WO2025075700A1 - Silicone – (meth)acrylate copolymer composition and emulsion formulation for textile treatment - Google Patents
Silicone – (meth)acrylate copolymer composition and emulsion formulation for textile treatment Download PDFInfo
- Publication number
- WO2025075700A1 WO2025075700A1 PCT/US2024/039475 US2024039475W WO2025075700A1 WO 2025075700 A1 WO2025075700 A1 WO 2025075700A1 US 2024039475 W US2024039475 W US 2024039475W WO 2025075700 A1 WO2025075700 A1 WO 2025075700A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- group
- alternatively
- subscript
- carbon atoms
- surfactant
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F220/00—Copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and only one being terminated by only one carboxyl radical or a salt, anhydride ester, amide, imide or nitrile thereof
- C08F220/02—Monocarboxylic acids having less than ten carbon atoms; Derivatives thereof
- C08F220/10—Esters
- C08F220/12—Esters of monohydric alcohols or phenols
- C08F220/16—Esters of monohydric alcohols or phenols of phenols or of alcohols containing two or more carbon atoms
- C08F220/18—Esters of monohydric alcohols or phenols of phenols or of alcohols containing two or more carbon atoms with acrylic or methacrylic acids
- C08F220/1818—C13or longer chain (meth)acrylate, e.g. stearyl (meth)acrylate
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F2/00—Processes of polymerisation
- C08F2/12—Polymerisation in non-solvents
- C08F2/16—Aqueous medium
- C08F2/22—Emulsion polymerisation
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F2/00—Processes of polymerisation
- C08F2/44—Polymerisation in the presence of compounding ingredients, e.g. plasticisers, dyestuffs, fillers
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F230/00—Copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and containing phosphorus, selenium, tellurium or a metal
- C08F230/04—Copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and containing phosphorus, selenium, tellurium or a metal containing a metal
- C08F230/08—Copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and containing phosphorus, selenium, tellurium or a metal containing a metal containing silicon
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D143/00—Coating compositions based on homopolymers or copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and containing boron, silicon, phosphorus, selenium, tellurium, or a metal; Coating compositions based on derivatives of such polymers
- C09D143/04—Homopolymers or copolymers of monomers containing silicon
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06M—TREATMENT, NOT PROVIDED FOR ELSEWHERE IN CLASS D06, OF FIBRES, THREADS, YARNS, FABRICS, FEATHERS OR FIBROUS GOODS MADE FROM SUCH MATERIALS
- D06M13/00—Treating fibres, threads, yarns, fabrics or fibrous goods made from such materials, with non-macromolecular organic compounds; Such treatment combined with mechanical treatment
- D06M13/10—Treating fibres, threads, yarns, fabrics or fibrous goods made from such materials, with non-macromolecular organic compounds; Such treatment combined with mechanical treatment with compounds containing oxygen
- D06M13/152—Treating fibres, threads, yarns, fabrics or fibrous goods made from such materials, with non-macromolecular organic compounds; Such treatment combined with mechanical treatment with compounds containing oxygen having a hydroxy group bound to a carbon atom of a six-membered aromatic ring
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06M—TREATMENT, NOT PROVIDED FOR ELSEWHERE IN CLASS D06, OF FIBRES, THREADS, YARNS, FABRICS, FEATHERS OR FIBROUS GOODS MADE FROM SUCH MATERIALS
- D06M15/00—Treating fibres, threads, yarns, fabrics, or fibrous goods made from such materials, with macromolecular compounds; Such treatment combined with mechanical treatment
- D06M15/19—Treating fibres, threads, yarns, fabrics, or fibrous goods made from such materials, with macromolecular compounds; Such treatment combined with mechanical treatment with synthetic macromolecular compounds
- D06M15/21—Macromolecular compounds obtained by reactions only involving carbon-to-carbon unsaturated bonds
- D06M15/244—Macromolecular compounds obtained by reactions only involving carbon-to-carbon unsaturated bonds of halogenated hydrocarbons
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06M—TREATMENT, NOT PROVIDED FOR ELSEWHERE IN CLASS D06, OF FIBRES, THREADS, YARNS, FABRICS, FEATHERS OR FIBROUS GOODS MADE FROM SUCH MATERIALS
- D06M15/00—Treating fibres, threads, yarns, fabrics, or fibrous goods made from such materials, with macromolecular compounds; Such treatment combined with mechanical treatment
- D06M15/19—Treating fibres, threads, yarns, fabrics, or fibrous goods made from such materials, with macromolecular compounds; Such treatment combined with mechanical treatment with synthetic macromolecular compounds
- D06M15/21—Macromolecular compounds obtained by reactions only involving carbon-to-carbon unsaturated bonds
- D06M15/263—Macromolecular compounds obtained by reactions only involving carbon-to-carbon unsaturated bonds of unsaturated carboxylic acids; Salts or esters thereof
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06M—TREATMENT, NOT PROVIDED FOR ELSEWHERE IN CLASS D06, OF FIBRES, THREADS, YARNS, FABRICS, FEATHERS OR FIBROUS GOODS MADE FROM SUCH MATERIALS
- D06M15/00—Treating fibres, threads, yarns, fabrics, or fibrous goods made from such materials, with macromolecular compounds; Such treatment combined with mechanical treatment
- D06M15/19—Treating fibres, threads, yarns, fabrics, or fibrous goods made from such materials, with macromolecular compounds; Such treatment combined with mechanical treatment with synthetic macromolecular compounds
- D06M15/21—Macromolecular compounds obtained by reactions only involving carbon-to-carbon unsaturated bonds
- D06M15/263—Macromolecular compounds obtained by reactions only involving carbon-to-carbon unsaturated bonds of unsaturated carboxylic acids; Salts or esters thereof
- D06M15/27—Macromolecular compounds obtained by reactions only involving carbon-to-carbon unsaturated bonds of unsaturated carboxylic acids; Salts or esters thereof of alkylpolyalkylene glycol esters of unsaturated carboxylic acids
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06M—TREATMENT, NOT PROVIDED FOR ELSEWHERE IN CLASS D06, OF FIBRES, THREADS, YARNS, FABRICS, FEATHERS OR FIBROUS GOODS MADE FROM SUCH MATERIALS
- D06M15/00—Treating fibres, threads, yarns, fabrics, or fibrous goods made from such materials, with macromolecular compounds; Such treatment combined with mechanical treatment
- D06M15/19—Treating fibres, threads, yarns, fabrics, or fibrous goods made from such materials, with macromolecular compounds; Such treatment combined with mechanical treatment with synthetic macromolecular compounds
- D06M15/21—Macromolecular compounds obtained by reactions only involving carbon-to-carbon unsaturated bonds
- D06M15/327—Macromolecular compounds obtained by reactions only involving carbon-to-carbon unsaturated bonds of unsaturated alcohols or esters thereof
- D06M15/333—Macromolecular compounds obtained by reactions only involving carbon-to-carbon unsaturated bonds of unsaturated alcohols or esters thereof of vinyl acetate; Polyvinylalcohol
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06M—TREATMENT, NOT PROVIDED FOR ELSEWHERE IN CLASS D06, OF FIBRES, THREADS, YARNS, FABRICS, FEATHERS OR FIBROUS GOODS MADE FROM SUCH MATERIALS
- D06M15/00—Treating fibres, threads, yarns, fabrics, or fibrous goods made from such materials, with macromolecular compounds; Such treatment combined with mechanical treatment
- D06M15/19—Treating fibres, threads, yarns, fabrics, or fibrous goods made from such materials, with macromolecular compounds; Such treatment combined with mechanical treatment with synthetic macromolecular compounds
- D06M15/21—Macromolecular compounds obtained by reactions only involving carbon-to-carbon unsaturated bonds
- D06M15/356—Macromolecular compounds obtained by reactions only involving carbon-to-carbon unsaturated bonds of other unsaturated compounds containing nitrogen, sulfur, silicon or phosphorus atoms
- D06M15/3568—Macromolecular compounds obtained by reactions only involving carbon-to-carbon unsaturated bonds of other unsaturated compounds containing nitrogen, sulfur, silicon or phosphorus atoms containing silicon
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06M—TREATMENT, NOT PROVIDED FOR ELSEWHERE IN CLASS D06, OF FIBRES, THREADS, YARNS, FABRICS, FEATHERS OR FIBROUS GOODS MADE FROM SUCH MATERIALS
- D06M2200/00—Functionality of the treatment composition and/or properties imparted to the textile material
- D06M2200/10—Repellency against liquids
- D06M2200/12—Hydrophobic properties
Definitions
- a composition includes a silicone - (meth)acrylate copolymer, a manganese ion source, and a phenolic compound; and methods for preparation and use of said composition are provided.
- the composition is useful in an emulsion formulation for treating textiles to provide the textiles with durable water repellency.
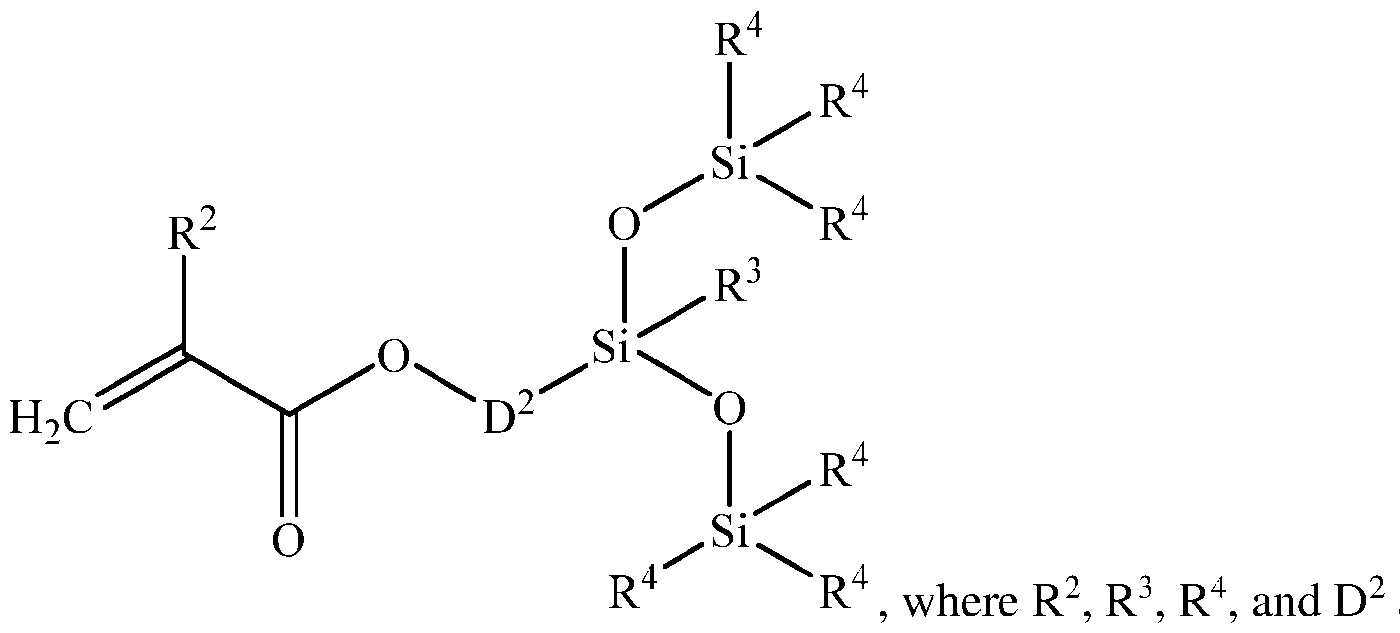
- each D 2 is a divalent hydrocarbon group of 2 to 12 carbon atoms.
- D 2 may have 2 to 10, alternatively 3 to 5, and alternatively 3 carbon atoms.
- Each D 3 and each D is independently a divalent hydrocarbon group of 1 to 12 carbon atoms.
- each D 3 and each D may be an alkylene group; alternatively ethylene.
- D 4 and D 1 are each independently an alkylene group of 2 to 4 carbon atoms or a divalent alkylarylene group.
- each R 8 is a crosslinkable group.
- Each R 8 may be independently selected from the group consisting of hydroxy, amino, epoxy, ureido, and acetoxy.
- each R 8 may be independently selected from the group consisting of hydroxy and ureido, and alternatively each R 8 may be hydroxy.
- each R 5 is an oxygen atom.
- Each R 6 is selected from the group consisting of -OH and alkoxy. Alternatively, the alkoxy group for R 6 may be -OCH3.
- each R 9 is a monovalent hydrocarbon group, which is free of aliphatic unsaturation and which may be linear, branched, or cyclic (i.e., monocyclic or polycyclic), or combinations thereof.
- R 9 may be an alkyl group or an aryl group, which may be monocyclic or polycyclic, and which may optionally have linear or branched groups.
- suitable alkyl groups for R 9 may include methyl, t-amyl, butyl (including t-butyl), cyclohexyl, iso-decyl, isobornyl, and 2-ethylhexyl.
- suitable aryl groups include phenyl, naphthyl, anthracyl, and benzyl.
- R 10 may be a halide, an acetate, or a monovalent hydrocarbon group, as described above for R 9 .
- the halide may be bromide (Br), chloride (Cl), fluoride (F) or iodide (I); alternatively Br, Cl or F; alternatively Br or Cl; and alternatively Cl.
- Subscript zl may be 0.
- subscript zl may be at least 0.5, alternatively at least 1, and alternatively at least 2; while at the same time, subscript zl may be up to 18, alternatively up to 15, alternatively up to 10, alternatively up to 8, and alternatively up to 5.
- subscript zl may be 0 to 18, alternatively > 0 to 18, alternatively 0.5 to 7, alternatively 1 to 6, and alternatively 2 to 5.
- Subscript z2 may be 0.
- subscript z2 may be at least 0.5, alternatively at least 1 , and alternatively at least 2; while at the same time, subscript z2 may be up to 8, alternatively up to 7, alternatively up to 6, alternatively up to 5, and alternatively up to 4.
- subscript z2 may be 0 to 8, alternatively > 0 to 8, alternatively 0.5 to 7, alternatively 1 to 6, and alternatively 2 to 5.
- the copolymer may be prepared by any convenient means such as that disclosed in PCT Patent Application Publication W02023/019044 to Jeletic et al. by using the manganese ion source and the phenolic compound described herein as inhibitors, instead of the inhibitor described therein.
- the copolymer may be prepared as described in U.S. Patent 10,047,199 to limura et al. by varying appropriate starting materials and their amounts and by including the manganese ion source and the phenolic compound described herein.
- PCT Patent Application Publication W02023/019044 and U.S. Patent 10,047,199 are hereby incorporated by reference.
- the starting materials used in the process to prepare the composition may optionally further comprise (H) a chain transfer agent; (J) an additional non- crystallizable monomer that is distinct from each of (A), (B) and (C); and a combination of both (H) and (J).
