WO2025064492A1 - Trpm8 antagonist for treating cancer - Google Patents
Trpm8 antagonist for treating cancer Download PDFInfo
- Publication number
- WO2025064492A1 WO2025064492A1 PCT/US2024/047191 US2024047191W WO2025064492A1 WO 2025064492 A1 WO2025064492 A1 WO 2025064492A1 US 2024047191 W US2024047191 W US 2024047191W WO 2025064492 A1 WO2025064492 A1 WO 2025064492A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- cancer
- difluoro
- diazaspiro
- decane
- dione
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/495—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with two or more nitrogen atoms as the only ring heteroatoms, e.g. piperazine or tetrazines
- A61K31/50—Pyridazines; Hydrogenated pyridazines
- A61K31/501—Pyridazines; Hydrogenated pyridazines not condensed and containing further heterocyclic rings
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/13—Amines
- A61K31/135—Amines having aromatic rings, e.g. ketamine, nortriptyline
- A61K31/138—Aryloxyalkylamines, e.g. propranolol, tamoxifen, phenoxybenzamine
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/41—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with two or more ring hetero atoms, at least one of which being nitrogen, e.g. tetrazole
- A61K31/4164—1,3-Diazoles
- A61K31/4166—1,3-Diazoles having oxo groups directly attached to the heterocyclic ring, e.g. phenytoin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/435—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with one nitrogen as the only ring hetero atom
- A61K31/44—Non condensed pyridines; Hydrogenated derivatives thereof
- A61K31/445—Non condensed piperidines, e.g. piperocaine
- A61K31/4523—Non condensed piperidines, e.g. piperocaine containing further heterocyclic ring systems
- A61K31/454—Non condensed piperidines, e.g. piperocaine containing further heterocyclic ring systems containing a five-membered ring with nitrogen as a ring hetero atom, e.g. pimozide, domperidone
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/495—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with two or more nitrogen atoms as the only ring heteroatoms, e.g. piperazine or tetrazines
- A61K31/505—Pyrimidines; Hydrogenated pyrimidines, e.g. trimethoprim
- A61K31/506—Pyrimidines; Hydrogenated pyrimidines, e.g. trimethoprim not condensed and containing further heterocyclic rings
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/495—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with two or more nitrogen atoms as the only ring heteroatoms, e.g. piperazine or tetrazines
- A61K31/505—Pyrimidines; Hydrogenated pyrimidines, e.g. trimethoprim
- A61K31/519—Pyrimidines; Hydrogenated pyrimidines, e.g. trimethoprim ortho- or peri-condensed with heterocyclic rings
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/70—Carbohydrates; Sugars; Derivatives thereof
- A61K31/7042—Compounds having saccharide radicals and heterocyclic rings
- A61K31/7052—Compounds having saccharide radicals and heterocyclic rings having nitrogen as a ring hetero atom, e.g. nucleosides, nucleotides
- A61K31/706—Compounds having saccharide radicals and heterocyclic rings having nitrogen as a ring hetero atom, e.g. nucleosides, nucleotides containing six-membered rings with nitrogen as a ring hetero atom
- A61K31/7064—Compounds having saccharide radicals and heterocyclic rings having nitrogen as a ring hetero atom, e.g. nucleosides, nucleotides containing six-membered rings with nitrogen as a ring hetero atom containing condensed or non-condensed pyrimidines
- A61K31/7068—Compounds having saccharide radicals and heterocyclic rings having nitrogen as a ring hetero atom, e.g. nucleosides, nucleotides containing six-membered rings with nitrogen as a ring hetero atom containing condensed or non-condensed pyrimidines having oxo groups directly attached to the pyrimidine ring, e.g. cytidine, cytidylic acid
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K33/00—Medicinal preparations containing inorganic active ingredients
- A61K33/24—Heavy metals; Compounds thereof
- A61K33/243—Platinum; Compounds thereof
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
Definitions
- Cancer is characterized by the development of abnormal cells that divide uncontrollably and have the ability to infiltrate and destroy normal body tissue. Cancer is a major public health problem in many parts of the world, which is the second-leading cause of death around the global. The survival rates are, however, improving for many types of cancer, due to the current advancement in cancer screening, treatment, and prevention.
- Transient receptor potential (TRP) channels are one of the largest groups of ion channels, and they are divided into 6 sub-families (TRPV, TRPM, TRPA, TRPC, TRPP and TRPMU). TRP channels are cation-selective channels that are activated by a variety of physical (e.g., temperature, osmolarity, mechanical) and chemical stimuli. Transient receptor potential melastatin-subfamily member 8 (TRPM8) is a member of TRP channel family.
- TRPM8 can sense temperature changes in the range of both innocuous cold (15-28 °C) (McKeny et al Nature 2002, 416, 52; Peier et al Cell 2002, 108, 705) and noxious cold ( ⁇ 15°C) as well as by chemical agents such as menthol and icilin (Chuang et al Neuron 2004, 43, 859).
- TRPM8 was expressed in various cancers, especially metastatic and resistant cancers.
- an increasing number of literatures have shown that TRPM8 involves in the initiation and progression of tumors and study found that TRPM8 expression upregulated markedly in prostate cancer (PC) and in many other tumors, suggesting an important role in carcinogenesis in human cancers.
- PC prostate cancer
- TRPM8 which plays prominent role in thermoregulation, could be one of the most promising novel therapeutic targets in cancer treatment.
- Compound 1 is in the form of a salt.
- Compound 1 is in the form of a besylate salt.
- the cancer is bladder cancer, breast cancer, cholangiocarcinoma, colorectal cancer, kidney cancer, liver cancer, lung cancer, lymphoma, myeloma, osteosarcoma, ovarian cancer, pancreatic cancer, prostate cancer, or skin cancer.
- the breast cancer is ER positive breast cancer, ER positive metastatic breast cancer, or triple negative breast cancer (TNB).
- TNB triple negative breast cancer
- the myeloma is multiple myeloma (MM).
- the skin cancer is melanoma.
- the lung cancer is small cell lung cancer (SCLC) or non-small cell lung cancer (NSCLC).
- SCLC small cell lung cancer
- NSCLC non-small cell lung cancer
- the method further comprises administering an additional therapeutic agent.
- the additional therapeutic agent is a targeted agent, a chemotherapy, an immunotherapy (IO), radiation therapy, a gene therapy, or a cell therapy, or any combination thereof.
- the additional therapeutic agent is a targeted agent selected from the group consisting of apalutamide, dabrafenib, enzalutamide, fulvestrant, ibrutinib, lenalidomide, palbociclib, pomalidomide, sorafenib, tamoxifen, and thalidomide.
- the additional therapeutic agent is a chemotherapy agent selected from the group consisting of 5 -fluorouracil, bleomycin, capecitabine, carboplatin, cisplatin, cyclophosphamide, dacarbazine, docetaxel, doxorubicin, epirubicin, etoposide, etoposide, folinic acid, gemcitabine, ifosfamide, methotrexate, mustine, oxaliplatin, prednisolone, procarbazine, vinorelbine, and vinorelbine.
- the additional therapeutic agent is a CTLA-4 blocker, a PD-1 blocker, or a PD-L1 blocker.
- the CTLA-4 blocker is ipilimumab or tremelimumab.
- the PD-1 blocker is nivolumab or pembrolizumab.
- the PD-L1 blocker is atezolizumab, avelumab, cemiplimab, or durvalumab.
- FIG. 1 shows the cellular proliferation inhibition in KMS-26 cell line of Compound 1, lenalidomide, and Compound 1 combined with lenalidomide.
- FIG. 2 shows the cellular proliferation inhibition in JJN-3 cell line of Compound 1, lenalidomide, and Compound 1 combined with lenalidomide.
- FIG. 3 shows the cellular proliferation inhibition in OPM-2 cell line of Compound 1, lenalidomide, and Compound 1 combined with lenalidomide.
- FIG. 4 shows the cellular proliferation inhibition in MCF7 cell line of Compound 1, tamoxifen, and Compound 1 combined with tamoxifen.
- FIG. 5 shows the cellular proliferation inhibition in MDA-MB-231 cell line that Compound 1, gemcitabine, and Compound 1 combined with gemcitabine.
- FIG. 6 shows the cellular proliferation inhibition in HMC-1-8 cell line that Compound 1, sorafenib, and Compound 1 combined with sorafenib.
- FIG. 7 shows the cellular proliferation inhibition in SK-MEL-28 cell line that Compound 1, dabrafenib, and Compound 1 combined with dabrafenib.
- FIG. 8 shows the cellular proliferation inhibition in A101D cell line that Compound 1, dabrafenib, and Compound 1 combined with dabrafenib.
- FIG. 9 shows the cellular proliferation inhibition in Hs 294T cell line that Compound 1, dabrafenib, and Compound 1 combined with dabrafenib.
- FIG. 10 shows the cellular proliferation inhibition in MDA-MB-435S cell line that Compound 1, dabrafenib, and Compound 1 combined with dabrafenib.
- FIG. 11 shows the cellular proliferation inhibition in HOS cell line that Compound 1, cisplatin, and Compound 1 combined with cisplatin.
- FIG. 12 shows the cellular proliferation inhibition in Saos-2 cell line that Compound 1, cisplatin, and Compound 1 combined with cisplatin.
- FIG. 13 shows the cellular proliferation inhibition in NCI-H2081 cell line that Compound 1, cisplatin, and Compound 1 combined with cisplatin.
- FIG. 14 shows the cellular proliferation inhibition in ACHN cell line that Compound 1, sorafenib, and Compound 1 combined with sorafenib.
- FIG. 15 shows the cellular proliferation inhibition in G-402 cell line that Compound 1, sorafenib, and Compound 1 combined with sorafenib.
- Examples include, but are not limited to methyl, ethyl, n-propyl, isopropyl, 2 -methyl- 1 -propyl, 2 -methyl -2 -propyl, 2-methyl-l- butyl, 3 -methyl- 1 -butyl, 2-methyl-3-butyl, 2,2-dimethyl-l -propyl, 2-methyl-l -pentyl, 3 -methyl- 1 -pentyl, 4-methyl-l -pentyl, 2-methyl-2-pentyl, 3 -methyl -2 -pentyl, 4-methyl-2-pentyl, 2,2-dimethyl-l -butyl, 3,3- dimethyl-1 -butyl, 2 -ethyl- 1 -butyl, n-butyl, isobutyl, sec-butyl, t-butyl, n-pentyl, isopentyl, neopentyl, tert-
- C2-C6 alkenyl means that the alkenyl group may consist of 2 carbon atoms, 3 carbon atoms, 4 carbon atoms, 5 carbon atoms or 6 carbon atoms, although the present definition also covers the occurrence of the term “alkenyl” where no numerical range is designated.
- an alkenyl group may be optionally substituted, for example, with one or more oxo, halogen, amino, nitrile, nitro, hydroxyl, alkyl, haloalkyl, alkoxy, carboxyl, carboxylate, aryl, cycloalkyl, heterocycloalkyl, or heteroaryl, and the like.
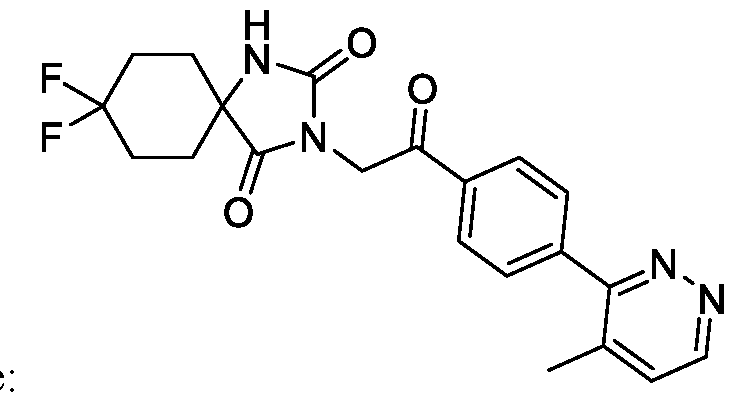
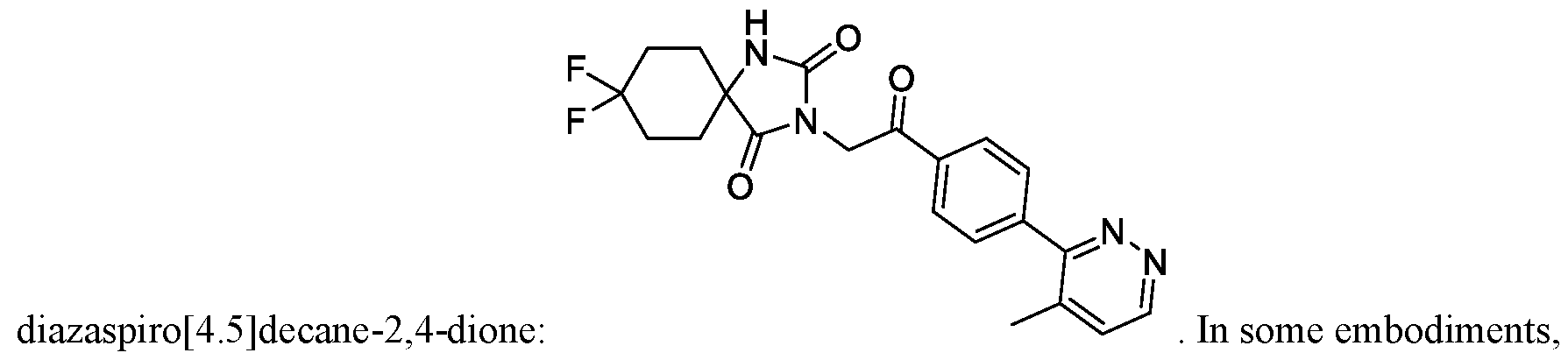
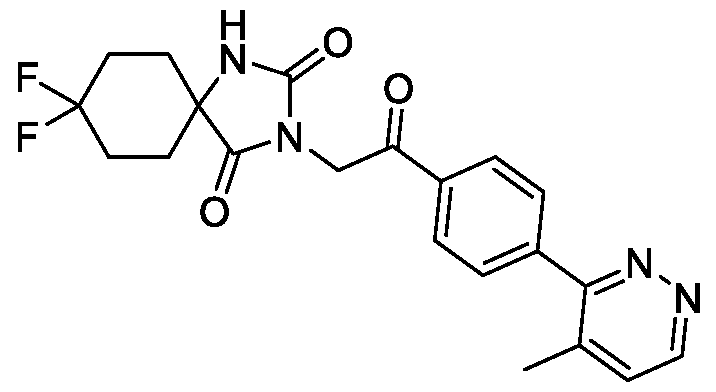
- the TRPM8 antagonist is Compound 1, or a pharmaceutically acceptable salt thereof.
- Compound 1 is 8, 8-difluoro-3-(2-(4-(4-methylpyridazin-3-yl)phenyl)-2 -oxoethyl)-!, 3-
- Compound 1 is in the form of a salt. In some embodiments, Compound 1 is in the form of a besylate salt.
- the TRPM8 antagonist is AMG-333, elismetrep, M8-B, PF-05105679,
- the TRPM8 antagonist i i
- the compounds described herein exist as geometric isomers. In some embodiments, the compounds described herein possess one or more double bonds. The compounds presented herein include all cis, trans, syn, anti,
- Z isomers as well as the corresponding mixtures thereof. In some situations, the compounds described herein possess one or more chiral centers and each center independently exists in the R configuration or S configuration. The compounds described herein include all diastereomeric, enantiomeric, and epimeric forms as well as the corresponding mixtures thereof.
- mixtures of enantiomers and/or diastereoisomers, resulting from a single preparative step, combination, or interconversion are useful for the applications described herein.
- the compounds described herein are prepared as their individual stereoisomers by reacting a racemic mixture of the compound with an optically active resolving agent to form a pair of diastereoisomeric compounds, separating the diastereomers and recovering the optically pure enantiomers.
- dissociable complexes are preferred.
- the diastereomers have distinct physical properties (e.g., melting points, boiling points, solubilities, reactivity, etc.) and are separated by taking advantage of these dissimilarities.
- the diastereomers are separated by chiral chromatography, or preferably, by separation/resolution techniques based upon differences in solubility.
- the optically pure enantiomer is then recovered, along with the resolving agent, by any practical means that would not result in racemization.
- the compounds described herein exist in their isotopically -labeled forms.
- the methods disclosed herein include methods of treating diseases by administering such isotopically-labeled compounds.
- the methods disclosed herein include methods of treating diseases by administering such isotopically-labeled compounds as pharmaceutical compositions.
- the compounds disclosed herein include isotopically-labeled compounds, which are identical to those recited herein, but for the fact that one or more atoms are replaced by an atom having an atomic mass or mass number different from the atomic mass or mass number usually found in nature.
- isotopes examples include isotopes of hydrogen, carbon, nitrogen, oxygen, phosphorous, sulfur, fluorine, and chlorine, such as 2 H, 3 H, 13 C, 14 C, 15 N, 18 0, 17 0, 31 P, 32 P, 35 S, 18 F, and 36 C1, respectively.
- Compounds described herein, and the pharmaceutically acceptable salts thereof which contain the aforementioned isotopes and/or other isotopes of other atoms are within the scope of this invention.
