WO2025067703A1 - Polyolefin composition for cable insulation - Google Patents
Polyolefin composition for cable insulation Download PDFInfo
- Publication number
- WO2025067703A1 WO2025067703A1 PCT/EP2024/058705 EP2024058705W WO2025067703A1 WO 2025067703 A1 WO2025067703 A1 WO 2025067703A1 EP 2024058705 W EP2024058705 W EP 2024058705W WO 2025067703 A1 WO2025067703 A1 WO 2025067703A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- polyolefin composition
- determined
- iso
- propylene
- range
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L23/00—Compositions of homopolymers or copolymers of unsaturated aliphatic hydrocarbons having only one carbon-to-carbon double bond; Compositions of derivatives of such polymers
- C08L23/02—Compositions of homopolymers or copolymers of unsaturated aliphatic hydrocarbons having only one carbon-to-carbon double bond; Compositions of derivatives of such polymers not modified by chemical after-treatment
- C08L23/10—Homopolymers or copolymers of propene
- C08L23/14—Copolymers of propene
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F210/00—Copolymers of unsaturated aliphatic hydrocarbons having only one carbon-to-carbon double bond
- C08F210/04—Monomers containing three or four carbon atoms
- C08F210/06—Propene
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01B—CABLES; CONDUCTORS; INSULATORS; SELECTION OF MATERIALS FOR THEIR CONDUCTIVE, INSULATING OR DIELECTRIC PROPERTIES
- H01B3/00—Insulators or insulating bodies characterised by the insulating materials; Selection of materials for their insulating or dielectric properties
- H01B3/18—Insulators or insulating bodies characterised by the insulating materials; Selection of materials for their insulating or dielectric properties mainly consisting of organic substances
- H01B3/30—Insulators or insulating bodies characterised by the insulating materials; Selection of materials for their insulating or dielectric properties mainly consisting of organic substances plastics; resins; waxes
- H01B3/44—Insulators or insulating bodies characterised by the insulating materials; Selection of materials for their insulating or dielectric properties mainly consisting of organic substances plastics; resins; waxes vinyl resins; acrylic resins
- H01B3/441—Insulators or insulating bodies characterised by the insulating materials; Selection of materials for their insulating or dielectric properties mainly consisting of organic substances plastics; resins; waxes vinyl resins; acrylic resins from alkenes
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L2203/00—Applications
- C08L2203/20—Applications use in electrical or conductive gadgets
- C08L2203/202—Applications use in electrical or conductive gadgets use in electrical wires or wirecoating
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L2207/00—Properties characterising the ingredient of the composition
- C08L2207/02—Heterophasic composition
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L2207/00—Properties characterising the ingredient of the composition
- C08L2207/06—Properties of polyethylene
- C08L2207/066—LDPE (radical process)
Definitions
- the present invention relates to a polyolefin composition, an article comprising said polyolefin composition, preferably a cable comprising an insulation layer comprising said polyolefin composition and the use of said polypropylene composition as cable insulation for medium and high voltage cables, especially for high voltage (HV) cable, extra high voltage (EHV) cables and ultra high voltage (UHV) cables.
- HV high voltage
- EHV extra high voltage
- UHV ultra high voltage
- thermoplastic ethylene polymer products PE and HDPE
- the maximum operation temperature being the maximum conductor operation temperature in normal operation for high voltage DC cables up to 320 kV, are limited to 80°C.
- thermoplastic propylene polymers as insulation material for medium, high and extra high voltage (MV, HV and EHV) cables as well as high-voltage direct current (HVDC) cables.
- MV, HV and EHV medium, high and extra high voltage cables
- HVDC high-voltage direct current cables
- WO 2022/200395 A1 and WO 2022/200396 A1 disclose flexible polypropylene compositions suitable for cable insulation, which both comprise a flexible heterophasic copolymer of propylene and ethylene. Said compositions show a superior balance of properties in regard of flexibility, impact strength and electric properties, which makes these compositions especially suitable for medium voltage cables.
- a polyolefin composition with a rather low amount of soluble fraction in crystallization extraction (CRYSTEX), which is a measure for the amorphous phase of the polyolefin composition, is proposed, which shows an improved balance of tensile and indentation properties and thermal conductivity and additionally shows a good performance during cable extrusion.
- the polyolefin composition thus is suitable for cable insulation, especially for meeting all CIGRE TB852 recommendations for HVDC cable systems.
- the present invention relates to a polyolefin composition having a total ethylene content of from more than 32.5 to 60.0 wt.-%, preferably from 33.0 to 55.0 wt.-%, more preferably from 33.5 to 52.5 wt.-%, still more preferably from 34.0 to 50.0 wt.-%, based on the total weight of the polyolefin composition and determined by FT-IR spectroscopy calibrated by quantitative 13 C-NMR spectroscopy from crystallization extraction (CRYSTEX); a melt flow rate MFR2 of from more than 2.5 to 6.0 g/10 min, preferably from 2.6 to 5.5 g/10 min, still more preferably from 2.7 to 5.0 g/10 min and most preferably from 2.8 to 4.5 g/10 min, determined according to ISO 1133 at 230°C and 2.16 kg; a crystalline fraction (CF) content, determined by crystallization extraction (CRYSTEX), in the range from 75.0 to 95.0 wt.-%,
- the present invention relates to an article comprising the polyolefin composition as described above or below.
- said article is a cable comprising an insulation layer comprising the polyolefin composition as described above or below.
- the present invention relates to the use of the polyolefin composition as described above or below as cable insulation for medium voltage cables, high voltage cables, extra high voltage cable and ultra high voltage cables, preferably for high voltage cables and extra high voltage cables, more preferably high voltage DC (HVDC) cables and extra high voltage DC (EHVDC) cables.
- HVDC high voltage DC
- EHVDC extra high voltage DC
- a heterophasic polypropylene is a propylene-based copolymer with a semi-crystalline matrix phase, which can be a propylene homopolymer or a random copolymer of propylene and at least one alpha-olefin comonomer, and an elastomeric phase dispersed therein.
- the elastomeric phase can be a propylene copolymer with a high amount of comonomer, which is not randomly distributed in the polymer chain but are distributed in a comonomer-rich block structure and a propylene-rich block structure.
- a heterophasic polypropylene usually differentiates from a one-phasic propylene copolymer in that it shows two distinct glass transition temperatures T g which are attributed to the matrix phase and the elastomeric phase.
- a propylene homopolymer is a polymer, which essentially consists of propylene monomer units. Due to impurities especially during commercial polymerization processes a propylene homopolymer can comprise up to 0.1 mol% comonomer units, preferably up to 0.05 mol% comonomer units and most preferably up to 0.01 mol% comonomer units.
- a propylene random copolymer is a copolymer of propylene monomer units and comonomer units in which the comonomer units are distributed randomly over the polypropylene chain.
- a propylene random copolymer includes a fraction, which is insoluble in xylene - xylene cold insoluble (XCI) fraction - in an amount of at least 85 wt.-%, most preferably of at least 88 wt.-%, based on the total amount of propylene random copolymer. Accordingly, the propylene random copolymer does not contain an elastomeric polymer phase dispersed therein.
- a propylene polymer comprising at least two propylene polymer fractions (components), which have been produced under different polymerization conditions resulting in different (weight average) molecular weights and/or different comonomer contents for the fractions, preferably produced by polymerizing in multiple polymerization stages with different polymerization conditions, is referred to as “multimodal”.
- multi relates to the number of different polymer fractions the propylene polymer is consisting of.
- a propylene polymer consisting of two fractions only is called “bimodal”
- a propylene polymer consisting of three fractions only is called “trimodal”.
- a unimodal propylene polymer only consists of one fraction.
- the term “different” means that the propylene polymer fractions differ from each other in at least one property, preferably in the weight average molecular weight - which can also be measured in different melt flow rates of the fractions - or comonomer content or both.
- An elastomer is a polymer with viscoelasticity and weak intermolecular forces.
- the term “elastomer” can be used interchangeably with “rubber”.
- Polyolefin based elastomers such as polypropylene based elastomers, i.e. an elastomer with a molar majority of olefin monomer units, such as propylene monomer units, are usually thermoplastic elastomers.
- Thermoplastic elastomers have both thermoplastic and elastomeric properties.
- Polyolefin based elastomers such as polypropylene based elastomers, usually show a low density and low viscosity. They can be propylene homopolymers or propylene-alpha olefin copolymers, such as propylene-ethylene copolymers.
- a specific class of polypropylene based elastomers are propylene homopolymers or propylene-alpha olefin copolymers, such as propylene-ethylene copolymers, which have been polymerized in the presence of a single site catalyst, usually in a solution polymerization process.
- Vis-breaking is a post reactor chemical process for modifying semi-crystalline polymers such as propylene polymers.
- the propylene polymer backbone is degraded, for example by means of peroxides, such as organic peroxides, via beta scission.
- the degradation is generally used for increasing the melt flow rate and narrowing the molecular weight distribution.
- a low density polyethylene is a polymer with predominately ethylene monomers, which is polymerized in a high pressure process by free radical polymerization.
- the low density polyethylene is characterized by long polymer side chains, which decrease the density of the polymer irrespective of the amount of comonomer.
- HV high voltage
- EHV Extra-high voltage
- ultra-high voltage consequently refers to voltages of more than 800 kV for DC systems.
- the polyolefin composition as described above or below has a total ethylene content of from more than 32.5 to 60.0 wt.-%, preferably from 33.0 to 55.0 wt.-%, more preferably from 33.5 to 52.5 wt.-%, still more preferably from 34.0 to 50.0 wt.-%, based on the total weight of the polyolefin composition and determined by FT-IR spectroscopy calibrated by quantitative 13 C-NMR spectroscopy from crystallization extraction (CRYSTEX); and a melt flow rate MFR2 of from more than 2.5 to 6.0 g/10 min, preferably from 2.6 to 5.5 g/10 min, still more preferably from 2.7 to 5.0 g/10 min and most preferably from 2.8 to 4.5 g/10 min, determined according to ISO 1133 at 230°C and 2.16 kg.
- the polymeric part of the polyolefin composition is characterized by crystallization extraction (CRYSTEX) using trichlorobenzene (TCB) as a solvent. This method is described below in the determination methods section.
- the crystalline fraction (CF) contains for the most part the matrix phase and only a small part of the elastomeric phase and the soluble fraction (SF) contains for the most part the elastomeric phase and only a small part of the matrix phase. In some cases, this method results in more useful data compared to xylene extraction, since the crystalline fraction (CF) and the soluble fraction (SF) more accurately correspond to the matrix and elastomeric phases, respectively.
- the crystalline fraction (CF) content and the soluble (SF) content of a composition only relate to its polymeric components, i.e. without other components, which are insoluble and therefore not part of the dissolution and crystallization cycles as described below in the determination method.
- the polyolefin composition is usually fully soluble in trichlorobenzene (TCB) so that the crystalline fraction (CF) content and the soluble (SF) content relate to the total content of the polyolefin composition.
- TBC trichlorobenzene
- the crystalline fraction (CF) content and the soluble fraction (SF) content preferably make up 100 wt.-% of the polyolefin composition.
- the polyolefin composition has a crystalline fraction (CF) content, determined by crystallization extraction (CRYSTEX), in the range from 75.0 to 95.0 wt.-%, preferably from 77.5 to 93.5 wt.-%, more preferably from 80.0 to 92.5 wt.-% and most preferably from 82.5 to 91 .0 wt.-%, based on the total weight amount of the polyolefin composition.
- CF crystalline fraction
- Said crystalline fraction (CF) has an ethylene content (C2(CF)), determined by FT-IR spectroscopy calibrated by quantitative 13 C-NMR spectroscopy, in the range from more than 32.5 to 62.5 wt.-%, preferably from 35.0 to 60.0 wt.-% and most preferably from 36.0 to 55.0 wt.-%, based on the total amount of monomer units in the crystalline fraction (CF).
- C2(CF) ethylene content
- said crystalline fraction (CF) has a propylene content (C3(CF)), determined by FT-IR spectroscopy calibrated by quantitative 13 C-NMR spectroscopy, in the range from 37.5 to less than 67.5 wt.-%, preferably from 40.0 to 65.0 wt.-% and most preferably from 45.0 to 64.0 wt.-%, based on the total amount of monomer units in the crystalline fraction (CF).
- C3(CF) propylene content
- said crystalline fraction (CF) preferably has an intrinsic viscosity (iV(CF)), determined according to ISO 1628-3, in the range from 100 to 250 cm 3 /g, more preferably from 125 to 225 cm 3 /g and most preferably from 140 to 210 cm 3 /g.
- iV(CF) intrinsic viscosity
- the polyolefin composition has a soluble fraction (SF) content, determined by crystallization extraction (CRYSTEX), in the range from 5.0 to 25.0 wt.-%, preferably from 6.5 to 22.5 wt.-%, more preferably from 7.5 to 20.0 wt.-% and most preferably from 9.0 to 17.5 wt.-%, based on the total weight amount of the polyolefin composition.
- SF soluble fraction
- Said soluble fraction (SF) has an intrinsic viscosity (iV(SF)), determined according to ISO 1628-3, in the range from 85 to 200 cm 3 /g, more preferably from 95 to 175 cm 3 /g and most preferably from 100 to 150 cm 3 /g.
- iV(SF) intrinsic viscosity
- said soluble fraction (SF) preferably has an ethylene content (C2(SF)), determined by FT-IR spectroscopy calibrated by quantitative 13 C-NMR spectroscopy, in the range from 22.5 to 42.5 wt.-%, more preferably from 25.0 to 40.0 wt.-% and most preferably from 30.0 to 35.0 wt.-%, based on the total amount of monomer units in the soluble fraction (SF).
- C2(SF) ethylene content
- said soluble fraction (SF) has a propylene content (C3(SF)), determined by FT-IR spectroscopy calibrated by quantitative 13 C-NMR spectroscopy, in the range from 57.5 to 77.5 wt.-%, preferably from 60.0 to 75.0 wt.-% and most preferably from 62.5 to 70.0 wt.-%, based on the total amount of monomer units in the soluble fraction (SF).
- C3(SF) propylene content
- the polyolefin composition preferably has a ratio of the ethylene content of the soluble fraction to the ethylene content of the crystalline fraction C2(SF)/C2(CF) of from 0.50 : 1.00 to 1 .00 : 1 .00, more preferably from 0.55 : 1 .00 to 0.95 : 1 .00, most preferably from 0.60 : 1.00 to 0.90 : 1.00.
- the polyolefin composition preferably has a ratio of the intrinsic viscosity of the soluble fraction to the intrinsic viscosity of the crystalline fraction iV(SF)/iV(CF) of from 0.40 : 1 .00 to 0.95 : 1 .00, more preferably from 0.50 : 1 .00 to 0.85 : 1 .00, most preferably from 0.55 : 1 .00 to 0.75 : 1 .00.
- the polyolefin composition preferably has an intrinsic viscosity, determined according to ISO 1628-3, of from 100 to 210 cm 3 /g, more preferably from 120 to 200 cm 3 /g, most preferably from 130 to 190 cm 3 /g.
