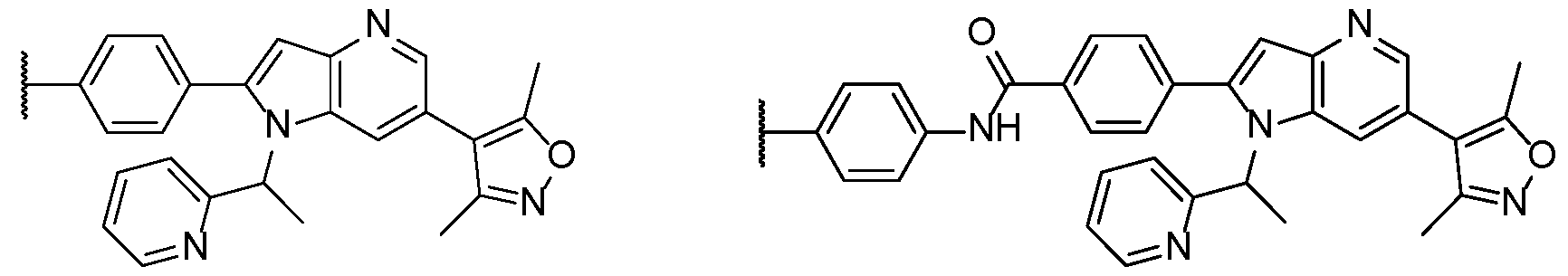
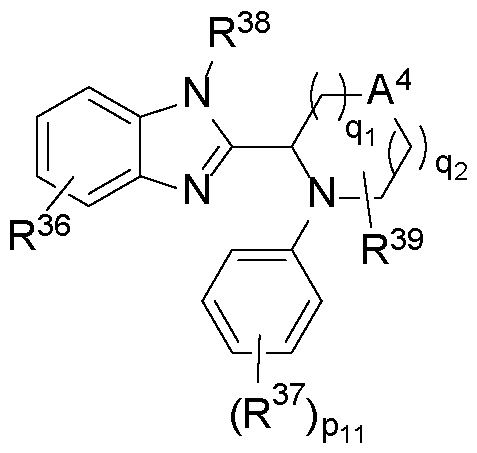
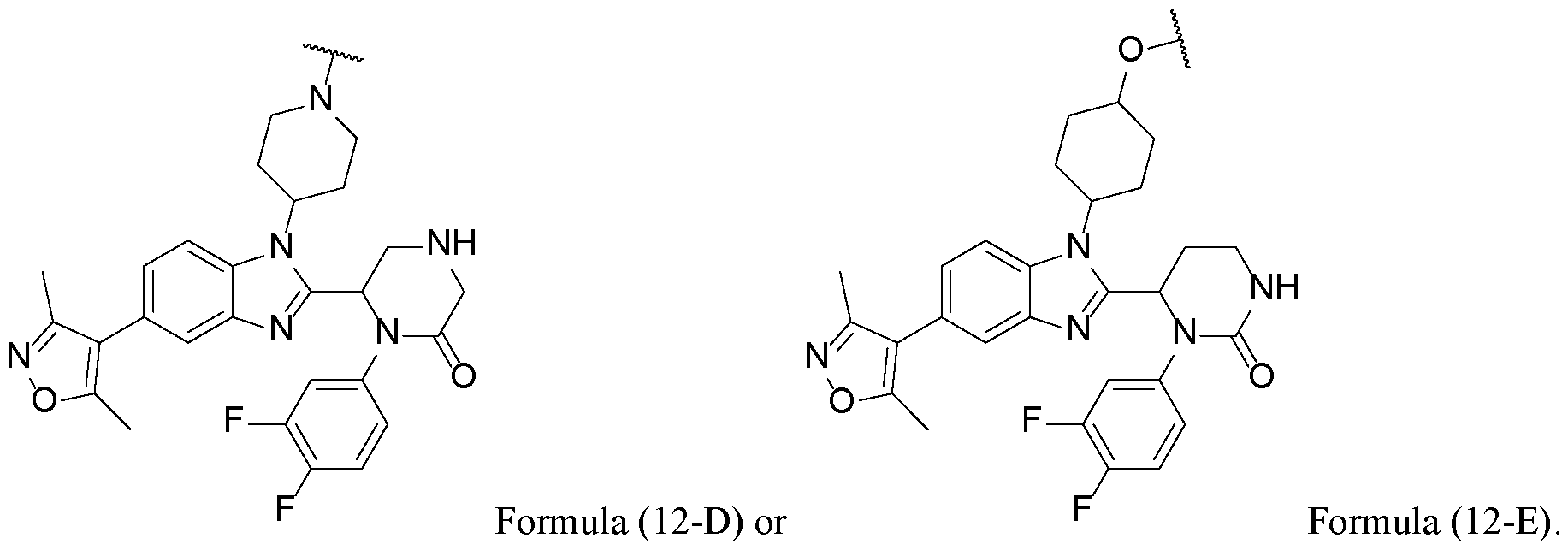
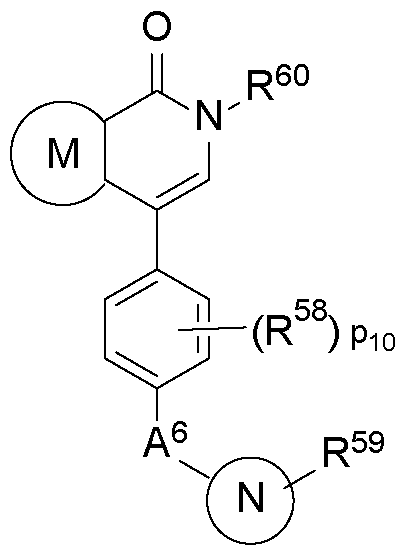
WO2025076219A1 - Methods and compounds for modulating huntington's disease - Google Patents
Methods and compounds for modulating huntington's disease Download PDFInfo
- Publication number
- WO2025076219A1 WO2025076219A1 PCT/US2024/049785 US2024049785W WO2025076219A1 WO 2025076219 A1 WO2025076219 A1 WO 2025076219A1 US 2024049785 W US2024049785 W US 2024049785W WO 2025076219 A1 WO2025076219 A1 WO 2025076219A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- optionally substituted
- alkyl
- hydrogen
- independently
- pharmaceutically acceptable
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D495/00—Heterocyclic compounds containing in the condensed system at least one hetero ring having sulfur atoms as the only ring hetero atoms
- C07D495/12—Heterocyclic compounds containing in the condensed system at least one hetero ring having sulfur atoms as the only ring hetero atoms in which the condensed system contains three hetero rings
- C07D495/14—Ortho-condensed systems
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/51—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent
- A61K47/54—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an organic compound
- A61K47/55—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an organic compound the modifying agent being also a pharmacologically or therapeutically active agent, i.e. the entire conjugate being a codrug, i.e. a dimer, oligomer or polymer of pharmacologically or therapeutically active compounds
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D401/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom
- C07D401/14—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing three or more hetero rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D413/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms
- C07D413/14—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms containing three or more hetero rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D471/00—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00
- C07D471/02—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00 in which the condensed system contains two hetero rings
- C07D471/04—Ortho-condensed systems
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/28—Drugs for disorders of the nervous system for treating neurodegenerative disorders of the central nervous system, e.g. nootropic agents, cognition enhancers, drugs for treating Alzheimer's disease or other forms of dementia
Definitions
- HD Huntington’s disease
- Pathology stems from a cascade of steps: production of poly-Q huntingtin, followed by fragmentation of the elongated huntingtin into smaller peptides, which bind together and accumulate in neurons.
- the effects of this cascade are pronounced in the basal ganglia and cortex of the brain.
- Huntington’s disease-like syndrome refers to a group of ailments whose symptoms are similar to those of Huntington’s disease, but which lack the characteristic mutation in the HTT gene.
- Huntington’s disease-like 2 syndrome (“HDL2”) is associated with a count of about 40 or more CAG trinucleotide repeat sequences in the junctophilin 3 Jph3) gene.
- HDL2 is a genetic disorder that has been seen in subjects with African lineage. Age of onset is inversely correlated with the number of trinucleotide repeat sequences. Symptoms of this syndrome include dystonia and chorea (uncontrolled movements), emotional disruptions, dysarthria, bradykinesia, inability to incorporate new learning, and difficulty in making decisions.
- the mechanism set forth above provides opportunity for an effective treatment for a disease or disorder which is characterized by the presence of an excessive count of CAG trinucleotide repeat sequences in a target gene.
- the pathology of the disease or disorder is due to the presence of mRNA containing an excessive count of CAG trinucleotide repeat sequences.
- the pathology of the disease or disorder is due to the presence of a translation product containing an excessive count of glutamine amino acid residues.
- the pathology of the disease or disorder is due to a loss of function in the translation product.
- the pathology of the disease or disorder is due to a gain of function in the translation product.
- the pathology of the disease or disorder can be alleviated by increasing the rate of transcription of the defective gene.
- the pathology of the disease or disorder can be alleviated by decreasing the rate of transcription of the defective gene.
- This disclosure utilizes regulatory molecules present in cell nuclei that control gene expression.
- Eukaryotic cells provide several mechanisms for controlling gene replication, transcription, and/or translation. Regulatory molecules that are produced by various biochemical mechanisms within the cell can modulate the various processes involved in the conversion of genetic information to cellular components.
- Regulatory molecules are known to modulate the production of mRNA and, if directed to the target gene (such as, HTT), would modulate the production of the target gene mRNA that causes diseases such as, for example, Huntington’s disease or Huntington’s disease-like syndrome, and thus reverse the progress of these diseases.
- transcription modulator molecules for recruiting a regulatory molecule into close proximity to the target gene.
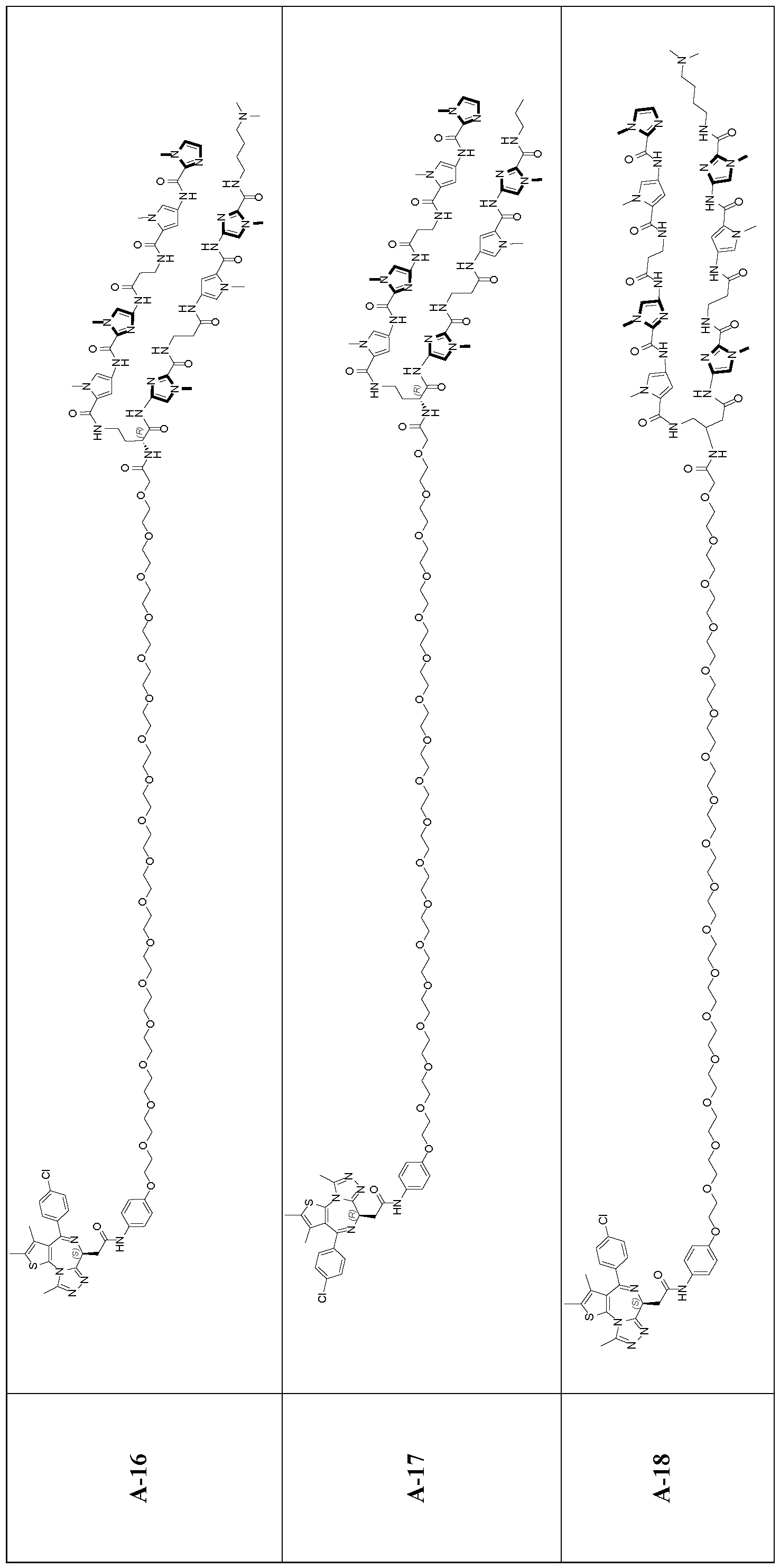
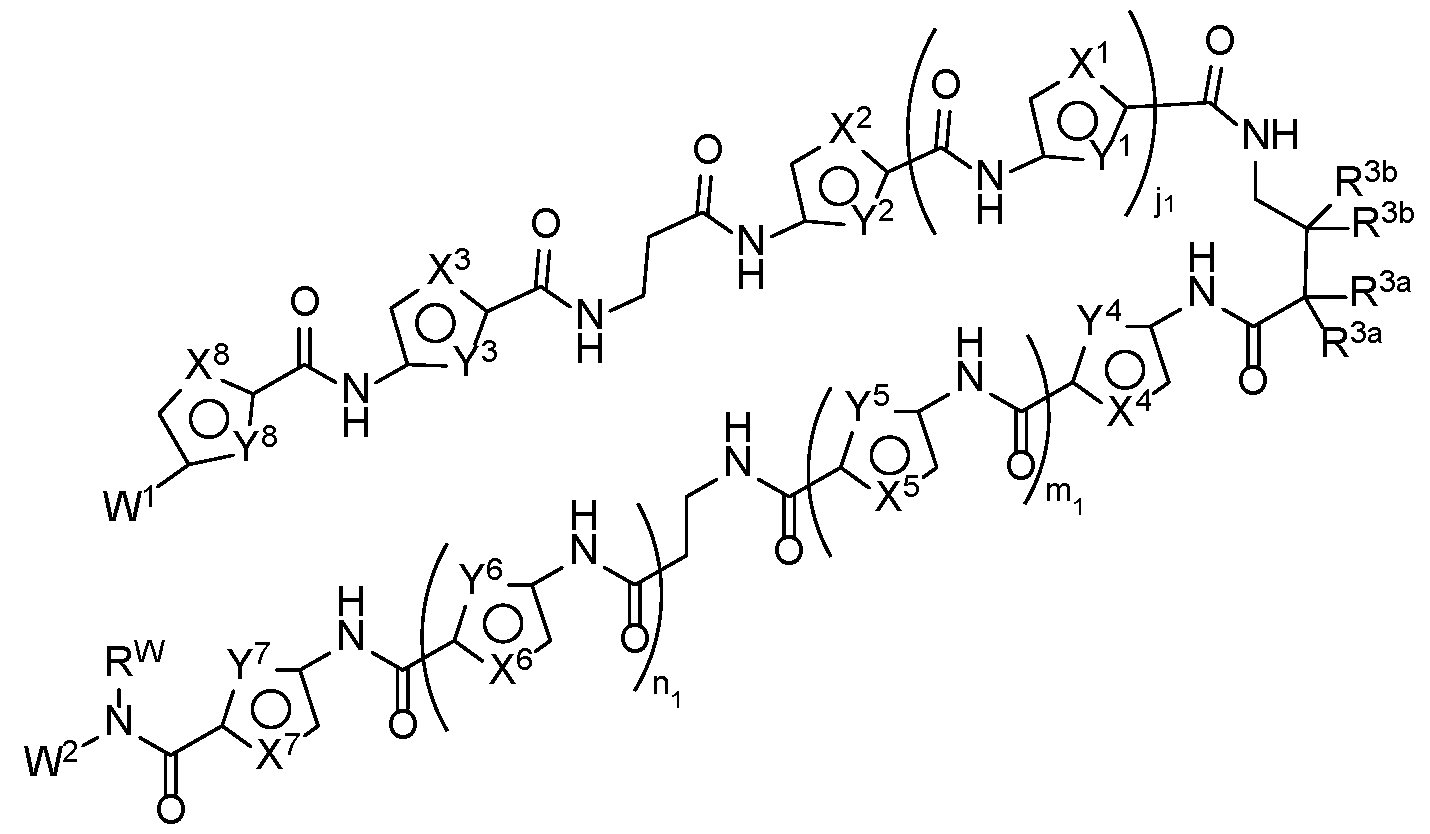
- a transcription modulator molecule having a first terminus, a second terminus, and an oligomeric backbone moiety, or a pharmaceutically acceptable salt thereof, wherein: a) the first terminus comprises a DNA-binding moiety having the structure of Formula (A-l):
- each R le is independently hydrogen or optionally substituted Ci-Cw alkyl
- each R lf is independently hydrogen, optionally substituted C1-C10 alkyl, optionally substituted C1-C10 heteroalkyl, AA P 2, or optionally substituted 5-membered heteroaryl, wherein each AA is an amino acid and p2 is an integer from 1-10;
- ZB is N or O; ps is 1-10;
- W 2 is optionally substituted C1-C20 alkyl, optionally substituted C1-C20 aminoalkyl, optionally substituted C1-C20 haloalkyl, optionally substituted C1-C20 heteroalkyl, or optionally substituted C1-C20 hydroxyalkyl; or W 2 is L’-Z-R 4 ; wherein
- L 1 is C1-C20 alkylene or C2-C20 heteroalkylene
- R 4 is C1-C3 alkyl, -OR 4b , or -NR 4a R 4b ; wherein
- R 4a is hydrogen, optionally substituted C1-C20 alkyl, or optionally substituted C1-C20 heteroalkyl;
- R 4b is optionally substituted C1-C20 alkyl, optionally substituted C2-C20 alkenyl, optionally substituted C2-C20 alkynyl, optionally substituted C1-C20 aminoalkyl, optionally substituted C1-C20 haloalkyl, optionally substituted C1-C20 heteroalkyl, optionally substituted C1-C20 hydroxyalkyl, optionally substituted C3-C8 cycloalkyl, optionally substituted 4 to 8-membered heterocycloalkyl, optionally substituted phenyl, or optionally substituted 5 to 10-membered heteroaryl; or
- R 4a and R 4b together with the nitrogen to which they are attached form an optionally substituted 3 to 8-membered heterocycloalkyl which is partially or fully saturated;
- R w is hydrogen or optionally substituted C1-C20 alkyl
- R w and W 2 combine together with the atoms to which they are attached to form an optionally substituted 4 to 8-membered heterocycloalkyl; each Y 1 , Y 2 , Y 3 , Y 4 , Y 5 , Y 6 , Y 7 , and Y 8 is independently N or CH; each X 1 , X 2 , X 3 , X 4 , X 5 , X 6 , X 7 , and X 8 is independently S, O, or NR 2 ; each R 2 is independently hydrogen, optionally substituted C1-C50 alkyl, optionally substituted C2-C50 alkenyl, optionally substituted C2-C50 alkynyl, optionally substituted C1-C50 heteroalkyl, optionally substituted C2-C50 heteroalkenyl, optionally substituted C2-C50 heteroalkynyl, optionally substituted C1-C50 haloalkyl, optionally substituted C3-C8 cycl
- R 3e is alkyl, PEG, cycloalkyl, heterocycloalkyl, or phenyl; or two R 3 together with the atoms to which they are attached form an optionally substituted C3-C6 cycloalkyl or 3 to 6 membered heterocycloalkyl; ji is 0 or 1; no is 0 or 1 ; mi and are each independently an integer from 0-3; and pi is 3 or 4; wherein the linker is attached at one of R 3 and the others of R 3 are each hydrogen; or the linker is attached at the cycloalkyl or heterocycloalkyl formed by two R 3 ; b) the second terminus comprises a protein-binding moiety capable of binding to a regulatory molecule that modulates expression of a gene; and c) the oligomeric backbone links the first terminus and the second terminus.
- the DNA binding moiety can selectively bind a trinucleotide repeat sequence in for example, HTT.
- the recruiting moiety, linked to the DNA binding moiety will thus be held in proximity to the target gene; will recruit the regulatory molecule into proximity with the gene; and the regulatory molecule will modulate expression, and therefore counteract the production of defective target gene by direct interaction with the target gene.
- This mechanism may provide an effective treatment for HD, which is caused by the expression of defective HTT, where correction of the expression of the defective target gene thus represents an effective method for the treatment for these diseases.
- the DNA binding moiety comprises a polyamide segment that will bind selectively to the target CAG sequence.
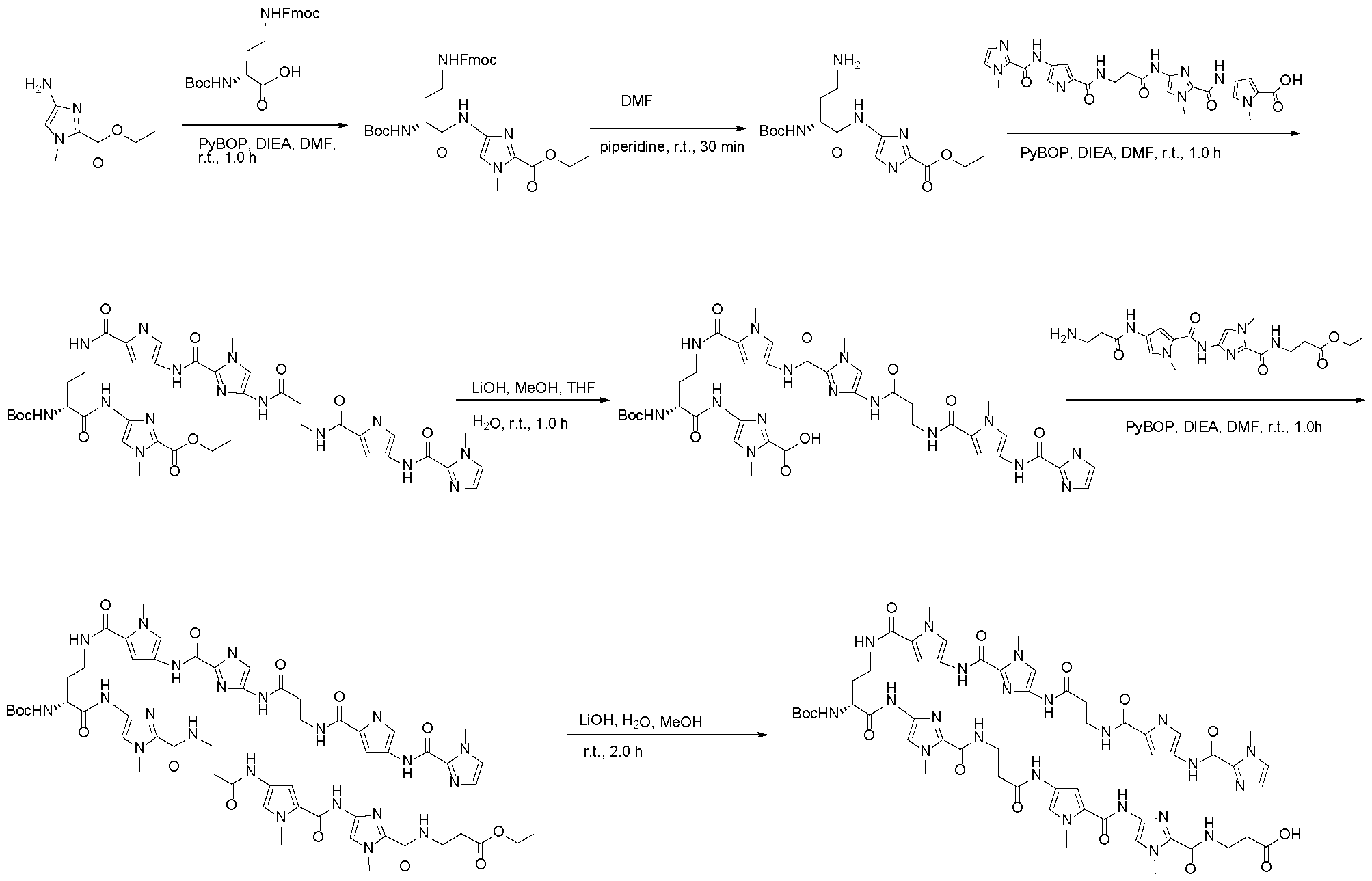
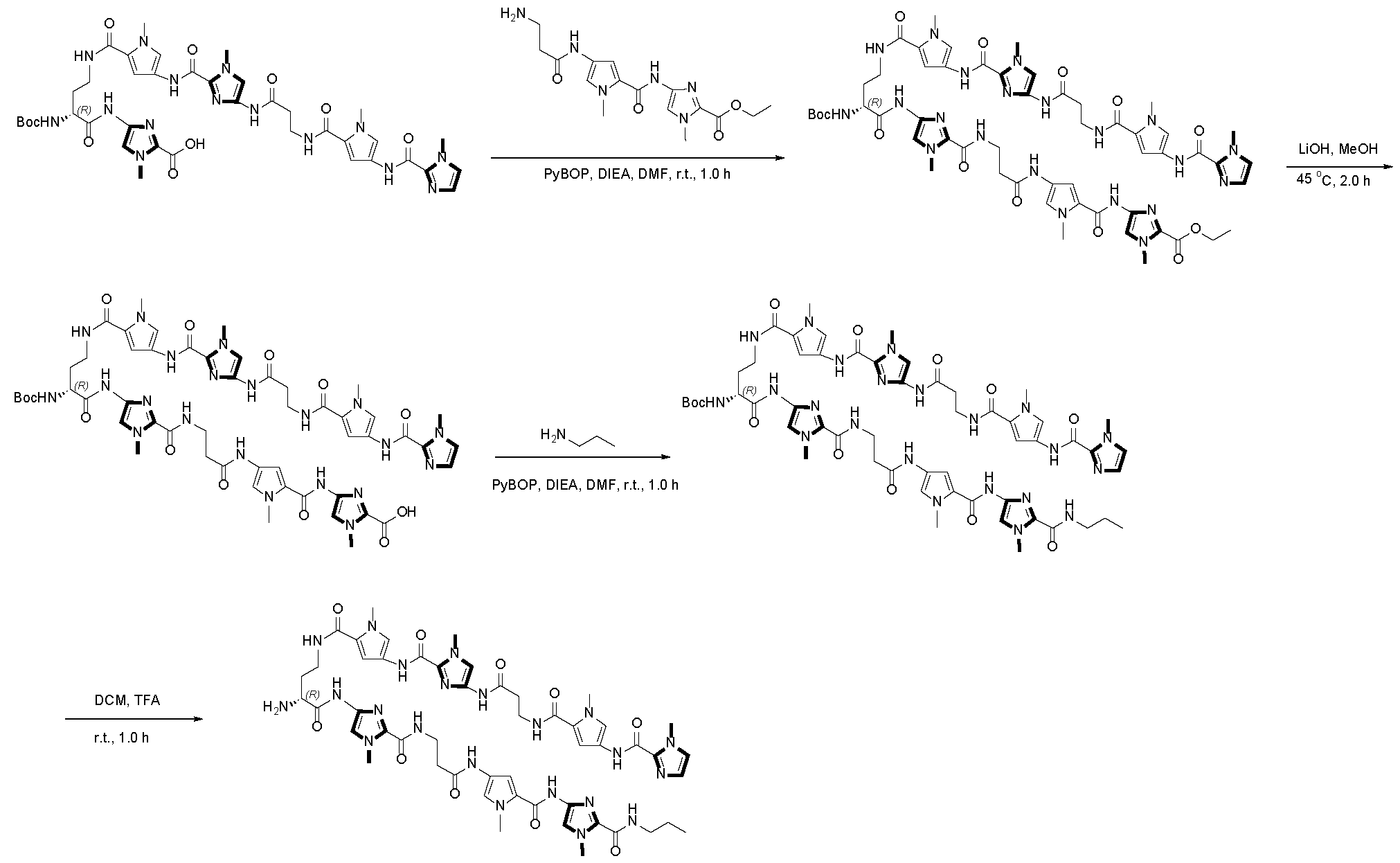
- Polyamides designed by for example Dervan (U.S. Patent Nos. 9,630,950 and 8,524,899) and others can selectively bind to selected DNA sequences. These polyamides sit in the minor groove of double helical DNA and form hydrogen bonding interactions with the Watson-Crick base pairs.
- Polyamides that selectively bind to particular DNA sequences can be designed by linking monoamide building blocks according to established chemical rules. One building block is provided for each DNA base pair, with each building block binding noncovalently and selectively to one of the DNA base pairs: A/T, T/A, G/C, and C/G. Following this guideline, trinucleotides bind to molecules with three amide units, i.e. tri -amides. In general, these polyamides can orient in either direction of a DNA sequence.
- the disclosure provides for transcription modulator molecules that comprise a DNA binding moiety (for example, a polyamide comprising multi-amine subunits) connected by spacers (for example, a linker moiety or oligomeric backbone) to a protein binding moiety.
- a DNA binding moiety for example, a polyamide comprising multi-amine subunits
- spacers for example, a linker moiety or oligomeric backbone
- Treatment of a subject with these compounds will modulate the expression of the defective target gene, and this can reduce the occurrence, severity, or frequency of symptoms associated with genetic disease (such as for example HD).
- the compounds described herein recruit the regulatory molecule to modulate the expression of the defective target gene and effectively treat and alleviate the symptoms associated with diseases.
- the compounds disclosed herein are transcription modulator molecules. They possess useful activity for modulating the transcription of a target gene (e.g., HTT) and may be used in the treatment or prophylaxis of a disease or condition in which the target gene plays an active role.
- a target gene e.g., HTT
- some embodiments also provide pharmaceutical compositions comprising one or more compounds disclosed herein together with a pharmaceutically acceptable carrier, as well as methods of making and using the compounds and compositions.
- a transcription modulator molecule having a first terminus, a second terminus, and a linker moiety, wherein: a) the first terminus comprises a DNA-binding moiety; b) the second terminus comprises a protein-binding moiety capable of binding to a regulatory molecule that modulates expression of a gene; and c) the oligomeric backbone links the first terminus and the second terminus.
- First terminus - DNA binding moiety The first terminus interacts and binds with the target gene at the target DNA-sequence, particularly within the minor groove.
- the molecules disclosed herein provide a polyamide sequence.
- the DNA-binding moiety comprises a polyamide of one or more of the following subunits selected from
- R’ is independently hydrogen, optionally substituted C1-C20 alkyl, optionally substituted C1-C20 heteroalkyl, optionally substituted C1-C20 haloalkyl, or optionally substituted C1-C20 alkylamino; and Z is H, NH2, Ci-Ce alkyl, Ci-Ce haloalkyl, or Ci-Ce alkyl -NH2.
- Z 1 is absent, -O-, or -NH-.
- Z B is N or O
- P3 is 1-10;
- W 2 is optionally substituted Ci-C 2 o alkyl, optionally substituted Ci-C 2 o aminoalkyl, optionally substituted Ci- C 20 haloalkyl, optionally substituted Ci-C 2 o heteroalkyl, or optionally substituted Ci-C 2 o hydroxyalkyl; or W 2 is L'-Z-R 4 ; wherein
- L 1 is Ci-C 2 o alkylene or C 2 -C 2 o heteroalkylene
- R 4 is C1-C3 alkyl, -OR 4b , or -NR 4a R 4b ; wherein
- R 4a is hydrogen, optionally substituted Ci-C 2 o alkyl, or optionally substituted Ci-C 2 o heteroalkyl;
- R 4b is optionally substituted Ci-C 2 o alkyl, optionally substituted C 2 -C 2 o alkenyl, optionally substituted C 2 -C 2 o alkynyl, optionally substituted Ci-C 2 o aminoalkyl, optionally substituted Ci- C 20 haloalkyl, optionally substituted Ci-C 2 o heteroalkyl, optionally substituted Ci-C 2 o hydroxyalkyl, optionally substituted C3-C8 cycloalkyl, optionally substituted 4 to 8-membered heterocycloalkyl, optionally substituted phenyl, or optionally substituted 5 to 10-membered heteroaryl; or R 4a and R 4b together with the nitrogen to which they are attached form an optionally substituted 3 to 10-membered heterocycloalkyl which is partially or fully saturated;
- R w is hydrogen or optionally substituted C1-C20 alkyl
- R w and W 2 combine together with the atoms to which they are attached to form an optionally substituted 4 to 10-membered heterocycloalkyl; each Y 1 , Y 2 , Y 3 , Y 4 , Y 5 , Y 6 , Y 7 , and Y 8 is independently N or CH; each X 1 , X 2 , X 3 , X 4 , X 5 , X 6 , X 7 , and X 8 is independently S, O, or NR 2 ; each R 2 is independently hydrogen, optionally substituted C1-C50 alkyl, optionally substituted C2-C50 alkenyl, optionally substituted C2-C50 alkynyl, optionally substituted C1-C50 heteroalkyl, optionally substituted C2-C50 heteroalkenyl, optionally substituted C2-C50 heteroalkynyl, optionally substituted C1-C50 haloalkyl, optionally substituted C3-C10 cyclo
- R 3e is alkyl, PEG, cycloalkyl, heterocycloalkyl, or phenyl; or two R 3 together with the atoms to which they are attached form an optionally substituted C3-C6 cycloalkyl or 3 to 6-membered heterocycloalkyl; ji is 0 or 1; no is 0 or 1 ; mi and are each independently an integer from 0-3; and pi is 2, 3, or 4, provided that when Z 1 is -O- or -NH- then pi is 2, and when Z 1 is absent then pi is 3 or 4; wherein the linker is attached at one of R 3 and the others of R 3 are each hydrogen; or the linker is attached at the cycloalkyl or heterocycloalkyl formed by two R 3 .
- Z 1 is absent. In some embodiments, Z 1 is -O-. In some embodiments, Z 1 is -NH-.
- the DNA-binding moiety comprises the structure of Formula (A-l), or a pharmaceutically acceptable salt thereof:
- Z B is N or O
- P3 is 1-10;
- W 2 is optionally substituted Ci-C 2 o alkyl, optionally substituted Ci-C 2 o aminoalkyl, optionally substituted Ci- C 20 haloalkyl, optionally substituted Ci-C 2 o heteroalkyl, or optionally substituted Ci-C 2 o hydroxyalkyl; or W 2 is L'-Z-R 4 ; wherein
- L 1 is Ci-C 2 o alkylene or C 2 -C 2 o heteroalkylene
- R 4 is C1-C3 alkyl, -OR 4b , or -NR 4a R 4b ; wherein
- R 4a is hydrogen, optionally substituted Ci-C 2 o alkyl, or optionally substituted Ci-C 2 o heteroalkyl;
- R 4b is optionally substituted Ci-C 2 o alkyl, optionally substituted C 2 -C 2 o alkenyl, optionally substituted C 2 -C 2 o alkynyl, optionally substituted Ci-C 2 o aminoalkyl, optionally substituted Ci- C 20 haloalkyl, optionally substituted Ci-C 2 o heteroalkyl, optionally substituted Ci-C 2 o hydroxyalkyl, optionally substituted C3-C8 cycloalkyl, optionally substituted 4 to 8-membered heterocycloalkyl, optionally substituted phenyl, or optionally substituted 5 to 10-membered heteroaryl; or
- R 4a and R 4b together with the nitrogen to which they are attached form an optionally substituted 3 to 10-membered heterocycloalkyl which is partially or fully saturated;
- R w is hydrogen or optionally substituted C1-C20 alkyl; or
- R w and W 2 combine together with the atoms to which they are attached to form an optionally substituted 4 to 10-membered heterocycloalkyl; each Y 1 , Y 2 , Y 3 , Y 4 , Y 5 , Y 6 , Y 7 , and Y 8 is independently N or CH; each X 1 , X 2 , X 3 , X 4 , X 5 , X 6 , X 7 , and X 8 is independently S, O, or NR 2 ; each R 2 is independently hydrogen, optionally substituted C1-C50 alkyl, optionally substituted C2-C50 alkenyl, optionally substituted C2-C50 alkynyl, optionally substituted C1-C50 heteroalkyl, optionally substituted C2-C50 heteroalkenyl, optionally substituted C2-C50 heteroalkynyl, optionally substituted C1-C50 haloalkyl, optionally substituted C3-C10 cyclo
- R 3c and R 3d are each independently hydrogen, alkyl, or PEG;
- R 3e is alkyl, PEG, cycloalkyl, heterocycloalkyl, or phenyl; or two R 3 together with the atoms to which they are attached form an optionally substituted C3-C6 cycloalkyl or 3 to 6-membered heterocycloalkyl; ji is 0 or 1; no is 0 or 1 ; mi and are each independently an integer from 0-3; and pi is 3 or 4; wherein the linker is attached at one of R 3 and the others of R 3 are each hydrogen; or the linker is attached at the cycloalkyl or heterocycloalkyl formed by two R 3 .
- each R 3 is independently hydrogen, halogen, Ci-Ce alkyl, -OR 3c , -NR 3c R 3d , or -NHC(O)R 3e , wherein R 3c and R 3d are each independently hydrogen, alkyl, or PEG; and R 3e is alkyl, PEG, cycloalkyl, heterocycloalkyl, or phenyl.
- each R 3 is independently -NR 3c R 3d .
- each R 3 is independently -NHC(O)R 3e .
- each R 3 is independently hydrogen.
- two R 3 together with the atoms to which they are attached form an optionally substituted C3-C6 cycloalkyl or 3 to 6-membered heterocycloalkyl.
- two R 3 together with the atom(s) to which they are attached form a C3-C6 cycloalkyl.
- two R 3 together with the atom(s) to which they are attached form a 4 to 6-membered heterocycloalkyl.
- two R 3 together with the atom(s) to which they are attached form a 4-membered heterocycloalkyl.
- two R 3 together with the atom(s) to which they are attached form a 5 -membered heterocycloalkyl. In some embodiments, two R 3 together with the atom(s) to which they are attached form a 6-membered heterocycloalkyl. In some embodiments, two R 3 together with the atom(s) to which they are attached form a cyclopropyl, cyclobutyl, or cyclopentyl.
- no is 1. In some embodiments, no is 0.
- the DNA-binding moiety comprises the structure of Formula (A-2), or a pharmaceutically acceptable salt thereof:
- W 2 is optionally substituted C1-C20 alkyl, optionally substituted C1-C20 aminoalkyl, optionally substituted Ci- C20 haloalkyl, optionally substituted C1-C20 heteroalkyl, or optionally substituted C1-C20 hydroxyalkyl; or W 2 is L’-Z-R 4 ; wherein
- L 1 is C1-C20 alkylene or C2-C20 heteroalkylene
- R 4 is C1-C3 alkyl, -OR 4b , or -NR 4a R 4b ; wherein
- R 4a is hydrogen, optionally substituted C1-C20 alkyl, or optionally substituted C1-C20 heteroalkyl;
- R 4b is optionally substituted C1-C20 alkyl, optionally substituted C2-C20 alkenyl, optionally substituted C2-C20 alkynyl, optionally substituted C1-C20 aminoalkyl, optionally substituted C1-C20 haloalkyl, optionally substituted C1-C20 heteroalkyl, optionally substituted C1-C20 hydroxyalkyl, optionally substituted C’s-Cs cycloalkyl, optionally substituted 4 to 8- membered heterocycloalkyl, optionally substituted phenyl, or optionally substituted 5 to 10- membered heteroaryl; or
- R 4a and R 4b together with the nitrogen to which they are attached form an optionally substituted 3 to 10-membered heterocycloalkyl which is partially or fully saturated;
- R w is hydrogen or optionally substituted C1-C20 alkyl
- R w and W 2 combine together with the atoms to which they are attached to form an optionally substituted 4 to 10-membered heterocycloalkyl; each Y 1 , Y 2 , Y 3 , Y 4 , Y 5 , Y 6 , Y 7 , and Y 8 is independently N or CH; each X 1 , X 2 , X 3 , X 4 , X 5 , X 6 , X 7 , and X 8 is independently S, O, or NR 2 ; each R 2 is independently hydrogen, optionally substituted C1-C50 alkyl, optionally substituted C2-C50 alkenyl, optionally substituted C2-C50 alkynyl, optionally substituted C1-C50 heteroalkyl, optionally substituted C2-C50 heteroalkenyl, optionally substituted C2-C50 heteroalkynyl, optionally substituted C1-C50 haloalkyl, optionally substituted C3-C10 cyclo
- R 3e is alkyl, PEG, cycloalkyl, heterocycloalkyl, or phenyl; or two R 3a or two R 3b together with the carbon atom to which they are attached form a CAC, cycloalkyl or 4 to 6-membered heterocycloalkyl; ji is 0 or 1; and mi and are each independently an integer from 0-3; wherein the linker is attached at R 3a or R 3b ; or at the cycloalkyl or heterocycloalkyl formed through the combination of two R 3a or two R 3b .
- each X 1 , X 2 , X 3 , X 4 , X 5 , X 6 , and X 7 is independently -NR 2 .
- each X 1 , X 2 , X 3 , X 4 , X 5 , X 6 , X 7 , and X 8 is independently O.
- each X 1 , X 2 , X 3 , X 4 , X 5 , X 6 , X 7 , and X 8 is independently S.
- each R 2 is independently hydrogen, optionally substituted C1-C50 alkyl, optionally substituted C2-C50 alkenyl, optionally substituted C2-C50 alkynyl, optionally substituted C1-C50 heteroalkyl, optionally substituted C2-C50 heteroalkenyl, optionally substituted C2-C50 heteroalkynyl, optionally substituted C1-C50 haloalkyl, optionally substituted C3-C10 cycloalkyl, optionally substituted 3 to 10-membered heterocycloalkyl, or optionally substituted PEG1.50.
- each R 2 is independently hydrogen, optionally substituted C1-C20 alkyl, optionally substituted C2-C20 alkenyl, optionally substituted C2-C20 alkynyl, optionally substituted C1-C20 heteroalkyl, optionally substituted C2-C20 heteroalkenyl, optionally substituted C2-C20 heteroalkynyl, optionally substituted C1-C20 haloalkyl, optionally substituted C3-C8 cycloalkyl, optionally substituted 3 to 8-membered heterocycloalkyl, or optionally substituted PEG1.20.
- each R 2 is independently hydrogen, optionally substituted C1-C10 alkyl, optionally substituted C1-C10 heteroalkyl, optionally substituted C1-C10 haloalkyl, optionally substituted C3-C8 cycloalkyl, optionally substituted 3 to 8-membered heterocycloalkyl, or optionally substituted PEG1 10.
- each R 2 is independently an optionally substituted C1-C20 alkyl. In some embodiments, each R 2 is independently an optionally substituted C1-C10 alkyl. In some embodiments, each R 2 is independently methyl, ethyl, isopropyl, isobutyl, sec-butyl, or tertbutyl. In some embodiments, each R 2 is independently hydrogen or methyl. In some embodiments, each R 2 is ethyl. In some embodiments, each R 2 is isopropyl. In some embodiments, each R 2 is methyl. In some embodiments, each R 2 is hydrogen.
- the DNA-binding moiety comprises a structure of Formula (A-3), or a pharmaceutically acceptable salt thereof:
- W 2 is optionally substituted C1-C20 alkyl, optionally substituted C1-C20 aminoalkyl, optionally substituted Ci- C20 haloalkyl, optionally substituted C1-C20 heteroalkyl, or optionally substituted C1-C20 hydroxyalkyl; or W 2 is L'-Z-R 4 ; wherein
- L 1 is C1-C20 alkylene or C2-C20 heteroalkylene
- R 4 is C1-C3 alkyl, -OR 4b , or -NR 4a R 4b ; wherein
- R 4a is hydrogen, optionally substituted C1-C20 alkyl, or optionally substituted C1-C20 heteroalkyl;
- R 4b is optionally substituted C1-C20 alkyl, optionally substituted C2-C20 alkenyl, optionally substituted C2-C20 alkynyl, optionally substituted C1-C20 aminoalkyl, optionally substituted C1-C20 haloalkyl, optionally substituted C1-C20 heteroalkyl, optionally substituted C1-C20 hydroxyalkyl, optionally substituted C’s-Cs cycloalkyl, optionally substituted 4 to 8- membered heterocycloalkyl, optionally substituted phenyl, or optionally substituted 5 to 10- membered heteroaryl; or
- R 4a and R 4b together with the nitrogen to which they are attached form an optionally substituted 3 to 10-membered heterocycloalkyl which is partially or fully saturated;
- R w is hydrogen or optionally substituted C1-C20 alkyl; or R w and W 2 combine together with the atoms to which they are attached to form an optionally substituted 4 to 10-membered heterocycloalkyl; each Y 1 , Y 2 , Y 3 , Y 4 , Y 5 , Y 6 , Y 7 , and Y 8 is independently N or CH; each R 2a , R 2b , R 2c , R 2d , R 2e , R 2f , R 2g , and R 2h is independently hydrogen, optionally substituted C1-C50 alkyl, optionally substituted C2-C50 alkenyl, optionally substituted C2-C50 alkynyl, optionally substituted C1-C50 heteroalkyl, optionally substituted C2-C50 heteroalkenyl, optionally substituted C2-C50 heteroalkynyl, optionally substituted C1-C50 haloalkyl, optionally substituted C3-
- R Za and R zb are each independently hydrogen, alkyl, or PEG;
- R Zc is alkyl, PEG, cycloalkyl, heterocycloalkyl, or phenyl; each R 3b is independently hydrogen, halogen, Ci-Cg alkyl, -OR 3c , -NR 3c R 3d , or -NHC(O)R 3e , wherein
- R 3c and R 3d are each independently hydrogen, alkyl, or PEG;
- R 3e is alkyl, PEG, cycloalkyl, heterocycloalkyl, or phenyl; or two R 3b together with the carbon atom to which they are attached form a C3-C6 cycloalkyl or 4 to 6- membered heterocycloalkyl; and and mi are each independently 0 or 1.
- the DNA-binding moiety comprises a structure of Formula (A-4), or a pharmaceutically acceptable salt thereof:
- W 2 is optionally substituted C1-C20 alkyl, optionally substituted C1-C20 aminoalkyl, optionally substituted Ci- C20 haloalkyl, optionally substituted C1-C20 heteroalkyl, or optionally substituted C1-C20 hydroxyalkyl; or W 2 is L'-Z-R 4 ; wherein
- L 1 is C1-C20 alkylene or C2-C20 heteroalkylene
- R 4 is C1-C3 alkyl, -OR 4b , or -NR 4a R 4b ; wherein R 4a is hydrogen, optionally substituted C1-C20 alkyl, or optionally substituted C1-C20 heteroalkyl;
- R 4b is optionally substituted C1-C20 alkyl, optionally substituted C2-C20 alkenyl, optionally substituted C2-C20 alkynyl, optionally substituted C1-C20 aminoalkyl, optionally substituted C1-C20 haloalkyl, optionally substituted C1-C20 heteroalkyl, optionally substituted C1-C20 hydroxyalkyl, optionally substituted Cs-Cs cycloalkyl, optionally substituted 4 to 8- membered heterocycloalkyl, optionally substituted phenyl, or optionally substituted 5 to 10- membered heteroaryl; or
- R 4a and R 4b together with the nitrogen to which they are attached form an optionally substituted 3 to 10-membered heterocycloalkyl which is partially or fully saturated;
- R w is hydrogen or optionally substituted C1-C20 alkyl; or R w and W 2 combine together with the atoms to which they are attached to form an optionally substituted 4 to 10-membered heterocycloalkyl; each Y 1 , Y 2 , Y 3 , Y 4 , Y 5 , Y 6 , Y 7 , and Y 8 is independently N or CH; each R 2a , R 2b , R 2c , R 2d , R 2e , R 2f , R 2g , and R 2h is independently hydrogen, optionally substituted C1-C50 alkyl, optionally substituted C2-C50 alkenyl, optionally substituted C2-C50 alkynyl, optionally substituted C1-C50 heteroalkyl, optionally substituted C2-C50 heteroalkenyl, optionally substituted C2-C50 heteroalkynyl, optionally substituted C1-C50 haloalkyl, optionally substituted C3-
- R Za and R zb are each independently hydrogen, alkyl, or PEG;
- R Zc is alkyl, PEG, cycloalkyl, heterocycloalkyl, or phenyl; each R 3a is independently hydrogen, halogen, Ci-Cg alkyl, -OR 3c , -NR 3c R 3d , or -NHC(O)R 3e , wherein
- R 3c and R 3d are each independently hydrogen, alkyl, or PEG;
- R 3e is alkyl, PEG, cycloalkyl, heterocycloalkyl, or phenyl; or two R 3a together with the carbon atom to which they are attached form a C3-C6 cycloalkyl or 4 to 6- membered heterocycloalkyl; and ni and mi are each independently 0 or 1.
- the DNA-binding moiety comprises a structure of Formula (A-5), or a pharmaceutically acceptable salt thereof:
- W 2 is optionally substituted C1-C20 alkyl, optionally substituted C1-C20 aminoalkyl, optionally substituted Ci- C20 haloalkyl, optionally substituted C1-C20 heteroalkyl, or optionally substituted C1-C20 hydroxyalkyl; or W 2 is L'-Z-R 4 ; wherein
- L 1 is C1-C20 alkylene or C2-C20 heteroalkylene
- R 4 is C1-C3 alkyl, -OR 4b , or -NR 4a R 4b ; wherein
- R 4a is hydrogen, optionally substituted C1-C20 alkyl, or optionally substituted C1-C20 heteroalkyl;
- R 4b is optionally substituted C1-C20 alkyl, optionally substituted C2-C20 alkenyl, optionally substituted C2-C20 alkynyl, optionally substituted C1-C20 aminoalkyl, optionally substituted C1-C20 haloalkyl, optionally substituted C1-C20 heteroalkyl, optionally substituted C1-C20 hydroxyalkyl, optionally substituted C’s-Cs cycloalkyl, optionally substituted 4 to 8- membered heterocycloalkyl, optionally substituted phenyl, or optionally substituted 5 to 10- membered heteroaryl; or
- R 4a and R 4b together with the nitrogen to which they are attached form an optionally substituted 3 to 10-membered heterocycloalkyl which is partially or fully saturated;
- R w is hydrogen or optionally substituted C1-C20 alkyl; or R w and W 2 combine together with the atoms to which they are attached to form an optionally substituted 4 to 10-membered heterocycloalkyl; each Y 5 and Y 6 is independently N or CH; each R 2a , R 2b , R 2c , R 2d , R 2e , R 2f , R 2g , and R 2h is independently hydrogen, optionally substituted C1-C50 alkyl, optionally substituted C2-C50 alkenyl, optionally substituted C2-C50 alkynyl, optionally substituted C1-C50 heteroalkyl, optionally substituted C2-C50 heteroalkenyl, optionally substituted C2-C50 heteroalkynyl, optionally substituted C1-C50 haloalkyl, optionally substituted C3-C10 cycloalkyl, optionally substituted 3 to 10-membered heterocycloalkyl, or optionally substituted P
- R Za and R zb are each independently hydrogen, alkyl, or PEG;
- R Zc is alkyl, PEG, cycloalkyl, heterocycloalkyl, or phenyl; and ni and mi are each independently 0 or 1.
- the DNA-binding moiety comprises a structure of Formula (A-6), or a pharmaceutically acceptable salt thereof:
- L 1 is C1-C20 alkylene or C2-C20 heteroalkylene
- R 4 is C1-C3 alkyl, -OR 4b , or -NR 4a R 4b ; wherein
- R 4a is hydrogen, optionally substituted C1-C20 alkyl, or optionally substituted C1-C20 heteroalkyl;
- R 4b is optionally substituted C1-C20 alkyl, optionally substituted C2-C20 alkenyl, optionally substituted C2-C20 alkynyl, optionally substituted C1-C20 aminoalkyl, optionally substituted C1-C20 haloalkyl, optionally substituted C1-C20 heteroalkyl, optionally substituted C1-C20 hydroxyalkyl, optionally substituted C’s-Cs cycloalkyl, optionally substituted 4 to 8- membered heterocycloalkyl, optionally substituted phenyl, or optionally substituted 5 to 10- membered heteroaryl; or R 4a and R 4b together with the nitrogen to which they are attached form an optionally substituted 3 to 10-membered heterocycloalkyl which is partially or fully saturated;
- R w is hydrogen or optionally substituted C1-C20 alkyl; or R w and W 2 combine together with the atoms to which they are attached to form an optionally substituted 4 to 10-membered heterocycloalkyl; each Y 5 and Y 6 is independently N or CH; each R 2a , R 2b , R 2c , R 2d , R 2e , R 2f , R 2g , and R 2h is independently hydrogen, optionally substituted C1-C50 alkyl, optionally substituted C2-C50 alkenyl, optionally substituted C2-C50 alkynyl, optionally substituted C1-C50 heteroalkyl, optionally substituted C2-C50 heteroalkenyl, optionally substituted C2-C50 heteroalkynyl, optionally substituted C1-C50 haloalkyl, optionally substituted C3-C10 cycloalkyl, optionally substituted 3 to 10-membered heterocycloalkyl, or optionally substituted P
- R Za and R zb are each independently hydrogen, alkyl, or PEG;
- R Zc is alkyl, PEG, cycloalkyl, heterocycloalkyl, or phenyl; and and mi are each independently 0 or 1.
- W 2 is optionally substituted Ci- C20 alkyl, optionally substituted C1-C20 aminoalkyl, optionally substituted C1-C20 haloalkyl, optionally substituted C1-C20 heteroalkyl, or optionally substituted C1-C20 hydroxyalkyl.
- W 2 is optionally substituted C1-C20 alkyl or optionally substituted C1-C20 heteroalkyl.
- W 2 is optionally substituted C1-C20 alkyl.
- W 2 is optionally substituted C1-C20 heteroalkyl.
- W 2 is methyl, ethyl, n-propyl, or isopropyl.
- W 2 is L’-Z-R 4 .
- R w is hydrogen. In some embodiments, R w is optionally substituted C1-C20 alkyl.
- W 2 and R w together with the nitrogen to which they are attached form an optionally substituted 4 to 10-membered heterocycloalkyl.
- W 2 and R w together with the nitrogen to which they are attached form an optionally substituted 4-membered heterocycloalkyl.
- W 2 and R w together with the nitrogen to which they are attached form an optionally substituted 5 -membered heterocycloalkyl.
- W 2 and R w together with the nitrogen to which they are attached form an optionally substituted 6-membered heterocycloalkyl.
- W 2 and R w together with the nitrogen to which they are attached form an optionally substituted 7-membered heterocycloalkyl.
- the DNA-binding moiety comprises a structure of Formula (A-7), or a pharmaceutically acceptable salt thereof:
- L 1 is C1-C20 alkylene or C2-C20 heteroalkylene
- R 4 is C1-C3 alkyl, -OR 4b , or -NR 4a R 4b ; wherein
- R 4a is hydrogen, optionally substituted C1-C20 alkyl, or optionally substituted C1-C20 heteroalkyl;
- R 4b is optionally substituted C1-C20 alkyl, optionally substituted C2-C20 alkenyl, optionally substituted C2-C20 alkynyl, optionally substituted C1-C20 aminoalkyl, optionally substituted C1-C20 haloalkyl, optionally substituted C1-C20 heteroalkyl, optionally substituted C1-C20 hydroxyalkyl, optionally substituted Cs-Cs cycloalkyl, optionally substituted 4 to 8-membered heterocycloalkyl, optionally substituted phenyl, or optionally substituted 5 to 10-membered heteroaryl; or
- R 4a and R 4b together with the nitrogen to which they are attached form an optionally substituted 3 to 10- membered heterocycloalkyl which is partially or fully saturated; each R z is independently -CN, -OH, -OR Za , -N 3 , -NR Za R zb , -CO(O)R Zc , -C(O)OR Zc , -C(O)NR Za R zb , - NHC(O)R Zc , -NHC(O)OR Zc , -OC(O)NR Za R zb , or optionally substituted 5 to 10-membered heteroaryl; wherein
- R Za and R zb are each independently hydrogen, alkyl, or PEG;
- R Zc is alkyl, PEG, cycloalkyl, heterocycloalkyl, or phenyl; each R 3b is independently hydrogen, halogen, Ci-Ce alkyl, -NR 3c R 3d , or -NHC(O)R 3e , wherein
- R 3c and R 3d are each independently hydrogen, alkyl, or PEG;
- R 3e is alkyl, PEG, cycloalkyl, heterocycloalkyl, or phenyl; or two R 3b together with the carbon atom to which they are attached form a C’s-C,, cycloalkyl or 4 to 6- membered heterocycloalkyl; and ni and mi are each independently 0 or 1.
- the DNA-binding moiety comprises a structure of Formula (A-8), or a pharmaceutically acceptable salt thereof:
- L 1 is C1-C20 alkylene or C2-C20 heteroalkylene
- R 4 is C1-C3 alkyl, -OR 4b , or -NR 4a R 4b ; wherein
- R 4a is hydrogen, optionally substituted C1-C20 alkyl, or optionally substituted C1-C20 heteroalkyl;
- R 4b is optionally substituted C1-C20 alkyl, optionally substituted C2-C20 alkenyl, optionally substituted C2-C20 alkynyl, optionally substituted C1-C20 aminoalkyl, optionally substituted C1-C20 haloalkyl, optionally substituted C1-C20 heteroalkyl, optionally substituted C1-C20 hydroxyalkyl, optionally substituted Cs-Cs cycloalkyl, optionally substituted 4 to 8-membered heterocycloalkyl, optionally substituted phenyl, or optionally substituted 5 to 10-membered heteroaryl; or
- R 4a and R 4b together with the nitrogen to which they are attached form an optionally substituted 3 to 10- membered heterocycloalkyl which is partially or fully saturated; each R z is independently -CN, -OH, -OR Za , -Ns, -NR Za R zb , -CO(O)R Zc , -C(O)OR Zc , -C(O)NR Za R zb , - NHC(O)R Zc , -NHC(O)OR Zc , -OC(O)NR Za R zb , or optionally substituted 5 to 10-membered heteroaryl; wherein
- R Za and R zb are each independently hydrogen, alkyl, or PEG;
- R Zc is alkyl, PEG, cycloalkyl, heterocycloalkyl, or phenyl; each R 3a is independently hydrogen, halogen, Ci-Cg alkyl, -NR 3c R 3d , or -NHC(O)R 3e , wherein
- R 3c and R 3d are each independently hydrogen, alkyl, or PEG;
- R 3e is alkyl, PEG, cycloalkyl, heterocycloalkyl, or phenyl; or two R 3a together with the carbon atom to which they are attached form a Cs-Ce cycloalkyl or 4 to 6- membered heterocycloalkyl; and and mi are each independently 0 or 1.
- the DNA-binding moiety comprises a structure of Formula (A-9), or a pharmaceutically acceptable salt thereof:
- L 1 is C1-C20 alkylene or C2-C20 heteroalkylene
- R 4 is C1-C3 alkyl, -OR 4b , or -NR 4a R 4b ; wherein
- R 4a is hydrogen, optionally substituted C1-C20 alkyl, or optionally substituted C1-C20 heteroalkyl;
- R 4b is optionally substituted C1-C20 alkyl, optionally substituted C2-C20 alkenyl, optionally substituted C2-C20 alkynyl, optionally substituted C1-C20 aminoalkyl, optionally substituted C1-C20 haloalkyl, optionally substituted C1-C20 heteroalkyl, optionally substituted C1-C20 hydroxyalkyl, optionally substituted Ci-C’s cycloalkyl, optionally substituted 4 to 8-membered heterocycloalkyl, optionally substituted phenyl, or optionally substituted 5 to 10-membered heteroaryl; or
- R 4a and R 4b together with the nitrogen to which they are attached form an optionally substituted 3 to 10- membered heterocycloalkyl which is partially or fully saturated; each R z is independently -CN, -OH, -OR Za , -N 3 , -NR Za R zb , -CO(O)R Zc , -C(O)OR Zc , -C(O)NR Za R zb , - NHC(O)R Zc , -NHC(O)OR Zc , -OC(O)NR Za R zb , or optionally substituted 5 to 10-membered heteroaryl; wherein
- R Za and R zb are each independently hydrogen, alkyl, or PEG;
- R Zc is alkyl, PEG, cycloalkyl, heterocycloalkyl, or phenyl; and and mi are each independently 0 or 1.
- the DNA-binding moiety comprises a structure of Formula (A- 10), or a pharmaceutically acceptable salt thereof:
- L 1 is C1-C20 alkylene or C2-C20 heteroalkylene
- R 4 is C1-C3 alkyl, -OR 4b , or -NR 4a R 4b ; wherein
- R 4a is hydrogen, optionally substituted C1-C20 alkyl, or optionally substituted C1-C20 heteroalkyl;
- R 4b is optionally substituted C1-C20 alkyl, optionally substituted C2-C20 alkenyl, optionally substituted C2-C20 alkynyl, optionally substituted C1-C20 aminoalkyl, optionally substituted C1-C20 haloalkyl, optionally substituted C1-C20 heteroalkyl, optionally substituted C1-C20 hydroxyalkyl, optionally substituted C3-C8 cycloalkyl, optionally substituted 4 to 8-membered heterocycloalkyl, optionally substituted phenyl, or optionally substituted 5 to 10-membered heteroaryl; or
- R 4a and R 4b together with the nitrogen to which they are attached form an optionally substituted 3 to 10- membered heterocycloalkyl which is partially or fully saturated; each R z is independently -CN, -OH, -OR Za , -N3, -NR Za R zb , -CO(O)R Zc , -C(O)OR Zc , -C(O)NR Za R zb , - NHC(O)R Zc , -NHC(O)OR Zc , -OC(O)NR Za R zb , or optionally substituted 5 to 10-membered heteroaryl; wherein
- R Za and R zb are each independently hydrogen, alkyl, or PEG;
- R Zc is alkyl, PEG, cycloalkyl, heterocycloalkyl, or phenyl; and and mi are each independently 0 or 1.
- Y 2 , Y 4 , and Y 7 are each independently N; and Y 1 and Y 3 are each independently CH.
- each Y 5 is independently CH. In some embodiments, each Y 5 is independently N.
- each Y 6 is independently CH. In some embodiments, each Y 6 is independently N.
- Y 8 is CH. In some embodiments, Y 8 is N.
- each R 2a , R 2b , R 2c , R 2d , R 2e , R 2f , R 2g , and R 2h is independently hydrogen, optionally substituted C1-C50 alkyl, optionally substituted C2-C50 alkenyl, optionally substituted C2-C50 alkynyl, optionally substituted C1-C50 heteroalkyl, optionally substituted C2-C50 heteroalkenyl, optionally substituted C2-C50 heteroalkynyl, optionally substituted C1-C50 haloalkyl, optionally substituted C3-C10 cycloalkyl, optionally substituted 3 to 10-membered heterocycloalkyl, or optionally substituted PEG1.50; each of which is optionally substituted with one or more R z .
- each R 2a , R 2b , R 2c , R 2d , R 2e , R 2f , R 2g , and R 2h is independently hydrogen, optionally substituted C1-C50 alkyl, optionally substituted C2-C50 alkenyl, optionally substituted C2-C50 alkynyl, optionally substituted C1-C50 heteroalkyl, optionally substituted C2-C50 heteroalkenyl, optionally substituted C2-C50 heteroalkynyl, optionally substituted C1-C50 haloalkyl, optionally substituted C3-G cycloalkyl, optionally substituted 3 to 8-membered heterocycloalkyl, or optionally substituted PEG1.50.
- each R 2a , R 2b , R 2c , R 2d , R 2e , R 2f , R 2g , and R 2h is independently hydrogen, optionally substituted C1-C20 alkyl, optionally substituted C2-C20 alkenyl, optionally substituted C2-C20 alkynyl, optionally substituted C1-C20 heteroalkyl, optionally substituted C2-C20 heteroalkenyl, optionally substituted C2-C20 heteroalkynyl, optionally substituted C1-C20 haloalkyl, optionally substituted Cs-Cs cycloalkyl, optionally substituted 3 to 8-membered heterocycloalkyl, or optionally substituted PEGI.20.
- each R 2a , R 2b , R 2c , R 2d , R 2e , R 2f , R 2g , and R 2h is independently optionally substituted C1-C10 alkyl, optionally substituted C1-C10 heteroalkyl, optionally substituted C1-C10 haloalkyl, optionally substituted Cs-Cs cycloalkyl, optionally substituted 3 to 8-membered heterocycloalkyl, or optionally substituted PEG1 10.
