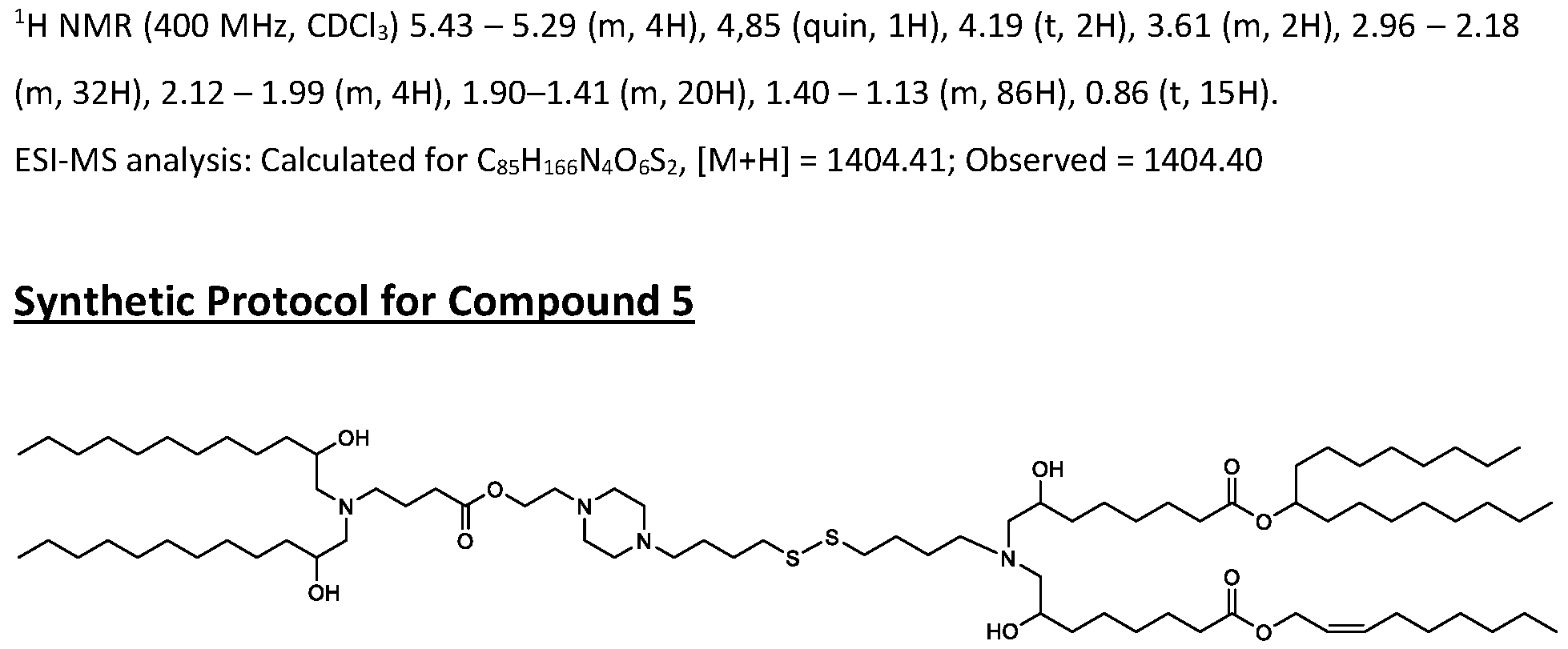
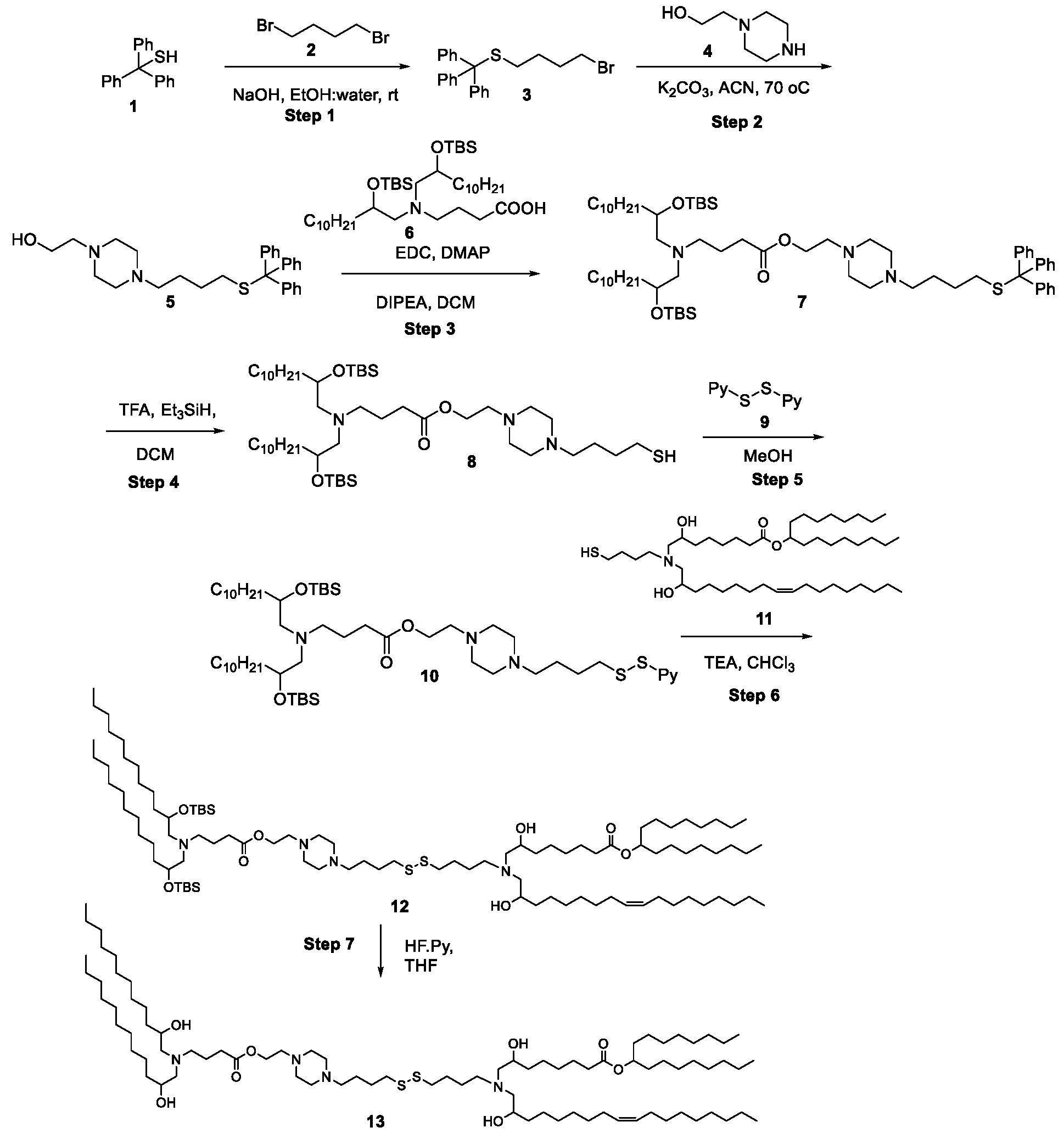
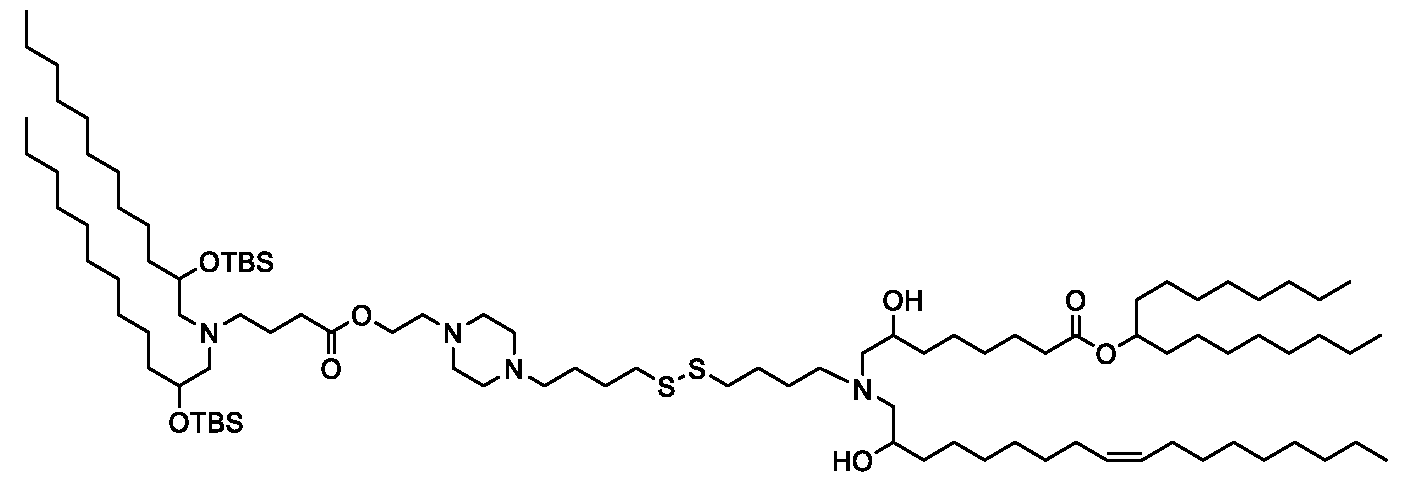
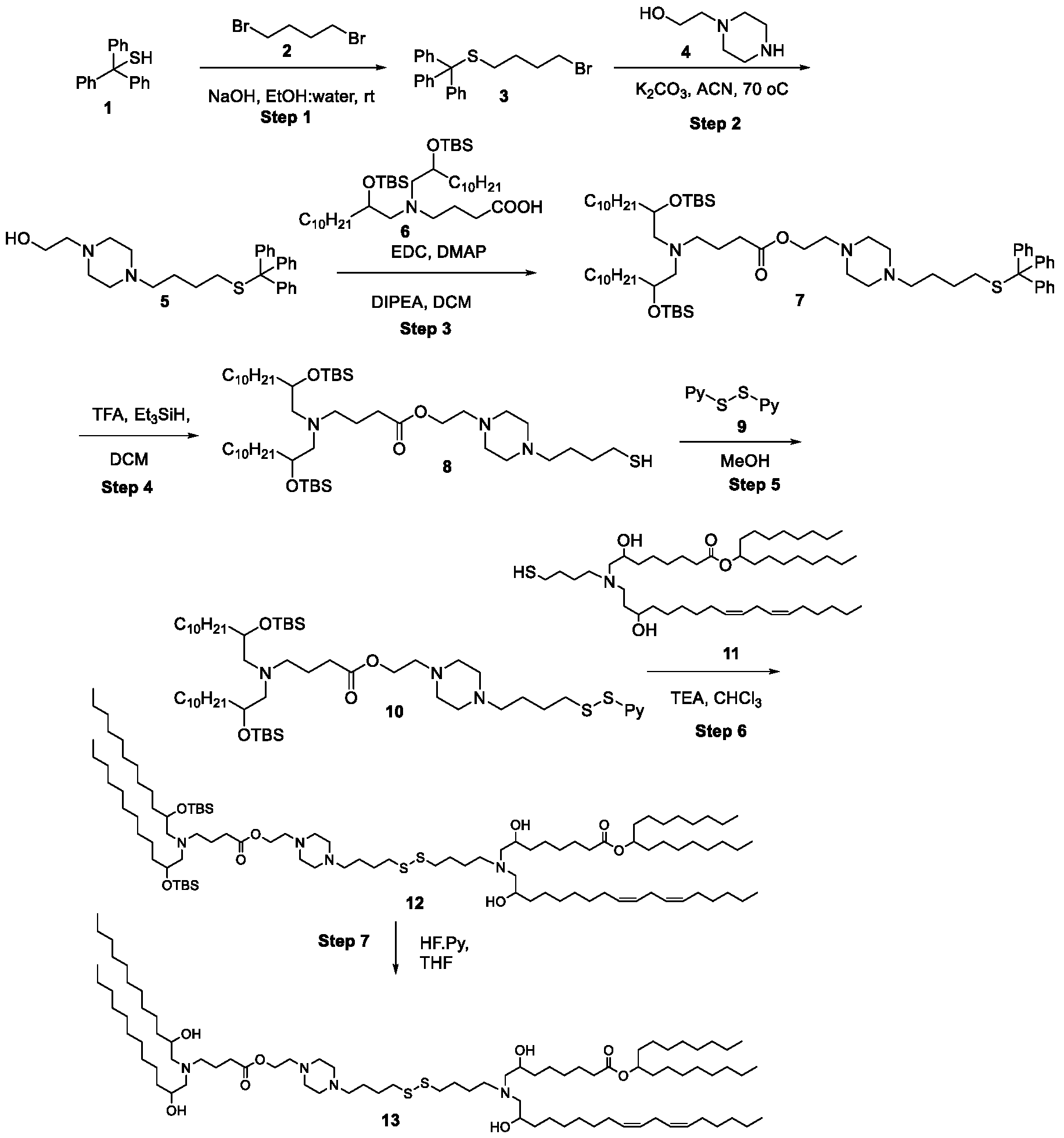
WO2025061967A1 - "good" buffer-based cationic lipids for nucleic acid delivery - Google Patents
"good" buffer-based cationic lipids for nucleic acid delivery Download PDFInfo
- Publication number
- WO2025061967A1 WO2025061967A1 PCT/EP2024/076467 EP2024076467W WO2025061967A1 WO 2025061967 A1 WO2025061967 A1 WO 2025061967A1 EP 2024076467 W EP2024076467 W EP 2024076467W WO 2025061967 A1 WO2025061967 A1 WO 2025061967A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- optionally substituted
- independently selected
- alkyl
- optionally
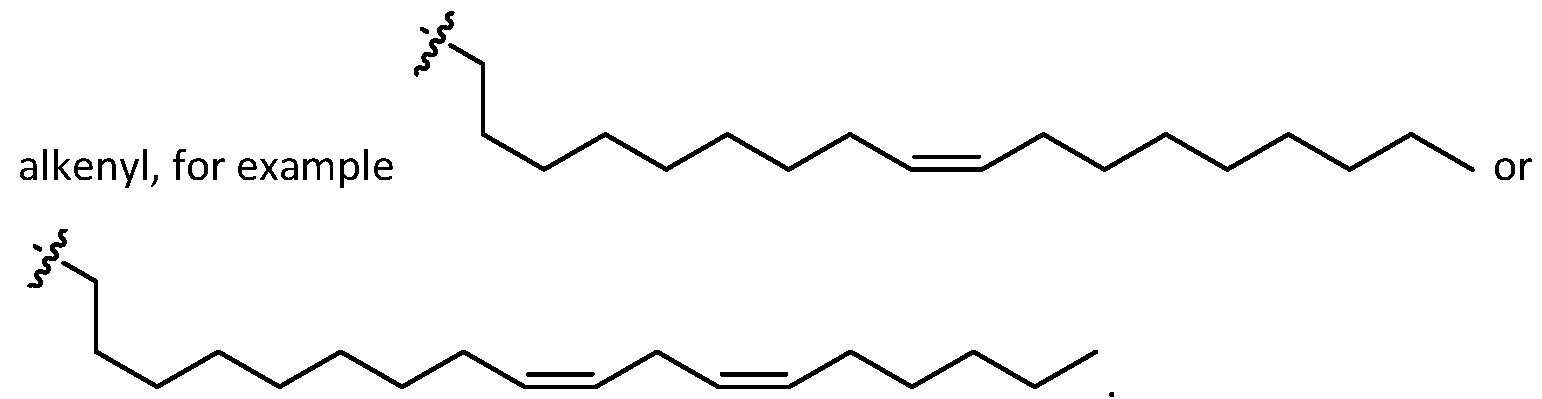
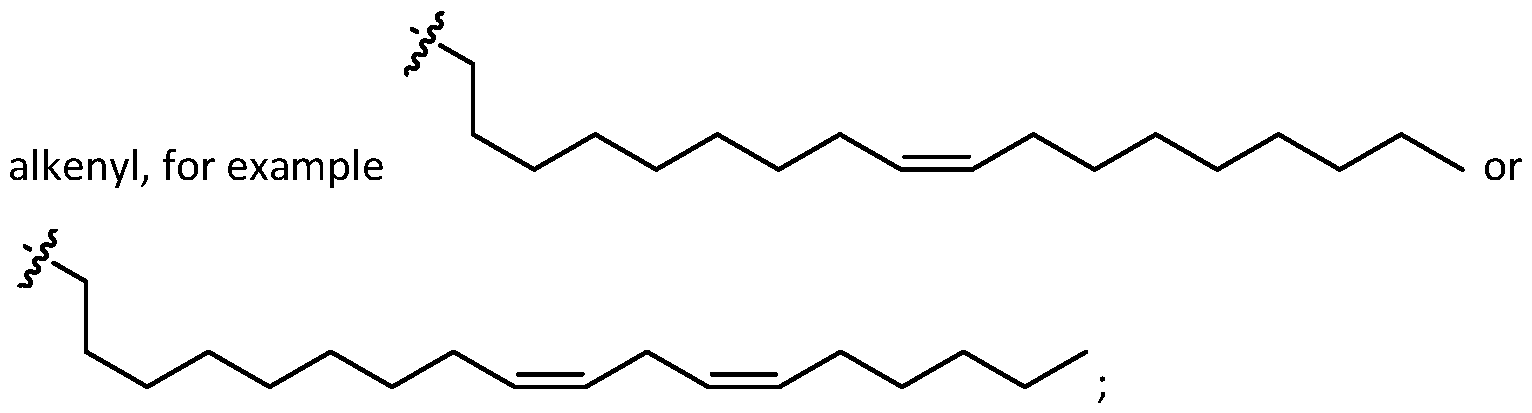
- alkenyl
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D295/00—Heterocyclic compounds containing polymethylene-imine rings with at least five ring members, 3-azabicyclo [3.2.2] nonane, piperazine, morpholine or thiomorpholine rings, having only hydrogen atoms directly attached to the ring carbon atoms
- C07D295/04—Heterocyclic compounds containing polymethylene-imine rings with at least five ring members, 3-azabicyclo [3.2.2] nonane, piperazine, morpholine or thiomorpholine rings, having only hydrogen atoms directly attached to the ring carbon atoms with substituted hydrocarbon radicals attached to ring nitrogen atoms
- C07D295/08—Heterocyclic compounds containing polymethylene-imine rings with at least five ring members, 3-azabicyclo [3.2.2] nonane, piperazine, morpholine or thiomorpholine rings, having only hydrogen atoms directly attached to the ring carbon atoms with substituted hydrocarbon radicals attached to ring nitrogen atoms substituted by singly bound oxygen or sulfur atoms
- C07D295/084—Heterocyclic compounds containing polymethylene-imine rings with at least five ring members, 3-azabicyclo [3.2.2] nonane, piperazine, morpholine or thiomorpholine rings, having only hydrogen atoms directly attached to the ring carbon atoms with substituted hydrocarbon radicals attached to ring nitrogen atoms substituted by singly bound oxygen or sulfur atoms with the ring nitrogen atoms and the oxygen or sulfur atoms attached to the same carbon chain, which is not interrupted by carbocyclic rings
- C07D295/088—Heterocyclic compounds containing polymethylene-imine rings with at least five ring members, 3-azabicyclo [3.2.2] nonane, piperazine, morpholine or thiomorpholine rings, having only hydrogen atoms directly attached to the ring carbon atoms with substituted hydrocarbon radicals attached to ring nitrogen atoms substituted by singly bound oxygen or sulfur atoms with the ring nitrogen atoms and the oxygen or sulfur atoms attached to the same carbon chain, which is not interrupted by carbocyclic rings to an acyclic saturated chain
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/10—Dispersions; Emulsions
- A61K9/127—Synthetic bilayered vehicles, e.g. liposomes or liposomes with cholesterol as the only non-phosphatidyl surfactant
- A61K9/1271—Non-conventional liposomes, e.g. PEGylated liposomes or liposomes coated or grafted with polymers
- A61K9/1272—Non-conventional liposomes, e.g. PEGylated liposomes or liposomes coated or grafted with polymers comprising non-phosphatidyl surfactants as bilayer-forming substances, e.g. cationic lipids or non-phosphatidyl liposomes coated or grafted with polymers
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/48—Preparations in capsules, e.g. of gelatin, of chocolate
- A61K9/50—Microcapsules having a gas, liquid or semi-solid filling; Solid microparticles or pellets surrounded by a distinct coating layer, e.g. coated microspheres, coated drug crystals
- A61K9/51—Nanocapsules; Nanoparticles
- A61K9/5107—Excipients; Inactive ingredients
- A61K9/5123—Organic compounds, e.g. fats, sugars
Definitions
- mRNA messenger RNA
- IM intramuscularly
- the cationic lipid component of a liposome plays an important role in facilitating effective encapsulation of the nucleic acid during the loading of liposomes.
- cationic lipids may play an important role in the efficient release of the nucleic acid cargo from the liposome into the cytoplasm of a target cell.
- Various cationic lipids suitable for in vivo use have been discovered. However, there remains a need to identify cationic lipids that are effective for e.g., intramuscular delivery of mRNA (e.g., in vaccines). There also remains a need to identify cationic lipids that can be synthesized efficiently and cheaply without the formation of potentially toxic by-products.
- Additional selection criteria included high solubility, lack of toxicity, limited interference with biochemical reactions, very low absorbance between 240 nm and 700 nm, enzymatic and hydrolytic stability, minimal changes due to temperature and concentration, limited effects due to ionic or salt composition of the solution, limited interaction with mineral cations, and limited permeability of biological membranes.
- the present invention provides, among other things, a novel class of cationic lipid compounds for in vivo delivery of therapeutic agents, such as nucleic acids.
