WO2025068452A1 - Guanfacine derivatives and their use in treating cancer - Google Patents
Guanfacine derivatives and their use in treating cancer Download PDFInfo
- Publication number
- WO2025068452A1 WO2025068452A1 PCT/EP2024/077199 EP2024077199W WO2025068452A1 WO 2025068452 A1 WO2025068452 A1 WO 2025068452A1 EP 2024077199 W EP2024077199 W EP 2024077199W WO 2025068452 A1 WO2025068452 A1 WO 2025068452A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- alkyl
- cycloalkyl
- optionally substituted
- halogen
- hydroxy
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C279/00—Derivatives of guanidine, i.e. compounds containing the group, the singly-bound nitrogen atoms not being part of nitro or nitroso groups
- C07C279/20—Derivatives of guanidine, i.e. compounds containing the group, the singly-bound nitrogen atoms not being part of nitro or nitroso groups containing any of the groups, X being a hetero atom, Y being any atom, e.g. acylguanidines
- C07C279/22—Y being a hydrogen or a carbon atom, e.g. benzoylguanidines
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C311/00—Amides of sulfonic acids, i.e. compounds having singly-bound oxygen atoms of sulfo groups replaced by nitrogen atoms, not being part of nitro or nitroso groups
- C07C311/01—Sulfonamides having sulfur atoms of sulfonamide groups bound to acyclic carbon atoms
- C07C311/02—Sulfonamides having sulfur atoms of sulfonamide groups bound to acyclic carbon atoms of an acyclic saturated carbon skeleton
- C07C311/08—Sulfonamides having sulfur atoms of sulfonamide groups bound to acyclic carbon atoms of an acyclic saturated carbon skeleton having the nitrogen atom of at least one of the sulfonamide groups bound to a carbon atom of a six-membered aromatic ring
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D209/00—Heterocyclic compounds containing five-membered rings, condensed with other rings, with one nitrogen atom as the only ring hetero atom
- C07D209/02—Heterocyclic compounds containing five-membered rings, condensed with other rings, with one nitrogen atom as the only ring hetero atom condensed with one carbocyclic ring
- C07D209/04—Indoles; Hydrogenated indoles
- C07D209/10—Indoles; Hydrogenated indoles with substituted hydrocarbon radicals attached to carbon atoms of the hetero ring
- C07D209/18—Radicals substituted by carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D211/00—Heterocyclic compounds containing hydrogenated pyridine rings, not condensed with other rings
- C07D211/04—Heterocyclic compounds containing hydrogenated pyridine rings, not condensed with other rings with only hydrogen or carbon atoms directly attached to the ring nitrogen atom
- C07D211/68—Heterocyclic compounds containing hydrogenated pyridine rings, not condensed with other rings with only hydrogen or carbon atoms directly attached to the ring nitrogen atom having one double bond between ring members or between a ring member and a non-ring member
- C07D211/70—Heterocyclic compounds containing hydrogenated pyridine rings, not condensed with other rings with only hydrogen or carbon atoms directly attached to the ring nitrogen atom having one double bond between ring members or between a ring member and a non-ring member with only hydrogen atoms, hydrocarbon or substituted hydrocarbon radicals, directly attached to ring carbon atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D231/00—Heterocyclic compounds containing 1,2-diazole or hydrogenated 1,2-diazole rings
- C07D231/02—Heterocyclic compounds containing 1,2-diazole or hydrogenated 1,2-diazole rings not condensed with other rings
- C07D231/10—Heterocyclic compounds containing 1,2-diazole or hydrogenated 1,2-diazole rings not condensed with other rings having two or three double bonds between ring members or between ring members and non-ring members
- C07D231/12—Heterocyclic compounds containing 1,2-diazole or hydrogenated 1,2-diazole rings not condensed with other rings having two or three double bonds between ring members or between ring members and non-ring members with only hydrogen atoms, hydrocarbon or substituted hydrocarbon radicals, directly attached to ring carbon atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D239/00—Heterocyclic compounds containing 1,3-diazine or hydrogenated 1,3-diazine rings
- C07D239/02—Heterocyclic compounds containing 1,3-diazine or hydrogenated 1,3-diazine rings not condensed with other rings
- C07D239/24—Heterocyclic compounds containing 1,3-diazine or hydrogenated 1,3-diazine rings not condensed with other rings having three or more double bonds between ring members or between ring members and non-ring members
- C07D239/26—Heterocyclic compounds containing 1,3-diazine or hydrogenated 1,3-diazine rings not condensed with other rings having three or more double bonds between ring members or between ring members and non-ring members with only hydrogen atoms, hydrocarbon or substituted hydrocarbon radicals, directly attached to ring carbon atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D261/00—Heterocyclic compounds containing 1,2-oxazole or hydrogenated 1,2-oxazole rings
- C07D261/02—Heterocyclic compounds containing 1,2-oxazole or hydrogenated 1,2-oxazole rings not condensed with other rings
- C07D261/06—Heterocyclic compounds containing 1,2-oxazole or hydrogenated 1,2-oxazole rings not condensed with other rings having two or more double bonds between ring members or between ring members and non-ring members
- C07D261/08—Heterocyclic compounds containing 1,2-oxazole or hydrogenated 1,2-oxazole rings not condensed with other rings having two or more double bonds between ring members or between ring members and non-ring members with only hydrogen atoms, hydrocarbon or substituted hydrocarbon radicals, directly attached to ring carbon atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D265/00—Heterocyclic compounds containing six-membered rings having one nitrogen atom and one oxygen atom as the only ring hetero atoms
- C07D265/28—1,4-Oxazines; Hydrogenated 1,4-oxazines
- C07D265/30—1,4-Oxazines; Hydrogenated 1,4-oxazines not condensed with other rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C2601/00—Systems containing only non-condensed rings
- C07C2601/02—Systems containing only non-condensed rings with a three-membered ring
Definitions
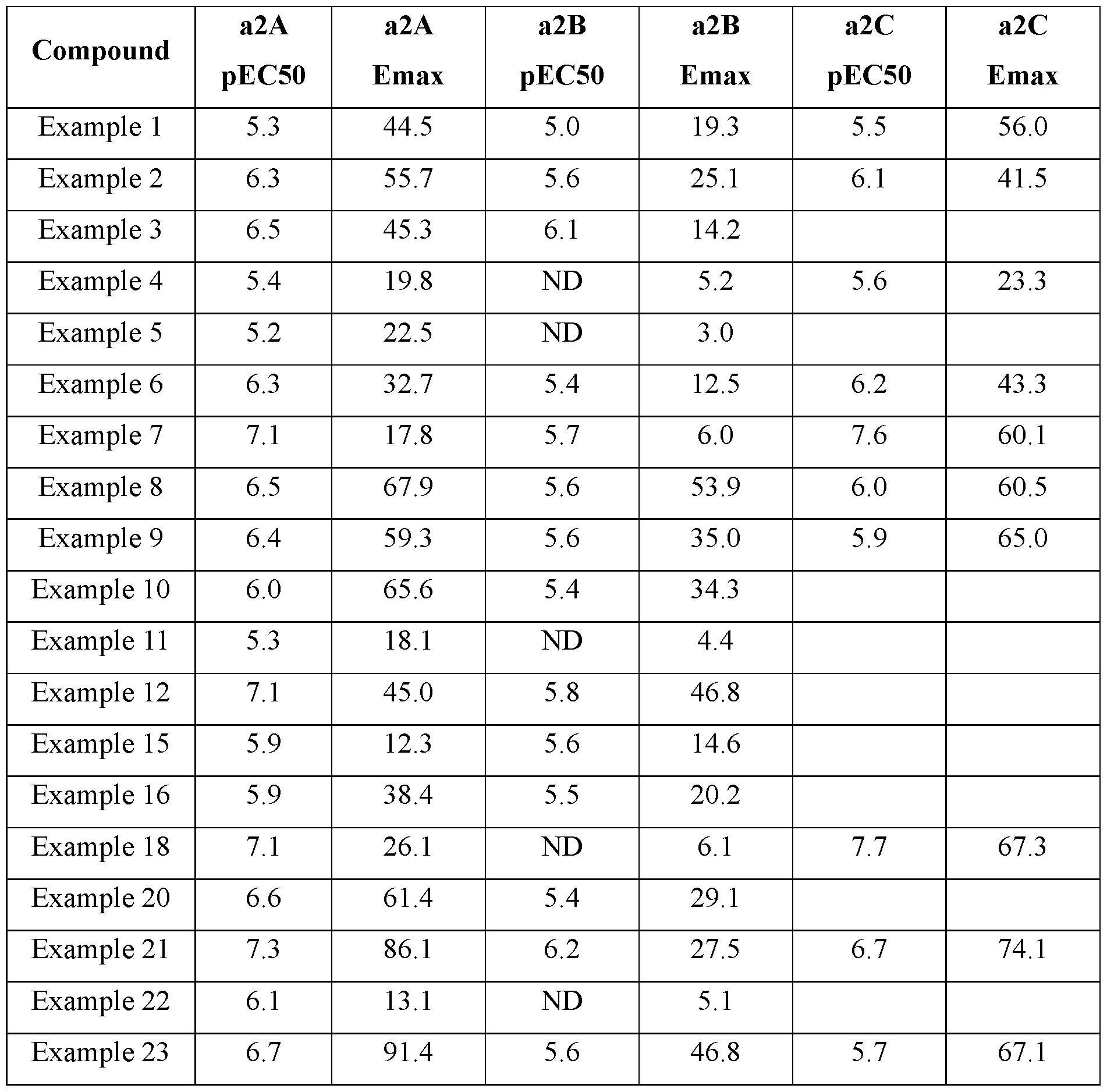
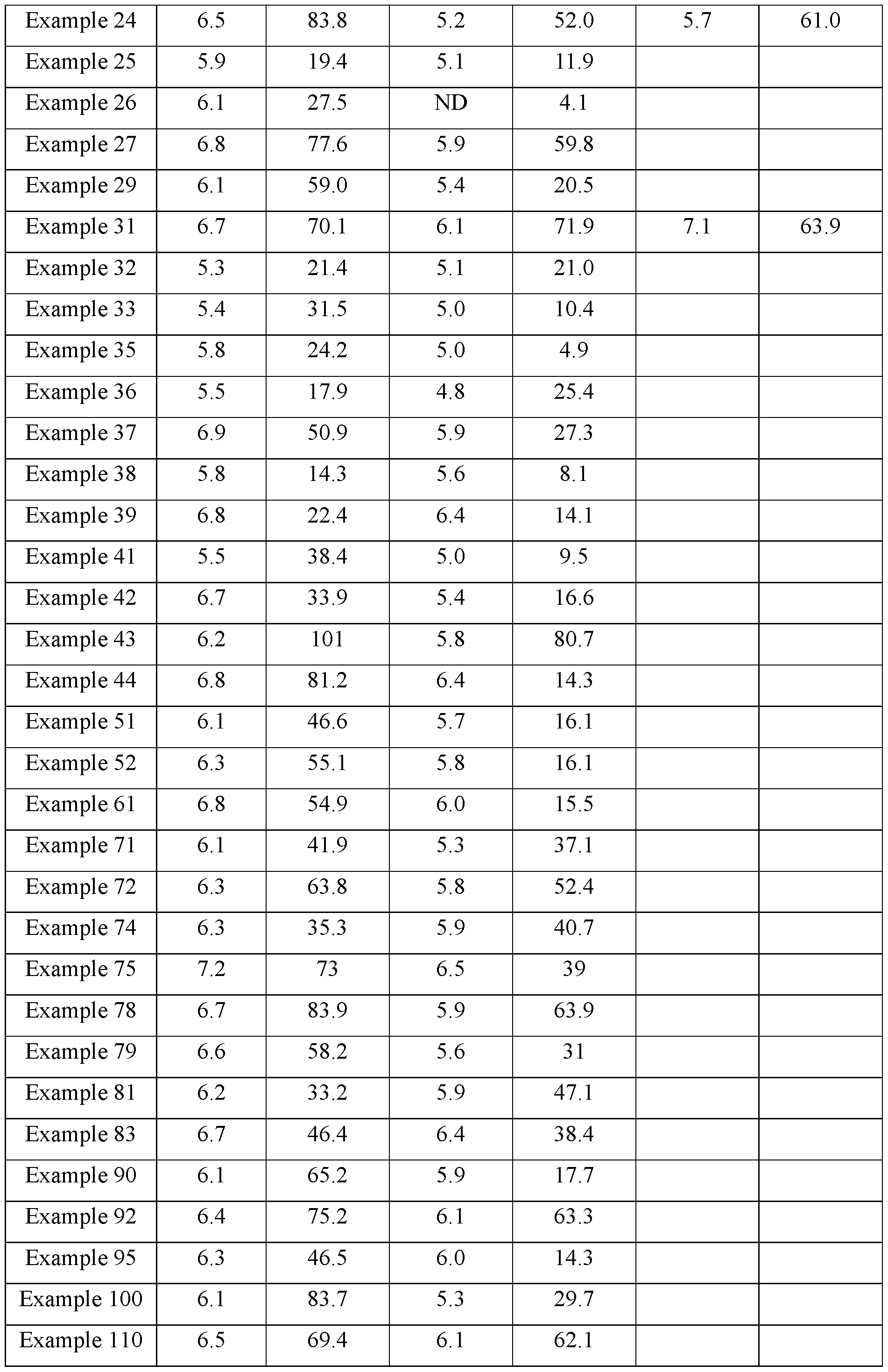
- the present invention relates to the field of medicine, in particular guanfacine derivatives and their uses for treating diseases. More particularly, the invention provides alpha2-adrenergic receptor ( ⁇ 2-AR) agonists for the treatment of cancer.
- ⁇ 2-AR alpha2-adrenergic receptor
- ⁇ 2-AR are a family of G-protein–coupled receptors with 3 pharmacological subtypes, ⁇ 2A, ⁇ 2B, and ⁇ 2C.
- Tumors from treated mice contained increased infiltrating CD8+ T lymphocytes and reduced infiltrating myeloid-derived suppressor cells (MDSC), which were more apoptotic.
- MDSC myeloid-derived suppressor cells
- Single-cell RNA sequencing revealed upregulation of innate and adaptive immune response pathways in macrophages and T cells. Consequently, the discovery and the synthesis of new ⁇ 2-AR agonists are relevant and promising approaches for anti-cancer therapies. More particularly, it could significantly improve clinical efficacy of cancer immunotherapy.
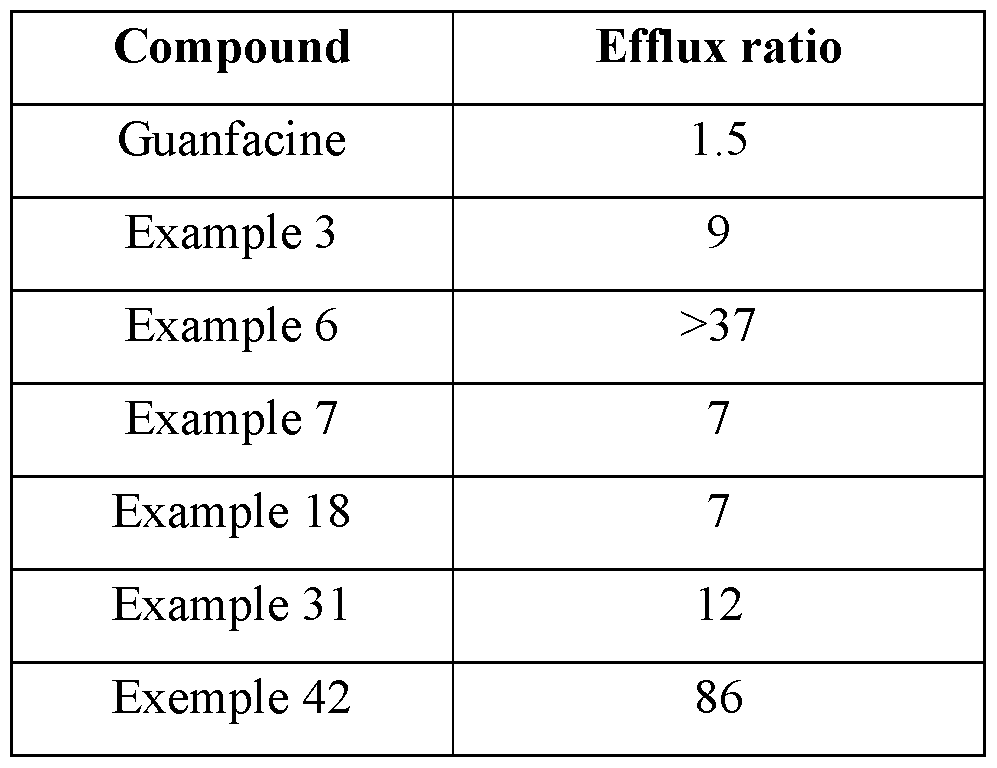
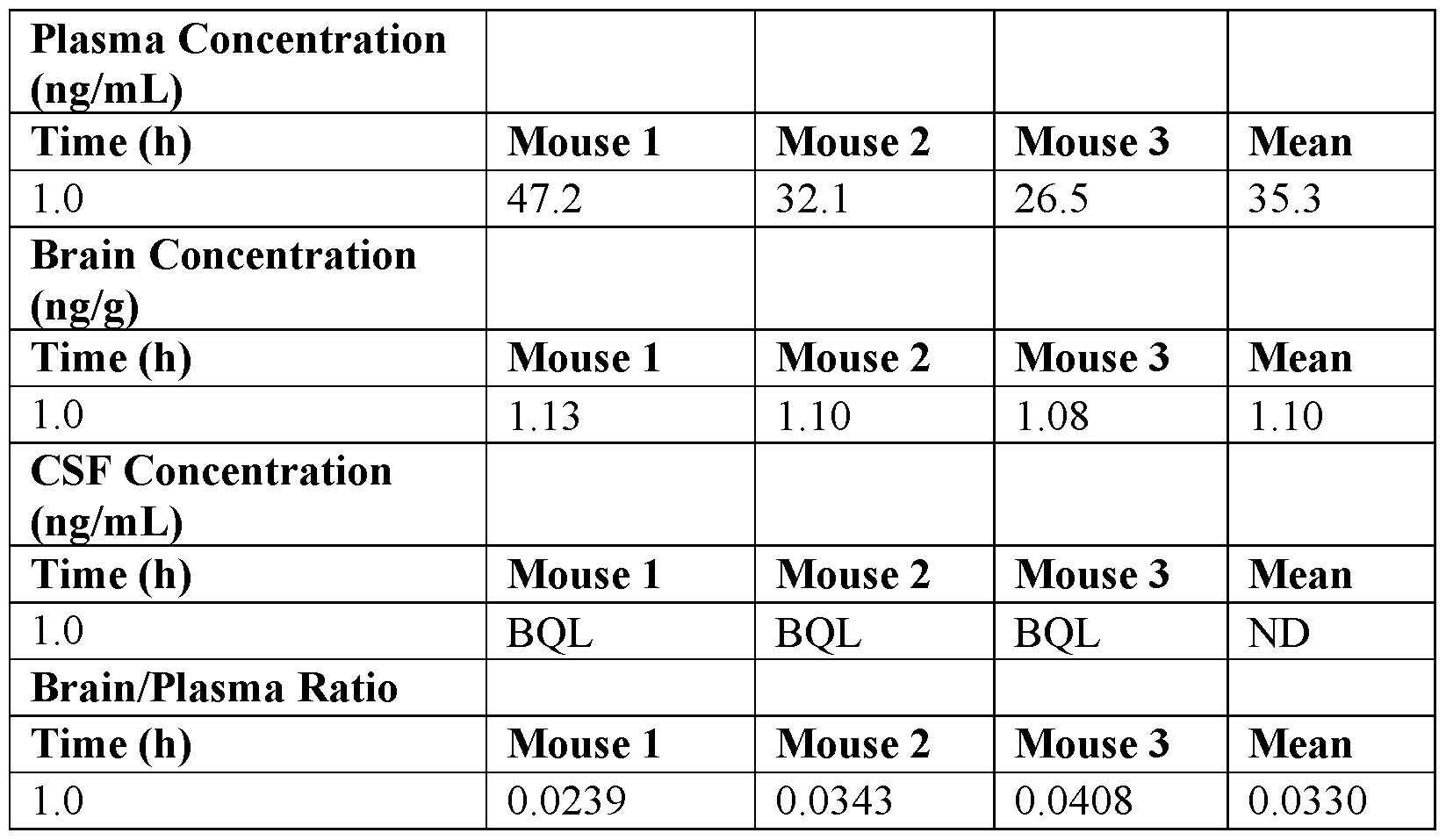
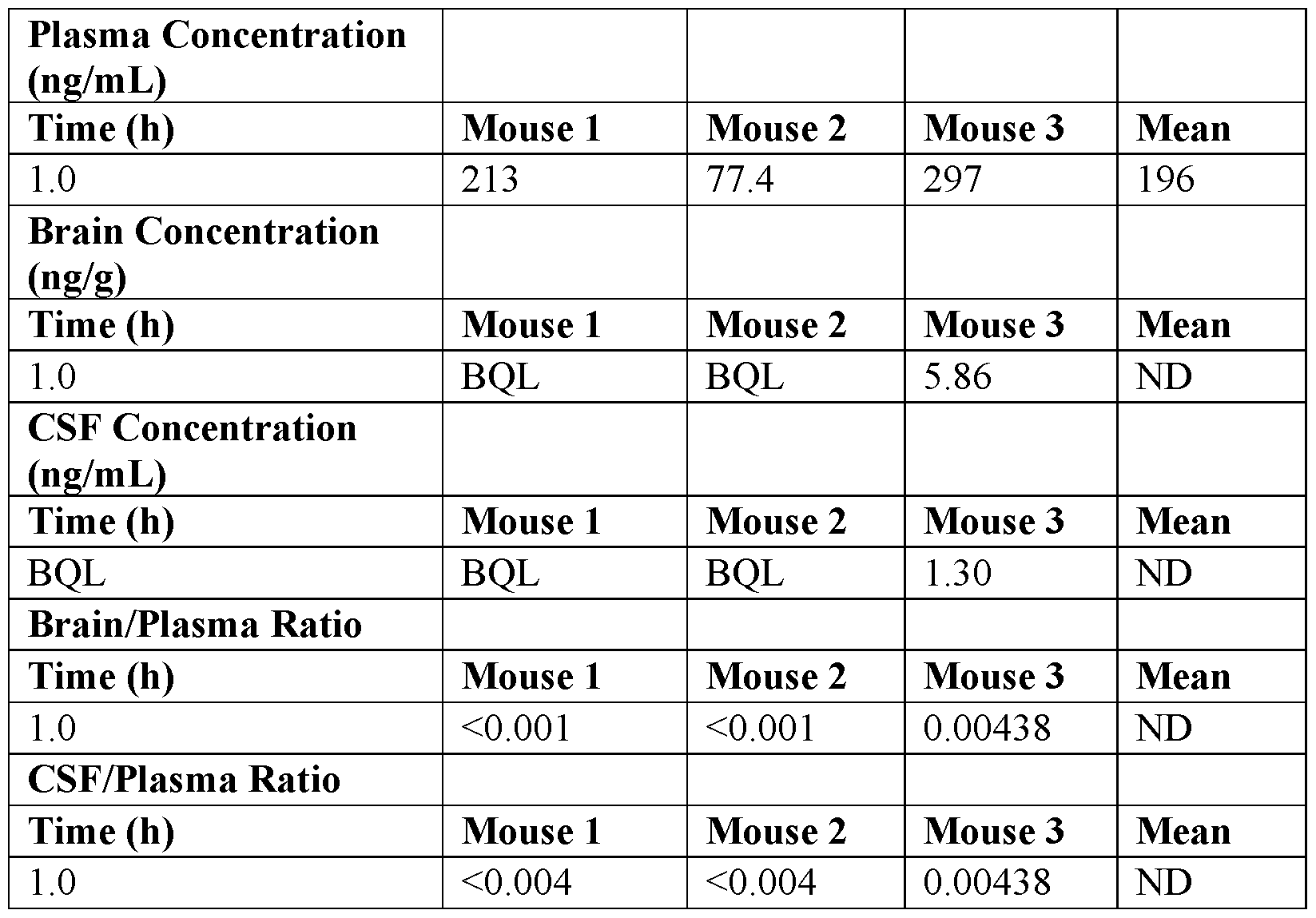
- the inventors have provided new guanfacine derivatives as efficient ⁇ 2-AR agonists while being not efficient in penetrating the BBB, demonstrating thereby the therapeutic interest of such derivatives in medicine, more particularly in anti-cancer therapies.
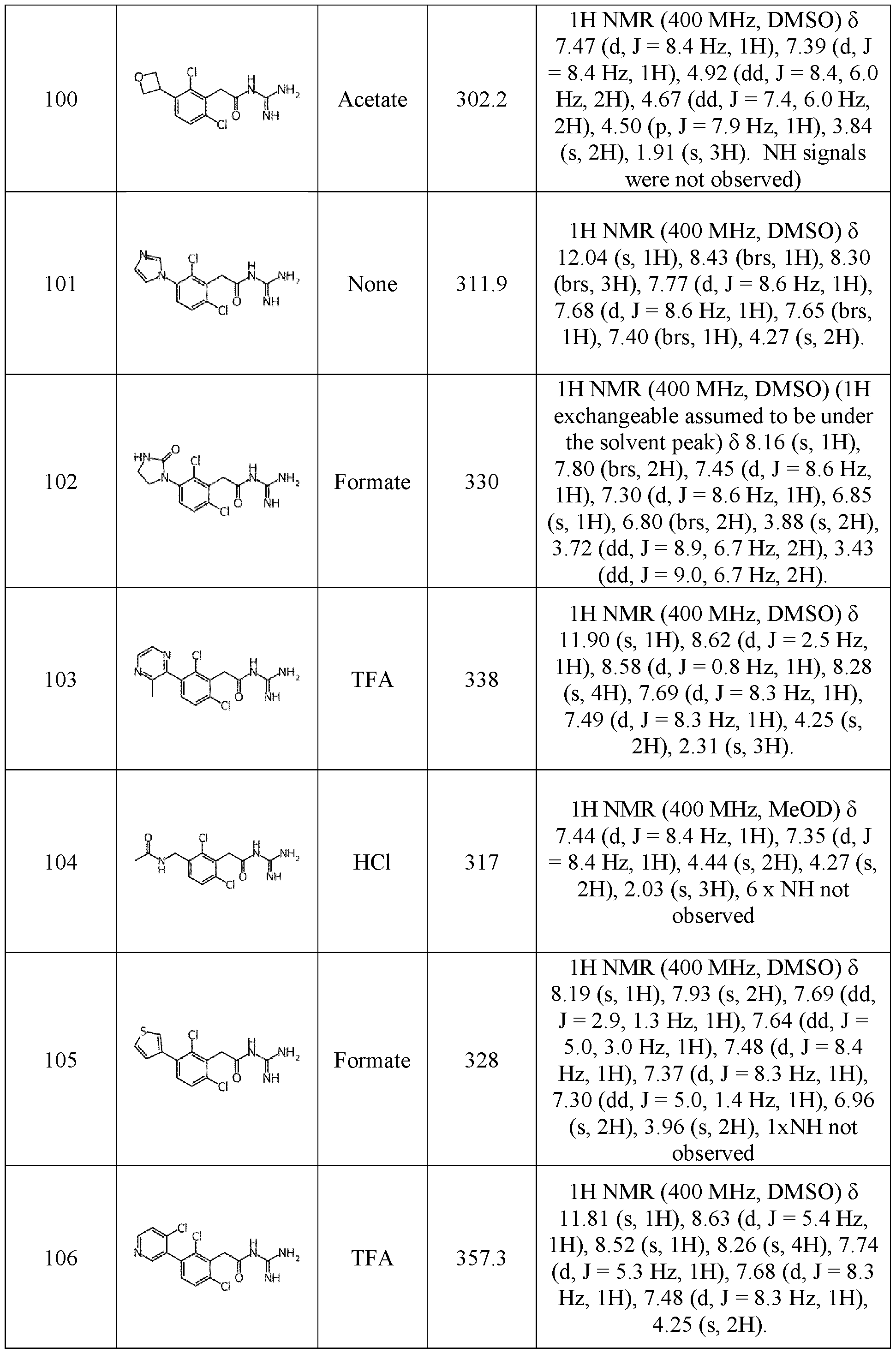
- ⁇ R 1 represents a radical selected in a group consisting of: • a hydrogen, • a (C 1 -C 6 )alkyl optionally substituted by a (C 1 -C 6 )alkyloxy, a hydroxy, at least one halogen, and a cycloalkyl, • a halogen, • a cyano, • a 3-14 membered ring, saturated, partially unsaturated, or unsaturated selected in a group consisting of a cycloalkyl, a heterocycloalkyl, an aryl, and an heteroaryl, said 3-14 membered ring is optionally substituted by a (C 1 -C 6 )alkyl, a hydroxy, a halogen, a -SO 2 -(C 1 -C 6 )alkyl, and a (C 1 -C 6 )alkyloxy, and • a (C 1 -C 6 )
- ⁇ R 1 represents a radical selected in a group consisting of: • a hydrogen, • a (C 1 -C 6 )alkyl optionally substituted by a (C 1 -C 6 )alkyloxy, a hydroxy, at least one halogen, and a cycloalkyl, • a halogen, • a 3-14 membered ring, saturated, partially unsaturated, or unsaturated selected in a group consisting of a cycloalkyl, a heterocycloalkyl, an aryl, and an heteroaryl, said 3-14 membered ring is optionally substituted by a (C 1 -C 6 )alkyl, a hydroxy, a halogen, a -SO 2 -(C 1 -C 6 )alkyl, and a (C 1 -C 6 )alkyloxy, and • a (C1-C6)alkyl
- the compound of formula (I) is such that n is 0. In a further particular embodiment, the compound of formula (I) is such that R4 and R5 represent H. In a further particular embodiment, the compound of formula (I) is such that R 6 , R 7 , and R 8 represent H.
- the compound of formula (I) is such that: ⁇ R 1 represents a (C 1 -C 6 )alkyl, preferably a methyl, a halogen, preferably a chlorine or a fluorine, more preferably a chlorine; and ⁇ R 2 represents a (C 1 -C 6 )alkyl, preferably a methyl, a halogen, preferably a chlorine or a fluorine, more preferably a chlorine, or a cyano.
- the compound of formula (I) is such that R 3 represents a 3- 14 membered ring, saturated, partially unsaturated, or unsaturated, selected in a group consisting of an aryl, a heteroaryl, a cycloalkyl, and a heterocycloalkyl, said 3-14 membered ring is optionally substituted by at least one radical A selected in a group consisting of: - a (C 1 -C 6 )alkyl optionally substituted by a halogen, a hydroxy, a cycloalkyl, a -CO-NH((C 1 -C 6 )alkyl), or a - CO-N((C 1 -C 6 )alkyl) 2 , - a heterocycloalkyl optionally substituted by a halogen, a (C 1 -C 6 )alkyloxy, or a hydroxy, - a cycloalkyl - a (C 1 -C 6 )
- R 3 represents a 3-14 membered ring, saturated, partially unsaturated, or unsaturated, selected in a group consisting of: o an aryl, particularly a phenyl, said aryl is optionally substituted by at least one radical selected in a group consisting of: - a (C 1 -C 6 )alkyl, particularly a methyl, - a (C 1 -C 6 )alkyloxy, particularly a methoxy or an ethoxy, - a cyano, - a hydroxy, - a halogen, particularly a chlorine, - a -NR d R e with R d and R e represent a hydrogen, and - a -NH-CO-(C 1 -C 6 )alkyl, particularly a -NH-CO-CH 3 , o a heteroaryl, particularly a pyrimidinyl, a pyrazinyl, a pyridaziny
- the compound of formula (I) is such that R 3 represents a radical selected in a group consisting of: o a (C 1 -C 6 )alkyl, particularly a methyl, an ethyl, an isopropyl, or an isobutyl, said (C 1 -C 6 )alkyl is optionally substituted by a (C 1 -C 6 )alkyloxy, particularly a 30 methoxy, a hydroxy, at least one halogen, or a -NH-CO-(C 1 -C 6 )alkyl, particularly a -NH-CO-CH 3 , o a (C 2 -C 6 )alkenyl, particularly an isopropenyl, o a (C 2 -C 6 )alkynyl optionally substituted by a heteroaryl, particularly a pyrazolyl, or a (C 1 -C 6 )alkyl optionally substituted by a radical selected in a group consist
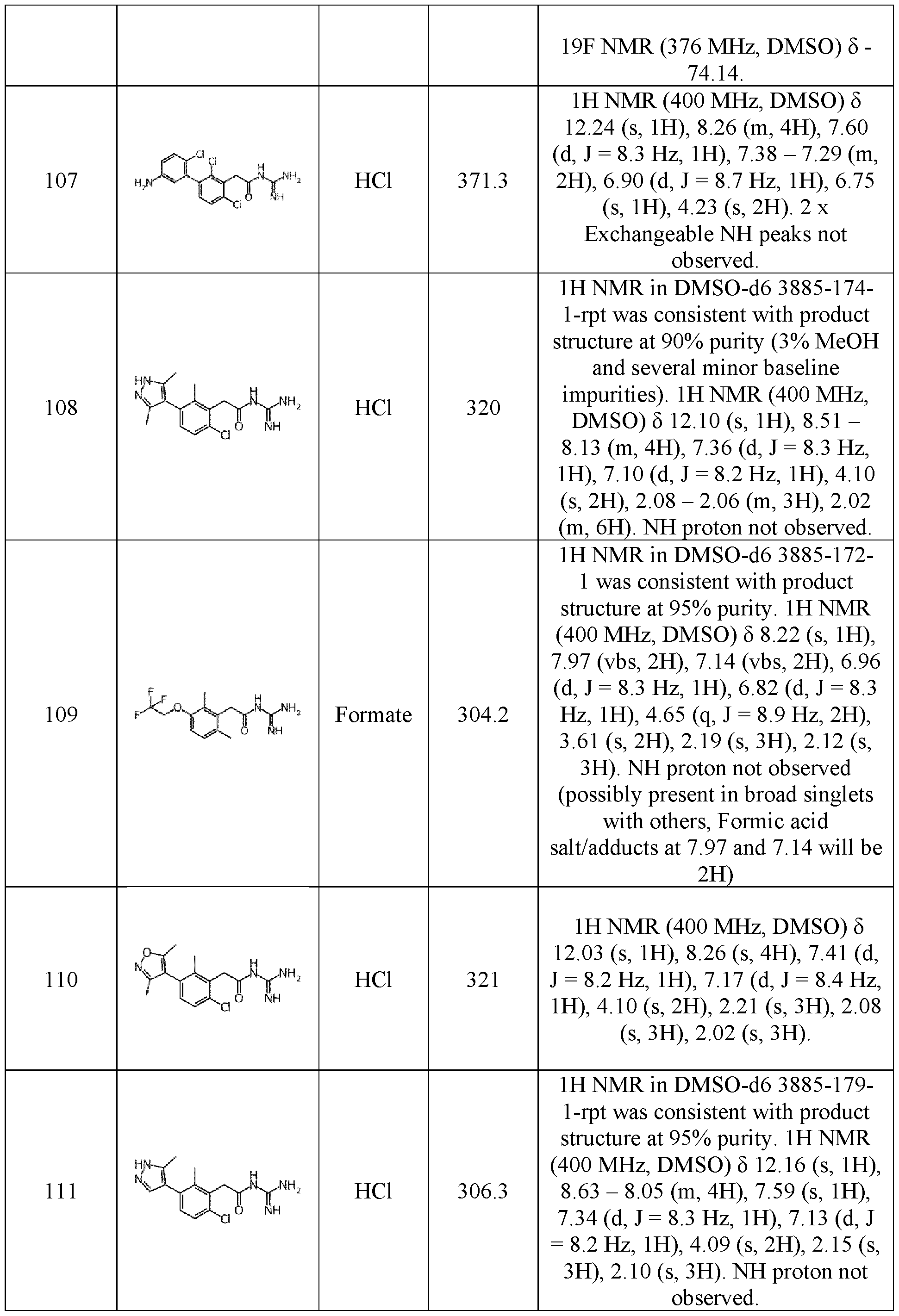
- a further object of the invention is a compound of formula (I) as defined herein for use as a medicine.
- a further object of the invention is a pharmaceutical composition comprising a compound of formula (I) as defined herein and a pharmaceutically acceptable excipient. 5
- a further object of the invention is a pharmaceutical composition as defined herein for use for treating a cancer.
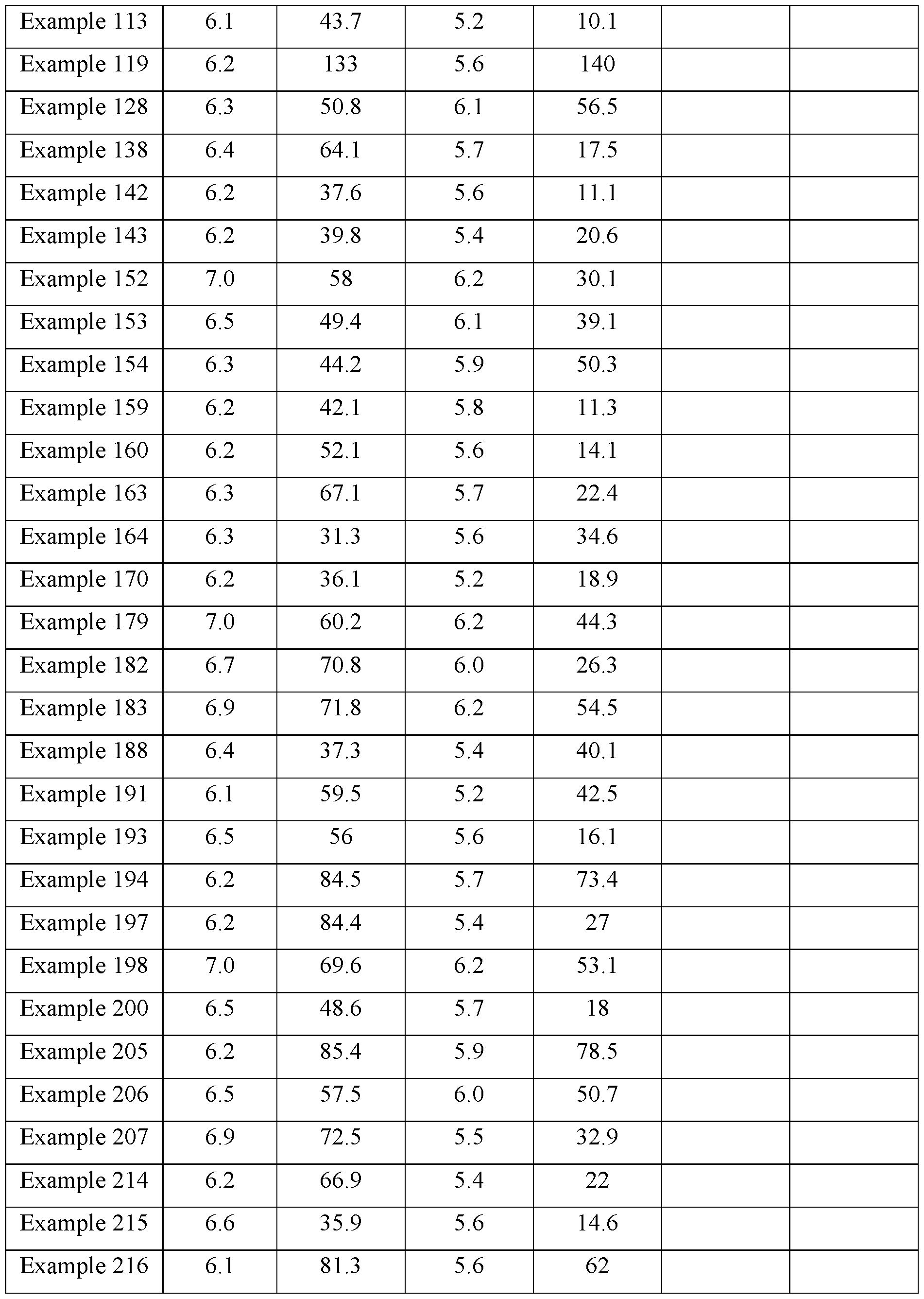
- the cancer is selected in a group consisting of myelofibrosis, acute lymphoblastic leukemia, acute myeloblastic leukemia adrenal gland carcinoma, bile duct cancer, bladder cancer, breast cancer, cervical cancer, colorectal cancer, endometrial cancer, esophageal cancer, gastric cancer, gastrointestinal stromal tumors, glioblastoma, head and neck 10 cancer, hepatocellular carcinoma, Hodgkin’s lymphoma, kidney cancer, lung cancer, melanoma, Merkel cell skin cancer, mesothelioma, multiple myeloma, myeloproliferative disorders, non-Hodgkin lymphoma, ovarian cancer, pancreatic cancer, prostate cancer, salivary gland cancer, sarcoma, squamous cell carcinoma, testicular cancer, thyroid cancer, urothelial carcinoma, and uveal melanoma.
- the pharmaceutical composition comprising a compound of formula (I) as defined herein is administered at a dose ranging from 0.001 mg/kg body weight to 30 mg/kg body weight.
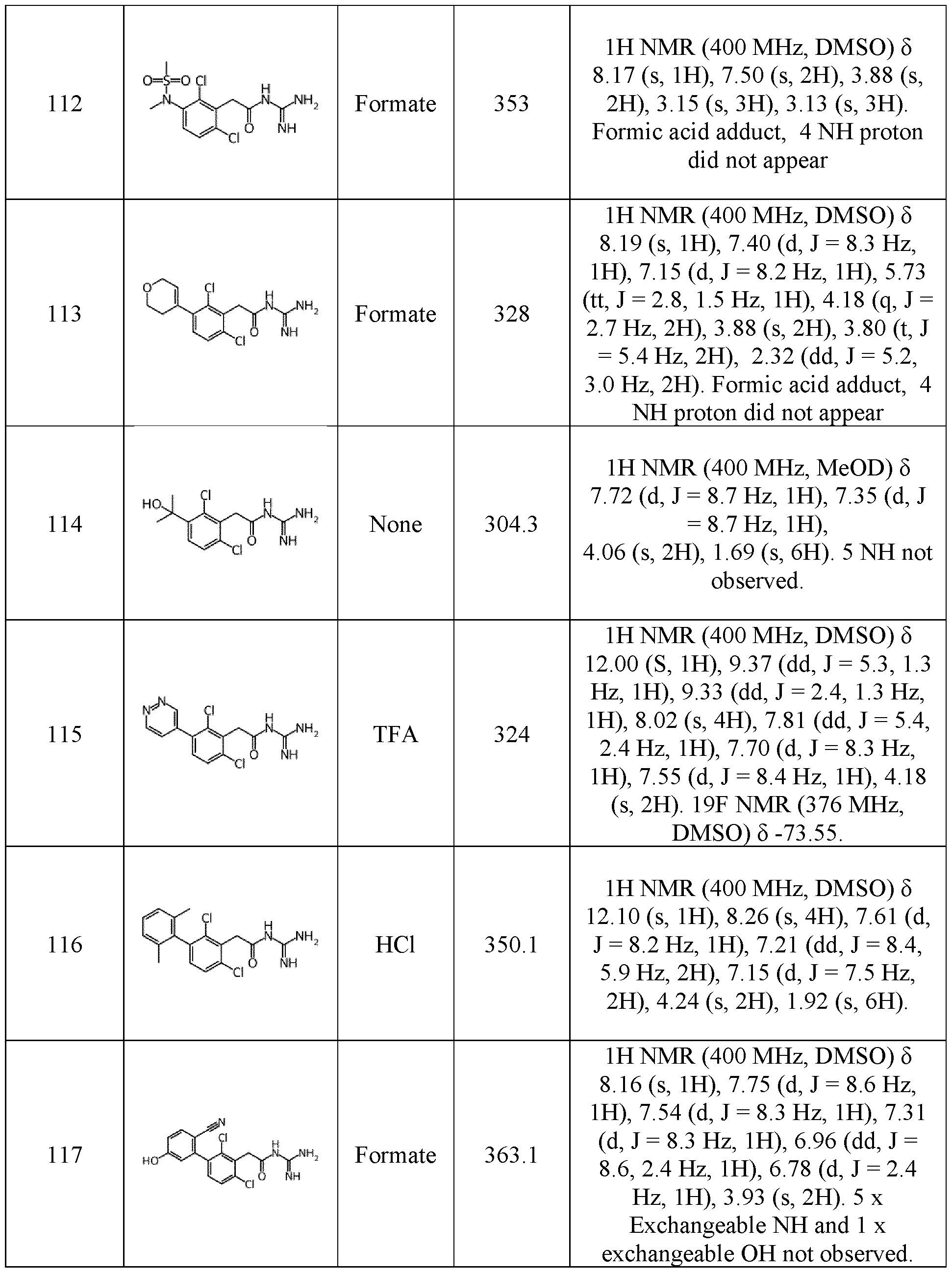
- the pharmaceutical composition is administered in combination with another antitumoral drug, especially chemotherapy, immunotherapy, hormonotherapy and/or radiotherapy, preferably immunotherapy.
- another antitumoral drug especially chemotherapy, immunotherapy, hormonotherapy and/or radiotherapy, preferably immunotherapy.
- the term C 1 -C 6 it means that the corresponding hydrocarbon chain may comprise from 1 to 6 carbon atoms, especially 1, 2, 3, 4, 5, or 6 carbon atoms. If, for example, the term C 1 -C 3 is used, it means that the corresponding hydrocarbon chain may comprise from 1 to 3 carbon atoms, especially 1, 2, or 3 carbon atoms.
- alkyl refers to a saturated, linear or branched aliphatic group.
- (C 1 - C 6 )alkyl more specifically means methyl, ethyl, propyl, isopropyl, butyl, isobutyl, pentyl, isopentyl, or hexyl.
- alkenyl refers to an unsaturated, linear or branched aliphatic group comprising at 5 least one carbon-carbon double bound.
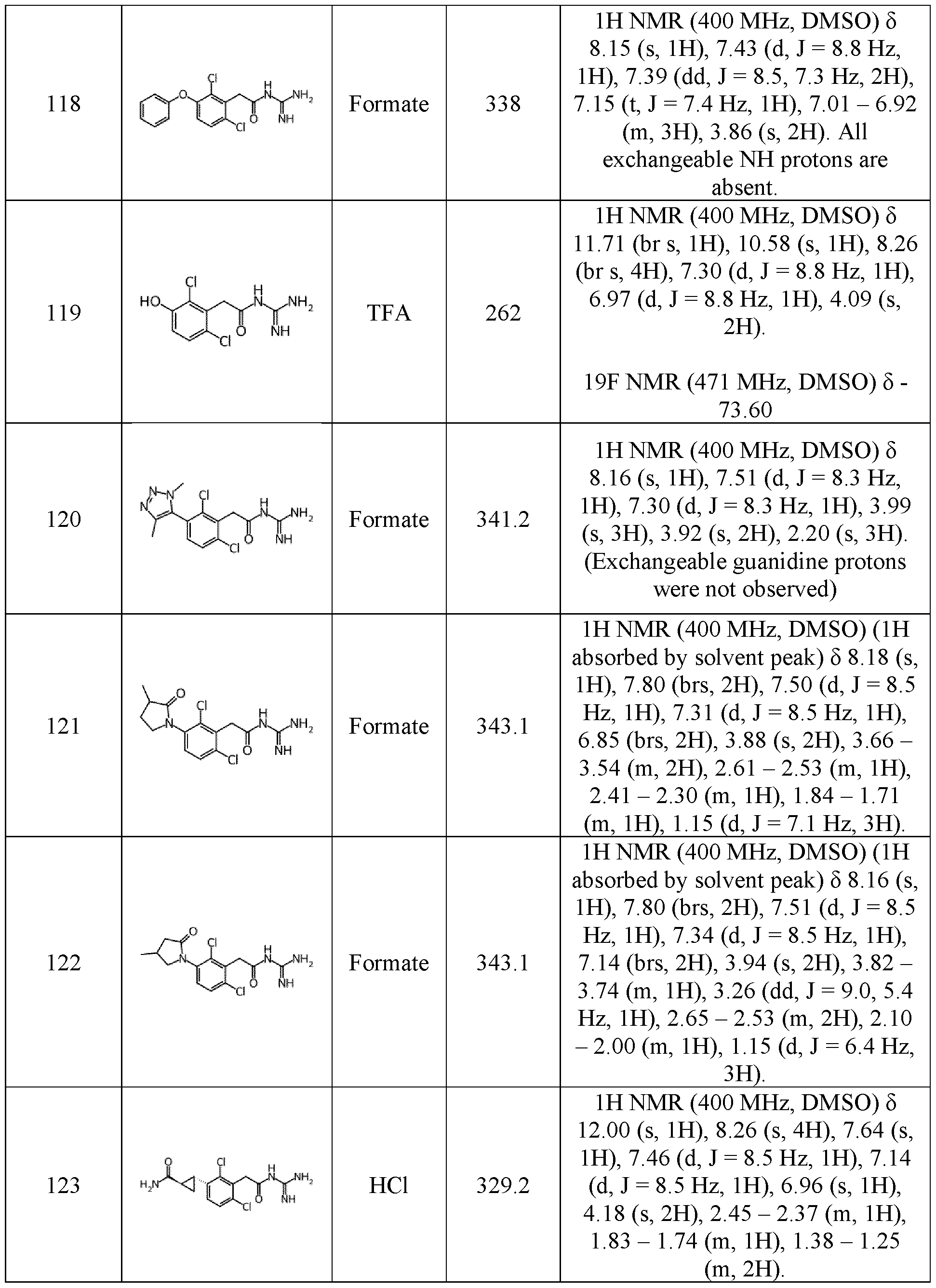
- (C2-C6)alkenyl more specifically means ethenyl, propenyl, isopropenyl, butenyl, isobutenyl, pentenyl, or hexenyl.
- alkynyl refers to an unsaturated, linear or branched aliphatic group comprising at least one carbon-carbon triple bound.
- (C 2 -C 6 )alkynyl more specifically means ethynyl, propynyl, isopropynyl, butynyl, isobutynyl, pentynyl, or hexynyl 10
- alkoxy or “alkyloxy” corresponds to the alkyl group as above defined bonded to the molecule by an -O- (ether) bond.
- (C 1 -C 6 )alkoxy or (C 1 -C 6 )alkyloxy includes methoxy or methyloxy, ethoxy or ethyloxy, propoxy or propyloxy, isopropoxy or isopropyloxy, butoxy or butyloxy, isobutoxy or isobutyloxy, pentoxy or pentyloxy, isopentoxy or isopentyloxy, and hexoxy or hexyloxy.
- the term “3-14 membered ring” corresponds to a ring having between 3 and 14 atoms.
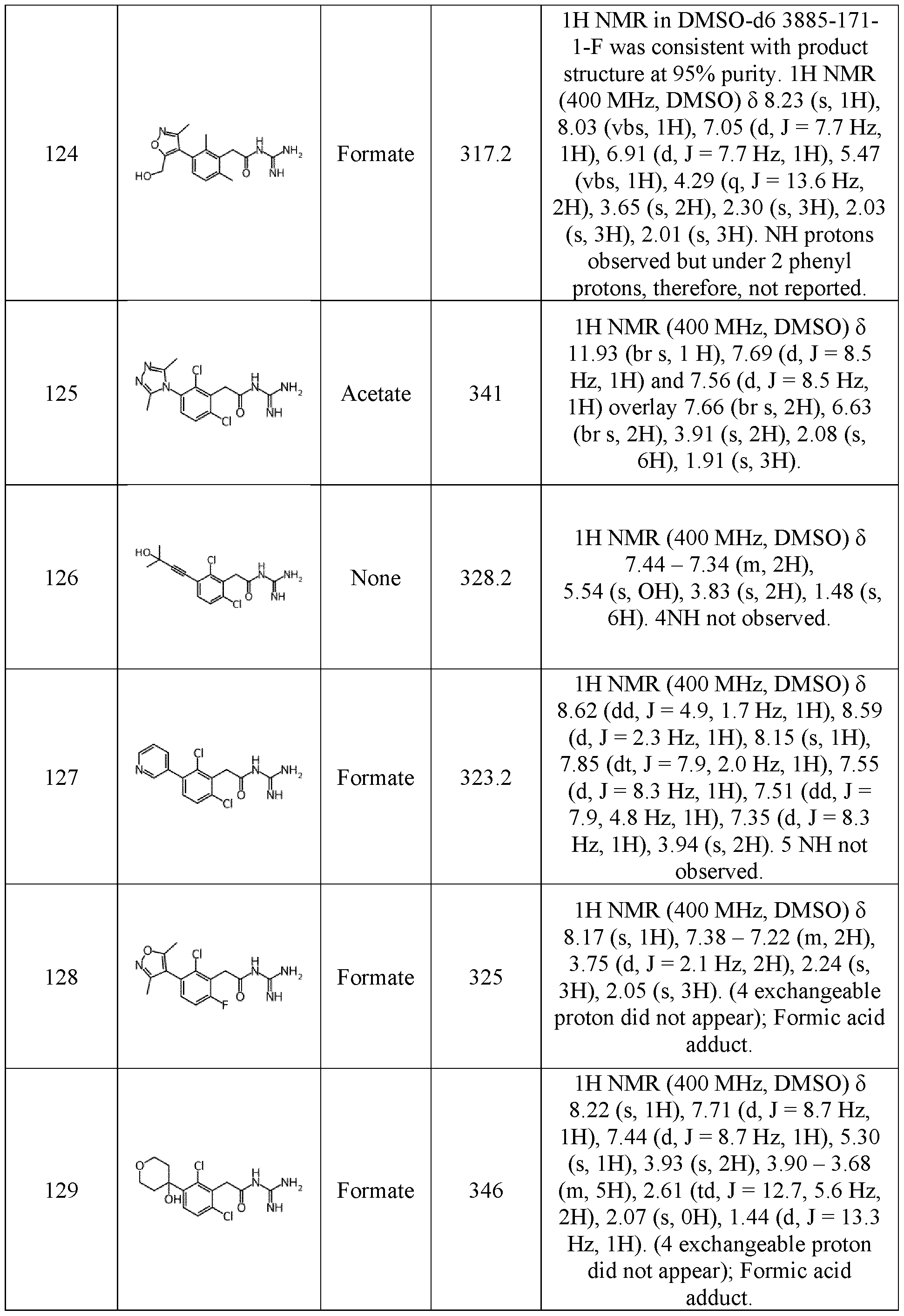
- Such a term includes, for instance, the term “5-14 membered ring” having between 5 and 14 atoms, and the term “5-7 membered ring” having between 5 and 7 atoms.
- the term “ring” corresponds to a mono-, bi, or tricycle, which can be saturated, partially unsaturated or unsaturated, and optionally comprises at least one heteroatom.
- the term “ring” includes a cycloalkyl, 20 a heterocycloalkyl, an aryl, and a heteroaryl.
