WO2025076498A1 - Treatment of osteogenesis imperfecta with c-type natriuretic peptide and analogs thereof - Google Patents
Treatment of osteogenesis imperfecta with c-type natriuretic peptide and analogs thereof Download PDFInfo
- Publication number
- WO2025076498A1 WO2025076498A1 PCT/US2024/050159 US2024050159W WO2025076498A1 WO 2025076498 A1 WO2025076498 A1 WO 2025076498A1 US 2024050159 W US2024050159 W US 2024050159W WO 2025076498 A1 WO2025076498 A1 WO 2025076498A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- cnp
- seq
- various embodiments
- subject
- bone
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P19/00—Drugs for skeletal disorders
- A61P19/08—Drugs for skeletal disorders for bone diseases, e.g. rachitism, Paget's disease
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K38/00—Medicinal preparations containing peptides
- A61K38/16—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- A61K38/17—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- A61K38/22—Hormones
- A61K38/2242—Atrial natriuretic factor complex: Atriopeptins, atrial natriuretic protein [ANP]; Cardionatrin, Cardiodilatin
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K14/00—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- C07K14/435—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- C07K14/575—Hormones
- C07K14/58—Atrial natriuretic factor complex; Atriopeptin; Atrial natriuretic peptide [ANP]; Cardionatrin; Cardiodilatin
Definitions
- the present disclosure relates, in general, to C-type natriuretic (CNP) therapy to treat skeletal dysplasia, such as osteogenesis imperfecta, and measures of efficacy in treatment.
- CNP C-type natriuretic
- C-type Natriuretic Peptide is a paracrine growth factor widely expressed across numerous tissues (Prickett et aL, Peptides 2020; 132:170363) with diverse functions including regulation of endochondral bone growth, blood flow and pressure in the microcirculation, antiinflammatory actions, gamete maturation and neurogenesis and connectivity (Kuhn M., Physiol Rev 2016; 96:751 -804). The best defined of these factors in humans is the crucial role of the hormone in skeletal growth in driving growth plate expansion.
- NTproCNP concentrations are raised where intra cellular CNP pathway activity is reduced (Olney et aL, J Clin Endocrinol Metab 2015; 100:E355-359; Wang et al., Hum Mutat 2015; 36:474-481 ) and are reduced where intra cellular activity is enhanced (Hannema et al., J Clin Endocrin Metab 2013; 98:E1988-1998; Boudin et aL, Am J Hum Genet 2018; 103:288-295; Miura et al. PloS one 2012; 7:e42180).
- the present disclosure is directed to treatment of skeletal dysplasias such as osteogenesis imperfecta in patients using CNP or variants thereof.
- a method of treating osteogenesis imperfecta (Ol) or alleviating one or more symptoms thereof comprising administering a C-type natriuretic peptide (CNP) to a subject in need thereof, wherein the CNP is administered less frequently than daily administration.
- the CNP is administered 5 times weekly, 4 times weekly, 3 times weekly, 2 times weekly, weekly, bi-weekly, monthly or less frequently.
- a method of treating osteogenesis imperfecta comprising administering a C-type natriuretic peptide (CNP) to a subject in need thereof, wherein the CNP is administered between 2 to 5 times per week.
- CNP C-type natriuretic peptide
- a method of reducing the frequency of bone fracture in a subject having osteogenesis imperfecta comprising administering a C-type natriuretic peptide (CNP) to a subject in need thereof, wherein the CNP is administered between 2 to 5 times per week.
- CNP C-type natriuretic peptide
- the disclosure provides a method of improving bone mineral density in a subject having osteogenesis imperfecta comprising administering a C-type natriuretic peptide (CNP) to a subject in need thereof, wherein the CNP is administered between 2 to 5 times per week.
- CNP C-type natriuretic peptide
- the disclosure provides a method of improving femoral length or vertebral growth in a subject having osteogenesis imperfecta comprising administering a C- type natriuretic peptide (CNP) to a subject in need thereof, wherein the CNP is administered between 2 to 5 times per week.
- CNP C- type natriuretic peptide
- a method of treating osteogenesis imperfecta comprising administering a C-type natriuretic peptide (CNP) to a subject in need thereof, wherein the CNP is administered weekly or bi-weekly.
- CNP C-type natriuretic peptide
- a method of reducing the frequency of bone fracture in a subject having osteogenesis imperfecta comprising administering a C-type natriuretic peptide (CNP) to a subject in need thereof, wherein the CNP is administered weekly or biweekly.
- CNP C-type natriuretic peptide
- the osteogenesis imperfecta is Type I, Type II, Type III, Type IV, or Type V Ol. In various embodiments, the osteogenesis imperfecta is Type VI -Type XXI Ol.
- the subject is further administered adjunct therapy to treat osteogenesis imperfecta.
- the adjunct therapy is alendronate, risedronate, ibandronate, zoledronic acid, denosumab, estrogen therapy or hormone therapy, raloxifene, apeledoxifene and estrogen, teriparatide, abaloparatide, or romosozumab.
- the adjunct therapy is bisphosphonate therapy.
- the bisphoshonate is selected from the group consisting of pamidronate, alendronate, ibandronate and risedronate, or combinations thereof.
- the bisphosphate is administered orally, intravenously or subcutaneously.
- the subject has a mutation in a gene selected from the group consisting of COL1A1 , COL1A2, COL2A1 , COL11 A1 , COL9A2, COL10, IFITM5, SERPINF1 , CRTAP, LEPRE1 , PPIB, SERPINH1 , FKBP10, SP7, BMP1 , TMEM38B, WNT1 , CREB3L1 , SPARC, FAM46A or combinations thereof.
- the CNP is administered intravenously, subcutaneously or orally.
- CNP therapy alleviates one or more symptoms of osteogenesis imperfecta, including but not limited to, femoral length, vertebral height, reduction in bone fracture frequency, cortical bone tissue mineral density (TMD), cortical bone mineral density (BMD), cortical bone thickness, cortical porosity, trabecular bone volume fraction (BVF), trabecular TMD, trabecular BMD, trabecular bone number and trabecular bone separation.
- TMD cortical bone tissue mineral density
- BMD cortical bone mineral density
- BMD cortical bone thickness
- cortical porosity cortical porosity
- BVF trabecular bone volume fraction
- the disclosure contemplates a method of treating a subject having osteogenesis imperfecta and receiving C-type natriuretic peptide (CNP) therapy, comprising i) administering CNP therapy to the subject; ii) obtaining a sample from the subject; iii) measuring levels of NTproCNP and/or N terminal fragment of collagen X (CXM) in a sample collected from the subject in (ii); and iv) altering or changing the dose of CNP to bring NTproCNP levels within +/- 2 SDS of mean NTproCNP for the population.
- CNP C-type natriuretic peptide
- the disclosure provides a method of improving and/or maintaining bone strength in a subject in need thereof comprising administering a C-type natriuretic peptide (CNP) to the subject.
- CNP C-type natriuretic peptide
- the subject has osteogenesis imperfecta.
- the CNP is administered daily, between 2 to 5 times per week, or weekly.
- CNP therapy dose level or frequency increases if the level of NTproCNP increases, or CNP therapy dose level decreases if the level of NTproCNP decreases.
- CNP therapy therapy increases the average growth velocity (AGV) in the subject.
- the average growth velocity (AGV) in the subject increases over 6 months, over 1 year or over 2 years, or more.
- increasing CNP therapy dose comprises increasing dose frequency or increasing dose amount.
- an increase in CNP therapy dose level and decrease in NTproCNP level correlate with improved Annualized Growth Velocity (AGV), reduces the frequency of bone fracture, improves bone mineral density, and/or improves femoral length or vertebral growth in subjects.
- AVG Annualized Growth Velocity
- an increase in CNP therapy dose level and decrease in NTproCNP level extends the duration of growth plate activity in the subject.
- the levels of NTproCNP are maintained between 2 standard deviations of mean NTproCNP levels based on population analysis. In various embodiments, the levels of NTproCNP are maintained between +/- 2 SDS of the mean NTproCNP for that population. In various embodiments, the NTproCNP is ⁇ 0.5, ⁇ 1 .0, ⁇ 1 .5 or ⁇ 2.0 standard deviations (SDS) of mean NTproCNP levels of a population to which the subject is grouped.
- SDS standard deviations
- the CNP therapy is titrated toward zero NTproCNP SDS if the NTproCNP SDS is below the mean. In various embodiments, the CNP therapy is titrated until zero NTproCNP SDS. In various embodiments, the CNP therapy is titrated until +0.5, + 1 .0, +1 .5 or +2.0 NTproCNP SDS for the population being treated is achieved. In various embodiments, the zero NTproCNP SDS predicts optimal effect size. Optimal size effect is a measure of the expected average normal growth rate of a subject based on population norms.
- the sample is blood, urine, plasma, saliva, or tissue.
- CNP therapy to treat a subject having osteogenesis imperfecta comprises administration of CNP variants, conjugates, salts or prodrugs thereof.
- the osteogenesis imperfecta is Type I, Type II, Type III, Type IV, or Type V Ol. In various embodiments, the osteogenesis imperfecta is Type VI -Type XXI Ol. In various embodiments, the osteogenesis imperfecta is associated with a mutation in a gene selected from the group consisting of COL1A1 , COL1 A2, COL2A1 , COL1 1 A1 , COL9A2, COL10, IFITM5, SERPINF1 , CRTAP, LEPRE1 , PPIB, SERPINH1 , FKBP10, SP7, BMP1 , TMEM38B, WNT1 , CREB3L1 , SPARC, FAM46A or combinations thereof.
- the CNP variants are useful to treat a subject with osteogenesis imperfecta having a height SDS of less than -1 .0, -1 .5, -2.0, -2.5, or -3.0, and having at least one parent with a height SDS of less than -1 .0, -1 .5, -2.0 or -2.5, optionally wherein the second parent has height within the normal range.
- the CNP variants are useful to treat a subject with short stature having a height SDS of between - 2.0 to -3.0.
- the CNP variants are useful to treat a subject with short stature having a height SDS of between -2.0 to -2.5.
- the CNP is a CNP variant selected from the group consisting of PGQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (Pro-Gly-CNP37) (SEQ ID NO: 1 ); GQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (Gly-CNP-37) (SEQ ID NO: 2);
- KSRAAWARLLQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC CNP-47 (SEQ ID NO: 12); SRAAWARLLQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-46) (SEQ ID NO:
- PGQEHPQARRYRGAQRRGLSRGCFGLKLDRIGSMSGLGC SEQ ID NO: 46
- PGQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC SEQ ID NO: 47
- PGQEHPNARRYRGANRRGLSRGCFGLKLDRIGSMSGLGC SEQ ID NO: 48
- PGQEHPQARKYKGAQKKGLSKGCFGLKLDRIGSMSGLGC SEQ ID NO:49.
- the variant peptide further comprises an acetyl group.
- the acetyl group is on the N-terminus of the peptide.
- the peptide further comprises an OH or an NH 2 group at the C-terminus.
- the CNP variant composition is an extended release composition.
- the composition is a sustained release composition.
- the sustained or extended release compositions comprises a CNP variant pro-drug.
- the variant peptide comprises a conjugate moiety.
- the conjugate moiety is on a residue of the CNP cyclic domain or at a site other than the CNP cyclic domain.
- the conjugate moiety is on a lysine residue.
- the conjugate moiety comprises one or more acid moieties.
- the acid moiety is a hydrophobic acid.
- the conjugate moiety comprises one or more acid moieties linked to a hydrophilic spacer.
- the hydrophilic spacer is any amino acid.
- the hydrophilic spacer is gamma glutamic acid (yGlu).
- the hydrophilic spacer is OEG (8-amino-3,6-dioxaoctanoic acid).
- the hydrophilic spacer is gamma glutamic acid (yGlu) or OEG (8-amino-3,6- dioxaoctanoic acid).
- the hydrophilic spacer is gamma glutamic acid (yGlu) linked to one or two or more OEG (8-amino-3,6-dioxaoctanoic acid).
- the acid moiety is a fatty acid.
- Exemplary fatty acids include short chain, medium chain, or long chain fatty acids, or a dicarboxylic fatty acid.
- the fatty acid is saturated or unsaturated. Contemplated are C-6 to C-20 fatty acids, including but not limited to, C-6, C-8, C-10, C-12, C-14, C-16, C-18 or C-20 fatty acids, saturated or unsaturated.
- the fatty acid is decanoic acid, dodecanoic acid, myristic acid, palmitic acid, stearic acid, arachidic acid, or diacids of the same.
- the acid moiety and the hydrophilic spacer have the structure AEEA-AEEA-yGlu-C18DA.
- the variant comprises one or more linker groups.
- the linker is on a residue of the CNP cyclic domain or at a site other than the CNP cyclic domain. In various embodiments, the linker is on a lysine residue.
- the linker is a hydrolysable linker.
- the conjugate moiety is a synthetic polymeric group.
- the variant comprises a synthetic polymeric group coupled to the variant through a hydrolysable linker.
- the synthetic polymeric group comprises a hydrophilic polymer moiety.
- the hydrophilic polymer moiety comprises polyethylene glycol (PEG).
- the hydrophilic polymer moiety comprises polyethylene glycol (PEG) having a 6 to 20 atom chain length.
- the CNP variant is PGQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (Pro-Gly-CNP37) (SEQ ID NO: 1 ); GQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (Gly-CNP-37) (SEQ ID NO: 2); or LQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-38) (SEQ ID NO: 21 ).
- the variant has the structure: PGQEHPQARRYRGAQRRGLSRGCFGLK(AEEA-AEEA-yGlu-C18DA)LDRIGSMSGLGC (SEQ ID NO: 46), or Ac-PGQEHPQARRYRGAQRRGLSRGCFGLK(AEEA-AEEA-yGlu- C18DA)LDRIGSMSGLGC-OH (SEQ ID NO: 46).
- the variant is selected from the group consisting of
- AC-PGQEHPNARRYRGANRRGLSRGCFGLKLDRIGSMSGLGC-NH 2 SEQ ID NO: 48
- AC-PGQEHPQARRYRGAQRRGLSRGCFGLKLDRIGSMSGLGC-NH 2 SEQ ID NO: 46
- the CNP variant includes a conjugate, salt or prodrug of the CNP variants described herein.
- levels of biomarkers involved in bone and/or collagen growth or formation are measured in a sample before and after administration of CNP.
- the biomarker is type II collagen (CTX), urinary N-telopeptide (NTX), tartrate resistant acid phosphatase (TRAP), bone-specific alkaline phosphatase (BSAP), osteocalcin (OC), N-terminal pro-peptide of type I procollagen (P1 NP), procollagen type 1 C-terminal propeptide (P1CP), NTproCNP or CXM.
- levels of NTproCNP or CXM are measured in a plasma sample, for example, before and after administration of a CNP variant.
- the subject is receiving between 0.5 pg/kg and 60 pg/kg CNP therapy. In various embodiments, the subject is receiving between 1.5 pg/kg and 30 pg/kg CNP therapy, between 2.5 pg/kg and 20 pg/kg CNP therapy, between 5 pg/kg and 20 pg/kg CNP therapy, or between 1 .5 pg/kg and 15 pg/kg CNP therapy. In various embodiments, the subject is receiving 15 pg/kg or 30 pg/kg CNP therapy. In various embodiments, the dose may be increased to 30 pg/kg or 60 pg/kg.
- the subject is receiving about 0.5 pg/kg, about 1 .0 pg/kg, about 1.5 pg/kg, about 2.0 pg/kg, about 2.5 pg/kg, about 3.0 pg/kg, about 3.5 pg/kg, about 4.0 pg/kg, about 4.5 pg/kg, about 5.0 pg/kg, about 6 pg/kg , about 7 pg/kg, about 7.5 pg/kg, about 10 pg/kg, about 12.5 pg/kg, about 15 pg/kg, about 20 pg/kg, about 25 pg/kg, or about 30 pg/kg of CNP or about 60 pg/kg.
- the biomarker is measured at least 4 hours after administration. In various embodiments, the level of biomarker is measured at least 3 months or 6 months after start of CNP therapy. In various embodiments, the level of biomarker is measured at least every 3 months, 6 months, or 1 year after start of CNP therapy. In various embodiments, the level of biomarker is measured for a duration of at least 3 months, 6 months, 1 year, 2 years, 3 years, 4 years, 5 years, or until puberty/close of growth plates after start of CNP therapy. [0052] In various embodiments, the level of biomarker a sample is compared to a baseline measurement taken prior to start of CNP therapy. In various embodiments, the level of biomarker in a sample is compared to average levels in normal control patients.
- CNP therapy dose or frequency is increased when a decrease in NTproCNP indicates an increase in AGV in the subject.
- the level of CXM in a sample is compared to a baseline measurement taken prior to start of CNP therapy. In various embodiments, the level of CXM in a sample is compared to average levels in normal control patients.
- the CXM increase indicates increased bone growth, and wherein the dose of CNP frequency or level is increased when there is CXM increase that enhances AGV, reduces the frequency of bone fracture, improves bone mineral density, improves femoral length and/or vertebral growth.
- the subject is a pediatric subject with open growth plates and receives a dose of about 0.5 pg/kg to about 30 pg/kg daily, five (5) times per week, four (4) times per week, three (3) times per week, two (2) times per week, weekly, bi-weekly, monthly, bi-monthly, or less frequently.
- the subject is in early adolescence and received a dose increase to 15 pg/kg, 30 pg/kg daily or 60 pg/kg daily, twice per week, three times per week, 4 times per week, 5 times, per week, weekly, bi-weekly, monthly, bi-monthly, or less frequently.
- the subject is an infant and received a dose increase to 30 pg/kg daily, twice per week, three times per week, 4 times per week, 5 times, per week, weekly, bi-weekly, monthly, bi-monthly, or less frequently.
- the subject receives 20 pg/kg CNP 5 days per week (20x5), 20 pg/kg CNP 3 days per week (20x3) or 10 pg/kg CNP 3 days per week.
- the subject is suffering from osteogenesis imperfecta.
- the disclosure also provides a method of selecting initiation of CNP therapy in a subject comprising: i) measuring NTproCNP in the subject at multiple timepoints to establish a baseline NTproCNP level; ii) determining if the NTproCNP levels indicate an SDS within ⁇ 2 of mean NTproCNP levels; and iii) starting treatment with CNP therapy when the subject has NTproCNP levels below mean NTproCNP SDS.
- the subject has an NTproCNP SDS of about -2.5, -2.0, -1.5, -1 .0 or -0.5.
- CNP therapy is adjusted such that the NTproCNP SDS of the subject is about -0.4, -0.3, -0.2, -0.1 , 0, 0.1 , 0.2, 0.3, 0.4, 0.5, 0.6, 0.7, 0.8, 0.9, 1 .0, 1 .1 , 1 .2, 1 .3, 1 .4, .5, .16, 1 .7, 1 .8, 1 .9 or 2.0 after a modulation in CNP therapy dose level and/or frequency.
- the disclosure contemplates a method of selecting initiation of CNP therapy in a subject having osteogenesis imperfecta comprising i) measuring NTproCNP in the subject at multiple timepoints to establish a baseline NTproCNP level; ii) determining if the NTproCNP levels indicate an SDS of zero, below zero or above zero; and iii) starting treatment with CNP therapy when the subject has NTproCNP levels above SDS zero.
- NTproCNP is measured at 2 weeks, one month, 3 months, and 6 months prior to CNP therapy to establish a baseline NTproCNP level. In various embodiments, NTproCNP is measured by radioimmunoassay.
- the subject has a PRS in the bottom 20%, 19%, 18%, 17.5%, 17%, 16.5%, 16%, 15.5%, 15%, 14.5%, 14%, 13.5%, 13%, 12.5%, 12%, 11%, 10%, 9%, 8%, 7.5%, 7%, 6%, 5%, 4%, 3%, 2.5%, 2% or 1 %.
- step iii) and iv) is a subject with a CNP variant if the subject has a LoF variant and a PRS in the bottom 12.5%.
- the PRS is calculated by a genome-wide association study (GWAS) of height.
- a PRS is an aggregate genetic score that consists of many, common variant effects of small effect each that are summed across the genome (Choi et al. Nat Protoc, 2020).
- GWAS Genome Wide Association Study
- association statistics are obtained to indicate the per-variant strength of association with height.
- PRS can be interpreted such that an individual with a low PRS carries a lower-than-average number of height-increasing genetic variants, and an individual with a high PRS carries a higher-than-average number of heightincreasing variants.
- the increase in facial volume, facial sinus volume, and foramen magnum area are measured by magnetic resonance imaging (MRI).
- MRI magnetic resonance imaging
- the change in facial volume, facial sinus volume, and foramen magnum area are compared to baseline levels, healthy control subjects or untreated control subjects.
- the CNP variant is administered subcutaneously. In various embodiments, the CNP variant is administered daily, twice per week, three times per week, 5 times per week, weekly, every 2 weeks, monthly, or less. In various embodiments, the CNP variant is administered at a dose of 30 pg/kg for 3 months, 6 months, 1 year or more. In various embodiments, the dose of CNP variant is decreased to 15 pg/kg when the subject is about 2 years old.
- each feature or embodiment, or combination, described herein is a non-limiting, illustrative example of any of the aspects of the invention and, as such, is meant to be combinable with any other feature or embodiment, or combination, described herein.
- each of these types of embodiments is a nonlimiting example of a feature that is intended to be combined with any other feature, or combination of features, described herein without having to list every possible combination.
- Figure 2 shows CNP levels (left) and NTproCNP levels (right) as measured by Z- scores compared to the normal ranges as a function of Ol clinical classification.
- Figures 3A and 3B illustrate femoral length in mouse model of osteogenesis imperfecta after treatment with CNP +bisphosphonate.
- Figure 4 shows the difference in trabecular and cortical bone of oim/oim animals receiving CNP+bisphosphonate therapy.
- Figure 5 shows micro-CT analysis of various parameters of cortical and trabecular bone in treated and untreated oim/oim mice.
- Figure 6 shows 3D images of trabecular bone in treated and untreated oim/oim mice.
- Figure 7 shows 3D images of cortical bone in treated and untreated oim/oim mice.
- Figure 8 shows mean cortical bone mineral density in treated and untreated oim/oim mice.
- Figure 9 shows mean cortical porosity in treated and untreated oim/oim mice.
- Figure 10 shows mean cortical thickness in treated and untreated oim/oim mice.
- Figure 11 shows mean trabecular bone mineral density in treated and untreated oim/oim mice.
- Figure 12 shows mean trabecular number in treated and untreated oim/oim mice.
- Figure 13 shows mean trabecular thickness in treated and untreated oim/oim mice.
- Figure 14 shows mean trabecular separation in treated and untreated oim/oim mice.
- Figure 15 shows femoral length in WT and oim/oim mice treated with CNP.
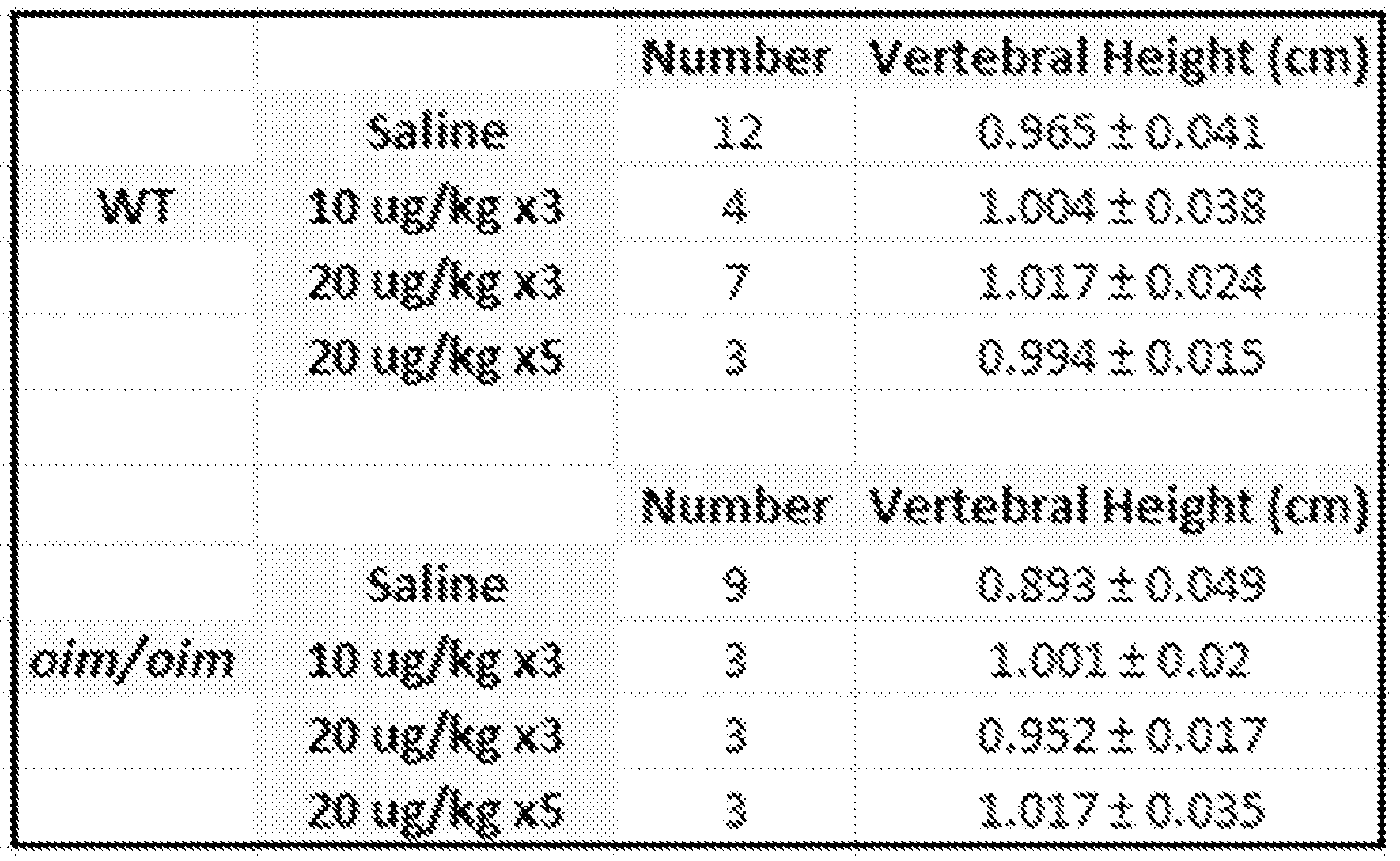
- Figure 16 shows vertebral height in WT and oim/oim mice treated with CNP.
- Figures 17A-17E show effects of CNP administration on WT cortical bone by micro-CT analysis: Figure 17A, cortical tissue mineral density; Figure 17B, cortical porosity; Figure 17C, cortical bone mineral density; Figure 17D, cortical thickness; Figure 17E, cortical minimum inertia.
- Figures 18A-18F show effects of CNP administration on WT trabecular bone by micro- CT analysis: Figure 18A, trabecular bone volume fraction; Figure 18B, trabecular tissue mineral density; Figure 18C, trabecular bone mineral density; Figure 18D, trabecular number; Figure 18E, trabecular thickness; Figure 18F, trabecular separation.
- Figures 19A-19E shows effects of CNP administration on oim/oim cortical bone by micro-CT analysis:
- Figure 19A cortical tissue mineral density
- Figure 19B cortical porosity
- Figure 19C cortical bone mineral density
- Figure 19D cortical thickness
- Figure 19E cortical minimum inertia.
- Figure 20A-20F shows effects of CNP administration on oim/oim trabecular bone by micro-CT analysis:
- Figure 20A trabecular bone volume fraction;
- Figure 20B trabecular tissue mineral density;
- Figure 20C trabecular bone mineral density;
- Figure 20D trabecular number;
- Figure 20E trabecular thickness;
- Figure 20F trabecular separation.
- Figure 21 is a table showing the effects of CNP therapy on WT and oim/oim mice in cortical and trabecular bone.
- CNP C-type natriuretic peptide
- CNP variants to patients having osteogenesis imperfecta can alleviate symptoms of diseases, such as bone mineral density, femoral length, and vertebral height.
- the term “about” or “approximately” means an acceptable error for a particular value as determined by one of ordinary skill in the art, which depends in part on how the value is measured or determined. In certain embodiments, the term “about” or “approximately” means within 1 , 2, 3, or 4 standard deviations. In certain embodiments, the term “about” or “approximately” means within 30%, 25%, 20%, 15%, 10%, 9%, 8%, 7%, 6%, 5%, 4%, 3%, 2%, 1%, 0.5%, or 0.05% of a given value or range. Whenever the term “about” or “approximately” precedes the first numerical value in a series of two or more numerical values, it is understood that the term “about” or “approximately” applies to each one of the numerical values in that series.
- C-type natriuretic peptide or “CNP” refers to a small, single chain peptide having a 17-amino acid loop structure at the C-terminal end (GenBank Accession No.
- NP 077720 for the CNP precursor protein, NPPC) and variants thereof.
- the 17-mer CNP loop structure is also referred to as CNP 17, the CNP ring, or CNP cyclic domain.
- CNP includes the active 53-amino acid peptide (CNP-53) and the mature 22-amino acid peptide (CNP-22), and peptides of varying lengths between the two peptides.
- a “CNP variant” is at least about 40%, 50%, 60%, 70%, 75%, 80%, 85%, 90%, or 95% homologous to the wild type NPPC over the same number of amino acid residues. It is further contemplated that a CNP variant peptide may comprise from about 1 to about 53, or 1 to 39, or 1 to 38, or 1 to 37, or 1 to 35, or 1 to 34, or 1 to 31 , or 1 to 27, or 1 to 22, or 10 to 35, or about 15 to about 37 residues of the NPPC polypeptide.
- a CNP variant may comprise a sequence of 1 , 2, 3, 4, 5, 6, 7, 8, 9, 10, 1 1 , 12, 13, 14, 15, 16, 17, 18, 19, 20, 21 , 22, 23, 24, 25, 26, 27, 28, 29, 30, 31 , 32, 33, 34, 35,36, 37, 38, 39, 40, 41 , 42, 43, 44, 45, 46, 47, 48, 49, 50, 51 , 52, or 53 amino acids derived from the NPPC polypeptide.
- CNP variant also includes conjugates, salts or prodrugs of CNP variants described herein.
- CNP therapy refers to administration of a CNP variant to treat a subject having a bone-related disorder, skeletal dysplasia or short stature as described herein.
- conjugate moiety refers to a moiety that is conjugated to the variant peptide.
- Conjugate moieties include a lipid, fatty acid, hydrophilic spacer, synthetic polymer, linker, or optionally, combinations thereof.
- the term “effective amount” refers to a dosage sufficient to produce a desired result on a health condition, pathology, or disease of a subject or for a diagnostic purpose.
- the desired result may comprise a subjective or objective improvement in the recipient of the dosage.
- “Therapeutically effective amount” refers to that amount of an agent effective to produce the intended beneficial effect on health.
- An appropriate “effective” amount in any individual case may be determined by one of ordinary skill in the art using routine experimentation.
- the specific dose level and frequency of dosage for any particular patient may be varied and will depend upon a variety of factors, including the activity of the specific compound employed; the bioavailability, metabolic stability, rate of excretion and length of action of that compound; the mode and time of administration of the compound; the age, body weight, general health, sex, and diet of the patient; and the severity of the particular condition.
- substantially pure or “isolated” means an object species is the predominant species present (/.e., on a molar basis, more abundant than any other individual macromolecular species in the composition), and a substantially purified fraction is a composition wherein the object species comprises at least about 50% (on a molar basis) of all macromolecular species present.
- a substantially pure composition means that the species of interest comprises at least about 70%, 75%, 80%, 85%, 90%, 95%, 98% or more of the macromolecular species present in the composition on a molar or weight basis.
- the object species is purified to essential homogeneity (contaminant species cannot be detected in the composition by conventional detection methods) if the composition consists essentially of a single macromolecular species.
- Solvent species, small molecules ( ⁇ 500 Daltons), stabilizers (e.g., BSA), and elemental ion species are not considered macromolecular species for purposes of this definition.
- the compounds of the disclosure are substantially pure or isolated.
- the compounds of the disclosure are substantially pure or isolated with respect to the macromolecular starting materials used in their production.
- the pharmaceutical compositions of the disclosure comprise a substantially pure or isolated CNP variant admixed with one or more pharmaceutically acceptable excipients, carriers or diluents, and optionally with another biologically active agent.
- Treatment refers to prophylactic treatment or therapeutic treatment or diagnostic treatment.
- treatment refers to administration of a compound or composition to a subject for therapeutic, prophylactic or diagnostic purposes.
- a "prophylactic" treatment is a treatment administered to a subject who does not exhibit signs of a disease or exhibits only early signs of the disease, for the purpose of decreasing the risk of developing pathology.
- the compounds or compositions of the disclosure may be given as a prophylactic treatment to reduce the likelihood of developing a pathology or to minimize the severity of the pathology, if developed.
- a "therapeutic” treatment is a treatment administered to a subject who exhibits signs or symptoms of pathology for the purpose of diminishing or eliminating those signs or symptoms.
- the signs or symptoms may be biochemical, cellular, histological, functional or physical, subjective or objective.
- the compounds of the disclosure may also be given as a therapeutic treatment or for diagnosis.
- Diagnostic means identifying the presence, extent and/or nature of a pathologic condition. Diagnostic methods differ in their specificity and selectivity. While a particular diagnostic method may not provide a definitive diagnosis of a condition, it suffices if the method provides a positive indication that aids in diagnosis.
- “Bone- or cartilage-associated biomarker” or “bone- or cartilage-associated marker” refers to a growth factor, enzyme, protein, or other detectable biological substance or moiety whose level is increased or decreased in association with, e.g., cartilage turnover, cartilage formation, cartilage growth, bone resorption, bone formation, bone growth, or combinations thereof. Such biomarkers may be measured before, during and/or after administration of a CNP variant as described herein.
- Exemplary bone- or cartilage-associated biomarkers include, but are not limited to, CNP, cGMP, propeptides of collagen type II and fragments thereof, collagen type II and fragments thereof, propeptides of collagen type I and fragments thereof, collagen type I and fragments thereof, osteocalcin (OC), proliferating cell nuclear antigen (PCNA), aggrecan chondroitin sulfate, collagen X, N terminal fragment of collagen X (CXM) alkaline phosphatase, type II collagen (CTX), urinary N-telopeptide (NTX), tartrate resistant acid phosphatase (TRAP), bone-specific alkaline phosphatase (BSAP), N-terminal pro-peptide of type I procollagen (P1 NP), procollagen type 1 C-terminal propeptide (P1 CP), or NTproCNP.
- Cartilage- and bone-associated biomarkers can be measured in any appropriate biological sample, including but not limited to tissues, blood, serum, plasma, cere
- composition refers to a composition suitable for pharmaceutical use in subject animal, including humans and mammals.
- a pharmaceutical composition comprises a therapeutically effective amount of CNP variant, optionally another biologically active agent, and optionally a pharmaceutically acceptable excipient, carrier or diluent.
- a pharmaceutical composition encompasses a composition comprising the active ingredient(s), and the inert ingredient(s) that make up the carrier, as well as any product that results, directly or indirectly, from combination, complexation or aggregation of any two or more of the ingredients, or from dissociation of one or more of the ingredients, or from other types of reactions or interactions of one or more of the ingredients.
- the pharmaceutical compositions of the present disclosure encompass any composition made by admixing a compound of the disclosure and a pharmaceutically acceptable excipient, carrier or diluent.
- “Pharmaceutically acceptable carrier” refers to any of the standard pharmaceutical carriers, buffers, and the like, such as a phosphate buffered saline solution, 5% aqueous solution of dextrose, and emulsions (e.g., an oil/water or water/oil emulsion).
