WO2025073561A1 - Herbicide/safener combination based on seed treatment with safeners belonging to the class of substituted [(1,5-diphenyl-1h-1,2,4-triazol-3-yl)oxy]acetic acid and salts thereof - Google Patents
Herbicide/safener combination based on seed treatment with safeners belonging to the class of substituted [(1,5-diphenyl-1h-1,2,4-triazol-3-yl)oxy]acetic acid and salts thereof Download PDFInfo
- Publication number
- WO2025073561A1 WO2025073561A1 PCT/EP2024/077001 EP2024077001W WO2025073561A1 WO 2025073561 A1 WO2025073561 A1 WO 2025073561A1 EP 2024077001 W EP2024077001 W EP 2024077001W WO 2025073561 A1 WO2025073561 A1 WO 2025073561A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- methyl
- formula
- oxy
- difluorophenyl
- fluorophenyl
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Classifications
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N25/00—Biocides, pest repellants or attractants, or plant growth regulators, characterised by their forms, or by their non-active ingredients or by their methods of application, e.g. seed treatment or sequential application; Substances for reducing the noxious effect of the active ingredients to organisms other than pests
- A01N25/32—Ingredients for reducing the noxious effect of the active substances to organisms other than pests, e.g. toxicity reducing compositions, self-destructing compositions
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/64—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with three nitrogen atoms as the only ring hetero atoms
- A01N43/647—Triazoles; Hydrogenated triazoles
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/72—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with nitrogen atoms and oxygen or sulfur atoms as ring hetero atoms
- A01N43/80—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with nitrogen atoms and oxygen or sulfur atoms as ring hetero atoms five-membered rings with one nitrogen atom and either one oxygen atom or one sulfur atom in positions 1,2
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01P—BIOCIDAL, PEST REPELLANT, PEST ATTRACTANT OR PLANT GROWTH REGULATORY ACTIVITY OF CHEMICAL COMPOUNDS OR PREPARATIONS
- A01P13/00—Herbicides; Algicides
- A01P13/02—Herbicides; Algicides selective
Definitions
- the invention relates to novel methods for preventing crop damage, which comprise seed treatment of a crop seed with a safener compound based on a substituted [(l,5-diphenyl-lH-l,2,4-triazol-3-yl)oxy]acetic acid or agrochemically acceptable salt thereof, followed by application of a herbicidal compound based on a substituted isoxazolincarboxamide or agrochemically acceptable salt thereof.
- a safener compound based on a substituted [(l,5-diphenyl-lH-l,2,4-triazol-3-yl)oxy]acetic acid or agrochemically acceptable salt thereof
- a herbicidal compound based on a substituted isoxazolincarboxamide or agrochemically acceptable salt thereof.
- Substituted isoxazolincarboxamides are already known as effective herbicides from WO2018/228985 and WO2019/145245.
- the activity of these compounds and/or their compatibility with crop plants are not entirely satisfactory under all conditions, such that the use of different safeners has been considered, e.g. described in W02021/245003, W02021/245004, W02021/245005, W02021/245006, W02021/245007, W02021/245008, W02021/245009.
- Substituted [(l,5-diphenyl-lH-l,2,4-triazol-3-yl)oxy]acetic acids and salts thereof are a novel class of safeners first described in W02021/105101.
- Combinations of substituted [(l,5-diphenyl-lH-l,2,4-triazol-3- yl)oxy]acetic acids with different classes of herbicides are furthermore described in WO2022/238194.
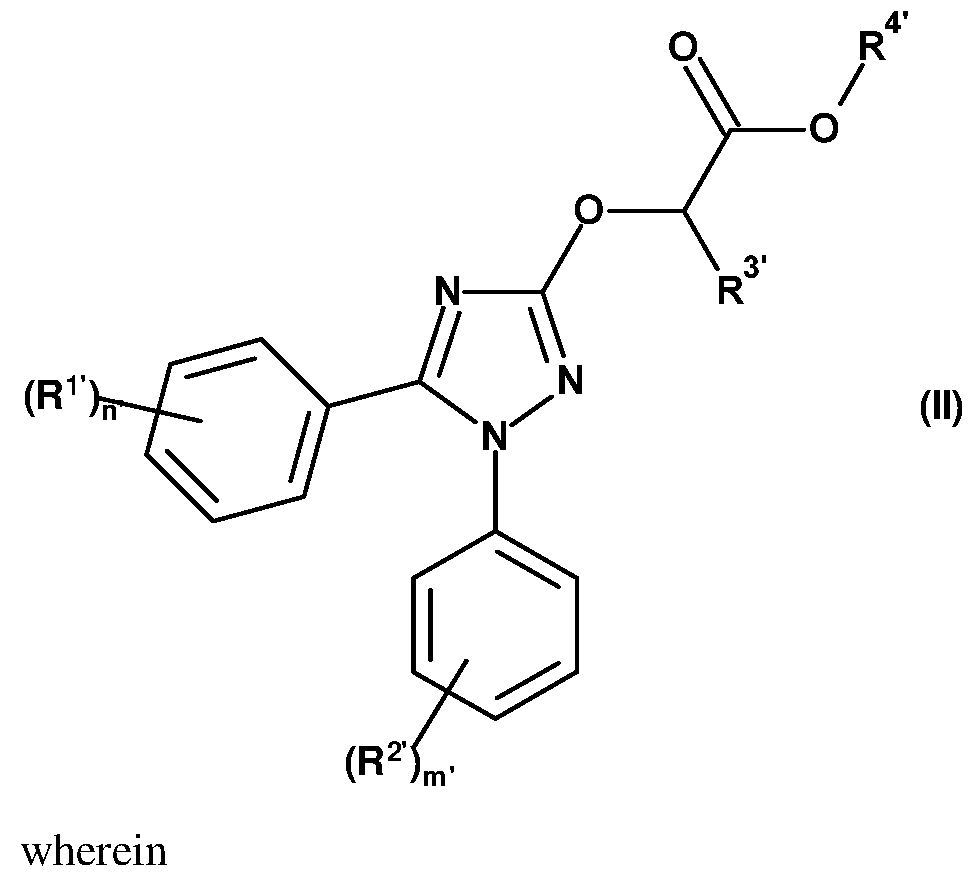
- the present invention thus provides a method for reducing crop damage, comprising treating seeds of the crop with a safener compound of formula (II) before sowing,
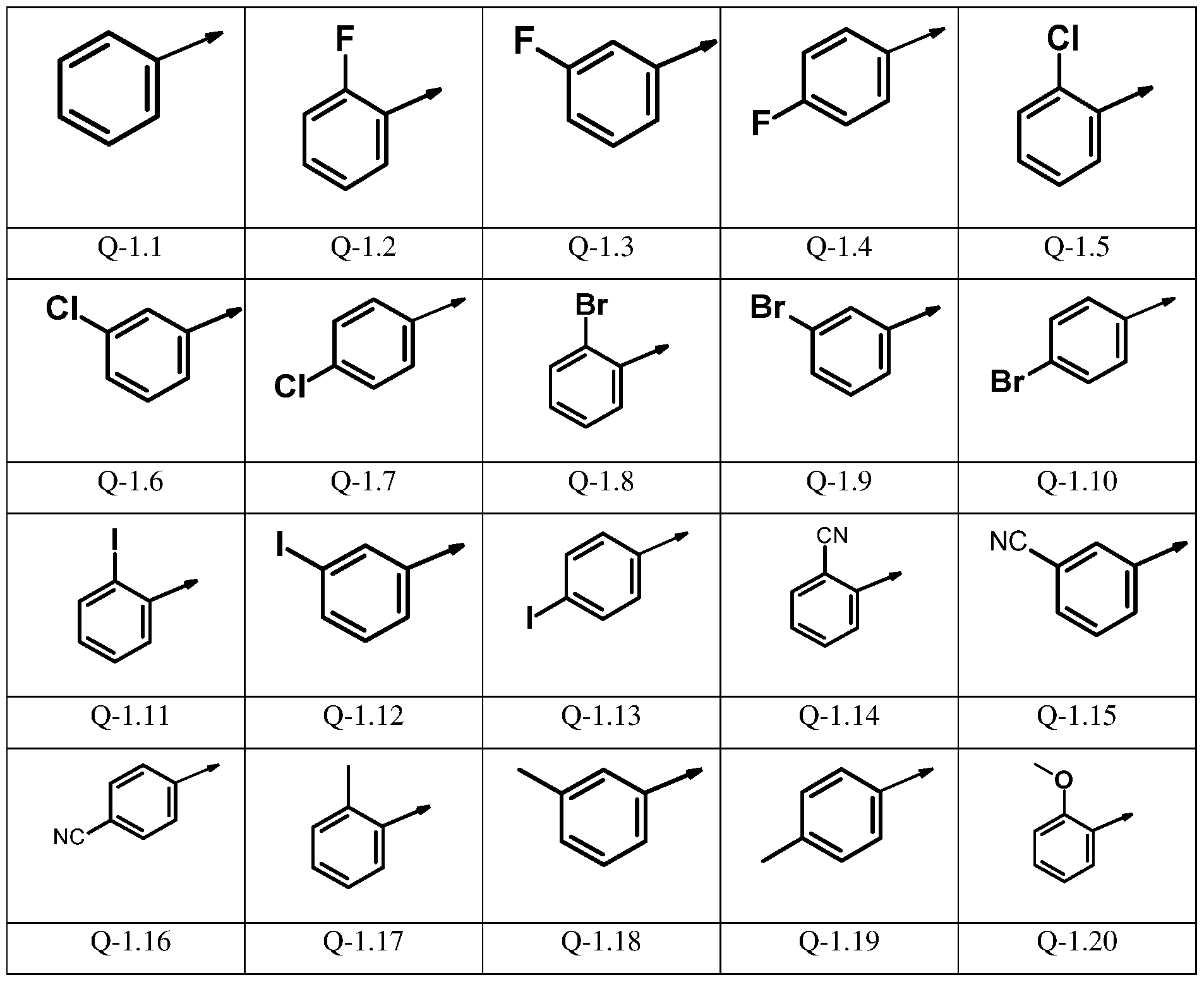
- (R 1 ) n ’ -phenyl is presented by one of the groups Q-l.l to Q-1.55 and (R 2 ) m -phenyl is selected from one of the groups Q-2.1 to Q-2.55
- R 3 is hydrogen
- R 4 is hydrogen, methyl, ethyl, n-propyl, i-propyl, n-butyl, n-pentyl, phenyl, benzyl, CH2(4-Cl-Ph), CH2(4-F-Ph), CH2(4-OMe-Ph), 2-methoxyethyl, tetrahydrofuran-2-yl-methyl, tetrahydrofuran-3-yl-methyl, tetrahydropyran-2-yl-methyl, tetrahydropyran-3-yl-methyl, tetrahydropyran-4-yl-methyl, methylpropionat-3-yl, ethylpropionat-3-yl, methylacetat-2-yl, ethylacetat-2-yl, methylpivalat-2-yl, ethylpivalat-3-yl, methyl-2-methylpropanoat-3-yl, methyl-2
- the compound of formula (I) is a compound of formula (la) or an agrochemically acceptable salt thereof, in which
- X 3 , X 5 , R 3 and G are as described above;
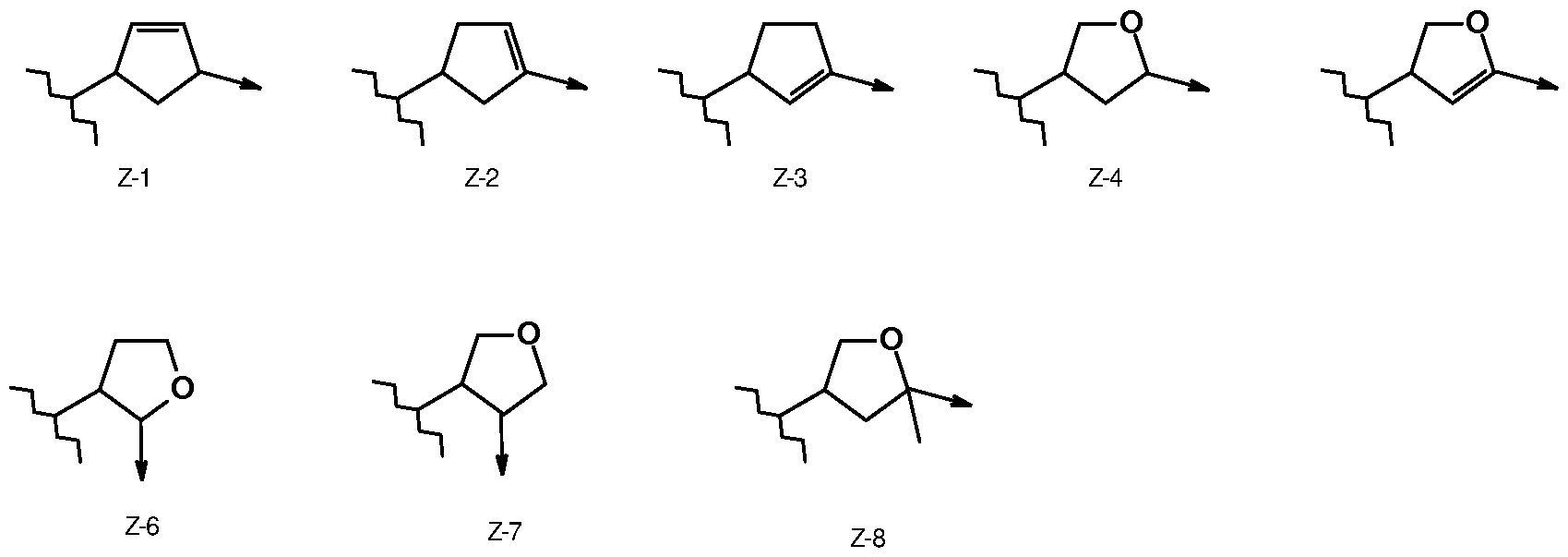
- Z means Z-la, Z-lb, Z-2a, Z-3a, Z-4a, Z-5a, Z-6a, Z-7a, or Z-8a,
- Z-8a wherein Z-4a means the mixture of both structures Z-4b and Z-4c and wherein Z-8a means the mixture of both structures Z-8b and Z-8c and wherein the arrow means a bond to the group CO-G in formula (la).