- the polyolefin composition preferably comprises a total propylene content of from 40.0 to less than 67.5 wt.-%, preferably from 45.0 to 67.0 wt.-%, more preferably from 47.5 to 66.5 wt.-%, still more preferably from 49.0 to 66.0 wt.-%, based on the total weight of the polyolefin composition and determined by FT-IR spectroscopy calibrated by quantitative 13 C-NMR spectroscopy from crystallization extraction (CRYSTEX).
- the polymeric part of the polyolefin composition preferably consists of propylene monomer units and ethylene monomer units.
- the polyolefin composition preferably has a good balance of properties in regard of tensile strength, strain at break and indentation as illustrated by the following properties:
- the polypropylene composition preferably has a tensile strength of from 8.5 to 50 MPa, preferably from 15 to 45 MPa, more preferably from 20 to 40 MPa, determined according to ISO 527-2 on 5A injection moulded test specimens at 23°C and a speed of 25 mm/min before ageing.
- the polypropylene composition preferably has a tensile strength of from 8.5 to 50 MPa, preferably from 15 to 45 MPa, more preferably from 20 to 40 MPa, determined according to ISO 527-2 on 5A injection moulded test specimens at 23°C and a speed of 25 mm/min after ageing for 10 days at 135°C.
- the polyolefin composition preferably has a strain at break of from 350 to 650 %, preferably from 360 to 600 %, more preferably from 375 to 575 %, determined according to ISO 527-2 on 5A injection moulded test specimens at 23°C and a speed of 25 mm/min before ageing.
- the polyolefin composition preferably has a strain at break of from 350 to 650 %, preferably from 360 to 600 %, more preferably from 375 to 575 %, determined according to ISO 527-2 on 5A injection moulded test specimens at 23°C and a speed of 25 mm/min after ageing for 10 days at 135°C.
- the polyolefin composition preferably has an indentation in the pressure test of from 0 to 50 %, preferably of from 0 to 45 %, determined on compression moulded plaques with a cylindric indentor according to method 1 pressure test in the CIGRE TB852 recommendations at 130°C for 1 h.
- the polyolefin composition preferably has a thermal conductivity of at least 0.245 W/Km, such as from 0.245 to 0.400 W/Km, preferably from 0.250 to 0.375 W/Km, more preferably from 0.260 to 0.350 W/Km, determined according to ISO 22007-2 at 22°C.
- the polyolefin composition preferably has a thermal conductivity of at least 0.240 W/Km, such as from 0.240 to 0.375 W/Km, preferably from 0.245 to 0.350 W/Km, more preferably from 0.248 to 0.325 W/Km, determined according to ISO 22007-2 at 22°C.
- the polyolefin composition thus preferably meets all all CIGRE TB852 recommendations for HVDC cable systems, also after ageing.
- polyolefin composition shows good thermal conductivity over a broad temperature range of 22 to 100°C.
- the polyolefin composition comprises from 40.0 to less than 70.0 wt.-%, preferably from 45.0 to 69.0 wt.-%, more preferably from 47.5 to 67.5 wt.-% of a copolymer of propylene and ethylene and from more than 30.0 to 60.0 wt.-%, preferably from 31 .0 to 55.0 wt.-%, more preferably from 32.5 to 52.5 wt.-% of a low density polyethylene, wherein all weight amounts are based on the total weight of the polyolefin composition.
- the polyolefin composition can further comprise polymeric components, which are different from the copolymer of propylene and ethylene and the low density polyethylene, in an amount of preferably 0.0 to 10.0 wt.-% based on the total weight of the polyolefin composition.
- the polymeric components of the polyolefin composition consist of copolymer of propylene and ethylene and the low density polyethylene.
- the polyolefin composition can comprise one or more additives in an amount of from 0.0 up to 5.0 wt.-%, based on the total weight of the polyolefin composition.
- the one or more additives are preferably selected from acid scavengers, antioxidants, alpha nucleating agents, beta nucleating agents, etc.
- Such additives are commercially available and for example described in “Plastic Additives Handbook”, 6 th edition 2009 of Hans Zweifel (pages 1141 to 1190).
- additives are added in quantities of 1 to 50000 ppm for each single component.
- the one or more additives can be added to the polymeric components in a blending step. Thereby, the one or more additives can be added to the polymeric components in form of master batches in which one or more additives are blended with a carrier polymer in concentrated amounts. Any optional carrier polymer is calculated to the amount of additives, based on the total weight of the polyolefin composition.
- the polyolefin composition contains from 0 to 5.00 wt.-%, more preferably from 0 to 2.50 wt.-% of the alpha-nucleating agent, based on the total amount of the polyolefin composition.
- the amount of pure alpha-nucleating agent in the polyolefin composition is preferably in the range of from 0 to 5000 ppm, more preferably from 0 to 4000 ppm, based on the total amount of the polyolefin composition.
- the alpha-nucleating agent is generally not restricted.
- the alpha-nucleating agent is selected from soluble alpha-nucleating agents and polymeric alpha-nucleating agents.
- the alpha-nucleating agent is preferably selected from the group consisting of
- dibenzylidenesorbitol e.g. 1 ,3 : 2,4 dibenzylidenesorbitol
- Ci-Cs-alkyl- substituted dibenzylidenesorbitol derivatives such as methyldibenzylidenesorbitol, ethyldibenzylidenesorbitol or dimethyldibenzylidenesorbitol (e.g. 1 ,3 : 2,4 di(methylbenzylidene) sorbitol), or substituted nonitol-derivatives, such as 1 ,2,3- trideoxy-4,6:5,7-bis-0-[(4-propylphenyl)methylene]-nonitol, and
- the alpha-nucleating agent is preferably selected from the group consisting of dibenzylidenesorbitol (e.g. 1 ,3 : 2,4 dibenzylidene sorbitol), dibenzylidenesorbitol derivative, preferably dimethyldibenzylidenesorbitol (e.g. 1 ,3 : 2,4 di(methylbenzylidene) sorbitol), or substituted nonitol-derivatives, such as 1 ,2,3-trideoxy-4,6:5,7-bis-0-[(4- propylphenyl)methylene]-nonitol, vinylcycloalkane polymer, vinylalkane polymer, and mixtures thereof.
- vinylcycloalkane polymers such as e.g. vinylcyclohexane (VCH) polymers.
- VCH vinylcyclohexane
- BNT Borealis Nucleation Technology
- the alpha-nucleating agent is a soluble alpha-nucleating agent, more preferably a soluble alpha-nucleating agent selected from dibenzylidenesorbitol (e.g. 1 ,3 : 2,4 dibenzylidenesorbitol) and Ci-Cs-alkyl-substituted dibenzylidenesorbitol derivatives, such as methyldibenzylidenesorbitol, ethyldibenzylidenesorbitol or dimethyldibenzylidenesorbitol (e.g.
- dibenzylidenesorbitol e.g. 1 ,3 : 2,4 dibenzylidenesorbitol
- Ci-Cs-alkyl-substituted dibenzylidenesorbitol derivatives such as methyldibenzylidenesorbitol, ethyldibenzylidenesorbitol or dimethyldibenz
- the alpha-nucleating agent can be added to the polyolefin composition as an isolated raw material or in a mixture with a carrier polymer, i.e. in a so-called master batch.
- the amount of the carrier polymer of the master batch thereby is calculated to the amount of the alpha-nucleating agent.
- the polyolefin composition comprises an alpha-nucleating agent.
- the pure amount of alpha-nucleating agent is preferably in the range of from 0.1 to 5000 ppm, more preferably from 1 to 5000 ppm.
- the polyolefin composition does not comprise an alphanucleating agent.
- the pure amount of alpha-nucleating agent is 0 ppm.
- the polyolefin composition is prepared by melt blending the copolymer of propylene and ethylene and the low density polyethylene, the optional additional polymeric components and the optional further additives, all as described above or below.
- the polyolefin composition is preferably not subjected to vis-breaking.
- the polyolefin composition does not comprise, i.e. is free of a dielectric fluid, such as e.g. described in EP 2 739 679.
- the polyolefin composition as described above or below preferably comprises a copolymer of propylene and ethylene.
- the copolymer of propylene and ethylene is preferably present in the polyolefin composition in an amount of from 40.0 to less than 70.0 wt.-%, preferably from 45.0 to 69.0 wt.-%, more preferably from 47.5 to 67.5 wt.-%, based on the total weight of the polyolefin composition.
- the copolymer of propylene and ethylene preferably has a total ethylene content of from 4.0 to 11 .0 wt.-%, more preferably from 5.0 to 10.0 wt.-%, still more preferably from 5.5 to 9.5 wt.-%, most preferably from 6.0 to 9.0 wt.-%, based on the total weight of the copolymer of propylene and ethylene and determined by FT-IR spectroscopy calibrated by quantitative 13 C-NMR spectroscopy from crystallization extraction (CRYSTEX).
- the copolymer of propylene and ethylene preferably has a total propylene content of from 89.0 to 96.0 wt.-%, more preferably from 90.0 to 95.0 wt.-%, still more preferably from 90.5 to 95.5 wt.-%, most preferably from 91 .0 to 94.0 wt.-%, based on the total weight of the copolymer of propylene and ethylene and determined by FT-IR spectroscopy calibrated by quantitative 13 C-NMR spectroscopy from crystallization extraction (CRYSTEX).
- the copolymer of propylene and ethylene preferably consists of propylene and ethylene. Thus, it is preferred that the total ethylene content and the total propylene content make up 100 wt.-% of the copolymer of propylene and ethylene.
- the copolymer of propylene and ethylene preferably has a melt flow rate MFR2 of from more than 2.5 to 6.0 g/10 min, more preferably from 2.8 to 5.5 g/10 min, still more preferably from 3.0 to 5.0 g/10 min and most preferably from 3.3 to 4.5 g/10 min, determined according to ISO 1133 at 230°C and 2.16 kg.
- the copolymer of propylene and ethylene is a heterophasic copolymer of propylene and ethylene.
- the heterophasic copolymer of propylene and ethylene has a matrix phase and an elastomeric phase dispersed in said matrix phase.
- the matrix phase is preferably a propylene-ethylene random copolymer.
- the matrix phase and the elastomeric phase usually cannot exactly be divided from each other.
- One method is the CRYSTEX method as described above for the polyolefin composition.
- the copolymer of propylene and ethylene preferably has a crystalline fraction (CF) content, determined by crystallization extraction (CRYSTEX), in the range from 75.0 to 90.0 wt.-%, more preferably from 77.0 to 88.0 wt.-%, still more preferably from 78.5 to 86.5 wt.-% and most preferably from 80.0 to 85.0 wt.-%, based on the total weight amount of the copolymer of propylene and ethylene.
- CF crystalline fraction
- Said crystalline fraction (CF) preferably has an ethylene content (C2(CF)), determined by FT-IR spectroscopy calibrated by quantitative 13 C-NMR spectroscopy, in the range from 1 .0 to 15.0 wt.-%, more preferably from 2.0 to 10.0 wt.-% and most preferably from 3.0 to 8.5 wt.-%, based on the total amount of monomer units in the crystalline fraction (CF).
- said crystalline fraction (CF) preferably has an intrinsic viscosity (iV(CF)), determined according to ISO 1628-3, in the range from 185 to 325 cm 3 /g, more preferably from 200 to 300 cm 3 /g and most preferably from 225 to 270 cm 3 /g.
- the copolymer of propylene and ethylene preferably has a soluble fraction (SF) content, determined by crystallization extraction (CRYSTEX), in the range from 10.0 to 25.0 wt.- %, more preferably from 12.0 to 23.0 wt.-%, still more preferably from 13.5 to 21 .5 wt.-% and most preferably from 15.0 to 20.0 wt.-%, based on the total weight amount of the copolymer of propylene and ethylene.
- SF soluble fraction
- Said soluble fraction (SF) preferably has an ethylene content (C2(SF)), determined by FT-IR spectroscopy calibrated by quantitative 13 C-NMR spectroscopy, in the range from 20.0 to 35.0 wt.-%, more preferably from 21 .5 to 32.5 wt.-% and most preferably from 23.0 to 30.0 wt.-%, based on the total amount of monomer units in the soluble fraction (SF).
- C2(SF) ethylene content
- said soluble fraction (SF) preferably has an intrinsic viscosity (iV(SF)), determined according to ISO 1628-3, in the range from 90 to 190 cm 3 /g, more preferably from 100 to 175 cm 3 /g and most preferably from 115 to 150 cm 3 /g.
- iV(SF) intrinsic viscosity
- the copolymer of propylene and ethylene preferably has a ratio of the ethylene content of the soluble fraction to the ethylene content of the crystalline fraction C2(SF)/C2(CF) of from 1.5 : 1.0 to 10.0 : 1.0, more preferably from 2.5 : 1.0 to 7.5 : 1.0, most preferably from 4.0 : 1.0 to 6.0 : 1.0.
- the copolymer of propylene and ethylene preferably has a ratio of the intrinsic viscosity of the soluble fraction to the intrinsic viscosity of the crystalline fraction iV(SF)/iV(CF) of from 0.35 : 1.00 to 0.90 : 1.00, preferably from 0.40 : 1.00 to 0.80 : 1.00, more preferably from 0.50 : 1 .00 to 0.70 : 1 .00.
- the copolymer of propylene and ethylene preferably has a total intrinsic viscosity (iV (total)) determined according to ISO 1628-3, in the range from 185 to 325 cm 3 /g, more preferably from 200 to 300 cm 3 /g and most preferably from 225 to 270 cm 3 /g.
- the crystalline fraction (CF) and the soluble fraction (SF) make up 100 wt.-% of the copolymer of propylene and ethylene.
- the copolymer of propylene and ethylene preferably has a flexural modulus of from more than 470 MPa to 750 MPa, more preferably of from 475 MPa to 700 MPa and most preferably of from 500 MPa to 650 MPa, determined according to ISO 178 method A.
- the copolymer of propylene and ethylene preferably has a Charpy notched impact strength at 23°C of from 5.0 to 30.0 kJ/m 2 , more preferably from 7.0 to 25.0 kJ/m 2 and most preferably from 9.0 to 20.0 kJ/m 2 , determined according to ISO 179-1/1 eA.
- the copolymer of propylene and ethylene preferably has a Charpy notched impact strength at -20°C of from 0.8 to 5.0 kJ/m 2 , more preferably from 0.8 to 4.0 kJ/m 2 and most preferably from 0.8 to 3.5 kJ/m 2 , determined according to ISO 179-1/1 eA.
- the copolymer of propylene and ethylene preferably has a melting temperature Tm of from 132 to 155°C, preferably from 135 to 150°C and most preferably from 137 to 147°C, determined by to DSC analysis according to ISO 11357 I part 3 /method C2.