- excipients include adjuvants, binders, fillers, diluents, disintegrants, emulsifying agents, wetting agents, lubricants, glidants, sweetening agents, flavoring agents, and coloring agents.
- Suitable pharmaceutical carriers, excipients and diluents are described in Remington's Pharmaceutical Sciences, 19th Ed.
- Preferred pharmaceutical carriers depend upon the intended mode of administration of the active agent. Typical modes of administration include enteral (e.g., oral) or parenteral (e.g., subcutaneous, intramuscular, intravenous or intraperitoneal injection; or topical, transdermal, or transmucosal administration).
- enteral e.g., oral
- parenteral e.g., subcutaneous, intramuscular, intravenous or intraperitoneal injection; or topical, transdermal, or transmucosal administration.
- a "pharmaceutically acceptable salt” is a salt that can be formulated into a compound for pharmaceutical use, including but not limited to metal salts (e.g., sodium, potassium, magnesium, calcium, etc.) and salts of ammonia or organic amines.
- metal salts e.g., sodium, potassium, magnesium, calcium, etc.
- salts of ammonia or organic amines e.g., sodium, potassium, magnesium, calcium, etc.
- pharmaceutically acceptable or “pharmacologically acceptable” is meant a material that is not biologically or otherwise undesirable, i.e., the material may be administered to an individual without causing any undesirable biological effects or without interacting in a deleterious manner with any of the components of the composition in which it is contained or with any components present on or in the body of the individual.
- Physiological conditions refer to conditions in the body of an animal (e.g., a human). Physiological conditions include, but are not limited to, body temperature and an aqueous environment of physiologic ionic strength, pH and enzymes. Physiological conditions also encompass conditions in the body of a particular subject which differ from the “normal” conditions present in the majority of subjects, e.g., which differ from the normal human body temperature of approximately 37 °C or differ from the normal human blood pH of approximately 7.4.
- physiological pH or a “pH in a physiological range” is meant a pH in the range of approximately 7.0 to 8.0 inclusive, more typically in the range of approximately 7.2 to 7.6 inclusive.
- the term “subject” encompasses mammals and non-mammals.
- mammals include, but are not limited to, any member of the mammalian class: humans, non-human primates such as chimpanzees, and other apes and monkey species; farm animals such as cattle, horses, sheep, goats, swine; domestic animals such as rabbits, dogs, and cats; laboratory animals including rodents, such as rats, mice and guinea pigs, and the like.
- non-mammals include, but are not limited to, birds, fish, and the like.
- the term does not denote a particular age or gender.
- the subject is human.
- the subject is a child or adolescent.
- the subject is an infant.
- the subject is older than 3, older than 2, older than 1 , or older than 6 months in age.
- C-type Natriuretic Peptide C-type natriuretic peptide (CNP) (Biochem. Biophys. Res. Common., 168: 863-870 (1990) (Gen Bank Accession No. NP 077720, for the CNP precursor protein, NPPC) (J. Hypertens., 10: 907-912 (1992)) is a small, single chain peptide in a family of peptides (ANP, BNP, CNP) having a 17-amino acid loop structure (Levin et al., N. Engl. J. Med., 339:863-870 (1998)) and have important roles in multiple biological processes.
- CNP interacts with natriuretic peptide receptor-B (NPR-B, GC-B) to stimulate the generation of cyclic- guanosine monophosphate (cGMP) (J. Hypertens., 10:11 11 -11 14 (1992)).
- CNP is expressed more widely, including in the central nervous system, reproductive tract, bone and endothelium of blood vessels (Gardner et al., Hypertension, 49:419-426 (2007)).
- CNP is initially produced from the natriuretic peptide precursor C (NPPC) gene as a single chain 126-amino acid pre-pro polypeptide (Sudoh et al., Biochem. Biophys. Res. Common., 168: 863-870 (1990)). Removal of the signal peptide yields pro-CNP, and further cleavage by the endoprotease furin generates an active 53-amino acid peptide (CNP- 53), which is secreted and cleaved again by an unknown enzyme to produce the mature 22- amino acid peptide (CNP-22) (Wu, J. Biol. Chem. 278: 25847-852 (2003)).
- NPPC natriuretic peptide precursor C
- CNP-53 and CNP- 22 differ in their distribution, with CNP-53 predominating in tissues, while CNP-22 is mainly found in plasma and cerebrospinal fluid (J. Alfonzo, Recept. Signal. Transduct. Res., 26: 269- 297 (2006)). Both CNP-53 and CNP-22 bind similarly to NPR-B.
- Downstream signaling mediated by cGMP generation influences a diverse array of biological processes that include endochondral ossification.
- knockout of either CNP or NPR-B in mouse models results in animals having a dwarfed phenotype with shorter long bones and vertebrae.
- Mutations in human NPR-B that block proper CNP signaling have been identified and result in dwarfism (Olney, et al., J. Clin. Endocrinol. Metab. 91 (4): 1229- 1232 (2006); Bartels, et al., Am. J. Hum. Genet. 75: 27-34 (2004)).
- mice engineered to produce elevated levels of CNP display elongated long bones and vertebrae.
- CNP of the disclosure includes truncated CNP ranging from human CNP-17 (hCNP-17) to human CNP-53 (hCNP-53), and having wild-type amino acid sequences derived from hCNP-53 and also variants thereof.
- CNP contemplated herein also includes neutral endopeptidase resistant CNP which are >30 amino acids, and CNP having a size >3 kDa.
- Truncated CNP peptides include:
- PGQEHPQARRYRGAQRRGLSRGCFGLKLDRIGSMSGLGC SEQ ID NO: 46
- PGQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC SEQ ID NO: 47
- PGQEHPNARRYRGANRRGLSRGCFGLKLDRIGSMSGLGC SEQ ID NO: 48
- PGQEHPQARKYKGAQKKGLSKGCFGLKLDRIGSMSGLGC SEQ ID NO: 49.
- the CNP variant peptides are modified CNP-37 or CNP-38 peptides, optionally having mutation(s)/substitution(s) at the furin cleavage site, and/or containing glycine or proline-glycine at the N-terminus.
- Exemplary CNP-37 variants include but are not limited to:
- GQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC GQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (Gly-CNP-37: SEQ ID NO: 2)
- GQEHPNARKYKGANPKGLSKGCFGLKLDRIGSMSGLGC SEQ ID NO: 50
- GQEHPNARKYKGANQKGLSKGCFGLKLDRIGSMSGLGC SEQ ID NO: 51 );
- CNP variants of the disclosure include PGQEHPQARRYRGAQRRGLSRGCFGLKLDRIGSMSGLGC (SEQ ID NO: 46); PGQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (SEQ ID NO: 47); PGQEHPNARRYRGANRRGLSRGCFGLKLDRIGSMSGLGC (SEQ ID NO: 48); or PGQEHPQARKYKGAQKKGLSKGCFGLKLDRIGSMSGLGC (SEQ ID NO: 49).
- the variant peptide may further comprise an acetyl group.
- the acetyl group is on the N-terminus of the peptide.
- the peptide further comprises an OH or an NH 2 group at the C-terminus.
- the variant peptide may comprise a conjugate moiety.
- the conjugate moiety is on a residue of the CNP cyclic domain or at a site other than the CNP cyclic domain.
- the conjugate moiety is on a lysine residue.
- the conjugate moiety comprises one or more acid moieties.
- the acid moiety is a hydrophobic acid.
- the variant has the structure: PGQEHPQARRYRGAQRRGLSRGCFGLK(AEEA-AEEA-yGlu-C18DA)LDRIGSMSGLGC (SEQ ID NO: 46), or Ac-PGQEHPQARRYRGAQRRGLSRGCFGLK(AEEA-AEEA-yGlu-
- the variant is selected from the group consisting of
- the CNP variant is Ac-
- the CNP variant is Ac-
- the CNP variant is PGQEHPNARKYKGANKKGLSKGCFGLK(AEEA-AEEA-yGlu-C18DA)LDRIGSMSGLGC-OH (SEQ ID NO: 47).
- the CNP variant is PGQEHPNARKYKGANKKGLSKGCFGLK(AEEA-AEEA-yGlu-C18DA)LDRIGSMSGLGC-OH (SEQ ID NO: 47).
- the CNP variant is conjugated to or is complexed to a moiety, e.g., a conjugate moiety, that confers increased stability or half-life.
- a conjugate moiety is complexed via a non-covalent bond or is attached by a covalent bond.
- the moiety may be non-covalently attached with the peptide via electrostatic interactions.
- the moiety may be covalently associated to the peptide via one or more linker moieties.
- Linkers can be cleavable and non-cleavable linkers.
- Cleavable linkers may be cleaved via enzymes, nucleophilic/basic reagents, reducing agents, photo-irradiation, electrophilic/acidic reagents, organometallic and metal reagents, or oxidizing reagents.
- Linkers may also be self-immolative linkers.
- linkers include, but are not limited to, N- succinim idyl-3-(2-pyridyldith iol) propionate (SPDP), iminothiolane (IT), bifunctional derivatives of imidoesters (such as dimethyl adipimidate HCL), active esters (such as disuccinimidyl suberate), aldehydes (such as glutaraldehyde), bis-azido compounds (such as bis (p- azidobenzoyl) hexanediamine), bis-diazonium derivatives (such as bis-(p-diazoniumbenzoyl)- ethylenediamine), diisocyanates (such as tolyene 2,6-diisocyanate), and bis-active fluorine compounds (such as 1 ,5-difluoro-2,4-dinitrobenzene), beta alanine, 4-aminobutyric acid (GABA), 2-aminoethoxy acid (G
- the linker is attached to a residue of the CNP variant within the CNP cyclic domain or at a site other than the CNP cyclic domain. In various embodiments, the linker is attached to a lysine residue. In various embodiments, the linker is attached to a lysine residue in the CNP cyclic domain.
- the CNP variant is attached to the conjugate moiety via the linker.
- the linker is attached to the conjugate moiety via the hydrophilic spacer of the conjugate moiety.
- the linker is a hydrolysable linker.
- the linker is a peptoid or electronic linker. In various embodiments the linker is a peptoid linker. In various embodiments the linker is an electronic linker. In various embodiments, the linker comprises an SO2 moiety. It is further contemplated that linkers are modified by substitution on the R groups.
- peptoid-type linkers include the structures as set out below:
- the moiety conjugated to the peptide is a synthetic polymer such as polyethylene glycol, a linker, a lipid moiety or fatty acid, or a combination thereof.
- the CNP variant is conjugated with a fatty acid, an amino acid, a spacer and a linker.
- the CNP variant is conjugated with a fatty acid, an amino acid, a polyethylene glycol spacer or a polyethylene glycol derivative spacer, and a linker.
- the CNP variant is conjugated with a fatty acid, an amino acid, a spacer, and a linker, wherein the spacer comprises a substituted C-6 to C-20 alkyl chain or any amino acid, or a combination of both, wherein the carbon atoms of the alkyl chain can be replaced by one or more of O, NH, N(C-1 to C-6 alkyl), or carbonyl groups.
- the CNP variant is conjugated with a fatty acid. It is hypothesized that the lipid technology increases the serum half-life of the CNP variant allowing for less frequent injections and/or improved oral delivery.
- the fatty acid is a short chain, medium chain, long chain fatty acid, or a dicarboxylic fatty acid.
- the fatty acid is saturated or unsaturated.
- the fatty acid is a C-6 to C-20 fatty acid.
- the fatty acid is a C-6, C-8, C-10, C-12, C- 14, C-16, C-18 or C-20 fatty acid.
- the fatty acid is decanoic acid, dodecanoic acid, myristic acid, palmitic acid, stearic acid, arachidic acid, or diacids of the same. In various embodiments, the fatty acid is conjugated to a lysine residue.
- the CNP variants described herein comprise a conjugate moiety as described herein. It is contemplated that the conjugate moiety is on a residue of the CNP cyclic domain or at a site other than the CNP cyclic domain. In various embodiments, the conjugate moiety is on a lysine residue. In various embodiments, the conjugate moiety comprises one or more acid moieties. In various embodiments, the acid moiety is a fatty acid. Exemplary CNP variants and peptide conjugates are described in International Patent Application No. PCT/US2020/051100 and USSN 17/642,150, incorporated by reference herein in their entirety. Variants, conjugates and salts of CNP are disclosed in USSN 17/634,034, herein incorporated by reference.
- the conjugate moiety comprises an acid moiety linked to a hydrophilic spacer.
- the hydrophilic spacer is a substituted C-6 to C-20 alkyl chain or any amino acid, or a combination of both, wherein the carbon atoms of the alkyl chain can be replaced by one or more of O, NH, N(C-1 to C-6 alkyl), or carbonyl groups.
- the hydrophilic spacer is any amino acid.
- the hydrophilic spacer is gamma glutamic acid (yGlu) .
- the hydrophilic spacer is a substituted C-6 to C-20 alkyl chain.
- the hydrophilic spacer is a substituted C-6, C-8, C-10, C-12, C-14, C-16, C-18 or C-20 alkyl chain. In various embodiments, the hydrophilic spacer is a substituted C-9 to C-18 alkyl chain. In various embodiments, the hydrophilic spacer is a substituted C-18 alkyl chain. In various embodiments, the hydrophilic spacer is a substituted C-9 alkyl chain. In various embodiments, the hydrophilic spacer is one or more OEG (8-amino-3,6-dioxaoctanoic acid) groups.
- the hydrophilic spacer is one or two OEG (8-amino-3,6-dioxaoctanoic acid) groups. In various embodiments, the hydrophilic spacer is OEG (8-amino-3,6-dioxaoctanoic acid). In various embodiments, the spacer is OEG (8-amino-3,6-dioxaoctanoic acid) or yGlu. In various embodiments, the hydrophilic spacer is gamma glutamic acid (yGlu) linked to one or more OEG (8-amino-3,6-dioxaoctanoic acid) groups.
- yGlu gamma glutamic acid
- the hydrophilic spacer is gamma glutamic acid (yGlu) linked to one or two OEG (8-amino-3,6-dioxaoctanoic acid) groups (diEG).
- yGlu gamma glutamic acid
- OEG 8-amino-3,6-dioxaoctanoic acid
- the acid moiety and the hydrophilic spacer have the structure AEEA-AEEA-yGlu-C18DA.
- CNP variants comprising hydrophilic or water soluble polymers (e.g., oxygenated alkyl chains, wherein the carbon atoms can be replaced with one or more oxygen atoms, such as polyethylene glycol (PEG) or polyethylene oxide (PEG) and the like).
- hydrophilic or water soluble polymers e.g., oxygenated alkyl chains, wherein the carbon atoms can be replaced with one or more oxygen atoms, such as polyethylene glycol (PEG) or polyethylene oxide (PEG) and the like.
- the water soluble polymers can vary in type (e.g., homopolymer or copolymer; random, alternating or block copolymer; linear or branched; monodispersed or polydispersed), linkage (e.g., hydrolysable or stable linkage such as, e.g., amide, imine, aminal, alkylene, or ester bond), conjugation site (e.g., at the N-terminus, internal, and/or C-terminus), and length (e.g., from about 0.2, 0.4 or 0.6 kDa to about 2, 5, 10, 25, 50 or 100 kDa).
- linkage e.g., hydrolysable or stable linkage such as, e.g., amide, imine, aminal, alkylene, or ester bond
- conjugation site e.g., at the N-terminus, internal, and/or C-terminus
- length e.g., from about 0.2, 0.4 or 0.6 kDa to about
- the hydrophilic or water-soluble polymer can be conjugated to the CNP variant by means of N-hydroxy succinimide (NHS)- or aldehyde-based chemistry or other chemistry, as is known in the art.
- N-hydroxy succinimide NHS
- negatively charged PEG-CNP variants can be designed for reduced renal clearance, including but not limited to use of carboxylated, sulfated and phosphorylated compounds (Caliceti, Adv. Drug Deliv. Rev., 55: 1261 -77 (2003); Perlman, J. Clin. Endo. Metab., 88: 3227-35 (2003); Pitkin, Antimicrob. Ag.
- the PEG (or PEG) moiety contains carboxyl group(s), sulfate group(s), and/or phosphate group(s).
- the hydrophilic polymer (e.g., PEG or PEG) moieties conjugated to the N-terminus, C-terminus and/or internal site(s) of CNP variants described herein contain one or more functional groups that are positively charged under physiological conditions. Such moieties are designed, inter alia, to improve distribution of such conjugated CNP variants to cartilage tissues.
- PEG moieties contain one or more primary, secondary or tertiary amino groups, quaternary ammonium groups, and/or other amine- containing (e.g., urea) groups.
- Achondroplasia is a result of an autosomal dominant mutation in the gene for fibroblast growth factor receptor 3 (FGFR-3), which causes an abnormality of cartilage formation.
- FGFR-3 normally has a negative regulatory effect on chondrocyte growth, and hence bone growth.
- the mutated form of FGFR-3 is constitutively active, which leads to severely shortened bones.
- activating mutations of FGFR-3 are the primary cause of genetic dwarfism.
- Mice having activated FGFR-3 serve as a model of achondroplasia, the most common form of the skeletal dysplasias, and overexpression of CNP rescues these animals from dwarfism. Accordingly, functional variants of CNP are potential therapeutics for treatment of the various skeletal dysplasias.
- the CNP variants of the disclosure are useful for treating mammals, including humans, suffering from a bone-related disorder, such as a skeletal dysplasia or short stature.
- Non-limiting examples of CNP-responsive bone-related disorders skeletal dysplasias and short stature disorders include achondroplasia, hypochondroplasia, short stature, idiopathic short stature, dwarfism, osteochondrodysplasias, thanatophoric dysplasia, osteogenesis imperfecta, osteogenesis congenita, achondrogenesis, homozygous achondroplasia, chondrodysplasia congenita, campomelic dysplasia, congenital lethal hypophosphatasia, perinatal lethal type of osteogenesis congenita, short-rib polydactyly syndromes, rhizomelic type of chondrodysplasia congenita, Jansen-type metaphyseal dysplasia, spondyloepiphyseal dysplasia congenital, atelosteogenesis, diastrophic dysplasia, congenital short femur, Langer-type mesomelic
- Additional short stature and growth plate disorders contemplated by the methods include disorders related to mutations in collagen (COL2A1 , COL11 A1 , COL9A2, COL10), aggrecan (ACAN), Indian hedgehog (IHH), PTPN11 , NPR2, NPPC, FGFR3, or IGF1 R SHOX, DTL, and pregnancy-associated plasma protein A2 (PAPPA2), or combinations thereof.
- the CNP variants are useful as an adjunct or alternative to growth hormone for treating idiopathic short stature and other skeletal dysplasias.
- Growth plate disorders include disorders that result in short stature or abnormal bone growth and that may be the result of a genetic mutation in a gene involved in bone growth, including collagen (COL2A1 , COL11A1 , COL9A2, COL10), aggrecan (ACAN), indian hedgehog (IHH), PTPN11 , NPR2, NPPC, FGFR3 or IGF1 R.
- the growth plate disorder or short stature is associated with one or more mutations in a gene associated with a RASopathy.
- a subject with a growth plate disorder is heterozygous for a mutation in a growth plate gene.
- the mutation is a loss-of-function mutation.
- the mutation is a gain-of-function mutation.
- Growth plate disorders include, but are not limited to, familial short stature, dominant familial short stature which is also known as dominant inherited short stature, or idiopathic short stature. See, e.g., Plachy et al., J Clin Endocrinol Metab 104: 4273-4281 , 2019.
- ACAN can give rise to familial osteochondritis dissecans and short stature and eventually osteoarthritis, characterized by areas of bone damage (or lesions) caused by the detachment of cartilage and sometimes bone from the end of the bone at a joint. It has been suggested that the disorganized cartilage network in growing bones impairs their growth, leading to short stature.
- a mutation associated with ACAN and short stature includes Val2303Met. See Stattin et al., Am J Hum Genet 86(2):126-37, 2010. It is contemplated that patients with a mutation in ACAN resulting in short stature would benefit from treatment with CNP as administration may be able to increase height in these patients by the known interaction of CNP with FGFR3.
- the natriuretic peptide system including receptor NPR2, has been shown to be involved in regulation of endochondral bone growth (Vasques et al., orm Res Pediat 82:222- 229, 2014). Studies have shown that homozygous or compound heterozygous loss-of-function mutations in NPR2 cause acromesomelic dysplasia type Maroteaux (AMDM), which is a skeletal dysplasia having extremely short stature (Vasquez et al., 2014, supra).
- AMDM acromesomelic dysplasia type Maroteaux
- NPR2 Heterozygous mutations of NPR2 are believed to result in idiopathic short stature and other forms of short stature. Mutations in the NPR2 gene are set out below and described in Amano et al., J Clin Endocrinol Metab 99:E713-718, 2014, Hisado-Oliva et al., J Clin Endocrinol Metab 100:E1 133-1 142, 2015 and Vasques et al., J Clin Endocrinol Metab 98:E1636-1644, 2013, hereby incorporated by reference.
- a subject having short stature to be treated with a CNP variant as described herein has a height SDS of less than -1 .0, -1.5, - 2.0, -2.5, or -3.0, and has at least one parent with a height SDS of less than -1.0, -1 .5, -2.0 or - 2.5, optionally wherein the second parent has height within the normal range.
- the CNP variants are useful to treat a subject with short stature having a height SDS of between -2.0 to -3.0.
- the CNP variants are useful to treat a subject with short stature having a height SDS of between -2.0 to -2.5.
- NPPC neuropeptide-derived protein
- NPPC knock out mouse showed severe disproportionate form of dwarfism including shortening of limbs and endochondral ossification (Hisado-Oliva et al., 2018, supra).
- Human genome wide studies have shown a link between NPPC and height (Hisado-Oliva et al., 2018, supra).
- CNP haploinsufficiency has been believed to be a cause of short stature in humans
- a recent study identified heterozygous mutations in families with short stature and hands (Hisado-Oliva et al., 2018, supra). These studies observed significant reduction in cGMP production as measured in heterozygous state (Hisado-Oliva et al., 2018, supra).
- Mutations in NPPC include a 355G>T missense mutation causing a Gly119Cys change and a 349C>G missense mutation causing a Arg1 17Gly change.
- a CNP variant rescuing CGMP production may provide therapeutic benefit in the management of a disorder in patients having heterozygous loss-of-function NPPC mutations.
- LWD Leri-Weill dyschondrosteosis
- SHOX short stature homeobox-containing gene or its regulatory elements located on the pseudoautosomal region 1 (PAR1 ) of the sex chromosomes.
- the disorder Langer mesomelic dysplasia arises when there are two SHOX mutations, and may result from a mutation on each chromosome, either a homozygous or compound heterozygous mutations.
- a subset of SHOX mutations give rise to idiopathic short stature.
- Turner syndrome results due to a deletion on the X chromosome that can include the SHOX gene.
- SHOX has been identified as involved in the regulation of FGFR3 transcription and contributes to control of bone growth (Marchini et al., Endocr Rev. 37: 417-448, 2016).
- SHOX deficiency leads to increased FGFR3 signaling, and there is some evidence to support that SHOX has direct interactions with CNP/NPR2 as well (Marchini, supra). Given the association of SHOX with FGFR3 and bone growth, it is contemplated that a subject having a homozygous or heterozygous SHOX mutation would benefit from treatment with CNP variants as described herein.
- RASopathies are a group of rare genetic conditions caused by mutations in genes of the Ras/mitogen-activated protein kinase (MAPK) pathway.
- RASopathies are a group of disorders characterized by increased signaling through RAS/MAPK pathway. This pathway leads to downstream activation of the RAF/MEK/ERK pathway. Short stature is a characteristic feature of certain RASopathies. For example, CNP signaling inhibits RAF and leads to decreased MEK and ERK activation.
- RASopathies associated with short stature include Noonan syndrome, Costello syndrome, Cardiofaciocutaneous syndrome, Neurofibromatosis Type 1 , and LEOPARD syndrome.
- Hereditary gingival fibromatosis type 1 is also a RASopathy contemplated herein.
- RASopathy patients include Noonan syndrome, Costello syndrome, Cardiofaciocutaneous syndrome, Neurofibromatosis Type 1 , LEOPARD syndrome, hereditary gingival fibromatosis type 1
- RASopathy patients include patients with heterozygous variants in one or more of the following genes: BRAF, CBL, HRAS, KRAS, LZTR1 , MAP2K1 , MAP2K2, MRAS, NF1 , NRAS, PPP1 CB, PTPN11 , RAF1 , RRAS, RIT1 , SHOC2, SOS1 , or SOS2 (Tajan et al. Endocr. Rev. 2018;39(5):676-700).
- CFC is caused by mutations in several genes in the Ras/MAPK signaling pathway, including K-Ras, B-Raf, Mek1 and Mek2.
- Costello syndrome also called faciocutaneoskeletal (FCS) syndrome is caused by activating mutations in the H-Ras gene.
- Hereditary gingival fibromatosis type I is caused by dominant mutations in the SOS1 gene (Son of Sevenless homolog 1 ), which encodes a guanine nucleotide exchange factor (SOS) that acts on the Ras subfamily of small GTPases.
- Neurofibromatosis type I is caused by mutations in the neurofibromin 1 gene, which encodes a negative regulator of the Ras/MAPK signaling pathway.
- Noonan syndrome is caused by mutations in one of several genes, including PTPN11 , which encodes SHP2, and SOS1 , as well as K-Ras and Raf-1 .
- CNP has been demonstrated to be an effective therapy in RASopathy models.
- Ono et al. generated mice deficient in Nf1 in type II collagen producing cells (Ono et aL, Hum. Mol. Genet. 2013;22(15):3048-62). These mice demonstrated constitutive ERK1/2 activation, and decreased chondrocyte proliferation, and maturation. Daily injections of CNP in these mice led to decreased ERK phosphorylation and corrected the short stature.
- a mouse model of Cardiofaciocutaneous syndrome using a Braf mutation (p.Q241 R) Inoue et al. Hum. Mol.
- Noonan syndrome which is characterized by short stature, heart defects, bleeding problems, and skeletal malformations. Mutations in the PTPN11 gene cause about half of all cases of Noonan’s syndrome. SOS1 gene mutations cause an additional 10 to 15 percent, and RAF1 and RIT1 genes each account for about 5 percent of cases. Mutations in other genes each account for a small number of cases. The cause of Noonan syndrome in 15 to 20 percent of people with this disorder is unknown.
- the PTPN11 , SOS1 , RAF1 , and RIT 1 genes all encode for proteins that are important in the RAS/MAPK cell signaling pathway, which is needed for cell division and growth (proliferation), differentiation, and cell migration. Many of the mutations in the genes associated with Noonan syndrome cause the resulting protein to be turned on (active) and this prolonged activation alters normal RAS/MAPK signaling, which disrupts the regulation of cell growth and division, leading to the characteristic features of Noonan syndrome. See, e.g., Chen et aL, Proc Natl Acad Sci U S A. 1 11 (31 ):11473-8, 2014, Romano et al., Pediatrics.
- a subject having mutations that activate the MAPK pathway would benefit from treatment with CNP variants as described herein to improve bone growth and short stature. It is also contemplated that a subject having mutations that activate the MAPK pathway would benefit from treatment with CNP variants as described herein to improve other comorbidities associated with an overactive MAPK pathway in other cells throughout the body where the NPR2 receptor is expressed on its surface.
- IHH Indian hedgehog
- IGF1 R Insulin-like growth factor 1 receptor
- 32 Insulin-like growth factor 1 receptor
- IGF1 R Insulin-like growth factor 1 receptor
- SGA Small for gestational age children
- individuals with familial short stature Kawashima et al., Endocrine J. 59:179-185, 2012.
- Mutations in IGF1 R associated with short stature include R108Q/K115N, R59T, R709Q, G1050K, R481 Q, V599E, and G1 125A (Kawashima, supra).
- Osteogenesis imperfecta also known as “osteogenesis congenita,” “osteogenesis imperfecta congenita” or “brittle bone disease”, is a heterogenous type 1 collagenopathy characterized by fragile bones. Osteogenesis imperfecta is found in approximately 1 :10000 to 1 :20000 individuals worldwide. Ol patients experience approximately 154 fractures per 1000 patient-years (Folkestad et al. J Bone Mineral Res. 31 : 2159-2166, 2016) and delayed healing was reported in -24% of fractures (Rauch, et al. J Bone Mineral Res.
- Type I subjects exhibit bones likely to break from mild to moderate trauma, with most broken bones occurring before puberty; no change or only slight changes to stature with aging; loose joints and muscle weakness; blue, purple, or gray tint to sclera (whites of the eyes); triangular face; curved spine with potential for compression of the vertebrae (spine bones) with aging; mild or no bone deformity; possible changes to the strength and color of teeth; possible hearing loss; normal collagen structure, but less than the normal amount is produced.
- Type II Ol is characterized by death at birth or shortly after, because of the inability to breathe; numerous broken bones that develop before birth while the baby is still in the womb; severe bone deformities; very small stature; underdeveloped lungs; blue, purple, or gray tint to sclera; improperly formed collagen.
- Skeletal symptoms include bone fractures, deformity of vertebrae that may result in the impairment of pulmonary function and short stature. Additional common (>50% patients) extraskeletal symptoms include fragility of vessels, hearing loss and discoloration in sclera (Steiner et aL, GeneReviews,” COL1 A1/2 Osteogenesis Imperfecta”; Etich et al. Mol Cell Pediatr. 2020).
- Type III Intermediate severity Ol is classified as Type III, Type VI or Type V, having similar characteristics to Type I disease, including, but not limited to different severity of short stature, bone density, bone deformity, ease of broken bones and bone fragility, and loose joints.
- Type III disease presents with moderate to severe bone fragility and more frequent fractures, extremely short stature, and more severe long bone deformity vs.
- Type I disease Type VI- Type XXI 01 are often classified based on the mutation shown to cause the disease. Z-scores are typically lowers in females, and are decreased with age (Robinson, European Soc for Paediatric Res 2023). Short stature is most severe in Type III and IV 01, and individuals with Type 1 can have a SD below -2.5.
- Bisphosphonates have been administered in order to decrease fracture incidence and are utilized to manage moderate to severe forms of Ol in children.
- Bisphosphonates include pamidronate, alendronate and risedronate.
- Bisphosphonates increase bone mineral density (BMD) and reduce biochemical markers of bone turnover, effects on preventing fractures are inconclusive (Raid et al., J Bone Mineral Res, 30:929-933, 2014; Liu et al., J Clin Endo Metab, 108:1787-179, 2023). Further, they do not improve bone quality and bone pain beyond 1 -year post-treatment (Liu et al., supra, Botor et aL, Biomolecules, 11 (10):1493, 2021).
- C-type natriuretic peptide (CNP) is produced in the growth plate and positively regulates linear bone growth. It is contemplated that administration of CNP variants or analogs will improve bone growth and reduce incidence and or severity of bone fracture in Ol patients.
- the subject is further administered adjunct therapy to treat osteogenesis imperfecta.
- the adjunct therapy is alendronate, risedronate, ibandronate, zoledronic acid, denosumab, romosozumab, estrogen therapy or hormone therapy, raloxifene, apeledoxifene and estrogen, teriparatide, or abaloparatide.
- the adjunct therapy is bisphosphonate therapy.
- Trabecular bone tissue is spongy and porous, comprising both hard and soft tissue components, and is found at the epiphyses and metaphyses of long bones and in the vertebral bodies.
- Trabecular bone in Ol is characterized by a lower trabecular number and impaired connectivity as well as decreased trabecular thickness and volumetric bone mass (Nijhuis et aL, J Child Orthop. 2019 13(1 ): 1-11 )
- CNP therapy alleviates one or more symptoms of osteogenesis imperfecta, including but not limited to, femoral length, vertebral height, reduction in bone fracture frequency, cortical bone tissue mineral density (TMD), cortical bone mineral density (BMD), cortical bone thickness, cortical porosity, trabecular bone volume fraction (BVF), trabecular TMD, trabecular BMD, trabecular bone number and trabecular bone separation.
- CNP therapy improves annualized growth velocity (AGV) and final adult height in a subject receiving therapy, e.g., a subject with osteogenesis imperfecta.
- CNP also improves bone strength of subjects with achondroplasia that receive long term CNP therapy.
- a method of improving and/or maintaining bone strength in a subject in need thereof comprising administering a C-type natriuretic peptide (CNP) to the subject.
- CNP C-type natriuretic peptide
- the CNP is administered daily, between 2 to 5 times per week or weekly.
- the subject has a bone-related disorder, such as skeletal dysplasia or short stature.
- Non-limiting examples of CNP-responsive bone-related disorders skeletal dysplasias and short stature disorders include achondroplasia, hypochondroplasia, short stature, idiopathic short stature, dwarfism, osteochondrodysplasias, thanatophoric dysplasia, osteogenesis imperfecta, osteogenesis congenita, achondrogenesis, homozygous achondroplasia, chondrodysplasia congenita, campomelic dysplasia, congenital lethal hypophosphatasia, perinatal lethal type of osteogenesis congenita, short-rib polydactyly syndromes, rhizomelic type of chondrodysplasia congenita, Jansen-type metaphyseal dysplasia, spondyloepiphyseal dysplasia congenital, atelosteogenesis, diastrophic dysplasia, congenital short femur, Langer-type mesomelic
- Height is a highly heritable trait that can be influenced by the combined effect of hundreds or thousands of genes (Wood et al, 2014, Nature Genetics, 46:1173-1189). Short stature in an individual can be the result of the combined effect of these genes, without a single gene being the primary contributor. It is contemplated that such individuals with short stature defined by a height SDS of less than -1 .0, -1 .5, -2.0, -2.5, or -3.0, can be beneficially treated with a CNP variant given the ability of CNP to increase the length of normal animals, for example, enhance bone growth and length of bones.
- the CNP variants are useful to treat a subject having a height SDS of less than -1 .0, -1 .5, -2.0, -2.5, or -3.0, and having at least one parent with a height SDS of less than -1 .0, -1 .5, -2.0 or -2.5, optionally wherein the second parent has height within the normal range.
- the CNP variants are useful to treat a subject having a height SDS of between -2.0 to -3.0.
- the CNP variants are useful to treat a subject having a height SDS of between -2.0 to -2.5.
- the short stature is associated with one or more mutations in a gene associated with short stature, such as, collagen (COL2A1 , COL11 A1 , COL9A2, COL10), aggrecan (ACAN), Indian hedgehog (IHH), PTPN1 1 , SHOX, NPR2, NPPC, FGFR3, or insulin growth factor 1 receptor (IGF1 R), DTL, PAPPA2, or combinations thereof, or associated with one or more mutations in a gene associated with a RASopathy.
- collagen COL2A1 , COL11 A1 , COL9A2, COL10
- aggrecan ACAN
- Indian hedgehog IHH
- PTPN1 1 PTPN1 1
- SHOX Indian hedgehog
- NPR2 NPPC
- FGFR3 FGFR3
- IGF1 R insulin growth factor 1 receptor
- the short stature is in a subject with osteogenesis imperfecta.
- the Ol may result from a mutation in one or more of the following genes: COL1 A1 , COL1 A2, COL2A1 , COL1 1A1 , COL9A2, COL10, IFITM5, SERPINF1 , CRTAP, LEPRE1 , PPIB, SERPINH1 , FKBP10, SP7, BMP1 , TMEM38B, WNT1 , CREB3L1 , SPARC, FAM46A or combinations thereof.
- the short stature is a result of mutations in multiple genes as determined by polygenic risk score (PRS).
- Polygenic risk scores are calculated for height using the largest published genome-wide association study (GWAS) meta-analysis for height that do not include any samples from the UK Biobank project as described in WO 2021/055497.
- the cohort may be divided into five PRS quintiles (PRS 1 being the lowest height, PRS 5 the tallest height).
- PRS 1 being the lowest height, PRS 5 the tallest height.
- the subject has a mutation in NPR2 and a low PRS.