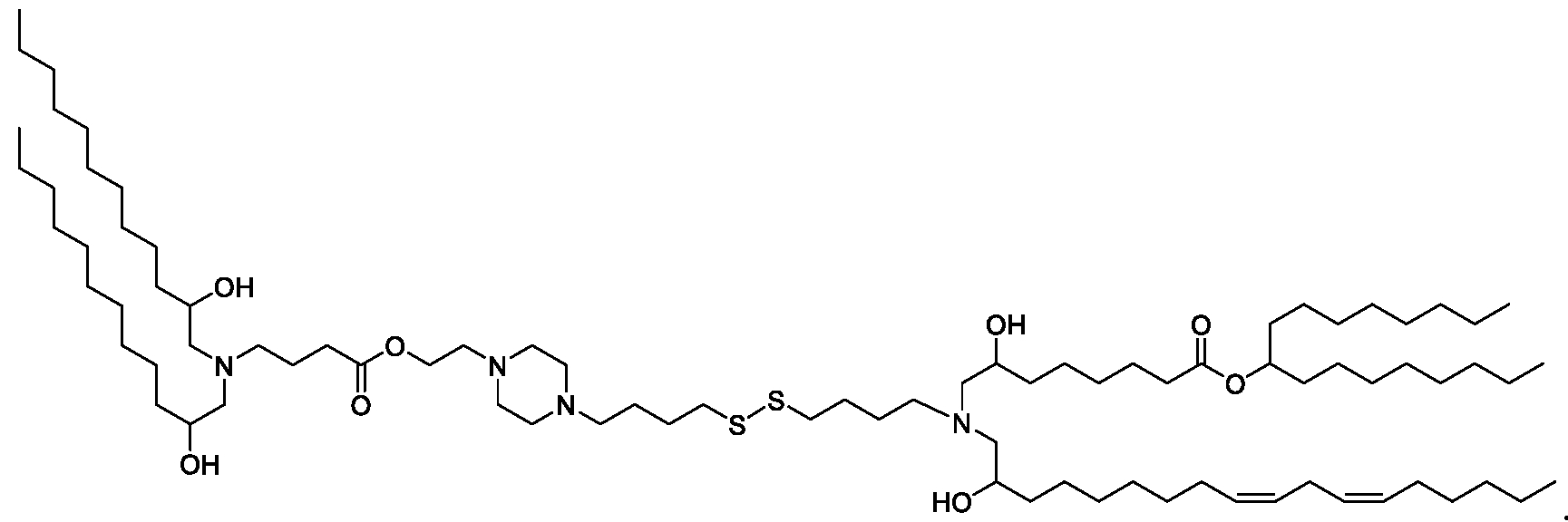
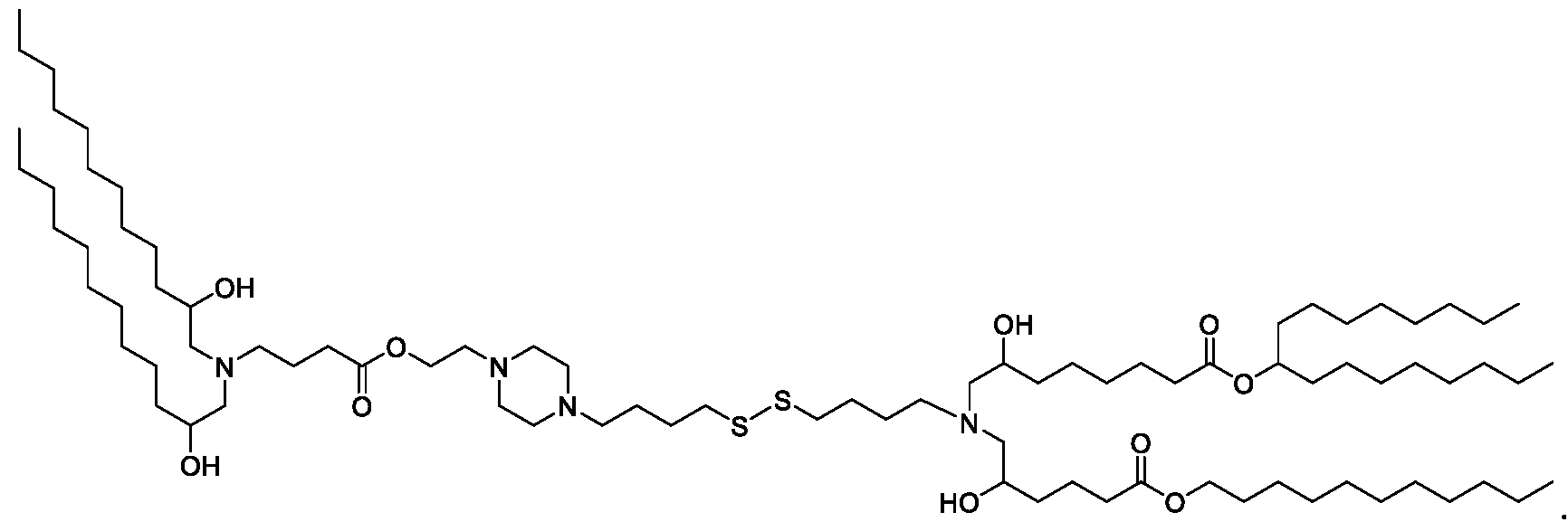
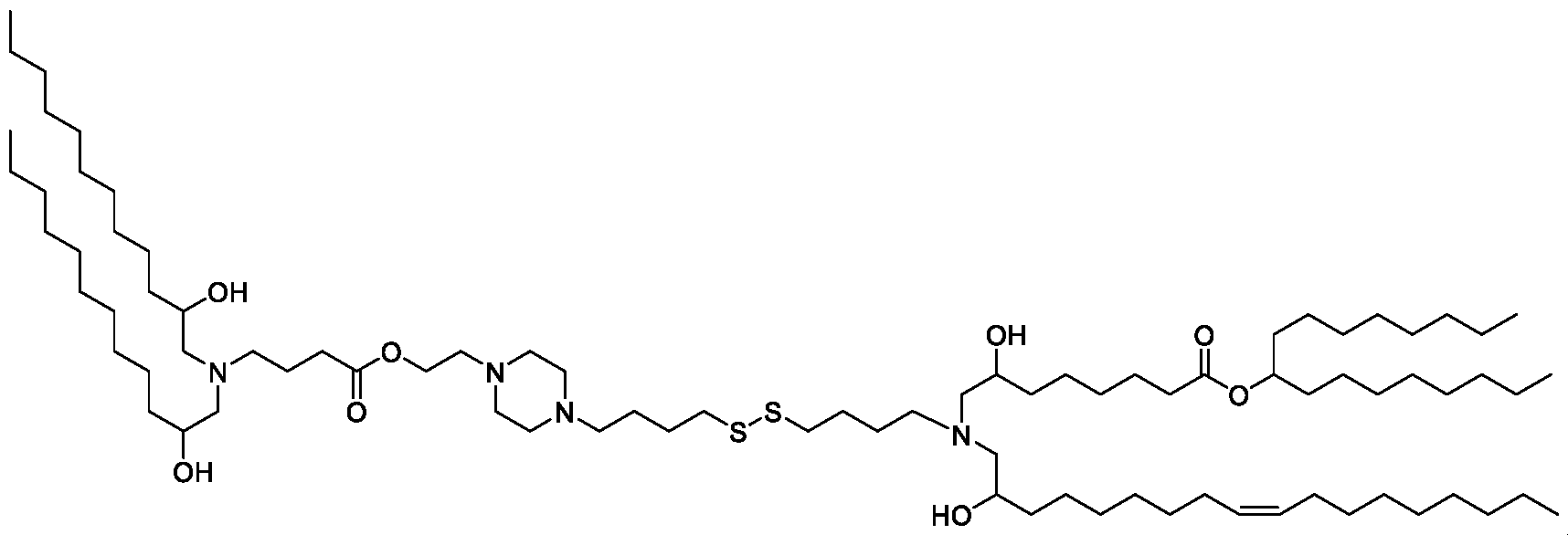
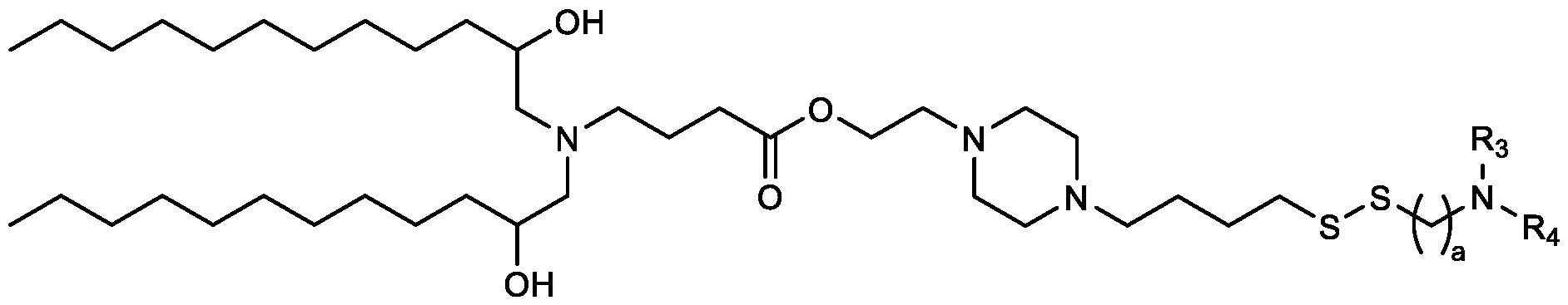
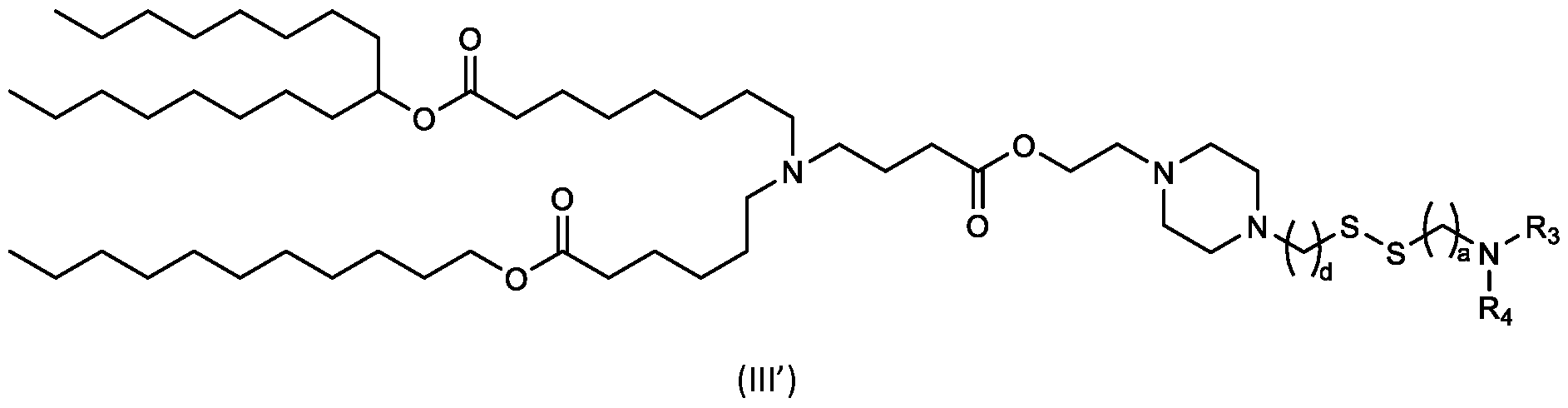
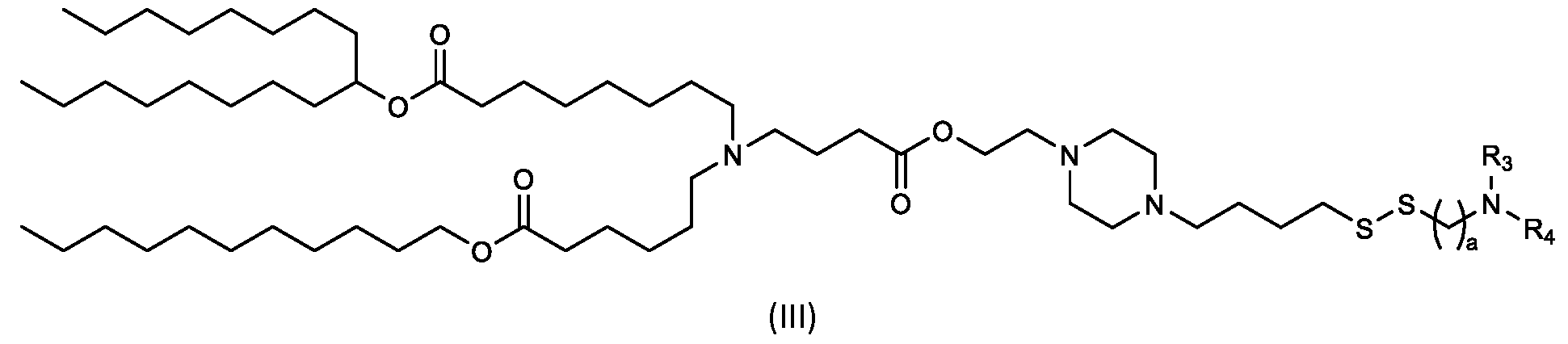
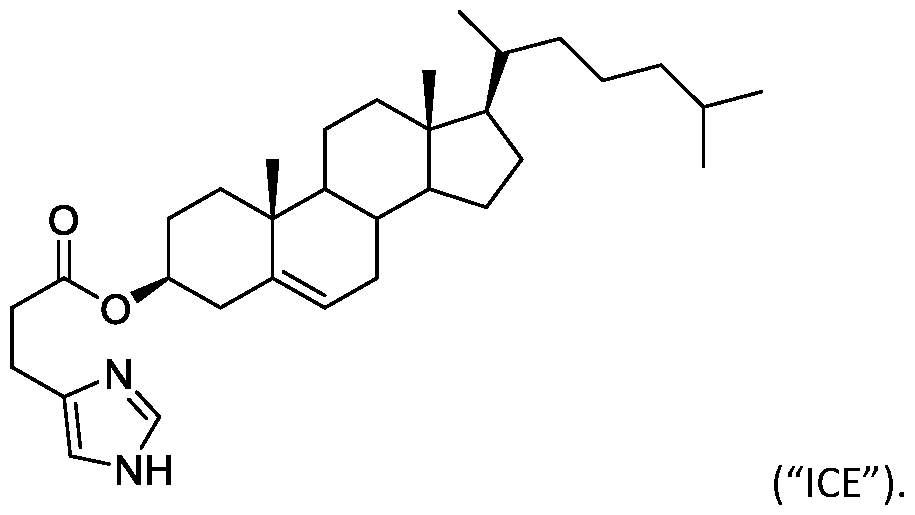
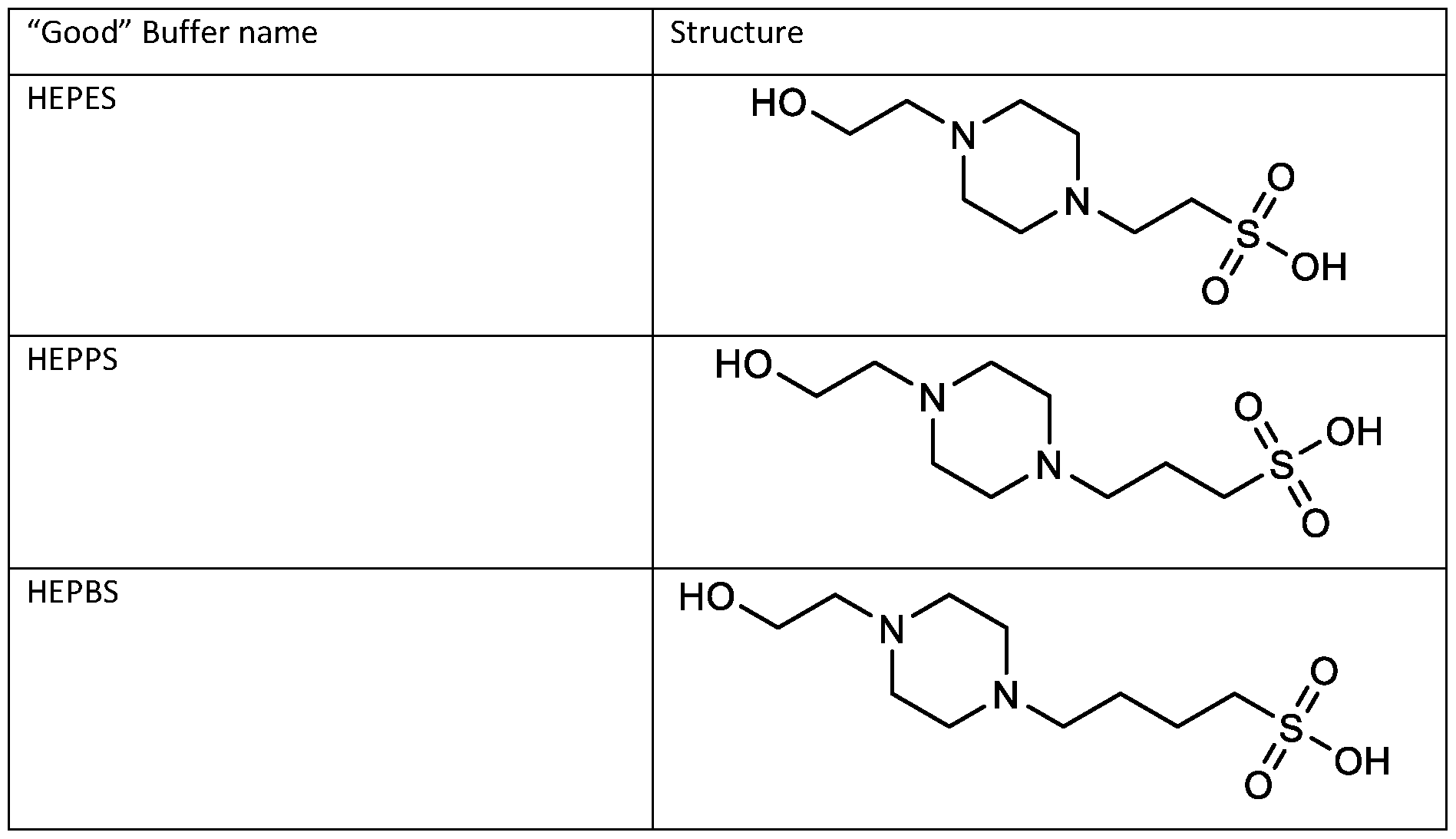
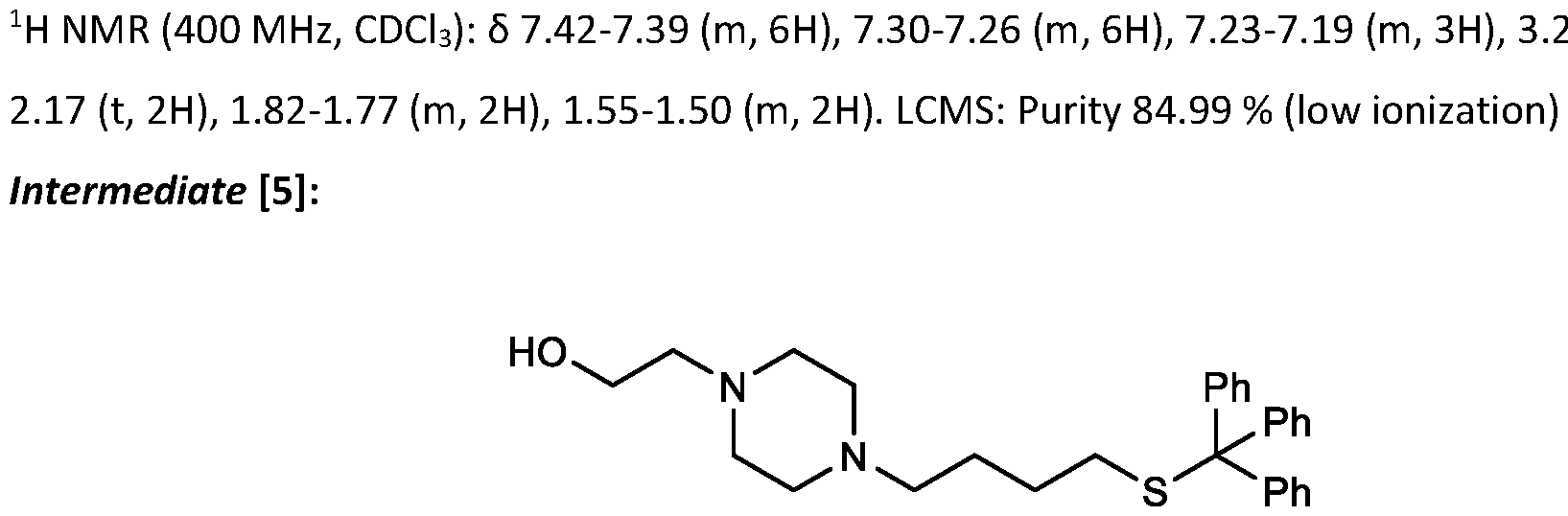
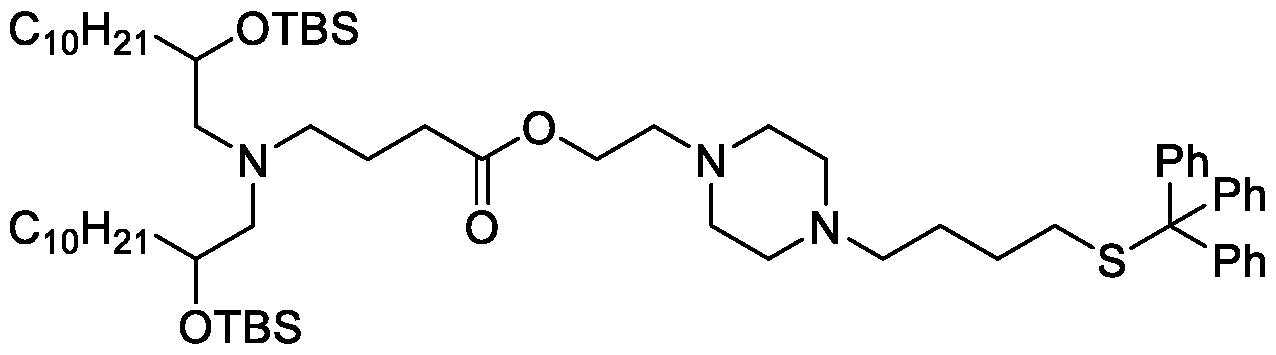
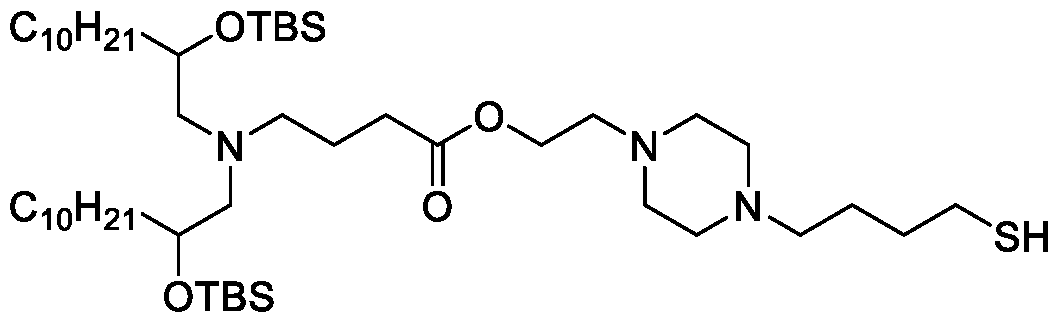
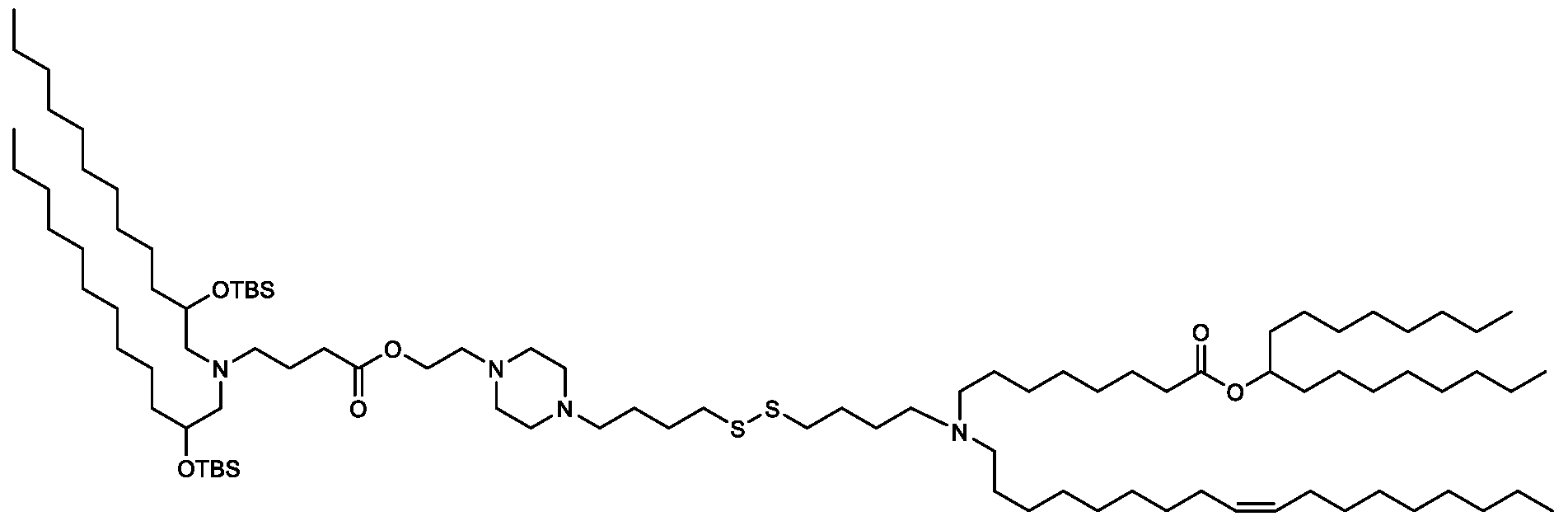
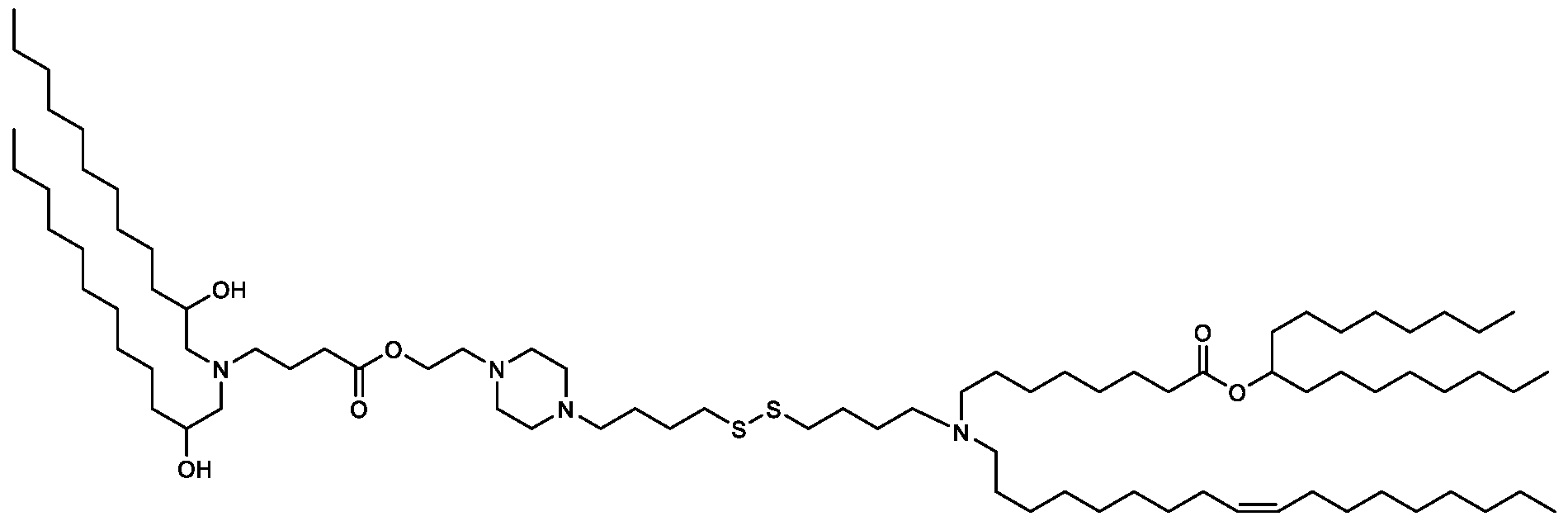
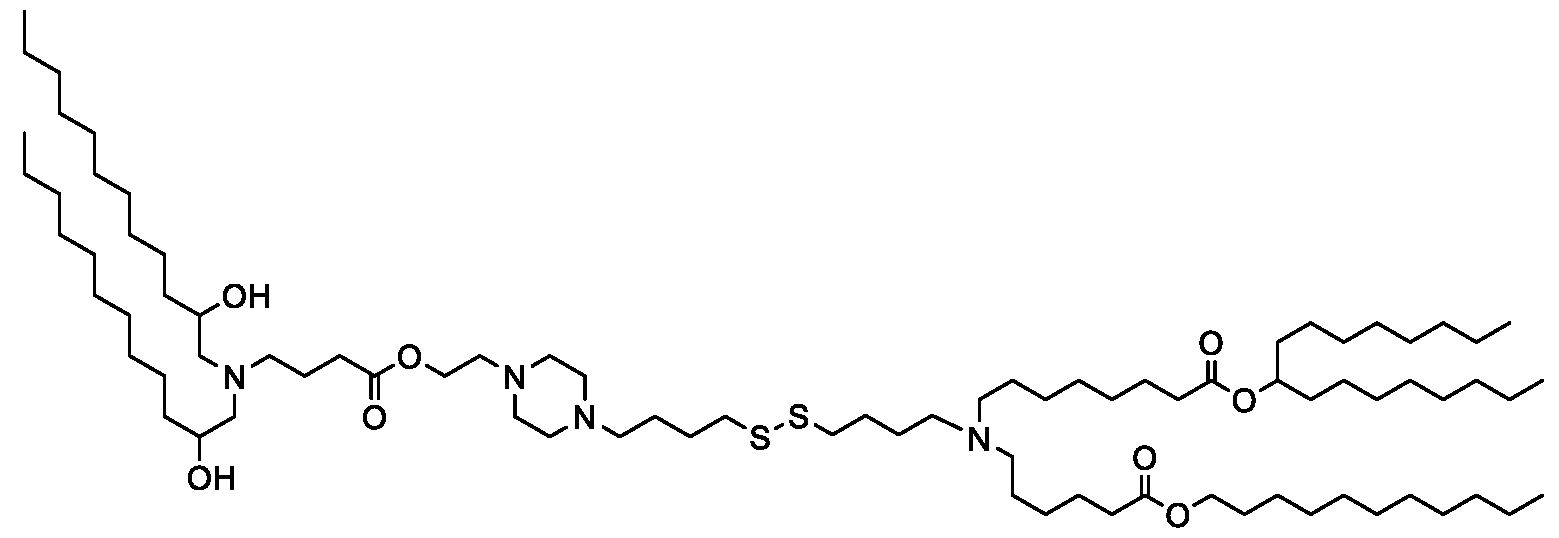
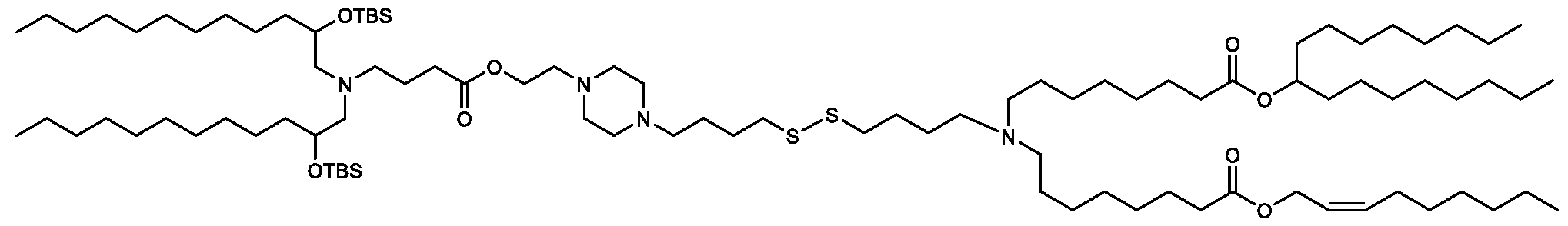
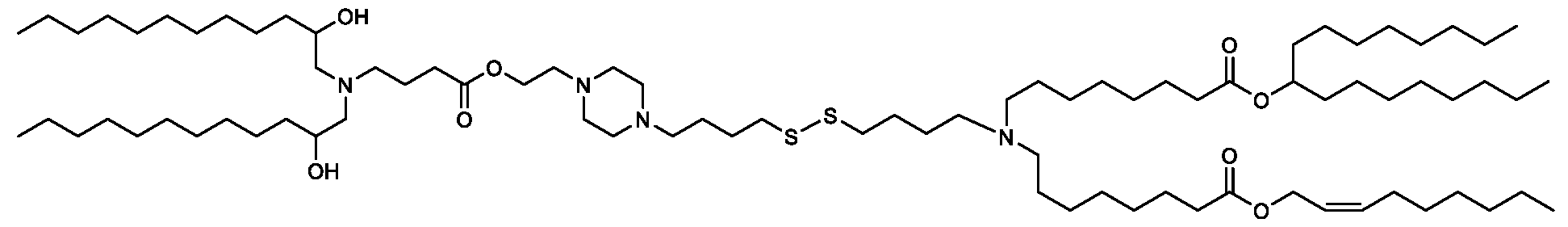
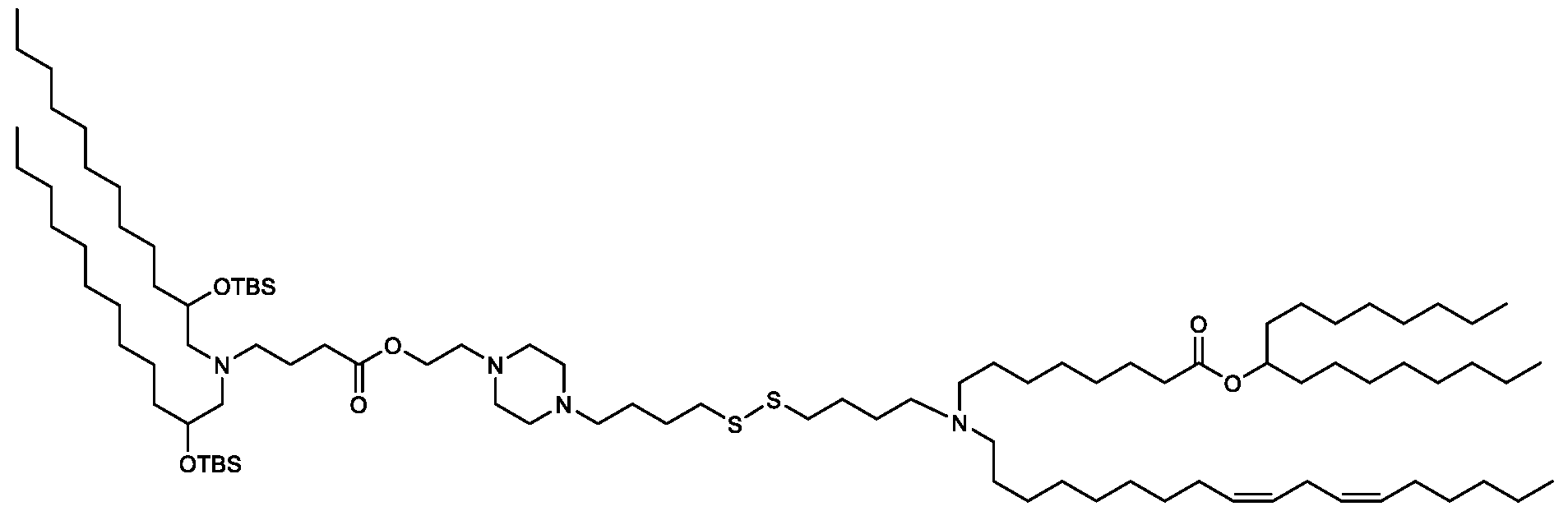