- Certain isotopically-labeled compounds for example those into which radioactive isotopes such as 3 H and 14 C are incorporated, are useful in drug and/or substrate tissue distribution assays.
- Tritiated, i.e., 3 H and carbon-14, i.e., 14 C, isotopes are particularly preferred for their ease of preparation and detectability. Further, substitution with heavy isotopes such as deuterium, i.e., 2 H, produces certain therapeutic advantages resulting from greater metabolic stability, for example increased in vivo half-life or reduced dosage requirements.
- one or more hydrogen in a compound disclosed herein has been replaced by a deuterium atom.
- one or more alkyl substituents in a compound disclosed herein has been replaced by a deuteroalkyl substituents.
- the compounds described herein are labeled by other means, including, but not limited to, the use of chromophores or fluorescent moieties, biolumine scent labels, or chemiluminescent labels.
- the compounds described herein exist as their pharmaceutically acceptable salts.
- the methods disclosed herein include methods of treating diseases by administering such pharmaceutically acceptable salts.
- the methods disclosed herein include methods of treating diseases by administering such pharmaceutically acceptable salts as pharmaceutical compositions.
- the compounds described herein possess acidic or basic groups and therefore react with any of a number of inorganic or organic bases, and inorganic and organic acids, to form a pharmaceutically acceptable salt.
- these salts are prepared in situ during the final isolation and purification of the compounds disclosed herein, or by separately reacting a purified compound in its free form with a suitable acid or base, and isolating the salt thus formed.
- Examples of pharmaceutically acceptable salts include those salts prepared by reaction of the compounds described herein with a mineral, organic acid or inorganic base, such salts including, but not limited to, acetate, acrylate, adipate, alginate, aspartate, benzoate, benzenesulfonate, bisulfate, bisulfite, bromide, butyrate, butyn-l,4-dioate, camphorate, camphorsulfonate, caproate, caprylate, chlorobenzoate, chloride, citrate, cyclopentanepropionate, decanoate, digluconate, gluconate, dihydrogenphosphate, dinitrobenzoate, dodecylsulfate, ethanesulfonate, formate, fumarate, glucoheptanoate, glycerophosphate, glycolate, hemisulfate, heptanoate, hexanoate, hexyn
- the compounds described herein can be prepared as pharmaceutically acceptable salts formed by reacting the free base form of the compound with a pharmaceutically acceptable inorganic or organic acid, including, but not limited to, inorganic acids such as hydrochloric acid, hydrobromic acid, sulfuric acid, nitric acid, phosphoric acid metaphosphoric acid, and the like; and organic acids such as acetic acid, propionic acid, hexanoic acid, cyclopentanepropionic acid, glycolic acid, pyruvic acid, lactic acid, malonic acid, succinic acid, malic acid, maleic acid, fumaric acid, p-toluenesulfonic acid, tartaric acid, trifluoroacetic acid, citric acid, benzoic acid, 3-(4-hydroxybenzoyl)benzoic acid, cinnamic acid, mandelic acid, arylsulfonic acid, methane sulfonic acid, ethanesulfonic acid, 1,
- those compounds described herein which comprise a free acid group react with a suitable base, such as the hydroxide, carbonate, bicarbonate, sulfate, of a pharmaceutically acceptable metal cation, with ammonia, or with a pharmaceutically acceptable organic primary, secondary, tertiary, or quaternary amine.
- a suitable base such as the hydroxide, carbonate, bicarbonate, sulfate, of a pharmaceutically acceptable metal cation, with ammonia, or with a pharmaceutically acceptable organic primary, secondary, tertiary, or quaternary amine.
- Representative salts include the alkali or alkaline earth salts, like lithium, sodium, potassium, calcium, and magnesium, and aluminum salts and the like.
- bases include sodium hydroxide, potassium hydroxide, choline hydroxide, sodium carbonate, N + (CI.C4 alkyl)4 hydroxide, and the like.
- Representative organic amines useful for the formation of base addition salts include ethylamine, diethylamine, ethylenediamine, ethanolamine, diethanolamine, piperazine and the like. It should be understood that the compounds described herein also include the quatemization of any basic nitrogencontaining groups they contain. In some embodiments, water or oil-soluble or dispersible products are obtained by such quatemization.
- Disclosed herein is a method of treating cancer in a subject in need thereof, the method comprising administering to the subject a TRPM8 antagonist.
- the cancer is bladder cancer, breast cancer, cholangiocarcinoma, colorectal cancer, kidney cancer, liver cancer, lung cancer, lymphoma, myeloma, osteosarcoma, ovarian cancer, pancreatic cancer, prostate cancer, or skin cancer.
- the cancer is prostate cancer.
- the breast cancer is ER positive breast cancer, ER positive metastatic breast cancer, or triple negative breast cancer (TNB).
- TNB triple negative breast cancer
- the myeloma is multiple myeloma (MM).
- the skin cancer is melanoma.
- the lung cancer is small cell lung cancer (SCLC) or non-small cell lung cancer (NSCLC).
- SCLC small cell lung cancer
- NSCLC non-small cell lung cancer
- the cancer is multiple myeloma.
- the cancer is breast cancer.
- the cancer is triple negative breast cancer
- the cancer is melanoma.
- the cancer is osteosarcoma.
- the cancer is small cell lung cancer (SCLC) .
- the cancer is kidney cancer. [0095] In some embodiments of a method of treating cancer, the cancer is bladder cancer. [0096] In some embodiments of a method of treating cancer, the cancer is ovarian cancer. [0097] In some embodiments of a method of treating cancer, the cancer is prostate cancer. [0098] In some embodiments of a method of treating cancer, the cancer is colorectal cancer. [0099] In some embodiments of a method of treating cancer, the cancer is lymphoma.
- the compounds and pharmaceutical compositions of the present disclosure are useful in the treatment or prevention of progression of cancer.
- the cancer is a hematologic malignancy or solid tumor.
- Hematologic malignancies include leukemias, lymphomas, multiple myeloma, and subtypes thereof.
- Lymphomas can be classified various ways, often based on the underlying type of malignant cell, including Hodgkin’s lymphoma (often cancers of Reed-Sternberg cells, but also sometimes originating in B cells; all other lymphomas are non-Hodgkin’s lymphomas), B- cell lymphomas, T-cell lymphomas, mantle cell lymphomas, Burkitt’s lymphoma, follicular lymphoma, and others as defined herein and known in the art.
- Hodgkin’s lymphoma often cancers of Reed-Sternberg cells, but also sometimes originating in B cells; all other lymphomas are non-Hodgkin’s lymphomas
- B- cell lymphomas B- cell lymphomas
- T-cell lymphomas T-cell lymphomas
- mantle cell lymphomas mantle cell lymphomas
- Burkitt’s lymphoma mantle cell lymphomas
- follicular lymphoma follicular lymph
- B-cell lymphomas include, but are not limited to, diffuse large B-cell lymphoma (DLBCL), chronic lymphocytic leukemia (CLL) /small lymphocytic lymphoma (SLL), and others as defined herein and known in the art.
- DLBCL diffuse large B-cell lymphoma
- CLL chronic lymphocytic leukemia
- SLL small lymphocytic lymphoma
- T-cell lymphomas include T-cell acute lymphoblastic leukemia/lymphoma (T-ALL), peripheral T-cell lymphoma (PTCL), T-cell chronic lymphocytic leukemia (T-CLL) Sezary syndrome, and others as defined herein and known in the art.
- T-ALL T-cell acute lymphoblastic leukemia/lymphoma
- PTCL peripheral T-cell lymphoma
- T-CLL T-cell chronic lymphocytic leukemia Sezary syndrome
- Leukemias include acute myeloid (or myelogenous) leukemia (AML), chronic myeloid (or myelogenous) leukemia (CML), acute lymphocytic (or lymphoblastic) leukemia (ALL), chronic lymphocytic leukemia (CLL) hairy cell leukemia (sometimes classified as a lymphoma) and others as defined herein and known in the art.
- AML acute myeloid (or myelogenous) leukemia
- CML chronic myeloid (or myelogenous) leukemia
- ALL acute lymphocytic leukemia
- CLL chronic lymphocytic leukemia
- Plasma cell malignancies include lymphoplasmacytic lymphoma, plasmacytoma, and multiple myeloma.
- Solid tumors include melanomas, neuroblastomas, gliomas or 5 carcinomas such as tumors of the brain, head and neck, breast, lung (e.g., non-small cell lung cancer, NSCLC), reproductive tract (e.g., ovary), upper digestive tract, pancreas, liver, renal system (e.g., kidneys), bladder, prostate, and colorectum.
- lung e.g., non-small cell lung cancer, NSCLC
- reproductive tract e.g., ovary
- upper digestive tract e.g., pancreas, liver, renal system (e.g., kidneys), bladder, prostate, and colorectum.
- Disclosed herein is a method of treating cancer in a subject in need thereof, the method comprising administering to the subject a TRPM8 antagonist in combination with an additional therapeutic agent.
- the additional therapeutic agent is a targeted agent selected from the group consisting of apalutamide, dabrafenib, enzalutamide, fulvestrant, ibrutinib, lenalidomide, palbociclib, pomalidomide, sorafenib, tamoxifen, and thalidomide.
- the additional therapeutic agent is dabrafenib.
- the additional therapeutic agent is enzalutamide.
- the additional therapeutic agent is ibrutinib.
- the additional therapeutic agent is lenalidomide.
- the additional therapeutic agent is sorafenib.
- the additional therapeutic agent is tamoxifen.
- the additional therapeutic agent is a chemotherapy agent selected from the group consisting of 5 -fluorouracil, bleomycin, capecitabine, carboplatin, cisplatin, cyclophosphamide, dacarbazine, docetaxel, doxorubicin, epirubicin, etoposide, etoposide, folinic acid, gemcitabine, ifosfamide, methotrexate, mustine, oxaliplatin, prednisolone, procarbazine, vinorelbine, and vinorelbine.
- a chemotherapy agent selected from the group consisting of 5 -fluorouracil, bleomycin, capecitabine, carboplatin, cisplatin, cyclophosphamide, dacarbazine, docetaxel, doxorubicin, epirubicin, etoposide, etoposide, folinic acid, gemcitabine, ifosfamide, met
- the additional therapeutic agent is cisplatin.
- the additional therapeutic agent is gemcitabine.
- the additional therapeutic agent is oxaliplatin.
- the additional therapeutic agent is a CTLA-4 blocker, a PD-1 blocker, or a PD-L1 blocker.
- the CTLA-4 blocker is ipilimumab or tremelimumab.
- the PD-1 blocker is nivolumab or pembrolizumab.
- the PD-L1 blocker is atezolizumab, avelumab, cemiplimab, or durvalumab.
- combination refers to either a fixed combination in one dosage unit form, or non-fixed combination, or a kit of parts for the combined administration where two or more therapeutic agents may be administered together, independently at the same time or separately within time intervals, especially where these time intervals allow that the combination partners show a cooperative, e.g., synergistic, effect.
- the term “combination therapy” refers to the administration of two or more therapeutic agents to treat a therapeutic condition or disorder described in the present disclosure. Such administration encompasses co-administration of these therapeutic agents in a substantially simultaneous manner, such as in a single formulation having a fixed ratio of active ingredients or in separate formulations (e.g. , capsules and/or intravenous formulations) for each active ingredient. In addition, such administration also encompasses use of each type of therapeutic agent in a sequential or separate manner, either at approximately the same time or at different times. Regardless of whether the active ingredients are administered as a single formulation or in separate formulations, the drugs are administered to the same patient as part of the same course of therapy. In any case, the treatment regimen will provide beneficial effects in treating the conditions or disorders described herein.
- the combination is for simultaneous, sequential, or separate administration.
- simultaneous therapeutic use within the meaning of the present embodiments is meant an administration of at least two active ingredients by the same route and at the same time or at substantially the same time.
- sequential therapeutic use is meant administration of at least two active ingredients at different times, the administration route being identical or different. More particularly by an administration method is meant according to which the whole administration of one of the active ingredients is carried out before administration of the other or others commences.
- the combination is a fixed combination. In some embodiments, the combination is a non-fixed combination.
- the terms “fixed combination,” “fixed dose” and “single formulation” as used herein refers to a single carrier or vehicle or dosage form formulated to deliver an amount, which is jointly therapeutically effective for the treatment of cancer, of both therapeutic agents to a patient.
- the single vehicle is designed to deliver an amount of each of the agents along with any pharmaceutically acceptable carriers or excipients.
- the vehicle is a tablet, capsule, pill, or a patch. In other embodiments, the vehicle is a solution or a suspension.
- non-fixed combination or “kit of parts” means that the therapeutic agents of the combinations disclosed herein are both administered to a patient as separate entities either simultaneously, concurrently, or sequentially with no specific time limits, wherein such administration provides therapeutically effective levels of the two compounds in the body of a subject in need thereof.
- cocktail therapy e.g., the administration of three or more active ingredients.
- Frozen vials of cell stocks were removed from liquid nitrogen and thawed rapidly by placing at 37°C in a water bath with gentle agitation for 1-2 minutes and then decontaminated by wiping with 70% ethanol before opening in a Class II biological safety cabinet.
- the vial contents were transferred dropwise into 10 mL of cell culture medium in a sterile 15mL conical tube (BD-Falcon, 352097).
- the cells were centrifuged at 200 x g for 5 minutes (Eppendorf, 5810R). The supernatant was aspirated, and the cell pellets were re-suspended in 1 mL of fresh cell culture medium and then transferred into a T75 flask containing cell culture medium.
- the cells were maintained with the addition of fresh medium or replacement of medium. The medium was renewed every 2-3 days. The cells were harvested from flask and then counted. The cells were diluted with culture medium to designed densities and inoculated 135 pL of cell suspension into each well of 96-well cell culture plate (Coming, 3599).
- Compound 1 besylate (10 pM and 30 pM), dabrafenib (1 pM), cisplatin (5 pM), gemcitabine (0.002 pM), lenalidomide (5 pM), enzalutamide (1 pM), sorafenib (6 pM), oxaliplatin (2 pM), tamoxifen (5 pM), carboplatin (2 pM), ibrutinib (5 pM), were prepared.
- the compound intermediate plate (10X) were prepared. Compound was diluted using culture medium. 15 pl of the compound-medium was added in each well from the 10X compound intermediate plate into the cells in 96-well assay plate according to the plate map. The final DMSO concentration was 0.1%. Compound 1 was tested in triplicate. The plate was incubated at 37°C for 5d, without changing medium during treatment.
- KMS-26 cell line was used in the proliferation assay as described in Example 1, using Compound 1 (10 pM and 30 pM), and lenalidomide (5 pM) (FIG. 1).
- Example 3 Cell proliferation assay with JJN-3 cell line (multiple myeloma)
- JJN-3 cell line was used in the proliferation assay as described in Example 1, using Compound 1 (10 pM and 30 pM), and lenalidomide (5 pM) (FIG. 2).
- Example 4 Cell proliferation assay with OPM-2 cell line (multiple myeloma)
- OPM-2 cell line was used in the proliferation assay as described in Example 1, using Compound 1 (10 pM and 30 pM), and lenalidomide (5 pM) (FIG. 3).
- Example 5 Cell proliferation assay with MCF7 cell line (breast cancer)
- MCF7 cell line was used in the proliferation assay as described in Example 1, using Compound 1 (10 pM and 30 pM), and tamoxifen (5 pM) (FIG. 4).
- Example 6 Cell proliferation assay with MDA-MB-231 cell line (TNB breast cancer)
- Example 7 Cell proliferation assay with HMC-1-8 cell line (breast cancer)
- HMC-1-8 cell line was used in the proliferation assay as described in Example 1, using Compound 1 (10 pM and 30 pM), and sorafenib (6 pM) (FIG. 6).
- SK-MEL-28 cell line was used in the proliferation assay as described in Example 1, using Compound 1 (10 pM and 30 pM), and dabrafenib (1 pM) (FIG. 7).
- Example 9 Cell proliferation assay with A101D cell line (melanoma)
- A101D cell line was used in the proliferation assay as described in Example 1, using Compound
- Example 10 Cell proliferation assay with Hs 294T cell line (melanoma)
- Hs 294T cell line was used in the proliferation assay as described in Example 1, using Compound 1 (10 pM and 30 pM), and dabrafenib (1 pM) (FIG. 9).
- Example 11 Cell proliferation assay with MDA-MB-435S cell line (melanoma)
- MDA-MB-435S cell line was used in the proliferation assay as described in Example 1, using Compound 1 (10 pM and 30 pM), and dabrafenib (1 pM) (FIG. 10).
- Example 12 Cell proliferation assay with HOS cell line (osteosarcoma)
- HOS cell line was used in the proliferation assay as described in Example 1, using Compound 1 (10 pM and 30 pM), and cisplatin (5 pM) (FIG. 11).
- Example 13 Cell proliferation assay with Saos-2 cell line (osteosarcoma)
- Saos-2 cell line was used in the proliferation assay as described in Example 1, using Compound 1 (10 pM and 30 pM), and cisplatin (5 pM) (FIG. 12).
