WO2025064982A1 - Platform of hybrid advanced molecular manufacturing regulator and applications thereof - Google Patents
Platform of hybrid advanced molecular manufacturing regulator and applications thereof Download PDFInfo
- Publication number
- WO2025064982A1 WO2025064982A1 PCT/US2024/047951 US2024047951W WO2025064982A1 WO 2025064982 A1 WO2025064982 A1 WO 2025064982A1 US 2024047951 W US2024047951 W US 2024047951W WO 2025064982 A1 WO2025064982 A1 WO 2025064982A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- platform according
- present
- cells
- certain embodiments
- cell
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B5/00—Measuring for diagnostic purposes; Identification of persons
- A61B5/48—Other medical applications
- A61B5/4836—Diagnosis combined with treatment in closed-loop systems or methods
- A61B5/4839—Diagnosis combined with treatment in closed-loop systems or methods combined with drug delivery
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B5/00—Measuring for diagnostic purposes; Identification of persons
- A61B5/68—Arrangements of detecting, measuring or recording means, e.g. sensors, in relation to patient
- A61B5/6846—Arrangements of detecting, measuring or recording means, e.g. sensors, in relation to patient specially adapted to be brought in contact with an internal body part, i.e. invasive
- A61B5/6847—Arrangements of detecting, measuring or recording means, e.g. sensors, in relation to patient specially adapted to be brought in contact with an internal body part, i.e. invasive mounted on an invasive device
- A61B5/686—Permanently implanted devices, e.g. pacemakers, other stimulators, biochips
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M31/00—Devices for introducing or retaining media, e.g. remedies, in cavities of the body
- A61M31/002—Devices for releasing a drug at a continuous and controlled rate for a prolonged period of time
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61N—ELECTROTHERAPY; MAGNETOTHERAPY; RADIATION THERAPY; ULTRASOUND THERAPY
- A61N1/00—Electrotherapy; Circuits therefor
- A61N1/18—Applying electric currents by contact electrodes
- A61N1/32—Applying electric currents by contact electrodes alternating or intermittent currents
- A61N1/36—Applying electric currents by contact electrodes alternating or intermittent currents for stimulation
- A61N1/36002—Cancer treatment, e.g. tumour
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B5/00—Measuring for diagnostic purposes; Identification of persons
- A61B5/145—Measuring characteristics of blood in vivo, e.g. gas concentration or pH-value ; Measuring characteristics of body fluids or tissues, e.g. interstitial fluid or cerebral tissue
- A61B5/14546—Measuring characteristics of blood in vivo, e.g. gas concentration or pH-value ; Measuring characteristics of body fluids or tissues, e.g. interstitial fluid or cerebral tissue for measuring analytes not otherwise provided for, e.g. ions, cytochromes
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B5/00—Measuring for diagnostic purposes; Identification of persons
- A61B5/145—Measuring characteristics of blood in vivo, e.g. gas concentration or pH-value ; Measuring characteristics of body fluids or tissues, e.g. interstitial fluid or cerebral tissue
- A61B5/1468—Measuring characteristics of blood in vivo, e.g. gas concentration or pH-value ; Measuring characteristics of body fluids or tissues, e.g. interstitial fluid or cerebral tissue using chemical or electrochemical methods, e.g. by polarographic means
- A61B5/1473—Measuring characteristics of blood in vivo, e.g. gas concentration or pH-value ; Measuring characteristics of body fluids or tissues, e.g. interstitial fluid or cerebral tissue using chemical or electrochemical methods, e.g. by polarographic means invasive, e.g. introduced into the body by a catheter
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2/00—Filters implantable into blood vessels; Prostheses, i.e. artificial substitutes or replacements for parts of the body; Appliances for connecting them with the body; Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/02—Prostheses implantable into the body
- A61F2/022—Artificial gland structures using bioreactors
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2/00—Filters implantable into blood vessels; Prostheses, i.e. artificial substitutes or replacements for parts of the body; Appliances for connecting them with the body; Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/82—Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/86—Stents in a form characterised by the wire-like elements; Stents in the form characterised by a net-like or mesh-like structure
- A61F2/90—Stents in a form characterised by the wire-like elements; Stents in the form characterised by a net-like or mesh-like structure characterised by a net-like or mesh-like structure
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2250/00—Special features of prostheses classified in groups A61F2/00 - A61F2/26 or A61F2/82 or A61F9/00 or A61F11/00 or subgroups thereof
- A61F2250/0058—Additional features; Implant or prostheses properties not otherwise provided for
- A61F2250/0067—Means for introducing or releasing pharmaceutical products into the body
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M37/00—Other apparatus for introducing media into the body; Percutany, i.e. introducing medicines into the body by diffusion through the skin
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B33—ADDITIVE MANUFACTURING TECHNOLOGY
- B33Y—ADDITIVE MANUFACTURING, i.e. MANUFACTURING OF THREE-DIMENSIONAL [3-D] OBJECTS BY ADDITIVE DEPOSITION, ADDITIVE AGGLOMERATION OR ADDITIVE LAYERING, e.g. BY 3-D PRINTING, STEREOLITHOGRAPHY OR SELECTIVE LASER SINTERING
- B33Y80/00—Products made by additive manufacturing
Definitions
- the present disclosure relates generally to the field of biomedical engineering, and more particularly to a platform for a hybrid advanced molecular manufacturing regulator in wireless communication with an external device, and applications of the same.
- Cancer immunotherapy represents perhaps the most important breakthrough in the field of oncology in the past half century, exhibiting the ability to induce durable responses even in patients with advanced-stage malignancies.
- These therapies fall into two categories: cell therapies and protein-based immunostimulatory agents.
- Cell therapies encompass approaches including adoptive transfer of ex vivo expanded autologous tumor- infiltrating lymphocytes (TILs), dendritic cell vaccines, and a range of engineered immune receptor lymphocytes such as transgenic TCR T cells, chimeric antigen receptor (CAR) T cells, and CAR-NK cells. While these approaches have yielded robust cures in a subset of hematological malignancies, they have yet to demonstrate durable responses in any solid organ malignancy.
- TILs tumor- infiltrating lymphocytes
- CAR chimeric antigen receptor
- Protein-based immunotherapies include immune checkpoint blockade (ICB) antibodies, vaccine, or administration of natural or engineered immunostimulatory cytokines. While ICB represents the greatest advance in solid tumors in decades, eliciting cures in a subset of patients with stage IV advanced cancers, most patients do not experience durable responses and many cancers, including most peritoneal malignancies, are unresponsive to ICB. Treatment with exogenous cytokines, such as high-dose IL-244 and IL-12, were among the first approaches in the field and elicited responses in a small subset of melanoma and renal cell carcinoma patients.
- cytokines such as high-dose IL-244 and IL-12
- the communication module comprises at least one antenna.
- the at least one antenna comprises a magnetoelectric (ME) component.
- ME magnetoelectric
- the ME component comprises at least one magnetostrictive layer attached to a piezoelectric layer.
- the at least one magnetostrictive layer comprises multiple magnetostrictive layers.
- the at least one antenna is in wireless communication with the at least one transmission coil.
- the at least one antenna is configured to receive a power transmission from the at least one transmission coil.
- the implantable device comprises a battery, wherein the battery is charged by the power transmission.
- the at least one antenna is configured to receive a download information transmission from the at least one transmission coil.
- the at least one transmission coil is configured to receive an upload information transmission from the at least one antenna.
- the at least one antenna comprises multiple sub-antennas.
- the at least one transmission coil comprises a power transmitter coil and a receiver pick-up coil.
- the at least one antenna is configured to receive a power transmission from the power transmitter coil.
- the receiver pick-up coil is configured to receive an upload information transmission from the at least one antenna, and the at least one antenna is configured to receive a download information transmission from the receiver pick-up coil.
- the molecular manufacturing regulator comprises a conductive scaffold in communication with a control module; a biochemical sensor module in communication with the control module; an oxygenator configured to produce oxygen and disposed inside the conductive scaffold; and at least one engineered cells encapsulation disposed in association with the conductive scaffold; wherein the at least one engineered cells encapsulation encapsulates engineered cells; wherein the engineered cells are configured to produce at least one cancer therapeutic agent.
- the conductive scaffold is configured to produce an electrical pulse, wherein the conductive scaffold is powered by the battery.
- control module comprises a machine learning algorithm for optimizing at least one of the power transmission, the upload information transmission, and the download information transmission.
- the implantable device comprises at least one of a field- programmable gate array (FPGA) circuit, an application specific integrated circuit (ASIC), and a microcontroller circuit; wherein the at least one of the FPGA, the ASIC, and the microcontroller circuit coordinates and controls an electrical stimulation of the conductive scaffold which is optimized for a production efficiency of the at least one cancer therapeutic agent and/or a longevity of battery.
- FPGA field- programmable gate array
- ASIC application specific integrated circuit
- microcontroller circuit coordinates and controls an electrical stimulation of the conductive scaffold which is optimized for a production efficiency of the at least one cancer therapeutic agent and/or a longevity of battery.
- the implantable device is powered by a primary cell battery.
- the primary cell battery is a lithium-based battery configured to provide a long-term power to the implantable device.
- the communication module of the implantable device comprises a Bluetooth communication module for wireless transmission to the external device.
- the communication module is configured to transmit and receive data related to an operational status of the molecular manufacturing regulator.
- the communication module of the implantable device incorporates a proprietary radio protocol for wireless communication with the external device.
- the proprietary radio protocol is configured to transmit and receive data related to the operational status of the molecular manufacturing regulator.
- the proprietary radio protocol is optimized for low-power operation to extend a life of a primary cell battery powering the implantable device.
- a primary cell battery is configured to power both the molecular manufacturing regulator and the communication module.
- Fig. 1 illustrates a Hybrid Advanced Molecular Manufacturing Regulator (HAMMR), related components and its application.
- HAMMR Hybrid Advanced Molecular Manufacturing Regulator
- Fig. 2 illustrates implementation and feedback of the HAMMR which is implanted in the intraperitoneal space through a small incision.
- Fig. 3 illustrates a list of the target therapeutics for the HAMMR.
- Fig. 4 illustrates light induced cytokine secretion from engineered ARPE-19 cells according to one embodiment of the present invention.
- Fig. 5A illustrates a design and function of engineered cells, in particular, the electrogenic processes and device.
- Fig. 5B illustrates the design and testing of various construct designs of the engineered cells.
- Fig. 6 illustrates an embodiment of printing conducting polymer composites.
- Fig. 7 illustrates microscopic images showing immunoisolating chamber and chemical coatings to protect against foreign body responses.
- Fig. 8 illustrates that ecCh arrays using sputtered IrCh (SIROF) catalyst are able to support the bioactivities of high- cell density capsules (60k cell mm-3) up to 21 days under hypoxic environment (1 % O2) as well as implanted in rodent for 10 days.
- SIROF sputtered IrCh
- FIG. 9 illustrates oxygenators of HAMMR and its application.
- Fig. 10 illustrates a schematic diagram of general model structure.
- Fig. 11 illustrates electrode engineering and performance of nano- structured electrode.
- Fig. 12 illustrates effect of bias stress on sensor stability.
- Fig. 13 illustrates a dual mode onboard NN engine for heart anomaly detection (top), and ultra small ME implant receiver.
- Fig. 14 illustrates models of tumor micro-environment and is to assist the design and control.
- Fig. 15 illustrates promoter construct designs for engineered Electro-cells to enable production of >1 pg/cell/day immunomodulator in vitro.
- Fig. 16 illustrates a chart reflecting hIL2 production of Electro- ARPE- 19 cells engineered with promoter construct designs stimulated with 80 mM KC1.
- Fig. 17A illustrates an optimized construct design for engineered Electro-cells.
- Fig. 17B illustrates charts reflecting 24-hr hIL2 production after KC1 stimulation and 24-hr hAnti-PDl production after KC1 stimulation by the Electro-ARPE-19 cells.
- Fig. 18A illustrates a workflow of in vitro activation of Electro-cells with selected scaffold material at >1 pg/cell/day.
- Fig. 18B illustrates a chart reflecting 24-hr hIL2 production by Electro-ARPE-19 cells after AC stimulation.
- Fig. 18C illustrates a chart reflecting 24-hr live cell counts (LCC) of Electro -ARPE- 19 cells after AC stimulation.
- Fig. 18D illustrates a chart reflecting 24-hr % viability of Electro -ARPE-19 cells after AC stimulation.
- Fig. 19A illustrates a down- sei ection of conducting scaffold material considering performance, stability, cell viability, and scalability.
- Fig. 19B illustrates chemical formulas of PDL, Fibronectin, Laminin, and RGD.
- Fig. 20 illustrates a selection of conducting scaffold material considering cell viability, particularly, PEDOT:PSS, Graphene, Platnium.
- Fig. 21 illustrates a selection of conducting scaffold material considering performance, particularly, IL-2 and H2O2 production on PETOD:PSS.
- Fig. 22 illustrates a selection of conducting scaffold material considering performance, stability, cell viability, and scalability, particularly, IL-2 and H2O2 production on Pt.
- Fig. 23 illustrates a photo of the conducting scaffold using the selected materials (Pt) and confocal images reflecting the cells growing on the conducting scaffold.
- Fig. 25 illustrates a schematic diagram of conducting scaffold with the oxygenator.
- Fig. 26 illustrates a diagram structure of three engineered IL-2s (upper panel) and a
- Fig. 27 illustrates charts reflecting immunostimulatory bias of engineered IL-2 via STAT5 signaling and biolayer interferometry-based receptor binding studies.
- Fig. 28 illustrates charts reflecting IL-2 effector stimulation bias in a quantitative form.
- Fig. 29A illustrates charts reflecting an aptamer sensor exposed to ascites from cancer patients, and PBS as control.
- Fig. 29B illustrates charts reflecting an aptamer sensor in a solution of increasing concentrations of albumin.
- Fig. 30 illustrates charts reflecting down selecting library of surface coating and electrode modification and select/confirm enhanced lifetime of electrode over conventional methods.
- Fig. 31 illustrates a photograph of a biosensor readout platform.
- Fig. 32 illustrates a CAD image of the design of the electronic device integrating all electronic components.
- Fig. 33 illustrates a schematic workflow process of the entire electronic device.
- Fig. 34 illustrates a degradation correction model that corrects the natural decay in the aptamer-based sensors.
- Fig. 35 illustrates a block diagram of initial signal compensation scheme, validation test bench setup for FPGA systems.
- Fig. 36 illustrates a prototype of the FPGA setup.
- Fig. 37A illustrates a humanized murine tumor model timeline.
- Fig. 37B illustrates images of tumor measured by IVIS imaging.
- Fig. 37C illustrates charts reflecting total flux and body weights.
- Fig. 38A illustrates charts reflecting flow cytometry results of CD45+, NK cells, and T cells for spleen harvested in BxPC3 humanized model.
- Fig. 38B illustrates a timeline for immune cell populations of the mice.
- Fig. 39 illustrates charts evaluating the efficacy of human IL12 cytokine factories in treating metastatic tumor model of PDAC lines.
- Fig. 40 illustrates charts evaluating the efficacy of human IL 12 cytokine factories in treating metastatic tumor model of OC lines.
- Fig. 41 illustrates charts reflecting the immune cell composition across the mouse conditions.
- Fig. 42 illustrates a chart reflecting which cytokines/proteins which are clinically relevant sensor targets.
- Fig. 43 illustrates a flowchart of a large language model to predict the effects of various cytokines.
- Fig. 44 illustrates the data generated by the large language model of Fig. 43.
- Fig. 45 illustrates a flowchart of a mathematical model for predicting protein production as a function of voltage applied.
- Fig. 46A illustrates that the mathematical model of Fig. 45 predicted that increasing NF AT expression inside the cells induces increased production of cytokines by the cells.
- Fig. 46B illustrates charts reflecting increasing NFAT3 in the cells does result increased production of IL2 by the cells.
- Fig. 47 illustrates a verification of the prediction of Fig. 46A with in vitro Protein Production Data.
- Fig. 48 illustrates that the mathematical model of Fig. 45 predicted that a 3 mm distance between electrodes for stimulation would result in the greatest stimulation of the cells.
- first, second, third etc. may be used herein to describe various elements, components, regions, layers and/or sections, these elements, components, regions, layers and/or sections should not be limited by these terms. These terms are only used to distinguish one element, component, region, layer or section from another element, component, region, layer or section. Thus, a first element, component, region, layer or section discussed below could be termed a second element, component, region, layer or section without departing from the teachings of the invention.
- relative terms such as “lower” or “bottom” and “upper” or “top,” may be used herein to describe one element’s relationship to another element as illustrated in the figures. It will be understood that relative terms are intended to encompass different orientations of the device in addition to the orientation depicted in the figures. For example, if the device in one of the figures is turned over, elements described as being on the “lower” side of other elements would then be oriented on “upper” sides of the other elements. The exemplary term “lower”, can therefore, encompasses both an orientation of “lower” and “upper,” depending of the particular orientation of the figure.
- “around”, “about”, “approximately” or “substantially” shall generally mean within 20 percent, preferably within 10 percent, and more preferably within 5 percent of a given value or range. Numerical quantities given herein are approximate, meaning that the term “around”, “about”, “approximately” or “substantially” can be inferred if not expressly stated.
- the phrase “at least one of A, B, and C” should be construed to mean a logical (A or B or C), using a non-exclusive logical OR.
- the term “and/or” includes any and all combinations of one or more of the associated listed items.
- the term “implantable” refers to an ability of a device to be positioned at a location within a body of a user, such as subcutaneously, within a body cavity, or etc.
- the terms “implantation” and “implanted” refer to the positioning of a device at a location within a body of a user, such as subcutaneously, within a body cavity, or etc.
- the term “wearable” refers to articles, adornments or items designed to be worn by a user, incorporated into another item worn by a user, act as an orthosis for the user, or interfacing with the contours of a user's body.
- “biocompatible” material is a material that is compatible with living tissue or a living system by not being toxic or injurious and not causing immunological rejection.
- therapeutics and “therapeutic agent” refer to any substance that provides therapeutic effects to a disease or symptom related thereto.
- a therapeutic agent refers to a substance that provides therapeutic effects to any diseases or biological or physiological responses to the diseases.
- the term “therapy” refers to any protocol, method, and/or agent that can be used in the management, treatment, and/or amelioration of a given disease, or a symptom related thereto.
- the terms “therapies” and “therapy” refer to a biological therapy, supportive therapy, and/or other therapies known to one of skill in the art, such as medical personnel, useful in the management or treatment of a given disease, or symptom related thereto.
- treat refers to the reduction or amelioration of the progression, severity, and/or duration of a given disease resulting from the administration of one or more therapies (including, but not limited to, the administration of microspheres disclosed herein). In certain embodiments, the terms refer to the reduction of pain associated with one or more diseases or conditions.
- engineered cell(s) refers herein to cells having been engineered, e.g., by the introduction of an exogenous nucleic acid sequence or specific alteration of an endogenous gene sequence.
- An exogenous nucleic acid sequence that is introduced may comprise a wild type sequence of any species that may be modified.
- An engineered cell may comprise genetic modifications such as one or more mutations, insertions and/or deletions in an endogenous gene and/or insertion of an exogenous nucleic acid (e.g., a genetic construct) in the genome.
- An engineered cell may refer to a cell in isolation or in culture.
- Engineered cells may be “transduced cells” wherein the cells have been infected with e.g., an engineered virus.
- a retroviral vector may be used, such as described in the examples, but other suitable viral vectors may also be contemplated such as lentiviruses.
- Non-viral methods may also be used, such as transfections or electroporation of DNA vectors.
- DNA vectors that may be used are transposon vectors.
- Engineered cells may thus also be “stably transfected cells” or “transiently transfected cells”. Transfection refers to non-viral methods to transfer DNA (or RNA) to cells such that a gene is expressed.
- Transfection methods are widely known in the art, such as calcium phosphate transfection, PEG transfection, and liposomal or lipoplex transfection of nucleic acids.
- Such a transfection may be transient but may also be a stable transfection wherein cells can be selected that have the gene construct integrated in their genome.
- Present system described herein features a next-generation bioelectronic platform for Targeted Hybrid Oncotherapeutic Regulation (THOR), based on a biohybrid system combining engineered cell factories that are activated and supported by bioelectronics.
- THOR Targeted Hybrid Oncotherapeutic Regulation
- regulation of production and dose are enabled by resilient sensors and sensing informatics integrated into the bioelectronic platform.
- the combined sensing and actuation implant, the Hybrid Advanced Molecular Manufacturing Regulator (HAMMR), as shown in Fig. 1, boosts response rates from less than 10% to over 50% by providing closed-loop patient-specific regulation of therapeutic immunomodulating molecules.
- the HAMMR of the present invention is minimally invasive implant that provides patient-specific electronically regulated dosing with minimal disruption to the patient’s lifestyle.
- the present invention involves breakthroughs in high-density loading of engineered cells, durable biosensors, and low-power electronics for signal processing and wireless data and power transfer.
- the resulting bioelectronic cancer therapy enables both systemic and local (tumor microenvironment) delivery of therapies.
- the HAMMR of the present invention With a diameter of ca. 1 cm, the HAMMR of the present invention is implanted in a minimally invasive out-patient surgery into the intraperitoneal space where it improves treatments for many of the deadliest treatment- refractory solid-organ malignancies including ovarian cancer, colorectal cancer, pancreatic cancer, hepatocellular carcinoma, and more.
- peritoneal malignancies account for approximately 34.8% of cancer deaths in the United States, and the ability to treat these diseases could save 187 thousand lives in the US annually. Furthermore, the vast majority of these malignancies are largely unresponsive to immunotherapies. Thus, the present invention improves outcomes for over a third of cancer patients who would otherwise succumb to their disease.
- the present invention develops an implantable actuation-only version of HAMMR applied to humanized mouse models of peritoneal and metastasis cancer.
- sensors are developed in vitro.
- sensors are developed in vivo.
- the present invention includes sensing/actuation integration, with validation of function, biocompatibility in large animals, and efficacy in humanized rodents, with a first-in-human trial.
- THOR’s HAMMR is built for clinical translation using the latest advances in synthetic biology, biomaterials, biosensing, and wireless communications/power and optimized for long-term (>60d) function in the complex tumor microenvironment.
- the present invention miniaturizes hardware, sensors, and computation to enable broader access and long-term effectiveness in diagnostic and therapeutic components.
- the present invention addresses Scalable Solutions and Resilient Systems, increasing access for underrepresented groups to life-saving care because these technologies will be available outside major cancer centers.
- automatically adjusting the dose for each patient's specific needs overcomes biases that can appear in drug development when trials are performed in small cohorts that may not represent the entire population of patients to accelerate clinical therapy discovery.
- Feedback-regulated control of immunotherapy can yield transformative improvements in boosting immunotherapy response rates across broader patient populations.
- the present invention provides a 5X increase in therapeutic efficacy, which is enabled by the following key innovations in synthetic biology, bioelectronics, and machine learning that allow personalized local delivery of therapeutic biomolecules.
- the present invention targets local and systemic therapies.
- immunotherapies need to activate both local immune responses in the tumor microenvironment as well as systemic immune responses in secondary lymphoid organs, as shown in Fig. 2.
- Most immunotherapies fail either because dose-limiting toxicity prevents their administration at concentrations necessary for efficacy or because they never generate systemic anti-tumor immune responses.
- the HAMMR system of the present invention expands the therapeutic window of systemically-toxic immunomodulatory molecules (e.g., IL-2 and IL- 12) through local delivery, while simultaneously facilitating systemic activation of adaptive immunity through secretion of systemic immunomodulatory molecules (e.g., anti-PD-1), engineered with enhanced biodistribution.
- systemically-toxic immunomodulatory molecules e.g., IL-2 and IL- 12
- systemic immunomodulatory molecules e.g., anti-PD-1
- the present invention has the unique ability to address both failure modes of immunotherapies to date.
- the present invention discloses patient-specific dose/feedback- regulated therapies to improve safety/efficacy of the device.
- a sense-and-respond device that adjusts output immunotherapy production to keep therapeutic doses near the levels needed for activation of immune receptors is critical for improving the objective response rates of therapy, as shown in Fig. 2.
- the present invention has opted for electrogenetic induction - compared to other cellular control mechanisms, this method enables fast response, tunable, localized induction properties, and is readily integrated into the platform with minimal power and size demands.
- HAMMR is designed to account for the natural patient-to-patient heterogeneity of the immune response and allow for variable patient bespoke dosing levels to achieve optimal therapy while avoiding toxicity.
- the platform integrates sensors that are engineered to sense and report extracellular levels of both the produced drug along with biomarkers of treatment efficacy and toxicity.
- the present invention combines validated PK/PD models and machine learning to optimize dosing. Feedback is achieved through modulation of electrogenetic cell activation.
- the present invention adopts electrogenetics for stimulating the engineered cells’ production of therapeutics.
- the present invention uses conductive scaffolds, as a 3D electrode to active the electrogenetic cells.
- the HAMMR system expands the therapeutic window of systemically-toxic immunomodulatory molecules (e.g., IL-2 and IL- 12) through local delivery, while simultaneously facilitating systemic activation of adaptive immunity through secretion of systemic immunomodulatory molecules (e.g., anti-PD-1), engineered with enhanced biodistribution.
- systemically-toxic immunomodulatory molecules e.g., IL-2 and IL- 12
- systemic immunomodulatory molecules e.g., anti-PD-1
- a sense-and-respond device that adjusts output immunotherapy production to keep therapeutic doses near the levels needed for activation of immune receptors is critical for improving the objective response rates of therapy.
- HAMMR is implanted in the intraperitoneal space through a small incision. Recharging and data offload can be done through a garment of or hub once daily.
- the present invention has opted for electrogenetic induction - compared to other cellular control mechanisms, this method enables fast response, tunable, localized induction properties, and is readily integrated into the platform with minimal power and size demands.
- HAMMR is designed to account for the natural patient-to-patient heterogeneity of the immune response and allow for variable patient bespoke dosing levels to achieve optimal therapy while avoiding toxicity.
- the device integrates sensors that are engineered to sense and report extracellular levels of both the produced drug along with biomarkers of treatment efficacy and toxicity.
- the present invention combines validated PK/PD models and machine learning to optimize dosing. Feedback is achieved through modulation of electrogenetic cell activation.
- Implanted biomaterials often induce foreign body responses, strong innate immune- mediated cascades consisting of inflammatory events, and wound-healing processes that lead to fibrosis and subsequent implant failure.
- the present invention overcomes critical limitations of allogeneic cell therapies and improve long-term cell survival by encapsulating cells in immunomodulatory-alginate derivatives.
- the present invention has identified lead formulations that protect encapsulated cells from the host immune system for at least 6 months. Furthermore, these capsules with allogeneic cells remained free of fibrotic deposition for up to 4 month in non-human primates, as compared to ⁇ 2w for SLG20 clinical grade alginates.
- alginate-based derivatives minimizes inflammatory responses and prolong in vivo cell-mediated protein delivery.
- a safe, highly-controlled on demand electrocatalytic on-site oxygenation platform is integrated.
- nanostructured and biocompatible sputtered iridium oxide is used to sustain cell viability while maintaining therapeutic peptide secretion with high-cell density (>60k/mm 3 ), enabling higher dosing.
- the present invention has successfully demonstrated this concept in hypoxic in vitro conditions (1% O2) for 3 weeks, and in vivo (rat) for 10 days.
- Biomarkers to guide personalized immunotherapy to maximize efficacy and limit toxicity uses a broad range of preclinical models, including non-human primates and humanized murine models, to validate the utility of biomarkers sensed by HAMMR in predicting efficacy/toxicity. Furthermore, the present invention establishes a computational framework to integrate the power of machine learning with multi-omics data from both clinical and preclinical results. Using this framework, one expects to identify novel biomarkers associated with antitumor activity and/or immune-related adverse effects. These biomarkers are used to optimize the implementation of cytokine-based immunotherapy.
- Lymph nodes can harbor sustained pools of tumor- reactive T cells, and the generation of these systemic durable immune responses within lymph nodes is the key feature distinguishing responders from non-responders.
- patients who fail to maintain durable responses following cessation of ICI treatment likely never generated systemic anti-tumor responses and memory within lymph nodes, and clinical trials (e.g., SWOG- 1801) indicate that commencing immunotherapy prior to lymph node resection (i.e., in the neoadjuvant setting) drastically improves survival.
- these treatment regimens are often as short as four weeks, indicating, in agreement with preclinical ICI and vaccination studies, that the generation of systemic immunity occurs rapidly.
- the HAMMR device of the present invention remains operational for 60 days, more than twice the length of the neoadjuvant treatments in the NSCLC trials.
- THOR is uniquely poised to elicit rapid, durable anti-tumor responses to some of the deadliest malignancies.
- the HAMMR is a novel platform for delivery of a wide range of therapeutics but developed here for cancer immunotherapy.
- the present invention develops and tests in humanized mice the efficacy of this approach, safety in large animals, and evaluates both safety and efficacy in a first-in-human clinical trial in refractory recurrent ovarian cancer patients.
- the present invention develops this new cancer therapy platform which reduces cancer- related deaths by > 50%.