- the term “cycloalkyl” corresponds to a saturated, partially unsaturated or unsaturated mono-, bi- or tri-cyclic alkyl group comprising between 3 and 14, preferably between 3 and 10 atoms of carbons.
- cycloalkyl includes for instance cyclopropyl, cyclobutyl, cyclopentyl, and cyclohexyl, 25 preferably cyclopropyl.
- heterocycloalkyl corresponds to a saturated, partially unsaturated or unsaturated cycloalkyl group as above defined further comprising at least one heteroatom such as nitrogen (N-heterocycloalkyl), oxygen (O-heterocycloalkyl), or sulphur atom (S-heterocycloalkyl). It also includes fused, bridged, or spiro-connected heterocycloalkyl groups.
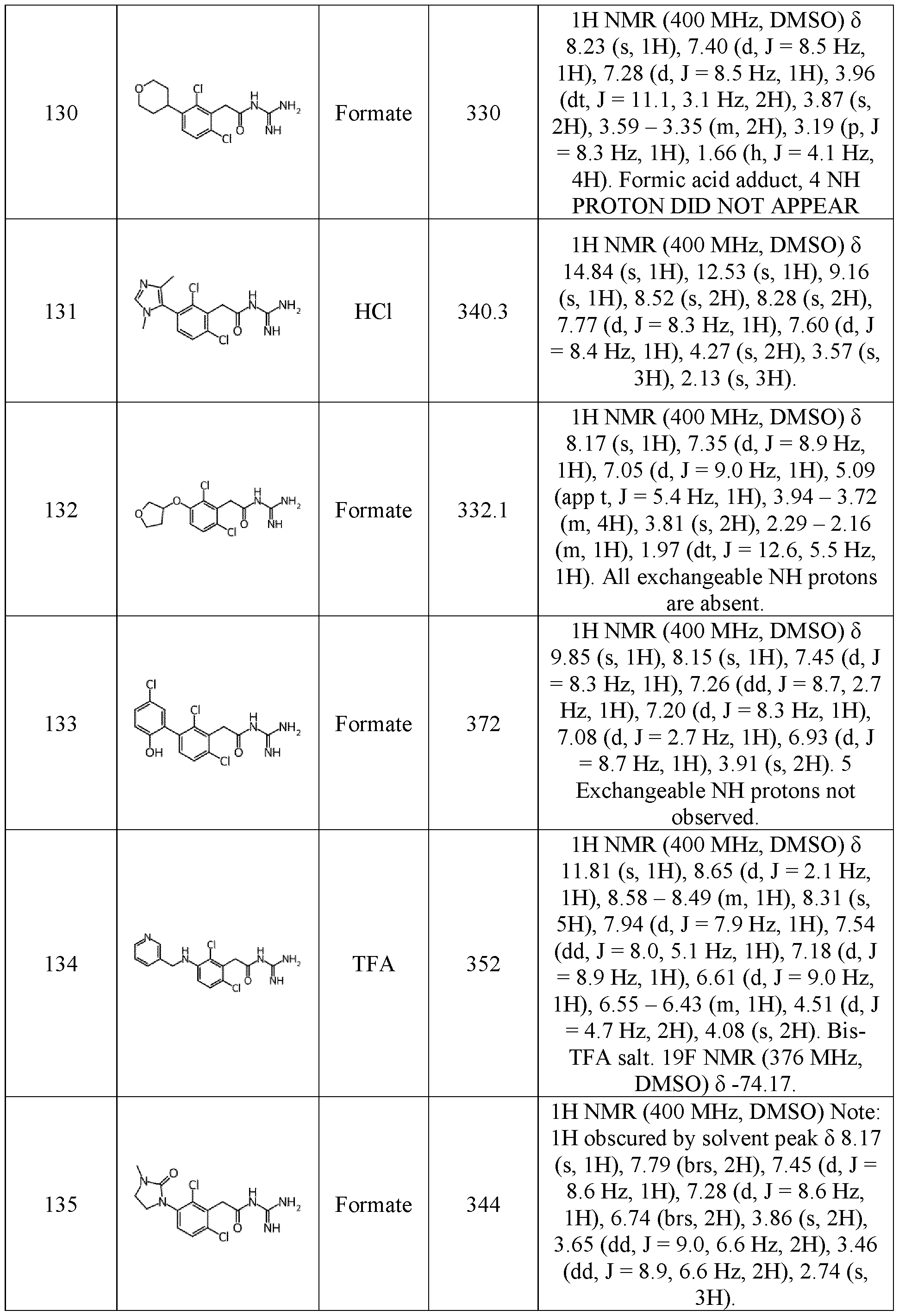
- heterocycloalkyl groups include, but are not limited to dioxolanyl, benzo [1,3] dioxolyl, azetidinyl, oxetanyl, thiomorpholinyl, pyrazolidinyl, piperidyl, piperazinyl, 1,4-dioxanyl, pyrrolinyl, pyrrolidinyl, piperidinyl, imidazolidinyl, morpholinyl, 1,4-dithianyl, pyrrolidinyl, oxozolinyl, oxazolidinyl, isoxazolinyl, isoxazolidinyl, thiazolinyl, thiazolidinyl, isothiazolinyl, isothiazolidinyl, tetrahydropyranyl, tetrahydrofuranyl, and tetrahydrothiophenyl.
- the heterocycloalkyl group is morpholinyl.
- “Cycloalkyl” and “heterocycloalkyl” also include cycloalkenyl and heterocycloalkenyl which correspond respectively to a partially unsaturated cycloalkyl and a partially unsaturated 5 heterocycloalkyl such as cyclohexenyl, imidazolinyl, dihydropyranyl, for instance 3,6-dihydro- 2H-pyranyl and 3,4-dihydro-2H-pyranyl, pyrazolinyl, azetinyl, pyranyl, and tetrahydropyridinyl, for instance 1,2,3-6-tetrahydropyridinyl.
- aryl corresponds to a mono- or bi-cyclic aromatic hydrocarbons having from 6 to 12 carbon atoms.
- the term “aryl” includes phenyl, biphenyl, naphthyl and 10 anthracenyl.
- the aryl is a phenyl.
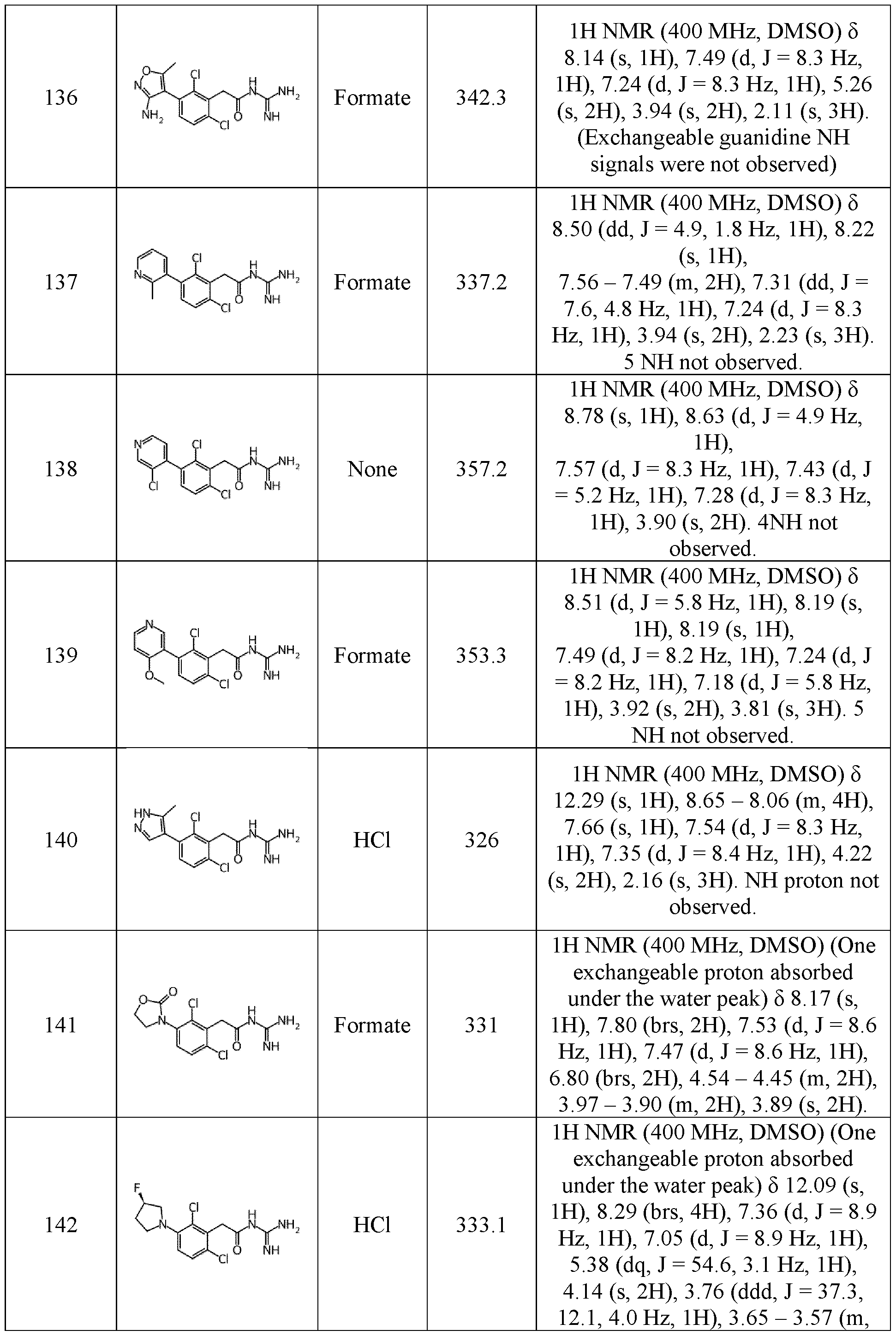
- heteroaryl as used herein corresponds to an aromatic, mono- or poly-cyclic group comprising between 3 and 20 atoms and comprising at least one heteroatom such as nitrogen, oxygen or sulphur atom.
- heteroaryl further includes the “fused arylheterocycloalkyl” and “fused heteroarylcycloalkyl”.
- fused 15 arylheterocycloalkyl and “fused heteroarylcycloalkyl” correspond to a bicyclic group in which an aryl as above defined or a heteroaryl is respectively bounded to the heterocycloalkyl or the cycloalkyl as above defined by at least two carbons. In other terms, the aryl or the heteroaryl shares a carbon bond with the heterocycloalkyl or the cycloalkyl.
- Examples of such mono- and poly-cyclic heteroaryl group, fused arylheterocycloalkyl and fused arylcycloalkyl may be: 20 pyridinyl, thiophenyl, furanyl, pyrrolyl, pyrazolyl, imidazolyl, oxazolyl, isoxazolyl, thiazolyl, isothiazolyl, triazolyl, tetrazolyl, oxadiazolyl, furazanyl, thiadiazolyl, tetrazolyl, benzofuranyl, thianaphthalenyl, indolyl, indolinyl, indanyl, quinolinyl, isoquinolinyl, benzimidazolyl, tetrahydroquinolinyl, tetrahydroisoquinolinyl, triazinyl, thianthrenyl, benzofuranyl, dihydrobenzofuranyl
- a fused arylheterocycloalkyl is for instance an indolinyl (phenyl fused to a pyrrolidinyl) and a dihydrobenzofuranyl (phenyl fused to a dihydrofuranyl).
- the heteroaryl group is a pyridinyl, a pyrazolyl, a pyrimidinyl, pyridazinyl, an indolyl, and an isoxazolyl.
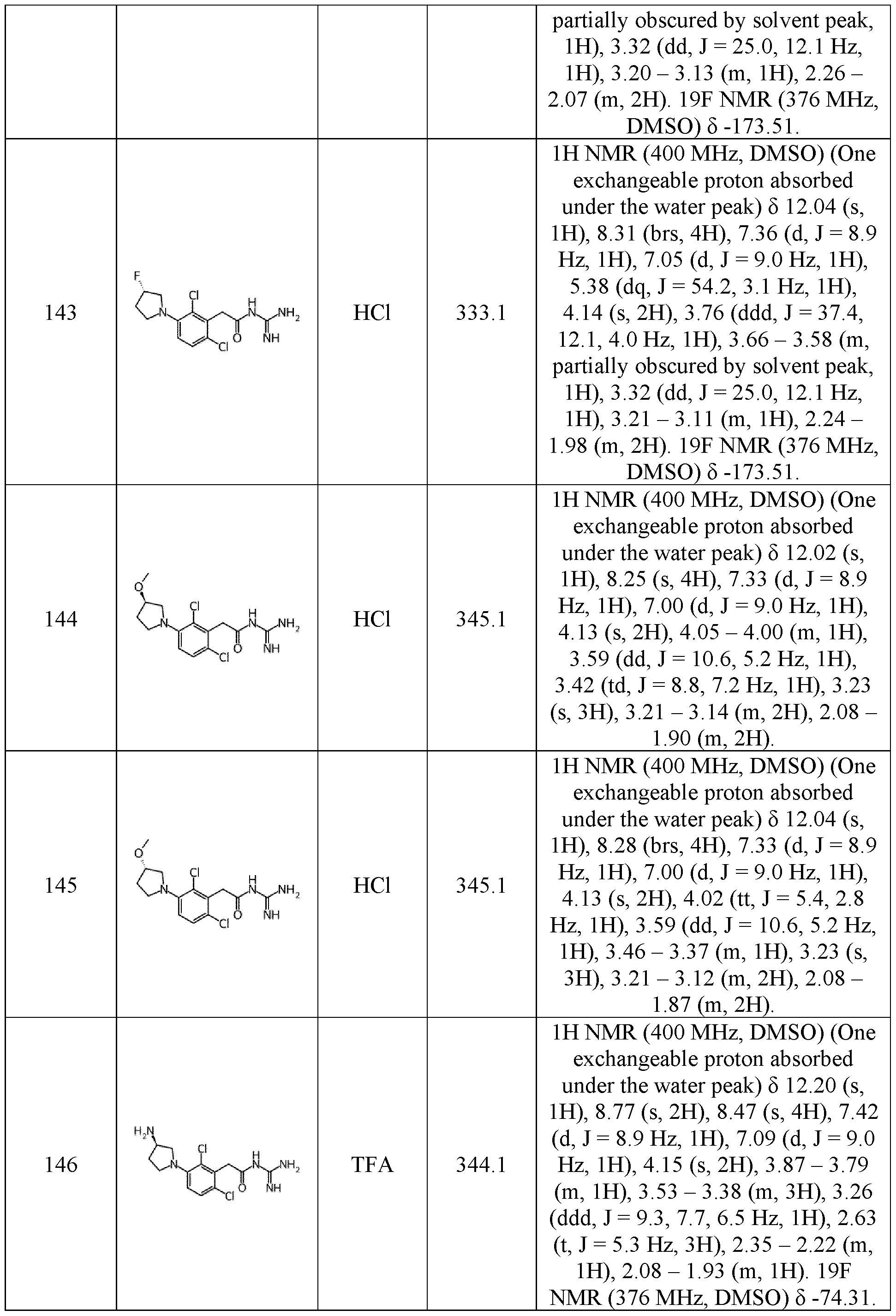
- halogen corresponds to a fluorine, chlorine, bromine, or iodine atom, preferably a fluorine, chlorine or bromine.
- the expression “substituted by at least” means that the radical is substituted by one or several groups of the list.
- a (C 1 -C 6 )alkyl substituted by at least one halogen, preferably a fluorine may include a fluoromethyl (-CH 2 F), a difluoromethyl (-CHF 2 ), or a trifluoromethyl (-CF 3 ).
- the expression “optionally substituted” means that the radical is not substituted or substituted 10 by one or several groups of the list.
- the “stereoisomers” are isomeric compounds that have the same molecular formula and sequence of bonded atoms, but differ in the 3D-dimensional orientations of their atoms in space.
- the stereoisomers include enantiomers, diastereoisomers, cis-trans and E-Z isomers, conformers, and anomers.
- the stereoisomers 15 include diastereoisomers and enantiomers.
- the “tautomers” are isomeric compounds that differ only in the position of the protons and the electrons.
- the “hydrates” are compounds further comprising at least one molecule of water. For instance, if the compound comprises one molecule of water, it corresponds to a monohydrate form. If the 20 compound comprises two molecules of water, it corresponds to a dihydrate form.
- the “pharmaceutically salts” include inorganic as well as organic acids salts.
- Suitable inorganic acids include hydrochloric, hydrobromic, hydroiodic, phosphoric, and the like.
- suitable organic acids include formic, acetic, trichloroacetic, trifluoroacetic, propionic, benzoic, cinnamic, citric, fumaric, maleic, 25 methanesulfonic and the like.
- pharmaceutically inorganic or organic acid addition salts include the pharmaceutically salts listed in J. Pharm. Sci. 1977, 66, 2, and in Handbook of Pharmaceutical Salts: Properties, Selection, and Use edited by P. Heinrich Stahl and Camille G. Wermuth 2002.
- the salt is selected from the group consisting of maleate, chlorhydrate, bromhydrate, and methanesulfonate.
- the 30 “pharmaceutically salts” also include inorganic as well as organic base salts.
- suitable inorganic bases include sodium or potassium salt, an alkaline earth metal salt, such as a calcium or magnesium salt, or an ammonium salt.
- treatment”, “treat” or “treating” refer to any act intended to ameliorate the health status of patients such as therapy, prevention, prophylaxis and retardation of a disease, in particular a cancer. In certain embodiments, such terms refer to the amelioration or eradication of the disease, or symptoms associated with it.
- this term refers to minimizing the spread or worsening of the disease, resulting from the administration of one or more therapeutic agents to a subject with such a disease.
- such terms 5 refer to decreased development of tumors, decreased tumor burden, tumor regression, and/or prevention or delay of metastasis occurrence and cancer relapse.
- the terms “subject”, “individual” or “patient” are interchangeable and refer to a mammal, even more preferably to a human, including adult, child, newborn and human at the prenatal stage.
- the term “subject” can also refer to non-human animals, in particular 10 mammals such as dogs, cats, horses, cows, pigs, sheep and non-human primates, among others.
- the subject is resistant to any other anticancer treatment.
- the subject is a chemoresistant subject.
- the subject is resistant to immunotherapy.
- the cancer is immune-resistant.
- Quantity,” “amount,” and “dose” are used interchangeably herein and may refer to 15 an absolute quantification of a molecule.
- active principle As used herein, the terms “active principle”, “active ingredient”, “active pharmaceutical ingredient”, “medicine”, and “drug” are equivalent and refers to a component of a pharmaceutical composition having a therapeutic effect.
- the term “therapeutic effect” refers to an effect induced by an active ingredient, 20 or a pharmaceutical composition according to the invention, capable to prevent or to delay the appearance or development of a cancer, or to cure or to attenuate the effects of a cancer.
- the term “effective amount” refers to a quantity of an active ingredient or of a pharmaceutical composition which prevents, removes or reduces the deleterious effects of a cancer. It is obvious that the quantity to be administered can be adapted by the man skilled in 25 the art according to the subject to be treated, to the nature of the cancer, etc.
- doses and regimen of administration may be function of the nature, of the stage and of the severity of the cancer to be treated, as well as of the weight, the age and the global health of the subject to be treated, as well as of the judgment of the doctor.
- pharmaceutically acceptable excipient refers to any ingredient except 30 active ingredients which are present in a pharmaceutical composition. Its addition may be aimed to confer a particular consistency or other physical or gustative properties to the final product. A pharmaceutically acceptable excipient must be devoid of any interaction, in particular chemical, with the active ingredients.
- the term “immunotherapy”, “immunotherapeutic agent” or “immunotherapy treatment” refers to a cancer therapeutic treatment using the immune system to reject cancer.
- the therapeutic treatment stimulates the patient's immune system to attack the malignant tumor cells. It includes immunization of the patient with tumor antigens (e.g., by administering a 5 cancer vaccine), in which case the patient's own immune system is trained to recognize tumor cells as targets to be destroyed, or administration of molecules stimulating the immune system such as cytokines, or administration of therapeutic antibodies as drugs, in which case the patient's immune system is recruited by the therapeutic antibodies to destroy tumor cells.
- tumor antigens e.g., by administering a 5 cancer vaccine
- the patient's own immune system is trained to recognize tumor cells as targets to be destroyed
- administration of molecules stimulating the immune system such as cytokines
- therapeutic antibodies as drugs
- antibodies are directed against specific antigens such as the unusual antigens that are 10 presented on the surfaces of tumors.
- kit defines especially a "kit of parts” in the sense that the combination partners (a) and (b), as defined in the present application can be dosed independently or by use of different fixed combinations with distinguished amounts of the combination partners (a) and (b), i.e., simultaneously or at 15 different time points.
- the parts of the kit of parts can then be administered simultaneously or chronologically staggered, that is at different time points for any part of the kit of parts.
- the ratio of the total amounts of the combination partner (a) to the combination partner (b) to be administered in the combined preparation can be varied.
- the combination partners (a) and (b) can be administered by the same route or by different routes.
- the term “simultaneous” refers to a pharmaceutical composition, a kit, a product or a combined preparation according to the invention in which the active ingredients are used or administered simultaneously, i.e., at the same time.
- the term “sequential” refers to a pharmaceutical composition, a kit, a product or a combined preparation according to the invention in which the active ingredients are used 25 or administered sequentially, i.e., one after the other.
- the administration is sequential, all the active ingredients are administered in less than about an hour, preferably less than about 10 minutes, even more preferably in less than about a minute.
- the term “separate” refers to a pharmaceutical composition, a kit, a product or a combined preparation according to the invention in which the active ingredients are used or 30 administered at distinct time of the day.
- the active ingredients are administered with an interval of about 1 hour to about 24 hours, preferably with an interval of about 1 hour and 15 hours, more preferably with an interval of about 1 hour and 8 hours, even more preferably with an interval of about 1 hour and 4 hours.
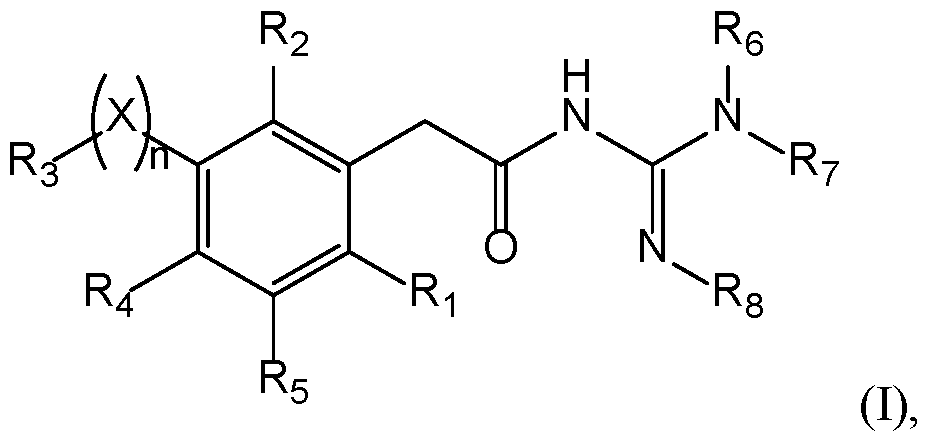
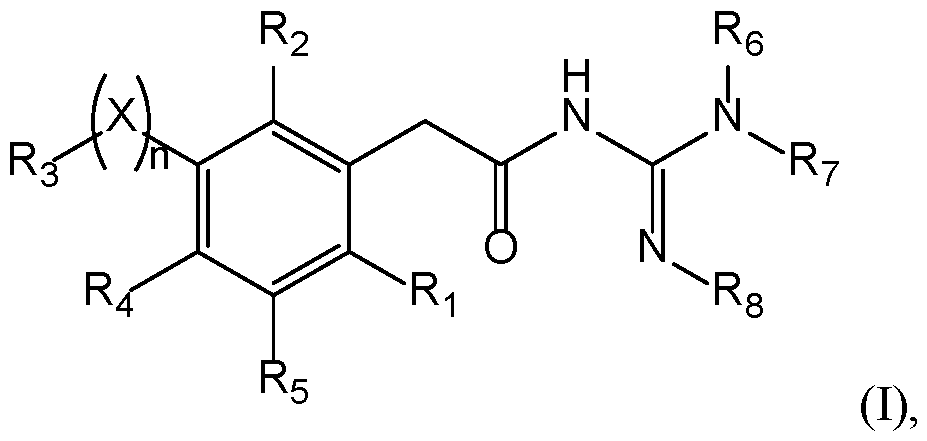
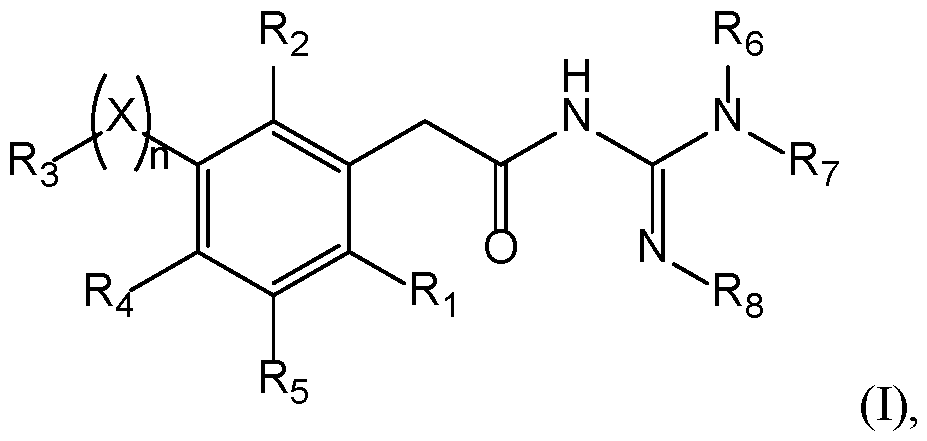
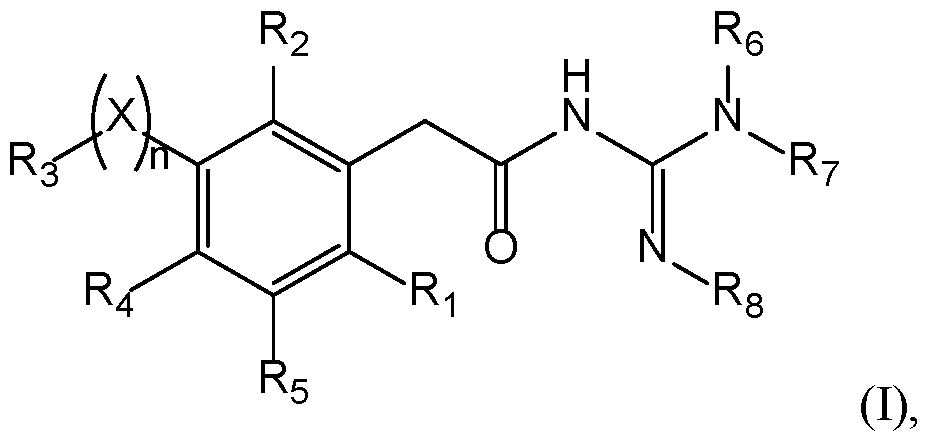
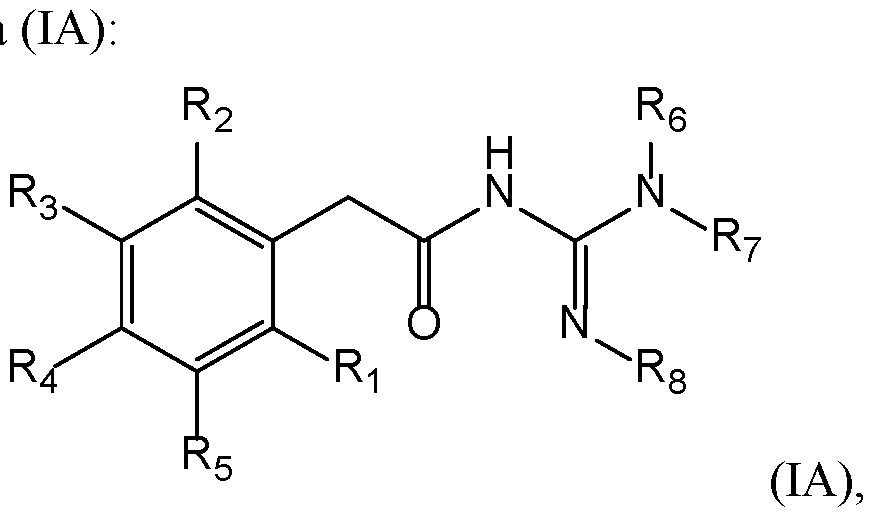
- Compounds The present invention provides new compounds of the following formula (I), including isomers and pharmaceutical acceptable salts thereof, of therapeutic interest.
- a compound has the following formula (I): wherein: ⁇ R 1 represents a radical selected in a group consisting of: 10 • a hydrogen, • a (C 1 -C 6 )alkyl optionally substituted by a (C 1 -C 6 )alkyloxy, a hydroxy, at least one halogen, and a cycloalkyl, • a halogen, • a cyano, 15 • a 3-14 membered ring, saturated, partially unsaturated, or unsaturated selected in a group consisting of a cycloalkyl, a heterocycloalkyl, an aryl, and an heteroaryl, said 3-14 membered ring is optionally substituted by a (C 1 -C 6 )alkyl, a hydroxy, a halogen, a -SO 2 -(C 1 -C 6 )alkyl, and a (C 1 -C 6 )alkyloxy, and •
- a compound has the following formula (I): 20 wherein: ⁇ R 1 represents a radical selected in a group consisting of: • a hydrogen, • a (C 1 -C 6 )alkyl optionally substituted by a (C 1 -C 6 )alkyloxy, a hydroxy, at least 25 one halogen, and a cycloalkyl, • a halogen, • a 3-14 membered ring, saturated, partially unsaturated, or unsaturated selected in a group consisting of a cycloalkyl, a heterocycloalkyl, an aryl, and an heteroaryl, said 3-14 membered ring is optionally substituted by a (C 1 -C 6 )alkyl, a hydroxy, a halogen, a -SO 2 -(C 1 -C 6 )alkyl, and a (C 1 -C 6 )alkyloxy, and • a (C 1 -C 6 )
- n is 0 or 1. If n is 1, X is present. If n is 0, X is absent. In a particular embodiment, n is 1. According to this embodiment, X is selected from the group consisting of -(CH 2 ) m - with m is from 1 to 3 optionally substituted by at least one R a , -O-, -S-, -CO-, -C(O)-O-, -O-C(O)-, -NR a -, -CO-NR a -, -NR a -CO-, -SO-, -SO 2 -, -SO 2 -NR a , -NR a -SO 2 -, 25 and -NR a -CO-NR a -, with R a being a hydrogen, a (C 1 -C 6 )alkyl optionally substituted by at least one radical C, a cycloalkyl optionally substituted by at least one
- X is selected from the group consisting of -(CH2)m- with m is from 1 to 3, particularly 1, -O-, -S-, -CO-, -C(O)-O-, -O-C(O)-, -SO-, and -SO 2 -.
- X is selected from the group consisting of -(CH 2 ) m - with m is from 1 to 3 substituted by at least one R a , -NR a -, -CO-NR a -, -NR a -CO-, -SO 2 -NR a , - NR a -SO 2 -, and -NR a -CO-NR a -, with R a being a hydrogen, a (C 1 -C 6 )alkyl optionally substituted 20 by at least one radical C, a cycloalkyl optionally substituted by at least one radical C, or a heterocycloalkyl optionally substituted by at least one radical C; said radical C being selected in a group consisting of: - a (C 1 -C 6 )alkyloxy, - a halogen, 25 - a -O-C(O)-(C 1 -C 6 )alkyl,
- R a is a hydrogen, a (C 1 -C 6 )alkyl, a cycloalkyl, or a heterocycloalkyl.
- R a is a (C 1 -C 6 )alkyl substituted by at least one radical C, a 10 cycloalkyl substituted by at least one radical C, or a heterocycloalkyl substituted by at least one radical C; said radical C being selected in a group as defined herein.
- R a is a hydrogen.
- X represents -NH-, -CO-NH-, -NH-CO-, -SO 2 -NH-, -NH-SO 2 -, or -NH-CO-NH-.
- n is 0.
- a compound of the invention 15 has the following formula with R 1 , R 2 , R 3 , R 4 , R 5 , R 6 , R 7 , and R 8 are as defined herein.
- R 4 and R 5 represent independently a radical selected in a group consisting of: 20 • a hydrogen, • a halogen, • a (C 1 -C 6 )alkyl optionally substituted by a halogen, a (C 1 -C 6 )alkyloxy, a hydroxy, or a cycloalkyl, • a (C 1 -C 6 )alkyloxy optionally substituted by a halogen, a (C 1 -C 6 )alkyloxy, a 25 hydroxy, or a cycloalkyl, • a hydroxy, • a -NR k R l with R k and R l represent independently a hydrogen, a (C 1 -C 6 )alkyl, a cycloalkyl, or R k and R l may form a N-heterocycloalkyl, • a nitro, • a cyano, • a -NH-(C 1 )
- R 4 and R 5 represent H.
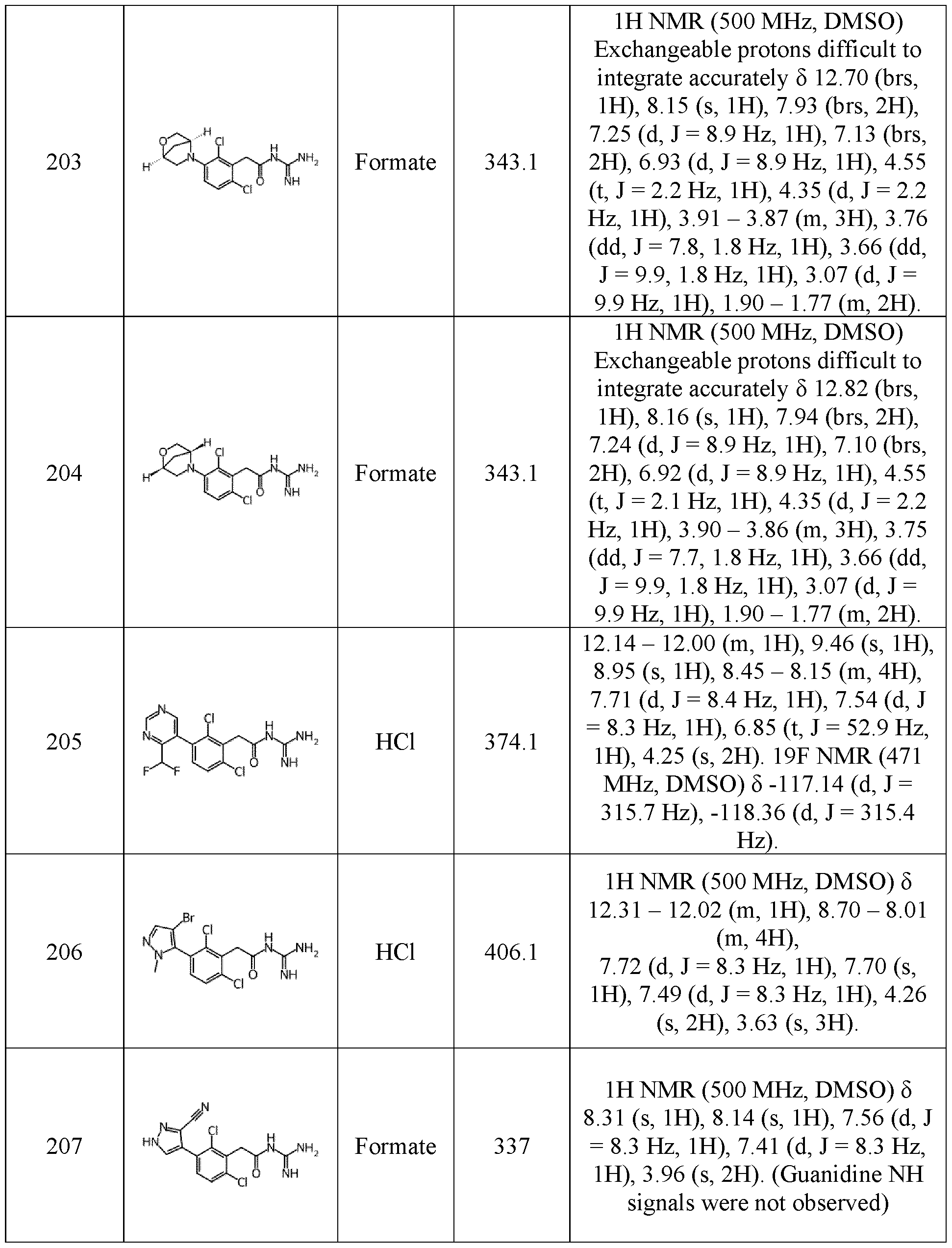
- a compound 5 of the invention has the following formula (IB): with X, n, R 1 , R 2 , R 3 , R 6 , R 7 , and R 8 are as defined herein.
- R 6 represents a hydrogen, or a (C 1 -C 6 )alkyl optionally substituted by a radical selected in a group consisting of a hydroxy, a (C 1 -C 6 )alkyloxy, a -NH(C 1 -C 6 )alkyl, 10 and a -N((C 1 -C 6 )alkyl) 2 .
- R 6 is a hydrogen.
- R 7 and R 8 represent independently a hydrogen, or a (C 1 -C 6 )alkyl optionally substituted by a radical selected in a group consisting of a hydroxy, a (C 1 - C 6 )alkyloxy, a -NH(C 1 -C 6 )alkyl, and a -N((C 1 -C 6 )alkyl) 2 , or R 7 and R 8 may form together a 5- 7 membered ring, partially unsaturated or saturated, N,N’-heterocycloalkyl optionally 15 substituted by at least one radical selected in a group consisting of a (C 1 -C 6 )alkyl, a hydroxy, a (C1-C6)alkyloxy, a -NH(C1-C6)alkyl, a -N((C1-C6)alkyl)2, and a ketone.
- R7 and/or R 8 are a hydrogen.
- R 7 and R 8 are a hydrogen.
- R 7 and R 8 form together an imidazolinyl optionally substituted by20 at least one radical selected in a group consisting of a (C 1 -C 6 )alkyl, a hydroxy, a (C 1 - C 6 )alkyloxy, a -NH(C 1 -C 6 )alkyl, a -N((C 1 -C 6 )alkyl) 2 , and a ketone.
- R 7 and R 8 form together an imidazolinyl substituted by a methyl and a ketone.
- R 6 , R 7 , and R 8 represent H.
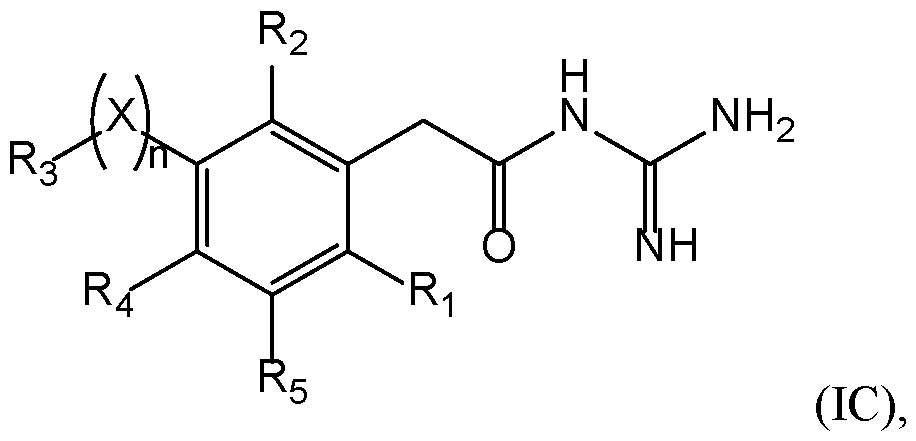
- a compound of the invention has the following formula (IC): 25 with X, n, R 1 , R 2 , R 3 , R 4 , and R 5 are as defined herein.
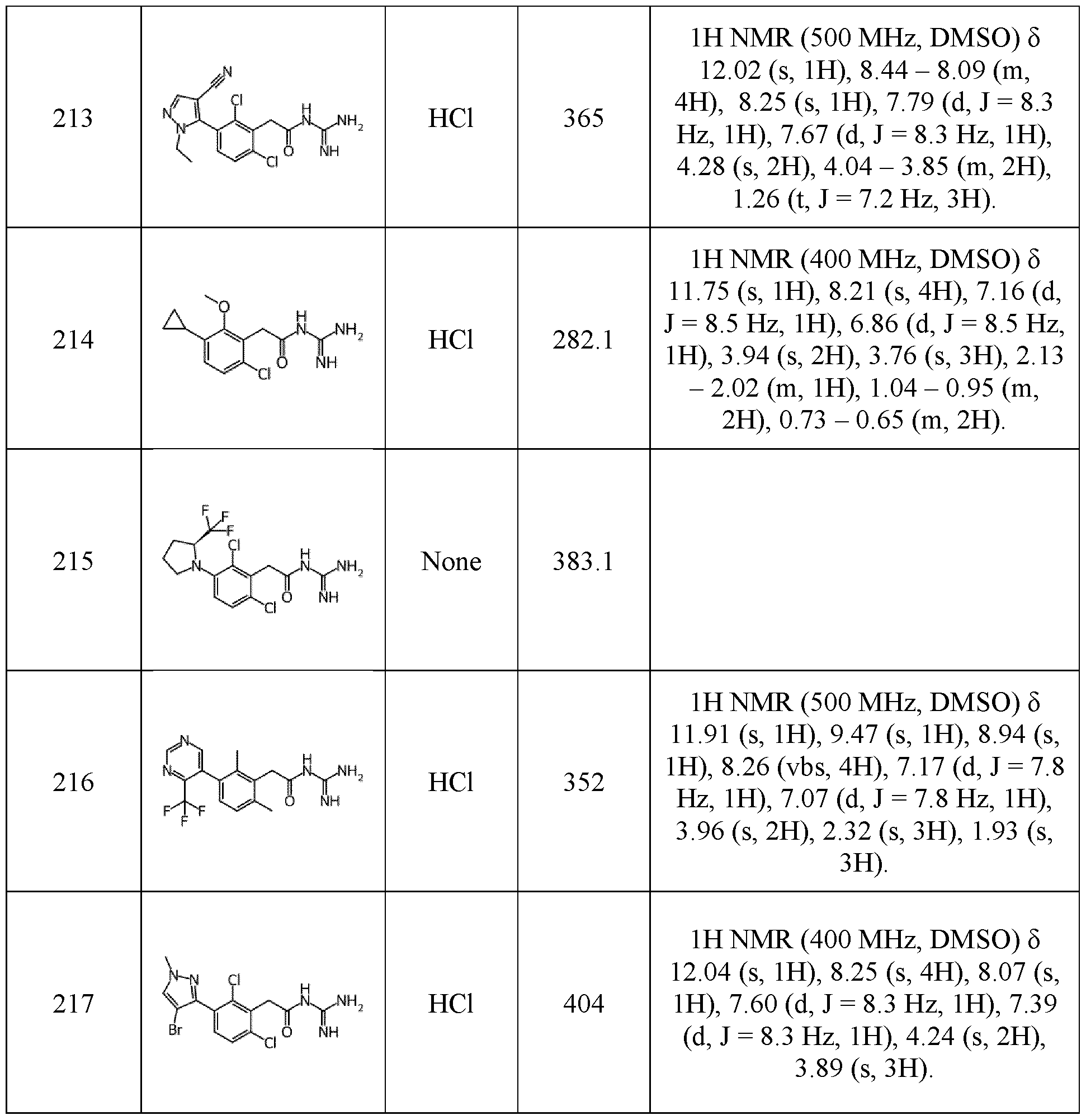
- R 1 and R 2 represent independently a radical selected in a group consisting of: • a hydrogen, • a (C 1 -C 6 )alkyl optionally substituted by a (C 1 -C 6 )alkyloxy, a hydroxy, at least 5 one halogen, and a cycloalkyl, • a halogen, • a cyano, • a 3-14 membered ring, saturated, partially unsaturated, or unsaturated selected in a group consisting of a cycloalkyl, a heterocycloalkyl, an aryl, and an 10 heteroaryl, said 3-14 membered ring is optionally substituted by a (C 1 -C 6 )alkyl, a hydroxy, a halogen, a -SO 2 -(C 1 -C 6 )alkyl, and a (C 1 -C 6 )alkyloxy, and • a (C 1 -C 6 )alkyl
- R 1 represents a hydrogen, a (C 1 -C 6 )alkyl, a halogen, or a (C 1 - C 6 )alkyloxy.
- R 1 represents a (C 1 -C 6 )alkyl or a halogen.
- R 1 is selected 15 from the group consisting of methyl, bromine, chlorine or fluorine, preferably methyl, chlorine or fluorine.
- R 1 represents a cyclopropyl, an optionally substituted phenyl (e.g., phenyl substituted by -(SO 2 )-(C 1 -C 6 )alkyl, (C 1 -C 6 )alkyloxy, or halogen, in particular -(SO 2 )-methyl, methoxy or chlorine), or an optionally substituted pyrazolyl or pyrimidinyl.
- phenyl e.g., phenyl substituted by -(SO 2 )-(C 1 -C 6 )alkyl, (C 1 -C 6 )alkyloxy, or halogen, in particular -(SO 2 )-methyl, methoxy or chlorine
- pyrazolyl or pyrimidinyl e.g., phenyl substituted by -(SO 2 )-(C 1 -C 6 )alkyl, (C 1 -C 6 )alkyloxy, or halogen,
- R 2 represent a hydrogen, a (C 1 -C 6 )alkyl optionally substituted by a (C 1 -C 6 )alkyloxy, a hydroxy, at least one halogen, a halogen, a cyano, a 3-14 membered ring, saturated, partially unsaturated, or unsaturated selected in a group consisting of a cycloalkyl, a heterocycloalkyl, an aryl, and an heteroaryl, said 3-14 membered ring is optionally substituted by a (C 1 -C 6 )alkyl, a hydroxy, a halogen, a -SO 2 -(C 1 -C 6 )alkyl, and a (C 1 - 25 C 6 )alkyloxy, or a (C 1 -C 6 )alkyloxy optionally substituted by at least one halogen.
- R 2 represents a (C 1 -C 6 )alkyl, a halogen, a cyano, a cyclopropyl, an optionally substituted phenyl, or an optionally substituted pyrazolyl or pyrimidinyl.
- R 2 is selected from the group consisting of methyl, bromine, chlorine, fluorine, a phenyl substituted by -(SO 2 )-(C 1 -C 6 )alkyl, (C 1 -C 6 )alkyloxy or halogen, in particular by -(SO 2 )-methyl, methoxy 30 or chlorine, or an optionally substituted pyrazolyl or pyrimidinyl.
- At least one among R1 and R2 comprises a halogen, for instance represents a halogen or a radical substituted by a halogen.
- both R 1 and R 2 comprise a halogen, for instance represent a halogen or a radical substituted by a halogen.
- the halogen is chlorine or fluorine, especially chlorine.
- R 1 and R 2 are identical.
- a compound of formula (I), (IA), (IB), or (IC) is such that: ⁇ R 1 represents a (C 1 -C 6 )alkyl, preferably a methyl, a halogen, preferably a chlorine or a fluorine, more preferably a chlorine; and ⁇ R 2 represents a (C 1 -C 6 )alkyl, preferably a methyl, a halogen, preferably a chlorine or a fluorine, more preferably a chlorine, or a cyano.
- R 3 represents a 3-14 membered ring, saturated, partially unsaturated, or unsaturated, optionally substituted by at least one radical A as defined herein, or a radical selected in a group consisting of a (C 1 -C 6 )alkyl, a cycloalkyl, a (C 2 -C 6 )alkenyl, a (C 2 -C 6 )alkynyl, or a (C 1 -C 6 )alkyloxy, optionally substituted by at least one radical B as defined 15 herein, a hydroxy, a cyano, a halogen, and a -NRf’Rg’ with Rf’ and Rg’, represent independently a hydrogen, a (C 1 -C 6 )alkyl optionally substituted by at least one radical B, a cycloalkyl optionally substituted by at least one radical B, a heterocycloalkyl optionally substituted by at least one radical B, with
- R 3 represents a 3-14 membered ring, saturated, partially unsaturated, or unsaturated, selected in a group consisting of an aryl, a heteroaryl, a cycloalkyl, and a heterocycloalkyl, said 3-14 membered ring is optionally substituted by at least one radical A selected in a group consisting of: - a (C 1 -C 6 )alkyl optionally substituted by a halogen, a 30 (C 1 -C 6 )alkyloxy, a hydroxy, or a -CO-NH 2 , - a cycloalkyl optionally substituted by a halogen, a (C 1 - C 6 )alkyloxy, a hydroxy, or a -CO-NH 2 , - a heterocycloalkyl optionally substituted by a halogen, a (C 1 -C 6 )alkyloxy, or a hydroxy,
- said at least one radical A is selected in a group consisting of: - a (C 1 -C 6 )alkyl optionally substituted by a halogen, a hydroxy, a cycloalkyl, a -CO-NH((C 1 -C 6 )alkyl), or a - 25 CO-N((C 1 -C 6 )alkyl) 2 , - a heterocycloalkyl optionally substituted by a halogen, a (C 1 -C 6 )alkyloxy, or a hydroxy, - a cycloalkyl - a (C 1 -C 6 )alkyloxy optionally substituted by a halogen, 30 - a halogen, - a -NR d R e with R d and R e represent a hydrogen, a (C 1 - C 6 )alkyl, or a cyclopropyl, - a cyano,
- R 3 represents a 3-14 membered ring which is a heteroaryl optionally substituted by at least one radical A as defined herein. 10
- R 3 represents a 3-14 membered ring, saturated, partially unsaturated, or unsaturated, selected in a group consisting of a phenyl, a pyrimidinyl, a pyrazinyl, a pyridazinyl, a pyrazolyl, an imidazolyl, a thiazolyl, an isoxazolyl, a pyridinyl, an indolyl, , an imidazolyl, a triazolyl, an indazolyl, a thiazolyl, a thiophenyl, a benzoxazolyl (1,3- benzoxazolyl), a benzofuranyl, a benzimidazolyl, a cyclopropyl, a cycl
- R 3 represents a 3-14 membered ring selected in a group consisting of a pyrimidinyl, a pyrazolyl, an imidazolyl, a thiazolyl, an isoxazolyl, a pyridinyl, an indolyl, a pyridazinyl, a 1,3-benzoxazolyl, a benzimidazolyl, and an indazolyl, particularly a pyrimidinyl, a pyrazolyl, an isoxazolyl, a pyridinyl, and an indolyl, said radical being optionally substituted by at least 25 one radical A as defined herein.
- R 3 represents a 3-14 membered ring, saturated, partially unsaturated, or unsaturated, selected in a group consisting of: o an aryl, particularly a phenyl, said aryl is optionally substituted by at least one radical selected in a group consisting of: 30 - a (C 1 -C 6 )alkyl, particularly a methyl, - a (C 1 -C 6 )alkyloxy, particularly a methoxy or an ethoxy, - a cyano, - a hydroxy, - a halogen, particularly a chlorine, - a -NR d R e with R d and R e represent a hydrogen, and - a -NH-CO-(C 1 -C 6 )alkyl, particularly a -NH-CO-CH 3 , o a heteroaryl, particularly a pyrimidinyl, a pyrazinyl, a pyri
- R 3 represents a 3-14 membered ring, which is a heteroaryl, particularly, a pyrimidinyl, a pyrazolyl, or an isoxazolyl, said heteroaryl is optionally substituted by at least one (C 1 -C 6 )alkyl, particularly a methyl, and/or one cyano.
- R 3 represents a radical selected in a group consisting of: o a (C 1 -C 6 )alkyl optionally substituted by at least one radical B or a cycloalkyl optionally substituted by at least one radical B, o a (C 2 -C 6 )alkenyl optionally substituted by at least one radical B, 5 o a (C2-C6)alkynyl optionally substituted by at least one radical B, o a (C 1 -C 6 )alkyloxy optionally substituted by at least one radical B, o a hydroxy, o a cyano, o a halogen, 10 o a -NR f’ R g’ with R f’ and R g’ , represent independently a hydrogen, a (C 1 -C 6 )alkyl optionally substituted by at least one radical B, a cycloalkyl optionally substituted by at least one radical B, a heterocycloal
- said at least one radical B is selected in a group consisting of: - a (C 1 -C 6 )alkyloxy, - a halogen, - a -O-CO-(C 1 -C 6 )alkyl, 20 - a hydroxy, and - a -NH-CO-(C 1 -C 6 )alkyl.
- R 3 represents a radical selected in a group consisting of: o a (C 1 -C 6 )alkyl, particularly a methyl, an ethyl, an isopropyl, or an isobutyl, said (C 1 -C 6 )alkyl is optionally substituted by a (C 1 - 25 C 6 )alkyloxy, particularly a methoxy, a hydroxy, at least one halogen, or a -NH-CO-(C 1 -C 6 )alkyl, particularly a -NH-CO-CH 3 , o a (C 2 -C 6 )alkenyl, particularly an isopropenyl, o a (C 2 -C 6 )alkynyl optionally substituted by a heteroaryl, particularly a pyrazolyl, or a (C 1 -C 6 )alkyl optionally substituted by a hydroxy, 30 o a (C 1 -C 6 )
- a compound of formula (I) is such that R 4 , R 5 , R 6 , R 7 , and R 8 represent a hydrogen.
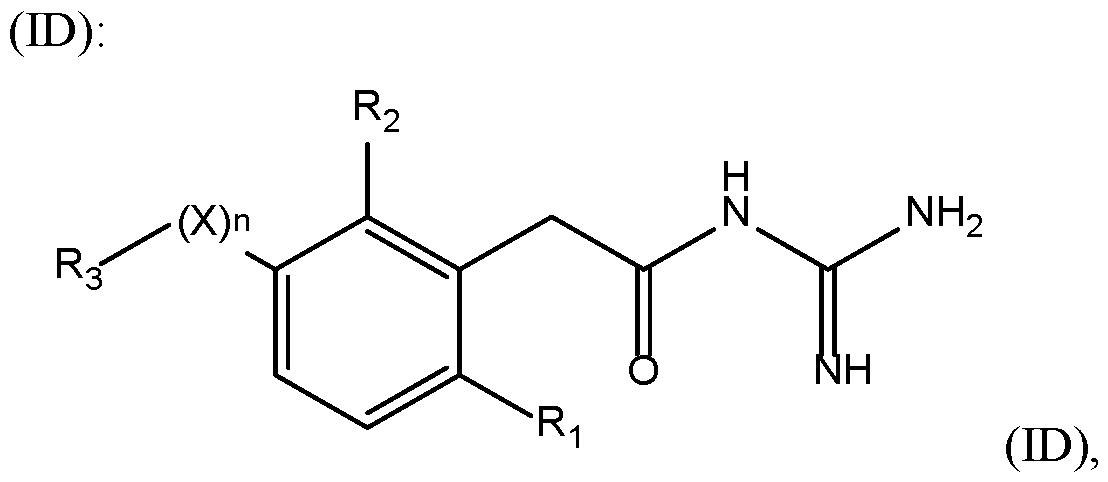
- a compound of the invention has the 15 following formula with X, n, R 1 , R 2 , and R 3 are as defined herein.