- Example 14 Cell proliferation assay with NCI-H2081 cell line (SCLC lung cancer) [00148] NCI-H208 cell line was used in the proliferation assay as described in Example 1, using Compound 1 (10 pM and 30 pM), and cisplatin (5 pM) (FIG. 13).
- Example 15 Cell proliferation assay with ACHN cell line (kidney cancer)
- ACHN cell line was used in the proliferation assay as described in Example 1, using Compound 1 (10 pM and 30 pM), and sorafenib (6 pM) (FIG. 14).
- Example 16 Cell proliferation assay with G-402 cell line (kidney cancer)
- G-402 cell line was used in the proliferation assay as described in Example 1, using Compound
- Example 17 Cell proliferation assay with T24 cell line (bladder cancer)
- T24 cell line was used in the proliferation assay as described in Example 1, using Compound 1
- Example 18 Cell proliferation assay with OAW28 cell line (ovarian cancer)
- OAW28 cell line was used in the proliferation assay as described in Example 1, using
- Example 19 Cell proliferation assay with LNCaP cell line (prostate cancer)
- LNCaP cell line was used in the proliferation assay as described in Example 1, using Compound 1 (10 pM and 30 pM), and enzalutamide (1 pM) (FIG. 18).
- Example 20 Cell proliferation assay with Caco-2 cell line (colorectal cancer)
- Caco-2 cell line was used in the proliferation assay as described in Example 1, using Compound
- Example 21 Cell proliferation assay with SU-DHL-1 cell line (lymphoma)
- SU-DHL-1 cell line was used in the proliferation assay as described in Example 1, using Compound 1 (10 pM and 30 pM), and ibrutinib (5 pM) (FIG. 20).
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Veterinary Medicine (AREA)
- Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Epidemiology (AREA)
- Inorganic Chemistry (AREA)
- Molecular Biology (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Organic Chemistry (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
A method of treating cancerin a subject, the method comprising administering a TRPM8 antagonist that is 8,8-difluoro-3-(2-(4-(4-methylpyridazin-3-y 1)pheny 1)-2 -oxoethyl)-1,3,diazaspiro[4.5]decane-2, 4-dione.
Description
TRPM8 ANTAGONIST FOR TREATING CANCER
CROSS-REFERENCE
[0001] This application claims the benefit of U.S. Provisional Application Serial No. 63/583,688 filed September 19, 2023; which is hereby incorporated by reference in its entirety.
BACKGROUND
[0002] Cancer is characterized by the development of abnormal cells that divide uncontrollably and have the ability to infiltrate and destroy normal body tissue. Cancer is a major public health problem in many parts of the world, which is the second-leading cause of death around the global. The survival rates are, however, improving for many types of cancer, due to the current advancement in cancer screening, treatment, and prevention.
[0003] Transient receptor potential (TRP) channels are one of the largest groups of ion channels, and they are divided into 6 sub-families (TRPV, TRPM, TRPA, TRPC, TRPP and TRPMU). TRP channels are cation-selective channels that are activated by a variety of physical (e.g., temperature, osmolarity, mechanical) and chemical stimuli. Transient receptor potential melastatin-subfamily member 8 (TRPM8) is a member of TRP channel family. TRPM8 can sense temperature changes in the range of both innocuous cold (15-28 °C) (McKeny et al Nature 2002, 416, 52; Peier et al Cell 2002, 108, 705) and noxious cold (<15°C) as well as by chemical agents such as menthol and icilin (Chuang et al Neuron 2004, 43, 859).
SUMMARY
[0004] It was discovered that TRPM8 was expressed in various cancers, especially metastatic and resistant cancers. In recent years, an increasing number of literatures have shown that TRPM8 involves in the initiation and progression of tumors and study found that TRPM8 expression upregulated markedly in prostate cancer (PC) and in many other tumors, suggesting an important role in carcinogenesis in human cancers. Accumulating evidence has emerged over the past decade that strongly supports the hypothesis that TRPM8 which plays prominent role in thermoregulation, could be one of the most promising novel therapeutic targets in cancer treatment.
[0005] Disclosed herein is a method of treating cancer in a subject in need thereof, the method comprising administering to the subject 8,8-difluoro-3-(2-(4-(4-methylpyridazin-3-yl)phenyl)-2- oxoethyl)- 1 , 3 -diazaspiro [4.5] decane -2,4-dione
(Compound 1), or a pharmaceutically acceptable salt thereof.
[0006] In some embodiments, Compound 1 is in the form of a salt.
[0007] In some embodiments, Compound 1 is in the form of a besylate salt.
[0008] In some embodiments, the cancer is bladder cancer, breast cancer, cholangiocarcinoma, colorectal cancer, kidney cancer, liver cancer, lung cancer, lymphoma, myeloma, osteosarcoma, ovarian cancer, pancreatic cancer, prostate cancer, or skin cancer.
[0009] In some embodiments, the breast cancer is ER positive breast cancer, ER positive metastatic breast cancer, or triple negative breast cancer (TNB).
[0010] In some embodiments, the myeloma is multiple myeloma (MM).
[0011] In some embodiments, the skin cancer is melanoma.
[0012] In some embodiments, the lung cancer is small cell lung cancer (SCLC) or non-small cell lung cancer (NSCLC).
[0013] In some embodiments, the method further comprises administering an additional therapeutic agent.
[0014] In some embodiments, the additional therapeutic agent is a targeted agent, a chemotherapy, an immunotherapy (IO), radiation therapy, a gene therapy, or a cell therapy, or any combination thereof. [0015] In some embodiments, the additional therapeutic agent is a targeted agent selected from the group consisting of apalutamide, dabrafenib, enzalutamide, fulvestrant, ibrutinib, lenalidomide, palbociclib, pomalidomide, sorafenib, tamoxifen, and thalidomide.
[0016] In some embodiments, the additional therapeutic agent is a chemotherapy agent selected from the group consisting of 5 -fluorouracil, bleomycin, capecitabine, carboplatin, cisplatin, cyclophosphamide, dacarbazine, docetaxel, doxorubicin, epirubicin, etoposide, etoposide, folinic acid, gemcitabine, ifosfamide, methotrexate, mustine, oxaliplatin, prednisolone, procarbazine, vinorelbine, and vinorelbine. [0017] In some embodiments, the additional therapeutic agent is a CTLA-4 blocker, a PD-1 blocker, or a PD-L1 blocker.
[0018] In some embodiments, the CTLA-4 blocker is ipilimumab or tremelimumab.
[0019] In some embodiments, the PD-1 blocker is nivolumab or pembrolizumab.
[0020] In some embodiments, the PD-L1 blocker is atezolizumab, avelumab, cemiplimab, or durvalumab.
INCORPORATION BY REFERENCE
[0021] All publications, patents, and patent applications mentioned in this specification are herein incorporated by reference to the same extent as if each individual publication, patent, or patent application was specifically and individually indicated to be incorporated by reference.
BRIEF DESCRIPTION OF THE DRAWINGS
[0022] The novel features of the present disclosure are set forth with particularity in the appended claims. A better understanding of the features and advantages of the present disclosure will be obtained by reference to the following detailed description that sets forth illustrative embodiments, in which the principles of the present disclosure are utilized, and the accompanying drawings of which:
[0023] FIG. 1 shows the cellular proliferation inhibition in KMS-26 cell line of Compound 1, lenalidomide, and Compound 1 combined with lenalidomide.
[0024] FIG. 2 shows the cellular proliferation inhibition in JJN-3 cell line of Compound 1, lenalidomide, and Compound 1 combined with lenalidomide.
[0025] FIG. 3 shows the cellular proliferation inhibition in OPM-2 cell line of Compound 1, lenalidomide, and Compound 1 combined with lenalidomide.
[0026] FIG. 4 shows the cellular proliferation inhibition in MCF7 cell line of Compound 1, tamoxifen, and Compound 1 combined with tamoxifen.
[0027] FIG. 5 shows the cellular proliferation inhibition in MDA-MB-231 cell line that Compound 1, gemcitabine, and Compound 1 combined with gemcitabine.
[0028] FIG. 6 shows the cellular proliferation inhibition in HMC-1-8 cell line that Compound 1, sorafenib, and Compound 1 combined with sorafenib.
[0029] FIG. 7 shows the cellular proliferation inhibition in SK-MEL-28 cell line that Compound 1, dabrafenib, and Compound 1 combined with dabrafenib.
[0030] FIG. 8 shows the cellular proliferation inhibition in A101D cell line that Compound 1, dabrafenib, and Compound 1 combined with dabrafenib.
[0031] FIG. 9 shows the cellular proliferation inhibition in Hs 294T cell line that Compound 1, dabrafenib, and Compound 1 combined with dabrafenib.
[0032] FIG. 10 shows the cellular proliferation inhibition in MDA-MB-435S cell line that Compound 1, dabrafenib, and Compound 1 combined with dabrafenib.
[0033] FIG. 11 shows the cellular proliferation inhibition in HOS cell line that Compound 1, cisplatin, and Compound 1 combined with cisplatin.
[0034] FIG. 12 shows the cellular proliferation inhibition in Saos-2 cell line that Compound 1, cisplatin, and Compound 1 combined with cisplatin.
[0035] FIG. 13 shows the cellular proliferation inhibition in NCI-H2081 cell line that Compound 1, cisplatin, and Compound 1 combined with cisplatin.
[0036] FIG. 14 shows the cellular proliferation inhibition in ACHN cell line that Compound 1, sorafenib, and Compound 1 combined with sorafenib.
[0037] FIG. 15 shows the cellular proliferation inhibition in G-402 cell line that Compound 1, sorafenib, and Compound 1 combined with sorafenib.
[0038] FIG. 16 shows the cellular proliferation inhibition in T24 cell line that Compound 1, gemcitabine, and Compound 1 combined with gemcitabine.
[0039] FIG.17 shows the cellular proliferation inhibition in OAW28 cell line that Compound 1, carboplatin, and Compound 1 combined with carboplatin.
[0040] FIG. 18 shows the cellular proliferation inhibition in LNCaP cell line that Compound 1, enzalutamide, and Compound 1 combined with enzalutamide.
[0041] FIG. 19 shows the cellular proliferation inhibition in Caco-2 cell line that Compound 1, oxaliplatin, and Compound 1 combined with oxaliplatin.
[0042] FIG. 20 shows the cellular proliferation inhibition in SU-DHL-1 cell line that Compound 1, ibrutinib, and Compound 1 combined with ibrutinib.
DETAILED DESCRIPTION
[0043] As used herein and in the appended claims, the singular forms “a,” “an,” and “the” include plural referents unless the context clearly dictates otherwise. Thus, for example, reference to “an agent” includes a plurality of such agents, and reference to “the cell” includes reference to one or more cells (or to a plurality of cells) and equivalents thereof known to those skilled in the art, and so forth. When ranges are used herein for physical properties, such as molecular weight, or chemical properties, such as chemical formulae, all combinations and subcombinations of ranges and specific embodiments therein are intended to be included. The term “about” when referring to a number or a numerical range means that the number or numerical range referred to is an approximation within experimental variability (or within statistical experimental error), and thus the number or numerical range, in some instances, will vary between 1% and 15% of the stated number or numerical range. The term “comprising” (and related terms such as “comprise” or “comprises” or “having” or “including”) is not intended to exclude that in other certain embodiments, for example, an embodiment of any composition of matter, composition, method, or process, or the like, described herein, “consist of’ or “consist essentially of’ the described features.
[0044] As used in the specification and appended claims, unless specified to the contrary, the following terms have the meaning indicated below.
[0045] As used herein, the term “therapeutic” means an agent utilized to treat, combat, ameliorate, prevent, or improve an unwanted condition or disease of a patient. In some embodiments, a therapeutic agent such as a TRPM8 antagonist to the treatment and/or the amelioration of cancers.
[0046] “Administering,” when used in conjunction with a therapeutic means to administer a therapeutic systemically or locally, as directly into or onto a target tissue, or to administer a therapeutic to a patient whereby the therapeutic positively impacts the tissue to which it is targeted. Thus, as used herein, the term “administering,” when used in conjunction with a composition described herein, can include, but is not limited to, providing a composition into or onto the target tissue; providing a composition systemically to a patient by, e.g., oral administration whereby the therapeutic reaches the target tissue or cells. “Administering” a composition may be accomplished by injection, topical administration, and oral administration or by other methods alone or in combination with other known techniques.
[0047] The term “animal” as used herein includes, but is not limited to, humans and non-human vertebrates such as wild, domestic and farm animals. As used herein, the terms “patient,” “subject” and “individual” are intended to include living organisms in which certain conditions as described herein can occur. Examples include humans, monkeys, cows, sheep, goats, dogs, cats, mice, rats, and transgenic species thereof. In a preferred embodiment, the patient is a primate. In certain embodiments, the primate or subject is a human. In certain instances, the human is an adult. In certain instances, the human is child. In further instances, the human is under the age of 12 years. In certain instances, the human is elderly. In
other instances, the human is 60 years of age or older. Other examples of subjects include experimental animals such as mice, rats, dogs, cats, goats, sheep, pigs, and cows. The experimental animal can be an animal model for a disorder, e.g., a transgenic mouse with hypertensive pathology.
[0048] By “pharmaceutically acceptable,” it is meant the carrier, diluent or excipient must be compatible with the other ingredients of the formulation and not deleterious to the recipient thereof.
[0049] The term “pharmaceutical composition” shall mean a composition comprising at least one active ingredient, whereby the composition is amenable to investigation for a specified, efficacious outcome in a mammal (for example, without limitation, a human). Those of ordinary skill in the art will understand and appreciate the techniques appropriate for determining whether an active ingredient has a desired efficacious outcome based upon the needs of the artisan.
[0050] A “therapeutically effective amount” or “effective amount” as used herein refers to the amount of active compound or pharmaceutical agent that elicits a biological or medicinal response in a tissue, system, animal, individual or human that is being sought by a researcher, veterinarian, medical doctor or other clinician, which includes one or more of the following: (1) preventing the disease; for example, preventing a disease, condition or disorder in an individual that may be predisposed to the disease, condition or disorder but does not yet experience or display the pathology or symptomatology of the disease, (2) inhibiting the disease; for example, inhibiting a disease, condition or disorder in an individual that is experiencing or displaying the pathology or symptomatology of the disease, condition or disorder (i.e., arresting further development of the pathology and/or symptomatology), and (3) ameliorating the disease; for example, ameliorating a disease, condition or disorder in an individual that is experiencing or displaying the pathology or symptomatology of the disease, condition or disorder (i.e., reversing the pathology and/or symptomatology).
[0051] The terms “treat,” “treated,” “treatment,” or “treating” as used herein refers to therapeutic treatment, wherein the object is to slow (lessen) an undesired physiological condition, disorder, or disease, or to obtain beneficial or desired clinical results. For the purposes described herein, beneficial or desired clinical results include, but are not limited to, alleviation of symptoms; diminishment of the extent of the condition, disorder or disease; stabilization (i.e., not worsening) of the state of the condition, disorder or disease; delay in onset or slowing of the progression of the condition, disorder or disease; amelioration of the condition, disorder or disease state; and remission (whether partial or total), whether detectable or undetectable, or enhancement or improvement of the condition, disorder or disease. Treatment includes eliciting a clinically significant response without excessive levels of side effects. Treatment also includes prolonging survival as compared to expected survival if not receiving treatment. [0052] The terms below, as used herein, have the following meanings, unless indicated otherwise: [0053] ‘ ‘oxo” refers to =0.
[0054] ‘ ‘Amine” refers to -NFF;
[0055] “hydroxy” refers to -OH;
[0056] “Carboxyl” refers to -COOH.
[0057] “Alkyl” refers to a straight-chain or branched-chain saturated hydrocarbon monoradical having from one to about ten carbon atoms, more preferably one to six carbon atoms. Examples include, but are not limited to methyl, ethyl, n-propyl, isopropyl, 2 -methyl- 1 -propyl, 2 -methyl -2 -propyl, 2-methyl-l- butyl, 3 -methyl- 1 -butyl, 2-methyl-3-butyl, 2,2-dimethyl-l -propyl, 2-methyl-l -pentyl, 3 -methyl- 1 -pentyl, 4-methyl-l -pentyl, 2-methyl-2-pentyl, 3 -methyl -2 -pentyl, 4-methyl-2-pentyl, 2,2-dimethyl-l -butyl, 3,3- dimethyl-1 -butyl, 2 -ethyl- 1 -butyl, n-butyl, isobutyl, sec-butyl, t-butyl, n-pentyl, isopentyl, neopentyl, tert-amyl and hexyl, and longer alkyl groups, such as heptyl, octyl and the like. Whenever it appears herein, a numerical range such as “Ci-Ce alkyl”, means that the alkyl group may consist of 1 carbon atom, 2 carbon atoms, 3 carbon atoms, 4 carbon atoms, 5 carbon atoms or 6 carbon atoms, although the present definition also covers the occurrence of the term “alkyl” where no numerical range is designated. In some embodiments, the alkyl is a Ci-Cio alkyl. In some embodiments, the alkyl is a Ci-Ce alkyl. In some embodiments, the alkyl is a C1-C5 alkyl. In some embodiments, the alkyl is a C1-C4 alkyl. In some embodiments, the alkyl is a C1-C3 alkyl. Unless stated otherwise specifically in the specification, an alkyl group may be optionally substituted, for example, with one or more oxo, halogen, amino, nitrile, nitro, hydroxyl, haloalkyl, alkoxy, carboxyl, carboxylate, aryl, cycloalkyl, heterocycloalkyl, or heteroaryl, and the like. In some embodiments, the alkyl is optionally substituted with one or more oxo, halogen, -CN, - COOH, -COOMe, -OH, -OMe, -NH2, or -NO2. In some embodiments, the alkyl is optionally substituted with one or more halogen, -CN, -OH, or -OMe. In some embodiments, the alkyl is optionally substituted with halogen.