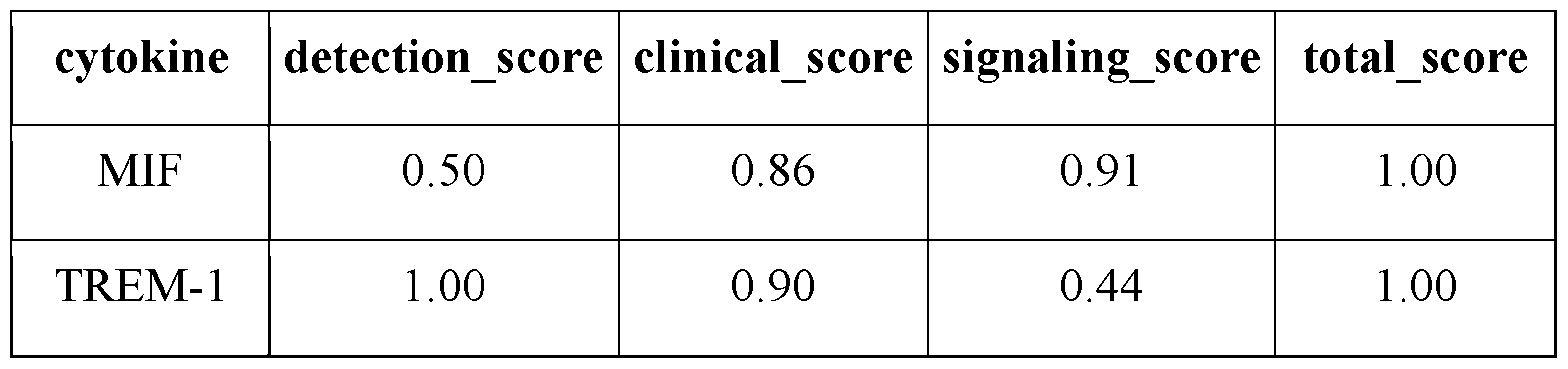
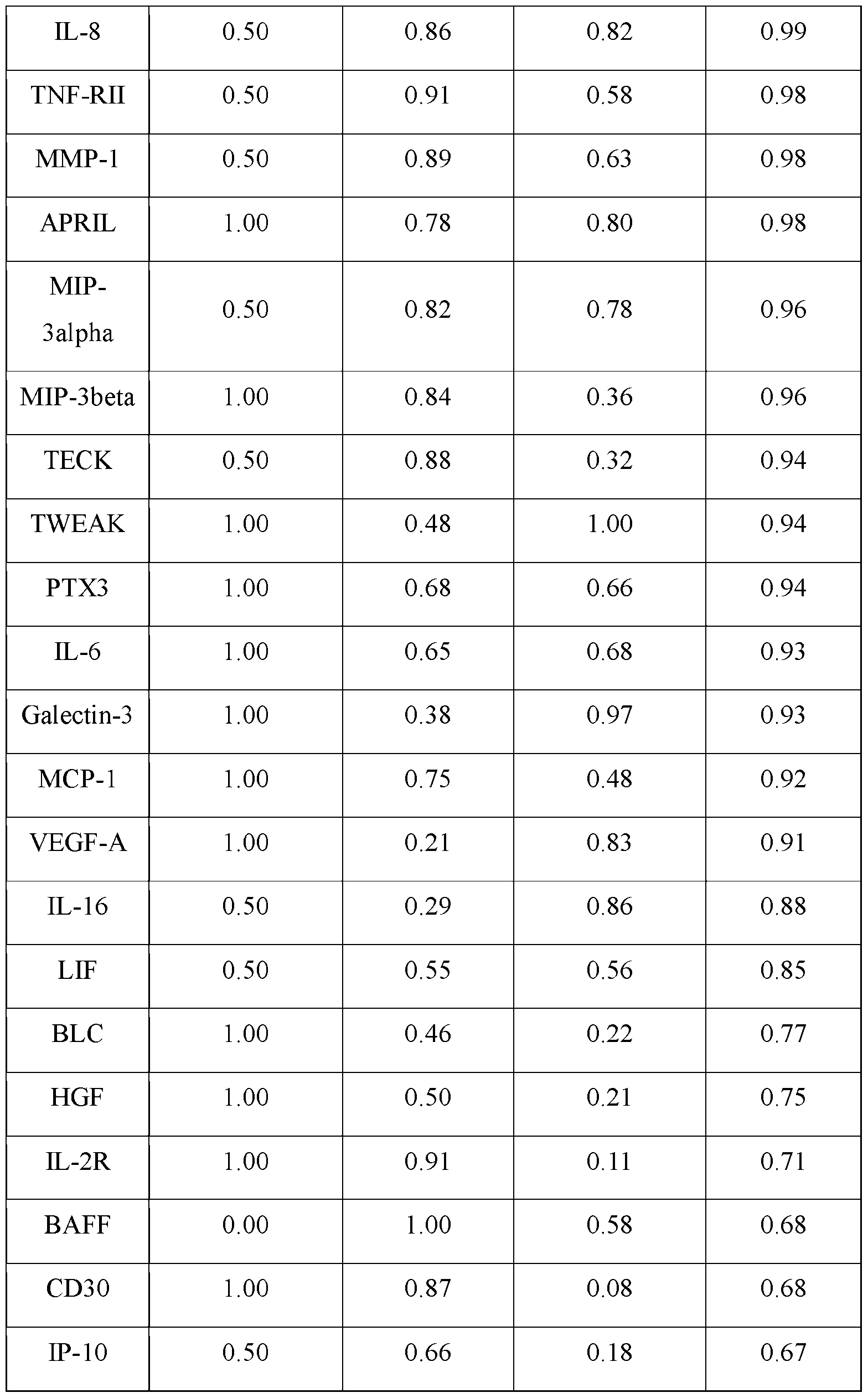
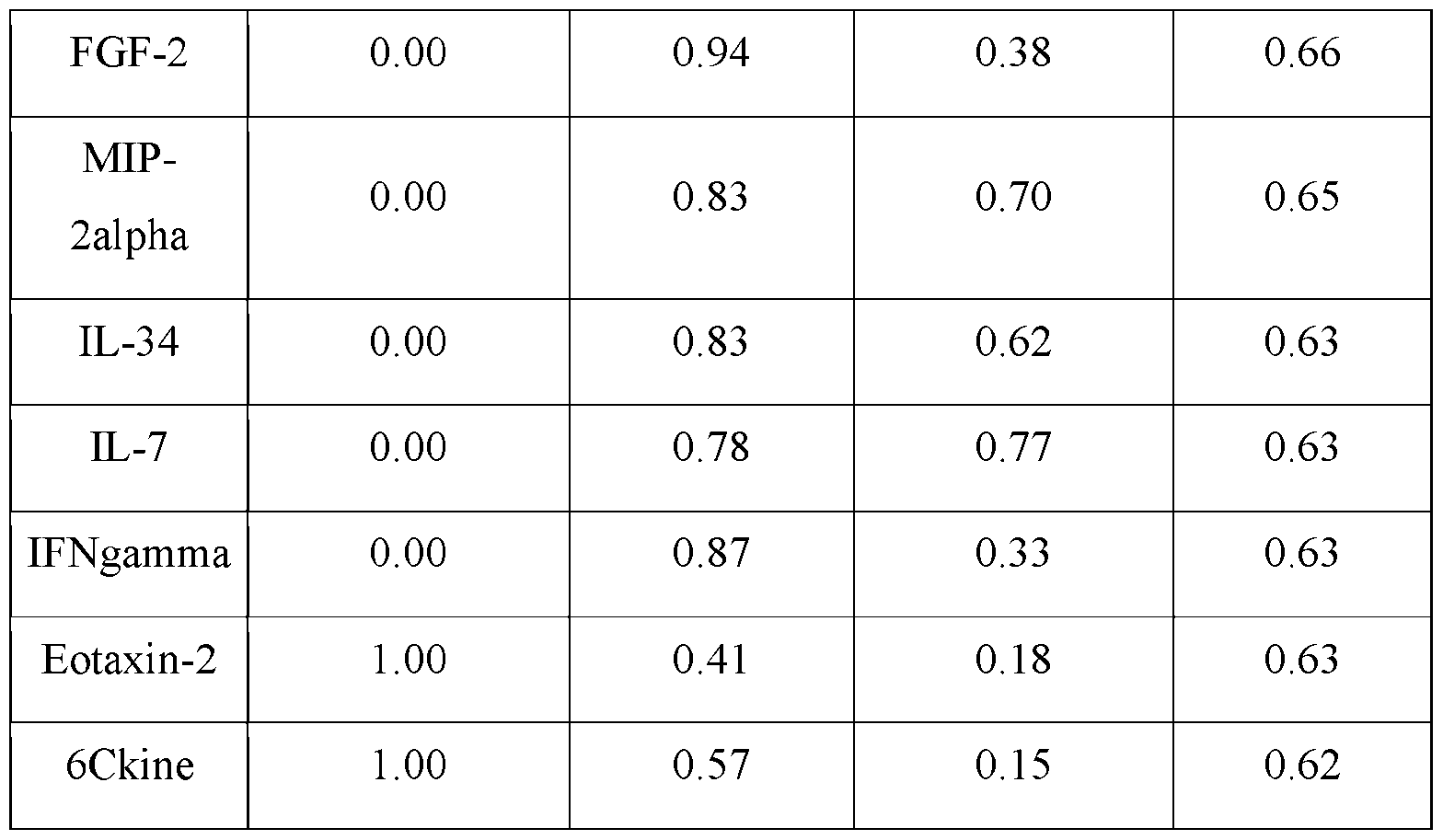
- the present invention targets the following immunomodulatory molecules for production by the cell factories: IL-2, IL-12, IL-18, IFNa, anti-CD40, anti-PD-1, anti-PD-Ll, and anti-Lag3, which were selected and prioritized for engineering based on criteria detailed in Fig. 3.
- immunostimulatory reagents such as IL-12, anti-CD40 could result in severe or unacceptable immune-related adverse effect (irAE), while irAEs from other types of immunotherapy, such as anti-PD-l/Ll are largely manageable.
- the present invention stratified immunomodulatory reagents into two categories based on expected locations for target dose: 1) Local (severe or unacceptable irAEs) and 2) Systemic (no or manageable irAEs). Then, the present invention pairs one type of local reagent with one type of systemic reagents to balance feasibility, novelty, and targeted cell types/pathways. Finally, the present invention ranks each pair based on readiness, expected efficacy, and toxicity. As such, the present invention discloses a target ranking of 5 pairs of immunoregulatory reagents. [00139] In one embodiment, HAMMR also senses an array of targets. For example, monitoring the therapeutic molecules above provides for direct feedback control to enable accurate and consistent dosing.
- the HAMMR device monitor patient responses by sensing an array of cytokines which have been associated with immunotherapy efficacy, toxicity, or both, particularly in the context of ICB and cytokine therapy.
- the present invention has implicated multiple cytokines. Specifically, efficacy is monitored with the T cell recruitment chemokine CXCL10, the immunosuppressive cytokine TGF-13 and angiogenesis promoting cytokine VEGF, all of which have been positively or negatively associated with ICB responses. TNF-a, IFN-y, and IL-8 are measured as elevated levels of these cytokines indicate response to multiple immunotherapies, while excess levels can indicate toxicity, including during cytokine therapy. Finally, IL-6 is included due to its strong association with toxicity in most immunotherapies.
- the specific animal model for various tasks is described herein.
- the present invention carefully considered the most appropriate animal model for each task. Live animals are used only for tests requiring fully intact physiology or behavior, with rodents being used for early cell and bioelectronic component development. Efficacy for human cancer immunotherapies can best be modeled in humanized mice.
- the present invention uses cynomolgus macaques because they are more immunologically similar to humans than rodents, and thus are a more appropriate surrogate model to study before translating to human patients.
- NHPs provide the greatest translational validity for safety and PD studies of any animal model.
- Electrogenetic Cell Factories The present invention discloses a drug delivery system for controlled release of immunomodulatory agents in response to electric signals, and engineers and validates cell factories using electrogenetic control.
- the present invention leverages innovative, recently described electrogenetic systems that use membrane depolarization to control the NF AT transcription factor and, in turn, regulation of user-defined target genes. These systems have been prioritized over other possible modalities due to their robustness, predictability, and demonstrated efficacy in human cells.
- the present invention uses human retinal pigmented epithelial cells, ARPE-19 cells because it has been shown that these cells are non-tumorigenic, display contact inhibited growth characteristics, are amenable to genetic modification, and have been shown safe in previous human trials (NCT03316300).
- the present invention has engineered ARPE-19 cells to produce a wide range of cytokines continuously and in response to light, as shown in Fig. 4.
- Encapsulated ARPE-19, IL-2 cytokine factories have advanced to human clinical trials for patients with high-grade serous adenocarcinoma of the ovary, primary peritoneum, or fallopian tube.
- the cell engineering research team has deep and demonstrable expertise in engineering human cells for biomedical utility, mammalian synthetic biology, protein engineering, and modeling drug dosing and synthetic biology circuit kinetics.
- the present invention employs electrogenetic strategies based on equipping human cells with calcium and potassium channels to achieve membrane depolarization- mediated control of the NF AT transcription factor in ARPE-19 cells, as shown in Fig 5, panel (a, top) and, in turn, regulation of immunomodulators shown in Fig. 3.
- the present invention first develops ARPE-19 cells that express the L-type voltage-gated calcium channel CaV1.2 and the potassium channel Kir2.1 (EiectroARPE-19), similar to previous reports. Briefly, electrical pulse triggers membrane depolarization and the opening of the calcium channel. This process results in calcium influx and activation of the calcineurin/calmodulin pathway, and translocation of dephosphorylated NF AT into the nucleus.
- Electro ARPE- 19 cells is validated and optimized using an expression vector encoding human IL-2 under the PNFAT3 promoter and quantifying IL-2 levels in the cell culture supernatant upon electrical pulse stimulation using ELISA.
- the present invention uses custom or commercial well plates and electronics hardware to deliver electrical stimulation with electrodes onto which cells are seeded (and a second electrode is immersed into the media), or a system whereby EiectroARPE-19 cells are seeded on transwell inserts, with platinum electrodes placed at opposite sides of the transwell fdter, as shown in Fig. 5A.
- the present invention measures IL-2 production from cells using ELISA and correlate voltage, pulse length, frequency and stimulation time with IL-2 expression. In one embodiment, the present invention achieves secretion of IL-2 within 60 min. Importantly, this workflow is compatible with any immunomodulator listed in Fig. 3 and/or any new engineered immunomodulators.
- the present invention constructs a coupled model describing the electrogenetic control system and pharmacokinetics. Expression of the therapeutic protein is tunable by modulating aspects of the electrical signal, e g., voltage or pulse length which lead to membrane depolarization.
- the model consists of ordinary differential equations describing membrane depolarization, calcium ion flux into the cell, subsequent activation of NF AT transcription factor, and expression of the protein of interest.
- the models predict the level of protein production and the kinetics (delays) of activation/deactivation as a function of the electric input parameters. Model predictions are used as an input to PK/PD modeling to optimize device design to maintain safe and efficacious levels of the therapeutic of interest and to design a control system.
- electrogenetic circuit responses might not be as fast as what has been previously described; faster response times may be needed for therapeutic molecules. Therefore, in one embodiment, the present invention explores other electrogenetic systems and/or implement a type 1 coherent feedforward loop motif with the electrogenetic circuit to reduce the delay of response to the electrical pulse signal and amplify transcriptional output.
- a second risk is that Electro ARPE- 19 cells may not perform similarly to cells in which electrogenetic systems have been optimized (i.e., HEK293T, pancreatic 0-cells).
- the present invention optimizes the performance of the electrogenetic platform in alternative cells if needed (i.e., HEK293T, MCF7, MSCs, HS27 cells) and subsequently port optimized circuitry into Electro ARPE- 19 cells.
- the present invention has demonstrated that the production of transgenic payloads can be enhanced using optimized construct designs.
- the present invention optimizes the expression of the electrogenetic systems from 1.1.1. driving immunomodulatory payloads to >lpg/cell/day from stimulated cells using these approaches.
- the present invention tests different combinations of modularized synthetic biology parts and plasmid construct architectures that have been observed to enhance mammalian gene expression, as shown in Fig. 5B, which involves testing various synthetic mammalian promoters (e.g., CMV, CAG, EFS, hUbC), optimized Kozak sequences, nuclear export signals (NESs), and/or polyA tails.
- various synthetic mammalian promoters e.g., CMV, CAG, EFS, hUbC
- optimized Kozak sequences e.g., nuclear export signals (NESs), and/or polyA tails.
- the present invention delivers the electrogenetic system to EiectroARPE-19 cells using multiple vectors and a single, all-in-one (AIO) vector.
- AIO all-in-one
- the present invention explores different arrangements of gene expression cassettes on individual vectors within each system variant. Optimization is initiated using IL-2 and the electrogenetic workflow described above. The present invention then evaluates that electrogenetically-induced IL-2 expression is sustained and tunable (i.e., on/off controllable in response to electrical input) over lOd in vitro.
- the optimized architecture for the expression of the IL-2 immunomodulator in EiectroARPE-19 cells is then evaluated for use with a second immunomodulator (anti-PD-1), the sustained and tunable expression of which is also measured over lOd in vitro.
- the present invention performs mixed cell experiments with these two engineered EiectroARPE-19 cells.
- the optimal levels and ratios of mixed engineered EiectroARPE-19 cells are defined based on the model predictions and in vitro data.
- Optimized construct designs/architectures is then used to create electrogenetically- responsive production of the remaining 6 immunomodulators (IL-12; anti-PDLl; INF a, anti- Lag3, anti-CD40 agonist, and DRIL-18) in EiectroARPE-19 cells and rank ordered combinations 2-5 as shown in Fig. 3, which are then assayed similarly in vitro.
- immunomodulators IL-12; anti-PDLl; INF a, anti- Lag3, anti-CD40 agonist, and DRIL-18
- the present invention uses high-throughput modular cloning platforms to optimize each immunomodulator in EiectroARPE-19 cells one by one. If ELISA is too slow for processing, the present invention discloses other systems in which the immunomodulator proteins are fused to a reporter (e g., SEAP/EGFP), allowing one to determine the immunomodulator protein expression level through corresponding quantification assays. Further, the present invention has also demonstrated that transgenes and endogenous gene products can be robustly activated using innovative CRISPR synthetic biology technologies. [00149] In certain embodiments, the present invention optimizes engineered human allogeneic cells to become potent factories of immunomodulators in response to electrogenetic stimulation.
- a reporter e g., SEAP/EGFP
- the present invention optimizes the Electro-cells to meet a production threshold of 1 pg/cell/day.
- the present invention can reach physiological drug levels with a limited number of cells.
- Different construct designs will be tested to find the system architecture that boosts production of individual immunomodulators.
- Expression of immunomodulators will be tested in vitro over 10 days to ensure that expression is both sustainable and tunable (i.e., on/off controllable in response to electrical input) over 10 days in vitro in these engineered cells.
- We will perform mixed cell experiments in vitro with two different engineered ElectroCells (each expressing a unique immunomodulator) and define optimal levels and ratios of mixed engineered ElectroCells for therapeutic use.
- the present invention discloses a Completion Criteria and Performance Metrics/Assessment.
- construct designs including the use of various synthetic promoters (e.g., CMV, CAG, EFS, or hUbC), plasmid architectures (delivery through individual vectors versus all-in-one delivery), and/or other genetic elements (e.g., polyA tails, Kozak sequences, nuclear export signals).
- the production features of each design will be evaluated by measuring protein expression levels (via ELISA) corresponding to the immunomodulator of interest.
- optimized architectures are tested using two lead immunomodulators (i.e., IL-2 and anti PD-1) and then ported to other i mmunomodul ators .
- the engineered cells used in the present invention are by design create pro- inflammatory environments via the production of high levels of potent immunomodulators. Recent studies have shown that cells expressing therapeutic payloads can be adversely impacted by inflammatory environments, such as those that may be produced by the engineered EiectroARPE-19. Although this is able to be controlled by tailoring electrogenetic on/off kinetics and thereby immunomodulator production, the present invention further improves EiectroARPE-19 cells to be durable and resistant to inflammatory environments.
- the proinfl ammatory cytokines IFNy, TNF-a and IL-ip act broadly and are each known to cause overall decreases in ARPE-19 function.
- the present invention also engineers ARPE-19 cells to be resistant to these signaling molecules using CRISPR/Cas9-mediated knockouts (KOs).
- KOs CRISPR/Cas9-mediated knockouts
- the present invention KO the receptors for IFNv (IFNGR1/2), TNF-a (TNFR1/2 and TRAF-1/2), and ZL-10 (IL-1R1/2 and IL-1RAP) in ARPE-19 cells and measure relative cell viability and function between WT and ARPE receptor KO cells using Annexin staining.
- engineered ARPE-19 KO cells are grown in media containing IFN-y, IL-ip, and TNF-a to mimic in vivo adverse immune environments. These experiments are carried out over 60 days.
- ARPE-19 cells with improved resilience to these pro-inflammatory molecules are ported into 1.1.2 to synergize with electrogenetic production efforts.
- KOs may be lethal or reduce overall cell fitness.
- the present invention assesses these phenomena and omit lethal KOs/KOs that reduce cell fitness.
- KOs of indicated receptors may lead to no significant benefits in cell production/viability, in which case the present invention explores CRISPR screening modalities to identify and utilize key drivers of improved immunomodulatory production in ARPE-19 cells.
- Naturally occurring proteins are limited in number and also by their pleiotropic effects, which hinders their efficacy as targeted therapeutics.
- engineered new proteins are produced by the engineered cells of the present invention for cancer therapy.
- target proteins are selectivity and circulation half-life.
- Locally delivered targets are intended to mobilize immune effector cells (i.e., CD4+ and CD8+ T cells and NK cells) to clear tumors.
- IL- 12 IFN-a, an anti-CD40 agonistic antibody, and decoyresistant IL-18 activate immunostimulatory and not immunosuppressive effects.
- IL-2 protein engineering for selectivity is required, as IL-2 activates both immune effector cells and regulatory T cells (Tregs), confounding its efficacy as an anti-cancer agent.
- the present invention engineers an IL-2/antibody fusion protein (F10 IC) that biases IL-2 towards activation of immune effector cells over Tregs.
- F10 IC IL-2/antibody fusion protein
- This molecule is based on a full-length antibody and has limited tumor penetration.
- the present invention designs an IL- 2/single chain variable fragment (scFv) fusion protein that recapitulates the bias of F10 IC in miniaturized format.
- the present invention performs IL-2 signal activation studies via flow cytometry and receptor binding studies via biolayer interferometry to validate the biased activity of our engineered IL-2 molecule.
- PK property engineering locally delivered targets are intended to be transient; thus, a short in vivo half-life is desired.
- Systemically delivered targets are intended to be available throughout the body for sustained time periods; thus, a long in vivo half-life is desired.
- the only locally delivered target with a long in vivo halflife is the anti-CD40 agonistic antibody, which has extended half-life due to engagement of its Fc domain with the neonatal Fc receptor (FcRn).
- the present invention mutates select residues in the antibody Fc domain to knock out FcRn interaction without impacting protein function.
- Biolayer interferometry -based binding studies confirm ablation of FcRn binding.
- Systemically delivered targets include antibodies against PD-1 (pembrolizumab), PD- L1 (atezolizumab), and LAG3 (relatlimab), all of which have extended half-life.
- the other 3 locally delivered molecules (IL- 12, IFN-alpha, and DR-18) can be substituted.
- the present invention engineers any of the locally delivered or systemically delivered proteins to alter their selectivity and/or PK behavior as needed.
- the present inventio tunes the selectivity, pharmacology, and immunogenicity of the delivered molecules. Selectivity can be tuned through modulation of target affinity.
- PK properties can be manipulated to extend or restrict serum halflife through Fc domain engineering of antibodies. Potentially immunogenic epitopes can be identified through computational algorithms to screen for class II MHC peptides, and the proteins can be mutated accordingly to mitigate these liabilities.
- Electrogenetic induction of therapy requires uniform actuation over a large volume of engineered cells. While planar electrodes have been used previously, it is known that stimulation electric field scales as r 2 where r is the distance from the electrode, thus the consistency of activation can be variable over the cell factory population.
- the present invention uses a conductive scaffold or mesh which is embedded within the cells or cell-laden hydrogel. In one embodiment, the present invention uses a conducting scaffolds of high surface area, scalability, and of sufficient conductivity to enable uniform electrogenetic actuation.
- the present invention develops high conductivity poly(3,4-ethylenedioxythiophene) (PEDOT) particles produced via dropwise addition to an acid bath, yielding particles with highly tunable internal surfactants (controlled by remaining PSS content from the initial PEDOT:PSS dispersion.) These particles form high performance inks with a slew of biomaterials, and can be printed or cast and crosslinked through a number of modalities, as shown in Fig. 6. In particular, Fig.
- PEDOT poly(3,4-ethylenedioxythiophene)
- PEDOT-based particles (410 S/cm) are highly dispersible within aqueous 3D- printable resins for (A) micro-continuous liquid interface production (pCLIP) stereolithography to generate conductive scaffolds; (B) Photo and SEM image of PEGDA stents (-100 pm diameter struts) with 10 wt% loading of PEDOT particles. Scale bars for the photographs and SEM images are 2 mm.
- micro continuous liquid interface printing pCLIP
- ice- templated, or fibrous conducting polymer scaffolds commercially available graphene foams or Pt wire meshes can be used.
- Activation is tested in modified well-plates with a control counter electrode (Pt) and compared to activation with flat metallic electrodes. While flat electrodes typically use stimulation parameters of 7.5V, 1ms pulses with a frequency of 1Hz, the electrodes of the present invention reduced average cell-electroactive scaffold distance lowers the stimulation voltages, while scaling current with overall surface area, which is part of the materials downselection selection approach. The tradeoff in power consumption vs. total cell activation is explored balancing therapy production and power draw. The connection to the stimulation hardware is separately be investigated to determine the robustness and durability using both physical clamping and conductive epoxy approaches, before integrating the device for in vitro /vivo validation, and integration. In one embodiment, the present invention also discloses the use of the oxygenation hardware as the CE electrode for stimulation.
- the hardware can probe for changes in scaffold impedance.
- the present invention discloses conductors like Pt meshes, or photolithographically patterned mesh electrodes that are monolithically integrated with the electronics hardware (i.e., on Flex PCB), which also address connect! on/integrati on issues.
- planar electrodes can be employed, and device dimensions reduced to account for non-uniformities - at the cost of accessible dose.
- Fibrosis is one of the major contributing factors for the failure of many implanted devices.
- Cells are of particular issue with fibrosis since they evoke a strong immune response, and a fibrotic capsule stops the flow of biomolecules to and from the cells and kill them.
- the present invention engineers immunomodulatory biomaterials with improved biocompatibility that mitigates foreign body response. This is accomplished by developing alginate-based hydrogel formulations that are immune modulating in nature and can host the engineered cells for prolonged duration, as shown in Fig. 7.
- Fig. 7 shows immunoisolating chamber and chemical coatings to protect against foreign body responses.
- Panel (A) shows engineered cells loaded in immunoisolation chamber;
- panel (B) shows high viability post loading (3d. Post encapsulation);
- panel (C) shows small molecule surface chemistry protects against host FBR (28d explants from mice); and
- panel (D) shows host tissue biocompatibility of surface modified devices (28d histology adjacent to implants).
- the present invention discloses a library of alginate-based hydrogels that could mitigate fibrosis. These hydrogels have been previously employed for the transplantation of xenogenic human pancreatic islets that led to the restoration of long term glycemia in profibrotic C57BL/6. These hydrogels prevent fibrosis and promote long term cell viability, as shown in Fig. 7 panels (B) and (C).
- the present invention discloses a carrier that mitigates fibrosis and supports the engineered cells.
- the present invention modifies the backbone of the sodium alginate polymer with immunomodulatory small molecules.
- the present invention performs a short in vivo study spanning over 14 days to evaluate the fibrosis and hence their biocompatibility.
- these materials successfully evaded fibrosis and supported viability for greater than 80 days when capsules fabricated from them were implanted in the intraperitoneal space of mice and NHPs. Since the device aims to host the cells for over 60 days, the present invention also evaluates the biocompatibility of these materials in vivo over extended time points of 30, 60 days.
- linkers are used for the library of alginate-based hydrogels that could mitigate fibrosis.
- the present invention engineers an electrocatalytic on-site oxygenation (ecOz) for implantable cell therapies. This is accomplished by electrocatalytic water splitting using integrated catalytic arrays in the cell-containing hydrogel.
- ecO2 arrays which adopts sputtered IrCh (SIROF) catalyst were able to support the bioactivities of high-cell density capsules (60k cell mm-3) up to 21 days under hypoxic environment (1 % O2) as well as implanted in rodent for 10 days, as shown in Fig. 8.
- SIROF sputtered IrCh
- Fig. 8 shows electrocatalytic onsite oxygenation (ecO2).
- Panel (a) shows linear sweep voltammetry (LSV) measurement in anodic scan for water oxidation; panel (b) shows cell viability (Live/dead assay) and peptide secretion (ELISA) as a function of time; panel (c) shows optical imaging of capsules after 21 -day hypoxic (1 %) incubation with or without oxygenation; and panel (d) shows immunostaining images after 10-day implantation in rats with or without oxygenation.
- LSV linear sweep voltammetry
- ELISA peptide secretion
- Microfabricated ecO2 arrays are translated to flexible substrates (e.g., polyimide), as shown in Fig. 9. Two arrays are facing back-to-back for whole-HAMMR-volume oxygenation. 3D-printed oxygenators with controlled morphologies, e.g., hierarchical helix or coiled electrodes are also tested.
- the present invention discloses 3D electronics employing nanoparticle-based 3D printing with high resolution ( ⁇ 10 pm).
- Catalytic ink is formulated by blending nanoparticles (e.g., IrCh, Pt) to fabricate 3D printed ecO2 as shown in Fig. 9.
- Fig. 9 top panel shows a double-side flexible catalytic arrays; and Fig. 9 bottom panel shows a 3D-printed double helix catalytic arrays; inset: 3D-printed spiral shape electrodes with controlled nano porosity.
- the present invention modifies the synthesis conditions such as the ratio of gas mixture (Ar and O2) synthesis pressure, applied power (DC or RF), and post-processing procedures (e.g., thermal annealing, sintering of 3D printed parts).
- IrO2 crystallinity improvement contributes to extended chronic oxygen generation.
- the present invention tailors crystallographic characteristics and hierarchical nanostructures of catalysts to achieve long-term stability (> 60 days), modulating crystallinity, dimensions of electrode, applied duty cycles, macro- and nanoporosity.
- In vitro production of oxygen to support high cell density is validated by oxygen measurement, oxygen sensitive dye imaging and cell viability (live/dead assay).
- the present invention engineers the therapeutic cell maintenance platform to allow at least 70 % viability for encapsulated 60k cell mm’ 3 under hypoxic conditions for at least 60d.
- Produced peptides is measured via ELISA.
- Oxygen generation platform is evaluated in vivo (rat) to achieve at least 70 % viability over 60d.
- Power supply for electrocatalysis and control unit are securely passivated using medical grade resin (e.g., Parylene C, epoxy, PDMS) to protect them from potential corrosion and water ingress damage.
- medical grade resin e.g., Parylene C, epoxy, PDMS
- IrO2 crystallinity in improve the stability of the catalyst, can be adjusted by modifying the synthesis conditions and/or annealing.
- nanocarbon materials e g., laser induced graphene (LIG) that exhibits exceptional electrochemical stability is used.
- LIG laser induced graphene
- twisted Pt/Pt+SIROF wires are adopted as working and counter electrodes.
- the present invention may use the decomposition of solid peroxides as an alternative.
- the present invention develops miniature electronics to stimulate the electrically- conductive scaffolds.
- the electronics platform is small enough to be implanted in the humanized mouse model, and have the ability to dynamically adjust to changing scaffold impedance to reliably actuate the engineered cells.
- the present invention uses a miniature antenna (RF, solenoid, or magnetoelectric receiver) and a magnetic or RF transmitter to enable contactless wireless power delivery to the implanted HAMMR for multi-day experiments and validation of the actuation- HAMMR. It has been shown that actuation is possible at 7.5 V.
- the present invention may use an off the shelf LCD controller for voltages up to 20V; if the required voltage exceeds 20V, in another embodiment, the present invention uses a Lomir rodent swivel -tether system and power the device via a programmable power supply instead of WPT.
- the present invention develops a computational model.
- the present invention plans to use a minimal physiologically based pharmacokinetic (mPBPK) model structure due to its relative simplicity.
- PK model structure is consistent across delivered therapeutics, whereas modeling PD is dependent on the therapeutic of interest.
- the present invention uses a model of IL-12 therapy where the production rate of IFNy is determined by the formation of complex between IL-12 and IL-12 receptor. This model is specific to IL- 12 and IFNy, but the present invention also uses a similar framework for all therapeutics.
- the PD of the delivered therapeutic is modeled as interaction between the therapeutic and its target and the resulting biological effects.
- the present invention constructs the model as a system of ordinary differential equations describing transport of the delivered therapeutics between compartments, interaction between the delivered therapeutic and its target and biological effects, cell viability within the HAMMR device, and electrogenetic control of the HAMMR device, as shown in Fig. 10.
- the present invention evaluates model performance and estimate parameters by optimizing fit to in vitro and in vivo preclinical data. In vitro data is used to estimate model parameters related to the HAMMR device, whereas in vivo data is used to estimate physiological parameters such as transport and clearance. Model performance is determined quantitatively (minimum weighted sum of error between model predictions and measurements) and qualitatively to ensure predictions are physiologically relevant with no overfitting. In one embodiment, the addition of peripheral tight and leaky tissue, such that the complex trends are also captured.
- the present invention assembles an antifouling strategy on different aspects: nanoscale, micron-scale and macro scale. It has been shown that flat electrode surfaces are more prone to fouling in comparison to nano/micro structured surfaces due to different surface adhesion energies. On the nano/micro scale, the present invention manipulates electrode surface architecture using electrochemical techniques to achieve various morphologies (e.g., pores, nanostars, etc.) and carefully select for surfaces that provide the maximal resistance to fouling. On the macro scale, the present invention, in one embodiment, uses porous hydrogel materials to protect the biofunctional surface from unwanted interaction with the biofluids to enhance surface lifetime. HAMMR sensors can also be inhibited by fibrosis.
- the present invention also identified small molecules that, when functionalized to a material surface, cause the material to resist fibrosis. These coatings have been shown to work on various materials (medical catheters, PDMS, and PTFE) and across multiple implant sites (IP space, subcutaneous space, and the brain), stopping or reducing fibrosis in implanted materials. To this end, the present invention down selects a library of materials/assemblies, show stable operation in complex environments on the benchtop, and show operation, and stable readings in vivo in rats for 30 days.
- biorecognition elements i.e., aptamer
- the present invention incorporates strategies, e.g., anchoring chemistries, electrode material engineering and redox reporter selection, as shown in Fig. 11 panel (a) to tackle bias stress. Operation of aptamer-based sensors at negative potentials strain the phosphate backbone of DNA and cause a reductive desorption of thiol-Au bond, i.e., desorption of aptamer from an Au electrode.
- the present invention selects stable anchoring chemistries from multi-thiol bonding, carbon-nitrogen bonding, multi TL-TI stacking, peptide self-assembly on graphene, and phenyl azide chemistry.
- Multidentate bonding like di-thiol or tri-thiol can potentially improve bonding strength and stability, at the expense of aptamer density.
- the present invention uses nanostructured electrodes with a large surface area for anchoring multidentate aptamers with high aptamer density (same device footprint).
- Nanostructured electrodes with extended electrical double layer over the electrode surface can enhance electron transfer rate and improve detection sensitivity.
- the present invention has synthesized tunable carbon- and metallic-based nanostructured materials such as microstructured gold, as shown in Fig. 11 panel (b), and CVD grown 3D fuzzy graphene with temperature/time controlled flake density, as shown in Fig. 11 panel (d).
- Metallic based nanostructured electrodes can be synthesized by electrochemical deposition and morphology is dependent on the precursor solution concentration and rate of deposition. Nanostructured electrodes are evaluated by cyclic voltammetry (CV) or electrochemical impedance spectroscopy (EIS) for electrochemical surface area characterization.
- CV cyclic voltammetry
- EIS electrochemical impedance spectroscopy
- microstructured gold electrode-based aptamer sensors have been demonstrated to improve the detection limit of TGF- bl compared with bare gold electrodes, as shown in Fig. 11 panel (c).
- modifying the work functions of the working electrode is a technique which can allow for a lower redox potential that ultimately reduces the operating voltage and bias stress.
- Chemical modifications of the surface, metal doping, and self-assembled monolayer formation can be used to modify the work function of the nanostructured electrode materials. Selecting redox reporters with lower redox potential or positive redox potential such as Os also allows stable operation under lower bias stress.
- electrochemical measurement techniques are chosen, i.e., electrochemical impedance spectroscopy (EIS, low amplitude of voltage ( ⁇ 10mV)) and pulsed chronoamperometry (CA, short duration ( ⁇ lms) of voltage) measurements cause less bias stress than the commonly used square wave voltammetry (SWV, high amplitude ( ⁇ 0.5V) and long duration ( ⁇ ls) of voltage) measurement.
- EIS electrochemical impedance spectroscopy
- CA pulsed chronoamperometry
- SWV high amplitude ( ⁇ 0.5V) and long duration ( ⁇ ls) of voltage
- the present invention performs SWV with longer interval time between each scan and narrow the scan range (limit potential range near the peak potential) to minimize the sensor bias stress.
- the present invention has demonstrated that the lifetime of the sensor can be improved by using optimized SWV scheme, as shown in Fig. 12. In particular, Fig.
- Panel (A) shows normalized redox peak vs. time during SWV with different biasing scheme. Sensors with short voltage scan range and long interval time between scans are stable over time; and panel (B) shows preliminary results of the sensor stability on pig wound using the biasing scheme in A.
- the present invention selects optimal redox reporter and attach aptamer through stable anchoring chemistry onto tunable nanostructured sensors, which is tested by optimized electrochemical method in vitro on bench top and allows for a 10-fold increased life-time compare with “standard” sensor.
- the present invention develops bio-recognition elements (i.e., aptamer, antibody, nanobody) with fine-tunable reactivity (association/dissociation) in biofluids through Kd engineering to enable continuous biosensing.