- the subject has a mutation in FGFR3 and a low PRS.
- the subject has a mutation in NPR2 and a low PRS.
- the subject has a mutation in IGF1 R and a low PRS. In various embodiments, the subject has a mutation in NPPC and a low PRS. In various embodiments, the subject has a mutation in SHOX and a low PRS. In various embodiments, the subject has one or more mutation in one or more of FGFR3, IGF1 R, NPPC, NPR2 and SHOX, and a low PRS. In various embodiments, the PRS is 1 or 2. In various embodiments, the PRS is 1 . In various embodiments, the PRS is 2.
- the CNP variants are useful for treating other bone-related conditions and disorders, such as rickets, hypophosphatemic rickets [including X-linked hypophosphatemic rickets (also called vitamin D-resistant rickets) and autosomal dominant hypophosphatemic rickets], and osteomalacia [including tumor-induced osteomalacia (also called oncogenic osteomalacia or oncogenic hypophosphatemic osteomalacia)].
- rickets including X-linked hypophosphatemic rickets (also called vitamin D-resistant rickets) and autosomal dominant hypophosphatemic rickets)
- osteomalacia including tumor-induced osteomalacia (also called oncogenic osteomalacia or oncogenic hypophosphatemic osteomalacia)].
- LoF Loss of Function
- GoF Gain of Function
- the subject has a PRS in the bottom 20%, 19%, 18%, 17.5%, 17%, 16.5%, 16%, 15.5%, 15%, 14.5%, 14%, 13.5%, 13%, 12.5%, 12%, 11%, 10%, 9%, 8%, 7.5%, 7%, 6%, 5%, 4%, 3%, 2.5%, 2% or 1 %.
- step iii) and iv) is a subject with a CNP variant if the subject has a LoF variant and a PRS in the bottom 12.5%.
- a LoF or GoF variant in a gene related to short stature is determined by a biological activity assay.
- a LoF or GoF variant may be predicted based on mapping to the predicted 3D structure and activity domain of a protein encoded by the gene, e.g., using AlphaForm 3D mapping or other protein mapping tools.
- the PRS is calculated by a genome-wide association study (GWAS) of height.
- GWAS genome-wide association study
- the CNP variants and compositions and formulations comprising the same of the present disclosure are useful for improving one or more of the symptom(s) or physiological consequences of a skeletal dysplasia, wherein the improvement may be increased absolute growth, increased growth velocity, increased qualitative computed tomography (QCT) bone mineral density, improvement in growth plate morphology, increased long bone growth, improvement in spinal morphology, improved elbow joint range of motion and/or decreased sleep apnea.
- QCT quantitative computed tomography
- the terms “improved”, “improvement”, “increase”, “decrease” and grammatical equivalents thereof are all relative terms that when used in relation to a symptom or physiological consequence of a disease state, refer to the state of the symptom or physiological consequence of the disease after treatment with a CNP variant (or composition or formulation comprising the same) of the present invention as compared to the same symptom or physiological consequence of the disease before treatment with a CNP variant (or composition or formulation comprising the same) of the present invention (i.e., as compared to "baseline”).
- a “baseline” state can be determined either through measurement of the state in the subject prior to treatment (which can subsequently be compared to the state in the same subject after treatment), or through measurement of that state in a population of subjects suffering from the same affliction that share the same or similar characteristics (e.g., age, sex and/or disease state or progression).
- FGFR-3 constitutively active mutant fibroblast growth factor receptor 3
- the disclosure provides CNP variants that in vitro or in vivo stimulate the production of at least about 50%, 60%, 70%, 80%, 90%, 100%, 110%, 120%, 130%, 140% or 150% of the cGMP level produced under the same concentration of wtCNP22 (e.g., 1 uM).
- the CNP variants of the disclosure in vitro or in vivo stimulate the production of at least about 50%, 60%, 70%, 80%, 90%, 100%, 1 10%, 120%, 130%, 140% or 150% of the cGMP level produced under the same concentration of wtCNP22 (e.g., 1 uM).
- an enhancement or increase in growth velocity is an increase in annualized growth velocity of at least about 25%, more preferably at least about 40%, change from baseline in the subject.
- an enhancement or increase in growth is an increase in bone growth of at least about 25%, more preferably at least about 40%, change from baseline in the subject.
- the CNP variant is PGQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (Pro-Gly-CNP-37) (SEQ ID NO: 1 ).
- the peptide further comprises an acetyl group.
- the acetyl group is on the N-terminus of the peptide.
- the peptide further comprises an OH or an NH2 group at the C-terminus.
- the variant comprises one or more linker groups as described herein.
- the linker is a hydrolysable linker, e.g., as described herein.
- the CNP variant is selected from the group consisting of PGQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (SEQ ID NO: 47);
- GQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC GQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (Gly-CNP-37: SEQ ID NO: 2)
- GQEHPNARKYKGANPKGLSKGCFGLKLDRIGSMSGLGC SEQ ID NO: 50;
- the CNP variant comprises a conjugate moiety.
- the conjugate moiety is a synthetic polymeric group.
- the variant comprises a synthetic polymeric group coupled to the variant through a hydrolysable linker.
- the synthetic polymeric group comprises a hydrophilic polymer moiety.
- the hydrophilic polymer moiety comprises polyethylene glycol (PEG).
- the hydrophilic polymer moiety comprises polyethylene glycol (PEG) having a 6 to 20 atom chain length.
- the conjugate moiety comprises one or more acid moieties linked to a hydrophilic spacer as described herein.
- the conjugate moiety comprises one or more acid moieties linked to a hydrophilic spacer.
- the hydrophilic spacer is any amino acid.
- the hydrophilic spacer is gamma glutamic acid (yGlu).
- the hydrophilic spacer is OEG (8-amino-3,6-dioxaoctanoic acid).
- the hydrophilic spacer is gamma glutamic acid (yGlu) or OEG (8-amino-3,6- dioxaoctanoic acid).
- the hydrophilic spacer is gamma glutamic acid (yGlu) linked to one or two or more OEG (8-amino-3,6-dioxaoctanoic acid).
- the acid moiety is a fatty acid.
- Exemplary fatty acids include short chain, medium chain, or long chain fatty acids, or a dicarboxylic fatty acid.
- the fatty acid is saturated or unsaturated. Contemplated are C-6 to C-20 fatty acids, including but not limited to, C-6, C-8, C-10, C-12, C-14, C-16, C-18 or C-20 fatty acids, saturated or unsaturated.
- the fatty acid is decanoic acid, dodecanoic acid, myristic acid, palmitic acid, stearic acid, arachidic acid, or diacids of the same.
- the variant comprises one or more linker groups.
- the linker is on a residue of the CNP cyclic domain or at a site other than the CNP cyclic domain. In various embodiments, the linker is on a lysine residue.
- Efficacy of treatment is measured by various parameters. In various embodiments, efficacy is assessed as the change in annualized growth velocity from the baseline period to the intervention period. Efficacy will also be assessed as the change in height SDS from baseline to end of treatment as measured using the GDC growth curves, and growth velocity SDS will be based on the Bone Mineral Density in Childhood Study (Kelly et al., J. Clin. Endocrinol. Metab. 2014;99(6) :2104-2112). Annualized growth velocity, body proportionality and bone growth/length can be measured as described in Savarirayan et al., (New England J Med 2019 181 : 25-35), herein incorporated by reference.
- Efficacy can also be measured using analysis of skull and brain morphology, e.g., using magnetic resonance imaging (MRI).
- MRI magnetic resonance imaging
- Foramen magnum stenosis has been implicated as the major underlying cause of an increased risk for sudden deaths observed in children less than age 5 years with achondroplasia (Pauli et al., J Pediatr 1984;104:342-8; Hashmi et al., Am J Med Genet A 2018;176:2359-64).
- Skull and brain morphology analysis include measurement of improvements in facial volume, sinus volume, and foramen magnum area in patients, e.g., younger patients less than 6 months old, treated with CNP variants.
- a method for increasing facial volume, facial sinus volume, and foramen magnum area in a subject 6 months old or less having a bone-related disorder, skeletal dysplasia or short stature comprising administering CNP variants, conjugates, salts or prodrugs thereof at a dose of at least 30 pg/kg.
- a method of decreasing the incidence of sudden infant death, sleep disordered breathing, and necessity for neurosurgical decompression of the foramen magnum in a subject 6 months old or less having a bone-related disorder, skeletal dysplasia or short stature comprising administering CNP variants, conjugates, salts or prodrugs thereof at a dose of at least 30 pg/kg.
- the CNP variant is administered at a dose of 30 pg/kg for 3 months, 6 months, 1 year or more.
- the dose of CNP variant is decreased to 15 pg/kg when the subject is about 2 years old.
- MRI magnetic resonance imaging
- QoLISSY the Quality of Life in Short Stature Youth, is assessed as directed (Quality of Life in Short Stature Youth - The QoLISSY Questionnaire User’s Manual. Lengerich: Pabst Science Publishers; 2013).
- Biomarker refers to a detectable biological substance or moiety whose level is increased or decreased in association with a particular disease condition or treatment regimen.
- biomarkers may be measured before, during and/or after administration of a CNP variant as described herein.
- Exemplary bone- or cartilage-associated biomarkers include, but are not limited to, NTproCNP, N terminal fragment of collagen X (CXM), CNP, cGMP, propeptides of collagen type II and fragments thereof, collagen type II and fragments thereof, propeptides of collagen type I and fragments thereof, collagen type I and fragments thereof, osteocalcin, proliferating cell nuclear antigen (PCNA), aggrecan chondroitin sulfate, collagen X, and alkaline phosphatase.
- Markers of bone resorption include cross-linked C-telopeptides of type II collagen (CTXII), urinary N-telopeptide (NTX) and tartrate resistant acid phosphatase (TRAP).
- Markers of bone formation include bone-specific alkaline phosphatase (BSAP), osteocalcin (OC), N-terminal pro-peptide of type I procollagen (PINP) and procollagen type 1 C-terminal propeptide (P1 CP).
- Cartilage- and bone-associated biomarkers can be measured in any appropriate biological sample, including but not limited to tissues, blood, serum, plasma, cerebrospinal fluid, synovial fluid and urine.
- NTproCNP is an amino-terminal propeptide (NTproCNP) of CNP that is released from cells at an equimolar ratio with CNP.
- the biologically active forms of CNP are found in plasma in low concentrations due to the quick clearance rate of the peptide.
- NTproCNP is not cleared via the same mechanism and it is found in the circulation at 20- to 50-fold higher concentration (Olney et al., Clin Endocrinol (Oxf). 2012, 77:416-422).
- NTproCNP levels are measured in a sample and doses of CNP altered or changed to bring NTproCNP levels within +/- 2 SDS of the mean NTproCNP for the population.
- NTproCNP mean levels for different populations have been studied in the following publications, herein incorporated by reference: Olneyet al. (2015).
- C-type natriuretic peptide plasma levels are elevated in subjects with achondroplasia, hypochondroplasia, and thanatophoric dysplasia.
- Olney 2016 shows that children between 6 to 10 years old with idiopathic short stature can have an average baseline NTproSDS of -0.6, ranging from -1.0 to 0.7.
- Olney 2012 reported NTproCNP levels in healthy children/adolescents during stages of growth.
- NTproCNP SDS can be calculated based on the average NTproCNP levels of the different age populations, and therefore +/- 2 SDS from this mean can also be calculated.
- NTproCNP levels of subjects with achondroplasia or hypochondroplasia are described in Olney 2015, showing that children approximately 3 to 8 years old have an NTproSDS average of 1 .4, ranging from 0.4 to 1 .8, while hypochondroplasia subjects (age 6.6 to 11 ) have an average NTproCNP SDS of 1 .9, ranging from 1 .8 to 2.3. Methods for determining NTproSDS levels are described herein and in the publications above.
- Collagen type X biomarker CXM is a degradation fragment of collagen type X, comprising intact trimeric noncollagenous 1 (NC1) domain of type X collagen. CXM is released by active growth plates and decreases in samples as subjects age. CXM levels have been correlated with growth velocity in children (Coghlan et al., Sci Transl Med 2017, 9(419):eaan4669).
- Bone-specific alkaline phosphatase is a bone growth biomarker produced by osteoblasts and osteoclasts in growth plates and mineralized bone. Changes in BSAP may reflect growth plate activity, bone growth, and I or bone remodeling activity.
- N-terminal pro-peptide of type I procollagen is a potential pharmacodynamic bone growth biomarker, released during production of type I collagen. Changes in PINP may reflect changes in growth plate activity, bone growth, and/or bone remodeling.
- CTXII Cross-linked C-telopeptides of type II collagen
- Changes in CTXII may reflect changes in growth plate activity, bone growth, bone remodeling, and/or articular cartilage remodeling.
- compositions including modified release compositions, comprising a CNP variant described herein, and one or more pharmaceutically acceptable excipients, carriers and/or diluents.
- the compositions further comprise one or more other biologically active agents (e.g., inhibitors of proteases, receptor tyrosine kinases, and/or the clearance receptor NPR-C).
- modified release compositions comprising a conjugate moiety as described herein.
- Modified-release compositions include those that deliver a drug with a delay after its administration (delayed-release dosage) or for a prolonged period of time (extended-release dosage).
- modified-release compositions such as extended release, sustained or controlled release, and delayed release.
- extended release composition refers to a composition formulated in a manner in order to make the active ingredient/drug available over an extended period of time following administration (US Pharmacopeia).
- Extended-release dosage include sustained-release (SR) or controlled-release (CR) forms in which.
- Sustained release maintains drug release over a sustained period but not necessarily at a constant rate, while CR maintains drug release over a sustained period at a nearly constant rate (Pharmaceutics: Drug Delivery and Targeting, Yvonne Perrie, Thomas Rades, Pharmaceutical Press, 2009). Delayed-release compositions or products are modified to delay release of the drug substance for some period of time after initial administration.
- the modified release composition is an extended release composition.
- the modified release composition is a sustained release composition.
- the sustained or extended release compositions comprises a CNP pro-drug.
- the composition comprises an excipient, diluent or carrier.
- the extended release composition comprises an excipient, diluent or carrier.
- the excipient, diluent or carrier is a pharmaceutically acceptable excipient, diluent or carrier.
- Non-limiting examples of excipients, carriers and diluents include vehicles, liquids, buffers, isotonicity agents, additives, stabilizers, preservatives, solubilizers, surfactants, emulsifiers, wetting agents, adjuvants, and so on.
- compositions can contain liquids (e.g., water, ethanol); diluents of various buffer content (e.g., Tris-HCI, phosphate, acetate buffers, citrate buffers), pH and ionic strength; detergents and solubilizing agents (e.g., Polysorbate 20, Polysorbate 80); anti-oxidants (e.g., methionine, ascorbic acid, sodium metabisulfite); preservatives (e.g., Thimerosol, benzyl alcohol, m-cresol); and bulking substances (e.g., lactose, mannitol, sucrose).
- buffer content e.g., Tris-HCI, phosphate, acetate buffers, citrate buffers
- detergents and solubilizing agents e.g., Polysorbate 20, Polysorbate 80
- anti-oxidants e.g., methionine, ascorbic acid, sodium metabisulfite
- preservatives e.g
- excipients, diluents and carriers in the formulation of pharmaceutical compositions is known in the art; see, e.g., Remington's Pharmaceutical Sciences, 18 th Edition, pages 1435-1712, Mack Publishing Co. (Easton, Pennsylvania (1990)), which is incorporated herein by reference in its entirety.
- carriers include without limitation diluents, vehicles and adjuvants, as well as implant carriers, and inert, non-toxic solid or liquid fillers and encapsulating materials that do not react with the active ingredient(s).
- Non-limiting examples of carriers include phosphate buffered saline, physiological saline, water, and emulsions (e.g., oil/water emulsions).
- a carrier can be a solvent or dispersing medium containing, e.g., ethanol, a polyol (e.g., glycerol, propylene glycol, liquid polyethylene glycol, and the like), a vegetable oil, and mixtures thereof.
- the compositions are liquid formulations.
- the formulations comprise a CNP variant in a concentration range from about 0.1 mg/ml to about 20 mg/ml, or from about 0.5 mg/ml to about 20 mg/ml, or from about 1 mg/ml to about 20 mg/ml, or from about 0.1 mg/ml to about 10 mg/ml, or from about 0.5 mg/ml to about 10 mg/ml, or from about 0.5 to 5 mg/ml, or from about 0.5 to 3 mg/ml, or from about 1 mg/ml to about 10 mg/ml.
- the CNP variant is in a concentration of 0.8 mg/ml to 2 mg/ml.
- the CNP variant is at a concentration of 0.8 mg/ml. In various embodiments, the CNP variant is at a concentration of 2.0 mg/ml. In various embodiments, the CNP variant is reconstituted from a lyophilized powder.
- the compositions comprise a buffer solution or buffering agent to maintain the pH of a CNP-containing solution or suspension within a desired range.
- buffer solutions include phosphate buffered saline, Tris buffered saline, and Hank's buffered saline.
- Buffering agents include without limitation sodium acetate, sodium phosphate, and sodium citrate. Mixtures of buffering agents can also be used.
- the buffering agent is acetic acid/acetate or citric acid/citrate. The amount of buffering agent suitable in a composition depends in part on the particular buffer used and the desired pH of the solution or suspension.
- the buffering agent has a concentration of about 10 mM ⁇ 5 mM.
- the pH of a composition is from about pH 3 to about pH 9, or from about pH 3 to about pH 7.5, or from about pH 3.5 to about pH 7, or from about pH 3.5 to about pH 6.5, or from about pH 4 to about pH 6, or from about pH 4 to about pH 5, or is at about pH 5.0 ⁇ 1 .0.
- the pH is about 5.0, 5.1 , 5.2, 5.3, 5.4, 5.5, 5.6, 5.7, 5.8, 5.9 or 6.0.
- the pH is 5.5.
- the compositions contain an isotonicity-adjusting agent to render the solution or suspension isotonic and more compatible for administration.
- isotonicity agents include NaCI, dextrose, glucose, glycerin, sorbitol, xylitol, and ethanol.
- the isotonicity agent is NaCI.
- NaCI is in a concentration of about 160 ⁇ 20 mM, or about 140 mM ⁇ 20 mM, or about 120 ⁇ 20 mM, or about 100 mM ⁇ 20 mM, or about 80 mM ⁇ 20 mM, or about 60 mM ⁇ 20 mM.
- the compositions comprise a preservative.
- Preservatives include, but are not limited to, m-cresol and benzyl alcohol.
- the preservative is in a concentration of about 0.4% ⁇ 0.2%, or about 1% ⁇ 0.5%, or about 1 .5% ⁇ 0.5%, or about 2.0% ⁇ 0.5%.
- the compositions contain an anti-adsorbent (e.g., to mitigate adsorption of a CNP variant to glass or plastic).
- Anti-adsorbents include without limitation benzyl alcohol, Polysorbate 20, and Polysorbate 80.
- the antiadsorbent is in a concentration from about 0.001% to about 0.5%, or from about 0.01% to about 0.5%, or from about 0.1% to about 1%, or from about 0.5% to about 1 %, or from about 0.5% to about 1 .5%, or from about 0.5% to about 2%, or from about 1% to about 2%.
- the compositions comprise a stabilizer.
- stabilizers include glycerin, glycerol, thioglycerol, methionine, and ascorbic acid and salts thereof.
- the stabilizer when the stabilizer is thioglycerol or ascorbic acid or a salt thereof, the stabilizer is in a concentration from about 0.1% to about 1%.
- the stabilizer when the stabilizer is methionine, the stabilizer is in a concentration from about 0.01% to about 0.5%, or from about 0.01 % to about 0.2%.
- the stabilizer when the stabilizer is glycerin, the stabilizer is in a concentration from about 5% to about 100% (neat).
- the compositions contain an antioxidant.
- antioxidants include without limitation methionine and ascorbic acid.
- the molar ratio of antioxidant to CNP is from about 0.1 :1 to about 15:1 , or from about 1 :1 to about 15:1 , or from about 0.5:1 to about 10:1 , or from about 1 :1 to about 10:1 or from about 3:1 to about 10:1 .
- compositions can be used in the compositions, including without limitation mineral acid salts (e.g., hydrochloride, hydrobromide, phosphate, sulfate), salts of organic acids (e.g., acetate, propionate, malonate, benzoate, mesylate, tosylate), and salts of amines (e.g., isopropylamine, trimethylamine, dicyclohexylamine, diethanolamine).
- mineral acid salts e.g., hydrochloride, hydrobromide, phosphate, sulfate
- organic acids e.g., acetate, propionate, malonate, benzoate, mesylate, tosylate
- salts of amines e.g., isopropylamine, trimethylamine, dicyclohexylamine, diethanolamine.
- compositions can be administered in various forms, such as tablets, capsules, granules, powders, solutions, suspensions, emulsions, ointments, and transdermal patches.
- the dosage forms of the compositions can be tailored to the desired mode of administration of the compositions.
- the compositions can take the form of, e.g., a tablet or capsule (including softgel capsule), or can be, e.g., an aqueous or nonaqueous solution, suspension or syrup.
- Tablets and capsules for oral administration can include one or more commonly used excipients, diluents and carriers, such as mannitol, lactose, glucose, sucrose, starch, corn starch, sodium saccharin, talc, cellulose, magnesium carbonate, and lubricating agents (e.g., magnesium stearate, sodium stearyl fumarate).
- lubricating agents e.g., magnesium stearate, sodium stearyl fumarate
- flavoring, coloring and/or sweetening agents can be added to the solid and liquid formulations.
- Other optional ingredients for oral formulations include without limitation preservatives, suspending agents, and thickening agents.
- Oral formulations can also have an enteric coating to protect the CNP variant from the acidic environment of the stomach. Methods of preparing solid and liquid dosage forms are known, or will be apparent, to those skilled in this art (see, e.g., Remington's Pharmaceutical Sciences, referenced above).
- Formulations for parenteral administration can be prepared, e.g., as liquid solutions or suspensions, as solid forms suitable for solubilization or suspension in a liquid medium prior to injection, or as emulsions.
- sterile injectable solutions and suspensions can be formulated according to techniques known in the art using suitable diluents, carriers, solvents (e.g., buffered aqueous solution, Ringer's solution, isotonic sodium chloride solution), dispersing agents, wetting agents, emulsifying agents, suspending agents, and the like.
- solvents e.g., buffered aqueous solution, Ringer's solution, isotonic sodium chloride solution
- dispersing agents e.g., buffered aqueous solution, Ringer's solution, isotonic sodium chloride solution
- dispersing agents e.g., buffered aqueous solution, Ringer's solution, isotonic sodium chloride solution
- dispersing agents e.g
- formulations for parenteral administration include aqueous sterile injectable solutions, which can contain antioxidants, buffers, bacteriostats, and solutes that render the formulation isotonic with the blood of the intended recipient; and aqueous and nonaqueous sterile suspensions, which can contain suspending agents and thickening agents.
- Exemplary CNP formulations are described in U.S. Patents 9,907,834 and 10,646,550. Use of CNP formulations having a pH in the range from about 4 to about 6 is contemplated.
- compositions comprising a CNP variant can also be lyophilized formulations.
- the lyophilized formulations comprise a buffer and bulking agent, and optionally an antioxidant.
- Exemplary buffers include without limitation acetate buffers and citrate buffers.
- Exemplary bulking agents include without limitation mannitol, sucrose, dextran, lactose, trehalose, and povidone (PVP K24).
- mannitol is in an amount from about 3% to about 10%, or from about 4% to about 8%, or from about 4% to about 6%.
- sucrose is in an amount from about 6% to about 20%, or from about 6% to about 15%, or from about 8% to about 12%.
- Exemplary anti-oxidants include, but are not limited to, methionine and ascorbic acid.
- the formulation comprises citric acid, sodium citrate, trehalose, mannitol, methionine, polysorbate 80, and optionally sterile water for injection (WFI).
- WFI sterile water for injection
- kits containing, e.g., bottles, vials, ampoules, tubes, cartridges and/or syringes that comprise a liquid (e.g., sterile injectable) formulation or a solid (e.g., lyophilized) formulation.
- the kits can also contain pharmaceutically acceptable vehicles or carriers (e.g., solvents, solutions and/or buffers) for reconstituting a solid (e.g., lyophilized) formulation into a solution or suspension for administration (e.g., by injection), including without limitation reconstituting a lyophilized formulation in a syringe for injection or for diluting concentrate to a lower concentration.
- extemporaneous injection solutions and suspensions can be prepared from, e.g., sterile powder, granules, or tablets comprising a CNP- containing composition.
- the kits can also include dispensing devices, such as aerosol or injection dispensing devices, pen injectors, autoinjectors, needleless injectors, syringes, and/or needles.
- a kit can include syringes having a single chamber or dual chambers.
- the single chamber can contain a liquid CNP formulation ready for injection, or a solid (e.g., lyophilized) CNP formulation or a liquid formulation of a CNP variant in a relatively small amount of a suitable solvent system (e.g., glycerin) that can be reconstituted into a solution or suspension for injection.
- a suitable solvent system e.g., glycerin
- one chamber can contain a pharmaceutically acceptable vehicle or carrier (e.g., solvent system, solution or buffer), and the other chamber can contain a solid (e.g., lyophilized) CNP formulation or a liquid formulation of a CNP variant in a relatively small amount of a suitable solvent system (e.g., glycerin) which can be reconstituted into a solution or suspension, using the vehicle or carrier from the first chamber, for injection.
- a pharmaceutically acceptable vehicle or carrier e.g., solvent system, solution or buffer
- a suitable solvent system e.g., glycerin
- a kit can include one or more pen injector or autoinjector devices, and dual-chamber cartridges.
- One chamber of a cartridge can contain a pharmaceutically acceptable vehicle or carrier (e.g., solvent system, solution or buffer), and the other chamber can contain a solid (e.g., lyophilized) CNP formulation or a liquid formulation of a CNP variant in a relatively small amount of a suitable solvent system (e.g., glycerin) which can be reconstituted into a solution or suspension, using the vehicle or carrier from the first chamber, for injection.
- a suitable solvent system e.g., glycerin
- a cartridge can comprise an amount of the CNP variant that is sufficient for dosing over a desired time period (e.g., 1 day, 2 days, 3 days, 1 week, 2 weeks, 3 weeks, 4 weeks, etc.).
- the pen injector or autoinjector can be adjusted to administer a desired amount of the CNP formulation from a cartridge.
- compositions comprising a CNP variant can be formulated as a slow release, controlled release or sustained release system for maintaining a relatively constant level of dosage over a desired time period, such as 1 week, 2 weeks, 3 weeks, 1 month, 2 months, or 3 months.
- Slow release, controlled release and sustained release formulations can be prepared using, e.g., biodegradable polymeric systems ⁇ which can comprise, e.g., hydrophilic polymers [e. ., polylactide, polyglycolide, poly(lactide-glycolide)] ⁇ , and can take the form of, e.g., microparticles, microspheres or liposomes, as is known in the art.
- Administration and Dosing can be prepared using, e.g., biodegradable polymeric systems ⁇ which can comprise, e.g., hydrophilic polymers [e. ., polylactide, polyglycolide, poly(lactide-glycolide)] ⁇ , and can take the form of,
- the term "therapeutically effective amount" of an active agent refers to an amount that provides therapeutic benefit to a patient.
- the amount may vary from one individual to another and may depend upon a number of factors, including the overall physical condition of the patient.
- a therapeutically effective amount of a CNP variant can be readily ascertained by one skilled in the art, using publicly available materials and procedures.
- the amount of a CNP variant used for therapy should give an acceptable rate of reversal of cartilage degeneration or increase in cartilage growth.
- the dosing frequency for a particular individual may vary depending upon various factors, including the disorder being treated and the condition and response of the individual to the therapy.
- a pharmaceutical composition containing a CNP variant is administered to a subject about one time per day, one time per two days, one time per three days, or one time per week, twice per week, three times per week, once every two weeks, or monthly.
- the CNP variant compositions described herein can be administered to patients in need thereof at therapeutically effective doses to treat, ameliorate or prevent bone-related disorders and short stature disorders (e.g., skeletal dysplasias, including achondroplasia, hypochondroplasia, osteogenesis imperfecta, etc.).
- the CNP variants contemplated for use herein can be administered to patients at therapeutically effective doses to treat, ameliorate or prevent osteoarthritis and other conditions having an osteoarthritis-associated symptom.
- the safety and therapeutic efficacy of the CNP variants can be determined by standard pharmacological procedures in cell cultures or experimental animals, such as, for example, by determining the LD50 (the dose lethal to 50% of the population) and the ED50 (the dose therapeutically effective in 50% of the population).
- the dose ratio between toxic and therapeutic effects is the therapeutic index and it can be expressed as the ratio LD50 /ED50. Active agents exhibiting a large therapeutic index are normally preferred.
- a method of treating osteogenesis imperfecta comprising administering a C-type natriuretic peptide (CNP) to a subject in need thereof, wherein the CNP is administered less frequently than daily administration.
- the CNP is administered 5 times weekly, 4 times weekly, 3 times weekly, 2 times weekly, weekly, bi-weekly, monthly or less frequently
- a method of treating osteogenesis imperfecta comprising administering a C-type natriuretic peptide (CNP) to a subject in need thereof, wherein the CNP is administered 5 times weekly, 4 times weekly, 3 times weekly, weekly or biweekly.
- a method of reducing the frequency of bone fracture in a subject having osteogenesis imperfecta comprising administering a C-type natriuretic peptide (CNP) to a subject in need thereof, wherein the CNP is administered 5 times weekly, 4 times weekly, 3 times weekly, weekly or biweekly.
- CNP C-type natriuretic peptide
- the CNP variant compositions described herein are administered at a dose in the range from about 3, 4, 5, 6, 7, 8, 9 or 10 nmol/kg to about 300 nmol/kg, or from about 20 nmol/kg to about 200 nmol/kg.
- the CNP compositions are administered at a dose of about 3, 4, 5, 6, 7, 8, 9, 10, 15, 20, 25, 30, 35, 40, 45, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 110, 120, 125, 130, 140, 150, 160, 170, 175, 180, 190, 200, 210, 220, 230, 240, 250, 260, 270, 280, 290, 300, 350, 400, 450, 500, 750, 1000, 1250, 1500, 1750 or 2000 nmol/kg or other dose deemed appropriate by the treating physician.
- the CNP variant compositions are administered at a dose of about 0.5, 1 , 2, 3, 4, 5, 6, 7, 8, 9, 10, 15, 20, 25, 30, 40, 50, 60, 70, 80, 90, 100, 110, 120, 130, 140, 150, 160, 170, 180, 190, 200, 250, 300, 350, 400, 450, 500, 550, 600, 650, 700, 750, 800, 850, 900, 950 or 1000 pg/kg, or about 0.5, 0.8, 1 .0, 1 .25, 1 .5, 2, 2.5, 3, 3.5, 4, 4.5, 5, 5.5, 6, 6.5, 7, 7.5, 8, 8.5, 9, 9.5 or 10 mg/kg, or other dose deemed appropriate by the treating physician.
- the doses of CNP or CNP variant described herein can be administered according to the dosing frequency/frequency of administration described herein, including without limitation daily, 2 or 3 times per week, 5 days a week, weekly, every 2 weeks, every 3 weeks, monthly, etc.
- the CNP or CNP variant is administered daily subcutaneously.
- the CNP or CNP variant is administered weekly subcutaneously.
- the CNP variant is administered at a dose of 0.5 pg/kg/day to 60 pg/kg/day, 1 .5 pg/kg/day to 30 pg/kg/day, 2.5 pg/kg/day to 15 pg/kg/day, 10pg/kg/day to 45 pg/kg/day, or 15pg/kg/day to 30 pg/kg/day.
- the CNP variant is administered at a dose of 15 pg/kg/day.
- the CNP variant is administered at a dose of 30 pg/kg/day.
- the CNP variant is administered 20 pg/kg 5 days per week, 20 pg/kg 3 days per week, or 10 pg/kg 3 days per week.
- the frequency of dosing/administration of a CNP variant for a particular subject may vary depending upon various factors, including the disorder being treated and the condition and response of the subject to the therapy.
- the CNP variant can be administered in a single dose or in multiple doses per dosing.
- the CNP variant composition is administered, in a single dose or in multiple doses, once daily, once weekly, once every two weeks, once every three weeks, once every 4 weeks, once every 6 weeks, once every two months, once every three months or once every six months, or as deemed appropriate by the treating physician.
- the CNP variant is administered for 3 month, 6 months, 12 months or more.
- a CNP variant composition is administered to a subject with osteogenesis imperfecta so as to allow for periods of growth (e.g., chondrogenesis), followed by a recovery period (e.g., osteogenesis).
- the CNP composition may be administered subcutaneously or by another mode daily, multiple times per week, or weekly for a period of time, followed by a period of no treatment, then the cycle is repeated.
- the initial period of treatment e.g., administration of the CNP variant composition daily, multiple times per week or weekly
- the initial period of treatment is for 3 days, 1 week, 2 weeks, 3 weeks, 4 weeks, 5 weeks, 6 weeks, 7 weeks, 8 weeks, 9 weeks, 10 weeks, 11 weeks or 12 weeks.
- the period of no treatment lasts for 3 days, 1 week, 2 weeks, 3 weeks or 4 weeks.
- the dosing regimen of the CNP variant compositions is daily for 3 days followed by 3 days off; or daily or multiple times per week for 1 week followed by 3 days or 1 week off; or daily or multiple times per week for 2 weeks followed by 1 or 2 weeks off; or daily, multiple times per week or weekly for 3 weeks followed by 1 , 2 or 3 weeks off; or daily, multiple times per week or weekly for 4, 5, 6, 7, 8, 9, 10, 11 or 12 weeks followed by 1 , 2, 3 or 4 weeks off. It is contemplated that a subject having osteogenesis imperfecta could still be receiving an adjunct therapy in periods when CNP is not being administered, or could be receiving no other treatment in periods when CNP is not being administered.
- the CNP variants, or pharmaceutical compositions comprising them can be administered to subjects in various ways such as, e.g., by injection subcutaneously, intraarticularly, intravenously, intra-arterially, intraperitoneally, intramuscularly, intradermally, or intrathecally, or by oral administration.
- the CNP variants are administered by a single subcutaneous, intraarticular, intravenous, intra-arterial, intraperitoneal, intramuscular, intradermal, or intrathecal injection.
- the CNP variants can be administered by implantation of a depot at the target site of action (e.g., an abnormal or degenerated joint or cartilage area).
- the CNP variants can be administered sublingually under the tongue (e.g., sublingual tablet) or by inhalation into the lungs (e.g., inhaler or aerosol spray), by delivery into the nasal cavity (e.g., intranasal spray), by delivery into the eye (e.g., eye drop), or by transdermal delivery (e.g., by means of a patch on the skin).
- inhaler or aerosol spray e.g., inhaler or aerosol spray
- delivery into the nasal cavity e.g., intranasal spray
- delivery into the eye e.g., eye drop
- transdermal delivery e.g., by means of a patch on the skin.
- the CNP variants may also be administered orally in the form of microspheres, microcapsules, liposomes (uncharged or charged (e.g., cationic)), polymeric microparticles (e.g., polyamides, polylactide, polyglycolide, poly(lactide-glycolide)), microemulsions, and the like.
- microspheres e.g., microspheres, microcapsules, liposomes (uncharged or charged (e.g., cationic)), polymeric microparticles (e.g., polyamides, polylactide, polyglycolide, poly(lactide-glycolide)), microemulsions, and the like.
- a further method of administration is by osmotic pump (e.g., an Alzet pump) or minipump (e.g., an Alzet mini-osmotic pump), which allows for controlled, continuous and/or slow- release delivery of the CNP variant or pharmaceutical composition over a pre-determined period.
