended result “Effective amount,” “Therapeutically effective amount”, or “pharmaceutically effective amount’ when used in the context of treating a patient or subject with a compound means that amount of the compound which, when administered to a subject or patient for treating or preventing a disease, is an amount sufficient to effect such treatment or prevention of the disease.
[0373] An “excipient” is a pharmaceutically acceptable substance formulated along with the active ingredients) of a medication, pharmaceutical composition, formulation, or drug delivery system. Excipients may be used, for example, to stabilize the composition, to bulk up the composition (thus often referred to as “bulking agents,” “fillers,” or “diluents” when used for this purpose), or to confer a therapeutic enhancement on the active ingredient in the final dosage form, such as facilitating drug absorption, reducing viscosity, or enhancing solubility. Excipients include pharmaceutically acceptable versions of antiadherents, binders, coatings, colors, disintegrants, flavors, glidants, lubricants, preservatives, sorbents, sweeteners, and vehicles. The main excipient that serves as a medium for conveying the active ingredient is usually called the vehicle. Excipients may also be used in the manufacturing process, for example, to aid in the handling of the active substance, such as by
fadlitating powder flowability or non-stick properties, in addition to aiding in vitro stability such as prevention of denaturation or aggregation over the expected shelf life. The suitability of an excipient will typically vary depending on the route of administration, the dosage form, the active ingredient, as well as other factors.
[0374] As used herein, the term “patienf ’ or “subject” refers to a living mammalian organism, such as a human, monkey, cow, horse, sheep, goat, dog, cat, mouse, rat, guinea pig, or transgenic species thereof. In certain embodiments, the patient or subject is a primate. Non-limiting examples of human patients are adults, juveniles, infants and fetuses.
[0375] As generally used herein “pharmaceutically acceptable” refers to those compounds, materials, compositions, and/or dosage forms which are, within the scope of sound medical judgment, suitable for use in contact with the tissues, organs, and/or bodily fluids of human beings and animals without excessive toxicity, irritation, allergic response, or other problems or complications commensurate with a reasonable benefit/risk ratio.
[0376] “Pharmaceutically acceptable salts” means salts of compounds of the present invention which are pharmaceutically acceptable, as defined above, and which possess the desired pharmacological activity. Non-limiting examples of such salts include add addition salts formed with inorganic acids such as hydrochloric acid, hydrobromic add, sulfuric acid, nitric add, and phosphoric add; or with organic adds such as 1,2-ethanedisulfonic acid, 2 -hydroxyethanesulfonic acid, 2-naphthalenesulfonic acid, 3-phenylpropionic acid, 4, 4'-methylenebis(3-hydroxy-2-ene-l -carboxylic acid), 4- methylbicyclo [2.2.2]oct-2-ene-l -carboxylic acid, acetic acid, aliphatic mono- and dicarboxylic adds, aliphatic sulfuric adds, aromatic sulfuric adds, benzenesulfonic acid, benzoic acid, camphorsulfonic acid, carbonic add, cinnamic add, dtric add, cyclopentanepropionic add, ethanesulfonic add, fumaric acid, glucoheptonic add, gluconic acid, glutamic acid, glycolic acid, heptanoic acid, hexanoic acid, hydroxynaphthoic add, lactic acid, laurylsulfuric acid, maldc acid, malic add, malonic add, mandelic add, methanesulfonic acid, muconic acid, o-(4-hydroxybenzoyl)benzoic add, oxalic add, p-chlorobenzenesulfonic acid, phenyl-substituted alkanoic adds, propionic add, p-toluenesulfonic acid, pyruvic add, salicylic acid, stearic acid, succinic add, tartaric add, tertiarybutylacetic acid, and trimethylacetic add. Pharmaceutically acceptable salts also include base addition salts which may be formed when addic protons present are capable of reacting with inorganic or organic bases. Acceptable inorganic bases include sodium hydroxide, sodium carbonate, potassium hydroxide, aluminum hydroxide and calcium hydroxide. Non-limiting examples of acceptable organic bases
include ethanolamine, diethanolamine, triethanolamine, tromethamine, and N-methylglucamine. It should be recognized that the particular anion or cation forming a part of any salt of this invention is not critical, so long as the salt, as a whole, is pharmacologically acceptable. Additional examples of pharmaceutically acceptable salts and their methods of preparation and use are presented in Handbook of Pharmaceutical Salts: Properties, and Use (P. H Stahl & C. G. Wermuth eds., Verlag Helvetica Chimica Acta, 2002).
[0377] A “pharmaceutically acceptable carrier,” “drug carrier,” or simply “carrier” is a pharmaceutically acceptable substance formulated along with the active ingredient medication that is involved in carrying, delivering and/or transporting a chemical agent Drug carriers may be used to improve the delivery and the effectiveness of drugs, including for example, controlled-release technology to modulate drug bioavailability, decrease drug metabolism, and/or reduce drug toxicity. Some drug carriers may increase the effectiveness of drug delivery to the specific target sites. Examples of carriers include: liposomes, microspheres (e.g., made of poly(lactic-co-glycolic) add), albumin microspheres, synthetic polymers, nanofibers, protein-DNA complexes, protein conjugates, erythrocytes, virosomes, and dendrimers.
[0378] A “pharmaceutical drug” (also referred to as a pharmaceutical, pharmaceutical agent, pharmaceutical preparation, pharmaceutical composition, pharmaceutical formulation, pharmaceutical product, medicinal product, medicine, medication, medicament, or simply a drug) is a drug used to diagnose, cure, treat, or prevent disease. An active ingredient (Al) (defined above) is the ingredient in a pharmaceutical drug or a pesticide that is biologically adive. The similar terms active pharmaceutical ingredient (API) and bulk adive are also used in medicine, and the term active substance may be used for pesticide formulations. Some medications and pesticide products may contain more than one active ingredient In contrast with the active ingredients, the inactive ingredients are usually called excipients (defined above) in pharmaceutical contexts.
[0379] A “stereoisomer” or “optical isomer” is an isomer of a given compound in which the same atoms are bonded to the same other atoms, but where the configuration of those atoms in three dimensions differs. “Enantiomers” are stereoisomers of a given compound that are mirror images of each other, like left and right hands. “Diastereomers” are stereoisomers of a given compound that are not enantiomers. Chiral molecules contain a chiral center, also referred to as a stereocenter or stereogenic center, which is any point, though not necessarily an atom, in a molecule bearing groups such that an interchanging of any two groups leads to a stereoisomer. In organic compounds, the chiral
center is typically a carbon, phosphorus or sulfur atom, though it is also possible for other atoms to be stereocenters in organic and inorganic compounds. A molecule can have multiple stereocenters, giving it many stereoisomers. In compounds whose stereoisomerism is due to tetrahedral stereogenic centers (e.g., tetrahedral carbon), the total number of hypothetically possible stereoisomers will not exceed 2n, where n is the number of tetrahedral stereocenters. Molecules with symmetry frequently have fewer than the maximinn possible number of stereoisomers. A 50:50 mixture of enantiomers is referred to as a racemic mixture. Alternatively, a mixture of enantiomers can be enantiomerically enriched so that one enantiomer is present in an amount greater than 50%. Typically, enantiomers and/or diastereomers can be resolved or separated using techniques known in the art It is contemplated that that for any stereocenter or axis of chirality for which stereochemistry has not been defined, that stereocenter or axis of chirality can be present in its R form, S form, or as a mixture of the R and S forms, including racemic and non-racemic mixtures. As used herein, the phrase “substantially free from other stereoisomers” means that the composition contains <15%, more preferably <10%, even more preferably <5%, or most preferably <1% of another stereoisomers).
[0380] “Prevention” or “preventing” includes: (1) inhibiting the onset of a disease in a subject or patient which may be at risk and/or predisposed to the disease but does not yet experience or display any or all of the pathology or symptomatology of the disease, and/or (2) slowing the onset of the pathology or symptomatology of a disease in a subject or patient which may be at risk and/or predisposed to the disease but does not yet experience or display any or all of the pathology or symptomatology of the disease.
[0381] “Treatment’ ’ or “treating” includes (1) inhibiting a disease in a subject or patient experiencing or displaying the pathology or symptomatology of the disease (e.g., arresting further development of the pathology and/or symptomatology), (2) ameliorating a disease in a subject or patient that is experiencing or displaying the pathology or symptomatology of the disease (e.g., reversing the pathology and/or symptomatology), and/or (3) effecting any measurable decrease in a disease in a subject or patient that is experiencing or displaying the pathology or symptomatology of the disease.
EXAMPLES
EXAMPLE 1
[0382] DESIGN OF ACYLATED HUMAN GROWTH HORMONE ANTAGONIST COMPLEXES
[0383] The goal of the present experiment is to design an albumin binding moiety of an hGH
antagonist This is designed to increase the longevity of the hGH antagonist Three principles were prioritized when designing a long-acting prodrug employing an albumin-binding probe. The first principle is that following its conjugation, the probe introduced into the drug should have sufficient affinity to albumin to manifest prolonged action in vivo.
[0384] Second, the conjugate’ s activity with respect to binding and properly activating the drug target receptor should be assessed. There are two approaches for probe conjugation that can be applied: (I) Covalent binding to the Albumin probe results in an inactive product; The latter should undergo slow reactivation at physiological conditions by applying reversible Fmoc linkers resulting in probe hydrolysis and release of the drug; (II) Conjugate retains its activity (to appropriate extend); the probe is not interfering with the drug activity, and its hydrolysis is not mandatory.
[0385] Lastly, most importantly, and many times overlooked, is the ability of the Albumin/conjugate complex to bind the FcRn receptor, that sequentially enables the recycle mechanism. The principles listed above were highlighted and considered to be the most important aspect during the designing the different albumin binding moieties disclosed below.
[0386] The general design of these newly developed acylated hGH antagonist utilizing an albumin binding technology to increase the longevity of the hormone is shown below. This design includes the protein design, hGHA, and the binder complex design, which is the binder, spacer, and optional
Linker GHR
Binder Spacer (optional) Antagonist linker.
[0387] The acylated hGH antagonists discussed in this example are also referred to as MOD -1202, long-acting human growth hormone (hGH) antagonists, modified hGH antagonists or modified human growth hormone receptor (GHR) antagonists.
[0388] The designed acylated GHR antagonists differ from each other in the following levels: protein (different mutations); linker (reversible, irreversible, or not present); spacer (differ in length); binders (different in fatty acid length).
[0389] Protein Design
[0390] A human growth hormone antagonist was selected as the target protein. In order to enhance the potential albumin binding characteristics of the hGH antagonist, several point mutations were used. A single mutation of G120K compared to native hGH was selected to convert hGH to an hGH antagonist
[0391 ] The mutation of either L129C or T135C was selected to introduce a cysteine as a conjugation site for the binder complex. The choice of these specific mutations was based on the hGH Receptor crystal structure (PDB code: 3HHR), and aimed to find an optional area for linker binding without interrupting the protein-receptor interaction, and while considering the amino acid conservation score. Outward-feeing amino acids were prioritized. The N-terminus and C-terminus hGH protein were evaluated as well for potential albumin linker binding sites, but in both cases, it seems that a large moiety such as albumin may cause structural interruptions to hGH antagonist protein and/or disrupt binding to the hGHR
[0392] Several point mutations were designed to increase the affinity of the molecule to the GH receptor. (Lowman HB and Wells JAin 1993). These point mutations vary between 4 and 8 mutations and increase binding affinity.
[0393] Based on the principles and strategies above, the following hGH antagonist polypeptide variants were designed and constructed
[0394] Variant 1: hGHL129C; G120K; R167N; D171S; E174S; I179T (SEQID NO: 5). TheL129C mutation introduced a cysteine as a conjugation site. The G120K mutation imparts antagonistic properties. 4 point mutations improve binding.
[0395] Variant 3: hGH L129C; H18D; H21N; F54P; R64K; G120K; R167N; D171S; E174S; I179T (SEQ ID NO: 6). The L129C mutation introduced a cysteine as a conjugation site. The G120K mutation imparts antagonistic properties. 8 point mutations improve binding.
[0396] Variant 4: hGHL129C; H18D; H21N; G120K; R167N; D171S; E174S; I179T (SEQ ID NO:
7). The L129C mutation introduced a cysteine as a conjugation site. The G120K mutation imparts antagonistic properties. 6 point mutations improve binding.
[0397] Variant 5: hGH T135C; H18D; H21N; G120K; R167N; D171S; E174S; I179T (SEQ ID NO:
8). The T135C mutation introduced a cysteine as a conjugation site. The G120K mutation imparts antagonistic properties. 6 point mutations improve binding.