- a compound of formula (I) is such that n is 0, and R 4 , R 5 , 20 R 6 , R 7 , and R 8 represent a hydrogen.
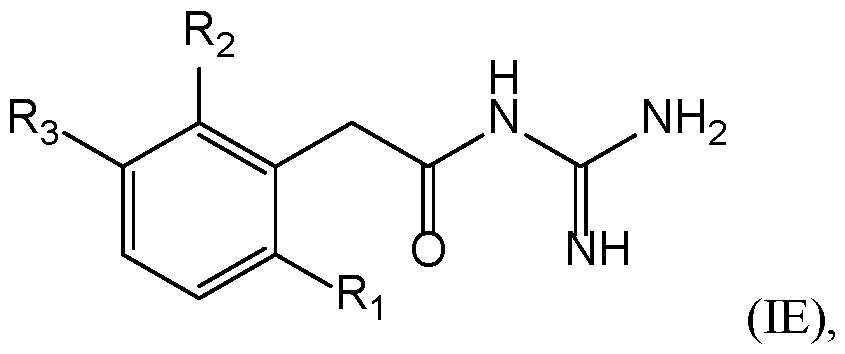
- a compound of the invention has the following formula (IE): with R 1 , R 2 , and R 3 are as defined herein.
- a compound of formula (I) is such that n is 1, and R 4 , R 5 , R 6 , R 7 , and R 8 represent a hydrogen.
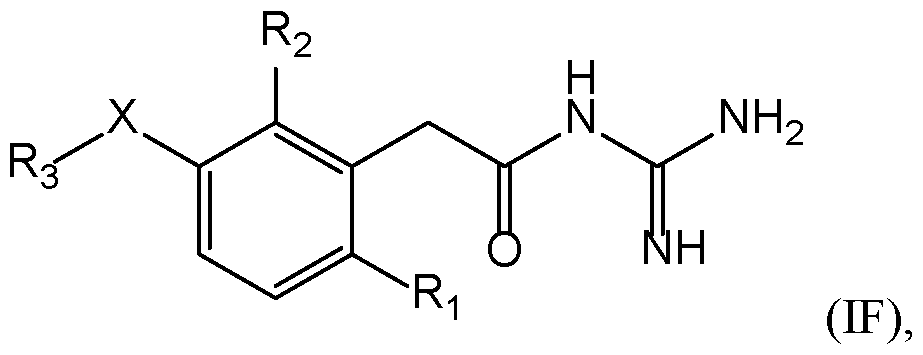
- a compound of the invention has the following formula (IF): with X, R 1 , R 2 , and R 3 are as defined herein.
- a compound of formula (ID) or (IF) is such that X is selected from 5 the group consisting of -(CH 2 ) m - with m is from 1 to 3, particularly 1, -O-, -S-, -CO-, -C(O)-O- , -O-C(O)-, -SO-, and -SO 2 -.
- X is selected from the group consisting of -(CH 2 ) m - with m is from 1 to 3 substituted by at least one R a , -NR a -, -CO-NR a -, -NR a -CO-, -SO 2 -NR a , - NR a -SO 2 -, and -NR a -CO-NR a -, with R a being a hydrogen, a (C 1 -C 6 )alkyl optionally substituted 10 by at least one radical C, a cycloalkyl optionally substituted by at least one radical C, or a heterocycloalkyl optionally substituted by at least one radical C; said radical C being selected in a group consisting of: - a (C 1 -C 6 )alkyloxy, - a halogen, 15 - a -O-C(O)-(C 1 -C 6 )alkyl,
- R a is a hydrogen, a (C 1 -C 6 )alkyl, a cycloalkyl, or a heterocycloalkyl.
- R a is a (C 1 -C 6 )alkyl substituted by at least one radical C, a cycloalkyl substituted by at least one radical C, or a heterocycloalkyl substituted by at least one 5 radical C; said radical C being selected in a group as defined herein.
- R a is a hydrogen.
- X represents -NH-, -CO-NH-, -NH-CO-, -SO 2 -NH-, -NH-SO 2 -, or -NH-CO-NH-.
- the compounds of formula (ID), (IE) or (IF) are such that10 R 1 and R 2 represent independently a radical selected in a group consisting of a hydrogen, a (C 1 - C 6 )alkyl optionally substituted by a (C 1 -C 6 )alkyloxy, a hydroxy, at least one halogen, and a cycloalkyl, a halogen, a cyano, a 3-14 membered ring, saturated, partially unsaturated, or unsaturated selected in a group consisting of a cycloalkyl, a heterocycloalkyl, an aryl, and an heteroaryl, said 3-14 membered ring is optionally substituted by a (C 1 -C 6
- R 1 represents a (C 1 -C 6 )alkyl or a halogen.
- R 1 is selected from the group consisting of methyl, bromine, chlorine or fluorine, preferably methyl, 20 chlorine or fluorine.
- R 1 represents a cyclopropyl, an optionally substituted phenyl (e.g., phenyl substituted by -(SO 2 )-(C 1 -C 6 )alkyl, (C 1 -C 6 )alkyloxy, or halogen, in particular -(SO 2 )-methyl, methoxy or chlorine), or an optionally substituted pyrazolyl or pyrimidinyl.
- R 2 represent a hydrogen, a (C 1 -C 6 )alkyl optionally 25 substituted by a (C 1 -C 6 )alkyloxy, a hydroxy, at least one halogen, and a cycloalkyl, a halogen, a cyano, a 3-14 membered ring, saturated, partially unsaturated, or unsaturated selected in a group consisting of a cycloalkyl, a heterocycloalkyl, an aryl, and an heteroaryl, said 3-14 membered ring is optionally substituted by a (C 1 -C 6 )alkyl, a hydroxy, a halogen, a -SO 2 -(C 1 - C 6 )alkyl, and a (C 1 -C 6 )alkyloxy, or a (C 1 -C 6 )alkyloxy optionally substituted by at least one 30 halogen.
- R 2 represents a (C 1 -C 6 )alkyl, a halogen, a cyano, a cyclopropyl, an optionally substituted phenyl, or an optionally substituted pyrazolyl or pyrimidinyl.
- R 2 is selected from the group consisting of methyl, bromine, chlorine, fluorine, a phenyl substituted by -(SO 2 )-(C 1 -C 6 )alkyl, (C 1 -C 6 )alkyloxy or halogen, in particular by -(SO 2 )-methyl, methoxy or chlorine, or an optionally substituted pyrazolyl or pyrimidinyl.
- At least one among R 1 and R 2 comprises a halogen, for instance represents a halogen or a radical substituted by a halogen.
- both R 1 and R 2 comprise a halogen, for 5 instance represent a halogen or a radical substituted by a halogen.
- the halogen is chlorine or fluorine, especially chlorine.
- R 1 and R 2 are identical.
- a compound of formula (ID), (IE) or (IF) is such that: ⁇ R 1 represents a (C 1 -C 6 )alkyl, preferably a methyl, a halogen, preferably a chlorine or a 10 fluorine, more preferably a chlorine; and ⁇ R 2 represents a (C 1 -C 6 )alkyl, preferably a methyl, a halogen, preferably a chlorine or a fluorine, more preferably a chlorine or a cyano.
- a compound of formula (ID), (IE) or (IF) is such that R 3 represents a 3-14 membered ring, saturated, partially unsaturated, or unsaturated, selected in a 15 group consisting of: o an aryl, particularly a phenyl, said aryl is optionally substituted by at least one radical selected in a group consisting of: - a (C 1 -C 6 )alkyl, particularly a methyl, - a (C 1 -C 6 )alkyloxy, particularly a methoxy or an ethoxy, 20 - a cyano, - a hydroxy, - a halogen, particularly a chlorine, - a -NR d R e with R d and R e represent a hydrogen, and - a -NH-CO-(C 1 -C 6 )alkyl, particularly a -NH-CO-CH 3 , 25 o a heteroaryl, particularly a
- a compound of formula (ID), (IE) or (IF) is such that R 3 represents a 3-14 membered ring, saturated, partially unsaturated, or unsaturated, which is a heteroaryl optionally substituted by at least one radical selected in a group consisting of: - a (C 1 -C 6 )alkyl, particularly a methyl, 25 - a (C 1 -C 6 )alkyloxy, particularly a methoxy, and - a cyano.
- R 3 represents a 3-14 membered ring selected in a group consisting of a pyrimidinyl, a pyrazolyl, an imidazolyl, a thiazolyl, an isoxazolyl, a pyridinyl, an indolyl, a pyridazinyl, a 1,3-benzoxazolyl, a benzimidazolyl, and an indazolyl, particularly a pyrimidinyl, a pyrazolyl, 30 and an isoxazolyl, said radical being optionally substituted by at least one radical A as defined herein.
- a compound of formula (ID) or (IE) is such that R 3 represents a 3-14 membered ring which is a heteroaryl, such as a pyrimidinyl, a pyrazolyl, or an isoxazolyl, said heteroaryl is optionally substituted by at least one (C 1 -C 6 )alkyl, particularly a methyl, and/or a cyano.
- a heteroaryl such as a pyrimidinyl, a pyrazolyl, or an isoxazolyl
- said heteroaryl is optionally substituted by at least one (C 1 -C 6 )alkyl, particularly a methyl, and/or a cyano.
- a compound of formula (ID) or (IE) is such that R 3 represents 5 a radical selected in a group consisting of: o a (C 1 -C 6 )alkyl, particularly a methyl, or an isobutyl, o a (C 2 -C 6 )alkenyl, particularly an isopropenyl, o a (C 1 -C 6 )alkyloxy, particularly a methoxy, or a butoxy, o a -NR f’ R g’ with R f’ and R g’ represent independently a hydrogen or a (C 1 - 10 C 6 )alkyl, particularly a methyl or an isopropyl, with the proviso that when one of R f’ or R g’ is a hydrogen, then the other is not a hydrogen, o a -NH-CO-(C 1 -C 6 )alkyl, particularly a -NH-CO-CH 3 , and
- a compound of formula (ID) or (IE) is such that: ⁇ R 1 represents a (C 1 -C 6 )alkyl, preferably a methyl, a halogen, preferably a chlorine or a fluorine, more preferably a chlorine; ⁇ R 2 represents a (C 1 -C 6 )alkyl, preferably a methyl, a halogen, preferably a chlorine or a fluorine, more preferably a chlorine, or a cyano; and 20 ⁇ R 3 represents a 3-14 membered ring, which is a heteroaryl optionally substituted by at least one radical A as defined herein, particularly a pyrimidinyl, a pyrazolyl, or an isoxazolyl, optionally substituted by at least one (C 1 -C 6 )alkyl, particularly a methyl, and/or a cyano.
- a compound of formula (ID) or (IE) is such that: ⁇ R 1 represents a (C 1 -C 6 )alkyl, preferably a methyl, a halogen, preferably a chlorine or a fluorine, more preferably a chlorine; ⁇ R 2 represents a (C 1 -C 6 )alkyl, preferably a methyl, a halogen, preferably a chlorine or a fluorine, more preferably a chlorine, or a cyano; and 30 ⁇ R 3 represents a 3-14 membered ring, which is a cycloalkyl optionally substituted by at least one radical A as defined herein, particularly a cyclopropyl.
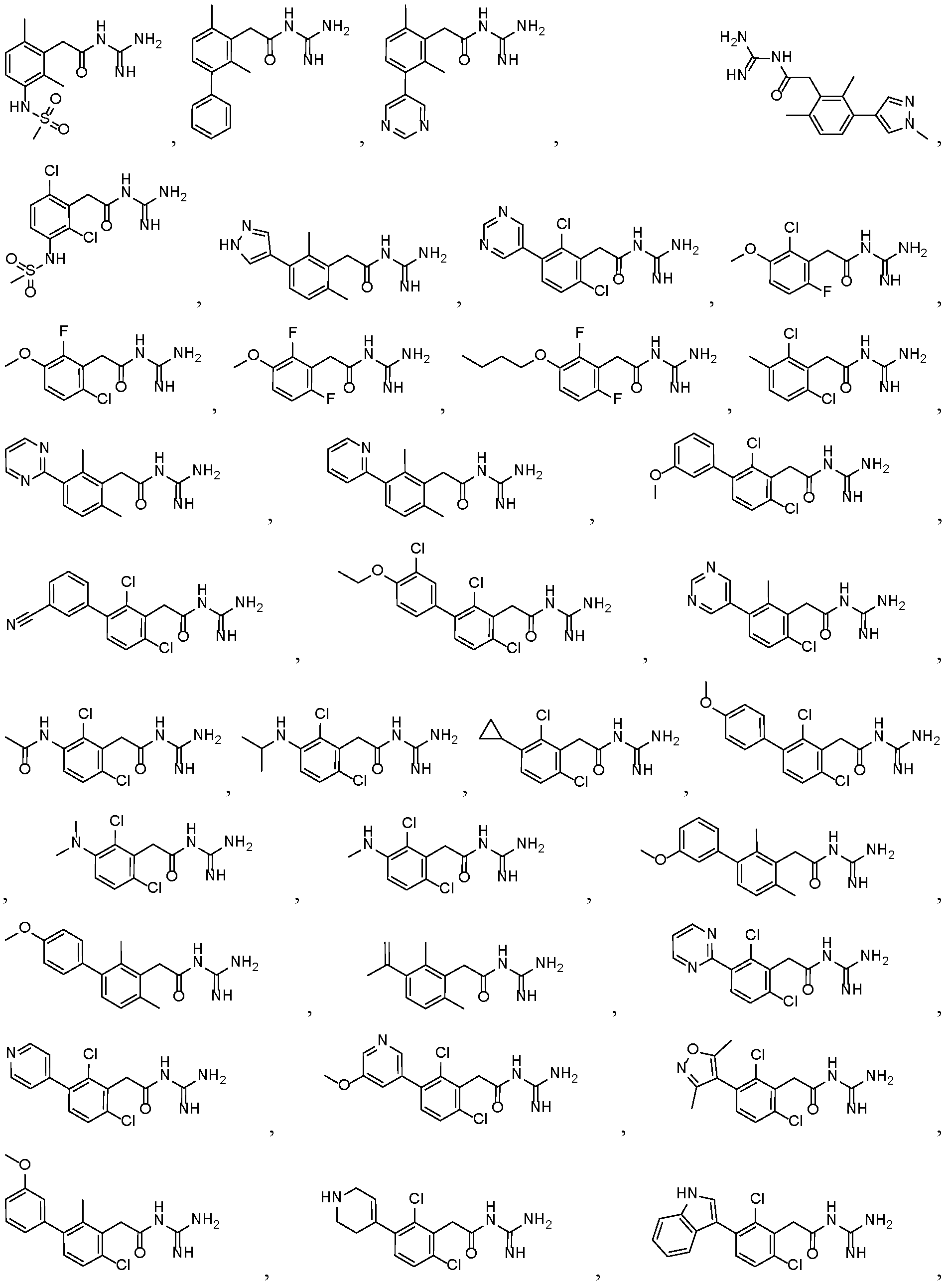
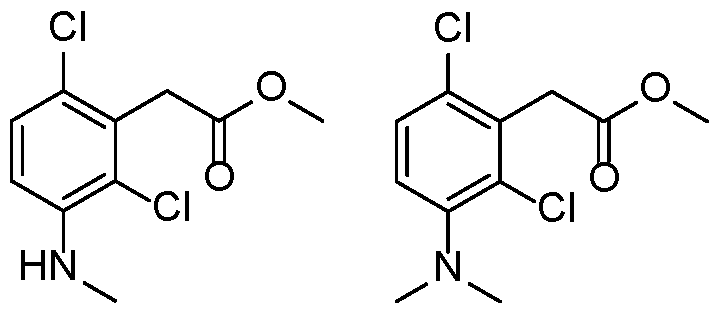
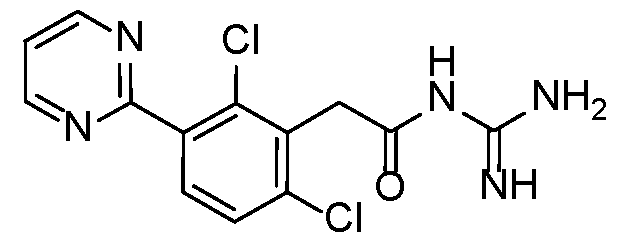
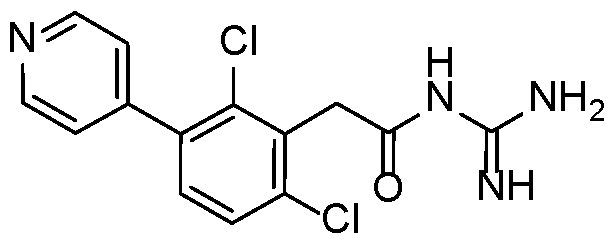
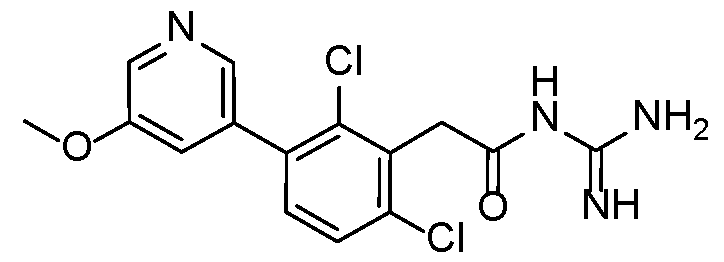
- a preferred compound of formula (I) or (IA) or (IB) or (IC) or (ID) or (IE) or (IF) according to the invention has the following formula selected in a group consisting of: 5 10
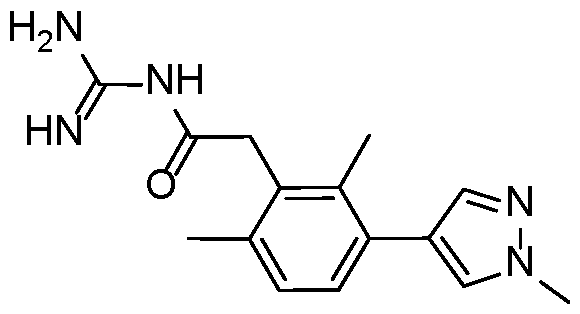
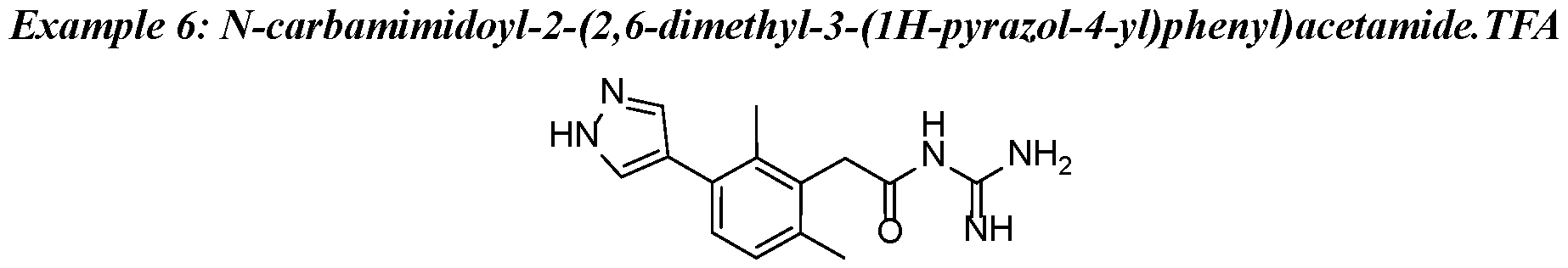
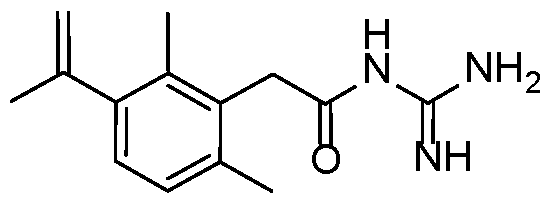
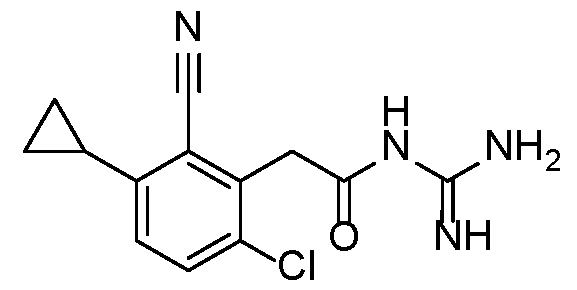
- a preferred compound of the invention is selected in a group consisting of: - N-carbamimidoyl-2-(2,6-dimethyl-3-(methylsulfonamido)phenyl)acetamide; - N-carbamimidoyl-2-(2,4-dimethyl-[1,1'-biphenyl]-3-yl)acetamide; - N-carbamimidoyl-2-(2,6-dimethyl-3-(pyrimidin-5-yl)phenyl)acetamide; 10 - N-carbamimidoyl-2-(2,6-dimethyl-3-(1-methyl-1H-pyrazol-4-yl)phenyl) acetamide; - N-carbamimidoyl-2-(2,6-dichloro-3-(methylsulfonamido)phenyl)acetamide; - N-carbamimido
- the present invention relates to a pharmaceutical or veterinary composition
- a pharmaceutical or veterinary composition comprising a compound of formula (I), (IA), (IB), (IC), (ID), (IE) or (IF) according to the invention or any particular compound as disclosed herein.
- the pharmaceutical composition further comprises a pharmaceutically or veterinary acceptable carrier or excipient.
- the present invention relates to the use of a compound of formula (I), (IA), (IB), (IC), (ID), 10 (IE) or (IF) according to the invention or any particular compound as disclosed herein as a drug or a medicine.
- the invention further relates to a method for treating a disease in a subject, wherein a therapeutically effective amount of a compound of formula (I), (IA), (IB), (IC), (ID), (IE) or (IF) according to the invention or any particular compound as disclosed herein, is administered to said subject in need thereof.
- the invention also relates to the use of a compound 15 of formula (I), (IA), (IB), (IC), (ID), (IE) or (IF) according to the invention or any particular compound as disclosed herein, for the manufacture of a medicine.
- the invention also relates to a pharmaceutical composition comprising a compound of formula (I), (IA), (IB), (IC), (ID), (IE) or (IF) according to the invention or any particular compound as disclosed herein for use as a drug.
- the present invention also concerns: - a compound of formula (I), (IA), (IB), (IC), (ID), (IE) or (IF) as defined above including any one of the disclosed embodiments and any particular compound as disclosed herein, or a pharmaceutical composition comprising such a compound for preventing and/or treating or for 25 use for preventing and/or treating a cancer; and/or - a pharmaceutical composition comprising a compound of formula (I), (IA), (IB), (IC), (ID), (IE) or (IF) as defined above including any one of the disclosed embodiments and any particular compound as disclosed herein, and an antitumor drug, in particular for the prevention and/or the treatment of cancer or for use in the prevention and/or the treatment of cancer; and/or 30 - a
- cancer refers to the presence of cells possessing characteristics typical of cancer-causing cells, such as uncontrolled proliferation, immortality, metastatic potential, rapid growth and proliferation rate, and certain characteristic morphological features.
- the cancer may be solid tumor or hematopoietic tumor.
- cancer examples include, but are 10 not limited to, solid tumors and hematological cancers, including carcinoma, lymphoma, blastoma (including medulloblastoma and retinoblastoma), sarcoma (including liposarcoma and synovial cell sarcoma), neuroendocrine tumors (including carcinoid tumors, gastrinoma, and islet cell cancer), mesothelioma, schwannoma (including acoustic neuroma), meningioma, adenocarcinoma, melanoma, and leukemia or lymphoid malignancies.
- solid tumors and hematological cancers including carcinoma, lymphoma, blastoma (including medulloblastoma and retinoblastoma), sarcoma (including liposarcoma and synovial cell sarcoma), neuroendocrine tumors (including carcinoid tumors, gastrinoma, and is
- cancers include chronic myeloid leukemia, acute lymphoblastic leukemia, Philadelphia chromosome positive acute lymphoblastic leukemia (Ph+ ALL), squamous cell carcinoma, lung cancer, small-cell lung cancer, non-small cell lung cancer, glioma, gastrointestinal cancer, renal cancer, ovarian cancer, bile duct cancer, liver cancer, colorectal cancer, endometrial cancer, kidney cancer, prostate cancer, melanoma, skin cancer, thyroid 20 cancer, neuroblastoma, osteosarcoma, pancreatic cancer, glioblastoma multiforme, cervical cancer, stomach cancer, bladder cancer, hepatoma, breast cancer, oesophagal cancer, colon cancer, head and neck cancer, brain cancer, gastric cancer, germ cell tumor, pediatric sarcoma, sinonasal natural killer, multiple myeloma, acute myelogenous leukemia (AML), chronic lymphocytic leukemia, mastocytosis and
- the cancer is chosen among of myelofibrosis, acute lymphoblastic leukemia, acute myeloblastic leukemia adrenal gland carcinoma, bile duct cancer, bladder cancer, breast cancer, cervical cancer, colorectal cancer, endometrial cancer, esophageal cancer, gastric cancer, gastrointestinal stromal tumors, glioblastoma, head and neck cancer, 30 hepatocellular carcinoma, Hodgkin’s lymphoma, kidney cancer, lung cancer, melanoma, Merkel cell skin cancer, mesothelioma, multiple myeloma, myeloproliferative disorders, non- Hodgkin lymphoma, ovarian cancer, pancreatic cancer, prostate cancer, salivary gland cancer, sarcoma, squamous cell carcinoma, testicular cancer, thyroid cancer, urothelial carcinoma, and uveal melanoma.
- the cancer is resistant or has a low response to immunotherapy. More specifically, the cancer can be resistant to a treatment with a checkpoint inhibitor such as an antibody against PD-1, PD-L1, CTLA-4 and the like.
- the administration route can be topical, transdermal, oral, rectal, sublingual, intranasal, intrathecal, intratumor or parenteral (including subcutaneous, intramuscular, intravenous and/or intradermal). Preferably, the administration route is parental, oral or topical.
- the pharmaceutical composition is adapted for one or several of the above-mentioned routes.
- the pharmaceutical composition, kit, product or combined preparation is preferably administered 10 by injection or by intravenous infusion or suitable sterile solutions, or in the form of liquid or solid doses via the alimentary canal.
- the pharmaceutical composition can be formulated as solutions in pharmaceutically compatible solvents or as emulsions, suspensions or dispersions in suitable pharmaceutical solvents or vehicles, or as pills, tablets or capsules that contain solid vehicles in a way known in the art.
- Formulations of the present invention suitable for oral administration may be in the form of discrete units as capsules, sachets, tablets or lozenges, each containing a predetermined amount of the active ingredient; in the form of a powder or granules; in the form of a solution or a suspension in an aqueous liquid or non-aqueous liquid; or in the form of an oil-in-water emulsion or a water-in-oil emulsion.
- Formulations for rectal administration may be in the form 20 of a suppository incorporating the active ingredient and carrier such as cocoa butter, or in the form of an enema.
- Formulations suitable for parenteral administration conveniently comprise a sterile oily or aqueous preparation of the active ingredient which is preferably isotonic with the blood of the recipient. Every such formulation can also contain other pharmaceutically compatible and nontoxic auxiliary agents, such as, e.g. stabilizers, antioxidants, binders, dyes, 25 emulsifiers or flavoring substances.
- the formulations of the present invention comprise an active ingredient in association with a pharmaceutically acceptable carrier therefore and optionally other therapeutic ingredients.
- the carrier must be "acceptable" in the sense of being compatible with the other ingredients of the formulations and not deleterious to the recipient thereof.
- the pharmaceutical compositions are advantageously applied by injection or 30 intravenous infusion of suitable sterile solutions or as oral dosage by the digestive tract.
- compositions according to the invention may be formulated to release the active drug substantially immediately upon administration or at any predetermined time or time period after administration.
- the treatment with the compound according to the invention or the pharmaceutical 5 composition according to the invention starts no longer than a month, preferably no longer than a week, after the diagnosis of the disease. In a particular embodiment, the treatment starts the day of the diagnosis.
- the compound according to the invention or the pharmaceutical composition according to the invention may be administered as a single dose or in multiple doses.
- the treatment is administered regularly, preferably between every day and every month, more preferably between every day and every two weeks, more preferably between every day and every week, even more preferably the treatment is administered every day.
- the treatment is administered several times a day, preferably 2 or 3 times a day, even more preferably 3 times a day.
- the duration of treatment with the compound according to the invention or the pharmaceutical composition according to the invention is preferably comprised between 1 day and 50 weeks, more preferably between 1 day and 30 weeks, still more preferably between 1 day and 15 weeks, even more preferably between 1 day and 10 weeks.
- the duration of the treatment is of about 1 week.
- the treatment may last as long as the disease 20 persists.
- the amount of compound according to the invention or of pharmaceutical composition according to the invention to be administered has to be determined by standard procedure well known by those of ordinary skills in the art. Physiological data of the patient (e.g. age, size, and weight) and the routes of administration have to be taken into account to determine the appropriate dosage, so as a therapeutically effective amount will be administered to the patient. 25 In a particular embodiment, is administered at a dose ranging from 0.001 mg/kg body weight to 30 mg/kg body weight. In one embodiment, the compound of the invention can be used in combination with another antitumor drug or antineoplastic agent.
- the additional antitumor drug can be selected in the non-exhaustive list of antitumor agents consisting of an inhibitor of topoisomerases I or II, an anti-mitotic agent, a DNA alkylating agent, an agent causing crosslinking of DNA, an anti-metabolic agent, a targeted agent such as a kinase inhibitor, a histone deacetylase inhibitor and an anti-EGFR agent and/or a therapeutical antibody designed to mediate cytotoxicity against the cancer cells or to modulate one of their key biological functions.
- antitumor agents consisting of an inhibitor of topoisomerases I or II, an anti-mitotic agent, a DNA alkylating agent, an agent causing crosslinking of DNA, an anti-metabolic agent, a targeted agent such as a kinase inhibitor, a histone deacetylase inhibitor and an anti-EGFR agent and/or a therapeutical antibody designed to mediate cytotoxicity against the cancer cells or to modulate one of their key biological functions
- Antimitotic agents include, but are not limited to, paclitaxel, docetaxel and analogs such as larotaxel (also called XRP9881; Sanofi-Aventis), XRP6258 (Sanofi-Aventis), BMS-184476 5 (Bristol-Meyer-Squibb), BMS-188797 (Bristol-Meyer-Squibb), BMS-275183 (Bristol-Meyer- Squibb), ortataxel (also called IDN 5109, BAY 59-8862 or SB-T-101131; Bristol-Meyer- Squibb), RPR 109881A (Bristol-Meyer-Squibb), RPR 116258 (Bristol-Meyer-Squibb), NBT- 287 (TAPESTRY), PG-paclitaxel (also called CT-2103, PPX, paclitaxel poliglumex, paclitaxel polyglutamate or Xyotax
- antimitotic agents are docetaxel, paclitaxel, and is more preferably docetaxel.
- Inhibitors of topoisomerases I and/or II include, but are not limited to etoposide, topotecan, 15 camptothecin, irinotecan, amsacrine, intoplicin, anthracyclines such as doxorubicin, epirubicin, daunorubicin, idarubicin and mitoxantrone.
- Inhibitors of topoisomerase I and II include, but are not limited to intoplicin.
- the additional antitumor agent can be alkylating agents including, without limitation, nitrogen mustards, ethylenimine derivatives, alkyl sulfonates, nitrosoureas, metal salts and triazenes.
- alkylating agents including, without limitation, nitrogen mustards, ethylenimine derivatives, alkyl sulfonates, nitrosoureas, metal salts and triazenes.
- Non-exhaustive examples thereof include uracil mustard, chlormethine, cyclophosphamide (CYTOXAN ® ), ifosfamide, melphalan, chlorambucil, pipobroman, triethylenemelamine, triethylenethiophosphoramine, busulfan, carmustine, lomustine, cisplatin, carboplatin, fotemustine, oxaliplatin, thiotepa, streptozocin, dacarbazine, and temozolomide.
- the DNA alkylating agent is preferably cisplatin, carboplatin, temozolomide, 25 fotemustine or dacarbazine.
- Anti-metabolic agents block the enzymes responsible for nucleic acid synthesis or become incorporated into DNA, which produces an incorrect genetic code and leads to apoptosis.
- Non- exhaustive examples thereof include, without limitation, folic acid antagonists, pyrimidine analogs, purine analogs and adenosine deaminase inhibitors, and more particularly 30 methotrexate, floxuridine, cytarabine, 6-mercaptopurine, 6-thioguanine, fludarabine phosphate, pentostatine, 5-fluorouracil, gemcitabine and capecitabine.
- such an agent is gemcitabine.
- the additional anti-tumor agent can also be a targeted agent, in particular a kinase inhibitor.
- the kinase may be selected from the group consisting of intracellular tyrosine or serine/threonine kinases, receptors tyrosine or serine/threonine kinase.
- the kinase could be selected among EGFR family, ALK, B-Raf, MEK, and mTOR.
- the agents may have ability to inhibit angiogenesis based on the inhibitory activities on VEGFR and PDGFR kinases.
- the targeted agent can be selected among the multiple kinase inhibitor 5 drugs which are already approved: Gleevec ® , which inhibits Bcr-Abl and c-Kit, and Iressa ® and Tarceva ® , which both inhibit EGFR, sorafenib (Nexavar ® , BAY 43-9006) which inhibits Raf, dasatinib (BMS-354825) and nilotinib (AMN-107, Tasigna ® ) which also inhibits Bcr-Abl, lapatinib which also inhibits EGFR, temsirolimus (Torisel ® , CCI-779) which targets the mTOR pathway, sunitinib (Student ® , SU11248) which inhibits several targets including VEGFR as10 well as specific antibodies inactivating kinase receptors: Herceptin ® and Avastin ® .
- the anti- EGFR agent can be selected among gefitinib, erlotinib, lapatinib, vandetanib, afatinib, osimertinib, neratinib, dacomitinib, brigatinib, canertinib, naquotinib, clawartinib, pelitinib, rociletinib, icotinib, AZD3759, AZ5104 (CAS 1421373-98-9), poziotinib, WZ4002, preferably is erlotinib or cetuximab.
- the ALK inhibitor can be selected among crizotinib, 15 entrectinib, ceritinib, alectinib, brigatinib, lorlatinib, TSR-011, CEP-37440, and ensartinib.
- the B-Raf inhibitor can be selected among vemurafenib, dabrafenib, regorafenib, and PLX4720.
- the MEK inhibitor can be selected among cobimetinib, trametinib, binimetinib, selumetinib, PD-325901, CI-1040, PD035901, U0126, TAK-733.
- the antitumor agent can be IDO1 inhibitors such as epacadostat.
- therapy refers to any type of treatment of cancer (i.e., antitumor therapy), including an adjuvant therapy and a neoadjuvant therapy. Therapy comprises radiotherapy and therapies, preferably systemic therapies such as hormone therapy, chemotherapy, immunotherapy and monoclonal antibody therapy.
- adjuvant therapy refers to any type of treatment of cancer given as additional treatment, usually after surgical resection of the primary tumor, in a patient affected with a cancer that is at risk of metastasizing and/or likely to recur. The aim of such an adjuvant treatment is to improve the prognosis.
- Adjuvant therapies comprise radiotherapy and therapy, preferably systemic therapy, such as hormone therapy, chemotherapy, immunotherapy and 30 monoclonal antibody therapy.
- hormone therapy or “hormonal therapy” or “hormonotherapy” refers to a cancer treatment having for purpose to block, add or remove hormones.
- hormone therapy is given to block estrogen and a non-exhaustive list commonly used drugs includes: tamoxifen, toremifene, anastrozole, exemestane, letrozole, goserelin, leuprolide, megestrol acetate, and fluoxymesterone.
- chemotherapeutic treatment refers to a cancer therapeutic treatment using chemical or biological substances, in particular using one or several 5 antineoplastic agents.
- radiationotherapeutic treatment or “radiotherapy” is a term commonly used in the art to refer to multiple types of radiation therapy including internal and external radiation therapies or radioimmunotherapy, and the use of various types of radiations including X-rays, gamma rays, alpha particles, beta particles, photons, electrons, neutrons, radioisotopes, and other forms 10 of ionizing radiations.
- therapeutic antibody refers to any antibody having an anti-tumoral effect.
- the therapeutical antibody is a monoclonal antibody.
- Therapeutic antibodies are generally specific for surface antigens, e.g., membrane antigens. Most preferred therapeutic antibodies are specific for tumor antigens (e.g., molecules specifically expressed by tumor 15 cells), such as CD20, CD52, ErbB2 (or HER2/Neu), CD33, CD22, CD25, MUC-1, CEA, KDR, aVb3, and the like.
- tumor antigens e.g., molecules specifically expressed by tumor 15 cells
- the therapeutical antibody includes, but is not limited to, antibodies such as trastuzumab (anti-HER2 antibody), rituximab (anti-CD20 antibody), alemtuzumab, gemtuzamab, cetuximab, pertuzumab, epratuzumab, basiliximab, daclizumab, labetuzumab, sevirumab, tuvurimab, palivizumab, infliximab, omalizumab, efalizumab, natalizumab, 20 clenoliximab, and bevacizumab.
- the antitumor agent can be an immunomodulator.
- the immunomodulator can be a cancer vaccine, molecules stimulating the immune system such as cytokines, therapeutic antibodies, preferably monoclonal antibodies, in particular antibodies directed 25 against antigens specifically presented or overexpressed at the membrane of tumor cells or directed against cell receptors which blockade prevent tumor growth, adoptive T-cell therapy, immune checkpoint inhibitor treatment, and any combination thereof.
- molecules stimulating the immune system such as cytokines, therapeutic antibodies, preferably monoclonal antibodies, in particular antibodies directed 25 against antigens specifically presented or overexpressed at the membrane of tumor cells or directed against cell receptors which blockade prevent tumor growth, adoptive T-cell therapy, immune checkpoint inhibitor treatment, and any combination thereof.
- the immunomodulator can be: - an immune checkpoint inhibitor (ICI), preferably an inhibitor of of PD-1 (programmed cell 30 death protein 1), PD-L1 (programmed cell death ligand), PD-L2, CTLA-4 (cytotoxic T lymphocyte associated protein 4), TIM-3 (T-cell immunoglobulin and mucin-domain containing-3), LAG-3 (Lymphocyte-activation gene 3), NKG2D, NKG2L, KIR, VISTA, BTLA (B- and T-lymphocyte attenuator), or TIGIT (T cell immunoreceptor with Ig and ITIM domains), , especially an antibody directed against an anti-CTLA-4 such as ipilimumab, an antibody directed against PD-1 such as nivolumab, pembrolizumab, or BGB-A317, an antibody directed against PDL1 such as atezolizumab, avelumab, or durvalumab, an antibody directed against LAG-3 such
- the anti-PD1 antibody can be selected from the group consisting 15 of Pembrolizumab (also known as Keytruda lambrolizumab, MK-3475), Nivolumab (Opdivo, MDX-1106, BMS-936558, ONO-4538), Pidilizumab (CT-011), Cemiplimab (Libtayo), Camrelizumab, AUNP12, AMP-224, AGEN-2034, BGB-A317 (Tisleizumab), PDR001 (spartalizumab), MK-3477, SCH-900475, PF-06801591, JNJ-63723283, genolimzumab (CBT- 501), LZM-009, BCD-100, SHR-1201, BAT-1306, AK-103 (HX-008), MEDI-0680 (also 20 known as AMP-51
- BI-754091 CBT-501, INCSHR1210 (also known as SHR-1210), TSR-042 (also known as ANB011), GLS-010 (also known as WBP3055), AM-0001 (Armo), STI-1110 (see WO 2014/194302), AGEN2034 (see WO 2017/040790), MGA012 (see WO 2017/19846), or IBI308 (see WO 2017/024465, WO 2017/025016, WO 2017/132825, and WO 2017/133540), 25 monoclonal antibodies 5C4, 17D8, 2D3, 4H1, 4A11, 7D3, and 5F4, described in WO 2006/121168.
- Bifunctional or bispecific molecules targeting PD-1 are also known such as RG7769 (Roche), XmAb20717 (Xencor), MEDI5752 (AstraZeneca), FS118 (F-star), SL- 279252 (Takeda) and XmAb23104 (Xencor).
- 30 Antibodies directed against CTLA-4 and bifunctional or bispecific molecules targeting CTLA- 4 are also known such as ipilimumab, tremelimumab, MK-1308, AGEN-1884, XmAb20717 (Xencor), MEDI5752 (AstraZeneca).
- Antibodies directed against TIGIT are also known in the art, such as BMS-986207 or AB154, BMS-986207 CPA.9.086, CHA.9.547.18, CPA.9.018, CPA.9.027, CPA.9.049, CPA.9.057, CPA.9.059, CPA.9.083, CPA.9.089, CPA.9.093, CPA.9.101, CPA.9.103, CHA.9.536.1, CHA.9.536.3, CHA.9.536.4, CHA.9.536.5, CHA.9.536.6, CHA.9.536.7, CHA.9.536.8, CHA.9.560.1, CHA.9.560.3, CHA.9.560.4, CHA.9.560.5, CHA.9.560.6, CHA.9.560.7, CHA.9.560.8, CHA.9.546.1, CHA.9.547.1, CHA.9.547.2, CHA.9.547.3, CHA.9.547.4, 5 CHA.9.547.6, CHA
- Anti-TIGIT antibodies are also disclosed in WO16028656, WO16106302, WO16191643, WO17030823, WO17037707, WO17053748, WO17152088, WO18033798, WO18102536, WO18102746, WO18160704, WO18200430, WO18204363, 10 WO19023504, WO19062832, WO19129221, WO19129261, WO19137548, WO19152574, WO19154415, WO19168382 and WO19215728.
- the LAG-3 inhibitor can be selected from LAG525 (Novartis), BMS-986016 (Bristol-Myers Squibb), or TSR-033 (Tesaro). Further known anti-LAG-3 antibodies include those described, 15 e.g., in WO 2008/132601, WO 2010/019570, WO 2014/140180, WO 2015/116539, WO 2015/200119, WO 2016/028672, US 9,244,059, US 9,505,839, which are incorporated herein by reference in their entirety.
- the TIM-3 inhibitor can be MGB453 (Novartis) or TSR-022 (Tesaro).
- anti- 20 TIM-3 antibodies include those described, e.g., in WO 2016/111947, WO 2016/071448, WO 2016/144803, US 8,552,156, US 8,841,418, and US 9,163,087, which are incorporated herein by reference in their entirety.
- the immunotherapy is selected from the group consisting of ipilimumab, nivolumab, 25 BGB-A317, pembrolizumab, atezolizumab, avelumab, or durvalumab, BMS-986016, and epacadostat, or any combination thereof.
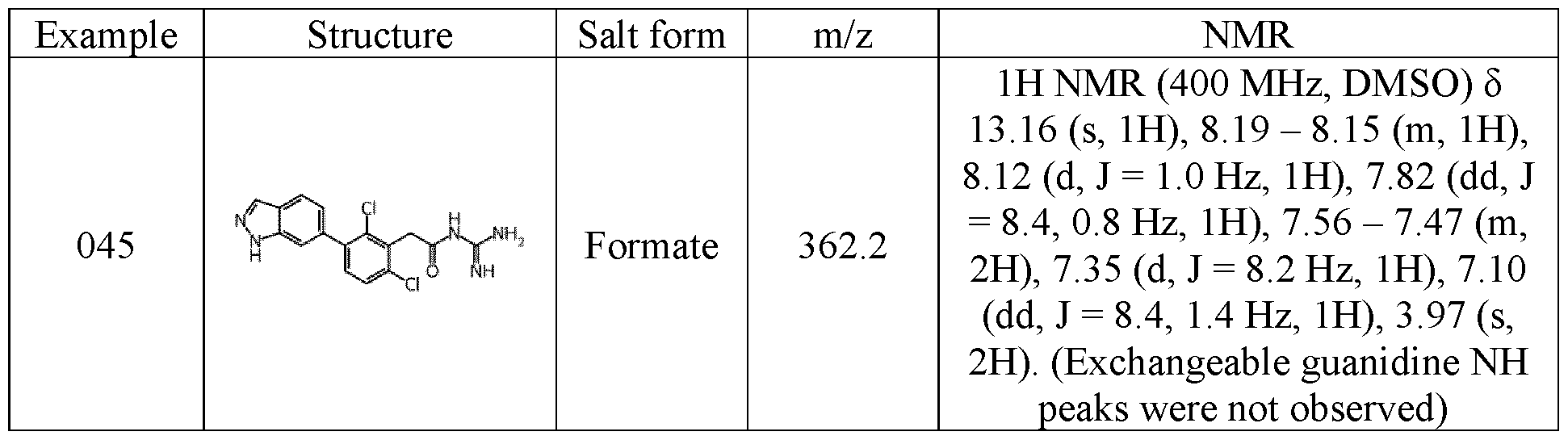
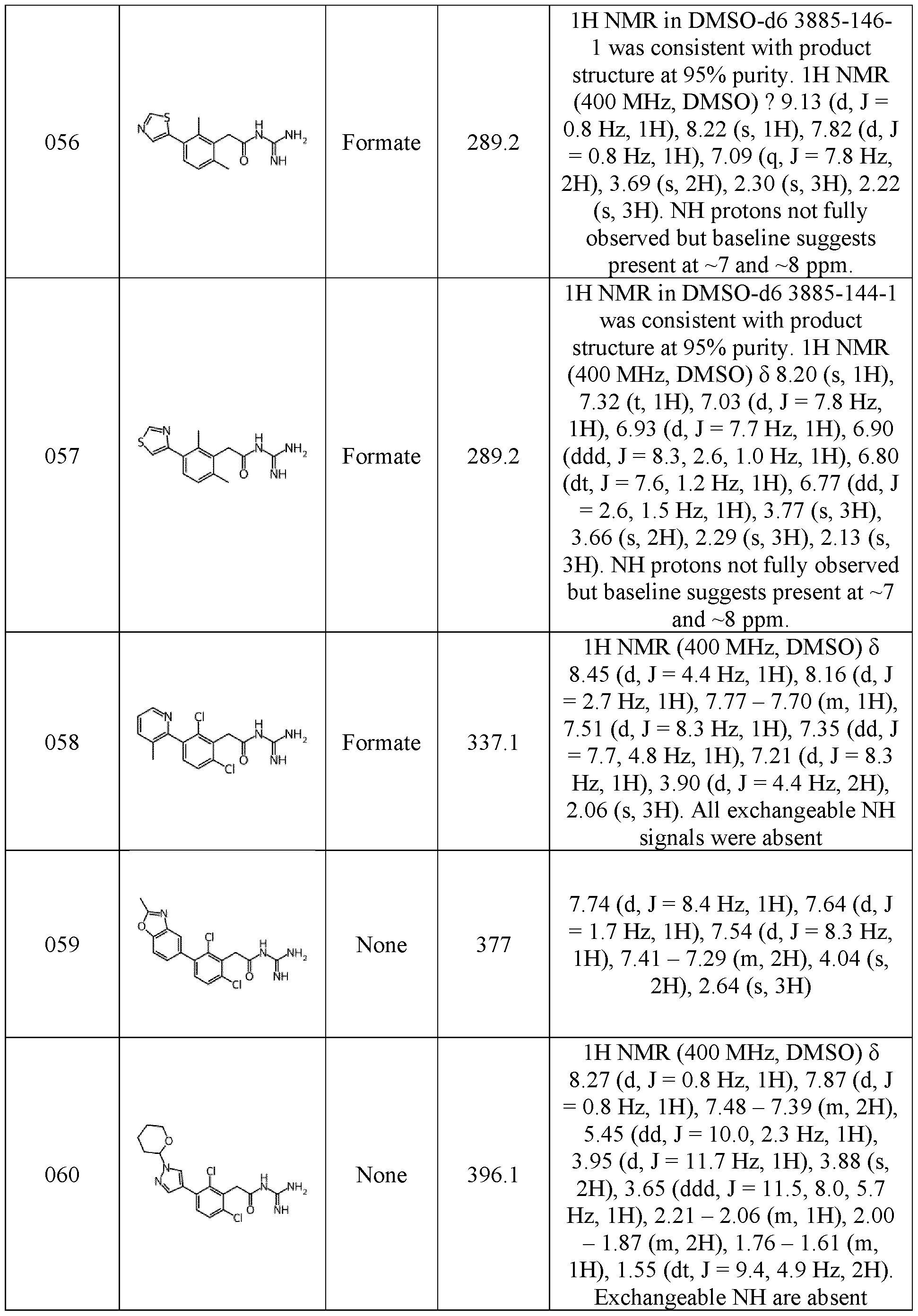
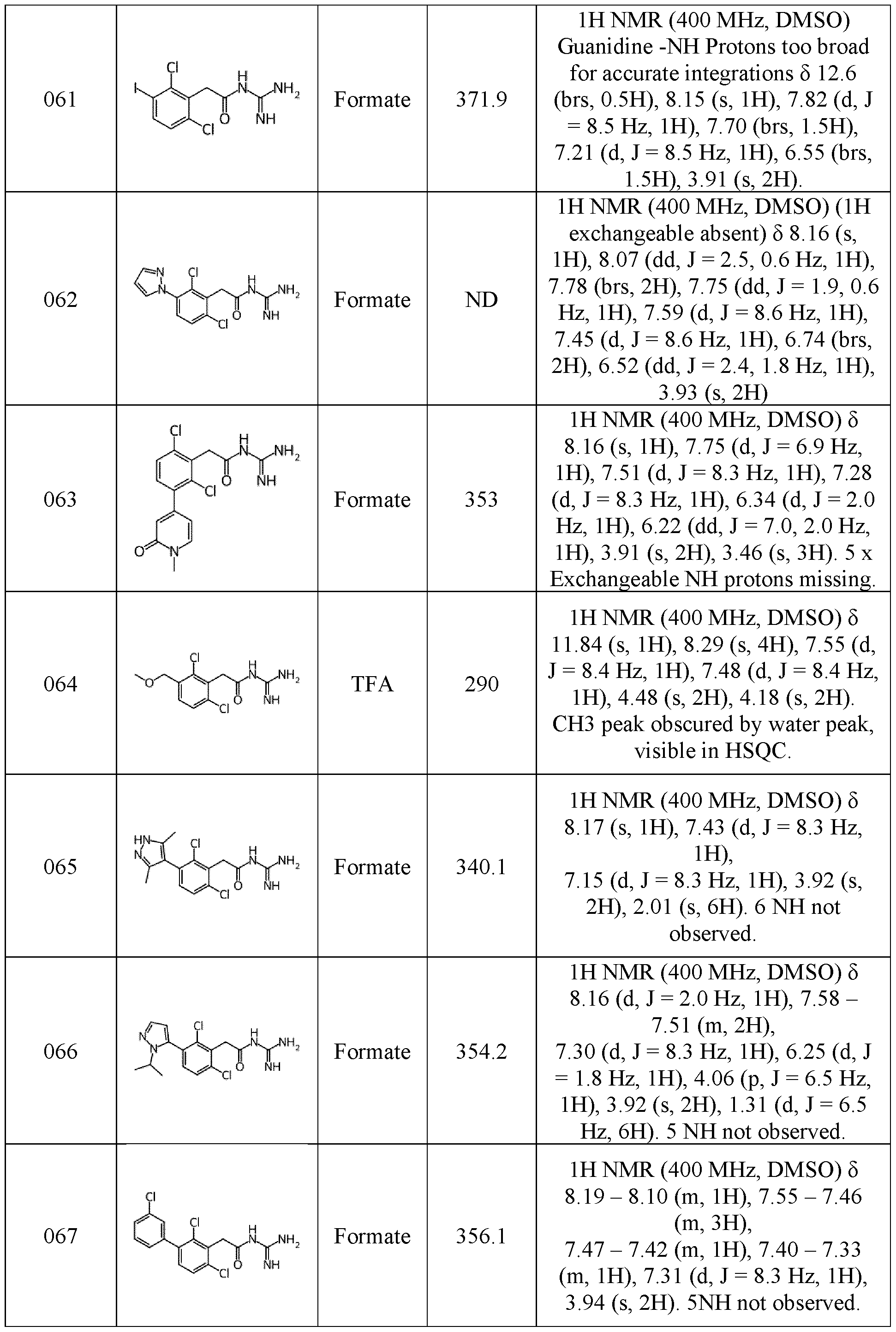
- NMR data provided in the examples described below were obtained as followed: NMR spectra were recorded using a Bruker 400MHz Avance Neo spectrometer fitted with a Bruker 5mm iProbe, or a Bruker 500MHz Avance III HD spectrometer equipped with a Bruker 5mm SmartProbe TM . Spectra were measured at 298 K, unless indicated otherwise, and were referenced relative to the solvent resonance. The chemical shifts are reported in parts per million. Data were acquired using Bruker TopSpin software and processed using MestreNova software.
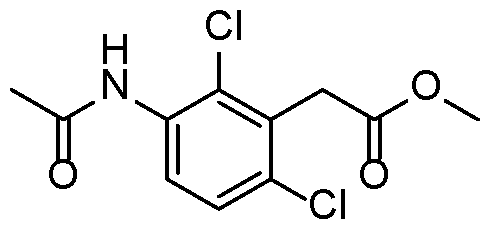
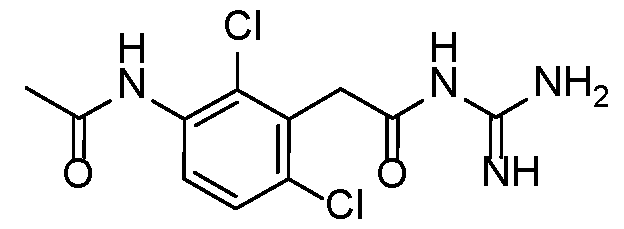
- Step 1 2-(2,6-dimethyl-3-(methylsulfonamido)phenyl)acetic acid 15
- Step 1 ethyl 2-(3-((tert-butoxycarbonyl)amino)-2,6-dimethylphenyl)acetate
- tert-butyl carbamate 148 mg, 0.0013 mol
- Cs 2 CO 3 548 mg, 0.0017 20 mol
- Pd 2 dba 3 39 mg, 0.0001 mol
- XantPhos 24 mg, 0.0001 mol
- reaction mass was degassed with nitrogen and heated to 100 °C for 18 h. After the reaction completion (LCMS), the reaction mixture was treated with water (30 mL) and extracted with EtOAc. The organic layer was dried over anhydrous MgSO 4 , filtered and concentrated under reduced pressure. The crude obtained was purified by column chromatography (EtOAc/Hexane) to afford ethyl 2-(3- 25 ((tert-butoxycarbonyl)amino)-2,6-dimethylphenyl)acetate as a yellow gum (200 mg, 77 %,). The material was used without analysis in the following step.