- each R 2a , R 2b , R 2c , R 2d , R 2e , R 2f , R 2g , and R 2h is independently optionally substituted C1-C10 alkyl, optionally substituted C1-C10 haloalkyl, optionally substituted Cs-Cs cycloalkyl, or optionally substituted 3 to 8-membered heterocycloalkyl.
- each R 2a , R 2b , R 2c , R 2d , R 2e , R 2f , R 2g , and R 2h is independently optionally substituted C1-C10 alkyl or optionally substituted Cs-Cs cycloalkyl.
- the cycloalkyl or heterocycloalkyl is monocyclic, bicyclic, or spirocyclic.
- each R 2a , R 2b , R 2c , R 2d , R 2e , R 2f , R 2g , and R 2h is independently an optionally substituted C1-C20 alkyl.
- each R 2a , R 2b , R 2c , R 2d , R 2e , R 2f , R 2g , and R 2h is independently an optionally substituted C1-C10 alkyl.
- each R 2a , R 2b , R 2c , R 2d , R 2e , R 2f , R 2g , and R 2h is independently methyl, ethyl, isopropyl, isobutyl, sec-butyl, or tert-butyl.
- each R 2 is independently hydrogen or methyl.
- each R 2a , R 2b , R 2c , R 2d , R 2e , R 2f , R 2g , and R 2h is ethyl.
- each R 2a , R 2b , R 2c , R 2d , R 2e , R 2f , R 2g , and R 2h is isopropyl. In some embodiments, each R 2a , R 2b , R 2c , R 2d , R 2e , R 2f , R 2g , and R 2h is methyl. In some embodiments, each R 2a , R 2b , R 2c , R 2d , R 2e , R 2f , R 2g , and R 2h is hydrogen.
- each R 3a is independently hydrogen, halogen, -OR 3c , -NR 3c R 3d , or -NHC(O)R 3e , wherein R 3c and R 3d are each independently hydrogen, alkyl, or PEG; and R 3e is alkyl, PEG, cycloalkyl, heterocycloalkyl, or phenyl.
- each R 3a is independently -NR 3c R 3d .
- each R 3a is independently -NHC(O)R 3e .
- each R 3a is -NH2.
- each R 3a is hydrogen.
- each R 3b is independently hydrogen, halogen, -OR 3c , -NR 3c R 3d , or -NHC(O)R 3e , wherein R 3c and R 3d are each independently hydrogen, alkyl, or PEG; and R 3e is alkyl, PEG, cycloalkyl, heterocycloalkyl, or phenyl.
- each R 3b is independently -NR 3c R 3d .
- each R 3b is independently -NHC(O)R 3e .
- each R 3b is -NH 2 .
- each R 3b is hydrogen.
- two R 3a together with the atoms to which they are attached combine together to form an optionally substituted C3-C, cycloalkyl or 3 to 6-membered heterocycloalkyl.
- two R 3a together with the atom(s) to which they are attached form a C’s-C,, cycloalkyl.
- two R 3a together with the atom(s) to which they are attached form a 4 to 6-membered heterocycloalkyl.
- two R 3a together with the atom(s) to which they are attached form a 4-membered heterocycloalkyl.
- two R 3a together with the atom(s) to which they are attached form a 5 -membered heterocycloalkyl. In some embodiments, two R 3a together with the atom(s) to which they are attached form a 6-membered heterocycloalkyl. In some embodiments, two R 3a together with the atom(s) to which they are attached form a cyclopropyl, cyclobutyl, or cyclopentyl.
- two R 3b together with the atoms to which they are attached combine together to form an optionally substituted Cs-Ce cycloalkyl or 3 to 6-membered heterocycloalkyl.
- two R 3b together with the atom(s) to which they are attached form a C’s-C,, cycloalkyl.
- two R 3b together with the atom(s) to which they are attached form a 4 to 6-membered heterocycloalkyl.
- two R 3b together with the atom(s) to which they are attached form a 4-membered heterocycloalkyl.
- two R 3b together with the atom(s) to which they are attached form a 5 -membered heterocycloalkyl. In some embodiments, two R 3b together with the atom(s) to which they are attached form a 6-membered heterocycloalkyl. In some embodiments, two R 3b together with the atom(s) to which they are attached form a cyclopropyl, cyclobutyl, or cyclopentyl.
- the linker is attached at R 3a .
- the linker is attached at R 3b .
- the linker is attached at the cycloalkyl or heterocycloalkyl formed through the combination of two R 3a or two R 3b
- R 3c and R 3d are each independently hydrogen, alkyl, or PEG. In some embodiments, R 3c and R 3d are each independently hydrogen, Ci-C2oalkyl, or PEG 1-20. In some embodiments, R and R are each independently Ci- C2oalkyl. In some embodiments, R 3c and R 3d are each independently hydrogen.
- R 3e is alkyl, PEG, cycloalkyl, heterocycloalkyl, or phenyl.
- R 3e is C1-C20 alkyl, PEG1.20, C3-C, cycloalkyl, 4 to 6-membered heterocycloalkyl, or phenyl.
- R 3e is C1-C20 alkyl or PEGi. 20.
- R 3e is C3-C, cycloalkyl, 4 to 6-membered heterocycloalkyl, or phenyl.
- R 3e is C1-C20 alkyl.
- L 1 is C1-C10 alkylene or C2-C10 heteroalkylene.
- L 1 is C1-C10 alkylene, Ci-Cs alkylene, Ci-Ce alkylene, C1-C5 alkylene, C1-C4 alkylene, C1-C3 alkylene, or C1-C2 alkylene.
- L 1 is C1-C4 alkylene.
- L 1 is C1-C3 alkylene.
- L 1 is C1-C2 alkylene.
- L 1 is C2-C10 heteroalkylene, C2-C8 heteroalkylene, C2-C6 heterolkylene, C2-C5 heteroalkylene, or C2-C4 heteroalkylene. In some embodiments, L 1 is C2-C10 heteroalkylene. In some embodiments, L 1 is C2-C8 heteroalkylene. In some embodiments, L 1 is C2-C6 heterolkylene. In some embodiments, L 1 is C2-C5 heteroalkylene. In some embodiments, L 1 is C2-C4 heteroalkylene.
- the heteroalkylene is polyethylene glycol.
- L 1 is PEG1.10.
- L 1 is PEGi s.
- L 1 is -(CIECFE-C ys-, wherein ys is an integer in the range of 1-10.
- ys is an integer in the range of 1-8.
- ys is an integer in the range of 1-6.
- ys is an integer in the range of 1-4.
- ys is 1-2.
- the heteroalkylene comprises - (CH 2 ) y 6N(R a )(CH2)y7- , wherein R a is hydrogen or an optionally substituted Ci-Ce alkyl; and each and y 7 is independently an integer in the range of 1-6.
- Z is -C(O)-; and R 4 is -OR 4b .
- Z is -C(O)-; and R 4 is -NR 4a R 4b .
- Z is absent; and R 4 is -OR 4b .
- Z is absent; and R 4 is - NR 4a R 4b .
- Z is -C(O)-; and R 4 is Ci-Ce alkyl.
- R 4 is C1-C3 alkyl or -NR 4a R 4b . In some embodiments, R 4 is C1-C3 alkyl. In some embodiments, R 4 is -NR 4a R 4b . In some embodiments, R 4 is - OR 4b .
- R 4a is hydrogen, optionally substituted C1-C20 alkyl, or optionally substituted C1-C20 heteroalkyl. In some embodiments, R 4a is an optionally substituted C1-C20 alkyl or optionally substituted C1-C20 heteroalkyl. In some embodiments, R 4a is an optionally substituted C1-C20 alkyl. In some embodiments, R 4a is an optionally substituted C1-C20 heteroalkyl. In some embodiments, R 4a is hydrogen.
- R 4b is optionally substituted Ci- C20 alkyl, optionally substituted C2-C20 alkenyl, optionally substituted C2-C20 alkynyl, optionally substituted C1-C20 aminoalkyl, optionally substituted C1-C20 haloalkyl, optionally substituted C1-C20 heteroalkyl, optionally substituted C1-C20 hydroxyalkyl, optionally substituted C’s-Cs cycloalkyl, optionally substituted 4 to 8-membered heterocycloalkyl, optionally substituted phenyl, or optionally substituted 5 to 10-membered heteroaryl.
- R 4b is optionally substituted C1-C20 alkyl, optionally substituted C1-C20 aminoalkyl, optionally substituted C1-C20 haloalkyl optionally substituted C1-C20 heteroalkyl, or optionally substituted C1-C20 hydroxyalkyl. In some embodiments, R 4b is optionally substituted C1-C20 alkyl or optionally substituted C1-C20 heteroalkyl. In some embodiments, R 4b is optionally substituted C1-C10 alkyl. In some embodiments, R 4b is optionally substituted C1-C10 heteroalkyl. In some embodiments, R 4b is optionally substituted C1-C10 haloalkyl.
- R 4a is hydrogen; and R 4b is C1-C10 alkyl or C1-C10 haloalkyl. In some embodiments, R 4a is hydrogen; and R 4b is C1-C10 alkyl. In some embodiments of, R 4a is hydrogen; and R 4b is C1-C10 alkyl or C1-C10 haloalkyl. In some embodiments, R 4a is hydrogen; and R 4b is methyl or ethyl.
- R 4a is C1-C10 alkyl and R 4b is Ci- Cio alkyl. In some embodiments of any of Formula (A-T) through (A-10), R 4a is methyl and R 4b is methyl. [0069] In some embodiments of any of Formula (A-T) through (A-10), R 4a and R 4b together with the nitrogen to which they are attached form an optionally substituted 4 to 10-membered heterocycloalkyl which is partially or fully saturated.
- R 4a and R 4b together with the nitrogen to which they are attached form an optionally substituted 4 to 10-membered heterocycloalkyl, wherein the heterocycloalkyl is a monocyclic, bicyclic, or spirocyclic heterocycloalkyl.
- R 4a and R 4b together with the nitrogen to which they are attached form an optionally substituted 4-membered heterocycloalkyl.
- R 4a and R 4b together with the nitrogen to which they are attached form an optionally substituted 5 -membered heterocycloalkyl.
- R 4a and R 4b together with the nitrogen to which they are attached form an optionally substituted 6-membered heterocycloalkyl.
- R 4a and R 4b together with the nitrogen to which they are attached form a piperidine, piperazine, or morpholine. In some embodiments, R 4a and R 4b together with the nitrogen to which they are attached form a piperidine. In some embodiments, R 4a and R 4b together with the nitrogen to which they are attached form a piperazine.
- R 4a is hydrogen or Ci-Cw alkyl and R 4b is Ci-Cw alkyl; or R 4a and R 4b together with the nitrogen to which they are attached form a 4 to 6- membered heterocycloalkyl.
- each R z is independently -CN, -OH, -OR Za , -N 3 , -NR Za R zb , -CO(O)R Zc , -C(O)OR Zc , -C(O)NR Za R zb , -NHC(O)R Zc , -NHC(O)OR Zc , - OC(O)NR Za R zb , or optionally substituted 5 to 10-membered heteroaryl.
- each R z is independently -CN, -OH, -OR Za , -Ns, or -NR Za R zb .
- each R z is independently - C(O)NR Za R zb , -NHC(O)R Zc , or -OC(O)NR Za R zb .
- each R z is independently an optionally substituted 5 to 10-membered heteroaryl.
- each R Za and R zb is independently hydrogen, alkyl, or PEG. In some embodiments, each R Za and R zb is independently hydrogen, C1-C20 alkyl, or PEG1.20. In some embodiments, each R Za and R zb is independently C1-C20 alkyl. In some embodiments, each R Za and R zb is independently hydrogen.
- R Zc is alkyl, PEG, cycloalkyl, heterocycloalkyl, or phenyl.
- R Zc is C1-C20 alkyl, PEG1.20, C 3 -Ce cycloalkyl, 4 to 6- membered heterocycloalkyl, or phenyl.
- R Zc is C1-C20 alkyl or PEG1.20.
- R Zc is C1-C20 alkyl.
- W 1 is hydrogen, optionally substituted C1-C10 alkyl, -NR le C(O)R lf , -NR le C(O)NR le R lf , -C(O)NR le R lf , -OC(O)NR le R lf , or -NR le C(O)OR lf .
- W 1 is hydrogen or optionally substituted C1-C10 alkyl.
- W 1 is - NR le C(O)R lf , -NR le C(O)NR le R lf , -C(O)NR le R lf , -OC(O)NR le R lf , or -NR le C(O)OR lf .
- W 1 is -ZB-PO(OR 16 )2, -ZB-(CH2) P 3-PO(OR le )2 , or -Z B -(CH 2 )p3-O-PO2(OR le )2, wherein ZB is O or N, and p3 is 1-10.
- W 1 is hydrogen
- each AA is a natural or unnatural amino acid.
- each R le is independently hydrogen or C1-C10 alkyl. In some embodiments, each R le is independently hydrogen or Ci-Cs alkyl. In some embodiments, each R le is independently hydrogen or Ci-Ce alkyl. In some embodiments, each R le is independently hydrogen or C1-C3 alkyl. In some embodiments, each R le is C1-C3 alkyl. In some embodiments, each R le is independently methyl. In some embodiments, each R le is independently hydrogen.
- each R lf is independently hydrogen, optionally substituted C1-C10 alkyl, optionally substituted C1-C10 heteroalkyl, or optionally substituted 5-membered heteroaryl. In some embodiments, each R lf is independently hydrogen or optionally substituted C1-C10 alkyl. In some embodiments, each R lf is independently hydrogen or optionally substituted Ci-Cs alkyl. In some embodiments, each R lf is independently hydrogen or optionally substituted Ci-Ce alkyl. In some embodiments, each R lf is independently optionally substituted Ci-Ce alkyl. In some embodiments, each R lf is independently methyl. In some embodiments, each R lf is independently hydrogen.
- no is 0. In some embodiments, no is 1.
- mi is 0. In some embodiments, mi is 1. In some embodiments, mi is 2. In some embodiments, mi is 3.
- any of Formula (A-T) through (A-10) is 0. In some embodiments, is 1. In some embodiments, is 2. In some embodiments, is 3.
- p2 is 1-10. In some embodiments, P2 is 1-8. In some embodiments, P2 is 1-6. In some embodiments, P2 is 1-4. In some embodiments, P2 is 1, 2, 3, or 4. In some embodiments, P2 is 1. In some embodiments, P2 is 2. In some embodiments, P2 is 3. In some embodiments, P2 is 4.
- p3 is 1-10. In some embodiments, p3 is 1-8. In some embodiments, p3 is 1-6. In some embodiments, p3 is 1-4. In some embodiments, p3 is 1, 2, 3, or 4. In some embodiments, p3 is 1. In some embodiments, p3 is 2. In some embodiments, p3 is 3. In some embodiments, P3 is 4.
- the binding affinity between the polyamide and the target gene can be adjusted based on the composition of the polyamide.
- the polyamide is capable of binding the DNA with an affinity of less than about 600 nM, about 500 nM, about 400 nM, about 300 nM, about 250 nM, about 200 nM, about 150 nM, about 100 nM, or about 50nM.
- the polyamide is capable of binding the DNA with an affinity of less than about 300 nM.
- the polyamide is capable of binding the DNA with an affinity of less than about 200 nM.
- the polyamide is capable of binding the DNA with an affinity of greater than about 200 nM, about 150 nM, about 100 nM, about 50 nM, about 10 nM, or about 1 nM. In some embodiments, the polyamide is capable of binding the DNA with an affinity in the range of about 1-600 nM, 10-500 nM, 20-500 nM, 50-400 nM, or 100-300 nM.
- the first terminus is capable of binding the DNA with an affinity of less than 500 nM.
- the binding affinity between the polyamide and the target DNA can be determined using a quantitative footprint titration experiment.
- the experiment involves measuring the dissociation constant Kd of the polyamide for the target sequence at either 24 °C or 37 °C, and using either standard polyamide assay solution conditions or approximate intracellular solution conditions.
- the binding affinity between the regulatory protein and the ligand on the second terminus can be determined using an assay suitable for the specific protein.
- the experiment involves measuring the dissociation constant Kd of the ligand for the protein and using either standard protein assay solution conditions or approximate intracellular solution conditions.
- the polyamide composed of a pre-selected combination of subunits can selectively bind to the DNA in the minor groove.
- antiparallel side-by-side pairings of two aromatic amino acids bind to DNA sequences, with a polyamide ring packed specifically against each DNA base.
- the second terminus comprises a protein-binding moiety capable of binding to a regulatory molecule that modulates expression of a gene having the expanded nucleotide repeat.
- the second terminus comprises a bromodomain binding moiety.
- the second terminus comprises a moiety capable of binding to a bromodomain and extra terminal domain (BET) family member.
- the BET family member is BRD2, BRD3, BRD4, or BRDT. In some embodiments, the BET family member is BRD2. In some embodiments, the BET family member is BRD3. In some embodiments, the BET family member is BRD4. In some embodiments, the BET family member is BRDT.
- the protein-binding moiety binds to CBP/p300, PCAF (P300/CBP- Associated Factor), CECR2 (cat eye syndrome chromosome region candidate 2), BRPF (bromodomain and PHD finger-containing protein), ATAD2/ATAD2B (chromatin remodeling proteins), TRIM24 (Tripartite motif-containing 24), BAZ2 (Bromodomain Adjacent to Zinc finger), TAF1 (TBP associated factors), BRD7/9, BPTF (Bromodomain PHD Finger Transcription Factor), SMARCA2/4, or PBRM1.
- PCAF P300/CBP- Associated Factor
- CECR2 cat eye syndrome chromosome region candidate 2
- BRPF bromodomain and PHD finger-containing protein
- ATAD2/ATAD2B chromatin remodeling proteins
- TRIM24 Tripartite motif-containing 24
- BAZ2 Bromodomain Adjacent to Zinc finger
- TAF1 TBP associated factors
- BRD7/9 BRD
- the regulatory molecule is CBP/p300.
- the regulatory molecule is PCAF (P300/CBP-Associated Factor).
- the regulatory molecule is CECR2 (cat eye syndrome chromosome region candidate 2).
- the regulatory molecule is BRPF (bromodomain and PHD fingercontaining protein).
- the regulatory molecule is a ATAD2 or ATAD2B chromatin remodeling protein.
- the regulatory molecule is BAZ2 (Bromodomain Adjacent Zinc Finger.
- the regulatory molecule is TAF1 (TBP associated factor).
- the regulatory molecule is TRIM24 (tripartite motif-containing 24).
- the regulatory molecule is BRD7/9.
- the regulatory molecule is BPTF (Bromodomain PHD Finger Transcription Factor).
- the regulatory molecule is SMARCA2/4.
- the regulatory molecule is PBRM1.
- the regulatory molecule modulates the rearrangement of histones.
- the regulatory molecule modulates the glycosylation, phosphorylation, alkylation, or acylation of histones.
- the regulatory molecule is a transcription factor.
- the regulatory molecule is an RNA polymerase.
- the regulatory molecule is a moiety that regulates the activity of RNA polymerase.
- the recruiting moiety binds to the regulatory molecule but does not inhibit the activity of the regulatory molecule. In some embodiments, the recruiting moiety binds to the regulatory molecule and inhibits the activity of the regulatory molecule. In some embodiments, the recruiting moiety binds to the regulatory molecule and increases the activity of the regulatory molecule.
- the recruiting moiety binds to the active site of the regulatory molecule. In certain embodiments, the recruiting moiety binds to a regulatory site of the regulatory molecule.
- the binding affinity between the regulatory protein and the second terminus can be adjusted based on the composition of the molecule or type of protein.
- the second terminus binds the regulatory molecule with an affinity of less than about 600 nM, about 500 nM, about 400 nM, about 300 nM, about 250 nM, about 200 nM, about 150 nM, about 100 nM, or about 50nM.
- the second terminus binds the regulatory molecule with an affinity of less than about 500 nM.
- the second terminus binds the regulatory molecule with an affinity of less than about 400 nM.
- the second terminus binds the regulatory molecule with an affinity of less than about 300 nM. In some embodiments, the second terminus binds the regulatory molecule with an affinity of less than about 250 nM. In some embodiments, the second terminus binds the regulatory molecule with an affinity of less than about 200 nM. In some embodiments, the second terminus binds the regulatory molecule with an affinity of less than about 150 nM. In some embodiments, the second terminus binds the regulatory molecule with an affinity of less than about 100 nM. In some embodiments, the second terminus binds the regulatory molecule with an affinity of less than about 50 nM.
- the second terminus has a triazolodiazepine structure. In some embodiments, the second terminus has a thiazolodiazepine structure.
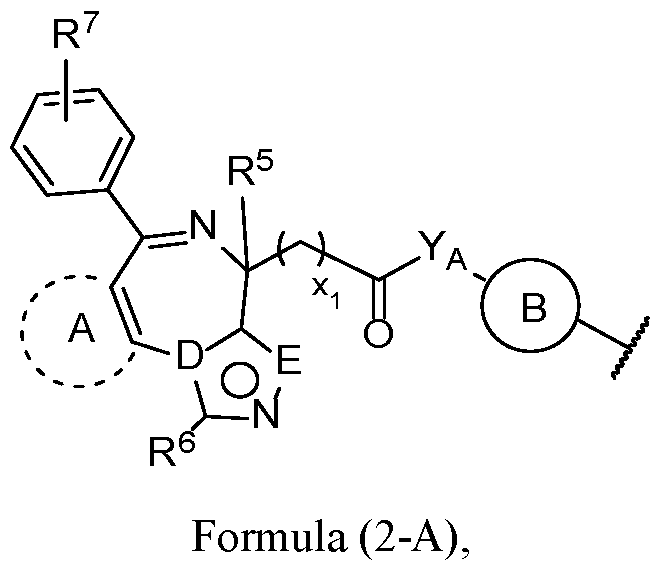
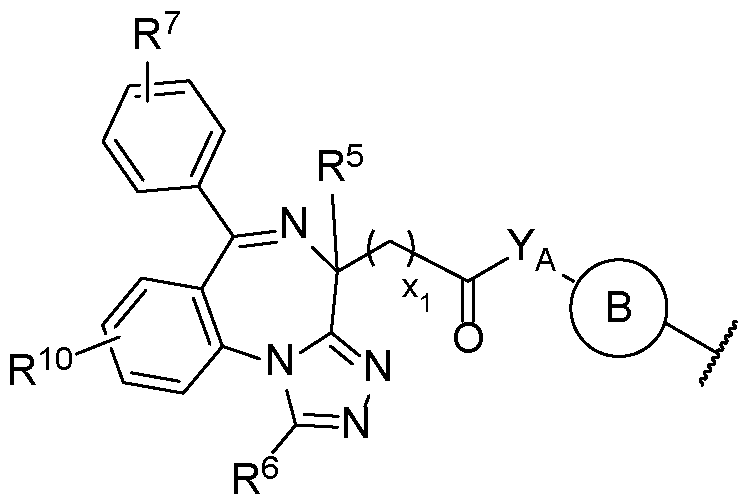
- the second terminus comprises the structure of Formula (2-A), or a pharmaceutically acceptable salt thereof:
- Ring A is an optionally substituted aryl or optionally substituted 5 to 6-membered heteroaryl
- E is O or N
- YA is -NH- or -O-;
- R 6 is selected from hydrogen, optionally substituted Ci-Ce alkyl, optionally substituted Ci-Ce haloalkyl, and optionally substituted Ci-Ce hydroxyalkyl;
- R 7 is selected from hydrogen, halogen, -NO2, -CN, optionally substituted aryl, optionally substituted C1-C20 alkyl, optionally substituted C1-C20 heteroalkyl, optionally substituted Ci-Ce haloalkyl, and optionally substituted Ci-Ce hydroxyalkyl; or R 7 is -NR 7A R 7B , wherein
- R 7A and R 7I are each independently hydrogen, optionally substituted C1-C20 alkyl, or optionally substituted C1-C20 heteroalkyl; and xi is an integer from 1-6.
- D is N and E is N. In some embodiments, D is C and E is O.
- the second terminus comprises the structure of Formula (2-B), or a pharmaceutically acceptable salt thereof:
- Ring B is absent or an optionally substituted 6-membered monocyclic aryl or heteroaryl
- R 5 is hydrogen or Ci-Ce alkyl
- R 6 is selected from hydrogen, optionally substituted Ci-Ce alkyl, optionally substituted Ci-Ce haloalkyl, and optionally substituted Ci-Ce hydroxyalkyl;
- YA is -NH- or -O-;
- R 5 is hydrogen or Ci-Ce alkyl
- R 7A and R 7I are each independently hydrogen, optionally substituted C1-C20 alkyl, or optionally substituted C1-C20 heteroalkyl;
- R8a j s hydrogen, C1-C20 alkyl, or C1-C20 heteroalkyl; and xi is an integer from 1-6.
- the second terminus comprising the structure of Formula (2-D), or a pharmaceutically acceptable salt thereof:
- Ring B is absent or an optionally substituted 6-membered monocyclic aryl or heteroaryl
- YA is -NH- or -O-;
- R 7 is selected from hydrogen, halogen, -NO2, -CN, optionally substituted aryl, optionally substituted C1-C20 alkyl, optionally substituted C1-C20 heteroalkyl, optionally substituted Ci-Ce haloalkyl, and optionally substituted Ci-Ce hydroxyalkyl; or R 7 is -NR 7A R 7B , wherein
- R 7A and R 7I are each independently hydrogen, optionally substituted C1-C20 alkyl, or optionally substituted C1-C20 heteroalkyl;
- R 7 is -NR 7A R 7I; . wherein R 7A and R 7I; are each independently hydrogen or optionally substituted Ci-Ce alkyl.
- R 10 is selected from optionally substituted Ci-Ce alkyl, optionally substituted Ci-Ce haloalkyl, or optionally substituted Ci-Ce hydroxyalkyl. In some embodiments, R 10 is selected from optionally substituted Ci-Ce alkyl. In some embodiments, R 10 is methyl, ethyl, or propyl. In some embodiments, R 10 is methyl. In some embodiments, R 10 is optionally substituted C1-6 hydroxyalkyl. In some embodiments, R 10 is -OMe.
- YA is -NH-. In some embodiments, YA is -O-.
- YA is NH and xi is 1.
- xi is an integer from 1-5, 1-4, 1-3, or 1-2. In some embodiments, xi is 1. In some embodiments, xi is 2.
- Ring B is an optionally substituted 6-membered monocyclic aryl or heteroaryl, each of which is optionally substituted with alkyl, amino, halogen, hydroxy, hydroxyalkyl, or PEG.
- Ring B is phenyl.
- Ring B is 6-membered monocyclic heteroaryl.
- Ring B is pyridine or pyrimidine.
- Ring B is absent.
- the second terminus comprises the structure of Formula (2-E), or a pharmaceutically acceptable salt thereof: Formula (2-E).
- the second terminus comprises the structure of Formula (2-F), or a pharmaceutically acceptable salt thereof: Formula (2-F).
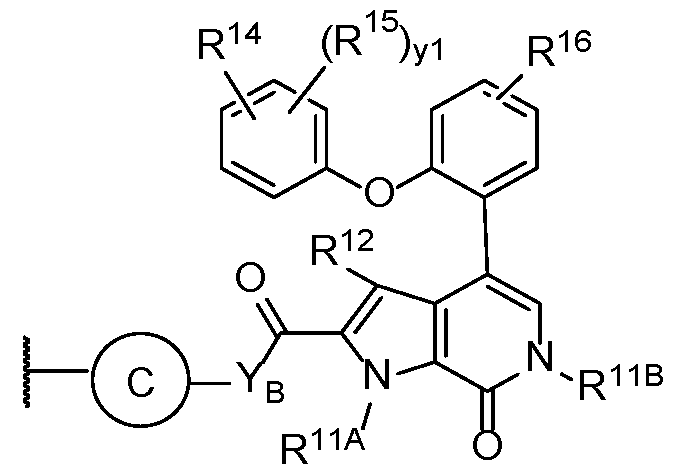
- the second terminus comprises the structure of Formula (2-G), or a pharmaceutically acceptable salt thereof:
- YB is -CH 2 NH-, -CH 2 O-, -NH-, or -O-;
- R 11A and R 11B are each independently hydrogen, or optionally substituted Ci-Ce alkyl;
- R 14 and R 15 are each independently hydrogen, halogen, -CN, -NO2, optionally substituted Ci-Ce alkyl, optionally substituted Ci-Ce haloalkyl, or optionally substituted Ci-Ce hydroxyalkyl; or R 14 is -NR A R B ;
- the second terminus comprises the structure of Formula (3-B), or a pharmaceutically acceptable salt thereof:
- R 12 is hydrogen, optionally substituted Ci-Ce alkyl, C(O)R A , or C(O)NR A R B ; wherein each R A and R B is independently hydrogen, optionally substituted Ci-Ce alkyl, or optionally substituted Ci-Ce heteroalkyl;
- the second terminus comprises the structure of Formula (3-B1), or a pharmaceutically acceptable salt thereof:
- R 11A and R 11B are each independently hydrogen or optionally substituted Ci-Ce alkyl
- R 13 is hydrogen, substituted aryl, substituted heteroaryl, or substituted oxydibenzene; and y2 is an integer from 0-2.
- R 13 is substituted aryl or substituted heteroaryl. In some embodiments, R 13 is hydrogen.
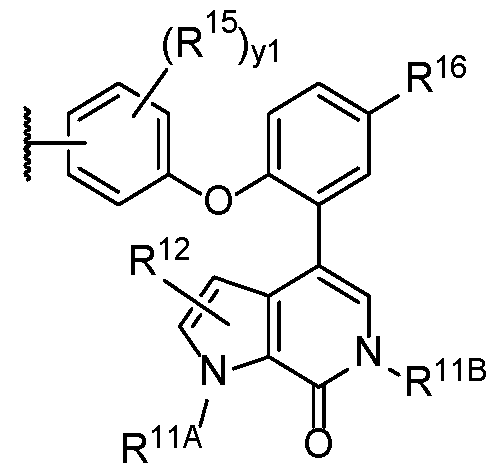
- the second terminus comprises the structure of Formula (3-C), or a pharmaceutically acceptable salt thereof: wherein:
- Ring C is absent, optionally substituted 5 to 6-membered monocyclic aryl or heteroaryl or 4 to 8-membered heterocycle;
- YB is -CH 2 NH-, -CH2O-, -NH-, or -O-;
- R 11A and R 11B are each independently hydrogen or optionally substituted Ci-Ce alkyl
- R 12 is hydrogen, halogen, -OH, optionally substituted Ci-Ce alkyl, optionally substituted Ci-Ce haloalkyl, or optionally substituted Ci-Ce hydroxyalkyl;
- R 16 is optionally substituted Ci-Ce alkyl, optionally substituted Ci-Ce heteroalkyl, optionally substituted C - C
- C-Ce C
- C-Ce C
- C-Ce C
- C-Ce C
- C-Ce C
- C-Ce C
- C-Ce C
- C-Ce C
- C-Ce C
- C-Ce C
- C-Ce C
- -Ce haloalkyl optionally substituted Ci-Ce hydroxyalkyl
- Cs-Ce cycloalkyl optionally substituted 4 to 6-membered heterocycloalkyl, or optionally substituted 5 to 6-membered heteroaryl
- yi is an integer from 1-3.
- Ring C is an optionally substituted 5 or 6-membered monocyclic aryl or heteroaryl, each of which is optionally substituted with alkyl, amino, halogen, hydroxy, hydroxyalkyl, or PEG.
- Ring C is a triazole. In some embodiments, Ring C is an imidazole or pyrrole. In some embodiments, an oxazole or oxadiazole. In some embodiments, Ring C is a thiazole or thiadiazole.
- Ring C is absent.
- R 11A and R 11B are each independently hydrogen or optionally substituted Ci-Ce alkyl
- R 12 is hydrogen or an optionally substituted Ci-Ce alkyl
- each R 15 is independently hydrogen, halogen, -CN, -NO2, optionally substituted Ci-Cg alkyl, optionally substituted Ci-Ce haloalkyl, or optionally substituted Ci-Ce hydroxyalkyl;
- R 11A is Ci-Ce alkyl, optionally substituted with haloalkyl or phosphorous hydroxide. In some embodiments, R 11A is Ci-Ce alkyl substituted with -OP(O)(OH)2. In some embodiments, R 11A is unsubstituted Ci-Ce alkyl. In some embodiments, R 11A is methyl, ethyl, or tert-butyl. In some embodiments, R 11A is methyl. In some embodiments, R 11A is hydrogen.
- R 12 is C(O)R A or C(O)NR A R B . In some embodiments, R 12 is C(O)NR A R B , wherein R A and R B are each independently hydrogen or optionally substituted Ci-Ce alkyl.
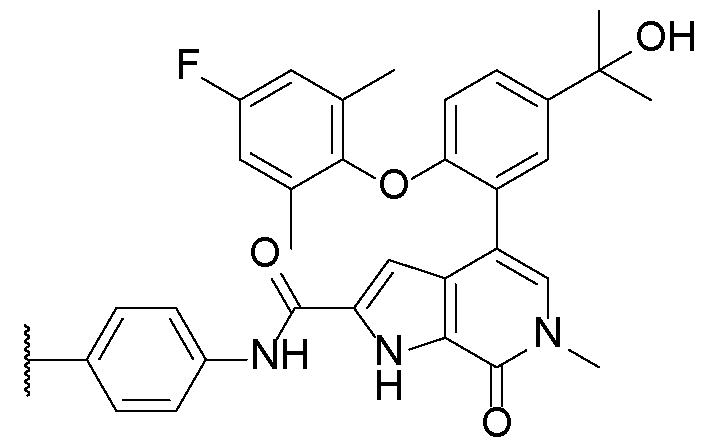
- R 14 and R 15 are each independently hydrogen, -CN, or -NO2. In some embodiments, R 14 and R 15 are each independently halogen or optionally substituted Ci-Ce alkyl. In some embodiments, R 14 and R 15 are each independently Br, Cl, F, methyl, or ethyl. In some embodiments, R 14 and R 15 are each independently F or methyl.
- R 16 is optionally substituted Ci-Ce alkyl, optionally substituted Ci-Ce heteroalkyl, or optionally substituted Ci-Ce hydroxyalkyl. In some embodiments, R 16 is Ci-Ce alkyl or Ci-Ce heteroalkyl, each or which optionally substituted with -CN, -NH2, -N3, -OH, CF3, or -OP(O)(OH)2.
- R 16 is -NHSO2R A , wherein R A is Ci-Ce alkyl. In some embodiments, R 16 is -NHSO2Et. In some embodiments, R 16 is -NHSO2Me.
- the second terminus comprising the structure of Formula (3-G) or Formula (3-H), or a pharmaceutically acceptable salt thereof: Formula (3-H).
- the second terminus comprising the structure of Formula (3-1), or a pharmaceutically acceptable salt thereof: Formula (3-1).
- the second terminus comprises the structure of Formula (4-A), or a pharmaceutically acceptable salt thereof:
- Ring D is absent, phenyl, or 5 to 6-membered heteroaryl
- L 2 is absent, optionally substituted alkylene, -O-, or -NR D -, wherein R D is hydrogen or optionally substituted C1-C3 alkyl;
- R 18 is an optionally substituted 5 to 6-membered heteroaryl
- R 19 is an optionally substituted C3-G cycloalkyl or optionally substituted 4 to 7-membered heteroaryl; each R 20 is independently hydrogen, halogen, -CN, -NO2, optionally substituted Ci-Ce alkyl, optionally substituted Ci-Ce haloalkyl, or optionally substituted Ci-Ce hydroxyalkyl;
- X3 is an integer from 1-3; and y4 is an integer from 1-4; wherein the attachment to the linker is at either R 19 or at one of R 20 .
- attachment to the linker is at R 19 .
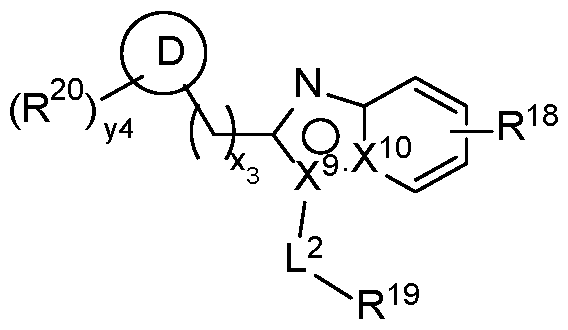
- the second terminus comprises the structure of Formula (4-B), or a pharmaceutically acceptable salt thereof:
- Ring D is absent, optionally substituted phenyl, or optionally substituted 5 to 6-membered heteroaryl;
- X 9 and X 10 are each independently C or N, wherein one of X 9 or X 10 is N;
- R 18 is an optionally substituted 5 to 6-membered heteroaryl
- X 9 is N; and X 10 is C. In some embodiments, X 9 is C; and X 10 is N. [00169] In some embodiments, the second terminus comprises the structure of Formula (4-C), or a pharmaceutically acceptable salt thereof:
- R 18 is an optionally substituted 5 to 6-membered heteroaryl
- R 20 is hydrogen, halogen, -CN, -NO2, optionally substituted Ci-Ce alkyl, optionally substituted Ci-Ce haloalkyl, or optionally substituted Ci-Ce hydroxyalkyl; x 3 is an integer from 1-3; and y4 is an integer from 1-4.
- L 2 is an optionally substituted alkylene. In some embodiments, L 2 is C2-C4 alkylene, optionally substituted with one or more C1-C3 alkyl. In some embodiments, L 2 is absent.
- y4 is 1 or 2. In some embodiments, y4 is 1. In some embodiments, y4 is 2.
- y4 is 3. In some embodiments, y4 is 4.
- the second terminus comprises the structure of Formula (4-E) or Formula (4-F), or a pharmaceutically acceptable salt thereof:
- the second terminus comprises the structure of Formula (4-G), or a pharmaceutically acceptable salt thereof:
- Ring E is absent. In some embodiments, Ring E is an optionally substituted phenyl. In some embodiments, Ring E is an optionally substituted 5 to 6-membered heteroaryl. In some embodiments, Ring E is a 5 -membered heteroaryl. In some embodiments, Ring E is a 6-membered heteroaryl.
- R 22 is halogen, optionally substituted Ci-Ce alkyl, optionally substituted Ci-Ce haloalkyl, or optionally substituted Ci-Ce hydroxyalkyl. In some embodiments, R 22 is CN, F, Cl, Br, or methyl.
- the second terminus comprises the structure of Formula (5-B) or Formula (5-C), or a pharmaceutically acceptable salt thereof:
- Ring G is absent or an optionally substituted C3-C, cycloalkyl or optionally substituted 4 to 6-membered heterocycloalkyl;
- L 6 is -O-(optionally substituted alkylene
- R 29 is optionally substituted Ci-Ce alkyl-(Ce-Cio aryl) or optionally substituted Ci-Ce alkyl-(6 to 10- membered heteroaryl);
- R 30 is optionally substituted Ci-Ce alkyl, optionally substituted Ci-Ce haloalkyl, optionally substituted Ci-Ce hydroxyalkyl, optionally substituted C3-C3 cycloalkyl, or optionally substituted 4 to 6-membered heterocycloalkyl.
- the second terminus comprises the structure of Formula (7-A), or a pharmaceutically acceptable salt thereof:
- a 3 is -O-, -NH-, or -CH 2 -;
- W is O or S; each R 31 is independently hydrogen, halogen, -OH, -CN, -NO2, -NH 2 , optionally substituted C1-C10 alkyl, optionally substituted C1-C10 haloalkyl, optionally substituted C1-C10 hydroxyalkyl, optionally substituted C 2 -Cio alkenyl, optionally substituted C 2 -Cio alkynyl, optionally substituted C3-G cycloalkyl, or optionally substituted 3 to 8-membered heterocycloalkyl; or two R 31 together with the atoms to which they are attached form an optionally substituted Cs-Cs cycloalkyl or optionally substituted 5 to 8-membered heterocycloalkyl;
- R 33 is hydrogen, halogen, -OH, -CN, -NO 2 , -NH 2 , optionally substituted C1-C10 alkyl, optionally substituted C1-C10 haloalkyl, or optionally substituted C1-C10 hydroxyalkyl; and qe is 0-4; wherein Formula (7-A) is atached to the linker is at R 32a or at one of R 31 .
- a 3 is -O-, -NH-, or -CH 2 -;
- Z 3 is CH or N
- W is O or S; each R 31 is hydrogen, halogen, -OH, -CN, -NO 2 , -NH 2 , optionally substituted C1-C10 alkyl, optionally substituted Ci-Cio haloalkyl, optionally substituted C1-C10 hydroxyalkyl, optionally substituted C 2 -Cio alkenyl, optionally substituted C 2 -Cio alkynyl, optionally substituted C-G-cycloalk l.
- R 32 is hydrogen or optionally substituted C1-C10 alkyl; and q 6 is 1-4.
- each R 31 is independently an optionally substituted C1-C10 alkyl, optionally substituted C1-C10 haloalkyl, or optionally substituted C1-C10 hydroxyalkyl. In some embodiments, each R 31 is independently an optionally substituted G-G -cycloalkyl or optionally substituted 3 to 8-membered heterocycloalkyl. In some embodiments, each R 31 is independently hydrogen, halogen, -OH, -CN, -NO 2 , or - NH 2 . In some embodiments, each R 31 is hydrogen.
- the second terminus comprises the structure of Formula (7-C), or a pharmaceutically acceptable salt thereof: Formula (7-C).
- the second terminus comprise the structure of Formula (7-D), or a pharmaceutically acceptable salt thereof:
- the second terminus comprises the structure of Formula (7-E), or a pharmaceutically acceptable salt thereof: Formula (7-E).
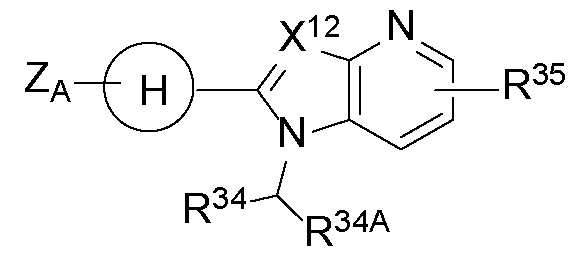
- X 12 is CH or N
- R 34A is hydrogen, halogen, or optionally substituted C1-C3 alkyl
- Ring H is an optionally substituted phenyl. In some embodiments, Ring H is an optionally substituted 6-membered heteroaryl.
- ZA is absent. In some embodiments, ZA is an optionally substituted phenyl formamide. In some embodiments, Z A is -C(O)NH-phenyl.
- R 34 is an optionally substituted phenyl. In some embodiments, R 34 is an optionally substituted 6-membered heteroaryl.
- the second terminus comprises the structure of Formula (8-B) or Formula (8-C), or a pharmaceutically acceptable salt thereof:
- the second terminus comprises the structure of Formula (12-A), or a pharmaceutically acceptable salt thereof:
- a 4 is -CR 40 R 40 - or -NR 40 -; wherein each R 40 is independently hydrogen or optionally substituted C1-C10 alkyl;
- R 36 is an optionally substituted 5 to 6-membered heteroaryl; each R 37 is independently hydrogen, halogen, Ci-Ce alkyl, or Ci-Ce haloalkyl;
- each R 40 is independently optionally substituted C1-C10 alkyl. In some embodiments, each R 40 is independently hydrogen.
- the second terminus comprises Formula (12-D) or (12-E), or a pharmaceutically acceptable salt thereof:
- Ring J is absent or optionally substituted 5 to 6-membered heteroaryl
- R 41 is optionally substituted Ci-Ce alkyl, optionally substituted C’s-G cycloalkyl, -C(O)R 41a , -C(O)-, or - C(O)NR 41a R 41b , wherein
- R 42 is an optionally substituted Ci-Cio alkyl, optionally substituted Ci-Cio haloalkyl, optionally substituted C 3 -C 8 cycloalkyl, or optionally substituted 3 to 8 membered heterocycloalkyl;
- R 43 is hydrogen or optionally substituted C1-C10 alkyl;
- each R 44 is independently hydrogen, halogen, -OH, -CN, -NO2, -NH2, optionally substituted C1-C10 alkyl, optionally substituted Ci-Ciohaloalkyl, C1-C10 hydroxyalkyl, optionally substituted C2-C10 alkenyl, optionally substituted C2-Cio alkynyl, optionally substituted G-G-cycloalkyl. or optionally substituted 3 to 8-membered heterocycloalkyl; or
- R 42 is optionally substituted G-Go alkyl or optionally substituted G-Go haloalkyl. In some embodiments, R 42 is optionally substituted G-G cycloalkyl or optionally substituted 3 to 8 membered heterocycloalkyl. In some embodiments, R 42 is optionally substituted 3- to 8-membered heterocycloalkyl ring.
- R 43 is optionally substituted G-Go alkyl. In some embodiments, R 43 is hydrogen.
- each R 44 is independently halogen, -OH, -CN, -NO2, -NH2, optionally substituted - o alkyl, optionally substituted G-Go haloalkyl, G-Go hydroxyalkyl, optionally substituted G-Go alkenyl, optionally substituted G-Go alkynyl, optionally substituted G-G-cycloalkyl. or optionally substituted 3 to 8-membered heterocycle.
- each R 44 is independently halogen or G- C10 haloalkyl.
- P12 is 3 or 4. In some embodiments, pn is 2. In some embodiments, pn is 1. [00237] In some embodiments, q, is 1. In some embodiments, q 3 is 0.
- Ring J is an optionally substituted 5 -membered heteroaryl. In some embodiments, Ring J is absent. [00239] In some embodiments, Formula (13-A) is connected to the linker at Ring J. In some embodiments, Formula (13-A) is connected to the linker at R 41 . In some embodiments, Formula (13-A) is connected to the linker at R 42 .
- the second terminus comprises the structure of Formula (13-C1) or Formula (13-C2), or a pharmaceutically acceptable salt thereof: .
- the second terminus comprises the structure of Formula (13-D1) or Formula (13-D2), or a pharmaceutically acceptable salt thereof: Formula (13-D1) or Formula (13-D2).
- Ring K is a 5 to 6-membered heterocycloalkyl
- a 5 is absent. In some embodiments, A 5 is -NH- or -O-. In some embodiments, A 5 is -NH-. In some embodiments, A 5 is -O-.
- each R 45 is independently halogen, -OH, -CN, -NO2, -NH2, optionally substituted C1-C10 alkyl, optionally substituted Ci-Ciohaloalkyl, C1-C10 hydroxyalkyl, optionally substituted C2-C10 alkenyl, or optionally substituted C2-C10 alkynyl.
- each R 45 is independently optionally substituted C1-C10 alkyl or optionally substituted C1-C10 hydroxyalkyl.
- each R 45 is independently C1-C10 hydroxyalkyl.
- each R 45 is independently -OCH3 or - OCH2CH3.
- R 47a is optionally substituted C1-C10 alkyl. In some embodiments, R 47a is optionally substituted C3-C8 cycloalkyl.
- qs is 2. In some embodiments, qs is 1. In some embodiments, qs is 0.
- the second terminus comprises the structure of Formula (14-B) or Formula (14-C), or a pharmaceutically acceptable salt thereof:
- the second terminus comprises the structure of Formula (15-B), or a pharmaceutically acceptable salt thereof:
- R 49 is hydrogen, optionally substituted C1-C10 alkyl, optionally substituted C1-C10 haloalkyl, optionally substituted C2-C10 alkenyl, or optionally substituted C2-Cio alkynyl. In some embodiments, R 49 is an optionally substituted C1-C10 alkyl. In some embodiments, R 49 is methyl, ethyl, isopropyl, or tert-butyl. In some embodiments, R 49 is hydrogen.
- B 5 is -O- or -S-. In some embodiments, B 5 is -O-. In some embodiments, B 5 is -S-.
- R 57 is halogen, -OH, -CN, -NO2, -NH2, or optionally substituted C1-C10 alkyl.
- pg is 3. In some embodiments, pg is 2. In some embodiments, pg is 1. [00279] In some embodiments, q? is 2. In some embodiments, q? is 1. In some embodiments, q? is 0. [00280] In some embodiments, the second terminus comprises the structure of Formula (16-B), or a pharmaceutically acceptable salt thereof: Formula (16-B).
- Ring M is optionally substituted aryl or optionally substituted heteroaryl; Ring N is absent or 4 to 8-membered heterocycloalkyl;
- a 6 is -O-, -NH-, or -CH 2 -; each R 58 is independently halogen, -OH, -CN, -NO 2 , -NH 2 , Ci-Cio alkyl, Ci-Ciohaloalkyl, or Ci-Cw hydroxyalkyl;
- R 60 is hydrogen or optionally substituted Ci-Cio alkyl
- a 6 is -O- or -NH-. In some embodiments, A 6 is -CH 2 -.
- B 7 is N or CH
- R 68 and R 69 are each independently an optionally substituted 5-membered heteroaryl; and x 5 and xe are each independently 0-4.
- B 7 is N. In some embodiments, B 7 is CH.
- R 68 is optionally substituted oxazole, oxadiazole, thiazole, thiadiazole, pyrrole, or pyrazole.
- R 68 is pyrrole or pyrazole.
- R 68 is pyrrole.
- R 68 is pyrazole.
- X5 is 2 or 3. In some embodiments, X5 is 1. In some embodiments, X5 is 0.
- xe is 3. In some embodiments, x flare is 2. In some embodiments, xe is 1. In some embodiments, xe is 0.
- the second terminus comprise Formula (18-B), or a pharmaceutically acceptable salt thereof: Formula (18-B).
- the second terminus comprises the structure of Formula (20-A), or a pharmaceutically acceptable salt thereof:
- a 7 is -NHC(O)- or -NHS(O) 2 -;
- a 8 is -O-. In some embodiments, A 8 is -NH-.
- each R 62 is independently optionally substituted C1-C10 alkyl, optionally substituted Ci-Cio haloalkyl, optionally substituted C1-C10 heteroalkyl, or optionally substituted C1-C10 hydroxyalkyl. In some embodiments, each R 62 is independently optionally substituted C1-C10 alkyl. In some embodiments, each R 62 is independently optionally substituted Ci-Ciohaloalkyl. In some embodiments, each R 62 is independently optionally substituted C1-C10 heteroalkyl. In some embodiments, each R 62 is independently optionally substituted C1-C10 hydroxyalkyl.
- a 9 is -O-. In some embodiments, A 9 is -NH-. In some embodiments, A 9 is - NHC(O)-.
- each R 65 is independently optionally substituted C1-C10 alkyl, optionally substituted Ci-C 10 haloalkyl, optionally substituted C1-C10 heteroalkyl, or optionally substituted C1-C10 hydroxyalkyl. In some embodiments, each R 65 is independently optionally substituted C1-C10 alkyl. In some embodiments, each R 65 is independently optionally substituted C1-C10 haloalkyl. In some embodiments, each R 65 is independently optionally substituted C1-C10 heteroalkyl. In some embodiments, each R 65 is independently optionally substituted C1-C10 hydroxyalkyl.
- R 68 and R 69 are each independently halogen, -CN, -NO2, -OH, optionally substituted C1-C10 alkyl, optionally substituted C1-C10 haloalkyl, or optionally substituted C1-C10 hydroxyalkyl;
- R 70 is -S(O)R 70a or -S(O)2R 70a , wherein R 70a is hydrogen or C1-C10 alkyl;
- R 71 is hydrogen or C1-C10 alkyl; wherein Formula (22-A) is attached to the linker at R 67 or R 71 .
- R 67 is hydrogen. In some embodiments, R 67 is -NH2, or -NHSO2CH3.
- R 70 is -S(O)2R 70a . In some embodiments, R 70 is -S(O)2CH3.
- Formula (22-A) is attached to the linker at R 71 .
- the second terminus comprises the structure of Formula (22-B) or Formula
- B 9 is -CH-CR B9 -, -N-, or NR B1 °;
- R 73 is hydrogen; wherein Formula (23-A) is attached to the linker at R B9 , R B1 °, or R 73 .
- B 9 is NR B1 ° and B 10 is -N-.
- Formula (23-A) is attached to the linker at R B9 or R B1 °. In some embodiments, Formula (23-A) is attached to the linker at R 73 .
- the second terminus comprises the structure of Formula (23-B) or Formula (23 -C), or a pharmaceutically acceptable salt thereof:
- the second terminus comprises the structure of Formula (23-D) or Formula (23 -E), or a pharmaceutically acceptable salt thereof:
- R 75 is -0-(Ci-Cio alkyl)-R 75a ; wherein R 75a is -C(O)O-alkyl;
- R 77a is hydrogen or Ci-Cw alkyl; wherein Formula (24-A) is attached to the linker at R 75a or R 76a .
- R 75 is -0-(Ci-Cio alkyl)-C(O)O-alkyl. In some embodiments, R 75 is -O-(Ci- C 4 alkyl)-C(O)O-Ci-C 4 alkyl.
- R 76 is -OH. In some embodiments, R 76 is -OCHs. In some embodiments, R 76 is -O-CH2CH3.
- R 77 is halogen. In some embodiments, R 77 is hydrogen. [00335] In some embodiments, R 77a is hydrogen or methyl. In some embodiments, R 77a is methyl. In some embodiments, R 77a is hydrogen.
- the second terminus comprises the structure of Formula (25-A), or a pharmaceutically acceptable salt thereof:
- R 78 is halogen, -CN, -NO2, -OH, or optionally substituted C1-C10 alkyl;
- R 79a and R 79b are each independently halogen, -CN, -NO2, -NH 2 , -N(CH3) 2 , or optionally substituted C1-C10 alkyl; or R 79a and R 79b together with the atoms to which they are attached form a 6-membered aryl; each R 80 is independently hydrogen, -O-C1-C3 alkyl, C1-C10 alkyl, or C1-C10 alkoxy;
- R 80a is Ci-C 6 alkyl; each R 80b is independently hydrogen or Ci-Ce alkyl; and r 7 is 1, 2, or 3; wherein Formula (25 -A) is attached to the linker at one of R 80 .
- R 79a and R 79b are each independently halogen, -NH2, or -N(CH3)2.
- R 79a and R 79b together with the atoms to which they are attached form a 6- membered aryl.
- each R 80 is independently hydrogen. In some embodiments, each R 80 is independently C1-C10 alkyl. In some embodiments, each R 80 is independently C1-C10 alkoxy. In some embodiments, each R 80 is independently -OCH3.
- the second terminus comprises the structure of Formula (26-A), or a pharmaceutically acceptable salt thereof:
- B n is -O- or -NR 82 -;
- a 10 is -NR 84 - or -NR 84 CH 2 -;
- R 83a is C1-C10 alkyl
- R 84 is hydrogen, C1-C3 alkyl, Cs-Ce cycloalkyl, or phenyl;
- R 85 is hydrogen or C1-C10 alkyl; and r4 is 1, 2, or 3; wherein Formula (26-A) is attached to the linker at one of R 83 .
- a 10 is -NR 84 -. In some embodiments, A 10 is -NR 84 CH2-.
- each R 83 is independently -OH or -OR 83a .
- R 84 is C3-C6 cycloalkyl or phenyl. In some embodiments, R 84 is phenyl.
- the second terminus comprises the structure of Formula (26-B) or Formula
- a 11 is Ci-Ce alkylene
- B 13 is N or CR 100 ;
- R 97 is Ci-C 6 alkyl
- R 98 is halogen
- R 100 is -OR 100a or optionally substituted C1-C10 alkyl; wherein R 100a is hydrogen or C1-C10 alkyl; or R" and R 100 together with the atoms to which they are attached from a heterocycloalkyl.
- a 11 is C1-C4 alkylene. In some embodiments, A 11 is -CH2CH2-.
- the second terminus comprises the structure of Formula (28-C), or a pharmaceutically acceptable salt thereof: Formula (28-C).
- the second terminus comprises the structure of Formula (29-A), or a pharmaceutically acceptable salt thereof: Formula (29-A).
- the second terminus comprises the structure of Formula (30-A), or a pharmaceutically acceptable salt thereof: Formula (30-A).
- the second terminus comprises the structure of Formula (31 -A), or a pharmaceutically acceptable salt thereof: Formula (31 -A), wherein:
- R94a j hydrogen or C1-C3 alkyl
- R 95 is hydrogen, halogen, -OH, -OCH3, or optionally substituted C1-C10 alkyl.
- the second terminus comprises the structure of Formula (33 -A), or a pharmaceutically acceptable salt thereof:
- the second terminus comprises the structure of Formula (34-A), or a pharmaceutically acceptable salt thereof:
- the second terminus comprises the structure of Formula (35-A), or a pharmaceutically acceptable salt thereof:
- R 89a and R 89b are each independently hydrogen or C1-C10 alkyl; or R 89a and R 89b together with the atom to which they are attached form a 5 to 6-membered heterocycloalkyl; R 96 is hydrogen or -CH 3 ; and r 5 and re are each independently 0, 1, 2, or 3.
- Ring T is a C -C, cycloalkyl. In some embodiments, Ring T is a 5 to 6- membered heterocycloalkyl. In some embodiments, Ring T is phenyl.
- R 87 is Cs-Ce cycloalkyl. In some embodiments, R 87 is phenyl.
- R 89a and R 89b are each independently C1-C10 alkyl. In some embodiments, R 89a and R 89b are each hydrogen.
- the second terminus comprises the structure of Formula (37-B) or Formula (37-C), or a pharmaceutically acceptable salt thereof: Formula (37-B) or (Formula 37-C).
- the oligomeric backbone comprises between 5 and 50 chain atoms.
- the oligomeric backbone comprises -(T 1 -V 1 ) a -(T 2 -V 2 )b-(T 3 -V 3 ) c -(T 4 -V 4 )d- (T 5 -V 5 ) e -, wherein a, b, c, d and e are each independently 0 or 1, and where the sum of a, b, c, d and e is 1 to 5;
- n is an integer from 1 to 30;
- (e) h is an integer from 1 to 12;
- V 1 , V 2 , V 3 , V 4 and V 5 are each independently selected from the group consisting of a bond, C(O)-, -NR la -, -C(O)NR la -, -NR la C(O)-, -CONR la -Ci-C 4 alkyl-, -NR la C(O)-Ci-C 4 alkyl-, -C(O)O-, -OC(O)-, -O-, -S-, -S(O)-, -S(O) 2 -, -S(O) 2 NR la -, -NR la S(O) 2 - and -P(O)OH-;
- each R la is independently hydrogen, optionally substituted Ci-Ce alkyl, or optionally substituted Ci-Ce haloalkyl;
- each R lb is independently selected from hydrogen, halogen, alkyl, substituted alkyl, alkenyl, substituted alkenyl, alkynyl, substituted alkynyl, halogen, alkoxy, substituted alkoxy, amino, substituted amino, carboxyl, carboxyl ester, acyl, acyloxy, acyl amino, amino acyl, alkylamide, substituted alkylamide, sulfonyl, thioalkoxy, substituted thioalkoxy, aryl, substituted aryl, heteroaryl, substituted heteroaryl, cycloalkyl, substituted cycloalkyl, heterocyclyl, and substituted heterocyclyl
- the a, b, c, d and e are each independently 0 or 1, where the sum of a, b, c, d and e is 4. In some embodiments, the a, b, c, d and e are each independently 0 or 1, where the sum of a, b, c, d and e is 5.
- n is 3-9. In some embodiments, n is 4-8. In some embodiments, n is 5 or 6. [00395] In some embodiments, T 1 , T 2 , T 3 , and T 4 , and T 5 are each independently selected from C1-C12 alkyl, substituted C1-C12 alkyl, (EA) W , (EDA) m , (PEG) n , (modified PEG) n , (AA) P , -(CR lb OH)h-, phenyl, substituted phenyl, piperidin-4-amino (P4A), para-amino-benzyloxycarbonyl (PABC), meta-amino- benzyloxycarbonyl (MABC), para-amino-benzyloxy (PABO), meta-amino-benzyloxy (MABO), paraaminobenzyl, an acetal group, a disulfide, a
- x is 2-3 and q is 1-3 for EA and EDA.
- R la is hydrogen or Ci-Ce alkyl.
- T 4 or T 5 is an optionally substituted Ce-Cio arylene.
- T 4 or T 5 is phenylene or substituted phenylene. In some embodiments, T 4 or T 5 is phenylene or phenylene substituted with 1-3 substituents selected from Ci-Ce alkyl, halogen, OH or amine. In some embodiments, T 4 or T 5 is 5 to 10-membered heteroarylene or substituted heteroarylene. In some embodiments, T 4 or T 5 is 4 to 10-membered heterocyclene or substituted heterocylcylene. In some embodiments, T 4 or T 5 is heteroarylene or heterocylene optionally substituted with 1-3 substituents selected from Ci-Ce alkyl, halogen, OH or amine.
- the oligomeric backbone comprises N(R la )(CH2) x N(R la )(CH2) x N-, wherein each R la is independently selected from hydrogen, optionally substituted Ci-Ce alkyl, or optionally substituted Ci-Ce haloalkyl; and each x is independently an integer in the range of 1-6.
- the oligomeric backbone comprises -(CH2-C(O)N(R la )-(CH 2 ) q -N(R la )- (CH 2 ) q -N(R la )C(O)-(CH 2 ) x -C(O)N(R la )-A-, -(CH2) x -C(O)N(R la )-(CH 2 CH 2 O) y (CH2) x -C(O)N(R la )-A-, or - C(O)N(R la )-(CH2) q -N(R la )-(CH2)q-N(R la )C(O)-(CH2) x -A-; wherein each q is independently an integer from 2 to 10; each x is independently an integer from 1-6; and each A is independently selected from a bond, an optionally substituted C1-C12 alkyl, an optionally substituted Ce-C
- the oligomeric backbone comprises -NH-(CH2CH2-O) X - or -NH-(CH2CH2-O) X -(CH2CH2)-NH-. In some embodiments, the oligomeric backbone comprises -NH-(CH 2 CH2-O) X -. In some embodiments, the oligomeric backbone comprises -NH- (CH 2 CH2-O) X -(CH 2 CH2)-NH-.
- the oligomeric backbone comprises polyethylene glycol (PEG). In some embodiments, the oligomeric backbone comprises 1-20 PEG units. In some embodiments, the oligomeric backbone comprises 1, 2, 3, 4,5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, or 20 PEG units.
- R 27 is C1-C50 alkyl. In some embodiments, R 27 is C1-C40 alkyl. In some embodiments, R 27 is C1-C30 alkyl. In some embodiments, R 27 is C1-C20 alkyl. In some embodiments, R 27 is C1-C10 alkyl. In some embodiments, R 27 is C1-C50 heteroalkyl. In some embodiments, R 26 is C1-C40 heteroalkyl. In some embodiments, R 27 is C1-C30 heteroalkyl. In some embodiments, R 27 is C1-C20 heteroalkyl. In some embodiments, R 27 is C1-C10 heteroalkyl. In some embodiments, the heteroalkyl is polyethylene glycol (PEG).
- PEG polyethylene glycol
- the oligomeric backbone comprises a moiety having a structure of Formula (C-l), or a pharmaceutically acceptable salt thereof:
- Ring P is absent, arylene, or heterocycloalkylene
- L 5 is absent, optionally substituted alkylene, or optionally substituted alkynylene;
- Ring P is absent. In some embodiments, Ring P is C4-C7 heterocycloalkylene .
- L 5 is absent.
- L 5 is alkylene or alkynylene.
- L 5 is absent, optionally substituted alkylene, or optionally substituted alkynylene;
- B 2 , B 3 and B 4 are each independently N or CH.
- L 5 is C1-C3 alkylene or C1-C3 alkynylene. In some embodiments, L 5 is Ci- C3 alkylene. In some embodiments, L 5 is C1-C3 alkynylene. In some embodiments, L 5 is -CH 2 -, -CH 2 CH 2 -, . In some embodiments, L 5 is -CH 2 - or -CH 2 CH 2 -. In some embodiments, L 5 ents,
- the oligomeric backbone comprises a moiety having the structure of Formula (C-3), or a pharmaceutically acceptable salt thereof:
- each R 1G is independently hydrogen. In some embodiments, R 1G is independently C1-C3 alkyl. In some embodiments, the C1-C3 alkyl is methyl, ethyl or propyl. In some embodiments, each R 1G is independently methyl.
- the oligomeric backbone comprises:
- the oligomeric backbone is joined with the first terminus and/or with the second terminus with a group selected from -C(O)-, -NR la -, -C(O)NR la -, -NR la C(O)-, -C(O)NR la Ci-C4alkyl- , -NR la C(O)-Ci-C 4 alkyl-, -C(O)O-, -OC(O)-, -O-, -S-, -S(O)-, -S(O) 2 -, -S(O) 2 NR la -, -NR la S(O) 2 -, -P(O)OH-, -((CH 2 ) X -O)-, -((CH 2 ) y -NR la )-, optionally substituted C1-C12 alkylene, optionally substituted C 2 -Cio alkenylene, optionally
- the oligomeric backbone is joined with the first terminus with a group selected from -O-, -C(O)-, -NR la -, C1-C12 alkyl, -C(O)NR la -, and -NR la C(O)-. In some embodiments, the oligomeric backbone is joined with the first terminus with a group selected from -O- or -NR la -.
- transcription modulator compounds described herein are presented below in Table 3 (next page).
- the gene is huntingtin (HIT). In some embodiments, the gene is mutant huntingtin.