[0398] Binder Complex
[0399] In addition to finding the hGH mutant variants, the inventors also found that there were
multiple design considerations to produce a pharmaceutically effective, long acting hGH antagonist First, a long fatty add chain was selected in order to effectively bind to a large albumin binding protein. Secondly, a diadd form of the fatty acid was found to be very important for the binding affinity to albumin Thirdly, the linker moiety was discovered to be important for the overall properties of the claimed molecules.
[0400] The modified GHR antagonists should minimally contain a single Cys or equivalent nucleophilic amino acid side chain reactant located at an odd numbered amino add position on the polypeptide to serve as an albumin-binder conjugation site. In one embodiment, the modified GHR antagonists have a Cys mutation located at either one of the following positions: 129 or 135. In these preferred positions, the Cys sulfur nucleophile is accessible to read with or couple to an albumin binding moiety. According to the structural analysis of the protein obtained via crystallography methodologies (data not shown), the proposed positions for albumin-binders coupling were the least probable to interfere with receptor binding.
[0401] For the binder part, fatty acid drains of various lengths were considered, including short, medium, and long fatty add chains. C2 to C30 fatty adds were considered. For example, the binder can be any of a C2 fatty acid, C4 fatty acid, C6 fatty add, C8 fatty acid, Cl 0 fatty acid, C12 fatty acid, C14 fatty add, C16 fatty acid, C18 fatty acid, C20 fatty add, C22 fatty acid, C24 fatty acid, C26 fatty acid, C28 fatty acid, or C30 fatty add. Three sizes of fatty add chains were recommended as candidates, C16, C18 and C20, as they were found to be the best analogues in different targets.
[0402] Additionally, two techniques for coupling Albumin binder to the hGH antagonist were developed: (1) reversible coupling, and (2) permanent coupling. For reversible coupling, also described as reversible acylation: the carbamate is hydrolyzed in the presence of water-based media at neutral-slight basic pHs (see formula XI & XII below). For permanent coupling, also described as permanent acylation: either MAL group or Br as a leaving group can serve for covalent reaction with free thiols (Michael addition, and Sn2 reactions, respectively). Formulas XHI and XIV exemplify the thioether bonds formed following the stable coupling using the MAL and Ac-Br functional groups.
[0403] In both the reversible and permanent linking/ coupling, the site of attachment to the hGH antagonist is designed to be either mutated amino acids T135C or E129C. Based on the overall design principles discussed above, a modulated Albumin-binder/spacer complex was developed in order to obtain both Albumin binding and drug potency. The full modulated Albumin binder/spacer/linker matrix is detailed in Tables 2-6.
Table 2: Full Matrix of hGH antagonist Binder-linker Complexes Designs Binding Type _ Binder Spacer Linker
Octadecanedioic
Reversible gGlu-Gliin-(AEEA)n-Cys-Glyn Mal-Fmoc-Propyl Bromide acid (Cl 8 diacid)
Cys (129 or Eicosanedioic acid 135) gGlu-Glun-(AEEA)n-Cys-Glyn Mal-Fmoc -Propyl Bromide (C20 diacid) _
Octadecanedioic gGlu-Glun-(AEEA)n-Lys Mal-NRFmoc-NHS acid (Cl 8 diacid) Eicosanedioic acid
Permanent gGlu-Glun-(AEEA)n-Lys Mal-NRFmoc-NHS (C20 diacid) _ Cys (129 or
Octadecanedioic gGlu-Glun-(AEEA)n-Lys(AcBr)- 135) N/A acid (Cl 8 diacid) Gly Eicosanedioic acid gGlu-Glun-(AEEA)n-Lys(AcBr)-
N/A (C20 diacid) Gly n=l, 2, or 3
Table 3: Acylated Hgh Antagonist Complexes (Binder Type 1202 A or “Type A”) hGHA Variant Acylated hGHA
Binder Spacer Linker _ Complex Name
Octadecanedioic Variant 1 (SEQ 1A gGlu-Glu-(AEEA)3-Lys Mal-NRFmoc-NHS acid (Cl 8 diacid) ID NO: 5) (or “1202-1 A) Octadecanedioic Variant 3 (SEQ 3A gGlu-Glu-(AEEA)3-Lys Mal-NRFmoc-NHS acid (Cl 8 diacid) ID NO: 6) (or 1202-3A) Octadecanedioic Variant 4 (SEQ 4A acid (Cl 8 diacid) ID NO: 7) (or 1202 -4 A) Octadecanedioic Variant 5 (SEQ 5A gGlu-Glu-(AEEA)3-Lys Mal-NRFmoc-NHS acid (Cl 8 diacid) ID NO: 8) (or 1202-5 A)
Binding Type for all Complexes above is Permanent at Cys Position 129 or 135 of the hGHA Variant
Table 4: Acylated Hgh Antagonist Complexes (Binder Type 1202 B or “Type B”)
Binder Spacer Linker
Complex Name
Eicosanedioic acid Variant 1 (SEQ IB gGlu-Glu-(AEEA)3-Lys Mal-NRFmoc-NHS (C20 diacid) ID NO: 5) (or 1202-1B)
Eicosanedioic acid Variant 3 (SEQ 3B gGlu-Glu-(AEEA)3-Lys Mal-NRFmoc-NHS (C20 diacid) ID NO: 6) (or 1202-3B)
Eicosanedioic acid Variant 4 (SEQ 4B (C20 diacid) ID NO: 7) (or 1202-4B)
Eicosanedioic acid Variant 5 (SEQ 5B gGlu-Glu-(AEEA)3-Lys Mal-NRFmoc-NHS (C20 diacid) ID NO: 8) (or 1202-5B)
Binding Type for all Complexes above is Permanent at Cys Position 129 or 135 of the hGHA Variant
Table 5: Acylated Hgh Antagonist Complexes (Binder Type 1202 C or “Type C”)
Hgha Variant Acylated Hgha
Binder Spacer Linker Complex Name
Octadecanedioic gGlu-Glu3-(AEEA)3- Variant 1 (SEQ 1C
None acid (Cl 8 diacid) Lys(AcBr)-Gly ID NO: 5) (or 1202-1C)
Octadecanedioic gGlu-Glu3-(AEEA)3- Variant 3 (SEQ 3C
None acid (Cl 8 diacid) Lys(AcBr)-Gly ID NO: 6) (or 1202-3C)
Octadecanedioic gGlu-Glu3-(AEEA)3- Variant 4 (SEQ 4C
None acid (Cl 8 diacid) Lys(AcBr)-Gly ID NO: 7) (or 1202-4C)
Octadecanedioic gGlu-Glu3-(AEEA)3- Variant 5 (SEQ 5C
None acid (Cl 8 diacid) Lys(AcBr)-Gly ID NO: 8) (or 1202-5C)
Binding Type for all Complexes above is Permanent at Cys Position 129 or 135 of the hGHA Variant
Table 6: Acylated Hgh Antagonist Complexes (Binder Type 1202 D or “Type D”) hGHA Variant Acylated hGHA
Binder Spacer Linker _ Complex Name
Eicosanedioic acid gGlu-Glu3-(AEEA)3- Variant 1 (SEQ ID
None (C20 diacid) _ Lys(AcBr)-Gly ID NO: 5) (or 1202-1D)
Eicosanedioic acid gGlu-Glu3-(AEEA)3- Variant 3 (SEQ 3D
None (C20 diacid) _ Lys(AcBr)-Gly ID NO: 6) (or 1202-3D)
Eicosanedioic acid gGlu-Glu3-(AEEA)3- Variant 4 (SEQ 4D
None (C20 diacid) _ Lys(AcBr)-Gly ID NO: 7) (or 1202-4D)
Eicosanedioic acid gGlu-Glu3-(AEEA)3- Variant 5 (SEQ 5D
None (C20 diacid) Lys(AcBr)-Gly ID NO: 8) (or 1202-5D)
Variant
[0404] The chemical formula for the acylated hGHA complex ID (1202-1D) described above in Table 6 is shown in the following formula.
(Formula XXVII)
[0405] In an embodiment, reversible albumin binding constructs having the designated binders, spacers and linkers or reactants thereof are shown in Table 7.
Table 7: Reversible Binder-linker Complexes Design-hGH
Complex Binder Spacer Linker _ 1 Eicosanedioic acid (C20 diacid) gGu-Glu-(AEEA)2- Mal-Fmoc-Propyl 2 Octadecanedioic acid (Cl 8 diacid) Cys-Gly bromide
Or Mal-Fmoc-Propyl Chloride Or
Mal-Fmoc-Propyl Iodide
All binders are conjugated to Cysteine at position 129 or 135
[0406] The chemical formula for complex 1 described above is represented in formula XI below.
Variant
(Formula XI)
[0407] The chemical formula for complex 2 described above is represented in formula XII below.
/j
HN-
MH
HO o
0 «i
'o hGHA Variant
(Formula XII)
[0408] After following the design principles stated above, the following permanent albumin binding constructs were selected for further study and are shown in Table 8.
Table 8
Complex Binder Spacer Linker Type A Octadecanedioic acid gGlu-Glu-(AEEA)3-Lys-Gly-OH Mal-NRFmoc-NHS (Cl 8 diacid)
B Eicosanedioic acid gGlu-Glu-(AEEA)3-Lys-Gly-OH Mal-NRFmoc-NHS (C20 diacid)
C Octadecanedioic acid gGlu-Glu-(AEEA)3-Lys(AcBr) - N/A (Cl 8 diacid) Gly-OH _
D Eicosanedioic acid gGlu-Glu-(AEEA)3-Lys(AcBr) - N/A (C20 diacid) Gly-OH
[0409] The chemical formula for a generic acylated hGHA with binder type D described above is represented in formula XHI below.
(Formula XIII)
[0410] The chemical formula for a generic acylated hGHA with binder type B described above is represented in formula XIV below.
(Formula XIV)
[0411] The chemical formula for a generic acylated hGHA with binder complex type A or type B described in Table 8 is represented in FIG. 2.
[0412] The acylated hGHA complex 1A (also “1202-1 A”) is represented in FIG. 2 where n is 15 and X is 1 or hGHA Variant 1 (SEQ ID NO: 5). The acylated hGHA complex 1A (also “1202-1A”) is also represented by the following chemical structure:
(Formula XV).
[0413] The acylated hGHA complex 3A (also “1202-3 A”) is represented in FIG. 2 where n is 15 and X is 3 or hGHA Variant 3 (SEQ ID NO: 6). The acylated hGHA complex 3A (also “1202-3A”) is also represented by the following chemical structure:
(Formula XVI).
[0414] The acylated hGHA complex 4A (also “1202-4A”) is represented in FIG. 2 where n is 15 and X is 4 or hGHA Variant 4 (SEQ ID NO: 7). The acylated hGHA complex 4A (also “1202-4A”) is also represented by the following chemical structure:
(Formula XVII).
[0415] The acylated hGHA complex 5A (also “1202-5 A”) is represented in FIG. 2 where n is 15 and X is 5 or hGHA Variant 5 (SEQ ID NO: 8). The acylated hGHA complex 5 A (also “1202-5 A”) is also represented by the following chemical structure:
(Formula XVm).
[0416] The acylated hGHA complex IB (also “1202-1B”) is represented in FIG. 2 where n is 17 and X is 1 or hGHA Variant 1 (SEQ ID NO: 5). The acylated hGHA complex IB (also “1202-1B”) is also represented by the following chemical structure:
(Formula XIX).
[0417] The acylated hGHA complex 3B (also “1202-3B”) is represented in FIG. 2 where n is 17 and X is 3 or hGHA Variant 3 (SEQ ID NO: 6). The acylated hGHA complex 3B (also “1202-3B”) is also represented by the following chemical structure:
(Formula XX).
[0418] The acylated hGHA complex 4B (also “1202-4B”) is represented in FIG. 2 where n is 17 and X is 4 or hGHA Variant 4 (SEQ ID NO: 7). The acylated hGHA complex 4B (also “1202^8”) is also represented by the following chemical structure:
(Formula XXI).
[0419] The acylated hGHA complex 5B (also “1202-5B”) is represented in FIG. 2 where n is 17 and X is 5 or hGHA Variant 5 (SEQ ID NO: 8). The acylated hGHA complex 5B (also “1202-5B”) is also represented by the following chemical structure:
(Formula XXII).
[0420] The chemical formula for a generic acylated hGHA with binder complex type C or type D described in Table 8 is represented in FIG. 3.
[0421] The acylated hGHA complex 1C (also “1202-1 C”) is represented in FIG. 3 where n is 15 and X is 1 or hGHA Variant 1 (SEQ ID NO: 5). The acylated hGHA complex 1C (also “1202-1 C”) is also represented by the following chemical structure:
(Formula XXHI).
[0422] The acylated hGHA complex 3C (also “1202-3C”) is represented in FIG. 3 where n is 15 and X is 3 or hGHA Variant 3 (SEQ ID NO: 6). The acylated hGHA complex 3C (also “1202-3C”) is also represented by the following chemical structure:
Q
(Formula XXIV).