- the osmotic pump or mini-pump can be implanted subcutaneously, or near the target site (e.g., the long bones of limbs, the epiphyses, etc.).
- CNP variants or compositions thereof can also be administered by other modes. Determination of the most effective mode of administration of the CNP variants or compositions thereof is within the skill of the skilled artisan.
- the CNP variants can be administered as pharmaceutical formulations suitable for, e.g., oral (including buccal and sub-lingual), rectal, nasal, topical, pulmonary, vaginal or parenteral (including intramuscular, intraarterial, intrathecal, subcutaneous, intraarticularly and intravenous) administration, or in a form suitable for administration by inhalation or insufflation.
- the pharmaceutical formulations can be in the form of solid, semi-solid or liquid dosage forms, such as tablets, suppositories, pills, capsules, powders, liquids, suspensions, emulsions, creams, ointments, lotions, and the like.
- the formulations can be provided in unit dosage form suitable for single administration of a precise dosage.
- the formulations comprise an effective amount of a CNP variant, and one or more pharmaceutically acceptable excipients, carriers and/or diluents, and optionally one or more other biologically active agents.
- the present Example evaluated treatment of young growing oim/oim mice, a model of moderate- to- severe osteogenesis imperfecta (Ol), with a CNP variant and the bisphosphonate alendronate (ALN) as measured by reduced fracture incidence, improved growth, increased bone mineral density (BMD), and improved bone strength.
- Ol moderate- to- severe osteogenesis imperfecta
- APN bisphosphonate alendronate
- mice are treated from 2 -14 weeks of age, for 12 weeks total. Bone quality is evaluated by radiographic analysis of fracture number, histological analyses, micro-CT, FTIR analysis of bone quality and mechanical testing of long bones. Growth is evaluated by weekly weighing and evaluation of long bones radiographically and by micro-CT. Further, muscle mass is assessed by micro CT.
- the oim/oim mouse is an established model of moderate to severe Ol that contains a naturally occurring mutation leading to deficiency of proa2(l) collagen. These mice are characterized by frequent fractures, small size, osteopenia and bone deformities (Chipman et al Proc Nat Acad Sci USA 1993, 90:1701 -1705). Heterozygous oim/+ mice have osteopenia but typically normal longitudinal growth and no spontaneous fractures, and have been used as a model of mild Ol (King, et al., Bone & Mineral Research 2005, 20, 987-993; McBride et al. Calcif Tissue Int 1998, 62, 172-176.).
- mice Homozygous oim/oim mice have been used in a number of studies to evaluate the effect of bisphosphonates (Camacho, et al., Calcified Tissue International 2001 , 69, 94-101 ; Evans et al., Bone 2003, 32, 268-274; McCarthy et al., Ped Res 2002, 52, 660-670.) and RANKL inhibition in Ol (Delos et al., Journal of Orthopaedic Research 2008, 26, 153-164; Bargman et al., Osteoporos Int 2012, 23, 1141 -1150; Bargman et al., Connect Tissue Res 2010, 51 , 123-131 ) 8-10.
- Alendronate (ALN), a bisphosphonate, has been shown to increase BMD, alter geometric and biomechanical properties of oim/oim bone and reduce fractures in these mice whereas RANKL inhibition was also found to increase BMD and alter geometric and biomechanical properties but not to reduce fracture incidence, when treatment started at 6 weeks of age (Bargman 2010, supra).
- Oim/oim mice are randomized for treatment with either saline as a control, CNP, CNP plus alendronate from 2 weeks to 14 weeks of age, additionally a dose range of CNP is analyzed.
- the ALN dose has been established in previous studies described above, and the number per group has been determined based on a power analysis from our previous studies with biomechanical changes as the main outcome.
- the CNP variant used in this study is an analog of Pro-Gly-CNP37 and differs by one less amino acid at its N-terminus.
- CNP-analog is administered at three dosage regimens given subcutaneously (20 ug/kg 5 days per week (20x5), 20 ug/kg 3 days per week (20x3), or 10 ug/kg 3 days per week (10x3)).
- the ALN is injected once per week at a dose of 0.21 mg/kg/week, and saline in an equal volume of 0.1 mL/10 g body weight as previously described (Bargman 2012, supra).
- Mice are also injected with calcein (15mg/kg) at the start and end of their treatments, and with alizarin red (50 mg/kg) at the midpoint, to evaluate bone growth.
- the mother will be placed in a separate cage temporarily while the injections are being given to the pups.
- oim/oim and WT mice will be faxitroned at 8 weeks of age for dose monitoring as well as at 14 weeks of age to determine fracture number at this age.
- mice will be weighed on a digital scale prior to administration of drug. Total body length is assessed tail to nose. To obtain information on femur growth, at sacrifice individual femora are measured in the AP and ML planes from digitized radiographs, and femur length is determined as the distance from the tip of the femoral head to the base of the condyles. Micro-CT is used to show femoral length.
- Radiographic Analyses -Fracture Counts Whole body digital radiographs are taken at sacrifice in the AP and ML planes. The AP radiographs are used for fracture counting. Fractures are counted in a blind fashion such that it will not be known if the particular mouse was treated or a control. Fractures are defined by any evidence of callus formation or obvious bone deformation. The humeri, femora, tibiae, and first 5 vertebrae of the tail of the mice are examined, and the total number of fractures per mouse obtained.
- Faxitron ULTRAFOCUS® Imaging (New York, NY, USA) was used to radiograph mice in the anterior-posterior and medial-lateral plane at 2 weeks (pre-treatment) for baseline images, 8 weeks (mid-treatment) for safety evaluation, and 14 weeks (post-treatment) for final treatment effect analysis. Images were produced at 46kV and 0.3mA. Two-week-old mice were anesthetized with 3% isoflurane and monitored throughout imaging. 14-week-old mice were imaged post-mortem. An aluminum density gradient was used to standardize length and intensity.
- the Faxitron radiographs were used to assess a variety of potential treatment effects, the first of which was fracture number and healing assessment.
- Two blinded investigators CLR and ED
- CLR and ED determined the number of fractures in the humeri, radii, femurs, tibiae, and first five proximal vertebrae of the tail. Fractures were defined as the observation of a fracture line, callus formation, or bone deformation. Radiographs taken at 2 weeks and 14 weeks were directly compared to assess fracture healing throughout the course of treatment.
- Femoral length was measured using Faxitron radiographs of the individual femurs post-sacrifice in the anterior-posterior plane, produced at 30kV and 0.3 mA.
- femoral length was defined as the distance from the base of the condyles to the tip of the femoral head.
- Full body post-sacrifice radiographs taken in the AP plane were used to measure vertebral height, which was defined as the distance from the distal endplate of L6 to the proximal endplate of L4.
- An aluminum density standard was used in the measurements of these individual femurs and full-body AP radiographs.
- Micro CT Bone Architecture Analyses of femurs on high-resolution cone-beam micro- CT scanner (SkyScan 1172; Broker microCT, Kontich, Belgium) is performed. Each scan is referenced to a phantom containing air, saline and a bone reference material (1.18 g/cm 3 ) for calibration of Houndsfield units to mineral density in g/cm 3 . Reconstruction of the individual projections to computed tomography volume data and analyses of the reconstructed scans are performed using instrument software. Specimen-specific thresholds are determined by first selecting a volume of interest, generating the attenuation histogram, and determining the threshold that segments mineralized tissue from background.
- Properties determined include bone mineral content, bone volume fraction, bone surface area, apparent density, tissue density, trabecular thickness, trabecular separation, trabecular number, and polar moment of inertia. Cortical bone was analyzed at the mid-diaphysis and trabecular bone was analyzed at the metaphysis.
- Micro CT Muscle Mass An index of muscle mass is determined after sacrifice by micro CT assessment as recently described (Pasetto et al., PLoS One 2018, 13, e0198089). Briefly, the distance from the upper extremity of the tibia to the medial malleolus (length of the tibia, L) is assessed; the perpendicular distance from the half-length of the tibia to the external margin of the hind limb muscle (thickness of the muscle, T) is assessed. These measurements are acquired by keeping the z axis fixed on the plane where the patella and the upper extremity of tibia are clearly identified. The Index of Muscle Mass (IMM) is defined as the ratio between T and L. The cross-sectional area of the muscle group at half length of the tibia is also measured using a ROI contour tool. For each mouse, the left and right leg are analyzed and averaged.
- ROI contour tool For each mouse, the left and right leg are analyzed and averaged.
- Histologic Analyses The left tibias are collected fresh from each animal, and preserved in routine formalin fixative for 18 hours at 4 °C. The bone is processed for paraffin embedding, sectioned and stained. The toluidine blue satin will show the growth plate. Osteoclastic numbers and activity are evaluated by tartrate resistant acid phosphatase (TRAP) activity after demineralization and paraffin processing. Additional morphometric analyses of the growth plate are done to assess total width and composition. Immunostaining is done for the Natriuretic peptide receptor B(NPRB). Right tibias will undergo PMMA embedding for staining with Goldner’s trichrome which will be used to show osteoid and new bone formation.
- TRIP tartrate resistant acid phosphatase
- FTIR Imaging Data Collection and Analysis FTIR Imaging data is collected from histological sections of long bones to assess molecular composition of the mineralized regions using a Perkin Elmer Spotlight 400 system in the 800-4000 cm spectral region with a spatial resolution of either 6 or 25 microns.
- the system is comprised of an FTIR spectrometer coupled with a light microscope and an 8 X 2 linear array detector, and allows data collection over a region up to ⁇ 10 mm 2 .
- Matlab based data processing software is utilized for imaging data analysis.
- Bone Biomechanics All femurs are evaluated radiographically and only those that lack fractures or obvious deformities are selected for mechanical testing. Three-point bend tests are conducted at room temperature using a Bose Electroforce mechanical testing machine as previously described (Bargman 2010 and 2012, supra). From these curves, the Young’s Modulus is determined by taking the slope of the elastic region of the curve. The yield point will be determined using a regression line over the linear region of the curve and adding data at successively higher stresses until the correlation coefficient (R 2 ) drops below 0.95. The elastic percentage of the curve is determined by taking the yield strain and dividing it by the total strain (last point before the femur breaks). The remaining portion of the curve is defined as the plastic region. The material property “brittleness” of the bone is defined as the elastic region strain divided by the total strain.
- mice in the 20x5 group had no new fractures as shown in Table 1 . In comparison to one fracture in one mouse in both the 20x3 and 10x3 groups.
- All treated groups also had an increase in vertebral height; both the 20x5 and 10x3 groups had higher vertebral heights than the 20x3 group but were not different from each other.
- mice There were no fractures observed in any of the WT mice either pre- treatment or postsacrifice.
- All of the untreated saline oim/oim mice had at least one fracture (1 .8 +/- 2.5 fractures); at sacrifice one mouse had two fractures while the others had none (Table 1 ).
- All of the mice in the high dosage group had at least one fracture at enrollment (4.14 +/- 0.45 fractures); at post-sacrifice there were no new fractures observed in any of the mice.
- All mice in the medium dosage group had at least one fracture at enrollment (3.16 +/- 0.65 fractures); at sacrifice two mice had one fracture each.
- All but one mouse in the low dosage group had at least one fracture at enrollment (2.5 +/- 1 .17 fractures); at sacrifice only two mice had one fracture (Table 4).
- Vertebral Height As seen in the femoral length results, for the WT mice, only the medium dosage group had a statistically significant increase in vertebral height (p ⁇ 0.05) compared to the saline mice. There was no significant difference between the saline, high dosage, and low dosage groups ( Figure 16). For the OIM mice, as seen in the femurs, both the high dosage and low dosage groups had statistically significant increases in vertebral height (p ⁇ 0.01 and p ⁇ 0.05 respectively) (8.6% increase and 10.6% increase respectively) compared to the saline mice. There was no significant difference between the saline and medium dosage groups ( Figure 16).
- Micro-CT In comparison to WT saline mice, all three WT dosage groups had an increase in cortical TMD, cortical minimum inertia, and trabecular TMD ( Figures 17-18, Figure 21 ). In comparison to WT saline mice and WT ALN only mice19, all three WT dosage groups had an additional increase in cortical BMD, cortical thickness, trabecular BVF, and trabecular thickness. None of the three WT dosage groups demonstrated increased trabecular number, trabecular BMD, or decreased trabecular separation that was demonstrated in the ALN only mice19. Only the WT medium dosage group showed an increase in cortical porosity compared to WT saline mice ( Figure 17, Figure 21 ).
- Table 5 Measurements of the second metacarpal (mean ⁇ SD) at each time point in treatment.
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Medicinal Chemistry (AREA)
- General Health & Medical Sciences (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Pharmacology & Pharmacy (AREA)
- Physical Education & Sports Medicine (AREA)
- Veterinary Medicine (AREA)
- Zoology (AREA)
- Public Health (AREA)
- Animal Behavior & Ethology (AREA)
- Molecular Biology (AREA)
- Endocrinology (AREA)
- Cardiology (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Gastroenterology & Hepatology (AREA)
- Immunology (AREA)
- Epidemiology (AREA)
- Toxicology (AREA)
- Genetics & Genomics (AREA)
- Biophysics (AREA)
- Orthopedic Medicine & Surgery (AREA)
- Biochemistry (AREA)
- Rheumatology (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
Abstract
The present disclosure relates, in general, to treatment of osteogenesis imperfecta with C-type natriuretic peptide (CNP) therapy.
Description
TREATMENT OF OSTEOGENESIS IMPERFECTA WITH C-TYPE NATRIURETIC PEPTIDE AND ANALOGS THEREOF
CORRS REFERENCE TO RELATED APPLICATIONS
[0001] The present application claims the priority benefit of U.S. Provisional Patent Application No. 63/542,816, filed October 6, 2023, U.S. Provisional Patent Application No. 63/544,006, filed October 13, 2024, U.S. Provisional Patent Application No. 63/564,067, filed March 12, 2024, and U.S. Provisional Patent Application No. 63/666,302, filed July 1 , 2024, herein incorporated by reference in their entireties.
INCORPORATION BY REFERENCE OF MATERIAL SUBMITTED ELECTRONICALLY
[0002] The Sequence Listing, which is a part of the present disclosure, is submitted concurrently with the specification as a text file. The name of the text file containing the Sequence Listing is “59396_SeqListing.xml", which was created on September 30, 2024, and is 48,423 bytes in size. The subject matter of the Sequence Listing is incorporated herein in its entirety by reference.
FIELD OF THE DISCLOSURE
[0003] The present disclosure relates, in general, to C-type natriuretic (CNP) therapy to treat skeletal dysplasia, such as osteogenesis imperfecta, and measures of efficacy in treatment.
BACKGROUND
[0004] C-type Natriuretic Peptide (CNP) is a paracrine growth factor widely expressed across numerous tissues (Prickett et aL, Peptides 2020; 132:170363) with diverse functions including regulation of endochondral bone growth, blood flow and pressure in the microcirculation, antiinflammatory actions, gamete maturation and neurogenesis and connectivity (Kuhn M., Physiol Rev 2016; 96:751 -804). The best defined of these factors in humans is the crucial role of the hormone in skeletal growth in driving growth plate expansion.
[0005] Studies in experimental animals show that it is the local production of CNP acting via its specific receptor NPR2 within growth plate tissues that determines physiological endochondral bone growth (Nakao et aL, Sci Rep 2015; 5:10554). Study of the dynamic role of CNP in the growth of children is challenging due to the rapid clearance of CNP and very low concentrations in plasma. However, an inactive portion of the synthesized product in tissues (proCNP) - amino terminal proCNP (NTproCNP) - is not subject to clearance or rapid degradation. Its level in plasma reflects variations in linear growth velocity throughout growth in both children and experimental animals (Espiner et aL, Horm Res Paediatr 2018; 90:345-357).
Notably in subjects with genetic disorders of skeletal growth affecting CNP pathway activity plasma NTproCNP concentrations are raised where intra cellular CNP pathway activity is reduced (Olney et aL, J Clin Endocrinol Metab 2015; 100:E355-359; Wang et al., Hum Mutat 2015; 36:474-481 ) and are reduced where intra cellular activity is enhanced (Hannema et al., J Clin Endocrin Metab 2013; 98:E1988-1998; Boudin et aL, Am J Hum Genet 2018; 103:288-295; Miura et al. PloS one 2012; 7:e42180). In achondroplasia, the normal reciprocal antagonism (Ozasa et al., Bone 2005 36:1056-1064) between FGFR3 pathway activity (inhibitory to endochondral bone growth) and CNP signaling (stimulating bone growth) is overridden by a gain of function mutation in FGFR3 (Yasoda etal. Nature Medicine 2004 10:80-86), reducing intracellular CNP activity, and is associated with modest elevations in concentrations of CNP products in plasma (Olney et aL, J Clin Endocrinol Metab 2015 100:E355-359).
[0006] Nothing is known of the dynamics or significance of feedback regulation of CNP during periods of active long bone growth. Further, it is unclear whether such feedback is direct or time-dependent on inter-cellular growth responses of skeletal tissues (indirect feedback). Direct feedback results from actions of the cell’s own product on CNP production whereas indirect feedback involves a longer loop mediated by cells other than those secreting the peptide. However in a recent report addressing these important issues in rodent pups, exogenous CNP administered at high concentrations continuously for 3 days inhibited CNP gene expression but only in tissues containing growth plates (Ueda et aL, PLoS One 2020, 15:e0240023).
SUMMARY
[0007] The present disclosure is directed to treatment of skeletal dysplasias such as osteogenesis imperfecta in patients using CNP or variants thereof.
[0008] Provided herein is a method of treating osteogenesis imperfecta (Ol) or alleviating one or more symptoms thereof comprising administering a C-type natriuretic peptide (CNP) to a subject in need thereof, wherein the CNP is administered less frequently than daily administration. For example, the CNP is administered 5 times weekly, 4 times weekly, 3 times weekly, 2 times weekly, weekly, bi-weekly, monthly or less frequently.
[0009] In various embodiments, provided herein is a method of treating osteogenesis imperfecta (Ol) comprising administering a C-type natriuretic peptide (CNP) to a subject in need thereof, wherein the CNP is administered between 2 to 5 times per week.
[0010] Also contemplated is a method of reducing the frequency of bone fracture in a subject having osteogenesis imperfecta (Ol) comprising administering a C-type natriuretic peptide
(CNP) to a subject in need thereof, wherein the CNP is administered between 2 to 5 times per week.
[0011] In various embodiments, the disclosure provides a method of improving bone mineral density in a subject having osteogenesis imperfecta comprising administering a C-type natriuretic peptide (CNP) to a subject in need thereof, wherein the CNP is administered between 2 to 5 times per week.
[0012] In various embodiments, the disclosure provides a method of improving femoral length or vertebral growth in a subject having osteogenesis imperfecta comprising administering a C- type natriuretic peptide (CNP) to a subject in need thereof, wherein the CNP is administered between 2 to 5 times per week.
[0013] In certain embodiments, provided is a method of treating osteogenesis imperfecta comprising administering a C-type natriuretic peptide (CNP) to a subject in need thereof, wherein the CNP is administered weekly or bi-weekly.
[0014] Provided is a method of reducing the frequency of bone fracture in a subject having osteogenesis imperfecta (Ol). improving bone mineral density in a subject having osteogenesis imperfecta (Ol) and/or improving femoral length or vertebral growth in a subject having osteogenesis imperfecta (Ol) comprising administering a C-type natriuretic peptide (CNP) to a subject in need thereof, wherein the CNP is administered weekly or biweekly.
[0015] In various embodiments, the osteogenesis imperfecta is Type I, Type II, Type III, Type IV, or Type V Ol. In various embodiments, the osteogenesis imperfecta is Type VI -Type XXI Ol.
[0016] In various embodiments, the subject is further administered adjunct therapy to treat osteogenesis imperfecta. In various embodiment, the adjunct therapy is alendronate, risedronate, ibandronate, zoledronic acid, denosumab, estrogen therapy or hormone therapy, raloxifene, bazedoxifene and estrogen, teriparatide, abaloparatide, or romosozumab. In various embodiments, the adjunct therapy is bisphosphonate therapy. In various embodiments, the bisphoshonate is selected from the group consisting of pamidronate, alendronate, ibandronate and risedronate, or combinations thereof.
[0017] In various embodiments, the bisphosphate is administered orally, intravenously or subcutaneously.
[0018] In various embodiments, the subject has a mutation in a gene selected from the group consisting of COL1A1 , COL1A2, COL2A1 , COL11 A1 , COL9A2, COL10, IFITM5, SERPINF1 , CRTAP, LEPRE1 , PPIB, SERPINH1 , FKBP10, SP7, BMP1 , TMEM38B, WNT1 , CREB3L1 , SPARC, FAM46A or combinations thereof.
[0019] In various embodiments, the CNP is administered intravenously, subcutaneously or orally.
[0020] In various embodiments, CNP therapy alleviates one or more symptoms of osteogenesis imperfecta, including but not limited to, femoral length, vertebral height, reduction in bone fracture frequency, cortical bone tissue mineral density (TMD), cortical bone mineral density (BMD), cortical bone thickness, cortical porosity, trabecular bone volume fraction (BVF), trabecular TMD, trabecular BMD, trabecular bone number and trabecular bone separation.
[0021] In various embodiments, the disclosure contemplates a method of treating a subject having osteogenesis imperfecta and receiving C-type natriuretic peptide (CNP) therapy, comprising i) administering CNP therapy to the subject; ii) obtaining a sample from the subject; iii) measuring levels of NTproCNP and/or N terminal fragment of collagen X (CXM) in a sample collected from the subject in (ii); and iv) altering or changing the dose of CNP to bring NTproCNP levels within +/- 2 SDS of mean NTproCNP for the population.
[0022] In various embodiments, the disclosure provides a method of improving and/or maintaining bone strength in a subject in need thereof comprising administering a C-type natriuretic peptide (CNP) to the subject. In various embodiments, the subject has osteogenesis imperfecta. In various embodiments, the CNP is administered daily, between 2 to 5 times per week, or weekly.
[0023] In various embodiments, CNP therapy dose level or frequency increases if the level of NTproCNP increases, or CNP therapy dose level decreases if the level of NTproCNP decreases.
[0024] Also provided is a method of treating a subject having a osteogenesis imperfecta and receiving C-type natriuretic peptide (CNP) therapy, comprising i) administering CNP therapy to the subject; ii) obtaining a sample from the subject; iii) measuring levels of N terminal fragment of collagen X (CXM) in a sample collected from the subject in (ii); and, iv) increasing CNP therapy dose level or frequency if the level of collagen X decreases.
[0025] In various embodiments, increasing the CNP therapy dose increases the average growth velocity (AGV) in the subject. In various embodiments, the average growth velocity (AGV) in the subject increases over 6 months, over 1 year or over 2 years, or more.
[0026] In various embodiments, increasing CNP therapy dose comprises increasing dose frequency or increasing dose amount.
[0027] In various embodiments, an increase in CNP therapy dose level and decrease in NTproCNP level correlate with improved Annualized Growth Velocity (AGV), reduces the frequency of bone fracture, improves bone mineral density, and/or improves femoral length or vertebral growth in subjects.
[0028] In various embodiments, an increase in CNP therapy dose level and decrease in NTproCNP level extends the duration of growth plate activity in the subject.
[0029] In various embodiments, the levels of NTproCNP are maintained between 2 standard deviations of mean NTproCNP levels based on population analysis. In various embodiments, the levels of NTproCNP are maintained between +/- 2 SDS of the mean NTproCNP for that population. In various embodiments, the NTproCNP is ± 0.5, ± 1 .0, ± 1 .5 or ± 2.0 standard deviations (SDS) of mean NTproCNP levels of a population to which the subject is grouped.
[0030] In various embodiments, the CNP therapy is titrated toward zero NTproCNP SDS if the NTproCNP SDS is below the mean. In various embodiments, the CNP therapy is titrated until zero NTproCNP SDS. In various embodiments, the CNP therapy is titrated until +0.5, + 1 .0, +1 .5 or +2.0 NTproCNP SDS for the population being treated is achieved. In various embodiments, the zero NTproCNP SDS predicts optimal effect size. Optimal size effect is a measure of the expected average normal growth rate of a subject based on population norms.
[0031] In various embodiments, the sample is blood, urine, plasma, saliva, or tissue.
[0032] It is contemplated that CNP therapy to treat a subject having osteogenesis imperfecta comprises administration of CNP variants, conjugates, salts or prodrugs thereof.
[0033] In various embodiments, the osteogenesis imperfecta is Type I, Type II, Type III, Type IV, or Type V Ol. In various embodiments, the osteogenesis imperfecta is Type VI -Type XXI Ol. In various embodiments, the osteogenesis imperfecta is associated with a mutation in a gene selected from the group consisting of COL1A1 , COL1 A2, COL2A1 , COL1 1 A1 , COL9A2, COL10, IFITM5, SERPINF1 , CRTAP, LEPRE1 , PPIB, SERPINH1 , FKBP10, SP7, BMP1 , TMEM38B, WNT1 , CREB3L1 , SPARC, FAM46A or combinations thereof.
[0034] In various embodiments, the CNP variants are useful to treat a subject with osteogenesis imperfecta having a height SDS of less than -1 .0, -1 .5, -2.0, -2.5, or -3.0, and having at least one parent with a height SDS of less than -1 .0, -1 .5, -2.0 or -2.5, optionally wherein the second parent has height within the normal range. In various embodiments, the CNP variants are useful to treat a subject with short stature having a height SDS of between - 2.0 to -3.0. In various embodiments, the CNP variants are useful to treat a subject with short stature having a height SDS of between -2.0 to -2.5.
[0035] In various embodiments, the CNP is a CNP variant selected from the group consisting of PGQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (Pro-Gly-CNP37) (SEQ ID NO: 1 ); GQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (Gly-CNP-37) (SEQ ID NO: 2);
GDLRVDTKSRAAWARLLQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (Gly- CNP53) (SEQ ID NO: 3);
PDLRVDTKSRAAWARLLQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (Pro- CNP53) (SEQ ID NO: 4);
MDLRVDTKSRAAWARLLQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (Met- CNP53) (SEQ ID NO: 5);
DLRVDTKSRAAWARLLQEHPNARKYKGANKKGLSKGCFGLKLDRIGSNSGLGC [CNP- 53(M48N)] (SEQ ID NO: 6);
LRVDTKSRAAWARLLQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-52) (SEQ ID NO: 7);
RVDTKSRAAWARLLQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-51 ) (SEQ ID NO: 8);
VDTKSRAAWARLLQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP- 50) (SEQ ID NO: 9);
DTKSRAAWARLLQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-49) (SEQ ID NO: 10);
TKSRAAWARLLQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-48) (SEQ ID NO: 11 );
KSRAAWARLLQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-47) (SEQ ID NO: 12);
SRAAWARLLQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-46) (SEQ ID NO:
13);
RAAWARLLQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-45) (SEQ ID NO:
14);
AAWARLLQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-44) (SEQ ID NO:
15);
AWARLLQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-43) (SEQ ID NO: 16);
WARLLQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-42) (SEQ ID NO: 17);
ARLLQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-41 ) (SEQ ID NO: 18);
RLLQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-40) (SEQ ID NO: 19);
LLQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-39) (SEQ ID NO: 20);
LQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-38) (SEQ ID NO: 21 );
QEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-37) (SEQ ID NO: 22);
EHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-36) (SEQ ID NO: 23);
HPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-35) (SEQ ID NO: 24);
PNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-34) (SEQ ID NO: 25);
NARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-33) (SEQ ID NO: 26);
ARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-32) (SEQ ID NO: 27) ;
RKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-31 ) (SEQ ID NO: 28);
KYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-30) (SEQ ID NO: 29);
YKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-29) (SEQ ID NO: 30);
KGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-28) (SEQ ID NO: 31 );
GANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-27) (SEQ ID NO: 32);
ANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-26) (SEQ ID NO: 33);
NKKGLSKGCFGLKLDRIGSMSGLGC (CNP-25) (SEQ ID NO: 34);
KKGLSKGCFGLKLDRIGSMSGLGC (CNP-24) (SEQ ID NO: 35);
KGLSKGCFGLKLDRIGSMSGLGC (CNP-23) (SEQ ID NO: 36);
LSKGCFGLKLDRIGSMSGLGC (CNP-21 ) (SEQ ID NO: 37);
SKGCFGLKLDRIGSMSGLGC (CNP-20) (SEQ ID NO: 38);
KGCFGLKLDRIGSMSGLGC (CNP- 19) (SEQ ID NO: 39);
GCFGLKLDRIGSMSGLGC (CNP-18) (SEQ ID NO: 40);
QEHPNARKYKGANKKGLSKGCFGLKLDRIGSNSGLGC [CNP-37(M32N)] (SEQ ID NO: 41 );
PQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (Pro-CNP-37) (SEQ ID NO: 42);
MQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (Met-CNP-37) (SEQ ID NO: 43);
GQEHPNARKYKGANKKGLSKGCFGLKLDRIGSNSGLGC [Gly-CNP-37(M32N)] (SEQ ID NO:
44);
MGQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (Met-Gly-CNP-37) (SEQ ID NO:
45);
PGQEHPQARRYRGAQRRGLSRGCFGLKLDRIGSMSGLGC (SEQ ID NO: 46); PGQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (SEQ ID NO: 47); PGQEHPNARRYRGANRRGLSRGCFGLKLDRIGSMSGLGC (SEQ ID NO: 48); and PGQEHPQARKYKGAQKKGLSKGCFGLKLDRIGSMSGLGC (SEQ ID NO:49).
[0036] In various embodiments, the variant peptide further comprises an acetyl group. In various embodiments, the acetyl group is on the N-terminus of the peptide. In various embodiments, the peptide further comprises an OH or an NH2 group at the C-terminus.
[0037] In various embodiments, the CNP variant composition is an extended release composition. In various embodiments, the composition is a sustained release composition. In various embodiments the sustained or extended release compositions comprises a CNP variant pro-drug.
[0038] In various embodiments, the variant peptide comprises a conjugate moiety. In various embodiments, the conjugate moiety is on a residue of the CNP cyclic domain or at a site other than the CNP cyclic domain. In various embodiments, the conjugate moiety is on a lysine residue. In various embodiments, the conjugate moiety comprises one or more acid moieties. In various embodiments, the acid moiety is a hydrophobic acid.
[0039] In various embodiments, the conjugate moiety comprises one or more acid moieties linked to a hydrophilic spacer. In various embodiments, the hydrophilic spacer is any amino acid. In various embodiments, the hydrophilic spacer is gamma glutamic acid (yGlu). In various embodiments, the hydrophilic spacer is OEG (8-amino-3,6-dioxaoctanoic acid). In various embodiments, the hydrophilic spacer is gamma glutamic acid (yGlu) or OEG (8-amino-3,6- dioxaoctanoic acid). In various embodiments, the hydrophilic spacer is gamma glutamic acid (yGlu) linked to one or two or more OEG (8-amino-3,6-dioxaoctanoic acid). In various embodiments, the acid moiety is a fatty acid. Exemplary fatty acids include short chain, medium
chain, or long chain fatty acids, or a dicarboxylic fatty acid. In various embodiments, the fatty acid is saturated or unsaturated. Contemplated are C-6 to C-20 fatty acids, including but not limited to, C-6, C-8, C-10, C-12, C-14, C-16, C-18 or C-20 fatty acids, saturated or unsaturated. In various embodiments, the fatty acid is decanoic acid, dodecanoic acid, myristic acid, palmitic acid, stearic acid, arachidic acid, or diacids of the same.
[0040] In various embodiments, the acid moiety and the hydrophilic spacer have the structure AEEA-AEEA-yGlu-C18DA.
[0041] In various embodiments, the variant comprises one or more linker groups. In various embodiments, the linker is on a residue of the CNP cyclic domain or at a site other than the CNP cyclic domain. In various embodiments, the linker is on a lysine residue.
[0042] In various embodiments, the linker is a hydrolysable linker.
[0043] In various embodiments, the conjugate moiety is a synthetic polymeric group. In various embodiments, the variant comprises a synthetic polymeric group coupled to the variant through a hydrolysable linker. In various embodiments, the synthetic polymeric group comprises a hydrophilic polymer moiety. In various embodiments, the hydrophilic polymer moiety comprises polyethylene glycol (PEG). In various embodiments, the hydrophilic polymer moiety comprises polyethylene glycol (PEG) having a 6 to 20 atom chain length.
[0044] In various embodiments, the CNP variant is PGQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (Pro-Gly-CNP37) (SEQ ID NO: 1 ); GQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (Gly-CNP-37) (SEQ ID NO: 2); or LQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-38) (SEQ ID NO: 21 ).
[0045] In various embodiments, the variant has the structure: PGQEHPQARRYRGAQRRGLSRGCFGLK(AEEA-AEEA-yGlu-C18DA)LDRIGSMSGLGC (SEQ ID NO: 46), or Ac-PGQEHPQARRYRGAQRRGLSRGCFGLK(AEEA-AEEA-yGlu- C18DA)LDRIGSMSGLGC-OH (SEQ ID NO: 46).
[0046] In various embodiments, the variant is selected from the group consisting of
Ac-PGQEHPQARRYRGAQRRGLSRGCFGLKLDRIGSMSGLGC-OH (SEQ ID NO: 46);
Ac-PGQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC-NH2 (SEQ ID NO: 47);
Ac-PGQEHPNARRYRGANRRGLSRGCFGLKLDRIGSMSGLGC-OH (SEQ ID NO: 48);
AC-PGQEHPNARRYRGANRRGLSRGCFGLKLDRIGSMSGLGC-NH2 (SEQ ID NO: 48);
AC-PGQEHPQARRYRGAQRRGLSRGCFGLKLDRIGSMSGLGC-NH2 (SEQ ID NO: 46);
Ac- PGQEHPQARKYKGAQKKGLSKGCFGLKLDRIGSMSGLGC-NH2 (SEQ ID NO: 49); and
Ac- PGQEHPQARKYKGAQKKGLSKGCFGLKLDRIGSMSGLGC-OH (SEQ ID NO: 49).
[0047] It is further contemplated that the CNP variant includes a conjugate, salt or prodrug of the CNP variants described herein.
[0048] In various embodiments, levels of biomarkers involved in bone and/or collagen growth or formation are measured in a sample before and after administration of CNP. In various embodiments, the biomarker is type II collagen (CTX), urinary N-telopeptide (NTX), tartrate resistant acid phosphatase (TRAP), bone-specific alkaline phosphatase (BSAP), osteocalcin (OC), N-terminal pro-peptide of type I procollagen (P1 NP), procollagen type 1 C-terminal propeptide (P1CP), NTproCNP or CXM. In various embodiments, levels of NTproCNP or CXM are measured in a plasma sample, for example, before and after administration of a CNP variant.
[0049] In various embodiments, the subject is receiving between 0.5 pg/kg and 60 pg/kg CNP therapy. In various embodiments, the subject is receiving between 1.5 pg/kg and 30 pg/kg CNP therapy, between 2.5 pg/kg and 20 pg/kg CNP therapy, between 5 pg/kg and 20 pg/kg CNP therapy, or between 1 .5 pg/kg and 15 pg/kg CNP therapy. In various embodiments, the subject is receiving 15 pg/kg or 30 pg/kg CNP therapy. In various embodiments, the dose may be increased to 30 pg/kg or 60 pg/kg.
[0050] In various embodiments, the subject is receiving about 0.5 pg/kg, about 1 .0 pg/kg, about 1.5 pg/kg, about 2.0 pg/kg, about 2.5 pg/kg, about 3.0 pg/kg, about 3.5 pg/kg, about 4.0 pg/kg, about 4.5 pg/kg, about 5.0 pg/kg, about 6 pg/kg , about 7 pg/kg, about 7.5 pg/kg, about 10 pg/kg, about 12.5 pg/kg, about 15 pg/kg, about 20 pg/kg, about 25 pg/kg, or about 30 pg/kg of CNP or about 60 pg/kg.
