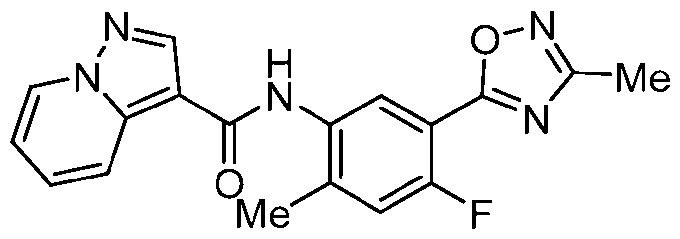
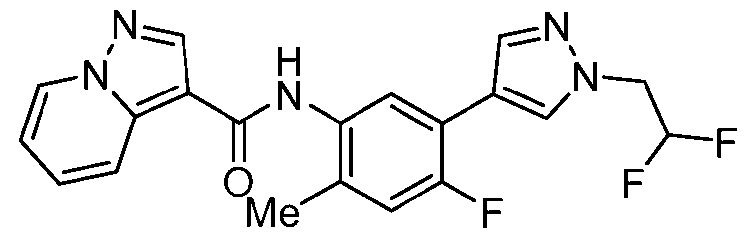
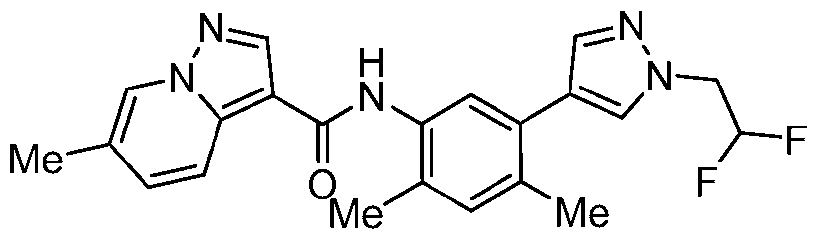
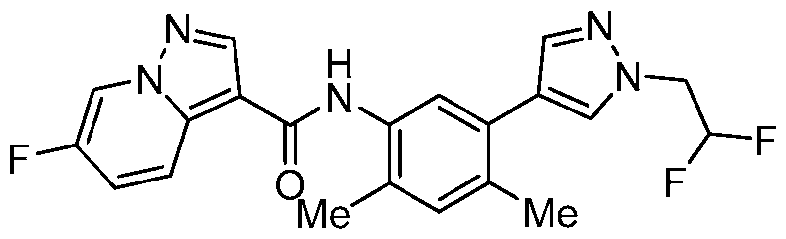
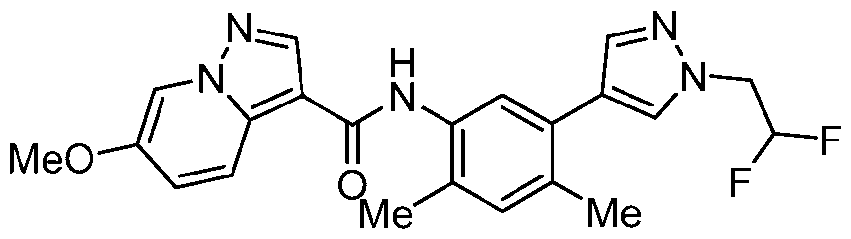
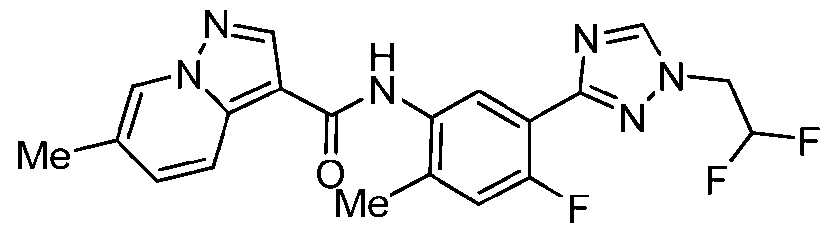
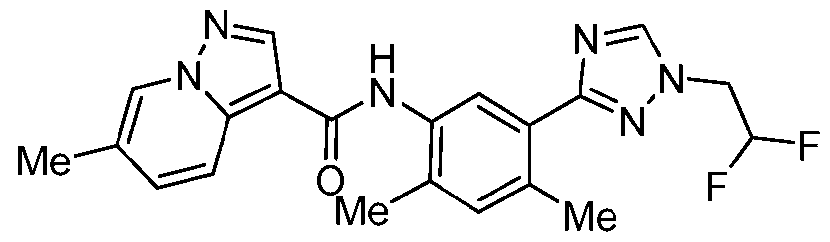
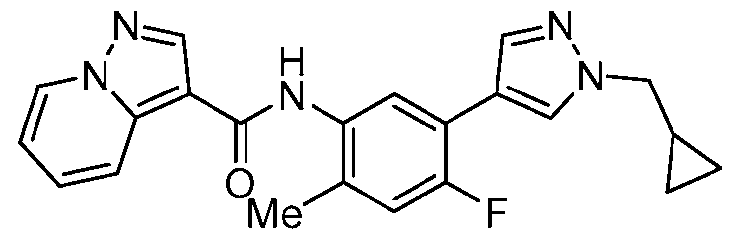
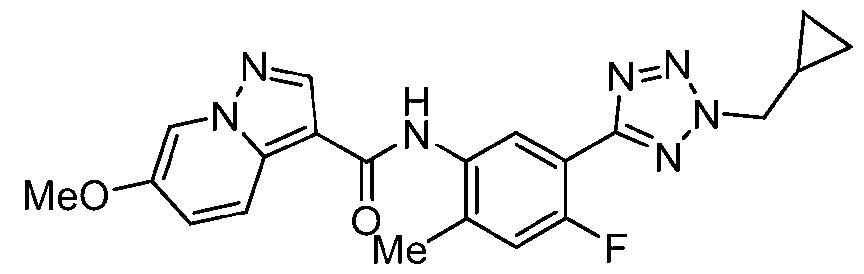
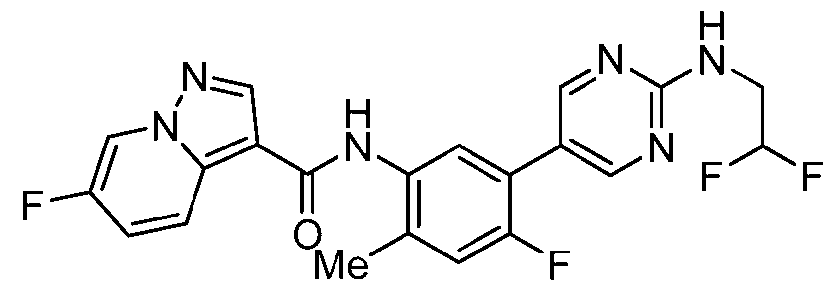
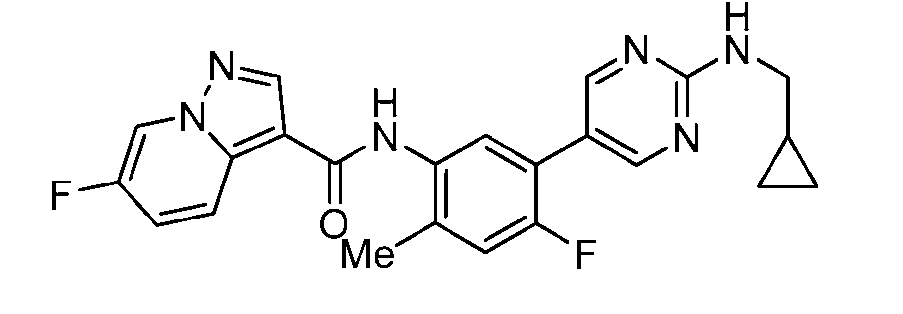
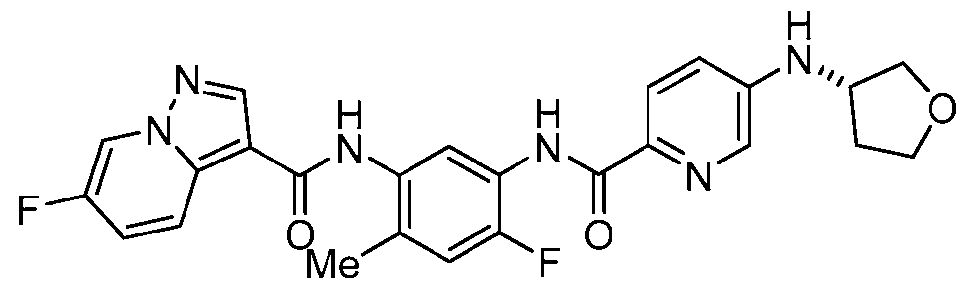
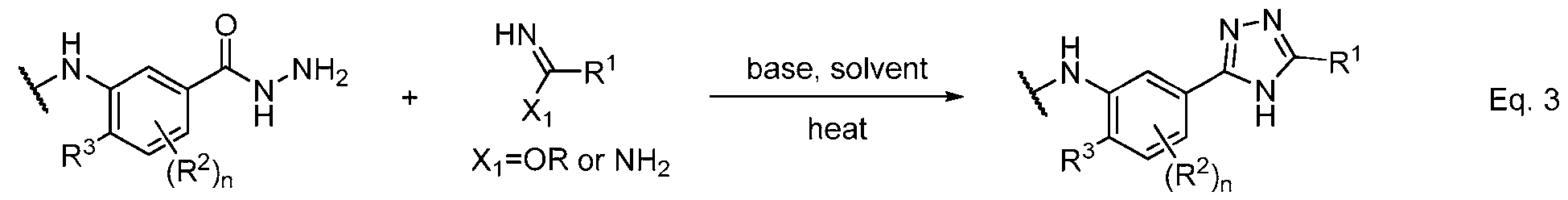WO2025072330A1 - Kit inhibitor compounds and methods of use thereof - Google Patents
Kit inhibitor compounds and methods of use thereof Download PDFInfo
- Publication number
- WO2025072330A1 WO2025072330A1 PCT/US2024/048409 US2024048409W WO2025072330A1 WO 2025072330 A1 WO2025072330 A1 WO 2025072330A1 US 2024048409 W US2024048409 W US 2024048409W WO 2025072330 A1 WO2025072330 A1 WO 2025072330A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- cycloalkyl
- pyridine
- disease
- alkyl
- cancer
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D471/00—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00
- C07D471/02—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00 in which the condensed system contains two hetero rings
- C07D471/04—Ortho-condensed systems
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
Definitions
- mast cells Upon activation, degranulation occurs, releasing these proinflammatory and immunomodulatory mediators into the surrounding tissues, generally in response to a perceived pathogen (e.g., parasitic, bacterial and viral infections, allergens, toxins, etc.).
- pathogen e.g., parasitic, bacterial and viral infections, allergens, toxins, etc.
- the activation of mast cells serves to induce an immune response to protect the body from pathogens, and to aid in wound healing, and tissue repair.
- misfunctioning mast cells underlie the etiology of many allergic and chronic inflammatory diseases and are implicated in a broad spectrum of conditions.
- SCF Stem Cell Factor
- the present disclosure relates to compounds that are inhibitors of the receptor tyrosine kinase KIT.
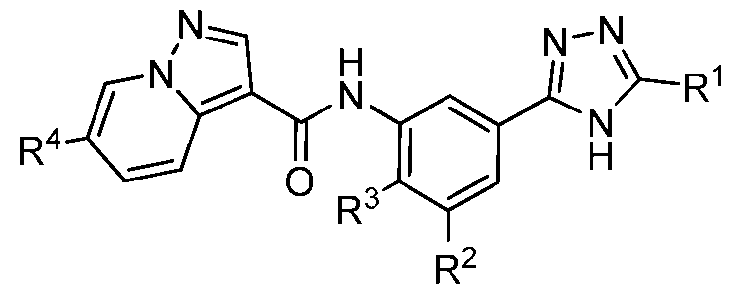
- the compounds are represented by Formula III and associated sub- formulas described herein: .
- this disclosure is directed to methods of inhibiting KIT in a subject comprising administering to the subject an effective amount of a compound of Formula III or an associated sub-formula described herein.
- this disclosure is directed to methods of reducing the activity and/or quantity of systemic mast cells in a subject comprising administering to the subject an effective amount of a compound of Formula III or an associated sub-formula described herein.
- this disclosure provides methods for treating a disease, disorder, or condition mediated at least in part by KIT in a subject, comprising administering to the subject a therapeutically effective amount of a compound of Formula III or an associated sub-formula described herein.
- KIT Diseases, disorders, and conditions mediated by KIT include e.g., an allergic disease, disorder, or condition; an inflammatory disease, disorder, or condition; a neuroinflammatory disease, disorder, or condition; a neurological disease, disorder, or condition; an immune related disease, disorder, or condition; an autoimmune related disease, disorder, or condition; a dermatological disease, disorder, or condition; a respiratory disease, disorder, or condition; a metabolic disease, disorder, or condition; a cardiovascular disease, disorder, or condition; a fibrotic disease, disorder, or condition; or cancer.
- Certain aspects of the present disclosure further comprise the administration of one or more additional therapeutic agents as set forth herein below.
- the near or approximating unrecited number can be a number which, in the context in which it is presented, provides the substantial equivalent of the specifically recited number.
- “about” means either within plus or minus 10% of the provided value, or rounded to the nearest significant figure, in all cases inclusive of the provided value. Where ranges are provided, they are inclusive of the boundary values.
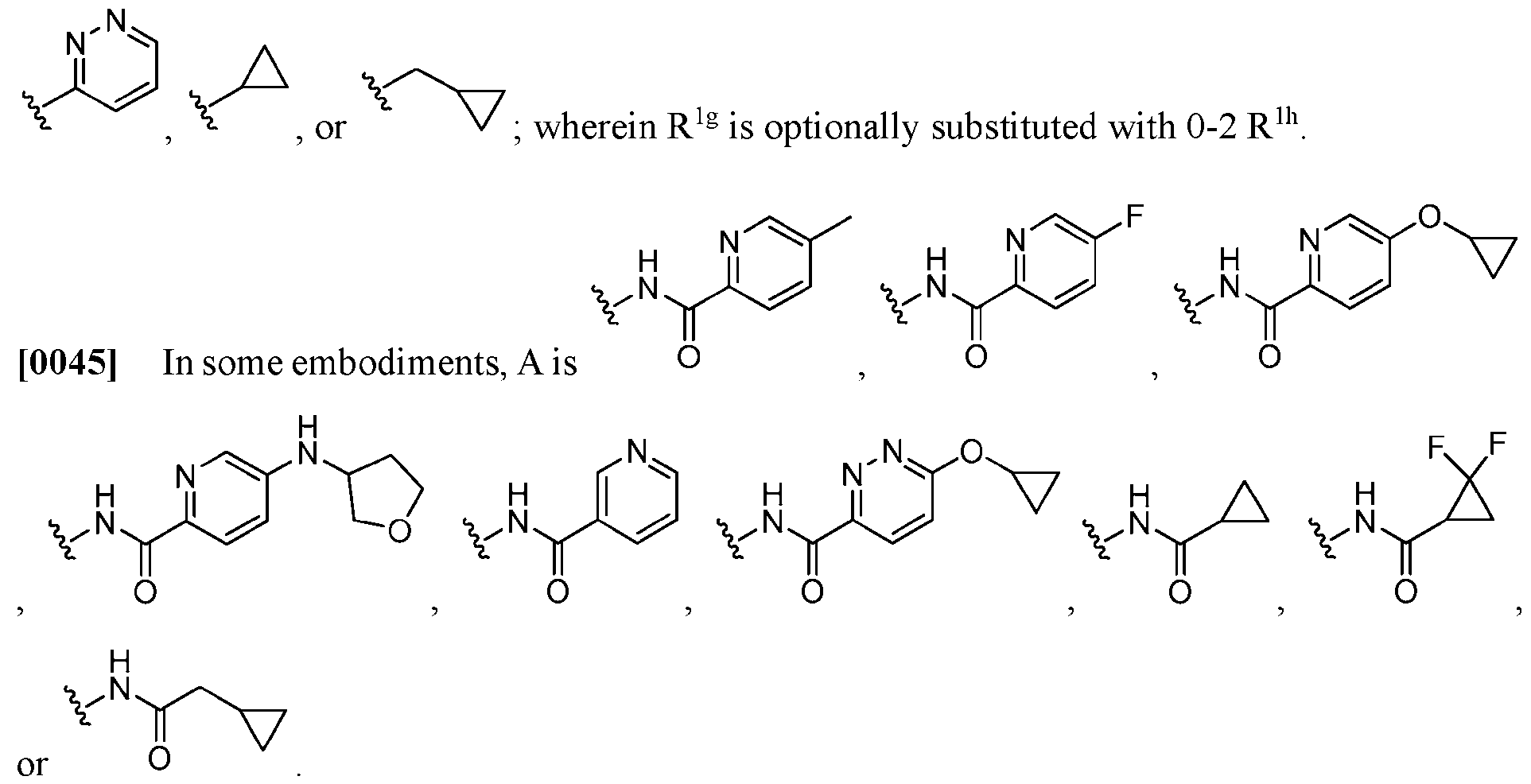
- alkyl by itself or as part of another substituent, means, unless otherwise stated, a saturated hydrocarbon radical, having, in some embodiments, one to eight (e.g., C1-C8- alkyl), or one to six (e.g., C 1 -C 6 -alkyl), or one to four carbon atoms (e.g., C 1 -C 4 -alkyl), or one to three carbon atoms (e.g., C 1 -C 3 -alkyl), respectively.
- alkyl encompasses straight and branched-chain hydrocarbon groups.
- alkyl groups include, but are not limited to, methyl (Me, -CH 3 ), ethyl (Et, -CH 2 CH 3 ), n-propyl, isopropyl, n-butyl, t-butyl, isobutyl, sec-butyl, isopentyl, tert-pentyl, n-pentyl, isohexyl, n-hexyl, n-heptyl, 4-isopropylheptane, n-octyl, and the like.
- the alkyl groups are C1-C4 alkyl groups (e.g., methyl, ethyl, n-propyl, isopropyl, n-butyl, isobutyl, sec-butyl, or t-butyl).
- alkylene refers to a straight or branched, saturated, hydrocarbon radical having, in some embodiments, one to six carbon atoms (e.g., C1-C6-alkylene), or one to four carbon atoms (e.g., C1-C4-alkylene), or one to three carbon atoms (e.g., C1-C3-alkylene) and linking at least two other groups, i.e., a divalent hydrocarbon radical.
- two moieties are linked to the alkylene they can be linked to the same carbon atom (i.e., geminal), or different carbon atoms of the alkylene group.
- a straight chain alkylene can be the bivalent radical of -(CH2)n- , where n is 1, 2, 3, 4, 5 or 6 (i.e., a C 1 -C 6 -alkylene).
- Representative alkylene groups include, but are not limited to, methylene, ethylene, propylene, isopropylene, butylene, isobutylene, secbutylene, pentylene, hexylene and the like.
- the alkylene groups are C1- 3 alkylene groups (e.g., methylene, ethylene, or propylene).
- cycloalkyl refers to a monocyclic, bicyclic or polycyclic hydrocarbon ring system having, in some embodiments, 3 to 14 carbon atoms (e.g., C3-C14-cycloalkyl), or 3 to 10 carbon atoms (e.g., C3-C10-cycloalkyl), or 3 to 8 carbon atoms (e.g., C3-C8-cycloalkyl), or 3 to 6 carbon atoms (e.g., C 3 -C 6 -cycloalkyl) or 5 to 6 carbon atoms (e.g., C 5 -C 6 -cycloalkyl).
- 3 to 14 carbon atoms e.g., C3-C14-cycloalkyl
- 10 carbon atoms e.g., C3-C10-cycloalkyl
- 3 to 8 carbon atoms e.g., C3-C8-cycloalkyl
- 3 to 6 carbon atoms e
- Cycloalkyl groups can be saturated or characterized by one or more points of unsaturation (i.e., carbon-carbon double and/or triple bonds), provided that the points of unsaturation do not result in an aromatic system.
- monocyclic cycloalkyl groups include, but are not limited to, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, cyclohexenyl, cyclohexynyl, cycloheptyl, cycloheptenyl, cycloheptadienyl, cyclooctyl, cyclooctenyl, cyclooctadienyl and the like.
- the rings of bicyclic and polycyclic cycloalkyl groups can be fused, bridged, or spirocyclic.
- Non-limiting examples of bicyclic, spirocyclic and polycyclic hydrocarbon groups include bicyclo[1.1.1]pentane, bicyclo[2.1.1]hexane, bicyclo[2.2.1]heptane, bicyclo[2.2.2]octane, adamantyl, spiro[5.5]undecane, spiro[2.2]pentane, spiro[2.2]pentadiene, spiro[2.5]octane, spiro[2.2]pentadiene, and the like.
- the cycloalkyl groups of the present disclosure are monocyclic C 3 -C 6 -cycloalkyl moieties (e.g., cyclopropyl, cyclobutyl, cyclopentyl, or cyclohexyl).
- a wavy line, " ", that intersects a single, double or triple bond in any chemical structure depicted herein, represents that the point of attachment of the single, double, or triple bond to the remainder of the molecule is through either one of the atoms that make up the single, double or triple bond.
- a bond extending from a substituent to the center of a ring is meant to indicate attachment of that substituent to the ring at any of the available ring vertices, i.e., such that attachment of the substituent to the ring results in a chemically stable arrangement.
- halogen or “halo” are used interchangeably and refer to, unless otherwise stated, a fluorine, chlorine, bromine, or iodine atom.
- haloalkyl refers to alkyl groups, as defined herein, that are substituted with one or more halogen(s) (e.g., 1-3 halogen(s)).
- C 1 -C 6 haloalkyl is meant to include trifluoromethyl, difluoromethyl, 2,2,2-trifluoroethyl, 4-chlorobutyl, 3-bromopropyl, and the like.
- hydroxyalkyl refers to an alkyl group, as defined herein, that is substituted with one or more hydroxyl groups (e.g., 1-3 hydroxyl groups).
- Exemplary hydroxyalkyl groups include methanol, ethanol, 1,2-propanediol, 1,2-hexanediol, glycerol, and the like.
- cyanoalkyl refers to an alkyl group, as defined herein, that is substituted with one cyano group.
- Exemplary cyanoalkyl groups include cyanomethyl, cyanoethyl, cyanopropyl, and the like.
- alkoxy refers to an alkyl group, as defined herein, that is attached to the remainder of the molecule via an oxygen atom (e.g., -O-C1-C12 alkyl, -O-C1-C8 alkyl, -O-C 1 -C 6 alkyl, or -O-C 1 -C 3 alkyl).
- oxygen atom e.g., -O-C1-C12 alkyl, -O-C1-C8 alkyl, -O-C 1 -C 6 alkyl, or -O-C 1 -C 3 alkyl.
- alkoxy groups include methoxy (OMe, -OCH 3 ), ethoxy (OEt, -OCH 2 CH 3 ), n-propoxy, iso-propoxy, n-butoxy, sec- butoxy, n-pentoxy, n-hexoxy, and the like.
- heteroaryl refers to monocyclic or fused bicyclic aromatic groups (or rings) having, in some embodiments, from 5 to 14 (i.e., 5- to 14-membered heteroaryl), or from 5 to 10 (i.e., 5- to 10-membered heteroaryl), or from 5 to 6 (i.e., 5- to 6-membered heteroaryl) members (i.e., ring vertices), and containing from one to five, one to four, one to three, one to two or one ring heteroatom independently selected from nitrogen (N), oxygen (O), and sulfur (S).
- N nitrogen
- O oxygen
- S sulfur
- a heteroaryl group can be attached to the remainder of the molecule through a carbon atom or a heteroatom of the heteroaryl group, when chemically permissible.
- heteroaryl groups include pyridyl, pyridazinyl, pyrazinyl, pyrimidinyl, triazinyl, purinyl, thienopyridinyl, thienopyrimidinyl, pyrazolopyrimidinyl, imidazopyridines, isothiazolyl, pyrazolyl, indazolyl, pteridinyl, imidazolyl, triazolyl, tetrazolyl, oxazolyl, isoxazolyl, thiadiazolyl, pyrrolyl, thiazolyl, furyl, thienyl and the like.
- the heteroaryl groups of the present disclosure are monocyclic 5- to 6-membered heteroaryl moieties having 1-4 ring nitrogen atoms (e.g., pyridinyl, pyrimidinyl, pyrazinyl, pyridazinyl, triazolyl, imidazolyl, pyrazolyl, tetrazolyl, and the like).
- the heteroaryl groups of the present disclosure are 5-membered heteroaryl moieties having 1-4 ring heteroatoms independently selected from N, O, and S (e.g., imidazolyl, pyrazolyl, thiazolyl, oxazolyl, oxadiazole, thiadiazolyl, triazolyl, tetrazolyl, and the like).
- heterocycloalkyl refers to a non-aromatic monocyclic, bicyclic or polycyclic cycloalkyl ring having, in some embodiments, 3 to 14 members (e.g., 3- to 14-membered heterocycle), or 3 to 10 members (e.g., 3- to 10-membered heterocycle), or 4 to 8 members (e.g., 4- to 8-membered heterocycle), or 4 to 6 members (e.g., 4- to 6-membered heterocycle), or 5 to 6 members (e.g., 5- to 6-membered heterocycle), and having from one to five, one to four, one to three, one to two or one ring heteroatom independently selected from nitrogen (N), oxygen (O), and sulfur (S).
- N nitrogen
- O oxygen
- S sulfur
- Heterocycloalkyl groups are saturated or characterized by one or more points of unsaturation (e.g., one or more carbon-carbon double bonds, carbon-carbon triple bonds, carbon- nitrogen double bonds, and/or nitrogen-nitrogen double bonds), provided that the points of unsaturation do not result in an aromatic system.
- the rings of bicyclic and polycyclic heterocycloalkyl groups can be fused, bridged, or spirocyclic.
- heterocycloalkyl groups include aziridine, oxirane, thiirane, azetidine, oxetane, pyrrolidine, imidazolidine, pyrazolidine, dioxolane, phthalimide, piperidine, 1,4-dioxane, morpholine, thiomorpholine, thiomorpholine-S-oxide, thiomorpholine-S,S-oxide, piperazine, 3,4,5,6- tetrahydropyridazine, pyran, decahydroisoquinoline, 3-pyrroline, thiopyran, tetrahydrofuran, tetrahydrothiophene, quinuclidine, 2,6-diazaspiro[3.3]heptane, 2-azaspiro[3.3]heptane, 1- oxaspiro[3.3]heptane, 6-azaspiro[3.4]octane,
- a heterocycloalkyl group can be attached to the remainder of the molecule through a ring carbon atom, or a ring heteroatom, when chemically permissible.
- the heterocycloalkyl groups of the present disclosure are monocyclic 4- to 8-membered heterocycloalkyl moieties having one or two heteroatoms independently selected from N, O, and S, (e.g., azetidine, oxetane, pyrrolidine, tetrahydrofuran, tetrahydropyran, piperidine, piperazine, morpholine, and the like).
- salts of the compounds according to this disclosure are prepared with suitably nontoxic acids or bases, depending on the particular substituents found on the compounds described herein.
- base addition salts can be obtained by contacting the neutral form of such compounds with a sufficient amount of the desired base, either neat or in a suitable inert solvent.
- salts derived from pharmaceutically-acceptable inorganic bases include aluminum, ammonium, calcium, copper, ferric, ferrous, lithium, magnesium, manganic, manganous, potassium, sodium, zinc and the like.
- Salts derived from pharmaceutically-acceptable organic bases include salts of primary, secondary and tertiary amines, including substituted amines, cyclic amines, naturally-occurring amines and the like, such as arginine, betaine, caffeine, choline, N,N’-dibenzylethylenediamine, diethylamine, 2-diethylaminoethanol, 2-dimethylaminoethanol, ethanolamine, ethylenediamine, N- ethylmorpholine, N-ethylpiperidine, glucamine, glucosamine, histidine, hydrabamine, isopropylamine, lysine, methylglucamine, morpholine, piperazine, piperidine, polyamine resins, procaine, purines, theobromine, triethylamine, trimethylamine, tripropylamine, tromethamine and the like.
- acid addition salts can be obtained by contacting the neutral form of such compounds with a sufficient amount of the desired acid, either neat or in a suitable inert solvent.
- suitable inert solvent examples include those derived from inorganic acids like hydrochloric, hydrobromic, nitric, carbonic, monohydrogencarbonic, phosphoric, monohydrogenphosphoric, dihydrogenphosphoric, sulfuric, monohydrogensulfuric, hydriodic, or phosphorous acids and the like, as well as the salts derived from suitably nontoxic organic acids like acetic, propionic, isobutyric, malonic, benzoic, succinic, suberic, fumaric, mandelic, phthalic, benzenesulfonic, p- tolylsulfonic, citric, tartaric, methanesulfonic, and the like.
- salts of amino acids such as arginate and the like, and salts of organic acids like glucuronic or galacturonic acids and the like (see, for example, Berge, S.M., et al, “Pharmaceutical Salts”, Journal of Pharmaceutical Science, 1977, 66, 1-19).
- Certain specific compounds of the present disclosure contain both basic and acidic functionalities that allow the compounds to be converted into either base or acid addition salts.
- the neutral forms of the compounds may be regenerated by contacting the salt with a base or acid and isolating the parent compound in the conventional manner.
- the parent form of the compound differs from the various salt forms in certain physical properties, such as solubility in polar solvents, but otherwise the salts are equivalent to the parent form of the compound for the purposes of the present disclosure.
- This disclosure also contemplates isomers of the compounds described herein (e.g., stereoisomers and/or atropisomers).
- certain compounds of the present disclosure possess asymmetric carbon atoms (chiral centers), or hindered rotation about a single bond; the racemates, diastereomers, and enantiomers, and atropisomers (e.g., Ra, Sa, P and M isomers) of which are all intended to be encompassed within the scope of the present disclosure.
- Stereoisomeric forms may be defined, in terms of absolute stereochemistry, as (R) or (S), depicted uses dashes and/or wedges, and/or in terms of the direction the stereoisomer rotates plane- polarized light (e.g., dextrorotary ((+) or (d)), or levorotary ((-) or (l))).
- stereochemical depiction e.g., using dashes, , and/or wedges,
- a stereochemical assignment e.g., using (R) and (S) notation, or (d) and (l) notation
- isomer(s) e.g., enantiomers and diastereomers, when present.
- “Substantially free of” other isomer(s) indicates at least an 70/30 ratio of the indicated isomer to the other isomer(s), more preferably 80/20, 90/10, or 95/5 or more.
- the indicated isomer will be present in an amount of at least 99%.
- a chemical bond to an asymmetric carbon that is depicted as a solid line ( ) indicates that all possible stereoisomers (e.g., enantiomers, diastereomers, racemic mixtures, etc.) at that carbon atom are included.
- the compound may be present as a racemic mixture, scalemic mixture, or a mixture of diastereomers.
- the compounds of the present disclosure may also contain unnatural proportions of atomic isotopes at one or more of the atoms that constitute such compounds. Unnatural proportions of an isotope may be defined as ranging from the amount found in nature to an amount consisting of 100% of the atom in question.
- the compounds may incorporate radioactive isotopes, such as for example tritium ( 3 H), iodine-125 ( 125 I) or carbon-14 ( 14 C), or non-radioactive isotopes, such as deuterium ( 2 H) or carbon-13 ( 13 C).
- radioactive isotopes such as for example tritium ( 3 H), iodine-125 ( 125 I) or carbon-14 ( 14 C), or non-radioactive isotopes, such as deuterium ( 2 H) or carbon-13 ( 13 C).
- isotopic variants of the compounds of the disclosure may find additional utility, including but not limited to, as diagnostic and/or imaging reagents, or as cytotoxic/radiotoxic therapeutic agents.
- isotopic variants of the compounds of the disclosure can have altered pharmacokinetic and pharmacodynamic characteristics which can contribute to enhanced safety, tolerability or efficacy during treatment.
- the compounds according to this disclosure are characterized by one or more deuterium atoms.
- patient or “subject” are used interchangeably to refer to a human or a non- human animal (e.g., a mammal).
- non- human animal e.g., a mammal.
- treat refers to a course of action that eliminates, reduces, suppresses, mitigates, ameliorates, or prevents the worsening of, either temporarily or permanently, a disease, disorder or condition to which the term applies, or at least one of the symptoms associated therewith.
- Treatment includes alleviation of symptoms, diminishment of extent of disease, inhibiting (e.g., arresting the development or further development of the disease, disorder or condition or clinical symptoms association therewith) an active disease, delaying or slowing of disease progression, improving the quality of life, and/or prolonging survival of a subject as compared to expected survival if not receiving treatment or as compared to a published standard of care therapy for a particular disease.
- the term “in need of treatment” as used herein refers to a judgment made by a physician or similar professional that a subject requires or will benefit from treatment. This judgment is made based on a variety of factors that are in the realm of the physician’s expertise, which may include a positive diagnosis of a disease, disorder or condition.
- the terms “prevent”, “preventing”, “prevention”, “prophylaxis” and the like refer to a course of action initiated in a manner (e.g., prior to the onset of a disease, disorder, condition or symptom thereof) so as to prevent, suppress, inhibit or reduce, either temporarily or permanently, a subject’s risk of developing a disease, disorder, condition or the like (as determined by, for example, the absence of clinical symptoms) or delaying the onset thereof, generally in the context of a subject predisposed to having a particular disease, disorder or condition. In certain instances, the terms also refer to slowing the progression of the disease, disorder or condition or inhibiting progression thereof to a harmful or otherwise undesired state.
- Prevention also refers to a course of action initiated in a subject after the subject has been treated for a disease, disorder, condition or a symptom associated therewith in order to prevent relapse of that disease, disorder, condition or symptom.
- the preventative course of action is taken based on anticipation of a condition or event.
- prevention refers to the prevention, suppression, inhibition or reduction of an allergic, immune, or autoimmune response in a subject suffering from an allergic, inflammatory, neuroinflammatory, neurological, immune, autoimmune, dermatological, respiratory, metabolic, cardiovascular or fibrotic disease, disorder, or condition.
- response may refer to a symptom initiated by an irritant or trigger (e.g., an antigen originating from within the body, or from the external environment) in a subject.
- a reaction e.g., hives, rash, welts, itchy skin, stinging skin, skin fissures, skin lesions, skin blisters, swelling (e.g., in joints, glands, or tissues), vertigo, fatigue, dizziness, fainting, lightheadedness, muscle weakness, headache, dry skin, dry eyes, hair loss, numbness or tingling in extremities, joint pain and/or stiffness, sneezing, runny nose, stuffy nose, chest tightness and/or pain, shortness of breath, wheezing, itchy eyes, watery eyes, blurred vision, sensitivity to light, stomach cramping, abdominal pain, bloating, diarrhea, constipation, indigestion,
- a reaction e.g., hives, rash, welts, itchy skin,
- Subjects susceptible to a trigger are generally those suffering from an allergic, inflammatory, neuroinflammatory, neurological, immune, autoimmune, dermatological, respiratory, metabolic, cardiovascular or fibrotic disease, disorder, or condition, such as those described elsewhere herein.
- the term “in need of prevention” as used herein refers to a judgment made by a physician or other caregiver that a subject requires or will benefit from preventative care. This judgment is made based on a variety of factors that are in the realm of a physician’s or caregiver’s expertise.
- “Substantially pure” indicates that a component (e.g., a compound according to this disclosure) makes up greater than about 50% of the total content of the composition, and typically greater than about 60% of the total content.
- substantially pure refers to compositions in which at least 75%, at least 85%, at least 90% or more of the total composition is the component of interest. In some cases, the component of interest will make up greater than about 90%, or greater than about 95% of the total content of the composition.
- inhibitor of KIT and “KIT inhibitor” may be used interchangeably, and refer to the ability of a molecule to decrease the activation of KIT either directly or indirectly, thereby decreasing activation and/or quantity of systemic mast cells.
- Compounds that are selective for KIT may be particularly useful in the treatment of certain disorders or may offer a reduced likelihood of undesired side effects.
- compounds of the present disclosure are selective over one or more other receptor tyrosine kinases.
- specific examples include, but are not limited to, PDGFR ⁇ , PDGFR ⁇ , CSF1R, and FLT3.
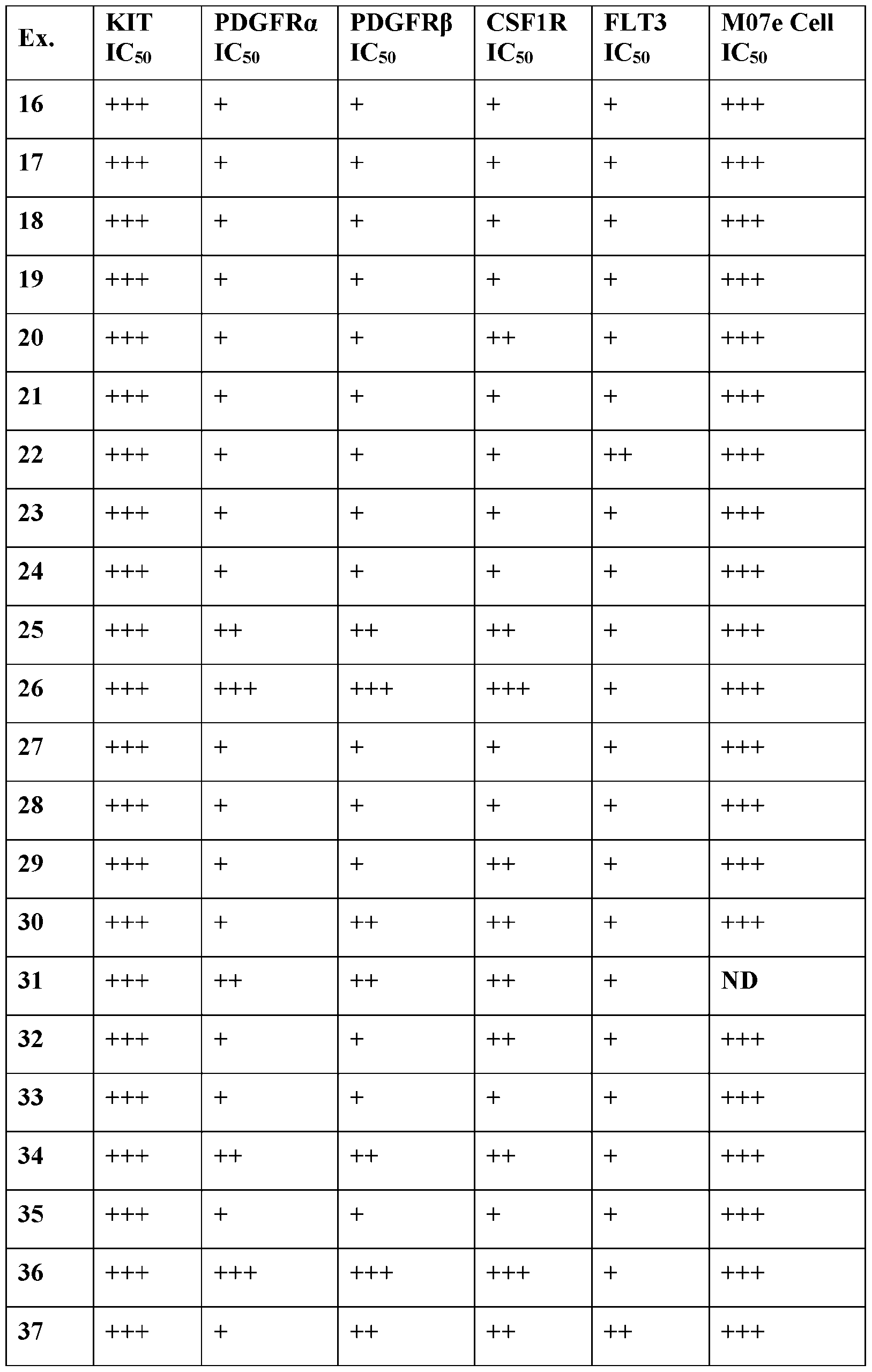
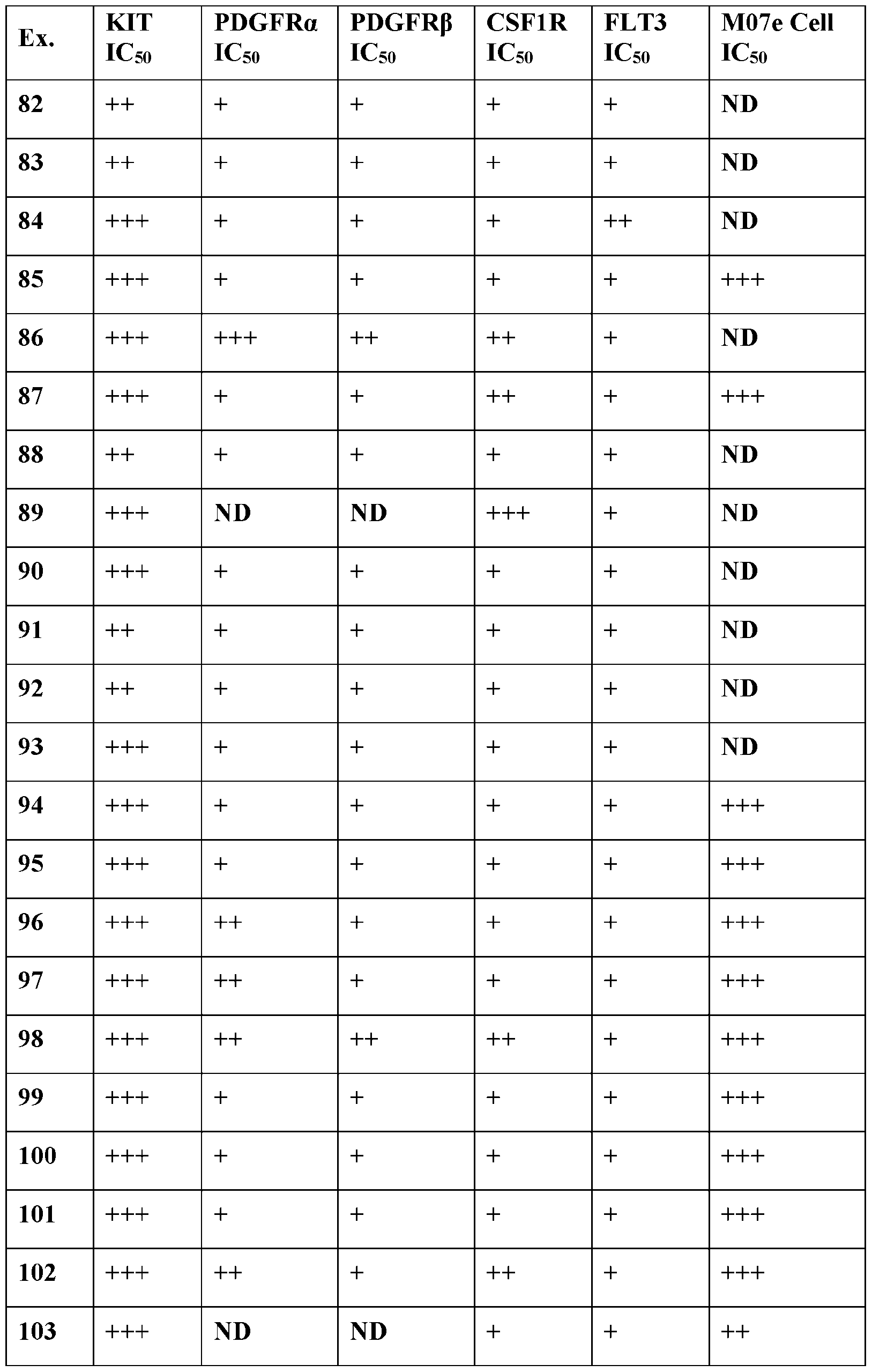
- Selectivity may be determined, for example, by comparing the inhibition of a compound as described herein against KIT against the inhibition of a compound as described herein against another kinase.
- the selective inhibition of a compound of Formula III or an associated sub-formula is at least 1000 times greater, 500 times greater, 100 times greater, 50 times greater, or 20 times greater than inhibition of one or more kinases selected from PDGFR ⁇ , PDGFR ⁇ , CSF1R, and FLT3.
- Compounds provided herein may have advantageous pharmacokinetic profiles including, for example, hepatocyte stability, clearance, inhibition against CYP, and inhibition against hERG.
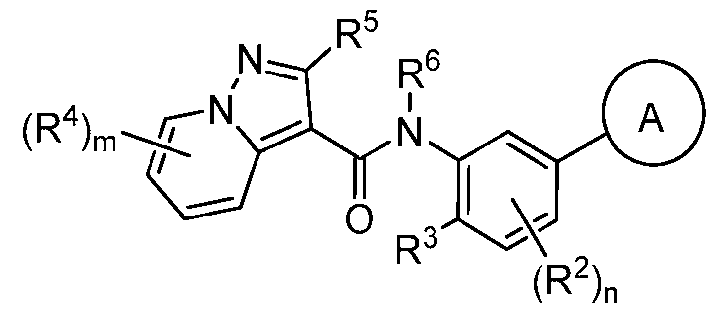
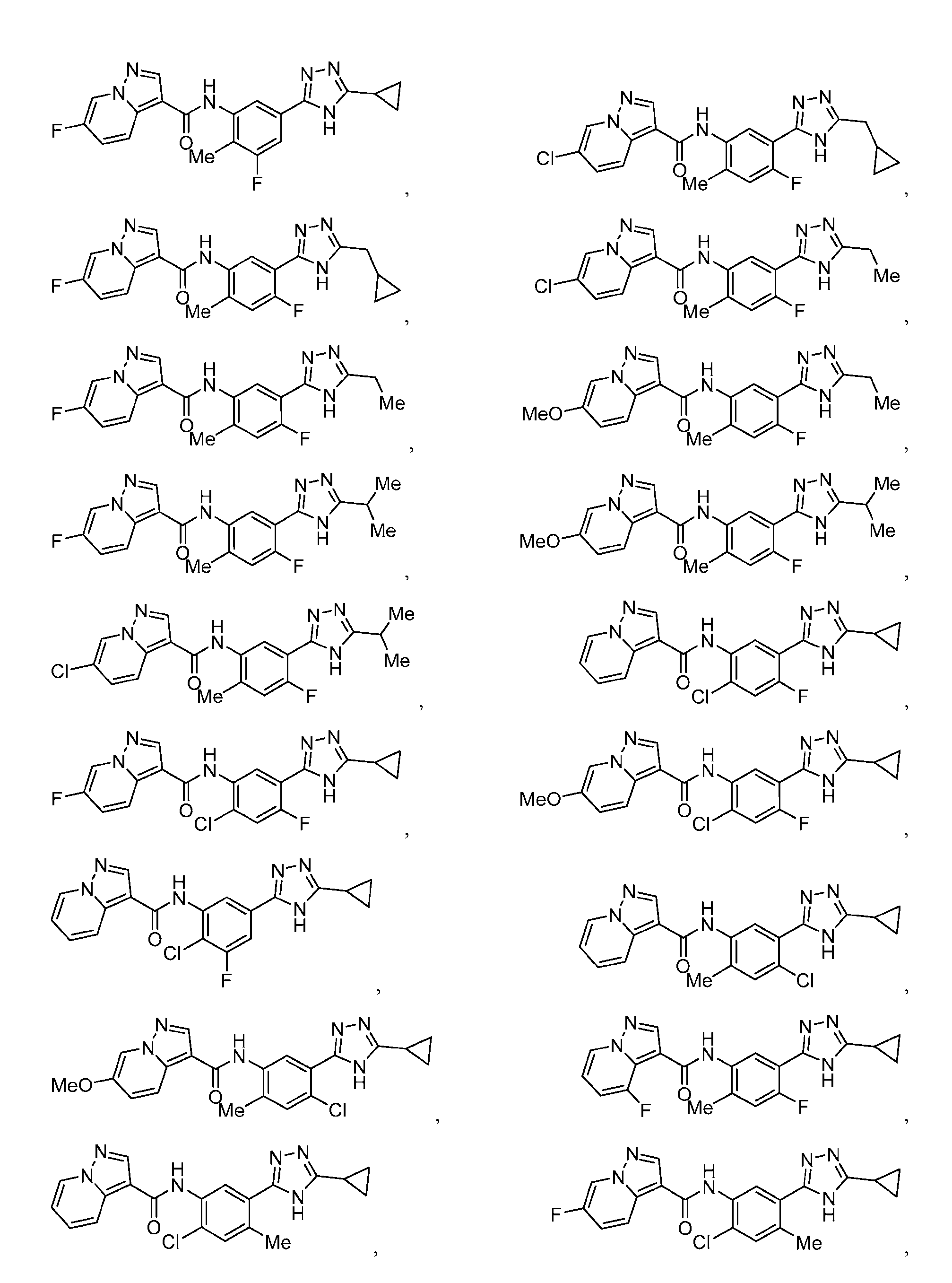
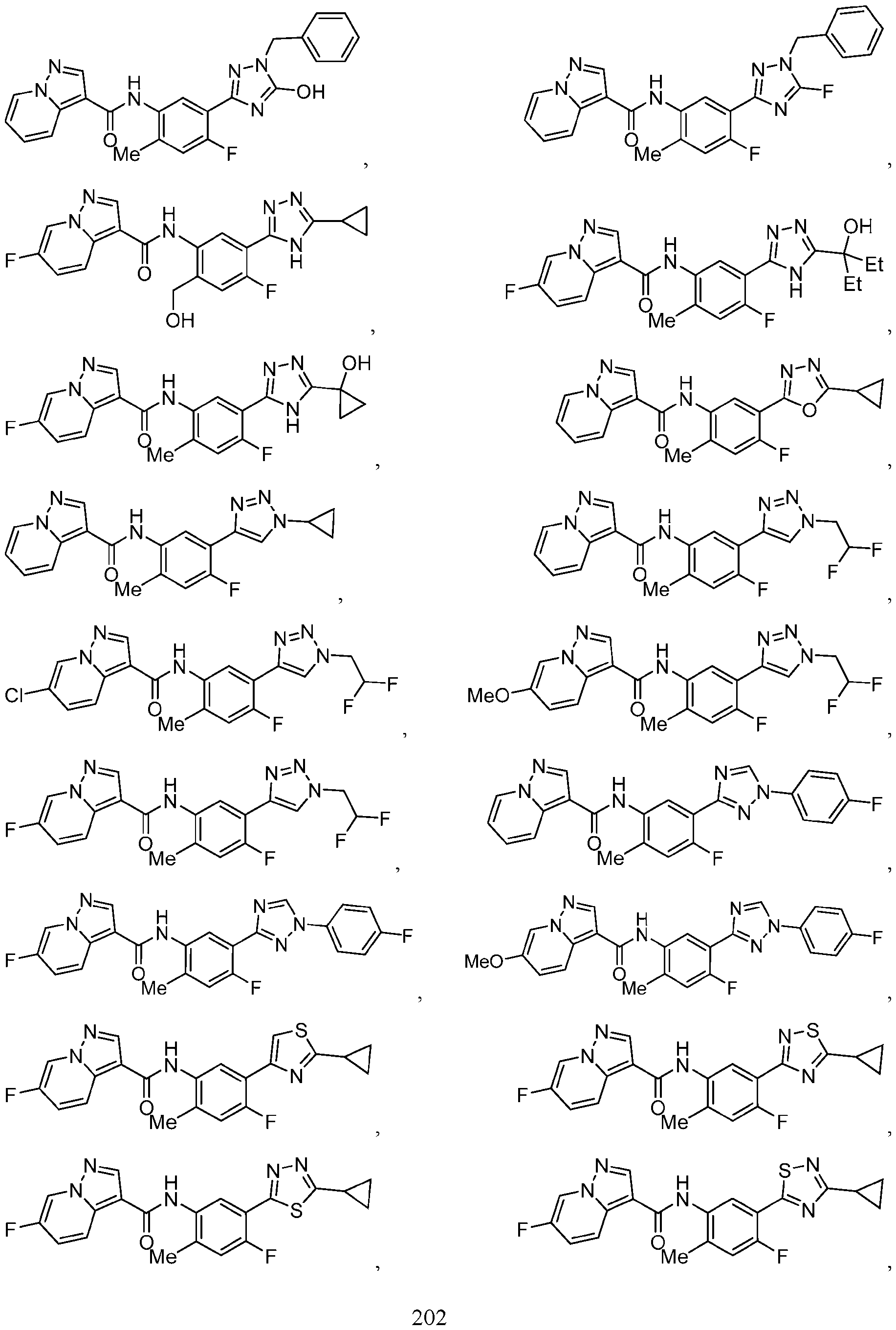
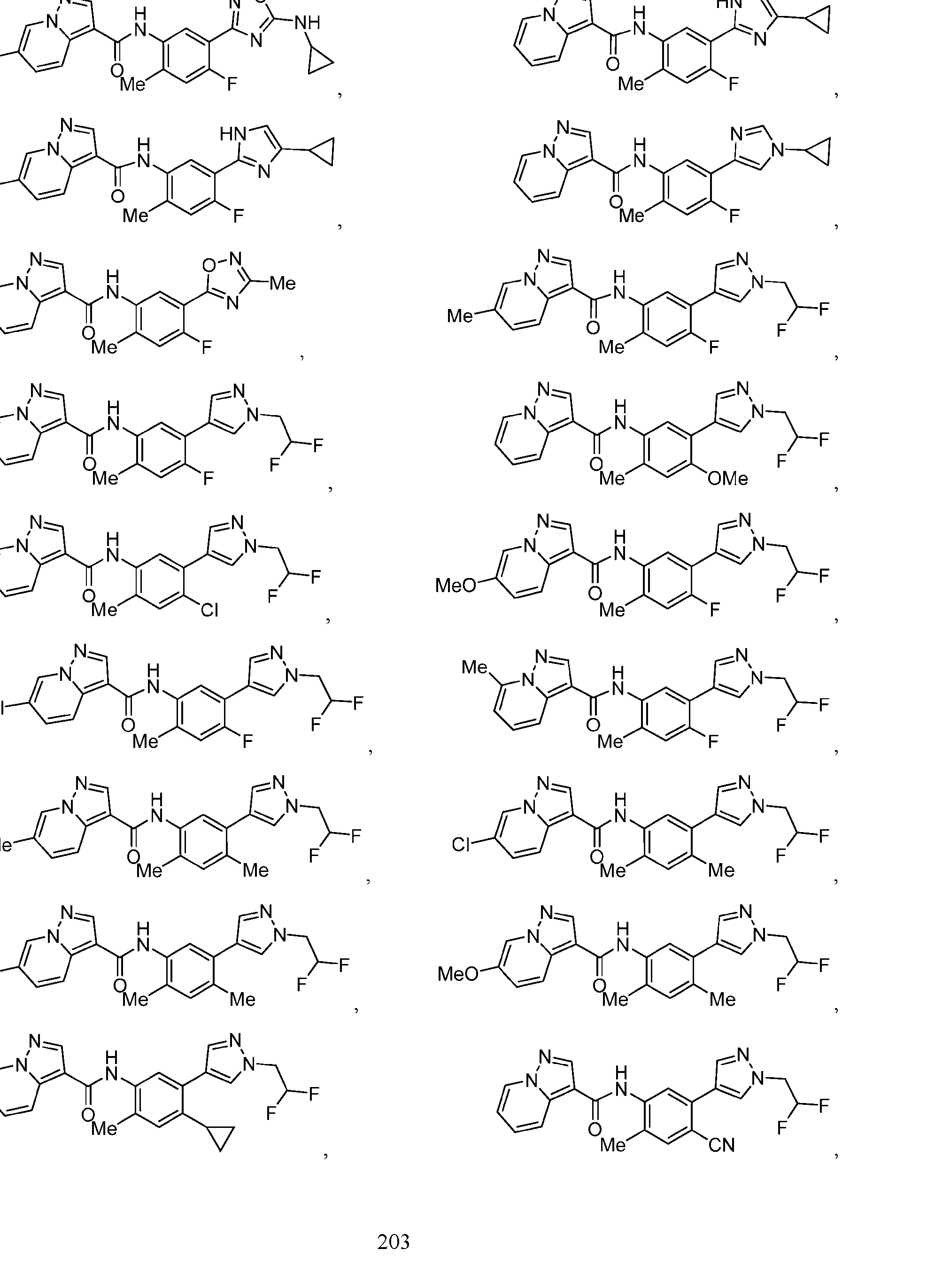
- Compounds of the Disclosure [0037] The present disclosure relates to compounds that inhibit the activity of KIT.
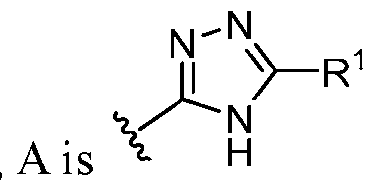
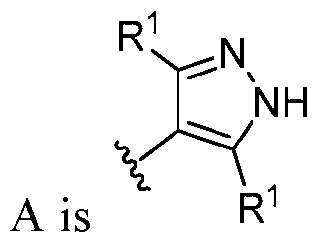
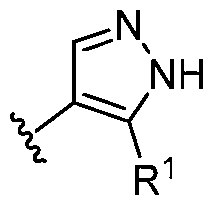
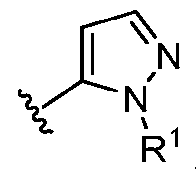
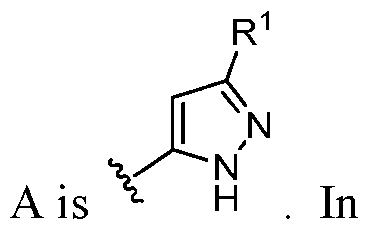
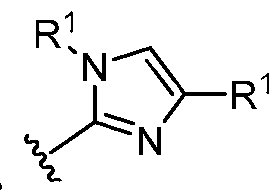
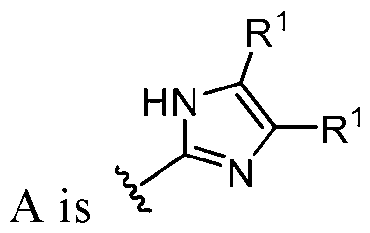
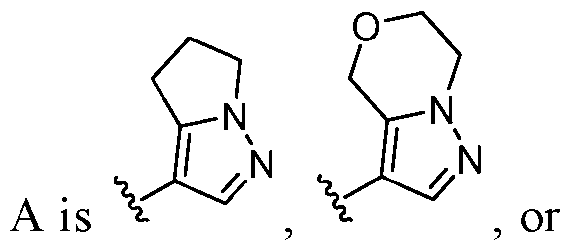
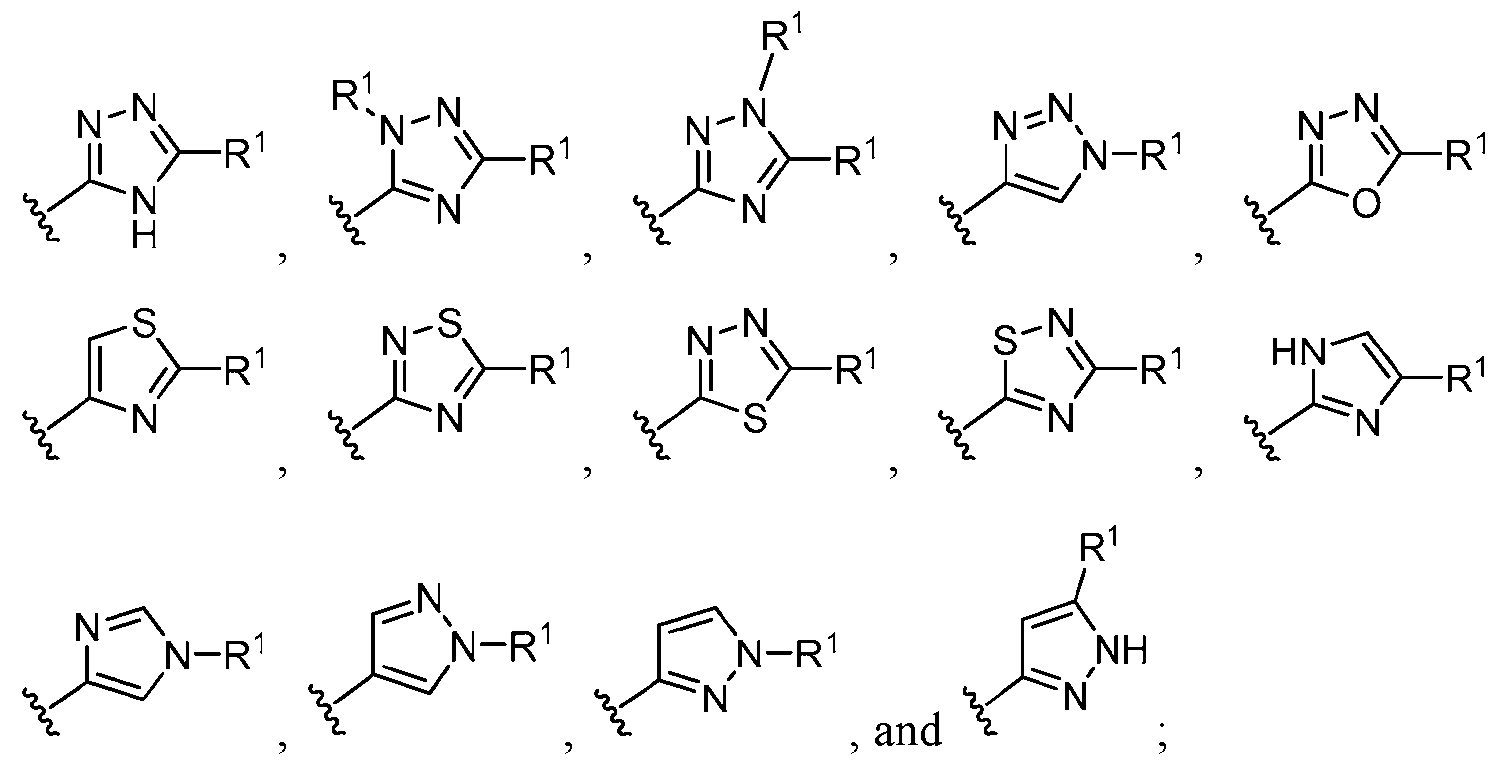
- this disclosure is directed to a compound having a structure according to Formula III: (Formula III) or a pharmaceutically acceptable salt thereof, wherein: A is: (i) selected from the group consisting of: , , membered heteroaryl is substituted with 0-2 R 1d ; or (iii) -NHC(O)-R 1g ; each R 1 is independently selected from the group consisting of halo, -CN, -OH, C 1 -C 6 - alkyl, C1-C6-haloalkyl, C1-C6-hydroxyalkyl, C1-C6-cyanoalkyl, C1-C6 alkoxy, C3-C6-cycloalkyl, - O-C3-C6-cycloalkyl, 4- to 8-membered heterocycloalkyl, -NR 1a R 1b , phenyl, 5- to 6-membered 12 heteroaryl, -X 1 -NR
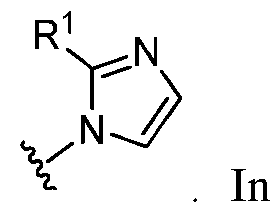
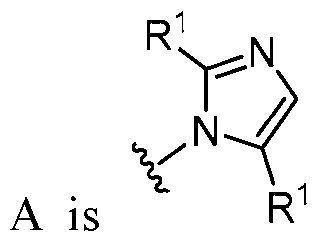
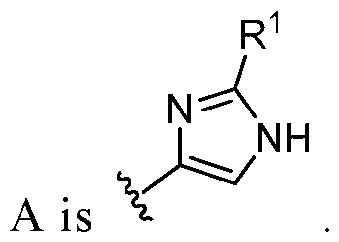
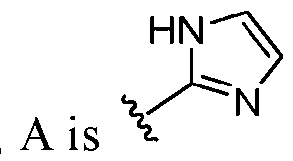
- A is selected from the group consisting of: , , , [0040] In some embodiments, . In some embodiments, A is some embodiments, A is . In some embodiments, A is . In some embodiments, A is . In some embodiments, A . In some embodiments, , . In some embodiments, A is . In some embodiments, A is . In some embodiments, A is . In some embodiments, A is . In some embodiments, A is . In some embodiments, A is . In some embodiments, A is . In some embodiments, A is . In some embodiments, A is . In some embodiments, A is . In some embodiments, A is . In some embodiments, A is . In some embodiments, A is . In some embodiments, A is . In some embodiments, A is . In some embodiments, A is . In some embodiments, A is . In some embodiments, A is . In some embodiments, A is . In some embodiments, A
- A is . In some embodiments, A is . some embodiments, A is . some embodiments, . In some embodiments, A is . In some embodiments, A is . In some embodiments, A is . In some embodiments, A is . In some embodiments, A is . In some embodiments, A is . In some embodiments, A is . In some embodiments, A is . In some embodiments, A is . In some embodiments, A is . , . , . In some embodiments, A is . In some embodiments, . In some embodiments, some embodiments, A is . In some embodiments, . In some embodiments, some embodiments, some embodiments, some embodiments, A is . In some embodiments, . In some embodiments, some embodiments, some embodiments, A is . , . In some embodiments, A is some embodiments, A is . In some embodiments, . In some embodiments, some embodiments, A is some embodiments, A is . In some embodiment
- A is . In some embodiments, A is . In some embodiments, A is . In some embodiments, A is . In some embodiments, A is . In some embodiments, A , . In some embodiments, A is . , . In some embodiments, . In some embodiments, . In some embodiments, . In some embodiments, A is . In some embodiments, . In some embodiments, A is some embodiments, A is . In some embodiments, . In some embodiments, . In some embodiments, A is .
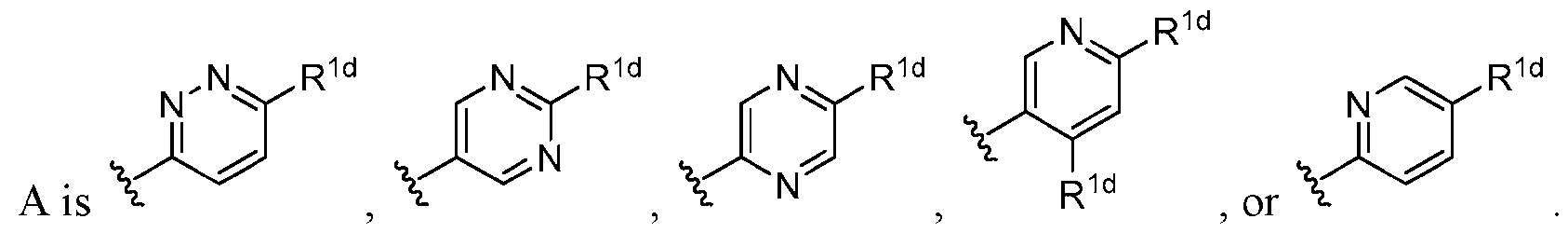
- A is a 6-membered heteroaryl having 1-3 ring nitrogen atoms, wherein the 6-membered heteroaryl is substituted with 0-2 R 1d .
- A is a 6- membered heteroaryl having 1-3 ring nitrogen atoms, wherein the 6-membered heteroaryl is substituted with 1-2 R 1d .
- A is pyridazinyl, pyrimidinyl, pyrazinyl, or pyridinyl, wherein A is substituted with 0-2 R 1d .
- A is pyridazinyl, pyrimidinyl, pyrazinyl, or pyridinyl, wherein A is substituted with 1-2 R 1d . In some embodiments, .
- each R 1d when present, is independently halo, C 1 -C 6 alkoxy, C 1 - C3 haloalkoxy, cyclopropyl, -O-cyclopropyl, or -NR 1e R 1f ; and R 1e and R 1f , when present, are independently H, C1-C6 haloalkyl, -(methylene)-(cyclopropyl), or tetrahydrofuranyl; or R 1e and R 1f together with the nitrogen atom to which they are attached form an azetidinyl group.
- A is -NHC(O)-R 1g .
- R 1g when present, is cyclopropyl, -(methylene)-(cyclopropyl), pyridinyl, or pyrazinyl; wherein R 1g is optionally substituted with 0-2 R 1h .
- R 1g when present, is , , , .
- this disclosure is directed to a compound having a structure according to Formula III: (Formula III) or a pharmaceutically acceptable salt thereof, wherein: ring A is a 5-membered heteroaryl having 1-4 ring heteroatoms independently selected from N, O, and S, wherein the heteroaryl is substituted with 0-2 R 1 ; each R 1 is independently selected from the group consisting of halo, -CN, -OH, C1-C6- alkyl, C 1 -C 6 -haloalkyl, C 1 -C 6 -hydroxyalkyl, C 1 -C 6 -cyanoalkyl, C 1 -C 6 alkoxy, C 3 -C 6 -cycloalkyl, - O-C 3 -C 6 -cycloalkyl, 4- to 8-membered heterocycloalkyl, -NR 1a R 1b , phenyl, 5- to 6-membered heteroaryl, -X 1 -NR 1
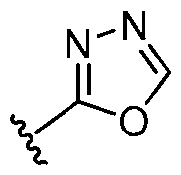
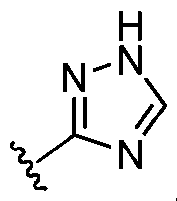
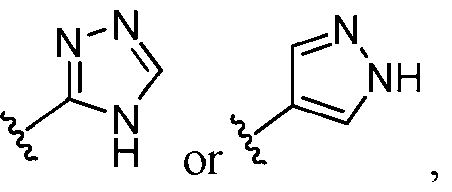
- ring A is selected from the group consisting of oxadiazolyl, thiazolyl, thiadiazolyl, triazolyl, imidazolyl, pyrazolyl, and tetrazolyl, each of which is substituted with 0-2 R 1 .
- ring A is selected from the group consisting of thiazolyl, thiadiazolyl, triazolyl, imidazolyl, pyrazolyl, and tetrazolyl, each of which is substituted with 0-2 R 1 .
- ring A is imidazolyl, pyrazolyl, triazolyl, or tetrazolyl, each of which is substituted with 0-2 R 1 .
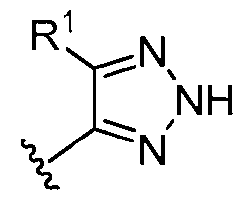
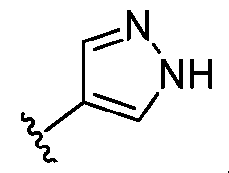
- ring A is selected from the group consisting of , , , , , , , , , , each of which is substituted with 0-2 R 1 .
- ring A , , with 0-2 R 1 ring A is selected from the group consisting of, , , , , , , , , , , , each of which is substituted with 0-2 R 1 .
- ring A is each of which is substituted with 0-2 R 1 . In some embodiments, ring A is substituted with 0-2 R 1 . In some embodiments, ring . [0049] In some embodiments, ring A is a 5-membered heteroaryl having 1-4 ring nitrogen atoms, wherein the heteroaryl is substituted with two adjacent R 1 groups that combine with the two ring atoms to which they are attached to form a 5- or 6-membered heterocycloalkyl group having 1-2 ring heteroatoms independently selected from N and O.
- ring A is pyrazolyl or triazolyl substituted with two adjacent R 1 groups that combine with the two ring atoms to which they are attached to form a 5- or 6-membered heterocycloalkyl group having 1-2 ring heteroatoms independently selected from N and O.
- ring r .
- R 6 is H, or C1-C3-alkyl. In some embodiments, R 6 is H or methyl. In some embodiments, R 6 is methyl. In some embodiments, R 6 is H.
- R 5 is H, C1-C3-alkyl, or -NR 5a R 5b ; wherein R 5a and R 5b are independently H or C1-C3 alkyl.
- R 5 is H, methyl, or -NH2.
- R 5 is H.
- R 5 is methyl.
- R 5 is -NR 5a R 5b , wherein at least one of R 5a and R 5b is H.
- R 5 is -NH2.
- R 5 and R 6 are H.
- m is 0, 1, or 2; and each R 4 , when present is independently halo, C1-C6 alkyl, C1-C6-alkoxy, C3-C6 cycloalkyl, 4- to 8-membered heterocycloalkyl having 1-3 ring heteroatoms independently selected from N, O, and S, or phenyl, wherein said phenyl is optionally substituted with 1-3 halo.
- m is 0.
- m is 1; and R 4 is halo, C 1 -C 6 alkyl, C 1 -C 6 -alkoxy, C 3 -C 6 cycloalkyl, 4- to 8-membered heterocycloalkyl having 1-3 ring heteroatoms independently selected from N, O, and S, or phenyl, wherein said phenyl is optionally substituted with 1-3 halo.
- m is 2; and each R 4 is independently halo, C 1 -C 6 alkyl, C 1 -C 6 -alkoxy, C 3 -C 6 cycloalkyl, 4- to 8-membered heterocycloalkyl having 1-3 ring heteroatoms independently selected from N, O, and S, or phenyl, wherein said phenyl is optionally substituted with 1-3 halo.
- m is 0, 1, or 2; and at least one R 4 , when present is halo.
- R 4 is -F, -Cl, -Br, or -I.
- R 4 is -F, or -Cl.
- R 4 is -F. In some embodiments, R 4 is -Cl. In some embodiments, m is 0, 1, or 2, and at least one R 4 , when present, is C1-C6 alkyl. In some embodiments, R 4 is C1- C 3 alkyl. In some embodiments, R 4 is methyl, ethyl, n-propyl, or isopropyl. In some embodiments, R 4 is methyl, or ethyl. In some embodiments, R 4 is methyl. In some embodiments, R 4 is ethyl. In some embodiments, m is 0, 1, or 2; and at least one R 4 , when present, is C1-C6-alkoxy.
- R 4 is C1-C3-alkoxy. In some embodiments, R 4 is methoxy, ethoxy, n-propoxy, or isopropoxy. In some embodiments, R 4 is methoxy or ethoxy. In some embodiments R 4 is methoxy. In some embodiments, R 4 is ethoxy. In some embodiments, m is 0, 1, or 2; and at least one R 4 , when present, is C3-C6 cycloalkyl. In some embodiments, R 4 is cyclopropyl, cyclobutyl, cyclopentyl, or cyclohexyl. In some embodiments, R 4 is cyclopropyl.
- m is 0, 1, or 2; and at least one R 4 , when present, is 4- to 8-membered heterocycloalkyl having 1-3 ring heteroatoms independently selected from N, O, and S.
- R 4 is a 5- to 6- membered heterocycloalkyl having 1-3 ring heteroatoms independently selected from N, O, and S.
- R 4 is a 6-membered heterocycloalkyl having 1-3 ring heteroatoms independently selected from N, and O.
- R 4 is piperidine, piperazine, or morpholine. In some embodiments, R 4 is morpholine.
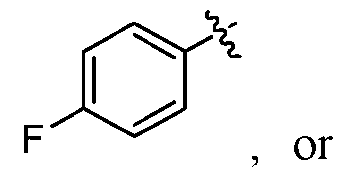
- m is 0, 1, or 2; and at least one R 4 , when present, is phenyl optionally substituted with 1-3 halo. In some embodiments, R 4 is phenyl substituted with one halo. In some embodiments, R 4 is phenyl substituted with -F, - Cl, -Br, or -I. In some embodiments, R 4 is phenyl substituted with -F. In some embodiments, m is 0, 1, or 2; and each R 4 , when present, is independently -F, -Cl, -CH 3 , -CH 2 CH 3 , -OCH 3 , - OCH 2 CH 3 , cyclopropyl, .
- m is 1; and R 4 , is independently -F, -Cl, -CH 3 , -CH 2 CH 3 , -OCH 3 , -OCH 2 CH 3 , cyclopropyl, r .
- m is 2; and each R 4 is independently -F, -Cl, -CH 3 , -CH 2 CH 3 , - OCH 3 , -OCH 2 CH 3 , cyclopropyl, .
- m is 0, 1, or 2; and each R 4 , when present is independently halo, C 1 -C 6 alkyl, C 1 -C 6 -alkoxy, or C 3 -C 6 cycloalkyl. In some embodiments, m is 0 or 1; and R 4 , when present is halo, C 1 -C 6 alkyl, C 1 -C 6 -alkoxy, or C 3 -C 6 cycloalkyl. In some embodiments, m is 0. In some embodiments, m is 1, and R 4 is halo, C1-C6 alkyl, C1-C6-alkoxy, or C3-C6 cycloalkyl.
- R 4 is halo, C1-C3-alkyl, C1-C3-alkoxy, or C3-C5-cycloalkyl. In some embodiments, R 4 is -F, -Cl, -CH 3 , -CH 2 CH 3 , -OCH 3 , -OCH 2 CH 3 , or cyclopropyl. [0055] In some embodiments, m is 0, 1, or 2; and each R 4 is independently halo, or C1-C6-alkoxy. In some embodiments, at least one R 4 is halo. In some embodiments, at least one R 4 is Cl, Br, or F. In some embodiments, at least one R 4 is Cl.
- At least one R 4 is C 1 -C 6 - alkoxy. In some embodiments, at least one R 4 is C 1 -C 3 -alkoxy. In some embodiments, at least one R 4 is -OCH3. In some embodiments, m is 0, 1, or 2; and each R 4 is independently Cl, or - OCH 3 . [0056] In some embodiments, R 3 is halo, C 1 -C 6 alkyl, or C 1 -C 6 hydroxyalkyl. In some embodiments, R 3 is halo, C1-C3 alkyl, or C1-C3 hydroxyalkyl. In some embodiments, R 3 is halo.
- R 3 is -F, -Cl, or -Br. In some embodiments, R 3 is -F, or -Cl. In some embodiments, R 3 is -F. In some embodiments, R 3 is -Cl. In some embodiments, R 3 is C1-C3 alkyl. In some embodiments, R 3 is -CH3. In some embodiments, R 3 is C1-C3 hydroxyalkyl. In some embodiments, R 3 is -CH 2 -OH, -CH 2 CH 2 -OH, or -CH 2 CH 2 CH 2 -OH. In some embodiments, R 3 is -CH2-OH.
- R 3 is -F, -Cl, -CH3, or -CH2-OH. [0057] In some embodiments, R 3 is halo or C1-C6-alkyl. In some embodiments, R 3 is halo, or C1- C 3 -alkyl. In some embodiments, R 3 is halo. In some embodiments, R 3 is -F, -Cl, or -Br. In some embodiments, R 3 is -Cl. In some embodiments, R 3 is C 1 -C 3 alkyl. In some embodiments, R 3 is - CH3. In some embodiments, R 3 is -Cl, or -CH3.
- n is 0 or 1, and R 2 , when present is halo, -CN, -C 1 -C 3 -alkyl, -C 1 - C 3 -alkoxy, or C 3 -C 6 -cycloalkyl. In some embodiments, n is 0. In some embodiments, n is 1. In some embodiments, R 2 , when present, is halo. In some embodiments, R 2 , when present, is -F, - Cl, -Br, or -I. In some embodiments, R 2 , when present, is -F, or -Cl. In some embodiments, R 2 , when present, is -F.
- R 2 when present, is -Cl. In some embodiments, R 2 , when present, is -C1-C3-alkyl. In some embodiments, R 2 , when present, is methyl, ethyl, n-propyl, or isopropyl. In some embodiments, R 2 , when present, is methyl. In some embodiments, R 2 , when present, is -C 1 -C 3 -alkoxy. In some embodiments, R 2 , when present, is methoxy, ethoxy, n- propoxyl, or isopropoxy. In some embodiments, R 2 , when present, is methoxy.
- R 2 when present, is C3-C6-cycloalkyl. In some embodiments, R 2 , when present, is cyclopropyl, cyclobutyl, cyclopentyl, or cyclohexyl. In some embodiments, R 2 , when present, is cyclopropyl. In some embodiments, n is 0 or 1, and R 2 , when present, is -F, -Cl, -CN, -CH 3 , - OCH3, or cyclopropyl. [0059] In some embodiments, n is 1, and R 2 is halo. In some embodiments, n is 1 and R 2 is -F or -Cl.
- each R 1 is independently halo, -CN, -OH, C1-C6-alkyl, C1-C6- haloalkyl, C1-C6-hydroxyalkyl, C1-C6 alkoxy, C3-C6-cycloalkyl, -O-C3-C6-cycloalkyl, 4- to 6- membered heterocycloalkyl, -NR 1a R 1b , phenyl, 6-membered heteroaryl, -X 1 -C(O)NR 1a R 1b , -X 1 -O- (C1-C3-alkyl), -X 1 -C3-C6-cycloalkyl, and -X 1 -phenyl; wherein each 6-membered heteroaryl has 1- 2 ring heteroatoms independently selected from N and O; each 4- to 6-membered heterocycloalkyl has 1-3 ring heteroatoms independently selected from N, O, and S;
- each R 1 is independently selected from the group consisting of C1-C6-alkyl, C1-C6-haloalkyl, C1-C6 alkoxy, C3-C6-cycloalkyl, 4- to 8-membered heterocycloalkyl having 1-2 ring heteroatoms independently selected from N and O, -NR 1a R 1b , and -X 1 -C3-C6- cycloalkyl; wherein X 1 is C 1 -C 3 -alkylene; and R 1a and R 1b , when present, are independently H, or C 1 -C 3 alkyl.