- step (1) copolymerizing starting materials (A), (B), (C), (F), and when present (H) and/or (J), in the presence of the (K) manganese ion source and (L) the phenolic compound, may be performed in a process such as that disclosed in U.S. Patent 10,047,199 to limura et al. by varying appropriate starting materials and their amounts.
- the resulting reaction product which comprises the copolymer, (K) the manganese ion source, and (L) the phenolic compound, may be solvent borne. When solvent is present, all or a portion of the solvent may be removed by any convenient means, such as by stripping or distillation with optionally with heat and/or optionally reduced pressure.
- the emulsion polymerization process described above may comprise forming an emulsion comprising starting materials (A), (B), (C), (D), (E), (K), and (L) (and optionally (H) and/or (J)), and thereafter adding (F) the initiator and copolymerizing.
- starting materials (K) and (L) may inhibit formation of acrylic radicals that can impact the formation of the copolymer during copolymerization in step (1).
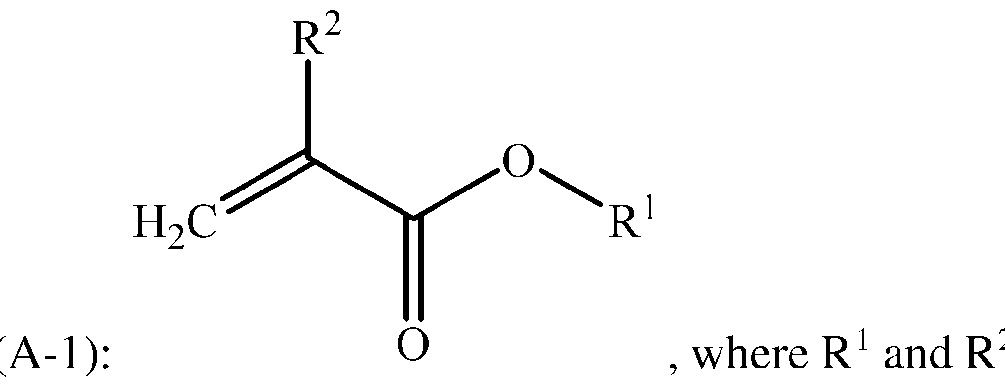
- Starting material (A) used to prepare the copolymer is a crystallizablc monomer of formula ( 2 are as described above.
- Examples of crystallizablc monomers for starting material (A) include stcaryl (mcth)acrylatc, eicosyl (meth)acrylate, behenyl (meth)acrylate, and combinations thereof.
- R 2 is hydrogen
- starting material (A) may be an acrylate selected from stearyl acrylate, eicosyl acrylate, behenyl acrylate, and combinations thereof.
- starting material (A) may be stearyl acrylate.
- Crystallizable monomers suitable for starting material (A) are commercially available, e.g., from Millipore Sigma of St. Louis, Missouri, USA and from BASF SE of Ludwigshafen, Germany. Crystallizable means that the stalling monomer has a melting point ) > 25 °C ⁇ 5 °C.
- starting material (B) may have a formula selected from the group consisting of: 4 are as described above.
- Stalling material (D) is a surfactant.
- the surfactant used in copolymerizing in step (1) of the process for making the aqueous emulsion is not specifically restricted and may be selected from the group consisting of anionic surfactants, amphoteric surfactants, cationic surfactants, nonionic surfactants, and combinations of two or more thereof.
- the anionic surfactants include (i) sulfonic acids and their salt derivatives, including alkyl, aralkyl, alkyl naphthalene, alkyl diphenyl ether sulfonic acids, and their salts, having at least 6 carbon atoms in the alkyl substituent, such as dodecyl benzene sulfonic acid, and its sodium salt or its amine salt; (ii) alkyl sulfates having at least 6 carbon atoms in the alkyl substituent, such as sodium lauryl sulfate; (iii) the sulfate esters of polyoxyethylene monoalkyl ethers; (iv) long chain carboxylic acid surfactants, such as lauric acid, steric acid, oleic acid, and their alkali metal and amine salts.
- sulfonic acids and their salt derivatives including alkyl, aralkyl, alkyl naphthalene, alkyl
- compositions of the latter type such as dodecyl benzene sulfonic acid, although a catalyst as noted above, can also function as the anionic surfactant when neutralized.
- suitable surfactants include sodium alkyl sulfonate such as HOSTAPURTM SAS-30, and triethanolamine dodecyl benzene sulfonate, such as BIO-SOFTTM N 300.
- Suitable amphoteric surfactants include amino acid surfactants, betaines (e.g., lauryl betaine, bis-(2-hydroxyethyl) tallow betaine, cocamidopropylbetaine, N-alkylamidobetaines, and derivatives thereof), proteins and derivatives thereof, glycinates (glycine derivatives, such as cocamphoglycinate, cocamphocarboxy-glycinates, and cocamphodipropionate), sultaines (e.g., cocamidopropylhydroxysultaine and lauryl sultaine), alkyl aminopropionates, alkyl polyaminocarboxylates and alkylamphoacetates, lecithin and hydrogenated lecithin, and combinations thereof.
- surfactants are commercially available from various suppliers under different tradenames. For example, REWOTERICTM AM TEG is produced by Evonik of Essen, Germany; AMPHOSOLTM CG is available from Step
- Cationic surfactants useful herein include compounds containing quaternary ammonium hydrophilic moieties in the molecule which are positively charged, such as quaternary ammonium salts, which may be represented by formula (D-2-1): R 12 R 13 R 14 R 15 N + X’" where R 12 to R 15 are alkyl groups containing 1-30 carbon atoms, or alkyl groups derived from tallow, coconut oil, or soy; and X’ is a halogen, e.g., chlorine or bromine.
- quaternary ammonium salts which may be represented by formula (D-2-1): R 12 R 13 R 14 R 15 N + X’" where R 12 to R 15 are alkyl groups containing 1-30 carbon atoms, or alkyl groups derived from tallow, coconut oil, or soy; and X’ is a halogen, e.g., chlorine or bromine.
- Representative quaternary ammonium halide salts are dodecyltrimethyl ammonium chloride/lauryltrimethyl ammonium chloride (LTAC), cetyltrimethyl ammonium chloride (CTAC), hexadeclyltrimethyl ammonium chloride, didodecyldimethyl ammonium bromide, dihexadecyldimethyl ammonium chloride, dihexadecyldimethyl ammonium bromide, dioctadecyldimethyl ammonium chloride, dieicosyldimethyl ammonium chloride, and didocosyldimethyl ammonium chloride.
- LTAC dodecyltrimethyl ammonium chloride/lauryltrimethyl ammonium chloride
- CTAC cetyltrimethyl ammonium chloride
- hexadeclyltrimethyl ammonium chloride didodecyldimethyl ammonium bromide
- the surfactant used in copolymerizing in step (1) may be (D-3) a nonionic surfactant.
- suitable nonionic surfactants which can be used include polyoxyalkylene copolymers, such as polyoxyethylene alkyl ethers, polyoxyethylene alkylphenol ethers, alkyl glycosides (e.g., alkylglucosides), polyoxyethylene fatty acid esters, polyoxyethylene lauryl ethers, sorbitan fatty acid esters, polyoxyethylene sorbitan fatty acid esters, polyoxyethylene sorbitan monooleates, polyoxyethylene alkyl esters, polyoxyethylene sorbitan alkyl esters, polyethylene glycol (such as polyethylene glycol having 23 cthylcnc-oxidc units), polypropylene glycol, diethylene glycol, ethoxylated trimethylnonanols, tristyrylphenol ethers (TSP’s), distyryl phenol
- alkyl-oxo alcohol polyglycol ethers such as GENAPOLTM UD 050, and GENAPOLTM UDI 10
- alkyl polyethylene glycol ether based on ClO-Guerbet alcohol and ethylene oxide such as LUTENSOLTM XP 79
- other alcohol ethoxylates for example, C11-15 alkyl alcohol ethoxylate with tradename ECOSURFTM EH, such as ECOSURFTM EH-40, from TDCC.
- TSPs and DSP’s are commercially available from Stepan.
- Suitable nonionic surfactants also include poly(oxyethylene)-poly(oxypropylene)- poly(oxyethylene) tri-block copolymers.
- Poly(oxyethylene)-poly(oxypropylene)- poly(oxyethylene) tri-block copolymers are also commonly known as Poloxamers. They are nonionic triblock copolymers composed of a central hydrophobic chain of polyoxypropylene (polypropylene oxide)) flanked by two hydrophilic chains of polyoxyethylene (poly(ethylene oxide)).
- Poly(oxyethylene)-poly(oxypropylene)-poly(oxyethylene) tri-block copolymers are commercially available from BASF of Florham Park, New Jersey, USA, and are sold under the tradename PLURONICTM, such as PLURONICTM L61, L62, L64, L81, P84.
- the amount of nonionic surfactant may be at least 0.1%, alternatively at least 0.2%, alternatively at least 0.3%, alternatively at least 0.4%, alternatively at least 0.5%, alternatively at least 1%; while at the same time the amount of nonionic surfactant may be up to 10%, alternatively up to 5%, alternatively up to 4%, alternatively up to 3%, alternatively up to 2%, alternatively up to 1%, on the same basis.
- the amount of nonionic surfactant may be 0.2% to 4%, alternatively 0.3% to 3%, alternatively 0.4 to 2.5%, and alternatively 1% to 2%; on the same basis.
- Starling material (E) is water.
- the water is not generally limited, for example, the water may be processed or unprocessed. Examples of processes that may be used for purifying the water include distilling, filtering, deionizing, and combinations of two or more thereof, such that the water may be deionized, distilled, and/or filtered. Alternatively, the water may be unprocessed (e.g. may be tap water, i.e., provided by a municipal water system or well water, used without further purification). The amount of water is sufficient to form an aqueous emulsion for copolymerizing in step (1) of the process described above.
- the peroxide compound may be a peroxide or a hydroperoxide, such as t-butylperoctoate, t-butyl perbenzoate, dicumyl peroxide, di-t-butyl peroxide, t-butyl hydroperoxide, cumene hydroperoxide, di-t-amyl peroxide and combinations of two or more thereof.
- di-peroxide initiators may be used alone or in combination with other initiators.
- the additional monomer may be present in an amount of at least 0.5%, alternatively at least 1%, and alternatively at least 2%; while at the same time the additional monomer may be present in an amount up to 18%, alternatively up to 15%, alternatively up to 10%, alternatively up to 8%, and alternatively up to 5%, on the same basis.
- the amount of (H) the additional monomer may be > 0 to 18%, alternatively 0.5% to 7%, alternatively 1% to 6%, and alternatively 2% to 5%, on the same basis.
- Starting material (J) is an optional reactive surfactant that may be used to facilitate emulsion copolymerization, and that may react to form part of the copolymer.
- the reactive surfactant may have formula:
- a reactive surfactant of formula subscript vl has a value sufficient to give the reactive surfactant a number average molecular weight of 300 g/mol to 950 g/mol is commercially available from Sigma- Aldrich, Inc. of St. Louis, Missouri, USA.
- the reactive surfactant is optional and may be added in an amount of 0 to 5 weight % based on combined weights of starting materials (A), (B), and (C), and when present (H), and (J).
- Reactive surfactants are commercially available, e.g., from Ethox.
- the reactive surfactant may be added in an amount of > 0 to 2 weight % on the same basis.
- the amount of the manganese ion source may be > 0 ppm, alternatively at least 0.5 ppm, alternatively at least 1 ppm, alternatively at least 1 .5; while at the same time, the amount of manganese ion source may be up to 10 ppm, alternatively up to 5 ppm, alternatively up to 4 ppm, and alternatively up to 3 ppm, and alternatively up to 2 ppm, based on combined weights of all starting materials in the emulsion formulation for treating the textile.
- Mixing under shear may be performed by any convenient means for forming an aqueous emulsion formulation, such as sonication and with subsequent microfluidization.
- Equipment for mixing under shear such as sonicators, homogenizers, microfluidizers, and speedmixers are known in the art and are commercially available.
- each D 5 is an independently selected alkylene oxide group of 2 or more carbon atoms.
- R 19 may be, for example, 2-ethylhexyl, lauryl, or other alkyl group of 8 to 15 carbon atoms.
- subscript n may be 23 to 40, alternatively 23 to 30.
- each D 5 may be independently selected from the group consisting of ethylene oxide and propylene oxide.
- each D 5 may be ethylene oxide.
- This process for preparing the emulsion formulation suitable for treating the textile may optionally further comprise adding a further additional starting material.
- the further additional starting material may be selected from the group consisting of (V) a wax, (VI) a biocide, (VII) additional water (which may be the same as starting material (E)), (VIII) a flame retardant, (IX) a wrinkle reducing agent, (X) an antistatic agent, (XI) a penetrating agent and a combination of two or more of (V) to (XI).
- the aqueous emulsion prepared in the process comprising step (1), described above may be used to prepare the emulsion formulation suitable for treating the textile.
- the process described above comprising step (1) further comprises step (2): adding starling material (IV), the water dispersible crosslinker, to the aqueous emulsion.
- This process for preparing the emulsion formulation suitable for treating the textile may optionally further comprise, after step (1), adding the further additional starting material.
- crosslinker (IV) examples include U.S. Patent Application 2017/0204558 to Knaup; U.S. Patent 9,777,105 to Hamajima et al., beginning at col. 11, line 54, which are hereby incorporated by reference for the purpose of describing suitable crosslinkers.
- the exact amount of crosslinker (IV) depends on various factors including the type and amount of (I) silicone - (meth)acrylate copolymer and the textile to be treated, however, the weight of the crosslinker (IV) may 0.25% to 3.75% on fabric weight, alternatively 0.25% to 1%, and alternatively 0.25% to 0.5%, on the same basis.
- Starting material (V) is a wax, which may optionally be added to provide improved water repellency or softness to the textile to which the emulsion formulation will be applied.
- the amount of wax will vary depending on factors including the type of wax selected, the benefit desired, and the fabric to be treated with the emulsion formulation. However, the amount of wax may be 0 to 75%, alternatively 0 to 50%, alternatively 25% to 50% based on weight of (I) the silicone - (meth)acrylate copolymer. Alternatively, when used, the amount of wax may be > 0%, alternatively at least 10%, and alternatively at least 25%, while at the same time the amount of wax may be up to 75%, alternatively up to 50% on the same basis.
- suitable waxes include paraffin waxes (e.g., n-paraffins, iso-paraffins, and/or cycloparaffins), silicone waxes such as silicone wax with long chain alkyl groups (e.g., alkyl methyl silicone wax) and/or amino-silicone wax, and a combination of two or more thereof.
- Suitable waxes are disclosed, for example, in U.S. Patent Application 2017/0204558 to Knaup and U.S. Patent 10,844,151 to Probst, et al.