[0058] “Alkenyl” refers to a straight-chain or branched-chain hydrocarbon monoradical having one or more carbon-carbon double-bonds and having from two to about ten carbon atoms, more preferably two to about six carbon atoms. The group may be in either the cis or trans or Z or E conformation about the double bond(s), and should be understood to include both isomers. Examples include, but are not limited to ethenyl (-CH=CH2), 1-propenyl (-CH2CH=CH2), isopropenyl [-C(CH3)=CH2], butenyl, 1,3-butadienyl and the like. Whenever it appears herein, a numerical range such as “C2-C6 alkenyl”, means that the alkenyl group may consist of 2 carbon atoms, 3 carbon atoms, 4 carbon atoms, 5 carbon atoms or 6 carbon atoms, although the present definition also covers the occurrence of the term “alkenyl” where no numerical range is designated. Unless stated otherwise specifically in the specification, an alkenyl group may be optionally substituted, for example, with one or more oxo, halogen, amino, nitrile, nitro, hydroxyl, alkyl, haloalkyl, alkoxy, carboxyl, carboxylate, aryl, cycloalkyl, heterocycloalkyl, or heteroaryl, and the like. In some embodiments, the alkenyl is optionally substituted with one or more oxo, halogen, -CN, -COOH, -COOMe, -OH, -OMe, -NH2, or -NO2. In some embodiments, the alkenyl is optionally substituted with one or more halogen, -CN, -OH, or -OMe. In some embodiments, the alkenyl is optionally substituted with halogen.
[0059] “Alkynyl” refers to a straight-chain or branched-chain hydrocarbon monoradical having one or more carbon-carbon triple-bonds and having from two to about ten carbon atoms, more preferably from two to about six carbon atoms. Examples include, but are not limited to ethynyl, 2-propynyl, 2-butynyl, 1,3-butadiynyl and the like. Whenever it appears herein, a numerical range such as “C2-C6 alkynyl”,
means that the alkynyl group may consist of 2 carbon atoms, 3 carbon atoms, 4 carbon atoms, 5 carbon atoms or 6 carbon atoms, although the present definition also covers the occurrence of the term “alkynyl” where no numerical range is designated. Unless stated otherwise specifically in the specification, an alkynyl group may be optionally substituted, for example, with one or more oxo, halogen, amino, nitrile, nitro, hydroxyl, alkyl, haloalkyl, alkoxy, carboxyl, carboxylate, aryl, cycloalkyl, heterocycloalkyl, or heteroaryl, and the like. In some embodiments, the alkynyl is optionally substituted with one or more oxo, halogen, -CN, -C00H, -COOMe, -OH, -OMe, -NH2, or -NO2. In some embodiments, the alkynyl is optionally substituted with one or more halogen, -CN, -OH, or -OMe. In some embodiments, the alkynyl is optionally substituted with halogen.
[0060] “Alkylene” refers to a straight or branched divalent hydrocarbon chain. Unless stated otherwise specifically in the specification, an alkylene group may be optionally substituted, for example, with one or more oxo, halogen, amino, nitrile, nitro, hydroxyl, alkyl, haloalkyl, alkoxy, carboxyl, carboxylate, aryl, cycloalkyl, heterocycloalkyl, or heteroaryl, and the like. In some embodiments, the alkylene is optionally substituted with one or more oxo, halogen, -CN, -C00H, -COOMe, -OH, -OMe, -NH2, or - NO2. In some embodiments, the alkylene is optionally substituted with one or more halogen, -CN, -OH, or -OMe. In some embodiments, the alkylene is optionally substituted with halogen.
[0061] “Alkoxy” refers to a radical of the formula -Oalkyl where alkyl is defined as above. Unless stated otherwise specifically in the specification, an alkoxy group may be optionally substituted, for example, with one or more oxo, halogen, amino, nitrile, nitro, hydroxyl, alkyl, haloalkyl, alkoxy, carboxyl, carboxylate, aryl, cycloalkyl, heterocycloalkyl, or heteroaryl, and the like. In some embodiments, the alkoxy is optionally substituted with one or more halogen, -CN, -COOH, -COOMe, - OH, -OMe, -NH2, or -NO2. In some embodiments, the alkoxy is optionally substituted with one or more halogen, -CN, -OH, or -OMe. In some embodiments, the alkoxy is optionally substituted with halogen. [0062] “Aryl” refers to a radical derived from a hydrocarbon ring system comprising 6 to 30 carbon atoms and at least one aromatic ring. The aryl radical may be a monocyclic, bicyclic, tricyclic, or tetracyclic ring system, which may include fused (when fused with a cycloalkyl or heterocycloalkyl ring, the aryl is bonded through an aromatic ring atom) or bridged ring systems. In some embodiments, the aryl is a 6- to 10-membered aryl. In some embodiments, the aryl is a 6-membered aryl (phenyl). Aryl radicals include, but are not limited to anthracenyl, naphthyl, phenanthrenyl, azulenyl, phenyl, chrysenyl, fluoranthenyl, fluorenyl, as-indacenyl, s-indacenyl, indanyl, indenyl, phenalenyl, phenanthrenyl, pleiadenyl, pyrenyl, and triphenylenyl. Unless stated otherwise specifically in the specification, an aryl may be optionally substituted, for example, with one or more halogen, amino, nitrile, nitro, hydroxyl, alkyl, alkenyl, alkynyl, haloalkyl, alkoxy, carboxyl, carboxylate, aryl, cycloalkyl, heterocycloalkyl, or heteroaryl, and the like. In some embodiments, the aryl is optionally substituted with one or more halogen, methyl, ethyl, -CN, -COOH, -COOMe, -CF3, -OH, -OMe, -NH2, or -NO2. In some embodiments, the aryl is optionally substituted with one or more halogen, methyl, ethyl, -CN, -CF3, -OH, or -OMe. In some embodiments, the aryl is optionally substituted with halogen.
[0063] “Cycloalkyl” refers to a partially or fully saturated, monocyclic, or polycyclic carbocyclic ring, which may include fused (when fused with an aryl or a heteroaryl ring, the cycloalkyl is bonded through a non-aromatic ring atom), spiro, and/or bridged ring systems. In some embodiments, the cycloalkyl is fully saturated. Representative cycloalkyls include, but are not limited to, cycloalkyls having from three to fifteen carbon atoms (e.g., C3-C15 fully saturated cycloalkyl or C3-C15 cycloalkenyl), from three to ten carbon atoms (e.g., C3-C10 fully saturated cycloalkyl or C3-C10 cycloalkenyl), from three to eight carbon atoms (e.g., C3-C8 fully saturated cycloalkyl or C3-C8 cycloalkenyl), from three to six carbon atoms (e.g., C3-C6 fully saturated cycloalkyl or C3-C6 cycloalkenyl), from three to five carbon atoms (e.g., C3-C5 fully saturated cycloalkyl or C3-C5 cycloalkenyl), or three to four carbon atoms (e.g., C3-C4 fully saturated cycloalkyl or C3-C4 cycloalkenyl). In some embodiments, the cycloalkyl is a 3 - to 10-membered fully saturated cycloalkyl or a 3- to 10-membered cycloalkenyl. In some embodiments, the cycloalkyl is a 3 - to 6-membered fully saturated cycloalkyl or a 3- to 6-membered cycloalkenyl. In some embodiments, the cycloalkyl is a 5 - to 6-membered fully saturated cycloalkyl or a 5 - to 6-membered cycloalkenyl. Monocyclic cycloalkyls include, for example, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, cycloheptyl, and cyclooctyl. Polycyclic cycloalkyls include, for example, adamantyl, norbomyl, decalinyl, bicyclo[3.3.0]octyl, bicyclo[4.3.0]nonyl, cis-decalinyl, trans-decalinyl, bicyclo [2.1.1] hexyl, bicyclo[2.2.1]heptyl, bicyclo[2.2.2]octyl, bicyclo[3.2.1]octyl, bicyclo[3.2.2]nonyl, and bicyclo[3.3.2]decyl, bicyclof l. l. l]pentyl, bicyclo[3.1.0]hexyl, bicyclo[3.1.1]heptyl, 7,7-dimethyl- bicyclo[2.2.1]heptanyl, Spiro[4.2]heptyl, spiro[4.3]octyl, spiro[5.2]octyl, spiro [3.3] heptyl, and spiro [5.3] nonyl. Partially saturated cycloalkyls include, for example cyclopentenyl, cyclohexenyl, cycloheptenyl, and cyclooctenyl. Unless stated otherwise specifically in the specification, a cycloalkyl is optionally substituted, for example, with one or more oxo, halogen, amino, nitrile, nitro, hydroxyl, alkyl, alkenyl, alkynyl, haloalkyl, alkoxy, carboxyl, carboxylate, aryl, cycloalkyl, heterocycloalkyl, or heteroaryl, and the like. In some embodiments, a cycloalkyl is optionally substituted with one or more oxo, halogen, methyl, ethyl, -CN, -COOH, -COOMe, -CF3, -OH, -OMe, -NH2, or -NO2. In some embodiments, a cycloalkyl is optionally substituted with one or more oxo, halogen, methyl, ethyl, -CN, - CF3, -OH, or -OMe. In some embodiments, the cycloalkyl is optionally substituted with halogen.
[0064] ‘ ‘Halo” or “halogen” refers to bromo, chloro, fluoro or iodo. In some embodiments, halogen is fluoro or chloro. In some embodiments, halogen is fluoro.
[0065] “Haloalkyl” refers to an alkyl radical, as defined above, that is substituted by one or more halo radicals, as defined above, e.g., trifluoromethyl, difluoromethyl, fluoromethyl, trichloromethyl, 2,2,2- trifluoroethyl, 1,2-difluoroethyl, 2-fluoroethyl, 3 -bromo-2 -fluoropropyl, 1,2-dibromoethyl, and the like. [0066] “Heterocycloalkyl” refers to a 3- to 24-membered partially or fully saturated ring radical comprising 2 to 23 carbon atoms and from one to 8 heteroatoms selected from the group consisting of nitrogen, oxygen, phosphorous, silicon, and sulfur. In some embodiments, the heterocycloalkyl is fully saturated. In some embodiments, the heterocycloalkyl is C-linked. In some embodiments, the heterocycloalkyl is N-linked. In some embodiments, the heterocycloalkyl comprises one to three heteroatoms selected from the group consisting of nitrogen, oxygen, and sulfur. In some embodiments,
the heterocycloalkyl comprises one to three heteroatoms selected from the group consisting of nitrogen and oxygen. In some embodiments, the heterocycloalkyl comprises one to three nitrogens. In some embodiments, the heterocycloalkyl comprises one or two nitrogens. In some embodiments, the heterocycloalkyl comprises one nitrogen. In some embodiments, the heterocycloalkyl comprises one nitrogen and one oxygen. Unless stated otherwise specifically in the specification, the heterocycloalkyl radical may be a monocyclic, bicyclic, tricyclic, or tetracyclic ring system, which may include fused (when fused with an aryl or a heteroaryl ring, the heterocycloalkyl is bonded through a non-aromatic ring atom), spiro, or bridged ring systems; and the nitrogen, carbon, or sulfur atoms in the heterocycloalkyl radical may be optionally oxidized; the nitrogen atom may be optionally quatemized. Representative heterocycloalkyls include, but are not limited to, heterocycloalkyls having from two to fifteen carbon atoms (e.g., C2-C15 fully saturated heterocycloalkyl or C2-C15 heterocycloalkenyl), from two to ten carbon atoms (e.g., C2-C10 fully saturated heterocycloalkyl or C2-C10 heterocycloalkenyl), from two to eight carbon atoms (e.g., C2-C8 fully saturated heterocycloalkyl or C2-C8 heterocycloalkenyl), from two to seven carbon atoms (e.g., C2-C7 fully saturated heterocycloalkyl or C2-C7 heterocycloalkenyl), from two to six carbon atoms (e.g., C2-C6 fully saturated heterocycloalkyl or C2-C7 heterocycloalkenyl), from two to five carbon atoms (e.g., C2-C5 fully saturated heterocycloalkyl or C2-C5 heterocycloalkenyl), or two to four carbon atoms (e.g., C2-C4 fully saturated heterocycloalkyl or C2-C4 heterocycloalkenyl). Examples of such heterocycloalkyl radicals include, but are not limited to, aziridinyl, azetidinyl, oxetanyl, dioxolanyl, thienyl[l,3]dithianyl, decahydroisoquinolyl, imidazolinyl, imidazolidinyl, isothiazolidinyl, isoxazolidinyl, morpholinyl, octahydroindolyl, octahydroisoindolyl, 2-oxopiperazinyl, 2-oxopiperidinyl, 2-oxopyrrolidinyl, oxazolidinyl, piperidinyl, piperazinyl, 4-piperidonyl, pyrrolidinyl, pyrazolidinyl, quinuclidinyl, thiazolidinyl, tetrahydrofuryl, trithianyl, tetrahydropyranyl, thiomorpholinyl, thiamorpholinyl, 1 -oxo-thiomorpholinyl, 1,1-dioxo-thiomorpholinyl, 1,3-dihydroisobenzofuran-l-yl, 3- oxo-1, 3-dihydroisobenzofuran-l-yl, methyl-2-oxo-l,3-dioxol-4-yl, and 2-oxo-l,3-dioxol-4-yl. The term heterocycloalkyl also includes all ring forms of the carbohydrates, including but not limited to the monosaccharides, the disaccharides, and the oligosaccharides. In some embodiments, heterocycloalkyls have from 2 to 10 carbons in the ring. It is understood that when referring to the number of carbon atoms in a heterocycloalkyl, the number of carbon atoms in the heterocycloalkyl is not the same as the total number of atoms (including the heteroatoms) that make up the heterocycloalkyl (i.e. skeletal atoms of the heterocycloalkyl ring). In some embodiments, the heterocycloalkyl is a 3- to 8-membered heterocycloalkyl. In some embodiments, the heterocycloalkyl is a 3 - to 7-membered heterocycloalkyl. In some embodiments, the heterocycloalkyl is a 3 - to 6-membered heterocycloalkyl. In some embodiments, the heterocycloalkyl is a 4- to 6-membered heterocycloalkyl. In some embodiments, the heterocycloalkyl is a 5- to 6-membered heterocycloalkyl. In some embodiments, the heterocycloalkyl is a 3- to 8- membered heterocycloalkenyl. In some embodiments, the heterocycloalkyl is a 3 - to 7-membered heterocycloalkenyl. In some embodiments, the heterocycloalkyl is a 3 - to 6-membered heterocycloalkenyl. In some embodiments, the heterocycloalkyl is a 4- to 6-membered heterocycloalkenyl. In some embodiments, the heterocycloalkyl is a 5 - to 6-membered
heterocycloalkenyl. Unless stated otherwise specifically in the specification, a heterocycloalkyl is optionally substituted, for example, with one or more oxo, halogen, amino, nitrile, nitro, hydroxyl, alkyl, alkenyl, alkynyl, haloalkyl, alkoxy, carboxyl, carboxylate, aryl, cycloalkyl, heterocycloalkyl, heteroaryl, and the like. In some embodiments, the heterocycloalkyl is optionally substituted with one or more oxo, halogen, methyl, ethyl, -CN, -COOH, -COOMe, -CF3, -OH, -OMe, -NH2, or -NO2. In some embodiments, the heterocycloalkyl is optionally substituted with one or more halogen, methyl, ethyl, - CN, -CF3, -OH, or -OMe. In some embodiments, the heterocycloalkyl is optionally substituted with halogen.