- R 1 is selected from the group consisting of C1-C6-alkyl, C1-C6- haloalkyl, C 3 -C 6 -cycloalkyl, and -X 1 -C 3 -C 6 -cycloalkyl, wherein X 1 is C 1 -C 3 -alkylene.
- R 1 is C 1 -C 6 -alkyl.
- R 1 is C 1 -C 4 -alkyl.
- R 1 is methyl, ethyl, n-propyl, iso-propyl, iso-butyl, or tert-butyl.
- R 1 is C1-C6-haloalkyl. In some embodiments, R 1 is C1-C3- haloalkyl. In some embodiments, R 1 is difluoromethyl, trifluoromethyl, trifluoroethyl, difluoroethyl, trifluoropropyl, or difluoropropyl. In some embodiments, R 1 is difluoromethyl, trifluoromethyl, trifluoroethyl, or trifluoropropyl. In some embodiments, R 1 is -CF2H, CF3, - CH 2 CF 2 H, -CH 2 CF 3 , or -CH 2 CH 2 CF 3 .
- R 1 is C 1 -C 6 alkoxy. In some embodiments, R 1 is C 1 -C 3 alkoxy. In some embodiments, R 1 is methoxy. [0066] In some embodiments, R 1 is C 3 -C 6 -cycloalkyl optionally substituted with 1-3 R 1c . In some embodiments, R 1 is C 3 -C 6 -cycloalkyl optionally substituted with 1-2 R 1c . In some embodiments, R 1 is C3-C5-cycloalkyl optionally substituted with 1-3 R 1c . In some embodiments, R 1 is C3-C5-cycloalkyl optionally substituted with 1-2 R 1c .
- R 1 is cyclopropyl, cyclobutyl, cyclopentyl, or cyclohexyl, each of which is optionally substituted with 1-3 R 1c .
- R 1 is cyclopropyl, cyclobutyl, cyclopentyl, or cyclohexyl, each of which is optionally substituted with 1-2 R 1c .
- R 1 is cyclopropyl, cyclobutyl, or cyclopentyl, each of which is optionally substituted with 1-3 R 1c .
- R 1 is cyclopropyl, cyclobutyl, or cyclopentyl, each of which is optionally substituted with 1-2 R 1c .
- each R 1c when present, is independently halo or -OH.
- each R 1c when present, is independently -F, or -OH.
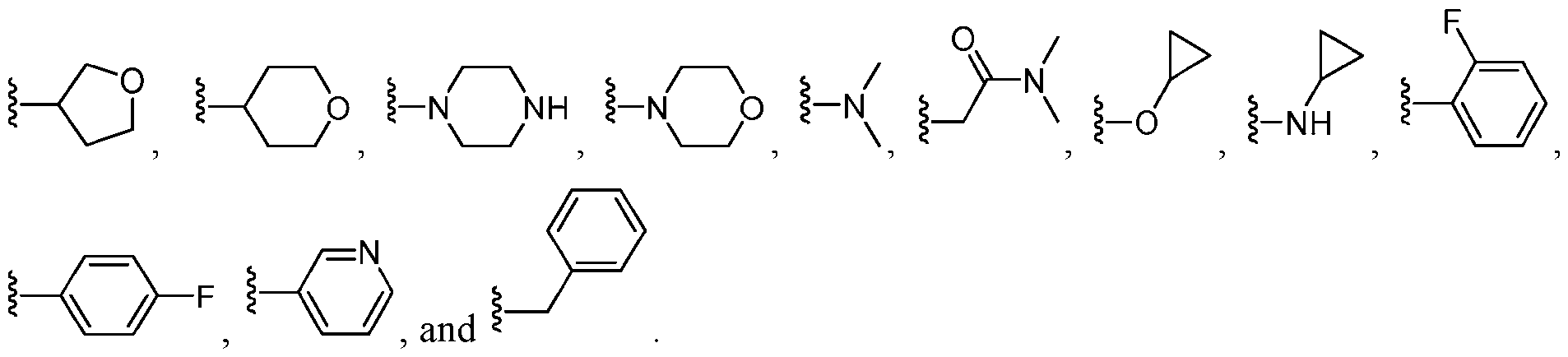
- R 1 is , . [0067]
- R 1 is C 3 -C 6 -cycloalkyl.
- R 1 is C 3 -C 5 - cycloalkyl.
- R 1 is cyclopropyl, cyclobutyl, cyclopentyl, or cyclohexyl.
- R 1 is cyclopropyl, cyclobutyl, or cyclopentyl.
- R 1 is -O-C 3 -C 6 -cycloalkyl.
- R 1 is -O- cyclopropyl, -O-cyclobutyl, -O-cyclopentyl, or -O-cyclohexyl.
- R 1 is -O- cyclopropyl.
- R 1 is 4- to 8-membered heterocycloalkyl having 1-2 ring heteroatoms independently selected from N and O.
- R 1 is 5- to 6-membered heterocycloalkyl having 1-2 ring heteroatoms independently selected from N and O.
- R 1 is pyrrolidinyl, tetrahydrofuranyl, tetrahydropyranyl, piperidinyl, or morpholinyl.
- R 1 is tetrahydrofuranyl, tetrahydropyranyl, or morpholinyl.
- R 1 is , , . In some embodiments, R 1 is .
- R 1 is -NR 1a R 1b , wherein R 1a and R 1b are independently H, C 1 -C 3 alkyl, or C 3 -C 6 cycloalkyl.
- R 1 is -NR 1a R 1b , wherein R 1a and R 1b are independently H, or C1-C3 alkyl.
- at least one of R 1a and R 1b is C1-C3 alkyl.
- both R 1a and R 1b are C1-C3 alkyl.
- at least one of R 1a and R 1b is C 3 -C 6 cycloalkyl.
- anti-inflammatory agents include non-steroidal anti-inflammatory drugs (NSAIDs) (e.g., ibuprofen, aspirin, naproxen, and celecoxib, etodolac, meloxicam, nabumetone, diclofenac, diflunisal, fenoprofen, and flurbiprofen); corticosteroids (e.g., cortisone, prednisone, prednisolone, methylprednisolone, dexamethasone, deflazacort, betamethasone, hydrocortisone, etc.); disease-modifying antirheumatic drugs (DMARDs) (e.g., methotrexate, sulfasalazine, hydroxychloroquine, and leflunomide); anti-tumor necrosis factor (anti-TNF) agents (e.g., infliximab, adalimumab, and
- the additional therapeutic agent comprises an antipsychotic.
- antipsychotics include haloperidol, loxapine, thioridazine, molindone, thiothixene, fluphenazine, mesoridazine, trifluoperazine, perphenazine, chlorpromazine, aripiprazole, clozapine, ziprasidone, risperidone, quetiapine, and olanzapine.
- the additional therapeutic agent comprises one or more anti- anxiety agents.
- anti-anxiety agents include alprazolam, chlordiazepoxide, clonazepam, diazepam, lorazepam, and buspirone.
- the additional therapeutic agent comprises one or more anticonvulsants (e.g., valproic acid, phenytoin, clonazepam, and carbamazepine).
- the additional therapeutic agent comprises one or more respiratory agents.
- the respiratory agent is a bronchodilator (e.g., adrenergic bronchodilator, anticholinergic bronchodilator, methylxanthines, and combinations thereof), an inhaled corticosteroid (e.g., beclomethasone, fluticasone, ciclesonide, mometasone, and budesonide), a beta andrenergic agonist (e.g., albuterol, metaproterenol, pirbuterol, terbutaline, isoetharine and levalbuterol), or leukotriene modifier (e.g., montelukast, zafirlukast, and zileuton).
- a bronchodilator e.g., adrenergic bronchodilator, anticholinergic bronchodilator, methylxanthines, and combinations thereof
- an inhaled corticosteroid e.g., beclomet
- the additional therapeutic agent comprises one or more nasal decongestants.
- exemplary decongestants include oxymetazoline, phenylephrine, and pseudoephedrine.
- the additional therapeutic agent comprises a cough suppressant.
- exemplary cough suppressants include dextromethorphan, guaifenesin, and codeine.
- the compounds according to this disclosure are combined with a proton pump inhibitor (PPI).
- PPIs include lansoprazole, omeprazole, pantoprazole, rebaprazole, and esomeprazole.
- the additional therapeutic agent comprises an agent that modulates cognitive function, e.g., cholinesterase inhibitors (e.g., donepezil, rivastigmine, galantamine), N-methyl-D-aspartate (NMDA) receptor antagonists (e.g., memantine), and agents targeting aggregated soluble and insoluble forms of amyloid beta (e.g., aducanumab).
- cholinesterase inhibitors e.g., donepezil, rivastigmine, galantamine
- NMDA N-methyl-D-aspartate
- amyloid beta e.g., aducanumab
- the additional therapeutic agent comprises an agent that targets thyroid function, such as, for example, anti-thyroid agents (e.g., radioiodine, propylthiouracil (PTU), and methimazole), or thyroid hormone replacement therapy (e.g., levothyrozine, or cytomel).
- anti-thyroid agents e.g., radioiodine, propylthiouracil (PTU), and methimazole
- thyroid hormone replacement therapy e.g., levothyrozine, or cytomel.
- the additional therapeutic agent comprises one or more agents useful in the treatment of diabetes, such as, e.g., insulin; synthetic glucagon; hyperglycemic agents (e.g., metformin, sulfonylureas, glinides, thiazolidinediones, dipeptidyl peptidase-4 (DPP- 4) inhibitors; anti-hyperglycemic agents (e.g., sodium glucose cotransporter-2 (SGLT2) inhibitors including, e.g., canagliflozin, dapagliflozin, and empagliflozin); and GLP-1 receptor agonists (e.g., semaglutide, exenatide, dulaglutide, liraglutide, or lixisenatide).
- agents useful in the treatment of diabetes such as, e.g., insulin; synthetic glucagon; hyperglycemic agents (e.g., metformin, sulfonylureas, glin
- the additional therapeutic agent comprises a diuretic.
- diuretics include spironolactone, bumetanide, torsemide, hydrochlorothiazide, furosemide, and metolazone, and aldosterone antagonists (e.g., spironolactone and eplerenone).
- the additional therapeutic agent comprises one or more of an antidiarrheal (e.g., eluxadoline, or alosetron), a laxative (lubiprostone, or a guanylate cyclase- C (GC-C agonist (e.g., linaclotide).
- an antidiarrheal e.g., eluxadoline, or alosetron
- laxative lubiprostone
- GC-C agonist e.g., linaclotide
- the additional therapeutic agent comprises a cholinergic modulator, such as a cholinergic agonist (e.g., chantix, pilocarpine, or bethanechol), or an anticholinergic agent (e.g., atropine, belladonna alkaloids, benztropine mesylate, clidinium, cyclopentolate, darifenacin, dicylomine, fesoterodine, flavoxate, glycopyrrolate, homatropine hydrobromide, hyoscyamine, ipratropium, orphenadrine, oxybutynin, propantheline, scopolamine, methscopolamine, solifenacin, tiotropium, tolterodine, trihexphenidyl, and trospium).
- a cholinergic modulator such as a cholinergic agonist (e.g., chantix, pilocarpine, or bethanechol), or
- the additional therapeutic agent comprises a vasodilator.
- vasodilators include, but are not limited to nitrates (e.g., nitroprusside, nitroglycerine, isosorbide, and amyl nitrate), hydralazine, treprostinil, minoxidil, angiotensin-converting enzyme (ACE) inhibitors (e.g., benazepril, captopril, enalapril, fosinopril, lisinopril, moexipril, perindopril, quinapril, ramipril, and trandolapril), and angiotensin receptor blockers (ARBs) (e.g., azilsartan, candesartan, eprosartan, irbesartan, losartan, olmesartan, telmisartan, and valsartan).
- ACE angiotensin-converting
- the additional therapeutic agent comprises a thrombolytic agent (e.g., streptokinase, alteplase, reteplase, Tenecteplase, urokinase, prourokinase, and anistreplase).
- a thrombolytic agent e.g., streptokinase, alteplase, reteplase, Tenecteplase, urokinase, prourokinase, and anistreplase.
- the additional therapeutic agent comprises an anticoagulant. Exemplary anticoagulants include rivaroxaban, dabigatran, apixaban, eboxaban, and warfarin.
- the additional therapeutic agent comprises an agent useful in the treatment of fibrosis. Certain such agents include pirfenidone and nintedanib.
- the additional therapeutic agent comprises a targeted agent useful in the treatment of pulmonary arterial hypertension.
- Targeted agents useful in the treatment of pulmonary arterial hypertension include phosphodiesterase-5 (PDE5) inhibitors (e.g., sildenafil, tadalafil and vardenafil); guanylate cyclase stimulators (GCS) (e.g., adempas, riociguat, vericiguat and verquvo); endothelin receptor antagonists (e.g., bosentan, ambrisentan, and macitentan), and prostacyclin and analogues thereof.
- PDE5 phosphodiesterase-5
- GCS guanylate cyclase stimulators
- endothelin receptor antagonists e.g., bosentan, ambrisentan, and macitentan
- Exemplary CFTR modulators include ivacaftor, elexacaftor, lumacaftor, and tezacaftor.
- the additional therapeutic agent comprises an antibiotic.
- Exemplary antibiotics include, but are not limited to phenoxymethylpenicillin, dicloxacillin, amoxicillin, ampicillin, nafcillin, oxacillin, penicillin, cefaclor, cefazolin, cefadroxil, cephalexin, cefuroxime, cefixime, ceroxitin, ceftriaxone, doxycycline, minocycline, sarecycline, erythromycin, clarithromycin, azithromycin, fidaxomicin, roxithromycin, ciprofloxacin, ofloxacin, levofloxacin, moxifloxacin, sulfamethoxazole with trimethoprim, sulfasalazine
- the additional therapeutic agent comprises one or more agents selected from the groups consisting of anti-inflammatory agents, analgesic agents, agents that target one or more cytokines, immunosuppressants, agents that targets one or more mast-cell derived immunomodulators, BTK inhibitors, IgE inhibitors, anti-depressants, anti-psychotics, anti-anxiety agents, anticonvulsants, respiratory agents, nasal decongestants, cough suppressants, proton pump inhibitors (PPIs), agents that modulate cognitive function, agents that target thyroid function, agents useful in the treatment of diabetes, diuretics, antidiarrheals, laxatives, GC-C agonists, cholinergic modulators, antiarrhythmics, vasodilators, cholesterol modifiers, thrombolytic agents, anticoagulants, agents useful in the treatment of fibrosis, agents useful in the treatment of arterial hypertension, mucolytic agents, pancreatic enzymes, CFTR modulators, and/or antibiotics.
- BTK inhibitors IgE inhibitors
- the additional therapeutic agent comprises one or more agents selected from the group consisting of an anti-inflammatory agent, an analgesic agent, an immunosuppressant, and/or an agent that targets one or more cytokines (e.g., IL-12, IL-17, and/or IL-23).
- the additional therapeutic agent comprises one or more agents selected from the group consisting of a respiratory agent, an anti-inflammatory agent, an agent that targets one or more cytokines (e.g., IL-4 and/or IL-13), a mast-cell stabilizer, and/or an agent that targets a mast-cell derived immunomodulator (e.g., leukotrienes).
- the additional therapeutic agent comprises one or more agents selected from the group consisting of an anti-depressant, an anti-psychotic, an anti-anxiety agent, an anticonvulsant, an agent that modulates cognitive function, an anti-CD20 antibody (e.g., ocrelizumab), an anti-inflammatory, and/or an immunosuppressant.
- the additional therapeutic agent comprises an anti-inflammatory and/or an immunosuppressant.
- the additional therapeutic agent comprises an anti-inflammatory agent, an immunosuppressant, and/or an agent that targets a mast-cell derived immunomodulator (e.g., antihistamine and/or leukotriene modulators).
- the additional therapeutic agent comprises an antihistamine, a BTK inhibitor, and/or an IgE inhibitor.
- the additional therapeutic agent comprises an antihistamine, an anti-inflammatory agent (e.g., a corticosteroid), and IgE inhibitor, and/or an immunosuppressant (e.g., cyclosporine).
- the additional therapeutic agent comprises an anti-inflammatory agent, an immunosuppressive agent, an agent that targets one or more cytokines, or a combination thereof.
- the additional therapeutic agent comprises a cholesterol modifier, a diuretic, an antiarrhythmic, a vasodilator, an anti-inflammatory, an analgesic agent, or any combination thereof.
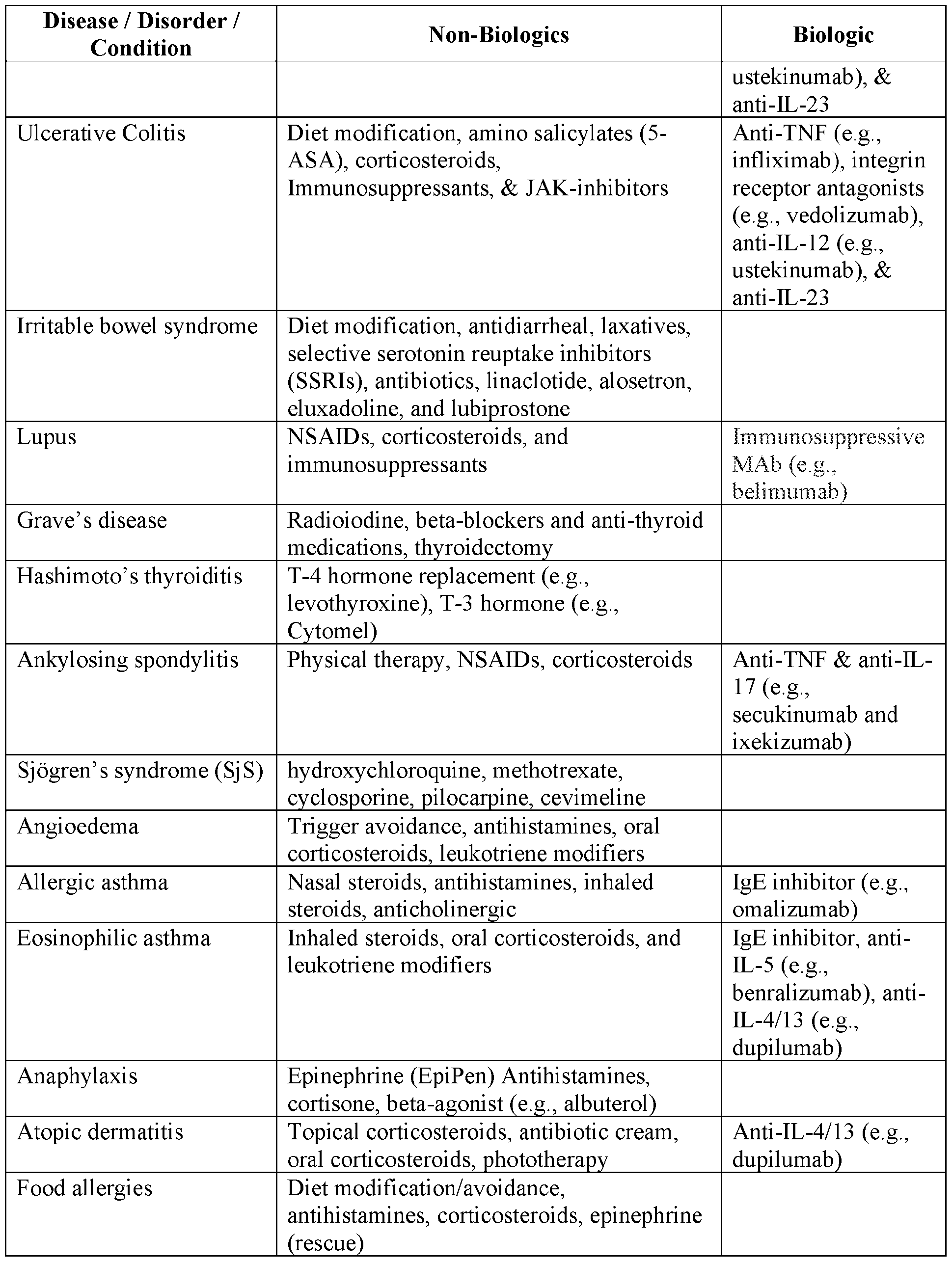
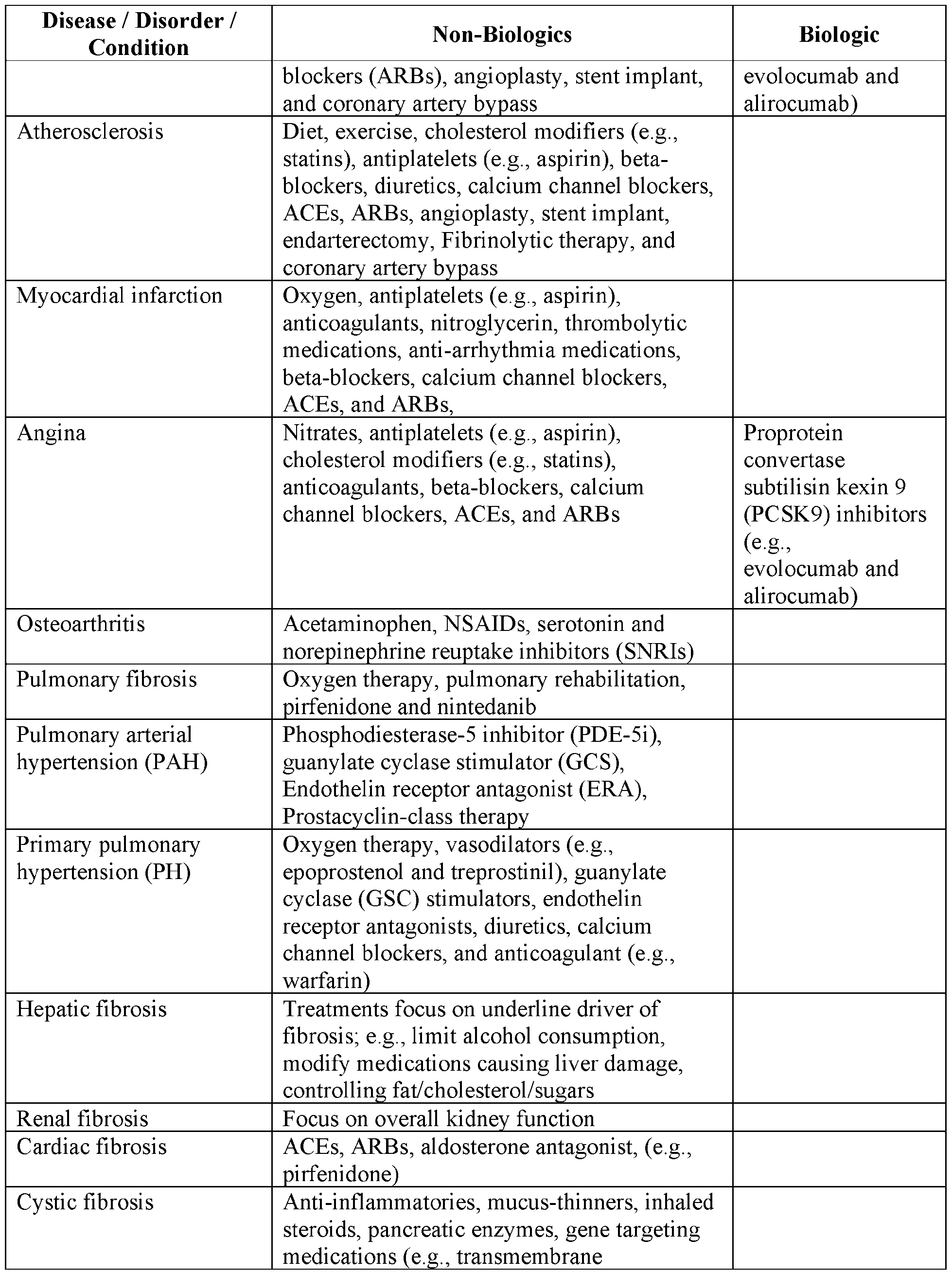
- the compounds described herein are combined with one or more additional therapeutic agents that are considered to be the standard of care (SOC) for one or more of the inflammatory, immune, and/or autoimmune-related indications described herein.
- SOC standard of care
- Exemplary SOC therapies for the indications described herein are summarized in Table 1 and Table 2 below. Table 1 66 Table 2 68 Cancer Therapies [0186]
- the present disclosure contemplates the use of the KIT inhibitors described herein in combination with one or more additional therapies useful in the treatment of cancer.
- one or more of the additional therapies is an additional treatment modality.
- Exemplary treatment modalities include but are not limited to surgical resection of a tumor, bone marrow transplant, radiation therapy, and photodynamic therapy.
- one or more of the additional therapies is a therapeutic agent.
- Exemplary therapeutic agents include chemotherapeutic agents, radiopharmaceuticals, hormone therapies, epigenetic modulators, ATP-adenosine axis-targeting agents (e.g., CD73 inhibitors, CD39 inhibitors, A 2A R inhibitors, and/or A 2B R inhibitors), signal transduction inhibitors (e.g., inhibitors of one or more of TYRO3, MERTK, EGFR, FGFR, VEGFR, HER-2, HER-3, BRAF, RET, MET, ABL, ALK, FLT-3, JAK, STAT, NF-kB), RAS signaling inhibitors (e.g., inhibitors of one or more of KRAS, HRAS, RAF, MEK, ERK, PTEN, SOS (e.g., SOS1), mTORC1, SHP2 (PTPN11), and AKT), PI3K inhibitors, arginase inhibitors, HIF inhibitors (e.g., inhibitors of HIF- 2 ⁇ ), A
- one or more of the additional therapeutic agents is a chemotherapeutic agent.
- chemotherapeutic agents include, but are not limited to, alkylating agents such as thiotepa and cyclosphosphamide; alkyl sulfonates such as busulfan, improsulfan and piposulfan; aziridines such as benzodopa, carboquone, meturedopa, and uredopa; ethylenimines and methylamelamines including altretamine, triethylenemelamine, triethylenephosphoramide, triethylenethiophosphoramide and trimethylolomelamime; nitrogen mustards such as chlorambucil, chlornaphazine, cholophosphamide, estramustine, ifosfamide, mechlorethamine, mechlorethamine oxide hydrochloride, melphalan, novembichin, phenesterine, prednimustine, tro
- combination therapy comprises a chemotherapy regimen that includes one or more chemotherapeutic agents.
- combination therapy comprises a chemotherapeutic regimen comprising FOLFOX (folinic acid, fluorouracil, and oxaliplatin), FOLFIRI (e.g., folinic acid, fluorouracil, and irinotecan), a taxoid (e.g., docetaxel, paclitaxel, nab- paclitaxel, etc.), and gemcitabine.
- FOLFOX folinic acid, fluorouracil, and oxaliplatin
- FOLFIRI e.g., folinic acid, fluorouracil, and irinotecan
- a taxoid e.g., docetaxel, paclitaxel, nab- paclitaxel, etc.
- gemcitabine e.g., gemcitabine.
- one or more of the additional therapeutic agents is an immune checkpoint inhibitor.
- immune checkpoint inhibitor refers to an antagonist of an inhibitory or co-inhibitory immune checkpoint.
- immunoreactive checkpoint inhibitor refers to an antagonist of an inhibitory or co-inhibitory immune checkpoint.
- checkpoint inhibitor checkpoint inhibitor
- CPI CPI-associated receptor -ligand binding and/or altering receptor signaling.
- immune checkpoints ligands and receptors
- PD-1 programmed cell death protein 1
- PD-L1 PD1 ligand
- BTLA B and T lymphocyte attenuator
- CTLA-4 cytotoxic T-lymphocyte associated antigen 4
- TIM-3 T cell immunoglobulin and mucin domain containing protein 3
- LAG-3 lymphocyte activation gene 3
- TIGIT T cell immunoreceptor with Ig and ITIM domains
- CD276 B7-H3
- PD-L2 Galectin 9, CEACAM-1, CD69, Galectin-1, CD113, GPR56, VISTA, 2B4, CD48, GARP, PD1H, LAIR1, TIM-1, and TIM-4, and Killer Inhibitory Receptors, which can be divided into two classes based on their structural features: i) killer cell immunoglobulin- like receptors (KIRs), and
- an immune checkpoint inhibitor is a CTLA-4 antagonist.
- the CTLA-4 antagonist can be an antagonistic CTLA-4 antibody.
- Suitable antagonistic CTLA-4 antibodies include, for example, monospecific antibodies such as ipilimumab or tremelimumab, as well as bispecific antibodies such as MEDI5752 and KN046.
- an immune checkpoint inhibitor is a PD-1 antagonist.
- the PD-1 antagonist can be an antagonistic PD-1 antibody, small molecule or peptide.
- Suitable antagonistic PD-1 antibodies include, for example, monospecific antibodies such as balstilimab, budigalimab, camrelizumab, cosibelimab, dostarlimab, cemiplimab, ezabenlimab (BI-754091), MEDI-0680 (AMP-514; WO2012/145493), nivolumab, pembrolizumab, pidilizumab (CT-011), pimivalimab, retifanlimab, sasanlimab, spartalizumab, sintilmab, tislelizumab, toripalimab, and zimberelimab; as well as bi-specific antibodies such as LY3434172.
- monospecific antibodies such as balstilimab, budigalimab, camrelizumab, cosibelimab, dostarlimab, cemiplimab, ezabenlimab (
- the PD-1 antagonist can be a recombinant protein composed of the extracellular domain of PD-L2 (B7-DC) fused to the Fc portion of IgGl (AMP-224).
- an immune checkpoint inhibitor is zimberelimab.
- an immune checkpoint inhibitor is a PD-L1 antagonist.
- the PD-L1 antagonist can be an antagonistic PD-L1 antibody.
- Suitable antagonistic PD-Ll antibodies include, for example, monospecific antibodies such as avelumab, atezolizumab, durvalumab, BMS-936559, and envafolimab as well as bi-specific antibodies such as LY3434172 and KN046.
- an immune checkpoint inhibitor is a TIGIT antagonist.
- the TIGIT antagonist can be an antagonistic TIGIT antibody.
- Suitable antagonistic anti-TIGIT antibodies include monospecific antibodies such as AGEN1327, AB308 (WO2021247591), BMS 986207, COM902, domvanalimab, EOS-448, etigilimab, IBI-929, JS006, M6223, ociperlimab, SEA-TGT, tiragolumab, vibostolimab; as well as bi-specific antibodies such as AGEN1777 and AZD2936.
- an immune checkpoint inhibitor is an antagonistic anti-TIGIT antibody disclosed in WO2017152088 or WO2021247591.
- an immune checkpoint inhibitor is domvanalimab or AB308.
- an immune checkpoint inhibitor is a LAG-3 antagonist.
- the LAG-3 antagonist can be an antagonistic LAG-3 antibody.
- Suitable antagonistic LAG-3 antibodies include, for example, BMS-986016 (WO10/19570, WO14/08218), or IMP-731 or IMP-321 (WO08/132601, WO09/44273).
- an immune checkpoint inhibitor is a B7-H3 antagonist.
- the B7-H3 antagonist is an antagonistic B7-H3 antibody.
- Suitable antagonist B7-H3 antibodies include, for example, MGA271 (WO11/109400), omburtumab, enoblituzumab, DS-7300a, ABBV-155, and SHR-A1811.
- one or more of the additional therapeutic agents activates a stimulatory or co-stimulatory immune checkpoint.
- stimulatory or co-stimulatory immune checkpoints include B7-1, B7-2, CD28, 4-1BB (CD137), 4-1BBL, ICOS, ICOS-L, OX40, OX40L, GITR, GITRL, CD70, CD27, CD40, DR3 and CD2.
- an agent that activates a stimulatory or co-stimulatory immune checkpoint is a CD137 (4-1BB) agonist.
- the CD137 agonist can be an agonistic CD137 antibody.
- Suitable CD137 antibodies include, for example, urelumab and PF- 05082566 (WO12/32433).
- an agent that activates a stimulatory or co- stimulatory immune checkpoint is a GITR agonist.
- the GITR agonist can be an agonistic GITR antibody.
- Suitable GITR antibodies include, for example, BMS-986153, BMS-986156, TRX-518 (WO06/105021, WO09/009116) and MK-4166 (WO11/028683).
- an agent that activates a stimulatory or co-stimulatory immune checkpoint is an OX40 agonist.
- the OX40 agonist can be an agonistic OX40 antibody.
- Suitable OX40 antibodies include, for example, MEDI-6383, MEDI-6469, MEDI-0562, PF- 04518600, GSK3174998, BMS-986178, and MOXR0916.
- an agent that activates a stimulatory or co-stimulatory immune checkpoint is a CD40 agonist.
- the CD40 agonist can be an agonistic CD40 antibody.
- an agent that activates a stimulatory or co-stimulatory immune checkpoint is a CD27 agonist.
- the CD27 agonist can be an agonistic CD27 antibody.
- Suitable CD27 antibodies include, for example, varlilumab.
- one or more of the additional therapeutic agents is an ATP- adenosine axis-targeting agent.
- an ATP-adenosine axis-targeting agent is an inhibitor of an ectonucleotidase involved in the conversion of ATP to adenosine or an antagonist of adenosine receptor, e.g., ectonucleoside triphosphate diphosphohydrolase 1 (ENTPD1, also known as CD39 or Cluster of Differentiation 39) and the ecto-5'-nucleotidase (NT5E or 5NT, also known as CD73 or Cluster of Differentiation 73).
- ENTPD1 ectonucleoside triphosphate diphosphohydrolase 1
- N5E or 5NT also known as CD73 or Cluster of Differentiation 73
- Exemplary small molecule CD73 inhibitors include CB-708, ORIC-533, LY3475070 and AB680.
- Exemplary anti-CD39 and anti-CD73 antibodies include ES002023, TTX-030, IPH-5201, SRF-617, CPI-006, oleclumab (MEDI9447), NZV930, IPH5301, GS-1423, uliledlimab (TJD5, TJ004309), BMS-986179, and AB598.
- the present disclosure contemplates combination of the compounds described herein with a CD73 inhibitor such as those described in WO 2017/120508, WO 2018/067424, WO 2018/094148, and WO 2020/046813.
- the CD73 inhibitor is quemliclustat (AB680).
- Adenosine can bind to and activate four different G-protein coupled receptors: A 1 R, A2AR, A2BR, and A3R.
- A2R antagonists include etrumadenant, inupadenant, taminadenant, caffeine citrate, NUV-1182, TT-702, DZD-2269, INCB-106385, EVOEXS-21546, AZD-4635, imaradenant, RVU-330, ciforadenant, PBF-509, PBF-999, PBF-1129, and CS-3005.
- the present disclosure contemplates the combination of the compounds described herein with an A2AR antagonist, an A2BR antagonist, or an antagonist of A2AR and A2BR.
- the present disclosure contemplates the combination of the compounds described herein with the adenosine receptor antagonists described in WO 2018/136700, WO 2018/204661, WO 2018/213377, or WO 2020/023846.
- the adenosine receptor antagonist is etrumadenant.
- one or more of the additional therapeutic agents is an inhibitor of a hypoxia-inducible factor (HIF) transcription factor, particularly HIF-2 ⁇ .
- HIF-2 ⁇ inhibitors include belzutifan, ARO-HIF2, PT-2385, and those described in WO 2021113436, WO 2021188769, and WO 2023077046.
- the HIF-2 ⁇ inhibitor is AB521.
- one or more of the additional therapeutic agents is an inhibitor of anexelekto (AXL).
- AXL anexelekto
- the AXL signaling pathway is associated with tumor growth and metastasis, and is believed to mediate resistance to a variety of cancer therapies.
- AXL inhibitors under development that also inhibit other kinases in the TAM family (i.e., TYRO3, MERTK), as well as other receptor tyrosine kinases including MET, FLT3, RON and AURORA, among others.
- Exemplary multikinase inhibitors include sitravatinib, rebastinib, glesatinib, gilteritinib, merestinib, cabozantinib, foretinib, BMS777607, LY2801653, S49076, and RXDX- 106.
- AXL specific inhibitors have also been developed, e.g., small molecule inhibitors including DS-1205, SGI-7079, SLC-391, TP-0903 (i.e., dubermatinib), BGB324 (i.e., bemcentinib), DP3975, and AB801; anti-AXL antibodies such as ADCT-601; and antibody drug conjugates (ADCs) such as BA3011.
- Another strategy to inhibit AXL signaling involves targeting AXL’s ligand, GAS6.
- batiraxcept (AVB-500) is under development as is a Fc fusion protein that binds the GAS6 ligand thereby inhibiting AXL signaling.
- the additional therapeutic agent is an AXL inhibitor described in WO 2022246177, WO 2022246179, or PCT/US2023/069124. In some embodiments, the AXL inhibitor is AB801. [0202] In some embodiments, the additional therapeutic agent comprises chemotherapy, radiation therapy, or both. [0203] In one or more embodiments, the additional therapeutic agent comprises domvanalimab, etrumadenant, quemliclustat, zimberelimab, AB308, AB521, AB598, or AB801, or any combinations thereof.
- the additional therapeutic agent comprises one or more of an immune checkpoint inhibitor, an A2R antagonist, a CD73 inhibitor, a HIF-2 ⁇ inhibitor, a chemotherapeutic agent, radiation therapy, or any combinations thereof.
- the immune checkpoint inhibitor comprises one or more inhibitors that block the activity of at least one of PD-1, PD-L1, BTLA, LAG-3, a B7 family member, TIM-3, TIGIT or CTLA-4,
- the immune checkpoint inhibitor comprises an inhibitor of PD-1 or PD-L1;
- the immune checkpoint inhibitor is selected from the group consisting of avelumab, atezolizumab, durvalumab, dostarlimab, cemiplimab, nivolumab, pembrolizumab, sintilmab, toripalimab, and zimberelimab;
- the immune checkpoint inhibitor is zimberelima
- NCN National Comprehensive Cancer Network
- NCCN Melanoma Cutaneous v2.2021, NCCN Melanoma: Uveal v2.2021, NCCN Prostate Cancer v1.2022, NCCN Squamous Cell Skin Cancer v1.2022, NCCN Hodgkin Lymphoma v2.2022, NCCN Acute Lymphoblastic Leukemia v4.2021, NCCN Chronic Lymphocytic Leukemia/Small Lymphocytic Lymphoma v2.2022, NCCN Chronic Myeloid Leukemia v3.2022, NCCN Hairy Cell Leukemia v1.2022, NCCN Pediatric Acute Lymphoblastic Leukemia v1.2022, NCNN Small Bowel Adenocarcinoma v1.2022, NCCN Thyroid Carcinoma v3.2021, NCCN Non-Small Cell Lung Cancer v3.2022, NCCN Small Cell Lung Cancer v2.2022, NCCN Breast Cancer v2.2022, NCCN Colon Cancer v3.2021, NCCN Hepatobiliary Cancer v5.2
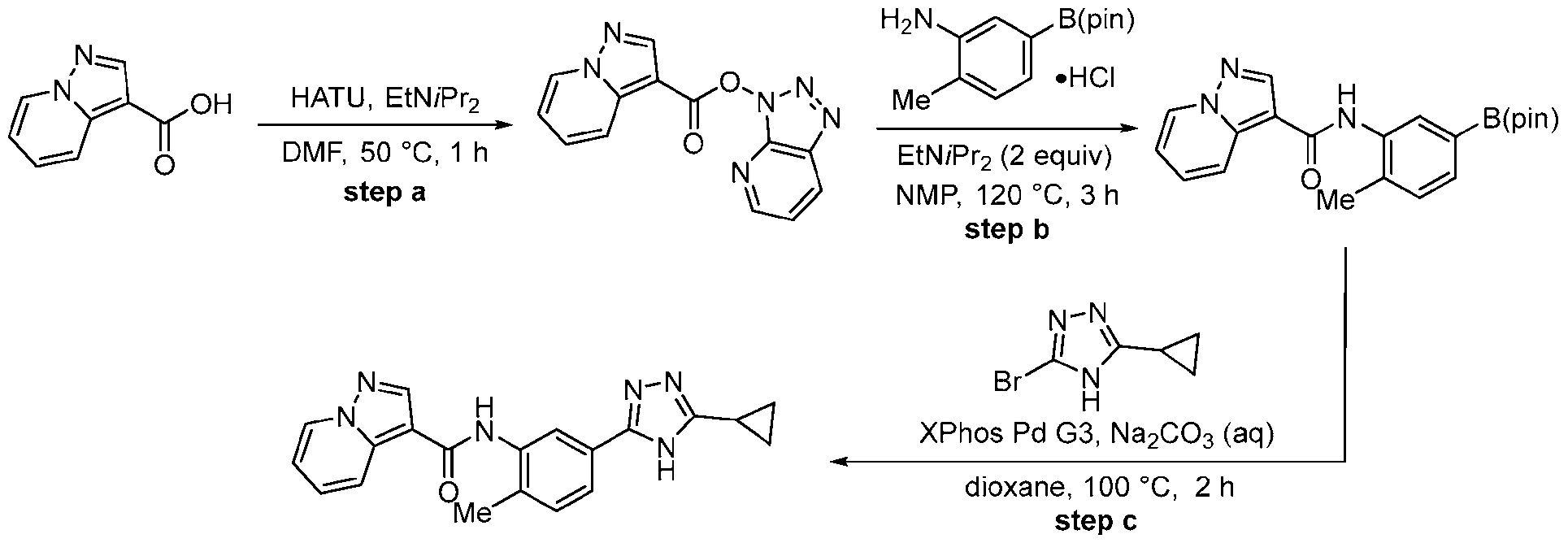
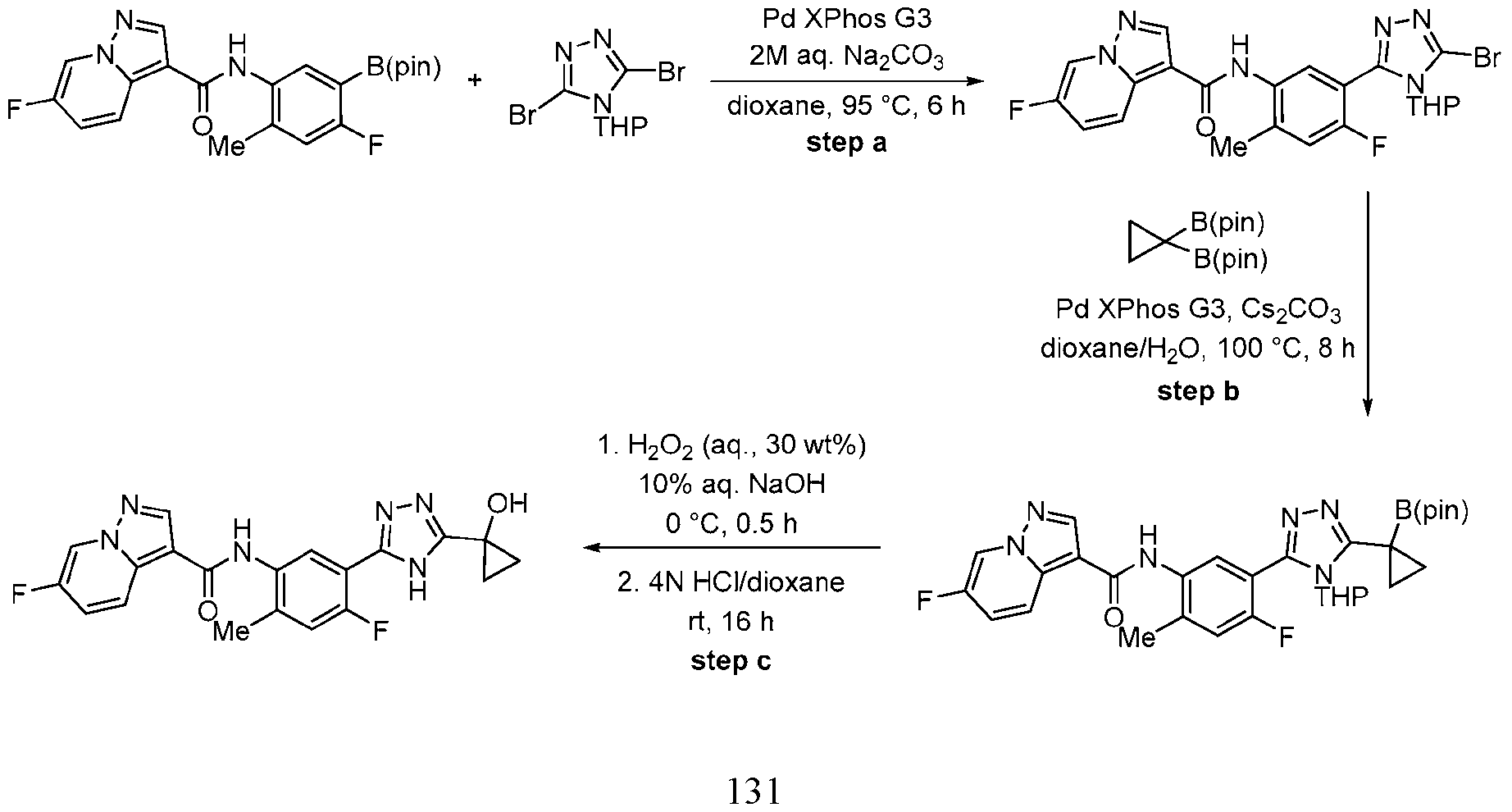
- Equation 1 illustrates a retrosynthetic disconnection of the compounds of the invention into fragments a, b and c, which are useful for the construction of the compounds according to this disclosure.
- Equation 2 demonstrates one method of forming the bond between fragments a and b.
- a readily available carboxylic acid derived from fragment a is converted to an activated intermediate, such as an acid chloride or an activated ester, using a suitable amide coupling reagent(s).
- carboxylic acids can be accomplished using a wide range of conditions and reagents, such as oxalyl chloride or thionyl chloride.
- carboxylic acids are readily converted to activated esters through the use of amide coupling reagents, such as HATU, CDI, EDC, HOBt, T3P or various other reagents (see, e.g., “Synthesis of amides” in https://www.organic- chemistry.org/synthesis/C1N/amides.shtm).
- the activated ester or acid chloride derived from fragment a can be coupled with a wide variety of amines, including but not limited to anilines (fragment b).
- the transformation may be assisted or accelerated by heating and/or addition of a base.
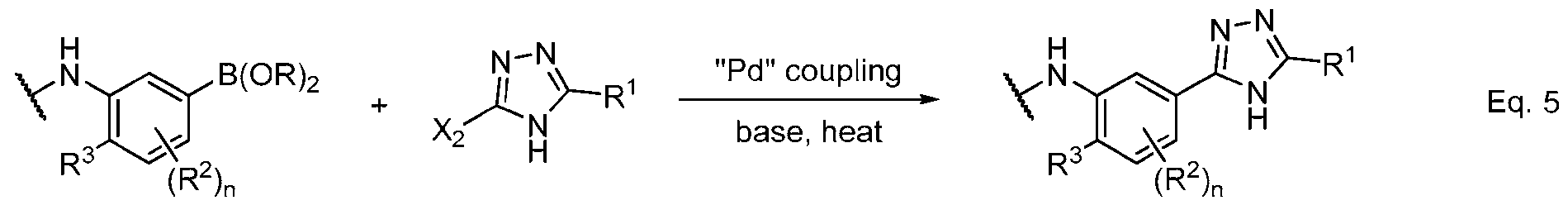
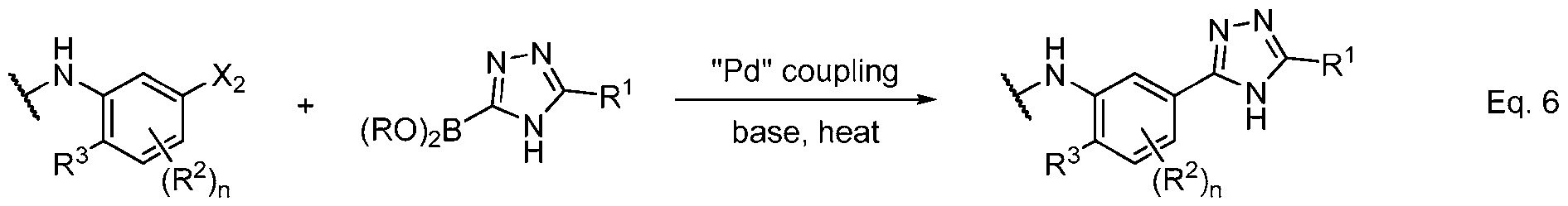
- Equations 3–6 demonstrate useful methods of synthesizing an appropriately substituted 1,2,4-triazole and connecting fragments b and c.
- an appropriately substituted aryl hydrazide readily condenses with an appropriately substituted carboximidamide or carboximidic acid ester to afford the corresponding 1,2,4-triazole, wherein “OR” typically consists of—but is not limited to—a methoxy or ethoxy group.
- Equation 5 demonstrates another method of forming the bond between fragments b and c via a Pd- catalyzed coupling reaction, such as a Suzuki reaction.
- -B(OR)2 represents a boronic acid or ester and X2 may be chosen from an appropriate halogen atom, such as Cl, Br, or I.
- the coupling is mediated by a transition metal catalyst, such as palladium with an appropriate ligand, and may be facilitated by the use of an organic or inorganic base and heating.
- a transition metal catalyst such as palladium with an appropriate ligand
- the functionalization of the coupling partners may be reversed, as exemplified in Eq. 6, and a wide variety of conditions are known in the art to effect these transformations.
- There are other possible combinations and synthetic sequences that will also give rise to the targeted products. Formation of the bond between fragments a and b may occur before or after connection of the b and c fragments, and each of these fragments may be further modified before or after connection of fragments a, b and c.
- ATP adenosine triphosphate
- BSA bovine serum albumin
- AcOH acetic acid
- DCM and CH 2 Cl 2 dichloromethane
- DIPEA and EtNiPr 2 N,N-diisopropylethylamine
- DMF N,N-dimethylformamide
- DMSO dimethyl sulfoxide
- DMSO-d6 perdeuterated dimethyl sulfoxide
- NMP N-methyl-2-pyrrolidone
- H 2 O lithium hydroxide monohydrate
- Cs2CO3 cesium carbonate
- POCl3 phosphoryl chloride
- MTBE tert-butyl methyl ether
- TBS-Cl tert-butyldimethylsilyl chloride
- Me3SiCF3 (trifluoromethyl)trimethylsilane
- TFA trifluoroacetic acid
- tBuBrettPhos 2-(di-tert- butylphosphino)-2′,4′,6′-triisopropyl-3,6-dimethoxy-1,1′-biphenyl
- PCy3 Pd G2 chloro[(tricyclohexylphosphine)-2-(2′-aminobiphenyl)]palladium(II)
- XPhos 2- dicyclohexylphosphino-2',4',6'-triisopropylbiphenyl
- XPhos Pd G3
- Flash chromatography was conducted on silica gel using an automated system (CombiFlash® RF+ manufactured by Teledyne ISCO), with detection wavelengths of 254 and 280 nm, and optionally equipped with an evaporative light scattering detector.
- Reverse phase preparative HPLC was conducted on an Agilent® 1260 or 1290 Infinity series HPLC.
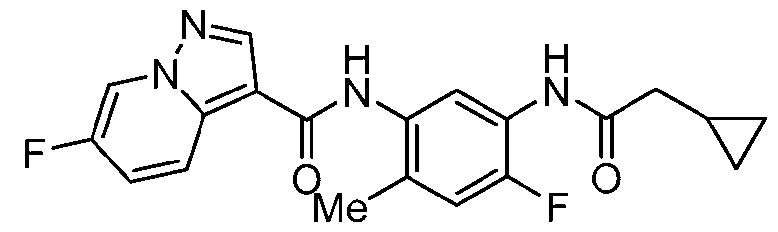
- Step a A 40 mL vial was charged with pyrazolo[1,5-a]pyridine-3-carboxylic acid (486 mg, 3.0 mmol, 1.0 equiv.), HATU (1.71 g, 4.5 mmol, 1.5 equiv.), EtNiPr2 (1.53 mL, 9.0 mmol, 3.0 equiv.) and DMF (8.6 mL, 0.35 M).
- Step b A mixture of 2-methyl-5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)aniline HCl (320 mg, 1.14 mmol, 1.0 equiv.), EtNiPr2 (0.36 ml, 2.0 equiv.) and triazolo[4,5-b]pyridin-3- yl pyrazolo[1,5-a]pyridine-3-carboxylate (280 mg, 1.04 mmol, 1.1 equiv.) in NMP (2 mL, 0.5 M) was stirred at 120 °C for approximately 3 h.
- step b Upon complete conversion, as judged by LCMS analysis, the reaction mixture was cooled to rt and diluted with water. The resulting precipitated solid was collected by vacuum filtration, rinsed with water, and dried in vacuo to afford N-[2- methyl-5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)phenyl]pyrazolo[1,5-a]pyridine-3- carboxamide, which was used directly in the next step without further purification.
- the product of step b could also be accessed via an alternative, two-step protocol involving an initial amidation with 5-bromo-2-methylaniline followed by Pd-catalyzed borylation.
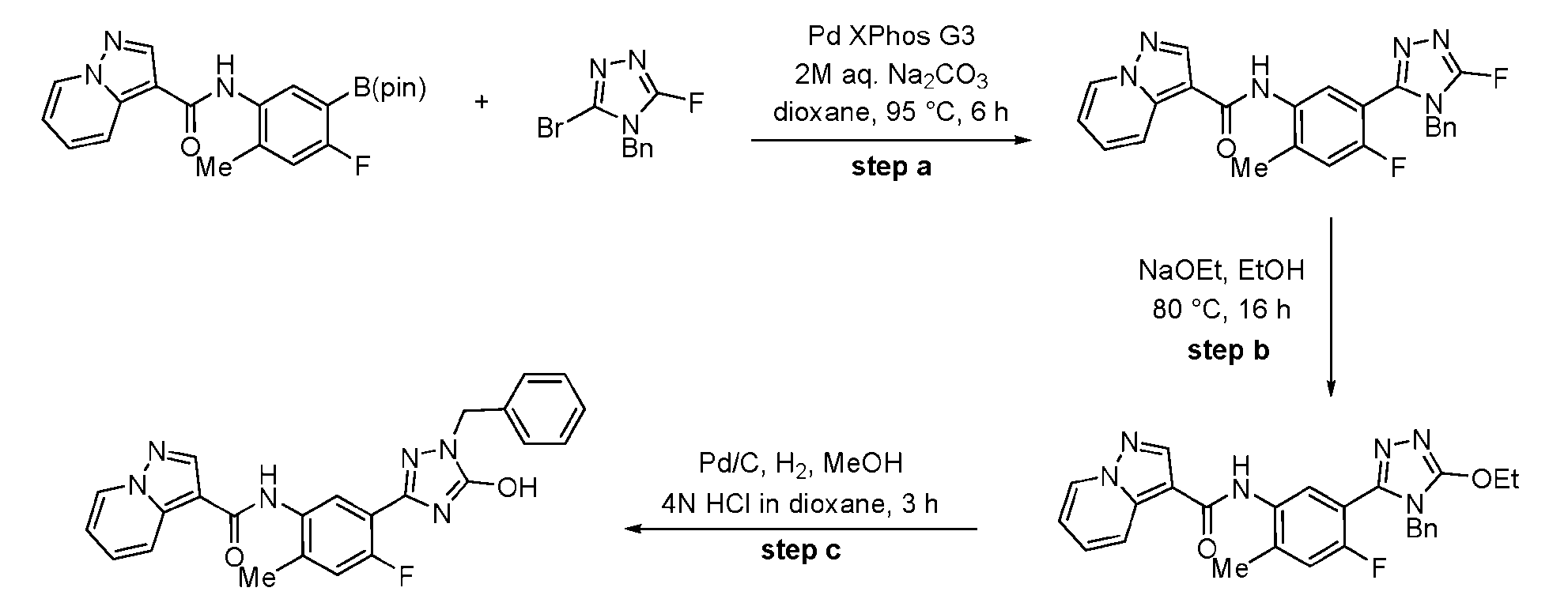
- Step c The crude intermediate obtained via step b (20 mg in 0.1 mL NMP, 0.053 mmol, 1.0 eq.) was combined with triazole bromide (20 mg, 0.106 mmol, 2.0 equiv.), XPhos Pd G3 (20 mg, 0.024 mmol, 45 mol%), 1 M aq. Na 2 CO 3 solution (0.4 mL, 0.4 mmol, 7.5 equiv.), and dioxane (0.4 mL, 0.07 M). The reaction mixture was briefly degassed by evacuation/back-filling with N2 3x. The reaction mixture was stirred at 100 °C for approximately 2 h, at which time LCMS analysis indicated complete consumption of starting material.
- Example 2 N-[5-(5-Ethyl-4H-1,2,4-triazol-3-yl)-2-methylphenyl]pyrazolo[1,5-a]pyridine-3- carboxamide [0216] The title compound was prepared from 3-bromo-5-ethyl-4H-1,2,4-triazole in a similar fashion to Ex.1.
- Example 4 N-[5-(5-tert-butyl-4H-1,2,4-triazol-3-yl)-2-methylphenyl]pyrazolo[1,5- a]pyridine-3-carboxamide [0218] The title compound was prepared from 3-bromo-5-tert-butyl-4H-1,2,4-triazole in a similar fashion to Ex.1.
- Example 6 N-[4-Fluoro-2-methyl-5-[5-(trifluoromethyl)-4H-1,2,4-triazol-3- yl]phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide [0220] The title compound was prepared from 3-bromo-5-(trifluoromethyl)-4H-1,2,4-triazole and 5-bromo-4-fluoro-2-methylaniline in a similar fashion to Ex. 1.
- Example 7 N-[5-(5-Cyclobutyl-4H-1,2,4-triazol-3-yl)-2-methylphenyl]pyrazolo[1,5- a]pyridine-3-carboxamide [0221] The title compound was prepared from 3-bromo-5-cyclobutyl-4H-1,2,4-triazole in a similar fashion to Ex.1.
- Step b A 40 mL vial was charged with the 5-[5-(cyclopropylmethyl)-4H-1,2,4-triazol- 3-yl]-4-fluoro-2-methylaniline obtained in step a (36 mg, 0.146 mmol, 1.0 equiv.), triazolo[4,5- b]pyridin-3-yl pyrazolo[1,5-a]pyridine-3-carboxylate (41 mg, 0.146 mmol, 1.0 equiv.), and NMP (0.150 mL, 1 M). The resulting mixture was stirred at 120 °C for 2 h.
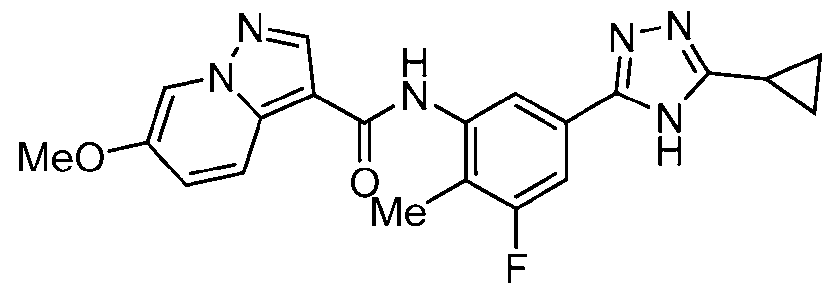
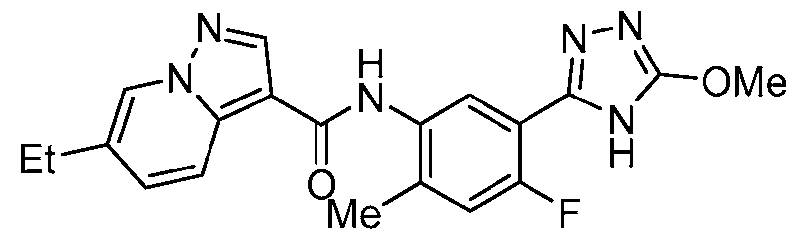
- Example 17 6-Fluoro-N-[4-fluoro-5-(5-methoxy-4H-1,2,4-triazol-3-yl)-2- methylphenyl]pyrazolo[1,5-a]pyridine-3-carboxamide
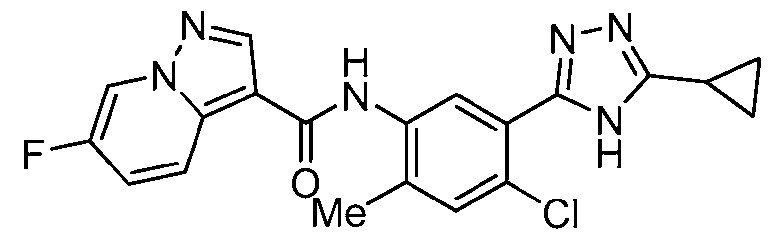
- the title compound was prepared from 6-fluoropyrazolo[1,5-a]pyridine-3-carboxylic acid, 3-bromo-5-methoxy-4H-1,2,4-triazole and 5-bromo-4-fluoro-2-methylaniline in a similar fashion to Ex.1.
- Example 18 N-[5-[5-(Dimethylamino)-4H-1,2,4-triazol-3-yl]-4-fluoro-2- methylphenyl]pyrazolo[1,5-a]pyridine-3-carboxamide [0235] The title compound was prepared from 5-bromo-N,N-dimethyl-4H-1,2,4-triazol-3-amine and 5-bromo-4-fluoro-2-methylaniline in a similar fashion to Ex. 1.
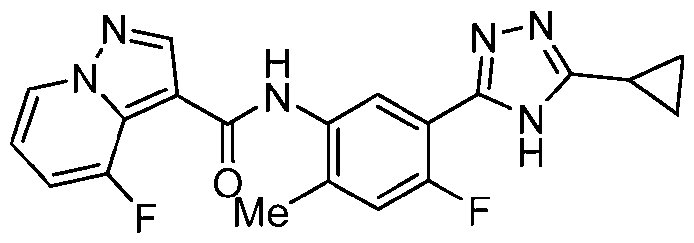
- Example 19 N-[5-(5-Cyclopropyl-4H-1,2,4-triazol-3-yl)-4-fluoro-2-methylphenyl]-6- fluoropyrazolo[1,5-a]pyridine-3-carboxamide
- the title compound was prepared from triazolo[4,5-b]pyridin-3-yl 6-fluoropyrazolo[1,5- a]pyridine-3-carboxylate in a similar fashion to Ex. 12, which was prepared from 6- fluoropyrazolo[1,5-a]pyridine-3-carboxylic acid in a similar fashion to Ex.1, step a.
- Example 23 N-[5-(5-Cyclopropyl-4H-1,2,4-triazol-3-yl)-4-fluoro-2-methylphenyl]-5- methoxypyrazolo[1,5-a]pyridine-3-carboxamide
- the title compound was prepared from triazolo[4,5-b]pyridin-3-yl 5- methoxypyrazolo[1,5-a]pyridine-3-carboxylate in a similar fashion to Ex.12, which was prepared from 5-methoxypyrazolo[1,5-a]pyridine-3-carboxylic acid in a similar fashion to Ex.1, step a.
- Step a To a solution of ethyl O-(2-mesitylenesulfonyl)acethydroxamate (2.00 g, 7.0 mmol) in dioxane (8 mL) was added 70% perchloric acid (1.2 mL) dropwise at 0 °C. The reaction was stirred at rt for 2 h, then the mixture was poured into ice/water (100 mL). The solution was stirred for 10 min until white solid precipitated.
- Step b To a mixture of O-(mesitylenesulfonyl)hydroxylamine (1.08 g, 5.0 mmol) and 3-ethylpyridine (536 mg, 5.0 mmol) was added DCM (20 mL), then the solution was stirred at rt for 36 h.
- Step c To a solution of 2-mesitylenesulfonate-1-amino-3-ethyl-pyridinium (5.0 mmol) in DMF (10 mL) at 0 °C, was added potassium carbonate (1.04 g, 7.5 mmol) and methyl propiolate (631 mg, 7.5 mmol). The reaction was stirred at rt for 18 h.
- Step d To a solution of methyl 6-ethylpyrazolo[1,5-a]pyridine-3-carboxylate (79 mg, 0.383 mmol) in 1:1:1 THF/MeOH/water (1.2 mL) at 0 °C, was added LiOH . H 2 O (81 mg, 1.92 mmol). The reaction was stirred at 50 °C for 2 h. Upon cooling to rt, the mixture was concentrated to remove THF and MeOH. The resultant mixture was adjusted to pH ⁇ 2 by addition of 2 M HCl (aq) . The mixture was diluted with EtOAc (10 mL) and washed with water (5 mL).
- Step e The desired product was prepared in a similar manner to Example 1, step a.
- Step f The desired product was prepared in a similar manner to Example 12, step c.
- Example 25 6-Cyclopropyl-N-[5-(5-cyclopropyl-4H-1,2,4-triazol-3-yl)-4-fluoro-2- methylphenyl]pyrazolo[1,5-a]pyridine-3-carboxamide [0247] The title compound was prepared from 3-cyclopropylpyridine in a similar fashion to Ex. 24.
- Step a To a mixture of 6-bromopyrazolo[1,5-a]pyridine (985 mg, 5.0 mmol), ethanol (0.58 mL, 10.0 mmol), palladium acetate (57 mg, 0.25 mmol), tBuBrettPhos (243 mg, 0.5 mmol) and cesium carbonate (3.26 g, 10.0 mmol) was added degassed toluene (10 mL).
- Step b To a solution of 6-ethoxypyrazolo[l,5-a]pyridine (747 mg, 4.6 mmol) in DMF (9.2 mL) at 0 °C, was added POCl 3 (1.29 mL, 13.8 mmol). The reaction was stirred at rt for 2 h.
- Step c To a solution of 6-ethoxypyrazolo[1,5-a]pyridine-3-carboxaldehyde (700 mg, 3.68 mmol) in water (20 mL), was added NaH2PO4 (1.33 g, 11.04 mmol). After 5 min, 1:1 THF/tBuOH (40 mL), 2-methyl-2-butene (2.34 mL, 22.08 mmol) and sodium chlorite (1.34 g, 14.72 mmol) were added sequentially. The reaction was stirred at rt for 24 h. The mixture was diluted with EtOAc (100 mL) and washed with water (50 mL) and brine (20 mL).
- Step d The desired product was prepared in a similar manner to Example 1, step a.
- Step e The desired product was prepared in a similar manner to Example 12, step c.
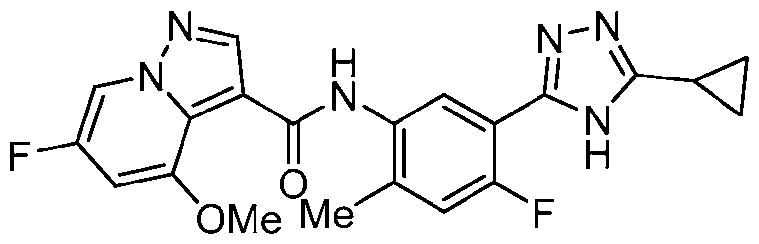
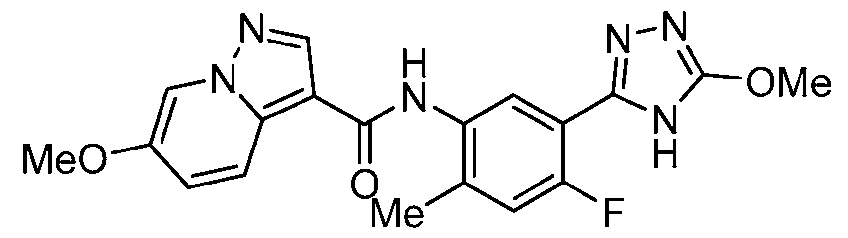
- Example 28 N-[4-Fluoro-2-methyl-5-(5-methyl-4H-1,2,4-triazol-3-yl)phenyl]-6- methoxypyrazolo[1,5-a]pyridine-3-carboxamide
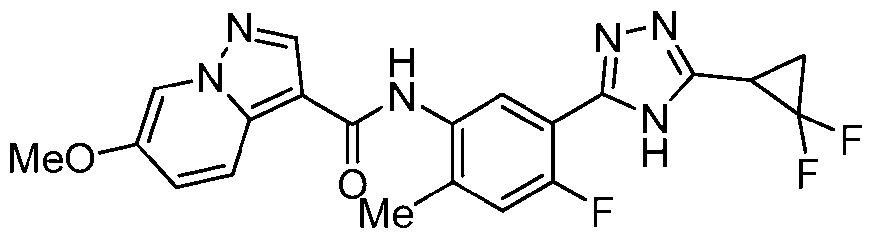
- the title compound was prepared in a similar fashion to Ex. 27 from triazolo[4,5- b]pyridin-3-yl 6-methoxypyrazolo[1,5-a]pyridine-3-carboxylate, which was prepared from 6- methoxypyrazolo[1,5-a]pyridine-3-carboxylic acid in a similar fashion to Ex.1, step a.
- Example 29 6-Fluoro-N-[4-fluoro-2-methyl-5-[5-(oxolan-2-yl)-4H-1,2,4-triazol-3- yl]phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide
- the title compound was prepared in a similar fashion to Ex. 12 from tetrahydro-2- furancarboximidamide and triazolo[4,5-b]pyridin-3-yl 6-fluoropyrazolo[1,5-a]pyridine-3- carboxylate, which was prepared from 6-fluoropyrazolo[1,5-a]pyridine-3-carboxylic acid in a similar fashion to Ex.1, step a.
- Example 32 N-[4-Fluoro-2-methyl-5-(5-propyl-4H-1,2,4-triazol-3-yl)phenyl]pyrazolo[1,5- a]pyridine-3-carboxamide [0258] The title compound was prepared from butyramidine hydrochloride in a similar fashion to Ex.12.
- Example 34 N-[5-[5-(Difluoromethyl)-4H-1,2,4-triazol-3-yl]-4-fluoro-2-methylphenyl]-6- methoxypyrazolo[1,5-a]pyridine-3-carboxamide [0260]
- the title compound was prepared from triazolo[4,5-b]pyridin-3-yl 6- methoxypyrazolo[1,5-a]pyridine-3-carboxylate (which was prepared from 6- methoxypyrazolo[1,5-a]pyridine-3-carboxylic acid in a similar fashion to Ex.1 step a) and 5-[5- (difluoromethyl)-4H-1,2,4-triazol-3-yl]-4-fluoro-2-methylaniline in a similar fashion to Ex.33.
- Example 35 N-[5-[5-(Difluoromethyl)-4H-1,2,4-triazol-3-yl]-4-fluoro-2-methylphenyl]-6- fluoropyrazolo[1,5-a]pyridine-3-carboxamide [0261]
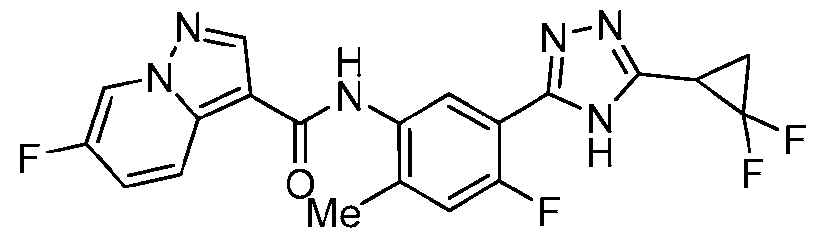
- the title compound was prepared from triazolo[4,5-b]pyridin-3-yl 6-fluoropyrazolo[1,5- a]pyridine-3-carboxylate (which was prepared from 6-fluoropyrazolo[1,5-a]pyridine-3-carboxylic acid in a similar fashion to Ex.
- Example 40 6-Chloro-N-[5-(5-ethyl-4H-1,2,4-triazol-3-yl)-4-fluoro-2- methylphenyl]pyrazolo[1,5-a]pyridine-3-carboxamide
- the title compound was prepared from triazolo[4,5-b]pyridin-3-yl 6-chloropyrazolo[1,5- a]pyridine-3-carboxylate (which was prepared from 6-chloropyrazolo[1,5-a]pyridine-3- carboxylic acid in a similar fashion to Ex. 1, step a) and propionimidamide hydrochloride in a similar fashion to Ex.12.
- Example 41 N-[5-(5-Ethyl-4H-1,2,4-triazol-3-yl)-4-fluoro-2-methylphenyl]-6- fluoropyrazolo[1,5-a]pyridine-3-carboxamide [0267]
- the title compound was prepared from triazolo[4,5-b]pyridin-3-yl 6-fluoropyrazolo[1,5- a]pyridine-3-carboxylate (which was prepared from 6-fluoropyrazolo[1,5-a]pyridine-3-carboxylic acid in a similar fashion to Ex.1, step a) and propionimidamide hydrochloride in a similar fashion to Ex.12.
- Example 42 N-[5-(5-Ethyl-4H-1,2,4-triazol-3-yl)-4-fluoro-2-methylphenyl]-6- methoxypyrazolo[1,5-a]pyridine-3-carboxamide
- the title compound was prepared from triazolo[4,5-b]pyridin-3-yl 6- methoxypyrazolo[1,5-a]pyridine-3-carboxylate (which was prepared from - methoxypyrazolo[1,5-a]pyridine-3-carboxylic acid in a similar fashion to Ex. 1, step a) and propionimidamide hydrochloride in a similar fashion to Ex.12.
- Example 43 6-Fluoro-N-[4-fluoro-2-methyl-5-(5-propan-2-yl-4H-1,2,4-triazol-3- yl)phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide
- the title compound was prepared in a similar fashion to Ex.12 using isobutyrimidamide hydrochloride and triazolo[4,5-b]pyridin-3-yl 6-fluoropyrazolo[1,5-a]pyridine-3-carboxylate, which was prepared from 6-fluoropyrazolo[1,5-a]pyridine-3-carboxylic acid in a similar fashion to Ex.1, step a.
- Example 45 6-Chloro-N-[4-fluoro-2-methyl-5-(5-propan-2-yl-4H-1,2,4-triazol-3- yl)phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide
- the title compound was prepared in a similar fashion to Ex.12 using isobutyrimidamide hydrochloride and triazolo[4,5-b]pyridin-3-yl 6-chloropyrazolo[1,5-a]pyridine-3-carboxylate, which was prepared from 6-chloropyrazolo[1,5-a]pyridine-3-carboxylic acid in a similar fashion to Ex.1, step a.
- Step a A round-bottom flask was charged with methyl 4-chloro-2-fluoro-5- nitrobenzoate (1.00 g, 4.28 mmol) and Fe (500 mg, 8.95 mmol, 2.1 equiv.) in AcOH (5 mL). The resulting mixture was then stirred at reflux under a nitrogen atmosphere for 2 h. Upon complete conversion, as judged by LCMS analysis, the reaction mixture was cooled to rt, diluted with EtOAc, filtered, and concentrated in vacuo. The residue was diluted with EtOAc and sat. aq. NaHCO3. The organic phase was separated, dried, and concentrated in vacuo.
- Steps b-d The title compound was prepared from methyl 5-amino-4-chloro-2- fluorobenzoate obtained in step a in a similar fashion to Ex.12.
- Step a A round-bottom flask was charged with methyl 5-amino-4-bromo-2- chlorobenzoate (1.0 g, 3.78 mmol, 1.0 equiv.), trimethylboroxine (0.58 mL, 4.15 mmol, 1.1 equiv.) and PdCl 2 (dppf) (277 mg, 0.378 mmol, 10 mol% equiv.) in 2 M Na 2 CO 3 (6 mL) and dioxane (18 mL).
- Steps b-d The title compound was prepared from methyl 5-amino-2-chloro-4- methylbenzoate obtained in step a in a similar fashion to Ex.12.
- Example 52 N-[5-(5-Cyclopropyl-4H-1,2,4-triazol-3-yl)-4-fluoro-2-methylphenyl]-4- fluoropyrazolo[1,5-a]pyridine-3-carboxamide [0281] The title compound was prepared from 4-fluoropyrazolo[1,5-a]pyridine-3-carboxylic acid in a similar fashion to Ex.12.
- Example 53 N-[2-Chloro-5-(5-cyclopropyl-4H-1,2,4-triazol-3-yl)-4- methylphenyl]pyrazolo[1,5-a]pyridine-3-carboxamide 109 [0282] The title compound was prepared from methyl 5-amino-2-bromo-4-chlorobenzoate in a similar fashion to Ex.50.
- Example 54 N-[2-Chloro-5-(5-cyclopropyl-4H-1,2,4-triazol-3-yl)-4-methylphenyl]-6- fluoropyrazolo[1,5-a]pyridine-3-carboxamide [0283] The title compound was prepared from methyl 5-amino-2-bromo-4-chlorobenzoate and 6-fluoropyrazolo[1,5-a]pyridine-3-carboxylic acid in a similar fashion to Ex. 50.
- Step a Methyl 6-bromopyrazolo[1,5-a]pyridine-3-carboxylate (510 mg, 2 mmol, 1.0 equiv.), RuPhos Pd G4 (340 mg, 0.4 mmol, 20 mol%) and Cs2CO3 (1.95 g, 6 mmol, 3.0 equiv.) were placed in a pressure vial. The vial was placed under N 2 , then dioxane (10 mL) and morpholine (348 mg, 4 mmol, 2.0 equiv.) were added. The reaction mixture was degassed again and stirred at 100 °C for 14 h.
- Step b A solution of the product from step a (450 mg, 1.72 mmol, 1.0 equiv.) in 2:1:1 THF:MeOH:H2O (20 mL) was treated with LiOH ⁇ H2O (206 mg, 8.62 mmol, 5.0 equiv). The thick mixture was allowed to stir at rt for 14 h. The resulting solution was diluted with water (6 mL) and treated with 1N aq. HCl (5 mL) dropwise over a few min. The mixture was allowed to stir for several additional min. to break up any clumps.
- Step a A mixture of methyl 6-bromopyrazolo[1,5-a]pyridine-3-carboxylate compound (510 mg, 2.0 mmol), B 2 pin 2 (762 mg, 1.5 mmol), Pd(dppf)Cl 2 (73 mg, 0.1 mmol), and KOAc (392 mg, 4 mmol) was placed under N 2 . Degassed dioxane (20 mL) was added and the reaction mixture was stirred at 100 °C for 4 h. The mixture was cooled to rt, concentrated, diluted with CH2Cl2 (50 mL), filtered through Celite® to remove solids, and again concentrated to afford the desired intermediate, which was used directly in part 2 of step a without further purification.
- Step b Ester hydrolysis was carried out similar to Ex.58, step b.
- Steps c-d The title compound was prepared in a similar fashion to Ex.24, steps e-f.
- Example 62 N-[5-[5-(2,2-Difluorocyclopropyl)-4H-1,2,4-triazol-3-yl]-4-fluoro-2- methylphenyl]-6-fluoropyrazolo[1,5-a]pyridine-3-carboxamide [0295] The title compound was prepared from 2,2-difluorocyclopropane-1-carboximidamide and triazolo[4,5-b]pyridin-3-yl 6-fluoropyrazolo[1,5-a]pyridine-3-carboxylate in a similar fashion to Ex.12.
- Example 63 N-[5-[5-(3,3-Difluorocyclobutyl)-4H-1,2,4-triazol-3-yl]-4-fluoro-2- methylphenyl]-6-fluoropyrazolo[1,5-a]pyridine-3-carboxamide [0296] The title compound was prepared from 3,3-difluorocyclobutane-1-carboximidamide and triazolo[4,5-b]pyridin-3-yl 6-fluoropyrazolo[1,5-a]pyridine-3-carboxylate in a similar fashion to Ex.12.
- Example 64 N-[5-[5-(3,3-Difluorocyclopentyl)-4H-1,2,4-triazol-3-yl]-4-fluoro-2- methylphenyl]-6-fluoropyrazolo[1,5-a]pyridine-3-carboxamide [0297] The title compound was prepared from 3,3-difluorocyclopentane-1-carboximidamide and triazolo[4,5-b]pyridin-3-yl 6-fluoropyrazolo[1,5-a]pyridine-3-carboxylate in a similar fashion to Ex.12.
- Example 66 4-Fluoro-N-[4-fluoro-2-methyl-5-(5-propan-2-yl-4H-1,2,4-triazol-3- yl)phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide [0299] The title compound was prepared from 2-methylpropionamidine and triazolo[4,5- b]pyridin-3-yl 4-fluoropyrazolo[1,5-a]pyridine-3-carboxylate in a similar fashion to Ex. 12.
- Example 68 N-[5-(5-Chloro-4H-1,2,4-triazol-3-yl)-4-fluoro-2-methylphenyl]pyrazolo[1,5- a]pyridine-3-carboxamide [0301] The title compound was prepared from N-[4-fluoro-2-methyl-5-(4,4,5,5-tetramethyl- 1,3,2-dioxaborolan-2-yl)phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide and 3-bromo-5-chloro- 4H-1,2,4-triazole in a similar fashion to Ex. 1 and Ex. 67.