- bio-recognition elements i.e., aptamer, antibody, nanobody
- association/dissociation fine-tunable reactivity
- the present invention generates libraries of recognition elements that are generated from original selected sequences (e.g., starting from aptamer sequences known from the literature, and determining Kd) and modified via mismatch, mutagenesis, chemical modification, and truncation.
- the present invention controls binding dynamics in molecular sensors.
- the present invention probes for fast-binding, highly specific and sensitive recognition species with high- release properties that can enable quick turnarounds and regeneration of the sensors for reusability in vivo.
- the present invention demonstrates sensor regeneration with retention times ⁇ 5 min where other sensor metrics are within a factor a 5 of unmodified aptamer. Sensing informatics
- the ultra-low power, high fidelity, low SNR chronoamperometry microelectronics present a significant amount of incoming data that is noisy, complex, and fast changing.
- the present invention is well positioned to employ sophisticated informatics onboard the implant to navigate both signal degradation concerns from fouling/degrading biosensors, as well as challenging therapy delivery actuation decisions.
- Computational methods can account for degradation via signal reconstruct on/sampling.
- the present invention uses computationally efficient compressed sensing techniques coupled with active learning (i.e., sensing behavioral and mechanistic changes) to reconstruct a lossy signal from degrading sensors.
- the present invention uses sensor data alongside simulated mechanistic traces, with random sampling, for validation purposes.
- Supervised regression-based and recurrent machine-learning techniques are applied to the datasets to learn the behavior of the biosensor as it degrades, allowing for the prediction of and compensation for sensor degradation.
- the present invention compares the error reduction for various model architectures and select a suitable machine learning/signal processing pipeline for execution on the FPGA/ASIC.
- the present invention achieves stable sensor readouts by using a control (i.e., scrambled aptamer), or housekeeping sensor (stable background biomarker determined from omics in 1.10) to yield stable differential signals.
- a control i.e., scrambled aptamer
- housekeeping sensor stable background biomarker determined from omics in 1.10
- the present invention performs SELEX from a randomized DNA library to select for desired aptamers using a microfluidic platform for selecting aptamers in a narrow-desired Kd window.
- unsupervised techniques informed by domain specific mechanisms and behaviors of biosensors, some of which are known a priori, are explored if the amount of data required to train a supervised model is not known.
- the present invention explores constellations of smaller, less accurate models as well as calibration routines to increase model accuracy.
- the present invention encapsulates different sets of sensors in slowly dissolvable protective polymeric scaffolds of differing thickness that will be activated at different timepoints, thus using multiple sensors for a shorter duration each.
- the present invention involves multiple parallel efforts regarding sensing validation, and when testing an approach, direct comparisons are made to a “standardized” version of a particular sensor (i.e., a starting aptamer sequence, bonded via monothiol to a flat gold working electrode) or to one that is properly unmodified (i.e., with and without a protective anti-fouling coating).
- a “standardized” version of a particular sensor i.e., a starting aptamer sequence, bonded via monothiol to a flat gold working electrode
- the ultimate goal is to address stability while down selecting and preparing a sensor set for implementation in HAMMR.
- in vitro validation progresses with a combination of benchtop testing of sensing metrics (sensitivity, limit of detection, lifetime) in spiked PBS, and in complex environment samples such as heparinized blood and patient samples.
- Patient samples are used to assess the accuracy of sensors compared to established analytical tools (e.g., ELISA), and are tested for 30-60 days on the benchtop in complex, heparinized blood. Due to the sheer volume of approaches and targets that must be explored and engineered to meet the strict timelines of THOR, one embodiment of the present invention only focuses on in vivo validation in the antifouling task.
- the present invention builds an in vivo testing device which is implantable (mm-scale) and low-power (tens- of-pW average power draw) capable of accurately reading data from the biosensors developed and sending it (wireless/tether) to a desktop/laptop application for data storage and analysis.
- the present invention develops an integrated hardware platform for cell life support and actuation, sensing and signal processing, energy (charging and usage), and communications. Developing this platform necessitates overcoming challenges in onboard computation, signal processing, and wireless power delivery.
- a bedside or clothes integrated transmitter charges the implant's battery and extracts biosensor data using magnetoelectric (ME) power transfer. This approach has been demonstrated with other WPT- implant systems and enables therapy progress reporting and continuous assessment in clinical trials.
- ME magnetoelectric
- ME power transfer has great potential for wireless mm bioelectronic implants. It offers low tissue absorption, high efficiency, small size, and low misalignment sensitivity. Using the same mechanism for power and communication is crucial for miniaturizing implants.
- the present invention has designed and demonstrated near-zero power magnetoelectric backscatter from mm-sized implants by exploiting the converse magnetostriction effects, in an 8.2-mm wireless implant > 1 kbps data rate at 2 cm and a bit error rate (BER) less than IE-3, as shown in Fig. 13.
- BER bit error rate
- the present invention investigates novel multi- antenna form factors for increased power to generate 5mW at 4 cm in-vivo and explores lossy coding and delay/interrupt tolerant communication protocols to overcome the higher error rate from the longer distance.
- the present invention explores hybrid automatic repeat request (HARQ) protocols used to combat channel fading (as in satellite networks).
- HARQ hybrid automatic repeat request
- the present invention enhances the existing model from 1.2 for PGA and ASIC implementation.
- the present invention has successfully demonstrated accelerator designs for biomedical implants that utilize embedded ML models to process high-rate data and make decisions, as shown in Fig. 13.
- the device includes feedback mechanisms to monitor that all parts of the scaffold are receiving appropriate waveforms.
- the device are used for humanized mice models and is an initial prototype for the fully integrated actuation-HAMMR device.
- the device receives power & commands via one of a few solutions: either off-the-shelf NFC charging, standard cages wrapped with wire for wireless power transfer, or a dedicated tether system, each standard with rodent/murine experimentation systems.
- Bluetooth Low Energy may be used in lieu of the above for communication and control.
- the device provides data offload via this to a smart phone or desktop for capture of experiments.
- the present invention develops primary and metastatic mouse models of peritoneal malignancies. Effective immunotherapies require activation of systemic immunity, and such immunity is ideally capable of clearing stage IV metastatic disease.
- the present invention develops mouse models of metastasis (predominant cause of cancer-associated deaths).
- the proposed immunomodulators are human-specific, the present invention develops humanized murine models suitable for testing immune activation and toxicity of the therapy combinations resulting in >30% reduction in tumor burden for phase 2 studies.
- the present invention monitors the tumor burden of 3 ovarian cancer (SKVO3, Igrov-1 and OVCAR4) and 3 pancreatic ductal adenocarcinoma (Panc-1, AsPCl, and BxPC3) lines, engineered to express luciferase, by bioluminescence imaging (BLI).
- Metastatic models consist of intrasplenic (liver) or lateral tail vein (lung) injections of tumor cells. Reconstitution of the human immune system can be achieved in NSG mice engrafted with human peripheral blood mononuclear cells (Hu-PBMC mice).
- the present invention selects 4 lines (2 for each cancer type) that exhibit 4 weeks of progressive growth in the peritoneum and metastatic sites.
- the present invention uses ARPE-19 cells secreting IL-2 and IL-12 to compare tumor and immune responses in the humanized models to previous syngeneic models.
- the present invention tests whether ARPE-19 cells expressing pairs of immunoregulatory agents listed in Fig. 3 can achieve target concentrations and exhibit 3-fold increase in activation cytokines (e.g., IFN-g), and a 2-fold increase in lymphocyte infiltration and/or proliferation.
- the present invention also confirms that designed biomarker sensors can achieve expected function and support down selection of therapies.
- the present invention evaluates toxicity of the actuation-HAMMR devices in 2 tumor models to ensure ⁇ 20% reduction in body weight and ⁇ 20% variation in liver function tests (LFT)for Phase 2 studies.
- LFT liver function tests
- the present invention focuses on two cancer types frequently causing peritoneal carcinomatosis.
- the present invention screens six lines and validate the findings in at least two humanized mice models in both peritoneal and metastatic settings by two research groups. As 2 weeks are needed to observe the antitumor effects, the present invention selects six tumor lines that are reported to grow well in NSG mice and conducts experiments to down-select four lines. Testing multiple lines mitigates the risk that these models lack therapeutic windows. In one embodiment, the present invention has eight additional OC and PDAC lines should these six not exhibit ideal growth kinetics.
- NHP studies to address PK/PD and safety are accomplished in coordination as an effort to make thoughtful and efficient use of these animals.
- Delivery/safety metrics are obtained from samples and data gathered from the same individual monkeys that are used for PK/PD testing.
- the fully assembled device is validated in rat models.
- the present invention immune-profiles the rat with cytokines delivered from the device to show that one can successfully modulate the immune system.
- Fully assembled devices are then be implanted intraperitoneally into the abdomen site of NHPs. Drug deliveries are determined through remote cell induction and monitoring of drug concentrations in blood serum and IP fluid. Through this study, the present invention demonstrates “turn on” and “turn off’ drug production kinetics at various time points to get data for the PK/PD model.
- the present invention when the dose required exceeds the capacity of the cell factories, the present invention uses higher cell loading densities (as enabled by O2 delivery) or add multiple implants to increase production.
- the present invention fits PK/PD data from large animal models and estimate safe and efficacious HAMMR dose regimens for clinical trials.
- human model parameters are estimated by fitting the model to clinical trial data of the corresponding therapeutics, e.g., data from a clinical trial of IP administration of IL-12. Allometric scaling or in vitro-in vivo extrapolation methods are used to estimate human parameters from in vivo estimated parameters otherwise. In vitro-in vivo extrapolation methods are more physiologically relevant; however, utility is dependent on the quality of data available. Conversely, allometric scaling methods are empirical in nature but simpler to employ. Large animal models more closely replicate PK/PD seen in humans.
- the present invention implements sensitivity analyses to determine which model parameters and assumptions are most important in final predictions.
- the present invention creates baseline omics profiles and models of cytokine molecules from ascites samples of ovarian cancer patients.
- the present invention proposes to develop a peritoneal tumor immune microenvironment agent-based model guided artificial intelligence (PTIME-ABM-AI) system that acts as the brain of the system to intelligently execute Closed-loop control of the PTIME through the cell factories based on input of the sensors, as shown in Fig. 14.
- PTIME-ABM-AI model guided artificial intelligence
- the present invention develops the PTIME- ABMs based on previous work such as the Ovarian Cancer Metastasis Simulation (OCMetSim) models and the Pressure Ulcer Agent Based Model, or PUABM using NetLogo or SPARK.
- OMetSim Ovarian Cancer Metastasis Simulation
- the present invention evaluates and guides the model design and optimization using real-world omics data.
- Multiplexed cytokine profiling data is obtained from around 30 ovarian cancer patient ascites samples by the Luminex assay, including 65 cytokine/soluble factors. Cytokine data is standardized and normalized to remove batch effects. Single-cell data of cellular components from ascites samples obtained through CyTOF and/or scRNA-seq are analyzed to identify cell types and cell states and corresponding RNA and protein molecular profiles.
- the present invention finds molecules that are consistent across longitudinal samples (for sensor calibration) and estimates baseline levels of the molecules.
- PTIME peritoneal tumor immune microenvironment
- ABM Agent Based Model
- the PTIME-ABM cannot well approximate the real- world data. This can occur when there is insufficient existing knowledge to mechanistically link measurements. If additional knowledge cannot be acquired from literature or data, the present invention may develop multiple versions of the PTIME-ABM, each instantiating a “best guess” at a putative mechanistic link. In one embodiment, the present invention tests all the plausible versions of the ABM in subsequent tasks and narrow down to those achieving consistently better results.
- the present invention discloses wireless power transmitters and miniature receivers to recharge or continuously power bioelectronics implants that will be in use throughout the project. Demands on device size, form factor, and functionality change throughout the project as testing moves from rodents into large mammals.
- power circuitry is used to facilitate communication between the implant device and external devices.
- the present invention discloses a foundational circuitry and design for eventual power delivery and low data rate communication into and out of the peritoneal cavity, from implant device to external charging device (or another transmitter).
- the present invention discloses energy management, storage, and battery recharging circuitry.
- a major challenge for implanted bioelectronics is efficient power and data transmission through biological tissue.
- energy harvesting antennas must also be made small compared to the wavelength of the electromagnetic waves typically used to power biomedical devices. These sub -wavelength devices do not efficiently capture electromagnetic energy.
- the transmitter power must be increased; however, tissue absorbs electromagnetic energy at these frequencies placing strict limits of the transmitter power to avoid potentially harmful heating of the tissue.
- Ultrasound provides an alternative for wireless power; yet ultrasound suffers impedance mismatches between air and tissue, and thus ultrasonic gel is typically required, which would be cumbersome during extended use.
- the present invention discloses magnetoelectric (ME) materials enable wireless power delivery more than 8 centimeters beneath bone and tissue, which is possible because the present invention uses low-frequency magnetic fields that are not absorbed or reflected by the body. Importantly, this allows the present invention to effectively deliver power and communicate with miniature devices in the body.
- ME magnetoelectric
- ME materials are fabricated and packaged with custom PCBs (supporting receiver circuitry) for testing power transfer efficiency and data link.
- the ME materials are fabricated by laminating a commercial magnetostrictive layer (Metglas) with a commercial piezoelectric layer (PZT or PVDF). The laminated films are then be cut to size using a laser cutter and used for power and data communication.
- the data uplink is accomplished via backscattering the transmitter ME signal, meaning a single antenna and system can be used for both power, downlink, and uplink communication, reducing size and complexity and obviating the need for a bulky NFC or other inductive antenna, which may not be feasible due to the long and skinny device form factor.
- the present invention discloses alternative antenna form factors, including multiple smaller antennas in series/parallel along the length, or wrapping the end of the implant in multiple smaller antennas, and using intelligent impedance matching circuitry to balance power delivery efficiency.
- testing of data and power transfer are performed by placing the prototype carrier in a tissue phantom simulating the peritoneal cavity using SynDaver abdominal Tissue Plates surrounding a bladder with composite fluids (or similar tissue phantom) to simulate an implant in the cavity (an approximate distance of 4-6 cm from the wearable transceiver).
- total power transfer is measured by the maximum resistive load that can be driven by ME and PCB/supporting circuitry, and/or the maximum charging speed of an energy storage capacitor.
- the data link bandwidth is measured by the maximum data rate of correctly decoded downlink data by an onboard MCU, uplink is measured by the transmitting device.
- the dimensions of ME materials can be optimized and/or more ME films can be added in parallel to increase harvested power without requiring a higher field strength.
- the present invention assembles the cell therapeutics, oxygenator, sensors and bioelectronic control unit, with an Application Specific Integrated Circuit (ASIC) machine learning accelerator into a hybrid bioelectronic implant for rat and NHP validation.
- ASIC Application Specific Integrated Circuit
- the electrogenetically actuated cell therapeutic chambers are deployed in a 3D printed cage with the conductive scaffold.
- An oxygenator platform is integrated with sensing chips and the encapsulated bioelectronic control unit.
- the oxygenator end of the construct is inserted into the cell/stimulator component, and the conductive scaffold is physically wired to the control unit board with encapsulation.
- the control unit is hermetically sealed for water ingress to allow continuous performance in the IP space.
- the HAMMR platform is able to charge and communicate with a transmitter/hub device that is tethered and embeddable in undergarments or in a bed for ease of charging and nightly data offload.
- the HAMMR ASIC conducts onboard machine learning for signal compensation, as well as host the control algorithm for PK/PD aware therapy delivery.
- the present invention then uses local O2 production to maintain high cell loading viability and records the stability of desired production levels across different HAMMR compartments.
- the bioelectronic feedback control system of the present invention maintains drug production levels in rats, which are measured by the multichannel HAMMR sensing unit.
- the present invention leverages the NHP testbed (Celltrans) to verify functionality of the fully integrated HAMMR system across a range of operating conditions, environments, and levels of physical activity.
- the present invention uses a pre-existing system from the DARPA to support NHP testing: the fully integrated, battery powered external hub (extHub) are mounted in a primate jacket against the abdomen. The extHub only functions to deliver power wirelessly to the implant (to recharge it) and as a data download source. The extHub is replaced once a day to offload data and replace/recharge its batteries.
- the HAMMR device is implanted in the peritoneal cavity and beneath the extHub.
- the present invention optimizes the dimensions of ME materials and/or add more ME films in parallel to increase harvested power without requiring a higher field strength.
- the present invention adopts other protocols for high error and lossy channels if communication rate is low. The flexibility of the design concept- recharging and data offload at night along with battery implant size, can all be modulated to ensure ME power delivery is sufficient.
- HAMMR As the integrated HAMMR is readied for translation, contingencies and fail-safes should be considered: In the event that HAMMR is not charged by the user and the device battery charge falls below a threshold, the device is programmed to turn off production regardless of current state. Should the user require device termination, one can (a) surgically remove the device, and/or (b) administer a pharmacological safety switch technology used in DARPA NTRAIN into the cell constructs, or alternatively the cells are responsive to chemotherapy.
- the present invention employs comprehensive immune profiling of blood and metastatic sites by mass cytometry.
- the present invention applies Statistical Scaffold Maps to evaluate changes in immune repertoire and activation across all immune subsets within a tissue.
- the present invention evaluates long-term efficacy of the treatments.
- Human CD34+ hematopoietic stem cell engrafted NSG mice is inoculated with tumor cells as above, devices are implanted, and survival of mice are evaluated with a primary goal of an increase of >2.5-week median overall survival.
- the generation of memory phenotypes in T cells are evaluated by flow cytometry.
- the present invention while graft-versus-host disease can develop in Hu-PBMC mice 4 to 8 weeks following PBMC engraftment, the present invention conducts a long-term survival study in huCD34- NSG mice, which exhibit stable engraftment for over one year. If GVHD limits the studies in the Hu-PBMC mice, the present invention switches to using Hu-PBMC mice with MHC-I/II double knockout, which increases engraftment stability for > 75-100 days. It is possible that the liver metastasis model, while a common site of metastasis, will not predict systemic immune responses given its proximity to the device. Thus, the present invention has included lung metastasis models to ensure adequate evaluation of systemic responses outside of the peritoneum.
- samples of NHP studies proposed in phase 2 are used to establish the safety thresholds for the clinical trial in Phase 3.
- the present invention assesses dose limiting toxicity in NHPs using samples collected from NHP studies (ip fluid and blood) at various time points post therapy.
- the present invention collects omics data from longitudinal NHP blood/ascites samples as well as the in situ HAMMR biomarker data, including cytokine/soluble factor profiling (around 7 cytokines in the device design table and biosensor readouts), cytokine profiling using Lumitex, flow cytometry (additional immune abundance/function markers) and bulk RNA-seq data.
- Toxicity readouts from the animals are obtained, which include general health measurements such as liver function, body weight, etc., indicative of immune cell abundance/states and irAE.
- RNA-seq data is preprocessed, quantified, and deconvoluted to estimate tumor burden, clonality, and immune cell type and abundance.
- the present invention constructs a safety score out of the toxicity readouts and perform multivariate regression analysis, which allows establishing dosage limits for each molecule and identifying subsets of sensor markers most predictive of safety.
- the present invention validates and optimizes the PTIME- ABMs using the above omics data, for example, whether the molecular parameters in the PTIME- ABM is consistent with observations from the omics data. This exercise may also reveal additional cytokines/soluble-factors that could be better candidates to implement on the HAMMRs.
- the present invention trains Al under the deep reinforcement learning Q-leaming framework using data generated by the optimized PTIME-ABM. For each device, the present invention performs regression to establish the functional relationship between the safety/efficacy metrics and the biomarker measurements.
- the present invention discretizes continuous cytokine levels to define the states and modulatory actions, given the constraints and the on-device biomarkers and immunomodulators.
- the present invention tests a variety of Al models, such as deep neural networks, XGBoost trees, etc.
- the granularity of cytokine/biomarker level discretization may affect the accuracy of the ABM as well as model complexity.
- the present invention tests a variety of granularity levels and determines the ones that achieve good tradeoffs.
- the present invention directly learns a pharmacodynamic model using SNODE, which has demonstrated stability and ability to integrate sparse training data.
- the present invention also examines the PK/PD models developed in Sections 3 and 4. In the scenario that the DRL is not successful (e.g., very slow convergence), the present invention adopts model predictive control (MPC) and Lyapunov based control methods to derive the Al models.
- MPC model predictive control
- Lyapunov based control methods to derive the Al models.
- the present invention conducts a pilot clinical trial to determine the safety and feasibility of HAMMR armed with a human IL-2 therapeutic module implanted intraperitoneally (IP) in patients with advanced solid tumors.
- the primary endpoint is safety assessed by adverse event reporting by CTCAE v.5.0.
- the secondary endpoints consists of: 1) Comparison of ascites cytokine (including IL2) levels as determined serially by direct sampling versus as assessed by the sensing module of the HAMMR; 2) Dynamic immunological changes in peripheral blood and in the peritoneal environment assessed by serial collection and investigation of peritoneal fluid and cells using Luminex assays, flow cytometry, and scRNAseq; 3) Changes in the tumor (assessed by spatial transcriptomics and immune profiling); and 4) Objective response rate (ORR) and clinical benefit rate (CBR).
- the present invention enrolls a maximum of 12 patients in this pilot study at a rate of one subject per month. The subjects are divided into cohorts of 3 patients with increasing duration of device implantation for each cohort.
- the first cohort undergoes surgical implantation of the HAMMR for two weeks.
- the duration of device implantation increases by two weeks, such that subjects in cohort 4 have the device for eight weeks.
- Ascites and blood are collected weekly, and response is evaluated by RECIST v.1.1 based on imaging every four weeks. All subjects undergo surgical removal of the device after completing the prescribed duration of therapy. Following removal of HAMMR, subjects enter long-term follow-up with every eight- week imaging until progression.
- Summary statistics are used to describe the demographic and clinical characteristics of the study population.
- the incidence of AEs is described by grade and relation to study drug overall and by dose level.
- the DLT rate are estimated along with a 90% credible interval overall and by dose level.
- the present invention also estimates the posterior probability that the DLT rate is greater than 25%.
- All biomarker data i.e., IP fluid, blood samples, gene expression
- Descriptive statistics such as the mean, standard deviation, coefficient of variation, median, minimum, and maximum values, are provided for continuous endpoints.
- the rates of binary endpoints are provided along with corresponding 2-sided 95% Cis using an exact method.
- the ORR is estimated along with a 95% CI.
- DCR is estimated in a similar fashion.
- DCR is defined as ORR+ SD.
- Time-to-event endpoints are summarized using the Kaplan-Meier method and displayed graphically when appropriate. Median event times and 2-sided 95% Cis for each time-to-event endpoint is provided.
- PFS is defined as the time from initial treatment to the earliest disease progression or death date. In the absence of disease progression or death at the time of data cutoff, subjects' PFS times are censored at the date last assessed for progression.
- the present invention performs multivariate regression analysis to predict safety scores from the molecule measurements, establishing dosage limits for each molecule and identifying subsets of sensor markers most predictive of patient safety.
- the ABM and the Al models are evaluated and calibrated by human data and be used to predict dose-adjustment to assist clinical decision making, and to assess the potential of executing full-autonomous tasks.
- Embodiment 1 Cell Engineering
- the present invention optimizes Electro-cells (engineered cells) to meet a production threshold of ⁇ 1 pg/cell/day. By maximizing immunomodulator output, the present invention achieves physiological drug levels with a limited number of cells. Different construct designs are tested to find the system architecture that boosts production of individual immunomodulators. Expression of immunomodulators are tested in vitro over 10 days to ensure that expression is both sustainable and tunable (i.e., on/off controllable in response to electrical input) over 10 days in vitro in these engineered cells.
- the present invention perform mixed cell experiments in vitro with two different engineered Electrocells (each expressing a unique immunomodulator) and define optimal levels and ratios of mixed engineered Electro-cells for therapeutic use.
- different construct designs are tested, including the use of various synthetic promoters (e g., NF AT and it variants, CMV, CAG, EFS, or hUbC), plasmid architectures (delivery through individual vectors versus all-in-one delivery), and/or other genetic elements (e.g., polyA tails, Kozak sequences, nuclear export signals).
- the production features of each design are evaluated by measuring protein expression levels corresponding to the immunomodulator of interest.
- optimized architectures are tested using two lead immunomodulators (i.e., IL-2 and anti PD-1) and then ported to other immunomodulators.
- Figs. 15-16 show promotor construct designs for engineered Electro-cells to enable production of >1 pg/cell/day immunomodulator in vitro.
- the present invention demonstrates that engineering the promoter sequence of NF AT can increase IL2 production.
- Fig. 15 shows various promoter construct designs of NF AT, where the additions listed (AP-1, OCT, GATA, etc.) are genetic motifs added around NF AT, as listed in Table 1.
- the vector/plasmid has a NF AT promotor which includes a NF AT sequence alone, or a NF AT sequence together with one or more additional sequences of AP-1, OCT, GAT A, C/EBP, MAF, IRF4, MEF2, T-be, etc.
- the NF AT promotor comprises 1-10 copies of a NF AT sequence, or 1-10 copies of a combined sequence having a NF AT sequence together with one or more additional sequences of AP-1, OCT, GAT A, C/EBP, MAF, IRF4, MEF2, T-be, etc.
- Figs. 17A shows optimized plasmid designs for calcium channels, potassium channels, and the therapeutics, e g. IL2, and Fig. 17B shows 24-hr hIL production and hAnti- PD1 production after KC1 stimulation.
- the present invention discloses various avenues to increase production of desired cytokines from the electrogenetic cells.
- ARPE-19 cells were engineered to have channels that activate in response to electric/KCl stimulation, which when stimulated, cause a signal cascade to activate the NF AT promoter which drives the trancriptions of the gene of interest (GOI), e.g. IL2 and antibodies.
- GOI gene of interest
- ARPE-19 cells were engineered with different ratios of the plasmids containing the channel proteins and the plasmid containing the NF AT promoter with GOI.
- a 1 : 1 : 1 ratio of the 2 channel proteins and the reporter protein as shown in Fig. 17A, had the best on/off ratio of the conditions.
- the ratio between the 2 channel proteins ranges from 1 : 10 to 10: 1. In certain embodiments, the ratio of the channel proteins in total with the reporter protein ranges from 1 : 10 to 10: 1.
- the plasmids were introduced in the cells using “PiggyBac” method.
- the cells were engineered to secrete IL12 and antibodies (IgG), highlighting that the device of the present invention can be adopted for producing a large plethora of factors.
- the present invention increases cytokine production by the engineered cells in response to electrical stimulation specifically.
- ARPE-19 cells were engineered to have channels which activate in response to electric stimulation, and which, when stimulated, cause a signal cascade to activate the NF AT promoter which drives a transcription of the GOI.
- the present invention stimulated the cells with AC, and changed the distance between the two electrode from 1 to 2 to 3 mm.
- 3 mm electrode difference induces similar production of IL2 by the engineered cells as the KC1 stimulation. Therefore, 3 mm is an optimal distance between electrodes for this AC stimulation condition.
- the optimal distance between electrodes for this AC stimulation ranges between 2.5 mm - 3.5 mm. In certain embodiments, the optimal distance between electrodes for this AC stimulation ranges between 2 mm - 4 mm.
- Embodiment 2 Hybrid Advanced Molecular Manufacturing Regulator
- the present invention discloses a conductive scaffold that can electrically stimulate the engineered cells disposed therein, the present invention discloses optimal material for a 3D conductive scaffold for culturing the engineered cells and providing electrical simulation to the engineered cells.
- the present invention discloses designing and constructing conductive scaffolds which host the electrogenetic cells with or without their supporting biomaterial hydrogel, which are activated electrically using a high conductivity scaffold that can be integrated with both the cells and the electronics/wireless hardware.
- the scaffolds includes one or more of the following materials a hydrophilic base polymer, e.g., alginate, PEDOT:PSS incorporated into the alginate, graphene foams (commercially available), lithographically defined flexible metallic constructs, or Pt meshes.
- a hydrophilic base polymer e.g., alginate, PEDOT:PSS incorporated into the alginate, graphene foams (commercially available), lithographically defined flexible metallic constructs, or Pt meshes.
- Figs. 19A-B platinum, graphene foam, and PEDOT:PSS are three candidate for conductive scaffold. Their advantages and disadvantages, relative forms, and chemical formulas are shown in Fig. 19A and 19B, respectively.
- Fig. 20 shows that ARPE-19 cells seeded on the graphene condition had the lowest cell viability, with Pt and PEDOT:PSS demonstrating higher cell viability.
- Fig. 21 shows that PEDOT :PSS is less efficient than Pt for stimulating the cells electronically, because no IL2 production after the cells were culture on the PEDOT:PSS material and a current run through the material. That is, cell viability on stimulated PEDOT scaffolds was -25%, which shows high cell mortality correlates with hydrogen peroxide formation when a voltage is applied to PEDOT:PSS in an aqueous environment.
- Fig. 22 shows that the platinum material has both high production of IL2 and viability of cells when the electro-active ARPE-19 cells are cultured on it and stimulated thereon. That is, Pt is one of the optimal materials for the 3D conductive scaffold to house the cells in the final device.
- Fig. 23 shows photos of Pt conductive scaffold c, e.g., balls of platinum mesh seen in the top left. Confocal imaging was also used to confirm cells growing on the platinum conductive scaffold.
- the conducive scaffold is made of Pt.
- the conducive scaffold is made of Pt in combination with one or more other materials, e g. PEDOT:PSS, graphene, or other metals.
- Embodiment 3 Oxygenator
- the present invention discloses a device that generates oxygen, which is necessary for higher cell viability of the engineered electrically active ARPE- 19 cells disposed in the device.
- the present invention utilizes standard microfabrication and additive manufacturing (e.g., aerojet printing), and assembly techniques to build a flexible and scalable oxygen evolving catalytic platform, which are placed within the cylindrical mesh form to ensure uniform 3-dimensional spatial distribution of the generated oxygen.
- standard microfabrication and additive manufacturing e.g., aerojet printing
- assembly techniques to build a flexible and scalable oxygen evolving catalytic platform, which are placed within the cylindrical mesh form to ensure uniform 3-dimensional spatial distribution of the generated oxygen.
- microfabrication techniques include the synthesis and patterning of nanostructured catalysts (e.g., sputtered iridium oxide) in planar substrates
- additive manufacturing allows the present invention to fabricate 3-dimensional electrodes which are beneficial to construct hierarchical nanostructures.
- the mixture of nanoparticles formulating with catalytic (e.g., IrOx nanoparticles) and conductive components (e.g., platinum nanoparticle) can be printed with high resolution ( ⁇ 10 pm) and controlled porosities. Sintered at elevated temperature, the printed nanoparticles are fused together and form interconnected nanoporous catalytic electrodes.
- finite element analysis can be adopted to estimate the produced oxygen profiles, considering practical challenges in manufacturing processes.
- the oxygenator includes conductive components (e.g., Pt or nanocarbons), catalysts (e.g., iridium oxide) and structural supports.
- conductive components e.g., Pt or nanocarbons
- catalysts e.g., iridium oxide
- applied materials will be fine-tuned to accomplish long-term stability (at least 60 days).
- the present invention generates highly controlled oxygen gradients to support the bioactivities of loaded cells, and the produced oxygen allows maintenance of the viability of implanted therapeutic cells (>70%) and to manage tumor tissues chronically (at least 60 days).
- key metrics include: (1) oxygen production, (2) lack of byproduct formation, and (3) the maintenance of cell bioactivity/viability. Performance should be assessed as a function of time. Once the catalytic arrays are designed and fabricated, they are analyzed electrochemically.
- Fig. 24A shows different geometries and coatings of the oxygenator, as well as the resulting changes in the current vs voltage applied.
- the present invention discloses oxygenator having higher current with lower voltage end, as that allows the oxygenator to generate oxygen with a lower power consumption.
- the lower panel of Fig. 24A shows the different models of oxygenator.
- the oxygenator is a wire, either in a straight form or a coiled form.
- the oxygenator has a 6.6 mm diameter 11 turns Pt with Pt black deposition (Pt black) coiled form.
- the oxygenator has a 6.6 mm diameter 11 turns Pt coiled form.
- the oxygenator has a 4 cm Pt black straight form. In certain embodiments, the oxygenator has a 4 cm Pt straight form.
- the oxygenator has a straight wire form having a length between about 3.0 cm - 3.5 cm. In certain embodiments, the oxygenator has a straight wire form having a length between about 3.5 cm - 4.0 cm. In certain embodiments, the oxygenator has a straight wire form having a length between about 4.5 cm - 4.5 cm. In certain embodiments, the oxygenator has a straight wire form having a length between about 4.5 cm - 5.0 cm.
- the oxygenator has a coiled wire form having a diameter between 6.0-6.2 cm. In certain embodiments, the oxygenator has a coiled wire form having a diameter between 6.2-6.4 cm. In certain embodiments, the oxygenator has a coiled wire form having a diameter between 6 4-6.6 cm. In certain embodiments, the oxygenator has a coiled wire form having a diameter between 6.6-6.8 cm. In certain embodiments, the oxygenator has a coiled wire form having a diameter between 6.8-7.0 cm. In certain embodiments, the oxygenator has a coiled wire form having a diameter between 7.0-7.2 cm.
- Fig. 24B shows in the upper panel that the oxygenator is capable of generating oxygen below 0.9 V applied to the device. This is significant because a voltage above 0.9 V leads to generation of toxic byproducts (e.g. chlorine), which impact the viability of the engineered cells, as shown in the lower panel.
- toxic byproducts e.g. chlorine
- Fig. 25 provides a schematic diagram, showing the structure of the fabricated hybrid device containing the oxygenator, the engineered cells, and the conductive scaffold for electrically stimulating the engineered cells.
- the CE and WE are in a form of a mesh cylindrical structure as shown in Fig. 25.