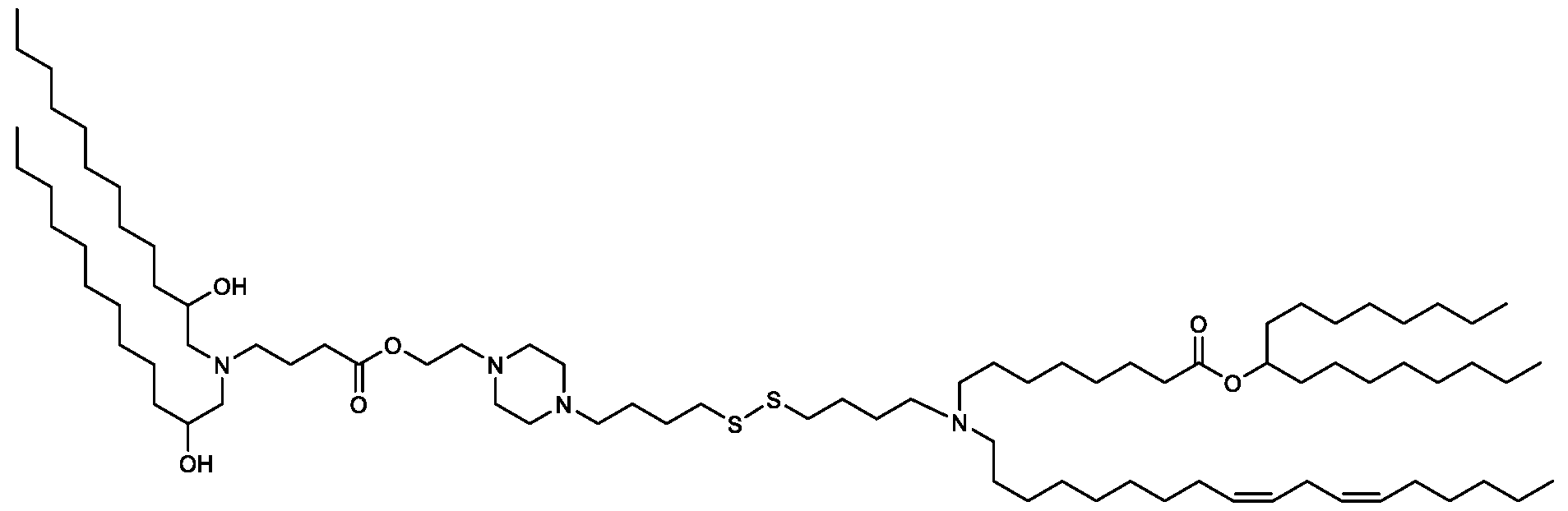
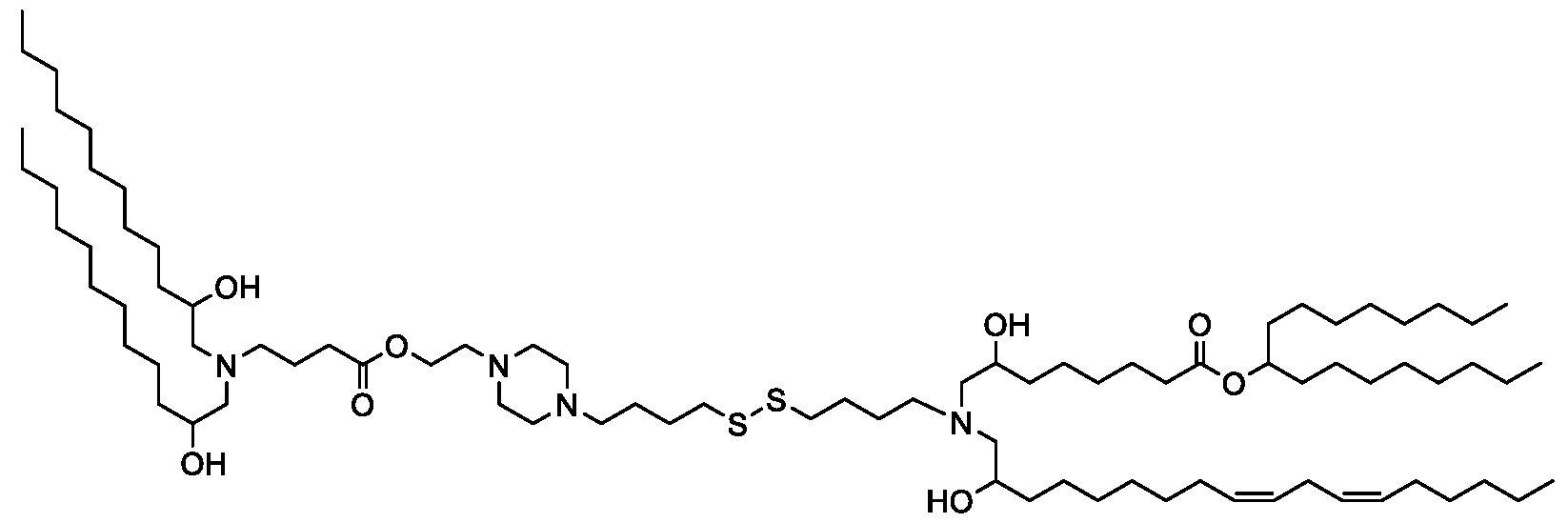
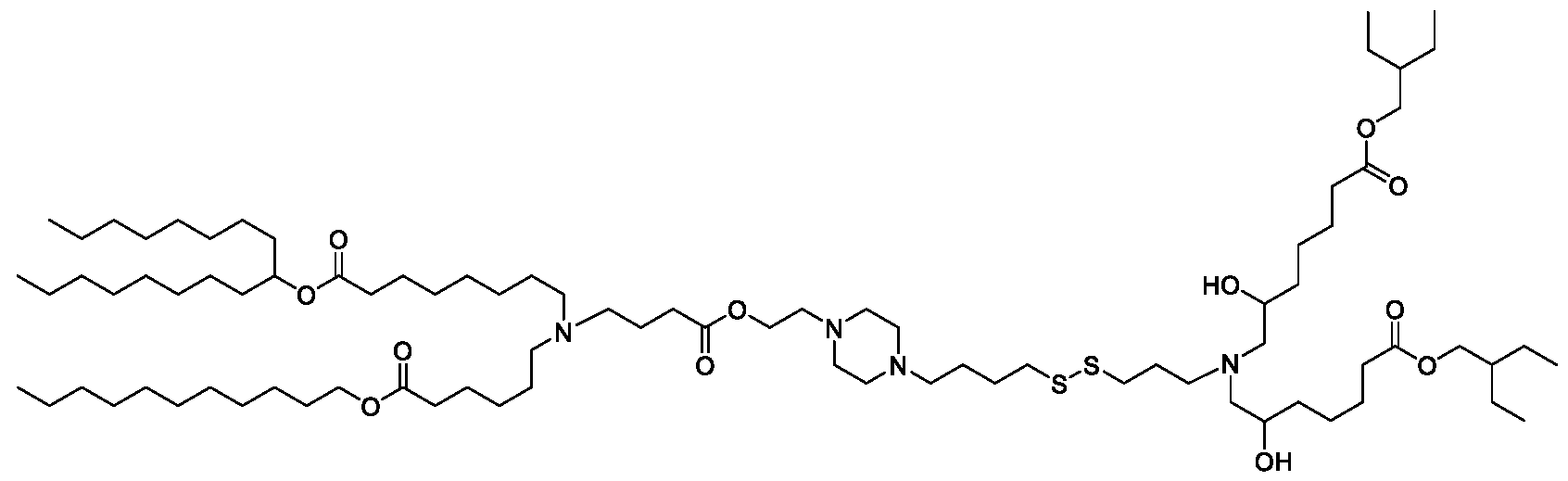
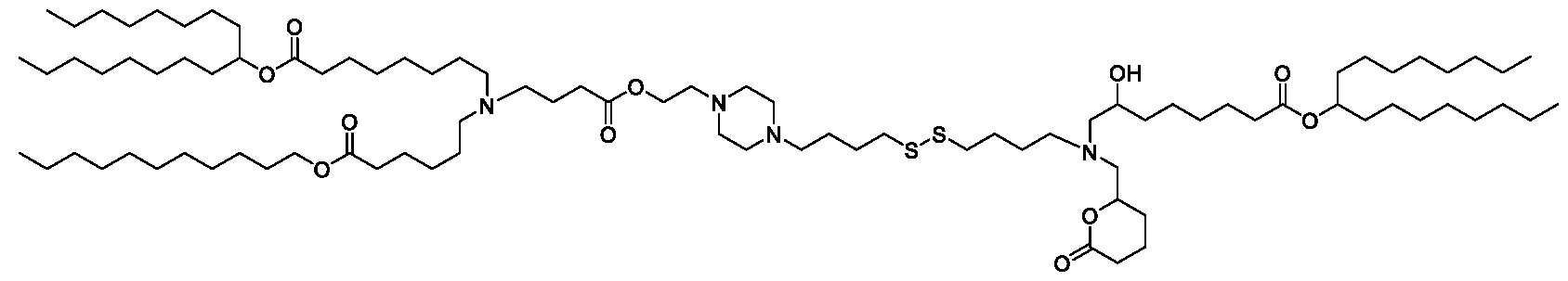
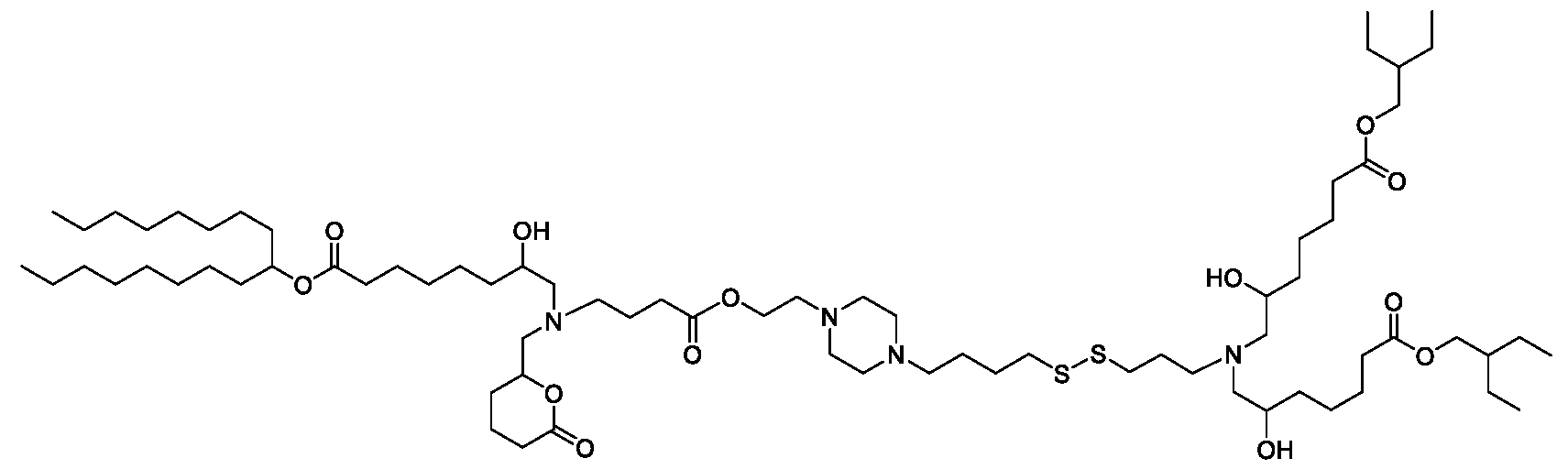
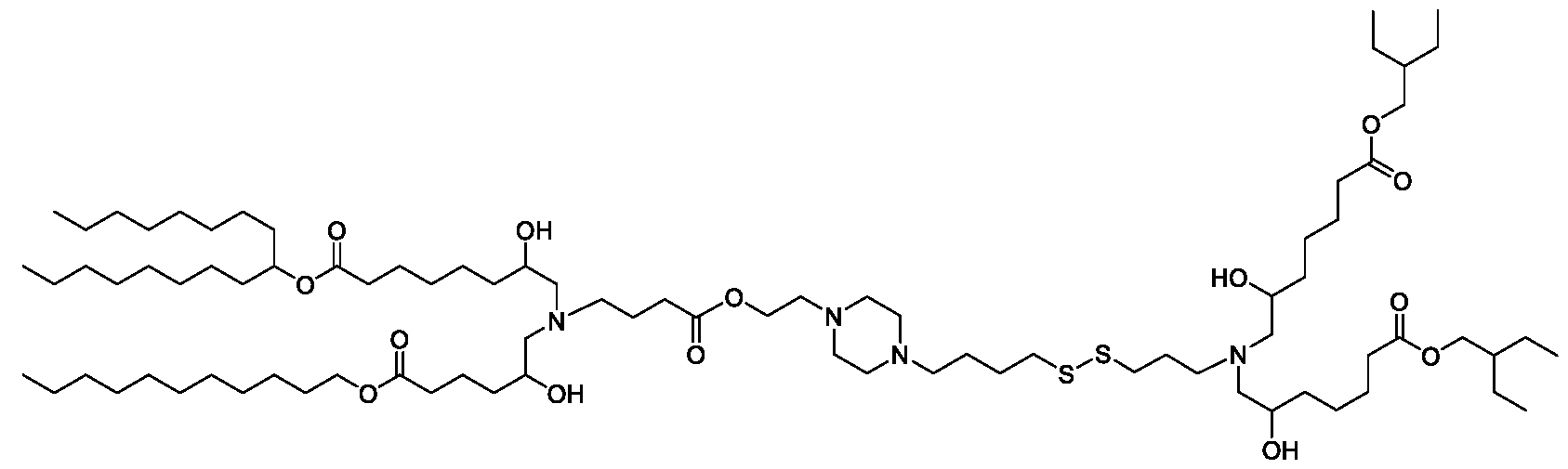
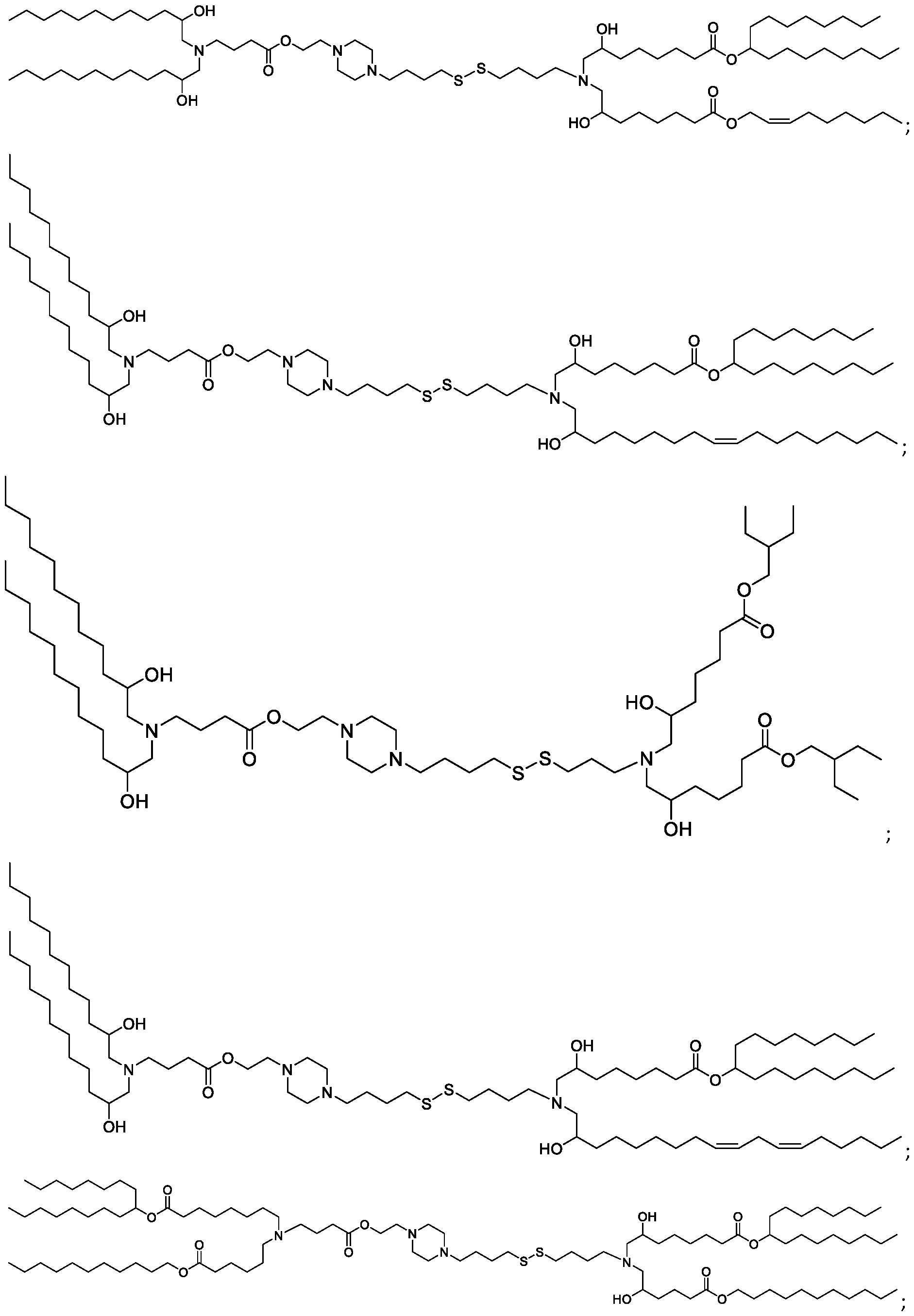
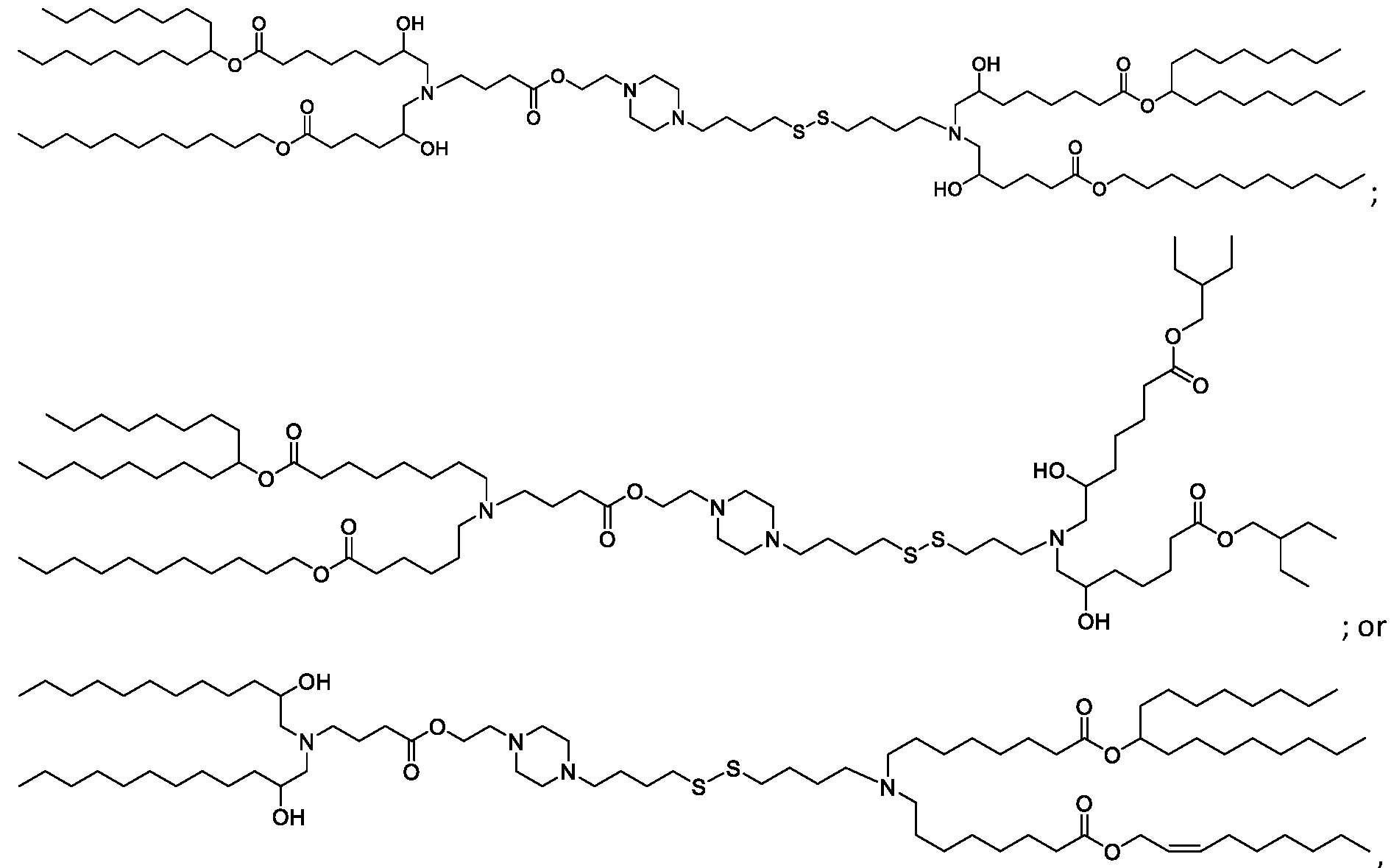
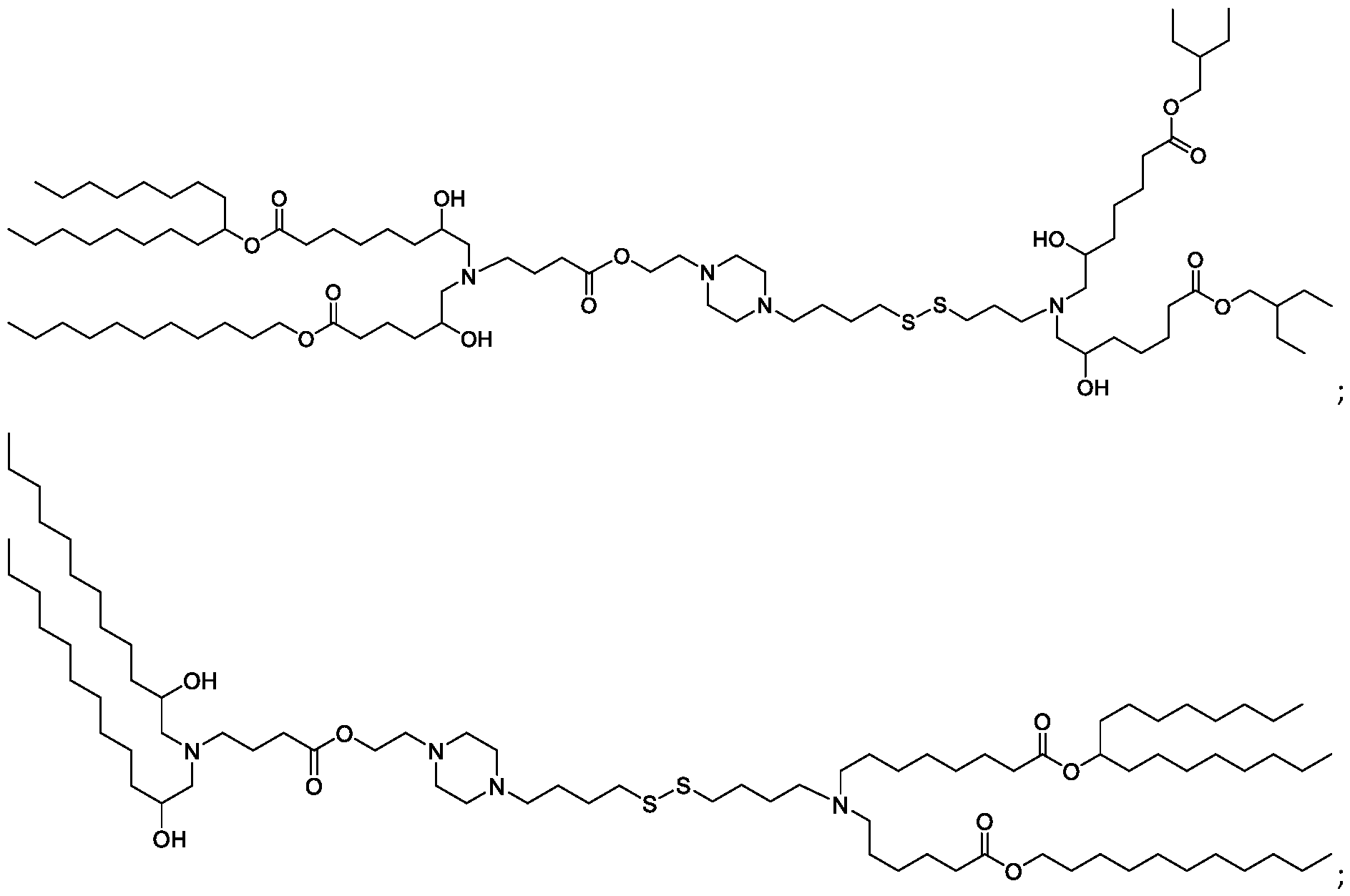
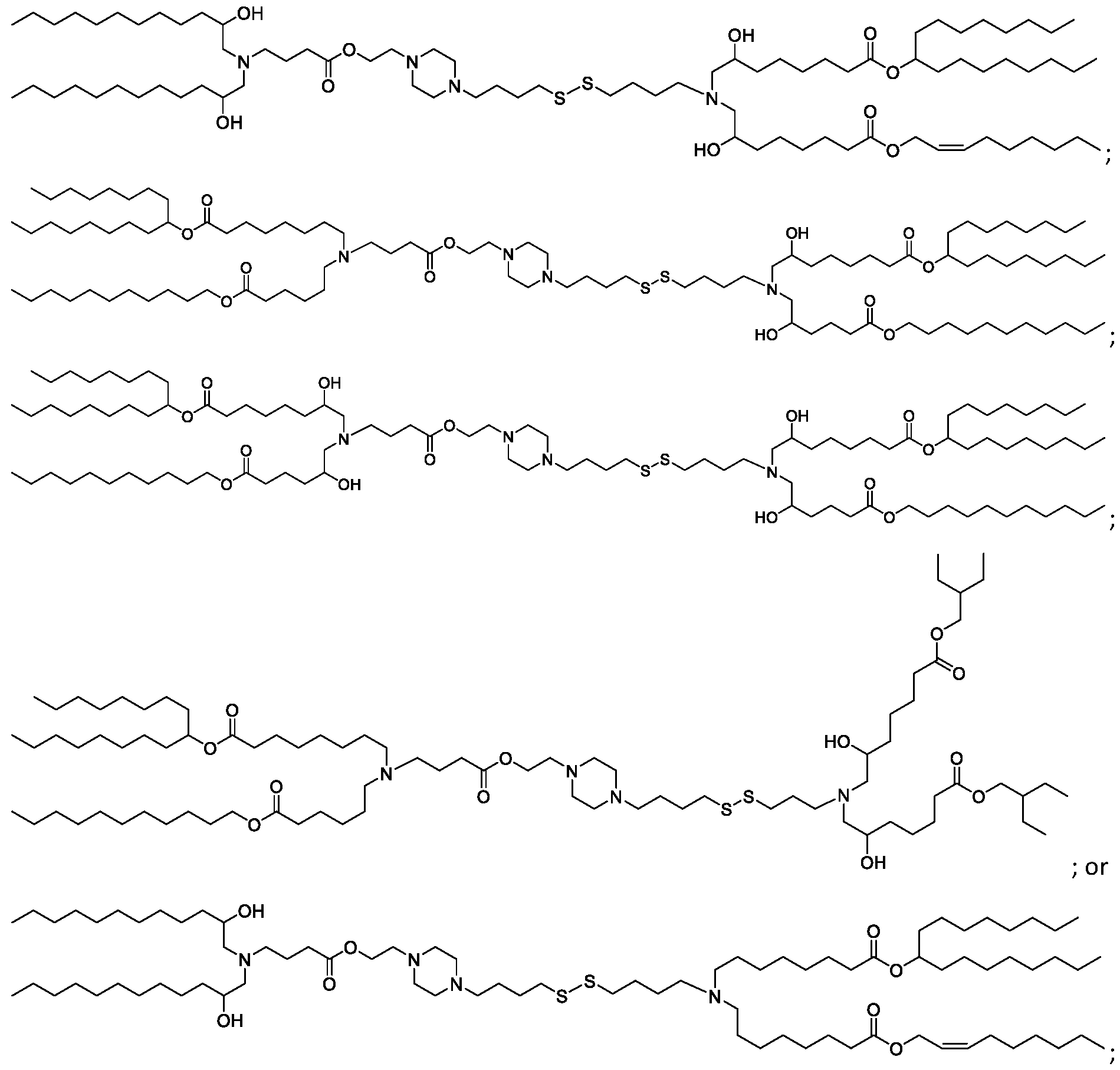
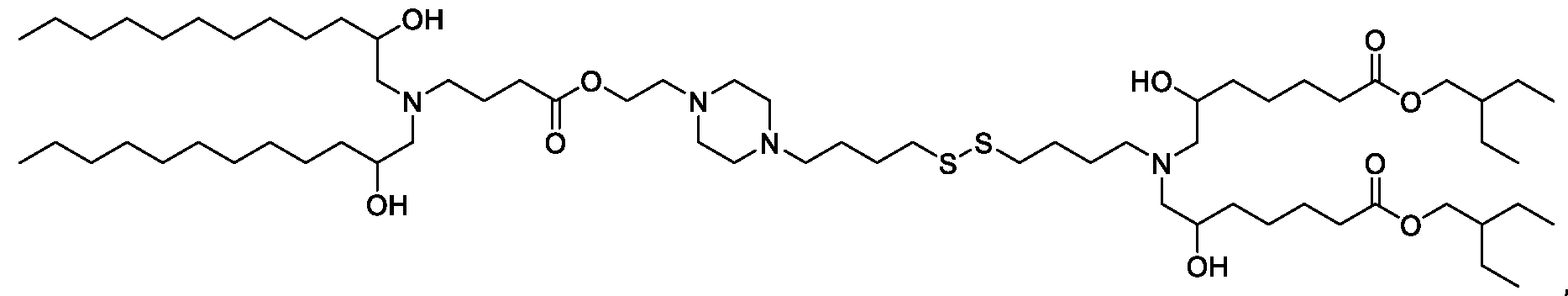
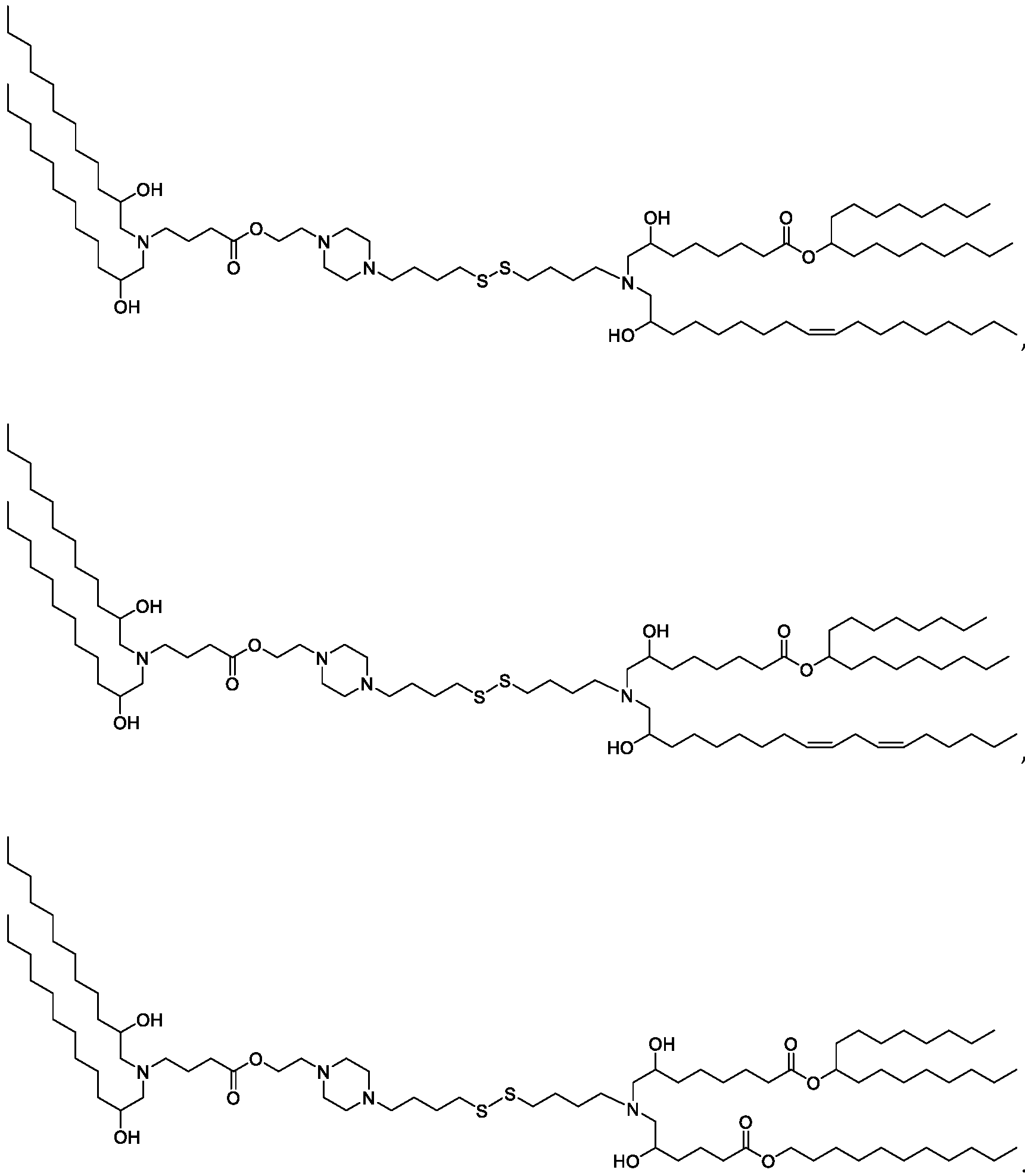
- the "Good" HEPES, HEPPS, and HEPBS buffers form the cores of the cationic lipids of the invention and were used to synthesize unique ionizable lipids containing different degradable moieties and carbon tails.