[0051] In various embodiments, the biomarker is measured at least 4 hours after administration. In various embodiments, the level of biomarker is measured at least 3 months or 6 months after start of CNP therapy. In various embodiments, the level of biomarker is measured at least every 3 months, 6 months, or 1 year after start of CNP therapy. In various embodiments, the level of biomarker is measured for a duration of at least 3 months, 6 months, 1 year, 2 years, 3 years, 4 years, 5 years, or until puberty/close of growth plates after start of CNP therapy.
[0052] In various embodiments, the level of biomarker a sample is compared to a baseline measurement taken prior to start of CNP therapy. In various embodiments, the level of biomarker in a sample is compared to average levels in normal control patients.
[0053] In various embodiments, CNP therapy dose or frequency is increased when a decrease in NTproCNP indicates an increase in AGV in the subject.
[0054] In various embodiments, the level of CXM in a sample is compared to a baseline measurement taken prior to start of CNP therapy. In various embodiments, the level of CXM in a sample is compared to average levels in normal control patients.
[0055] In various embodiments, the CXM increase indicates increased bone growth, and wherein the dose of CNP frequency or level is increased when there is CXM increase that enhances AGV, reduces the frequency of bone fracture, improves bone mineral density, improves femoral length and/or vertebral growth.
[0056] In various embodiments, the subject is a pediatric subject with open growth plates and receives a dose of about 0.5 pg/kg to about 30 pg/kg daily, five (5) times per week, four (4) times per week, three (3) times per week, two (2) times per week, weekly, bi-weekly, monthly, bi-monthly, or less frequently. In various embodiments, the subject is in early adolescence and received a dose increase to 15 pg/kg, 30 pg/kg daily or 60 pg/kg daily, twice per week, three times per week, 4 times per week, 5 times, per week, weekly, bi-weekly, monthly, bi-monthly, or less frequently. In various embodiments, the subject is an infant and received a dose increase to 30 pg/kg daily, twice per week, three times per week, 4 times per week, 5 times, per week, weekly, bi-weekly, monthly, bi-monthly, or less frequently.
[0057] In various embodiments, the subject receives 20 pg/kg CNP 5 days per week (20x5), 20 pg/kg CNP 3 days per week (20x3) or 10 pg/kg CNP 3 days per week. In various embodiments, the subject is suffering from osteogenesis imperfecta.
[0058] The disclosure also provides a method of selecting initiation of CNP therapy in a subject comprising: i) measuring NTproCNP in the subject at multiple timepoints to establish a baseline NTproCNP level; ii) determining if the NTproCNP levels indicate an SDS within ± 2 of mean NTproCNP levels; and iii) starting treatment with CNP therapy when the subject has NTproCNP levels below mean NTproCNP SDS. In various embodiments, the subject has an NTproCNP SDS of about -2.5, -2.0, -1.5, -1 .0 or -0.5. In various embodiments, CNP therapy is adjusted such that the NTproCNP SDS of the subject is about -0.4, -0.3, -0.2, -0.1 , 0, 0.1 , 0.2,
0.3, 0.4, 0.5, 0.6, 0.7, 0.8, 0.9, 1 .0, 1 .1 , 1 .2, 1 .3, 1 .4, .5, .16, 1 .7, 1 .8, 1 .9 or 2.0 after a modulation in CNP therapy dose level and/or frequency.
[0059] In certain embodiments, the disclosure contemplates a method of selecting initiation of CNP therapy in a subject having osteogenesis imperfecta comprising i) measuring NTproCNP in the subject at multiple timepoints to establish a baseline NTproCNP level; ii) determining if the NTproCNP levels indicate an SDS of zero, below zero or above zero; and iii) starting treatment with CNP therapy when the subject has NTproCNP levels above SDS zero.
[0060] In various embodiments, NTproCNP is measured at 2 weeks, one month, 3 months, and 6 months prior to CNP therapy to establish a baseline NTproCNP level. In various embodiments, NTproCNP is measured by radioimmunoassay.
[0061] Also provided is a method of treating a subject having osteogenesis imperfecta described herein comprising,
[0062] i) identifying whether a subject has a Loss of Function (LoF) or Gain of Function (GoF) variant of a gene related to short stature;
[0063] ii) calculating a polygenic risk score (PRS) of the subject;
[0064] iii) determining if the subject has a LoF variant and a PRS in the bottom 20%; and
[0065] iv) treating the subject with a CNP variant if the subject has a LoF variant and a PRS in the bottom 20%.
[0066] In various embodiments, the subject has a PRS in the bottom 20%, 19%, 18%, 17.5%, 17%, 16.5%, 16%, 15.5%, 15%, 14.5%, 14%, 13.5%, 13%, 12.5%, 12%, 11%, 10%, 9%, 8%, 7.5%, 7%, 6%, 5%, 4%, 3%, 2.5%, 2% or 1 %. In various embodiments, step iii) and iv) is a subject with a CNP variant if the subject has a LoF variant and a PRS in the bottom 12.5%.
[0067] In various embodiments, the PRS is calculated by a genome-wide association study (GWAS) of height. A PRS is an aggregate genetic score that consists of many, common variant effects of small effect each that are summed across the genome (Choi et al. Nat Protoc, 2020). To calculate a height PRS, Genome Wide Association Study (GWAS) association statistics are obtained to indicate the per-variant strength of association with height. These effect sizes are then applied to an independent sample, here the clinical population, by weighing the number of each height-associated allele (0, 1 or 2) by the GWAS effect size, and summing this weighted count across the genome for each clinical sample. PRS can be interpreted such that an individual with a low PRS carries a lower-than-average number of height-increasing genetic
variants, and an individual with a high PRS carries a higher-than-average number of heightincreasing variants.
[0068] In various embodiments, the increase in facial volume, facial sinus volume, and foramen magnum area are measured by magnetic resonance imaging (MRI). In various embodiments, the change in facial volume, facial sinus volume, and foramen magnum area are compared to baseline levels, healthy control subjects or untreated control subjects.
[0069] In various embodiments, the CNP variant is administered subcutaneously. In various embodiments, the CNP variant is administered daily, twice per week, three times per week, 5 times per week, weekly, every 2 weeks, monthly, or less. In various embodiments, the CNP variant is administered at a dose of 30 pg/kg for 3 months, 6 months, 1 year or more. In various embodiments, the dose of CNP variant is decreased to 15 pg/kg when the subject is about 2 years old.
[0070] It is understood that each feature or embodiment, or combination, described herein is a non-limiting, illustrative example of any of the aspects of the invention and, as such, is meant to be combinable with any other feature or embodiment, or combination, described herein. For example, where features are described with language such as “one embodiment”, “some embodiments”, “certain embodiments”, “further embodiment”, “specific exemplary embodiments”, and/or “another embodiment”, each of these types of embodiments is a nonlimiting example of a feature that is intended to be combined with any other feature, or combination of features, described herein without having to list every possible combination.
[0071] Such features or combinations of features apply to any of the aspects of the invention. Where examples of values falling within ranges are disclosed, any of these examples are contemplated as possible endpoints of a range, any and all numeric values between such endpoints are contemplated, and any and all combinations of upper and lower endpoints are envisioned.
BRIEF DESCRIPTION OF THE DRAWINGS
[0072] Figure 1 shows height velocity Z-scores by Ol clinical classification. (N=9 for mild, 10 for moderate and 7 for severe).
[0073] Figure 2 shows CNP levels (left) and NTproCNP levels (right) as measured by Z- scores compared to the normal ranges as a function of Ol clinical classification.
[0074] Figures 3A and 3B illustrate femoral length in mouse model of osteogenesis imperfecta after treatment with CNP +bisphosphonate.
[0075] Figure 4 shows the difference in trabecular and cortical bone of oim/oim animals receiving CNP+bisphosphonate therapy.
[0076] Figure 5 shows micro-CT analysis of various parameters of cortical and trabecular bone in treated and untreated oim/oim mice.
[0077] Figure 6 shows 3D images of trabecular bone in treated and untreated oim/oim mice.
[0078] Figure 7 shows 3D images of cortical bone in treated and untreated oim/oim mice.
[0079] Figure 8 shows mean cortical bone mineral density in treated and untreated oim/oim mice.
[0080] Figure 9 shows mean cortical porosity in treated and untreated oim/oim mice.
[0081] Figure 10 shows mean cortical thickness in treated and untreated oim/oim mice.
[0082] Figure 11 shows mean trabecular bone mineral density in treated and untreated oim/oim mice.
[0083] Figure 12 shows mean trabecular number in treated and untreated oim/oim mice.
[0084] Figure 13 shows mean trabecular thickness in treated and untreated oim/oim mice.
[0085] Figure 14 shows mean trabecular separation in treated and untreated oim/oim mice.
[0086] Figure 15 shows femoral length in WT and oim/oim mice treated with CNP.
[0087] Figure 16 shows vertebral height in WT and oim/oim mice treated with CNP.
[0088] Figures 17A-17E show effects of CNP administration on WT cortical bone by micro-CT analysis: Figure 17A, cortical tissue mineral density; Figure 17B, cortical porosity; Figure 17C, cortical bone mineral density; Figure 17D, cortical thickness; Figure 17E, cortical minimum inertia.
[0089] Figures 18A-18F show effects of CNP administration on WT trabecular bone by micro- CT analysis: Figure 18A, trabecular bone volume fraction; Figure 18B, trabecular tissue mineral density; Figure 18C, trabecular bone mineral density; Figure 18D, trabecular number; Figure 18E, trabecular thickness; Figure 18F, trabecular separation.
[0090] Figures 19A-19E shows effects of CNP administration on oim/oim cortical bone by micro-CT analysis: Figure 19A, cortical tissue mineral density; Figure 19B, cortical porosity;
Figure 19C, cortical bone mineral density; Figure 19D, cortical thickness; Figure 19E, cortical minimum inertia.
[0091] Figure 20A-20F shows effects of CNP administration on oim/oim trabecular bone by micro-CT analysis: Figure 20A, trabecular bone volume fraction; Figure 20B, trabecular tissue mineral density; Figure 20C, trabecular bone mineral density; Figure 20D, trabecular number; Figure 20E, trabecular thickness; Figure 20F, trabecular separation.
[0092] Figure 21 is a table showing the effects of CNP therapy on WT and oim/oim mice in cortical and trabecular bone.
DETAILED DESCRIPTION
[0093] The present application pertains to the discovery that administration of C-type natriuretic peptide (CNP) or CNP variants to patients having osteogenesis imperfecta can alleviate symptoms of diseases, such as bone mineral density, femoral length, and vertebral height.
Definitions
[0094] As used in the specification and the appended claims, the indefinite articles “a” and “an” and the definite article “the” include plural as well as singular referents unless the context clearly dictates otherwise.
[0095] The term “about” or “approximately” means an acceptable error for a particular value as determined by one of ordinary skill in the art, which depends in part on how the value is measured or determined. In certain embodiments, the term “about” or “approximately” means within 1 , 2, 3, or 4 standard deviations. In certain embodiments, the term “about” or “approximately” means within 30%, 25%, 20%, 15%, 10%, 9%, 8%, 7%, 6%, 5%, 4%, 3%, 2%, 1%, 0.5%, or 0.05% of a given value or range. Whenever the term “about” or “approximately” precedes the first numerical value in a series of two or more numerical values, it is understood that the term “about” or “approximately” applies to each one of the numerical values in that series.
[0096] The term “C-type natriuretic peptide” or “CNP” refers to a small, single chain peptide having a 17-amino acid loop structure at the C-terminal end (GenBank Accession No.
NP 077720, for the CNP precursor protein, NPPC) and variants thereof. The 17-mer CNP loop structure, is also referred to as CNP 17, the CNP ring, or CNP cyclic domain. CNP includes the
active 53-amino acid peptide (CNP-53) and the mature 22-amino acid peptide (CNP-22), and peptides of varying lengths between the two peptides.
[0097] In various embodiments, a “CNP variant” is at least about 40%, 50%, 60%, 70%, 75%, 80%, 85%, 90%, or 95% homologous to the wild type NPPC over the same number of amino acid residues. It is further contemplated that a CNP variant peptide may comprise from about 1 to about 53, or 1 to 39, or 1 to 38, or 1 to 37, or 1 to 35, or 1 to 34, or 1 to 31 , or 1 to 27, or 1 to 22, or 10 to 35, or about 15 to about 37 residues of the NPPC polypeptide. In one embodiment, a CNP variant may comprise a sequence of 1 , 2, 3, 4, 5, 6, 7, 8, 9, 10, 1 1 , 12, 13, 14, 15, 16, 17, 18, 19, 20, 21 , 22, 23, 24, 25, 26, 27, 28, 29, 30, 31 , 32, 33, 34, 35,36, 37, 38, 39, 40, 41 , 42, 43, 44, 45, 46, 47, 48, 49, 50, 51 , 52, or 53 amino acids derived from the NPPC polypeptide. CNP variant also includes conjugates, salts or prodrugs of CNP variants described herein.
“CNP therapy” refers to administration of a CNP variant to treat a subject having a bone-related disorder, skeletal dysplasia or short stature as described herein.
[0098] The term “conjugate moiety” refers to a moiety that is conjugated to the variant peptide. Conjugate moieties include a lipid, fatty acid, hydrophilic spacer, synthetic polymer, linker, or optionally, combinations thereof.
[0099] The term “effective amount” refers to a dosage sufficient to produce a desired result on a health condition, pathology, or disease of a subject or for a diagnostic purpose. The desired result may comprise a subjective or objective improvement in the recipient of the dosage. "Therapeutically effective amount" refers to that amount of an agent effective to produce the intended beneficial effect on health. An appropriate “effective” amount in any individual case may be determined by one of ordinary skill in the art using routine experimentation. It will be understood that the specific dose level and frequency of dosage for any particular patient may be varied and will depend upon a variety of factors, including the activity of the specific compound employed; the bioavailability, metabolic stability, rate of excretion and length of action of that compound; the mode and time of administration of the compound; the age, body weight, general health, sex, and diet of the patient; and the severity of the particular condition.
[0100] "Substantially pure" or "isolated" means an object species is the predominant species present (/.e., on a molar basis, more abundant than any other individual macromolecular species in the composition), and a substantially purified fraction is a composition wherein the object species comprises at least about 50% (on a molar basis) of all macromolecular species present. In one embodiment, a substantially pure composition means that the species of
interest comprises at least about 70%, 75%, 80%, 85%, 90%, 95%, 98% or more of the macromolecular species present in the composition on a molar or weight basis. The object species is purified to essential homogeneity (contaminant species cannot be detected in the composition by conventional detection methods) if the composition consists essentially of a single macromolecular species. Solvent species, small molecules (<500 Daltons), stabilizers (e.g., BSA), and elemental ion species are not considered macromolecular species for purposes of this definition. In an embodiment, the compounds of the disclosure are substantially pure or isolated. In another embodiment, the compounds of the disclosure are substantially pure or isolated with respect to the macromolecular starting materials used in their production. In yet another embodiment, the pharmaceutical compositions of the disclosure comprise a substantially pure or isolated CNP variant admixed with one or more pharmaceutically acceptable excipients, carriers or diluents, and optionally with another biologically active agent.
[0101] "Treatment" refers to prophylactic treatment or therapeutic treatment or diagnostic treatment. In certain embodiments, “treatment” refers to administration of a compound or composition to a subject for therapeutic, prophylactic or diagnostic purposes.
[0102] A "prophylactic" treatment is a treatment administered to a subject who does not exhibit signs of a disease or exhibits only early signs of the disease, for the purpose of decreasing the risk of developing pathology. The compounds or compositions of the disclosure may be given as a prophylactic treatment to reduce the likelihood of developing a pathology or to minimize the severity of the pathology, if developed.
[0103] A "therapeutic" treatment is a treatment administered to a subject who exhibits signs or symptoms of pathology for the purpose of diminishing or eliminating those signs or symptoms. The signs or symptoms may be biochemical, cellular, histological, functional or physical, subjective or objective. The compounds of the disclosure may also be given as a therapeutic treatment or for diagnosis.
[0104] “Diagnostic" means identifying the presence, extent and/or nature of a pathologic condition. Diagnostic methods differ in their specificity and selectivity. While a particular diagnostic method may not provide a definitive diagnosis of a condition, it suffices if the method provides a positive indication that aids in diagnosis.
[0105] “Bone- or cartilage-associated biomarker” or “bone- or cartilage-associated marker” refers to a growth factor, enzyme, protein, or other detectable biological substance or moiety whose level is increased or decreased in association with, e.g., cartilage turnover, cartilage
formation, cartilage growth, bone resorption, bone formation, bone growth, or combinations thereof. Such biomarkers may be measured before, during and/or after administration of a CNP variant as described herein. Exemplary bone- or cartilage-associated biomarkers include, but are not limited to, CNP, cGMP, propeptides of collagen type II and fragments thereof, collagen type II and fragments thereof, propeptides of collagen type I and fragments thereof, collagen type I and fragments thereof, osteocalcin (OC), proliferating cell nuclear antigen (PCNA), aggrecan chondroitin sulfate, collagen X, N terminal fragment of collagen X (CXM) alkaline phosphatase, type II collagen (CTX), urinary N-telopeptide (NTX), tartrate resistant acid phosphatase (TRAP), bone-specific alkaline phosphatase (BSAP), N-terminal pro-peptide of type I procollagen (P1 NP), procollagen type 1 C-terminal propeptide (P1 CP), or NTproCNP. Cartilage- and bone-associated biomarkers can be measured in any appropriate biological sample, including but not limited to tissues, blood, serum, plasma, cerebrospinal fluid, synovial fluid and urine.
[0106] "Pharmaceutical composition" or "formulation" refers to a composition suitable for pharmaceutical use in subject animal, including humans and mammals. A pharmaceutical composition comprises a therapeutically effective amount of CNP variant, optionally another biologically active agent, and optionally a pharmaceutically acceptable excipient, carrier or diluent. In an embodiment, a pharmaceutical composition encompasses a composition comprising the active ingredient(s), and the inert ingredient(s) that make up the carrier, as well as any product that results, directly or indirectly, from combination, complexation or aggregation of any two or more of the ingredients, or from dissociation of one or more of the ingredients, or from other types of reactions or interactions of one or more of the ingredients. Accordingly, the pharmaceutical compositions of the present disclosure encompass any composition made by admixing a compound of the disclosure and a pharmaceutically acceptable excipient, carrier or diluent.
[0107] "Pharmaceutically acceptable carrier" refers to any of the standard pharmaceutical carriers, buffers, and the like, such as a phosphate buffered saline solution, 5% aqueous solution of dextrose, and emulsions (e.g., an oil/water or water/oil emulsion). Non-limiting examples of excipients include adjuvants, binders, fillers, diluents, disintegrants, emulsifying agents, wetting agents, lubricants, glidants, sweetening agents, flavoring agents, and coloring agents. Suitable pharmaceutical carriers, excipients and diluents are described in Remington's Pharmaceutical Sciences, 19th Ed. (Mack Publishing Co., Easton, 1995). Preferred pharmaceutical carriers depend upon the intended mode of administration of the active agent.
Typical modes of administration include enteral (e.g., oral) or parenteral (e.g., subcutaneous, intramuscular, intravenous or intraperitoneal injection; or topical, transdermal, or transmucosal administration).
[0108] A "pharmaceutically acceptable salt" is a salt that can be formulated into a compound for pharmaceutical use, including but not limited to metal salts (e.g., sodium, potassium, magnesium, calcium, etc.) and salts of ammonia or organic amines.
[0109] By "pharmaceutically acceptable” or “pharmacologically acceptable” is meant a material that is not biologically or otherwise undesirable, i.e., the material may be administered to an individual without causing any undesirable biological effects or without interacting in a deleterious manner with any of the components of the composition in which it is contained or with any components present on or in the body of the individual.
[0110] “Physiological conditions” refer to conditions in the body of an animal (e.g., a human). Physiological conditions include, but are not limited to, body temperature and an aqueous environment of physiologic ionic strength, pH and enzymes. Physiological conditions also encompass conditions in the body of a particular subject which differ from the “normal” conditions present in the majority of subjects, e.g., which differ from the normal human body temperature of approximately 37 °C or differ from the normal human blood pH of approximately 7.4.
[0111] By “physiological pH” or a “pH in a physiological range” is meant a pH in the range of approximately 7.0 to 8.0 inclusive, more typically in the range of approximately 7.2 to 7.6 inclusive.
[0112] As used herein, the term “subject” encompasses mammals and non-mammals. Examples of mammals include, but are not limited to, any member of the mammalian class: humans, non-human primates such as chimpanzees, and other apes and monkey species; farm animals such as cattle, horses, sheep, goats, swine; domestic animals such as rabbits, dogs, and cats; laboratory animals including rodents, such as rats, mice and guinea pigs, and the like. Examples of non-mammals include, but are not limited to, birds, fish, and the like. The term does not denote a particular age or gender. In various embodiments, the subject is human. In various embodiments the subject is a child or adolescent. In various embodiments, the subject is an infant. In various embodiments, the subject is older than 3, older than 2, older than 1 , or older than 6 months in age.
C-type Natriuretic Peptide
[0113] C-type natriuretic peptide (CNP) (Biochem. Biophys. Res. Common., 168: 863-870 (1990) (Gen Bank Accession No. NP 077720, for the CNP precursor protein, NPPC) (J. Hypertens., 10: 907-912 (1992)) is a small, single chain peptide in a family of peptides (ANP, BNP, CNP) having a 17-amino acid loop structure (Levin et al., N. Engl. J. Med., 339:863-870 (1998)) and have important roles in multiple biological processes. CNP interacts with natriuretic peptide receptor-B (NPR-B, GC-B) to stimulate the generation of cyclic- guanosine monophosphate (cGMP) (J. Hypertens., 10:11 11 -11 14 (1992)). CNP is expressed more widely, including in the central nervous system, reproductive tract, bone and endothelium of blood vessels (Gardner et al., Hypertension, 49:419-426 (2007)).
[0114] In humans, CNP is initially produced from the natriuretic peptide precursor C (NPPC) gene as a single chain 126-amino acid pre-pro polypeptide (Sudoh et al., Biochem. Biophys. Res. Common., 168: 863-870 (1990)). Removal of the signal peptide yields pro-CNP, and further cleavage by the endoprotease furin generates an active 53-amino acid peptide (CNP- 53), which is secreted and cleaved again by an unknown enzyme to produce the mature 22- amino acid peptide (CNP-22) (Wu, J. Biol. Chem. 278: 25847-852 (2003)). CNP-53 and CNP- 22 differ in their distribution, with CNP-53 predominating in tissues, while CNP-22 is mainly found in plasma and cerebrospinal fluid (J. Alfonzo, Recept. Signal. Transduct. Res., 26: 269- 297 (2006)). Both CNP-53 and CNP-22 bind similarly to NPR-B.
[0115] Downstream signaling mediated by cGMP generation influences a diverse array of biological processes that include endochondral ossification. For example, knockout of either CNP or NPR-B in mouse models results in animals having a dwarfed phenotype with shorter long bones and vertebrae. Mutations in human NPR-B that block proper CNP signaling have been identified and result in dwarfism (Olney, et al., J. Clin. Endocrinol. Metab. 91 (4): 1229- 1232 (2006); Bartels, et al., Am. J. Hum. Genet. 75: 27-34 (2004)). In contrast, mice engineered to produce elevated levels of CNP display elongated long bones and vertebrae.
[0116] Natural CNP gene and polypeptide have been previously described. U.S. Patent No. 5,352,770 discloses isolated and purified CNP-22 from porcine brain identical in sequence to human CNP and its use in treating cardiovascular indications. U.S. Patent No. 6,034,231 discloses the human gene and polypeptide of pre-proCNP (126 amino acids) and the human CNP-53 gene and polypeptide. The mature CNP is a 22-amino acid peptide (CNP-22). Certain CNP variants are disclosed in US Patent 8,198,242, incorporated by reference herein.
[0117] In various embodiments, CNP of the disclosure includes truncated CNP ranging from human CNP-17 (hCNP-17) to human CNP-53 (hCNP-53), and having wild-type amino acid
sequences derived from hCNP-53 and also variants thereof. CNP contemplated herein also includes neutral endopeptidase resistant CNP which are >30 amino acids, and CNP having a size >3 kDa. Truncated CNP peptides include:
PGQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (Pro-Gly-CNP37) (SEQ ID NO: 1 ); GQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (Gly-CNP-37) (SEQ ID NO: 2);
GDLRVDTKSRAAWARLLQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (Gly- CNP53) (SEQ ID NO: 3); PDLRVDTKSRAAWARLLQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (Pro- CNP53) (SEQ ID NO: 4);
MDLRVDTKSRAAWARLLQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (Met- CNP53) (SEQ ID NO: 5);
DLRVDTKSRAAWARLLQEHPNARKYKGANKKGLSKGCFGLKLDRIGSNSGLGC [CNP- 53(M48N)] (SEQ ID NO: 6);
LRVDTKSRAAWARLLQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-52) (SEQ ID NO: 7);
RVDTKSRAAWARLLQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-51 ) (SEQ ID NO: 8);
VDTKSRAAWARLLQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP- 50) (SEQ ID NO: 9);
DTKSRAAWARLLQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-49) (SEQ ID NO: 10);
TKSRAAWARLLQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-48) (SEQ ID NO: 11 );
KSRAAWARLLQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-47) (SEQ ID NO: 12);
SRAAWARLLQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-46) (SEQ ID NO:
13);
RAAWARLLQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-45) (SEQ ID NO:
14);
AAWARLLQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-44) (SEQ ID NO:
15);
AWARLLQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-43) (SEQ ID NO: 16);
WARLLQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-42) (SEQ ID NO: 17);
ARLLQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-41) (SEQ ID NO: 18);
RLLQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-40) (SEQ ID NO: 19);
LLQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-39) (SEQ ID NO: 20);
LQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-38) (SEQ ID NO: 21 );
QEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-37) (SEQ ID NO: 22);
EHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-36) (SEQ ID NO: 23);
HPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-35) (SEQ ID NO: 24);
PNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-34) (SEQ ID NO: 25);
NARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-33) (SEQ ID NO: 26);
ARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-32) (SEQ ID NO: 27);
RKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-31 ) (SEQ ID NO: 28);
KYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-30) (SEQ ID NO: 29);
YKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-29) (SEQ ID NO: 30);
KGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-28) (SEQ ID NO: 31 );
GANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-27) (SEQ ID NO: 32);
ANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-26) (SEQ ID NO: 33);
NKKGLSKGCFGLKLDRIGSMSGLGC (CNP-25) (SEQ ID NO: 34);
KKGLSKGCFGLKLDRIGSMSGLGC (CNP-24) (SEQ ID NO: 35);
KGLSKGCFGLKLDRIGSMSGLGC (CNP-23) (SEQ ID NO: 36);
LSKGCFGLKLDRIGSMSGLGC (CNP-21 ) (SEQ ID NO: 37);
SKGCFGLKLDRIGSMSGLGC (CNP-20) (SEQ ID NO: 38);
KGCFGLKLDRIGSMSGLGC (CNP-19) (SEQ ID NO: 39);
GCFGLKLDRIGSMSGLGC (CNP-18) (SEQ ID NO: 40);
QEHPNARKYKGANKKGLSKGCFGLKLDRIGSNSGLGC [CNP-37(M32N)] (SEQ ID NO: 41 );
PQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (Pro-CNP-37) (SEQ ID NO: 42);
MQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (Met-CNP-37) (SEQ ID NO: 43);
GQEHPNARKYKGANKKGLSKGCFGLKLDRIGSNSGLGC [Gly-CNP-37(M32N)] (SEQ ID NO: 44);
MGQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (Met-Gly-CNP-37) (SEQ ID NO: 45);
PGQEHPQARRYRGAQRRGLSRGCFGLKLDRIGSMSGLGC (SEQ ID NO: 46); PGQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (SEQ ID NO: 47); PGQEHPNARRYRGANRRGLSRGCFGLKLDRIGSMSGLGC (SEQ ID NO: 48); and PGQEHPQARKYKGAQKKGLSKGCFGLKLDRIGSMSGLGC (SEQ ID NO: 49).
[0118] In various embodiments, the CNP variant peptides are modified CNP-37 or CNP-38 peptides, optionally having mutation(s)/substitution(s) at the furin cleavage site, and/or containing glycine or proline-glycine at the N-terminus. Exemplary CNP-37 variants include but are not limited to:
QEHPNARKYKGANKKGLSKGCFGLKLDRIGSNSGLGC [CNP-37(M32N); SEQ ID NO: 41]; MQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (Met-CNP-37; SEQ ID NO: 43); PQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (Pro-CNP-37; SEQ ID NO: 42); GQEHPNARKYKGANKKGLSKGCFGLKLDRIGSNSGLGC [Gly-CNP-37 (M32N); SEQ ID NO:
44];
PGQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (Pro-Gly-CNP-37; SEQ ID NO:1 ); MGQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (Met-Gly-CNP-37; SEQ ID NO:
45);
GQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (Gly-CNP-37: SEQ ID NO: 2) GQEHPNARKYKGANPKGLSKGCFGLKLDRIGSMSGLGC (SEQ ID NO: 50); GQEHPNARKYKGANQKGLSKGCFGLKLDRIGSMSGLGC (SEQ ID NO: 51 );
GQEHPNARKYKGANQQGLSKGCFGLKLDRIGSMSGLGC (SEQ ID NO: 52); and GQEHPNARKYKGANKPGLSKGCFGLKLDRIGSMSGLGC (SEQ ID NO: 53);
[0119] In various embodiments, CNP variants of the disclosure include PGQEHPQARRYRGAQRRGLSRGCFGLKLDRIGSMSGLGC (SEQ ID NO: 46); PGQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (SEQ ID NO: 47); PGQEHPNARRYRGANRRGLSRGCFGLKLDRIGSMSGLGC (SEQ ID NO: 48); or PGQEHPQARKYKGAQKKGLSKGCFGLKLDRIGSMSGLGC (SEQ ID NO: 49).
[0120] The variant peptide may further comprise an acetyl group. In various embodiments, the acetyl group is on the N-terminus of the peptide. In various embodiments, the peptide further comprises an OH or an NH2 group at the C-terminus.
[0121] The variant peptide may comprise a conjugate moiety. In various embodiments, the conjugate moiety is on a residue of the CNP cyclic domain or at a site other than the CNP cyclic domain. In various embodiments, the conjugate moiety is on a lysine residue. In various embodiments, the conjugate moiety comprises one or more acid moieties. In various embodiments, the acid moiety is a hydrophobic acid.
[0122] In various embodiments, the variant has the structure: PGQEHPQARRYRGAQRRGLSRGCFGLK(AEEA-AEEA-yGlu-C18DA)LDRIGSMSGLGC (SEQ ID NO: 46), or Ac-PGQEHPQARRYRGAQRRGLSRGCFGLK(AEEA-AEEA-yGlu-
C18DA)LDRIGSMSGLGC-OH (SEQ ID NO: 46).
[0123] In various embodiments, the variant is selected from the group consisting of
Ac-PGQEHPQARRYRGAQRRGLSRGCFGLKLDRIGSMSGLGC-OH (SEQ ID NO: 46);
AC-PGQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC-NH2 (SEQ ID NO: 47);
Ac-PGQEHPNARRYRGANRRGLSRGCFGLKLDRIGSMSGLGC-OH (SEQ ID NO: 48);
AC-PGQEHPNARRYRGANRRGLSRGCFGLKLDRIGSMSGLGC-NH2 (SEQ ID NO: 48);
AC-PGQEHPQARRYRGAQRRGLSRGCFGLKLDRIGSMSGLGC-NH2 (SEQ ID NO: 46);
Ac- PGQEHPQARKYKGAQKKGLSKGCFGLKLDRIGSMSGLGC-NH2 (SEQ ID NO: 49); and
Ac- PGQEHPQARKYKGAQKKGLSKGCFGLKLDRIGSMSGLGC-OH (SEQ ID NO: 49).
[0124] In various embodiments, the CNP variant is Ac-
PGQEHPQARRYRGAQRRGLSRGCFGLK(AEEA-AEEA-yGlu-C18DA)LDRIGSMSGLGC-OH (SEQ ID NO: 46). In various embodiments, the CNP variant is Ac-
PGQEHPNARKYKGANKKGLSKGCFGLK(AEEA-AEEA-yGlu-C18DA)LDRIGSMSGLGC-OH (SEQ ID NO: 47). In various embodiments, the CNP variant is
PGQEHPNARKYKGANKKGLSKGCFGLK(AEEA-AEEA-yGlu-C18DA)LDRIGSMSGLGC-OH (SEQ ID NO: 47).
[0125] It is further contemplated that the CNP variant is conjugated to or is complexed to a moiety, e.g., a conjugate moiety, that confers increased stability or half-life. In various embodiments, the conjugate moiety is complexed via a non-covalent bond or is attached by a covalent bond. The moiety may be non-covalently attached with the peptide via electrostatic interactions. Alternatively, the moiety may be covalently associated to the peptide via one or more linker moieties. Linkers can be cleavable and non-cleavable linkers. Cleavable linkers may be cleaved via enzymes, nucleophilic/basic reagents, reducing agents, photo-irradiation,
electrophilic/acidic reagents, organometallic and metal reagents, or oxidizing reagents. Linkers may also be self-immolative linkers. Exemplary linkers include, but are not limited to, N- succinim idyl-3-(2-pyridyldith iol) propionate (SPDP), iminothiolane (IT), bifunctional derivatives of imidoesters (such as dimethyl adipimidate HCL), active esters (such as disuccinimidyl suberate), aldehydes (such as glutaraldehyde), bis-azido compounds (such as bis (p- azidobenzoyl) hexanediamine), bis-diazonium derivatives (such as bis-(p-diazoniumbenzoyl)- ethylenediamine), diisocyanates (such as tolyene 2,6-diisocyanate), and bis-active fluorine compounds (such as 1 ,5-difluoro-2,4-dinitrobenzene), beta alanine, 4-aminobutyric acid (GABA), 2-aminoethoxy acid (AEA), aminoethoxy-2-ethoxy acetic acid (AEEA), 5 aminovaleric acid (AVA), 6-aminocaproic acid (Abx), a vicinal diol cleavable linker, Trimethyl Lock Lactonization, p-alkoxyphenyl carbamate, bicin, peptoid or bicin-type linkers, and electronic linkers as described herein.
[0126] It is contemplated that the linker is attached to a residue of the CNP variant within the CNP cyclic domain or at a site other than the CNP cyclic domain. In various embodiments, the linker is attached to a lysine residue. In various embodiments, the linker is attached to a lysine residue in the CNP cyclic domain.
[0127] In various embodiments, the CNP variant is attached to the conjugate moiety via the linker. In various embodiments, the linker is attached to the conjugate moiety via the hydrophilic spacer of the conjugate moiety.
[0128] In various embodiments, the linker is a hydrolysable linker.
[0129] In various embodiments the linker is a peptoid or electronic linker. In various embodiments the linker is a peptoid linker. In various embodiments the linker is an electronic linker. In various embodiments, the linker comprises an SO2 moiety. It is further contemplated that linkers are modified by substitution on the R groups. For example, peptoid-type linkers include the structures as set out below:
[0130] In various embodiments, the moiety conjugated to the peptide is a synthetic polymer such as polyethylene glycol, a linker, a lipid moiety or fatty acid, or a combination thereof. In various embodiments, the CNP variant is conjugated with a fatty acid, an amino acid, a spacer and a linker. In various embodiments, the CNP variant is conjugated with a fatty acid, an amino acid, a polyethylene glycol spacer or a polyethylene glycol derivative spacer, and a linker. In various embodiments, the CNP variant is conjugated with a fatty acid, an amino acid, a spacer, and a linker, wherein the spacer comprises a substituted C-6 to C-20 alkyl chain or any amino acid, or a combination of both, wherein the carbon atoms of the alkyl chain can be replaced by one or more of O, NH, N(C-1 to C-6 alkyl), or carbonyl groups.