- Step 2 ethyl 2-(3-amino-2,6-dimethylphenyl)acetate
- ethyl 2-(3-((tert-butoxycarbonyl)amino)-2,6-dimethylphenyl)acetate 200 mg, 0.0005 mol
- DCM DCM
- HCl 1.63 mL of a 4 N solution in 1,4-dioxane, 30 0.0065 mol
- the reaction mixture was stirred for 2 h at RT. After the reaction completion (LCMS), the reaction mixture was concentrated under reduced pressure to afford ethyl 2-(3- amino-2,6-dimethylphenyl)acetate, HCl (212 mg, quant).
- Step 3 2-(2,6-dimethyl-3-(methylsulfonamido)phenyl)acetic acid
- HCl 212 mg, 0.0005 mol
- NEt 3 115 ⁇ L, 0.0008 mol
- methanesulfonyl chloride 51 ⁇ L, 0.0007 mol
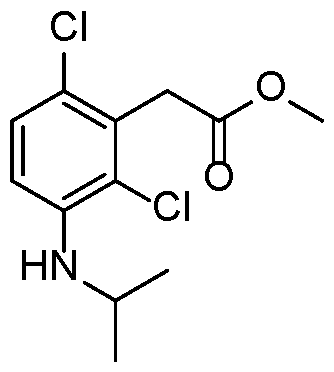
- Step 3 methyl 2-(3-amino-2,6-dichlorophenyl)acetate
- THF THF
- ammonium chloride 614 mg, 11.5 mmol
- zinc 1.00 g, 8, 15.3 mmol
- the reaction was stirred at RT for 16 h.
- the mixture was filtered through Celite washing with EtOAc (30 mL). The filtrate was diluted with water (40 mL) and EtOAc (20 mL) and transferred to a separating funnel.
- Step 4 methyl 2-(2,6-dichloro-3-(methylsulfonamido)phenyl)acetate
- methyl 2-(3-amino-2,6-dichlorophenyl)acetate 300 mg, 1.282 15 mmol
- DCM dimethyl sulfoxide
- triethylamine 0.39 mL, 2.82 mmol
- methanesulfonyl chloride 0.22 mL, 2.82 mmol
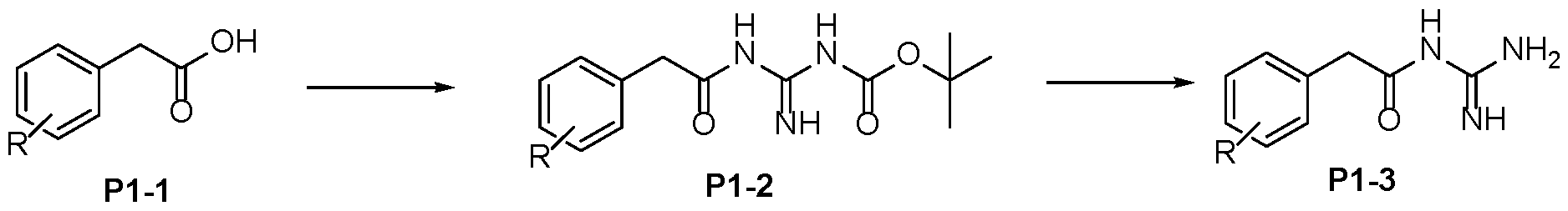
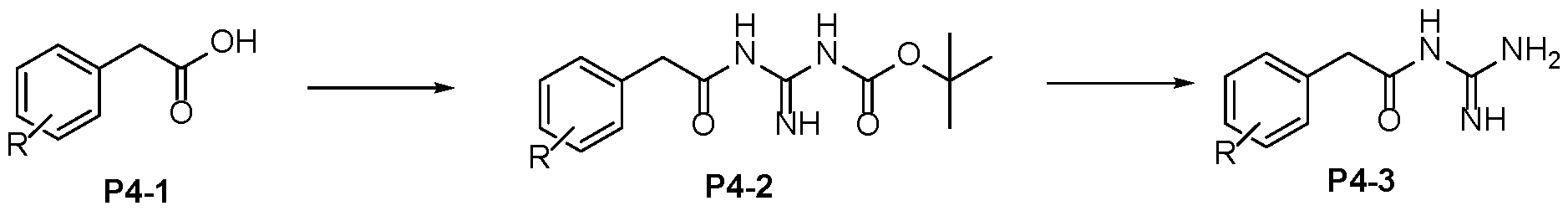
- Step 2 To a stirred solution of carboxylic acid P3-2 in DMF (0.1 M) was added N-Boc guanidine (1.1- 1.2 eq), DIPEA (2-3 eq) and HATU (1.4-1.5 eq) at RT under a nitrogen atmosphere. After reaction completion (LCMS), the reaction mixture was quenched with saturated sodium bicarbonate solution. The reaction mixture was extracted with EtOAc and the organic layer 5 dried over anhydrous MgSO4, filtered and concentrated under reduced pressure. The crude obtained was purified by column chromatography or used directly for subsequent chemistry. Alternatively, the reaction was quenched with water and the product isolated directly by filtration to afford P3-3.
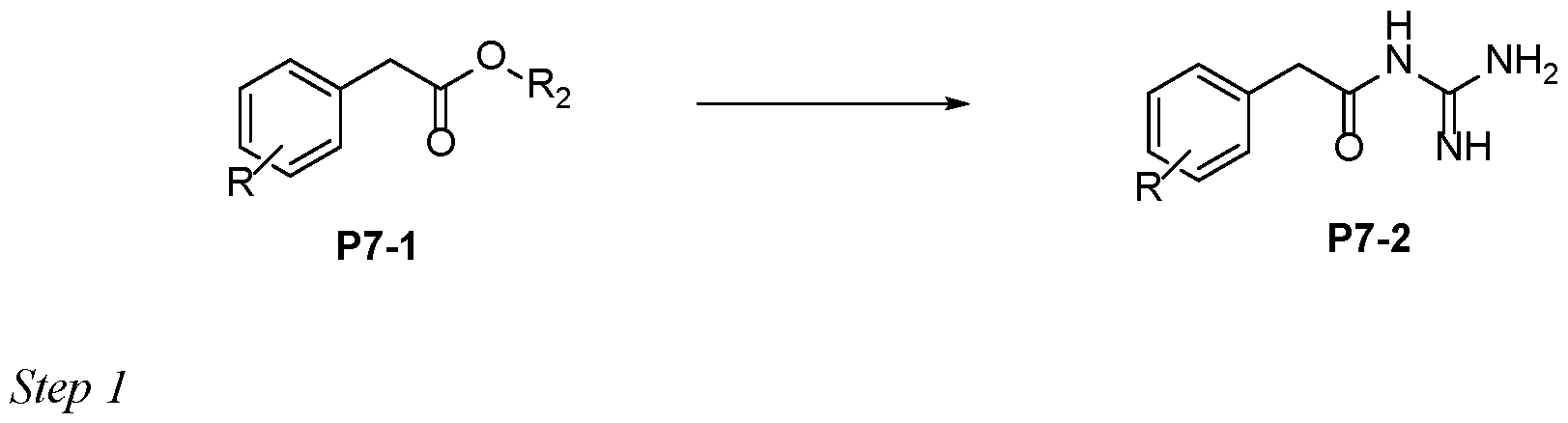
- Step 3 10 To a stirred solution of Boc-protected-acyl-guanidine P3-3 in dichloromethane (20-50 vol) was added Trifluoroacetic acid (10-20 eq). After reaction completion (LCMS), the reaction mixture was concentrated under reduced pressure. The crude product could be further purified by trituration or preparative HPLC to afford P3-4 15 Protocol 4 Step 1 To a stirred solution of carboxylic acid P4-1 in DMF (0.1 M) was added N-Boc guanidine (1.1- 20 1.2 eq), DIPEA (2-3 eq) and HATU (1.4-1.5 eq) at RT under a nitrogen atmosphere. After reaction completion (LCMS), the reaction mixture was quenched with saturated sodium bicarbonate solution.
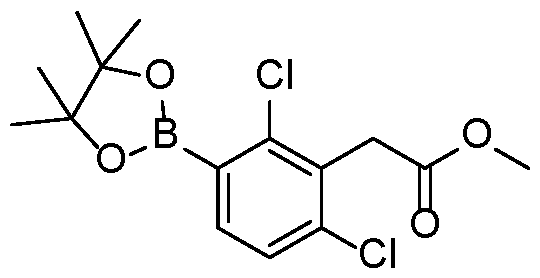
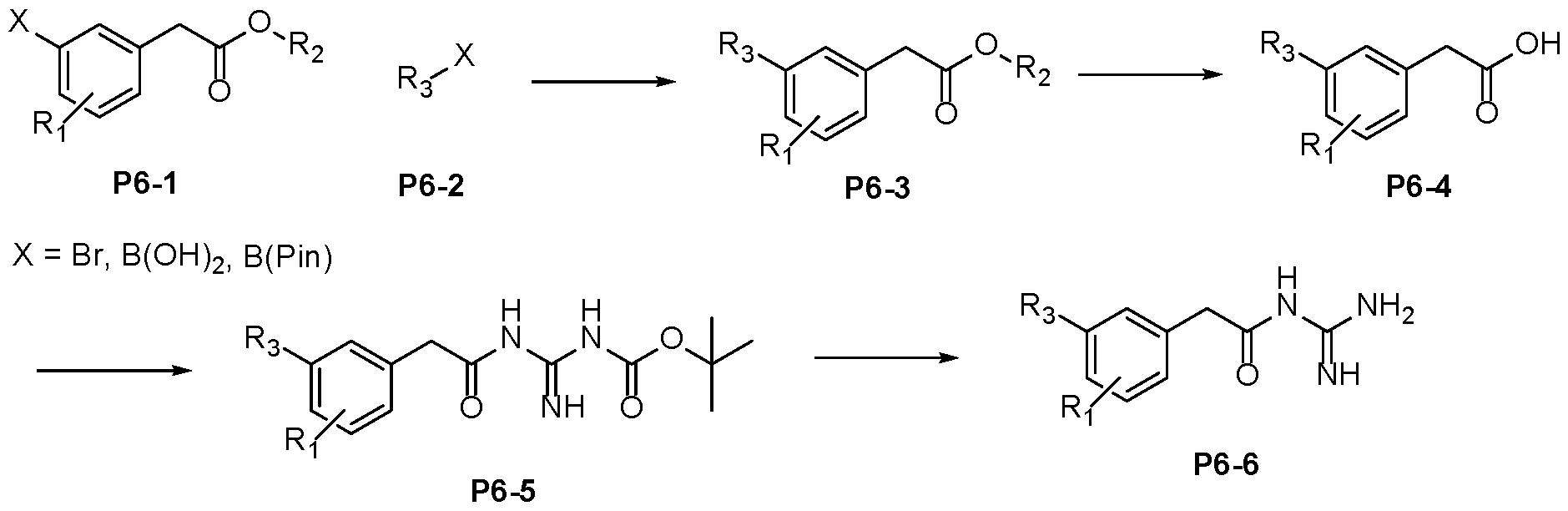
- Protocol 5 Step 1 To a solution of bromide P5-1 (1 eq), boronate P5-2 (1.5 eq) and caesium or potassium 5 carbonate (3 eq) in Acetonitrile/Water (4:1 ratio) was added SPhos Pd G3 (10 mol%). Alternatively, PdCl2dppf or Pd(PPh3)Cl2 was used. The reaction mixture was degassed with nitrogen and heated to 80 °C under nitrogen. After the reaction completion (LCMS), the mixture was allowed to cool and subjected to aqueous work up.
- Step 3 To a stirred solution of carboxylic acid P6-4 in DMF (0.1 M) was added N-Boc guanidine (1.1- 1.2 eq), DIPEA (2-3 eq) and HATU (1.4-1.5 eq) at RT under a nitrogen atmosphere. After reaction completion (LCMS), the reaction mixture was quenched with saturated sodium 20 bicarbonate solution. The reaction mixture was extracted with EtOAc and the organic layer dried over anhydrous MgSO 4 , filtered and concentrated under reduced pressure. The crude obtained was purified by column chromatography or used directly for subsequent chemistry. Alternatively, the reaction was quenched with water and the product isolated directly by filtration to afford P6-5.
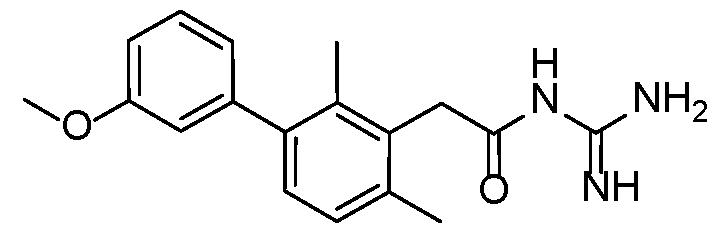
- Example 2 N-carbamimidoyl-2-(2,4-dimethyl-[1,1'-biphenyl]-3-yl)acetamide.TFA 5
- the title compound was prepared from Intermediate 2 using Protocol 2 and phenylboronic acid.
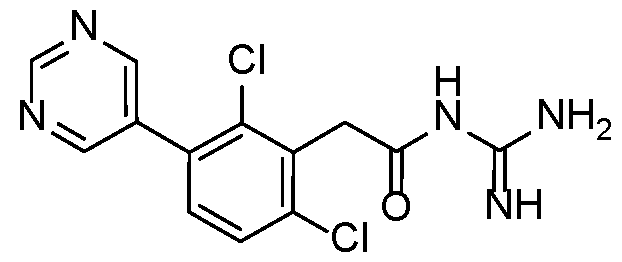
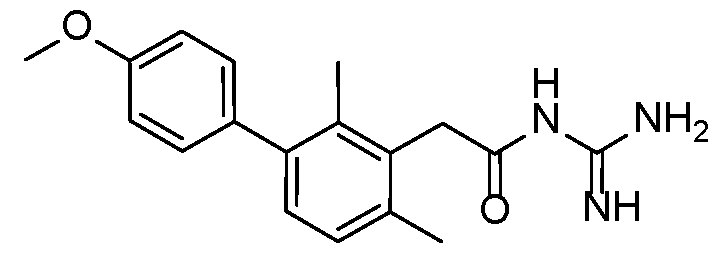
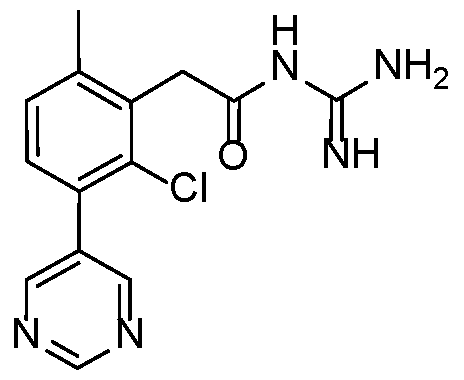
- Example 3 N-carbamimidoyl-2-(2,6-dimethyl-3-(pyrimidin-5-yl)phenyl)acetamide.TFA 15
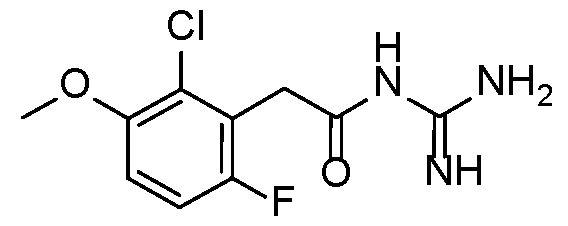
- Example 8 N-carbamimidoyl-2-(2-chloro-6-fluoro-3-methoxyphenyl)acetamide.TFA 5
- the title compound was prepared from 2-chloro-6-fluoro-3-methoxyphenyl)acetic acid using Protocol 1. m/z: 260.1 @ 0.81 min, Method 2.
- Example 9 N-carbamimidoyl-2-(6-chloro-2-fluoro-3-methoxyphenyl)acetamide.
- Example 10 N-carbamimidoyl-2-(2,6-difluoro-3-methoxyphenyl)acetamide.HCl 25
- the title compound was prepared from 2-(2,6-difluoro-3-methoxyphenyl)acetic acid using Protocol 4. m/z: 243.9 @ 0.74 min, Method 2.
- Example 11 2-(3-butoxy-2,6-difluorophenyl)-N-carbamimidoylacetamide.HCl
- the title compound was prepared from 2-(3-butoxy-2,6-difluorophenyl)acetic acid using 5 Protocol 4.
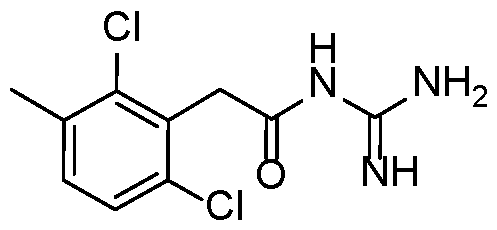
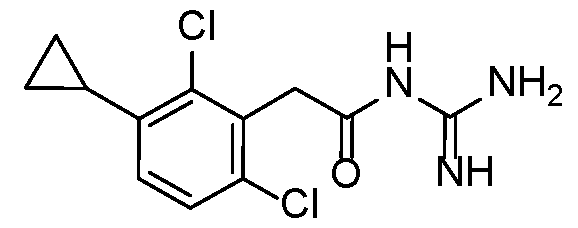
- Example 12 N-carbamimidoyl-2-(2,6-dichloro-3-methylphenyl)acetamide.TFA
- the title compound was prepared from Intermediate 5 using Protocol 5 with trimethylboroxine. 15
- Example 13 N-carbamimidoyl-2-(2,6-dimethyl-3-(pyrimidin-2-yl)phenyl)acetamide.TFA 20
- Example 14 N-carbamimidoyl-2-(2,6-dimethyl-3-(pyridin-2-yl)phenyl)acetamide.TFA 30
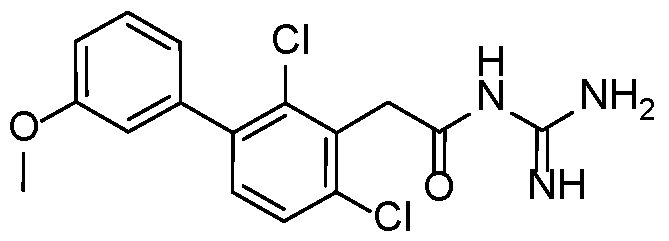
- Example 15 N-carbamimidoyl-2-(2,4-dichloro-3'-methoxy-[1,1'-biphenyl]-3-yl)acetamide.
- HCOOH 10 The title compound was prepared from Intermediate 5 using Protocol 5 with (3- methoxyphenyl)boronic acid and PdCl 2 dppf in the Suzuki coupling.
- Example 16 N-carbamimidoyl-2-(2,4-dichloro-3'-cyano-[1,1'-biphenyl]-3-yl)acetamide.
- HCOOH 20 The title compound was prepared from Intermediate 5 using Protocol 5 with (3- cyanophenyl)boronic acid and PdCl2dppf in the Suzuki coupling.
- Example 17 N-carbamimidoyl-2-(2,3',4-trichloro-4'-ethoxy-[1,1'-biphenyl]-3- yl)acetamide.TFA 30
- the title compound was prepared from Intermediate 5 using Protocol 6 with (3-chloro-4- ethoxyphenyl)boronic acid and PdCl 2 dppf in the Suzuki coupling.
- Example 19 2-(3-acetamido-2,6-dichlorophenyl)-N-carbamimidoylacetamide.TFA 20
- Example 20 N-carbamimidoyl-2-(2,6-dichloro-3-(isopropylamino)phenyl)acetamide.
- Example 22 N-carbamimidoyl-2-(2,4-dichloro-4'-methoxy-[1,1'-biphenyl]-3-yl)acetamide.
- Example 23 N-carbamimidoyl-2-(2,6-dichloro-3-(dimethylamino)phenyl)acetamide. HCOOH 30
- Example 24 N-carbamimidoyl-2-(2,6-dichloro-3-(methylamino)phenyl)acetamide. 5 HCOOH
- Example 25 N-carbamimidoyl-2-(3'-methoxy-2,4-dimethyl-[1,1'-biphenyl]-3-yl)acetamide. 15 HCOOH 20
- Example 26 N-carbamimidoyl-2-(4'-methoxy-2,4-dimethyl-[1,1'-biphenyl]-3-yl)acetamide.
- Example 28 N-carbamimidoyl-2-(2,6-dichloro-3-(pyrimidin-2-yl)phenyl)acetamide.TFA 20
- Example 29 N-carbamimidoyl-2-(2,6-dichloro-3-(pyridin-4-yl)phenyl)acetamide.
- Example 31 N-carbamimidoyl-2-(2,6-dichloro-3-(3,5-dimethylisoxazol-4- yl)phenyl)acetamide.
- Example 32 N-carbamimidoyl-2-(4-chloro-3'-methoxy-2-methyl-[1,1'-biphenyl]-3- 30 yl)acetamide 5
- the title compound was prepared from Intermediate 7 using Protocol 5 with (3- methoxyphenyl)boronic acid and PdCl 2 dppf in the Suzuki coupling.
- TFA 15 The title compound was prepared from Intermediate 5 using Protocol 6 with tert-butyl 4- (4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-3,6-dihydropyridine-1(2H)-carboxylate and PdCl 2 dppf in the Suzuki coupling.
- Example 38 N-carbamimidoyl-2-(2-chloro-3'-methoxy-4-methyl-[1,1'-biphenyl]-3- yl)acetamide. HCOOH 10
- the title compound was prepared from Intermediate 15 using Protocol 5 with (3- methoxyphenyl)boronic acid and PdCl 2 dppf in the Suzuki coupling. m/z: 332.2 @ 1.31 min, Method 2.
- Example 42 N-carbamimidoyl-2-(2,6-dichloro-3-(1H-pyrazol-4-yl)phenyl)acetamide, HCl 25
- Example 43 N-carbamimidoyl-2-(2,6-dichloro-3-(4-cyano-1-methyl-1H-pyrazol-5- yl)phenyl)acetamide, HCl 5
- the title compound was prepared from Intermediate 12 using Protocol 5 with 5-bromo-1- methyl-1H-pyrazole-4-carbonitrile and Pd-170 (Johnson and Matthey) m/z: 351 @ 0.70 min, Method 3 10
- Example 44 N-carbamimidoyl-2-(6-chloro-2-cyano-3-cyclopropylphenyl)acetamide, HCl 15
- the title compound was prepared from
- HTRF GTP binding kit was purchased from Perkin Elmer (62GTPPEC). Methods Membrane preparations 10 Cells were detached in Versene for up to 15 minutes with agitation. All collected cells were centrifuged at 300 xg for 5 minutes (RT) and supernatants were discarded.
- Cell pellets were resuspended in ice-cold HE buffer: 20 mM HEPES (H0887, Merck), 10 mM EDTA (E7889, Merck), pH 7.4.
- the cell suspension was then homogenised using a Dounce homogeniser with 20x strokes of pestle A, followed by 20x strokes with pestle B. Homogenates were centrifuged 15 for 600 xg for 10 minutes at 4 °C. The supernatants were then transferred to high-speed centrifuge tubes (NC654, Appleton Woods).
- Pellets were again resuspended in ice-cold HE buffer: 20 mM HEPES (H0887, Merck), 10 mM EDTA (E7889, Merck), pH 7.4and homogenised using a Dounce homogeniser with 20x strokes of pestle A, followed by 20x strokes with pestle B, then centrifuged for 600 xg for 10 minutes at 4 °C. Supernatants were 20 pooled and centrifuged at 40,000xg for 1 h at 4 °C. Each pellet was resuspended in 400 ⁇ l of 20 mM HEPES with 10 % (w/v) glycerol (332031000, ThermoScientific). All steps were performed on ice.
- Membrane suspensions were passed 10x through the insulin syringe (324892, BD Microfine), aliquoted and stored at -80 °C until further use. Protein content was established using the BCA kit (23225, ThermoFisher) with bovine serum albumin standards. 25 HTRF GTP binding assay To evaluate the potential agonists of ⁇ 2 adrenergic receptors ( ⁇ 2A , ⁇ 2B and ⁇ 2C ) HTRF GTP binding kit was used with some modification to the manufacturer’s protocol. Tested compounds were dispensed into 384 white ProxiPlates Plus (6008280, Perkin Elmer) using an ECHO acoustic dispenser with automatic DMSO backfill.
- Guanfacine (HU-17416, Medchemtronica 30 AB) was used as a reference compound. All compounds were run in duplicates on each plate. For plate controls, 30 ⁇ M norepinephrine (N5785, Merck) and vehicle control (DMSO) were used. Then, the assay mix was prepared by mixing stimulation buffer #3, MgCl 2 , 1x GTP Eu Cryptate reagent, 1x GTP d2 antibody and membrane preparation. MgCl 2 concentration per well was 50 mM for ⁇ 2A and ⁇ 2C , 8 mM for ⁇ 2B . The amount of membrane proteins was established for each membrane preparation in a validation experiment (range 0.5-2 ⁇ g of protein per well).
- the compounds of the invention have to exhibit a limited, if any, CNS penetrance, in order to avoid deleterious side effects that can occur if these compounds penetrate significantly into the brain.
- the present assay aims at showing that the compounds of the invention do not have any 10 significant CNS penetrance by showing that they are substrates of transporters that efflux them from brain.
- Efflux ratio Papp (A-B)/Papp(B-A) where Papp (A-B) and Papp (B-A) are the Papp values of compound in Apical to Basolateral and Basolateral to Apical directional transport, respectively. 20 Results. Compounds are considered to be P-Glycoprotein substrates when the value of efflux ratio is > 3. Compounds of the present invention typically have efflux ratios > 3 whereas guanfacine has an efflux ratio ⁇ 3 (1.5), as evidenced in Table 3 below. 25 Table 3: III.2.
- the present assay aims at showing that the compounds of the invention do not have any 5 significant CNS penetrance by determining the concentration of these compounds in brain and cerebrospinal fluid (CSF) compared to plasma.
- Method. 7-9 weems old female Balb-c mice (obtained from SLAC Laboratory Animal Co. Ltd., Shanghai, China or SIPPR-B&K Laboratory Animal Co.
- Example 7, 42, and 43 of the invention were dosed at 5 mg/kg by oral route and concentrations in brain or CSF of mice were found to be significantly lower compared to the concentrations present in plasma (brain/plasma ratio ⁇ 0.1).
- Individual and Mean Concentration of Example 7 after PO Administration (5 mg/kg) are provided in Table 4 below.
- Table 5 Individual and Mean Concentration of Example 43 after PO Administration (5 mg/kg) are provided in Table 6 below.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Pharmacology & Pharmacy (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
The present invention relates to guanfacine derivatives of formula (I), and the isomers and pharmaceutical acceptable salts thereof. The invention further relates to pharmaceutical compositions comprising such compounds of formula (I) and their use for treating a cancer.
Description
GUANFACINE DERIVATIVES AND THEIR USES FIELD OF THE INVENTION The present invention relates to the field of medicine, in particular guanfacine derivatives and their uses for treating diseases. More particularly, the invention provides alpha2-adrenergic receptor (α2-AR) agonists for the treatment of cancer. BACKGROUND OF THE INVENTION Cancer is the second leading cause of death worldwide. Indeed, nearly 1 in 6 deaths is due to cancer. The prevalence of cancer is also extremely high as more than 15 million new cases are diagnosed each year, and the number of new cases is expected to rise by about 70% over the next 2 decades. Among the most common cancers, lung cancers account for 1.69 million deaths per year, colorectal cancer for 774000 deaths per year, and breast cancer for 571000 deaths per year. Many treatment options exist nowadays for cancer, including for example surgery, chemotherapy, radiation therapy, hormonal therapy, targeted therapy, immunotherapy and palliative care. The choice of the best treatments depends on the type, location and grade of the cancer as well as the patient's health and preferences. The α2-AR are a family of G-protein–coupled receptors with 3 pharmacological subtypes, α2A, α2B, and α2C. They have been used for decades to treat common medical conditions such as hypertension; attention-deficit/hyperactivity disorder; various pain and panic disorders; symptoms of opioid, and alcohol withdrawal; and for sedation and to reduce anesthetic requirements. Centrally acting alpha-2 adrenergic receptors agonists stimulate α2-AR in the central nervous system (brain and spinal cord). When α2-AR are stimulated, sympathetic nervous system activity decreases. This decreased sympathetic activity leads to a drop in blood pressure and heart rate. WO 2021/214129 and J. Zhu, et al. (Nature, 618, 607–615 (2023)) have currently shown that agonists of α2-AR, such as Guanabenz, Clonidine or Guanfacine, have surprisingly strong anti- tumor activity when used as monotherapies in multiple immunocompetent murine tumor models, including ICB-resistant models, but not in immunodeficient models. It was also observed striking effects in human tumor xenografts implanted in mice reconstituted with
human peripheral blood mononuclear cells. The anti-tumor effects of α2-AR agonists were reverted by α2-AR antagonists, and were absent in α2-AR knockout mice, demonstrating on- target action exerted on host cells, not tumor cells. Tumors from treated mice contained increased infiltrating CD8+ T lymphocytes and reduced infiltrating myeloid-derived suppressor cells (MDSC), which were more apoptotic. Single-cell RNA sequencing revealed upregulation of innate and adaptive immune response pathways in macrophages and T cells. Consequently, the discovery and the synthesis of new α2-AR agonists are relevant and promising approaches for anti-cancer therapies. More particularly, it could significantly improve clinical efficacy of cancer immunotherapy. However, many of the effects of centrally active α2-AR agonists are mediated by decreasing sympathetic activity and lead to side hypotensive and sedative effects that could be harmful to patients and/or limit the dose of α2- AR agonists that could be safely administered to patients, thereby potentially limiting the therapeutic anti-cancer benefit. Therefore, there remains a need to identify further α2-AR agonists having an efficient effect on α-2 AR while limited CNS penetrance. The development of new peripherally restricted α2-AR agonists could be of interest as anti-cancer therapies and, more specifically anti-cancer immunotherapies. The present invention seeks to meet these and other needs. SUMMARY OF THE INVENTION In this context, the inventors have provided new guanfacine derivatives as efficient α2-AR agonists while being not efficient in penetrating the BBB, demonstrating thereby the therapeutic interest of such derivatives in medicine, more particularly in anti-cancer therapies. The present invention thus provides new compounds of formula (I):
wherein:
^ R1 represents a radical selected in a group consisting of: • a hydrogen, • a (C1-C6)alkyl optionally substituted by a (C1-C6)alkyloxy, a hydroxy, at least one halogen, and a cycloalkyl, • a halogen, • a cyano, • a 3-14 membered ring, saturated, partially unsaturated, or unsaturated selected in a group consisting of a cycloalkyl, a heterocycloalkyl, an aryl, and an heteroaryl, said 3-14 membered ring is optionally substituted by a (C1-C6)alkyl, a hydroxy, a halogen, a -SO2-(C1-C6)alkyl, and a (C1-C6)alkyloxy, and • a (C1-C6)alkyloxy optionally substituted by at least one halogen; ^ R2 represents a radical selected in a group consisting of: • a hydrogen, • a (C1-C6)alkyl optionally substituted by a (C1-C6)alkyloxy, a hydroxy, at least one halogen, and a cycloalkyl, • a halogen, • a cyano, • a 3-14 membered ring, saturated, partially unsaturated, or unsaturated selected in a group consisting of a cycloalkyl, a heterocycloalkyl, an aryl, and an heteroaryl, said 3-14 membered ring is optionally substituted by a (C1-C6)alkyl, a hydroxy, a halogen, a -SO2-(C1-C6)alkyl, and a (C1-C6)alkyloxy, and • a (C1-C6)alkyloxy optionally substituted by at least one halogen; ^ X is selected from the group consisting of -(CH2)m- with m is from 1 to 3 optionally substituted by at least one Ra, -O-, -S-, -CO-, -C(O)-O-, -O-C(O)-, -NRa-, -CO-NRa-, - NRa-CO-, -SO-, -SO2-, -SO2-NRa, -NRa-SO2-, and -NRa-CO-NRa-, with Ra being a hydrogen, a (C1-C6)alkyl optionally substituted by at least one radical C, a cycloalkyl optionally substituted by at least one radical C, or a heterocycloalkyl optionally substituted by at least one radical C; said radical C being selected in a group consisting of: - a (C1-C6)alkyloxy, - a halogen, - a -O-C(O)-(C1-C6)alkyl, a -C(O)-O-(C1-C6)alkyl, a - COOH,
- a -O-C(O)-cycloalkyl, a -C(O)-O-cycloalkyl, - a -CO-(C1-C6)alkyl, - a -CO-cycloalkyl, - a hydroxy, - a -NRbRc with Rb and Rc represent independently a hydrogen, a (C1-C6)alkyl, a cycloalkyl, or Rb and Rc may form together a N-heterocycloalkyl, - a nitro, - a cyano, - a -NH-CO-(C1-C6)alkyl, a -CO-NH-(C1-C6)alkyl, - a -NH-CO-cycloalkyl, a -CO-NH-cycloalkyl, - a -NH-SO2-(C1-C6)alkyl, a -SO2-NH-(C1-C6)alkyl, a - NH-SO2-cycloalkyl, a -SO2-NH-cycloalkyl, - a -SO2-(C1-C6)alkyl, a -SO2-cycloalkyl, - a -SO-(C1-C6)alkyl, and a -SO-cycloalkyl; ^ n is 0 or 1; ^ R3 represents a radical selected in a group consisting of: • a 3-14 membered ring, saturated, partially unsaturated, or unsaturated, selected in a group consisting of an aryl, a heteroaryl, a cycloalkyl, and a heterocycloalkyl, said 3-14 membered ring is optionally substituted by at least one radical A selected in a group consisting of: - a (C1-C6)alkyl optionally substituted by a halogen, a (C1-C6)alkyloxy, or a hydroxy, - a cycloalkyl optionally substituted by a halogen, a (C1- C6)alkyloxy, a hydroxy, or a -CO-NH2, - a heterocycloalkyl optionally substituted by a halogen, a (C1-C6)alkyloxy, or a hydroxy, - a (C2-C6)alkenyl, - a (C2-C6)alkynyl, - a (C1-C6)alkyloxy optionally substituted by a halogen, - a halogen, - a cyano,
- a -NRdRe with Rd and Re represent independently a hydrogen, a (C1-C6)alkyl, a cycloalkyl, or Rd and Re may form together a N-heterocycloalkyl, - a nitro, - a hydroxy, - a ketone, - a -NH-CO-(C1-C6)alkyl, a -CO-NH-(C1-C6)alkyl, a - CO-N((C1-C6)alkyl)2, a -NH-CO-cycloalkyl, a -CO- NH-cycloalkyl, - a -NH-SO2-(C1-C6)alkyl, a -SO2-NH-(C1-C6)alkyl, a - NH-SO2-cycloalkyl, a -SO2-NH-cycloalkyl, - a -C(O)-O-(C1-C6)alkyl, a -O-C(O)-(C1-C6)alkyl, a - COOH, a -C(O)-O-cycloalkyl, -O-C(O)-cycloalkyl, - a -CO-(C1-C6)alkyl, a -CO-cycloalkyl, - a -SO2-(C1-C6)alkyl, a -SO2-cycloalkyl, - a -SO-(C1-C6)alkyl, and a -SO-cycloalkyl, and • a radical selected in a group consisting of: o a (C1-C6)alkyl optionally substituted by at least one radical B or a cycloalkyl optionally substituted by at least one radical B, o a (C2-C6)alkenyl optionally substituted by at least one radical B, o a (C2-C6)alkynyl optionally substituted by at least one radical B, o a (C1-C6)alkyloxy optionally substituted by at least one radical B, o a hydroxy, o a cyano, o a halogen, o a -NRf’Rg’ with Rf’ and Rg’, represent independently a hydrogen, a (C1-C6)alkyl optionally substituted by at least one radical B, a cycloalkyl optionally substituted by at least one radical B, a heterocycloalkyl optionally substituted by at least one radical B, with the proviso that when one of Rf’ or Rg’ is a hydrogen, then the other is not a hydrogen, or Rf’ and Rg’ may form together a N- heterocycloalkyl, and o a -N(Rf)-CO-Rg, a-CO-N(Rf)-Rg, a -CO-Rf, a -C(O)-O-Rf, a -O- C(O)-Rf, a -SO2-Rf, a -SO-Rf, a -SO2-N(Rf)-Rg, a -N(Rf)-SO2-Rg,
and a -N(Rh)-CO-NRfRg, with Rf, Rg, and Rh represent independently a hydrogen, a (C1-C6)alkyl optionally substituted by at least one radical B, a cycloalkyl optionally substituted by at least one radical B, a heterocycloalkyl optionally substituted by at least one radical B, or Rf and Rg may form together a N- heterocycloalkyl, said radical B being selected in a group consisting of: - a (C1-C6)alkyloxy, - a (C1-C6)alkyl optionally substituted by a hydroxy, - a halogen, - a -O-C(O)-(C1-C6)alkyl optionally substituted by at least one halogen, a -C(O)-O-(C1-C6)alkyl, a -COOH, - a -O-C(O)-cycloalkyl, a -C(O)-O-cycloalkyl, - a -CO-(C1-C6)alkyl, - a cycloalkyl, - a heterocycloalkyl, - a -CO-cycloalkyl, - a hydroxy, - a -NRiRj with Ri and Rj represent independently a hydrogen, a (C1-C6)alkyl, a cycloalkyl, or Ri and Rj may form together a N-heterocycloalkyl, - a nitro, - a cyano, - an aryl, - a heteroaryl, - a -NH-CO-(C1-C6)alkyl, a -CO-NH-(C1-C6)alkyl, - a -NH-CO-cycloalkyl; -CO-NH-cycloalkyl, - a -NH-SO2-(C1-C6)alkyl, a -SO2-NH-(C1-C6)alkyl, - a -NH-SO2-cycloalkyl, a -SO2-NH-cycloalkyl, - a -SO2-(C1-C6)alkyl, a -SO2-cycloalkyl, - a -SO-(C1-C6)alkyl, and a -SO-cycloalkyl; ^ R4 represents a radical selected in a group consisting of: • a hydrogen, • a halogen,
• a (C1-C6)alkyl optionally substituted by a halogen, a (C1-C6)alkyloxy, a hydroxy, or a cycloalkyl, • a (C1-C6)alkyloxy optionally substituted by a halogen, a (C1-C6)alkyloxy, a hydroxy, or a cycloalkyl, • a hydroxy, • a -NRkRl with Rk and Rl represent independently a hydrogen, a (C1-C6)alkyl, a cycloalkyl, or Rk and Rl may form a N-heterocycloalkyl, • a nitro, • a cyano, • a -NH-(C1-C6)alkyl, a -NH-cycloalkyl; and • a -NH-CO-(C1-C6)alkyl, and a -NH-CO-cycloalkyl; ^ R5 represents a radical selected in a group consisting of: • a hydrogen, • a halogen, • a (C1-C6)alkyl optionally substituted by a halogen, a (C1-C6)alkyloxy, a hydroxy, or a cycloalkyl, • a (C1-C6)alkyloxy optionally substituted by a halogen, a (C1-C6)alkyloxy, a hydroxy, or a cycloalkyl, • a hydroxy, • a -NRkRl with Rk and Rl represent independently a hydrogen, a (C1-C6)alkyl, a cycloalkyl, or Rk and Rl may form a N-heterocycloalkyl, • a nitro, • a cyano, • a -NH-(C1-C6)alkyl, a -NH-cycloalkyl; and • a -NH-CO-(C1-C6)alkyl, and a -NH-CO-cycloalkyl; ^ R6 represents a hydrogen, or a (C1-C6)alkyl optionally substituted by a radical selected in a group consisting of a hydroxy, a (C1-C6)alkyloxy, a -NH(C1-C6)alkyl, and a -N((C1- C6)alkyl)2; ^ R7 represents a hydrogen, or a (C1-C6)alkyl optionally substituted by a radical selected in a group consisting of a hydroxy, a (C1-C6)alkyloxy, a -NH(C1-C6)alkyl, and a -N((C1- C6)alkyl)2; and
^ R8 represents a hydrogen, or a (C1-C6)alkyl optionally substituted by a radical selected in a group consisting of a hydroxy, a (C1-C6)alkyloxy, a -NH(C1-C6)alkyl, and a -N((C1- C6)alkyl)2; or R7 and R8 may form together a 5-7 membered ring, partially unsaturated or saturated, N,N’-heterocycloalkyl optionally substituted by at least one radical selected in a group consisting of a (C1-C6)alkyl, a hydroxy, a (C1-C6)alkyloxy, a -NH(C1-C6)alkyl, a - N((C1-C6)alkyl)2, and a ketone; and the isomers, stereoisomers and pharmaceutical acceptable salts thereof. Particularly, the present invention provides new compounds of formula (I):
wherein: ^ R1 represents a radical selected in a group consisting of: • a hydrogen, • a (C1-C6)alkyl optionally substituted by a (C1-C6)alkyloxy, a hydroxy, at least one halogen, and a cycloalkyl, • a halogen, • a 3-14 membered ring, saturated, partially unsaturated, or unsaturated selected in a group consisting of a cycloalkyl, a heterocycloalkyl, an aryl, and an heteroaryl, said 3-14 membered ring is optionally substituted by a (C1-C6)alkyl, a hydroxy, a halogen, a -SO2-(C1-C6)alkyl, and a (C1-C6)alkyloxy, and • a (C1-C6)alkyloxy optionally substituted by at least one halogen; ^ R2 represents a radical selected in a group consisting of: • a hydrogen, • a (C1-C6)alkyl optionally substituted by a (C1-C6)alkyloxy, a hydroxy, at least one halogen, and a cycloalkyl, • a halogen, • a 3-14 membered ring, saturated, partially unsaturated, or unsaturated selected in a group consisting of a cycloalkyl, a heterocycloalkyl, an aryl, and an
heteroaryl, said 3-14 membered ring is optionally substituted by a (C1-C6)alkyl, a hydroxy, a halogen, a -SO2-(C1-C6)alkyl, and a (C1-C6)alkyloxy, and • a (C1-C6)alkyloxy optionally substituted by at least one halogen; ^ X is selected from the group consisting of -(CH2)m- with m is from 1 to 3 optionally substituted by at least one Ra, -O-, -S-, -CO-, -C(O)-O-, -O-C(O)-, -NRa-, -CO-NRa-, - NRa-CO-, -SO-, -SO2-, -SO2-NRa, -NRa-SO2-, and -NRa-CO-NRa-, with Ra being a hydrogen, a (C1-C6)alkyl optionally substituted by at least one radical C, a cycloalkyl optionally substituted by at least one radical C, or a heterocycloalkyl optionally substituted by at least one radical C; said radical C being selected in a group consisting of: - a (C1-C6)alkyloxy, - a halogen, - a -O-C(O)-(C1-C6)alkyl, a -C(O)-O-(C1-C6)alkyl, a - COOH, - a -O-C(O)-cycloalkyl, a -C(O)-O-cycloalkyl, - a -CO-(C1-C6)alkyl, - a -CO-cycloalkyl, - a hydroxy, - a -NRbRc with Rb and Rc represent independently a hydrogen, a (C1-C6)alkyl, a cycloalkyl, or Rb and Rc may form together a N-heterocycloalkyl, - a nitro, - a cyano, - a -NH-CO-(C1-C6)alkyl, a -CO-NH-(C1-C6)alkyl, - a -NH-CO-cycloalkyl, a -CO-NH-cycloalkyl, - a -NH-SO2-(C1-C6)alkyl, a -SO2-NH-(C1-C6)alkyl, a - NH-SO2-cycloalkyl, a -SO2-NH-cycloalkyl, - a -SO2-(C1-C6)alkyl, a -SO2-cycloalkyl, - a -SO-(C1-C6)alkyl, and a -SO-cycloalkyl; ^ n is 0 or 1; ^ R3 represents a radical selected in a group consisting of: • a 3-14 membered ring, saturated, partially unsaturated, or unsaturated, selected in a group consisting of an aryl, a heteroaryl, a cycloalkyl, and a
heterocycloalkyl, said 3-14 membered ring is optionally substituted by at least one radical A selected in a group consisting of: - a (C1-C6)alkyl optionally substituted by a halogen, a (C1-C6)alkyloxy, or a hydroxy, - a cycloalkyl optionally substituted by a halogen, a (C1- C6)alkyloxy, or a hydroxy, - a (C2-C6)alkenyl, - a (C2-C6)alkynyl, - a (C1-C6)alkyloxy optionally substituted by a halogen, - a halogen, - a cyano, - a -NRdRe with Rd and Re represent independently a hydrogen, a (C1-C6)alkyl, a cycloalkyl, or Rd and Re may form together a N-heterocycloalkyl, - a nitro, - a hydroxy, - a ketone, - a -NH-CO-(C1-C6)alkyl, a -CO-NH-(C1-C6)alkyl, a - NH-CO-cycloalkyl, a -CO-NH-cycloalkyl, - a -NH-SO2-(C1-C6)alkyl, a -SO2-NH-(C1-C6)alkyl, a - NH-SO2-cycloalkyl, a -SO2-NH-cycloalkyl, - a -C(O)-O-(C1-C6)alkyl, a -O-C(O)-(C1-C6)alkyl, a - COOH, a -C(O)-O-cycloalkyl, -O-C(O)-cycloalkyl, - a -CO-(C1-C6)alkyl, a -CO-cycloalkyl, - a -SO2-(C1-C6)alkyl, a -SO2-cycloalkyl, - a -SO-(C1-C6)alkyl, and a -SO-cycloalkyl, and • a radical selected in a group consisting of: o a (C1-C6)alkyl optionally substituted by at least one radical B or a cycloalkyl optionally substituted by at least one radical B, o a (C2-C6)alkenyl optionally substituted by at least one radical B, o a (C2-C6)alkynyl optionally substituted by at least one radical B, o a (C1-C6)alkyloxy optionally substituted by at least one radical B, o a cyano,
o a -NRf’Rg’ with Rf’ and Rg’, represent independently a hydrogen, a (C1-C6)alkyl optionally substituted by at least one radical B, a cycloalkyl optionally substituted by at least one radical B, a heterocycloalkyl optionally substituted by at least one radical B, with the proviso that when one of Rf’ or Rg’ is a hydrogen, then the other is not a hydrogen, or Rf’ and Rg’ may form together a N- heterocycloalkyl, and o a -N(Rf)-CO-Rg, a-CO-N(Rf)-Rg, a -CO-Rf, a -C(O)-O-Rf, a -O- C(O)-Rf, a -SO2-Rf, a -SO-Rf, a -SO2-N(Rf)-Rg, a -N(Rf)-SO2-Rg, and a -N(Rh)-CO-NRfRg, with Rf, Rg, and Rh represent independently a hydrogen, a (C1-C6)alkyl optionally substituted by at least one radical B, a cycloalkyl optionally substituted by at least one radical B, a heterocycloalkyl optionally substituted by at least one radical B, or Rf and Rg may form together a N- heterocycloalkyl, said radical B being selected in a group consisting of: - a (C1-C6)alkyloxy, - a halogen, - a -O-C(O)-(C1-C6)alkyl, a -C(O)-O-(C1-C6)alkyl, a - COOH, - a -O-C(O)-cycloalkyl, a -C(O)-O-cycloalkyl, - a -CO-(C1-C6)alkyl, - a -CO-cycloalkyl, - a hydroxy, - a -NRiRj with Ri and Rj represent independently a hydrogen, a (C1-C6)alkyl, a cycloalkyl, or Ri and Rj may form together a N-heterocycloalkyl, - a nitro, - a cyano, - a -NH-CO-(C1-C6)alkyl, a -CO-NH-(C1-C6)alkyl, - a -NH-CO-cycloalkyl; -CO-NH-cycloalkyl, - a -NH-SO2-(C1-C6)alkyl, a -SO2-NH-(C1-C6)alkyl, - a -NH-SO2-cycloalkyl, a -SO2-NH-cycloalkyl, - a -SO2-(C1-C6)alkyl, a -SO2-cycloalkyl,
- a -SO-(C1-C6)alkyl, and a -SO-cycloalkyl; ^ R4 represents a radical selected in a group consisting of: • a hydrogen, • a halogen, • a (C1-C6)alkyl optionally substituted by a halogen, a (C1-C6)alkyloxy, a hydroxy, or a cycloalkyl, • a (C1-C6)alkyloxy optionally substituted by a halogen, a (C1-C6)alkyloxy, a hydroxy, or a cycloalkyl, • a hydroxy, • a -NRkRl with Rk and Rl represent independently a hydrogen, a (C1-C6)alkyl, a cycloalkyl, or Rk and Rl may form a N-heterocycloalkyl, • a nitro, • a cyano, • a -NH-(C1-C6)alkyl, a -NH-cycloalkyl; and • a -NH-CO-(C1-C6)alkyl, and a -NH-CO-cycloalkyl; ^ R5 represents a radical selected in a group consisting of: • a hydrogen, • a halogen, • a (C1-C6)alkyl optionally substituted by a halogen, a (C1-C6)alkyloxy, a hydroxy, or a cycloalkyl, • a (C1-C6)alkyloxy optionally substituted by a halogen, a (C1-C6)alkyloxy, a hydroxy, or a cycloalkyl, • a hydroxy, • a -NRkRl with Rk and Rl represent independently a hydrogen, a (C1-C6)alkyl, a cycloalkyl, or Rk and Rl may form a N-heterocycloalkyl, • a nitro, • a cyano, • a -NH-(C1-C6)alkyl, a -NH-cycloalkyl; and • a -NH-CO-(C1-C6)alkyl, and a -NH-CO-cycloalkyl; ^ R6 represents a hydrogen, or a (C1-C6)alkyl optionally substituted by a radical selected in a group consisting of a hydroxy, a (C1-C6)alkyloxy, a -NH(C1-C6)alkyl, and a -N((C1- C6)alkyl)2;
^ R7 represents a hydrogen, or a (C1-C6)alkyl optionally substituted by a radical selected in a group consisting of a hydroxy, a (C1-C6)alkyloxy, a -NH(C1-C6)alkyl, and a -N((C1- C6)alkyl)2; and ^ R8 represents a hydrogen, or a (C1-C6)alkyl optionally substituted by a radical selected in a group consisting of a hydroxy, a (C1-C6)alkyloxy, a -NH(C1-C6)alkyl, and a -N((C1- C6)alkyl)2; or R7 and R8 may form together a 5-7 membered ring, partially unsaturated or saturated, N,N’-heterocycloalkyl optionally substituted by at least one radical selected in a group consisting of a (C1-C6)alkyl, a hydroxy, a (C1-C6)alkyloxy, a -NH(C1-C6)alkyl, a - N((C1-C6)alkyl)2, and a ketone; and the isomers, stereoisomers and pharmaceutical acceptable salts thereof. In a particular embodiment, the compound of formula (I) is such that n is 0. In a further particular embodiment, the compound of formula (I) is such that R4 and R5 represent H. In a further particular embodiment, the compound of formula (I) is such that R6, R7, and R8 represent H. In a further particular embodiment, the compound of formula (I) is such that: ^ R1 represents a (C1-C6)alkyl, preferably a methyl, a halogen, preferably a chlorine or a fluorine, more preferably a chlorine; and ^ R2 represents a (C1-C6)alkyl, preferably a methyl, a halogen, preferably a chlorine or a fluorine, more preferably a chlorine, or a cyano. In a further particular embodiment, the compound of formula (I) is such that R3 represents a 3- 14 membered ring, saturated, partially unsaturated, or unsaturated, selected in a group consisting of an aryl, a heteroaryl, a cycloalkyl, and a heterocycloalkyl, said 3-14 membered ring is optionally substituted by at least one radical A selected in a group consisting of: - a (C1-C6)alkyl optionally substituted by a halogen, a hydroxy, a cycloalkyl, a -CO-NH((C1-C6)alkyl), or a - CO-N((C1-C6)alkyl)2,
- a heterocycloalkyl optionally substituted by a halogen, a (C1-C6)alkyloxy, or a hydroxy, - a cycloalkyl - a (C1-C6)alkyloxy optionally substituted by a halogen, - a halogen, - a -NRdRe with Rd and Re represent a hydrogen, a (C1- C6)alkyl, or a cyclopropyl, - a cyano, - a hydroxy, - a ketone, - a -CO-NH2, - a -COOH, - a -CO-(C1-C6)alkyl, - a -C(O)-O-(C1-C6)alkyl, - a -SO2-(C1-C6)alkyl, and - a -NH-CO-(C1-C6)alkyl. Particularly, R3 represents a 3-14 membered ring, saturated, partially unsaturated, or unsaturated, selected in a group consisting of: o an aryl, particularly a phenyl, said aryl is optionally substituted by at least one radical selected in a group consisting of: - a (C1-C6)alkyl, particularly a methyl, - a (C1-C6)alkyloxy, particularly a methoxy or an ethoxy, - a cyano, - a hydroxy, - a halogen, particularly a chlorine, - a -NRdRe with Rd and Re represent a hydrogen, and - a -NH-CO-(C1-C6)alkyl, particularly a -NH-CO-CH3, o a heteroaryl, particularly a pyrimidinyl, a pyrazinyl, a pyridazinyl, a pyrazolyl, an isoxazolyl, a pyridinyl, an indolyl, an imidazolyl, a triazolyl, an indazolyl, a thiazolyl, a thiophenyl, a benzoxazolyl, or a benzofuranyl, said heteroaryl is optionally substituted by at least one radical selected in a group consisting of:
- a (C1-C6)alkyl, particularly a methyl, an ethyl, an isopropyl, or a tert-butyl, said (C1-C6)alkyl is optionally substituted by at least one halogen, a hydroxy, or a cyclopropyl, - a (C1-C6)alkyloxy, particularly a methoxy, 5 - a cyano, - a halogen, particularly a chlorine, a bromine, or a iodine, - a -NRdRe with Rd and Re represent a hydrogen, - a tetrahydropyranyl, - a cyclopropyl, 10 - a hydroxy, and - a ketone, o a cycloalkyl, particularly a cyclopropyl, said cycloalkyl is optionally substituted by a -CO-NH2, a -COOH, a -C(O)-O-(C1-C6)alkyl, a (C1- C6)alkyl optionally substituted by a hydroxy, a -CO-CH3, a -CO- 15 NH(CH3), or a -CO-N(CH3)2, and o a heterocycloalkyl, particularly a 1,2,3-6-tetrahydropyridinyl, a morpholinyl, a pyrrolidinyl, an imidazolidinyl, a 3,4-dihydro-2H- pyranyl, a tetrahydropyranyl, a tetrahydrofuranyl, a morpholinyl, an azetidinyl, or an oxetanyl, said heterocycloalkyl is optionally substituted 20 by a -CO-(C1-C6)alkyl, particularly a -CO-CH3, a (C1-C6)alkyl optionally substituted by at least one halogen, particularly a methyl or a trifluromethyl, a (C1-C6)alkyloxy, particularly a methoxy, a halogen, particularly a fluorine, a -NRdRe with Rd and Re represent a hydrogen, a ketone, a -SO2-(C1-C6)alkyl, or a hydroxy.. 25 In a further particular embodiment, the compound of formula (I) is such that R3 represents a radical selected in a group consisting of: o a (C1-C6)alkyl, particularly a methyl, an ethyl, an isopropyl, or an isobutyl, said (C1-C6)alkyl is optionally substituted by a (C1-C6)alkyloxy, particularly a 30 methoxy, a hydroxy, at least one halogen, or a -NH-CO-(C1-C6)alkyl, particularly a -NH-CO-CH3, o a (C2-C6)alkenyl, particularly an isopropenyl, o a (C2-C6)alkynyl optionally substituted by a heteroaryl, particularly a pyrazolyl, or a (C1-C6)alkyl optionally substituted by a hydroxy,
o a (C1-C6)alkyloxy, particularly a methoxy, an ethoxy, an isopropoxy, or a butoxy, said (C1-C6)alkyloxy is optionally substituted by a phenyl, a hydroxy, or by at least one halogen, o a hydroxy, 5 o a phenoxy, o a halogen, particularly a chlorine or an iodine, o a cyano, o a -NRf’Rg’ with Rf’ and Rg’ represent independently a hydrogen or a (C1-C6)alkyl, particularly a methyl or an isopropyl, optionally substituted by a cycloalkyl or a 10 heterocycloalkyl, with the proviso that when one of Rf’ or Rg’ is a hydrogen, then the other is not a hydrogen, o a -NH-CO-(C1-C6)alkyl, particularly a -NH-CO-CH3, o a -NH-SO2-(C1-C6)alkyl, particularly a -NH-SO2-CH3, o a -SO2-Rf with Rf represents a (C1-C6)alkyl, particularly a methyl, and 15 o a -N(Rh)-CO-NRfRg, a -N(Rf)-CO-Rg, a-CO-N(Rf)-Rg, or a -N(Rf)-SO2-Rg, with Rf, Rg, and Rh represent independently a hydrogen, or a methyl. In a particular embodiment, the compound of the invention has the following formula: 20
5
, , , , ,
, , , , 5 ,
, , and , and a pharmaceutical acceptable salt thereof.