[0067] “Heteroaryl” refers to a 5- to 14-membered ring system radical comprising one to thirteen carbon atoms, one to six heteroatoms selected from the group consisting of nitrogen, oxygen, phosphorous, and sulfur, and at least one aromatic ring. In some embodiments, the heteroaryl comprises one to three heteroatoms selected from the group consisting of nitrogen, oxygen, and sulfur. In some embodiments, the heteroaryl comprises one to three heteroatoms selected from the group consisting of nitrogen and oxygen. In some embodiments, the heteroaryl comprises one to three nitrogens. In some embodiments, the heteroaryl comprises one or two nitrogens. In some embodiments, the heteroaryl comprises one nitrogen. In some embodiments, the heteroaryl is C-linked. In some embodiments, the heteroaryl is N- linked. The heteroaryl radical may be a monocyclic, bicyclic, tricyclic, or tetracyclic ring system, which may include fused (when fused with a cycloalkyl or heterocycloalkyl ring, the heteroaryl is bonded through an aromatic ring atom) or bridged ring systems; and the nitrogen, carbon, or sulfur atoms in the heteroaryl radical may be optionally oxidized; the nitrogen atom may be optionally quatemized. In some embodiments, the heteroaryl is a 5- to 10-membered heteroaryl comprising 1, 2, or 3 heteroatoms selected from the group consisting of oxygen, nitrogen, and sulfur. In some embodiments, the heteroaryl is a 5 - to 6-membered heteroaryl comprising 1, 2, or 3 heteroatoms selected from the group consisting of oxygen, nitrogen, and sulfur. In some embodiments, the heteroaryl is a 6-membered heteroaryl comprising 1, 2, or 3 heteroatoms selected from the group consisting of oxygen, nitrogen, and sulfur. In some embodiments, the heteroaryl is a 5 -membered heteroaryl comprising 1, 2, or 3 heteroatoms selected from the group consisting of oxygen, nitrogen, and sulfur. Examples include, but are not limited to, azepinyl, acridinyl, benzimidazolyl, benzothiazolyl, benzindolyl, benzodioxolyl, benzofuranyl, benzoxazolyl, benzothiadiazolyl, benzo[b][l,4]dioxepinyl, 1,4-benzodioxanyl, benzonaphthofuranyl, benzodioxinyl, benzopyranyl, benzopyranonyl, benzofuranyl, benzofuranonyl, benzothienyl (benzothiophenyl), benzotriazolyl, benzo[4,6]imidazo[l,2-a]pyridinyl, carbazolyl, cinnolinyl, dibenzofuranyl, dibenzothiophenyl, furanyl, isothiazolyl, imidazolyl, indazolyl, indolyl, , isoindolyl, indolinyl, isoindolinyl, isoquinolyl, indolizinyl, isoxazolyl, naphthyridinyl, oxadiazolyl, 2-oxoazepinyl, oxazolyl, 1-oxidopyridinyl, 1 -oxidopyrimidinyl, 1-oxidopyrazinyl, 1-oxidopyridazinyl, 1 -phenyl- 1H- pyrrolyl, phenazinyl, phenothiazinyl, phenoxazinyl, phthalazinyl, pteridinyl, purinyl, pyrrolyl, pyrazolyl, pyridinyl, pyrazinyl, pyrimidinyl, pyridazinyl, quinazolinyl, quinoxalinyl, quinolinyl, isoquinolinyl, tetrahydroquinolinyl, thiazolyl, thiadiazolyl, triazolyl, tetrazolyl, triazinyl, and thiophenyl (i.e., thienyl). Unless stated otherwise specifically in the specification, a heteroaryl is optionally substituted, for
example, with one or more halogen, amino, nitrile, nitro, hydroxyl, alkyl, alkenyl, alkynyl, haloalkyl, alkoxy, carboxyl, carboxylate, aryl, cycloalkyl, heterocycloalkyl, or heteroaryl, and the like. In some embodiments, the heteroaryl is optionally substituted with one or more halogen, methyl, ethyl, -CN, - COOH, -COOMe, -CF3, -OH, -OMe, -NH2, or -NO2. In some embodiments, the heteroaryl is optionally substituted with one or more halogen, methyl, ethyl, -CN, -CF3, -OH, or -OMe. In some embodiments, the heteroaryl is optionally substituted with halogen.
TRPM8 Antagonist
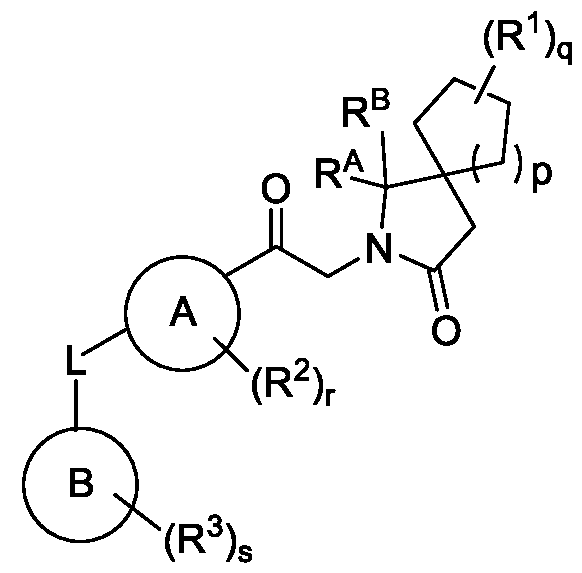
Formula (I); wherein:
A is aryl and heteroaryl;
B is aryl and heteroaryl;
L is independently selected from the group consisting of a chemical bond, oxygen, sulfur, -NR4-, - (CRcRD)t-, -O(CRcRD)t-, -(CRcRD)tO-, -N(R4)(CRcRD)t-, -(CRcRD)tN(R4)-, -N(R4)(CRcRD)tO-, and - O(CRcRD)tN(R4)-
X is independently selected from the group consisting of -CH2-, oxygen, sulfur, and NH;
RA and RB are independently selected from the group consisting of;
(1) hydrogen, (2) halogen, (3) (Ci-Cio)alkyl, (4) (C3-Cio)cycloalkyl and (5) (Ci-Cio)haloalkyl; or RA and RB may form oxo group (=0); or RA and RB may form a 3 to 8 membered ring which may contain one or more heteroatoms independently selected from oxygen, sulfur and nitrogen; and said ring is optionally substituted with 1 to 6 substituents independently selected from (1) hydrogen, (2) halogen, (3) hydroxy, (4) (Ci-Cio)alkyl, (5) (C3-Cio)cycloalkyl, (6) (Ci-Cio)haloalkyl, (7) (Ci- Cio)alkoxy and (8) ((Ci-Cio)haloalkoxy;
Rc and RD are independently selected from the group consisting of (1) hydrogen, (2) halogen, (3) (Ci- Cio)alkyl, (4) (C3-Cio)cycloalkyl and (5) (Ci-Cio)haloalkyl; or Rc and RD may form a 3 to 8 membered ring which may contain one or more heteroatoms independently selected from oxygen, sulfur and nitrogen; and said ring is optionally substituted with 1 to 6 substituents independently selected from (1) hydrogen, (2) halogen, (3) hydroxy, (4) (Ci-Cio)alkyl, (5) (C3-Cio)cycloalky, (6) (Ci-Cio)haloalkyl, (7) (Ci-Cio)alkoxy and (8) (Ci-Cio)haloalkoxy;
R1 is independently selected from the group consisting of (1) hydrogen, (2) halogen, (3) amino, (4) cyano, (5) hydroxyl, (6) (Ci-Cio)alkyl, (7) (C3-Cio)cycloalkyl, (8) (Ci-Cio)haloalkyl, (9) (Ci- Cio)alkoxy and (10) (Ci-Cio)haloalkoxy; two Ri on the same carbon or the different carbons are possible to form a 3 to 8 membered ring which may contain an atom selected from oxygen, sulfur and nitrogen; and said ring is optionally substituted with 1 to 6 substituents independently selected from (1) hydrogen, (2) halogen, (3) hydroxy, (4) (Ci-Cio)alkyl, (5) (C3-Cio)cycloalkyl, (6) (Ci- Cio)haloalkyl, (7) (Ci-Cio)alkoxy, and (8) (Ci-Cio)haloalkoxy;
R2 is independently selected from the group consisting of (1) hydrogen, (2) halogen, (3) amino, (4) - NH(Ci-Ce)alkyl, (5) -N[(Ci-Ce)alkyl]2 wherein the alkyl is same or different, (6) cyano, (7) hydroxyl, (8) nitro, (9) (Ci-C6)alkylthio, (10) (Ci-Cio)alkyl, (11) (C3-Cio)cycloalkyl, (12) (Ci-Cio)alkoxy, (13) (Ci-Cio)haloalkyl and (14) (Ci-Cio)haloalkoxy;
R3 is independently selected from the group consisting of (1) hydrogen, (2) halogen, (3) cyano, (4) nitro, (5) hydroxyl, (6) (Ci-C6)akylthio, (7) (Ci-C6)alkylsulfinyl, (8) (Ci-C6)alkylsulfonyl, (9) -NR5R6, (10) -C(=O)NR5R6, (11) tri(Ci-C6)alkylsilyl, (12) (Ci-Cio)alkyl, (13) (C3-Cio)cycloalkyl, (14) (Ci- C6)alkoxy(C0-C6)alkyl, (15) (C3-Cio)cycloalkoxy, (16) -C(=O)(Ci-C6)alkyl, (17) -C(=O)O(Ci- C6)alkyl and (18) -C(=O)OH; said (Ci-Cio)alkyl, (C3-Cio)cycloalkyl, (Ci-C6)alkoxy(C0-C6)alkyl and (C3-Cio)cycloalkoxy are optionally substituted with 1 to 6 substituents independently selected from (1) hydrogen, (2) halogen, (3) hydroxyl, (4) cyano, (5) (C3-Cio)cycloalkyl, (6) (Ci-Cio)haloalkyl, (7) (Ci-Cio)alkoxy, (8) (Cl-Cio)haloalkoxy and (9) -NR6R5; wherein R5 and R6, together with nitrogen atom to which they are attached, may form a 3 to 10 membered ring which may contain an atom selected from oxygen, sulfur and nitrogen; and said ring is optionally substituted with 1 to 6 substituents independently selected from (1) hydrogen, (2) halogen, (3) hydroxyl, (4) (Ci-Cio)alkyl, (5) (C3-Cio)cycloalkyl, (6) (Ci-Cio)haloalkyl, (7) (Ci- Cio)alkoxy and (8) (Cl-ClO)haloalkoxy;
R4, R5 and R6 are independently selected from the group consisting of (1) hydrogen, (2) (Ci-Cio)alkyl, (3) (C3-Cio)cycloalkyl, (4) (Ci-Cio)haloalkyl, (5) hydroxyl(Ci-Cio)alkyl, (6) (Ci-Cio)alkoxy(Ci-Cio)alkyl, (7) H2N-(Ci-Cio)alkyl, (8) [(Ci-Cio)alkyl]NH-( Ci-Cio)alkyl, (9) [(Ci-Cio)alkyl]2N-(Ci-Cio)alkyl, (10) (Ci-Cio)alkylcarbonyl and (11) (Ci-Cio)alkylsulfonyl; p is 1, 2, 3, or 4; q is 1, 2, 3, or 4; when q is two or more than two, R1 is same or different, r is 1, 2, 3, or 4; when r is two or more than two, R2 is same or different, s is 1, 2, 3, 4, 5, 6, or 7; when s is two or more than two, R3 is same or different, t is 1, 2, or 3; when t is two or more than two, Rc and RD are same or different, or a pharmaceutically acceptable salt thereof.
[0069] In some embodiments, the TRPM8 antagonist is a compound selected from the group consisting of:
3-(2-(2,5-dimethyl-l-(5-methylisoxazol-3-yl)-lH-pyrrol-3-yl)-2-oxoethyl)-8,8-difluoro-l,3- diazaspiro[4.5]decane-2, 4-dione;
3-(2-(2,5-dimethyl-l-phenyl-lH-imidazol-4-yl)-2-oxoethyl)-8,8-difluoro-l,3-diazaspiro-[4.5]decane-2,4- dione;
6-(4-(2-(2,4-dioxo-l,3-diazaspiro[4.5]decan-3-yl)acetyl)phenyl)picolinonitrile;
3-(2-(l-(3-chlorophenyl)-2,5-dimethyl-lH-imidazol-4-yl)-2-oxoethyl)-8,8-difluoro-l,3-di- azaspiro [4.5] decane-2, 4-dione ;
8.8-difluoro-3-(2-(l-(3-fluorophenyl)-2,5-dimethyl-lH-imidazol-4-yl)-2-oxoethyl)-l,3-di- azaspiro [4.5] decane-2, 4-dione ;
3-(2-(l,4-dimethyl-5-phenyl-lH-pyrazol-3-yl)-2-oxoethyl)-8,8-difluoro-l,3-diazaspiro-[4.5]decane-2,4- dione;
8.8-difluoro-3-(2-(4-(6-methylpyrazin-2-yl)phenyl)-2-oxoethyl)-l,3-diazaspiro[4.5]decane-2, 4-dione;
6-(4-(2-(8,8-difluoro-2,4-dioxo-l,3-diazaspiro[4.5]decan-3-yl)acetyl)phenyl)picolinonitrile;
8.8-difluoro-3-(2-(2'-(hydroxymethyl)-[l,r-biphenyl]-4-yl)-2-oxoethyl)-l,3-diazaspiro-[4.5]decane-2,4- dione;
8.8-difluoro-3-(2-(4-(3-methylpyrazin-2-yl)phenyl)-2-oxoethyl)-l,3-diazaspiro[4.5]decane-2, 4-dione;
3-(2-(2,5-dimethyl-l-phenyl-lH-imidazol-4-yl)-2-oxoethyl)-8,8-difluoro-l-oxa-3-azaspi-ro[4.5]decane-
2,4-dione;
8.8-difluoro-3-(2-(4-(2-(hydroxymethyl)pyridin-3-yl)phenyl)-2-oxoethyl)-l,3-diazaspiro-[4.5]decane-
2,4-dione;
8.8-difluoro-3-(2-(4-(4-(hydroxymethyl)pyridin-3-yl)phenyl)-2-oxoethyl)-l,3-diazaspiro-[4.5]decane-
2,4-dione;
3 -(2-(2, 5 -dimethyl -l-(5 -methylisoxazol-3 -yl)- lH-pyrrol-3 -yl)-2-oxoethyl)-8, 8-difluoro-l-oxa-3 - azaspiro [4.5] decane-2, 4-dione ;
8.8-difluoro-3-(2-(5-(2-(hydroxymethyl)phenyl)-4-methylthiophen-2-yl)-2-oxoethyl)-l,3- diazaspiro[4.5]decane-2, 4-dione;
3-(2-(4-(3-(hydroxymethyl)pyrazin-2-yl)phenyl)-2-oxoethyl)-l,3-diazaspiro[4.5]decane-2, 4-dione;
3-(2-(l,4-dimethyl-5-phenyl-lH-pyrazol-3-yl)-2-oxoethyl)-8,8-difluoro-l-oxa-3-azaspiro-[4.5]decane-
2,4-dione;
8.8-difluoro-3-(2-(4-(4-methylpyridazin-3-yl)phenyl)-2-oxoethyl)-l,3-diazaspiro[4.5]decane-2, 4-dione;
3-(4-(2-(8,8-difluoro-2,4-dioxo-l,3-diazaspiro[4.5]decan-3-yl)acetyl)phenyl)pyrazine-2 -carbonitrile;
3-(2-(l,4-dimethyl-5-phenyl-lH-pyrrol-2-yl)-2-oxoethyl)-8,8-difluoro-l,3-diazaspiro[4.5]-decane-2,4- dione;
8.8-difluoro-3-(2-(4-(3-(hydroxymethyl)pyrazin-2-yl)phenyl)-2-oxoethyl)-l,3-diazaspiro-[4.5]decane-
2,4-dione;
3-(2-(6-(methyl(pyridin-2-yl)amino)pyridin-3-yl)-2-oxoethyl)-l,3-diazaspiro[4.5]decane-2, 4-dione;
3-(4-(2-(8,8-difluoro-2,4-dioxo-l,3-diazaspiro[4.5]decan-3-yl)acetyl)phenyl)picolinonitrile;