- Waxes may be delivered as water-based dispersions, for example Michelman wax 743 and others from Michelman of Cincinnati, Ohio, U.S.A.
- Other waxes are also commercially available, for example, from Sasol Wax of Hamburg, Germany, and silicone waxes, such as DOWSILTM AMS-C30, are available from Dow Silicones Corporation of Midland, Michigan, U.S.A.
- the Bundesmann test may be carried out using the ISO 9865 method.
- An SDL Atlas M230 Bundesmann test apparatus may be used to collect face rating data after 0, 1, 10, and 20 washes. The instrument may be calibrated so that each cup collected between 190 and 210mL after 150s. Face Ratings may be assigned based on the images in the ISO 9865 method using half increments as intermediate assignments. Face Ratings may be repeated and reported as averages. A passing value may be > 4.5 to 5.
- any ranges and subranges relied upon in describing the present invention independently and collectively fall within the scope of the appended claims, and are understood to describe and contemplate all ranges including whole and/or fractional values therein, even if such values are not expressly written herein.
- One of skill in the art readily recognizes that the enumerated ranges and subranges sufficiently describe and enable various embodiments of the present invention, and such ranges and subranges may be further delineated into relevant halves, thirds, quarters, fifths, and any other subrange subsumed within the range.
Landscapes
- Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Engineering & Computer Science (AREA)
- Textile Engineering (AREA)
- Organic Chemistry (AREA)
- Medicinal Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Polymers & Plastics (AREA)
- Wood Science & Technology (AREA)
- Materials Engineering (AREA)
- Life Sciences & Earth Sciences (AREA)
- Addition Polymer Or Copolymer, Post-Treatments, Or Chemical Modifications (AREA)
- Treatments For Attaching Organic Compounds To Fibrous Goods (AREA)
- Compositions Of Macromolecular Compounds (AREA)
Abstract
A composition includes a silicone – (meth)acrylate copolymer, a manganese ion source, and a phenolic compound. The composition may be used with additional starting materials including a surfactant, water, and a water dispersible crosslinker in an emulsion formulation. The emulsion formulation may be used for treating a textile. Methods for preparation of the composition and the emulsion formulation are provided.
Description
SILICONE - (METH) ACRYLATE COPOLYMER COMPOSITION AND EMULSION
FORMULATION FOR TEXTILE TREATMENT
CROSS REFERENCE TO RELATED APPLICATIONS
[0001] This application claims the benefit of U.S. Provisional Patent Application Serial No. 63/588050 filed on 5 October 2023 under 35 U.S.C. § 119 (e). U.S. Provisional Patent Application Serial No. 63/588050 is hereby incorporated by reference.
TECHNICAL FIELD
[0002] A composition includes a silicone - (meth)acrylate copolymer, a manganese ion source, and a phenolic compound; and methods for preparation and use of said composition are provided. The composition is useful in an emulsion formulation for treating textiles to provide the textiles with durable water repellency.
BACKGROUND
[0003] In water repellent textile treatment applications, fluorocarbon materials have dominated the market due to their ability to provide excellent durable water repellency. However, regulatory and customer pressures are contributing to an industry need for nonfluorocarbon based textile treatments. Previously disclosed fluorocarbon-free textile treatments suffer the drawback of providing poor durability, where water repellency of textiles treated therewith decreases significantly after multiple washings.
SUMMARY
[0004] A composition comprising a silicone - (meth) acrylate copolymer, a manganese ion source, and a phenolic compound; and a method for preparing the composition, are described. An emulsion formulation comprising the silicone - (meth)acrylate copolymer, the manganese ion source, the phenolic compound, a surfactant, and water; and methods for preparing and using the emulsion formulation, are also described. The emulsion formulation is suitable for treating a textile.
DETAILED DESCRIPTION
[0005] As introduced above, starting material (I) in the composition, and in the emulsion formulation, is the silicone - (meth) acrylate copolymer (copolymer). The copolymer comprises unit formula:
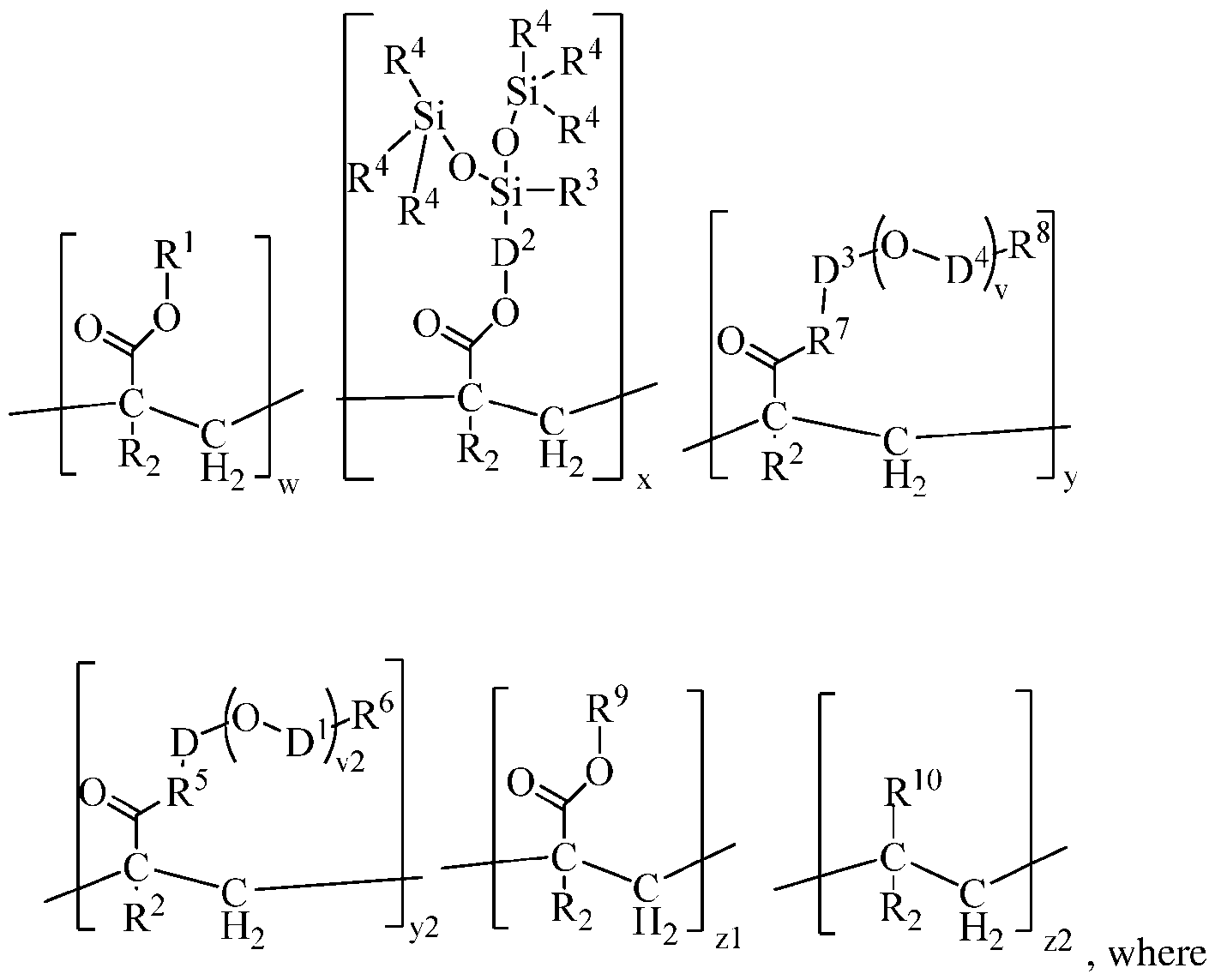
independently selected alkyl group of 16 to 24 carbon atoms; each R2 is independently selected from the group consisting of H and methyl; each D2 is a divalent hydrocarbon group of 2 to 12 carbon atoms; each R3 is independently selected from the group consisting of R4 and a group of formula -OSi(R4)s; each R4 is an independently selected monovalent hydrocarbon group of 1 to 12 carbon atoms; each R7 is independently selected from the group consisting of an oxygen atom and NH; D3 is a divalent hydrocarbon group of 1 to 12 carbon atoms; D4 is an alkylene group of 2 to 4 carbon atoms or a divalent alkylarylene group; subscript v represents the number of units of formula (OD4) in the unit with subscript y, and subscript v has a value of 0 to 12; each R8 is a crosslinkable group; each R5 is independently selected from the group consisting of an oxygen atom and NH; D is a divalent hydrocarbon group of 1 to 12 carbon atoms; each D1 is an alkylene group of 2 to 4 carbon atoms or a divalent alkylarylene group; subscript v2 represents the number of units of formula (OD1) in the unit with subscript y2, and subscript v2 has a value of 0 to 20; each R6 is independently selected from the group consisting of a hydroxyl group and an alkoxy group (e.g., methoxy); each R9 is a monovalent hydrocarbon group of 1 to 14 carbon atoms; each R10 is independently selected from the group consisting of a halogen (e.g., chloride), an acetate group, or a monovalent hydrocarbon group of 1 to 14 carbon atoms; subscripts w, x, y, y2, zl, and z2 represent relative weights of each unit in the copolymer, subscript w has a value of
80 to 98; subscript x has a value of 1 to 15; subscript y has a value of 1 to 5; subscript y2 has a value of 0 to 5; subscript zl has a value of 0 to 18; and subscript z2 has a value of 0 to 18, and a quantity (w + x + y + y2 + zl + z2) = 100. The copolymer further comprises a terminal moiety. [0006] In the unit formula above, R1 has 16 to 24 carbon atoms. Alternatively, R1 may have at least 16, alternatively at least 17, alternatively at least 18 carbon atoms, while at the same time R1 may have up to 24, alternatively up to 23, and alternatively up to 22 carbon atoms. Alternatively, R1 may have 17 to 23, carbon atoms, alternatively 16 to 22 carbon atoms, alternatively 17 to 24 carbon atoms, and alternatively 18 to 22 carbon atoms. R1 may be selected from the group consisting of stearyl, eicosyl, and behenyl. Alternatively, R1 may be stearyl. [0007] In the unit formula above, the unit with subscript x is a silicone - (meth)acrylate macromonomer unit. In the silicone - methacrylate macromonomer unit, each R3 is independently selected from the group consisting of R4 and a group of formula -OSi(R4)3; where each R4 is an independently selected monovalent hydrocarbon group of 1 to 12 carbon atoms. In the silicone - (meth)acrylate macromonomer unit, the monovalent hydrocarbon group for R4 may be an alkyl group, such as an alkyl group of 1 to 6 carbon atoms. Alternatively, the alkyl groups may have 1 to 3 carbon atoms, alternatively 1 to 2 carbon atoms. Alternatively, each R4 may be methyl.
[0008] In the unit formula above, each D2 is a divalent hydrocarbon group of 2 to 12 carbon atoms. Alternatively, D2 may have 2 to 10, alternatively 3 to 5, and alternatively 3 carbon atoms. Each D3 and each D is independently a divalent hydrocarbon group of 1 to 12 carbon atoms. Alternatively, each D3 and each D may be an alkylene group; alternatively ethylene. D4 and D1 are each independently an alkylene group of 2 to 4 carbon atoms or a divalent alkylarylene group.
[0009] The divalent hydrocarbon group for D4 and D1 may be exemplified by an alkylene group such as ethylene, propylene, or butylene; an arylene group such as phenylene, or an alkylarylene group such as:
each subscript u is independently 1 to 6, alternatively 1 to 2. Alternatively, the divalent hydrocarbon group may be alkylene, and alternatively the divalent hydrocarbon group may be ethylene. The divalent hydrocarbon group for D, D2 and D3 may be as described above, and alternatively may be methylene. Alternatively, D2 may be methylene, ethylene or propylene; alternatively propylene. Alternatively, D2 may be linear, e.g., -(Cth - or -(Cth -.
[0010] In the unit formula above, each R7 is independently selected from the group consisting of an oxygen atom and NH. Alternatively, each R7 may be oxygen.
[0011] In the unit formula above, each R8 is a crosslinkable group. Each R8 may be independently selected from the group consisting of hydroxy, amino, epoxy, ureido, and acetoxy. Alternatively, each R8 may be independently selected from the group consisting of hydroxy and ureido, and alternatively each R8 may be hydroxy.
[0012] In the unit formula above each R5 is an oxygen atom. Each R6 is selected from the group consisting of -OH and alkoxy. Alternatively, the alkoxy group for R6 may be -OCH3.
[0013] In the unit formula above, each R9 is a monovalent hydrocarbon group, which is free of aliphatic unsaturation and which may be linear, branched, or cyclic (i.e., monocyclic or polycyclic), or combinations thereof. R9 may be an alkyl group or an aryl group, which may be monocyclic or polycyclic, and which may optionally have linear or branched groups. Examples of suitable alkyl groups for R9 may include methyl, t-amyl, butyl (including t-butyl), cyclohexyl, iso-decyl, isobornyl, and 2-ethylhexyl. Examples of suitable aryl groups include phenyl, naphthyl, anthracyl, and benzyl.
[0014] In the unit formula above, R10 may be a halide, an acetate, or a monovalent hydrocarbon group, as described above for R9. The halide may be bromide (Br), chloride (Cl), fluoride (F) or iodide (I); alternatively Br, Cl or F; alternatively Br or Cl; and alternatively Cl.
[0015] In the unit formula above, subscripts w, x, y, y2, zl, and z2 are relative weights of each unit, and a quantity (w + x + y + y2 + zl + z2) may total 100. Subscript w has a value of 80 to 98. Alternatively, subscript w may be at least 80, alternatively at least 81, alternatively at least
82, alternatively at least 83, alternatively at least 84, alternatively at least 85, alternatively at least 86, alternatively at least 87, and alternatively at least 88; while at the same time, subscript w is up to 98, alternatively up to 97, alternatively up to 96, alternatively up to 95, alternatively up to 94, alternatively up to 93, alternatively up to 92, alternatively up to 91, alternatively up to 90, alternatively up to 89, and alternatively up to 88. Alternatively, subscript w may be 84 to 92, alternatively 85 to 91, alternatively 86 to 90, alternatively 87 to 89, and alternatively 88.
[0016] Subscript x has a value of 1 to 15, alternatively 5 to 15. Alternatively, subscript x may be at least 1, alternatively at least 5, alternatively at least 6, alternatively at least 7, alternatively at least 8, alternatively at least 9, alternatively at least 10; while at the same time, subscript x may be up to 15, alternatively up to 14, alternatively up to 13, alternatively up to 12, alternatively up to 11, and alternatively up to 10. Alternatively, subscript x may be 1 to 14, alternatively 2 to 13, alternatively 3 to 12, alternatively 4 to 11, alternatively 5 to 10, alternatively 7 to 13; and alternatively 10.