A further object of the invention is a compound of formula (I) as defined herein for use as a medicine. A further object of the invention is a pharmaceutical composition comprising a compound of formula (I) as defined herein and a pharmaceutically acceptable excipient. 5 A further object of the invention is a pharmaceutical composition as defined herein for use for treating a cancer. Preferably, the cancer is selected in a group consisting of myelofibrosis, acute lymphoblastic leukemia, acute myeloblastic leukemia adrenal gland carcinoma, bile duct cancer, bladder cancer, breast cancer, cervical cancer, colorectal cancer, endometrial cancer, esophageal cancer, gastric cancer, gastrointestinal stromal tumors, glioblastoma, head and neck 10 cancer, hepatocellular carcinoma, Hodgkin’s lymphoma, kidney cancer, lung cancer, melanoma, Merkel cell skin cancer, mesothelioma, multiple myeloma, myeloproliferative disorders, non-Hodgkin lymphoma, ovarian cancer, pancreatic cancer, prostate cancer, salivary gland cancer, sarcoma, squamous cell carcinoma, testicular cancer, thyroid cancer, urothelial carcinoma, and uveal melanoma. 15 In a particular embodiment, the pharmaceutical composition comprising a compound of formula (I) as defined herein is administered at a dose ranging from 0.001 mg/kg body weight to 30 mg/kg body weight. 20 In a further particular embodiment, the pharmaceutical composition is administered in combination with another antitumoral drug, especially chemotherapy, immunotherapy, hormonotherapy and/or radiotherapy, preferably immunotherapy. DETAILED DESCRIPTION OF THE INVENTION 25 Definitions According to the present invention, the terms below have the following meanings: The terms mentioned herein with prefixes such as for example C1-C6, can also be used with 30 lower numbers of carbon atoms such as C1-C2. If, for example, the term C1-C6 is used, it means that the corresponding hydrocarbon chain may comprise from 1 to 6 carbon atoms, especially 1, 2, 3, 4, 5, or 6 carbon atoms. If, for example, the term C1-C3 is used, it means that the corresponding hydrocarbon chain may comprise from 1 to 3 carbon atoms, especially 1, 2, or 3 carbon atoms.
The term “alkyl” refers to a saturated, linear or branched aliphatic group. The term “(C1- C6)alkyl” more specifically means methyl, ethyl, propyl, isopropyl, butyl, isobutyl, pentyl, isopentyl, or hexyl. The term “alkenyl” refers to an unsaturated, linear or branched aliphatic group comprising at 5 least one carbon-carbon double bound. The term “(C2-C6)alkenyl” more specifically means ethenyl, propenyl, isopropenyl, butenyl, isobutenyl, pentenyl, or hexenyl. The term “alkynyl” refers to an unsaturated, linear or branched aliphatic group comprising at least one carbon-carbon triple bound. The term “(C2-C6)alkynyl” more specifically means ethynyl, propynyl, isopropynyl, butynyl, isobutynyl, pentynyl, or hexynyl 10 The term “alkoxy” or “alkyloxy” corresponds to the alkyl group as above defined bonded to the molecule by an -O- (ether) bond. (C1-C6)alkoxy or (C1-C6)alkyloxy includes methoxy or methyloxy, ethoxy or ethyloxy, propoxy or propyloxy, isopropoxy or isopropyloxy, butoxy or butyloxy, isobutoxy or isobutyloxy, pentoxy or pentyloxy, isopentoxy or isopentyloxy, and hexoxy or hexyloxy. 15 The term “3-14 membered ring” corresponds to a ring having between 3 and 14 atoms. Such a term includes, for instance, the term “5-14 membered ring” having between 5 and 14 atoms, and the term “5-7 membered ring” having between 5 and 7 atoms. The term “ring” corresponds to a mono-, bi, or tricycle, which can be saturated, partially unsaturated or unsaturated, and optionally comprises at least one heteroatom. Particularly, the term “ring” includes a cycloalkyl, 20 a heterocycloalkyl, an aryl, and a heteroaryl. The term “cycloalkyl” corresponds to a saturated, partially unsaturated or unsaturated mono-, bi- or tri-cyclic alkyl group comprising between 3 and 14, preferably between 3 and 10 atoms of carbons. It also includes fused, bridged, or spiro-connected cycloalkyl groups. The term “cycloalkyl” includes for instance cyclopropyl, cyclobutyl, cyclopentyl, and cyclohexyl, 25 preferably cyclopropyl. The term “heterocycloalkyl” corresponds to a saturated, partially unsaturated or unsaturated cycloalkyl group as above defined further comprising at least one heteroatom such as nitrogen (N-heterocycloalkyl), oxygen (O-heterocycloalkyl), or sulphur atom (S-heterocycloalkyl). It also includes fused, bridged, or spiro-connected heterocycloalkyl groups. Representative 30 heterocycloalkyl groups include, but are not limited to dioxolanyl, benzo [1,3] dioxolyl, azetidinyl, oxetanyl, thiomorpholinyl, pyrazolidinyl, piperidyl, piperazinyl, 1,4-dioxanyl, pyrrolinyl, pyrrolidinyl, piperidinyl, imidazolidinyl, morpholinyl, 1,4-dithianyl, pyrrolidinyl, oxozolinyl, oxazolidinyl, isoxazolinyl, isoxazolidinyl, thiazolinyl, thiazolidinyl, isothiazolinyl,
isothiazolidinyl, tetrahydropyranyl, tetrahydrofuranyl, and tetrahydrothiophenyl. In a particular embodiment, the heterocycloalkyl group is morpholinyl. "Cycloalkyl" and "heterocycloalkyl" also include cycloalkenyl and heterocycloalkenyl which correspond respectively to a partially unsaturated cycloalkyl and a partially unsaturated 5 heterocycloalkyl such as cyclohexenyl, imidazolinyl, dihydropyranyl, for instance 3,6-dihydro- 2H-pyranyl and 3,4-dihydro-2H-pyranyl, pyrazolinyl, azetinyl, pyranyl, and tetrahydropyridinyl, for instance 1,2,3-6-tetrahydropyridinyl. The term “aryl” corresponds to a mono- or bi-cyclic aromatic hydrocarbons having from 6 to 12 carbon atoms. For instance, the term “aryl” includes phenyl, biphenyl, naphthyl and 10 anthracenyl. In a particular embodiment, the aryl is a phenyl. The term “heteroaryl” as used herein corresponds to an aromatic, mono- or poly-cyclic group comprising between 3 and 20 atoms and comprising at least one heteroatom such as nitrogen, oxygen or sulphur atom. As used herein, the term “heteroaryl” further includes the “fused arylheterocycloalkyl” and “fused heteroarylcycloalkyl”. The terms “fused 15 arylheterocycloalkyl” and “fused heteroarylcycloalkyl” correspond to a bicyclic group in which an aryl as above defined or a heteroaryl is respectively bounded to the heterocycloalkyl or the cycloalkyl as above defined by at least two carbons. In other terms, the aryl or the heteroaryl shares a carbon bond with the heterocycloalkyl or the cycloalkyl. Examples of such mono- and poly-cyclic heteroaryl group, fused arylheterocycloalkyl and fused arylcycloalkyl may be: 20 pyridinyl, thiophenyl, furanyl, pyrrolyl, pyrazolyl, imidazolyl, oxazolyl, isoxazolyl, thiazolyl, isothiazolyl, triazolyl, tetrazolyl, oxadiazolyl, furazanyl, thiadiazolyl, tetrazolyl, benzofuranyl, thianaphthalenyl, indolyl, indolinyl, indanyl, quinolinyl, isoquinolinyl, benzimidazolyl, tetrahydroquinolinyl, tetrahydroisoquinolinyl, triazinyl, thianthrenyl, benzofuranyl, dihydrobenzofuranyl, isobenzofuranyl, benzothiophenyl, isobenzothiophenyl, chromenyl, 25 xanthenyl, phenoxanthinyl, pyrazinyl, pyridazinyl, indolizinyl, isoindolyl, indazolyl, purinyl, quinolizinyl, phtalazinyl, naphthyridinyl, quinoxalinyl, quinazolinyl, cinnolinyl, pteridinyl, carbazolyl, β-carbolinyl, phenanthridinyl, acridinyl, pyrimidinyl, phenanthrolinyl, phenazinyl, phenothiazinyl, furazanyl, phenoxazinyl, isochromanyl, chromanyl, imidazolidinyl, imidazolinyl, pyrazolidinyl, pyrazolinyl, indolinyl, isoindolinyl, oxazolidinyl, benzotriazolyl, 30 benzoisoxazolyl, oxindolyl, benzoxazolyl, benzoxazolinyl, benzoxazinyl, benzothienyl, benzothiazolyl, benzodiazepinyl, benzazepinyl, benzoxazepinyl, isatinyl, dihydrobenzodioxepinyl, dihydropyridyl, pyrimidinyl, s-triazinyl, oxazolyl, or thiofuranyl. A fused arylheterocycloalkyl is for instance an indolinyl (phenyl fused to a pyrrolidinyl) and a
dihydrobenzofuranyl (phenyl fused to a dihydrofuranyl). In a particular aspect, the heteroaryl group is a pyridinyl, a pyrazolyl, a pyrimidinyl, pyridazinyl, an indolyl, and an isoxazolyl. The term “halogen” corresponds to a fluorine, chlorine, bromine, or iodine atom, preferably a fluorine, chlorine or bromine. 5 The expression “substituted by at least” means that the radical is substituted by one or several groups of the list. For instance, the expression “a (C1-C6)alkyl substituted by at least one halogen, preferably a fluorine” may include a fluoromethyl (-CH2F), a difluoromethyl (-CHF2), or a trifluoromethyl (-CF3). The expression “optionally substituted” means that the radical is not substituted or substituted 10 by one or several groups of the list. The “stereoisomers” are isomeric compounds that have the same molecular formula and sequence of bonded atoms, but differ in the 3D-dimensional orientations of their atoms in space. The stereoisomers include enantiomers, diastereoisomers, cis-trans and E-Z isomers, conformers, and anomers. In a particular embodiment of the invention, the stereoisomers 15 include diastereoisomers and enantiomers. The “tautomers” are isomeric compounds that differ only in the position of the protons and the electrons. The “hydrates” are compounds further comprising at least one molecule of water. For instance, if the compound comprises one molecule of water, it corresponds to a monohydrate form. If the 20 compound comprises two molecules of water, it corresponds to a dihydrate form. The “pharmaceutically salts” include inorganic as well as organic acids salts. Representative examples of suitable inorganic acids include hydrochloric, hydrobromic, hydroiodic, phosphoric, and the like. Representative examples of suitable organic acids include formic, acetic, trichloroacetic, trifluoroacetic, propionic, benzoic, cinnamic, citric, fumaric, maleic, 25 methanesulfonic and the like. Further examples of pharmaceutically inorganic or organic acid addition salts include the pharmaceutically salts listed in J. Pharm. Sci. 1977, 66, 2, and in Handbook of Pharmaceutical Salts: Properties, Selection, and Use edited by P. Heinrich Stahl and Camille G. Wermuth 2002. In a particular embodiment, the salt is selected from the group consisting of maleate, chlorhydrate, bromhydrate, and methanesulfonate. The 30 “pharmaceutically salts” also include inorganic as well as organic base salts. Representative examples of suitable inorganic bases include sodium or potassium salt, an alkaline earth metal salt, such as a calcium or magnesium salt, or an ammonium salt. As used herein, the terms “treatment”, “treat” or “treating” refer to any act intended to ameliorate the health status of patients such as therapy, prevention, prophylaxis and retardation
of a disease, in particular a cancer. In certain embodiments, such terms refer to the amelioration or eradication of the disease, or symptoms associated with it. In other embodiments, this term refers to minimizing the spread or worsening of the disease, resulting from the administration of one or more therapeutic agents to a subject with such a disease. In particular, such terms 5 refer to decreased development of tumors, decreased tumor burden, tumor regression, and/or prevention or delay of metastasis occurrence and cancer relapse. As used herein, the terms “subject”, “individual” or “patient” are interchangeable and refer to a mammal, even more preferably to a human, including adult, child, newborn and human at the prenatal stage. However, the term "subject" can also refer to non-human animals, in particular 10 mammals such as dogs, cats, horses, cows, pigs, sheep and non-human primates, among others. In a particular embodiment, the subject is resistant to any other anticancer treatment. Optionally, the subject is a chemoresistant subject. In a particular aspect, the subject is resistant to immunotherapy. In other words, the cancer is immune-resistant. The terms “quantity,” “amount,” and “dose” are used interchangeably herein and may refer to 15 an absolute quantification of a molecule. As used herein, the terms "active principle", "active ingredient", "active pharmaceutical ingredient", “medicine”, and “drug” are equivalent and refers to a component of a pharmaceutical composition having a therapeutic effect. As used herein, the term “therapeutic effect” refers to an effect induced by an active ingredient, 20 or a pharmaceutical composition according to the invention, capable to prevent or to delay the appearance or development of a cancer, or to cure or to attenuate the effects of a cancer. As used herein, the term “effective amount” refers to a quantity of an active ingredient or of a pharmaceutical composition which prevents, removes or reduces the deleterious effects of a cancer. It is obvious that the quantity to be administered can be adapted by the man skilled in 25 the art according to the subject to be treated, to the nature of the cancer, etc. In particular, doses and regimen of administration may be function of the nature, of the stage and of the severity of the cancer to be treated, as well as of the weight, the age and the global health of the subject to be treated, as well as of the judgment of the doctor. As used herein, the term "pharmaceutically acceptable excipient" refers to any ingredient except 30 active ingredients which are present in a pharmaceutical composition. Its addition may be aimed to confer a particular consistency or other physical or gustative properties to the final product. A pharmaceutically acceptable excipient must be devoid of any interaction, in particular chemical, with the active ingredients.
As used herein, the term “immunotherapy”, “immunotherapeutic agent” or “immunotherapy treatment” refers to a cancer therapeutic treatment using the immune system to reject cancer. The therapeutic treatment stimulates the patient's immune system to attack the malignant tumor cells. It includes immunization of the patient with tumor antigens (e.g., by administering a 5 cancer vaccine), in which case the patient's own immune system is trained to recognize tumor cells as targets to be destroyed, or administration of molecules stimulating the immune system such as cytokines, or administration of therapeutic antibodies as drugs, in which case the patient's immune system is recruited by the therapeutic antibodies to destroy tumor cells. In particular, antibodies are directed against specific antigens such as the unusual antigens that are 10 presented on the surfaces of tumors. The terms “kit”, “product” or "combined preparation", as used herein, defines especially a "kit of parts" in the sense that the combination partners (a) and (b), as defined in the present application can be dosed independently or by use of different fixed combinations with distinguished amounts of the combination partners (a) and (b), i.e., simultaneously or at 15 different time points. The parts of the kit of parts can then be administered simultaneously or chronologically staggered, that is at different time points for any part of the kit of parts. The ratio of the total amounts of the combination partner (a) to the combination partner (b) to be administered in the combined preparation can be varied. The combination partners (a) and (b) can be administered by the same route or by different routes. 20 As used herein, the term “simultaneous” refers to a pharmaceutical composition, a kit, a product or a combined preparation according to the invention in which the active ingredients are used or administered simultaneously, i.e., at the same time. As used herein, the term “sequential” refers to a pharmaceutical composition, a kit, a product or a combined preparation according to the invention in which the active ingredients are used 25 or administered sequentially, i.e., one after the other. Preferably, when the administration is sequential, all the active ingredients are administered in less than about an hour, preferably less than about 10 minutes, even more preferably in less than about a minute. As used herein, the term “separate” refers to a pharmaceutical composition, a kit, a product or a combined preparation according to the invention in which the active ingredients are used or 30 administered at distinct time of the day. Preferably, when the administration is separate, the active ingredients are administered with an interval of about 1 hour to about 24 hours, preferably with an interval of about 1 hour and 15 hours, more preferably with an interval of about 1 hour and 8 hours, even more preferably with an interval of about 1 hour and 4 hours.
Compounds The present invention provides new compounds of the following formula (I), including isomers and pharmaceutical acceptable salts thereof, of therapeutic interest. 5 According to the invention, a compound has the following formula (I):
wherein: ^ R1 represents a radical selected in a group consisting of: 10 • a hydrogen, • a (C1-C6)alkyl optionally substituted by a (C1-C6)alkyloxy, a hydroxy, at least one halogen, and a cycloalkyl, • a halogen, • a cyano, 15 • a 3-14 membered ring, saturated, partially unsaturated, or unsaturated selected in a group consisting of a cycloalkyl, a heterocycloalkyl, an aryl, and an heteroaryl, said 3-14 membered ring is optionally substituted by a (C1-C6)alkyl, a hydroxy, a halogen, a -SO2-(C1-C6)alkyl, and a (C1-C6)alkyloxy, and • a (C1-C6)alkyloxy optionally substituted by at least one halogen; 20 ^ R2 represents a radical selected in a group consisting of: • a hydrogen, • a (C1-C6)alkyl optionally substituted by a (C1-C6)alkyloxy, a hydroxy, at least one halogen, and a cycloalkyl, • a halogen, 25 • a cyano, • a 3-14 membered ring, saturated, partially unsaturated, or unsaturated selected in a group consisting of a cycloalkyl, a heterocycloalkyl, an aryl, and an heteroaryl, said 3-14 membered ring is optionally substituted by a (C1-C6)alkyl, a hydroxy, a halogen, a -SO2-(C1-C6)alkyl, and a (C1-C6)alkyloxy, and
• a (C1-C6)alkyloxy optionally substituted by at least one halogen; ^ X is selected from the group consisting of -(CH2)m- with m is from 1 to 3 optionally substituted by at least one Ra, -O-, -S-, -CO-, -C(O)-O-, -O-C(O)-, -NRa-, -CO-NRa-, - NRa-CO-, -SO-, -SO2-, -SO2-NRa, -NRa-SO2-, and -NRa-CO-NRa-, with Ra being a 5 hydrogen, a (C1-C6)alkyl optionally substituted by at least one radical C, a cycloalkyl optionally substituted by at least one radical C, or a heterocycloalkyl optionally substituted by at least one radical C; said radical C being selected in a group consisting of: - a (C1-C6)alkyloxy, 10 - a halogen, - a -O-C(O)-(C1-C6)alkyl, a -C(O)-O-(C1-C6)alkyl, a - COOH, - a -O-C(O)-cycloalkyl, a -C(O)-O-cycloalkyl, - a -CO-(C1-C6)alkyl, 15 - a -CO-cycloalkyl, - a hydroxy, - a -NRbRc with Rb and Rc represent independently a hydrogen, a (C1-C6)alkyl, a cycloalkyl, or Rb and Rc may form together a N-heterocycloalkyl, 20 - a nitro, - a cyano, - a -NH-CO-(C1-C6)alkyl, a -CO-NH-(C1-C6)alkyl, - a -NH-CO-cycloalkyl, a -CO-NH-cycloalkyl, - a -NH-SO2-(C1-C6)alkyl, a -SO2-NH-(C1-C6)alkyl, a - 25 NH-SO2-cycloalkyl, a -SO2-NH-cycloalkyl, - a -SO2-(C1-C6)alkyl, a -SO2-cycloalkyl, - a -SO-(C1-C6)alkyl, and a -SO-cycloalkyl; ^ n is 0 or 1; ^ R3 represents a radical selected in a group consisting of: 30 • a 3-14 membered ring, saturated, partially unsaturated, or unsaturated, selected in a group consisting of an aryl, a heteroaryl, a cycloalkyl, and a heterocycloalkyl, said 3-14 membered ring is optionally substituted by at least one radical A selected in a group consisting of:
- a (C1-C6)alkyl optionally substituted by a halogen, a (C1-C6)alkyloxy, or a hydroxy, - a cycloalkyl optionally substituted by a halogen, a (C1- C6)alkyloxy, a hydroxy, or a -CO-NH2, 5 - a heterocycloalkyl optionally substituted by a halogen, a (C1-C6)alkyloxy, or a hydroxy, - a (C2-C6)alkenyl, - a (C2-C6)alkynyl, - a (C1-C6)alkyloxy optionally substituted by a halogen, 10 - a halogen, - a cyano, - a -NRdRe with Rd and Re represent independently a hydrogen, a (C1-C6)alkyl, a cycloalkyl, or Rd and Re may form together a N-heterocycloalkyl, 15 - a nitro, - a hydroxy, - a ketone, - a -NH-CO-(C1-C6)alkyl, a -CO-NH-(C1-C6)alkyl, a - CO-N((C1-C6)alkyl)2, a -NH-CO-cycloalkyl, a -CO- 20 NH-cycloalkyl, - a -NH-SO2-(C1-C6)alkyl, a -SO2-NH-(C1-C6)alkyl, a - NH-SO2-cycloalkyl, a -SO2-NH-cycloalkyl, - a -C(O)-O-(C1-C6)alkyl, a -O-C(O)-(C1-C6)alkyl, a - COOH, a -C(O)-O-cycloalkyl, -O-C(O)-cycloalkyl, 25 - a -CO-(C1-C6)alkyl, a -CO-cycloalkyl, - a -SO2-(C1-C6)alkyl, a -SO2-cycloalkyl, - a -SO-(C1-C6)alkyl, and a -SO-cycloalkyl, and • a radical selected in a group consisting of: o a (C1-C6)alkyl optionally substituted by at least one radical B or 30 a cycloalkyl optionally substituted by at least one radical B, o a (C2-C6)alkenyl optionally substituted by at least one radical B, o a (C2-C6)alkynyl optionally substituted by at least one radical B, o a (C1-C6)alkyloxy optionally substituted by at least one radical B, o a hydroxy,
o a cyano, o a halogen, o a -NRf’Rg’ with Rf’ and Rg’, represent independently a hydrogen, a (C1-C6)alkyl optionally substituted by at least one radical B, a 5 cycloalkyl optionally substituted by at least one radical B, a heterocycloalkyl optionally substituted by at least one radical B, with the proviso that when one of Rf’ or Rg’ is a hydrogen, then the other is not a hydrogen, or Rf’ and Rg’ may form together a N- heterocycloalkyl, and 10 o a -N(Rf)-CO-Rg, a-CO-N(Rf)-Rg, a -CO-Rf, a -C(O)-O-Rf, a -O- C(O)-Rf, a -SO2-Rf, a -SO-Rf, a -SO2-N(Rf)-Rg, a -N(Rf)-SO2-Rg, and a -N(Rh)-CO-NRfRg, with Rf, Rg, and Rh represent independently a hydrogen, a (C1-C6)alkyl optionally substituted by at least one radical B, a cycloalkyl optionally substituted by at 15 least one radical B, a heterocycloalkyl optionally substituted by at least one radical B, or Rf and Rg may form together a N- heterocycloalkyl, said radical B being selected in a group consisting of: - a (C1-C6)alkyloxy, 20 - a (C1-C6)alkyl optionally substituted by a hydroxy, - a halogen, - a -O-C(O)-(C1-C6)alkyl optionally substituted by at least one halogen, a -C(O)-O-(C1-C6)alkyl, a -COOH, - a -O-C(O)-cycloalkyl, a -C(O)-O-cycloalkyl, 25 - a -CO-(C1-C6)alkyl, - a cycloalkyl, - a heterocycloalkyl, - a -CO-cycloalkyl, - a hydroxy, 30 - a -NRiRj with Ri and Rj represent independently a hydrogen, a (C1-C6)alkyl, a cycloalkyl, or Ri and Rj may form together a N-heterocycloalkyl, - a nitro, - a cyano,
- an aryl, - a heteroaryl, - a -NH-CO-(C1-C6)alkyl, a -CO-NH-(C1-C6)alkyl, - a -NH-CO-cycloalkyl; -CO-NH-cycloalkyl, 5 - a -NH-SO2-(C1-C6)alkyl, a -SO2-NH-(C1-C6)alkyl, - a -NH-SO2-cycloalkyl, a -SO2-NH-cycloalkyl, - a -SO2-(C1-C6)alkyl, a -SO2-cycloalkyl, - a -SO-(C1-C6)alkyl, and a -SO-cycloalkyl; ^ R4 represents a radical selected in a group consisting of: 10 • a hydrogen, • a halogen, • a (C1-C6)alkyl optionally substituted by a halogen, a (C1-C6)alkyloxy, a hydroxy, or a cycloalkyl, • a (C1-C6)alkyloxy optionally substituted by a halogen, a (C1-C6)alkyloxy, a 15 hydroxy, or a cycloalkyl, • a hydroxy, • a -NRkRl with Rk and Rl represent independently a hydrogen, a (C1-C6)alkyl, a cycloalkyl, or Rk and Rl may form a N-heterocycloalkyl, • a nitro, 20 • a cyano, • a -NH-(C1-C6)alkyl, a -NH-cycloalkyl; and • a -NH-CO-(C1-C6)alkyl, and a -NH-CO-cycloalkyl; ^ R5 represents a radical selected in a group consisting of: • a hydrogen, 25 • a halogen, • a (C1-C6)alkyl optionally substituted by a halogen, a (C1-C6)alkyloxy, a hydroxy, or a cycloalkyl, • a (C1-C6)alkyloxy optionally substituted by a halogen, a (C1-C6)alkyloxy, a hydroxy, or a cycloalkyl, 30 • a hydroxy, • a -NRkRl with Rk and Rl represent independently a hydrogen, a (C1-C6)alkyl, a cycloalkyl, or Rk and Rl may form a N-heterocycloalkyl, • a nitro,
• a cyano, • a -NH-(C1-C6)alkyl, a -NH-cycloalkyl; and • a -NH-CO-(C1-C6)alkyl, and a -NH-CO-cycloalkyl; ^ R6 represents a hydrogen, or a (C1-C6)alkyl optionally substituted by a radical selected 5 in a group consisting of a hydroxy, a (C1-C6)alkyloxy, a -NH(C1-C6)alkyl, and a -N((C1- C6)alkyl)2; ^ R7 represents a hydrogen, or a (C1-C6)alkyl optionally substituted by a radical selected in a group consisting of a hydroxy, a (C1-C6)alkyloxy, a -NH(C1-C6)alkyl, and a -N((C1- C6)alkyl)2; and 10 ^ R8 represents a hydrogen, or a (C1-C6)alkyl optionally substituted by a radical selected in a group consisting of a hydroxy, a (C1-C6)alkyloxy, a -NH(C1-C6)alkyl, and a -N((C1- C6)alkyl)2; or R7 and R8 may form together a 5-7 membered ring, partially unsaturated or saturated, N,N’-heterocycloalkyl optionally substituted by at least one radical selected in a group15 consisting of a (C1-C6)alkyl, a hydroxy, a (C1-C6)alkyloxy, a -NH(C1-C6)alkyl, a - N((C1-C6)alkyl)2, and a ketone; and the isomers, stereoisomers and pharmaceutical acceptable salts thereof. Particularly, a compound has the following formula (I): 20
wherein: ^ R1 represents a radical selected in a group consisting of: • a hydrogen, • a (C1-C6)alkyl optionally substituted by a (C1-C6)alkyloxy, a hydroxy, at least 25 one halogen, and a cycloalkyl, • a halogen, • a 3-14 membered ring, saturated, partially unsaturated, or unsaturated selected in a group consisting of a cycloalkyl, a heterocycloalkyl, an aryl, and an
heteroaryl, said 3-14 membered ring is optionally substituted by a (C1-C6)alkyl, a hydroxy, a halogen, a -SO2-(C1-C6)alkyl, and a (C1-C6)alkyloxy, and • a (C1-C6)alkyloxy optionally substituted by at least one halogen; ^ R2 represents a radical selected in a group consisting of: 5 • a hydrogen, • a (C1-C6)alkyl optionally substituted by a (C1-C6)alkyloxy, a hydroxy, at least one halogen, and a cycloalkyl, • a halogen, • a 3-14 membered ring, saturated, partially unsaturated, or unsaturated selected 10 in a group consisting of a cycloalkyl, a heterocycloalkyl, an aryl, and an heteroaryl, said 3-14 membered ring is optionally substituted by a (C1-C6)alkyl, a hydroxy, a halogen, a -SO2-(C1-C6)alkyl, and a (C1-C6)alkyloxy, and • a (C1-C6)alkyloxy optionally substituted by at least one halogen; ^ X is selected from the group consisting of -(CH2)m- with m is from 1 to 3 optionally15 substituted by at least one Ra, -O-, -S-, -CO-, -C(O)-O-, -O-C(O)-, -NRa-, -CO-NRa-, - NRa-CO-, -SO-, -SO2-, -SO2-NRa, -NRa-SO2-, and -NRa-CO-NRa-, with Ra being a hydrogen, a (C1-C6)alkyl optionally substituted by at least one radical C, a cycloalkyl optionally substituted by at least one radical C, or a heterocycloalkyl optionally substituted by at least one radical C; said radical C being selected in a group consisting 20 of: - a (C1-C6)alkyloxy, - a halogen, - a -O-C(O)-(C1-C6)alkyl, a -C(O)-O-(C1-C6)alkyl, a - COOH, 25 - a -O-C(O)-cycloalkyl, a -C(O)-O-cycloalkyl, - a -CO-(C1-C6)alkyl, - a -CO-cycloalkyl, - a hydroxy, - a -NRbRc with Rb and Rc represent independently a 30 hydrogen, a (C1-C6)alkyl, a cycloalkyl, or Rb and Rc may form together a N-heterocycloalkyl, - a nitro, - a cyano,
- a -NH-CO-(C1-C6)alkyl, a -CO-NH-(C1-C6)alkyl, - a -NH-CO-cycloalkyl, a -CO-NH-cycloalkyl, - a -NH-SO2-(C1-C6)alkyl, a -SO2-NH-(C1-C6)alkyl, a - NH-SO2-cycloalkyl, a -SO2-NH-cycloalkyl, 5 - a -SO2-(C1-C6)alkyl, a -SO2-cycloalkyl, and - a -SO-(C1-C6)alkyl or a -SO-cycloalkyl; ^ n is 0 or 1; ^ R3 represents a radical selected in a group consisting of: • a 3-14 membered ring, saturated, partially unsaturated, or unsaturated, selected 10 in a group consisting of an aryl, a heteroaryl, a cycloalkyl, and a heterocycloalkyl, said 3-14 membered ring is optionally substituted by at least one radical A selected in a group consisting of: - a (C1-C6)alkyl optionally substituted by a halogen, a (C1-C6)alkyloxy, or a hydroxy, 15 - a cycloalkyl optionally substituted by a halogen, a (C1- C6)alkyloxy, or a hydroxy, - a (C2-C6)alkenyl, - a (C2-C6)alkynyl, - a (C1-C6)alkyloxy optionally substituted by a halogen, 20 - a halogen, - a cyano, - a -NRdRe with Rd and Re represent independently a hydrogen, a (C1-C6)alkyl, a cycloalkyl, or Rd and Re may form together a N-heterocycloalkyl, 25 - a nitro, - a hydroxy, - a ketone, - a -NH-CO-(C1-C6)alkyl, a -CO-NH-(C1-C6)alkyl, a - NH-CO-cycloalkyl, a -CO-NH-cycloalkyl, 30 - a -NH-SO2-(C1-C6)alkyl, a -SO2-NH-(C1-C6)alkyl, a - NH-SO2-cycloalkyl, a -SO2-NH-cycloalkyl, - a -C(O)-O-(C1-C6)alkyl, a -O-C(O)-(C1-C6)alkyl, a - COOH, a -C(O)-O-cycloalkyl, -O-C(O)-cycloalkyl, - a -CO-(C1-C6)alkyl, a -CO-cycloalkyl,
- a -SO2-(C1-C6)alkyl, a -SO2-cycloalkyl, - a -SO-(C1-C6)alkyl, and a -SO-cycloalkyl, and • a radical selected in a group consisting of: o a (C1-C6)alkyl optionally substituted by at least one radical B or 5 a cycloalkyl optionally substituted by at least one radical B, o a (C2-C6)alkenyl optionally substituted by at least one radical B, o a (C2-C6)alkynyl optionally substituted by at least one radical B, o a (C1-C6)alkyloxy optionally substituted by at least one radical B, o a cyano, 10 o a -NRf’Rg’ with Rf’ and Rg’ represent independently a hydrogen, a (C1-C6)alkyl optionally substituted by at least one radical B, a cycloalkyl optionally substituted by at least one radical B, a heterocycloalkyl optionally substituted by at least one radical B with the proviso that when one of Rf’ or Rg’ is a hydrogen, then15 the other is not a hydrogen, or Rf’ and Rg’ may form together a N- heterocycloalkyl, and o a -N(Rf)-CO-Rg, a-CO-N(Rf)-Rg, , a -CO-Rf, a -C(O)-O-Rf, a -O- C(O)-Rf, a -SO2-Rf, a -SO-Rf, a -SO2-N(Rf)-Rg, a -N(Rf)-SO2-Rg, and a -N(Rh)-CO-NRfRg, with Rf, Rg, and Rh represent 20 independently a hydrogen, a (C1-C6)alkyl optionally substituted by at least one radical B, a cycloalkyl optionally substituted by at least one radical B, a heterocycloalkyl optionally substituted by at least one radical B, or Rf and Rg may form together a N- heterocycloalkyl, 25 said radical B being selected in a group consisting of: - a (C1-C6)alkyloxy, - a halogen, - a -O-C(O)-(C1-C6)alkyl, a -C(O)-O-(C1-C6)alkyl, a - COOH, 30 - a -O-C(O)-cycloalkyl, a -C(O)-O-cycloalkyl, - a -CO-(C1-C6)alkyl, - a -CO-cycloalkyl, - a hydroxy,
- a -NRiRj with Ri and Rj represent independently a hydrogen, a (C1-C6)alkyl, a cycloalkyl, or Ri and Rj may form together a N-heterocycloalkyl, - a nitro, 5 - a cyano, - a -NH-CO-(C1-C6)alkyl, a -CO-NH-(C1-C6)alkyl, - a -NH-CO-cycloalkyl; -CO-NH-cycloalkyl, - a -NH-SO2-(C1-C6)alkyl, a -SO2-NH-(C1-C6)alkyl, - a -NH-SO2-cycloalkyl, a -SO2-NH-cycloalkyl, 10 - a -SO2-(C1-C6)alkyl, a -SO2-cycloalkyl, - a -SO-(C1-C6)alkyl, and a -SO-cycloalkyl; ^ R4 represents a radical selected in a group consisting of: • a hydrogen, • a halogen, 15 • a (C1-C6)alkyl optionally substituted by a halogen, a (C1-C6)alkyloxy, a hydroxy, or a cycloalkyl, • a (C1-C6)alkyloxy optionally substituted by a halogen, a (C1-C6)alkyloxy, a hydroxy, or a cycloalkyl, • a hydroxy, 20 • a -NRkRl with Rk and Rl represent independently a hydrogen, a (C1-C6)alkyl, a cycloalkyl, or Rk and Rl may form a N-heterocycloalkyl, • a nitro, • a cyano, • a -NH-(C1-C6)alkyl, a -NH-cycloalkyl; and 25 • a -NH-CO-(C1-C6)alkyl, and a -NH-CO-cycloalkyl; ^ R5 represents a radical selected in a group consisting of: • a hydrogen, • a halogen, • a (C1-C6)alkyl optionally substituted by a halogen, a (C1-C6)alkyloxy, a 30 hydroxy, or a cycloalkyl, • a (C1-C6)alkyloxy optionally substituted by a halogen, a (C1-C6)alkyloxy, a hydroxy, or a cycloalkyl, • a hydroxy,
• a -NRkRl with Rk and Rl represent independently a hydrogen, a (C1-C6)alkyl, a cycloalkyl, or Rk and Rl may form a N-heterocycloalkyl, • a nitro, • a cyano, 5 • a -NH-(C1-C6)alkyl, a -NH-cycloalkyl; and • a -NH-CO-(C1-C6)alkyl, and a -NH-CO-cycloalkyl; ^ R6 represents a hydrogen, or a (C1-C6)alkyl optionally substituted by a radical selected in a group consisting of a hydroxy, a (C1-C6)alkyloxy, a -NH(C1-C6)alkyl, and a -N((C1- C6)alkyl)2; 10 ^ R7 represents a hydrogen, or a (C1-C6)alkyl optionally substituted by a radical selected in a group consisting of a hydroxy, a (C1-C6)alkyloxy, a -NH(C1-C6)alkyl, and a -N((C1- C6)alkyl)2; and ^ R8 represents a hydrogen, or a (C1-C6)alkyl optionally substituted by a radical selected in a group consisting of a hydroxy, a (C1-C6)alkyloxy, a -NH(C1-C6)alkyl, and a -N((C1- 15 C6)alkyl)2; or R7 and R8 may form together a 5-7 membered ring, partially unsaturated or saturated, N,N’-heterocycloalkyl optionally substituted by at least one radical selected in a group consisting of a (C1-C6)alkyl, a hydroxy, a (C1-C6)alkyloxy, a -NH(C1-C6)alkyl, a - N((C1-C6)alkyl)2, and a ketone. 20 According to the invention, n is 0 or 1. If n is 1, X is present. If n is 0, X is absent. In a particular embodiment, n is 1. According to this embodiment, X is selected from the group consisting of -(CH2)m- with m is from 1 to 3 optionally substituted by at least one Ra, -O-, -S-, -CO-, -C(O)-O-, -O-C(O)-, -NRa-, -CO-NRa-, -NRa-CO-, -SO-, -SO2-, -SO2-NRa, -NRa-SO2-, 25 and -NRa-CO-NRa-, with Ra being a hydrogen, a (C1-C6)alkyl optionally substituted by at least one radical C, a cycloalkyl optionally substituted by at least one radical C, or a heterocycloalkyl optionally substituted by at least one radical C; said radical C being selected in a group consisting of: - a (C1-C6)alkyloxy, 30 - a halogen, - a -O-C(O)-(C1-C6)alkyl, a -C(O)-O-(C1-C6)alkyl, a - COOH, - a -O-C(O)-cycloalkyl, a -C(O)-O-cycloalkyl,
- a -CO-(C1-C6)alkyl, - a -CO-cycloalkyl, - a hydroxy, - a -NRbRc with Rb and Rc represent independently a 5 hydrogen, a (C1-C6)alkyl, a cycloalkyl, or Rb and Rc may form together a N-heterocycloalkyl, - a nitro, - a cyano, - a -NH-CO-(C1-C6)alkyl, a -CO-NH-(C1-C6)alkyl, 10 - a -NH-CO-cycloalkyl, a -CO-NH-cycloalkyl, - a -NH-SO2-(C1-C6)alkyl, a -SO2-NH-(C1-C6)alkyl, a - NH-SO2-cycloalkyl, a -SO2-NH-cycloalkyl, - a -SO2-(C1-C6)alkyl, a -SO2-cycloalkyl, - a -SO-(C1-C6)alkyl, and a -SO-cycloalkyl. 15 In a more particular embodiment, X is selected from the group consisting of -(CH2)m- with m is from 1 to 3, particularly 1, -O-, -S-, -CO-, -C(O)-O-, -O-C(O)-, -SO-, and -SO2-. In an alternative particular embodiment, X is selected from the group consisting of -(CH2)m- with m is from 1 to 3 substituted by at least one Ra, -NRa-, -CO-NRa-, -NRa-CO-, -SO2-NRa, - NRa-SO2-, and -NRa-CO-NRa-, with Ra being a hydrogen, a (C1-C6)alkyl optionally substituted 20 by at least one radical C, a cycloalkyl optionally substituted by at least one radical C, or a heterocycloalkyl optionally substituted by at least one radical C; said radical C being selected in a group consisting of: - a (C1-C6)alkyloxy, - a halogen, 25 - a -O-C(O)-(C1-C6)alkyl, a -C(O)-O-(C1-C6)alkyl, a - COOH, - a -O-C(O)-cycloalkyl, a -C(O)-O-cycloalkyl, - a -CO-(C1-C6)alkyl, - a -CO-cycloalkyl, 30 - a hydroxy, - a -NRbRc with Rb and Rc represent independently a hydrogen, a (C1-C6)alkyl, a cycloalkyl, or Rb and Rc may form together a N-heterocycloalkyl, - a nitro,
- a cyano, - a -NH-CO-(C1-C6)alkyl, a -CO-NH-(C1-C6)alkyl, - a -NH-CO-cycloalkyl, a -CO-NH-cycloalkyl, - a -NH-SO2-(C1-C6)alkyl, a -SO2-NH-(C1-C6)alkyl, a - 5 NH-SO2-cycloalkyl, a -SO2-NH-cycloalkyl, - a -SO2-(C1-C6)alkyl, a -SO2-cycloalkyl, - a -SO-(C1-C6)alkyl, and a -SO-cycloalkyl. In an embodiment, Ra is a hydrogen, a (C1-C6)alkyl, a cycloalkyl, or a heterocycloalkyl. In an alternative embodiment, Ra is a (C1-C6)alkyl substituted by at least one radical C, a 10 cycloalkyl substituted by at least one radical C, or a heterocycloalkyl substituted by at least one radical C; said radical C being selected in a group as defined herein. In a more specific embodiment, Ra is a hydrogen. According to this more specific embodiment, X represents -NH-, -CO-NH-, -NH-CO-, -SO2-NH-, -NH-SO2-, or -NH-CO-NH-. In a particular embodiment, n is 0. According to this embodiment, a compound of the invention 15 has the following formula
with R1, R2, R3, R4, R5, R6, R7, and R8 are as defined herein. According to the invention, R4 and R5 represent independently a radical selected in a group consisting of: 20 • a hydrogen, • a halogen, • a (C1-C6)alkyl optionally substituted by a halogen, a (C1-C6)alkyloxy, a hydroxy, or a cycloalkyl, • a (C1-C6)alkyloxy optionally substituted by a halogen, a (C1-C6)alkyloxy, a 25 hydroxy, or a cycloalkyl, • a hydroxy, • a -NRkRl with Rk and Rl represent independently a hydrogen, a (C1-C6)alkyl, a cycloalkyl, or Rk and Rl may form a N-heterocycloalkyl, • a nitro,
• a cyano, • a -NH-(C1-C6)alkyl, a -NH-cycloalkyl; and • a -NH-CO-(C1-C6)alkyl, and a -NH-CO-cycloalkyl. In a particular embodiment, R4 and R5 represent H. According to this embodiment, a compound 5 of the invention has the following formula (IB):
with X, n, R1, R2, R3, R6, R7, and R8 are as defined herein. According to the invention, R6 represents a hydrogen, or a (C1-C6)alkyl optionally substituted by a radical selected in a group consisting of a hydroxy, a (C1-C6)alkyloxy, a -NH(C1-C6)alkyl, 10 and a -N((C1-C6)alkyl)2. In a particular embodiment, R6 is a hydrogen. According to the invention, R7 and R8 represent independently a hydrogen, or a (C1-C6)alkyl optionally substituted by a radical selected in a group consisting of a hydroxy, a (C1- C6)alkyloxy, a -NH(C1-C6)alkyl, and a -N((C1-C6)alkyl)2, or R7 and R8 may form together a 5- 7 membered ring, partially unsaturated or saturated, N,N’-heterocycloalkyl optionally 15 substituted by at least one radical selected in a group consisting of a (C1-C6)alkyl, a hydroxy, a (C1-C6)alkyloxy, a -NH(C1-C6)alkyl, a -N((C1-C6)alkyl)2, and a ketone. Optionally, R7 and/or R8 are a hydrogen. In a particular embodiment, R7 and R8 are a hydrogen. In a particular embodiment, R7 and R8 form together an imidazolinyl optionally substituted by20 at least one radical selected in a group consisting of a (C1-C6)alkyl, a hydroxy, a (C1- C6)alkyloxy, a -NH(C1-C6)alkyl, a -N((C1-C6)alkyl)2, and a ketone. In a specific embodiment, R7 and R8 form together an imidazolinyl substituted by a methyl and a ketone. In a particular embodiment, R6, R7, and R8 represent H. According to this embodiment, a compound of the invention has the following formula (IC): 25
with X, n, R1, R2, R3, R4, and R5 are as defined herein.
According to the invention, R1 and R2 represent independently a radical selected in a group consisting of: • a hydrogen, • a (C1-C6)alkyl optionally substituted by a (C1-C6)alkyloxy, a hydroxy, at least 5 one halogen, and a cycloalkyl, • a halogen, • a cyano, • a 3-14 membered ring, saturated, partially unsaturated, or unsaturated selected in a group consisting of a cycloalkyl, a heterocycloalkyl, an aryl, and an 10 heteroaryl, said 3-14 membered ring is optionally substituted by a (C1-C6)alkyl, a hydroxy, a halogen, a -SO2-(C1-C6)alkyl, and a (C1-C6)alkyloxy, and • a (C1-C6)alkyloxy optionally substituted by at least one halogen. In a particular embodiment, R1 represents a hydrogen, a (C1-C6)alkyl, a halogen, or a (C1- C6)alkyloxy. Optionally, R1 represents a (C1-C6)alkyl or a halogen. Optionally, R1 is selected 15 from the group consisting of methyl, bromine, chlorine or fluorine, preferably methyl, chlorine or fluorine. Optionally, R1 represents a cyclopropyl, an optionally substituted phenyl (e.g., phenyl substituted by -(SO2)-(C1-C6)alkyl, (C1-C6)alkyloxy, or halogen, in particular -(SO2)-methyl, methoxy or chlorine), or an optionally substituted pyrazolyl or pyrimidinyl. 20 In a further particular embodiment, R2 represent a hydrogen, a (C1-C6)alkyl optionally substituted by a (C1-C6)alkyloxy, a hydroxy, at least one halogen, a halogen, a cyano, a 3-14 membered ring, saturated, partially unsaturated, or unsaturated selected in a group consisting of a cycloalkyl, a heterocycloalkyl, an aryl, and an heteroaryl, said 3-14 membered ring is optionally substituted by a (C1-C6)alkyl, a hydroxy, a halogen, a -SO2-(C1-C6)alkyl, and a (C1- 25 C6)alkyloxy, or a (C1-C6)alkyloxy optionally substituted by at least one halogen. Optionally, R2 represents a (C1-C6)alkyl, a halogen, a cyano, a cyclopropyl, an optionally substituted phenyl, or an optionally substituted pyrazolyl or pyrimidinyl. Optionally, R2 is selected from the group consisting of methyl, bromine, chlorine, fluorine, a phenyl substituted by -(SO2)-(C1-C6)alkyl, (C1-C6)alkyloxy or halogen, in particular by -(SO2)-methyl, methoxy 30 or chlorine, or an optionally substituted pyrazolyl or pyrimidinyl. Optionally, at least one among R1 and R2 comprises a halogen, for instance represents a halogen or a radical substituted by a halogen. Optionally, both R1 and R2 comprise a halogen, for
instance represent a halogen or a radical substituted by a halogen. Preferably, the halogen is chlorine or fluorine, especially chlorine. In another particular embodiment, R1 and R2 are identical. 5 In a preferred embodiment, a compound of formula (I), (IA), (IB), or (IC) is such that: ^ R1 represents a (C1-C6)alkyl, preferably a methyl, a halogen, preferably a chlorine or a fluorine, more preferably a chlorine; and ^ R2 represents a (C1-C6)alkyl, preferably a methyl, a halogen, preferably a chlorine or a fluorine, more preferably a chlorine, or a cyano. 10 According to the invention, R3 represents a 3-14 membered ring, saturated, partially unsaturated, or unsaturated, optionally substituted by at least one radical A as defined herein, or a radical selected in a group consisting of a (C1-C6)alkyl, a cycloalkyl, a (C2-C6)alkenyl, a (C2-C6)alkynyl, or a (C1-C6)alkyloxy, optionally substituted by at least one radical B as defined 15 herein, a hydroxy, a cyano, a halogen, and a -NRf’Rg’ with Rf’ and Rg’, represent independently a hydrogen, a (C1-C6)alkyl optionally substituted by at least one radical B, a cycloalkyl optionally substituted by at least one radical B, a heterocycloalkyl optionally substituted by at least one radical B, with the proviso that when one of Rf’ or Rg’ is a hydrogen, then the other is not a hydrogen, or Rf’ and Rg’ may form together a N-heterocycloalkyl, a -N(Rf)-CO-Rg, a-CO-20 N(Rf)-Rg, a -C(O)-O-Rf, a -O-C(O)-Rf, a -SO2-Rf, a -SO-Rf, a -SO2-N(Rf)-Rg, a -N(Rf)-SO2- Rg, and a -N(Rh)-CO-NRfRg, with Rf, Rg, and Rh represent independently a hydrogen, a (C1- C6)alkyl, a cycloalkyl, a heterocycloalkyl, optionally substituted by at least one radical B as defined herein, or Rf and Rg may form together a N-heterocycloalkyl. 25 In a particular embodiment, R3 represents a 3-14 membered ring, saturated, partially unsaturated, or unsaturated, selected in a group consisting of an aryl, a heteroaryl, a cycloalkyl, and a heterocycloalkyl, said 3-14 membered ring is optionally substituted by at least one radical A selected in a group consisting of: - a (C1-C6)alkyl optionally substituted by a halogen, a 30 (C1-C6)alkyloxy, a hydroxy, or a -CO-NH2, - a cycloalkyl optionally substituted by a halogen, a (C1- C6)alkyloxy, a hydroxy, or a -CO-NH2, - a heterocycloalkyl optionally substituted by a halogen, a (C1-C6)alkyloxy, or a hydroxy,
- a (C2-C6)alkenyl, - a (C2-C6)alkynyl, - a (C1-C6)alkyloxy optionally substituted by a halogen, - a halogen, 5 - a cyano, - a -NRdRe with Rd and Re represent independently a hydrogen, a (C1-C6)alkyl, a cycloalkyl, or Rd and Re may form together a N-heterocycloalkyl, - a nitro, 10 - a hydroxy, - a ketone, - a -NH-CO-(C1-C6)alkyl, a -CO-NH-(C1-C6)alkyl, a - CO-N((C1-C6)alkyl)2, a -NH-CO-cycloalkyl, a -CO- NH-cycloalkyl, 15 - a -NH-SO2-(C1-C6)alkyl, a -SO2-NH-(C1-C6)alkyl, a - NH-SO2-cycloalkyl, a -SO2-NH-cycloalkyl, - a -C(O)-O-(C1-C6)alkyl, a -O-C(O)-(C1-C6)alkyl, a - COOH, a -C(O)-O-cycloalkyl, -O-C(O)-cycloalkyl, - a -CO-(C1-C6)alkyl, a -CO-cycloalkyl, 20 - a -SO2-(C1-C6)alkyl, a -SO2-cycloalkyl, and - a -SO-(C1-C6)alkyl, and a -SO-cycloalkyl. Particularly, said at least one radical A is selected in a group consisting of: - a (C1-C6)alkyl optionally substituted by a halogen, a hydroxy, a cycloalkyl, a -CO-NH((C1-C6)alkyl), or a - 25 CO-N((C1-C6)alkyl)2, - a heterocycloalkyl optionally substituted by a halogen, a (C1-C6)alkyloxy, or a hydroxy, - a cycloalkyl - a (C1-C6)alkyloxy optionally substituted by a halogen, 30 - a halogen, - a -NRdRe with Rd and Re represent a hydrogen, a (C1- C6)alkyl, or a cyclopropyl, - a cyano, - a hydroxy,
- a ketone, - a -CO-NH2, - a -COOH, - a -CO-(C1-C6)alkyl, 5 - a -C(O)-O-(C1-C6)alkyl, - a -SO2-(C1-C6)alkyl, and - a -NH-CO-(C1-C6)alkyl. In a more particular embodiment, R3 represents a 3-14 membered ring which is a heteroaryl optionally substituted by at least one radical A as defined herein. 10 In a more particular embodiment, R3 represents a 3-14 membered ring, saturated, partially unsaturated, or unsaturated, selected in a group consisting of a phenyl, a pyrimidinyl, a pyrazinyl, a pyridazinyl, a pyrazolyl, an imidazolyl, a thiazolyl, an isoxazolyl, a pyridinyl, an indolyl, , an imidazolyl, a triazolyl, an indazolyl, a thiazolyl, a thiophenyl, a benzoxazolyl (1,3- benzoxazolyl), a benzofuranyl, a benzimidazolyl, a cyclopropyl, a cyclobutyl, an oxatenyl, an 15 azetidinyl, a tetrahydrofuranyl, a 1,2,3,6-tetrahydropyridinyl, a piperidinyl, an imidazolidinyl, a 3,6-dihydro-2H-pyranyl, a 3,4-dihydro-2H-pyranyl, a tetrahydropyranyl, a tetrahydrofuranyl, a pyrrolidinyl, a morpholinyl, and a piperazinyl, particularly a phenyl, a pyrimidinyl, a pyrazolyl, an isoxazolyl, a pyridinyl, an indolyl, a cyclopropyl, a 1,2,3,6-tetrahydropyridinyl, and morpholinyl, said radical being optionally substituted by at least one radical A as defined 20 herein. Optionally, R3 represents a 3-14 membered ring selected in a group consisting of a pyrimidinyl, a pyrazolyl, an imidazolyl, a thiazolyl, an isoxazolyl, a pyridinyl, an indolyl, a pyridazinyl, a 1,3-benzoxazolyl, a benzimidazolyl, and an indazolyl, particularly a pyrimidinyl, a pyrazolyl, an isoxazolyl, a pyridinyl, and an indolyl, said radical being optionally substituted by at least 25 one radical A as defined herein. In a specific embodiment, R3 represents a 3-14 membered ring, saturated, partially unsaturated, or unsaturated, selected in a group consisting of: o an aryl, particularly a phenyl, said aryl is optionally substituted by at least one radical selected in a group consisting of: 30 - a (C1-C6)alkyl, particularly a methyl, - a (C1-C6)alkyloxy, particularly a methoxy or an ethoxy, - a cyano, - a hydroxy, - a halogen, particularly a chlorine,
- a -NRdRe with Rd and Re represent a hydrogen, and - a -NH-CO-(C1-C6)alkyl, particularly a -NH-CO-CH3, o a heteroaryl, particularly a pyrimidinyl, a pyrazinyl, a pyridazinyl, a pyrazolyl, an isoxazolyl, a pyridinyl, an indolyl, an imidazolyl, a 5 triazolyl, an indazolyl, a thiazolyl, a thiophenyl, a benzoxazolyl, or a benzofuranyl, said heteroaryl is optionally substituted by at least one radical selected in a group consisting of: - a (C1-C6)alkyl, particularly a methyl, an ethyl, an isopropyl, or a tert- butyl, said (C1-C6)alkyl is optionally substituted by at least one 10 halogen, a hydroxy, or a cyclopropyl, - a (C1-C6)alkyloxy, particularly a methoxy, - a cyano, - a halogen, particularly a chlorine, a bromine, or a iodine, - a -NRdRe with Rd and Re represent a hydrogen, 15 - a tetrahydropyranyl, - a cyclopropyl, - a hydroxy, and - a ketone, o a cycloalkyl, particularly a cyclopropyl, said cycloalkyl is optionally20 substituted by a -CO-NH2, a -COOH, a -C(O)-O-(C1-C6)alkyl, a (C1- C6)alkyl optionally substituted by a hydroxy, a -CO-CH3, a -CO- NH(CH3), or a -CO-N(CH3)2, and o a heterocycloalkyl, particularly a 1,2,3-6-tetrahydropyridinyl, a morpholinyl, a pyrrolidinyl, an imidazolidinyl, a 3,4-dihydro-2H- 25 pyranyl, a tetrahydropyranyl, a tetrahydrofuranyl, a morpholinyl, an azetidinyl, or an oxetanyl, said heterocycloalkyl is optionally substituted by a -CO-(C1-C6)alkyl, particularly a -CO-CH3, a (C1-C6)alkyl optionally substituted by at least one halogen, particularly a methyl or a trifluromethyl, a (C1-C6)alkyloxy, particularly a methoxy, a halogen, 30 particularly a fluorine, a -NRdRe with Rd and Re represent a hydrogen, a ketone, a -SO2-(C1-C6)alkyl, or a hydroxy. In a more specific embodiment, R3 represents a 3-14 membered ring, which is a heteroaryl, particularly, a pyrimidinyl, a pyrazolyl, or an isoxazolyl, said heteroaryl is optionally substituted by at least one (C1-C6)alkyl, particularly a methyl, and/or one cyano.