- Example 69 N-[5-(5-Bromo-4H-1,2,4-triazol-3-yl)-4-fluoro-2-methylphenyl]pyrazolo[1,5- a]pyridine-3-carboxamide [0302] The title compound was prepared from N-[4-fluoro-2-methyl-5-(4,4,5,5-tetramethyl- 1,3,2-dioxaborolan-2-yl)phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide and 3,5-dibromo-4- (oxan-2-yl)-1,2,4-triazole in a similar fashion to Ex.1 and Ex.67 and followed by removal of the THP protecting group.
- Example 70 N-[4-Fluoro-5-[5-(methoxymethyl)-4H-1,2,4-triazol-3-yl]-2- methylphenyl]pyrazolo[1,5-a]pyridine-3-carboxamide [0303] The title compound was prepared from 2-methoxyethanimidamide in a similar fashion to Ex.12.
- Example 71 N-[5-[5-[2-(Dimethylamino)-2-oxoethyl]-4H-1,2,4-triazol-3-yl]-4-fluoro-2- methylphenyl]pyrazolo[1,5-a]pyridine-3-carboxamide [0304]
- the title compound was prepared from 2-cyano-N,N-dimethylacetamide in a similar fashion to Ex.15.
- Example 73 N-[4-Fluoro-5-[5-(4-fluorophenyl)-4H-1,2,4-triazol-3-yl]-2- methylphenyl]pyrazolo[1,5-a]pyridine-3-carboxamide [0306] The title compound was prepared from 4-fluorobenzenecarboximidamide in a similar fashion to Ex.12.
- Example 74 N-[4-Fluoro-2-methyl-5-(5-pyridin-3-yl-4H-1,2,4-triazol-3- yl)phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide [0307] The title compound was prepared from pyridine-3-carboximidamide in a similar fashion to Ex.12.
- Example 77 N-[5-[5-(Dimethylamino)-4H-1,2,4-triazol-3-yl]-4-fluoro-2-methylphenyl]-5- fluoropyrazolo[1,5-a]pyridine-3-carboxamide [0310] The title compound was prepared in a similar fashion to Ex.
- Example 78 N-[5-[5-(Cyclopropylamino)-4H-1,2,4-triazol-3-yl]-4-fluoro-2- methylphenyl]pyrazolo[1,5-a]pyridine-3-carboxamide [0311] The title compound was prepared from 1-cyclopropylguanidine in a similar fashion to Ex.12.
- Example 79 N-[4-Fluoro-2-methyl-5-(5-pyrrolidin-1-yl-4H-1,2,4-triazol-3- yl)phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide [0312] The title compound was prepared from N-[4-fluoro-2-methyl-5-(4,4,5,5-tetramethyl- 1,3,2-dioxaborolan-2-yl)phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide and 3-bromo-5- pyrrolidin-1-yl-4H-1,2,4-triazole in a similar fashion to Ex. 1 and Ex. 67.
- Example 80 N-[4-Fluoro-2-methyl-5-(5-piperazin-1-yl-4H-1,2,4-triazol-3- yl)phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide [0313] The title compound was prepared from N-[4-fluoro-2-methyl-5-(4,4,5,5-tetramethyl- 1,3,2-dioxaborolan-2-yl)phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide and 1-(5-bromo-4H- 1,2,4-triazol-3-yl)piperazine in a similar fashion to Ex. 1 and Ex. 67.
- Example 81 N-[5-[5-(Cyclopropylmethyl)-4H-1,2,4-triazol-3-yl]-4-fluoro-2-methylphenyl]- 6-methoxypyrazolo[1,5-a]pyridine-3-carboxamide [0314] The title compound was prepared from triazolo[4,5-b]pyridin-3-yl 6- methoxypyrazolo[1,5-a]pyridine-3-carboxylate in a similar fashion to Ex.15.
- Example 83 N-[4-Fluoro-2-methyl-5-(5-methyl-4H-1,2,4-triazol-3-yl)phenyl]-6- methylpyrazolo[1,5-a]pyridine-3-carboxamide [0316] The title compound was prepared from ethanimidamide and triazolo[4,5-b]pyridin-3-yl 6-methylpyrazolo[1,5-a]pyridine-3-carboxylate in a similar fashion to Ex.12.
- Example 84 6-Ethyl-N-[4-fluoro-5-(5-methoxy-4H-1,2,4-triazol-3-yl)-2- methylphenyl]pyrazolo[1,5-a]pyridine-3-carboxamide
- the title compound was prepared in a similar fashion to Ex.1 from 3-bromo-5-methoxy- 4H-1,2,4-triazole and 6-ethyl-N-[4-fluoro-2-methyl-5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2- yl)phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide, which was accessed from 6-ethylpyrazolo[1,5- a]pyridine-3-carboxylic acid and 5-bromo-4-fluoro-2-methylaniline in a similar fashion to step b of Ex.1.
- Example 85 5-Fluoro-N-[4-fluoro-5-(5-methoxy-4H-1,2,4-triazol-3-yl)-2- methylphenyl]pyrazolo[1,5-a]pyridine-3-carboxamide
- the title compound was prepared from 5-fluoro-N-[4-fluoro-2-methyl-5-(4,4,5,5- tetramethyl-1,3,2-dioxaborolan-2-yl)phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide and 3- bromo-5-methoxy-4H-1,2,4-triazole in a similar fashion to Ex.1 and Ex.77.
- Example 86 N-[4-Fluoro-5-(5-methoxy-4H-1,2,4-triazol-3-yl)-2-methylphenyl]-6- methoxypyrazolo[1,5-a]pyridine-3-carboxamide
- the title compound was prepared in a similar fashion to Ex.1 from 3-bromo-5-methoxy- 4H-1,2,4-triazole and N-[4-fluoro-2-methyl-5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2- yl)phenyl]-6-methoxypyrazolo[1,5-a]pyridine-3-carboxamide, which was accessed from 6- methoxypyrazolo[1,5-a]pyridine-3-carboxylic acid and 5-bromo-4-fluoro-2-methylaniline in a similar fashion to step b, Ex.1.
- Example 87 6-Fluoro-N-[4-fluoro-2-methyl-5-[5-(oxolan-3-yl)-4H-1,2,4-triazol-3- yl]phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide
- the title compound was prepared from oxolane-3-carboximidamide and triazolo[4,5- b]pyridin-3-yl 6-fluoropyrazolo[1,5-a]pyridine-3-carboxylate in a similar fashion to Ex. 12.
- Example 88 N-[5-(5-Cyclopropyl-2-methyl-1,2,4-triazol-3-yl)-4-fluoro-2- methylphenyl]pyrazolo[1,5-a]pyridine-3-carboxamide [0321] The title compound was prepared from 5-bromo-3-cyclopropyl-1-methyl-1,2,4-triazole and N-[4-fluoro-2-methyl-5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)phenyl]pyrazolo[1,5- a]pyridine-3-carboxamide in a similar fashion to Ex.1 and Ex.67.
- Step a A vial was charged with N-[4-fluoro-2-methyl-5-(4,4,5,5-tetramethyl-1,3,2- dioxaborolan-2-yl)phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide (494 mg, 1.25 mmol, 1.0 equiv.), 4-benzyl-3-bromo-5-fluoro-1,2,4-triazole (318 mg, 1.25 mmol, 1.0 equiv.), Pd XPhos G3 (105 mg, 0.12 mmol, 10 mol%), 2M Na2CO3 (aq.
- Step b A vial was charged with the product from step a (75 mg, 0.17 mmol, 1.0 equiv.), NaOEt (68 mg, 1.02 mmol, 6.0 equiv.) and EtOH (1 mL). The reaction mixture was stirred at 80 °C for approximately 16 h, at which time LCMS analysis indicated 70% consumption of starting material.
- Step c A reactor jar was charged with the product from step b (65 mg, 0.13 mmol, 1.0 equiv.) and Pd/C (13 mg, 20 wt%) in MeOH (10 mL) containing a few drops of 4N HCl in dioxane. The reaction mixture was shaken under 30 psi of hydrogen gas for 3 h, then filtered over Celite® and concentrated under reduced pressure to afford the crude product. Purification by reverse phase HPLC afforded the title compound.
- Example 90 N-[5-(1-Benzyl-5-fluoro-1,2,4-triazol-3-yl)-4-fluoro-2- methylphenyl]pyrazolo[1,5-a]pyridine-3-carboxamide [0325] The title compound was prepared from N-[4-fluoro-2-methyl-5-(4,4,5,5-tetramethyl- 1,3,2-dioxaborolan-2-yl)phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide and 1-benzyl-3-bromo- 5-fluoro-1,2,4-triazole in a similar fashion to Ex.1 and Ex.67.
- Step a A vial was charged with methyl 2-amino-4-bromo-5-fluorobenzoate (1.5 g, 6 mmol, 1.0 equiv.) and THF (10 mL). To the reaction mixture was added LiBH4 in THF (2M, 6 mL, 12 mmol, 2.0 equiv.).
- reaction mixture was stirred at 50 °C for 2 h, at which time LCMS analysis indicated complete consumption of starting material.
- the reaction mixture was cooled to rt, diluted with water and stirred for 30 min., during which time a few drops of conc. HCl were added.
- the mixture was diluted with EtOAc.
- the organic phase was separated, washed with brine (2x), dried over Na 2 SO 4 and concentrated in vacuo.
- the crude product was purified by column chromatography (0% to 90% EtOAc in hexanes) to provide the desired (2-amino-4-bromo-5- fluorophenyl) methanol.
- Step b A vial was charged with product from step a (1.3 g, 5.9 mmol, 1.0 equiv.), imidazole (1.0 g, 14.7 mmol, 2.5 equiv.) and DMF (15 mL). TBS-Cl (979 mg, 6.5 mmol, 1.1 equiv.) was added to the mixture. The reaction mixture was stirred for 5 h at 23 °C, at which time LCMS analysis indicated complete consumption of starting material. The reaction mixture was diluted with EtOAc, the layers were separated, and the organic layer was washed successively with sat. aq. NH4Cl (2x) and brine (2x).
- Step c A mixture of the product from step b (2.0 g, 5.9 mmol, 1.0 equiv.), and triazolo[4,5-b]pyridin-3-yl pyrazolo[1,5-a]pyridine-3-carboxylate (1.7 g, 5.9 mmol, 1.0 equiv.) in NMP (15.0 mL) was stirred at 120 °C for 1 h. Upon complete conversion, as judged by LCMS analysis, the reaction mixture was cooled to rt and diluted with water. The resulting precipitated solid was collected by vacuum filtration, rinsed with water, and dried in vacuo.
- Step d A vial was charged with the product from step c (1.0 g, 2.0 mmol, 1 equiv.), 4,4,4',4',5,5,5',5'-octamethyl-2,2'-bi(l,3,2-dioxaborolane) (615 mg, 2.4 mmol, 1.2 equiv.), KOAc (600 mg, 6.1 mmol, 3 equiv.) and 1,4-dioxane (15 mL). The reaction mixture was degassed with N 2 for 30 min. before Pd(dppf)C1 2 (74 mg, 0.1 mmol, 5 mmol%) was added.
- reaction mixture was then heated at 100 °C for 12 h, after which time LCMS analysis showed complete conversion of starting material.
- the reaction mixture was cooled to rt and diluted with EtOAc.
- the organic layer was filtered over Celite® and the filtrate was concentrated in vacuo.
- the crude product was purified by column chromatography (0% to 60% EtOAc in hexanes) to provide the desired N-[2- [[tert-butyl(dimethyl)silyl]oxymethyl]-4-fluoro-5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2- yl)phenyl]-6-fluoropyrazolo[1,5-a]pyridine-3-carboxamide.
- Step e A vial was charged with the product from step d (150 mg, 0.27 mmol, 1.0 equiv.), 3-bromo-5-cyclopropyl-4H-1,2,4-triazole (104 mg, 0.55 mmol, 2.0 equiv.), XPhos Pd G3 (47 mg, 0.055 mmol, 20 mol%), K 3 CO 3 (114 mg, 0.83 mmol, 3.0 equiv.), 1,4-dioxane (2.0 mL) and water (0.2 mL). The reaction mixture was degassed with N 2 and then stirred at 100 °C for 16 h, at which time LCMS analysis indicated complete consumption of starting material.
- Step f A vial was charged with the product from step e (145 mg, 0.276 mmol, 1.0 equiv.) and THF (2 mL). The mixture was cooled to 0 °C and TBAF (1M soln. in THF, 0.83 mL, 0.83 mmol, 3.0 equiv.) was added. The reaction mixture was allowed to warm to rt and stirred for 8 h until complete consumption of starting material was determined by LCMS analysis. The reaction mixture was diluted with water and EtOAc, the layers were separated, and the organic layer was washed with brine (2x). The combined organic layers were collected, dried over Na2SO4 and concentrated in vacuo.
- Step a To a solution of methyl 5-bromo-1H-1,2,4-triazole-3-carboxylate (2.96 g, 14.4 mmol, 1.0 equiv.) and pTsOH ⁇ H2O (547 mg, 2.88 mmol, 20 mol%) in THF (21.8 mL, 0.7 M) under N2 at rt was added dihydropyran (3.94 mL, 43.2 mmol, 3.0 equiv.) portion-wise over 5 min. The resulting mixture was heated at reflux for 6 h, then cooled to rt and concentrated under reduced pressure. The resulting residue was treated with sat. aq.
- Step b A vial was charged with one of the regioisomeric products from step a (211.7 mg, 0.730 mmol, 1.0 equiv.) in THF (2.0 mL, 0.36 M) and placed under N 2 .
- Step c A vial was charged successively with the crude product obtained in step b (51.9 mg, 0.180 mmol, 1.0 equiv.), 6-fluoro-N-[4-fluoro-2-methyl-5-(4,4,5,5-tetramethyl-1,3,2- dioxaborolan-2-yl)phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide (74.4 mg, 0.180 mmol, 1.0 equiv., prepared from 6-fluoropyrazolo[1,5-a]pyridine-3-carboxylic acid and 5-bromo-4-fluoro-2- methylaniline according to Ex.1, steps a-b), Pd XPhos G3 (15.2 mg, 0.018 mmol, 10 mol%), 1M aq.
- Step d The product obtained in step c was taken up in 4N HCl in dioxane (1.5 mL) and the mixture was stirred at rt for 14 h.
- Step b A vial was charged with the product from step a (60 mg, 0.117 mmol, 1.0 equiv.), 4,4,5,5-tetramethyl-2-[1-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)cyclopropyl]-1,3,2- dioxaborolane (51 mg, 0.17 mmol, 1.5 equiv.), Pd XPhos G3 (10 mg, 0.01 mmol, 10 mol%), Cs2CO3 (76 mg, 0.23 mmol, 2.0 equiv.), dioxane (2.0 mL) and water (0.5 mL).
- Step c To a solution of the crude product from step b (0.117 mmol, 1.0 equiv.) in 10% aq. NaOH (1 mL) at 0 °C was added H 2 O 2 (30 wt% in water, 0.16 mL, 28 equiv.) dropwise.
- reaction mixture was allowed to stir for 0.5 h, then the reaction was quenched by addition of sat. aq. Na2S2O3.
- the mixture was diluted with EtOAc and water.
- the organic layer was separated, dried over Na 2 SO 4 and concentrated in vacuo.
- the crude product was purified by column chromatography (0% to 100% EtOAc in hexanes) to provide 6-fluoro-N-[4-fluoro-5-[5-(1- hydroxycyclopropyl)-4-(oxan-2-yl)-1,2,4-triazol-3-yl]-2-methylphenyl]pyrazolo[1,5-a]pyridine- 3-carboxamide, which was treated with 4N HCl in dioxane (2 mL) and stirred overnight to afford the title compound.
- Example 94 N-[5-(5-Cyclopropyl-1,3,4-oxadiazol-2-yl)-4-fluoro-2- methylphenyl]pyrazolo[1,5-a]pyridine-3-carboxamide [0339] A vial was charged with triazolo[4,5-b]pyridin-3-yl pyrazolo[1,5-a]pyridine-3- carboxylate (22.5 mg, 0.080 mmol, 1.0 equiv.), 5-(5-cyclopropyl-1,3,4-oxadiazol-2-yl)-4-fluoro- 2-methylaniline (18.7 mg, 0.080 mmol, 1.0 equiv.), which was obtained as a minor byproduct in the synthesis of Ex.12 (step b), and NMP (0.2 mL, 0.5 M).
- Example 96 N-[5-[1-(2,2-Difluoroethyl)triazol-4-yl]-4-fluoro-2-methylphenyl]pyrazolo[1,5- a]pyridine-3-carboxamide [0341] The title compound was prepared from 4-bromo-1-(2,2-difluoroethyl)triazole and N-[4- fluoro-2-methyl-5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)phenyl]pyrazolo[1,5-a]pyridine- 3-carboxamide in a similar fashion to Ex.1 and Ex.67.
- Example 99 N-[5-[1-(2,2-Difluoroethyl)triazol-4-yl]-4-fluoro-2-methylphenyl]-6- fluoropyrazolo[1,5-a]pyridine-3-carboxamide [0344]
- the title compound was prepared from 4-bromo-1-(2,2-difluoroethyl)triazole and 6- fluoro-N-[4-fluoro-2-methyl-5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2- yl)phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide in a similar fashion to Ex. 1 and Ex. 76.
- Example 100 N-[4-Fluoro-5-[1-(4-fluorophenyl)-1,2,4-triazol-3-yl]-2- methylphenyl]pyrazolo[1,5-a]pyridine-3-carboxamide [0345] The title compound was prepared from 3-bromo-1-(4-fluorophenyl)-1H-1,2,4-triazole and N-[4-fluoro-2-methyl-5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)phenyl]pyrazolo[1,5- a]pyridine-3-carboxamide in a similar fashion to Ex.1 and Ex.67.
- Example 102 N-[4-Fluoro-5-[1-(4-fluorophenyl)-1,2,4-triazol-3-yl]-2-methylphenyl]-6- methoxypyrazolo[1,5-a]pyridine-3-carboxamide [0347] The title compound was prepared from 3-bromo-1-(4-fluorophenyl)-1H-1,2,4-triazole and N-[4-fluoro-2-methyl-5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)phenyl]-6- methoxypyrazolo[1,5-a]pyridine-3-carboxamide in a similar fashion to Ex.1 and Ex.86.
- Example 103 N-[5-(2-Cyclopropyl-1,3-thiazol-4-yl)-4-fluoro-2-methylphenyl]-6- fluoropyrazolo[1,5-a]pyridine-3-carboxamide [0348] The title compound was prepared from 4-bromo-2-cyclopropyl-1,3-thiazole and 6- fluoro-N-[4-fluoro-2-methyl-5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2- yl)phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide in a similar fashion to Ex. 1 and Ex. 76.
- Example 104 N-[5-(5-Cyclopropyl-1,2,4-thiadiazol-3-yl)-4-fluoro-2-methylphenyl]-6- fluoropyrazolo[1,5-a]pyridine-3-carboxamide [0349] The title compound was prepared from 3-bromo-5-cyclopropyl-1,2,4-thiadiazole and 6- fluoro-N-[4-fluoro-2-methyl-5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2- yl)phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide in a similar fashion to Ex.
- Example 105 N-[5-(5-Cyclopropyl-1,3,4-thiadiazol-2-yl)-4-fluoro-2-methylphenyl]-6- fluoropyrazolo[1,5-a]pyridine-3-carboxamide [0350] The title compound was prepared from 2-bromo-5-cyclopropyl-1,3,4-thiadiazole and 6- fluoro-N-[4-fluoro-2-methyl-5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2- yl)phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide in a similar fashion to Ex.
- Example 106 N-[5-(3-Cyclopropyl-1,2,4-thiadiazol-5-yl)-4-fluoro-2-methylphenyl]-6- fluoropyrazolo[1,5-a]pyridine-3-carboxamide [0351] The title compound was prepared from 5-bromo-3-cyclopropyl-1,2,4-thiadiazole and 6- fluoro-N-[4-fluoro-2-methyl-5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2- yl)phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide in a similar fashion to Ex.
- Example 108 N-[5-(4-Cyclopropyl-1H-imidazol-2-yl)-4-fluoro-2- methylphenyl]pyrazolo[1,5-a]pyridine-3-carboxamide [0353] The title compound was prepared from 2-bromo-4-cyclopropyl-1H-imidazole and N-[4- fluoro-2-methyl-5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)phenyl]pyrazolo[1,5-a]pyridine- 3-carboxamide in a similar fashion to Ex.1 and Ex.67.
- Example 110 N-[5-(1-Cyclopropylimidazol-4-yl)-4-fluoro-2-methylphenyl]pyrazolo[1,5- a]pyridine-3-carboxamide [0355] The title compound was prepared from 4-bromo-1-cyclopropylimidazole and N-[4- fluoro-2-methyl-5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)phenyl]pyrazolo[1,5-a]pyridine- 3-carboxamide in a similar fashion to Ex.1 and Ex.67.
- Example 111 N-[4-Fluoro-2-methyl-5-(3-methyl-1,2,4-oxadiazol-5-yl)phenyl]pyrazolo[1,5- a]pyridine-3-carboxamide [0356] The title compound was prepared from 5-bromo-3-methyl-1,2,4-oxadiazole and N-[4- fluoro-2-methyl-5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)phenyl]pyrazolo[1,5-a]pyridine- 3-carboxamide e in a similar fashion to Ex.1 and Ex.67.
- Example 112 N-[5-[1-(2,2-Difluoroethyl)pyrazol-4-yl]-4-fluoro-2-methylphenyl]-6- methylpyrazolo[1,5-a]pyridine-3-carboxamide
- Step a A 20 mL vial was charged with 5-bromo-4-fluoro-2-methylaniline (0.395 g, 1.938 mmol, 1.0 equiv.), 1-(2,2-difluoroethyl)-4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2- yl)pyrazole (0.5 g, 1.938 mmol, 1.0 equiv.), Pd(dppf)Cl 2 (0.141 g, 0.193 mmol, 10 mol%), and Na2CO3 (2.91 mL, 2M aq. solution, 3.0 equiv.) in of 1,4-dioxane (6 mL).
- Step b A mixture of 5-[1-(2,2-difluoroethyl)pyrazol-4-yl]-4-fluoro-2-methylaniline (0.090 g, 0.306 mmol, 1.0 equiv.) and triazolo[4,5-b]pyridin-3-yl 6-methylpyrazolo[1,5- a]pyridine-3-carboxylate (0.078 g, 0.306 mmol, 1.0 equiv., prepared according to Ex.1, step a) in NMP (2.0 mL) was stirred at 120 °C for 6 h.
- NMP 2.0 mL
- Example 120 6-Chloro-N-[5-[1-(2,2-difluoroethyl)pyrazol-4-yl]-2,4- dimethylphenyl]pyrazolo[1,5-a]pyridine-3-carboxamide [0366] The title compound was prepared from 5-bromo-2,4-dimethylaniline and triazolo[4,5- b]pyridin-3-yl 6-chloropyrazolo[1,5-a]pyridine-3-carboxylate in a similar fashion to Ex.112.
- Example 121 N-[5-[1-(2,2-Difluoroethyl)pyrazol-4-yl]-2,4-dimethylphenyl]-6- fluoropyrazolo[1,5-a]pyridine-3-carboxamide [0367] The title compound was prepared from 5-bromo-2,4-dimethylaniline and triazolo[4,5- b]pyridin-3-yl 6-fluoropyrazolo[1,5-a]pyridine-3-carboxylate in a similar fashion to Ex.112.
- Example 122 N-[5-[1-(2,2-Difluoroethyl)pyrazol-4-yl]-2,4-dimethylphenyl]-6- methoxypyrazolo[1,5-a]pyridine-3-carboxamide [0368] The title compound was prepared from 5-bromo-2,4-dimethylaniline and triazolo[4,5- b]pyridin-3-yl 6-methoxypyrazolo[1,5-a]pyridine-3-carboxylate in a similar fashion to Ex. 112.
- Step a A vial was charged with 5-bromo-4-chloro-2-methylaniline (340 mg, 1.55 mmol, 1.0 equiv.), 1-(2,2-difluoroethyl)-4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)pyrazole (400 mg, 1.55 mmol, 1.0 equiv.), Pd(dppf)Cl 2 (117 mg, 0.16 mmol, 10 mol%), Na 2 CO 3 (328 mg, 3.10 mmol, 2.0 equiv.), di
- reaction mixture was degassed with N2, and it was stirred at 95 °C for 2 h, at which time LCMS analysis indicated complete consumption of starting material.
- the reaction mixture was cooled to rt and diluted with EtOAc.
- the organic layer was separated, filtered over Celite®, and concentrated in vacuo.
- the crude product was purified by column chromatography (0% to 60% EtOAc in hexanes) to provide the desired 4-chloro-5-[1-(2,2-difluoroethyl)pyrazol-4-yl]-2-methylaniline.
- Step b A vial was charged with product from step a (100 mg, 0.37 mmol, 1.0 equiv.), cyclopropylboronic acid (47 mg, 0.55 mmol, 1.5 equiv.), PCy3 Pd G2 (22 mg, 0.037 mmol, 10 mol%), K3PO4 (234 mg, 1.10 mmol, 3.0 equiv.), toluene (2.0 mL) and water (0.2 mL). The reaction mixture was degassed with N 2, and it was stirred at 100 °C for 3 h, at which time LCMS analysis indicated partial consumption of starting material. The reaction mixture was cooled to rt and diluted with EtOAc.
- Step c A mixture of 4-cyclopropyl-5-[1-(2,2-difluoroethyl)pyrazol-4-yl]-2- methylaniline (28 mg, 0.10 mmol, 1.0 equiv.), and triazolo[4,5-b]pyridin-3-yl pyrazolo[1,5- a]pyridine-3-carboxylate (28 mg, 0.10 mmol, 1.0 equiv.) in NMP (1.0 mL) was stirred at 120 °C for 1 h. Upon complete conversion, as judged by LCMS analysis, the reaction mixture was cooled to rt and diluted with water.
- Example 125 N-[5-[1-(2,2-Difluoroethyl)-1,2,4-triazol-3-yl]-4-fluoro-2- methylphenyl]pyrazolo[1,5-a]pyridine-3-carboxamide [0373] The title compound was prepared from 1-(2,2-difluoroethyl)-3-(4,4,5,5-tetramethyl- 1,3,2-dioxaborolan-2-yl)-1,2,4-triazole and triazolo[4,5-b]pyridin-3-yl pyrazolo[1,5-a]pyridine- 3-carboxylate in a similar fashion to Ex.112.
- Example 126 N-[5-[1-(2,2-Difluoroethyl)-1,2,4-triazol-3-yl]-2,4-dimethylphenyl]-6- methoxypyrazolo[1,5-a]pyridine-3-carboxamide [0374] The title compound was prepared from 1-(2,2-difluoroethyl)-3-(4,4,5,5-tetramethyl- 1,3,2-dioxaborolan-2-yl)-1,2,4-triazole, 5-bromo-2,4-dimethylaniline, and triazolo[4,5-b]pyridin- 3-yl 6-methoxypyrazolo[1,5-a]pyridine-3-carboxylate in a similar fashion to Ex.
- Example 127 N-[5-[1-(2,2-Difluoroethyl)-1,2,4-triazol-3-yl]-4-fluoro-2-methylphenyl]-6- methylpyrazolo[1,5-a]pyridine-3-carboxamide [0375]
- the title compound was prepared from 1-(2,2-difluoroethyl)-3-(4,4,5,5-tetramethyl- 1,3,2-dioxaborolan-2-yl)-1,2,4-triazole in a similar fashion to Ex. 112.
- Example 128 N-[5-[1-(2,2-Difluoroethyl)-1,2,4-triazol-3-yl]-2,4-dimethylphenyl]-6- methylpyrazolo[1,5-a]pyridine-3-carboxamide [0376]
- the title compound was prepared from 1-(2,2-difluoroethyl)-3-(4,4,5,5-tetramethyl- 1,3,2-dioxaborolan-2-yl)-1,2,4-triazole and 5-bromo-2,4-dimethylaniline in a similar fashion to Ex.112.
- Example 129 N-[5-(1-Cyclopropylpyrazol-4-yl)-4-fluoro-2-methylphenyl]pyrazolo[1,5- a]pyridine-3-carboxamide [0377] The title compound was prepared from 1-cyclopropyl-4-(4,4,5,5-tetramethyl-1,3,2- dioxaborolan-2-yl)pyrazole and triazolo[4,5-b]pyridin-3-yl pyrazolo[1,5-a]pyridine-3- carboxylate.
- Example 132 N-[5-[1-(Cyclopropylmethyl)pyrazol-4-yl]-4-fluoro-2- methylphenyl]pyrazolo[1,5-a]pyridine-3-carboxamide [0380] The title compound was prepared from 1-(cyclopropylmethyl)-4-(4,4,5,5-tetramethyl- 1,3,2-dioxaborolan-2-yl)pyrazole and triazolo[4,5-b]pyridin-3-yl pyrazolo[1,5-a]pyridine-3- carboxylate in a similar fashion to Ex.112.
- Example 133 N-[5-[1-(Cyclopropylmethyl)pyrazol-4-yl]-2,4-difluorophenyl]pyrazolo[1,5- a]pyridine-3-carboxamide [0381] The title compound was prepared from 1-(cyclopropylmethyl)-4-(4,4,5,5-tetramethyl- 1,3,2-dioxaborolan-2-yl)pyrazole, 5-bromo-2,4-difluoroaniline, and triazolo[4,5-b]pyridin-3-yl pyrazolo[1,5-a]pyridine-3-carboxylate in a similar fashion to Ex.
- Example 134 N-[4-Chloro-5-[1-(cyclopropylmethyl)pyrazol-4-yl]-2- methylphenyl]pyrazolo[1,5-a]pyridine-3-carboxamide [0382] The title compound was prepared from 1-(cyclopropylmethyl)-4-(4,4,5,5-tetramethyl- 1,3,2-dioxaborolan-2-yl)-1H-pyrazole, 5-bromo-4-chloro-2-methylaniline, and triazolo[4,5- b]pyridin-3-yl pyrazolo[1,5-a]pyridine-3-carboxylate in a similar fashion to Ex. 112.
- Example 135 N-[2-Chloro-5-[1-(cyclopropylmethyl)pyrazol-4-yl]-4- fluorophenyl]pyrazolo[1,5-a]pyridine-3-carboxamide [0383] The title compound was prepared from 1-(cyclopropylmethyl)-4-(4,4,5,5-tetramethyl- 1,3,2-dioxaborolan-2-yl)-1H-pyrazole, 5-bromo-2-chloro-4-fluoroaniline, and triazolo[4,5- b]pyridin-3-yl pyrazolo[1,5-a]pyridine-3-carboxylate in a similar fashion to Ex.
- Example 136 N-[5-[1-(Cyclopropylmethyl)pyrazol-4-yl]-2,4-dimethylphenyl]pyrazolo[1,5- a]pyridine-3-carboxamide [0384] The title compound was prepared from 1-(cyclopropylmethyl)-4-(4,4,5,5-tetramethyl- 1,3,2-dioxaborolan-2-yl)-1H-pyrazole, 5-bromo-2,4-dimethylaniline, and triazolo[4,5-b]pyridin- 3-yl pyrazolo[1,5-a]pyridine-3-carboxylate in a similar fashion to Ex.
- Example 138 N-[4-Fluoro-2-methyl-5-(1-propan-2-ylpyrazol-4-yl)phenyl]pyrazolo[1,5- a]pyridine-3-carboxamide [0386] The title compound was prepared from 1-propan-2-yl-4-(4,4,5,5-tetramethyl-1,3,2- dioxaborolan-2-yl)pyrazole and triazolo[4,5-b]pyridin-3-yl pyrazolo[1,5-a]pyridine-3- carboxylate in a similar fashion to Ex.112.
- Example 139 N-[4-Fluoro-2-methyl-5-[1-(trifluoromethyl)pyrazol-4- yl]phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide [0387] The title compound was prepared from 4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)- 1-(trifluoromethyl)pyrazole and triazolo[4,5-b]pyridin-3-yl pyrazolo[1,5-a]pyridine-3- carboxylate in a similar fashion to Ex.112.
- Example 140 N-[4-Fluoro-2-methyl-5-[1-(oxan-4-yl)pyrazol-4-yl]phenyl]pyrazolo[1,5- a]pyridine-3-carboxamide [0388] The title compound was prepared from 1-(oxan-4-yl)-4-(4,4,5,5-tetramethyl-1,3,2- dioxaborolan-2-yl)pyrazole and triazolo[4,5-b]pyridin-3-yl pyrazolo[1,5-a]pyridine-3- carboxylate in a similar fashion to Ex.112.
- Example 141 N-[4-Fluoro-2-methyl-5-[1-(2,2,2-trifluoroethyl)pyrazol-4- yl]phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide [0389] The title compound was prepared from 4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)- 1-(2,2,2-trifluoroethyl)pyrazole and triazolo[4,5-b]pyridin-3-yl pyrazolo[1,5-a]pyridine-3- carboxylate in a similar fashion to Ex.112.
- Example 142 N-[4-Fluoro-2-methyl-5-[1-(3,3,3-trifluoropropyl)pyrazol-4- yl]phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide [0390] The title compound was prepared from 4-bromo-1-(3,3,3-trifluoropropyl)pyrazole and N-[4-fluoro-2-methyl-5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)phenyl]pyrazolo[1,5- a]pyridine-3-carboxamide in a similar fashion to Ex.1 and Ex.67.
- Step a A 40 mL vial was charged with 1-ethenyl-4-iodopyrazole (500 mg, 2.27 mmol, 1.0 equiv.), NaI (2.1 g, 13.9 mmol, 6.1 equiv.), and MeCN (15 mL, 0.15 M) under an N2 atmosphere. Me 3 SiCF 3 (2.2 mL, 14.9 mmol, 6.6 equiv.) was added over 45 min to the reaction mixture at 80 °C, and the reaction mixture was stirred at 80 °C for an additional 3 h. Upon complete conversion, as judged by LCMS analysis, the reaction mixture was cooled to rt.
- Step b The crude intermediate obtained in step a was converted to the title compound in a similar fashion to Ex.1.
- Example 144 N-[5-[1-(2,2-Difluorocyclopropyl)pyrazol-4-yl]-4-fluoro-2-methylphenyl]-6- fluoropyrazolo[1,5-a]pyridine-3-carboxamide [0393] The title compound was prepared from 6-fluoro-N-[4-fluoro-2-methyl-5-(4,4,5,5- tetramethyl-1,3,2-dioxaborolan-2-yl)phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide in a similar fashion to Ex. 143.
- Step a N-[4-Fluoro-5-[1-(3-hydroxycyclobutyl)pyrazol-4-yl]-2- methylphenyl]pyrazolo[1,5-a]pyridine-3-carboxamide
- Step a A round-bottom flask was charged with 4-bromo-1H-pyrazole (2.00 g, 13.6 mmol, 1.0 equiv.), 3-bromocyclobutan-1-one (2.60 g, 17.7 mmol, 1.3 equiv.), K 2 CO 3 (2.60 g, 17.7 mmol, 1.30 equiv.), and DMF (39 mL, 0.35 M), and the resulting mixture was stirred at 25 °C for 18 h.
- Step b A 40 mL vial was charged with the crude product obtained in step a (2.7 g) and MeOH (14 mL). NaBH4 (0.573 g, 15.1 mmol) was added to the mixture in small portions at 0 °C, and the resulting mixture was allowed to warm to 25 °C and stir for 4 h.
- Step c The crude intermediate obtained in step b was converted to the title compound in a similar fashion to Ex.1.
- Example 149 N-[4-Chloro-5-[1-(4-fluorophenyl)pyrazol-4-yl]-2-methylphenyl]-6- methoxypyrazolo[1,5-a]pyridine-3-carboxamide [0400]
- the title compound was prepared from [1-(4-fluorophenyl)-1H-pyrazol-4-yl]boronic acid, 5-bromo-4-chloro-2-methylaniline, and triazolo[4,5-b]pyridin-3-yl 6-methoxypyrazolo[1,5- a]pyridine-3-carboxylate in a similar fashion to Ex.112.
- Example 150 N-[4-Fluoro-2-methyl-5-(1-methylpyrazol-3-yl)phenyl]pyrazolo[1,5- a]pyridine-3-carboxamide [0401] The title compound was prepared from 1-methyl-3-(4,4,5,5-tetramethyl-1,3,2- dioxaborolan-2-yl)pyrazole and triazolo[4,5-b]pyridin-3-yl pyrazolo[1,5-a]pyridine-3- carboxylate in a similar fashion to Ex.112.
- Example 151 N-[5-(5-Cyclopropyl-1H-pyrazol-3-yl)-4-fluoro-2-methylphenyl]pyrazolo[1,5- a]pyridine-3-carboxamide [0402] The title compound was prepared from 5-cyclopropyl-3-iodo-1H-pyrazole and N-[4- fluoro-2-methyl-5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)phenyl]pyrazolo[1,5-a]pyridine- 3-carboxamide in a similar fashion to Ex.1 and Ex.67.
- Example 153 N-[5-(5-Cyclopropyl-1H-pyrazol-3-yl)-4-fluoro-2-methylphenyl]-6- methoxypyrazolo[1,5-a]pyridine-3-carboxamide [0404] The title compound was prepared from 5-cyclopropyl-3-iodo-1H-pyrazole and N-[4- fluoro-2-methyl-5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)phenyl]-6-methoxypyrazolo[1,5- a]pyridine-3-carboxamide in a similar fashion to Ex.1 and Ex.86.
- Step a A mixture of triazolo[4,5-b]pyridin-3-yl pyrazolo[1,5-a]pyridine-3-carboxylate (500 mg, 1.78 mmol, 1.0 equiv.), and 5-amino-2-fluoro-4-methylbenzonitrile (268 mg, 1.78 mmol, 1.0 equiv.) in NMP (6.0 mL) was stirred at 120 °C for 6 h. The reaction mixture was cooled to rt and diluted with water.
- Step b A vial was charged with product from step a (495 mg, 1.68 mmol, 1.0 equiv.), sodium azide (219 mg, 3.36 mmol, 2.0 equiv.), ammonium chloride (180 mg, 3.36 mmol, 2.0 equiv.) and DMF (8 mL). The reaction mixture was stirred at 110 °C for 16 h, at which time LCMS analysis indicated full consumption of starting material.
- Step c To a solution of product from step b (75 mg, 0.22 mmol, 1.0 equiv.) and K 2 CO 3 (60 mg, 0.44 mmol, 2.0 equiv.) in DMF (1 mL) was added bromomethylcyclopropane (59 mg, 0.44 mmol, 2.0 equiv.).
- Example 156 6-Fluoro-N-[4-fluoro-2-methyl-5-(2-methyltetrazol-5-yl)phenyl]pyrazolo[1,5- a]pyridine-3-carboxamide
- the title compound was prepared from triazolo[4,5-b]pyridin-3-yl 6-fluoropyrazolo[1,5- a]pyridine-3-carboxylate and iodomethane in a similar fashion to Ex. 154.
- Example 157 6-Fluoro-N-[4-fluoro-2-methyl-5-(1-methyltetrazol-5-yl)phenyl]pyrazolo[1,5- a]pyridine-3-carboxamide
- the title compound was obtained as a minor byproduct in the synthesis of Ex.157, which was prepared from triazolo[4,5-b]pyridin-3-yl 6-fluoropyrazolo[1,5-a]pyridine-3-carboxylate and iodomethane in a similar fashion to Ex.154.
- Example 158 N-[5-(2-Ethyltetrazol-5-yl)-4-fluoro-2-methylphenyl]pyrazolo[1,5-a]pyridine- 3-carboxamide [0411] The title compound was prepared from bromoethane in a similar fashion to Ex.154.
- Example 161 N-[5-(6-cyclopropylpyridazin-3-yl)-4-fluoro-2-methylphenyl]pyrazolo[1,5- a]pyridine-3-carboxamide [0414] The title compound was prepared from N-[4-fluoro-2-methyl-5-(4,4,5,5-tetramethyl- 1,3,2-dioxaborolan-2-yl)phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide and 3-bromo-6- cyclopropylpyridazine in a similar fashion to Ex. 89 (step a).
- Example 162 N-[5-(2-cyclopropylpyrimidin-5-yl)-4-fluoro-2-methylphenyl]pyrazolo[1,5- a]pyridine-3-carboxamide [0415] The title compound was prepared from N-[4-fluoro-2-methyl-5-(4,4,5,5-tetramethyl- 1,3,2-dioxaborolan-2-yl)phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide and 5-bromo-2- cyclopropylpyrimidine in a similar fashion to Ex. 89 (step a).
- Example 163 N-[5-(5-cyclopropylpyrazin-2-yl)-4-fluoro-2-methylphenyl]pyrazolo[1,5- a]pyridine-3-carboxamide [0416] The title compound was prepared from N-[4-fluoro-2-methyl-5-(4,4,5,5-tetramethyl- 1,3,2-dioxaborolan-2-yl)phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide and 2-bromo-5- cyclopropylpyrazine in a similar fashion to Ex.89 (step a).
- Example 164 N-[5-(2-cyclopropyloxypyrimidin-5-yl)-4-fluoro-2-methylphenyl]-6- fluoropyrazolo[1,5-a]pyridine-3-carboxamide [0417] The title compound was prepared from 6-fluoro-N-[4-fluoro-2-methyl-5-(4,4,5,5- tetramethyl-1,3,2-dioxaborolan-2-yl)phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide and 5- bromo-2-cyclopropyloxypyrimidine in a similar fashion to Ex.92 (step c).
- Example 165 6-fluoro-N-[4-fluoro-2-methyl-5-[2-[[(3S)-oxolan-3-yl]amino]pyrimidin-5- yl]phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide
- Step a A round-bottom flask was charged with 5-bromo-2-chloropyrimidine (1.93 g, 10.0 mmol, 1.0 equiv.), (3S)-oxolan-3-amine hydrochloride (2.47 g, 20.0 mmol, 2.0 equiv.), K 2 CO 3 (5.53 g, 40.0 mmol, 40.0 equiv.), and DMF (20 mL, 0.5 M), and the resulting mixture was stirred at 70 °C for 12 h.
- Step b A vial was charged successively with the crude product obtained in step a (55 mg, 0.225 mmol, 1.5 equiv.), 6-fluoro-N-[4-fluoro-2-methyl-5-(4,4,5,5-tetramethyl-1,3,2- dioxaborolan-2-yl)phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide (62 mg, 0.15 mmol, 1.0 equiv., prepared from 6-fluoropyrazolo[1,5-a]pyridine-3-carboxylic acid and 5-bromo-4-fluoro-2- methylaniline according to Ex.1, steps a-b), Pd(dppf)Cl2 (11 mg, 0.015 mmol, 10 mol%), 2M aq.
- Example 166 N-[5-[2-(2,2-difluoroethylamino)pyrimidin-5-yl]-4-fluoro-2-methylphenyl]-6- fluoropyrazolo[1,5-a]pyridine-3-carboxamide [0420] The title compound was prepared from 6-fluoro-N-[4-fluoro-2-methyl-5-(4,4,5,5- tetramethyl-1,3,2-dioxaborolan-2-yl)phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide and 5- bromo-N-(2,2-difluoroethyl)pyrimidin-2-amine (prepared from 5-bromo-2-chloropyrimidine and 2,2-difluoroethanamine hydrochloride according to Ex.165, step a) in a similar fashion to Ex.165.
- Example 167 6-chloro-N-[5-[1-(2,2-difluoroethyl)triazol-4-yl]-4-fluoro-2- methylphenyl]pyrazolo[1,5-a]pyridine-3-carboxamide [0421] The title compound was prepared from 6-fluoro-N-[4-fluoro-2-methyl-5-(4,4,5,5- tetramethyl-1,3,2-dioxaborolan-2-yl)phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide and 5- bromo-N-(cyclopropylmethyl)pyrimidin-2-amine (prepared from 5-bromo-2-chloropyrimidine and cyclopropylmethanamine according to Ex.
- Example 168 6-fluoro-N-[4-fluoro-2-methyl-5-[6-[[(3S)-oxolan-3-yl]amino]pyridazin-3- yl]phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide
- the title compound was prepared from 6-fluoro-N-[4-fluoro-2-methyl-5-(4,4,5,5- tetramethyl-1,3,2-dioxaborolan-2-yl)phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide and 6- bromo-N-[(3S)-oxolan-3-yl]pyridazin-3-amine (prepared from 3-bromo-6-chloropyridazine and (3S)-oxolan-3-amine hydrochloride according to Ex.165,
- Example 170 N-[5-(6-cyclopropyloxypyridazin-3-yl)-4-fluoro-2-methylphenyl]-6- fluoropyrazolo[1,5-a]pyridine-3-carboxamide [0424] The title compound was prepared from 6-fluoro-N-[4-fluoro-2-methyl-5-(4,4,5,5- tetramethyl-1,3,2-dioxaborolan-2-yl)phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide and 3- bromo-6-cyclopropyloxypyridazine (prepared from 3-bromo-6-fluoropyridazine and cyclopropanol according to Ex.165, step a) in a similar fashion to Ex.165.
- Step a A 40 mL vial was charged with 5-bromo-2-chloro-4-fluoropyridine (250 mg, 0.95 mmol., 1.0 equiv.), cyclopropyl zinc bromide (443 mg, 1.9 mmol., 2 equiv.), Pd(PPh 3 ) 4 (59 mg, 0.05 mmol., 5 mol%) and THF (6.0 mL). The resulting mixture was stirred at 50 °C for 5 h. Upon complete conversion, as judged by LCMS analysis, the reaction mixture was cooled to rt and filtered over Celite® and concentrated in vacuo.
- Step b A vial was charged successively with the product obtained in step a, 5-bromo-2- cyclopropyl-4-fluoropyridine (78 mg, 0.363 mmol., 1.0 equiv.), 6-fluoro-N-[4-fluoro-2-methyl-5- (4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide (100 mg, 0.242 mmol, 1.0 equiv., prepared from 6-fluoropyrazolo[1,5-a]pyridine-3-carboxylic acid and 5-bromo-4-fluoro-2-methylaniline according to Ex.
- Example 172 N-[2,4-dimethyl-5-(6-propan-2-yloxypyridazin-3-yl)phenyl]-6- fluoropyrazolo[1,5-a]pyridine-3-carboxamide [0427] The title compound was prepared from 6-fluoro-N-[4-fluoro-2-methyl-5-(4,4,5,5- tetramethyl-1,3,2-dioxaborolan-2-yl)phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide and 3- bromo-6-propan-2-yloxypyridazine in a similar fashion to Ex.
- Example 173 N-[5-(5-difluoromethoxy-2-pyridyl)-4-fluoro-2-tolyl]-1,7a-diaza-3- indenecarboxamide [0428] The title compound was prepared from 6-fluoro-N-[4-fluoro-2-methyl-5-(4,4,5,5- tetramethyl-1,3,2-dioxaborolan-2-yl)phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide and 2- bromo-5-(difluoromethoxy)pyridine according to Ex.165 (step a), in a similar fashion to Ex.165.
- Step a A solution of cyclopropanol (280 mg, 4.8 mmol, 1.5 equiv.) in DMF (5.0 mL) was added NatBuO (470 mg, 4.8 mmol, 1.5 equiv.).
- Step b A mixture of product from step a with KOH (10.0 equiv.) in tBuOH (10mL) and water (2mL) was stirred at 105 °C for 48h, then cooled to room temperature. The reaction mixture was acidified with 1N HCl to PH ⁇ 3 and then extracted with EtOAc. The organic layer was separated, dried over Na2SO4 and concentrated in vacuo. The crude product was used directly in the next step without further purification.
- Step c The title compound was prepared from 5-cyclopropyloxypyridine-2-carboxylic acid and 6-N-(5-amino-4-fluoro-2-methylphenyl)-6-fluoropyrazolo[1,5-a]pyridine-3- carboxamide in a similar fashion to Ex. 176.
- Example 175 6-fluoro-N-[4-fluoro-2-methyl-5-[[5-[[(3S)-oxolan-3-yl]amino]pyridine-2- carbonyl]amino]phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide [0432] The title compound was prepared from 5-[[(3S)-oxolan-3-yl]amino]pyridine-2- carboxylic acid and 6-N-(5-amino-4-fluoro-2-methylphenyl)-6-fluoropyrazolo[1,5-a]pyridine-3- carboxamide in a similar fashion to Ex.176.
- Step a A mixture of triazolo[4,5-b]pyridin-3-yl 6-fluoropyrazolo[1,5-a]pyridine-3- carboxylate (298 mg, 1.0 mmol, 1.0 equiv.) and 5-bromo-4-fluoro-2-methylaniline (204 mg, 1.0 mmol, 1.0 equiv.) in NMP (6.0 mL) was stirred at 120 °C for 4 h. The reaction mixture was cooled to rt and diluted with water.
- Step b A vial was charged with product from step a (260 mg, 0.72 mmol, 1.0 equiv.), diphenylmethanimine (155 mg, 0.85 mmol, 1.2 equiv.), Pd 2 (dba) 3 (66 mg, 0.072 mmol, 0.1 equiv.), XantPhos (84 mg, 0.44 mmol, 0.2 equiv.), Cs 2 CO 3 (705 mg, 2.16 mmol, 3.0 equiv.) and dioxane (8 mL). The reaction mixture was stirred at 100 °C for 16 h under N2, at which time LCMS analysis indicated full consumption of starting material.
- Step c To a mixture of product from step b (220 mg, 0.47 mmol, 1.0 equiv.), NaOAc (97 mg, 1.18 mmol, 2.5 equiv.) in MeOH (5 mL) was added NH2OH HCl (66 mg, 0.94 mmol, 2.0 equiv.).
- Step d The desired product was prepared from 5-fluoropyridine-2-carboxylic acid in a similar manner to Example 1, step a.
- Example 177 6-fluoro-N-[4-fluoro-2-methyl-5-[(5-methylpyridine-2- carbonyl)amino]phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide
- the title compound was prepared from 5-methylpyridine-2-carboxylic acid and 6-N-(5- amino-4-fluoro-2-methylphenyl)-6-fluoropyrazolo[1,5-a]pyridine-3-carboxamide in a similar fashion to Ex.176.
- Example 178 6-fluoro-N-[4-fluoro-2-methyl-5-(pyridine-3- carbonylamino)phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide
- the title compound was prepared from pyridine-2-carboxylic acid and 6-N-(5-amino-4- fluoro-2-methylphenyl)-6-fluoropyrazolo[1,5-a]pyridine-3-carboxamide in a similar fashion to Ex.176.
- Example 180 N-[5-[(2,2-difluorocyclopropanecarbonyl)amino]-4-fluoro-2-methylphenyl]- 6-fluoropyrazolo[1,5-a]pyridine-3-carboxamide [0440] The title compound was prepared from 2,2-difluorocyclopropane-1-carboxylic acid and 6-N-(5-amino-4-fluoro-2-methylphenyl)-6-fluoropyrazolo[1,5-a]pyridine-3-carboxamide in a similar fashion to Ex. 176.
- Example 181 N-[5-(cyclopropanecarbonylamino)-4-fluoro-2-methylphenyl]-6- fluoropyrazolo[1,5-a]pyridine-3-carboxamide [0441] The title compound was prepared from cyclopropanecarboxylic acid and 6-N-(5-amino- 4-fluoro-2-methylphenyl)-6-fluoropyrazolo[1,5-a]pyridine-3-carboxamide in a similar fashion to Ex.176.
- Activity was determined as a function of phosphorylated biotinylated TK peptide generated by the transfer of phosphate from ATP as measured by the use of the HTRF KinEASE-TK assay kit (Cisbio, Product #: 62TK0PEJ).
- Levels of phosphorylated biotinylated TK peptide (Part 61TK0BLC of KinEASE assay kit) are quantified by its capture by phosphor-TK-Antibody-Cryptate (Part of KinEASE assay kit) and XL665-labeled Streptavidin (Part 610SAXLG of KinEASE assay kit) followed by measurement of Time-Resolved Fluorescence Resonance Energy Transfer (TR- FRET) signal.
- TR- FRET Time-Resolved Fluorescence Resonance Energy Transfer
- Enzyme solution was prepared for each of the tyrosine kinases in 50 mM HEPES, pH 7.4, 5 mM MgCl2 for c-Kit or 10 mM MgCl2 for FLT3, CSF1R, PDGFR ⁇ and PDGFR ⁇ , 2 mM MnCl2, 0.01% Brij-35 and 0.01% BSA.
- M07e cells (DSMZ, catalog # ACC 104) were serum starved. Cells were centrifuged and cell pellet resuspended in OptiMEM (Gibco, catalog # 31985062) to a density of 1x10 6 - 2.5x10 6 cells per mL. Cells were then incubated overnight at 5% CO2 and 37 °C in an appropriately sized flask. On the day of the experiment, an 11 point, half log titration of test compound was pre-dispensed into 96-well round bottom polypropylene plates (Corning, catalog # 3365).
- Cell pellets were resuspended with either OptiMEM or human serum (Innovative Research, catalog # ISERAB1000ML) to a cell density of 1x10 6 cells per mL. Cells were then added to the appropriate compound plate at 100,000 cells per well in 100 ⁇ L and mixed. Plates were incubated at 5% CO2 and 37 °C for 50 min. Stem Cell Factor, SCF (R&D Systems, #255-SC/CF) was diluted in OptiMEM to 1 ⁇ g/mL and added to all wells as 10 ⁇ L for a final concentration of 90.9 ng/mL. Plates were incubated at 5% CO2 and 37 °C for 10 min.
- SCF Stem Cell Factor
- Plates were centrifuged, supernatant removed by flicking, and cells resuspended in 50 ⁇ L primary antibody, anti-phospho- c-KIT (Tyr703) (Cell Signaling, catalog # 3073), diluted to 1 ⁇ g/mL with permeabilization buffer. Plates were incubated at rt for 1 h. Plates were centrifuged, supernatant removed, and cells resuspended with 200 ⁇ L DPBS + 0.5% BSA.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- General Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Pharmacology & Pharmacy (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
The present disclosure is directed to compounds that are inhibitors of KIT having a structure according to Formula (III), and compositions containing those compounds. Methods of using the compounds for the treatment of diseases, disorders, or conditions are also described.
Description
KIT INHIBITOR COMPOUNDS AND METHODS OF USE THEREOF CROSS-REFERENCE TO RELATED APPLICATIONS [0001] This application claims the benefit of priority to U.S. Provisional Patent Application No. 63/540,604, filed on September 26, 2023, the entire content of which is incorporated by reference herein. BACKGROUND [0002] The following discussion is provided to aid the reader in understanding the disclosure and is not admitted to describe or constitute prior art thereto. [0003] Mast cells contain granules containing proinflammatory and immunomodulatory mediators. Upon activation, degranulation occurs, releasing these proinflammatory and immunomodulatory mediators into the surrounding tissues, generally in response to a perceived pathogen (e.g., parasitic, bacterial and viral infections, allergens, toxins, etc.). The activation of mast cells serves to induce an immune response to protect the body from pathogens, and to aid in wound healing, and tissue repair. However, misfunctioning mast cells underlie the etiology of many allergic and chronic inflammatory diseases and are implicated in a broad spectrum of conditions. [0004] The activation of receptor tyrosine kinase KIT on mast cells by its ligand, Stem Cell Factor (SCF), is required for mast cell differentiation, maturation, and survival. For certain diseases, mast cell activation may play a central role in the onset and progression of the disease. Accordingly, inhibition of KIT may lead to mast cell depletion, and provide a promising therapeutic approach for mast cell-driven diseases. Thus, there is a need to develop inhibitors of KIT. SUMMARY [0005] In one aspect, the present disclosure relates to compounds that are inhibitors of the receptor tyrosine kinase KIT. The compounds are represented by Formula III and associated sub- formulas described herein:
. [0006] In another aspect, this disclosure is directed to methods of inhibiting KIT in a subject comprising administering to the subject an effective amount of a compound of Formula III or an associated sub-formula described herein. [0007] In another aspect, this disclosure is directed to methods of reducing the activity and/or quantity of systemic mast cells in a subject comprising administering to the subject an effective amount of a compound of Formula III or an associated sub-formula described herein. [0008] In yet another aspect, this disclosure provides methods for treating a disease, disorder, or condition mediated at least in part by KIT in a subject, comprising administering to the subject a therapeutically effective amount of a compound of Formula III or an associated sub-formula described herein. Diseases, disorders, and conditions mediated by KIT include e.g., an allergic disease, disorder, or condition; an inflammatory disease, disorder, or condition; a neuroinflammatory disease, disorder, or condition; a neurological disease, disorder, or condition; an immune related disease, disorder, or condition; an autoimmune related disease, disorder, or condition; a dermatological disease, disorder, or condition; a respiratory disease, disorder, or condition; a metabolic disease, disorder, or condition; a cardiovascular disease, disorder, or condition; a fibrotic disease, disorder, or condition; or cancer. [0009] Certain aspects of the present disclosure further comprise the administration of one or more additional therapeutic agents as set forth herein below. DETAILED DESCRIPTION OF THE DISCLOSURE [0010] Before the present disclosure is further described, it is to be understood that the disclosure is not limited to the particular embodiments set forth herein, and it is also to be understood that the terminology used herein is for the purpose of describing particular embodiments only, and is not intended to be limiting.
Definitions [0011] Unless otherwise defined, all terms of art, notations and other scientific terms or terminology used herein are intended to have the meanings commonly understood by those of skill in the art to which this disclosure pertains. [0012] The term “about” as used herein has its original meaning of approximately and is to provide literal support for the exact number that it precedes, as well as a number that is near to or approximately the number that the term precedes. In determining whether a number is near to or approximately a specifically recited number, the near or approximating unrecited number can be a number which, in the context in which it is presented, provides the substantial equivalent of the specifically recited number. For example, if the degree of approximation is not otherwise clear from the context, “about” means either within plus or minus 10% of the provided value, or rounded to the nearest significant figure, in all cases inclusive of the provided value. Where ranges are provided, they are inclusive of the boundary values. [0013] The term "alkyl", by itself or as part of another substituent, means, unless otherwise stated, a saturated hydrocarbon radical, having, in some embodiments, one to eight (e.g., C1-C8- alkyl), or one to six (e.g., C1-C6-alkyl), or one to four carbon atoms (e.g., C1-C4-alkyl), or one to three carbon atoms (e.g., C1-C3-alkyl), respectively. The term “alkyl” encompasses straight and branched-chain hydrocarbon groups. Examples of alkyl groups include, but are not limited to, methyl (Me, -CH3), ethyl (Et, -CH2CH3), n-propyl, isopropyl, n-butyl, t-butyl, isobutyl, sec-butyl, isopentyl, tert-pentyl, n-pentyl, isohexyl, n-hexyl, n-heptyl, 4-isopropylheptane, n-octyl, and the like. In some embodiments, the alkyl groups are C1-C4 alkyl groups (e.g., methyl, ethyl, n-propyl, isopropyl, n-butyl, isobutyl, sec-butyl, or t-butyl). [0014] The term “alkylene” refers to a straight or branched, saturated, hydrocarbon radical having, in some embodiments, one to six carbon atoms (e.g., C1-C6-alkylene), or one to four carbon atoms (e.g., C1-C4-alkylene), or one to three carbon atoms (e.g., C1-C3-alkylene) and linking at least two other groups, i.e., a divalent hydrocarbon radical. When two moieties are linked to the alkylene they can be linked to the same carbon atom (i.e., geminal), or different carbon atoms of the alkylene group. For instance, a straight chain alkylene can be the bivalent radical of -(CH2)n- , where n is 1, 2, 3, 4, 5 or 6 (i.e., a C1-C6-alkylene). Representative alkylene groups include, but are not limited to, methylene, ethylene, propylene, isopropylene, butylene, isobutylene,
secbutylene, pentylene, hexylene and the like. In some embodiments, the alkylene groups are C1- 3 alkylene groups (e.g., methylene, ethylene, or propylene). [0015] The term "cycloalkyl" refers to a monocyclic, bicyclic or polycyclic hydrocarbon ring system having, in some embodiments, 3 to 14 carbon atoms (e.g., C3-C14-cycloalkyl), or 3 to 10 carbon atoms (e.g., C3-C10-cycloalkyl), or 3 to 8 carbon atoms (e.g., C3-C8-cycloalkyl), or 3 to 6 carbon atoms (e.g., C3-C6-cycloalkyl) or 5 to 6 carbon atoms (e.g., C5-C6-cycloalkyl). Cycloalkyl groups can be saturated or characterized by one or more points of unsaturation (i.e., carbon-carbon double and/or triple bonds), provided that the points of unsaturation do not result in an aromatic system. Examples of monocyclic cycloalkyl groups include, but are not limited to, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, cyclohexenyl, cyclohexynyl, cycloheptyl, cycloheptenyl, cycloheptadienyl, cyclooctyl, cyclooctenyl, cyclooctadienyl and the like. The rings of bicyclic and polycyclic cycloalkyl groups can be fused, bridged, or spirocyclic. Non-limiting examples of bicyclic, spirocyclic and polycyclic hydrocarbon groups include bicyclo[1.1.1]pentane, bicyclo[2.1.1]hexane, bicyclo[2.2.1]heptane, bicyclo[2.2.2]octane, adamantyl, spiro[5.5]undecane, spiro[2.2]pentane, spiro[2.2]pentadiene, spiro[2.5]octane, spiro[2.2]pentadiene, and the like. In some embodiments, the cycloalkyl groups of the present disclosure are monocyclic C3-C6-cycloalkyl moieties (e.g., cyclopropyl, cyclobutyl, cyclopentyl, or cyclohexyl). [0016] As used herein, a wavy line, " ", that intersects a single, double or triple bond in any chemical structure depicted herein, represents that the point of attachment of the single, double, or triple bond to the remainder of the molecule is through either one of the atoms that make up the single, double or triple bond. Additionally, a bond extending from a substituent to the center of a ring (e.g., a phenyl ring or heteroaryl ring) is meant to indicate attachment of that substituent to the ring at any of the available ring vertices, i.e., such that attachment of the substituent to the ring results in a chemically stable arrangement. [0017] The terms "halogen," or “halo” are used interchangeably and refer to, unless otherwise stated, a fluorine, chlorine, bromine, or iodine atom. Additionally, terms such as "haloalkyl," refer to alkyl groups, as defined herein, that are substituted with one or more halogen(s) (e.g., 1-3 halogen(s)). For example, the term "C1-C6 haloalkyl" is meant to include trifluoromethyl, difluoromethyl, 2,2,2-trifluoroethyl, 4-chlorobutyl, 3-bromopropyl, and the like.
[0018] The term “hydroxyalkyl” refers to an alkyl group, as defined herein, that is substituted with one or more hydroxyl groups (e.g., 1-3 hydroxyl groups). Exemplary hydroxyalkyl groups include methanol, ethanol, 1,2-propanediol, 1,2-hexanediol, glycerol, and the like. [0019] The term “cyanoalkyl” refers to an alkyl group, as defined herein, that is substituted with one cyano group. Exemplary cyanoalkyl groups include cyanomethyl, cyanoethyl, cyanopropyl, and the like. [0020] As used herein, the term “alkoxy” refers to an alkyl group, as defined herein, that is attached to the remainder of the molecule via an oxygen atom (e.g., -O-C1-C12 alkyl, -O-C1-C8 alkyl, -O-C1-C6 alkyl, or -O-C1-C3 alkyl). Non-limiting examples of alkoxy groups include methoxy (OMe, -OCH3), ethoxy (OEt, -OCH2CH3), n-propoxy, iso-propoxy, n-butoxy, sec- butoxy, n-pentoxy, n-hexoxy, and the like. [0021] The term "heteroaryl" refers to monocyclic or fused bicyclic aromatic groups (or rings) having, in some embodiments, from 5 to 14 (i.e., 5- to 14-membered heteroaryl), or from 5 to 10 (i.e., 5- to 10-membered heteroaryl), or from 5 to 6 (i.e., 5- to 6-membered heteroaryl) members (i.e., ring vertices), and containing from one to five, one to four, one to three, one to two or one ring heteroatom independently selected from nitrogen (N), oxygen (O), and sulfur (S). A heteroaryl group can be attached to the remainder of the molecule through a carbon atom or a heteroatom of the heteroaryl group, when chemically permissible. Non-limiting examples of heteroaryl groups include pyridyl, pyridazinyl, pyrazinyl, pyrimidinyl, triazinyl, purinyl, thienopyridinyl, thienopyrimidinyl, pyrazolopyrimidinyl, imidazopyridines, isothiazolyl, pyrazolyl, indazolyl, pteridinyl, imidazolyl, triazolyl, tetrazolyl, oxazolyl, isoxazolyl, thiadiazolyl, pyrrolyl, thiazolyl, furyl, thienyl and the like. In some embodiments, the heteroaryl groups of the present disclosure are monocyclic 5- to 6-membered heteroaryl moieties having 1-4 ring nitrogen atoms (e.g., pyridinyl, pyrimidinyl, pyrazinyl, pyridazinyl, triazolyl, imidazolyl, pyrazolyl, tetrazolyl, and the like). In some embodiments, the heteroaryl groups of the present disclosure are 5-membered heteroaryl moieties having 1-4 ring heteroatoms independently selected from N, O, and S (e.g., imidazolyl, pyrazolyl, thiazolyl, oxazolyl, oxadiazole, thiadiazolyl, triazolyl, tetrazolyl, and the like). [0022] The term "heterocycloalkyl" refers to a non-aromatic monocyclic, bicyclic or polycyclic cycloalkyl ring having, in some embodiments, 3 to 14 members (e.g., 3- to 14-membered
heterocycle), or 3 to 10 members (e.g., 3- to 10-membered heterocycle), or 4 to 8 members (e.g., 4- to 8-membered heterocycle), or 4 to 6 members (e.g., 4- to 6-membered heterocycle), or 5 to 6 members (e.g., 5- to 6-membered heterocycle), and having from one to five, one to four, one to three, one to two or one ring heteroatom independently selected from nitrogen (N), oxygen (O), and sulfur (S). Heterocycloalkyl groups are saturated or characterized by one or more points of unsaturation (e.g., one or more carbon-carbon double bonds, carbon-carbon triple bonds, carbon- nitrogen double bonds, and/or nitrogen-nitrogen double bonds), provided that the points of unsaturation do not result in an aromatic system. The rings of bicyclic and polycyclic heterocycloalkyl groups can be fused, bridged, or spirocyclic. Non-limiting examples of heterocycloalkyl groups include aziridine, oxirane, thiirane, azetidine, oxetane, pyrrolidine, imidazolidine, pyrazolidine, dioxolane, phthalimide, piperidine, 1,4-dioxane, morpholine, thiomorpholine, thiomorpholine-S-oxide, thiomorpholine-S,S-oxide, piperazine, 3,4,5,6- tetrahydropyridazine, pyran, decahydroisoquinoline, 3-pyrroline, thiopyran, tetrahydrofuran, tetrahydrothiophene, quinuclidine, 2,6-diazaspiro[3.3]heptane, 2-azaspiro[3.3]heptane, 1- oxaspiro[3.3]heptane, 6-azaspiro[3.4]octane, and the like. A heterocycloalkyl group can be attached to the remainder of the molecule through a ring carbon atom, or a ring heteroatom, when chemically permissible. In some embodiments, the heterocycloalkyl groups of the present disclosure are monocyclic 4- to 8-membered heterocycloalkyl moieties having one or two heteroatoms independently selected from N, O, and S, (e.g., azetidine, oxetane, pyrrolidine, tetrahydrofuran, tetrahydropyran, piperidine, piperazine, morpholine, and the like). [0023] As referred to herein, "pharmaceutically acceptable salt" is meant to include salts of the compounds according to this disclosure that are prepared with suitably nontoxic acids or bases, depending on the particular substituents found on the compounds described herein. When compounds of the present disclosure contain suitably acidic functionalities, base addition salts can be obtained by contacting the neutral form of such compounds with a sufficient amount of the desired base, either neat or in a suitable inert solvent. Examples of salts derived from pharmaceutically-acceptable inorganic bases include aluminum, ammonium, calcium, copper, ferric, ferrous, lithium, magnesium, manganic, manganous, potassium, sodium, zinc and the like. Salts derived from pharmaceutically-acceptable organic bases include salts of primary, secondary and tertiary amines, including substituted amines, cyclic amines, naturally-occurring amines and the like, such as arginine, betaine, caffeine, choline, N,N’-dibenzylethylenediamine, diethylamine,
2-diethylaminoethanol, 2-dimethylaminoethanol, ethanolamine, ethylenediamine, N- ethylmorpholine, N-ethylpiperidine, glucamine, glucosamine, histidine, hydrabamine, isopropylamine, lysine, methylglucamine, morpholine, piperazine, piperidine, polyamine resins, procaine, purines, theobromine, triethylamine, trimethylamine, tripropylamine, tromethamine and the like. When compounds of the present disclosure contain suitably basic functionalities, acid addition salts can be obtained by contacting the neutral form of such compounds with a sufficient amount of the desired acid, either neat or in a suitable inert solvent. Examples of pharmaceutically acceptable acid addition salts include those derived from inorganic acids like hydrochloric, hydrobromic, nitric, carbonic, monohydrogencarbonic, phosphoric, monohydrogenphosphoric, dihydrogenphosphoric, sulfuric, monohydrogensulfuric, hydriodic, or phosphorous acids and the like, as well as the salts derived from suitably nontoxic organic acids like acetic, propionic, isobutyric, malonic, benzoic, succinic, suberic, fumaric, mandelic, phthalic, benzenesulfonic, p- tolylsulfonic, citric, tartaric, methanesulfonic, and the like. Also included are salts of amino acids such as arginate and the like, and salts of organic acids like glucuronic or galacturonic acids and the like (see, for example, Berge, S.M., et al, “Pharmaceutical Salts”, Journal of Pharmaceutical Science, 1977, 66, 1-19). Certain specific compounds of the present disclosure contain both basic and acidic functionalities that allow the compounds to be converted into either base or acid addition salts. [0024] The neutral forms of the compounds may be regenerated by contacting the salt with a base or acid and isolating the parent compound in the conventional manner. The parent form of the compound differs from the various salt forms in certain physical properties, such as solubility in polar solvents, but otherwise the salts are equivalent to the parent form of the compound for the purposes of the present disclosure. [0025] This disclosure also contemplates isomers of the compounds described herein (e.g., stereoisomers and/or atropisomers). For example, certain compounds of the present disclosure possess asymmetric carbon atoms (chiral centers), or hindered rotation about a single bond; the racemates, diastereomers, and enantiomers, and atropisomers (e.g., Ra, Sa, P and M isomers) of which are all intended to be encompassed within the scope of the present disclosure. Stereoisomeric forms may be defined, in terms of absolute stereochemistry, as (R) or (S), depicted uses dashes and/or wedges, and/or in terms of the direction the stereoisomer rotates plane-
polarized light (e.g., dextrorotary ((+) or (d)), or levorotary ((-) or (l))). When a stereochemical depiction (e.g., using dashes, , and/or wedges, ) is shown in a chemical structure, or a stereochemical assignment (e.g., using (R) and (S) notation, or (d) and (l) notation) is made in a chemical name, it is meant to indicate that the depicted/referred to isomer is present and substantially free of one or more other isomer(s) (e.g., enantiomers and diastereomers, when present). “Substantially free of” other isomer(s) indicates at least an 70/30 ratio of the indicated isomer to the other isomer(s), more preferably 80/20, 90/10, or 95/5 or more. In some embodiments, the indicated isomer will be present in an amount of at least 99%. A chemical bond to an asymmetric carbon that is depicted as a solid line ( ) indicates that all possible stereoisomers (e.g., enantiomers, diastereomers, racemic mixtures, etc.) at that carbon atom are included. In such instances, the compound may be present as a racemic mixture, scalemic mixture, or a mixture of diastereomers. [0026] The compounds of the present disclosure may also contain unnatural proportions of atomic isotopes at one or more of the atoms that constitute such compounds. Unnatural proportions of an isotope may be defined as ranging from the amount found in nature to an amount consisting of 100% of the atom in question. For example, the compounds may incorporate radioactive isotopes, such as for example tritium (3H), iodine-125 (125I) or carbon-14 (14C), or non-radioactive isotopes, such as deuterium (2H) or carbon-13 (13C). Such isotopic variations can provide additional utilities to those described elsewhere herein. For instance, isotopic variants of the compounds of the disclosure may find additional utility, including but not limited to, as diagnostic and/or imaging reagents, or as cytotoxic/radiotoxic therapeutic agents. Additionally, isotopic variants of the compounds of the disclosure can have altered pharmacokinetic and pharmacodynamic characteristics which can contribute to enhanced safety, tolerability or efficacy during treatment. In some embodiments, the compounds according to this disclosure are characterized by one or more deuterium atoms. [0027] The terms “patient” or “subject” are used interchangeably to refer to a human or a non- human animal (e.g., a mammal). [0028] The terms “treat”, “treating”, treatment” and the like refer to a course of action that eliminates, reduces, suppresses, mitigates, ameliorates, or prevents the worsening of, either temporarily or permanently, a disease, disorder or condition to which the term applies, or at least
one of the symptoms associated therewith. Treatment includes alleviation of symptoms, diminishment of extent of disease, inhibiting (e.g., arresting the development or further development of the disease, disorder or condition or clinical symptoms association therewith) an active disease, delaying or slowing of disease progression, improving the quality of life, and/or prolonging survival of a subject as compared to expected survival if not receiving treatment or as compared to a published standard of care therapy for a particular disease. [0029] The term “in need of treatment” as used herein refers to a judgment made by a physician or similar professional that a subject requires or will benefit from treatment. This judgment is made based on a variety of factors that are in the realm of the physician’s expertise, which may include a positive diagnosis of a disease, disorder or condition. [0030] The terms “prevent”, “preventing”, “prevention”, “prophylaxis” and the like refer to a course of action initiated in a manner (e.g., prior to the onset of a disease, disorder, condition or symptom thereof) so as to prevent, suppress, inhibit or reduce, either temporarily or permanently, a subject’s risk of developing a disease, disorder, condition or the like (as determined by, for example, the absence of clinical symptoms) or delaying the onset thereof, generally in the context of a subject predisposed to having a particular disease, disorder or condition. In certain instances, the terms also refer to slowing the progression of the disease, disorder or condition or inhibiting progression thereof to a harmful or otherwise undesired state. Prevention also refers to a course of action initiated in a subject after the subject has been treated for a disease, disorder, condition or a symptom associated therewith in order to prevent relapse of that disease, disorder, condition or symptom. In one or more embodiments, the preventative course of action is taken based on anticipation of a condition or event. In one embodiment, prevention refers to the prevention, suppression, inhibition or reduction of an allergic, immune, or autoimmune response in a subject suffering from an allergic, inflammatory, neuroinflammatory, neurological, immune, autoimmune, dermatological, respiratory, metabolic, cardiovascular or fibrotic disease, disorder, or condition. [0031] As used herein, the term “response” may refer to a symptom initiated by an irritant or trigger (e.g., an antigen originating from within the body, or from the external environment) in a subject. For example, “response” may refer to a reaction (e.g., hives, rash, welts, itchy skin, stinging skin, skin fissures, skin lesions, skin blisters, swelling (e.g., in joints, glands, or tissues), vertigo, fatigue, dizziness, fainting, lightheadedness, muscle weakness, headache, dry skin, dry
eyes, hair loss, numbness or tingling in extremities, joint pain and/or stiffness, sneezing, runny nose, stuffy nose, chest tightness and/or pain, shortness of breath, wheezing, itchy eyes, watery eyes, blurred vision, sensitivity to light, stomach cramping, abdominal pain, bloating, diarrhea, constipation, indigestion, heartburn, excessive flatulence, frequent urination, difficulty swallowing, muscle spasm, muscle tremor, unexpected weight gain or loss, increased thirst, increased hunger, irritability, temperature sensitivity, low blood pressure, constriction of airways, weak pulse, rapid pulse, nausea, vomiting, anemia, depression, and the like) caused by an external or internal trigger in a subject susceptible to the trigger. Subjects susceptible to a trigger are generally those suffering from an allergic, inflammatory, neuroinflammatory, neurological, immune, autoimmune, dermatological, respiratory, metabolic, cardiovascular or fibrotic disease, disorder, or condition, such as those described elsewhere herein. [0032] The term “in need of prevention” as used herein refers to a judgment made by a physician or other caregiver that a subject requires or will benefit from preventative care. This judgment is made based on a variety of factors that are in the realm of a physician’s or caregiver’s expertise. [0033] “Substantially pure” indicates that a component (e.g., a compound according to this disclosure) makes up greater than about 50% of the total content of the composition, and typically greater than about 60% of the total content. More typically, “substantially pure” refers to compositions in which at least 75%, at least 85%, at least 90% or more of the total composition is the component of interest. In some cases, the component of interest will make up greater than about 90%, or greater than about 95% of the total content of the composition. [0034] The terms “inhibitor of KIT” and “KIT inhibitor” may be used interchangeably, and refer to the ability of a molecule to decrease the activation of KIT either directly or indirectly, thereby decreasing activation and/or quantity of systemic mast cells. [0035] Compounds that are selective for KIT may be particularly useful in the treatment of certain disorders or may offer a reduced likelihood of undesired side effects. In one embodiment, compounds of the present disclosure are selective over one or more other receptor tyrosine kinases. Specific examples include, but are not limited to, PDGFRα, PDGFRβ, CSF1R, and FLT3. Selectivity may be determined, for example, by comparing the inhibition of a compound as described herein against KIT against the inhibition of a compound as described herein against another kinase. In one embodiment, the selective inhibition of a compound of Formula III or an
associated sub-formula is at least 1000 times greater, 500 times greater, 100 times greater, 50 times greater, or 20 times greater than inhibition of one or more kinases selected from PDGFRα, PDGFRβ, CSF1R, and FLT3. [0036] Compounds provided herein may have advantageous pharmacokinetic profiles including, for example, hepatocyte stability, clearance, inhibition against CYP, and inhibition against hERG. Compounds of the Disclosure [0037] The present disclosure relates to compounds that inhibit the activity of KIT. [0038] In one aspect, this disclosure is directed to a compound having a structure according to Formula III:
(Formula III) or a pharmaceutically acceptable salt thereof, wherein: A is: (i) selected from the group consisting of: , , ,
membered heteroaryl is substituted with 0-2 R1d; or (iii) -NHC(O)-R1g; each R1 is independently selected from the group consisting of halo, -CN, -OH, C1-C6- alkyl, C1-C6-haloalkyl, C1-C6-hydroxyalkyl, C1-C6-cyanoalkyl, C1-C6 alkoxy, C3-C6-cycloalkyl, - O-C3-C6-cycloalkyl, 4- to 8-membered heterocycloalkyl, -NR1aR1b, phenyl, 5- to 6-membered 12
heteroaryl, -X1-NR1aR1b, -X1-C(O)NR1aR1b, -X1-O-(C1-C3-alkyl), -X1-C3-C6-cycloalkyl, -X1- phenyl, and -X2-(4- to 8-membered heterocycloalkyl), wherein each 5- to 6-membered heteroaryl has 1-2 ring heteroatoms independently selected from N and O; each 4- to 8-membered heterocycloalkyl has 1-3 ring heteroatoms independently selected from N, O, and S, and said 4- to 8-membered heterocycloalkyl is optionally substituted with one oxo; and each R1 is optionally substituted with 1-3 R1c; or two adjacent R1 groups are combined with the two ring atoms to which they are attached to form a 5- or 6-membered heterocycloalkyl group having 1-2 ring heteroatoms independently selected from N, and O; each X1 is C1-C3-alkylene; each X2 is C1-C3-alkylene, or –(C1-C3-alkylene)-C(O)-; R1a and R1b, when present, are independently H, C1-C6 alkyl, or C3-C6 cycloalkyl; each R1c, when present, is independently halo, -OH, -C1-C6-alkyl, -C1-C6-alkoxy, or -C(O)- (C1-C6-alkyl); each R1d, when present, is independently halo, C1-C6 alkoxy, C1-C3 haloalkoxy, C3-C6- cycloalkyl, -O-C3-C6-cycloalkyl, or -NR1eR1f; R1e and R1f, when present, are independently H, C1-C6 haloalkyl, -(C1-C3-alkylene)-(C3- C6-cycloalkyl), or 4- to 8-membered heterocycloalkyl having 1-3 ring heteroatoms independently selected from N, O, and S; or R1e and R1f together with the nitrogen atom to which they are attached form a 4- to 6-membered heterocycloalkyl group having 1-2 ring heteroatoms independently selected from N, and O; R1g, when present, is C3-C6-cycloalkyl, -(C1-C3-alkylene)-(C3-C6-cycloalkyl), or 6- membered heteroaryl having 1-3 ring nitrogen atoms; wherein R1g is optionally substituted with 0-2 R1h; each R1h, when present, is independently halo, C1-C6 alkyl, -O-C3-C6-cycloalkyl, or -NH- (4- to 8-membered heterocycloalkyl), wherein the 4- to 8-membered heterocycloalkyl has 1-2 ring heteroatoms independently selected from N, O, and S; n is 0 or 1; m is 0, 1 or 2; R2, when present, is halo, -CN, -C1-C3-alkyl, -C1-C3-alkoxy, or C3-C6-cycloalkyl;
R3 is -H, halo, -C1-C6-alkyl, -C1-C6-haloalkyl, -C1-C6-hydroxyalkyl, -C1-C6-alkoxy, or - C(O)NR3aR3b; R3a and R3b, when present, are independently H, or C1-C3 alkyl; each R4, when present, is independently halo, C1-C6 alkyl, C1-C6-alkoxy, C3-C6 cycloalkyl, 4- to 8-membered heterocycloalkyl having 1-3 ring heteroatoms independently selected from N, O, and S, or phenyl, wherein said phenyl is optionally substituted with 1-3 halo; R5 is H, C1-C3-alkyl, or -NR5aR5b; R5a and R5b, when present, are independently H or C1-C3 alkyl; and R6 is H, or C1-C3-alkyl. [0039] In some embodiments, A is selected from the group consisting of:
, , ,
[0040] In some embodiments,
. In some embodiments, A is
some embodiments, A is
. In some embodiments, A is
. In some embodiments, A is . In some embodiments, A
. In some embodiments,
, . In some embodiments, A is
. In some embodiments, A is
. In some embodiments, A is
. In some embodiments, A is
. In some embodiments, A is
. In some embodiments,
A is
. In some embodiments,
. In some embodiments, A is
. In some embodiments, A is
. In some embodiments, A is
. In some embodiments, A is
. In some embodiments, A is
. In some embodiments, . In some embodiments, A is
. In some embodiments, A is
. some embodiments,
. In some embodiments, A is
. In some embodiments, A is
. In some embodiments, A is
. In some embodiments, A is
. In some embodiments, A is
. In some embodiments, A is
. , . , . In some embodiments, A is
. In some embodiments,
. some embodiments, A is
. , . In some embodiments,
. In some embodiments, A is
. In some embodiments,
. In some embodiments,
some embodiments,
some embodiments, A is
. , . In some embodiments, A is
some embodiments, A is
. In some embodiments,
. In some embodiments, A is
. In some embodiments, A is
. In some embodiments, A is
. In some embodiments, A is
. In some embodiments, A
, . In some embodiments, A is
. , . In some embodiments,
. In some embodiments,
. In some embodiments,
. In some
embodiments, A is
. In some embodiments,
. In some embodiments, A is
some embodiments, A is
. In some embodiments,
. In some embodiments,
. In some embodiments,
. In some embodiments, A is
. [0041] In some embodiments, A is a 6-membered heteroaryl having 1-3 ring nitrogen atoms, wherein the 6-membered heteroaryl is substituted with 0-2 R1d. In some embodiments, A is a 6- membered heteroaryl having 1-3 ring nitrogen atoms, wherein the 6-membered heteroaryl is substituted with 1-2 R1d. In some embodiments, A is pyridazinyl, pyrimidinyl, pyrazinyl, or pyridinyl, wherein A is substituted with 0-2 R1d. In some embodiments, A is pyridazinyl, pyrimidinyl, pyrazinyl, or pyridinyl, wherein A is substituted with 1-2 R1d. In some embodiments,
. [0042] In some embodiments, each R1d, when present, is independently halo, C1-C6 alkoxy, C1- C3 haloalkoxy, cyclopropyl, -O-cyclopropyl, or -NR1eR1f; and R1e and R1f, when present, are independently H, C1-C6 haloalkyl, -(methylene)-(cyclopropyl), or tetrahydrofuranyl; or R1e and R1f together with the nitrogen atom to which they are attached form an azetidinyl group.