- the expression of the gene is at least about 10%, at least about 20%, at least about 30%, at least about 40%, at least about 50%, at least about 60%, at least about 70%, at least about 80%, at least about 90%, at least about 95%, or more.
- a method of treating Huntington’s disease (HD) in a subject in need thereof comprising administering to the subject a molecule described herein, or a pharmaceutically acceptable salt thereof.
- a method of treating Huntington’s disease-like syndrome [00439] in a subject in need thereof comprising administering to the subject a molecule described herein, or a pharmaceutically acceptable salt thereof.
- provided herein is a method of treating Juvenile Huntington’s disease in a subject in need thereof, the method comprising administering to the subject a molecule described herein, or a pharmaceutically acceptable salt thereof.
- the method reduces one or more symptoms of Huntington’s disease (HD).
- the one or more symptoms are selected from chorea, cognitive decline, abnormal libido, abnormal eye movement, abnormal sense of smell, aggression, agitation, anxiety, apathy, bradykinesia, bradyphrenia, clumsiness, delusions, depression, difficulty walking, disinhibition, dystonia, gait imbalance, muscle weakness, hallucinations, hostility, hypokinesia, irritability, memory impairment, myoclonus, obsessive-compulsive behavior, poor fine motor coordination, seizure, speech articulation difficulties, staring gaze, weight loss, abnormal cholesterol metabolism, abnormal cerebral white matter, alcoholism, Babinski sign, caudate atrophy, cerebral atrophy, choking, clonus, degeneration of the striatum, excessive daytime sleepiness, impaired visuospatial constructive cognition, inability to walk
- the compounds described herein are administered to a subject in need thereof, either alone or in combination with pharmaceutically acceptable carriers, excipients, or diluents, in a pharmaceutical composition, according to standard pharmaceutical practice. In some embodiments, the compounds described herein are administered to animals.
- compositions comprising a compound described herein, or a pharmaceutically acceptable salt thereof, and at least one pharmaceutically acceptable
- compositions are formulated in a conventional manner using one or more pharmaceutically acceptable excipients that facilitate processing of the active compounds into preparations that can be used pharmaceutically. Proper formulation is dependent upon the route of administration chosen.
- a summary of pharmaceutical compositions described herein can be found, for example, in Remington: The Science and Practice of Pharmacy, Nineteenth Ed (Easton, Pa.: Mack Publishing Company, 1995); Hoover, John E., Remington’s Pharmaceutical Sciences, Mack Publishing Co., Easton, Pennsylvania 1975;
- the dose of a pharmaceutical agent described herein for treating a disease or disorder may depend upon the subject’s condition, that is, stage of the disease, severity of symptoms caused by the disease, general health status, as well as age, gender, and weight, and other factors apparent to a person skilled in the medical art.
- Pharmaceutical compositions may be administered in a manner appropriate to the disease to be treated as determined by persons skilled in the medical arts.
- suitable duration and frequency of administration of the pharmaceutical agent may also be determined or adjusted by such factors as the condition of the patient, the type and severity of the patient’s disease, the particular form of the active ingredient, and the method of administration.
- Optimal doses of an agent may generally be determined using experimental models and/or clinical trials.
- the optimal dose may depend upon the body mass, weight, or blood volume of the subject. The use of the minimum dose that is sufficient to provide effective therapy is usually preferred. Design and execution of pre-clinical and clinical studies for a pharmaceutical agent, including when administered for prophylactic benefit, described herein are well within the skill of a person skilled in the relevant art.
- the optimal dose of each pharmaceutical agent may be different, such as less than when either agent is administered alone as a single agent therapy.
- two pharmaceutical agents in combination may act synergistically or additively, and either agent may be used in a lesser amount than if administered alone.
- the range “from 2 to 6 carbons” is intended to include two, three, four, five, and six carbons, since carbons come in integer units. Compare, by way of example, the range “from 1 to 3 pM (micromolar),” which is intended to include 1 pM, 3 pM, and everything in between to any number of significant figures (e.g., 1.255 pM, 2. 1 pM, 2.9999 pM, etc.).
- Carboxyl refers to -COOH.
- Cyano refers to -CN.
- Alkyl refers to a straight-chain, or branched-chain saturated hydrocarbon monoradical having from one to about ten carbon atoms, more preferably one to six carbon atoms. Examples include, but are not limited to methyl, ethyl, n-propyl, isopropyl, 2-methyl-l -propyl, 2-methyl-2 -propyl, 2-methyl-l -butyl, 3- methyl-1 -butyl, 2-methyl-3 -butyl, 2,2-dimethyl-l -propyl, 2-methyl-l -pentyl, 3 -methyl- 1 -pentyl, 4-methyl-l- pentyl, 2-methyl-2-pentyl, 3 -methyl -2 -pentyl, 4-methyl-2-pentyl, 2,2-dimethyl-l -butyl, 3, 3 -dimethyl- 1- butyl, 2 -ethyl- 1 -butyl, n-
- the alkyl is a Ci-Cioalkyl.
- the alkyl is a Ci-Cealkyl.
- the alkyl is a Ci-Csalkyl.
- the alkyl is a Ci-C4alkyl.
- the alkyl is a Ci-Csalkyl.
- an alkyl group may be optionally substituted, for example, with oxo, halogen, amino, nitrile, nitro, hydroxyl, haloalkyl, alkoxy, carboxyl, carboxylate, aryl, cycloalkyl, heterocycloalkyl, heteroaryl, and the like.
- the alkyl is optionally substituted with oxo, halogen, -Ns, -CN, -C(O)OH, -C(O)OMe, -OH, - OMe, -NH2, or -NO2.
- the alkyl is optionally substituted with halogen, -CN, -OH, or - OMe.
- the alkyl is optionally substituted with halogen.
- Alkenyl refers to a straight-chain, or branched-chain hydrocarbon monoradical having one or more carbon-carbon double-bonds and having from two to about ten carbon atoms, more preferably two to about six carbon atoms.
- C2-C6 alkenyl or “C2-6alkenyl”
- alkenyl group may consist of 2 carbon atoms, 3 carbon atoms, 4 carbon atoms, 5 carbon atoms or 6 carbon atoms, although the present definition also covers the occurrence of the term “alkenyl” where no numerical range is designated.
- an alkenyl group may be optionally substituted, for example, with oxo, halogen, amino, nitrile, nitro, hydroxyl, haloalkyl, alkoxy, carboxyl, carboxylate, aryl, cycloalkyl, heterocycloalkyl, heteroaryl, and the like.
- the alkenyl is optionally substituted with oxo, halogen, -N3, -CN, -C(O)OH, -C(O)OMe, -OH, -OMe, -NH 2 , or - NO2.
- the alkenyl is optionally substituted with halogen, -CN, -OH, or -OMe.
- the alkenyl is optionally substituted with halogen.
- Alkynyl refers to a straight-chain or branched-chain hydrocarbon monoradical having one or more carbon-carbon triple-bonds and having from two to about ten carbon atoms, more preferably from two to about six carbon atoms. Examples include, but are not limited to ethynyl, 2-propynyl, 2-butynyl, 1,3- butadiynyl and the like.
- a numerical range such as “C2-C6 alkynyl” or “C 2 - ealkynyl”, means that the alkynyl group may consist of 2 carbon atoms, 3 carbon atoms, 4 carbon atoms, 5 carbon atoms or 6 carbon atoms, although the present definition also covers the occurrence of the term “alkynyl” where no numerical range is designated.
- an alkynyl group may be optionally substituted, for example, with oxo, halogen, amino, nitrile, nitro, hydroxyl, haloalkyl, alkoxy, carboxyl, carboxylate, aryl, cycloalkyl, heterocycloalkyl, heteroaryl, and the like.
- the alkynyl is optionally substituted with oxo, halogen, -N3, -CN, -C(O)OH, C(O)OMe, -OH, -OMe, -NH 2 , or -NO2.
- alkynyl is optionally substituted with halogen, -CN, -OH, or -OMe. In some embodiments, the alkynyl is optionally substituted with halogen.
- Alkylene refers to a straight or branched divalent hydrocarbon chain. Unless stated otherwise specifically in the specification, an alkylene group may be optionally substituted, for example, with oxo, halogen, amino, nitrile, nitro, hydroxyl, haloalkyl, alkoxy, carboxyl, carboxylate, aryl, cycloalkyl, heterocycloalkyl, heteroaryl, and the like.
- the alkylene is optionally substituted with oxo, halogen, -N3, -CN, -C(O)OH, C(O)OMe, -OH, -OMe, -NH 2 , or -NO2. In some embodiments, the alkylene is optionally substituted with halogen, -CN, -OH, or -OMe. In some embodiments, the alkylene is optionally substituted with halogen.
- the aryl radical may be a monocyclic, bicyclic, tricyclic, or tetracyclic ring system, which may include fused (when fused with a cycloalkyl or heterocycloalkyl ring, the aryl is bonded through an aromatic ring atom) or bridged ring systems.
- the aryl is a 6- to 10-membered aryl.
- the aryl is a 6-membered aryl (phenyl).
- Aryl radicals include, but are not limited to, aryl radicals derived from the hydrocarbon ring systems of anthrylene, naphthylene, phenanthrylene, anthracene, azulene, benzene, chrysene, fluoranthene, fluorene, as-indacene, s-indacene, indane, indene, naphthalene, phenalene, phenanthrene, pleiadene, pyrene, and triphenylene.
- an aryl may be optionally substituted, for example, with halogen, amino, nitrile, nitro, hydroxyl, alkyl, alkenyl, alkynyl, haloalkyl, alkoxy, carboxyl, carboxylate, aryl, cycloalkyl, heterocycloalkyl, heteroaryl, and the like.
- the aryl is optionally substituted with halogen, methyl, ethyl, -N 3 , -CN, -C(O)OH, C(O)OMe, -CF 3 , -OH, -OMe, -NH 2 , or -NO2.
- Cycloalkyl refers to a partially or fully saturated, monocyclic, or polycyclic carbocyclic ring, which may include fused (when fused with an aryl or a heteroaryl ring, the cycloalkyl is bonded through a non-aromatic ring atom), spiro, or bridged ring systems. In some embodiments, the cycloalkyl is fully saturated.
- Representative cycloalkyls include, but are not limited to, cycloalkyls having from three to fifteen carbon atoms (e.g., C 3 -Cis fully saturated cycloalkyl or C 3 -Cis cycloalkenyl), from three to ten carbon atoms (e.g., C 3 -Cio fully saturated cycloalkyl or C 3 -Cio cycloalkenyl), from three to eight carbon atoms (e.g., C 3 -Cs fully saturated cycloalkyl or C 3 -Cs cycloalkenyl), from three to six carbon atoms (e.g., C 3 -Ce fully saturated cycloalkyl or C 3 -Ce cycloalkenyl), from three to five carbon atoms (e.g., C 3 -C 3 fully saturated cycloalkyl or C 3 -C 3 cycloalkenyl), or three to four carbon atoms (e.g
- the cycloalkyl is a 3- to 10-membered fully saturated cycloalkyl or a 3- to 10-membered cycloalkenyl. In some embodiments, the cycloalkyl is a 3 - to 6-membered fully saturated cycloalkyl or a 3- to 6-membered cycloalkenyl. In some embodiments, the cycloalkyl is a 5 - to 6-membered fully saturated cycloalkyl or a 5- to 6-membered cycloalkenyl.
- Monocyclic cycloalkyls include, for example, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, cycloheptyl, and cyclooctyl.
- Polycyclic cycloalkyls include, for example, adamantyl, norbomyl, decalinyl, bicyclo[3.3.0]octane, bicyclo[4.3.0]nonane, cisdecalin, trans-decalin, bicyclo [2.1.1] hexane, bicyclo[2.2.1]heptane, bicyclo[2.2.2]octane, bicyclo[3.2.2]nonane, and bicyclo[3.3.2]decane, and 7,7-dimethyl-bicyclo[2.2.1]heptanyl.
- Partially saturated cycloalkyls include, for example cyclopentenyl, cyclohexenyl, cycloheptenyl, and cyclooctenyl.
- a cycloalkyl is optionally substituted, for example, with oxo, halogen, amino, nitrile, nitro, hydroxyl, alkyl, alkenyl, alkynyl, haloalkyl, alkoxy, carboxyl, carboxylate, aryl, cycloalkyl, heterocycloalkyl, heteroaryl, and the like.
- a cycloalkyl is optionally substituted with oxo, halogen, methyl, ethyl, -Ns, -CN, -C(0)0H, C(0)0Me, -CF 3 , -OH, -OMe, -NH 2 , or -N0 2 .
- a cycloalkyl is optionally substituted with oxo, halogen, methyl, ethyl, -CN, -CF 3 , -OH, or -OMe.
- the cycloalkyl is optionally substituted with halogen.
- Halo or “halogen” refers to bromo, chloro, fluoro or iodo. In some embodiments, halogen is fluoro or chloro. In some embodiments, halogen is fluoro.
- haloalkyl or “haloalkane” refers to an alkyl radical, as defined above, that is substituted by one or more halogen radicals, for example, trifluoromethyl, dichloromethyl, bromomethyl, 2,2,2-trifluoroethyl, 1 -fluoromethyl -2 -fluoroethyl, and the like.
- the alkyl part of the fluoroalkyl radical is optionally further substituted.
- haloalkanes examples include halomethane (e.g., chloromethane, bromomethane, fluoromethane, iodomethane), di-and trihalomethane (e.g., trichloromethane, tribromomethane, trifluoromethane, triiodomethane), 1- haloethane, 2-haloethane, 1,2-dihaloethane, 1-halopropane, 2-halopropane, 3-halopropane, 1,2- dihalopropane, 1,3-dihalopropane, 2,3-dihalopropane, 1,2,3-trihalopropane, and any other suitable combinations of alkanes (or substituted alkanes) and halogens (e.g., Cl, Br, F, I, etc.).
- halogen substituted alkanes e.g., Cl, Br, F, I, etc.
- each halogen may be independently selected e.g., 1- chloro,2-fluoroethane .
- fluoroalkyl refers to an alkyl radical, as defined above, that is substituted by one or more fluoro radicals, for example, trifluoromethyl, difluoromethyl, fluoromethyl, 2,2,2-trifluoroethyl, l-fluoromethyl-2-fluoroethyl, and the like.
- “Hydroxyalkyl” refers to an alkyl radical, as defined above, that is substituted by one or more hydroxyls. In some embodiments, the alkyl is substituted with one hydroxyl. In some embodiments, the alkyl is substituted with one, two, or three hydroxyls. Hydroxyalkyl include, for example, hydroxymethyl, hydroxyethyl, hydroxypropyl, hydroxybutyl, or hydroxypentyl. In some embodiments, the hydroxyalkyl is hydroxymethyl.
- Aminoalkyl refers to an alkyl radical, as defined above, that is substituted by one or more amines. In some embodiments, the alkyl is substituted with one amine. In some embodiments, the alkyl is substituted with one, two, or three amines. Aminoalkyl include, for example, aminomethyl, aminoethyl, aminopropyl, aminobutyl, or aminopentyl. In some embodiments, the aminoalkyl is aminomethyl.
- Heteroalkyl refers to an alkyl group in which one or more skeletal atoms of the alkyl are selected from an atom other than carbon, e.g., oxygen, nitrogen (e.g., -NH-, -N(alkyl)-), sulfur, phosphorus, or combinations thereof.
- a heteroalkyl is attached to the rest of the molecule at a carbon atom of the heteroalkyl.
- a heteroalkyl is a Ci-Ce heteroalkyl wherein the heteroalkyl is comprised of 1 to 6 carbon atoms and one or more atoms other than carbon, e.g., oxygen, nitrogen (e.g., -NH-, -N(alkyl)-), sulfur, phosphorus, or combinations thereof wherein the heteroalkyl is attached to the rest of the molecule at a carbon atom of the heteroalkyl.
- heteroalkyl examples include, for example, -CH2OCH3, - CH2CH2OCH3, -CH2CH2OCH2CH2OCH3, -CH(CH 3 )OCH 3 , -CH2NHCH3, -CH 2 N(CH 3 )2, -CH2CH2NHCH3, or -CH 2 CH2N(CH3)2.
- a heteroalkyl is optionally substituted for example, with oxo, halogen, amino, nitrile, nitro, hydroxyl, alkyl, alkenyl, alkynyl, haloalkyl, alkoxy, aryl, cycloalkyl, heterocycloalkyl, heteroaryl, and the like.
- a heteroalkyl is optionally substituted with oxo, halogen, methyl, ethyl, -CN, -CF3, -OH, -OMe, -NH2, or -NO2.
- a heteroalkyl is optionally substituted with oxo, halogen, methyl, ethyl, -CN, -CF3, -OH, or - OMe. In some embodiments, the heteroalkyl is optionally substituted with halogen.
- Heterocycloalkyl refers to a 3- to 24-membered partially or fully saturated ring radical comprising 2 to 23 carbon atoms and from one to 8 heteroatoms selected from the group consisting of nitrogen, oxygen, phosphorous, silicon, and sulfur. In some embodiments, the heterocycloalkyl is fully saturated. In some embodiments, the heterocycloalkyl comprises one to three heteroatoms selected from the group consisting of nitrogen, oxygen, and sulfur. In some embodiments, the heterocycloalkyl comprises one to three heteroatoms selected from the group consisting of nitrogen and oxygen. In some embodiments, the heterocycloalkyl comprises one to three nitrogens. In some embodiments, the heterocycloalkyl comprises one or two nitrogens.
- the heterocycloalkyl comprises one nitrogen. In some embodiments, the heterocycloalkyl comprises one nitrogen and one oxygen.
- the heterocycloalkyl radical may be a monocyclic, bicyclic, tricyclic, or tetracyclic ring system, which may include fused (when fused with an aryl or a heteroaryl ring, the heterocycloalkyl is bonded through a non-aromatic ring atom), spiro, or bridged ring systems; and the nitrogen, carbon, or sulfur atoms in the heterocycloalkyl radical may be optionally oxidized; the nitrogen atom may be optionally quatemized.
- heterocycloalkyls include, but are not limited to, heterocycloalkyls having from two to fifteen carbon atoms (e.g., C2-C15 fully saturated heterocycloalkyl or C2-C15 heterocycloalkenyl), from two to ten carbon atoms (e.g., C2-C10 fully saturated heterocycloalkyl or C2-C10 heterocycloalkenyl), from two to eight carbon atoms (e.g., C2-C8 fully saturated heterocycloalkyl or C2-C8 heterocycloalkenyl), from two to seven carbon atoms (e.g., C2-C7 fully saturated heterocycloalkyl or C2-C7 heterocycloalkenyl), from two to six carbon atoms (e.g., C2-C6 fully saturated heterocycloalkyl or C2- C, heterocycloalkenyl), from two to five carbon atoms (e.g., C2-C5 fully saturated heterocycloalkyl or C2-C5 heterocycloal
- heterocycloalkyl radicals include, but are not limited to, aziridinyl, azetidinyl, oxetanyl, dioxolanyl, thienyl[l,3]dithianyl, decahydroisoquinolyl, imidazolinyl, imidazolidinyl, isothiazolidinyl, isoxazolidinyl, morpholinyl, octahydroindolyl, octahydroisoindolyl, 2-oxopiperazinyl, 2- oxopiperidinyl, 2-oxopyrrolidinyl, oxazolidinyl, piperidinyl, piperazinyl, 4-piperidonyl, pyrrolidinyl, pyrazolidinyl, quinuclidinyl, thiazolidinyl, tetrahydrofuryl, trithianyl, tetrahydro
- the heterocycloalkyl is a 3 - to 7-membered fully saturated heterocycloalkyl. In some embodiments, the heterocycloalkyl is a 3 - to 6-membered fully saturated heterocycloalkyl. In some embodiments, the heterocycloalkyl is a 4- to 6-membered fully saturated heterocycloalkyl. In some embodiments, the heterocycloalkyl is a 5 - to 6-membered fully saturated heterocycloalkyl. In some embodiments, the heterocycloalkyl is a 3- to 8-membered heterocycloalkenyl. In some embodiments, the heterocycloalkyl is a 3- to 7-membered heterocycloalkenyl.
- the heterocycloalkyl is a 3- to 6-membered heterocycloalkenyl. In some embodiments, the heterocycloalkyl is a 4- to 6-membered heterocycloalkenyl. In some embodiments, the heterocycloalkyl is a 5 - to 6-membered heterocycloalkenyl.
- a heterocycloalkyl may be optionally substituted as described below, for example, with oxo, halogen, amino, nitrile, nitro, hydroxyl, alkyl, alkenyl, alkynyl, haloalkyl, alkoxy, carboxyl, carboxylate, aryl, cycloalkyl, heterocycloalkyl, heteroaryl, and the like.
- the heterocycloalkyl is optionally substituted with oxo, halogen, methyl, ethyl, -CN, -C(O)OH, C(O)OMe, -CF3, -OH, -OMe, -NH2, or -NO2.
- the heterocycloalkyl is optionally substituted with halogen, methyl, ethyl, -CN, -CF3, -OH, or -OMe. In some embodiments, the heterocycloalkyl is optionally substituted with halogen.
- Heteroaryl refers to a 5- to 14-membered ring system radical comprising one to thirteen carbon atoms, one to six heteroatoms selected from the group consisting of nitrogen, oxygen, phosphorous, and sulfur, and at least one aromatic ring.
- the heteroaryl comprises one to three heteroatoms selected from the group consisting of nitrogen, oxygen, and sulfur.
- the heteroaryl comprises one to three heteroatoms selected from the group consisting of nitrogen and oxygen.
- the heteroaryl comprises one to three nitrogens.
- the heteroaryl comprises one or two nitrogens.
- the heteroaryl comprises one nitrogen.
- the heteroaryl is a 5- membered heteroaryl.
- examples include, but are not limited to, azepinyl, acridinyl, benzimidazolyl, benzothiazolyl, benzindolyl, benzodioxolyl, benzofuranyl, benzooxazolyl, benzothiazolyl, benzothiadiazolyl, benzo[b][l,4]dioxepinyl, 1,4-benzodioxanyl, benzonaphthofuranyl, benzoxazolyl, benzodioxolyl, benzodioxinyl, benzopyranyl, benzopyranonyl, benzofuranyl, benzofuranonyl, benzothienyl (benzothiophenyl), benzotriazolyl, benzo[4,6]imidazo[l,2-a]pyridinyl, carbazolyl, cinnolin
- the heteroaryl is optionally substituted with halogen, methyl, ethyl, -CN, -CF 3 , -OH, or -OMe. In some embodiments, the heteroaryl is optionally substituted with halogen.
- oligonucleotide sequence refers to a plurality of nucleic acids having a defined sequence and length (e.g., 2, 3, 4, 5, 6, or even more nucleotides).
- oligonucleotide repeat sequence refers to a contiguous expansion of oligonucleotide sequences.
- transcription well known in the art, refers to the synthesis of RNA (i.e., ribonucleic acid) by DNA-directed RNA polymerase.
- modulate transcription refers to a change in transcriptional level which can be measured by methods well known in the art, for example, assay of mRNA, the product of transcription. In certain embodiments, modulation is an increase in transcription. In other embodiments, modulation is a decrease in transcription.
- polyamide refers to polymers of linkable units chemically bound by amide (i.e., CONH) linkages; optionally, polyamides include chemical probes conjugated therewith.
- Polyamides may be synthesized by stepwise condensation of carboxylic acids (COOH) with amines (RR’NH) using methods known in the art. Alternatively, polyamides may be formed using enzymatic reactions in vitro, or by employing fermentation with microorganisms.
- linkable unit refers to methylimidazoles, methylpyrroles, and straight and branched chain aliphatic functionalities (e.g., methylene, ethylene, propylene, butylene, and the like) which optionally contain nitrogen Substituents, and chemical derivatives thereof.
- the aliphatic functionalities of linkable units can be provided, for example, by condensation of B-alanine or dimethylaminopropylamine during synthesis of the polyamide by methods well known in the art.
- linker refers to a chain of at least 10 contiguous atoms. In certain embodiments, the linker contains no more than 20 non-hydrogen atoms. The terms linker and oligomeric backbone can be used interchangeably. In some embodiments, the linker contains no more than 40 non-hydrogen atoms. In some embodiments, the linker contains no more than 60 non-hydrogen atoms. In certain embodiments, the linker contains atoms chosen from C, H, N, O, and S. In some embodiments, every non-hydrogen atom is chemically bonded either to 2 neighboring atoms in the linker, or one neighboring atom in the linker and a terminus of the linker.
- “optionally substituted” is a substituted group is derived from the unsubstituted parent group in which there has been an exchange of one or more hydrogen atoms for another atom or group. Unless otherwise indicated, when a group is deemed to be “substituted” or “optionally substituted” it is meant that the group is substituted with one or more substituents independently selected from Ci-Ce alkyl, Ci-Ce alkenyl, Ci-Ce alkynyl, Ci-Ce heteroalkyl, C3-C7 carbocyclyl (optionally substituted with halo, Ci-Ce alkyl, Ci-Ce alkoxy, Ci-Ce haloalkyl, and Ci-Ce haloalkoxy), Cs-Cv-carbocyclyl-Ci-Ce-alkyl (optionally substituted with halo, Ci-Ce alkyl, Ci-Ce alkoxy, Ci-Ce haloal
- Chemical entities having carbon-carbon double bonds or carbon-nitrogen double bonds may exist in Z- or E- form (or cis- or trans- form). Furthermore, some chemical entities may exist in various tautomeric forms. Unless otherwise specified, compounds described herein are intended to include all Z-, E- and tautomeric forms as well.
- the compounds disclosed herein are used in different enriched isotopic forms, e.g., enriched in the content of 2 H, 3 H, n C, 13 C and/or 14 C.
- the compound is deuterated in at least one position.
- deuterated forms can be made by the procedure described in U.S. Patent Nos. 5,846,514 and 6,334,997. As described in U.S. Patent Nos. 5,846,514 and 6,334,997, deuteration can improve the metabolic stability and or efficacy, thus increasing the duration of action of drugs.
- compounds described herein are intended to include compounds which differ only in the presence of one or more isotopically enriched atoms.
- compounds having the present structures except for the replacement of a hydrogen by a deuterium or tritium, or the replacement of a carbon by 13 C- or 14 C-enriched carbon are within the scope of the present disclosure.
- the compounds of the present disclosure optionally contain unnatural proportions of atomic isotopes at one or more atoms that constitute such compounds.
- the compounds may be labeled with isotopes, such as for example, deuterium ( 2 H), tritium ( 3 H), iodine-125 ( 125 I) or carbon-14 ( 14 C).
- Isotopic substitution with 2 H, n C, 13 C, 14 C, 15 C, 12 N, 13 N, 15 N, 16 N, 16 0, 17 0, 14 F, 15 F, 16 F, 17 F, 18 F, 33 S, 34 S, 35 S, 36 S, 35 C1, 37 C1, 79 Br, 81 Br, and 125 I are all contemplated. All isotopic variations of the compounds of the present invention, whether radioactive or not, are encompassed within the scope of the present invention. In some embodiments, where isotopic variations are illustrated, the remaining atoms of the compound may optionally contain unnatural portions of atomic isotopes.
- the compounds disclosed herein have some or all of the ’H atoms replaced with 2 H atoms.
- the methods of synthesis for deuterium-containing compounds are known in the art and include, by way of non-limiting example only, the following synthetic methods.
- Deuterium substituted compounds are synthesized using various methods such as described in: Dean, Dennis C.; Editor. Recent Advances in the Synthesis and Applications of Radiolabeled Compounds for Drug Discovery and Development. [In: Curr., Pharm. Des., 2000; 6(10)] 2000, 110 pp; George W.; Varma, Rajender S. The Synthesis of Radiolabeled Compounds via Organometallic Intermediates, Tetrahedron, 1989, 45(21), 6601-21; and Evans, E. Anthony. Synthesis of radiolabeled compounds, J. Radioanal. Chem., 1981, 64(1-2), 9-32.
- Deuterated starting materials are readily available and are subjected to the synthetic methods described herein to provide for the synthesis of deuterium-containing compounds.
- Large numbers of deuterium-containing reagents and building blocks are available commercially from chemical vendors, such as Aldrich Chemical Co.
- one or more of the substituent groups comprise deuterium at a percentage higher than the natural abundance of deuterium.
- one or more hydrogens are replaced with one or more deuteriums.
- the abundance of deuterium in each of the substituents is independently at least 1%, at least 10%, at least 20%, at least 30%, at least 40%, at least 50%, at least 60%, at least 70%, at least 80%, at least 90%, or 100% of a total number of hydrogen and deuterium.
- Compounds of the present disclosure also include crystalline and amorphous forms of those compounds, pharmaceutically acceptable salts, and active metabolites of these compounds having the same type of activity, including, for example, polymorphs, pseudopolymorphs, solvates, hydrates, unsolvated polymorphs (including anhydrates), conformational polymorphs, and amorphous forms of the compounds, as well as mixtures thereof.
- the compounds described herein may in some cases exist as diastereomers, enantiomers, or other stereoisomeric forms. Where absolute stereochemistry is not specified, the compounds presented herein include all diastereomeric, enantiomeric, and epimeric forms as well as the appropriate mixtures thereof. Separation of stereoisomers may be performed by chromatography or by forming diastereomers and separating by recrystallization, or chromatography, or any combination thereof. (Jean Jacques, Andre Collet, Samuel H. Wilen, “Enantiomers, Racemates and Resolutions”, John Wiley And Sons, Inc., 1981, herein incorporated by reference for this disclosure). Stereoisomers may also be obtained by stereoselective synthesis.
- salt or “pharmaceutically acceptable salt” refers to salts derived from a variety of organic and inorganic counter ions well known in the art.
- Pharmaceutically acceptable acid addition salts can be formed with inorganic acids and organic acids.
- Inorganic acids from which salts can be derived include, for example, hydrochloric acid, hydrobromic acid, sulfuric acid, nitric acid, phosphoric acid, and the like.
- Organic acids from which salts can be derived include, for example, acetic acid, propionic acid, glycolic acid, pyruvic acid, oxalic acid, maleic acid, malonic acid, succinic acid, fumaric acid, tartaric acid, citric acid, benzoic acid, cinnamic acid, mandelic acid, methanesulfonic acid, ethanesulfonic acid, p- toluene sulfonic acid, salicylic acid, and the like.
- Pharmaceutically acceptable base addition salts can be formed with inorganic and organic bases.
- Inorganic bases from which salts can be derived include, for example, sodium, potassium, lithium, ammonium, calcium, magnesium, iron, zinc, copper, manganese, aluminum, and the like.
- Organic bases from which salts can be derived include, for example, primary, secondary, and tertiary amines, substituted amines including naturally occurring substituted amines, cyclic amines, basic ion exchange resins, and the like, specifically such as isopropylamine, trimethylamine, diethylamine, triethylamine, tripropylamine, and ethanolamine.
- the pharmaceutically acceptable base addition salt is chosen from ammonium, potassium, sodium, calcium, and magnesium salts.
- phrases “pharmaceutically acceptable” is employed herein to refer to those compounds, materials, compositions, and/or dosage forms which are, within the scope of sound medical judgment, suitable for use in contact with the tissues of human beings and animals without excessive toxicity, irritation, allergic response, or other problem or complication, commensurate with a reasonable benefit/risk ratio.
- phrases “pharmaceutically acceptable excipient” or “pharmaceutically acceptable carrier” as used herein means a pharmaceutically acceptable material, composition, or vehicle, such as a liquid or solid filler, diluent, excipient, solvent, or encapsulating material. Each carrier must be “acceptable” in the sense of being compatible with the other ingredients of the formulation and not injurious to the patient.
- an “effective amount” or “therapeutically effective amount” refers to an amount of a compound administered to a mammalian subject, either as a single dose or as part of a series of doses, which is effective to produce a desired therapeutic effect.
- treat include alleviating, abating, or ameliorating at least one symptom of a disease or condition, preventing additional symptoms, inhibiting the disease or condition, e.g., arresting the development of the disease or condition, relieving the disease or condition, causing regression of the disease or condition, relieving a condition caused by the disease or condition, or stopping the symptoms of the disease or condition.
- the term “patient” is generally synonymous with the term “subject” and includes all mammals including humans. Examples of patients include humans, livestock such as cows, goats, sheep, pigs, and rabbits, and companion animals such as dogs, cats, rabbits, and horses. Preferably, the patient is a human.
- the term “contacting” refers to bringing the compound (e.g., a transcription molecular molecule of the present disclosure) into proximity of the desired target gene. The contacting may result in the binding to or result in a conformational change of the target moiety.
- the methods and compositions described herein include the use of amorphous forms as well as crystalline forms (also known as polymorphs).
- the compounds described herein may be in the form of pharmaceutically acceptable salts.
- active metabolites of these compounds having the same type of activity are included in the scope of the present disclosure.
- the compounds described herein can exist in unsolvated as well as solvated forms with pharmaceutically acceptable solvents such as water, ethanol, and the like.
- the solvated forms of the compounds presented herein are also considered to be disclosed herein.
- AC2O acetic anhydride
- AcCl acetyl chloride
- AcOH acetic acid
- AIBN azobisisobutyronitrile
- aq. aqueous
- BusSnH tributyltin hydride
- Boc tert-butyloxy carbonyl
- CD3OD deuterated methanol
- CDCE deuterated chloroform
- CDI 1,1 '-Carbonyldiimidazole
- DBU 1,8- diazabicyclo[5.4.0]undec-7-ene
- DCM dichloromethane
- DEAD diethyl azodicarboxylate
- DIBAL-H di-iso-butyl aluminium hydride
- DMAP 4- dimethylaminopyridine
- DMF N,N-dimethylformamide
- DMSO-de deuterated di
- Example 1 Synthesis of 3-[(4- ⁇ 4-[3-( ⁇ 4-[(2R)-2-[(tert-butoxycarbonyl)amino]-4-( ⁇ l-methyl-4- H-methyl-4-(3-Hl-methyl-4-(l-methylimidazole-2-amido)pyrrol-2- yl]formamidoIpropanamido)imidazole-2-amido1pyrrol-2-vHformamido)butanamido1-l- methylimidazol-2-yl ⁇ formamido)propanamido]-l-methylpyrrole-2-amido ⁇ -l-methylimidazol-2- yl)formamido]propanoic acid
- Step 1 To a stirred solution of (2R)-2-[(tert-butoxycarbonyl)amino]-4- ⁇ [(9H-fluoren-9- ylmethoxy)carbonyl] amino [butanoic acid (3.00 g, 6.80 mmol, 0.80 equiv) in DMF (50.00 mb) was added DIEA (5.50 g, 42.55 mmol, 5.00 equiv), ethyl 4-amino-l-methylimidazole-2 -carboxylate (1.44 g, 8.51 mmol, 1.00 equiv) and PyBOP (5.76 g, 11.06 mmol, 1.30 equiv) in portions at 0 °C.
- DIEA 5.50 g, 42.55 mmol, 5.00 equiv
- ethyl 4-amino-l-methylimidazole-2 -carboxylate (1.44 g, 8.51 mmol, 1.00 equi
- Step 2 Into a 50 ml flask was added ethyl 4-[(2R)-2-[(tert-butoxycarbonyl)amino]-4- ⁇ [(9H- fluoren-9-ylmethoxy)carbonyl]amino ⁇ butanamido]-l-methylimidazole-2-carboxylate (500.00 mg, 0.85 mmol, 1.00 equiv) in DMF (5.00 mb) and piperidine (1.00 mb). The reaction was stirred at r.t. for 30 mins.
- Step 3 The procedure was the same as ethyl 4-[(2R)-2-[(tert-butoxycarbonyl)amino]-4- ⁇ [(9H- fluoren-9-ylmethoxy)carbonyl]amino ⁇ butanamido]-l-methylimidazole-2-carboxylate (Example 1 Step 1). 230.00 mg of ethyl 4-[(2R)-4-amino-2-[(tert- butoxycarbonyl)amino]butanamido]-l-methylimidazole-2- carboxylate was used and 200.00 mg of desired product was obtained as a yellow solid (68.39% yield). LC/MS: mass calcd. For C41H53N15O10: 915.41, found: 916.75[M+H] + .
- Step 4 To a stirred solution of ethyl 4-[(2R)-2-[(tert-butoxycarbonyl) amino]-4-( ⁇ l-methyl-4-[l- methyl-4-(3- ⁇ [l -methyl -4-(l-methylimidazole-2 -amido) pyrrol-2-yl]formamido ⁇ propanamido)imidazole-2- amido]pyrrol-2-yl ⁇ formamido)butanamido]-l-methylimidazole-2 -carboxylate (570 mg, 1.00 equiv) in a mixture of solvents MeOH/THF (5:3), was added 2 M LiOH in water (6.00 equiv) at room temperature.
- Step 5 The procedure was the same as ethyl 4-[(2R)-2-[(tert-butoxycarbonyl)amino]-4- ⁇ [(9H- fluoren-9-yhnethoxy)carbonyl]amino ⁇ butanamido]-l-methylimidazole-2-carboxylate (Example 1 Step 1).
- Step 2 To a stirred solution of tert-butyl N-[(lR)-l-( ⁇ 2-[(2- ⁇ [5-( ⁇ 2-[(2- ⁇ [5- (dimethylamino)pentyl]carbamoyl ⁇ ethyl)carbamoyl] - 1 -methylimidazol-4-yl ⁇ carbamoyl)- 1 -methylpyrrol-3 - yl]carbamoyl ⁇ ethyl)carbamoyl] - 1 -methylimidazol-4-yl ⁇ carbamoyl)-3 -( ⁇ 1 -methyl -4-[ 1 -methyl -4-(3 - ⁇ [ 1 - methyl-4-(l-methylimidazole-2-amido)pyrrol-2-yl] formamido ⁇ propanamido)imidazole-2-amido]pyrrol-2- yl ⁇ formamido)propyl]
- Step 1 The procedure was the same as tert-butyl N-[( 1R)- 1-( ⁇ 2-[(2- ⁇ [5-( ⁇ 2-[(2- ⁇ [5- (dimethylamino)pentyl] carbamoyl ⁇ ethyl)carbamoyl] - 1 -methylimidazol-4-yl ⁇ carbamoyl)- 1 -methylpyrrol-3 - yl]carbamoyl ⁇ ethyl)carbamoyl] - 1 -methylimidazol-4-yl ⁇ carbamoyl)-3 -( ⁇ 1 -methyl -4-[ 1 -methyl -4-(3 - ⁇ [ 1 - methyl-4-(l-methylimidazole-2-amido)pyrrol-2-yl]formamido ⁇ propanamido)imidazole-2-amido]pyrrol-2- yl ⁇ formamido)propyl] carba
- Step 2 To a stirred solution of ethyl 4- ⁇ 4-[3-( ⁇ 4-[(2R)-2-[(tert-butoxycarbonyl)amino]- 4-( ⁇ l- methyl-4-[ 1 -methyl -4-(3 - ⁇ [ 1 -methyl -4-( 1 -methylimidazole-2-amido)pyrrol-2- yl]formamido ⁇ propanamido)imidazole-2-amido]pyrrol-2-yl ⁇ formamido)butanamido]- l-methylimidazol-2- yl ⁇ formamido)propanamido]-l-methylpyrrole-2-amido ⁇ -l-methylimidazole-2 -carboxylate (1.30 g, 1.05 mmol, 1.00 equiv.) in MeOH (13.00 mL) was added 2M LiOH.H 2 O (2.11 mL, 4.22 mmol,
- Step 3 The procedure was the same as tert-butyl N-[(lR)-l-( ⁇ 2-[(2- ⁇ [5-( ⁇ 2-[(2- ⁇ [5- (dimethylamino)pentyl] carbamoyl ⁇ ethyl)carbamoyl] - 1 -methylimidazol-4-yl ⁇ carbamoyl)- 1 -methylpyrrol-3 - yl]carbamoyl ⁇ ethyl)carbamoyl] - 1 -methylimidazol-4-yl ⁇ carbamoyl)-3 -( ⁇ 1 -methyl -4-[ 1 -methyl -4-(3 - ⁇ [ 1 - methyl-4-(l-methylimidazole-2-amido)pyrrol-2-yl]formamido ⁇ propanamido)imidazole-2-amido]pyrrol-2- yl ⁇ formamido)propyl] carb
- Step 4 The procedure was the same as N-(5- ⁇ [(3R)-3-amino-3-( ⁇ 2-[(2- ⁇ [5-( ⁇ 2-[(2- ⁇ [5- (dimethylamino)pentyl] carbamoyl ⁇ ethyl) carbamoyl] - 1 -methylimidazol-4-yl ⁇ carbamoyl)- 1 -methylpyrrol-3 - yl] carbamoyl ⁇ ethyl)carbamoyl] - 1 -methylimidazol-4-yl ⁇ carbamoyl)propyl] carbamoyl ⁇ - 1 -methylpyrrol-3 - yl)- 1 -methyl -4-(3 - ⁇ [ 1 -methyl -4-( 1 -methylimidazole-2-amido)pyrrol-2- yl]formamido ⁇ propanamido)imidazole-2 -carboxamide (Ex
- Step 1 To a stirred solution of (2R)-2-[[(9H-fluoren-9-ylmethoxy)carbonyl]amino]-4-methoxy- 4-oxobutanoic acid (5.00 g, 13.54 mmol, 1.00 equiv) in DMF (60.00 mb) was added NMI (3.33 g, 40.61 mmol, 3.00 equiv), TCFH (5.70 g, 20.31 mmol, 1.50 equiv) and 3-(4-chlorobenzoyl)-4,5-dimethylthiophen- 2-amine (3.96 g, 14.90 mmol, 1.10 equiv) in portions at 0 °C.
- Step 2 To a solution of methyl (3R)-3-amino-3-[[3-(4-chlorobenzoyl)-4,5-dimethylthiophen-2- yl] carbamoyl] propanoate (4.95 g) in DCM was treated with EtsN followed by AcOH treatment in DCE to give 2.10 g of methyl 2-[(3R)-5-(4-chlorophenyl)-6,7-dimethyl-2-oxo-lH,3H-thieno[2,3-e][l,4]diazepin-3- yl]acetate as a yellow solid (70.00% yield of two steps).
- LCMS mass calcd. For C18H17CIN2O3S: 376.06, found: 377.15 [M+H] + .
- Step 3 To a solution of methyl 2-[(3R)-5-(4-chlorophenyl)-6,7-dimethyl-2-oxo-lH,3H- thieno[2,3-e][l,4]diazepin-3-yl]acetate (1.10 g, 2.92 mmol, 1.00 equiv) in toluene (25.00 ml) was added P2S5 (1.30 g, 0.006 mmol, 2.00 equiv) and ISfeCCE (0.62 g, 5.85 mmol, 2.00 equiv). The reaction was stirred at 110 °C for 17 h.
- Step 4 To a solution of methyl 2-[(3R)-5-(4-chlorophenyl)-6,7-dimethyl-2-sulfanylidene-lH,3H- thieno[2,3-e][l,4]diazepin-3-yl]acetate (900.00 mg, 2.29 mmol, 1.00 equiv) in THF (6.00 mb) was added NH2NH2.H2O (344.00 mg, 6.87 mmol, 3.00 equiv) dropwise at 0 °C. The reaction was stirred at 0 °C for 1 h.
- Step 5 A solution of methyl (R)-2-(4-(4-chlorophenyl)-2,3,9-trimethyl-6H-thieno[3,2-f] [l,2,4]triazolo[4,3-a][l,4]diazepin-6-yl)acetate (400.00 mg, 0.96 mmol, 1.00 equiv) in LiOH (2M, 2.00 mL, 4.00 equiv) and MeOH (2.00 mL) was stirred at 40 °C for 2 h. The mixture was acidified with HC1 (2M) and concentrated. The residue was dissolved in DMF (2 mL) and purified by reverse phase column with the following conditions: column, C18 column; mobile phase, MeCN in water (0.05% TFA),
- Step 1 A solution of 2,4-difluorophenol (2.66 g, 20.45 mmol, 1.50 equiv) and 2-bromo-l-fluoro- 4-nitrobenzene (3.00 g, 13.64 mmol, 1.00 equiv) in DMF (50.00 mb) was stirred at 85 °C for 3.0 h. The reaction was cooled to room temperature and was then diluted with water (150 mL). The precipitate was filtered and the filter cake was washed with water and dried to afford 2-bromo-l-(2,4-difluorophenoxy)-4- nitrobenzene (4.40 g, 98.21%) as light a yellow solid.
- Step 2 Iron powder (7.78 g, 139.36 mmol, 10.00 equiv) was added in portions to a stirred solution of 2-bromo-l-(2,4-difluorophenoxy)-4-nitrobenzene (4.40 g, 13.37 mmol, 1.00 equiv) in EtOH (20.00 mL) and THF (20.00 mL), NH4CI (7.45 g, 139.36 mmol, 10.00 equiv), and H2O (20.00 mL) at room temperature. The mixture was heated at 70 °C for 2.0 h. The reaction mixture was cooled to room temperature and was then filtered through a pad of Celite.
- Step 3 To a mixture of 3-bromo-4-(2,4-difluorophenoxy)aniline (2.51 g, 8.36 mmol, 1.00 equiv) and EtsN (3.39 g, 33.45 mmol, 4.00 equiv) in DCM (10.00 mL) a solution of ethane sulfonyl chloride (3.23 g, 25.09 mmol, 3.00 equiv) in DCM (10.00 mL) was added dropwise at 0 °C. The reaction mixture was stirred at room temperature for 3.0 h. The reaction was concentrated under vacuum, diluted with water (20 mL), and extracted with EA (20 mLx3).
- Step 4 A mixture of N-[3-bromo-4-(2,4-difluorophenoxy)phenyl]ethanesulfonamide (1.00 g, 2.55 mmol, 1.00 equiv) and ethyl 6-methyl-7-oxo-4-(4,4,5,5-tetramethyl-l,3,2-dioxaborolan-2-yl)-lH- pyrrolo[2,3-c]pyridine-2-carboxylate (970.00 mg, 2.80 mmol, 1.10 equiv), Pd(dtbpf)C12 (166.00 mg, 0.25 mmol, 0.10 equiv), and K3PO4 (1.10 g, 5.18 mmol, 2.03 equiv) in toluene (8.00 mL) and H2O (2.00 mL) was stirred for 1.0 h at 75 °C under nitrogen atmosphere.
- Step 5 The procedure was the same as 4- ⁇ 4-[3-( ⁇ 4-[(2R)-2-[(tert-butoxycarbonyl)amino]-4-( ⁇ l- methyl-4-[ 1 -methyl -4-(3 - ⁇ [ 1 -methyl -4-( 1 -methylimidazole-2-amido)pyrrol-2- yl]formamido ⁇ propanamido)imidazole-2-amido]pyrrol-2-yl ⁇ formamido)butanamido]-l-methylimidazol-2- yl ⁇ formamido)propanamido]-l-methylpyrrole-2-amido ⁇ -l-methylimidazole-2-carboxylic acid (Example 3 Step 2).
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Epidemiology (AREA)
- Pharmacology & Pharmacy (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Medicinal Chemistry (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
The present disclosure relates to transcription modulator molecules having a first terminus, a second terminus, and an oligomeric backbone and methods for treating Huntington's disease (HD).
Description
METHODS AND COMPOUNDS FOR MODULATING HUNTINGTON’S DISEASE CROSS REFERENCE
[0001] This application claims the benefit of U.S. Provisional Application No. 63/587,615 filed on October 3, 2023 which is hereby incorporated by reference in its entirety.
FIELD OF THE DISCLOSURE
[0002] Disclosed herein are new chimeric heterocyclic polyamide compounds and compositions and their application as pharmaceuticals for the treatment of disease. Methods to modulate the expression of a target gene comprising the CAG trinucleotide repeat sequence in a subject are also provided for the treatment of diseases such as Huntington’s disease (“HD”).
BACKGROUND OF THE DISCLOSURE
[0003] Huntington’s disease (“HD”) was first identified in the late 19th century as an autosomal dominant, neurodegenerative disorder. The symptoms of HD, which include a range of movement, cognitive and psychiatric disorders, generally appear in adulthood. HD is associated with the presence of the CAG trinucleotide repeat sequence in the huntingin gene (HTT), which codes for a protein termed huntingtin. Subjects with more than about 36 trinucleotide repeat sequences generally present with symptoms of HD, with a larger number of trinucleotide repeat sequences associated with an earlier onset of symptoms. Pathology stems from a cascade of steps: production of poly-Q huntingtin, followed by fragmentation of the elongated huntingtin into smaller peptides, which bind together and accumulate in neurons. The effects of this cascade are pronounced in the basal ganglia and cortex of the brain.
[0004] Huntington’s disease-like syndrome refers to a group of ailments whose symptoms are similar to those of Huntington’s disease, but which lack the characteristic mutation in the HTT gene. Huntington’s disease-like 2 syndrome (“HDL2”) is associated with a count of about 40 or more CAG trinucleotide repeat sequences in the junctophilin 3 Jph3) gene. HDL2 is a genetic disorder that has been seen in subjects with African lineage. Age of onset is inversely correlated with the number of trinucleotide repeat sequences. Symptoms of this syndrome include dystonia and chorea (uncontrolled movements), emotional disruptions, dysarthria, bradykinesia, inability to incorporate new learning, and difficulty in making decisions. Life expectancy can range from a few years post diagnosis to over a decade. The current theory holds that a poly- Q protein that is coded by the Jph3 gene forms aggregates in neuronal cells that is responsible for the pathology of the disease. However, evidence suggesting toxic gain-of-function of mRNA has also been uncovered, indicating a possible dual pathway for pathology.
[0005] In some embodiments, the mechanism set forth above provides opportunity for an effective treatment for a disease or disorder which is characterized by the presence of an excessive count of CAG trinucleotide repeat sequences in a target gene. In some embodiments, the pathology of the disease or disorder is due to the presence of mRNA containing an excessive count of CAG trinucleotide repeat sequences. In some embodiments, the pathology of the disease or disorder is due to the presence of a translation product containing an excessive count of glutamine amino acid residues. In some embodiments,
the pathology of the disease or disorder is due to a loss of function in the translation product. In some embodiments, the pathology of the disease or disorder is due to a gain of function in the translation product. In some embodiments, the pathology of the disease or disorder can be alleviated by increasing the rate of transcription of the defective gene. In some embodiments, the pathology of the disease or disorder can be alleviated by decreasing the rate of transcription of the defective gene.
SUMMARY OF THE DISCLOSURE
[0006] This disclosure utilizes regulatory molecules present in cell nuclei that control gene expression. Eukaryotic cells provide several mechanisms for controlling gene replication, transcription, and/or translation. Regulatory molecules that are produced by various biochemical mechanisms within the cell can modulate the various processes involved in the conversion of genetic information to cellular components. Several regulatory molecules are known to modulate the production of mRNA and, if directed to the target gene (such as, HTT), would modulate the production of the target gene mRNA that causes diseases such as, for example, Huntington’s disease or Huntington’s disease-like syndrome, and thus reverse the progress of these diseases.
[0007] Provided herein are transcription modulator molecules for recruiting a regulatory molecule into close proximity to the target gene. In some embodiments, provided herein is a transcription modulator molecule having a first terminus, a second terminus, and an oligomeric backbone moiety, or a pharmaceutically acceptable salt thereof, wherein: a) the first terminus comprises a DNA-binding moiety having the structure of Formula (A-l):
Formula (A-l), wherein:
W1 is hydrogen, optionally substituted C1-C10 alkyl, optionally substituted C1-C10 haloalkyl, - NRleRlf, -NRleC(O)Rlf, -NRleC(O)NRleRlf, -C(O)NRleRlf, -OC(O)NRleRlf, -NRleC(O)ORlf, - N=C(N(Rle)2)2. -ZB-PO(ORle)2, -ZB-(CH2)P3-PO(ORle)2, or -ZB-(CH2)p3-O-PO(ORle)2, wherein each Rle is independently hydrogen or optionally substituted Ci-Cw alkyl;
each Rlf is independently hydrogen, optionally substituted C1-C10 alkyl, optionally substituted C1-C10 heteroalkyl, AAP2, or optionally substituted 5-membered heteroaryl, wherein each AA is an amino acid and p2 is an integer from 1-10;
ZB is N or O; ps is 1-10;
W2 is optionally substituted C1-C20 alkyl, optionally substituted C1-C20 aminoalkyl, optionally substituted C1-C20 haloalkyl, optionally substituted C1-C20 heteroalkyl, or optionally substituted C1-C20 hydroxyalkyl; or W2 is L’-Z-R4; wherein
L1 is C1-C20 alkylene or C2-C20 heteroalkylene;
Z is absent, -C(O)-, or -C(=NH)-;
R4 is C1-C3 alkyl, -OR4b, or -NR4aR4b; wherein
R4a is hydrogen, optionally substituted C1-C20 alkyl, or optionally substituted C1-C20 heteroalkyl;
R4b is optionally substituted C1-C20 alkyl, optionally substituted C2-C20 alkenyl, optionally substituted C2-C20 alkynyl, optionally substituted C1-C20 aminoalkyl, optionally substituted C1-C20 haloalkyl, optionally substituted C1-C20 heteroalkyl, optionally substituted C1-C20 hydroxyalkyl, optionally substituted C3-C8 cycloalkyl, optionally substituted 4 to 8-membered heterocycloalkyl, optionally substituted phenyl, or optionally substituted 5 to 10-membered heteroaryl; or
R4a and R4b together with the nitrogen to which they are attached form an optionally substituted 3 to 8-membered heterocycloalkyl which is partially or fully saturated;
Rw is hydrogen or optionally substituted C1-C20 alkyl; or
Rw and W2 combine together with the atoms to which they are attached to form an optionally substituted 4 to 8-membered heterocycloalkyl; each Y1, Y2, Y3, Y4, Y5, Y6, Y7, and Y8 is independently N or CH; each X1, X2, X3, X4, X5, X6, X7, and X8 is independently S, O, or NR2; each R2 is independently hydrogen, optionally substituted C1-C50 alkyl, optionally substituted C2-C50 alkenyl, optionally substituted C2-C50 alkynyl, optionally substituted C1-C50 heteroalkyl, optionally substituted C2-C50 heteroalkenyl, optionally substituted C2-C50 heteroalkynyl, optionally substituted C1-C50 haloalkyl, optionally substituted C3-C8 cycloalkyl, optionally substituted 3 to 8-membered heterocycloalkyl, or optionally substituted PEG1.50; each R3 is independently hydrogen, halogen, Ci-Ce alkyl, -OR3c, -NR3cR3d, or -NHC(O)R3e, wherein R3c and R3d are each independently hydrogen, alkyl, or PEG;
R3e is alkyl, PEG, cycloalkyl, heterocycloalkyl, or phenyl; or two R3 together with the atoms to which they are attached form an optionally substituted C3-C6 cycloalkyl or 3 to 6 membered heterocycloalkyl; ji is 0 or 1;
no is 0 or 1 ; mi and are each independently an integer from 0-3; and pi is 3 or 4; wherein the linker is attached at one of R3 and the others of R3 are each hydrogen; or the linker is attached at the cycloalkyl or heterocycloalkyl formed by two R3; b) the second terminus comprises a protein-binding moiety capable of binding to a regulatory molecule that modulates expression of a gene; and c) the oligomeric backbone links the first terminus and the second terminus.
[0008] In some embodiments, the DNA binding moiety can selectively bind a trinucleotide repeat sequence in for example, HTT. The recruiting moiety, linked to the DNA binding moiety, will thus be held in proximity to the target gene; will recruit the regulatory molecule into proximity with the gene; and the regulatory molecule will modulate expression, and therefore counteract the production of defective target gene by direct interaction with the target gene. This mechanism may provide an effective treatment for HD, which is caused by the expression of defective HTT, where correction of the expression of the defective target gene thus represents an effective method for the treatment for these diseases.
[0009] The DNA binding moiety comprises a polyamide segment that will bind selectively to the target CAG sequence. Polyamides designed by for example Dervan (U.S. Patent Nos. 9,630,950 and 8,524,899) and others can selectively bind to selected DNA sequences. These polyamides sit in the minor groove of double helical DNA and form hydrogen bonding interactions with the Watson-Crick base pairs. Polyamides that selectively bind to particular DNA sequences can be designed by linking monoamide building blocks according to established chemical rules. One building block is provided for each DNA base pair, with each building block binding noncovalently and selectively to one of the DNA base pairs: A/T, T/A, G/C, and C/G. Following this guideline, trinucleotides bind to molecules with three amide units, i.e. tri -amides. In general, these polyamides can orient in either direction of a DNA sequence.
[0010] In principle, longer DNA sequences can be targeted with higher specificity and/or higher affinity by combining a larger number of monoamide building blocks into longer polyamide chains. Ideally, the binding affinity for a polyamide would simply be equal to the sum of each individual monoamide/DNA base pair interaction. In practice, however, due to the geometric mismatch between the fairly rigid polyamide and DNA structures, longer polyamide sequences do not bind to longer DNA sequences as tightly as would be expected from a simple additive contribution. The geometric mismatch between longer polyamide sequences and longer DNA sequences induces an unfavorable geometric strain that subtracts from the binding affinity than would be otherwise expected.
[0011] The disclosure provides for transcription modulator molecules that comprise a DNA binding moiety (for example a polyamide comprising multi-amine subunits) that is connected by a spacer (for example a linker moiety or oligomeric backbone) to a protein binding moiety. The spacer can alleviate the geometric strain that would otherwise decrease binding affinity of a larger polyamide sequence.
[0012] It should be understood, however, that the detailed description and the specific examples, while indicating specific embodiments, are given by way of illustration only, since various changes and modifications within the spirit and scope of the instant disclosure will become apparent to those skilled in the art from this detailed description.
INCORPORATION BY REFERENCE
[0013] All publications, patents, and patent applications mentioned in this specification are herein incorporated by reference to the same extent as if each individual publication, patent, or patent application was specifically and individually indicated to be incorporated by reference. To the extent publications and patents or patent applications incorporated by reference contradict the disclosure contained in the specification, the specification is intended to supersede and/or take precedence over any such contradictory material.
DETAILED DESCRIPTION
[0014] The disclosure provides for transcription modulator molecules that comprise a DNA binding moiety (for example, a polyamide comprising multi-amine subunits) connected by spacers (for example, a linker moiety or oligomeric backbone) to a protein binding moiety. The spacers can alleviate the geometric strain that would otherwise decrease binding affinity of a larger polyamide sequences.
[0015] Treatment of a subject with these compounds will modulate the expression of the defective target gene, and this can reduce the occurrence, severity, or frequency of symptoms associated with genetic disease (such as for example HD). The compounds described herein recruit the regulatory molecule to modulate the expression of the defective target gene and effectively treat and alleviate the symptoms associated with diseases.
Compounds - Transcription modulator molecules
[0016] The compounds disclosed herein are transcription modulator molecules. They possess useful activity for modulating the transcription of a target gene (e.g., HTT) and may be used in the treatment or prophylaxis of a disease or condition in which the target gene plays an active role. Thus, in broad aspects, some embodiments also provide pharmaceutical compositions comprising one or more compounds disclosed herein together with a pharmaceutically acceptable carrier, as well as methods of making and using the compounds and compositions.
[0017] In an aspect, provided herein is a transcription modulator molecule having a first terminus, a second terminus, and a linker moiety, wherein: a) the first terminus comprises a DNA-binding moiety; b) the second terminus comprises a protein-binding moiety capable of binding to a regulatory molecule that modulates expression of a gene; and c) the oligomeric backbone links the first terminus and the second terminus.
First terminus - DNA binding moiety
[0018] The first terminus interacts and binds with the target gene at the target DNA-sequence, particularly within the minor groove. In an aspect, the molecules disclosed herein provide a polyamide sequence.
[0019] In some embodiments, the DNA-binding moiety comprises a polyamide of one or more of the following subunits selected from
C(O)-, -NH-phenylene-C(O)-, -NH-pyridinylene-C(O)-, -NH-piperidinylene-C(O)-, -NH-pyrimidinylene-
R’ is independently hydrogen, optionally substituted C1-C20 alkyl, optionally substituted C1-C20 heteroalkyl, optionally substituted C1-C20 haloalkyl, or optionally substituted C1-C20 alkylamino; and Z is H, NH2, Ci-Ce alkyl, Ci-Ce haloalkyl, or Ci-Ce alkyl -NH2.
[0020] In some embodiments, the DNA-binding moiety comprises the structure of Formula (A-l’), or a pharmaceutically acceptable salt thereof:
Formula (A-T), wherein:
Z1 is absent, -O-, or -NH-.
W1 is hydrogen, optionally substituted C1-C10 alkyl, optionally substituted C1-C10 haloalkyl, -NRleRlf, - NRleC(O)Rlf, -NRleC(O)NRleRlf, -C(O)NRleRlf, -OC(O)NRleRlf, -NRleC(O)ORlf, -N=C(N(Rle)2)2, -ZB- PO(ORle)2, -ZB-(CH2)p3-PO(ORle)2, or -ZB-(CH2)P3-O-PO(ORle)2, wherein each Rle is independently hydrogen or optionally substituted C1-C10 alkyl; each Rlf is independently hydrogen, optionally substituted C1-C10 alkyl, optionally substituted Ci- Cio heteroalkyl, AAp2, or optionally substituted 5 -membered heteroaryl, wherein each AA is an amino acid and p2 is an integer from 1-10;
ZB is N or O;
P3 is 1-10;
W2is optionally substituted Ci-C2o alkyl, optionally substituted Ci-C2o aminoalkyl, optionally substituted Ci- C20 haloalkyl, optionally substituted Ci-C2o heteroalkyl, or optionally substituted Ci-C2o hydroxyalkyl; or W2is L'-Z-R4; wherein
L1 is Ci-C2o alkylene or C2-C2o heteroalkylene;
Z is absent, -C(O)-, or -C(=NH)-;
R4 is C1-C3 alkyl, -OR4b, or -NR4aR4b; wherein
R4a is hydrogen, optionally substituted Ci-C2o alkyl, or optionally substituted Ci-C2o heteroalkyl;
R4b is optionally substituted Ci-C2o alkyl, optionally substituted C2-C2o alkenyl, optionally substituted C2-C2o alkynyl, optionally substituted Ci-C2o aminoalkyl, optionally substituted Ci- C20 haloalkyl, optionally substituted Ci-C2o heteroalkyl, optionally substituted Ci-C2o hydroxyalkyl, optionally substituted C3-C8 cycloalkyl, optionally substituted 4 to 8-membered heterocycloalkyl, optionally substituted phenyl, or optionally substituted 5 to 10-membered heteroaryl; or
R4a and R4b together with the nitrogen to which they are attached form an optionally substituted 3 to 10-membered heterocycloalkyl which is partially or fully saturated;
Rw is hydrogen or optionally substituted C1-C20 alkyl; or
Rw and W2 combine together with the atoms to which they are attached to form an optionally substituted 4 to 10-membered heterocycloalkyl; each Y1, Y2, Y3, Y4, Y5, Y6, Y7, and Y8 is independently N or CH; each X1, X2, X3, X4, X5, X6, X7, and X8 is independently S, O, or NR2; each R2 is independently hydrogen, optionally substituted C1-C50 alkyl, optionally substituted C2-C50 alkenyl, optionally substituted C2-C50 alkynyl, optionally substituted C1-C50 heteroalkyl, optionally substituted C2-C50 heteroalkenyl, optionally substituted C2-C50 heteroalkynyl, optionally substituted C1-C50 haloalkyl, optionally substituted C3-C10 cycloalkyl, optionally substituted 3 to 10-membered heterocycloalkyl, or optionally substituted PEG1.50; each R3 is independently hydrogen, halogen, Ci-Cg alkyl, -OR3c, -NR3cR3d, or -NHC(O)R3e, wherein
R3c and R3d are each independently hydrogen, alkyl, or PEG;
R3e is alkyl, PEG, cycloalkyl, heterocycloalkyl, or phenyl; or two R3 together with the atoms to which they are attached form an optionally substituted C3-C6 cycloalkyl or 3 to 6-membered heterocycloalkyl; ji is 0 or 1; no is 0 or 1 ; mi and are each independently an integer from 0-3; and pi is 2, 3, or 4, provided that when Z1 is -O- or -NH- then pi is 2, and when Z1 is absent then pi is 3 or 4; wherein the linker is attached at one of R3 and the others of R3 are each hydrogen; or the linker is attached at the cycloalkyl or heterocycloalkyl formed by two R3.
[0021] In some embodiments of Formula (A-l’), Z1 is absent. In some embodiments, Z1 is -O-. In some embodiments, Z1 is -NH-.