[0423] The acylated hGHA complex 4C (also “1202-4C”) is represented in FIG. 3 where n is 15 and X is 4 or hGHA Variant 4 (SEQ ID NO: 7). The acylated hGHA complex 4C (also “1202-4C”) is also represented by the following chemical structure:
(Formula XXV).
[0424] The acylated hGHA complex 5C (also “1202-5C”) is represented in FIG. 3 where n is 15 and X is 5 or hGHA Variant 5 (SEQ ID NO: 8). The acylated hGHA complex 5C (also “1202-5C”) is also represented by the following chemical structure:
(Formula XXVI).
[0425] The acylated hGHA complex ID (also “1202-1D”) is represented in FIG. 3 where n is 17 and X is 1 or hGHA Variant 1 (SEQ ID NO: 5). The acylated hGHA complex ID (also “1202-1D”) is also represented by the following chemical structure:
(Formula XXVII).
[0426] The acylated hGHA complex 3D (also “1202-3D”) is represented in FIG. 3 where n is 17 and X is 3 or hGHA Variant 3 (SEQ ID NO: 6). The acylated hGHA complex 3D (also “1202-3D”) is also represented by the following chemical structure:
(Formula XXVIII).
[0427] The acylated hGHA complex 4D (also “1202-4D”) is represented in FIG. 3 where n is 17 and X is 4 or hGHA Variant 4 (SEQ ID NO: 7). The acylated hGHA complex 4D (also “1202^0”) is also represented by the following chemical structure:
(Formula XXIX).
[0428] The acylated hGHA complex 5D (also “1202-5D”) is represented in FIG. 3 where n is 17 and X is 5 or hGHA Variant 5 (SEQ ID NO: 8). The acylated hGHA complex 5D (also “1202-5D”) is also represented by the following chemical structure:
(Formula XXX).
EXAMPLE !
[0429] MANUFACTURE OF THE ACYLATED HGH ANTAGONISTS
[0430] Conjugation of Binder Complex to hGH Antagonist
[0431] Conjugation of a binder complex or albumin binder complex to the hGH antagonist is a key step in the production of the acylated hGH antagonists.
[0432] A schematic illustration of the Albumin binder complex production routes is presented in FIG. 16. The Albumin binding complex is a three-components entity composed of (1) Fatty acid, (2) amino acid sequence (gGlu, Glu, AETA, Lys, and (Sy; For structure, please see Table 8), and (3) linking moiety enabling its coupling to the hGH antagonist polypeptide. The three-component binder complex was synthesized on resin, using a standard SPPS techniques. A TFA-containing cleavage cocktail was used for binder cleavage from the resin: 95% TFA, 5% HzO; ratio of 10ml cleavage cocktail to lgr resin. Cleavage was performed for 2hr at RT, followed by evaporation of -80% of the volume.
[0433] Below are two schemes showing the on resin synthesis and the final deprotected binders D andB.
/
§ -A. w* A/* 3 2 I » 2
A V sA t- •><- I V* K v <k X .8. '^yvA
[0435] An organic antisolvent was used for the crude binder precipitation: Diisopropyl ether, ratio of 10ml antisolvent to lml of evaporated cleavage cocktail.
[0436] Binder purification was enabled via preparative HPLC purification cycles, and purified material was finally lyophilized in order to obtain a stable powder containing a TFA-salt purified Albumin binder complex
[0437] Description of the Binder Backbone
[0438] tBu-Fatty acid-yGlu(C)tBu)-Glu(C)tBu)-(AEEA)i-Lys(Mtt)-Gly-Wang resin.
[0439] The fatty add in the binder complex can be, but is not restricted to, either Eicosanedioic or Octadecandioic, and the flexible AEEA spacer tandem unit repetitions can be either 2 or 3, as detailed in the formula below:
n = 15,17 i ~ 2,3
(Formula XXXI).
[0440] Following the removal of the Lys side chain protecting group, a linking moiety is coupled to the primary amine via carbodiimide-initiated condensation reaction A general structure of the two types of the Albumin binding complexes, differ by their linking moiety are described as Albumin binder complex with either Acetyl-Br or MAL-NRFmoc as a linking moiety enabling the coupling to Hgh antagonist polypeptide:
• tBu-Fatty acid-gGlu(C)tBu)-Glu(C)tBu)-(AEEA)i-Lys(Ac-Br)-Gly-Wang resin (where i is 2 or 3).
• tBu-Fatty acid-gGlu(C)tBu)-Glu(C)tBu)-(AEEA)i-Lys(MAL-NRFmoc)-Gly-Wang resin (where i is 2 or 3).
[0441] Chemical schemes of the two Albumin binding complexes possessing an active linking moieties for hGH antagonist polypeptides are presented below.
Q,
HN Br
HO, ,O
O
H o’ O
HO. N H
NH N
H OH
O O Jj H
O
HO O n * 15,17 i * 2,3
(Formula XXXII)
Q NH
O1
HN O
N
HO, O O O
H O O
HO, N n N H OH
O O
6
HO O n « 15,17 i » 2.3
(Formula XXXm)
[0442] Process Steps
[0443] 1. Sequence elongation via SPPS:
[0444] The binder backbone was synthesized with a CEM automatic synthesizer.
[0445] The synthesis was performed with a 2.5 eq excess of AA, Oxymapure and DIC, on Fmoc-dy- Wang resin (preloaded- 0.7mmol/gr) while applying microwave.
[0446] The fatty add conjugation was performed manually, with a 2.5 eq excess of Fatty add, HOBt and DIC without the use of microwave. Fatty add coupling was performed overnight coupling was monitored with Kaiser test In order to assess the fetty acid coupling, yield, a small-scale cleavage was performed before and after fetty acid coupling. FIG. 17 represents a coupling step of the fatty acid Eiocosanedioc to the Fmoc-ydu(OtBu)-du(C)tBu)-(AEEA)3-Lys(mtt)-Gly-Wang resin (peptide on resin). The fatty add-conjugated binder complex was eluted from the C- 18 RP-HPLC column using higher percentage of organic solvent (in this case ACN used in the mobile phase) compared to the non-conjugated peptide.
[0447] FIG. 17 shows RP-HPCD chromatograms. The top chromatogram shows the binder backbone before fatty add coupling (TFmoc), and the bottom chromatogram shows binder 1 backbone after fatty add coupling (8AA).
[0448] 2. Mtt removal:
[0449] In order to couple the linking moiety to the albumin binding complex (either Ac-Br, or the MAL-NRFmoc), the Lys side chain was deprotected. The Mtt protecting group was removed from
the Lys side chain by applying 1%TFA in DCM wash cycles. The washing cycles continued until the entire Mtt was removed. Reaction monitoring was performed by addition of TFA to a few beads. If the beads turn orange/yellow immediately, an additional cycle is required. If the beads do not change color, the cleavage has ended, and no further cycle is necessary.
[0450] 3. Lysine side chain conjugation:
[0451] Below are two methods of conjugating the Lys side chain of the albumin binding complex on a resin to a linking moiety, depending on whether the linker moiety is MAL-NRFmoc-NHS or Ac-Br (Bromoacetic anhydride). Step 3(A) outlines the Lys side chain conjugation method for the Mal- NRFmoc-NHS linker. Step 3(B) outlines the Lys side chain conjugation method for the Bromoacetic anhydride linker.
[0452] 3(A): ForMal-NRFmoc-NHS linker conjugation
[0453] MAL-NRFmoc-NHS linker was reconstituted in DMF to obtain a lOOmg/ml solution (85% of the total calculated volume was used). The concentration was determined by O.D. measurements at 280nm after dilution x500 & xlOOO in (20% 0.1%TFA/HaO:80% ACM). (EC: 21200 ml/(mmol*cm), MW: 501 mg/mmol). The concentration of the linker was adjusted with DMF according to the results obtained. DCM was added to the linker solution to obtain a 50mg/ml linker stock solution pH of the solution was verified to be around pH of 5.
[0454] The reactor was drained, and 2-3eq of linker were added to the reactor and stirred. The pH of the reaction was verified to be around a pH of 5.0. If the pH was lower, 10%DIPEA in (DMF DCM) was added slowly to obtain the required pH of 5.0. The reaction progress was monitored by Kaiser test FIG. 18 represents a conjugation step of MAL-NRFmoc-NHS to the tBu-Eiocosanedioc- ydu(C)tBu)-Glu(C)tBu)-(AEEA)3-Lys-Gly-Wang resin. The MAL-NRFmoc-conjugated binder complex is eluted from the C-18 RP-HPLC column using higher percentage of organic solvent (in this case ACN used in the mobile phase) compared to the non-conjugated binding complex.
[0455] FIG. 18 shows RP-HPCD chromatograms. The top chromatogram shows the binder before MAL-NRFmoc linker conjugation, and the bottom chromatogram shows the binder after linker conjugation.
[0456] At the end of the synthesis, the P-R containing the tBu-Eiocosanedioc-yGlu(C)tBu)- Glu(OtBu)-(AEEA)3-Lys-Gly sequence was washed- 3*DCM, 3*IPE and dried in a vacuum oven (30°C, overnight).
[0457] 3(B): Bromoacetic anhydride conjugation
[0458] 20eq of Bromoacetic anhydride (Ac-Br) were dissolved in DCM, and 20eq of DIPEA were added to the solution. The reactor was drained, and the Bromoacetic anhydride solution was added to the reactor for lhr while stirring. The reaction progress was monitored by Kaiser test FIG. 19 represents a conjugation step of Ac-Br to the tBu- Eiocosanedioc-yGlu(C)tBu)-Glu(C)tBu)-(AEEA)3- Lys-Gly-Wang resin. The Ac-Br -conjugated binder complex is eluted (as expected) from the C-18 RP-HPLC column using higher percentage of organic solvent (in this case ACN used in the mobile phase) compared to the non-conjugated binding complex.
[0459] FIG. 19 shows RP-HPCD chromatograms that represent the step of conjugating Ac-Br to the tBu- Eiocosanedioc-yGlu(C)tBu)-Glu(C)tBu)-(AEEA)3-Lys-Gly-Wang resin. The top chromatogram shows the binder before Ac-Br linker conjugation, and the bottom chromatogram shows binder after linker conjugation
[0460] 4. Cleavage, precipitation & reconstitution:
[0461] Following full synthesis (and identity/purity tests) the Albumin binding peptide complex was cleaved from the Wang peptide in order to be further purified on reparative RP-HPLC system.
[0462] Cleavage was performed with a cocktail containing 95%TFA: 5% H2O, with a ratio of 10ml cleavage cocktail: lgr peptide-resin. After 2hr of cleavage, about 80% of the cleavage cocktail was evaporated (using N2), and then remaining solution was precipitated in cold IPE (-20°C), with a ratio of 10ml IPE: 1 ml of evaporated cleavage cocktail. The binder precipitants were kept at -20°C overnight to achieve full precipitation After a full precipitation, the binders were washed with IPE 3 more times and dried under N2, and the pellet was weighed. Assuming about 50% residual solvents in the pellet, the pellet was reconstituted in DMF to obtain a concentration of ~100mg/ml of stable binder prior to purification and was kept at -80°C until purification started.
[0463] 5. Preparative HPLC purification:
[0464] RP-HPLC was performed in order to obtain the purified binders. Luna Cl 8(2) lOum, 100 A 2.12*25cm column with a loading density of 0.5-3 mg/ml was used.
[0465] Preparation of binder crudes for purification cycles: the DMF dissolved crude binders were diluted xl0-15 with a solution of 30%AcOH:70%ACN. This solution was then diluted x2-5 with
0.1% TFA/HzO to obtain a final solution containing ~2mg/ml of binder crudes.
[0466] The different binders elute from the preparative column at about 60-70% MPB. At the end of each purification cycle, samples of the fractions were taken for RP-HPLC analysis, and the fractions were kept at -80°C until further process.
[0467] 6. Lyophilization:
[0468] Preparative HPLC fractions with purity >90% were pooled. Rotor evaporation was used in order to decrease the ACN content (from 50-60% to <5%), and to increase the pool concentration The final pool was filtered through a 0.22mm PVDF filter before lyophilization and analyzed for purity using RP-HPLC. The concentration and purity before and after filtration were similar suggesting no filter losses. Final lyophilized powders were dissolved and analyzed for purity using RP-HPLC and identity using LC-MS (See FIGS. 20-22).
[0469] FIG. 20 shows RP-HPCD chromatograms of purified Eicosanedioic-yGlu-Glu-(AEEA)3- Lys(Ac-Br)-Gly-OH in the top chromatogram, and Eicosanedioic-yGlu-Glu-(AEEA)3-Lys(MAL- NRFmoc)-Gly-OH in the bottom chromatogram.