[0017] Subscript y has a value of 1 to 5. Alternatively, subscript y may be at least 1, alternatively at least 1.25, alternatively at least 1.5, alternatively at least 1.75, and alternatively at least 2; while at the same time, subscript y may be up to 5, alternatively up to 4, alternatively up to 3, alternatively up to 2.75, alternatively up to 2.5, and alternatively up to 2.25. Alternatively, subscript y may be 1 to 3, alternatively 1 to 2, alternatively 1.5 to 2.5, alternatively 1.75 to 2.25, and alternatively 2.
[0018] Subscript y2 has a value of 0 to 5. Alternatively, subscript y2 may be at least 1, alternatively at least 1.25, alternatively at least 1.5, alternatively at least 1.75, and alternatively at least 2; while at the same time, subscript y2 may be up to 5, alternatively up to 4, alternatively up to 3, alternatively up to 2.75, alternatively up to 2.5, and alternatively up to 2.25. Alternatively, subscript y2 may be 0 to 3, alternatively 1 to 2, alternatively 1.5 to 2.5, alternatively 1.75 to 2.25, and alternatively 2.
[0019] Subscript zl may be 0. Alternatively, subscript zl may be at least 0.5, alternatively at least 1, and alternatively at least 2; while at the same time, subscript zl may be up to 18, alternatively up to 15, alternatively up to 10, alternatively up to 8, and alternatively up to 5. Alternatively, subscript zl may be 0 to 18, alternatively > 0 to 18, alternatively 0.5 to 7, alternatively 1 to 6, and alternatively 2 to 5.
[0020] Subscript z2 may be 0. Alternatively, subscript z2 may be at least 0.5, alternatively at
least 1 , and alternatively at least 2; while at the same time, subscript z2 may be up to 8, alternatively up to 7, alternatively up to 6, alternatively up to 5, and alternatively up to 4. Alternatively, subscript z2 may be 0 to 8, alternatively > 0 to 8, alternatively 0.5 to 7, alternatively 1 to 6, and alternatively 2 to 5.
[0021] The total number of units per molecule of copolymer is not specifically restricted. The units shown above may be in any order, e.g., the copolymer may be a random copolymer or a block copolymer.
[0022] One skilled in the ail would recognize that the copolymer may be prepared by radical polymerization, via a process as described below, and that this process would form the terminal moiety for the copolymer. The copolymer further comprises a terminal moiety which may be derived from an initiator, a chain transfer agent, or both, as described, for example in Odian, George (2004). Principles of Polymerization (4th ed.). New York: Wiley-Interscience. ISBN 978-0-471-27400-1.
[0023] The copolymer may be prepared by any convenient means such as that disclosed in PCT Patent Application Publication W02023/019044 to Jeletic et al. by using the manganese ion source and the phenolic compound described herein as inhibitors, instead of the inhibitor described therein. Alternatively, the copolymer may be prepared as described in U.S. Patent 10,047,199 to limura et al. by varying appropriate starting materials and their amounts and by including the manganese ion source and the phenolic compound described herein. PCT Patent Application Publication W02023/019044 and U.S. Patent 10,047,199 are hereby incorporated by reference.
[0024] For example, a composition according to this invention may be prepared via a process comprising:
1) copolymerizing starting materials comprising
80 weight % to 98 weight % of (A) a crystallizable monomer of formula
, where R1 is an alkyl group of 16 to 24 carbon atoms as described above, and R2 is selected from the group consisting of H and methyl, as described above;
1 weight % to 15 weight % of (B) a silicone - (meth)acrylate macromonomer of formula
described above, each R3 is independently selected from the group consisting of R4 and a group of formula -OSilR4)?; each R4 is an independently selected monovalent hydrocarbon group of 1 to 12 carbon atoms; and
1 weight % to 5 weight % of C) a crosslinkable (meth)acrylate monomer of formula
independently selected from oxygen and NH, D3 is a divalent hydrocarbon group of 1 to 12 carbons, D4 is a divalent group of 2 to 4 carbon atoms, subscript v represents the number of units of formula (OD4) in the unit with subscript y, and subscript v is 0 to 12, and R8 is a crosslinkable group, each as described above; where the amounts of starting materials (A), (B), and (C) shown above total 100 weight% based on combined amounts of starting materials (A), (B), and (C). The starting materials used in step 1) of the process to prepare the composition further comprise:
(F) an initiator;
(K) a manganese ion source, and
(L) a phenolic compound. The starting materials used in the process to prepare the composition may optionally further comprise (H) a chain transfer agent; (J) an additional non- crystallizable monomer that is distinct from each of (A), (B) and (C); and a combination of both (H) and (J).
[0025] Copolymerizing, as introduced above, may comprise preparing the copolymer described above by a method comprising dissolving one or more of starting materials (A), (B), (C), and when present (H) and/or (J), in the presence of the (K) manganese ion source and (L)
the phenolic compound, in a solvent (e.g., an organic solvent such as a monohydric alcohol). Without wishing to be bound by theory, it is thought that during processing to combine (A), (B), (C), (H), and (J), stalling materials (K) and (L) may inhibit formation of acrylic radicals that can impact the formation of the copolymer during copolymerization in step 1). In step (1) copolymerizing starting materials (A), (B), (C), (F), and when present (H) and/or (J), in the presence of the (K) manganese ion source and (L) the phenolic compound, may be performed in a process such as that disclosed in U.S. Patent 10,047,199 to limura et al. by varying appropriate starting materials and their amounts. The resulting reaction product, which comprises the copolymer, (K) the manganese ion source, and (L) the phenolic compound, may be solvent borne. When solvent is present, all or a portion of the solvent may be removed by any convenient means, such as by stripping or distillation with optionally with heat and/or optionally reduced pressure. The resulting composition comprises the copolymer, (K) the manganese ion source, and (L) the phenolic compound. An aqueous emulsion may be prepared by a process comprising emulsifying said composition by mixing, under shear, the composition described above and additional starting materials comprising (D) a surfactant and (E) water. The resulting aqueous emulsion comprises the silicone - (meth) acrylate copolymer, (K) the manganese ion source, (L) the phenolic compound, (D) the surfactant, and (E) the water.
[0026] Alternatively, the process for preparing the composition may comprise an emulsion polymerization, e.g., the copolymer may be prepared via an emulsion polymerization process comprising:
(1) copolymerizing starting materials comprising
1 weight % to 15 weight % of (B) a silicone - (meth)acrylate macromonomer of formula
1 weight % to 5 weight % of (C) a crosslinkable (meth) acrylate monomer of formula
subscript v are as described above; where amounts of starting materials (A), (B), and (C) total 100 weight% based on combined amounts of starting materials (A), (B), and (C); and starting materials (A), (B), and (C) are copolymerized in an emulsion further comprising
(D) a surfactant;
(E) water;
(F) an initiator;
(K) a manganese ion source; and
(L) a phenolic compound; and thereby forming an aqueous emulsion comprising the silicone - (meth)acrylate copolymer, (D) the surfactant, (E) the water, (K) the manganese ion source, and (L) the phenolic compound. The process may optionally further comprise adding an additional starting material in step (1), where the additional starting material is selected from the group consisting of (G) a chain transfer agent; (H) an additional monomer that is distinct from starting materials (A), (B), and (C); (J) a reactive surfactant, and a combination of two or more of (G), (H), and (K).
[0027] The emulsion polymerization process described above may comprise forming an emulsion comprising starting materials (A), (B), (C), (D), (E), (K), and (L) (and optionally (H)
and/or (J)), and thereafter adding (F) the initiator and copolymerizing. Without wishing to be bound by theory, it is thought that during processing to combine and emulsify (A), (B), (C), (D), (E), (H), and (J), starting materials (K) and (L) may inhibit formation of acrylic radicals that can impact the formation of the copolymer during copolymerization in step (1).
[0028] If starting material (A) is a solid at RT, the starting materials may be heated to a temperature and for a time sufficient to melt starling material (A), e.g., 30 °C to 50 °C for 5 minutes to 15 minutes. The resulting starting materials may be mixed under shear to form the aqueous emulsion. Mixing under shear may be performed by any convenient means for forming an aqueous emulsion, such as sonication and with subsequent microfluidization. Equipment for mixing under shear, such as sonicators, homogenizers, microfluidizers, and speedmixers are known in the ail and are commercially available. Without wishing to be bound by theory, it is thought that mixing under shear may be used to obtain a submicron particle size in the emulsion. In step (1), starting materials comprising (A), (B), (C), (F), (K), and (L) (and when present (G), (H), and (J)) copolymerize to form the silicone - (meth)acrylate copolymer in the aqueous emulsion with starting materials (D) the surfactant, (E) the water, (K) the manganese ion source, and (L) the phenolic compound.
[0029] Before step (1) the starting materials comprising (A), (B), (C), (K), (L), and when present (H) and (J) may be combined under aerobic or anaerobic conditions, optionally with heating for extended times. For example, the starting materials comprising (A), (B), (C), (K), (L), and when present (H) and (J) may be emulsified with (D) and (E) before adding initiator and copolymerizing in step (1). In step (1), copolymerizing (either in solvent or via emulsion polymerization) in the process described above may be performed on a commercial scale under anaerobic or aerobic conditions optionally at elevated temperature, e.g., up to 100 °C, alternatively 40 °C to 80 °C, and alternatively 45 °C to 50 °C. Copolymerizing may be performed in a batch process with a residence time of 15 minutes to 24 hours, alternatively 30 minutes to 12 hours, alternatively 40 minutes to 8 hours, and alternatively 40 minutes to 2 hours. For purposes of this application, aerobic or anaerobic conditions means that oxygen is not required to be present in the gas in the headspace of the reactor where copolymerizing takes place, or dissolved in the liquid where copolymerizing takes place. The balance of the gas in the headspace could be an inert gas such as nitrogen or argon. The starting materials used herein will be further described below.
(A) Crystallizable Monomer
[0030] Starting material (A) used to prepare the copolymer is a crystallizablc monomer of formula (
2 are as described above. Examples of crystallizablc monomers for starting material (A) include stcaryl (mcth)acrylatc, eicosyl (meth)acrylate, behenyl (meth)acrylate, and combinations thereof. Alternatively, when R2 is hydrogen, starting material (A) may be an acrylate selected from stearyl acrylate, eicosyl acrylate, behenyl acrylate, and combinations thereof. Alternatively, starting material (A) may be stearyl acrylate. Crystallizable monomers suitable for starting material (A) are commercially available, e.g., from Millipore Sigma of St. Louis, Missouri, USA and from BASF SE of Ludwigshafen, Germany. Crystallizable means that the stalling monomer has a melting point ) > 25 °C ± 5 °C.
[0031] Starting material (A) is used in an amount of 80% to 98%, based on combined weights of starting materials (A) the crystallizable monomer, (B) the silicone - (meth)acrylate macromonomer, and (C) the crosslinkable (meth)acrylate monomer, and when present (H) the additional monomer and/or (J) the reactive surfactant. The amount of starting material (A) may be at least 80%, alternatively at least 81%, alternatively at least 82%, alternatively at least 83%, alternatively at least 84%, alternatively at least 85%, alternatively at least 86%, alternatively at least 87%, and alternatively at least 88% on the same basis. At the same time, the amount of starting material (A) may be up to 93%, alternatively up to 92%, alternatively up to 91%, alternatively up to 90%, alternatively up to 89%, and alternatively up to 88, on the same basis. Alternatively, the amount of starting material (A) may be 84% to 92%, alternatively 85% to 91 %, alternatively 86% to 90%, alternatively 87% to 89%, and alternatively 88%; on the same basis.
(B) Silicone - (Meth)acrylate Macromonomer
[0032] Starting material (B) is a silicone - (meth)acrylate macromonomer of formula
are as described above.
Alternatively, starting material (B) may have a formula selected from the group consisting of:
4 are as described above.
Alternatively, starting material (B) may have formula:
3-(l,l,l,5,5,5-hexamethyl-3-((trimethylsilyl)oxy)trisiloxan-3-yl)propyl methacrylate.
Starting material (B) may be prepared by known methods, such as those disclosed in PCT Publication WO2020/142388 and U.S. Patent 6,420,504.
[0033] Starling material (B) is used in an amount of 1% to 15%, based on combined weights of starting materials (A), (B), and (C), and when present (H) the additional monomer and/or (J) the reactive surfactant. Alternatively, starting material (B) may be used in an amount of at least
5%, alternatively at least 6%, alternatively at least 7%, alternatively at least 8%, alternatively at least 9%, and alternatively at least 10%, on the same basis. At the same time, starting material
(B) may be present in an amount up to 15%, alternatively up to 14%, alternatively up to 13%, alternatively up to 12%, alternatively up to 11%, and alternatively up to 10%, on the same basis. Alternatively, the amount of starting material (B) may be 6% to 14%, alternatively 7% to 13%, alternatively 8% to 12%, alternatively 9% to 11%, and alternatively 10%, on the same basis described above.
(C) Crosslinkable (Meth)acrylate Monomer
[0034] Starting material (C) is a crosslinkable (meth)acrylate monomer of formula (C-l):
subscript v are as described above. Examples of suitable crosslinkable (meth)acrylates for starting material (C) include (2-acetoacetoxy)ethyl methacrylate, hydroxybutyl (meth)acrylate, hydroxyethyl (meth)acrylate, hydroxyethylcaprolactone (meth)acrylate, hydroxypropyl (meth)acrylate, ureido (meth)acrylate, glycidyl (meth)acrylate (GMA), poly(ethylene glycol) (meth)acrylate (PEGMA), and combinations thereof. The ureido (meth) acrylate monomer may have formula:
oxygen atom or an NH moiety. Examples of ureido monomers are known in the art and are disclosed, for example, in U.S. Patent 9,212,292 to Pressley, ct al. Other crosslinkablc (mcth)acrylatc monomers arc known in the ail and are commercially available, e.g., from BASF SE. Other crosslinkable (meth)acrylates are commercially available as Sipomer WAM1 and 2.
[0035] Starting material (C) is used in an amount 1% to 5%, based on combined weights of starting materials (A), (B), and (C), and when present, (H) and/or (J). The amount of starting material (C) may be at least 1%, alternatively at least 1.25%, alternatively at least 1.5%,
alternatively at least 1 .75%, and alternatively at least 2%, on the same basis. At the same time, the amount of starting material (C) may be up to 5%, alternatively up to 4%, alternatively up to 3%, alternatively up to 2.75%, alternatively up to 2.5%, and alternatively up to 2.25%, on the same basis. Alternatively, the amount of starting material (C) may be 1% to 3%, alternatively 1% to 2%, alternatively 1.5% to 2.5%, alternatively 1.75% to 2.25%, and alternatively 2%; on the same basis.