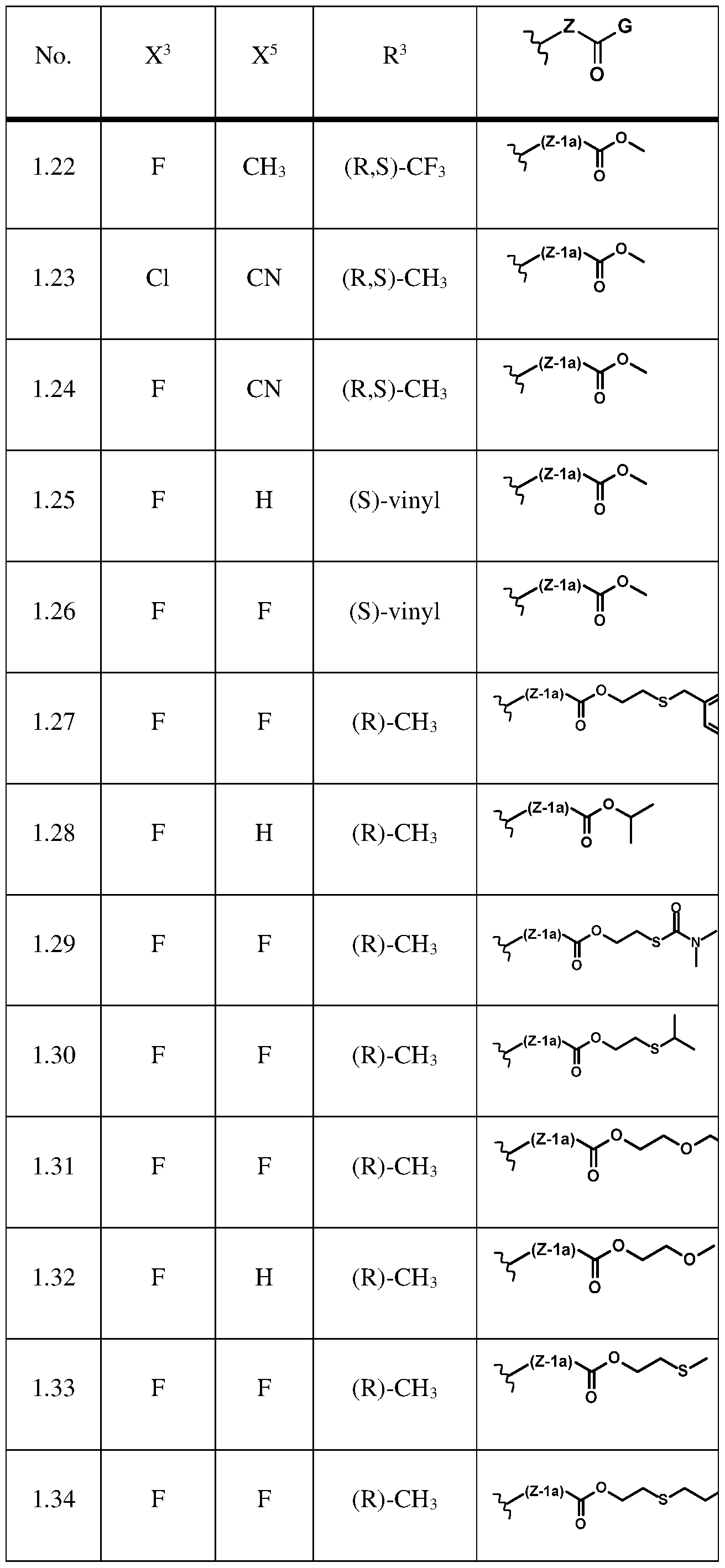
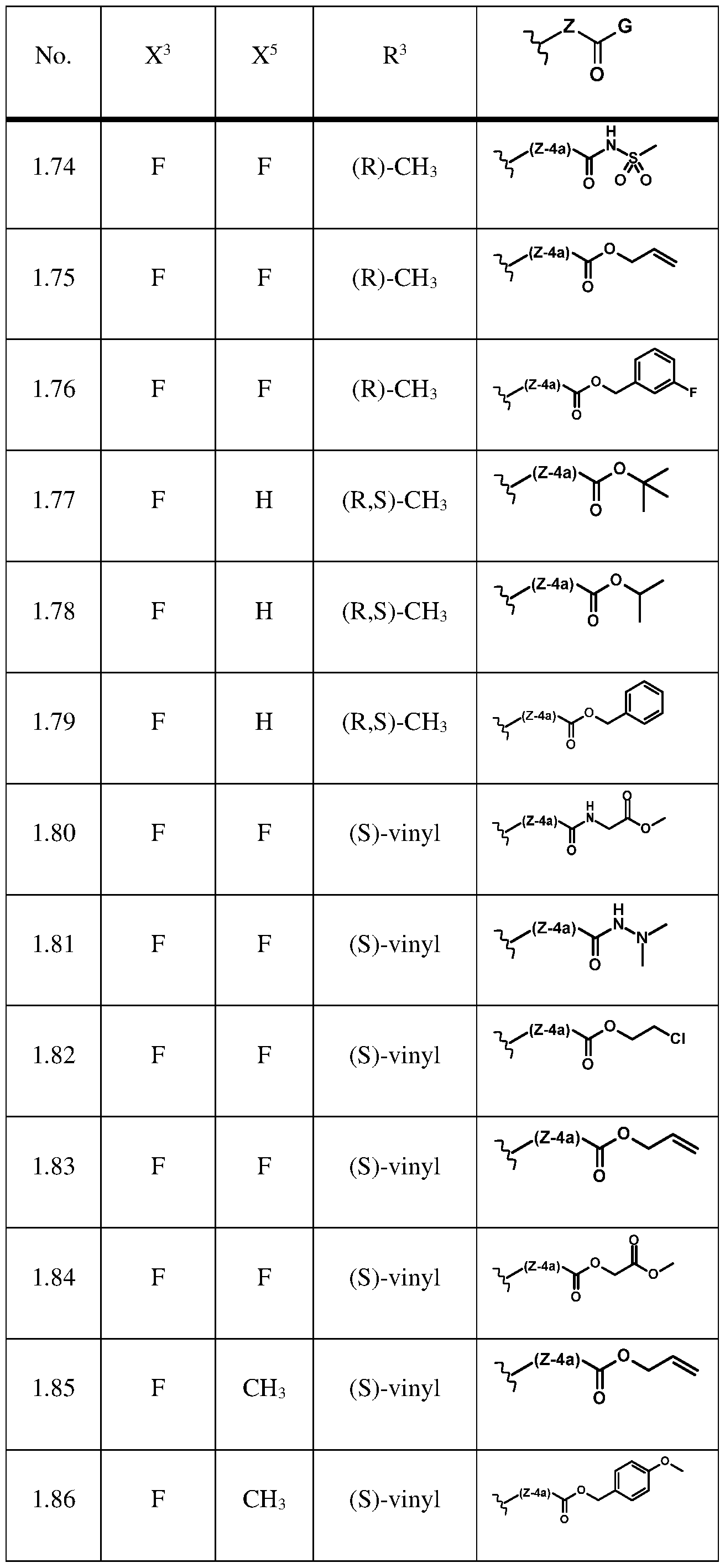
- the compound of formula (la) is selected from the compounds of below table 1 or agrochemically acceptable salts thereof.
- the compounds of formula (I) are known from WO2018/228985 and WO2019/145245 and can be synthesized according to the processes described therein. Even further preferably, the compound of formula (I) is selected from
- the compound of formula (II) is selected from
- Such compounds of formula (II) are for example described in W02021/105101 and can be synthesized according to the processes described therein.
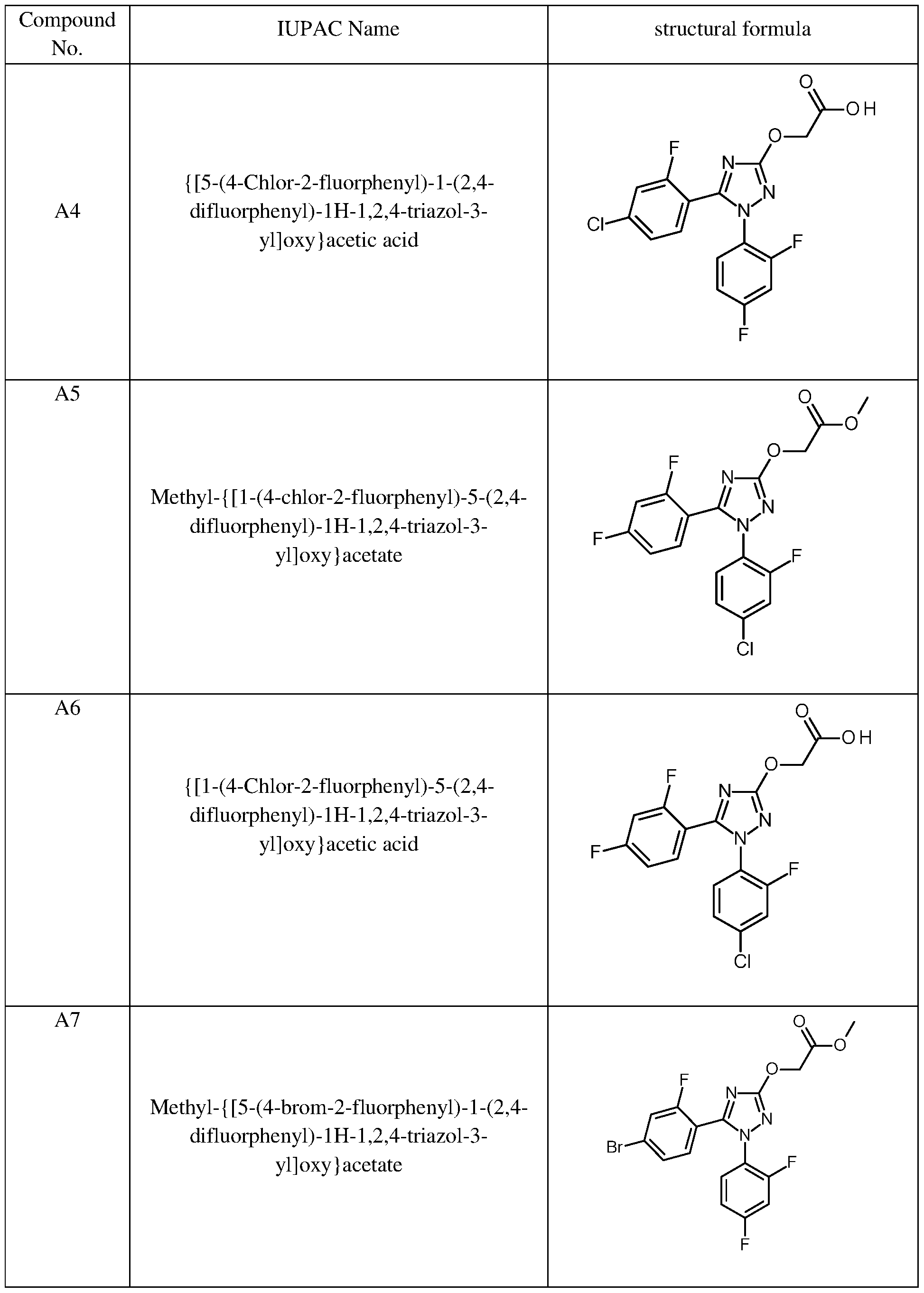
- preferred safener compounds of formula (II) in the context of the present invention are compounds Al, A3, A5, A7 and A9 of the previous table 2.
- the herbicidal compound of formula (I) is Methyl (2R* ,4R*)-4- [ [(5 S) -3 -(3 ,5 -difluorophenyl)-5 - vinyl-4H-isoxazol-5 -carbonyl] amino] tetrahydrofuran- 2-carboxylate; and the safener compound of formula (II) is selected from
- the herbicidal compound of formula (I) is
- the herbicidal compound of formula (I) is applied in a pre-emergent treatment.
- the herbicidal compound of formula (I) is applied in a post-emergent treatment, preferably in an early or mid post-emergent treatment.
- the crop is a genetically modified plant.
- the safener compound (II) is applied to the seeds in an application rate of between 0.01 and 2 g per kg seed, preferably between 0.1 and 1 g per kg seed, further preferably between 0.2 and 0.8 g per kg seed, even further preferably between 0.2 and 0.5 g per kg seed.
- the herbicidal compound of formula (I) is applied at an application rate of between 0.1 and 1000 g per ha, preferably between 0.5 and 500 g per ha, further preferably between 0.7 and 100 g per ha, even further preferably between 1 and 50 g per ha.
- the method further comprises applying the safener compound of formula (II) after sowing in a in a pre-emergent or post-emergent treatment.
- the safener compound of formula (II) is applied in a tank-mix together with the herbicidal compound of formula (I).
- the safener compound of formula (II) is applied in a tank-mix together with the herbicidal compound of formula (I) at an application rate of between 1 and 1000 g per ha, preferably between 5 and 500 g per ha, further preferably between 10 and 100 g per ha, even further preferably between 25 and 75 g per ha.
- Halogen represents radicals of fluorine, chlorine, bromine and iodine. Preference is given to the radicals of fluorine and chlorine.
- Alkyl means saturated straight-chain or branched hydrocarbyl radicals having the number of carbon atoms specified in each case, e.g. Ci-Ce-alkyl such as methyl, ethyl, propyl, 1 -methylethyl, butyl, 1- methylpropyl, 2-methylpropyl, 1,1 -dimethylethyl, pentyl, 1 -methylbutyl, 2-methylbutyl, 3-methylbutyl,
- Alkenyl means unsaturated straight-chain or branched hydrocarbyl radicals having the number of carbon atoms specified in each case and one double bond in any position, e.g. Cz-Ce-alkenyl such as ethenyl, 1- propenyl, 2-propenyl, 1 -methylethenyl, 1-butenyl, 2-butenyl, 3-butenyl, 1 -methyl- 1 -propenyl, 2-methyl- 1 -propenyl, l-methyl-2-propenyl, 2-methyl-2-propenyl, 1 -pentenyl, 2-pentenyl, 3-pentenyl, 4-pentenyl, 1 -methyl- 1-butenyl, 2-methyl- 1-butenyl, 3-methyl- 1-butenyl, l-methyl-2-butenyl, 2-methyl-2-butenyl, 3-methyl-2-butenyl, l-methyl-3-butenyl, 2-methyl-3-butenyl, 3-
- Alkynyl represents a straight-chain or branched hydrocarbyl groups having 2 to 8, preferably 2 to 6, carbon atoms and one triple bond in any position.
- Non-limiting examples include ethynyl, prop-l-ynyl, prop-2-ynyl, but-l-ynyl, but-2-ynyl, but-3-ynyl, l-methylprop-2-ynyl, pent-l-ynyl, pent-2-ynyl, pent-3- ynyl, pent-4-ynyl, l-methylbut-2-ynyl, l-methylbut-3-ynyl, 2-methylbut-3-ynyl, 3-methylbut-l-ynyl, l,l-dimethylprop-2-ynyl, l-ethylprop-2-ynyl, hex-l-ynyl, hex-2-ynyl, hex-3-ynyl
- Cycloalkyl means a carbocyclic saturated ring system having preferably 3-8 ring carbon atoms, for example cyclopropyl, cyclobutyl, cyclopentyl or cyclohexyl.
- cyclic systems with substituents are included, also including substituents with a double bond on the cycloalkyl radical, for example an alkylidene group such as methylidene.
- Alkoxy means saturated straight-chain or branched alkoxy radicals having the number of carbon atoms specified in each case, for example Ci-Ce-alkoxy such as methoxy, ethoxy, propoxy, 1 -methylethoxy, butoxy, 1 -methylpropoxy, 2-methylpropoxy, 1,1 -dimethylethoxy, pentoxy, 1 -methylbutoxy, 2- methylbutoxy, 3-methylbutoxy, 2,2-dimethylpropoxy, 1 -ethylpropoxy, hexoxy, 1,1 -dimethylpropoxy,
- Ci-Ce-alkoxy such as methoxy, ethoxy, propoxy, 1 -methylethoxy, butoxy, 1 -methylpropoxy, 2-methylpropoxy, 1,1 -dimethylethoxy, pentoxy, 1 -methylbutoxy, 2- methylbutoxy, 3-methylbutoxy, 2,2-dimethylpropoxy, 1 -ethylpropoxy, hexoxy, 1,1 -dimethylprop
- Halogen-substituted alkoxy means straight-chain or branched alkoxy radicals having the number of carbon atoms specified in each case, where some or all of the hydrogen atoms in these groups may be replaced by halogen atoms as specified above, e.g.