In a particular embodiment, R3 represents a radical selected in a group consisting of: o a (C1-C6)alkyl optionally substituted by at least one radical B or a cycloalkyl optionally substituted by at least one radical B, o a (C2-C6)alkenyl optionally substituted by at least one radical B, 5 o a (C2-C6)alkynyl optionally substituted by at least one radical B, o a (C1-C6)alkyloxy optionally substituted by at least one radical B, o a hydroxy, o a cyano, o a halogen, 10 o a -NRf’Rg’ with Rf’ and Rg’, represent independently a hydrogen, a (C1-C6)alkyl optionally substituted by at least one radical B, a cycloalkyl optionally substituted by at least one radical B, a heterocycloalkyl optionally substituted by at least one radical B, with the proviso that when one of Rf’ or Rg’ is a hydrogen, then15 the other is not a hydrogen, or Rf’ and Rg’ may form together a N- heterocycloalkyl, and o a -N(Rf)-CO-Rg, a-CO-N(Rf)-Rg, a -CO-Rf, a -C(O)-O-Rf, a -O- C(O)-Rf, a -SO2-Rf, a -SO-Rf, a -SO2-N(Rf)-Rg, a -N(Rf)-SO2-Rg, and a -N(Rh)-CO-NRfRg, with Rf, Rg, and Rh represent 20 independently a hydrogen, a (C1-C6)alkyl optionally substituted by at least one radical B, a cycloalkyl optionally substituted by at least one radical B, a heterocycloalkyl optionally substituted by at least one radical B, or Rf and Rg may form together a N- heterocycloalkyl, 25 said radical B being selected in a group consisting of: - a (C1-C6)alkyloxy, - a (C1-C6)alkyl optionally substituted by a hydroxy, - a halogen, - a -O-C(O)-(C1-C6)alkyl optionally substituted by at 30 least one halogen, a -C(O)-O-(C1-C6)alkyl, a -COOH, - a -O-C(O)-cycloalkyl, a -C(O)-O-cycloalkyl, - a -CO-(C1-C6)alkyl, - a cycloalkyl, - a heterocycloalkyl,
- a -CO-cycloalkyl, - a hydroxy, - a -NRiRj with Ri and Rj represent independently a hydrogen, a (C1-C6)alkyl, a cycloalkyl, or Ri and Rj 5 may form together a N-heterocycloalkyl, - a nitro, - a cyano, - an aryl, - a heteroaryl, 10 - a -NH-CO-(C1-C6)alkyl, a -CO-NH-(C1-C6)alkyl, - a -NH-CO-cycloalkyl; -CO-NH-cycloalkyl, - a -NH-SO2-(C1-C6)alkyl, a -SO2-NH-(C1-C6)alkyl, - a -NH-SO2-cycloalkyl, a -SO2-NH-cycloalkyl, - a -SO2-(C1-C6)alkyl, a -SO2-cycloalkyl, 15 - a -SO-(C1-C6)alkyl, and a -SO-cycloalkyl. Particularly, said at least one radical B is selected in a group consisting of: - a (C1-C6)alkyloxy, - a halogen, - a -O-CO-(C1-C6)alkyl, 20 - a hydroxy, and - a -NH-CO-(C1-C6)alkyl. In a more particular embodiment, R3 represents a radical selected in a group consisting of: o a (C1-C6)alkyl, particularly a methyl, an ethyl, an isopropyl, or an isobutyl, said (C1-C6)alkyl is optionally substituted by a (C1- 25 C6)alkyloxy, particularly a methoxy, a hydroxy, at least one halogen, or a -NH-CO-(C1-C6)alkyl, particularly a -NH-CO-CH3, o a (C2-C6)alkenyl, particularly an isopropenyl, o a (C2-C6)alkynyl optionally substituted by a heteroaryl, particularly a pyrazolyl, or a (C1-C6)alkyl optionally substituted by a hydroxy, 30 o a (C1-C6)alkyloxy, particularly a methoxy, an ethoxy, an isopropoxy, or a butoxy, said (C1-C6)alkyloxy is optionally substituted by a phenyl, a hydroxy, or by at least one halogen, o a hydroxy, o a phenoxy,
o a halogen, particularly a chlorine or an iodine, o a cyano, o a -NRf’Rg’ with Rf’ and Rg’ represent independently a hydrogen or a (C1- C6)alkyl, particularly a methyl or an isopropyl, optionally substituted by 5 a cycloalkyl or a heterocycloalkyl, with the proviso that when one of Rf’ or Rg’ is a hydrogen, then the other is not a hydrogen, o a -NH-CO-(C1-C6)alkyl, particularly a -NH-CO-CH3, o a -NH-SO2-(C1-C6)alkyl, particularly a -NH-SO2-CH3, o a -SO2-Rf with Rf represents a (C1-C6)alkyl, particularly a methyl, and10 o a -N(Rh)-CO-NRfRg, a -N(Rf)-CO-Rg, a-CO-N(Rf)-Rg, or a -N(Rf)-SO2- Rg, with Rf, Rg, and Rh represent independently a hydrogen, or a methyl. In an embodiment of the invention, a compound of formula (I) is such that R4, R5, R6, R7, and R8 represent a hydrogen. According to this embodiment, a compound of the invention has the 15 following formula
with X, n, R1, R2, and R3 are as defined herein. In an embodiment of the invention, a compound of formula (I) is such that n is 0, and R4, R5, 20 R6, R7, and R8 represent a hydrogen. According to this embodiment, a compound of the invention has the following formula (IE):
with R1, R2, and R3 are as defined herein. 25 In an embodiment of the invention, a compound of formula (I) is such that n is 1, and R4, R5, R6, R7, and R8 represent a hydrogen. According to this embodiment, a compound of the invention has the following formula (IF):
with X, R1, R2, and R3 are as defined herein. In a particular embodiment, a compound of formula (ID) or (IF) is such that X is selected from 5 the group consisting of -(CH2)m- with m is from 1 to 3, particularly 1, -O-, -S-, -CO-, -C(O)-O- , -O-C(O)-, -SO-, and -SO2-. In an alternative particular embodiment, X is selected from the group consisting of -(CH2)m- with m is from 1 to 3 substituted by at least one Ra, -NRa-, -CO-NRa-, -NRa-CO-, -SO2-NRa, - NRa-SO2-, and -NRa-CO-NRa-, with Ra being a hydrogen, a (C1-C6)alkyl optionally substituted 10 by at least one radical C, a cycloalkyl optionally substituted by at least one radical C, or a heterocycloalkyl optionally substituted by at least one radical C; said radical C being selected in a group consisting of: - a (C1-C6)alkyloxy, - a halogen, 15 - a -O-C(O)-(C1-C6)alkyl, a -C(O)-O-(C1-C6)alkyl, a - COOH, - a -O-C(O)-cycloalkyl, a -C(O)-O-cycloalkyl, - a -CO-(C1-C6)alkyl, - a -CO-cycloalkyl, 20 - a hydroxy, - a -NRbRc with Rb and Rc represent independently a hydrogen, a (C1-C6)alkyl, a cycloalkyl, or Rb and Rc may form together a N-heterocycloalkyl, - a nitro, 25 - a cyano, - a -NH-CO-(C1-C6)alkyl, a -CO-NH-(C1-C6)alkyl, - a -NH-CO-cycloalkyl, a -CO-NH-cycloalkyl, - a -NH-SO2-(C1-C6)alkyl, a -SO2-NH-(C1-C6)alkyl, a - NH-SO2-cycloalkyl, a -SO2-NH-cycloalkyl, 30 - a -SO2-(C1-C6)alkyl, a -SO2-cycloalkyl,
- a -SO-(C1-C6)alkyl, and a -SO-cycloalkyl. In an embodiment, Ra is a hydrogen, a (C1-C6)alkyl, a cycloalkyl, or a heterocycloalkyl. In an alternative embodiment, Ra is a (C1-C6)alkyl substituted by at least one radical C, a cycloalkyl substituted by at least one radical C, or a heterocycloalkyl substituted by at least one 5 radical C; said radical C being selected in a group as defined herein. In a more specific embodiment, Ra is a hydrogen. According to this more specific embodiment, X represents -NH-, -CO-NH-, -NH-CO-, -SO2-NH-, -NH-SO2-, or -NH-CO-NH-. In a further particular embodiment, the compounds of formula (ID), (IE) or (IF) are such that10 R1 and R2 represent independently a radical selected in a group consisting of a hydrogen, a (C1- C6)alkyl optionally substituted by a (C1-C6)alkyloxy, a hydroxy, at least one halogen, and a cycloalkyl, a halogen, a cyano, a 3-14 membered ring, saturated, partially unsaturated, or unsaturated selected in a group consisting of a cycloalkyl, a heterocycloalkyl, an aryl, and an heteroaryl, said 3-14 membered ring is optionally substituted by a (C1-C6)alkyl, a hydroxy, a 15 halogen, a -SO2-(C1-C6)alkyl, and a (C1-C6)alkyloxy, and a (C1-C6)alkyloxy optionally substituted by at least one halogen; In a further particular embodiment, R1 represent a hydrogen, a (C1-C6)alkyl, a halogen, or a (C1-C6)alkyloxy. Optionally, R1 represents a (C1-C6)alkyl or a halogen. Optionally, R1 is selected from the group consisting of methyl, bromine, chlorine or fluorine, preferably methyl, 20 chlorine or fluorine. Optionally, R1 represents a cyclopropyl, an optionally substituted phenyl (e.g., phenyl substituted by -(SO2)-(C1-C6)alkyl, (C1-C6)alkyloxy, or halogen, in particular -(SO2)-methyl, methoxy or chlorine), or an optionally substituted pyrazolyl or pyrimidinyl. In a further particular embodiment, R2 represent a hydrogen, a (C1-C6)alkyl optionally 25 substituted by a (C1-C6)alkyloxy, a hydroxy, at least one halogen, and a cycloalkyl, a halogen, a cyano, a 3-14 membered ring, saturated, partially unsaturated, or unsaturated selected in a group consisting of a cycloalkyl, a heterocycloalkyl, an aryl, and an heteroaryl, said 3-14 membered ring is optionally substituted by a (C1-C6)alkyl, a hydroxy, a halogen, a -SO2-(C1- C6)alkyl, and a (C1-C6)alkyloxy, or a (C1-C6)alkyloxy optionally substituted by at least one 30 halogen. Optionally, R2 represents a (C1-C6)alkyl, a halogen, a cyano, a cyclopropyl, an optionally substituted phenyl, or an optionally substituted pyrazolyl or pyrimidinyl. Optionally, R2 is selected from the group consisting of methyl, bromine, chlorine, fluorine, a phenyl substituted
by -(SO2)-(C1-C6)alkyl, (C1-C6)alkyloxy or halogen, in particular by -(SO2)-methyl, methoxy or chlorine, or an optionally substituted pyrazolyl or pyrimidinyl. Optionally, at least one among R1 and R2 comprises a halogen, for instance represents a halogen or a radical substituted by a halogen. Optionally, both R1 and R2 comprise a halogen, for 5 instance represent a halogen or a radical substituted by a halogen. Preferably, the halogen is chlorine or fluorine, especially chlorine. In another particular embodiment, R1 and R2 are identical. In a specific embodiment, a compound of formula (ID), (IE) or (IF) is such that: ^ R1 represents a (C1-C6)alkyl, preferably a methyl, a halogen, preferably a chlorine or a 10 fluorine, more preferably a chlorine; and ^ R2 represents a (C1-C6)alkyl, preferably a methyl, a halogen, preferably a chlorine or a fluorine, more preferably a chlorine or a cyano. In a further particular embodiment, a compound of formula (ID), (IE) or (IF) is such that R3 represents a 3-14 membered ring, saturated, partially unsaturated, or unsaturated, selected in a 15 group consisting of: o an aryl, particularly a phenyl, said aryl is optionally substituted by at least one radical selected in a group consisting of: - a (C1-C6)alkyl, particularly a methyl, - a (C1-C6)alkyloxy, particularly a methoxy or an ethoxy, 20 - a cyano, - a hydroxy, - a halogen, particularly a chlorine, - a -NRdRe with Rd and Re represent a hydrogen, and - a -NH-CO-(C1-C6)alkyl, particularly a -NH-CO-CH3, 25 o a heteroaryl, particularly a pyrimidinyl, a pyrazinyl, a pyridazinyl, a pyrazolyl, an isoxazolyl, a pyridinyl, an indolyl, an imidazolyl, a triazolyl, an indazolyl, a thiazolyl, a thiophenyl, a benzoxazolyl, or a benzofuranyl, said heteroaryl is optionally substituted by at least one radical selected in a group consisting of: 30 - a (C1-C6)alkyl, particularly a methyl, an ethyl, an isopropyl, or a tert- butyl, said (C1-C6)alkyl is optionally substituted by at least one halogen, a hydroxy, or a cyclopropyl, - a (C1-C6)alkyloxy, particularly a methoxy, - a cyano,
- a halogen, particularly a chlorine, a bromine, or a iodine, - a -NRdRe with Rd and Re represent a hydrogen, - a tetrahydropyranyl, - a cyclopropyl, 5 - a hydroxy, and - a ketone, o a cycloalkyl, particularly a cyclopropyl, said cycloalkyl is optionally substituted by a -CO-NH2, a -COOH, a -C(O)-O-(C1-C6)alkyl, a (C1- C6)alkyl optionally substituted by a hydroxy, a -CO-CH3, a -CO- 10 NH(CH3), or a -CO-N(CH3)2, and o a heterocycloalkyl, particularly a 1,2,3-6-tetrahydropyridinyl, a morpholinyl, a pyrrolidinyl, an imidazolidinyl, a 3,4-dihydro-2H- pyranyl, a tetrahydropyranyl, a tetrahydrofuranyl, a morpholinyl, an azetidinyl, or an oxetanyl, said heterocycloalkyl is optionally substituted 15 by a -CO-(C1-C6)alkyl, particularly a -CO-CH3, a (C1-C6)alkyl optionally substituted by at least one halogen, particularly a methyl or a trifluromethyl, a (C1-C6)alkyloxy, particularly a methoxy, a halogen, particularly a fluorine, a -NRdRe with Rd and Re represent a hydrogen, a ketone, a -SO2-(C1-C6)alkyl, or a hydroxy. 20 In a specific embodiment, a compound of formula (ID), (IE) or (IF) is such that R3 represents a 3-14 membered ring, saturated, partially unsaturated, or unsaturated, which is a heteroaryl optionally substituted by at least one radical selected in a group consisting of: - a (C1-C6)alkyl, particularly a methyl, 25 - a (C1-C6)alkyloxy, particularly a methoxy, and - a cyano. Optionally, R3 represents a 3-14 membered ring selected in a group consisting of a pyrimidinyl, a pyrazolyl, an imidazolyl, a thiazolyl, an isoxazolyl, a pyridinyl, an indolyl, a pyridazinyl, a 1,3-benzoxazolyl, a benzimidazolyl, and an indazolyl, particularly a pyrimidinyl, a pyrazolyl, 30 and an isoxazolyl, said radical being optionally substituted by at least one radical A as defined herein. More specifically, a compound of formula (ID) or (IE) is such that R3 represents a 3-14 membered ring which is a heteroaryl, such as a pyrimidinyl, a pyrazolyl, or an isoxazolyl, said
heteroaryl is optionally substituted by at least one (C1-C6)alkyl, particularly a methyl, and/or a cyano. In a further specific embodiment, a compound of formula (ID) or (IE) is such that R3 represents 5 a radical selected in a group consisting of: o a (C1-C6)alkyl, particularly a methyl, or an isobutyl, o a (C2-C6)alkenyl, particularly an isopropenyl, o a (C1-C6)alkyloxy, particularly a methoxy, or a butoxy, o a -NRf’Rg’ with Rf’ and Rg’ represent independently a hydrogen or a (C1- 10 C6)alkyl, particularly a methyl or an isopropyl, with the proviso that when one of Rf’ or Rg’ is a hydrogen, then the other is not a hydrogen, o a -NH-CO-(C1-C6)alkyl, particularly a -NH-CO-CH3, and o a -NH-SO2-(C1-C6)alkyl, particularly a -NH-SO2-CH3. 15 In a further embodiment, a compound of formula (ID) or (IE) is such that: ^ R1 represents a (C1-C6)alkyl, preferably a methyl, a halogen, preferably a chlorine or a fluorine, more preferably a chlorine; ^ R2 represents a (C1-C6)alkyl, preferably a methyl, a halogen, preferably a chlorine or a fluorine, more preferably a chlorine, or a cyano; and 20 ^ R3 represents a 3-14 membered ring, which is a heteroaryl optionally substituted by at least one radical A as defined herein, particularly a pyrimidinyl, a pyrazolyl, or an isoxazolyl, optionally substituted by at least one (C1-C6)alkyl, particularly a methyl, and/or a cyano. 25 In a further embodiment, a compound of formula (ID) or (IE) is such that: ^ R1 represents a (C1-C6)alkyl, preferably a methyl, a halogen, preferably a chlorine or a fluorine, more preferably a chlorine; ^ R2 represents a (C1-C6)alkyl, preferably a methyl, a halogen, preferably a chlorine or a fluorine, more preferably a chlorine, or a cyano; and 30 ^ R3 represents a 3-14 membered ring, which is a cycloalkyl optionally substituted by at least one radical A as defined herein, particularly a cyclopropyl.
A preferred compound of formula (I) or (IA) or (IB) or (IC) or (ID) or (IE) or (IF) according to the invention has the following formula selected in a group consisting of: 5 10
, ,
, , and , 5 and a pharmaceutical acceptable salt thereof. A preferred compound of the invention is selected in a group consisting of: - N-carbamimidoyl-2-(2,6-dimethyl-3-(methylsulfonamido)phenyl)acetamide; - N-carbamimidoyl-2-(2,4-dimethyl-[1,1'-biphenyl]-3-yl)acetamide; - N-carbamimidoyl-2-(2,6-dimethyl-3-(pyrimidin-5-yl)phenyl)acetamide; 10 - N-carbamimidoyl-2-(2,6-dimethyl-3-(1-methyl-1H-pyrazol-4-yl)phenyl) acetamide; - N-carbamimidoyl-2-(2,6-dichloro-3-(methylsulfonamido)phenyl)acetamide; - N-carbamimidoyl-2-(2,6-dimethyl-3-(1H-pyrazol-4-yl)phenyl)acetamide; - N-carbamimidoyl-2-(2,6-dichloro-3-(pyrimidin-5-yl)phenyl)acetamide; - N-carbamimidoyl-2-(2-chloro-6-fluoro-3-methoxyphenyl)acetamide; 15 - N-carbamimidoyl-2-(6-chloro-2-fluoro-3-methoxyphenyl)acetamide; - N-carbamimidoyl-2-(2,6-difluoro-3-methoxyphenyl)acetamide; - 2-(3-butoxy-2,6-difluorophenyl)-N-carbamimidoylacetamide; - N-carbamimidoyl-2-(2,6-dichloro-3-methylphenyl)acetamide; - N-carbamimidoyl-2-(2,6-dimethyl-3-(pyrimidin-2-yl)phenyl)acetamide; 20 - N-carbamimidoyl-2-(2,6-dimethyl-3-(pyridin-2-yl)phenyl)acetamide; - N-carbamimidoyl-2-(2,4-dichloro-3'-methoxy-[1,1'-biphenyl]-3-yl)acetamide; - N-carbamimidoyl-2-(2,4-dichloro-3'-cyano-[1,1'-biphenyl]-3-yl)acetamide;
- N-carbamimidoyl-2-(2,3',4-trichloro-4'-ethoxy-[1,1'-biphenyl]-3-yl)acetamide; - N-carbamimidoyl-2-(6-chloro-2-methyl-3-(pyrimidin-5-yl)phenyl)acetamide; - 2-(3-acetamido-2,6-dichlorophenyl)-N-carbamimidoylacetamide; - N-carbamimidoyl-2-(2,6-dichloro-3-(isopropylamino)phenyl)acetamide; 5 - N-carbamimidoyl-2-(2,6-dichloro-3-cyclopropylphenyl)acetamide; - N-carbamimidoyl-2-(2,4-dichloro-4'-methoxy-[1,1'-biphenyl]-3-yl)acetamide; - N-carbamimidoyl-2-(2,6-dichloro-3-(dimethylamino)phenyl)acetamide; - N-carbamimidoyl-2-(2,6-dichloro-3-(methylamino)phenyl)acetamide; - N-carbamimidoyl-2-(3'-methoxy-2,4-dimethyl-[1,1'-biphenyl]-3-yl)acetamide; 10 - N-carbamimidoyl-2-(4'-methoxy-2,4-dimethyl-[1,1'-biphenyl]-3-yl)acetamide; - N-carbamimidoyl-2-(2,6-dimethyl-3-(prop-1-en-2-yl)phenyl)acetamide; - N-carbamimidoyl-2-(2,6-dichloro-3-(pyrimidin-2-yl)phenyl)acetamide; - N-carbamimidoyl-2-(2,6-dichloro-3-(pyridin-4-yl)phenyl)acetamide; - N-carbamimidoyl-2-(2,6-dichloro-3-(5-methoxypyridin-3-yl)phenyl)acetamide; 15 - N-carbamimidoyl-2-(2,6-dichloro-3-(3,5-dimethylisoxazol-4-yl)phenyl)acetamide; - N-carbamimidoyl-2-(4-chloro-3'-methoxy-2-methyl-[1,1'-biphenyl]-3-yl)acetamide; - N-carbamimidoyl-2-(2,6-dichloro-3-(1,2,3,6-tetrahydropyridin-4-yl)phenyl)acetamide; - N-carbamimidoyl-2-(2,6-dichloro-3-(1H-indol-3-yl)phenyl)acetamide; - N-carbamimidoyl-2-(2,6-dichloro-3-morpholinophenyl)acetamide; 20 -2-(3-(1-acetyl-1,2,3,6-tetrahydropyridin-4-yl)-2,6-dichlorophenyl)-N-carbamimidoyl acetamide; - N-carbamimidoyl-2-(2,6-dichloro-3-isobutylphenyl)acetamide; - N-carbamimidoyl-2-(2-chloro-3'-methoxy-4-methyl-[1,1'-biphenyl]-3-yl)acetamide; - N-carbamimidoyl-2-(2-chloro-6-methyl-3-(pyrimidin-5-yl)phenyl)acetamide; 25 - 2-(2'-acetamido-2,4-dichloro-[1,1'-biphenyl]-3-yl)-N-carbamimidoylacetamide; - N-carbamimidoyl-2-(2,6-dichloro-3-(pyridin-2-yl)phenyl)acetamide; - N-carbamimidoyl-2-(2,6-dichloro-3-(1H-pyrazol-4-yl)phenyl)acetamide; - N-carbamimidoyl-2-(2,6-dichloro-3-(4-cyano-1-methyl-1H-pyrazol-5-yl)phenyl)acetamide; and 30 - N-carbamimidoyl-2-(6-chloro-2-cyano-3-cyclopropylphenyl)acetamide; and a pharmaceutical acceptable salt therof.
Therapeutic applications As illustrated by examples, the inventors have demonstrated the therapeutic interest of the new compounds of the invention. 5 Accordingly, the present invention relates to a pharmaceutical or veterinary composition comprising a compound of formula (I), (IA), (IB), (IC), (ID), (IE) or (IF) according to the invention or any particular compound as disclosed herein. Preferably, the pharmaceutical composition further comprises a pharmaceutically or veterinary acceptable carrier or excipient. The present invention relates to the use of a compound of formula (I), (IA), (IB), (IC), (ID), 10 (IE) or (IF) according to the invention or any particular compound as disclosed herein as a drug or a medicine. The invention further relates to a method for treating a disease in a subject, wherein a therapeutically effective amount of a compound of formula (I), (IA), (IB), (IC), (ID), (IE) or (IF) according to the invention or any particular compound as disclosed herein, is administered to said subject in need thereof. The invention also relates to the use of a compound 15 of formula (I), (IA), (IB), (IC), (ID), (IE) or (IF) according to the invention or any particular compound as disclosed herein, for the manufacture of a medicine. The invention also relates to a pharmaceutical composition comprising a compound of formula (I), (IA), (IB), (IC), (ID), (IE) or (IF) according to the invention or any particular compound as disclosed herein for use as a drug. 20 The present invention also concerns: - a compound of formula (I), (IA), (IB), (IC), (ID), (IE) or (IF) as defined above including any one of the disclosed embodiments and any particular compound as disclosed herein, or a pharmaceutical composition comprising such a compound for preventing and/or treating or for 25 use for preventing and/or treating a cancer; and/or - a pharmaceutical composition comprising a compound of formula (I), (IA), (IB), (IC), (ID), (IE) or (IF) as defined above including any one of the disclosed embodiments and any particular compound as disclosed herein, and an antitumor drug, in particular for the prevention and/or the treatment of cancer or for use in the prevention and/or the treatment of cancer; and/or 30 - a compound of formula (I), (IA), (IB), (IC), (ID), (IE) or (IF) including any one of the disclosed embodiments and any particular compound as disclosed herein, or a pharmaceutical composition comprising such a compound, for preventing and/or treating a cancer or for use for preventing and/or treating a cancer in combination with an antitumor drug such as
chemotherapy, immunotherapy, and/or hormonotherapy, and/or with radiotherapy, optionally before, simultaneously and/or after surgery (e.g., tumor resection); and/or - a kit comprising (a) a compound of formula (I), (IA), (IB), (IC), (ID), (IE) or (IF) as defined above including any one of the disclosed embodiments and any particular compound as 5 disclosed herein; and (b) an antitumor drug as a combined preparation for simultaneous, separate or sequential use, for preventing and/or treating cancer or for use for preventing and/or treating a cancer; and/or - the use of a compound of formula (I), (IA), (IB), (IC), (ID), (IE) or (IF) as defined above including any one of the disclosed embodiments and any particular compound as disclosed 10 herein, or a pharmaceutical composition comprising such a compound, for the manufacture of a medicament, a medicine or a drug for the prevention and/or the treatment of a cancer; and/or - the use of a pharmaceutical composition comprising a compound of formula (I), (IA), (IB), (IC) (ID), (IE) or (IF) as defined above including any one of the disclosed embodiments and any particular compound as disclosed herein, and an additional antitumor drug, for the 15 manufacture of a medicament, a medicine or a drug for the prevention and/or the treatment of a cancer; and/or - the use of a compound of formula (I), (IA), (IB), (IC), (ID), (IE) or (IF) as defined above including any one of the disclosed embodiments and any particular compound as disclosed herein, or a pharmaceutical composition comprising such a compound, for the manufacture of 20 a medicament, a medicine or a drug for the prevention and/or the treatment of a cancer in combination with a treatment with an antitumor drug such as chemotherapy, immunotherapy, and/or hormonotherapy, and/or with radiotherapy, optionally before, simultaneously and/or after surgery (e.g., tumor resection); and/or - a method for treating a cancer, in a subject in need thereof, comprising administering an 25 effective amount of a compound of formula (I), (IA), (IB), (IC), (ID), (IE) or (IF) as defined herein including any one of the disclosed embodiments and any particular compound as disclosed herein, or a pharmaceutical composition comprising such a compound; - a method for treating a cancer, in a subject in need thereof, comprising administering an effective amount of a compound of formula (I), (IA), (IB), (IC), (ID), (IE) or (IF) as defined 30 herein including any one of the disclosed embodiments and any particular compound as disclosed herein, or a pharmaceutical composition comprising such a compound, and an additional antitumor drug, optionally with radiotherapy; - a method for treating a cancer, in a subject in need thereof, comprising administering an effective amount of a compound of formula (I), (IA), (IB), (IC), (ID), (IE) or (IF) as defined
herein including any one of the disclosed embodiments and any particular compound as disclosed herein, or a pharmaceutical composition comprising such a compound; the method further comprises chemotherapy, immunotherapy, hormonotherapy and/or radiotherapy, optionally before, simultaneously and/or after surgery (e.g., tumor resection). 5 The term “cancer”, as used herein, refers to the presence of cells possessing characteristics typical of cancer-causing cells, such as uncontrolled proliferation, immortality, metastatic potential, rapid growth and proliferation rate, and certain characteristic morphological features. The cancer may be solid tumor or hematopoietic tumor. Examples of cancer include, but are 10 not limited to, solid tumors and hematological cancers, including carcinoma, lymphoma, blastoma (including medulloblastoma and retinoblastoma), sarcoma (including liposarcoma and synovial cell sarcoma), neuroendocrine tumors (including carcinoid tumors, gastrinoma, and islet cell cancer), mesothelioma, schwannoma (including acoustic neuroma), meningioma, adenocarcinoma, melanoma, and leukemia or lymphoid malignancies. More particular 15 examples of such cancers include chronic myeloid leukemia, acute lymphoblastic leukemia, Philadelphia chromosome positive acute lymphoblastic leukemia (Ph+ ALL), squamous cell carcinoma, lung cancer, small-cell lung cancer, non-small cell lung cancer, glioma, gastrointestinal cancer, renal cancer, ovarian cancer, bile duct cancer, liver cancer, colorectal cancer, endometrial cancer, kidney cancer, prostate cancer, melanoma, skin cancer, thyroid 20 cancer, neuroblastoma, osteosarcoma, pancreatic cancer, glioblastoma multiforme, cervical cancer, stomach cancer, bladder cancer, hepatoma, breast cancer, oesophagal cancer, colon cancer, head and neck cancer, brain cancer, gastric cancer, germ cell tumor, pediatric sarcoma, sinonasal natural killer, multiple myeloma, acute myelogenous leukemia (AML), chronic lymphocytic leukemia, mastocytosis and any symptom associated with mastocytosis. 25 In a particular aspect, the cancer is chosen among of myelofibrosis, acute lymphoblastic leukemia, acute myeloblastic leukemia adrenal gland carcinoma, bile duct cancer, bladder cancer, breast cancer, cervical cancer, colorectal cancer, endometrial cancer, esophageal cancer, gastric cancer, gastrointestinal stromal tumors, glioblastoma, head and neck cancer, 30 hepatocellular carcinoma, Hodgkin’s lymphoma, kidney cancer, lung cancer, melanoma, Merkel cell skin cancer, mesothelioma, multiple myeloma, myeloproliferative disorders, non- Hodgkin lymphoma, ovarian cancer, pancreatic cancer, prostate cancer, salivary gland cancer, sarcoma, squamous cell carcinoma, testicular cancer, thyroid cancer, urothelial carcinoma, and uveal melanoma.
In a particular aspect, the cancer is resistant or has a low response to immunotherapy. More specifically, the cancer can be resistant to a treatment with a checkpoint inhibitor such as an antibody against PD-1, PD-L1, CTLA-4 and the like. 5 The administration route can be topical, transdermal, oral, rectal, sublingual, intranasal, intrathecal, intratumor or parenteral (including subcutaneous, intramuscular, intravenous and/or intradermal). Preferably, the administration route is parental, oral or topical. The pharmaceutical composition is adapted for one or several of the above-mentioned routes. The pharmaceutical composition, kit, product or combined preparation is preferably administered 10 by injection or by intravenous infusion or suitable sterile solutions, or in the form of liquid or solid doses via the alimentary canal. The pharmaceutical composition can be formulated as solutions in pharmaceutically compatible solvents or as emulsions, suspensions or dispersions in suitable pharmaceutical solvents or vehicles, or as pills, tablets or capsules that contain solid vehicles in a way known in the art. 15 Formulations of the present invention suitable for oral administration may be in the form of discrete units as capsules, sachets, tablets or lozenges, each containing a predetermined amount of the active ingredient; in the form of a powder or granules; in the form of a solution or a suspension in an aqueous liquid or non-aqueous liquid; or in the form of an oil-in-water emulsion or a water-in-oil emulsion. Formulations for rectal administration may be in the form 20 of a suppository incorporating the active ingredient and carrier such as cocoa butter, or in the form of an enema. Formulations suitable for parenteral administration conveniently comprise a sterile oily or aqueous preparation of the active ingredient which is preferably isotonic with the blood of the recipient. Every such formulation can also contain other pharmaceutically compatible and nontoxic auxiliary agents, such as, e.g. stabilizers, antioxidants, binders, dyes, 25 emulsifiers or flavoring substances. The formulations of the present invention comprise an active ingredient in association with a pharmaceutically acceptable carrier therefore and optionally other therapeutic ingredients. The carrier must be "acceptable" in the sense of being compatible with the other ingredients of the formulations and not deleterious to the recipient thereof. The pharmaceutical compositions are advantageously applied by injection or 30 intravenous infusion of suitable sterile solutions or as oral dosage by the digestive tract. Methods for the safe and effective administration of most of these chemotherapeutic agents are known to those skilled in the art. In addition, their administration is described in the standard literature.
Pharmaceutical compositions according to the invention may be formulated to release the active drug substantially immediately upon administration or at any predetermined time or time period after administration. Preferably, the treatment with the compound according to the invention or the pharmaceutical 5 composition according to the invention starts no longer than a month, preferably no longer than a week, after the diagnosis of the disease. In a particular embodiment, the treatment starts the day of the diagnosis. The compound according to the invention or the pharmaceutical composition according to the invention may be administered as a single dose or in multiple doses. 10 Preferably, the treatment is administered regularly, preferably between every day and every month, more preferably between every day and every two weeks, more preferably between every day and every week, even more preferably the treatment is administered every day. In a particular embodiment, the treatment is administered several times a day, preferably 2 or 3 times a day, even more preferably 3 times a day. 15 The duration of treatment with the compound according to the invention or the pharmaceutical composition according to the invention is preferably comprised between 1 day and 50 weeks, more preferably between 1 day and 30 weeks, still more preferably between 1 day and 15 weeks, even more preferably between 1 day and 10 weeks. In a particular embodiment, the duration of the treatment is of about 1 week. Alternatively, the treatment may last as long as the disease 20 persists. The amount of compound according to the invention or of pharmaceutical composition according to the invention to be administered has to be determined by standard procedure well known by those of ordinary skills in the art. Physiological data of the patient (e.g. age, size, and weight) and the routes of administration have to be taken into account to determine the appropriate dosage, so as a therapeutically effective amount will be administered to the patient. 25 In a particular embodiment, is administered at a dose ranging from 0.001 mg/kg body weight to 30 mg/kg body weight. In one embodiment, the compound of the invention can be used in combination with another antitumor drug or antineoplastic agent. 30 The additional antitumor drug can be selected in the non-exhaustive list of antitumor agents consisting of an inhibitor of topoisomerases I or II, an anti-mitotic agent, a DNA alkylating agent, an agent causing crosslinking of DNA, an anti-metabolic agent, a targeted agent such as a kinase inhibitor, a histone deacetylase inhibitor and an anti-EGFR agent and/or a therapeutical
antibody designed to mediate cytotoxicity against the cancer cells or to modulate one of their key biological functions. Antimitotic agents include, but are not limited to, paclitaxel, docetaxel and analogs such as larotaxel (also called XRP9881; Sanofi-Aventis), XRP6258 (Sanofi-Aventis), BMS-184476 5 (Bristol-Meyer-Squibb), BMS-188797 (Bristol-Meyer-Squibb), BMS-275183 (Bristol-Meyer- Squibb), ortataxel (also called IDN 5109, BAY 59-8862 or SB-T-101131; Bristol-Meyer- Squibb), RPR 109881A (Bristol-Meyer-Squibb), RPR 116258 (Bristol-Meyer-Squibb), NBT- 287 (TAPESTRY), PG-paclitaxel (also called CT-2103, PPX, paclitaxel poliglumex, paclitaxel polyglutamate or XyotaxTM), ABRAXANE® (also called Nab-paclitaxel; ABRAXIS 10 BIOSCIENCE), tesetaxel (also called DJ-927), IDN 5390 (INDENA), taxoprexin (also called docosahexanoic acid-paclitaxel; PROTARGA), DHA-paclitaxel (also called Taxoprexin®), and MAC-321 (WYETH). Preferably, antimitotic agents are docetaxel, paclitaxel, and is more preferably docetaxel. Inhibitors of topoisomerases I and/or II include, but are not limited to etoposide, topotecan, 15 camptothecin, irinotecan, amsacrine, intoplicin, anthracyclines such as doxorubicin, epirubicin, daunorubicin, idarubicin and mitoxantrone. Inhibitors of topoisomerase I and II include, but are not limited to intoplicin. The additional antitumor agent can be alkylating agents including, without limitation, nitrogen mustards, ethylenimine derivatives, alkyl sulfonates, nitrosoureas, metal salts and triazenes. 20 Non-exhaustive examples thereof include uracil mustard, chlormethine, cyclophosphamide (CYTOXAN®), ifosfamide, melphalan, chlorambucil, pipobroman, triethylenemelamine, triethylenethiophosphoramine, busulfan, carmustine, lomustine, cisplatin, carboplatin, fotemustine, oxaliplatin, thiotepa, streptozocin, dacarbazine, and temozolomide. In a particular embodiment, the DNA alkylating agent is preferably cisplatin, carboplatin, temozolomide, 25 fotemustine or dacarbazine. Anti-metabolic agents block the enzymes responsible for nucleic acid synthesis or become incorporated into DNA, which produces an incorrect genetic code and leads to apoptosis. Non- exhaustive examples thereof include, without limitation, folic acid antagonists, pyrimidine analogs, purine analogs and adenosine deaminase inhibitors, and more particularly 30 methotrexate, floxuridine, cytarabine, 6-mercaptopurine, 6-thioguanine, fludarabine phosphate, pentostatine, 5-fluorouracil, gemcitabine and capecitabine. In a particular embodiment, such an agent is gemcitabine. The additional anti-tumor agent can also be a targeted agent, in particular a kinase inhibitor. The kinase may be selected from the group consisting of intracellular tyrosine or
serine/threonine kinases, receptors tyrosine or serine/threonine kinase. The kinase could be selected among EGFR family, ALK, B-Raf, MEK, and mTOR. For instance, the agents may have ability to inhibit angiogenesis based on the inhibitory activities on VEGFR and PDGFR kinases. In particular, the targeted agent can be selected among the multiple kinase inhibitor 5 drugs which are already approved: Gleevec®, which inhibits Bcr-Abl and c-Kit, and Iressa® and Tarceva®, which both inhibit EGFR, sorafenib (Nexavar®, BAY 43-9006) which inhibits Raf, dasatinib (BMS-354825) and nilotinib (AMN-107, Tasigna®) which also inhibits Bcr-Abl, lapatinib which also inhibits EGFR, temsirolimus (Torisel®, CCI-779) which targets the mTOR pathway, sunitinib (Student®, SU11248) which inhibits several targets including VEGFR as10 well as specific antibodies inactivating kinase receptors: Herceptin® and Avastin®. The anti- EGFR agent can be selected among gefitinib, erlotinib, lapatinib, vandetanib, afatinib, osimertinib, neratinib, dacomitinib, brigatinib, canertinib, naquotinib, nazartinib, pelitinib, rociletinib, icotinib, AZD3759, AZ5104 (CAS
1421373-98-9), poziotinib, WZ4002, preferably is erlotinib or cetuximab. The ALK inhibitor can be selected among crizotinib, 15 entrectinib, ceritinib, alectinib, brigatinib, lorlatinib, TSR-011, CEP-37440, and ensartinib. The B-Raf inhibitor can be selected among vemurafenib, dabrafenib, regorafenib, and PLX4720. The MEK inhibitor can be selected among cobimetinib, trametinib, binimetinib, selumetinib, PD-325901, CI-1040, PD035901, U0126, TAK-733. The antitumor agent can be IDO1 inhibitors such as epacadostat. 20 The term “therapy”, as used herein, refers to any type of treatment of cancer (i.e., antitumor therapy), including an adjuvant therapy and a neoadjuvant therapy. Therapy comprises radiotherapy and therapies, preferably systemic therapies such as hormone therapy, chemotherapy, immunotherapy and monoclonal antibody therapy. 25 The term “adjuvant therapy”, as used herein, refers to any type of treatment of cancer given as additional treatment, usually after surgical resection of the primary tumor, in a patient affected with a cancer that is at risk of metastasizing and/or likely to recur. The aim of such an adjuvant treatment is to improve the prognosis. Adjuvant therapies comprise radiotherapy and therapy, preferably systemic therapy, such as hormone therapy, chemotherapy, immunotherapy and 30 monoclonal antibody therapy. The term “hormone therapy” or “hormonal therapy” or “hormonotherapy” refers to a cancer treatment having for purpose to block, add or remove hormones. For instance, in breast cancer, the female hormones estrogen and progesterone can promote the growth of some breast cancer cells. So, in these patients, hormone therapy is given to block estrogen and a non-exhaustive
list commonly used drugs includes: tamoxifen, toremifene, anastrozole, exemestane, letrozole, goserelin, leuprolide, megestrol acetate, and fluoxymesterone. As used herein, the term “chemotherapeutic treatment” or “chemotherapy” refers to a cancer therapeutic treatment using chemical or biological substances, in particular using one or several 5 antineoplastic agents. The term “radiotherapeutic treatment” or “radiotherapy” is a term commonly used in the art to refer to multiple types of radiation therapy including internal and external radiation therapies or radioimmunotherapy, and the use of various types of radiations including X-rays, gamma rays, alpha particles, beta particles, photons, electrons, neutrons, radioisotopes, and other forms 10 of ionizing radiations. The term “therapeutical antibody” refers to any antibody having an anti-tumoral effect. Preferably, the therapeutical antibody is a monoclonal antibody. Therapeutic antibodies are generally specific for surface antigens, e.g., membrane antigens. Most preferred therapeutic antibodies are specific for tumor antigens (e.g., molecules specifically expressed by tumor 15 cells), such as CD20, CD52, ErbB2 (or HER2/Neu), CD33, CD22, CD25, MUC-1, CEA, KDR, aVb3, and the like. The therapeutical antibody includes, but is not limited to, antibodies such as trastuzumab (anti-HER2 antibody), rituximab (anti-CD20 antibody), alemtuzumab, gemtuzamab, cetuximab, pertuzumab, epratuzumab, basiliximab, daclizumab, labetuzumab, sevirumab, tuvurimab, palivizumab, infliximab, omalizumab, efalizumab, natalizumab, 20 clenoliximab, and bevacizumab. In a particular aspect, the antitumor agent can be an immunomodulator. The immunomodulator can be a cancer vaccine, molecules stimulating the immune system such as cytokines, therapeutic antibodies, preferably monoclonal antibodies, in particular antibodies directed 25 against antigens specifically presented or overexpressed at the membrane of tumor cells or directed against cell receptors which blockade prevent tumor growth, adoptive T-cell therapy, immune checkpoint inhibitor treatment, and any combination thereof. For instance, the immunomodulator can be: - an immune checkpoint inhibitor (ICI), preferably an inhibitor of of PD-1 (programmed cell 30 death protein 1), PD-L1 (programmed cell death ligand), PD-L2, CTLA-4 (cytotoxic T lymphocyte associated protein 4), TIM-3 (T-cell immunoglobulin and mucin-domain containing-3), LAG-3 (Lymphocyte-activation gene 3), NKG2D, NKG2L, KIR, VISTA, BTLA (B- and T-lymphocyte attenuator), or TIGIT (T cell immunoreceptor with Ig and ITIM domains), , especially an antibody directed against an anti-CTLA-4 such as ipilimumab, an
antibody directed against PD-1 such as nivolumab, pembrolizumab, or BGB-A317, an antibody directed against PDL1 such as atezolizumab, avelumab, or durvalumab, an antibody directed against LAG-3 such as BMS-986016, an antibody directed against TIM-3, an antibody directed against TIGIT, an antibody directed against BLTA, or a combination thereof; or 5 - an activator of a costimulatory molecule, in particular an agonist of OX40, CD2, CD27, CDS, ICAM-1, LFA-1 (CD11a/CD18), ICOS (CD278), 4-1 BB (CD137), GITR, CD30, CD40, BAFFR, HVEM, CD7, LIGHT, NKG2C, SLAMF7, NKp80, CD160, B7-H3 or CD83 ligand; In some embodiments, the PD-1 inhibitor is selected from PDR001 (Novartis), Nivolumab (Bristol-Myers Squibb), Pembrolizumab (Merck & Co), Pidilizumab (CureTech), MEDI068010 (Medimmune), REGN2810 (Regeneron), TSR-042 (Tesaro), PF-06801591 (Pfizer), BGB- A317 (Beigene), BGB-108 (Beigene), INCSHR1210 (Incyte), or AMP-224 (Amplimmune). Several anti-PD-1 antibodies are already clinically approved and others are still in clinical developments. For instance, the anti-PD1 antibody can be selected from the group consisting 15 of Pembrolizumab (also known as Keytruda lambrolizumab, MK-3475), Nivolumab (Opdivo, MDX-1106, BMS-936558, ONO-4538), Pidilizumab (CT-011), Cemiplimab (Libtayo), Camrelizumab, AUNP12, AMP-224, AGEN-2034, BGB-A317 (Tisleizumab), PDR001 (spartalizumab), MK-3477, SCH-900475, PF-06801591, JNJ-63723283, genolimzumab (CBT- 501), LZM-009, BCD-100, SHR-1201, BAT-1306, AK-103 (HX-008), MEDI-0680 (also 20 known as AMP-514) MEDI0608, JS001 (see Si-Yang Liu et al., J. Hematol. Oncol.10:136 (2017)), BI-754091, CBT-501, INCSHR1210 (also known as SHR-1210), TSR-042 (also known as ANB011), GLS-010 (also known as WBP3055), AM-0001 (Armo), STI-1110 (see WO 2014/194302), AGEN2034 (see WO 2017/040790), MGA012 (see WO 2017/19846), or IBI308 (see WO 2017/024465, WO 2017/025016, WO 2017/132825, and WO 2017/133540), 25 monoclonal antibodies 5C4, 17D8, 2D3, 4H1, 4A11, 7D3, and 5F4, described in WO 2006/121168. Bifunctional or bispecific molecules targeting PD-1 are also known such as RG7769 (Roche), XmAb20717 (Xencor), MEDI5752 (AstraZeneca), FS118 (F-star), SL- 279252 (Takeda) and XmAb23104 (Xencor). 30 Antibodies directed against CTLA-4 and bifunctional or bispecific molecules targeting CTLA- 4 are also known such as ipilimumab, tremelimumab, MK-1308, AGEN-1884, XmAb20717 (Xencor), MEDI5752 (AstraZeneca). Antibodies directed against TIGIT are also known in the art, such as BMS-986207 or AB154, BMS-986207 CPA.9.086, CHA.9.547.18, CPA.9.018, CPA.9.027, CPA.9.049, CPA.9.057,
CPA.9.059, CPA.9.083, CPA.9.089, CPA.9.093, CPA.9.101, CPA.9.103, CHA.9.536.1, CHA.9.536.3, CHA.9.536.4, CHA.9.536.5, CHA.9.536.6, CHA.9.536.7, CHA.9.536.8, CHA.9.560.1, CHA.9.560.3, CHA.9.560.4, CHA.9.560.5, CHA.9.560.6, CHA.9.560.7, CHA.9.560.8, CHA.9.546.1, CHA.9.547.1, CHA.9.547.2, CHA.9.547.3, CHA.9.547.4, 5 CHA.9.547.6, CHA.9.547.7, CHA.9.547.8, CHA.9.547.9, CHA.9.547.13, CHA.9.541.1, CHA.9.541.3, CHA.9.541.4, CHA.9.541.5, CHA.9.541.6, CHA.9.541.7, and CHA.9.541.8 as disclosed in WO19232484. Anti-TIGIT antibodies are also disclosed in WO16028656, WO16106302, WO16191643, WO17030823, WO17037707, WO17053748, WO17152088, WO18033798, WO18102536, WO18102746, WO18160704, WO18200430, WO18204363, 10 WO19023504, WO19062832, WO19129221, WO19129261, WO19137548, WO19152574, WO19154415, WO19168382 and WO19215728. The LAG-3 inhibitor can be selected from LAG525 (Novartis), BMS-986016 (Bristol-Myers Squibb), or TSR-033 (Tesaro). Further known anti-LAG-3 antibodies include those described, 15 e.g., in WO 2008/132601, WO 2010/019570, WO 2014/140180, WO 2015/116539, WO 2015/200119, WO 2016/028672, US 9,244,059, US 9,505,839, which are incorporated herein by reference in their entirety. The TIM-3 inhibitor can be MGB453 (Novartis) or TSR-022 (Tesaro). Further known anti- 20 TIM-3 antibodies include those described, e.g., in WO 2016/111947, WO 2016/071448, WO 2016/144803, US 8,552,156, US 8,841,418, and US 9,163,087, which are incorporated herein by reference in their entirety. Preferably, the immunotherapy is selected from the group consisting of ipilimumab, nivolumab, 25 BGB-A317, pembrolizumab, atezolizumab, avelumab, or durvalumab, BMS-986016, and epacadostat, or any combination thereof. Further aspects and advantages of the invention will be disclosed in the following experimental section, which should be regarded as illustrative and not limiting. 30
EXAMPLES I. CHEMISTRY EXAMPLES 5 The purity data provided in the examples described below were obtained using the following methods: Method 1 – UPLC Acidic Method Apparatus: Waters HClass; Binary Solvent Pump, SM-FTN, CMA, PDA, QDa; Column: Waters ACQUITY UPLC® CSH C18, 1.7 µm, 2.1 x 30 mm at 40 °C; Detection: UV at 210- 10 400 nm unless otherwise indicated, MS by electrospray ionisation; Solvents and Gradient: 0.1% Formic in water / MeCN 98/2 to 0/100 over 2.5 minutes. Flow rate 0.77 mL/Min. Method 2 – UPLC Basic Method Apparatus: Waters HClass; Binary Solvent Pump, SM-FTN, CMA, PDA, QDa; Column:15 Waters ACQUITY UPLC® BEH C18, 1.7 µm, 2.1 x 30 mm at 40 °C; Detection: UV at 210- 400 nm unless otherwise indicated, MS by electrospray ionisation; Solvents and Gradient: 0.1% Ammonia in water / MeCN 98/2 to 0/100 over 2.5 minutes. Flow rate 0.77 mL/Min. Method 3 – LCMS Acidic Method 20 Apparatus: Agilent 1260; Binary Pump, HiP Sampler, Column Compartment, DAD:, G6150 MSD; Column: Waters Cortecs C18, 30 x 2.1 mm, 2.7μm, at 40 °C; Detection: UV at 260nm +/- 90nm unless otherwise indicated, MS by electrospray ionisation; Solvents and Gradient: 0.1% Formic in water / MeCN 98/2 to 0/100 over 2.5 minutes. Flow rate 1.35 mL/Min. 25 Method 4 – LCMS Basic Method Apparatus: Agilent 1260; Binary Pump, HiP Sampler, Column Compartment, DAD:, G6150 MSD; Column: Phenomenex Evo C18, 30 x 2.1 mm, 2.6μm, at 40 °C; Detection: UV at 260nm +/- 90nm unless otherwise indicated, MS by electrospray ionisation; 0.1% Ammonia in water / MeCN 98/2 to 0/100 over 2.5 minutes. Flow rate 1.35 mL/Min. 30 The NMR data provided in the examples described below were obtained as followed: NMR spectra were recorded using a Bruker 400MHz Avance Neo spectrometer fitted with a Bruker 5mm iProbe, or a Bruker 500MHz Avance III HD spectrometer equipped with a Bruker 5mm SmartProbeTM. Spectra were measured at 298 K, unless indicated otherwise, and were
referenced relative to the solvent resonance. The chemical shifts are reported in parts per million. Data were acquired using Bruker TopSpin software and processed using MestreNova software. Abbreviations for multiplicities observed in NMR spectra are as follows: s (singlet), d (doublet), t (triplet), q (quadruplet), m (multiplet), br (broad). 5 Solvents, reagents and starting materials were purchased and used as received from commercial vendors unless otherwise specified. The intermediates and compounds described below were named using ChemDraw® version 21.0.0 (PerkinElmer). 10 I.1. SYNTHESIS OF INTERMEDIATE COMPOUNDS Intermediate 1: 2-(3-bromo-2,6-dimethylphenyl)acetic acid 15
To a stirred solution of 2-(2,6-dimethylphenyl)acetic acid (300 mg, 0.0018 mol) in acetic acid (3 mL) was added a solution of bromine (99 µL, 0.0019 mol) in acetic acid (1.5 mL) dropwise 20 over 1 h at 45 °C under nitrogen. After complete addition the mixture was stirred for 18 h at 45 °C. After the reaction completion (LCMS), the reaction mixture was concentrated under reduced pressure. The crude obtained was purified by column chromatography (EtOAc/Hexane) to afford 2-(3-bromo-2,6-dimethylphenyl)acetic acid as a cream solid (302 mg, 58%). 25 m/z: 243.1 @ 1.32 min, Method 3.1H-NMR (400 MHz, DMSO-d6): δ 12.36 (s, 1H), 7.39 (d, J = 8.2 Hz, 1H), 6.98 (d, J = 8.2 Hz, 1H), 3.70 (s, 2H), 2.33 (s, 3H), 2.22 (s, 3H). Intermediate 2: ethyl 2-(3-bromo-2,6-dimethylphenyl)acetate 30
To a stirred solution of 2-(3-bromo-2,6-dimethylphenyl)acetic acid (302 mg, 0.0001 mol) in EtOH (10 mL) was added thionyl chloride (95 μL, 0.0001 mol) portion wise at 0 °C under a
nitrogen atmosphere. The reaction mass was stirred for 1 h at 0 °C and then stirred for 18 h at 80 °C. After the reaction completion (LCMS), the reaction mixture was concentrated under reduced pressure. The crude obtained was dissolved in water (10 mL) and extracted with EtOAc. The organic layer was washed with saturated brine solution, dried over anhydrous 5 MgSO4, filtered and concentrated under reduced pressure to afford ethyl 2-(3-bromo-2,6- dimethylphenyl)acetate as a clear oil (278 mg, 74%). m/z: 271.0 @ 1.77 min, Method 3.1H-NMR (400 MHz, DMSO-d6): δ 7.40 (d, J = 8.1 Hz, 1H), 6.99 (d, J = 8.2 Hz, 1H), 4.09 (q, J = 7.1 Hz, 2H), 3.78 (s, 2H), 2.33 (s, 3H), 2.22 (s, 3H), 1.18 (t, J = 7.1 Hz, 3H). 10 Intermediate 3: 2-(2,6-dimethyl-3-(methylsulfonamido)phenyl)acetic acid 15
Step 1: ethyl 2-(3-((tert-butoxycarbonyl)amino)-2,6-dimethylphenyl)acetate To a solution of ethyl 2-(3-bromo-2,6-dimethylphenyl)acetate (228 mg, 0.0008 mol) in 1,4- dioxane (3 mL) was added tert-butyl carbamate (148 mg, 0.0013 mol), Cs2CO3 (548 mg, 0.0017 20 mol), Pd2dba3 (39 mg, 0.0001 mol) and XantPhos (24 mg, 0.0001 mol). The reaction mass was degassed with nitrogen and heated to 100 °C for 18 h. After the reaction completion (LCMS), the reaction mixture was treated with water (30 mL) and extracted with EtOAc. The organic layer was dried over anhydrous MgSO4, filtered and concentrated under reduced pressure. The crude obtained was purified by column chromatography (EtOAc/Hexane) to afford ethyl 2-(3- 25 ((tert-butoxycarbonyl)amino)-2,6-dimethylphenyl)acetate as a yellow gum (200 mg, 77 %,). The material was used without analysis in the following step. Step 2: ethyl 2-(3-amino-2,6-dimethylphenyl)acetate To a stirred solution of ethyl 2-(3-((tert-butoxycarbonyl)amino)-2,6-dimethylphenyl)acetate (200 mg, 0.0005 mol) in DCM (5 mL) was added HCl (1.63 mL of a 4 N solution in 1,4-dioxane, 30 0.0065 mol). The reaction mixture was stirred for 2 h at RT. After the reaction completion (LCMS), the reaction mixture was concentrated under reduced pressure to afford ethyl 2-(3- amino-2,6-dimethylphenyl)acetate, HCl (212 mg, quant). The material was used without analysis in the following step. Step 3: 2-(2,6-dimethyl-3-(methylsulfonamido)phenyl)acetic acid
To a stirred solution of ethyl 2-(3-amino-2,6-dimethylphenyl)acetate, HCl (212 mg, 0.0005 mol) in DCM (2.5 mL) was added NEt3 (115 µL, 0.0008 mol) and methanesulfonyl chloride (51 µL, 0.0007 mol) at 0 °C under a nitrogen atmosphere. The reaction mixture was stirred for 4 h at RT. EtOH (1 mL) and NaOH (3 mL of a 2 M aq solution) was added and the reaction 5 mixture was stirred for 0.5 h. After the reaction completion (LCMS), the reaction mixture was treated with water (2 mL), acidified to pH 4 with 1 M aq HCl and extracted with EtOAc. The organic layer was dried over anhydrous MgSO4, filtered and concentrated under reduced pressure to afford 2-(2,6-dimethyl-3-(methylsulfonamido)phenyl)acetic acid (176 mg, quant.). The material was used without analysis in the following step. 10 Intermediate 4: methyl 2-(2,6-dichloro-3-(methylsulfonamido)phenyl)acetate 15
Step 1: 2-(2,6-dichloro-3-nitrophenyl)acetic acid To a stirred solution of 2-(2,6-dichloro-3-nitrophenyl)acetonitrile (prepared as described WO2005/34869, 2005, A2) (1.50 g, 70% wt, 4.54 mmol) under a nitrogen atmosphere was 20 added HCl (1.1 mL of a 35% aq solution, 36.4 mmol). The reaction mixture was stirred and heated to 70 ºC for 8 h. After the completion of the reaction (LCMS), the mixture was carefully treated with ice water (50 mL) and the precipitate formed was filtered to afford 2- (2,6-dichloro-3-nitrophenyl)acetic acid (0.987 g, 52 %, 60% Purity) as a white solid. m/z: no ion @ 1.16 min, Method 2.1H NMR (400 MHz, DMSO) δ 13.0 - 12.60 (bs, 1H), 8.03 25 (d, J = 8.8 Hz, 1H), 7.79 (d, J = 8.8 Hz, 1H), 4.02 (s, 2H) Step 2: methyl 2-(2,6-dichloro-3-nitrophenyl)acetate TMS-Cl (1.26 mL, 9.93 mmol) was added to MeOH (20 mL) and stirred for 30 mins before adding a solution of 2-(2,6-dichloro-3-nitrophenyl)acetic acid (828 mg, 60% Wt, 1.99 mmol) in MeOH (5 mL). The reaction mixture was stirred at rt for 16 h. After the completion 30 of the reaction (LCMS), the reaction mixture was concentrated under reduced pressure. The residue was purified by column chromatography (EtOAc/iso-hexane) to afford the title compound as a colourless solid (0.510 g, 96%). m/z: no ion @ 1.44 min, Method 3.1H NMR (400 MHz, DMSO) δ 8.06 (d, J = 8.8 Hz, 1H), 7.81 (d, J = 8.8 Hz, 1H), 4.13 (s, 2H), 3.67 (s, 3H).