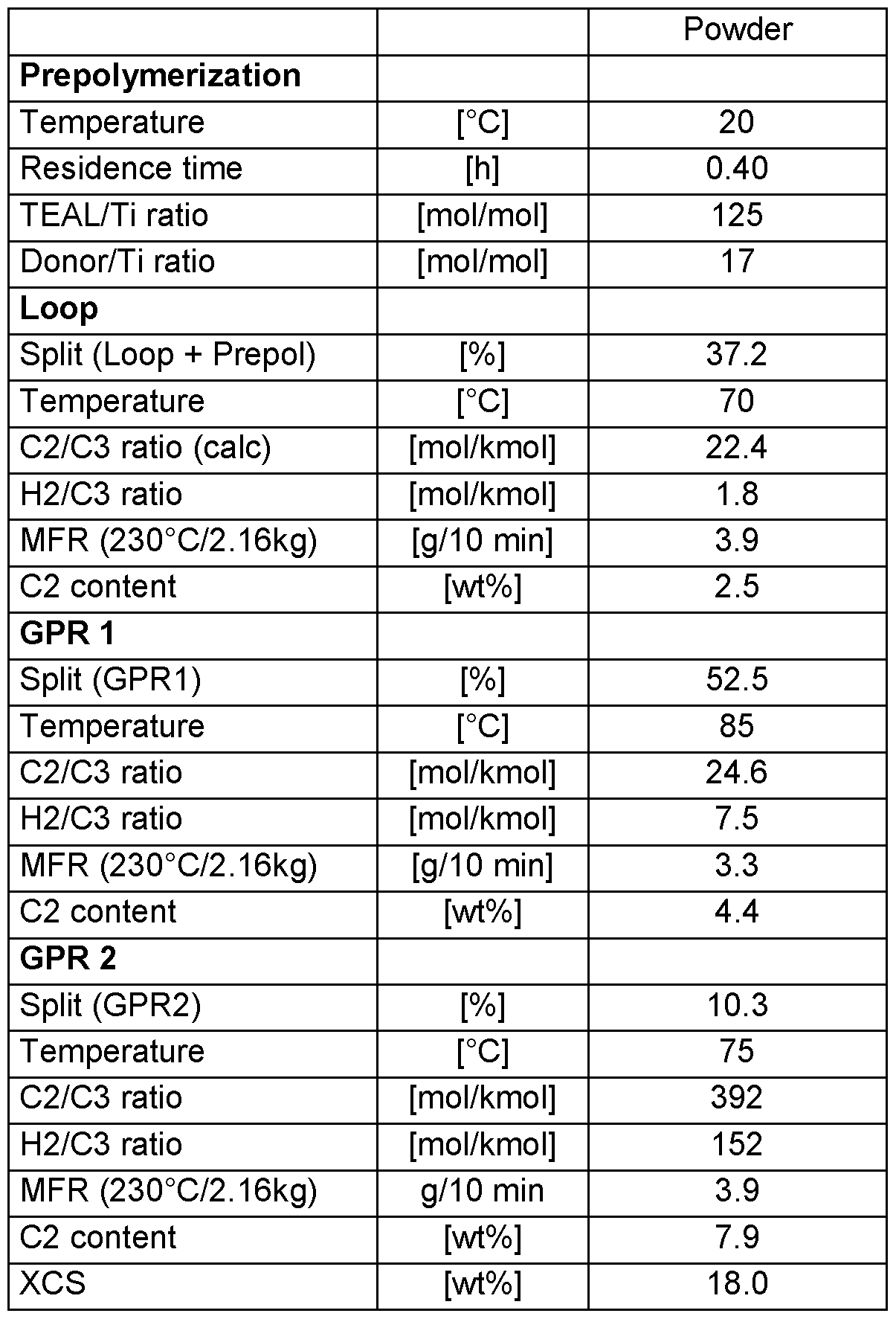
- Suitable sequential polymerization processes for polymerizing the copolymer of propylene and ethylene, preferably the heterophasic copolymer of propylene and ethylene, are e.g. disclosed in WO 2015/117948.
Landscapes
- Chemical & Material Sciences (AREA)
- Health & Medical Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Medicinal Chemistry (AREA)
- Polymers & Plastics (AREA)
- Organic Chemistry (AREA)
- Physics & Mathematics (AREA)
- Spectroscopy & Molecular Physics (AREA)
- Compositions Of Macromolecular Compounds (AREA)
Abstract
The present invention relates to a polyolefin composition having a total ethylene content of from more than 32.5 to 60.0 wt.-%, based on the total weight of the polyolefin composition and determined by FT-IR spectroscopy calibrated by quantitative 13C-NMR spectroscopy from crystallization extraction (CRYSTEX); a melt flow rate MFR2 of from more than 2.5 to 6.0 g/10 min, determined according to ISO 1133 at 230°C and 2.16 kg; a crystalline fraction (CF) content, determined by crystallization extraction (CRYSTEX), in the range from 75.0 to 95.0 wt.-%, based on the total weight amount of the polyolefin composition, said crystalline fraction (CF) having an ethylene content (C2(CF)), determined by FT-IR spectroscopy calibrated by quantitative 13C-NMR spectroscopy, in the range from more than 32.5 to 62.5 wt.-%, based on the total amount of monomer units in the crystalline fraction (CF), and a soluble fraction (SF) content, determined by crystallization extraction (CRYSTEX), in the range from 5.0 to 25.0 wt.-%, based on the total weight amount of the polyolefin composition, said soluble fraction (SF) having an intrinsic viscosity (iV(SF)), determined according to ISO 1628-3, in the range from 85 to 200 cm³/g, an article comprising said polyolefin composition, preferably a cable comprising an insulation layer comprising said polyolefin composition and the use of said polyolefin composition as cable insulation for medium voltage cables, high voltage cables, extra high voltage cable and ultra high voltage cables.
Description
Polyolefin composition for cable insulation
The present invention relates to a polyolefin composition, an article comprising said polyolefin composition, preferably a cable comprising an insulation layer comprising said polyolefin composition and the use of said polypropylene composition as cable insulation for medium and high voltage cables, especially for high voltage (HV) cable, extra high voltage (EHV) cables and ultra high voltage (UHV) cables.
Technical background
Nowadays, ethylene polymer products are used as insulation and semiconducting shields for medium, high and extra high voltage cables, due to easy processability and their beneficial electrical properties. However, for thermoplastic ethylene polymer products (PE and HDPE) according to IEC 62895 the maximum operation temperature, being the maximum conductor operation temperature in normal operation for high voltage DC cables up to 320 kV, are limited to 80°C.
Therefore, attempts were made using thermoplastic propylene polymers as insulation material for medium, high and extra high voltage (MV, HV and EHV) cables as well as high-voltage direct current (HVDC) cables. Propylene polymers offer a benefit especially for high and extra high voltage cables due to their higher maximum operating temperature.
WO 2022/200395 A1 and WO 2022/200396 A1 disclose flexible polypropylene compositions suitable for cable insulation, which both comprise a flexible heterophasic copolymer of propylene and ethylene. Said compositions show a superior balance of properties in regard of flexibility, impact strength and electric properties, which makes these compositions especially suitable for medium voltage cables.
However, for high voltage, extra-high voltage and ultra-high voltage cables, especially for high voltage DC cables specific minimum mechanical requirements are needed, as described in the CIGRE TB852 recommendations for HVDC cable systems.
In the present invention a polyolefin composition with a rather low amount of soluble fraction in crystallization extraction (CRYSTEX), which is a measure for the amorphous phase of the polyolefin composition, is proposed, which shows an improved balance of tensile and indentation properties and thermal conductivity and additionally shows a good performance during cable extrusion. The polyolefin composition thus is suitable for
cable insulation, especially for meeting all CIGRE TB852 recommendations for HVDC cable systems.
Summary of the invention
In one aspect the present invention relates to a polyolefin composition having a total ethylene content of from more than 32.5 to 60.0 wt.-%, preferably from 33.0 to 55.0 wt.-%, more preferably from 33.5 to 52.5 wt.-%, still more preferably from 34.0 to 50.0 wt.-%, based on the total weight of the polyolefin composition and determined by FT-IR spectroscopy calibrated by quantitative 13C-NMR spectroscopy from crystallization extraction (CRYSTEX); a melt flow rate MFR2 of from more than 2.5 to 6.0 g/10 min, preferably from 2.6 to 5.5 g/10 min, still more preferably from 2.7 to 5.0 g/10 min and most preferably from 2.8 to 4.5 g/10 min, determined according to ISO 1133 at 230°C and 2.16 kg; a crystalline fraction (CF) content, determined by crystallization extraction (CRYSTEX), in the range from 75.0 to 95.0 wt.-%, preferably from 77.5 to 93.5 wt.-%, more preferably from 80.0 to 92.5 wt.-% and most preferably from 82.5 to 91 .0 wt.-%, based on the total weight amount of the polyolefin composition, said crystalline fraction (CF) having an ethylene content (C2(CF)), determined by FT-IR spectroscopy calibrated by quantitative 13C-NMR spectroscopy, in the range from more than 32.5 to 62.5 wt.-%, preferably from 35.0 to 60.0 wt.-% and most preferably from 36.0 to 55.0 wt.-%, based on the total amount of monomer units in the crystalline fraction (CF); and a soluble fraction (SF) content, determined by crystallization extraction (CRYSTEX), in the range from 5.0 to 25.0 wt.-%, preferably from 6.5 to 22.5 wt.-%, more preferably from 7.5 to 20.0 wt.-% and most preferably from 9.0 to 17.5 wt.-%, based on the total weight amount of the polyolefin composition, said soluble fraction (SF) having an intrinsic viscosity (iV(SF)), determined according to ISO 1628-3, in the range from 85 to 200 cm3/g, more preferably from 95 to 175 cm3/g and most preferably from 100 to 150 cm3/g.
In another aspect, the present invention relates to an article comprising the polyolefin composition as described above or below.
Preferably, said article is a cable comprising an insulation layer comprising the polyolefin composition as described above or below.
In yet another aspect the present invention relates to the use of the polyolefin composition as described above or below as cable insulation for medium voltage cables, high voltage cables, extra high voltage cable and ultra high voltage cables, preferably for high voltage cables and extra high voltage cables, more preferably high voltage DC (HVDC) cables and extra high voltage DC (EHVDC) cables.
Definitions
A heterophasic polypropylene is a propylene-based copolymer with a semi-crystalline matrix phase, which can be a propylene homopolymer or a random copolymer of propylene and at least one alpha-olefin comonomer, and an elastomeric phase dispersed therein. The elastomeric phase can be a propylene copolymer with a high amount of comonomer, which is not randomly distributed in the polymer chain but are distributed in a comonomer-rich block structure and a propylene-rich block structure. A heterophasic polypropylene usually differentiates from a one-phasic propylene copolymer in that it shows two distinct glass transition temperatures Tg which are attributed to the matrix phase and the elastomeric phase.
A propylene homopolymer is a polymer, which essentially consists of propylene monomer units. Due to impurities especially during commercial polymerization processes a propylene homopolymer can comprise up to 0.1 mol% comonomer units, preferably up to 0.05 mol% comonomer units and most preferably up to 0.01 mol% comonomer units.
A propylene random copolymer is a copolymer of propylene monomer units and comonomer units in which the comonomer units are distributed randomly over the polypropylene chain. Thereby, a propylene random copolymer includes a fraction, which is insoluble in xylene - xylene cold insoluble (XCI) fraction - in an amount of at least 85 wt.-%, most preferably of at least 88 wt.-%, based on the total amount of propylene
random copolymer. Accordingly, the propylene random copolymer does not contain an elastomeric polymer phase dispersed therein.
Usually, a propylene polymer comprising at least two propylene polymer fractions (components), which have been produced under different polymerization conditions resulting in different (weight average) molecular weights and/or different comonomer contents for the fractions, preferably produced by polymerizing in multiple polymerization stages with different polymerization conditions, is referred to as “multimodal”. The prefix “multi” relates to the number of different polymer fractions the propylene polymer is consisting of. As an example of multimodal propylene polymer, a propylene polymer consisting of two fractions only is called “bimodal”, whereas a propylene polymer consisting of three fractions only is called “trimodal”.
A unimodal propylene polymer only consists of one fraction.
Thereby, the term “different” means that the propylene polymer fractions differ from each other in at least one property, preferably in the weight average molecular weight - which can also be measured in different melt flow rates of the fractions - or comonomer content or both.
An elastomer is a polymer with viscoelasticity and weak intermolecular forces. The term “elastomer” can be used interchangeably with “rubber”.
Polyolefin based elastomers, such as polypropylene based elastomers, i.e. an elastomer with a molar majority of olefin monomer units, such as propylene monomer units, are usually thermoplastic elastomers.
Thermoplastic elastomers have both thermoplastic and elastomeric properties. Polyolefin based elastomers, such as polypropylene based elastomers, usually show a low density and low viscosity. They can be propylene homopolymers or propylene-alpha olefin copolymers, such as propylene-ethylene copolymers. A specific class of polypropylene based elastomers are propylene homopolymers or propylene-alpha olefin copolymers, such as propylene-ethylene copolymers, which have been polymerized in the presence of a single site catalyst, usually in a solution polymerization process.
Vis-breaking is a post reactor chemical process for modifying semi-crystalline polymers such as propylene polymers. During the vis-breaking process, the propylene polymer backbone is degraded, for example by means of peroxides, such as organic peroxides, via beta scission. The degradation is generally used for increasing the melt flow rate and narrowing the molecular weight distribution.
A low density polyethylene is a polymer with predominately ethylene monomers, which is polymerized in a high pressure process by free radical polymerization. The low density polyethylene is characterized by long polymer side chains, which decrease the density of the polymer irrespective of the amount of comonomer.
According to CIGRE TB852 recommendations for HVDC transmission cable systems high voltage (HV) refers to voltages up to and including 400 kV for DC systems. Extra-high voltage (EHV) refers to voltages above 400 kV to and including 800 kV for DC systems.
Although not explicitly mentioned in said recommendations, ultra-high voltage (UHV) consequently refers to voltages of more than 800 kV for DC systems.
In the following amounts are given in % by weight (wt.-%) unless it is stated otherwise.
Detailed description of the invention
Polyolefin composition
The polyolefin composition as described above or below has a total ethylene content of from more than 32.5 to 60.0 wt.-%, preferably from 33.0 to 55.0 wt.-%, more preferably from 33.5 to 52.5 wt.-%, still more preferably from 34.0 to 50.0 wt.-%, based on the total weight of the polyolefin composition and determined by FT-IR spectroscopy calibrated by quantitative 13C-NMR spectroscopy from crystallization extraction (CRYSTEX); and a melt flow rate MFR2 of from more than 2.5 to 6.0 g/10 min, preferably from 2.6 to 5.5 g/10 min, still more preferably from 2.7 to 5.0 g/10 min and most preferably from 2.8 to 4.5 g/10 min, determined according to ISO 1133 at 230°C and 2.16 kg.
The polymeric part of the polyolefin composition is characterized by crystallization extraction (CRYSTEX) using trichlorobenzene (TCB) as a solvent. This method is described below in the determination methods section. The crystalline fraction (CF) contains for the most part the matrix phase and only a small part of the elastomeric phase and the soluble fraction (SF) contains for the most part the elastomeric phase and only a small part of the matrix phase. In some cases, this method results in more useful data compared to xylene extraction, since the crystalline fraction (CF) and the soluble fraction (SF) more accurately correspond to the matrix and elastomeric phases, respectively. Due to the differences in the separation methods of xylene extraction and crystallization extraction (CRYSTEX) the properties of XCS/XCI fractions on the one hand and crystalline/soluble (CF/SF) fractions on the other hand are not exactly the same, meaning that the amounts of matrix phase and elastomeric phase can differ as well as the properties.