Starting Material (D) Surfactant
[0036] Stalling material (D) is a surfactant. The surfactant used in copolymerizing in step (1) of the process for making the aqueous emulsion is not specifically restricted and may be selected from the group consisting of anionic surfactants, amphoteric surfactants, cationic surfactants, nonionic surfactants, and combinations of two or more thereof.
[0037] The anionic surfactants include (i) sulfonic acids and their salt derivatives, including alkyl, aralkyl, alkyl naphthalene, alkyl diphenyl ether sulfonic acids, and their salts, having at least 6 carbon atoms in the alkyl substituent, such as dodecyl benzene sulfonic acid, and its sodium salt or its amine salt; (ii) alkyl sulfates having at least 6 carbon atoms in the alkyl substituent, such as sodium lauryl sulfate; (iii) the sulfate esters of polyoxyethylene monoalkyl ethers; (iv) long chain carboxylic acid surfactants, such as lauric acid, steric acid, oleic acid, and their alkali metal and amine salts. Some other examples of anionic surfactants are alkali metal sulfosuccinates; sulfonated glyceryl esters of fatty acids such as sulfonated monoglycerides of coconut oil acids; salts of sulfonated monovalent alcohol esters such as sodium oleyl isothionate; amides of amino sulfonic acids such as the sodium salt of oleyl methyl tauride; sulfonated products of fatty acid nitriles such as palmitonitrile sulfonate; sulfonated aromatic hydrocarbons such as sodium alpha-naphthalene monosulfonate; condensation products of naphthalene sulfonic acids with formaldehyde; sodium octahydro anthracene sulfonate; alkali metal alkyl sulfates; ether sulfates having alkyl groups of eight or more carbon atoms such as sodium lauryl ether sulfate; and alkylaryl sulfonates having one or more alkyl groups of eight or more carbon atoms such as neutral salts of hexadecylbenzene sulfonic acid and C20 alkylbenzene sulfonic acid.
[0038] Commercial anionic surfactants which can be used include the sodium salt of dodecyl benzene sulfonic acid sold under the trademark SIPONATE™ DS- 10 by Alcolac Inc., Baltimore, Maryland; sodium salt of alkyl alkoxylatc sulfate sold under the trademark DOWFAX™ AS-801
by The Dow Chemical Company of Midland, Michigan, USA; sodium n-hexadecyl diphcnyloxidc disulfonatc sold under the trademark DOWFAX™ 8390 by The Dow Chemical Company, Midland, Michigan; the sodium salt of a secondary alkane sulfonate sold under the trademark HOSTAPUR™ SAS 60 by Clariant Corporation, Charlotte, North Carolina; N-acyl taurates such as sodium N-lauroyl methyl taurate sold under the trademark NIKKOL LMT™ by Nikko Chemicals Company, Ltd., Tokyo, Japan; and linear alkyl benzene sulfonic acids sold under the trademark BIO-SOFT™ S-100 by the Stepan Company, Northfield, Illinois. Compositions of the latter type such as dodecyl benzene sulfonic acid, although a catalyst as noted above, can also function as the anionic surfactant when neutralized. Other suitable surfactants include sodium alkyl sulfonate such as HOSTAPUR™ SAS-30, and triethanolamine dodecyl benzene sulfonate, such as BIO-SOFT™ N 300.
[0039] Suitable amphoteric surfactants include amino acid surfactants, betaines (e.g., lauryl betaine, bis-(2-hydroxyethyl) tallow betaine, cocamidopropylbetaine, N-alkylamidobetaines, and derivatives thereof), proteins and derivatives thereof, glycinates (glycine derivatives, such as cocamphoglycinate, cocamphocarboxy-glycinates, and cocamphodipropionate), sultaines (e.g., cocamidopropylhydroxysultaine and lauryl sultaine), alkyl aminopropionates, alkyl polyaminocarboxylates and alkylamphoacetates, lecithin and hydrogenated lecithin, and combinations thereof. These surfactants are commercially available from various suppliers under different tradenames. For example, REWOTERIC™ AM TEG is produced by Evonik of Essen, Germany; AMPHOSOL™ CG is available from Stepan Company of Northfield, Illinois, USA.
[0040] Alternatively, (D) the surfactant used in copolymerizing in step (1) may be selected from the group consisting of (D-l) an anionic surfactant as described above, (D-2) a cationic surfactant, (D-3) a nonionic surfactant, (D-4) an amphoteric surfactant, as described above, and (D-5) a combination of two or more of (D-l) to (D-4). Cationic surfactants useful herein include compounds containing quaternary ammonium hydrophilic moieties in the molecule which are positively charged, such as quaternary ammonium salts, which may be represented by formula (D-2-1): R12R13R14R15N+X’" where R12 to R15 are alkyl groups containing 1-30 carbon atoms, or alkyl groups derived from tallow, coconut oil, or soy; and X’ is a halogen, e.g., chlorine or bromine. Alternatively, the quaternary ammonium compounds may be alkyl trimethyl ammonium and dialkyldimcthylammonium halides, or acetates, having at least 8 carbon atoms in each alkyl
substituent. Dialkyl dimethyl ammonium salts can be used and are represented by formula (D-2- 2): R16R17N+(CH3)2X’- where R16 and R17 are alkyl groups containing 12-30 carbon atoms or alkyl groups derived from tallow, coconut oil, or soy; and X’ is halogen. Monoalkyl trimethyl ammonium salts can be used and are represented by formula (D-l-3): R18N+(CH3)3X”‘ where R18 is an alkyl group containing 12-30 carbon atoms or an alkyl group derived from tallow, coconut oil, or soy; and X” is halogen or acetate.
[0041] Representative quaternary ammonium halide salts are dodecyltrimethyl ammonium chloride/lauryltrimethyl ammonium chloride (LTAC), cetyltrimethyl ammonium chloride (CTAC), hexadeclyltrimethyl ammonium chloride, didodecyldimethyl ammonium bromide, dihexadecyldimethyl ammonium chloride, dihexadecyldimethyl ammonium bromide, dioctadecyldimethyl ammonium chloride, dieicosyldimethyl ammonium chloride, and didocosyldimethyl ammonium chloride. These quaternary ammonium salts are commercially available under trademarks such as ADOGEN™, ARQUAD™, TOMAH™, and VARIQUAT™. [0042] Other suitable cationic surfactants which can be used include fatty acid amines and amides and their salts and derivatives, such as aliphatic fatty amines and their derivatives. Such cationic surfactants that are commercially available include compositions sold under the names ARQUAD™ T27 W, ARQUAD™ 16-29, by Akzo Nobel Chemicals Inc., Chicago, Illinois; and Ammonyx Cetac-30 by the Stepan Company, Northfield, Illinois, USA.
[0043] The amount of (D-2) the cationic surfactant may be 0.1 % to 5%, based on weight of (I) the silicone - (meth)acrylate copolymer in the aqueous emulsion. Alternatively, the amount of cationic surfactant may be at least 0.1%, alternatively at least 0.2%, alternatively at least 0.3%, alternatively at least 0.4%, alternatively at least 0.5%; while at the same time the amount of cationic surfactant may be up to 5%, alternatively up to 4%, alternatively up to 3%, alternatively up to 2%, alternatively up to 1%, on the same basis. Alternatively, the amount of cationic surfactant may be 0.2% to 4%, alternatively 0.3% to 3%, alternatively 0.4% to 2.5%, and alternatively 0.5% to 2%; on the same basis.
[0044] Alternatively, (D) the surfactant used in copolymerizing in step (1) may be (D-3) a nonionic surfactant. Some suitable nonionic surfactants which can be used include polyoxyalkylene copolymers, such as polyoxyethylene alkyl ethers, polyoxyethylene alkylphenol ethers, alkyl glycosides (e.g., alkylglucosides), polyoxyethylene fatty acid esters, polyoxyethylene lauryl ethers, sorbitan fatty acid esters, polyoxyethylene sorbitan fatty acid
esters, polyoxyethylene sorbitan monooleates, polyoxyethylene alkyl esters, polyoxyethylene sorbitan alkyl esters, polyethylene glycol (such as polyethylene glycol having 23 cthylcnc-oxidc units), polypropylene glycol, diethylene glycol, ethoxylated trimethylnonanols, tristyrylphenol ethers (TSP’s), distyryl phenol ethers (DSP’s), and polyoxyalkylene glycol modified polysiloxane surfactants. Nonionic surfactants which are commercially available include compositions such as (i) 2,6,8-trimethyl-4-nonyl polyoxyethylene ether sold under the names TERGITOL™ TMN-6 and TERGITOL™ TMN-10; (ii) the C11-15 secondary alcohol ethoxylates such as those sold under the names TERGITOL™ 15-S-7, TERGITOL™ 15-S-9, TERGITOL™ 15-S-12, TERGITOL™ 15-S-15, TERGITOL™ 15-S-20, TERGITOL™ 15-S-30, and TERGITOL™ 15-S-40, by the Dow Chemical Company, of Midland, Michigan, USA; octylphenyl polyoxyethylene (40) ether sold under the name TRITON™ X405 by the Dow Chemical Company; (iii) nonylphenyl polyoxyethylene (10) ether sold under the name MAKON™ 10 by the Stepan Company; (iv) ethoxylated alcohols sold under the name Trycol 5953 by Henkel Corp./Emery Group, of Cincinnati, Ohio, USA; (v) ethoxylated alcohols sold under the name BRII™ L23 (polyoxyethylene 23 lauryl ether or Laureth-23) and BRII™ L4 by Croda Inc. of Edison, New Jersey, USA, (vi) alkyl-oxo alcohol polyglycol ethers such as GENAPOL™ UD 050, and GENAPOL™ UDI 10, (vii) alkyl polyethylene glycol ether based on ClO-Guerbet alcohol and ethylene oxide such as LUTENSOL™ XP 79; (viii) other alcohol ethoxylates, for example, C11-15 alkyl alcohol ethoxylate with tradename ECOSURF™ EH, such as ECOSURF™ EH-40, from TDCC. TSPs and DSP’s are commercially available from Stepan. [0045] Suitable nonionic surfactants also include poly(oxyethylene)-poly(oxypropylene)- poly(oxyethylene) tri-block copolymers. Poly(oxyethylene)-poly(oxypropylene)- poly(oxyethylene) tri-block copolymers are also commonly known as Poloxamers. They are nonionic triblock copolymers composed of a central hydrophobic chain of polyoxypropylene (polypropylene oxide)) flanked by two hydrophilic chains of polyoxyethylene (poly(ethylene oxide)). Poly(oxyethylene)-poly(oxypropylene)-poly(oxyethylene) tri-block copolymers are commercially available from BASF of Florham Park, New Jersey, USA, and are sold under the tradename PLURONIC™, such as PLURONIC™ L61, L62, L64, L81, P84.
[0046] The nonionic surfactant may also be a silicone poly ether (SPE). The silicone poly ether as an emulsifier may have a rake type structure wherein the polyoxyethylene or polyoxyethylene-polyoxypropylene copolymeric units are grafted onto the siloxane backbone, or
the SPE can have an ABA block copolymeric structure wherein A represents the polyether portion and B the siloxane portion of an ABA structure. Suitable SPE’s include DOWSIL™ OFX-5329 Fluid from Dow Silicones Corporation of Midland, Michigan, USA. Alternatively, the nonionic surfactant may be selected from polyoxyalkylene-substituted silicones, silicone alkanolamides, silicone esters and silicone glycosides. Such silicone-based surfactants may be used to form such aqueous emulsions and are known in the art, and have been described, for example, in U.S. Patent 4,122,029 to Gee et al., U.S. Patent 5,387,417 to Rentsch, and U.S. Patent 5,811,487 to Schulz et al.
[0047] Starting material (D-3) the nonionic surfactant may be delivered in a dilution, and the amount used may be sufficient to provide 0.1% to 10% of the surfactant, based on weight of (I) the silicone - (meth)acrylate copolymer in the aqueous emulsion. Alternatively, the amount of nonionic surfactant may be at least 0.1%, alternatively at least 0.2%, alternatively at least 0.3%, alternatively at least 0.4%, alternatively at least 0.5%, alternatively at least 1%; while at the same time the amount of nonionic surfactant may be up to 10%, alternatively up to 5%, alternatively up to 4%, alternatively up to 3%, alternatively up to 2%, alternatively up to 1%, on the same basis. Alternatively, the amount of nonionic surfactant may be 0.2% to 4%, alternatively 0.3% to 3%, alternatively 0.4 to 2.5%, and alternatively 1% to 2%; on the same basis. Alternatively, starting materials (D-l) the anionic surfactant, (D-2) the cationic surfactant, and (D-3) the nonionic surfactant may be present in combined amounts < 10%, based on weight of (I) the silicone - (meth)acrylate copolymer in the aqueous emulsion.
Starting Material (E) Water
[0048] Starling material (E) is water. The water is not generally limited, for example, the water may be processed or unprocessed. Examples of processes that may be used for purifying the water include distilling, filtering, deionizing, and combinations of two or more thereof, such that the water may be deionized, distilled, and/or filtered. Alternatively, the water may be unprocessed (e.g. may be tap water, i.e., provided by a municipal water system or well water, used without further purification). The amount of water is sufficient to form an aqueous emulsion for copolymerizing in step (1) of the process described above. The amount of water used in step (1) varies depending on various factors including the types and amounts of starting materials (A), (B), and (C), and whether (H) the additional monomer is present. However, the water may be added in an amount of 20% to 97%, alternatively 30% to 90%, and alternatively
40% to 80%, alternatively 50% to 97%, alternatively 50% to 90%, and alternatively 60% to 80%; based on combined weights of all starting materials in step (1). Alternatively, the water may be added in an amount of at least 20%, alternatively at least 30%, alternatively at least 40%, alternatively at least 50%, and alternatively at least 60%; while at the same time the amount of water may be up to 97%, alternatively up to 96%, alternatively up to 95%, and alternatively up to 80%, on the same basis.
[0049] Without wishing to be bound by theory, it is thought that starting materials (A), (B), and (C), and when present (H) and/or (J), copolymerize to form the silicone - (meth)acrylate copolymer described above. It is further thought that starting materials (D) surfactant and (E) water do not participate in the copolymerization reaction, however, a copolymer including one or both of starting materials (D) and (E) is not excluded from the scope herein.
[0050] Additional water may be added after step (1). For example, the aqueous emulsion prepared as described above may be diluted with additional water to achieve a desired amount of starting materials before preparing the emulsion formulation and/or treating a textile with the emulsion formulation. Starting material (III), the water, in the emulsion formulation suitable for treating the textile may be the same as (E) the water described above, and may be introduced when the emulsion copolymerization process described above is used to prepare the aqueous emulsion in step (1).