- the shape and size of the mesh can be customized.
- the mesh is structured into a form other than cylindrical structure.
- the engineered cells are cultured in-between each CE and WE pair, and a current can be run between the CE and WE to stimulate the engineered cells, while the oxygenator locating in the center provides oxygen to the engineered cells.
- the oxygenator run through the center of concentric cylinders containing the counter electrodes (CE) and the working electrode (WE).
- the engineered cells are cultured in-between each CE and WE pair, and a current can be run between the CE and WE to stimulate the engineered cells, while the oxygenator locating in the center provides oxygen to the engineered cells.
- the WE comprises multiple mesh cylindrical structure WEs and the CE comprises multiple mesh cylindrical structure CEs.
- Multiple mesh cylindrical structure WEs and the multiple mesh cylindrical structure CEs are arranged alternatively at intervals and in a concentric arrangement.
- the Pt conductive mesh is modified to Pt black to increase the surface area.
- the implantable device has a length between about 3.0 cm -3.5 cm. In certain embodiment, the implantable device has a length between about 3.5 cm -4 cm. In certain embodiment, the implantable device has a length between about 4.0 cm -4.5 cm.
- the cell stimulation chamber within the implantable device has a length between about 1.4 cm -1.6 cm. In certain embodiments, the cell stimulation chamber within the implantable device has a length between about 1.6 cm -1.8 cm. In certain embodiments, the cell stimulation chamber within the implantable device has a length between about 1.8 cm -2.0 cm. In certain embodiments, the cell stimulation chamber within the implantable device has a length between about 2.0 cm -2.2 cm. In certain embodiments, the cell stimulation chamber within the implantable device has a length between about 2.2 cm -2.4 cm. [00275] In certain embodiments, the cell stimulation chamber within the implantable device has a diameter between about 0.8 cm - 0.9 cm.
- the cell stimulation chamber within the implantable device has a diameter between about 0.9 cm - 1.0 cm. In certain embodiments, the cell stimulation chamber within the implantable device has a diameter between about 1.0 cm - 1.1 cm. In certain embodiments, the cell stimulation chamber within the implantable device has a diameter between about 1.1 cm - 1.2 cm. In certain embodiments, the cell stimulation chamber within the implantable device has a diameter between about 1.2 cm - 1.3 cm.
- electrocatalytic oxygen evolution bioelectronics may be able to maintain both cellular viability and peptide-producibility with precisely controlled oxygen generation.
- electrocatalytic oxygen evolution bioelectronics may be able to maintain both cellular viability and peptide-producibility with precisely controlled oxygen generation.
- the capabilities of planar platforms to 3D spaces may be translated, interfacing with tissues in a “vasculature-like” manner.
- spatial and/or temporal oxygen distribution may be predicted and/or manipulated, enabled by simulation-driven geometry design.
- prestress in the design structures may be computed, which allows for anticipation of structural deformation and/or geometrical conversion for producing out-of-plane 3D flexible electronics.
- catalytic platforms may be able to form stable interfaces to tissues with minimized irritation.
- catalytic platforms may be introduced on compliant substrates including, but not limited to, polymeric substrates, thin metal wires and/or meshes.
- flexible devices may be integrated into tissues.
- the flexible platform may be fabricated as multi-side devices to support tissues/cells around the devices, without leaving oxygen-depleted areas.
- flexible 2-dimensional devices may map out 3-dimensional spaces with controlled pre-stress in the device construction.
- bioresorbability may facilitate surgical treatment and/or device implantation, since extra surgery may not be needed to remove implantation once objectives of the devices are accomplished.
- a flexible platform may be constructed utilizing additive manufacturing methods.
- the porosity of fabricated structures may be modulated by processing conditions including, but not limited to, processing temperature, annealing time, the size of contents and/or the formulation of precursors.
- 3D catalytic platforms may manage tissue-level of cell clusters in the aspects of high cell density as well as large scales beyond the limitation of diffusion from 2D-based systems. 3D catalytic platforms may have large interfacial areas with environments, which may support this feature.
- the tailored mechanical properties may allow formation of stable biointerfaces with various types of tissues.
- flexible and/or 3-dimensional oxygen supply devices may enable not only the modulation of physiological processes such as revascularization, cell migration and/or inflammatory responses, but also support large scale tissue with high cell density by integration as artificial vasculatures for oxygen delivery.
- Embodiment 4 Protein Engineering
- the present invention discloses engineered versions of naturally occurring cytokines and clinical antibodies for integration into the HAMMR platform.
- the selectivity and pharmacokinetic properties of these molecules are tailored to the specifications required in this system.
- the repertoire of naturally occurring proteins is finite, and many molecules induce multiple confounding effects, limiting their efficacy as therapeutics.
- the present invention implements a structure-based engineering approach to optimize the biochemical and biophysical properties of the cytokines and antibodies that are integrated into the HAMMR system. This interdisciplinary work at the interface of synthetic biology and protein engineering empowers the controlled delivery of immunomodulatory signals, both spatially and temporally.
- the present invention discloses engineering the properties of the interleukin-2 (IL-2) cytokine to bias its activities towards immune effector cells (i.e., CD4+ and CD8+ T cells and natural killer [NK] cells) and also engineer the constant domain of anti-CD40 agonist antibody to shorten its serum half-life and thereby restrict its local availability.
- IL-2 interleukin-2
- a combination of directed evolution and protein design technologies are employed to ensure that the engineered cytokines and antibodies incorporated in the HAMMR are suited for the system.
- signaling activity are assessed via activation assays carried on cytokine- responsive human NK cells. Phosphorylation of signal transducer and activator of transcription 5 (STAT5) are used as a readout for IL-2 signaling. Binding studies are conducted on an Octet® biolayer interferometry instrument to verify the interactions between the engineered cytokine and its cognate receptor subunits. The interaction of the engineered anti-CD40 antibody with the neonatal Fc receptor (FcRn) are also assessed through biolayer interferometry studies using an Octet® instrument to confirm that the antibody does not interact with FcRn and thus does not experience extended serum half-life.
- Phosphorylation of signal transducer and activator of transcription 5 STAT5
- Binding studies are conducted on an Octet® biolayer interferometry instrument to verify the interactions between the engineered cytokine and its cognate receptor subunits.
- Natural IL2 generally binds to 2 receptors: one on CD8 T-cells, and one on Regulator T-cells (T Reg). T Reg binding of IL2 does not have a significant biological affect, while CD8 T- cell binding does.
- the present invention discloses an engineered IL2 molecule that does not bind to the T Reg receptor, thus biasing the IL2 to bind to the CD8 T-cell receptor, which causes a greater biological effect.
- Fig. 26 shows a summary of constructs for the engineered IL2 in the upper panel.
- LN35 and LN15 are peptide linkers of either 35 amino acids or 15 amino acids in length.
- hIL-2 is human IL2, and the Vh and VI components are parts of an antibody. These antibody components are intended to sterically block the IL2 from binding to the T Reg receptor.
- Fig. 26 lower panel shows the receptors of the T Reg and the CD8 T-cells (effector).
- the IL-2Ralpha component is the component causes the steric hinderance on the receptor side.
- the present invention discloses biochemical sensors that can resist or reduce biofouling over 60 days in complex tissue environments.
- Biofouling is a major concern for development of implantable devices, especially with functional devices where long term performance requires calibration-free measurements.
- the present invention discloses fouling-resist biosensors via multiple grounds.
- the present invention designs a “housekeeping” sensor (designed not to bind, or one that binds a housekeeping molecule) and locate it in proximity to the working sensor that will account for the systematic drift caused by long-term implantation.
- a BSA-bound DNA (similar to a scrambled aptamer) does not bind a target from biofluids, and can be used as an internal calibrant in the sensing system to account for drift and can play this role as a “control”.
- the present invention builds sensors on nanostructured electrodes that are less prone to fouling due to their function to prevent cell surface adhesion.
- the present invention optimizes sensor longevity as a function of surface roughness by building micro-electrodes (dome, star/dendrite, cube shapes) decorated with nanostructures (pores carved out in nanoscale) using current potential amperometry (DCPA) to control electrode morphology.
- DCPA current potential amperometry
- the present invention assembles a library of surface coating molecules as self- assembled monolayers (SAM) spacers (e.g., Fluorosilane, bromotrimethylsilane, Zwitterion, and 8-mercapto-l -octanol (MCO)) that can help prevent fouling and examines their effectiveness in comparison to conventional spacers that are prone to bio-fouling and desorption
- SAM self- assembled monolayers
- the present invention incorporates polymer coatings or polymeric micro scaffolds with optimized pore size to allow for target penetration.
- Microelectrode array (MEA) with planar structure is fabricated using standard microfabrication techniques and will be used as sensors. Before applying the anti-fouling coating, the MEA of working electrode sensors are modified with specific biorecognition elements and backfilled with the spacer molecule mentioned above.
- the present invention discloses aptamer-based sensors to sense targets relevant to peritoneal cancer. This particular sensor being testing is for sensing Albumin, which is being used as a housekeeping gene.
- the aptamer sensors are engineered to sense other targets relevant to cancer as well, such as TNF-a.
- Fig. 29B shows the same sensor placed in a solution of increasing concentrations of albumin. At -1000 micrograms/mL the sensor begins to detect the albumin, evident by the change in the normalized current (y-axis). A lower frequency applied typically results in a lower signal as the concentration increases, while a 60 or 120 frequency applied results in a higher signal as the concentration increases. This result is expected, and the delta between baseline normalized current and the normalized current when the sensor senses albumin is the important metric which demonstrates proper function of the aptamer.
- the present invention discloses a method to mitigate this degradation effect by coating the electrodes used in the aptamer-based sensors.
- Fig. 30 shows the change in current density over times with different chemicals functionalized to the electrode surfaces.
- the ideal electrode demonstrates a stable current density over time. If the current density drops, it suggests that the electrode is unstable.
- the aptamer-based sensor has a coating of MUO-MCH. In certain embodiments, the aptamer-based sensor has a coating of MUO-TA. In certain embodiments, the aptamer-based sensor has a coating of MHx3-MPCl. In certain embodiments, the aptamer-based sensor has a coating of one of MPO, MCH, MPC, Z1A3, MHX:MPC (ratio 3: 1), BG53, BG42, MBO, MUO, TA, Z4A10, MUO & TA, MCH55, BG53, BG44.
- the aptamer-based sensor has a coating of a mixture comprises at least two of MPO, MCH, MPC, Z1A3, MHX:MPC (ratio 3:1), BG53, BG42, MBO, MUO, TA, Z4A10, MCH55, BG53, BG44.
- Embodiment 6 Electronic Hardware of Integrated Component
- Figs. 31-32 represent the electronic device which integrates all electronic components of the present invention.
- the electronic device integrating the electronic components including the aptamer-based sensors, the oxygenator, and the electronic stimulator for the cells.
- This integration allows the communication between the electronic components, i.e. sensing the dose of the cancer therapeutics with the aptamer-based sensors, and then relay the dose information to the stimulator to either start stimulating the engineered cells for producing a specific dose of cancer therapeutics depending on the dose was sensed, or stop the cancer therapeutics production.
- Fig. 31 shows a photograph of biosensor readout platform which has been implemented as a vertical stack for modularity.
- the biosensor readout platform has dimensions of 2 cm x 2 cm x 1.5 cm.
- the biosensor readout platform has a width between about 1-3 cm.
- the biosensor readout platform has a length between about 1-3 cm.
- the biosensor readout platform has a depth between about 1-3 cm.
- the biosensor readout platform comprises multiple PCBs. In certain embodiments, the biosensor readout platform comprises a single PCB.
- the electronic device includes a wireless communication component to provide data of the therapeutics and patient’s response to the therapeutics to an external hub, which shows the data the doctor/patient.
- the wireless communication module also allows for wireless charging of a battery on the electronic device.
- Embodiment 7 Software for Integrated Hardware
- the present invention discloses a software which is used in association with the electronic device.
- the software collects the data from the aptamer-based sensors, computes the amount and the dose of the cancer therapeutics needed, and relay this information to the electrical stimulator/conductive scaffold to turn the engineered cells on to secrete the cancer therapeutics at the needed amount and dose.
- the software assists modeling the aptamer-based sensor decay and correcting for this decay.
- Fig. 33 shows a schematic workflow process of the entire electronic device.
- the left side of Fig. 33 shows the software used by the controller for controlling therapeutics delivery and sensor degradation compensation, and power consumption control.
- Fig. 34 shows the degradation correction model that corrects for the natural decay in the aptamer-based sensors.
- the degradation correction model includes a polynomial model, which provides high accuracy and efficiency.
- the degradation correction model includes a neural network model which provides high generalizability.
- the degradation correction model combines the polynomial model and the neural network model, which allows for accurate prediction while reducing the need to calibrate and lowering the battery power consumption.
- the FPGA is the hardware to house the software of Figs. 33- 34.
- Fig. 36 shows a prototype of the FPGA board and its setup, where the “upstream machine” is simulating input into the FPGA (i.e. information obtained by an aptamer-based sensor), and the output of the FPGA is being read by the “logic analyzer” so as to determine that the FPGA is correctly accounting for the modeled degradation of the aptamer-based sensor.
- the “upstream machine” is simulating input into the FPGA (i.e. information obtained by an aptamer-based sensor)
- the output of the FPGA is being read by the “logic analyzer” so as to determine that the FPGA is correctly accounting for the modeled degradation of the aptamer-based sensor.
- Embodiment 8 Humanized Cancer Model Data
- the present invention demonstrates that the cytokines produced by the engineered ARPE-19 cells can effectively treat cancer.
- the present invention uses humanized mouse models, in which human immune cells are grafted into the mice. This allows the present invention to get as close to a human immune system in a mouse model as possible.
- Figs. 38A-B show immune cell populations of the mice. Most notable is that the delivered IL2 increases CD8+ T-cells, a T-cell known to be beneficial in fighting tumors.
- the tumors were injected into the lymph nodes of the mice instead of directly into the intraperitoneal space. This is intended to model metastasis cancer.
- the present invention uses ARPE-19 cells that are engineered to secrete cytokines constitutively, rather than in response to electrical stimulation, to allow for ease of testing.
- Fig. 39 shows the timeline of the mouse studies, where the mice are first injected with a cancer cell line, either ovarian, SKOV3 and IGROV, or pancreatic, AsPC-1 and BxPC-3, then engrafted with human immune cells (PBMC injection), followed by implantation with the ARPE-19 cells engineered to secrete IL2 encapsulated in alginate, by surgery.
- a cancer cell line either ovarian, SKOV3 and IGROV, or pancreatic, AsPC-1 and BxPC-3
- PBMC injection human immune cells
- ARPE-19 cells engineered to secrete IL2 encapsulated in alginate
- the tumors are luminescent, so their size can be tracked via flux.
- Fig. 39 shows that cytokine treatment can effectively reduce tumor burden in AsPC-1 tumor mice (middle left panel).
- Fig. 40 shows that cytokine treatment can effectively reduce tumor burden in SKOV3 tumor mice (middle left panel).
- Fig. 41 shows immune cell composition across the mouse conditions. There are no consistent changes across conditions, suggesting that the immune response responsible for treatment of the cancer is not in the set of immune cells analyzed here.
- Embodiment 9 Cancer Omics Data
- the present invention discloses a method to analyze patient ascites data with omics to identify specifics cytokines/proteins that can be used as biomarkers of the cancer state. Any identified cytokines/proteins can be used as biomarkers for being sensed by the aptamer-based sensors, and therefore providing information regarding toxicity and efficacy of the cancer therapeutics delivered.
- a scoring system is used to determine which cytokines/proteins are clinically relevant biomarkers for sensing the toxicity and efficacy of the cancer therapeutics.
- the scoring system comprises three parts: a detection score, a clinical score, and a signaling score.
- the biomarker should have: (a) absolute difference between partial response (PR) and stable disease (SD) patients at baseline (C0D1); (b) absolute difference after combination therapy treatment (C3D1) versus at baseline (C0D1); (c) absolute difference between PR and SD after treatment; (d) absolute difference in change between PR and SD after treatment.
- PR partial response
- SD stable disease
- the present invention screened 147 molecules in total, with 80 from Luminex assay; 17 from Cytosig database, and 50 from CellChat secretory ligand database.
- Table 2 shows the top 30 molecules with highest total scores when analyzing ascites from ovarian cancer patients.
- Fig. 42 shows a chart reflecting these molecule on the signaling and clinical score for ease of comparison and visualization.
- Galectin-3, MIF, IL-8, IL-2R, VEGF-A, TWEAK, APRIL, and PTX3 were all identified as good candidate molecules for sensing.
- Fig. 43 shows an additional metric that was added to the 3 scores described above.
- this additional metric is using a large language model to predict the effects of various cytokines, e g. clinical relevance and functional effect from literature databases.
- Fig. 43 demonstrates how the model work: (1) the cytokines analyzed are searched for on literature databases, e.g. PubMed, (2) the top 30 relevant papers for each cytokine are retrieved, and (3) associations are extracted from the manuscripts to determine the role the cytokines as the tumor progresses.
- Fig. 44 shows the data generated by the large language model, and the important sensing biomarkers identified. MIF, MCP-1, and IL-6 were the top molecules suggested by the model that would be important biomarkers for being sensed.
- one of Galectin-3, MIF, IL-8, IL-2R, VEGF-A, TWEAK, APRIL, PTX3, MIF, MCP-1, and IL-6 is sensed by the aptamer-based sensor for determining the toxicity and/or efficacy of the cancer therapeutics.
- a combination of any of Galectin-3, MIF, IL-8, IL-2R, VEGF-A, TWEAK, APRIL, PTX3, MIF, MCP-1, and IL-6 is sensed by the aptamer-based sensor for determining the toxicity and/or efficacy of the cancer therapeutics.
- Embodiment 10 Mathematics and Computation Modeling of the Electronic Device
- the present invention discloses an overall mathematical model of the engineered ARPE-19 cells in the stimulation environment. This overall mathematical model assist the controlled of the electronic device to determine the degree of the stimulation for the engineered cells to produce the desired amount / dose of the cancer therapeutics.
- Fig. 45 depicts an overview of the mathematical model.
- the left side shows a finite element/electrophysiology model relating stimulus to membrane depolarization, which is a physics-based model.
- the right side shows a cell signaling model for relating membrane depolarization and protein production, which reflects the processes/reactions took places inside the engineered ARPE-19 cells after the cells are electrically stimulated, starting with the electroactive channels opening, and ending with cytokine secretion.
- Figs. 46A-B show that this mathematical model predicted that increasing NF AT expression inside the cells will in turn increase production of cytokines from the engineered ARPE-19 cells.
- Fig. 47 shows that the predicted production by the mathematical model was correct.
- the upper panel shows that increasing NFAT3 in the engineered ARPE-19 cells does result increased production of IL2 by engineered ARPE-19 cells, while increasing the other NFATs, NF ATI and NFAT2, does not increase cellular production, suggesting NFAT3 is the most important.
- the mathematical model also predicted that a 3 mm distance between electrodes for stimulation would result in the greatest stimulation of the engineered ARPE-19 cells, by comparing the bottom right chart with the top left chart. This prediction was validated in Fig. 18B, with 3 mm distance between the electrodes results in the greatest production from the cells.
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Engineering & Computer Science (AREA)
- Animal Behavior & Ethology (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- General Health & Medical Sciences (AREA)
- Biomedical Technology (AREA)
- Heart & Thoracic Surgery (AREA)
- Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Surgery (AREA)
- Medical Informatics (AREA)
- Physics & Mathematics (AREA)
- Pathology (AREA)
- Biophysics (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Pharmacology & Pharmacy (AREA)
- Molecular Biology (AREA)
- Anesthesiology (AREA)
- Hospice & Palliative Care (AREA)
- Oncology (AREA)
- Hematology (AREA)
- Radiology & Medical Imaging (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
- Medicines Containing Material From Animals Or Micro-Organisms (AREA)
Abstract
A platform of a hybrid advanced molecular manufacturing regulator device containing engineered cells delivering cancer therapeutics to a subject comprises an implantable device disposed inside an internal cavity of a subject; and an external device disposed outside the internal cavity of the subject and in wireless communication with the implantable device, wherein the implantable device comprises a molecular manufacturing regulator and a communication module; wherein the external device comprises at least one transmission coil in wireless communication with the communication module.
Description
PLATFORM OF HYBRID ADVANCED MOLECULAR MANUFACTURING
REGULATOR AND APPLICATIONS THEREOF
STATEMENT AS TO RIGHTS UNDER FEDERALLY-SPONSORED RESEARCH
[0001] This invention was made with government support under FA8650-21-2-7119 awarded by the Air Force Research Laboratory. The government has certain rights in the invention.
CROSS-REFERENCE TO RELATED APPLICATIONS
[0002] This application claims priority to U.S. Provisional Patent Application Serial No. 63/540,004 filed on September 22, 2023, and U.S. Provisional Patent Application Serial No. 63/539,658 filed on September 21, 2023. The disclosures of each of these applications are incorporated herein by reference.
[0003] This application is also a continuation-in-part of PCT application No. PCT/US23/36276 filed on October 30, 2023, which further claimed its priority to U.S. Provisional Patent Application Serial No. 63/420,219 filed on October 28, 2022 and U.S. Provisional Patent Application Serial No. 63/545,996 filed October 27, 2023. PCT application No. PCT/US23/36276 was also a continuation-in-part of PCT applications PCT/US22/25686, PCT/US22/25706, and PCT/US22/25724 filed on April 21, 2022, which further claimed priority of U.S. Provisional Patent Application Serial No. 63/177,806 filed on April 21, 2021. The disclosures of each of these applications are also incorporated herein by reference.
[0004] This application is also a continuation-in-part of U.S. Patent Application Serial No. 18/287,671, U.S. Patent Application Serial No. 18/287,684, and U.S. Patent Application Serial No. 18/287,709, filed on October 20, 2023, which are U.S. national stage applications of PCT applications PCT/US22/25686, PCT/US22/25706, and PCT/US22/25724 filed on April 21, 2022, respectively. The disclosures of each of these applications are also incorporated herein by reference.
[0005] This application is also related to co-pending PCT patent applications, entitled “Hybrid Advanced Molecular Manufacturing Regulator and Applications Thereof’ with Attorney Docket No. 0116936.297WO22, and “Engineered Cells Producing Oncotherapeutics and Applications Thereof’ with Attorney Docket No. 0116936.297WO23, respectively, which
are filed on the same day that this application is filed, and with the same applicant as that of this application. The disclosures of each of these applications are also incorporated herein by reference.
SEQUENCE LISTING
[0006] A Sequence Listing conforming to the rules of WIPO Standard ST.26 is hereby incorporated by reference. Said Sequence Listing has been filed as an electronic document via PatentCenter encoded as XML in UTF-8 text. The electronic document, created on September 20, 2024, is entitled “0116936.297WO24. xml”, and is 15,138 bytes in size.
FIELD OF THE INVENTION
[0007] The present disclosure relates generally to the field of biomedical engineering, and more particularly to a platform for a hybrid advanced molecular manufacturing regulator in wireless communication with an external device, and applications of the same.
BACKGROUND OF THE INVENTION
[0008] The background description provided herein is for the purpose of generally presenting the context of the present invention. The subject matter discussed in the background of the invention section should not be assumed to be prior art merely as a result of its mention in the background of the invention section. Similarly, a problem mentioned in the background of the invention section or associated with the subject matter of the background of the invention section should not be assumed to have been previously recognized in the prior art. The subject matter in the background of the invention section merely represents different approaches, which in and of themselves may also be inventions.
[0009] Cancer immunotherapy represents perhaps the most important breakthrough in the field of oncology in the past half century, exhibiting the ability to induce durable responses even in patients with advanced-stage malignancies. These therapies fall into two categories: cell therapies and protein-based immunostimulatory agents. Cell therapies encompass approaches including adoptive transfer of ex vivo expanded autologous tumor- infiltrating lymphocytes (TILs), dendritic cell vaccines, and a range of engineered immune receptor lymphocytes such as transgenic TCR T cells, chimeric antigen receptor (CAR) T cells, and CAR-NK cells. While
these approaches have yielded robust cures in a subset of hematological malignancies, they have yet to demonstrate durable responses in any solid organ malignancy. Protein-based immunotherapies include immune checkpoint blockade (ICB) antibodies, vaccine, or administration of natural or engineered immunostimulatory cytokines. While ICB represents the greatest advance in solid tumors in decades, eliciting cures in a subset of patients with stage IV advanced cancers, most patients do not experience durable responses and many cancers, including most peritoneal malignancies, are unresponsive to ICB. Treatment with exogenous cytokines, such as high-dose IL-244 and IL-12, were among the first approaches in the field and elicited responses in a small subset of melanoma and renal cell carcinoma patients. Yet systemic administration of these cytokines results in severe dose-limiting toxicity, and both cytokine therapy and ICB consequently induce a range of immune-related adverse events (iRAEs). The ability of these therapies to elicit systemic anti-tumor immune responses is essential for longterm efficacy, yet the ability to do so while avoiding systemic toxicity has been the major roadblock for these approaches. Furthermore, while significant amount of investment has been spent in cancer immunotherapies, the majority of the investment has been dedicated to further development of these existing approaches, including additional cell therapies, cytokines, new checkpoints, and combination immunotherapies, none of which address the challenge of generating systemic immunity while avoiding toxicity.
[0010] For example, most patients (85-95%) with solid tumor malignancy do not fully respond to current immunotherapy approaches due to inadequate activation in secondary lymphoid organs. The generation of systemic immunity within such sites at levels required for effective immune responses frequently results in dose-limiting toxicity for the current immunotherapy treatment modalities.
[0011] Therefore, a heretofore unaddressed need exists in the art to address the aforementioned deficiencies and inadequacies.
SUMMARY OF THE INVENTION
[0012] In light of the foregoing, this invention discloses a platform of a hybrid advanced molecular manufacturing regulator containing engineered cells delivering cancer therapeutics to a subject comprises an implantable device disposed inside an internal cavity of a subject; and an external device disposed outside the internal cavity of the subject and in wireless communication
with the implantable device, wherein the implantable device comprises a molecular manufacturing regulator and a communication module; wherein the external device comprises at least one transmission coil.
[0013] In one embodiment, the communication module comprises at least one antenna.
[0014] In one embodiment, the at least one antenna comprises a magnetoelectric (ME) component.
[0015] In one embodiment, the ME component comprises at least one magnetostrictive layer attached to a piezoelectric layer.
[0016] In one embodiment, the at least one magnetostrictive layer comprises multiple magnetostrictive layers.
[0017] In one embodiment, the at least one antenna is in wireless communication with the at least one transmission coil.
[0018] In one embodiment, the at least one antenna is configured to receive a power transmission from the at least one transmission coil.
[0019] In one embodiment, the implantable device comprises a battery, wherein the battery is charged by the power transmission.
[0020] In one embodiment, the at least one antenna is configured to receive a download information transmission from the at least one transmission coil.
[0021] In one embodiment, the at least one transmission coil is configured to receive an upload information transmission from the at least one antenna.
[0022] In one embodiment, the at least one antenna comprises multiple sub-antennas.
[0023] In one embodiment, each of the multiple sub-antennas are configured to receive the power transmission and the download information transmission, and provides the upload information transmission to the at least one transmission coil.
[0024] In one embodiment, the at least one transmission coil comprises a power transmitter coil and a receiver pick-up coil.
[0025] In one embodiment, the at least one antenna is configured to receive a power transmission from the power transmitter coil.
[0026] In one embodiment, the receiver pick-up coil is configured to receive an upload information transmission from the at least one antenna, and the at least one antenna is configured to receive a download information transmission from the receiver pick-up coil.
[0027] In one embodiment, the molecular manufacturing regulator comprises a conductive scaffold in communication with a control module; a biochemical sensor module in communication with the control module; an oxygenator configured to produce oxygen and disposed inside the conductive scaffold; and at least one engineered cells encapsulation disposed in association with the conductive scaffold; wherein the at least one engineered cells encapsulation encapsulates engineered cells; wherein the engineered cells are configured to produce at least one cancer therapeutic agent.
[0028] In one embodiment, the conductive scaffold is configured to produce an electrical pulse, wherein the conductive scaffold is powered by the battery.
[0029] In one embodiment, the control module comprises a machine learning algorithm for optimizing at least one of the power transmission, the upload information transmission, and the download information transmission.
[0030] In one embodiment, the implantable device comprises at least one of a field- programmable gate array (FPGA) circuit, an application specific integrated circuit (ASIC), and a microcontroller circuit; wherein the at least one of the FPGA, the ASIC, and the microcontroller circuit coordinates and controls an electrical stimulation of the conductive scaffold which is optimized for a production efficiency of the at least one cancer therapeutic agent and/or a longevity of battery.
[0031] In one embodiment, the implantable device is powered by a primary cell battery.
[0032] In one embodiment, the primary cell battery is a lithium-based battery configured to provide a long-term power to the implantable device.
[0033] In one embodiment, the communication module of the implantable device comprises a Bluetooth communication module for wireless transmission to the external device.
[0034] In one embodiment, the communication module is configured to transmit and receive data related to an operational status of the molecular manufacturing regulator.
[0035] In one embodiment, the communication module of the implantable device incorporates a proprietary radio protocol for wireless communication with the external device. [0036] In one embodiment, the proprietary radio protocol is configured to transmit and receive data related to the operational status of the molecular manufacturing regulator.
[0037] In one embodiment, the proprietary radio protocol is optimized for low-power operation to extend a life of a primary cell battery powering the implantable device.
[0038] In one embodiment, a primary cell battery is configured to power both the molecular manufacturing regulator and the communication module.
BRIEF DESCRIPTION OF THE DRAWINGS
[0039] The accompanying drawings illustrate one or more embodiments of the invention and together with the written description, serve to explain the principles of the invention. Wherever possible, the same reference numbers are used throughout the drawings to refer to the same or like elements of an embodiment.
[0040] Fig. 1 illustrates a Hybrid Advanced Molecular Manufacturing Regulator (HAMMR), related components and its application.
[0041] Fig. 2 illustrates implementation and feedback of the HAMMR which is implanted in the intraperitoneal space through a small incision.
[0042] Fig. 3 illustrates a list of the target therapeutics for the HAMMR.
[0043] Fig. 4 illustrates light induced cytokine secretion from engineered ARPE-19 cells according to one embodiment of the present invention.
[0044] Fig. 5A illustrates a design and function of engineered cells, in particular, the electrogenic processes and device.
[0045] Fig. 5B illustrates the design and testing of various construct designs of the engineered cells.
[0046] Fig. 6 illustrates an embodiment of printing conducting polymer composites.
[0047] Fig. 7 illustrates microscopic images showing immunoisolating chamber and chemical coatings to protect against foreign body responses.
[0048] Fig. 8 illustrates that ecCh arrays using sputtered IrCh (SIROF) catalyst are able to support the bioactivities of high- cell density capsules (60k cell mm-3) up to 21 days under hypoxic environment (1 % O2) as well as implanted in rodent for 10 days.
[0049] Fig. 9 illustrates oxygenators of HAMMR and its application.
[0050] Fig. 10 illustrates a schematic diagram of general model structure.
[0051] Fig. 11 illustrates electrode engineering and performance of nano- structured electrode.
[0052] Fig. 12 illustrates effect of bias stress on sensor stability.
[0053] Fig. 13 illustrates a dual mode onboard NN engine for heart anomaly detection (top),
and ultra small ME implant receiver.
[0054] Fig. 14 illustrates models of tumor micro-environment and is to assist the design and control.
[0055] Fig. 15 illustrates promoter construct designs for engineered Electro-cells to enable production of >1 pg/cell/day immunomodulator in vitro.
[0056] Fig. 16 illustrates a chart reflecting hIL2 production of Electro- ARPE- 19 cells engineered with promoter construct designs stimulated with 80 mM KC1.
[0057] Fig. 17A illustrates an optimized construct design for engineered Electro-cells.
[0058] Fig. 17B illustrates charts reflecting 24-hr hIL2 production after KC1 stimulation and 24-hr hAnti-PDl production after KC1 stimulation by the Electro-ARPE-19 cells.
[0059] Fig. 18A illustrates a workflow of in vitro activation of Electro-cells with selected scaffold material at >1 pg/cell/day.
[0060] Fig. 18B illustrates a chart reflecting 24-hr hIL2 production by Electro-ARPE-19 cells after AC stimulation.
[0061] Fig. 18C illustrates a chart reflecting 24-hr live cell counts (LCC) of Electro -ARPE- 19 cells after AC stimulation.
[0062] Fig. 18D illustrates a chart reflecting 24-hr % viability of Electro -ARPE-19 cells after AC stimulation.
[0063] Fig. 19A illustrates a down- sei ection of conducting scaffold material considering performance, stability, cell viability, and scalability.
[0064] Fig. 19B illustrates chemical formulas of PDL, Fibronectin, Laminin, and RGD.
[0065] Fig. 20 illustrates a selection of conducting scaffold material considering cell viability, particularly, PEDOT:PSS, Graphene, Platnium.
[0066] Fig. 21 illustrates a selection of conducting scaffold material considering performance, particularly, IL-2 and H2O2 production on PETOD:PSS.
[0067] Fig. 22 illustrates a selection of conducting scaffold material considering performance, stability, cell viability, and scalability, particularly, IL-2 and H2O2 production on Pt.
[0068] Fig. 23 illustrates a photo of the conducting scaffold using the selected materials (Pt) and confocal images reflecting the cells growing on the conducting scaffold.
[0069] Fig. 24A illustrates a chart reflecting surface area of Pt counter electrode of the
oxygenator (upper panel) and several oxygenator models designed with different Pt (n=3) (lower panel).
[0070] Fig. 24B illustrates charts reflecting wire oxygenator oxygen measurement (n=l) (upper panel) and wire oxygenator byproduct measurements (lower panel).