- Ci- Cz-haloalkoxy such as chloromethoxy, bromomethoxy, dichloromethoxy, trichloromethoxy, fluoromethoxy, difluoromethoxy, trifluoromethoxy, chlorofluoromethoxy, dichlorofluoromethoxy, chlorodifluoromethoxy, 1 -chloroethoxy, 1 -bromoethoxy, 1 -fluoroethoxy, 2-fluoroethoxy, 2,2- difluoroethoxy, 2,2,2-trifluoroethoxy, 2-chloro-2-fluoroethoxy, 2-chloro-l,2-difluoroethoxy, 2,2- dichloro-2-fluoroethoxy, 2,2,2-trichloroethoxy, pentafluoroethoxy and l,l,l-trifluoroprop-2-oxy.
- the safenening effect of the compounds of formula (II) is higher compared to the safening effect of other known known safeners or antidotes which are generally capable of antagonizing the damaging effect of a herbicide on the crop plants, when followed by herbicidal treatment with substituted isoxazolincarboxamides of the general formula (I).
- Emphasis is given here to the particularly advantageous effect of a crop seed treatment with compounds of formula (II) with respect to sparing cereal plants, such as, for example, wheat, barley and rye, but also maize and rice, as crop plants.
- the active compound combinations according to the invention can be used, for example, in connection with the following plants:
- Dicotyledonous crops of the genera Gossypium, Glycine, Beta, Daucus, Phaseolus, Pisum, Solanum, Linum, Ipomoea, Vicia, Nicotiana, Lycopersicon, Arachis, Brassica, Lactuca, Cucumis, Cuburbita, Helianthus.
- Monocotyledonous crops of the genera Oryza, Zea, Triticum, Hordeum, Avena, Secale, Sorghum, Panicum, Saccharum, Ananas, Asparagus, Allium.
- crop plants are all plants and plant varieties including transgenic plants and plant varieties, where on transgenic plants and plant varieties it is also possible for synergistic effects to occur.
- compositions within the context of the present invention comprise in addition to the herbicide compound of formula (I) according to the invention one or more further component(s) which include, but are not limited to the following: formulation auxiliaries, additives customary in crop protection, further agrochemically active compounds (e.g. fungicides and insecticides). Such compositions may also be applied in the method according to the invention.
- the method according to the present invention comprises applying at least one further herbicide (c) wherein (c) is chosen from the list which includes but is not limited to the following: Acetochlor, acifluorfen, acifluorfen-methyl, acifluorfen-sodium, aclonifen, alachlor, allidochlor, alloxydim, alloxydim-sodium, ametryn, amicarbazone, amidochlor, amidosulfuron, 4-amino-3-chloro-6- (4-chloro-2-fluoro-3-methylphenyl)-5-fluoropyridine-2-carboxylic acid, aminocyclopyrachlor, aminocyclopyrachlor-potassium, aminocyclopyrachlor-methyl, aminopyralid, aminopyralid- dimethylammonium, aminopyralid- tripromine, amitrole, ammoniumsulfamate, anilofos, asulam, asulam-potassium, asulam sodium
- dicamba-biproamine dicamba-N,N-Bis(3-aminopropyl)methylamine, dicamba-butotyl, dicamba-choline, dicamba-digly colamine, dicamba-dimethylammonium, dicamba- diethanolaminemmonium, dicamba-diethylammonium, dicamba-isopropylammonium, dicamba-methyl, dicamba-monoethanolaminedicamba-olamine, dicamba-potassium, dicamba-sodium, dicamba- triethanolamine, dichlobenil, 2-(2,4-dichlorobenzyl)-4,4-dimethyl- 1 ,2-oxazolidin-3-one, 2-(2,5- dichlorobenzyl)-4,4-dimethyl- 1 ,2-oxazolidin-3-one, dichlorprop, dichlorprop-butotyl, dichlroprop- dimethylammonium, dichhlorprop-
- COs sometimes referred to as N-acetylchitooligosaccharides, are also composed of GlcNAc residues but have side chain decorations that make them different from chitin molecules [(CsHoNOsln, CAS No. 1398-61-4] and chitosan molecules [(CsHnNOrln, CAS No.
- chitinous compounds chlormequat chloride, cloprop, cyclanilide, 3-(Cycloprop-l-enyl)propionic acid, daminozide, dazomet, dazomet-sodium, n-decanol, dikegulac, dikegulac-sodium, endothal, endothal-dipotassium, -disodium, and mono(N,N-dimethylalkylammonium), ethephon, flumetralin, flurenol, flurenol-butyl, flurenol- methyl, flurprimidol, forchlorfenuron, gibberellic acid, inabenfide, indol-3-acetic acid (IAA), 4-indol-3- ylbutyric acid, isoprothiolane, probenazole, jasmonic acid, Jasmonic acid or derivatives thereof (e.g.
- LCO lipo-chitooligosaccharides
- Nod symbiotic nodulation
- Myc factors consist of an oligosaccharide backbone of P-l,4-linked N-acetyl-D-glucosamine (“GlcNAc”) residues with an N-linked fatty acyl chain condensed at the non-reducing end.
- LCOs differ in the number of GlcNAc residues in the backbone, in the length and degree of saturation of the fatty acyl chain and in the substitutions of reducing and non-reducing sugar residues), linoleic acid or derivatives thereof, linolenic acid or derivatives thereof, maleic hydrazide, mepiquat chloride, mepiquat pentaborate, 1 -methylcyclopropene, 3 ’-methyl abscisic acid, 2-(l-naphthyl)acetamide, 1 -naphthylacetic acid, 2- naphthyloxyacetic acid, nitrophenolate-mixture, 4-Oxo-4[(2-phenylethyl)amino]butyric acid, paclobutrazol, 4-phenylbutyric acid, N-phenylphthalamic acid, prohexadione, prohexadione-calcium, prohydrojasmon, salicylic
- Pre-emergent application refers to a herbicide which is applied to an area of interest (e.g., a field or area of cultivation) before a plant emerges visibly from the soil.
- Post-emergent application refers to a herbicide which is applied to an area after a plant emerges visibly from the soil.
- pre- emergent and post-emergent may be defined via the BBCH code.
- the BBCH code commonly known to the person skilled in the art, provides information on the stage of morphological development of a plant.
- the abbreviation officially represents the Federal Department of Biology, Federal Register of Plant Varieties and Chemical Industry.
- PE prior to seed germination or up to BBCH stage 09
- Post-emergent (PO) application on the aboveground parts of the plant; BBCH stages 10-31.
- Late post-emergent BBCH stages 29-31.
- the BBCH code may apply to both the emergence of a weed or of a planted crop.
- the BBCH code refers to the emergence of the crop. Examples:
- TRZAW winter wheat
- the safener compound A3 (see table 2 above) according to the invention, as well as the comparative safeners mefenpyr-diethyl (CAS 135590-91-9) and cloquintocet-mexyl (CAS 99607- 70-2) were used.
- each safener was tank-mixed with water.
- the water is used to ensure right kernel distribution of the compound on it.
- the slurry volume (water+safener) is 800 ml/100 kg seeds.
- the seeds to be treated were put in a mixing chamber of the equipment for seed treatment. Once seeds are placed in the equipment, the slurry volume was introduced in the mixing chamber for a duration of 20 seconds (to complete the seed treatment). Treated seeds were collected then from the mixing chamber and dried in a paper bag.
- the dose rate of the seed treatment was 50 g safener / 100 kg seeds.
- Seeds of the eight varieties of TRZAW treated with safeners as described under 1. above as well as untreated seeds were planted in strips in the field with 4 rows of TRZAW per each of the treatments in 4 trials around Europe (DE, FR, PL, IT).
- % crop damage from the herbicide application observed on the different strips was scored visually in comparison to control strips (that had not received a herbicide application but containing seed treatment and not seed treatment).
- [Average crop damage] % (herbicide damage on strips (with and without safener seed treatment) treated with the herbicide - herbicide damage on strips (with and without safener seed treatment) non treated with the herbicide) *100
- the herbicidal activity (%) on Alopecurus myosuroides (ALOMY) and Avena sativa (AVESA) was determined by the overall difference (%) of the biomass and other symptoms (i.e. chlorosis, necrosis, stunting) between the treated plots with the herbicide and the untreated plots.
- the safener compound A3 provides for a higher safening function of the crop compared to the safeners mefenpyr-diethyl or cloquintocet-mexyl, as shown in the tables above. At the same time, the herbicidal activity of HER is not affected by the safener compound A3. B. Efficacy of different modes of safener application
- test plants were treated post-emergent in growth stage BBCH11.
- HER Metal (2R*,4R*)-4-[[(5S)-3-(3,5-difluorophenyl)-5-vinyl-4H-isoxazol-5- carbonyl] amino] tetrahydrofuran-2-carboxylate: EC 10g/l
- the application rate of HER was 2 g /ha; the application rate of compound A3 was 50 g/ha.
- the plants were further cultivated as described above. 28 days after application, the herbicide damage was scored visually. The damage was evaluated visually on a scale of 0-100% in comparison to control plants without herbicide and safener treatment.
- the crop damage could be unexpectedly reduced by applying the safener as seed treatment when compared to tank- mixing the safener with the herbicides.
- An even further unexpected reduction of crop damage was achieved by applying the safener twice, namely both in terms of seed treatment and as tank-mix partner to the herbicide.
Landscapes
- Life Sciences & Earth Sciences (AREA)
- Environmental Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Engineering & Computer Science (AREA)
- Health & Medical Sciences (AREA)
- Pest Control & Pesticides (AREA)
- Plant Pathology (AREA)
- Wood Science & Technology (AREA)
- Zoology (AREA)
- Dentistry (AREA)
- Agronomy & Crop Science (AREA)
- General Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Chemical & Material Sciences (AREA)
- Toxicology (AREA)
- Agricultural Chemicals And Associated Chemicals (AREA)
Abstract
The invention relates to novel methods of reducing crop damage comprising seed treatment applications of safeners followed by pre- or post-emergent treatment with herbicidally acting substituted isoxazolincarboxamides.
Description
Herbicide/safener combination based on seed treatment with safeners belonging to the class of substituted r(l,5-diphenyl-lH-l,2,4-triazol-3-yl)oxy1acetic acid and salts thereof
The invention relates to novel methods for preventing crop damage, which comprise seed treatment of a crop seed with a safener compound based on a substituted [(l,5-diphenyl-lH-l,2,4-triazol-3-yl)oxy]acetic acid or agrochemically acceptable salt thereof, followed by application of a herbicidal compound based on a substituted isoxazolincarboxamide or agrochemically acceptable salt thereof. The combination of seed treated crops and herbicidal treatment after planting allows for a highly selective control of weeds in various crops of useful plants.
Substituted isoxazolincarboxamides are already known as effective herbicides from WO2018/228985 and WO2019/145245. However, the activity of these compounds and/or their compatibility with crop plants are not entirely satisfactory under all conditions, such that the use of different safeners has been considered, e.g. described in W02021/245003, W02021/245004, W02021/245005, W02021/245006, W02021/245007, W02021/245008, W02021/245009.
Substituted [(l,5-diphenyl-lH-l,2,4-triazol-3-yl)oxy]acetic acids and salts thereof are a novel class of safeners first described in W02021/105101. Combinations of substituted [(l,5-diphenyl-lH-l,2,4-triazol-3- yl)oxy]acetic acids with different classes of herbicides are furthermore described in WO2022/238194.
While different options to safen crops from the herbicidal activity of substituted isoxazolincarboxamides have been suggested in the art, there is still a need to further improve the safening methods and thereby to decrease the level of crop damage.
Surprisingly, it has now been found that seeds of crops treated with substituted [( 1,5-diphenyl-lH- 1,2,4- triazol-3-yl)oxy] acetic acids and salts thereof can be effectively combined with a herbicide treatment with certain substituted isoxazolincarboxamides. The seed treatment provides for a high safening effect on the crop, preventing its damage extremely well, while the herbicidal action on unwanted weeds remains high.