[ , ,
[0044] In some embodiments, A is -NHC(O)-R1g. In some embodiments, R1g, when present, is cyclopropyl, -(methylene)-(cyclopropyl), pyridinyl, or pyrazinyl; wherein R1g is optionally substituted with 0-2 R1h. In some embodiments, R1g, when present, is
, , ,
. [0046] In another aspect, this disclosure is directed to a compound having a structure according to Formula III:
(Formula III) or a pharmaceutically acceptable salt thereof, wherein: ring A is a 5-membered heteroaryl having 1-4 ring heteroatoms independently selected from N, O, and S, wherein the heteroaryl is substituted with 0-2 R1; each R1 is independently selected from the group consisting of halo, -CN, -OH, C1-C6- alkyl, C1-C6-haloalkyl, C1-C6-hydroxyalkyl, C1-C6-cyanoalkyl, C1-C6 alkoxy, C3-C6-cycloalkyl, - O-C3-C6-cycloalkyl, 4- to 8-membered heterocycloalkyl, -NR1aR1b, phenyl, 5- to 6-membered heteroaryl, -X1-NR1aR1b, -X1-C(O)NR1aR1b, -X1-O-(C1-C3-alkyl), -X1-C3-C6-cycloalkyl, -X1- phenyl, and -X2-(4- to 8-membered heterocycloalkyl), wherein each 5- to 6-membered heteroaryl has 1-2 ring heteroatoms independently selected from N and O; each 4- to 8-membered heterocycloalkyl has 1-3 ring heteroatoms independently selected from N, O, and S, and said 4- to 8-membered heterocycloalkyl is optionally substituted with one oxo; and each R1 is optionally substituted with 1-3 R1c; or two adjacent R1 groups are combined with the two ring atoms to which they are attached to form a 5- or 6-membered heterocycloalkyl group having 1-2 ring heteroatoms independently selected from N, and O; each X1 is C1-C3-alkylene; each X2 is C1-C3-alkylene, or –(C1-C3-alkylene)-C(O)-; R1a and R1b, when present, are independently H, C1-C6 alkyl, or C3-C6 cycloalkyl; R1c, when present, is halo, -OH, -C1-C6-alkyl, -C1-C6-alkoxy, or -C(O)-(C1-C6-alkyl); n is 0 or 1; m is 0, 1 or 2; R2, when present, is halo, -CN, -C1-C3-alkyl, -C1-C3-alkoxy, or C3-C6-cycloalkyl; R3 is -H, halo, -C1-C6-alkyl, -C1-C6-haloalkyl, -C1-C6-hydroxyalkyl, -C1-C6-alkoxy, or - C(O)NR3aR3b; R3a and R3b, when present, are independently H, or C1-C3 alkyl;
each R4, when present, is independently halo, C1-C6 alkyl, C1-C6-alkoxy, C3-C6 cycloalkyl, 4- to 8-membered heterocycloalkyl having 1-3 ring heteroatoms independently selected from N, O, and S, or phenyl, wherein said phenyl is optionally substituted with 1-3 halo; R5 is H, C1-C3-alkyl, or -NR5aR5b; R5a and R5b, when present, are independently H or C1-C3 alkyl; and R6 is H, or C1-C3-alkyl. [0047] In some embodiments, ring A is selected from the group consisting of oxadiazolyl, thiazolyl, thiadiazolyl, triazolyl, imidazolyl, pyrazolyl, and tetrazolyl, each of which is substituted with 0-2 R1. In some embodiments, ring A is selected from the group consisting of thiazolyl, thiadiazolyl, triazolyl, imidazolyl, pyrazolyl, and tetrazolyl, each of which is substituted with 0-2 R1. In some embodiments, ring A is imidazolyl, pyrazolyl, triazolyl, or tetrazolyl, each of which is substituted with 0-2 R1. [0048] In some embodiments, ring A is selected from the group consisting of
, ,
, , , , , , , ,
, , each of which is substituted with 0-2 R1. In some embodiments, ring A , ,
with 0-2 R1. In some embodiments, ring A is selected from the group consisting of,
,
, , , , , , , ,
, each of which is substituted with 0-2 R1. In some embodiments, ring A is
each of which is substituted with 0-2 R1. In some embodiments, ring A is
substituted with 0-2 R1. In some embodiments, ring
. [0049] In some embodiments, ring A is a 5-membered heteroaryl having 1-4 ring nitrogen atoms, wherein the heteroaryl is substituted with two adjacent R1 groups that combine with the two ring atoms to which they are attached to form a 5- or 6-membered heterocycloalkyl group having 1-2 ring heteroatoms independently selected from N and O. In some embodiments, ring A is pyrazolyl or triazolyl substituted with two adjacent R1 groups that combine with the two ring atoms to which they are attached to form a 5- or 6-membered heterocycloalkyl group having 1-2 ring heteroatoms independently selected from N and O. In some embodiments, ring
r
. [0050] In some embodiments, R6 is H, or C1-C3-alkyl. In some embodiments, R6 is H or methyl. In some embodiments, R6 is methyl. In some embodiments, R6 is H.
[0051] In some embodiments, R5 is H, C1-C3-alkyl, or -NR5aR5b; wherein R5a and R5b are independently H or C1-C3 alkyl. In some embodiments, R5 is H, methyl, or -NH2. In some embodiments, R5 is H. In some embodiments, R5 is methyl. In some embodiments, R5 is -NR5aR5b, wherein at least one of R5a and R5b is H. In some embodiments, R5 is -NH2. [0052] In some embodiments, R5 and R6 are H. [0053] In some embodiments, m is 0, 1, or 2; and each R4, when present is independently halo, C1-C6 alkyl, C1-C6-alkoxy, C3-C6 cycloalkyl, 4- to 8-membered heterocycloalkyl having 1-3 ring heteroatoms independently selected from N, O, and S, or phenyl, wherein said phenyl is optionally substituted with 1-3 halo. In some embodiments, m is 0. In some embodiments, m is 1; and R4 is halo, C1-C6 alkyl, C1-C6-alkoxy, C3-C6 cycloalkyl, 4- to 8-membered heterocycloalkyl having 1-3 ring heteroatoms independently selected from N, O, and S, or phenyl, wherein said phenyl is optionally substituted with 1-3 halo. In some embodiments, m is 2; and each R4 is independently halo, C1-C6 alkyl, C1-C6-alkoxy, C3-C6 cycloalkyl, 4- to 8-membered heterocycloalkyl having 1-3 ring heteroatoms independently selected from N, O, and S, or phenyl, wherein said phenyl is optionally substituted with 1-3 halo. In some embodiments, m is 0, 1, or 2; and at least one R4, when present is halo. In some embodiments, R4 is -F, -Cl, -Br, or -I. In some embodiments, R4 is -F, or -Cl. In some embodiments, R4 is -F. In some embodiments, R4 is -Cl. In some embodiments, m is 0, 1, or 2, and at least one R4, when present, is C1-C6 alkyl. In some embodiments, R4 is C1- C3 alkyl. In some embodiments, R4 is methyl, ethyl, n-propyl, or isopropyl. In some embodiments, R4 is methyl, or ethyl. In some embodiments, R4 is methyl. In some embodiments, R4 is ethyl. In some embodiments, m is 0, 1, or 2; and at least one R4, when present, is C1-C6-alkoxy. In some embodiments, R4 is C1-C3-alkoxy. In some embodiments, R4 is methoxy, ethoxy, n-propoxy, or isopropoxy. In some embodiments, R4 is methoxy or ethoxy. In some embodiments R4 is methoxy. In some embodiments, R4 is ethoxy. In some embodiments, m is 0, 1, or 2; and at least one R4, when present, is C3-C6 cycloalkyl. In some embodiments, R4 is cyclopropyl, cyclobutyl, cyclopentyl, or cyclohexyl. In some embodiments, R4 is cyclopropyl. In some embodiments, m is 0, 1, or 2; and at least one R4, when present, is 4- to 8-membered heterocycloalkyl having 1-3 ring heteroatoms independently selected from N, O, and S. In some embodiments, R4 is a 5- to 6- membered heterocycloalkyl having 1-3 ring heteroatoms independently selected from N, O, and S. In some embodiments R4 is a 6-membered heterocycloalkyl having 1-3 ring heteroatoms
independently selected from N, and O. In some embodiments, R4 is piperidine, piperazine, or morpholine. In some embodiments, R4 is morpholine. In some embodiments, m is 0, 1, or 2; and at least one R4, when present, is phenyl optionally substituted with 1-3 halo. In some embodiments, R4 is phenyl substituted with one halo. In some embodiments, R4 is phenyl substituted with -F, - Cl, -Br, or -I. In some embodiments, R4 is phenyl substituted with -F. In some embodiments, m is 0, 1, or 2; and each R4, when present, is independently -F, -Cl, -CH3, -CH2CH3, -OCH3, - OCH2CH3, cyclopropyl,
. In some embodiments, m is 1; and R4, is independently -F, -Cl, -CH3, -CH2CH3, -OCH3, -OCH2CH3, cyclopropyl,
r
. In some embodiments, m is 2; and each R4 is independently -F, -Cl, -CH3, -CH2CH3, - OCH3, -OCH2CH3, cyclopropyl,
. [0054] In some embodiments, m is 0, 1, or 2; and each R4, when present is independently halo, C1-C6 alkyl, C1-C6-alkoxy, or C3-C6 cycloalkyl. In some embodiments, m is 0 or 1; and R4, when present is halo, C1-C6 alkyl, C1-C6-alkoxy, or C3-C6 cycloalkyl. In some embodiments, m is 0. In some embodiments, m is 1, and R4 is halo, C1-C6 alkyl, C1-C6-alkoxy, or C3-C6 cycloalkyl. In some embodiments, R4 is halo, C1-C3-alkyl, C1-C3-alkoxy, or C3-C5-cycloalkyl. In some embodiments, R4 is -F, -Cl, -CH3, -CH2CH3, -OCH3, -OCH2CH3, or cyclopropyl. [0055] In some embodiments, m is 0, 1, or 2; and each R4 is independently halo, or C1-C6-alkoxy. In some embodiments, at least one R4 is halo. In some embodiments, at least one R4 is Cl, Br, or F. In some embodiments, at least one R4 is Cl. In some embodiments at least one R4 is C1-C6- alkoxy. In some embodiments, at least one R4 is C1-C3-alkoxy. In some embodiments, at least one R4 is -OCH3. In some embodiments, m is 0, 1, or 2; and each R4 is independently Cl, or - OCH3. [0056] In some embodiments, R3 is halo, C1-C6 alkyl, or C1-C6 hydroxyalkyl. In some embodiments, R3 is halo, C1-C3 alkyl, or C1-C3 hydroxyalkyl. In some embodiments, R3 is halo. In some embodiments, R3 is -F, -Cl, or -Br. In some embodiments, R3 is -F, or -Cl. In some
embodiments, R3 is -F. In some embodiments, R3 is -Cl. In some embodiments, R3 is C1-C3 alkyl. In some embodiments, R3 is -CH3. In some embodiments, R3 is C1-C3 hydroxyalkyl. In some embodiments, R3 is -CH2-OH, -CH2CH2-OH, or -CH2CH2CH2-OH. In some embodiments, R3 is -CH2-OH. In some embodiments R3 is -F, -Cl, -CH3, or -CH2-OH. [0057] In some embodiments, R3 is halo or C1-C6-alkyl. In some embodiments, R3 is halo, or C1- C3-alkyl. In some embodiments, R3 is halo. In some embodiments, R3 is -F, -Cl, or -Br. In some embodiments, R3 is -Cl. In some embodiments, R3 is C1-C3 alkyl. In some embodiments, R3 is - CH3. In some embodiments, R3 is -Cl, or -CH3. [0058] In some embodiments, n is 0 or 1, and R2, when present is halo, -CN, -C1-C3-alkyl, -C1- C3-alkoxy, or C3-C6-cycloalkyl. In some embodiments, n is 0. In some embodiments, n is 1. In some embodiments, R2, when present, is halo. In some embodiments, R2, when present, is -F, - Cl, -Br, or -I. In some embodiments, R2, when present, is -F, or -Cl. In some embodiments, R2, when present, is -F. In some embodiments, R2, when present, is -Cl. In some embodiments, R2, when present, is -C1-C3-alkyl. In some embodiments, R2, when present, is methyl, ethyl, n-propyl, or isopropyl. In some embodiments, R2, when present, is methyl. In some embodiments, R2, when present, is -C1-C3-alkoxy. In some embodiments, R2, when present, is methoxy, ethoxy, n- propoxyl, or isopropoxy. In some embodiments, R2, when present, is methoxy. In some embodiments, R2, when present, is C3-C6-cycloalkyl. In some embodiments, R2, when present, is cyclopropyl, cyclobutyl, cyclopentyl, or cyclohexyl. In some embodiments, R2, when present, is cyclopropyl. In some embodiments, n is 0 or 1, and R2, when present, is -F, -Cl, -CN, -CH3, - OCH3, or cyclopropyl. [0059] In some embodiments, n is 1, and R2 is halo. In some embodiments, n is 1 and R2 is -F or -Cl. [0060] In some embodiments, each R1 is independently halo, -CN, -OH, C1-C6-alkyl, C1-C6- haloalkyl, C1-C6-hydroxyalkyl, C1-C6 alkoxy, C3-C6-cycloalkyl, -O-C3-C6-cycloalkyl, 4- to 6- membered heterocycloalkyl, -NR1aR1b, phenyl, 6-membered heteroaryl, -X1-C(O)NR1aR1b, -X1-O- (C1-C3-alkyl), -X1-C3-C6-cycloalkyl, and -X1-phenyl; wherein each 6-membered heteroaryl has 1- 2 ring heteroatoms independently selected from N and O; each 4- to 6-membered heterocycloalkyl has 1-3 ring heteroatoms independently selected from N, O, and S; and each R1 is unsubstituted or
substituted with 1-3 R1c; each R1a and R1b is independently H, C1-C3 alkyl, or C3-C6 cycloalkyl; each R1c is independently halo or -OH; and each X1 is C1-C3-alkylene. [0061] In some embodiments, each R1 is independently selected from the group consisting of C1-C6-alkyl, C1-C6-haloalkyl, C1-C6 alkoxy, C3-C6-cycloalkyl, 4- to 8-membered heterocycloalkyl having 1-2 ring heteroatoms independently selected from N and O, -NR1aR1b, and -X1-C3-C6- cycloalkyl; wherein X1 is C1-C3-alkylene; and R1a and R1b, when present, are independently H, or C1-C3 alkyl. [0062] In some embodiments, R1 is selected from the group consisting of C1-C6-alkyl, C1-C6- haloalkyl, C3-C6-cycloalkyl, and -X1-C3-C6-cycloalkyl, wherein X1 is C1-C3-alkylene. [0063] In some embodiments, R1 is C1-C6-alkyl. In some embodiments, R1 is C1-C4-alkyl. In some embodiments, R1 is methyl, ethyl, n-propyl, iso-propyl, iso-butyl, or tert-butyl. [0064] In some embodiments, R1 is C1-C6-haloalkyl. In some embodiments, R1 is C1-C3- haloalkyl. In some embodiments, R1 is difluoromethyl, trifluoromethyl, trifluoroethyl, difluoroethyl, trifluoropropyl, or difluoropropyl. In some embodiments, R1 is difluoromethyl, trifluoromethyl, trifluoroethyl, or trifluoropropyl. In some embodiments, R1 is -CF2H, CF3, - CH2CF2H, -CH2CF3, or -CH2CH2CF3. [0065] In some embodiments, R1 is C1-C6 alkoxy. In some embodiments, R1 is C1-C3 alkoxy. In some embodiments, R1 is methoxy. [0066] In some embodiments, R1 is C3-C6-cycloalkyl optionally substituted with 1-3 R1c. In some embodiments, R1 is C3-C6-cycloalkyl optionally substituted with 1-2 R1c. In some embodiments, R1 is C3-C5-cycloalkyl optionally substituted with 1-3 R1c. In some embodiments, R1 is C3-C5-cycloalkyl optionally substituted with 1-2 R1c. In some embodiments, R1 is cyclopropyl, cyclobutyl, cyclopentyl, or cyclohexyl, each of which is optionally substituted with 1-3 R1c. In some embodiments, R1 is cyclopropyl, cyclobutyl, cyclopentyl, or cyclohexyl, each of which is optionally substituted with 1-2 R1c. In some embodiments, R1 is cyclopropyl, cyclobutyl, or cyclopentyl, each of which is optionally substituted with 1-3 R1c. In some embodiments, R1 is cyclopropyl, cyclobutyl, or cyclopentyl, each of which is optionally substituted with 1-2 R1c. In some embodiments, each R1c when present, is independently halo or -OH. In some embodiments,
each R1c, when present, is independently -F, or -OH. In some embodiments, R1 is
,
. [0067] In some embodiments, R1 is C3-C6-cycloalkyl. In some embodiments, R1 is C3-C5- cycloalkyl. In some embodiments, R1 is cyclopropyl, cyclobutyl, cyclopentyl, or cyclohexyl. In some embodiments, R1 is cyclopropyl, cyclobutyl, or cyclopentyl. [0068] In some embodiments, R1 is -O-C3-C6-cycloalkyl. In some embodiments, R1 is -O- cyclopropyl, -O-cyclobutyl, -O-cyclopentyl, or -O-cyclohexyl. In some embodiments, R1 is -O- cyclopropyl. [0069] In some embodiments, R1 is 4- to 8-membered heterocycloalkyl having 1-2 ring heteroatoms independently selected from N and O. In some embodiments, R1 is 5- to 6-membered heterocycloalkyl having 1-2 ring heteroatoms independently selected from N and O. In some embodiments, R1 is pyrrolidinyl, tetrahydrofuranyl, tetrahydropyranyl, piperidinyl, or morpholinyl. In some embodiments, R1 is tetrahydrofuranyl, tetrahydropyranyl, or morpholinyl. In some embodiments, R1 is
, , . In some embodiments, R1 is
. [0070] In some embodiments, R1 is -NR1aR1b, wherein R1a and R1b are independently H, C1-C3 alkyl, or C3-C6 cycloalkyl. In some embodiments, R1 is -NR1aR1b, wherein R1a and R1b are independently H, or C1-C3 alkyl. In some embodiments, at least one of R1a and R1b is C1-C3 alkyl. In some embodiments, both R1a and R1b are C1-C3 alkyl. In some embodiments, at least one of R1a and R1b is C3-C6 cycloalkyl. In some embodiments, at least one of R1a and R1b is cyclopropyl. In some embodiments, R1 is -NH2. In some embodiments, R1 is -N(CH3)2. In some embodiments, R1 i
. [0071] In some embodiments, R1 is phenyl optionally substituted with 1-3 R1c. In some embodiments, R1 is phenyl optionally substituted with 1-2 R1c. In some embodiments, R1 is phenyl
optionally substituted with 1 R1c. In some embodiments R1 is phenyl optionally substituted with 1-3 R1c, wherein each R1c is independently halo. In some embodiments R1 is phenyl optionally substituted with 1-3 R1c, wherein each R1c is independently -F. In some embodiments, R1 is
. [0072] In some embodiments, R1 is 5- to 6-membered heteroaryl having 1-2 ring heteroatoms independently selected from N, and O. In some embodiments, R1 is a 6-membered heteroaryl having 1-2 ring heteroatoms that are independently N. In some embodiments, R1 is pyridinyl, pyrimidinyl, pyrazinyl, or pyridazinyl. In some embodiments, R1 is pyridyl. In some embodiments,
[0073] In some embodiments, R1 is -X1-phenyl; wherein X1 is -C1-C3-alkylene. In some embodiments, R1 is -X1-phenyl, wherein X1 is methylene, ethylene, n-propylene, or iso-propylene. In some embodiments,
. [0074] In some embodiments, R1 is -X1-C3-C6-cycloalkyl; wherein X1 is C1-C3-alkylene. In some embodiments, R1 is -X1-C3-C6-cycloalkyl; wherein X1 is methylene, ethylene, n-propylene, or iso-propylene. In some embodiments, R1 is -X1-cyclopropyl. In some embodiments, R1 is
. [0075] In some embodiments, each R1 is independently selected from the group consisting of - F, -Cl, -Br, -CN, -OH, , , , , , , , , , , , , , , , , , , , , , , , , , ,
,
[0076] In some embodiments, the compound, or a pharmaceutically acceptable salt thereof, has a structure according to Formula III:
(Formula III) wherein: A is: (i) selected from the group consisting of: , ,
(ii) a 6-membered heteroaryl having 1 or 2 ring nitrogen atoms, wherein the 6- membered heteroaryl is substituted with 0-2 R1d; or (iii) -NHC(O)-R1g; each R1 is independently selected from the group consisting of halo, CN, -OH, C1-C6-alkyl, C1-C6-haloalkyl, C1-C6-hydroxyalkyl, C1-C6 alkoxy, C3-C6-cycloalkyl, -O-C3-C6-cycloalkyl, 4- to
8-membered heterocycloalkyl, -NR1aR1b, phenyl, 5- to 6-membered heteroaryl, -X1-C(O)NR1aR1b, -X1-O-(C1-C3-alkyl), -X1-C3-C6-cycloalkyl, and -X1-phenyl, wherein each 5- to 6-membered heteroaryl has 1-2 ring heteroatoms independently selected from N and O; each 4- to 8-membered heterocycloalkyl has 1-3 ring heteroatoms independently selected from N, O, and S; and each R1 is optionally substituted with 1-3 R1c; each X1 is C1-C3-alkylene; R1a and R1b, when present, are independently H, C1-C6 alkyl, or C3-C6 cycloalkyl; each R1c, when present, is independently halo, or -OH; each R1d, when present, is independently halo, C1-C6 alkoxy, C3-C6-cycloalkyl, -O-C3-C6- cycloalkyl, or -NR1eR1f; R1e and R1f, when present, are independently H, C1-C6 haloalkyl, -(C1-C3-alkylene)-(C3- C6-cycloalkyl), or 4- to 8-membered heterocycloalkyl having 1-3 ring heteroatoms independently selected from N, O, and S; or R1e and R1f together with the nitrogen atom to which they are attached form a 4- to 6-membered heterocycloalkyl group having 1-2 ring heteroatoms independently selected from N, and O; R1g, when present, is C3-C6-cycloalkyl, -(C1-C3-alkylene)-(C3-C6-cycloalkyl), or 6- membered heteroaryl having 1 or 2 ring nitrogen atoms; wherein R1g is optionally substituted with 0-2 R1h; each R1h, when present, is independently halo, C1-C6 alkyl, -O-C3-C6-cycloalkyl, or -NH- (4- to 8-membered heterocycloalkyl), wherein the 4- to 8-membered heterocycloalkyl has 1-2 ring heteroatoms independently selected from N, O, and S; n is 0 or 1; m is 0, 1 or 2; R2, when present, is halo, -CN, -C1-C3-alkyl, -C1-C3-alkoxy, or C3-C6-cycloalkyl; R3 is halo, C1-C3 alkyl, or C1-C3 hydroxyalkyl; each R4, when present, is independently halo, C1-C6 alkyl, C1-C6-alkoxy, C3-C6 cycloalkyl, 4- to 8-membered heterocycloalkyl having 1-3 ring heteroatoms independently selected from N, O, and S, or phenyl, wherein said phenyl is optionally substituted with 1-3 halo; and R5 and R6 are H. [0077] In some embodiments, the compound, or a pharmaceutically acceptable salt thereof, has a structure according to Formula III:
(Formula III) wherein: ring A is a 5-membered heteroaryl having 1-4 ring heteroatoms independently selected from N, O, and S, wherein the heteroaryl is substituted with 0-2 R1; each R1 is independently selected from the group consisting of halo, CN, -OH, C1-C6-alkyl, C1-C6-haloalkyl, C1-C6-hydroxyalkyl, C1-C6 alkoxy, C3-C6-cycloalkyl, -O-C3-C6-cycloalkyl, 4- to 8-membered heterocycloalkyl, -NR1aR1b, phenyl, 5- to 6-membered heteroaryl, -X1-C(O)NR1aR1b, -X1-O-(C1-C3-alkyl), -X1-C3-C6-cycloalkyl, and -X1-phenyl, wherein each 5- to 6-membered heteroaryl has 1-2 ring heteroatoms independently selected from N and O; each 4- to 8-membered heterocycloalkyl has 1-3 ring heteroatoms independently selected from N, O, and S; and each R1 is optionally substituted with 1-3 R1c; each X1 is C1-C3-alkylene; R1a and R1b, when present, are independently H, C1-C6 alkyl, or C3-C6 cycloalkyl; R1c, when present, is halo, or -OH; n is 0 or 1; m is 0, 1 or 2; R2, when present, is halo, -CN, -C1-C3-alkyl, -C1-C3-alkoxy, or C3-C6-cycloalkyl; R3 is halo, C1-C3 alkyl, or C1-C3 hydroxyalkyl; each R4, when present, is independently halo, C1-C6 alkyl, C1-C6-alkoxy, C3-C6 cycloalkyl, 4- to 8-membered heterocycloalkyl having 1-3 ring heteroatoms independently selected from N, O, and S, or phenyl, wherein said phenyl is optionally substituted with 1-3 halo; and R5 and R6 are H. [0078] In some embodiments, the compound, or a pharmaceutically acceptable salt thereof, has a structure according to Formula III:
(Formula III) wherein: ring A is a 5-membered heteroaryl having 1-4 ring nitrogen atoms, wherein the heteroaryl is substituted with 0-2 R1; each R1 is independently selected from the group consisting of halo, -CN, C1-C6-alkyl, C1- C6-haloalkyl, C1-C6-hydroxyalkyl, C1-C6-cyanoalkyl, C1-C6 alkoxy, C3-C6-cycloalkyl, 4- to 8- membered heterocycloalkyl, -NR1aR1b, phenyl, 5- to 6-membered heteroaryl, -X1-NR1aR1b, -X1- C(O)NR1aR1b, -X1-O-(C1-C3-alkyl), -X1-C3-C6-cycloalkyl, -X1-phenyl, and -X2-(4- to 8-membered heterocycloalkyl), wherein each 5- to 6-membered heteroaryl has 1-2 ring heteroatoms independently selected from N and O; each 4- to 8-membered heterocycloalkyl has 1-3 ring heteroatoms independently selected from N, O, and S, and said heterocycloalkyl is optionally substituted with one oxo; and each R1 is optionally substituted with one R1c; or two adjacent R1 groups are combined with the two ring atoms to which they are attached to form a 5- or 6-membered heterocycloalkyl group having 1-2 ring heteroatoms independently selected from N and O; each X1 is C1-C3-alkylene; each X2 is C1-C3-alkylene, or –(C1-C3-alkylene)-C(O)-; R1a and R1b, when present, are independently H, C1-C6 alkyl, or C3-C6 cycloalkyl; R1c, when present, is halo, -OH, -C1-C6-alkyl, -C1-C6-alkoxy, or -C(O)-(C1-C6-alkyl); n is 0 or 1; m is 0, 1 or 2; R2, when present, is halo, -CN, -C1-C3-alkyl, -C1-C3-alkoxy, or C3-C6-cycloalkyl; R3 is -H, halo, -C1-C6-alkyl, -C1-C6-haloalkyl, C1-C6-alkoxy, or -C(O)NR3aR3b; R3a and R3b, when present, are independently H, or C1-C3 alkyl; each R4, when present, is independently halo, C1-C6 alkyl, C1-C6-alkoxy, or C3-C6 cycloalkyl; R5 is H, C1-C3-alkyl, or -NR5aR5b;
R5a and R5b, when present, are independently H or C1-C3 alkyl; and R6 is H, or C1-C3-alkyl. [0079] In some embodiments, the compound, or a pharmaceutically acceptable salt thereof, has a structure according to Formula IIIa:
. [0080] In some embodiments, the compound, or a pharmaceutically acceptable salt thereof, has a structure according to Formula II:
(Formula II) wherein: R1 is selected from the group consisting of C1-C6-alkyl, C1-C6-haloalkyl, C1-C6 alkoxy, C3- C6-cycloalkyl, 4- to 8-membered heterocycloalkyl, -NR1aR1b, and -X1-C3-C6-cycloalkyl, wherein said heterocycloalkyl has 1-2 ring heteroatoms independently selected from N and O; X1 is C1-C3-alkylene; R1a and R1b, when present, are independently H, or C1-C3 alkyl; n is 0 or 1; m is 0, 1 or 2; R2, when present, is halo; R3 is halo or C1-C6-alkyl; and each R4, when present, is independently halo, C1-C6 alkyl, C1-C6-alkoxy, or C3-C6 cycloalkyl. [0081] In some embodiments, the compound, or a pharmaceutically acceptable salt thereof, has a structure according to Formula I:
(Formula I) wherein: R1 is selected from the group consisting of C1-C6-alkyl, C1-C6-haloalkyl, C3-C6-cycloalkyl, and -X1-C3-C6-cycloalkyl, wherein X1 is C1-C3-alkylene; n is 0 or 1; R2, when present, is halo; R3 is C1-C6-alkyl; and R4 is H, halo, or C1-C6-alkoxy. [0082] In some embodiments, the compound has a structure according to Formula Ia:
(Formula Ia). [0083] In some embodiments, the compound has a structure according to Formula Ib:
(Formula Ib). [0084] In some embodiments, the compound has a structure according to Formula Ic:
(Formula Ic). [0085] In some embodiments, the compound, or a pharmaceutically acceptable salt thereof, has a structure according to Formula IV:
(Formula IV) wherein: R1 is selected from the group consisting of C1-C6-alkyl; C1-C6-haloalkyl; C3-C6-cycloalkyl; 4- to 8-membered heterocycloalkyl having 1-3 ring heteroatoms independently selected from N, O, and S; phenyl; and -X1-C3-C6-cycloalkyl; wherein X1 is C1-C3-alkylene; and the phenyl and C3- C6-cycloalkyl are optionally substituted with 1-3 R1c; each R1c, when present, is independently halo or -OH; n is 0 or 1; m is 0, 1 or 2; R2, when present, is halo, -CN, -C1-C3-alkyl, -C1-C3-alkoxy, or C3-C6-cycloalkyl; R3 is halo or C1-C6-alkyl; and R4 is H, halo, C1-C6 alkyl, or C1-C6-alkoxy. [0086] In some embodiments, the compound, or a pharmaceutically acceptable salt thereof, has a structure according to Formula IVa:
. [0087] In some embodiments, the compound is selected from the group consisting of:
[0088] In some embodiments, the compound is selected from the group consisting of:
, , ,
[0089] In some embodiments, the compounds according to this disclosure is a pharmaceutically acceptable salt of a compound described above or described in the Examples. Methods of Use [0090] The present disclosure provides methods for using the compounds described herein in the preparation of a medicament for inhibition of KIT. In some embodiments, the medicament is for inhibiting KIT. As used herein, the terms “inhibit”, “inhibition” and the like refer to the ability of an antagonist to decrease the function or activity of a particular target, e.g., KIT. The decrease is preferably at least a 50% and may be, for example, at least about 55%, at least about 60%, at least about 65%, at least about 70%, at least about 75%, at least about 80%, at least about 85%, at least about 90%, or at least about 95%. The present disclosure also encompasses the use of the compounds described herein in the preparation of a medicament for the treatment or prevention of diseases, disorders, and/or conditions that would benefit from inhibition of KIT. As one example, the present disclosure encompasses the use of the compounds described herein in the preparation of a medicament for the treatment of an allergic, inflammatory, neuroinflammatory, neurological, autoimmune, dermatological, respiratory, metabolic, cardiovascular or a fibrotic disease, disorder or condition. In some embodiments of the aforementioned methods, the compounds described
herein are used in combination with at least one additional therapy, examples of which are set forth elsewhere herein. [0091] In some embodiments, the compounds of this disclosure are inhibitors of wild type KIT. In other embodiments, the compounds are inhibitors of mutant forms of KIT. Exemplary mutant forms of KIT include, but are not limited to, KIT (L576P), KIT (V559D), KIT (V559D, T670I), KIT (V559D, V654A), KIT (D816V), KIT (D816H), and KIT (A829P). In some embodiments, the compounds of the disclosure inhibit both wild type KIT and one or more mutant forms selected from KIT (L576P), KIT (V559D), KIT (V559D, T670I), KIT (V559D, V654A), KIT (D816V), KIT (D816H), and KIT (A829P). [0092] As demonstrated herein, the compounds according to this disclosure potently inhibit the receptor tyrosine kinase (RTK) KIT. Mast cells release proinflammatory and immunomodulatory mediators upon activation via binding of Stem Cell Factor (SCF) to KIT. Mast cells respond to antigens when activated and are often identified by the co-expression of KIT and FcεRI. KIT signaling is necessary for mast cell differentiation, maturation and survival. For certain diseases, mast cell activation may play a central role in the onset and progression of the disease. Accordingly, inhibition of KIT may lead to mast cell depletion, and/or reduced mast cell activation, and provide a promising therapeutic approach for mast cell-driven diseases. [0093] Diseases, disorders, and/or conditions that would benefit from KIT inhibition may include those in which mast cells play a contributory or vital role, or which are mediated, at least in part, by mast cell degranulation or mast cell activation. [0094] Accordingly, in some embodiments, the compounds described herein are administered to a subject in need thereof in an amount effective to inhibit KIT. KIT inhibition may be assessed using a peripheral serum sample, blood sample or a tissue sample obtained from the subject. Activity may be determined, for example, by comparison to a previous sample obtained from the subject (i.e., prior to administration of the compound described herein) or by comparison to a reference value for a control group (e.g., standard of care, a placebo, etc.). [0095] Alternatively or in addition, in some embodiments, the compounds described herein are administered to a subject in need thereof in an amount effective to diminish the activity and/or quantity of systemic mast cells in the subject. Mast cell activity and quantity may be assessed
using a peripheral serum sample, blood sample or a tissue sample obtained from the subject. Activity may be determined, for example, by comparison to a previous sample obtained from the subject (i.e., prior to administration of the compound) or by comparison to a reference value for a control group (e.g., standard of care, a placebo, etc.). As a specific example, mast cell quantity can be assessed by measuring tryptase levels in a suitable sample (e.g., a blood or serum sample) from a subject to determine mast cell burden on the subject. In one embodiment, the compounds described herein are administered to a subject identified as having a high mast cell burden. [0096] Alternatively or in addition, in some embodiments, the compounds described herein are administered to a subject in need thereof to treat and/prevent an allergic, inflammatory, neuroinflammatory, neurological, immune, autoimmune, dermatological, respiratory, metabolic, cardiovascular, or fibrotic disease, disorder, or condition. In some embodiments, the compounds described herein are administered in combination with one or more additional therapeutic agents, examples of which are set forth elsewhere herein. [0097] Alternatively or in addition, in some embodiments, the compounds described herein are administered to a subject in need thereof to treat and/prevent a symptom or response associated with an allergic, inflammatory, neuroinflammatory, neurological, immune, autoimmune, dermatological, respiratory, metabolic, cardiovascular, or fibrotic disease, disorder, or condition. In some embodiments, the compounds described herein are administered in combination with one or more additional therapeutic agents, examples of which are set forth elsewhere herein. [0098] Alternatively or in addition, in some embodiments, the compounds described herein are administered to a subject suffering from an allergic, inflammatory, neuroinflammatory, neurological, immune, autoimmune, dermatological, respiratory, metabolic, cardiovascular, or fibrotic disease, disorder, or condition in order to treat and/or prevent a response or symptom associated therewith. In some embodiments, the compounds described herein are administered in combination with one or more additional therapeutic agents, examples of which are set forth elsewhere herein. [0099] Alternatively or in addition, in some embodiments, the compounds described herein are administered to a subject in need thereof to treat and/or prevent cancer or a cancer-related disease, disorder or condition. In some embodiments, the compounds described herein are administered to
a subject in need thereof to treat cancer, optionally in combination with at least one additional therapy, examples of which are set forth elsewhere herein. Inflammatory, immune, and autoimmune indications [0100] In one or more embodiments, the compounds described herein are useful in the treatment and/or prophylaxis of inflammatory, immune, and autoimmune-related diseases, disorders and conditions. Inflammatory, immune and autoimmune-related diseases, disorders and conditions include allergic, neuroinflammatory, neurological, dermatological, respiratory, metabolic, fibrotic, and cardiovascular diseases, disorders and conditions. A non-limiting list of inflammatory, immune, and autoimmune-related diseases, disorders and conditions which may be treated or prevented with the compounds and compositions of the present disclosure include allergies (e.g., food allergy, drug allergy, insect allergy, latex allergy, mold allergy, pet allergy, pollen allergy, hay-fever, ragweed allergy, allergic rhinitis, allergic conjunctivitis, eosinophilic esophagitis, and the like), arthritis (e.g., rheumatoid arthritis, inflammatory arthritis, psoriatic arthritis, osteoarthritis), asthma, eosinophilic asthma, bullous dermatosis, alopecia areata, chronic rhinosinusitis with nasal polyps (CRSwNP), coeliac disease, systemic sclerosis, netherton syndrome, idiopathic anaphylaxis, migraine, chronic graft verse host disease, multiple sclerosis, Alzheimer’s disease, autism, psoriasis, inflammatory bowel disease (e.g., Chrohn’s disease and ulcerative colitis), irritable bowel syndrome, lupus, Grave’s disease, Hashimoto’s thyroiditis, ankylosing spondylitis, Sjӧgren’s syndrome (SjS), angioedema, anaphylaxis, atopic dermatitis, urticaria (e.g., chronic spontaneous urticaria (CSU), acute urticaria, or physical urticaria such as popular urticaria, cold urticaria, cholinergic urticaria, solar urticaria, scleroderma, and dermatographic urticaria), mastocytosis, dermographism, systemic mastocytosis, mast cell activation syndrome, mast cell gastrointestinal disease, dermatitis herpetiformis, dermatosis, dermatitis, allergic contact dermatitis, eosinophilic gastrointestinal (GI) disease, type I diabetes, type II diabetes, prurigo nodularis, coronary heart disease, atherosclerosis, myocardial infarction, angina, pulmonary fibrosis, idiopathic pulmonary fibrosis, pulmonary arterial hypertension, primary pulmonary hypertension, chronic obstructive pulmonary disease, acute respiratory distress syndrome, hepatic fibrosis, renal fibrosis, cardiac fibrosis, cystic fibrosis, and bronchitis. [0101] In some embodiments, the inflammatory immune, or autoimmune-related disease, disorder or condition is allergies (e.g., food allergy, drug allergy, insect allergy, latex allergy, mold
allergy, pet allergy, pollen allergy, hay-fever, ragweed allergy, allergic rhinitis, allergic conjunctivitis, and eosinophilic esophagitis), allergic asthma, eosinophilic asthma, asthma, multiple sclerosis, inflammatory bowel disease (e.g., Chrohn’s disease and ulcerative colitis), lupus, Grave’s disease, Hashimoto’s thyroiditis, ankylosing spondylitis, Sjӧgren’s syndrome (SjS), urticaria (e.g., chronic spontaneous urticaria (CSU), acute urticaria, or physical urticaria including popular urticaria, cold urticaria, cholinergic urticaria, solar urticaria, scleroderma, and dermatographic urticaria), or anaphylaxis. [0102] In some embodiments, the inflammatory immune, or autoimmune-related disease, disorder, or condition is urticaria (e.g., chronic spontaneous urticaria (CSU), acute urticaria, or physical urticaria including popular urticaria, cold urticaria, cholinergic urticaria, solar urticaria, scleroderma, and dermatographic urticaria), allergic asthma, eosinophilic asthma, atopic dermatitis, allergies (e.g., food allergy, drug allergy, insect allergy, latex allergy, mold allergy, pet allergy, pollen allergy, hay-fever, ragweed allergy, allergic rhinitis, allergic conjunctivitis, and eosinophilic esophagitis), or mastocytosis. [0103] In some embodiments, the inflammatory immune, or autoimmune-related disease, disorder, or condition is urticaria (e.g., chronic spontaneous urticaria (CSU), acute urticaria, or physical urticaria including popular urticaria, cold urticaria, cholinergic urticaria, solar urticaria, scleroderma, and dermatographic urticaria), allergic asthma, eosinophilic asthma, atopic dermatitis, or allergies (e.g., food allergy, drug allergy, insect allergy, latex allergy, mold allergy, pet allergy, pollen allergy, hay-fever, ragweed allergy, allergic rhinitis, allergic conjunctivitis, and eosinophilic esophagitis). [0104] In some embodiments, the inflammatory immune, or autoimmune-related disease, disorder, or condition is urticaria (e.g., chronic spontaneous urticaria (CSU), acute urticaria, or physical urticaria including popular urticaria, cold urticaria, cholinergic urticaria, solar urticaria, scleroderma, and dermatographic urticaria), or allergies (e.g., food allergy, drug allergy, insect allergy, latex allergy, mold allergy, pet allergy, pollen allergy, hay-fever, ragweed allergy, allergic rhinitis, allergic conjunctivitis, and eosinophilic esophagitis). [0105] In some embodiments, the inflammatory immune, or autoimmune-related disease, disorder, or condition is urticaria (e.g., chronic spontaneous urticaria (CSU), acute urticaria, or
physical urticaria including popular urticaria, cold urticaria, cholinergic urticaria, solar urticaria, scleroderma, and dermatographic urticaria). [0106] In some embodiments, the inflammatory immune, or autoimmune-related disease, disorder, or condition is allergies (e.g., food allergy, drug allergy, insect allergy, latex allergy, mold allergy, pet allergy, pollen allergy, hay-fever, ragweed allergy, allergic rhinitis, allergic conjunctivitis, and eosinophilic esophagitis). [0107] In some embodiments, the inflammatory immune, or autoimmune-related disease, disorder, or condition is a cardiovascular disease (e.g., coronary heart disease or atherosclerosis). [0108] In some embodiments, the inflammatory immune, or autoimmune-related disease, disorder, or condition is a fibrotic disease (e.g., myocardial infarction, angina, chronic obstructive pulmonary disease, acute respiratory distress syndrome, osteoarthritis, pulmonary fibrosis, renal fibrosis, cystic fibrosis, bronchitis, or asthma). [0109] In some embodiments, the inflammatory immune, or autoimmune-related disease, disorder or condition is arthritis, asthma, multiple sclerosis, psoriasis, inflammatory bowel disease, (e.g., Chrohn’s disease and ulcerative colitis), Grave’s disease, Hashimoto’s thyroiditis, or ankylosing spondylitis. [0110] In one or more embodiments, the inflammatory immune, or autoimmune-related disease, disorder, or condition is antihistamine-refractory. In some embodiments, the inflammatory immune, or autoimmune-related disease, disorder, or condition is refractory to IgE inhibitor therapy. In some embodiments, the inflammatory immune, or autoimmune-related disease, disorder, or condition is contraindicated or resistant to antihistamines. Oncology Indications [0111] In one or more embodiments, the compounds described herein are useful in the treatment and/or prophylaxis of cancer (e.g., carcinomas, sarcomas, leukemias, lymphomas, myelomas, etc.). In certain embodiments, the cancer may be locally advanced and/or unresectable, metastatic, or at risk of becoming metastatic. Alternatively, or in addition, the cancer may be recurrent or no longer responding to a treatment, such as a standard of care treatment known to one of skill in the art. Exemplary types of cancer contemplated by this disclosure include melanoma, prostate cancer,
pancreatic cancer, squamous cell carcinoma, Hodgkin lymphoma, leukemia (e.g., chronic myeloid leukemia, chronic eosinophilic leukemia, chronic myelomonocytic leukemia, myeloid leukemia, acute myeloid leukemia, acute megakaryoblastic leukemia, mast cell leukemia, acute lymphocytic leukemia), gastric cancer (e.g., gastrointestinal stromal cancer), small bowel cancer, salivary gland cancer, adrenocortical cancer, thyroid cancer, breast cancer, endometrial cancer, cervical cancer, testicular cancer, esophageal cancer, lung cancer (e.g., small cell and non-small cell lung cancer), colorectal cancer, prostate cancer, liver cancer, bile duct cancer, gallbladder cancer, appendiceal cancer, urothelial cancer, neuroendocrine tumors, kidney cancer, head and neck cancer, bone cancer, brain cancer (e.g., glioblastoma, medulloblastoma), mesothelioma, and soft tissue sarcoma. [0112] In the aforementioned embodiments, the methods of the present disclosure may be practiced in an adjuvant setting or neoadjuvant setting. The methods described herein may be indicated as a first line, second line, third line, or greater line of treatment. [0113] The present disclosure also provides methods of treating or preventing other cancer- related diseases, disorders or conditions. The use of the term(s) cancer-related diseases, disorders and conditions is meant to refer broadly to conditions that are associated, directly or indirectly, with cancer and non-cancerous proliferative disease, and includes, e.g., angiogenesis, precancerous conditions such as dysplasia, and non-cancerous proliferative diseases disorders or conditions, such as benign proliferative breast disease and papillomas. For clarity, the term(s) cancer-related disease, disorder and condition do not include cancer per se. [0114] In general, the disclosed methods for treating or preventing cancer, or a cancer-related disease, disorder or condition, in a subject in need thereof comprise administering to the subject a compound according to this disclosure. In some embodiments, the present disclosure provides methods for treating or preventing cancer, or a cancer-related disease, disorder or condition with a compound disclosed herein and at least one additional therapy, examples of which are set forth elsewhere herein. Routes of Administration [0115] In some embodiments, pharmaceutical compositions containing a compound according to this disclosure may be in a form suitable for oral administration. Oral administration may involve swallowing the formulation thereby allowing the compound to be absorbed into the
bloodstream in the gastrointestinal tract. Alternatively, oral administration may involve buccal, lingual or sublingual administration, thereby allowing the compound to be absorbed into the blood stream through oral mucosa. [0116] In another embodiment, the pharmaceutical compositions containing a compound according to this disclosure may be in a form suitable for parenteral administration. Forms of parenteral administration include, but are not limited to, intravenous, intraarterial, intramuscular, intradermal, intraperitoneal, intrathecal, intracisternal, intracerebral, intracerebroventricular, intraventricular, and subcutaneous. Pharmaceutical compositions suitable for parenteral administration may be formulated using suitable aqueous or non-aqueous carriers. Depot injections, which are generally administered subcutaneously or intramuscularly, may also be utilized to release the compounds disclosed herein over a defined period of time. [0117] In another embodiment, the pharmaceutical compositions containing a compound according to this disclosure may be in a form suitable for administration via inhalation. Pharmaceutical compositions suitable for administration via inhalation are formulated such that the compound is dispersed via an aerosol spray, mist, or powder that can be inhaled into the airways. Administration via inhalation may be, for example, via an inhaler, or nebulizer. [0118] In another embodiment, the pharmaceutical compositions containing a compound according to this disclosure may be in a form suitable for topical administration to body surfaces such as the skin or mucous membranes. Forms of topical administration include transdermal, ophthalmic, otic, nasal, intraocular, vaginal, and rectal administration. [0119] Particular embodiments of the present disclosure contemplate oral administration or parenteral administration. Pharmaceutical Compositions [0120] The compounds of the present disclosure may be in the form of compositions suitable for administration to a subject. In one embodiment the disclosure provides a pharmaceutical composition comprising a compound of Formula III or an associated sub-formula (i.e., Formula I, Ia, Ib, Ic, II, IIIa, IV, or IVa), or a pharmaceutically acceptable salt thereof, and a pharmaceutically acceptable excipient. In certain embodiments, the compound may be present in the pharmaceutical composition in an effective amount. The pharmaceutical compositions may be used in the methods
of the present disclosure; thus, for example, the pharmaceutical compositions comprising a compound according to this disclosure, or a pharmaceutically acceptable salt thereof, can be administered to a subject in order to practice the therapeutic and prophylactic methods and uses described herein. [0121] The pharmaceutical compositions of the present disclosure can be formulated to be compatible with the intended method or route of administration. Routes of administration may include those known in the art. Exemplary routes of administration are oral, parenteral, topical, or via inhalation. Furthermore, the pharmaceutical compositions may be used in combination with one or more other therapies described herein in order to treat or prevent the diseases, disorders and conditions as contemplated by the present disclosure. In one embodiment, one or more other therapeutic agents contemplated by this disclosure are included in the same pharmaceutical composition that comprises the compound according to this disclosure. In another embodiment, the one or more other therapeutic agents are in a composition that is separate from the pharmaceutical composition comprising the compound according to this disclosure. [0122] In one aspect, the compounds described herein may be administered orally. Oral administration may be via, for example, capsule or tablets. In making the pharmaceutical compositions that include the compound of Formula III or an associated sub-formula, or a pharmaceutically acceptable salt thereof, the tablet or capsule typically includes at least one pharmaceutically acceptable excipient. An oral dosage form may alternatively be formulated as a solution or suspension. [0123] In another aspect, the compounds described herein may be administered parenterally, for example by intravenous injection. A pharmaceutical composition appropriate for parenteral administration may be formulated in solution for injection or may be reconstituted for injection in an appropriate system such as a physiological solution. Such solutions may include sterile water for injection, salts, buffers, and tonicity excipients in amounts appropriate to achieve isotonicity with the appropriate physiology. [0124] In another aspect, the compounds described herein may be administered via inhalation. A pharmaceutical composition appropriate for inhalation may be formulated as an aerosol, dry powder, or other form suitable for administration with nebulizers, pressurized metered-dose inhalers, or dry powder inhalers.
[0125] In another aspect, the compounds described herein may be administered topically. Pharmaceutical compositions suitable for topical administration may be formulated as, for example, a lotion, gel, patch, powder, paste, cream, foam, ointment, oil, spray, liniment, aerosol, or liquid. [0126] The pharmaceutical compositions described herein may be stored in an appropriate sterile container or containers. In some embodiments, the container is designed to maintain stability for the pharmaceutical composition over a given period of time. Administration [0127] In general, the disclosed methods comprise administering a compound of Formula III or an associated sub-formula described herein, or a composition thereof, in an effective amount to a subject in need thereof. An “effective amount” with reference to a KIT inhibitor of the present disclosure means an amount of the compound that is sufficient to engage the target (by inhibiting, or antagonizing the target) at a level that is indicative of the potency of the molecule. For KIT, target engagement can be determined by one or more biochemical or cellular assays resulting in an EC50, ED50, EC90, IC50, or similar value which can be used as one assessment of the potency of the compound. Assays for determining target engagement include, but are not limited to, those described in the Examples. The effective amount may be administered as a single quantity or as multiple, smaller quantities (e.g., as one tablet with “x” amount, as two tablets each with “x/2” amount, etc.). [0128] In some embodiments, the disclosed methods comprise administering a therapeutically effective amount of a compound of Formula III or an associated sub-formula described herein to a subject in need thereof. As used herein, the phrase “therapeutically effective amount” with reference to KIT inhibition means a dose regimen (i.e., amount and interval) of the compound that provides the specific pharmacological effect for which the compound is administered to a subject in need of such treatment. For prophylactic use, a therapeutically effective amount may be effective to eliminate or reduce the risk, lessen the severity, or delay the onset of the disease, including biochemical, histological and/or behavioral signs or symptoms of the disease. For treatment, a therapeutically effective amount may be effective to reduce, ameliorate, or eliminate one or more signs or symptoms associated with a disease, delay disease progression, prolong survival, decrease the dose of other medication(s) required to treat the disease, or a combination thereof. In one
embodiment, a therapeutically effective amount may be effective to reduce mast cell burden in the subject. With respect to cancer specifically, a therapeutically effective amount may, for example, result in the killing of cancer cells, reduce cancer cell counts, reduce tumor burden, eliminate tumors or metastasis, or reduce metastatic spread. A therapeutically effective amount of a KIT inhibitor need not always be effective in treating every individual subject to be deemed to be a therapeutically effective amount by those of skill in the art. A therapeutically effective amount may vary based on, for example, one or more of the following: the age and weight of the subject, the subject’s overall health, the stage of the subject’s disease, the route of administration, and prior or concomitant treatments. [0129] Administration may comprise one or more (e.g., one, two, or three or more) dosing cycles. [0130] In certain embodiments, the compounds of Formula III or an associated sub-formula contemplated by the present disclosure may be administered (e.g., orally, parenterally, topically, via inhalation, etc.) at about 0.01 mg/kg to about 50 mg/kg, or about 1 mg/kg to about 25 mg/kg, of subject’s body weight per day, one or more times a day, a week, or a month, to obtain the desired effect. In some embodiments, a suitable weight-based dose of a compound contemplated by the present disclosure is used to determine a dose that is administered independent of a subject’s body weight (i.e., a fixed-dose). In certain embodiments, a compound of the present disclosure is administered (e.g., orally, parenterally, etc.) at a fixed dosage levels of about 1 mg to about 1000 mg, particularly 1, 3, 5, 10, 15, 20, 25, 50, 75, 100, 150, 200, 250, 300, 400, 500, 600, 750, 800, 900, or 1000 mg, one or more times a day, a week, or a month, to obtain the desired effect. [0131] In certain embodiments, the compound of Formula III or an associated sub-formula is contained in a “unit dosage form”. The phrase “unit dosage form” refers to physically discrete units, each unit containing a predetermined amount of the compound, either alone or in combination with one or more additional agents, sufficient to produce the desired effect. It will be appreciated that the parameters of a unit dosage form will depend on the particular agent and the effect to be achieved.
Combination Therapy [0132] The present disclosure contemplates the use of the KIT inhibitors described herein alone, or in combination with one or more additional therapeutic agents. The use of the KIT inhibitors described herein in combination with one or more additional therapies may have a synergistic therapeutic or prophylactic effect on the underlying disease, disorder, or condition. In addition or alternatively, the combination therapy may allow for a dose reduction of one or more of the therapies, thereby ameliorating, reducing or eliminating adverse effects associated with one or more of the agents. [0133] In embodiments comprising one or more additional therapeutic agents, the KIT inhibitor described herein can be administered before, during, or after treatment with the additional therapeutic agents. In embodiments comprising one or more additional therapeutic agents, the therapeutic agents used in such combination therapy can be formulated as a single composition or as separate compositions. If administered separately, each therapeutic agent in the combination can be given at or around the same time, or at different times. Furthermore, the therapeutic agents are administered “in combination” even if they have different forms of administration (e.g., oral capsule and intravenous), they are given at different dosing intervals, one therapeutic agent is given at a constant dosing regimen while another is titrated up, titrated down or discontinued, or each therapeutic agent in the combination is independently titrated up, titrated down, increased or decreased in dosage, or discontinued and/or resumed during a patient’s course of therapy. If the combination is formulated as separate compositions, in some embodiments, the separate compositions are provided together in a kit. Inflammatory, immune and autoimmune-related diseases, disorders and conditions [0134] The present disclosure contemplates the use of the KIT inhibitors described herein in combination with one or more additional therapies useful in the treatment of inflammatory and immune-related diseases, disorders and conditions, such as those described elsewhere herein. [0135] In some embodiments, one or more of the additional therapies is an additional treatment modality such as, for example, diet modification, physical therapy, skin hydration, oxygen therapy, exercise, plasmapheresis, phototherapy, use of a humidifier, surgery (e.g., coronary artery bypass
graft surgery, angioplasty, stent implant, endarterectomy, and thyroidectomy), and behavioral intervention such as avoidance of external triggers (e.g., allergens) or harmful substances. [0136] In one or more embodiments, the compounds according to the disclosure can be combined with one or more anti-inflammatory agents. A non-limiting list of anti-inflammatory agents include non-steroidal anti-inflammatory drugs (NSAIDs) (e.g., ibuprofen, aspirin, naproxen, and celecoxib, etodolac, meloxicam, nabumetone, diclofenac, diflunisal, fenoprofen, and flurbiprofen); corticosteroids (e.g., cortisone, prednisone, prednisolone, methylprednisolone, dexamethasone, deflazacort, betamethasone, hydrocortisone, etc.); disease-modifying antirheumatic drugs (DMARDs) (e.g., methotrexate, sulfasalazine, hydroxychloroquine, and leflunomide); anti-tumor necrosis factor (anti-TNF) agents (e.g., infliximab, adalimumab, and certolizumab pegol); interferons (e.g., interferon alfa); integrin receptor antagonists (e.g., vedolizumab); mast-cell stabilizers (e.g., cromolyn sodium, nedocromil, and lodoxamide); and aminosalicylates (5-ASA) (e.g., balsalazide, mesalazine, olsalazine, and sulfasalazine). [0137] In some embodiments, the additional therapeutic agent comprises an analgesic agent, e.g., acetaminophen, NSAIDs, cyclooxygenase-2 (COX-2) inhibitors (e.g., celecoxib), tramadol, and opiates. In one embodiment, the analgesic agent is acetaminophen. [0138] In some embodiments, the additional therapeutic agent comprises an agent that targets one or more cytokines, such as, e.g., interleukin-4, interleukin-5, interleukin-12, interleukin-13, interleukin-17, and/or interleukin-23. In some embodiments, the agent blocks one or more pro- inflammatory cytokines (e.g., interleukin-12, interleukin,-17, and/or interleukin-23) (e.g., secukinumab, ixekizumab, brodalumab, ustekinumab, guselkumab, tildrakizumab, and risankizumab). In some embodiments, the agent blocks a cytokine that regulates allergic inflammation, e.g., interleukin-4 and/or interleukin 13 (e.g., dupilumab). In some embodiments, the agent blocks a cytokine that mediates eosinophil activation, e.g., interleukin-5 (e.g., benralizumab, mepolizumab, or reslizumab). [0139] In some embodiments, the at least one additional therapeutic agent comprises an agent that targets CD20, e.g., an anti-CD20 antibody (e.g., ocrelizumab) [0140] In one or more embodiments, the additional therapeutic agent comprises one or more immunosuppressants. Exemplary immunosuppressants include, but are not limited to,
azathioprine, cyclosporine, leflunomide, JAK-inhibitors (e.g., abrocitinib, baricitinib, tofacitinib, and upadacitinib), and targeted immunosuppressive antibodies (e.g., belimumab). [0141] In some embodiments, the additional therapeutic agent comprises one or more agents that target mast-cell derived immunomodulators, such as, for example, histamines, and/or leukotrienes. Exemplary antihistamines include brompheniramine, cetirizine, chloropheniramine, clemastine, diphenyldramine, fexofenadine, azelastine, carbinoxamine, cyproheptadine, desloratadine, emedastine, hydroxyzine, levocabastine, levocetirizine, and loratadine. Exemplary leukotriene modifiers include montelukast, zafirlukast, and zileuton. [0142] In one or more embodiments, the additional therapeutic agent comprises a Bruton’s tyrosine kinase (BTK) inhibitor. Exemplary BTK inhibitors include ibrutinib, acalabrutinib, remibrutinib, and zanubrutinib. [0143] In one or more embodiments, the additional therapeutic agent comprises an immunoglobulin E (IgE) inhibitor, e.g., an anti-IgE inhibitor (omalizumab, TNX-901, or ligelizumab). [0144] In one or more embodiments, the additional therapeutic agent comprises an agent that targets the cytokine IL-13, e.g., an anti-IL13 antibody (tralokinumab). [0145] In one or more embodiments, the additional therapeutic agent comprises a TSLP antagonist, e.g., an anti-TSLP antibody (Tezepelumab). [0146] In one or more embodiments, the additional therapeutic agent comprises a costimulation inhibitor of CD80 and CD86. Exemplary costimulation inhibitors of CD80 and CD86 include, but are not limited to, abatacept. [0147] In one or more embodiments, the additional therapeutic agent comprises an antagonist of siglec-8, e.g., an anti-siglec-8 antibody (lirentelimab). [0148] In one or more embodiments, the additional therapeutic agent comprises an immunotherapeutic agent. Exemplary immunotherapeutic agents include, but are not limited to, PALFORZIA®. [0149] In one or more embodiments, the additional therapeutic agent comprises an antagonist of IL-31RA, e.g., an anti-IL-31RA antibody (nemolizumab).
[0150] In some embodiments, the additional therapeutic agent comprises an anti-depressant. Exemplary anti-depressants contemplated include, but are not limited to, serotonin and norepinephrine reuptake inhibitors (SNRIs) (e.g., desvenlafaxine, duloxetine, levomilnacipran, milnacipran, and venlafaxine), selective serotonin reuptake inhibitors (SSRIs) (e.g., citalopram, escitalopram, fluoxetine, fluvoxamine, and sertraline), tricyclic antidepressants (e.g., imipramine, nortriptyline, amitriptyline, doxepin, and desipramine), and monoamine oxidase inhibitors (MAOIs) (e.g., tranylcypromine, phenelzine, and isocarboxazid). [0151] In some embodiments, the additional therapeutic agent comprises an antipsychotic. Exemplary antipsychotics include haloperidol, loxapine, thioridazine, molindone, thiothixene, fluphenazine, mesoridazine, trifluoperazine, perphenazine, chlorpromazine, aripiprazole, clozapine, ziprasidone, risperidone, quetiapine, and olanzapine. [0152] In some embodiments, the additional therapeutic agent comprises one or more anti- anxiety agents. Exemplary anti-anxiety agents include alprazolam, chlordiazepoxide, clonazepam, diazepam, lorazepam, and buspirone. [0153] In some embodiments, the additional therapeutic agent comprises one or more anticonvulsants (e.g., valproic acid, phenytoin, clonazepam, and carbamazepine). [0154] In some embodiments, the additional therapeutic agent comprises one or more respiratory agents. In some embodiments, the respiratory agent is a bronchodilator (e.g., adrenergic bronchodilator, anticholinergic bronchodilator, methylxanthines, and combinations thereof), an inhaled corticosteroid (e.g., beclomethasone, fluticasone, ciclesonide, mometasone, and budesonide), a beta andrenergic agonist (e.g., albuterol, metaproterenol, pirbuterol, terbutaline, isoetharine and levalbuterol), or leukotriene modifier (e.g., montelukast, zafirlukast, and zileuton). [0155] In one or more embodiments, the additional therapeutic agent comprises one or more nasal decongestants. Exemplary decongestants include oxymetazoline, phenylephrine, and pseudoephedrine. [0156] In some embodiments, the additional therapeutic agent comprises a cough suppressant. Exemplary cough suppressants include dextromethorphan, guaifenesin, and codeine.
[0157] In some embodiments, the compounds according to this disclosure are combined with a proton pump inhibitor (PPI). Exemplary PPIs include lansoprazole, omeprazole, pantoprazole, rebaprazole, and esomeprazole. [0158] In some embodiments, the additional therapeutic agent comprises an agent that modulates cognitive function, e.g., cholinesterase inhibitors (e.g., donepezil, rivastigmine, galantamine), N-methyl-D-aspartate (NMDA) receptor antagonists (e.g., memantine), and agents targeting aggregated soluble and insoluble forms of amyloid beta (e.g., aducanumab). [0159] In some embodiments, the additional therapeutic agent comprises an agent that targets thyroid function, such as, for example, anti-thyroid agents (e.g., radioiodine, propylthiouracil (PTU), and methimazole), or thyroid hormone replacement therapy (e.g., levothyrozine, or cytomel). [0160] In one or more embodiments, the additional therapeutic agent comprises one or more agents useful in the treatment of diabetes, such as, e.g., insulin; synthetic glucagon; hyperglycemic agents (e.g., metformin, sulfonylureas, glinides, thiazolidinediones, dipeptidyl peptidase-4 (DPP- 4) inhibitors; anti-hyperglycemic agents (e.g., sodium glucose cotransporter-2 (SGLT2) inhibitors including, e.g., canagliflozin, dapagliflozin, and empagliflozin); and GLP-1 receptor agonists (e.g., semaglutide, exenatide, dulaglutide, liraglutide, or lixisenatide). [0161] In some embodiments, the additional therapeutic agent comprises a diuretic. Exemplary diuretics include spironolactone, bumetanide, torsemide, hydrochlorothiazide, furosemide, and metolazone, and aldosterone antagonists (e.g., spironolactone and eplerenone). [0162] In one or more embodiments, the additional therapeutic agent comprises one or more of an antidiarrheal (e.g., eluxadoline, or alosetron), a laxative (lubiprostone, or a guanylate cyclase- C (GC-C agonist (e.g., linaclotide). [0163] In some embodiments, the additional therapeutic agent comprises a cholinergic modulator, such as a cholinergic agonist (e.g., chantix, pilocarpine, or bethanechol), or an anticholinergic agent (e.g., atropine, belladonna alkaloids, benztropine mesylate, clidinium, cyclopentolate, darifenacin, dicylomine, fesoterodine, flavoxate, glycopyrrolate, homatropine hydrobromide, hyoscyamine, ipratropium, orphenadrine, oxybutynin, propantheline, scopolamine, methscopolamine, solifenacin, tiotropium, tolterodine, trihexphenidyl, and trospium).
[0164] In another embodiment, the additional therapeutic agent comprises an antiarrhythmic agent. Antiarrhythmic agents include calcium channel blocking agents (e.g., amlodipine, diltiazem, felodipine, isradipine, nicardipine, nifedipine, nisoldipine, and verapamil), beta- adrenergic blocking agents (i.e., beta blockers) (e.g., atenolol, bisoprolol, carvedilol, labetalol, metoprolol, propranolol, and sotalol), potassium-channel blockers (e.g., amiodarone, dronedarone, dofetilide, ibutilide, azimilide, bretylium, clofilium, nifekalant, tedisamil, and sematilide), adenosine, electrolyte supplements, atropine, and digitalis compounds. [0165] In one or more embodiments, the additional therapeutic agent comprises a vasodilator. Exemplary vasodilators include, but are not limited to nitrates (e.g., nitroprusside, nitroglycerine, isosorbide, and amyl nitrate), hydralazine, treprostinil, minoxidil, angiotensin-converting enzyme (ACE) inhibitors (e.g., benazepril, captopril, enalapril, fosinopril, lisinopril, moexipril, perindopril, quinapril, ramipril, and trandolapril), and angiotensin receptor blockers (ARBs) (e.g., azilsartan, candesartan, eprosartan, irbesartan, losartan, olmesartan, telmisartan, and valsartan). [0166] In some embodiments, the additional therapeutic agent comprises a cholesterol modifier. Cholesterol modifiers include statins (atorvastatin, Fluvastatin, lovastatin, pitavastatin, pravastatin, rosuvastatin, and simvastatin), cholesterol absorption inhibitors (e.g., ezetimibe), proprotein convertase subtilisin kexin 9 (PCSK9) inhibitors (e.g., alirocumab, and evolocumab), citrate lyase inhibitors (e.g., bempedoic acid, and benpedoic acid-ezetimibe), bile acid sequestrants (e.g., cholestyramine, colesevelam, and colestipol), fibrates (e.g., fenofibrate, and gemfibrozil), niacin, and omega-3 fatty acids. [0167] In some embodiments, the additional therapeutic agent comprises a thrombolytic agent (e.g., streptokinase, alteplase, reteplase, Tenecteplase, urokinase, prourokinase, and anistreplase). [0168] In another embodiment, the additional therapeutic agent comprises an anticoagulant. Exemplary anticoagulants include rivaroxaban, dabigatran, apixaban, eboxaban, and warfarin. [0169] In some embodiments, the additional therapeutic agent comprises an agent useful in the treatment of fibrosis. Certain such agents include pirfenidone and nintedanib. [0170] In another embodiment, the additional therapeutic agent comprises a targeted agent useful in the treatment of pulmonary arterial hypertension. Targeted agents useful in the treatment of pulmonary arterial hypertension include phosphodiesterase-5 (PDE5) inhibitors (e.g., sildenafil,
tadalafil and vardenafil); guanylate cyclase stimulators (GCS) (e.g., adempas, riociguat, vericiguat and verquvo); endothelin receptor antagonists (e.g., bosentan, ambrisentan, and macitentan), and prostacyclin and analogues thereof. [0171] In some embodiments, the additional therapeutic agent includes a mucolytic, e.g., guaifenesin, carbocisteine, erdosteine, mecysteine, bromhexine, hyperosmolar saline, mannitol powder, and dornase alfa. [0172] In some embodiments, the additional therapeutic agent comprises a pancreatic enzyme, e.g., creon. [0173] In some embodiments, the additional therapeutic agent comprises an agent that targets a mutation in the cystic fibrosis transmembrane conductance regulator (CFTR) gene. Exemplary CFTR modulators include ivacaftor, elexacaftor, lumacaftor, and tezacaftor. [0174] In some embodiments, the additional therapeutic agent comprises an antibiotic. Exemplary antibiotics include, but are not limited to phenoxymethylpenicillin, dicloxacillin, amoxicillin, ampicillin, nafcillin, oxacillin, penicillin, cefaclor, cefazolin, cefadroxil, cephalexin, cefuroxime, cefixime, ceroxitin, ceftriaxone, doxycycline, minocycline, sarecycline, erythromycin, clarithromycin, azithromycin, fidaxomicin, roxithromycin, ciprofloxacin, ofloxacin, levofloxacin, moxifloxacin, sulfamethoxazole with trimethoprim, sulfasalazine, sulfacetamide, sulfadiazine silver, vancomycin, dalbavancin, oritavancin, and telavancin. [0175] In some embodiments, the additional therapeutic agent comprises one or more agents selected from the groups consisting of anti-inflammatory agents, analgesic agents, agents that target one or more cytokines, immunosuppressants, agents that targets one or more mast-cell derived immunomodulators, BTK inhibitors, IgE inhibitors, anti-depressants, anti-psychotics, anti-anxiety agents, anticonvulsants, respiratory agents, nasal decongestants, cough suppressants, proton pump inhibitors (PPIs), agents that modulate cognitive function, agents that target thyroid function, agents useful in the treatment of diabetes, diuretics, antidiarrheals, laxatives, GC-C agonists, cholinergic modulators, antiarrhythmics, vasodilators, cholesterol modifiers, thrombolytic agents, anticoagulants, agents useful in the treatment of fibrosis, agents useful in the treatment of arterial hypertension, mucolytic agents, pancreatic enzymes, CFTR modulators, and/or antibiotics.
[0176] In some embodiments, the additional therapeutic agent comprises one or more agents selected from the group consisting of an anti-inflammatory agent, an analgesic agent, an immunosuppressant, and/or an agent that targets one or more cytokines (e.g., IL-12, IL-17, and/or IL-23). [0177] In some embodiments, the additional therapeutic agent comprises one or more agents selected from the group consisting of a respiratory agent, an anti-inflammatory agent, an agent that targets one or more cytokines (e.g., IL-4 and/or IL-13), a mast-cell stabilizer, and/or an agent that targets a mast-cell derived immunomodulator (e.g., leukotrienes). [0178] In some embodiments, the additional therapeutic agent comprises one or more agents selected from the group consisting of an anti-depressant, an anti-psychotic, an anti-anxiety agent, an anticonvulsant, an agent that modulates cognitive function, an anti-CD20 antibody (e.g., ocrelizumab), an anti-inflammatory, and/or an immunosuppressant. [0179] In some embodiments, the additional therapeutic agent comprises an anti-inflammatory and/or an immunosuppressant. [0180] In some embodiments, the additional therapeutic agent comprises an anti-inflammatory agent, an immunosuppressant, and/or an agent that targets a mast-cell derived immunomodulator (e.g., antihistamine and/or leukotriene modulators). [0181] In some embodiments, the additional therapeutic agent comprises an antihistamine, a BTK inhibitor, and/or an IgE inhibitor. [0182] In some embodiments, the additional therapeutic agent comprises an antihistamine, an anti-inflammatory agent (e.g., a corticosteroid), and IgE inhibitor, and/or an immunosuppressant (e.g., cyclosporine). [0183] In some embodiments, the additional therapeutic agent comprises an anti-inflammatory agent, an immunosuppressive agent, an agent that targets one or more cytokines, or a combination thereof. [0184] In some embodiments, the additional therapeutic agent comprises a cholesterol modifier, a diuretic, an antiarrhythmic, a vasodilator, an anti-inflammatory, an analgesic agent, or any combination thereof.
[0185] In one or more embodiments, the compounds described herein are combined with one or more additional therapeutic agents that are considered to be the standard of care (SOC) for one or more of the inflammatory, immune, and/or autoimmune-related indications described herein. Exemplary SOC therapies for the indications described herein are summarized in Table 1 and Table 2 below. Table 1
66
Table 2
68
Cancer Therapies [0186] The present disclosure contemplates the use of the KIT inhibitors described herein in combination with one or more additional therapies useful in the treatment of cancer. [0187] In some embodiments, one or more of the additional therapies is an additional treatment modality. Exemplary treatment modalities include but are not limited to surgical resection of a tumor, bone marrow transplant, radiation therapy, and photodynamic therapy. [0188] In some embodiments, one or more of the additional therapies is a therapeutic agent. Exemplary therapeutic agents include chemotherapeutic agents, radiopharmaceuticals, hormone therapies, epigenetic modulators, ATP-adenosine axis-targeting agents (e.g., CD73 inhibitors, CD39 inhibitors, A2AR inhibitors, and/or A2BR inhibitors), signal transduction inhibitors (e.g., inhibitors of one or more of TYRO3, MERTK, EGFR, FGFR, VEGFR, HER-2, HER-3, BRAF, RET, MET, ABL, ALK, FLT-3, JAK, STAT, NF-kB), RAS signaling inhibitors (e.g., inhibitors of one or more of KRAS, HRAS, RAF, MEK, ERK, PTEN, SOS (e.g., SOS1), mTORC1, SHP2 (PTPN11), and AKT), PI3K inhibitors, arginase inhibitors, HIF inhibitors (e.g., inhibitors of HIF- 2α), AXL inhibitors, PAK4 inhibitors, immunotherapeutic agents, cellular therapies, gene therapies, immune checkpoint inhibitors (e.g., inhibitors of one or more of PD-1, PD-L1, TIGIT, CTLA-4, BTLA, LAG-3, and TIM-3), and agonists of stimulatory or co-stimulatory immune checkpoints. [0189] In some embodiments, one or more of the additional therapeutic agents is a chemotherapeutic agent. Examples of chemotherapeutic agents include, but are not limited to, alkylating agents such as thiotepa and cyclosphosphamide; alkyl sulfonates such as busulfan,
improsulfan and piposulfan; aziridines such as benzodopa, carboquone, meturedopa, and uredopa; ethylenimines and methylamelamines including altretamine, triethylenemelamine, triethylenephosphoramide, triethylenethiophosphoramide and trimethylolomelamime; nitrogen mustards such as chlorambucil, chlornaphazine, cholophosphamide, estramustine, ifosfamide, mechlorethamine, mechlorethamine oxide hydrochloride, melphalan, novembichin, phenesterine, prednimustine, trofosfamide, uracil mustard; nitrosureas such as carmustine, chlorozotocin, fotemustine, lomustine, nimustine, ranimustine; antibiotics such as aclacinomysins, actinomycin, authramycin, azaserine, bleomycins, cactinomycin, calicheamicin, carabicin, caminomycin, carzinophilin, chromomycins, dactinomycin, daunorubicin, detorubicin, 6-diazo-5-oxo-L- norleucine, doxorubicin, epirubicin, esorubicin, idarubicin, marcellomycin, mitomycins, mycophenolic acid, nogalamycin, olivomycins, pomalidomide, peplomycin, potfiromycin, puromycin, quelamycin, rodorubicin, streptonigrin, streptozocin, tubercidin, ubenimex, zinostatin, zorubicin; anti-metabolites such as methotrexate and 5-fluorouracil (5-FU); folic acid analogs such as denopterin, methotrexate, pteropterin, trimetrexate; purine analogs such as fludarabine, 6- mercaptopurine, thiamiprine, thioguanine; pyrimidine analogs such as ancitabine, azacitidine, 6- azauridine, carmofur, cytarabine, dideoxyuridine, doxifluridine, enocitabine, floxuridine, 5-FU; androgens such as calusterone, dromostanolone propionate, epitiostanol, mepitiostane, testolactone; anti-adrenals such as aminoglutethimide, mitotane, trilostane; folic acid replenisher such as folinic acid; aceglatone; aldophosphamide glycoside; aminolevulinic acid; amsacrine; bestrabucil; bisantrene; edatraxate; defofamine; demecolcine; diaziquone; elformithine; elliptinium acetate; etoglucid; gallium nitrate; hydroxyurea; lentinan; lonidamine; mitoguazone; mitoxantrone; mopidamol; nitracrine; pentostatin; phenamet; pirarubicin; podophyllinic acid; 2- ethylhydrazide; procarbazine; razoxane; sizofiran; spirogermanium; tenuazonic acid; triaziquone; 2,2',2''-trichlorotriethylamine; urethan; vindesine; dacarbazine; mannomustine; mitobronitol; mitolactol; pipobroman; gacytosine; arabinoside (Ara-C); cyclophosphamide; thiotepa; taxoids, e.g., paclitaxel, nab paclitaxel, and docetaxel; chlorambucil; gemcitabine; 6-thioguanine; mercaptopurine; methotrexate; platinum and platinum coordination complexes such as cisplatin, carboplatin and oxaliplatin; vinblastine; etoposide (VP-16); ifosfamide; mitomycin C; mitoxantrone; vincristine; vinorelbine; navelbine; novantrone; teniposide; daunomycin; aminopterin; xeloda; ibandronate; CPT11; proteasome inhibitors such as bortezomib, carfilzomib and ixazomib; topoisomerase inhibitors such as irinotecan, topotecan, etoposide, mitoxantrone,
teniposide; difluoromethylornithine (DMFO); retinoic acid; esperamicins; capecitabine; anthracyclines and pharmaceutically acceptable salts, acids or derivatives of any of the above. In certain embodiments, combination therapy comprises a chemotherapy regimen that includes one or more chemotherapeutic agents. In one embodiment, combination therapy comprises a chemotherapeutic regimen comprising FOLFOX (folinic acid, fluorouracil, and oxaliplatin), FOLFIRI (e.g., folinic acid, fluorouracil, and irinotecan), a taxoid (e.g., docetaxel, paclitaxel, nab- paclitaxel, etc.), and gemcitabine. [0190] In some embodiments, one or more of the additional therapeutic agents is an immune checkpoint inhibitor. As used herein, the term “immune checkpoint inhibitor” refers to an antagonist of an inhibitory or co-inhibitory immune checkpoint. The terms “immune checkpoint inhibitor”, “checkpoint inhibitor” and “CPI” may be used herein interchangeably. Immune checkpoint inhibitors may antagonize an inhibitory or co-inhibitory immune checkpoint by interfering with receptor -ligand binding and/or altering receptor signaling. Examples of immune checkpoints (ligands and receptors), some of which are selectively upregulated in various types of cancer cells, that can be antagonized include PD-1 (programmed cell death protein 1); PD-L1 (PD1 ligand); BTLA (B and T lymphocyte attenuator); CTLA-4 (cytotoxic T-lymphocyte associated antigen 4); TIM-3 (T cell immunoglobulin and mucin domain containing protein 3); LAG-3 (lymphocyte activation gene 3); TIGIT (T cell immunoreceptor with Ig and ITIM domains); CD276 (B7-H3), PD-L2, Galectin 9, CEACAM-1, CD69, Galectin-1, CD113, GPR56, VISTA, 2B4, CD48, GARP, PD1H, LAIR1, TIM-1, and TIM-4, and Killer Inhibitory Receptors, which can be divided into two classes based on their structural features: i) killer cell immunoglobulin- like receptors (KIRs), and ii) C-type lectin receptors (members of the type II transmembrane receptor family). Also contemplated are other less well-defined immune checkpoints that have been described in the literature, including both receptors (e.g., the 2B4 (also known as CD244) receptor) and ligands (e.g., certain B7 family inhibitory ligands such B7-H3 (also known as CD276) and B7-H4 (also known as B7-S1, B7x and VCTN1)). [See Pardoll, (April 2012) Nature Rev. Cancer 12:252-64]. [0191] In some embodiments, an immune checkpoint inhibitor is a CTLA-4 antagonist. In further embodiments, the CTLA-4 antagonist can be an antagonistic CTLA-4 antibody. Suitable
antagonistic CTLA-4 antibodies include, for example, monospecific antibodies such as ipilimumab or tremelimumab, as well as bispecific antibodies such as MEDI5752 and KN046. [0192] In some embodiments, an immune checkpoint inhibitor is a PD-1 antagonist. In further embodiments, the PD-1 antagonist can be an antagonistic PD-1 antibody, small molecule or peptide. Suitable antagonistic PD-1 antibodies include, for example, monospecific antibodies such as balstilimab, budigalimab, camrelizumab, cosibelimab, dostarlimab, cemiplimab, ezabenlimab (BI-754091), MEDI-0680 (AMP-514; WO2012/145493), nivolumab, pembrolizumab, pidilizumab (CT-011), pimivalimab, retifanlimab, sasanlimab, spartalizumab, sintilmab, tislelizumab, toripalimab, and zimberelimab; as well as bi-specific antibodies such as LY3434172. In still further embodiments, the PD-1 antagonist can be a recombinant protein composed of the extracellular domain of PD-L2 (B7-DC) fused to the Fc portion of IgGl (AMP-224). In certain embodiments, an immune checkpoint inhibitor is zimberelimab. [0193] In some embodiments, an immune checkpoint inhibitor is a PD-L1 antagonist. In further embodiments, the PD-L1 antagonist can be an antagonistic PD-L1 antibody. Suitable antagonistic PD-Ll antibodies include, for example, monospecific antibodies such as avelumab, atezolizumab, durvalumab, BMS-936559, and envafolimab as well as bi-specific antibodies such as LY3434172 and KN046. [0194] In some embodiments, an immune checkpoint inhibitor is a TIGIT antagonist. In further embodiments, the TIGIT antagonist can be an antagonistic TIGIT antibody. Suitable antagonistic anti-TIGIT antibodies include monospecific antibodies such as AGEN1327, AB308 (WO2021247591), BMS 986207, COM902, domvanalimab, EOS-448, etigilimab, IBI-929, JS006, M6223, ociperlimab, SEA-TGT, tiragolumab, vibostolimab; as well as bi-specific antibodies such as AGEN1777 and AZD2936. In certain embodiments, an immune checkpoint inhibitor is an antagonistic anti-TIGIT antibody disclosed in WO2017152088 or WO2021247591. In certain embodiments, an immune checkpoint inhibitor is domvanalimab or AB308. [0195] In some embodiments, an immune checkpoint inhibitor is a LAG-3 antagonist. In further embodiments, the LAG-3 antagonist can be an antagonistic LAG-3 antibody. Suitable antagonistic LAG-3 antibodies include, for example, BMS-986016 (WO10/19570, WO14/08218), or IMP-731 or IMP-321 (WO08/132601, WO09/44273).