[0022] In some embodiments, the DNA-binding moiety comprises the structure of Formula (A-l), or a pharmaceutically acceptable salt thereof:
Formula (A-l), wherein:
W1 is hydrogen, optionally substituted C1-C10 alkyl, optionally substituted C1-C10 haloalkyl, -NRleRlf, - NRleC(O)Rlf, -NRleC(O)NRleRlf, -C(O)NRleRlf, -OC(O)NRleRlf, -NRleC(O)ORlf, -N=C(N(Rle)2)2, -ZB- PO(ORle)2, -ZB-(CH2)p3-PO(ORle)2, or -ZB-(CH2)P3-O-PO(ORle)2, wherein each Rle is independently hydrogen or optionally substituted C1-C10 alkyl; each Rlf is independently hydrogen, optionally substituted C1-C10 alkyl, optionally substituted Ci- Cio heteroalkyl, AAp2, or optionally substituted 5 -membered heteroaryl, wherein each AA is an amino acid and p2 is an integer from 1-10;
ZB is N or O;
P3 is 1-10;
W2 is optionally substituted Ci-C2o alkyl, optionally substituted Ci-C2o aminoalkyl, optionally substituted Ci- C20 haloalkyl, optionally substituted Ci-C2o heteroalkyl, or optionally substituted Ci-C2o hydroxyalkyl; or W2 is L'-Z-R4; wherein
L1 is Ci-C2o alkylene or C2-C2o heteroalkylene;
Z is absent, -C(O)-, or -C(=NH)-;
R4 is C1-C3 alkyl, -OR4b, or -NR4aR4b; wherein
R4a is hydrogen, optionally substituted Ci-C2o alkyl, or optionally substituted Ci-C2o heteroalkyl;
R4b is optionally substituted Ci-C2o alkyl, optionally substituted C2-C2o alkenyl, optionally substituted C2-C2o alkynyl, optionally substituted Ci-C2o aminoalkyl, optionally substituted Ci- C20 haloalkyl, optionally substituted Ci-C2o heteroalkyl, optionally substituted Ci-C2o hydroxyalkyl, optionally substituted C3-C8 cycloalkyl, optionally substituted 4 to 8-membered heterocycloalkyl, optionally substituted phenyl, or optionally substituted 5 to 10-membered heteroaryl; or
R4a and R4b together with the nitrogen to which they are attached form an optionally substituted 3 to 10-membered heterocycloalkyl which is partially or fully saturated;
Rw is hydrogen or optionally substituted C1-C20 alkyl; or
Rw and W2 combine together with the atoms to which they are attached to form an optionally substituted 4 to 10-membered heterocycloalkyl; each Y1, Y2, Y3, Y4, Y5, Y6, Y7, and Y8 is independently N or CH; each X1, X2, X3, X4, X5, X6, X7, and X8 is independently S, O, or NR2; each R2 is independently hydrogen, optionally substituted C1-C50 alkyl, optionally substituted C2-C50 alkenyl, optionally substituted C2-C50 alkynyl, optionally substituted C1-C50 heteroalkyl, optionally substituted C2-C50 heteroalkenyl, optionally substituted C2-C50 heteroalkynyl, optionally substituted C1-C50 haloalkyl, optionally substituted C3-C10 cycloalkyl, optionally substituted 3 to 10-membered heterocycloalkyl, or optionally substituted PEG1.50; each R3 is independently hydrogen, halogen, Ci-Ce alkyl, -OR3c, -NR3cR3d, or -NHC(0)R3e, wherein
R3c and R3d are each independently hydrogen, alkyl, or PEG;
R3e is alkyl, PEG, cycloalkyl, heterocycloalkyl, or phenyl; or two R3 together with the atoms to which they are attached form an optionally substituted C3-C6 cycloalkyl or 3 to 6-membered heterocycloalkyl; ji is 0 or 1; no is 0 or 1 ; mi and are each independently an integer from 0-3; and pi is 3 or 4; wherein the linker is attached at one of R3 and the others of R3 are each hydrogen; or the linker is attached at the cycloalkyl or heterocycloalkyl formed by two R3.
[0023] In some embodiments of Formula (A-l’) or (A-l), each R3 is independently hydrogen, halogen, Ci-Ce alkyl, -OR3c, -NR3cR3d, or -NHC(O)R3e, wherein R3c and R3d are each independently hydrogen, alkyl, or PEG; and R3e is alkyl, PEG, cycloalkyl, heterocycloalkyl, or phenyl. In some embodiments, each R3 is independently -NR3cR3d. In some embodiments, each R3 is independently -NHC(O)R3e. In some embodiments, each R3 is independently hydrogen.
[0024] In some embodiments of Formula (A-l’) or (A-l), two R3 together with the atoms to which they are attached form an optionally substituted C3-C6 cycloalkyl or 3 to 6-membered heterocycloalkyl. In some embodiments, two R3 together with the atom(s) to which they are attached form a C3-C6 cycloalkyl. In some embodiments, two R3 together with the atom(s) to which they are attached form a 4 to 6-membered heterocycloalkyl. In some embodiments, two R3 together with the atom(s) to which they are attached form a 4-membered heterocycloalkyl. In some embodiments, two R3 together with the atom(s) to which they are attached form a 5 -membered heterocycloalkyl. In some embodiments, two R3 together with the atom(s) to which they are attached form a 6-membered heterocycloalkyl. In some embodiments, two R3 together with the atom(s) to which they are attached form a cyclopropyl, cyclobutyl, or cyclopentyl.
[0025] In some embodiments of Formula (A-l’) or (A-l), no is 1. In some embodiments, no is 0.
[0026] In some embodiments of Formula (A-l’) or (A-l), pi is 4. In some embodiments, pi is 3.
[0027] In some embodiments, the DNA-binding moiety comprises the structure of Formula (A-2), or a pharmaceutically acceptable salt thereof:
Formula (A-2), wherein:
W1 is hydrogen or -N=C(N(Rle)2)2, wherein each Rle is independently hydrogen or C1-C3 alkyl;
W2 is optionally substituted C1-C20 alkyl, optionally substituted C1-C20 aminoalkyl, optionally substituted Ci- C20 haloalkyl, optionally substituted C1-C20 heteroalkyl, or optionally substituted C1-C20 hydroxyalkyl; or W2 is L’-Z-R4; wherein
L1 is C1-C20 alkylene or C2-C20 heteroalkylene;
Z is absent, -C(O)-, or -C(=NH)-;
R4 is C1-C3 alkyl, -OR4b, or -NR4aR4b; wherein
R4a is hydrogen, optionally substituted C1-C20 alkyl, or optionally substituted C1-C20 heteroalkyl;
R4b is optionally substituted C1-C20 alkyl, optionally substituted C2-C20 alkenyl, optionally substituted C2-C20 alkynyl, optionally substituted C1-C20 aminoalkyl, optionally substituted C1-C20 haloalkyl, optionally substituted C1-C20 heteroalkyl, optionally substituted C1-C20 hydroxyalkyl, optionally substituted C’s-Cs cycloalkyl, optionally substituted 4 to 8- membered heterocycloalkyl, optionally substituted phenyl, or optionally substituted 5 to 10- membered heteroaryl; or
R4a and R4b together with the nitrogen to which they are attached form an optionally substituted 3 to 10-membered heterocycloalkyl which is partially or fully saturated;
Rw is hydrogen or optionally substituted C1-C20 alkyl; or
Rw and W2 combine together with the atoms to which they are attached to form an optionally substituted 4 to 10-membered heterocycloalkyl; each Y1, Y2, Y3, Y4, Y5, Y6, Y7, and Y8 is independently N or CH; each X1, X2, X3, X4, X5, X6, X7, and X8 is independently S, O, or NR2;
each R2 is independently hydrogen, optionally substituted C1-C50 alkyl, optionally substituted C2-C50 alkenyl, optionally substituted C2-C50 alkynyl, optionally substituted C1-C50 heteroalkyl, optionally substituted C2-C50 heteroalkenyl, optionally substituted C2-C50 heteroalkynyl, optionally substituted C1-C50 haloalkyl, optionally substituted C3-C10 cycloalkyl, optionally substituted 3 to 10-membered heterocycloalkyl, or optionally substituted PEG1.50; each R3a and R3b is independently hydrogen, halogen, Ci-Ce alkyl, -OR3c, -NR3cR3d, or -NHC(O)R3e, wherein R3c and R3d are each independently hydrogen, alkyl, or PEG;
R3e is alkyl, PEG, cycloalkyl, heterocycloalkyl, or phenyl; or two R3a or two R3b together with the carbon atom to which they are attached form a CAC, cycloalkyl or 4 to 6-membered heterocycloalkyl; ji is 0 or 1; and mi and are each independently an integer from 0-3; wherein the linker is attached at R3a or R3b; or at the cycloalkyl or heterocycloalkyl formed through the combination of two R3a or two R3b.
[0028] In some embodiments of Formula (A-l ’) or (A-l), each X1, X2, X3, X4, X5, X6, and X7 is independently -NR2. In some embodiments, each X1, X2, X3, X4, X5, X6, X7, and X8 is independently O. In some embodiments, each X1, X2, X3, X4, X5, X6, X7, and X8 is independently S.
[0029] In some embodiments of Formula (A-l’) or (A-l), each R2 is independently hydrogen, optionally substituted C1-C50 alkyl, optionally substituted C2-C50 alkenyl, optionally substituted C2-C50 alkynyl, optionally substituted C1-C50 heteroalkyl, optionally substituted C2-C50 heteroalkenyl, optionally substituted C2-C50 heteroalkynyl, optionally substituted C1-C50 haloalkyl, optionally substituted C3-C10 cycloalkyl, optionally substituted 3 to 10-membered heterocycloalkyl, or optionally substituted PEG1.50. In some embodiments, each R2 is independently hydrogen, optionally substituted C1-C20 alkyl, optionally substituted C2-C20 alkenyl, optionally substituted C2-C20 alkynyl, optionally substituted C1-C20 heteroalkyl, optionally substituted C2-C20 heteroalkenyl, optionally substituted C2-C20 heteroalkynyl, optionally substituted C1-C20 haloalkyl, optionally substituted C3-C8 cycloalkyl, optionally substituted 3 to 8-membered heterocycloalkyl, or optionally substituted PEG1.20. In some embodiments, each R2 is independently hydrogen, optionally substituted C1-C10 alkyl, optionally substituted C1-C10 heteroalkyl, optionally substituted C1-C10 haloalkyl, optionally substituted C3-C8 cycloalkyl, optionally substituted 3 to 8-membered heterocycloalkyl, or optionally substituted PEG1 10.
[0030] In some embodiments of Formula (A-l’) or (A-l), each R2 is independently an optionally substituted C1-C20 alkyl. In some embodiments, each R2 is independently an optionally substituted C1-C10 alkyl. In some embodiments, each R2 is independently methyl, ethyl, isopropyl, isobutyl, sec-butyl, or tertbutyl. In some embodiments, each R2 is independently hydrogen or methyl. In some embodiments, each R2 is ethyl. In some embodiments, each R2 is isopropyl. In some embodiments, each R2 is methyl. In some embodiments, each R2 is hydrogen.
[0031] In some embodiments of Formula (A-l’) or (A-l), ji is 1. In some embodiments, ji is 0.
[0032] In some embodiments, the DNA-binding moiety comprises a structure of Formula (A-3), or a pharmaceutically acceptable salt thereof:
Formula (A-3), wherein:
W1 is hydrogen or -N=C(N(Rle)2)2, wherein each Rle is independently hydrogen or C1-C3 alkyl;
W2 is optionally substituted C1-C20 alkyl, optionally substituted C1-C20 aminoalkyl, optionally substituted Ci- C20 haloalkyl, optionally substituted C1-C20 heteroalkyl, or optionally substituted C1-C20 hydroxyalkyl; or W2 is L'-Z-R4; wherein
L1 is C1-C20 alkylene or C2-C20 heteroalkylene;
Z is absent, -C(O)-, or -C(=NH)-;
R4 is C1-C3 alkyl, -OR4b, or -NR4aR4b; wherein
R4a is hydrogen, optionally substituted C1-C20 alkyl, or optionally substituted C1-C20 heteroalkyl;
R4b is optionally substituted C1-C20 alkyl, optionally substituted C2-C20 alkenyl, optionally substituted C2-C20 alkynyl, optionally substituted C1-C20 aminoalkyl, optionally substituted C1-C20 haloalkyl, optionally substituted C1-C20 heteroalkyl, optionally substituted C1-C20 hydroxyalkyl, optionally substituted C’s-Cs cycloalkyl, optionally substituted 4 to 8- membered heterocycloalkyl, optionally substituted phenyl, or optionally substituted 5 to 10- membered heteroaryl; or
R4a and R4b together with the nitrogen to which they are attached form an optionally substituted 3 to 10-membered heterocycloalkyl which is partially or fully saturated;
Rw is hydrogen or optionally substituted C1-C20 alkyl; or Rw and W2 combine together with the atoms to which they are attached to form an optionally substituted 4 to 10-membered heterocycloalkyl; each Y1, Y2, Y3, Y4, Y5, Y6, Y7, and Y8 is independently N or CH; each R2a, R2b, R2c, R2d, R2e, R2f, R2g, and R2h is independently hydrogen, optionally substituted C1-C50 alkyl, optionally substituted C2-C50 alkenyl, optionally substituted C2-C50 alkynyl, optionally substituted C1-C50
heteroalkyl, optionally substituted C2-C50 heteroalkenyl, optionally substituted C2-C50 heteroalkynyl, optionally substituted C1-C50 haloalkyl, optionally substituted C3-C10 cycloalkyl, optionally substituted 3 to 10-membered heterocycloalkyl, or optionally substituted PEG1.50; each of which is optionally substituted with one or more Rz; each Rz is independently -CN, -OH, -ORZa, -N3, -NRZaRzb, -CO(O)RZc, -C(O)ORZc, -C(O)NRZaRzb, - NHC(O)RZc, -NHC(O)ORZc, -OC(O)NRZaRzb, or optionally substituted 5 to 10-membered heteroaryl; wherein
RZa and Rzb are each independently hydrogen, alkyl, or PEG;
RZc is alkyl, PEG, cycloalkyl, heterocycloalkyl, or phenyl; each R3b is independently hydrogen, halogen, Ci-Cg alkyl, -OR3c, -NR3cR3d, or -NHC(O)R3e, wherein
R3c and R3d are each independently hydrogen, alkyl, or PEG;
R3e is alkyl, PEG, cycloalkyl, heterocycloalkyl, or phenyl; or two R3b together with the carbon atom to which they are attached form a C3-C6 cycloalkyl or 4 to 6- membered heterocycloalkyl; and and mi are each independently 0 or 1.
[0033] In some embodiments, the DNA-binding moiety comprises a structure of Formula (A-4), or a pharmaceutically acceptable salt thereof:
Formula (A-4), wherein:
W1 is hydrogen or -N=C(N(Rle)2)2, wherein each Rle is independently hydrogen or C1-C3 alkyl;
W2 is optionally substituted C1-C20 alkyl, optionally substituted C1-C20 aminoalkyl, optionally substituted Ci- C20 haloalkyl, optionally substituted C1-C20 heteroalkyl, or optionally substituted C1-C20 hydroxyalkyl; or W2 is L'-Z-R4; wherein
L1 is C1-C20 alkylene or C2-C20 heteroalkylene;
Z is absent, -C(O)-, or -C(=NH)-;
R4 is C1-C3 alkyl, -OR4b, or -NR4aR4b; wherein
R4a is hydrogen, optionally substituted C1-C20 alkyl, or optionally substituted C1-C20 heteroalkyl;
R4b is optionally substituted C1-C20 alkyl, optionally substituted C2-C20 alkenyl, optionally substituted C2-C20 alkynyl, optionally substituted C1-C20 aminoalkyl, optionally substituted C1-C20 haloalkyl, optionally substituted C1-C20 heteroalkyl, optionally substituted C1-C20 hydroxyalkyl, optionally substituted Cs-Cs cycloalkyl, optionally substituted 4 to 8- membered heterocycloalkyl, optionally substituted phenyl, or optionally substituted 5 to 10- membered heteroaryl; or
R4a and R4b together with the nitrogen to which they are attached form an optionally substituted 3 to 10-membered heterocycloalkyl which is partially or fully saturated;
Rw is hydrogen or optionally substituted C1-C20 alkyl; or Rw and W2 combine together with the atoms to which they are attached to form an optionally substituted 4 to 10-membered heterocycloalkyl; each Y1, Y2, Y3, Y4, Y5, Y6, Y7, and Y8 is independently N or CH; each R2a, R2b, R2c, R2d, R2e, R2f, R2g, and R2h is independently hydrogen, optionally substituted C1-C50 alkyl, optionally substituted C2-C50 alkenyl, optionally substituted C2-C50 alkynyl, optionally substituted C1-C50 heteroalkyl, optionally substituted C2-C50 heteroalkenyl, optionally substituted C2-C50 heteroalkynyl, optionally substituted C1-C50 haloalkyl, optionally substituted C3-C10 cycloalkyl, optionally substituted 3 to 10-membered heterocycloalkyl, or optionally substituted PEG1.50; each of which is optionally substituted with one or more Rz; each Rz is independently -CN, -OH, -ORZa, -N3, -NRZaRzb, -CO(O)RZc, -C(O)ORZc, -C(O)NRZaRzb, - NHC(O)RZc, -NHC(O)ORZc, -OC(O)NRZaRzb, or optionally substituted 5 to 10-membered heteroaryl; wherein
RZa and Rzb are each independently hydrogen, alkyl, or PEG;
RZc is alkyl, PEG, cycloalkyl, heterocycloalkyl, or phenyl; each R3a is independently hydrogen, halogen, Ci-Cg alkyl, -OR3c, -NR3cR3d, or -NHC(O)R3e, wherein
R3c and R3d are each independently hydrogen, alkyl, or PEG;
R3e is alkyl, PEG, cycloalkyl, heterocycloalkyl, or phenyl; or two R3a together with the carbon atom to which they are attached form a C3-C6 cycloalkyl or 4 to 6- membered heterocycloalkyl; and ni and mi are each independently 0 or 1.
[0034] In some embodiments, the DNA-binding moiety comprises a structure of Formula (A-5), or a pharmaceutically acceptable salt thereof:
Formula (A-5), wherein:
W1 is hydrogen or -N=C(N(Rle)2)2, wherein each Rle is independently hydrogen or C1-C3 alkyl;
W2 is optionally substituted C1-C20 alkyl, optionally substituted C1-C20 aminoalkyl, optionally substituted Ci- C20 haloalkyl, optionally substituted C1-C20 heteroalkyl, or optionally substituted C1-C20 hydroxyalkyl; or W2 is L'-Z-R4; wherein
L1 is C1-C20 alkylene or C2-C20 heteroalkylene;
Z is absent, -C(O)-, or -C(=NH)-;
R4 is C1-C3 alkyl, -OR4b, or -NR4aR4b; wherein
R4a is hydrogen, optionally substituted C1-C20 alkyl, or optionally substituted C1-C20 heteroalkyl;
R4b is optionally substituted C1-C20 alkyl, optionally substituted C2-C20 alkenyl, optionally substituted C2-C20 alkynyl, optionally substituted C1-C20 aminoalkyl, optionally substituted C1-C20 haloalkyl, optionally substituted C1-C20 heteroalkyl, optionally substituted C1-C20 hydroxyalkyl, optionally substituted C’s-Cs cycloalkyl, optionally substituted 4 to 8- membered heterocycloalkyl, optionally substituted phenyl, or optionally substituted 5 to 10- membered heteroaryl; or
R4a and R4b together with the nitrogen to which they are attached form an optionally substituted 3 to 10-membered heterocycloalkyl which is partially or fully saturated;
Rw is hydrogen or optionally substituted C1-C20 alkyl; or Rw and W2 combine together with the atoms to which they are attached to form an optionally substituted 4 to 10-membered heterocycloalkyl; each Y5 and Y6 is independently N or CH; each R2a, R2b, R2c, R2d, R2e, R2f, R2g, and R2h is independently hydrogen, optionally substituted C1-C50 alkyl, optionally substituted C2-C50 alkenyl, optionally substituted C2-C50 alkynyl, optionally substituted C1-C50 heteroalkyl, optionally substituted C2-C50 heteroalkenyl, optionally substituted C2-C50 heteroalkynyl, optionally substituted C1-C50 haloalkyl, optionally substituted C3-C10 cycloalkyl, optionally substituted 3
to 10-membered heterocycloalkyl, or optionally substituted PEG1.50; each of which is optionally substituted with one or more Rz; each Rz is independently -CN, -OH, -ORZa, -N3, -NRZaRzb, -CO(O)RZc, -C(O)ORZc, -C(O)NRZaRzb, - NHC(O)RZc, -NHC(O)ORZc, -OC(O)NRZaRzb, or optionally substituted 5 to 10-membered heteroaryl; wherein
RZa and Rzb are each independently hydrogen, alkyl, or PEG;
RZc is alkyl, PEG, cycloalkyl, heterocycloalkyl, or phenyl; and ni and mi are each independently 0 or 1.
[0035] In some embodiments, the DNA-binding moiety comprises a structure of Formula (A-6), or a pharmaceutically acceptable salt thereof:
Formula (A-6), wherein:
W1 is hydrogen or -N=C(N(Rle)2)2, wherein each Rle is independently hydrogen or C1-C3 alkyl;
W2 is optionally substituted C1-C20 alkyl, optionally substituted C1-C20 aminoalkyl, optionally substituted Ci- C20 haloalkyl, optionally substituted C1-C20 heteroalkyl, or optionally substituted C1-C20 hydroxyalkyl; or W2 is L’-Z-R4; wherein
L1 is C1-C20 alkylene or C2-C20 heteroalkylene;
Z is absent, -C(O)-, or -C(=NH)-;
R4 is C1-C3 alkyl, -OR4b, or -NR4aR4b; wherein
R4a is hydrogen, optionally substituted C1-C20 alkyl, or optionally substituted C1-C20 heteroalkyl;
R4b is optionally substituted C1-C20 alkyl, optionally substituted C2-C20 alkenyl, optionally substituted C2-C20 alkynyl, optionally substituted C1-C20 aminoalkyl, optionally substituted C1-C20 haloalkyl, optionally substituted C1-C20 heteroalkyl, optionally substituted C1-C20 hydroxyalkyl, optionally substituted C’s-Cs cycloalkyl, optionally substituted 4 to 8- membered heterocycloalkyl, optionally substituted phenyl, or optionally substituted 5 to 10- membered heteroaryl; or
R4a and R4b together with the nitrogen to which they are attached form an optionally substituted 3 to 10-membered heterocycloalkyl which is partially or fully saturated;
Rw is hydrogen or optionally substituted C1-C20 alkyl; or Rw and W2 combine together with the atoms to which they are attached to form an optionally substituted 4 to 10-membered heterocycloalkyl; each Y5 and Y6 is independently N or CH; each R2a, R2b, R2c, R2d, R2e, R2f, R2g, and R2h is independently hydrogen, optionally substituted C1-C50 alkyl, optionally substituted C2-C50 alkenyl, optionally substituted C2-C50 alkynyl, optionally substituted C1-C50 heteroalkyl, optionally substituted C2-C50 heteroalkenyl, optionally substituted C2-C50 heteroalkynyl, optionally substituted C1-C50 haloalkyl, optionally substituted C3-C10 cycloalkyl, optionally substituted 3 to 10-membered heterocycloalkyl, or optionally substituted PEG1.50; each of which is optionally substituted with one or more Rz; each Rz is independently -CN, -OH, -ORZa, -N3, -NRZaRzb, -CO(O)RZc, -C(O)ORZc, -C(O)NRZaRzb, - NHC(O)RZc, -NHC(O)ORZc, -OC(O)NRZaRzb, or optionally substituted 5 to 10-membered heteroaryl; wherein
RZa and Rzb are each independently hydrogen, alkyl, or PEG;
RZc is alkyl, PEG, cycloalkyl, heterocycloalkyl, or phenyl; and and mi are each independently 0 or 1.
[0036] In some embodiments of any of Formulas (A-l’) through (A-6), W2 is optionally substituted Ci- C20 alkyl, optionally substituted C1-C20 aminoalkyl, optionally substituted C1-C20 haloalkyl, optionally substituted C1-C20 heteroalkyl, or optionally substituted C1-C20 hydroxyalkyl. In some embodiments, W2 is optionally substituted C1-C20 alkyl or optionally substituted C1-C20 heteroalkyl. In some embodiments, W2 is optionally substituted C1-C20 alkyl. In some embodiments, W2 is optionally substituted C1-C20 heteroalkyl. In some embodiments, W2 is methyl, ethyl, n-propyl, or isopropyl.
[0037] In some embodiments of any of Formulas (A-l’) through (A-6), W2 is L’-Z-R4.
[0038] In some embodiments of any of Formula (A-l’) through (A-6), Rw is hydrogen. In some embodiments, Rw is optionally substituted C1-C20 alkyl.
[0039] In some embodiments of any of Formula (A-l’) through (A-6), W2 and Rw together with the nitrogen to which they are attached form an optionally substituted 4 to 10-membered heterocycloalkyl. In some embodiments, W2 and Rw together with the nitrogen to which they are attached form an optionally substituted 4-membered heterocycloalkyl. In some embodiments, W2 and Rw together with the nitrogen to which they are attached form an optionally substituted 5 -membered heterocycloalkyl. In some embodiments, W2 and Rw together with the nitrogen to which they are attached form an optionally substituted 6-membered heterocycloalkyl. In some embodiments, W2 and Rw together with the nitrogen to which they are attached form an optionally substituted 7-membered heterocycloalkyl.
[0040] In some embodiments, the DNA-binding moiety comprises a structure of Formula (A-7), or a pharmaceutically acceptable salt thereof:
Formula (A-7), wherein:
W1 is hydrogen or -N=C(N(Rle)2)2, wherein each Rle is independently hydrogen or C1-C3 alkyl; each Y1, Y2, Y3, Y4, Y5, Y6, Y7, and Y8 is independently N or CH; each R2a, R2b, R2c, R2d, R2e, R2f, R2g, and R2h is independently hydrogen, optionally substituted C1-C50 alkyl, optionally substituted C2-C50 alkenyl, optionally substituted C2-C50 alkynyl, optionally substituted C1-C50 heteroalkyl, optionally substituted C2-C50 heteroalkenyl, optionally substituted C2-C50 heteroalkynyl, optionally substituted C1-C50 haloalkyl, optionally substituted C3-C10 cycloalkyl, optionally substituted 3 to 10-membered heterocycloalkyl, or optionally substituted PEG1.50; each of which is optionally substituted with one or more Rz;
L1 is C1-C20 alkylene or C2-C20 heteroalkylene;
Z is absent, -C(O)-, or -C(=NH)-;
R4 is C1-C3 alkyl, -OR4b, or -NR4aR4b; wherein
R4a is hydrogen, optionally substituted C1-C20 alkyl, or optionally substituted C1-C20 heteroalkyl;
R4b is optionally substituted C1-C20 alkyl, optionally substituted C2-C20 alkenyl, optionally substituted C2-C20 alkynyl, optionally substituted C1-C20 aminoalkyl, optionally substituted C1-C20 haloalkyl, optionally substituted C1-C20 heteroalkyl, optionally substituted C1-C20 hydroxyalkyl, optionally substituted Cs-Cs cycloalkyl, optionally substituted 4 to 8-membered heterocycloalkyl, optionally substituted phenyl, or optionally substituted 5 to 10-membered heteroaryl; or
R4a and R4b together with the nitrogen to which they are attached form an optionally substituted 3 to 10- membered heterocycloalkyl which is partially or fully saturated; each Rz is independently -CN, -OH, -ORZa, -N3, -NRZaRzb, -CO(O)RZc, -C(O)ORZc, -C(O)NRZaRzb, - NHC(O)RZc, -NHC(O)ORZc, -OC(O)NRZaRzb, or optionally substituted 5 to 10-membered heteroaryl; wherein
RZa and Rzb are each independently hydrogen, alkyl, or PEG;
RZc is alkyl, PEG, cycloalkyl, heterocycloalkyl, or phenyl;
each R3b is independently hydrogen, halogen, Ci-Ce alkyl, -NR3cR3d, or -NHC(O)R3e, wherein
R3c and R3d are each independently hydrogen, alkyl, or PEG;
R3e is alkyl, PEG, cycloalkyl, heterocycloalkyl, or phenyl; or two R3b together with the carbon atom to which they are attached form a C’s-C,, cycloalkyl or 4 to 6- membered heterocycloalkyl; and ni and mi are each independently 0 or 1.
[0041] In some embodiments, the DNA-binding moiety comprises a structure of Formula (A-8), or a pharmaceutically acceptable salt thereof:
Formula (A-8), wherein:
W1 is hydrogen or -N=C(N(Rle)2)2, wherein each Rle is independently hydrogen or C1-C3 alkyl; each Y1, Y2, Y3, Y4, Y5, Y6, Y7, and Y8 is independently N or CH; each R2a, R2b, R2c, R2d, R2e, R2f, R2g, and R2h is independently hydrogen, optionally substituted C1-C50 alkyl, optionally substituted C2-C50 alkenyl, optionally substituted C2-C50 alkynyl, optionally substituted C1-C50 heteroalkyl, optionally substituted C2-C50 heteroalkenyl, optionally substituted C2-C50 heteroalkynyl, optionally substituted C1-C50 haloalkyl, optionally substituted C3-C10 cycloalkyl, optionally substituted 3 to 10-membered heterocycloalkyl, or optionally substituted PEG1.50; each of which is optionally substituted with one or more Rz;
L1 is C1-C20 alkylene or C2-C20 heteroalkylene;
Z is absent, -C(O)-, or -C(=NH)-;
R4 is C1-C3 alkyl, -OR4b, or -NR4aR4b; wherein
R4a is hydrogen, optionally substituted C1-C20 alkyl, or optionally substituted C1-C20 heteroalkyl;
R4b is optionally substituted C1-C20 alkyl, optionally substituted C2-C20 alkenyl, optionally substituted C2-C20 alkynyl, optionally substituted C1-C20 aminoalkyl, optionally substituted C1-C20 haloalkyl, optionally substituted C1-C20 heteroalkyl, optionally substituted C1-C20 hydroxyalkyl, optionally
substituted Cs-Cs cycloalkyl, optionally substituted 4 to 8-membered heterocycloalkyl, optionally substituted phenyl, or optionally substituted 5 to 10-membered heteroaryl; or
R4a and R4b together with the nitrogen to which they are attached form an optionally substituted 3 to 10- membered heterocycloalkyl which is partially or fully saturated; each Rz is independently -CN, -OH, -ORZa, -Ns, -NRZaRzb, -CO(O)RZc, -C(O)ORZc, -C(O)NRZaRzb, - NHC(O)RZc, -NHC(O)ORZc, -OC(O)NRZaRzb, or optionally substituted 5 to 10-membered heteroaryl; wherein
RZa and Rzb are each independently hydrogen, alkyl, or PEG;
RZc is alkyl, PEG, cycloalkyl, heterocycloalkyl, or phenyl; each R3a is independently hydrogen, halogen, Ci-Cg alkyl, -NR3cR3d, or -NHC(O)R3e, wherein
R3c and R3d are each independently hydrogen, alkyl, or PEG;
R3e is alkyl, PEG, cycloalkyl, heterocycloalkyl, or phenyl; or two R3a together with the carbon atom to which they are attached form a Cs-Ce cycloalkyl or 4 to 6- membered heterocycloalkyl; and and mi are each independently 0 or 1.
[0042] In some embodiments, the DNA-binding moiety comprises a structure of Formula (A-9), or a pharmaceutically acceptable salt thereof:
Formula (A-9), wherein:
W1 is hydrogen or -N=C(N(Rle)2)2, wherein each Rle is independently hydrogen or C1-C3 alkyl; each Y5 and Y6 is independently N or CH; each R2a, R2b, R2c, R2d, R2e, R2f, R2g, and R2h is independently hydrogen, optionally substituted C1-C50 alkyl, optionally substituted C2-C50 alkenyl, optionally substituted C2-C50 alkynyl, optionally substituted C1-C50 heteroalkyl, optionally substituted C2-C50 heteroalkenyl, optionally substituted C2-C50 heteroalkynyl, optionally substituted C1-C50 haloalkyl, optionally substituted C3-C10 cycloalkyl, optionally substituted 3
to 10-membered heterocycloalkyl, or optionally substituted PEG1.50; each of which is optionally substituted with one or more Rz;
L1 is C1-C20 alkylene or C2-C20 heteroalkylene;
Z is absent, -C(O)-, or -C(=NH)-;
R4 is C1-C3 alkyl, -OR4b, or -NR4aR4b; wherein
R4a is hydrogen, optionally substituted C1-C20 alkyl, or optionally substituted C1-C20 heteroalkyl;
R4b is optionally substituted C1-C20 alkyl, optionally substituted C2-C20 alkenyl, optionally substituted C2-C20 alkynyl, optionally substituted C1-C20 aminoalkyl, optionally substituted C1-C20 haloalkyl, optionally substituted C1-C20 heteroalkyl, optionally substituted C1-C20 hydroxyalkyl, optionally substituted Ci-C’s cycloalkyl, optionally substituted 4 to 8-membered heterocycloalkyl, optionally substituted phenyl, or optionally substituted 5 to 10-membered heteroaryl; or
R4a and R4b together with the nitrogen to which they are attached form an optionally substituted 3 to 10- membered heterocycloalkyl which is partially or fully saturated; each Rz is independently -CN, -OH, -ORZa, -N3, -NRZaRzb, -CO(O)RZc, -C(O)ORZc, -C(O)NRZaRzb, - NHC(O)RZc, -NHC(O)ORZc, -OC(O)NRZaRzb, or optionally substituted 5 to 10-membered heteroaryl; wherein
RZa and Rzb are each independently hydrogen, alkyl, or PEG;
RZc is alkyl, PEG, cycloalkyl, heterocycloalkyl, or phenyl; and and mi are each independently 0 or 1.
[0043] In some embodiments, the DNA-binding moiety comprises a structure of Formula (A- 10), or a pharmaceutically acceptable salt thereof:
Formula (A- 10), wherein:
W1 is hydrogen or -N=C(N(Rle)2)2, wherein each Rle is independently hydrogen or C1-C3 alkyl; each Y5 and Y6 is independently N or CH;
each R2a, R2b, R2c, R2d, R2e, R2f, R2g, and R2h is independently hydrogen, optionally substituted C1-C50 alkyl, optionally substituted C2-C50 alkenyl, optionally substituted C2-C50 alkynyl, optionally substituted C1-C50 heteroalkyl, optionally substituted C2-C50 heteroalkenyl, optionally substituted C2-C50 heteroalkynyl, optionally substituted C1-C50 haloalkyl, optionally substituted C3-C10 cycloalkyl, optionally substituted 3 to 10-membered heterocycloalkyl, or optionally substituted PEG1.50; each of which is optionally substituted with one or more Rz;
L1 is C1-C20 alkylene or C2-C20 heteroalkylene;
Z is absent, -C(O)-, or -C(=NH)-;
R4 is C1-C3 alkyl, -OR4b, or -NR4aR4b; wherein
R4a is hydrogen, optionally substituted C1-C20 alkyl, or optionally substituted C1-C20 heteroalkyl;
R4b is optionally substituted C1-C20 alkyl, optionally substituted C2-C20 alkenyl, optionally substituted C2-C20 alkynyl, optionally substituted C1-C20 aminoalkyl, optionally substituted C1-C20 haloalkyl, optionally substituted C1-C20 heteroalkyl, optionally substituted C1-C20 hydroxyalkyl, optionally substituted C3-C8 cycloalkyl, optionally substituted 4 to 8-membered heterocycloalkyl, optionally substituted phenyl, or optionally substituted 5 to 10-membered heteroaryl; or
R4a and R4b together with the nitrogen to which they are attached form an optionally substituted 3 to 10- membered heterocycloalkyl which is partially or fully saturated; each Rz is independently -CN, -OH, -ORZa, -N3, -NRZaRzb, -CO(O)RZc, -C(O)ORZc, -C(O)NRZaRzb, - NHC(O)RZc, -NHC(O)ORZc, -OC(O)NRZaRzb, or optionally substituted 5 to 10-membered heteroaryl; wherein
RZa and Rzb are each independently hydrogen, alkyl, or PEG;
RZc is alkyl, PEG, cycloalkyl, heterocycloalkyl, or phenyl; and and mi are each independently 0 or 1.
[0044] In some embodiments of any of Formulas (A-l’), ( A-l), (A-2), (A-3), (A-4), (A-7), or (A-8), Y2, Y4, and Y7 are each independently N; and Y1 and Y3 are each independently CH.
[0045] In some embodiments of any of Formula (A-l’ ) through (A- 10, each Y5 is independently CH. In some embodiments, each Y5 is independently N.
[0046] In some embodiments of any of Formula (A-l’) through (A- 10), each Y6 is independently CH. In some embodiments, each Y6 is independently N.
[0047] In some embodiments of any of Formula (A-l’), ( A-l), (A-2), (A-3), (A-4), (A-7), or (A-8), Y8 is CH. In some embodiments, Y8 is N.
[0048] In some embodiments of any of Formula (A-3) through (A- 10), each R2a, R2b, R2c, R2d, R2e, R2f, R2g, and R2h is independently hydrogen, optionally substituted C1-C50 alkyl, optionally substituted C2-C50 alkenyl, optionally substituted C2-C50 alkynyl, optionally substituted C1-C50 heteroalkyl, optionally substituted C2-C50 heteroalkenyl, optionally substituted C2-C50 heteroalkynyl, optionally substituted C1-C50 haloalkyl, optionally substituted C3-C10 cycloalkyl, optionally substituted 3 to 10-membered heterocycloalkyl, or optionally substituted PEG1.50; each of which is optionally substituted with one or more
Rz. In some embodiments, each R2a, R2b, R2c, R2d, R2e, R2f, R2g, and R2h is independently hydrogen, optionally substituted C1-C50 alkyl, optionally substituted C2-C50 alkenyl, optionally substituted C2-C50 alkynyl, optionally substituted C1-C50 heteroalkyl, optionally substituted C2-C50 heteroalkenyl, optionally substituted C2-C50 heteroalkynyl, optionally substituted C1-C50 haloalkyl, optionally substituted C3-G cycloalkyl, optionally substituted 3 to 8-membered heterocycloalkyl, or optionally substituted PEG1.50. In some embodiments, each R2a, R2b, R2c, R2d, R2e, R2f, R2g, and R2h is independently hydrogen, optionally substituted C1-C20 alkyl, optionally substituted C2-C20 alkenyl, optionally substituted C2-C20 alkynyl, optionally substituted C1-C20 heteroalkyl, optionally substituted C2-C20 heteroalkenyl, optionally substituted C2-C20 heteroalkynyl, optionally substituted C1-C20 haloalkyl, optionally substituted Cs-Cs cycloalkyl, optionally substituted 3 to 8-membered heterocycloalkyl, or optionally substituted PEGI.20. In some embodiments, each R2a, R2b, R2c, R2d, R2e, R2f, R2g, and R2h is independently optionally substituted C1-C10 alkyl, optionally substituted C1-C10 heteroalkyl, optionally substituted C1-C10 haloalkyl, optionally substituted Cs-Cs cycloalkyl, optionally substituted 3 to 8-membered heterocycloalkyl, or optionally substituted PEG1 10. In some embodiments, each R2a, R2b, R2c, R2d, R2e, R2f, R2g, and R2h is independently optionally substituted C1-C10 alkyl, optionally substituted C1-C10 haloalkyl, optionally substituted Cs-Cs cycloalkyl, or optionally substituted 3 to 8-membered heterocycloalkyl. In some embodiments, each R2a, R2b, R2c, R2d, R2e, R2f, R2g, and R2h is independently optionally substituted C1-C10 alkyl or optionally substituted Cs-Cs cycloalkyl. In some embodiments, the cycloalkyl or heterocycloalkyl is monocyclic, bicyclic, or spirocyclic.
[0049] In some embodiments of any of Formula (A-3) through (A- 10), each R2a, R2b, R2c, R2d, R2e, R2f, R2g, and R2h is independently an optionally substituted C1-C20 alkyl. In some embodiments, each R2a, R2b, R2c, R2d, R2e, R2f, R2g, and R2h is independently an optionally substituted C1-C10 alkyl. In some embodiments, each R2a, R2b, R2c, R2d, R2e, R2f, R2g, and R2h is independently methyl, ethyl, isopropyl, isobutyl, sec-butyl, or tert-butyl. In some embodiments, each R2 is independently hydrogen or methyl. In some embodiments, each R2a, R2b, R2c, R2d, R2e, R2f, R2g, and R2h is ethyl. In some embodiments, each R2a, R2b, R2c, R2d, R2e, R2f, R2g, and R2h is isopropyl. In some embodiments, each R2a, R2b, R2c, R2d, R2e, R2f, R2g, and R2h is methyl. In some embodiments, each R2a, R2b, R2c, R2d, R2e, R2f, R2g, and R2h is hydrogen.
[0050] In some embodiments Formula (A-2), (A-4), or (A-8), each R3a is independently hydrogen, halogen, -OR3c, -NR3cR3d, or -NHC(O)R3e, wherein R3c and R3d are each independently hydrogen, alkyl, or PEG; and R3e is alkyl, PEG, cycloalkyl, heterocycloalkyl, or phenyl. In some embodiments, each R3a is independently -NR3cR3d. In some embodiments, each R3a is independently -NHC(O)R3e. In some embodiments, each R3a is -NH2. In some embodiments, each R3a is hydrogen.
[0051] In some embodiments Formula (A-2), (A-3), or (A-7), each R3b is independently hydrogen, halogen, -OR3c, -NR3cR3d, or -NHC(O)R3e, wherein R3c and R3d are each independently hydrogen, alkyl, or PEG; and R3e is alkyl, PEG, cycloalkyl, heterocycloalkyl, or phenyl. In some embodiments, each R3b is independently -NR3cR3d. In some embodiments, each R3b is independently -NHC(O)R3e. In some embodiments, each R3b is -NH2. In some embodiments, each R3b is hydrogen.
[0052] In some embodiments of Formula (A-2), (A-4), or (A-8), two R3a together with the atoms to which they are attached combine together to form an optionally substituted C3-C, cycloalkyl or 3 to 6-membered heterocycloalkyl. In some embodiments, two R3a together with the atom(s) to which they are attached form a C’s-C,, cycloalkyl. In some embodiments, two R3a together with the atom(s) to which they are attached form a 4 to 6-membered heterocycloalkyl. In some embodiments, two R3a together with the atom(s) to which they are attached form a 4-membered heterocycloalkyl. In some embodiments, two R3a together with the atom(s) to which they are attached form a 5 -membered heterocycloalkyl. In some embodiments, two R3a together with the atom(s) to which they are attached form a 6-membered heterocycloalkyl. In some embodiments, two R3a together with the atom(s) to which they are attached form a cyclopropyl, cyclobutyl, or cyclopentyl.
[0053] In some embodiments of Formula (A-2), (A-3), or (A-7), two R3b together with the atoms to which they are attached combine together to form an optionally substituted Cs-Ce cycloalkyl or 3 to 6-membered heterocycloalkyl. In some embodiments, two R3b together with the atom(s) to which they are attached form a C’s-C,, cycloalkyl. In some embodiments, two R3b together with the atom(s) to which they are attached form a 4 to 6-membered heterocycloalkyl. In some embodiments, two R3b together with the atom(s) to which they are attached form a 4-membered heterocycloalkyl. In some embodiments, two R3b together with the atom(s) to which they are attached form a 5 -membered heterocycloalkyl. In some embodiments, two R3b together with the atom(s) to which they are attached form a 6-membered heterocycloalkyl. In some embodiments, two R3b together with the atom(s) to which they are attached form a cyclopropyl, cyclobutyl, or cyclopentyl.
[0054] In some embodiments of Formula (A-2), the linker is attached at R3a.
[0055] In some embodiments of Formula (A-2), the linker is attached at R3b.
[0056] In some embodiments of Formula (A-2), the linker is attached at the cycloalkyl or heterocycloalkyl formed through the combination of two R3a or two R3b
[0057] In some embodiments of any of Formulas (A-2), (A-3), (A-4), (A-7), or (A-8), R3c and R3d are each independently hydrogen, alkyl, or PEG. In some embodiments, R3c and R3d are each independently hydrogen, Ci-C2oalkyl, or PEG 1-20. In some embodiments, R and R are each independently Ci- C2oalkyl. In some embodiments, R3c and R3d are each independently hydrogen.
[0058] In some embodiments of any of Formulas (A-2), (A-3), (A-4), (A-7), or (A-8), R3e is alkyl, PEG, cycloalkyl, heterocycloalkyl, or phenyl. In some embodiments, R3e is C1-C20 alkyl, PEG1.20, C3-C, cycloalkyl, 4 to 6-membered heterocycloalkyl, or phenyl. In some embodiments, R3e is C1-C20 alkyl or PEGi. 20. In some embodiments, R3e is C3-C, cycloalkyl, 4 to 6-membered heterocycloalkyl, or phenyl. In some embodiments, R3e is C1-C20 alkyl.
[0059] In some embodiments of any of Formulas (A-E) through (A-10), L1 is C1-C10 alkylene or C2-C10 heteroalkylene. In some embodiments, L1 is C1-C10 alkylene, Ci-Cs alkylene, Ci-Ce alkylene, C1-C5 alkylene, C1-C4 alkylene, C1-C3 alkylene, or C1-C2 alkylene. In some embodiments, L1 is C1-C4 alkylene. In some embodiments, L1 is C1-C3 alkylene. In some embodiments, L1 is C1-C2 alkylene. In some embodiments, L1 is C2-C10 heteroalkylene, C2-C8 heteroalkylene, C2-C6 heterolkylene, C2-C5 heteroalkylene, or C2-C4 heteroalkylene. In some embodiments, L1 is C2-C10 heteroalkylene. In some embodiments, L1 is C2-C8
heteroalkylene. In some embodiments, L1 is C2-C6 heterolkylene. In some embodiments, L1 is C2-C5 heteroalkylene. In some embodiments, L1 is C2-C4 heteroalkylene.
[0060] In some embodiments of any of Formula (A-E) through (A- 10), the heteroalkylene is polyethylene glycol. In some embodiments, L1 is PEG1.10. In some embodiments, L1 is PEGi s. In some embodiments, L1 is -(CIECFE-C ys-, wherein ys is an integer in the range of 1-10. In some embodiments, ys is an integer in the range of 1-8. In some embodiments, ys is an integer in the range of 1-6. In some embodiments, ys is an integer in the range of 1-4. In some embodiments, ys is 1-2.
[0061] In some embodiments of any of Formulas (A-E) through (A- 10), the heteroalkylene comprises - (CH2) y6N(Ra)(CH2)y7- , wherein Ra is hydrogen or an optionally substituted Ci-Ce alkyl; and each and y7 is independently an integer in the range of 1-6.
[0062] In some embodiments of any of Formulas (A-E) through (A-10), Z is -C(O)-; and R4 is -OR4b. In some embodiments, Z is -C(O)-; and R4 is -NR4aR4b. In some embodiments, Z is -C(=NH)-; and R4 is - NR4aR4b. In some embodiments, Z is absent; and R4 is -OR4b. In some embodiments, Z is absent; and R4 is - NR4aR4b. In some embodiments, Z is -C(O)-; and R4 is Ci-Ce alkyl.
[0063] In some embodiments of any of Formulas (A-E) through (A-10), Z is absent. In some embodiments, Z is -C(O)-. In some embodiments, Z is -C(=NH)-.
[0064] In some embodiments of any of Formulas (A-E) through (A-10), R4 is C1-C3 alkyl or -NR4aR4b. In some embodiments, R4 is C1-C3 alkyl. In some embodiments, R4 is -NR4aR4b. In some embodiments, R4 is - OR4b.
[0065] In some embodiments of any of Formulas (A-E) through (A-10), R4a is hydrogen, optionally substituted C1-C20 alkyl, or optionally substituted C1-C20 heteroalkyl. In some embodiments, R4a is an optionally substituted C1-C20 alkyl or optionally substituted C1-C20 heteroalkyl. In some embodiments, R4a is an optionally substituted C1-C20 alkyl. In some embodiments, R4a is an optionally substituted C1-C20 heteroalkyl. In some embodiments, R4a is hydrogen.
[0066] In some embodiments of any of Formulas (A-E) through (A-10), R4b is optionally substituted Ci- C20 alkyl, optionally substituted C2-C20 alkenyl, optionally substituted C2-C20 alkynyl, optionally substituted C1-C20 aminoalkyl, optionally substituted C1-C20 haloalkyl, optionally substituted C1-C20 heteroalkyl, optionally substituted C1-C20 hydroxyalkyl, optionally substituted C’s-Cs cycloalkyl, optionally substituted 4 to 8-membered heterocycloalkyl, optionally substituted phenyl, or optionally substituted 5 to 10-membered heteroaryl. In some embodiments, R4b is optionally substituted C1-C20 alkyl, optionally substituted C1-C20 aminoalkyl, optionally substituted C1-C20 haloalkyl optionally substituted C1-C20 heteroalkyl, or optionally substituted C1-C20 hydroxyalkyl. In some embodiments, R4b is optionally substituted C1-C20 alkyl or optionally substituted C1-C20 heteroalkyl. In some embodiments, R4b is optionally substituted C1-C10 alkyl. In some embodiments, R4b is optionally substituted C1-C10 heteroalkyl. In some embodiments, R4b is optionally substituted C1-C10 haloalkyl.
[0067] In some embodiments of any of Formula (A-E) through (A-10), R4a is hydrogen; and R4b is C1-C10 alkyl or C1-C10 haloalkyl. In some embodiments, R4a is hydrogen; and R4b is C1-C10 alkyl. In some
embodiments of, R4a is hydrogen; and R4b is C1-C10 alkyl or C1-C10 haloalkyl. In some embodiments, R4a is hydrogen; and R4b is methyl or ethyl.
[0068] In some embodiments of any of Formula (A-T) through (A-10), R4a is C1-C10 alkyl and R4b is Ci- Cio alkyl. In some embodiments of any of Formula (A-T) through (A-10), R4a is methyl and R4b is methyl. [0069] In some embodiments of any of Formula (A-T) through (A-10), R4a and R4b together with the nitrogen to which they are attached form an optionally substituted 4 to 10-membered heterocycloalkyl which is partially or fully saturated. In some embodiments, R4a and R4b together with the nitrogen to which they are attached form an optionally substituted 4 to 10-membered heterocycloalkyl, wherein the heterocycloalkyl is a monocyclic, bicyclic, or spirocyclic heterocycloalkyl. In some embodiments, R4a and R4b together with the nitrogen to which they are attached form an optionally substituted 4-membered heterocycloalkyl. In some embodiments, R4a and R4b together with the nitrogen to which they are attached form an optionally substituted 5 -membered heterocycloalkyl. In some embodiments, R4a and R4b together with the nitrogen to which they are attached form an optionally substituted 6-membered heterocycloalkyl. In some embodiments, R4a and R4b together with the nitrogen to which they are attached form a piperidine, piperazine, or morpholine. In some embodiments, R4a and R4b together with the nitrogen to which they are attached form a piperidine. In some embodiments, R4a and R4b together with the nitrogen to which they are attached form a piperazine.
[0070] In some embodiments of any of Formula (A-T) through (A-10), R4a is hydrogen or Ci-Cw alkyl and R4b is Ci-Cw alkyl; or R4a and R4b together with the nitrogen to which they are attached form a 4 to 6- membered heterocycloalkyl.
[0071] In some embodiments of any of Formulas (A-3) through (A-10), each Rz is independently -CN, - OH, -ORZa, -N3, -NRZaRzb, -CO(O)RZc, -C(O)ORZc, -C(O)NRZaRzb, -NHC(O)RZc, -NHC(O)ORZc, - OC(O)NRZaRzb, or optionally substituted 5 to 10-membered heteroaryl. In some embodiments, each Rz is independently -CN, -OH, -ORZa, -Ns, or -NRZaRzb. In some embodiments, each Rz is independently - C(O)NRZaRzb, -NHC(O)RZc, or -OC(O)NRZaRzb. In some embodiments, each Rz is independently an optionally substituted 5 to 10-membered heteroaryl.
[0072] In some embodiments of any of Formulas (A-3) through (A-10), each RZa and Rzb is independently hydrogen, alkyl, or PEG. In some embodiments, each RZa and Rzb is independently hydrogen, C1-C20 alkyl, or PEG1.20. In some embodiments, each RZa and Rzb is independently C1-C20 alkyl. In some embodiments, each RZa and Rzb is independently hydrogen.
[0073] In some embodiments of Formula (A-3) through (A-10) RZc is alkyl, PEG, cycloalkyl, heterocycloalkyl, or phenyl. In some embodiments, RZc is C1-C20 alkyl, PEG1.20, C3-Ce cycloalkyl, 4 to 6- membered heterocycloalkyl, or phenyl. In some embodiments, RZc is C1-C20 alkyl or PEG1.20. In some embodiments, RZc is C1-C20 alkyl.
[0074] In some embodiments of Formula (A-I’) or (A-l), W1 is hydrogen, optionally substituted C1-C10 alkyl, -NRleC(O)Rlf, -NRleC(O)NRleRlf, -C(O)NRleRlf, -OC(O)NRleRlf, or -NRleC(O)ORlf. In some
embodiments, W1 is hydrogen or optionally substituted C1-C10 alkyl. In some embodiments, W1 is - NRleC(O)Rlf, -NRleC(O)NRleRlf, -C(O)NRleRlf, -OC(O)NRleRlf, or -NRleC(O)ORlf.
[0075] In some embodiments of Formula (A-l’) or (A-l), W1 is -ZB-PO(OR16)2, -ZB-(CH2)P3-PO(ORle)2 , or -ZB-(CH2)p3-O-PO2(ORle)2, wherein ZB is O or N, and p3 is 1-10.
[0076] In some embodiments of any of Formulas (A-T) through (A- 10), W1 is hydrogen or - N=C(N(Rle)2)2, wherein each Rle is independently hydrogen or C1-C3 alkyl. In some embodiments, W1 is hydrogen or -N=C(N(Rle)2)2, wherein each Rle is independently hydrogen or methyl.
[0077] In some embodiments of any of Formulas (A-T) through (A- 10), W1 is hydrogen.
[0078] In some embodiments of Formula (A-T) or (A-l), each AA is a natural or unnatural amino acid. [0079] In some embodiments of any of Formula (A-T) through (A- 10), each Rle is independently hydrogen or C1-C10 alkyl. In some embodiments, each Rle is independently hydrogen or Ci-Cs alkyl. In some embodiments, each Rle is independently hydrogen or Ci-Ce alkyl. In some embodiments, each Rle is independently hydrogen or C1-C3 alkyl. In some embodiments, each Rle is C1-C3 alkyl. In some embodiments, each Rle is independently methyl. In some embodiments, each Rle is independently hydrogen. [0080] In some embodiments of Formula (A-T) or (A-l), each Rlf is independently hydrogen, optionally substituted C1-C10 alkyl, optionally substituted C1-C10 heteroalkyl, or optionally substituted 5-membered heteroaryl. In some embodiments, each Rlf is independently hydrogen or optionally substituted C1-C10 alkyl. In some embodiments, each Rlf is independently hydrogen or optionally substituted Ci-Cs alkyl. In some embodiments, each Rlf is independently hydrogen or optionally substituted Ci-Ce alkyl. In some embodiments, each Rlf is independently optionally substituted Ci-Ce alkyl. In some embodiments, each Rlf is independently methyl. In some embodiments, each Rlf is independently hydrogen.
[0081] In some embodiments of any of Formulas (A-T) through (A-l), no is 0. In some embodiments, no is 1.
[0082] In some embodiments of any of Formulas (A-T) through (A-10), mi is 0. In some embodiments, mi is 1. In some embodiments, mi is 2. In some embodiments, mi is 3.
[0083] In some embodiments of any of Formula (A-T) through (A-10), is 0. In some embodiments, is 1. In some embodiments, is 2. In some embodiments, is 3.
[0084] In some embodiments of Formula (A-T) or (A-l), p2 is 1-10. In some embodiments, P2 is 1-8. In some embodiments, P2 is 1-6. In some embodiments, P2 is 1-4. In some embodiments, P2 is 1, 2, 3, or 4. In some embodiments, P2 is 1. In some embodiments, P2 is 2. In some embodiments, P2 is 3. In some embodiments, P2 is 4.
[0085] In some embodiments of Formula (A-T) or (A-l), p3 is 1-10. In some embodiments, p3 is 1-8. In some embodiments, p3 is 1-6. In some embodiments, p3 is 1-4. In some embodiments, p3 is 1, 2, 3, or 4. In some embodiments, p3 is 1. In some embodiments, p3 is 2. In some embodiments, p3 is 3. In some embodiments, P3 is 4.
[0086] The binding affinity between the polyamide and the target gene can be adjusted based on the composition of the polyamide. In some embodiments, the polyamide is capable of binding the DNA with an
affinity of less than about 600 nM, about 500 nM, about 400 nM, about 300 nM, about 250 nM, about 200 nM, about 150 nM, about 100 nM, or about 50nM. In some embodiments, the polyamide is capable of binding the DNA with an affinity of less than about 300 nM. In some embodiments, the polyamide is capable of binding the DNA with an affinity of less than about 200 nM. In some embodiments, the polyamide is capable of binding the DNA with an affinity of greater than about 200 nM, about 150 nM, about 100 nM, about 50 nM, about 10 nM, or about 1 nM. In some embodiments, the polyamide is capable of binding the DNA with an affinity in the range of about 1-600 nM, 10-500 nM, 20-500 nM, 50-400 nM, or 100-300 nM.
[0087] In some embodiments, the first terminus is capable of binding the DNA with an affinity of less than 500 nM.
[0088] The binding affinity between the polyamide and the target DNA can be determined using a quantitative footprint titration experiment. The experiment involves measuring the dissociation constant Kd of the polyamide for the target sequence at either 24 °C or 37 °C, and using either standard polyamide assay solution conditions or approximate intracellular solution conditions.
[0089] The binding affinity between the regulatory protein and the ligand on the second terminus can be determined using an assay suitable for the specific protein. The experiment involves measuring the dissociation constant Kd of the ligand for the protein and using either standard protein assay solution conditions or approximate intracellular solution conditions.
[0090] The polyamide composed of a pre-selected combination of subunits can selectively bind to the DNA in the minor groove. In their hairpin structure, antiparallel side-by-side pairings of two aromatic amino acids bind to DNA sequences, with a polyamide ring packed specifically against each DNA base.
Second Terminus -Regulatory Protein Binding Moiety
[0091] In some embodiments, the second terminus comprises a protein-binding moiety capable of binding to a regulatory molecule that modulates expression of a gene having the expanded nucleotide repeat.
[0092] In some embodiments, the second terminus comprises a bromodomain binding moiety. [0093] In some embodiments, the second terminus comprises a moiety capable of binding to a bromodomain and extra terminal domain (BET) family member.
[0094] In some embodiments, the BET family member is BRD2, BRD3, BRD4, or BRDT. In some embodiments, the BET family member is BRD2. In some embodiments, the BET family member is BRD3. In some embodiments, the BET family member is BRD4. In some embodiments, the BET family member is BRDT.
[0095] In some embodiments, the protein-binding moiety binds to CBP/p300, PCAF (P300/CBP- Associated Factor), CECR2 (cat eye syndrome chromosome region candidate 2), BRPF (bromodomain and PHD finger-containing protein), ATAD2/ATAD2B (chromatin remodeling proteins), TRIM24 (Tripartite motif-containing 24), BAZ2 (Bromodomain Adjacent to Zinc finger), TAF1 (TBP associated factors), BRD7/9, BPTF (Bromodomain PHD Finger Transcription Factor), SMARCA2/4, or PBRM1.
[0096] In some embodiments, the regulatory molecule is CBP/p300.
[0097] In some embodiments, the regulatory molecule is PCAF (P300/CBP-Associated Factor).
[0098] In some embodiments, the regulatory molecule is CECR2 (cat eye syndrome chromosome region candidate 2).
[0099] In some embodiments, the regulatory molecule is BRPF (bromodomain and PHD fingercontaining protein).
[00100] In some embodiments, the regulatory molecule is a ATAD2 or ATAD2B chromatin remodeling protein.
[00101] In some embodiments, the regulatory molecule is BAZ2 (Bromodomain Adjacent Zinc Finger.
[00102] In some embodiments, the regulatory molecule is TAF1 (TBP associated factor).
[00103] In some embodiments, the regulatory molecule is TRIM24 (tripartite motif-containing 24).
[00104] In some embodiments, the regulatory molecule is BRD7/9.
[00105] In some embodiments, the regulatory molecule is BPTF (Bromodomain PHD Finger Transcription Factor).
[00106] In some embodiments, the regulatory molecule is SMARCA2/4.
[00107] In some embodiments, the regulatory molecule is PBRM1.
[00108] In some embodiments, the regulatory molecule modulates the rearrangement of histones.
[00109] In some embodiments, the regulatory molecule modulates the glycosylation, phosphorylation, alkylation, or acylation of histones.
[00110] In some embodiments, the regulatory molecule is a transcription factor.
[00111] In some embodiments, the regulatory molecule is an RNA polymerase.
[00112] In some embodiments, the regulatory molecule is a moiety that regulates the activity of RNA polymerase.
[00113] In some embodiments, the recruiting moiety binds to the regulatory molecule but does not inhibit the activity of the regulatory molecule. In some embodiments, the recruiting moiety binds to the regulatory molecule and inhibits the activity of the regulatory molecule. In some embodiments, the recruiting moiety binds to the regulatory molecule and increases the activity of the regulatory molecule.
[00114] In some embodiments, the recruiting moiety binds to the active site of the regulatory molecule. In certain embodiments, the recruiting moiety binds to a regulatory site of the regulatory molecule.
[00115] The binding affinity between the regulatory protein and the second terminus can be adjusted based on the composition of the molecule or type of protein. In some embodiments, the second terminus binds the regulatory molecule with an affinity of less than about 600 nM, about 500 nM, about 400 nM, about 300 nM, about 250 nM, about 200 nM, about 150 nM, about 100 nM, or about 50nM. In some embodiments, the second terminus binds the regulatory molecule with an affinity of less than about 500 nM. In some embodiments, the second terminus binds the regulatory molecule with an affinity of less than about 400 nM. In some embodiments, the second terminus binds the regulatory molecule with an affinity of less than about 300 nM. In some embodiments, the second terminus binds the regulatory molecule with an affinity of less
than about 250 nM. In some embodiments, the second terminus binds the regulatory molecule with an affinity of less than about 200 nM. In some embodiments, the second terminus binds the regulatory molecule with an affinity of less than about 150 nM. In some embodiments, the second terminus binds the regulatory molecule with an affinity of less than about 100 nM. In some embodiments, the second terminus binds the regulatory molecule with an affinity of less than about 50 nM.
[00116] In some embodiments, the second terminus comprises a diazine or diazepine ring, wherein the diazine or diazepine ring is fused with a Ce-Cio aryl or a 5 to 10-membered heteroaryl ring comprising one or more heteroatoms selected from S, N and O. In some embodiments, the second terminus comprises an optionally substituted bicyclic or tricyclic structure.
[00117] In some embodiments, the second terminus has a triazolodiazepine structure. In some embodiments, the second terminus has a thiazolodiazepine structure.
[00118] In some embodiments, the second terminus comprises the structure of Formula (2-A), or a pharmaceutically acceptable salt thereof:
Formula (2-A), wherein:
Ring A is an optionally substituted aryl or optionally substituted 5 to 6-membered heteroaryl;
Ring B is absent or an optionally substituted 6-membered monocyclic aryl or heteroaryl;
D is C or N;
E is O or N;
YA is -NH- or -O-;
R5 is hydrogen or Ci-Ce alkyl;
R6 is selected from hydrogen, optionally substituted Ci-Ce alkyl, optionally substituted Ci-Ce haloalkyl, and optionally substituted Ci-Ce hydroxyalkyl;
R7 is selected from hydrogen, halogen, -NO2, -CN, optionally substituted aryl, optionally substituted C1-C20 alkyl, optionally substituted C1-C20 heteroalkyl, optionally substituted Ci-Ce haloalkyl, and optionally substituted Ci-Ce hydroxyalkyl; or R7 is -NR7AR7B, wherein
R7A and R7I; are each independently hydrogen, optionally substituted C1-C20 alkyl, or optionally substituted C1-C20 heteroalkyl; and xi is an integer from 1-6.
[00119] In some embodiments, D is N and E is N. In some embodiments, D is C and E is O.
[00120] In some embodiments, the second terminus comprises the structure of Formula (2-B), or a pharmaceutically acceptable salt thereof:
Formula (2-B), wherein:
Ring A is an optionally substituted aryl or optionally substituted 5 to 6-membered heteroaryl;
Ring B is absent or an optionally substituted 6-membered monocyclic aryl or heteroaryl;
YA is -NH- or -O-;
R5 is hydrogen or Ci-Ce alkyl;
R6 is selected from hydrogen, optionally substituted Ci-Ce alkyl, optionally substituted Ci-Ce haloalkyl, and optionally substituted Ci-Ce hydroxyalkyl;
R7 is selected from hydrogen, halogen, -NO2, -CN, optionally substituted aryl, optionally substituted C1-C20 alkyl, optionally substituted C1-C20 heteroalkyl, optionally substituted Ci-Ce haloalkyl, and optionally substituted Ci-Ce hydroxyalkyl; or R7 is -NR7AR7B, wherein
R7A and R7I; are each independently hydrogen, optionally substituted C1-C20 alkyl, or optionally substituted C1-C20 heteroalkyl; and xi is an integer from 1-6.