Generally, the crystalline fraction (CF) content and the soluble (SF) content of a composition only relate to its polymeric components, i.e. without other components, which are insoluble and therefore not part of the dissolution and crystallization cycles as described below in the determination method.
In the present case the polyolefin composition is usually fully soluble in trichlorobenzene (TCB) so that the crystalline fraction (CF) content and the soluble (SF) content relate to the total content of the polyolefin composition. Thus, the crystalline fraction (CF) content and the soluble fraction (SF) content preferably make up 100 wt.-% of the polyolefin composition.
The polyolefin composition has a crystalline fraction (CF) content, determined by crystallization extraction (CRYSTEX), in the range from 75.0 to 95.0 wt.-%, preferably from 77.5 to 93.5 wt.-%, more preferably from 80.0 to 92.5 wt.-% and most preferably from 82.5 to 91 .0 wt.-%, based on the total weight amount of the polyolefin composition.
Said crystalline fraction (CF) has an ethylene content (C2(CF)), determined by FT-IR spectroscopy calibrated by quantitative 13C-NMR spectroscopy, in the range from more than 32.5 to 62.5 wt.-%, preferably from 35.0 to 60.0 wt.-% and most preferably from
36.0 to 55.0 wt.-%, based on the total amount of monomer units in the crystalline fraction (CF).
It is preferred that said crystalline fraction (CF) has a propylene content (C3(CF)), determined by FT-IR spectroscopy calibrated by quantitative 13C-NMR spectroscopy, in the range from 37.5 to less than 67.5 wt.-%, preferably from 40.0 to 65.0 wt.-% and most preferably from 45.0 to 64.0 wt.-%, based on the total amount of monomer units in the crystalline fraction (CF).
Further, said crystalline fraction (CF) preferably has an intrinsic viscosity (iV(CF)), determined according to ISO 1628-3, in the range from 100 to 250 cm3/g, more preferably from 125 to 225 cm3/g and most preferably from 140 to 210 cm3/g.
The polyolefin composition has a soluble fraction (SF) content, determined by crystallization extraction (CRYSTEX), in the range from 5.0 to 25.0 wt.-%, preferably from 6.5 to 22.5 wt.-%, more preferably from 7.5 to 20.0 wt.-% and most preferably from 9.0 to 17.5 wt.-%, based on the total weight amount of the polyolefin composition.
Said soluble fraction (SF) has an intrinsic viscosity (iV(SF)), determined according to ISO 1628-3, in the range from 85 to 200 cm3/g, more preferably from 95 to 175 cm3/g and most preferably from 100 to 150 cm3/g.
Further, said soluble fraction (SF) preferably has an ethylene content (C2(SF)), determined by FT-IR spectroscopy calibrated by quantitative 13C-NMR spectroscopy, in the range from 22.5 to 42.5 wt.-%, more preferably from 25.0 to 40.0 wt.-% and most preferably from 30.0 to 35.0 wt.-%, based on the total amount of monomer units in the soluble fraction (SF).
It is preferred that said soluble fraction (SF) has a propylene content (C3(SF)), determined by FT-IR spectroscopy calibrated by quantitative 13C-NMR spectroscopy, in the range from 57.5 to 77.5 wt.-%, preferably from 60.0 to 75.0 wt.-% and most
preferably from 62.5 to 70.0 wt.-%, based on the total amount of monomer units in the soluble fraction (SF).
The polyolefin composition preferably has a ratio of the ethylene content of the soluble fraction to the ethylene content of the crystalline fraction C2(SF)/C2(CF) of from 0.50 : 1.00 to 1 .00 : 1 .00, more preferably from 0.55 : 1 .00 to 0.95 : 1 .00, most preferably from 0.60 : 1.00 to 0.90 : 1.00.
Further, the polyolefin composition preferably has a ratio of the intrinsic viscosity of the soluble fraction to the intrinsic viscosity of the crystalline fraction iV(SF)/iV(CF) of from 0.40 : 1 .00 to 0.95 : 1 .00, more preferably from 0.50 : 1 .00 to 0.85 : 1 .00, most preferably from 0.55 : 1 .00 to 0.75 : 1 .00.
The polyolefin composition preferably has an intrinsic viscosity, determined according to ISO 1628-3, of from 100 to 210 cm3/g, more preferably from 120 to 200 cm3/g, most preferably from 130 to 190 cm3/g.
It is preferred that the polyolefin composition preferably comprises a total propylene content of from 40.0 to less than 67.5 wt.-%, preferably from 45.0 to 67.0 wt.-%, more preferably from 47.5 to 66.5 wt.-%, still more preferably from 49.0 to 66.0 wt.-%, based on the total weight of the polyolefin composition and determined by FT-IR spectroscopy calibrated by quantitative 13C-NMR spectroscopy from crystallization extraction (CRYSTEX).
The polymeric part of the polyolefin composition preferably consists of propylene monomer units and ethylene monomer units.
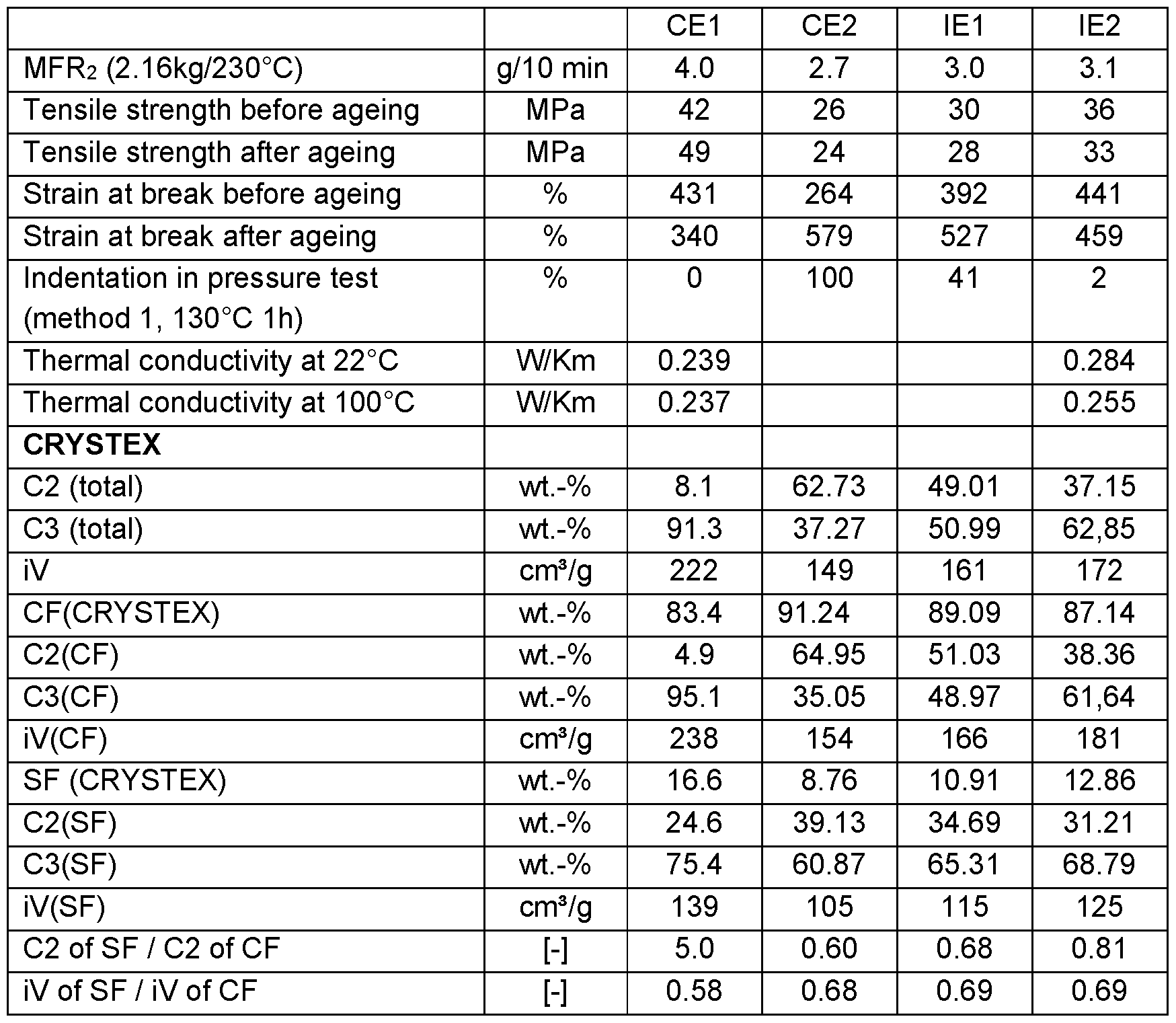
The polyolefin composition preferably has a good balance of properties in regard of tensile strength, strain at break and indentation as illustrated by the following properties:
The polypropylene composition preferably has a tensile strength of from 8.5 to 50 MPa, preferably from 15 to 45 MPa, more preferably from 20 to 40 MPa, determined
according to ISO 527-2 on 5A injection moulded test specimens at 23°C and a speed of 25 mm/min before ageing.
The polypropylene composition preferably has a tensile strength of from 8.5 to 50 MPa, preferably from 15 to 45 MPa, more preferably from 20 to 40 MPa, determined according to ISO 527-2 on 5A injection moulded test specimens at 23°C and a speed of 25 mm/min after ageing for 10 days at 135°C.
Further, the polyolefin composition preferably has a strain at break of from 350 to 650 %, preferably from 360 to 600 %, more preferably from 375 to 575 %, determined according to ISO 527-2 on 5A injection moulded test specimens at 23°C and a speed of 25 mm/min before ageing.
Still further, the polyolefin composition preferably has a strain at break of from 350 to 650 %, preferably from 360 to 600 %, more preferably from 375 to 575 %, determined according to ISO 527-2 on 5A injection moulded test specimens at 23°C and a speed of 25 mm/min after ageing for 10 days at 135°C.
Furthermore, the polyolefin composition preferably has an indentation in the pressure test of from 0 to 50 %, preferably of from 0 to 45 %, determined on compression moulded plaques with a cylindric indentor according to method 1 pressure test in the CIGRE TB852 recommendations at 130°C for 1 h.
Additionally, the polyolefin composition preferably has a thermal conductivity of at least 0.245 W/Km, such as from 0.245 to 0.400 W/Km, preferably from 0.250 to 0.375 W/Km, more preferably from 0.260 to 0.350 W/Km, determined according to ISO 22007-2 at 22°C.
Further, the polyolefin composition preferably has a thermal conductivity of at least 0.240 W/Km, such as from 0.240 to 0.375 W/Km, preferably from 0.245 to 0.350 W/Km, more preferably from 0.248 to 0.325 W/Km, determined according to ISO 22007-2 at 22°C.
The polyolefin composition thus preferably meets all all CIGRE TB852 recommendations for HVDC cable systems, also after ageing.
Additionally the polyolefin composition shows good thermal conductivity over a broad temperature range of 22 to 100°C.
It is preferred that the polyolefin composition comprises from 40.0 to less than 70.0 wt.-%, preferably from 45.0 to 69.0 wt.-%, more preferably from 47.5 to 67.5 wt.-% of a copolymer of propylene and ethylene and from more than 30.0 to 60.0 wt.-%, preferably from 31 .0 to 55.0 wt.-%, more preferably from 32.5 to 52.5 wt.-% of a low density polyethylene, wherein all weight amounts are based on the total weight of the polyolefin composition.
The polyolefin composition can further comprise polymeric components, which are different from the copolymer of propylene and ethylene and the low density polyethylene, in an amount of preferably 0.0 to 10.0 wt.-% based on the total weight of the polyolefin composition.
In a preferred embodiment the polymeric components of the polyolefin composition consist of copolymer of propylene and ethylene and the low density polyethylene. Besides these polymeric components the polyolefin composition can comprise one or more additives in an amount of from 0.0 up to 5.0 wt.-%, based on the total weight of the polyolefin composition. The one or more additives are preferably selected from acid scavengers, antioxidants, alpha nucleating agents, beta nucleating agents, etc. Such additives are commercially available and for example described in “Plastic Additives Handbook”, 6th edition 2009 of Hans Zweifel (pages 1141 to 1190).
Usually, these additives are added in quantities of 1 to 50000 ppm for each single component.
The one or more additives can be added to the polymeric components in a blending step.
Thereby, the one or more additives can be added to the polymeric components in form of master batches in which one or more additives are blended with a carrier polymer in concentrated amounts. Any optional carrier polymer is calculated to the amount of additives, based on the total weight of the polyolefin composition.
Preferably, the polyolefin composition contains from 0 to 5.00 wt.-%, more preferably from 0 to 2.50 wt.-% of the alpha-nucleating agent, based on the total amount of the polyolefin composition.
The amount of pure alpha-nucleating agent in the polyolefin composition (without optional carrier polymer of a master batch) is preferably in the range of from 0 to 5000 ppm, more preferably from 0 to 4000 ppm, based on the total amount of the polyolefin composition.
The alpha-nucleating agent is generally not restricted.
Preferably the alpha-nucleating agent is selected from soluble alpha-nucleating agents and polymeric alpha-nucleating agents.
The alpha-nucleating agent is preferably selected from the group consisting of
(i) dibenzylidenesorbitol (e.g. 1 ,3 : 2,4 dibenzylidenesorbitol) and Ci-Cs-alkyl- substituted dibenzylidenesorbitol derivatives, such as methyldibenzylidenesorbitol, ethyldibenzylidenesorbitol or dimethyldibenzylidenesorbitol (e.g. 1 ,3 : 2,4 di(methylbenzylidene) sorbitol), or substituted nonitol-derivatives, such as 1 ,2,3- trideoxy-4,6:5,7-bis-0-[(4-propylphenyl)methylene]-nonitol, and
(ii) vinylcycloalkane polymer and vinylalkane polymer (as discussed in more detail below), and
(iii) mixtures thereof.
The alpha-nucleating agent is preferably selected from the group consisting of dibenzylidenesorbitol (e.g. 1 ,3 : 2,4 dibenzylidene sorbitol), dibenzylidenesorbitol derivative, preferably dimethyldibenzylidenesorbitol (e.g. 1 ,3 : 2,4 di(methylbenzylidene) sorbitol), or substituted nonitol-derivatives, such as 1 ,2,3-trideoxy-4,6:5,7-bis-0-[(4- propylphenyl)methylene]-nonitol, vinylcycloalkane polymer, vinylalkane polymer, and mixtures thereof.
Especially preferred are vinylcycloalkane polymers such as e.g. vinylcyclohexane (VCH) polymers. Such polymers can be added e.g. using Borealis Nucleation Technology (BNT).