[0196] In certain embodiments, an immune checkpoint inhibitor is a B7-H3 antagonist. In further embodiments, the B7-H3 antagonist is an antagonistic B7-H3 antibody. Suitable antagonist B7-H3 antibodies include, for example, MGA271 (WO11/109400), omburtumab, enoblituzumab, DS-7300a, ABBV-155, and SHR-A1811. [0197] In some embodiments, one or more of the additional therapeutic agents activates a stimulatory or co-stimulatory immune checkpoint. Examples of stimulatory or co-stimulatory immune checkpoints (ligands and receptors) include B7-1, B7-2, CD28, 4-1BB (CD137), 4-1BBL, ICOS, ICOS-L, OX40, OX40L, GITR, GITRL, CD70, CD27, CD40, DR3 and CD2. [0198] In some embodiments, an agent that activates a stimulatory or co-stimulatory immune checkpoint is a CD137 (4-1BB) agonist. In further embodiments, the CD137 agonist can be an agonistic CD137 antibody. Suitable CD137 antibodies include, for example, urelumab and PF- 05082566 (WO12/32433). In some embodiments, an agent that activates a stimulatory or co- stimulatory immune checkpoint is a GITR agonist. In further embodiments, the GITR agonist can be an agonistic GITR antibody. Suitable GITR antibodies include, for example, BMS-986153, BMS-986156, TRX-518 (WO06/105021, WO09/009116) and MK-4166 (WO11/028683). In some embodiments, an agent that activates a stimulatory or co-stimulatory immune checkpoint is an OX40 agonist. In further embodiments, the OX40 agonist can be an agonistic OX40 antibody. Suitable OX40 antibodies include, for example, MEDI-6383, MEDI-6469, MEDI-0562, PF- 04518600, GSK3174998, BMS-986178, and MOXR0916. In some embodiments, an agent that activates a stimulatory or co-stimulatory immune checkpoint is a CD40 agonist. In further embodiments, the CD40 agonist can be an agonistic CD40 antibody. In some embodiments, an agent that activates a stimulatory or co-stimulatory immune checkpoint is a CD27 agonist. In further embodiments, the CD27 agonist can be an agonistic CD27 antibody. Suitable CD27 antibodies include, for example, varlilumab. [0199] In some embodiments, one or more of the additional therapeutic agents is an ATP- adenosine axis-targeting agent. In certain embodiments, an ATP-adenosine axis-targeting agent is an inhibitor of an ectonucleotidase involved in the conversion of ATP to adenosine or an antagonist of adenosine receptor, e.g., ectonucleoside triphosphate diphosphohydrolase 1 (ENTPD1, also known as CD39 or Cluster of Differentiation 39) and the ecto-5'-nucleotidase (NT5E or 5NT, also known as CD73 or Cluster of Differentiation 73). Exemplary small molecule CD73 inhibitors
include CB-708, ORIC-533, LY3475070 and AB680. Exemplary anti-CD39 and anti-CD73 antibodies include ES002023, TTX-030, IPH-5201, SRF-617, CPI-006, oleclumab (MEDI9447), NZV930, IPH5301, GS-1423, uliledlimab (TJD5, TJ004309), BMS-986179, and AB598. In one embodiment, the present disclosure contemplates combination of the compounds described herein with a CD73 inhibitor such as those described in WO 2017/120508, WO 2018/067424, WO 2018/094148, and WO 2020/046813. In further embodiments, the CD73 inhibitor is quemliclustat (AB680). Adenosine can bind to and activate four different G-protein coupled receptors: A1R, A2AR, A2BR, and A3R. A2R antagonists include etrumadenant, inupadenant, taminadenant, caffeine citrate, NUV-1182, TT-702, DZD-2269, INCB-106385, EVOEXS-21546, AZD-4635, imaradenant, RVU-330, ciforadenant, PBF-509, PBF-999, PBF-1129, and CS-3005. In some embodiments, the present disclosure contemplates the combination of the compounds described herein with an A2AR antagonist, an A2BR antagonist, or an antagonist of A2AR and A2BR. In some embodiments, the present disclosure contemplates the combination of the compounds described herein with the adenosine receptor antagonists described in WO 2018/136700, WO 2018/204661, WO 2018/213377, or WO 2020/023846. In one embodiment, the adenosine receptor antagonist is etrumadenant. [0200] In some embodiments, one or more of the additional therapeutic agents is an inhibitor of a hypoxia-inducible factor (HIF) transcription factor, particularly HIF-2α. Exemplary HIF-2α inhibitors include belzutifan, ARO-HIF2, PT-2385, and those described in WO 2021113436, WO 2021188769, and WO 2023077046. In some embodiments, the HIF-2α inhibitor is AB521. [0201] In some embodiments, one or more of the additional therapeutic agents is an inhibitor of anexelekto (AXL). The AXL signaling pathway is associated with tumor growth and metastasis, and is believed to mediate resistance to a variety of cancer therapies. There are a variety of AXL inhibitors under development that also inhibit other kinases in the TAM family (i.e., TYRO3, MERTK), as well as other receptor tyrosine kinases including MET, FLT3, RON and AURORA, among others. Exemplary multikinase inhibitors include sitravatinib, rebastinib, glesatinib, gilteritinib, merestinib, cabozantinib, foretinib, BMS777607, LY2801653, S49076, and RXDX- 106. AXL specific inhibitors have also been developed, e.g., small molecule inhibitors including DS-1205, SGI-7079, SLC-391, TP-0903 (i.e., dubermatinib), BGB324 (i.e., bemcentinib), DP3975, and AB801; anti-AXL antibodies such as ADCT-601; and antibody drug conjugates
(ADCs) such as BA3011. Another strategy to inhibit AXL signaling involves targeting AXL’s ligand, GAS6. For example, batiraxcept (AVB-500) is under development as is a Fc fusion protein that binds the GAS6 ligand thereby inhibiting AXL signaling. In some embodiments, the additional therapeutic agent is an AXL inhibitor described in WO 2022246177, WO 2022246179, or PCT/US2023/069124. In some embodiments, the AXL inhibitor is AB801. [0202] In some embodiments, the additional therapeutic agent comprises chemotherapy, radiation therapy, or both. [0203] In one or more embodiments, the additional therapeutic agent comprises domvanalimab, etrumadenant, quemliclustat, zimberelimab, AB308, AB521, AB598, or AB801, or any combinations thereof. [0204] In one or more embodiments, the additional therapeutic agent comprises one or more of an immune checkpoint inhibitor, an A2R antagonist, a CD73 inhibitor, a HIF-2α inhibitor, a chemotherapeutic agent, radiation therapy, or any combinations thereof. In further embodiments of the above; (a) the immune checkpoint inhibitor comprises one or more inhibitors that block the activity of at least one of PD-1, PD-L1, BTLA, LAG-3, a B7 family member, TIM-3, TIGIT or CTLA-4, (b) the immune checkpoint inhibitor comprises an inhibitor of PD-1 or PD-L1; (c) the immune checkpoint inhibitor is selected from the group consisting of avelumab, atezolizumab, durvalumab, dostarlimab, cemiplimab, nivolumab, pembrolizumab, sintilmab, toripalimab, and zimberelimab; (d) the immune checkpoint inhibitor is zimberelimab; (e) the immune checkpoint inhibitor comprises an inhibitor that blocks the activity of TIGIT; (f) the immune checkpoint inhibitor is domvanalimab or AB308; (g) the A2R antagonist is selected from the group consisting of etrumadenant, inupadenant, taminadenant, caffeine citrate, NUV-1182, TT-702, DZD-2269, INCB-106385, EVOEXS-21546, AZD-4635, imaradenant, RVU-330, ciforadenant, PBF-509, PBF-999, PBF-1129, and CS-3005; (h) the A2R antagonist is etrumadenant; (i) the CD73 inhibitor is selected from the group consisting of CB-708, ORIC-533, LY3475070 and quemliclustat; (j) the CD73 inhibitor is quemliclustat; (k) the HIF-2α inhibitor is selected from the group consisting of belzutifan, ARO-HIF2, PT-2385, and AB521, (l) the inhibitor of HIF-2α is AB521; (m) the at least one additional therapeutic agent comprises a chemotherapeutic agent; (n) the chemotherapeutic agent comprises a platinum-based, taxoid-based, or anthracycline-based chemotherapeutic agent; (o) the chemotherapeutic agent is selected from the group consisting of
cisplatin, carboplatin, oxaliplatin, doxorubicin, docetaxel, and paclitaxel; and (p) the at least one additional therapeutic agent comprises radiation therapy. [0205] Selection of the additional therapeutic agent(s) may be informed by current standard of care for a particular cancer and/or mutational status of a subject’s cancer and/or stage of disease. Detailed standard of care guidelines are published, for example, by National Comprehensive Cancer Network (NCCN). See, for instance, NCCN Melanoma: Cutaneous v2.2021, NCCN Melanoma: Uveal v2.2021, NCCN Prostate Cancer v1.2022, NCCN Squamous Cell Skin Cancer v1.2022, NCCN Hodgkin Lymphoma v2.2022, NCCN Acute Lymphoblastic Leukemia v4.2021, NCCN Chronic Lymphocytic Leukemia/Small Lymphocytic Lymphoma v2.2022, NCCN Chronic Myeloid Leukemia v3.2022, NCCN Hairy Cell Leukemia v1.2022, NCCN Pediatric Acute Lymphoblastic Leukemia v1.2022, NCNN Small Bowel Adenocarcinoma v1.2022, NCCN Thyroid Carcinoma v3.2021, NCCN Non-Small Cell Lung Cancer v3.2022, NCCN Small Cell Lung Cancer v2.2022, NCCN Breast Cancer v2.2022, NCCN Colon Cancer v3.2021, NCCN Hepatobiliary Cancer v5.2021, NCCN Kidney Cancer, v3.2022, NCCN NSCLC v7.2021, NCCN Pancreatic Adenocarcinoma v2.2021, NCCN Esophageal and Esophagogastric Junction Cancers v4.2021, NCCN Gastric Cancer v5.2021, NCCN Gastrointestinal Stromal Tumors (GIST) v1.2022, NCCN Cervical Cancer v1.2022, NCCN Ovarian Cancer /Fallopian Tube Cancer /Primary Peritoneal Cancer v3.2021, NCCN Testicular Cancer v2.2022, NCCN Kidney Cancer v4.2021, NCCN Head and Neck Cancers v1.2022, NCCN Malignant Peritoneal Mesothelioma v1.2022, NCCN Malignant Pleural Mesothelioma v1.2022, NCCN Soft Tissue Sarcoma v3.2021, and NCCN Bone Cancer v2.2022. Methods of Synthesis General chemistry and methods for the preparation of compounds of the claims [0206] The exemplary molecules described herein and represented in the claims can be synthesized by the methods outlined in the following experimental section, and by various methods that are known to those skilled in the art. The exemplary molecules were synthesized from starting materials that are commercially available or readily prepared by standard synthetic methods. Equation 1 illustrates a retrosynthetic disconnection of the compounds of the invention into fragments a, b and c, which are useful for the construction of the compounds according to this disclosure. In general, compounds represented in the claims can be constructed using methods that
consist of three parts, which may be done in any order: modification of the functional groups present in fragments a, b or c; connection of the a and b fragments; and connection of fragments b and c.
[0207] There are several exemplary methods for the preparation of claimed compounds. Equation 2 demonstrates one method of forming the bond between fragments a and b. In the case of eq.2, a readily available carboxylic acid derived from fragment a is converted to an activated intermediate, such as an acid chloride or an activated ester, using a suitable amide coupling reagent(s). One skilled in the art will recognize that the conversion of carboxylic acids to acid chlorides can be accomplished using a wide range of conditions and reagents, such as oxalyl chloride or thionyl chloride. Alternatively, carboxylic acids are readily converted to activated esters through the use of amide coupling reagents, such as HATU, CDI, EDC, HOBt, T3P or various other reagents (see, e.g., “Synthesis of amides” in https://www.organic- chemistry.org/synthesis/C1N/amides.shtm). The activated ester or acid chloride derived from fragment a can be coupled with a wide variety of amines, including but not limited to anilines (fragment b). The transformation may be assisted or accelerated by heating and/or addition of a base.
[0208] Equations 3–6 demonstrate useful methods of synthesizing an appropriately substituted 1,2,4-triazole and connecting fragments b and c. In the case of Eq.3, an appropriately substituted aryl hydrazide readily condenses with an appropriately substituted carboximidamide or carboximidic acid ester to afford the corresponding 1,2,4-triazole, wherein “OR” typically consists of—but is not limited to—a methoxy or ethoxy group. The transformation may be assisted by the use of an organic or inorganic base and/or by heating. In the case of Eq.4, the functionalization of the reactants is reversed, wherein an appropriately substituted arylcarboximidamide or arylcarboximidic acid ester readily condenses with an appropriately substituted hydrazide. Equation 5 demonstrates another method of forming the bond between fragments b and c via a Pd-
catalyzed coupling reaction, such as a Suzuki reaction. In the case of Eq.5, -B(OR)2 represents a boronic acid or ester and X2 may be chosen from an appropriate halogen atom, such as Cl, Br, or I. The coupling is mediated by a transition metal catalyst, such as palladium with an appropriate ligand, and may be facilitated by the use of an organic or inorganic base and heating. The functionalization of the coupling partners may be reversed, as exemplified in Eq. 6, and a wide variety of conditions are known in the art to effect these transformations.
[0209] There are other possible combinations and synthetic sequences that will also give rise to the targeted products. Formation of the bond between fragments a and b may occur before or after connection of the b and c fragments, and each of these fragments may be further modified before or after connection of fragments a, b and c. A variety of the methods described above have been used to prepare compounds of the invention, some of which are exemplified in the examples. EXPERIMENTAL [0210] The following examples are put forth so as to provide those of ordinary skill in the art with a complete disclosure and description of how to make and use the present disclosure, and are
not intended to limit the scope of what the inventors regard as their invention. Additional compounds within the scope of this disclosure may be made using methods based on those illustrated in these examples, or based on other methods known in the art. Efforts have been made to ensure accuracy with respect to numbers used (e.g., amounts, temperature, etc.), but some experimental errors and deviations should be accounted for. [0211] Unless indicated otherwise, parts are parts by weight, molecular weight is average molecular weight, temperature is in degrees Celsius (°C), and pressure is at or near atmospheric. Standard abbreviations are used, including the following: ATP=adenosine triphosphate; BSA=bovine serum albumin; AcOH = acetic acid; DCM and CH2Cl2=dichloromethane; DIPEA and EtNiPr2 =N,N-diisopropylethylamine; DMF=N,N-dimethylformamide; DMSO=dimethyl sulfoxide; DMSO-d6=perdeuterated dimethyl sulfoxide; NMP = N-methyl-2-pyrrolidone; h or hr=hour(s); s or sec=second(s); min=minute(s); ng=nanogram; μg=microgram; mg=milligram; g=gram; kg=kilogram; μl or μL=microliter; ml or mL=milliliter; l or L=liter; μM=micromolar; mM=millimolar; M=molar; nm = nanometers; rt = room temperature; anhyd. = anhydrous; sat. = saturated; aq.=aqueous; SiO2=silica gel; EtOAc=ethyl acetate; EtOH=ethanol; equiv.=equivalents; MeCN = acetonitrile; MeOH=methanol; THF = tetrahydrofuran; N2=nitrogen gas; HATU=N- [(dimethylamino)-1H-1,2,3-triazolo-[4,5-b]pyridin-1-ylmethylene]-N-methylmethanaminium hexafluorophosphate N-oxide; Methanol-d4=perdeuterated methanol; CDI = 1,1’- carbonyldiimidazole; EDC = 1-Ethyl-3-(3-dimethylaminopropyl)carbodiimide; HOBt = hydroxybenzotriazole; T3P = propylphosphonic anhydride; NaBH4=sodium borohydride; NaCl = sodium chloride; NaOAc = sodium acetate; NaOH = sodium hydroxide; NaH2PO4 = sodium phosphate monobasic; NaHCO3 = sodium bicarbonate; Na2CO3 = sodium carbonate; K2CO3 = potassium carbonate; MgSO4 = magnesium sulfate; MgCl2 = magnesium chloride; LiOH.H2O = lithium hydroxide monohydrate; Cs2CO3 = cesium carbonate; POCl3 = phosphoryl chloride; MTBE=tert-butyl methyl ether; TBS-Cl = tert-butyldimethylsilyl chloride; Me3SiCF3 = (trifluoromethyl)trimethylsilane; TFA = trifluoroacetic acid; tBuBrettPhos = 2-(di-tert- butylphosphino)-2′,4′,6′-triisopropyl-3,6-dimethoxy-1,1′-biphenyl; PCy3 Pd G2= chloro[(tricyclohexylphosphine)-2-(2′-aminobiphenyl)]palladium(II); XPhos=2- dicyclohexylphosphino-2',4',6'-triisopropylbiphenyl; XPhos Pd G3 = (2-dicyclohexylphosphino- 2′,4′,6′-triisopropyl-1,1′-biphenyl)[2-(2′-amino-1,1′-biphenyl)]palladium(II) methanesulfonate; Xantphos=4,5-bis(diphenylphosphino)-9,9-dimethylxanthene; Pd2(dba)3 =
tris(dibenxylideneacetone)dipalladium(0); PdCl2(dppf) = [1,1′-bis(diphenylphosphino)ferrocene] dichloropalladium(II); K4Fe(CN)6•3H2O=potassium hexacyanoferrate(II) trihydrate; MHz=megahertz; Hz=hertz; ppm=parts per million; ESI MS = electrospray ionization mass spectrometry; NMR=nuclear magnetic resonance; HPLC=high performance liquid chromatography; TLC = thin layer chromatography, LCMS = liquid chromatography-mass spectrometry. [0212] All reactions were performed using a Teflon-coated magnetic stir bar at the indicated temperature and were conducted under an inert atmosphere when stated. Purchased starting materials and reagents were generally used as received. Reactions were monitored by TLC (silica gel 60 with fluorescence F254, visualized with a short wave/long wave UV lamp) and/or LCMS (Agilent® 1100 or 1200 series LCMS with UV detection at 254 or 280 nm using a binary solvent system [0.1% formic acid in MeCN/0.1% formic acid in H2O] using one of the following columns: Agilent® Eclipse Plus C18 [3.5 μm, 4.6 mm i.d. × 100 mm], Waters™ XSelect HSS C18 [3.5 μm, 2.1 mm i.d. × 75 mm]). Flash chromatography was conducted on silica gel using an automated system (CombiFlash® RF+ manufactured by Teledyne ISCO), with detection wavelengths of 254 and 280 nm, and optionally equipped with an evaporative light scattering detector. Reverse phase preparative HPLC was conducted on an Agilent® 1260 or 1290 Infinity series HPLC. Samples were eluted using a binary solvent system (MeCN/H2O with an acid modifier as needed – for example 0.1% TFA or 0.1% formic acid) with gradient elution on a Gemini C18110 Å column (21.2 mm i.d. ×x 250 mm) with variable wavelength detection. Final compounds obtained through preparative HPLC were concentrated through lyophilization. All assayed compounds were purified to ≥95% purity as determined by 1H NMR or LCMS (Agilent® 1100 or 1200 series LCMS with UV detection at 254 or 280 nm using a binary solvent system [0.1% formic acid in MeCN/0.1% formic acid in H2O] using one of the following columns: Agilent® Eclipse Plus C18 [3.5 μm, 4.6 mm i.d. × 100 mm], Waters™ XSelect HSS C18 [3.5 μm, 2.1 mm i.d. × 75 mm]). 1H NMR spectra were recorded on a Varian 400 MHz NMR spectrometer equipped with an Oxford AS400 magnet or a Bruker® AVANCE NEO 400 MHz NMR. Chemical shifts (δ) are reported as parts per million (ppm) relative to residual undeuterated solvent as an internal reference. The abbreviations s, br s, d, t, q, dd, dt, ddd, dddd, and m stand for singlet, broad singlet, doublet, triplet, quartet, doublet of doublets, doublet of triplets, doublet of doublet of doublets, doublet of doublet of doublet of doublet, and multiplet, respectively.
General Procedure exemplified for N-[5-(5-Cyclopropyl-4H-1,2,4-triazol-3-yl)-2- methylphenyl]pyrazolo[1,5-a]pyridine-3-carboxamide, Ex.1
[0213] Step a: A 40 mL vial was charged with pyrazolo[1,5-a]pyridine-3-carboxylic acid (486 mg, 3.0 mmol, 1.0 equiv.), HATU (1.71 g, 4.5 mmol, 1.5 equiv.), EtNiPr2 (1.53 mL, 9.0 mmol, 3.0 equiv.) and DMF (8.6 mL, 0.35 M). The resulting mixture was stirred at 50 °C for 1 h. Upon complete conversion, as judged by LCMS analysis, the reaction mixture was cooled to rt and diluted with water. The resulting precipitated solid was collected by vacuum filtration, rinsed with water, and dried in vacuo to afford triazolo[4,5-b]pyridin-3-yl pyrazolo[1,5-a]pyridine-3- carboxylate as a tan solid (765 mg, 91% yield), which was used directly in the next step without further purification. [0214] Step b: A mixture of 2-methyl-5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)aniline HCl (320 mg, 1.14 mmol, 1.0 equiv.), EtNiPr2 (0.36 ml, 2.0 equiv.) and triazolo[4,5-b]pyridin-3- yl pyrazolo[1,5-a]pyridine-3-carboxylate (280 mg, 1.04 mmol, 1.1 equiv.) in NMP (2 mL, 0.5 M) was stirred at 120 °C for approximately 3 h. Upon complete conversion, as judged by LCMS analysis, the reaction mixture was cooled to rt and diluted with water. The resulting precipitated solid was collected by vacuum filtration, rinsed with water, and dried in vacuo to afford N-[2- methyl-5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)phenyl]pyrazolo[1,5-a]pyridine-3- carboxamide, which was used directly in the next step without further purification. The product of step b could also be accessed via an alternative, two-step protocol involving an initial amidation with 5-bromo-2-methylaniline followed by Pd-catalyzed borylation.
[0215] Step c: The crude intermediate obtained via step b (20 mg in 0.1 mL NMP, 0.053 mmol, 1.0 eq.) was combined with triazole bromide (20 mg, 0.106 mmol, 2.0 equiv.), XPhos Pd G3 (20 mg, 0.024 mmol, 45 mol%), 1 M aq. Na2CO3 solution (0.4 mL, 0.4 mmol, 7.5 equiv.), and dioxane (0.4 mL, 0.07 M). The reaction mixture was briefly degassed by evacuation/back-filling with N2 3x. The reaction mixture was stirred at 100 °C for approximately 2 h, at which time LCMS analysis indicated complete consumption of starting material. The reaction mixture was cooled to rt and diluted with EtOAc. The organic layer was separated, filtered over Celite®, and concentrated in vacuo. The crude product was purified by HPLC to provide the title compound as the HCl salt (5.0 mg, 25% yield).1H NMR (400 MHz, Methanol-d4) δ 8.73 – 8.62 (m, 2H), 8.29 (d, J = 8.9 Hz, 1H), 8.11 (d, J = 1.8 Hz, 1H), 7.80 (dd, J = 8.0, 1.8 Hz, 1H), 7.60 – 7.44 (m, 2H), 7.17 – 7.06 (m, 1H), 2.46 (s, 3H), 2.36 (ddd, J = 11.0, 7.4, 3.8 Hz, 1H), 1.48 – 1.40 (m, 2H), 1.34 – 1.23 (m, 2H). ESI MS [M+H]+ for C20H19N6O, calcd 359.2, found 359.2. Example 2: N-[5-(5-Ethyl-4H-1,2,4-triazol-3-yl)-2-methylphenyl]pyrazolo[1,5-a]pyridine-3- carboxamide
[0216] The title compound was prepared from 3-bromo-5-ethyl-4H-1,2,4-triazole in a similar fashion to Ex.1.1H NMR (400 MHz, Methanol-d4) δ 8.59 – 8.53 (m, 2H), 8.28 (dt, J = 8.8, 1.2 Hz, 1H), 7.96 (d, J = 1.9 Hz, 1H), 7.75 (dd, J = 8.1, 1.9 Hz, 1H), 7.44 (ddd, J = 9.0, 6.8, 1.1 Hz, 1H), 7.39 (d, J = 8.0 Hz, 1H), 7.03 (td, J = 6.9, 1.4 Hz, 1H), 2.86 (q, J = 7.7 Hz, 2H), 2.38 (s, 3H), 1.37 (t, J = 7.7 Hz, 3H). ESI MS [M+H]+ for C19H19N6O, calcd 347.2, found 347.2. Example 3: N-[5-(5-Ethyl-4H-1,2,4-triazol-3-yl)-4-fluoro-2-methylphenyl]pyrazolo[1,5- a]pyridine-3-carboxamide
[0217] The title compound was prepared from 3-bromo-5-ethyl-4H-1,2,4-triazole and 5-bromo- 4-fluoro-2-methylaniline in a similar fashion to Ex.1.1H NMR (400 MHz, Methanol-d4) δ 8.57 –
8.53 (m, 2H), 8.27 (dt, J = 9.0, 1.2 Hz, 1H), 7.88 (d, J = 7.0 Hz, 1H), 7.43 (ddd, J = 9.0, 6.8, 1.1 Hz, 1H), 7.19 – 7.13 (m, 1H), 7.03 (td, J = 6.9, 1.4 Hz, 1H), 2.85 (q, J = 7.6 Hz, 2H), 2.37 (s, 3H), 1.36 (t, J = 7.7 Hz, 3H). ESI MS [M+H]+ for C19H18FN6O, calcd 365.1, found 365.1. Example 4: N-[5-(5-tert-butyl-4H-1,2,4-triazol-3-yl)-2-methylphenyl]pyrazolo[1,5- a]pyridine-3-carboxamide
[0218] The title compound was prepared from 3-bromo-5-tert-butyl-4H-1,2,4-triazole in a similar fashion to Ex.1.1H NMR (400 MHz, DMSO-d6) δ 13.60 (s, 1H), 9.71 (s, 1H), 8.81 (dt, J = 7.0, 1.0 Hz, 1H), 8.74 (s, 1H), 8.20 (dt, J = 8.9, 1.3 Hz, 1H), 7.96 (d, J = 1.7 Hz, 1H), 7.74 (dd, J = 7.8, 1.8 Hz, 1H), 7.48 (ddd, J = 8.9, 6.8, 1.1 Hz, 1H), 7.32 (d, J = 7.9 Hz, 1H), 7.08 (td, J = 6.9, 1.5 Hz, 1H), 2.26 (s, 3H), 1.32 (s, 9H). ESI MS [M+H]+ for C21H23N6O, calcd 375.2, found 375.2. Example 5: N-[2-Methyl-5-[5-(trifluoromethyl)-4H-1,2,4-triazol-3-yl]phenyl]pyrazolo[1,5- a]pyridine-3-carboxamide
[0219] The title compound was prepared from 3-bromo-5-(trifluoromethyl)-4H-1,2,4-triazole in a similar fashion to Ex.1.1H NMR (400 MHz, Methanol-d4) δ 8.59 – 8.53 (m, 2H), 8.28 (dt, J = 8.9, 1.3 Hz, 1H), 7.99 (d, J = 1.9 Hz, 1H), 7.74 (dd, J = 7.9, 1.9 Hz, 1H), 7.47 – 7.39 (m, 2H), 7.03 (td, J = 6.9, 1.4 Hz, 1H), 2.39 (s, 3H). ESI MS [M+H]+ for C18H14F3N6O, calcd 387.1, found 387.1. Example 6: N-[4-Fluoro-2-methyl-5-[5-(trifluoromethyl)-4H-1,2,4-triazol-3- yl]phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide
[0220] The title compound was prepared from 3-bromo-5-(trifluoromethyl)-4H-1,2,4-triazole and 5-bromo-4-fluoro-2-methylaniline in a similar fashion to Ex. 1. 1H NMR (400 MHz, Methanol-d4) δ 8.57 – 8.53 (m, 2H), 8.27 (dt, J = 9.0, 1.2 Hz, 1H), 8.01 (d, J = 7.0 Hz, 1H), 7.43 (ddd, J = 9.0, 6.9, 1.2 Hz, 1H), 7.20 (d, J = 11.2 Hz, 1H), 7.03 (td, J = 6.9, 1.4 Hz, 1H), 2.38 (s, 3H). ESI MS [M+H]+ for C18H13F4N6O, calcd 405.1, found 405.1. Example 7: N-[5-(5-Cyclobutyl-4H-1,2,4-triazol-3-yl)-2-methylphenyl]pyrazolo[1,5- a]pyridine-3-carboxamide
[0221] The title compound was prepared from 3-bromo-5-cyclobutyl-4H-1,2,4-triazole in a similar fashion to Ex.1.1H NMR (400 MHz, DMSO-d6) δ 13.64 (s, 1H), 9.67 (s, 1H), 8.81 (d, J = 6.9 Hz, 1H), 8.74 (s, 1H), 8.21 (dt, J = 8.9, 1.3 Hz, 1H), 7.99 (s, 1H), 7.74 (d, J = 8.0 Hz, 1H), 7.52 – 7.45 (m, 1H), 7.30 (d, J = 8.0 Hz, 1H), 7.08 (t, J = 7.0 Hz, 1H), 3.72 – 3.52 (m, 1H), 2.34 – 2.23 (m, 7H), 2.05 – 1.80 (m, 2H). ESI MS [M+H]+ for C21H21N6O, calcd 373.2, found 373.2. Example 8: N-[5-(5-Cyclopentyl-4H-1,2,4-triazol-3-yl)-2-methylphenyl]pyrazolo[1,5- a]pyridine-3-carboxamide
[0222] The title compound was prepared from 3-bromo-5-cyclopentyl-4H-1,2,4-triazole in a similar fashion to Ex.1.1H NMR (400 MHz, DMSO-d6) δ 13.60 (s, 1H), 9.68 (s, 1H), 8.81 (dt, J = 6.9, 1.1 Hz, 1H), 8.74 (s, 1H), 8.20 (dt, J = 8.8, 1.3 Hz, 1H), 7.97 (d, J = 1.7 Hz, 1H), 7.73 (d, J = 8.2 Hz, 1H), 7.48 (ddd, J = 8.9, 6.8, 1.1 Hz, 1H), 7.30 (d, J = 8.0 Hz, 1H), 7.08 (td, J = 6.9, 1.4 Hz, 1H), 3.23 – 3.08 (m, 1H), 2.26 (s, 3H), 2.06 – 1.89 (m, 2H), 1.83 – 1.52 (m, 6H). ESI MS [M+H]+ for C22H23N6O, calcd 387.2, found 387.2. Example 9: N-[2-Methyl-5-[5-(2,2,2-trifluoroethyl)-4H-1,2,4-triazol-3- yl]phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide
[0223] The title compound was prepared from 3-chloro-5-(2,2,2-trifluoroethyl)-4H-1,2,4- triazole in a similar fashion to Ex. 1. 1H NMR (400 MHz, Methanol-d4) δ 8.59 – 8.51 (m, 2H), 8.31 – 8.26 (m, 1H), 7.95 (d, J = 1.9 Hz, 1H), 7.74 (dd, J = 8.0, 1.9 Hz, 1H), 7.48 – 7.36 (m, 2H), 7.07 – 6.99 (m, 1H), 3.64 (q, J = 10.4 Hz, 2H), 2.39 (s, 3H). ESI MS [M+H]+ for C19H16F3N6O, calcd 401.1, found 401.1. Example 10: N-[2-Methyl-5-[5-(2-methylpropyl)-4H-1,2,4-triazol-3-yl]phenyl]pyrazolo[1,5- a]pyridine-3-carboxamide
[0224] The title compound was prepared from 3-bromo-5-(2-methylpropyl)-4H-1,2,4-triazole in a similar fashion to Ex.1.1H NMR (400 MHz, Methanol-d4) δ 8.60 – 8.52 (m, 2H), 8.28 (ddt, J = 9.0, 2.2, 1.3 Hz, 1H), 7.97 (d, J = 1.9 Hz, 1H), 7.76 (dd, J = 7.9, 1.9 Hz, 1H), 7.46 – 7.38 (m, 2H), 7.03 (td, J = 7.0, 1.4 Hz, 1H), 2.70 (d, J = 7.3 Hz, 2H), 2.38 (s, 3H), 2.13 (dt, J = 13.6, 6.8 Hz, 1H), 1.01 – 0.96 (m, 6H). ESI MS [M+H]+ for C21H23N6O, calcd 375.2, found 375.2. Example 11: N-[5-(5-Cyclopropyl-4H-1,2,4-triazol-3-yl)-3-fluoro-2- methylphenyl]pyrazolo[1,5-a]pyridine-3-carboxamide
[0225] N-[5-[5-Cyclopropyl-4-(2-trimethylsilylethoxymethyl)-1,2,4-triazol-3-yl]-3-fluoro-2- methylphenyl]pyrazolo[1,5-a]pyridine-3-carboxamide (140 mg, 0.276 mmol, 1.0 equiv.), prepared from 5-bromo-3-fluoro-2-methylaniline in a similar fashion to Ex. 1, was dissolved in CH2Cl2 (0.4 mL) and treated with TFA (0.2 mL). The resulting mixture was stirred at 60 °C for 2 h, then concentrated in vacuo. The crude product was re-dissolved in 7M NH3 in MeOH (0.6 mL),
and the resulting mixture was stirred at 60 °C for 1 h, then concentrated. The crude residue was then recrystallized from CH2Cl2 to provide the title compound (100 mg, 99% yield).1H NMR (400 MHz, Methanol-d4) δ 8.70 (dt, J = 7.0, 1.0 Hz, 2H), 8.65 (s, 1H), 8.29 (dt, J = 8.9, 1.3 Hz, 2H), 7.91 (s, 1H), 7.63 (dd, J = 10.0, 1.7 Hz, 2H), 7.53 (ddd, J = 9.0, 6.9, 1.1 Hz, 2H), 7.13 (td, J = 6.9, 1.4 Hz, 2H), 2.30 (d, J = 2.1 Hz, 5H), 2.22 (ddd, J = 13.6, 8.9, 5.2 Hz, 1H), 1.31 – 1.23 (m, 3H), 1.20 – 1.15 (m, 3H). ESI MS [M+H]+ for C20H18FN6O, calcd 377.1, found 377.1. General procedure exemplified for Example 12: N-[5-(5-Cyclopropyl-4H-1,2,4-triazol-3-yl)- 4-fluoro-2-methylphenyl]pyrazolo[1,5-a]pyridine-3-carboxamide
[0226] Step a: A round-bottom flask was charged with methyl 5-amino-2-fluoro-4- methylbenzoate (1.83 g, 10 mmol, 1.0 equiv.) and EtOH (20 mL, 0.5 M). Hydrazine monohydrate (3.0 mL, 40 mmol, 4.0 equiv) was added via syringe and the reaction mixture was allowed to stir at 65 °C for 60 h. Upon complete conversion, as judged by LCMS analysis, the reaction mixture was cooled to rt. The resulting precipitate was collected by vacuum filtration and rinsed with MTBE. The filtrate was concentrated in vacuo and the resulting residue was taken up in a minimal amount of EtOH. The resulting precipitate was collected by vacuum filtration, rinsing with additional EtOH followed by MTBE. The combined solids were dried under vacuum to afford 5- amino-2-fluoro-4-methylbenzohydrazide (1.52 g, 83% yield), which was used directly in the next step without further purification.
[0227] Step b: A mixture of the product obtained in step a (183 mg, 1.0 mmol, 1.0 equiv.), cyclopropylcarbamidine HCl (145 mg, 1.2 mmol, 1.2 equiv.), iPr2NEt (0.34 mL, 2.0 mmol, 2.0 equiv.) in 2-methoxyethanol (10 mL, 0.1 M) was stirred at 100 °C for 14 h. Upon complete conversion, as judged by LCMS analysis, the reaction mixture was concentrated under vacuum. The crude product was purified by column chromatography (SiO2, 60 to 100% EtOAc in hexanes) to afford 5-(5-cyclopropyl-4H-1,2,4-triazol-3-yl)-4-fluoro-2-methylaniline (139 mg, 60% yield). [0228] Step c: A 40 mL vial was charged with triazolo[4,5-b]pyridin-3-yl pyrazolo[1,5- a]pyridine-3-carboxylate (138 mg, 0.49 mmol, 1.0 equiv., see Ex.1, step a), 5-(5-cyclopropyl-4H- 1,2,4-triazol-3-yl)-4-fluoro-2-methylaniline (114 mg, 0.49 mmol, 1.0 equiv.), and NMP (5.0 mL, 0.1 M). The resulting mixture was stirred at 120 °C for 3 h, then cooled to rt and diluted with water. The resulting precipitated solid was collected by vacuum filtration, rinsed with water, and dried in vacuo. The crude product was purified by column chromatography (SiO2, 60 to 100% EtOAc in hexanes) followed by reverse phase HPLC to afford the title compound (35 mg, 19% yield).1H NMR (400 MHz, DMSO-d6) δ 13.75 (s, 1H), 9.71 (s, 1H), 8.81 (dt, J = 7.0, 1.1 Hz, 1H), 8.71 (s, 1H), 8.18 (dt, J = 8.9, 1.3 Hz, 1H), 7.86 (d, J = 7.2 Hz, 1H), 7.48 (ddd, J = 8.9, 6.8, 1.1 Hz, 1H), 7.23 (d, J = 11.4 Hz, 1H), 7.08 (td, J = 6.9, 1.5 Hz, 1H), 3.13 (s, 1H), 2.25 (s, 2H), 2.05 – 2.00 (m, 1H), 1.00 - 0.95 (m, 2H), 0.90 – 0.86 (m, 2H). ESI MS [M+H]+ for C20H18FN6O, calcd 377.2, found 377.2. Example 13: N-[5-(5-Cyclopropyl-4H-1,2,4-triazol-3-yl)-4-fluoro-2-methylphenyl]-6- methoxypyrazolo[1,5-a]pyridine-3-carboxamide
[0229] The title compound was prepared from triazolo[4,5-b]pyridin-3-yl 6- methoxypyrazolo[1,5-a]pyridine-3-carboxylate in a similar fashion to Ex.12, which was prepared from 6-methoxypyrazolo[1,5-a]pyridine-3-carboxylic acid in a similar fashion to Ex.1, step a.1H NMR (400 MHz, DMSO-d6) δ 13.75 (s, 1H), 9.67 (s, 1H), 8.61 (s, 1H), 8.51 (dd, J = 2.2, 0.7 Hz, 1H), 8.07 (dd, J = 9.7, 0.7 Hz, 1H), 7.85 (d, J = 7.2 Hz, 1H), 7.28 (dd, J = 9.6, 2.2 Hz, 1H), 7.22 (d, J = 11.3 Hz, 1H), 3.83 (s, 3H), 2.25 (s, 3H), 2.05 – 2.01 (m, 1H), 0.99-0.95 (m, 2H), 0.90 – 0.86 (m, 2H). ESI MS [M+H]+ for C21H20FN6O2, calcd 407.2, found 407.2.
Example 14: 6-Chloro-N-[5-(5-cyclopropyl-4H-1,2,4-triazol-3-yl)-4-fluoro-2- methylphenyl]pyrazolo[1,5-a]pyridine-3-carboxamide
[0230] The title compound was prepared from triazolo[4,5-b]pyridin-3-yl 6-chloropyrazolo[1,5- a]pyridine-3-carboxylate in a similar fashion to Ex. 12, which was prepared from 6- chloropyrazolo[1,5-a]pyridine-3-carboxylic acid in a similar fashion to Ex. 1, step a. 1H NMR (400 MHz, DMSO-d6) δ 13.75 (s, 1H), 9.79 (s, 1H), 9.19 (s, 1H), 8.74 (s, 1H), 8.18 (dd, J = 9.5, 0.9 Hz, 1H), 7.85 (d, J = 7.2 Hz, 1H), 7.56 (dd, J = 9.5, 1.9 Hz, 1H), 7.20 (d, J = 11.5 Hz, 1H), 2.24 (s, 3H), 2.09 – 1.98 (m, 1H), 1.06 – 0.95 (m, 2H), 0.94 – 0.86 (m, 2H). ESI MS [M+H]+ for C20H17 ClFN6O, calcd 411.1, found 411.1. Example 15: N-[5-[5-(Cyclopropylmethyl)-4H-1,2,4-triazol-3-yl]-4-fluoro-2- methylphenyl]pyrazolo[1,5-a]pyridine-3-carboxamide
[0231] Step a: A 4 mL vial was charged with 5-amino-2-fluoro-4-methylbenzohydrazide (275 mg, 1.5 mmol, 1.5 mmol, 1.5 equiv.), cyclopropylacetonitrile (81 mg, 1.0 mmol, 1.0 equiv.), K2CO3 (138 mg, 1.0 mmol, 1.0 equiv) and n-BuOH (2 mL, 0.5 M). The resulting mixture was stirred at 130 °C for 20 h, then concentrated in vacuo. The crude product was purified by column chromatography (SiO2, 0 to 100% EtOAc in CH2Cl2) to afford 5-[5-(cyclopropylmethyl)-4H- 1,2,4-triazol-3-yl]-4-fluoro-2-methylaniline (124 mg, 50% yield).
[0232] Step b: A 40 mL vial was charged with the 5-[5-(cyclopropylmethyl)-4H-1,2,4-triazol- 3-yl]-4-fluoro-2-methylaniline obtained in step a (36 mg, 0.146 mmol, 1.0 equiv.), triazolo[4,5- b]pyridin-3-yl pyrazolo[1,5-a]pyridine-3-carboxylate (41 mg, 0.146 mmol, 1.0 equiv.), and NMP (0.150 mL, 1 M). The resulting mixture was stirred at 120 °C for 2 h. Upon complete conversion, as judged by LCMS analysis, the reaction mixture was cooled to rt and diluted with water. The resulting precipitated solid was collected by vacuum filtration, rinsed with water, and dried in vacuo. The crude product was purified by column chromatography (SiO2, 0 to 100% EtOAc in CH2Cl2) to afford the title compound (31.7 mg, 56% yield).1H NMR (400 MHz, DMSO-d6) δ 9.75 (s, 1H), 8.84 (dt, J = 7.0, 1.1 Hz, 1H), 8.75 (s, 1H), 8.22 (dt, J = 8.9, 1.3 Hz, 1H), 7.98 (d, J = 7.2 Hz, 1H), 7.52 (ddd, J = 8.9, 6.8, 1.1 Hz, 1H), 7.31 (d, J = 11.4 Hz, 1H), 7.11 (td, J = 6.9, 1.4 Hz, 1H), 2.71 (d, J = 7.0 Hz, 2H), 2.32 (s, 3H), 1.17 – 1.06 (m, 1H), 0.56 – 0.47 (m, 2H), 0.26 (dt, J = 6.1, 4.3 Hz, 2H). ESI MS [M+H]+ for C21H20FN6O, calcd 391.2, found 391.2. Example 16: N-[4-Fluoro-5-(5-methoxy-4H-1,2,4-triazol-3-yl)-2-methylphenyl]pyrazolo [1,5-a]pyridine-3-carboxamide
[0233] The title compound was prepared from 3-bromo-5-methoxy-4H-1,2,4-triazole and 5- bromo-4-fluoro-2-methylaniline in a similar fashion to Ex.1.1H NMR (400 MHz, Methanol-d4) δ 8.55 (d, J = 5.7 Hz, 2H), 8.27 (d, J = 8.9 Hz, 1H), 7.84 (d, J = 7.0 Hz, 1H), 7.47 – 7.39 (m, 1H), 7.14 (d, J = 11.4 Hz, 1H), 7.06 – 6.99 (m, 1H), 4.00 (s, 3H), 2.35 (s, 3H). ESI MS [M+H]+ for C18H16FN6O2, calcd 367.1, found 367.1. Example 17: 6-Fluoro-N-[4-fluoro-5-(5-methoxy-4H-1,2,4-triazol-3-yl)-2- methylphenyl]pyrazolo[1,5-a]pyridine-3-carboxamide
[0234] The title compound was prepared from 6-fluoropyrazolo[1,5-a]pyridine-3-carboxylic acid, 3-bromo-5-methoxy-4H-1,2,4-triazole and 5-bromo-4-fluoro-2-methylaniline in a similar
fashion to Ex.1.1H NMR (400 MHz, Methanol-d4) δ 8.56 (dd, J = 4.4, 2.1 Hz, 2H), 8.33 – 8.26 (m, 1H), 7.84 (d, J = 7.0 Hz, 1H), 7.39 (ddd, J = 9.9, 7.9, 2.2 Hz, 1H), 7.14 (d, J = 11.3 Hz, 1H), 4.00 (s, 3H), 2.35 (s, 3H). ESI MS [M+H]+ for C18H15F2N6O2, calcd 385.1, found 385.1. Example 18: N-[5-[5-(Dimethylamino)-4H-1,2,4-triazol-3-yl]-4-fluoro-2- methylphenyl]pyrazolo[1,5-a]pyridine-3-carboxamide
[0235] The title compound was prepared from 5-bromo-N,N-dimethyl-4H-1,2,4-triazol-3-amine and 5-bromo-4-fluoro-2-methylaniline in a similar fashion to Ex. 1. 1H NMR (400 MHz, Methanol-d4) δ 8.54 (d, J = 7.7 Hz, 2H), 8.27 (d, J = 8.8 Hz, 1H), 7.81 (dd, J = 7.1, 2.0 Hz, 1H), 7.45 – 7.40 (m, 1H), 7.09 (d, J = 11.4 Hz, 1H), 7.01 (t, J = 6.7 Hz, 1H), 3.02 (s, 6H), 2.33 (s, 3H). ESI MS [M+H]+ for C19H19FN7O, calcd 380.2, found 380.2. Example 19: N-[5-(5-Cyclopropyl-4H-1,2,4-triazol-3-yl)-4-fluoro-2-methylphenyl]-6- fluoropyrazolo[1,5-a]pyridine-3-carboxamide
[0236] The title compound was prepared from triazolo[4,5-b]pyridin-3-yl 6-fluoropyrazolo[1,5- a]pyridine-3-carboxylate in a similar fashion to Ex. 12, which was prepared from 6- fluoropyrazolo[1,5-a]pyridine-3-carboxylic acid in a similar fashion to Ex.1, step a.1H NMR (400 MHz, DMSO-d6) δ 13.78 (s, 1H), 9.82 (s, 1H), 9.19 (ddd, J = 4.7, 2.3, 0.8 Hz, 1H), 8.77 (s, 1H), 8.24 (ddd, J = 9.8, 5.9, 0.8 Hz, 1H), 7.90 (dd, J = 7.1, 1.7 Hz, 1H), 7.64 (dddd, J = 9.5, 8.2, 2.3, 1.1 Hz, 1H), 7.28 (d, J = 11.4 Hz, 1H), 2.29 (d, J = 1.9 Hz, 3H), 2.15 – 2.04 (m, 1H), 1.09 – 1.00 (m, 2H), 1.00 – 0.91 (m, 2H). ESI MS [M+H]+ for C20H17F2N6O, calcd 395.1, found 395.1. Example 20: N-[5-(5-Cyclopropyl-4H-1,2,4-triazol-3-yl)-4-fluoro-2-methylphenyl]-6- methylpyrazolo[1,5-a]pyridine-3-carboxamide
[0237] The title compound was prepared from triazolo[4,5-b]pyridin-3-yl 6- methylpyrazolo[1,5-a]pyridine-3-carboxylate in a similar fashion to Ex.12, which was prepared from 6-methylpyrazolo[1,5-a]pyridine-3-carboxylic acid in a similar fashion to Ex.1, step a.1H NMR (400 MHz, DMSO-d6) δ 13.78 (s, 1H), 9.68 (s, 1H), 8.67 (d, J = 6.0 Hz, 2H), 8.11 (d, J = 9.0 Hz, 1H), 7.88 (d, J = 7.5 Hz, 1H), 7.39 (d, J = 9.2 Hz, 1H), 7.22 (d, J = 11.4 Hz, 1H), 2.35 (s, 3H), 2.27 (s, 3H), 2.15 – 2.02 (m, 1H), 1.09 – 1.01 (m, 1H), 0.98 – 0.89 (m, 2H), 0.89 – 0.80 (m, 1H). ESI MS [M+H]+ for C21H20FN6O, calcd 391.2, found 391.1. Example 21: N-[5-(5-Cyclopropyl-4H-1,2,4-triazol-3-yl)-4-fluoro-2-methylphenyl]-5- fluoropyrazolo[1,5-a]pyridine-3-carboxamide
[0238] The title compound was prepared from triazolo[4,5-b]pyridin-3-yl 5-fluoropyrazolo[1,5- a]pyridine-3-carboxylate in a similar fashion to Ex. 12, which was prepared from 5- fluoropyrazolo[1,5-a]pyridine-3-carboxylic acid in a similar fashion to Ex.1, step a.1H NMR (400 MHz, Methanol-d4) δ 8.59 – 8.52 (m, 2H), 7.90 (dd, J = 8.9, 2.8 Hz, 1H), 7.82 (d, J = 6.9 Hz, 1H), 7.12 (d, J = 11.2 Hz, 1H), 6.89 (td, J = 7.2, 2.9 Hz, 1H), 2.33 (s, 3H), 2.10 – 2.00 (m, 1H), 1.03 (d, J = 7.5 Hz, 4H). ESI MS [M+H]+ for C20H17F2N6O, calcd 395.1, found 395.1. Example 22: N-[5-(5-Cyclopropyl-4H-1,2,4-triazol-3-yl)-4-fluoro-2-methylphenyl]-5- methylpyrazolo[1,5-a]pyridine-3-carboxamide
[0239] The title compound was prepared from triazolo[4,5-b]pyridin-3-yl 5- methylpyrazolo[1,5-a]pyridine-3-carboxylate in a similar fashion to Ex.12, which was prepared from 5-methylpyrazolo[1,5-a]pyridine-3-carboxylic acid in a similar fashion to Ex.1, step a.1H NMR (400 MHz, Methanol-d4) δ 8.48 (s, 1H), 8.41 (dd, J = 7.1, 0.9 Hz, 1H), 8.05 (dt, J = 2.1, 1.1 Hz, 1H), 7.83 (d, J = 7.0 Hz, 1H), 7.11 (d, J = 11.2 Hz, 1H), 6.85 (dd, J = 7.1, 1.9 Hz, 1H), 2.43 (d, J = 1.1 Hz, 3H), 2.34 (s, 3H), 2.09 – 2.01 (m, 1H), 1.02 (d, J = 4.9 Hz, 4H). ESI MS [M+H]+ for C21H20FN6O, calcd 391.2, found 391.2. Example 23: N-[5-(5-Cyclopropyl-4H-1,2,4-triazol-3-yl)-4-fluoro-2-methylphenyl]-5- methoxypyrazolo[1,5-a]pyridine-3-carboxamide
[0240] The title compound was prepared from triazolo[4,5-b]pyridin-3-yl 5- methoxypyrazolo[1,5-a]pyridine-3-carboxylate in a similar fashion to Ex.12, which was prepared from 5-methoxypyrazolo[1,5-a]pyridine-3-carboxylic acid in a similar fashion to Ex.1, step a.1H NMR (400 MHz, Methanol-d4) δ 8.44 (s, 1H), 8.34 (dd, J = 7.5, 0.6 Hz, 1H), 7.83 (d, J = 7.0 Hz, 1H), 7.58 (dd, J = 2.8, 0.6 Hz, 1H), 7.11 (d, J = 11.2 Hz, 1H), 6.66 (dd, J = 7.6, 2.8 Hz, 1H), 3.88 (s, 3H), 2.35 (s, 3H), 2.10 – 2.00 (m, 1H), 1.02 (s, 4H). ESI MS [M+H]+ for C21H20FN6O2, calcd 407.2, found 407.2. Example 24: N-[5-(5-Cyclopropyl-4H-1,2,4-triazol-3-yl)-4-fluoro-2-methylphenyl]-6- ethylpyrazolo[1,5-a]pyridine-3-carboxamide
[0241] Step a: To a solution of ethyl O-(2-mesitylenesulfonyl)acethydroxamate (2.00 g, 7.0 mmol) in dioxane (8 mL) was added 70% perchloric acid (1.2 mL) dropwise at 0 °C. The reaction was stirred at rt for 2 h, then the mixture was poured into ice/water (100 mL). The solution was stirred for 10 min until white solid precipitated. The solids were collected by filtration, washed with cold water (100 mL) and hexane (100 mL), and dried under vacuum to afford O- (mesitylenesulfonyl)hydroxylamine as a white solid (1.26 g, 83% yield). [0242] Step b: To a mixture of O-(mesitylenesulfonyl)hydroxylamine (1.08 g, 5.0 mmol) and 3-ethylpyridine (536 mg, 5.0 mmol) was added DCM (20 mL), then the solution was stirred at rt for 36 h. Upon complete conversion, as judged by LCMS analysis, the solution was concentrated and dried under vacuum to afford 2-mesitylenesulfonate-1-amino-3-ethyl-pyridinium as a white foam, which was taken crude into the next step. [0243] Step c: To a solution of 2-mesitylenesulfonate-1-amino-3-ethyl-pyridinium (5.0 mmol) in DMF (10 mL) at 0 °C, was added potassium carbonate (1.04 g, 7.5 mmol) and methyl propiolate (631 mg, 7.5 mmol). The reaction was stirred at rt for 18 h. The mixture was diluted with 100 mL
EtOAc and washed with water (20 mL). The aqueous layer was extracted with EtOAc (2 x 40 mL). The combined organic layer was dried over anhyd. MgSO4, filtered and concentrated. The crude material was purified by flash column chromatography (0-30% EtOAc in hexanes) to afford methyl 6-ethylpyrazolo[1,5-a]pyridine-3-carboxylate as a yellow solid (245 mg, 24% over 2 steps). [0244] Step d: To a solution of methyl 6-ethylpyrazolo[1,5-a]pyridine-3-carboxylate (79 mg, 0.383 mmol) in 1:1:1 THF/MeOH/water (1.2 mL) at 0 °C, was added LiOH.H2O (81 mg, 1.92 mmol). The reaction was stirred at 50 °C for 2 h. Upon cooling to rt, the mixture was concentrated to remove THF and MeOH. The resultant mixture was adjusted to pH ~2 by addition of 2 M HCl(aq). The mixture was diluted with EtOAc (10 mL) and washed with water (5 mL). The aqueous layer was extracted with EtOAc (2 x 10 mL). The combined organic layer was dried over anhyd. MgSO4, filtered and concentrated to afford 6-ethylpyrazolo[1,5-a]pyridine-3-carboxylic acid as an off-white solid (72 mg, 99% yield). [0245] Step e: The desired product was prepared in a similar manner to Example 1, step a. [0246] Step f: The desired product was prepared in a similar manner to Example 12, step c.1H NMR (400 MHz, DMSO-d6) δ 13.74 (s, 1H), 9.80 (s, 1H), 8.64 (d, J = 6.7 Hz, 1H), 8.47 (s, 1H), 7.87 (d, J = 7.3 Hz, 1H), 7.37 – 7.15 (m, 2H), 6.99 (t, J = 6.9 Hz, 1H), 3.10 (q, J = 7.6 Hz, 2H), 2.26 (s, 3H), 2.12 – 1.96 (m, 1H), 1.12 (t, J = 7.5 Hz, 3H), 1.06 – 0.76 (m, 4H). ESI MS [M+H]+ for C21H22FN6O, calcd 405.2, found 405.2. Example 25: 6-Cyclopropyl-N-[5-(5-cyclopropyl-4H-1,2,4-triazol-3-yl)-4-fluoro-2- methylphenyl]pyrazolo[1,5-a]pyridine-3-carboxamide
[0247] The title compound was prepared from 3-cyclopropylpyridine in a similar fashion to Ex. 24.1H NMR (400 MHz, DMSO-d6) δ 13.74 (s, 1H), 9.64 (s, 1H), 8.63 (d, J = 5.1 Hz, 2H), 8.06 (d, J = 9.2 Hz, 1H), 7.85 (d, J = 6.8 Hz, 1H), 7.42 – 7.05 (m, 2H), 2.24 (s, 3H), 2.01 (ddd, J = 13.4, 8.5, 4.9 Hz, 2H), 1.05 – 0.73 (m, 8H). ESI MS [M+H]+ for C23H22FN6O, calcd 417.2, found 417.2.
Example 26: N-[5-(5-Cyclopropyl-4H-1,2,4-triazol-3-yl)-4-fluoro-2-methylphenyl]-6- ethoxypyrazolo[1,5-a]pyridine-3-carboxamide
[0248] Step a: To a mixture of 6-bromopyrazolo[1,5-a]pyridine (985 mg, 5.0 mmol), ethanol (0.58 mL, 10.0 mmol), palladium acetate (57 mg, 0.25 mmol), tBuBrettPhos (243 mg, 0.5 mmol) and cesium carbonate (3.26 g, 10.0 mmol) was added degassed toluene (10 mL). The resulting mixture was stirred at 90° C for 8 h. Upon cooling, EtOAc (100 mL) was added and the mixture was filtered through Celite®. The filtrate was concentrated and purified by flash column chromatography (0-20% EtOAc in hexanes) to afford 6-ethoxypyrazolo[l,5-a]pyridine (747 mg, 92%) as a colorless oil. [0249] Step b: To a solution of 6-ethoxypyrazolo[l,5-a]pyridine (747 mg, 4.6 mmol) in DMF (9.2 mL) at 0 °C, was added POCl3 (1.29 mL, 13.8 mmol). The reaction was stirred at rt for 2 h. The solution was cooled to 0 °C and adjusted to pH ~14 by addition of 1 M NaOH(aq). The mixture was diluted with MeOH (20 mL) and refluxed for an additional 2 h. Upon cooling to rt, the mixture was concentrated to remove MeOH and extracted with CH2Cl2 (2 x 40 mL). The combined organic
layer was dried over anhyd. MgSO4, filtered and concentrated. The crude material was purified by flash column chromatography (0-50% EtOAc in hexanes) to afford 6-ethoxypyrazolo[1,5- a]pyridine-3-carboxaldehyde as a yellow solid (700 mg, 80%). [0250] Step c: To a solution of 6-ethoxypyrazolo[1,5-a]pyridine-3-carboxaldehyde (700 mg, 3.68 mmol) in water (20 mL), was added NaH2PO4 (1.33 g, 11.04 mmol). After 5 min, 1:1 THF/tBuOH (40 mL), 2-methyl-2-butene (2.34 mL, 22.08 mmol) and sodium chlorite (1.34 g, 14.72 mmol) were added sequentially. The reaction was stirred at rt for 24 h. The mixture was diluted with EtOAc (100 mL) and washed with water (50 mL) and brine (20 mL). The aqueous layer was extracted with EtOAc (2 x 50 mL). The combined organic layer was dried over anhyd. MgSO4, filtered and concentrated to afford 6-ethoxypyrazolo[1,5-a]pyridine-3-carboxylic acid as an off-white solid, which was used in the next step without further purification. [0251] Step d: The desired product was prepared in a similar manner to Example 1, step a. [0252] Step e: The desired product was prepared in a similar manner to Example 12, step c.1H NMR (400 MHz, DMSO-d6) δ 13.74 (s, 1H), 9.65 (s, 1H), 8.60 (s, 1H), 8.49 (dd, J = 2.2, 0.8 Hz, 1H), 8.06 (dd, J = 9.6, 0.7 Hz, 1H), 7.85 (d, J = 7.1 Hz, 1H), 7.39 – 7.12 (m, 2H), 4.08 (q, J = 7.0 Hz, 2H), 2.24 (s, 3H), 2.04 (s, 1H), 1.34 (t, J = 7.0 Hz, 3H), 0.96 (d, J = 40.3 Hz, 4H). ESI MS [M+H]+ for C22H22FN6O2, calcd 421.2, found 421.2. Example 27: N-[4-Fluoro-2-methyl-5-(5-methyl-4H-1,2,4-triazol-3-yl)phenyl]pyrazolo[1,5- a]pyridine-3-carboxamide
[0253] The title compound was prepared in a similar fashion to Ex. 12 using acetamidine hydrochloride.1H NMR (400 MHz, Methanol-d4) δ 8.58 – 8.52 (m, 2H), 8.27 (dt, J = 8.9, 1.2 Hz, 1H), 7.85 (d, J = 7.0 Hz, 1H), 7.42 (ddd, J = 9.0, 6.8, 1.1 Hz, 1H), 7.12 (d, J = 11.3 Hz, 1H), 7.02 (td, J = 6.9, 1.4 Hz, 1H), 2.45 (s, 3H), 2.35 (s, 3H). ESI MS [M+H]+ for C18H16FN6O, calcd 351.1, found 351.1.
Example 28: N-[4-Fluoro-2-methyl-5-(5-methyl-4H-1,2,4-triazol-3-yl)phenyl]-6- methoxypyrazolo[1,5-a]pyridine-3-carboxamide
[0254] The title compound was prepared in a similar fashion to Ex. 27 from triazolo[4,5- b]pyridin-3-yl 6-methoxypyrazolo[1,5-a]pyridine-3-carboxylate, which was prepared from 6- methoxypyrazolo[1,5-a]pyridine-3-carboxylic acid in a similar fashion to Ex.1, step a.1H NMR (400 MHz, Methanol-d4) δ 8.43 (s, 1H), 8.16 – 8.11 (m, 2H), 7.83 (d, J = 7.0 Hz, 1H), 7.20 (dd, J = 9.8, 2.1 Hz, 1H), 7.13 (d, J = 11.3 Hz, 1H), 3.87 (s, 3H), 2.45 (s, 3H), 2.34 (s, 3H). ESI MS [M+H]+ for C19H18FN6O2, calcd 381.1, found 381.1. Example 29: 6-Fluoro-N-[4-fluoro-2-methyl-5-[5-(oxolan-2-yl)-4H-1,2,4-triazol-3- yl]phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide
[0255] The title compound was prepared in a similar fashion to Ex. 12 from tetrahydro-2- furancarboximidamide and triazolo[4,5-b]pyridin-3-yl 6-fluoropyrazolo[1,5-a]pyridine-3- carboxylate, which was prepared from 6-fluoropyrazolo[1,5-a]pyridine-3-carboxylic acid in a similar fashion to Ex.1, step a.1H NMR (400 MHz, DMSO-d6) δ 9.82 (s, 1H), 9.22 – 9.16 (m, 1H), 8.78 (s, 1H), 8.25 (dd, J = 9.8, 5.8 Hz, 1H), 7.97 (d, J = 7.2 Hz, 1H), 7.64 (ddd, J = 10.1, 8.2, 2.2 Hz, 1H), 7.30 (d, J = 11.4 Hz, 1H), 5.03 (t, J = 6.7 Hz, 1H), 3.99 – 3.87 (m, 1H), 3.85 – 3.78 (m, 1H), 2.30 (s, 3H), 2.30 – 2.22 (m, 1H), 2.19 – 2.06 (m, 1H), 2.06 – 1.87 (m, 2H). ESI MS [M+H]+ for C21H19F2N6O2, calcd 425.1, found 425.1. Example 30: N-[4-Fluoro-2-methyl-5-[5-(oxan-4-yl)-4H-1,2,4-triazol-3- yl]phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide
[0256] The title compound was prepared from tetrahydropyran-4-carboxamidine hydrochloride in a similar fashion to Ex.12.1H NMR (400 MHz, DMSO-d6) δ 13.83 (s, 1H), 9.71 (s, 1H), 8.81 (dt, J = 6.9, 1.1 Hz, 1H), 8.72 (s, 1H), 8.19 (dt, J = 8.9, 1.3 Hz, 1H), 7.93 (d, J = 7.2 Hz, 1H), 7.48 (ddd, J = 8.9, 6.8, 1.1 Hz, 1H), 7.36 – 7.17 (m, 1H), 7.08 (td, J = 6.9, 1.5 Hz, 1H), 3.92 – 3.83 (m, 2H), 3.42 (td, J = 11.5, 2.3 Hz, 2H), 3.13 (d, J = 5.2 Hz, 1H), 2.27 (s, 3H), 1.92 – 1.81 (m, 2H), 1.81 – 1.67 (m, 2H). ESI MS [M+H]+ for C22H22FN6O2, calcd 421.2, found 421.2. Example 31: 6-Fluoro-N-[4-fluoro-2-methyl-5-(5-morpholin-4-yl-4H-1,2,4-triazol-3- yl)phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide
[0257] The title compound was prepared in a similar fashion to Ex. 12 from morpholine-4- carboximidamide hydrobromide and triazolo[4,5-b]pyridin-3-yl 6-fluoropyrazolo[1,5-a]pyridine- 3-carboxylate, which was prepared from 6-fluoropyrazolo[1,5-a]pyridine-3-carboxylic acid in a similar fashion to Ex.1, step a.1H NMR (400 MHz, CD3OD) δ 8.80 (ddd, J = 4.2, 2.2, 0.8 Hz, 1H), 8.62 (s, 1H), 8.30 (ddd, J = 9.8, 5.7, 0.8 Hz, 1H), 7.93 (brs, 1H), 7.52 (ddd, J = 10.1, 8.1, 2.2 Hz, 1H), 7.23 (brs, 1H), 3.84 – 3.74 (m, 4H), 3.45 – 3.39 (m, 4H), 2.37 (s, 3H). ESI MS [M+H]+ for C21H20F2N7O2, calcd 440.2, found 440.2. Example 32: N-[4-Fluoro-2-methyl-5-(5-propyl-4H-1,2,4-triazol-3-yl)phenyl]pyrazolo[1,5- a]pyridine-3-carboxamide
[0258] The title compound was prepared from butyramidine hydrochloride in a similar fashion to Ex.12.1H NMR (400 MHz, DMSO-d6) δ 13.78 (brs, 1H), 9.72 (brs, 1H), 8.84 (d, J = 6.9 Hz,
1H), 8.75 (s, 1H), 8.24 – 8.20 (m, 1H), 7.97 (d, J = 7.3 Hz, 1H), 7.55 – 7.47 (m, 1H), 7.27 – 7.21 (m, 1H), 7.15 – 7.08 (m, 1H), 2.73 (t, J = 7.6 Hz, 2H), 2.29 (s, 3H), 1.79 – 1.66 (m, 2H), 0.93 (t, J = 7.4 Hz, 3H). ESI MS [M+H]+ for C20H20FN6O, calcd 379.2, found 379.2. Example 33: N-[5-[5-(Difluoromethyl)-4H-1,2,4-triazol-3-yl]-4-fluoro-2- methylphenyl]pyrazolo[1,5-a]pyridine-3-carboxamide
[0259] The title compound was prepared from 5-[5-(difluoromethyl)-4H-1,2,4-triazol-3-yl]-4- fluoro-2-methylaniline in a similar fashion to Ex. 12. 1H NMR (400 MHz, Methanol-d4) δ 8.56 (dd, J = 4.4, 2.1 Hz, 2H), 8.33 – 8.26 (m, 1H), 7.84 (d, J = 7.0 Hz, 1H), 7.39 (ddd, J = 9.9, 7.9, 2.2 Hz, 1H), 7.14 (d, J = 11.3 Hz, 1H), 4.00 (s, 3H), 2.35 (s, 3H). ESI MS [M+H]+ for C18H14F3N6O, calcd 387.1, found 387.1. Example 34: N-[5-[5-(Difluoromethyl)-4H-1,2,4-triazol-3-yl]-4-fluoro-2-methylphenyl]-6- methoxypyrazolo[1,5-a]pyridine-3-carboxamide
[0260] The title compound was prepared from triazolo[4,5-b]pyridin-3-yl 6- methoxypyrazolo[1,5-a]pyridine-3-carboxylate (which was prepared from 6- methoxypyrazolo[1,5-a]pyridine-3-carboxylic acid in a similar fashion to Ex.1 step a) and 5-[5-
(difluoromethyl)-4H-1,2,4-triazol-3-yl]-4-fluoro-2-methylaniline in a similar fashion to Ex.33.1H NMR (400 MHz, Methanol-d4) δ 8.45 (s, 1H), 8.14 (d, J = 8.1 Hz, 2H), 7.95 (d, J = 7.0 Hz, 1H), 7.24 – 7.16 (m, 2H), 6.77 (t, J = 53.5 Hz, 1H), 3.88 (s, 3H), 2.37 (s, 3H). ESI MS [M+H]+ for C19H16F3N6O2, calcd 417.1, found 417.1. Example 35: N-[5-[5-(Difluoromethyl)-4H-1,2,4-triazol-3-yl]-4-fluoro-2-methylphenyl]-6- fluoropyrazolo[1,5-a]pyridine-3-carboxamide
[0261] The title compound was prepared from triazolo[4,5-b]pyridin-3-yl 6-fluoropyrazolo[1,5- a]pyridine-3-carboxylate (which was prepared from 6-fluoropyrazolo[1,5-a]pyridine-3-carboxylic acid in a similar fashion to Ex. 1, step a) and 5-[5-(difluoromethyl)-4H-1,2,4-triazol-3-yl]-4- fluoro-2-methylaniline in a similar fashion to Ex. 33. 1H NMR (400 MHz, Methanol-d4) δ 8.57 (ddd, J = 4.0, 2.2, 0.8 Hz, 2H), 8.29 (ddd, J = 9.8, 5.7, 0.8 Hz, 1H), 7.97 (d, J = 7.0 Hz, 1H), 7.39 (ddd, J = 9.9, 7.9, 2.2 Hz, 1H), 7.19 (d, J = 11.3 Hz, 1H), 6.77 (t, J = 53.5 Hz, 1H), 2.37 (s, 3H). ESI MS [M+H]+ for C18H13F4N6O, calcd 405.1, found 405.1. Example 36: N-[5-(5-Cyclopropyl-4H-1,2,4-triazol-3-yl)-3-fluoro-2-methylphenyl]-6- methoxypyrazolo[1,5-a]pyridine-3-carboxamide
[0262] The title compound was prepared in a similar fashion to Ex. 12 using triazolo[4,5- b]pyridin-3-yl 6-methoxypyrazolo[1,5-a]pyridine-3-carboxylate (which was prepared from 6- methoxypyrazolo[1,5-a]pyridine-3-carboxylic acid in a similar fashion to Ex. 1, step a) and 5- bromo-3-fluoro-2-methylaniline.1H NMR (400 MHz, Methanol-d4) δ 8.53 (s, 1H), 8.34 (dd, J = 2.2, 0.7 Hz, 1H), 8.16 (dd, J = 9.7, 0.7 Hz, 1H), 7.83 (s, 1H), 7.60 (dd, J = 10.1, 1.6 Hz, 1H), 7.30 (dd, J = 9.7, 2.2 Hz, 1H), 3.91 (s, 3H), 2.26 (d, J = 2.1 Hz, 3H), 2.15 – 2.05 (m, 1H), 1.16 – 1.08 (m, 2H), 1.08 – 1.02 (m, 2H). ESI MS [M+H]+ for C21H20FN6O2, calcd 407.2, found 407.1.