8.8-difluoro-3-(2-oxo-2-(4-(quinolin-8-yl)phenyl)ethyl)-l,3-diazaspiro[4.5]decane-2, 4-dione;
3-(2-(4-(lH-indol-4-yl)phenyl)-2-oxoethyl)-8,8-difluoro-l,3-diazaspiro[4.5]decane-2, 4-dione;
8.8-difluoro-3-(2-oxo-2-(4-(quinolin-2-yl)phenyl)ethyl)-l,3-diazaspiro[4.5]decane-2, 4-dione;
8.8-difluoro-3-(2-(4-(isoquinolin-8-yl)phenyl)-2-oxoethyl)-l,3-diazaspiro[4.5]decane-2, 4-dione;
8.8-difluoro-3-(2-(4-(isoquinolin-l-yl)phenyl)-2 -oxoethyl)- l,3-diazaspiro[4.5]decane-2, 4-dione;
8.8-difluoro-3-(2-(4-(furo[3,2-c]pyridin-4-yl)phenyl)-2-oxoethyl)-l,3-diazaspiro[4.5]decane-2, 4-dione;
8.8-difluoro-3-(2-(6-(methyl(pyridin-2-yl)amino)pyridin-3-yl)-2-oxoethyl)-l,3-diazaspiro[4.5]decane-
2,4-dione;
8.8-difluoro-3 -(2-(6-(methyl(phenyl)amino)pyridin-3 -yl)-2 -oxoethyl)- 1 ,3 -diazaspiro [4.5] -decane-2, 4- dione;
8.8-difluoro-3-(2-(4-(3-fluoropyridin-2-yl)phenyl)-2-oxoethyl)-l,3-diazaspiro[4.5]decane-2, 4-dione;
3-(4-(2-(8,8-difluoro-2,4-dioxo-l,3-diazaspiro[4.5]decan-3-yl)acetyl)phenyl)isonicotino-nitrile;
8.8-difluoro-3-(2-(4-(2-methoxypyridin-3-yl)phenyl)-2-oxoethyl)-l,3-diazaspiro[4.5]decane-2, 4-dione;
8.8-difluoro-3-(2-(4-(4-methoxypyridin-3-yl)phenyl)-2-oxoethyl)-l,3-diazaspiro[4.5]decane-2, 4-dione;
8.8-difluoro-3-(2-oxo-2-(4-(2-oxoindolin-4-yl)phenyl)ethyl)-l,3-diazaspiro[4.5]decane-2, 4-dione;
3-(2-(4-(7H-pyrrolo[2,3-d]pyrimidin-4-yl)phenyl)-2-oxoethyl)-8,8-difluoro-l,3-diazaspiro[4.5]decane-
2,4-dione;
3-(2-(4-(lH-pyrrolo[3,2-c]pyridin-4-yl)phenyl)-2-oxoethyl)-8,8-difluoro-l,3-diazaspiro-[4.5]decane-2,4- dione;
3-(2-(4-(lH-pyrrolo[2,3-b]pyridin-4-yl)phenyl)-2-oxoethyl)-8,8-difluoro-l,3-diazaspiro-[4.5]decane-2,4- dione;
3-(2-(4-(3-chloropyridin-2-yl)phenyl)-2-oxoethyl)-8,8-difluoro-l,3-diazaspiro[4.5]decane-2, 4-dione;
8.8-difluoro-3-(2-(4-(2-methyl-lH-benzo[d]imidazol-l-yl)phenyl)-2-oxoethyl)-l,3- diazaspiro[4.5]decane-2, 4-dione;
3-(2-(4-(lH-indazol-4-yl)phenyl)-2-oxoethyl)-8,8-difluoro-l,3-diazaspiro[4.5]decane-2, 4-dione;
3-(2-(6-(lH-indazol-4-yl)pyridin-3-yl)-2-oxoethyl)-8,8-difluoro-l,3-diazaspiro[4.5]decane-2, 4-dione;
3-(2-(4-(lH-benzo[d]imidazol-l-yl)phenyl)-2-oxoethyl)-8,8-difluoro-l,3-diazaspiro[4.5]-decane-2,4- dione;
3-(5-(2-(8,8-difluoro-2,4-dioxo-l,3-diazaspiro[4.5]decan-3-yl)acetyl)-l,3-dimethyl-lH-pyrrol-2- yl)benzonitrile;
3-(2-(4-(lH-pyrrolo[2,3-c]pyridin-4-yl)phenyl)-2-oxoethyl)-8,8-difluoro-l,3-diazaspiro-[4.5]decane-2,4- dione;
8.8-difluoro-3-(2-(3-fluoro-4-(quinolin-8-yl)phenyl)-2-oxoethyl)-l,3-diazaspiro[4.5]decane-2, 4-dione;
3-(5-(2-(8,8-difluoro-2,4-dioxo-l,3-diazaspiro[4.5]decan-3-yl)acetyl)-l-methyl-lH-pyrrol-2- yl)benzonitrile;
3-(5-(2-(8,8-difluoro-2,4-dioxo-l,3-diazaspiro[4.5]decan-3-yl)acetyl)-3-methylthiophen-2- yl)benzonitrile;
8.8-difluoro-3-(2-(4-(2-(2-hydroxyethoxy)pyridin-3-yl)phenyl)-2-oxoethyl)-l,3-diazaspiro[4.5]decane-
2,4-dione;
8.8-difluoro-3-(2-(5-(3-fluorophenyl)-l,4-dimethyl-lH-pyrrol-2-yl)-2-oxoethyl)-l,3- diazaspiro[4.5]decane-2, 4-dione;
3 -(2-(5 -(3 -chlorophenyl)- 1 ,4-dimethyl- lH-pyrrol-2-yl)-2-oxoethyl)-8, 8-difluoro- 1,3- diazaspiro[4.5]decane-2, 4-dione;
3-(5-(2-(8,8-difluoro-2,4-dioxo-l,3-diazaspiro[4.5]decan-3-yl)acetyl)-l,3-dimethyl-lH-pyrrol-2- yl)benzamide;
8.8-difluoro-3-(2-(5-(2-fluorophenyl)-l,4-dimethyl-lH-pyrrol-2-yl)-2-oxoethyl)-l,3- diazaspiro[4.5]decane-2, 4-dione;
3-(2-(4-(lH-pyrazolo[3,4-b]pyridin-4-yl)phenyl)-2-oxoethyl)-8,8-difluoro-l,3-diazaspiro-[4.5]decane-
2,4-dione;
3-(2-(4-(lH-pyrazolo[4,3-c]pyridin-4-yl)phenyl)-2-oxoethyl)-8,8-difluoro-l,3-diazaspiro-[4.5]decane-
2,4-dione;
3-(2-(4-(lH-indazol-l-yl)phenyl)-2-oxoethyl)-8,8-difluoro-l,3-diazaspiro[4.5]decane-2, 4-dione;
8.8-difluoro-3-(2-(5-(3-fluorophenyl)-l-methyl-lH-imidazol-2-yl)-2-oxoethyl)-l,3- diazaspiro [4.5] decane -2,4-dione ;
8.8-difluoro-3-(2-(4-(2-methyl-3H-imidazo[4,5-b]pyridin-3-yl)phenyl)-2-oxoethyl)-l,3- diazaspiro[4.5]decane-2, 4-dione;
8.8-difluoro-3-(2-oxo-2-(4-(pyridin-2-yloxy)phenyl)ethyl)-l,3-diazaspiro[4.5]decane-2, 4-dione;
3-(2-(5-(3,5-difluorophenyl)-l,4-dimethyl-lH-pyrrol-2-yl)-2-oxoethyl)-8,8-difluoro-l,3- diazaspiro[4.5]decane-2, 4-dione;
8.8-difluoro-3-(2-(2'-methyl-[3,3'-bipyridin]-6-yl)-2-oxoethyl)-l,3-diazaspiro[4.5]decane-2, 4-dione;
3-(2-(4-(lH-pyrazolo[3,4-d]pyrimidin-4-yl)phenyl)-2-oxoethyl)-8,8-difluoro-l,3-diazaspiro[4.5]decane-
2,4-dione;
3-(2-(6-(lH-pyrrolo[2,3-c]pyridin-4-yl)pyridin-3-yl)-2-oxoethyl)-8,8-difluoro-l,3-diazaspiro[4.5]decane-
2,4-dione;
8.8-difluoro-3-(2-(4-(3-(2-hydroxyethoxy)pyrazin-2-yl)phenyl)-2-oxoethyl)-l,3-diazaspiro[4.5]decane-
2,4-dione;
8.8-difluoro-3-(2-oxo-2-(4-(phthalazin-l-yl)phenyl)ethyl)-l,3-diazaspiro[4.5]decane-2, 4-dione;
3 -(2-(4-(3H-imidazo[4,5 -b]pyridin-3 -yl)phenyl)-2-oxoethyl)-8, 8-difluoro- 1 ,3 -diazaspiro-[4.5] decane-
2, 4-dione;
3-(2-(2-(8,8-difluoro-2,4-dioxo-l,3-diazaspiro[4.5]decan-3-yl)acetyl)-4-methylthiazol-5- yl)benzonitrile;
3-(2-(l,4-dimethyl-5-phenyl-lH-imidazol-2-yl)-2-oxoethyl)-8,8-difluoro-l,3-diazaspiro-[4.5]decane-2,4- dione;
8.8-difluoro-3-(2-(5-(3-fluorophenyl)-l,4-dimethyl-lH-imidazol-2-yl)-2-oxoethyl)-l,3- diazaspiro [4.5] decane -2,4-dione ;
3-(2-(2-(8,8-difluoro-2,4-dioxo-l,3-diazaspiro[4.5]decan-3-yl)acetyl)-l,4-dimethyl-lH-imidazol-5- yl)benzonitrile;
8.8-difluoro-3-(2-(5-(isoquinolin-8-yl)-l,4-dimethyl-lH-imidazol-2-yl)-2-oxoethyl)-l,3- diazaspiro[4.5]decane-2, 4-dione;
8.8-difluoro-3-(2-(5-(2-(hydroxymethyl)phenyl)-l,4-dimethyl-lH-imidazol-2-yl)-2-oxo-ethyl)-l,3- diazaspiro[4.5]decane-2, 4-dione;
8.8-difluoro-3-(2-(4-(2-(hydroxymethyl)-lH-benzo[d]imidazol-l-yl)phenyl)-2-oxoethyl)-l,3- diazaspiro[4.5]decane-2, 4-dione;
8.8-difluoro-3-(2-(5-(3-fluorophenyl)-l,4-dimethyl-lH-pyrrol-3-yl)-2-oxoethyl)-l,3- diazaspiro[4.5]decane-2, 4-dione;
3-(4-(2-(8,8-difluoro-2,4-dioxo-l,3-diazaspiro[4.5]decan-3-yl)acetyl)-l,3-dimethyl-lH-pyrrol-2- yl)benzonitrile;
3-(2-(5-(lH-benzo[d]imidazol-l-yl)pyrazin-2-yl)-2-oxoethyl)-8,8-difluoro-l,3-diazaspiro-[4.5]decane-
2,4-dione;
3-(2-(4-(2,7-naphthyridin-l-yl)phenyl)-2-oxoethyl)-8,8-difluoro-l,3-diazaspiro[4.5]decane-2, 4-dione;
8.8-difluoro-3 -(2-oxo-2-(4-(2-oxo-2,3 -dihydro- IH-benzo [d] imidazol- 1 -yl)phenyl)ethyl)- 1 ,3- diazaspiro[4.5]decane-2, 4-dione;
8.8-difluoro-3-(2-(5-(2-(hydroxymethyl)phenyl)pyrazin-2-yl)-2-oxoethyl)-l,3-diazaspiro[4.5]decane-2,4- dione;
8.8-difluoro-3-(2-(5-(4-methoxypyridin-3-yl)pyrazin-2-yl)-2-oxoethyl)-l,3-diazaspiro-[4.5]decane-2,4- dione;
3-(5-(2-(8,8-difluoro-2,4-dioxo-l,3-diazaspiro[4.5]decan-3-yl)acetyl)-2,4-dimethylthiophen-3- yl)benzamide;
3 -(2-(5 -(3 ,5 -difluorophenyl)- 1 ,4-dimethyl- lH-imidazol-2-yl)-2-oxoethyl)-8, 8-difluoro- 1,3- diazaspiro[4.5]decane-2, 4-dione;
8.8-difluoro-3-(2-oxo-2-(4-(pyridazin-3-yloxy)phenyl)ethyl)-l,3-diazaspiro[4.5]decane-2, 4-dione;
8.8-difluoro-3-(2-oxo-2-(4-(2-oxo-lH-imidazo[4,5-b]pyridin-3(2H)-yl)phenyl)ethyl)-l,3- diazaspiro[4.5]decane-2, 4-dione;
3-(2-(5-(3,5-difluorophenyl)-4-methylthiazol-2-yl)-2-oxoethyl)-8,8-difluoro-l,3-diazaspiro[4.5]decane-
2,4-dione;
4'-(2-(8,8-difluoro-2,4-dioxo-l,3-diazaspiro[4.5]decan-3-yl)acetyl)-2'-methoxy-[l,r-biphenyl]-2- carbonitrile;
2-(4-(2-(8,8-difluoro-2,4-dioxo-l,3-diazaspiro[4.5]decan-3-yl)acetyl)phenoxy)nicotinonitrile;
3 -(2-(4-((3 -chloropyridin-2-yl)oxy)phenyl)-2-oxoethyl)-8, 8-difluoro- 1 ,3 -diazaspiro [4.5] -decane-2, 4- dione;
8.8-difluoro-3-(2-(4-(3-(hydroxymethyl)pyridin-2-yl)phenyl)-2-oxoethyl)-l,3-diazaspiro-[4.5]decane-
2,4-dione;
3-(2-(2'-(aminomethyl)-[l,r-biphenyl]-4-yl)-2-oxoethyl)-8,8-difluoro-l,3-diazaspiro[4.5]-decane-2,4- dione;
8.8-difluoro-3-(2-oxo-2-(6-(quinolin-8-yl)pyridin-3-yl)ethyl)-l,3-diazaspiro[4.5]decane-2, 4-dione;
8.8-difluoro-3 -(2-(5 -(2-methylpyridin-3 -yl)pyrazin-2-yl)-2 -oxoethyl)- 1 ,3 -diazaspiro [4.5] -decane-2, 4- dione;
3-(2-(4-(2,7-naphthyridin-l-yl)phenyl)-2-oxoethyl)-8,8-difluoro-l-oxa-3-azaspiro[4.5]decane-2, 4-dione; 3-(4-(2-(8,8-difluoro-2,4-dioxo-l,3-diazaspiro[4.5]decan-3-yl)acetyl)phenyl)-2-methyl-3H-imidazo[4,5 - b] pyridine -5 -carbonitrile ;
8.8-difluoro-3-(2-oxo-2-(4-(2-oxobenzo[d]oxazol-3(2H)-yl)phenyl)ethyl)-l,3-diazaspiro-[4.5]decane- 2,4-dione;
3-(2-(4-(2,5-dimethyl-3H-imidazo[4,5-b]pyridin-3-yl)phenyl)-2-oxoethyl)-8,8-difluoro-l,3- diazaspiro[4.5]decane-2, 4-dione;
8.8-difluoro-3-(2-(5-(2-methyl-lH-benzo[d]imidazol-l-yl)pyrazin-2-yl)-2-oxoethyl)-l,3- diazaspiro[4.5]decane-2, 4-dione;
8.8-difluoro-3 -(2-(4-(2-methoxy-5 -methyl-3H-imidazo [4,5 -b]pyridin-3 -yl)phenyl)-2 -oxo-ethyl)- 1 ,3- diazaspiro[4.5]decane-2, 4-dione;
8.8-difluoro-3 -(2-(4-(5 -methyl -2-(trifluoromethyl)-3H-imidazo [4,5 -b]pyridin-3 -yl)phenyl)-2-oxoethyl)-
1 ,3 -diazaspiro [4.5] decane-2, 4-dione;
3-(2-(4-(2-(difluoromethyl)-5-methyl-3H-imidazo[4,5-b]pyridin-3-yl)phenyl)-2-oxoethyl)-8,8-difluoro-
1 ,3 -diazaspiro [4.5] decane-2, 4-dione;
8.8-difluoro-3-(2-(4-(5-methyl-2-oxo-lH-imidazo[4,5-b]pyridin-3(2H)-yl)phenyl)-2-oxo-ethyl)-l,3- diazaspiro[4.5]decane-2, 4-dione;
6-(4-(2-(8,8-difluoro-2,4-dioxo-l,3-diazaspiro[4.5]decan-3-yl)acetyl)phenoxy)picolinonitrile;
8.8-difluoro-3 -(2-(4-(5 -methyl-3H-imidazo[4,5 -b]pyridin-3 -yl)phenyl)-2 -oxoethyl)- 1,3- diazaspiro[4.5]decane-2, 4-dione;
8.8-difluoro-3-(2-(4-(3-methoxypyrazin-2-yl)phenyl)-2-oxoethyl)-l,3-diazaspiro[4.5]decane-2, 4-dione;
3-(4-(2-(8,8-difluoro-2,4-dioxo-l,3-diazaspiro[4.5]decan-3-yl)acetyl)-3-fluorophenyl)pyrazine-2- carbonitrile;
8.8-difluoro-3-(2-(4-(imidazo[l,2-b]pyridazin-3-yl)phenyl)-2-oxoethyl)-l,3-diazaspiro-[4.5]decane-2,4- dione;
8.8-difluoro-3-(2-(2'-(2-hydroxyethyl)-[l,r-biphenyl]-4-yl)-2-oxoethyl)-l,3-diazaspiro-[4.5]decane-2,4- dione;
2-(4'-(2-(8,8-difluoro-2,4-dioxo-l,3-diazaspiro[4.5]decan-3-yl)acetyl)-[l,r-biphenyl]-2-yl)acetonitrile;
3-(2-(4-(lH-imidazo[4,5-b]pyrazin-l-yl)phenyl)-2-oxoethyl)-8,8-difluoro-l,3-diazaspiro-[4.5]decane- 2,4-dione;
8.8-difluoro-3-(2-(4-(4-methylpyridazin-3-yl)phenyl)-2-oxoethyl)-l-oxa-3-azaspiro[4.5]-decane-2,4- dione;
3-(4-(2-(8,8-difluoro-2,4-dioxo-l,3-diazaspiro[4.5]decan-3-yl)acetyl)phenoxy)pyridazine-4-carbonitrile;
8.8-difluoro-3-(2-(4-(2-(hydroxymethyl)-5-methyl-3H-imidazo[4,5-b]pyridin-3-yl)phenyl)-2 -oxoethyl)-
1 ,3 -diazaspiro [4.5] decane-2, 4-dione;
8.8-difluoro-3-(2-oxo-2-(4-(pyrazolo[l,5-a]pyrimidin-3-yl)phenyl)ethyl)-l,3-diazaspiro-[4.5]decane-2,4- dione;
4-(4-(2-(8,8-difluoro-2,4-dioxo-l,3-diazaspiro[4.5]decan-3-yl)acetyl)phenyl)nicotinonitrile;
8.8-difluoro-3-(2-(2-fluoro-4-(4-methylpyridazin-3-yl)phenyl)-2-oxoethyl)-l,3-diazaspiro[4.5]decane-
2,4-dione;
8.8-difluoro-3-(2-(2-fluoro-4-(3-(hydroxymethyl)pyrazin-2-yl)phenyl)-2-oxoethyl)-l,3-di- azaspiro [4.5] decane-2, 4-dione ; and
2-(4-(2-(8,8-difluoro-2,4-dioxo-l,3-diazaspiro[4.5]decan-3-yl)acetyl)phenyl)nicotinonitrile; or a pharmaceutically acceptable salt thereof.