[0470] FIG. 21 shows mass spectrum of Eicosanedioic-yGlu-Glu-(AEEA)3-Lys(Ac-Br)-Gly-OH [0471] FIG. 22 shows mass spectrum of Eicosanedioic-yGlu-Glu-(AEEA)3-Lys(MAL-NRFmoc)- Gly-OH
[0472] Methods
[0473] Two main analytical methods were used for purity and identity of the Albumin binding peptide complexes. The RP-HPLC method used for purity is detailed in Table 9, and LCMS method used for identity is detailed in Table 10.
Table 9: Analytical HPLC Conditions
Column Acquity UPLC CSH C18, 50X2.1mm, 1.7um Flow 0.4ml/min
MPA 0.1% TFA/H2O Column Temp. 45°C
MPI.1% TFA Z ACN Wave Length 220nm
Gradient 5% -> 5% in 0.2min -> 20% in 0.3min -> 90%
Injection Volume 1— 0ml
(MPB) in 7min
Table 10: LCMS Conditions
Column Acquity UPLC CSH C18, 100X2.1mm, 1.7um Flow 0.4ml/min
IPB 0.1% FA / ACN Wave Length 214nm
Gradient
1% -> 1% in 2.5min -> 95% in 5min Injection Volume l-5ml
(MPB)
[0474] Binder Conjugation to a hGH antagonist polypeptides
[0475] The binders presented in this Example were dissolved in a water containing buffer systems (e.g. Na-Pi) and were further conjugated to the hGH antagonist polypeptides thought their unpaired Cys side chains (thiol groups).
EXAMPLE 3
[0476] CONJUGATION OF TYPE A OR TYPE B BINDER COMPLEXES TO HGH ANTAGONISTS [0477] Conjugation of an Albumin binder complex to the hGH antagonist is a key step in the production of acylated hGH antagonists.
[0478] The conjugation step was performed between the MAL group of the Albumin binder complex, and thiol group located on the Cys side chain of the hGH antagonist (See Scheme 1). The reaction was performed in a water-based media for 1-2 hours, and quenching of unreacted MAL groups was achieved via lOOmM L-Cys stock.
[0479] Scheme 1 below is a schematic representation of hGH antagonist conjugation to an Albumin binder complex via a bi-functional Fmoc linker (MAL).
NH
O'
HN o
HQ >0
H O
O' o
HO. N IH o H o « o
♦ o HS n * 15,17 i « 2,3 1202* x « 1 <3,4,5
O, NH
O'
HN I o
HQ .0 if °l 0^
HO.
H
O O 2i sj J, I o ( 1202x
HO O n = 15,17 i = 2,3 x » 1.3, 4,5
[0480] The acylated hGH antagonists described throughout the application can be conjugated applying the same outlined above. Moreover, the length of the fatty add chain (C-20 or C-18 for example), the number of spacer units (for example 2-3 instances of AEEA) has no effect on the reaction yields.
[0481] Procedure of Binder Conjugation to AN hGH Antagonist
[0482] hGH Antagonist Handling and preparation:
[0483] The hGH antagonist protein was thawed to room temperature on the bench, or in a 22 ± 3°C water bath. The thawed protein was diluted to -1-1.5 mg/mL with a dilution buffer that contains:
20mM nA-Pi, 200Mm naCl, pH 7. the hGHA solution pH was adjusted to 7 using drop-wise of nAoh (IN). Diluted hGHA was filtered using a 0.22 |im PVDF/PES filter to remove any undesired precipitants. Filtered hGHA was analyzed for concentration using A280 via NanoDrop (EC= 7.96). 1 volume unit of reaction buffer containing: 0.05% HPcd, 20mM na-Pi, 200mM NaCl pH 7 were added to 9 volume units of the hGHA, and a sample was withdrawn for analysis using RP-HPLC (see FIG. 23). The protein solution was kept at room temperature until the reaction was started, [0484] Conjugation:
[0485] The binder solution WAs added to the hGHA solution (1.25eq binder: leq protein). Reaction was kept at RT for 2 hours and analyzed via RP-HPLC for reaction progress every 1 hour (See FIG. 23). At the end of the reaction, the residual binder was quenched by addition of 1 volume unit of quenching solution (lOOmM L-Cys, pH 7) to 100 volume units of the reaction solution. The conjugated hGHA sample was filtrated using 0.22 |im PVDF/PES filter and analyzed for identity using LC-MS (See FIG. 24). The filtrated sample was kept at 4°C until further processed via UFDF for concentration and buffer exchange.
[0486] FIG. 23 shows RP-HPLc Chromatograms of hGHA conjugation reaction to an Albumin binder complex The top chromatogram shows the hGHA solution; the middle chromatogram shows the Binder solution; and the bottom chromatogram shows the end of reaction (the acylated hGHA). The hGHA is 1202-1, the binder is type B (C-20, MAL), and the acylated hGHA is 1202-1D
[0487] FIG. 24 shows LC-ms Analysis to the hGHA and the conjugated protein. The top chromatogram shows hGHA deconvoluted mass (SEQ ID No: 5) and the bottom chromatogram shows the conjugated protein (1202-1D) deconvoluted mass.
[0488] Sample preparation for RP-HPLC:
[0489] One volume of lOOmM L-Cys quenching solution was added to 10 volumes of the analytic sample withdrawn from the reaction. The quenched sample was analyzed using RP-HPLC on Phenomenex Jupiter 5um Cl 8300A column with a gradient of 58-80% 0.1% TFA/CAN.
EXAMPLE 4
[0490] CONJUGATION OF TYPE COR TYPED BINDER COMPLEXES TO HGH ANTAGONISTS [0491] Conjugation of an Albumin binder complex to the hGH antagonist is a key step in the production of acylated hGH antagonists.
[0492] The conjugation step was performed between the Acetyl Bromide group of the Albumin
binder complex, and thiol group located on the Cys side chain of the hGH antagonist (See Scheme 2). The reaction was performed in a water-based media for 3-5 hours, and quenching was performed via lOOmML-Cys stock.
[0493] Reaction progress was monitored (real time progress) via RP-HPLC and LC-MS analysis.
[0494] Scheme 2 below is a schematic representation of hGH antagonist conjugation to Albumin binder complex (Acetyl Bromide).
O
HO o n= 15,17 ♦ HBr i = 2,3 x« 1,3, 4, 5
[0495] Procedure of Binder Conjugation to AN hGH Antagonist
[0496] hGH Antagonist Handling and preparation:
[0497] The hGH antagonist was thawed to RT on the bench or in a 22 ± 3°C water bath. The thawed protein was diluted to ~1 -1.5 mg/mL with a dilution buffer that contains: 20mM na-Pi, 200Mm NaCl pH 7. THE hGHA solution’s pH was adjusted to 7 using NaOH (IN). Diluted MOD-1202x was filtered using a 0.22 |im PVDF/PES filter in order to remove any undesired precipitants. Filtered
hGHA was analyzed for concentration using A.280 via NanoDrop (EC= 7.96). 1 volume unit of reaction buffer containing: 0.05%HPcd, 20mM na-Pi, 200mM NaCl pH 7 were added to 9 volume units of the hGHA, and a sample was withdrawn for analysis using RP-HPLC (See FIG. 25). The protein solution was kept at RT until the reaction was started.
[0498] Binder handling and preparation:
[0499] Lyophilized binder was thawed on the bench for 1 hour. The thawed binder was reconstituted in reaction buffer containing: 0.005%HPcd, 20mM na-Pi, 200mM NaCl pH 7 to reach a concentration of 2-4 mg/mL (wt/V). The binder concentration was analyzed using A.205 via NanoDrop, and a sample was taken for RP-HPLC analysis (See FIG. 26).
[0500] Conjugation:
[0501] The binder solution WAs added to the hGHA solution (3eq binder: leq protein). Reaction was kept at RT and analyzed via RP-HPLC for reaction progress every 1 hour (See FIG. 26). At the end of the reaction, the residual binder was quenched by addition of 1 volume unit of quenching solution (lOOmM L-Cys, pH 7) to 20 volume units of the reaction solution The conjugated hGHA sample was filtrated using 0.22 |im PVDF/PES filter. The filtrated sample was kept at 4°C until further processed via UFDF for concentration and buffer exchange.
[0502] FIG. 25 shows RP-HPLc Chromatograms of hGHA conjugation reaction to an Albumin binder complex. The top chromatogram shows the hGHA solution; the middle chromatogram shows the Binder solution; and the bottom chromatogram shows the end of reaction (the acylated hGHA). The hGHA is 1201, the binder is type D (C-20 Ac-BR), and the acylated hGHA is 1202-1D.
[0503] FIG. 26 shows LC-ms Analysis to the hGHA and the conjugated protein. The top chromatogram shows hGHA deconvoluted mass. The bottom chromatogram shows the conjugated protein deconvoluted mass.
[0504] Sample preparation for RP-HPLC:
[0505] One volume of lOOmM L-Cys quenching solution was added to 10 volumes of the analytic sample withdrawn from the reaction. The quenched sample was analyzed using RP-HPLC on Phenomenex Jupiter 5um Cl 8300A column with a gradient of 58-80% 0.1% TFA/CAN.
EXAMPLE S
[0506] DETERMINATION OF THE IN VITRO ACTIVITY OF ACYLATED HGH ANTAGONISTS USING A BINDING ASSAY
[0507] A Binding assay was developed and used to evaluate the in vitro potency of MOD-1202 variants, while measuring the variants’ ability to inhibit the hGH/ hGHR interaction, as compared to Somavert (Pegvisomant), which is the current available treatment or as compared to an internal reference standard, which is used in order to compare the results of independent assays.
[0508] Briefly, a MaxiSorp plate was coated with human growth hormone receptor (hGHR). After a short blocking procedure with skim milk (a general blocking procedure), purified standard (in serial dilutions to create a STD curve) and tested samples (also in serial dilutions to create inhibition curve) were applied together with a constant concentration of biotinylated human growth Hormone (labeled hGH). Finally, Streptavidin conjugated HRP was added and bound to the biotinylated hGH A TMB substrate, which was applied thereafter, developed a soluble blue reaction product, that was proportional to the level of the hGH that bound the hGHR. The intensity of the blue color was inversely proportional to the concentration of the tested sample (since MOD-1202 is a competitive inhibitor to the binding of hGH to hGHR). Addition of sulfuric acid to stop the enzymatic reaction of the HRP, changes the color to yellow, and enable accurate measurement of the intensity at 450 run using a spectrophotometer or plate reader.
[0509] FIGS. 4A-4D show a representative binding assay results of different hGHA variants with two binder types (Binder Type A which is a C18 albumin binding moiety and Binder Type B which is a C20 albumin binding moiety) and without a binder (naked variants)..
[0510] FIG. 4A shows the binding assay results of the reference standard (RS), hGHA Variant 1 (without any binder complex attached), acylated hGHA complex la, and acylated hGHA complex IB. FIG.4B shows the binding assay results of the reference standard (RS), hGHA Variant 3 (without any binder complex attached), acylated hGHA complex 3a, and acylated hGHA complex 3B. FIG. 4C shows the binding assay results of the reference standard (RS), hGHA Variant 4 (without any binder complex attached), acylated hGHA complex 4A, acylated hGHA complex 4B. FIG.4D shows the binding assay results of the reference standard (RS), hGHA Variant 5 (without any binder complex attached), acylated hGHA complex 5a, and acylated hGHA complex 5B. These results show that the addition of binders leads to a minor decrease in potency compared to the naked variant
[0511] IC50 was calculated using GraphPad Prism software and is summarized in Tables 11 and 12 All acylated hGHA complexes showed similar potency. All acylated hGHA complexes were examined after pre-incubation with HSA and showed approximately the same IC50 with and without HSA There was no significant difference between THE ICSO’s of different hGHA variants and
different binders.
Table 11: Binding assay results of Complexes IB and IDto Somavert
Somavert 1202-1B 1202-1D
IC50 [nm] 124 4.04 4.62
% Relative IC50 100 3069.3 2684.0
Table 12: Binding assay results of Complexes 5B and 5D to Somavert
Somavert 1202-5B 1202-5D
IC50 [nm] 120 4.24 4.5 % Relative IC50 100 2830 2667
[0512] FIGS. 5A and SB show representative binding assay results of different acylated hGH antagonists To Somavert Acylated hGHA complexes IB and ID (Also 1202- IB and 1202-1D respectively) exhibit an increase in potency compared to Somavert (FIG. SA). Acylated hGHA complexes 5B and 5D (also 1202-5B and 1202-5D respectively) exhibit an increase in potency compared to Somavert (FIG SB).
[0513] IC50 was calculated using GraphPad Prism software and is summarized in Table 13. All acylated hGHA complexes variants show 30-fold increase in potency.