In a preferred embodiment the alpha-nucleating agent is a soluble alpha-nucleating agent, more preferably a soluble alpha-nucleating agent selected from dibenzylidenesorbitol (e.g. 1 ,3 : 2,4 dibenzylidenesorbitol) and Ci-Cs-alkyl-substituted dibenzylidenesorbitol derivatives, such as methyldibenzylidenesorbitol, ethyldibenzylidenesorbitol or dimethyldibenzylidenesorbitol (e.g. 1 ,3 : 2,4 di(methylbenzylidene) sorbitol), or substituted nonitol-derivatives, such as 1 ,2,3- trideoxy-4,6:5,7-bis-0-[(4-propylphenyl)methylene]-nonitol and mixtures thereof.
The alpha-nucleating agent can be added to the polyolefin composition as an isolated raw material or in a mixture with a carrier polymer, i.e. in a so-called master batch. The amount of the carrier polymer of the master batch thereby is calculated to the amount of the alpha-nucleating agent.
In one embodiment the polyolefin composition comprises an alpha-nucleating agent. In said embodiment, the pure amount of alpha-nucleating agent is preferably in the range of from 0.1 to 5000 ppm, more preferably from 1 to 5000 ppm.
In another embodiment, the polyolefin composition does not comprise an alphanucleating agent.
In said embodiment, the pure amount of alpha-nucleating agent is 0 ppm. Preferably, the polyolefin composition is prepared by melt blending the copolymer of propylene and ethylene and the low density polyethylene, the optional additional polymeric components and the optional further additives, all as described above or below.
The polyolefin composition is preferably not subjected to vis-breaking.
In one embodiment the polyolefin composition does not comprise, i.e. is free of a dielectric fluid, such as e.g. described in EP 2 739 679.
In the following, the copolymer of propylene and ethylene and the low density polyethylene are described in more detail.
The polyolefin composition as described above or below preferably comprises a copolymer of propylene and ethylene.
The copolymer of propylene and ethylene is preferably present in the polyolefin composition in an amount of from 40.0 to less than 70.0 wt.-%, preferably from 45.0 to 69.0 wt.-%, more preferably from 47.5 to 67.5 wt.-%, based on the total weight of the polyolefin composition.
The copolymer of propylene and ethylene preferably has a total ethylene content of from 4.0 to 11 .0 wt.-%, more preferably from 5.0 to 10.0 wt.-%, still more preferably from 5.5 to 9.5 wt.-%, most preferably from 6.0 to 9.0 wt.-%, based on the total weight of the copolymer of propylene and ethylene and determined by FT-IR spectroscopy calibrated by quantitative 13C-NMR spectroscopy from crystallization extraction (CRYSTEX).
Further, the copolymer of propylene and ethylene preferably has a total propylene content of from 89.0 to 96.0 wt.-%, more preferably from 90.0 to 95.0 wt.-%, still more preferably from 90.5 to 95.5 wt.-%, most preferably from 91 .0 to 94.0 wt.-%, based on the total weight of the copolymer of propylene and ethylene and determined by FT-IR spectroscopy calibrated by quantitative 13C-NMR spectroscopy from crystallization extraction (CRYSTEX).
The copolymer of propylene and ethylene preferably consists of propylene and ethylene. Thus, it is preferred that the total ethylene content and the total propylene content make up 100 wt.-% of the copolymer of propylene and ethylene.
The copolymer of propylene and ethylene preferably has a melt flow rate MFR2 of from more than 2.5 to 6.0 g/10 min, more preferably from 2.8 to 5.5 g/10 min, still more preferably from 3.0 to 5.0 g/10 min and most preferably from 3.3 to 4.5 g/10 min, determined according to ISO 1133 at 230°C and 2.16 kg.
It is preferred that the copolymer of propylene and ethylene is a heterophasic copolymer of propylene and ethylene.
The heterophasic copolymer of propylene and ethylene has a matrix phase and an elastomeric phase dispersed in said matrix phase.
The matrix phase is preferably a propylene-ethylene random copolymer.
In the copolymer of propylene and ethylene, such as a heterophasic copolymer of propylene and ethylene, the matrix phase and the elastomeric phase usually cannot exactly be divided from each other. In order to characterize the matrix phase and the elastomeric phase of a heterophasic copolymer of propylene and ethylene several methods are known. One method is the CRYSTEX method as described above for the polyolefin composition.
The copolymer of propylene and ethylene preferably has a crystalline fraction (CF) content, determined by crystallization extraction (CRYSTEX), in the range from 75.0 to 90.0 wt.-%, more preferably from 77.0 to 88.0 wt.-%, still more preferably from 78.5 to 86.5 wt.-% and most preferably from 80.0 to 85.0 wt.-%, based on the total weight amount of the copolymer of propylene and ethylene.
Said crystalline fraction (CF) preferably has an ethylene content (C2(CF)), determined by FT-IR spectroscopy calibrated by quantitative 13C-NMR spectroscopy, in the range from 1 .0 to 15.0 wt.-%, more preferably from 2.0 to 10.0 wt.-% and most preferably from 3.0 to 8.5 wt.-%, based on the total amount of monomer units in the crystalline fraction (CF).
Further, said crystalline fraction (CF) preferably has an intrinsic viscosity (iV(CF)), determined according to ISO 1628-3, in the range from 185 to 325 cm3/g, more preferably from 200 to 300 cm3/g and most preferably from 225 to 270 cm3/g.
The copolymer of propylene and ethylene preferably has a soluble fraction (SF) content, determined by crystallization extraction (CRYSTEX), in the range from 10.0 to 25.0 wt.- %, more preferably from 12.0 to 23.0 wt.-%, still more preferably from 13.5 to 21 .5 wt.-% and most preferably from 15.0 to 20.0 wt.-%, based on the total weight amount of the copolymer of propylene and ethylene.
Said soluble fraction (SF) preferably has an ethylene content (C2(SF)), determined by FT-IR spectroscopy calibrated by quantitative 13C-NMR spectroscopy, in the range from 20.0 to 35.0 wt.-%, more preferably from 21 .5 to 32.5 wt.-% and most preferably from 23.0 to 30.0 wt.-%, based on the total amount of monomer units in the soluble fraction (SF).
Further, said soluble fraction (SF) preferably has an intrinsic viscosity (iV(SF)), determined according to ISO 1628-3, in the range from 90 to 190 cm3/g, more preferably from 100 to 175 cm3/g and most preferably from 115 to 150 cm3/g.
The copolymer of propylene and ethylene preferably has a ratio of the ethylene content of the soluble fraction to the ethylene content of the crystalline fraction C2(SF)/C2(CF) of from 1.5 : 1.0 to 10.0 : 1.0, more preferably from 2.5 : 1.0 to 7.5 : 1.0, most preferably from 4.0 : 1.0 to 6.0 : 1.0.
Further, the copolymer of propylene and ethylene preferably has a ratio of the intrinsic viscosity of the soluble fraction to the intrinsic viscosity of the crystalline fraction iV(SF)/iV(CF) of from 0.35 : 1.00 to 0.90 : 1.00, preferably from 0.40 : 1.00 to 0.80 : 1.00, more preferably from 0.50 : 1 .00 to 0.70 : 1 .00.
The copolymer of propylene and ethylene preferably has a total intrinsic viscosity (iV (total)) determined according to ISO 1628-3, in the range from 185 to 325 cm3/g, more preferably from 200 to 300 cm3/g and most preferably from 225 to 270 cm3/g.
It is preferred that the crystalline fraction (CF) and the soluble fraction (SF) make up 100 wt.-% of the copolymer of propylene and ethylene.
The copolymer of propylene and ethylene preferably has a flexural modulus of from more than 470 MPa to 750 MPa, more preferably of from 475 MPa to 700 MPa and most preferably of from 500 MPa to 650 MPa, determined according to ISO 178 method A.
Further, the copolymer of propylene and ethylene preferably has a Charpy notched impact strength at 23°C of from 5.0 to 30.0 kJ/m2, more preferably from 7.0 to 25.0 kJ/m2 and most preferably from 9.0 to 20.0 kJ/m2, determined according to ISO 179-1/1 eA.
Still further, the copolymer of propylene and ethylene preferably has a Charpy notched impact strength at -20°C of from 0.8 to 5.0 kJ/m2, more preferably from 0.8 to 4.0 kJ/m2 and most preferably from 0.8 to 3.5 kJ/m2, determined according to ISO 179-1/1 eA.
Furthermore, the copolymer of propylene and ethylene preferably has a melting temperature Tm of from 132 to 155°C, preferably from 135 to 150°C and most preferably from 137 to 147°C, determined by to DSC analysis according to ISO 11357 I part 3 /method C2.
Further, the copolymer of propylene and ethylene preferably has a crystallization temperature Tc of from 85 to 115°C, preferably from 90 to 110°C and most preferably from 95 to 107°C, determined by to DSC analysis according to ISO 11357 I part 3 /method C2.
The difference of the melting temperature to the crystallization temperature Tm-Tc is preferably in the range of from 35 to 55°C, more preferably 37 to 52°C and most preferably from 39 to 50°C.
The copolymer of propylene and ethylene preferably has a complex viscosity at a frequency of 100 rad/s eta*ioo rad/s of from 650 to 900 Pa s, more preferably from 700 to 850 Pa s, most preferably from 725 to 800 Pa s.
The copolymer of propylene and ethylene can be polymerized in a sequential multistage polymerization process, i.e. in a polymerization process in which two or more polymerization reactors are connected in series. Preferably, in the sequential multistage polymerization process, two or more, more preferably three or more, such as three or four, polymerization reactors are connected in series. The term “polymerization reactor” shall indicate that the main polymerization takes place. Thus in case the process consists of four polymerization reactors, this definition does not exclude the option that the overall process comprises for instance a pre-polymerization step in a prepolymerization reactor.
When the copolymer of propylene and ethylene is a heterophasic copolymer of propylene and ethylene, the matrix phase of the heterophasic copolymer of propylene and ethylene is polymerized in first polymerization reactor for producing a unimodal matrix phase or in the first and second polymerization reactor for producing a multimodal matrix phase.
The elastomeric phase of the heterophasic copolymer of propylene and ethylene is preferably polymerized in the subsequent one or two polymerization reactor(s) in the presence of the matrix phase for producing a unimodal elastomeric phase or a multimodal elastomeric phase.
Preferably, the polymerization reactors are selected from slurry phase reactors, such as loop reactors and/or gas phase reactors such as fluidized bed reactors, more preferably from loop reactors and fluidized bed reactors.
A preferred sequential multistage polymerization process is a “loop-gas phase”-process, such as developed by Borealis A/S, Denmark (known as BORSTAR® technology) described e.g. in patent literature, such as in EP 0 887 379, WO 92/12182
WO 2004/000899, WO 2004/111095, WO 99/24478, WO 99/24479 or in WO 00/68315.
A further suitable slurry-gas phase process is the Spheripol® process of LyondellBasell.
Suitable sequential polymerization processes for polymerizing the copolymer of propylene and ethylene, preferably the heterophasic copolymer of propylene and ethylene, are e.g. disclosed in WO 2015/117948.
The copolymer of propylene and ethylene, preferably the heterophasic copolymer of propylene and ethylene can be polymerized in the presence of a Ziegler-Natta catalyst. Suitable Ziegler-Natta catalysts are e.g. disclosed in WO 2015/117948.
The copolymer of propylene and ethylene is preferably not subjected to a visbreaking step as e.g. described in WO 2013/092620 A1 .
Heterophasic propylene copolymer resins suitable as copolymer of propylene and ethylene are also commercially available. These resins are usually already additivated with stabilizer packages. Thus, when using commercially available resins as copolymer of propylene and ethylene the addition of additives as described above might have to be adjusted to the already present additives.
In case of a commercially available copolymer of propylene and ethylene the above stated properties can be measured using a common measurement method or verified by the technical documentation provided by the supplier.
Low density polyethylene
The polyolefin composition as described above or below preferably comprises a low density polyethylene.
The low density polyethylene. is preferably present in the polyolefin composition in an amount of from 30.0 to 60.0 wt.-%, preferably from 31 .0 to 55.0 wt.-%, more preferably from 32.5 to 52.5 wt.-%, based on the total weight of the polyolefin composition.
It is preferred that the low density polyethylene is an ethylene homopolymer.
The low density polyethylene preferably has a melt flow rate MFR2 of from 0.1 to 2.5 g/10 min, preferably from 0.2 to 2.3 g/10 min, still more preferably from 0.3 to 2.0 g/10 min and most preferably from 0.5 to 1 .5 g/10 min, determined according to ISO 1133 at 190°C and 2.16 kg.
Further, the low density polyethylene preferably has a density of from 915 to 932 kg/m3, preferably from 917 to 930 kg/m3, more preferably from 918 to 928 kg/m3, determined according to ISO 1183.
Still further, the low density polyethylene preferably has a melting temperature Tm of from 105 to 125°C, preferably from 107 to 120°C, more preferably from 109 to 117°C, determined by to DSC analysis according to ISO 113571 part 3 /method C2.
Furthermore, the low density polyethylene preferably has a Vicat A50 softening temperature of from 85 to 105°C, preferably from 87 to 102°C, more preferably from 90 to 100°C, determined according to ISO 306 at 10 N.
The low density polyethylene preferably has a complex viscosity at a frequency of 100 rad/s eta*ioo rad/s of from 600 to 800 Pa s, more preferably from 625 to 775 Pa s, most preferably from 650 to 750 Pa s.
The low density polyethylene is preferably selected as such that the difference between the complex viscosity at a frequency of 100 rad/s of the copolymer of propylene (eta*wo rad/s (PP)) and the complex viscosity at a frequency of 100 rad/s of the low density polyethylene (eta* 100 rad/s (LDPE)) | eta*wo rad/s (PP) - eta*wo rad/s (LDPE)| is not more than ± 20 % of eta*wo rad/s (PP), preferably not more than ± 15 % of eta*wo rad/s (PP).
The low density polyethylene is polymerized in a high pressure polymerization process by means of free radical polymerization, preferably a tubular high pressure polymerization process. Said high pressure polymerization processes, especially tubular high pressure polymerization processes are well known in the art.
Low density polyethylene resins suitable as low density polyethylene are also commercially available. These resins are usually already additivated with stabilizer packages. Thus, when using commercially available resins as low density polyethylene the addition of additives as described above might have to be adjusted to the already present additives.
In case of a commercially available low density polyethylene the above stated properties can be measured using a common measurement method or verified by the technical documentation provided by the supplier.
Article
In a further aspect, the present invention further relates to an article comprising the polyolefin composition as defined above or below.
The article is preferably a cable, preferably a medium voltage cable voltage cables, high voltage cables, extra high voltage cable or ultra high voltage cable, more preferably a high voltage cable or an extra high voltage cable, even more preferably a high voltage DC (HVDC) cable or an extra high voltage DC (EHVDC) cable, comprising an insulation layer comprising the polyolefin composition as described above or below.
The insulation layer preferably comprises from 90 to 100 wt.-%, preferably from 95 to 100 wt.-%, still more preferably from 99 to 100 wt.-%, based on the total weight of the insulation layer, most preferably consists of the polyolefin composition.
It is preferred that the article meets all CIGRE TB852 recommendations for HVDC cable systems.
The cable usually comprises of at least one conductor and at least one insulation layer comprising the polyolefin composition as described above or below.