[0071] Fig. 25 illustrates a schematic diagram of conducting scaffold with the oxygenator.
[0072] Fig. 26 illustrates a diagram structure of three engineered IL-2s (upper panel) and a
3D structure of engineered !L-2(lower panel).
[0073] Fig. 27 illustrates charts reflecting immunostimulatory bias of engineered IL-2 via STAT5 signaling and biolayer interferometry-based receptor binding studies.
[0074] Fig. 28 illustrates charts reflecting IL-2 effector stimulation bias in a quantitative form.
[0075] Fig. 29A illustrates charts reflecting an aptamer sensor exposed to ascites from cancer patients, and PBS as control.
[0076] Fig. 29B illustrates charts reflecting an aptamer sensor in a solution of increasing concentrations of albumin.
[0077] Fig. 30 illustrates charts reflecting down selecting library of surface coating and electrode modification and select/confirm enhanced lifetime of electrode over conventional methods.
[0078] Fig. 31 illustrates a photograph of a biosensor readout platform.
[0079] Fig. 32 illustrates a CAD image of the design of the electronic device integrating all electronic components.
[0080] Fig. 33 illustrates a schematic workflow process of the entire electronic device.
[0081] Fig. 34 illustrates a degradation correction model that corrects the natural decay in the aptamer-based sensors.
[0082] Fig. 35 illustrates a block diagram of initial signal compensation scheme, validation test bench setup for FPGA systems.
[0083] Fig. 36 illustrates a prototype of the FPGA setup.
[0084] Fig. 37A illustrates a humanized murine tumor model timeline.
[0085] Fig. 37B illustrates images of tumor measured by IVIS imaging.
[0086] Fig. 37C illustrates charts reflecting total flux and body weights.
[0087] Fig. 38A illustrates charts reflecting flow cytometry results of CD45+, NK cells, and
T cells for spleen harvested in BxPC3 humanized model.
[0088] Fig. 38B illustrates a timeline for immune cell populations of the mice.
[0089] Fig. 39 illustrates charts evaluating the efficacy of human IL12 cytokine factories in treating metastatic tumor model of PDAC lines.
[0090] Fig. 40 illustrates charts evaluating the efficacy of human IL 12 cytokine factories in treating metastatic tumor model of OC lines.
[0091] Fig. 41 illustrates charts reflecting the immune cell composition across the mouse conditions.
[0092] Fig. 42 illustrates a chart reflecting which cytokines/proteins which are clinically relevant sensor targets.
[0093] Fig. 43 illustrates a flowchart of a large language model to predict the effects of various cytokines.
[0094] Fig. 44 illustrates the data generated by the large language model of Fig. 43.
[0095] Fig. 45 illustrates a flowchart of a mathematical model for predicting protein production as a function of voltage applied.
[0096] Fig. 46A illustrates that the mathematical model of Fig. 45 predicted that increasing NF AT expression inside the cells induces increased production of cytokines by the cells.
[0097] Fig. 46B illustrates charts reflecting increasing NFAT3 in the cells does result increased production of IL2 by the cells.
[0098] Fig. 47 illustrates a verification of the prediction of Fig. 46A with in vitro Protein Production Data.
[0099] Fig. 48 illustrates that the mathematical model of Fig. 45 predicted that a 3 mm distance between electrodes for stimulation would result in the greatest stimulation of the cells.
DETAILED DESCRIPTION OF THE INVENTION
[00100] The invention will now be described more fully hereinafter with reference to the accompanying drawings, in which exemplary embodiments of the invention are shown. This invention may, however, be embodied in many different forms and should not be construed as limited to the embodiments set forth herein. Rather, these embodiments are provided so that this invention will be thorough and complete, and will fully convey the scope of the invention to those skilled in the art. Like reference numerals refer to like elements throughout.
[00101] The terms used in this specification generally have their ordinary meanings in the art, within the context of the invention, and in the specific context where each term is used. Certain terms that are used to describe the invention are discussed below, or elsewhere in the specification, to provide additional guidance to the practitioner regarding the description of the invention. For convenience, certain terms may be highlighted, for example using italics and/or quotation marks. The use of highlighting has no influence on the scope and meaning of a term; the scope and meaning of a term is the same, in the same context, whether or not it is highlighted. It will be appreciated that same thing can be said in more than one way. Consequently, alternative language and synonyms may be used for any one or more of the terms discussed herein, nor is any special significance to be placed upon whether or not a term is elaborated or discussed herein. Synonyms for certain terms are provided. A recital of one or more synonyms does not exclude the use of other synonyms. The use of examples anywhere in this specification including examples of any terms discussed herein is illustrative only, and in no way limits the scope and meaning of the invention or of any exemplified term. Likewise, the invention is not limited to various embodiments given in this specification.
[00102] One of ordinary skill in the art will appreciate that starting materials, biological materials, reagents, synthetic methods, purification methods, analytical methods, assay methods, and biological methods other than those specifically exemplified can be employed in the practice of the invention without resort to undue experimentation. All art-known functional equivalents, of any such materials and methods are intended to be included in this invention. The terms and expressions which have been employed are used as terms of description and not of limitation, and there is no intention that in the use of such terms and expressions of excluding any equivalents of the features shown and described or portions thereof, but it is recognized that various modifications are possible within the scope of the invention claimed. Thus, it should be understood that although the invention has been specifically disclosed by preferred embodiments and optional features, modification and variation of the concepts herein disclosed may be resorted to by those skilled in the art, and that such modifications and variations are considered to be within the scope of this invention as defined by the appended claims.
[00103] Whenever a range is given in the specification, for example, a temperature range, a time range, or a composition or concentration range, all intermediate ranges and subranges, as well as all individual values included in the ranges given are intended to be included in the
invention. It will be understood that any subranges or individual values in a range or subrange that are included in the description herein can be excluded from the claims herein.
[00104] It will be understood that, as used in the description herein and throughout the claims that follow, the meaning of “a”, “an”, and “the” includes plural reference unless the context clearly dictates otherwise. Thus, for example, reference to “a cell” includes a plurality of such cells and equivalents thereof known to those skilled in the art. As well, the terms “a” (or “an”), “one or more” and “at least one” can be used interchangeably herein. It is also to be noted that the terms “comprising”, “including”, and “having” can be used interchangeably.
[00105] It will be understood that when an element is referred to as being “on”, “attached” to, “connected” to, “coupled” with, “contacting”, etc., another element, it can be directly on, attached to, connected to, coupled with or contacting the other element or intervening elements may also be present. In contrast, when an element is referred to as being, for example, “directly on”, “directly attached” to, “directly connected” to, “directly coupled” with or “directly contacting” another element, there are no intervening elements present. It will also be appreciated by those of skill in the art that references to a structure or feature that is disposed “adjacent” another feature may have portions that overlap or underlie the adjacent feature.
[00106] It will be understood that, although the terms first, second, third etc. may be used herein to describe various elements, components, regions, layers and/or sections, these elements, components, regions, layers and/or sections should not be limited by these terms. These terms are only used to distinguish one element, component, region, layer or section from another element, component, region, layer or section. Thus, a first element, component, region, layer or section discussed below could be termed a second element, component, region, layer or section without departing from the teachings of the invention.
[00107] Furthermore, relative terms, such as “lower” or “bottom” and “upper” or “top,” may be used herein to describe one element’s relationship to another element as illustrated in the figures. It will be understood that relative terms are intended to encompass different orientations of the device in addition to the orientation depicted in the figures. For example, if the device in one of the figures is turned over, elements described as being on the “lower” side of other elements would then be oriented on “upper” sides of the other elements. The exemplary term “lower”, can therefore, encompasses both an orientation of “lower” and “upper,” depending of the particular orientation of the figure. Similarly, if the device in one of the figures is turned
over, elements described as “below” or “beneath” other elements would then be oriented “above” the other elements. The exemplary terms “below” or “beneath” can, therefore, encompass both an orientation of above and below.
[00108] It will be further understood that the terms “comprises” and/or “comprising”, or “includes” and/or “including”, or “has” and/or “having”, or “carry” and/or “carrying”, or “contain” and/or “containing”, or “involve” and/or “involving”, “characterized by”, and the like are to be open-ended, i.e., to mean including but not limited to. When used in this disclosure, they specify the presence of stated features, regions, integers, steps, operations, elements, and/or components, but do not preclude the presence or addition of one or more other features, regions, integers, steps, operations, elements, components, and/or groups thereof.
[00109] Unless otherwise defined, all terms (including technical and scientific terms) used herein have the same meaning as commonly understood by one of ordinary skill in the art to which this invention belongs. It will be further understood that terms, such as those defined in commonly used dictionaries, should be interpreted as having a meaning that is consistent with their meaning in the context of the relevant art and the invention, and will not be interpreted in an idealized or overly formal sense unless expressly so defined herein.
[00110] As used in the disclosure, “around”, “about”, “approximately” or “substantially” shall generally mean within 20 percent, preferably within 10 percent, and more preferably within 5 percent of a given value or range. Numerical quantities given herein are approximate, meaning that the term “around”, “about”, “approximately” or “substantially” can be inferred if not expressly stated.
[00111] As used in the disclosure, the phrase “at least one of A, B, and C” should be construed to mean a logical (A or B or C), using a non-exclusive logical OR. As used herein, the term “and/or” includes any and all combinations of one or more of the associated listed items.
[00112] As used in the disclosure, the term “implantable” refers to an ability of a device to be positioned at a location within a body of a user, such as subcutaneously, within a body cavity, or etc. Furthermore, the terms “implantation” and “implanted” refer to the positioning of a device at a location within a body of a user, such as subcutaneously, within a body cavity, or etc.
[00113] As used in the disclosure, the term “wearable” refers to articles, adornments or items designed to be worn by a user, incorporated into another item worn by a user, act as an orthosis for the user, or interfacing with the contours of a user's body.
[00114] As used in the disclosure, “biocompatible” material is a material that is compatible with living tissue or a living system by not being toxic or injurious and not causing immunological rejection.
[00115] As used in the disclosure, “therapeutics” and “therapeutic agent” refer to any substance that provides therapeutic effects to a disease or symptom related thereto. In certain embodiments, a therapeutic agent refers to a substance that provides therapeutic effects to any diseases or biological or physiological responses to the diseases.
[00116] As used in the disclosure, the term “therapy” refers to any protocol, method, and/or agent that can be used in the management, treatment, and/or amelioration of a given disease, or a symptom related thereto. In certain embodiments, the terms “therapies” and “therapy” refer to a biological therapy, supportive therapy, and/or other therapies known to one of skill in the art, such as medical personnel, useful in the management or treatment of a given disease, or symptom related thereto.
[00117] As used in the disclosure, “treat”, “treatment”, and “treating” refer to the reduction or amelioration of the progression, severity, and/or duration of a given disease resulting from the administration of one or more therapies (including, but not limited to, the administration of microspheres disclosed herein). In certain embodiments, the terms refer to the reduction of pain associated with one or more diseases or conditions.
[00118] As used in the disclosure, “engineered cell(s)” refers herein to cells having been engineered, e.g., by the introduction of an exogenous nucleic acid sequence or specific alteration of an endogenous gene sequence. An exogenous nucleic acid sequence that is introduced may comprise a wild type sequence of any species that may be modified. An engineered cell may comprise genetic modifications such as one or more mutations, insertions and/or deletions in an endogenous gene and/or insertion of an exogenous nucleic acid (e.g., a genetic construct) in the genome. An engineered cell may refer to a cell in isolation or in culture. Engineered cells may be “transduced cells” wherein the cells have been infected with e.g., an engineered virus. For example, a retroviral vector may be used, such as described in the examples, but other suitable viral vectors may also be contemplated such as lentiviruses. Non-viral methods may also be used, such as transfections or electroporation of DNA vectors. DNA vectors that may be used are transposon vectors. Engineered cells may thus also be “stably transfected cells” or “transiently transfected cells”. Transfection refers to non-viral methods to transfer DNA (or RNA) to cells
such that a gene is expressed. Transfection methods are widely known in the art, such as calcium phosphate transfection, PEG transfection, and liposomal or lipoplex transfection of nucleic acids. Such a transfection may be transient but may also be a stable transfection wherein cells can be selected that have the gene construct integrated in their genome.
Overview of Hybrid Advanced Molecular Manufacturing Regulator Device
[00119] Present system described herein features a next-generation bioelectronic platform for Targeted Hybrid Oncotherapeutic Regulation (THOR), based on a biohybrid system combining engineered cell factories that are activated and supported by bioelectronics.
[00120] In certain embodiments, regulation of production and dose are enabled by resilient sensors and sensing informatics integrated into the bioelectronic platform. The combined sensing and actuation implant, the Hybrid Advanced Molecular Manufacturing Regulator (HAMMR), as shown in Fig. 1, boosts response rates from less than 10% to over 50% by providing closed-loop patient-specific regulation of therapeutic immunomodulating molecules. The HAMMR of the present invention is minimally invasive implant that provides patient-specific electronically regulated dosing with minimal disruption to the patient’s lifestyle.
[00121] The present invention involves breakthroughs in high-density loading of engineered cells, durable biosensors, and low-power electronics for signal processing and wireless data and power transfer. The resulting bioelectronic cancer therapy enables both systemic and local (tumor microenvironment) delivery of therapies. With a diameter of ca. 1 cm, the HAMMR of the present invention is implanted in a minimally invasive out-patient surgery into the intraperitoneal space where it improves treatments for many of the deadliest treatment- refractory solid-organ malignancies including ovarian cancer, colorectal cancer, pancreatic cancer, hepatocellular carcinoma, and more. Together, peritoneal malignancies account for approximately 34.8% of cancer deaths in the United States, and the ability to treat these diseases could save 187 thousand lives in the US annually. Furthermore, the vast majority of these malignancies are largely unresponsive to immunotherapies. Thus, the present invention improves outcomes for over a third of cancer patients who would otherwise succumb to their disease.
[00122] In one embodiment, the present invention develops an implantable actuation-only version of HAMMR applied to humanized mouse models of peritoneal and metastasis cancer. In one embodiment, sensors are developed in vitro. In another embodiment, sensors are developed in vivo. In one embodiment, the present invention includes sensing/actuation integration, with
validation of function, biocompatibility in large animals, and efficacy in humanized rodents, with a first-in-human trial. In one embodiment, THOR’s HAMMR is built for clinical translation using the latest advances in synthetic biology, biomaterials, biosensing, and wireless communications/power and optimized for long-term (>60d) function in the complex tumor microenvironment.
[00123] In one embodiment, the present invention miniaturizes hardware, sensors, and computation to enable broader access and long-term effectiveness in diagnostic and therapeutic components. The present invention addresses Scalable Solutions and Resilient Systems, increasing access for underrepresented groups to life-saving care because these technologies will be available outside major cancer centers. Furthermore, automatically adjusting the dose for each patient's specific needs overcomes biases that can appear in drug development when trials are performed in small cohorts that may not represent the entire population of patients to accelerate clinical therapy discovery.
[00124] Feedback-regulated control of immunotherapy can yield transformative improvements in boosting immunotherapy response rates across broader patient populations. The present invention provides a 5X increase in therapeutic efficacy, which is enabled by the following key innovations in synthetic biology, bioelectronics, and machine learning that allow personalized local delivery of therapeutic biomolecules.
[00125] In one embodiment, the present invention targets local and systemic therapies. To elicit effective anti-tumor responses, immunotherapies need to activate both local immune responses in the tumor microenvironment as well as systemic immune responses in secondary lymphoid organs, as shown in Fig. 2. Most immunotherapies fail either because dose-limiting toxicity prevents their administration at concentrations necessary for efficacy or because they never generate systemic anti-tumor immune responses. To address these shortcomings, the HAMMR system of the present invention expands the therapeutic window of systemically-toxic immunomodulatory molecules (e.g., IL-2 and IL- 12) through local delivery, while simultaneously facilitating systemic activation of adaptive immunity through secretion of systemic immunomodulatory molecules (e.g., anti-PD-1), engineered with enhanced biodistribution. Thus, the present invention has the unique ability to address both failure modes of immunotherapies to date.
[00126] In one embodiment, the present invention discloses patient-specific dose/feedback- regulated therapies to improve safety/efficacy of the device. A sense-and-respond device that adjusts output immunotherapy production to keep therapeutic doses near the levels needed for activation of immune receptors is critical for improving the objective response rates of therapy, as shown in Fig. 2. For control and feedback, the present invention has opted for electrogenetic induction - compared to other cellular control mechanisms, this method enables fast response, tunable, localized induction properties, and is readily integrated into the platform with minimal power and size demands. HAMMR is designed to account for the natural patient-to-patient heterogeneity of the immune response and allow for variable patient bespoke dosing levels to achieve optimal therapy while avoiding toxicity. To assess and properly account for patient pharmacokinetics (PK) and pharmacodynamics (PD), the platform integrates sensors that are engineered to sense and report extracellular levels of both the produced drug along with biomarkers of treatment efficacy and toxicity. To further improve the therapeutic and safety profiles of the treatment, the present invention combines validated PK/PD models and machine learning to optimize dosing. Feedback is achieved through modulation of electrogenetic cell activation.
[00127] In one embodiment, the present invention adopts electrogenetics for stimulating the engineered cells’ production of therapeutics. In contrast to existing technologies which involves simply load cells between two electrodes and stimulate, the present invention uses conductive scaffolds, as a 3D electrode to active the electrogenetic cells.
[00128] The existing technology stimulates the electrogenetics, and only some engineered cells locate on the electrode. That is, the activation is not uniform. The engineered cells at the electrode are subject to higher potentials/fields, and thus are activated more easily. Likely, to activate more cells in a bulk hydrogel, one would need to boost up the potentials, which would still lead to varied activation levels, but might also adversely affect the cells near the electrodes. [00129] In contrast, the present invention loads a high density of many cells into a conductive scaffold, and therefore no cell is more than 10-100 um from the electrode. Simulation is then much more effective and safe for the cells, providing more predictable, and stable, and long term production of cell-based biologies.
Targeting local and systemic therapies
[00130] As shown in Fig. 1, to elicit effective anti-tumor responses, immunotherapies need to
activate both local immune responses in the tumor microenvironment as well as systemic immune responses in secondary lymphoid organs. Most immunotherapies fail either because dose-limiting toxicity prevents their administration at concentrations necessary for efficacy or because they never generate systemic anti-tumor immune responses. To address these shortcomings, the HAMMR system expands the therapeutic window of systemically-toxic immunomodulatory molecules (e.g., IL-2 and IL- 12) through local delivery, while simultaneously facilitating systemic activation of adaptive immunity through secretion of systemic immunomodulatory molecules (e.g., anti-PD-1), engineered with enhanced biodistribution. Thus, the present invention has the unique ability to address both failure modes of immunotherapies to date.
Patient-specific dose/feedback-regulated therapies to improve safety/efficacy
[00131] A sense-and-respond device that adjusts output immunotherapy production to keep therapeutic doses near the levels needed for activation of immune receptors is critical for improving the objective response rates of therapy. As shown in Figs. 1-2, HAMMR is implanted in the intraperitoneal space through a small incision. Recharging and data offload can be done through a garment of or hub once daily. For control and feedback, the present invention has opted for electrogenetic induction - compared to other cellular control mechanisms, this method enables fast response, tunable, localized induction properties, and is readily integrated into the platform with minimal power and size demands. HAMMR is designed to account for the natural patient-to-patient heterogeneity of the immune response and allow for variable patient bespoke dosing levels to achieve optimal therapy while avoiding toxicity. To assess and properly account for patient pharmacokinetics (PK) and pharmacodynamics (PD), the device integrates sensors that are engineered to sense and report extracellular levels of both the produced drug along with biomarkers of treatment efficacy and toxicity. To further improve the therapeutic and safety profiles of the treatment, the present invention combines validated PK/PD models and machine learning to optimize dosing. Feedback is achieved through modulation of electrogenetic cell activation.
Resilient sensing in vivo
[00132] Continuously monitoring the biomarker concentration in vivo is critical for feedback control and personalized medicine by tracking efficacy and toxicity. However, the lack of stability and longevity in current bioanalysis platforms is a grand challenge. Long-term stability
is affected by many factors including bio-fouling and the desorption of bio-recognition elements during repeated voltametric measurements. Continuous sensing is also challenging, requiring a finely-tunable bio-recognition element that possesses balanced association/dissociation characteristics to the target. Innovation in THOR addresses long-term stability through a multifaceted approach: 1. Addressing antifouling via surface chemistry and surface manipulation; 2. Overcoming instability caused by bias stress by modifying sensor operation (bias, time/duty cycle), and by controlling electrode material, structure, anchoring chemistry, redox reporter selection; 3. Manipulating the binding and dissociation energies (Kd engineering) by modifying recognition elements; and 4. Hardware integrating advanced signal processing methodologies to quantify and mitigate the impacts of sensor degradation, employing large-scale data analytics techniques such as machine learning.
Biomaterials and oxygenation for long-term performance of cell factories
[00133] Implanted biomaterials often induce foreign body responses, strong innate immune- mediated cascades consisting of inflammatory events, and wound-healing processes that lead to fibrosis and subsequent implant failure. The present invention overcomes critical limitations of allogeneic cell therapies and improve long-term cell survival by encapsulating cells in immunomodulatory-alginate derivatives. In one embodiment, the present invention has identified lead formulations that protect encapsulated cells from the host immune system for at least 6 months. Furthermore, these capsules with allogeneic cells remained free of fibrotic deposition for up to 4 month in non-human primates, as compared to <2w for SLG20 clinical grade alginates. Thus, using these alginate-based derivatives minimizes inflammatory responses and prolong in vivo cell-mediated protein delivery. To increase cell loading for higher dosing potential, a safe, highly-controlled on demand electrocatalytic on-site oxygenation platform is integrated. To enhance oxygen production and reduce energetic cost, nanostructured and biocompatible sputtered iridium oxide is used to sustain cell viability while maintaining therapeutic peptide secretion with high-cell density (>60k/mm3), enabling higher dosing. The present invention has successfully demonstrated this concept in hypoxic in vitro conditions (1% O2) for 3 weeks, and in vivo (rat) for 10 days.
Long lifetime and robust operation via onboard machine learning and wireless power [00134] Making sense of complex and multi-modal biosensor data is computationally challenging. Machine learning techniques are well suited to this problem, but off-the-shelf
machine learning hardware requires Watts of power, which is prohibitive for implantable bioelectronic systems: bulky batteries, cooling systems, and table-top graphical processing units (GPUs) are impracticable for this application. THOR of the present invention builds tiny machine learning acceleration for signal compensation and interpretation, on-board, extending battery lifetimes and providing flexible computing power for changing disease progression. Using magnetoelectric fdms, which provides the highest power density for wireless bioelectronics, the present invention is able to create small implants that can be delivered minimally invasively and efficiently charged overnight. The result is a therapy that is almost unnoticed by the patient allowing them to move freely about their day without the need to wear bulky battery powered external devices.
Biomarkers to guide personalized immunotherapy to maximize efficacy and limit toxicity [00135] The present invention uses a broad range of preclinical models, including non-human primates and humanized murine models, to validate the utility of biomarkers sensed by HAMMR in predicting efficacy/toxicity. Furthermore, the present invention establishes a computational framework to integrate the power of machine learning with multi-omics data from both clinical and preclinical results. Using this framework, one expects to identify novel biomarkers associated with antitumor activity and/or immune-related adverse effects. These biomarkers are used to optimize the implementation of cytokine-based immunotherapy.
Uniqueness towards advancing the current state of the art
[00136] The key hallmark of the breakthrough successes of modern immunotherapies over other treatment modalities is their capacity to generate cures in patients with advanced malignancies. When effective, these therapies generate sustained pools of tumor-reactive lymphocytes that reside systematically throughout the patient. While it remains unclear how long to maintain patients on immune checkpoint inhibitors (ICTs) following tumor clearance, emerging evidence reveals that activation of treatment responses, when effective, occurs rapidly following onset of treatment and within secondary lymphoid organs. In cancer patients exhibiting complete remissions following tumor-reactive T cell infusions or ICI treatment, responses were detectable for over a decade. Lymph nodes can harbor sustained pools of tumor- reactive T cells, and the generation of these systemic durable immune responses within lymph nodes is the key feature distinguishing responders from non-responders. Thus, patients who fail to maintain durable responses following cessation of ICI treatment likely never generated
systemic anti-tumor responses and memory within lymph nodes, and clinical trials (e.g., SWOG- 1801) indicate that commencing immunotherapy prior to lymph node resection (i.e., in the neoadjuvant setting) drastically improves survival. Of note, these treatment regimens are often as short as four weeks, indicating, in agreement with preclinical ICI and vaccination studies, that the generation of systemic immunity occurs rapidly. Thus, the HAMMR device of the present invention remains operational for 60 days, more than twice the length of the neoadjuvant treatments in the NSCLC trials. By combining on-demand activation to generate robust tumor killing with systemic activation to generate stem and memory pools of anti-tumor lymphocytes, as well as personalized sensing feedback regulation of dosing, THOR is uniquely poised to elicit rapid, durable anti-tumor responses to some of the deadliest malignancies.
[00137] The HAMMR is a novel platform for delivery of a wide range of therapeutics but developed here for cancer immunotherapy. Through this effort, the present invention develops and tests in humanized mice the efficacy of this approach, safety in large animals, and evaluates both safety and efficacy in a first-in-human clinical trial in refractory recurrent ovarian cancer patients. The present invention develops this new cancer therapy platform which reduces cancer- related deaths by > 50%.
1. Biochemical targets for sensing & actuation
[00138] In one embodiment, the present invention targets the following immunomodulatory molecules for production by the cell factories: IL-2, IL-12, IL-18, IFNa, anti-CD40, anti-PD-1, anti-PD-Ll, and anti-Lag3, which were selected and prioritized for engineering based on criteria detailed in Fig. 3. Briefly, as reported in clinical and/or preclinical studies of systemic administration, immunostimulatory reagents such as IL-12, anti-CD40 could result in severe or unacceptable immune-related adverse effect (irAE), while irAEs from other types of immunotherapy, such as anti-PD-l/Ll are largely manageable. Therefore, the present invention stratified immunomodulatory reagents into two categories based on expected locations for target dose: 1) Local (severe or unacceptable irAEs) and 2) Systemic (no or manageable irAEs). Then, the present invention pairs one type of local reagent with one type of systemic reagents to balance feasibility, novelty, and targeted cell types/pathways. Finally, the present invention ranks each pair based on readiness, expected efficacy, and toxicity. As such, the present invention discloses a target ranking of 5 pairs of immunoregulatory reagents.
[00139] In one embodiment, HAMMR also senses an array of targets. For example, monitoring the therapeutic molecules above provides for direct feedback control to enable accurate and consistent dosing. In addition, the HAMMR device monitor patient responses by sensing an array of cytokines which have been associated with immunotherapy efficacy, toxicity, or both, particularly in the context of ICB and cytokine therapy. The present invention has implicated multiple cytokines. Specifically, efficacy is monitored with the T cell recruitment chemokine CXCL10, the immunosuppressive cytokine TGF-13 and angiogenesis promoting cytokine VEGF, all of which have been positively or negatively associated with ICB responses. TNF-a, IFN-y, and IL-8 are measured as elevated levels of these cytokines indicate response to multiple immunotherapies, while excess levels can indicate toxicity, including during cytokine therapy. Finally, IL-6 is included due to its strong association with toxicity in most immunotherapies.
2. Validation in Animal Models
[00140] The specific animal model for various tasks is described herein. The present invention carefully considered the most appropriate animal model for each task. Live animals are used only for tests requiring fully intact physiology or behavior, with rodents being used for early cell and bioelectronic component development. Efficacy for human cancer immunotherapies can best be modeled in humanized mice. To evaluate the clinical translation of the present invention, the present invention uses cynomolgus macaques because they are more immunologically similar to humans than rodents, and thus are a more appropriate surrogate model to study before translating to human patients. Similarly, the cynomolgus macaques’ foreign body response to immunostimulatory implants more closely resembles that in humans due to their exposure to a more diverse set of antigens than inbred rodents. Thus, NHPs provide the greatest translational validity for safety and PD studies of any animal model.
[00141] In one embodiment, a total of N=9 NHPs are used over the course of the project (Phase 2) across multiple tasks. Specific details regarding experimental procedures and sample sizes are provided in the tasks below.
Phase 1: Development of HAMMR
3.1. Electrogenetic Cell Factories
[00142] The present invention discloses a drug delivery system for controlled release of immunomodulatory agents in response to electric signals, and engineers and validates cell factories using electrogenetic control.
[00143] Specifically, the present invention leverages innovative, recently described electrogenetic systems that use membrane depolarization to control the NF AT transcription factor and, in turn, regulation of user-defined target genes. These systems have been prioritized over other possible modalities due to their robustness, predictability, and demonstrated efficacy in human cells. In one embodiment, the present invention uses human retinal pigmented epithelial cells, ARPE-19 cells because it has been shown that these cells are non-tumorigenic, display contact inhibited growth characteristics, are amenable to genetic modification, and have been shown safe in previous human trials (NCT03316300). In one embodiment, the present invention has engineered ARPE-19 cells to produce a wide range of cytokines continuously and in response to light, as shown in Fig. 4. Encapsulated ARPE-19, IL-2 cytokine factories, have advanced to human clinical trials for patients with high-grade serous adenocarcinoma of the ovary, primary peritoneum, or fallopian tube. The cell engineering research team has deep and demonstrable expertise in engineering human cells for biomedical utility, mammalian synthetic biology, protein engineering, and modeling drug dosing and synthetic biology circuit kinetics.
Electrogenetic Regulation in Human Cells
[00144] The present invention employs electrogenetic strategies based on equipping human cells with calcium and potassium channels to achieve membrane depolarization- mediated control of the NF AT transcription factor in ARPE-19 cells, as shown in Fig 5, panel (a, top) and, in turn, regulation of immunomodulators shown in Fig. 3. The present invention first develops ARPE-19 cells that express the L-type voltage-gated calcium channel CaV1.2 and the potassium channel Kir2.1 (EiectroARPE-19), similar to previous reports. Briefly, electrical pulse triggers membrane depolarization and the opening of the calcium channel. This process results in calcium influx and activation of the calcineurin/calmodulin pathway, and translocation of dephosphorylated NF AT into the nucleus. There, a transgene under an NFAT-sensitive promoter is expressed, completing an adapted electrogenetic circuit previously described. The performance of Electro ARPE- 19 cells is validated and optimized using an expression vector encoding human IL-2 under the PNFAT3 promoter and quantifying IL-2 levels in the cell culture supernatant upon electrical pulse stimulation using ELISA. The present invention uses custom or commercial
well plates and electronics hardware to deliver electrical stimulation with electrodes onto which cells are seeded (and a second electrode is immersed into the media), or a system whereby EiectroARPE-19 cells are seeded on transwell inserts, with platinum electrodes placed at opposite sides of the transwell fdter, as shown in Fig. 5A. In one embodiment, the present invention measures IL-2 production from cells using ELISA and correlate voltage, pulse length, frequency and stimulation time with IL-2 expression. In one embodiment, the present invention achieves secretion of IL-2 within 60 min. Importantly, this workflow is compatible with any immunomodulator listed in Fig. 3 and/or any new engineered immunomodulators.
Modeling
[00145] To optimize design of electrical signals and cell mixtures, the present invention constructs a coupled model describing the electrogenetic control system and pharmacokinetics. Expression of the therapeutic protein is tunable by modulating aspects of the electrical signal, e g., voltage or pulse length which lead to membrane depolarization. In one embodiment, the model consists of ordinary differential equations describing membrane depolarization, calcium ion flux into the cell, subsequent activation of NF AT transcription factor, and expression of the protein of interest. The models predict the level of protein production and the kinetics (delays) of activation/deactivation as a function of the electric input parameters. Model predictions are used as an input to PK/PD modeling to optimize device design to maintain safe and efficacious levels of the therapeutic of interest and to design a control system.
[00146] In one embodiment, electrogenetic circuit responses might not be as fast as what has been previously described; faster response times may be needed for therapeutic molecules. Therefore, in one embodiment, the present invention explores other electrogenetic systems and/or implement a type 1 coherent feedforward loop motif with the electrogenetic circuit to reduce the delay of response to the electrical pulse signal and amplify transcriptional output. A second risk is that Electro ARPE- 19 cells may not perform similarly to cells in which electrogenetic systems have been optimized (i.e., HEK293T, pancreatic 0-cells). Although this risk is low, the present invention optimizes the performance of the electrogenetic platform in alternative cells if needed (i.e., HEK293T, MCF7, MSCs, HS27 cells) and subsequently port optimized circuitry into Electro ARPE- 19 cells.
Optimized potency and production
[00147] The present invention has demonstrated that the production of transgenic payloads can be enhanced using optimized construct designs. The present invention optimizes the expression of the electrogenetic systems from 1.1.1. driving immunomodulatory payloads to >lpg/cell/day from stimulated cells using these approaches. The present invention tests different combinations of modularized synthetic biology parts and plasmid construct architectures that have been observed to enhance mammalian gene expression, as shown in Fig. 5B, which involves testing various synthetic mammalian promoters (e.g., CMV, CAG, EFS, hUbC), optimized Kozak sequences, nuclear export signals (NESs), and/or polyA tails. To compare plasmid architectures, the present invention delivers the electrogenetic system to EiectroARPE-19 cells using multiple vectors and a single, all-in-one (AIO) vector. The present invention explores different arrangements of gene expression cassettes on individual vectors within each system variant. Optimization is initiated using IL-2 and the electrogenetic workflow described above. The present invention then evaluates that electrogenetically-induced IL-2 expression is sustained and tunable (i.e., on/off controllable in response to electrical input) over lOd in vitro. The optimized architecture for the expression of the IL-2 immunomodulator in EiectroARPE-19 cells is then evaluated for use with a second immunomodulator (anti-PD-1), the sustained and tunable expression of which is also measured over lOd in vitro. The present invention performs mixed cell experiments with these two engineered EiectroARPE-19 cells. The optimal levels and ratios of mixed engineered EiectroARPE-19 cells are defined based on the model predictions and in vitro data. Optimized construct designs/architectures is then used to create electrogenetically- responsive production of the remaining 6 immunomodulators (IL-12; anti-PDLl; INF a, anti- Lag3, anti-CD40 agonist, and DRIL-18) in EiectroARPE-19 cells and rank ordered combinations 2-5 as shown in Fig. 3, which are then assayed similarly in vitro.