Table 13: ic50 Summary Acylated hGHA Variant 1
Reference Variant 1
IB 1A standard (Naked)
IC50 (nM) 10.3 5.22 6.37 6.7 w\o HSA Relative IC50 100 197 162 154 IC50 (nM) 10.2 4.35 6.09 5.13
With HSA Relative ic50 100 234 167 199
Acylated hGHA Variant 3 Reference Variant s
3B 3A standard (Naked) w\o HSA IC50 (nM) 9.87 5.63 7.44 6.50
Relative IC50 100 175 133 152
IC50 (nM) 9.00 5.57 9.96 7.83
With HSA
Relative IC50 100 162 90 116
Acylated hGHA Variant 4
Reference variant 4
4B 4A standard (Naked)
IC50 (nM) 6.49 4.33 4.1 4.53 w\o HSA
Relative IC50 100 149 158 143
IC50 (nM) 5.78 3.94 5.03 5.38
With HSA
Relative IC50 100 147 115 107
Acylated hGHA Variant 5
Reference Variant 5
5B 5A standard (Naked)
IC50 (nM) 11.8 4.73 7 7.21 w\o HSA
Relative IC50 100 249 169 164
IC50 (nM) 13.4 5.96 6.91 6.75
With HSA
Relative IC50 100 225 164 199
EXAMPLE 6
[0514] PHARMACOKINETICS (PK) AND PHARMACOLOGY EFFECT (PD) STUDIES IN RATS
[0515] The pharmacokinetic profiles and pharmacology effects of acylated hGH antagonist complexes administered to rats were measured in a PK/PD study (Internal Study Ref. Nos. 12219 and
12220) following administration OF a single SC injection of the acylated hGHA complexes, as compared to Somavert
[0516] Healthy male SD rats (7-8 weeks of age at study initiation) were assigned to groups of six rats per group (Table 15) based on their body weight On Study Day 1 animals were administered with a single SC injection of TA Serum samples were collected at several timepoints and analyzed for
acylated HGH antagonist serum level using HGH ELISA kit (Roche) and IGF-1 levels using IGF1 ELISA kit (R&D Systems). The design of the experiments is detailed in Table 14 and Table 15.
Linker Linker hGHA No. of
Acylated hGHA conjugati Binder Variant mutations type on
1A C18
NRFmoc
IB 1 4 + G120K E129C C20
ID N/A C20
5A C18
NRFmoc
5B 5 6 + G120K T135C C20
5D N/A C20
Table 15: MOD-1202 PK studies
Study # Test article Study description Dose (mg/kg) & bleeding timepoints
Somavert
Acylated Antagonists: IGF-1 reduction of
Study 1 1A Acylated hGh Bleeding timeooints: (#12219)
IB antagonists to Pre-dose, 0.5, 1, 2, 4, 8, 12, 18, 24, 36, 48, 72,
5A Somavert in rats 96, 120, 144, 168 hours
5B
Acylated hGH Antagonists: 1 mg/kg, 3 mg/kg,
Compare the
Acylated hGH 9 mg/kg biological half-life and
Study 2 Antagonists: IGF-1 reduction of (#12220) IB Bleeding timeooints: Acylated hGH
5B Pre-dose, 0.5, 1, 2, 4, 8, 12, 18, 24, 36, 48, 72, Antagonists 96, 120, 144, 168 hours
Acylated hGH Antagonists: 1 mg/kg, 3 mg/kg,
Compare the
Acylated hGH 9 mg/kg biological half-life and
Study 3 Antagonists: IGF-1 reduction of
(#12222) ID Acylated hGH
5D Pre-dose, 0.5, 1, 2, 4, 8, 12, 18, 24, 36, 48, 72, Antagonists 96, 120, 144, 168 hours
[0517] Study 1 (#12219)
[0518] The study results for #12219 are presented in FIGS. 6-8 and summarized in Tables 16-18 below.
[0519] FIG. 6 shows the pharmacokinetic (PK) results of the acylated hGH antagonists (1202-1 A, 1202-1B, 1202-5A, and 1202-5B) versus Somavert The results in FIG. 6 are based on noncompartmental pharmacokinetic analysis of PK study #12219. The PK parameters are summarized in
Table 16 below.
Table 16: Pharmacokinetic parameters of the analyzed compounds, based on noncompartmental pharmacokinetic analysis of PK study #12219
Compound
Parameter Units
1A IB 5A 5b Somavert
Dose mg/kg _ 5 5 5 5 3 Cmix _ ng/mL _ 4789 5733 6115 6438 10996
Tmix _ h _ 4 18 4 4 48
AUCo-t ng/mL*h _ 72528 113277 101993 138575 941299 AUCo-inf ng/mL*h _ 72574 113369 102055 138630 1093675 tl/2 _ h _ 8.5 11.3 8.4 10.6 51.6
AUMCo-faf ng/mL*hA2 _ 935692 2129433 1292614 2231012 103068630 MRTp-faf h _ 12.9 18.8 12.7 16.1 94.2 V/F (mg/kg)/(ng/mL) 0.0008 0.0007 0.0006 0.0006 0.0003 CL/F (mg/kg)/(ng/mL)/h 0.000069 0.000044 0.000049 0.000036 0.000005 AUCo- kgh/mL 0.0145 0.0227 0.0204 0.0277 0.3646 taf/Dose
[0520] As summarized in Table 16, the obtained half-life values of acylated hGH antagonists type B, that contain the eicosanedioic (C20) fatty-acid binder (IB and 5B) obtained longer half-life values compared to the acylated hGH type A antagonists (1 A and 5 A) that contain the octadecanedioic (Cl 8) fatty-acid binder: 11.3 h vs. 8.5 h and 10.6 h vs. 8.4 h. The obtained half-life values of the acylated hGH antagonists (8.4-11.3 h) were lower by 4.6-6.1 compared to Somavert (51.6 h) resulting in a higher systemic exposure to Somavert compared to the acylated hGH antagonists (&e the dose- normalized AUCo-inf values in Table 16).
[0521] The changes in IGF-1 level (PD effects), following single SC injection of the different compounds, are depicted in FIG. 7 and FIG. 8 and summarized in Table 17 and Table 18 below.
Table 17: IGF-1 serum levels (ng/ml) in SD rats - study #12219
Time-point (h.) 0 8 12 18 24 36 48 72
1202-1A 1205 1135 1014 813 876 1093 1254 1454
1202-1B 1326 1366 1014 868 708 964 1199 1401
1202-5A 1225 1065 912 846 924 1112 1314 1268
1202-5B 1221 1027 1064 872 745 962 1210 1310
Somavert 1334 1262 1269 1362 1184
Table 18: Tabulated IGF-1 %change in SD rats - study #12219
Time-point (h.) 0 8 12 18 24 36 48 72
1202- 1A 0 -8.0 -13.6 -34.0 -25.6 -11.5 6.5 17.9
1202- IB 0 2.5 -21.1 -34.5 -45.2 -27.1 -7.1 6.1
1202-5A 0 -10.9 -27.0 -29.1 -25.9 -6.8 5.2 6.2
1202-5B 0 -10.4 -17.7 -23.7 -42.2 -15.9 -6.1 14.1
Somavert 0 -10.5 0.9 -3.4 -5.8
[0522] The results strongly indicate that acylated hGH antagonists according to the invention have an antagonist effect on the rat GH receptor that is expressed as a decrease in IGF1 levels. Importantly, despite the lower half-lives of these compounds compared to Somavert (Table 16 above), they were much more potent, with respect to IGF-1 reduction. The maximal response for the acylated hGH antagonists according to the invention was observed at 18-24 h from drug administration It should be noted that the type B variants (C20) had a higher (~45% vs. —30%) response compared to the type A variants (Cl 8) with a more prolonged effects (IGF-1 level returned to baseline after 36-48 h for the Cl 8 variants, and at 48-72 h for the C20 variants). Finally, the complexes containing hGHA Variant 1, containing only 4 point mutations (in addition to the G120K and site of conjugation), was not inferior to the complexes containing hGHA Variant 5, containing additional two more point mutations (-34.0% for 1202-1 A compared to -29.1% for 1202-5Aand -45.2% for 1202-1B compared to -42.2% for 1202-5B).
[0523] Study 2 (#12220)
[0524] Based on the results of study #12219, study #12220 was designed to further explore the
PK/PD profiles of the type-B compounds, containing the C20 fatty-acid binder and a non-reversible
Fmoc linker, at three dose levels as detailed in Table 15. Study results are presented in FIG. 9, FIG.
10, FIG. 11 and summarized in Table 19, Table 20, and Table 21 below.
[0525] FIG. 9 shows the PK results based on non-compartmental Pharmacokinetic analysis of the acylated hGH antagonists (1202-1B and 1202-5B) at different dosages. The PK parameters are summarized in Table 19 below.
Table 19: Pharmacokinetic parameters of the analyzed compounds, based on non-compartmental pharmacokinetic analysis of PK study #12220
Compound
Parameter Units
IB SB
Dose mg/kg 1 3 9 1 3 9
Cea* ng/mL 1230 5654 11360 447 6527 8500 h 4 4 4 4 4 4
AUCM Hg/mL*h 15808 69942 170336 6669 58157 127096
AUCe-hf ng/mL*h 15935 70021 170401 6713 58417 127185 tlfl h 9.9 9.4 10.5 9.5 8.7 8.5
MRTO-M h 15.4 14.2 16.7 15.7 12.3 14.4
V/F (mg/kg)/(ng/mL) 0.0009 0.0006 0.0008 0.0020 0.0006 0.0009
CL/F (mg/kg)/(ng/mL)/h 0.000063 0.000043 0.000053 0.000149 0.000051 0.000071
AUCMMIDO* kgh/mL 0.0159 0.0234 0.0189 0.0067 0.0195 0.0141
[0526] As summarized in Table 19, exposure levels were dose proportional for both tested variants, with generally higher and almost dose-linear exposure of IB compared to 5B (see the dose- normalized AUCo-irf values). The obtained half-life values of the two type-B compounds (IB and 5B), were comparable to the half-life obtained in study #12219.
[0527] The changes in IGF-1 level (PD effects), following single SC injection of the different compounds at three dose levels, are depicted in FIG. 10 and FIG. 11 and summarized in Table 20 and Table 21. FIG. 10 shows the changes in IGF-1 serum levels (PD effects) in SD rats, following single SC injection of the different acylated hGHA complexes and dosages. FIG. 11 shows the percentage changes in IGF-1 levels in SD rats from Study #12220, following single SC injection of the different acylated hGHA complexes and dosages.
Table 20: IGF-1 serum levels (ng/ml) in SD rats - study #12220
Time-point (h.)
Dose
Compound (mg/kg)
0 8 12 18 24 36 48 72
1 1113 1189 1187 1164 1359 1249 1335 1258
IB 3 1339 1242 1260 1045 1107 1142 1337 1167
9 1120 1186 1021 931 825 907 1275 1208
1 1310 1186 1411 1030 1196 1278 1330 1246
SB 3 1271 1278 1155 1201 1185 1220 1426 1117
9 1276 1260 1175 1068 947 971 1158 1300
Table 21: Tabulated IGF-1 %change in SD rats - study #12220
Time-point (h.)
Dose
Compound (mg/kg)
0 8 12 18 24 36 48 72
1 0 4.4 9.5 2.3 25.8 9.7 24.0 10.5
IB 3 0 -3.0 -9.3 -18.4 -20.6 -11.1 -3.7 -8.9
9 0 6.7 -8.2 -16.0 -25.2 -18.3 14.3 9.0
1 0 -8.3 7.8 -19.4 -9.0 -0.2 1.1 -3.2
5B 3 0 0.5 -8.9 -5.6 -6.4 -4.1 12.2 -12.1
9 0 -0.2 -8.4 -15.4 -26.8 -23.0 -10.2 3.0
[0528] Comparable to study #12219, the results indicated that the acylated hGH antagonists have an antagonist effect on the rat GH receptor that is expressed as a decrease in IGF1 levels. While no to mild effects on IGF-1 levels were noted at the 1 mg/kg and 3 mg/kg doses, the 9 mg/kg dose resulted in similar % IGF-1 decrease (-25%) for both tested variants. This demonstrates again that IB, containing only 4 point mutations (in addition to the G120K and site of conjugation), is not inferior to the 5B, containing additional two more point mutations. The maximal response for these
compounds administered at 9 mg/kg was observed at 24 h from drug administration Normalization of IGF-1 levels occurred 48-72 h post-administration.
[0529] Study 3 (#12222)
[0530] Study #12222 was designed to explore the PK/PD profiles of the type-D molecules, containing the C20 fatty -acid binder and an AC-Br linker, at three dose levels as detailed in Table 15. Study results are presented in FIG. 12, FIG. 13, FIG. 14 and summarized in Table 22, Table 23, and Table 24 below.