- the core structure with a hydroxyl and sulfonic acid group on either side allows for the ionizable lipids to contain both ester and disulfide degradable moieties.
- lipid nanoparticles comprising a cationic lipid derived from HEPES, HEPPS, and HEPBS buffers, for example which possesses at least one pair of asymmetric lipid tails on either arm of the lipid molecule, exhibit improved properties relative to lipid nanoparticles comprising other cationic lipids derived from "Good" buffers, such as those in WO 2022/221688 Al, incorporated herein by reference and are very effective for the intramuscular delivery of mRNA encapsulated in said lipid nanoparticles.
- lipid nanoparticles comprising the cationic lipids of the present invention have demonstrated high levels of peptide or protein expression when delivering mRNA encoding for said peptide or protein by intramuscular delivery.
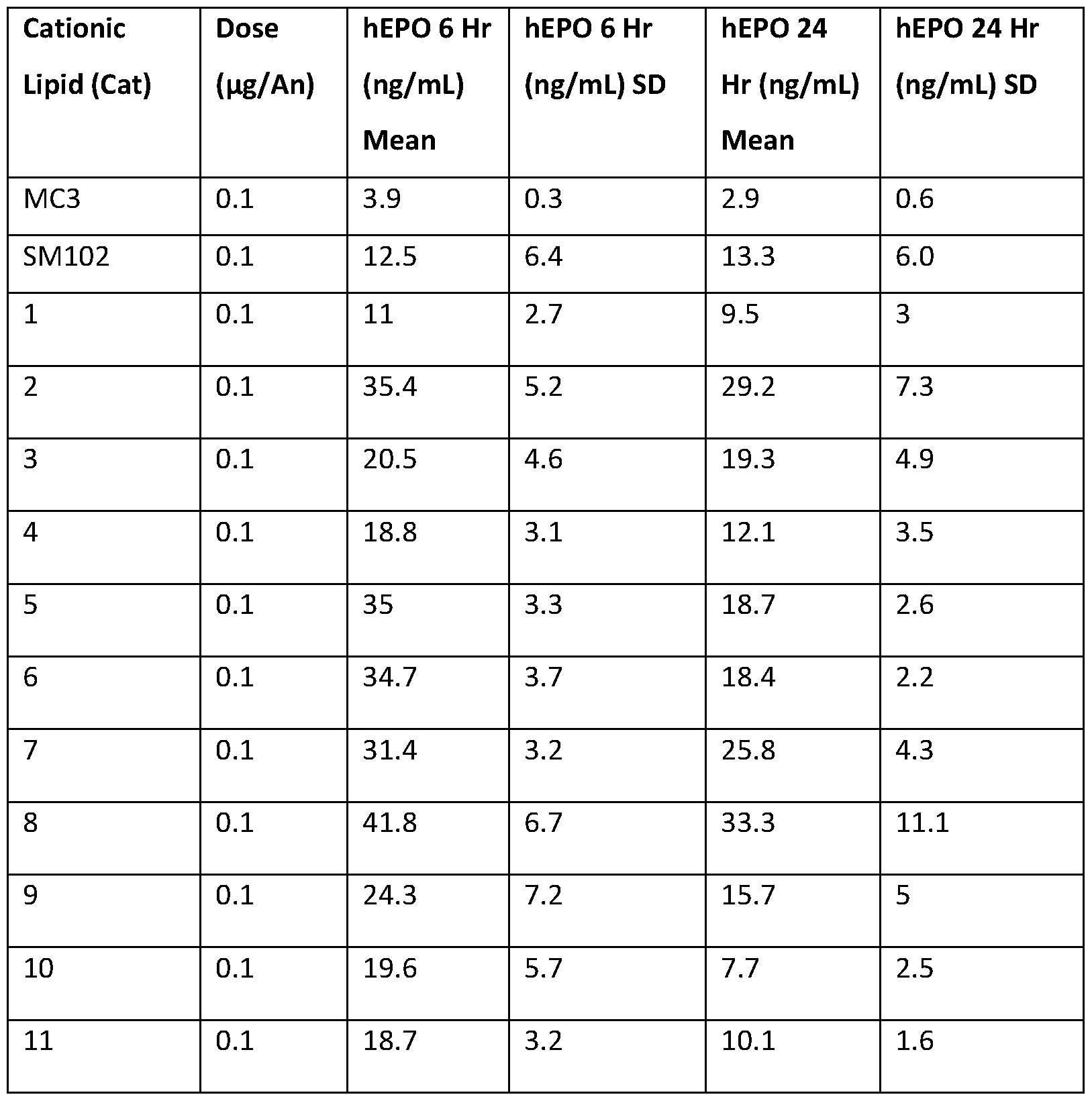
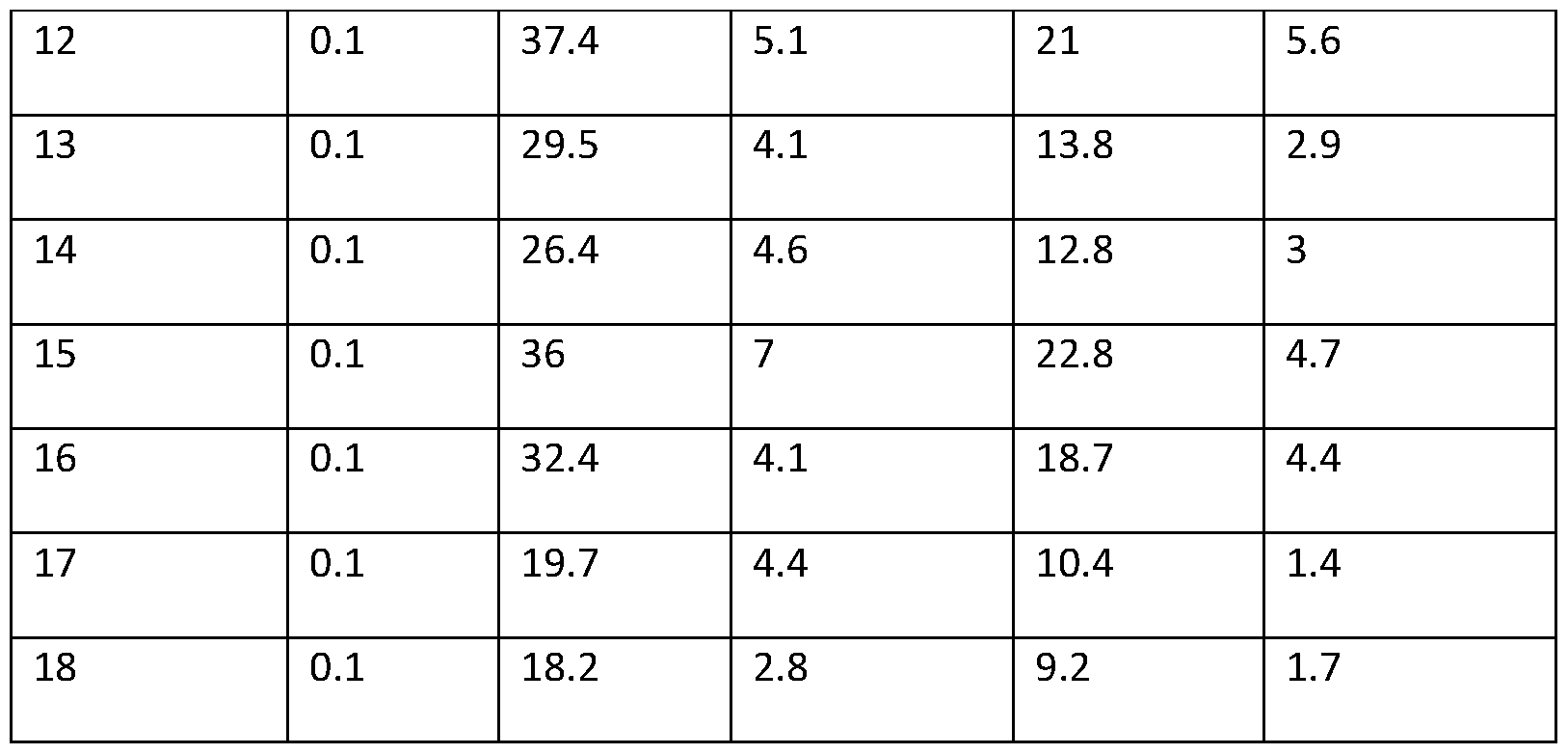
- lipid nanoparticles comprising cationic lipids of the present invention and encapsulating human erythropoietin (hEPO) mRNA achieved improved expression of hEPO mRNA when administered to mice by intramuscular delivery to lipid nanoparticles comprising MC3, which is currently used for in vivo delivery of e.g. siRNA (see W02010/144740).
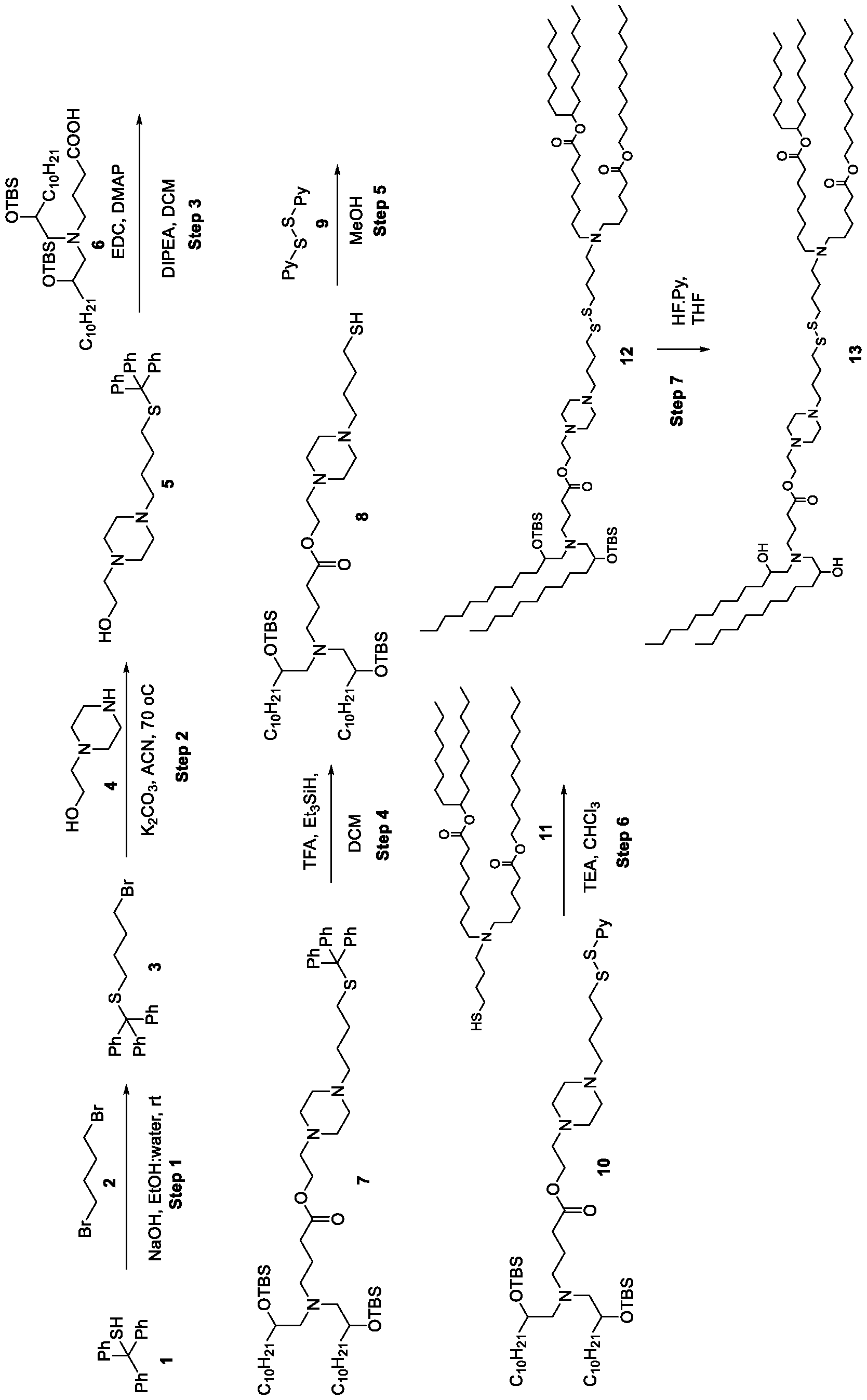
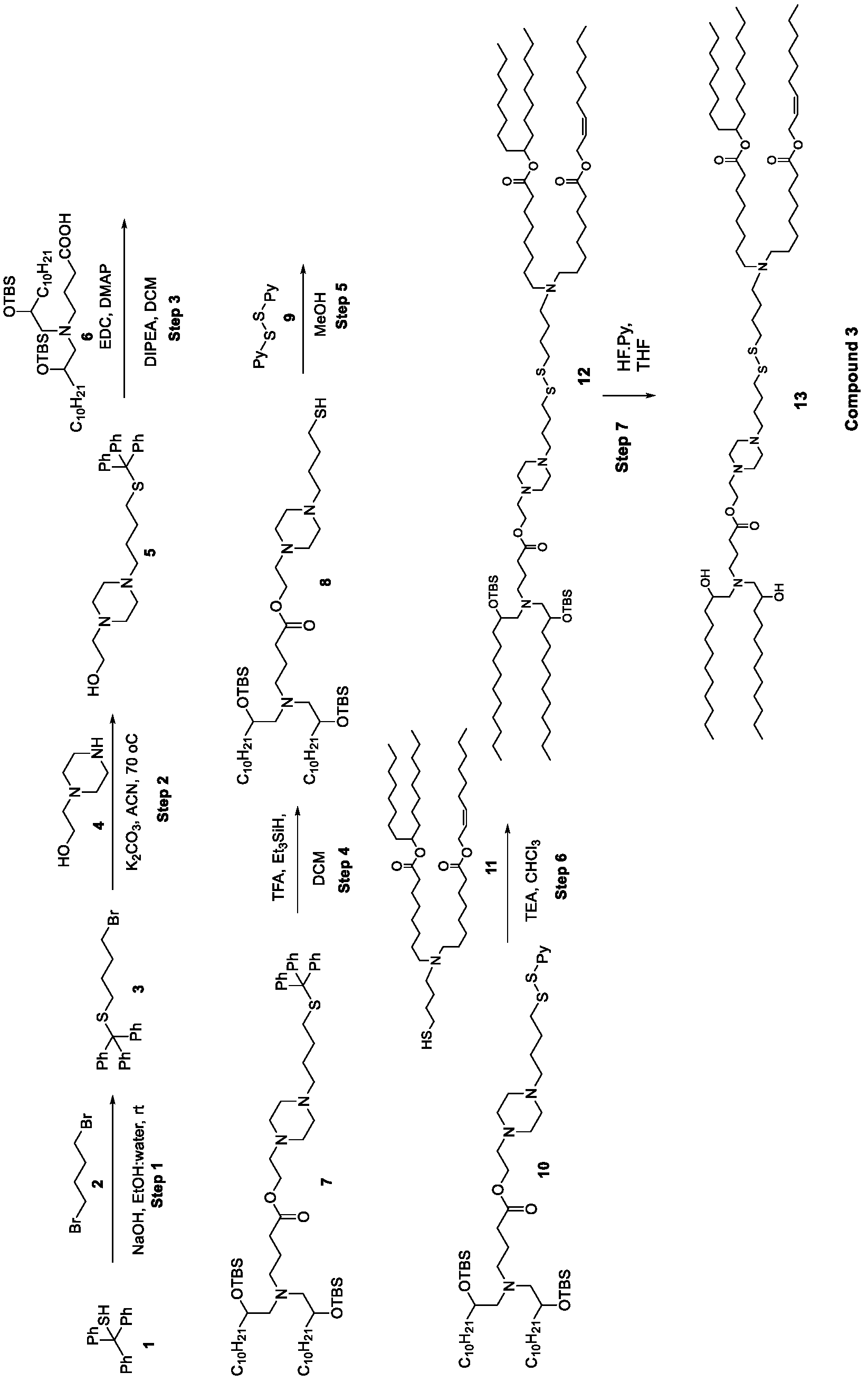
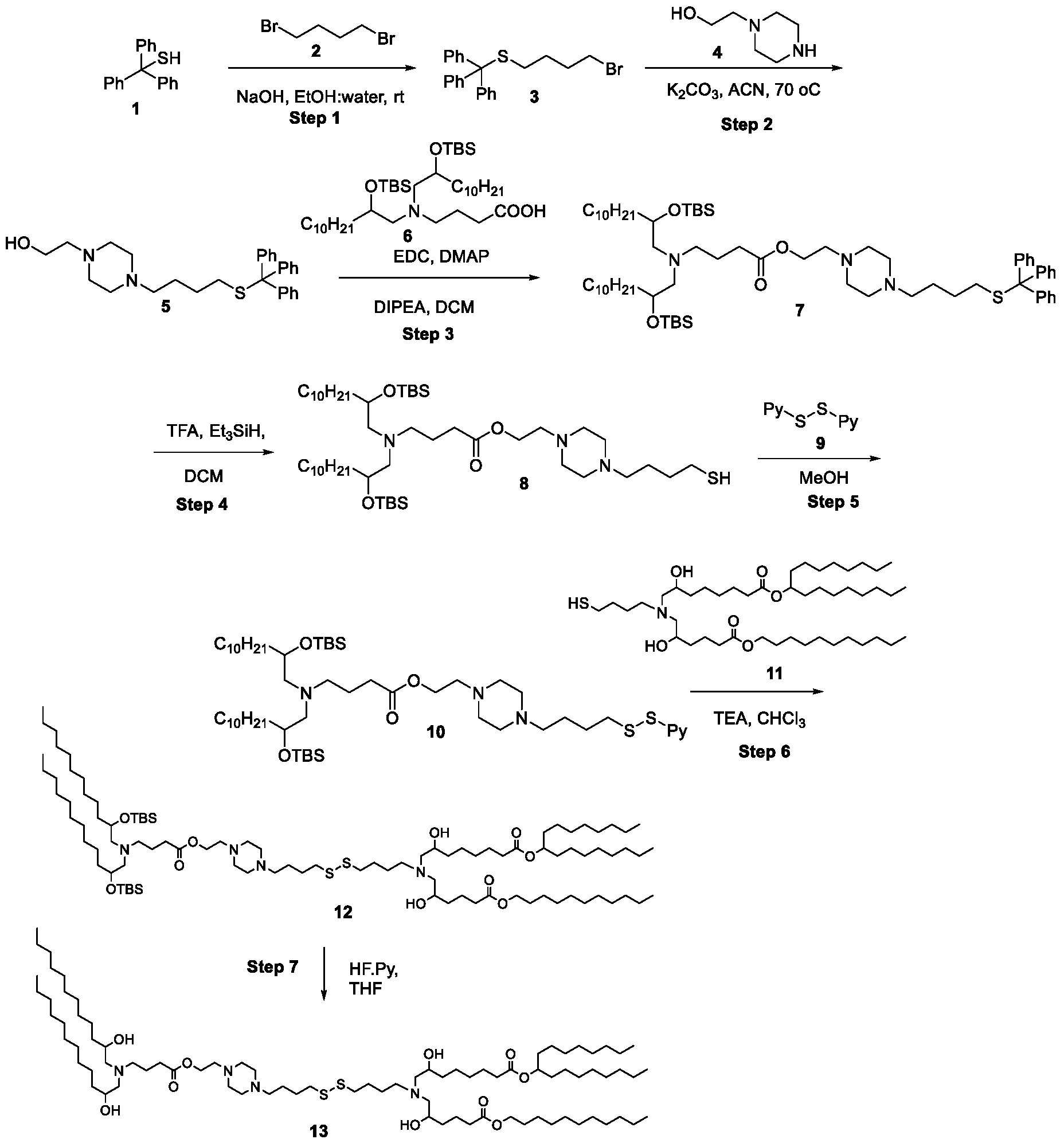
- the cationic lipids of the present invention are also more straightforward to synthesize than other cationic lipids, such as MC3. Indeed, the synthesis of MC3 involves a six-step process and requires handling of a Grignard reagent. In contrast, the cationic lipids of the present invention can be synthesized from readily available starting reagents, such as "Good's" buffers (see Table A).
- the cationic lipids of the present invention also comprise cleavable groups (e.g., esters, thioesters, disulphides, carbonates, carbamates and thiocarbamates) that are contemplated to improve biodegradability and thus contribute to their favorable safety profile.
- cleavable groups e.g., esters, thioesters, disulphides, carbonates, carbamates and thiocarbamates
- these compounds are capable of highly effective in vivo intramuscular delivery of various therapeutic agents and vaccines (e.g., therapeutic agents and/or vaccines for use in Flu, acne, chlamydia, or a disease or disorder induced by infection with P. gingivalis). It is also contemplated that lipid nanoparticles comprising these cationic lipid compounds are capable of highly effective in vivo delivery while maintaining a favorable safety profile. It is also contemplated that lipid nanoparticles comprising these cationic lipid compounds may exhibit improved degradation in vivo.
- therapeutic agents and vaccines e.g., therapeutic agents and/or vaccines for use in Flu, acne, chlamydia, or a disease or disorder induced by infection with P. gingivalis.