Step 3: methyl 2-(3-amino-2,6-dichlorophenyl)acetate To a solution of methyl 2-(2,6-dichloro-3-nitrophenyl)acetate (0.51 g, 1.91 mmol) in THF (15 mL) and Water (5 mL) was added ammonium chloride (614 mg, 11.5 mmol) and zinc (1.00 g, 8, 15.3 mmol) and the reaction was stirred at RT for 16 h. After the completion of the reaction 5 (LCMS), the mixture was filtered through Celite washing with EtOAc (30 mL). The filtrate was diluted with water (40 mL) and EtOAc (20 mL) and transferred to a separating funnel. The phases were separated and the aqueous phase extracted with EtOAc (20 mL). The combined organic extracts were washed with brine (20 mL), dried over MgSO4, filtered and concentrated in vacuo. The crude product was purified by column chromatography (EtOAc/iso-hexane) to 10 afford the title compound as a waxy tan solid. (0.350 g, 74%) m/z: no ion @ 1.26 min, Method 2.1H NMR (400 MHz, DMSO) δ 7.12 (d, J = 8.8 Hz, 1H), 6.76 (d, J = 8.7 Hz, 1H), 5.54 (s, 2H), 3.89 (s, 2H), 3.63 (s, 3H). Step 4: methyl 2-(2,6-dichloro-3-(methylsulfonamido)phenyl)acetate To a stirred solution of methyl 2-(3-amino-2,6-dichlorophenyl)acetate (300 mg, 1.282 15 mmol) in DCM (20 mL) under a nitrogen atmosphere at 0 °C was added triethylamine (0.39 mL, 2.82 mmol) and methanesulfonyl chloride (0.22 mL, 2.82 mmol). The reaction mixture was stirred at 25 °C for 18 h. After completion of the reaction (LCMS), the mixture was evaporated to dryness and dissolved in MeOH (10 mL) and treated with 2 M aq NaOH (2 mL). The mixture was stirred at RT for 2 h before acidifying with 1 M aq HCl and extracting with 20 EtOAc (100 mL). The organic phase was dried (MgSO4), filtered and concentrated to dryness. The crude product was purified by column chromatography (EtOAc/iso-hexane) to afford title compound as a yellow oil (0.275 g, 62%). m/z: no ion @ 0.64 min, Method 21H NMR (400 MHz, DMSO) δ 9.61 (s, 1H), 7.52 (d, J = 8.8 Hz, 1H), 7.44 (d, J = 8.8 Hz, 1H), 4.02 (s, 2H), 3.66 (s, 3H), 3.06 (s, 3H). 25 Intermediate 5: methyl 2-(3-bromo-2,6-dichlorophenyl)acetate 30
To a stirred solution of methyl 2-(2,6-dichlorophenyl)acetate (16.70 g, 76.23 mmol) in conc. sulfuric acid (60 mL) was added NBS (13.57 g, 76.23 mmol) in small portions. The reaction was stirred 1 hour at 20 °C. The reaction mixture was slowly poured into ice cold water (100
mL) and stirred for 15 min. The white precipitate was filtered and washed with water (50 mL) and dried in the oven to afford the title compound (22.2 g, 95%) as a white solid. m/z: 296.8 @ 1.66 min, Method 3. 1H NMR (400 MHz, DMSO) δ 7.78 (d, J = 8.7 Hz, 1H), 7.48 (d, J = 8.7 Hz, 1H), 4.08 (s, 2H), 3.66 (s, 3H). 5 Intermediate 6: ethyl 2-(2,6-dimethyl-3-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2- yl)phenyl)acetate 10
A vial was charged with ethyl 2-(3-bromo-2,6-dimethylphenyl)acetate (391 mg, 1.154 mmol), bis(pinacolato)diboron (322 mg, 1.269 mmol), Pd-170 (15.5 mg, 0.023 mmol) and X-Phos (11 15 mg, 0.023 mmol). The vial was purged with nitrogen and under a nitrogen atmosphere a solution of 0.5 M potassium 2-ethylhexanoate in isopropyl acetate (4.6 mL, 2.307 mmol) was added. The mixture was stirred at 35 °C for 18 h. The mixture was concentrated under reduced pressure to afford crude ethyl 2-(2,6-dimethyl-3-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2- yl)phenyl)acetate as a brown gum, used directly for subsequent chemistry assuming 20 quantitative yield. Intermediate 7: methyl 2-(3-bromo-6-chloro-2-methylphenyl)acetate 25
To a stirred solution of methyl 2-(2-chloro-6-methylphenyl)acetate (83 mg, 0.42 mmol) in conc. sulfuric acid (0.4 mL) was added NBS (78 mg, 0.44 mmol) in small portions. After 30 min, the reaction mixture was cooled with an ice bath then diluted with cold water and extracted with EtOAc (3 x 5 mL). The combined organic layers were dried over Na2SO4, 30 filtered and concentrated in vacuo. The crude product was purified by column chromatography (MTBE/iso-hexane) to afford methyl 2-(3-bromo-6-chloro-2- methylphenyl)acetate as the major product (85 mg, 66%) m/z: 277.0 @ 1.69 min, Method 3. 1H NMR (400 MHz, DMSO) δ 7.57 (d, J = 8.6 Hz, 1H), 7.29 (d, J = 8.6 Hz, 1H), 3.98 (s, 2H), 3.64 (s, 3H), 2.37 (s, 3H).
Intermediate 8: methyl 2-(3-acetamido-2,6-dichlorophenyl)acetate
5 A solution of methyl 2-(3-amino-2,6-dichlorophenyl)acetate (50 mg, 0.21 mmol) in pyridine (0.50 mL) was treated with acetic anhydride (20 μL, 0.21 mmol) and stirred at RT. After 1 h a further portion of acetic anhydride (20 μL, 0.21 mmol) was added. After a further 18 h, the reaction mixture was diluted with EtOAc (10 mL) then washed successively with 1 M HCl aq. (2 x 10 mL) and brine (10 mL) before drying over MgSO4, filtering and evaporating solvent. 10 The crude product was purified by column chromatography (EtOAc/iso-hexane) to afford the title compound as a colourless solid (25 mg, 42 %,). m/z: 275.8 @ 1.10 min, Method 2. 1H NMR (400 MHz, DMSO) δ 9.61 (s, 1H), 7.69 (d, J = 8.8 Hz, 1H), 7.47 (d, J = 8.8 Hz, 1H), 4.01 (s, 2H), 3.65 (s, 3H), 2.10 (s, 3H). 15 Intermediate 9: methyl 2-(2,6-dichloro-3-(isopropylamino)phenyl)acetate
20 To a solution of methyl 2-(3-amino-2,6-dichlorophenyl)acetate (200 mg, 0.85 mmol) and acetone (125 μL, 1.709 mmol) in DCM (1 mL) and AcOH (500 μL) under a nitrogen atmosphere was added borane (2.56 mL of a 1 M solution in THF, 2.563 mmol) at 0 °C. The reaction mixture was stirred at 0 °C for 30 min. After completion of the reaction (LCMS), sat. 25 aq. NaHCO3 (5 mL) was added carefully followed by EtOAc (10 mL). The phases were separated and the aqueous layer was extracted with EtOAc (3 x 10 mL). The combined organic layers were dried over Na2SO4 and concentrated to dryness to afford crude methyl 2-(2,6- dichloro-3-(isopropylamino)phenyl)acetate (230 mg, 92 %,) as a brown gum. m/z: 276.4 @ 1.80 min, Method 2. 1H NMR (400 MHz, DMSO) δ 7.24 (d, J = 8.9 Hz, 1H), 30 6.72 (d, J = 9.0 Hz, 1H), 4.91 (d, J = 8.2 Hz, 1H), 3.91 (s, 2H), 3.63 (s, 3H), 1.18 (d, J = 6.3 Hz, 6H). Intermediate 10: methyl 2-(2,6-dichloro-3-(methylamino)phenyl)acetate and methyl 2-(2,6- dichloro-3-(dimethylamino)phenyl)acetate
5 To a stirred solution of methyl 2-(3-amino-2,6-dichlorophenyl)acetate (50 mg, 214 μmol) in DMF (0.50 mL) at 0 °C was added sodium bicarbonate (35.9 mg, 427 μmol) then methyl iodide (60 mg, 27 μL, 428 μmol). The reaction mixture was stirred at 25 °C for 5 h. After completion (LCMS), the reaction mixture was added to water (5 mL) and extracted with EtOAc (20 mL). The organic layer was dried over Na2SO4 and concentrated to afford a mixture of the 10 title compounds (approx.1:9 ratio) used directly for subsequent chemistry. Intermediate 11: methyl 2-(3-bromo-2,6-dimethylphenyl)acetate 15
To a stirred solution of 2-(3-bromo-2,6-dimethylphenyl)acetic acid (1.415 g, 5.53 mmol) in MeOH (10 mL) was added conc. sulfuric acid (295 μL, 5.53 mmol) portion wise at RT under a nitrogen atmosphere. The reaction mass was stirred for 18 h at 65 °C. After the reaction 20 completion (LCMS), the MeOH was evaporated under reduced pressure. The residue was diluted in water (20 mL) and extracted with EtOAc. The organic layer was dried over anhydrous MgSO4, filtered and concentrated under reduced pressure to afford methyl 2-(3-bromo-2,6- dimethylphenyl)acetate as a clear oil (1.40 g, 94%). m/z: 257.0 @ 1.64 min, Method 2. 1H-NMR (400 MHz, DMSO-d6): 1H NMR (400 MHz, 25 DMSO) δ 7.40 (d, J = 8.2 Hz, 1H), 6.99 (d, J = 8.2 Hz, 1H), 3.80 (s, 2H), 3.62 (s, 3H), 2.32 (s, 3H), 2.22 (s, 3H). Intermediate 12: methyl 2-(2,6-dichloro-3-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2- yl)phenyl)acetate 30
To a degassed solution of potassium acetate (299 mg, 3.043 mmol), bis(pinacolato)diborane (284 mg, 1.118 mmol) and methyl 2-(3-bromo-2,6-dichlorophenyl)acetate (303 mg, 1.018 mmol) in 1,4-dioxane (10 mL) was added 1,1'-bis(diphenylphosphino)ferrocene-palladium(II) dichloride (80 mg, 110 μmol )and the mixture heated at 70 °C. Upon completion (LCMS), the 5 reaction mixture was concentrated under reduced pressure onto silica gel and the residue purified by column chromatography (EtOAc/iso-hexane) to afford the title compound as a clear colourless oil (220 mg, 58%). m/z: 345.2 @ 1.92 min, Method 3. 1H NMR (400 MHz, DMSO) δ 7.55 (d, J = 8.1 Hz, 1H), 7.50 (d, J = 8.1 Hz, 1H), 4.01 (s, 2H), 3.64 (s, 3H), 1.31 (s, 12H). 10 Intermediate 13: methyl 2-(2,6-dichloro-3-morpholinophenyl)acetate
15 To a stirred mixture of cesium carbonate (315 mg, 967 μmol), xantphos (14 mg, 24.2 μmol), methyl 2-(3-bromo-2,6-dichlorophenyl)acetate Intermediate 5 (150 mg, 483 μmol) and Pd-177 (18.4 mg, 0.05 Eq, 24.2 μmol) and 1,4-dioxane (2 mL) under a nitrogen atmosphere at 25 oC was added morpholine (50 μL, 580 μmol). The reaction mixture was stirred 20 at 60 °C for 2 h then 80 °C for 16 h. After completion of the reaction (LCMS), the mixture was filtered on a pad of celite, washing with with DCM, and the solvent was removed under vacuum to obtain a yellow oil. The crude compound was purified by column chromatography on silica to afford methyl 2-(2,6-dichloro-3-morpholinophenyl)acetate (63 mg, 0.21 mmol, 42 %, 99% Purity) as an off-white solid. 25 m/z: 304.2 @ 2.23 min, Method 2. 1H NMR (400 MHz, DMSO) δ 7.46 (d, J = 8.7 Hz, 1H), 7.17 (d, J = 8.7 Hz, 1H), 4.00 (s, 2H), 3.77 – 3.70 (m, 4H), 3.64 (s, 3H), 2.98 – 2.91 (m, 4H). Intermediate 14: methyl 2-(2,6-dichloro-3-isobutylphenyl)acetate 30
To a stirred suspension of zinc (329 mg, 5.03 mmol) in THF (5 mL) was added TMS-Cl (64 μL, 503 μmol). The mixture was stirred at 55 °C for 15 min whereupon 1-iodo-2-
methylpropane (290 μL, 2.52 mmol) was added dropwise. The mixture was then stirred at 55 °C for 20 min to afford an organozinc solution used as detailed below. Another vial was charged with methyl 2-(3-bromo-2,6-dichlorophenyl)acetate Intermediate 5 (150 mg, 503 μmol), Pd(dppf)Cl2 (37 mg, 50.3 μmol) and THF (2 mL). The solution was 5 degassed for 5 min with a nitrogen flow under magnetic stirring. The organozinc solution was added and the mixture heated to 65 °C for 20 h. The reaction mixture was quenched with saturated aq. NH4Cl (5 mL) and extracted with EtOAc (2 x 10 mL). The combined organic layers were dried over MgSO4, filtered and concentrated in vacuo. The crude product was purified by column chromatography (iso-hexane/MTBE) to afford the title 10 compound (40 mg, 28%) as a colourless oil. m/z: no ion @ 0.80 min, Method 3. 1H NMR (400 MHz, CDCl3) δ 7.18 (d, J = 8.4 Hz, 1H), 7.00 (d, J = 8.2 Hz, 1H), 3.99 (s, 2H), 3.65 (s, 3H), 2.52 (d, J = 7.2 Hz, 2H), 1.88 (dp, J = 13.6, 6.8 Hz, 1H), 0.85 (d, J = 6.6 Hz, 6H). 15 Intermediate 15: ethyl 2-(3-bromo-2-chloro-6-methylphenyl)acetate
20 Following the procedure described for Intermediate 6, ethyl 2-(2-chloro-6- methylphenyl)acetate (1.0 g, 4.6 mmol) gave ethyl 2-(3-bromo-2-chloro-6- methylphenyl)acetate (381 mg, 28%) as the minor product. m/z: 291.0 @ 1.77 min, Method 3.1H NMR (400 MHz, CDCl3) δ 7.45 (d, J = 8.2 Hz, 1H), 6.98 (d, J = 8.2 Hz, 1H), 4.18 (q, J = 7.1 Hz, 2H), 3.90 (s, 2H), 2.30 (s, 3H), 1.26 (t, J = 7.1 Hz, 3H). 25 I.2. SYNTHESIS OF COMPOUNDS OF THE INVENTION I.2.1 GENERAL PROTOCOLS FOR THE SYNTHESIS OF FINAL COMPOUNDS 30 Protocol 1
Step 1 To a stirred solution of carboxylic acid P1-1 in DMF (0.1 M) was added N-Boc guanidine (1.1- 1.2 eq), DIPEA (2-3 eq) and HATU (1.4-1.5 eq) at RT under a nitrogen atmosphere. After 5 reaction completion (LCMS), the reaction mixture was quenched with saturated sodium bicarbonate solution. The reaction mixture was extracted with EtOAc and the organic layer dried over anhydrous MgSO4, filtered and concentrated under reduced pressure. The crude obtained was purified by column chromatography or used directly for subsequent chemistry. Alternatively, the reaction was quenched with water and the product isolated directly by 10 filtration to afford P1-2. Step 2 To a stirred solution of Boc-protected-acyl-guanidine P1-2 in DCM (20-50 vol) was added TFA (10-20 eq). After reaction completion (LCMS), the reaction mixture was concentrated under reduced pressure. The crude product could be further purified by trituration or preparative 15 HPLC to afford P1-3 Protocol 2 20 25
To a solution of bromide or boronate P2-1 (1 eq), boronate or bromide P2-2 (1.5 eq) and caesium or potassium carbonate (3 eq) in Acetonitrile/Water (4:1 ratio) was added SPhos Pd 30 G3 (10 mol%). Alternatively, PdCl2dppf or Pd(PPh3)Cl2 was used. The reaction mixture was
degassed with nitrogen and heated to 80 °C under nitrogen. After the reaction completion (LCMS), the mixture was allowed to cool and subjected to aqueous work up. The crude product was purified by column chromatography or used directly to afford P2-3 Step 2 5 To a stirred solution of carboxylic acid P2-3 in DMF (0.1 M) was added N-Boc guanidine (1.1- 1.2 eq), DIPEA (2-3 eq) and HATU (1.4-1.5 eq) at RT under a nitrogen atmosphere. After reaction completion (LCMS), the reaction mixture was quenched with saturated sodium bicarbonate solution. The reaction mixture was extracted with EtOAc and the organic layer dried over anhydrous MgSO4, filtered and concentrated under reduced pressure. The crude 10 obtained was purified by column chromatography or used directly for subsequent chemistry. Alternatively, the reaction was quenched with water and the product isolated directly by filtration to afford P2-4. Step 3 To a stirred solution of Boc-protected-acyl-guanidine P2-4 in DCM (20-50 vol) was added TFA 15 (10-20 eq). After reaction completion (LCMS), the reaction mixture was concentrated under reduced pressure. The crude product could be further purified by trituration or preparative HPLC to afford P2-5 Protocol 3
20 Step 1 To a stirred solution of ester P3-1 in MeOH or EtOH (30 vol) or a mixture of THF/MeOH (2/1) was added sodium hydroxide (2 -5 Eq,). The reaction was treated with water (0-6 vol) and stirred at rt or heated as required. After completion of the reaction, the mixture was acidified with 1 M aq HCl and extracted with EtOAc The organic phase was dried (MgSO4), filtered and 25 concentrated to give the carboxylic acid P3-2. Step 2
To a stirred solution of carboxylic acid P3-2 in DMF (0.1 M) was added N-Boc guanidine (1.1- 1.2 eq), DIPEA (2-3 eq) and HATU (1.4-1.5 eq) at RT under a nitrogen atmosphere. After reaction completion (LCMS), the reaction mixture was quenched with saturated sodium bicarbonate solution. The reaction mixture was extracted with EtOAc and the organic layer 5 dried over anhydrous MgSO4, filtered and concentrated under reduced pressure. The crude obtained was purified by column chromatography or used directly for subsequent chemistry. Alternatively, the reaction was quenched with water and the product isolated directly by filtration to afford P3-3. Step 3 10 To a stirred solution of Boc-protected-acyl-guanidine P3-3 in dichloromethane (20-50 vol) was added Trifluoroacetic acid (10-20 eq). After reaction completion (LCMS), the reaction mixture was concentrated under reduced pressure. The crude product could be further purified by trituration or preparative HPLC to afford P3-4 15 Protocol 4
Step 1 To a stirred solution of carboxylic acid P4-1 in DMF (0.1 M) was added N-Boc guanidine (1.1- 20 1.2 eq), DIPEA (2-3 eq) and HATU (1.4-1.5 eq) at RT under a nitrogen atmosphere. After reaction completion (LCMS), the reaction mixture was quenched with saturated sodium bicarbonate solution. The reaction mixture was extracted with EtOAc and the organic layer dried over anhydrous MgSO4, filtered and concentrated under reduced pressure. The crude obtained was purified by column chromatography or used directly for subsequent chemistry. 25 Alternatively, the reaction was quenched with water and the product isolated directly by filtration to afford P4-2. Step 2 To a stirred solution of Boc-protected-acyl-guanidine P4-2 in dioxane (10 vol) was added HCl (15 eq of a 4 M solution in dioxane). After reaction completion (LCMS), the reaction mixture 30 was concentrated under reduced pressure. The crude product could be further purified by trituration or preparative HPLC to afford P4-3.
Protocol 5
Step 1 To a solution of bromide P5-1 (1 eq), boronate P5-2 (1.5 eq) and caesium or potassium 5 carbonate (3 eq) in Acetonitrile/Water (4:1 ratio) was added SPhos Pd G3 (10 mol%). Alternatively, PdCl2dppf or Pd(PPh3)Cl2 was used. The reaction mixture was degassed with nitrogen and heated to 80 °C under nitrogen. After the reaction completion (LCMS), the mixture was allowed to cool and subjected to aqueous work up. The crude product was purified by column chromatography or used directly to afford P5-3 10 Step 2 To a stirred suspension of guanidine hydrochloride (2-2.5 eq) in DMF (10-30 vol) at 35 °C was added sodium methoxide (2 eq of a 30% Wt solution in MeOH) or potassium tert-butoxide (2 eq). After, 5 mins the ester P5-3 was added was added. If required, a further charge of reagents was added. After completion (LCMS), the mixture was quenched with water and the precipitate 15 collected by filtration to afford crude material. This was purified by preparative HPLC or column chromatography to afford the product P5-4. 20 25
Protocol 6
Step 1 To a solution of boronate or bromide P6-1 (1 eq), bromide or boronate P6-2 (1.5 eq) and 5 caesium or potassium carbonate (3 eq) in Acetonitrile/Water (4:1 ratio) was added SPhos Pd G3 (10 mol%). Alternatively, PdCl2dppf or Pd(PPh3)Cl2 was used. The reaction mixture was degassed with nitrogen and heated to 80 °C under nitrogen. After the reaction completion (LCMS), the mixture was allowed to cool and subjected to aqueous work up. The crude product was purified by column chromatography or used directly to afford P6-3 10 Step 2 To a stirred solution of ester P6-3 in MeOH or EtOH (30 vol) or a mixture of THF/MeOH (2/1) was added sodium hydroxide (2 -5 Eq,). The reaction was treated with water (0-6 vol) and stirred at rt or heated as required. After completion of the reaction, the mixture was acidified with 1M HCl and extracted with EtOAc The organic phase was dried (MgSO4), filtered and 15 concentrated to give the carboxylic acid P6-4. Step 3 To a stirred solution of carboxylic acid P6-4 in DMF (0.1 M) was added N-Boc guanidine (1.1- 1.2 eq), DIPEA (2-3 eq) and HATU (1.4-1.5 eq) at RT under a nitrogen atmosphere. After reaction completion (LCMS), the reaction mixture was quenched with saturated sodium 20 bicarbonate solution. The reaction mixture was extracted with EtOAc and the organic layer dried over anhydrous MgSO4, filtered and concentrated under reduced pressure. The crude obtained was purified by column chromatography or used directly for subsequent chemistry. Alternatively, the reaction was quenched with water and the product isolated directly by filtration to afford P6-5. 25 Step 4
To a stirred solution of Boc-protected-acyl-guanidine P6-5 in dichloromethane (20-50 vol) was added Trifluoroacetic acid (10-20 eq). After reaction completion (LCMS), the reaction mixture was concentrated under reduced pressure. The crude product could be further purified by trituration or preparative HPLC to afford P6-6 5 Protocol 7 10
To a stirred suspension of guanidine hydrochloride (2-2.5 eq) in DMF (10-30 vol) at 35 °C was added sodium methoxide (2 eq of a 30% Wt solution in MeOH) or potassium tert-butoxide (2 eq). After, 5 mins the ester P7-1 was added was added. If required, a further charge of reagents 15 was added. After completion (LCMS), the mixture was quenched with water and the precipitate collected by filtration to afford crude material. This was purified by preparative HPLC or column chromatography to afford the product P7-2. I.2.2 EXAMPLES 20 Example 1: N-carbamimidoyl-2-(2,6-dimethyl-3-(methylsulfonamido)phenyl)acetamide. TFA 25
The title compound was prepared from Intermediate 3 using Protocol 1 m/z: 299.1 @ 0.40 min, Method 3.1H NMR (500 MHz, DMSO) δ 9.01 (s, 1H), 8.25 (s, 4H), 7.12 (d, J = 8.0 Hz, 1H), 7.06 (d, J = 8.1 Hz, 1H), 3.88 (s, 2H), 2.92 (s, 3H), 2.22 (app d, J = 30 10.5 Hz, 6H). NH proton not observed.
Example 2: N-carbamimidoyl-2-(2,4-dimethyl-[1,1'-biphenyl]-3-yl)acetamide.TFA 5
The title compound was prepared from Intermediate 2 using Protocol 2 and phenylboronic acid. m/z: 282.2 @ 1.04 min, Method 3.1H NMR (400 MHz, DMSO) δ 11.77 (s, 1H), 8.36 (s, 4H), 10 7.46 – 7.39 (m, 2H), 7.38 – 7.32 (m, 1H), 7.28 – 7.23 (m, 2H), 7.11 (d, J = 7.8 Hz, 1H), 7.03 (d, J = 7.7 Hz, 1H), 3.94 (s, 2H), 2.28 (s, 3H), 2.10 (s, 3H). Example 3: N-carbamimidoyl-2-(2,6-dimethyl-3-(pyrimidin-5-yl)phenyl)acetamide.TFA 15
The title compound was prepared from Intermediate 2 using Protocol 2 and pyrimidin-5- 20 ylboronic acid. m/z: 284.1 @ 0.64 min, Method 3.1H NMR (400 MHz, DMSO) δ 11.73 (s, 1H), 9.20 (s, 1H), 8.78 (s, 2H), 8.32 (s, 4H), 7.21 (d, J = 7.8 Hz, 1H), 7.16 (d, J = 7.8 Hz, 1H), 3.97 (s, 2H), 2.31 (s, 3H), 2.13 (s, 3H). 25 Example 4: N-carbamimidoyl-2-(2,6-dimethyl-3-(1-methyl-1H-pyrazol-4- yl)phenyl)acetamide.TFA 30
The title compound was prepared from Intermediate 2 using Protocol 2 and 1-methyl-4- (4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-1H-pyrazole
m/z: 286.2 @ 0.72 min, Method 3.1H NMR (400 MHz, DMSO) δ 11.69 (s, 1H), 8.33 (s, 4H), 7.80 (s, 1H), 7.53 – 7.48 (m, 1H), 7.12 (d, J = 7.8 Hz, 1H), 7.05 (d, J = 7.9 Hz, 1H), 3.92 (s, 2H), 3.87 (s, 3H), 2.24 (s, 3H), 2.23 (s, 3H). 5 Example 5: N-carbamimidoyl-2-(2,6-dichloro-3-(methylsulfonamido)phenyl)acetamide 10
The title compound was prepared from Intermediate 4 using Protocol 3. m/z: 339.1 @ 0.48 min, Method 2. 1H NMR (400 MHz, DMSO) δ 7.40 (d, J = 8.7 Hz, 1H), 7.33 (d, J = 8.7 Hz, 1H), 3.83 (s, 2H), 3.00 (s, 3H), 6 x NH broad and partially exchanged. 15
The title compound was prepared from Intermediate 2 using Protocol 2 and tert-butyl 4- 20 (4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-1H-pyrazole-1-carboxylate m/z: 272.1 @ 0.64 min, Method 3.1H NMR (500 MHz, DMSO) δ 11.59 (s, 1H), 8.28 (s, 4H), 7.70 (s, 2H), 7.14 (d, J = 7.8 Hz, 1H), 7.05 (d, J = 7.9 Hz, 1H), 3.92 (s, 2H), 2.25 (s, 3H), 2.23 (s, 3H). NH protons not observed. 25 Example 7: N-carbamimidoyl-2-(2,6-dichloro-3-(pyrimidin-5-yl)phenyl)acetamide.TFA
30 The title compound was prepared from Intermediate 5 using Protocol 2 and pyrimidin-5- ylboronic acid. m/z: 324.1 @ 0.79 min, Method 2.1H NMR (400 MHz, DMSO) δ 11.87 (s, NH), 9.27 (s, 1H), 8.92 (s, 2H), 8.28 (s, 2NH), 7.72 (d, J = 8.4 Hz, 1H), 7.59 (d, J = 8.4 Hz, 1H), 4.26 (s, 2H). One NH not observed.
Example 8: N-carbamimidoyl-2-(2-chloro-6-fluoro-3-methoxyphenyl)acetamide.TFA 5
The title compound was prepared from 2-chloro-6-fluoro-3-methoxyphenyl)acetic acid using Protocol 1. m/z: 260.1 @ 0.81 min, Method 2. 1H NMR (400 MHz, DMSO) δ 11.84 (s, NH), 8.32 (s, 3NH), 7.26 (t, J = 9.1 Hz, 1H), 7.16 (dd, J = 9.1, 4.9 Hz, 1H), 4.00 (d, J = 1.7 Hz, 2H), 3.85 (s, 10 3H). Example 9: N-carbamimidoyl-2-(6-chloro-2-fluoro-3-methoxyphenyl)acetamide.TFA 15
The title compound was prepared from 2-(6-chloro-2-fluoro-3-methoxyphenyl)acetic acid using Protocol 1. m/z: 260.2 @ 0.85 min, Method 2.1H NMR (400 MHz, DMSO) δ 11.95 (br s, 1H), 8.39 (br s, 4H), 7.31 (dd, J = 9.0, 1.8 Hz, 1H), 7.19 (t, J = 9.0 Hz, 1H), 3.98 (d, J = 2.0 Hz, 2H), 3.85 (s, 20 3H). Example 10: N-carbamimidoyl-2-(2,6-difluoro-3-methoxyphenyl)acetamide.HCl 25
The title compound was prepared from 2-(2,6-difluoro-3-methoxyphenyl)acetic acid using Protocol 4. m/z: 243.9 @ 0.74 min, Method 2. 1H NMR (400 MHz, DMSO) δ 11.96 (s, 1H), 8.25 (s, 30 4H), 7.17 (td, J = 9.4, 5.4 Hz, 1H), 7.06 (td, J = 9.1, 1.9 Hz, 1H), 3.89 (s, 2H), 3.83 (s, 3H). Example 11: 2-(3-butoxy-2,6-difluorophenyl)-N-carbamimidoylacetamide.HCl
The title compound was prepared from 2-(3-butoxy-2,6-difluorophenyl)acetic acid using 5 Protocol 4. m/z: 286.2 @ 1.21 min, Method 2.1H NMR (400 MHz, DMSO) δ 11.94 (s, 1H), 8.22 (s, 4H), 7.17 (td, J = 9.4, 5.3 Hz, 1H), 7.04 (td, J = 9.1, 1.9 Hz, 1H), 4.03 (t, J = 6.4 Hz, 2H), 3.89 (s, 2H), 1.69 (dq, J = 8.3, 6.4 Hz, 2H), 1.50 – 1.37 (m, 2H), 0.93 (t, J = 7.4 Hz, 3H). 10 Example 12: N-carbamimidoyl-2-(2,6-dichloro-3-methylphenyl)acetamide.TFA
The title compound was prepared from Intermediate 5 using Protocol 5 with trimethylboroxine. 15 The Suzuki coupling used potassium carbonate and Pd-118 (Johnson and Matthey). m/z: 260.1 @ 1.04 min, Method 2.1H NMR (400 MHz, DMSO) δ 12.17 (br. s, 1H), 7.84 (s, 4H), 7.38 (d, J = 8.2 Hz, 1H), 7.32 (d, J = 8.3 Hz, 1H), 4.02 (s, 2H), 2.33 (s, 3H). Example 13: N-carbamimidoyl-2-(2,6-dimethyl-3-(pyrimidin-2-yl)phenyl)acetamide.TFA 20
The title compound was prepared from Intermediate 6 using Protocol 6 with 2- bromopyrimidine. 25 m/z: 284.2 @ 0.54 min, Method 3.1H NMR (400 MHz, DMSO) δ 11.61 (s, 1H), 8.91 (d, J = 4.9 Hz, 2H), 8.28 (s, 4H), 7.49 – 7.42 (m, 2H), 7.18 (d, J = 7.9 Hz, 1H), 3.97 (s, 2H), 2.31 (s, 3H), 2.28 (s, 3H). Example 14: N-carbamimidoyl-2-(2,6-dimethyl-3-(pyridin-2-yl)phenyl)acetamide.TFA 30
The title compound was prepared from Intermediate 6 using Protocol 6 with 2-bromopyridine.
m/z: 283.2 @ 0.27 min, Method 3.1H NMR (400 MHz, DMSO) δ 11.64 (s, 1H), 8.70 – 8.64 (m, 1H), 8.29 (s, 4H), 7.95 (t, J = 7.7 Hz, 1H), 7.48 (d, J = 7.9 Hz, 1H), 7.46 – 7.41 (m, 1H), 7.21 – 7.15 (m, 2H), 3.96 (s, 2H), 2.31 (s, 3H), 2.15 (s, 3H). 5 Example 15: N-carbamimidoyl-2-(2,4-dichloro-3'-methoxy-[1,1'-biphenyl]-3-yl)acetamide. HCOOH
10 The title compound was prepared from Intermediate 5 using Protocol 5 with (3- methoxyphenyl)boronic acid and PdCl2dppf in the Suzuki coupling. m/z: 352 @ 1.02 min, Method 3.1H NMR (400 MHz, DMSO) δ 8.17 (s, 1H), 7.49 (d, J = 8.3 Hz, 1H), 7.37 (t, J = 7.9 Hz, 1H), 7.27 (d, J = 8.4 Hz, 1H), 6.98 (ddd, J = 8.3, 2.6, 1.0 Hz, 1H), 6.94 (dt, J = 7.5, 1.3 Hz, 1H), 6.90 (dd, J = 2.6, 1.6 Hz, 1H), 3.93 (s, 2H), 3.79 (s, 3H). NH not 15 observed. Example 16: N-carbamimidoyl-2-(2,4-dichloro-3'-cyano-[1,1'-biphenyl]-3-yl)acetamide. HCOOH 20
The title compound was prepared from Intermediate 5 using Protocol 5 with (3- cyanophenyl)boronic acid and PdCl2dppf in the Suzuki coupling. m/z: 347 @ 0.93 min, Method 3.1H NMR (400 MHz, DMSO) δ 8.17 (s, 1H), 7.94 – 7.85 (m, 25 2H), 7.75 (dt, J = 7.9, 1.5 Hz, 1H), 7.68 (t, J = 7.7 Hz, 1H), 7.54 (d, J = 8.3 Hz, 1H), 7.34 (d, J = 8.3 Hz, 1H), 3.93 (s, 2H). NH not observed. Example 17: N-carbamimidoyl-2-(2,3',4-trichloro-4'-ethoxy-[1,1'-biphenyl]-3- yl)acetamide.TFA 30
The title compound was prepared from Intermediate 5 using Protocol 6 with (3-chloro-4- ethoxyphenyl)boronic acid and PdCl2dppf in the Suzuki coupling. m/z: 400 @ 1.18 min, Method 3.1H NMR (400 MHz, DMSO) δ 11.82 (s, 1H), 8.26 (s, 3H), 7.59 (d, J = 8.3 Hz, 1H), 7.47 – 7.38 (m, 2H), 7.32 (dd, J = 8.6, 2.2 Hz, 1H), 7.23 (d, J = 8.6 5 Hz, 1H), 4.21 (s, 2H), 4.16 (d, J = 7.0 Hz, 2H), 1.39 (t, J = 7.0 Hz, 3H). Example 18: N-carbamimidoyl-2-(6-chloro-2-methyl-3-(pyrimidin-5-yl)phenyl)acetamide. HCOOH 10
The title compound was prepared from Intermediate 7 using Protocol 5 with 5-pyrimidinyl boronic acid, potassium carbonate and Pd(PPh3)2Cl2 in the Suzuki coupling. 15 m/z: 304.2 @ 0.85 min, Method 2.1H NMR (400 MHz, DMSO) δ 9.22 (s, 1H), 8.81 (s, 2H), 8.19 (s, 1H), 7.41 (d, J = 8.3 Hz, 1H), 7.20 (d, J = 8.3 Hz, 1H), 3.84 (s, 2H), 2.17 (s, 3H). Example 19: 2-(3-acetamido-2,6-dichlorophenyl)-N-carbamimidoylacetamide.TFA 20
The title compound was prepared from Intermediate 8 using Protocol 3. m/z: 303.1 @ 0.67 min, Method 2.1H NMR (400 MHz, DMSO) δ 11.77 (s, 1H), 9.62 (s, 1H), 25 8.26 (s, 5H), 7.71 (d, J = 8.8 Hz, 1H), 7.50 (d, J = 8.8 Hz, 1H), 4.17 (s, 2H), 2.10 (s, 3H). Example 20: N-carbamimidoyl-2-(2,6-dichloro-3-(isopropylamino)phenyl)acetamide. HCOOH 30
The title compound was prepared from Intermediate 9 using Protocol 7.
m/z: 303.1 @ 0.67 min, Method 2. 1H NMR (400 MHz, DMSO) δ 7.05 (d, J = 8.7 Hz, 1H), 6.69 (d, J = 8.7 Hz, 1H), 5.39 (s, 2H), 3.76 (s, 2H), 3.35 (s, 3H). Example 21: N-carbamimidoyl-2-(2,6-dichloro-3-cyclopropylphenyl)acetamide. HCOOH 5
The title compound was prepared from Intermediate 5 using Protocol 5 with 10 cyclopropylboronic acid and PdCl2(dppf) in the Suzuki coupling m/z: 286.1 @ 0.90 min, Method 3.1H NMR (400 MHz, DMSO) δ 8.18 (s, 1H), 7.31 (d, J = 8.4 Hz, 1H), 6.94 (d, J = 8.5 Hz, 1H), 3.89 (s, 2H), 2.11 (tt, J = 8.5, 5.3 Hz, 1H), 1.08 – 0.84 (m, 2H), 0.78 – 0.55 (m, 2H). Exchangeable NH protons appear as two broad peaks which overlap with aromatic protons. 15 Example 22: N-carbamimidoyl-2-(2,4-dichloro-4'-methoxy-[1,1'-biphenyl]-3-yl)acetamide. HCOOH 20
The title compound was prepared from Intermediate 5 using Protocol 5 with (4- methoxyphenyl)boronic acid and PdCl2(dppf) in the Suzuki coupling m/z: 352 @ 1.04 min, Method 3.1H NMR (400 MHz, DMSO) δ 8.14 (s, 1H),7.52 (d, J = 8.4 25 Hz, 1H), 7.36 – 7.27 (m, 3H), 7.06 – 6.98 (m, 2H), 4.06 (s, 2H), 3.80 (s, 3H). 4 x NH peak appears as a very broad peak at 7.78 which overlaps with aromatic protons. Example 23: N-carbamimidoyl-2-(2,6-dichloro-3-(dimethylamino)phenyl)acetamide. HCOOH 30
The title compound was prepared from Intermediate 10 using Protocol 7
m/z: 289.1 @ 0.96 min, Method 2. 1H NMR (400 MHz, DMSO) δ 7.34 (d, J = 8.7 Hz, 1H), 7.08 (d, J = 8.8 Hz, 1H), 3.87 (s, 2H), 2.69 (s, 6H). (4 exchangeable Proton did not appear) Example 24: N-carbamimidoyl-2-(2,6-dichloro-3-(methylamino)phenyl)acetamide. 5 HCOOH
The title compound was prepared from Intermediate 10 using Protocol 7 10 m/z: 275 @ 0.67 min, Method 3.1H NMR (400 MHz, DMSO) δ 7.19 (d, J = 8.8 Hz, 1H), 6.53 (d, J = 8.9 Hz, 1H), 5.55 (q, J = 4.9 Hz, 1H), 3.79 (s, 2H), 2.74 (d, J = 4.8 Hz, 3H). (4 exchangeable Proton did not appear) Example 25: N-carbamimidoyl-2-(3'-methoxy-2,4-dimethyl-[1,1'-biphenyl]-3-yl)acetamide. 15 HCOOH
20 The title compound was prepared from Intermediate 11 using Protocol 5 with (3- methoxyphenyl)boronic acid m/z: 312.2 @ 1.02 min, Method 3.1H NMR (400 MHz, DMSO) δ 8.20 (s, 1H), 7.32 (t, 1H), 7.03 (d, J = 7.8 Hz, 1H), 6.93 (d, J = 7.7 Hz, 1H), 6.90 (ddd, J = 8.3, 2.6, 1.0 Hz, 1H), 6.80 (dt, J = 7.6, 1.2 Hz, 1H), 6.77 (dd, J = 2.6, 1.5 Hz, 1H), 3.77 (s, 3H), 3.66 (s, 2H), 2.29 (s, 3H), 2.13 25 (s, 3H). NH protons not observed. Example 26: N-carbamimidoyl-2-(4'-methoxy-2,4-dimethyl-[1,1'-biphenyl]-3-yl)acetamide. HCOOH 30
The title compound was prepared from Intermediate 11 using Protocol 5 with (4- methoxyphenyl)boronic acid
m/z: 312.2 @ 1.03 min, Method 3.1H NMR (400 MHz, DMSO) δ 8.20 (s, 1H), 7.21 – 7.12 (m, 2H), 7.01 (d, J = 7.8 Hz, 1H), 6.99 – 6.95 (m, 2H), 6.90 (d, J = 7.7 Hz, 1H), 3.79 (s, 3H), 3.64 (s, 2H), 2.28 (s, 3H), 2.12 (s, 3H). NH protons not observed. 5 Example 27: N-carbamimidoyl-2-(2,6-dimethyl-3-(prop-1-en-2-yl)phenyl)acetamide. HCOOH
10 The title compound was prepared from Intermediate 11 using Protocol 5 with potassium isopropenyltrifluoroborate m/z: 246.2 @ 0.92 min, Method 3.1H NMR (400 MHz, DMSO) δ 8.21 (s, 1H), 6.93 (d, J = 7.7 Hz, 1H), 6.83 (d, J = 7.7 Hz, 1H), 5.19 – 5.12 (m, 1H), 4.75 – 4.69 (m, 1H), 3.62 (s, 2H), 2.23 15 (s, 3H), 2.17 (s, 3H), 1.98 – 1.93 (m, 3H). NH protons not observed. Example 28: N-carbamimidoyl-2-(2,6-dichloro-3-(pyrimidin-2-yl)phenyl)acetamide.TFA 20
The title compound was prepared from Intermediate 12 using Protocol 6 with 2- bromopyrimidine m/z: 324.0 @ 0.62 min, Method 3.1H NMR (400 MHz, DMSO) δ 12.01 (s, 1H), 8.97 (d, J = 25 4.9 Hz, 2H), 8.37 (s, 4H), 7.67 (s, 2H), 7.58 (t, J = 4.9 Hz, 1H), 4.27 (s, 2H). Example 29: N-carbamimidoyl-2-(2,6-dichloro-3-(pyridin-4-yl)phenyl)acetamide. HCOOH 30
The title compound was prepared from Intermediate 5 using Protocol 5 with 4-(4,4,5,5- tetramethyl-1,3,2-dioxaborolan-2-yl)pyridine and PdCl2dppf in the Suzuki coupling
m/z: 323 @ 0.87 min, Method 3.1H NMR (400 MHz, DMSO) δ 8.70 – 8.64 (m, 2H), 8.15 (s, 1H), 7.56 (d, J = 8.3 Hz, 1H), 7.46 – 7.40 (m, 2H), 7.32 (d, J = 8.3 Hz, 1H), 3.92 (s, 2H). 4 exchangeable NH protons missing. Mono-formic acid salt. 5 Example 30: N-carbamimidoyl-2-(2,6-dichloro-3-(5-methoxypyridin-3- yl)phenyl)acetamide.HCOOH
10 The title compound was prepared from Intermediate 5 using Protocol 5 with 3-methoxy-5- (4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)pyridine and PdCl2dppf in the Suzuki coupling m/z: 353 @ 0.72 min, Method 3.1H NMR (400 MHz, DMSO) δ 8.15 (s, 1H), 7.52 (d, J = 8.3 Hz, 1H), 7.29 (d, J = 8.3 Hz, 1H), 3.90 (s, 2H), 2.25 (s, 3H), 2.06 (s, 3H). 4 exchangeable 15 protons missing. Mono-formic acid salt. Example 31: N-carbamimidoyl-2-(2,6-dichloro-3-(3,5-dimethylisoxazol-4- yl)phenyl)acetamide. HCOOH 20
The title compound was prepared from Intermediate 5 using Protocol 5 with 3,5-dimethyl-4- (4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)isoxazole and PdCl2dppf in the Suzuki coupling 25 m/z: 341.1 @ 0.87 min, Method 3.1H NMR (400 MHz, DMSO) δ 8.15 (s, 1H), 7.52 (d, J = 8.3 Hz, 1H), 7.29 (d, J = 8.3 Hz, 1H), 3.90 (s, 2H), 2.25 (s, 3H), 2.06 (s, 3H). 4 exchangeable protons missing. Mono-formic acid salt. Example 32: N-carbamimidoyl-2-(4-chloro-3'-methoxy-2-methyl-[1,1'-biphenyl]-3- 30 yl)acetamide
5 The title compound was prepared from Intermediate 7 using Protocol 5 with (3- methoxyphenyl)boronic acid and PdCl2dppf in the Suzuki coupling. m/z: 332.2 @ 1.37 min, Method 2.1H NMR (400 MHz, MeOD) δ 7.36 – 7.24 (m, 2H), 7.06 (d, J = 8.2 Hz, 1H), 6.91 (ddd, J = 8.3, 2.5, 1.0 Hz, 1H), 6.87 – 6.78 (m, 2H), 3.95 (s, 2H), 3.81 (s, 3H), 2.16 (s, 3H). 10 Example 33: N-carbamimidoyl-2-(2,6-dichloro-3-(1,2,3,6-tetrahydropyridin-4- yl)phenyl)acetamide. TFA 15
The title compound was prepared from Intermediate 5 using Protocol 6 with tert-butyl 4- (4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-3,6-dihydropyridine-1(2H)-carboxylate and PdCl2dppf in the Suzuki coupling. 20 m/z: 327.2 @ 0.90 min, Method 2.1H NMR (400 MHz, DMSO) δ 12.27 (s, 1H), 8.95 (s, 1H), 8.49 (s, 3H), 7.56 (d, J = 8.3 Hz, 1H), 7.24 (d, J = 8.3 Hz, 1H), 5.77 – 5.71 (m, 1H), 4.19 (s, 2H), 3.75 (bs, 2H), 3.31 (bs, 2H), 2.56 – 2.51 (m, 2H). Example 34: N-carbamimidoyl-2-(2,6-dichloro-3-(1H-indol-3-yl)phenyl)acetamide. 25 HCOOH
30 The title compound was prepared from Intermediate 5 using Protocol 6 with tert-butyl 3- (4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-1H-indole-1-carboxylateand PdCl2dppf in the Suzuki coupling.
m/z: 361.2 @ 1.26 min, Method 2.1H NMR (400 MHz, DMSO) δ 11.43 (s, NH), 8.18 (s, 1H), 7.56 (d, J = 2.6 Hz, 1H), 7.52 – 7.35 (m, 4H), 7.15 (ddd, J = 8.2, 6.9, 1.2 Hz, 1H), 7.05 (ddd, J = 8.0, 6.9, 1.1 Hz, 1H), 3.98 (d, J = 4.2 Hz, 2H).4NH not observed. 5 Example 35: N-carbamimidoyl-2-(2,6-dichloro-3-morpholinophenyl)acetamide. HCOOH
10 The title compound was prepared from Intermediate 13 using Protocol 7. m/z: 331@ 0.93 min, Method 2.1H NMR (400 MHz, DMSO) δ 8.16 (s, 1H), 7.37 (d, J = 8.6 Hz, 1H), 7.08 (d, J = 8.7 Hz, 1H), 3.85 (s, 2H), 3.76 – 3.70 (m, 4H), 2.96 – 2.89 (m, 4H).4NH not observed. 15 Example 36: 2-(3-(1-acetyl-1,2,3,6-tetrahydropyridin-4-yl)-2,6-dichlorophenyl)-N- carbamimidoylacetamide. HCOOH 20
The title compound was prepared from Intermediate 5 using Protocol 6 with 1-(4-(4,4,5,5- tetramethyl-1,3,2-dioxaborolan-2-yl)-3,6-dihydropyridin-1(2H)-yl)ethan-1-one and PdCl2dppf in the Suzuki coupling. m/z: 371.3 @ 0.92 min, Method 2.1H NMR (400 MHz, DMSO) δ 8.16 (s, 1H), 7.40 (d, J = 8.3 25 Hz, 1H), 7.14 (d, J = 8.3 Hz, 1H), 5.69 (s, 1H), 4.16 – 4.08 (m, 1H), 4.07 – 4.00 (m, 1H), 3.87 (s, 2H), 3.62 (dt, J = 11.6, 5.6 Hz, 2H), 2.43 – 2.38 (m, 1H), 2.33 – 2.28 (m, 1H), 2.15 – 2.00 (m, 3H).4NH not observed. Example 37: N-carbamimidoyl-2-(2,6-dichloro-3-isobutylphenyl)acetamide. HCOOH 30
The title compound was prepared from Intermediate 14 using Protocol 7.
m/z: 302.2 @ 1.35 min, Method 2.1H NMR (400 MHz, DMSO) δ 8.15 (s, 1H), 7.35 (d, J = 8.3 Hz, 1H), 7.19 (d, J = 8.3 Hz, 1H), 3.88 (s, 2H), 2.57 (d, J = 7.2 Hz, 2H), 1.88 (dq, J = 13.0, 6.6 Hz, 1H), 0.88 (d, J = 6.6 Hz, 6H). (exchangeable NH signals were not observed). 5 Example 38: N-carbamimidoyl-2-(2-chloro-3'-methoxy-4-methyl-[1,1'-biphenyl]-3- yl)acetamide. HCOOH 10
The title compound was prepared from Intermediate 15 using Protocol 5 with (3- methoxyphenyl)boronic acid and PdCl2dppf in the Suzuki coupling. m/z: 332.2 @ 1.31 min, Method 2.1H NMR (400 MHz, DMSO) δ 8.18 (s, 1H), 7.34 (t, J = 7.9 15 Hz, 1H), 7.19 (d, J = 7.7 Hz, 1H), 7.12 (d, J = 7.9 Hz, 1H), 6.98 – 6.85 (m, 3H), 3.84 – 3.79 (m, 2H), 3.78 (s, 3H), 2.30 (s, 3H). (exchangeable NH protons were not observed) Example 39: N-carbamimidoyl-2-(2-chloro-6-methyl-3-(pyrimidin-5-yl)phenyl)acetamide. HCOOH 20
25 The title compound was prepared from Intermediate 15 using Protocol 5 with pyrimidin-5- ylboronic acid and PdCl2dppf in the Suzuki coupling. m/z: 304.2 @ 0.78 min, Method 2. 1H NMR (400 MHz, DMSO) δ 9.21 (s, 1H), 8.87 (s, 2H), 8.14 (s, 1H), 7.31 (s, 2H), 3.87 (s, 2H), 2.35 (s, 3H). (exchangeable NH protons were not observed) 30 Example 40: 2-(2'-acetamido-2,4-dichloro-[1,1'-biphenyl]-3-yl)-N- carbamimidoylacetamide. HCOOH
5 The title compound was prepared from Intermediate 5 using Protocol 5 with N-(2-(4,4,5,5- tetramethyl-1,3,2-dioxaborolan-2-yl)phenyl)acetamide and PdCl2dppf in the Suzuki coupling. m/z: 379.2 @ 1.03 min, Method 2. 1H NMR (400 MHz, DMSO) δ 8.91 (s, 1H), 8.16 (s, 1H), 7.58 (d, J = 8.0 Hz, 1H), 7.48 (dd, J = 8.2, 2.3 Hz, 1H), 7.42 – 7.33 (m, 1H), 7.24 (t, J = 7.4 Hz, 10 1H), 7.21 – 7.10 (m, 2H), 3.91 (s, 2H), 1.84 (s, 3H).4NH not observed. Example 41: N-carbamimidoyl-2-(2,6-dichloro-3-(pyridin-2-yl)phenyl)acetamide. TFA 15
The title compound was prepared from Intermediate 12 using Protocol 6 with 2-bromopyridine m/z: 323 @ 0.64 min, Method 3.1H NMR (400 MHz, DMSO) δ 11.70 (br s, 1H), 8.72 – 8.66 (m, 1H), 8.24 (s, 4H), 7.93 (td, J = 7.8, 1.9 Hz, 1H), 7.68 – 7.57 (m, 2H), 7.54 (d, J = 8.4 Hz, 20 1H), 7.46 (ddd, J = 7.6, 4.8, 1.1 Hz, 1H), 4.26 (s, 2H). Example 42: N-carbamimidoyl-2-(2,6-dichloro-3-(1H-pyrazol-4-yl)phenyl)acetamide, HCl 25
The title compound was prepared from Intermediate 5 using Protocol 2 with 1-(tetrahydro-2H- pyran-2-yl)-4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-1H-pyrazole and Pd-118 (Johnson and Matthey) 30 m/z: 312 @ 0.66 min, Method 3 1H NMR (500 MHz, DMSO) δ 12.49 (s, 1H), 8.64 – 8.18 (m, 4H), 8.04 (s, 2H), 7.58 (d, J = 8.4 Hz, 1H), 7.51 (d, J = 8.5 Hz, 1H), 4.22 (s, 2H), NH not observed.