[0531] FIG. 12 below shows the PK results of the ID and 5D acylated hGH antagonists at various dosages. The PK parameters are summarized in Table 22 below.
Table 22: Pharmacokinetic parameters of the analyzed compounds, based on non-compartmental _ pharmacokinetic analysis of PK study #12222 _ Compound
Parameter Units
ID 5D
Dose mg/kg 1 3 9 1 3 9
C.„ ng/mL 643 2200 6521 494 2876 10903
T.„ h 24 18 8 18 8 18
AUQM ng/mL*h 19733 56366 225571 12101 64143 301363
AUCO-M ng/mL*h 19841 56480 225644 12225 64217 301478 tl/2 H 10.6 12.2 13.3 6.4 6.5 7.1
AUMCwrf ng/mL*hA2 464417 1205804 5440268 215512 1083038 6197942
MRTo-taf h 23.4 21.3 24.1 17.6 16.9 20.6
V/F (mg/kg)/(ng/mL) 0.0008 0.0009 0.0008 0.0007 0.0004 0.0003
CL/F (mg/kg)/(ng/mL)/h 0.00050 0.000053 0.000040 0.000082 0.000047 0.000030
AUCo- kgh/mL 0.0198 0.0188 0.0251 0.0122 0.0214 0.0335 taf/Doee
[0532] As summarized in Table 22, the obtained half-life value of ID was ~13 h, i.e., 2-fold higher compared to compound 5D with only -6-7 h of half-life. Exposure levels were dose proportional for both tested variants, with almost dose-linear exposure of variant 1 (see the dose-normalized AUC0- inf values). Despite the higher half-life, exposure levels of compound 5D were generally higher compared to compound ID (see the dose-normalized AUCo-inf values).
[0533] The changes in IGF-1 level (PD effects), following single SC injection of the different D-type molecules, at 3 dose levels, are depicted in FIG. 13 and FIG. 14 and summarized in Table 23 and Table 24 below.
Table 23: IGF-1 serum levels (ng/ml) in SD rats - study #12222
Time-point (h.)
Dose
Compound (mg/kg)
0 8 12 18 24 36 48 72
1 1214 1244 1093 1010 1319 1260 1287 NA
ID 3 1162 1230 1086 935 1088 1158 1340 1234
9 1264 1069 937 676 571 776 1139 1377
1 1239 1440 1376 1225 1387 1394 1378 NA
5D 3 1232 1126 1076 930 1160 1157 1193 1293
9 1164 1099 1006 869 665 840 1170 1293
Table 24: Tabulated IGF-1 %change in SD rats - study #12222
Tune-point (h.)
Dose
Compound (mg/kg)
0 8 12 18 24 36 48 72
1 0 1.5 -8.7 -17.2 10.5 3.9 8.2 NA
ID 3 0 7.9 -8.1 -17.9 -8.2 1.7 13.2 8.3
9 0 -12.9 -27.6 -44.8 -55.9 -36.8 -11.9 12.5
1 0 11.7 16.3 -4.9 18.6 8.2 16.6 NA
5D 3 0 -6.4 -12.5 -23.3 -6.7 -4.0 -2.7 7.0
9 0 -7.9 -11.5 -26.6 -41.5 -28.8 2.4 9.4
[0534] Comparable to the previous studies, these results indicate that the acylated hGH antagonists have an antagonist effect on the rat GH receptor that is expressed as a decrease in IGF1 levels. As in study #12220, while no to mild effects on IGF-1 levels were noted at the 1 mg/kg and 3 mg/kg doses, the 9 mg/kg dose yielded a significant decrease in IGF-1 levels, up to —56%. As in the two previous studies (#12219 and #12220) ID, with fewer point mutations was not inferior to compound 5D, containing additional two point-mutations and actually demonstrated somewhat of a superiority with -56% decrease compared to -42% decrease for variant 5. The maximal response for these compounds
administered at 9 mg/kg was observed at 24 h from drug administration Normalization of IGF-1 levels occurred 48-72 h post-administration.
EXAMPLE ?
[0535] PHARMACOKINETICS (PK) AND PHARMACOLOGY EFFECT (PD) STUDIES IN RABBITS
[0536] The pharmacokinetic profiles and pharmacology effects of acylated hGH antagonist complexes administered to rabbits were measured in PK and PD studies following administration of a single SC or IV injection (Internal Study Ref. No. OPK-OIO-PD) as well as following repeated SC administration (Internal Study Ref. No. OPK-009-PD) of the acylated hGHA complexes, compared to Somavert The design of the experiments is detailed in Table 25 and Table 26.
Table 25: MOD-1202 Tested Acylated hGHA Complexes in PK studies
Linker Linker hGHA No. of
Acylated hGHA conjugati Binder Variant mutations type on
IB NRFmoc C20
1 4 + G120K E129C
ID N/A C20
Table 26: MOD-1202 PK/PD studies
Study# Test article Study description Dose (mg/kg) & bleeding timepoints
Study 1 Somavert & Compare IGF-1 Acylated hGH Antagonists: 3 mg/kg dosed
(#OPK- Acylated hGH
009-PD) Antagonists: hGh antagonists to Somavert: 3 mg/kg, dosed daily IB Somavert in rabbits
ID Bleeding timeooints:
Pre-dose, and daily (pre-dose on dosing days) for up to 8 days post last administration (Day 21)
Study 2 Somavert & Compare the Acylated hGH Antagonists:
(#OPK- Acylated hGH biological half-life SC: 1 mg/kg, 3 mg/kg, 9 mg/kg; IV: 3 mg/kg
010-PD) Antagonist: and IGF-1 reduction Somavert: 3 mg/kg
ID of Acylated hGH
Antagonist (ID) to Somavert in rabbits Pre-dose, 0.5, 1, 2, 4, 6, 8, 11, 16, 24, 33, 48, and to estimate the 72, 96, 120, 144, 168 hours bioavailability of compound ID
[0537] Study 1 (#QPK-009-PD)
[0538] Study &OPK-009-PD was designed to explore the PK/PD profiles of the type-B and D acylated hGHA complexes containing the C20 fatty-acid binder as detailed in Table 26.
[0539] Healthy male NZW rabbits were assigned to groups of three rabbits per group based on their body weight Compounds MOD-1202-1B and MOD-1202-1D were administrated repeatedly every 48 h, for 13 days, subcutaneously (SC). Somavert, as a comparator, was administered daily for 13 days, SC. Serum samples were collected at several timepoints and analyzed for acylated hGH antagonist serum level (IB and ID only) using hGH ELISA kit (Roche) and IGF-1 levels using IGF1 ELISA kit (Mediagnost, Catalog #E20).
[0540] Study results are presented in FIG. 27, FIG. 28, FIG. 29 and summarized in Table 27 and Table 28, below.
[0541] FIG. 27 shows the mean serum concentration of the IB and ID acylated hGH antagonists.
As observed, the serum level of MOD-1202-1D and MOD-1202-1B following repeated administration reach peak levels 24 hours post-injection, followed by a decrease to trough levels before the next injection Although both compounds were administered at the same dose level, measured concentration of MOD-1202-1B were generally lower compared to MOD-1202-1D, with MOD-1202-1B displaying less stable serum levels throughout the study. At the beginning of the study, no major differences in the profile patterns of the compounds were observed, excluding the different serum level concentration. However, on study days 9-13, the difference between the compounds became more pronounced, with IB levels decreasing compared to earlier study days. Accordingly, in animals treated with compound ID, serum levels were detectable for up to 8 days post last injection, while compound IB could be detected for only 5 days post last injection
[0542] The changes in IGF-1 level (PD effects), following repeated SC injection of the different compounds, are depicted in FIG. 28 and FIG. 29 and summarized in Table 27 and Table 28 below.
Table 27: Tabulated IGF-1 serum levels (ng/ml) in NZW rabbits - study #OPK-009-PD
Compound
Study Days Somavert Somavert 1202 vehicle
IB ID vehide
Baseline 691 405 565 458 471 Day 2 598 406 414 317 302 Day 3 438 407 417 267 141 Day 4 361 443 450 244 135 Day 5 285 440 433 258 120 Day 6 241 399 417 213 119 Day 7 287 379 452 153 126 Day 8 291 358 396 140 107 Day 9 246 383 486 253 105 Day 10 296 414 402 199 97 Day 11 265 387 394 318 162 Day 12 248 402 428 274 114 Day 13 _ 209 432 415 346 219
Day 14 241 469 414 246 166 Day 15 231 418 436 336 215 Day 16 244 455 424 356 365 Day 17 270 453 391 367 406 Day 18 _ 262 330 388 370 377
Day 19 287 ND ND 363 ND Day 20 310 ND ND 354 ND Day 21 339 ND ND 326 ND
ND = Not Determined
Table 28: Tabulated IGF-1 %change in NZW rabbits - study #OPK-009-PD
Compound
Study Days Somavert Somavert 1202 vehicle
IB ID vehide
Baseline 0.0 0.0 0.0 0.0 0.0
Day 2 -12.6 0.0 -27.0 -31.0 -35.9
Day 3 -34.8 0.7 -26.7 -42.5 -70.2
Day 4 -46.9 9.5 -21.0 -46.9 -71.5
Day 5 -58.4 8.6 -23.8 -44.3 -74.7
Day 6 -64.9 -1.5 -26.5 -53.8 -74.9
Day 7 -58.4 -6.7 -20.3 -66.7 -73.5
Day 8 -57.5 -11.7 -30.2 -69.5 -77.6
Day 9 -63.8 -5.7 -12.1 -44.7 -77.8
Day 10 -56.2 2.0 -19.1 -50.2 -79.5
Day 11 -61.1 -4.5 -20.1 -21.0 -65.2
Day 12 -63.2 -0.8 -13.6 -31.7 -75.7
Day 13 -69.2 7.2 -15.9 -13.9 -52.9
Day 14 -64.3 15.4 -16.4 -39.6 -64.6
Day 15 -66.1 3.0 -12.2 -16.4 -53.8
Day 16 -63.7 12.1 -14.7 -11.3 -22.4
Day 17 -60.0 11.7 -21.3 -9.3 -13.6
Day 18 -60.9 -18.7 -21.6 -7.9 -26.2
Day 19 -57.7 ND ND -10.3 ND
Day 20 -54.1 ND ND -12.8 ND
Day 21 -50.1 ND ND -19.2 ND
ND = Not Determined
[0543] As in the rat model, the results depicted in FIG. 28 and FIG. 29 indicate that the acylated hGH antagonists have indeed an antagonist effect on the rabbit GH receptor that is expressed as a decrease in IGF1 levels. Overall, the effect on IGF1 levels is well correlated to serum levels of the antagonists. MOD-1202-1D administered every 48 hours, maintained relatively stable and constant low IGF1 levels reaching up to -80% reduction, compared to both MOD-1202-1B (reaching up to -70% reduction) at the same dosing regimen, and also displaying less stable effects, and Somavert (reaching up to -65% reduction) administered every day at the same dose level.
[0544] Study 2 (#QPK-010-PD)
[0545] Study &OPK-010-PD was designed to explore the PK/PD profiles of the MOD-1202-1D containing the C20 fatty-add binder and an AC-Br linker, following a single SC injection, at three dose levels as detailed in Table 26.
[0546] Healthy male NZW rabbits were assigned to groups of three rabbits per group based on their body weight MOD-1202-1D was administrated once on Study Day 1 subcutaneously at different dose levels, or intravenously at a single dose level as described in Table 26. As a comparator, Somavert, was administered once subcutaneously at 3 mg/kg. Serum samples were collected at several timepoints and analyzed for acylated hGH antagonist serum level using an MSD based, inhouse developed immunosorbent assay for MOD-1202-1D or using a PEG ELISA kit (Abeam, cat #ab215546) for Somavert IGF-1 levels were measured by Endokrinologisdies Labor, in Munich Germany, using the automated IDS-iSYS immunoassay.
[0547] Study results are presented in FIG.30, FIG.31, FIG.32, and summarized in Table 29, Table
30, and Table 31, below.
[0548] FIG. 30 shows the PK results of MOD-1202-1D at various dosages compared to Somavert.
The PK parameters are summarized in Table 29 below.