The term "conductor" means herein above and below that the conductor comprises one or more wires. The wire can be for any use and be e.g. telecommunication or electrical wire. Moreover, the cable may comprise one or more such conductors. Preferably the conductor is an electrical conductor and comprises one or more metal wires. The cable is preferably a power cable. A power cable is defined to be a cable transferring energy operating at any voltage, typically operating at voltages higher than 1 kV. The voltage applied to the power cable can be alternating (AC), direct (DC), or transient (impulse). The polyolefin composition of the invention is very suitable for power cables.
The term "cable" as used herein, is intended to denote a cable comprising at least one cable core, optionally two cable cores or three cable cores. Each “cable core” as used herein, comprises a conductor surrounded by at least an inner semiconductive layer, an insulation layer and an outer semiconductive layer. The one or more cable cores in the cable may be surrounded by at least one reinforcing layer and/or an armouring layer adapted for mechanical protection of the cable. The armouring layer may comprise metallic wires, braid, sheath or low loss armour. These variations and cable constructions are familiar to the person skilled in the art. The armouring layer may extend across parts of the cable.
The term “DC cable” refers to a direct current (DC) cable generally comprising one or more cable cores, preferably one or two cable cores.
The cable according to the present invention is very advantageously a DC power cable, which can be e.g. a medium voltage (MV), a high voltage (HV) or an extra high voltage (EHV) or an ultra-high voltage (UHV) DC cable, which terms, as well known, indicate the level of operating voltage.
The DC power cable may operate at voltages of at least 30kV, such as a HVDC, EHVDC or UHVDC cable. For HVDC, EHVDC or UHVDC cables the operating voltage is defined herein as the electric voltage between ground and the conductor of the high voltage cable.
The cable may be an HVDC, EHVDC or UHVDC power cable operating at voltages of 40 kV or higher, even at voltages of 50 kV or higher even 60 kV or higher. In some embodiments, the operating voltage may be higher than 80 kV. The upper limit is not limited. A practical upper limit can be up to 1100 kV.
As well known the cable can optionally comprise further layers, e.g. layers surrounding the outer semiconductive layers, such as screen(s), a jacketing layer, other protective layer(s) or any combinations thereof.
In yet another aspect, the present invention relates to the use of the polyolefin composition as described above or below as cable insulation for medium voltage cables, high voltage cables, extra high voltage cable and ultra high voltage cables, preferably for high voltage cables and extra high voltage cables, more preferably high voltage DC (HVDC) cables and extra high voltage DC (EHVDC) cables.
Benefits of the invention:
The polyolefin composition shows a good balance of tensile and indentation properties which allow the use as cable insulation e.g. for medium voltage cables, high voltage cables, extra high voltage cable and ultra high voltage cables at high operation temperatures, in particular for high voltage DC (HVDC) cables and extra high voltage DC (EHVDC) cables.
The rather low soluble fraction (SF) content of the polyolefin composition of not more than 25.0 wt.-% allows for improved extrudability during cable extrusion.
By adding the low density polyethylene to the polyolefin composition the strain to break can be improved whereby the indentation in pressure test remains low even after ageing so that the polyolefin composition meets all CIGRE TB852 recommendations for HVDC cable systems.
Additionally, the addition of the low density polyethylene improved the thermal conductivity of the polyolefin composition over a broad temperature range.
Examples
The following definitions of terms and determination methods apply for the above general description of the invention as well as to the below examples unless otherwise defined.
1. Measurement methods a) Melt Flow Rate (MFR2)
The melt flow rate is the quantity of polymer in grams which the test apparatus standardized to ISO 1133 extrudes within 10 minutes at a certain temperature under a certain load.
The melt flow rate MFR2 of propylene based polymers and the polyolefin composition is measured at 230°C with a load of 2.16 kg according to ISO 1133.
The melt flow rate MFRs of the polyolefin composition is measured at 230°C with a load of 5.0 kg according to ISO 1133.
The melt flow rate MFR2 of the ethylene based polymers is measured at 190°C with a load of 2.16 kg according to ISO 1133. b) Density
The density is measured according to ISO 1183-1 :2004 Method A on compression moulded specimen prepared according to EN ISO 1872-2 (Feb 2007) and is given in kg/m3. c) Comonomer content
Comonomer content quantification of poly(propylene-co-ethylene) copolymers Quantitative 13C{1H} NMR spectra were recorded in the solution-state using a Bruker Avance NEO 400 NMR spectrometer operating at 400.15 and 100.62 MHz for 1H and 13C respectively. All spectra were recorded using a 13C optimised 10 mm extended temperature probe head at 125°C using nitrogen gas for all pneumatics. Approximately 200 mg of material was dissolved in 3 ml of 7,2-tetrachloroethane-c/2 (TCE-cfe) along with chromium-(lll)-acetylacetonate (Cr(acac)3) resulting in a 60 mM solution of relaxation agent in solvent {8} and with approximately 3 mg BHT (2 ,6-di-tert-butyl-4- methylphenol CAS 128-37-0) . To ensure a homogenous solution, after initial sample
preparation in a heat block, the NMR tube was further heated in a rotatory oven for at least 1 hour. Upon insertion into the magnet the tube was spun at 10 Hz. This setup was chosen primarily for the high resolution and quantitatively needed for accurate ethylene content quantification. Standard single-pulse excitation was employed without NOE, using an optimised tip angle, 1 s recycle delay and a bi-level WALTZ16 decoupling scheme {3, 4}. A total of 6144 (6k) transients were acquired per spectra.
Quantitative 13C{1H} NMR spectra were processed, integrated and relevant quantitative properties determined from the integrals using proprietary computer programs. All chemical shifts were indirectly referenced to the central methylene group of the ethylene block (EEE) at 30.00 ppm using the chemical shift of the solvent. This approach allowed comparable referencing even when this structural unit was not present. Characteristic signals corresponding to the incorporation of ethylene were observed {7}.
The comonomer fraction was quantified using the method of Wang et. al. {6} through integration of multiple signals across the whole spectral region in the 13C{1H} spectra. This method was chosen for its robust nature and ability to account for the presence of regiodefects when needed. Integral regions were slightly adjusted to increase applicability across the whole range of encountered comonomer contents.
For systems where only isolated ethylene in PPEPP sequences was observed the method of Wang et al. was modified to reduce the influence of non-zero integrals of sites that are known to not be present. This approach reduced the overestimation of ethylene content for such systems and was achieved by reduction of the number of sites used to determine the absolute ethylene content to:
E = 0.5 (Spp + Spy + Spb + 0.5( Sap + Say))
Through the use of this set of sites the corresponding integral equation becomes: E = 0.5 (IH +IG + 0.5(lc + ID)) using the same notation used in the article of Wang et al. {6}. Equations used for absolute propylene content were not modified.
The mole percent comonomer incorporation was calculated from the mole fraction:
E [mol%] = 100 * fE
The weight percent comonomer incorporation was calculated from the mole fraction: E [wt%] = 100 * (fE * 28.06 ) / ( (fE * 28.06) + ((1-fE) * 42.08) ) Bibliographic references:
1) Busico, V., Cipullo, R., Prog. Polym. Sci. 26 (2001) 443.
2) Busico, V., Cipullo, R., Monaco, G., Vacatello, M., Segre, A.L., Macromolecules 30 (1997) 6251.
3) Zhou, Z., Kuemmerle, R., Qiu, X., Redwine, D., Cong, R., Taha, A., Baugh, D. Winniford, B., J. Mag. Reson. 187 (2007) 225.
4) Busico, V., Carbonniere, P., Cipullo, R., Pellecchia, R., Severn, J., Talarico, G., Macromol. Rapid Commun. 2007, 28, 1128.
5) Resconi, L., Cavallo, L., Fait, A., Piemontesi, F., Chem. Rev. 2000, 100, 1253.
6) Wang, W-J., Zhu, S., Macromolecules 33 (2000), 1157.
7) Cheng, H. N., Macromolecules 17 (1984), 1950.
8) Singh, G., Kothari, A., Gupta, V., Polymer Testing 28 5 (2009), 475.
9) Kakugo, M., Naito, Y., Mizunuma, K., Miyatake, T. Macromolecules 15 (1982) 1150.
10) Randall, J. Macromol. Sci., Rev. Macromol. Chem. Phys. 1989, C29, 201.
11) Resconi, L., Cavallo, L., Fait, A., Piemontesi, F., Chem. Rev. 2000, 100, 1253. d) Differential scanning calorimetry (DSC) analysis, melting temperature (Tm) and crystallization temperature (Tc): measured with a TA Instrument Q2000 differential scanning calorimetry (DSC) on 5 to 7 mg samples. DSC is run according to ISO 11357 I part 3 /method C2 in a heat I cool /heat cycle with a scan rate of 10°C/min in the temperature range of -30°C to +225°C. Crystallization temperature and heat of crystallization (He) are determined from the cooling step, while melting temperature and heat of fusion (Hf) are determined from the second heating step.
When a sample shows two or more melting temperatures and/or crystallization temperatures only the main melting temperature (at the highest Hf) and main crystallization temperature (at the highest He) are displayed in the accordant table. The difference of melting temperature and crystallization temperature (Tm-Tc) is given for the main melting temperature and the main crystallization temperature. e) Crystallisation extraction (CRYSTEX) analysis
Note: Crystallization extraction (CRYSTEX) analyses the polymeric part of each component, with non-polymeric parts, such as any fillers or particulate pigments, not contributing to the reported CRYSTEX data presented.
Determination of Crystalline and soluble fractions and their respective properties (iV and Ethylene content)
The crystalline (CF) and soluble fractions (SF) of the polypropylene (PP) compositions as well as the comonomer content and intrinsic viscosities of the respective fractions were analyzed by use of the Crystex (crystallisation extraction) method. Potential instruments that can be used are Crystex QC or Crystex 42 (Polymer Char; Valencia, Spain). Details of the technique and the method can be found in literature (Ljiljana Jeremie, Andreas Albrecht, Martina Sandholzer & Markus Gahleitner (2020): Rapid characterization of high-impact ethylene-propylene copolymer composition by crystallization extraction separation: comparability to standard separation methods, International Journal of Polymer Analysis and Characterization, 25:8, 581 - 596).
The crystalline and amorphous fractions are separated through temperature cycles of dissolution at 160°C, crystallization at 40°C and re-dissolution in 1 ,2,4-trichlorobenzene at 160°C. Quantification of SF and CF and determination of ethylene content (C2) are achieved by means of an integrated infrared detector (IR4) and for the determination of the intrinsic viscosity (IV) an online 2-capillary viscometer is used.
IR4 detector is a multiple wavelength detector measuring IR absorbance at two different bands (CH3 stretching vibration (centred at app. 2960 cm-1) and the CH stretching vibration (2700-3000 cm'1) that are serving for the determination of the concentration and the Ethylene content in Ethylene-Propylene copolymers. IR4 detector is calibrated with series of 8 EP copolymers with known Ethylene content in the range of 2 wt.-% to 69 wt.-% (determined by 13C-NMR) and each at various concentrations, in the range of 2 and 13mg/ml. To encounter for both features, concentration and ethylene content at the same time for various polymer concentration expected during Crystex analyses the following calibration equations were applied:
Cone = a + b*Abs(CH) + c*(Abs(CH))2 + d*Abs(CH3) + e*(Abs(CH3)2 + f*Abs(CH)*Abs(CH3) (Equation 1)
CH3/1000C = a + b*Abs(CH) + c* Abs(CH3) + d * (Abs(CH3)/Abs(CH)) + e * (Abs(CH3)/Abs(CH))2 (Equation 2)
The constants a to f for equation 1 and a to e for equation 2 were determined by using least square regression analysis.
The CH3/1000C is converted to the ethylene content in wt.-% using following relationship: wt.-% (Ethylene in EP Copolymers) = 100 - CH3/1000TC * 0.3 (Equation 3)
Amount of Soluble Fraction (SF) and Crystalline Fraction (CF) are correlated through the XS calibration to the “Xylene Cold Soluble” (XCS) quantity and respectively Xylene Cold Insoluble (XCI) fractions, determined according to standard gravimetric method as per ISO16152. XS calibration is achieved by testing various EP copolymers with XS content in the range 2-31 wt.-%. A linear calibration curve is used.
Intrinsic viscosity (iV) of the parent EP copolymer and its soluble and crystalline fractions are determined with a use of an online 2-capillary viscometer and are correlated to corresponding IV’s determined by standard method in decalin according to ISO 1628-3. Calibration is achieved with various EP copolymers and PP polymers with iV = 2-4 dL/g. The determined calibration curve is linear.
The samples to be analyzed are weighed out in concentrations of 10 mg/ml to 20 mg/ml. After automated filling of the vial with 1 ,2,4-TCB containing 250 mg/l 2,6-tert-butyl-4- methylphenol (BHT) as antioxidant, the sample is dissolved at 170°C until complete dissolution is achieved with either constant stirring or gentle shaking. To avoid sample degradation, polymer solution is blanketed with the N2 atmosphere during dissolution. For PP composition containing inorganic fillers or pigments or any other non-TCB soluble polymeric substances removal of these is required. This can be done by hot filtration prior injection.
A defined volume of the polymer solution is injected into the column filled with inert support where the crystallization of the sample and separation of the soluble fraction from the crystalline fraction is taking place. This process is repeated two times. During the first injection the whole sample is measured at high temperature, determining the iV [dl/g] and the C2 [wt.-%] of the PP composition. During the second injection the soluble fraction (at low temperature) and the crystalline fraction (at high temperature) with the crystallization cycle are measured (wt.-% SF, wt.-% C2, iV). f) Intrinsic viscosity (iV)
The reduced viscosity (also known as viscosity number), qred, and intrinsic viscosity, iV, are determined according to ISO 1628-3: “Determination of the viscosity of polymers in dilute solution using capillary viscometers”.
Relative viscosities of a diluted polymer solution with concentration of 1 mg/ml and of the pure solvent (decahydronaphthalene stabilized with 200 ppm 2,6-bis(1 ,1 - dimethylethyl)-4-methylphenol) are determined in an automated capillary viscometer (Lauda PVS1) equipped with 4 Ubbelohde capillaries placed in a thermostatic bath filled with silicone oil. The bath temperature is maintained at 135 °C. The sample is dissolved with constant stirring until complete dissolution is achieved (typically within 90 min). The efflux time of the polymer solution as well as of the pure solvent are measured several times until three consecutive readings do not differ for more than 0.2s (standard deviation).
The relative viscosity of the polymer solution is determined as the ratio of averaged efflux times in seconds obtained for both, polymer solution and solvent: rei r ■■ ■ . q [dimensionless n ]
Reduced viscosity (qred) is calculated using the equation:
m where C is the polymer solution concentration at 135°C: C= — , p
and m is the polymer mass, V is the solvent volume, and y is the ratio of solvent densities at 20°C and 135°C (y=p2o/pi35=1.1 O7).
The calculation of intrinsic viscosity iV is performed by using the Schulz-Blaschke equation from the single concentration measurement:
where K is a coefficient depending on the polymer structure and concentration. For calculation of the approximate value for IV, K=0.27. g) Xylene cold solubles (XCS) content
The quantity of xylene soluble matter in polypropylene is detemined according to the ISO16152 (first edition; 2005-07-01).