The present invention, thus provides a method for reducing crop damage, comprising treating seeds of the crop with a safener compound of formula (II) before sowing,
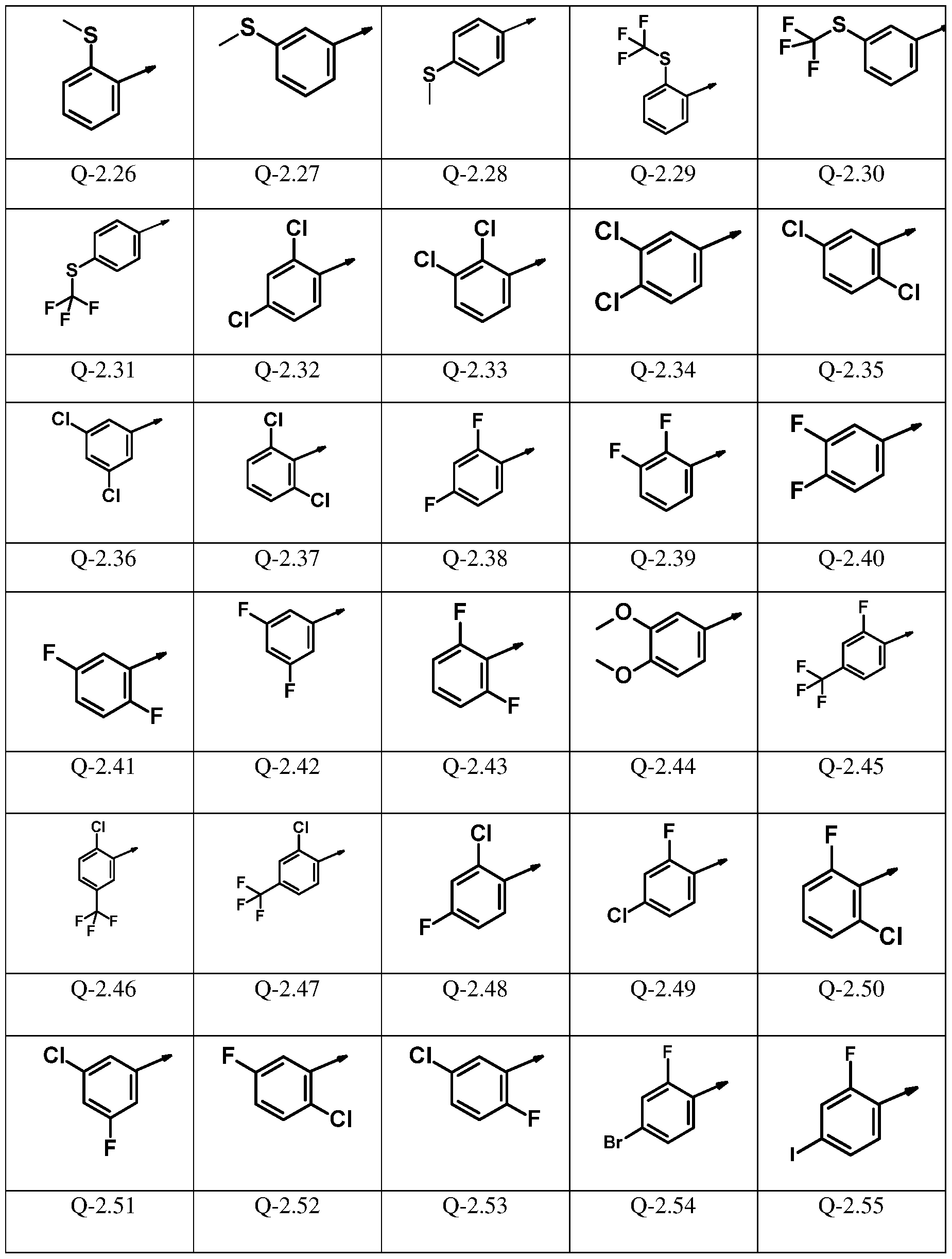
(R1 )n’ -phenyl is presented by one of the groups Q-l.l to Q-1.55
and (R2 )m -phenyl is selected from one of the groups Q-2.1 to Q-2.55
R3 is hydrogen, and
R4 is hydrogen, methyl, ethyl, n-propyl, i-propyl, n-butyl, n-pentyl, phenyl, benzyl, CH2(4-Cl-Ph), CH2(4-F-Ph), CH2(4-OMe-Ph), 2-methoxyethyl, tetrahydrofuran-2-yl-methyl, tetrahydrofuran-3-yl-methyl, tetrahydropyran-2-yl-methyl, tetrahydropyran-3-yl-methyl, tetrahydropyran-4-yl-methyl, methylpropionat-3-yl, ethylpropionat-3-yl, methylacetat-2-yl, ethylacetat-2-yl, methylpivalat-2-yl, ethylpivalat-3-yl, methyl-2-methylpropanoat-3-yl, methyl-2,2-dimethylpropanoat-3-yl, ethyl-2-methylpropanoat-3-yl, methyl-2-propanoat-2-yl, ethyl-2-propanoat-2-yl, methyl-acetat-2yl, ethyl-acetat-2yl, methyl- 1- methylcyclopropancarboxylat-2yl, ethyl- 1 -methylcyclopropan-carboxylat-2yl, 2- (dimethylamino)ethyl, oxetan-3-yl, (3-methyloxetan-3-yl)methyl, 2,2,2-trifluorethyl, 2,2- difluorethyl, 2-fluorethyl, 2,2,3,3,3-pentafluorpropyl, cyclopropylmethyl, 1 -cyclopropyl-
ethyl, (1-methyl-cyclopropyl)-methyl, (2,2-dichlorcyclopropyl)-methyl, (2,2-dimethyl- cyclopropyl)-methyl, allyl, propargyl (prop-2-in-1-yl), 2-chlorprop-2-en-1-yl, 3-phenylprop- 2-in-1-yl, 3,3-dichlorprop-2-en-1-yl, 3,3-dichlor-2-fluor-prop-2-en-1-yl, methylprop-2-in-1- yl, 2-methylprop-2-en-1-yl, but-2-en-1-yl, but-3-en-1-yl, but-2-in-1-yl, but-3-in-1-yl, 4-chlor- but-2-in-1-yl, 3-methyl-but-2-en-1-yl, 3-methyl-but-1-en-1-yl, 1- (2e)-1-methylbut-2-en-1-yl, (e)-pent-3-en-2-yl or (z)-pent-3-en-2-yl, cyclobutylmethyl, cyclopentylmethyl, cyclohexylmethyl, heptan-2-yl, iso-butyl, 1,3-dioxolan-2-ylmethyl oder 1-ethyl-5-methyl- 1hpyrazol-4-methyl; followed by applying a herbicidal compound of formula (I) or agrochemical acceptable salt thereof after sowing,
in which G represents OR4 or NR7R8; R1 and R2 each represent hydrogen; R3 represents (C1-C5)-alkyl, (C3-C6)-cycloalkyl, (C2-C5)-alkenyl, (C2-C5)-alkinyl or (C1-C5)- alkoxy each optionally substitued “m” times by substituents from the group consisting of halogen, cyano, (C1-C5)-alkoxy and hydroxy; R4 represents hydrogen, or represents (C1-C12)-alkyl, (C3-C7)-cycloalkyl, (C3-C7)-cycloalkyl-(C1-C8)-alkyl, (C2-C8)- alkenyl, (C5-C6)-cycloalkenyl, (C1-C4)-alkylphenyl or (C2-C8)-alkinyl each optionally substitued “m” times by substituents from the group consisting of halogen, cyano, (C1-C6)- alkoxy, (C1-C6)-alkoxycarbonyl, hydroxy, S(O)n R5; R5 represents (C1-C8)-alkyl, (C2-C8)-alkenyl, (C3-C6)-cycloalkyl, benzyl, CON((C1-C3)-alkyl)2 or (C1-C8)-alkyl-C(O)-(C1-C8)-alkyl each optionally substitued “m” times by substituents from the group consisting of halogen and cyano; R6 represents hydrogen,
or represents (C1-C8)-alkyl, (C3-C6)-cycloalkyl, (C3-C8)-alkenyl or (C3-C8)-alkinyl each optionally substitued “m” times by substituents from the group consisting of halogen, cyano und (C1-C2)-alkoxy; R7, R8 independently of each other represent hydrogen, (C1-C6)-alkoxycarbonyl-(C1-C6)- alkyl, N((C1-C3)-alkyl)2, S(O)n R5, or R7 and R8 together with the nitrogen atom to which they are attached form a saturated or partially or fully unsaturated five-, six-, or seven-membered ring which may contain apart from the nitrogen ataom “r” carbon atoms, “o” oxygen atoms and is optionally substitued “m” times by substituents from the group consisting of halogen, (C1-C6)-alkyl, halogen-(C1-C6)-alkyl, oxo, CO2R6; Z represents a residue selected from any of Z-1 to Z-8: 0
whereas the arrow represents the bonding to the group CO-G of the formula (I); X2, X4 and X6 independently of one another represent hydrogen or fluorine; X3 and X5 independently of one another represent hydrogen, chlorine, cyano or fluorine; or represent (C1-C3)-Alkyl, (C1-C3)-Alkoxy each optionally substitued “m” times by substituents from the group consisting of fluorine or chlorine; m represents 0, 1, 2, 3, 4 or 5; n represents 0, 1 or 2; o represents 0, 1 or 2;
r represents 3, 4, 5 or 6.
Preferably, the compound of formula (I) is a compound of formula (la) or an agrochemically acceptable salt thereof,
in which
X3, X5, R3 and G are as described above;
Z-8a wherein Z-4a means the mixture of both structures Z-4b and Z-4c
and wherein Z-8a means the mixture of both structures Z-8b and Z-8c
and wherein the arrow means a bond to the group CO-G in formula (la).
Further preferably, the compound of formula (la) is selected from the compounds of below table 1 or agrochemically acceptable salts thereof.
(la)
The compounds of formula (I) are known from WO2018/228985 and WO2019/145245 and can be synthesized according to the processes described therein. Even further preferably, the compound of formula (I) is selected from
Methyl rel-(2R,4R)-4-[[3-(3,5-dichlorophenyl)-5-methoxy-4H-isoxazol-5- carbonyl]amino]tetrahydrofuran-2-carboxylate,
Methyl rel-(2R,4R)-4-[[3-(3,5-dichlorophenyl)-5-vinyl-4H-isoxazol-5- carbonyl]amino]tetrahydrofuran-2-carboxylate,
Methyl (2R* ,4R*)-4- [ [(5 S) -3 -(3 ,5 -difluorophenyl)-5 - vinyl-4H-isoxazol-5 - carbonyl]amino]tetrahydrofuran-2-carboxylate,
Isopropyl rel-(2R,4R)-4-[[3-(3-fluorophenyl)-5-methyl-4H-isoxazol-5- carbonyl]amino]tetrahydrofuran-2-carboxylate,
Methyl (3R)-3-[[(5S)-3-(3,5-difluorophenyl)-5-vinyl-4H-isoxazol-5-carbonyl]amino]-2,3- dihydrofuran-5 -carboxylate, and
Methyl (3R)-3-[[(5R)-3-(3,5-difluorophenyl)-5-methyl-4H-isoxazol-5-carbonyl]amino]-2,3- dihydrofuran-5 -carboxylate.
In a further embodiment, the compound of formula (II) is selected from
Methyl- { [1 ,5-bis(4-chloro-2-fluorophenyl)- 1H- 1 ,2,4-triazole-3-yl]oxy } acetate,
{ [ 1 ,5-Bis(4-chloro-2-fluorophenyl)- 1H- 1 ,2,4-triazole-3-yl]oxy } acetic acid,
Methyl- { [5-(4-chloro-2-fluorophenyl)- 1 -(2,4-difluorophenyl)- 1H- 1 ,2,4-triazole-3- yl]oxy} acetate,
{ [5-(4-Chloro-2-fluorophenyl)- 1 -(2,4-difluorophenyl)-lH- 1 ,2,4-triazole-3-yl]oxy } acetic acid,
Methyl- { [ 1 -(4-chloro-2-fluorophenyl)-5-(2,4-difluorophenyl)- 1 H- 1 ,2,4-triazole-3- yl]oxy} acetate,
{ [l-(4-Chloro-2-fhiorophenyl)-5-(2,4-difluorophenyl)-lH-l,2,4-triazole-3-yl]oxy}acetic acid,
Methyl- { [5-(4-bromo-2-fluorophenyl)- 1 -(2,4-difluorophenyl)- 1H- 1 ,2,4-triazole-3- yl]oxy} acetate,
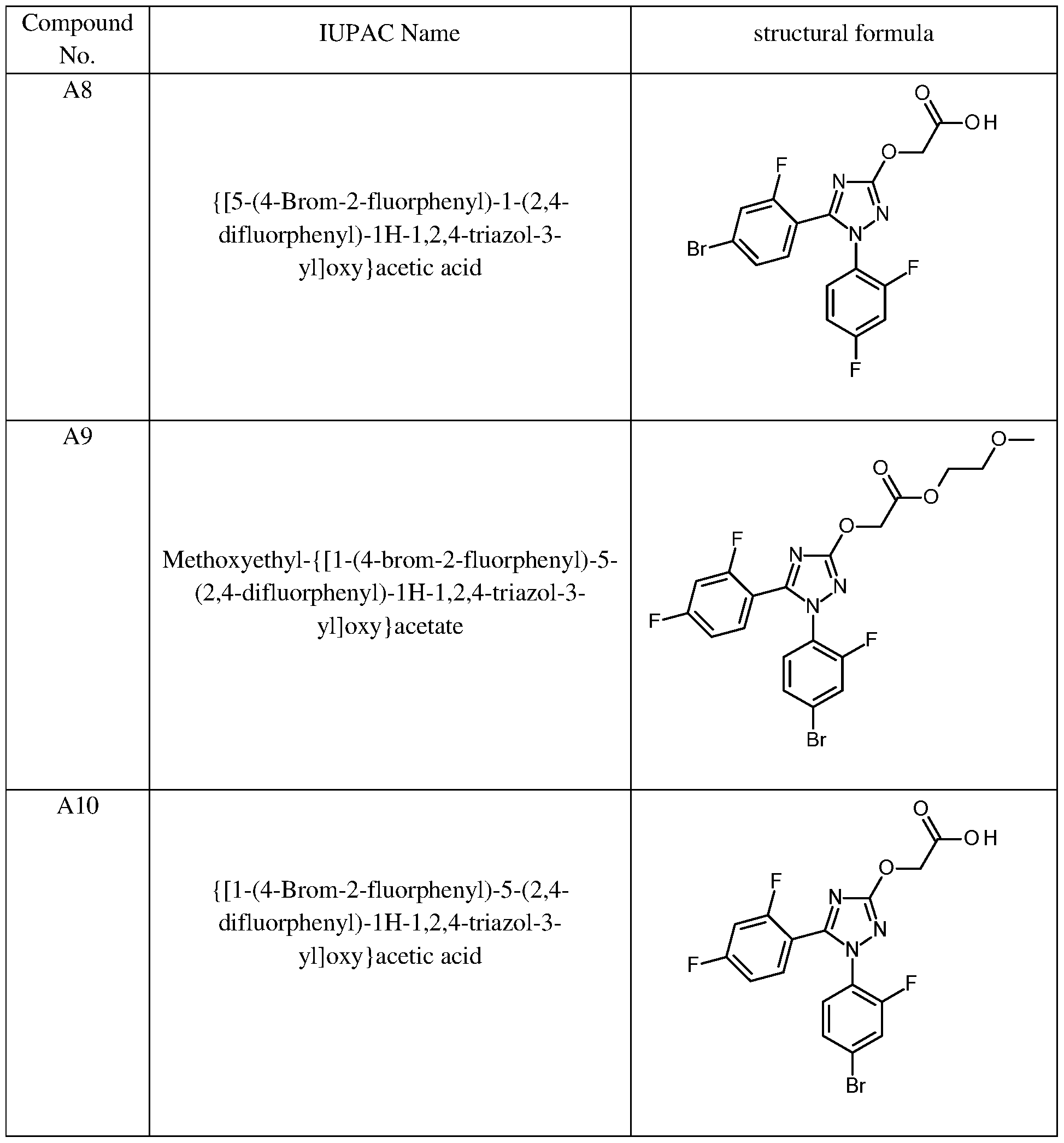
{ [5-(4-Bromo-2-fluorophenyl)- 1 -(2,4-difluorophenyl)- 1H- 1 ,2,4-triazole-3-yl]oxy } acetic acid,
Methoxyethyl- { [ 1 -(4-bromo-2-fluorophenyl)-5-(2,4-difluorophenyl)- 1H- 1 ,2,4-triazole-3- yl]oxy} acetate, and
{ [ l-(4-Bromo-2-fhrorophenyl)-5-(2,4-difluorophenyl)- 1H- 1 ,2,4-triazole-3-yl]oxy } acetic acid.
These preferred compounds of formula (II) are described in the following table 2.
Table 2: Preferred examples for compounds of the formula (II)
Such compounds of formula (II) are for example described in W02021/105101 and can be synthesized according to the processes described therein.
In particular preferred safener compounds of formula (II) in the context of the present invention are compounds Al, A3, A5, A7 and A9 of the previous table 2.
In a further embodiment, the herbicidal compound of formula (I) is
Methyl (2R* ,4R*)-4- [ [(5 S) -3 -(3 ,5 -difluorophenyl)-5 - vinyl-4H-isoxazol-5 -carbonyl] amino] tetrahydrofuran- 2-carboxylate; and the safener compound of formula (II) is selected from
Methyl- { [1 ,5-bis(4-chloro-2-fluorophenyl)- 1H- 1 ,2,4-triazole-3-yl]oxy } acetate,
Methyl- { [5-(4-chloro-2-fluorophenyl)- 1 -(2,4-difluorophenyl)- 1H- 1 ,2,4-triazole-3-yl]oxy } acetate, Methyl- { [1 -(4-chloro-2-fluorophenyl)-5-(2,4-difluorophenyl)- 1H- 1 ,2,4-triazole-3-yl]oxy } acetate, Methyl- { [5-(4-bromo-2-fluorophenyl)- 1 -(2,4-difluorphenyl)- 1H- 1 ,2,4-triazole-3-yl]oxy } acetate, and Methoxyethyl- { [ 1 -(4-bromo-2-fluorophenyl)-5-(2,4-difluorophenyl)- 1H- 1 ,2,4-triazole-3-yl]oxy } acetate.
In a further embodiment, the herbicidal compound of formula (I) is
Methyl (2R* ,4R*)-4- [ [(5 S) -3 -(3 ,5 -difluorophenyl)-5 - vinyl-4H-isoxazol-5 -carbonyl] amino] tetrahydrofuran- 2-carboxylate; and the safener compound of formula (II) is Methyl-] [5-(4-chlor-2-fluorphenyl)-l -(2,4- difluorphenyl)- 1 H- 1 ,2,4-triazol-3-yl] oxy } acetate.