[00121] In some embodiments, Ring A is an optionally substituted aryl ring. In some embodiments, Ring A is an optionally substituted phenyl. In some embodiments, Ring A is an optionally substituted 5 membered heteroaryl. In some embodiments, Ring A is an optionally substituted oxazolyl. In some embodiments, Ring A is an optionally substituted furanyl. In some embodiments, Ring A is an optionally substituted thiophenyl.
[00122] In some embodiments, the second terminus comprising the structure of Formula (2-C), or a pharmaceutically acceptable salt thereof:
Formula (2-C), wherein:
Ring B is absent or an optionally substituted 6-membered monocyclic aryl or heteroaryl;
YA is -NH- or -O-;
R5 is hydrogen or Ci-Ce alkyl;
R6 is selected from hydrogen, optionally substituted Ci-Ce alkyl, optionally substituted Ci-Ce haloalkyl, and optionally substituted Ci-Ce hydroxyalkyl;
R7 is selected from hydrogen, halogen, -NO2, -CN, optionally substituted aryl, optionally substituted C1-C20 alkyl, optionally substituted C1-C20 heteroalkyl, optionally substituted Ci-Ce haloalkyl, and optionally substituted Ci-Cg hydroxyalkyl; or R7 is -NR7AR7B, wherein
R7A and R7I; are each independently hydrogen, optionally substituted C1-C20 alkyl, or optionally substituted C1-C20 heteroalkyl;
R8 and R9 are each independently selected from hydrogen, -C(O)OR8a, optionally substituted Ci-Ce alkyl, optionally substituted Ci-Ce haloalkyl, or optionally substituted Ci-Ce hydroxyalkyl; and
R8a js hydrogen, C1-C20 alkyl, or C1-C20 heteroalkyl; and xi is an integer from 1-6.
[00123] In some embodiments, R8 and R9 are each independently selected from optionally substituted Ci- Ce alkyl, Ci-Ce haloalkyl, or Ci-Ce hydroxyalkyl. In some embodiments, R8 and R9 are each independently selected from optionally substituted Ci-Ce alkyl. In some embodiments, R8 and R9 are each independently methyl, ethyl, or propyl. In some embodiments, R8 and R9 are each independently methyl. In some embodiments, R8 and R9 are each independently ethyl. In some embodiments, R8 and R9 are each independently propyl.
[00124] In some embodiments, the second terminus comprising the structure of Formula (2-D), or a pharmaceutically acceptable salt thereof:
Formula (2-D), wherein:
Ring B is absent or an optionally substituted 6-membered monocyclic aryl or heteroaryl;
YA is -NH- or -O-;
R5 is hydrogen or Ci-Ce alkyl;
R6 is selected from hydrogen, optionally substituted Ci-Ce alkyl, optionally substituted Ci-Ce haloalkyl, and optionally substituted Ci-Ce hydroxyalkyl;
R7 is selected from hydrogen, halogen, -NO2, -CN, optionally substituted aryl, optionally substituted C1-C20 alkyl, optionally substituted C1-C20 heteroalkyl, optionally substituted Ci-Ce haloalkyl, and optionally substituted Ci-Ce hydroxyalkyl; or R7 is -NR7AR7B, wherein
R7A and R7I; are each independently hydrogen, optionally substituted C1-C20 alkyl, or optionally substituted C1-C20 heteroalkyl;
R10 is selected from hydrogen, optionally substituted Ci-Ce alkyl, optionally substituted Ci-Ce haloalkyl, or optionally substituted Ci-Cg hydroxyalkyl; and xi is an integer from 1-6.
[00125] In some embodiments, R5 is Ci-Ce alkyl. In some embodiments, R5 is methyl or ethyl. In some embodiments, R5 is methyl. In some embodiments, R5 is ethyl. In some embodiments, R5 is hydrogen. [00126] In some embodiments, R7 is selected from hydrogen, halogen, optionally substituted Ci-Ce alkyl, Ci-Ce haloalkyl, or Ci-Ce hydroxyalkyl. In some embodiments, R7 is halogen. In some embodiments, R7 is Br, Cl, or F. In some embodiments, R7 is Cl. In some embodiments, R7 is F. In some embodiments, R7 is Br.
[00127] In some embodiments, R7 is -NR7AR7I;. wherein R7A and R7I; are each independently hydrogen or optionally substituted Ci-Ce alkyl.
[00128] In some embodiments, R10 is selected from optionally substituted Ci-Ce alkyl, optionally substituted Ci-Ce haloalkyl, or optionally substituted Ci-Ce hydroxyalkyl. In some embodiments, R10 is selected from optionally substituted Ci-Ce alkyl. In some embodiments, R10 is methyl, ethyl, or propyl. In some embodiments, R10 is methyl. In some embodiments, R10 is optionally substituted C1-6 hydroxyalkyl. In some embodiments, R10 is -OMe.
[00129] In some embodiments, R6 is selected from optionally substituted Ci-Ce alkyl, optionally substituted Ci-Ce haloalkyl, or optionally substituted Ci-Ce hydroxyalkyl. In some embodiments, R6 is an optionally substituted Ci-Ce alkyl. In some embodiments, R6 is methyl, ethyl, or propyl. In some embodiments, R6 is methyl. In some embodiments, R6 is ethyl. In some embodiments, R6 is propyl. In some embodiments, R6 is hydrogen.
[00130] In some embodiments, YA is -NH-. In some embodiments, YA is -O-.
[00131] In some embodiments, YA is NH and xi is 1.
[00132] In some embodiments, xi is an integer from 1-5, 1-4, 1-3, or 1-2. In some embodiments, xi is 1. In some embodiments, xi is 2.
[00133] In some embodiments, Ring B is an optionally substituted 6-membered monocyclic aryl or heteroaryl, each of which is optionally substituted with alkyl, amino, halogen, hydroxy, hydroxyalkyl, or PEG. In some embodiments Ring B is phenyl. In some embodiments, Ring B is 6-membered monocyclic heteroaryl. In some embodiments, Ring B is pyridine or pyrimidine. In some embodiments, Ring B is absent.
[00134] In some embodiments, the second terminus comprises the structure of Formula (2-E), or a pharmaceutically acceptable salt thereof:
Formula (2-E).
[00135] In some embodiments, the second terminus comprises the structure of Formula (2-F), or a pharmaceutically acceptable salt thereof:
Formula (2-F).
[00136] In some embodiments, the second terminus comprises the structure of Formula (2-G), or a pharmaceutically acceptable salt thereof:
[00137] In some embodiments, the second terminus comprises the structure of Formula (3-A), or a pharmaceutically acceptable salt thereof:
Formula (3-A), wherein:
YB is -CH2NH-, -CH2O-, -NH-, or -O-;
R11A and R11B are each independently hydrogen, or optionally substituted Ci-Ce alkyl;
R12 is hydrogen, halogen, -OH, optionally substituted Ci-Ce alkyl, optionally substituted Ci-Ce haloalkyl, or optionally substituted Ci-Ce hydroxyalkyl;
R14 and R15 are each independently hydrogen, halogen, -CN, -NO2, optionally substituted Ci-Ce alkyl, optionally substituted Ci-Ce haloalkyl, or optionally substituted Ci-Ce hydroxyalkyl; or R14 is -NRARB;
R16 is optionally substituted Ci-Ce alkyl, optionally substituted Ci-Ce heteroalkyl, optionally substituted C2- C, alkenyl, optionally substituted C2-C6 alkynyl, optionally substituted Ci-Ce hydroxyalkyl, - S(O)(=NH)RA, -SO2RA, or -NHSO2RA;
RYA is hydrogen, optionally substituted Ci-Cg alkyl, optionally substituted Ci-Cg haloalkyl, or optionally substituted 5-6-membered monocyclic aryl or heteroaryl; each RA and RB is independently hydrogen, optionally substituted Ci-Ce alkyl, optionally substituted Ci-Ce heteroalkyl, optionally substituted Ci-Cg haloalkyl, optionally substituted Ci-Cg hydroxyalkyl, optionally substituted C3-C6 cycloalkyl, optionally substituted 4 to 6-membered heterocycloalkyl, or optionally substituted 5 to 6-membered heteroaryl; and yi is 1-3; and wherein the attachment to the linker is at either R14 or at RYA.
[00138] In some embodiments, the second terminus comprises the structure of Formula (3-B), or a pharmaceutically acceptable salt thereof:
Formula (3-B), wherein:
Ring C is absent, optionally substituted 5 to 6-membered monocyclic aryl or heteroaryl or 4 to 8-membered heterocycle;
YB is -NH-, -CH2NH-, -CH2O-, or -O-;
R11A and R11B are each independently hydrogen, or optionally substituted Ci-Ce alkyl;
R12 is hydrogen, optionally substituted Ci-Ce alkyl, C(O)RA, or C(O)NRARB; wherein each RA and RB is independently hydrogen, optionally substituted Ci-Ce alkyl, or optionally substituted Ci-Ce heteroalkyl;
R13 is hydrogen, substituted aryl, substituted heteroaryl, or substituted oxydibenzene; and y2 is an integer from 0-2.
[00139] In some embodiments, the second terminus comprises the structure of Formula (3-B1), or a pharmaceutically acceptable salt thereof:
Formula (3-B1), wherein:
R11A and R11B are each independently hydrogen or optionally substituted Ci-Ce alkyl;
R13 is hydrogen, substituted aryl, substituted heteroaryl, or substituted oxydibenzene; and y2 is an integer from 0-2.
[00140] In some embodiments, y2 is 0. In some embodiments, y2 is 1. In some embodiments, y2 is 2.
[00141] In some embodiments, R13 is substituted aryl or substituted heteroaryl. In some embodiments, R13 is hydrogen.
[00142] In some embodiments, R13 is substituted oxydibenzene.
R14 and R15 are each independently hydrogen, halogen, -CN, -NO2, optionally substituted Ci-Ce alkyl, optionally substituted Ci-Ce haloalkyl, or optionally substituted Ci-Ce hydroxyalkyl; or R14 is -NRARB;
R16 is optionally substituted Ci-Ce alkyl, optionally substituted Ci-Ce heteroalkyl, optionally substituted C2- C, alkenyl, optionally substituted C2-C6 alkynyl, optionally substituted Ci-Ce hydroxyalkyl, - S(O)(=NH)RA, -SO2RA, or -NHSO2RA; each RA and RB is independently hydrogen, optionally substituted Ci-Ce alkyl, optionally substituted Ci-Ce heteroalkyl, optionally substituted Ci-Ce haloalkyl, optionally substituted Ci-Ce hydroxyalkyl, optionally substituted Cs-Ce cycloalkyl, optionally substituted 4 to 6-membered heterocycloalkyl, or optionally substituted 5 to 6-membered heteroaryl; and yi is 1-3.
[00144] In some embodiments, the second terminus comprises the structure of Formula (3-C), or a pharmaceutically acceptable salt thereof:
wherein:
Ring C is absent, optionally substituted 5 to 6-membered monocyclic aryl or heteroaryl or 4 to 8-membered heterocycle;
YB is -CH2NH-, -CH2O-, -NH-, or -O-;
R11A and R11B are each independently hydrogen or optionally substituted Ci-Ce alkyl;
R12 is hydrogen, halogen, -OH, optionally substituted Ci-Ce alkyl, optionally substituted Ci-Ce haloalkyl, or optionally substituted Ci-Ce hydroxyalkyl;
R14 and R15 are each independently hydrogen, halogen, -CN, -NO2, optionally substituted Ci-Ce alkyl, optionally substituted Ci-Ce haloalkyl, or optionally substituted Ci-Ce hydroxyalkyl; or R14 is -NRARB;
R16 is optionally substituted Ci-Ce alkyl, optionally substituted Ci-Ce heteroalkyl, optionally substituted C - C„ alkenyl, optionally substituted C2-C6 alkynyl, optionally substituted Ci-Ce hydroxyalkyl, - S(O)(=NH)RA, -SO2RA, or -NHSO2RA; each RA and RB is independently hydrogen, optionally substituted Ci-Ce alkyl, optionally substituted Ci-Ce heteroalkyl, optionally substituted Ci-Ce haloalkyl, optionally substituted Ci-Ce hydroxyalkyl, optionally substituted Cs-Ce cycloalkyl, optionally substituted 4 to 6-membered heterocycloalkyl, or optionally substituted 5 to 6-membered heteroaryl; and yi is an integer from 1-3.
[00145] In some embodiments, YB is -NH-. In some embodiments, YB is -CH2NH-. In some embodiments, YB is -CH2O-. In some embodiments, YB is -O-.
[00146] In some embodiments, Ring C is an optionally substituted 5 or 6-membered monocyclic aryl or heteroaryl, each of which is optionally substituted with alkyl, amino, halogen, hydroxy, hydroxyalkyl, or PEG.
[00147] . In some embodiments, Ring C is phenyl. In some embodiments, Ring C is a 6-membered heteroaryl. In some embodiments, Ring C is pyridine, pyrazine, or triazine. In some embodiments, Ring C is pyridine. In some embodiments, Ring C is pyrazine. In some embodiments, Ring C is triazine. In some embodiments, Ring C is a 5 -membered heteroaryl. In some embodiments, Ring C is a pyrazole. In some embodiments, Ring C is a triazole, pyrrole, imidazole, oxazole, oxadiazole, thiazole, or thiadiazole. In some embodiments, Ring C is a triazole. In some embodiments, Ring C is an imidazole or pyrrole. In some embodiments, an oxazole or oxadiazole. In some embodiments, Ring C is a thiazole or thiadiazole.
[00148] In some embodiments, Ring C is absent.
[00149] In some embodiments, the second terminus comprises the structure of Formula (3-D), or a pharmaceutically acceptable salt thereof:
Formula (3-D), wherein:
R11A and R11B are each independently hydrogen or optionally substituted Ci-Ce alkyl;
R12 is hydrogen or an optionally substituted Ci-Ce alkyl; each R15 is independently hydrogen, halogen, -CN, -NO2, optionally substituted Ci-Cg alkyl, optionally substituted Ci-Ce haloalkyl, or optionally substituted Ci-Ce hydroxyalkyl;
R16 is optionally substituted Ci-Ce alkyl, optionally substituted Ci-Ce heteroalkyl, optionally substituted C2- C, alkenyl, optionally substituted C2-C6 alkynyl, optionally substituted Ci-Ce hydroxyalkyl, - S(O)(=NH)RA, -SO2RA, or -NHSO2RA;
RA is hydrogen, optionally substituted Ci-Ce alkyl, optionally substituted Ci-Ce heteroalkyl, optionally substituted Ci-Ce haloalkyl, optionally substituted Ci-Ce hydroxyalkyl, optionally substituted C3-C,, cycloalkyl, optionally substituted 4 to 6-membered heterocycloalkyl, or optionally substituted 5 to 6- membered heteroaryl; and yi is an integer from 1-3.
[00150] In some embodiments, R11A and R11B are each independently optionally substituted Ci-Ce alkyl. In some embodiments, R11A and R11B are each independently methyl, ethyl, propyl, or tert-butyl. In some embodiments, R11A and R11B are each independently methyl. In some embodiments, R11A and R11B are each independently hydrogen.
[00151] In some embodiments, R11A is Ci-Ce alkyl, optionally substituted with haloalkyl or phosphorous hydroxide. In some embodiments, R11A is Ci-Ce alkyl substituted with -OP(O)(OH)2. In some embodiments, R11A is unsubstituted Ci-Ce alkyl. In some embodiments, R11A is methyl, ethyl, or tert-butyl. In some embodiments, R11A is methyl. In some embodiments, R11A is hydrogen.
[00152] In some embodiments, R12 is optionally substituted Ci-Ce alkyl. In some embodiments, R12 is hydrogen.
[00153] In some embodiments, R12 is C(O)RA or C(O)NRARB. In some embodiments, R12 is C(O)NRARB, wherein RA and RB are each independently hydrogen or optionally substituted Ci-Ce alkyl.
[00154] In some embodiments, R14 and R15 are each independently hydrogen, -CN, or -NO2. In some embodiments, R14 and R15 are each independently halogen or optionally substituted Ci-Ce alkyl. In some embodiments, R14 and R15 are each independently Br, Cl, F, methyl, or ethyl. In some embodiments, R14 and R15 are each independently F or methyl.
[00155] In some embodiments, R16 is optionally substituted Ci-Ce alkyl, optionally substituted Ci-Ce heteroalkyl, optionally substituted C2-C6 alkenyl, optionally substituted C2-C6 alkynyl, or optionally substituted Ci-Ce hydroxyalkyl, each of which is optionally substituted with amido, alkyl, alkynyl, azido, amino, halogen, haloalkyl, hydroxy, nitro, oxo (=0), phosphorous hydroxide, or PEG.
[00156] In some embodiments, R16 is optionally substituted Ci-Ce alkyl, optionally substituted Ci-Ce heteroalkyl, or optionally substituted Ci-Ce hydroxyalkyl. In some embodiments, R16 is Ci-Ce alkyl or Ci-Ce heteroalkyl, each or which optionally substituted with -CN, -NH2, -N3, -OH, CF3, or -OP(O)(OH)2.
[00157] In some embodiments, R16 is -SO2RA, wherein RA is Ci-Ce alkyl. In some embodiments, R16 is - S02Et. In some embodiments, R16 is -SO2Me.
[00158] In some embodiments, R16 is -NHSO2RA, wherein RA is Ci-Ce alkyl. In some embodiments, R16 is -NHSO2Et. In some embodiments, R16 is -NHSO2Me.
[00159] In some embodiments, yi is 1. In some embodiments, yi is 2. In some embodiments, yi is 3. [00160] In some embodiments, the second terminus comprises the structure of Formula (3-E), or a pharmaceutically acceptable salt thereof:
Formula (3-E).
[00161] In some embodiments, the second terminus comprising the structure of Formula (3-F), or a pharmaceutically acceptable salt thereof:
Formula (3-F).
[00162] In some embodiments, the second terminus comprising the structure of Formula (3-G) or Formula (3-H), or a pharmaceutically acceptable salt thereof:
Formula (3-H).
[00163] In some embodiments, the second terminus comprising the structure of Formula (3-1), or a pharmaceutically acceptable salt thereof:
Formula (3-1).
[00164] In some embodiments, the second terminus comprises the structure of Formula (4-A), or a pharmaceutically acceptable salt thereof:
Formula (4-A), wherein:
Ring D is absent, phenyl, or 5 to 6-membered heteroaryl;
X9 and X10 are each independently C or N, wherein one of X9 or X10 is N;
L2 is absent, optionally substituted alkylene, -O-, or -NRD-, wherein RD is hydrogen or optionally substituted C1-C3 alkyl;
R18 is an optionally substituted 5 to 6-membered heteroaryl;
R19 is an optionally substituted C3-G cycloalkyl or optionally substituted 4 to 7-membered heteroaryl; each R20 is independently hydrogen, halogen, -CN, -NO2, optionally substituted Ci-Ce alkyl, optionally substituted Ci-Ce haloalkyl, or optionally substituted Ci-Ce hydroxyalkyl;
X3 is an integer from 1-3; and y4 is an integer from 1-4; wherein the attachment to the linker is at either R19 or at one of R20.
[00165] In some embodiments, attachment to the linker is at R19.
[00166] In some embodiments, attachment to the linker is at one of R20.
[00167] In some embodiments, the second terminus comprises the structure of Formula (4-B), or a pharmaceutically acceptable salt thereof:
Formula (4-B),
wherein:
Ring D is absent, optionally substituted phenyl, or optionally substituted 5 to 6-membered heteroaryl;
X9 and X10 are each independently C or N, wherein one of X9 or X10 is N;
L2 is absent, optionally substituted alkylene, -O-, or -NRD-, wherein RD is hydrogen or optionally substituted C1-C3 alkyl;
R18 is an optionally substituted 5 to 6-membered heteroaryl;
R19 is an optionally substituted C-G cycloalkyl or optionally substituted 4 to 7-membered heteroaryl; and x3 is an integer from 1-3.
[00168] In some embodiments, X9 is N; and X10 is C. In some embodiments, X9 is C; and X10 is N. [00169] In some embodiments, the second terminus comprises the structure of Formula (4-C), or a pharmaceutically acceptable salt thereof:
Formula (4-C).
[00170] In some embodiments, Ring D is an optionally substituted monocyclic 6-membered aryl or 5 to 6- membered heteroaryl. In some embodiments, Ring D is an optionally substituted monocyclic 6-membered aryl. In some embodiments, Ring D is an optionally substituted phenyl.
[00171] In some embodiments, R19 is an optionally substituted C3-C8 cycloalkyl. In some embodiments, R19 is optionally substituted 4 to 7-membered heteroaryl.
[00172] In some embodiments, the second terminus comprises the structure of Formula (4-D), or a pharmaceutically acceptable salt thereof:
Formula (4-D), wherein:
L2 is an optionally substituted alkylene, -O-, or -NRD-, wherein RD is hydrogen or optionally substituted Ci- C3 alkyl;
R18 is an optionally substituted 5 to 6-membered heteroaryl;
R20 is hydrogen, halogen, -CN, -NO2, optionally substituted Ci-Ce alkyl, optionally substituted Ci-Ce haloalkyl, or optionally substituted Ci-Ce hydroxyalkyl; x3 is an integer from 1-3; and
y4 is an integer from 1-4.
[00173] In some embodiments, L2 is an optionally substituted alkylene. In some embodiments, L2 is C2-C4 alkylene, optionally substituted with one or more C1-C3 alkyl. In some embodiments, L2 is absent.
[00174] In some embodiments, L2 is -NRD-. In some embodiments, L2 is -NH-.
[00175] In some embodiments, R18 is an optionally substituted 5 -membered heteroaryl. In some embodiments, R18 is optionally substituted oxazole, oxadiazole, thiazole, thiadiazole, pyrrole, or pyrazole. In some embodiments, R18 is optionally substituted oxazole.
[00176] In some embodiments, R20 is halogen, -CN, -NO2, optionally substituted Ci-Ce alkyl, optionally substituted Ci-Ce haloalkyl, or optionally substituted Ci-Ce hydroxyalkyl.
[00177] In some embodiments, x3 is 1. In some embodiments, x3 is 2. In some embodiments, x3 is 3.
[00178] In some embodiments, y4 is 1 or 2. In some embodiments, y4 is 1. In some embodiments, y4 is 2.
In some embodiments, y4 is 3. In some embodiments, y4 is 4.
[00179] In some embodiments, the second terminus comprises the structure of Formula (4-E) or Formula (4-F), or a pharmaceutically acceptable salt thereof:
Formula (4E) or Formula (4F).
[00180] In some embodiments, the second terminus comprises the structure of Formula (4-G), or a pharmaceutically acceptable salt thereof:
[00181] In some embodiments, the second terminus comprises the structure of Formula (5-A), or a pharmaceutically acceptable salt thereof:
wherein:
Ring E is absent or an optionally substituted phenyl or optionally substituted 5 to 6-membered heteroaryl;
X11 is CH orN;
L3 is -NRE- or -CRERE-, wherein each RE is independently hydrogen, or optionally substituted C1-C3 alkyl;
R21 is Ci-Ce alkyl or Cs-Ce cycloalkyl; and
R22 is halogen, CN, NO2, optionally substituted Ci-Ce alkyl, optionally substituted Ci-Ce haloalkyl, or optionally substituted Ci-Ce hydroxyalkyl.
[00182] In some embodiments, Ring E is absent. In some embodiments, Ring E is an optionally substituted phenyl. In some embodiments, Ring E is an optionally substituted 5 to 6-membered heteroaryl. In some embodiments, Ring E is a 5 -membered heteroaryl. In some embodiments, Ring E is a 6-membered heteroaryl.
[00183] In some embodiments, X11 is CH and L3 is -NRE-. In some embodiments, X11 is N and L3 is - CRERE-.
[00184] In some embodiments, R21 is Ci-Ce alkyl. In some embodiments, R21 is methyl.
[00185] In some embodiments, R22 is halogen, optionally substituted Ci-Ce alkyl, optionally substituted Ci-Ce haloalkyl, or optionally substituted Ci-Ce hydroxyalkyl. In some embodiments, R22 is CN, F, Cl, Br, or methyl.
[00186] In some embodiments, the second terminus comprises the structure of Formula (5-B) or Formula (5-C), or a pharmaceutically acceptable salt thereof:
[00187] In some embodiments, the second terminus comprises the structure of Formula (6-A), or a pharmaceutically acceptable salt thereof:
Formula (6-A), wherein:
Ring G is absent or an optionally substituted C3-C, cycloalkyl or optionally substituted 4 to 6-membered heterocycloalkyl;
L6 is -O-(optionally substituted alkylene);
R28 is an optionally substituted 5 to 6-membered heteroaryl;
R29 is optionally substituted Ci-Ce alkyl-(Ce-Cio aryl) or optionally substituted Ci-Ce alkyl-(6 to 10- membered heteroaryl); and
R30 is optionally substituted Ci-Ce alkyl, optionally substituted Ci-Ce haloalkyl, optionally substituted Ci-Ce hydroxyalkyl, optionally substituted C3-C3 cycloalkyl, or optionally substituted 4 to 6-membered heterocycloalkyl.
[00188] In some embodiments, the second terminus comprises the structure of Formula (7-A), or a pharmaceutically acceptable salt thereof:
Formula (7-A), wherein:
A3 is -O-, -NH-, or -CH2-;
Z2 is CH or N;
W is O or S; each R31 is independently hydrogen, halogen, -OH, -CN, -NO2, -NH2, optionally substituted C1-C10 alkyl, optionally substituted C1-C10 haloalkyl, optionally substituted C1-C10 hydroxyalkyl, optionally substituted C2-Cio alkenyl, optionally substituted C2-Cio alkynyl, optionally substituted C3-G cycloalkyl, or optionally substituted 3 to 8-membered heterocycloalkyl; or two R31 together with the atoms to which they are attached form an optionally substituted Cs-Cs cycloalkyl or optionally substituted 5 to 8-membered heterocycloalkyl;
R32 is hydrogen or optionally substituted C1-C10 alkyl;
R32a is hydrogen or optionally substituted C1-C10 alkyl;
R33 is hydrogen, halogen, -OH, -CN, -NO2, -NH2, optionally substituted C1-C10 alkyl, optionally substituted C1-C10 haloalkyl, or optionally substituted C1-C10 hydroxyalkyl; and
qe is 0-4; wherein Formula (7-A) is atached to the linker is at R32a or at one of R31.
[00189] In some embodiments, Z2 is CH. In some embodiments, Z2 is N.
[00190] In some embodiments, the second terminus comprises the structure of Formula (7-B), or a pharmaceutically acceptable salt thereof:
Formula (7-B), wherein:
Ring F is an optionally substituted 5 to 6-membered heteroaryl;
A3 is -O-, -NH-, or -CH2-;
Z3 is CH or N;
W is O or S; each R31 is hydrogen, halogen, -OH, -CN, -NO2, -NH2, optionally substituted C1-C10 alkyl, optionally substituted Ci-Cio haloalkyl, optionally substituted C1-C10 hydroxyalkyl, optionally substituted C2-Cio alkenyl, optionally substituted C2-Cio alkynyl, optionally substituted C-G-cycloalk l. or optionally substituted 3- to 8-membered heterocycloalkyl; or two R31 together with the atoms to which they are attached form an optionally substituted - cycloalkyl or optionally substituted 5 to 8-membered heterocycloalkyl;
R32 is hydrogen or optionally substituted C1-C10 alkyl; and q6 is 1-4.
[00191] In some embodiments, A3 is -O-. In some embodiments, A3 is -NH-. In some embodiments, A3 is - CH2-.
[00192] In some embodiments, Z3 is CH. In some embodiments, Z3 is N.
[00193] In some embodiments, W is O. In some embodiments, W is S.
[00194] In some embodiments, Ring F is an optionally substituted 5 -membered heteroaryl. In some embodiments, Ring F is an optionally substituted 6-membered heteroaryl.
[00195] In some embodiments, each R31 is independently an optionally substituted C1-C10 alkyl, optionally substituted C1-C10 haloalkyl, or optionally substituted C1-C10 hydroxyalkyl. In some embodiments, each R31 is independently an optionally substituted G-G -cycloalkyl or optionally substituted 3 to 8-membered heterocycloalkyl. In some embodiments, each R31 is independently hydrogen, halogen, -OH, -CN, -NO2, or - NH2. In some embodiments, each R31 is hydrogen.
[00196] In some embodiments, R32 is an optionally substituted C1-C10 alkyl. In some embodiments, R32 is methyl. In some embodiments, R32 is hydrogen.
[00197] In some embodiments, R32a is an optionally substituted Ci-Cio alkyl. In some embodiments, R32a is hydrogen.
[00198] In some embodiments, R33 is hydrogen, halogen, -OH, -CN, -NO2, or -NH2. In some embodiments, R33 is an optionally substituted C1-C10 alkyl, optionally substituted C1-C10 haloalkyl, or optionally substituted C1-C10 hydroxyalkyl.
[00199] In some embodiments, Formula (7-A) is attached to the linker is at R32a. In some embodiments, Formula (7-A) is attached to the linker at one of R31.
[00200] In some embodiments, the second terminus comprises the structure of Formula (7-C), or a pharmaceutically acceptable salt thereof:
Formula (7-C).
[00201] In some embodiments, the second terminus comprise the structure of Formula (7-D), or a pharmaceutically acceptable salt thereof:
[00202] In some embodiments, the second terminus comprises the structure of Formula (7-E), or a pharmaceutically acceptable salt thereof:
Formula (7-E).
[00203] In some embodiments, the second terminus comprises the structure of Formula (8-A), or a pharmaceutically acceptable salt thereof:
Formula (8-A), wherein:
Ring H is an optionally substituted phenyl or optionally substituted 6-membered heteroaryl; or Ring
ZA is absent or an optionally substituted phenyl formamide;
X12 is CH or N;
R34 is an optionally substituted phenyl or optionally substituted 6-membered heteroaryl;
R34A is hydrogen, halogen, or optionally substituted C1-C3 alkyl; and
R35 is independently halogen, optionally substituted C1-C10 alkyl, or optionally substituted 5 to 6-membered heteroaryl; wherein the attachment to the linker is at R35, ZA, or Ring H.
[00204] In some embodiments, Ring H is an optionally substituted phenyl. In some embodiments, Ring H is an optionally substituted 6-membered heteroaryl.
[00206] In some embodiments, ZA is absent. In some embodiments, ZA is an optionally substituted phenyl formamide. In some embodiments, ZA is -C(O)NH-phenyl.
[00207] In some embodiments, X12 is CH. In some embodiments, X12 is N.
[00208] In some embodiments, R34 is an optionally substituted phenyl. In some embodiments, R34 is an optionally substituted 6-membered heteroaryl.
[00209] In some embodiments, R34A is hydrogen or halogen. In some embodiments, R34A is an optionally substituted C1-C3 alkyl. In some embodiments, R34A is methyl.
[00210] In some embodiments, Formula (8-A) is attachment to the linker is at R35. In some embodiments, Formula (8-A) is attached to the linker at ZB. In some embodiments, Formula (8-A) is attached to the linker at Ring H.
[00211] In some embodiments, the second terminus comprises the structure of Formula (8-B) or Formula (8-C), or a pharmaceutically acceptable salt thereof:
Formula (8-B) or Formula (8-C).
[00212] In some embodiments, the second terminus comprises the structure of Formula (8-D), or a pharmaceutically acceptable salt thereof:
[00213] In some embodiments, the second terminus comprises the structure of Formula (9-A), or a pharmaceutically acceptable salt thereof:
Formula (9-A).
[00214] In some embodiments, the second terminus comprises the structure of Formula (10-A) or Formula (10-B), or a pharmaceutically acceptable salt thereof:
[00215] In some embodiments, the second terminus comprises the structure of Formula (11-A), or a pharmaceutically acceptable salt thereof:
Formula (11-A).
[00216] In some embodiments, the second terminus comprises the structure of Formula (12-A), or a pharmaceutically acceptable salt thereof:
Formula (12-A), wherein:
A4 is -CR40R40- or -NR40-; wherein each R40 is independently hydrogen or optionally substituted C1-C10 alkyl;
R36 is an optionally substituted 5 to 6-membered heteroaryl; each R37 is independently hydrogen, halogen, Ci-Ce alkyl, or Ci-Ce haloalkyl;
R38 is optionally substituted C1-C10 alkyl, optionally substituted Cs-Cs cycloalkyl, or optionally substituted 3 to 8-membered heterocycloalkyl;
R39 is hydrogen, halogen, -OH, -CN, -NO2, -NH2, oxo (=0), =S, C1-C10 haloalkyl, or C1-C10 hydroxyalkyl; pn is 1-4; and qi and q2are each independently 0-2; wherein the linker is attached to Formula (12-A) at either R38 or R40.
[00217] In some embodiments, R36 is an optionally substituted 5 -membered heteroaryl. In some embodiments, R36 is optionally substituted oxazole, oxadiazole, thiazole, thiadiazole, pyrrole, or pyrazole. In some embodiments, R36 is optionally substituted oxazole.
[00218] In some embodiments, each R37 is independently halogen, Ci-Ce alkyl, or Ci-Ce haloalkyl. In some embodiments, each R37 is independently halogen.
[00219] In some embodiments, R38 is an optionally substituted C1-C10 alkyl. In some embodiments, R38 is an optionally substituted Cs-Cs cycloalkyl or optionally substituted 3 to 8-membered heterocycloalkyl. In some embodiments, R38 is a 3 to 8-membered heterocycloalkyl.
[00220] In some embodiments, R39 is hydrogen, halogen, -OH, -CN, -NO2, -NH2, C1-C10 haloalkyl, or Ci- C10 hydroxyalkyl. In some embodiments, R39 is oxo or =S. In some embodiments, R39 is oxo. In some embodiments, R39 is =S.
[00221] In some embodiments, A4 is -NR40. In some embodiments, A4 is -NH. In some embodiments, A4 is -NCH3. In some embodiments, A4 is -CR40R40. In some embodiments, A4 is -CH2-.
[00222] In some embodiments, each R40 is independently optionally substituted C1-C10 alkyl. In some embodiments, each R40 is independently hydrogen.
[00223] In some embodiments, pn is 3 or 4. In some embodiments, pn is 2. In some embodiments, pn is 1. [00224] In some embodiments, qi is 1 and q2 is 1. In some embodiments, qi is 2 and q2 is 0.
[00225] In some embodiments, the linker is attached to Formula (12-A) through R38. In some embodiments, the linker is attached to Formula (12-A) through R40.
[00226] In some embodiments, the second terminus comprises Formula (12-B) or (12-C), or a
[00227] In some embodiments, the second terminus comprises Formula (12-D) or (12-E), or a pharmaceutically acceptable salt thereof:
[00228] In some embodiments, the second terminus comprises the structure of Formula (13-A), or a pharmaceutically acceptable salt thereof:
Formula (13-A), wherein:
Ring J is absent or optionally substituted 5 to 6-membered heteroaryl;
R41 is optionally substituted Ci-Ce alkyl, optionally substituted C’s-G cycloalkyl, -C(O)R41a, -C(O)-, or - C(O)NR41aR41b, wherein
R41a and R41b are each independently optionally substituted Ci-Cio alkyl or optionally substituted Cs-Cs cycloalkyl;
R42 is an optionally substituted Ci-Cio alkyl, optionally substituted Ci-Cio haloalkyl, optionally substituted C3-C8 cycloalkyl, or optionally substituted 3 to 8 membered heterocycloalkyl;
R43 is hydrogen or optionally substituted C1-C10 alkyl; each R44 is independently hydrogen, halogen, -OH, -CN, -NO2, -NH2, optionally substituted C1-C10 alkyl, optionally substituted Ci-Ciohaloalkyl, C1-C10 hydroxyalkyl, optionally substituted C2-C10 alkenyl, optionally substituted C2-Cio alkynyl, optionally substituted G-G-cycloalkyl. or optionally substituted 3 to 8-membered heterocycloalkyl; or
R43 and one of R44 together with the atoms to which they are attached form an optionally substituted 5 to 8- membered heterocycloalkyl;
P12 is 1-4; and qs is 0 or 1 ; wherein Formula (13-A) is connected to the linker at Ring J, R41, or R42.
[00229] In some embodiments, R41 is optionally substituted Ci-Ce alkyl or optionally substituted G-G cycloalkyl. In some embodiments, R41 is -C(O)R41a. In some embodiments, R41 is -C(O)CH3 or - C(O)CH2CH3. In some embodiments, R41 is -C(O)-NR41aR41b.
[00230] In some embodiments, R41a is optionally substituted C1-C10 alkyl. In some embodiments, R41a is optionally substituted G-G cycloalkyl.
[00231] In some embodiments, R41b is optionally substituted C1-C10 alkyl. In some embodiments, R41b is optionally substituted G-G cycloalkyl.
[00232] In some embodiments, R42 is optionally substituted G-Go alkyl or optionally substituted G-Go haloalkyl. In some embodiments, R42 is optionally substituted G-G cycloalkyl or optionally substituted 3 to 8 membered heterocycloalkyl. In some embodiments, R42 is optionally substituted 3- to 8-membered heterocycloalkyl ring.
[00233] In some embodiments, R43 is optionally substituted G-Go alkyl. In some embodiments, R43 is hydrogen.
[00234] In some embodiments, each R44 is independently halogen, -OH, -CN, -NO2, -NH2, optionally substituted - o alkyl, optionally substituted G-Go haloalkyl, G-Go hydroxyalkyl, optionally substituted G-Go alkenyl, optionally substituted G-Go alkynyl, optionally substituted G-G-cycloalkyl. or optionally substituted 3 to 8-membered heterocycle. In some embodiments, each R44 is independently halogen or G- C10 haloalkyl.
[00235] In some embodiments, R43 and one of R44 together with the atoms to which they are attached form an optionally substituted 5 to 8-membered heterocycloalkyl. In some embodiments, R43 and one of R44 together with the atoms to which they are attached form a 5, 6, 7, or 8-membered heterocycloalkyl.
[00236] In some embodiments, P12 is 3 or 4. In some embodiments, pn is 2. In some embodiments, pn is 1. [00237] In some embodiments, q, is 1. In some embodiments, q3 is 0.
[00238] In some embodiments, Ring J is an optionally substituted 5 -membered heteroaryl. In some embodiments, Ring J is absent.
[00239] In some embodiments, Formula (13-A) is connected to the linker at Ring J. In some embodiments, Formula (13-A) is connected to the linker at R41. In some embodiments, Formula (13-A) is connected to the linker at R42.
[00240] In some embodiments, the second terminus comprises the structure of Formula (13-B), or a pharmaceutically acceptable salt thereof:
Formula (13-B).
[00241] In some embodiments, the second terminus comprises the structure of Formula (13-C1) or Formula (13-C2), or a pharmaceutically acceptable salt thereof:
.
[00242] In some embodiments, the second terminus comprises the structure of Formula (13-D1) or Formula (13-D2), or a pharmaceutically acceptable salt thereof:
Formula (13-D1) or
Formula (13-D2).
[00243] In some embodiments, the second terminus comprises the structure of Formula ( 13-E), or a pharmaceutically acceptable salt thereof:
Formula (13-E).
[00244] In some embodiments, the second terminus comprises the structure of Formula (14-A), or a pharmaceutically acceptable salt thereof:
Formula (14-A), wherein:
Ring K is a 5 to 6-membered heterocycloalkyl;
A5 is absent, CH2, -NH-, or -O-;
L4 is alkylene or heteroalkylene; each R45 is independently halogen, -OH, -CN, -NO2, -NH2, optionally substituted C1-C10 alkyl, optionally substituted Ci-Ciohaloalkyl, optionally substituted C1-C10 heteroalkyl, C1-C10 hydroxyalkyl, optionally substituted C2-C10 alkenyl, optionally substituted C2-Cioalkynyl, optionally substituted Cs-Cs-cycloalkyl, or optionally substituted 3 to 8-membered heterocycloalkyl; each R46 is independently hydrogen, halogen, -OH, -CN, -NO2, -NH2, optionally substituted C1-C10 alkyl, optionally substituted Ci-Ciohaloalkyl, optionally substituted C1-C10 hydroxyalkyl, optionally substituted C2-C10 alkenyl, optionally substituted C2-Cioalkynyl, optionally substituted Cs-Cs cycloalkyl, or optionally substituted 3 to 8-membered heterocycloalkyl;
R47 is optionally substituted Ci-Cw alkyl, -C(O)R47a, or -C(O)-NR47aR47b, wherein
R47a and R47b are each independently optionally substituted C1-C10 alkyl or optionally substituted C’s-G cycloalkyl; q4 is 2-3; and qs is 0-2; wherein the Formula (14-A) is connected to the linker through Ring K or through one of R45.
[00245] In some embodiments, A5 is absent. In some embodiments, A5 is -NH- or -O-. In some embodiments, A5 is -NH-. In some embodiments, A5 is -O-.
[00246] In some embodiments, L4 is alkylene. In some embodiments, In some embodiments, L4 is C1-C5 alkylene.
[00247] In some embodiments, L4 is heteroalkylene. In some embodiments, L4 is C1-C4 heteroalkylene-. In some embodiments, L4 is -O-CH2- or -O-CH2CH2-.
[00248] In some embodiments, each R45 is independently halogen, -OH, -CN, -NO2, -NH2, optionally substituted C1-C10 alkyl, optionally substituted Ci-Ciohaloalkyl, C1-C10 hydroxyalkyl, optionally substituted C2-C10 alkenyl, or optionally substituted C2-C10 alkynyl. In some embodiments, each R45 is independently optionally substituted C1-C10 alkyl or optionally substituted C1-C10 hydroxyalkyl. In some embodiments, each R45 is independently C1-C10 hydroxyalkyl. In some embodiments, each R45 is independently -OCH3 or - OCH2CH3.
[00249] In some embodiments, each R46 is independently hydrogen, halogen, -OH, -CN, -NO2, -NH2, optionally substituted C1-C10 alkyl, optionally substituted Ci-Ciohaloalkyl, or optionally substituted C1-C10 hydroxyalkyl. In some embodiments, each R46 is independently hydrogen.
[00250] In some embodiments, R47 is optionally substituted C1-C10 alkyl. In some embodiments, R47is - C(O)R47a. In some embodiments, R47 is -C(O)CH3 or -C(O)CH2CH3. In some embodiments, -C(O)- NR47aR47b.
[00251] In some embodiments, R47a is optionally substituted C1-C10 alkyl. In some embodiments, R47a is optionally substituted C3-C8 cycloalkyl.
[00252] In some embodiments, R47b is optionally substituted C1-C10 alkyl. In some embodiments, R47b is an optionally substituted C3-C8 cycloalkyl.
[00253] In some embodiments, Ring K is a 6-membered heterocycloalkyl.
[00254] In some embodiments, q4 is 3. In some embodiments, q4 is 2.
[00255] In some embodiments, qs is 2. In some embodiments, qs is 1. In some embodiments, qs is 0.
[00256] In some embodiments, Formula (14-A) is connected to the linker through Ring K. In some embodiments, Formula (14-A) is connected to the linker through one of R45.
[00257] In some embodiments, the second terminus comprises the structure of Formula (14-B) or Formula (14-C), or a pharmaceutically acceptable salt thereof:
[00258] In some embodiments, the second terminus comprises the structure of Formula (15-A), or a pharmaceutically acceptable salt thereof:
Formula (15-A), wherein:
Ring L is an aryl or heteroaryl; each R48 is hydrogen, halogen, -OH, -CN, -NO2, -NH2, C1-C10 alkyl, Ci-Ciohaloalkyl, or C1-C10 hydroxyalkyl;
R49 and R50 are each independently hydrogen, optionally substituted C1-C10 alkyl, optionally substituted Ci- Ciohaloalkyl, optionally substituted C2-C10 alkenyl, or optionally substituted C2-Cio alkynyl;
R51 is hydrogen, halogen, -OH, -CN, -NO2, -NH2, optionally substituted C1-C10 alkyl, optionally substituted Ci-Ciohaloalkyl, optionally substituted C1-C10 hydroxyalkyl, optionally substituted C2-C10 alkenyl, or optionally substituted C2-Cio alkynyl;
R52 is hydrogen, halogen, -OH, -CN, -NO2, -NH2, optionally substituted C1-C10 alkyl, optionally substituted Ci-Ciohaloalkyl, or optionally substituted C1-C10 hydroxyalkyl; and p? is 1-4.
[00259] In some embodiments, Ring L is an aryl. In some embodiments, the aryl is phenyl. In some embodiments, Ring L is heteroaryl. In some embodiments, Ring L is a bicyclic heteroaryl comprising 1, 2, or 3 heteroatoms selected from N and O.
[00260] In some embodiments, the second terminus comprises the structure of Formula (15-B), or a pharmaceutically acceptable salt thereof:
Formula (15-B), wherein: each R48 is hydrogen, halogen, -OH, -CN, -NO2, -NH2, C1-C10 alkyl, Ci-Ciohaloalkyl, or C1-C10 hydroxyalkyl;
R49 and R50 are each independently hydrogen, optionally substituted C1-C10 alkyl, optionally substituted Ci- Ciohaloalkyl, optionally substituted C2-C10 alkenyl, or optionally substituted C2-Cio alkynyl;
R51 is hydrogen, halogen, -OH, -CN, -NO2, -NH2, optionally substituted C1-C10 alkyl, optionally substituted Ci-Ciohaloalkyl, optionally substituted C1-C10 hydroxyalkyl, optionally substituted C2-C10 alkenyl, or optionally substituted C2-Cio alkynyl; and p? is 1-4.
[00261] In some embodiments, wherein the second terminus comprises the structure of Formula ( 15-C), or a pharmaceutically acceptable salt thereof:
Formula (15-C), wherein:
X is CR48 or N; each R48 is hydrogen, halogen, -OH, -CN, -NO2, -NH2, C1-C10 alkyl, Ci-Ciohaloalkyl, or C1-C10 hydroxyalkyl;
R49 and R50 are each independently hydrogen, optionally substituted C1-C10 alkyl, optionally substituted Ci- Ciohaloalkyl, optionally substituted C2-C10 alkenyl, or optionally substituted C2-Cio alkynyl;
R51 is hydrogen, halogen, -OH, -CN, -NO2, -NH2, optionally substituted C1-C10 alkyl, optionally substituted Ci-Ciohaloalkyl, optionally substituted C1-C10 hydroxyalkyl, optionally substituted C2-C10 alkenyl, or optionally substituted C2-Cio alkynyl;
R53 is hydrogen or optionally substituted C1-C10 alkyl; and
p? is 1-3.
[00262] In some embodiments, each R48 is hydrogen, halogen, -OH, -CN, -NO2, -NH2, C1-C10 alkyl, C1-C10 haloalkyl, or optionally substituted C1-C10 hydroxyalkyl.
[00263] In some embodiments, R49 is hydrogen, optionally substituted C1-C10 alkyl, optionally substituted C1-C10 haloalkyl, optionally substituted C2-C10 alkenyl, or optionally substituted C2-Cio alkynyl. In some embodiments, R49 is an optionally substituted C1-C10 alkyl. In some embodiments, R49 is methyl, ethyl, isopropyl, or tert-butyl. In some embodiments, R49 is hydrogen.
[00264] In some embodiments, R50 is hydrogen, optionally substituted C1-C10 alkyl, optionally substituted C1-C10 haloalkyl, optionally substituted C2-C10 alkenyl, or optionally substituted C2-Cio alkynyl. In some embodiments, R50 is an optionally substituted C1-C10 alkyl or optionally substituted C2-C10 alkenyl. In some embodiments, R50 is hydrogen.
[00265] In some embodiments, R51 is hydrogen, halogen, -OH, -CN, -NO2, -NH2, optionally substituted C1-C10 alkyl, optionally substituted C1-C10 haloalkyl, optionally substituted C1-C10 hydroxyalkyl, optionally substituted C2-C10 alkenyl, or optionally substituted C2-Cioalkynyl.
[00266] In some embodiments, R52 is hydrogen, halogen, -OH, -CN, -NO2, -NH2, optionally substituted C1-C10 alkyl, optionally substituted C1-C10 haloalkyl, or optionally substituted C1-C10 hydroxyalkyl. In some embodiments, R52 is hydrogen.
[00267] In some embodiments, R53 is hydrogen or optionally substituted C1-C10 alkyl. In some embodiments, R53 is an optionally substituted C1-C10 alkyl. In some embodiments, R53 is methyl, ethyl, isopropyl, or tert-butyl. In some embodiments, R53 is hydrogen.
[00268] In some embodiments, p? is 4. In some embodiments, p? is 3. In some embodiments, p? is 2. In some embodiments, p? is 1.
[00269] In some embodiments, the second terminus comprises the structure of Formula (15-D1), (15-D2), or (15-D3), or a pharmaceutically acceptable salt thereof:
[00270] In some embodiments, the second terminus comprises the structure of Formula (15-E1), (15-E2), or (15-E3), or a pharmaceutically acceptable salt thereof:
[00271] In some embodiments, the second terminus comprises the structure of Formula (16-A), or a pharmaceutically acceptable salt thereof:
Formula (16-A), wherein:
B5 is -O-, -NH-, or S;
B6 is N or CH;
R54 is optionally substituted aryl or optionally substituted heteroaryl; each R55 is independently halogen, -OH, -CN, -NO2, -NH2, optionally substituted C1-C10 alkyl, optionally substituted C1-C10 haloalkyl, optionally substituted C1-C10 heteroalkyl, or optionally substituted C1-C10 hydroxyalkyl;
R56 is hydrogen, optionally substituted C1-C10 alkyl, optionally substituted Ci-Ciohaloalkyl, or optionally substituted C1-C10 hydroxyalkyl;
R57 is halogen, -OH, -CN, -NO2, -NH2, optionally substituted C1-C10 alkyl, optionally substituted C1-C10 haloalkyl, or optionally substituted C1-C10 hydroxyalkyl;
pg is 1-3; and q? is 0-2.
[00272] In some embodiments, B5 is -O- or -S-. In some embodiments, B5 is -O-. In some embodiments, B5 is -S-.
[00273] In some embodiments, B6 is N. In some embodiments, B6 is CH.
[00274] In some embodiments, R54 is an optionally substituted aryl. In some embodiments, R54 is phenyl optionally substituted with one or more halogen, -CN, -NH2, -OH, C1-C10 alkyl, Ci-Ciohaloalkyl, or C1-C10 hydroxyalkyl.
[00275] In some embodiments, each R55 is independently halogen, -OH, -CN, -NH2, optionally substituted C1-C10 alkyl, optionally substituted Ci-Ciohaloalkyl, or optionally substituted C1-C10 hydroxyalkyl.
[00276] In some embodiments, R56 is optionally substituted C1-C10 alkyl, optionally substituted C1-C10 haloalkyl, or optionally substituted C1-C10 hydroxyalkyl. In some embodiments, R56 is optionally substituted C1-C10 alkyl.
[00277] In some embodiments, R57 is halogen, -OH, -CN, -NO2, -NH2, or optionally substituted C1-C10 alkyl.
[00278] In some embodiments, pg is 3. In some embodiments, pg is 2. In some embodiments, pg is 1. [00279] In some embodiments, q? is 2. In some embodiments, q? is 1. In some embodiments, q? is 0. [00280] In some embodiments, the second terminus comprises the structure of Formula (16-B), or a pharmaceutically acceptable salt thereof:
Formula (16-B).
[00281] In some embodiments, the second terminus comprises the structure of Formula (17-A), or a pharmaceutically acceptable salt thereof:
Formula (17-A), wherein:
Ring M is optionally substituted aryl or optionally substituted heteroaryl;
Ring N is absent or 4 to 8-membered heterocycloalkyl;
A6 is -O-, -NH-, or -CH2-; each R58 is independently halogen, -OH, -CN, -NO2, -NH2, Ci-Cio alkyl, Ci-Ciohaloalkyl, or Ci-Cw hydroxyalkyl;
R59 is hydrogen, -OH, -NH2, Ci-Cio alkyl, Ci-Ciohaloalkyl, Ci-Cio hydroxyalkyl, or -NH-Ci-Cio alkyl;
R60 is hydrogen or optionally substituted Ci-Cio alkyl; and
Pio is 1-4; wherein Formula (17-A) is connected to the linker through R59.
[00282] In some embodiments, Ring M is an aryl, optionally substituted with one or more halogen, CN, NH2, OH, CI-CIO alkyl, Ci-Ciohaloalkyl, or C1-C10 hydroxyalkyl. In some embodiments, Ring M is phenyl. In some embodiments, Ring M is an optionally substituted 6-membered heteroaryl, optionally substituted with one or more halogen, CN, NH2, OH, Ci-Cio alkyl, Ci-Ciohaloalkyl, or Ci-Cio hydroxyalkyl. In some embodiments, Ring M is an optionally substituted pyridine.
[00283] In some embodiments, Ring N is 4 to 8-membered heterocycloalkyl. In some embodiment, Ring N is a 4-membered heterocycloalkyl. In some embodiments, Ring N is a 5 -membered heterocycloalkyl. In some embodiments, Ring N is a 6-membered heterocycloalkyl. In some embodiments, Ring N is absent.
[00284] In some embodiments, A6 is -O- or -NH-. In some embodiments, A6 is -CH2-.
[00285] In some embodiments, each R58 is independently -OH, -NH2, Ci-Cio alkyl, Ci-Ciohaloalkyl, or Ci- Cio hydroxyalkyl. In some embodiments, each R58 is independently Ci-Cio alkyl, or Ci-Cio hydroxyalkyl. In some embodiments, each R58 is independently Ci-Cio hydroxyalkyl.
[00286] In some embodiments, R59 is -OH, -NH2, Ci-Cio hydroxyalkyl, or -NH-Ci-Cio alkyl. In some embodiments, R59 is hydrogen.
[00287] In some embodiments, R60 is optionally substituted Ci-Cio alkyl. In some embodiments, R60 is methyl. In some embodiments, R60 is hydrogen.
[00288] In some embodiments, pio is 3 or 4. In some embodiments, pio is 2. In some embodiments, pio is 1. [00289] In some embodiments, the second terminus comprises the structure of Formula (17-B) of Formula (17-C), or a pharmaceutically acceptable salt thereof:
[00290] In some embodiments, the second terminus comprises the structure of Formula (18-A), or a pharmaceutically acceptable salt thereof:
Formula (18-A), wherein:
B7 is N or CH;
R67 is hydrogen, halogen, -OH, -CN, -NO2, -NH2, optionally substituted C1-C10 alkyl, optionally substituted Ci-Ciohaloalkyl, optionally substituted C1-C10 heteroalkyl, or optionally substituted C1-C10 hydroxyalkyl;
R68 and R69 are each independently an optionally substituted 5-membered heteroaryl; and x5 and xe are each independently 0-4.
[00291] In some embodiments, B7 is N. In some embodiments, B7 is CH.
[00292] In some embodiments, R67 is halogen, -OH, -CN, -NO2, -NH2, optionally substituted C1-C10 alkyl, Ci-Ciohaloalkyl, or optionally substituted Ci-Ciohydroxyalkyl. In some embodiments, R67 is halogen, -OH, -CN, -NO2, -NH2, or -CH3.
[00293] In some embodiments, R68 is optionally substituted oxazole, oxadiazole, thiazole, thiadiazole, pyrrole, or pyrazole. In some embodiments, R68 is pyrrole or pyrazole. In some embodiments, R68 is pyrrole. In some embodiments, R68 is pyrazole.
[00294] In some embodiments, R69 is optionally substituted oxazole, oxadiazole, thiazole, thiadiazole, pyrrole, or pyrazole. In some embodiments, R69 is pyrrole or pyrazole. In some embodiments, R69 is pyrrole. In some embodiments, R69 is pyrazole.
[00295] In some embodiments, X5 is 2 or 3. In some embodiments, X5 is 1. In some embodiments, X5 is 0.
[00296] In some embodiments, xe is 3. In some embodiments, x„ is 2. In some embodiments, xe is 1. In some embodiments, xe is 0.
[00297] In some embodiments, the second terminus comprise Formula (18-B), or a pharmaceutically acceptable salt thereof:
Formula (18-B).
[00298] In some embodiments, the second terminus comprises Formula (19-A), or a pharmaceutically acceptable salt thereof:
Formula (19-A).
[00299] In some embodiments, the second terminus comprises the structure of Formula (20-A), or a pharmaceutically acceptable salt thereof:
Formula (20-A), wherein:
A7 is -NHC(O)- or -NHS(O)2-;
A8 is -O- or -NH-; each R61 is independently hydrogen or Ci-Ce alkyl; each R62 is independently halogen, -CN, -NO2, -OH, -OR62a, -NR62aR62b, -C(O)R62a, optionally substituted C1-C10 alkyl, optionally substituted Ci-Ciohaloalkyl, optionally substituted C1-C10 heteroalkyl, or optionally substituted C1-C10 hydroxyalkyl; wherein each R62a and R62b is independently hydrogen or C1-C10 alkyl, wherein each R63 is independently -OR63a, -NR63aR63b, optionally substituted C1-C10 alkyl, optionally substituted Ci- Ciohaloalkyl, optionally substituted C1-C10 heteroalkyl, or optionally substituted C1-C10 hydroxyalkyl; wherein each R63a and R63b is independently hydrogen, C1-C10 alkyl, C1-C10 alkylamino, Ci-Ciohaloalkyl, or Ci -C10 hydroxyalkyl; pis is 1-3; and pi4 is 1-3; wherein the linker is connected to Formula (20-A) through one of R62 or one of R63.
[00300] In some embodiments, A7 is -NHC(O)-. In some embodiments, A7 is -NHS(O)2-.
[00301] In some embodiments, A8 is -O-. In some embodiments, A8 is -NH-.
[00302] In some embodiments, each R61 is independently Ci-Ce alkyl. In some embodiments, each R61 is methyl. In some embodiments, each R61 is hydrogen.
[00303] In some embodiments, each R62 is independently optionally substituted C1-C10 alkyl, optionally substituted Ci-Cio haloalkyl, optionally substituted C1-C10 heteroalkyl, or optionally substituted C1-C10 hydroxyalkyl. In some embodiments, each R62 is independently optionally substituted C1-C10 alkyl. In some
embodiments, each R62 is independently optionally substituted Ci-Ciohaloalkyl. In some embodiments, each R62 is independently optionally substituted C1-C10 heteroalkyl. In some embodiments, each R62 is independently optionally substituted C1-C10 hydroxyalkyl.
[00304] In some embodiments, each R63 is independently optionally substituted C1-C10 alkyl, optionally substituted Ci-Cio haloalkyl, optionally substituted C1-C10 heteroalkyl, or optionally substituted Ci-Cw hydroxyalkyl. In some embodiments, each R63 is independently optionally substituted C1-C10 alkyl. In some embodiments, each R66 is independently optionally substituted Ci-Ciohaloalkyl. In some embodiments, each R63 is independently optionally substituted C1-C10 heteroalkyl. In some embodiments, each R63 is independently optionally substituted C1-C10 hydroxyalkyl.
[00305] In some embodiments, pis is 1. In some embodiments, pis is 2. In some embodiments, pis is 3. [00306] In some embodiments, pi4 is 1. In some embodiments, pi4 is 2. In some embodiments, pi4 is 3. [00307] In some embodiments, the linker is connected to Formula (20-A) through one of R62. In some embodiments, the linker is connected to Formula (20-A) through one of R63.
[00308] In some embodiments, the second terminus comprises the structure of Formula (20-B) or Formula (20-B), or a pharmaceutically acceptable salt thereof:
[00309] In some embodiments, the second terminus comprises the structure of Formula (21 -A), or a pharmaceutically acceptable salt thereof:
Formula (21-A), wherein:
Ring Q is Cs-Cs cycloalkyl or 4 to 8-membered heterocycloalkyl;
A9 is -O-, -NH-, or -NHC(O)-; each R64 is independently hydrogen or Ci-Ce alkyl; each R65 is independently halogen, -CN, -NO2, -OH, -OR62a, -NR62aR62b, optionally substituted C1-C10 alkyl, optionally substituted Ci-Ciohaloalkyl, optionally substituted C1-C10 heteroalkyl, or optionally substituted C1-C10 hydroxyalkyl; wherein each R62a and R62b is independently hydrogen or C1-C10 alkyl; each R66 is independently halogen, optionally substituted C1-C10 alkyl, optionally substituted Ci-Cw haloalkyl, optionally substituted C1-C10 heteroalkyl, or optionally substituted C1-C10 hydroxyalkyl; pis is 0-3; and
Pi6 is 1-3; wherein Formula (21 -A) is connected to the linker at one of R65 or one of R66.
[00310] In some embodiments, A9 is -O-. In some embodiments, A9 is -NH-. In some embodiments, A9 is - NHC(O)-.
[00311] In some embodiments, Ring Q is Cs-Cs cycloalkyl. In some embodiments, Ring Q is 4 to 8- membered heterocycloalkyl.
[00312] In some embodiments, each R64 is independently Ci-Ce alkyl. In some embodiments, each R64 is methyl. In some embodiments, each R64 is hydrogen.
[00313] In some embodiments, each R65 is independently optionally substituted C1-C10 alkyl, optionally substituted Ci-C 10 haloalkyl, optionally substituted C1-C10 heteroalkyl, or optionally substituted C1-C10 hydroxyalkyl. In some embodiments, each R65 is independently optionally substituted C1-C10 alkyl. In some embodiments, each R65 is independently optionally substituted C1-C10 haloalkyl. In some embodiments, each R65 is independently optionally substituted C1-C10 heteroalkyl. In some embodiments, each R65 is independently optionally substituted C1-C10 hydroxyalkyl.
[00314] In some embodiments, each R66 is independently optionally substituted C1-C10 alkyl, optionally substituted Ci-C 10 haloalkyl, optionally substituted C1-C10 heteroalkyl, or optionally substituted C1-C10 hydroxyalkyl. In some embodiments, each R66 is independently optionally substituted C1-C10 alkyl. In some embodiments, each R66 is independently optionally substituted C1-C10 haloalkyl. In some embodiments, each R66 is independently optionally substituted C1-C10 heteroalkyl. In some embodiments, each R66 is independently optionally substituted C1-C10 hydroxyalkyl.
[00315] In some embodiments, pis is 0. In some embodiments, pis is 1. In some embodiments, pis is 2. In some embodiments, pis is 3.
[00316] In some embodiments, pie is 1. In some embodiments, pie is 2. In some embodiments, pie is 3. [00317] In some embodiments, Formula (21-A) is connected to the linker at one of R65. In some embodiments, Formula (21-A) is connected to the linker at one of R66.
[00318] In some embodiments, the second terminus comprises the structure of Formula (21-B), or a pharmaceutically acceptable salt thereof:
Formula (21-B).
[00319] In some embodiments, the second terminus comprises the structure of Formula (22-A), or a pharmaceutically acceptable salt thereof:
Formula (22-A), wherein:
R67 is hydrogen, -NH2, or -NHSO2CH3;
R68 and R69 are each independently halogen, -CN, -NO2, -OH, optionally substituted C1-C10 alkyl, optionally substituted C1-C10 haloalkyl, or optionally substituted C1-C10 hydroxyalkyl;
R70 is -S(O)R70a or -S(O)2R70a, wherein R70a is hydrogen or C1-C10 alkyl; and
R71 is hydrogen or C1-C10 alkyl; wherein Formula (22-A) is attached to the linker at R67 or R71.
[00320] In some embodiments, R67 is hydrogen. In some embodiments, R67 is -NH2, or -NHSO2CH3.
[00321] In some embodiments, R68 and R69 are each independently optionally substituted C1-C10 alkyl. In some embodiments, R68 and R69 are each methyl.
[00322] In some embodiments, R70 is -S(O)2R70a. In some embodiments, R70 is -S(O)2CH3.
[00323] In some embodiments, Formula (22-A) is attached to the linker at R67. In some embodiments,
Formula (22-A) is attached to the linker at R71.
[00324] In some embodiments, the second terminus comprises the structure of Formula (22-B) or Formula
Formula (23-A) wherein:
B9 is -CH-CRB9-, -N-, or NRB1°;
B10 is -N- or NRB1°, wherein
RB9 is hydrogen or halogen; and RB1° is hydrogen;
R72 is halogen, -CN, -NO2, -OH, optionally substituted C1-C10 alkyl, optionally substituted C1-C10 haloalkyl, or optionally substituted C1-C10 hydroxyalkyl; and
R73 is hydrogen; wherein Formula (23-A) is attached to the linker at RB9, RB1°, or R73.
[00326] In some embodiments, B9 is -CH-CRB9- and B10 is -N-. In some embodiments, B9 is -N- and B10 is
NRB1°. In some embodiments, B9 is NRB1° and B10 is -N-.
[00327] In some embodiments, R72 is an optionally substituted C1-C10 alkyl. In some embodiments, R72 is methyl.
[00328] In some embodiments, Formula (23-A) is attached to the linker at RB9 or RB1°. In some embodiments, Formula (23-A) is attached to the linker at R73.
[00329] In some embodiments, the second terminus comprises the structure of Formula (23-B) or Formula (23 -C), or a pharmaceutically acceptable salt thereof:
[00330] In some embodiments, the second terminus comprises the structure of Formula (23-D) or Formula (23 -E), or a pharmaceutically acceptable salt thereof:
[00331] In some embodiments, the second terminus comprises the structure of Formula (24-A), or a pharmaceutically acceptable salt thereof:
Formula (24-A), wherein:
R74 is -CH2-(optionally substituted 5 -membered heteroaryl);
R75 is -0-(Ci-Cio alkyl)-R75a; wherein R75a is -C(O)O-alkyl;
R76 is -OR76a, wherein R76a is hydrogen or alkyl;
R77 is hydrogen or halogen; and
R77a is hydrogen or Ci-Cw alkyl; wherein Formula (24-A) is attached to the linker at R75a or R76a.