Compound 1
[0070] In some embodiments, the TRPM8 antagonist is Compound 1, or a pharmaceutically acceptable salt thereof. Compound 1 is 8, 8-difluoro-3-(2-(4-(4-methylpyridazin-3-yl)phenyl)-2 -oxoethyl)-!, 3-
Compound 1 is in the form of a salt. In some embodiments, Compound 1 is in the form of a besylate salt.
[0071] In some embodiments, the TRPM8 antagonist is AMG-333, elismetrep, M8-B, PF-05105679,
Further Forms of Compounds Disclosed Herein
Isomers/Stereoisomers
[0072] In some embodiments, the compounds described herein exist as geometric isomers. In some embodiments, the compounds described herein possess one or more double bonds. The compounds
presented herein include all cis, trans, syn, anti, entgegen (E), and zusammen (Z) isomers as well as the corresponding mixtures thereof. In some situations, the compounds described herein possess one or more chiral centers and each center independently exists in the R configuration or S configuration. The compounds described herein include all diastereomeric, enantiomeric, and epimeric forms as well as the corresponding mixtures thereof. In additional embodiments of the compounds and methods provided herein, mixtures of enantiomers and/or diastereoisomers, resulting from a single preparative step, combination, or interconversion are useful for the applications described herein. In some embodiments, the compounds described herein are prepared as their individual stereoisomers by reacting a racemic mixture of the compound with an optically active resolving agent to form a pair of diastereoisomeric compounds, separating the diastereomers and recovering the optically pure enantiomers. In some embodiments, dissociable complexes are preferred. In some embodiments, the diastereomers have distinct physical properties (e.g., melting points, boiling points, solubilities, reactivity, etc.) and are separated by taking advantage of these dissimilarities. In some embodiments, the diastereomers are separated by chiral chromatography, or preferably, by separation/resolution techniques based upon differences in solubility. In some embodiments, the optically pure enantiomer is then recovered, along with the resolving agent, by any practical means that would not result in racemization.
Labeled compounds
[0073] In some embodiments, the compounds described herein exist in their isotopically -labeled forms. In some embodiments, the methods disclosed herein include methods of treating diseases by administering such isotopically-labeled compounds. In some embodiments, the methods disclosed herein include methods of treating diseases by administering such isotopically-labeled compounds as pharmaceutical compositions. Thus, in some embodiments, the compounds disclosed herein include isotopically-labeled compounds, which are identical to those recited herein, but for the fact that one or more atoms are replaced by an atom having an atomic mass or mass number different from the atomic mass or mass number usually found in nature. Examples of isotopes that can be incorporated into compounds disclosed herein include isotopes of hydrogen, carbon, nitrogen, oxygen, phosphorous, sulfur, fluorine, and chlorine, such as 2H, 3H, 13C, 14C, 15N, 180, 170, 31P, 32P, 35S, 18F, and 36C1, respectively. Compounds described herein, and the pharmaceutically acceptable salts thereof which contain the aforementioned isotopes and/or other isotopes of other atoms are within the scope of this invention. Certain isotopically-labeled compounds, for example those into which radioactive isotopes such as 3H and 14C are incorporated, are useful in drug and/or substrate tissue distribution assays. Tritiated, i.e., 3H and carbon-14, i.e., 14C, isotopes are particularly preferred for their ease of preparation and detectability. Further, substitution with heavy isotopes such as deuterium, i.e., 2H, produces certain therapeutic advantages resulting from greater metabolic stability, for example increased in vivo half-life or reduced dosage requirements. In some embodiments, one or more hydrogen in a compound disclosed herein has been replaced by a deuterium atom. In some embodiments, one or more alkyl substituents in a compound disclosed herein has been replaced by a deuteroalkyl substituents.
[0074] In some embodiments, the compounds described herein are labeled by other means, including, but not limited to, the use of chromophores or fluorescent moieties, biolumine scent labels, or chemiluminescent labels.
Pharmaceutically acceptable salts
[0075] In some embodiments, the compounds described herein exist as their pharmaceutically acceptable salts. In some embodiments, the methods disclosed herein include methods of treating diseases by administering such pharmaceutically acceptable salts. In some embodiments, the methods disclosed herein include methods of treating diseases by administering such pharmaceutically acceptable salts as pharmaceutical compositions.
[0076] In some embodiments, the compounds described herein possess acidic or basic groups and therefore react with any of a number of inorganic or organic bases, and inorganic and organic acids, to form a pharmaceutically acceptable salt. In some embodiments, these salts are prepared in situ during the final isolation and purification of the compounds disclosed herein, or by separately reacting a purified compound in its free form with a suitable acid or base, and isolating the salt thus formed.
[0077] Examples of pharmaceutically acceptable salts include those salts prepared by reaction of the compounds described herein with a mineral, organic acid or inorganic base, such salts including, but not limited to, acetate, acrylate, adipate, alginate, aspartate, benzoate, benzenesulfonate, bisulfate, bisulfite, bromide, butyrate, butyn-l,4-dioate, camphorate, camphorsulfonate, caproate, caprylate, chlorobenzoate, chloride, citrate, cyclopentanepropionate, decanoate, digluconate, gluconate, dihydrogenphosphate, dinitrobenzoate, dodecylsulfate, ethanesulfonate, formate, fumarate, glucoheptanoate, glycerophosphate, glycolate, hemisulfate, heptanoate, hexanoate, hexyne- 1,6-dioate, hydroxybenzoate, y-hydroxybutyrate, hydrochloride, hydrobromide, hydroiodide, 2-hydroxyethanesulfonate, iodide, isobutyrate, lactate, maleate, malonate, methanesulfonate, mandelate metaphosphate, methoxybenzoate, methylbenzoate, monohydrogenphosphate, 1-napthalenesulfonate, 2-napthalenesulfonate, nicotinate, nitrate, palmoate, pectinate, persulfate, 3 -phenylpropionate, phosphate, picrate, pivalate, propionate, pyrosulfate, pyrophosphate, propiolate, phthalate, phenylacetate, phenylbutyrate, propanesulfonate, salicylate, succinate, sulfate, sulfite, succinate, suberate, sebacate, sulfonate, tartrate, thiocyanate, tosylate, undecanoate, and xylenesulfonate.
[0078] Further, the compounds described herein can be prepared as pharmaceutically acceptable salts formed by reacting the free base form of the compound with a pharmaceutically acceptable inorganic or organic acid, including, but not limited to, inorganic acids such as hydrochloric acid, hydrobromic acid, sulfuric acid, nitric acid, phosphoric acid metaphosphoric acid, and the like; and organic acids such as acetic acid, propionic acid, hexanoic acid, cyclopentanepropionic acid, glycolic acid, pyruvic acid, lactic acid, malonic acid, succinic acid, malic acid, maleic acid, fumaric acid, p-toluenesulfonic acid, tartaric acid, trifluoroacetic acid, citric acid, benzoic acid, 3-(4-hydroxybenzoyl)benzoic acid, cinnamic acid, mandelic acid, arylsulfonic acid, methane sulfonic acid, ethanesulfonic acid, 1,2-ethanedisulfonic acid, 2- hydroxyethanesulfonic acid, benzenesulfonic acid, 2-naphthalenesulfonic acid, 4-methylbicyclo- [2.2.2]oct-2-ene-l-carboxylic acid, glucoheptonic acid, 4,4 ’-methylenebis-(3 -hydroxy-2 -ene-1-
carboxylic acid), 3 -phenylpropionic acid, trimethylacetic acid, tertiary butylacetic acid, lauryl sulfuric acid, gluconic acid, glutamic acid, hydroxynaphthoic acid, salicylic acid, stearic acid and muconic acid. In some embodiments, other acids, such as oxalic, while not in themselves pharmaceutically acceptable, are employed in the preparation of salts useful as intermediates in obtaining the compounds disclosed herein and their pharmaceutically acceptable acid addition salts.
[0079] In some embodiments, those compounds described herein which comprise a free acid group react with a suitable base, such as the hydroxide, carbonate, bicarbonate, sulfate, of a pharmaceutically acceptable metal cation, with ammonia, or with a pharmaceutically acceptable organic primary, secondary, tertiary, or quaternary amine. Representative salts include the alkali or alkaline earth salts, like lithium, sodium, potassium, calcium, and magnesium, and aluminum salts and the like. Illustrative examples of bases include sodium hydroxide, potassium hydroxide, choline hydroxide, sodium carbonate, N+(CI.C4 alkyl)4 hydroxide, and the like.
[0080] Representative organic amines useful for the formation of base addition salts include ethylamine, diethylamine, ethylenediamine, ethanolamine, diethanolamine, piperazine and the like. It should be understood that the compounds described herein also include the quatemization of any basic nitrogencontaining groups they contain. In some embodiments, water or oil-soluble or dispersible products are obtained by such quatemization.
Method of treatment
[0081] Disclosed herein is a method of treating cancer in a subject in need thereof, the method comprising administering to the subject a TRPM8 antagonist.
[0082] In some embodiments of a method of treating cancer, the cancer is bladder cancer, breast cancer, cholangiocarcinoma, colorectal cancer, kidney cancer, liver cancer, lung cancer, lymphoma, myeloma, osteosarcoma, ovarian cancer, pancreatic cancer, prostate cancer, or skin cancer.
[0083] In some embodiments of a method of treating cancer, the cancer is prostate cancer.
[0084] In some embodiments of a method of treating cancer, the breast cancer is ER positive breast cancer, ER positive metastatic breast cancer, or triple negative breast cancer (TNB).
[0085] In some embodiments of a method of treating cancer, the myeloma is multiple myeloma (MM). [0086] In some embodiments of a method of treating cancer, the skin cancer is melanoma.
[0087] In some embodiments of a method of treating cancer, the lung cancer is small cell lung cancer (SCLC) or non-small cell lung cancer (NSCLC).
[0088] In some embodiments of a method of treating cancer, the cancer is multiple myeloma.
[0089] In some embodiments of a method of treating cancer, the cancer is breast cancer.
[0090] In some embodiments of a method of treating cancer, the cancer is triple negative breast cancer
(TNB).
[0091] In some embodiments of a method of treating cancer, the cancer is melanoma.
[0092] In some embodiments of a method of treating cancer, the cancer is osteosarcoma.
[0093] In some embodiments of a method of treating cancer, the cancer is small cell lung cancer (SCLC) .
[0094] In some embodiments of a method of treating cancer, the cancer is kidney cancer. [0095] In some embodiments of a method of treating cancer, the cancer is bladder cancer. [0096] In some embodiments of a method of treating cancer, the cancer is ovarian cancer. [0097] In some embodiments of a method of treating cancer, the cancer is prostate cancer. [0098] In some embodiments of a method of treating cancer, the cancer is colorectal cancer. [0099] In some embodiments of a method of treating cancer, the cancer is lymphoma.
[00100] In some embodiments, the compounds and pharmaceutical compositions of the present disclosure are useful in the treatment or prevention of progression of cancer. In some embodiments, the cancer is a hematologic malignancy or solid tumor. Hematologic malignancies include leukemias, lymphomas, multiple myeloma, and subtypes thereof. Lymphomas can be classified various ways, often based on the underlying type of malignant cell, including Hodgkin’s lymphoma (often cancers of Reed-Sternberg cells, but also sometimes originating in B cells; all other lymphomas are non-Hodgkin’s lymphomas), B- cell lymphomas, T-cell lymphomas, mantle cell lymphomas, Burkitt’s lymphoma, follicular lymphoma, and others as defined herein and known in the art.
[00101] B-cell lymphomas include, but are not limited to, diffuse large B-cell lymphoma (DLBCL), chronic lymphocytic leukemia (CLL) /small lymphocytic lymphoma (SLL), and others as defined herein and known in the art.
[00102] T-cell lymphomas include T-cell acute lymphoblastic leukemia/lymphoma (T-ALL), peripheral T-cell lymphoma (PTCL), T-cell chronic lymphocytic leukemia (T-CLL) Sezary syndrome, and others as defined herein and known in the art.
[00103] Leukemias include acute myeloid (or myelogenous) leukemia (AML), chronic myeloid (or myelogenous) leukemia (CML), acute lymphocytic (or lymphoblastic) leukemia (ALL), chronic lymphocytic leukemia (CLL) hairy cell leukemia (sometimes classified as a lymphoma) and others as defined herein and known in the art.
[00104] Plasma cell malignancies include lymphoplasmacytic lymphoma, plasmacytoma, and multiple myeloma.
[00105] Solid tumors include melanomas, neuroblastomas, gliomas or 5 carcinomas such as tumors of the brain, head and neck, breast, lung (e.g., non-small cell lung cancer, NSCLC), reproductive tract (e.g., ovary), upper digestive tract, pancreas, liver, renal system (e.g., kidneys), bladder, prostate, and colorectum.
[00106] In some embodiments of a method of treating cancer, the cancer is metastatic. In some embodiments of a method of treating cancer, the cancer is a resistant cancer.
Combinations
[00107] Disclosed herein is a method of treating cancer in a subject in need thereof, the method comprising administering to the subject a TRPM8 antagonist in combination with an additional therapeutic agent.
[00108] In some embodiments, the additional therapeutic agent is a targeted agent, a chemotherapy, an immunotherapy (IO), radiation therapy, a gene therapy, or a cell therapy, or any combination thereof. In some embodiments, the additional therapeutic agent is a targeted agent. In some embodiments, the additional therapeutic agent is a chemotherapy. In some embodiments, the additional therapeutic agent is an immunotherapy (IO). In some embodiments, the additional therapeutic agent is radiation therapy. In some embodiments, the additional therapeutic agent is a gene therapy. In some embodiments, the additional therapeutic agent is a cell therapy.
[00109] In some embodiments, the additional therapeutic agent is a targeted agent selected from the group consisting of apalutamide, dabrafenib, enzalutamide, fulvestrant, ibrutinib, lenalidomide, palbociclib, pomalidomide, sorafenib, tamoxifen, and thalidomide.
[00110] In some embodiments, the additional therapeutic agent is dabrafenib.
[00111] In some embodiments, the additional therapeutic agent is enzalutamide.
[00112] In some embodiments, the additional therapeutic agent is ibrutinib.
[00113] In some embodiments, the additional therapeutic agent is lenalidomide.
[00114] In some embodiments, the additional therapeutic agent is sorafenib.
[00115] In some embodiments, the additional therapeutic agent is tamoxifen.
[00116] In some embodiments, the additional therapeutic agent is a chemotherapy agent selected from the group consisting of 5 -fluorouracil, bleomycin, capecitabine, carboplatin, cisplatin, cyclophosphamide, dacarbazine, docetaxel, doxorubicin, epirubicin, etoposide, etoposide, folinic acid, gemcitabine, ifosfamide, methotrexate, mustine, oxaliplatin, prednisolone, procarbazine, vinorelbine, and vinorelbine.
[00117] In some embodiments, the additional therapeutic agent is cisplatin.
[00118] In some embodiments, the additional therapeutic agent is gemcitabine.
[00119] In some embodiments, the additional therapeutic agent is oxaliplatin.
[00120] In some embodiments, the additional therapeutic agent is a CTLA-4 blocker, a PD-1 blocker, or a PD-L1 blocker.
[00121] In some embodiments, the CTLA-4 blocker is ipilimumab or tremelimumab.
[00122] In some embodiments, the PD-1 blocker is nivolumab or pembrolizumab.
[00123] In some embodiments, the PD-L1 blocker is atezolizumab, avelumab, cemiplimab, or durvalumab.
[00124] The terms “combination,” “therapeutic combination” or “pharmaceutical combination” as used herein refer to either a fixed combination in one dosage unit form, or non-fixed combination, or a kit of parts for the combined administration where two or more therapeutic agents may be administered
together, independently at the same time or separately within time intervals, especially where these time intervals allow that the combination partners show a cooperative, e.g., synergistic, effect.
[00125] The term “combination therapy” refers to the administration of two or more therapeutic agents to treat a therapeutic condition or disorder described in the present disclosure. Such administration encompasses co-administration of these therapeutic agents in a substantially simultaneous manner, such as in a single formulation having a fixed ratio of active ingredients or in separate formulations (e.g. , capsules and/or intravenous formulations) for each active ingredient. In addition, such administration also encompasses use of each type of therapeutic agent in a sequential or separate manner, either at approximately the same time or at different times. Regardless of whether the active ingredients are administered as a single formulation or in separate formulations, the drugs are administered to the same patient as part of the same course of therapy. In any case, the treatment regimen will provide beneficial effects in treating the conditions or disorders described herein.