(F) Initiator
[0051] Starting material (F), an initiator, is also added to facilitate copolymerizing in step (1) described above. Suitable initiators include azo compounds and peroxide compounds. For example, the azo compound may be an aliphatic azo compound such as 1-t-amylazo-l- cyanocyclohexane, azo-bis-isobutyronitrile and 1-t-butylazo-cy anocyclohexane, 2,2'-azo- bis-(2- methyl)butyronitrile, 2,2’-azobis(2-methylpropionitrile), 2,2’-azobis(2-methylpropionamidine) dihydrochloride, 2,2’-azobis(cyanovaleric acid), or a combination of two or more thereof. Azo compounds are known in the art and are commercially available, e.g., under the tradename VAZO™ WSP from The Chemours Company of Wilmington, Delaware, USA. The peroxide compound may be a peroxide or a hydroperoxide, such as t-butylperoctoate, t-butyl perbenzoate, dicumyl peroxide, di-t-butyl peroxide, t-butyl hydroperoxide, cumene hydroperoxide, di-t-amyl peroxide and combinations of two or more thereof. Additionally, di-peroxide initiators may be used alone or in combination with other initiators. Such di-peroxide initiators include, but are not
limited to, l ,4-bis-(t-butyl peroxycarbo)cyclohexane, 1 ,2-di(t-butyl peroxy)cyclohexane, and 2,5-di(t-butyl pcroxy)-3-hcxync. Suitable peroxide compounds arc known in the art and arc commercially available from various sources, such as Sigma-Aldrich, Inc. Alternatively, the initiator may comprise isoascorbic acid.
[0052] An initiator may be used alone as starting material (F). Alternatively, starting material
(F) may be a redox pair, which comprises an initiator as the oxidizing component and a reducing component. Alternatively, a redox pair including isoascorbic acid and a hydrophobic organic hydroperoxide such as t-amyl hydroperoxide or t-butyl hydroperoxide may be used as starting material (F). Examples of suitable initiators and/or redox pairs for starting material (F) are disclosed in U.S. Patent 6,576,051 to Bardman et al., beginning at col. 11, line 16. How the initiator is added depends on various factors including whether the initiator is water soluble and the type of initiator (e.g., whether a thermal initiator or a redox pair is used). Typically, when a thermal initiator is used, all the initiator is added at once at the beginning of step (1).
Alternatively, when a redox pair is used, it may be metered in over time.
[0053] Alternatively, the initiator may optionally further comprise Iron(II) sulfate hexahydrate, Potassium persulfate, or a combination thereof. The initiator (F) may be used in an amount sufficient to provide 0.01% to 3%, alternatively 0.1% to 1.5%, based on weight of (I) the silicone - (meth)acrylate copolymer.
(G) Chain Transfer Agent
[0054] An additional starting material that may be added to facilitate copolymerizing in step (1) of the process described above comprises (G) a chain transfer agent. Suitable chain transfer agents include mercaptans such as alkyl mercaptans, e.g., n-octyl mercaptan, n-dodecyl mercaptan, dodecyl mercaptan (dodecane thiol), and/or 2,2-dimethyldecyl mercaptan. Alternatively, the chain transfer agent may be water soluble, such as mercaptoacetic acid and/or 2-mercaptoethanol. Suitable chain transfer agents are known in the art and have been disclosed, for example, in “Radical Polymerization in Industry” by Peter Nesvadba, Performance Chemical Research, GASF Schweiz AG, Basel, Switzerland, Encyclopedia of Radicals in Chemistry, Biology and Materials, Online © 2012 John Wiley & Sons, Ltd.
[0055] Starting material (G), the chain transfer agent, is optional and may be added in an amount of 0 to 10%, based on combined weights of stalling materials (A), (B), and (C) (and when present (H) and/or (J)). Alternatively, (G) the chain transfer agent may be used in an
amount of 5% to 10% on the same basis.
(H) Additional Monomer
[0056] Stalling material (H) is an optional additional monomer that may be added for copolymerizing in step (1). Starting material (H) may be a non-crystallizable monomer that is distinct from starting materials (A), (B), and (C), described above. The additional monomer, when present, may be used in an amount of > 0 to 18 weight % based on weight of (I) the silicone - (meth) acrylate copolymer. Suitable monomers include (meth)acrylate monomers such as methyl methacrylate, t-amyl methacrylate, butyl (meth)acrylate such as t-butyl methacrylate, cyclohexyl (meth)acrylate, iso-decyl (meth)acrylate, isobomyl(meth)acrylate, 2-naphthyl acrylate, benzyl (meth)acrylate, 2-ethylhexyl(meth)acrylate, and combinations of two or more thereof. Alternatively the additional monomer may be styrene or vinyl chloride. Suitable monomers for starting material (H) are known in the art and are commercially available, e.g., from Polysciences, Inc. Alternatively, the additional monomer (H) may be selected from the group consisting of isobomyl methacrylate (IB MA), isobomyl acrylate (IBA), and a combination thereof. The additional monomer is optional, and may be present in an amount of 0 to 18%, based on combined weights of starting materials (A), (B), and (C), and when present (H) and (J). Alternatively, (H) the additional monomer may be present in an amount of at least 0.5%, alternatively at least 1%, and alternatively at least 2%; while at the same time the additional monomer may be present in an amount up to 18%, alternatively up to 15%, alternatively up to 10%, alternatively up to 8%, and alternatively up to 5%, on the same basis. Alternatively, the amount of (H) the additional monomer may be > 0 to 18%, alternatively 0.5% to 7%, alternatively 1% to 6%, and alternatively 2% to 5%, on the same basis.
(I) Reactive Surfactant
[0057] Starting material (J) is an optional reactive surfactant that may be used to facilitate emulsion copolymerization, and that may react to form part of the copolymer. The reactive surfactant may have formula:
R6 are as described above, and subscript vl has a value sufficient to give the reactive surfactant a
number average molecular weight of 300 to 950 g/mole, alternatively 350 g/mol to 900 g/mole.
For example, a reactive surfactant of formula
subscript vl has a value sufficient to give the reactive surfactant a number average molecular weight of 300 g/mol to 950 g/mol is commercially available from Sigma- Aldrich, Inc. of St. Louis, Missouri, USA. The reactive surfactant is optional and may be added in an amount of 0 to 5 weight % based on combined weights of starting materials (A), (B), and (C), and when present (H), and (J). Reactive surfactants are commercially available, e.g., from Ethox. Alternatively, the reactive surfactant may be added in an amount of > 0 to 2 weight % on the same basis.
Starting Material (K) Manganese Ion Source
[0058] Stalling material (K) is a manganese ion source, which may be a manganese (II) compound. Suitable manganese compounds include manganese (II) acetate, manganese (II) nitrite, manganese (II) propionate, manganese (II) oxide, manganese (II) hydroxide, manganese (II) chloride, manganese (II) phosphate, manganese (II) perchlorate, hydrates thereof (e.g., manganese (II) tetrahydrate) and combinations thereof. Alternatively, the manganese ion source may comprise manganese (II) acetate or manganese (II) tetrahydrate, or a combination thereof. Suitable manganese ion sources are commercially available from Millipore Sigma of St. Louis, Missouri, USA, Fisher Scientific of Waltham, Massachusetts, USA, and City Chemical LLC of Connecticut, USA. The amount of manganese ion source depends on various factors including the selections and amounts of other starting materials used, however the amount may be 0.1 ppm to 5,000 ppm based on combined weights of starting materials (A), (B), and (C). Alternatively, the amount of the manganese ion source may be > 0 ppm, alternatively at least 0.5 ppm, alternatively at least 1 ppm, alternatively at least 1 .5; while at the same time, the amount of manganese ion source may be up to 10 ppm, alternatively up to 5 ppm, alternatively up to 4 ppm, and alternatively up to 3 ppm, and alternatively up to 2 ppm, based on combined weights of all starting materials in the emulsion formulation for treating the textile.
(L) Phenolic Compound
[0059] Starting material (L) is a phenolic compound. Suitable phenolic compounds include hydroquinone (HQ), dihydroxybcnzcnc (catechol), resorcinol, dihydroxyxylcnc, mcthoxyphcnols such as guaiacol, p-methoxyphenol (also called methyl ether of hydroquinone or MeHQ), tertbutyl hydroquinone (tBuHQ), pyrogallol, methylpyrogallol, cresol, phenol, xylenols, and combinations thereof. Alternatively, the phenolic compound may be selected from the group consisting of HQ, MeHQ, tBuHQ, and a combination of two or more thereof. Suitable phenolic compounds are commercially available, e.g., from Millipore Sigma of St. Louis, Missouri, USA. The amount of phenolic compound source depends on various factors including the selections and amounts of other starting materials used, however the amount may be 5 ppm to 5,000 ppm based on combined weights of starting materials (A), (B), and (C). Alternatively, the amount of the phenolic compound may be at least 5 ppm, alternatively at least 50 ppm, alternatively at least 100 ppm, alternatively at least 150 ppm; while at the same time, the amount of phenolic compound may be up to 500 ppm, alternatively up to 400 ppm, alternatively up to 350 ppm, and alternatively up to 320 ppm, based on combined weights of all starling materials in the emulsion formulation for treating the textile.
Method for Preparing Emulsion Formulation Suitable for Treating a Textile
[0060] The copolymer prepared as described above and/or the aqueous emulsion prepared by the process comprising step (1), described above, can be used to prepare the emulsion formulation suitable for treating the textile, described above. For example, preparing the emulsion formulation may be performed by a process comprising mixing starting materials comprising (I) the copolymer, (K) the manganese ion source, (L) the phenolic compound, (II) the nonionic surfactant, (III) the water (which may be as described above for starting material (E)), and (IV) the water dispersible crosslinker under shear (e.g., when the copolymer is formed by the solvent borne method described above, e.g., by a method comprising dissolving one or more of the starting materials in an organic solvent and copolymerizing starting materials (A) the crystallizable monomer, (B) silicone - (meth) acrylate macromonomer, (C) the crosslinkable monomer, (F) the initiator, and when present (G) the chain transfer agent and/or (H) the additional monomer with subsequent solvent removal) in the presence of (K) the manganese ion source, and (L) the phenolic compound. Mixing under shear may be performed by any convenient means for forming an aqueous emulsion formulation, such as sonication and with subsequent microfluidization. Equipment for mixing under shear, such as sonicators,
homogenizers, microfluidizers, and speedmixers are known in the art and are commercially available.
[0061] In the emulsion formulation, (II) the nonionic surfactant comprises an alcohol
alkoxylate of formula (II- 1) ' , where R 19 is a branched or linear alkyl group with
8 to 15 carbon atoms, subscript n is at least 23, and each D5 is an independently selected alkylene oxide group of 2 or more carbon atoms. Alternatively, R19 may be, for example, 2-ethylhexyl, lauryl, or other alkyl group of 8 to 15 carbon atoms. Alternatively, subscript n may be 23 to 40, alternatively 23 to 30. Alternatively, each D5 may be independently selected from the group consisting of ethylene oxide and propylene oxide. Alternatively, each D5 may be ethylene oxide. Suitable alcohol ethoxylates of the formula above include C11-15 secondary alkyl alcohol ethoxylate (such as TERGITOL™ 15-S-30 or 15-S-40, as described above); 2-ethylhexyl primary alcohol alkoxylate, (such as 2-ethylhexyl ethylene oxide/propylene oxide, sold under the tradename ECOSURF EH-30 and ECOSURF EH-40 as described above), a C12-40 primary alcohol ethoxylate (such as laureth-23, sold under the tradename BRU™ L23 as described above); or a combination thereof. The emulsion formulation may optionally further comprise an additional nonionic cosurfactant, which may be any of the nonionic surfactants described above as starting material (D-3) that differs from formula (11-1) above.
[0062] This process for preparing the emulsion formulation suitable for treating the textile may optionally further comprise adding a further additional starting material. The further additional starting material may be selected from the group consisting of (V) a wax, (VI) a biocide, (VII) additional water (which may be the same as starting material (E)), (VIII) a flame retardant, (IX) a wrinkle reducing agent, (X) an antistatic agent, (XI) a penetrating agent and a combination of two or more of (V) to (XI).
[0063] Alternatively, the aqueous emulsion prepared in the process comprising step (1), described above, may be used to prepare the emulsion formulation suitable for treating the textile. To prepare the emulsion formulation, the process described above comprising step (1) further comprises step (2): adding starling material (IV), the water dispersible crosslinker, to the aqueous emulsion. This process for preparing the emulsion formulation suitable for treating the textile may optionally further comprise, after step (1), adding the further additional starting material. The further additional starting material may be selected from the group consisting of
(V) a wax, (VT) a biocide, (VII) additional water (which may be the same as starting material (D)), (VIII) a flame retardant, (IX) a wrinkle reducing agent, (X) an antistatic agent, (XI) a penetrating agent and a combination of two or more of (V) to (XI).
[0064] Step (2) of this process may be performed by any convenient means, such as mixing using in a jacketed vessel equipped with an agitator. Step (1) and step (2), and any optional additional steps as described above may be performed sequentially in the same vessel. Alternatively, step (1) and step (2) may be performed in different equipment. Step (2) may be performed at RT or elevated temperature, e.g., up to 100°C, alternatively 40°C to 80°C. Alternatively, heating may be performed in step (1), and step (2) may be performed at RT. Alternatively, step (2) may be performed at lower temperatures and elevated pressures, such as up to 5 atmospheres.
[0065] The process for preparing the emulsion formulation suitable for treating the textile may optionally further comprise adding additional surfactant. Alternatively, when the process for preparing the copolymer includes a surfactant that is not nonionic (e.g., (D-l) the anionic surfactant, (D-4) the amphoteric surfactant, and/or (D-2) the cationic surfactant, described above), the process for preparing the emulsion formulation may further comprise (3): removing (D) the surfactant (used in the process to make the aqueous emulsion comprising the copolymer) and replacing (D) the surfactant with (II) the nonionic surfactant (which may be as described above for D-3). Starting material (D-l) the anionic surfactant, (D-2) the cationic surfactant, and (D-4) the amphoteric surfactant may be removed by any convenient means such as adsorption, contact with an ion exchange resin, and/or filtration, such as diafiltration and/or ultrafiltration. Alternatively, when the process for preparing the copolymer employs as starting material (D-3) a nonionic surfactant other than the nonionic surfactant of formula (II- 1) above, the process may further comprise removing all or a portion of starting material (D-3) and replacing with the nonionic surfactant of formula (II- 1) by the means as described above.
[0066] After the process for preparing the emulsion formulation is complete, the emulsion formulation suitable for treating the textile is free of cationic surfactant. Alternatively, the emulsion formulation may be free of anionic surfactant, amphoteric surfactant, and cationic surfactant. “Free of anionic surfactant, amphoteric surfactant and cationic surfactant” means that the surfactant in the emulsion formulation is only a nonionic surfactant, or the emulsion formulation contains either a non-detectable amount of another surfactant, or an amount of
another surfactant insufficient to detrimentally affect the durable water repellency of a textile treated with the emulsion formulation.