[00332] In some embodiments, R75 is -0-(Ci-Cio alkyl)-C(O)O-alkyl. In some embodiments, R75 is -O-(Ci- C4 alkyl)-C(O)O-Ci-C4 alkyl.
[00333] In some embodiments, R76 is -OH. In some embodiments, R76 is -OCHs. In some embodiments, R76 is -O-CH2CH3.
[00334] In some embodiments, R77 is halogen. In some embodiments, R77 is hydrogen.
[00335] In some embodiments, R77a is hydrogen or methyl. In some embodiments, R77a is methyl. In some embodiments, R77a is hydrogen.
[00336] In some embodiments, Formula (24-A) is attached to the linker at R75a. In some embodiments, Formula (24-A) is attached to the linker at R76a.
[00337] In some embodiments, the second terminus comprises the structure of Formula (24-B) or Formula
[00338] In some embodiments, the second terminus comprises the structure of Formula (25-A), or a pharmaceutically acceptable salt thereof:
Formula (25 -A), wherein:
B12 is -NR80a- or -C(R80b)2-;
R78 is halogen, -CN, -NO2, -OH, or optionally substituted C1-C10 alkyl;
R79a and R79b are each independently halogen, -CN, -NO2, -NH2, -N(CH3)2, or optionally substituted C1-C10 alkyl; or R79a and R79b together with the atoms to which they are attached form a 6-membered aryl;
each R80 is independently hydrogen, -O-C1-C3 alkyl, C1-C10 alkyl, or C1-C10 alkoxy;
R80a is Ci-C6 alkyl; each R80b is independently hydrogen or Ci-Ce alkyl; and r7 is 1, 2, or 3; wherein Formula (25 -A) is attached to the linker at one of R80.
[00339] In some embodiments, B12is -NR80a-. In some embodiments, B12is -C(R80b)2-.
[00340] In some embodiments, R78 is -CN, -NO2, or -OH. In some embodiments, R78 is -OH. In some embodiments, R78 is halogen. In some embodiments, R78 is an optionally substituted C1-C10 alkyl.
[00341] In some embodiments, R79a and R79b are each independently halogen, -NH2, or -N(CH3)2.
[00342] In some embodiments, R79a and R79b together with the atoms to which they are attached form a 6- membered aryl.
[00343] In some embodiments, each R80 is independently hydrogen. In some embodiments, each R80 is independently C1-C10 alkyl. In some embodiments, each R80 is independently C1-C10 alkoxy. In some embodiments, each R80 is independently -OCH3.
[00344] In some embodiments, the second terminus comprises the structure of Formula (25-B) or Formula (25 -C), or a pharmaceutically acceptable salt thereof:
u .
[00345] In some embodiments, the second terminus comprises the structure of Formula (26-A), or a pharmaceutically acceptable salt thereof:
Formula (26-A), wherein:
Bn is -O- or -NR82-;
A10 is -NR84- or -NR84CH2-;
R81a and R81b are each independently C1-C10 alkyl; or R81a and R81b combine together to form oxo (=0);
R82 is hydrogen or C1-C3 alkyl; each R83 is independently halogen, -CN, -NO2, -OH, -OR83a, or optionally substituted C1-C10 alkyl; wherein
R83a is C1-C10 alkyl;
R84 is hydrogen, C1-C3 alkyl, Cs-Ce cycloalkyl, or phenyl;
R85 is hydrogen or C1-C10 alkyl; and r4 is 1, 2, or 3; wherein Formula (26-A) is attached to the linker at one of R83.
[00346] In some embodiments, B11 is -O-. In some embodiments, B11 is -NR82-. In some embodiments, B11 is NCH3.
[00347] In some embodiments, A10 is -NR84-. In some embodiments, A10 is -NR84CH2-.
[00348] In some embodiments, R81a and R81b are each methyl. In some embodiments, R81a and R81b combine together to form oxo (=0).
[00349] In some embodiments, each R83 is independently -CN, -OH, -OR83a, or optionally substituted Ci-
C10 alkyl. In some embodiments, each R83 is independently -OH or -OR83a.
[00350] In some embodiments, R84 is C3-C6 cycloalkyl or phenyl. In some embodiments, R84 is phenyl.
[00351] In some embodiments, R85 is C1-C10 alkyl. In some embodiments, R85 is methyl. In some embodiments, R85 is hydrogen.
[00352] In some embodiments, the second terminus comprises the structure of Formula (26-B) or Formula
[00353] In some embodiments, the second terminus comprises the structure of Formula (26-D) or Formula (26-E), or a pharmaceutically acceptable salt thereof:
Formula (26-E).
[00354] In some embodiments, the second terminus comprises the structure of Formula (27-A), or a pharmaceutically acceptable salt thereof:
Formula (27-A).
[00355] In some embodiments, the second terminus comprises the structure of Formula (28-A), or a pharmaceutically acceptable salt thereof:
Formula (28-A), wherein:
A11 is Ci-Ce alkylene;
B13 is N or CR100;
R97 is Ci-C6 alkyl;
R98 is halogen;
R" is hydrogen, halogen, or optionally substituted C1-C10 alkyl; and
R100 is -OR100a or optionally substituted C1-C10 alkyl; wherein R100a is hydrogen or C1-C10 alkyl; or R" and R100 together with the atoms to which they are attached from a heterocycloalkyl.
[00356] In some embodiments, A11 is C1-C4 alkylene. In some embodiments, A11 is -CH2CH2-.
[00357] In some embodiments, B13 is N. In some embodiments, B13 is CR100.
[00358] In some embodiments, R97 is methyl.
[00359] In some embodiments, R98 is -Cl.
[00360] In some embodiments, R100 is -OR100a. In some embodiments, R100 is an optionally substituted Ci- C10 alkyl.
[00361] In some embodiments, R" and R100 together with the atoms to which they are attached from a heterocycloalkyl.
[00362] In some embodiments, the second terminus comprises the structure of Formula (28-B), or a pharmaceutically acceptable salt thereof:
Formula (28-B).
[00363] In some embodiments, the second terminus comprises the structure of Formula (28-C), or a pharmaceutically acceptable salt thereof:
Formula (28-C).
[00364] In some embodiments, the second terminus comprises the structure of Formula (29-A), or a pharmaceutically acceptable salt thereof:
Formula (29-A).
[00365] In some embodiments, the second terminus comprises the structure of Formula (30-A), or a pharmaceutically acceptable salt thereof:
Formula (30-A).
[00366] In some embodiments, the second terminus comprises the structure of Formula (31 -A), or a pharmaceutically acceptable salt thereof:
Formula (31 -A), wherein:
R94 is halogen;
R94a js hydrogen or C1-C3 alkyl; and
R95 is hydrogen, halogen, -OH, -OCH3, or optionally substituted C1-C10 alkyl.
[00367] In some embodiments, R94is -Cl.
[00368] In some embodiments, R94ais C1-C3 alkyl. In some embodiments, R94ais methyl. In some embodiments, R94ais hydrogen.
[00369] In some embodiments, R95 is halogen, -OH, -OCH3, or optionally substituted C1-C10 alkyl. In some embodiments, R95 is hydrogen.
[00370] In some embodiments, the second terminus comprises the structure of Formula (31-B) or Formula (31-C), or a pharmaceutically acceptable salt thereof:
Formula (31-B) or Formula (31-C).
[00371] In some embodiments, the second terminus comprises the structure of Formula (32-A), or a pharmaceutically acceptable salt thereof:
Formula (32-A).
[00372] In some embodiments, the second terminus comprises the structure of Formula (33 -A), or a pharmaceutically acceptable salt thereof:
[00373] In some embodiments, the second terminus comprises the structure of Formula (34-A), or a pharmaceutically acceptable salt thereof:
[00374] In some embodiments, the second terminus comprises the structure of Formula (35-A), or a pharmaceutically acceptable salt thereof:
[00375] In some embodiments, the second terminus comprises the structure of Formula (36-A) or Formula (36-B), or a pharmaceutically acceptable salt thereof:
[00376] In some embodiments, the second terminus comprises the structure of Formula (37-A), or a pharmaceutically acceptable salt thereof:
Formula (37-A), wherein:
Ring T is a Cs-Ce cycloalkyl, 5 to 6-membered heterocycloalkyl, or phenyl; each R86 is independently halogen, -CN, -NO2, -OH, -OR86a, -N(R86b)2, or optionally substituted C1-C10 alkyl; wherein
each R86a is independently C1-C10 alkyl; each R86b is independently hydrogen or C1-C10 alkyl;
R87 is Cs-Ce cycloalkyl or phenyl; each R88 is independently halogen, -CN, -NO2, -OH, -OR88a, -NR88bR88b, or optionally substituted C1-C10 alkyl; wherein each R88a is independently C1-C10 alkyl; each R88b is independently hydrogen or C1-C10 alkyl;
R89a and R89b are each independently hydrogen or C1-C10 alkyl; or R89a and R89b together with the atom to which they are attached form a 5 to 6-membered heterocycloalkyl; R96 is hydrogen or -CH3; and r5 and re are each independently 0, 1, 2, or 3.
[00377] In some embodiments, Ring T is a C -C, cycloalkyl. In some embodiments, Ring T is a 5 to 6- membered heterocycloalkyl. In some embodiments, Ring T is phenyl.
[00378] In some embodiments, each R86 is independently halogen, -OH, -OR86a, or -N(R86b)2. In some embodiments, each R86 is independently halogen or -N(R86b)2. In some embodiments, each R86 is independently -N(R86b)2.
[00379] In some embodiments, R87 is Cs-Ce cycloalkyl. In some embodiments, R87 is phenyl.
[00380] In some embodiments, R89a and R89b are each independently C1-C10 alkyl. In some embodiments, R89a and R89b are each hydrogen.
[00381] In some embodiments, R89a and R89b together with the atom to which they are attached form a 5 to 6-membered heterocycloalkyl.
[00382] In some embodiments, R96 is -CH3. In some embodiments, R96 is hydrogen.
[00383] In some embodiments, rs and re are each independently 0, 1, or 2.
[00384] In some embodiments, the second terminus comprises the structure of Formula (37-B) or Formula (37-C), or a pharmaceutically acceptable salt thereof:
Formula (37-B) or
(Formula 37-C).
o o o
or a pharmaceutically acceptable salt thereof.
[00386] In some embodiments, the second terminus is selected from a moiety described in Table 1, or a pharmaceutically acceptable salt thereof.
Oligomeric Backbone - Linker Moiety
[00387] The oligomeric backbone is a linker that connects the first terminus and the second terminus and brings the regulatory molecule in proximity to the target gene to modulate gene expression.
[00388] The length of the linker depends on the type of regulatory protein and also the target gene. In some embodiments, the linker has a length of less than about 50 Angstroms. In some embodiments, the linker has a length of about 20 to 30 Angstroms.
[00389] In some embodiments, the oligomeric backbone comprises between 5 and 50 chain atoms. [00390] In some embodiments, the oligomeric backbone comprises a multimer having 2 to 50 spacing moieties, wherein each spacing moiety is independently selected from the group consisting of -((CRlbRlb)x-O)y-, - ((CRlbRlb)x-NRla)y-, -((CRlbRlb)x-CH=CH-(CRlbRlb)x-O)y-, optionally substituted C1-C12 alkyl, optionally substituted C2-C10 alkenyl, optionally substituted C2-Cio alkynyl, optionally substituted Ce-C 10 arylene, optionally substituted C3-C7 cycloalkylene, optionally substituted 5- to 10-membered heteroarylene, optionally substituted 4- to 10-membered heterocycloalkylene, amino acid residue, -O-, -C(O)NRla-, - NRlaC(O)-, -C(O)-, -NRla-, -C(O)O-, -S-, -S(O)-, -S(O)2-, -S(O)2NRla-, -NRlaS(O)2-, and -P(O)OH-, and any combinations thereof; wherein
each x is independently 2-4; each y is independently 1-10; each Rla is independently, hydrogen, optionally substituted Ci-Ce alkyl, or optionally substituted Ci- C„ haloalkyl; and each Rlb is independently selected from hydrogen, halogen, optionally substituted alkyl, optionally substituted alkenyl, optionally substituted alkynyl, optionally substituted alkoxy, optionally substituted amino, carboxyl, carboxyl ester, acyl, acyloxy, acyl amino, amino acyl, optionally substituted alkylamide, sulfonyl, optionally substituted thioalkoxy, optionally substituted aryl, optionally substituted heteroaryl, optionally substituted cycloalkyl, and optionally substituted heterocyclyl.
[00391] In some embodiments, the oligomeric backbone comprises a multimer having 2 to 50 spacing moieties, wherein each spacing moiety is independently selected from the group consisting of optionally substituted C1-C12 alkyl, -((CH2)X-O)y-, -((CH2)x-NH)y-, -O-, -C(O)NH-, -NH-, and any combinations thereof.
[00392] In some embodiments, the oligomeric backbone comprises -(T1-V1)a-(T2-V2)b-(T3-V3)c-(T4-V4)d- (T5-V5)e-, wherein a, b, c, d and e are each independently 0 or 1, and where the sum of a, b, c, d and e is 1 to 5;
T1, T2, T3, T4 and T5 are each independently selected from an optionally substituted C1-C12 alkylene, optionally substituted alkenylene, optionally substituted alkynylene, (EA)W, (EDA)m, (PEG)n, (modified PEG) n, (AA)P, -(CRlbOH)h-, optionally substituted Ce-Cio arylene, optionally substituted C3-C7 cycloalkylene, optionally substituted 5- to 10-membered heteroarylene, optionally substituted 4- to 10- membered heterocycloalkylene, an acetal group, a disulfide, a hydrazine, a carbohydrate, a beta-lactam, and an ester,
(a) w is an integer from 1 to 20;
(b) m is an integer from 1 to 20;
(c) n is an integer from 1 to 30;
(d) p is an integer from 1 to 20;
(e) h is an integer from 1 to 12;
(g) EDA has the following structure:
wherein each q is independently an integer from 1 to 6, each x is independently an integer from 1 to 4, and each r is independently 0 or 1 ;
(h) (PEG)n has the structure of-(CRlbRlb-CRlbRlb-O)n-CRlbRlb-;
(i) (modified PEG)n has the structure of replacing at least one -(CRlbRlb-CRlbRlb-O)- in (PEG)n with -(CH2-CRlb=CRlb-CH2-O)- or -(CRlbRlb-CRlbRlb-S)-;
(j) AA is an amino acid residue;
(k) V1, V2, V3, V4 and V5 are each independently selected from the group consisting of a bond, C(O)-, -NRla-, -C(O)NRla-, -NRlaC(O)-, -CONRla-Ci-C4 alkyl-, -NRlaC(O)-Ci-C4 alkyl-, -C(O)O-, -OC(O)-, -O-, -S-, -S(O)-, -S(O)2-, -S(O)2NRla-, -NRlaS(O)2- and -P(O)OH-;
(l) each Rla is independently hydrogen, optionally substituted Ci-Ce alkyl, or optionally substituted Ci-Ce haloalkyl; and
(m) each Rlb is independently selected from hydrogen, halogen, alkyl, substituted alkyl, alkenyl, substituted alkenyl, alkynyl, substituted alkynyl, halogen, alkoxy, substituted alkoxy, amino, substituted amino, carboxyl, carboxyl ester, acyl, acyloxy, acyl amino, amino acyl, alkylamide, substituted alkylamide, sulfonyl, thioalkoxy, substituted thioalkoxy, aryl, substituted aryl, heteroaryl, substituted heteroaryl, cycloalkyl, substituted cycloalkyl, heterocyclyl, and substituted heterocyclyl
[00393] In some embodiments, the a, b, c, d and e are each independently 0 or 1, where the sum of a, b, c, d and e is 1. In some embodiments, the a, b, c, d and e are each independently 0 or 1, where the sum of a, b, c, d and e is 2. In some embodiments, the a, b, c, d and e are each independently 0 or 1, where the sum of a, b, c, d and e is 3. In some embodiments, the a, b, c, d and e are each independently 0 or 1, where the sum of a, b, c, d and e is 4. In some embodiments, the a, b, c, d and e are each independently 0 or 1, where the sum of a, b, c, d and e is 5.
[00394] In some embodiments, n is 3-9. In some embodiments, n is 4-8. In some embodiments, n is 5 or 6. [00395] In some embodiments, T1, T2, T3, and T4, and T5 are each independently selected from C1-C12 alkyl, substituted C1-C12 alkyl, (EA)W, (EDA)m, (PEG)n, (modified PEG)n, (AA)P, -(CRlbOH)h-, phenyl, substituted phenyl, piperidin-4-amino (P4A), para-amino-benzyloxycarbonyl (PABC), meta-amino- benzyloxycarbonyl (MABC), para-amino-benzyloxy (PABO), meta-amino-benzyloxy (MABO), paraaminobenzyl, an acetal group, a disulfide, a hydrazine, a carbohydrate, a beta-lactam, an ester, (AA)P- MABC-(AA)p, (AA)p-MABO-(AA)p, (AA)p-PABO-(AA)p and (AA)p-PABC-(AA)p. In some embodiments, piperidin-4-amino (P4A) is
, wherein Rla is hydrogen or Ci-Ce alkyl.
[00396] In some embodiments, T1, T2, T3, T4 and T5 are each independently selected from (Ci-Ci2)alkyl, substituted C1-C12 alkyl, (EA)W, (EDA)m, (PEG)n, (modified PEG)n, (AA)p,-(CR2aOH)h-, optionally substituted Ce-Cio arylene, 4 to 10-membered heterocycloalkene, optionally substituted 5 to 10-membered heteroarylene. In some embodiments, EA has the following structure:
[00397] In some embodiments, x is 2-3 and q is 1-3 for EA and EDA. In some embodiments, Rla is hydrogen or Ci-Ce alkyl.
[00398] In some embodiments, T4or T5 is an optionally substituted Ce-Cio arylene.
[00399] In some embodiments, T4or T5 is phenylene or substituted phenylene. In some embodiments, T4 or T5 is phenylene or phenylene substituted with 1-3 substituents selected from Ci-Ce alkyl, halogen, OH or amine. In some embodiments, T4or T5 is 5 to 10-membered heteroarylene or substituted heteroarylene. In some embodiments, T4 or T5 is 4 to 10-membered heterocyclene or substituted heterocylcylene. In some embodiments, T4or T5 is heteroarylene or heterocylene optionally substituted with 1-3 substituents selected from Ci-Ce alkyl, halogen, OH or amine.
[00400] In some embodiments, T1, T2, T3, T4 and T5 and V1, V2, V3, V4 and V5 are selected from the following Table 2.
[00401] In some embodiments, the oligomeric backbone comprises N(Rla)(CH2)xN(Rla)(CH2)xN-, wherein each Rla is independently selected from hydrogen, optionally substituted Ci-Ce alkyl, or optionally substituted Ci-Ce haloalkyl; and each x is independently an integer in the range of 1-6.
[00402] In some embodiments, the oligomeric backbone comprises -(CH2-C(O)N(Rla)-(CH2)q-N(Rla)- (CH2)q-N(Rla)C(O)-(CH2)x-C(O)N(Rla)-A-, -(CH2)x-C(O)N(Rla)-(CH2CH2O)y(CH2)x-C(O)N(Rla)-A-, or - C(O)N(Rla)-(CH2)q-N(Rla)-(CH2)q-N(Rla)C(O)-(CH2)x-A-; wherein each q is independently an integer from 2 to 10; each x is independently an integer from 1-6; and each A is independently selected from a bond, an optionally substituted C1-C12 alkyl, an optionally substituted Ce-Cio arylene, optionally substituted C3-C7 cycloalkylene, optionally substituted 5 to 10-membered heteroarylene, and optionally substituted 4 to 10- membered heterocycloalkylene.
[00403] In some embodiments, the oligomeric backbone comprises -(CH2CH2-O)X7- or -(CH2CH2-O)xs-A- (CH2CH2-O)X9-, wherein A is an optionally substituted 4 to 10-membered heterocycloalkylene or spirocyclene, and each x7, x§, and xg is independently an integer from 1-15.
[00404] In some embodiments, the oligomeric backbone comprises -NRla-(CH2CH2O)y(CH2)x- or -NRla- (CH2)q-C(O)NRla(CH2CH2O)y(CH2)x-, wherein q is 2-10, x is 1-4, y is 1-50, and each R4a is independently hydrogen or an optionally substituted Ci-Ce alkyl. In some embodiments, the oligomeric backbone comprises -NRla-(CH2CH2O)y(CH2)x,. In some embodiments, the oligomeric backbone comprises -NRla- (CH2)q-C(O)NRla(CH2CH2O)y(CH2)x-.
[00405] In some embodiments, the oligomeric backbone comprises -(CH2CH2-O)X-, -(CH2CH2-O)X- (CH2CH2)-NH-, -NH-(CH2CH2-O)X-, -NH-(CH2CH2-O)X-(CH2CH2)-NH-, -(CH2CH2-O)X-(CH2CH2)- NHC(O)-, or -NH-(CH2CH2-O)X-(CH2CH2)-NHC(O)-. In some embodiments, the oligomeric backbone comprises -NH-(CH2CH2-O)X- or -NH-(CH2CH2-O)X-(CH2CH2)-NH-. In some embodiments, the oligomeric backbone comprises -NH-(CH2CH2-O)X-. In some embodiments, the oligomeric backbone comprises -NH- (CH2CH2-O)X-(CH2CH2)-NH-.
[00406] In some embodiments, the oligomeric backbone comprises polyethylene glycol (PEG). In some embodiments, the oligomeric backbone comprises 1-20 PEG units. In some embodiments, the oligomeric backbone comprises 1, 2, 3, 4,5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, or 20 PEG units.
[00408] In some embodiments, A comprises a moiety having the structure, or a pharmaceutically acceptable salt thereof:
wherein:
A2 is absent or -C(O)-; and
R27 is an optionally substituted C1-C50 alkyl or optionally substituted C1-C50 heteroalkyl.
[00409] In some embodiments, A2 is -C(O)-. In some embodiments, A2 is absent.
[00410] In some embodiments, R27 is C1-C50 alkyl. In some embodiments, R27 is C1-C40 alkyl. In some embodiments, R27 is C1-C30 alkyl. In some embodiments, R27 is C1-C20 alkyl. In some embodiments, R27 is C1-C10 alkyl. In some embodiments, R27 is C1-C50 heteroalkyl. In some embodiments, R26 is C1-C40 heteroalkyl. In some embodiments, R27 is C1-C30 heteroalkyl. In some embodiments, R27 is C1-C20 heteroalkyl. In some embodiments, R27 is C1-C10 heteroalkyl. In some embodiments, the heteroalkyl is polyethylene glycol (PEG).
[00411] In some embodiments, the oligomeric backbone comprises a moiety having a structure of Formula (C-l), or a pharmaceutically acceptable salt thereof:
Formula (C-l), wherein:
Ring P is absent, arylene, or heterocycloalkylene;
L5 is absent, optionally substituted alkylene, or optionally substituted alkynylene;
B1 and B2 are each independently CH orN; si and S2 are each independently 0-3; and
** denotes attachment to the second terminus.
[00412] In some embodiments, Ring P is absent. In some embodiments, Ring P is C4-C7 heterocycloalkylene .
[00413] In some embodiments, B1 is N. In some embodiments, B1 is CH.
[00414] In some embodiments, B2 is N. In some embodiments, B2 is CH.
[00415] In some embodiments, L5 is absent.
[00416] In some embodiments, L5 is alkylene or alkynylene.
[00417] In some embodiments, L5 is -(CR1GR1G)xio-(alkylene)2-(CR1GR1G)yio-; wherein xio and yio are each independently 0 or 1; and each R1G is hydrogen or C1-C3 alkyl.
[00418] In some embodiments, the oligomeric backbone comprises a moiety having a structure of Formula (C-2), or a pharmaceutically acceptable salt thereof:
Formula (C-2), wherein:
L5 is absent, optionally substituted alkylene, or optionally substituted alkynylene; and
B2, B3 and B4 are each independently N or CH.
[00419] In some embodiments, each of B3 and B4 is independently N or CH; and B2 is N.
[00420] In some embodiments, L5 is C1-C3 alkylene or C1-C3 alkynylene. In some embodiments, L5 is Ci- C3 alkylene. In some embodiments, L5 is C1-C3 alkynylene. In some embodiments, L5 is -CH2-, -CH2CH2-, . In some embodiments, L5 is -CH2- or -CH2CH2-. In some embodiments, L5
ents,
[00421] In some embodiments, L5 is -(CR1GR1G)xio-(alkylene)2-(CR1GR1G)yio-; wherein xio and yio are each independently 0 or 1; and each R1G is hydrogen or C1-C3 alkyl.
[00422] In some embodiments, the oligomeric backbone comprises a moiety having the structure of Formula (C-3), or a pharmaceutically acceptable salt thereof:
Formula (C-3), wherein:
Si and s2 are each independently 0-3; n is an integer from 1-3;
R26 is an optionally substituted Ci-C2o alkylene or optionally substituted C2-C2o heteroalkylene; each R1G is independently hydrogen or C1-C3 alkyl; and
** denotes attachment to the second terminus.
[00423] In some embodiments, R26 is an optionally substituted Ci-C2o heteroalkylene. In some embodiments, R26 is PEG.
[00424] In some embodiments, each R1G is independently hydrogen. In some embodiments, R1G is independently C1-C3 alkyl. In some embodiments, the C1-C3 alkyl is methyl, ethyl or propyl. In some embodiments, each R1G is independently methyl.
[00425] In some embodiments, si and s2 are each independently is 0, 1, or 2. In some embodiments, si and s2 are each independently 0. In some embodiments, si and s2 are each independently 1.
[00426] In some embodiments, n is 1 or 2. In some embodiments, n is 1. In some embodiments, n is 2.
[00427] In some embodiments, the oligomeric backbone comprises:
[00428] In some embodiments, the oligomeric backbone is joined with the first terminus and/or with the second terminus with a group selected from -C(O)-, -NRla-, -C(O)NRla-, -NRlaC(O)-, -C(O)NRlaCi-C4alkyl- , -NRlaC(O)-Ci-C4alkyl-, -C(O)O-, -OC(O)-, -O-, -S-, -S(O)-, -S(O)2-, -S(O)2NRla-, -NRlaS(O)2-, -P(O)OH-, -((CH2)X-O)-, -((CH2)y-NRla)-, optionally substituted C1-C12 alkylene, optionally substituted C2-Cio alkenylene, optionally substituted C2-Cio alkynylene, optionally substituted Ce-Cio arylene, optionally substituted C3-C7 cycloalkylene, optionally substituted 5 to 10-membered heteroarylene, and optionally substituted 4 to 10-membered heterocycloalkylene, wherein each x is independently 1-4, each y is independently 1-4, and each Rla is independently a hydrogen, optionally substituted Ci-Ce alkyl, or optionally substituted Ci-Ce haloalkyl .
[00429] In some embodiments, the oligomeric backbone is joined with the first terminus with a group selected from -O-, -C(O)-, -NRla-, C1-C12 alkyl, -C(O)NRla-, and -NRlaC(O)-. In some embodiments, the oligomeric backbone is joined with the first terminus with a group selected from -O- or -NRla-.
[00430] In some embodiments, the oligomeric backbone is joined with the second terminus with a group selected from -C(O)-, -NRla-, -C(O)NRla-, -NRlaC(O)-, -((CH2)X-O)-, -((CH2)y-NRla)-, -O-, optionally substituted C1-C12 alkyl, optionally substituted Ce-Cio arylene, optionally substituted C3-C7 cycloalkylene, optionally substituted 5- to 10-membered heteroarylene, and optionally substituted 4- to 10-membered heterocycloalkylene, wherein each x is independently 1-4, each y is independently 1-4, and each Rla is independently a hydrogen, optionally substituted Ci-Ce alkyl, or optionally substituted Ci-Ce haloalkyl.
[00431] In some embodiments, the oligomeric backbone is joined with the second terminus with a group selected from -O-, -C(O)-, -NRla-, C1-C12 alkyl, -C(O)NRla-, and -NRlaC(O)-. In some embodiments, the oligomeric backbone is joined with the second terminus with a group selected from -O- or -NRla-. In some embodiments, the oligomeric backbone is joined with the second terminus with -O-. In some embodiments,
the oligomeric backbone is joined with the second terminus with -NRla-. In some embodiments, the oligomeric backbone is joined with the second terminus with -NH-.
[00432] Any combination of the groups described above for the various variables is contemplated herein. Throughout the specification, groups and substituents thereof are chosen by one skilled in the field to provide stable moieties and compounds.
[00433] In some embodiments, non-limiting examples of the transcription modulator compounds described herein are presented below in Table 3 (next page).
Methods of Use
[00434] In another aspect, provided herein is a method of decreasing expression of a gene in a cell, the method comprising contacting the cell with an effective amount of a molecule disclosed herein, or a pharmaceutically acceptable salt thereof.
[00435] In some embodiments, the gene is huntingtin (HIT). In some embodiments, the gene is mutant huntingtin.
[00436] In some embodiments, the expression of the gene is at least about 10%, at least about 20%, at least about 30%, at least about 40%, at least about 50%, at least about 60%, at least about 70%, at least about 80%, at least about 90%, at least about 95%, or more.
[00437] In another aspect, provided herein is a method of treating Huntington’s disease (HD) in a subject in need thereof, the method comprising administering to the subject a molecule described herein, or a pharmaceutically acceptable salt thereof.
[00438] In another aspect, provided herein is a method of treating Huntington’s disease-like syndrome [00439] in a subject in need thereof, the method comprising administering to the subject a molecule described herein, or a pharmaceutically acceptable salt thereof.
[00440] In another aspect, provided herein is a method of treating Juvenile Huntington’s disease in a subject in need thereof, the method comprising administering to the subject a molecule described herein, or a pharmaceutically acceptable salt thereof.
[00441] In some embodiments, the method reduces one or more symptoms of Huntington’s disease (HD). [00442] In some embodiments, the one or more symptoms are selected from chorea, cognitive decline, abnormal libido, abnormal eye movement, abnormal sense of smell, aggression, agitation, anxiety, apathy, bradykinesia, bradyphrenia, clumsiness, delusions, depression, difficulty walking, disinhibition, dystonia, gait imbalance, muscle weakness, hallucinations, hostility, hypokinesia, irritability, memory impairment, myoclonus, obsessive-compulsive behavior, poor fine motor coordination, seizure, speech articulation difficulties, staring gaze, weight loss, abnormal cholesterol metabolism, abnormal cerebral white matter, alcoholism, Babinski sign, caudate atrophy, cerebral atrophy, choking, clonus, degeneration of the striatum, excessive daytime sleepiness, impaired visuospatial constructive cognition, inability to walk, insomnia, mutism, oral-pharyngeal dysphagia, rigidity, suicidal ideation, cerebellar atrophy, dementia, gate ataxia, gliosis, hyperreflexia, neuronal loss, or personality changes.
Pharmaceutical Compositions and Administration
[00443] The compounds described herein are administered to a subject in need thereof, either alone or in combination with pharmaceutically acceptable carriers, excipients, or diluents, in a pharmaceutical composition, according to standard pharmaceutical practice. In some embodiments, the compounds described herein are administered to animals.
[00444] In another aspect, provided herein are pharmaceutical compositions comprising a compound described herein, or a pharmaceutically acceptable salt thereof, and at least one pharmaceutically acceptable
Ill
excipient. Pharmaceutical compositions are formulated in a conventional manner using one or more pharmaceutically acceptable excipients that facilitate processing of the active compounds into preparations that can be used pharmaceutically. Proper formulation is dependent upon the route of administration chosen. A summary of pharmaceutical compositions described herein can be found, for example, in Remington: The Science and Practice of Pharmacy, Nineteenth Ed (Easton, Pa.: Mack Publishing Company, 1995); Hoover, John E., Remington’s Pharmaceutical Sciences, Mack Publishing Co., Easton, Pennsylvania 1975;
Liberman, H.A. and Lachman, L., Eds., Pharmaceutical Dosage Forms, Marcel Decker, New York, (N.Y., 1980; and Pharmaceutical Dosage Forms and Drug Delivery Systems, Seventh Ed. (Lippincott Williams & Wilkins 1999), herein incorporated by reference for such disclosure.
[00445] In some embodiments, the pharmaceutically acceptable excipient is selected from carriers, binders, filling agents, suspending agents, flavoring agents, sweetening agents, disintegrating agents, dispersing agents, surfactants, lubricants, colorants, diluents, solubilizers, moistening agents, plasticizers, stabilizers, penetration enhancers, wetting agents, anti-foaming agents, antioxidants, preservatives, and any combinations thereof.
[00446] The dose of a pharmaceutical agent described herein for treating a disease or disorder may depend upon the subject’s condition, that is, stage of the disease, severity of symptoms caused by the disease, general health status, as well as age, gender, and weight, and other factors apparent to a person skilled in the medical art. Pharmaceutical compositions may be administered in a manner appropriate to the disease to be treated as determined by persons skilled in the medical arts. In addition to the factors described herein and above related to use of pharmaceutical agent for treating a disease or disorder, suitable duration and frequency of administration of the pharmaceutical agent may also be determined or adjusted by such factors as the condition of the patient, the type and severity of the patient’s disease, the particular form of the active ingredient, and the method of administration. Optimal doses of an agent may generally be determined using experimental models and/or clinical trials. The optimal dose may depend upon the body mass, weight, or blood volume of the subject. The use of the minimum dose that is sufficient to provide effective therapy is usually preferred. Design and execution of pre-clinical and clinical studies for a pharmaceutical agent, including when administered for prophylactic benefit, described herein are well within the skill of a person skilled in the relevant art. When two or more pharmaceutical agents are administered to treat a disease or disorder, the optimal dose of each pharmaceutical agent may be different, such as less than when either agent is administered alone as a single agent therapy. In certain particular embodiments, two pharmaceutical agents in combination may act synergistically or additively, and either agent may be used in a lesser amount than if administered alone. An amount of a pharmaceutical agent that may be administered per day may be, for example, between about 0.01 mg/kg and 100 mg/kg, e.g., between about 0.1 to 1 mg/kg, between about 1 to 10 mg/kg, between about 10-50 mg/kg, between about 50-100 mg/kg body weight. In other embodiments, the amount of a pharmaceutical agent that may be administered per day is between about 0.01 mg/kg and 1000 mg/kg, between about 100-500 mg/kg, or between about 500-1000 mg/kg body weight. The optimal
dose, per day or per course of treatment, may be different for the disease or disorder to be treated and may also vary with the administrative route and therapeutic regimen.
Definitions
[00447] Unless defined otherwise, all technical and scientific terms used herein have the same meaning as is commonly understood by one of skill in the art to which this invention belongs.
[00448] Unless the context requires otherwise, throughout the specification and claims which follow, the word “comprise” and variations thereof, such as, “comprises” and “comprising” are to be construed in an open, inclusive sense, that is, as “including, but not limited to.” Further, headings provided herein are for convenience only and do not interpret the scope or meaning of the claimed invention.
[00449] As used in this specification and the appended claims, the singular forms “a,” “an,” and “the” include plural referents unless the content clearly dictates otherwise. It should also be noted that the term “or” is generally employed in its sense including “and/or” unless the content clearly dictates otherwise. [00450] When ranges of values are disclosed, and the notation “from ... to n2” or “between . . . and n2” is used, where and n2 are the numbers, then unless otherwise specified, this notation is intended to include the numbers themselves and the range between them. This range may be integral or continuous between and including the end values. By way of example, the range “from 2 to 6 carbons” is intended to include two, three, four, five, and six carbons, since carbons come in integer units. Compare, by way of example, the range “from 1 to 3 pM (micromolar),” which is intended to include 1 pM, 3 pM, and everything in between to any number of significant figures (e.g., 1.255 pM, 2. 1 pM, 2.9999 pM, etc.). [00451] The terms below, as used herein, have the following meanings, unless indicated otherwise: [00452] “oxo” refers to =0.
[00453] “Carboxyl” refers to -COOH.
[00454] “Cyano” refers to -CN.
[00455] “Alkyl” refers to a straight-chain, or branched-chain saturated hydrocarbon monoradical having from one to about ten carbon atoms, more preferably one to six carbon atoms. Examples include, but are not limited to methyl, ethyl, n-propyl, isopropyl, 2-methyl-l -propyl, 2-methyl-2 -propyl, 2-methyl-l -butyl, 3- methyl-1 -butyl, 2-methyl-3 -butyl, 2,2-dimethyl-l -propyl, 2-methyl-l -pentyl, 3 -methyl- 1 -pentyl, 4-methyl-l- pentyl, 2-methyl-2-pentyl, 3 -methyl -2 -pentyl, 4-methyl-2-pentyl, 2,2-dimethyl-l -butyl, 3, 3 -dimethyl- 1- butyl, 2 -ethyl- 1 -butyl, n-butyl, isobutyl, sec-butyl, t-butyl, n-pentyl, isopentyl, neopentyl, tert-amyl and hexyl, and longer alkyl groups, such as heptyl, octyl and the like. Whenever it appears herein, a numerical range such as “Ci-Ce alkyl” or “Ci -ealkyl”, means that the alkyl group may consist of 1 carbon atom, 2 carbon atoms, 3 carbon atoms, 4 carbon atoms, 5 carbon atoms or 6 carbon atoms, although the present definition also covers the occurrence of the term “alkyl” where no numerical range is designated. In some embodiments, the alkyl is a Ci-Cioalkyl. In some embodiments, the alkyl is a Ci-Cealkyl. In some embodiments, the alkyl is a Ci-Csalkyl. In some embodiments, the alkyl is a Ci-C4alkyl. In some embodiments, the alkyl is a Ci-Csalkyl. Unless stated otherwise specifically in the specification, an alkyl
group may be optionally substituted, for example, with oxo, halogen, amino, nitrile, nitro, hydroxyl, haloalkyl, alkoxy, carboxyl, carboxylate, aryl, cycloalkyl, heterocycloalkyl, heteroaryl, and the like. In some embodiments, the alkyl is optionally substituted with oxo, halogen, -Ns, -CN, -C(O)OH, -C(O)OMe, -OH, - OMe, -NH2, or -NO2. In some embodiments, the alkyl is optionally substituted with halogen, -CN, -OH, or - OMe. In some embodiments, the alkyl is optionally substituted with halogen.
[00456] “Alkenyl” refers to a straight-chain, or branched-chain hydrocarbon monoradical having one or more carbon-carbon double-bonds and having from two to about ten carbon atoms, more preferably two to about six carbon atoms. The group may be in either the cis or trans conformation about the double bond(s), and should be understood to include both isomers. Examples include, but are not limited to ethenyl (- CH=CH2), 1-propenyl (-CH2CH=CH2), isopropenyl [-C(CHs)=CH2], butenyl, 1,3-butadienyl and the like. Whenever it appears herein, a numerical range such as “C2-C6 alkenyl” or “C2-6alkenyl”, means that the alkenyl group may consist of 2 carbon atoms, 3 carbon atoms, 4 carbon atoms, 5 carbon atoms or 6 carbon atoms, although the present definition also covers the occurrence of the term “alkenyl” where no numerical range is designated. Unless stated otherwise specifically in the specification, an alkenyl group may be optionally substituted, for example, with oxo, halogen, amino, nitrile, nitro, hydroxyl, haloalkyl, alkoxy, carboxyl, carboxylate, aryl, cycloalkyl, heterocycloalkyl, heteroaryl, and the like. In some embodiments, the alkenyl is optionally substituted with oxo, halogen, -N3, -CN, -C(O)OH, -C(O)OMe, -OH, -OMe, -NH2, or - NO2. In some embodiments, the alkenyl is optionally substituted with halogen, -CN, -OH, or -OMe. In some embodiments, the alkenyl is optionally substituted with halogen.
[00457] “Alkynyl” refers to a straight-chain or branched-chain hydrocarbon monoradical having one or more carbon-carbon triple-bonds and having from two to about ten carbon atoms, more preferably from two to about six carbon atoms. Examples include, but are not limited to ethynyl, 2-propynyl, 2-butynyl, 1,3- butadiynyl and the like. Whenever it appears herein, a numerical range such as “C2-C6 alkynyl” or “C2- ealkynyl”, means that the alkynyl group may consist of 2 carbon atoms, 3 carbon atoms, 4 carbon atoms, 5 carbon atoms or 6 carbon atoms, although the present definition also covers the occurrence of the term “alkynyl” where no numerical range is designated. Unless stated otherwise specifically in the specification, an alkynyl group may be optionally substituted, for example, with oxo, halogen, amino, nitrile, nitro, hydroxyl, haloalkyl, alkoxy, carboxyl, carboxylate, aryl, cycloalkyl, heterocycloalkyl, heteroaryl, and the like. In some embodiments, the alkynyl is optionally substituted with oxo, halogen, -N3, -CN, -C(O)OH, C(O)OMe, -OH, -OMe, -NH2, or -NO2. In some embodiments, the alkynyl is optionally substituted with halogen, -CN, -OH, or -OMe. In some embodiments, the alkynyl is optionally substituted with halogen. [00458] “Alkylene” refers to a straight or branched divalent hydrocarbon chain. Unless stated otherwise specifically in the specification, an alkylene group may be optionally substituted, for example, with oxo, halogen, amino, nitrile, nitro, hydroxyl, haloalkyl, alkoxy, carboxyl, carboxylate, aryl, cycloalkyl, heterocycloalkyl, heteroaryl, and the like. In some embodiments, the alkylene is optionally substituted with oxo, halogen, -N3, -CN, -C(O)OH, C(O)OMe, -OH, -OMe, -NH2, or -NO2. In some embodiments, the
alkylene is optionally substituted with halogen, -CN, -OH, or -OMe. In some embodiments, the alkylene is optionally substituted with halogen.
[00459] “Alkoxy” refers to a radical of the formula -ORa where Ra is an alkyl radical as defined. Unless stated otherwise specifically in the specification, an alkoxy group may be optionally substituted, for example, with oxo, halogen, amino, nitrile, nitro, hydroxyl, haloalkyl, alkoxy, carboxyl, carboxylate, aryl, cycloalkyl, heterocycloalkyl, heteroaryl, and the like. In some embodiments, the alkoxy is optionally substituted with halogen, -Ns, -CN, -C(O)OH, C(O)OMe, -OH, -OMe, -NH2, or -NO2. In some embodiments, the alkoxy is optionally substituted with halogen, -CN, -OH, or -OMe. In some embodiments, the alkoxy is optionally substituted with halogen.
[00460] "Aryl" refers to a radical derived from an aromatic monocyclic or aromatic multicyclic hydrocarbon ring system by removing a hydrogen atom from a ring carbon atom. The aromatic monocyclic or aromatic multicyclic hydrocarbon ring system can contain only hydrogen and carbon and from five to eighteen carbon atoms, where at least one of the rings in the ring system is aromatic, i.e., it contains a cyclic, delocalized (4n+2) K-cIcctron system in accordance with the Htickel theory. The ring system from which aryl groups are derived include, but are not limited to, groups such as benzene, fluorene, indane, indene, tetralin and naphthalene. The aryl radical may be a monocyclic, bicyclic, tricyclic, or tetracyclic ring system, which may include fused (when fused with a cycloalkyl or heterocycloalkyl ring, the aryl is bonded through an aromatic ring atom) or bridged ring systems. In some embodiments, the aryl is a 6- to 10-membered aryl. In some embodiments, the aryl is a 6-membered aryl (phenyl). Aryl radicals include, but are not limited to, aryl radicals derived from the hydrocarbon ring systems of anthrylene, naphthylene, phenanthrylene, anthracene, azulene, benzene, chrysene, fluoranthene, fluorene, as-indacene, s-indacene, indane, indene, naphthalene, phenalene, phenanthrene, pleiadene, pyrene, and triphenylene. Unless stated otherwise specifically in the specification, an aryl may be optionally substituted, for example, with halogen, amino, nitrile, nitro, hydroxyl, alkyl, alkenyl, alkynyl, haloalkyl, alkoxy, carboxyl, carboxylate, aryl, cycloalkyl, heterocycloalkyl, heteroaryl, and the like. In some embodiments, the aryl is optionally substituted with halogen, methyl, ethyl, -N3, -CN, -C(O)OH, C(O)OMe, -CF3, -OH, -OMe, -NH2, or -NO2. In some embodiments, the aryl is optionally substituted with halogen, methyl, ethyl, -CN, -CF3, -OH, or -OMe. In some embodiments, the aryl is optionally substituted with halogen.
[00461] “Cycloalkyl” refers to a partially or fully saturated, monocyclic, or polycyclic carbocyclic ring, which may include fused (when fused with an aryl or a heteroaryl ring, the cycloalkyl is bonded through a non-aromatic ring atom), spiro, or bridged ring systems. In some embodiments, the cycloalkyl is fully saturated. Representative cycloalkyls include, but are not limited to, cycloalkyls having from three to fifteen carbon atoms (e.g., C3-Cis fully saturated cycloalkyl or C3-Cis cycloalkenyl), from three to ten carbon atoms (e.g., C3-Cio fully saturated cycloalkyl or C3-Cio cycloalkenyl), from three to eight carbon atoms (e.g., C3-Cs fully saturated cycloalkyl or C3-Cs cycloalkenyl), from three to six carbon atoms (e.g., C3-Ce fully saturated cycloalkyl or C3-Ce cycloalkenyl), from three to five carbon atoms (e.g., C3-C3 fully saturated cycloalkyl or C3-C3 cycloalkenyl), or three to four carbon atoms (e.g., C3-C4 fully saturated cycloalkyl or C3-C4
cycloalkenyl). In some embodiments, the cycloalkyl is a 3- to 10-membered fully saturated cycloalkyl or a 3- to 10-membered cycloalkenyl. In some embodiments, the cycloalkyl is a 3 - to 6-membered fully saturated cycloalkyl or a 3- to 6-membered cycloalkenyl. In some embodiments, the cycloalkyl is a 5 - to 6-membered fully saturated cycloalkyl or a 5- to 6-membered cycloalkenyl. Monocyclic cycloalkyls include, for example, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, cycloheptyl, and cyclooctyl. Polycyclic cycloalkyls include, for example, adamantyl, norbomyl, decalinyl, bicyclo[3.3.0]octane, bicyclo[4.3.0]nonane, cisdecalin, trans-decalin, bicyclo [2.1.1] hexane, bicyclo[2.2.1]heptane, bicyclo[2.2.2]octane, bicyclo[3.2.2]nonane, and bicyclo[3.3.2]decane, and 7,7-dimethyl-bicyclo[2.2.1]heptanyl. Partially saturated cycloalkyls include, for example cyclopentenyl, cyclohexenyl, cycloheptenyl, and cyclooctenyl. Unless stated otherwise specifically in the specification, a cycloalkyl is optionally substituted, for example, with oxo, halogen, amino, nitrile, nitro, hydroxyl, alkyl, alkenyl, alkynyl, haloalkyl, alkoxy, carboxyl, carboxylate, aryl, cycloalkyl, heterocycloalkyl, heteroaryl, and the like. In some embodiments, a cycloalkyl is optionally substituted with oxo, halogen, methyl, ethyl, -Ns, -CN, -C(0)0H, C(0)0Me, -CF3, -OH, -OMe, -NH2, or -N02. In some embodiments, a cycloalkyl is optionally substituted with oxo, halogen, methyl, ethyl, -CN, -CF3, -OH, or -OMe. In some embodiments, the cycloalkyl is optionally substituted with halogen.
[00462] "Cycloalkenyl" refers to an unsaturated non-aromatic monocyclic or polycyclic hydrocarbon radical consisting solely of carbon and hydrogen atoms, which includes fused or bridged ring systems, preferably having from three to twelve carbon atoms and comprising at least one double bond. In certain embodiments, a cycloalkenyl comprises three to ten carbon atoms. In other embodiments, a cycloalkenyl comprises five to seven carbon atoms. The cycloalkenyl may be attached to the rest of the molecule by a single bond. Examples of monocyclic cycloalkenyls includes, e.g., cyclopentenyl, cyclohexenyl, cycloheptenyl, and cyclooctenyl.
[00463] “Halo” or “halogen” refers to bromo, chloro, fluoro or iodo. In some embodiments, halogen is fluoro or chloro. In some embodiments, halogen is fluoro.
[00464] As used herein, the term "haloalkyl" or “haloalkane” refers to an alkyl radical, as defined above, that is substituted by one or more halogen radicals, for example, trifluoromethyl, dichloromethyl, bromomethyl, 2,2,2-trifluoroethyl, 1 -fluoromethyl -2 -fluoroethyl, and the like. In some embodiments, the alkyl part of the fluoroalkyl radical is optionally further substituted. Examples of halogen substituted alkanes (“haloalkanes”) include halomethane (e.g., chloromethane, bromomethane, fluoromethane, iodomethane), di-and trihalomethane (e.g., trichloromethane, tribromomethane, trifluoromethane, triiodomethane), 1- haloethane, 2-haloethane, 1,2-dihaloethane, 1-halopropane, 2-halopropane, 3-halopropane, 1,2- dihalopropane, 1,3-dihalopropane, 2,3-dihalopropane, 1,2,3-trihalopropane, and any other suitable combinations of alkanes (or substituted alkanes) and halogens (e.g., Cl, Br, F, I, etc.). When an alkyl group is substituted with more than one halogen radicals, each halogen may be independently selected e.g., 1- chloro,2-fluoroethane .
[00465] "Fluoroalkyl" refers to an alkyl radical, as defined above, that is substituted by one or more fluoro radicals, for example, trifluoromethyl, difluoromethyl, fluoromethyl, 2,2,2-trifluoroethyl, l-fluoromethyl-2-fluoroethyl, and the like.
[00466] “Hydroxyalkyl” refers to an alkyl radical, as defined above, that is substituted by one or more hydroxyls. In some embodiments, the alkyl is substituted with one hydroxyl. In some embodiments, the alkyl is substituted with one, two, or three hydroxyls. Hydroxyalkyl include, for example, hydroxymethyl, hydroxyethyl, hydroxypropyl, hydroxybutyl, or hydroxypentyl. In some embodiments, the hydroxyalkyl is hydroxymethyl.
[00467] “Aminoalkyl” refers to an alkyl radical, as defined above, that is substituted by one or more amines. In some embodiments, the alkyl is substituted with one amine. In some embodiments, the alkyl is substituted with one, two, or three amines. Aminoalkyl include, for example, aminomethyl, aminoethyl, aminopropyl, aminobutyl, or aminopentyl. In some embodiments, the aminoalkyl is aminomethyl.
[00468] “Heteroalkyl” refers to an alkyl group in which one or more skeletal atoms of the alkyl are selected from an atom other than carbon, e.g., oxygen, nitrogen (e.g., -NH-, -N(alkyl)-), sulfur, phosphorus, or combinations thereof. A heteroalkyl is attached to the rest of the molecule at a carbon atom of the heteroalkyl. In one aspect, a heteroalkyl is a Ci-Ce heteroalkyl wherein the heteroalkyl is comprised of 1 to 6 carbon atoms and one or more atoms other than carbon, e.g., oxygen, nitrogen (e.g., -NH-, -N(alkyl)-), sulfur, phosphorus, or combinations thereof wherein the heteroalkyl is attached to the rest of the molecule at a carbon atom of the heteroalkyl. Examples of such heteroalkyl are, for example, -CH2OCH3, - CH2CH2OCH3, -CH2CH2OCH2CH2OCH3, -CH(CH3)OCH3, -CH2NHCH3, -CH2N(CH3)2, -CH2CH2NHCH3, or -CH2CH2N(CH3)2. Unless stated otherwise specifically in the specification, a heteroalkyl is optionally substituted for example, with oxo, halogen, amino, nitrile, nitro, hydroxyl, alkyl, alkenyl, alkynyl, haloalkyl, alkoxy, aryl, cycloalkyl, heterocycloalkyl, heteroaryl, and the like. In some embodiments, a heteroalkyl is optionally substituted with oxo, halogen, methyl, ethyl, -CN, -CF3, -OH, -OMe, -NH2, or -NO2. In some embodiments, a heteroalkyl is optionally substituted with oxo, halogen, methyl, ethyl, -CN, -CF3, -OH, or - OMe. In some embodiments, the heteroalkyl is optionally substituted with halogen.
[00469] “Heterocycloalkyl” refers to a 3- to 24-membered partially or fully saturated ring radical comprising 2 to 23 carbon atoms and from one to 8 heteroatoms selected from the group consisting of nitrogen, oxygen, phosphorous, silicon, and sulfur. In some embodiments, the heterocycloalkyl is fully saturated. In some embodiments, the heterocycloalkyl comprises one to three heteroatoms selected from the group consisting of nitrogen, oxygen, and sulfur. In some embodiments, the heterocycloalkyl comprises one to three heteroatoms selected from the group consisting of nitrogen and oxygen. In some embodiments, the heterocycloalkyl comprises one to three nitrogens. In some embodiments, the heterocycloalkyl comprises one or two nitrogens. In some embodiments, the heterocycloalkyl comprises one nitrogen. In some embodiments, the heterocycloalkyl comprises one nitrogen and one oxygen. Unless stated otherwise specifically in the specification, the heterocycloalkyl radical may be a monocyclic, bicyclic, tricyclic, or tetracyclic ring system, which may include fused (when fused with an aryl or a heteroaryl ring, the
heterocycloalkyl is bonded through a non-aromatic ring atom), spiro, or bridged ring systems; and the nitrogen, carbon, or sulfur atoms in the heterocycloalkyl radical may be optionally oxidized; the nitrogen atom may be optionally quatemized. Representative heterocycloalkyls include, but are not limited to, heterocycloalkyls having from two to fifteen carbon atoms (e.g., C2-C15 fully saturated heterocycloalkyl or C2-C15 heterocycloalkenyl), from two to ten carbon atoms (e.g., C2-C10 fully saturated heterocycloalkyl or C2-C10 heterocycloalkenyl), from two to eight carbon atoms (e.g., C2-C8 fully saturated heterocycloalkyl or C2-C8 heterocycloalkenyl), from two to seven carbon atoms (e.g., C2-C7 fully saturated heterocycloalkyl or C2-C7 heterocycloalkenyl), from two to six carbon atoms (e.g., C2-C6 fully saturated heterocycloalkyl or C2- C, heterocycloalkenyl), from two to five carbon atoms (e.g., C2-C5 fully saturated heterocycloalkyl or C2-C5 heterocycloalkenyl), or two to four carbon atoms (e.g., C2-C4 fully saturated heterocycloalkyl or C2-C4 heterocycloalkenyl). Examples of such heterocycloalkyl radicals include, but are not limited to, aziridinyl, azetidinyl, oxetanyl, dioxolanyl, thienyl[l,3]dithianyl, decahydroisoquinolyl, imidazolinyl, imidazolidinyl, isothiazolidinyl, isoxazolidinyl, morpholinyl, octahydroindolyl, octahydroisoindolyl, 2-oxopiperazinyl, 2- oxopiperidinyl, 2-oxopyrrolidinyl, oxazolidinyl, piperidinyl, piperazinyl, 4-piperidonyl, pyrrolidinyl, pyrazolidinyl, quinuclidinyl, thiazolidinyl, tetrahydrofuryl, trithianyl, tetrahydropyranyl, thiomorpholinyl, thiamorpholinyl, 1-oxo-thiomorpholinyl, 1,1-dioxo-thiomorpholinyl, 1,3-dihydroisobenzofuran-l-yl, 3-oxo- 1,3-dihydroisobenzofuran-l-yl, methyl-2-oxo-l,3-dioxol-4-yl, and 2-oxo-l,3-dioxol-4-yl. The term heterocycloalkyl also includes all ring forms of the carbohydrates, including but not limited to the monosaccharides, the disaccharides, and the oligosaccharides. In some embodiments, heterocycloalkyls have from 2 to 10 carbons in the ring. It is understood that when referring to the number of carbon atoms in a heterocycloalkyl, the number of carbon atoms in the heterocycloalkyl is not the same as the total number of atoms (including the heteroatoms) that make up the heterocycloalkyl (i.e. skeletal atoms of the heterocycloalkyl ring). In some embodiments, the heterocycloalkyl is a 3- to 8-membered fully saturated heterocycloalkyl. In some embodiments, the heterocycloalkyl is a 3 - to 7-membered fully saturated heterocycloalkyl. In some embodiments, the heterocycloalkyl is a 3 - to 6-membered fully saturated heterocycloalkyl. In some embodiments, the heterocycloalkyl is a 4- to 6-membered fully saturated heterocycloalkyl. In some embodiments, the heterocycloalkyl is a 5 - to 6-membered fully saturated heterocycloalkyl. In some embodiments, the heterocycloalkyl is a 3- to 8-membered heterocycloalkenyl. In some embodiments, the heterocycloalkyl is a 3- to 7-membered heterocycloalkenyl. In some embodiments, the heterocycloalkyl is a 3- to 6-membered heterocycloalkenyl. In some embodiments, the heterocycloalkyl is a 4- to 6-membered heterocycloalkenyl. In some embodiments, the heterocycloalkyl is a 5 - to 6-membered heterocycloalkenyl. Unless stated otherwise specifically in the specification, a heterocycloalkyl may be optionally substituted as described below, for example, with oxo, halogen, amino, nitrile, nitro, hydroxyl, alkyl, alkenyl, alkynyl, haloalkyl, alkoxy, carboxyl, carboxylate, aryl, cycloalkyl, heterocycloalkyl, heteroaryl, and the like. In some embodiments, the heterocycloalkyl is optionally substituted with oxo, halogen, methyl, ethyl, -CN, -C(O)OH, C(O)OMe, -CF3, -OH, -OMe, -NH2, or -NO2. In some embodiments,
the heterocycloalkyl is optionally substituted with halogen, methyl, ethyl, -CN, -CF3, -OH, or -OMe. In some embodiments, the heterocycloalkyl is optionally substituted with halogen.
[00470] “Heteroaryl” refers to a 5- to 14-membered ring system radical comprising one to thirteen carbon atoms, one to six heteroatoms selected from the group consisting of nitrogen, oxygen, phosphorous, and sulfur, and at least one aromatic ring. In some embodiments, the heteroaryl comprises one to three heteroatoms selected from the group consisting of nitrogen, oxygen, and sulfur. In some embodiments, the heteroaryl comprises one to three heteroatoms selected from the group consisting of nitrogen and oxygen. In some embodiments, the heteroaryl comprises one to three nitrogens. In some embodiments, the heteroaryl comprises one or two nitrogens. In some embodiments, the heteroaryl comprises one nitrogen. The heteroaryl radical may be a monocyclic, bicyclic, tricyclic, or tetracyclic ring system, which may include fused (when fused with a cycloalkyl or heterocycloalkyl ring, the heteroaryl is bonded through an aromatic ring atom) or bridged ring systems; and the nitrogen, carbon, or sulfur atoms in the heteroaryl radical may be optionally oxidized; the nitrogen atom may be optionally quatemized. In some embodiments, the heteroaryl is a 5 - to 10-membered heteroaryl. In some embodiments, the heteroaryl is a 5 - to 6-membered heteroaryl. In some embodiments, the heteroaryl is a 6-membered heteroaryl. In some embodiments, the heteroaryl is a 5- membered heteroaryl. Examples include, but are not limited to, azepinyl, acridinyl, benzimidazolyl, benzothiazolyl, benzindolyl, benzodioxolyl, benzofuranyl, benzooxazolyl, benzothiazolyl, benzothiadiazolyl, benzo[b][l,4]dioxepinyl, 1,4-benzodioxanyl, benzonaphthofuranyl, benzoxazolyl, benzodioxolyl, benzodioxinyl, benzopyranyl, benzopyranonyl, benzofuranyl, benzofuranonyl, benzothienyl (benzothiophenyl), benzotriazolyl, benzo[4,6]imidazo[l,2-a]pyridinyl, carbazolyl, cinnolinyl, dibenzofuranyl, dibenzothiophenyl, furanyl, furanonyl, isothiazolyl, imidazolyl, indazolyl, indolyl, isoindolyl, indolinyl, isoindolinyl, isoquinolyl, indolizinyl, isoxazolyl, naphthyridinyl, oxadiazolyl, 2- oxoazepinyl, oxazolyl, oxiranyl, 1-oxidopyridinyl, 1-oxidopyrimidinyl, 1-oxidopyrazinyl, 1- oxidopyridazinyl, 1 -phenyl- IH-pyrrolyl, phenazinyl, phenothiazinyl, phenoxazinyl, phthalazinyl, pteridinyl, purinyl, pyrrolyl, pyrazolyl, pyridinyl, pyrazinyl, pyrimidinyl, pyridazinyl, quinazolinyl, quinoxalinyl, quinolinyl, quinuclidinyl, isoquinolinyl, tetrahydroquinolinyl, thiazolyl, thiadiazolyl, triazolyl, tetrazolyl, triazinyl, and thiophenyl (i.e., thienyl). Unless stated otherwise specifically in the specification, a heteroaryl may be optionally substituted, for example, with halogen, amino, nitrile, nitro, hydroxyl, alkyl, alkenyl, alkynyl, haloalkyl, alkoxy, carboxyl, carboxylate, aryl, cycloalkyl, heterocycloalkyl, heteroaryl, and the like. In some embodiments, the heteroaryl is optionally substituted with halogen, methyl, ethyl, -CN, -C(O)OH, C(O)OMe, -CF3, -OH, -OMe, -NH2, or -NO2. In some embodiments, the heteroaryl is optionally substituted with halogen, methyl, ethyl, -CN, -CF3, -OH, or -OMe. In some embodiments, the heteroaryl is optionally substituted with halogen.
[00471] The term “oligonucleotide sequence” refers to a plurality of nucleic acids having a defined sequence and length (e.g., 2, 3, 4, 5, 6, or even more nucleotides). The term “oligonucleotide repeat sequence” refers to a contiguous expansion of oligonucleotide sequences.
[00472] The term “transcription,” well known in the art, refers to the synthesis of RNA (i.e., ribonucleic acid) by DNA-directed RNA polymerase. The term “modulate transcription” refers to a change in transcriptional level which can be measured by methods well known in the art, for example, assay of mRNA, the product of transcription. In certain embodiments, modulation is an increase in transcription. In other embodiments, modulation is a decrease in transcription.
[00473] The term “polyamide” refers to polymers of linkable units chemically bound by amide (i.e., CONH) linkages; optionally, polyamides include chemical probes conjugated therewith. Polyamides may be synthesized by stepwise condensation of carboxylic acids (COOH) with amines (RR’NH) using methods known in the art. Alternatively, polyamides may be formed using enzymatic reactions in vitro, or by employing fermentation with microorganisms.
[00474] The term “linkable unit” refers to methylimidazoles, methylpyrroles, and straight and branched chain aliphatic functionalities (e.g., methylene, ethylene, propylene, butylene, and the like) which optionally contain nitrogen Substituents, and chemical derivatives thereof. The aliphatic functionalities of linkable units can be provided, for example, by condensation of B-alanine or dimethylaminopropylamine during synthesis of the polyamide by methods well known in the art.
[00475] The term “linker” or “oligomeric backbone” refers to a chain of at least 10 contiguous atoms. In certain embodiments, the linker contains no more than 20 non-hydrogen atoms. The terms linker and oligomeric backbone can be used interchangeably. In some embodiments, the linker contains no more than 40 non-hydrogen atoms. In some embodiments, the linker contains no more than 60 non-hydrogen atoms. In certain embodiments, the linker contains atoms chosen from C, H, N, O, and S. In some embodiments, every non-hydrogen atom is chemically bonded either to 2 neighboring atoms in the linker, or one neighboring atom in the linker and a terminus of the linker. In some embodiments, the linker forms an amide bond with at least one of the two other groups to which it is attached. In certain embodiments, the linker forms an ester or ether bond with at least one of the two other groups to which it is attached. In some embodiments, the linker forms a thioester or thioether bond with at least one of the two other groups to which it is attached. In some embodiments, the linker forms a direct carbon-carbon bond with at least one of the two other groups to which it is attached. In some embodiments, the linker forms an amine or amide bond with at least one of the two other groups to which it is attached. In some embodiments, the linker comprises -(CH2OCH2)- units. In some embodiments, the linker comprises -(CH(CH3)OCH2)- units. In some embodiments, the linker comprises -(CH2NRNCH2) units, for RN = Ci-4alkyl. In some embodiments, the linker comprises an arylene, cycloalkylene, or heterocycloalkylene moiety.
[00476] The term “bond” refers to a covalent linkage between two atoms, or two moieties when the atoms joined by the bond are considered to be part of larger substructure. A bond may be single, double, or triple unless otherwise specified. A dashed line between two atoms in a drawing of a molecule indicates that an additional bond may be present or absent at that position.