In a further embodiment, the herbicidal compound of formula (I) is applied in a pre-emergent treatment.
In a further embodiment, the herbicidal compound of formula (I) is applied in a post-emergent treatment, preferably in an early or mid post-emergent treatment.
In a further embodiment, the crop is a genetically modified plant.
In a further embodiment, the safener compound (II) is applied to the seeds in an application rate of between 0.01 and 2 g per kg seed, preferably between 0.1 and 1 g per kg seed, further preferably between 0.2 and 0.8 g per kg seed, even further preferably between 0.2 and 0.5 g per kg seed.
In a further embodiment, the herbicidal compound of formula (I) is applied at an application rate of between 0.1 and 1000 g per ha, preferably between 0.5 and 500 g per ha, further preferably between 0.7 and 100 g per ha, even further preferably between 1 and 50 g per ha.
In a further embodiment, the method further comprises applying the safener compound of formula (II) after sowing in a in a pre-emergent or post-emergent treatment. Preferably, the safener compound of formula (II) is applied in a tank-mix together with the herbicidal compound of formula (I). Preferably, the safener compound of formula (II) is applied in a tank-mix together with the herbicidal compound of formula (I) at an application rate of between 1 and 1000 g per ha, preferably between 5 and 500 g per ha, further preferably between 10 and 100 g per ha, even further preferably between 25 and 75 g per ha.
By such dual application of the safener compound, i.e. both during seed treatment and (after sowing) in a pre- or post-emergent application, the safening action is even further increased.
Definitions
Halogen represents radicals of fluorine, chlorine, bromine and iodine. Preference is given to the radicals of fluorine and chlorine.
Alkyl means saturated straight-chain or branched hydrocarbyl radicals having the number of carbon atoms specified in each case, e.g. Ci-Ce-alkyl such as methyl, ethyl, propyl, 1 -methylethyl, butyl, 1- methylpropyl, 2-methylpropyl, 1,1 -dimethylethyl, pentyl, 1 -methylbutyl, 2-methylbutyl, 3-methylbutyl,
2.2-dimethylpropyl, 1 -ethylpropyl, hexyl, 1,1 -dimethylpropyl, l,2-dimethylpropyl,l -methylpentyl, 2- methylpentyl, 3-methylpentyl, 4-methylpentyl, 1,1 -dimethylbutyl, 1 ,2-dimethylbutyl, 1,3-dimethylbutyl,
2.2-dimethylbutyl, 2,3-dimethylbutyl, 3,3-dimethylbutyl, 1-ethylbutyl, 2-ethylbutyl, 1,1,2- trimethylpropyl, 1 ,2,2-trimethylpropyl, 1 -ethyl- 1 -methylpropyl and l-ethyl-2-methylpropyl.
Alkenyl means unsaturated straight-chain or branched hydrocarbyl radicals having the number of carbon atoms specified in each case and one double bond in any position, e.g. Cz-Ce-alkenyl such as ethenyl, 1- propenyl, 2-propenyl, 1 -methylethenyl, 1-butenyl, 2-butenyl, 3-butenyl, 1 -methyl- 1 -propenyl, 2-methyl- 1 -propenyl, l-methyl-2-propenyl, 2-methyl-2-propenyl, 1 -pentenyl, 2-pentenyl, 3-pentenyl, 4-pentenyl, 1 -methyl- 1-butenyl, 2-methyl- 1-butenyl, 3-methyl- 1-butenyl, l-methyl-2-butenyl, 2-methyl-2-butenyl, 3-methyl-2-butenyl, l-methyl-3-butenyl, 2-methyl-3-butenyl, 3-methyl-3-butenyl, l,l-dimethyl-2- propenyl, 1,2-dimethyl-l -propenyl, 1 ,2-dimethyl-2-propenyl, 1 -ethyl- 1 -propenyl, l-ethyl-2-propenyl, 1- hexenyl, 2-hexenyl, 3-hexenyl, 4-hexenyl, 5-hexenyl, 1 -methyl- 1 -pentenyl, 2-methyl- 1 -pentenyl, 3- methyl-1 -pentenyl, 4-methyl-l -pentenyl, l-methyl-2-pentenyl, 2-methyl-2-pentenyl, 3-methyl-2- pentenyl, 4-methyl-2-pentenyl, l-methyl-3-pentenyl, 2-methyl-3-pentenyl, 3-methyl-3-pentenyl, 4- methyl-3-pentenyl, l-methyl-4-pentenyl, 2-methyl-4-pentenyl, 3-methyl-4-pentenyl, 4-methyl-4- pentenyl, l,l-dimethyl-2-butenyl, l,l-dimethyl-3-butenyl, 1 ,2-dimethyl- 1-butenyl, 1 ,2-dimethyl-2- butenyl, l,2-dimethyl-3-butenyl, 1,3-dimethyl- 1-butenyl, l,3-dimethyl-2-butenyl, l,3-dimethyl-3- butenyl, 2,2-dimethyl-3-butenyl, 2, 3-dimethyl- 1-butenyl, 2,3-dimethyl-2-butenyl, 2,3-dimethyl-3- butenyl, 3,3-dimethyl-l-butenyl, 3,3-dimethyl-2-butenyl, 1 -ethyl- 1-butenyl, l-ethyl-2-butenyl, 1-ethyl- 3-butenyl, 2-ethyl- 1-butenyl, 2-ethyl-2-butenyl, 2-ethyl-3-butenyl, l,l,2-trimethyl-2-propenyl, 1-ethyl- l-methyl-2-propenyl, l-ethyl-2-methyl-l -propenyl and l-ethyl-2-methyl-2-propenyl.
Alkynyl represents a straight-chain or branched hydrocarbyl groups having 2 to 8, preferably 2 to 6, carbon atoms and one triple bond in any position. Non-limiting examples include ethynyl, prop-l-ynyl, prop-2-ynyl, but-l-ynyl, but-2-ynyl, but-3-ynyl, l-methylprop-2-ynyl, pent-l-ynyl, pent-2-ynyl, pent-3- ynyl, pent-4-ynyl, l-methylbut-2-ynyl, l-methylbut-3-ynyl, 2-methylbut-3-ynyl, 3-methylbut-l-ynyl, l,l-dimethylprop-2-ynyl, l-ethylprop-2-ynyl, hex-l-ynyl, hex-2-ynyl, hex-3-ynyl, hex-4-ynyl, hex-5-
ynyl, l-methylpent-2-ynyl, l-methylpent-3-ynyl, l-methylpent-4-ynyl, 2-methylpent-3-ynyl, 2- methylpent-4-ynyl, 3-methylpent-l-ynyl, 3-methylpent-4-ynyl, 4-methylpent-l-ynyl, 4-methylpent-2- ynyl, l,l-dimethylbut-2-ynyl, l,l-dimethylbut-3-ynyl, l,2-dimethylbut-3-ynyl, 2,2-dimethylbut-3-ynyl, 3,3-dimethylbut-l-ynyl, l-ethylbut-2-ynyl, l-ethylbut-3-ynyl, 2-ethylbut-3-ynyl and 1-ethyl-l- methylprop-2-ynyl.
Cycloalkyl means a carbocyclic saturated ring system having preferably 3-8 ring carbon atoms, for example cyclopropyl, cyclobutyl, cyclopentyl or cyclohexyl. In the case of optionally substituted cycloalkyl, cyclic systems with substituents are included, also including substituents with a double bond on the cycloalkyl radical, for example an alkylidene group such as methylidene.
Alkoxy means saturated straight-chain or branched alkoxy radicals having the number of carbon atoms specified in each case, for example Ci-Ce-alkoxy such as methoxy, ethoxy, propoxy, 1 -methylethoxy, butoxy, 1 -methylpropoxy, 2-methylpropoxy, 1,1 -dimethylethoxy, pentoxy, 1 -methylbutoxy, 2- methylbutoxy, 3-methylbutoxy, 2,2-dimethylpropoxy, 1 -ethylpropoxy, hexoxy, 1,1 -dimethylpropoxy,
1.2-dimethylpropoxy, 1 -methylpentoxy, 2-methylpentoxy, 3-methylpentoxy, 4-methylpentoxy, 1,1- dimethylbutoxy, 1 ,2-dimethylbutoxy, 1,3-dimethylbutoxy, 2,2-dimethylbutoxy, 2,3-dimethylbutoxy,
3.3-dimethylbutoxy, 1 -ethylbutoxy, 2-ethylbutoxy, 1,1,2-trimethylpropoxy, 1 ,2,2-trimethylpropoxy, 1- ethyl-1 -methylpropoxy and l-ethyl-2-methylpropoxy. Halogen-substituted alkoxy means straight-chain or branched alkoxy radicals having the number of carbon atoms specified in each case, where some or all of the hydrogen atoms in these groups may be replaced by halogen atoms as specified above, e.g. Ci- Cz-haloalkoxy such as chloromethoxy, bromomethoxy, dichloromethoxy, trichloromethoxy, fluoromethoxy, difluoromethoxy, trifluoromethoxy, chlorofluoromethoxy, dichlorofluoromethoxy, chlorodifluoromethoxy, 1 -chloroethoxy, 1 -bromoethoxy, 1 -fluoroethoxy, 2-fluoroethoxy, 2,2- difluoroethoxy, 2,2,2-trifluoroethoxy, 2-chloro-2-fluoroethoxy, 2-chloro-l,2-difluoroethoxy, 2,2- dichloro-2-fluoroethoxy, 2,2,2-trichloroethoxy, pentafluoroethoxy and l,l,l-trifluoroprop-2-oxy.
Surprisingly, it has been found that crop seeds treated with safener compounds of the general formula (II) are extremely well protected from crop damage upon application of herbicidal compounds, namely of substituted isoxazolincarboxamides of the general formula (I) and/or their salts.
Unexpectedly, the safenening effect of the compounds of formula (II) is higher compared to the safening effect of other known known safeners or antidotes which are generally capable of antagonizing the damaging effect of a herbicide on the crop plants, when followed by herbicidal treatment with substituted isoxazolincarboxamides of the general formula (I).
Emphasis is given here to the particularly advantageous effect of a crop seed treatment with compounds of formula (II) with respect to sparing cereal plants, such as, for example, wheat, barley and rye, but also maize and rice, as crop plants.
The active compound combinations according to the invention can be used, for example, in connection with the following plants:
Dicotyledonous weeds of the genera: Sinapis, Lepidium, Galium, Stellaria, Matricaria, Anthemis, Galinsoga, Chenopodium, Urtica, Senecio, Amaranthus, Portulaca, Xanthium, Convolvulus, Ipomoea, Polygonum, Sesbania, Ambrosia, Cirsium, Carduus, Sonchus, Solanum, Rorippa, Rotala, Lindemia, Lamium, Veronica, Abutilon, Emex, Datura, Viola, Galeopsis, Papaver, Centaurea, Trifolium, Ranunculus, Taraxacum.
Dicotyledonous crops of the genera: Gossypium, Glycine, Beta, Daucus, Phaseolus, Pisum, Solanum, Linum, Ipomoea, Vicia, Nicotiana, Lycopersicon, Arachis, Brassica, Lactuca, Cucumis, Cuburbita, Helianthus.
Monocotyledonous weeds of the genera: Echinochloa, Setaria, Panicum, Digitaria, Phleum, Poa, Festuca, Eleusine, Brachiaria, Lolium, Bromus, Avena, Cyperus, Sorghum, Agropyron, Cynodon, Monochoria, Fimbristylis, Sagittaria, Eleocharis, Scirpus, Paspalum, Ischaemum, Sphenoclea, Dactyloctenium, Agrostis, Alopecurus, Apera.
Monocotyledonous crops of the genera: Oryza, Zea, Triticum, Hordeum, Avena, Secale, Sorghum, Panicum, Saccharum, Ananas, Asparagus, Allium.
However, the use of the active compounds according to the invention is in no way restricted to these genera, but also extends in the same manner to other plants. According to the invention, crop plants are all plants and plant varieties including transgenic plants and plant varieties, where on transgenic plants and plant varieties it is also possible for synergistic effects to occur.
Compositions within the context of the present invention comprise in addition to the herbicide compound of formula (I) according to the invention one or more further component(s) which include, but are not limited to the following: formulation auxiliaries, additives customary in crop protection, further agrochemically active compounds (e.g. fungicides and insecticides). Such compositions may also be applied in the method according to the invention.