Example 37: N-[5-(5-Cyclopropyl-4H-1,2,4-triazol-3-yl)-3-fluoro-2-methylphenyl]-6- fluoropyrazolo[1,5-a]pyridine-3-carboxamide
[0263] The title compound was prepared in a similar fashion to Ex. 12 using triazolo[4,5- b]pyridin-3-yl 6-fluoropyrazolo[1,5-a]pyridine-3-carboxylate (which was prepared from 6- fluoroypyrazolo[1,5-a]pyridine-3-carboxylic acid in a similar fashion to Ex. 1, step a) and 5- bromo-3-fluoro-2-methylaniline. 1H NMR (400 MHz, Methanol-d4) δ 8.83 – 8.80 (m, 1H), 8.64 (s, 1H), 8.31 (ddd, J = 9.8, 5.7, 0.8 Hz, 1H), 7.83 (s, 1H), 7.62 (d, J = 10.3 Hz, 1H), 7.53 (ddd, J = 10.1, 8.2, 2.3 Hz, 1H), 2.25 (s, 3H), 2.15 – 2.06 (m, 1H), 1.13 (d, J = 8.2 Hz, 2H), 1.10 – 0.95 (m, 2H). ESI MS [M+H]+ for C20H17F2N6O, calcd 395.1, found 395.1. Example 38: 6-Chloro-N-[5-[5-(cyclopropylmethyl)-4H-1,2,4-triazol-3-yl]-4-fluoro-2- methylphenyl]pyrazolo[1,5-a]pyridine-3-carboxamide
[0264] The title compound was prepared in a similar fashion to Ex. 15 using triazolo[4,5- b]pyridin-3-yl 6-chloropyrazolo[1,5-a]pyridine-3-carboxylate, which was prepared from 6- chloropyrazolo[1,5-a]pyridine-3-carboxylic acid in a similar fashion to Ex. 1, step a. 1H NMR (400 MHz, DMSO-d6) δ 13.83 (s, 1H), 9.80 (s, 1H), 9.20 (ddd, J = 4.6, 2.2, 0.8 Hz, 1H), 8.77 (s, 1H), 8.25 (ddd, J = 9.8, 5.9, 0.8 Hz, 1H), 7.97 (d, J = 7.2 Hz, 1H), 7.64 (ddd, J = 9.8, 8.3, 2.2 Hz, 1H), 7.29 (d, J = 11.4 Hz, 1H), 2.71 – 2.63 (m, 2H), 2.30 (s, 3H), 1.10 (s, 1H), 0.54 – 0.45 (m, 2H), 0.29 – 0.20 (m, 2H). ESI MS [M+H]+ for C21H19ClFN6O, calcd 425.1, found 425.2. Example 39: N-[5-[5-(Cyclopropylmethyl)-4H-1,2,4-triazol-3-yl]-4-fluoro-2-methylphenyl]- 6-fluoropyrazolo[1,5-a]pyridine-3-carboxamide
[0265] The title compound was prepared in a similar fashion to Ex. 15 using triazolo[4,5- b]pyridin-3-yl 6-fluoropyrazolo[1,5-a]pyridine-3-carboxylate, which was prepared from 6- fluoropyrazolo[1,5-a]pyridine-3-carboxylic acid in a similar fashion to Ex.1, step a.1H NMR (400 MHz, Methanol-d4) δ 8.81 (s, 1H), 8.63 (s, 1H), 8.30 (ddd, J = 9.8, 5.7, 0.8 Hz, 1H), 8.02 – 7.88 (m, 1H), 7.52 (t, J = 9.2 Hz, 1H), 7.34 – 7.18 (m, 1H), 2.80 – 2.64 (m, 2H), 2.38 (s, 3H), 1.25 – 1.11 (m, 1H), 0.65 – 0.49 (m, 2H), 0.36 – 0.23 (m, 2H). ESI MS [M+H]+ for C21H19F2N6O, calcd 409.2, found 409.1. Example 40: 6-Chloro-N-[5-(5-ethyl-4H-1,2,4-triazol-3-yl)-4-fluoro-2- methylphenyl]pyrazolo[1,5-a]pyridine-3-carboxamide
[0266] The title compound was prepared from triazolo[4,5-b]pyridin-3-yl 6-chloropyrazolo[1,5- a]pyridine-3-carboxylate (which was prepared from 6-chloropyrazolo[1,5-a]pyridine-3- carboxylic acid in a similar fashion to Ex. 1, step a) and propionimidamide hydrochloride in a similar fashion to Ex.12.1H NMR (400 MHz, Methanol-d4) δ 8.85 (dd, J = 1.8, 0.8 Hz, 1H), 8.60 (s, 1H), 8.24 (dd, J = 9.5, 0.9 Hz, 1H), 7.89 (s, 1H), 7.50 (dd, J = 9.5, 1.8 Hz, 1H), 7.19 (d, J = 11.6 Hz, 1H), 2.85 (d, J = 8.1 Hz, 2H), 2.35 (s, 3H), 1.34 (t, J = 7.8 Hz, 3H). ESI MS [M+H]+ for C19H17ClFN6O, calcd 399.1, found 399.1. Example 41: N-[5-(5-Ethyl-4H-1,2,4-triazol-3-yl)-4-fluoro-2-methylphenyl]-6- fluoropyrazolo[1,5-a]pyridine-3-carboxamide
[0267] The title compound was prepared from triazolo[4,5-b]pyridin-3-yl 6-fluoropyrazolo[1,5- a]pyridine-3-carboxylate (which was prepared from 6-fluoropyrazolo[1,5-a]pyridine-3-carboxylic acid in a similar fashion to Ex.1, step a) and propionimidamide hydrochloride in a similar fashion to Ex.12.1H NMR (400 MHz, Methanol-d4) δ 8.79 (dd, J = 4.2, 2.2 Hz, 1H), 8.60 (s, 1H), 8.28 (ddd, J = 9.8, 5.7, 0.8 Hz, 1H), 7.99 – 7.84 (m, 1H), 7.54 – 7.45 (m, 1H), 7.23 (dd, J = 35.8, 11.7 Hz, 1H), 2.85 (d, J = 7.9 Hz, 2H), 2.35(s, 3H), 1.35 (t, J = 7.6 Hz, 2H). ESI MS [M+H]+ for C19H17F2N6O, calcd 383.1, found 383.1. Example 42: N-[5-(5-Ethyl-4H-1,2,4-triazol-3-yl)-4-fluoro-2-methylphenyl]-6- methoxypyrazolo[1,5-a]pyridine-3-carboxamide
[0268] The title compound was prepared from triazolo[4,5-b]pyridin-3-yl 6- methoxypyrazolo[1,5-a]pyridine-3-carboxylate (which was prepared from
- methoxypyrazolo[1,5-a]pyridine-3-carboxylic acid in a similar fashion to Ex. 1, step a) and propionimidamide hydrochloride in a similar fashion to Ex.12.1H NMR (400 MHz, Methanol-d4) δ 8.51 (d, J = 14.5 Hz, 1H), 8.31 (dd, J = 2.2, 0.8 Hz, 1H), 8.13 (dd, J = 9.7, 0.7 Hz, 1H), 7.90 (d, J = 7.0 Hz, 1H), 7.33 – 7.16 (m, 2H), 3.89 (s, 3H), 2.82 (d, J = 7.6 Hz, 2H), 2.35 (s, 3H), 1.34 (t, J = 7.6 Hz, 3H). ESI MS [M+H]+ for C20H20FN6O2, calcd 395.2, found 395.1. Example 43: 6-Fluoro-N-[4-fluoro-2-methyl-5-(5-propan-2-yl-4H-1,2,4-triazol-3- yl)phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide
[0269] The title compound was prepared in a similar fashion to Ex.12 using isobutyrimidamide hydrochloride and triazolo[4,5-b]pyridin-3-yl 6-fluoropyrazolo[1,5-a]pyridine-3-carboxylate, which was prepared from 6-fluoropyrazolo[1,5-a]pyridine-3-carboxylic acid in a similar fashion to Ex.1, step a.1H NMR (400 MHz, DMSO-d6) δ 13.80 – 13.67 (br., 1H), 9.78 (s, 1H), 9.16 (ddd, J = 4.6, 2.2, 0.8 Hz, 1H), 8.74 (s, 1H), 8.22 (ddd, J = 9.8, 5.9, 0.8 Hz, 1H), 7.91 (d, J = 7.1 Hz,
1H), 7.61 (ddd, J = 9.7, 8.2, 2.2 Hz, 1H), 7.33 – 7.13 (m, 1H), 3.06 (m, 1H), 2.26 (s, 3H), 1.26 (d, J = 7.0 Hz, 6H). ESI MS [M+H]+ for C20H19F2N6O, calcd 397.2, found 397.2. Example 44: N-[4-Fluoro-2-methyl-5-(5-propan-2-yl-4H-1,2,4-triazol-3-yl)phenyl]-6- methoxypyrazolo[1,5-a]pyridine-3-carboxamide
[0270] The title compound was prepared in a similar fashion to Ex.12 using isobutyrimidamide hydrochloride and triazolo[4,5-b]pyridin-3-yl 6-methoxypyrazolo[1,5-a]pyridine-3-carboxylate, which was prepared from 6-methoxypyrazolo[1,5-a]pyridine-3-carboxylic acid in a similar fashion to Ex.1, step a.1H NMR (400 MHz, 1H NMR (400 MHz, DMSO-d6) δ 9.67 (s, 1H), 8.61 (s, 1H), 8.51 (dd, J = 2.2, 0.8 Hz, 1H), 8.07 (dd, J = 9.7, 0.8 Hz, 1H), 7.91 (d, J = 7.1 Hz, 1H), 7.32 – 7.19 (m, 2H), 3.83 (d, J = 0.9 Hz, 3H), 3.05 (s, 1H), 2.26 (s, 3H), 1.26 (d, J = 7.0 Hz, 6H). ESI MS [M+H]+ for C21H22FN6O2, calcd 409.2, found 409.1. Example 45: 6-Chloro-N-[4-fluoro-2-methyl-5-(5-propan-2-yl-4H-1,2,4-triazol-3- yl)phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide
[0271] The title compound was prepared in a similar fashion to Ex.12 using isobutyrimidamide hydrochloride and triazolo[4,5-b]pyridin-3-yl 6-chloropyrazolo[1,5-a]pyridine-3-carboxylate, which was prepared from 6-chloropyrazolo[1,5-a]pyridine-3-carboxylic acid in a similar fashion to Ex.1, step a.1H NMR (400 MHz, DMSO-d6) δ 13.72 (br., 1H), 9.81 (s, 1H), 9.19 (dd, J = 1.9, 0.9 Hz, 1H), 8.75 (s, 1H), 8.19 (dd, J = 9.5, 0.9 Hz, 1H), 7.91 (d, J = 7.1 Hz, 1H), 7.56 (dd, J = 9.5, 1.8 Hz, 1H), 7.24 (d, J = 11.4 Hz, 1H), 3.06 (p, J = 6.9 Hz, 1H), 2.26 (s, 3H), 1.26 (d, J = 7.0 Hz, 6H). ESI MS [M+H]+ for C20H19ClFN6O, calcd 413.1, found 413.1. Example 46: N-[2-Chloro-5-(5-cyclopropyl-4H-1,2,4-triazol-3-yl)-4- fluorophenyl]pyrazolo[1,5-a]pyridine-3-carboxamide
[0272] Step a: A round-bottom flask was charged with methyl 4-chloro-2-fluoro-5- nitrobenzoate (1.00 g, 4.28 mmol) and Fe (500 mg, 8.95 mmol, 2.1 equiv.) in AcOH (5 mL). The resulting mixture was then stirred at reflux under a nitrogen atmosphere for 2 h. Upon complete conversion, as judged by LCMS analysis, the reaction mixture was cooled to rt, diluted with EtOAc, filtered, and concentrated in vacuo. The residue was diluted with EtOAc and sat. aq. NaHCO3. The organic phase was separated, dried, and concentrated in vacuo. Purification by column chromatography (SiO2, 10 to 90% EtOAc in hexanes) provided methyl 5-amino-4-chloro- 2-fluorobenzoate as a clear oil (800 mg, 90% yield). [0273] Steps b-d: The title compound was prepared from methyl 5-amino-4-chloro-2- fluorobenzoate obtained in step a in a similar fashion to Ex.12.1H NMR (400 MHz, DMSO-d6) δ 13.90 (s, 1H), 9.89 (s, 1H), 8.82 (dt, J = 7.0, 1.1 Hz, 1H), 8.74 (s, 1H), 8.18 (dt, J = 8.9, 1.3 Hz, 1H), 8.10 (d, J = 7.5 Hz, 1H), 7.64 (d, J = 10.2 Hz, 1H), 7.51 (ddd, J = 8.9, 6.8, 1.1 Hz, 1H), 7.10 (td, J = 6.9, 1.4 Hz, 1H), 2.06 (m, 1H), 1.06 – 0.96 (m, 2H), 0.96 – 0.86 (m, 2H). ESI MS [M+H]+ for C19H15ClFN6O, calcd 397.1, found 397.1. Example 47: N-[2-Chloro-5-(5-cyclopropyl-4H-1,2,4-triazol-3-yl)-4-fluorophenyl]-6- fluoropyrazolo[1,5-a]pyridine-3-carboxamide
[0274] The title compound was prepared in a similar fashion to Ex. 46 using triazolo[4,5- b]pyridin-3-yl 6-fluoropyrazolo[1,5-a]pyridine-3-carboxylate, which was prepared from 6- fluoropyrazolo[1,5-a]pyridine-3-carboxylic acid in a similar fashion to Ex.1, step a.1H NMR (400 MHz, DMSO-d6) δ 13.90 (s, 1H), 9.97 (s, 1H), 9.18 (ddd, J = 4.6, 2.2, 0.8 Hz, 1H), 8.76 (s, 1H), 8.21 (ddd, J = 9.8, 5.9, 0.8 Hz, 1H), 8.08 (d, J = 7.4 Hz, 1H), 7.63 (ddd, J = 9.7, 8.2, 2.2 Hz, 1H), 2.06 (m, 1H), 1.06 – 0.96 (m, 2H), 0.96 – 0.81 (m, 2H). ESI MS [M+H]+ for C19H14ClF2N6O, calcd 415.1, found 415.1. Example 48: N-[2-Chloro-5-(5-cyclopropyl-4H-1,2,4-triazol-3-yl)-4-fluorophenyl]-6- methoxypyrazolo[1,5-a]pyridine-3-carboxamide
[0275] The title compound was prepared in a similar fashion to Ex. 46 using triazolo[4,5- b]pyridin-3-yl 6-methoxypyrazolo[1,5-a]pyridine-3-carboxylate, which was prepared from 6- methoxypyrazolo[1,5-a]pyridine-3-carboxylic acid in a similar fashion to Ex.1, step a.1H NMR (400 MHz, DMSO-d6) δ 13.90 (s, 1H), 9.84 (s, 1H), 8.64 (s, 1H), 8.53 (dd, J = 2.2, 0.7 Hz, 1H), 8.15 – 8.00 (m, 2H), 7.64 (d, J = 10.2 Hz, 1H), 7.31 (dd, J = 9.6, 2.2 Hz, 1H), 3.83 (s, 3H), 2.06 (m, 1H), 1.06 – 0.97 (m, 2H), 0.94 – 0.86 (m, 2H). ESI MS [M+H]+ for C20H17ClFN6O2, calcd 427.1, found 427.1. Example 49: N-[2-Chloro-5-(5-cyclopropyl-4H-1,2,4-triazol-3-yl)-3- fluorophenyl]pyrazolo[1,5-a]pyridine-3-carboxamide
[0276] Step a: A round-bottom flask was charged with methyl 3-bromo-4-chloro-5- fluorobenzoate (1.07 g, 4.0 mmol, 1.0 equiv.), benzophenone imine (869 mg, 4.8 mmol, 1.2 equiv.), Pd2(dba)3 (366 mg, 0.4 mmol, 10 mol%), XantPhos (464 mg, 0.8 mmol, 20 mol%) and Cs2CO3 (3.92 g, 12 mmol, 3.0 equiv.) in THF (16 mL). The resulting mixture was degassed by evacuation/back-filling with nitrogen three times. The mixture was then stirred at 100 °C under a nitrogen atmosphere for 16 h. Upon complete conversion, as judged by LCMS analysis, the reaction mixture was cooled to rt, diluted with EtOAc, filtered over Celite®, and concentrated in vacuo. The crude product was purified by column chromatography (SiO2, 0 to 30% EtOAc in hexanes) to provide the intermediate imine as a yellowish solid (1.0 g), which was combined with NaOAc (557 mg, 6.8 mmol, 2.5 equiv.) and hydroxylamine hydrochloride (378 mg, 5.4 mmol, 2.0 equiv.) in MeOH (15 mL). The resulting mixture was stirred at rt for 2 h. Upon complete conversion, as judged by LCMS analysis, the reaction mixture was concentrated in vacuo, diluted with EtOAc and sat. aq. NaHCO3. The organic phase was separated, dried, and concentrated in vacuo. Purification by column chromatography (SiO2, 10 to 50% EtOAc in hexanes) provided methyl 3-amino-4-chloro-5-fluorobenzoate as a clear oil (480 mg, 58% yield over two steps). [0277] Steps b-d: The title compound was prepared from methyl 3-amino-4-chloro-5- fluorobenzoate obtained in step a in a similar fashion to Ex.12.1H NMR (400 MHz, Methanol- d4) δ 8.68 (dt, J = 7.0, 1.1 Hz, 1H), 8.63 (s, 1H), 8.29 (dt, J = 8.9, 1.3 Hz, 1H), 8.26 (dd, J = 1.9, 1.4 Hz, 1H), 7.71 (dd, J = 9.8, 1.9 Hz, 1H), 7.52 (ddd, J = 8.9, 6.9, 1.1 Hz, 2H), 7.11 (td, J = 6.9,
1.4 Hz, 1H), 2.09 (tt, J = 8.3, 5.1 Hz, 1H), 1.16 – 1.00 (m, 6H). ESI MS [M+H]+ for C19H15ClFN6O, calcd 397.1, found 397.1. Example 50: N-[4-Chloro-5-(5-cyclopropyl-4H-1,2,4-triazol-3-yl)-2- methylphenyl]pyrazolo[1,5-a]pyridine-3-carboxamide
[0278] Step a: A round-bottom flask was charged with methyl 5-amino-4-bromo-2- chlorobenzoate (1.0 g, 3.78 mmol, 1.0 equiv.), trimethylboroxine (0.58 mL, 4.15 mmol, 1.1 equiv.) and PdCl2(dppf) (277 mg, 0.378 mmol, 10 mol% equiv.) in 2 M Na2CO3 (6 mL) and dioxane (18 mL). The resulting mixture was degassed by evacuation/back-filling with nitrogen three times. The mixture was then stirred at 100 °C under a nitrogen atmosphere for 3 h. Upon complete conversion, as judged by LCMS analysis, the reaction mixture was cooled to rt, and diluted with EtOAc and sat. aq. NaCl. The organic phase was separated, dried, and concentrated in vacuo. Purification by column chromatography (SiO2, 10 to 50% EtOAc in hexanes) provided methyl 5- amino-2-chloro-4-methylbenzoate as a clear oil (750 mg, 98% yield). [0279] Steps b-d: The title compound was prepared from methyl 5-amino-2-chloro-4- methylbenzoate obtained in step a in a similar fashion to Ex.12.1H NMR (400 MHz, DMSO-d6) δ 13.88 – 13.64 (br., 1H), 9.72 (s, 1H), 8.81 (dt, J = 7.0, 1.1 Hz, 1H), 8.72 (s, 1H), 8.18 (dt, J = 8.9, 1.3 Hz, 1H), 7.81 (s, 1H), 7.48 (ddd, J = 8.9, 6.8, 1.1 Hz, 1H), 7.08 (td, J = 6.9, 1.4 Hz, 1H),
2.26 (s, 3H), 2.09 – 1.99 (m, 1H), 1.06 – 0.80 (m, 4H). ESI MS [M+H]+ for C20H18ClN6O, calcd 393.1, found 393.1. Example 51: N-[4-Chloro-5-(5-cyclopropyl-4H-1,2,4-triazol-3-yl)-2-methylphenyl]-6- methoxypyrazolo[1,5-a]pyridine-3-carboxamide
[0280] The title compound was prepared in a similar fashion to Ex. 50 using triazolo[4,5- b]pyridin-3-yl 6-methoxypyrazolo[1,5-a]pyridine-3-carboxylate, which was prepared from 6- methoxypyrazolo[1,5-a]pyridine-3-carboxylic acid in a similar fashion to Ex.1, step a.1H NMR (400 MHz, DMSO-d6) δ 13.76 (s, 1H), 9.67 (s, 1H), 8.61 (s, 1H), 8.51 (dd, J = 2.3, 0.8 Hz, 1H), 8.06 (dd, J = 9.6, 0.7 Hz, 2H), 7.80 (s, 1H), 7.44 (s, 1H), 7.28 (dd, J = 9.6, 2.2 Hz, 1H), 3.83 (s, 3H), 2.26 (s, 3H), 2.07 – 2.01 (m, 1H), 0.99 (s, 2H), 0.89 (s, 2H). ESI MS [M+H]+ for C21H20ClN6O2, calcd 423.1, found 423.1. Example 52: N-[5-(5-Cyclopropyl-4H-1,2,4-triazol-3-yl)-4-fluoro-2-methylphenyl]-4- fluoropyrazolo[1,5-a]pyridine-3-carboxamide
[0281] The title compound was prepared from 4-fluoropyrazolo[1,5-a]pyridine-3-carboxylic acid in a similar fashion to Ex.12. 1H NMR (400 MHz, DMSO-d6) δ 13.78 (s, 1H), 9.90 – 9.57 (m, 1H), 8.87 – 8.67 (m, 1H), 8.66 – 8.47 (m, 1H), 8.00 (dd, J = 12.9, 7.1 Hz, 1H), 7.43 – 7.17 (m, 2H), 7.09 (td, J = 7.4, 4.9 Hz, 1H), 2.34 – 2.26 (m, 3H), 2.08 (td, J = 8.6, 4.3 Hz, 1H), 1.09 – 0.81 (m, 4H). ESI MS [M+H]+ for C20H17F2N6O, calcd 395.1, found 395.1. Example 53: N-[2-Chloro-5-(5-cyclopropyl-4H-1,2,4-triazol-3-yl)-4- methylphenyl]pyrazolo[1,5-a]pyridine-3-carboxamide 109
[0282] The title compound was prepared from methyl 5-amino-2-bromo-4-chlorobenzoate in a similar fashion to Ex.50.1H NMR (400 MHz, DMSO-d6) δ 13.79 (s, 1H), 9.82 (s, 1H), 8.84 (dt, J = 7.0, 1.1 Hz, 1H), 8.78 (s, 1H), 8.22 (dt, J = 8.9, 1.3 Hz, 1H), 8.08 (s, 1H), 7.57 – 7.44 (m, 2H), 7.12 (td, J = 6.9, 1.4 Hz, 1H), 2.58 (s, 3H), 2.08 (m, 1H), 1.10 - 0.88 (m, 4H). ESI MS [M+H]+ for C20H18ClN6O, calcd 393.1, found 393.1. Example 54: N-[2-Chloro-5-(5-cyclopropyl-4H-1,2,4-triazol-3-yl)-4-methylphenyl]-6- fluoropyrazolo[1,5-a]pyridine-3-carboxamide
[0283] The title compound was prepared from methyl 5-amino-2-bromo-4-chlorobenzoate and 6-fluoropyrazolo[1,5-a]pyridine-3-carboxylic acid in a similar fashion to Ex. 50. 1H NMR (400 MHz, DMSO-d6) δ 13.79 (s, 1H), 9.90 (s, 1H), 9.19 (ddd, J = 4.6, 2.3, 0.7 Hz, 1H), 8.80 (s, 1H), 8.30 – 8.20 (m, 1H), 8.06 (s, 1H), 7.65 (ddd, J = 10.3, 8.3, 2.3 Hz, 1H), 7.49 (s, 1H), 2.58 (s, 3H), 2.15 – 2.00 (m, 1H), 0.99 (d, J = 37.7 Hz, 4H). 19F NMR (376 MHz, DMSO-d6) δ -138.54. ESI MS [M+H]+ for C20H17ClFN6O, calcd 411.1, found 411.2. Example 55: N-[5-(5-Cyclopropyl-4H-1,2,4-triazol-3-yl)-4-fluoro-2-methylphenyl]-6-fluoro- 4-methoxypyrazolo[1,5-a]pyridine-3-carboxamide
[0284] The title compound was prepared from 3,5-difluoropyridine in a similar fashion to Ex. 24, wherein the C4-fluoro group of methyl 4,6-difluoropyrazolo[1,5-a]pyridine-3-carboxylate was displaced by MeOH in step d to afford 6-fluoro-4-methoxypyrazolo[1,5-a]pyridine-3-carboxylic acid.1H NMR (400 MHz, DMSO-d6) δ 13.77 (s, 1H), 9.53 (s, 1H), 8.80 (dd, J = 4.2, 1.9 Hz, 1H),
8.39 (d, J = 5.3 Hz, 1H), 8.14 (dd, J = 13.9, 7.3 Hz, 1H), 7.21 (dd, J = 11.0, 3.8 Hz, 2H), 4.06 (s, 3H), 2.31 (s, 3H), 2.09 (td, J = 8.6, 4.3 Hz, 1H), 1.09 – 1.02 (m, 1H), 1.00 – 0.89 (m, 2H), 0.85 (s, 1H). ESI MS [M+H]+ for C21H19F2N6O2, calcd 425.2, found 425.0. Example 56: N-[5-(5-Cyclopropyl-4H-1,2,4-triazol-3-yl)-4-fluoro-2-methylphenyl]-4- ethoxypyrazolo[1,5-a]pyridine-3-carboxamide
[0285] The title compound was prepared from 3-ethoxypyridine in a similar fashion to Ex.24. 1H NMR (400 MHz, DMSO-d6) δ 13.74 (s, 1H), 9.64 (s, 1H), 8.43 (dd, J = 6.5, 1.1 Hz, 1H), 8.33 (s, 1H), 7.95 (d, J = 7.4 Hz, 1H), 7.19 (d, J = 11.4 Hz, 1H), 7.05 – 6.91 (m, 2H), 4.26 (q, J = 6.9 Hz, 2H), 2.25 (s, 3H), 2.11 – 1.92 (m, 1H), 1.27 (t, J = 6.9 Hz, 3H), 1.06 – 0.77 (m, 4H). ESI MS [M+H]+ for C22H22FN6O2, calcd 421.2, found 421.2. Example 57: N-[4-Chloro-5-(5-cyclopropyl-4H-1,2,4-triazol-3-yl)-2-methylphenyl]-6- fluoropyrazolo[1,5-a]pyridine-3-carboxamide
[0286] The title compound was prepared from 6-fluoropyrazolo[1,5-a]pyridine-3-carboxylic acid in a similar fashion to Ex.50.1H NMR (400 MHz, Methanol-d4) δ 8.78 (ddd, J = 4.2, 2.2, 0.8 Hz, 1H), 8.59 (s, 1H), 8.27 (ddd, J = 9.9, 5.7, 0.8 Hz, 1H), 7.70 (s, 1H), 7.54 – 7.44 (m, 2H), 2.35 (s, 3H), 2.09 (tt, J = 8.3, 5.0 Hz, 1H), 1.04 (dd, J = 21.9, 6.2 Hz, 4H).19F NMR (376 MHz, DMSO- d6) δ -140.13. ESI MS [M+H]+ for C20H17ClFN6O, calcd 411.1, found 411.2. Example 58: N-[5-(5-Cyclopropyl-4H-1,2,4-triazol-3-yl)-4-fluoro-2-methylphenyl]-6- morpholin-4-ylpyrazolo[1,5-a]pyridine-3-carboxamide
[0287] Step a: Methyl 6-bromopyrazolo[1,5-a]pyridine-3-carboxylate (510 mg, 2 mmol, 1.0 equiv.), RuPhos Pd G4 (340 mg, 0.4 mmol, 20 mol%) and Cs2CO3 (1.95 g, 6 mmol, 3.0 equiv.) were placed in a pressure vial. The vial was placed under N2, then dioxane (10 mL) and morpholine (348 mg, 4 mmol, 2.0 equiv.) were added. The reaction mixture was degassed again and stirred at 100 °C for 14 h. The reaction mixture was filtered and the filtrate was concentrated under reduced pressure and purified by column chromatography to afford the coupled product. [0288] Step b: A solution of the product from step a (450 mg, 1.72 mmol, 1.0 equiv.) in 2:1:1 THF:MeOH:H2O (20 mL) was treated with LiOH ^H2O (206 mg, 8.62 mmol, 5.0 equiv). The thick mixture was allowed to stir at rt for 14 h. The resulting solution was diluted with water (6 mL) and treated with 1N aq. HCl (5 mL) dropwise over a few min. The mixture was allowed to stir for several additional min. to break up any clumps. The mixture was then filtered and the filter cake was rinsed with water, followed by hexanes. The resulting solid was dried under vacuum overnight to provide 6-morpholin-4-ylpyrazolo[1,5-a]pyridine-3-carboxylic acid. [0289] Steps c-d: The title compound was prepared in a similar fashion to Ex.24, steps e-f.1H NMR (400 MHz, DMSO-d6) δ 13.74 (s, 1H), 9.61 (s, 1H), 8.57 (s, 1H), 8.27 – 8.10 (m, 1H), 8.03 (dd, J = 9.7, 0.7 Hz, 1H), 7.93 – 7.75 (m, 1H), 7.52 (dd, J = 9.7, 2.1 Hz, 1H), 7.18 (d, J = 11.4 Hz, 1H), 3.81 – 3.65 (m, 4H), 3.16 – 3.00 (m, 4H), 2.24 (s, 3H), 2.05 (s, 1H), 1.08 – 0.79 (m, 4H). ESI MS [M+H]+ for C24H25FN7O2, calcd 462.2, found 462.0. Example 59: N-[5-(5-Cyclopropyl-4H-1,2,4-triazol-3-yl)-4-fluoro-2-methylphenyl]-6-(4- fluorophenyl)pyrazolo[1,5-a]pyridine-3-carboxamide
[0290] Step a: A mixture of methyl 6-bromopyrazolo[1,5-a]pyridine-3-carboxylate compound (510 mg, 2.0 mmol), B2pin2 (762 mg, 1.5 mmol), Pd(dppf)Cl2 (73 mg, 0.1 mmol), and KOAc (392 mg, 4 mmol) was placed under N2. Degassed dioxane (20 mL) was added and the reaction mixture was stirred at 100 °C for 4 h. The mixture was cooled to rt, concentrated, diluted with CH2Cl2 (50 mL), filtered through Celite® to remove solids, and again concentrated to afford the desired intermediate, which was used directly in part 2 of step a without further purification. A mixture of the product from part 1 of step a (2 mmol assumed), 4-bromofluorobenzene (700 mg, 4 mmol), Pd(dppf)Cl2 (73 mg, 0.1 mmol), and K2CO3 (552 mg, 8 mmol) was placed under N2. Degassed dioxane (10 mL) and degassed water (10 mL) were added and the reaction mixture was stirred at 100 °C for 2 h. The mixture was cooled to rt, concentrated, diluted with CH2Cl2 (25 mL), dried over MgSO4, and concentrated. The crude material was purified by column chromatography (0 to 20% EtOAc in hexanes) to provide the desired product as a colorless liquid. [0291] Step b: Ester hydrolysis was carried out similar to Ex.58, step b. [0292] Steps c-d: The title compound was prepared in a similar fashion to Ex.24, steps e-f.1H NMR (400 MHz, DMSO-d6) δ 13.75 (s, 1H), 9.74 (s, 1H), 9.17 (d, J = 1.8 Hz, 1H), 8.75 (s, 1H), 8.24 (dd, J = 9.3, 0.9 Hz, 1H), 7.93 – 7.78 (m, 3H), 7.40 – 7.26 (m, 2H), 7.20 (d, J = 11.5 Hz, 1H), 3.26 (dd, J = 7.4, 6.7 Hz, 1H), 2.66 (t, J = 0.7 Hz, 1H), 2.26 (s, 1H), 2.14 (dd, J = 8.6, 7.6 Hz, 1H), 2.10 – 1.96 (m, 1H), 1.92 – 1.80 (m, 1H), 1.07 – 0.96 (m, 1H), 0.96 – 0.83 (m, 2H). ESI MS [M+H]+ for C26H21F2N6O, calcd 471.2, found 471.0. Example 60: N-[5-[5-(2,2-Difluorocyclopropyl)-4H-1,2,4-triazol-3-yl]-4-fluoro-2- methylphenyl]pyrazolo[1,5-a]pyridine-3-carboxamide
[0293] The title compound was prepared from 2,2-difluorocyclopropane-1-carboximidamide in a similar fashion to Ex.12.1H NMR (400 MHz, Methanol-d4) δ 8.56 – 8.50 (m, 2H), 8.24 (dt, J = 8.9, 1.2 Hz, 1H), 7.87 (d, J = 6.9 Hz, 1H), 7.40 (ddd, J = 8.9, 6.8, 1.0 Hz, 1H), 7.12 (d, J = 11.2 Hz, 1H), 7.00 (td, J = 6.9, 1.4 Hz, 1H), 2.85 (d, J = 10.6 Hz, 1H), 2.33 (s, 3H), 2.12 (q, J = 9.0, 8.2 Hz, 1H), 1.88 (s, 1H). ESI MS [M+H]+ for C20H16F3N6O, calcd 413.1, found 413.0. Example 61: N-[5-[5-(2,2-Difluorocyclopropyl)-4H-1,2,4-triazol-3-yl]-4-fluoro-2- methylphenyl]-6-methoxypyrazolo[1,5-a]pyridine-3-carboxamide
[0294] The title compound was prepared from 2,2-difluorocyclopropane-1-carboximidamide and triazolo[4,5-b]pyridin-3-yl 6-methoxypyrazolo[1,5-a]pyridine-3-carboxylate in a similar fashion to Ex.12.1H NMR (400 MHz, Methanol-d4) δ 8.42 (s, 1H), 8.14 – 8.09 (m, 2H), 7.86 (d, J = 7.0 Hz, 1H), 7.18 (dd, J = 9.8, 2.1 Hz, 1H), 7.12 (d, J = 11.3 Hz, 1H), 3.84 (s, 3H), 2.85 (d, J = 10.4 Hz, 1H), 2.33 (s, 3H), 2.09 (dd, J = 13.5, 7.6 Hz, 1H), 1.89 (d, J = 14.3 Hz, 1H). ESI MS [M+H]+ for C21H18F3N6O2, calcd 443.1, found 443.0. Example 62: N-[5-[5-(2,2-Difluorocyclopropyl)-4H-1,2,4-triazol-3-yl]-4-fluoro-2- methylphenyl]-6-fluoropyrazolo[1,5-a]pyridine-3-carboxamide
[0295] The title compound was prepared from 2,2-difluorocyclopropane-1-carboximidamide and triazolo[4,5-b]pyridin-3-yl 6-fluoropyrazolo[1,5-a]pyridine-3-carboxylate in a similar fashion to Ex.12.1H NMR (400 MHz, Methanol-d4) δ 8.52 (td, J = 2.5, 1.1 Hz, 2H), 8.26 (ddd, J = 9.9, 5.7, 0.8 Hz, 1H), 7.92 – 7.85 (m, 1H), 7.35 (ddd, J = 10.0, 7.8, 2.2 Hz, 1H), 7.12 (d, J = 11.2 Hz,
1H), 2.83 (d, J = 0.7 Hz, 1H), 2.33 (s, 3H), 2.10 (s, 1H), 1.86 (s, 1H). ESI MS [M+H]+ for C20H15F4N6O, calcd 431.1, found 431.0. Example 63: N-[5-[5-(3,3-Difluorocyclobutyl)-4H-1,2,4-triazol-3-yl]-4-fluoro-2- methylphenyl]-6-fluoropyrazolo[1,5-a]pyridine-3-carboxamide
[0296] The title compound was prepared from 3,3-difluorocyclobutane-1-carboximidamide and triazolo[4,5-b]pyridin-3-yl 6-fluoropyrazolo[1,5-a]pyridine-3-carboxylate in a similar fashion to Ex.12.1H NMR (400 MHz, DMSO-d6) δ 9.80 (s, 1H), 9.16 (ddd, J = 4.6, 2.2, 0.8 Hz, 1H), 8.74 (s, 1H), 8.22 (ddd, J = 9.8, 5.9, 0.8 Hz, 1H), 7.94 (d, J = 7.1 Hz, 1H), 7.61 (ddd, J = 9.8, 8.3, 2.3 Hz, 1H), 7.29 (d, J = 11.4 Hz, 1H), 3.55 – 3.45 (m, 1H), 3.08 – 2.81 (m, 4H), 2.27 (s, 3H). ESI MS [M+H]+ for C21H17F4N6O, calcd 445.1, found 445.1. Example 64: N-[5-[5-(3,3-Difluorocyclopentyl)-4H-1,2,4-triazol-3-yl]-4-fluoro-2- methylphenyl]-6-fluoropyrazolo[1,5-a]pyridine-3-carboxamide
[0297] The title compound was prepared from 3,3-difluorocyclopentane-1-carboximidamide and triazolo[4,5-b]pyridin-3-yl 6-fluoropyrazolo[1,5-a]pyridine-3-carboxylate in a similar fashion to Ex.12.1H NMR (400 MHz, DMSO-d6) δ 9.80 (s, 1H), 9.16 (ddd, J = 4.6, 2.2, 0.8 Hz, 1H), 8.74 (s, 1H), 8.21 (ddd, J = 9.8, 5.9, 0.8 Hz, 1H), 7.92 (d, J = 7.1 Hz, 1H), 7.61 (ddd, J = 9.8, 8.2, 2.2 Hz, 1H), 7.27 (s, 1H), 3.50 (s, 1H), 2.66 – 2.49 (m, 1H), 2.44 – 2.32 (m, 1H), 2.27 (s, 3H), 2.26 – 2.09 (m, 3H), 2.06 – 1.95 (m, 1H). ESI MS [M+H]+ for C22H19F4N6O, calcd 459.2, found 459.1. Example 65: N-[4-Fluoro-2-methyl-5-(5-propan-2-yl-4H-1,2,4-triazol-3- yl)phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide
[0298] The title compound was prepared from 2-methylpropionamidine HCl in a similar fashion to Ex.12.1H NMR (400 MHz, DMSO-d6) δ 9.72 (s, 1H), 8.81 (dt, J = 7.0, 1.1 Hz, 1H), 8.72 (s, 1H), 8.19 (dt, J = 8.9, 1.3 Hz, 1H), 7.92 (d, J = 7.2 Hz, 1H), 7.48 (ddd, J = 8.9, 6.8, 1.1 Hz, 1H), 7.24 (d, J = 11.4 Hz, 1H), 7.08 (td, J = 6.9, 1.5 Hz, 1H), 3.06 (m, 1H), 2.27 (s, 3H), 1.27 (d, J = 7.0 Hz, 6H).19F NMR (376 MHz, DMSO-d6) δ -74.71. ESI MS [M+H]+ for C20H20FN6O, calcd 379.1, found 379.2. Example 66: 4-Fluoro-N-[4-fluoro-2-methyl-5-(5-propan-2-yl-4H-1,2,4-triazol-3- yl)phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide
[0299] The title compound was prepared from 2-methylpropionamidine and triazolo[4,5- b]pyridin-3-yl 4-fluoropyrazolo[1,5-a]pyridine-3-carboxylate in a similar fashion to Ex. 12. 1H NMR (400 MHz, DMSO-d6) δ 9.73 (s, 1H), 8.71 (dd, J = 6.9, 0.8 Hz, 1H), 8.55 (s, 1H), 8.02 (d, J = 7.1 Hz, 1H), 7.34 (ddd, J = 11.0, 7.8, 0.8 Hz, 1H), 7.23 (d, J = 11.4 Hz, 1H), 7.06 (ddd, J = 7.8, 6.9, 4.9 Hz, 1H), 3.06 (m, 1H), 2.28 (s, 3H), 1.27 (d, J = 6.9 Hz, 6H).19F NMR (376 MHz, DMSO- d6) δ -74.31, -117.40. ESI MS [M+H]+ for C20H19F2N6O, calcd 397.1, found 397.2. Example 67: N-[5-(5-Cyano-4H-1,2,4-triazol-3-yl)-4-fluoro-2-methylphenyl]pyrazolo[1,5- a]pyridine-3-carboxamide
[0300] The title compound was prepared in a similar fashion to Ex.1 from 5-bromo-4H-1,2,4- triazole-3-carbonitrile and N-[4-fluoro-2-methyl-5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2- yl)phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide, which was accessed from 5-bromo-4-fluoro-2-
methylaniline in a fashion similar to step b of Ex.1.1H NMR (400 MHz, Methanol-d4) δ 8.54 – 8.49 (m, 2H), 8.27 – 8.22 (m, 1H), 7.91 (d, J = 6.9 Hz, 1H), 7.40 (ddd, J = 9.0, 6.9, 1.1 Hz, 1H), 7.14 (d, J = 11.2 Hz, 1H), 7.00 (td, J = 6.8, 1.5 Hz, 1H), 2.34 (s, 3H). ESI MS [M+H]+ for C18H13FN7O, calcd 362.1, found 362.1. Example 68: N-[5-(5-Chloro-4H-1,2,4-triazol-3-yl)-4-fluoro-2-methylphenyl]pyrazolo[1,5- a]pyridine-3-carboxamide
[0301] The title compound was prepared from N-[4-fluoro-2-methyl-5-(4,4,5,5-tetramethyl- 1,3,2-dioxaborolan-2-yl)phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide and 3-bromo-5-chloro- 4H-1,2,4-triazole in a similar fashion to Ex. 1 and Ex. 67. 1H NMR (400 MHz, Methanol-d4) δ 8.54 – 8.51 (m, 2H), 8.24 (dt, J = 8.9, 1.3 Hz, 1H), 7.87 (d, J = 7.0 Hz, 1H), 7.41 (ddd, J = 9.0, 6.9, 1.1 Hz, 1H), 7.14 (d, J = 11.4 Hz, 1H), 7.00 (td, J = 6.9, 1.4 Hz, 1H), 2.34 (s, 3H). ESI MS [M+H]+ for C17H13ClFN6O, calcd 371.1, found 371.1. Example 69: N-[5-(5-Bromo-4H-1,2,4-triazol-3-yl)-4-fluoro-2-methylphenyl]pyrazolo[1,5- a]pyridine-3-carboxamide
[0302] The title compound was prepared from N-[4-fluoro-2-methyl-5-(4,4,5,5-tetramethyl- 1,3,2-dioxaborolan-2-yl)phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide and 3,5-dibromo-4- (oxan-2-yl)-1,2,4-triazole in a similar fashion to Ex.1 and Ex.67 and followed by removal of the THP protecting group.1H NMR (400 MHz, Methanol-d4) δ 8.52 (dt, J = 7.0, 1.1 Hz, 2H), 8.24 (dt, J = 8.9, 1.2 Hz, 1H), 7.88 (d, J = 7.0 Hz, 1H), 7.41 (ddd, J = 9.0, 6.8, 1.1 Hz, 1H), 7.14 (d, J = 11.5 Hz, 1H), 7.00 (td, J = 7.0, 1.4 Hz, 1H), 2.34 (s, 3H). ESI MS [M+H]+ for C17H13BrFN6O, calcd 415.0, found 415.0.
Example 70: N-[4-Fluoro-5-[5-(methoxymethyl)-4H-1,2,4-triazol-3-yl]-2- methylphenyl]pyrazolo[1,5-a]pyridine-3-carboxamide
[0303] The title compound was prepared from 2-methoxyethanimidamide in a similar fashion to Ex.12.1H NMR (400 MHz, Methanol-d4) δ 8.54 – 8.50 (m, 2H), 8.23 (dt, J = 9.0, 1.3 Hz, 1H), 7.89 (d, J = 7.0 Hz, 1H), 7.39 (ddd, J = 9.0, 6.8, 1.1 Hz, 1H), 7.12 (dd, J = 11.2, 0.9 Hz, 1H), 6.99 (td, J = 6.9, 1.4 Hz, 1H), 4.56 (s, 2H), 3.41 (s, 3H), 2.33 (s, 3H). ESI MS [M+H]+ for C19H18FN6O2, calcd 381.1, found 381.2. Example 71: N-[5-[5-[2-(Dimethylamino)-2-oxoethyl]-4H-1,2,4-triazol-3-yl]-4-fluoro-2- methylphenyl]pyrazolo[1,5-a]pyridine-3-carboxamide
[0304] The title compound was prepared from 2-cyano-N,N-dimethylacetamide in a similar fashion to Ex.15.1H NMR (400 MHz, DMSO-d6) δ 13.93 (s, 1H), 9.73 (s, 1H), 8.84 (dt, J = 7.0, 1.1 Hz, 1H), 8.75 (s, 1H), 8.23 (dt, J = 8.9, 1.3 Hz, 1H), 7.98 (d, J = 7.2 Hz, 1H), 7.52 (ddd, J = 8.9, 6.8, 1.1 Hz, 1H), 7.30 (d, J = 11.4 Hz, 1H), 7.11 (td, J = 6.9, 1.5 Hz, 1H), 3.91 (s, 2H), 3.08 (s, 3H), 2.85 (s, 3H), 2.31 (s, 3H). ESI MS [M+H]+ for C21H21FN7O2, calcd 422.2, found 422.1. Example 72: N-[4-Fluoro-5-[5-(2-fluorophenyl)-4H-1,2,4-triazol-3-yl]-2- methylphenyl]pyrazolo[1,5-a]pyridine-3-carboxamide
[0305] The title compound was prepared from 2-fluorobenzenecarboximidamide in a similar fashion to Ex.12.1H NMR (400 MHz, Methanol-d4) δ 8.52 (dt, J = 7.0, 1.1 Hz, 2H), 8.25 (dt, J =
8.9, 1.2 Hz, 1H), 8.05 – 7.98 (m, 2H), 7.45 – 7.37 (m, 2H), 7.28 – 7.12 (m, 3H), 7.00 (td, J = 6.9, 1.5 Hz, 1H), 2.35 (d, J = 0.7 Hz, 3H). ESI MS [M+H]+ for C23H17F2N6O, calcd 431.1, found 431.2. Example 73: N-[4-Fluoro-5-[5-(4-fluorophenyl)-4H-1,2,4-triazol-3-yl]-2- methylphenyl]pyrazolo[1,5-a]pyridine-3-carboxamide
[0306] The title compound was prepared from 4-fluorobenzenecarboximidamide in a similar fashion to Ex.12.1H NMR (400 MHz, Methanol-d4) δ 8.59 – 8.56 (m, 2H), 8.29 – 8.24 (m, 1H), 8.09 – 8.03 (m, 3H), 7.44 (ddd, J = 9.0, 6.9, 1.1 Hz, 1H), 7.19 (d, J = 11.4 Hz, 1H), 7.14 (t, J = 8.7 Hz, 2H), 7.04 (td, J = 7.0, 1.4 Hz, 1H), 2.37 (s, 3H). ESI MS [M+H]+ for C23H17F2N6O, calcd 431.1, found 431.1. Example 74: N-[4-Fluoro-2-methyl-5-(5-pyridin-3-yl-4H-1,2,4-triazol-3- yl)phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide
[0307] The title compound was prepared from pyridine-3-carboximidamide in a similar fashion to Ex.12.1H NMR (400 MHz, Methanol-d4) δ 9.34 (d, J = 2.0 Hz, 1H), 8.85 (dt, J = 8.0, 1.8 Hz, 1H), 8.67 (dd, J = 5.3, 1.6 Hz, 1H), 8.59 – 8.52 (m, 2H), 8.26 (dd, J = 8.9, 1.3 Hz, 1H), 8.14 (d, J = 6.9 Hz, 1H), 7.82 (dd, J = 8.1, 5.3 Hz, 1H), 7.45 – 7.39 (m, 1H), 7.18 (d, J = 11.3 Hz, 1H), 7.02 (td, J = 6.9, 1.4 Hz, 1H), 2.37 (s, 3H). ESI MS [M+H]+ for C22H17FN7O, calcd 414.1, found 414.2. Example 75: N-[5-(5-Cyclopropyloxy-4H-1,2,4-triazol-3-yl)-4-fluoro-2- methylphenyl]pyrazolo[1,5-a]pyridine-3-carboxamide
[0308] The title compound was prepared from N-[4-fluoro-2-methyl-5-(4,4,5,5-tetramethyl- 1,3,2-dioxaborolan-2-yl)phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide and 4-benzyl-3-bromo- 5-cyclopropyloxy-1,2,4-triazole in a similar fashion to Ex. 1 and Ex. 67, followed by debenzylation.1H NMR (400 MHz, Methanol-d4) δ 8.51 (dt, J = 7.0, 1.1 Hz, 2H), 8.24 (dt, J = 8.9, 1.2 Hz, 1H), 7.79 (d, J = 7.0 Hz, 1H), 7.39 (ddd, J = 9.0, 6.8, 1.1 Hz, 1H), 7.13 – 7.09 (m, 1H), 6.99 (td, J = 6.9, 1.4 Hz, 1H), 4.16 (tt, J = 6.0, 3.0 Hz, 1H), 2.32 (s, 3H), 0.79 – 0.72 (m, 4H). ESI MS [M+H]+ for C20H18FN6O2, calcd 393.1, found 393.0. Example 76: N-[5-[5-(Dimethylamino)-4H-1,2,4-triazol-3-yl]-4-fluoro-2-methylphenyl]-6- fluoropyrazolo[1,5-a]pyridine-3-carboxamide
[0309] The title compound was prepared in a similar fashion to Ex. 1 from 5-bromo-N,N- dimethyl-4H-1,2,4-triazol-3-amine and 6-fluoro-N-[4-fluoro-2-methyl-5-(4,4,5,5-tetramethyl- 1,3,2-dioxaborolan-2-yl)phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide, which was accessed from 6-fluoropyrazolo[1,5-a]pyridine-3-carboxylic acid and 5-bromo-4-fluoro-2-methylaniline in a similar fashion to step b, Ex.1.1H NMR (400 MHz, Methanol-d4) δ 8.52 (s, 2H), 8.26 (dd, J = 9.8, 5.8 Hz, 1H), 7.79 (d, J = 7.0 Hz, 1H), 7.35 (t, J = 8.8 Hz, 1H), 7.07 (d, J = 11.3 Hz, 1H), 3.00 (s, 6H), 2.30 (s, 3H). ESI MS [M+H]+ for C19H18F2N7O, calcd 398.2, found 398.0. Example 77: N-[5-[5-(Dimethylamino)-4H-1,2,4-triazol-3-yl]-4-fluoro-2-methylphenyl]-5- fluoropyrazolo[1,5-a]pyridine-3-carboxamide
[0310] The title compound was prepared in a similar fashion to Ex. 1 from 5-bromo-N,N- dimethyl-4H-1,2,4-triazol-3-amine and 5-fluoro-N-[4-fluoro-2-methyl-5-(4,4,5,5-tetramethyl- 1,3,2-dioxaborolan-2-yl)phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide, which was accessed from 5-fluoropyrazolo[1,5-a]pyridine-3-carboxylic acid and 5-bromo-4-fluoro-2-methylaniline in
a similar fashion to step b of Ex.1.1H NMR (400 MHz, Methanol-d4) δ 8.53 (dd, J = 7.6, 4.8 Hz, 2H), 7.90 – 7.86 (m, 1H), 7.79 (d, J = 7.1 Hz, 1H), 7.08 (d, J = 11.3 Hz, 1H), 6.89 – 6.84 (m, 1H), 3.00 (s, 6H), 2.30 (s, 3H). ESI MS [M+H]+ for C19H18F2N7O, calcd 398.2, found 398.0. Example 78: N-[5-[5-(Cyclopropylamino)-4H-1,2,4-triazol-3-yl]-4-fluoro-2- methylphenyl]pyrazolo[1,5-a]pyridine-3-carboxamide
[0311] The title compound was prepared from 1-cyclopropylguanidine in a similar fashion to Ex.12.1H NMR (400 MHz, Methanol-d4) δ 8.51 (d, J = 7.3 Hz, 2H), 8.27 – 8.21 (m, 1H), 7.73 (d, J = 7.0 Hz, 1H), 7.42 – 7.36 (m, 1H), 7.07 (d, J = 11.4 Hz, 1H), 6.98 (td, J = 6.9, 1.4 Hz, 1H), 2.55 (tt, J = 6.8, 3.5 Hz, 1H), 2.30 (s, 3H), 0.73 (td, J = 6.9, 4.9 Hz, 2H), 0.58 – 0.53 (m, 2H). ESI MS [M+H]+ for C20H19FN7O, calcd 392.2, found 392.1. Example 79: N-[4-Fluoro-2-methyl-5-(5-pyrrolidin-1-yl-4H-1,2,4-triazol-3- yl)phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide
[0312] The title compound was prepared from N-[4-fluoro-2-methyl-5-(4,4,5,5-tetramethyl- 1,3,2-dioxaborolan-2-yl)phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide and 3-bromo-5- pyrrolidin-1-yl-4H-1,2,4-triazole in a similar fashion to Ex. 1 and Ex. 67. 1H NMR (400 MHz, Methanol-d4) δ 8.53 – 8.49 (m, 2H), 8.24 (d, J = 8.9 Hz, 1H), 7.76 (d, J = 7.0 Hz, 1H), 7.41 – 7.37 (m, 1H), 7.07 (d, J = 11.4 Hz, 1H), 7.01 – 6.96 (m, 1H), 3.45 – 3.39 (m, 4H), 2.31 (s, 3H), 2.02 – 1.94 (m, 4H). ESI MS [M+H]+ for C21H21F1N7O, calcd 406.2, found 406.0. Example 80: N-[4-Fluoro-2-methyl-5-(5-piperazin-1-yl-4H-1,2,4-triazol-3- yl)phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide
[0313] The title compound was prepared from N-[4-fluoro-2-methyl-5-(4,4,5,5-tetramethyl- 1,3,2-dioxaborolan-2-yl)phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide and 1-(5-bromo-4H- 1,2,4-triazol-3-yl)piperazine in a similar fashion to Ex. 1 and Ex. 67. 1H NMR (400 MHz, Methanol-d4) δ 8.55 – 8.49 (m, 1H), 8.42 (s, 1H), 8.27 – 8.22 (m, 1H), 7.90 (d, J = 7.0 Hz, 1H), 7.43 – 7.37 (m, 1H), 7.10 (d, J = 11.3 Hz, 1H), 7.00 (td, J = 7.0, 1.4 Hz, 1H), 3.58 (t, J = 5.1 Hz, 4H), 3.13 (t, J = 5.2 Hz, 4H), 2.33 (s, 3H). ESI MS [M+H]+ for C21H22FN8O, calcd 421.2, found 421.1. Example 81: N-[5-[5-(Cyclopropylmethyl)-4H-1,2,4-triazol-3-yl]-4-fluoro-2-methylphenyl]- 6-methoxypyrazolo[1,5-a]pyridine-3-carboxamide
[0314] The title compound was prepared from triazolo[4,5-b]pyridin-3-yl 6- methoxypyrazolo[1,5-a]pyridine-3-carboxylate in a similar fashion to Ex.15.1H NMR (400 MHz, Methanol-d4) δ 8.54 (s, 1H), 8.35 (dd, J = 2.2, 0.7 Hz, 1H), 8.16 (dd, J = 9.7, 0.7 Hz, 1H), 8.05 (d, J = 6.9 Hz, 1H), 7.40 (d, J = 11.1 Hz, 1H), 7.31 (dd, J = 9.7, 2.2 Hz, 1H), 3.91 (s, 3H), 2.98 (d, J = 7.2 Hz, 2H), 2.44 (s, 3H), 1.31 – 1.18 (m, 1H), 0.77 – 0.68 (m, 2H), 0.41 (dt, J = 6.0, 4.7 Hz, 2H). ESI MS [M+H]+ for C22H22FN6O2, calcd 421.2, found 421.2. Example 82: 6-Fluoro-N-[4-fluoro-2-methyl-5-(5-methyl-4H-1,2,4-triazol-3- yl)phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide
[0315] The title compound was prepared from ethanimidamide and triazolo[4,5-b]pyridin-3-yl 6-fluoropyrazolo[1,5-a]pyridine-3-carboxylate in a similar fashion to Ex.12.1H NMR (400 MHz,
Methanol-d4) δ 8.52 (dt, J = 3.7, 1.5 Hz, 2H), 8.26 (dd, J = 9.8, 5.6 Hz, 1H), 7.81 (d, J = 7.0 Hz, 1H), 7.34 (ddd, J = 9.9, 7.9, 2.2 Hz, 1H), 7.09 (d, J = 11.3 Hz, 1H), 2.42 (s, 3H), 2.31 (s, 3H). ESI MS [M+H]+ for C18H15F2N6O, calcd 369.1, found 369.0. Example 83: N-[4-Fluoro-2-methyl-5-(5-methyl-4H-1,2,4-triazol-3-yl)phenyl]-6- methylpyrazolo[1,5-a]pyridine-3-carboxamide
[0316] The title compound was prepared from ethanimidamide and triazolo[4,5-b]pyridin-3-yl 6-methylpyrazolo[1,5-a]pyridine-3-carboxylate in a similar fashion to Ex.12.1H NMR (400 MHz, Methanol-d4) δ 8.44 (s, 1H), 8.31 (q, J = 1.2 Hz, 1H), 8.12 (dd, J = 9.1, 1.0 Hz, 1H), 7.81 (d, J = 7.0 Hz, 1H), 7.26 (dd, J = 9.1, 1.5 Hz, 1H), 7.09 (d, J = 11.3 Hz, 1H), 2.42 (s, 3H), 2.36 (d, J = 1.1 Hz, 3H), 2.31 (s, 3H). ESI MS [M+H]+ for C19H18FN6O, calcd 365.1, found 365.1. Example 84: 6-Ethyl-N-[4-fluoro-5-(5-methoxy-4H-1,2,4-triazol-3-yl)-2- methylphenyl]pyrazolo[1,5-a]pyridine-3-carboxamide
[0317] The title compound was prepared in a similar fashion to Ex.1 from 3-bromo-5-methoxy- 4H-1,2,4-triazole and 6-ethyl-N-[4-fluoro-2-methyl-5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2- yl)phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide, which was accessed from 6-ethylpyrazolo[1,5- a]pyridine-3-carboxylic acid and 5-bromo-4-fluoro-2-methylaniline in a similar fashion to step b of Ex.1.1H NMR (400 MHz, Methanol-d4) δ 8.41 – 8.33 (m, 2H), 7.81 (d, J = 7.0 Hz, 1H), 7.19 – 7.15 (m, 1H), 7.11 (d, J = 11.4 Hz, 1H), 6.92 (t, J = 7.0 Hz, 1H), 3.98 (s, 3H), 3.13 (q, J = 7.4 Hz, 2H), 2.34 (s, 3H), 1.19 (t, J = 7.4 Hz, 3H). ESI MS [M+H]+ for C20H20FN6O2, calcd 395.2, found 395.0. Example 85: 5-Fluoro-N-[4-fluoro-5-(5-methoxy-4H-1,2,4-triazol-3-yl)-2- methylphenyl]pyrazolo[1,5-a]pyridine-3-carboxamide
[0318] The title compound was prepared from 5-fluoro-N-[4-fluoro-2-methyl-5-(4,4,5,5- tetramethyl-1,3,2-dioxaborolan-2-yl)phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide and 3- bromo-5-methoxy-4H-1,2,4-triazole in a similar fashion to Ex.1 and Ex.77.1H NMR (400 MHz, Methanol-d4) δ 8.56 – 8.50 (m, 2H), 7.88 (dd, J = 8.9, 2.8 Hz, 1H), 7.80 (d, J = 7.0 Hz, 1H), 7.11 (d, J = 11.4 Hz, 1H), 6.86 (td, J = 7.3, 2.9 Hz, 1H), 3.97 (s, 3H), 2.31 (s, 3H). ESI MS [M+H]+ for C18H15F2N6O2, calcd 385.1, found 385.0. Example 86: N-[4-Fluoro-5-(5-methoxy-4H-1,2,4-triazol-3-yl)-2-methylphenyl]-6- methoxypyrazolo[1,5-a]pyridine-3-carboxamide
[0319] The title compound was prepared in a similar fashion to Ex.1 from 3-bromo-5-methoxy- 4H-1,2,4-triazole and N-[4-fluoro-2-methyl-5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2- yl)phenyl]-6-methoxypyrazolo[1,5-a]pyridine-3-carboxamide, which was accessed from 6- methoxypyrazolo[1,5-a]pyridine-3-carboxylic acid and 5-bromo-4-fluoro-2-methylaniline in a similar fashion to step b, Ex.1.1H NMR (400 MHz, Methanol-d4) δ 8.39 (s, 1H), 8.13 – 8.08 (m, 2H), 7.78 (d, J = 7.0 Hz, 1H), 7.16 (dd, J = 9.6, 2.2 Hz, 1H), 7.09 (d, J = 11.4 Hz, 1H), 3.97 (s, 3H), 3.84 (s, 3H), 2.31 (s, 3H). ESI MS [M+H]+ for C19H18FN6O3, calcd 397.1, found 397.2. Example 87: 6-Fluoro-N-[4-fluoro-2-methyl-5-[5-(oxolan-3-yl)-4H-1,2,4-triazol-3- yl]phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide
[0320] The title compound was prepared from oxolane-3-carboximidamide and triazolo[4,5- b]pyridin-3-yl 6-fluoropyrazolo[1,5-a]pyridine-3-carboxylate in a similar fashion to Ex. 12. 1H
NMR (400 MHz, DMSO-d6) δ 9.79 (s, 1H), 9.16 (ddd, J = 4.6, 2.3, 0.8 Hz, 1H), 8.74 (s, 1H), 8.22 (ddd, J = 9.8, 5.9, 0.8 Hz, 1H), 7.92 (d, J = 7.1 Hz, 1H), 7.61 (ddd, J = 9.8, 8.3, 2.3 Hz, 1H), 7.27 (d, J = 11.4 Hz, 1H), 4.02 (t, J = 7.9 Hz, 1H), 3.84 (td, J = 8.1, 5.6 Hz, 1H), 3.81 – 3.72 (m, 2H), 3.55 (p, J = 7.3 Hz, 1H), 2.31 – 2.21 (m, 4H), 2.19 – 2.10 (m, 1H). ESI MS [M+H]+ for C21H19F2N6O2, calcd 425.2, found 425.1. Example 88: N-[5-(5-Cyclopropyl-2-methyl-1,2,4-triazol-3-yl)-4-fluoro-2- methylphenyl]pyrazolo[1,5-a]pyridine-3-carboxamide
[0321] The title compound was prepared from 5-bromo-3-cyclopropyl-1-methyl-1,2,4-triazole and N-[4-fluoro-2-methyl-5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)phenyl]pyrazolo[1,5- a]pyridine-3-carboxamide in a similar fashion to Ex.1 and Ex.67.1H NMR (400 MHz, DMSO- d6) δ 9.71 (s, 1H), 8.81 (dt, J = 7.0, 1.1 Hz, 1H), 8.71 (s, 1H), 8.17 (dt, J = 8.9, 1.3 Hz, 1H), 7.55 – 7.44 (m, 2H), 7.36 (d, J = 10.9 Hz, 1H), 7.08 (td, J = 6.9, 1.5 Hz, 1H), 3.68 (d, J = 1.9 Hz, 3H), 2.31 (s, 3H), 2.01 – 1.86 (m, 1H), 0.93 – 0.84 (m, 2H), 0.83 – 0.73 (m, 2H). ESI MS [M+H]+ for C21H20FN6O, calcd 391.2, found 391.1. Example 89: N-[5-(1-Benzyl-5-hydroxy-1,2,4-triazol-3-yl)-4-fluoro-2- methylphenyl]pyrazolo[1,5-a]pyridine-3-carboxamide
[0322] Step a: A vial was charged with N-[4-fluoro-2-methyl-5-(4,4,5,5-tetramethyl-1,3,2- dioxaborolan-2-yl)phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide (494 mg, 1.25 mmol, 1.0 equiv.), 4-benzyl-3-bromo-5-fluoro-1,2,4-triazole (318 mg, 1.25 mmol, 1.0 equiv.), Pd XPhos G3 (105 mg, 0.12 mmol, 10 mol%), 2M Na2CO3 (aq. soln., 1.25 mL, 2.0 equiv.) and dioxane (6 mL). The reaction mixture was degassed with N2, and then stirred at 95 °C for 6 h, at which time LCMS analysis indicated complete consumption of starting material. The reaction mixture was cooled to rt and diluted with EtOAc and water. The organic layer was separated, dried over Na2SO4 and concentrated in vacuo. The crude product was purified by column chromatography (0% to 70% EtOAc in hexanes) to provide the desired N-[5-(4-benzyl-5-fluoro-1,2,4-triazol-3-yl)-4-fluoro-2- methylphenyl]pyrazolo[1,5-a]pyridine-3-carboxamide. [0323] Step b: A vial was charged with the product from step a (75 mg, 0.17 mmol, 1.0 equiv.), NaOEt (68 mg, 1.02 mmol, 6.0 equiv.) and EtOH (1 mL). The reaction mixture was stirred at 80 °C for approximately 16 h, at which time LCMS analysis indicated 70% consumption of starting material. The reaction mixture was cooled to rt and diluted with water, which caused precipitation of the product. The solid was collected by vacuum filtration and dried in vacuo to afford the crude product, which was used directly in the next step without further purification. [0324] Step c: A reactor jar was charged with the product from step b (65 mg, 0.13 mmol, 1.0 equiv.) and Pd/C (13 mg, 20 wt%) in MeOH (10 mL) containing a few drops of 4N HCl in dioxane. The reaction mixture was shaken under 30 psi of hydrogen gas for 3 h, then filtered over Celite® and concentrated under reduced pressure to afford the crude product. Purification by reverse phase HPLC afforded the title compound.1H NMR (400 MHz, Methanol-d4) δ 8.52 (dq, J = 6.6, 1.1 Hz, 2H), 8.23 (dt, J = 8.9, 1.2 Hz, 1H), 7.76 (d, J = 7.0 Hz, 1H), 7.40 (ddd, J = 9.0, 6.9, 1.1 Hz, 1H), 7.32 – 7.19 (m, 5H), 7.08 (dd, J = 11.4, 0.9 Hz, 1H), 7.00 (td, J = 6.9, 1.4 Hz, 1H), 4.95 (s, 2H), 2.31 (s, 3H). ESI MS [M+H]+ for C24H20FN6O2, calcd 443.2, found 443.0. Example 90: N-[5-(1-Benzyl-5-fluoro-1,2,4-triazol-3-yl)-4-fluoro-2- methylphenyl]pyrazolo[1,5-a]pyridine-3-carboxamide
[0325] The title compound was prepared from N-[4-fluoro-2-methyl-5-(4,4,5,5-tetramethyl- 1,3,2-dioxaborolan-2-yl)phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide and 1-benzyl-3-bromo- 5-fluoro-1,2,4-triazole in a similar fashion to Ex.1 and Ex.67.1H NMR (400 MHz, DMSO-d6) δ 8.84 (dt, J = 6.9, 1.1 Hz, 1H), 8.74 (s, 1H), 8.21 (dt, J = 8.9, 1.3 Hz, 1H), 7.90 (d, J = 7.3 Hz, 1H), 7.52 (ddd, J = 8.9, 6.8, 1.1 Hz, 1H), 7.45 – 7.32 (m, 6H), 7.29 (d, J = 11.7 Hz, 1H), 7.11 (td, J = 6.9, 1.5 Hz, 1H), 5.38 (s, 2H), 2.31 (s, 3H). ESI MS [M+H]+ for C24H19F2N6O, calcd 445.2, found 445.2. Example 91: N-[5-(5-Cyclopropyl-4H-1,2,4-triazol-3-yl)-4-fluoro-2- (hydroxymethyl)phenyl]-6-fluoropyrazolo[1,5-a]pyridine-3-carboxamide
[0326] Step a: A vial was charged with methyl 2-amino-4-bromo-5-fluorobenzoate (1.5 g, 6 mmol, 1.0 equiv.) and THF (10 mL). To the reaction mixture was added LiBH4 in THF (2M, 6 mL, 12 mmol, 2.0 equiv.). The reaction mixture was stirred at 50 °C for 2 h, at which time LCMS analysis indicated complete consumption of starting material. The reaction mixture was cooled to rt, diluted with water and stirred for 30 min., during which time a few drops of conc. HCl were added. The mixture was diluted with EtOAc. The organic phase was separated, washed with brine (2x), dried over Na2SO4 and concentrated in vacuo. The crude product was purified by column chromatography (0% to 90% EtOAc in hexanes) to provide the desired (2-amino-4-bromo-5- fluorophenyl) methanol. [0327] Step b: A vial was charged with product from step a (1.3 g, 5.9 mmol, 1.0 equiv.), imidazole (1.0 g, 14.7 mmol, 2.5 equiv.) and DMF (15 mL). TBS-Cl (979 mg, 6.5 mmol, 1.1 equiv.) was added to the mixture. The reaction mixture was stirred for 5 h at 23 °C, at which time LCMS analysis indicated complete consumption of starting material. The reaction mixture was diluted with EtOAc, the layers were separated, and the organic layer was washed successively with sat. aq. NH4Cl (2x) and brine (2x). The combined organic layers were collected, dried over Na2SO4 and concentrated in vacuo. The crude product was purified by column chromatography (0% to 90% EtOAc in hexanes) to provide the desired 5-bromo-2-[[tert-butyl(dimethyl)silyl]oxymethyl]- 4-fluoroaniline. [0328] Step c: A mixture of the product from step b (2.0 g, 5.9 mmol, 1.0 equiv.), and triazolo[4,5-b]pyridin-3-yl pyrazolo[1,5-a]pyridine-3-carboxylate (1.7 g, 5.9 mmol, 1.0 equiv.) in NMP (15.0 mL) was stirred at 120 °C for 1 h. Upon complete conversion, as judged by LCMS analysis, the reaction mixture was cooled to rt and diluted with water. The resulting precipitated solid was collected by vacuum filtration, rinsed with water, and dried in vacuo. The crude product was purified by column chromatography (0% to 80% EtOAc in hexanes) to provide the desired N- [5-bromo-2-[[tert-butyl(dimethyl)silyl]oxymethyl]-4-fluorophenyl]-6-fluoropyrazolo[1,5- a]pyridine-3-carboxamide. [0329] Step d: A vial was charged with the product from step c (1.0 g, 2.0 mmol, 1 equiv.), 4,4,4',4',5,5,5',5'-octamethyl-2,2'-bi(l,3,2-dioxaborolane) (615 mg, 2.4 mmol, 1.2 equiv.), KOAc (600 mg, 6.1 mmol, 3 equiv.) and 1,4-dioxane (15 mL). The reaction mixture was degassed with N2 for 30 min. before Pd(dppf)C12 (74 mg, 0.1 mmol, 5 mmol%) was added. The reaction mixture
was then heated at 100 °C for 12 h, after which time LCMS analysis showed complete conversion of starting material. The reaction mixture was cooled to rt and diluted with EtOAc. The organic layer was filtered over Celite® and the filtrate was concentrated in vacuo. The crude product was purified by column chromatography (0% to 60% EtOAc in hexanes) to provide the desired N-[2- [[tert-butyl(dimethyl)silyl]oxymethyl]-4-fluoro-5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2- yl)phenyl]-6-fluoropyrazolo[1,5-a]pyridine-3-carboxamide. [0330] Step e: A vial was charged with the product from step d (150 mg, 0.27 mmol, 1.0 equiv.), 3-bromo-5-cyclopropyl-4H-1,2,4-triazole (104 mg, 0.55 mmol, 2.0 equiv.), XPhos Pd G3 (47 mg, 0.055 mmol, 20 mol%), K3CO3 (114 mg, 0.83 mmol, 3.0 equiv.), 1,4-dioxane (2.0 mL) and water (0.2 mL). The reaction mixture was degassed with N2 and then stirred at 100 °C for 16 h, at which time LCMS analysis indicated complete consumption of starting material. The reaction mixture was cooled to rt and diluted with EtOAc. The organic layer was separated, filtered over Celite®, and concentrated in vacuo. The crude product was purified by column chromatography (0% to 100% EtOAc in hexanes) to provide the desired N-[2-[[tert-butyl(dimethyl)silyl]oxymethyl]-5-(5- cyclopropyl-4H-1,2,4-triazol-3-yl)-4-fluorophenyl]-6-fluoropyrazolo[1,5-a]pyridine-3- carboxamide. [0331] Step f: A vial was charged with the product from step e (145 mg, 0.276 mmol, 1.0 equiv.) and THF (2 mL). The mixture was cooled to 0 °C and TBAF (1M soln. in THF, 0.83 mL, 0.83 mmol, 3.0 equiv.) was added. The reaction mixture was allowed to warm to rt and stirred for 8 h until complete consumption of starting material was determined by LCMS analysis. The reaction mixture was diluted with water and EtOAc, the layers were separated, and the organic layer was washed with brine (2x). The combined organic layers were collected, dried over Na2SO4 and concentrated in vacuo. The crude product was purified by HPLC to provide the title compound. 1H NMR (400 MHz, DMSO-d6) δ 13.78 (s, 1H), 9.86 (s, 1H), 9.15 (ddd, J = 4.6, 2.3, 0.7 Hz, 1H), 8.66 (s, 1H), 8.35 – 8.10 (m, 1H), 7.99 (d, J = 6.9 Hz, 1H), 7.60 (ddd, J = 10.2, 8.3, 2.3 Hz, 1H), 7.34 (d, J = 11.5 Hz, 1H), 5.49 (s, 1H), 4.55 (d, J = 2.8 Hz, 2H), 2.14 – 1.87 (m, 1H), 1.16 – 0.69 (m, 4H). ESI MS [M+H]+ for C20H17F2N6O2, calcd 411.1, found 411.0. Example 92: 6-Fluoro-N-[4-fluoro-5-[5-(3-hydroxypentan-3-yl)-4H-1,2,4-triazol-3-yl]-2- methylphenyl]pyrazolo[1,5-a]pyridine-3-carboxamide
[0332] Step a: To a solution of methyl 5-bromo-1H-1,2,4-triazole-3-carboxylate (2.96 g, 14.4 mmol, 1.0 equiv.) and pTsOH ^H2O (547 mg, 2.88 mmol, 20 mol%) in THF (21.8 mL, 0.7 M) under N2 at rt was added dihydropyran (3.94 mL, 43.2 mmol, 3.0 equiv.) portion-wise over 5 min. The resulting mixture was heated at reflux for 6 h, then cooled to rt and concentrated under reduced pressure. The resulting residue was treated with sat. aq. NaHCO3 (20 mL) and extracted with CH2Cl2 (3x). The combined organic extracts were dried over Na2SO4, filtered, and concentrated in vacuo to afford the crude product, which contained a mixture of three regioisomers. Purification by column chromatography (0 to 100% EtOAc/hexanes) afforded the desired regioisomeric products, the first eluting of which was used directly in step b. [0333] Step b: A vial was charged with one of the regioisomeric products from step a (211.7 mg, 0.730 mmol, 1.0 equiv.) in THF (2.0 mL, 0.36 M) and placed under N2. To this solution was added Ti(OiPr)4 (0.30 mL, 1.02 mmol, 1.4 equiv.), followed by dropwise addition of EtMgBr (3M in Et2O, 0.68 mL, 2.04 mmol, 2.8 equiv.) over 0.5 h at rt. After the addition of EtMgBr was complete, the reaction mixture was stirred an additional 14 h at rt and then quenched by the dropwise addition of H2O. The mixture was diluted with EtOAc and filtered over Celite®. The
organic phase was separated, dried (Na2SO4), filtered and concentrated in vacuo to afford the crude product, which was used directly in step c without further purification. [0334] Step c: A vial was charged successively with the crude product obtained in step b (51.9 mg, 0.180 mmol, 1.0 equiv.), 6-fluoro-N-[4-fluoro-2-methyl-5-(4,4,5,5-tetramethyl-1,3,2- dioxaborolan-2-yl)phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide (74.4 mg, 0.180 mmol, 1.0 equiv., prepared from 6-fluoropyrazolo[1,5-a]pyridine-3-carboxylic acid and 5-bromo-4-fluoro-2- methylaniline according to Ex.1, steps a-b), Pd XPhos G3 (15.2 mg, 0.018 mmol, 10 mol%), 1M aq. Na2CO3 (0.36 mL, 0.36 mmol, 2.0 equiv.) and dioxane (1.8 mL, 0.1 M). The reaction mixture was evacuated and back-filled with N2 (3x) and then heated at 100 ºC for 1 h. The mixture was then cooled to rt, diluted with EtOAc, filtered over Celite® and concentrated in vacuo. Column chromatography (0 to 100% EtOAc/hexanes) afforded the THP-protected product. [0335] Step d: The product obtained in step c was taken up in 4N HCl in dioxane (1.5 mL) and the mixture was stirred at rt for 14 h. The mixture was then concentrated in vacuo to afford the title compound.1H NMR (400 MHz, DMSO-d6) δ 9.84 (s, 1H), 9.22 – 9.17 (m, 1H), 8.77 (s, 1H), 8.25 (dd, J = 9.9, 5.9 Hz, 1H), 7.92 (d, J = 7.2 Hz, 1H), 7.64 (ddd, J = 10.1, 8.2, 2.2 Hz, 1H), 7.26 (d, J = 11.5 Hz, 1H), 2.28 (s, 3H), 1.82 (dh, J = 28.4, 7.2 Hz, 4H), 0.74 (t, J = 7.4 Hz, 6H). ESI MS [M+H]+ for C22H23F2N6O2, calcd 441.2, found 441.2. Example 93: 6-Fluoro-N-[4-fluoro-5-[5-(1-hydroxycyclopropyl)-4H-1,2,4-triazol-3-yl]-2- methylphenyl]pyrazolo[1,5-a]pyridine-3-carboxamide
[0336] Step a: A vial was charged with 6-fluoro-N-[4-fluoro-2-methyl-5-(4,4,5,5-tetramethyl- 1,3,2-dioxaborolan-2-yl)phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide (500 mg, 1.21 mmol, 1.0 equiv.), 3,5-dibromo-4-(oxan-2-yl)-1,2,4-triazole (747 mg, 2.4 mmol, 2.0 equiv.), Pd XPhos G3 (102 mg, 0.12 mmol, 10 mol%), 2M Na2CO3 (aq. soln., 1.21 mL, 2.0 equiv.) and dioxane (6 mL). The reaction mixture was degassed with N2 and stirred at 95 °C for 6 h, at which time LCMS analysis indicated complete consumption of starting material. The reaction mixture was cooled to rt and diluted with EtOAc and water. The organic layer was separated, dried over Na2SO4 and concentrated in vacuo. The crude product was purified by column chromatography (0% to 70% EtOAc in hexanes) to provide the desired N-[5-[5-bromo-4-(oxan-2-yl)-1,2,4-triazol-3-yl]-4- fluoro-2-methylphenyl]-6-fluoropyrazolo[1,5-a]pyridine-3-carboxamide. [0337] Step b: A vial was charged with the product from step a (60 mg, 0.117 mmol, 1.0 equiv.), 4,4,5,5-tetramethyl-2-[1-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)cyclopropyl]-1,3,2- dioxaborolane (51 mg, 0.17 mmol, 1.5 equiv.), Pd XPhos G3 (10 mg, 0.01 mmol, 10 mol%), Cs2CO3 (76 mg, 0.23 mmol, 2.0 equiv.), dioxane (2.0 mL) and water (0.5 mL). The reaction mixture was degassed with N2 and stirred at 100 °C for 8 h, at which time LCMS analysis indicated partial consumption of starting material. The reaction mixture was cooled to rt, filtered over Celite® and diluted with EtOAc and water. The organic layer was separated, concentrated in vacuo, and the crude product was used directly in the next step without further purification. [0338] Step c: To a solution of the crude product from step b (0.117 mmol, 1.0 equiv.) in 10% aq. NaOH (1 mL) at 0 °C was added H2O2 (30 wt% in water, 0.16 mL, 28 equiv.) dropwise. The reaction mixture was allowed to stir for 0.5 h, then the reaction was quenched by addition of sat. aq. Na2S2O3. The mixture was diluted with EtOAc and water. The organic layer was separated, dried over Na2SO4 and concentrated in vacuo. The crude product was purified by column chromatography (0% to 100% EtOAc in hexanes) to provide 6-fluoro-N-[4-fluoro-5-[5-(1- hydroxycyclopropyl)-4-(oxan-2-yl)-1,2,4-triazol-3-yl]-2-methylphenyl]pyrazolo[1,5-a]pyridine- 3-carboxamide, which was treated with 4N HCl in dioxane (2 mL) and stirred overnight to afford the title compound.1H NMR (400 MHz, Methanol-d4) δ 8.53 (ddd, J = 4.2, 2.4, 0.9 Hz, 2H), 8.30 – 8.22 (m, 1H), 7.85 (dd, J = 29.0, 6.8 Hz, 1H), 7.35 (ddt, J = 10.0, 7.9, 2.4 Hz, 1H), 7.09 (d, J = 11.2 Hz, 1H), 2.31 (s, 3H), 1.24 (d, J = 22.1 Hz, 4H). ESI MS [M+H]+ for C20H17F2N6O2, calcd 411.1, found 411.0.
Example 94: N-[5-(5-Cyclopropyl-1,3,4-oxadiazol-2-yl)-4-fluoro-2- methylphenyl]pyrazolo[1,5-a]pyridine-3-carboxamide
[0339] A vial was charged with triazolo[4,5-b]pyridin-3-yl pyrazolo[1,5-a]pyridine-3- carboxylate (22.5 mg, 0.080 mmol, 1.0 equiv.), 5-(5-cyclopropyl-1,3,4-oxadiazol-2-yl)-4-fluoro- 2-methylaniline (18.7 mg, 0.080 mmol, 1.0 equiv.), which was obtained as a minor byproduct in the synthesis of Ex.12 (step b), and NMP (0.2 mL, 0.5 M). The mixture was heated at 130 ºC for 1 h, then cooled to rt and purified by column chromatography (0 to 50% EtOAc in CH2Cl2). The solid thus obtained was washed successively with hot MeOH (3x) and hot EtOAc (2x), then dried in vacuo to afford the title compound.1H NMR (400 MHz, DMSO-d6) δ 9.79 (s, 1H), 8.85 (d, J = 7.0 Hz, 1H), 8.76 (s, 1H), 8.22 (d, J = 8.9 Hz, 1H), 7.97 (d, J = 6.9 Hz, 1H), 7.58 – 7.49 (m, 1H), 7.44 (d, J = 11.4 Hz, 1H), 7.17 – 7.06 (m, 1H), 2.35 (s, 3H), 2.34 – 2.28 (m, 1H), 1.18 (dt, J = 8.4, 3.1 Hz, 2H), 1.13 – 1.07 (m, 2H). ESI MS [M+H]+ for C20H17FN5O2, calcd 378.1, found 378.1. Example 95: N-[5-(1-Cyclopropyltriazol-4-yl)-4-fluoro-2-methylphenyl]pyrazolo[1,5- a]pyridine-3-carboxamide
[0340] The title compound was prepared from 4-bromo-1-cyclopropyltriazole and N-[4-fluoro- 2-methyl-5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)phenyl]pyrazolo[1,5-a]pyridine-3- carboxamide in a similar fashion to Ex.1 and Ex.67.1H NMR (400 MHz, DMSO-d6) δ 9.68 (s, 1H), 8.81 (dt, J = 7.0, 1.1 Hz, 1H), 8.72 (s, 1H), 8.19 (dt, J = 8.9, 1.3 Hz, 1H), 8.07 (d, J = 3.7 Hz, 1H), 7.90 (d, J = 7.3 Hz, 1H), 7.49 (ddd, J = 8.9, 6.8, 1.1 Hz, 1H), 7.29 (d, J = 11.7 Hz, 1H), 7.08 (td, J = 6.9, 1.4 Hz, 1H), 6.06 (ddt, J = 17.0, 10.3, 5.9 Hz, 1H), 5.31 – 5.02 (m, 4H), 2.27 (s, 3H). ESI MS [M+H]+ for C20H18FN6O, calcd 377.2, found 377.2.