[0067] In addition to (I) the copolymer, the emulsion formulation suitable for treating the textile comprises (II) nonionic surfactant, which is as described above for starting material (D-3) and (III) water, which is as described above for starting material (E), (K) the manganese ion source, and (L) the phenolic compound. Starting material (IV) the water dispersible crosslinker, and the further additional starting materials, which may be selected from the group consisting of (V) a wax, (VI) a biocide, (VII) additional water (which may be the same as starting material (E)), (VIII) a flame retardant, (IX) a wrinkle reducing agent, (X) an antistatic agent, (XI) a penetrating agent, are described below.
Starting Material (IV) Water Dispersible Crosslinker
[0068] Starting material (IV) is a water dispersible crosslinker (crosslinker) that may be added to the emulsion formulation for treating the textile. Suitable crosslinkers include blocked isocyanates, polycarbodiimides, polyepoxy compounds, polycarboxy compounds, polyamines, and diols. The term “blocked isocyanates” encompasses mono-, di- and polyisocyanates in which an isocyanate group has been reacted with a blocking agent, which upon heating, release the isocyanate and the blocking agent. Suitable blocking agents are known in the art such as amines, amides, compounds having an active hydrogen atom, alcohols, or oximes. Blocked isocyanates are commercially available, such as ARKOPHOB™ DAN and ARKOPHOB™ SR from Archroma of Reinach, Switzerland; RUCO-GUARD™ WEB from Rudolf GmbH of Geretsreid, Bayern, Germany, and PHOBOL™ UXN extender and PHOBOL™ XAN extender from Archroma. Diols include, for example, 1,2-propoanediol; 1,3-propanediol; 1,2-butanediol; 1,3-butanediol; 1,4-butanediol; 2, 3 -butanediol; 2-methyl-l,2-propanediol; 1,5 -pentanediol; 2- methyl-2,3-butanediol; 1,6-hexanediol; 1,2-hexanediol; 2,5-hexanediol; 2-methyl-2,4- pentanediol; 2,3-dimethyl-2,3-butanediol; 2-ethylhexanediol; 1,2-octanediol; 1,2-decanediol; 2,2,4-trimethylpentanediol; 2-butyl-2-ethyl- 1 ,3-propanediol; 2,2-diethyl- 1 ,3-propanediol. Examples of suitable crosslinkers are known in the art and are disclosed, for example, in U.S. Patent Application 2017/0204558 to Knaup; U.S. Patent 9,777,105 to Hamajima et al., beginning at col. 11, line 54, which are hereby incorporated by reference for the purpose of describing suitable crosslinkers. The exact amount of crosslinker (IV) depends on various factors including the type and amount of (I) silicone - (meth)acrylate copolymer and the textile to be treated,
however, the weight of the crosslinker (IV) may 0.25% to 3.75% on fabric weight, alternatively 0.25% to 1%, and alternatively 0.25% to 0.5%, on the same basis.
Starting Material ( V) Wax
[0069] Starting material (V) is a wax, which may optionally be added to provide improved water repellency or softness to the textile to which the emulsion formulation will be applied. The amount of wax will vary depending on factors including the type of wax selected, the benefit desired, and the fabric to be treated with the emulsion formulation. However, the amount of wax may be 0 to 75%, alternatively 0 to 50%, alternatively 25% to 50% based on weight of (I) the silicone - (meth)acrylate copolymer. Alternatively, when used, the amount of wax may be > 0%, alternatively at least 10%, and alternatively at least 25%, while at the same time the amount of wax may be up to 75%, alternatively up to 50% on the same basis. Examples of suitable waxes include paraffin waxes (e.g., n-paraffins, iso-paraffins, and/or cycloparaffins), silicone waxes such as silicone wax with long chain alkyl groups (e.g., alkyl methyl silicone wax) and/or amino-silicone wax, and a combination of two or more thereof. Suitable waxes are disclosed, for example, in U.S. Patent Application 2017/0204558 to Knaup and U.S. Patent 10,844,151 to Probst, et al. Waxes may be delivered as water-based dispersions, for example Michelman wax 743 and others from Michelman of Cincinnati, Ohio, U.S.A. Other waxes are also commercially available, for example, from Sasol Wax of Hamburg, Germany, and silicone waxes, such as DOWSIL™ AMS-C30, are available from Dow Silicones Corporation of Midland, Michigan, U.S.A.
Starting Material (VI) Biocide
[0070] Starling material (VI) is an optional biocide. The amount of biocide will vary depending on factors including the type of biocide selected and the benefit desired. However, when used, the amount of biocide may be > 0% to 5% based on the combined weights of all starting materials in the emulsion formulation. Starting material (VI) is exemplified by (VI- 1) a fungicide, (VI-2) an herbicide, (VI-3) a pesticide, (VI-4) an antimicrobial agent, or a combination thereof. Suitable biocides are disclosed, for example, in U.S. Patent 9,480,977. Starting Material (XI) Penetrating Agent
[0071] Starting material (XI) is a penetrating agent. Suitable penetrating agents are exemplified by glycol ethers, which are commercially available from The Dow Chemical Company and include DOWANOL™ DPM, TPM, PPh, EPh, Methyl CARBITOL™, and Butyl
CARBITOL™.
[0072] When selecting starting materials to add to the aqueous emulsion described above in step (1) and the emulsion formulation formed described above, there may be overlap between types of starting materials because certain starting materials described herein may have more than one function. The starting materials used in aqueous emulsion and/or the emulsion formulation, may be distinct from one another.
[0073] The emulsion formulation suitable for treating a textile comprises: may be formulated with stalling materials that are fluorocarbon-free. For example, the emulsion formulation may be free of any starting material that contains a fluorine atom covalently bonded to a carbon atom. [0074] An additional starting material may optionally be used in the process described above. For example, a chelating agent such as ethylene diamine tetraacetate may be used when making the copolymer.
Process for Treating Textiles
[0075] The emulsion formulation prepared as described above may be used for treating a textile. For example, a method for treating a textile comprises: I) coating the textile with the emulsion formulation described above, and II) heating the textile. Step I) may be performed by any convenient method, such as padding, dipping, or spraying the textile with the emulsion formulation. However, the method should be sufficient to deliver on fabric weight of 0.25 weight % to 10 weight % of (I), the silicone - (meth) aery late copolymer, and on fabric weight of 0.1 weight % to 3.75 weight %, alternatively 0.25% to 1%, of (IV), the water dispersible crosslinker, both based on weight of the textile.
[0076] Step II) may be performed by any convenient method, such as placing the textile in an oven. Heating the textile may be performed to remove all or a portion of the water and/or cure the emulsion formulation. The exact temperature depends on various factors including the temperature sensitivity of the type of textile selected and the desired drying time. However, heating may be performed at a temperature > 100 °C to remove water. Alternatively, the temperature may be > 100 °C to 200 °C for a time sufficient to remove all or a portion of the water, de-block the blocked isocyanate crosslinker, and/or cure (I) the silicone - (meth)acrylate copolymer.
[0077] The textile to be treated is not specifically restricted. Suitable textiles include naturally derived textiles such as fabrics of cotton, silk, linen, and/or wool; textiles derived from synthetic
sources such as rayon, acetate, polyesters, polyamides (such as Nylons), polyacrylonitriles, and polyolefins such as polyethylenes and/or polypropylenes, and combinations of two or more thereof (e.g., blends such as polyester/cotton blend). The form of the textile is also not specifically restricted. The emulsion formulation described herein is suitable for use on textiles in any form, e.g., woven fabrics, knitted fabrics, or nonwoven textiles.
Durable water repellent testing:
[0078] The Bundesmann test may be carried out using the ISO 9865 method. An SDL Atlas M230 Bundesmann test apparatus may be used to collect face rating data after 0, 1, 10, and 20 washes. The instrument may be calibrated so that each cup collected between 190 and 210mL after 150s. Face Ratings may be assigned based on the images in the ISO 9865 method using half increments as intermediate assignments. Face Ratings may be repeated and reported as averages. A passing value may be > 4.5 to 5.
PROBLEMS TO BE ADDRESSED
[0079] There is an industry need for non-fluorocarbon-based textile treatments that impart durable water repellency to various fabrics. Furthermore, there is an industry need for fluorocarbon-free solutions with high durability as measured by the Bundesmann test, described above. There is a further need to produce commercial scale quantities of non-fluorocarbon-based textile treatments and to stabilize the monomer emulsion at higher temperatures for long processing times while maintaining excellent performance properties.
INDUSTRIAL APPLICABILITY
[0080] Without wishing to be bound by theory, it is thought that the emulsion formulation and process described herein can produce a fabric having durable water repellency as measured by the Bundesmann test described herein. More specifically, the fabric can have Bundesmann face rating > 4.5 after initial treatment and can maintain the face rating > 4.5 after 1, 5, 10, and 20 washes. Without wishing to be bound by theory, it is thought that using the manganese ion source and the phenolic compound, will lead to better monomer emulsion stability and will impart improved post polymerization emulsion formulation water repellency to fabric (as compared to an monomer emulsions that did not contain one or both of the manganese ion source and the phenolic compound).
DEFINITIONS AND USAGE OF TERMS
[0081] All amounts, ratios, and percentages herein are by weight, unless otherwise indicated. The SUMMARY and ABSTRACT arc hereby incorporated by reference. The articles, “a”, “an”, and “the” each refer to one or more, unless otherwise indicated by the context of the specification. The transitional phrases “comprising”, “consisting essentially of’, and “consisting of’ are used as described in the Manual of Patent Examining Procedure Ninth Edition, Revision 08.2017, Last Revised January 2018 at section §2111.03 I., II., and III. The use of “for example,” “e.g.,” “such as,” and “including” to list illustrative examples does not limit to only the listed examples. Thus, “for example” or “such as” means “for example, but not limited to” or “such as, but not limited to” and encompasses other similar or equivalent examples. The abbreviations used herein have the definitions in Table 5.
[0082] The invention has been described in an illustrative manner, and it is to be understood that the terminology which has been used is intended to be in the nature of words of description rather than of limitation. With respect to any Markush groups relied upon herein for describing
particular features or aspects, different, special, and/or unexpected results may be obtained from each member of the respective Markush group independent from all other Markush members. Each member of a Markush group may be relied upon individually and or in combination and provides adequate support for specific embodiments within the scope of the appended claims. [0083] Furthermore, any ranges and subranges relied upon in describing the present invention independently and collectively fall within the scope of the appended claims, and are understood to describe and contemplate all ranges including whole and/or fractional values therein, even if such values are not expressly written herein. One of skill in the art readily recognizes that the enumerated ranges and subranges sufficiently describe and enable various embodiments of the present invention, and such ranges and subranges may be further delineated into relevant halves, thirds, quarters, fifths, and any other subrange subsumed within the range. As just one example, a range of “16 to 24” may be further delineated into a lower third, i.e., 16 to 18, a middle third, i.e., 19 to 21, and an upper third, i.e., from 22 to 24, and alternatively, the range “16 to 24” includes the subrange “18 to 22”, each which individually and collectively are within the scope of the appended claims, and may be relied upon individually and/or collectively and provide adequate support for specific embodiments within the scope of the appended claims. In addition, with respect to the language which defines or modifies a range, such as “at least,” “greater than,” “less than,” “no more than,” and the like, it is to be understood that such language includes subranges and/or an upper or lower limit.
Claims
1. A composition comprising:
(I) a silicone - (meth) acrylate copolymer comprising unit formula
each R1 is an independently selected alkyl group of 16 to 24 carbon atoms; each R2 is independently selected from the group consisting of H and methyl; each D2 is a divalent hydrocarbon group of 2 to 12 carbon atoms; each R3 is independently selected from the group consisting of R4 and a group of formula -OSi(R4)3; each R4 is an independently selected monovalent hydrocarbon group of 1 to 12 carbon atoms; each R7 is independently selected from the group consisting of an oxygen atom and NH;
D3 is a divalent hydrocarbon group of 1 to 12 carbon atoms; each D4 is an alkylene group of 2 to 4 carbon atoms or a divalent alkylarylene group; subscript v represents the number of units of formula (OD4) in the unit with subscript y, subscript v has a value of 0 to 12; each R8 is a crosslinkable group;
each R5 is oxygen atom;
D is a divalent hydrocarbon group of 1 to 12 carbon atoms; each D1 is an alkylene group of 2 to 4 carbon atoms or a divalent alkylarylene group; subscript v2 represents the number of units of formula (OD1) in the unit with subscript y2, subscript v2 has a value of 0 to 20; each R6 is an alkoxy group or a hydroxy group; each R9 is a monovalent hydrocarbon group of 1 to 14 carbon atoms; each R10 is independently selected from the group consisting of a halogen, an acetate group, or a monovalent hydrocarbon group of 1 to 14 carbon atoms; subscripts w, x, y, y2, zl, and z2 represent relative weights of each unit in the copolymer, where a quantity (w + x + y + y2 + zl + z2) = 100,
80 < w < 98,
1 < x < 5, l < y < 5,
0 < y2 < 5,
0 < zl < 18, and
0 < z2 < 18; a manganese ion source, and a phenolic compound.
2. The composition of claim 1, wherein the manganese ion source comprises manganese (II) acetate, manganese (II) acetate tetrahydrate, or a combination thereof.
3. The composition of claim 1 or claim 2, wherein the phenolic compound is selected from the group consisting of hydroquinone, monomethyl ether of hydroquinone, tert-butylhydroquinone, and a combination of two or more thereof.
4. A process for preparing the composition of any one of claims 1 to 3, wherein the process comprises:
(1) copolymerizing starting materials comprising
1 weight % to 15 weight % of (B) a silicone - (mcth)acrylatc macromonomcr of formula
4 are as described above;
1 weight % to 5 weight % of (C) a crosslinkable (meth) acrylate monomer of formula
subscript v are as described above; where amounts of starting materials (A), (B), and (C) total 100 weight% based on combined amounts of starting materials (A), (B), and (C); and starting materials (A), (B), and (C) are copolymerized in the presence of additional starting materials, wherein the additional starting materials comprise: optionally a chain transfer agent; an initiator; optionally an additional monomer distinct from starting materials (A), (B), and (C); and a manganese ion source, and
a phenolic compound; thereby forming the composition comprising the silicone - (mcth)acrylatc copolymer, the manganese ion source, and the phenolic compound.
5. The process of claim 4, further comprising adding a solvent during step 1).
6. The process of claim 5, further comprising: removing the solvent after forming the copolymer, and forming an aqueous emulsion comprising the silicone - (meth)acrylate copolymer, the manganese ion source, the phenolic compound, a surfactant, and water.