In another embodiment the method according to the present invention comprises applying at least one further herbicide (c) wherein (c) is chosen from the list which includes but is not limited to the following:
Acetochlor, acifluorfen, acifluorfen-methyl, acifluorfen-sodium, aclonifen, alachlor, allidochlor, alloxydim, alloxydim-sodium, ametryn, amicarbazone, amidochlor, amidosulfuron, 4-amino-3-chloro-6- (4-chloro-2-fluoro-3-methylphenyl)-5-fluoropyridine-2-carboxylic acid, aminocyclopyrachlor, aminocyclopyrachlor-potassium, aminocyclopyrachlor-methyl, aminopyralid, aminopyralid- dimethylammonium, aminopyralid- tripromine, amitrole, ammoniumsulfamate, anilofos, asulam, asulam-potassium, asulam sodium, atrazine, azafenidin, azimsulfuron, beflubutamid, (S)-(-)- beflubutamid, beflubutamid-M, benazolin, benazolin-ethyl, benazolin-dimethylammonium, benazolin- potassium, benfluralin, benfuresate, bensulfuron, bensulfuron-methyl, bensulide, bentazone, bentazone- sdium, benzobicyclon, benzofenap, bicyclopyrone, bifenox, bilanafos, bilanafos-sodium, bipyrazone, bispyribac, bispyribac-sodium, bixlozone, bromacil, bromacil-lithium, bromacil-sodium, bromobutide, bromofenoxim, bromoxynil, bromoxynil-butyrate, -potassium, -heptanoate und -octanoate, busoxinone, butachlor, butafenacil, butamifos, butenachlor, butralin, butroxydim, butylate, cafenstrole, cambendichlor, carbetamide, carfentrazone, carfentrazone-ethyl, chloramben, chloramben-ammonium, chloramben-diolamine, chlroamben-methyl, chloramben-methylammonium, chloramben-sodium, chlorbromuron, chlorfenac, chlorfenac-ammonium, chlorfenac-sodium, chlorfenprop, chlorfenpropmethyl, chlorflurenol, chlorflurenol-methyl, chloridazon, chlorimuron, chlorimuron-ethyl, chlorophthalim, chlorotoluron, chlorsulfuron, chlorthal, chlorthal-dimethyl, chlorthal-monomethyl, cinidon, cinidon-ethyl, cinmethylin, , exo-(+)-cinmethylin, i.e. (lR,2S,4S)-4-isopropyl-l-methyl-2-[(2- methylbenzyl)oxy]-7-oxabicyclo[2.2.1]heptane, exo-(-)-cinmethylin, i.e. (lR,2S,4S)-4-isopropyl-l- methyl-2-[(2-methylbenzyl)oxy]-7-oxabicyclo[2.2.1]heptane, cinosulfuron, clacyfos, clethodim, clodinafop, clodinafop-ethyl, clodinafop-propargyl, clomazone, clomeprop, clopyralid, clopyralid- methyl, clopyralid-olamine, clopyralid-potassium, clopyralid-tripomine, cloransulam, cloransulam- methyl, cumyluron, cyanamide, cyanazine, cycloate, cyclopyranil, cyclopyrimorate, cyclosulfamuron, cycloxydim, cyhalofop, cyhalofop-butyl, cyprazine, 2,4-D (including theammonium, butotyl, -butyl, choline, diethylammonium, -dimethylammonium, -diolamine, -doboxyl, -dodecylammonium, etexyl, ethyl, 2-ethylhexyl, heptylammonium, isobutyl, isooctyl, isopropyl, isopropylammonium, lithium, meptyl, methyl, potassium, tetradecylammonium, triethylammonium, triisopropanolammonium, tripromine and trolamine salt thereof), 2,4-DB, 2,4-DB-butyl, -dimethylammonium, isooctyl, -potassium und -sodium, daimuron (dymron), dalapon, dalapon-calcium, dalapon-magnesium, dalapon-sodium, dazomet, dazomet-sodium, n-decanol, 7-deoxy-D-sedoheptulose, desmedipham, detosyl-pyrazolate (DTP), dicamba and its salts, e. g. dicamba-biproamine, dicamba-N,N-Bis(3-aminopropyl)methylamine, dicamba-butotyl, dicamba-choline, dicamba-digly colamine, dicamba-dimethylammonium, dicamba- diethanolaminemmonium, dicamba-diethylammonium, dicamba-isopropylammonium, dicamba-methyl, dicamba-monoethanolaminedicamba-olamine, dicamba-potassium, dicamba-sodium, dicamba- triethanolamine, dichlobenil, 2-(2,4-dichlorobenzyl)-4,4-dimethyl- 1 ,2-oxazolidin-3-one, 2-(2,5- dichlorobenzyl)-4,4-dimethyl- 1 ,2-oxazolidin-3-one, dichlorprop, dichlorprop-butotyl, dichlroprop- dimethylammonium, dichhlorprop-etexyl, dichlorprop-ethylammonium, dichlorprop-isoctyl,
dichlorprop-methyl, dichlorprop-postassium, dichlorprop-sodium, dichlorprop-P, dichlorprop-P- dimethylammonium, dichlorprop-P-etexyl, dichlorprop-P-potassium, dichlorprop-sodium, diclofop, diclofop-methyl, diclofop-P, diclofop-P-methyl, diclosulam, difenzoquat, difenzoquat-metilsulfate, diflufenican, diflufenzopyr, diflufenzopyr-sodium, dimefuron, dimepiperate, dimesulfazet, dimethachlor, dimethametryn, dimethenamid, dimethenamid-P, dimetrasulfuron, dinitramine, dinoterb, dinoterb-acetate, diphenamid, diquat, diquat-dibromid, diquat-dichloride, dithiopyr, diuron, DNOC, DNOC-ammonium, DNOC-potassium, DNOC-sodium, endothal, endothal-diammonium, endothal- dipotassium, endothal-disodium, Epyrifenacil (S-3100), EPTC, esprocarb, ethalfluralin, ethametsulfuron, ethametsulfuron-methyl, ethiozin, ethofumesate, ethoxyfen, ethoxyfen-ethyl, ethoxysulfuron, etobenzanid, F-5231, i.e. N-[2-Chlor-4-fluor-5-[4-(3-fluorpropyl)-4,5-dihydro-5-oxo- IH-tetrazol-l-yl] -phenyl] -ethansulfonamid, F-7967, i.e. 3-[7-Chlor-5-fhior-2-(trifhrormethyl)-lH- benzimidazol-4-yl]-l-methyl-6-(trifluormethyl)pyrimidin-2,4(lH,3H)-dion, fenoxaprop, fenoxaprop-P, fenoxaprop-ethyl, fenoxaprop-P-ethyl, fenoxasulfone, fenpyrazone, fenquinotrione, fentrazamide, flamprop, flamprop-isoproyl, flamprop-methyl, flamprop-M-isopropyl, flamprop-M-methyl, flazasulfuron, florasulam, florpyrauxifen, florpyrauxifen-benzyl, fluazifop, fluazifop-butyl, fluazifop- methyl, fluazifop-P, fluazifop-P-butyl, flucarbazone, flucarbazone-sodium, flucetosulfuron, fluchloralin, flufenacet, flufenpyr, flufenpyr-ethyl, flumetsulam, flumiclorac, flumiclorac -pentyl, flumioxazin, fluometuron, flurenol, flurenol-butyl, -dimethylammonium und -methyl, fluoroglycofen, fluoroglycofen- ethyl, flupropanate, flupropanate-sdium, flupyrsulfuron, flupyrsulfuron-methyl, flupyrsulfuron-methyl- sodium, fluridone, flurochloridone, fluroxypyr, fluroxypyr-butometyl, fluroxypyr-meptyl, flurtamone, fluthiacet, fluthiacet-methyl, fomesafen, fomesafen-sodium, foramsulfuron, foramsulfuron sodium salt, fosamine, fosamine-ammonium, glufosinate, glufosinate-ammonium, glufosinate-sodium, L-glufosinate- ammonium, L-glufosiante-sodium, glufosinate-P-sodium, glufosinate-P-ammonium, glyphosate, glyphosate-ammonium, -isopropylammonium, -diammonium, -dimethylammonium, -potassium, - sodium, sesquisodium and -trimesium, H-9201, i.e. O-(2,4-Dimethyl-6-nitrophenyl)-O-ethyl- isopropylphosphoramidothioat, halauxifen, halauxifen-methyl, halosafen, halosulfuron, halosulfuron- methyl, haloxyfop, haloxyfop-P, haloxyfop-ethoxyethyl, haloxyfop-P-ethoxyethyl, haloxyfop-methyl, haloxyfop-P-methyl, haloxifop-sodium, hexazinone, HNPC-A8169, i.e. prop-2-yn-l-yl (2S)-2-{3-[(5- tert-butylpyridin-2-yl)oxy]phenoxy}propanoate, HW-02, i.e. l-(Dimethoxyphosphoryl)-ethyl-(2,4- dichlorphenoxy)acetat, hydantocidin, imazamethabenz, imazamethabenz-methyl, imazamox, imazamox- ammonium, imazapic, imazapic-ammonium, imazapyr, imazapyr-isopropylammonium, imazaquin, imazaquin-ammonium, imazaquin.methyl, imazethapyr, imazethapyr-immonium, imazosulfuron, indanofan, indaziflam, iodosulfuron, iodosulfuron-methyl, iodosulfuron-methyl-sodium, ioxynil, ioxynil-lithium, -octanoate, -potassium und sodium, ipfencarbazone, isoproturon, isouron, isoxaben, isoxaflutole, karbutilate, KUH-043, i.e. 3-({[5-(Difluormethyl)-l-methyl-3-(trifluormethyl)-lH-pyrazol- 4-yl]methyl}sulfonyl)-5,5-dimethyl-4,5-dihydro-l,2-oxazol, ketospiradox, ketospiradox-potassium, lactofen, lenacil, linuron, MCPA, MCPA-butotyl, -butyl, -dimethylammonium, -diolamine, -2-
ethylhexyl, -ethyl, -isobutyl, isoctyl, -isopropyl, -isopropylammonium, -methyl, olamine, -potassium, - sodium and -trolamine, MCPB, MCPB-methyl, -ethyl und -sodium, mecoprop, mecoprop-butotyl, mecoprop- demethylammonium, mecoprop-diolamine, mecoprop-etexyl, mecoprop-ethadyl, mecoprop- isoctyl, mecoprop-methyl, mecoprop-potassium, mecoprop-sodium, and mecoprop-trolamine, mecoprop-P, mecoprop-P-butotyl, -dimethylammonium, -2-ethylhexyl and -potassium, mefenacet, mefluidide, mefluidide-diolamine, mefluidide-potassium, mesosulfuron, mesosulfuron-methyl, mesosulfuron sodium salt, mesotrione, methabenzthiazuron, metam, metamifop, metamitron, metazachlor, metazosulfuron, methabenzthiazuron, methiopyrsulfuron, methiozolin, methyl isothiocyanate, metobromuron, metolachlor, S-metolachlor, metosulam, metoxuron, metribuzin, metsulfuron, metsulfuron-methyl, molinate, monolinuron, monosulfuron, monosulfuron-methyl, MT- 5950, i.e. N-[3-chlor-4-(l-methylethyl)-phenyl]-2-methylpentanamid, NGGC-011, napropamide, NC- 310, i.e. 4-(2,4-Dichlorbenzoyl)-l-methyl-5-benzyloxypyrazol, NC-656, i.e. 3- [(isopropylsulfonyl)methyl]-N-(5-methyl-l,3,4-oxadiazol-2-yl)-5-(trifluoromethyl)[l,2,4]triazolo[4,3- a]pyridine-8-carboxamide, neburon, nicosulfuron, nonanoic acid (pelargonic acid), norflurazon, oleic acid (fatty acids), orbencarb, orthosulfamuron, oryzalin, oxadiargyl, oxadiazon, oxasulfuron, oxaziclomefone, oxyfluorfen, paraquat, paraquat-dichloride, paraquat-dimethylsulfate, pebulate, pendimethalin, penoxsulam, pentachlorphenol, pentoxazone, pethoxamid, petroleum oils, phenmedipham, phenmedipham-ethyl, picloram, picloram-dimethylammonium, picloram-etexyl, picloram-isoctyl, picloram-methyl, picloram-olamine, picloram-potassium, picloram-triethylammonium, picloram- tripromine, picloram- trolamine, picolinafen, pinoxaden, piperophos, pretilachlor, primisulfuron, primisulfuron-methyl, prodiamine, profoxydim, prometon, prometryn, propachlor, propanil, propaquizafop, propazine, propham, propisochlor, propoxycarbazone, propoxycarbazonesodium, propyrisulfuron, propyzamide, prosulfocarb, prosulfuron, pyraclonil, pyraflufen, pyraflufen- ethyl, pyrasulfotole, pyrazolynate (pyrazolate), pyrazosulfuron, pyrazosulfuron-ethyl, pyrazoxyfen, pyribambenz, pyribambenz-isopropyl, pyribambenz-propyl, pyribenzoxim, pyributicarb, pyridafol, pyridate, pyriftalid, pyriminobac, pyriminobac-methyl, pyrimisulfan, pyrithiobac, pyrithiobac-sodium, pyroxasulfone, pyroxsulam, quinclorac, quinclorac-dimethylammonium, quinclorac-methyl, quinmerac, quinoclamine, quizalofop, quizalofop-ethyl, quizalofop-P, quizalofop-P-ethyl, quizalofop-P-tefuryl, QYM201 , i.e. 1 - { 2-chloro-3- [(3-cyclopropyl-5-hydroxy- 1 -methyl- 1 H-pyrazol-4-yl)carbonyl] -6- (trifluoromethyl)phenyl}piperidin-2-one, rimsulfuron, saflufenacil, sethoxydim, siduron, simazine, simetryn, SL-261, sulcotrione, sulfentrazone, sulfometuron, sulfometuron-methyl, sulfosulfuron, , SYP- 249, i.e. 1 -Ethoxy-3-methyl- 1 -oxobut-3-en-2-yl-5-[2-chlor-4-(trifluormethyl)phenoxy] -2-nitrobenzoat, SYP-300, i.e. l-[7-Fluor-3-oxo-4-(prop-2-in-l-yl)-3,4-dihydro-2H-l,4-benzoxazin-6-yl]-3-propyl-2- thioxoimidazolidin-4,5-dion, 2,3,6-TBA, TCA (trichloro acetic acid) and its salts, e.g. TCA-ammonium, TCA-calcium, TCA-ethyl, TCA-magnesium, TCA-sodium, tebuthiuron, tefuryltrione, tembotrione, tepraloxydim, terbacil, terbucarb, terbumeton, terbuthylazine, terbutryn, tetflupyrolimet, thaxtomin, thenylchlor, thiazopyr, thiencarbazone, thiencarbazone-methyl, thifensulfuron, thifensulfuron-methyl,
thiobencarb, tiafenacil, tolpyralate, topramezone, tralkoxydim, triafamone, tri-allate, triasulfuron, triaziflam, tribenuron, tribenuron-methyl, triclopyr, triclopyr-butotyl, triclopyr-choline, triclopyr-ethyl, triclopyr-triethylammonium, trietazine, trifloxysulfuron, trifloxysulfuron-sodium, trifludimoxazin, trifluralin, triflusulfuron, triflusulfuron-methyl, tritosulfuron, urea sulfate, vernolate, XDE-848, ZJ-0862, i.e. 3,4-Dichlor-N-{2-[(4,6-dimethoxypyrimidin-2-yl)oxy]benzyl}anilin, 3-(2-chloro-4-fluoro-5-(3- methyl-2,6-dioxo-4-trifluoromethyl-3,6-dihydropyrimidin-l (2H)-yl)phenyl)-5-methyl-4,5- dihydroisoxazole-5-carboxylic acid ethyl ester, 3-chloro-2-[3-(difluoromethyl)isoxazolyl-5-yl]phenyl-5- chloropyrimidin-2-yl ether, 2-(3 ,4-dimethoxyphenyl)-4- [(2-hydroxy-6-oxocyclohex- 1 -en- 1 - yl)carbonyl]-6-methylpyridazine-3(2H)-one, 2-({2-[(2-methoxyethoxy)methyl]-6-methylpyridin-3- yl } carbonyl)cyclohexane- 1 ,3-dione, (5-hydroxy- 1 -methyl- 1 H-pyrazol-4-yl)(3 ,3 ,4-trimethyl- 1 , 1 -dioxido- 2,3-dihydro- 1 -benzothiophen-5-yl)methanone, 1 -methyl-4-[(3,3,4-trimethyl- 1 , 1 -dioxido-2,3-dihydro- 1 - benzothiophen-5-yl)carbonyl] - 1 H-pyrazol-5-yl propane- 1 -sulfonate, 4- { 2-chloro-3- [(3 ,5-dimethyl- 1 H- pyrazol- 1 -yl)methyl] -4-(methylsulfonyl)benzoyl } - 1 -methyl- 1 H-pyrazol-5 -yl 1,3 -dimethyl- 1 H-pyrazole- 4-carboxylate; cyanomethyl 4-amino-3-chloro-5-fluoro-6-(7-fluoro-lH-indol-6-yl)pyridine-2- carboxylate, prop-2-yn-l-yl 4-amino-3-chloro-5-fluoro-6-(7-fluoro-lH-indol-6-yl)pyridine-2- carboxylate, methyl 4-amino-3-chloro-5-fluoro-6-(7-fluoro-lH-indol-6-yl)pyridine-2-carboxylate, 4- amino-3-chloro-5-fluoro-6-(7-fluoro-lH-indol-6-yl)pyridine-2-carboxylic acid, benzyl 4-amino-3- chloro-5-fluoro-6-(7-fluoro-lH-indol-6-yl)pyridine-2-carboxylate, ethyl 4-amino-3-chloro-5-fluoro-6- (7-fhroro-lH-indol-6-yl)pyridine-2-carboxylate, methyl 4-amino-3-chloro-5-fluoro-6-(7-fluoro-l- isobutyryl- 1 H-indol-6-yl)pyridine-2-carboxylate, methyl 6-( 1 -acetyl-7 -fluoro- 1 H-indol-6-yl)-4-amino- 3-chloro-5-fluoropyridine-2-carboxylate, methyl 4-amino-3-chloro-6-[l-(2,2-dimethylpropanoyl)-7- fluoro- 1 H-indol-6-yl] -5-fluoropyridine-2-carboxylate, methyl 4-amino-3-chloro-5-fluoro-6- [7 -fluoro- 1 - (methoxyacetyl)-lH-indol-6-yl]pyridine-2-carboxylate, potassium 4-amino-3-chloro-5-fluoro-6-(7- fluoro-lH-indol-6-yl)pyridine-2-carboxylate, sodium 4-amino-3-chloro-5-fluoro-6-(7-fluoro-lH-indol- 6-yl)pyridine-2-carboxylate, butyl 4-amino-3-chloro-5-fhroro-6-(7-fluoro-lH-indol-6-yl)pyridine-2- carboxylate, 4-hydroxy-l-methyl-3-[4-(trifluoromethyl)pyridin-2-yl]imidazolidin-2-one, 3-(5-tert-butyl- l,2-oxazol-3-yl)-4-hydroxy-l-methylimidazolidin-2-one, abscisic acid, acibenzolar, acibenzolar-S- methyl, 1-aminocyclopro-l-yl carboxylic acid and derivatives thereof, 5 -Aminolevulinic acid, ancymidol, 6-benzylaminopurine, brassinolide, brassinolide-ethyl, catechin, chitooligosaccharides (CO; COs differ from LCOs in that they lack the pendant fatty acid chain that is characteristic of LCOs. COs, sometimes referred to as N-acetylchitooligosaccharides, are also composed of GlcNAc residues but have side chain decorations that make them different from chitin molecules [(CsHoNOsln, CAS No. 1398-61-4] and chitosan molecules [(CsHnNOrln, CAS No. 9012-76-4]), chitinous compounds, chlormequat chloride, cloprop, cyclanilide, 3-(Cycloprop-l-enyl)propionic acid, daminozide, dazomet, dazomet-sodium, n-decanol, dikegulac, dikegulac-sodium, endothal, endothal-dipotassium, -disodium, and mono(N,N-dimethylalkylammonium), ethephon, flumetralin, flurenol, flurenol-butyl, flurenol- methyl, flurprimidol, forchlorfenuron, gibberellic acid, inabenfide, indol-3-acetic acid (IAA), 4-indol-3-
ylbutyric acid, isoprothiolane, probenazole, jasmonic acid, Jasmonic acid or derivatives thereof (e.g. Jasmonic acid methyl ester), lipo-chitooligosaccharides (LCO, sometimes referred to as symbiotic nodulation (Nod) signals (or Nod factors) or as Myc factors, consist of an oligosaccharide backbone of P-l,4-linked N-acetyl-D-glucosamine (“GlcNAc”) residues with an N-linked fatty acyl chain condensed at the non-reducing end. As understood in the art, LCOs differ in the number of GlcNAc residues in the backbone, in the length and degree of saturation of the fatty acyl chain and in the substitutions of reducing and non-reducing sugar residues), linoleic acid or derivatives thereof, linolenic acid or derivatives thereof, maleic hydrazide, mepiquat chloride, mepiquat pentaborate, 1 -methylcyclopropene, 3 ’-methyl abscisic acid, 2-(l-naphthyl)acetamide, 1 -naphthylacetic acid, 2- naphthyloxyacetic acid, nitrophenolate-mixture, 4-Oxo-4[(2-phenylethyl)amino]butyric acid, paclobutrazol, 4-phenylbutyric acid, N-phenylphthalamic acid, prohexadione, prohexadione-calcium, prohydrojasmon, salicylic acid, , salicylic acid methyl ester, strigolacton, tecnazene, thidiazuron, triacontanol, trinexapac, trinexapac- ethyl, tsitodef, uniconazole, uniconazole-P, 2-fluoro-N-(3-methoxyphenyl)-9H-purin-6-amine.