Example 96: N-[5-[1-(2,2-Difluoroethyl)triazol-4-yl]-4-fluoro-2-methylphenyl]pyrazolo[1,5- a]pyridine-3-carboxamide
[0341] The title compound was prepared from 4-bromo-1-(2,2-difluoroethyl)triazole and N-[4- fluoro-2-methyl-5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)phenyl]pyrazolo[1,5-a]pyridine- 3-carboxamide in a similar fashion to Ex.1 and Ex.67.1H NMR (400 MHz, DMSO-d6) δ 9.72 (s, 1H), 8.81 (dt, J = 6.9, 1.1 Hz, 1H), 8.73 (s, 1H), 8.45 (d, J = 3.6 Hz, 1H), 8.20 (dt, J = 8.9, 1.3 Hz, 1H), 8.09 (d, J = 7.3 Hz, 1H), 7.49 (ddd, J = 8.9, 6.8, 1.1 Hz, 1H), 7.34 – 7.25 (m, 1H), 7.08 (td, J = 6.9, 1.5 Hz, 1H), 6.71 – 6.31 (m, 1H), 5.01 (td, J = 15.5, 3.4 Hz, 2H), 2.27 (s, 3H). ESI MS [M+H]+ for C19H16F3N6O, calcd 401.1, found 401.1. Example 97: 6-Chloro-N-[5-[1-(2,2-difluoroethyl)triazol-4-yl]-4-fluoro-2- methylphenyl]pyrazolo[1,5-a]pyridine-3-carboxamide
[0342] The title compound was prepared in a similar fashion to Ex. 1 from 4-bromo-1-(2,2- difluoroethyl)triazole and 6-chloro-N-[4-fluoro-2-methyl-5-(4,4,5,5-tetramethyl-1,3,2- dioxaborolan-2-yl)phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide, which was accessed from 6- chloropyrazolo[1,5-a]pyridine-3-carboxylic acid and 5-bromo-4-fluoro-2-methylaniline in a similar fashion to step b, Ex.1.1H NMR (400 MHz, DMSO-d6) δ 9.81 (s, 1H), 9.19 (dd, J = 1.9, 0.9 Hz, 1H), 8.76 (s, 1H), 8.45 (d, J = 3.6 Hz, 1H), 8.20 (dd, J = 9.5, 0.9 Hz, 1H), 8.07 (d, J = 7.2 Hz, 1H), 7.60 – 7.51 (m, 1H), 7.29 (d, J = 11.6 Hz, 1H), 6.51 (tt, J = 54.3, 3.4 Hz, 1H), 5.01 (td, J = 15.5, 3.4 Hz, 2H), 2.27 (s, 3H). ESI MS [M+H]+ for C19H15ClF3N6O, calcd 435.1, found 435.1. Example 98: N-[5-[1-(2,2-Difluoroethyl)triazol-4-yl]-4-fluoro-2-methylphenyl]-6- methoxypyrazolo[1,5-a]pyridine-3-carboxamide
[0343] The title compound was prepared from 4-bromo-1-(2,2-difluoroethyl)triazole and N-[4- fluoro-2-methyl-5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)phenyl]-6-methoxypyrazolo[1,5- a]pyridine-3-carboxamide in a similar fashion to Ex.1 and Ex.86.1H NMR (400 MHz, DMSO- d6) δ 9.68 (s, 1H), 8.62 (s, 1H), 8.52 (dd, J = 2.2, 0.8 Hz, 1H), 8.45 (d, J = 3.6 Hz, 1H), 8.12 – 8.05 (m, 2H), 7.33 – 7.22 (m, 2H), 6.52 (tt, J = 54.4, 3.4 Hz, 1H), 5.01 (td, J = 15.5, 3.4 Hz, 2H), 3.83 (s, 3H), 2.26 (s, 3H). ESI MS [M+H]+ for C20H18F3N6O2, calcd 431.1, found 431.1. Example 99: N-[5-[1-(2,2-Difluoroethyl)triazol-4-yl]-4-fluoro-2-methylphenyl]-6- fluoropyrazolo[1,5-a]pyridine-3-carboxamide
[0344] The title compound was prepared from 4-bromo-1-(2,2-difluoroethyl)triazole and 6- fluoro-N-[4-fluoro-2-methyl-5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2- yl)phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide in a similar fashion to Ex. 1 and Ex. 76. 1H NMR (400 MHz, DMSO-d6) δ 9.79 (s, 1H), 9.17 (ddd, J = 4.6, 2.3, 0.8 Hz, 1H), 8.75 (s, 1H), 8.45 (d, J = 3.6 Hz, 1H), 8.23 (ddd, J = 9.8, 5.9, 0.8 Hz, 1H), 8.08 (d, J = 7.3 Hz, 1H), 7.61 (ddd, J = 9.7, 8.3, 2.3 Hz, 1H), 7.29 (d, J = 11.6 Hz, 1H), 6.68 – 6.36 (m, 1H), 5.01 (td, J = 15.5, 3.4 Hz, 2H), 2.27 (s, 3H). ESI MS [M+H]+ for C19H15F4N6O, calcd 419.1, found 419.1. Example 100: N-[4-Fluoro-5-[1-(4-fluorophenyl)-1,2,4-triazol-3-yl]-2- methylphenyl]pyrazolo[1,5-a]pyridine-3-carboxamide
[0345] The title compound was prepared from 3-bromo-1-(4-fluorophenyl)-1H-1,2,4-triazole and N-[4-fluoro-2-methyl-5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)phenyl]pyrazolo[1,5-
a]pyridine-3-carboxamide in a similar fashion to Ex.1 and Ex.67.1H NMR (400 MHz, Methanol- d4) δ 9.08 (s, 1H), 8.66 (dt, J = 7.0, 1.1 Hz, 1H), 8.61 (s, 1H), 8.27 (dt, J = 9.0, 1.2 Hz, 1H), 8.05 (d, J = 7.0 Hz, 1H), 7.94 – 7.85 (m, 2H), 7.49 (ddd, J = 9.0, 6.8, 1.1 Hz, 1H), 7.35 – 7.25 (m, 2H), 7.23 (d, J = 11.3 Hz, 1H), 7.09 (td, J = 6.9, 1.4 Hz, 1H), 2.38 (s, 3H). 19F NMR (376 MHz, DMSO- d6) δ -115.62, -117.23. ESI MS [M+H]+ for C23H17F2N6O, calcd 431.1, found 431.1. Example 101: 6-Fluoro-N-[4-fluoro-5-[1-(4-fluorophenyl)-1,2,4-triazol-3-yl]-2- methylphenyl]pyrazolo[1,5-a]pyridine-3-carboxamide
[0346] The title compound was prepared from 3-bromo-1-(4-fluorophenyl)-1H-1,2,4-triazole and 6-fluoro-N-[4-fluoro-2-methyl-5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2- yl)phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide in a similar fashion to Ex. 1 and Ex. 67. 1H NMR (400 MHz, DMSO-d6) δ 9.81 (s, 1H), 9.34 (s, 1H), 9.17 (ddd, J = 4.7, 2.3, 0.8 Hz, 1H), 8.76 (s, 1H), 8.23 (ddd, J = 9.7, 5.9, 0.8 Hz, 1H), 8.04 (d, J = 7.2 Hz, 1H), 7.99 – 7.89 (m, 2H), 7.61 (ddd, J = 9.8, 8.3, 2.2 Hz, 1H), 7.48 – 7.37 (m, 2H), 7.31 (d, J = 11.4 Hz, 1H), 2.29 (s, 3H).19F NMR (376 MHz, DMSO-d6) δ -114.23, -116.11, -138.65. ESI MS [M+H]+ for C23H16F3N6O, calcd 449.1, found 449.1. Example 102: N-[4-Fluoro-5-[1-(4-fluorophenyl)-1,2,4-triazol-3-yl]-2-methylphenyl]-6- methoxypyrazolo[1,5-a]pyridine-3-carboxamide
[0347] The title compound was prepared from 3-bromo-1-(4-fluorophenyl)-1H-1,2,4-triazole and N-[4-fluoro-2-methyl-5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)phenyl]-6- methoxypyrazolo[1,5-a]pyridine-3-carboxamide in a similar fashion to Ex.1 and Ex.86.1H NMR (400 MHz, DMSO-d6) δ 9.69 (s, 1H), 9.34 (s, 1H), 8.63 (s, 1H), 8.52 (dd, J = 2.2, 0.7 Hz, 1H), 8.12 – 8.00 (m, 2H), 7.99 – 7.89 (m, 2H), 7.48 – 7.36 (m, 2H), 7.34 – 7.25 (m, 2H), 3.83 (s, 3H),
2.29 (s, 3H). 19F NMR (376 MHz, DMSO-d6) δ -114.24, -115.31. ESI MS [M+H]+ for C24H19F2N6O2, calcd 461.1, found 461.1. Example 103: N-[5-(2-Cyclopropyl-1,3-thiazol-4-yl)-4-fluoro-2-methylphenyl]-6- fluoropyrazolo[1,5-a]pyridine-3-carboxamide
[0348] The title compound was prepared from 4-bromo-2-cyclopropyl-1,3-thiazole and 6- fluoro-N-[4-fluoro-2-methyl-5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2- yl)phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide in a similar fashion to Ex. 1 and Ex. 76. 1H NMR (400 MHz, DMSO-d6) δ 9.81 (s, 1H), 9.16 (ddd, J = 4.6, 2.3, 0.7 Hz, 1H), 8.74 (s, 1H), 8.21 (ddd, J = 9.8, 5.9, 0.7 Hz, 1H), 7.97 (d, J = 7.5 Hz, 1H), 7.68 (d, J = 2.5 Hz, 1H), 7.60 (ddd, J = 9.8, 8.3, 2.2 Hz, 1H), 7.27 – 7.19 (m, 1H), 2.45 – 2.38 (m, 1H), 2.23 (s, 3H), 1.14 – 1.08 (m, 2H), 1.01 – 0.93 (m, 2H). ESI MS [M+H]+ for C21H17F2N4OS, calcd 411.1, found 411.1. Example 104: N-[5-(5-Cyclopropyl-1,2,4-thiadiazol-3-yl)-4-fluoro-2-methylphenyl]-6- fluoropyrazolo[1,5-a]pyridine-3-carboxamide
[0349] The title compound was prepared from 3-bromo-5-cyclopropyl-1,2,4-thiadiazole and 6- fluoro-N-[4-fluoro-2-methyl-5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2- yl)phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide in a similar fashion to Ex. 1 and Ex. 76. 1H NMR (400 MHz, DMSO-d6) δ 9.83 (s, 1H), 9.16 (ddd, J = 4.6, 2.2, 0.8 Hz, 1H), 8.74 (s, 1H), 8.21 (ddd, J = 9.8, 5.9, 0.8 Hz, 1H), 8.01 (d, J = 7.3 Hz, 1H), 7.61 (ddd, J = 9.8, 8.3, 2.3 Hz, 1H), 7.29 (dd, J = 11.5, 0.8 Hz, 1H), 2.74 (tt, J = 8.1, 4.7 Hz, 1H), 2.28 (s, 3H), 1.32 – 1.25 (m, 2H), 1.16 – 1.10 (m, 2H). ESI MS [M+H]+ for C20H16F2N5OS, calcd 412.1, found 412.1. Example 105: N-[5-(5-Cyclopropyl-1,3,4-thiadiazol-2-yl)-4-fluoro-2-methylphenyl]-6- fluoropyrazolo[1,5-a]pyridine-3-carboxamide
[0350] The title compound was prepared from 2-bromo-5-cyclopropyl-1,3,4-thiadiazole and 6- fluoro-N-[4-fluoro-2-methyl-5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2- yl)phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide in a similar fashion to Ex. 1 and Ex. 76. 1H NMR (400 MHz, DMSO-d6) δ 9.85 (s, 1H), 9.17 (ddd, J = 4.6, 2.3, 0.8 Hz, 1H), 8.75 (s, 1H), 8.22 (ddd, J = 9.8, 5.9, 0.8 Hz, 1H), 8.16 (d, J = 7.0 Hz, 1H), 7.62 (ddd, J = 9.8, 8.2, 2.2 Hz, 1H), 7.41 (dd, J = 11.6, 0.8 Hz, 1H), 2.57 (tt, J = 8.3, 4.9 Hz, 1H), 2.31 (s, 3H), 1.26 – 1.20 (m, 2H), 1.10 – 1.04 (m, 2H). ESI MS [M+H]+ for C20H16F2N5OS, calcd 412.1, found 412.1. Example 106: N-[5-(3-Cyclopropyl-1,2,4-thiadiazol-5-yl)-4-fluoro-2-methylphenyl]-6- fluoropyrazolo[1,5-a]pyridine-3-carboxamide
[0351] The title compound was prepared from 5-bromo-3-cyclopropyl-1,2,4-thiadiazole and 6- fluoro-N-[4-fluoro-2-methyl-5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2- yl)phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide in a similar fashion to Ex. 1 and Ex. 76. 1H NMR (400 MHz, DMSO-d6) δ 9.90 (s, 1H), 9.17 (ddd, J = 4.6, 2.3, 0.8 Hz, 1H), 8.76 (s, 1H), 8.22 (ddd, J = 9.8, 5.9, 0.8 Hz, 1H), 8.16 (d, J = 7.0 Hz, 1H), 7.62 (ddd, J = 9.8, 8.2, 2.3 Hz, 1H), 7.47 (dd, J = 11.6, 0.8 Hz, 1H), 2.41 – 2.33 (m, 1H), 2.32 (s, 3H), 1.11 – 1.02 (m, 4H). ESI MS [M+H]+ for C20H16F2N5OS, calcd 412.1, found 412.1. Example 107: N-[5-[5-(Cyclopropylamino)-1,2,4-thiadiazol-3-yl]-4-fluoro-2-methylphenyl]- 6-fluoropyrazolo[1,5-a]pyridine-3-carboxamide
[0352] The title compound was prepared from 3-bromo-N-cyclopropyl-1,2,4-thiadiazol-5- amine and 6-fluoro-N-[4-fluoro-2-methyl-5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)phenyl] pyrazolo[1,5-a]pyridine-3-carboxamide in a similar fashion to Ex. 1 and Ex. 76. 1H NMR (400 MHz, DMSO-d6) δ 9.75 (s, 1H), 9.16 (ddd, J = 4.6, 2.3, 0.8 Hz, 1H), 9.06 (s, 1H), 8.74 (s, 1H), 8.21 (ddd, J = 9.8, 5.9, 0.8 Hz, 1H), 7.97 (d, J = 7.3 Hz, 1H), 7.61 (ddd, J = 9.8, 8.3, 2.3 Hz, 1H), 7.23 (d, J = 11.7 Hz, 1H), 2.61 (s, 1H), 2.27 (s, 3H), 0.75 (td, J = 6.9, 4.7 Hz, 2H), 0.61 – 0.54 (m, 2H). ESI MS [M+H]+ for C20H17F2N6OS, calcd 427.1, found 427.1. Example 108: N-[5-(4-Cyclopropyl-1H-imidazol-2-yl)-4-fluoro-2- methylphenyl]pyrazolo[1,5-a]pyridine-3-carboxamide
[0353] The title compound was prepared from 2-bromo-4-cyclopropyl-1H-imidazole and N-[4- fluoro-2-methyl-5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)phenyl]pyrazolo[1,5-a]pyridine- 3-carboxamide in a similar fashion to Ex.1 and Ex.67.1H NMR (400 MHz, Methanol-d4) δ 8.69 (dt, J = 7.0, 1.1 Hz, 1H), 8.63 (s, 1H), 8.33 – 8.26 (m, 1H), 7.92 (d, J = 7.0 Hz, 1H), 7.53 (ddd, J = 8.9, 6.9, 1.1 Hz, 1H), 7.45 – 7.39 (m, 1H), 7.33 (d, J = 0.9 Hz, 1H), 7.12 (td, J = 6.9, 1.4 Hz, 1H), 2.45 (t, J = 0.5 Hz, 3H), 2.06 – 1.96 (m, 1H), 1.15 – 1.06 (m, 2H), 0.91 – 0.79 (m, 2H). ESI MS [M+H]+ for C21H19FN5O, calcd 376.2, found 376.1. Example 109: N-[5-(4-Cyclopropyl-1H-imidazol-2-yl)-4-fluoro-2-methylphenyl]-6- fluoropyrazolo[1,5-a]pyridine-3-carboxamide
[0354] The title compound was prepared from 2-bromo-4-cyclopropyl-1H-imidazole and 6- fluoro-N-[4-fluoro-2-methyl-5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2- yl)phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide in a similar fashion to Ex. 1 and Ex. 76. 1H NMR (400 MHz, Methanol-d4) δ 8.59 (dd, J = 6.9, 0.8 Hz, 1H), 8.51 (s, 1H), 8.06 (d, J = 7.0 Hz,
1H), 7.41 (dd, J = 11.2, 0.8 Hz, 1H), 7.37 – 7.27 (m, 2H), 7.13 – 7.03 (m, 1H), 2.46 (s, 3H), 2.05 – 1.96 (m, 1H), 1.14 – 1.04 (m, 2H), 0.91 – 0.83 (m, 2H). ESI MS [M+H]+ for C21H18F2N5O, calcd 394.1, found 394.1. Example 110: N-[5-(1-Cyclopropylimidazol-4-yl)-4-fluoro-2-methylphenyl]pyrazolo[1,5- a]pyridine-3-carboxamide
[0355] The title compound was prepared from 4-bromo-1-cyclopropylimidazole and N-[4- fluoro-2-methyl-5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)phenyl]pyrazolo[1,5-a]pyridine- 3-carboxamide in a similar fashion to Ex.1 and Ex.67.1H NMR (400 MHz, DMSO-d6) δ 9.66 (s, 1H), 8.80 (dt, J = 7.0, 1.1 Hz, 1H), 8.71 (s, 1H), 8.19 (dt, J = 8.9, 1.3 Hz, 1H), 7.94 (d, J = 7.5 Hz, 1H), 7.78 (s, 1H), 7.51 – 7.43 (m, 2H), 7.19 – 7.11 (m, 1H), 7.07 (td, J = 6.9, 1.4 Hz, 1H), 3.53 (tt, J = 7.3, 3.6 Hz, 1H), 2.21 (s, 3H), 1.04 – 0.90 (m, 4H). ESI MS [M+H]+ for C21H19FN5O, calcd 376.2, found 376.1. Example 111: N-[4-Fluoro-2-methyl-5-(3-methyl-1,2,4-oxadiazol-5-yl)phenyl]pyrazolo[1,5- a]pyridine-3-carboxamide
[0356] The title compound was prepared from 5-bromo-3-methyl-1,2,4-oxadiazole and N-[4- fluoro-2-methyl-5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)phenyl]pyrazolo[1,5-a]pyridine- 3-carboxamide e in a similar fashion to Ex.1 and Ex.67.1H NMR (400 MHz, DMSO-d6) δ 9.74 (s, 1H), 8.82 (dt, J = 7.0, 1.0 Hz, 1H), 8.73 (s, 1H), 8.20 (dt, J = 8.9, 1.3 Hz, 1H), 8.14 (d, J = 7.0 Hz, 1H), 7.54 – 7.41 (m, 2H), 7.10 (td, J = 6.9, 1.4 Hz, 1H), 2.40 (s, 3H), 2.36 (s, 3H). ESI MS [M+H]+ for C18H15FN5O2, calcd 352.1, found 352.1. Example 112: N-[5-[1-(2,2-Difluoroethyl)pyrazol-4-yl]-4-fluoro-2-methylphenyl]-6- methylpyrazolo[1,5-a]pyridine-3-carboxamide
[0357] Step a: A 20 mL vial was charged with 5-bromo-4-fluoro-2-methylaniline (0.395 g, 1.938 mmol, 1.0 equiv.), 1-(2,2-difluoroethyl)-4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2- yl)pyrazole (0.5 g, 1.938 mmol, 1.0 equiv.), Pd(dppf)Cl2 (0.141 g, 0.193 mmol, 10 mol%), and Na2CO3 (2.91 mL, 2M aq. solution, 3.0 equiv.) in of 1,4-dioxane (6 mL). The resulting mixture was degassed by evacuation and back-filling with N2 (3x). The mixture was then stirred at 95 °C under N2 for 2 h. Upon complete conversion, as judged by LCMS analysis, the reaction mixture was cooled to rt and filtered over Celite®. The filtrate was concentrated in vacuo. The crude product was purified by column chromatography (0% to 80% EtOAc in hexanes) to afford the desired 5-[1-(2,2-difluoroethyl)pyrazol-4-yl]-4-fluoro-2-methylaniline. [0358] Step b: A mixture of 5-[1-(2,2-difluoroethyl)pyrazol-4-yl]-4-fluoro-2-methylaniline (0.090 g, 0.306 mmol, 1.0 equiv.) and triazolo[4,5-b]pyridin-3-yl 6-methylpyrazolo[1,5- a]pyridine-3-carboxylate (0.078 g, 0.306 mmol, 1.0 equiv., prepared according to Ex.1, step a) in NMP (2.0 mL) was stirred at 120 °C for 6 h. Upon complete conversion, as judged by LCMS analysis, the reaction mixture was cooled to rt and diluted with EtOAc (20 mL) and H2O (20 mL). The organic phase was separated, dried over Na2SO4, filtered and concentrated in vacuo. The crude product was purified by HPLC to provide the title compound.1H NMR (400 MHz, Methanol-d4) δ 8.57 – 8.44 (m, 2H), 8.16 (dd, J = 9.1, 0.9 Hz, 1H), 8.09 (d, J = 1.9 Hz, 1H), 7.94 (dd, J = 1.4, 0.7 Hz, 1H), 7.62 (d, J = 7.4 Hz, 1H), 7.38 (dd, J = 9.1, 1.5 Hz, 1H), 7.16 – 7.05 (m, 1H), 6.19 (tt, J = 55.2, 3.9 Hz, 1H), 4.59 (td, J = 14.4, 3.9 Hz, 2H), 2.39 (d, J = 1.0 Hz, 3H), 2.30 (d, J = 0.6 Hz, 3H). ESI MS [M+H]+ for C21H19F3N5O, calcd 414.2, found 414.1.
Example 113: N-[5-[1-(2,2-Difluoroethyl)pyrazol-4-yl]-4-fluoro-2- methylphenyl]pyrazolo[1,5-a]pyridine-3-carboxamide
[0359] The title compound was prepared from triazolo[4,5-b]pyridin-3-yl pyrazolo[1,5- a]pyridine-3-carboxylate in a similar fashion to Ex.112.1H NMR (400 MHz, DMSO-d6) δ 9.68 (s, 1H), 8.83 (dt, J = 7.0, 1.1 Hz, 1H), 8.75 (s, 1H), 8.26 – 8.19 (m, 2H), 7.99 (s, 1H), 7.68 (d, J = 7.6 Hz, 1H), 7.51 (ddd, J = 8.9, 6.8, 1.1 Hz, 1H), 7.22 (d, J = 11.7 Hz, 1H), 7.11 (td, J = 6.9, 1.5 Hz, 1H), 6.41 (tt, J = 55.0, 3.8 Hz, 1H), 4.69 (td, J = 15.1, 3.8 Hz, 2H), 2.25 (s, 3H).19F NMR (376 MHz, DMSO-d6) δ -119.21, -122.72. ESI MS [M+H]+ for C20H17F3N5O, calcd 400.1, found 400.1. Example 114: N-[5-[1-(2,2-Difluoroethyl)pyrazol-4-yl]-4-methoxy-2- methylphenyl]pyrazolo[1,5-a]pyridine-3-carboxamide
[0360] The title compound was prepared from 5-bromo-4-methoxy-2-methylaniline and triazolo[4,5-b]pyridin-3-yl pyrazolo[1,5-a]pyridine-3-carboxylate in a similar fashion to Ex.112. 1H NMR (400 MHz, DMSO-d6) δ 9.55 (s, 1H), 8.79 (dt, J = 7.0, 1.1 Hz, 1H), 8.71 (s, 1H), 8.22 – 8.15 (m, 2H), 7.94 (d, J = 0.7 Hz, 1H), 7.51 – 7.42 (m, 2H), 7.06 (td, J = 6.9, 1.4 Hz, 1H), 6.96 (s, 1H), 6.35 (tt, J = 55.0, 3.8 Hz, 1H), 4.61 (td, J = 15.0, 3.9 Hz, 2H), 3.86 (s, 3H), 2.22 (s, 3H). ESI MS [M+H]+ for C21H20F2N5O2, calcd 412.2, found 412.1. Example 115: N-[4-Chloro-5-[1-(2,2-difluoroethyl)pyrazol-4-yl]-2- methylphenyl]pyrazolo[1,5-a]pyridine-3-carboxamide
[0361] The title compound was prepared from 5-bromo-4-chloro-2-methylaniline and triazolo[4,5-b]pyridin-3-yl pyrazolo[1,5-a]pyridine-3-carboxylate in a similar fashion to Ex.112. 1H NMR (400 MHz, DMSO-d6) δ 9.68 (s, 1H), 8.84 (dt, J = 6.9, 1.1 Hz, 1H), 8.76 (s, 1H), 8.30 – 8.18 (m, 2H), 7.93 (d, J = 0.7 Hz, 1H), 7.64 (s, 1H), 7.52 (ddd, J = 9.0, 6.8, 1.1 Hz, 1H), 7.46 (d, J = 0.8 Hz, 1H), 7.12 (td, J = 6.9, 1.4 Hz, 1H), 6.41 (tt, J = 54.9, 3.9 Hz, 1H), 4.69 (td, J = 15.1, 3.8 Hz, 2H), 2.27 (s, 3H). 19F NMR (376 MHz, DMSO-d6) δ -122.71. ESI MS [M+H]+ for C20H17ClF2N5O, calcd 416.1, found 416.1. Example 116: N-[5-[1-(2,2-Difluoroethyl)pyrazol-4-yl]-4-fluoro-2-methylphenyl]-6- methoxypyrazolo[1,5-a]pyridine-3-carboxamide
[0362] The title compound was prepared from triazolo[4,5-b]pyridin-3-yl 6- methoxypyrazolo[1,5-a]pyridine-3-carboxylate in a similar fashion to Ex. 112. 1H NMR (400 MHz, Methanol-d4) δ 8.47 (s, 1H), 8.27 (dd, J = 2.2, 0.7 Hz, 1H), 8.15 – 8.03 (m, 2H), 7.92 (dd, J = 1.5, 0.7 Hz, 1H), 7.61 (d, J = 7.4 Hz, 1H), 7.24 (dd, J = 9.7, 2.2 Hz, 1H), 7.08 (dd, J = 11.6, 0.9 Hz, 1H), 6.18 (tt, J = 55.2, 3.9 Hz, 1H), 4.58 (td, J = 14.4, 3.9 Hz, 2H), 3.87 (s, 3H), 2.28 (d, J = 0.6 Hz, 3H). ESI MS [M+H]+ for C21H19F3N5O2, calcd 430.1, found 430.1. Example 117: 6-Chloro-N-[5-[1-(2,2-Difluoroethyl)pyrazol-4-yl]-4-fluoro-2- methylphenyl]pyrazolo[1,5-a]pyridine-3-carboxamide
[0363] The title compound was prepared from triazolo[4,5-b]pyridin-3-yl 6-chloropyrazolo[1,5- a]pyridine-3-carboxylate in a similar fashion to Ex.112.1H NMR (400 MHz, DMSO-d6) δ 9.75 (s, 1H), 9.19 (dd, J = 1.9, 0.8 Hz, 1H), 8.76 (s, 1H), 8.27 – 8.12 (m, 2H), 7.96 (t, J = 1.0 Hz, 1H), 7.64 (dd, J = 7.6, 2.4 Hz, 1H), 7.55 (dd, J = 9.5, 1.9 Hz, 1H), 7.26 – 7.12 (m, 1H), 6.58 – 6.20 (m,
1H), 4.65 (td, J = 15.1, 3.8 Hz, 2H), 2.25 – 2.14 (m, 3H). ESI MS [M+H]+ for C20H16ClF3N5O, calcd 434.1, found 434.1. Example 118: N-[5-[1-(2,2-Difluoroethyl)pyrazol-4-yl]-4-fluoro-2-methylphenyl]-7- methylpyrazolo[1,5-a]pyridine-3-carboxamide
[0364] The title compound was prepared from triazolo[4,5-b]pyridin-3-yl 7- methylpyrazolo[1,5-a]pyridine-3-carboxylate in a similar fashion to Ex.112.1H NMR (400 MHz, Methanol-d4) δ 8.64 (s, 1H), 8.18 (ddd, J = 8.9, 1.4, 0.7 Hz, 1H), 8.12 – 8.06 (m, 1H), 7.95 (dd, J = 1.4, 0.7 Hz, 1H), 7.63 (d, J = 7.4 Hz, 1H), 7.43 (dd, J = 8.9, 6.9 Hz, 1H), 7.11 (dd, J = 11.6, 0.9 Hz, 1H), 6.97 (dt, J = 6.9, 1.2 Hz, 1H), 6.19 (tt, J = 55.2, 3.9 Hz, 1H), 4.59 (td, J = 14.4, 3.9 Hz, 2H), 2.78 (d, J = 0.8 Hz, 3H), 2.31 (d, J = 0.6 Hz, 3H). ESI MS [M+H]+ for C21H19F3N5O, calcd 414.2, found 414.1. Example 119: N-[5-[1-(2,2-Difluoroethyl)pyrazol-4-yl]-2,4-dimethylphenyl]-6- methylpyrazolo[1,5-a]pyridine-3-carboxamide
[0365] The title compound was prepared from 5-bromo-2,4-dimethylaniline and triazolo[4,5- b]pyridin-3-yl 6-methylpyrazolo[1,5-a]pyridine-3-carboxylate in a similar fashion to Ex.112.1H NMR (400 MHz, DMSO-d6) δ 9.51 (s, 1H), 8.63 (dd, J = 2.4, 1.2 Hz, 2H), 8.08 (dd, J = 9.1, 0.9 Hz, 1H), 8.00 (d, J = 0.7 Hz, 1H), 7.73 (d, J = 0.8 Hz, 1H), 7.36 – 7.30 (m, 2H), 7.12 (s, 1H), 6.37 (tt, J = 55.0, 3.9 Hz, 1H), 4.63 (td, J = 15.0, 3.9 Hz, 2H), 2.33 – 2.29 (m, 6H), 2.19 (s, 3H). ESI MS [M+H]+ for C22H22F2N5O, calcd 410.2, found 410.2. Example 120: 6-Chloro-N-[5-[1-(2,2-difluoroethyl)pyrazol-4-yl]-2,4- dimethylphenyl]pyrazolo[1,5-a]pyridine-3-carboxamide
[0366] The title compound was prepared from 5-bromo-2,4-dimethylaniline and triazolo[4,5- b]pyridin-3-yl 6-chloropyrazolo[1,5-a]pyridine-3-carboxylate in a similar fashion to Ex.112.1H NMR (400 MHz, DMSO-d6) δ 9.66 (s, 1H), 9.17 (dd, J = 1.9, 0.9 Hz, 1H), 8.75 (s, 1H), 8.18 (dd, J = 9.5, 0.9 Hz, 1H), 8.00 (d, J = 0.8 Hz, 1H), 7.74 (d, J = 0.8 Hz, 1H), 7.54 (dd, J = 9.5, 1.8 Hz, 1H), 7.33 (s, 1H), 7.13 (s, 1H), 6.37 (tt, J = 55.0, 3.8 Hz, 1H), 4.63 (td, J = 15.0, 3.9 Hz, 2H), 2.32 (s, 3H), 2.19 (s, 3H). ESI MS [M+H]+ for C21H19ClF2N5O, calcd 430.1, found 430.1. Example 121: N-[5-[1-(2,2-Difluoroethyl)pyrazol-4-yl]-2,4-dimethylphenyl]-6- fluoropyrazolo[1,5-a]pyridine-3-carboxamide
[0367] The title compound was prepared from 5-bromo-2,4-dimethylaniline and triazolo[4,5- b]pyridin-3-yl 6-fluoropyrazolo[1,5-a]pyridine-3-carboxylate in a similar fashion to Ex.112.1H NMR (400 MHz, DMSO-d6) δ 9.56 (s, 1H), 8.70 (dd, J = 6.9, 0.7 Hz, 1H), 8.52 (s, 1H), 7.99 (d, J = 0.9 Hz, 1H), 7.71 (d, J = 0.8 Hz, 1H), 7.44 (s, 1H), 7.32 (ddd, J = 11.1, 7.8, 0.8 Hz, 1H), 7.12 (s, 1H), 7.07 – 7.00 (m, 1H), 6.38 (tt, J = 55.0, 3.9 Hz, 1H), 4.63 (td, J = 15.0, 3.9 Hz, 2H), 2.31 (s, 3H), 2.21 (s, 3H). ESI MS [M+H]+ for C21H19F3N5O, calcd 414.2, found 414.1. Example 122: N-[5-[1-(2,2-Difluoroethyl)pyrazol-4-yl]-2,4-dimethylphenyl]-6- methoxypyrazolo[1,5-a]pyridine-3-carboxamide
[0368] The title compound was prepared from 5-bromo-2,4-dimethylaniline and triazolo[4,5- b]pyridin-3-yl 6-methoxypyrazolo[1,5-a]pyridine-3-carboxylate in a similar fashion to Ex. 112. 1H NMR (400 MHz, DMSO-d6) δ 9.52 (s, 1H), 8.61 (s, 1H), 8.50 (dd, J = 2.2, 0.8 Hz, 1H), 8.07
(dd, J = 9.7, 0.7 Hz, 1H), 8.00 (d, J = 0.8 Hz, 1H), 7.73 (d, J = 0.8 Hz, 1H), 7.33 (s, 1H), 7.26 (dd, J = 9.6, 2.2 Hz, 1H), 7.12 (s, 1H), 6.37 (tt, J = 55.0, 3.8 Hz, 1H), 4.63 (td, J = 15.0, 3.9 Hz, 2H), 3.83 (s, 3H), 2.32 (s, 3H), 2.19 (s, 3H). ESI MS [M+H]+ for C22H22F2N5O2, calcd 426.2, found 426.1. Example 123: N-[4-Cyclopropyl-5-[1-(2,2-difluoroethyl)pyrazol-4-yl]-2- methylphenyl]pyrazolo[1,5-a]pyridine-3-carboxamide
[0369] Step a: A vial was charged with 5-bromo-4-chloro-2-methylaniline (340 mg, 1.55 mmol, 1.0 equiv.), 1-(2,2-difluoroethyl)-4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)pyrazole (400 mg, 1.55 mmol, 1.0 equiv.), Pd(dppf)Cl2 (117 mg, 0.16 mmol, 10 mol%), Na2CO3 (328 mg, 3.10 mmol, 2.0 equiv.), dioxane (6 mL) and water (1.5 mL). The reaction mixture was degassed with N2, and it was stirred at 95 °C for 2 h, at which time LCMS analysis indicated complete consumption of starting material. The reaction mixture was cooled to rt and diluted with EtOAc. The organic layer was separated, filtered over Celite®, and concentrated in vacuo. The crude product was purified by column chromatography (0% to 60% EtOAc in hexanes) to provide the desired 4-chloro-5-[1-(2,2-difluoroethyl)pyrazol-4-yl]-2-methylaniline. [0370] Step b: A vial was charged with product from step a (100 mg, 0.37 mmol, 1.0 equiv.), cyclopropylboronic acid (47 mg, 0.55 mmol, 1.5 equiv.), PCy3 Pd G2 (22 mg, 0.037 mmol, 10 mol%), K3PO4 (234 mg, 1.10 mmol, 3.0 equiv.), toluene (2.0 mL) and water (0.2 mL). The reaction mixture was degassed with N2, and it was stirred at 100 °C for 3 h, at which time LCMS analysis
indicated partial consumption of starting material. The reaction mixture was cooled to rt and diluted with EtOAc. The organic layer was separated, filtered over Celite®, and concentrated in vacuo. The crude product was purified by column chromatography (0% to 25% EtOAc in CH2Cl2) to provide the desired 4-cyclopropyl-5-[1-(2,2-difluoroethyl)pyrazol-4-yl]-2-methylaniline. [0371] Step c: A mixture of 4-cyclopropyl-5-[1-(2,2-difluoroethyl)pyrazol-4-yl]-2- methylaniline (28 mg, 0.10 mmol, 1.0 equiv.), and triazolo[4,5-b]pyridin-3-yl pyrazolo[1,5- a]pyridine-3-carboxylate (28 mg, 0.10 mmol, 1.0 equiv.) in NMP (1.0 mL) was stirred at 120 °C for 1 h. Upon complete conversion, as judged by LCMS analysis, the reaction mixture was cooled to rt and diluted with water. The resulting precipitated solid was collected by vacuum filtration, rinsed with water, and dried in vacuo. The crude product was purified by HPLC to provide the title compound.1H NMR (400 MHz, DMSO-d6) δ 9.55 (s, 1H), 8.79 (dt, J = 7.0, 1.1 Hz, 1H), 8.71 (s, 1H), 8.17 (dt, J = 8.9, 1.3 Hz, 1H), 8.07 (d, J = 0.8 Hz, 1H), 7.79 (d, J = 0.7 Hz, 1H), 7.46 (ddd, J = 8.9, 6.8, 1.1 Hz, 1H), 7.34 (s, 1H), 7.06 (td, J = 6.9, 1.5 Hz, 1H), 6.92 (s, 1H), 6.37 (tt, J = 55.0, 3.9 Hz, 1H), 4.68 – 4.59 (m, 2H), 2.19 (s, 3H), 1.96 (tt, J = 8.5, 5.4 Hz, 1H), 0.96 – 0.87 (m, 2H), 0.70 – 0.61 (m, 2H). ESI MS [M+H]+ for C23H22F2N5O, calcd 422.2, found 422.2. Example 124: N-[4-Cyano-5-[1-(2,2-difluoroethyl)pyrazol-4-yl]-2- methylphenyl]pyrazolo[1,5-a]pyridine-3-carboxamide
[0372] A vial was charged with N-[4-chloro-5-[1-(2,2-difluoroethyl)pyrazol-4-yl]-2- methylphenyl] pyrazolo[1,5-a]pyridine-3-carboxamide (98.5 mg, 0.237 mmol, 1.0 equiv.), K4Fe(CN)6 .3H2O (70.1 mg, 0.166 mmol, 0.7 equiv.), XPhos Pd G3 (20 mg, 0.0237 mmol, 10 mol%), and XPhos (11.2 mg, 0.028 mmol, 10 mol%) in 1,4-dioxane (2 mL) and H2O (2 mL). The reaction mixture was degassed by evacuation and back-filling with N2 (3x). The mixture was then stirred at 100 °C under N2 for 5 h. Upon complete conversion, as judged by LCMS analysis, the reaction mixture was cooled to rt and filtered over Celite®. The filtrate was concentrated in vacuo. The crude product was purified by column chromatography (0% to 100% EtOAc in hexanes) to afford the title compound.1H NMR (400 MHz, DMSO-d6) δ 9.72 (s, 1H), 8.83 (dt, J = 6.9, 1.1
Hz, 1H), 8.77 (s, 1H), 8.32 (d, J = 0.8 Hz, 1H), 8.20 (dt, J = 8.9, 1.3 Hz, 1H), 8.00 (d, J = 0.8 Hz, 1H), 7.88 (s, 1H), 7.77 (d, J = 1.0 Hz, 1H), 7.52 (ddd, J = 8.9, 6.8, 1.1 Hz, 1H), 7.11 (td, J = 6.9, 1.5 Hz, 1H), 6.39 (tt, J = 54.9, 3.8 Hz, 1H), 4.71 (td, J = 15.1, 3.7 Hz, 2H), 2.31 (s, 3H). ESI MS [M+H]+ for C21H17F2N6O, calcd 407.1, found 407.1. Example 125: N-[5-[1-(2,2-Difluoroethyl)-1,2,4-triazol-3-yl]-4-fluoro-2- methylphenyl]pyrazolo[1,5-a]pyridine-3-carboxamide
[0373] The title compound was prepared from 1-(2,2-difluoroethyl)-3-(4,4,5,5-tetramethyl- 1,3,2-dioxaborolan-2-yl)-1,2,4-triazole and triazolo[4,5-b]pyridin-3-yl pyrazolo[1,5-a]pyridine- 3-carboxylate in a similar fashion to Ex.112.1H NMR (400 MHz, DMSO-d6) δ 9.70 (s, 1H), 8.81 (dt, J = 6.9, 1.1 Hz, 1H), 8.72 (s, 1H), 8.65 (s, 1H), 8.24 – 8.13 (m, 1H), 7.97 (d, J = 7.2 Hz, 1H), 7.48 (ddd, J = 8.9, 6.8, 1.1 Hz, 1H), 7.26 (d, J = 11.7 Hz, 1H), 7.08 (td, J = 6.9, 1.4 Hz, 1H), 6.44 (tt, J = 54.6, 3.4 Hz, 1H), 4.80 (td, J = 15.2, 3.5 Hz, 2H), 2.28 (s, 3H). ESI MS [M+H]+ for C19H16F3N6O, calcd 401.1, found 401.1. Example 126: N-[5-[1-(2,2-Difluoroethyl)-1,2,4-triazol-3-yl]-2,4-dimethylphenyl]-6- methoxypyrazolo[1,5-a]pyridine-3-carboxamide
[0374] The title compound was prepared from 1-(2,2-difluoroethyl)-3-(4,4,5,5-tetramethyl- 1,3,2-dioxaborolan-2-yl)-1,2,4-triazole, 5-bromo-2,4-dimethylaniline, and triazolo[4,5-b]pyridin- 3-yl 6-methoxypyrazolo[1,5-a]pyridine-3-carboxylate in a similar fashion to Ex. 112. 1H NMR (400 MHz, Methanol-d4) δ 8.54 (s, 1H), 8.48 (s, 2H), 8.35 – 8.24 (m, 1H), 8.12 (dd, J = 9.7, 0.7 Hz, 1H), 7.80 (s, 1H), 7.26 (d, J = 2.2 Hz, 1H), 7.25 – 7.21 (m, 1H), 6.50 – 6.05 (m, 1H), 4.72 (td, J = 14.3, 3.8 Hz, 2H), 3.88 (s, 3H), 2.55 (s, 3H), 2.31 (s, 3H). ESI MS [M+H]+ for C21H21F2N6O2, calcd 427.2, found 427.3.
Example 127: N-[5-[1-(2,2-Difluoroethyl)-1,2,4-triazol-3-yl]-4-fluoro-2-methylphenyl]-6- methylpyrazolo[1,5-a]pyridine-3-carboxamide
[0375] The title compound was prepared from 1-(2,2-difluoroethyl)-3-(4,4,5,5-tetramethyl- 1,3,2-dioxaborolan-2-yl)-1,2,4-triazole in a similar fashion to Ex. 112. 1H NMR (400 MHz, DMSO-d6) δ 9.64 (s, 1H), 8.65 (d, J = 2.4 Hz, 3H), 8.09 (dd, J = 9.0, 0.9 Hz, 1H), 7.96 (d, J = 7.2 Hz, 1H), 7.36 (dd, J = 9.2, 1.5 Hz, 1H), 7.25 (d, J = 11.5 Hz, 1H), 6.59 – 6.27 (m, 1H), 4.80 (td, J = 15.3, 3.5 Hz, 2H), 2.32 (d, J = 1.2 Hz, 3H), 2.27 (s, 3H). ESI MS [M+H]+ for C20H18F3N6O, calcd 415.1, found 415.2. Example 128: N-[5-[1-(2,2-Difluoroethyl)-1,2,4-triazol-3-yl]-2,4-dimethylphenyl]-6- methylpyrazolo[1,5-a]pyridine-3-carboxamide
[0376] The title compound was prepared from 1-(2,2-difluoroethyl)-3-(4,4,5,5-tetramethyl- 1,3,2-dioxaborolan-2-yl)-1,2,4-triazole and 5-bromo-2,4-dimethylaniline in a similar fashion to Ex.112.1H NMR (400 MHz, DMSO-d6) δ 9.58 (s, 1H), 8.71 – 8.54 (m, 2H), 8.09 (dd, J = 9.0, 0.9 Hz, 1H), 7.89 (s, 1H), 7.34 (dd, J = 9.1, 1.5 Hz, 1H), 7.18 (s, 1H), 6.62 – 6.23 (m, 2H), 4.77 (td, J = 15.3, 3.6 Hz, 2H), 2.53 (s, 3H), 2.32 (d, J = 1.1 Hz, 3H), 2.23 (s, 3H). ESI MS [M+H]+ for C21H21F2N6O, calcd 411.2, found 411.3. Example 129: N-[5-(1-Cyclopropylpyrazol-4-yl)-4-fluoro-2-methylphenyl]pyrazolo[1,5- a]pyridine-3-carboxamide
[0377] The title compound was prepared from 1-cyclopropyl-4-(4,4,5,5-tetramethyl-1,3,2- dioxaborolan-2-yl)pyrazole and triazolo[4,5-b]pyridin-3-yl pyrazolo[1,5-a]pyridine-3- carboxylate.1H NMR (400 MHz, DMSO-d6) δ 9.69 (s, 1H), 8.84 (dt, J = 7.0, 1.1 Hz, 1H), 8.75 (s, 1H), 8.26 – 8.15 (m, 2H), 7.85 (dd, J = 1.9, 0.8 Hz, 1H), 7.64 (d, J = 7.6 Hz, 1H), 7.51 (ddd, J = 8.9, 6.8, 1.1 Hz, 1H), 7.20 (d, J = 11.9 Hz, 1H), 7.11 (td, J = 6.9, 1.4 Hz, 1H), 3.78 (tt, J = 7.5, 3.9 Hz, 1H), 2.23 (s, 3H), 1.12 – 1.06 (m, 2H), 1.00 – 0.94 (m, 2H).19F NMR (376 MHz, DMSO-d6) δ -119.16. ESI MS [M+H]+ for C21H19FN5O, calcd 376.2, found 376.2. Example 130: N-[5-(1-Cyclopropylpyrazol-4-yl)-2,4-dimethylphenyl]pyrazolo[1,5- a]pyridine-3-carboxamide
[0378] The title compound was prepared from 1-cyclopropyl-4-(4,4,5,5-tetramethyl-1,3,2- dioxaborolan-2-yl)-1H-pyrazole, 5-bromo-2,4-dimethylaniline, and triazolo[4,5-b]pyridin-3-yl pyrazolo[1,5-a]pyridine-3-carboxylate in a similar fashion to Ex. 112. 1H NMR (400 MHz, Methanol-d4) δ 8.65 (dt, J = 7.0, 1.1 Hz, 1H), 8.58 (s, 1H), 8.26 (dt, J = 9.0, 1.3 Hz, 1H), 7.86 (s, 1H), 7.65 (s, 1H), 7.48 (ddd, J = 9.0, 6.8, 1.1 Hz, 1H), 7.32 (s, 1H), 7.16 (s, 1H), 7.07 (td, J = 6.9, 1.4 Hz, 1H), 3.68 (tt, J = 7.3, 3.8 Hz, 1H), 2.36 (s, 3H), 2.28 (s, 3H), 1.17 – 1.00 (m, 4H). ESI MS [M+H]+ for C22H22N5O, calcd 372.1, found 372.1. Example 131: N-[5-(1-Cyclopropylpyrazol-4-yl)-4-fluoro-2-methylphenyl]-6- fluoropyrazolo[1,5-a]pyridine-3-carboxamide
[0379] The title compound was prepared from 1-cyclopropyl-4-(4,4,5,5-tetramethyl-1,3,2- dioxaborolan-2-yl)pyrazole, 5-bromo-4-fluoro-2-methylaniline, and triazolo[4,5-b]pyridin-3-yl 6- fluoropyrazolo[1,5-a]pyridine-3-carboxylate in a similar fashion to Ex.112.1H NMR (400 MHz, DMSO-d6) δ 9.70 (s, 1H), 8.74 (dd, J = 6.9, 0.7 Hz, 1H), 8.58 (s, 1H), 8.23 – 8.15 (m, 1H), 7.82 (dd, J = 1.8, 0.8 Hz, 1H), 7.69 (d, J = 7.6 Hz, 1H), 7.37 (ddd, J = 10.9, 7.8, 0.8 Hz, 1H), 7.25 –
7.16 (m, 1H), 7.09 (td, J = 7.4, 4.8 Hz, 1H), 3.79 (tt, J = 7.5, 3.9 Hz, 1H), 2.25 (s, 3H), 1.12 – 1.07 (m, 2H), 1.01 – 0.94 (m, 2H).19F NMR (376 MHz, DMSO-d6) δ -116.86 – -117.34 (m), -119.22 – -119.73 (m). ESI MS [M+H]+ for C21H18F2N5O, calcd 394.1, found 394.2. Example 132: N-[5-[1-(Cyclopropylmethyl)pyrazol-4-yl]-4-fluoro-2- methylphenyl]pyrazolo[1,5-a]pyridine-3-carboxamide
[0380] The title compound was prepared from 1-(cyclopropylmethyl)-4-(4,4,5,5-tetramethyl- 1,3,2-dioxaborolan-2-yl)pyrazole and triazolo[4,5-b]pyridin-3-yl pyrazolo[1,5-a]pyridine-3- carboxylate in a similar fashion to Ex.112.1H NMR (400 MHz, Methanol-d4) δ 8.68 (dt, J = 7.0, 1.1 Hz, 1H), 8.62 (s, 1H), 8.29 (dt, J = 9.0, 1.3 Hz, 1H), 8.10 (dd, J = 2.0, 0.8 Hz, 1H), 7.89 (dd, J = 1.6, 0.8 Hz, 1H), 7.64 (d, J = 7.4 Hz, 1H), 7.51 (ddd, J = 9.0, 6.8, 1.1 Hz, 1H), 7.15 – 7.08 (m, 2H), 4.04 (d, J = 7.1 Hz, 2H), 2.33 (d, J = 0.6 Hz, 3H), 1.40 – 1.28 (m, 1H), 0.67 – 0.60 (m, 2H), 0.43 (dt, J = 6.1, 4.6 Hz, 2H). ESI MS [M+H]+ for C22H21FN5O, calcd 390.2, found 390.2. Example 133: N-[5-[1-(Cyclopropylmethyl)pyrazol-4-yl]-2,4-difluorophenyl]pyrazolo[1,5- a]pyridine-3-carboxamide
[0381] The title compound was prepared from 1-(cyclopropylmethyl)-4-(4,4,5,5-tetramethyl- 1,3,2-dioxaborolan-2-yl)pyrazole, 5-bromo-2,4-difluoroaniline, and triazolo[4,5-b]pyridin-3-yl pyrazolo[1,5-a]pyridine-3-carboxylate in a similar fashion to Ex. 112. 1H NMR (400 MHz, DMSO-d6) δ 9.92 (s, 1H), 8.85 (d, J = 7.0 Hz, 1H), 8.78 (s, 1H), 8.26 – 8.18 (m, 2H), 7.95 – 7.84 (m, 2H), 7.54 (ddd, J = 8.9, 6.8, 1.1 Hz, 1H), 7.44 (t, J = 10.6 Hz, 1H), 7.13 (td, J = 6.9, 1.4 Hz, 1H), 4.02 (d, J = 7.2 Hz, 2H), 1.35 – 1.21 (m, 1H), 0.57 – 0.50 (m, 2H), 0.42 – 0.36 (m, 2H).19F NMR (376 MHz, DMSO-d6) δ -114.73 – -115.27 (m), -120.04 – -120.19 (m). ESI MS [M+H]+ for C21H18F2N5O, calcd 394.1, found 394.1.
Example 134: N-[4-Chloro-5-[1-(cyclopropylmethyl)pyrazol-4-yl]-2- methylphenyl]pyrazolo[1,5-a]pyridine-3-carboxamide
[0382] The title compound was prepared from 1-(cyclopropylmethyl)-4-(4,4,5,5-tetramethyl- 1,3,2-dioxaborolan-2-yl)-1H-pyrazole, 5-bromo-4-chloro-2-methylaniline, and triazolo[4,5- b]pyridin-3-yl pyrazolo[1,5-a]pyridine-3-carboxylate in a similar fashion to Ex. 112. 1H NMR (400 MHz, Methanol-d4) δ 8.66 (dt, J = 7.0, 1.1 Hz, 1H), 8.60 (s, 1H), 8.27 (dt, J = 8.9, 1.2 Hz, 1H), 8.11 (d, J = 0.8 Hz, 1H), 7.84 (d, J = 0.8 Hz, 1H), 7.57 (s, 1H), 7.49 (ddd, J = 9.0, 6.8, 1.1 Hz, 1H), 7.40 (d, J = 0.8 Hz, 1H), 7.09 (td, J = 6.9, 1.4 Hz, 1H), 4.03 (d, J = 7.1 Hz, 2H), 2.31 (s, 3H), 1.39 – 1.24 (m, 2H), 0.66 – 0.55 (m, 2H), 0.48 – 0.37 (m, 2H). ESI MS [M+H]+ for C22H21ClN5O, calcd 406.1, found 406.1. Example 135: N-[2-Chloro-5-[1-(cyclopropylmethyl)pyrazol-4-yl]-4- fluorophenyl]pyrazolo[1,5-a]pyridine-3-carboxamide
[0383] The title compound was prepared from 1-(cyclopropylmethyl)-4-(4,4,5,5-tetramethyl- 1,3,2-dioxaborolan-2-yl)-1H-pyrazole, 5-bromo-2-chloro-4-fluoroaniline, and triazolo[4,5- b]pyridin-3-yl pyrazolo[1,5-a]pyridine-3-carboxylate in a similar fashion to Ex. 112. 1H NMR (400 MHz, Methanol-d4) δ 8.67 (dt, J = 7.0, 1.1 Hz, 1H), 8.62 (s, 1H), 8.28 (dt, J = 8.9, 1.2 Hz, 1H), 8.14 (d, J = 1.8 Hz, 1H), 7.99 (d, J = 7.7 Hz, 1H), 7.91 (t, J = 1.2 Hz, 1H), 7.51 (ddd, J = 8.9, 6.8, 1.1 Hz, 2H), 7.40 (d, J = 10.5 Hz, 1H), 7.10 (td, J = 6.9, 1.4 Hz, 1H), 4.04 (d, J = 7.2 Hz, 2H), 1.36 – 1.24 (m, 1H), 0.66 – 0.57 (m, 2H), 0.41 (dt, J = 6.2, 4.6 Hz, 2H). 19F NMR (376 MHz, DMSO-d6) δ -117.78. ESI MS [M+H]+ for C21H18ClFN5O, calcd 410.1, found 410.1. Example 136: N-[5-[1-(Cyclopropylmethyl)pyrazol-4-yl]-2,4-dimethylphenyl]pyrazolo[1,5- a]pyridine-3-carboxamide
[0384] The title compound was prepared from 1-(cyclopropylmethyl)-4-(4,4,5,5-tetramethyl- 1,3,2-dioxaborolan-2-yl)-1H-pyrazole, 5-bromo-2,4-dimethylaniline, and triazolo[4,5-b]pyridin- 3-yl pyrazolo[1,5-a]pyridine-3-carboxylate in a similar fashion to Ex. 112. 1H NMR (400 MHz, Methanol-d4) δ 8.64 (dt, J = 7.0, 1.1 Hz, 1H), 8.25 (dt, J = 9.0, 1.2 Hz, 1H), 7.89 (s, 1H), 7.69 (s, 1H), 7.47 (ddd, J = 8.9, 6.8, 1.1 Hz, 1H), 7.34 (s, 1H), 7.19 – 7.14 (m, 1H), 7.07 (td, J = 6.9, 1.4 Hz, 1H), 4.03 (d, J = 7.1 Hz, 2H), 2.37 (s, 3H), 2.28 (s, 3H), 1.39 – 1.24 (m, 1H), 0.68 – 0.55 (m, 2H), 0.48 – 0.34 (m, 2H). ESI MS [M+H]+ for C23H24N5O, calcd 386.1, found 386.1. Example 137: N-[5-[1-(Cyclopropylmethyl)pyrazol-4-yl]-4-fluoro-2-methylphenyl]-6- methylpyrazolo[1,5-a]pyridine-3-carboxamide
[0385] The title compound was prepared from 1-(cyclopropylmethyl)-4-(4,4,5,5-tetramethyl- 1,3,2-dioxaborolan-2-yl)pyrazole in a similar fashion to Ex.112.1H NMR (400 MHz, DMSO-d6) δ 9.61 (s, 1H), 8.68 – 8.60 (m, 2H), 8.20 – 8.12 (m, 1H), 8.08 (dd, J = 8.9, 0.9 Hz, 1H), 7.84 (dd, J = 1.7, 0.8 Hz, 1H), 7.61 (d, J = 7.6 Hz, 1H), 7.40 – 7.28 (m, 1H), 7.16 (d, J = 11.8 Hz, 1H), 3.97 (d, J = 7.1 Hz, 2H), 2.32 (s, 3H), 2.20 (s, 3H), 1.23 (dtd, J = 12.8, 7.5, 6.4, 3.7 Hz, 1H), 0.56 – 0.44 (m, 2H), 0.42 – 0.31 (m, 2H). ESI MS [M+H]+ for C23H23FN5O, calcd 404.2, found 404.2. Example 138: N-[4-Fluoro-2-methyl-5-(1-propan-2-ylpyrazol-4-yl)phenyl]pyrazolo[1,5- a]pyridine-3-carboxamide
[0386] The title compound was prepared from 1-propan-2-yl-4-(4,4,5,5-tetramethyl-1,3,2- dioxaborolan-2-yl)pyrazole and triazolo[4,5-b]pyridin-3-yl pyrazolo[1,5-a]pyridine-3-
carboxylate in a similar fashion to Ex.112.1H NMR (400 MHz, Methanol-d4) δ 8.68 (dt, J = 6.9, 1.0 Hz, 1H), 8.62 (s, 1H), 8.29 (dt, J = 8.9, 1.3 Hz, 1H), 8.07 (dd, J = 1.9, 0.8 Hz, 1H), 7.88 (dd, J = 1.8, 0.8 Hz, 1H), 7.64 (d, J = 7.4 Hz, 1H), 7.51 (ddd, J = 9.0, 6.9, 1.1 Hz, 1H), 7.15 – 7.07 (m, 2H), 4.58 (h, J = 6.8 Hz, 1H), 2.32 (s, 3H), 1.53 (d, J = 6.7 Hz, 6H). ESI MS [M+H]+ for C21H21FN5O, calcd 378.2, found 378.1. Example 139: N-[4-Fluoro-2-methyl-5-[1-(trifluoromethyl)pyrazol-4- yl]phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide
[0387] The title compound was prepared from 4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)- 1-(trifluoromethyl)pyrazole and triazolo[4,5-b]pyridin-3-yl pyrazolo[1,5-a]pyridine-3- carboxylate in a similar fashion to Ex.112.1H NMR (400 MHz, Methanol-d4) δ 8.50 (d, J = 7.4 Hz, 2H), 8.29 – 8.21 (m, 2H), 8.09 (s, 1H), 7.61 (d, J = 7.2 Hz, 1H), 7.39 (dd, J = 8.9, 6.9 Hz, 1H), 7.04 (d, J = 11.4 Hz, 1H), 6.98 (t, J = 6.8 Hz, 1H), 2.29 (s, 3H). ESI MS [M+H]+ for C19H14F4N5O, calcd 404.1, found 404.2. Example 140: N-[4-Fluoro-2-methyl-5-[1-(oxan-4-yl)pyrazol-4-yl]phenyl]pyrazolo[1,5- a]pyridine-3-carboxamide
[0388] The title compound was prepared from 1-(oxan-4-yl)-4-(4,4,5,5-tetramethyl-1,3,2- dioxaborolan-2-yl)pyrazole and triazolo[4,5-b]pyridin-3-yl pyrazolo[1,5-a]pyridine-3- carboxylate in a similar fashion to Ex.112.1H NMR (400 MHz, Methanol-d4) δ 8.54 – 8.49 (m, 2H), 8.26 (dt, J = 8.9, 1.3 Hz, 1H), 7.89 (dd, J = 2.0, 0.8 Hz, 1H), 7.82 (dd, J = 1.4, 0.8 Hz, 1H), 7.56 (d, J = 7.3 Hz, 1H), 7.42 – 7.37 (m, 1H), 7.02 – 6.96 (m, 2H), 4.40 – 4.30 (m, 1H), 4.06 (dt, J = 12.5, 3.6 Hz, 2H), 3.59 – 3.48 (m, 2H), 2.27 (d, J = 0.7 Hz, 3H), 2.06 (ddd, J = 10.3, 7.0, 3.8 Hz, 4H). ESI MS [M+H]+ for C23H23FN5O2, calcd 420.2, found 420.2.
Example 141: N-[4-Fluoro-2-methyl-5-[1-(2,2,2-trifluoroethyl)pyrazol-4- yl]phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide
[0389] The title compound was prepared from 4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)- 1-(2,2,2-trifluoroethyl)pyrazole and triazolo[4,5-b]pyridin-3-yl pyrazolo[1,5-a]pyridine-3- carboxylate in a similar fashion to Ex.112.1H NMR (400 MHz, DMSO-d6) δ 9.70 (s, 1H), 8.84 (dt, J = 7.0, 1.1 Hz, 1H), 8.76 (s, 1H), 8.29 (d, J = 2.2 Hz, 1H), 8.22 (dt, J = 8.9, 1.3 Hz, 1H), 8.07 (d, J = 1.1 Hz, 1H), 7.70 (d, J = 7.6 Hz, 1H), 7.51 (ddd, J = 8.9, 6.8, 1.1 Hz, 1H), 7.24 (d, J = 11.8 Hz, 1H), 7.11 (td, J = 6.9, 1.5 Hz, 1H), 5.20 (q, J = 9.1 Hz, 2H), 2.25 (s, 3H).19F NMR (376 MHz, DMSO-d6) δ -70.21 (t, J = 9.4 Hz), -119.10 – -119.33 (m). ESI MS [M+H]+ for C20H16F4N5O, calcd 418.1, found 418.1. Example 142: N-[4-Fluoro-2-methyl-5-[1-(3,3,3-trifluoropropyl)pyrazol-4- yl]phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide
[0390] The title compound was prepared from 4-bromo-1-(3,3,3-trifluoropropyl)pyrazole and N-[4-fluoro-2-methyl-5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)phenyl]pyrazolo[1,5- a]pyridine-3-carboxamide in a similar fashion to Ex.1 and Ex.67.1H NMR (400 MHz, Methanol- d4) δ 8.68 (dt, J = 7.0, 1.1 Hz, 1H), 8.62 (s, 1H), 8.29 (dt, J = 8.9, 1.3 Hz, 1H), 8.10 (dd, J = 2.0, 0.7 Hz, 1H), 7.93 (dd, J = 1.5, 0.8 Hz, 1H), 7.64 (d, J = 7.4 Hz, 1H), 7.51 (ddd, J = 8.9, 6.8, 1.1 Hz, 1H), 7.13 (dd, J = 12.0, 1.0 Hz, 1H), 7.13 – 7.08 (m, 1H), 4.48 (t, J = 7.1 Hz, 2H), 2.84 (qt, J = 10.7, 7.0 Hz, 2H), 2.33 (s, 3H). ESI MS [M+H]+ for C21H18F4N5O, calcd 432.1, found 432.2. Example 143: N-[5-[1-(2,2-Difluorocyclopropyl)pyrazol-4-yl]-4-fluoro-2- methylphenyl]pyrazolo[1,5-a]pyridine-3-carboxamide
[0391] Step a: A 40 mL vial was charged with 1-ethenyl-4-iodopyrazole (500 mg, 2.27 mmol, 1.0 equiv.), NaI (2.1 g, 13.9 mmol, 6.1 equiv.), and MeCN (15 mL, 0.15 M) under an N2 atmosphere. Me3SiCF3 (2.2 mL, 14.9 mmol, 6.6 equiv.) was added over 45 min to the reaction mixture at 80 °C, and the reaction mixture was stirred at 80 °C for an additional 3 h. Upon complete conversion, as judged by LCMS analysis, the reaction mixture was cooled to rt. The insoluble salts were filtered and washed with CH2Cl2, and the filtrate was concentrated to dryness to afford 1- (2,2-difluorocyclopropyl)-4-iodopyrazole, which was used directly in the next step without further purification. [0392] Step b: The crude intermediate obtained in step a was converted to the title compound in a similar fashion to Ex.1.1H NMR (400 MHz, Methanol-d4) δ 8.68 (dt, J = 7.0, 1.1 Hz, 1H), 8.62 (s, 1H), 8.28 (dt, J = 9.0, 1.3 Hz, 1H), 8.15 (d, J = 1.8 Hz, 1H), 7.96 (d, J = 0.8 Hz, 1H), 7.65 (d, J = 7.4 Hz, 1H), 7.51 (ddd, J = 8.9, 6.8, 1.0 Hz, 1H), 7.16 – 7.08 (m, 2H), 4.37 (dddd, J = 10.6, 8.1, 5.9, 1.9 Hz, 1H), 2.33 (s, 3H), 2.40 – 2.18 (m, 2H). ESI MS [M+H]+ for C21H17F3N5O, calcd 412.1, found 412.1. Example 144: N-[5-[1-(2,2-Difluorocyclopropyl)pyrazol-4-yl]-4-fluoro-2-methylphenyl]-6- fluoropyrazolo[1,5-a]pyridine-3-carboxamide
[0393] The title compound was prepared from 6-fluoro-N-[4-fluoro-2-methyl-5-(4,4,5,5- tetramethyl-1,3,2-dioxaborolan-2-yl)phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide in a similar fashion to Ex. 143. 1H NMR (400 MHz, Methanol-d4) δ 8.59 (d, J = 7.0 Hz, 1H), 8.50 (s, 1H), 8.15 (s, 1H), 7.96 (s, 1H), 7.76 (dd, J = 13.5, 7.3 Hz, 1H), 7.34 – 7.24 (m, 1H), 7.17 – 7.01 (m, 2H), 4.38 (q, J = 8.9, 8.3 Hz, 1H), 2.35 (s, 3H), 2.40 – 2.18 (m, 2H). ESI MS [M+H]+ for C21H16F4N5O, calcd 430.1, found 430.1. Example 145: N-[4-Fluoro-5-[1-(3-hydroxycyclobutyl)pyrazol-4-yl]-2- methylphenyl]pyrazolo[1,5-a]pyridine-3-carboxamide
[0394] Step a: A round-bottom flask was charged with 4-bromo-1H-pyrazole (2.00 g, 13.6 mmol, 1.0 equiv.), 3-bromocyclobutan-1-one (2.60 g, 17.7 mmol, 1.3 equiv.), K2CO3 (2.60 g, 17.7 mmol, 1.30 equiv.), and DMF (39 mL, 0.35 M), and the resulting mixture was stirred at 25 °C for 18 h. The reaction mixture was diluted with water and EtOAc, and extracted with EtOAc (3x). The combined organic layers were washed with brine, dried over Na2SO4, and concentrated. The crude product, 3-(4-bromopyrazol-1-yl)cyclobutan-1-one was used in directly the next step without further purification. [0395] Step b: A 40 mL vial was charged with the crude product obtained in step a (2.7 g) and MeOH (14 mL). NaBH4 (0.573 g, 15.1 mmol) was added to the mixture in small portions at 0 °C, and the resulting mixture was allowed to warm to 25 °C and stir for 4 h. The reaction was quenched with water and the mixture was extracted with EtOAc (3x). The combined organic layers were
washed with brine, dried over Na2SO4, and concentrated to afford 3-(4-bromopyrazol-1- yl)cyclobutan-1-ol, which was used directly in the next step without further purification. [0396] Step c: The crude intermediate obtained in step b was converted to the title compound in a similar fashion to Ex.1.1H NMR (400 MHz, Methanol-d4) δ 8.68 (dt, J = 6.9, 1.1 Hz, 1H), 8.62 (s, 1H), 8.29 (dt, J = 9.0, 1.2 Hz, 1H), 8.14 – 8.12 (m, 1H), 8.05 – 8.00 (m, 1H), 7.96 – 7.93 (m, 1H), 7.65 (d, J = 7.4 Hz, 1H), 7.54 – 7.44 (m, 1H), 7.13 – 7.07 (m, 1H), 4.45 (ddd, J = 16.4, 8.9, 7.4 Hz, 1H), 4.13 (tt, J = 7.9, 6.7 Hz, 1H), 2.90 (dddd, J = 11.9, 9.2, 6.1, 2.8 Hz, 2H), 2.46 (tdd, J = 9.2, 7.9, 2.9 Hz, 2H), 2.33 (s, 3H). ESI MS [M+H]+ for C22H21FN5O2, calcd 406.2, found 406.1. Example 146: N-[4-Fluoro-5-[1-(4-fluorophenyl)pyrazol-4-yl]-2- methylphenyl]pyrazolo[1,5-a]pyridine-3-carboxamide
[0397] The title compound was prepared from [1-(4-fluorophenyl)-1H-pyrazol-4-yl] boronic acid and triazolo[4,5-b]pyridin-3-yl pyrazolo[1,5-a]pyridine-3-carboxylate in a similar fashion to Ex.112.1H NMR (400 MHz, DMSO-d6) δ 9.72 (s, 1H), 8.90 (t, J = 1.0 Hz, 1H), 8.81 (dt, J = 7.0, 1.1 Hz, 1H), 8.74 (s, 1H), 8.20 (dt, J = 8.9, 1.3 Hz, 1H), 8.15 (dd, J = 2.0, 0.6 Hz, 1H), 7.98 – 7.88 (m, 2H), 7.74 (d, J = 7.5 Hz, 1H), 7.48 (ddd, J = 8.9, 6.8, 1.1 Hz, 1H), 7.40 – 7.28 (m, 2H), 7.28 – 7.20 (m, 1H), 7.08 (td, J = 6.9, 1.5 Hz, 1H), 2.23 (s, 3H). 19F NMR (376 MHz, DMSO-d6) δ - 116.40, -118.51. ESI MS [M+H]+ for C24H18F2N5O, calcd 430.1, found 430.1. Example 147: N-[4-Fluoro-5-[1-(4-chlorophenyl)pyrazol-4-yl]-2- methylphenyl]pyrazolo[1,5-a]pyridine-3-carboxamide
[0398] The title compound was prepared from [1-(4-fluorophenyl)-1H-pyrazol-4-yl] boronic acid, 5-bromo-4-chloro-2-methylaniline, and triazolo[4,5-b]pyridin-3-yl pyrazolo[1,5-a]pyridine- 3-carboxylate in a similar fashion to Ex.112.1H NMR (400 MHz, DMSO-d6) δ 9.74 (s, 1H), 8.89
(d, J = 0.7 Hz, 1H), 8.84 (dt, J = 6.9, 1.1 Hz, 1H), 8.77 (s, 1H), 8.22 (dt, J = 8.9, 1.3 Hz, 1H), 8.16 (d, J = 0.6 Hz, 1H), 8.01 – 7.91 (m, 2H), 7.72 (s, 1H), 7.57 – 7.47 (m, 2H), 7.43 – 7.32 (m, 2H), 7.12 (td, J = 6.9, 1.5 Hz, 1H), 2.29 (s, 3H).19F NMR (376 MHz, DMSO-d6) δ -116.24. ESI MS [M+H]+ for C24H18ClFN5O, calcd 446.1, found 446.1. Example 148: 6-Fluoro-N-[4-fluoro-5-[1-(4-fluorophenyl)pyrazol-4-yl]-2- methylphenyl]pyrazolo[1,5-a]pyridine-3-carboxamide
[0399] The title compound was prepared from [1-(4-fluorophenyl)-1H-pyrazol-4-yl] boronic acid and triazolo[4,5-b]pyridin-3-yl 6-fluoropyrazolo[1,5-a]pyridine-3-carboxylate in a similar fashion to Ex.112.1H NMR (400 MHz, DMSO-d6) δ 9.81 (s, 1H), 9.19 (ddd, J = 4.7, 2.3, 0.7 Hz, 1H), 8.93 (d, J = 1.2 Hz, 1H), 8.79 (s, 1H), 8.30 – 8.22 (m, 1H), 8.19 (d, J = 2.0 Hz, 1H), 8.01 – 7.92 (m, 2H), 7.77 (d, J = 7.5 Hz, 1H), 7.64 (ddd, J = 9.7, 8.2, 2.3 Hz, 1H), 7.43 – 7.33 (m, 2H), 7.28 (d, J = 11.8 Hz, 1H), 2.27 (s, 3H). 19F NMR (376 MHz, DMSO-d6) δ -116.39, -118.29, - 138.54. ESI MS [M+H]+ for C24H17F3N5O, calcd 448.1, found 448.2. Example 149: N-[4-Chloro-5-[1-(4-fluorophenyl)pyrazol-4-yl]-2-methylphenyl]-6- methoxypyrazolo[1,5-a]pyridine-3-carboxamide
[0400] The title compound was prepared from [1-(4-fluorophenyl)-1H-pyrazol-4-yl]boronic acid, 5-bromo-4-chloro-2-methylaniline, and triazolo[4,5-b]pyridin-3-yl 6-methoxypyrazolo[1,5- a]pyridine-3-carboxylate in a similar fashion to Ex.112.1H NMR (400 MHz, DMSO-d6) δ 9.67 (s, 1H), 8.85 (d, J = 0.7 Hz, 1H), 8.63 (s, 1H), 8.51 (dd, J = 2.3, 0.7 Hz, 1H), 8.12 (d, J = 0.7 Hz, 1H), 8.07 (dd, J = 9.6, 0.7 Hz, 1H), 7.97 – 7.87 (m, 2H), 7.67 (s, 1H), 7.46 (d, J = 0.9 Hz, 1H), 7.39 – 7.31 (m, 2H), 7.29 (dd, J = 9.6, 2.2 Hz, 1H), 3.83 (s, 3H), 2.25 (s, 3H). 19F NMR (376 MHz, DMSO-d6) δ -116.36. ESI MS [M+H]+ for C25H20ClFN5O2, calcd 476.1, found 476.1.
Example 150: N-[4-Fluoro-2-methyl-5-(1-methylpyrazol-3-yl)phenyl]pyrazolo[1,5- a]pyridine-3-carboxamide
[0401] The title compound was prepared from 1-methyl-3-(4,4,5,5-tetramethyl-1,3,2- dioxaborolan-2-yl)pyrazole and triazolo[4,5-b]pyridin-3-yl pyrazolo[1,5-a]pyridine-3- carboxylate in a similar fashion to Ex.112.1H NMR (400 MHz, Methanol-d4) δ 8.67 (dt, J = 7.0, 1.3 Hz, 1H), 8.61 (s, 1H), 8.28 (dt, J = 8.9, 1.3 Hz, 1H), 7.85 (d, J = 7.3 Hz, 1H), 7.63 (d, J = 2.2 Hz, 1H), 7.50 (dddd, J = 8.9, 6.8, 1.9, 1.1 Hz, 1H), 7.13 (d, J = 12.0 Hz, 1H), 7.09 (dt, J = 6.9, 1.5 Hz, 1H), 6.65 (ddd, J = 3.6, 2.4, 1.2 Hz, 1H), 3.94 (d, J = 0.9 Hz, 3H), 2.34 (s, 3H). ESI MS [M+H]+ for C19H17FN5O, calcd 350.1, found 350.1. Example 151: N-[5-(5-Cyclopropyl-1H-pyrazol-3-yl)-4-fluoro-2-methylphenyl]pyrazolo[1,5- a]pyridine-3-carboxamide
[0402] The title compound was prepared from 5-cyclopropyl-3-iodo-1H-pyrazole and N-[4- fluoro-2-methyl-5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)phenyl]pyrazolo[1,5-a]pyridine- 3-carboxamide in a similar fashion to Ex.1 and Ex.67.1H NMR (400 MHz, Methanol-d4) δ 8.67 (dt, J = 7.0, 1.1 Hz, 1H), 8.61 (s, 1H), 8.28 (dt, J = 8.9, 1.3 Hz, 1H), 7.76 (d, J = 7.1 Hz, 1H), 7.50 (ddd, J = 9.0, 6.8, 1.1 Hz, 1H), 7.14 (d, J = 11.7 Hz, 1H), 7.10 (td, J = 6.9, 1.5 Hz, 1H), 6.33 (d, J = 3.2 Hz, 1H), 2.34 (s, 3H), 1.96 (tt, J = 8.5, 5.1 Hz, 1H), 0.99 (d, J = 7.9 Hz, 2H), 0.81 – 0.71 (m, 2H). ESI MS [M+H]+ for C21H19FN5O, calcd 376.2, found 376.1. Example 152: N-[5-(5-Cyclopropyl-1H-pyrazol-3-yl)-4-fluoro-2-methylphenyl]-6- methylpyrazolo[1,5-a]pyridine-3-carboxamide
[0403] The title compound was prepared in a similar fashion to Ex. 1 from 5-cyclopropyl-3- iodo-1H-pyrazole and N-[4-fluoro-2-methyl-5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2- yl)phenyl]-6-methylpyrazolo[1,5-a]pyridine-3-carboxamide, which was accessed from triazolo[4,5-b]pyridin-3-yl 6-methylpyrazolo[1,5-a]pyridine-3-carboxylate and 5-bromo-4- fluoro-2-methyl aniline in a similar fashion to step b, Ex.1.1H NMR (400 MHz, Methanol-d4) δ 8.54 (s, 1H), 8.51 – 8.46 (m, 1H), 8.21 – 8.11 (m, 1H), 7.75 (d, J = 7.3 Hz, 1H), 7.39 (dd, J = 9.1, 1.5 Hz, 1H), 7.14 (d, J = 11.7 Hz, 1H), 6.33 (d, J = 3.3 Hz, 1H), 2.41 (s, 3H), 2.33 (s, 3H), 1.96 (tt, J = 8.5, 5.0 Hz, 1H), 1.02 – 0.95 (m, 2H), 0.79 – 0.74 (m, 2H). ESI MS [M+H]+ for C22H21FN5O, calcd 390.2, found 390.1. Example 153: N-[5-(5-Cyclopropyl-1H-pyrazol-3-yl)-4-fluoro-2-methylphenyl]-6- methoxypyrazolo[1,5-a]pyridine-3-carboxamide
[0404] The title compound was prepared from 5-cyclopropyl-3-iodo-1H-pyrazole and N-[4- fluoro-2-methyl-5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)phenyl]-6-methoxypyrazolo[1,5- a]pyridine-3-carboxamide in a similar fashion to Ex.1 and Ex.86.1H NMR (400 MHz, Methanol- d4) δ 8.50 (s, 1H), 8.32 (dd, J = 2.2, 0.7 Hz, 1H), 8.15 (dd, J = 9.7, 0.7 Hz, 1H), 7.75 (d, J = 7.3 Hz, 1H), 7.28 (dd, J = 9.7, 2.2 Hz, 1H), 7.13 (d, J = 11.6 Hz, 1H), 6.32 (d, J = 3.2 Hz, 1H), 3.91 (s, 3H), 2.33 (s, 3H), 2.00 – 1.91 (m, 1H), 1.04 – 0.93 (m, 2H), 0.79 – 0.72 (m, 2H). ESI MS [M+H]+ for C22H21FN5O2, calcd 406.2, found 406.1. Example 154: N-[5-[2-(Cyclopropylmethyl)tetrazol-5-yl]-4-fluoro-2- methylphenyl]pyrazolo[1,5-a]pyridine-3-carboxamide
[0405] Step a: A mixture of triazolo[4,5-b]pyridin-3-yl pyrazolo[1,5-a]pyridine-3-carboxylate (500 mg, 1.78 mmol, 1.0 equiv.), and 5-amino-2-fluoro-4-methylbenzonitrile (268 mg, 1.78 mmol, 1.0 equiv.) in NMP (6.0 mL) was stirred at 120 °C for 6 h. The reaction mixture was cooled to rt and diluted with water. The resulting precipitated solid was collected by vacuum filtration, rinsed with water, and dried under vacuum to afford N-(5-cyano-4-fluoro-2-methylphenyl)pyrazolo[1,5- a]pyridine-3-carboxamide, which was used directly in the next step without further purification. [0406] Step b: A vial was charged with product from step a (495 mg, 1.68 mmol, 1.0 equiv.), sodium azide (219 mg, 3.36 mmol, 2.0 equiv.), ammonium chloride (180 mg, 3.36 mmol, 2.0 equiv.) and DMF (8 mL). The reaction mixture was stirred at 110 °C for 16 h, at which time LCMS analysis indicated full consumption of starting material. The reaction mixture was cooled to rt and concentrated under reduced pressure. The crude product was purified by column chromatography (0% to 20% EtOAc in MeOH) to provide the desired N-[4-fluoro-2-methyl-5-(2H-tetrazol-5- yl)phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide. [0407] Step c: To a solution of product from step b (75 mg, 0.22 mmol, 1.0 equiv.) and K2CO3 (60 mg, 0.44 mmol, 2.0 equiv.) in DMF (1 mL) was added bromomethylcyclopropane (59 mg, 0.44 mmol, 2.0 equiv.). The reaction mixture was stirred at 40 °C for 2 h, then diluted with EtOAc and water. The organic layer was separated, filtered over Celite®, and concentrated under reduced pressure. Purification by reverse phase HPLC afforded the title compound.1H NMR (400 MHz, Methanol-d4) δ 8.51 (dt, J = 7.0, 1.2 Hz, 2H), 8.25 (dt, J = 8.9, 1.3 Hz, 1H), 8.04 (dd, J = 6.9, 2.4 Hz, 1H), 7.40 (ddd, J = 9.0, 6.8, 1.1 Hz, 1H), 7.14 (dd, J = 11.0, 1.0 Hz, 1H), 6.99 (td, J = 6.9, 1.4
Hz, 1H), 4.49 (d, J = 7.4 Hz, 2H), 2.35 (d, J = 0.8 Hz, 3H), 1.49- 1.40 (tt, 1H), 0.68 – 0.62 (m, 2H), 0.48 (dt, J = 6.4, 4.8 Hz, 2H). ESI MS [M+H]+ for C20H19FN7O, calcd 392.2, found 392.1. Example 155: N-[5-[2-(Cyclopropylmethyl)tetrazol-5-yl]-4-fluoro-2-methylphenyl]-6- methoxypyrazolo[1,5-a]pyridine-3-carboxamide
[0408] The title compound was prepared from triazolo[4,5-b]pyridin-3-yl 6- methoxypyrazolo[1,5-a]pyridine-3-carboxylate in a similar fashion to Ex. 154. 1H NMR (400 MHz, Methanol-d4) δ 8.43 (s, 1H), 8.15 – 8.10 (m, 2H), 8.04 (d, J = 6.8 Hz, 1H), 7.20 – 7.13 (m, 2H), 4.50 (d, J = 7.4 Hz, 2H), 3.85 (s, 3H), 2.35 (s, 3H), 1.48 – 1.41 (m, 1H), 0.68 – 0.63 (m, 2H), 0.49 (q, J = 5.5 Hz, 2H). ESI MS [M+H]+ for C21H21FN7O2, calcd 422.2, found 422.1. Example 156: 6-Fluoro-N-[4-fluoro-2-methyl-5-(2-methyltetrazol-5-yl)phenyl]pyrazolo[1,5- a]pyridine-3-carboxamide
[0409] The title compound was prepared from triazolo[4,5-b]pyridin-3-yl 6-fluoropyrazolo[1,5- a]pyridine-3-carboxylate and iodomethane in a similar fashion to Ex. 154. 1H NMR (400 MHz, Methanol-d4) δ 8.53 (ddd, J = 3.9, 2.2, 0.8 Hz, 2H), 8.27 (ddt, J = 9.7, 5.6, 0.9 Hz, 1H), 8.03 (d, J = 6.9 Hz, 1H), 7.35 (ddd, J = 10.0, 7.9, 2.2 Hz, 1H), 7.15 (dd, J = 11.0, 0.9 Hz, 1H), 4.39 (s, 3H), 2.34 (d, J = 0.7 Hz, 3H). ESI MS [M+H]+ for C17H14F2N7O, calcd 370.1, found 370.0. Example 157: 6-Fluoro-N-[4-fluoro-2-methyl-5-(1-methyltetrazol-5-yl)phenyl]pyrazolo[1,5- a]pyridine-3-carboxamide
[0410] The title compound was obtained as a minor byproduct in the synthesis of Ex.157, which was prepared from triazolo[4,5-b]pyridin-3-yl 6-fluoropyrazolo[1,5-a]pyridine-3-carboxylate and iodomethane in a similar fashion to Ex.154.1H NMR (400 MHz, Methanol-d4) δ 8.55 – 8.51 (m, 2H), 8.26 (ddd, J = 9.8, 5.7, 0.8 Hz, 1H), 7.62 (d, J = 6.7 Hz, 1H), 7.35 (ddd, J = 9.9, 7.9, 2.2 Hz, 1H), 7.26 – 7.21 (m, 1H), 4.08 (d, J = 2.1 Hz, 3H), 2.39 (d, J = 0.7 Hz, 3H). ESI MS [M+H]+ for C17H14F2N7O, calcd 370.1, found 370.0. Example 158: N-[5-(2-Ethyltetrazol-5-yl)-4-fluoro-2-methylphenyl]pyrazolo[1,5-a]pyridine- 3-carboxamide
[0411] The title compound was prepared from bromoethane in a similar fashion to Ex.154.1H NMR (400 MHz, Methanol-d4) δ 8.52 (dq, J = 7.0, 1.1 Hz, 2H), 8.25 (dq, J = 8.9, 1.2 Hz, 1H), 8.04 (dd, J = 6.8, 0.9 Hz, 1H), 7.40 (ddt, J = 9.0, 6.8, 1.1 Hz, 1H), 7.14 (d, J = 11.1 Hz, 1H), 7.00 (tt, J = 6.9, 1.2 Hz, 1H), 4.70 (qd, J = 7.4, 1.0 Hz, 2H), 2.35 (s, 3H), 1.64 (td, J = 7.4, 1.0 Hz, 3H). ESI MS [M+H]+ for C18H17FN7O, calcd 366.1, found 366.1. Example 159: N-[5-(2-Ethyltetrazol-5-yl)-4-fluoro-2-methylphenyl]-6-fluoropyrazolo[1,5- a]pyridine-3-carboxamide
[0412] The title compound was prepared from triazolo[4,5-b]pyridin-3-yl 6-fluoropyrazolo[1,5- a]pyridine-3-carboxylate and bromoethane in a similar fashion to Ex. 154. 1H NMR (400 MHz, Methanol-d4) δ 8.52 (dq, J = 7.0, 1.1 Hz, 2H), 8.25 (dq, J = 8.9, 1.2 Hz, 1H), 8.04 (dd, J = 6.8, 0.9 Hz, 1H), 7.40 (ddt, J = 9.0, 6.8, 1.1 Hz, 1H), 7.14 (d, J = 11.1 Hz, 1H), 7.00 (tt, J = 6.9, 1.2 Hz, 1H), 4.70 (qd, J = 7.4, 1.0 Hz, 2H), 2.35 (s, 3H), 1.64 (td, J = 7.4, 1.0 Hz, 3H). ESI MS [M+H]+ for C18H16F2N7O, calcd 384.1, found 384.0.