[00148] When optimization for one immunomodulator may not be consistent across molecules, the present invention uses high-throughput modular cloning platforms to optimize each immunomodulator in EiectroARPE-19 cells one by one. If ELISA is too slow for processing, the present invention discloses other systems in which the immunomodulator proteins are fused to a reporter (e g., SEAP/EGFP), allowing one to determine the immunomodulator protein expression level through corresponding quantification assays. Further, the present invention has also demonstrated that transgenes and endogenous gene products can be robustly activated using innovative CRISPR synthetic biology technologies.
[00149] In certain embodiments, the present invention optimizes engineered human allogeneic cells to become potent factories of immunomodulators in response to electrogenetic stimulation. [00150] In certain embodiments, the present invention optimizes the Electro-cells to meet a production threshold of 1 pg/cell/day. By maximizing immunomodulator output, the present invention can reach physiological drug levels with a limited number of cells. Different construct designs will be tested to find the system architecture that boosts production of individual immunomodulators. Expression of immunomodulators will be tested in vitro over 10 days to ensure that expression is both sustainable and tunable (i.e., on/off controllable in response to electrical input) over 10 days in vitro in these engineered cells. We will perform mixed cell experiments in vitro with two different engineered ElectroCells (each expressing a unique immunomodulator) and define optimal levels and ratios of mixed engineered ElectroCells for therapeutic use.
[00151] In certain embodiments, the present invention discloses a Completion Criteria and Performance Metrics/Assessment. In particular, different construct designs are tested, including the use of various synthetic promoters (e.g., CMV, CAG, EFS, or hUbC), plasmid architectures (delivery through individual vectors versus all-in-one delivery), and/or other genetic elements (e.g., polyA tails, Kozak sequences, nuclear export signals). The production features of each design will be evaluated by measuring protein expression levels (via ELISA) corresponding to the immunomodulator of interest. To ensure system modularity, optimized architectures are tested using two lead immunomodulators (i.e., IL-2 and anti PD-1) and then ported to other i mmunomodul ators .
Durability and Resilience
[00152] The engineered cells used in the present invention are by design create pro- inflammatory environments via the production of high levels of potent immunomodulators. Recent studies have shown that cells expressing therapeutic payloads can be adversely impacted by inflammatory environments, such as those that may be produced by the engineered EiectroARPE-19. Although this is able to be controlled by tailoring electrogenetic on/off kinetics and thereby immunomodulator production, the present invention further improves EiectroARPE-19 cells to be durable and resistant to inflammatory environments. The proinfl ammatory cytokines IFNy, TNF-a and IL-ip act broadly and are each known to cause overall decreases in ARPE-19 function. Therefore, in one embodiment, the present invention also engineers ARPE-19 cells to
be resistant to these signaling molecules using CRISPR/Cas9-mediated knockouts (KOs). Specifically, the present invention KO the receptors for IFNv (IFNGR1/2), TNF-a (TNFR1/2 and TRAF-1/2), and ZL-10 (IL-1R1/2 and IL-1RAP) in ARPE-19 cells and measure relative cell viability and function between WT and ARPE receptor KO cells using Annexin staining. In these experiments, engineered ARPE-19 KO cells are grown in media containing IFN-y, IL-ip, and TNF-a to mimic in vivo adverse immune environments. These experiments are carried out over 60 days. ARPE-19 cells with improved resilience to these pro-inflammatory molecules are ported into 1.1.2 to synergize with electrogenetic production efforts.
[00153] Some KOs may be lethal or reduce overall cell fitness. The present invention assesses these phenomena and omit lethal KOs/KOs that reduce cell fitness. KOs of indicated receptors may lead to no significant benefits in cell production/viability, in which case the present invention explores CRISPR screening modalities to identify and utilize key drivers of improved immunomodulatory production in ARPE-19 cells.
Molecular Engineering
[00154] Naturally occurring proteins are limited in number and also by their pleiotropic effects, which hinders their efficacy as targeted therapeutics. There is growing interest in manipulating existing proteins or engineering entirely new proteins in order to overcome the deficiencies of molecules found in nature or to improve existing clinical drugs. In one embodiment, engineered new proteins are produced by the engineered cells of the present invention for cancer therapy.
[00155] The two aspects of target proteins are selectivity and circulation half-life. Locally delivered targets are intended to mobilize immune effector cells (i.e., CD4+ and CD8+ T cells and NK cells) to clear tumors. IL- 12, IFN-a, an anti-CD40 agonistic antibody, and decoyresistant IL-18 activate immunostimulatory and not immunosuppressive effects. In the case of IL-2, protein engineering for selectivity is required, as IL-2 activates both immune effector cells and regulatory T cells (Tregs), confounding its efficacy as an anti-cancer agent. In one embodiment, the present invention engineers an IL-2/antibody fusion protein (F10 IC) that biases IL-2 towards activation of immune effector cells over Tregs. However, this molecule is based on a full-length antibody and has limited tumor penetration. The present invention designs an IL- 2/single chain variable fragment (scFv) fusion protein that recapitulates the bias of F10 IC in miniaturized format. The present invention performs IL-2 signal activation studies via flow
cytometry and receptor binding studies via biolayer interferometry to validate the biased activity of our engineered IL-2 molecule. With respect to PK property engineering, locally delivered targets are intended to be transient; thus, a short in vivo half-life is desired. Systemically delivered targets are intended to be available throughout the body for sustained time periods; thus, a long in vivo half-life is desired. The only locally delivered target with a long in vivo halflife is the anti-CD40 agonistic antibody, which has extended half-life due to engagement of its Fc domain with the neonatal Fc receptor (FcRn). In one embodiment, the present invention mutates select residues in the antibody Fc domain to knock out FcRn interaction without impacting protein function. Biolayer interferometry -based binding studies confirm ablation of FcRn binding. Systemically delivered targets include antibodies against PD-1 (pembrolizumab), PD- L1 (atezolizumab), and LAG3 (relatlimab), all of which have extended half-life.
[00156] In another embodiment, the other 3 locally delivered molecules (IL- 12, IFN-alpha, and DR-18) can be substituted. Moreover, in yet another embodiment, the present invention engineers any of the locally delivered or systemically delivered proteins to alter their selectivity and/or PK behavior as needed. In one embodiment the present inventio tunes the selectivity, pharmacology, and immunogenicity of the delivered molecules. Selectivity can be tuned through modulation of target affinity. PK properties can be manipulated to extend or restrict serum halflife through Fc domain engineering of antibodies. Potentially immunogenic epitopes can be identified through computational algorithms to screen for class II MHC peptides, and the proteins can be mutated accordingly to mitigate these liabilities.
Cell scaffolds for activation
[00157] Electrogenetic induction of therapy requires uniform actuation over a large volume of engineered cells. While planar electrodes have been used previously, it is known that stimulation electric field scales as r 2 where r is the distance from the electrode, thus the consistency of activation can be variable over the cell factory population. The present invention uses a conductive scaffold or mesh which is embedded within the cells or cell-laden hydrogel. In one embodiment, the present invention uses a conducting scaffolds of high surface area, scalability, and of sufficient conductivity to enable uniform electrogenetic actuation. In one embodiment, the present invention develops high conductivity poly(3,4-ethylenedioxythiophene) (PEDOT) particles produced via dropwise addition to an acid bath, yielding particles with highly tunable internal surfactants (controlled by remaining PSS content from the initial PEDOT:PSS
dispersion.) These particles form high performance inks with a slew of biomaterials, and can be printed or cast and crosslinked through a number of modalities, as shown in Fig. 6. In particular, Fig. 6 shows that PEDOT-based particles (410 S/cm) are highly dispersible within aqueous 3D- printable resins for (A) micro-continuous liquid interface production (pCLIP) stereolithography to generate conductive scaffolds; (B) Photo and SEM image of PEGDA stents (-100 pm diameter struts) with 10 wt% loading of PEDOT particles. Scale bars for the photographs and SEM images are 2 mm. For example, micro continuous liquid interface printing (pCLIP) yields scaffolds with features of ca. 10’s-100s of um. Alternatively, in another embodiment, ice- templated, or fibrous conducting polymer scaffolds, commercially available graphene foams or Pt wire meshes can be used.
[00158] Activation is tested in modified well-plates with a control counter electrode (Pt) and compared to activation with flat metallic electrodes. While flat electrodes typically use stimulation parameters of 7.5V, 1ms pulses with a frequency of 1Hz, the electrodes of the present invention reduced average cell-electroactive scaffold distance lowers the stimulation voltages, while scaling current with overall surface area, which is part of the materials downselection selection approach. The tradeoff in power consumption vs. total cell activation is explored balancing therapy production and power draw. The connection to the stimulation hardware is separately be investigated to determine the robustness and durability using both physical clamping and conductive epoxy approaches, before integrating the device for in vitro /vivo validation, and integration. In one embodiment, the present invention also discloses the use of the oxygenation hardware as the CE electrode for stimulation.
[00159] In one embodiment, the hardware can probe for changes in scaffold impedance. In another embodiment, the present invention discloses conductors like Pt meshes, or photolithographically patterned mesh electrodes that are monolithically integrated with the electronics hardware (i.e., on Flex PCB), which also address connect! on/integrati on issues. In another embodiment, planar electrodes can be employed, and device dimensions reduced to account for non-uniformities - at the cost of accessible dose.
3.2. Cell life support
Biomaterials
[00160] Fibrosis is one of the major contributing factors for the failure of many implanted devices. Cells are of particular issue with fibrosis since they evoke a strong immune response,
and a fibrotic capsule stops the flow of biomolecules to and from the cells and kill them. To address this, the present invention engineers immunomodulatory biomaterials with improved biocompatibility that mitigates foreign body response. This is accomplished by developing alginate-based hydrogel formulations that are immune modulating in nature and can host the engineered cells for prolonged duration, as shown in Fig. 7. In particular, Fig. 7 shows immunoisolating chamber and chemical coatings to protect against foreign body responses. Panel (A) shows engineered cells loaded in immunoisolation chamber; panel (B) shows high viability post loading (3d. Post encapsulation); panel (C) shows small molecule surface chemistry protects against host FBR (28d explants from mice); and panel (D) shows host tissue biocompatibility of surface modified devices (28d histology adjacent to implants).
[00161] In one embodiment, the present invention discloses a library of alginate-based hydrogels that could mitigate fibrosis. These hydrogels have been previously employed for the transplantation of xenogenic human pancreatic islets that led to the restoration of long term glycemia in profibrotic C57BL/6. These hydrogels prevent fibrosis and promote long term cell viability, as shown in Fig. 7 panels (B) and (C). In one embodiment the present invention discloses a carrier that mitigates fibrosis and supports the engineered cells. In one embodiment, the present invention modifies the backbone of the sodium alginate polymer with immunomodulatory small molecules. To validate the anti-fibrotic properties of these materials, the present invention performs a short in vivo study spanning over 14 days to evaluate the fibrosis and hence their biocompatibility. In one embodiment, these materials successfully evaded fibrosis and supported viability for greater than 80 days when capsules fabricated from them were implanted in the intraperitoneal space of mice and NHPs. Since the device aims to host the cells for over 60 days, the present invention also evaluates the biocompatibility of these materials in vivo over extended time points of 30, 60 days.
[00162] In one embodiment, different linkers are used for the library of alginate-based hydrogels that could mitigate fibrosis.
Oxygenation
[00163] To better oxygenate chronically transplanted high-density cell therapies, the present invention engineers an electrocatalytic on-site oxygenation (ecOz) for implantable cell therapies. This is accomplished by electrocatalytic water splitting using integrated catalytic arrays in the cell-containing hydrogel. In one embodiment, the present invention discloses that ecO2 arrays
which adopts sputtered IrCh (SIROF) catalyst were able to support the bioactivities of high-cell density capsules (60k cell mm-3) up to 21 days under hypoxic environment (1 % O2) as well as implanted in rodent for 10 days, as shown in Fig. 8. ecCh lowers the operational power requirement and minimizes the generation of detrimental byproducts. It has a safe potential range up to 1.9 V without production of reactive oxygen species (e.g., hydrogen peroxide) and chlorine (from oxidation of chloride). Fig. 8 shows electrocatalytic onsite oxygenation (ecO2). Panel (a) shows linear sweep voltammetry (LSV) measurement in anodic scan for water oxidation; panel (b) shows cell viability (Live/dead assay) and peptide secretion (ELISA) as a function of time; panel (c) shows optical imaging of capsules after 21 -day hypoxic (1 %) incubation with or without oxygenation; and panel (d) shows immunostaining images after 10-day implantation in rats with or without oxygenation.
Fabrication of oxygen generating modules
[00164] Microfabricated ecO2 arrays are translated to flexible substrates (e.g., polyimide), as shown in Fig. 9. Two arrays are facing back-to-back for whole-HAMMR-volume oxygenation. 3D-printed oxygenators with controlled morphologies, e.g., hierarchical helix or coiled electrodes are also tested. In one embodiment, the present invention discloses 3D electronics employing nanoparticle-based 3D printing with high resolution (< 10 pm). Catalytic ink is formulated by blending nanoparticles (e.g., IrCh, Pt) to fabricate 3D printed ecO2 as shown in Fig. 9. In particular, Fig. 9 top panel shows a double-side flexible catalytic arrays; and Fig. 9 bottom panel shows a 3D-printed double helix catalytic arrays; inset: 3D-printed spiral shape electrodes with controlled nano porosity.
Device optimization and evaluation
[00165] To improve the stability of the catalysts (SIROF or EO2), in one embodiment, the present invention modifies the synthesis conditions such as the ratio of gas mixture (Ar and O2) synthesis pressure, applied power (DC or RF), and post-processing procedures (e.g., thermal annealing, sintering of 3D printed parts). In one embodiment, IrO2 crystallinity improvement contributes to extended chronic oxygen generation. The present invention tailors crystallographic characteristics and hierarchical nanostructures of catalysts to achieve long-term stability (> 60 days), modulating crystallinity, dimensions of electrode, applied duty cycles, macro- and nanoporosity. In vitro production of oxygen to support high cell density (60k cell mm'3) is validated by oxygen measurement, oxygen sensitive dye imaging and cell viability (live/dead assay). In
one embodiment, the present invention engineers the therapeutic cell maintenance platform to allow at least 70 % viability for encapsulated 60k cell mm’3 under hypoxic conditions for at least 60d. Produced peptides is measured via ELISA. Oxygen generation platform is evaluated in vivo (rat) to achieve at least 70 % viability over 60d. Power supply for electrocatalysis and control unit are securely passivated using medical grade resin (e.g., Parylene C, epoxy, PDMS) to protect them from potential corrosion and water ingress damage.
[00166] In one embodiment, in improve the stability of the catalyst, IrO2 crystallinity can be adjusted by modifying the synthesis conditions and/or annealing. In another embodiment, nanocarbon materials, e g., laser induced graphene (LIG) that exhibits exceptional electrochemical stability is used. In one embodiment, twisted Pt/Pt+SIROF wires are adopted as working and counter electrodes. In another embodiment, when there is insufficient oxygen supply due to a restricted power budget or limited electrolysis, the present invention may use the decomposition of solid peroxides as an alternative.
3.3. Validation of actuation HAMMR
Integration of hardware and cells
[00167] The present invention develops miniature electronics to stimulate the electrically- conductive scaffolds. The electronics platform is small enough to be implanted in the humanized mouse model, and have the ability to dynamically adjust to changing scaffold impedance to reliably actuate the engineered cells. In one embodiment, the present invention uses a miniature antenna (RF, solenoid, or magnetoelectric receiver) and a magnetic or RF transmitter to enable contactless wireless power delivery to the implanted HAMMR for multi-day experiments and validation of the actuation- HAMMR. It has been shown that actuation is possible at 7.5 V. However, if the voltages needed to induce the cells is higher than expected, in one embodiment, the present invention may use an off the shelf LCD controller for voltages up to 20V; if the required voltage exceeds 20V, in another embodiment, the present invention uses a Lomir rodent swivel -tether system and power the device via a programmable power supply instead of WPT.
PK/PD Modeling
[00168] To predict HAMMR pharmacokinetics (PK) and pharmacodynamics (PD), the present invention develops a computational model. In one embodiment, the present invention plans to use a minimal physiologically based pharmacokinetic (mPBPK) model structure due to its relative simplicity. In one embodiment, PK model structure is consistent across delivered
therapeutics, whereas modeling PD is dependent on the therapeutic of interest. For example, in one embodiment, the present invention uses a model of IL-12 therapy where the production rate of IFNy is determined by the formation of complex between IL-12 and IL-12 receptor. This model is specific to IL- 12 and IFNy, but the present invention also uses a similar framework for all therapeutics. The PD of the delivered therapeutic is modeled as interaction between the therapeutic and its target and the resulting biological effects.
[00169] The present invention constructs the model as a system of ordinary differential equations describing transport of the delivered therapeutics between compartments, interaction between the delivered therapeutic and its target and biological effects, cell viability within the HAMMR device, and electrogenetic control of the HAMMR device, as shown in Fig. 10. The present invention evaluates model performance and estimate parameters by optimizing fit to in vitro and in vivo preclinical data. In vitro data is used to estimate model parameters related to the HAMMR device, whereas in vivo data is used to estimate physiological parameters such as transport and clearance. Model performance is determined quantitatively (minimum weighted sum of error between model predictions and measurements) and qualitatively to ensure predictions are physiologically relevant with no overfitting. In one embodiment, the addition of peripheral tight and leaky tissue, such that the complex trends are also captured.
4.1. Resilient Sensing
Antifouling strategies
[00170] The present invention assembles an antifouling strategy on different aspects: nanoscale, micron-scale and macro scale. It has been shown that flat electrode surfaces are more prone to fouling in comparison to nano/micro structured surfaces due to different surface adhesion energies. On the nano/micro scale, the present invention manipulates electrode surface architecture using electrochemical techniques to achieve various morphologies (e.g., pores, nanostars, etc.) and carefully select for surfaces that provide the maximal resistance to fouling. On the macro scale, the present invention, in one embodiment, uses porous hydrogel materials to protect the biofunctional surface from unwanted interaction with the biofluids to enhance surface lifetime. HAMMR sensors can also be inhibited by fibrosis. Sensors are therefore also in need of being engineered to be immune-evasive. In addition to immunomodulatory alginates, the present invention also identified small molecules that, when functionalized to a material surface, cause the material to resist fibrosis. These coatings have been shown to work on various materials
(medical catheters, PDMS, and PTFE) and across multiple implant sites (IP space, subcutaneous space, and the brain), stopping or reducing fibrosis in implanted materials. To this end, the present invention down selects a library of materials/assemblies, show stable operation in complex environments on the benchtop, and show operation, and stable readings in vivo in rats for 30 days.
Overcoming Bias Stress
[00171] The desorption of biorecognition elements (i.e., aptamer) leads to sensor instability. The present invention incorporates strategies, e.g., anchoring chemistries, electrode material engineering and redox reporter selection, as shown in Fig. 11 panel (a) to tackle bias stress. Operation of aptamer-based sensors at negative potentials strain the phosphate backbone of DNA and cause a reductive desorption of thiol-Au bond, i.e., desorption of aptamer from an Au electrode. In one embodiment, the present invention selects stable anchoring chemistries from multi-thiol bonding, carbon-nitrogen bonding, multi TL-TI stacking, peptide self-assembly on graphene, and phenyl azide chemistry. Multidentate bonding like di-thiol or tri-thiol can potentially improve bonding strength and stability, at the expense of aptamer density. In one embodiment, the present invention uses nanostructured electrodes with a large surface area for anchoring multidentate aptamers with high aptamer density (same device footprint).
Nanostructured electrodes with extended electrical double layer over the electrode surface can enhance electron transfer rate and improve detection sensitivity. In one embodiment, the present invention has synthesized tunable carbon- and metallic-based nanostructured materials such as microstructured gold, as shown in Fig. 11 panel (b), and CVD grown 3D fuzzy graphene with temperature/time controlled flake density, as shown in Fig. 11 panel (d). Metallic based nanostructured electrodes can be synthesized by electrochemical deposition and morphology is dependent on the precursor solution concentration and rate of deposition. Nanostructured electrodes are evaluated by cyclic voltammetry (CV) or electrochemical impedance spectroscopy (EIS) for electrochemical surface area characterization. In one embodiment, microstructured gold electrode-based aptamer sensors have been demonstrated to improve the detection limit of TGF- bl compared with bare gold electrodes, as shown in Fig. 11 panel (c). In addition to surface chemistry and electrode engineering, modifying the work functions of the working electrode is a technique which can allow for a lower redox potential that ultimately reduces the operating voltage and bias stress. Chemical modifications of the surface, metal doping, and self-assembled
monolayer formation can be used to modify the work function of the nanostructured electrode materials. Selecting redox reporters with lower redox potential or positive redox potential such as Os also allows stable operation under lower bias stress. In one embodiment, electrochemical measurement techniques are chosen, i.e., electrochemical impedance spectroscopy (EIS, low amplitude of voltage (~10mV)) and pulsed chronoamperometry (CA, short duration (~lms) of voltage) measurements cause less bias stress than the commonly used square wave voltammetry (SWV, high amplitude (±0.5V) and long duration (~ls) of voltage) measurement. If use of SWV is inevitable, the present invention performs SWV with longer interval time between each scan and narrow the scan range (limit potential range near the peak potential) to minimize the sensor bias stress. In one embodiment, the present invention has demonstrated that the lifetime of the sensor can be improved by using optimized SWV scheme, as shown in Fig. 12. In particular, Fig. 12 shows effect of bias stress on sensor stability. Panel (A) shows normalized redox peak vs. time during SWV with different biasing scheme. Sensors with short voltage scan range and long interval time between scans are stable over time; and panel (B) shows preliminary results of the sensor stability on pig wound using the biasing scheme in A.
[00172] In one embodiment, the present invention selects optimal redox reporter and attach aptamer through stable anchoring chemistry onto tunable nanostructured sensors, which is tested by optimized electrochemical method in vitro on bench top and allows for a 10-fold increased life-time compare with “standard” sensor.
Kd engineering
[00173] The present invention develops bio-recognition elements (i.e., aptamer, antibody, nanobody) with fine-tunable reactivity (association/dissociation) in biofluids through Kd engineering to enable continuous biosensing. To do so, the present invention generates libraries of recognition elements that are generated from original selected sequences (e.g., starting from aptamer sequences known from the literature, and determining Kd) and modified via mismatch, mutagenesis, chemical modification, and truncation. In one embodiment, the present invention controls binding dynamics in molecular sensors. In this aim, the present invention probes for fast-binding, highly specific and sensitive recognition species with high- release properties that can enable quick turnarounds and regeneration of the sensors for reusability in vivo. Thus, the present invention demonstrates sensor regeneration with retention times <5 min where other sensor metrics are within a factor a 5 of unmodified aptamer.
Sensing informatics
[00174] The ultra-low power, high fidelity, low SNR chronoamperometry microelectronics present a significant amount of incoming data that is noisy, complex, and fast changing. With the advent of ultra-low power accelerated machine learning cores alongside state- of-the-art signal processing and learning algorithms, the present invention is well positioned to employ sophisticated informatics onboard the implant to navigate both signal degradation concerns from fouling/degrading biosensors, as well as challenging therapy delivery actuation decisions. Computational methods can account for degradation via signal reconstruct on/sampling. In one embodiment, the present invention uses computationally efficient compressed sensing techniques coupled with active learning (i.e., sensing behavioral and mechanistic changes) to reconstruct a lossy signal from degrading sensors. In one embodiment, the present invention uses sensor data alongside simulated mechanistic traces, with random sampling, for validation purposes. Supervised regression-based and recurrent machine-learning techniques are applied to the datasets to learn the behavior of the biosensor as it degrades, allowing for the prediction of and compensation for sensor degradation. The present invention compares the error reduction for various model architectures and select a suitable machine learning/signal processing pipeline for execution on the FPGA/ASIC.
[00175] In another embodiment, the present invention achieves stable sensor readouts by using a control (i.e., scrambled aptamer), or housekeeping sensor (stable background biomarker determined from omics in 1.10) to yield stable differential signals. In yet another embodiment, the present invention performs SELEX from a randomized DNA library to select for desired aptamers using a microfluidic platform for selecting aptamers in a narrow-desired Kd window. [00176] In one embodiment, with respect to computation, unsupervised techniques, informed by domain specific mechanisms and behaviors of biosensors, some of which are known a priori, are explored if the amount of data required to train a supervised model is not known. In one embodiment, the present invention explores constellations of smaller, less accurate models as well as calibration routines to increase model accuracy. In one embodiment, the present invention encapsulates different sets of sensors in slowly dissolvable protective polymeric scaffolds of differing thickness that will be activated at different timepoints, thus using multiple sensors for a shorter duration each.
4.2. Sensing validation
[00177] The present invention involves multiple parallel efforts regarding sensing validation, and when testing an approach, direct comparisons are made to a “standardized” version of a particular sensor (i.e., a starting aptamer sequence, bonded via monothiol to a flat gold working electrode) or to one that is properly unmodified (i.e., with and without a protective anti-fouling coating). However, the ultimate goal is to address stability while down selecting and preparing a sensor set for implementation in HAMMR. As such, in vitro validation progresses with a combination of benchtop testing of sensing metrics (sensitivity, limit of detection, lifetime) in spiked PBS, and in complex environment samples such as heparinized blood and patient samples. Patient samples are used to assess the accuracy of sensors compared to established analytical tools (e.g., ELISA), and are tested for 30-60 days on the benchtop in complex, heparinized blood. Due to the sheer volume of approaches and targets that must be explored and engineered to meet the strict timelines of THOR, one embodiment of the present invention only focuses on in vivo validation in the antifouling task. To validate in vivo in a rat, the present invention builds an in vivo testing device which is implantable (mm-scale) and low-power (tens- of-pW average power draw) capable of accurately reading data from the biosensors developed and sending it (wireless/tether) to a desktop/laptop application for data storage and analysis.
4.3. Platform engineering
[00178] The present invention develops an integrated hardware platform for cell life support and actuation, sensing and signal processing, energy (charging and usage), and communications. Developing this platform necessitates overcoming challenges in onboard computation, signal processing, and wireless power delivery. When the wearer is asleep, a bedside or clothes integrated transmitter charges the implant's battery and extracts biosensor data using magnetoelectric (ME) power transfer. This approach has been demonstrated with other WPT- implant systems and enables therapy progress reporting and continuous assessment in clinical trials.
[00179] ME power transfer has great potential for wireless mm bioelectronic implants. It offers low tissue absorption, high efficiency, small size, and low misalignment sensitivity. Using the same mechanism for power and communication is crucial for miniaturizing implants. The present invention has designed and demonstrated near-zero power magnetoelectric backscatter from mm-sized implants by exploiting the converse magnetostriction effects, in an 8.2-mm
wireless implant > 1 kbps data rate at 2 cm and a bit error rate (BER) less than IE-3, as shown in Fig. 13.
[00180] In one embodiment, the present invention investigates novel multi- antenna form factors for increased power to generate 5mW at 4 cm in-vivo and explores lossy coding and delay/interrupt tolerant communication protocols to overcome the higher error rate from the longer distance. In one embodiment, in case that increasing transmission power may not be possible due to safety, the present invention explores hybrid automatic repeat request (HARQ) protocols used to combat channel fading (as in satellite networks). To address the computational load of degrading biosensors and complex streaming data, the present invention enhances the existing model from 1.2 for PGA and ASIC implementation. In one embodiment, the present invention has successfully demonstrated accelerator designs for biomedical implants that utilize embedded ML models to process high-rate data and make decisions, as shown in Fig. 13. The FPGA circuit/architecture holds the entire signal/machine-leaming pipeline for the overall system validation and demonstration. To ensure a less than lOmW FPGA power consumption, the present invention integrates several low-power techniques (e.g., event-driven adaptive compensation, optimized dataflow, and micro- architecture) without compromising the signal/machine-leaming compensation performance. This computational design also enables the controller for regulating therapy. In one embodiment the present invention adapts the mPBPK model into a software/firmware/hardware controller, with tunings entered from in-vivo data via ML on-device, using the same FPGA/ASIC to conserve space, and power.
[00181] In certain embodiments, the present invention uses commercial off-the-shelf (CoTs) integrated circuits to develop a miniature (approximately 5 mm x 5 mm) PCB capable of sending short pulses of an as-yet unspecified voltage (likely >10V) to the conductive scaffold.
[00182] In certain embodiments, the device includes feedback mechanisms to monitor that all parts of the scaffold are receiving appropriate waveforms. The device are used for humanized mice models and is an initial prototype for the fully integrated actuation-HAMMR device.
[00183] In certain embodiments, the device receives power & commands via one of a few solutions: either off-the-shelf NFC charging, standard cages wrapped with wire for wireless power transfer, or a dedicated tether system, each standard with rodent/murine experimentation systems. Bluetooth Low Energy may be used in lieu of the above for communication and control. The device provides data offload via this to a smart phone or desktop for capture of experiments.
5.1 Humanized Models
[00184] To evaluate the capacity of the various immunomodulatory agents to elicit potent systemic anti- tumor immune responses and select lead candidates, the present invention develops primary and metastatic mouse models of peritoneal malignancies. Effective immunotherapies require activation of systemic immunity, and such immunity is ideally capable of clearing stage IV metastatic disease. In one embodiment, the present invention develops mouse models of metastasis (predominant cause of cancer-associated deaths). As the proposed immunomodulators are human-specific, the present invention develops humanized murine models suitable for testing immune activation and toxicity of the therapy combinations resulting in >30% reduction in tumor burden for phase 2 studies.
[00185] In one embodiment, the present invention monitors the tumor burden of 3 ovarian cancer (SKVO3, Igrov-1 and OVCAR4) and 3 pancreatic ductal adenocarcinoma (Panc-1, AsPCl, and BxPC3) lines, engineered to express luciferase, by bioluminescence imaging (BLI). Metastatic models consist of intrasplenic (liver) or lateral tail vein (lung) injections of tumor cells. Reconstitution of the human immune system can be achieved in NSG mice engrafted with human peripheral blood mononuclear cells (Hu-PBMC mice). Based on growth kinetics in Hu- PBMC mice, the present invention selects 4 lines (2 for each cancer type) that exhibit 4 weeks of progressive growth in the peritoneum and metastatic sites. In one embodiment, the present invention uses ARPE-19 cells secreting IL-2 and IL-12 to compare tumor and immune responses in the humanized models to previous syngeneic models. The present invention then tests whether ARPE-19 cells expressing pairs of immunoregulatory agents listed in Fig. 3 can achieve target concentrations and exhibit 3-fold increase in activation cytokines (e.g., IFN-g), and a 2-fold increase in lymphocyte infiltration and/or proliferation. The present invention also confirms that designed biomarker sensors can achieve expected function and support down selection of therapies. The present invention then evaluates toxicity of the actuation-HAMMR devices in 2 tumor models to ensure <20% reduction in body weight and <20% variation in liver function tests (LFT)for Phase 2 studies.
[00186] In one embodiment, because heterogeneity between cancer types and patients yields variability in treatment responses that are difficult to model preclinically, the present invention focuses on two cancer types frequently causing peritoneal carcinomatosis. The present invention screens six lines and validate the findings in at least two humanized mice models in both
peritoneal and metastatic settings by two research groups. As 2 weeks are needed to observe the antitumor effects, the present invention selects six tumor lines that are reported to grow well in NSG mice and conducts experiments to down-select four lines. Testing multiple lines mitigates the risk that these models lack therapeutic windows. In one embodiment, the present invention has eight additional OC and PDAC lines should these six not exhibit ideal growth kinetics.
5.2 Animal model validation
[00187] NHP studies to address PK/PD and safety are accomplished in coordination as an effort to make thoughtful and efficient use of these animals. Delivery/safety metrics are obtained from samples and data gathered from the same individual monkeys that are used for PK/PD testing. The fully assembled device is validated in rat models. Additionally, the present invention immune-profiles the rat with cytokines delivered from the device to show that one can successfully modulate the immune system. Fully assembled devices are then be implanted intraperitoneally into the abdomen site of NHPs. Drug deliveries are determined through remote cell induction and monitoring of drug concentrations in blood serum and IP fluid. Through this study, the present invention demonstrates “turn on” and “turn off’ drug production kinetics at various time points to get data for the PK/PD model. Multiple tests are performed over a 60-day period to ensure consistent and predictable drug delivery profiles at therapeutically relevant levels. In one embodiment, the present invention assesses that biofouling or foreign body responses does not decrease efficacy of the carrier nor significantly affect dosing for 10 days in NHP, and that the device supports encapsulated cell factory viability and sensor functionality for > 60 days in NHPs (2.2.2.M2). To determine the proper dosage during these biocompatibility tests, 3 separate dosages are tested in a total of 9 NHP (n = 3 per condition).
[00188] In one embodiment, when the dose required exceeds the capacity of the cell factories, the present invention uses higher cell loading densities (as enabled by O2 delivery) or add multiple implants to increase production.