[00477] As used herein, “optionally substituted” is a substituted group is derived from the unsubstituted parent group in which there has been an exchange of one or more hydrogen atoms for another atom or
group. Unless otherwise indicated, when a group is deemed to be “substituted” or “optionally substituted” it is meant that the group is substituted with one or more substituents independently selected from Ci-Ce alkyl, Ci-Ce alkenyl, Ci-Ce alkynyl, Ci-Ce heteroalkyl, C3-C7 carbocyclyl (optionally substituted with halo, Ci-Ce alkyl, Ci-Ce alkoxy, Ci-Ce haloalkyl, and Ci-Ce haloalkoxy), Cs-Cv-carbocyclyl-Ci-Ce-alkyl (optionally substituted with halo, Ci-Ce alkyl, Ci-Ce alkoxy, Ci-Ce haloalkyl, and Ci-Ce haloalkoxy), 3-10 membered heterocyclyl (optionally substituted with halo, Ci-Ce alkyl, Ci-Ce alkoxy, Ci-Ce haloalkyl, and Ci-Ce haloalkoxy), 3-10 membered heterocyclyl-Ci-Ce-alkyl (optionally substituted with halo, Ci-Ce alkyl, Ci-Ce alkoxy, Ci-Ce haloalkyl, and Ci-Ce haloalkoxy), aryl (optionally substituted with halo, Ci-Ce alkyl, Ci-Ce alkoxy, Ci-Ce haloalkyl, and Ci-Ce haloalkoxy), aryl(Ci-Ce)alkyl (optionally substituted with halo, Ci-Ce alkyl, Ci-Cg alkoxy, Ci-Cg haloalkyl, and Ci-Cg haloalkoxy), 5-10 membered heteroaryl (optionally substituted with halo, Ci-Ce alkyl, Ci-Ce alkoxy, Ci-Ce haloalkyl, and Ci-Ce haloalkoxy), 5-10 membered heteroaryl(Ci-Ce)alkyl (optionally substituted with halo, Ci-Ce alkyl, Ci-Ce alkoxy, Ci-Ce haloalkyl, and Ci- Ce haloalkoxy), halo, cyano, hydroxy, Ci-Cg alkoxy, Ci-Cg alkoxy(Ci-Ce)alkyl (i.e., ether), aryloxy, sulfhydryl (mercapto), halo(Ci-Ce)alkyl (e.g., -CF3), halo(Ci-Ce)alkoxy (e.g., -OCF3), Ci-Ce alkylthio, arylthio, amino, amino(Ci-Ce)alkyl, nitro, O-carbamyl, N-carbamyl, O-thiocarbamyl, N-thiocarbamyl, C- amido, N-amido, S-sulfonamido, N-sulfonamido, C-carboxy, O-carboxy, acyl, cyanato, isocyanato, thiocyanate, isothiocyanate, sulfinyl, sulfonyl, and oxo (=0). Wherever a group is described as “optionally substituted” that group can be substituted with the above substituents.
[00478] The term “one or more” when referring to an optional substituent means that the subject group is optionally substituted with one, two, three, or four substituents. In some embodiments, the subject group is optionally substituted with one, two, or three substituents. In some embodiments, the subject group is optionally substituted with one or two substituents. In some embodiments, the subject group is optionally substituted with one substituent. In some embodiments, the subject group is optionally substituted with two substituents.
[00479] Chemical entities having carbon-carbon double bonds or carbon-nitrogen double bonds may exist in Z- or E- form (or cis- or trans- form). Furthermore, some chemical entities may exist in various tautomeric forms. Unless otherwise specified, compounds described herein are intended to include all Z-, E- and tautomeric forms as well.
[00480] The compounds disclosed herein, in some embodiments, are used in different enriched isotopic forms, e.g., enriched in the content of 2H, 3H, nC, 13C and/or 14C. In one particular embodiment, the compound is deuterated in at least one position. Such deuterated forms can be made by the procedure described in U.S. Patent Nos. 5,846,514 and 6,334,997. As described in U.S. Patent Nos. 5,846,514 and 6,334,997, deuteration can improve the metabolic stability and or efficacy, thus increasing the duration of action of drugs.
[00481] Unless otherwise stated, compounds described herein are intended to include compounds which differ only in the presence of one or more isotopically enriched atoms. For example, compounds having the
present structures except for the replacement of a hydrogen by a deuterium or tritium, or the replacement of a carbon by 13C- or 14C-enriched carbon are within the scope of the present disclosure.
[00482] The compounds of the present disclosure optionally contain unnatural proportions of atomic isotopes at one or more atoms that constitute such compounds. For example, the compounds may be labeled with isotopes, such as for example, deuterium (2H), tritium (3H), iodine-125 (125I) or carbon-14 (14C).
Isotopic substitution with 2H, nC, 13C, 14C, 15C, 12N, 13N, 15N, 16N, 160, 170, 14F, 15F, 16F, 17F, 18F, 33S, 34S, 35S, 36S, 35C1, 37C1, 79Br, 81Br, and 125I are all contemplated. All isotopic variations of the compounds of the present invention, whether radioactive or not, are encompassed within the scope of the present invention. In some embodiments, where isotopic variations are illustrated, the remaining atoms of the compound may optionally contain unnatural portions of atomic isotopes.
[00483] In certain embodiments, the compounds disclosed herein have some or all of the ’H atoms replaced with 2H atoms. The methods of synthesis for deuterium-containing compounds are known in the art and include, by way of non-limiting example only, the following synthetic methods.
[00484] Deuterium substituted compounds are synthesized using various methods such as described in: Dean, Dennis C.; Editor. Recent Advances in the Synthesis and Applications of Radiolabeled Compounds for Drug Discovery and Development. [In: Curr., Pharm. Des., 2000; 6(10)] 2000, 110 pp; George W.; Varma, Rajender S. The Synthesis of Radiolabeled Compounds via Organometallic Intermediates, Tetrahedron, 1989, 45(21), 6601-21; and Evans, E. Anthony. Synthesis of radiolabeled compounds, J. Radioanal. Chem., 1981, 64(1-2), 9-32.
[00485] Deuterated starting materials are readily available and are subjected to the synthetic methods described herein to provide for the synthesis of deuterium-containing compounds. Large numbers of deuterium-containing reagents and building blocks are available commercially from chemical vendors, such as Aldrich Chemical Co.
[00486] In some embodiments of a compound disclosed herein, one or more of the substituent groups comprise deuterium at a percentage higher than the natural abundance of deuterium. In some embodiments of a compound disclosed herein, one or more hydrogens are replaced with one or more deuteriums.
[00487] In some embodiments of a compound disclosed herein, the abundance of deuterium in each of the substituents is independently at least 1%, at least 10%, at least 20%, at least 30%, at least 40%, at least 50%, at least 60%, at least 70%, at least 80%, at least 90%, or 100% of a total number of hydrogen and deuterium. [00488] Compounds of the present disclosure also include crystalline and amorphous forms of those compounds, pharmaceutically acceptable salts, and active metabolites of these compounds having the same type of activity, including, for example, polymorphs, pseudopolymorphs, solvates, hydrates, unsolvated polymorphs (including anhydrates), conformational polymorphs, and amorphous forms of the compounds, as well as mixtures thereof.
[00489] The compounds described herein may in some cases exist as diastereomers, enantiomers, or other stereoisomeric forms. Where absolute stereochemistry is not specified, the compounds presented herein include all diastereomeric, enantiomeric, and epimeric forms as well as the appropriate mixtures thereof.
Separation of stereoisomers may be performed by chromatography or by forming diastereomers and separating by recrystallization, or chromatography, or any combination thereof. (Jean Jacques, Andre Collet, Samuel H. Wilen, “Enantiomers, Racemates and Resolutions”, John Wiley And Sons, Inc., 1981, herein incorporated by reference for this disclosure). Stereoisomers may also be obtained by stereoselective synthesis.
[00490] The term “salt” or “pharmaceutically acceptable salt” refers to salts derived from a variety of organic and inorganic counter ions well known in the art. Pharmaceutically acceptable acid addition salts can be formed with inorganic acids and organic acids. Inorganic acids from which salts can be derived include, for example, hydrochloric acid, hydrobromic acid, sulfuric acid, nitric acid, phosphoric acid, and the like. Organic acids from which salts can be derived include, for example, acetic acid, propionic acid, glycolic acid, pyruvic acid, oxalic acid, maleic acid, malonic acid, succinic acid, fumaric acid, tartaric acid, citric acid, benzoic acid, cinnamic acid, mandelic acid, methanesulfonic acid, ethanesulfonic acid, p- toluene sulfonic acid, salicylic acid, and the like. Pharmaceutically acceptable base addition salts can be formed with inorganic and organic bases. Inorganic bases from which salts can be derived include, for example, sodium, potassium, lithium, ammonium, calcium, magnesium, iron, zinc, copper, manganese, aluminum, and the like. Organic bases from which salts can be derived include, for example, primary, secondary, and tertiary amines, substituted amines including naturally occurring substituted amines, cyclic amines, basic ion exchange resins, and the like, specifically such as isopropylamine, trimethylamine, diethylamine, triethylamine, tripropylamine, and ethanolamine. In some embodiments, the pharmaceutically acceptable base addition salt is chosen from ammonium, potassium, sodium, calcium, and magnesium salts. [00491] The phrase “pharmaceutically acceptable” is employed herein to refer to those compounds, materials, compositions, and/or dosage forms which are, within the scope of sound medical judgment, suitable for use in contact with the tissues of human beings and animals without excessive toxicity, irritation, allergic response, or other problem or complication, commensurate with a reasonable benefit/risk ratio.
[00492] The phrase “pharmaceutically acceptable excipient” or “pharmaceutically acceptable carrier” as used herein means a pharmaceutically acceptable material, composition, or vehicle, such as a liquid or solid filler, diluent, excipient, solvent, or encapsulating material. Each carrier must be “acceptable” in the sense of being compatible with the other ingredients of the formulation and not injurious to the patient. Some examples of materials which can serve as pharmaceutically acceptable carriers include: (1) sugars, such as lactose, glucose and sucrose; (2) starches, such as com starch and potato starch; (3) cellulose, and its derivatives, such as sodium carboxymethyl cellulose, ethyl cellulose and cellulose acetate; (4) powdered tragacanth; (5) malt; (6) gelatin; (7) talc; (8) excipients, such as cocoa butter and suppository waxes; (9) oils, such as peanut oil, cottonseed oil, safflower oil, sesame oil, olive oil, com oil and soybean oil; (10) glycols, such as propylene glycol; (11) polyols, such as glycerin, sorbitol, mannitol and polyethylene glycol; (12) esters, such as ethyl oleate and ethyl laurate; (13) agar; (14) buffering agents, such as magnesium hydroxide and aluminum hydroxide; (15) alginic acid; (16) pyrogen-free water; (17) isotonic saline; (18) Ringer's
solution; (19) ethyl alcohol; (20) phosphate buffer solutions; and (21) other non-toxic compatible substances employed in pharmaceutical formulations.
[00493] An “effective amount” or “therapeutically effective amount” refers to an amount of a compound administered to a mammalian subject, either as a single dose or as part of a series of doses, which is effective to produce a desired therapeutic effect.
[00494] The terms “treat,” “treating” or “treatment,” as used herein, include alleviating, abating, or ameliorating at least one symptom of a disease or condition, preventing additional symptoms, inhibiting the disease or condition, e.g., arresting the development of the disease or condition, relieving the disease or condition, causing regression of the disease or condition, relieving a condition caused by the disease or condition, or stopping the symptoms of the disease or condition.
[00495] The term “patient” is generally synonymous with the term “subject” and includes all mammals including humans. Examples of patients include humans, livestock such as cows, goats, sheep, pigs, and rabbits, and companion animals such as dogs, cats, rabbits, and horses. Preferably, the patient is a human. [00496] The term “contacting” refers to bringing the compound (e.g., a transcription molecular molecule of the present disclosure) into proximity of the desired target gene. The contacting may result in the binding to or result in a conformational change of the target moiety.
[00497] The methods and compositions described herein include the use of amorphous forms as well as crystalline forms (also known as polymorphs). The compounds described herein may be in the form of pharmaceutically acceptable salts. As well, in some embodiments, active metabolites of these compounds having the same type of activity are included in the scope of the present disclosure. In addition, the compounds described herein can exist in unsolvated as well as solvated forms with pharmaceutically acceptable solvents such as water, ethanol, and the like. The solvated forms of the compounds presented herein are also considered to be disclosed herein.
EXAMPLES
[00498] The following examples are given for the purpose of illustrating various embodiments of the invention and are not meant to limit the present invention in any fashion. The present examples, along with the methods described herein are presently representative of preferred embodiments, are exemplary, and are not intended as limitations on the scope of the invention. Changes therein and other uses which are encompassed within the spirit of the invention as defined by the scope of the claims will be known to those skilled in the art.
Compound Synthesis
[00499] Compounds of the present disclosure can be prepared using methods illustrated in general synthetic schemes and experimental procedures detailed below. General synthetic schemes and experimental procedures are presented for purposes of illustration and are not intended to be limiting. Starting materials used to prepare compounds of the present disclosure are commercially available or can be prepared using routine methods known in the art.
[00500] Synthetic chemistry transformations and methodologies useful in synthesizing the compounds described herein are known in the art and include, for example, those described in R.
Larock, Comprehensive Organic Transformations (1989); T. W. Greene and P. G. M. Wuts, Protective Groups in Organic Synthesis, 2d. Ed. (1991); L. Fieser and M. Fieser, Fieser and Fieser ’s Reagents for Organic Synthesis (1994); and L. Paquette, ed., Encyclopedia of Reagents for Organic Synthesis (1995).
List of Abbreviations
[00501] AC2O = acetic anhydride; AcCl = acetyl chloride; AcOH = acetic acid; AIBN = azobisisobutyronitrile; aq. = aqueous; BusSnH = tributyltin hydride; Boc = tert-butyloxy carbonyl; CD3OD = deuterated methanol; CDCE = deuterated chloroform; CDI = 1,1 '-Carbonyldiimidazole; DBU = 1,8- diazabicyclo[5.4.0]undec-7-ene; DCM = dichloromethane; DEAD = diethyl azodicarboxylate; DIBAL-H = di-iso-butyl aluminium hydride; DIEA = DIPEA = N,N-diisopropylethylamine; DMAP = 4- dimethylaminopyridine; DMF = N,N-dimethylformamide; DMSO-de = deuterated dimethyl sulfoxide;
DMSO = dimethyl sulfoxide; DPPA = diphenylphosphoryl azide; EDC.HC1 = EDCI.HC1 = l-ethyl-3-(3- dimethylaminopropyl)carbodiimide hydrochloride; Et2O = diethyl ether; EtOAc = ethyl acetate; EtOH = ethanol; h = hour; HATU=2-(lH-7-azabenzotriazol-l-yl)-l,l,3,3-tetramethyl uronium hexafluorophosphate methanaminium; HMDS = hexamethyldisilazane; HOBT = 1 -hydroxybenzotriazole; i-PrOH = isopropanol; LAH = lithium aluminium hydride; LiHMDS = Lithium bis(trimethylsilyl)amide; MeCN = acetonitrile; Mel = methyl iodide; MeOH = methanol; MP -carbonate resin = macroporous triethylammonium methylpolystyrene carbonate resin; MsCl = mesyl chloride; MTBE = methyl tertiary butyl ether; MW = microwave irradiation ; n-BuLi = n-butyllithium; NaHMDS = Sodium bis(trimethylsilyl)amide; NaOMe = sodium methoxide; NaOtBu = sodium t-butoxide; NBS = N-bromosuccinimide; NCS = N-chloro- succinimide; NMI = 1 -methylimidazole; NMP = N-Methyl -2 -pyrrolidone; OAc = acetoxy; Pd(Ph3)4 = tetrakis(triphenylphosphine)palladium(0); Pd2(dba)3 = tris(dibenzylideneacetone)dipalladium(0);
PdCl2(PPh3)2 = bis(triphenylphosphine)palladium(II) dichloride; PG = protecting group; prep-HPLC = preparative high-performance liquid chromatography; PyBop = (benzotriazol- 1-yloxy)- tripyrrolidinophosphonium hexafluorophosphate; Pyr = pyridine; RT = room temperature; RuPhos = 2- dicyclohexylphosphino-2',6'-diisopropoxybiphenyl; sat. = saturated; ss = saturated solution; t-BuOH = tertbutanol; T3P = Propylphosphonic Anhydride; TBS = TBDMS = tert-butyldimethylsilyl; TBSC1 = TBDMSC1 = tert-butyldimethylchlorosilane; TEA = EtsN = triethylamine; TCFH = chloro-N, N, N’, N’- tetramethylformamidium hexafluorophosphate; TFA = trifluoroacetic acid; TFAA = trifluoroacetic anhydride; THF = tetrahydrofuran; Tol = toluene; TsCl = tosyl chloride; XPhos = 2-dicyclohexylphosphino- 2',4',6'-triisopropylbiphenyl.
SYNTHESIS OF REPRESENTATIVE POLYAMIDES
[00502] Example 1. Synthesis of 3-[(4-{4-[3-({4-[(2R)-2-[(tert-butoxycarbonyl)amino]-4-({l-methyl-4- H-methyl-4-(3-Hl-methyl-4-(l-methylimidazole-2-amido)pyrrol-2- yl]formamidoIpropanamido)imidazole-2-amido1pyrrol-2-vHformamido)butanamido1-l-
methylimidazol-2-yl}formamido)propanamido]-l-methylpyrrole-2-amido}-l-methylimidazol-2- yl)formamido]propanoic acid
[00504] Step 1: To a stirred solution of (2R)-2-[(tert-butoxycarbonyl)amino]-4-{[(9H-fluoren-9- ylmethoxy)carbonyl] amino [butanoic acid (3.00 g, 6.80 mmol, 0.80 equiv) in DMF (50.00 mb) was added DIEA (5.50 g, 42.55 mmol, 5.00 equiv), ethyl 4-amino-l-methylimidazole-2 -carboxylate (1.44 g, 8.51 mmol, 1.00 equiv) and PyBOP (5.76 g, 11.06 mmol, 1.30 equiv) in portions at 0 °C. The mixture was stirred for 1.0 h at r.t.. The reaction mixture was quenched with ice water (200 m ) at 0 °C. The precipitated solids were collected by filtration and washed with water (3x50 m ) and Et2O (3x30 mb) and then dried under vacuum. This resulted in ethyl 4-[(2R)-2-[(tert-butoxycarbonyl)amino]-4-{[(9H-fluoren-9- ylmethoxy)carbonyl] amino [butanamido] -1 -methylimidazole -2 -carboxylate (3.50 g, 86.94%) as a light yellow solid. EC/MS: mass calcd. For C31H37N5O7: 591.27, found: 592.50[M+H]+.
[00505] Step 2: Into a 50 ml flask was added ethyl 4-[(2R)-2-[(tert-butoxycarbonyl)amino]-4-{[(9H- fluoren-9-ylmethoxy)carbonyl]amino}butanamido]-l-methylimidazole-2-carboxylate (500.00 mg, 0.85 mmol, 1.00 equiv) in DMF (5.00 mb) and piperidine (1.00 mb). The reaction was stirred at r.t. for 30 mins. Piperidine was removed and the reaction mixture was purified by reverse flash chromatography with the following conditions: column, C18 column; mobile phase, MeCN in water (0.5% NH4HCO3), 10% to 50% gradient in 30 min; detector, UV 254 nm. The fractions were combined and concentrated. This resulted in ethyl 4-[(2R)-4-amino-2-[(tert- butoxy carbonyl)amino]butanamido]-l-methylimidazole-2 -carboxylate (250.00 mg, 96.10%) as a yellow oil. EC/MS: mass calcd. For C16H27N5O5: 369.20, found: 370.35[M+H]+.
[00506] Step 3: The procedure was the same as ethyl 4-[(2R)-2-[(tert-butoxycarbonyl)amino]-4-{[(9H- fluoren-9-ylmethoxy)carbonyl]amino}butanamido]-l-methylimidazole-2-carboxylate (Example 1 Step 1). 230.00 mg of ethyl 4-[(2R)-4-amino-2-[(tert- butoxycarbonyl)amino]butanamido]-l-methylimidazole-2- carboxylate was used and 200.00 mg of desired product was obtained as a yellow solid (68.39% yield). LC/MS: mass calcd. For C41H53N15O10: 915.41, found: 916.75[M+H]+.
[00507] Step 4: To a stirred solution of ethyl 4-[(2R)-2-[(tert-butoxycarbonyl) amino]-4-({ l-methyl-4-[l- methyl-4-(3-{[l -methyl -4-(l-methylimidazole-2 -amido) pyrrol-2-yl]formamido}propanamido)imidazole-2- amido]pyrrol-2-yl}formamido)butanamido]-l-methylimidazole-2 -carboxylate (570 mg, 1.00 equiv) in a mixture of solvents MeOH/THF (5:3), was added 2 M LiOH in water (6.00 equiv) at room temperature. The reaction mixture was stirred at room temperature for 2.0 h. The solvent was removed under reduced pressure and the residue was dissolved in H2O (20 m ). The mixture was acidified to pH 3~5 with 2 M HC1 at 0 °C. The precipitated solids were collected by filtration, washed with H2O (3x30 mb), and dried under vacuum to provide 370.00 mg of desired product as yellow solid (66.90% yield). LC/MS: mass calcd. For C39H49N15O10: 887.38, found: 888.85[M+H]+.
[00508] Step 5: The procedure was the same as ethyl 4-[(2R)-2-[(tert-butoxycarbonyl)amino]-4-{[(9H- fluoren-9-yhnethoxy)carbonyl]amino}butanamido]-l-methylimidazole-2-carboxylate (Example 1 Step 1). 380.00 mg of 4-[(2R)-2-[(tert-butoxycarbonyl)amino]-4-({l- methyl-4-[l-methyl-4-(3-{[l-methyl-4-(l- methylimidazole-2-amido)pyrrol-2-yl]formamido}propanamido)imidazole-2-amido]pyrrol-2- yl}formamido)butanamido]-l-methylimidazole-2 -carboxylic acid was used and 521.00 mg of the desired product was obtained as a white solid (91.75% yield). HRMS: mass calcd. For C5sH74N22Oi4: 1302.5755, found: 1303.5867[M+H]+.
[00509] Step 6: The procedure was the same as 4-[(2R)-2-[(tert-butoxycarbonyl)amino]-4-({l-methyl-4- [l-methyl-4-(3-{[l-methyl-4-(l-methylimidazole-2-amido)pyrrol-2-yl]formamido}propanamido)imidazole- 2-amido]pyrrol-2-yl}formamido)butanamido]-l-methylimidazole-2 -carboxylic acid, (Example 1 Step 4), but the reaction was run at room temperature. 150.00 mg of ethyl 3-[(4-{4-[3-({4-[(2R)-2-[(tert- butoxycarbonyl)amino]-4-({ l-methyl-4-[l-methyl-4-(3-{[l-methyl-4-(l-methylimidazole-2-amido)pyrrol-2- yl]formamido}propanamido)imidazole-2-amido]pyrrol-2-yl}formamido)butanamido]-l-methylimidazol-2- yl } formamido)propanamido] - 1 -methylpyrrole-2 -amido } - 1 -methylimidazol-2-yl)formamido]propanoate was used, and 146.00 mg crude of desired product was obtained as a white solid. LC/MS: mass calcd. For C56H7ON22OI4: 1274.54 found: 638.85[M/2+H]+.
[00510] Example 2. Synthesis of N-(5-{[(3R)-3-amino-3-({2-[(2-{[5-({2-[(2-{[5-(dimethylamino)pentyl] carbamoyl }ethyl)carbamoyl]-l-methylimidazol-4-yl}carbamoyl)-l- methylpyrrol-3- yl] carbarn oyl}ethyl)carbamoyl]-l-methylimidazol-4-yl} carbarn ovDpropyl] carbarn oyll-1- methylpyrrol-3-yl)-l-methyl-4-(3- {[l-methyl-4-(l-methylimidazole-2-amido)pyrrol-2-yl]formamidol propanamido) imidazole-2-carboxamide
[00511] Scheme 2.
[00512] Step 1: To a stirred mixture of 3-[(4-{4-[3-({4-[(2R)-2-[(tert-butoxycarbonyl)amino]- 4-({ 1- methyl-4-[ 1 -methyl -4-(3 - { [ 1 -methyl -4-( 1 -methylimidazole-2-amido)pyrrol-2- yl]formamido}propanamido)imidazole-2-amido]pyrrol-2-yl}formamido)butanamido]-l-methylimidazol-2- yl } formamido)propanamido] - 1 -methylpyrrole-2 -amido } - 1 -methylimidazol-2-yl)formamido]propanoic acid (432.00 mg, 0.33 mmol, 1.00 equiv) in DMF (4.00 mL) was added PyBOP (211.54 mg, 0.40 mmol, 1.20 equiv) and DIEA (131.34 mg, 1.01 mmol, 3.00 equiv) in portions at 0 °C. Then (5- aminopentyl)dimethylamine (48.53 mg, 0.37 mmol, 1.10 equiv) was added in portions at 0 °C. The resulting mixture was stirred for additional 1.0 h at room temperature. The reaction was poured into ice water (10 mL). The precipitated solids were collected by filtration, the filter cake was washed with H2O (3x5 mL), and dried under vacuum. This resulted in tert-butyl N-[(lR)-l-({2-[(2-{[5-({2-[(2-{[5- (dimethylamino)pentyl] carbamoyl } ethyl)carbamoyl] - 1 -methylimidazol-4-yl } carbamoyl)- 1 -methylpyrrol-3 - yl]carbamoyl }ethyl)carbamoyl] - 1 -methylimidazol-4-yl } carbamoyl)-3 -({ 1 -methyl -4-[ 1 -methyl -4-(3 -{[ 1 - methyl-4-(l-methylimidazole-2-amido)pyrrol-2-yl]formamido}propanamido)imidazole-2-amido]pyrrol-2- yl}formamido)propyl] carbamate (305.00 mg, 64.89%) as a yellow solid. LC/MS: mass calcd. For C63H86N24Oi3: 1386.68 found:694.70 [M/2+H]+.
[00513] Step 2: To a stirred solution of tert-butyl N-[(lR)-l-({2-[(2-{[5-({2-[(2-{[5- (dimethylamino)pentyl]carbamoyl } ethyl)carbamoyl] - 1 -methylimidazol-4-yl } carbamoyl)- 1 -methylpyrrol-3 - yl]carbamoyl }ethyl)carbamoyl] - 1 -methylimidazol-4-yl } carbamoyl)-3 -({ 1 -methyl -4-[ 1 -methyl -4-(3 -{[ 1 - methyl-4-(l-methylimidazole-2-amido)pyrrol-2-yl] formamido}propanamido)imidazole-2-amido]pyrrol-2- yl}formamido)propyl] carbamate (60.00 mg, 0.04 mmol, 1.00 equiv.) in DCM (2.00 mL) was added TFA (0.40 mL) dropwise at room temperature. The resulting mixture was stirred for 1.0 h at room temperature and was then concentrated under vacuum. N-(5-{[(3R)-3-amino-3-({2-[(2-{[5-({2-[(2-{[5-(dimethylamino) pentyl] carbamoyl} ethyl) carbamoyl] -l-methylimidazol-4-yl} carbamoyl)- 1 -methylpyrrol-3 -yl] carbamoyl} ethyl) carbamoyl] -l-methylimidazol-4-yl} carbamoyl) propyl] carbamoyl} -1 -methylpyrrol-3 -yl)-l -methyl - 4-(3-{[l-methyl-4-(l-methylimidazole-2-amido) pyrrol-2-yl] formamido} propanamido) imidazole-2-
carboxamide (60.00 mg crude) was obtained as a yellow oil. LC/MS: mass calcd. For C58H78N24O11: 1286.63, found: 430.20 [M/3+H]+.
[00514] Example 3. Synthesis of N-(5-f[(3R)-3-amino-3-f[l-methyl-2-(f2-[(l-methyl-5-f[l-methyl-2- (propylcarbamoyl)imidazol-4-yl]carbamoyl}pyrrol-3-yl)carbamoyl]ethyl}carbamoyl)imidazol-4- yl]carbamoyl}propyl]carbamoyl}-l-methylpyrrol-3-yl)-l-methyl-4-(3-{[l-methyl-4-(l- methylimidazole-2-amido)pyrrol-2-yl]formamido}propanamido)imidazole-2-carboxamide
[00516] Step 1: The procedure was the same as tert-butyl N-[( 1R)- 1-({2-[(2-{ [5-({2-[(2-{ [5- (dimethylamino)pentyl] carbamoyl } ethyl)carbamoyl] - 1 -methylimidazol-4-yl } carbamoyl)- 1 -methylpyrrol-3 - yl]carbamoyl }ethyl)carbamoyl] - 1 -methylimidazol-4-yl } carbamoyl)-3 -({ 1 -methyl -4-[ 1 -methyl -4-(3 -{[ 1 - methyl-4-(l-methylimidazole-2-amido)pyrrol-2-yl]formamido}propanamido)imidazole-2-amido]pyrrol-2- yl}formamido)propyl] carbamate (Example 2 Step 1), but the crude was purified by silica gel column chromatography. 965.00 mg of 4-[(2R)-2-[(tert-butoxycarbonyl) amino]-4-({ l-methyl-4-[l-methyl-4-(3-{[l- methyl-4-(l-methylimidazole-2 -amido) pyrrol-2-yl] formamido} propanamido) imidazole-2 -amido] pyrrol- 2-yl} formamido) butanamido]-l-methylimidazole-2 -carboxylic acid was used and 1.30 g of desired product was obtained as a white solid (83.48% yield). LC/MS: mass calcd. For C55H69N21O13: 1231.54, found: 617.15 [M/2+H]+.
[00517] Step 2: To a stirred solution of ethyl 4-{4-[3-({4-[(2R)-2-[(tert-butoxycarbonyl)amino]- 4-({ l- methyl-4-[ 1 -methyl -4-(3 - { [ 1 -methyl -4-( 1 -methylimidazole-2-amido)pyrrol-2- yl]formamido}propanamido)imidazole-2-amido]pyrrol-2-yl}formamido)butanamido]- l-methylimidazol-2-
yl}formamido)propanamido]-l-methylpyrrole-2-amido}-l-methylimidazole-2 -carboxylate (1.30 g, 1.05 mmol, 1.00 equiv.) in MeOH (13.00 mL) was added 2M LiOH.H2O (2.11 mL, 4.22 mmol, 4.00 equiv.) dropwise at room temperature. The mixture was stirred for 2.0 h at 45 °C. The mixture was concentrated under reduced pressure and the residue was dissolved in H2O (20.00 mL). The mixture was acidified to pH 3~5 with 2M HC1. The precipitated solids were collected by filtration, washed with H2O (3x5.00 mL), and dried under vacuum to yield 4-{4-[3-({4-[(2R)-2-[(tert-butoxycarbonyl)amino]-4-({ 1 -methyl -4-[l- methyl- 4-(3 - { [ 1 -methyl -4-( 1 -methylimidazole-2-amido)pyrrol-2-yl] formamido } propanamido)imidazole-2- amido]pyrrol-2-yl } formamido)butanamido] - 1 - methylimidazol-2-yl } formamido)propanamido] - 1 - methylpyrrole-2-amido}-l- methylimidazole -2 -carboxylic acid (1.20 g, 81.86%) as a white solid. LC/MS: mass calcd. For C53H65N2iOi3: 1203.51, found: 1204.45 [M+H]+.
[00518] Step 3: The procedure was the same as tert-butyl N-[(lR)-l-({2-[(2-{[5-({2-[(2-{[5- (dimethylamino)pentyl] carbamoyl } ethyl)carbamoyl] - 1 -methylimidazol-4-yl } carbamoyl)- 1 -methylpyrrol-3 - yl]carbamoyl }ethyl)carbamoyl] - 1 -methylimidazol-4-yl } carbamoyl)-3 -({ 1 -methyl -4-[ 1 -methyl -4-(3 -{[ 1 - methyl-4-(l-methylimidazole-2-amido)pyrrol-2-yl]formamido}propanamido)imidazole-2-amido]pyrrol-2- yl}formamido)propyl] carbamate (Example 2 Step 1). 400.00 mg of 4-{4-[3-({4-[(2R)-2-[(tert- butoxycarbonyl)amino]-4-({l-methyl-4-[l-methyl-4-(3-{[l-methyl-4-(l-methylimidazole-2-amido)pyrrol-2- yl]formamido}propanamido)imidazole-2-amido]pyrrol-2-yl}formamido)butanamido]-l-methylimidazol-2- yl}formamido)propanamido]-l-methylpyrrole-2-amido}-l-methylimidazole-2-carboxylic acid was used and 320.00 mg of desired product was obtained as a white solid (77.36% yield). LC/MS: mass calcd. For C56H72N22OI2: 1244.57, found: 1245.65 [M+H]+.
[00519] Step 4: The procedure was the same as N-(5-{[(3R)-3-amino-3-({2-[(2-{[5-({2-[(2-{[5- (dimethylamino)pentyl] carbamoyl } ethyl) carbamoyl] - 1 -methylimidazol-4-yl } carbamoyl)- 1 -methylpyrrol-3 - yl] carbamoyl }ethyl)carbamoyl] - 1 -methylimidazol-4-yl } carbamoyl)propyl] carbamoyl } - 1 -methylpyrrol-3 - yl)- 1 -methyl -4-(3 - { [ 1 -methyl -4-( 1 -methylimidazole-2-amido)pyrrol-2- yl]formamido}propanamido)imidazole-2 -carboxamide (Example 2 Step 2). 59.80 mg of tert-butyl N-[(1R)- 1 -{[ 1 -methyl -2-( {2-[( 1 -methyl -5 -{[ 1 -methyl -2-(propylcarbamoyl)imidazol-4-yl] carbamoyl }pyrrol-3 - yl)carbamoyl]ethyl } carbamoyl)imidazol-4-yl] carbamoyl } -3 -( { 1 -methyl -4-[ 1 -methyl -4-(3 - { [ 1 -methyl -4-( 1 - methylimidazole-2-amido)pyrrol-2-yl]formamido}propanamido)imidazole-2-amido]pyrrol-2- yl}formamido)propyl] carbamate was used and 59.80 mg of the desired product was obtained as a yellow oil. LC/MS: mass calcd. For C51H64N22O10: 1144.52, found: 1145.50 [M+H]+.
SYNTHESIS OF TARGETING LIGAND
[00520] Example 4. Synthesis of (R)-2-(4-(4-chlorophenyl)-2,3,9-trimethyl-6H-thieno[3,2- f|[l,2,4]triazolo[4,3-a][l,4] diazepin-6-yl)-N-(4-hvdroxyphenyl)acetamide
[00521] Scheme 4.
[00522] Step 1: To a stirred solution of (2R)-2-[[(9H-fluoren-9-ylmethoxy)carbonyl]amino]-4-methoxy- 4-oxobutanoic acid (5.00 g, 13.54 mmol, 1.00 equiv) in DMF (60.00 mb) was added NMI (3.33 g, 40.61 mmol, 3.00 equiv), TCFH (5.70 g, 20.31 mmol, 1.50 equiv) and 3-(4-chlorobenzoyl)-4,5-dimethylthiophen- 2-amine (3.96 g, 14.90 mmol, 1.10 equiv) in portions at 0 °C. The resulting mixture was stirred for 2 h at room temperature. The reaction was poured into water/ice (200 mb) and the precipitated solids were collected by fdtration, washed with water (3x30 mb), and dried under vacuum. The solid was purified by silica gel column chromatography, eluted with PE/EA (3: 1) to afford methyl (3R)-3-[[3-(4-chlorobenzoyl)- 4,5- dimethylthiophen-2-yl]carbamoyl]-3-[[(9H-fluoren-9-ylmethoxy)carbonyl]amino]propanoate (4.95 g, 56.00%) as a yellow solid. LCMS: mass calcd. For C33H29CIN2O6S: 616.14, found: 617.05 [M+H]+ [00523] Step 2: To a solution of methyl (3R)-3-amino-3-[[3-(4-chlorobenzoyl)-4,5-dimethylthiophen-2- yl] carbamoyl] propanoate (4.95 g) in DCM was treated with EtsN followed by AcOH treatment in DCE to give 2.10 g of methyl 2-[(3R)-5-(4-chlorophenyl)-6,7-dimethyl-2-oxo-lH,3H-thieno[2,3-e][l,4]diazepin-3- yl]acetate as a yellow solid (70.00% yield of two steps). LCMS: mass calcd. For C18H17CIN2O3S: 376.06, found: 377.15 [M+H]+.
[00524] Step 3: To a solution of methyl 2-[(3R)-5-(4-chlorophenyl)-6,7-dimethyl-2-oxo-lH,3H- thieno[2,3-e][l,4]diazepin-3-yl]acetate (1.10 g, 2.92 mmol, 1.00 equiv) in toluene (25.00 ml) was added P2S5 (1.30 g, 0.006 mmol, 2.00 equiv) and ISfeCCE (0.62 g, 5.85 mmol, 2.00 equiv). The reaction was stirred at 110 °C for 17 h. The solid was fdtered and the fdtrate was concentrated. The residue was purified by silica gel column chromatography, eluted with PE: EA = 1: 1 to afford methyl 2-[(3R)-5-(4- chlorophenyl)-6,7-dimethyl-2-sulfanylidene-lH,3H-thieno[2,3-e][l,4]diazepin-3-yl]acetate (2 g, crude) as a yellow solid. LCMS: mass calcd. For C18H17CIN2O2S2: 392.04, found: 393.10 [M+H]+.
[00525] Step 4: To a solution of methyl 2-[(3R)-5-(4-chlorophenyl)-6,7-dimethyl-2-sulfanylidene-lH,3H- thieno[2,3-e][l,4]diazepin-3-yl]acetate (900.00 mg, 2.29 mmol, 1.00 equiv) in THF (6.00 mb) was added NH2NH2.H2O (344.00 mg, 6.87 mmol, 3.00 equiv) dropwise at 0 °C. The reaction was stirred at 0 °C for 1 h. AcCl (539.41 mg, 6.87 mmol, 3.00 equiv) and EtsN (695.34 mg, 6.87 mmol, 3.00 equiv) were added and the mixture was stirred at 0 °C for 30 mins. The reaction mixture was then quenched with H2O (15 mb), extracted with EA (3x15 mb), washed with brine (20 mb), and dried over Na2SO4. The solid was filtrated
and the filtrate was concentrated to dryness. The crude product was dissolved in AcOH (10.0 mL) and stirred at 40 °C for 4 h. The solvent was removed and the residue was purified by reverse flash chromatography with the following conditions: column, C18 column; mobile phase, MeCN in water (0.05% TFA), 5% to 50% gradient in 40 min; detector, UV 254 nm to afford methyl (R)-2-(4-(4-chlorophenyl)- 2,3,9-trimethyl-6H-thieno[3,2-f][l,2,4]triazolo[4,3-a][l,4]diazepin-6-yl)acetate(400.00 mg, 42.09%) as a yellow solid. LCMS: mass calcd. For C20H19CIN4O2S: 414.09, found: 415.10 [M+H]+.
[00526] Step 5: A solution of methyl (R)-2-(4-(4-chlorophenyl)-2,3,9-trimethyl-6H-thieno[3,2-f] [l,2,4]triazolo[4,3-a][l,4]diazepin-6-yl)acetate (400.00 mg, 0.96 mmol, 1.00 equiv) in LiOH (2M, 2.00 mL, 4.00 equiv) and MeOH (2.00 mL) was stirred at 40 °C for 2 h. The mixture was acidified with HC1 (2M) and concentrated. The residue was dissolved in DMF (2 mL) and purified by reverse phase column with the following conditions: column, C18 column; mobile phase, MeCN in water (0.05% TFA),
5% to 50% gradient in 40 min; detector, UV 254 nm to afford (R)-2-(4-(4-chlorophenyl)-2,3,9-trimethyl- 6H-thieno[3,2-f][l,2,4]triazolo[4,3-a][l,4]diazepin-6-yl)acetic acid (340.00 mg, 87.97% yield) as a white solid. LCMS: mass calcd. For C19H17CIN4O2S: 400.09, found: 401.10 [M+H]+.
[00527] Example 5. Synthesis of 4 difluorophenoxy)-5-ethanesulfonamidophenyl]-6-methyl-7-
oxo-lH-pyrrolo[2,3-c]pyridine-2-carboxylic acid
[00528] Scheme 5.
[00529] Step 1: A solution of 2,4-difluorophenol (2.66 g, 20.45 mmol, 1.50 equiv) and 2-bromo-l-fluoro- 4-nitrobenzene (3.00 g, 13.64 mmol, 1.00 equiv) in DMF (50.00 mb) was stirred at 85 °C for 3.0 h. The reaction was cooled to room temperature and was then diluted with water (150 mL). The precipitate was filtered and the filter cake was washed with water and dried to afford 2-bromo-l-(2,4-difluorophenoxy)-4- nitrobenzene (4.40 g, 98.21%) as light a yellow solid. 'HNMR (300 MHz, DMSO-t/e) 5: 8.56-8.58 (m, 1H), 8.18-8.22 (m, 1H), 7.48-7.63 (m, 2H), 7.22-7.28(m, 1H), 6.99 (d, J= 9.0 Hz, 1H).
[00530] Step 2: Iron powder (7.78 g, 139.36 mmol, 10.00 equiv) was added in portions to a stirred solution of 2-bromo-l-(2,4-difluorophenoxy)-4-nitrobenzene (4.40 g, 13.37 mmol, 1.00 equiv) in EtOH (20.00 mL) and THF (20.00 mL), NH4CI (7.45 g, 139.36 mmol, 10.00 equiv), and H2O (20.00 mL) at room temperature. The mixture was heated at 70 °C for 2.0 h. The reaction mixture was cooled to room temperature and was then filtered through a pad of Celite. The filtrate was concentrated under vacuum and the resulting residue was diluted with water (20 mL) and extracted with EA (50 mLx3). The combined organic layers were washed with brine (100 mL), dried over Na2SO4, and concentrated to afford 3-bromo-4- (2,4-difluorophenoxy)aniline (3.77 g, 65.28%) as a brown solid. LC/MS: mass calcd. For CpHsB^NO: 298.98, found: 299.90, 301.90 [M+H, M+H+2]+.
[00531] Step 3: To a mixture of 3-bromo-4-(2,4-difluorophenoxy)aniline (2.51 g, 8.36 mmol, 1.00 equiv) and EtsN (3.39 g, 33.45 mmol, 4.00 equiv) in DCM (10.00 mL) a solution of ethane sulfonyl chloride (3.23 g, 25.09 mmol, 3.00 equiv) in DCM (10.00 mL) was added dropwise at 0 °C. The reaction mixture was stirred at room temperature for 3.0 h. The reaction was concentrated under vacuum, diluted with water (20 mL), and extracted with EA (20 mLx3). The combined organic layers were washed with brine, dried over Na2SC>4, and concentrated under vacuum. The resulting crude was suspended in 1,4-dioxane (30.00 mL) and 10% NaOH (6.00 mL) and the mixture was heated at 100 °C for 2.0 h. The reaction was concentrated under vacuum, diluted with water (20 mL), and extracted with EA (20 mLx3). The combined organic layers were washed with brine, dried over ISfeSCL and concentrated to afford N-[3-bromo-4-(2,4- difluorophenoxy)phenyl] ethane sulfonamide (3.00 g, 92.02% yield) as a brown solid. LC/MS: mass calcd. For Ci4Hi2BrF2NO3S: 390.97, found: 392.21 [M+H]+. ’H NMR (300 MHz, DMSO-t/6) 5: 9.94(s, 1H), 7.46- 7.53 (m, 2H), 7.20-7.24 (m, 1H), 7.07-7.15(m, 2H), 6.88-7.04 (m, 1H), 3.09-3.16(m, 2H), 1.21(t, J= 7.2 Hz, 3H).
[00532] Step 4: A mixture of N-[3-bromo-4-(2,4-difluorophenoxy)phenyl]ethanesulfonamide (1.00 g, 2.55 mmol, 1.00 equiv) and ethyl 6-methyl-7-oxo-4-(4,4,5,5-tetramethyl-l,3,2-dioxaborolan-2-yl)-lH- pyrrolo[2,3-c]pyridine-2-carboxylate (970.00 mg, 2.80 mmol, 1.10 equiv), Pd(dtbpf)C12 (166.00 mg, 0.25 mmol, 0.10 equiv), and K3PO4 (1.10 g, 5.18 mmol, 2.03 equiv) in toluene (8.00 mL) and H2O (2.00 mL) was stirred for 1.0 h at 75 °C under nitrogen atmosphere. The reaction mixture was cooled to room temperature and was T quenched with water (70.00 mL). The mixture was extracted with EA (50 mLx3) and the combined organic layers were washed with water (40 mLx2), dried over anhydrous Na2SC>4 and concentrated under reduced pressure. The residue was purified by silica gel column chromatography, eluted with QLCL/MeOH (15: 1) to afford ethyl 4-[2-(2,4-difluorophenoxy)-5-ethanesulfonamidophenyl]-6-
methyl-7-oxo-lH-pyrrolo[2,3-c]pyridine-2-carboxylate (1.20 g, 88.55%) as a brown solid. LC/MS: mass calcd. For C25H23F2N3O6S: 531.13, found: 532.10 [M+H]+.
[00533] Step 5: The procedure was the same as 4-{4-[3-({4-[(2R)-2-[(tert-butoxycarbonyl)amino]-4-({l- methyl-4-[ 1 -methyl -4-(3 - { [ 1 -methyl -4-( 1 -methylimidazole-2-amido)pyrrol-2- yl]formamido}propanamido)imidazole-2-amido]pyrrol-2-yl}formamido)butanamido]-l-methylimidazol-2- yl}formamido)propanamido]-l-methylpyrrole-2-amido}-l-methylimidazole-2-carboxylic acid (Example 3 Step 2). 1.10 g of ethyl 4-[2-(2,4-difluorophenoxy)-5-ethanesulfonamidophenyl]-6-methyl-7-oxo-lH- pyrrolo[2,3-c]pyridine-2 -carboxylate was used and 1.00 g of the desired product was obtained as a grey solid (95.97% yield). LC/MS: mass calcd. For C23Hi9F2N3O6S: 503.10, found: 504.10 [M+H]+.
[00534] Example 6. Synthesis of tert-butyl (3R,5R)-3-((5-bromo-2-methyl-3-oxo-2,3- dihvdropyridazin-4-yl)amino)-5-phenylpiperidine-l-carboxylate
[00536] Step 1: To a solution of 4,5-dibromo-2H-pyridazin-3-one (5.00 g, 19.69 mmol, 1.00 equiv.) in DMF (50.00 mL) was added methyl iodide (8.39 g, 59.08 mmol, 3.00 equiv.) and K2CO3 (6.80 g, 49.24 mmol, 2.50 equiv.). The resulting mixture was stirred for 17.0 h at room temperature. The reaction mixture was poured into water (200 mL). The precipitated solids were collected by fdtration and washed with water (150.00 mL) and dried to afford 4,5-dibromo-2-methylpyridazin-3-one (3.30 g, 62.54%) as a yellow solid. LC/MS: mass calcd. For CMLB ^O: 265.87, found: 266.85, 268.85 [M+H, M+H+2]+.
[00537] Step 2: To a stirred solution of picolinic acid (10.14 g, 82.38 mmol, 1.10 equiv.) in DCM (100 mL) was added HATU (34.17 g, 89.87 mmol, 1.20 equiv.) and DIEA (10.65 g, 82.38 mmol, 1.10 equiv.). The resulting mixture was stirred at 25 °C for 0.5 h. The tert-butyl (3R)-3-aminopiperidine-l-carboxylate (15.00 g, 74.89 mmol, 1.00 equiv.) and DIEA (19.36 g, 149.79 mmol, 2.00 equiv.) were added in portions
and the resulting mixture was stirred at 25 °C for 1.0 h. Then the resulting mixture was concentrated under vacuum and the residue was purified by silica gel column chromatography (30-50% EA/PE) to afford tertbutyl (3R)-3-(pyridine-2-amido)piperidine-l-carboxylate (22.00 g, 96.19%) as a yellow solid. LC/MS: mass calcd. For C16H23N3O3: 305.17, found: 306.10 [M+H]+.
[00538] Step 3: To a stirred solution of tert-butyl (3R)-3-(pyridine-2-amido)piperidine-l-carboxylate (19.00 g, 62.22 mmol, 1.00 equiv.) in 2-methylpropan-2-ol (100 mL) was added iodophenyl (63.47 g, 311.09 mmol, 5.00 equiv.), 2,6-dimethylbenzoic acid (2.34 g, 15.58 mmol, 0.25 equiv.), Pd(OAc)2 (1.40 g, 6.22 mmol, 0.10 equiv.) and Ag2CC>3 (17.16 g, 62.22 mmol, 1.00 equiv.) and the resulting mixture was stirred at 100 °C for 16.0 h. Then the resulting mixture was concentrated under vacuum and the residue was purified by silica gel column chromatography (30-50% EA/PE) to afford tert-butyl (3R,5R)-3-phenyl-5- (pyridine-2-amido)piperidine-l -carboxylate (10.70 g, 45.08%) as a yellow solid. LC/MS: mass calcd. For C22H27N3O3 : 381.21, found: 326.15 [M+H-56]+.
[00539] Step 4: To a stirred solution of tert-butyl (3R,5R)-3-phenyl-5-(pyridine-2-amido)piperidine-l- carboxylate (10.70 g, 28.05 mmol, 1.00 equiv) in i-PrOH (100 mL) was added NaOH (11.22 g, 280.49 mmol, 10.00 equiv.) and the reaction mixture was stirred for 16.0 h at 85 °C. The reaction was filtered and the filtrate was concentrated. The resulting residue was purified by reverse flash chromatography with the following conditions: column, C18 silica gel; mobile phase, MeCN in water (0.05% NH4HCO3), 30% to 50% gradient in 30 min; Detector, UV 254 nm. The fractions were combined and concentrated to afford tertbutyl (3R,5R)-3-amino-5-phenylpiperidine-l-carboxylate (6.50 g, 83.85%) as a white solid. LC/MS: mass calcd. For C16H24N2O2: 276.18, found: 277.10 [M+H]+.
[00540] Step 5: To a stirred solution of tert-butyl (3R,5R)-3-amino-5-phenylpiperidine-l-carboxylate (6.00 g, 21.71 mmol, 1.00 equiv.) in DMF (50 mL) was added 4,5-dibromo-2-methylpyridazin-3-one (5.82 g, 21.71 mmol, 1.00 equiv.) and DIEA (11.22 g, 86.84 mmol, 4.00 equiv.). The reaction mixture was irradiated with microwave radiation for 10.0 h at 120 °C and then 100.00 mL H2O was added. The resulting mixture was extracted with EA (100 mLx3), the organic layers were combined, dried over anhydrous sodium sulfate, filtered, and concentrated. The residue was purified by silica gel column chromatography (30-50% EA/PE) to afford tert-butyl (3R,5R)-3-[(5-bromo-l-methyl-6-oxopyridazin-4-yl)amino]-5- phenylpiperidine-1 -carboxylate (5.02 g, 46.83%) as a yellow solid and tert-butyl (3R,5R)-3-((5-bromo-2- methyl-3-oxo-2,3-dihydropyridazin-4-yl)amino)-5-phenylpiperidine-l-carboxylate (2.00 g, 19.88%) as a yellow solid. LC/MS: mass calcd. For C2iH2?BrN4O3: 462.13, found: 463.10, 465.10 [M+H, M+H+2]+.
[00541] Example 7. Synthesis of (S)-2-chloro-4-(2-(5-(3,5-dimethylisoxazol-4-yl)-l-(2- morpholinopropyl)-lH-benzo[d1imidazol-2-yl)ethyl)phenol
[00542] Scheme 7.
[00543] Step 1: Into a 500 ml flask was added 4-bromo-l-fluoro-2-nitorbenzene (15.40 g, 70.00 mmol, 1.00 equiv), 3, 5 -dimethyl- l,2-oxazol-4-ylboronic acid (9.87 g, 70.001 mmol, 1.00 equiv), NaHC’CF (11.76 g, 140.00 mmol, 2.00 equiv), Pd(dppf)C12.CH2C12 (2.86 g, 3.500 mmol, 0.05 equiv), DME (100.00 mb), and H2O (10.00 mL) and the reaction mixture was stirred at 95 °C for 3.0 h under N2 atmosphere. The reaction was concentrated and the residue was purified by silica gel column with PE/A (1 : 1) to yield 4-(4-fluoro-3- nitrophenyl)-3,5-dimethyl-l,2-oxazole (15.7 g , 90.21% yield) as a white solid. LC/MS: mass calcd. For CnH9FN2O3:236.06, found: 237.10[M+H]+.
[00544] Step 2: Into a 500 ml flask was added 4-(4-fluoro-3-nitrophenyl)-3,5-dimethyl-l,2- oxazole (12.70 g, 53.77 mmol, 1.00 equiv), (2S)-propane-I,2-diamine (3.99 g, 53.92 mmol, 1.00 equiv), K2CO3 (14.86 g, 107.54 mmol, 2.00 equiv), and CH3CN (300.00 mL) and the reaction was stirred at 50 °C for 24.0 h. The reaction was filtered and washed with CH3CN. The filtrate was concentrated and the residue was purified by silica gel column with PE/EA (1: 1) to afford (2S)-Nl-[4-(3,5-dimethyl-l,2-oxazol-4-yl)-2- nitrophenyl]propane-I,2- diamine (6.10 g, 37.01% yield) as a yellow oil. LC/MS: mass calcd. For CnH9FN2O3:290.14, found: 291.05[M+H]+.
[00545] Step 3: Into a 500 ml flask was added (2S)-NI-[4-(3,5-dimethyl-l,2-oxazol-4-yl)-2- nitrophenyl]propane-I,2-diamine (6.10 g, 21.01 mmol, 1.00 equiv) and CH3CN (200.00 mL). Then 1- bromo-2-(2-bromoethoxy)ethane (5.36 g, 23.11 mmol, 1.10 equiv), K2CO3 (8.71 g, 63.03mmol, 3.00 equiv), and KI (4.19 g, 25.21 mmol, 1.20 equiv) were added and the reaction mixture was stirred at 80 °C for 60.0 h. The reaction was fdtered and the fdtrate was concentrated. The residue was purified by silica gel column chromatography with PE/EA (1: 1) to afford 4-(3,5-dimethyl-l,2-oxazol-4-yl)-N-[(2S)-2-(morpholin-4-
yl)propyl] -2 -nitroaniline (6.00 g, 71.31% yield) as a red solid. LC/MS: mass calcd. For C18H24N4O4: 360.18, found: 361.10[M+H]+.
[00546] Step 4: Into a 500 ml flask was added 4-(3,5-dimethyl-l,2-oxazol-4-yl)-N-[(2S)-2-(morpholin-4- yl)propyl] -2 -nitroaniline (3.00 g, 8.32 mmol, 1.00 equiv), EtOH (50.00 mL), H2O (50.00 mL), and Na2S2C>4 (7.25 g, 41.62 mmol, 5.00 equiv) and the reaction was stirred at 80 °C for 1.0 h. The reaction was then concentrated under the reduced pressure and the residue was extracted with EA (50 mLx3). The combined organic phase was washed with NaCl (100 mL), dried over anhydrous ISfeSCE, and concentrated. The residue (1.70 g, 61.19% yield) was used directly in the next step without further purification. LC/MS: mass calcd. For C18H26N4O2: 330.21, found: 331.15[M+H]+.
[00547] Step 5: Into a 250 ml flask was added 4-(3,5-dimethyl-l,2-oxazol-4-yl)-NI-[(2S)-2-(morpholin- 4- yl)propyl] benzene- 1,2-diamine (1.70 g, 5.15 mmol, 1.00 equiv), EA (60.00 mL), 3-(3-chloro-4- hydroxyphenyl) propanoic acid (1.03 g, 5.15 mmol, 1.00 equiv), T3P (2.46 g, 7.72 mmol, 1.50 equiv), and DIEA (2.66 g, 20.58 mmol, 4.00 equiv) and the reaction mixture was stirred at 80 °C for 1.0 h. Then the reaction mixture was washed with H2O (20 ml x 3) and the combined organic phase was dried with anhydrous Na2SO4 and concentrated. The residue (1.10 g, 27.92% yield) was used directly in the next step without further purification. LC/MS: mass calcd. For C27H33CIN4O4: 512.22, found: 513.15 [M+H]+.
[00548] Step 6: Into a 100 ml flask was added 3-(3-chloro-4-hydroxyphenyl)-N-[5-(3,5-dimethyl-l,2- oxazol-4-yl)-2-[[(2S)-2-(morpholin-4-yl)propyl]amino]phenyl]propanamide (1.10 g, 2.14 mmol, 1.00 equiv) and HC1 (20.00 mL) and the reaction mixture was stirred at 65 °C for 40 mins. The reaction was concentrated and the residue was purified by reverse flash chromatography with the following conditions: Column: C18 column; mobile phase: MeCN in water (0.05% NH4HCO3), 10% to 50% gradient in 30 min; Detector: UV 254 nm. The fractions were combined and concentrated to afford 2-chloro-4-[2-[5 -(3,5- dimethyl-l,2-oxazol-4-yl)-l-[(2S)-2-(morpholin-4-yl)propyl]-I,3-benzodiazol-2-yl]ethyl]phenol (490.00 mg, 46.00% yield) as a white oil. LC/MS: mass calcd. For C27H31CIN4O3: 494.21, found: 495.05[M+H]+.
[00549] 'HNMR (400 MHz, Methanol-d4) 5: 7.52 - 7.56 (m, 2H), 7.17 - 7.23 (m, 2H), 6.94 (d, J= 8.3 Hz, 1H), 6.81 (d, J= 8.2 Hz, 1H), 4.16 - 4.21 (m, 1H), 3.92 - 3.97 (m, 1H), 3.58 - 3.66 (m, 4H), 3.21 - 3.25 (m, 2H), 3.12 - 3.16 (m, 2H), 2.82 - 2.90 (m, 1H), 2.66 - 2.72 (m, 2H), 2.39 - 2.59 (m, 5H), 2.29 (s, 3H), 0.99 (d, J= 6.7 Hz, 3H).
[00550] 13C NMR (75 MHz, MeOD) 5: 165.35, 158.86, 155.96, 151.51, 141.90, 134.59, 132.82, 129.54, 127.68, 123.98, 123.50, 120.25, 118.50, 117.01, 116.32, 110.56, 78.27, 77.83, 77.40, 66.86, 59.47, 32.55, 29.25, 10.79, 10.20, 9.51.
SYNTHESIS OF REPRESENTATIVE COMPOUNDS
[00551] Example 8. Synthesis of Compound A-l [00552] Scheme 8.
[00553] Step 1: A solution of 50-(4-nitrophenoxy)-3,6,9,12,15,18,21,24,27,30,33,36,39,42,45,48- hexadecaoxapentacontan-l-ol (1 g) and tert-butoxypotassium (2.00 equiv) in DCM (10.00 mL) was stirred for 3.0 h at room temperature. The reaction was quenched with ice-water (20 mL) and extracted with DCM (30 mLx3). The combined organic layers were dried over anhydrous ISfeSCL and concentrated under reduced pressure. The residue was purified to afford the desired product as a yellow oil (1.00 g, 86.70% yield). LC/MS: mass calcd. For C46H83NO22: 1001.54, found: 1024.45 [M+Na]+.
[00554] Step 2: To a solution of tert-butyl 53-(4-nitrophenoxy) 3,6,9,12,15,18,21,24,27,30,33,36,39,42,45,48,51-heptadecaoxatripentacontanoate, in EA was added Pd/C (25% w/w) and the reaction was stirred for 6.0 h at room temperature under H2 atmosphere. The mixture was filtered and the filtrate was concentrated to afford the desired product was obtained as a yellow oil (965.00 mg, 90.43% yield). LC/MS: mass calcd. For C46H85NO20: 971.57, found: 972.70 [M+H]+.
[00555] Step 3: The procedure was the same as tert-butyl N-[( 1R)- 1-({2-[(2-{ [5-({2-[(2-{ [5- (dimethylamino)pentyl] carbamoyl } ethyl)carbamoyl] - 1 -methylimidazol-4-yl } carbamoyl)- 1 -methylpyrrol-3 - yl]carbamoyl }ethyl)carbamoyl] - 1 -methylimidazol-4-yl } carbamoyl)-3 -({ 1 -methyl -4-[ 1 -methyl -4-(3 -{[ 1 - methyl-4-(l-methylimidazole-2-amido)pyrrol-2-yl]formamido}propanamido)imidazole-2-amido]pyrrol-2- yl}formamido)propyl] carbamate (Example 2 Step 1), but the crude was purified by reverse phase column. 450.00 mg of tert-butyl 53-(4-aminophenoxy)-3,6,9,12,15,18,21,24,27,30,33,36,39,42,45,48,51- heptadecaoxatripentacontanoate was used and 610.00 mg of the desired product was obtained as a yellow oil (90.41% yield). LC/MS: mass calcd. For C69H102F2N4O25S: 1457.63, found: 701.80 [M/2-28+H]+.
[00556] Step 4: The procedure was the same as l-(4-nitrophenoxy)-3,6,9,12,15,18,21, 24,27,30,33,36- dodecaoxanonatriacontan-39-oic acid (Example 2 Step 2). 70.00 mg of tert-butyl 53-(4-{4-[2-(2,4- difluorophenoxy)-5-ethanesulfonamidophenyl]- 6-methyl-7-oxo-lH-pyrrolo[2,3-c]pyridine-2-
amido}phenoxy)-3,6,9,12,15,18,21,24,27,30,33,36,39,42,45,48,51-heptadecaoxatripentacontanoate was used, 70.00 mg crude of desired product was obtained as yellow oil. LC/MS: mass calcd. For C65H94F2N4O25S: 1401.53, found: 701.60 [M/2+H]+.
[00557] Step 5: Synthesis of Compound A-l
[00558] The procedure was the same as tert-butyl N-[(1R)-1 -({2-[(2-{ [5-({2-[(2-{ [5- (dimethylamino)pentyl]carbamoyl } ethyl)carbamoyl] - 1 -methylimidazol-4-yl } carbamoyl)- 1 -methylpyrrol-3 - yl]carbamoyl }ethyl)carbamoyl] - 1 -methylimidazol-4-yl } carbamoyl)-3 -({ 1 -methyl -4-[ 1 -methyl -4-(3 -{[ 1 - methyl-4-(l-methylimidazole-2-amido)pyrrol-2-yl]formamido}propanamido)imidazole-2-amido]pyrrol-2- yl}formamido)propyl] carbamate (Example 2 Step 2), but the reaction mixture was purified by Prep-HPLC.
59.22 mg of N-(5-{[(3R)-3-amino-3-[(l-methyl-2-{[2-({ l-methyl-5-[(l-methyl-2-{[2- (propylcarbamoyl)ethyl] carbamoyl } imidazol-4-yl)carbamoyl]pyrrol-3 - yl } carbamoyl)ethyl]carbamoyl } imidazol-4-yl)carbamoyl]propyl]carbamoyl }- 1 -methylpyrrol-3 -yl)- 1 - methyl-4-(3-{[l-methyl-4-(l-methylimidazole-2-amido)pyrrol-2-yl]formamido}propanamido)imidazole-2- carboxamide was used, 4.00 mg of desired product was obtained as white solid (2.82% yield). HRMS: mass calcd. For C119H161F2N27O35S: 2598.1337, found: 2599.1426 [M+H]+.
[00559] Example 9. General synthesis and purification of the compounds of the disclosure
[00560] Compounds of the disclosure were made by methods similar to Examples 1-8. The compounds were subsequently purified by HRMS methods A or B.
[00561] Method A: Instrument: Waters Acquity I Class UPLC with Xevo G2-XSQ Tof HRMS; Column: ACQUITY UPLC BEH-C18, 2.1 x 50 mm, 2.7 pm; mobile phase A: H2O (0.1% HCOOH), mobile B, CAN (0. 1% HCOOH); Flow rate:0.4mL/min; Gradient: 10% B to 95% B in 1.5 min, hold 95% for another 0.5 min, then down to 10% B in 0.3 min, hold 10% B for another 0.7 min; detector: 254nm.
[00562] Method B: Instrument: Waters Acquity I Class UPLC with Xevo G2-XS Q Tof HRMS; Column: ACQUITY UPLC BEH-C18, 2.1 x 50 mm, 2.7pm; mobile phase A: H2O (0.1% HCOOH), mobile B, CAN (0.1% HCOOH); Flow rate:0.4 mL/min; Gradient: 5% B to 40% B in 2.0min, to 95% in another 1.5 min, hold 95% for 1.5 min, then down to 5% B in 0.3 min, hold 5% B for another 0.7 min; detector: 254nm.
[00563] Experimental data for the compounds of the disclosure purified by Method A are provided in Table 4.
BIOLOGICAL EXAMPLES
[00564] Example B-l. Activity
[00565] Fibroblast: a cell type derived from a skin biopsy of a patient. These cells are not altered genetically, so they serve as a primary cell culture model of disease.
[00566] iPSC: induced pluripotent stem cell, a cell type that results as a reprogramming of another cell type (typically skin cells or blood cells) into a more embryonic-like state that enables the development of other cell types to model therapeutic effects of drugs in vitro.