[00126] In some embodiments the combination is for simultaneous, sequential, or separate administration. By simultaneous therapeutic use, within the meaning of the present embodiments is meant an administration of at least two active ingredients by the same route and at the same time or at substantially the same time.
[00127] By separate use, within the meaning of the present embodiments is meant in particular an administration of at least two active ingredients at the same time or at substantially the same time by different routes.
[00128] By sequential therapeutic use is meant administration of at least two active ingredients at different times, the administration route being identical or different. More particularly by an administration method is meant according to which the whole administration of one of the active ingredients is carried out before administration of the other or others commences.
[00129] In some embodiments, the combination is a fixed combination. In some embodiments, the combination is a non-fixed combination. The terms “fixed combination,” “fixed dose” and “single formulation” as used herein refers to a single carrier or vehicle or dosage form formulated to deliver an amount, which is jointly therapeutically effective for the treatment of cancer, of both therapeutic agents to a patient. The single vehicle is designed to deliver an amount of each of the agents along with any pharmaceutically acceptable carriers or excipients. In some embodiments, the vehicle is a tablet, capsule, pill, or a patch. In other embodiments, the vehicle is a solution or a suspension.
[00130] The term “non-fixed combination” or “kit of parts” means that the therapeutic agents of the combinations disclosed herein are both administered to a patient as separate entities either simultaneously, concurrently, or sequentially with no specific time limits, wherein such administration provides therapeutically effective levels of the two compounds in the body of a subject in need thereof. The latter also applies to cocktail therapy, e.g., the administration of three or more active ingredients.
Examples
[00131] The present invention is explained in more detail in the following Examples, which are not to be construed as limitative.
Example 1: Cell proliferation assay:
[00132] 15 mL of cell culture medium was placed into a T75 flask (Coming, 430639), and the flask was placed in a humidified 37°C/5% CO2 incubator (ThermoFisher, 371) for 15 minutes to allow medium to equilibrate to the proper pH and temperature.
[00133] Frozen vials of cell stocks were removed from liquid nitrogen and thawed rapidly by placing at 37°C in a water bath with gentle agitation for 1-2 minutes and then decontaminated by wiping with 70% ethanol before opening in a Class II biological safety cabinet. The vial contents were transferred dropwise into 10 mL of cell culture medium in a sterile 15mL conical tube (BD-Falcon, 352097). The cells were centrifuged at 200 x g for 5 minutes (Eppendorf, 5810R). The supernatant was aspirated, and the cell pellets were re-suspended in 1 mL of fresh cell culture medium and then transferred into a T75 flask containing cell culture medium. The cells were maintained with the addition of fresh medium or replacement of medium. The medium was renewed every 2-3 days. The cells were harvested from flask and then counted. The cells were diluted with culture medium to designed densities and inoculated 135 pL of cell suspension into each well of 96-well cell culture plate (Coming, 3599).
[00134] Compound 1 besylate (10 pM and 30 pM), dabrafenib (1 pM), cisplatin (5 pM), gemcitabine (0.002 pM), lenalidomide (5 pM), enzalutamide (1 pM), sorafenib (6 pM), oxaliplatin (2 pM), tamoxifen (5 pM), carboplatin (2 pM), ibrutinib (5 pM), were prepared. The compound intermediate plate (10X) were prepared. Compound was diluted using culture medium. 15 pl of the compound-medium was added in each well from the 10X compound intermediate plate into the cells in 96-well assay plate according to the plate map. The final DMSO concentration was 0.1%. Compound 1 was tested in triplicate. The plate was incubated at 37°C for 5d, without changing medium during treatment.
[00135] After day 0, day 1, day 2, day 3, day 4, day 5, the plate was removed from incubators and equilibrated at room temperature for 15 minutes. The CellTiter Gio reagents (Promega, G7571) was equilibrated to room temperature before the experiment. 50 pL medium was slowly removed from each well using a multichannel pipet and not touching the tip at the bottom of each well. 100 pL of CellTiter- Glo reagent was added into each well to be detected (at 1: 1 to culture medium). The plates were kept at room temperature for 30 min followed by reading on EnVision (Perkin Elmer). The cell viability was calculated following the formula below:
Y=(LumTest- Lumslank)/ (LumDMSO- Lumsiank)
Blank: culture medium
Example 2: Cell proliferation assay with KMS-26 cell line (multiple myeloma)
[00136] KMS-26 cell line was used in the proliferation assay as described in Example 1, using Compound 1 (10 pM and 30 pM), and lenalidomide (5 pM) (FIG. 1).
Example 3: Cell proliferation assay with JJN-3 cell line (multiple myeloma)
[00137] JJN-3 cell line was used in the proliferation assay as described in Example 1, using Compound 1 (10 pM and 30 pM), and lenalidomide (5 pM) (FIG. 2).
Example 4: Cell proliferation assay with OPM-2 cell line (multiple myeloma)
[00138] OPM-2 cell line was used in the proliferation assay as described in Example 1, using Compound 1 (10 pM and 30 pM), and lenalidomide (5 pM) (FIG. 3).
Example 5: Cell proliferation assay with MCF7 cell line (breast cancer)
[00139] MCF7 cell line was used in the proliferation assay as described in Example 1, using Compound 1 (10 pM and 30 pM), and tamoxifen (5 pM) (FIG. 4).
Example 6: Cell proliferation assay with MDA-MB-231 cell line (TNB breast cancer)
[00140] MDA-MB-231 cell line was used in the proliferation assay as described in Example 1, using Compound 1 (10 pM and 30 pM), and gemcitabine (0.002 pM) (FIG. 5).
Example 7: Cell proliferation assay with HMC-1-8 cell line (breast cancer)
[00141] HMC-1-8 cell line was used in the proliferation assay as described in Example 1, using Compound 1 (10 pM and 30 pM), and sorafenib (6 pM) (FIG. 6).
Example 8: Cell proliferation assay with SK-MEL-28 cell line (melanoma)
[00142] SK-MEL-28 cell line was used in the proliferation assay as described in Example 1, using Compound 1 (10 pM and 30 pM), and dabrafenib (1 pM) (FIG. 7).
Example 9: Cell proliferation assay with A101D cell line (melanoma)
[00143] A101D cell line was used in the proliferation assay as described in Example 1, using Compound
1 (10 pM and 30 pM), and dabrafenib (1 pM) (FIG. 8).
Example 10: Cell proliferation assay with Hs 294T cell line (melanoma)
[00144] Hs 294T cell line was used in the proliferation assay as described in Example 1, using Compound 1 (10 pM and 30 pM), and dabrafenib (1 pM) (FIG. 9).
Example 11: Cell proliferation assay with MDA-MB-435S cell line (melanoma)
[00145] MDA-MB-435S cell line was used in the proliferation assay as described in Example 1, using Compound 1 (10 pM and 30 pM), and dabrafenib (1 pM) (FIG. 10).
Example 12: Cell proliferation assay with HOS cell line (osteosarcoma)
[00146] HOS cell line was used in the proliferation assay as described in Example 1, using Compound 1 (10 pM and 30 pM), and cisplatin (5 pM) (FIG. 11).
Example 13: Cell proliferation assay with Saos-2 cell line (osteosarcoma)
[00147] Saos-2 cell line was used in the proliferation assay as described in Example 1, using Compound 1 (10 pM and 30 pM), and cisplatin (5 pM) (FIG. 12).
Example 14: Cell proliferation assay with NCI-H2081 cell line (SCLC lung cancer)
[00148] NCI-H208 cell line was used in the proliferation assay as described in Example 1, using Compound 1 (10 pM and 30 pM), and cisplatin (5 pM) (FIG. 13).
Example 15: Cell proliferation assay with ACHN cell line (kidney cancer)
[00149] ACHN cell line was used in the proliferation assay as described in Example 1, using Compound 1 (10 pM and 30 pM), and sorafenib (6 pM) (FIG. 14).
Example 16: Cell proliferation assay with G-402 cell line (kidney cancer)
[00150] G-402 cell line was used in the proliferation assay as described in Example 1, using Compound
1 (10 pM and 30 pM), and sorafenib (6 pM) (FIG. 15).
Example 17: Cell proliferation assay with T24 cell line (bladder cancer)
[00151] T24 cell line was used in the proliferation assay as described in Example 1, using Compound 1
(10 pM and 30 pM), and gemcitabine (0.002 pM) (FIG. 16).
Example 18: Cell proliferation assay with OAW28 cell line (ovarian cancer)
[00152] OAW28 cell line was used in the proliferation assay as described in Example 1, using
Compound 1 (10 pM and 30 pM), and carboplatin (2 pM) (FIG. 17).
Example 19: Cell proliferation assay with LNCaP cell line (prostate cancer)
[00153] LNCaP cell line was used in the proliferation assay as described in Example 1, using Compound 1 (10 pM and 30 pM), and enzalutamide (1 pM) (FIG. 18).
Example 20: Cell proliferation assay with Caco-2 cell line (colorectal cancer)
[00154] Caco-2 cell line was used in the proliferation assay as described in Example 1, using Compound
1 (10 pM and 30 pM), and oxaliplatin (2 pM) (FIG. 19).
Example 21: Cell proliferation assay with SU-DHL-1 cell line (lymphoma)
[00155] SU-DHL-1 cell line was used in the proliferation assay as described in Example 1, using Compound 1 (10 pM and 30 pM), and ibrutinib (5 pM) (FIG. 20).
Claims
What is claimed is:
2. The method of claim 1, wherein Compound 1 is in the form of a salt.
3. The method of claim 1, wherein Compound 1 is in the form of a besylate salt.
4. The method of any one of claims 1-3, wherein the cancer is bladder cancer, breast cancer, cholangiocarcinoma, colorectal cancer, kidney cancer, liver cancer, lung cancer, lymphoma, myeloma, osteosarcoma, ovarian cancer, pancreatic cancer, prostate cancer, or skin cancer.
5. The method of claim 4, wherein the breast cancer is ER positive breast cancer, ER positive metastatic breast cancer, or triple negative breast cancer (TNB).
6. The method of claim 4, wherein the myeloma is multiple myeloma (MM).
7. The method of claim 4, wherein the skin cancer is melanoma.
8. The method of claim 4, wherein the lung cancer is small cell lung cancer (SCLC) or non-small cell lung cancer (NSCLC).
9. The method of any one of claims 1-8 further comprising administering an additional therapeutic agent.
10. The method of claim 9, wherein the additional therapeutic agent is a targeted agent, a chemotherapy, an immunotherapy (IO), radiation therapy, a gene therapy, or a cell therapy, or any combination thereof.
11. The method of claim 9, wherein the additional therapeutic agent is a targeted agent selected from the group consisting of apalutamide, dabrafenib, enzalutamide, fulvestrant, ibrutinib, lenalidomide, palbociclib, pomalidomide, sorafenib, tamoxifen, and thalidomide.
12. The method of claim 9, wherein the additional therapeutic agent is a chemotherapy agent selected from the group consisting of 5 -fluorouracil, bleomycin, capecitabine, carboplatin, cisplatin, cyclophosphamide, dacarbazine, docetaxel, doxorubicin, epirubicin, etoposide, etoposide, folinic acid, gemcitabine, ifosfamide, methotrexate, mustine, oxaliplatin, prednisolone, procarbazine, vinorelbine, and vinorelbine.
13. The method of claim 9, wherein the additional therapeutic agent is a CTLA-4 blocker, a PD-1 blocker, or a PD-L1 blocker.
14. The method of claim 13, wherein the CTLA-4 blocker is ipilimumab or tremelimumab.
15. The method of claim 13, wherein the PD-1 blocker is nivolumab or pembrolizumab.
16. The method of claim 13, wherein the PD-L1 blocker is atezolizumab, avelumab, cemiplimab, or durvalumab.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US202363583688P | 2023-09-19 | 2023-09-19 | |
| US63/583,688 | 2023-09-19 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2025064492A1 true WO2025064492A1 (en) | 2025-03-27 |
Family
ID=95072066
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2024/047191 Pending WO2025064492A1 (en) | 2023-09-19 | 2024-09-18 | Trpm8 antagonist for treating cancer |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2025064492A1 (en) |
Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2015136947A1 (en) * | 2014-03-14 | 2015-09-17 | Raqualia Pharma Inc. | Azaspiro derivatives as trpm8 antagonists |
-
2024
- 2024-09-18 WO PCT/US2024/047191 patent/WO2025064492A1/en active Pending
Patent Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2015136947A1 (en) * | 2014-03-14 | 2015-09-17 | Raqualia Pharma Inc. | Azaspiro derivatives as trpm8 antagonists |
Non-Patent Citations (3)
| Title |
|---|
| LIU TAO, FANG ZHIHAI, WANG GANG, SHI MINGJUN, WANG XIAO, JIANG KUN, YANG ZHONGHUA, CAO RUI, TAO HUANGHENG, WANG XINGHUAN, ZHOU JIA: "Anti-tumor activity of the TRPM8 inhibitor BCTC in prostate cancer DU145 cells", ONCOLOGY LETTERS, SPANDIDOS PUBLICATIONS, GR, vol. 11, no. 1, 1 January 2016 (2016-01-01), GR , pages 182 - 188, XP093295697, ISSN: 1792-1074, DOI: 10.3892/ol.2015.3854 * |
| OKAMOTO Y ET AL.: "Blockade of TRPM8 activity reduces the invasion potential of oral squamous carcinoma cell lines", INTERNATIONAL JOURNAL OF ONCOLOGY, vol. 40, no. 5, 2012, pages 1431 - 40, XP008160951, DOI: 10.3892/ijo.2012.1340 * |
| VALERO MARÍA LL, MELLO DE QUEIROZ FERNANDA, STÜHMER WALTER, VIANA FÉLIX, PARDO LUIS A.: "TRPM8 Ion Channels Differentially Modulate Proliferation and Cell Cycle Distribution of Normal and Cancer Prostate Cells", PLOS ONE, PUBLIC LIBRARY OF SCIENCE, US, vol. 7, no. 12, 1 December 2012 (2012-12-01), US , pages e51825 - e51825, XP093295699, ISSN: 1932-6203, DOI: 10.1371/journal.pone.0051825 * |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP7503380B2 (en) | Combination medicine comprising an ALK inhibitor and an SHP2 inhibitor | |
| CN114599361B (en) | Pharmaceutical compositions of PRMT5 inhibitors | |
| JP6669744B2 (en) | Substituted pyrrolopyridines as inhibitors of bromodomains | |
| WO2021248095A1 (en) | Heterocyclic compounds and methods of use thereof | |
| CN107108637B (en) | Triazolopyrimidine compound and application thereof | |
| CA2915838C (en) | Bicyclic bromodomain inhibitors | |
| KR101546693B1 (en) | Substituted quinoline compounds and methods of use | |
| EP2291375B1 (en) | Pyrrolopyridinylpyrimidin-2-ylamine derivatives | |
| TW202313588A (en) | Checkpoint kinase 1 (chk1) inhibitors and uses thereof | |
| US20140357636A1 (en) | Treatment of Skeletal-Related Disorders | |
| EP3868761A1 (en) | Imidazolonylchinolines and their use as atm kinase inhibitors | |
| AU2014219256B2 (en) | Heteroaromatic compounds as PI3 kinase modulators | |
| JP2021054843A (en) | Tropomyosin-related kinase (trk) inhibitors | |
| RS61058B1 (en) | Treatment of b-cell malignancies by a combination jak and pi3k inhibitor | |
| BR112015006726B1 (en) | COMPOUND, PHARMACEUTICAL COMPOSITION, AND, USE OF A COMPOUND OR A PHARMACEUTICAL COMPOSITION | |
| EP2635574A1 (en) | 1h-pyrrolo[2,3-b]pyridine derivatives | |
| EP2516431B1 (en) | Pyrrolopyridinyl-pyrimidin-2-ylamine derivatives | |
| EP2516442B1 (en) | Pyrrolo[2,3-d]pyrazin-7-yl pyrimidine compounds | |
| WO2025064492A1 (en) | Trpm8 antagonist for treating cancer | |
| WO2024217563A1 (en) | Lysine acetyltransferase 6a (kat6a) inhibitor, combinations and uses thereof | |
| WO2025007955A1 (en) | Ectonucleotide pyrophosphatase/phosphodiesterase 1 (enpp1) inhibitor combinations and uses thereof | |
| CN105461709B (en) | Substituted urea derivatives and their application in medicine | |
| RU2809631C2 (en) | Pyrazine compounds and their use | |
| BR122021014786B1 (en) | COMPOUND, PHARMACEUTICAL COMPOSITION, AND, USE OF A COMPOUND OR A PHARMACEUTICAL COMPOSITION | |
| BR122021024771B1 (en) | USE OF A COMBINATION OF JAK1/2 AND PI3KSIGMA INHIBITORS IN THE TREATMENT OF MYELOFIBROSIS |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 24869055 Country of ref document: EP Kind code of ref document: A1 |