PK/PD In Large Animal Model
[00189] In one embodiment, the present invention fits PK/PD data from large animal models and estimate safe and efficacious HAMMR dose regimens for clinical trials. In one embodiment, human model parameters are estimated by fitting the model to clinical trial data of the corresponding therapeutics, e.g., data from a clinical trial of IP administration of IL-12. Allometric scaling or in vitro-in vivo extrapolation methods are used to estimate human
parameters from in vivo estimated parameters otherwise. In vitro-in vivo extrapolation methods are more physiologically relevant; however, utility is dependent on the quality of data available. Conversely, allometric scaling methods are empirical in nature but simpler to employ. Large animal models more closely replicate PK/PD seen in humans.
[00190] In one embodiment, the present invention implements sensitivity analyses to determine which model parameters and assumptions are most important in final predictions.
5.3 Omics/AI models
[00191] The present invention creates baseline omics profiles and models of cytokine molecules from ascites samples of ovarian cancer patients. To model the complex biological context, the present invention proposes to develop a peritoneal tumor immune microenvironment agent-based model guided artificial intelligence (PTIME-ABM-AI) system that acts as the brain of the system to intelligently execute Closed-loop control of the PTIME through the cell factories based on input of the sensors, as shown in Fig. 14. In one embodiment, the present invention develops the PTIME- ABMs based on previous work such as the Ovarian Cancer Metastasis Simulation (OCMetSim) models and the Pressure Ulcer Agent Based Model, or PUABM using NetLogo or SPARK. In one embodiment, the present invention evaluates and guides the model design and optimization using real-world omics data. Multiplexed cytokine profiling data is obtained from around 30 ovarian cancer patient ascites samples by the Luminex assay, including 65 cytokine/soluble factors. Cytokine data is standardized and normalized to remove batch effects. Single-cell data of cellular components from ascites samples obtained through CyTOF and/or scRNA-seq are analyzed to identify cell types and cell states and corresponding RNA and protein molecular profiles. The present invention finds molecules that are consistent across longitudinal samples (for sensor calibration) and estimates baseline levels of the molecules. These data are used to develop a peritoneal tumor immune microenvironment (PTIME) Agent Based Model (ABM) to simulate the development of ascites in peritoneal cavity of ovarian cancer patients and the growth and spread of cancer cells, as well as the interactions between cancer cells and the immune system. Knowledge about the PTIME such as cell types, cytokines and soluble factors, their means, ranges, and putative functional relations are curated from literature and from experience gained in past preclinical and clinical studies, and be coded as agents, links, parameters and equations into the model. The ABM aims to approximate the real- world PTIME, capturing the homeostasis, heterogeneity, and stochasticity of the environment,
and can be used to generate synthetic data points to expand the sparse data measured from the real -world experiments.
[00192] In one embodiment, it is possible that the PTIME-ABM cannot well approximate the real- world data. This can occur when there is insufficient existing knowledge to mechanistically link measurements. If additional knowledge cannot be acquired from literature or data, the present invention may develop multiple versions of the PTIME-ABM, each instantiating a “best guess” at a putative mechanistic link. In one embodiment, the present invention tests all the plausible versions of the ABM in subsequent tasks and narrow down to those achieving consistently better results.
6. Wireless power and information communication
6.1 Implant Wireless Power Transfer and Communication
[00193] In certain embodiments, the present invention discloses wireless power transmitters and miniature receivers to recharge or continuously power bioelectronics implants that will be in use throughout the project. Demands on device size, form factor, and functionality change throughout the project as testing moves from rodents into large mammals.
[00194] In certain embodiments, power circuitry is used to facilitate communication between the implant device and external devices. In certain embodiments, the present invention discloses a foundational circuitry and design for eventual power delivery and low data rate communication into and out of the peritoneal cavity, from implant device to external charging device (or another transmitter). In certain embodiments, the present invention discloses energy management, storage, and battery recharging circuitry.
[00195] A major challenge for implanted bioelectronics is efficient power and data transmission through biological tissue. When the device size is made small enough to be compatible with miniature bioelectronic implants, energy harvesting antennas must also be made small compared to the wavelength of the electromagnetic waves typically used to power biomedical devices. These sub -wavelength devices do not efficiently capture electromagnetic energy. To compensate for the reduced power transfer efficiencies, the transmitter power must be increased; however, tissue absorbs electromagnetic energy at these frequencies placing strict limits of the transmitter power to avoid potentially harmful heating of the tissue. Ultrasound provides an alternative for wireless power; yet ultrasound suffers impedance mismatches
between air and tissue, and thus ultrasonic gel is typically required, which would be cumbersome during extended use.
[00196] In certain embodiments, the present invention discloses magnetoelectric (ME) materials enable wireless power delivery more than 8 centimeters beneath bone and tissue, which is possible because the present invention uses low-frequency magnetic fields that are not absorbed or reflected by the body. Importantly, this allows the present invention to effectively deliver power and communicate with miniature devices in the body.
[00197] In certain embodiments, ME materials are fabricated and packaged with custom PCBs (supporting receiver circuitry) for testing power transfer efficiency and data link.
[00198] In certain embodiments, the ME materials are fabricated by laminating a commercial magnetostrictive layer (Metglas) with a commercial piezoelectric layer (PZT or PVDF). The laminated films are then be cut to size using a laser cutter and used for power and data communication. The data uplink is accomplished via backscattering the transmitter ME signal, meaning a single antenna and system can be used for both power, downlink, and uplink communication, reducing size and complexity and obviating the need for a bulky NFC or other inductive antenna, which may not be feasible due to the long and skinny device form factor. [00199] In certain embodiments, the present invention discloses alternative antenna form factors, including multiple smaller antennas in series/parallel along the length, or wrapping the end of the implant in multiple smaller antennas, and using intelligent impedance matching circuitry to balance power delivery efficiency.
[00200] In certain embodiments, testing of data and power transfer are performed by placing the prototype carrier in a tissue phantom simulating the peritoneal cavity using SynDaver abdominal Tissue Plates surrounding a bladder with composite fluids (or similar tissue phantom) to simulate an implant in the cavity (an approximate distance of 4-6 cm from the wearable transceiver).
[00201] In certain embodiments, total power transfer is measured by the maximum resistive load that can be driven by ME and PCB/supporting circuitry, and/or the maximum charging speed of an energy storage capacitor.
[00202] In certain embodiments, the data link bandwidth is measured by the maximum data rate of correctly decoded downlink data by an onboard MCU, uplink is measured by the transmitting device.
[00203] In certain embodiments, if ME induced power is lower than anticipated, the dimensions of ME materials can be optimized and/or more ME films can be added in parallel to increase harvested power without requiring a higher field strength.
Phase 2
7. Integration
[00204] In Phase 2, the present invention assembles the cell therapeutics, oxygenator, sensors and bioelectronic control unit, with an Application Specific Integrated Circuit (ASIC) machine learning accelerator into a hybrid bioelectronic implant for rat and NHP validation. Briefly, the electrogenetically actuated cell therapeutic chambers are deployed in a 3D printed cage with the conductive scaffold. An oxygenator platform is integrated with sensing chips and the encapsulated bioelectronic control unit. The oxygenator end of the construct is inserted into the cell/stimulator component, and the conductive scaffold is physically wired to the control unit board with encapsulation. In one embodiment, the control unit is hermetically sealed for water ingress to allow continuous performance in the IP space. In one embodiment, the HAMMR platform is able to charge and communicate with a transmitter/hub device that is tethered and embeddable in undergarments or in a bed for ease of charging and nightly data offload. The HAMMR ASIC conducts onboard machine learning for signal compensation, as well as host the control algorithm for PK/PD aware therapy delivery. The present invention then uses local O2 production to maintain high cell loading viability and records the stability of desired production levels across different HAMMR compartments. The bioelectronic feedback control system of the present invention maintains drug production levels in rats, which are measured by the multichannel HAMMR sensing unit. These experiments are performed first using wired or battery powered devices before implementing a fully wireless version that integrates the custom circuitry for power regulation, communication, and wireless power. Finally, the ASIC development builds off the FPGA design/circuit developed in Phase 1, where individual control and machine learning functions were designed and tested first as separate flows: these validations de-risk the fully integrated accelerator solution that will minimize the form factor and power requirements. [00205] In one embodiment, the present invention leverages the NHP testbed (Celltrans) to verify functionality of the fully integrated HAMMR system across a range of operating conditions, environments, and levels of physical activity. In one embodiment, the present invention uses a pre-existing system from the DARPA to support NHP testing: the fully
integrated, battery powered external hub (extHub) are mounted in a primate jacket against the abdomen. The extHub only functions to deliver power wirelessly to the implant (to recharge it) and as a data download source. The extHub is replaced once a day to offload data and replace/recharge its batteries. The HAMMR device is implanted in the peritoneal cavity and beneath the extHub.
[00206] In one embodiment, the present invention optimizes the dimensions of ME materials and/or add more ME films in parallel to increase harvested power without requiring a higher field strength. In one embodiment, the present invention adopts other protocols for high error and lossy channels if communication rate is low. The flexibility of the design concept- recharging and data offload at night along with battery implant size, can all be modulated to ensure ME power delivery is sufficient.
[00207] As the integrated HAMMR is readied for translation, contingencies and fail-safes should be considered: In the event that HAMMR is not charged by the user and the device battery charge falls below a threshold, the device is programmed to turn off production regardless of current state. Should the user require device termination, one can (a) surgically remove the device, and/or (b) administer a pharmacological safety switch technology used in DARPA NTRAIN into the cell constructs, or alternatively the cells are responsive to chemotherapy.
8. Cancer model
8.1. Cancer model efficacy:
[00208] In Phase 2, the present invention evaluates the therapeutic potential of the lead candidate device in short- and long-term humanized mouse models and demonstrate that it can achieve >30% reduction in tumor burden and <20% loss of body weight. The present invention also identifies immunological characteristics associated with efficacy and toxicity to aid in translation. To evaluate efficacy, HAMMR devices are implanted one week following injection of tumor cells into the peritoneal cavity (primary) or into the spleens or lateral tail veins (liver/lung metastatic), and tumor growth is evaluated for three weeks by BLI, with a goal of >30% reduction in tumor burden. Systemic cellular and molecular biomarkers provide insights into responses throughout treatment. To identify potential clinical biomarkers of response, the present invention employs comprehensive immune profiling of blood and metastatic sites by mass cytometry. In one embodiment, the present invention applies Statistical Scaffold Maps to
evaluate changes in immune repertoire and activation across all immune subsets within a tissue. Finally, the present invention evaluates long-term efficacy of the treatments. Human CD34+ hematopoietic stem cell engrafted NSG mice is inoculated with tumor cells as above, devices are implanted, and survival of mice are evaluated with a primary goal of an increase of >2.5-week median overall survival. The generation of memory phenotypes in T cells are evaluated by flow cytometry.
[00209] In one embodiment, while graft-versus-host disease can develop in Hu-PBMC mice 4 to 8 weeks following PBMC engraftment, the present invention conducts a long-term survival study in huCD34- NSG mice, which exhibit stable engraftment for over one year. If GVHD limits the studies in the Hu-PBMC mice, the present invention switches to using Hu-PBMC mice with MHC-I/II double knockout, which increases engraftment stability for > 75-100 days. It is possible that the liver metastasis model, while a common site of metastasis, will not predict systemic immune responses given its proximity to the device. Thus, the present invention has included lung metastasis models to ensure adequate evaluation of systemic responses outside of the peritoneum.
8.2 Establishment of safety thresholds
[00210] To meet requirements for IND application, samples of NHP studies proposed in phase 2 are used to establish the safety thresholds for the clinical trial in Phase 3. The present invention assesses dose limiting toxicity in NHPs using samples collected from NHP studies (ip fluid and blood) at various time points post therapy. In one embodiment, the present invention collects omics data from longitudinal NHP blood/ascites samples as well as the in situ HAMMR biomarker data, including cytokine/soluble factor profiling (around 7 cytokines in the device design table and biosensor readouts), cytokine profiling using Lumitex, flow cytometry (additional immune abundance/function markers) and bulk RNA-seq data. Toxicity readouts from the animals are obtained, which include general health measurements such as liver function, body weight, etc., indicative of immune cell abundance/states and irAE. RNA-seq data is preprocessed, quantified, and deconvoluted to estimate tumor burden, clonality, and immune cell type and abundance. The present invention constructs a safety score out of the toxicity readouts and perform multivariate regression analysis, which allows establishing dosage limits for each molecule and identifying subsets of sensor markers most predictive of safety. The present invention validates and optimizes the PTIME- ABMs using the above omics data, for
example, whether the molecular parameters in the PTIME- ABM is consistent with observations from the omics data. This exercise may also reveal additional cytokines/soluble-factors that could be better candidates to implement on the HAMMRs.
[002111 In one embodiment, the present invention trains Al under the deep reinforcement learning Q-leaming framework using data generated by the optimized PTIME-ABM. For each device, the present invention performs regression to establish the functional relationship between the safety/efficacy metrics and the biomarker measurements. The present invention discretizes continuous cytokine levels to define the states and modulatory actions, given the constraints and the on-device biomarkers and immunomodulators. In one embodiment, the present invention tests a variety of Al models, such as deep neural networks, XGBoost trees, etc.
[00212] The granularity of cytokine/biomarker level discretization may affect the accuracy of the ABM as well as model complexity. Thus, in one embodiment, the present invention tests a variety of granularity levels and determines the ones that achieve good tradeoffs. As an alternative to ABM, in another embodiment, the present invention directly learns a pharmacodynamic model using SNODE, which has demonstrated stability and ability to integrate sparse training data. The present invention also examines the PK/PD models developed in Sections 3 and 4. In the scenario that the DRL is not successful (e.g., very slow convergence), the present invention adopts model predictive control (MPC) and Lyapunov based control methods to derive the Al models.
Phase 3
9. Human trial
[00213] Based on the above-described rationale predicated by supportive pre-clinical and nonhuman primate data, it is planned conduct a pilot study to assess the safety and feasibility of using the HAMMR in humans. The present invention anticipates using cell factories engineered to produce IL2 and anti-PD 1 as an initial payload for several reasons. First, anti-tumor efficacy, IP administration tolerability, and downstream immunological effects are well-characterized and predictable. Second, the ongoing Phase 1/2 study of an encapsulated cell product engineered to produce native human interleukin-2 have established a recommended phase 2 dose (RP2D). In assessing the most reasonable approach, balancing the realistic regulatory requirements and timeline, it is believed a pilot clinical test of the combined product is required.
[00214] IP administration of HAMMR in patients with recurrent/refractory solid tumors with carcinomatosis are associated with an acceptable safety profile.
[00215] The present invention conducts a pilot clinical trial to determine the safety and feasibility of HAMMR armed with a human IL-2 therapeutic module implanted intraperitoneally (IP) in patients with advanced solid tumors. The primary endpoint is safety assessed by adverse event reporting by CTCAE v.5.0. The secondary endpoints consists of: 1) Comparison of ascites cytokine (including IL2) levels as determined serially by direct sampling versus as assessed by the sensing module of the HAMMR; 2) Dynamic immunological changes in peripheral blood and in the peritoneal environment assessed by serial collection and investigation of peritoneal fluid and cells using Luminex assays, flow cytometry, and scRNAseq; 3) Changes in the tumor (assessed by spatial transcriptomics and immune profiling); and 4) Objective response rate (ORR) and clinical benefit rate (CBR). The present invention enrolls a maximum of 12 patients in this pilot study at a rate of one subject per month. The subjects are divided into cohorts of 3 patients with increasing duration of device implantation for each cohort. The first cohort undergoes surgical implantation of the HAMMR for two weeks. For subsequent cohorts, the duration of device implantation increases by two weeks, such that subjects in cohort 4 have the device for eight weeks. Ascites and blood are collected weekly, and response is evaluated by RECIST v.1.1 based on imaging every four weeks. All subjects undergo surgical removal of the device after completing the prescribed duration of therapy. Following removal of HAMMR, subjects enter long-term follow-up with every eight- week imaging until progression.
[00216] Summary statistics are used to describe the demographic and clinical characteristics of the study population. The incidence of AEs is described by grade and relation to study drug overall and by dose level. The DLT rate are estimated along with a 90% credible interval overall and by dose level. The present invention also estimates the posterior probability that the DLT rate is greater than 25%. All biomarker data (i.e., IP fluid, blood samples, gene expression) are summarized descriptively. Descriptive statistics, such as the mean, standard deviation, coefficient of variation, median, minimum, and maximum values, are provided for continuous endpoints. The rates of binary endpoints are provided along with corresponding 2-sided 95% Cis using an exact method. The ORR is estimated along with a 95% CI. DCR is estimated in a similar fashion. DCR is defined as ORR+ SD. Time-to-event endpoints are summarized using the Kaplan-Meier method and displayed graphically when appropriate. Median event times and
2-sided 95% Cis for each time-to-event endpoint is provided. PFS is defined as the time from initial treatment to the earliest disease progression or death date. In the absence of disease progression or death at the time of data cutoff, subjects' PFS times are censored at the date last assessed for progression.
8.1 Immune Profiling and Biomarker Discovery
[00217] Longitudinal omics data is used to characterize immunological effects of HAMMR. By the end of this phase, the present invention validates the induction and maintenance of antitumor immune responses by HAMMR and identify biomarkers associated with clinical responses and/or toxicity. The present invention obtains longitudinal data including tumor growth, adverse effect, cytokine levels in serum and ascites (by Luminex), and abundance/function of immune subsets (by flow cytometry, CyTOF and RNA-seq, etc.) from enrolled patients. The present invention leverages these data and efficacy outcomes to better predict which biomarkers correlate with efficacy and toxicity. Toxicity metrics in humans such as liver function, body weight, irAE, etc. are obtained to construct a safety score. In one embodiment, the present invention performs multivariate regression analysis to predict safety scores from the molecule measurements, establishing dosage limits for each molecule and identifying subsets of sensor markers most predictive of patient safety. The ABM and the Al models are evaluated and calibrated by human data and be used to predict dose-adjustment to assist clinical decision making, and to assess the potential of executing full-autonomous tasks.
Embodiment 1: Cell Engineering
[00218] In certain embodiments, the present invention optimizes Electro-cells (engineered cells) to meet a production threshold of ^1 pg/cell/day. By maximizing immunomodulator output, the present invention achieves physiological drug levels with a limited number of cells. Different construct designs are tested to find the system architecture that boosts production of individual immunomodulators. Expression of immunomodulators are tested in vitro over 10 days to ensure that expression is both sustainable and tunable (i.e., on/off controllable in response to electrical input) over 10 days in vitro in these engineered cells. In certain embodiments, the present invention perform mixed cell experiments in vitro with two different engineered Electrocells (each expressing a unique immunomodulator) and define optimal levels and ratios of mixed engineered Electro-cells for therapeutic use.
[00219] In certain embodiments, different construct designs are tested, including the use of various synthetic promoters (e g., NF AT and it variants, CMV, CAG, EFS, or hUbC), plasmid architectures (delivery through individual vectors versus all-in-one delivery), and/or other genetic elements (e.g., polyA tails, Kozak sequences, nuclear export signals). The production features of each design are evaluated by measuring protein expression levels corresponding to the immunomodulator of interest. To ensure system modularity, optimized architectures are tested using two lead immunomodulators (i.e., IL-2 and anti PD-1) and then ported to other immunomodulators.
[00220] Figs. 15-16 show promotor construct designs for engineered Electro-cells to enable production of >1 pg/cell/day immunomodulator in vitro.
[00221] In certain embodiments, the present invention provides the engineer cells to secrete high amounts of cytokine (i.e. IL2) in response to KC1 stimulation, which is a proxy for electrical stimulation. ARPE-19 cells were engineered to have channels which would be activated in response to electric/KCl stimulation. Such activation cause a signal cascade to activate the NF AT promoter which drives the transcription of the cytokines of interest, e.g., IL2.
[00222] In certain embodiments, the present invention demonstrates that engineering the promoter sequence of NF AT can increase IL2 production. Fig. 15 shows various promoter construct designs of NF AT, where the additions listed (AP-1, OCT, GATA, etc.) are genetic motifs added around NF AT, as listed in Table 1.
[00223] These constructs (pNFATrepVl-12)(SEQ ID No: 2-13) were then transfected into ARPE-19 cells, respectively, and productions of IL2 in response to KC1 stimulation were shown in Fig. 16.
[00224] As it can be seen in Fig. 16, pNFATrep V3, (AP-1 + NFAT)x9, increased production of IL2 most.
[00225] In certain embodiments, the vector/plasmid has a NF AT promotor which includes a NF AT sequence alone, or a NF AT sequence together with one or more additional sequences of AP-1, OCT, GAT A, C/EBP, MAF, IRF4, MEF2, T-be, etc.
[00226] In certain embodiments, the NF AT promotor comprises 1-10 copies of a NF AT sequence, or 1-10 copies of a combined sequence having a NF AT sequence together with one or more additional sequences of AP-1, OCT, GAT A, C/EBP, MAF, IRF4, MEF2, T-be, etc.
[00227] Figs. 17A shows optimized plasmid designs for calcium channels, potassium channels, and the therapeutics, e g. IL2, and Fig. 17B shows 24-hr hIL production and hAnti- PD1 production after KC1 stimulation.
[00228] In certain embodiments, the present invention discloses various avenues to increase production of desired cytokines from the electrogenetic cells. ARPE-19 cells were engineered to have channels that activate in response to electric/KCl stimulation, which when stimulated, cause a signal cascade to activate the NF AT promoter which drives the trancriptions of the gene of interest (GOI), e.g. IL2 and antibodies.
[00229] ARPE-19 cells were engineered with different ratios of the plasmids containing the channel proteins and the plasmid containing the NF AT promoter with GOI.
[00230] In certain embodiments, a 1 : 1 : 1 ratio of the 2 channel proteins and the reporter protein, as shown in Fig. 17A, had the best on/off ratio of the conditions.
[00231] In certain embodiments, the ratio between the 2 channel proteins ranges from 1 : 10 to 10: 1. In certain embodiments, the ratio of the channel proteins in total with the reporter protein ranges from 1 : 10 to 10: 1.
[00232] In certain embodiments, the plasmids were introduced in the cells using “PiggyBac” method.
[00233] In certain embodiments, the cells were engineered to secrete IL12 and antibodies (IgG), highlighting that the device of the present invention can be adopted for producing a large plethora of factors.
[00234] In certain embodiments, the present invention increases cytokine production by the engineered cells in response to electrical stimulation specifically.
[00235] Figs. 18A-D show cytokine production, by the engineered cells, LCC, and % viability in response to AC stimulation.
[00236] In certain embodiments, ARPE-19 cells were engineered to have channels which activate in response to electric stimulation, and which, when stimulated, cause a signal cascade to activate the NF AT promoter which drives a transcription of the GOI.
[00237] In certain embodiments, the present invention stimulated the cells with AC, and changed the distance between the two electrode from 1 to 2 to 3 mm. As shown in Figs. 18B-D, 3 mm electrode difference induces similar production of IL2 by the engineered cells as the KC1 stimulation. Therefore, 3 mm is an optimal distance between electrodes for this AC stimulation
condition. In certain embodiments, the optimal distance between electrodes for this AC stimulation ranges between 2.5 mm - 3.5 mm. In certain embodiments, the optimal distance between electrodes for this AC stimulation ranges between 2 mm - 4 mm.
Embodiment 2: Hybrid Advanced Molecular Manufacturing Regulator
[00238] In certain embodiments, the present invention discloses a conductive scaffold that can electrically stimulate the engineered cells disposed therein, the present invention discloses optimal material for a 3D conductive scaffold for culturing the engineered cells and providing electrical simulation to the engineered cells.
[00239] In certain embodiments, the present invention discloses designing and constructing conductive scaffolds which host the electrogenetic cells with or without their supporting biomaterial hydrogel, which are activated electrically using a high conductivity scaffold that can be integrated with both the cells and the electronics/wireless hardware.
[00240] In certain embodiment, the scaffolds includes one or more of the following materials a hydrophilic base polymer, e.g., alginate, PEDOT:PSS incorporated into the alginate, graphene foams (commercially available), lithographically defined flexible metallic constructs, or Pt meshes.
[00241] As shown in Figs. 19A-B, platinum, graphene foam, and PEDOT:PSS are three candidate for conductive scaffold. Their advantages and disadvantages, relative forms, and chemical formulas are shown in Fig. 19A and 19B, respectively.
[00242] Fig. 20 shows that ARPE-19 cells seeded on the graphene condition had the lowest cell viability, with Pt and PEDOT:PSS demonstrating higher cell viability.
[00243] Fig. 21 shows that PEDOT :PSS is less efficient than Pt for stimulating the cells electronically, because no IL2 production after the cells were culture on the PEDOT:PSS material and a current run through the material. That is, cell viability on stimulated PEDOT scaffolds was -25%, which shows high cell mortality correlates with hydrogen peroxide formation when a voltage is applied to PEDOT:PSS in an aqueous environment.
[00244] Fig. 21 also highlights that there is a high amount of H2O2 produced during the electrical stimulation, which leads to cell death.
[00245] Fig. 22 shows that the platinum material has both high production of IL2 and viability of cells when the electro-active ARPE-19 cells are cultured on it and stimulated thereon. That is,
Pt is one of the optimal materials for the 3D conductive scaffold to house the cells in the final device.
[00246] Fig. 23 shows photos of Pt conductive scaffold c, e.g., balls of platinum mesh seen in the top left. Confocal imaging was also used to confirm cells growing on the platinum conductive scaffold.
[00247] In certain embodiments, the conducive scaffold is made of Pt.
[00248] In certain embodiments, the conducive scaffold is made of Pt in combination with one or more other materials, e g. PEDOT:PSS, graphene, or other metals.
Embodiment 3: Oxygenator
[00249] In certain embodiments, the present invention discloses a device that generates oxygen, which is necessary for higher cell viability of the engineered electrically active ARPE- 19 cells disposed in the device.
[00250] In certain embodiments, the present invention utilizes standard microfabrication and additive manufacturing (e.g., aerojet printing), and assembly techniques to build a flexible and scalable oxygen evolving catalytic platform, which are placed within the cylindrical mesh form to ensure uniform 3-dimensional spatial distribution of the generated oxygen.
[00251] In certain embodiments, while microfabrication techniques include the synthesis and patterning of nanostructured catalysts (e.g., sputtered iridium oxide) in planar substrates, additive manufacturing allows the present invention to fabricate 3-dimensional electrodes which are beneficial to construct hierarchical nanostructures. In particular, the mixture of nanoparticles formulating with catalytic (e.g., IrOx nanoparticles) and conductive components (e.g., platinum nanoparticle) can be printed with high resolution (<10 pm) and controlled porosities. Sintered at elevated temperature, the printed nanoparticles are fused together and form interconnected nanoporous catalytic electrodes.
[00252] In certain embodiments, to design effective geometries, finite element analysis can be adopted to estimate the produced oxygen profiles, considering practical challenges in manufacturing processes.
[00253] In certain embodiments, the oxygenator includes conductive components (e.g., Pt or nanocarbons), catalysts (e.g., iridium oxide) and structural supports.
[00254] In certain embodiment, applied materials will be fine-tuned to accomplish long-term stability (at least 60 days). The present invention generates highly controlled oxygen gradients to
support the bioactivities of loaded cells, and the produced oxygen allows maintenance of the viability of implanted therapeutic cells (>70%) and to manage tumor tissues chronically (at least 60 days).
[00255] In certain embodiments, to evaluate the effectiveness of the system, key metrics include: (1) oxygen production, (2) lack of byproduct formation, and (3) the maintenance of cell bioactivity/viability. Performance should be assessed as a function of time. Once the catalytic arrays are designed and fabricated, they are analyzed electrochemically.
[00256] In certain embodiment, linear sweep voltammetry (LSV) and chronoamperometry (CA) are essential analytic methods determining catalytic performances such as onset, overpotential and the stability of catalysts. Based on the basic characterization, the generated oxygen are measured at various potentials using oxygen-sensitive probes (e.g., optical oxygen sensor or Clark style probe).
[00257] Fig. 24A shows different geometries and coatings of the oxygenator, as well as the resulting changes in the current vs voltage applied. The present invention discloses oxygenator having higher current with lower voltage end, as that allows the oxygenator to generate oxygen with a lower power consumption. The lower panel of Fig. 24A shows the different models of oxygenator.
[00258] In certain embodiments, the oxygenator is a wire, either in a straight form or a coiled form. In certain embodiments, the oxygenator has a 6.6 mm diameter 11 turns Pt with Pt black deposition (Pt black) coiled form. In certain embodiments, the oxygenator has a 6.6 mm diameter 11 turns Pt coiled form. In certain embodiments, the oxygenator has a 4 cm Pt black straight form. In certain embodiments, the oxygenator has a 4 cm Pt straight form.
[00259] As shown in the upper panel of Fig. 24A, that coiling the wire and depositing Pt black increase the current generated at lower voltage the best (green line).
[00260] In certain embodiments, the oxygenator has a straight wire form having a length between about 3.0 cm - 3.5 cm. In certain embodiments, the oxygenator has a straight wire form having a length between about 3.5 cm - 4.0 cm. In certain embodiments, the oxygenator has a straight wire form having a length between about 4.5 cm - 4.5 cm. In certain embodiments, the oxygenator has a straight wire form having a length between about 4.5 cm - 5.0 cm.
[00261] In certain embodiments, the oxygenator has a coiled wire form having a diameter between 6.0-6.2 cm. In certain embodiments, the oxygenator has a coiled wire form having a
diameter between 6.2-6.4 cm. In certain embodiments, the oxygenator has a coiled wire form having a diameter between 6 4-6.6 cm. In certain embodiments, the oxygenator has a coiled wire form having a diameter between 6.6-6.8 cm. In certain embodiments, the oxygenator has a coiled wire form having a diameter between 6.8-7.0 cm. In certain embodiments, the oxygenator has a coiled wire form having a diameter between 7.0-7.2 cm.
[00262] In certain embodiments, the oxygenator has a coiled wire form having 5-10 turns. In certain embodiments, the oxygenator has a coiled wire form having 11-15 turns.
[00263] Fig. 24B shows in the upper panel that the oxygenator is capable of generating oxygen below 0.9 V applied to the device. This is significant because a voltage above 0.9 V leads to generation of toxic byproducts (e.g. chlorine), which impact the viability of the engineered cells, as shown in the lower panel.
[00264] Fig. 25 provides a schematic diagram, showing the structure of the fabricated hybrid device containing the oxygenator, the engineered cells, and the conductive scaffold for electrically stimulating the engineered cells.
[00265] In certain embodiments, the CE and WE are in a form of a mesh cylindrical structure as shown in Fig. 25. In certain embodiments, the shape and size of the mesh can be customized. In certain embodiments, the mesh is structured into a form other than cylindrical structure. The engineered cells are cultured in-between each CE and WE pair, and a current can be run between the CE and WE to stimulate the engineered cells, while the oxygenator locating in the center provides oxygen to the engineered cells.
[00266] In certain embodiments, the oxygenator run through the center of concentric cylinders containing the counter electrodes (CE) and the working electrode (WE). The engineered cells are cultured in-between each CE and WE pair, and a current can be run between the CE and WE to stimulate the engineered cells, while the oxygenator locating in the center provides oxygen to the engineered cells.
[00267] In certain embodiments, collagen sponge spacers are disposed between the CE and WE to maintain the electrode spacing for optimal stimulation, and preventing the shorting.
[00268] In certain embodiments, there is only one concentric cylinder separated by the WE and CE.
[00269] In certain embodiments, there are multiple concentric cylinders separated by multiple WEs and CEs, as shown in Fig. 25.
[00270] In one embodiment, the WE comprises multiple mesh cylindrical structure WEs and the CE comprises multiple mesh cylindrical structure CEs. Multiple mesh cylindrical structure WEs and the multiple mesh cylindrical structure CEs are arranged alternatively at intervals and in a concentric arrangement.
[00271] In certain embodiments, each of the concentric cylinders shown in Fig. 25 houses various cell populations, each of the cell populations produces different therapeutic agents, e.g., immunomodulatory cytokines.
[00272] In certain embodiment, the Pt conductive mesh is modified to Pt black to increase the surface area.
[00273] In certain embodiment, the implantable device has a length between about 3.0 cm -3.5 cm. In certain embodiment, the implantable device has a length between about 3.5 cm -4 cm. In certain embodiment, the implantable device has a length between about 4.0 cm -4.5 cm.
[00274] In certain embodiments, the cell stimulation chamber within the implantable device has a length between about 1.4 cm -1.6 cm. In certain embodiments, the cell stimulation chamber within the implantable device has a length between about 1.6 cm -1.8 cm. In certain embodiments, the cell stimulation chamber within the implantable device has a length between about 1.8 cm -2.0 cm. In certain embodiments, the cell stimulation chamber within the implantable device has a length between about 2.0 cm -2.2 cm. In certain embodiments, the cell stimulation chamber within the implantable device has a length between about 2.2 cm -2.4 cm. [00275] In certain embodiments, the cell stimulation chamber within the implantable device has a diameter between about 0.8 cm - 0.9 cm. In certain embodiments, the cell stimulation chamber within the implantable device has a diameter between about 0.9 cm - 1.0 cm. In certain embodiments, the cell stimulation chamber within the implantable device has a diameter between about 1.0 cm - 1.1 cm. In certain embodiments, the cell stimulation chamber within the implantable device has a diameter between about 1.1 cm - 1.2 cm. In certain embodiments, the cell stimulation chamber within the implantable device has a diameter between about 1.2 cm - 1.3 cm.
[00276] In some embodiments, electrocatalytic oxygen evolution bioelectronics may be able to maintain both cellular viability and peptide-producibility with precisely controlled oxygen generation.
[00277] In some embodiments, to exploit the versatility and benefits of catalytic oxygen generators in biomedical applications, the capabilities of planar platforms to 3D spaces may be translated, interfacing with tissues in a “vasculature-like” manner.