[00567] SNP: Single Nucleotide Polymorphism, a variation in a single base pair in a DNA sequence
[00568] Molecular Biology Toolkit:
• qPCR primer probe sets: o RNA input normalization was assessed utilizing human glyceraldehyde 3-phosphate dehydrogenase (hGAPDH) TaqMan assay (ThermoFisher cat# 4351370) or Human Cyclophillin (IAPP) TaqMan assay (ThermoFisher cat# 4351372) o Total HTT detection was assessed utilizing human Htt TaqMan assay (ThermoFisher cat# 4331182) o Allele-specific detection of human HTT expression in HD cells containing the SNP rs362331C/T
(Exon 50): for each assay, allele-specific probes to detect the SNP variant contained locked nucleic acid bases to improve allele discrimination, as compared to unmodified DNA probes.
■ 362331-F (331 forward primer)
■ 362331-R (331 reverse primer)
■ 362331 -C probe : TCC CTC ATC + C + AC TGT GT
■ 362331-T probe: CTC + A + T + C + T + A + C TGT GT o qPCR was performed using Agpath ID one-step reverse transcriptase polymerase chain reaction (RT-PCR) reagent
[00569] Protein measurements were performed via western blots probing with antibody MW1 (polyQ specific) to assess reduction of mtHTT alone. D7F7 (a.a surrounding Prol218) was used to visualize both wtHTT and mtHTT. Lysates were standardized by DC prior to separation on a 3-8% Tris-acetate gel and transferred via wet transfer method onto nitrocellulose membranes. Blots were probed with the previously
mentioned antibodies and complementary fluorescent secondary antibodies and imaged on the Li-Cor Odyssey® DLx Imaging system.
[00570] Antibody pairing of 2B7 (a.a. 1-17) and MW1 (polyQ specific) will be used to track mtHTT levels while pairing of MAB2166 (a.a. 181-810) and MAB5490 (a.a. 115-129) will be employed to track total full- length HTT.
[00571] Screening of HD molecules methods: GM09197 and/or GM04022 fibroblasts were cultured in T175 flasks incubated at 37 °C and 5% CO2. Once confluency was reached, the media was removed, the cells were washed with IX PBS, and cells were dissociated using TrypLE™ Express Enzyme. Media was added to the enzyme and collected into a 15-mL conical tube and centrifuged at 500xg for 5 minutes to pellet the cells. Media and enzyme were aspirated using a serological pipette. Cells were resuspended in fresh media and counted using a Countess 3 Automated Cell Counter. Cells were plated at a density of 15,000 cells/well in a tissue culture -treated polystyrene 96-well dish and incubated at 37 °C and 5% CO2 overnight. The next day, media was removed using an 8-channel aspirator. 200 pL media/well are added back into the plate. The molecules are formulated to 1 mM and are dispensed using a Multidrop™ Pico 8 Digital Dispenser. After a 48-hour incubation with compounds, media was removed from plates, cells were washed with IX PBS, and cells were lysed in 40 pL per well guanidinium thiocyanate buffer. RNA was isolated and purified in 382-well glass fiber column plates using chaotropic salts. Human mtHTT, wtHTT, and GAPDH mRNA were measured via RT-PCR using the ThermoFisher QuantStudio™ 7 Flex System in 384-well format. Results of HTT levels were normalized to GAPDH mRNA levels. Normalized HTT mRNA levels were expressed relative to vehicle-treated samples to assess fold change after molecule treatment.
[00572] iPSC-Neuron Duration of Action of HD molecules methods: Fibroblasts isolated from HD patients were reprogrammed into iPSCs expanded in the presence of cytokines and transduced with the Sendai virus, a cytoplasmic RNA vector. These iPSCs expressed stem cell markers and have normal karyotypes and express the pluripotent markers Nanog, Tra-1-60, and SSeA-44. iPSC-derived neuron differentiation methodology followed standard protocols for mixed cortical neuron differentiation resulting in immunohistochemical staining of iPS-Neuron of Tuj 1 and Map2. iPSC-neuronal precursor cells were plated at 300,000 cells/well in a PLO/Laminin-521 coated culture-treated polystyrene 96-well dish and incubated at 37 °C and 5% CO2. The next day, media was changed to allow neuron precursor cells to continue maturation into neurons. Four days later, media was refreshed, and cells were treated with mitotic inhibitor to remove any remaining dividing cells, resulting in a pure neuronal culture. Three days later, media was removed. 200 pL media/well were added back into the plate. The molecules were formulated to 1 mM and dispensed using a Multidrop™ Pico 8 Digital Dispenser. After 96-hour incubation with compounds, media was removed and refreshed and the cells were retreated. After 7 days of compound exposure, media was removed from plates, and cells were lysed in 60 pL of Ambion Lysis buffer. RNA is isolated using PureLink™ RNA isolation kits. cDNA is synthesized with Agilent Superscript II kit. Human mtHTT, wtHTT, and GAPDH mRNA were measured via RT-PCR using the ThermoFisher QuantStudio™ 7 Flex
System in 384-well format. Results of HTT levels were normalized to GAPDH mRNA levels. Normalized HTT mRNA levels were expressed relative to vehicle-treated samples to assess fold change after treatment with the HD compounds.
[00573] Representative in vitro biochemical data is presented in Table 5, where A < 100 nM, B > 100 nM to < 1 pM, C > 1 pM.
[00574] While preferred embodiments of the present invention have been shown and described herein, it will be obvious to those skilled in the art that such embodiments are provided by way of example only. Numerous variations, changes, and substitutions will now occur to those skilled in the art without departing from the invention. It should be understood that various alternatives to the embodiments of the invention described herein may be employed in practicing the invention. It is intended that the following claims define the scope of the invention and that methods and structures within the scope of these claims and their equivalents be covered thereby.
Claims
WHAT IS CLAIMED IS:
1. A transcription modulator molecule having a first terminus, a second terminus, and an oligomeric backbone moiety, or a pharmaceutically acceptable salt thereof, wherein: a) the first terminus comprises a DNA-binding moiety having the structure of Formula (A-l):
Formula (A-l), wherein:
W1 is hydrogen, halogen, optionally substituted C1-C10 alkyl, -NRleRlf, -NRleC(O)Rlf, - NRleC(O)NRleRlf, -C(O)NRleRlf, -OC(O)NRleRlf, -NRleC(O)ORlf, -N=C(N(Rle)2)2, -ZB- PO(ORle)2, -ZB-(CH2)p3-PO(ORle)2, or -ZB-(CH2)P3-O-PO(ORle)2, wherein each Rle is independently hydrogen or optionally substituted C1-C10 alkyl; each Rlf is independently hydrogen, optionally substituted C1-C10 alkyl, optionally substituted C1-C10 heteroalkyl, AAp2, or optionally substituted 5-membered heteroaryl, wherein each AA is an amino acid and p2 is an integer from 1-10;
ZB is N or O;
P3 is 1-10;
W2 is optionally substituted Ci-C2o alkyl, optionally substituted Ci-C2o aminoalkyl, optionally substituted Ci-C2o haloalkyl, optionally substituted Ci-C2o heteroalkyl, or optionally substituted Ci-C2o hydroxyalkyl; or W2 is L’-Z-R4;
Rw is hydrogen or optionally substituted Ci-C2o alkyl; or
Rw and W2 combine together with the atoms to which they are attached to form an optionally substituted 4 to 10-membered heterocycloalkyl; each Y1, Y2, Y3, Y4, Y5, Y6, Y7, and Y8 is independently N or CH; each X1, X2, X3, X4, X5, X6, X7, and X8 is independently S, O, or NR2; each R2 is independently hydrogen, optionally substituted C1-C50 alkyl, optionally substituted -Cso alkenyl, optionally substituted C2-C5o alkynyl, optionally substituted C1-C50 heteroalkyl,
optionally substituted C2-C50 heteroalkenyl, optionally substituted C2-C50 heteroalkynyl, optionally substituted C1-C50 haloalkyl, optionally substituted C3-C10 cycloalkyl, optionally substituted 3 to 10-membered heterocycloalkyl, or optionally substituted PEG1.50;
L1 is C1-C20 alkylene or C2-C20 heteroalkylene;
Z is absent, -C(O)-, or -C(=NH)-;
R4 is C1-C3 alkyl, -OR4b, or -NR4aR4b; wherein
R4a is hydrogen, optionally substituted C1-C20 alkyl, or optionally substituted C1-C20 heteroalkyl;
R4b is optionally substituted C1-C20 alkyl, optionally substituted C2-C20 alkenyl, optionally substituted C2-C20 alkynyl, optionally substituted C1-C20 aminoalkyl, optionally substituted C1-C20 haloalkyl, optionally substituted C1-C20 heteroalkyl, optionally substituted C1-C20 hydroxyalkyl, optionally substituted C3-C8 cycloalkyl, optionally substituted 4 to 8- membered heterocycloalkyl, optionally substituted phenyl, or optionally substituted 5 to 10- membered heteroaryl; or
R4a and R4b together with the nitrogen to which they are attached form an optionally substituted 3 to 10-membered heterocycloalkyl which is partially or fully saturated; each R3 is independently hydrogen, halogen, Ci-Ce alkyl, -OR3c, -NR3cR3d, or -NHC(O)R3e, wherein R3c and R3d are each independently hydrogen, alkyl, or PEG;
R3e is alkyl, PEG, cycloalkyl, heterocycloalkyl, or phenyl; or two R3 together with the atoms to which they are attached form an optionally substituted C3-C6 cycloalkyl or 3 to 6 membered heterocycloalkyl; ji is 0 or 1; no is 0 or 1 ; mi and are each independently an integer from 0-3; and pi is 3 or 4; wherein the linker is attached at one of R3 and the others of R3 are each hydrogen; or the linker is attached at the cycloalkyl or heterocycloalkyl formed by two R3; b) the second terminus comprises a protein-binding moiety capable of binding to a regulatory molecule that modulates expression of a gene; and c) the oligomeric backbone links the first terminus and the second terminus.
The molecule of claim 1, or a pharmaceutically acceptable salt thereof, wherein the DNA-binding moiety comprises a structure of Formula (A-2):
Formula (A-2), wherein:
W1 is hydrogen or -N=C(N(Rle)2)2, wherein each Rle is independently hydrogen or C1-C3 alkyl;
W2 is optionally substituted C1-C20 alkyl, optionally substituted C1-C20 aminoalkyl, optionally substituted C1-C20 haloalkyl, optionally substituted C1-C20 heteroalkyl, or optionally substituted C1-C20 hydroxyalkyl; or W2 is L’-Z-R4; wherein
L1 is C1-C20 alkylene or C2-C20 heteroalkylene;
Z is absent, -C(O)-, or -C(=NH)-;
R4 is C1-C3 alkyl, -OR4b, or -NR4aR4b; wherein
R4a is hydrogen, optionally substituted C1-C20 alkyl, or optionally substituted C1-C20 heteroalkyl;
R4b is optionally substituted C1-C20 alkyl, optionally substituted C2-C20 alkenyl, optionally substituted C2-C20 alkynyl, optionally substituted C1-C20 aminoalkyl, optionally substituted C1-C20 haloalkyl, optionally substituted C1-C20 heteroalkyl, optionally substituted C1-C20 hydroxyalkyl, optionally substituted C’s-Cs cycloalkyl, optionally substituted 4 to 8-membered heterocycloalkyl, optionally substituted phenyl, or optionally substituted 5 to 10-membered heteroaryl; or
R4a and R4b together with the nitrogen to which they are attached form an optionally substituted 3 to 10-membered heterocycloalkyl which is partially or fully saturated;
Rw is hydrogen or optionally substituted C1-C20 alkyl; or
Rw and W2 combine together with the atoms to which they are attached to form an optionally substituted 4 to 10-membered heterocycloalkyl; each Y1, Y2, Y3, Y4, Y5, Y6, Y7, and Y8 is independently N or CH; each X1, X2, X3, X4, X5, X6, X7, and X8 is independently S, O, or NR2;
each R2 is independently hydrogen, optionally substituted C1 -C50 alkyl, optionally substituted C2-C50 alkenyl, optionally substituted C2-C50 alkynyl, optionally substituted C1 -C50 heteroalkyl, optionally substituted C2-C50 heteroalkenyl, optionally substituted C2-C50 heteroalkynyl, optionally substituted C1 -C50 haloalkyl, optionally substituted C3-C10 cycloalkyl, optionally substituted 3 to 10-membered heterocycloalkyl, or optionally substituted PEG1.50;
R3a and R3b are each independently hydrogen, halogen, Ci-Ce alkyl, -OR3c, -NR3cR3d, or - NHC(O)R3e, wherein
R3c and R3d are each independently hydrogen, alkyl, or PEG;
R3e is alkyl, PEG, cycloalkyl, heterocycloalkyl, or phenyl; or two R3a or two R3b together with the carbon atom to which they are attached form a C3-C6 cycloalkyl or 4 to 6-membered heterocycloalkyl; ji is 0 or 1; and mi and ni are each independently an integer from 0-3; wherein the linker is attached at R3a or R3b; or at the cycloalkyl or heterocycloalkyl formed through the combination of two R3a or two R3b. The molecule of claim 1 or 2, or a pharmaceutically acceptable salt thereof, wherein each X1, X2, X3, X4, X5, X6, X7, and X8 is independently NR2. The molecule of any one of claims 1-3, or a pharmaceutically acceptable salt thereof, wherein ji is 0. The molecule of any one of claims 1 -3 , or a pharmaceutically acceptable salt thereof, wherein j 1 is 1. The molecule of any one of claims 1-5, or a pharmaceutically acceptable salt thereof, wherein mi and are each independently 2 or 3. The molecule of claim 1, or a pharmaceutically acceptable salt thereof, wherein the DNA-binding moiety comprises a structure of Formula (A-3):
Formula (A-3), wherein:
W1 is hydrogen or -N=C(N(Rle)2)2, wherein
each Rle is independently hydrogen or C1-C3 alkyl;
W2 is optionally substituted C1-C20 alkyl, optionally substituted C1-C20 aminoalkyl, optionally substituted C1-C20 haloalkyl, optionally substituted C1-C20 heteroalkyl, or optionally substituted C1-C20 hydroxyalkyl; or W2 is L’-Z-R4; wherein
L1 is C1-C20 alkylene or C2-C20 heteroalkylene;
Z is absent, -C(O)-, or -C(=NH)-;
R4 is C1-C3 alkyl, -OR4b, or -NR4aR4b; wherein
R4a is hydrogen, optionally substituted C1-C20 alkyl, or optionally substituted C1-C20 heteroalkyl;
R4b is optionally substituted C1-C20 alkyl, optionally substituted C2-C20 alkenyl, optionally substituted C2-C20 alkynyl, optionally substituted C1-C20 aminoalkyl, optionally substituted C1-C20 haloalkyl, optionally substituted C1-C20 heteroalkyl, optionally substituted C1-C20 hydroxyalkyl, optionally substituted C’s-Cs cycloalkyl, optionally substituted 4 to 8-membered heterocycloalkyl, optionally substituted phenyl, or optionally substituted 5 to 10-membered heteroaryl; or
R4a and R4b together with the nitrogen to which they are attached form an optionally substituted 3 to 10-membered heterocycloalkyl which is partially or fully saturated;
Rw is hydrogen or optionally substituted C1-C20 alkyl; or Rw and W2 combine together with the atoms to which they are attached to form an optionally substituted 4 to 10-membered heterocycloalkyl; each Y1, Y2, Y3, Y4, Y5, Y6, Y7, and Y8 is independently N or CH; each R2a, R2b, R2c, R2d, R2e, R2f, R2g, and R2h is independently hydrogen, optionally substituted C1-C50 alkyl, optionally substituted C2-C50 alkenyl, optionally substituted C2-C50 alkynyl, optionally substituted C1-C50 heteroalkyl, optionally substituted C2-C50 heteroalkenyl, optionally substituted C2-C50 heteroalkynyl, optionally substituted C1-C50 haloalkyl, optionally substituted C3-C10 cycloalkyl, optionally substituted 3 to 10-membered heterocycloalkyl, or optionally substituted PEG1.50; each of which is optionally substituted with one or more Rz; each Rz is independently -CN, -OH, -ORZa, -N3, -NRZaRzb, -CO(O)RZc, -C(O)ORZc, -C(O)NRZaRzb, - NHC(O)RZc, -NHC(O)ORZc, -OC(O)NRZaRzb, or optionally substituted 5 to 10-membered heteroaryl; wherein
RZa and Rzb are each independently hydrogen, alkyl, or PEG;
RZc is alkyl, PEG, cycloalkyl, heterocycloalkyl, or phenyl; each R3b is independently hydrogen, halogen, Ci-Ce alkyl, -OR3c, -NR3cR3d, or -NHC(O)R3e, wherein R3c and R3d are each independently hydrogen, alkyl, or PEG;
R3e is alkyl, PEG, cycloalkyl, heterocycloalkyl, or phenyl; or two R3b together with the carbon atom to which they are attached form a C’s-C,, cycloalkyl or 4 to 6-membered heterocycloalkyl; and
ni and mi are each independently 0 or 1.
8. The molecule of claim 1, or a pharmaceutically acceptable salt thereof, wherein the DNA-binding moiety comprises a structure of Formula (A-4):
Formula (A-4), wherein:
W1 is hydrogen or -N=C(N(Rle)2)2, wherein each Rle is independently hydrogen or C1-C3 alkyl;
W2 is optionally substituted C1-C20 alkyl, optionally substituted C1-C20 aminoalkyl, optionally substituted C1-C20 haloalkyl, optionally substituted C1-C20 heteroalkyl, or optionally substituted C1-C20 hydroxyalkyl; or W2 is L’-Z-R4; wherein
L1 is C1-C20 alkylene or C2-C20 heteroalkylene;
Z is absent, -C(O)-, or -C(=NH)-;
R4 is C1-C3 alkyl, -OR4b, or -NR4aR4b; wherein
R4a is hydrogen, optionally substituted C1-C20 alkyl, or optionally substituted C1-C20 heteroalkyl;
R4b is optionally substituted C1-C20 alkyl, optionally substituted C2-C20 alkenyl, optionally substituted C2-C20 alkynyl, optionally substituted C1-C20 aminoalkyl, optionally substituted C1-C20 haloalkyl, optionally substituted C1-C20 heteroalkyl, optionally substituted C1-C20 hydroxyalkyl, optionally substituted C’s-Cs cycloalkyl, optionally substituted 4 to 8-membered heterocycloalkyl, optionally substituted phenyl, or optionally substituted 5 to 10-membered heteroaryl; or
R4a and R4b together with the nitrogen to which they are attached form an optionally substituted 3 to 10-membered heterocycloalkyl which is partially or fully saturated;
Rw is hydrogen or optionally substituted C1-C20 alkyl; or Rw and W2 combine together with the atoms to which they are attached to form an optionally substituted 4 to 10-membered heterocycloalkyl;
each Y1, Y2, Y3, Y4, Y5, Y6, Y7, and Y8 is independently N or CH; each R2a, R2b, R2c, R2d, R2e, R2f, R2g, and R2h is independently hydrogen, optionally substituted C1-C50 alkyl, optionally substituted C2-C50 alkenyl, optionally substituted C2-C50 alkynyl, optionally substituted C1-C50 heteroalkyl, optionally substituted C2-C50 heteroalkenyl, optionally substituted C2-C50 heteroalkynyl, optionally substituted C1-C50 haloalkyl, optionally substituted C3-C10 cycloalkyl, optionally substituted 3 to 10-membered heterocycloalkyl, or optionally substituted PEG1.50; each of which is optionally substituted with one or more Rz; each Rz is independently -CN, -OH, -ORZa, -N3, -NRZaRzb, -CO(O)RZc, -C(O)ORZc, -C(O)NRZaRzb, - NHC(O)RZc, -NHC(O)ORZc, -OC(O)NRZaRzb, or optionally substituted 5 to 10-membered heteroaryl; wherein
RZa and Rzb are each independently hydrogen, alkyl, or PEG;
RZc is alkyl, PEG, cycloalkyl, heterocycloalkyl, or phenyl; each R3a is independently hydrogen, halogen, Ci-Cg alkyl, -OR3c, -NR3cR3d, or -NHC(O)R3e, wherein R3c and R3d are each independently hydrogen, alkyl, or PEG;
R3e is alkyl, PEG, cycloalkyl, heterocycloalkyl, or phenyl; or two R3a together with the carbon atom to which they are attached form a G-C, cycloalkyl or 4 to 6-membered heterocycloalkyl; and and mi are each independently 0 or 1. The molecule of claim 7 or 8, or a pharmaceutically acceptable salt thereof, wherein Y2, Y4, and Y7 are each independently N; and Y1 and Y3 are each independently CH. The molecule of any one of claims 7-9, or a pharmaceutically acceptable salt thereof, wherein Y8 is N. The molecule of claim 7, or a pharmaceutically acceptable salt thereof, wherein the DNA-binding moiety comprises a structure of Formula (A-5):
Formula (A-5), wherein:
W1 is hydrogen or -N=C(N(Rle)2)2, wherein each Rle is independently hydrogen or C1-C3 alkyl;
W2 is optionally substituted C1-C20 alkyl, optionally substituted C1-C20 aminoalkyl, optionally substituted C1-C20 haloalkyl, optionally substituted C1-C20 heteroalkyl, or optionally substituted C1-C20 hydroxyalkyl; or W2 is L’-Z-R4; wherein
L1 is C1-C20 alkylene or C2-C20 heteroalkylene;
Z is absent, -C(O)-, or -C(=NH)-;
R4 is C1-C3 alkyl, -OR4b, or -NR4aR4b; wherein
R4a is hydrogen, optionally substituted C1-C20 alkyl, or optionally substituted C1-C20 heteroalkyl;
R4b is optionally substituted C1-C20 alkyl, optionally substituted C2-C20 alkenyl, optionally substituted C2-C20 alkynyl, optionally substituted C1-C20 aminoalkyl, optionally substituted C1-C20 haloalkyl, optionally substituted C1-C20 heteroalkyl, optionally substituted C1-C20 hydroxyalkyl, optionally substituted C’s-Cs cycloalkyl, optionally substituted 4 to 8-membered heterocycloalkyl, optionally substituted phenyl, or optionally substituted 5 to 10-membered heteroaryl; or
R4a and R4b together with the nitrogen to which they are attached form an optionally substituted 3 to 10-membered heterocycloalkyl which is partially or fully saturated;
Rw is hydrogen or optionally substituted C1-C20 alkyl; or Rw and W2 combine together with the atoms to which they are attached to form an optionally substituted 4 to 10-membered heterocycloalkyl; each Y5 and Y6 is independently N or CH; each R2a, R2b, R2c, R2d, R2e, R2f, R2g, and R2h is independently hydrogen, optionally substituted C1-C50 alkyl, optionally substituted C2-C50 alkenyl, optionally substituted C2-C50 alkynyl, optionally substituted C1-C50 heteroalkyl, optionally substituted C2-C50 heteroalkenyl, optionally substituted C2-C50 heteroalkynyl, optionally substituted C1-C50 haloalkyl, optionally substituted C3-C10 cycloalkyl, optionally substituted 3 to 10-membered heterocycloalkyl, or optionally substituted PEG1.50; each of which is optionally substituted with one or more Rz; each Rz is independently -CN, -OH, -ORZa, -N3, -NRZaRzb, -CO(O)RZc, -C(O)ORZc, -C(O)NRZaRzb, - NHC(O)RZc, -NHC(O)ORZc, -OC(O)NRZaRzb, or optionally substituted 5 to 10-membered heteroaryl; wherein
RZa and Rzb are each independently hydrogen, alkyl, or PEG;
RZc is alkyl, PEG, cycloalkyl, heterocycloalkyl, or phenyl; and and mi are each independently 0 or 1.
12. The molecule of claim 8, or a pharmaceutically acceptable salt thereof, wherein the DNA-binding moiety comprises a structure of Formula (A-6):
Formula (A-6), wherein:
W1 is hydrogen or -N=C(N(Rle)2)2, wherein each Rle is independently hydrogen or C1-C3 alkyl;
W2 is optionally substituted C1-C20 alkyl, optionally substituted C1-C20 aminoalkyl, optionally substituted C1-C20 haloalkyl, optionally substituted C1-C20 heteroalkyl, or optionally substituted C1-C20 hydroxyalkyl; or W2 is L’-Z-R4; wherein
L1 is C1-C20 alkylene or C2-C20 heteroalkylene;
Z is absent, -C(O)-, or -C(=NH)-;
R4 is C1-C3 alkyl, -OR4b, or -NR4aR4b; wherein
R4a is hydrogen, optionally substituted C1-C20 alkyl, or optionally substituted C1-C20 heteroalkyl;
R4b is optionally substituted C1-C20 alkyl, optionally substituted C2-C20 alkenyl, optionally substituted C2-C20 alkynyl, optionally substituted C1-C20 aminoalkyl, optionally substituted C1-C20 haloalkyl, optionally substituted C1-C20 heteroalkyl, optionally substituted C1-C20 hydroxyalkyl, optionally substituted Cs-Cs cycloalkyl, optionally substituted 4 to 8-membered heterocycloalkyl, optionally substituted phenyl, or optionally substituted 5 to 10-membered heteroaryl; or
R4a and R4b together with the nitrogen to which they are attached form an optionally substituted 3 to 10-membered heterocycloalkyl which is partially or fully saturated;
Rw is hydrogen or optionally substituted C1-C20 alkyl; or Rw and W2 combine together with the atoms to which they are attached to form an optionally substituted 4 to 10-membered heterocycloalkyl; each Y5 and Y6 is independently N or CH; each R2a, R2b, R2c, R2d, R2e, R2f, R2g, and R2h is independently hydrogen, optionally substituted C1-C50 alkyl, optionally substituted C2-C50 alkenyl, optionally substituted C2-C50 alkynyl, optionally
substituted C1-C50 heteroalkyl, optionally substituted C2-C50 heteroalkenyl, optionally substituted C2-C50 heteroalkynyl, optionally substituted C1-C50 haloalkyl, optionally substituted C3-C10 cycloalkyl, optionally substituted 3 to 10-membered heterocycloalkyl, or optionally substituted PEG1.50; each of which is optionally substituted with one or more Rz; each Rz is independently -CN, -OH, -ORZa, -N3, -NRZaRzb, -CO(O)RZc, -C(O)ORZc, -C(O)NRZaRzb, - NHC(O)RZc, -NHC(O)ORZc, -OC(O)NRZaRzb, or optionally substituted 5 to 10-membered heteroaryl; wherein
RZa and Rzb are each independently hydrogen, alkyl, or PEG;
RZc is alkyl, PEG, cycloalkyl, heterocycloalkyl, or phenyl; and ni and mi are each independently 0 or 1.
13. The molecule of any one of claims 1-12, or a pharmaceutically acceptable salt thereof, wherein Rw and W2 combine together with the atoms to which they are attached to form an optionally substituted 4 to 10-membered heterocycloalkyl.
14. The molecule of any one of claims 1-12, or a pharmaceutically acceptable salt thereof, wherein W2 is optionally substituted C1-C20 alkyl or optionally substituted C1-C20 haloalkyl.
15. The molecule of claim 13, or a pharmaceutically acceptable salt thereof, wherein W2 is Ci-Ce alkyl.
16. The molecule of any one of claims 1-12, 14, or 15, or a pharmaceutically acceptable salt thereof, wherein Rw is hydrogen.
17. The molecule of any one of claims 11-16, or a pharmaceutically acceptable salt thereof, wherein each R2a, R2b, R2c, R2d, R2e, R2f, R2g, and R2h is independently hydrogen, optionally substituted C1-C20 alkyl, or optionally substituted C1-C20 haloalkyl, each of which is optionally substituted with one or more Rz.
18. The molecule of any one of claims 11-16, or a pharmaceutically acceptable salt thereof, wherein each R2a, R2b, R2c, R2d, R2e, R2f, R2g, and R2h is independently C1-C20 alkyl.
19. The molecule of claim 18, or a pharmaceutically acceptable salt thereof, wherein each R2a, R2b, R2c, R2d, R2e, R2f, R2g, and R2h is independently methyl, ethyl, or propyl.
20. The molecule of any one of claims 1-10, or a pharmaceutically acceptable salt thereof, wherein Rw is hydrogen and W2is L’-Z-R4.
21. The molecule of claim 7, or a pharmaceutically acceptable salt thereof, wherein the DNA-binding moiety comprises a structure of Formula (A-7):
Formula (A-7), wherein:
W1 is hydrogen or -N=C(N(Rle)2)2, wherein each Rle is independently hydrogen or C1-C3 alkyl; each Y1, Y2, Y3, Y4, Y5, Y6, Y7, and Y8 is independently N or CH; each R2a, R2b, R2c, R2d, R2e, R2f, R2g, and R2h is independently hydrogen, optionally substituted C1-C50 alkyl, optionally substituted C2-C50 alkenyl, optionally substituted C2-C50 alkynyl, optionally substituted C1-C50 heteroalkyl, optionally substituted C2-C50 heteroalkenyl, optionally substituted C2-C50 heteroalkynyl, optionally substituted C1-C50 haloalkyl, optionally substituted C3-C10 cycloalkyl, optionally substituted 3 to 10-membered heterocycloalkyl, or optionally substituted PEG1.50; each of which is optionally substituted with one or more Rz;
L1 is C1-C20 alkylene or C2-C20 heteroalkylene;
Z is absent, -C(O)-, or -C(=NH)-;
R4 is C1-C3 alkyl, -OR4b, or -NR4aR4b; wherein
R4a is hydrogen, optionally substituted C1-C20 alkyl, or optionally substituted C1-C20 heteroalkyl;
R4b is optionally substituted C1-C20 alkyl, optionally substituted C2-C20 alkenyl, optionally substituted C2-C20 alkynyl, optionally substituted C1-C20 aminoalkyl, optionally substituted C1-C20 haloalkyl, optionally substituted C1-C20 heteroalkyl, optionally substituted C1-C20 hydroxyalkyl, optionally substituted Cs-Cs cycloalkyl, optionally substituted 4 to 8- membered heterocycloalkyl, optionally substituted phenyl, or optionally substituted 5 to 10- membered heteroaryl; or
R4a and R4b together with the nitrogen to which they are attached form an optionally substituted 3 to 10-membered heterocycloalkyl which is partially or fully saturated; each Rz is independently -CN, -OH, -ORZa, -N3, -NRZaRzb, -CO(O)RZc, -C(O)ORZc, -C(O)NRZaRzb, - NHC(O)RZc, -NHC(O)ORZc, -OC(O)NRZaRzb, or optionally substituted 5 to 10-membered heteroaryl; wherein
RZa and Rzb are each independently hydrogen, alkyl, or PEG;
RZc is alkyl, PEG, cycloalkyl, heterocycloalkyl, or phenyl; each R3b is independently hydrogen, halogen, Ci-Ce alkyl, -NR3cR3d, or -NHC(O)R3e, wherein
R3c and R3d are each independently hydrogen, alkyl, or PEG;
R3e is alkyl, PEG, cycloalkyl, heterocycloalkyl, or phenyl; or two R3b together with the carbon atom to which they are attached form a C3-C6 cycloalkyl or 4 to 6-membered heterocycloalkyl; and ni and mi are each independently 0 or 1.
22. The molecule of claim 8, or a pharmaceutically acceptable salt thereof, wherein the DNA-binding moiety comprises a structure of Formula (A-8):
Formula (A-8), wherein:
W1 is hydrogen or -N=C(N(Rle)2)2, wherein each Rle is independently hydrogen or C1-C3 alkyl; each Y1, Y2, Y3, Y4, Y5, Y6, Y7, and Y8 is independently N or CH; each R2a, R2b, R2c, R2d, R2e, R2f, R2g, and R2h is independently hydrogen, optionally substituted C1-C50 alkyl, optionally substituted C2-C50 alkenyl, optionally substituted C2-C50 alkynyl, optionally substituted C1-C50 heteroalkyl, optionally substituted C2-C50 heteroalkenyl, optionally substituted C2-C50 heteroalkynyl, optionally substituted C1-C50 haloalkyl, optionally substituted C3-C10 cycloalkyl, optionally substituted 3 to 10-membered heterocycloalkyl, or optionally substituted PEG1.50; each of which is optionally substituted with one or more Rz;
L1 is C1-C20 alkylene or C2-C20 heteroalkylene;
Z is absent, -C(O)-, or -C(=NH)-;
R4 is C1-C3 alkyl, -OR4b, or -NR4aR4b; wherein
R4a is hydrogen, optionally substituted C1-C20 alkyl, or optionally substituted C1-C20 heteroalkyl;
R4b is optionally substituted C1-C20 alkyl, optionally substituted C2-C20 alkenyl, optionally substituted C2-C20 alkynyl, optionally substituted C1-C20 aminoalkyl, optionally substituted C1-C20 haloalkyl, optionally substituted C1-C20 heteroalkyl, optionally substituted C1-C20 hydroxyalkyl, optionally substituted Cs-Cs cycloalkyl, optionally substituted 4 to 8- membered heterocycloalkyl, optionally substituted phenyl, or optionally substituted 5 to 10- membered heteroaryl; or
R4a and R4b together with the nitrogen to which they are attached form an optionally substituted 3 to 10-membered heterocycloalkyl which is partially or fully saturated; each Rz is independently -CN, -OH, -ORZa, -N3, -NRZaRzb, -CO(O)RZc, -C(O)ORZc, -C(O)NRZaRzb, - NHC(O)RZc, -NHC(O)ORZc, -OC(O)NRZaRzb, or optionally substituted 5 to 10-membered heteroaryl; wherein
RZa and Rzb are each independently hydrogen, alkyl, or PEG;
RZc is alkyl, PEG, cycloalkyl, heterocycloalkyl, or phenyl; each R3a is independently hydrogen, halogen, Ci-Ce alkyl, -NR3cR3d, or -NHC(O)R3e, wherein R3c and R3d are each independently hydrogen, alkyl, or PEG;
R3e is alkyl, PEG, cycloalkyl, heterocycloalkyl, or phenyl; or two R3a together with the carbon atom to which they are attached form a G-C, cycloalkyl or 4 to 6-membered heterocycloalkyl; and and mi are each independently 0 or 1.
23. The molecule of claim 21 or 22, or a pharmaceutically acceptable salt thereof, wherein Y2, Y4, and Y7 are each independently N; and Y1 and Y3 are each independently CH.
24. The molecule of any one of claims 21-23, or a pharmaceutically acceptable salt thereof, wherein Y8 is N.
25. The molecule of claim 21, or a pharmaceutically acceptable salt thereof, wherein the DNA-binding moiety comprises a structure of Formula (A-9):
Formula (A-9),
wherein:
W1 is hydrogen or -N=C(N(Rle)2)2, wherein each Rle is independently hydrogen or C1-C3 alkyl; each Y5 and Y6 is independently N or CH; each R2a, R2b, R2c, R2d, R2e, R2f, R2g, and R2h is independently hydrogen, optionally substituted C1-C50 alkyl, optionally substituted C2-C50 alkenyl, optionally substituted C2-C50 alkynyl, optionally substituted C1-C50 heteroalkyl, optionally substituted C2-C50 heteroalkenyl, optionally substituted C2-C50 heteroalkynyl, optionally substituted C1-C50 haloalkyl, optionally substituted C3-C10 cycloalkyl, optionally substituted 3 to 10-membered heterocycloalkyl, or optionally substituted PEG1.50; each of which is optionally substituted with one or more Rz;
L1 is C1-C20 alkylene or C2-C20 heteroalkylene;
Z is absent, -C(O)-, or -C(=NH)-;
R4 is C1-C3 alkyl, -OR4b, or -NR4aR4b; wherein
R4a is hydrogen, optionally substituted C1-C20 alkyl, or optionally substituted C1-C20 heteroalkyl;
R4b is optionally substituted C1-C20 alkyl, optionally substituted C2-C20 alkenyl, optionally substituted C2-C20 alkynyl, optionally substituted C1-C20 aminoalkyl, optionally substituted C1-C20 haloalkyl, optionally substituted C1-C20 heteroalkyl, optionally substituted C1-C20 hydroxyalkyl, optionally substituted Cs-Cs cycloalkyl, optionally substituted 4 to 8- membered heterocycloalkyl, optionally substituted phenyl, or optionally substituted 5 to 10- membered heteroaryl; or
R4a and R4b together with the nitrogen to which they are attached form an optionally substituted 3 to 10-membered heterocycloalkyl which is partially or fully saturated; each Rz is independently -CN, -OH, -ORZa, -N3, -NRZaRzb, -CO(O)RZc, -C(O)ORZc, -C(O)NRZaRzb, - NHC(O)RZc, -NHC(O)ORZc, -OC(O)NRZaRzb, or optionally substituted 5 to 10-membered heteroaryl; wherein
RZa and Rzb are each independently hydrogen, alkyl, or PEG;
RZc is alkyl, PEG, cycloalkyl, heterocycloalkyl, or phenyl; and ni and mi are each independently 0 or 1.
26. The molecule of claim 22, or a pharmaceutically acceptable salt thereof, wherein the DNA-binding moiety comprises a structure of Formula (A-10):
Formula (A- 10), wherein:
W1 is hydrogen or -N=C(N(Rle)2)2, wherein each Rle is independently hydrogen or C1-C3 alkyl; each Y5 and Y6 is independently N or CH; each R2a, R2b, R2c, R2d, R2e, R2f, R2g, and R2h is independently hydrogen, optionally substituted C1-C50 alkyl, optionally substituted C2-C50 alkenyl, optionally substituted C2-C50 alkynyl, optionally substituted C1-C50 heteroalkyl, optionally substituted C2-C50 heteroalkenyl, optionally substituted C2-C50 heteroalkynyl, optionally substituted C1-C50 haloalkyl, optionally substituted C3-C10 cycloalkyl, optionally substituted 3 to 10-membered heterocycloalkyl, or optionally substituted PEG1.50; each of which is optionally substituted with one or more Rz;
L1 is C1-C20 alkylene or C2-C20 heteroalkylene;
Z is absent, -C(O)-, or -C(=NH)-;
R4 is C1-C3 alkyl, -OR4b, or -NR4aR4b; wherein
R4a is hydrogen, optionally substituted C1-C20 alkyl, or optionally substituted C1-C20 heteroalkyl;
R4b is optionally substituted C1-C20 alkyl, optionally substituted C2-C20 alkenyl, optionally substituted C2-C20 alkynyl, optionally substituted C1-C20 aminoalkyl, optionally substituted C1-C20 haloalkyl, optionally substituted C1-C20 heteroalkyl, optionally substituted C1-C20 hydroxyalkyl, optionally substituted Cs-Cs cycloalkyl, optionally substituted 4 to 8- membered heterocycloalkyl, optionally substituted phenyl, or optionally substituted 5 to 10- membered heteroaryl; or
R4a and R4b together with the nitrogen to which they are attached form an optionally substituted 3 to 10-membered heterocycloalkyl which is partially or fully saturated; each Rz is independently -CN, -OH, -ORZa, -N3, -NRZaRzb, -CO(O)RZc, -C(O)ORZc, -C(O)NRZaRzb, - NHC(O)RZc, -NHC(O)ORZc, -OC(O)NRZaRzb, or optionally substituted 5 to 10-membered heteroaryl; wherein
RZa and Rzb are each independently hydrogen, alkyl, or PEG;
RZc is alkyl, PEG, cycloalkyl, heterocycloalkyl, or phenyl; and ni and mi are each independently 0 or 1.
27. The molecule of any one of claims 21-26, or a pharmaceutically acceptable salt thereof, wherein each R2a, R2b, R2c, R2d, R2e, R2f, R2g, and R2h is independently hydrogen, optionally substituted C1-C20 alkyl, or optionally substituted C1-C20 haloalkyl, each of which is optionally substituted with one or more Rz.
28. The molecule of any one of claims 21-26, or a pharmaceutically acceptable salt thereof, wherein each R2a, R2b, R2c, R2d, R2e, R2f, R2g, and R2h is independently C1-C20 alkyl.
29. The molecule of claim 27, or a pharmaceutically acceptable salt thereof, wherein each R2a, R2b, R2c, R2d, R2e, R2f, R2g, and R2h is independently methyl, ethyl, or propyl.
30. The molecule of any one of claims 21-28, or a pharmaceutically acceptable salt thereof, wherein L1 is C1-C20 alkylene or C2-C20 heteroalkylene;
Z is absent or -C(O)-; and R4 is -NR4aR4b.
31. The molecule of claim 30, or a pharmaceutically acceptable salt thereof, wherein Z is absent.
32. The molecule of claim 30, or a pharmaceutically acceptable salt thereof, wherein Z is -C(O)-.
33. The molecule of any one of claims 30-32, or a pharmaceutically acceptable salt thereof, wherein L1 is C1-C10 alkylene.
34. The molecule of any one of claims 21-33, or a pharmaceutically acceptable salt thereof, wherein R4a is hydrogen, optionally substituted C1-C20 alkyl, or optionally substituted C1-C20 heteroalkyl; and R4b is optionally substituted C1-C20 alkyl or optionally substituted C1-C20 heteroalkyl.
35. The molecule of claim 34, or a pharmaceutically acceptable salt thereof, wherein R4a is optionally substituted Ci-Ce alkyl; and R4b is optionally substituted Ci-Ce alkyl.
36. The molecule of any one of claims 21-33, or a pharmaceutically acceptable salt thereof, wherein R4a and R4b together with the nitrogen to which they are attached form an optionally substituted 3 to 10- membered heterocycloalkyl.
37. The molecule of any one of claims 1-36, or a pharmaceutically acceptable salt thereof, wherein ni is 1.
38. The molecule of any one of claims 1-36, or a pharmaceutically acceptable salt thereof, wherein ni is 0.
39. The molecule of any one of claims 1-37, or a pharmaceutically acceptable salt thereof, wherein each Y6 is independently CH.
40. The molecule of any one of claims 1-37 or a pharmaceutically acceptable salt thereof, wherein each Y6 is independently N.
41. The molecule of any one of claims 1 -40, or a pharmaceutically acceptable salt thereof, wherein mi is 1.
42. The molecule of any one of claims 1-40, or a pharmaceutically acceptable salt thereof, wherein mi is 0.
43. The molecule of any one of claims 1-42, or a pharmaceutically acceptable salt thereof, wherein each Y5 is independently CH.
44. The molecule of any one of claims 1-42, or a pharmaceutically acceptable salt thereof, wherein each Y5 is independently N.
45. The molecule of any one of claims 1-44, or a pharmaceutically acceptable salt thereof, wherein the first terminus is capable of binding the DNA with an affinity of less than 500 nM.
46. The molecule of any one of claims 1-44, or a pharmaceutically acceptable salt thereof, wherein the oligomeric backbone is a linker having a length of less than about 50 Angstroms.
47. The molecule of any one of claims 1-44, or a pharmaceutically acceptable salt thereof, wherein the oligomeric backbone is a linker having a length of about 10 to 60 Angstroms.
48. The molecule of any one of claims 1-47, or a pharmaceutically acceptable salt thereof, wherein the oligomeric backbone comprises a multimer having 2 to 50 spacing moieties, wherein each spacing moiety is independently selected from the group consisting of -((CRlbRlb)x-O)y-, - ((CRlbRlb)x-NRla)y-, -((CRlbRlb)x-CH=CH-(CRlbRlb)x-O)y-, optionally substituted Ci-Cw alkyl, optionally substituted C2-C10 alkenyl, optionally substituted C2-Cio alkynyl, optionally substituted Ce-Cio arylene, optionally substituted C3-C7 cycloalkylene, optionally substituted 5 to 10- membered heteroarylene, optionally substituted 4 to 10-membered heterocycloalkylene, amino acid residue, -O-, -C(O)NRla-, -NRlaC(O)-, -C(O)-, -NRla-, -C(O)O-, -S-, -S(O)-, -S(O)2-, -S(O)2NRla-, - NRlaS(O)2-, and -P(O)OH-, and any combinations thereof; wherein each x is independently 2-4; each y is independently 1-10; each Rla is independently hydrogen, optionally substituted Ci-Ce alkyl, or optionally substituted Ci-Cg haloalkyl; and each Rlb is independently hydrogen, halogen, optionally substituted Ci-Ce alkyl, or optionally substituted Ci-Ce haloalkyl.
49. The molecule of claim 48, or a pharmaceutically acceptable salt thereof, wherein the oligomeric backbone comprises a multimer having 2 to 50 spacing moieties, wherein each spacing moiety is independently selected from the group consisting of -((CH2)X-O)y-, -((CH2)x-NH)y-, -O-, -C(O)NH-, -NH-, and any combinations thereof.
50. The molecule of claim 48, or a pharmaceutically acceptable salt thereof, wherein the oligomeric backbone comprises a multimer having 2 to 50 spacing moieties selected from -NHC(O)((CH2)X- O)y-, -C(O)NH((CH2)X-O)y-.
51. The molecule of any one of claims 1-50, or a pharmaceutically acceptable salt thereof, wherein the second terminus comprises a bromodomain binding moiety.
52. The molecule of any one of claims 1-50, or a pharmaceutically acceptable salt thereof, wherein the second terminus comprises a moiety capable of binding to a bromodomain and extra terminal domain (BET) family member.
53. The molecule of claim 52, or a pharmaceutically acceptable salt thereof, wherein the BET family member is BRD2, BRD3, BRD4, or BRDT.
54. The molecule of any one of claims 1-50, or a pharmaceutically acceptable salt thereof, wherein the second terminus comprises a moiety capable of binding to CBP/p300, PCAF (P300/CBP-Associated Factor), CECR2 (cat eye syndrome chromosome region candidate 2), BRPF (bromodomain and PHD finger-containing protein), ATAD2/ATAD2B (chromatin remodeling proteins), TRIM24 (Tripartite motif-containing 24), BAZ2 (Bromodomain Adjacent to Zinc finger), TAF1 (TBP associated factors), BRD7/9, BPTF (Bromodomain PHD Finger Transcription Factor), SMARCA2/4, or PBRM1.
55. The molecule of claim 54, or a pharmaceutically acceptable salt thereof, wherein the second terminus comprises a CBP/p300 binding moiety.
56. The molecule of claim 54, or a pharmaceutically acceptable salt thereof, wherein the second terminus comprises a PCAF binding moiety.
57. The molecule of any of claims 1-50, wherein the second terminus comprises the structure of Formula (4-A), or a pharmaceutically acceptable salt thereof:
Formula (4-A), wherein:
Ring D is absent, phenyl, or 5 to 6-membered heteroaryl;
X9 and X10 are each independently C or N, wherein one of X9 or X10 is N;
L2 is absent, optionally substituted alkylene, -O-, or -NRD-, wherein RD is hydrogen or optionally substituted C1-C3 alkyl;
R18 is an optionally substituted 5 to 6-membered heteroaryl;
R19 is an optionally substituted C-G cycloalkyl or optionally substituted 4 to 7-membered heteroaryl; each R20 is independently hydrogen, halogen, -CN, -NO2, optionally substituted Ci-Ce alkyl, optionally substituted Ci-Ce haloalkyl, or optionally substituted Ci-Ce hydroxyalkyl;
X3 is an integer from 1-3; and y4 is an integer from 1-4; wherein the attachment to the linker is at either R19 or at one of R20.
58. The molecule of claim 57, wherein the second terminus comprises the structure of Formula (4-B), or a pharmaceutically acceptable salt thereof:
Formula (4-B), wherein:
Ring D is absent, optionally substituted phenyl, or optionally substituted 5 to 6-membered heteroaryl;
X9 and X10 are each independently C or N, wherein one of X9 or X10 is N;
L2 is absent, optionally substituted alkylene, -O-, or -NRD-, wherein RD is hydrogen or optionally substituted C1-C3 alkyl;
R18 is an optionally substituted 5 to 6-membered heteroaryl;
R19 is an optionally substituted C3-G cycloalkyl or optionally substituted 4 to 7-membered heteroaryl; and x3 is an integer from 1-3.
59. The molecule of claim 57, wherein the second terminus comprises the structure of Formula (4-D), or a pharmaceutically acceptable salt thereof:
Formula (4-D), wherein:
L2 is an optionally substituted alkylene, -O-, or -NRD-, wherein RD is hydrogen or optionally substituted C1-C3 alkyl;
R18 is an optionally substituted 5 to 6-membered heteroaryl;
R20 is hydrogen, halogen, -CN, -NO2, optionally substituted Ci-Ce alkyl, optionally substituted Ci-Ce haloalkyl, or optionally substituted Ci-Ce hydroxyalkyl;
X3 is an integer from 1-3; and y4 is an integer from 1-4.
60. The molecule of any of claims 1-50, wherein the second terminus comprises the structure of
Formula (7-A), wherein:
A3 is -O-, -NH-, or -CH2-;
Z2 is CH or N;
W is O or S; each R31 is independently hydrogen, halogen, -OH, -CN, -NO2, -NH2, optionally substituted C1-C10 alkyl, optionally substituted C1-C10 haloalkyl, optionally substituted C1-C10 hydroxyalkyl, optionally substituted C2-Cio alkenyl, optionally substituted C2-Cio alkynyl, optionally substituted Cs-Cs cycloalkyl, or optionally substituted 3 to 8-membered heterocycloalkyl; or two R31 together with the atoms to which they are attached form an optionally substituted Cs-Cs cycloalkyl or optionally substituted 5 to 8-membered heterocycloalkyl;
R32 is hydrogen or optionally substituted C1-C10 alkyl;
R32a is hydrogen or optionally substituted C1-C10 alkyl;
R33 is hydrogen, halogen, -OH, -CN, -NO2, -NH2, optionally substituted C1-C10 alkyl, optionally substituted C1-C10 haloalkyl, or optionally substituted C1-C10 hydroxyalkyl; and qe is 0-4; wherein Formula (7-A) is attached to the linker is at R32a or at one of R31.
61. The molecule of any one of claims 1-50, wherein the second terminus comprises the structure of Formula (2 -A), or a pharmaceutically acceptable salt thereof:
wherein:
Ring A is an optionally substituted aryl or optionally substituted 5 to 6-membered heteroaryl;
Ring B is absent or an optionally substituted 6-membered monocyclic aryl or heteroaryl;
D is C or N;
E is O or N;
YA is -NH- or -O-;
R5 is hydrogen or Ci-Ce alkyl;
R6 is selected from hydrogen, optionally substituted Ci-Ce alkyl, optionally substituted Ci-Ce haloalkyl, and optionally substituted Ci-Ce hydroxyalkyl;
R7 is selected from hydrogen, halogen, -NO2, -CN, optionally substituted aryl, optionally substituted C1-C20 alkyl, optionally substituted C1-C20 heteroalkyl, optionally substituted Ci-Ce haloalkyl, and optionally substituted Ci-Ce hydroxyalkyl; or R7 is -NR7AR7B, wherein
R7A and R7I; are each independently hydrogen, optionally substituted C1-C20 alkyl, or optionally substituted C1-C20 heteroalkyl; and xi is an integer from 1-6.
62. The molecule of claim 61, wherein the second terminus comprises the structure of Formula (2-B), or a pharmaceutically acceptable salt thereof:
Formula (2-B), wherein:
Ring A is an optionally substituted aryl or optionally substituted 5 to 6-membered heteroaryl;
Ring B is absent or an optionally substituted 6-membered monocyclic aryl or heteroaryl;
YA is -NH- or -O-;
R5 is hydrogen or Ci-Ce alkyl;
R6 is selected from hydrogen, optionally substituted Ci-Ce alkyl, optionally substituted Ci-Ce haloalkyl, and optionally substituted Ci-Ce hydroxyalkyl;
R7 is selected from hydrogen, halogen, -NO2, -CN, optionally substituted aryl, optionally substituted C1-C20 alkyl, optionally substituted C1-C20 heteroalkyl, optionally substituted Ci-Ce haloalkyl, and optionally substituted Ci-Ce hydroxyalkyl; or R7 is -NR7AR7B, wherein
R7A and R7I; are each independently hydrogen, optionally substituted C1-C20 alkyl, or optionally substituted C1-C20 heteroalkyl; and xi is an integer from 1-6.
63. The molecule of claim 61, wherein the second terminus comprises the structure of Formula (2-C), or a pharmaceutically acceptable salt thereof:
Formula (2-C), wherein:
Ring B is absent or an optionally substituted 6-membered monocyclic aryl or heteroaryl;
YA is -NH- or -O-;
R5 is hydrogen or Ci-Ce alkyl;
R6 is selected from hydrogen, optionally substituted Ci-Ce alkyl, optionally substituted Ci-Ce haloalkyl, and optionally substituted Ci-Ce hydroxyalkyl;
R7 is selected from hydrogen, halogen, -NO2, -CN, optionally substituted aryl, optionally substituted C1-C20 alkyl, optionally substituted C1-C20 heteroalkyl, optionally substituted Ci-Ce haloalkyl, and optionally substituted Ci-Ce hydroxyalkyl; or R7 is -NR7AR7B, wherein
R7A and R7I; are each independently hydrogen, optionally substituted C1-C20 alkyl, or optionally substituted C1-C20 heteroalkyl;
R8 and R9 are each independently selected from hydrogen, -C(O)OR8a, optionally substituted Ci-Ce alkyl, optionally substituted Ci-Ce haloalkyl, or optionally substituted Ci-Ce hydroxyalkyl; and R8a is hydrogen, C1-C20 alkyl, or C1-C20 heteroalkyl; and xi is an integer from 1-6.
64. The molecule of claim 61, wherein the second terminus comprises the structure of Formula (2-D), or a pharmaceutically acceptable salt thereof:
Formula (2-D), wherein:
Ring B is absent or an optionally substituted 6-membered monocyclic aryl or heteroaryl;
YA is -NH- or -O-;
R5 is hydrogen or Ci-Ce alkyl;
R6 is selected from hydrogen, optionally substituted Ci-Ce alkyl, optionally substituted Ci-Ce haloalkyl, and optionally substituted Ci-Ce hydroxyalkyl;
R7 is selected from hydrogen, halogen, -NO2, -CN, optionally substituted aryl, optionally substituted C1-C20 alkyl, optionally substituted C1-C20 heteroalkyl, optionally substituted Ci-Ce haloalkyl, and optionally substituted Ci-Ce hydroxyalkyl; or R7 is -NR7AR7B, wherein
R7A and R7I; are each independently hydrogen, optionally substituted C1-C20 alkyl, or optionally substituted C1-C20 heteroalkyl;
R10 is selected from hydrogen, optionally substituted Ci-Ce alkyl, optionally substituted Ci-Ce haloalkyl, or optionally substituted Ci-Cg hydroxyalkyl; and xi is an integer from 1-6.
65. The molecule of any of claims 1-50, wherein the second terminus comprises the structure of
Formula (3-A), wherein:
YB is -CH2NH-, -CH2O-, -NH-, or -O-;
R11A and R11B are each independently hydrogen or optionally substituted Ci-Ce alkyl;
R12 is hydrogen, halogen, -OH, optionally substituted Ci-Ce alkyl, optionally substituted Ci-Ce haloalkyl, or optionally substituted Ci-Ce hydroxyalkyl;
R14 and R15 are each independently hydrogen, halogen, -CN, -NO2, optionally substituted Ci-Ce alkyl, optionally substituted Ci-Ce haloalkyl, or optionally substituted Ci-Ce hydroxyalkyl; or R14 is -NRARB;
R16 is an optionally substituted Ci-Ce alkyl, optionally substituted Ci-Ce heteroalkyl, optionally substituted C2-C6 alkenyl, optionally substituted C2-C6 alkynyl, optionally substituted Ci-Ce hydroxyalkyl, -S(O)(=NH)RA, -SO2RA, or -NHSO2RA;
RYA is hydrogen, optionally substituted Ci-Ce alkyl, optionally substituted Ci-Ce haloalkyl, or optionally substituted 5-6-membered monocyclic aryl or heteroaryl; each RA and RB is independently hydrogen, optionally substituted Ci-Cg alkyl, optionally substituted Ci-Ce heteroalkyl, optionally substituted Ci-Ce haloalkyl, optionally substituted Ci-Ce hydroxyalkyl, optionally substituted C3-C6 cycloalkyl, optionally substituted 4 to 6-membered heterocycloalkyl, or optionally substituted 5 to 6-membered heteroaryl; and
yi is 1-3; wherein the attachment to the linker is at either R14 or at RYA.
66. The molecule of any one of claims 1-50, wherein the second terminus comprises the structure of Formula (3-B), or a pharmaceutically acceptable salt thereof:
Formula (3-B), wherein:
Ring C is absent, optionally substituted 5 to 6-membered monocyclic aryl or heteroaryl or 4 to 8- membered heterocycle;
YB is -NH-, -CH2NH-, -CH2O-, or -O-;
R11A and R11B are each independently hydrogen or optionally substituted Ci-Ce alkyl;
R12 is hydrogen, optionally substituted Ci-Ce alkyl, C(O)RA, or C(O)NRARB; wherein each RA and RB is independently hydrogen, optionally substituted Ci-Ce alkyl, or optionally substituted Ci-Ce heteroalkyl;
R13 is hydrogen, substituted aryl, substituted heteroaryl, or substituted oxydibenzene; and y2 is an integer from 0-2.
67. The molecule of claim 65, wherein the second terminus comprises the structure of Formula (3-C), or a pharmaceutically acceptable salt thereof:
Formula (3-C), wherein:
Ring C is absent, optionally substituted 5 to 6-membered monocyclic aryl or heteroaryl or 4 to 8- membered heterocycle;
YB is -CH2NH-, -CH2O-, -NH-, or -O-;
R11A and R11B are each independently hydrogen or optionally substituted Ci-Cg alkyl;
R12 is hydrogen, halogen, -OH, optionally substituted Ci-Ce alkyl, optionally substituted Ci-Ce haloalkyl, or optionally substituted Ci-Ce hydroxyalkyl;
R14 and R15 are each independently hydrogen, halogen, -CN, -NO2, optionally substituted Ci-Ce alkyl, optionally substituted Ci-Ce haloalkyl, or optionally substituted Ci-Ce hydroxyalkyl;
or R14 is -NRARB;
R16 is optionally substituted Ci-Ce alkyl, optionally substituted Ci-Ce heteroalkyl, optionally substituted C2-C6 alkenyl, optionally substituted C2-C6 alkynyl, optionally substituted Ci-Ce hydroxyalkyl, -S(O)(=NH)RA, -SC>2RA, or -NHSC>2RA; each RA and RB is independently hydrogen, optionally substituted Ci-Ce alkyl, optionally substituted Ci-Ce heteroalkyl, optionally substituted Ci-Ce haloalkyl, optionally substituted Ci-Ce hydroxyalkyl, optionally substituted C3-C6 cycloalkyl, optionally substituted 4 to 6-membered heterocycloalkyl, or optionally substituted 5 to 6-membered heteroaryl; and yi is an integer from 1-3.
68. The molecule of claim 65, wherein the second terminus comprises the structure of Formula (3-D), or a pharmaceutically acceptable salt thereof:
Formula (3-D), wherein:
R11A and R11B are each independently hydrogen or optionally substituted Ci-Ce alkyl;
R12 is hydrogen or an optionally substituted Ci-Ce alkyl; each R15 is independently hydrogen, halogen, -CN, -NO2, optionally substituted Ci-Ce alkyl, optionally substituted Ci-Ce haloalkyl, or optionally substituted Ci-Ce hydroxyalkyl;
R16 is an optionally substituted Ci-Ce alkyl, optionally substituted Ci-Ce heteroalkyl, optionally substituted C2-C6 alkenyl, optionally substituted C2-C6 alkynyl, optionally substituted Ci-Ce hydroxyalkyl, -S(O)(=NH)RA, -SO2RA, or -NHSO2RA;
RA is hydrogen, optionally substituted Ci-Ce alkyl, optionally substituted Ci-Ce heteroalkyl, optionally substituted Ci-Ce haloalkyl, optionally substituted Ci-Ce hydroxyalkyl, optionally substituted C3-C6 cycloalkyl, optionally substituted 4 to 6-membered heterocycloalkyl, or optionally substituted 5 to 6-membered heteroaryl; and yi is an integer from 1-3.
69. The molecule of any of claims 1-50, wherein the second terminus is selected from the group consisting of, or a pharmaceutically acceptable salt thereof:
70. A molecule selected from Table 3, or a pharmaceutically acceptable salt thereof.
71. A pharmaceutical composition comprising the molecule of any one of claims 1-70, or a pharmaceutically acceptable salt thereof and a pharmaceutically acceptable excipient.
72. A method of decreasing expression of a gene in a cell, the method comprising contacting the cell with an effective amount of a molecule of any one of claims 1-70, or a pharmaceutically acceptable salt thereof.
73. The method of claim 72, wherein the gene is HTT.
74. A method of treating Huntington’s disease (HD) in a subject in need thereof, the method comprising administering to the subject a molecule of any one of claims 1-70, or a pharmaceutically acceptable salt thereof.
75. The method of claim 74, wherein the method reduces one or more symptoms of Huntington’s disease.
76. The method of claim 75, wherein the one or more symptoms are selected from chorea, cognitive decline, abnormal libido, abnormal eye movement, abnormal sense of smell, aggression, agitation, anxiety, apathy, bradykinesia, bradyphrenia, clumsiness, delusions, depression, difficulty walking,
disinhibition, dystonia, gait imbalance, muscle weakness, hallucinations, hostility, hypokinesia, irritability, memory impairment, myoclonus, obsessive-compulsive behavior, poor fine motor coordination, seizure, speech articulation difficulties, staring gaze, weight loss, abnormal cholesterol metabolism, abnormal cerebral white matter, alcoholism, Babinski sign, caudate atrophy, cerebral atrophy, choking, clonus, degeneration of the striatum, excessive daytime sleepiness, impaired visuospatial constructive cognition, inability to walk, insomnia, mutism, oral-pharyngeal dysphagia, rigidity, suicidal ideation, cerebellar atrophy, dementia, gate ataxia, gliosis, hyperreflexia, neuronal loss, or personality changes.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US202363587615P | 2023-10-03 | 2023-10-03 | |
| US63/587,615 | 2023-10-03 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2025076219A1 true WO2025076219A1 (en) | 2025-04-10 |
Family
ID=95283894
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2024/049785 Pending WO2025076219A1 (en) | 2023-10-03 | 2024-10-03 | Methods and compounds for modulating huntington's disease |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2025076219A1 (en) |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US9630950B2 (en) * | 2007-04-23 | 2017-04-25 | California Institute Of Technology | Inhibitors for steroid response elements and RNA polymerase II and related methods |
| WO2023014793A1 (en) * | 2021-08-04 | 2023-02-09 | Design Therapeutics, Inc. | Process for polyamide synthesis |
| WO2023133284A2 (en) * | 2022-01-06 | 2023-07-13 | Design Therapeutics, Inc. | Compounds and methods for treating friedreich's ataxia |
-
2024
- 2024-10-03 WO PCT/US2024/049785 patent/WO2025076219A1/en active Pending
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US9630950B2 (en) * | 2007-04-23 | 2017-04-25 | California Institute Of Technology | Inhibitors for steroid response elements and RNA polymerase II and related methods |
| WO2023014793A1 (en) * | 2021-08-04 | 2023-02-09 | Design Therapeutics, Inc. | Process for polyamide synthesis |
| WO2023133284A2 (en) * | 2022-01-06 | 2023-07-13 | Design Therapeutics, Inc. | Compounds and methods for treating friedreich's ataxia |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2019014460A1 (en) | Eaat2 enhancing molecules | |
| WO2023137453A1 (en) | Prodrugs of 3,4-methylenedioxy-n-methcathinone and uses thereof | |
| TW202334148A (en) | Compounds and methods for treating friedreich's ataxia | |
| WO2022060943A1 (en) | Compositions for modulating splicing | |
| EP4377314A1 (en) | Bicyclic pgdh inhibitors and methods of making and using | |
| WO2025076219A1 (en) | Methods and compounds for modulating huntington's disease | |
| WO2023244682A1 (en) | Methods and compounds for modulating inherited genetic diseases | |
| US11420962B2 (en) | Indene derivatives useful in treating pain and inflammation | |
| WO2025076181A1 (en) | Methods and compounds for modulating huntington's disease | |
| WO2025076236A1 (en) | Methods and compounds for modulating huntington's disease | |
| TW202404642A (en) | Methods and compounds for modulating huntington's disease | |
| WO2023215377A1 (en) | Methods of treating neuroinflammatory conditions | |
| JP2025512487A (en) | Novel uses of pyrazolopyrimidine compounds | |
| WO2023137446A1 (en) | Prodrugs of 3,4-methylenedioxy- n-ethyl-amphetamine (mdea) and uses thereof | |
| WO2026015815A1 (en) | Methods of treating haploinsufficiency disorders | |
| WO2026024888A1 (en) | Pgdh inhibitors and methods of making and using | |
| WO2025122762A1 (en) | Compounds and methods for treating sickle cell disease | |
| WO2024158931A1 (en) | Pgdh inhibitors and methods of making and using | |
| WO2024158929A1 (en) | Pgdh inhibitors and methods of making and using | |
| WO2023212237A1 (en) | Compositions useful for modulating splicing | |
| WO2023219939A1 (en) | Compounds and methods for treating friedreich's ataxia |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 24875378 Country of ref document: EP Kind code of ref document: A1 |