Example 43: N-carbamimidoyl-2-(2,6-dichloro-3-(4-cyano-1-methyl-1H-pyrazol-5- yl)phenyl)acetamide, HCl 5
The title compound was prepared from Intermediate 12 using Protocol 5 with 5-bromo-1- methyl-1H-pyrazole-4-carbonitrile and Pd-170 (Johnson and Matthey) m/z: 351 @ 0.70 min, Method 3 10 1H NMR (400 MHz, DMSO) δ 12.05 – 11.85 (m, 1H), 8.34 – 8.16 (m, 5H), 7.79 (d, J = 8.3 Hz, 1H), 7.66 (d, J = 8.3 Hz, 1H), 4.28 (s, 2H), 3.69 (s, 3H) Example 44: N-carbamimidoyl-2-(6-chloro-2-cyano-3-cyclopropylphenyl)acetamide, HCl 15
The title compound was prepared from Intermediate 16 using Protocol 7 m/z: 277 @ 0.81 min, Method 3 20 1H NMR (400 MHz, DMSO) δ 12.12 (s, 1H), 8.26 (s, 4H), 7.71 (d, J = 8.6 Hz, 1H), 7.10 (d, J = 8.6 Hz, 1H), 4.19 (s, 2H), 2.18 (tt, J = 8.3, 5.0 Hz, 1H), 1.19 – 1.12 (m, 2H), 0.89 – 0.81 (m, 2H). The following examples were prepared according to similar procedures as those described for Examples above: 25 Table 1:
II. BIOLOGY EXAMPLES 5 II.1. ASSAYS FOR ADRENERGIC a2 FUNCTIONAL ACTIVITIES II.1.A. Stimulation of GTP binding in CHO cells Materials 10
CHO-K1 cells stably transfected with plasmid containing the human sequence for adrenergic receptors: α2A, α2B and α2C were purchased from Perkin Elmer (EC-030-C, ES-31-C and ES- 032-C, respectively). Cells were maintained in Ham’s F12 (11765054, Gibco) with addition of 10 % (v/v) FBS and 0.4 mg/ml Geneticin (10131027, Gibco) at 37 °C, 5 % CO2 and passaged 5 twice weekly. HTRF GTP binding kit was purchased from Perkin Elmer (62GTPPEC). Methods Membrane preparations 10 Cells were detached in Versene for up to 15 minutes with agitation. All collected cells were centrifuged at 300 xg for 5 minutes (RT) and supernatants were discarded. Cell pellets were resuspended in ice-cold HE buffer: 20 mM HEPES (H0887, Merck), 10 mM EDTA (E7889, Merck), pH 7.4. The cell suspension was then homogenised using a Dounce homogeniser with 20x strokes of pestle A, followed by 20x strokes with pestle B. Homogenates were centrifuged 15 for 600 xg for 10 minutes at 4 °C. The supernatants were then transferred to high-speed centrifuge tubes (NC654, Appleton Woods). Pellets were again resuspended in ice-cold HE buffer: 20 mM HEPES (H0887, Merck), 10 mM EDTA (E7889, Merck), pH 7.4and homogenised using a Dounce homogeniser with 20x strokes of pestle A, followed by 20x strokes with pestle B, then centrifuged for 600 xg for 10 minutes at 4 °C. Supernatants were 20 pooled and centrifuged at 40,000xg for 1 h at 4 °C. Each pellet was resuspended in 400 µl of 20 mM HEPES with 10 % (w/v) glycerol (332031000, ThermoScientific). All steps were performed on ice. Membrane suspensions were passed 10x through the insulin syringe (324892, BD Microfine), aliquoted and stored at -80 °C until further use. Protein content was established using the BCA kit (23225, ThermoFisher) with bovine serum albumin standards. 25 HTRF GTP binding assay To evaluate the potential agonists of α2 adrenergic receptors (α2A, α2B and α2C) HTRF GTP binding kit was used with some modification to the manufacturer’s protocol. Tested compounds were dispensed into 384 white ProxiPlates Plus (6008280, Perkin Elmer) using an ECHO acoustic dispenser with automatic DMSO backfill. Guanfacine (HU-17416, Medchemtronica 30 AB) was used as a reference compound. All compounds were run in duplicates on each plate. For plate controls, 30 µM norepinephrine (N5785, Merck) and vehicle control (DMSO) were used. Then, the assay mix was prepared by mixing stimulation buffer #3, MgCl2, 1x GTP Eu Cryptate reagent, 1x GTP d2 antibody and membrane preparation. MgCl2 concentration per well was 50 mM for α2A and α2C, 8 mM for α2B. The amount of membrane proteins was
established for each membrane preparation in a validation experiment (range 0.5-2 µg of protein per well). 10 µl of assay mix was added per well and assay plates were briefly centrifuged. Then plates were sealed with the optically clear plate seals (4311971, Applied Biosystems) and incubated for 6 h at room temperature. Plates were read on Pherastar FSX (BMG Labtech) using 5 the Pherastar HTRF module. Fluorescence was read at two channels: 665 nm and 620 nm and the ratio between channels was calculated. Data was expressed as the percentage activation versus 30 µM norepinephrine. Results. As evidenced in Table 2 below, the compounds of the present invention stimulate GTP 10 binding on a2-overexpressing CHO cells. Table 2:
ND: Not Determined
III. PHARMACOKINETIC EXAMPLES III.1. DETERMINATION OF PERMEABILITY AND EFFLUX IN CACO-2 CELLS 5 As mentioned in the introduction, the compounds of the invention have to exhibit a limited, if any, CNS penetrance, in order to avoid deleterious side effects that can occur if these compounds penetrate significantly into the brain. The present assay aims at showing that the compounds of the invention do not have any 10 significant CNS penetrance by showing that they are substrates of transporters that efflux them from brain. Indeed, it is well known in the art that xenobiotics that are substrates of transporters such as P- Glycoprotein are not efficient in penetrating the Blood-Brain Barrier, and are thus less effective 15 in the Central Nervous System (Alfred H. Schinkel, “P-Glycoprotein, a gatekeeper in the blood–brain barrier”, Advanced Drug Delivery Reviews 36 (1999) 179–194). The present assay thus aims at showing that the compounds of the present invention are substrates to such transporters present in the Caco-2 cell line and thus do not cross the Blood- Brain Barrier. 20 Protocol Summary The permeability of test compounds across confluent Caco-2 monolayers were assessed using 96-well Transwell plates by adding compound into the apical and basolateral compartments (bidirectional) and determining test compound levels in the opposing compartment after 2 hours 25 at 37°C in a CO2 incubator. Following sampling at T=0 and 2hrs from both compartments, the samples are analysed by UPLC-MS. Experimental Procedure The Caco-2 Permeability assay used cells purchased from ECACC (86010202) and cultured 30 inhouse. Caco-2 cells are plated on a 96-transwell permeable system as a single monolayer and allowed to differentiate for 21 days to phenotypically mimic intestinal epithelial cells. Dilutions of a 10 mM test compound DMSO stock solution were prepared in buffer (supplemented HBSS, pH6 or 7.4 for apical; supplemented HBSS, pH7.4 for basolateral)) so that the final DMSO concentration was 0.1% and the final test compound concentration tested was 10 uM. Following
addition of test compound (in duplicate) into either the apical or basolateral compartment, the samples were incubated for 2 hours at 37°C in a CO2 incubator. Aliquots were sampled at T=0 and 2hrs and mixed with acetonitrile (containing internal standard). All the samples were mixed, centrifuged and the supernatants analysed by UPLC-MS with quantification against a 5 calibration line prepared in matched HBSS buffer. Lucifer Yellow permeability was performed during the incubation period to assess monolayer integrity. Data Analysis Test Compound was quantified in each compartment and concentrations determined and against 10 a calibration curve prepared in matched HBSS buffer. The apparent permeability coefficient (Papp) was calculated using the following equation: Papp=VR/(Area*Time)*(CR/C0) wherein VR is the solution volume in the receiver chamber; Area is the surface area for the transport; Time is incubation time, expressed in seconds; C0 is the initial concentration in the 15 donor chamber; CR is the final concentrations in receiver chamber. The efflux ratio was calculated using the following equation: Efflux ratio = Papp (A-B)/Papp(B-A) where Papp (A-B) and Papp (B-A) are the Papp values of compound in Apical to Basolateral and Basolateral to Apical directional transport, respectively. 20 Results. Compounds are considered to be P-Glycoprotein substrates when the value of efflux ratio is > 3. Compounds of the present invention typically have efflux ratios > 3 whereas guanfacine has an efflux ratio < 3 (1.5), as evidenced in Table 3 below. 25 Table 3:
III.2. DETERMINATION OF THE CONCENTRATION A2A ANTAGONISTS IN BRAIN AND CEREBROSPINAL FLUID COMPARED TO PLASMA The present assay aims at showing that the compounds of the invention do not have any 5 significant CNS penetrance by determining the concentration of these compounds in brain and cerebrospinal fluid (CSF) compared to plasma. Method. 7-9 weems old female Balb-c mice (obtained from SLAC Laboratory Animal Co. Ltd., Shanghai, China or SIPPR-B&K Laboratory Animal Co. Ltd., Shanghai, China) were dosed orally at a dose of 10 mg/kg, as a 1.00 mg/mL suspension in 10 10%DMSO+10%solutol+80% water, adjusted to pH 3-4 (homogenous opaque suspension). Animals were fasted at least 12 hours prior to the administration. All animals had access to Certified Rodent Diet (Catalog # M01-F, SLAC Laboratory Animal Cl. Ltd., Shanghai, China) ad libitum 4 hours post dosing. Serial bleeding (about 30 μL blood per time point) were performed from submandibular or saphenous vein. Those samples were transferred into 15 prechilled microcentrifuge tubes containing 2 μL of K2EDTA (0.5M) as anti-coagulant and placed on wet ice for further treatment. Immediately after blood collection, the whole brain was harvested immediately at the designed time points. At selected timepoints post-dose, CSF was collected from cisterna magna. Blood samples were processed for plasma by centrifugation at approximately 4 °C, 3000g 15 20 min within half an hour of collection. Plasma samples were stored in polypropylene tubes, quick frozen over dry ice and kept at -70 °C until LC/MS/MS analysis. Brain samples were weighed, rinsed in cold distilled water to remove blood, and homogenized using pre-cooled water at the ratio of 1:4 (1 g brain used 4mL water). And the brain homogenization was kept at -70 °C until LC/MSMS analysis. CSF was quick frozen over dry ice and kept at -70 °C until LC/MS/MS 25 analysis. Results. Examples 7, 42, and 43 of the invention were dosed at 5 mg/kg by oral route and concentrations in brain or CSF of mice were found to be significantly lower compared to the concentrations present in plasma (brain/plasma ratio <0.1).
Individual and Mean Concentration of Example 7 after PO Administration (5 mg/kg) are provided in Table 4 below. Table 4:
5 Individual and Mean Concentration of Example 42 after PO Administration (5 mg/kg) are provided in Table 4 below. Table 5:
Individual and Mean Concentration of Example 43 after PO Administration (5 mg/kg) are provided in Table 6 below. Table 6:
Claims
CLAIMS 1. A compound of formula (I):
wherein: ^ R1 represents a radical selected in a group consisting of: • a hydrogen, • a (C1-C6)alkyl optionally substituted by a (C1-C6)alkyloxy, a hydroxy, at least one halogen, and a cycloalkyl, • a halogen, • a cyano, • a 3-14 membered ring, saturated, partially unsaturated, or unsaturated selected in a group consisting of a cycloalkyl, a heterocycloalkyl, an aryl, and an heteroaryl, said 3-14 membered ring is optionally substituted by a (C1-C6)alkyl, a hydroxy, a halogen, a -SO2-(C1-C6)alkyl, and a (C1-C6)alkyloxy, and • a (C1-C6)alkyloxy optionally substituted by at least one halogen; ^ R2 represents a radical selected in a group consisting of: • a hydrogen, • a (C1-C6)alkyl optionally substituted by a (C1-C6)alkyloxy, a hydroxy, at least one halogen, and a cycloalkyl, • a halogen, • a cyano, • a 3-14 membered ring, saturated, partially unsaturated, or unsaturated selected in a group consisting of a cycloalkyl, a heterocycloalkyl, an aryl, and an heteroaryl, said 3-14 membered ring is optionally substituted by a (C1-C6)alkyl, a hydroxy, a halogen, a -SO2-(C1-C6)alkyl, and a (C1-C6)alkyloxy, and • a (C1-C6)alkyloxy optionally substituted by at least one halogen; ^ X is selected from the group consisting of -(CH2)m- with m is from 1 to 3 optionally substituted by at least one Ra, -O-, -S-, -CO-, -C(O)-O-, -O-C(O)-, -NRa-, -CO-NRa-, -
NRa-CO-, -SO-, -SO2-, -SO2-NRa, -NRa-SO2-, and -NRa-CO-NRa-, with Ra being a hydrogen, a (C1-C6)alkyl optionally substituted by at least one radical C, a cycloalkyl optionally substituted by at least one radical C, or a heterocycloalkyl optionally substituted by at least one radical C; said radical C being selected in a group consisting of: - a (C1-C6)alkyloxy, - a halogen, - a -O-C(O)-(C1-C6)alkyl, a -C(O)-O-(C1-C6)alkyl, a - COOH, - a -O-C(O)-cycloalkyl, a -C(O)-O-cycloalkyl, - a -CO-(C1-C6)alkyl, - a -CO-cycloalkyl, - a hydroxy, - a -NRbRc with Rb and Rc represent independently a hydrogen, a (C1-C6)alkyl, a cycloalkyl, or Rb and Rc may form together a N-heterocycloalkyl, - a nitro, - a cyano, - a -NH-CO-(C1-C6)alkyl, a -CO-NH-(C1-C6)alkyl, - a -NH-CO-cycloalkyl, a -CO-NH-cycloalkyl, - a -NH-SO2-(C1-C6)alkyl, a -SO2-NH-(C1-C6)alkyl, a - NH-SO2-cycloalkyl, a -SO2-NH-cycloalkyl, - a -SO2-(C1-C6)alkyl, a -SO2-cycloalkyl, - a -SO-(C1-C6)alkyl, and a -SO-cycloalkyl; ^ n is 0 or 1; ^ R3 represents a radical selected in a group consisting of: • a 3-14 membered ring, saturated, partially unsaturated, or unsaturated, selected in a group consisting of an aryl, a heteroaryl, a cycloalkyl, and a heterocycloalkyl, said 3-14 membered ring is optionally substituted by at least one radical A selected in a group consisting of: - a (C1-C6)alkyl optionally substituted by a halogen, a (C1-C6)alkyloxy, or a hydroxy, - a cycloalkyl optionally substituted by a halogen, a (C1- C6)alkyloxy, a hydroxy, or a -CO-NH2,
- a heterocycloalkyl optionally substituted by a halogen, a (C1-C6)alkyloxy, or a hydroxy, - a (C2-C6)alkenyl, - a (C2-C6)alkynyl, - a (C1-C6)alkyloxy optionally substituted by a halogen, - a halogen, - a cyano, - a -NRdRe with Rd and Re represent independently a hydrogen, a (C1-C6)alkyl, a cycloalkyl, or Rd and Re may form together a N-heterocycloalkyl, - a nitro, - a hydroxy, - a ketone, - a -NH-CO-(C1-C6)alkyl, a -CO-NH-(C1-C6)alkyl, a - CO-N((C1-C6)alkyl)2, a -NH-CO-cycloalkyl, a -CO- NH-cycloalkyl, - a -NH-SO2-(C1-C6)alkyl, a -SO2-NH-(C1-C6)alkyl, a - NH-SO2-cycloalkyl, a -SO2-NH-cycloalkyl, - a -C(O)-O-(C1-C6)alkyl, a -O-C(O)-(C1-C6)alkyl, a - COOH, a -C(O)-O-cycloalkyl, -O-C(O)-cycloalkyl, - a -CO-(C1-C6)alkyl, a -CO-cycloalkyl, - a -SO2-(C1-C6)alkyl, a -SO2-cycloalkyl, - a -SO-(C1-C6)alkyl, and a -SO-cycloalkyl, and • a radical selected in a group consisting of: o a (C1-C6)alkyl optionally substituted by at least one radical B or a cycloalkyl optionally substituted by at least one radical B, o a (C2-C6)alkenyl optionally substituted by at least one radical B, o a (C2-C6)alkynyl optionally substituted by at least one radical B, o a (C1-C6)alkyloxy optionally substituted by at least one radical B, o a hydroxy, o a cyano, o a halogen, o a -NRf’Rg’ with Rf’ and Rg’, represent independently a hydrogen, a (C1-C6)alkyl optionally substituted by at least one radical B, a
cycloalkyl optionally substituted by at least one radical B, a heterocycloalkyl optionally substituted by at least one radical B, with the proviso that when one of Rf’ or Rg’ is a hydrogen, then the other is not a hydrogen, or Rf’ and Rg’ may form together a N- heterocycloalkyl, and o a -N(Rf)-CO-Rg, a-CO-N(Rf)-Rg, a -CO-Rf, a -C(O)-O-Rf, a -O- C(O)-Rf, a -SO2-Rf, a -SO-Rf, a -SO2-N(Rf)-Rg, a -N(Rf)-SO2-Rg, and a -N(Rh)-CO-NRfRg, with Rf, Rg, and Rh represent independently a hydrogen, a (C1-C6)alkyl optionally substituted by at least one radical B, a cycloalkyl optionally substituted by at least one radical B, a heterocycloalkyl optionally substituted by at least one radical B, or Rf and Rg may form together a N- heterocycloalkyl, said radical B being selected in a group consisting of: - a (C1-C6)alkyloxy, - a (C1-C6)alkyl optionally substituted by a hydroxy, - a halogen, - a -O-C(O)-(C1-C6)alkyl optionally substituted by at least one halogen, a -C(O)-O-(C1-C6)alkyl, a -COOH, - a -O-C(O)-cycloalkyl, a -C(O)-O-cycloalkyl, - a -CO-(C1-C6)alkyl, - a cycloalkyl, - a heterocycloalkyl, - a -CO-cycloalkyl, - a hydroxy, - a -NRiRj with Ri and Rj represent independently a hydrogen, a (C1-C6)alkyl, a cycloalkyl, or Ri and Rj may form together a N-heterocycloalkyl, - a nitro, - a cyano, - an aryl, - a heteroaryl, - a -NH-CO-(C1-C6)alkyl, a -CO-NH-(C1-C6)alkyl, - a -NH-CO-cycloalkyl; -CO-NH-cycloalkyl,
- a -NH-SO2-(C1-C6)alkyl, a -SO2-NH-(C1-C6)alkyl, - a -NH-SO2-cycloalkyl, a -SO2-NH-cycloalkyl, - a -SO2-(C1-C6)alkyl, a -SO2-cycloalkyl, - a -SO-(C1-C6)alkyl, and a -SO-cycloalkyl; ^ R4 represents a radical selected in a group consisting of: • a hydrogen, • a halogen, • a (C1-C6)alkyl optionally substituted by a halogen, a (C1-C6)alkyloxy, a hydroxy, or a cycloalkyl, • a (C1-C6)alkyloxy optionally substituted by a halogen, a (C1-C6)alkyloxy, a hydroxy, or a cycloalkyl, • a hydroxy, • a -NRkRl with Rk and Rl represent independently a hydrogen, a (C1-C6)alkyl, a cycloalkyl, or Rk and Rl may form a N-heterocycloalkyl, • a nitro, • a cyano, • a -NH-(C1-C6)alkyl, a -NH-cycloalkyl; and • a -NH-CO-(C1-C6)alkyl, and a -NH-CO-cycloalkyl; ^ R5 represents a radical selected in a group consisting of: • a hydrogen, • a halogen, • a (C1-C6)alkyl optionally substituted by a halogen, a (C1-C6)alkyloxy, a hydroxy, or a cycloalkyl, • a (C1-C6)alkyloxy optionally substituted by a halogen, a (C1-C6)alkyloxy, a hydroxy, or a cycloalkyl, • a hydroxy, • a -NRkRl with Rk and Rl represent independently a hydrogen, a (C1-C6)alkyl, a cycloalkyl, or Rk and Rl may form a N-heterocycloalkyl, • a nitro, • a cyano, • a -NH-(C1-C6)alkyl, a -NH-cycloalkyl; and • a -NH-CO-(C1-C6)alkyl, and a -NH-CO-cycloalkyl;
^ R6 represents a hydrogen, or a (C1-C6)alkyl optionally substituted by a radical selected in a group consisting of a hydroxy, a (C1-C6)alkyloxy, a -NH(C1-C6)alkyl, and a -N((C1- C6)alkyl)2; ^ R7 represents a hydrogen, or a (C1-C6)alkyl optionally substituted by a radical selected in a group consisting of a hydroxy, a (C1-C6)alkyloxy, a -NH(C1-C6)alkyl, and a -N((C1- C6)alkyl)2; and ^ R8 represents a hydrogen, or a (C1-C6)alkyl optionally substituted by a radical selected in a group consisting of a hydroxy, a (C1-C6)alkyloxy, a -NH(C1-C6)alkyl, and a -N((C1- C6)alkyl)2; or R7 and R8 may form together a 5-7 membered ring, partially unsaturated or saturated, N,N’-heterocycloalkyl optionally substituted by at least one radical selected in a group consisting of a (C1-C6)alkyl, a hydroxy, a (C1-C6)alkyloxy, a -NH(C1-C6)alkyl, a - N((C1-C6)alkyl)2, and a ketone; and the isomers, stereoisomers and pharmaceutical acceptable salts thereof.
2. The compound according to claim 1, wherein n is 0.
3. The compound according to claim 1 or 2, wherein R4 and R5 represent H.
4. The compound according to any one of claims 1 to 3, wherein R6, R7, and R8 represent H.
5. The compound according to any one of claims 1 to 4, wherein: ^ R1 represents a (C1-C6)alkyl, preferably a methyl, a halogen, preferably a chlorine or a fluorine, more preferably a chlorine; and ^ R2 represents a (C1-C6)alkyl, preferably a methyl, a halogen, preferably a chlorine or a fluorine, more preferably a chlorine, or a cyano.
6. The compound according to any one of claims 1 to 5, wherein, R3 represents a 3-14 membered ring, saturated, partially unsaturated, or unsaturated, selected in a group consisting of an aryl, a heteroaryl, a cycloalkyl, and a heterocycloalkyl, said 3-14 membered ring is optionally substituted by at least one radical A selected in a group consisting of: - a (C1-C6)alkyl optionally substituted by a halogen, a hydroxy, a cycloalkyl, a -CO-NH((C1-C6)alkyl), or a - CO-N((C1-C6)alkyl)2,
- a heterocycloalkyl optionally substituted by a halogen, a (C1-C6)alkyloxy, or a hydroxy, - a cycloalkyl - a (C1-C6)alkyloxy optionally substituted by a halogen, - a halogen, - a -NRdRe with Rd and Re represent a hydrogen, a (C1- C6)alkyl, or a cyclopropyl, - a cyano, - a hydroxy, - a ketone, - a -CO-NH2, - a -COOH, - a -CO-(C1-C6)alkyl, - a -C(O)-O-(C1-C6)alkyl, - a -SO2-(C1-C6)alkyl, and - a -NH-CO-(C1-C6)alkyl.
7. The compound according to any one of claims 1 to 6, wherein R3 represents a 3-14 membered ring, saturated, partially unsaturated, or unsaturated, selected in a group consisting of: o an aryl, particularly a phenyl, said aryl is optionally substituted by at least one radical selected in a group consisting of: - a (C1-C6)alkyl, particularly a methyl, - a (C1-C6)alkyloxy, particularly a methoxy or an ethoxy, - a cyano, - a hydroxy, - a halogen, particularly a chlorine, - a -NRdRe with Rd and Re represent a hydrogen, and - a -NH-CO-(C1-C6)alkyl, particularly a -NH-CO-CH3, o a heteroaryl, particularly a pyrimidinyl, a pyrazinyl, a pyridazinyl, a pyrazolyl, an isoxazolyl, a pyridinyl, an indolyl, an imidazolyl, a triazolyl, an indazolyl, a thiazolyl, a thiophenyl, a benzoxazolyl, or a benzofuranyl, said heteroaryl is optionally substituted by at least one radical selected in a group consisting of:
- a (C1-C6)alkyl, particularly a methyl, an ethyl, an isopropyl, or a tert- butyl, said (C1-C6)alkyl is optionally substituted by at least one halogen, a hydroxy, or a cyclopropyl, - a (C1-C6)alkyloxy, particularly a methoxy, - a cyano, - a halogen, particularly a chlorine, a bromine, or a iodine, - a -NRdRe with Rd and Re represent a hydrogen, - a tetrahydropyranyl, - a cyclopropyl, - a hydroxy, and - a ketone, o a cycloalkyl, particularly a cyclopropyl, said cycloalkyl is optionally substituted by a -CO-NH2, a -COOH, a -C(O)-O-(C1-C6)alkyl, a (C1- C6)alkyl optionally substituted by a hydroxy, a -CO-CH3, a -CO- NH(CH3), or a -CO-N(CH3)2, and o a heterocycloalkyl, particularly a 1,2,3-6-tetrahydropyridinyl, a morpholinyl, a pyrrolidinyl, an imidazolidinyl, a 3,4-dihydro-2H- pyranyl, a tetrahydropyranyl, a tetrahydrofuranyl, a morpholinyl, an azetidinyl, or an oxetanyl, said heterocycloalkyl is optionally substituted by a -CO-(C1-C6)alkyl, particularly a -CO-CH3, a (C1-C6)alkyl optionally substituted by at least one halogen, particularly a methyl or a trifluromethyl, a (C1-C6)alkyloxy, particularly a methoxy, a halogen, particularly a fluorine, a -NRdRe with Rd and Re represent a hydrogen, a ketone, a -SO2-(C1-C6)alkyl, or a hydroxy.
8. The compound according to any one of claims 1 to 5, wherein R3 represents a radical selected in a group consisting of: o a (C1-C6)alkyl, particularly a methyl, an ethyl, an isopropyl, or an isobutyl, said (C1-C6)alkyl is optionally substituted by a (C1-C6)alkyloxy, particularly a methoxy, a hydroxy, at least one halogen, or a -NH-CO-(C1-C6)alkyl, particularly a -NH-CO-CH3, o a (C2-C6)alkenyl, particularly an isopropenyl, o a (C2-C6)alkynyl optionally substituted by a heteroaryl, particularly a pyrazolyl, or a (C1-C6)alkyl optionally substituted by a hydroxy,
o a (C1-C6)alkyloxy, particularly a methoxy, an ethoxy, an isopropoxy, or a butoxy, said (C1-C6)alkyloxy is optionally substituted by a phenyl, a hydroxy, or by at least one halogen, o a hydroxy, o a phenoxy, o a halogen, particularly a chlorine or an iodine, o a cyano, o a -NRf’Rg’ with Rf’ and Rg’ represent independently a hydrogen or a (C1-C6)alkyl, particularly a methyl or an isopropyl, optionally substituted by a cycloalkyl or a heterocycloalkyl, with the proviso that when one of Rf’ or Rg’ is a hydrogen, then the other is not a hydrogen, o a -NH-CO-(C1-C6)alkyl, particularly a -NH-CO-CH3, o a -NH-SO2-(C1-C6)alkyl, particularly a -NH-SO2-CH3, o a -SO2-Rf with Rf represents a (C1-C6)alkyl, particularly a methyl, and o a -N(Rh)-CO-NRfRg, a -N(Rf)-CO-Rg, a-CO-N(Rf)-Rg, or a -N(Rf)-SO2-Rg, with Rf, Rg, and Rh represent independently a hydrogen, or a methyl.
9. The compound according to claim 1, wherein said compound has the following formula selected in a group consisting of:
Cl , 5
, , and and a pharmaceutical acceptable salt thereof.
10. The compound according to any one of claims 1 to 9, for use as a medicine.
11. A pharmaceutical composition comprising a compound as defined in any one of claims 1 to 9, and a pharmaceutically acceptable excipient.
12. The pharmaceutical composition according to claim 11, for use for treating a cancer.
13. The pharmaceutical composition for use according to claim 12, wherein the cancer is selected in a group consisting of myelofibrosis, acute lymphoblastic leukemia, acute myeloblastic leukemia adrenal gland carcinoma, bile duct cancer, bladder cancer, breast cancer, cervical cancer, colorectal cancer, endometrial cancer, esophageal cancer, gastric cancer, gastrointestinal stromal tumors, glioblastoma, head and neck cancer, hepatocellular carcinoma, Hodgkin’s lymphoma, kidney cancer, lung cancer, melanoma, Merkel cell skin cancer, mesothelioma, multiple myeloma, myeloproliferative disorders, non-Hodgkin lymphoma, ovarian cancer, pancreatic cancer, prostate cancer, salivary gland cancer, sarcoma, squamous cell carcinoma, testicular cancer, thyroid cancer, urothelial carcinoma, and uveal melanoma.
14. The pharmaceutical composition for use according to claim 12 or 13, wherein the compound is administered at a dose ranging from 0.001 mg/kg body weight to 30 mg/kg body weight.
15. The pharmaceutical composition for use according to any one of claims 12 to 14, wherein said pharmceutical composition is administered in combination with another antitumoral drug,
especially chemotherapy, immunotherapy, hormonotherapy and/or radiotherapy, preferably immunotherapy.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP23200710.4 | 2023-09-29 | ||
| EP23200710 | 2023-09-29 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2025068452A1 true WO2025068452A1 (en) | 2025-04-03 |
Family
ID=88296980
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2024/077199 Pending WO2025068452A1 (en) | 2023-09-29 | 2024-09-27 | Guanfacine derivatives and their use in treating cancer |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2025068452A1 (en) |
Citations (46)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4602041A (en) * | 1982-02-10 | 1986-07-22 | Beecham Group P.L.C. | Benzylideneamino- and phenylacetylguanidines and their pharmaceutical uses |
| WO2006121168A1 (en) | 2005-05-09 | 2006-11-16 | Ono Pharmaceutical Co., Ltd. | Human monoclonal antibodies to programmed death 1(pd-1) and methods for treating cancer using anti-pd-1 antibodies alone or in combination with other immunotherapeutics |
| WO2008132601A1 (en) | 2007-04-30 | 2008-11-06 | Immutep | Cytotoxic anti-lag-3 monoclonal antibody and its use in the treatment or prevention of organ transplant rejection and autoimmune disease |
| WO2010019570A2 (en) | 2008-08-11 | 2010-02-18 | Medarex, Inc. | Human antibodies that bind lymphocyte activation gene-3 (lag-3), and uses thereof |
| US8552156B2 (en) | 2010-06-11 | 2013-10-08 | Kyowa Hakko Kirin Co., Ltd | Anti-TIM-3 antibody |
| WO2014140180A1 (en) | 2013-03-15 | 2014-09-18 | Glaxosmithkline Intellectual Property Development Limited | Anti-lag-3 binding proteins |
| US8841418B2 (en) | 2011-07-01 | 2014-09-23 | Cellerant Therapeutics, Inc. | Antibodies that specifically bind to TIM3 |
| WO2014194302A2 (en) | 2013-05-31 | 2014-12-04 | Sorrento Therapeutics, Inc. | Antigen binding proteins that bind pd-1 |
| WO2015116539A1 (en) | 2014-01-28 | 2015-08-06 | Bristol-Myers Squibb Company | Anti-lag-3 antibodies to treat hematological malignancies |
| US9163087B2 (en) | 2010-06-18 | 2015-10-20 | The Brigham And Women's Hospital, Inc. | Bi-specific antibodies against TIM-3 and PD-1 for immunotherapy in chronic immune conditions |
| WO2015200119A1 (en) | 2014-06-26 | 2015-12-30 | Macrogenics, Inc. | Covalently bonded diabodies having immunoreactivity with pd-1 and lag-3, and methods of use thereof |
| US9244059B2 (en) | 2007-04-30 | 2016-01-26 | Immutep Parc Club Orsay | Cytotoxic anti-LAG-3 monoclonal antibody and its use in the treatment or prevention of organ transplant rejection and autoimmune disease |
| WO2016028656A1 (en) | 2014-08-19 | 2016-02-25 | Merck Sharp & Dohme Corp. | Anti-tigit antibodies |
| WO2016028672A1 (en) | 2014-08-19 | 2016-02-25 | Merck Sharp & Dohme Corp. | Anti-lag3 antibodies and antigen-binding fragments |
| WO2016071448A1 (en) | 2014-11-06 | 2016-05-12 | F. Hoffmann-La Roche Ag | Anti-tim3 antibodies and methods of use |
| WO2016106302A1 (en) | 2014-12-23 | 2016-06-30 | Bristol-Myers Squibb Company | Antibodies to tigit |
| WO2016111947A2 (en) | 2015-01-05 | 2016-07-14 | Jounce Therapeutics, Inc. | Antibodies that inhibit tim-3:lilrb2 interactions and uses thereof |
| WO2016144803A2 (en) | 2015-03-06 | 2016-09-15 | Sorrento Therapeutics, Inc. | Antibody therapeutics that bind tim3 |
| US9505839B2 (en) | 2012-07-02 | 2016-11-29 | Bristol-Myers Squibb Company | Optimization of antibodies that bind lymphocyte activation gene-3 (LAG-3), and uses thereof |
| WO2016191643A2 (en) | 2015-05-28 | 2016-12-01 | Oncomed Pharmaceuticals, Inc. | Tigit-binding agents and uses thereof |
| WO2017019846A1 (en) | 2015-07-30 | 2017-02-02 | Macrogenics, Inc. | Pd-1-binding molecules and methods use thereof |
| WO2017024465A1 (en) | 2015-08-10 | 2017-02-16 | Innovent Biologics (Suzhou) Co., Ltd. | Pd-1 antibodies |
| WO2017030823A2 (en) | 2015-08-14 | 2017-02-23 | Merck Sharp & Dohme Corp. | Anti-tigit antibodies |
| WO2017037707A1 (en) | 2015-09-02 | 2017-03-09 | Yissum Research Development Company Of The Hebrew University Of Jerusalem Ltd. | Antibodies specific to human t-cell immunoglobulin and itim domain (tigit) |
| WO2017040790A1 (en) | 2015-09-01 | 2017-03-09 | Agenus Inc. | Anti-pd-1 antibodies and methods of use thereof |
| WO2017053748A2 (en) | 2015-09-25 | 2017-03-30 | Genentech, Inc. | Anti-tigit antibodies and methods of use |
| WO2017132825A1 (en) | 2016-02-02 | 2017-08-10 | 华为技术有限公司 | Emission power verification method, user equipment, and base station |
| WO2017133540A1 (en) | 2016-02-02 | 2017-08-10 | Innovent Biologics (Suzhou) Co., Ltd. | Pd-1 antibodies |
| WO2017152088A1 (en) | 2016-03-04 | 2017-09-08 | JN Biosciences, LLC | Antibodies to tigit |
| WO2018033798A1 (en) | 2016-08-17 | 2018-02-22 | Compugen Ltd. | Anti-tigit antibodies, anti-pvrig antibodies and combinations thereof |
| WO2018102746A1 (en) | 2016-12-02 | 2018-06-07 | Rigel Pharmaceuticals, Inc. | Antigen binding molecules to tigit |
| WO2018102536A1 (en) | 2016-11-30 | 2018-06-07 | Oncomed Pharmaceuticals, Inc. | Methods for treatment of cancer comprising tigit-binding agents |
| WO2018160704A1 (en) | 2017-02-28 | 2018-09-07 | Adimab Llc | Anti-tigit antibodies |
| WO2018200430A1 (en) | 2017-04-26 | 2018-11-01 | Bristol-Myers Squibb Company | Methods of antibody production that minimize disulfide bond reduction |
| WO2018204363A1 (en) | 2017-05-01 | 2018-11-08 | Agenus Inc. | Anti-tigit antibodies and methods of use thereof |
| WO2019023504A1 (en) | 2017-07-27 | 2019-01-31 | Iteos Therapeutics Sa | Anti-tigit antibodies |
| WO2019062832A1 (en) | 2017-09-29 | 2019-04-04 | 江苏恒瑞医药股份有限公司 | Tigit antibody, antigen-binding fragment thereof, and medical use thereof |
| WO2019129261A1 (en) | 2017-12-30 | 2019-07-04 | Beigene, Ltd. | Anti-tigit antibodies and their use as therapeutics and diagnostics |
| WO2019129221A1 (en) | 2017-12-28 | 2019-07-04 | Nanjing Legend Biotech Co., Ltd. | Single-domain antibodies and variants thereof against tigit |
| WO2019137548A1 (en) | 2018-01-15 | 2019-07-18 | Nanjing Legend Biotech Co., Ltd. | Antibodies and variants thereof against tigit |
| WO2019152574A1 (en) | 2018-02-01 | 2019-08-08 | Merck Sharp & Dohme Corp. | Methods for treating cancer or infection using a combination of an anti-pd-1 antibody, an anti-lag3 antibody, and an anti-tigit antibody |
| WO2019154415A1 (en) | 2018-02-06 | 2019-08-15 | I-Mab | Antibodies to t cell immunoreceptor with ig and itim domains (tigit) and uses thereof |
| WO2019168382A1 (en) | 2018-02-28 | 2019-09-06 | Yuhan Corporation | Anti-tigit antibodies and uses thereof |
| WO2019215728A1 (en) | 2018-05-09 | 2019-11-14 | Yissum Research Development Company Of The Hebrew University Of Jerusalem Ltd. | Antibodies specific to human nectin4 |
| WO2019232484A1 (en) | 2018-06-01 | 2019-12-05 | Compugen Ltd | Anti-pvrig/anti-tigit bispecific antibodies and methods of use |
| WO2021214129A1 (en) | 2020-04-21 | 2021-10-28 | Université Catholique de Louvain | Alpha-2 adrenergic receptor agonists for the treatment of cancer |
-
2024
- 2024-09-27 WO PCT/EP2024/077199 patent/WO2025068452A1/en active Pending
Patent Citations (47)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4602041A (en) * | 1982-02-10 | 1986-07-22 | Beecham Group P.L.C. | Benzylideneamino- and phenylacetylguanidines and their pharmaceutical uses |
| WO2006121168A1 (en) | 2005-05-09 | 2006-11-16 | Ono Pharmaceutical Co., Ltd. | Human monoclonal antibodies to programmed death 1(pd-1) and methods for treating cancer using anti-pd-1 antibodies alone or in combination with other immunotherapeutics |
| WO2008132601A1 (en) | 2007-04-30 | 2008-11-06 | Immutep | Cytotoxic anti-lag-3 monoclonal antibody and its use in the treatment or prevention of organ transplant rejection and autoimmune disease |
| US9244059B2 (en) | 2007-04-30 | 2016-01-26 | Immutep Parc Club Orsay | Cytotoxic anti-LAG-3 monoclonal antibody and its use in the treatment or prevention of organ transplant rejection and autoimmune disease |
| WO2010019570A2 (en) | 2008-08-11 | 2010-02-18 | Medarex, Inc. | Human antibodies that bind lymphocyte activation gene-3 (lag-3), and uses thereof |
| US8552156B2 (en) | 2010-06-11 | 2013-10-08 | Kyowa Hakko Kirin Co., Ltd | Anti-TIM-3 antibody |
| US9163087B2 (en) | 2010-06-18 | 2015-10-20 | The Brigham And Women's Hospital, Inc. | Bi-specific antibodies against TIM-3 and PD-1 for immunotherapy in chronic immune conditions |
| US8841418B2 (en) | 2011-07-01 | 2014-09-23 | Cellerant Therapeutics, Inc. | Antibodies that specifically bind to TIM3 |
| US9505839B2 (en) | 2012-07-02 | 2016-11-29 | Bristol-Myers Squibb Company | Optimization of antibodies that bind lymphocyte activation gene-3 (LAG-3), and uses thereof |
| WO2014140180A1 (en) | 2013-03-15 | 2014-09-18 | Glaxosmithkline Intellectual Property Development Limited | Anti-lag-3 binding proteins |
| WO2014194302A2 (en) | 2013-05-31 | 2014-12-04 | Sorrento Therapeutics, Inc. | Antigen binding proteins that bind pd-1 |
| WO2015116539A1 (en) | 2014-01-28 | 2015-08-06 | Bristol-Myers Squibb Company | Anti-lag-3 antibodies to treat hematological malignancies |
| WO2015200119A1 (en) | 2014-06-26 | 2015-12-30 | Macrogenics, Inc. | Covalently bonded diabodies having immunoreactivity with pd-1 and lag-3, and methods of use thereof |
| WO2016028672A1 (en) | 2014-08-19 | 2016-02-25 | Merck Sharp & Dohme Corp. | Anti-lag3 antibodies and antigen-binding fragments |
| WO2016028656A1 (en) | 2014-08-19 | 2016-02-25 | Merck Sharp & Dohme Corp. | Anti-tigit antibodies |
| WO2016071448A1 (en) | 2014-11-06 | 2016-05-12 | F. Hoffmann-La Roche Ag | Anti-tim3 antibodies and methods of use |
| WO2016106302A1 (en) | 2014-12-23 | 2016-06-30 | Bristol-Myers Squibb Company | Antibodies to tigit |
| WO2016111947A2 (en) | 2015-01-05 | 2016-07-14 | Jounce Therapeutics, Inc. | Antibodies that inhibit tim-3:lilrb2 interactions and uses thereof |
| WO2016144803A2 (en) | 2015-03-06 | 2016-09-15 | Sorrento Therapeutics, Inc. | Antibody therapeutics that bind tim3 |
| WO2016191643A2 (en) | 2015-05-28 | 2016-12-01 | Oncomed Pharmaceuticals, Inc. | Tigit-binding agents and uses thereof |
| WO2017019846A1 (en) | 2015-07-30 | 2017-02-02 | Macrogenics, Inc. | Pd-1-binding molecules and methods use thereof |
| WO2017025016A1 (en) | 2015-08-10 | 2017-02-16 | Innovent Biologics (Suzhou) Co., Ltd. | Pd-1 antibodies |
| WO2017024465A1 (en) | 2015-08-10 | 2017-02-16 | Innovent Biologics (Suzhou) Co., Ltd. | Pd-1 antibodies |
| WO2017030823A2 (en) | 2015-08-14 | 2017-02-23 | Merck Sharp & Dohme Corp. | Anti-tigit antibodies |
| WO2017040790A1 (en) | 2015-09-01 | 2017-03-09 | Agenus Inc. | Anti-pd-1 antibodies and methods of use thereof |
| WO2017037707A1 (en) | 2015-09-02 | 2017-03-09 | Yissum Research Development Company Of The Hebrew University Of Jerusalem Ltd. | Antibodies specific to human t-cell immunoglobulin and itim domain (tigit) |
| WO2017053748A2 (en) | 2015-09-25 | 2017-03-30 | Genentech, Inc. | Anti-tigit antibodies and methods of use |
| WO2017132825A1 (en) | 2016-02-02 | 2017-08-10 | 华为技术有限公司 | Emission power verification method, user equipment, and base station |
| WO2017133540A1 (en) | 2016-02-02 | 2017-08-10 | Innovent Biologics (Suzhou) Co., Ltd. | Pd-1 antibodies |
| WO2017152088A1 (en) | 2016-03-04 | 2017-09-08 | JN Biosciences, LLC | Antibodies to tigit |
| WO2018033798A1 (en) | 2016-08-17 | 2018-02-22 | Compugen Ltd. | Anti-tigit antibodies, anti-pvrig antibodies and combinations thereof |
| WO2018102536A1 (en) | 2016-11-30 | 2018-06-07 | Oncomed Pharmaceuticals, Inc. | Methods for treatment of cancer comprising tigit-binding agents |
| WO2018102746A1 (en) | 2016-12-02 | 2018-06-07 | Rigel Pharmaceuticals, Inc. | Antigen binding molecules to tigit |
| WO2018160704A1 (en) | 2017-02-28 | 2018-09-07 | Adimab Llc | Anti-tigit antibodies |
| WO2018200430A1 (en) | 2017-04-26 | 2018-11-01 | Bristol-Myers Squibb Company | Methods of antibody production that minimize disulfide bond reduction |
| WO2018204363A1 (en) | 2017-05-01 | 2018-11-08 | Agenus Inc. | Anti-tigit antibodies and methods of use thereof |
| WO2019023504A1 (en) | 2017-07-27 | 2019-01-31 | Iteos Therapeutics Sa | Anti-tigit antibodies |
| WO2019062832A1 (en) | 2017-09-29 | 2019-04-04 | 江苏恒瑞医药股份有限公司 | Tigit antibody, antigen-binding fragment thereof, and medical use thereof |
| WO2019129221A1 (en) | 2017-12-28 | 2019-07-04 | Nanjing Legend Biotech Co., Ltd. | Single-domain antibodies and variants thereof against tigit |
| WO2019129261A1 (en) | 2017-12-30 | 2019-07-04 | Beigene, Ltd. | Anti-tigit antibodies and their use as therapeutics and diagnostics |
| WO2019137548A1 (en) | 2018-01-15 | 2019-07-18 | Nanjing Legend Biotech Co., Ltd. | Antibodies and variants thereof against tigit |
| WO2019152574A1 (en) | 2018-02-01 | 2019-08-08 | Merck Sharp & Dohme Corp. | Methods for treating cancer or infection using a combination of an anti-pd-1 antibody, an anti-lag3 antibody, and an anti-tigit antibody |
| WO2019154415A1 (en) | 2018-02-06 | 2019-08-15 | I-Mab | Antibodies to t cell immunoreceptor with ig and itim domains (tigit) and uses thereof |
| WO2019168382A1 (en) | 2018-02-28 | 2019-09-06 | Yuhan Corporation | Anti-tigit antibodies and uses thereof |
| WO2019215728A1 (en) | 2018-05-09 | 2019-11-14 | Yissum Research Development Company Of The Hebrew University Of Jerusalem Ltd. | Antibodies specific to human nectin4 |
| WO2019232484A1 (en) | 2018-06-01 | 2019-12-05 | Compugen Ltd | Anti-pvrig/anti-tigit bispecific antibodies and methods of use |
| WO2021214129A1 (en) | 2020-04-21 | 2021-10-28 | Université Catholique de Louvain | Alpha-2 adrenergic receptor agonists for the treatment of cancer |
Non-Patent Citations (8)
| Title |
|---|
| "Handbook of Pharmaceutical Salts: Properties, Selection, and Use", 2002 |
| ALFRED H. SCHINKEL: "P-Glycoprotein, a gatekeeper in the blood-brain barrier", ADVANCED DRUG DELIVERY REVIEWS, vol. 36, 1999, pages 179 - 194, XP003004977, DOI: 10.1016/S0169-409X(98)00085-4 |
| DATABASE CAS STNext [online] American Chemical Society; 1 January 1980 (1980-01-01), SHOJI KAMIMURA ET AL: "Metabolic studies on N-amidino-2-(2,6-dichlorophenyl) acetamidehydrochloride (guanfacine), a new antihypertensive agent. 2. Metabolites in blood, liver and excreta of rats after single and repeated oral administration", XP093133538, retrieved from CAPLUS accession no. 78433-86-0 Database accession no. 1981:508334 * |
| J. PHARM. SCI., vol. 66, 1977, pages 2 |
| J. ZHU ET AL., NATURE, vol. 618, 2023, pages 607 - 615 |
| SHOJI KAMIMURA: "Metabolic studies on N-amidino-2-(2,6-dichlorophenyl) acetamidehydrochloride (guanfacine), a new antihypertensive agent. 2. Metabolites in blood, liver and excreta of rats after single and repeated oral administration", OYO YAKURI, vol. 20, no. 5, 1980, pages 745 - 751, ISSN: 0369-8033 * |
| SI-YANG LIU ET AL., J. HEMATOL. ONCOL., vol. 10, 2017, pages 136 |
| XING GANG ET AL: "Recent progress in the development of [beta]2 adrenergic receptor agonists: a patent review (2015-2020)", EXPERT OPINION ON THERAPEUTIC PATENTS, vol. 31, no. 3, 23 December 2020 (2020-12-23), GB, pages 239 - 246, XP093132564, ISSN: 1354-3776, DOI: 10.1080/13543776.2021.1865312 * |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN107922431B (en) | HPK1 inhibitors and methods of use | |
| CN115279771B (en) | MAP4K1 inhibitors | |
| BR112021000049A2 (en) | DERIVATIVES OF 3- (5-AMINO-1-OXOISOINDOLIN-2-IL) PIPERIDINE-2,6-DIONA AND ITS USE IN THE TREATMENT OF ZINC FINGER-RELATED DISEASES OF THE IKAROS 2 FAMILY (IKZF2) | |
| BR102018007822A2 (en) | compound, methods for inhibiting pd-1, pd-11 and / or interaction of pd-1 / pd-11 and for cancer treatment, pharmaceutical composition, and kit for treating or preventing cancer or a disease or condition | |
| JP2020536971A (en) | Cyclic dinucleotide as an anticancer drug | |
| AU2021383928A1 (en) | Fgfr inhibitors and methods of making and using the same | |
| KR20220004066A (en) | Ionic liquids for drug delivery | |
| EP3836916A1 (en) | Benzothiophene estrogen receptor modulators to treat medical disorders | |
| JP2020526520A (en) | Amorphous and crystalline forms of IDO inhibitors | |
| CA3220155A1 (en) | Tricyclic heterocycles as fgfr inhibitors | |
| JP2024521791A (en) | EGFR degraders for treating cancer metastasis to the brain or CNS | |
| EP3719021B1 (en) | Inhibitors of adapter associated kinase 1, compositions comprising them, and methods of their use | |
| US12173008B2 (en) | Substituted piperazines as BTK inhibitors | |
| WO2025068452A1 (en) | Guanfacine derivatives and their use in treating cancer | |
| EP4587429A1 (en) | Substituted oxoisoindolinyl piperidine-2,6-dione compounds | |
| WO2025064197A1 (en) | Substituted azetidinyl oxoisoindolinyl piperidine-2,6-dione compounds | |
| CN112608330A (en) | A2A and/or A2B receptor inhibitors | |
| WO2025068461A1 (en) | Guanfacine derivatives and their use in treating cancer | |
| JP2021515016A (en) | Cyclic dinucleotide as an anticancer drug | |
| WO2025262250A1 (en) | Guanfacine derivatives and their uses | |
| TWI768781B (en) | TRANSFORMING GROWTH FACTOR-β RECEPTOR INHIBITOR | |
| CN116507331A (en) | TLR7/8 antagonists and uses thereof | |
| RU2828748C2 (en) | Heterocyclic compounds, methods for production and use thereof | |
| WO2025229177A1 (en) | Crystalline forms of an n-substituted indole derivative | |
| WO2025132831A1 (en) | N-heteroaryl derivatives and uses thereof for treating cancer |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 24776978 Country of ref document: EP Kind code of ref document: A1 |