[0131] In various embodiments, the CNP variant is conjugated with a fatty acid. It is hypothesized that the lipid technology increases the serum half-life of the CNP variant allowing for less frequent injections and/or improved oral delivery. In various embodiments, the fatty acid is a short chain, medium chain, long chain fatty acid, or a dicarboxylic fatty acid. In various embodiments, the fatty acid is saturated or unsaturated. In various embodiments, the fatty acid is a C-6 to C-20 fatty acid. In various embodiments, the fatty acid is a C-6, C-8, C-10, C-12, C- 14, C-16, C-18 or C-20 fatty acid. In various embodiments, the fatty acid is decanoic acid, dodecanoic acid, myristic acid, palmitic acid, stearic acid, arachidic acid, or diacids of the same. In various embodiments, the fatty acid is conjugated to a lysine residue.
[0132] In various embodiments, it is contemplated that the CNP variants described herein comprise a conjugate moiety as described herein. It is contemplated that the conjugate moiety is on a residue of the CNP cyclic domain or at a site other than the CNP cyclic domain. In various embodiments, the conjugate moiety is on a lysine residue. In various embodiments, the conjugate moiety comprises one or more acid moieties. In various embodiments, the acid
moiety is a fatty acid. Exemplary CNP variants and peptide conjugates are described in International Patent Application No. PCT/US2020/051100 and USSN 17/642,150, incorporated by reference herein in their entirety. Variants, conjugates and salts of CNP are disclosed in USSN 17/634,034, herein incorporated by reference.
[0133] In various embodiments, the conjugate moiety comprises an acid moiety linked to a hydrophilic spacer. In various embodiments, the hydrophilic spacer is a substituted C-6 to C-20 alkyl chain or any amino acid, or a combination of both, wherein the carbon atoms of the alkyl chain can be replaced by one or more of O, NH, N(C-1 to C-6 alkyl), or carbonyl groups. In various embodiments, the hydrophilic spacer is any amino acid. In various embodiments, the hydrophilic spacer is gamma glutamic acid (yGlu) . In various embodiments, the hydrophilic spacer is a substituted C-6 to C-20 alkyl chain. In various embodiments, the hydrophilic spacer is a substituted C-6, C-8, C-10, C-12, C-14, C-16, C-18 or C-20 alkyl chain. In various embodiments, the hydrophilic spacer is a substituted C-9 to C-18 alkyl chain. In various embodiments, the hydrophilic spacer is a substituted C-18 alkyl chain. In various embodiments, the hydrophilic spacer is a substituted C-9 alkyl chain. In various embodiments, the hydrophilic spacer is one or more OEG (8-amino-3,6-dioxaoctanoic acid) groups. In various embodiments, the hydrophilic spacer is one or two OEG (8-amino-3,6-dioxaoctanoic acid) groups. In various embodiments, the hydrophilic spacer is OEG (8-amino-3,6-dioxaoctanoic acid). In various embodiments, the spacer is OEG (8-amino-3,6-dioxaoctanoic acid) or yGlu. In various embodiments, the hydrophilic spacer is gamma glutamic acid (yGlu) linked to one or more OEG (8-amino-3,6-dioxaoctanoic acid) groups. In various embodiments, the hydrophilic spacer is gamma glutamic acid (yGlu) linked to one or two OEG (8-amino-3,6-dioxaoctanoic acid) groups (diEG). In various embodiments, the acid moiety and the hydrophilic spacer have the structure AEEA-AEEA-yGlu-C18DA.
[0134] In various embodiments, the disclosure contemplates use of CNP variants comprising hydrophilic or water soluble polymers (e.g., oxygenated alkyl chains, wherein the carbon atoms can be replaced with one or more oxygen atoms, such as polyethylene glycol (PEG) or polyethylene oxide (PEG) and the like). In various embodiments, the water soluble polymers can vary in type (e.g., homopolymer or copolymer; random, alternating or block copolymer; linear or branched; monodispersed or polydispersed), linkage (e.g., hydrolysable or stable linkage such as, e.g., amide, imine, aminal, alkylene, or ester bond), conjugation site (e.g., at the N-terminus, internal, and/or C-terminus), and length (e.g., from about 0.2, 0.4 or 0.6 kDa to about 2, 5, 10, 25, 50 or 100 kDa). The hydrophilic or water-soluble polymer can be conjugated
to the CNP variant by means of N-hydroxy succinimide (NHS)- or aldehyde-based chemistry or other chemistry, as is known in the art. In various embodiments, negatively charged PEG-CNP variants can be designed for reduced renal clearance, including but not limited to use of carboxylated, sulfated and phosphorylated compounds (Caliceti, Adv. Drug Deliv. Rev., 55: 1261 -77 (2003); Perlman, J. Clin. Endo. Metab., 88: 3227-35 (2003); Pitkin, Antimicrob. Ag.
Chemo., 29: 440-444 (1986); Vehaskari, Kidney Int’l, 22: 127-135 (1982)). In one embodiment, the PEG (or PEG) moiety contains carboxyl group(s), sulfate group(s), and/or phosphate group(s).
[0135] In another embodiment, the hydrophilic polymer (e.g., PEG or PEG) moieties conjugated to the N-terminus, C-terminus and/or internal site(s) of CNP variants described herein contain one or more functional groups that are positively charged under physiological conditions. Such moieties are designed, inter alia, to improve distribution of such conjugated CNP variants to cartilage tissues. In one embodiment, PEG moieties contain one or more primary, secondary or tertiary amino groups, quaternary ammonium groups, and/or other amine- containing (e.g., urea) groups.
Methods of Using CNP Variants
[0136] Achondroplasia is a result of an autosomal dominant mutation in the gene for fibroblast growth factor receptor 3 (FGFR-3), which causes an abnormality of cartilage formation. FGFR-3 normally has a negative regulatory effect on chondrocyte growth, and hence bone growth. In achondroplasia, the mutated form of FGFR-3 is constitutively active, which leads to severely shortened bones. In humans activating mutations of FGFR-3 are the primary cause of genetic dwarfism. Mice having activated FGFR-3 serve as a model of achondroplasia, the most common form of the skeletal dysplasias, and overexpression of CNP rescues these animals from dwarfism. Accordingly, functional variants of CNP are potential therapeutics for treatment of the various skeletal dysplasias.
[0137] By stimulating matrix production, proliferation and differentiation of chondrocytes and increasing long bone growth, the CNP variants of the disclosure are useful for treating mammals, including humans, suffering from a bone-related disorder, such as a skeletal dysplasia or short stature. Non-limiting examples of CNP-responsive bone-related disorders skeletal dysplasias and short stature disorders include achondroplasia, hypochondroplasia, short stature, idiopathic short stature, dwarfism, osteochondrodysplasias, thanatophoric dysplasia, osteogenesis imperfecta, osteogenesis congenita, achondrogenesis, homozygous achondroplasia, chondrodysplasia congenita, campomelic dysplasia, congenital lethal
hypophosphatasia, perinatal lethal type of osteogenesis congenita, short-rib polydactyly syndromes, rhizomelic type of chondrodysplasia congenita, Jansen-type metaphyseal dysplasia, spondyloepiphyseal dysplasia congenital, atelosteogenesis, diastrophic dysplasia, congenital short femur, Langer-type mesomelic dysplasia, Nievergelt-type mesomelic dysplasia, Robinow syndrome, Reinhardt syndrome, acrodysostosis, peripheral dysostosis, Kniest dysplasia, fibrochondrogenesis, Roberts syndrome, acromesomelic dysplasia, micromelia, Morquio syndrome, Kniest syndrome, metatrophic dysplasia, and spondyloepimetaphyseal dysplasia, disorders related to NPR2 mutation, SHOX mutation (Turner’s syndrome/Leri Weill), PTPN11 mutations (Noonan’s syndrome) and IGF1 R mutation. In various embodiments, the bone related disorder is osteogenesis imperfecta.
[0138] Additional short stature and growth plate disorders contemplated by the methods include disorders related to mutations in collagen (COL2A1 , COL11 A1 , COL9A2, COL10), aggrecan (ACAN), Indian hedgehog (IHH), PTPN11 , NPR2, NPPC, FGFR3, or IGF1 R SHOX, DTL, and pregnancy-associated plasma protein A2 (PAPPA2), or combinations thereof.
[0139] Further, the CNP variants are useful as an adjunct or alternative to growth hormone for treating idiopathic short stature and other skeletal dysplasias.
[0140] Growth plate disorders include disorders that result in short stature or abnormal bone growth and that may be the result of a genetic mutation in a gene involved in bone growth, including collagen (COL2A1 , COL11A1 , COL9A2, COL10), aggrecan (ACAN), indian hedgehog (IHH), PTPN11 , NPR2, NPPC, FGFR3 or IGF1 R. In various embodiments, the growth plate disorder or short stature is associated with one or more mutations in a gene associated with a RASopathy. In various embodiments, a subject with a growth plate disorder is heterozygous for a mutation in a growth plate gene. In various embodiments, the mutation is a loss-of-function mutation. In various embodiments, the mutation is a gain-of-function mutation. Growth plate disorders include, but are not limited to, familial short stature, dominant familial short stature which is also known as dominant inherited short stature, or idiopathic short stature. See, e.g., Plachy et al., J Clin Endocrinol Metab 104: 4273-4281 , 2019.
[0141] Mutations in ACAN can give rise to familial osteochondritis dissecans and short stature and eventually osteoarthritis, characterized by areas of bone damage (or lesions) caused by the detachment of cartilage and sometimes bone from the end of the bone at a joint. It has been suggested that the disorganized cartilage network in growing bones impairs their growth, leading to short stature. A mutation associated with ACAN and short stature includes Val2303Met. See Stattin et al., Am J Hum Genet 86(2):126-37, 2010. It is contemplated that
patients with a mutation in ACAN resulting in short stature would benefit from treatment with CNP as administration may be able to increase height in these patients by the known interaction of CNP with FGFR3.
[0142] The natriuretic peptide system, including receptor NPR2, has been shown to be involved in regulation of endochondral bone growth (Vasques et al., orm Res Pediat 82:222- 229, 2014). Studies have shown that homozygous or compound heterozygous loss-of-function mutations in NPR2 cause acromesomelic dysplasia type Maroteaux (AMDM), which is a skeletal dysplasia having extremely short stature (Vasquez et al., 2014, supra). There are reports implicating heterozygous loss-of-function (such as dominant negative) NPR2 mutations as a cause of short stature, whereas gain-of-function NPR2 heterozygous mutations have been found to be responsible for tall stature (Vasquez et al., 2014, supra). In view of CNP’s interaction with NPR2 to stimulate cGMP generation, increasing cGMP levels is desirable in these conditions and would have therapeutic benefit in the management of the complications from these diseases and conditions.
[0143] Heterozygous mutations of NPR2 are believed to result in idiopathic short stature and other forms of short stature. Mutations in the NPR2 gene are set out below and described in Amano et al., J Clin Endocrinol Metab 99:E713-718, 2014, Hisado-Oliva et al., J Clin Endocrinol Metab 100:E1 133-1 142, 2015 and Vasques et al., J Clin Endocrinol Metab 98:E1636-1644, 2013, hereby incorporated by reference. It is contemplated that a subject having short stature to be treated with a CNP variant as described herein has a height SDS of less than -1 .0, -1.5, - 2.0, -2.5, or -3.0, and has at least one parent with a height SDS of less than -1.0, -1 .5, -2.0 or - 2.5, optionally wherein the second parent has height within the normal range. In various embodiments, the CNP variants are useful to treat a subject with short stature having a height SDS of between -2.0 to -3.0. In various embodiments, the CNP variants are useful to treat a subject with short stature having a height SDS of between -2.0 to -2.5. However, because de novo mutations in NPR2 can result in short stature as defined by a height SDS of less than -1 .5, -2.0, -2.5, or -3.0, treatment of individuals who are heterozygous carriers of a deleterious mutation in NPR2 with neither parent having short stature is also contemplated. Further contemplated is treatment of individuals who are heterozygous for deleterious mutations in other growth plate genes with CNP to improve stature and/or enhance bone growth.
[0144] Exemplary mutations in NPR2 are disclosed in International Patent Publication WO 2021/055497, incorporated herein by reference.
[0145] NPPC’s role in skeletal growth is well documented (Hisado-Oliva et al., Genetics Medicine 20:91 -97, 2018). The NPPC knock out mouse showed severe disproportionate form of dwarfism including shortening of limbs and endochondral ossification (Hisado-Oliva et al., 2018, supra). Human genome wide studies have shown a link between NPPC and height (Hisado-Oliva et al., 2018, supra). Although CNP haploinsufficiency has been believed to be a cause of short stature in humans, a recent study identified heterozygous mutations in families with short stature and hands (Hisado-Oliva et al., 2018, supra). These studies observed significant reduction in cGMP production as measured in heterozygous state (Hisado-Oliva et al., 2018, supra). Mutations in NPPC include a 355G>T missense mutation causing a Gly119Cys change and a 349C>G missense mutation causing a Arg1 17Gly change. A CNP variant rescuing CGMP production may provide therapeutic benefit in the management of a disorder in patients having heterozygous loss-of-function NPPC mutations.
[0146] Leri-Weill dyschondrosteosis (LWD) is a rare genetic disorder characterized by shortening of the forearms and lower legs, abnormal misalignment of the wrist (Madelung deformity of the wrist), and associated short stature. LWD is caused by a heterozygous mutation in the short stature homeobox-containing (SHOX) gene or its regulatory elements located on the pseudoautosomal region 1 (PAR1 ) of the sex chromosomes. (See the Rare Disease Database and Carmona et al., Hum Mol Genet 20:1547-1559, 2011 ). The disorder Langer mesomelic dysplasia arises when there are two SHOX mutations, and may result from a mutation on each chromosome, either a homozygous or compound heterozygous mutations. A subset of SHOX mutations give rise to idiopathic short stature. Turner syndrome results due to a deletion on the X chromosome that can include the SHOX gene. SHOX has been identified as involved in the regulation of FGFR3 transcription and contributes to control of bone growth (Marchini et al., Endocr Rev. 37: 417-448, 2016). SHOX deficiency leads to increased FGFR3 signaling, and there is some evidence to support that SHOX has direct interactions with CNP/NPR2 as well (Marchini, supra). Given the association of SHOX with FGFR3 and bone growth, it is contemplated that a subject having a homozygous or heterozygous SHOX mutation would benefit from treatment with CNP variants as described herein.
[0147] RASopathies are a group of rare genetic conditions caused by mutations in genes of the Ras/mitogen-activated protein kinase (MAPK) pathway. RASopathies are a group of disorders characterized by increased signaling through RAS/MAPK pathway. This pathway leads to downstream activation of the RAF/MEK/ERK pathway. Short stature is a characteristic
feature of certain RASopathies. For example, CNP signaling inhibits RAF and leads to decreased MEK and ERK activation.
[0148] Treatment of RASopathies is contemplated herein. RASopathies associated with short stature include Noonan syndrome, Costello syndrome, Cardiofaciocutaneous syndrome, Neurofibromatosis Type 1 , and LEOPARD syndrome. Hereditary gingival fibromatosis type 1 is also a RASopathy contemplated herein. RASopathy patients (including Noonan syndrome, Costello syndrome, Cardiofaciocutaneous syndrome, Neurofibromatosis Type 1 , LEOPARD syndrome, hereditary gingival fibromatosis type 1) include patients with heterozygous variants in one or more of the following genes: BRAF, CBL, HRAS, KRAS, LZTR1 , MAP2K1 , MAP2K2, MRAS, NF1 , NRAS, PPP1 CB, PTPN11 , RAF1 , RRAS, RIT1 , SHOC2, SOS1 , or SOS2 (Tajan et al. Endocr. Rev. 2018;39(5):676-700).
[0149] CFC is caused by mutations in several genes in the Ras/MAPK signaling pathway, including K-Ras, B-Raf, Mek1 and Mek2. Costello syndrome, also called faciocutaneoskeletal (FCS) syndrome is caused by activating mutations in the H-Ras gene. Hereditary gingival fibromatosis type I (HGF) is caused by dominant mutations in the SOS1 gene (Son of Sevenless homolog 1 ), which encodes a guanine nucleotide exchange factor (SOS) that acts on the Ras subfamily of small GTPases. Neurofibromatosis type I (NF1 ) is caused by mutations in the neurofibromin 1 gene, which encodes a negative regulator of the Ras/MAPK signaling pathway. Noonan syndrome (NS) is caused by mutations in one of several genes, including PTPN11 , which encodes SHP2, and SOS1 , as well as K-Ras and Raf-1 .
[0150] CNP has been demonstrated to be an effective therapy in RASopathy models. Ono et al. generated mice deficient in Nf1 in type II collagen producing cells (Ono et aL, Hum. Mol. Genet. 2013;22(15):3048-62). These mice demonstrated constitutive ERK1/2 activation, and decreased chondrocyte proliferation, and maturation. Daily injections of CNP in these mice led to decreased ERK phosphorylation and corrected the short stature. A mouse model of Cardiofaciocutaneous syndrome using a Braf mutation (p.Q241 R) (Inoue et al. Hum. Mol.
Genet. 2019;28(1 ):74— 83). exhibited decreased body length and reduced growth plate width with smaller proliferative and hypertrophic zones compared to wild type, and CNP administration led to increases in body length in these animals.
[0151] Mutations in multiple genes can cause Noonan syndrome, which is characterized by short stature, heart defects, bleeding problems, and skeletal malformations. Mutations in the PTPN11 gene cause about half of all cases of Noonan’s syndrome. SOS1 gene mutations cause an additional 10 to 15 percent, and RAF1 and RIT1 genes each account for about 5
percent of cases. Mutations in other genes each account for a small number of cases. The cause of Noonan syndrome in 15 to 20 percent of people with this disorder is unknown.
[0152] The PTPN11 , SOS1 , RAF1 , and RIT 1 genes all encode for proteins that are important in the RAS/MAPK cell signaling pathway, which is needed for cell division and growth (proliferation), differentiation, and cell migration. Many of the mutations in the genes associated with Noonan syndrome cause the resulting protein to be turned on (active) and this prolonged activation alters normal RAS/MAPK signaling, which disrupts the regulation of cell growth and division, leading to the characteristic features of Noonan syndrome. See, e.g., Chen et aL, Proc Natl Acad Sci U S A. 1 11 (31 ):11473-8, 2014, Romano et al., Pediatrics. 126(4)746-59, 2010, and Milosavljevic et aL, Am J Med Genet 170(7):1874-80, 2016. It is contemplated that a subject having mutations that activate the MAPK pathway would benefit from treatment with CNP variants as described herein to improve bone growth and short stature. It is also contemplated that a subject having mutations that activate the MAPK pathway would benefit from treatment with CNP variants as described herein to improve other comorbidities associated with an overactive MAPK pathway in other cells throughout the body where the NPR2 receptor is expressed on its surface.
[0153] Mutations in the PTPN11 gene, which encodes the non-receptor protein tyrosine phosphatase SHP-2, lead to disorders characterized by short stature such as Noonan’s Syndrome (Musente et aL, Eur J Hum Genet 1 1 :201-206 (2003). Musente (supra) identifies numerous mutations in the PTPN11 gene that lead to short stature. Gain of function mutations lead to overactive signaling through SHP2 and inhibit Growth Hormone-induced IGF-1 release, thereby contributing to a decrease in bone growth (Rocca Serra-Nedelec, PNAS 109:4257- 4262, 2012). It is contemplated that a subject having a homozygous or heterozygous PTPN1 1 mutation would benefit from treatment with CNP variants as described herein to improve bone growth and short stature.
[0154] Mutations in the Indian hedgehog (IHH) gene, which is related to regulation of endochondral ossification, have also been associated with short stature syndromes (Vasques et aL, J Clin Endocrinol Metab. 103:604-614, 2018). Many IHH mutations identified segregate with short stature in a dominant inheritance pattern. Given the association of IHH with bone growth and ossification, it is contemplated that subjects having a homozygous or heterozygous IHH mutation will benefit from treatment with a CNP variant as described herein.
[0155] Mutations in FGFR3, including N540K and K650N, lead to short stature and hypochondroplasia.
[0156] Insulin-like growth factor 1 receptor (IGF1 R) is a heterotetrameric (a2|32) transmembrane glycoprotein with an intrinsic kinase activity. IGF1 R has been shown to have a role in prenatal and postnatal growth. Heterozygous mutations in IGF1 R have been identified in Small for gestational age children (SGA) and individuals with familial short stature (Kawashima et al., Endocrine J. 59:179-185, 2012). Mutations in IGF1 R associated with short stature include R108Q/K115N, R59T, R709Q, G1050K, R481 Q, V599E, and G1 125A (Kawashima, supra).
[0157] Osteogenesis imperfecta (Ol), also known as “osteogenesis congenita,” “osteogenesis imperfecta congenita” or “brittle bone disease”, is a heterogenous type 1 collagenopathy characterized by fragile bones. Osteogenesis imperfecta is found in approximately 1 :10000 to 1 :20000 individuals worldwide. Ol patients experience approximately 154 fractures per 1000 patient-years (Folkestad et al. J Bone Mineral Res. 31 : 2159-2166, 2016) and delayed healing was reported in -24% of fractures (Rauch, et al. J Bone Mineral Res. 24(7): 1282-89, 2009) Loss of function mutations in the COL1 A1 or COL1A2 genes lead to improper collagen formation, reduced levels of Type 1 collagen and result in the majority of cases of Ol. However, up to 20 different genes have been known to cause a form of Ol (van Dijk et al., Am J Hum Genet. 2020 107(5) :989-999). Type I (less severe) and Type II (most severe) Ol are diagnosed using the following criteria. Type I subjects exhibit bones likely to break from mild to moderate trauma, with most broken bones occurring before puberty; no change or only slight changes to stature with aging; loose joints and muscle weakness; blue, purple, or gray tint to sclera (whites of the eyes); triangular face; curved spine with potential for compression of the vertebrae (spine bones) with aging; mild or no bone deformity; possible changes to the strength and color of teeth; possible hearing loss; normal collagen structure, but less than the normal amount is produced. Type II Ol is characterized by death at birth or shortly after, because of the inability to breathe; numerous broken bones that develop before birth while the baby is still in the womb; severe bone deformities; very small stature; underdeveloped lungs; blue, purple, or gray tint to sclera; improperly formed collagen. Skeletal symptoms include bone fractures, deformity of vertebrae that may result in the impairment of pulmonary function and short stature. Additional common (>50% patients) extraskeletal symptoms include fragility of vessels, hearing loss and discoloration in sclera (Steiner et aL, GeneReviews,” COL1 A1/2 Osteogenesis Imperfecta”; Etich et al. Mol Cell Pediatr. 2020).
[0158] Intermediate severity Ol is classified as Type III, Type VI or Type V, having similar characteristics to Type I disease, including, but not limited to different severity of short stature, bone density, bone deformity, ease of broken bones and bone fragility, and loose joints. Type III
disease presents with moderate to severe bone fragility and more frequent fractures, extremely short stature, and more severe long bone deformity vs. Type I disease. Type VI- Type XXI 01 are often classified based on the mutation shown to cause the disease. Z-scores are typically lowers in females, and are decreased with age (Robinson, European Soc for Paediatric Res 2023). Short stature is most severe in Type III and IV 01, and individuals with Type 1 can have a SD below -2.5.
[0159] Bisphosphonates have been administered in order to decrease fracture incidence and are utilized to manage moderate to severe forms of Ol in children. Bisphosphonates include pamidronate, alendronate and risedronate. Bisphosphonates increase bone mineral density (BMD) and reduce biochemical markers of bone turnover, effects on preventing fractures are inconclusive (Raid et al., J Bone Mineral Res, 30:929-933, 2014; Liu et al., J Clin Endo Metab, 108:1787-179, 2023). Further, they do not improve bone quality and bone pain beyond 1 -year post-treatment (Liu et al., supra, Botor et aL, Biomolecules, 11 (10):1493, 2021). C-type natriuretic peptide (CNP) is produced in the growth plate and positively regulates linear bone growth. It is contemplated that administration of CNP variants or analogs will improve bone growth and reduce incidence and or severity of bone fracture in Ol patients.
[0160] Other therapies that have been attempted with Ol include anti-RANKL antibody, Human 1 -34 parathyroid hormone inhibitors, or sclerostin inhibitors.
[0161] In various embodiments, the subject is further administered adjunct therapy to treat osteogenesis imperfecta. In various embodiment, the adjunct therapy is alendronate, risedronate, ibandronate, zoledronic acid, denosumab, romosozumab, estrogen therapy or hormone therapy, raloxifene, bazedoxifene and estrogen, teriparatide, or abaloparatide. In various embodiments, the adjunct therapy is bisphosphonate therapy.
[0162] Trabecular bone tissue is spongy and porous, comprising both hard and soft tissue components, and is found at the epiphyses and metaphyses of long bones and in the vertebral bodies. Trabecular bone in Ol is characterized by a lower trabecular number and impaired connectivity as well as decreased trabecular thickness and volumetric bone mass (Nijhuis et aL, J Child Orthop. 2019 13(1 ): 1-11 )
[0163] It is contemplated that CNP therapy alleviates one or more symptoms of osteogenesis imperfecta, including but not limited to, femoral length, vertebral height, reduction in bone fracture frequency, cortical bone tissue mineral density (TMD), cortical bone mineral density (BMD), cortical bone thickness, cortical porosity, trabecular bone volume fraction (BVF),
trabecular TMD, trabecular BMD, trabecular bone number and trabecular bone separation. In various embodiments, CNP therapy improves annualized growth velocity (AGV) and final adult height in a subject receiving therapy, e.g., a subject with osteogenesis imperfecta.
[0164] It is reported herein that CNP also improves bone strength of subjects with achondroplasia that receive long term CNP therapy. Provided herein is a method of improving and/or maintaining bone strength in a subject in need thereof comprising administering a C-type natriuretic peptide (CNP) to the subject. In various embodiments, the CNP is administered daily, between 2 to 5 times per week or weekly. In various embodiments, the subject has a bone-related disorder, such as skeletal dysplasia or short stature. Non-limiting examples of CNP-responsive bone-related disorders skeletal dysplasias and short stature disorders include achondroplasia, hypochondroplasia, short stature, idiopathic short stature, dwarfism, osteochondrodysplasias, thanatophoric dysplasia, osteogenesis imperfecta, osteogenesis congenita, achondrogenesis, homozygous achondroplasia, chondrodysplasia congenita, campomelic dysplasia, congenital lethal hypophosphatasia, perinatal lethal type of osteogenesis congenita, short-rib polydactyly syndromes, rhizomelic type of chondrodysplasia congenita, Jansen-type metaphyseal dysplasia, spondyloepiphyseal dysplasia congenital, atelosteogenesis, diastrophic dysplasia, congenital short femur, Langer-type mesomelic dysplasia, Nievergelt-type mesomelic dysplasia, Robinow syndrome, Reinhardt syndrome, acrodysostosis, peripheral dysostosis, Kniest dysplasia, fibrochondrogenesis, Roberts syndrome, acromesomelic dysplasia, micromelia, Morquio syndrome, Kniest syndrome, metatrophic dysplasia, spondyloepimetaphyseal dysplasia, disorders related to NPR2 mutation, SHOX mutation (Turner’s syndrome/Leri Weill), PTPN1 1 mutations (Noonan’s syndrome), IGF1 R mutation, rickets, hypophosphatemic rickets [including X-linked hypophosphatemic rickets (also called vitamin D-resistant rickets) and autosomal dominant hypophosphatemic rickets], and osteomalacia [including tumor-induced osteomalacia (also called oncogenic osteomalacia or oncogenic hypophosphatemic osteomalacia.
[0165] Height is a highly heritable trait that can be influenced by the combined effect of hundreds or thousands of genes (Wood et al, 2014, Nature Genetics, 46:1173-1189). Short stature in an individual can be the result of the combined effect of these genes, without a single gene being the primary contributor. It is contemplated that such individuals with short stature defined by a height SDS of less than -1 .0, -1 .5, -2.0, -2.5, or -3.0, can be beneficially treated with a CNP variant given the ability of CNP to increase the length of normal animals, for example, enhance bone growth and length of bones.
[0166] In various embodiments, the CNP variants are useful to treat a subject having a height SDS of less than -1 .0, -1 .5, -2.0, -2.5, or -3.0, and having at least one parent with a height SDS of less than -1 .0, -1 .5, -2.0 or -2.5, optionally wherein the second parent has height within the normal range. In various embodiments, the CNP variants are useful to treat a subject having a height SDS of between -2.0 to -3.0. In various embodiments, the CNP variants are useful to treat a subject having a height SDS of between -2.0 to -2.5. In various embodiments, the short stature is associated with one or more mutations in a gene associated with short stature, such as, collagen (COL2A1 , COL11 A1 , COL9A2, COL10), aggrecan (ACAN), Indian hedgehog (IHH), PTPN1 1 , SHOX, NPR2, NPPC, FGFR3, or insulin growth factor 1 receptor (IGF1 R), DTL, PAPPA2, or combinations thereof, or associated with one or more mutations in a gene associated with a RASopathy.
[0167] In various embodiments the short stature is in a subject with osteogenesis imperfecta. In various embodiments, the Ol may result from a mutation in one or more of the following genes: COL1 A1 , COL1 A2, COL2A1 , COL1 1A1 , COL9A2, COL10, IFITM5, SERPINF1 , CRTAP, LEPRE1 , PPIB, SERPINH1 , FKBP10, SP7, BMP1 , TMEM38B, WNT1 , CREB3L1 , SPARC, FAM46A or combinations thereof.
[0168] In various embodiments, the short stature is a result of mutations in multiple genes as determined by polygenic risk score (PRS). Polygenic risk scores (PRS) are calculated for height using the largest published genome-wide association study (GWAS) meta-analysis for height that do not include any samples from the UK Biobank project as described in WO 2021/055497. The cohort may be divided into five PRS quintiles (PRS 1 being the lowest height, PRS 5 the tallest height). In various embodiments, the subject has a mutation in NPR2 and a low PRS. In various embodiments, the subject has a mutation in FGFR3 and a low PRS. In various embodiments, the subject has a mutation in NPR2 and a low PRS. In various embodiments, the subject has a mutation in IGF1 R and a low PRS. In various embodiments, the subject has a mutation in NPPC and a low PRS. In various embodiments, the subject has a mutation in SHOX and a low PRS. In various embodiments, the subject has one or more mutation in one or more of FGFR3, IGF1 R, NPPC, NPR2 and SHOX, and a low PRS. In various embodiments, the PRS is 1 or 2. In various embodiments, the PRS is 1 . In various embodiments, the PRS is 2.
[0169] In addition, the CNP variants are useful for treating other bone-related conditions and disorders, such as rickets, hypophosphatemic rickets [including X-linked hypophosphatemic rickets (also called vitamin D-resistant rickets) and autosomal dominant hypophosphatemic
rickets], and osteomalacia [including tumor-induced osteomalacia (also called oncogenic osteomalacia or oncogenic hypophosphatemic osteomalacia)].
[0170] Disclosed herein is a method of treating a subject having a bone-related disorder, skeletal dysplasia or short stature described herein comprising,
[0171] i) identifying whether a subject has a Loss of Function (LoF) or Gain of Function (GoF) variant of a gene related to short stature, such as osteogenesis imperfecta;
[0172] ii) calculating a polygenic risk score (PRS) of the subject;
[0173] iii) determining if the subject has a LoF variant and a PRS in the bottom 20%; and
[0174] iv) treating the subject with a CNP variant if the subject has a LoF variant and a PRS in the bottom 20%.
[0175] In various embodiments, the subject has a PRS in the bottom 20%, 19%, 18%, 17.5%, 17%, 16.5%, 16%, 15.5%, 15%, 14.5%, 14%, 13.5%, 13%, 12.5%, 12%, 11%, 10%, 9%, 8%, 7.5%, 7%, 6%, 5%, 4%, 3%, 2.5%, 2% or 1 %. In various embodiments, step iii) and iv) is a subject with a CNP variant if the subject has a LoF variant and a PRS in the bottom 12.5%.
[0176] The existence of a LoF or GoF variant in a gene related to short stature is determined by a biological activity assay. In various embodiments, a LoF or GoF variant may be predicted based on mapping to the predicted 3D structure and activity domain of a protein encoded by the gene, e.g., using AlphaForm 3D mapping or other protein mapping tools.
[0177] In an exemplary method, the PRS is calculated by a genome-wide association study (GWAS) of height.
[0178] In certain embodiments, the CNP variants and compositions and formulations comprising the same of the present disclosure are useful for improving one or more of the symptom(s) or physiological consequences of a skeletal dysplasia, wherein the improvement may be increased absolute growth, increased growth velocity, increased qualitative computed tomography (QCT) bone mineral density, improvement in growth plate morphology, increased long bone growth, improvement in spinal morphology, improved elbow joint range of motion and/or decreased sleep apnea. In this regard, it is noted that the terms "improved", "improvement", "increase", "decrease" and grammatical equivalents thereof are all relative terms that when used in relation to a symptom or physiological consequence of a disease state, refer to the state of the symptom or physiological consequence of the disease after treatment with a CNP variant (or composition or formulation comprising the same) of the present invention
as compared to the same symptom or physiological consequence of the disease before treatment with a CNP variant (or composition or formulation comprising the same) of the present invention (i.e., as compared to "baseline"). As described above, a "baseline" state can be determined either through measurement of the state in the subject prior to treatment (which can subsequently be compared to the state in the same subject after treatment), or through measurement of that state in a population of subjects suffering from the same affliction that share the same or similar characteristics (e.g., age, sex and/or disease state or progression).
[0179] Also provided is a method of overcoming cell growth arrest induced by a constitutively active mutant fibroblast growth factor receptor 3 (FGFR-3) comprising contacting a cell expressing the constitutively active FGFR-3 with a CNP variant or a composition as described herein.
[0180] In yet another embodiment, the disclosure provides CNP variants that in vitro or in vivo stimulate the production of at least about 50%, 60%, 70%, 80%, 90%, 100%, 110%, 120%, 130%, 140% or 150% of the cGMP level produced under the same concentration of wtCNP22 (e.g., 1 uM). In a still further embodiment, the CNP variants of the disclosure in vitro or in vivo stimulate the production of at least about 50%, 60%, 70%, 80%, 90%, 100%, 1 10%, 120%, 130%, 140% or 150% of the cGMP level produced under the same concentration of wtCNP22 (e.g., 1 uM).
[0181] The disclosure also contemplates that modulation of treatment with CNP as described herein enhances or increases growth in the range of 25%-50% change from baseline in the subject. In one embodiment, an enhancement or increase in growth velocity is an increase in annualized growth velocity of at least about 25%, more preferably at least about 40%, change from baseline in the subject. In one embodiment, an enhancement or increase in growth is an increase in bone growth of at least about 25%, more preferably at least about 40%, change from baseline in the subject.
[0182] It is contemplated that any of the CNP variants, including conjugates, salts or prodrugs thereof, described herein are useful in the methods.
[0183] In various embodiments, the CNP variant is PGQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (Pro-Gly-CNP-37) (SEQ ID NO: 1 ). In various embodiments, the peptide further comprises an acetyl group. In various embodiments, the acetyl group is on the N-terminus of the peptide. In various embodiments, the peptide further comprises an OH or an NH2 group at the C-terminus. In various
embodiments, the variant comprises one or more linker groups as described herein. In various embodiments, the linker is a hydrolysable linker, e.g., as described herein.