Table 29: Pharmacokinetic parameters of the analyzed compounds, based on non-compartmental _ pharmacokinetic analysis of PK study #QPK-010-PD
Compound
Parameter Units
ID Somavert
Dose mg/kg 1 3 9 3 IV 3
C.„ ng/mL 681 8932 34700 84997 28470
T.„ h 2 - 8 0.5 - 24 6 - 24 0.5* 48*
AUQM ng/mL*h 16444 299753 1198100 876771 2541156
AUCO-M ng/mL*h 16522 299843 1198261 876873 2757187 tl/2 H 13.8 16.0 18.3 18.2 35.3
MRTo-taf h 20.9 23.0 23.9 10.0 77.6
V/F (mg/kg)/(ng/mL) 0.0013 0.0003 0.0002 0.0001“ 0.0001
CL/F (mg/kg)/(ng/mL)/h 0.000064 0.000011 0.000008 0.000003 0.000001
AUCo- kgh/mL 0.0165 0.0999 0.1331 0.2923 0.9191 taf/Doee
Data arc presented as mean for Cmax, Tl/2, AUC, Vz/F and Cl/F values, and as a range for Tmax values. * Same value for all rabbits; ** Absolute V and CL are reported for the IV group
[0549] As summarized in Table 29, the obtained half-life of MOD-1202-1D was found to be 13.8-
18.3 h compared to -35 h for Somavert Exposure levels were dose correlated; however, they were not proportional (See the dose-normalized AUCo-irf values), indicating non-linear pharmacokinetics properties at lower doses (6-fold higher dose-normalized exposure values for the 3 mg/kg compared to 1 mg/kg). However, at higher doses (9 mg/kg over 3 mg/kg), exposure parameters ratios were almost linear (only 1.3-fold higher dose-normalized exposure values for the 9 mg/kg compared to 3 mg/kg).
[0550] The bioavailability of MOD-1202- ID following SC administration was calculated from the dose-adjusted exposure levels comparison by both routes of administration (SC and TV) and was found to be variable and dose dependent ranging from 5.6% (for the low 1 mg/kg dose) to as much as 45.5% (for the high 9 mg/kg dose). Comparing the exposure levels of the 3 mg/kg dose by both routes yields -34% bioavailability.
[0551] The changes in IGF-1 level (PD effects), following single SC injection of the different compounds, are depicted in FIG. 31 and FIG. 32 and summarized in Table 30 and Table 31 below.
Table 30: Tabulated IGF-1 serum levels (ng/ml) in NZW rabbits - study #OPK-010-PD
Compound
Time-point ID Somavert (h.)
1 mg/kg 3 mg/kg 9 mg/kg 3 IV mg/kg 3 mg/kg
0 (baseline) 310 354 288 317 279
0.5 320 341 286 317 278
1 311 349 283 312 287
2 311 347 283 313 280
4 328 353 298 328 287
6 339 357 301 322 290
8 340 336 290 311 293
11 338 329 280 290 297
16 342 284 254 251 285
24 350 239 217 211 275
33 371 201 190 187 267
48 357 192 166 224 261
72 313 299 181 295 267
96 296 308 241 294 290
120 307 295 247 298 316
144 286 291 231 295 304
168 290 292 239 283 292
Tab e 31: Tabulated IGF-1 %change in NZW rabbits - study #QPK-010-PD Compound
Time-point ID Somavert (h.)
1 mg/kg 3 mg/kg 9 mg/kg 3 IV mg/kg 3 mg/kg
0 (baseline) 0.0 65 65 OO 0.0 05 3.2 3J -0.5 03 -0.1 1 0.0 -0.8 -1.6 -L2 3.0 2 0.3 -L2 0.6 4 5.8 02 3/7 33 2.8 6 9.1 L6 43 2J 4.1 8 9.5 -4.7 L2 T5 5.4 n 8.9 55 -2.6 55 6.6 16 10.3 -19.5 -11.2 503 2.6 24 13.3 -32.3 -24.0 32/7
33 20.7 -42.7 -33.6 -40.3 -3.7
48 16.1 -45.2 -41.7 -28.1 ZT
72 1.6 -15.0 -35.4 -5.9 TTJ
96 -3.9 -12.3 -15.3 -6.3 Ze"
120 0.0 -16.1 -13.4 TZe
144 -6.9 -16.8 -19.1 -5.3 103
168 -16.7 -16.2 -9.8 TT
[0552] Despite its relatively long half-life (Table 29), Somavert administered as a single SC dose of 3 mg/kg, had no effect on IGF1 level. MOD-1202-1D, however, administered at 3 mg/kg and 9 mg/kg single SC injections, yielded comparable IGF-1 maximal decrease (-45%) 48 h post injection Despite its shorter half-life compared to Somavert, the effect was maintained for up to 48 h and 72 h postdose in animals treated with MOD-1202-1D at 3 mg/kg and 9 mg/kg dose, respectively. In animals treated with 3 mg/kg given via the IV route, a similar maximal effect on IGF1 levels was observed; however, the duration of the effect was shorter and lasted for 33-48 h post administration
[0553] Conclusion
[0554] Taking all the previous Examples and studies together, the acylated hGHA complexes containing hGHA Variant 1, which has 6 point mutations, exhibited better PK and PD effects compared to other acylated hGHA complexes studied. Specifically, the acylated hGHA complexes containing hGha variant 1 were superior to the acylated hGHA complexes containing hGHA Variant 5, which has 8 point mutations.
[0555] With respect to the binder complexes in the acylated hGH antagonists and in view of the previous studies, the acylated hGH antagonists having bromide as a linker (D-type binder complexes) showed comparable results compared to the acylated hGH antagonists having NRFmoc as a linker (B-type binder complexes).
[0556] Together with CMC considerations, including but not limited to simplicity of production, analytical characterization, and stability-related issues, the acylated hGHA complex MOD-1202-1D shows the greatest potential as a therapeutic protein. This acylated hGHA has the ability to harness the benefits of albumin binding through a fatty add moiety, leading to prolonged half-life (~ 13 h in rats and -16 h in rabbits) while reaching the desired PD effects, i.e., decreasing IGF-1 serum levels (up to - 56% reduction in rats following single administration and - 80% in rabbits following repeated administration). A head-to-head comparison of MOD-1202-1D with Somavert, the commercially
available drag, show clear advantage of MOD-1202-1D following a single or repeated SC administration in rabbits. Among the different linker types that were tested, the D-type linker, composed of only Acetyl-Bromide group is the more promising linker with respect to both production processes and biocompatibility.
Claims
What is claimed is:
1. A human growth hormone receptor antagonist (“GHR antagonisf’ or “hGHA"), comprising growth hormone receptor antagonist G120K (SEQ ID NO: 2) and at least one of the following amino acid mutations: H18D, H21N, F54P, R64K, R167N, D171S, E174S, I179T.
2. The GHR antagonist of claim 1, further comprising at least one amino acid mutation to cysteine and wherein the amino acid mutated to cysteine is T3, E39, P48, Q69, N99, L129, T135, T142, Hl 51 or combinations thereof.
3. The GHR antagonist of claim 1, wherein the GHR antagonist has the following amino acid mutations:
(a) G120K, L129C, R167N, D171S, E174S, andI179T;
(b) H18D, H21N, F54P, R64K, G120K, L129C, R167N, D171S, E174S, andI179T;
(c) H18D, H21N, G120K, L129C, R167N, D171S, E174S, andI179T; or
(d) H18D, H21N, G120K, T135C, R167N, D171S, E174S, andI179T.
4. The GHR antagonist of claim 3, wherein the GHR antagonist comprises the amino acid sequence of SEQ ID NO: 5, SEQ ID NO: 6, SEQ ID NO: 7, or SEQ ID NO: 8.
A modified human growth hormone receptor antagonist (“modified GHR antagonisf ’ or “modified hGHA”) having the structure of formula (I):
W-X-Y-Z (I) wherein:
W represents a binder;
X represents a spacer;
Y represents an optional linker;
Z represents a human growth hormone receptor antagonist (“GHR antagonist”); or a pharmaceutically acceptable salt thereof.
6. The modified GHR antagonist of claim 5, wherein the GHR antagonist comprises any of one of claims 1 to 4.
7. The modified GHR antagonist of any one of claims 5 to 6, wherein W is a fatty acid.
8. The modified GHR antagonist of any one of claims 5 to 6, wherein W is octadecanedioic acid (Cl 8 diacid) or is eicosanedioic acid (C20 diacid).
9. The modified GHR antagonist of claim 8, wherein octadecanedioic acid (Cl 8 diacid) is represented by Formula II:
O
OH
HO o or is represented by Formula II-A:
O
HO
; or wherein eicosanedioic acid (C20 diacid) and is represented by Formula HI: o
OH
HO
O or is represented by Formula IH-A: o
HO o
10. The modified GHR antagonist of any one of claims 5 to 9, wherein X is gGlu-Glun- (AEEA)m-Cys-Glyp, gGlu-Glun-(AEEA)m-Lys(AcBr)-Glyp, or gGlu-Glun-(AEEA)m-Lys- Glyp and n is 1, 2, or 3; m is 1, 2, or 3; and p is 1, 2, or 3.
11. The modified GHR antagonist of any one of claims 5 to 9, wherein X is gGlu-Glu- (AEEA)m-Cys-Gly, m is 1, 2, or 3, and is represented by Formula IV:
n® 1. 2, 3 or is represented by Formula IV-E;
n« 1, 2, 3
12. The modified GHR antagonist of any one of claims 5 to 9, wherein X is gGlu-Glu- (AEEA)m-Lys(AcBr)-Gly, m is 1, 2, or 3, and is represented by Formula V:
13. The modified GHR antagonist of any one of claims 5 to 9, wherein X is gGlu-Glu- (AEEA)m-Lys-Gly, m is 1, 2, or 3, and is represented by Formula VI:
NH2
HQ o O
H O' o .N
HaN OH o O
14. The modified GHR antagonist of any one of claims 5 to 13, wherein ¥ is Chloropropane- 2-one-Fmoc-Mal.
15. The modified GHR antagonist of claim 14, wherein Chloropropane-2-one-Fmoc-Mal is represented by Formula VII:
or is represented by Formula VII-C:
O o
N H o
O
HN o o
16. The modified GHR antagonist of any one of claims 5 to 13, wherein Y is Mal-NRFmoc- NHS.
17. The modified GHR antagonist of claim 16, wherein Mal-NRFmoc-NHS is represented by Formula X:
or is represented by Formula X-C:
18. The modified GHR antagonist of any one of claims 16 to 17, wherein the bond between the GHR antagonist and the linker is a stable covalent bond.
19. The modified GHR antagonist of any one of claims 14 to 15, wherein the bond between the GHR antagonist and the linker is a reversible covalent bond.
20. The modified GHR antagonist of any one of claims 5-19, wherein the modified GHR antagonist comprises any one of the following formulas:
(Formula XXX).
21. A method of inhibiting human growth hormone (hGH) action in a subject, the method comprising: administering to a subject in need thereof an effective amount of the GHR antagonist of any one of claims 1 to 4 or the modified GHR antagonist of any one of claims 5 to 20, to inhibit hGH action in the subject.
22. The method of claim 21, wherein the method comprises administering to the subject said GHR antagonist or the modified GHR antagonist in an amount effective to treat, prevent, or ameliorate a hGH related disorder in the subject
23. The method of claim 22, wherein the hGH related disorder is one or more of: acromegaly; age-related macular degeneration; cancer; diabetes; gigantism, vascular eye diseases, diabetic retinopathy, retinopathy of prematurity, and retinopathy of sickle-cell anemia; and, nephropathy.
24. A method of treating, preventing, or ameliorating a hGH related disorder, the method comprising: administering to a subject in need thereof an effective amount of the GHR antagonist of any one of claims 1 to 4 or the modified GHR antagonist of any one of claims 5 to 20, to treat, prevent, or ameliorate a hGH related disorder in the subject.
25. A method of producing an albumin binding complex on a resin, the steps comprising:
(a) Synthesizing a protected binder backbone of Formula XXXI on a resin
HNH
Or
O O
H
NH N o OH
O
HO o n - 15,17 i « 1,2,3
(Formula XXXI);
(b) Removing the methyltrityl (Mtt) resulting in a deprotected binder backbone;
(c) Coupling a linking moiety to the deprotected binder backbone resulting in an albumin binding peptide complex;
(d) Cleaving the albumin binding peptide complex from the resin;
(e) Purifying the albumin binding peptide complex; and
(f) Lyophilizing the albumin binding peptide complex.
26. The method of claim 25, wherein said synthesizing of a binder backbone is done using solid-phase peptide synthesis (SPPS).