Example 160: N-[4-Fluoro-5-[2-(2-methoxyethyl)tetrazol-5-yl]-2- methylphenyl]pyrazolo[1,5-a]pyridine-3-carboxamide
[0413] The title compound was prepared from 1-bromo-2-methoxyethane in a similar fashion to Ex.154.1H NMR (400 MHz, Methanol-d4) δ 8.52 (dq, J = 7.0, 0.9 Hz, 2H), 8.25 (dt, J = 8.9, 1.2 Hz, 1H), 8.05 (d, J = 6.8 Hz, 1H), 7.40 (ddt, J = 8.9, 6.9, 0.9 Hz, 1H), 7.15 (d, J = 11.0 Hz, 1H), 7.00 (td, J = 6.9, 1.4 Hz, 1H), 4.82 (dd, J = 5.7, 4.8 Hz, 2H), 3.97 – 3.92 (m, 2H), 3.31 (d, J = 0.7 Hz, 3H), 2.35 (s, 3H). ESI MS [M+H]+ for C19H19FN7O2, calcd 396.2, found 396.0. Example 161: N-[5-(6-cyclopropylpyridazin-3-yl)-4-fluoro-2-methylphenyl]pyrazolo[1,5- a]pyridine-3-carboxamide
[0414] The title compound was prepared from N-[4-fluoro-2-methyl-5-(4,4,5,5-tetramethyl- 1,3,2-dioxaborolan-2-yl)phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide and 3-bromo-6- cyclopropylpyridazine in a similar fashion to Ex. 89 (step a).1H NMR (400 MHz, DMSO-d6) δ 9.73 (s, 1H), 8.81 (dt, J = 6.9, 1.1 Hz, 1H), 8.73 (s, 1H), 8.18 (dt, J = 8.9, 1.3 Hz, 1H), 7.95 (d, J = 7.6 Hz, 1H), 7.86 (dd, J = 8.9, 2.1 Hz, 1H), 7.57 (d, J = 8.9 Hz, 1H), 7.48 (ddd, J = 8.9, 6.8, 1.1 Hz, 1H), 7.31 (d, J = 11.8 Hz, 1H), 7.08 (td, J = 6.9, 1.5 Hz, 1H), 2.34 – 2.23 (m, 4H), 1.17 – 0.99 (m, 4H). ESI MS [M+H]+ for C22H19FN5O, calcd 388.2, found 388.1. Example 162: N-[5-(2-cyclopropylpyrimidin-5-yl)-4-fluoro-2-methylphenyl]pyrazolo[1,5- a]pyridine-3-carboxamide
[0415] The title compound was prepared from N-[4-fluoro-2-methyl-5-(4,4,5,5-tetramethyl- 1,3,2-dioxaborolan-2-yl)phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide and 5-bromo-2- cyclopropylpyrimidine in a similar fashion to Ex. 89 (step a).1H NMR (400 MHz, DMSO-d6) δ 9.72 (s, 1H), 8.83 – 8.77 (m, 3H), 8.73 (s, 1H), 8.18 (dt, J = 8.9, 1.3 Hz, 1H), 7.58 (d, J = 7.7 Hz, 1H), 7.48 (ddd, J = 8.9, 6.8, 1.1 Hz, 1H), 7.31 (d, J = 11.4 Hz, 1H), 7.08 (td, J = 6.9, 1.5 Hz, 1H), 2.28 (s, 3H), 2.23 (tt, J = 8.0, 4.9 Hz, 1H), 1.09 – 0.98 (m, 4H). ESI MS [M+H]+ for C22H19FN5O, calcd 388.2, found 388.1. Example 163: N-[5-(5-cyclopropylpyrazin-2-yl)-4-fluoro-2-methylphenyl]pyrazolo[1,5- a]pyridine-3-carboxamide
[0416] The title compound was prepared from N-[4-fluoro-2-methyl-5-(4,4,5,5-tetramethyl- 1,3,2-dioxaborolan-2-yl)phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide and 2-bromo-5- cyclopropylpyrazine in a similar fashion to Ex.89 (step a).1H NMR (400 MHz, DMSO-d6) δ 9.72 (s, 1H), 8.86 – 8.77 (m, 2H), 8.75 – 8.69 (m, 2H), 8.19 (dt, J = 8.9, 1.3 Hz, 1H), 7.94 (d, J = 7.6 Hz, 1H), 7.48 (ddd, J = 8.9, 6.8, 1.1 Hz, 1H), 7.30 (d, J = 12.0 Hz, 1H), 7.08 (td, J = 6.9, 1.4 Hz, 1H), 2.30 (s, 3H), 2.28 – 2.20 (m, 1H), 1.09 – 1.02 (m, 2H), 0.99 (dt, J = 4.6, 2.8 Hz, 2H). ESI MS [M+H]+ for C22H19FN5O, calcd 388.2, found 388.1. Example 164: N-[5-(2-cyclopropyloxypyrimidin-5-yl)-4-fluoro-2-methylphenyl]-6- fluoropyrazolo[1,5-a]pyridine-3-carboxamide
[0417] The title compound was prepared from 6-fluoro-N-[4-fluoro-2-methyl-5-(4,4,5,5- tetramethyl-1,3,2-dioxaborolan-2-yl)phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide and 5- bromo-2-cyclopropyloxypyrimidine in a similar fashion to Ex.92 (step c). 1H NMR (400 MHz, DMSO-d6) δ 9.79 (s, 1H), 9.21 – 9.08 (m, 1H), 8.79 (d, J = 1.4 Hz, 2H), 8.75 (s, 1H), 8.21 (ddd, J = 9.7, 5.9, 0.8 Hz, 1H), 7.68 – 7.52 (m, 2H), 7.31 (d, J = 11.4 Hz, 1H), 4.32 (tt, J = 6.5, 3.2 Hz, 1H), 2.27 (s, 3H), 0.83 – 0.69 (m, 4H). ESI MS [M+H]+ for C22H18F2N5O2, calcd 422.1, found 422.1. Example 165: 6-fluoro-N-[4-fluoro-2-methyl-5-[2-[[(3S)-oxolan-3-yl]amino]pyrimidin-5- yl]phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide
[0418] Step a: A round-bottom flask was charged with 5-bromo-2-chloropyrimidine (1.93 g, 10.0 mmol, 1.0 equiv.), (3S)-oxolan-3-amine hydrochloride (2.47 g, 20.0 mmol, 2.0 equiv.), K2CO3 (5.53 g, 40.0 mmol, 40.0 equiv.), and DMF (20 mL, 0.5 M), and the resulting mixture was stirred at 70 °C for 12 h. The reaction mixture was diluted with water and EtOAc, and extracted with EtOAc (3x). The combined organic layers were washed with brine, dried over Na2SO4, and concentrated. The crude product was purified by column chromatography (0% to 100% EtOAc in hexanes) to afford the desired 5-bromo-N-[(3S)-oxolan-3-yl]pyrimidin-2-amine. [0419] Step b: A vial was charged successively with the crude product obtained in step a (55 mg, 0.225 mmol, 1.5 equiv.), 6-fluoro-N-[4-fluoro-2-methyl-5-(4,4,5,5-tetramethyl-1,3,2- dioxaborolan-2-yl)phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide (62 mg, 0.15 mmol, 1.0 equiv., prepared from 6-fluoropyrazolo[1,5-a]pyridine-3-carboxylic acid and 5-bromo-4-fluoro-2- methylaniline according to Ex.1, steps a-b), Pd(dppf)Cl2 (11 mg, 0.015 mmol, 10 mol%), 2M aq. Na2CO3 (0.15 mL, 0.3 mmol, 2.0 equiv.) and dioxane (0.6 mL, 0.2 M). The reaction mixture was evacuated and back-filled with N2 (3x) and then heated at 100 ºC for 12 h. The mixture was then cooled to rt, diluted with EtOAc, filtered over Celite® and concentrated in vacuo. The crude
product was purified by reverse phase HPLC to provide the title compound.1H NMR (400 MHz, DMSO-d6) δ 9.74 (s, 1H), 9.16 (ddd, J = 4.6, 2.3, 0.7 Hz, 1H), 8.74 (s, 1H), 8.47 (d, J = 1.4 Hz, 2H), 8.21 (ddd, J = 9.8, 5.9, 0.8 Hz, 1H), 7.66 (d, J = 6.4 Hz, 1H), 7.60 (ddd, J = 9.8, 8.3, 2.3 Hz, 1H), 7.47 (d, J = 7.8 Hz, 1H), 7.24 (d, J = 11.4 Hz, 1H), 4.44 – 4.31 (m, 1H), 3.89 – 3.77 (m, 2H), 3.69 (td, J = 8.1, 5.6 Hz, 1H), 3.54 (dd, J = 8.8, 4.2 Hz, 1H), 2.24 (s, 3H), 2.12 (dq, J = 12.4, 7.6 Hz, 1H), 1.87 (ddt, J = 12.5, 7.4, 5.2 Hz, 1H). ESI MS [M+H]+ for C23H21F2N6O2, calcd 451.2, found 451.1. Example 166: N-[5-[2-(2,2-difluoroethylamino)pyrimidin-5-yl]-4-fluoro-2-methylphenyl]-6- fluoropyrazolo[1,5-a]pyridine-3-carboxamide
[0420] The title compound was prepared from 6-fluoro-N-[4-fluoro-2-methyl-5-(4,4,5,5- tetramethyl-1,3,2-dioxaborolan-2-yl)phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide and 5- bromo-N-(2,2-difluoroethyl)pyrimidin-2-amine (prepared from 5-bromo-2-chloropyrimidine and 2,2-difluoroethanamine hydrochloride according to Ex.165, step a) in a similar fashion to Ex.165. 1H NMR (400 MHz, DMSO-d6) δ 9.75 (s, 1H), 9.16 (dd, J = 4.8, 2.2 Hz, 1H), 8.74 (s, 1H), 8.50 (d, J = 1.3 Hz, 2H), 8.21 (dd, J = 9.8, 5.9 Hz, 1H), 7.75 (t, J = 6.2 Hz, 1H), 7.60 (ddd, J = 10.2, 8.2, 2.3 Hz, 1H), 7.48 (d, J = 7.8 Hz, 1H), 7.25 (d, J = 11.4 Hz, 1H), 6.10 (tt, J = 56.2, 4.1 Hz, 1H), 3.70 (tdd, J = 15.1, 6.2, 4.2 Hz, 2H), 2.25 (s, 3H). ESI MS [M+H]+ for C21H17F4N6O, calcd 445.1, found 445.1. Example 167: 6-chloro-N-[5-[1-(2,2-difluoroethyl)triazol-4-yl]-4-fluoro-2- methylphenyl]pyrazolo[1,5-a]pyridine-3-carboxamide
[0421] The title compound was prepared from 6-fluoro-N-[4-fluoro-2-methyl-5-(4,4,5,5- tetramethyl-1,3,2-dioxaborolan-2-yl)phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide and 5- bromo-N-(cyclopropylmethyl)pyrimidin-2-amine (prepared from 5-bromo-2-chloropyrimidine and cyclopropylmethanamine according to Ex. 165, step a) in a similar fashion to Ex. 165. 1H NMR (400 MHz, DMSO-d6) δ 9.74 (s, 1H), 9.16 (dd, J = 4.7, 2.2 Hz, 1H), 8.74 (s, 1H), 8.43 (d, J = 1.5 Hz, 2H), 8.26 – 8.15 (m, 1H), 7.60 (ddd, J = 10.2, 8.2, 2.3 Hz, 1H), 7.54 – 7.42 (m, 2H), 7.23 (d, J = 11.5 Hz, 1H), 3.16 (t, J = 6.3 Hz, 2H), 2.24 (s, 3H), 1.12 – 0.96 (m, 1H), 0.46 – 0.32 (m, 2H), 0.23 – 0.16 (m, 2H). ESI MS [M+H]+ for C23H21F2N6O, calcd 435.2, found 435.2. Example 168: 6-fluoro-N-[4-fluoro-2-methyl-5-[6-[[(3S)-oxolan-3-yl]amino]pyridazin-3- yl]phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide
[0422] The title compound was prepared from 6-fluoro-N-[4-fluoro-2-methyl-5-(4,4,5,5- tetramethyl-1,3,2-dioxaborolan-2-yl)phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide and 6- bromo-N-[(3S)-oxolan-3-yl]pyridazin-3-amine (prepared from 3-bromo-6-chloropyridazine and (3S)-oxolan-3-amine hydrochloride according to Ex.165, step a) in a similar fashion to Ex. 165. 1H NMR (400 MHz, DMSO-d6) δ 9.74 (s, 1H), 9.16 (ddd, J = 4.6, 2.3, 0.8 Hz, 1H), 8.74 (s, 1H), 8.28 – 8.16 (m, 1H), 7.85 (d, J = 7.7 Hz, 1H), 7.60 (ddt, J = 8.3, 5.2, 2.5 Hz, 2H), 7.32 – 7.19 (m, 2H), 6.88 (d, J = 9.4 Hz, 1H), 4.55 – 4.41 (m, 1H), 3.89 (dd, J = 9.0, 5.8 Hz, 1H), 3.83 (q, J = 7.5 Hz, 1H), 3.71 (td, J = 8.2, 5.6 Hz, 1H), 3.57 (dd, J = 8.9, 3.7 Hz, 1H), 2.27 (s, 3H), 2.20 (dq, J = 12.6, 7.5 Hz, 1H), 1.90 – 1.76 (m, 1H). ESI MS [M+H]+ for C23H21F2N6O2, calcd 451.2, found 451.2. Example 169: N-[5-[6-(azetidin-1-yl)pyridazin-3-yl]-4-fluoro-2-methylphenyl]-6- fluoropyrazolo[1,5-a]pyridine-3-carboxamide
[0423] The title compound was prepared from 6-fluoro-N-[4-fluoro-2-methyl-5-(4,4,5,5- tetramethyl-1,3,2-dioxaborolan-2-yl)phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide and 3- (azetidin-1-yl)-6-bromopyridazine (prepared from 3-bromo-6-chloropyridazine and azetidine hydrochloride according to Ex.165, step a) in a similar fashion to Ex.165.1H NMR (400 MHz, DMSO-d6) δ 9.78 (s, 1H), 9.22 – 9.13 (m, 1H), 8.74 (s, 1H), 8.25 – 8.16 (m, 1H), 7.83 (d, J = 7.7 Hz, 1H), 7.68 (dd, J = 9.3, 2.2 Hz, 1H), 7.60 (ddd, J = 10.1, 8.2, 2.3 Hz, 1H), 7.25 (d, J = 11.9 Hz, 1H), 6.82 (d, J = 9.4 Hz, 1H), 4.07 (t, J = 7.5 Hz, 4H), 2.38 (dq, J = 9.5, 7.4 Hz, 2H), 2.27 (s, 3H). ESI MS [M+H]+ for C22H19F2N6O, calcd 421.2, found 421.1. Example 170: N-[5-(6-cyclopropyloxypyridazin-3-yl)-4-fluoro-2-methylphenyl]-6- fluoropyrazolo[1,5-a]pyridine-3-carboxamide
[0424] The title compound was prepared from 6-fluoro-N-[4-fluoro-2-methyl-5-(4,4,5,5- tetramethyl-1,3,2-dioxaborolan-2-yl)phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide and 3- bromo-6-cyclopropyloxypyridazine (prepared from 3-bromo-6-fluoropyridazine and cyclopropanol according to Ex.165, step a) in a similar fashion to Ex.165.1H NMR (400 MHz, DMSO-d6) δ 9.79 (s, 1H), 9.16 (dd, J = 4.7, 2.2 Hz, 1H), 8.75 (d, J = 1.7 Hz, 1H), 8.22 (dd, J = 9.8, 5.8 Hz, 1H), 7.95 (dd, J = 9.2, 2.1 Hz, 1H), 7.90 (d, J = 7.7 Hz, 1H), 7.61 (ddd, J = 10.1, 8.2, 2.2 Hz, 1H), 7.38 – 7.26 (m, 2H), 4.42 (tt, J = 6.3, 3.0 Hz, 1H), 2.30 (s, 3H), 0.82 (tt, J = 7.3, 2.6 Hz, 2H), 0.79 – 0.70 (m, 2H). ESI MS [M+H]+ for C22H18F2N5O2, calcd 422.1, found 422.1. Example 171: N-[5-(6-cyclopropyl-4-fluoropyridin-3-yl)-4-fluoro-2-methylphenyl]-6- fluoropyrazolo[1,5-a]pyridine-3-carboxamide
[0425] Step a: A 40 mL vial was charged with 5-bromo-2-chloro-4-fluoropyridine (250 mg, 0.95 mmol., 1.0 equiv.), cyclopropyl zinc bromide (443 mg, 1.9 mmol., 2 equiv.), Pd(PPh3)4 (59 mg, 0.05 mmol., 5 mol%) and THF (6.0 mL). The resulting mixture was stirred at 50 °C for 5 h. Upon complete conversion, as judged by LCMS analysis, the reaction mixture was cooled to rt and filtered over Celite® and concentrated in vacuo. The crude product was purified by column chromatography (SiO2, 0 to 100% EtOAc in hexanes) to afford 5-bromo-2-cyclopropyl-4- fluoropyridine. [0426] Step b: A vial was charged successively with the product obtained in step a, 5-bromo-2- cyclopropyl-4-fluoropyridine (78 mg, 0.363 mmol., 1.0 equiv.), 6-fluoro-N-[4-fluoro-2-methyl-5- (4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide (100 mg, 0.242 mmol, 1.0 equiv., prepared from 6-fluoropyrazolo[1,5-a]pyridine-3-carboxylic acid and 5-bromo-4-fluoro-2-methylaniline according to Ex. 1, steps a-b), Pd(PPh3)4 (42.0 mg, 0.036 mmol., 10 mol%), K2CO3 (100 mg, 0.73 mmol., 2.0 equiv.) and dioxane : water (4:1, 4 mL). The reaction mixture was stirred at 100 °C for approximately 2 h, at which time LCMS analysis indicated complete consumption of starting material. The reaction mixture was cooled to rt and diluted with EtOAc. The organic layer was separated, filtered over Celite®, and concentrated in vacuo. The crude product was purified by HPLC to provide the title compound. 1H NMR (400 MHz, Chloroform-d1) δ 8.47 (ddd, J = 3.8, 2.2, 0.8 Hz, 1H), 8.39 – 8.31 (m, 1H), 8.30 – 8.17 (m, 2H), 7.56 (s, 1H), 7.48 (dd, J = 11.4, 1.3 Hz, 1H), 7.32 (ddd, J = 9.9, 7.8, 2.2 Hz, 1H), 7.04 (d, J = 11.7 Hz, 1H), 2.34 (s, 3H), 2.01 (tt, J = 8.6, 5.3 Hz, 1H), 1.17 – 0.95 (m, 2H), 0.95 – 0.65 (m, 2H). ESI MS [M+H]+ for C23H18F3N4O1, calcd 423.1, found 423.0. Example 172: N-[2,4-dimethyl-5-(6-propan-2-yloxypyridazin-3-yl)phenyl]-6- fluoropyrazolo[1,5-a]pyridine-3-carboxamide
[0427] The title compound was prepared from 6-fluoro-N-[4-fluoro-2-methyl-5-(4,4,5,5- tetramethyl-1,3,2-dioxaborolan-2-yl)phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide and 3- bromo-6-propan-2-yloxypyridazine in a similar fashion to Ex. 165. 1H NMR (400 MHz, Chloroform-d1) δ 8.81 (d, J = 40.6 Hz, 1H), 8.44 (ddd, J = 3.9, 2.2, 0.8 Hz, 1H), 8.29 (ddd, J = 9.8, 5.8, 0.8 Hz, 1H), 8.00 (d, J = 7.5 Hz, 1H), 7.74 (dd, J = 9.2, 2.4 Hz, 1H), 7.38 – 7.10 (m, 1H), 6.96 (d, J = 9.2 Hz, 1H), 6.86 (d, J = 11.4 Hz, 1H), 5.63 (p, J = 6.1 Hz, 1H), 2.09 (s, 3H), 1.45 (d, J = 6.1 Hz, 6H). ESI MS [M+H]+ for C22H20F2N5O2, calcd 424.1, found 424.1. Example 173: N-[5-(5-difluoromethoxy-2-pyridyl)-4-fluoro-2-tolyl]-1,7a-diaza-3- indenecarboxamide
[0428] The title compound was prepared from 6-fluoro-N-[4-fluoro-2-methyl-5-(4,4,5,5- tetramethyl-1,3,2-dioxaborolan-2-yl)phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide and 2- bromo-5-(difluoromethoxy)pyridine according to Ex.165 (step a), in a similar fashion to Ex.165. 1H NMR (400 MHz, DMSO-d6) δ 9.74 (s, 1H), 8.84 (dt, J = 7.0, 1.1 Hz, 1H), 8.76 (s, 1H), 8.61 (d, J = 2.8 Hz, 1H), 8.22 (dt, J = 8.9, 1.2 Hz, 1H), 7.97 (d, J = 7.8 Hz, 1H), 7.90 (dd, J = 8.8, 1.6 Hz, 1H), 7.79 (dd, J = 8.7, 2.9 Hz, 1H), 7.51 (ddd, J = 8.9, 6.8, 1.1 Hz, 1H), 7.39 (s, 1H), 7.31 (d, J = 12.2 Hz, 1H), 7.11 (td, J = 6.9, 1.5 Hz, 1H), 2.33 (s, 3H). ESI MS [M+H]+ for C21H16F3N4O2, calcd 413.1, found 413.1. Example 174: N-[5-[(5-cyclopropyloxypyridine-2-carbonyl)amino]-4-fluoro-2- methylphenyl]-6-fluoropyrazolo[1,5-a]pyridine-3-carboxamide
step c [0429] Step a: A solution of cyclopropanol (280 mg, 4.8 mmol, 1.5 equiv.) in DMF (5.0 mL) was added NatBuO (470 mg, 4.8 mmol, 1.5 equiv.). Let it stir at room temperature for 30min.5- fluoropyridine-2-carbonitrile (394 mg, 3.2 mmol, 1.0 equiv.) was added. The reaction mixture was stirred at 75 °C for 30min, cooled to room temperature and then diluted with EtOAc and water. The organic layer was separated, dried over Na2SO4 and concentrated in vacuo. The crude product was purified by column chromatography (10% to 70% EtOAc in hexanes) to provide the desired 5-fluoropyridine-2-carbonitrile. [0430] Step b: A mixture of product from step a with KOH (10.0 equiv.) in tBuOH (10mL) and water (2mL) was stirred at 105 °C for 48h, then cooled to room temperature. The reaction mixture was acidified with 1N HCl to PH~3 and then extracted with EtOAc. The organic layer was separated, dried over Na2SO4 and concentrated in vacuo. The crude product was used directly in the next step without further purification. [0431] Step c: The title compound was prepared from 5-cyclopropyloxypyridine-2-carboxylic acid and 6-N-(5-amino-4-fluoro-2-methylphenyl)-6-fluoropyrazolo[1,5-a]pyridine-3- carboxamide in a similar fashion to Ex. 176. 1H NMR (400 MHz, Methanol-d4) δ 8.78 (dd, J = 4.4, 2.2 Hz, 1H), 8.61 (s, 1H), 8.41 (d, J = 2.8 Hz, 1H), 8.35 – 8.25 (m, 2H), 8.15 (d, J = 8.7 Hz, 1H), 7.69 (dd, J = 8.7, 2.8 Hz, 1H), 7.50 (ddd, J = 10.1, 8.1, 2.2 Hz, 1H), 7.17 (d, J = 11.6 Hz, 1H), 3.98 (m, 1H), 2.29 (s, 3H), 0.93 – 0.85 (m, 2H), 0.78 (m, 2H). ESI MS [M+H]+ for C24H20F2N5O3, calcd 464.1, found 464.2.
Example 175: 6-fluoro-N-[4-fluoro-2-methyl-5-[[5-[[(3S)-oxolan-3-yl]amino]pyridine-2- carbonyl]amino]phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide
[0432] The title compound was prepared from 5-[[(3S)-oxolan-3-yl]amino]pyridine-2- carboxylic acid and 6-N-(5-amino-4-fluoro-2-methylphenyl)-6-fluoropyrazolo[1,5-a]pyridine-3- carboxamide in a similar fashion to Ex.176. 1H NMR (400 MHz, DMSO-d6) δ 9.95 (s, 1H), 9.76 (s, 1H), 9.18 – 9.12 (m, 1H), 8.74 (s, 1H), 8.24 – 8.19 (m, 1H), 8.03 (d, J = 2.7 Hz, 1H), 7.86 (d, J = 8.6 Hz, 1H), 7.64 – 7.56 (m, 1H), 7.23 (d, J = 11.7 Hz, 1H), 7.05 (dd, J = 8.7, 2.7 Hz, 1H), 6.98 (d, J = 6.4 Hz, 1H), 4.09 (m, 1H), 3.92 – 3.76 (m, 3H), 3.72 (td, J = 8.3, 5.4 Hz, 2H), 3.61 – 3.51 (m, 2H), 2.19 (s, 3H). ESI MS [M+H]+ for C25H23F2N6O3, calcd 493.1, found 493.2. Example 176: 6-fluoro-N-[4-fluoro-5-[(5-fluoropyridine-2-carbonyl)amino]-2- methylphenyl]pyrazolo[1,5-a]pyridine-3-carboxamide
[0433] Step a: A mixture of triazolo[4,5-b]pyridin-3-yl 6-fluoropyrazolo[1,5-a]pyridine-3- carboxylate (298 mg, 1.0 mmol, 1.0 equiv.) and 5-bromo-4-fluoro-2-methylaniline (204 mg, 1.0 mmol, 1.0 equiv.) in NMP (6.0 mL) was stirred at 120 °C for 4 h. The reaction mixture was cooled to rt and diluted with water. The resulting precipitated solid was collected by vacuum filtration, rinsed with water, and dried under vacuum to afford N-(5-bromo-4-fluoro-2-methylphenyl)-6- fluoropyrazolo[1,5-a]pyridine-3-carboxamide, which was used directly in the next step without further purification. [0434] Step b: A vial was charged with product from step a (260 mg, 0.72 mmol, 1.0 equiv.), diphenylmethanimine (155 mg, 0.85 mmol, 1.2 equiv.), Pd2(dba)3 (66 mg, 0.072 mmol, 0.1 equiv.), XantPhos (84 mg, 0.44 mmol, 0.2 equiv.), Cs2CO3 (705 mg, 2.16 mmol, 3.0 equiv.) and dioxane (8 mL). The reaction mixture was stirred at 100 °C for 16 h under N2, at which time LCMS analysis indicated full consumption of starting material. The reaction mixture was cooled to rt and concentrated under reduced pressure. The crude product was purified by column chromatography (10% to 70% EtOAc in MeOH) to provide the desired N-[5-(benzhydrylideneamino)-4-fluoro-2- methylphenyl]-6-fluoropyrazolo[1,5-a]pyridine-3-carboxamide. [0435] Step c: To a mixture of product from step b (220 mg, 0.47 mmol, 1.0 equiv.), NaOAc (97 mg, 1.18 mmol, 2.5 equiv.) in MeOH (5 mL) was added NH2OH HCl (66 mg, 0.94 mmol, 2.0 equiv.). The reaction mixture was stirred at 45 °C for 3 h, then diluted with EtOAc and NaHCO3 aq. The organic layer was separated, dried over Na2SO4 and concentrated in vacuo. The crude product was purified by column chromatography (10% to 70% EtOAc in hexanes) to provide the desired N-(5-amino-4-fluoro-2-methylphenyl)-6-fluoropyrazolo[1,5-a]pyridine-3-carboxamide. [0436] Step d: The desired product was prepared from 5-fluoropyridine-2-carboxylic acid in a similar manner to Example 1, step a.1H NMR (400 MHz, DMSO-d6) δ 10.25 (s, 1H), 9.77 (s, 1H), 9.16 (ddd, J = 4.6, 2.2, 0.7 Hz, 1H), 8.74 (d, J = 2.7 Hz, 1H), 8.26 – 8.17 (m, 2H), 8.04 – 7.92 (m, 2H), 7.61 (ddd, J = 10.2, 8.2, 2.2 Hz, 1H), 7.26 (d, J = 11.5 Hz, 1H), 6.51 (s, 1H), 2.22 (s, 3H). ESI MS [M+H]+ for C21H15F3N5O2, calcd 426.1, found 426.2. Example 177: 6-fluoro-N-[4-fluoro-2-methyl-5-[(5-methylpyridine-2- carbonyl)amino]phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide
[0437] The title compound was prepared from 5-methylpyridine-2-carboxylic acid and 6-N-(5- amino-4-fluoro-2-methylphenyl)-6-fluoropyrazolo[1,5-a]pyridine-3-carboxamide in a similar fashion to Ex.176. 1H NMR (400 MHz, DMSO-d6) δ 10.28 (s, 1H), 9.77 (s, 1H), 9.16 (dd, J = 4.7, 2.2 Hz, 1H), 8.74 (s, 1H), 8.56 (s, 1H), 8.21 (dd, J = 9.8, 5.9 Hz, 1H), 8.10 (d, J = 7.7 Hz, 1H), 8.03 (d, J = 7.9 Hz, 1H), 7.91 – 7.83 (m, 1H), 7.61 (ddd, J = 10.1, 8.3, 2.2 Hz, 1H), 7.26 (d, J = 11.6 Hz, 1H), 2.39 (s, 3H), 2.21 (s, 3H). ESI MS [M+H]+ for C22H18F2N5O2, calcd 422.1, found 422.2. Example 178: 6-fluoro-N-[4-fluoro-2-methyl-5-(pyridine-3- carbonylamino)phenyl]pyrazolo[1,5-a]pyridine-3-carboxamide
[0438] The title compound was prepared from pyridine-2-carboxylic acid and 6-N-(5-amino-4- fluoro-2-methylphenyl)-6-fluoropyrazolo[1,5-a]pyridine-3-carboxamide in a similar fashion to Ex.176. 1H NMR (400 MHz, Methanol-d4) δ 9.13 – 9.07 (m, 1H), 8.81 – 8.71 (m, 2H), 8.60 (s, 1H), 8.41 (dt, J = 8.1, 1.9 Hz, 1H), 8.28 (ddd, J = 9.8, 5.6, 0.7 Hz, 1H), 7.81 (d, J = 7.3 Hz, 1H), 7.64 (ddd, J = 8.0, 5.0, 0.9 Hz, 1H), 7.50 (ddt, J = 9.9, 8.0, 2.0 Hz, 1H), 7.18 (d, J = 11.0 Hz, 1H), 2.32 (s, 3H). ESI MS [M+H]+ for C21H16F2N5O2, calcd 408.1, found 408.2. Example 179: N-[5-[(6-cyclopropyloxypyridazine-3-carbonyl)amino]-4-fluoro-2- methylphenyl]-6-fluoropyrazolo[1,5-a]pyridine-3-carboxamide
[0439] The title compound was prepared from 6-cyclopropyloxypyridazine-3-carboxylic acid and 6-N-(5-amino-4-fluoro-2-methylphenyl)-6-fluoropyrazolo[1,5-a]pyridine-3-carboxamide in a similar fashion to Ex.176. 1H NMR (400 MHz, DMSO-d6) δ 10.50 (s, 1H), 9.76 (s, 1H), 9.16 (dd, J = 4.6, 2.2 Hz, 1H), 8.74 (s, 1H), 8.25 – 8.18 (m, 2H), 7.88 (d, J = 7.5 Hz, 1H), 7.61 (ddd, J = 10.1, 8.2, 2.3 Hz, 1H), 7.45 (d, J = 9.2 Hz, 1H), 7.26 (d, J = 11.3 Hz, 1H), 4.47 (m, 1H), 2.23 (s, 3H), 0.92 – 0.74 (m, 4H). ESI MS [M+H]+ for C23H19F2N6O3, calcd 465.1, found 465.2. Example 180: N-[5-[(2,2-difluorocyclopropanecarbonyl)amino]-4-fluoro-2-methylphenyl]- 6-fluoropyrazolo[1,5-a]pyridine-3-carboxamide
[0440] The title compound was prepared from 2,2-difluorocyclopropane-1-carboxylic acid and 6-N-(5-amino-4-fluoro-2-methylphenyl)-6-fluoropyrazolo[1,5-a]pyridine-3-carboxamide in a similar fashion to Ex. 176. 1H NMR (400 MHz, DMSO-d6) δ 10.20 (s, 1H), 9.70 (s, 1H), 9.15 (ddd, J = 4.6, 2.3, 0.8 Hz, 1H), 8.71 (s, 1H), 8.19 (ddd, J = 9.8, 5.9, 0.8 Hz, 1H), 7.86 (d, J = 7.7 Hz, 1H), 7.60 (ddd, J = 9.8, 8.2, 2.3 Hz, 1H), 7.19 (d, J = 11.6 Hz, 1H), 3.03 – 2.90 (m, 1H), 2.18 (s, 3H), 2.01 – 1.88 (m, 2H). ESI MS [M+H]+ for C19H15F4N4O2, calcd 407.1, found 407.2. Example 181: N-[5-(cyclopropanecarbonylamino)-4-fluoro-2-methylphenyl]-6- fluoropyrazolo[1,5-a]pyridine-3-carboxamide
[0441] The title compound was prepared from cyclopropanecarboxylic acid and 6-N-(5-amino- 4-fluoro-2-methylphenyl)-6-fluoropyrazolo[1,5-a]pyridine-3-carboxamide in a similar fashion to Ex.176. 1H NMR (400 MHz, DMSO-d6) δ 9.92 (s, 1H), 9.67 (s, 1H), 9.15 (ddd, J = 4.7, 2.3, 0.8 Hz, 1H), 8.71 (s, 1H), 8.19 (ddd, J = 9.8, 5.9, 0.8 Hz, 1H), 7.84 (d, J = 7.7 Hz, 1H), 7.59 (ddd, J = 9.8, 8.2, 2.3 Hz, 1H), 7.15 (d, J = 11.6 Hz, 1H), 2.16 (s, 3H), 1.99 – 1.89 (m, 1H), 0.75 (d, J = 6.1 Hz, 4H). ESI MS [M+H]+ for C19H17F2N4O2, calcd 371.1, found 371.2.
Example 182: N-[5-[(2-cyclopropylacetyl)amino]-4-fluoro-2-methylphenyl]-6- fluoropyrazolo[1,5-a]pyridine-3-carboxamide
[0442] The title compound was prepared from 2-cyclopropyl acetic acid and 6-N-(5-amino-4- fluoro-2-methylphenyl)-6-fluoropyrazolo[1,5-a]pyridine-3-carboxamide in a similar fashion to Ex.176. 1H NMR (400 MHz, DMSO-d6) δ 9.69 (s, 1H), 9.53 (s, 1H), 9.15 (ddd, J = 4.6, 2.3, 0.8 Hz, 1H), 8.72 (s, 1H), 8.20 (ddd, J = 9.8, 6.0, 0.8 Hz, 1H), 7.83 (d, J = 7.7 Hz, 1H), 7.60 (ddd, J = 9.8, 8.3, 2.3 Hz, 1H), 7.15 (d, J = 11.6 Hz, 1H), 2.23 (d, J = 7.0 Hz, 2H), 2.17 (s, 3H), 1.08 – 0.93 (m, 1H), 0.48 – 0.37 (m, 2H), 0.23 – 0.12 (m, 2H). ESI MS [M+H]+ for C20H19F2N4O2, calcd 385.1, found 385.2. Example 183: N-[5-(cyclopropanecarbonylamino)-4-fluoro-2-methylphenyl]pyrazolo[1,5- a]pyridine-3-carboxamide
[0443] The title compound was prepared from cyclopropanecarboxylic acid and triazolo[4,5- b]pyridin-3-yl pyrazolo[1,5-a]pyridine-3-carboxylate in a similar fashion to Ex. 176. 1H NMR (400 MHz, DMSO-d6) δ 9.92 (s, 1H), 9.61 (s, 1H), 8.79 (dt, J = 7.0, 1.1 Hz, 1H), 8.69 (s, 1H), 8.16 (dt, J = 8.9, 1.3 Hz, 1H), 7.84 (d, J = 7.7 Hz, 1H), 7.47 (ddd, J = 8.9, 6.8, 1.1 Hz, 1H), 7.15 (d, J = 11.8 Hz, 1H), 7.07 (td, J = 6.9, 1.4 Hz, 1H), 2.17 (s, 3H), 1.94 (q, J = 6.2 Hz, 1H), 0.75 (d, J = 6.2 Hz, 4H). ESI MS [M+H]+ for C19H18FN4O2, calcd 353.1, found 353.2. Biological Example Inhibition of c-Kit, PDGFRα, PDGFRβ, CSF1R and FLT3 kinase activity [0444] Compounds were evaluated to determine the potency with which they inhibit the kinase activity of the following panel of tyrosine kinases: c-Kit (SignalChem, Product #: K06-11BG), PDGFRα (SignalChem, Product #: P12-18G), PDGFRβ (SignalChem, Product #: P13-11G),
CSF1R (SignalChem, Product #: C74-11G) and FLT3 (Invitrogen, Product #: PR4666C). Activity was determined as a function of phosphorylated biotinylated TK peptide generated by the transfer of phosphate from ATP as measured by the use of the HTRF KinEASE-TK assay kit (Cisbio, Product #: 62TK0PEJ). Levels of phosphorylated biotinylated TK peptide (Part 61TK0BLC of KinEASE assay kit) are quantified by its capture by phosphor-TK-Antibody-Cryptate (Part of KinEASE assay kit) and XL665-labeled Streptavidin (Part 610SAXLG of KinEASE assay kit) followed by measurement of Time-Resolved Fluorescence Resonance Energy Transfer (TR- FRET) signal. On the day of the assay, compounds were solubilized in DMSO and dispensed into a 384-well white Opti-plate (PerkinElmer, catalog # 6007290) to generate a 22 point 1:2 titration. Enzyme solution was prepared for each of the tyrosine kinases in 50 mM HEPES, pH 7.4, 5 mM MgCl2 for c-Kit or 10 mM MgCl2 for FLT3, CSF1R, PDGFRα and PDGFRβ, 2 mM MnCl2, 0.01% Brij-35 and 0.01% BSA. Working enzyme concentrations were prepared for c-Kit at 2 nM (2x), PDGFRα and PDGFRβ at 10 nM (2x), CSF1R at 2 nM (2x) and FLT3 at 0.4 nM (2x). Five microliters of enzyme dilution of each tyrosine kinase were added to their respective 384-well white Opti-plate pre-dispensed with compound and allowed to incubate for 1 h at rt. Utilizing the same enzyme buffer recipe, 2x substrate mixes were prepared for each enzyme as follows: For c- Kit, 1.6 μM (2x) TK substrate and 16 µM (2x) ATP (Promega, catalog # V915). For PDGFRα, 3.2 μM TK substrate and 1 μM ATP. For PDGFRβ, 3.2 μM TK substrate and 40 μM ATP. For CSF1R, 3.2 μM TK substrate and 20 μM ATP. For FLT3, 3.2 μM TK substrate and 100 μM ATP. Reactions were initiated by addition of 5 µL of the respective 2x substrate mix to each well of the plates containing the respective tyrosine kinase enzymes and were allowed to proceed for 120 minutes at rt, except for FLT3, the reaction time is 45 min. Following the reaction, 10 μL of detection mix consisting of 0.2 μM (2x) of Streptavidin-XL665 and 2 μM (2x) of TK-Antibody-Cryptate prepared in detection buffer (Part 62SDBRDF of KinEASE assay kit) was added and then allowed to incubate for 60 min. The TR-FRET signal was quantified by measuring the ratio of emission at 665 nm to 620 nm after excitation at 320 nm by reading on a PerkinElmer Envision multimode reader. Compound potencies (IC50 values) were determined using a standard 4-parameter non- linear regression fit.
Phospho-KIT (Y703) Cellular Assay in M07e Cells [0445] The day prior to assay, M07e cells (DSMZ, catalog # ACC 104) were serum starved. Cells were centrifuged and cell pellet resuspended in OptiMEM (Gibco, catalog # 31985062) to a density of 1x106 - 2.5x106 cells per mL. Cells were then incubated overnight at 5% CO2 and 37 °C in an appropriately sized flask. On the day of the experiment, an 11 point, half log titration of test compound was pre-dispensed into 96-well round bottom polypropylene plates (Corning, catalog # 3365). The serum starved M07e cells were centrifuged and resuspended to a cell density of 1x106 cells per mL with DPBS (Gibco, catalog # 14190-144). Live/Dead Green Fixable viability dye (Invitrogen, catalog # L34970) was added as 1 µL/ 1 mL of cell suspension. Cells were then incubated in the dark at 4 °C for 30 min. Cells were washed by centrifugation and resuspension with DPBS + 0.5% BSA (Gemini Bio Products, catalog # 700110/100, 30% BSA solution). Cells were resuspended, separated into two equal volumes and again centrifuged. Cell pellets were resuspended with either OptiMEM or human serum (Innovative Research, catalog # ISERAB1000ML) to a cell density of 1x106 cells per mL. Cells were then added to the appropriate compound plate at 100,000 cells per well in 100 μL and mixed. Plates were incubated at 5% CO2 and 37 °C for 50 min. Stem Cell Factor, SCF (R&D Systems, #255-SC/CF) was diluted in OptiMEM to 1 µg/mL and added to all wells as 10 µL for a final concentration of 90.9 ng/mL. Plates were incubated at 5% CO2 and 37 °C for 10 min. Incubation was stopped by addition of 110 µL 4% paraformaldehyde solution in PBS (Life Technologies, catalog # J19943.K2). Plates were incubated at rt for at least 15 min. Plates were then centrifuged and supernatant removed by flicking. Cells were washed once by resuspension in 200 µL DPBS + 0.5% BSA, centrifuged, and supernatant removed by flicking. Cells were resuspended in 100 µL 1X Permeabilization buffer (Invitrogen, catalog # 88882400) and plates incubated at rt for 30 min. Plates were centrifuged, supernatant removed by flicking, and cells resuspended in 50 µL primary antibody, anti-phospho- c-KIT (Tyr703) (Cell Signaling, catalog # 3073), diluted to 1 µg/mL with permeabilization buffer. Plates were incubated at rt for 1 h. Plates were centrifuged, supernatant removed, and cells resuspended with 200 µL DPBS + 0.5% BSA. Plates were again centrifuged, supernatant removed by flicking, and cells resuspended in 100 µL secondary antibody, goat anti-rabbit AlexaFluor647 (AF647) (Invitrogen, catalog # A21244) diluted to 1 µg/mL with DPBS + 0.5% BSA. Plates were incubated at rt for 30 min. followed by centrifugation, supernatant removal, and cell resuspension with 200 µL DPBS + 0.5% BSA. Cells were washed again by centrifugation, supernatant removal,
and cell resuspension with 200 µL DPBS + 0.5% BSA. Samples were then read by flow cytometry. The Forward:Side scatter plot was gated and that population was gated for live cells based on the Live/Dead Green staining. The geometric mean of AF647 of the live cell population was recorded. Percentage maximum activity in each test well was calculated based on DMSO (100% activity) and positive control treated cell wells (0% activity). The potencies (IC50 values) of test compounds were determined using a standard 4-parameter non-linear regression fit. Table 3: Biochemical and cellular potency of specific examples vs. KIT, PDGFRα, PDGFRβ, CSF1R, and FLT3 (IC50: + means > 1 μM, ++ means 100 nM to 1 μM, +++ means < 100 nM). ND=not determined.
185
[0446] Although the foregoing disclosure has been described in some detail by way of illustration and Example for purposes of clarity of understanding, one of skill in the art will appreciate that certain changes and modifications may be practiced within the scope of the appended claims.
Claims
CLAIMS What is claimed is: 1. A compound having a structure according to Formula III:
(Formula III) or a pharmaceutically acceptable salt thereof, wherein: A is: (i) selected from the group consisting of: ,
, , , , ,
r (ii) a 6-membered heteroaryl having 1-3 ring nitrogen atoms, wherein the 6- membered heteroaryl is substituted with 0-2 R1d; or (iii) -NHC(O)-R1g; each R1 is independently selected from the group consisting of halo, -CN, -OH, C1- C6-alkyl, C1-C6-haloalkyl, C1-C6-hydroxyalkyl, C1-C6-cyanoalkyl, C1-C6 alkoxy, C3-C6- cycloalkyl, -O-C3-C6-cycloalkyl, 4- to 8-membered heterocycloalkyl, -NR1aR1b, phenyl, 5- to 6-membered heteroaryl, -X1-NR1aR1b, -X1-C(O)NR1aR1b, -X1-O-(C1-C3-alkyl), -X1-C3- C6-cycloalkyl, -X1-phenyl, and -X2-(4- to 8-membered heterocycloalkyl), wherein each 5- to 6-membered heteroaryl has 1-2 ring heteroatoms independently selected from N and O; each 4- to 8-membered heterocycloalkyl has 1-3 ring heteroatoms independently selected
from N, O, and S, and said 4- to 8-membered heterocycloalkyl is optionally substituted with one oxo; and each R1 is optionally substituted with 1-3 R1c; or two adjacent R1 groups are combined with the two ring atoms to which they are attached to form a 5- or 6-membered heterocycloalkyl group having 1-2 ring heteroatoms independently selected from N, and O; each X1 is C1-C3-alkylene; each X2 is C1-C3-alkylene, or –(C1-C3-alkylene)-C(O)-; R1a and R1b, when present, are independently H, C1-C6 alkyl, or C3-C6 cycloalkyl; each R1c, when present, is independently halo, -OH, -C1-C6-alkyl, -C1-C6-alkoxy, or -C(O)-(C1-C6-alkyl); each R1d, when present, is independently halo, C1-C6 alkoxy, C1-C3 haloalkoxy, C3- C6-cycloalkyl, -O-C3-C6-cycloalkyl, or -NR1eR1f; R1e and R1f, when present, are independently H, C1-C6 haloalkyl, -(C1-C3-alkylene)- (C3-C6-cycloalkyl), or 4- to 8-membered heterocycloalkyl having 1-3 ring heteroatoms independently selected from N, O, and S; or R1e and R1f together with the nitrogen atom to which they are attached form a 4- to 6-membered heterocycloalkyl group having 1-2 ring heteroatoms independently selected from N, and O; R1g, when present, is C3-C6-cycloalkyl, -(C1-C3-alkylene)-(C3-C6-cycloalkyl), or 6- membered heteroaryl having 1-3 ring nitrogen atoms; wherein R1g is optionally substituted with 0-2 R1h; each R1h, when present, is independently halo, C1-C6 alkyl, -O-C3-C6-cycloalkyl, or -NH-(4- to 8-membered heterocycloalkyl), wherein the 4- to 8-membered heterocycloalkyl has 1-2 ring heteroatoms independently selected from N, O, and S; n is 0 or 1; m is 0, 1, or 2; R2, when present, is halo, -CN, -C1-C3-alkyl, -C1-C3-alkoxy, or C3-C6-cycloalkyl; R3 is -H, halo, -C1-C6-alkyl, -C1-C6-haloalkyl, -C1-C6-hydroxyalkyl, -C1-C6-alkoxy, or -C(O)NR3aR3b; R3a and R3b, when present, are independently H, or C1-C3 alkyl; each R4, when present, is independently halo, C1-C6 alkyl, C1-C6-alkoxy, C3-C6 cycloalkyl, 4- to 8-membered heterocycloalkyl having 1-3 ring heteroatoms independently
selected from N, O, and S, or phenyl, wherein said phenyl is optionally substituted with 1- 3 halo; R5 is H, C1-C3-alkyl, or -NR5aR5b; R5a and R5b, when present, are independently H or C1-C3 alkyl; and R6 is H, or C1-C3-alkyl. 2. The compound according to claim 1, or a pharmaceutically acceptable salt thereof, wherein R6 is H. 3. The compound according to claim 1 or claim 2, or a pharmaceutically acceptable salt thereof, wherein R5 is H. 4. The compound according to claim any one of claims 1-3, or a pharmaceutically acceptable salt thereof, wherein: m is 0, 1, or 2; and each R4, when present, is independently -F, -Cl, -CH3, -CH2CH3, -OCH3, -OCH2CH3, cyclopropyl,
. 5. The compound according to any one of claims 1-4, or a pharmaceutically acceptable salt thereof, wherein R3 is halo, C1-C3 alkyl, or C1-C3 hydroxyalkyl. 6. The compound according to any one of claims 1-5, or a pharmaceutically acceptable salt thereof, wherein R3 is -F, -Cl, -CH3, or -CH2-OH. 7. The compound according to any one of claims 1-6, or a pharmaceutically acceptable salt thereof, wherein n is 0 or 1, and R2, when present, is -F, -Cl, -CN, -CH3, -OCH3, or cyclopropyl. 8. The compound according to claim 7, wherein n is 1. 9. The compound according to any one of claims 1-8, or a pharmaceutically acceptable salt thereof, wherein:
each R1 is independently halo, -CN, -OH, C1-C6-alkyl, C1-C6-haloalkyl, C1-C6- hydroxyalkyl, C1-C6 alkoxy, C3-C6-cycloalkyl, -O-C3-C6-cycloalkyl, 4- to 6-membered heterocycloalkyl, -NR1aR1b, phenyl, 6-membered heteroaryl, -X1-C(O)NR1aR1b, -X1-O- (C1-C3-alkyl), -X1-C3-C6-cycloalkyl, and -X1-phenyl; wherein each 6-membered heteroaryl has 1-2 ring heteroatoms independently selected from N and O; each 4- to 6-membered heterocycloalkyl has 1-3 ring heteroatoms independently selected from N, O, and S; and each R1 is unsubstituted or substituted with 1-3 R1c; each R1a and R1b is independently H, C1-C3 alkyl, or C3-C6 cycloalkyl; each R1c is independently halo or -OH; and and each X1 is C1-C3-alkylene. 10. The compound according to any one of claims 1-9, or a pharmaceutically acceptable salt thereof, wherein R1 is selected from the group consisting of -F, -Cl, -Br, -CN, -OH, , , , , ,
11. The compound according to claim 1, having a structure according to Formula III:
or a pharmaceutically acceptable salt thereof, wherein: A is (i) selected from the group consisting of: , ,
(ii) a 6-membered heteroaryl having 1 or 2 ring nitrogen atoms, wherein the 6- membered heteroaryl is substituted with 0-2 R1d; or (iii) -NHC(O)-R1g; each R1 is independently selected from the group consisting of halo, CN, -OH, C1- C6-alkyl, C1-C6-haloalkyl, C1-C6-hydroxyalkyl, C1-C6 alkoxy, C3-C6-cycloalkyl, -O-C3- C6-cycloalkyl, 4- to 8-membered heterocycloalkyl, -NR1aR1b, phenyl, 5- to 6-membered heteroaryl, -X1-C(O)NR1aR1b, -X1-O-(C1-C3-alkyl), -X1-C3-C6-cycloalkyl, and -X1-phenyl, wherein each 5- to 6-membered heteroaryl has 1-2 ring heteroatoms independently selected from N and O; each 4- to 8-membered heterocycloalkyl has 1-3 ring heteroatoms independently selected from N, O, and S; and each R1 is optionally substituted with 1-3 R1c; each X1 is C1-C3-alkylene; R1a and R1b, when present, are independently H, C1-C6 alkyl, or C3-C6 cycloalkyl; each R1c, when present, is independently halo, or -OH;
each R1d, when present, is independently halo, C1-C6 alkoxy, C3-C6-cycloalkyl, -O-C3-C6-cycloalkyl, or -NR1eR1f; R1e and R1f, when present, are independently H, C1-C6 haloalkyl, -(C1-C3-alkylene)- (C3-C6-cycloalkyl), or 4- to 8-membered heterocycloalkyl having 1-3 ring heteroatoms independently selected from N, O, and S; or R1e and R1f together with the nitrogen atom to which they are attached form a 4- to 6-membered heterocycloalkyl group having 1-2 ring heteroatoms independently selected from N, and O; R1g, when present, is C3-C6-cycloalkyl, -(C1-C3-alkylene)-(C3-C6-cycloalkyl), or 6- membered heteroaryl having 1 or 2 ring nitrogen atoms; wherein R1g is optionally substituted with 0-2 R1h; each R1h, when present, is independently halo, C1-C6 alkyl, -O-C3-C6-cycloalkyl, or -NH-(4- to 8-membered heterocycloalkyl), wherein the 4- to 8-membered heterocycloalkyl has 1-2 ring heteroatoms independently selected from N, O, and S; n is 0 or 1; m is 0, 1, or 2; R2, when present, is halo, -CN, -C1-C3-alkyl, -C1-C3-alkoxy, or C3-C6-cycloalkyl; R3 is halo, C1-C3 alkyl, or C1-C3 hydroxyalkyl; each R4, when present, is independently halo, C1-C6 alkyl, C1-C6-alkoxy, C3-C6 cycloalkyl, 4- to 8-membered heterocycloalkyl having 1-3 ring heteroatoms independently selected from N, O, and S, or phenyl, wherein said phenyl is optionally substituted with 1- 3 halo; and R5 and R6 are H. 12. The compound according to claim 11, having a structure according to Formula IIIa:
) or a pharmaceutically acceptable salt thereof. 13. A compound selected from the group consisting of:
, , , , , , , , ,
F
, , , , , ,
15. A pharmaceutical composition comprising a compound according to any one of claims 1-14, or a pharmaceutically acceptable salt thereof, and a pharmaceutically acceptable excipient. 16. A method of treating a disease, disorder, or condition, mediated at least in part by mast cell activation, said method comprising administering a compound of any one of claims 1-14, or
a pharmaceutically acceptable salt thereof, or the pharmaceutical composition according to claim 15, to a subject in need thereof. 17. The method according to claim 16, wherein the compound or pharmaceutical composition is administered in an effective amount to inhibit KIT. 18. The method according to claim 16 or 17, wherein the compound or pharmaceutical composition is administered in an amount sufficient to diminish the activity and/or quantity of systemic mast cells in the subject. 19. The method according to any one of claims 16-18, wherein the disease, disorder, or condition is an allergic, inflammatory, neuroinflammatory, neurological, immune, autoimmune, dermatological, respiratory, or metabolic disease, disorder, or condition. 20. The method according to claim 19, wherein the disease, disorder, or condition is arthritis, rheumatoid arthritis, inflammatory arthritis, osteoarthritis, asthma, bullous dermatosis, alopecia areata, chronic rhinosinusitis with nasal polyps (CRSwNP), coeliac disease, systemic sclerosis, netherton syndrome, idiopathic anaphylaxis, migraine, chronic graft verse host disease, multiple sclerosis, Alzheimer’s disease, autism, psoriasis, Crohn’s disease, inflammatory bowel disease, irritable bowel syndrome, lupus, Grave’s disease, Hashimoto’s thyroiditis, ankylosing spondylitis, Sjӧgren’s syndrome (SjS), angioedema, allergic asthma, eosinophilic asthma, anaphylaxis, atopic dermatitis, food allergies, allergic conjunctivitis, allergic rhinitis, urticaria (e.g., chronic spontaneous urticaria (CSU), acute urticaria, or physical urticaria including popular urticaria, cold urticaria, cholinergic urticaria, solar urticaria, scleroderma, and dermatographic urticaria), mastocytosis, dermographism, systemic mastocytosis, mast cell activation syndrome, mast cell gastrointestinal disease, dermatitis herpetiformis, dermatosis, dermatitis, allergic contact dermatitis, eosinophilic gastrointestinal disease, eosinophilic esophagitis, type I diabetes, type II diabetes, or prurigo nodularis. 21. The method according to any one of claims 16-18, wherein the disease, disorder, or condition is a cardiovascular or fibrotic disease, disorder, or condition.
22. The method according to claim 21, wherein the cardiovascular or fibrotic disease, disorder, or condition is coronary heart disease, atherosclerosis, myocardial infarction, angina, osteoarthritis, pulmonary fibrosis, idiopathic pulmonary fibrosis, pulmonary arterial hypertension, primary pulmonary hypertension, hepatic fibrosis, renal fibrosis, cardiac fibrosis, cystic fibrosis, bronchitis, or asthma. 23. The method according to any one of claims 16-22, wherein the disease, disorder or condition is antihistamine-refractory. 24. The method according to any one of claims 16-22, wherein the disease, disorder or condition is refractory to IgE inhibitor therapy. 25. The method according to any one of claims 16-22, wherein the disease, disorder or condition is contraindicated or resistant to antihistamines. 26. The method according to any one of claims 16-25, further comprising administering at least one additional therapeutic agent to the subject. 27. The method according to claim 26, wherein the at least one additional therapeutic agent comprises one or more of an antihistamine, a BTK inhibitor, and/or an IgE inhibitor. 28. The method according to claim 27, wherein the IgE inhibitor is omalizumab, or ligelizumab. 29. The method according to claim 27 or claim 28, wherein the BTK inhibitor is ibrutinib, acalabrutinib, remibrutinib, or zanubrutinib. 30. The method according to any one of claims 26-29, wherein the additional therapeutic agent comprises an anti-inflammatory agent, an immunosuppressive agent, an agent that targets one or more cytokines, or combinations thereof. 31. The method according to claim 30, wherein the anti-inflammatory agent is a corticosteroid. 32. The method according to claim 30 or claim 31, wherein the immunosuppressive agent is cyclosporine.
33. The method according to any one of claims 30-32, wherein the agent that targets one or more cytokines targets IL-4 and/or IL-13. 34. The method according to claim 33, wherein the agent that targets one or more cytokines is dupilumab. 35. The method according to any one of claims 26-34, wherein the additional therapeutic agent comprises a leukotriene modifier. 36. The method according to any one of claims 16-18, 21, or 22, further comprising administering at least one additional therapeutic agent to the subject. 37. The method according to claim 36, wherein the additional therapeutic agent comprises a cholesterol modifier, a diuretic, an antiarrhythmic, a vasodilator, an anti-inflammatory, an analgesic agent, or any combination thereof. 38. The method according to any one of claims 16-18, wherein the disease, disorder, or condition is cancer. 39. The method according to claim 38, wherein the cancer is melanoma, prostate cancer, pancreatic cancer, squamous cell carcinoma, Hodgkin lymphoma, leukemia (e.g., chronic myeloid leukemia, chronic eosinophilic leukemia, chronic myelomonocytic leukemia, myeloid leukemia, chronic myeloid leukemia, acute myeloid leukemia, acute megakaryoblastic leukemia, mast cell leukemia, acute lymphocytic leukemia), gastric cancer (e.g., gastrointenstinal stromal cancer), thyroid cancer, breast cancer, endometrial cancer, cervical cancer, esophageal cancer, lung cancer (e.g., small cell and non-small cell lung cancer), colorectal cancer, prostate cancer, liver cancer, bile duct cancer, gallbladder cancer, neuroendocrine tumors, kidney cancer, head and neck cancer, bone cancer, brain cancer (e.g., glioblastoma, medulloblastoma), mesothelioma, or soft tissue sarcoma. 40. The method according to claim 38 or claim 39, further comprising administering at least one additional therapeutic agent.
41. The method according to claim 40, wherein the at least one additional therapeutic agent comprises an immune checkpoint inhibitor, an A2R antagonist, a CD73 inhibitor, a HIF-2α inhibitor, a chemotherapeutic agent, radiation therapy, or any combinations thereof. 42. The method according to claim 41, wherein the immune checkpoint inhibitor comprises one or more inhibitors that block the activity of at least one of PD-1, PD-L1, BTLA, LAG-3, a B7 family member, TIM-3, TIGIT or CTLA-4. 43. The method according to claim 42, wherein said immune checkpoint inhibitor comprises an inhibitor of PD-1 or PD-L1. 44. The method according to claim 43, wherein said immune checkpoint inhibitor is selected from the group consisting of avelumab, atezolizumab, durvalumab, dostarlimab, cemiplimab, nivolumab, pembrolizumab, sintilmab, toripalimab, and zimberelimab. 45. The method according to claim 44, wherein said immune checkpoint inhibitor is zimberelimab. 46. The method according to any one of claims 42-45, wherein said immune checkpoint inhibitor comprises an inhibitor that blocks the activity of TIGIT. 47. The method according to claim 46, wherein the immune checkpoint inhibitor is domvanalimab or AB308. 48. A method of preventing an allergic, immune, or autoimmune response in a subject in need thereof, said method comprising administering a compound according to any one of claims 1-14, or a pharmaceutically acceptable salt thereof, or the pharmaceutical composition according to claim 15 to the subject. 49. The method according to claim 48, wherein the subject suffers from an allergic, immune, or autoimmune disease, disorder or condition. 50. The method according to claim 49, wherein the allergic, immune, or autoimmune disease, disorder or condition is arthritis, asthma, bullous dermatosis, alopecia areata, chronic rhinosinusitis with nasal polyps (CRSwNP), coeliac disease, systemic sclerosis, netherton
syndrome, idiopathic anaphylaxis, migraine, chronic graft verse host disease, multiple sclerosis, psoriasis, Crohn’s disease, inflammatory bowel disease, irritable bowel syndrome, lupus, Grave’s disease, Hashimoto’s thyroiditis, ankylosing spondylitis, Sjӧgren’s syndrome (SjS), angioedema, allergic asthma, eosinophilic asthma, anaphylaxis, atopic dermatitis, food allergies, allergic conjunctivitis, allergic rhinitis, urticaria (e.g., chronic spontaneous urticaria (CSU), acute urticaria, or physical urticaria including popular urticaria, cold urticaria, cholinergic urticaria, solar urticaria, scleroderma, and dermatographic urticaria), mastocytosis, dermographism, systemic mastocytosis, systemic mastocytosis, mast cell activation syndrome, mast cell gastrointestinal disease, dermatitis herpetiformis, dermatosis, dermatitis, allergic contact dermatitis, eosinophilic gastrointestinal disease, eosinophilic esophagitis, type I diabetes, type II diabetes, or prurigo nodularis. 51. The method according to any one of claims 48-50, wherein the disease, disorder or condition is antihistamine-refractory. 52. The method according to any one of claims 48-50, wherein the disease, disorder or condition is refractory to IgE inhibitor therapy. 53. The method according to any one of claims 48-50, wherein the disease, disorder or condition is contraindicated or resistant to antihistamines. 54. The method according to any one of claims 48-53, wherein the method further comprises administering at least one additional therapeutic agent to the subject. 55. The method according to claim 54, wherein the at least one additional therapeutic agent comprises one or more of an antihistamine, a BTK inhibitor, and/or an IgE inhibitor. 56. The method according to claim 55, wherein the IgE inhibitor is omalizumab, or ligelizumab. 57. The method according to claim 55, wherein the BTK inhibitor is ibrutinib, acalabrutinib, remibrutinib, or zanubrutinib. 58. The method according to any one of claims 54-57, wherein the additional therapeutic agent comprises an anti-inflammatory agent, an immunosuppressive agent, an agent that targets one or more cytokines, or combinations thereof.
59. The method according to claim 58, wherein the anti-inflammatory agent is a corticosteroid. 60. The method according to claim 58 or claim 59, wherein the immunosuppressive agent is cyclosporine. 61. The method according to any one of claims 58-60, wherein the agent that targets one or more cytokines targets IL-4 and/or IL-13. 62. The method according to claim 61, wherein the agent that targets one or more cytokines is dupilumab. 63. The method according to any one of claims 54-62, wherein the additional therapeutic agent comprises a leukotriene modifier.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US202363540604P | 2023-09-26 | 2023-09-26 | |
| US63/540,604 | 2023-09-26 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2025072330A1 true WO2025072330A1 (en) | 2025-04-03 |
Family
ID=93010717
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2024/048409 Pending WO2025072330A1 (en) | 2023-09-26 | 2024-09-25 | Kit inhibitor compounds and methods of use thereof |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2025072330A1 (en) |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US12448379B2 (en) | 2022-11-30 | 2025-10-21 | Blueprint Medicines Corporation | Wild type kit inhibitors |
| WO2025256552A1 (en) * | 2024-06-11 | 2025-12-18 | 上海翰森生物医药科技有限公司 | Pyrazole derivative inhibitor, preparation method therefor and use thereof |
Citations (29)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2006105021A2 (en) | 2005-03-25 | 2006-10-05 | Tolerrx, Inc. | Gitr binding molecules and uses therefor |
| WO2008009487A1 (en) * | 2006-03-10 | 2008-01-24 | Novartis Ag | Heterobicyclic carboxamides as inhibitors for kinases |
| WO2008132601A1 (en) | 2007-04-30 | 2008-11-06 | Immutep | Cytotoxic anti-lag-3 monoclonal antibody and its use in the treatment or prevention of organ transplant rejection and autoimmune disease |
| WO2009009116A2 (en) | 2007-07-12 | 2009-01-15 | Tolerx, Inc. | Combination therapies employing gitr binding molecules |
| WO2009044273A2 (en) | 2007-10-05 | 2009-04-09 | Immutep | Use of recombinant lag-3 or the derivatives thereof for eliciting monocyte immune response |
| WO2010019570A2 (en) | 2008-08-11 | 2010-02-18 | Medarex, Inc. | Human antibodies that bind lymphocyte activation gene-3 (lag-3), and uses thereof |
| WO2011028683A1 (en) | 2009-09-03 | 2011-03-10 | Schering Corporation | Anti-gitr antibodies |
| WO2011109400A2 (en) | 2010-03-04 | 2011-09-09 | Macrogenics,Inc. | Antibodies reactive with b7-h3, immunologically active fragments thereof and uses thereof |
| WO2012032433A1 (en) | 2010-09-09 | 2012-03-15 | Pfizer Inc. | 4-1bb binding molecules |
| WO2012145493A1 (en) | 2011-04-20 | 2012-10-26 | Amplimmune, Inc. | Antibodies and other molecules that bind b7-h1 and pd-1 |
| WO2013033167A1 (en) * | 2011-09-01 | 2013-03-07 | Irm Llc | Compounds and compositions as c-kit kinase inhibitors |
| WO2014008218A1 (en) | 2012-07-02 | 2014-01-09 | Bristol-Myers Squibb Company | Optimization of antibodies that bind lymphocyte activation gene-3 (lag-3), and uses thereof |
| WO2017120508A1 (en) | 2016-01-08 | 2017-07-13 | Arcus Biosciences, Inc. | Modulators of 5'-nucleotidase, ecto and the use thereof |
| WO2017152088A1 (en) | 2016-03-04 | 2017-09-08 | JN Biosciences, LLC | Antibodies to tigit |
| WO2018067424A1 (en) | 2016-10-03 | 2018-04-12 | Arcus Biosciences, Inc. | Inhibitors of adenosine 5'-nucleotidase |
| WO2018094148A1 (en) | 2016-11-18 | 2018-05-24 | Arcus Biosciences, Inc. | Inhibitors of cd73-mediated immunosuppression |
| WO2018136700A1 (en) | 2017-01-20 | 2018-07-26 | Arcus Biosciences, Inc. | Azolopyrimidine for the treatment of cancer-related disorders |
| WO2018204661A1 (en) | 2017-05-05 | 2018-11-08 | Arcus Biosciences, Inc. | Quinazoline-pyridine derivatives for the treatment of cancer-related disorders |
| WO2018213377A1 (en) | 2017-05-17 | 2018-11-22 | Arcus Biosciences, Inc. | Quinazoline-pyrazole derivatives for the treatment of cancer-related disorders |
| WO2020023846A1 (en) | 2018-07-27 | 2020-01-30 | Arcus Biosciences, Inc. | Pyridone a2r antagonists |
| WO2020046813A1 (en) | 2018-08-27 | 2020-03-05 | Arcus Biosciences, Inc. | Cd73 inhibitors |
| WO2021113436A1 (en) | 2019-12-04 | 2021-06-10 | Arcus Biosciences, Inc. | Inhibitors of hif-2alpha |
| WO2021188769A1 (en) | 2020-03-19 | 2021-09-23 | Arcus Biosciences, Inc. | Tetralin and tetrahydroquinoline compounds as inhibitors of hif-2alpha |
| WO2021247591A1 (en) | 2020-06-02 | 2021-12-09 | Arcus Biosciences, Inc. | Antibodies to tigit |
| WO2022246177A1 (en) | 2021-05-21 | 2022-11-24 | Arcus Biosciences, Inc. | Axl compounds |
| WO2022246179A1 (en) | 2021-05-21 | 2022-11-24 | Arcus Biosciences, Inc. | Axl inhibitor compounds |
| US20230069124A1 (en) | 2021-08-24 | 2023-03-02 | Red Hat, Inc. | Schema based type-coercion for structured documents |
| WO2023077046A1 (en) | 2021-10-29 | 2023-05-04 | Arcus Biosciences, Inc. | Inhibitors of hif-2alpha and methods of use thereof |
| WO2024118887A1 (en) * | 2022-11-30 | 2024-06-06 | Blueprint Medicines Corporation | N-phenyl-pyrazolo[1,5-a]pyridine-3-carboxamide derivatives as wild type c-kit kinase inhibitors for the treatment of urticaria |
-
2024
- 2024-09-25 WO PCT/US2024/048409 patent/WO2025072330A1/en active Pending
Patent Citations (29)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2006105021A2 (en) | 2005-03-25 | 2006-10-05 | Tolerrx, Inc. | Gitr binding molecules and uses therefor |
| WO2008009487A1 (en) * | 2006-03-10 | 2008-01-24 | Novartis Ag | Heterobicyclic carboxamides as inhibitors for kinases |
| WO2008132601A1 (en) | 2007-04-30 | 2008-11-06 | Immutep | Cytotoxic anti-lag-3 monoclonal antibody and its use in the treatment or prevention of organ transplant rejection and autoimmune disease |
| WO2009009116A2 (en) | 2007-07-12 | 2009-01-15 | Tolerx, Inc. | Combination therapies employing gitr binding molecules |
| WO2009044273A2 (en) | 2007-10-05 | 2009-04-09 | Immutep | Use of recombinant lag-3 or the derivatives thereof for eliciting monocyte immune response |
| WO2010019570A2 (en) | 2008-08-11 | 2010-02-18 | Medarex, Inc. | Human antibodies that bind lymphocyte activation gene-3 (lag-3), and uses thereof |
| WO2011028683A1 (en) | 2009-09-03 | 2011-03-10 | Schering Corporation | Anti-gitr antibodies |
| WO2011109400A2 (en) | 2010-03-04 | 2011-09-09 | Macrogenics,Inc. | Antibodies reactive with b7-h3, immunologically active fragments thereof and uses thereof |
| WO2012032433A1 (en) | 2010-09-09 | 2012-03-15 | Pfizer Inc. | 4-1bb binding molecules |
| WO2012145493A1 (en) | 2011-04-20 | 2012-10-26 | Amplimmune, Inc. | Antibodies and other molecules that bind b7-h1 and pd-1 |
| WO2013033167A1 (en) * | 2011-09-01 | 2013-03-07 | Irm Llc | Compounds and compositions as c-kit kinase inhibitors |
| WO2014008218A1 (en) | 2012-07-02 | 2014-01-09 | Bristol-Myers Squibb Company | Optimization of antibodies that bind lymphocyte activation gene-3 (lag-3), and uses thereof |
| WO2017120508A1 (en) | 2016-01-08 | 2017-07-13 | Arcus Biosciences, Inc. | Modulators of 5'-nucleotidase, ecto and the use thereof |
| WO2017152088A1 (en) | 2016-03-04 | 2017-09-08 | JN Biosciences, LLC | Antibodies to tigit |
| WO2018067424A1 (en) | 2016-10-03 | 2018-04-12 | Arcus Biosciences, Inc. | Inhibitors of adenosine 5'-nucleotidase |
| WO2018094148A1 (en) | 2016-11-18 | 2018-05-24 | Arcus Biosciences, Inc. | Inhibitors of cd73-mediated immunosuppression |
| WO2018136700A1 (en) | 2017-01-20 | 2018-07-26 | Arcus Biosciences, Inc. | Azolopyrimidine for the treatment of cancer-related disorders |
| WO2018204661A1 (en) | 2017-05-05 | 2018-11-08 | Arcus Biosciences, Inc. | Quinazoline-pyridine derivatives for the treatment of cancer-related disorders |
| WO2018213377A1 (en) | 2017-05-17 | 2018-11-22 | Arcus Biosciences, Inc. | Quinazoline-pyrazole derivatives for the treatment of cancer-related disorders |
| WO2020023846A1 (en) | 2018-07-27 | 2020-01-30 | Arcus Biosciences, Inc. | Pyridone a2r antagonists |
| WO2020046813A1 (en) | 2018-08-27 | 2020-03-05 | Arcus Biosciences, Inc. | Cd73 inhibitors |
| WO2021113436A1 (en) | 2019-12-04 | 2021-06-10 | Arcus Biosciences, Inc. | Inhibitors of hif-2alpha |
| WO2021188769A1 (en) | 2020-03-19 | 2021-09-23 | Arcus Biosciences, Inc. | Tetralin and tetrahydroquinoline compounds as inhibitors of hif-2alpha |
| WO2021247591A1 (en) | 2020-06-02 | 2021-12-09 | Arcus Biosciences, Inc. | Antibodies to tigit |
| WO2022246177A1 (en) | 2021-05-21 | 2022-11-24 | Arcus Biosciences, Inc. | Axl compounds |
| WO2022246179A1 (en) | 2021-05-21 | 2022-11-24 | Arcus Biosciences, Inc. | Axl inhibitor compounds |
| US20230069124A1 (en) | 2021-08-24 | 2023-03-02 | Red Hat, Inc. | Schema based type-coercion for structured documents |
| WO2023077046A1 (en) | 2021-10-29 | 2023-05-04 | Arcus Biosciences, Inc. | Inhibitors of hif-2alpha and methods of use thereof |
| WO2024118887A1 (en) * | 2022-11-30 | 2024-06-06 | Blueprint Medicines Corporation | N-phenyl-pyrazolo[1,5-a]pyridine-3-carboxamide derivatives as wild type c-kit kinase inhibitors for the treatment of urticaria |
Non-Patent Citations (2)
| Title |
|---|
| BERGE, S.M. ET AL.: "Journal of Pharmaceutical Science", vol. 66, 1977, article "Pharmaceutical Salts", pages: 1 - 19 |
| PARDOLL, NATURE REV. CANCER, vol. 12, April 2012 (2012-04-01), pages 252 - 64 |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US12448379B2 (en) | 2022-11-30 | 2025-10-21 | Blueprint Medicines Corporation | Wild type kit inhibitors |
| WO2025256552A1 (en) * | 2024-06-11 | 2025-12-18 | 上海翰森生物医药科技有限公司 | Pyrazole derivative inhibitor, preparation method therefor and use thereof |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US12410170B2 (en) | 2,3,5-trisubstituted pyrazolo[1,5-A]pyrimidine compounds | |
| US12103907B2 (en) | Tetralin and tetrahydroquinoline compounds as inhibitors of HIF-2alpha | |
| EP4069212A1 (en) | Inhibitors of hif-2alpha | |
| US20230339902A1 (en) | Tricyclic ligands for degradation of ikzf2 or ikzf4 | |
| WO2025072330A1 (en) | Kit inhibitor compounds and methods of use thereof | |
| KR102874347B1 (en) | compound | |
| US20240246967A1 (en) | Axl compounds | |
| AU2018210272A1 (en) | Azolopyrimidine for the treatment of cancer-related disorders | |
| RS56657B1 (en) | Novel amino pyrimidine derivatives | |
| TW202448871A (en) | Azolylpyridine pyridazinone amides as sos1 inhibitors | |
| CA3161339A1 (en) | Cyclic compounds and methods of using same | |
| TW201902895A (en) | 6-5 fused ring as a C5a inhibitor | |
| CA3090150A1 (en) | Ahr modulators | |
| US11396502B2 (en) | Substituted heterocyclic derivatives as PI3K inhibitors | |
| US20260028336A1 (en) | Cbl-b inhibitors and methods of use thereof | |
| US20240300936A1 (en) | Preparation of substituted 1,2-diaminoheterocyclic compound derivatives and their use as pharmaceutical agents | |
| US12384773B2 (en) | Thiazole compounds and methods of use thereof | |
| WO2025054339A1 (en) | Triazolopyridine compounds as inhibitors of kit | |
| RU2800063C2 (en) | Compounds | |
| US20250011318A1 (en) | Cbl-b inhibitors and methods of use thereof | |
| US20250352549A1 (en) | Substituted heterocyclic compound derivatives and their pharmaceutical use | |
| HK40116573A (en) | Compounds as inhibitors of axl | |
| HK40076614A (en) | Inhibitors of hif-2alpha | |
| HK40068316A (en) | 2,3,5-trisubstituted pyrazolo[1,5-a]pyrimidine compounds | |
| HK40027765B (en) | Quinazoline-pyrazole derivatives for the treatment of cancer-related disorders |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 24786333 Country of ref document: EP Kind code of ref document: A1 |