“Pre-emergent” application refers to a herbicide which is applied to an area of interest (e.g., a field or area of cultivation) before a plant emerges visibly from the soil. “Post-emergent” application refers to a herbicide which is applied to an area after a plant emerges visibly from the soil. The terms “pre- emergent” and “post-emergent” may be defined via the BBCH code.
The BBCH code, commonly known to the person skilled in the art, provides information on the stage of morphological development of a plant. The abbreviation officially represents the Federal Department of Biology, Federal Register of Plant Varieties and Chemical Industry.
Pre-emergent (PE) = prior to seed germination or up to BBCH stage 09
Post-emergent (PO) = application on the aboveground parts of the plant; BBCH stages 10-31.
Early post-emergent = BBCH stages 10-12.
Mid post-emergent = BBCH stages 13-28.
Late post-emergent = BBCH stages 29-31.
The BBCH code may apply to both the emergence of a weed or of a planted crop. Preferably, the BBCH code refers to the emergence of the crop.
Examples:
A. Efficacy of safening winter wheat
1. Seed treatment of winter wheat (TRZAW)
For seed treatment, the safener compound A3 (see table 2 above) according to the invention, as well as the comparative safeners mefenpyr-diethyl (CAS 135590-91-9) and cloquintocet-mexyl (CAS 99607- 70-2) were used.
After the calculation of the right amount of safener compound needed (WP and WG formulations were used), each safener was tank-mixed with water. The water is used to ensure right kernel distribution of the compound on it. Usually the slurry volume (water+safener) is 800 ml/100 kg seeds. The seeds to be treated were put in a mixing chamber of the equipment for seed treatment. Once seeds are placed in the equipment, the slurry volume was introduced in the mixing chamber for a duration of 20 seconds (to complete the seed treatment). Treated seeds were collected then from the mixing chamber and dried in a paper bag.
In all cases, the dose rate of the seed treatment was 50 g safener / 100 kg seeds.
2. Field trials
Seeds of the eight varieties of TRZAW treated with safeners as described under 1. above as well as untreated seeds were planted in strips in the field with 4 rows of TRZAW per each of the treatments in 4 trials around Europe (DE, FR, PL, IT).
The herbicidal compound Methyl (2R*,4R*)-4-[[(5S)-3-(3,5-difluorophenyl)-5-vinyl-4H-isoxazol-5- carbonyl] amino] tetrahydrofuran-2-carboxylate (in the following: “HER”) formulated as WP 20 was sprayed on early post-emergence (12 BBCH crop) and mid post-emergence (13-31 BBCH crop) at an application rate of 2 g ai/ha and at an application volume of between 200-300 1/ha. The application of the herbicide was done in perpendicular to the planted strips containing the different seed treatments.
% crop damage from the herbicide application observed on the different strips (containing seed treatment and not seed treatment) was scored visually in comparison to control strips (that had not received a herbicide application but containing seed treatment and not seed treatment).
Values in the table below are mean values of two replicates.
The effectiveness of the safener treatment is displayed in below tables 3 and 4 as:
[Average crop damage] % = (herbicide damage on strips (with and without safener seed treatment) treated with the herbicide - herbicide damage on strips (with and without safener seed treatment) non treated with the herbicide) *100
The herbicidal activity (%) on Alopecurus myosuroides (ALOMY) and Avena sativa (AVESA), was determined by the overall difference (%) of the biomass and other symptoms (i.e. chlorosis, necrosis, stunting) between the treated plots with the herbicide and the untreated plots.
Unexpectedly, the safener compound A3 provides for a higher safening function of the crop compared to the safeners mefenpyr-diethyl or cloquintocet-mexyl, as shown in the tables above. At the same time, the herbicidal activity of HER is not affected by the safener compound A3.
B. Efficacy of different modes of safener application
Seeds of the winter wheat variety RGT Reform were used for the following experiments.
1) Seeds treated with safener in SC formulation, corresponding to 0.5 g safener per kilogram of seed.
2) Seeds treated with the corresponding amount of SC formulation without safener.
5 grains per pot of treated seeds were laid out on soil in plastic pots with a volume of 3 liters, covered with a soil-sand mixture and grown in a climatic chamber (day temperature: 16°C, night temperature: 12°C, RH: 50% day length: 15 hours).
Once growth stage BBCH10 was reached, the plants were transferred to a climate chamber with different climatic settings (maximum daily temperature: 15 °C , temperature lowering at night to 8 °C, RH during the day: 50-65%, RH at night: 70%, length of day: 12 hours).
The test plants were treated post-emergent in growth stage BBCH11.
The herbicides and safeners used were formulated as follows:
“HER” (Methyl (2R*,4R*)-4-[[(5S)-3-(3,5-difluorophenyl)-5-vinyl-4H-isoxazol-5- carbonyl] amino] tetrahydrofuran-2-carboxylate): EC 10g/l
“compound A3” (see table 2 above) : WP20
These formulations were mixed according to the indicated dose and sprayed as an aqueous suspension with a water application rate corresponding to 200 1/ha with the addition of a wetting agent (Mero, 1.5 1/ha) onto the above-ground parts of the plant.
The application rate of HER was 2 g /ha; the application rate of compound A3 was 50 g/ha.
After application, the plants were further cultivated as described above. 28 days after application, the herbicide damage was scored visually. The damage was evaluated visually on a scale of 0-100% in comparison to control plants without herbicide and safener treatment.
0% = no observable effect compared to the untreated plant
20% = the treated plant population is 20% damaged compared to the untreated control population (e.g. growth height, leaf damage, etc.)
100% = treated plants are completely damaged or dead.
The mean values from 3 replicates (= 3 pots with 5 plants each) are shown in Table 5 below. A clear superiority of the combination of seed treatment and subsequent foliar application is shown compared to the corresponding single treatments with safener.
The crop damage could be unexpectedly reduced by applying the safener as seed treatment when compared to tank- mixing the safener with the herbicides. An even further unexpected reduction of crop damage was achieved by applying the safener twice, namely both in terms of seed treatment and as tank-mix partner to the herbicide.
Claims
1. Method for reducing crop damage, comprising treating seeds of the crop with a safener compound of formula (II) before sowing,
wherein
(R1 )n’ -phenyl is presented by one of the groups Q-l.l to Q-1.55
and (R2 )m -phenyl is selected from one of the groups Q-2.1 to Q-2.55
R3 is hydrogen, and
R4 is hydrogen, methyl, ethyl, n-propyl, i-propyl, n-butyl, n-pentyl, phenyl, benzyl, CH2(4-Cl-Ph), CH2(4-F-Ph), CH2(4-OMe-Ph), 2-methoxyethyl, tetrahydrofuran-2-yl-methyl, tetrahydrofuran-3-yl-methyl, tetrahydropyran-2-yl-methyl, tetrahydropyran-3-yl-methyl, tetrahydropyran-4-yl-methyl, methylpropionat-3-yl, ethylpropionat-3-yl, methylacetat-2-yl, ethylacetat-2-yl, methylpivalat-2-yl, ethylpivalat-3-yl, methyl-2-methylpropanoat-3-yl, methyl-2,2-dimethylpropanoat-3-yl, ethyl-2-methylpropanoat-3-yl, methyl-2-propanoat-2-yl, ethyl-2-propanoat-2-yl, methyl-acetat-2yl, ethyl-acetat-2yl, methyl- 1- methylcyclopropancarboxylat-2yl, ethyl- 1 -methylcyclopropan-carboxylat-2yl, 2- (dimethylamino)ethyl, oxetan-3-yl, (3-methyloxetan-3-yl)methyl, 2,2,2-trifluorethyl, 2,2- difluorethyl, 2-fluorethyl, 2,2,3,3,3-pentafluorpropyl, cyclopropylmethyl, 1-cyclopropyl- ethyl, (l-methyl-cyclopropyl)-methyl, (2,2-dichlorcyclopropyl)-methyl, (2,2-dimethyl- cyclopropyl)-methyl, allyl, propargyl (prop-2-in-l-yl), 2-chlorprop-2-en-l-yl, 3-phenylprop- 2-in-l-yl, 3,3-dichlorprop-2-en-l-yl, 3,3-dichlor-2-fluor-prop-2-en-l-yl, methylprop-2-in-l- yl, 2-methylprop-2-en-l-yl, but-2-en-l-yl, but-3-en-l-yl, but-2-in-l-yl, but-3-in-l-yl, 4-chlor- but-2-in-l-yl, 3-methyl-but-2-en-l-yl, 3-methyl-but-l-en-l-yl, 1- (2e)-l-methylbut-2-en-l-yl, (e)-pent-3-en-2-yl or (z)-pent-3-en-2-yl, cyclobutylmethyl, cyclopentylmethyl, cyclohexylmethyl, heptan-2-yl, iso-butyl, l,3-dioxolan-2-ylmethyl oder l-ethyl-5-methyl- 1 hpyrazol-4-methyl ; followed by applying a herbicidal compound of formula (I) or agrochemically acceptable salt thereof after sowing,
G represents OR4 or NR7R8
R1 and R2 each represent hydrogen;
R3 represents (Ci-Cs)-alkyl, (Cs-Cel-cycloalkyl, (C2-Cs)-alkenyl, (C2-Cs)-alkinyl or (C1-C5)- alkoxy each optionally substitued “m” times by substituents from the group consisting of halogen, cyano, (Ci-Cs)-alkoxy and hydroxy;
R4 represents hydrogen,
or represents (C1-C12)-alkyl, (C3-C7)-cycloalkyl, (C3-C7)-cycloalkyl-(C1-C8)-alkyl, (C2-C8)- alkenyl, (C5-C6)-cycloalkenyl, (C1-C4)-alkylphenyl or (C2-C8)-alkinyl each optionally substitued “m” times by substituents from the group consisting of halogen, cyano, (C1-C6)- alkoxy, (C1-C6)-alkoxycarbonyl, hydroxy, S(O)n R5; R5 represents (C1-C8)-alkyl, (C2-C8)-alkenyl, (C3-C6)-cycloalkyl, benzyl, CON((C1-C3)-alkyl)2 or (C1-C8)-alkyl-C(O)-(C1-C8)-alkyl each optionally substitued “m” times by substituents from the group consisting of halogen and cyano; R6 represents hydrogen, or represents (C1-C8)-alkyl, (C3-C6)-cycloalkyl, (C3-C8)-alkenyl or (C3-C8)-alkinyl each optionally substitued “m” times by substituents from the group consisting of halogen, cyano und (C1-C2)-alkoxy; R7, R8 independently of each other represent hydrogen, (C1-C6)-alkoxycarbonyl-(C1-C6)- alkyl, N((C1-C3)-alkyl)2, S(O)n R5, or R7 and R8 together with the nitrogen atom to which they are attached form a saturated or partially or fully unsaturated five-, six-, or seven-membered ring which may contain apart from the nitrogen ataom “r” carbon atoms, “o” oxygen atoms and is optionally substitued “m” times by substituents from the group consisting of halogen, (C1-C6)-alkyl, halogen-(C1-C6)-alkyl, oxo, CO2R6; Z represents Z-1 to Z-8: 0‹ ‹ ‹
v whereas the arrow represents the bonding to the group CO-G of the formula (I); X2, X4 and X6 independently of one another represent hydrogen or fluorine;
X3 and X5 independently of one another represent hydrogen, chlorine, cyano or fluorine; or represents (C1-C3)- Alkyl, (C1-C3)- Alkoxy each optionally substitued “m” times by substituents from the group consisting of fluorine or chlorine; m represents 0, 1, 2, 3, 4 or 5; n represents 0, 1 or 2; o represents 0, 1 or 2; r represents 3, 4, 5 or 6.