27. The method of claim 25 or 26, wherein:
1. said binder backbone is tBu-Fatty acid-gGhi(OtBu)-Ghi(OtBu)-(AEEA)i- Lys(Mtt)-Gly-Wang resin, wherein i is 2 or 3 and the Fatty acid is eicosanedioic or octadecandioic; ii. said removing in step (b) is done by applying 1%TFA in DCM wash cycles; ill. said removing in step (b) deprotects the Lys side chain; iv. the intermediate composition following step (b) is the tBu-eiocosanedioc- yGlu(OtBu)-Glu(OtBu)-(AEEA)3-Lys-Gly-Wang resin, tBu-eiocosanedioc- yGlu(OtBu)-Glu(OtBu)-(AEEA)2-Lys-Gly-Wang resin, tBu-octadecandioic- yGlu(OtBu)-Glu(OtBu)-(AEEA)3-Lys-Gly-Wang resin, or tBu-octadecandioic- yGlu(OtBu)-Glu(OtBu)-(AEEA)2-Lys-Gly-Wang resin;
V. wherein the linker moiety and deprotected binder backbone are added to a reactor and stirred;
vi. the linker moiety is coupled to the primary amine of the deprotected Lys side chain of the peptide backbone; vii. said cleaving in step (d) is done with a cocktail containing 95%TFA: 5% H2O, with a ratio of 10ml cleavage cocktail: lgr peptide-resin; or vni. wherein said purifying in step (e) is done on RP-HPLC column; or any combinations thereof.
28. The method of any one of claims 25-27, wherein said resin is Wang resin.
29. The method of any one of claims 25-28, wherein said linking moiety in step (c) is Acetyl- Br.
30. The method of claim 29, wherein around 20eq of Bromoacetic anhydride is dissolved in DCM, and 20eq of DIPEA is added to the solution resulting in a Bromoacetic anhydride solution ready for Lys side chain conjugation.
31. The method of claim 30, wherein said Bromoacetic anhydride solution is added to the reactor and stirred.
32. The method of claim 31, wherein said stirring comprises 1 hour.
33. The method of any one of claims 25-28, wherein said linking moiety in step (c) is MAL- NRFmoc.
34. The method of claim 33, wherein around 2-3eq of the MAL-NRFmoc is added to the reactor.
35. The method of any one of claims 33-34, wherein the pH in the reactor is around 5.0.
36. The method of any one of claims 25-32, wherein the albumin binding complex is Formula XXXII:
Q,
HN Br
HO, ,O
O
H N o' O
HO. H
NH N
H OH
O O
O
O
HO n « 15,17 i * 1,2,3
(Formula XXXII).
37. The method of any one of claims 25-32, wherein the albumin binding complex is tBu- eicosanedioic-yGlu(OtBu)-Glu(OtBu)-(AEEA)3-Lys(Ac-Br)-Gly-Wang resin, tBu- eicosanedioic-yGlu(OtBu)-Glu(OtBu)-(AEEA)2-Lys(Ac-Br)-Gly-Wang, resin tBu- octadecandioic-yGlu(OtBu)-Glu(OtBu)-(AEEA)3-Lys(Ac-Br)-Gly-Wang resin, or tBu- octadecandioic-yGlu(OtBu)-Glu(OtBu)-(AEEA)2-Lys(Ac-Br)-Gly-Wang resin.
38. The method of any one of claims 25-28 or 33-35, wherein the albumin binding complex is Formula XXXHI:
, ,
(Formula XXXHI).
39. The method of any of claims 25-28 or 33-35, wherein the albumin binding complex is tBu-eicosanedioic-yGlu(OtBu)-Glu(OtBu)-(AEEA)3-Lys(MAL-NRFmoc)-Gly-Wang resin, tBu-eicosanedioic-yGlu(OtBu)-Glu(OtBu)-(AEEA)2-Lys(MAL-NRFmoc)-Gly- Wang resin, tBu-octadecandioic-yGlu(OtBu)-Glu(OtBu)-(AEEA)3-Lys(MAL-NRFmoc)- Gly-Wang resin, or tBu-octadecandioic-yGlu(OtBu)-Glu(OtBu)-(AEEA)2-Lys(MAL- NRFmoc)-Gly-Wang resin.
40. An albumin binding complex comprising Formula XXXII:
O,
HN Br
H N
O
O
HO ■o 15,17 ,2,3
(Formula XXXII).
41. An albumin binding complex comprising tBu-eicosanedioic-yGlu(OtBu)-Glu(OtBu)- (AEEA)3-Lys(Ac-Br)-Gly-Wang resin, tBu-eicosanedioic-yGlu(OtBu)-Glu(OtBu)- (AEEA)2-Lys(Ac-Br)-Gly-Wang resin, tBu-octadecandioic-yGlu(OtBu)-Glu(OtBu)- (AEEA)3-Lys(Ac-Br)-Gly-Wang resin, or tBu-octadecandioic-yGlu(OtBu)-Glu(OtBu)- (AEEA)2-Lys(Ac-Br)-Gly-Wang resin.
42. A method of conjugating an albumin binding complex of any one of claims 40-41 to an hGH antagonist by adding said binding complex to said hGHA in solution
43. The method of claim 42, wherein: said hGH antagonist is any one of claims 1-4; ii. said binder complex is 3 eq; said hGHA is leq; iv. the solution was kept at room temperature; v. once the conjugation is completed the solution is quenched; or
VI. once the conjugation is completed the solution is quenched and said quenching solution is 1 OOmM L-Cys at a pH of 7; or any combinations thereof.
44. The method of claim 43, wherein 1 volume unit of the quenching solution is added to 20 volume units of the reaction solution.
(Formula XXXHI).
46. An albumin binding complex comprising tBu-eicosanedioic-yGlu(OtBu)-Glu(OtBu)- (AEEA)s-Lys(MAL-NRFmoc)-Gly-Wang resin, tBu-eicosanedioic-yGlu(OtBu)- Glu(OtBu)-(AEEA)2-Lys(MAL-NRFmoc)-Gly-Wang resin, tBu-octadecandioic- yGlu(OtBu)-Glu(OtBu)-(AEEA)3-Lys(MAL-NRFmoc)-Gly-Wang resin, or tBu- octadecandioic-yGlu(OtBu)-Glu(OtBu)-(AEEA)2-Lys(MAL-NRFmoc)-Gly-Wang resin.
47. A method of conjugating an albumin binding complex of any one of claims 45-46 to an hGH antagonist by adding said binding complex to said hGHA in solution
48. The method of claim 47, wherein
1. said hGH antagonist is any one of claims 1-4; said binder complex is 1.25eq; iii. said hGHA is leq;
IV. wherein the solution was kept at room temperature; once the conjugation is completed the solution is quenched; or vi. once the conjugation is completed the solution is quenched and said quenching solution is lOOmM L-Cys at a pH of 7; or any combinations thereof.
49. The method of claim 48, wherein 1 volume unit of the quenching solution is added to 100 volume units of the reaction solution.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US202363592760P | 2023-10-24 | 2023-10-24 | |
| US63/592,760 | 2023-10-24 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO2025088605A2 true WO2025088605A2 (en) | 2025-05-01 |
| WO2025088605A3 WO2025088605A3 (en) | 2025-06-12 |
Family
ID=93566438
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/IL2024/051024 Pending WO2025088605A2 (en) | 2023-10-24 | 2024-10-22 | Human growth hormone receptor antagonists and methods of use thereof |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2025088605A2 (en) |
Family Cites Families (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| BRPI0507436A (en) * | 2004-02-09 | 2007-07-03 | Pharmacia Corp | chemically modified human growth hormone receptor antagonist conjugates |
| KR102167827B1 (en) * | 2017-12-20 | 2020-10-20 | 주식회사 알테오젠 | Novel Growth Hormone Receptor Antagonists and Fusion Proteins Thereof |
| WO2019211842A1 (en) * | 2018-04-30 | 2019-11-07 | Opko Biologics Ltd. | Long-acting human growth hormone-antagonists and methods of producing the same |
| WO2020257243A1 (en) * | 2019-06-17 | 2020-12-24 | Molecular Technologies Laboratories Llc | Therapeutic pegylated growth hormone antagonists |
| WO2023141527A1 (en) * | 2022-01-21 | 2023-07-27 | Applied Molecular Transport Inc. | Furin-cleavable delivery constructs |
-
2024
- 2024-10-22 WO PCT/IL2024/051024 patent/WO2025088605A2/en active Pending
Non-Patent Citations (16)
| Title |
|---|
| ALEX F. MULLER: "Growth Hormone Receptor Antagonists", THE JOURNAL OF CLINICAL ENDOCRINOLOGY & METABOLISM, vol. 89, 1 April 2004 (2004-04-01), pages 1503 - 1511 |
| CUNNINGHAM ET AL., SCIENCE, vol. 244, 1989, pages 1081 |
| CUNNINGHAM ET AL., SCIENCE, vol. 255, 1991, pages 306 |
| DM. CHEN ET AL., ENDOCRINOLOGY, vol. 136, 1995, pages 660 - 667 |
| GERICH ET AL., NEW ENGLAND J. MED., vol. 310, 1984, pages 848 - 850 |
| GHUMAN, JAMIE ET AL.: "Structural basis of the drug-binding specificity of human serum albumin.", JOURNAL OF MOLECULAR BIOLOGY, vol. 353, no. 1, 2005, pages 38 - 52, XP005086293, DOI: 10.1016/j.jmb.2005.07.075 |
| HOUSSAYBIASOTTI, REV. SOC. ARGENT. BIOL., vol. 6, 1930, pages 251 - 296 |
| LARSENMAJA THIM ET AL.: "Albumin-based drug delivery: harnessing nature to cure disease.", MOLECULAR AND CELLULAR THERAPIES, vol. 4, no. 1, 2016, pages 1 - 12 |
| LOWMAN, HENRY B.JAMES A. WELLS.: "Affinity maturation of human growth hormone by monovalent phage display.", JOURNAL OF MOLECULAR BIOLOGY, vol. 234, no. 3, 1993, pages 564 - 578 |
| MELMED ET AL., AMER. J. MED., vol. 97, 1994, pages 468 - 473 |
| RYMASZEWSKI ET AL., PROC. NATL. ACAD. SCI. USA, vol. 88, 1991, pages 617 - 621 |
| SMITH ET AL., ABSTRACT OF PRESENTATION AT ARVO MEETING, May 1995 (1995-05-01) |
| SPECTOR AA, J LIPID RES, vol. 16, 1975, pages 165 - 79 |
| VAN DER LELY ET AL., THE LANCET, vol. 358, 2001, pages 1754 - 1759 |
| WELLS ET AL., ANNU. REV. BIOPHYS. BIOMOL. STRUCT., vol. 22, no. 329, 1993 |
| ZORZIALESSANDROSARA LINCIANOALESSANDRO ANGELINI: "Non-covalent albumin-binding ligands for extending the circulating half-life of small biotherapeutics.", MEDCHEMCOMM, vol. 10, no. 7, 2019, pages 1068 - 1081, XP055652580, DOI: 10.1039/C9MD00018F |
Also Published As
| Publication number | Publication date |
|---|---|
| WO2025088605A3 (en) | 2025-06-12 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN100425284C (en) | Methods for glucagon suppression | |
| KR101352225B1 (en) | Novel Exendin variant and conjugate thereof | |
| CN103649115B (en) | Polypeptide | |
| US6451970B1 (en) | Peptide derivatives | |
| CN103596583B (en) | Novel Glucagon Analogs | |
| CN102197049B (en) | Amylin derivatives | |
| CN112043835B (en) | Bioreversible introducing group for nitrogen and hydroxyl-containing drugs | |
| EP3518982B1 (en) | Controlled-release pth compounds | |
| EP2528942B1 (en) | Novel compounds and their effects on feeding behaviour | |
| JP2020535138A (en) | Long-acting coagonists of glucagon and GLP-1 receptors | |
| CN101400698A (en) | Amylin derivatives | |
| CN103459416A (en) | Novel compounds and their effects on feeding behavior | |
| TW200529868A (en) | N-terminally monopegylated human growth hormone conjugates, process for their preparation, and methods of use thereof | |
| JP2023506952A (en) | Incretin analogues and uses thereof | |
| JP7443315B2 (en) | Acylated oxyntomodulin peptide analogs | |
| JP2024501467A (en) | new compound | |
| JP7279061B2 (en) | glucagon-like peptide | |
| WO2025088605A2 (en) | Human growth hormone receptor antagonists and methods of use thereof | |
| JP7539835B2 (en) | PYY Analogs | |
| TW201808346A (en) | Long-acting oxyntomodulin formulation and methods of producing and administering same | |
| EP2389198A1 (en) | Site-specific monoconjugated insulinotropic glp-1 peptides | |
| CN117417430A (en) | Bullfrog GLP-1 analogues with agonistic activity on GLP-1 and glucagon receptor and application thereof | |
| CN117417431A (en) | A class of polypeptides with agonistic activity on GLP-1, glucagon and GIP receptors and their applications | |
| CN107365375B (en) | A kind of pair of GLP-1 receptor has the polypeptide compound and its preparation method and application of high-affinity | |
| WO2022177018A1 (en) | Method for producing long-acting adrenomedullin derivative |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 24809074 Country of ref document: EP Kind code of ref document: A2 |