- lipid nanoparticles comprising these cationic lipid compounds are capable of highly effective in vivo delivery while maintaining a favorable safety profile. It is also contemplated that lipid nanoparticles comprising these cationic lipid compounds may exhibit improved degradation in
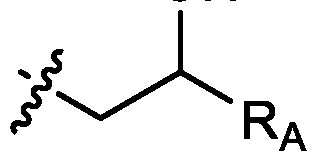
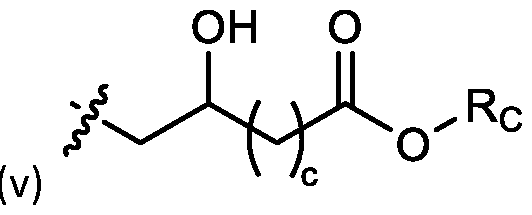
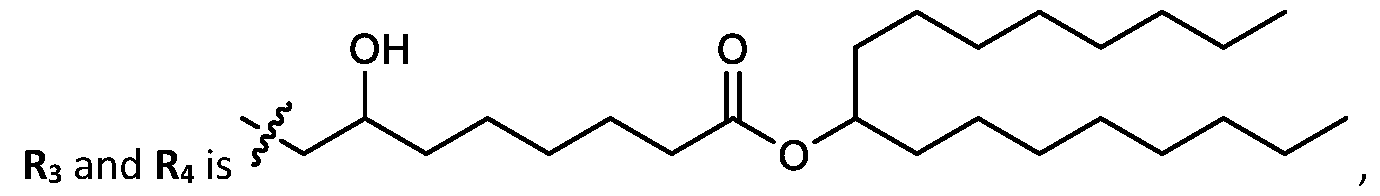
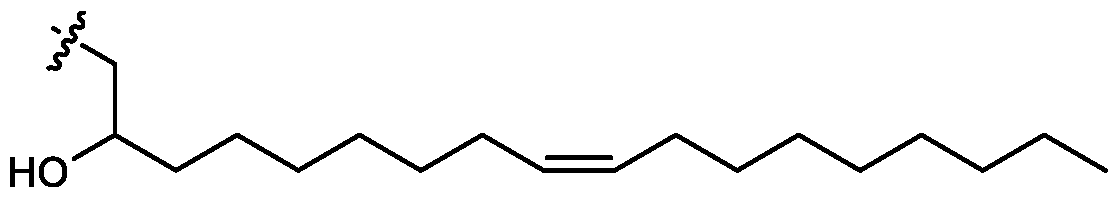
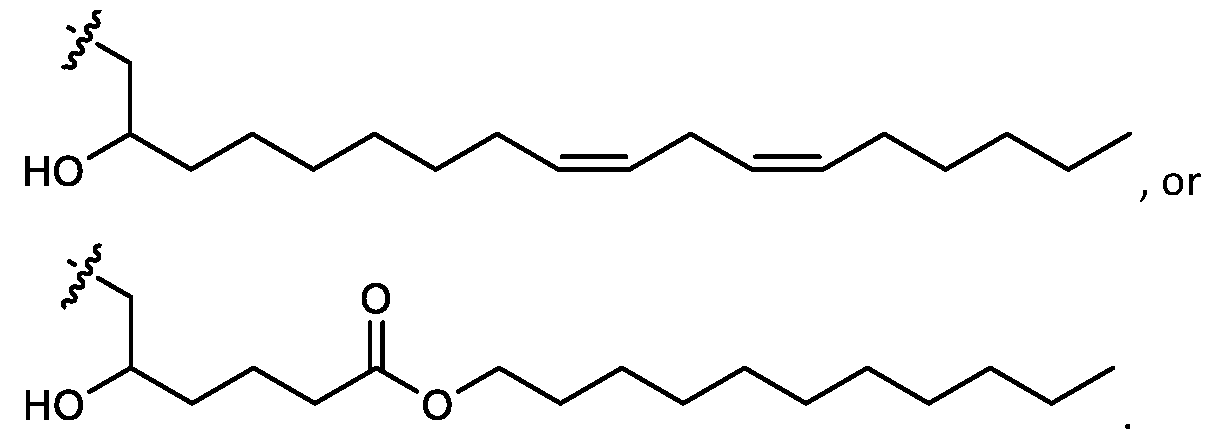
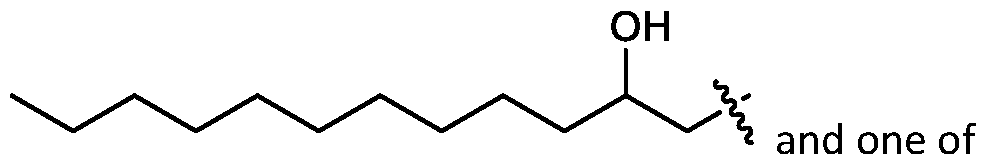
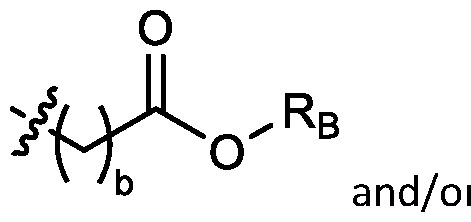
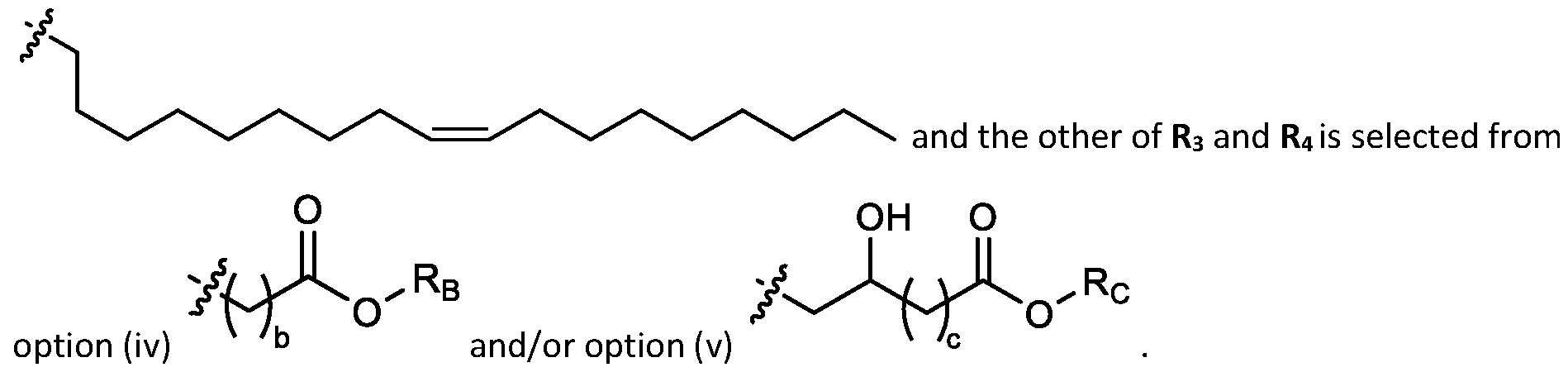
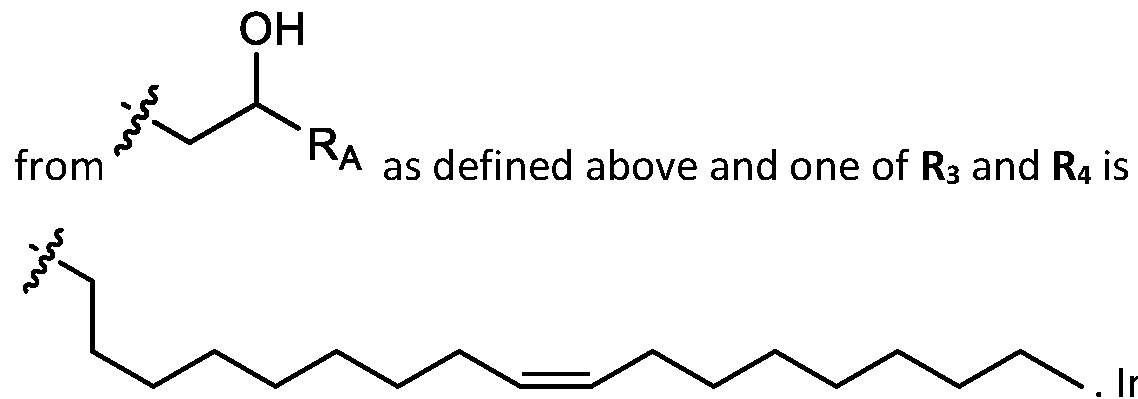
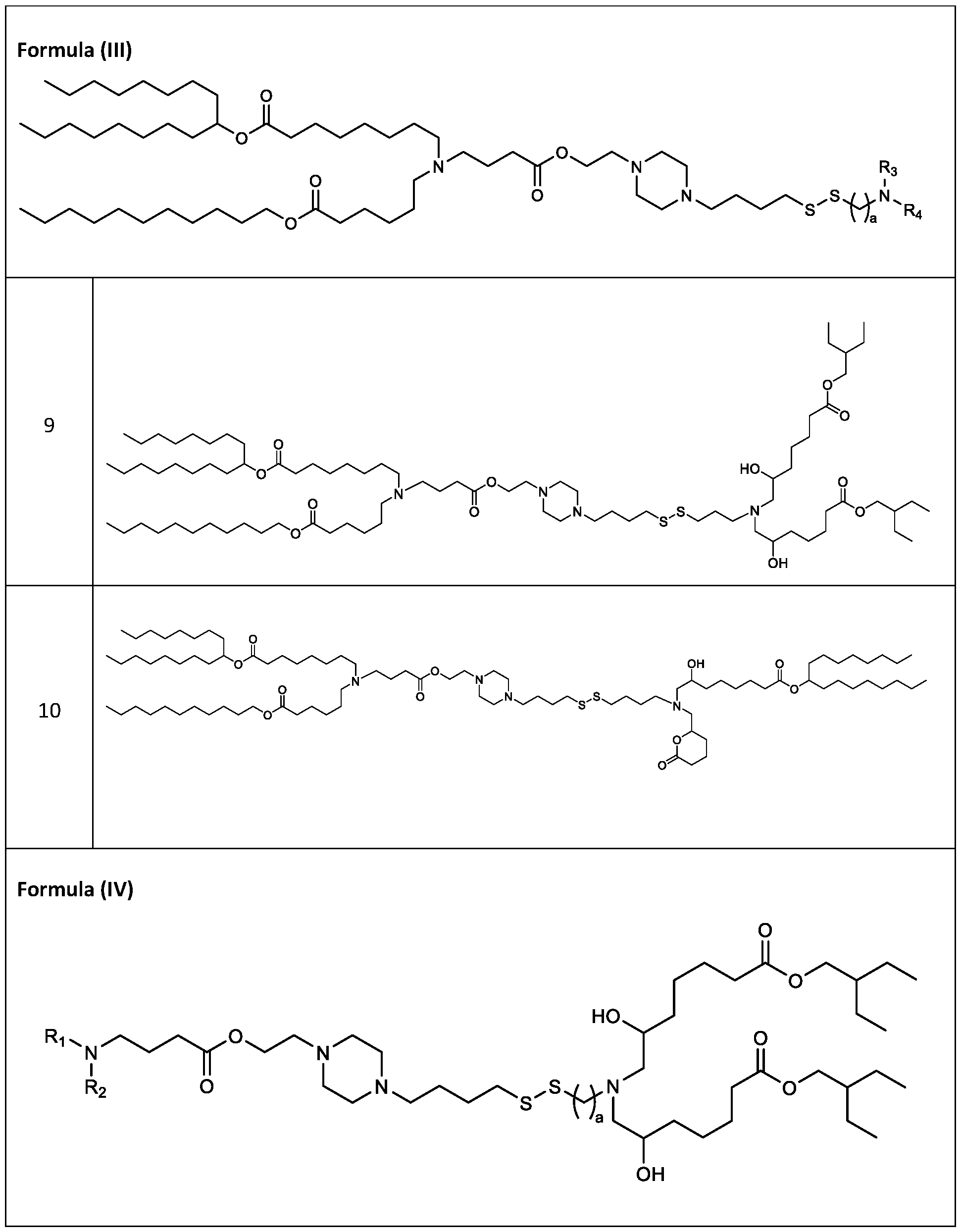
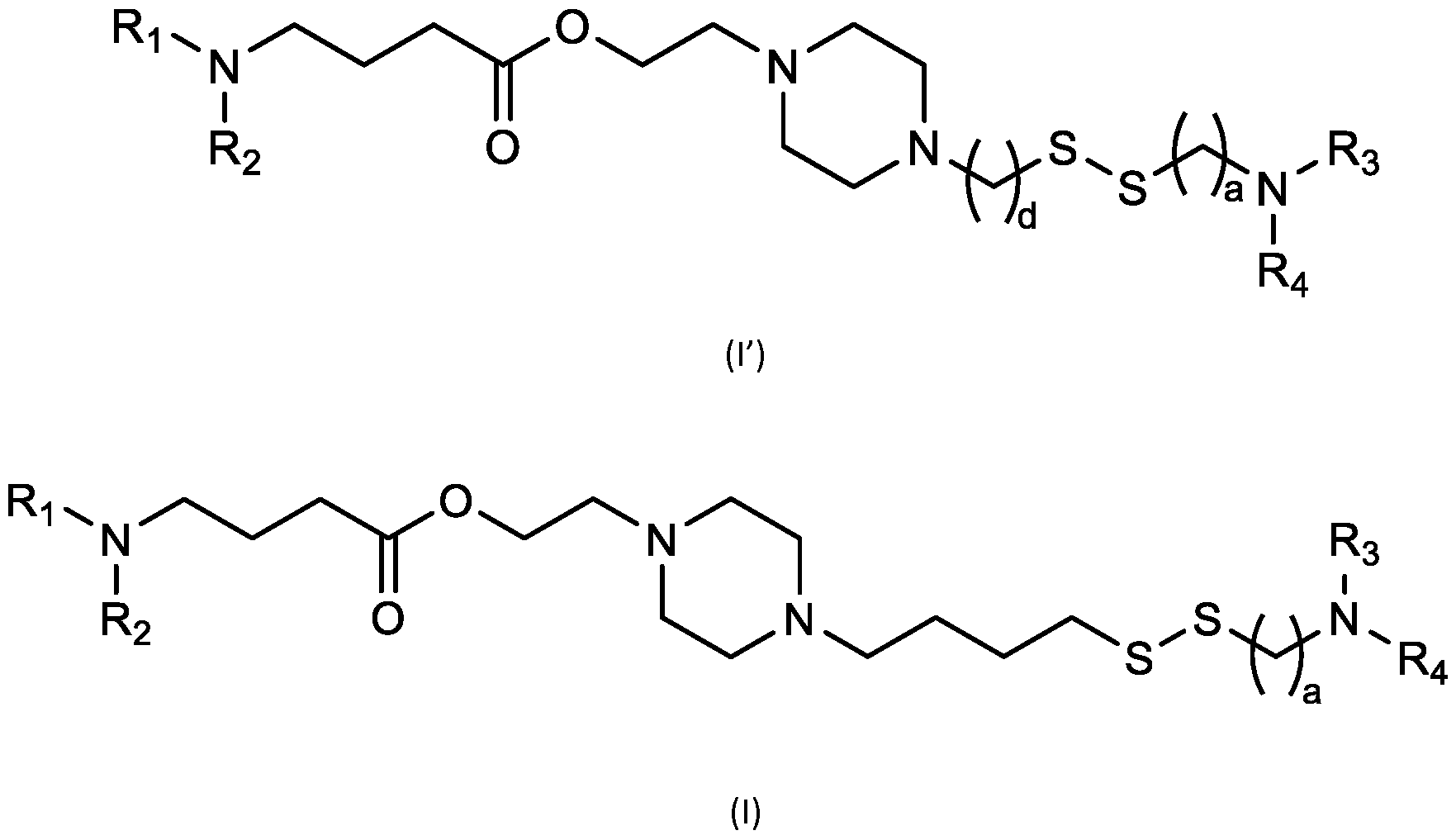
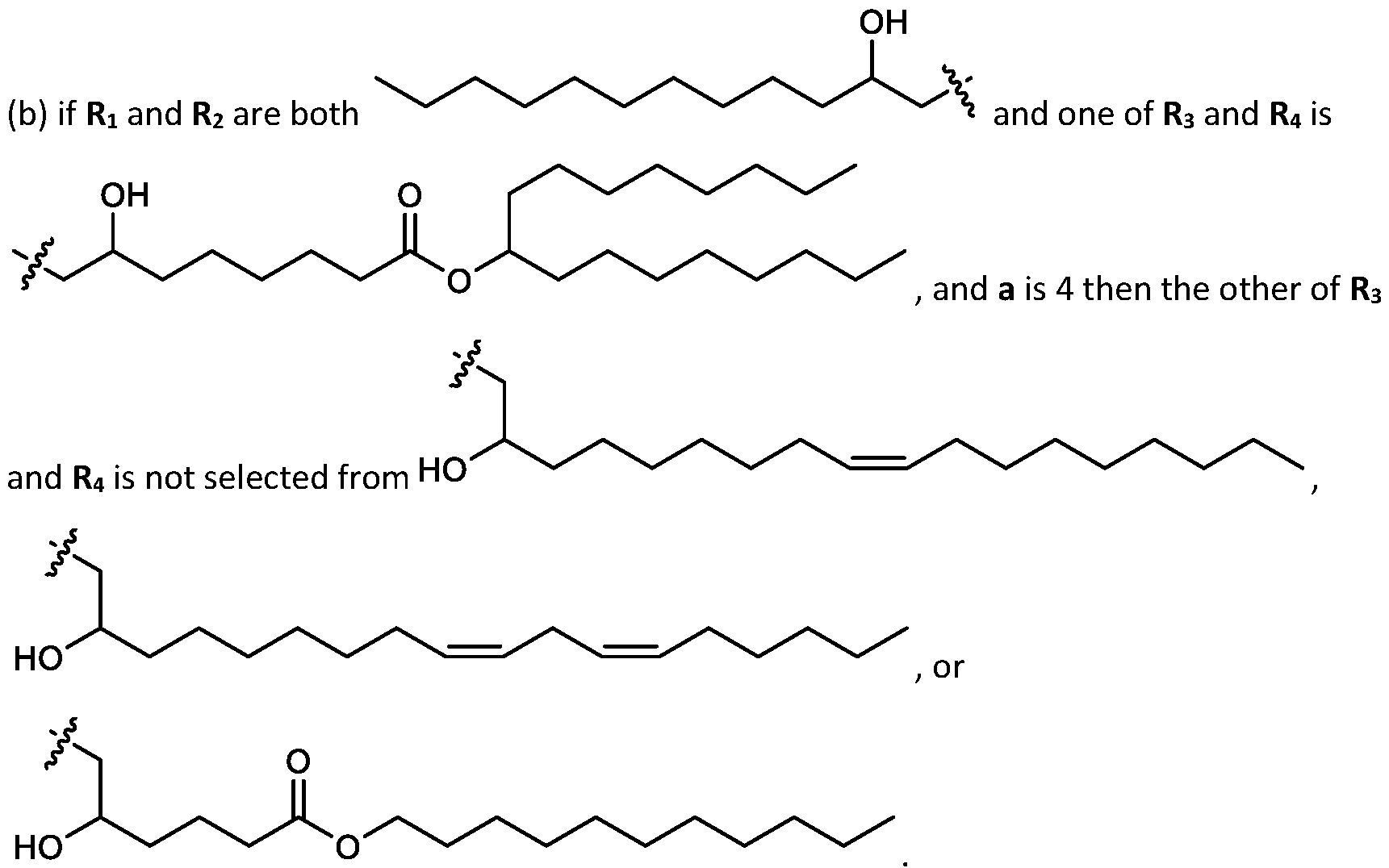
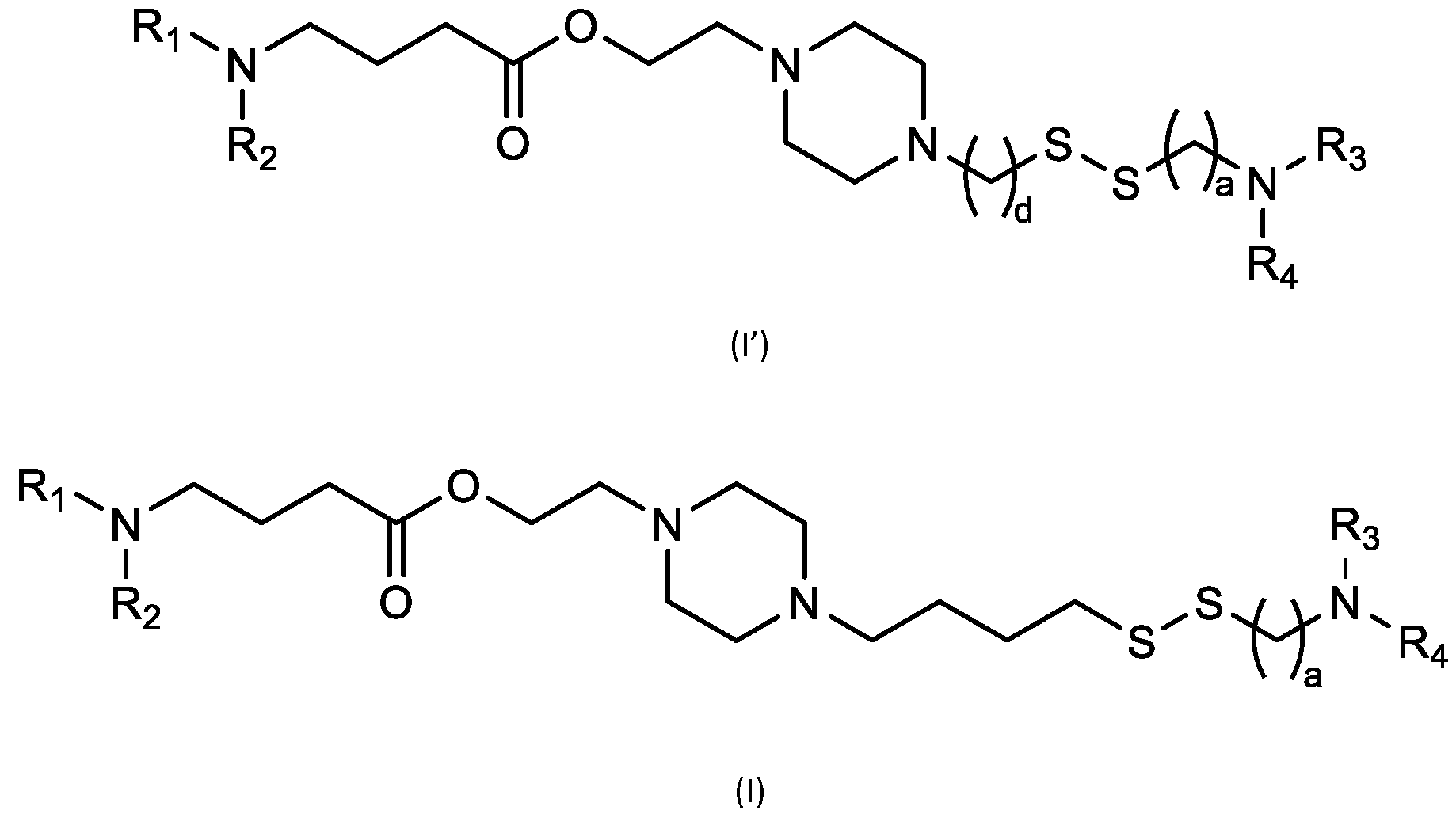
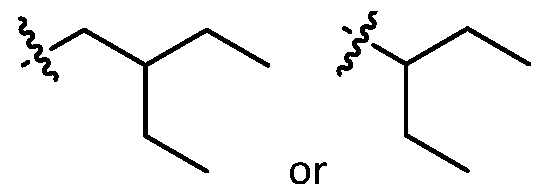
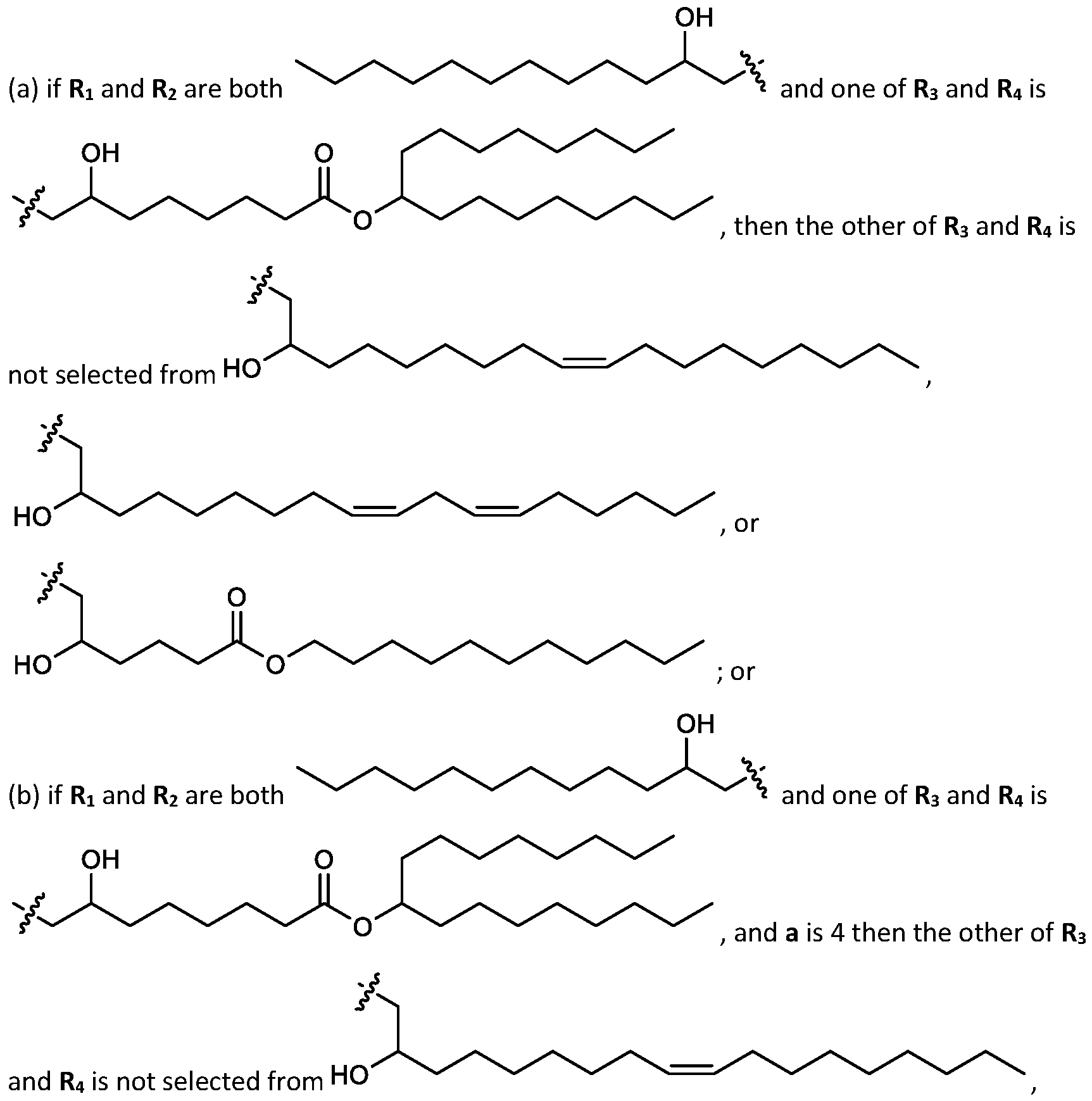
- cationic lipids having a structure according to Formula (I'), optionally Formula (I): or a pharmaceutically acceptable salt thereof, wherein: a is 3 or 4; each of Ri, R2, R3, and R 4 is independently selected from:
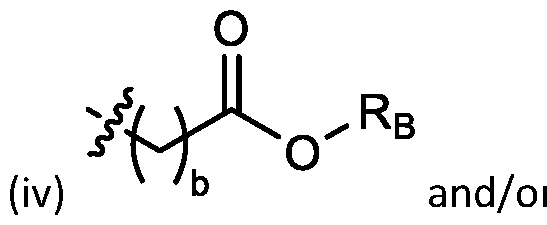
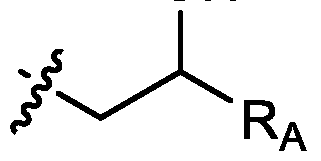
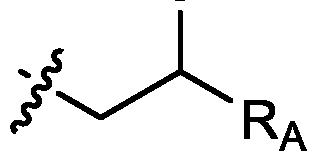
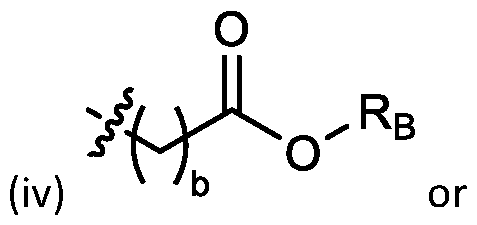
- each R A is independently selected from optionally substituted C3-C28 alkyl, optionally substituted C3-C28 alkenyl, and -W 1 -C(O)-O-X 1 , wherein each W 1 is independently selected from optionally substituted C3-C5 alkylene and optionally substituted C3-C5 alkenylene, and each X 1 is independently selected from optionally substituted C5-C30 alkyl and optionally substituted C5-C30 alkenyl; o
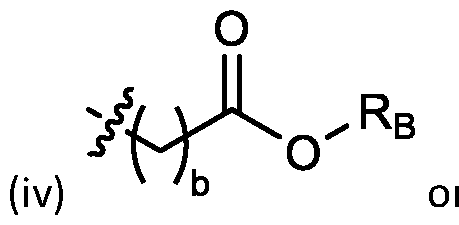
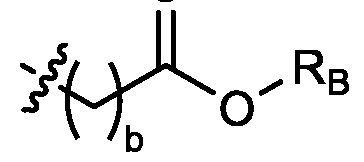
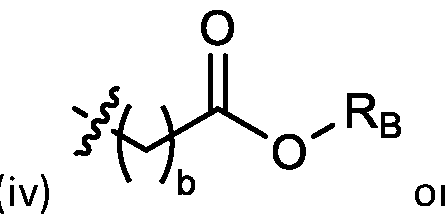
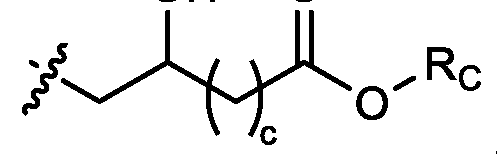
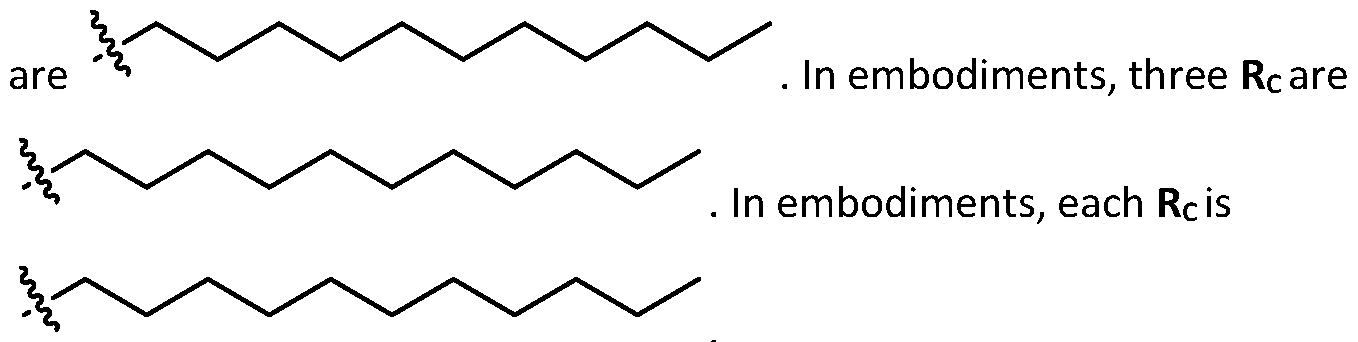
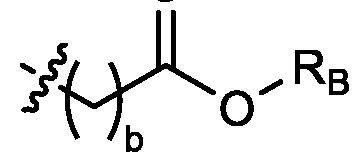
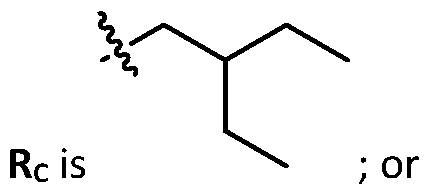
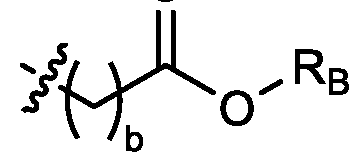
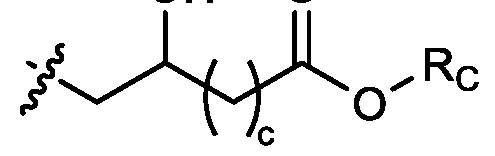
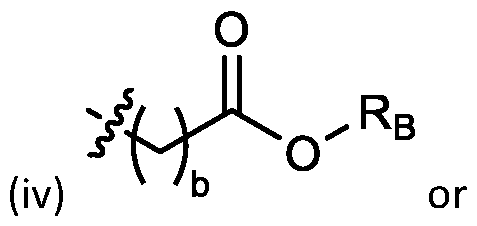
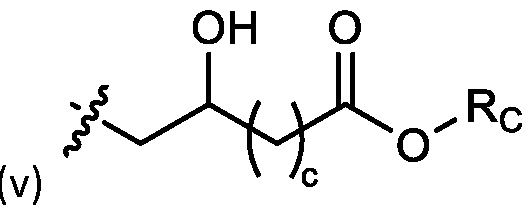
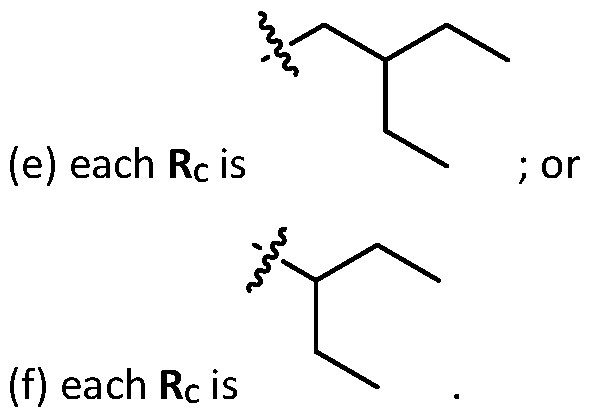
- each R B is independently selected from optionally substituted C5-C30 alkyl, optionally substituted C5-C30 alkenyl; and (v) , wherein each R C independently selected from optionally substituted C 5 -C 30 alkyl, optionally substituted C 5 -C 30 alkenyl; each b is independently selected from 5, 6, and 7; each c is independently selected from 3, 4, and 5; d is 2, 3, or 4; and wherein R 1 and R 2 are not identical to one another and/or R 3 and R 4 are not identical to one another; and wherein at least one of R 1 , R 2 , R 3 , and R 4 , is independently selected from option r option above.