[0184] In various embodiments, the CNP variant is selected from the group consisting of PGQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (SEQ ID NO: 47);
PGQEHPQARRYRGAQRRGLSRGCFGLKLDRIGSMSGLGC (SEQ ID NO: 46); PGQEHPNARRYRGANRRGLSRGCFGLKLDRIGSMSGLGC (SEQ ID NO: 48); PGQEHPQARKYKGAQKKGLSKGCFGLKLDRIGSMSGLGC (SEQ ID NO: 49) QEHPNARKYKGANKKGLSKGCFGLKLDRIGSNSGLGC [CNP-37(M32N); SEQ ID NO: 41]; MQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (Met-CNP-37; SEQ ID NO: 43); PQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (Pro-CNP-37; SEQ ID NO: 42); GQEHPNARKYKGANKKGLSKGCFGLKLDRIGSNSGLGC [Gly-CNP-37 (M32N); SEQ ID NO:
44];
MGQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (Met-Gly-CNP-37; SEQ ID NO:
45);
GQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (Gly-CNP-37: SEQ ID NO: 2) GQEHPNARKYKGANPKGLSKGCFGLKLDRIGSMSGLGC (SEQ ID NO: 50);
GQEHPNARKYKGANQKGLSKGCFGLKLDRIGSMSGLGC (SEQ ID NO: 51 ); GQEHPNARKYKGANQQGLSKGCFGLKLDRIGSMSGLGC (SEQ ID NO: 52); GQEHPNARKYKGANKPGLSKGCFGLKLDRIGSMSGLGC (SEQ ID NO: 53); and LQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-38) (SEQ ID NO: 21 ), wherein the CNP variant comprises a conjugate moiety. In various embodiments, the conjugate moiety is a synthetic polymeric group.
[0185] In various embodiments, the variant comprises a synthetic polymeric group coupled to the variant through a hydrolysable linker. In various embodiments, the synthetic polymeric group comprises a hydrophilic polymer moiety. In various embodiments, the hydrophilic polymer moiety comprises polyethylene glycol (PEG). In various embodiments, the hydrophilic polymer moiety comprises polyethylene glycol (PEG) having a 6 to 20 atom chain length. In various embodiments, the conjugate moiety comprises one or more acid moieties linked to a hydrophilic spacer as described herein.
[0186] In various embodiments, the conjugate moiety comprises one or more acid moieties linked to a hydrophilic spacer. In various embodiments, the hydrophilic spacer is any amino acid. In various embodiments, the hydrophilic spacer is gamma glutamic acid (yGlu). In various embodiments, the hydrophilic spacer is OEG (8-amino-3,6-dioxaoctanoic acid). In various
embodiments, the hydrophilic spacer is gamma glutamic acid (yGlu) or OEG (8-amino-3,6- dioxaoctanoic acid). In various embodiments, the hydrophilic spacer is gamma glutamic acid (yGlu) linked to one or two or more OEG (8-amino-3,6-dioxaoctanoic acid). In various embodiments, the acid moiety is a fatty acid. Exemplary fatty acids include short chain, medium chain, or long chain fatty acids, or a dicarboxylic fatty acid. In various embodiments, the fatty acid is saturated or unsaturated. Contemplated are C-6 to C-20 fatty acids, including but not limited to, C-6, C-8, C-10, C-12, C-14, C-16, C-18 or C-20 fatty acids, saturated or unsaturated. In various embodiments, the fatty acid is decanoic acid, dodecanoic acid, myristic acid, palmitic acid, stearic acid, arachidic acid, or diacids of the same.
[0187] In various embodiments, the variant comprises one or more linker groups. In various embodiments, the linker is on a residue of the CNP cyclic domain or at a site other than the CNP cyclic domain. In various embodiments, the linker is on a lysine residue.
[0188] Efficacy of treatment is measured by various parameters. In various embodiments, efficacy is assessed as the change in annualized growth velocity from the baseline period to the intervention period. Efficacy will also be assessed as the change in height SDS from baseline to end of treatment as measured using the GDC growth curves, and growth velocity SDS will be based on the Bone Mineral Density in Childhood Study (Kelly et al., J. Clin. Endocrinol. Metab. 2014;99(6) :2104-2112). Annualized growth velocity, body proportionality and bone growth/length can be measured as described in Savarirayan et al., (New England J Med 2019 181 : 25-35), herein incorporated by reference.
[0189] Efficacy can also be measured using analysis of skull and brain morphology, e.g., using magnetic resonance imaging (MRI). At birth, children with achondroplasia have abnormalities of the base of the skull and boundaries of the foramen magnum due to defective endochondral ossification, resulting in stenosis of the foramen magnum and compression of the vital neural and vascular structures passing through it. Foramen magnum stenosis has been implicated as the major underlying cause of an increased risk for sudden deaths observed in children less than age 5 years with achondroplasia (Pauli et al., J Pediatr 1984;104:342-8; Hashmi et al., Am J Med Genet A 2018;176:2359-64). Skull and brain morphology analysis include measurement of improvements in facial volume, sinus volume, and foramen magnum area in patients, e.g., younger patients less than 6 months old, treated with CNP variants.
[0190] Provided herein is a method for increasing facial volume, facial sinus volume, and foramen magnum area in a subject 6 months old or less having a bone-related disorder, skeletal dysplasia or short stature comprising administering CNP variants, conjugates, salts or prodrugs
thereof at a dose of at least 30 pg/kg. Also provided is a method of decreasing the incidence of sudden infant death, sleep disordered breathing, and necessity for neurosurgical decompression of the foramen magnum in a subject 6 months old or less having a bone-related disorder, skeletal dysplasia or short stature comprising administering CNP variants, conjugates, salts or prodrugs thereof at a dose of at least 30 pg/kg. In various embodiments, the CNP variant is administered at a dose of 30 pg/kg for 3 months, 6 months, 1 year or more. In various embodiments, the dose of CNP variant is decreased to 15 pg/kg when the subject is about 2 years old.
[0191] Change in facial volume, facial sinus volume, and foramen magnum area are measured by magnetic resonance imaging (MRI), and can be compared to baseline levels, healthy control subjects or untreated control subjects.
[0192] QoLISSY, the Quality of Life in Short Stature Youth, is assessed as directed (Quality of Life in Short Stature Youth - The QoLISSY Questionnaire User’s Manual. Lengerich: Pabst Science Publishers; 2013).
Biomarkers
[0193] Biomarker refers to a detectable biological substance or moiety whose level is increased or decreased in association with a particular disease condition or treatment regimen. In the present disclosure, biomarkers may be measured before, during and/or after administration of a CNP variant as described herein. Exemplary bone- or cartilage-associated biomarkers include, but are not limited to, NTproCNP, N terminal fragment of collagen X (CXM), CNP, cGMP, propeptides of collagen type II and fragments thereof, collagen type II and fragments thereof, propeptides of collagen type I and fragments thereof, collagen type I and fragments thereof, osteocalcin, proliferating cell nuclear antigen (PCNA), aggrecan chondroitin sulfate, collagen X, and alkaline phosphatase. Markers of bone resorption (osteoclast activity) include cross-linked C-telopeptides of type II collagen (CTXII), urinary N-telopeptide (NTX) and tartrate resistant acid phosphatase (TRAP). Markers of bone formation (osteoblast activity) include bone-specific alkaline phosphatase (BSAP), osteocalcin (OC), N-terminal pro-peptide of type I procollagen (PINP) and procollagen type 1 C-terminal propeptide (P1 CP). Cartilage- and bone-associated biomarkers can be measured in any appropriate biological sample, including but not limited to tissues, blood, serum, plasma, cerebrospinal fluid, synovial fluid and urine. In some embodiments, the biomarkers are measured in blood, plasma or serum from animals undergoing efficacy/pharmacodynamic in vivo studies and/or from the conditioned media of ex vivo studies.
[0194] NTproCNP is an amino-terminal propeptide (NTproCNP) of CNP that is released from cells at an equimolar ratio with CNP. The biologically active forms of CNP are found in plasma in low concentrations due to the quick clearance rate of the peptide. NTproCNP is not cleared via the same mechanism and it is found in the circulation at 20- to 50-fold higher concentration (Olney et al., Clin Endocrinol (Oxf). 2012, 77:416-422).
[0195] It is contemplated that assessment of the effects of CNP therapy as described herein on bone growth are measured in relation to NTproCNP levels. For example, NTproCNP levels are measured in a sample and doses of CNP altered or changed to bring NTproCNP levels within +/- 2 SDS of the mean NTproCNP for the population. NTproCNP mean levels for different populations have been studied in the following publications, herein incorporated by reference: Olneyet al. (2015). C-type natriuretic peptide plasma levels are elevated in subjects with achondroplasia, hypochondroplasia, and thanatophoric dysplasia. J Clin Endocrinol Metab, 100(2), E355-359; Prickett et al., (2013). Impact of age, phenotype and cardio-renal function on plasma C-type and B-type natriuretic peptide forms in an adult population. Clin Endocrinol (Oxf), 78(5), 783-789; Espiner et al. (2018). Plasma C-Type Natriuretic Peptide: Emerging Applications in Disorders of Skeletal Growth. Norm Res Paediatr, 90(6), 345-357; Olney et al. (2012). Amino-terminal propeptide of C-type natriuretic peptide (NTproCNP) predicts height velocity in healthy children. Clin Endocrinol (Oxf), 77(3), 416-422; Olney et al., (2007). Aminoterminal propeptide of C-type natriuretic peptide and linear growth in children: effects of puberty, testosterone, and growth hormone. J Clin Endocrinol Metab, 92(11 ), 4294-4298; and Olney et al. (2016). Dynamic response of C-type natriuretic peptide and its aminoterminal propeptide (NTproCNP) to growth hormone treatment in children with short stature. Clin Endocrinol (Oxf), 85(4), 561 -568.
[0196] For example, Olney 2016 shows that children between 6 to 10 years old with idiopathic short stature can have an average baseline NTproSDS of -0.6, ranging from -1.0 to 0.7. Olney 2012 reported NTproCNP levels in healthy children/adolescents during stages of growth. NTproCNP SDS can be calculated based on the average NTproCNP levels of the different age populations, and therefore +/- 2 SDS from this mean can also be calculated. NTproCNP levels of subjects with achondroplasia or hypochondroplasia are described in Olney 2015, showing that children approximately 3 to 8 years old have an NTproSDS average of 1 .4, ranging from 0.4 to 1 .8, while hypochondroplasia subjects (age 6.6 to 11 ) have an average NTproCNP SDS of 1 .9, ranging from 1 .8 to 2.3. Methods for determining NTproSDS levels are described herein and in the publications above.
[0197] Collagen type X biomarker (CXM) is a degradation fragment of collagen type X, comprising intact trimeric noncollagenous 1 (NC1) domain of type X collagen. CXM is released by active growth plates and decreases in samples as subjects age. CXM levels have been correlated with growth velocity in children (Coghlan et al., Sci Transl Med 2017, 9(419):eaan4669).
[0198] Bone-specific alkaline phosphatase (BSAP or BAP) is a bone growth biomarker produced by osteoblasts and osteoclasts in growth plates and mineralized bone. Changes in BSAP may reflect growth plate activity, bone growth, and I or bone remodeling activity.
[0199] N-terminal pro-peptide of type I procollagen (PINP) is a potential pharmacodynamic bone growth biomarker, released during production of type I collagen. Changes in PINP may reflect changes in growth plate activity, bone growth, and/or bone remodeling.
[0200] Cross-linked C-telopeptides of type II collagen (CTXII) is a potential pharmacodynamic bone growth biomarker released during degradation of type II collagen. Changes in CTXII may reflect changes in growth plate activity, bone growth, bone remodeling, and/or articular cartilage remodeling.
Formulations
[0201] The disclosure provides pharmaceutical compositions, including modified release compositions, comprising a CNP variant described herein, and one or more pharmaceutically acceptable excipients, carriers and/or diluents. In certain embodiments, the compositions further comprise one or more other biologically active agents (e.g., inhibitors of proteases, receptor tyrosine kinases, and/or the clearance receptor NPR-C).
[0202] The disclosure provides for modified release compositions comprising a conjugate moiety as described herein. Modified-release compositions include those that deliver a drug with a delay after its administration (delayed-release dosage) or for a prolonged period of time (extended-release dosage). Various embodiments of a CNP peptide conjugate provided herein include modified-release compositions, such as extended release, sustained or controlled release, and delayed release. The term “extended release composition” refers to a composition formulated in a manner in order to make the active ingredient/drug available over an extended period of time following administration (US Pharmacopeia). Extended-release dosage include sustained-release (SR) or controlled-release (CR) forms in which. Sustained release maintains drug release over a sustained period but not necessarily at a constant rate, while CR maintains drug release over a sustained period at a nearly constant rate (Pharmaceutics: Drug Delivery and
Targeting, Yvonne Perrie, Thomas Rades, Pharmaceutical Press, 2009). Delayed-release compositions or products are modified to delay release of the drug substance for some period of time after initial administration.
[0203] In various embodiments, the modified release composition is an extended release composition. In various embodiments, the modified release composition is a sustained release composition. In various embodiments the sustained or extended release compositions comprises a CNP pro-drug.
[0204] In various embodiments, the composition comprises an excipient, diluent or carrier. In various embodiments, the extended release composition comprises an excipient, diluent or carrier. In various embodiments, the excipient, diluent or carrier is a pharmaceutically acceptable excipient, diluent or carrier.
[0205] Non-limiting examples of excipients, carriers and diluents include vehicles, liquids, buffers, isotonicity agents, additives, stabilizers, preservatives, solubilizers, surfactants, emulsifiers, wetting agents, adjuvants, and so on. The compositions can contain liquids (e.g., water, ethanol); diluents of various buffer content (e.g., Tris-HCI, phosphate, acetate buffers, citrate buffers), pH and ionic strength; detergents and solubilizing agents (e.g., Polysorbate 20, Polysorbate 80); anti-oxidants (e.g., methionine, ascorbic acid, sodium metabisulfite); preservatives (e.g., Thimerosol, benzyl alcohol, m-cresol); and bulking substances (e.g., lactose, mannitol, sucrose). The use of excipients, diluents and carriers in the formulation of pharmaceutical compositions is known in the art; see, e.g., Remington's Pharmaceutical Sciences, 18th Edition, pages 1435-1712, Mack Publishing Co. (Easton, Pennsylvania (1990)), which is incorporated herein by reference in its entirety.
[0206] For example, carriers include without limitation diluents, vehicles and adjuvants, as well as implant carriers, and inert, non-toxic solid or liquid fillers and encapsulating materials that do not react with the active ingredient(s). Non-limiting examples of carriers include phosphate buffered saline, physiological saline, water, and emulsions (e.g., oil/water emulsions). A carrier can be a solvent or dispersing medium containing, e.g., ethanol, a polyol (e.g., glycerol, propylene glycol, liquid polyethylene glycol, and the like), a vegetable oil, and mixtures thereof.
[0207] In some embodiments, the compositions are liquid formulations. In certain embodiments, the formulations comprise a CNP variant in a concentration range from about 0.1 mg/ml to about 20 mg/ml, or from about 0.5 mg/ml to about 20 mg/ml, or from about 1 mg/ml to
about 20 mg/ml, or from about 0.1 mg/ml to about 10 mg/ml, or from about 0.5 mg/ml to about 10 mg/ml, or from about 0.5 to 5 mg/ml, or from about 0.5 to 3 mg/ml, or from about 1 mg/ml to about 10 mg/ml. In various embodiments, the CNP variant is in a concentration of 0.8 mg/ml to 2 mg/ml. In various embodiments, the CNP variant is at a concentration of 0.8 mg/ml. In various embodiments, the CNP variant is at a concentration of 2.0 mg/ml. In various embodiments, the CNP variant is reconstituted from a lyophilized powder.
[0208] In further embodiments, the compositions comprise a buffer solution or buffering agent to maintain the pH of a CNP-containing solution or suspension within a desired range. Nonlimiting examples of buffer solutions include phosphate buffered saline, Tris buffered saline, and Hank's buffered saline. Buffering agents include without limitation sodium acetate, sodium phosphate, and sodium citrate. Mixtures of buffering agents can also be used. In certain embodiments, the buffering agent is acetic acid/acetate or citric acid/citrate. The amount of buffering agent suitable in a composition depends in part on the particular buffer used and the desired pH of the solution or suspension. In some embodiments, the buffering agent has a concentration of about 10 mM ± 5 mM. In certain embodiments, the pH of a composition is from about pH 3 to about pH 9, or from about pH 3 to about pH 7.5, or from about pH 3.5 to about pH 7, or from about pH 3.5 to about pH 6.5, or from about pH 4 to about pH 6, or from about pH 4 to about pH 5, or is at about pH 5.0 ± 1 .0. In various embodiments, the pH is about 5.0, 5.1 , 5.2, 5.3, 5.4, 5.5, 5.6, 5.7, 5.8, 5.9 or 6.0. In various embodiments, the pH is 5.5.
[0209] In other embodiments, the compositions contain an isotonicity-adjusting agent to render the solution or suspension isotonic and more compatible for administration. Non-limiting examples of isotonicity agents include NaCI, dextrose, glucose, glycerin, sorbitol, xylitol, and ethanol. In certain embodiments, the isotonicity agent is NaCI. In certain embodiments, NaCI is in a concentration of about 160 ± 20 mM, or about 140 mM ± 20 mM, or about 120 ± 20 mM, or about 100 mM ± 20 mM, or about 80 mM ± 20 mM, or about 60 mM ± 20 mM.
[0210] In yet other embodiments, the compositions comprise a preservative. Preservatives include, but are not limited to, m-cresol and benzyl alcohol. In certain embodiments, the preservative is in a concentration of about 0.4% ± 0.2%, or about 1% ± 0.5%, or about 1 .5% ± 0.5%, or about 2.0% ± 0.5%.
[0211] In still other embodiments, the compositions contain an anti-adsorbent (e.g., to mitigate adsorption of a CNP variant to glass or plastic). Anti-adsorbents include without limitation benzyl alcohol, Polysorbate 20, and Polysorbate 80. In certain embodiments, the antiadsorbent is in a concentration from about 0.001% to about 0.5%, or from about 0.01% to about
0.5%, or from about 0.1% to about 1%, or from about 0.5% to about 1 %, or from about 0.5% to about 1 .5%, or from about 0.5% to about 2%, or from about 1% to about 2%.
[0212] In additional embodiments, the compositions comprise a stabilizer. Non-limiting examples of stabilizers include glycerin, glycerol, thioglycerol, methionine, and ascorbic acid and salts thereof. In some embodiments, when the stabilizer is thioglycerol or ascorbic acid or a salt thereof, the stabilizer is in a concentration from about 0.1% to about 1%. In other embodiments, when the stabilizer is methionine, the stabilizer is in a concentration from about 0.01% to about 0.5%, or from about 0.01 % to about 0.2%. In still other embodiments, when the stabilizer is glycerin, the stabilizer is in a concentration from about 5% to about 100% (neat).
[0213] In further embodiments, the compositions contain an antioxidant. Exemplary antioxidants include without limitation methionine and ascorbic acid. In certain embodiments, the molar ratio of antioxidant to CNP is from about 0.1 :1 to about 15:1 , or from about 1 :1 to about 15:1 , or from about 0.5:1 to about 10:1 , or from about 1 :1 to about 10:1 or from about 3:1 to about 10:1 .
[0214] Pharmaceutically acceptable salts can be used in the compositions, including without limitation mineral acid salts (e.g., hydrochloride, hydrobromide, phosphate, sulfate), salts of organic acids (e.g., acetate, propionate, malonate, benzoate, mesylate, tosylate), and salts of amines (e.g., isopropylamine, trimethylamine, dicyclohexylamine, diethanolamine). A thorough discussion of pharmaceutically acceptable salts is found in Remington's Pharmaceutical Sciences, 18th Edition, Mack Publishing Company, (Easton, Pennsylvania (1990)).
[0215] The pharmaceutical compositions can be administered in various forms, such as tablets, capsules, granules, powders, solutions, suspensions, emulsions, ointments, and transdermal patches. The dosage forms of the compositions can be tailored to the desired mode of administration of the compositions. For oral administration, the compositions can take the form of, e.g., a tablet or capsule (including softgel capsule), or can be, e.g., an aqueous or nonaqueous solution, suspension or syrup. Tablets and capsules for oral administration can include one or more commonly used excipients, diluents and carriers, such as mannitol, lactose, glucose, sucrose, starch, corn starch, sodium saccharin, talc, cellulose, magnesium carbonate, and lubricating agents (e.g., magnesium stearate, sodium stearyl fumarate). If desired, flavoring, coloring and/or sweetening agents can be added to the solid and liquid formulations. Other optional ingredients for oral formulations include without limitation preservatives, suspending agents, and thickening agents. Oral formulations can also have an enteric coating to protect the CNP variant from the acidic environment of the stomach. Methods of preparing
solid and liquid dosage forms are known, or will be apparent, to those skilled in this art (see, e.g., Remington's Pharmaceutical Sciences, referenced above).
[0216] Formulations for parenteral administration can be prepared, e.g., as liquid solutions or suspensions, as solid forms suitable for solubilization or suspension in a liquid medium prior to injection, or as emulsions. For example, sterile injectable solutions and suspensions can be formulated according to techniques known in the art using suitable diluents, carriers, solvents (e.g., buffered aqueous solution, Ringer's solution, isotonic sodium chloride solution), dispersing agents, wetting agents, emulsifying agents, suspending agents, and the like. In addition, sterile fixed oils, fatty esters, polyols and/or other inactive ingredients can be used. As further examples, formulations for parenteral administration include aqueous sterile injectable solutions, which can contain antioxidants, buffers, bacteriostats, and solutes that render the formulation isotonic with the blood of the intended recipient; and aqueous and nonaqueous sterile suspensions, which can contain suspending agents and thickening agents.
[0217] Exemplary CNP formulations are described in U.S. Patents 9,907,834 and 10,646,550. Use of CNP formulations having a pH in the range from about 4 to about 6 is contemplated.
[0218] Compositions comprising a CNP variant can also be lyophilized formulations. In certain embodiments, the lyophilized formulations comprise a buffer and bulking agent, and optionally an antioxidant. Exemplary buffers include without limitation acetate buffers and citrate buffers. Exemplary bulking agents include without limitation mannitol, sucrose, dextran, lactose, trehalose, and povidone (PVP K24). In certain embodiments, mannitol is in an amount from about 3% to about 10%, or from about 4% to about 8%, or from about 4% to about 6%. In certain embodiments, sucrose is in an amount from about 6% to about 20%, or from about 6% to about 15%, or from about 8% to about 12%. Exemplary anti-oxidants include, but are not limited to, methionine and ascorbic acid.
[0219] In various embodiments, the formulation comprises citric acid, sodium citrate, trehalose, mannitol, methionine, polysorbate 80, and optionally sterile water for injection (WFI).
[0220] The disclosure also provides kits containing, e.g., bottles, vials, ampoules, tubes, cartridges and/or syringes that comprise a liquid (e.g., sterile injectable) formulation or a solid (e.g., lyophilized) formulation. The kits can also contain pharmaceutically acceptable vehicles or carriers (e.g., solvents, solutions and/or buffers) for reconstituting a solid (e.g., lyophilized) formulation into a solution or suspension for administration (e.g., by injection), including without
limitation reconstituting a lyophilized formulation in a syringe for injection or for diluting concentrate to a lower concentration. Furthermore, extemporaneous injection solutions and suspensions can be prepared from, e.g., sterile powder, granules, or tablets comprising a CNP- containing composition. The kits can also include dispensing devices, such as aerosol or injection dispensing devices, pen injectors, autoinjectors, needleless injectors, syringes, and/or needles.
[0221] As a non-limiting example, a kit can include syringes having a single chamber or dual chambers. For single-chamber syringes, the single chamber can contain a liquid CNP formulation ready for injection, or a solid (e.g., lyophilized) CNP formulation or a liquid formulation of a CNP variant in a relatively small amount of a suitable solvent system (e.g., glycerin) that can be reconstituted into a solution or suspension for injection. For dual-chamber syringes, one chamber can contain a pharmaceutically acceptable vehicle or carrier (e.g., solvent system, solution or buffer), and the other chamber can contain a solid (e.g., lyophilized) CNP formulation or a liquid formulation of a CNP variant in a relatively small amount of a suitable solvent system (e.g., glycerin) which can be reconstituted into a solution or suspension, using the vehicle or carrier from the first chamber, for injection.
[0222] As a further example, a kit can include one or more pen injector or autoinjector devices, and dual-chamber cartridges. One chamber of a cartridge can contain a pharmaceutically acceptable vehicle or carrier (e.g., solvent system, solution or buffer), and the other chamber can contain a solid (e.g., lyophilized) CNP formulation or a liquid formulation of a CNP variant in a relatively small amount of a suitable solvent system (e.g., glycerin) which can be reconstituted into a solution or suspension, using the vehicle or carrier from the first chamber, for injection. A cartridge can comprise an amount of the CNP variant that is sufficient for dosing over a desired time period (e.g., 1 day, 2 days, 3 days, 1 week, 2 weeks, 3 weeks, 4 weeks, etc.). The pen injector or autoinjector can be adjusted to administer a desired amount of the CNP formulation from a cartridge.
[0223] In addition, pharmaceutical compositions comprising a CNP variant can be formulated as a slow release, controlled release or sustained release system for maintaining a relatively constant level of dosage over a desired time period, such as 1 week, 2 weeks, 3 weeks, 1 month, 2 months, or 3 months. Slow release, controlled release and sustained release formulations can be prepared using, e.g., biodegradable polymeric systems {which can comprise, e.g., hydrophilic polymers [e. ., polylactide, polyglycolide, poly(lactide-glycolide)]}, and can take the form of, e.g., microparticles, microspheres or liposomes, as is known in the art.
Administration and Dosing
[0224] As used herein, the term "therapeutically effective amount" of an active agent (e.g., a CNP variant) refers to an amount that provides therapeutic benefit to a patient. The amount may vary from one individual to another and may depend upon a number of factors, including the overall physical condition of the patient. A therapeutically effective amount of a CNP variant can be readily ascertained by one skilled in the art, using publicly available materials and procedures. For example, the amount of a CNP variant used for therapy should give an acceptable rate of reversal of cartilage degeneration or increase in cartilage growth.
[0225] The dosing frequency for a particular individual may vary depending upon various factors, including the disorder being treated and the condition and response of the individual to the therapy. In certain embodiments, a pharmaceutical composition containing a CNP variant is administered to a subject about one time per day, one time per two days, one time per three days, or one time per week, twice per week, three times per week, once every two weeks, or monthly.
[0226] The CNP variant compositions described herein can be administered to patients in need thereof at therapeutically effective doses to treat, ameliorate or prevent bone-related disorders and short stature disorders (e.g., skeletal dysplasias, including achondroplasia, hypochondroplasia, osteogenesis imperfecta, etc.). The CNP variants contemplated for use herein can be administered to patients at therapeutically effective doses to treat, ameliorate or prevent osteoarthritis and other conditions having an osteoarthritis-associated symptom. The safety and therapeutic efficacy of the CNP variants can be determined by standard pharmacological procedures in cell cultures or experimental animals, such as, for example, by determining the LD50 (the dose lethal to 50% of the population) and the ED50 (the dose therapeutically effective in 50% of the population). The dose ratio between toxic and therapeutic effects is the therapeutic index and it can be expressed as the ratio LD50 /ED50. Active agents exhibiting a large therapeutic index are normally preferred.
[0227] In certain embodiments, provided is a method of treating osteogenesis imperfecta comprising administering a C-type natriuretic peptide (CNP) to a subject in need thereof, wherein the CNP is administered less frequently than daily administration. For example, the CNP is administered 5 times weekly, 4 times weekly, 3 times weekly, 2 times weekly, weekly, bi-weekly, monthly or less frequently
[0228] In certain embodiments, provided is a method of treating osteogenesis imperfecta comprising administering a C-type natriuretic peptide (CNP) to a subject in need thereof, wherein the CNP is administered 5 times weekly, 4 times weekly, 3 times weekly, weekly or biweekly.
[0229] Provided is a method of reducing the frequency of bone fracture in a subject having osteogenesis imperfecta (Ol). improving bone mineral density in a subject having osteogenesis imperfecta (Ol) and/or improving femoral length or vertebral growth in a subject having osteogenesis imperfecta (Ol) comprising administering a C-type natriuretic peptide (CNP) to a subject in need thereof, wherein the CNP is administered 5 times weekly, 4 times weekly, 3 times weekly, weekly or biweekly.
[0230] In certain embodiments, the CNP variant compositions described herein are administered at a dose in the range from about 3, 4, 5, 6, 7, 8, 9 or 10 nmol/kg to about 300 nmol/kg, or from about 20 nmol/kg to about 200 nmol/kg. In some embodiments, the CNP compositions are administered at a dose of about 3, 4, 5, 6, 7, 8, 9, 10, 15, 20, 25, 30, 35, 40, 45, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 110, 120, 125, 130, 140, 150, 160, 170, 175, 180, 190, 200, 210, 220, 230, 240, 250, 260, 270, 280, 290, 300, 350, 400, 450, 500, 750, 1000, 1250, 1500, 1750 or 2000 nmol/kg or other dose deemed appropriate by the treating physician. In other embodiments, the CNP variant compositions are administered at a dose of about 0.5, 1 , 2, 3, 4, 5, 6, 7, 8, 9, 10, 15, 20, 25, 30, 40, 50, 60, 70, 80, 90, 100, 110, 120, 130, 140, 150, 160, 170, 180, 190, 200, 250, 300, 350, 400, 450, 500, 550, 600, 650, 700, 750, 800, 850, 900, 950 or 1000 pg/kg, or about 0.5, 0.8, 1 .0, 1 .25, 1 .5, 2, 2.5, 3, 3.5, 4, 4.5, 5, 5.5, 6, 6.5, 7, 7.5, 8, 8.5, 9, 9.5 or 10 mg/kg, or other dose deemed appropriate by the treating physician. The doses of CNP or CNP variant described herein can be administered according to the dosing frequency/frequency of administration described herein, including without limitation daily, 2 or 3 times per week, 5 days a week, weekly, every 2 weeks, every 3 weeks, monthly, etc. In various embodiments, the CNP or CNP variant is administered daily subcutaneously. In various embodiments, the CNP or CNP variant is administered weekly subcutaneously. In various embodiments, the CNP variant is administered at a dose of 0.5 pg/kg/day to 60 pg/kg/day, 1 .5 pg/kg/day to 30 pg/kg/day, 2.5 pg/kg/day to 15 pg/kg/day, 10pg/kg/day to 45 pg/kg/day, or 15pg/kg/day to 30 pg/kg/day. In various embodiments, the CNP variant is administered at a dose of 15 pg/kg/day. In various embodiments, the CNP variant is administered at a dose of 30 pg/kg/day. In various embodiments, the CNP variant is administered 20 pg/kg 5 days per week, 20 pg/kg 3 days per week, or 10 pg/kg 3 days per week.
[0231 ] The frequency of dosing/administration of a CNP variant for a particular subject may vary depending upon various factors, including the disorder being treated and the condition and response of the subject to the therapy. The CNP variant can be administered in a single dose or in multiple doses per dosing. In certain embodiments, the CNP variant composition is administered, in a single dose or in multiple doses, once daily, once weekly, once every two weeks, once every three weeks, once every 4 weeks, once every 6 weeks, once every two months, once every three months or once every six months, or as deemed appropriate by the treating physician. In various embodiments, the CNP variant is administered for 3 month, 6 months, 12 months or more.
[0232] In some embodiments, a CNP variant composition is administered to a subject with osteogenesis imperfecta so as to allow for periods of growth (e.g., chondrogenesis), followed by a recovery period (e.g., osteogenesis). For example, the CNP composition may be administered subcutaneously or by another mode daily, multiple times per week, or weekly for a period of time, followed by a period of no treatment, then the cycle is repeated. In some embodiments, the initial period of treatment (e.g., administration of the CNP variant composition daily, multiple times per week or weekly) is for 3 days, 1 week, 2 weeks, 3 weeks, 4 weeks, 5 weeks, 6 weeks, 7 weeks, 8 weeks, 9 weeks, 10 weeks, 11 weeks or 12 weeks. In a related embodiment, the period of no treatment lasts for 3 days, 1 week, 2 weeks, 3 weeks or 4 weeks. In certain embodiments, the dosing regimen of the CNP variant compositions is daily for 3 days followed by 3 days off; or daily or multiple times per week for 1 week followed by 3 days or 1 week off; or daily or multiple times per week for 2 weeks followed by 1 or 2 weeks off; or daily, multiple times per week or weekly for 3 weeks followed by 1 , 2 or 3 weeks off; or daily, multiple times per week or weekly for 4, 5, 6, 7, 8, 9, 10, 11 or 12 weeks followed by 1 , 2, 3 or 4 weeks off. It is contemplated that a subject having osteogenesis imperfecta could still be receiving an adjunct therapy in periods when CNP is not being administered, or could be receiving no other treatment in periods when CNP is not being administered.
[0233] The CNP variants, or pharmaceutical compositions comprising them, can be administered to subjects in various ways such as, e.g., by injection subcutaneously, intraarticularly, intravenously, intra-arterially, intraperitoneally, intramuscularly, intradermally, or intrathecally, or by oral administration. In one embodiment, the CNP variants are administered by a single subcutaneous, intraarticular, intravenous, intra-arterial, intraperitoneal, intramuscular, intradermal, or intrathecal injection.
[0234] The CNP variants can be administered by implantation of a depot at the target site of action (e.g., an abnormal or degenerated joint or cartilage area). Alternatively, the CNP variants can be administered sublingually under the tongue (e.g., sublingual tablet) or by inhalation into the lungs (e.g., inhaler or aerosol spray), by delivery into the nasal cavity (e.g., intranasal spray), by delivery into the eye (e.g., eye drop), or by transdermal delivery (e.g., by means of a patch on the skin). The CNP variants may also be administered orally in the form of microspheres, microcapsules, liposomes (uncharged or charged (e.g., cationic)), polymeric microparticles (e.g., polyamides, polylactide, polyglycolide, poly(lactide-glycolide)), microemulsions, and the like.
[0235] A further method of administration is by osmotic pump (e.g., an Alzet pump) or minipump (e.g., an Alzet mini-osmotic pump), which allows for controlled, continuous and/or slow- release delivery of the CNP variant or pharmaceutical composition over a pre-determined period. The osmotic pump or mini-pump can be implanted subcutaneously, or near the target site (e.g., the long bones of limbs, the epiphyses, etc.).
[0236] It will be apparent to one skilled in the art that the CNP variants or compositions thereof can also be administered by other modes. Determination of the most effective mode of administration of the CNP variants or compositions thereof is within the skill of the skilled artisan.
[0237] The CNP variants can be administered as pharmaceutical formulations suitable for, e.g., oral (including buccal and sub-lingual), rectal, nasal, topical, pulmonary, vaginal or parenteral (including intramuscular, intraarterial, intrathecal, subcutaneous, intraarticularly and intravenous) administration, or in a form suitable for administration by inhalation or insufflation. Depending on the intended mode of administration, the pharmaceutical formulations can be in the form of solid, semi-solid or liquid dosage forms, such as tablets, suppositories, pills, capsules, powders, liquids, suspensions, emulsions, creams, ointments, lotions, and the like. The formulations can be provided in unit dosage form suitable for single administration of a precise dosage. The formulations comprise an effective amount of a CNP variant, and one or more pharmaceutically acceptable excipients, carriers and/or diluents, and optionally one or more other biologically active agents.
[0238] Additional aspects and details of the disclosure will be apparent from the following examples, which are intended to be illustrative rather than limiting.