A weighed amount of a sample is dissolved in hot xylene under reflux conditions at 135°C. The solution is then cooled down under controlled conditions and maintained at 25°C for 30 minutes to ensure controlled crystallization of the insoluble fraction. This insoluble fraction is then separated by filtration. Xylene is evaporated from the filtrate leaving the soluble fraction as a residue. The percentage of this fraction is determined gravi metrically.
where mo is the mass of the sample test portion weighed, in grams r is the mass of residue, in grams vo is the original volume of solvent taken vi is the volume of the aliquot taken for determination. h) Rheology:
Dynamic rheological measurements were carried out with Rheometrics RDA-II QC on compression molded samples under nitrogen atmosphere at 200°C using 25 mm - diameter plate and plate geometry. The oscillatory shear experiments were done within the linear viscoelastic range of strain at frequencies from 0.01 to 500 rad/s. (ISO6721-1)
The values of storage modulus (G ') , loss modulus (G”), complex modulus (G*) and complex viscosity (q*) were obtained as a function of frequency (co).
The Zero shear viscosity (qo) was calculated using complex fluidity defined as the reciprocal of complex viscosity. Its real and imaginary part are thus defined by f'(co) = q'( co)/ [q'( co)2 +q” (co)2 ] and f’ ( w)= q”( w)/ [q'( w)2 + q” (w)2 ] From the following equations q' = G” / co G7 co f'( co) = G” (co co)2 +G” (co)2 ] f’( co) = G' (co
co)2 +G” (co)2 ] i) Flexural Modulus
The flexural modulus was determined acc. to ISO 178 method A (3-point bending test) on 80 mm x 10 mm x 4 mm specimens. Following the standard, a test speed of 2 mm/min and a span length of 16 times the thickness was used. The testing temperature was 23±2° C. Injection moulding was carried out according to ISO 19069-2 using a melt temperature of 230°C for all materials irrespective of material melt flow rate. j) Charpy notched impact strength
The Charpy notched impact strength was determined acc. to ISO 179-1/1 eA on notched 80 mm x 10 mm x 4 mm specimens (specimens were prepared according to ISO 179- 1/1eA). Testing temperatures were 23±2° C or -20±2° C. Injection moulding was carried out acc. to ISO 19069-2 using a melt temperature of 230°C for all materials irrespective of material melt flow rate. k) Tensile strength and strain at break
Tensile strength and strain at break were determined on 5A injection moulding tensile specimens according to ISO 527-2. The tensile test was performed at +23°C with a speed of 25 mm/min as suggested by the IEC 60811-501 Edition 1.0 2012-03. The 5A injection moulding tensile specimens were aged for 10 days at 135 °C for the tensile strength after ageing and the strain at break after ageing.
I) Indentation in pressure test
The indentation properties at high temperature were determined on compression moulded plaques at 130°C for 1 h with a cylindric indentor according to method 1 pressure test in the CIGRE TB852 recommendations. m) Thermal conductivity
The thermal conductivities at 22°C and at 100°C were determined according to ISO 22007-2 on 20 mm x 20 mm x 3 mm compression moulded specimens.
2. Polyolefin composition
The following resins were used for the preparation of the polyolefin compositions of the examples:
Preparation of the heterophasic propylene copolymer HECO
• Catalysts
The catalyst used in the polymerization process for the heterophasic propylene copolymer powder was a Ziegler-Natta catalyst, which is described in the example section of WO 2015/117948. As co-catalyst triethyl-aluminium (TEAL) and as donor dicyclo pentyl dimethoxy silane (D-donor) was used.
• Polymerization of the heterophasic propylene copolymer powder
Heterophasic propylene copolymer powder was produced in a Borstar™ plant in the presence of the above described polymerization catalyst using one liquid-phase loop reactor and two gas phase reactors connected in series under conditions as shown in Table 1 . The first reaction zone was a loop reactor and the second and third reaction zones were gas phase reactors. The matrix phase was polymerized in the loop and first gas phase reactor and the elastomeric phase was polymerized in the second gas phase reactor. The catalyst as described above was fed into a prepolymerization reactor, which precedes the first reaction zone.
Table 1 : Polymerization conditions of the heterophasic propylene copolymer powder:
• Compounding conditions and properties of compounded HECO
The heterophasic propylene copolymer powder from the polymerization reaction was compounded in a twin screw extruder together with a stabilizer package to obtain the compounded HECO.
An overview of the compounding conditions of the HECO are shown in Table 2.
Table 2: Compounding of HECO in a twin screw extruder:
Stabiliser onepack 1 consists of 25.6 wt% Pentaerythrityl-tetrakis(3-(3’,5’-di-tert. butyl-4- hydroxyphenyl)-propionate (CAS-No. 6683-19-8), 51.3wt% Tris (2,4-di-f-butylphenyl) phosphite (CAS-No. 31570-04-4) and 23.1 wt% synthetic hydrotalcite (CAS-No. 11097- 59-9), all commercially available from a variety of companies.
The properties of HECO are listed in Table 3.
Preparation of polyolefin compositions
For the production of the polyolefin compositions of the inventive examples IE1 and IE2 and comparative example CE2, HECO was compounded with a LDPE in a
compounding step in a Coperion ZSK18 intermeshing, co-rotating twin screw 18 mm L/D 40.
Comparative example CE1 reflects the compounded HECO without additional compounding step.
An overview of polyolefin compositions CE1 , CE2, IE1 and IE2 are shown in Table 4.
The properties of CE1 , CE2, IE1 and IE2 are shown in Table 5.
• LDPE is a low density ethylene homopolymer produced in a commercial high pressure tubular reactor.
In general, high pressure radical initiated polymerization of ethylene can be effected in a tubular or an autoclave reactor at pressures in the range of 1200 to 3500 bars and a temperature in the range of 120 to 350 °C. Further details about high pressure radical polymerization is given in Encyclopedia of Polymer Science and Engineering, Vol 6 (1986), pp. 383-410 and in Encyclopedia of Materials: Science and Technology, Elsevier Science Ltd.: 'Polyethylene: High-pressure', R. Klimesch, D. Littmann and F.-O. Mahling, pp. 7181-7184 (2001), which are herewith incorporated as reference.
The LDPE has a density of 923 kg/m3 a melt flow rate MFR2 (190°C, 2.16 kg) of 0.75 g/10 min, a melting temperature Tm of 112°C, Vicat A50 softening temperature of 95°C and a complex viscosity eta*ioo rad/s of 682 Pa s.
The LDPE has been chosen because of its complex viscosity eta*ioo rad/s of 682 Pa s. The HECO has a complex viscosity eta*wo rad/s of 760 Pa s so that the difference between eta*wo rad/s of the HECO and eta*wo rad/s of the LDPE eta*wo rad/s (HECO) - eta*wo rad/s (LDPE) is 10.3 % of eta*wo rad/s (HECO).
Properties of the polyolefin compositions
The results, calculated as median of at least 5 and 3 measurements for tensile and pressure tests, respectively, are reported in Table 5.
As can be observed, CE1 shows high tensile strength and no indentation, but the strain to break after ageing is below the accepted limit of 350%.
When adding 65 wt.-% of LDPE (CE2), the strain to break before ageing is too low and the indentation is 100 %. For IE1 and IE2 with 50 wt.-% and 35 wt.-% of LDPE, respectively, the tensile strength and the indentation are still fulfilling the indentation requirements, while at the same time the strain to break before ageing is higher than 350 % and it even increases after ageing.
Claims
1. A polyolefin composition having a total ethylene content of from more than 32.5 to 60.0 wt.-%, preferably from 33.0 to 55.0 wt.-%, more preferably from 33.5 to 52.5 wt.-%, still more preferably from 34.0 to 50.0 wt.-%, based on the total weight of the polyolefin composition and determined by FT-IR spectroscopy calibrated by quantitative 13C-NMR spectroscopy from crystallization extraction (CRYSTEX); a melt flow rate MFR2 of from more than 2.5 to 6.0 g/10 min, preferably from 2.6 to 5.5 g/10 min, still more preferably from 2.7 to 5.0 g/10 min and most preferably from 2.8 to 4.5 g/10 min, determined according to ISO 1133 at 230°C and 2.16 kg; a crystalline fraction (CF) content, determined by crystallization extraction (CRYSTEX), in the range from 75.0 to 95.0 wt.-%, preferably from 77.5 to 93.5 wt.- %, more preferably from 80.0 to 92.5 wt.-% and most preferably from 82.5 to 91 .0 wt.-%, based on the total weight amount of the polyolefin composition, said crystalline fraction (CF) having an ethylene content (C2(CF)), determined by FT-IR spectroscopy calibrated by quantitative 13C-NMR spectroscopy, in the range from more than 32.5 to 62.5 wt.-%, preferably from 35.0 to 60.0 wt.-% and most preferably from 36.0 to 55.0 wt.-%, based on the total amount of monomer units in the crystalline fraction (CF), and a soluble fraction (SF) content, determined by crystallization extraction (CRYSTEX), in the range from 5.0 to 25.0 wt.-%, preferably from 6.5 to 22.5 wt.-%, more preferably from 7.5 to 20.0 wt.-% and most preferably from 9.0 to 17.5 wt.-%, based on the total weight amount of the polyolefin composition, said soluble fraction (SF) having an intrinsic viscosity (iV(SF)), determined according to ISO 1628-3, in the range from 85 to 200 cm3/g, more preferably from 95 to 175 cm3/g and most preferably from 100 to 150 cm3/g.
2. The polyolefin composition according to claim 1 , wherein the crystalline fraction has an intrinsic viscosity (iV(CF)), determined according to ISO 1628-3, in the range from 100 to 250 cm3/g, more preferably from 125 to 225 cm3/g and most preferably from 140 to 210 cm3/g; and/or a propylene content (C3(CF)), determined by FT-IR
spectroscopy calibrated by quantitative 13C-NMR spectroscopy, in the range from 37.5 to less than 67.5 wt.-%, preferably from 40.0 to 65.0 wt.-% and most preferably from 45.0 to 64.0 wt.-%, based on the total amount of monomer units in the crystalline fraction (CF); and/or the soluble fraction (SF) has an ethylene content (C2(SF)), determined by FT-IR spectroscopy calibrated by quantitative 13C-NMR spectroscopy, in the range from 22.5 to 42.5 wt.-%, preferably from 25.0 to 40.0 wt.- % and most preferably from 30.0 to 35.0 wt.-%, and/or a propylene content (C3(SF)), determined by FT-IR spectroscopy calibrated by quantitative 13C-NMR spectroscopy, in the range from 57.5 to 77.5 wt.-%, preferably from 60.0 to 75.0 wt.- % and most preferably from 62.5 to 70.0 wt.-%, both based on the total amount of monomer units in the soluble fraction (SF).
3. The polyolefin composition according to claims 1 or 2 having a ratio of the ethylene content of the soluble fraction to the ethylene content of the crystalline fraction C2(SF)/C2(CF) of from 0.50 : 1.00 to 1.00 : 1.00, more preferably from 0.55 : 1.00 to 0.95 : 1 .00, most preferably from 0.60 : 1 .00 to 0.90 : 1 .00, and/or ratio of the intrinsic viscosity of the soluble fraction to the intrinsic viscosity of the crystalline fraction iV(SF)/iV(CF) of from 0.40 : 1.00 to 0.95 : 1.00, more preferably from 0.50 : 1 .00 to 0.85 : 1 .00, most preferably from 0.55 : 1 .00 to 0.75 : 1 .00.
4. The polyolefin composition according to any one claims 1 to 3 comprising a total propylene content of from 40.0 to less than 67.5 wt.-%, preferably from 45.0 to 67.0 wt.-%, more preferably from 47.5 to 66.5 wt.-%, still more preferably from 49.0 to 66.0 wt.-%, based on the total weight of the polyolefin composition and determined by FT-IR spectroscopy calibrated by quantitative 13C-NMR spectroscopy from crystallization extraction (CRYSTEX).
5. The polyolefin composition according to any one of claims 1 to 4 having all of the following properties:
• a tensile strength of from 8.5 to 50 MPa, preferably from 15 to 45 MPa, more preferably from 20 to 40 MPa, determined according to ISO 527-2 on 5A
injection moulded test specimens at 23°C and a speed of 25 mm/min before ageing;
• a tensile strength of from 8.5 to 50 MPa, preferably from 15 to 45 MPa, more preferably from 20 to 40 MPa, determined according to ISO 527-2 on 5A injection moulded test specimens at 23°C and a speed of 25 mm/min after ageing for 10 days at 135°C;
• a strain at break of from 350 to 650 %, preferably from 360 to 600 %, more preferably from 375 to 575 %, determined according to ISO 527-2 on 5A injection moulded test specimens at 23°C and a speed of 25 mm/min before ageing;
• a strain at break of from 350 to 650 %, preferably from 360 to 600 %, more preferably from 375 to 575 %, determined according to ISO 527-2 on 5A injection moulded test specimens at 23°C and a speed of 25 mm/min after ageing for 10 days at 135°C; and
• an indentation in the pressure test of from 0 to 50 %, preferably of from 0 to 45 %, determined on compression moulded plaques with a cylindric indentor according to method 1 pressure test in the CIGRE TB852 recommendations at 130°C for 1 h.
6. The polyolefin composition according to any one of claims 1 to 5 comprising from 40.0 to less than 70.0 wt.-%, preferably from 45.0 to 69.0 wt.-%, more preferably from 47.5 to 67.5 wt.-% of a copolymer of propylene and ethylene and from more than 30.0 to 60.0 wt.-%, preferably from 31 .0 to 55.0 wt.-%, more preferably from 32.5 to 52.5 wt.-% of a low density polyethylene, wherein all weight amounts are based on the total weight of the polyolefin composition.