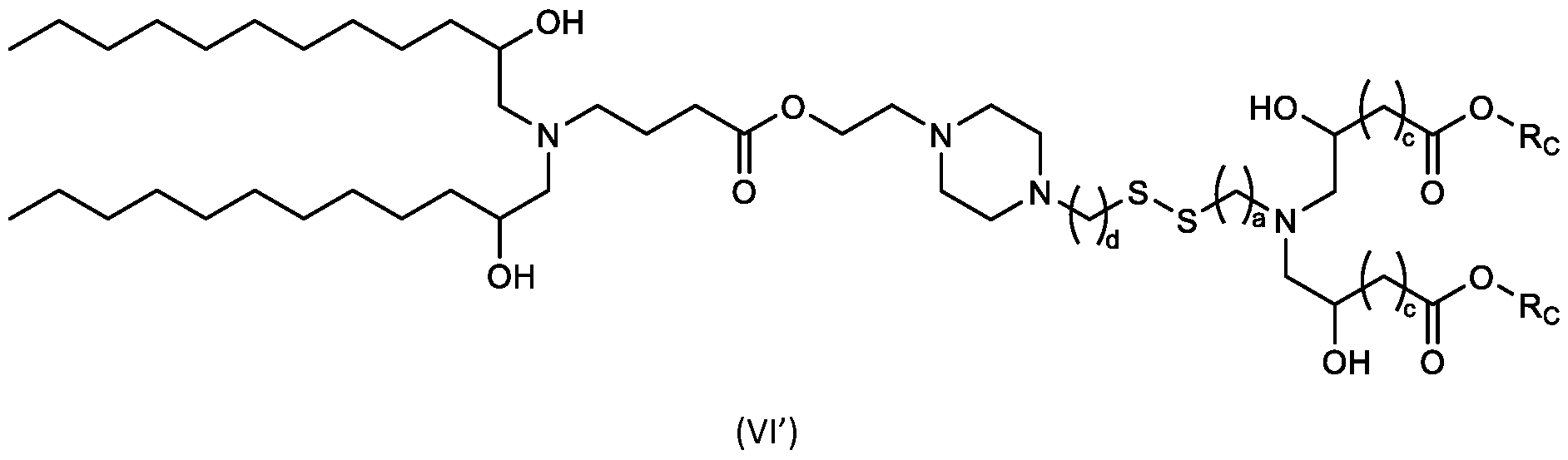
- cationic lipids having a structure according to Formula (VI’), or a pharmaceutically acceptable salt thereof, wherein: a is 3 or 4; d is 2, 3, or 4; each c is independently selected from 3 and 4; and each Rc is independently selected from
- composition comprising one or more cationic lipids of the present invention or pharmaceutically acceptable salts thereof, one or more non-cationic lipids, one or more cholesterol-based lipids and one or more PEG-modified lipids.
- compositions of the invention for use in methods of treating or preventing a disease or disorder amenable to treatment or prevention by a peptide or protein encoded by mRNA that is encapsulated within said composition.
- Methods of treating or preventing a disease or disorder wherein said method comprises administering to a subject in need thereof a composition of the invention and wherein the disease is amenable to treatment or prevention by a peptide or protein encoded by mRNA that is encapsulated within said composition are also provided.
- Figure 1 shows HAI Titers after 35 days of post intramuscular injection (0.4 pg/50 pL) in mice of mRNA encoded influenza antigen encapsulated within lipid nanoparticles comprising cationic lipids of the present invention.
- amino acid in its broadest sense, refers to any compound and/or substance that can be incorporated into a polypeptide chain.
- an amino acid has the general structure H2N-C(H)(R)-COOH.
- an amino acid is a naturally occurring amino acid.
- an amino acid is a synthetic amino acid; in some embodiments, an amino acid is a d-amino acid; in some embodiments, an amino acid is an l-amino acid.
- Standard amino acid refers to any of the twenty standard l-amino acids commonly found in naturally occurring peptides.
- Nonstandard amino acid refers to any amino acid, other than the standard amino acids, regardless of whether it is prepared synthetically or obtained from a natural source.
- synthetic amino acid encompasses chemically modified amino acids, including but not limited to salts, amino acid derivatives (such as amides), and/or substitutions.
- Amino acids, including carboxy- and/or amino-terminal amino acids in peptides can be modified by methylation, amidation, acetylation, protecting groups, and/or substitution with other chemical groups that can change the peptide's circulating half-life without adversely affecting their activity. Amino acids may participate in a disulfide bond.
- Amino acids may comprise one or post-translational modifications, such as association with one or more chemical entities (e.g., methyl groups, acetate groups, acetyl groups, phosphate groups, formyl moieties, isoprenoid groups, sulfate groups, polyethylene glycol moieties, lipid moieties, carbohydrate moieties, biotin moieties, etc.).
- chemical entities e.g., methyl groups, acetate groups, acetyl groups, phosphate groups, formyl moieties, isoprenoid groups, sulfate groups, polyethylene glycol moieties, lipid moieties, carbohydrate moieties, biotin moieties, etc.
- amino acid is used interchangeably with "amino acid residue,” and may refer to a free amino acid and/or to an amino acid residue of a peptide. It will be apparent from the context in which the term is used whether it refers to a free amino acid or a residue of
- animal refers to any member of the animal kingdom. In some embodiments, “animal” refers to humans, at any stage of development. In some embodiments, “animal” refers to non-human animals, at any stage of development. In certain embodiments, the non-human animal is a mammal (e.g., a rodent, a mouse, a rat, a rabbit, a monkey, a dog, a cat, a sheep, a bovine, a primate, and/or a pig). In some embodiments, animals include, but are not limited to, mammals, birds, reptiles, amphibians, fish, insects, and/or worms. In some embodiments, an animal may be a transgenic animal, genetically engineered animal, and/or a clone.
- mammal e.g., a rodent, a mouse, a rat, a rabbit, a monkey, a dog, a cat, a sheep, a bovine, a primate, and/or
- biologically active refers to a characteristic of any agent that has activity in a biological system, and particularly in an organism. For instance, an agent that, when administered to an organism, has a biological effect on that organism, is considered to be biologically active.
- delivery encompasses both local and systemic delivery.
- delivery of mRNA encompasses situations in which an mRNA is delivered to a target tissue and the encoded protein is expressed and retained within the target tissue (also referred to as “local distribution” or “local delivery”), and situations in which an mRNA is delivered to a target tissue and the encoded protein is expressed and secreted into patient's circulation system (e.g., serum) and systematically distributed and taken up by other tissues (also referred to as “systemic distribution” or “systemic delivery”).
- patient's circulation system e.g., serum
- expression refers to translation of an mRNA into a polypeptide, assemble multiple polypeptides into an intact protein (e.g., enzyme) and/or post-translational modification of a polypeptide or fully assembled protein (e.g., enzyme).
- intact protein e.g., enzyme
- post-translational modification e.g., enzyme
- a "functional" biological molecule is a biological molecule in a form in which it exhibits a property and/or activity by which it is characterized.
- Half-life As used herein, the term "half-life" is the time required for a quantity such as nucleic acid or protein concentration or activity to fall to half of its value as measured at the beginning of a time period.
- Helper lipid refers to any neutral or zwitterionic lipid material including cholesterol. Without wishing to be held to a particular theory, helper lipids may add stability, rigidity, and/or fluidity within lipid bilayers/nanoparticles.
- improve, increase, or reduce As used herein, the terms “improve,” “increase,” or “reduce,” or grammatical equivalents, indicate values that are relative to a baseline measurement, such as a measurement in the same individual prior to initiation of the treatment described herein, or a measurement in a control subject (or multiple control subject) in the absence of the treatment described herein.
- a “control subject” is a subject afflicted with the same form of disease as the subject being treated, who is about the same age as the subject being treated.
- in vitro refers to events that occur in an artificial environment, e.g., in a test tube or reaction vessel, in cell culture, etc., rather than within a multicellular organism.
- in vivo refers to events that occur within a multi-cellular organism, such as a human and a non-human animal. In the context of cell-based systems, the term may be used to refer to events that occur within a living cell (as opposed to, for example, in vitro systems).
- Liposome refers to any lamellar, multilamellar, or solid nanoparticle vesicle.
- a liposome as used herein can be formed by mixing one or more lipids or by mixing one or more lipids and polymer(s).
- a liposome suitable for the present invention contains a cationic lipid(s) and optionally further comprises:
- messenger RNA As used herein, the term "messenger RNA (mRNA)" or “mRNA” refers to a polynucleotide that encodes at least one polypeptide. mRNA as used herein encompasses both modified and unmodified RNA. The term “modified mRNA” related to mRNA comprising at least one chemically modified nucleotide. mRNA may contain one or more coding and non-coding regions. mRNA can be purified from natural sources, produced using recombinant expression systems and optionally purified, chemically synthesized, etc.
- mRNA can comprise nucleoside analogs such as analogs having chemically modified bases or sugars, backbone modifications, etc.
- An mRNA sequence is presented in the 5' to 3' direction unless otherwise indicated.
- an mRNA is or comprises natural nucleosides (e.g., adenosine, guanosine, cytidine, uridine); nucleoside analogs (e.g., 2-aminoadenosine, 2-thiothymidine, inosine, pyrrolo-pyrimidine, 3-methyl adenosine, 5-methylcytidine, C5-propynyl-cytidine, C5-propynyl-uridine, 2-aminoadenosine, C5-bromouridine, C5-fluorouridine, C5-iodouridine, C5-propynyl-uridine, C5-propynyl-cytidine, C5-methylcytidine, 2- aminoadenosine, 7-deazaadenosine, 7-deazaguanosine, 8-oxoadenosine, 8-oxoguanosine, 0(6)-
- nucleic acid refers to any compound and/or substance that is or can be incorporated into a polynucleotide chain.
- a nucleic acid is a compound and/or substance that is or can be incorporated into a polynucleotide chain via a phosphodiester linkage.
- nucleic acid refers to individual nucleic acid residues (e.g., nucleotides and/or nucleosides).
- nucleic acid refers to a polynucleotide chain comprising individual nucleic acid residues.
- nucleic acid encompasses RNA as well as single and/or double-stranded DNA and/or cDNA.
- “nucleic acid” encompasses ribonucleic acids (RNA), including but not limited to any one or more of interference RNAs (RNAi), small interfering RNA (siRNA), short hairpin RNA (shRNA), antisense RNA (aRNA), messenger RNA (mRNA), modified messenger RNA (mmRNA), long non-coding RNA (IncRNA), micro-RNA (miRNA) multimeric coding nucleic acid (MCNA), polymeric coding nucleic acid (PCNA), guide RNA (gRNA) and CRISPR RNA (crRNA).
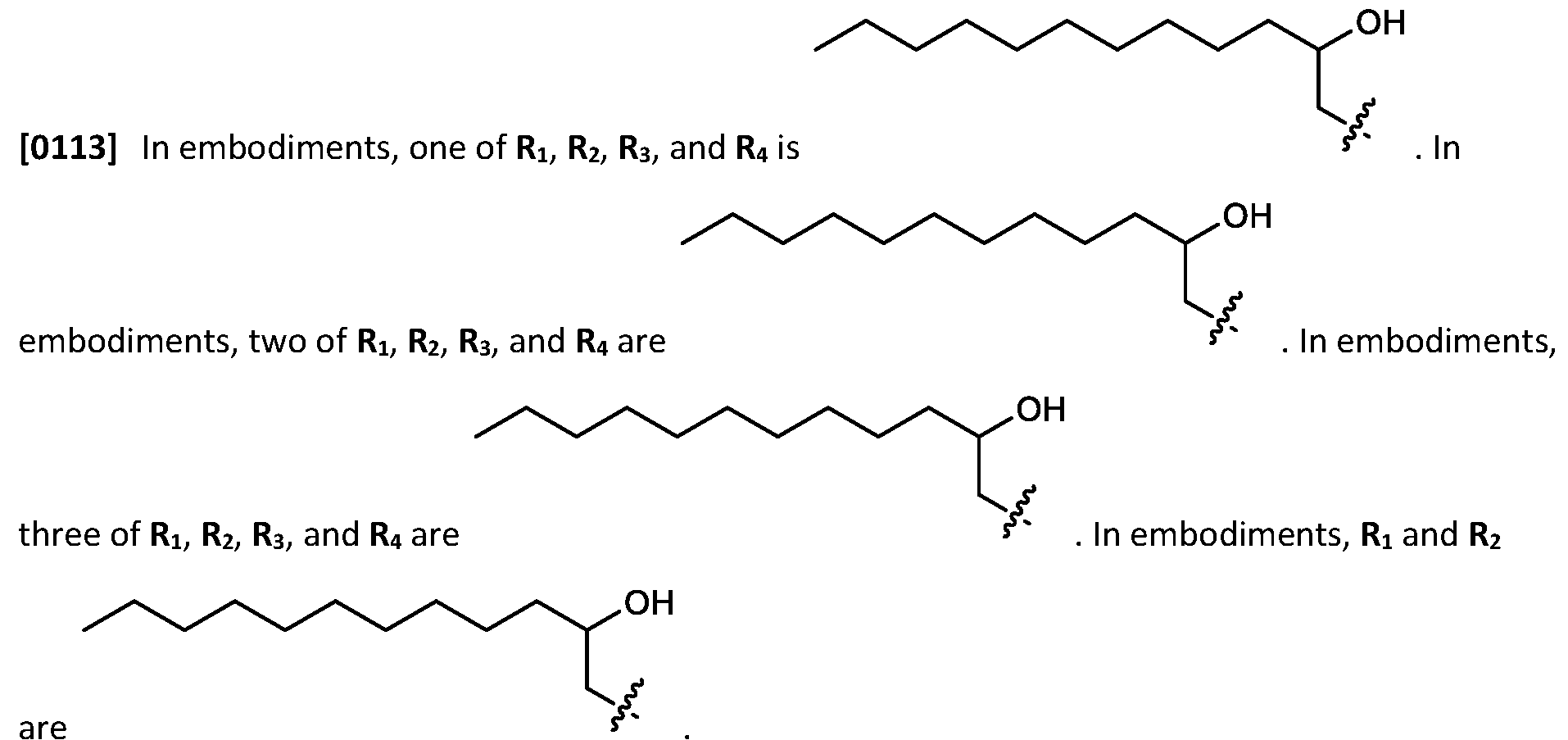
- RNAi interference RNAs
- siRNA small interfering RNA
- shRNA short hairpin RNA
- aRNA antisense RNA
- mRNA messenger RNA
- mmRNA modified messenger RNA
- IncRNA micro-RNA
- miRNA multimeric coding nucleic acid
- PCNA
- nucleic acid encompasses deoxyribonucleic acid (DNA), including but not limited to any one or more of single-stranded DNA (ssDNA), double-stranded DNA (dsDNA) and complementary DNA (cDNA). In some embodiments, "nucleic acid” encompasses both RNA and DNA.
- DNA may be in the form of antisense DNA, plasmid DNA, parts of a plasmid DNA, pre-condensed DNA, a product of a polymerase chain reaction (PCR), vectors (e.g., Pl, PAC, BAC, YAC, artificial chromosomes), expression cassettes, chimeric sequences, chromosomal DNA, or derivatives of these groups.
- RNA may be in the form of messenger RNA (mRNA), ribosomal RNA (rRNA), signal recognition particle RNA (7 SL RNA or SRP RNA), transfer RNA (tRNA), transfer-messenger RNA (tmRNA), small nuclear RNA (snRNA), small nucleolar RNA (snoRNA), SmY RNA, small Cajal body-specific RNA (scaRNA), guide RNA (gRNA), ribonuclease P (RNase P), Y RNA, telomerase RNA component (TERC), spliced leader RNA (SL RNA), antisense RNA (aRNA or asRNA), cis-natural antisense transcript (cis-NAT), CRISPR RNA (crRNA), long noncoding RNA (IncRNA), micro- RNA (miRNA), piwi-interacting RNA (piRNA), small interfering RNA (siRNA), transacting siRNA (tasiRNA), repeat associated siRNA (rasiRNA),
- patient refers to any organism to which a provided composition may be administered, e.g., for experimental, diagnostic, prophylactic, cosmetic, and/or therapeutic purposes. Typical patients include animals (e.g., mammals such as mice, rats, rabbits, non-human primates, and/or humans). In some embodiments, a patient is a human. A human includes pre- and post-natal forms.
- compositions that, within the scope of sound medical judgment, are suitable for use in contact with the tissues of human beings and animals without excessive toxicity, irritation, allergic response, or other problem or complication, commensurate with a reasonable benefit/risk ratio.
- Pharmaceutically acceptable salts are well known in the art. For example, S. M. Berge et al., describes pharmaceutically acceptable salts in detail in J. Pharmaceutical Sciences (1977) 66:1-19. Pharmaceutically acceptable salts of the compounds of this invention include those derived from suitable inorganic and organic acids and bases.
- Examples of pharmaceutically acceptable, non-toxic acid addition salts are salts of an amino group formed with inorganic acids such as hydrochloric acid, hydrobromic acid, phosphoric acid, sulfuric acid, and perchloric acid or with organic acids such as acetic acid, oxalic acid, maleic acid, tartaric acid, citric acid, succinic acid, or malonic acid, or by using other methods used in the art such as ion exchange.
- inorganic acids such as hydrochloric acid, hydrobromic acid, phosphoric acid, sulfuric acid, and perchloric acid
- organic acids such as acetic acid, oxalic acid, maleic acid, tartaric acid, citric acid, succinic acid, or malonic acid, or by using other methods used in the art such as ion exchange.
- salts include adipate, alginate, ascorbate, aspartate, benzenesulfonate, benzoate, bisulfate, borate, butyrate, camphorate, camphorsulfonate, citrate, cyclopentanepropionate, digluconate, dodecylsulfate, ethanesulfonate, formate, fumarate, glucoheptonate, glycerophosphate, gluconate, hemisulfate, heptanoate, hexanoate, hydroiodide, 2- hydroxy-ethanesulfonate, lactobionate, lactate, laurate, lauryl sulfate, malate, maleate, malonate, methanesulfonate, 2-naphthalenesulfonate, nicotinate, nitrate, oleate, oxalate, palmitate, pamoate, pectinate
- Salts derived from appropriate bases include alkali metal, alkaline earth metal, ammonium and N + (CI- 4 alkyl) 4 salts.
- Representative alkali or alkaline earth metal salts include sodium, lithium, potassium, calcium, magnesium, and the like.
- Further pharmaceutically acceptable salts include, when appropriate, nontoxic ammonium. Quaternary ammonium, and amine cations formed using counterions such as halide, hydroxide, carboxylate, sulfate, phosphate, nitrate, sulfonate, and aryl sulfonate.
- Further pharmaceutically acceptable salts include salts formed from the quarternization of an amine using an appropriate electrophile, e.g., an alkyl halide, to form a quarternized alkylated amino salt.
- Systemic distribution or delivery As used herein, the terms “systemic distribution” or “systemic delivery,” or grammatical equivalents thereof, refer to a delivery or distribution mechanism or approach that affect the entire body or an entire organism. Typically, systemic distribution or delivery is accomplished via body's circulation system, e.g., blood stream. Compared to the definition of "local distribution or delivery.”
- Subject refers to a human or any non-human animal (e.g., mouse, rat, rabbit, dog, cat, cattle, swine, sheep, horse or primate).
- a human includes pre- and post-natal forms.
- a subject is a human being.
- a subject can be a patient, which refers to a human presenting to a medical provider for diagnosis or treatment of a disease.
- the term "subject” is used herein interchangeably with “individual” or "patient.”
- a subject can be afflicted with or is susceptible to a disease or disorder but may or may not display symptoms of the disease or disorder.
- the term “substantially” refers to the qualitative condition of exhibiting total or near-total extent or degree of a characteristic or property of interest.
- One of ordinary skill in the biological arts will understand that biological and chemical phenomena rarely, if ever, go to completion and/or proceed to completeness or achieve or avoid an absolute result.
- the term “substantially” is therefore used herein to capture the potential lack of completeness inherent in many biological and chemical phenomena.
- Target tissues refers to any tissue that is affected by a disease to be treated.
- target tissues include those tissues that display disease-associated pathology, symptom, or feature.
- therapeutically effective amount As used herein, the term "therapeutically effective amount" of a therapeutic agent means an amount that is sufficient, when administered to a subject suffering from or susceptible to a disease, disorder, and/or condition, to treat, diagnose, prevent, and/or delay the onset of the symptom(s) of the disease, disorder, and/or condition. It will be appreciated by those of ordinary skill in the art that a therapeutically effective amount is typically administered via a dosing regimen comprising at least one unit dose.
- Treating refers to any method used to partially or completely alleviate, ameliorate, relieve, inhibit, prevent, delay onset of, reduce severity of and/or reduce incidence of one or more symptoms or features of a particular disease, disorder, and/or condition. Treatment may be administered to a subject who does not exhibit signs of a disease and/or exhibits only early signs of the disease for the purpose of decreasing the risk of developing pathology associated with the disease.
- Aliphatic refers to (C1-C50) hydrocarbons and includes both saturated and unsaturated hydrocarbons.
- An aliphatic may be linear, branched, or cyclic.
- (C1-C20) aliphatics can include (C1-C20) alkyls (e.g., linear or branched (C1-C20) saturated alkyls), (C2-C20) alkenyls (e.g., linear or branched (C4-C20) dienyls, linear or branched (C6-C20) trienyls, and the like), and (C2-C2o)alkynyls (e.g., linear or branched (C2-C20) alkynyls).
- (C1-C20) aliphatics can include (C3-C20) cyclic aliphatics (e.g., (C3-C2o)cycloalkyls, (C4-C20) cycloalkenyls, or (C8-C20) cycloalkynyls).
- the aliphatic may comprise one or more cyclic aliphatic and/or one or more heteroatoms such as oxygen, nitrogen, or sulfur and may optionally be substituted with one or more substituents such as alkyl, halo, alkoxyl, hydroxy, amino, aryl, ether, ester or amide.
- An aliphatic group is unsubstituted or substituted with one or more substituent groups as described herein.