7. The process of claim 4, wherein step 1) comprises an emulsion polymerization reaction; the additional starting materials further comprise a surfactant and water; and the product of step 1) comprises an aqueous emulsion comprising the silicone - (meth)acrylate copolymer, the manganese ion source, the phenolic compound, the surfactant, and water.
8. The process of claim 6 or claim 7, where starting material (A) is selected from the group consisting of stearyl acrylate, stearyl methacrylate, behenyl methacrylate and behenyl acrylate.
9. The process of any one of claims 6 to 8, where starting material (B) is selected from the group consisting of 3-(l,l,l,3,5,5,5-heptamethyltrisiloxan-3-yl)propyl methacrylate; 3-(l, 1,1, 5,5,5- hexamethyl-3-((trimethylsilyl)oxy)trisiloxan-3-yl)propyl methacrylate; and a combination thereof.
10. The process of any one of claims 6 to 9, where in starting material (C), where each R8 is independently selected from the group consisting of hydroxy, amino, epoxy, ureido, and acetoxy.
11. The process of any one of claims 6 to 10, where starting material (C) is 2-
hydroxyethylmethacrylate.
12. An emulsion formulation suitable for treating a textile, where the emulsion formulation comprises:
(I) a silicone - (meth)acrylate copolymer comprising unit formula
each R1 is an independently selected alkyl group of 16 to 24 carbon atoms; each R2 is independently selected from the group consisting of H and methyl; each D2 is a divalent hydrocarbon group of 2 to 12 carbon atoms; each R3 is independently selected from the group consisting of R4 and a group of formula -Osi(R4)3; each R4 is an independently selected monovalent hydrocarbon group of 1 to 12 carbon atoms; each R7 is independently selected from the group consisting of an oxygen atom and NH;
D3 is a divalent hydrocarbon group of 1 to 12 carbon atoms; each D4 is an alkylene group of 2 to 4 carbon atoms or a divalent alkylarylene group; subscript v represents the number of units of formula (OD4) in the unit with subscript y, subscript v has a value of 0 to 12;
each R8 is a crosslinkable group; each R5 is oxygen atom;
D is a divalent hydrocarbon group of 1 to 12 carbon atoms; each D1 is an alkylene group of 2 to 4 carbon atoms or a divalent alkylarylene group; subscript v2 represents the number of units of formula (OD1) in the unit with subscript y2, subscript v2 has a value of 0 to 20; each R6 is an alkoxy group or a hydroxy group; each R9 is a monovalent hydrocarbon group of 1 to 14 carbon atoms; each R10 is independently selected from the group consisting of a halogen, an acetate group, or a monovalent hydrocarbon group of 1 to 14 carbon atoms; subscripts w, x, y, y2, zl, and z2 represent relative weights of each unit in the copolymer, where a quantity (w + x + y + y2 + zl + z2) = 100, 80 < w < 98, 1 < x < 5, l < y < 5, 0 < y2 < 5, 0 < zl < 18, and 0 < z2 < 18; a manganese ion source; a phenolic compound;
(II) a nonionic surfactant comprising an alcohol alkoxylate of formula
where R19 is a branched or linear alkyl group with 8 to 15 carbon atoms, subscript n is at least 23, and each D5 is an independently selected alkylene oxide group of 2 or more carbon atoms;
(III) water; and
(IV) a water dispersible crosslinker;
With the proviso that the emulsion formulation is free of cationic surfactant.
13. The emulsion formulation of claim 12, where R1 is a stearyl group; 83 < w < 93; R3 is
selected from the group consisting of -CH3 and -OSi(CHa)3; R4 is methyl; D2 is -(C kh-; 5 < x < 15; R7 is O, subscript v = 0; R8 is OH; subscript y = 2; subscript zl= 0; and subscript z2 = 0.
14. The emulsion formulation of claim 12 or claim 13, further comprising an additional starting material selected from the group consisting of a wax, a biocide, a flame retardant, a wrinkle reducing agent, an antistatic agent, a penetrating agent or a combination of two or more thereof.
15. The emulsion formulation of claim 12 or claim 13, where (II) the nonionic surfactant comprises: a Cii-15 secondary alkyl alcohol ethoxylate, a 2-ethylhexyl primary alcohol alkoxylate, a C12-40 primary alcohol ethoxylate; or a combination thereof.
16. The emulsion formulation of claim 12 or claim 13, where (IV) the water dispersible crosslinker comprises a blocked isocyanate.
17. A method for preparing the emulsion formulation of claim 12 or claim 13, the method comprising:
(1) copolymerizing starting materials comprising
1 weight % to 15 weight % of (B) a silicone - (meth)acrylate macromonomer of formula.
1 weight % to 5 weight % of (C) a crosslinkable (meth) acrylate monomer of formula
subscript v are as described above; where amounts of starting materials (A), (B), and (C) total 100 weight% based on combined amounts of starting materials (A), (B), and (C); and starting materials (A), (B), and (C) are copolymerized in an emulsion further comprising a manganese ion source; a phenolic compound;
(D) a surfactant;
(TIT) water; an initiator; and thereby forming an aqueous emulsion comprising (I) the silicone - (meth)acrylate copolymer, (D) the surfactant and (E) the water; and
(2) combining the aqueous emulsion and an additional starting material comprising (TV) the water dispersible crosslinker.
18. The process of claim 17, where (D) the surfactant is not nonionic, and the method further comprises (3) removing (D) the surfactant and replacing with (IT) the nonionic surfactant.
19. The process of claim 17 or claim 18, further comprising adding an additional starting material in step (1), where the additional stalling material is selected from the group consisting of (G) a chain transfer agent; (H) an additional monomer that is distinct from starting materials (A), (B), and (C); (J) a reactive surfactant that is distinct from starting material (D); and a combination of two or more thereof.
21. A process for treating a textile, the process comprising:
I) coating the textile with the emulsion formulation of any one of claims 12 to 16; and
II) heating the textile.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US202363588050P | 2023-10-05 | 2023-10-05 | |
| US63/588,050 | 2023-10-05 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2025075700A1 true WO2025075700A1 (en) | 2025-04-10 |
Family
ID=92456509
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2024/039475 Pending WO2025075700A1 (en) | 2023-10-05 | 2024-07-25 | Silicone – (meth)acrylate copolymer composition and emulsion formulation for textile treatment |
Country Status (2)
| Country | Link |
|---|---|
| TW (1) | TW202515924A (en) |
| WO (1) | WO2025075700A1 (en) |
Citations (15)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4122029A (en) | 1977-07-27 | 1978-10-24 | Dow Corning Corporation | Emulsion compositions comprising a siloxane-oxyalkylene copolymer and an organic surfactant |
| US5387417A (en) | 1991-08-22 | 1995-02-07 | Dow Corning Corporation | Non-greasy petrolatum emulsion |
| US5811487A (en) | 1996-12-16 | 1998-09-22 | Dow Corning Corporation | Thickening silicones with elastomeric silicone polyethers |
| US6420504B1 (en) | 1997-06-12 | 2002-07-16 | Dow Corning Toray Silicone Co., Ltd. | Branched siloxane/silalkylene copolymer, silicone-containing organic polymer, and process for producing the same |
| US6576051B2 (en) | 2000-11-21 | 2003-06-10 | Rohm And Haas Company | Polymer-pigment composites |
| US9212292B2 (en) | 2013-07-30 | 2015-12-15 | Rohm And Haas Company | Ureido-functionalized aqueous polymeric dispersion |
| US9480977B2 (en) | 2011-09-20 | 2016-11-01 | Dow Corning Corporation | Ruthenium containing hydrosilylation catalysts and compositions containing the catalysts |
| US20170204558A1 (en) | 2014-07-04 | 2017-07-20 | Archroma Ip Gmbh | Fluorine-free water-repellent composition |
| US9777105B2 (en) | 2015-05-18 | 2017-10-03 | Shin-Etsu Chemical Co., Ltd. | Blocked polyisocyanate-containing curable silicone composition and textile treatment using the same |
| JP2017218713A (en) * | 2017-04-21 | 2017-12-14 | 明成化学工業株式会社 | Water repellent containing no fluorine, water repellent processing method, water repellent fiber product |
| US10047199B2 (en) | 2009-12-24 | 2018-08-14 | Dow Corning Toray Co., Ltd. | Copolymer having carbosiloxane dendrimer structure, and composition and cosmetic containing the same |
| US20190382580A1 (en) * | 2017-02-27 | 2019-12-19 | Showa Denko K.K. | Curable aqueous resin emulsion composition |
| WO2020142388A1 (en) | 2018-12-31 | 2020-07-09 | Dow Silicones Corporation | Composition, method of preparing copolymer, and methods and end uses thereof |
| US10844151B2 (en) | 2016-09-23 | 2020-11-24 | Huntsman Textile Effects (Germany) Gmbh | Fluorine-free aqueous dispersions for the finishing of textile fabrics |
| WO2023019044A1 (en) | 2021-08-10 | 2023-02-16 | Dow Silicones Corporation | Silicone-(meth)acrylate copolymer emulsion formulation for textile treatment |
-
2024
- 2024-07-25 WO PCT/US2024/039475 patent/WO2025075700A1/en active Pending
- 2024-09-02 TW TW113133052A patent/TW202515924A/en unknown
Patent Citations (15)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4122029A (en) | 1977-07-27 | 1978-10-24 | Dow Corning Corporation | Emulsion compositions comprising a siloxane-oxyalkylene copolymer and an organic surfactant |
| US5387417A (en) | 1991-08-22 | 1995-02-07 | Dow Corning Corporation | Non-greasy petrolatum emulsion |
| US5811487A (en) | 1996-12-16 | 1998-09-22 | Dow Corning Corporation | Thickening silicones with elastomeric silicone polyethers |
| US6420504B1 (en) | 1997-06-12 | 2002-07-16 | Dow Corning Toray Silicone Co., Ltd. | Branched siloxane/silalkylene copolymer, silicone-containing organic polymer, and process for producing the same |
| US6576051B2 (en) | 2000-11-21 | 2003-06-10 | Rohm And Haas Company | Polymer-pigment composites |
| US10047199B2 (en) | 2009-12-24 | 2018-08-14 | Dow Corning Toray Co., Ltd. | Copolymer having carbosiloxane dendrimer structure, and composition and cosmetic containing the same |
| US9480977B2 (en) | 2011-09-20 | 2016-11-01 | Dow Corning Corporation | Ruthenium containing hydrosilylation catalysts and compositions containing the catalysts |
| US9212292B2 (en) | 2013-07-30 | 2015-12-15 | Rohm And Haas Company | Ureido-functionalized aqueous polymeric dispersion |
| US20170204558A1 (en) | 2014-07-04 | 2017-07-20 | Archroma Ip Gmbh | Fluorine-free water-repellent composition |
| US9777105B2 (en) | 2015-05-18 | 2017-10-03 | Shin-Etsu Chemical Co., Ltd. | Blocked polyisocyanate-containing curable silicone composition and textile treatment using the same |
| US10844151B2 (en) | 2016-09-23 | 2020-11-24 | Huntsman Textile Effects (Germany) Gmbh | Fluorine-free aqueous dispersions for the finishing of textile fabrics |
| US20190382580A1 (en) * | 2017-02-27 | 2019-12-19 | Showa Denko K.K. | Curable aqueous resin emulsion composition |
| JP2017218713A (en) * | 2017-04-21 | 2017-12-14 | 明成化学工業株式会社 | Water repellent containing no fluorine, water repellent processing method, water repellent fiber product |
| WO2020142388A1 (en) | 2018-12-31 | 2020-07-09 | Dow Silicones Corporation | Composition, method of preparing copolymer, and methods and end uses thereof |
| WO2023019044A1 (en) | 2021-08-10 | 2023-02-16 | Dow Silicones Corporation | Silicone-(meth)acrylate copolymer emulsion formulation for textile treatment |
Non-Patent Citations (3)
| Title |
|---|
| "Manual of Patent Examining Procedure", January 2018 |
| ODIAN, GEORGE: "Principles of Polymerization", 2004, WILEY-INTERSCIENCE |
| PETER NESVADBA: "Encyclopedia of Radicals in Chemistry, Biology and Materials", 2012, JOHN WILEY & SONS, LTD, article "Radical Polymerization in Industry" |
Also Published As
| Publication number | Publication date |
|---|---|
| TW202515924A (en) | 2025-04-16 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP4384560B1 (en) | Silicone-(meth)acrylate copolymer emulsion formulation for textile treatment | |
| US20240068158A1 (en) | Silicone - (meth)acrylate copolymer formulation and processes for preparation and use thereof | |
| KR20240097929A (en) | Textile treatment emulsion formulations comprising silicone-(meth)acrylate copolymers and silicone additives | |
| JP5439743B2 (en) | Water and oil repellent | |
| EP3464401B1 (en) | Aqueous polymer dispersion and preparation method thereof | |
| EP3594305A1 (en) | Oil-repellent composition | |
| WO2025075700A1 (en) | Silicone – (meth)acrylate copolymer composition and emulsion formulation for textile treatment | |
| CN116096958A (en) | Silicone polyether polymer treatment for fibrous substrates | |
| WO2025075701A1 (en) | Silicone – (meth)acrylate copolymer composition containing a silicone additive and methods for the preparation and use thereof | |
| WO2025075699A1 (en) | Silicone – (meth)acrylate copolymer formulation and processes for preparation and use thereof | |
| WO2025090192A1 (en) | Textile treatment using non-crystalline silicone – (meth)acrylate copolymer, crystalline (meth)acrylate based (co)polymer and blocked isocyanate | |
| WO2025090193A1 (en) | Textile treatment with silicone – (meth)acrylate (co)polymer, poly[poly(alkylene glycol) (meth)acrylate], and blocked isocyanate | |
| KR20260002711A (en) | Silicone-(meth)acrylate copolymer emulsion and method for producing the same, and use of the emulsion for imparting oil repellency to fabrics | |
| JP6651970B2 (en) | Fiber sizing agent | |
| WO2025090189A1 (en) | Silicone – (meth)acrylate copolymer emulsion and preparation thereof and use of the emulsion to impart oil repellency to textiles | |
| WO2025183828A1 (en) | Textile treatment composition containing a silicone – (meth)acrylate copolymer and methods for the preparation and use thereof | |
| CN109503742B (en) | Silicon Hybrid Polyacrylate Dispersion and Its Application | |
| WO2025090822A1 (en) | Treating paper with composition including silicone - (meth)acrylate copolymer | |
| JPWO2018180538A1 (en) | Water repellant for fabric and fabric | |
| WO2025226624A1 (en) | Treating paper with silicone – (meth)acrylate copolymer | |
| JP2000136495A (en) | Composition for treating paper and treated paper |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 24758071 Country of ref document: EP Kind code of ref document: A1 |