7. The polyolefin composition according to claim 6, wherein the copolymer of propylene and ethylene has one or more or all of the following properties:
• a total ethylene content of from 4.0 to 11 .0 wt.-%, preferably from 5.0 to 10.0 wt.- %, more preferably from 5.5 to 9.5 wt.-%, still more preferably from 6.0 to 9.0 wt.-%, based on the total weight of the copolymer of propylene and ethylene and
determined by FT-IR spectroscopy calibrated by quantitative 13C-NMR spectroscopy from crystallization extraction (CRYSTEX);
• a crystalline fraction (CF) content, determined by crystallization extraction (CRYSTEX), in the range from 75.0 to 90.0 wt.-%, preferably from 77.0 to 88.0 wt.-%, more preferably from 78.5 to 86.5 wt.-% and most preferably from 80.0 to 85.0 wt.-%, based on the total weight amount of the polyolefin composition,
• said crystalline fraction (CF) having an ethylene content (C2(CF)), determined by FT-IR spectroscopy calibrated by quantitative 13C-NMR spectroscopy, in the range from 1.0 to 15.0 wt.-%, preferably from 2.0 to 10.0 wt.-% and most preferably from 3.0 to 8.5 wt.-%, based on the total amount of monomer units in the crystalline fraction (CF), and
• an intrinsic viscosity (iV(CF)), determined according to ISO 1628-3, in the range from 185 to 325 cm3/g, more preferably from 200 to 300 cm3/g and most preferably from 225 to 270 cm3/g; and
• a soluble fraction (SF) content, determined by crystallization extraction (CRYSTEX), in the range from 10.0 to 25.0 wt.-%, preferably from 12.0 to 23.0 wt.-%, more preferably from 13.5 to 21 .5 wt.-% and most preferably from 15.0 to 20.0 wt.-%, based on the total weight amount of the copolymer of propylene and ethylene,
• said soluble fraction (SF) having an ethylene content (C2(SF)), determined by FT-IR spectroscopy calibrated by quantitative 13C-NMR spectroscopy, in the range from 20.0 to 35.0 wt.-%, preferably from 21 .5 to 32.5 wt.-% and most preferably from 23.0 to 30.0 wt.-%, based on the total amount of monomer units in the soluble fraction (SF), and
• an intrinsic viscosity (iV(SF)), determined according to ISO 1628-3, in the range from 90 to 190 cm3/g, more preferably from 100 to 175 cm3/g and most preferably from 115 to 150 cm3/g;
• a melt flow rate MFR2 of from more than 2.5 to 6.0 g/10 min, preferably from 2.8 to 5.5 g/10 min, still more preferably from 3.0 to 5.0 g/10 min and most preferably from 3.3 to 4.5 g/10 min, determined according to ISO 1133 at 230°C and 2.16 kg; and/or
• a flexural modulus of from more than 470 MPa to 750 MPa, more preferably of from 475 MPa to 700 MPa and most preferably of from 500 MPa to 650 MPa, determined according to ISO 178 method A; and/or
• a Charpy notched impact strength at 23°C of from 5.0 to 30.0 kJ/m2, more preferably from 7.0 to 25.0 kJ/m2 and most preferably from 9.0 to 20.0 kJ/m2, determined according to ISO 179-1/1 eA; and/or
• a Charpy notched impact strength at -20°C of from 0.8 to 5.0 kJ/m2, more preferably from 0.8 to 4.0 kJ/m2 and most preferably from 0.8 to 3.5 kJ/m2, determined according to ISO 179-1/1 eA; and/or
• a melting temperature Tm of from 132 to 155°C, preferably from 135 to 150°C and most preferably from 137 to 147°C, determined by to DSC analysis according to ISO 11357 I part 3 /method C2; and/or
• a crystallization temperature Tc of from 85 to 115°C, preferably from 90 to 110°C and most preferably from 95 to 107°C, determined by to DSC analysis according to ISO 113571 part 3 /method C2; and/or
• a difference of the melting temperature to the crystallization temperature Tm-Tc in the range of from 35 to 55°C, preferably 37 to 52°C and most preferably from 39 to 50°C; and/or
• a complex viscosity at a frequency of 100 rad/s eta*wo rad/s of from 650 to 900 Pa s, more preferably from 700 to 850 Pa s, most preferably from 725 to 800 Pa s.
8. The polyolefin composition according to claim 6 or 7, wherein the copolymer of propylene and ethylene is not subjected to vis-breaking.
9. The polyolefin composition according to any one of claims 6 to 8, wherein the low density polyethylene is polymerized in a tubular high pressure polymerization process and has one or more or all of the following properties:
• a melt flow rate MFR2 of from 0.1 to 2.5 g/10 min, preferably from 0.2 to 2.3 g/10 min, still more preferably from 0.3 to 2.0 g/10 min and most preferably from 0.5 to 1.5 g/10 min, determined according to ISO 1133 at 190°C and 2.16 kg; and/or
• a density of from 915 to 932 kg/m3, preferably from 917 to 930 kg/m3, more preferably from 918 to 928 kg/m3, determined according to ISO 1183; and/or
• a melting temperature Tm of from 105 to 125°C, preferably from 107 to 120°C, more preferably from 109 to 117°C, determined by to DSC analysis according to ISO 113571 part 3 /method C2; and/or
• a Vicat A50 softening temperature of from 85 to 105°C, preferably from 87 to 102°C, more preferably from 90 to 100°C, determined according to ISO 306 at 10 N; and/or
• a complex viscosity at a frequency of 100 rad/s eta*wo rad/s of from 600 to 800 Pa s, more preferably from 625 to 775 Pa s, most preferably from 650 to 750 Pa s.
10. The polyolefin composition according to any one of claims 6 to 9, wherein the difference between the complex viscosity at a frequency of 100 rad/s of the copolymer of propylene (eta*wo rad/s (PP)) and the complex viscosity at a frequency of 100 rad/s of the low density polyethylene (eta* o rad/s (LDPE)) | eta*wo rad/s (PP) - eta*wo rad/s (LDPE)| is not more than ± 20 % of eta* o rad/s (PP), preferably not more than ± 15 % of eta*wo rad/s (PP).
11 . The polyolefin composition according to any one of claims 6 to 10 further comprising from 0 to 5000 ppm, preferably from 0 to 4000 ppm of an alphanucleating agent, preferably a soluble or polymeric alpha-nucleating agent, more preferably from a soluble alpha-nucleating agent selected from dibenzylidenesorbitol (e.g. 1 ,3 : 2,4 dibenzylidenesorbitol) and Ci-Cs-alkyl- substituted dibenzylidenesorbitol derivatives, such as methyldibenzylidenesorbitol, ethyldibenzylidenesorbitol or dimethyldibenzylidenesorbitol (e.g. 1 ,3 : 2,4 di(methylbenzylidene) sorbitol), or substituted nonitol-derivatives, such as 1 ,2,3- trideoxy-4,6:5,7-bis-0-[(4-propylphenyl)methylene]-nonitol and mixtures thereof.
12. The polyolefin composition according to any one of claims 1 to 11 being free of a dielectric fluid.
13. An article comprising the polyolefin composition according to any one of the preceding claims.
14. The article according to claim 13 being a cable, preferably a medium voltage cable or high voltage cable, more preferably a high voltage cable, even more preferably a high voltage DC (HVDC) cable, comprising an insulation layer comprising the polyolefin composition, preferably wherein the insulation layer comprises from 90 to 100 wt.-%, preferably from 95 to 100 wt.-%, still more preferably from 99 to 100 wt.- %, based on the total weight of the insulation layer, most preferably consists of the polyolefin composition..
15. The article according to claim 14 meeting all CIGRE TB852 recommendations for HVDC cable systems.
16. The use of a polyolefin composition according to any one of claims 1 to 12 as cable insulation for medium voltage cables, high voltage cables, extra high voltage cable and ultra high voltage cables, preferably for high voltage cables and extra high voltage cables, more preferably high voltage DC (HVDC) cables and extra high voltage DC (EHVDC) cables.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP23200624 | 2023-09-28 | ||
| EP23200624.7 | 2023-09-28 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2025067703A1 true WO2025067703A1 (en) | 2025-04-03 |
Family
ID=88236513
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2024/058705 Pending WO2025067703A1 (en) | 2023-09-28 | 2024-03-28 | Polyolefin composition for cable insulation |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2025067703A1 (en) |
Citations (13)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1992012182A1 (en) | 1990-12-28 | 1992-07-23 | Neste Oy | Multi-stage process for producing polyethylene |
| EP0887379A1 (en) | 1997-06-24 | 1998-12-30 | Borealis A/S | Process and apparatus for preparing propylene homopolymers and copolymers |
| WO1999024479A1 (en) | 1997-11-07 | 1999-05-20 | Borealis A/S | Novel propylene polymers and products thereof |
| WO1999024478A1 (en) | 1997-11-07 | 1999-05-20 | Borealis A/S | Process for preparing polypropylene |
| WO2000068315A1 (en) | 1999-05-07 | 2000-11-16 | Borealis Technology Oy | High-stiffness propylene polymers and a process for the preparation thereof |
| WO2004000899A1 (en) | 2002-06-25 | 2003-12-31 | Borealis Technology Oy | Polyolefin with improved scratch resistance and process for producing the same |
| WO2004111095A1 (en) | 2003-06-06 | 2004-12-23 | Borealis Technology Oy | Process for the catalytic polymerization of olefins, a reactor system, its use in the process, the polyolefins obtained and their use |
| WO2013092620A1 (en) | 2011-12-23 | 2013-06-27 | Borealis Ag | Propylene copolymer for injection molded articles or films |
| EP2739679A1 (en) | 2011-08-04 | 2014-06-11 | Prysmian S.p.A. | Energy cable having a thermoplastic electrically insulating layer |
| WO2015117948A1 (en) | 2014-02-06 | 2015-08-13 | Borealis Ag | Soft and transparent impact copolymers |
| WO2022200396A1 (en) | 2021-03-25 | 2022-09-29 | Borealis Ag | Polypropylene composition for cable insulation |
| WO2022200395A1 (en) | 2021-03-25 | 2022-09-29 | Borealis Ag | Polypropylene composition for cable insulation |
| WO2023099448A1 (en) * | 2021-12-01 | 2023-06-08 | Borealis Ag | Polypropylene composition suitable for packaging applications |
-
2024
- 2024-03-28 WO PCT/EP2024/058705 patent/WO2025067703A1/en active Pending
Patent Citations (13)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1992012182A1 (en) | 1990-12-28 | 1992-07-23 | Neste Oy | Multi-stage process for producing polyethylene |
| EP0887379A1 (en) | 1997-06-24 | 1998-12-30 | Borealis A/S | Process and apparatus for preparing propylene homopolymers and copolymers |
| WO1999024479A1 (en) | 1997-11-07 | 1999-05-20 | Borealis A/S | Novel propylene polymers and products thereof |
| WO1999024478A1 (en) | 1997-11-07 | 1999-05-20 | Borealis A/S | Process for preparing polypropylene |
| WO2000068315A1 (en) | 1999-05-07 | 2000-11-16 | Borealis Technology Oy | High-stiffness propylene polymers and a process for the preparation thereof |
| WO2004000899A1 (en) | 2002-06-25 | 2003-12-31 | Borealis Technology Oy | Polyolefin with improved scratch resistance and process for producing the same |
| WO2004111095A1 (en) | 2003-06-06 | 2004-12-23 | Borealis Technology Oy | Process for the catalytic polymerization of olefins, a reactor system, its use in the process, the polyolefins obtained and their use |
| EP2739679A1 (en) | 2011-08-04 | 2014-06-11 | Prysmian S.p.A. | Energy cable having a thermoplastic electrically insulating layer |
| WO2013092620A1 (en) | 2011-12-23 | 2013-06-27 | Borealis Ag | Propylene copolymer for injection molded articles or films |
| WO2015117948A1 (en) | 2014-02-06 | 2015-08-13 | Borealis Ag | Soft and transparent impact copolymers |
| WO2022200396A1 (en) | 2021-03-25 | 2022-09-29 | Borealis Ag | Polypropylene composition for cable insulation |
| WO2022200395A1 (en) | 2021-03-25 | 2022-09-29 | Borealis Ag | Polypropylene composition for cable insulation |
| WO2023099448A1 (en) * | 2021-12-01 | 2023-06-08 | Borealis Ag | Polypropylene composition suitable for packaging applications |
Non-Patent Citations (13)
| Title |
|---|
| BUSICO, V.CARBONNIERE, P.CIPULLO, R.PELLECCHIA, R.SEVERN, J.TALARICO, G.MACROMOL., RAPID COMMUN., vol. 28, 2007, pages 1128 |
| BUSICO, V.CIPULLO, R., MONACOG., VACATELLO, M.SEGRE, A.L., MACROMOLECULES, vol. 30, 1997, pages 6251 |
| BUSICO, V.CIPULLO, R., PROG. POLYM. SCI., vol. 26, no. 128-37-0, 2001, pages 443 |
| CHENG, H. N., MACROMOLECULES, vol. 17, 1950, pages 1984 |
| ENCYCLOPEDIA OF POLYMER SCIENCE AND ENGINEERING, vol. 6, 1986, pages 383 - 410 |
| KAKUGO, M.NAITO, Y.MIZUNUMA, K.MIYATAKE, T., MACROMOLECULES, vol. 15, 1982, pages 1150 |
| LJILJANA JEREMICANDREAS ALBRECHTMARTINA SANDHOLZERMARKUS GAHLEITNER: "Rapid characterization of high-impact ethylene-propylene copolymer composition by crystallization extraction separation: comparability to standard separation methods", INTERNATIONAL JOURNAL OF POLYMER ANALYSIS AND CHARACTERIZATION, vol. 25, no. 8, 2020, pages 581 - 596 |
| R. KLIMESCHD. LITTMANNF.-O. MAHLING: "Encyclopedia of Materials: Science and Technology", 2001, ELSEVIER SCIENCE LTD., article "Polyethylene: High-pressure", pages: 7181 - 7184 |
| RANDALL, J., MACROMOL. SCI., REV. MACROMOL. CHEM. PHYS., vol. C29, 1989, pages 201 |
| RESCONI, L.CAVALLO, L.FAIT, A.PIEMONTESI, F., CHEM. REV., vol. 100, 2000, pages 1253 |
| SINGH, G.KOTHARI, A.GUPTA, V., POLYMER TESTING, vol. 28, no. 5, 2009, pages 1141 - 1190 |
| WANGW-J.ZHU, S., MACROMOLECULES, vol. 33, 2000, pages 1157 |
| ZHOU, Z.KUEMMERLE, R.QIU, X.REDWINE, D.CONG, R.TAHA, A.BAUGH, DWINNIFORD, B., J. MAG. RESON., vol. 187, 2007, pages 225 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP4314152B1 (en) | Polypropylene composition for cable insulation | |
| US20240158623A1 (en) | Polypropylene compostion for cable insulation | |
| WO2024068577A1 (en) | Polypropylene composition for cable insulation | |
| WO2024068580A1 (en) | Polypropylene composition for cable insulation | |
| US20240002647A1 (en) | Semiconductive polypropylene composition | |
| WO2025068332A1 (en) | Cable comprising polypropylene composition | |
| WO2025068333A1 (en) | Semiconductive polypropylene composition | |
| WO2025067703A1 (en) | Polyolefin composition for cable insulation | |
| WO2025067702A1 (en) | Polyolefin composition for cable insulation | |
| WO2026008736A1 (en) | Propylene copolymer for cable insulation | |
| WO2026008730A1 (en) | Polymer composition for cable insulation | |
| WO2026008734A1 (en) | Polymer composition for cable insulation | |
| WO2026008735A1 (en) | Polymer composition for cable insulation | |
| WO2024068579A1 (en) | Polypropylene composition for cable insulation | |
| WO2024068576A1 (en) | Polypropylene composition for cable insulation | |
| RU2837167C1 (en) | Polypropylene composition for cable insulation | |
| WO2026008731A1 (en) | Polymer composition for cable insulation | |
| RU2837167C9 (en) | Polypropylene composition for cable insulation | |
| WO2025202325A1 (en) | Polypropylene composition for cable insulation | |
| WO2026027750A1 (en) | Polypropylene composition for fiber applications | |
| WO2024068578A1 (en) | Polypropylene composition for cable insulation | |
| EP4606826A1 (en) | Heterophasic polypropylene compositions with high c2 content | |
| WO2025176814A1 (en) | Polypropylene composition with improved low temperature impact performance |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 24716731 Country of ref document: EP Kind code of ref document: A1 |