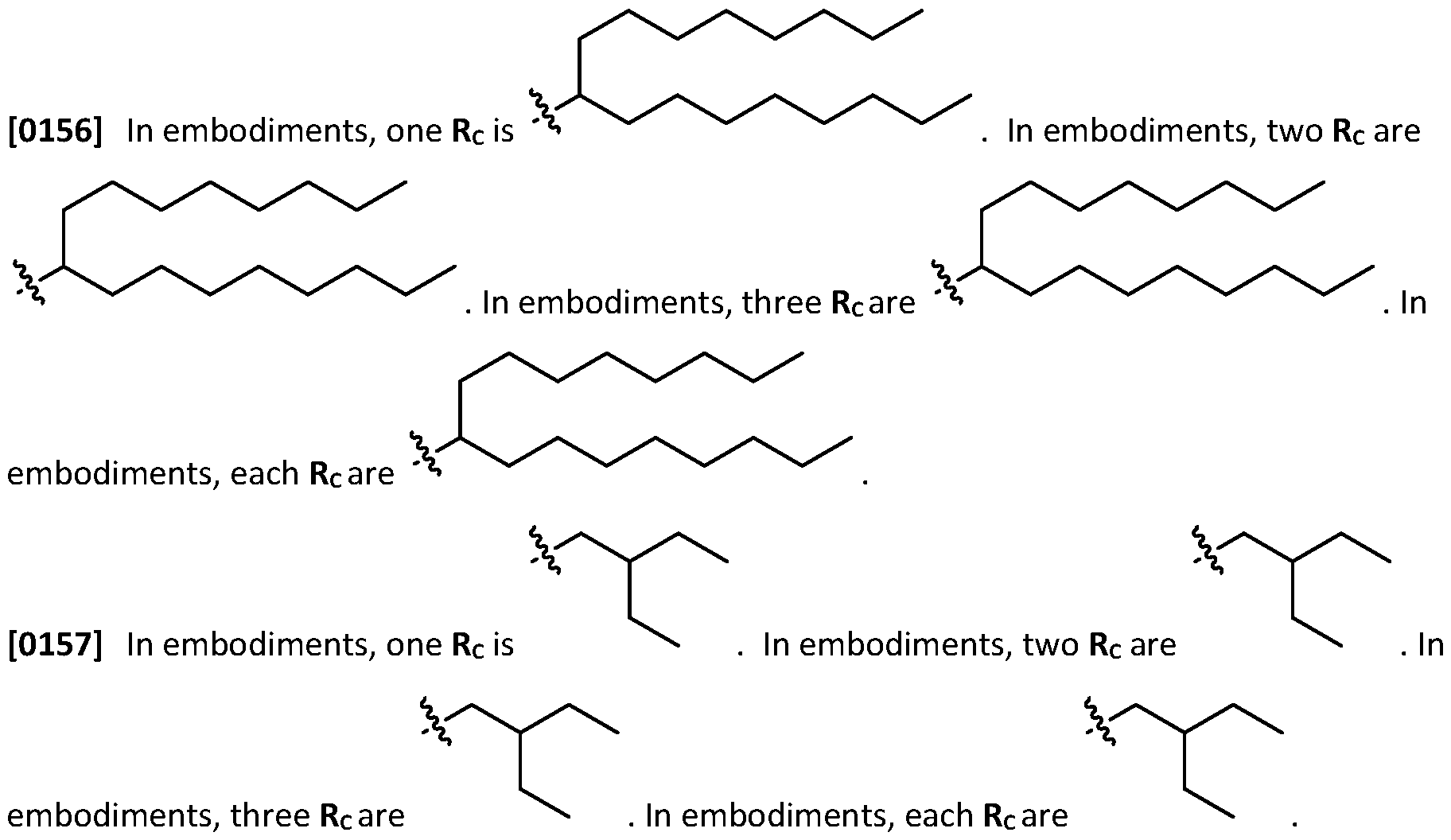
- an aliphatic may be substituted with one or more (e.g., 1, 2, 3, 4, 5, or 6 independently selected substituents) of halogen, -COR", -CO2H, -CO2R", -CN, -OH, - OR", -OCOR", -OCO2R", -NH2, -NHR", -N(R")2, -SR” or -SO2R", wherein each instance of R" independently is (C1-C20) aliphatic (e.g., (C1-C20) alkyl, (C1-C15) alkyl, (C1-C10) alkyl, or (C1-C3) alkyl).
- R independently is (C1-C20) aliphatic (e.g., (C1-C20) alkyl, (C1-C15) alkyl, (C1-C10)
- R" independently is an unsubstituted alkyl (e.g., unsubstituted (C1-C20) alkyl, (C1-C15) alkyl, (C1-C10) alkyl, or (C1-C3) alkyl). In embodiments, R" independently is unsubstituted (C1-C3) alkyl. In embodiments, the aliphatic is unsubstituted. In embodiments, the aliphatic does not include any heteroatoms.
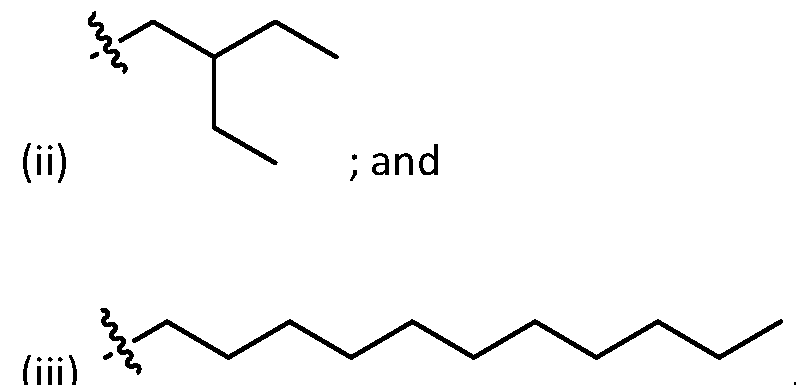
- alkyl means acyclic linear and branched hydrocarbon groups, e.g. "(C1-C30) alkyl” refers to alkyl groups having 1-30 carbons.
- An alkyl group may be linear or branched. Examples of alkyl groups include, but are not limited to, methyl, ethyl, n-propyl, isopropyl, butyl, isobutyl, sec-butyl, tert-butyl, pentyl, isopentyl tert-pentylhexyl, isohexyl, etc.
- an alkyl group may be substituted with one or more (e.g., 1, 2, 3, 4, 5, or 6 independently selected substituents) of halogen, -COR", -CO2H, -CO2R", -CN, -OH, -OR", - OCOR", -OCO2R", -NH2, -NHR", -N(R") 2 , -SR” or -SO2R", wherein each instance of R" independently is (C1-C20) aliphatic (e.g., (C1-C20) alkyl, (C1-C15) alkyl, (C1-C10) alkyl, or (C1-C3) alkyl).
- R independently is (C1-C20) aliphatic (e.g., (C1-C20) alkyl, (C1-C15) alkyl, (C1-C10) alkyl, or (C1-C3) alkyl).
- alkyl also refers to a radical of a straight-chain or branched saturated hydrocarbon group having from 1 to 50 carbon atoms (“(C1-C50) alkyl”). In some embodiments, an alkyl group has 1 to 40 carbon atoms (“(C1-C40) alkyl”). In some embodiments, an alkyl group has 1 to 30 carbon atoms (“(C1-C30) alkyl”). In some embodiments, an alkyl group has 1 to 20 carbon atoms (“(C1-C20) alkyl”). In some embodiments, an alkyl group has 1 to 10 carbon atoms (“(C1-C10) alkyl").
- an alkyl group has 1 to 9 carbon atoms ("(C1-C9) alkyl”). In some embodiments, an alkyl group has 1 to 8 carbon atoms ("(Ci-Cg) alkyl”). In some embodiments, an alkyl group has 1 to 7 carbon atoms (“(C1-C7) alkyl”). In some embodiments, an alkyl group has 1 to 6 carbon atoms (“(Ci-Cg) alkyl”). In some embodiments, an alkyl group has 1 to 5 carbon atoms (“(C1-C5) alkyl"). In some embodiments, an alkyl group has 1 to 4 carbon atoms (“(C1-C4) alkyl").
- Examples of (Ci-Cg) alkyl groups include, without limitation, methyl (Ci), ethyl (C2), n-propyl (C3), isopropyl (C3), n- butyl (C 4 ), tert-butyl (C 4 ), sec-butyl (C 4 ), iso-butyl (C 4 ), n-pentyl (C 5 ), 3-pentanyl (C 5 ), amyl (C 5 ), neopentyl (C 5 ), 3-methyl-2-butanyl (C 5 ), tertiary amyl (C 5 ), and n-hexyl (Cg).
- alkyl groups include n-heptyl (C 7 ), n-octyl (Cg) and the like. Unless otherwise specified, each instance of an alkyl group is independently unsubstituted (an "unsubstituted alkyl") or substituted (a "substituted alkyl") with one or more substituents. In certain embodiments, the alkyl group is an unsubstituted (C1-C50) alkyl. In certain embodiments, the alkyl group is a substituted (C1-C50) alkyl.
- Affixing the suffix "-ene" to a group indicates the group is a divalent moiety, e.g., arylene is the divalent moiety of aryl, and heteroarylene is the divalent moiety of heteroaryl.
- Alkylene represents a saturated divalent straight or branched chain hydrocarbon group and is exemplified by methylene, ethylene, isopropylene and the like.
- alkenylene represents an unsaturated divalent straight or branched chain hydrocarbon group having one or more unsaturated carbon-carbon double bonds that may occur in any stable point along the chain
- alkynylene herein represents an unsaturated divalent straight or branched chain hydrocarbon group having one or more unsaturated carbon-carbon triple bonds that may occur in any stable point along the chain.
- an alkylene, alkenylene, or alkynylene group may comprise one or more cyclic aliphatic and/or one or more heteroatoms such as oxygen, nitrogen, or sulfur and may optionally be substituted with one or more substituents such as alkyl, halo, alkoxyl, hydroxy, amino, aryl, ether, ester or amide.
- an alkylene, alkenylene, or alkynylene may be substituted with one or more (e.g., 1, 2, 3, 4, 5, or 6 independently selected substituents) of halogen, -COR", -CO2H, -CO2R", -CN, -OH, -OR", -OCOR", -OCO 2 R", -NH 2 , -NHR", -N(R") 2 , -SR" or -SO 2 R", wherein each instance of R" independently is (C1-C20) aliphatic (e.g., (C1-C20) alkyl, (C1-C15) alkyl, (C1-C10) alkyl, or (C1-C3) alkyl).
- R independently is (C1-C20) aliphatic (e.g., (C1-C20) alkyl, (C1-C15) alkyl, (C1-C10) alkyl, or (C1-C3) alkyl).
- R" independently is an unsubstituted alkyl (e.g., unsubstituted (C1-C20) alkyl, (C1-C15) alkyl, (C1-C10) alkyl, or (C1-C3) alkyl). In embodiments, R" independently is unsubstituted (C1-C3) alkyl. In certain embodiments, an alkylene, alkenylene, or alkynylene is unsubstituted. In certain embodiments, an alkylene, alkenylene, or alkynylene does not include any heteroatoms.
- alkenyl means any linear or branched hydrocarbon chains having one or more unsaturated carbon-carbon double bonds that may occur in any stable point along the chain, e.g. "(C2-C30) alkenyl” refers to an alkenyl group having 2-30 carbons.
- an alkenyl group includes prop-2-enyl, but-2-enyl, but-3-enyl, 2-methylprop-2-enyl, hex-2-enyl, hex-5-enyl, 2,3- dimethylbut-2-enyl, and the like.
- the alkenyl comprises 1, 2, or 3 carbon-carbon double bond.
- the alkenyl comprises a single carbon-carbon double bond. In embodiments, multiple double bonds (e.g., 2 or 3) are conjugated.
- An alkenyl group may be unsubstituted or substituted with one or more substituent groups as described herein.
- an alkenyl group may be substituted with one or more (e.g., 1, 2, 3, 4, 5, or 6 independently selected substituents) of halogen, -COR’’, -CO2H, -CO2R’’, -CN, -OH, -OR’’, -OCOR’’, -OCO2R’’, -NH2, -NHR’’, - N(R’’) 2 , -SR’’ or -SO 2 R’’, wherein each instance of R’’ independently is (C 1 -C 20 ) aliphatic (e.g., (C 1 -C 20 ) alkyl, (C 1 -C 15 ) alkyl, (C 1 -C 10 ) alkyl, or (C 1 -C 3 ) alkyl).
- R’ independently is (C 1 -C 20 ) aliphatic (e.g., (C 1 -C 20 ) alkyl, (C 1 -C 15 )
- R’’ independently is an unsubstituted alkyl (e.g., unsubstituted (C1-C20) alkyl, (C1-C15) alkyl, (C1-C10) alkyl, or (C1-C3) alkyl). In embodiments, R’’ independently is unsubstituted (C1-C3) alkyl. In embodiments, the alkenyl is unsubstituted. In embodiments, the alkenyl is substituted (e.g., with 1, 2, 3, 4, 5, or 6 substituent groups as described herein).
- an alkenyl group has 2 to 7 carbon atoms (“(C2-C7) alkenyl”). In some embodiments, an alkenyl group has 2 to 6 carbon atoms (“(C2-C6) alkenyl”). In some embodiments, an alkenyl group has 2 to 5 carbon atoms (“(C2-C5) alkenyl”). In some embodiments, an alkenyl group has 2 to 4 carbon atoms (“(C2-C4) alkenyl”). In some embodiments, an alkenyl group has 2 to 3 carbon atoms (“(C2-C3) alkenyl”). In some embodiments, an alkenyl group has 2 carbon atoms (“(C 2 ) alkenyl”).
- alkenyl examples include heptenyl (C7), octenyl (C8), octatrienyl (C8), and the like. Unless otherwise specified, each instance of an alkenyl group is independently unsubstituted (an “unsubstituted alkenyl”) or substituted (a “substituted alkenyl”) with one or more substituents. In certain embodiments, the alkenyl group is an unsubstituted (C2- C50) alkenyl. In certain embodiments, the alkenyl group is a substituted (C2-C50) alkenyl.
- An alkynyl group may be unsubstituted or substituted with one or more substituent groups as described herein.
- an alkynyl group may be substituted with one or more (e.g., 1, 2, 3, 4, 5, or 6 independently selected substituents) of halogen, -COR", -CO2H, -CO2R", -CN, -OH, -OR", -OCOR", - OCO2R", -NH2, -NHR", -N(R") 2 , -SR” or -SO2R", wherein each instance of R" independently is (C1-C20) aliphatic (e.g., (C1-C20) alkyl, (C1-C15) alkyl, (C1-C10) alkyl, or (C1-C3) alkyl).
- alkynyl also refers to a radical of a straight-chain or branched hydrocarbon group having from 2 to 50 carbon atoms and one or more carbon-carbon triple bonds (e.g., 1, 2, 3, or 4 triple bonds) and optionally one or more double bonds (e.g., 1, 2, 3, or 4 double bonds) ("(C2-C50) alkynyl").
- An alkynyl group that has one or more triple bonds, and one or more double bonds is also referred to as an "ene-yne”.
- an alkynyl group has 2 to 40 carbon atoms ("(C2- C40) alkynyl").
- an alkynyl group has 2 to 30 carbon atoms ("(C2-C30) alkynyl”). In some embodiments, an alkynyl group has 2 to 20 carbon atoms ("(C2-C20) alkynyl”). In some embodiments, an alkynyl group has 2 to 10 carbon atoms (“(C2-C10) alkynyl”). In some embodiments, an alkynyl group has 2 to 9 carbon atoms (“(C2-C9) alkynyl”). In some embodiments, an alkynyl group has 2 to 8 carbon atoms ("(C2-Cg) alkynyl").
- an alkynyl group has 2 to 7 carbon atoms ("(C2-C7) alkynyl”). In some embodiments, an alkynyl group has 2 to 6 carbon atoms ("(C 2 -C 6 ) alkynyl”). In some embodiments, an alkynyl group has 2 to 5 carbon atoms (“(C2-C5) alkynyl”). In some embodiments, an alkynyl group has 2 to 4 carbon atoms (“(C2-C4) alkynyl”). In some embodiments, an alkynyl group has 2 to 3 carbon atoms (“(C2-C3) alkynyl").
- an alkynyl group has 2 carbon atoms ("(C2) alkynyl”).
- the one or more carbon- carbon triple bonds can be internal (such as in 2-butynyl) or terminal (such as in 1-butynyl).
- each instance of an alkynyl group is independently unsubstituted (an “unsubstituted alkynyl”) or substituted (a “substituted alkynyl”) with one or more substituents.
- the alkynyl group is an unsubstituted (C2-C50) alkynyl. In certain embodiments, the alkynyl group is a substituted (C2-C50) alkynyl.
- Aryl refers to a monocyclic, bicyclic, or tricyclic carbocyclic ring system having a total of six to fourteen ring members, wherein said ring system has a single point of attachment to the rest of the molecule, at least one ring in the system is aromatic and wherein each ring in the system contains 4 to 7 ring members.
- an aryl group has 6 ring carbon atoms (“(C6) aryl,” e.g., phenyl).
- an aryl group has 10 ring carbon atoms (“(C10) aryl,” e.g., naphthyl such as 1-naphthyl and 2-naphthyl).
- an aryl group has 14 ring carbon atoms (“(C 14 ) aryl,” e.g., anthracyl).
- “Aryl” also includes ring systems wherein the aryl ring, as defined above, is fused with one or more carbocyclyl or heterocyclyl groups wherein the radical or point of attachment is on the aryl ring, and in such instances, the number of carbon atoms continue to designate the number of carbon atoms in the aryl ring system.
- aryls include phenyl, naphthyl, and anthracene.
- aryl also refers to a radical of a monocyclic or polycyclic (e.g., bicyclic or tricyclic) 4n+2 aromatic ring system (e.g., having 6, 10, or 14 ⁇ electrons shared in a cyclic array) having 6-14 ring carbon atoms and zero heteroatoms provided in the aromatic ring system (“(C 6 -C 14 ) aryl”).
- an aryl group has 6 ring carbon atoms (“(C 6 ) aryl”; e.g., phenyl).
- an aryl group has 10 ring carbon atoms (“(C 10 ) aryl”; e.g., naphthyl such as 1- naphthyl and 2-naphthyl).
- an aryl group has 14 ring carbon atoms (“(C14) aryl”; e.g., anthracyl).
- Aryl also includes ring systems wherein the aryl ring, as defined above, is fused with one or more carbocyclyl or heterocyclyl groups wherein the radical or point of attachment is on the aryl ring, and in such instances, the number of carbon atoms continue to designate the number of carbon atoms in the aryl ring system.
- each instance of an aryl group is independently unsubstituted (an “unsubstituted aryl”) or substituted (a “substituted aryl”) with one or more substituents.
- the aryl group is an unsubstituted (C6-C14) aryl.
- the aryl group is a substituted (C6-C14) aryl.
- Arylene The term “arylene” as used herein refers to an aryl group that is divalent (that is, having two points of attachment to the molecule). Exemplary arylenes include phenylene (e.g., unsubstituted phenylene or substituted phenylene).
- Carbocyclyl As used herein, “carbocyclyl” or “carbocyclic” refers to a radical of a non- aromatic cyclic hydrocarbon group having from 3 to 10 ring carbon atoms (“(C 3 -C 10 ) carbocyclyl”) and zero heteroatoms in the non-aromatic ring system. In some embodiments, a carbocyclyl group has 3 to 8 ring carbon atoms (“(C3-C8) carbocyclyl”). In some embodiments, a carbocyclyl group has 3 to 7 ring carbon atoms (“(C3-C7) carbocyclyl”).
- a carbocyclyl group has 3 to 6 ring carbon atoms (“(C3-C6) carbocyclyl”). In some embodiments, a carbocyclyl group has 4 to 6 ring carbon atoms (“(C 4 -C 6 ) carbocyclyl”). In some embodiments, a carbocyclyl group has 5 to 6 ring carbon atoms (“(C 5 -C 6 ) carbocyclyl”). In some embodiments, a carbocyclyl group has 5 to 10 ring carbon atoms (“(C5-C10) carbocyclyl”).
- Exemplary (C3-C6) carbocyclyl groups include, without limitation, cyclopropyl (C3), cyclopropenyl (C3), cyclobutyl (C4), cyclobutenyl (C4), cyclopentyl (C5), cyclopentenyl (C5), cyclohexyl (C6), cyclohexenyl (C6), cyclohexadienyl (C6), and the like.
- Exemplary (C3-C8) carbocyclyl groups include, without limitation, the aforementioned (C3-C6) carbocyclyl groups as well as cycloheptyl (C 7 ), cycloheptenyl (C 7 ), cycloheptadienyl (C 7 ), cycloheptatrienyl (C 7 ), cyclooctyl (C 8 ), cyclooctenyl (C 8 ), bicyclo[2.2.1]heptanyl (C 7 ), bicyclo[2.2.2]octanyl (C 8 ), and the like.
- Exemplary (C 3 -C 10 ) carbocyclyl groups include, without limitation, the aforementioned (C 3 -C 8 ) carbocyclyl groups as well as cyclononyl (C9), cyclononenyl (C9), cyclodecyl (C10), cyclodecenyl (C10), octahydro-1H-indenyl (C9), decahydronaphthalenyl (C10), spiro[4.5]decanyl (C10), and the like.
- the carbocyclyl group is either monocyclic (“monocyclic carbocyclyl”) or polycyclic (e.g., containing a fused, bridged or spiro ring system such as a bicyclic system (“bicyclic carbocyclyl”) or tricyclic system (“tricyclic carbocyclyl”)) and can be saturated or can contain one or more carbon-carbon double or triple bonds.
- Carbocyclyl also includes ring systems wherein the carbocyclyl ring, as defined above, is fused with one or more aryl or heteroaryl groups wherein the point of attachment is on the carbocyclyl ring, and in such instances, the number of carbons continue to designate the number of carbons in the carbocyclic ring system.
- each instance of a carbocyclyl group is independently unsubstituted (an “unsubstituted carbocyclyl”) or substituted (a “substituted carbocyclyl”) with one or more substituents.
- the carbocyclyl group is an unsubstituted C 3 -C 10 carbocyclyl.
- a cycloalkyl group has 4 to 6 ring carbon atoms (“(C 4 -C 6 ) cycloalkyl”). In some embodiments, a cycloalkyl group has 5 to 6 ring carbon atoms (“(C 5 -C 6 ) cycloalkyl”). In some embodiments, a cycloalkyl group has 5 to 10 ring carbon atoms (“(C 5 -C 10 ) cycloalkyl”). Examples of (C5-C6) cycloalkyl groups include cyclopentyl (C5) and cyclohexyl (C5).
- Examples of (C3-C6) cycloalkyl groups include the aforementioned (C5-C6) cycloalkyl groups as well as cyclopropyl (C3) and cyclobutyl (C4).
- Examples of (C3-C8) cycloalkyl groups include the aforementioned (C3-C6) cycloalkyl groups as well as cycloheptyl (C7) and cyclooctyl (C8).
- each instance of a cycloalkyl group is independently unsubstituted (an “unsubstituted cycloalkyl”) or substituted (a “substituted cycloalkyl”) with one or more substituents.
- the cycloalkyl group is an unsubstituted (C3-C10) cycloalkyl. In certain embodiments, the cycloalkyl group is a substituted (C3-C10) cycloalkyl.
- Heteroalkyl The term “heteroalkyl” is meant a branched or unbranched alkyl, alkenyl, or alkynyl group having from 1 to 14 carbon atoms in addition to 1, 2, 3 or 4 heteroatoms independently selected from the group consisting of N, O, S, and P.
- Heteroalkyls include tertiary amines, secondary amines, ethers, thioethers, amides, thioamides, carbamates, thiocarbamates, hydrazones, imines, phosphodiesters, phosphoramidates, sulfonamides, and disulfides.
- a heteroalkyl group may optionally include monocyclic, bicyclic, or tricyclic rings, in which each ring desirably has three to six members. Examples of heteroalkyls include polyethers, such as methoxymethyl and ethoxyethyl.
- Heteroalkylene The term “heteroalkylene,” as used herein, represents a divalent form of a heteroalkyl group as described herein.
- Heteroaryl The term “heteroaryl,” as used herein, represents a fully unsaturated heteroatom-containing ring wherein at least one ring atom is a heteroatom such as, but not limited to, nitrogen and oxygen.
- heteroaryl also refers to a radical of a 5-14 membered monocyclic or polycyclic (e.g., bicyclic or tricyclic) 4n+2 aromatic ring system (e.g., having 6, 10, or 14 ⁇ electrons shared in a cyclic array) having ring carbon atoms and 1 or more (e.g., 1, 2, 3, or 4 ring heteroatoms) ring heteroatoms provided in the aromatic ring system, wherein each heteroatom is independently selected from oxygen, sulfur, nitrogen, boron, silicon, and phosphorus (“5-14 membered heteroaryl”).
- heteroaryl groups that contain one or more nitrogen atoms
- the point of attachment can be a carbon or nitrogen atom, as valency permits.
- Heteroaryl polycyclic ring systems can include one or more heteroatoms in one or both rings.
- “Heteroaryl” includes ring systems wherein the heteroaryl ring, as defined above, is fused with one or more carbocyclyl or heterocyclyl groups wherein the point of attachment is on the heteroaryl ring, and in such instances, the number of ring members continue to designate the number of ring members in the heteroaryl ring system.
- Heteroaryl also includes ring systems wherein the heteroaryl ring, as defined above, is fused with one or more aryl groups wherein the point of attachment is either on the aryl or heteroaryl ring, and in such instances, the number of ring members designates the number of ring members in the fused polycyclic (aryl/heteroaryl) ring system.
- a heteroaryl group is a 5-10 membered aromatic ring system having ring carbon atoms and 1 or more (e.g., 1, 2, 3, or 4) ring heteroatoms provided in the aromatic ring system, wherein each heteroatom is independently selected from oxygen, sulfur, nitrogen, boron, silicon, and phosphorus (“5-10 membered heteroaryl”).
- a heteroaryl group is a 5-8 membered aromatic ring system having ring carbon atoms and 1 or more (e.g., 1, 2, 3, or 4) ring heteroatoms provided in the aromatic ring system, wherein each heteroatom is independently selected from oxygen, sulfur, nitrogen, boron, silicon, and phosphorus (“5-8 membered heteroaryl”).
- a heteroaryl group is a 5-6 membered aromatic ring system having ring carbon atoms and 1 or more (e.g., 1, 2, 3, or 4) ring heteroatoms provided in the aromatic ring system, wherein each heteroatom is independently selected from oxygen, sulfur, nitrogen, boron, silicon, and phosphorus (“5-6 membered heteroaryl”).
- the 5-6 membered heteroaryl has 1 or more (e.g., 1, 2, or 3) ring heteroatoms selected from oxygen, sulfur, nitrogen, boron, silicon, and phosphorus.
- the 5-6 membered heteroaryl has 1 or 2 ring heteroatoms selected from oxygen, sulfur, nitrogen, boron, silicon, and phosphorus.
- Exemplary 5-membered heteroaryl groups containing 2 heteroatoms include, without limitation, imidazolyl, pyrazolyl, oxazolyl, isoxazolyl, thiazolyl, and isothiazolyl.
- Exemplary 5-membered heteroaryl groups containing 3 heteroatoms include, without limitation, triazolyl, oxadiazolyl, and thiadiazolyl.
- Exemplary 5-membered heteroaryl groups containing 4 heteroatoms include, without limitation, tetrazolyl.
- Exemplary 6-membered heteroaryl groups containing 1 heteroatom include, without limitation, pyridinyl.
- Exemplary 6,6-bicyclic heteroaryl groups include, without limitation, naphthyridinyl, pteridinyl, quinolinyl, isoquinolinyl, cinnolinyl, quinoxalinyl, phthalazinyl, and quinazolinyl.
- Exemplary tricyclic heteroaryl groups include, without limitation, phenanthridinyl, dibenzofuranyl, carbazolyl, acridinyl, phenothiazinyl, phenoxazinyl and phenazinyl.
- heterocyclyl or “heterocyclic” refers to a radical of a 3- to 14-membered non-aromatic ring system having ring carbon atoms and 1 or more (e.g., 1, 2, 3, or 4) ring heteroatoms, wherein each heteroatom is independently selected from oxygen, sulfur, nitrogen, boron, silicon, and phosphorus (“3-14 membered heterocyclyl”).
- the point of attachment can be a carbon or nitrogen atom, as valency permits.
- each instance of heterocyclyl is independently unsubstituted (an “unsubstituted heterocyclyl”) or substituted (a “substituted heterocyclyl”) with one or more substituents.
- the heterocyclyl group is an unsubstituted 3-14 membered heterocyclyl. In certain embodiments, the heterocyclyl group is a substituted 3-14 membered heterocyclyl.
- a heterocyclyl group is a 5-10 membered non-aromatic ring system having ring carbon atoms and 1 or more (e.g., 1, 2, 3, or 4) ring heteroatoms, wherein each heteroatom is independently selected from oxygen, sulfur, nitrogen, boron, silicon, and phosphorus (“5-10 membered heterocyclyl”).
- a heterocyclyl group is a 5-8 membered non-aromatic ring system having ring carbon atoms and 1 or more (e.g., 1, 2, 3, or 4) ring heteroatoms, wherein each heteroatom is independently selected from oxygen, sulfur, nitrogen, boron, silicon, and phosphorus (“5-8 membered heterocyclyl”).
- Exemplary 5-membered heterocyclyl groups containing 1 heteroatom include, without limitation, tetrahydrofuranyl, dihydrofuranyl, tetrahydrothiophenyl, dihydrothiophenyl, pyrrolidinyl, dihydropyrrolyl and pyrrolyl- 2,5-dione.
- Exemplary 5- membered heterocyclyl groups containing 2 heteroatoms include, without limitation, dioxolanyl, oxathiolanyl and dithiolanyl.
- Exemplary 5-membered heterocyclyl groups containing 3 heteroatoms include, without limitation, triazolinyl, oxadiazolinyl, and thiadiazolinyl.
- Exemplary 6-membered heterocyclyl groups containing 1 heteroatom include, without limitation, piperidinyl, tetrahydropyranyl, dihydropyridinyl, and thianyl.
- Exemplary 6-membered heterocyclyl groups containing 2 heteroatoms include, without limitation, piperazinyl, morpholinyl, dithianyl, dioxanyl.
- Exemplary 6-membered heterocyclyl groups containing 2 heteroatoms include, without limitation, triazinanyl.
- Exemplary 7-membered heterocyclyl groups containing 1 heteroatom include, without limitation, azepanyl, oxepanyl and thiepanyl.
- Exemplary 8-membered heterocyclyl groups containing 1 heteroatom include, without limitation, azocanyl, oxecanyl and thiocanyl.