[00278] In some embodiments, spatial and/or temporal oxygen distribution may be predicted and/or manipulated, enabled by simulation-driven geometry design. In some embodiments, prestress in the design structures may be computed, which allows for anticipation of structural deformation and/or geometrical conversion for producing out-of-plane 3D flexible electronics. [00279] In some embodiments, catalytic platforms may be able to form stable interfaces to tissues with minimized irritation. In some embodiments, catalytic platforms may be introduced on compliant substrates including, but not limited to, polymeric substrates, thin metal wires and/or meshes.
[00280] In some embodiments, flexible devices may be integrated into tissues.
[00281] In some embodiments, oxygen-generating catalytic arrays may be constructed on flexible substrates, and/or may take advantage of flexible biomedical devices.
[00282] In some embodiments, for effective and efficient oxygenation, the flexible platform may be fabricated as multi-side devices to support tissues/cells around the devices, without leaving oxygen-depleted areas.
[00283] In some embodiments, flexible 2-dimensional devices may map out 3-dimensional spaces with controlled pre-stress in the device construction.
[00284] In some embodiments, bioresorbability may facilitate surgical treatment and/or device implantation, since extra surgery may not be needed to remove implantation once objectives of the devices are accomplished.
[00285] In some embodiments, a flexible platform may be constructed utilizing additive manufacturing methods.
[00286] In some embodiments, 3D printed nano/microstructured catalytic materials may form high-aspect-ratio flexible structures with or without matrices.
[00287] In some embodiments, to maximize the catalytic activities and/or the area of interfaces, the porosity of fabricated structures may be modulated by processing conditions including, but not limited to, processing temperature, annealing time, the size of contents and/or the formulation of precursors.
[00288] In some embodiments, 3D catalytic platforms may manage tissue-level of cell clusters in the aspects of high cell density as well as large scales beyond the limitation of diffusion from 2D-based systems. 3D catalytic platforms may have large interfacial areas with environments, which may support this feature.
[00289] In some embodiments, flexibility of the system may be fine-tuned via manufacturing processes.
[00290] In some embodiments, the tailored mechanical properties may allow formation of stable biointerfaces with various types of tissues.
[00291] In some embodiments, flexible and/or 3-dimensional oxygen supply devices may enable not only the modulation of physiological processes such as revascularization, cell migration and/or inflammatory responses, but also support large scale tissue with high cell density by integration as artificial vasculatures for oxygen delivery.
Embodiment 4: Protein Engineering
[00292] In certain embodiments, the present invention discloses engineered versions of naturally occurring cytokines and clinical antibodies for integration into the HAMMR platform. The selectivity and pharmacokinetic properties of these molecules are tailored to the specifications required in this system.
[00293] The repertoire of naturally occurring proteins is finite, and many molecules induce multiple confounding effects, limiting their efficacy as therapeutics. The present invention implements a structure-based engineering approach to optimize the biochemical and biophysical properties of the cytokines and antibodies that are integrated into the HAMMR system. This interdisciplinary work at the interface of synthetic biology and protein engineering empowers the controlled delivery of immunomodulatory signals, both spatially and temporally. In particular, the present invention discloses engineering the properties of the interleukin-2 (IL-2) cytokine to bias its activities towards immune effector cells (i.e., CD4+ and CD8+ T cells and natural killer [NK] cells) and also engineer the constant domain of anti-CD40 agonist antibody to shorten its serum half-life and thereby restrict its local availability. In certain embodiments, a combination of directed evolution and protein design technologies are employed to ensure that the engineered cytokines and antibodies incorporated in the HAMMR are suited for the system.
[00294] In certain embodiments, for the engineered IL-2, signaling activity are assessed via activation assays carried on cytokine- responsive human NK cells. Phosphorylation of signal
transducer and activator of transcription 5 (STAT5) are used as a readout for IL-2 signaling. Binding studies are conducted on an Octet® biolayer interferometry instrument to verify the interactions between the engineered cytokine and its cognate receptor subunits. The interaction of the engineered anti-CD40 antibody with the neonatal Fc receptor (FcRn) are also assessed through biolayer interferometry studies using an Octet® instrument to confirm that the antibody does not interact with FcRn and thus does not experience extended serum half-life.
[00295] In certain embodiments, the present invention discloses engineered cytokine IL2 which is more potent in eliciting an immune response comparing to natural IL2. Generally, IL2 requires a very high dose to be medically effective, the present invention discloses an engineered cytokine IL2 whose dose is significantly reduced for being medically effective.
[00296] Natural IL2 generally binds to 2 receptors: one on CD8 T-cells, and one on Regulator T-cells (T Reg). T Reg binding of IL2 does not have a significant biological affect, while CD8 T- cell binding does. The present invention discloses an engineered IL2 molecule that does not bind to the T Reg receptor, thus biasing the IL2 to bind to the CD8 T-cell receptor, which causes a greater biological effect.
[00297] Fig. 26 shows a summary of constructs for the engineered IL2 in the upper panel. LN35 and LN15 are peptide linkers of either 35 amino acids or 15 amino acids in length. hIL-2 is human IL2, and the Vh and VI components are parts of an antibody. These antibody components are intended to sterically block the IL2 from binding to the T Reg receptor. Fig. 26 lower panel shows the receptors of the T Reg and the CD8 T-cells (effector). The IL-2Ralpha component is the component causes the steric hinderance on the receptor side.
[00298] Fig. 27 shows that the engineered constructs of Fig. 26 have normal CD8 T-cell binding capability (upper panel), while demonstrate reduced binding to the T Reg receptor (lower panel). In particular, the F10 mini ICs constructs (2HL and HL2) showed expected signaling responses (pSTAT5) on human primary cells, and comparable signaling to human IL-2 on CD8+ T cells, as well as weaker signaling compared to hIL-2 on Treg cells. Meanwhile, the control mini IC behaved comparably to hIL-2, as expected.
[00299] Fig. 28 substantiates the results achieved by the engineered IL2 constructs shown in Fig. 27 with a quantitative test, particularly, the EC50 ratio of the engineered IL2s binding to the receptors. This clearly shows that the engineered IL2s (2HL and HL2) now have a biased to bind to the CD8 T-cell receptor and cause a greater immune response with a lower dose.
Embodiment 5: Aptamer Based Sensor Data
[00300] In certain embodiments, the present invention discloses biochemical sensors that can resist or reduce biofouling over 60 days in complex tissue environments.
[00301] Biofouling is a major concern for development of implantable devices, especially with functional devices where long term performance requires calibration-free measurements.
[00302] The present invention discloses fouling-resist biosensors via multiple grounds.
[00303] In certain embodiments, the present invention designs a “housekeeping” sensor (designed not to bind, or one that binds a housekeeping molecule) and locate it in proximity to the working sensor that will account for the systematic drift caused by long-term implantation. For example, a BSA-bound DNA (similar to a scrambled aptamer) does not bind a target from biofluids, and can be used as an internal calibrant in the sensing system to account for drift and can play this role as a “control”.
[00304] In certain embodiments, the present invention builds sensors on nanostructured electrodes that are less prone to fouling due to their function to prevent cell surface adhesion. [00305] In certain embodiments, the present invention optimizes sensor longevity as a function of surface roughness by building micro-electrodes (dome, star/dendrite, cube shapes) decorated with nanostructures (pores carved out in nanoscale) using current potential amperometry (DCPA) to control electrode morphology.
[00306] In certain embodiments, the present invention assembles a library of surface coating molecules as self- assembled monolayers (SAM) spacers (e.g., Fluorosilane, bromotrimethylsilane, Zwitterion, and 8-mercapto-l -octanol (MCO)) that can help prevent fouling and examines their effectiveness in comparison to conventional spacers that are prone to bio-fouling and desorption
[00307] In certain embodiments, the present invention incorporates polymer coatings or polymeric micro scaffolds with optimized pore size to allow for target penetration.
Microelectrode array (MEA) with planar structure is fabricated using standard microfabrication techniques and will be used as sensors. Before applying the anti-fouling coating, the MEA of working electrode sensors are modified with specific biorecognition elements and backfilled with the spacer molecule mentioned above.
[00308] In certain embodiments, the present invention discloses aptamer-based sensors to sense targets relevant to peritoneal cancer. This particular sensor being testing is for sensing
Albumin, which is being used as a housekeeping gene. In certain embodiments, the aptamer sensors are engineered to sense other targets relevant to cancer as well, such as TNF-a.
[00309] As shown in Fig. 29A, the sensors are exposed to ascites obtained from cancer patients, which contain biomarkers from cancer as well as albumin. The three charts in Fig. 29A shows that the aptamer sensors work effectively. In particular, the difference in the current (I on the y-axis) of the dip at -0.25 E between the PBS and ascites conditions shows that the sensor is sensing Albumin in the ascites fluid. These three charts represent different applied frequencies (10, 60, and 120 Hz), which increases the robustness of the dataset.
[00310] Fig. 29B shows the same sensor placed in a solution of increasing concentrations of albumin. At -1000 micrograms/mL the sensor begins to detect the albumin, evident by the change in the normalized current (y-axis). A lower frequency applied typically results in a lower signal as the concentration increases, while a 60 or 120 frequency applied results in a higher signal as the concentration increases. This result is expected, and the delta between baseline normalized current and the normalized current when the sensor senses albumin is the important metric which demonstrates proper function of the aptamer.
[00311] However, a major complication with the aptamer-based sensors is that they degrade over time through various mechanisms. The present invention discloses a method to mitigate this degradation effect by coating the electrodes used in the aptamer-based sensors.
[00312] Fig. 30 shows the change in current density over times with different chemicals functionalized to the electrode surfaces. The ideal electrode demonstrates a stable current density over time. If the current density drops, it suggests that the electrode is unstable.
[00313] The upper panel and the lower panel represent the same data, with different time units on the x-axis. The upper panel demonstrates actual time on x-axis, and the lower panel demonstrates number of stimulation cycles the electrode went through.
[00314] Among all coating agents shown in Fig. 30, MUO-MCH, MUO-TA, and MHx3- MPC1 were picked as the optimal coatings as these electrodes had the most stable current density, chemical formulas of which are shown in Fig. 30.
[00315] In certain embodiments, the aptamer-based sensor has a coating of MUO-MCH. In certain embodiments, the aptamer-based sensor has a coating of MUO-TA. In certain embodiments, the aptamer-based sensor has a coating of MHx3-MPCl. In certain embodiments, the aptamer-based sensor has a coating of one of MPO, MCH, MPC, Z1A3, MHX:MPC (ratio
3: 1), BG53, BG42, MBO, MUO, TA, Z4A10, MUO & TA, MCH55, BG53, BG44. In certain embodiments, the aptamer-based sensor has a coating of a mixture comprises at least two of MPO, MCH, MPC, Z1A3, MHX:MPC (ratio 3:1), BG53, BG42, MBO, MUO, TA, Z4A10, MCH55, BG53, BG44.
Embodiment 6: Electronic Hardware of Integrated Component
[00316] Figs. 31-32 represent the electronic device which integrates all electronic components of the present invention.
[00317] In certain embodiment, the electronic device integrating the electronic components including the aptamer-based sensors, the oxygenator, and the electronic stimulator for the cells. This integration allows the communication between the electronic components, i.e. sensing the dose of the cancer therapeutics with the aptamer-based sensors, and then relay the dose information to the stimulator to either start stimulating the engineered cells for producing a specific dose of cancer therapeutics depending on the dose was sensed, or stop the cancer therapeutics production.
[00318] Fig. 31 shows a photograph of biosensor readout platform which has been implemented as a vertical stack for modularity. In certain embodiments, the biosensor readout platform has dimensions of 2 cm x 2 cm x 1.5 cm. In certain embodiment, the biosensor readout platform has a width between about 1-3 cm. In certain embodiment, the biosensor readout platform has a length between about 1-3 cm. In certain embodiment, the biosensor readout platform has a depth between about 1-3 cm.
[00319] In certain embodiments, the biosensor readout platform comprises multiple PCBs. In certain embodiments, the biosensor readout platform comprises a single PCB.
[00320] In certain embodiments, the electronic device includes a wireless communication component to provide data of the therapeutics and patient’s response to the therapeutics to an external hub, which shows the data the doctor/patient.
[00321] In certain embodiment, the wireless communication module also allows for wireless charging of a battery on the electronic device.
Embodiment 7: Software for Integrated Hardware
[00322] In certain embodiments, the present invention discloses a software which is used in association with the electronic device.
[00323] In certain embodiments, the software collects the data from the aptamer-based sensors, computes the amount and the dose of the cancer therapeutics needed, and relay this information to the electrical stimulator/conductive scaffold to turn the engineered cells on to secrete the cancer therapeutics at the needed amount and dose.
[00324] In certain embodiments, the software assists modeling the aptamer-based sensor decay and correcting for this decay.
[00325] Fig. 33 shows a schematic workflow process of the entire electronic device. The left side of Fig. 33, in particular, shows the software used by the controller for controlling therapeutics delivery and sensor degradation compensation, and power consumption control. [00326] Fig. 34 shows the degradation correction model that corrects for the natural decay in the aptamer-based sensors. In certain embodiments, the degradation correction model includes a polynomial model, which provides high accuracy and efficiency. In certain embodiments, the degradation correction model includes a neural network model which provides high generalizability. In certain embodiments, the degradation correction model combines the polynomial model and the neural network model, which allows for accurate prediction while reducing the need to calibrate and lowering the battery power consumption.
[00327] In certain embodiments, the FPGA is the hardware to house the software of Figs. 33- 34.
[00328] Fig. 35 shows a schematic diagram of the FPGA, and that the signal compensation software is implemented in the FPGA.
[00329] Fig. 36 shows a prototype of the FPGA board and its setup, where the “upstream machine” is simulating input into the FPGA (i.e. information obtained by an aptamer-based sensor), and the output of the FPGA is being read by the “logic analyzer” so as to determine that the FPGA is correctly accounting for the modeled degradation of the aptamer-based sensor.
Embodiment 8: Humanized Cancer Model Data
[00330] The present invention demonstrates that the cytokines produced by the engineered ARPE-19 cells can effectively treat cancer. In particular, the present invention uses humanized mouse models, in which human immune cells are grafted into the mice. This allows the present invention to get as close to a human immune system in a mouse model as possible.
[00331] In certain embodiments, the ARPE-19 cells that are engineered to secrete cytokines constitutively, rather than in response to electrical stimulation, to allow for ease of testing.
[00332] Fig. 37A shows a timeline of the mouse studies, where the mice are first injected with ovarian cancer (SKOV3) into the intraperitoneal space, then engrafted with human immune cells (PBMC injection), followed by implantation with the ARPE-19 cells engineered to secrete IL2 encapsulated in alginate. The tumors are luminescent, so their size can be tracked via flux. As shown in Fig. 37B that condition A, which is 12 IL2 capsules implanted, results in a decreased tumor burden.
[00333] Figs. 38A-B show immune cell populations of the mice. Most notable is that the delivered IL2 increases CD8+ T-cells, a T-cell known to be beneficial in fighting tumors. [00334] In certain embodiments, the tumors were injected into the lymph nodes of the mice instead of directly into the intraperitoneal space. This is intended to model metastasis cancer. [00335] For these studies of metastasis cancer, the present invention uses ARPE-19 cells that are engineered to secrete cytokines constitutively, rather than in response to electrical stimulation, to allow for ease of testing.
[00336] Fig. 39 shows the timeline of the mouse studies, where the mice are first injected with a cancer cell line, either ovarian, SKOV3 and IGROV, or pancreatic, AsPC-1 and BxPC-3, then engrafted with human immune cells (PBMC injection), followed by implantation with the ARPE-19 cells engineered to secrete IL2 encapsulated in alginate, by surgery.
[00337] The tumors are luminescent, so their size can be tracked via flux.
[00338] Fig. 39 shows that cytokine treatment can effectively reduce tumor burden in AsPC-1 tumor mice (middle left panel).
[00339] Fig. 40 shows that cytokine treatment can effectively reduce tumor burden in SKOV3 tumor mice (middle left panel).
[00340] Fig. 41 shows immune cell composition across the mouse conditions. There are no consistent changes across conditions, suggesting that the immune response responsible for treatment of the cancer is not in the set of immune cells analyzed here.
Embodiment 9: Cancer Omics Data
[00341] In certain embodiments, the present invention discloses a method to analyze patient ascites data with omics to identify specifics cytokines/proteins that can be used as biomarkers of the cancer state. Any identified cytokines/proteins can be used as biomarkers for being sensed by the aptamer-based sensors, and therefore providing information regarding toxicity and efficacy of the cancer therapeutics delivered.
[00342] In certain embodiments, a scoring system is used to determine which cytokines/proteins are clinically relevant biomarkers for sensing the toxicity and efficacy of the cancer therapeutics.
[00343] In certain embodiments, the scoring system comprises three parts: a detection score, a clinical score, and a signaling score.
[00344] With respect to the detection score, in certain embodiments, the biomarker should be: (a) measurable in > about 50% of patients in ascites samples; (b) measurable in > about 50% of patients in peritoneal fluid samples; (c) average observed concentration in ascites > about 100 pg/ml; (d) average observed concentration in peritoneal fluid > about 100 pg/ml.
[00345] With respect to the clinical score, in certain embodiments, the biomarker should have: (a) absolute difference between partial response (PR) and stable disease (SD) patients at baseline (C0D1); (b) absolute difference after combination therapy treatment (C3D1) versus at baseline (C0D1); (c) absolute difference between PR and SD after treatment; (d) absolute difference in change between PR and SD after treatment.
[00346] With respect to the signaling score, in certain embodiments, the biomarker should have/be: (1) correlation between observed average concentration measured by Luminex assay and scRNA-seq expression; (b) expressed in tumor cells of more than about 50% of ascites patient samples; (c) average CellChat signaling probability from cancer cells to immune cells;
(d) average Cytosig score (cytokine induced signaling) in immune cells.
[00347] Using the scoring system described above, the present invention screened 147 molecules in total, with 80 from Luminex assay; 17 from Cytosig database, and 50 from CellChat secretory ligand database.
[00348] Table 2 shows the top 30 molecules with highest total scores when analyzing ascites from ovarian cancer patients.
Table 2. Top 30 molecules with highest total score based on detectability, clinical relevance, and signaling effects.
[00349] Fig. 42 shows a chart reflecting these molecule on the signaling and clinical score for ease of comparison and visualization. Galectin-3, MIF, IL-8, IL-2R, VEGF-A, TWEAK, APRIL, and PTX3 were all identified as good candidate molecules for sensing.
[00350] Fig. 43 shows an additional metric that was added to the 3 scores described above. In particular, this additional metric is using a large language model to predict the effects of various cytokines, e g. clinical relevance and functional effect from literature databases. In particular, Fig. 43 demonstrates how the model work: (1) the cytokines analyzed are searched for on literature databases, e.g. PubMed, (2) the top 30 relevant papers for each cytokine are retrieved, and (3) associations are extracted from the manuscripts to determine the role the cytokines as the tumor progresses.
[00351] Fig. 44 shows the data generated by the large language model, and the important sensing biomarkers identified. MIF, MCP-1, and IL-6 were the top molecules suggested by the model that would be important biomarkers for being sensed.
[00352] In certain embodiments, one of Galectin-3, MIF, IL-8, IL-2R, VEGF-A, TWEAK, APRIL, PTX3, MIF, MCP-1, and IL-6 is sensed by the aptamer-based sensor for determining the toxicity and/or efficacy of the cancer therapeutics. In certain embodiments, a combination of any of Galectin-3, MIF, IL-8, IL-2R, VEGF-A, TWEAK, APRIL, PTX3, MIF, MCP-1, and IL-6 is sensed by the aptamer-based sensor for determining the toxicity and/or efficacy of the cancer therapeutics.
Embodiment 10: Mathematics and Computation Modeling of the Electronic Device [00353] In certain embodiments, the present invention discloses an overall mathematical model of the engineered ARPE-19 cells in the stimulation environment. This overall mathematical model assist the controlled of the electronic device to determine the degree of the stimulation for the engineered cells to produce the desired amount / dose of the cancer therapeutics.
[00354] Fig. 45 depicts an overview of the mathematical model. The left side shows a finite element/electrophysiology model relating stimulus to membrane depolarization, which is a physics-based model. The right side shows a cell signaling model for relating membrane depolarization and protein production, which reflects the processes/reactions took places inside the engineered ARPE-19 cells after the cells are electrically stimulated, starting with the electroactive channels opening, and ending with cytokine secretion.
[00355] Figs. 46A-B show that this mathematical model predicted that increasing NF AT expression inside the cells will in turn increase production of cytokines from the engineered ARPE-19 cells. Fig. 47 shows that the predicted production by the mathematical model was correct. The upper panel shows that increasing NFAT3 in the engineered ARPE-19 cells does result increased production of IL2 by engineered ARPE-19 cells, while increasing the other NFATs, NF ATI and NFAT2, does not increase cellular production, suggesting NFAT3 is the most important.
[00356] As shown in Fig. 48, the mathematical model also predicted that a 3 mm distance between electrodes for stimulation would result in the greatest stimulation of the engineered ARPE-19 cells, by comparing the bottom right chart with the top left chart. This prediction was validated in Fig. 18B, with 3 mm distance between the electrodes results in the greatest production from the cells.
[00357] The foregoing description of the exemplary embodiments of the invention has been presented only for the purposes of illustration and description and is not intended to be exhaustive or to limit the invention to the precise forms disclosed. Many modifications and variations are possible in light of the above teaching.
[00358] While there has been shown several and alternate embodiments of the present invention, it is to be understood that certain changes can be made as would be known to one skilled in the art without departing from the underlying scope of the invention as is discussed and
set forth above and below including claims and drawings. Furthermore, the embodiments described above and claims set forth below are only intended to illustrate the principles of the present invention and are not intended to limit the scope of the invention to the disclosed elements.
[00359] Some references, which may include patents, patent applications and various publications, are cited and discussed in the description of this invention is incorporated herein by reference in its entireties for background information only. The citation and/or discussion of such references is provided merely to clarify the description of the present invention and is not an admission that any such reference is “prior art” to the invention described herein. All references cited and discussed in the description of this invention are incorporated herein by reference in their entireties and to the same extent as if each reference was individually incorporated by reference.
Claims
1. A platform of a hybrid advanced molecular manufacturing regulator containing engineered cells delivering cancer therapeutics to a subject, the platform comprising: an implantable device disposed inside an internal cavity of a subject; and an external device disposed outside the internal cavity of the subject and in wireless communication with the implantable device, wherein the implantable device comprises a molecular manufacturing regulator and a communication module; wherein the external device comprises at least one transmission coil.
2. The platform according to claim 1, wherein the communication module comprises at least one antenna.
3. The platform according to claim 2, wherein the at least one antenna comprises a magnetoelectric (ME) component.
4. The platform according to claim 3, wherein the ME component comprises at least one magnetostrictive layer attached to a piezoelectric layer.
5. The platform according to claim 4, wherein the at least one magnetostrictive layer comprises multiple magnetostrictive layers.
6. The platform according to claim 4, wherein the at least one antenna is in wireless communication with the at least one transmission coil.
7. The platform according to claim 6, wherein the at least one antenna is configured to
receive a power transmission from the at least one transmission coil.
8. The platform according to claim 7, wherein the implantable device comprises a battery, wherein the battery is charged by the power transmission.
9. The platform according to claim 6, wherein the at least one antenna is configured to receive a download information transmission from the at least one transmission coil.
10. The platform according to claim 6, wherein the at least one transmission coil is configured to receive an upload information transmission from the at least one antenna.
11. The platform according to claim 10, wherein the at least one antenna comprises multiple sub-antennas.
12. The platform according to claim 11, wherein each of the multiple sub-antennas are configured to receive the power transmission and the download information transmission, and provides the upload information transmission to the at least one transmission coil.
13. The platform according to claim 6, wherein the at least one transmission coil comprises a power transmitter coil and a receiver pick-up coil.
14. The platform according to claim 13, wherein the at least one antenna is configured to receive a power transmission from the power transmitter coil.
15. The platform according to claim 13, wherein the receiver pick-up coil is configured to receive an upload information transmission from the at least one antenna, and the at least one antenna is configured to receive a download information transmission from the receiver pick-up coil.
16. The platform according to claim 10, wherein the molecular manufacturing regulator comprises a conductive scaffold in communication with a control module; a biochemical sensor
module in communication with the control module; an oxygenator configured to produce oxygen and disposed inside the conductive scaffold; and at least one engineered cells encapsulation disposed in association with the conductive scaffold; wherein the at least one engineered cells encapsulation encapsulates engineered cells; wherein the engineered cells are configured to produce at least one cancer therapeutic agent.
17. The platform according to claim 16, wherein the conductive scaffold is configured to produce an electrical pulse, wherein the conductive scaffold is powered by the battery.
18. The platform according to claim 16, wherein the control module comprises a machine learning algorithm for optimizing at least one of the power transmission, the upload information transmission, and the download information transmission.
19. The platform according to claim 16, wherein the implantable device comprises at least one of a field-programmable gate array (FPGA) circuit, an application specific integrated circuit (ASIC), and a microcontroller circuit; wherein the at least one of the FPGA, the ASIC, and the microcontroller circuit coordinates and controls an electrical stimulation of the conductive scaffold which is optimized for a production efficiency of the at least one cancer therapeutic agent and/or a longevity of battery.
20. The platform according to claim 1, wherein the implantable device is powered by a primary cell battery.
21. The platform according to claim 20, wherein the primary cell battery is a lithium-based battery configured to provide a long-term power to the implantable device.
22. The platform according to claim 1, wherein the communication module of the implantable device comprises a Bluetooth communication module for wireless transmission to the external device.
23. The platform according to claim 22, wherein the communication module is configured to
transmit and receive data related to an operational status of the molecular manufacturing regulator.
24. The platform according to claim 23, wherein the communication module of the implantable device incorporates a proprietary radio protocol for wireless communication with the external device.
25. The platform according to claim 24, wherein the proprietary radio protocol is configured to transmit and receive data related to the operational status of the molecular manufacturing regulator.
26. The platform according to claim 24, wherein the proprietary radio protocol is optimized for low-power operation to extend a life of a primary cell battery powering the implantable device.
27. The platform according to claim 1, wherein a primary cell battery is configured to power both the molecular manufacturing regulator and the communication module.
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US202363539658P | 2023-09-21 | 2023-09-21 | |
| US63/539,658 | 2023-09-21 | ||
| US202363540004P | 2023-09-22 | 2023-09-22 | |
| US63/540,004 | 2023-09-22 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2025064982A1 true WO2025064982A1 (en) | 2025-03-27 |
Family
ID=95072230
Family Applications (4)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2024/047951 Pending WO2025064982A1 (en) | 2023-09-21 | 2024-09-23 | Platform of hybrid advanced molecular manufacturing regulator and applications thereof |
| PCT/US2024/047943 Pending WO2025064980A1 (en) | 2023-09-21 | 2024-09-23 | Engineered cells producing oncotherapeutics and applications thereof |
| PCT/US2024/047927 Pending WO2025064970A1 (en) | 2023-09-21 | 2024-09-23 | Targeted hybrid oncotherapeutics regulation system and applications thereof |
| PCT/US2024/047934 Pending WO2025064974A1 (en) | 2023-09-21 | 2024-09-23 | Hybrid advanced molecular manufacturing regulator and applications thereof |
Family Applications After (3)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2024/047943 Pending WO2025064980A1 (en) | 2023-09-21 | 2024-09-23 | Engineered cells producing oncotherapeutics and applications thereof |
| PCT/US2024/047927 Pending WO2025064970A1 (en) | 2023-09-21 | 2024-09-23 | Targeted hybrid oncotherapeutics regulation system and applications thereof |
| PCT/US2024/047934 Pending WO2025064974A1 (en) | 2023-09-21 | 2024-09-23 | Hybrid advanced molecular manufacturing regulator and applications thereof |
Country Status (1)
| Country | Link |
|---|---|
| WO (4) | WO2025064982A1 (en) |
Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20160270729A1 (en) * | 2013-11-17 | 2016-09-22 | Ramot At Tel-Aviv University Ltd. | Electronic scaffold and uses thereof |
| US20190101551A1 (en) * | 2016-03-21 | 2019-04-04 | The Regents Of The University Of California | Real-time and Continuous Measurement in Vivo Using Aptamer-Based Biosensors |
| WO2022140578A1 (en) * | 2020-12-24 | 2022-06-30 | Conductivebio Incorporated | Conductive scaffolds formed by absorbable composite biomaterials and use thereof |
| WO2022226146A1 (en) * | 2021-04-21 | 2022-10-27 | Northwestern University | Hybrid bioelectronic/engineered cell implantable system for therapeutic agents delivery and applications thereof |
| WO2022266234A1 (en) * | 2021-06-15 | 2022-12-22 | William Marsh Rice University | Methods for in vivo delivery of microbes to human microenvironments |
Family Cites Families (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| RU2009132949A (en) * | 2007-02-02 | 2011-03-10 | Юниверсити Оф Майами (Us) | THERAPEUTIC HYBRID IMPLANTED DEVICES |
| WO2017181027A1 (en) * | 2016-04-14 | 2017-10-19 | The Regents Of The University Of California | Closed-loop actuating and sensing epidermal systems |
| MX2019000677A (en) * | 2016-07-18 | 2019-06-10 | Eth Zuerich | B-cell-mimetic cells. |
| AU2018285539B2 (en) * | 2017-06-14 | 2024-10-31 | Vertex Pharmaceuticals Incorporated | Devices and methods for delivering therapeutics |
| US11834640B2 (en) * | 2019-09-15 | 2023-12-05 | Board Of Regents, The University Of Texas System | Device and uses thereof for treating diabetes |
-
2024
- 2024-09-23 WO PCT/US2024/047951 patent/WO2025064982A1/en active Pending
- 2024-09-23 WO PCT/US2024/047943 patent/WO2025064980A1/en active Pending
- 2024-09-23 WO PCT/US2024/047927 patent/WO2025064970A1/en active Pending
- 2024-09-23 WO PCT/US2024/047934 patent/WO2025064974A1/en active Pending
Patent Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20160270729A1 (en) * | 2013-11-17 | 2016-09-22 | Ramot At Tel-Aviv University Ltd. | Electronic scaffold and uses thereof |
| US20190101551A1 (en) * | 2016-03-21 | 2019-04-04 | The Regents Of The University Of California | Real-time and Continuous Measurement in Vivo Using Aptamer-Based Biosensors |
| WO2022140578A1 (en) * | 2020-12-24 | 2022-06-30 | Conductivebio Incorporated | Conductive scaffolds formed by absorbable composite biomaterials and use thereof |
| WO2022226146A1 (en) * | 2021-04-21 | 2022-10-27 | Northwestern University | Hybrid bioelectronic/engineered cell implantable system for therapeutic agents delivery and applications thereof |
| WO2022266234A1 (en) * | 2021-06-15 | 2022-12-22 | William Marsh Rice University | Methods for in vivo delivery of microbes to human microenvironments |
Also Published As
| Publication number | Publication date |
|---|---|
| WO2025064970A1 (en) | 2025-03-27 |
| WO2025064980A1 (en) | 2025-03-27 |
| WO2025064974A1 (en) | 2025-03-27 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US20240207508A1 (en) | Hybrid bioelectronic/engineered cell implantable system for therapeutic agents delivery and applications thereof | |
| Wikswo et al. | Engineering challenges for instrumenting and controlling integrated organ-on-chip systems | |
| CN101184989B (en) | Devices and methods for use in assessing a flow condition of a fluid | |
| Park et al. | Materials chemistry of neural interface technologies and recent advances in three-dimensional systems | |
| JP6858769B2 (en) | Cell culture, transport, and investigation | |
| US20110251471A1 (en) | Implantable power sources and sensors | |
| Baj-Rossi et al. | Full fabrication and packaging of an implantable multi-panel device for monitoring of metabolites in small animals | |
| US20200369996A1 (en) | Bioreactor Screening Platform for Modelling Human Systems Biology and for Screening for Inotropic Effects of Agents on the Heart | |
| Pankratov et al. | Ex vivo electric power generation in human blood using an enzymatic fuel cell in a vein replica | |
| Zajdel et al. | Come together: On-chip bioelectric wound closure | |
| WO2007124448A2 (en) | Method and apparatus for the management of diabetes | |
| Bhave et al. | Distributed sensor and actuator networks for closed-loop bioelectronic medicine | |
| Cao et al. | Graphene oxide based recyclable in vivo device for amperometric monitoring of interferon-γ in inflammatory mice | |
| Rivnay et al. | Integrating bioelectronics with cell-based synthetic biology | |
| Zou et al. | Active chemical messenger-driven immune activation via electrochemical patch for in-situ tumor vaccination | |
| Wei et al. | Stability enhancement of galvanic redox potentiometry by optimizing the redox couple in counterpart poles | |
| WO2025064982A1 (en) | Platform of hybrid advanced molecular manufacturing regulator and applications thereof | |
| Man et al. | Antidiabetic Close Loop Based on Wearable DNA–Hydrogel Glucometer and Implantable Optogenetic Cells | |
| US20210220558A1 (en) | Bio-electroceutical device using cell cluster | |
| Ding et al. | Tumor microenvironment–activated Zn//MnO2 battery for sustained and local electrochemical immunotherapy | |
| Fisher et al. | Controlling tissue microenvironments: biomimetics, transport phenomena, and reacting systems | |
| US20150343093A1 (en) | Controlled death of implanted cells | |
| US20230038480A1 (en) | System for produciton of high yield of recombinant proteins | |
| Zajdel et al. | Come together: bioelectric healing-on-a-chip | |
| WO2024091700A1 (en) | Hybrid bioelectronic/engineered cell implantable system for therapeutic agents delivery and applications thereof |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 24869365 Country of ref document: EP Kind code of ref document: A1 |