EXAMPLES
Example 1-CNP Variants to Treat Osteogenesis Imperfecta
[0239] The present Example evaluated treatment of young growing oim/oim mice, a model of moderate- to- severe osteogenesis imperfecta (Ol), with a CNP variant and the bisphosphonate alendronate (ALN) as measured by reduced fracture incidence, improved growth, increased bone mineral density (BMD), and improved bone strength.
[0240] Mice are treated from 2 -14 weeks of age, for 12 weeks total. Bone quality is evaluated by radiographic analysis of fracture number, histological analyses, micro-CT, FTIR analysis of bone quality and mechanical testing of long bones. Growth is evaluated by weekly weighing and evaluation of long bones radiographically and by micro-CT. Further, muscle mass is assessed by micro CT.
[0241] In an initial study, NTproCNP and CNP levels have been measured and plasma studied plasma in patients with Ol. An interesting pattern has emerged, showing that individuals with the most severe form of the condition have therefore shortest stature and lowest growth velocities (Figure 1 ). Corresponding with this data are NTproCNP levels which appear to follow the same pattern with the lowest levels seen in the most severely affected patients (Figure 2). This observation suggests to us that the poor linear growth in individuals with moderate and severe Ol, may be due to growth plate dysfunction and decreased CNP production.
[0242] The oim/oim mouse is an established model of moderate to severe Ol that contains a naturally occurring mutation leading to deficiency of proa2(l) collagen. These mice are characterized by frequent fractures, small size, osteopenia and bone deformities (Chipman et al Proc Nat Acad Sci USA 1993, 90:1701 -1705). Heterozygous oim/+ mice have osteopenia but typically normal longitudinal growth and no spontaneous fractures, and have been used as a model of mild Ol (King, et al., Bone & Mineral Research 2005, 20, 987-993; McBride et al. Calcif Tissue Int 1998, 62, 172-176.). Homozygous oim/oim mice have been used in a number of studies to evaluate the effect of bisphosphonates (Camacho, et al., Calcified Tissue International 2001 , 69, 94-101 ; Evans et al., Bone 2003, 32, 268-274; McCarthy et al., Ped Res 2002, 52, 660-670.) and RANKL inhibition in Ol (Delos et al., Journal of Orthopaedic Research 2008, 26, 153-164; Bargman et al., Osteoporos Int 2012, 23, 1141 -1150; Bargman et al., Connect Tissue Res 2010, 51 , 123-131 ) 8-10. Alendronate (ALN), a bisphosphonate, has been shown to increase BMD, alter geometric and biomechanical properties of oim/oim bone and reduce fractures in these mice whereas RANKL inhibition was also found to increase BMD and
alter geometric and biomechanical properties but not to reduce fracture incidence, when treatment started at 6 weeks of age (Bargman 2010, supra).
[0243] Initiation of treatment at two weeks of age with either RANK-Fc or alendronate produced equivalent increases in BMD and equivalent reduction in fracture numbers9 Alendronate treatment has not been shown to increase growth in oim/oim mouse model studies Based on these recent studies to evaluate therapeutics, the use of the oim/oim mouse model will allow evaluation of the effect of the CNP analog on growth, fracture reduction and bone quality in Ol (Bargman 2012, supra).
[0244] Treatment: Oim/oim mice are randomized for treatment with either saline as a control, CNP, CNP plus alendronate from 2 weeks to 14 weeks of age, additionally a dose range of CNP is analyzed. The ALN dose has been established in previous studies described above, and the number per group has been determined based on a power analysis from our previous studies with biomechanical changes as the main outcome. The CNP variant used in this study is an analog of Pro-Gly-CNP37 and differs by one less amino acid at its N-terminus. CNP-analog is administered at three dosage regimens given subcutaneously (20 ug/kg 5 days per week (20x5), 20 ug/kg 3 days per week (20x3), or 10 ug/kg 3 days per week (10x3)). The ALN is injected once per week at a dose of 0.21 mg/kg/week, and saline in an equal volume of 0.1 mL/10 g body weight as previously described (Bargman 2012, supra). Mice are also injected with calcein (15mg/kg) at the start and end of their treatments, and with alizarin red (50 mg/kg) at the midpoint, to evaluate bone growth. At 2 weeks of age, the mother will be placed in a separate cage temporarily while the injections are being given to the pups. In addition, oim/oim and WT mice will be faxitroned at 8 weeks of age for dose monitoring as well as at 14 weeks of age to determine fracture number at this age.
[0245] Growth of mice: Mice will be weighed on a digital scale prior to administration of drug. Total body length is assessed tail to nose. To obtain information on femur growth, at sacrifice individual femora are measured in the AP and ML planes from digitized radiographs, and femur length is determined as the distance from the tip of the femoral head to the base of the condyles. Micro-CT is used to show femoral length.
[0246] Radiographic Analyses -Fracture Counts. Whole body digital radiographs are taken at sacrifice in the AP and ML planes. The AP radiographs are used for fracture counting. Fractures are counted in a blind fashion such that it will not be known if the particular mouse was treated or a control. Fractures are defined by any evidence of callus formation or obvious bone
deformation. The humeri, femora, tibiae, and first 5 vertebrae of the tail of the mice are examined, and the total number of fractures per mouse obtained.
[0247] Faxitron ULTRAFOCUS® Imaging (New York, NY, USA) was used to radiograph mice in the anterior-posterior and medial-lateral plane at 2 weeks (pre-treatment) for baseline images, 8 weeks (mid-treatment) for safety evaluation, and 14 weeks (post-treatment) for final treatment effect analysis. Images were produced at 46kV and 0.3mA. Two-week-old mice were anesthetized with 3% isoflurane and monitored throughout imaging. 14-week-old mice were imaged post-mortem. An aluminum density gradient was used to standardize length and intensity.
[0248] The Faxitron radiographs were used to assess a variety of potential treatment effects, the first of which was fracture number and healing assessment. Two blinded investigators (CLR and ED) determined the number of fractures in the humeri, radii, femurs, tibiae, and first five proximal vertebrae of the tail. Fractures were defined as the observation of a fracture line, callus formation, or bone deformation. Radiographs taken at 2 weeks and 14 weeks were directly compared to assess fracture healing throughout the course of treatment.
[0249] Femoral length was measured using Faxitron radiographs of the individual femurs post-sacrifice in the anterior-posterior plane, produced at 30kV and 0.3 mA. In the AP plane, femoral length was defined as the distance from the base of the condyles to the tip of the femoral head. Full body post-sacrifice radiographs taken in the AP plane were used to measure vertebral height, which was defined as the distance from the distal endplate of L6 to the proximal endplate of L4. An aluminum density standard was used in the measurements of these individual femurs and full-body AP radiographs.
[0250] Micro CT Bone Architecture'. Analyses of femurs on high-resolution cone-beam micro- CT scanner (SkyScan 1172; Broker microCT, Kontich, Belgium) is performed. Each scan is referenced to a phantom containing air, saline and a bone reference material (1.18 g/cm3) for calibration of Houndsfield units to mineral density in g/cm3. Reconstruction of the individual projections to computed tomography volume data and analyses of the reconstructed scans are performed using instrument software. Specimen-specific thresholds are determined by first selecting a volume of interest, generating the attenuation histogram, and determining the threshold that segments mineralized tissue from background. Properties determined include bone mineral content, bone volume fraction, bone surface area, apparent density, tissue density, trabecular thickness, trabecular separation, trabecular number, and polar moment of
inertia. Cortical bone was analyzed at the mid-diaphysis and trabecular bone was analyzed at the metaphysis.
[0251] Micro CT Muscle Mass: An index of muscle mass is determined after sacrifice by micro CT assessment as recently described (Pasetto et al., PLoS One 2018, 13, e0198089). Briefly, the distance from the upper extremity of the tibia to the medial malleolus (length of the tibia, L) is assessed; the perpendicular distance from the half-length of the tibia to the external margin of the hind limb muscle (thickness of the muscle, T) is assessed. These measurements are acquired by keeping the z axis fixed on the plane where the patella and the upper extremity of tibia are clearly identified. The Index of Muscle Mass (IMM) is defined as the ratio between T and L. The cross-sectional area of the muscle group at half length of the tibia is also measured using a ROI contour tool. For each mouse, the left and right leg are analyzed and averaged.
[0252] Histologic Analyses: The left tibias are collected fresh from each animal, and preserved in routine formalin fixative for 18 hours at 4 °C. The bone is processed for paraffin embedding, sectioned and stained. The toluidine blue satin will show the growth plate. Osteoclastic numbers and activity are evaluated by tartrate resistant acid phosphatase (TRAP) activity after demineralization and paraffin processing. Additional morphometric analyses of the growth plate are done to assess total width and composition. Immunostaining is done for the Natriuretic peptide receptor B(NPRB). Right tibias will undergo PMMA embedding for staining with Goldner’s trichrome which will be used to show osteoid and new bone formation.
[0253] FTIR Imaging Data Collection and Analysis: FTIR Imaging data is collected from histological sections of long bones to assess molecular composition of the mineralized regions using a Perkin Elmer Spotlight 400 system in the 800-4000 cm spectral region with a spatial resolution of either 6 or 25 microns. The system is comprised of an FTIR spectrometer coupled with a light microscope and an 8 X 2 linear array detector, and allows data collection over a region up to ~10 mm2. Matlab based data processing software is utilized for imaging data analysis. Spectral absorbances related to protein and mineral are evaluated for differences in relative mineral amount, collagen maturity, and mineral crystallinity, as previously described (Boskey et al., Biomaterials 2007, 28, 2465-2478; Boskey et al., Clin Orthop Relat Res 2015, 473, 2587-2598; Gollwitzer et aL, J Spectrosc (Hindawi) 2015, 2015.
[0254] Bone Biomechanics: All femurs are evaluated radiographically and only those that lack fractures or obvious deformities are selected for mechanical testing. Three-point bend tests are conducted at room temperature using a Bose Electroforce mechanical testing machine as previously described (Bargman 2010 and 2012, supra). From these curves, the Young’s
Modulus is determined by taking the slope of the elastic region of the curve. The yield point will be determined using a regression line over the linear region of the curve and adding data at successively higher stresses until the correlation coefficient (R2) drops below 0.95. The elastic percentage of the curve is determined by taking the yield strain and dividing it by the total strain (last point before the femur breaks). The remaining portion of the curve is defined as the plastic region. The material property “brittleness” of the bone is defined as the elastic region strain divided by the total strain.
[0255] Statistical Analysis and Outcomes: Statistical analyses is performed using SigmaStat software (SPSS inc., Chicago, IL) and GraphPad Prism (GraphPad Software, Boston, Massachusetts USA) software. The Grubbs Outlier test was used to identify and remove outliers from the datasets. Comparisons are made among treatment groups and between oim/oim and +/+ animals. For the fracture counting, a non-parametric measurement, the Mann-Whitney test, is utilized with differences considered significant at p < 0.05. Parametric data is analyzed by two-factor analysis of variance (ANOVA) to test for the simultaneous effects of genotype and treatment on the outcome variables. If significant differences are found for any factors, multiple comparisons are performed using the BonFerroni post hoc test. Values are considered significantly different at p < 0.05.
[0256] Starting at two weeks of age, growing oim/oim mice (N = 17) were divided into 4 groups, saline-treated (controls, n=8), and three groups of treated oim/oim mice where all received weekly ALN and divided into 3 CNP dosage groups: 20 pg/kg 5 days per week (20x5) (N = 3), 20 pg/kg 3 days per week (20x3) (N = 3), or 10 pg/kg 3 days per week (10x3) (N = 3). Faxitron images were taken at 2 and 14 weeks (sacrifice) to evaluate fracture incidence. Femurs and vertebrae (L3-L6) were analyzed post-sacrifice to assess femoral length, vertebral height, and other bone microstructural parameters by microcomputed tomography (micro-CT) analysis.
[0257] At sacrifice, all of the mice in the 20x5 group had no new fractures as shown in Table 1 . In comparison to one fracture in one mouse in both the 20x3 and 10x3 groups.
[0258] All three treatment groups had increased femoral lengths in comparison to the untreated oim/oim mice; the greatest increase was in the 20x5 group with the 20x3 and 10x3 having similar increases (Figure 3, Table 2).
[0259] All treated groups also had an increase in vertebral height; both the 20x5 and 10x3 groups had higher vertebral heights than the 20x3 group but were not different from each other.
(Table 3, Figure 4).
Table 3. Vertebral height
[0260] Micro-CT analysis of the cortical and trabecular bone tissue and bone density, porosity bone volume fraction, and thickness are illustrated in Figure 5. 3D images of trabecular bone (Figure 6) and cortical bone (Figure 7) in treated and untreated mice show that CNP administration improves bone growth in these areas, which was not observed with bisphosphonates alone as reported in previous studies. In comparison to untreated oim/oim mice, all three dosage groups had increased cortical bone tissue mineral density (TMD) (Figure 5), cortical bone mineral density (BMD) (Figure 8), cortical porosity (Figure 9), cortical bone thickness (Figure 10), trabecular bone volume fraction (BVF) (Figure 5), trabecular TMD (Figure 5), trabecular BMD (Figure 11 ), and trabecular bone number (Figure 5, Figure 12), and trabecular thickness (Figure 13). Furthermore, all three dosage groups had decreased trabecular bone separation (Figure 14).
[0261] Data points were collected from additional subjects receiving treatment and similar results were observed.
[0262] There were no fractures observed in any of the WT mice either pre- treatment or postsacrifice. At baseline enrollment, all of the untreated saline oim/oim mice had at least one fracture (1 .8 +/- 2.5 fractures); at sacrifice one mouse had two fractures while the others had none (Table 1 ). All of the mice in the high dosage group had at least one fracture at enrollment (4.14 +/- 0.45 fractures); at post-sacrifice there were no new fractures observed in any of the mice. All mice in the medium dosage group had at least one fracture at enrollment (3.16 +/- 0.65 fractures); at sacrifice two mice had one fracture each. All but one mouse in the low dosage
group had at least one fracture at enrollment (2.5 +/- 1 .17 fractures); at sacrifice only two mice had one fracture (Table 4).
[0263] Femoral Length: In the WT mice, only the medium dosage group had a significant increase in femoral length (p<0.001 ) in comparison to the saline control group (Figure 1 ). The high dosage and low dosage OIM CNP treatment groups had statistically significant increases in femoral length in comparison to the control OIM group (p<0.01 and p<0.05 respectively) (Figure 15). The medium dosage group did not have a statistically significant increase in femoral length. Therefore, for OIM mice, the high dosage group had the greatest increase in femoral length (8.2% increase), with the low dosage groups having a smaller increase (7.1% increase) (Figure 15).
[0264] Vertebral Height: As seen in the femoral length results, for the WT mice, only the medium dosage group had a statistically significant increase in vertebral height (p<0.05) compared to the saline mice. There was no significant difference between the saline, high dosage, and low dosage groups (Figure 16). For the OIM mice, as seen in the femurs, both the high dosage and low dosage groups had statistically significant increases in vertebral height (p<0.01 and p<0.05 respectively) (8.6% increase and 10.6% increase respectively) compared to the saline mice. There was no significant difference between the saline and medium dosage groups (Figure 16).
[0265] Micro-CT: In comparison to WT saline mice, all three WT dosage groups had an increase in cortical TMD, cortical minimum inertia, and trabecular TMD (Figures 17-18, Figure 21 ). In comparison to WT saline mice and WT ALN only mice19, all three WT dosage groups had an additional increase in cortical BMD, cortical thickness, trabecular BVF, and trabecular
thickness. None of the three WT dosage groups demonstrated increased trabecular number, trabecular BMD, or decreased trabecular separation that was demonstrated in the ALN only mice19. Only the WT medium dosage group showed an increase in cortical porosity compared to WT saline mice (Figure 17, Figure 21 ).
[0266] In comparison to OIM saline mice, all three OIM dosage groups had an increase in cortical TMD and trabecular TMD; there was no difference in cortical porosity or cortical minimum inertia (Figures 19-21). In comparison to OIM saline mice and OIM ALN only mice19, all three OIM dosage groups had an additional increase in cortical BMD, cortical thickness, and trabecular BMD (Figures 19-21). None of the three OIM dosage groups demonstrated increased trabecular BVF, trabecular number, trabecular thickness, or decreased trabecular separation that was demonstrated in the ALN only mice.
[0267] In this preliminary study, it appears that the adjunctive treatment of a CNP analog results in an overall increase in both femoral and vertebral heights without compromising fracture reduction. In addition, the combination treatment increases bone length (the key unmet need in Ol patients) and appears to have an additive beneficial effect on both trabecular and cortical bone quality; the latter of which was not seen with bisphosphonates alone. These results are useful to further study therapeutic doses of the CNP analog and carry out additional animal studies to support the use of CNP therapy in human patients with Ol.
Example 2. Improvement of Bone Strength using CNP
[0268] The primary objective of this study was to determine whether CNP variant affects both length and development of bone strength in children with achondroplasia using measurements of the second metacarpal.
[0269] This study included 103 deidentified AP hand/wrist radiographs from 30 children with achondroplasia (13M, 17F; ages 7.8-16 years). Proprietary data included hand films collected at four time points: at week 104 (at rollover into the phase II extension study), week 156, week 208 and week 260 on treatment. Second metacarpal length and midshaft width, cortical thickness, robustness (total area/length), and cortical area (correlated with strength) were measured. Measurements were compared to 378 radiographs from 1 14 average-stature controls (61 M, 53F; ages 6-16 years) via non-parametric Kruskal-Wallis tests (p<0.05).
[0270] Children with achondroplasia at week 208 and week 260 of treatment demonstrated longer metacarpals with increased cortical area and cortical thickness compared to week 104 (all p<0.05). There was no significant change in RCA and robustness across the treatment time
points (Table 5). Furthermore, no differences were seen between males and females with achondroplasia at any of the timepoints. Compared to the controls, children with achondroplasia had higher robustness and cortical area throughout treatment (p<0.001).
Table 5: Measurements of the second metacarpal (mean±SD) at each time point in treatment.
Baseline Week 52 Week 104 Week 156 Controls
(n=29) (n=29) (n=28)
T.Ar (mm2) 42.84±1 1.82A 45.79±12.49° 49.80±14.63° 54.29±17.64*° 39.01 ±11 .79#tA
Ct.Ar (mm2) 25.60±6.59#A° 28.12±6.72A 32.06±7.93* 35.60±9.59*#° 29.64±9.57*A
Marrow Area
(mm2) 17.24+9.38° 17.67±10.54° 17.73±12.15° 18.68±12.06° 9.37±4.81 *#tA
Cortical
Thickness
(mm) 1.39±0.35i ' ° 1 .49±0.37° 1.67+0.43*° 1.77±0.41 *° 1 .10±0.35*#tA
RCA 0.61 ±0.12° 0.63±0.12° 0.66±0.12° 0.67±0.12° 0.76±0.10*#tA
T.Ar/Le (mm) 1 .08±0.30° 1 .09±0.29° 1.13±0.32° 1 .20±0.37° 0.68±0.13*#tA
*significantly different (p<0.05) than baseline, Ssignificantly different (p<0.05) than w52, ^significantly different (p<0.05) than wl04, Significantly different (p<0.05) than wl56, "significantly different (p<0.05) than controls"
[0271] It was observed that an additional 2-3 years of vosoritide treatment was associated with increases in bone length compared to the initial timepoint (week 104) as well as increases in metacarpal cortical area, which is correlated with strength. This study suggested this bone lengthening treatment did not adversely affect bone strength in children with achondroplasia. The lack of a significant difference in robustness after treatment indicated that periosteal expansion continued outward at a pace which maintains robustness, allowing the bone to remain strong as it lengthened. It was also recently reported children with achondroplasia treated for 4 and 5 years with CNP analog, e.g., vosoritide, demonstrated longer metacarpals with increased cortical area compared to baseline; those at year 5 also displayed increased cortical thickness (DeRocher et al., “Does Vosoritide Treatment Affect Bone Strength in Children with Achondroplasia?” poster, International Skeletal Dysplasia Society Conference, Madrid, Spain, Sept. 18, 2024). Future work comparing treated and untreated children with achondroplasia is necessary to more rigorously confirm whether the treatment did not adversely affect the development of bone strength. Overall, this work may have important clinical implications in terms of treatment choices for children with achondroplasia.
[0272] It is understood that every embodiment of the disclosure described herein may optionally be combined with any one or more of the other embodiments described herein. Every patent literature and every non-patent literature cited herein are incorporated herein by reference in their entirety.
[0273] It is understood, therefore, that this invention is not limited to the particular embodiments disclosed, but is intended to cover all modifications which are within the spirit and scope of the invention as defined by the appended claims; the above description, and/or shown in the attached drawings. Consequently only such limitations as appear in the appended claims should be placed on the disclosure.
Claims
1 . A method of treating osteogenesis imperfecta comprising administering a C-type natriuretic peptide (CNP) to a subject in need thereof, wherein the CNP is administered between 2 to 5 times per week or weekly.
2. A method of reducing the frequency of bone fracture in a subject having osteogenesis imperfecta (Ol) comprising administering a C-type natriuretic peptide (CNP) to a subject in need thereof, wherein the CNP is administered between 2 to 5 times per week or weekly.
3. A method of improving bone mineral density in a subject having osteogenesis imperfecta comprising administering a C-type natriuretic peptide (CNP) to a subject in need thereof, wherein the CNP is administered between 2 to 5 times per week or weekly.
4. A method of improving femoral length or vertebral growth in a subject having osteogenesis imperfecta comprising administering a C-type natriuretic peptide (CNP) to a subject in need thereof, wherein the CNP is administered between 2 to 5 times per week or weekly.
5. The method of any one of claims 1 to 4 wherein the osteogenesis imperfecta is Type I, Type II, Type III, Type IV, or Type V Ol.
6. The method of any one of claims 1 to 4, wherein the osteogenesis imperfecta is Type VI -Type XXI Ol.
7. The method of any one of claims 1 to 6, wherein the CNP is administered intravenously or subcutaneously.
8. The method of any one of claims 1 to 7, wherein the subject is further administered a bisphosphonate.
9. The method of claim 8, wherein the bisphosphonate is selected from the group consisting of pamidronate, alendronate and risedronate, or combinations thereof.
10. The method of claim 8 or 9, wherein the bisphosphate is administered orally, intravenously or subcutaneously.
11 . The method of any one of claims 1 -10, wherein the subject has a mutation in a gene selected from the group consisting of COL1 A1 , COL1 A2, COL2A1 , COL11 A1 , COL9A2, COL10, IFITM5, SERPINF1 , CRTAP, LEPRE1 , PPIB, SERPINH1 , FKBP10, SP7, BMP1 , TMEM38B, WNT1 , CREB3L1 , SPARC, FAM46A or combinations thereof.
12. The method of any one of claims 1 to 1 1 , wherein the CNP is a CNP variant selected from the group consisting of
PGQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (Pro-Gly-CNP37) (SEQ ID NO: 1 ); GQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (Gly-CNP-37) (SEQ ID NO: 2);
GDLRVDTKSRAAWARLLQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (Gly- CNP53) (SEQ ID NO: 3);
PDLRVDTKSRAAWARLLQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (Pro- CNP53) (SEQ ID NO: 4);
MDLRVDTKSRAAWARLLQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (Met- CNP53) (SEQ ID NO: 5);
DLRVDTKSRAAWARLLQEHPNARKYKGANKKGLSKGCFGLKLDRIGSNSGLGC [CNP- 53(M48N)] (SEQ ID NO: 6);
LRVDTKSRAAWARLLQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-52) (SEQ ID NO: 7);
RVDTKSRAAWARLLQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-51 ) (SEQ ID NO: 8);
VDTKSRAAWARLLQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP- 50) (SEQ ID NO: 9);
DTKSRAAWARLLQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-49) (SEQ ID NO: 10);
TKSRAAWARLLQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-48) (SEQ ID NO: 11 );
KSRAAWARLLQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-47) (SEQ ID NO: 12);
SRAAWARLLQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-46) (SEQ ID NO:
13);
RAAWARLLQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-45) (SEQ ID NO:
14);
AAWARLLQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-44) (SEQ ID NO:
15);
AWARLLQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-43) (SEQ ID NO: 16);
WARLLQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-42) (SEQ ID NO: 17);
ARLLQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-41 ) (SEQ ID NO: 18);
RLLQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-40) (SEQ ID NO: 19);
LLQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-39) (SEQ ID NO: 20);
LQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-38) (SEQ ID NO: 21 );
QEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-37) (SEQ ID NO: 22);
EHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-36) (SEQ ID NO: 23);
HPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-35) (SEQ ID NO: 24);
PNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-34) (SEQ ID NO: 25);
NARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-33) (SEQ ID NO: 26);
ARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-32) (SEQ ID NO: 27) ;
RKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-31 ) (SEQ ID NO: 28);
KYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-30) (SEQ ID NO: 29);
YKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-29) (SEQ ID NO: 30);
KGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-28) (SEQ ID NO: 31 );
GANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-27) (SEQ ID NO: 32);
ANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-26) (SEQ ID NO: 33); NKKGLSKGCFGLKLDRIGSMSGLGC (CNP-25) (SEQ ID NO: 34);
KKGLSKGCFGLKLDRIGSMSGLGC (CNP-24) (SEQ ID NO: 35);
KGLSKGCFGLKLDRIGSMSGLGC (CNP-23) (SEQ ID NO: 36);
LSKGCFGLKLDRIGSMSGLGC (CNP-21 ) (SEQ ID NO: 37);
SKGCFGLKLDRIGSMSGLGC (CNP-20) (SEQ ID NO: 38);
KGCFGLKLDRIGSMSGLGC (CNP- 19) (SEQ ID NO: 39);
GCFGLKLDRIGSMSGLGC (CNP-18) (SEQ ID NO: 40);
QEHPNARKYKGANKKGLSKGCFGLKLDRIGSNSGLGC [CNP-37(M32N)] (SEQ ID NO: 41 );
PQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (Pro-CNP-37) (SEQ ID NO: 42);
MQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (Met-CNP-37) (SEQ ID NO: 43);
GQEHPNARKYKGANKKGLSKGCFGLKLDRIGSNSGLGC [Gly-CNP-37(M32N)] (SEQ ID NO:
44);
MGQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (Met-Gly-CNP-37) (SEQ ID NO:
45);
PGQEHPQARRYRGAQRRGLSRGCFGLKLDRIGSMSGLGC (SEQ ID NO: 46); PGQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (SEQ ID NO: 47);
PGQEHPNARRYRGANRRGLSRGCFGLKLDRIGSMSGLGC (SEQ ID NO: 48); and PGQEHPQARKYKGAQKKGLSKGCFGLKLDRIGSMSGLGC (SEQ ID NO: 49).
13. The method of claim 12, wherein the CNP variant is PGQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (Pro-Gly-CNP37) (SEQ ID NO: 1); GQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (Gly-CNP-37) (SEQ ID NO: 2); or LQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-38) (SEQ ID NO: 21 ).
14. The method of any one of claims 1 to 13, wherein the subject is receiving between 0.5 pg/kg and 30 pg/kg CNP therapy.
15. The method of any one of claims 1 to 14, wherein the subject is receiving 10 pg/kg or 20 pg/kg CNP therapy.
16. The method of any one of claims 1 to 15, wherein the CNP is administered in a dose of 20 pg/kg 5 times weekly, a dose of 20 pg/kg 3 times weekly or a dose of 10 pg/kg 3 times weekly.
17. The method of any one of claims 1 to 16 wherein CNP therapy alleviates one or more symptoms of osteogenesis imperfecta, including but not limited to, femoral length, vertebral height, reduction in bone fracture frequency, cortical bone tissue mineral density (TMD), cortical bone mineral density (BMD), cortical bone thickness, cortical porosity, trabecular bone volume fraction (BVF), trabecular TMD, trabecular BMD, trabecular bone number and trabecular bone separation.
18. A method for improving bone strength in a subject in need thereof comprising administering a C-type natriuretic peptide (CNP) to the subject.
19. The method of claim 18, wherein the CNP is a CNP variant selected from the group consisting of
PGQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (Pro-Gly-CNP37) (SEQ ID NO: 1 ); GQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (Gly-CNP-37) (SEQ ID NO: 2);
GDLRVDTKSRAAWARLLQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (Gly- CNP53) (SEQ ID NO: 3);
PDLRVDTKSRAAWARLLQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (Pro- CNP53) (SEQ ID NO: 4);
MDLRVDTKSRAAWARLLQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (Met- CNP53) (SEQ ID NO: 5);
DLRVDTKSRAAWARLLQEHPNARKYKGANKKGLSKGCFGLKLDRIGSNSGLGC [CNP- 53(M48N)] (SEQ ID NO: 6);
LRVDTKSRAAWARLLQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-52) (SEQ ID NO: 7);
RVDTKSRAAWARLLQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-51 ) (SEQ ID NO: 8);
VDTKSRAAWARLLQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP- 50) (SEQ ID NO: 9);
DTKSRAAWARLLQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-49) (SEQ ID NO: 10);
TKSRAAWARLLQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-48) (SEQ ID NO: 11 );
KSRAAWARLLQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-47) (SEQ ID NO: 12);
SRAAWARLLQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-46) (SEQ ID NO:
13);
RAAWARLLQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-45) (SEQ ID NO:
14);
AAWARLLQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-44) (SEQ ID NO:
15);
AWARLLQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-43) (SEQ ID NO: 16);
WARLLQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-42) (SEQ ID NO: 17);
ARLLQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-41 ) (SEQ ID NO: 18);
RLLQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-40) (SEQ ID NO: 19);
LLQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-39) (SEQ ID NO: 20);
LQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-38) (SEQ ID NO: 21 );
QEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-37) (SEQ ID NO: 22);
EHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-36) (SEQ ID NO: 23);
HPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-35) (SEQ ID NO: 24);
PNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-34) (SEQ ID NO: 25);
NARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-33) (SEQ ID NO: 26);
ARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-32) (SEQ ID NO: 27) ;
RKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-31 ) (SEQ ID NO: 28);
KYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-30) (SEQ ID NO: 29);
YKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-29) (SEQ ID NO: 30);
KGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-28) (SEQ ID NO: 31 );
GANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-27) (SEQ ID NO: 32);
ANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-26) (SEQ ID NO: 33);
NKKGLSKGCFGLKLDRIGSMSGLGC (CNP-25) (SEQ ID NO: 34);
KKGLSKGCFGLKLDRIGSMSGLGC (CNP-24) (SEQ ID NO: 35);
KGLSKGCFGLKLDRIGSMSGLGC (CNP-23) (SEQ ID NO: 36);
LSKGCFGLKLDRIGSMSGLGC (CNP-21 ) (SEQ ID NO: 37);
SKGCFGLKLDRIGSMSGLGC (CNP-20) (SEQ ID NO: 38);
KGCFGLKLDRIGSMSGLGC (CNP- 19) (SEQ ID NO: 39);
GCFGLKLDRIGSMSGLGC (CNP-18) (SEQ ID NO: 40);
QEHPNARKYKGANKKGLSKGCFGLKLDRIGSNSGLGC [CNP-37(M32N)] (SEQ ID NO: 41 );
PQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (Pro-CNP-37) (SEQ ID NO: 42);
MQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (Met-CNP-37) (SEQ ID NO: 43);
GQEHPNARKYKGANKKGLSKGCFGLKLDRIGSNSGLGC [Gly-CNP-37(M32N)] (SEQ ID NO:
44);
MGQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (Met-Gly-CNP-37) (SEQ ID NO:
45);
PGQEHPQARRYRGAQRRGLSRGCFGLKLDRIGSMSGLGC (SEQ ID NO: 46);
PGQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (SEQ ID NO: 47);
PGQEHPNARRYRGANRRGLSRGCFGLKLDRIGSMSGLGC (SEQ ID NO: 48); and PGQEHPQARKYKGAQKKGLSKGCFGLKLDRIGSMSGLGC (SEQ ID NO: 49).
20. The method of claim 19, wherein the CNP variant is
PGQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (Pro-Gly-CNP37) (SEQ ID NO: 1);
GQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (Gly-CNP-37) (SEQ ID NO: 2); or LQEHPNARKYKGANKKGLSKGCFGLKLDRIGSMSGLGC (CNP-38) (SEQ ID NO: 21 ).
Applications Claiming Priority (8)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US202363542816P | 2023-10-06 | 2023-10-06 | |
| US63/542,816 | 2023-10-06 | ||
| US202363544006P | 2023-10-13 | 2023-10-13 | |
| US63/544,006 | 2023-10-13 | ||
| US202463564067P | 2024-03-12 | 2024-03-12 | |
| US63/564,067 | 2024-03-12 | ||
| US202463666302P | 2024-07-01 | 2024-07-01 | |
| US63/666,302 | 2024-07-01 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2025076498A1 true WO2025076498A1 (en) | 2025-04-10 |
Family
ID=95284139
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2024/050159 Pending WO2025076498A1 (en) | 2023-10-06 | 2024-10-07 | Treatment of osteogenesis imperfecta with c-type natriuretic peptide and analogs thereof |
Country Status (2)
| Country | Link |
|---|---|
| TW (1) | TW202532429A (en) |
| WO (1) | WO2025076498A1 (en) |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20150125457A1 (en) * | 2012-04-25 | 2015-05-07 | Daiichi Sankyo Company, Limited | Bone repair promoter |
| US20190216943A1 (en) * | 2016-08-11 | 2019-07-18 | Precithera, Inc. | Tgf-beta antagonist conjugates |
| US20230192799A1 (en) * | 2019-09-16 | 2023-06-22 | Biomarin Pharmaceutical Inc. | CNP Variants and Conjugates Thereof |
-
2024
- 2024-10-07 WO PCT/US2024/050159 patent/WO2025076498A1/en active Pending
- 2024-10-07 TW TW113138102A patent/TW202532429A/en unknown
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20150125457A1 (en) * | 2012-04-25 | 2015-05-07 | Daiichi Sankyo Company, Limited | Bone repair promoter |
| US20190216943A1 (en) * | 2016-08-11 | 2019-07-18 | Precithera, Inc. | Tgf-beta antagonist conjugates |
| US20230192799A1 (en) * | 2019-09-16 | 2023-06-22 | Biomarin Pharmaceutical Inc. | CNP Variants and Conjugates Thereof |
Non-Patent Citations (1)
| Title |
|---|
| MEDLINEPLUS: "Osteogenesis imperfecta", MEDLINEPLUS, 8 June 2023 (2023-06-08), pages 1 - 6, XP093302834, Retrieved from the Internet <URL:https://web.archive.org/web/20230608035812/https://medlineplus.gov/genetics/condition/osteogenesis-imperfecta/#references> * |
Also Published As
| Publication number | Publication date |
|---|---|
| TW202532429A (en) | 2025-08-16 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US12514906B2 (en) | Use of C-type natriuretic peptide variants to treat skeletal dysplasia | |
| JP7758663B2 (en) | CNP variants and conjugates thereof | |
| US12233106B2 (en) | C-type natriuretic peptide variants to treat skeletal dysplasia in children | |
| US20250032576A1 (en) | CNP Therapy | |
| WO2025076498A1 (en) | Treatment of osteogenesis imperfecta with c-type natriuretic peptide and analogs thereof | |
| WO2026030743A1 (en) | Treatment of short stature with cnp variants | |
| TW202535449A (en) | Cnp variants, conjugates and formulations thereof | |
| CN117881416A (en) | C-type natriuretic peptide variants for the treatment of skeletal dysplasia in children | |
| HK40114833A (en) | Use of c-type natriuretic peptide variants to treat skeletal dysplasia | |
| HK40075682A (en) | Cnp variants and conjugates thereof | |
| HK1255864B (en) | Use of c-type natriuretic peptide variants to treat skeletal dysplasia |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 24875568 Country of ref document: EP Kind code of ref document: A1 |