2. Method according to claim 1 , wherein the compound of formula (I) is a compound of formula (la) or an agrochemical acceptable salts thereof,
in which
X3, X5, R3 and G are as described above;
3. Method according to claim 1 or 2, wherein the compound of formula (I) is selected from
Methyl rel-(2R,4R)-4-[[3-(3,5-dichlorophenyl)-5-methoxy-4H-isoxazol-5- carbonyl]amino]tetrahydrofuran-2-carboxylate,
Methyl rel-(2R,4R)-4-[[3-(3,5-dichlorophenyl)-5-vinyl-4H-isoxazol-5- carbonyl]amino]tetrahydrofuran-2-carboxylate,
Methyl (2R* ,4R*)-4- [ [(5 S) -3 -(3 ,5 -difluorophenyl)-5 - vinyl-4H-isoxazol-5 - carbonyl]amino]tetrahydrofuran-2-carboxylate,
Isopropyl rel-(2R,4R)-4-[[3-(3-fluorophenyl)-5-methyl-4H-isoxazol-5- carbonyl]amino]tetrahydrofuran-2-carboxylate,
Methyl (3R)-3-[[(5S)-3-(3,5-difluorophenyl)-5-vinyl-4H-isoxazol-5-carbonyl]amino]-2,3- dihydrofuran-5 -carboxylate, and
Methyl (3R)-3-[[(5R)-3-(3,5-difluorophenyl)-5-methyl-4H-isoxazol-5-carbonyl]amino]-2,3- dihydrofuran-5 -carboxylate.
4. Method according to any one of the preceding claims, wherein the compound of formula (II) is selected from
Methyl- { [1 ,5-bis(4-chloro-2-fluorophenyl)- 1H- 1 ,2,4-triazole-3-yl]oxy } acetate,
{ [ 1 ,5-Bis(4-chloro-2-fluorophenyl)- 1H- 1 ,2,4-triazole-3-yl]oxy } acetic acid,
Methyl- { [5-(4-chloro-2-fluorophenyl)- 1 -(2,4-difluorophenyl)- 1H- 1 ,2,4-triazole-3- yl]oxy} acetate,
{ [5-(4-Chloro-2-fluorophenyl)- 1 -(2,4-difluorophenyl)-lH- 1 ,2,4-triazole-3-yl]oxy } acetic acid,
Methyl- { [ 1 -(4-chloro-2-fluorophenyl)-5-(2,4-difluorophenyl)- 1 H- 1 ,2,4-triazole-3- yl]oxy} acetate,
{[l-(4-Chloro-2-fluorophenyl)-5-(2,4-difluorophenyl)-lH-l,2,4-triazole-3-yl]oxy}acetic acid,
Methyl- { [5-(4-bromo-2-fluorophenyl)- 1 -(2,4-difluorophenyl)- 1H- 1 ,2,4-triazole-3- yl]oxy} acetate,
{ [5-(4-Bromo-2-fluorophenyl)- 1 -(2,4-difluorophenyl)- 1H- 1 ,2,4-triazole-3-yl]oxy } acetic acid,
Methoxyethyl- { [ 1 -(4-bromo-2-fluorophenyl)-5-(2,4-difluorophenyl)- 1H- 1 ,2,4-triazole-3- yl]oxy} acetate, and
{ [ l-(4-Bromo-2-fluorophenyl)-5-(2,4-difluorophenyl)- 1H- 1 ,2,4-triazole-3-yl]oxy } acetic acid.
5. Method according to any one of the preceding claims, wherein the herbicidal compound of formula (I) is Methyl (2R*,4R*)-4-[[(5S)-3-(3,5-difluorophenyl)-5-vinyl-4H-isoxazol-5- carbonyl]amino]tetrahydrofuran-2-carboxylate; and/or wherein the safener compound of formula (II) is selected from
Methyl- { [1 ,5-bis(4-chloro-2-fluorophenyl)- 1H- 1 ,2,4-triazole-3-yl]oxy } acetate,
Methyl- { [5-(4-chloro-2-fluorophenyl)- 1 -(2,4-difluorophenyl)- 1H- 1 ,2,4-triazole-3- yl]oxy} acetate,
Methyl- { [ 1 -(4-chloro-2-fluorophenyl)-5-(2,4-difluorophenyl)- 1 H- 1 ,2,4-triazole-3- yl]oxy} acetate,
Methyl- { [5-(4-bromo-2-fluorophenyl)- 1 -(2,4-difluorophenyl)- 1H- 1 ,2,4-triazole-3- yl]oxy} acetate, and
Methoxyethyl- { [ 1 -(4-bromo-2-fluorophenyl)-5-(2,4-difluorophenyl)- 1H- 1 ,2,4-triazole-3- yl]oxy} acetate.
6. Method according to claim 5, wherein the herbicidal compound of formula (I) is Methyl (2R*,4R*)-4-[[(5S)-3-(3,5-difluorophenyl)-5-vinyl-4H-isoxazol-5- carbonyl]amino]tetrahydrofuran-2-carboxylate, and wherein the safener compound of formula
(II) is Methyl-{ [5-(4-chlor-2-fluorphenyl)-l-(2,4-difluorphenyl)-lH-l,2,4-triazol-3- yl]oxy} acetate.
7. Method according to any one of claims 1 to 6, wherein the herbicidal compound of formula (I) is applied in a pre-emergent treatment.
8. Method according to any one of claims 1 to 6, wherein the herbicidal compound of formula (I) is applied in a post-emergent treatment.
9. Method according to any one of the preceding claims, wherein the crop is a genetically modified plant.
10. Method according to any one of the preceding claims, wherein the safener compound of formula (II) is applied to the seeds in an application rate of between 0.01 and 2 g per kg seed, preferably between 0.1 and 1 g per kg seed.
11. Method according to any one of the preceding claims, wherein the herbicidal compound of formula (I) is applied at an application rate of between 0.1 and 1000 g per ha, preferably between 1 and 50 g per ha.
12. Method according to any one of the preceding claims, further comprising applying the safener compound of formula (II) after sowing in a pre-emergent or post-emergent treatment.
13. Method according to claim 12, wherein the safener compound of formula (II) is applied in a tank-mix together with the herbicidal compound of formula (I).
14. Method according to any of the preceding claims, wherein the crop is selected from wheat, triticale, barley, and rye.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP23201166 | 2023-10-02 | ||
| EP23201166.8 | 2023-10-02 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2025073561A1 true WO2025073561A1 (en) | 2025-04-10 |
Family
ID=88237411
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2024/077001 Pending WO2025073561A1 (en) | 2023-10-02 | 2024-09-26 | Herbicide/safener combination based on seed treatment with safeners belonging to the class of substituted [(1,5-diphenyl-1h-1,2,4-triazol-3-yl)oxy]acetic acid and salts thereof |
Country Status (2)
| Country | Link |
|---|---|
| AR (1) | AR134007A1 (en) |
| WO (1) | WO2025073561A1 (en) |
Citations (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2018228985A1 (en) | 2017-06-13 | 2018-12-20 | Bayer Aktiengesellschaft | Herbicidally active 3-phenylisoxazoline-5-carboxamides of tetrahydro and dihydrofuran carboxylic acids and esters |
| WO2019145245A1 (en) | 2018-01-25 | 2019-08-01 | Bayer Aktiengesellschaft | Herbicidally active 3-phenylisoxazoline-5-carboxamides of cyclopentenyl carboxylic acid derivatives |
| WO2021105101A1 (en) | 2019-11-26 | 2021-06-03 | Bayer Aktiengesellschaft | [(1,5-diphenyl-1h-1,2,4-triazol-3-yl)oxy]acetic acid derivatives and salts thereof, crop protection compositions comprising them, methods for producing them and use thereof as safeners |
| WO2021245004A1 (en) | 2020-06-02 | 2021-12-09 | Bayer Aktiengesellschaft | Selective herbicides based on substituted isoxazolin carboxamides and cloquintocet-mexyl |
| WO2021245003A1 (en) | 2020-06-02 | 2021-12-09 | Bayer Aktiengesellschaft | Selective herbicides based on substituted isoxazolin carboxamides and isoxadifen-ethyl |
| WO2021245005A1 (en) | 2020-06-02 | 2021-12-09 | Bayer Aktiengesellschaft | Selective herbicides based on substituted isoxazolin carboxamides and cyprosulfamide |
| WO2021245009A1 (en) | 2020-06-02 | 2021-12-09 | Bayer Aktiengesellschaft | Selective herbicides based on substituted isoxazolin carboxamides and metcamifen |
| WO2021245006A1 (en) | 2020-06-02 | 2021-12-09 | Bayer Aktiengesellschaft | Selective herbicides based on substituted isoxazolin carboxamides and furilazole |
| WO2021245007A1 (en) | 2020-06-02 | 2021-12-09 | Bayer Aktiengesellschaft | Selective herbicides based on substituted isoxazolin carboxamides and mefenpyr-diethyl |
| WO2021245008A1 (en) | 2020-06-02 | 2021-12-09 | Bayer Aktiengesellschaft | Selective herbicides based on substituted isoxazolin carboxamides and benoxacor |
| WO2022238194A1 (en) | 2021-05-10 | 2022-11-17 | Bayer Aktiengesellschaft | Herbicide/safener combination based on safeners from the class of substituted [(1,5-diphenyl-1h-1,2,4-triazol-3-yl)oxy]acetic acids and their salts |
| WO2023001679A1 (en) * | 2021-07-19 | 2023-01-26 | Bayer Aktiengesellschaft | Method for spot-treatment application against weeds |
-
2024
- 2024-09-26 WO PCT/EP2024/077001 patent/WO2025073561A1/en active Pending
- 2024-10-01 AR ARP240102662A patent/AR134007A1/en unknown
Patent Citations (21)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2018228985A1 (en) | 2017-06-13 | 2018-12-20 | Bayer Aktiengesellschaft | Herbicidally active 3-phenylisoxazoline-5-carboxamides of tetrahydro and dihydrofuran carboxylic acids and esters |
| US11597724B2 (en) * | 2017-06-13 | 2023-03-07 | Bayer Aktiengesellschaft | Herbicidally active 3-phenylisoxazoline-5-carboxamides of tetrahydro and dihydrofuran carboxylic acids and esters |
| WO2019145245A1 (en) | 2018-01-25 | 2019-08-01 | Bayer Aktiengesellschaft | Herbicidally active 3-phenylisoxazoline-5-carboxamides of cyclopentenyl carboxylic acid derivatives |
| US20220306591A1 (en) * | 2018-01-25 | 2022-09-29 | Bayer Aktiengesellschaft | Herbicidally active 3-phenylisoxazoline-5-carboxamides of cyclopentenylcarboxylic acid derivatives |
| WO2021105101A1 (en) | 2019-11-26 | 2021-06-03 | Bayer Aktiengesellschaft | [(1,5-diphenyl-1h-1,2,4-triazol-3-yl)oxy]acetic acid derivatives and salts thereof, crop protection compositions comprising them, methods for producing them and use thereof as safeners |
| WO2021245005A1 (en) | 2020-06-02 | 2021-12-09 | Bayer Aktiengesellschaft | Selective herbicides based on substituted isoxazolin carboxamides and cyprosulfamide |
| US20230200390A1 (en) * | 2020-06-02 | 2023-06-29 | Bayer Aktiengesellschaft | Selective herbicides based on substituted isoxazolin carboxamides and mefenpyr-diethyl |
| WO2021245006A1 (en) | 2020-06-02 | 2021-12-09 | Bayer Aktiengesellschaft | Selective herbicides based on substituted isoxazolin carboxamides and furilazole |
| WO2021245007A1 (en) | 2020-06-02 | 2021-12-09 | Bayer Aktiengesellschaft | Selective herbicides based on substituted isoxazolin carboxamides and mefenpyr-diethyl |
| WO2021245008A1 (en) | 2020-06-02 | 2021-12-09 | Bayer Aktiengesellschaft | Selective herbicides based on substituted isoxazolin carboxamides and benoxacor |
| WO2021245003A1 (en) | 2020-06-02 | 2021-12-09 | Bayer Aktiengesellschaft | Selective herbicides based on substituted isoxazolin carboxamides and isoxadifen-ethyl |
| US20230276792A1 (en) * | 2020-06-02 | 2023-09-07 | Bayer Aktiengesellschaft | Selective herbicides based on substituted isoxazolin carboxamides and cloquintocet-mexyl |
| US20230240297A1 (en) * | 2020-06-02 | 2023-08-03 | Bayer Aktiengesellschaft | Selective herbicides based on substituted isoxazolin carboxamides and cyprosulfamide |
| WO2021245004A1 (en) | 2020-06-02 | 2021-12-09 | Bayer Aktiengesellschaft | Selective herbicides based on substituted isoxazolin carboxamides and cloquintocet-mexyl |
| US20230189808A1 (en) * | 2020-06-02 | 2023-06-22 | Bayer Aktiengesellschaft | Selective herbicides based on substituted isoxazolin carboxamides and furilazole |
| WO2021245009A1 (en) | 2020-06-02 | 2021-12-09 | Bayer Aktiengesellschaft | Selective herbicides based on substituted isoxazolin carboxamides and metcamifen |
| US20230200393A1 (en) * | 2020-06-02 | 2023-06-29 | Bayer Aktiengesellschaft | Selective herbicides based on substituted isoxazolin carboxamides and metcamifen |
| US20230200394A1 (en) * | 2020-06-02 | 2023-06-29 | Bayer Aktiengesellschaft | Selective herbicides based on substituted isoxazolin carboxamides and benoxacor |
| US20230217926A1 (en) * | 2020-06-02 | 2023-07-13 | Bayer Aktiengesellschaft | Selective herbicides based on substituted isoxazolin carboxamides and isoxadifen-ethyl |
| WO2022238194A1 (en) | 2021-05-10 | 2022-11-17 | Bayer Aktiengesellschaft | Herbicide/safener combination based on safeners from the class of substituted [(1,5-diphenyl-1h-1,2,4-triazol-3-yl)oxy]acetic acids and their salts |
| WO2023001679A1 (en) * | 2021-07-19 | 2023-01-26 | Bayer Aktiengesellschaft | Method for spot-treatment application against weeds |
Non-Patent Citations (1)
| Title |
|---|
| no. 9012-76-4 |
Also Published As
| Publication number | Publication date |
|---|---|
| AR134007A1 (en) | 2025-11-26 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US20230276792A1 (en) | Selective herbicides based on substituted isoxazolin carboxamides and cloquintocet-mexyl | |
| AU2021285006A1 (en) | Selective herbicides based on substituted isoxazolin carboxamides and mefenpyr-diethyl | |
| AU2021286151A1 (en) | Selective herbicides based on substituted isoxazolin carboxamides and isoxadifen-ethyl | |
| WO2021245009A1 (en) | Selective herbicides based on substituted isoxazolin carboxamides and metcamifen | |
| US20230189808A1 (en) | Selective herbicides based on substituted isoxazolin carboxamides and furilazole | |
| AU2021285005A1 (en) | Selective herbicides based on substituted isoxazolin carboxamides and cyprosulfamide | |
| US20230200394A1 (en) | Selective herbicides based on substituted isoxazolin carboxamides and benoxacor | |
| WO2025073561A1 (en) | Herbicide/safener combination based on seed treatment with safeners belonging to the class of substituted [(1,5-diphenyl-1h-1,2,4-triazol-3-yl)oxy]acetic acid and salts thereof | |
| EA048095B1 (en) | SELECTIVE HERBICIDES BASED ON SUBSTITUTED ISOXAZOLINE CARBOXAMIDES AND CYPROSULFAMIDE |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 24776305 Country of ref document: EP Kind code of ref document: A1 |