- Exemplary bicyclic heterocyclyl groups include, without limitation, indolinyl, isoindolinyl, dihydrobenzofuranyl, dihydrobenzothienyl, tetrahydrobenzothienyl, tetrahydrobenzofuranyl, tetrahydroindolyl, tetrahydroquinolinyl, tetrahydroisoquinolinyl, decahydroquinolinyl, decahydroisoquinolinyl, octahydrochromenyl, octahydroisochromenyl, decahydronaphthyridinyl, decahydro-1,8- naphthyridinyl, octahydropyrrolo[3,2-b]pyrrole,
- Heterocycloalkyl is a non-aromatic ring wherein at least one atom is a heteroatom such as, but not limited to, nitrogen, oxygen, sulfur, or phosphorus, and the remaining atoms are carbon.
- the heterocycloalkyl group can be substituted or unsubstituted.
- alkyl, alkenyl, alkynyl, acyl, carbocyclyl, heterocyclyl, aryl, and heteroaryl groups, as defined herein are, in certain embodiments, optionally substituted.
- Optionally substituted refers to a group which may be substituted or unsubstituted (e.g., “substituted” or “unsubstituted” alkyl, “substituted” or “unsubstituted” alkenyl, “substituted” or “unsubstituted” alkynyl, “substituted” or “unsubstituted” heteroalkyl, “substituted” or “unsubstituted” heteroalkenyl, “substituted” or ’unsubstituted” heteroalkynyl, “substituted” or “unsubstituted” carbocyclyl, “substituted” or “unsubstituted” heterocyclyl, “substituted” or “unsubstituted” aryl or “substituted” or “unsubstituted” heteroaryl group.
- substituted or unsubstituted
- substituted means that at least one hydrogen present on a group is replaced with a permissible substituent, e.g., a substituent which upon substitution results in a stable compound, e.g., a compound which does not spontaneously undergo transformation such as by rearrangement, cyclization, elimination, or other reaction.
- a “substituted” group has a substituent at one or more substitutable positions of the group, and when more than one position in any given structure is substituted, the substituent is either the same or different at each position.
- Exemplary counterions include halide ions (e.g., F-, Cl-, Br-, I-), NO 3 -, ClO 4 -, OH-, H 2 PO 4 -, HSO 4 -, sulfonate ions (e.g., methansulfonate, trifluoromethanesulfonate, p-toluenesulfonate, benzenesulfonate, 10-camphor sulfonate, naphthalene-2-sulfonate, naphthalene-l-sulfonic acid-5-sulfonate, ethan-1-sulfonic acid-2-sulfonate, and the like), and carboxylate ions (e.g., acetate, ethanoate, propanoate, benzoate, glycerate, lactate, tartrate, glycolate, and the like).
- carboxylate ions e.g., acetate, ethanoate
- Nitrogen atoms can be substituted or unsubstituted as valency permits, and include primary, secondary, tertiary, and quaternary nitrogen atoms.
- the substituent present on a nitrogen atom is a nitrogen protecting group (also referred to as an amino protecting group).
- Nitrogen protecting groups are well known in the art and include those described in detail in Protecting Groups in Organic Synthesis, T. W. Greene and P. G. M. Wuts, 3rd edition, John Wiley & Sons, 1999, incorporated herein by reference.
- Nitrogen protecting groups such as sulfonamide groups include, but are not limited to, p-toluenesulfonamide (Ts), benzenesulfonamide, 2,3,6,-trimethyl-4- methoxybenzenesulfonamide (Mtr), 2,4,6-trimethoxybenzenesulfonamide (Mtb), 2,6-dimethyl-4- methoxybenzenesulfonamide (Pme), 2,3,5,6-tetramethyl-4-methoxybenzenesulfonamide (Mte), 4- methoxybenzenesulfonamide (Mbs), 2,4,6- trimethylbenzenesulfonamide (Mts), 2,6-dimethoxy-4- methylbenzenesulfonamide (iMds), 2,2,5,7,8-pentamethylchroman-6-sulfonamide (Pmc), methan
- nitrogen protecting groups include, but are not limited to, phenothiazinyl-(10)-acyl derivative, N’-p-toluenesulfonylaminoacyl derivative, N’ -phenylaminothioacyl derivative, N- benzoylphenylalanyl derivative, N-acetylmethionine derivative, 4,5-diphenyl-3-oxazolin-2-one, N- phthalimide, N-dithiasuccinimide (Dts), N-2,3-diphenylmaleimide, N-2,5-dimethylpyrrole, N-1,1,4,4- tetramethyldisilylazacyclopentane adduct (STABASE), 5-substituted 1,3-dimethyl-1,3,5- triazacyclohexan-2-one, 5-substituted 1,3-dibenzyl-1,3,5-triazacyclohexan-2-one, 1- substituted 3,5-
- the substituent present on an oxygen atom is an oxygen protecting group (also referred to as a hydroxyl protecting group).
- Oxygen protecting groups are well known in the art and include those described in detail in Protecting Groups in Organic Synthesis, T. W. Greene and P. G. M. Wuts, 3rd edition, John Wiley & Sons, 1999, incorporated herein by reference.
- oxygen protecting groups include, but are not limited to, methyl, methoxylmethyl (MOM), methylthiomethyl (MTM), t-butylthiomethyl, (phenyldimethylsilyl)methoxymethyl (SMOM), benzyloxymethyl (BOM), p-methoxybenzyloxymethyl (PMBM), (4-methoxyphenoxy)methyl (p-AOM), guaiacolmethyl (GUM), t-butoxymethyl, 4-pentenyloxymethyl (POM), siloxymethyl, 2- methoxyethoxymethyl (MEM), 2,2,2-trichloroethoxymethyl, bis(2-chloroethoxy)methyl, 2- (trimethylsilyl)ethoxymethyl (SEMOR), tetrahydropyranyl (THP), 3-bromotetrahydropyranyl, tetrahydrothiopyranyl, 1-methoxycyclohexyl, 4- methoxytetrahydropyranyl (MT), methyl,
- the substituent present on a sulfur atom is a sulfur protecting group (also referred to as a thiol protecting group).
- Sulfur protecting groups are well known in the art and include those described in detail in Protecting Groups in Organic Synthesis, T. W. Greene and P. G. M. Wuts, 3rd edition, John Wiley & Sons, 1999, incorporated herein by reference.
- Exemplary sulfur protecting groups include, but are not limited to, alkyl, benzyl, p- methoxybenzyl, 2,4,6-trimethylbenzyl, 2,4,6-trimethoxybenzyl, o-hydroxybenzyl, p-hydroxybenzyl, o- acetoxybenzyl, p-acetoxybenzyl, p-nitrobenzyl, 4-picolyl, 2-quinolinylmethyl, 2-picolyl N-oxido, 9- anthrylmethyl, 9-fluorenylmethyl, xanthenyl, ferrocenylmethyl, diphenylmethyl, bis(4- methoxyphenyl)methyl, 5-dibenzosuberyl, triphenylmethyl, diphenyl-4-pyridylmethyl, phenyl, 2,4- dinitrophenyl, t-butyl, 1-adamantyl, methoxymethyl (MOM), isobutoxymethyl, benzyloxymethyl,
- Liposomal-based vehicles are considered as an attractive carrier for therapeutic agents and remain subject to continued development efforts. While liposomal-based vehicles that comprise certain lipid components have shown promising results with regard to encapsulation, stability and site localization, there remains a great need for improvement of liposomal-based delivery systems. For example, a significant drawback of liposomal delivery systems relates to the construction of liposomes that have sufficient cell culture or in vivo stability to reach desired target cells and/or intracellular compartments, and the ability of such liposomal delivery systems to efficiently release their encapsulated materials to such target cells.
- cationic lipids that are effective for intramuscular delivery of mRNA (e.g., for preventing or treating Flu, acne, chlamydia, or a disease or disorder induced by infection with P. gingivalis).
- improved lipids compounds that demonstrate improved pharmacokinetic properties, and which are capable of delivering macromolecules, such as nucleic acids, to a wide variety cell types and tissues with enhanced efficiency.
- novel lipid compounds that are characterized as having improved safety profiles and are capable of efficiently delivering encapsulated nucleic acids and polynucleotides to targeted cells, tissues and organs.
- compounds of the invention as described herein can be characterized as having one or more properties that afford such compounds advantages relative to other similarly classified lipids.
- compounds disclosed herein can allow for the control and tailoring of the properties of liposomal compositions (e.g., lipid nanoparticles) of which they are a component.
- compounds disclosed herein can be characterized by enhanced transfection efficiencies and their ability to provoke specific biological outcomes. Such outcomes can include, for example enhanced cellular uptake, endosomal/lysosomal disruption capabilities and/or promoting the release of encapsulated materials (e.g., polynucleotides) intracellularly.
- the compounds disclosed herein can also be characterized by achieving high levels of peptide or protein expression when delivering mRNA encoding for said peptide or protein by intravenous, intrathecal, intramuscular, intranasal, sublingual, or by pulmonary delivery, optionally through nebulization.
- the compounds disclosed herein can also be characterized by achieving high Hemagglutination Inhibition (HAI) when delivering mRNA encoding an influenza antigen by intramuscular delivery.
- HAI Hemagglutination Inhibition
- the compounds disclosed herein have advantageous pharmacokinetic properties, biodistribution, and efficiency.
- the cationic lipids of the present invention have cleavable groups such as ester groups. These cleavable groups (e.g.
- the cationic lipids of the present invention are capable of highly effective in vivo intramuscular delivery of therapeutic agents and vaccines (e.g., for treating or preventing Flu, acne, chlamydia, or a disease or disorder induced by infection with P. gingivalis). It is also contemplated that lipid nanoparticles comprising the cationic lipids of the present invention are capable of highly effective in vivo delivery while maintaining a favorable safety profile.
- lipid nanoparticles comprising the cationic lipids of the present invention may exhibit improved degradation in vivo.
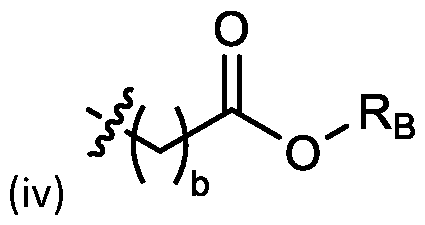
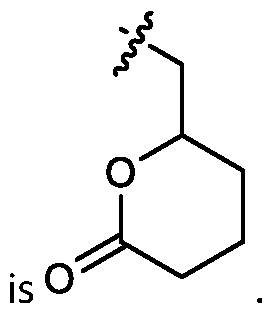
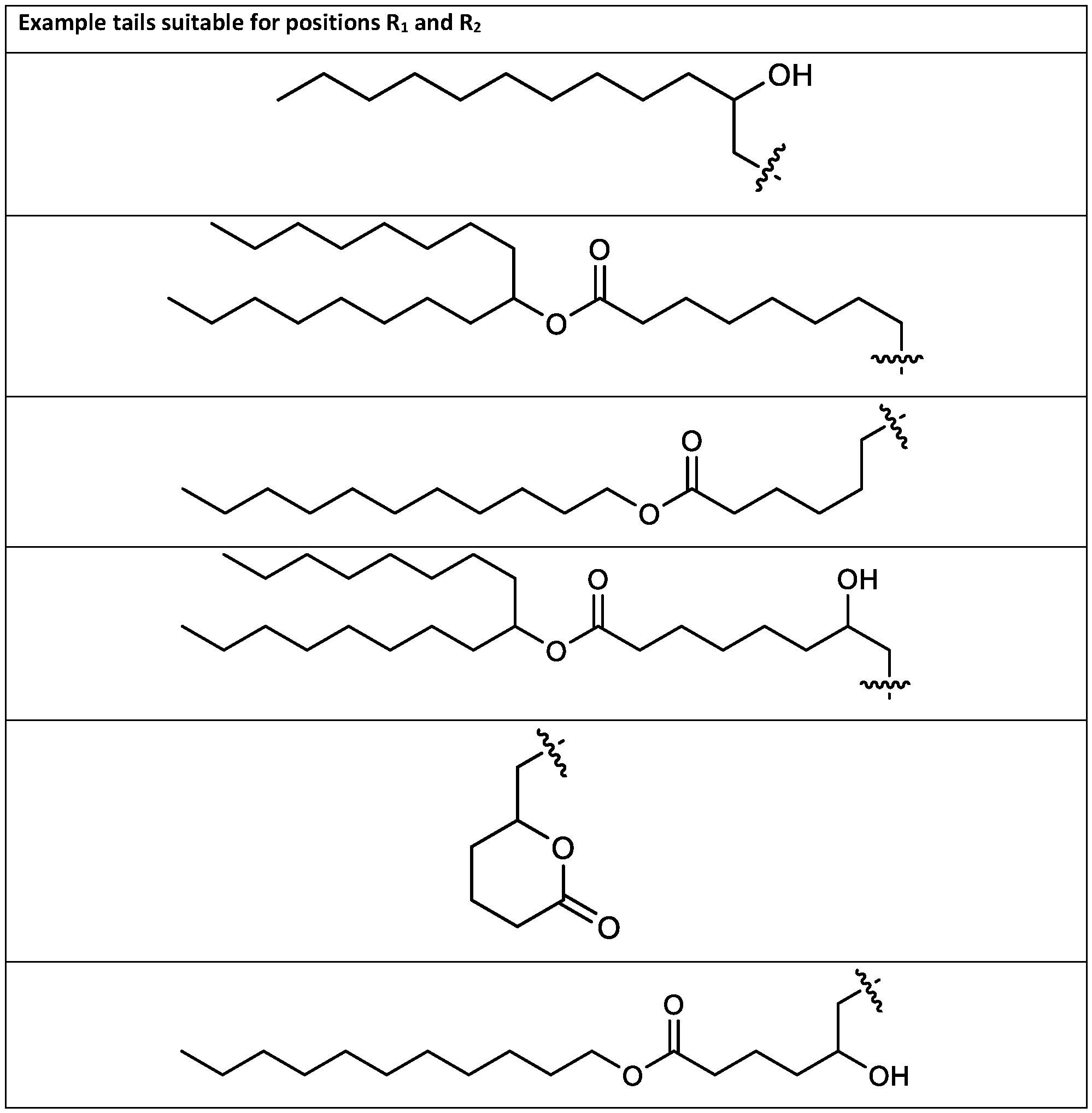
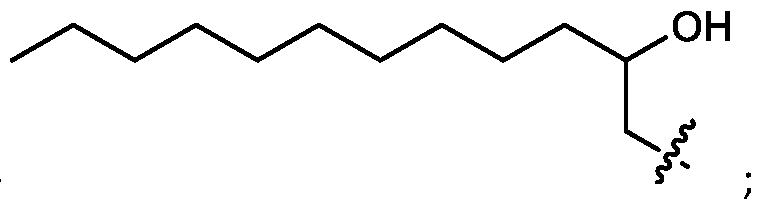
- the cationic lipids include compounds having a structure according to Formula (I’): or a pharmaceutically acceptable salt thereof, wherein: a is 3 or 4; each of R1, R2, R3, and R4 is independently selected from: (i) optionally substituted C5-C30 alkyl, and optionally substituted C5-C30 alkenyl; (ii) optionally substituted C 1-5 alkyl which is substituted with an optionally substituted heterocyclyl moiety; (iii) , wherein each R A is independently selected from optionally substituted C3-C28 alkyl, optionally substituted C3-C28 alkenyl, and -W 1 -C(O)-O-X 1 , wherein each W 1 is independently selected from optionally substituted C 3 -C
- the cationic lipids include compounds having a structure according to Formula (I): or a pharmaceutically acceptable salt thereof, wherein: a is 3 or 4; each of R1, R2, R3, and R4 is independently selected from: (i) optionally substituted C5-C30 alkyl, and optionally substituted C5-C30 alkenyl; (ii) optionally substituted C 1-5 alkyl which is substituted with an optionally substituted heterocyclyl moiety; , wherein each R A is independently selected from optionally substituted C 3 -C 28 alkyl, optionally substituted C 3 -C 28 alkenyl, and -W 1 -C(O)-O-X 1 , wherein each W 1 is independently selected from optionally substituted C 3 -C 5 a lkylene and optionally substituted C 3 -C 5 alkenylene, and each X1 is independently selected from optionally substituted C5-C30 alkyl and optionally substituted C5-C30
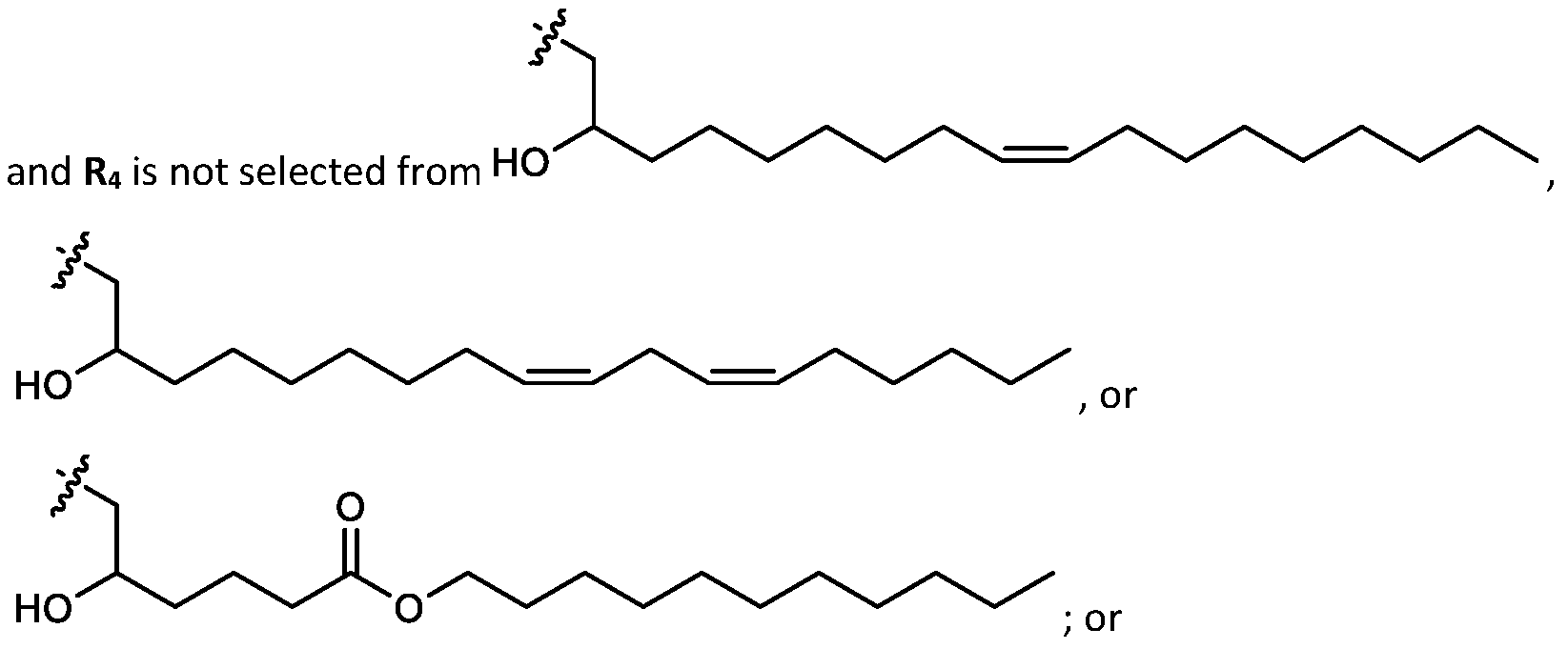
- the cationic lipids do not include the following compound: . [099] In embodiments, the cationic lipids do not include the following compound: . [0100] In embodiments, the cationic lipids do not include the following compound: . [0101] In embodiments, the cationic lipids do not include the following compounds: ,
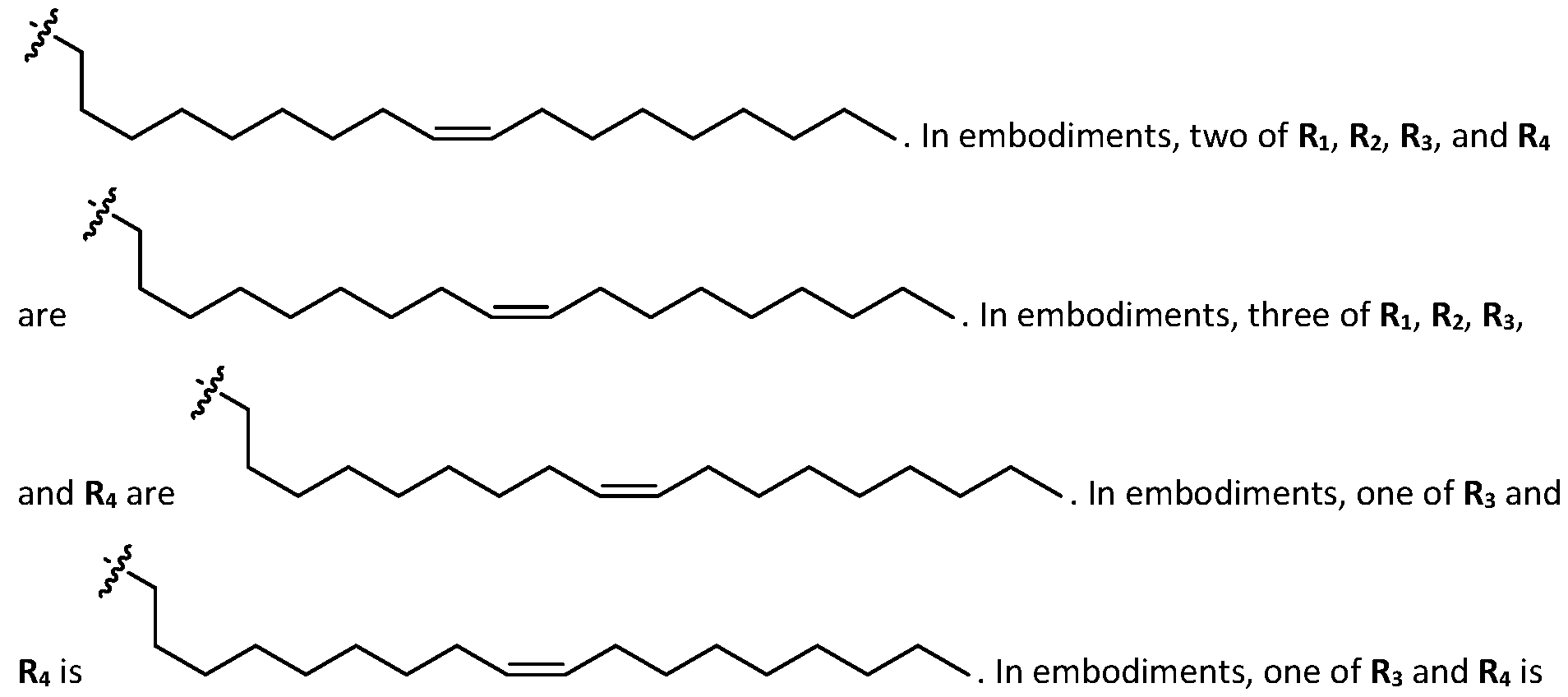
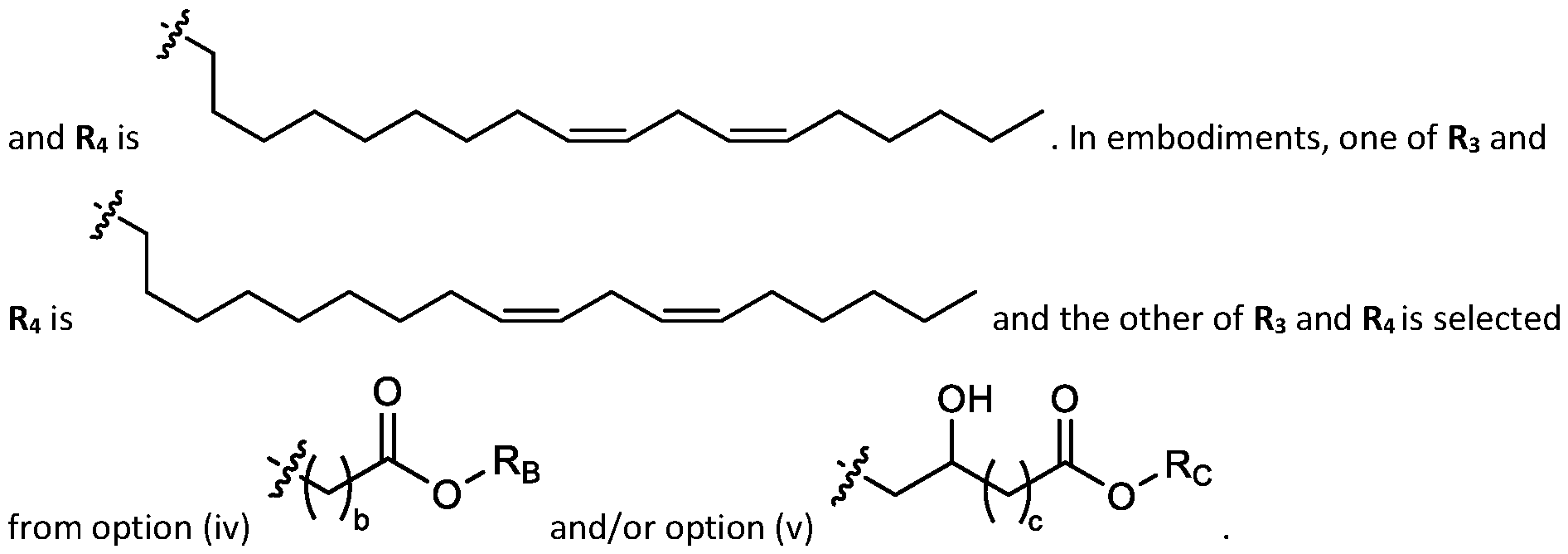
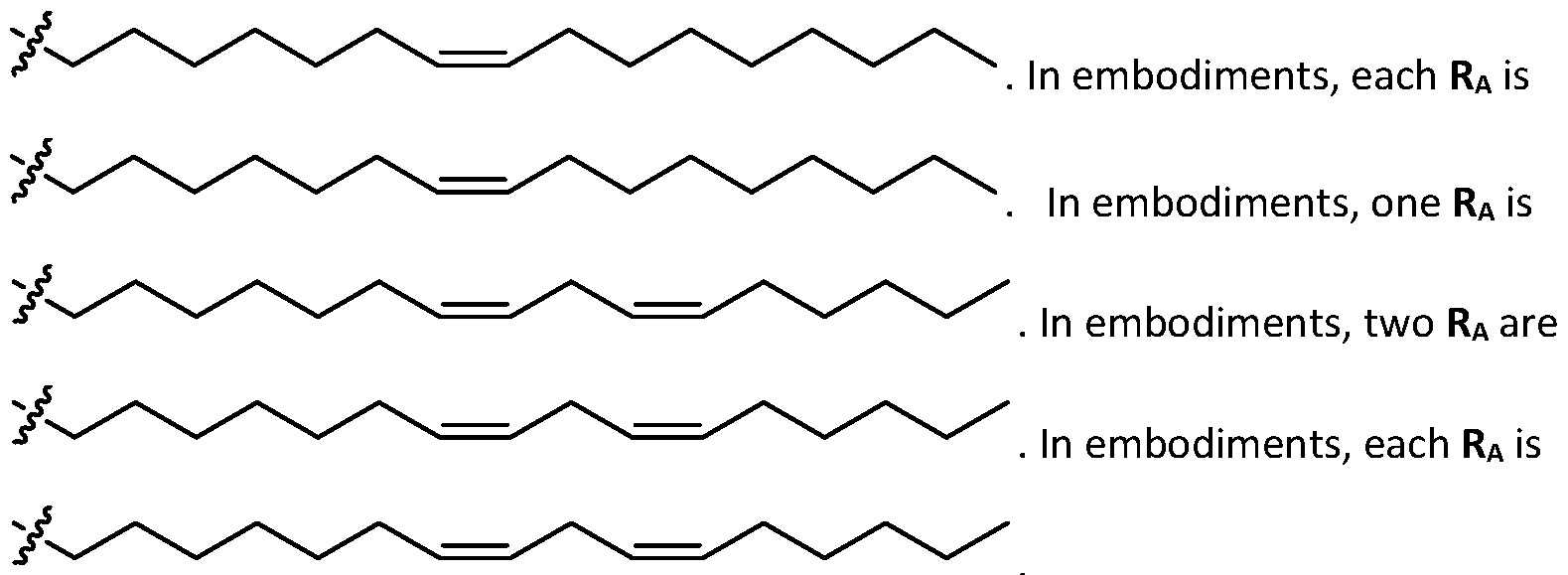
- a is 3. In embodiments, a is 4. [0103] In embodiments, b is 5. In embodiments, b is 6. In embodiments, b is 7. In embodiments, each b is independently selected from 5 or 6. In embodiments, each b is independently selected from 6 or 7. In one embodiment, each b is independently selected from 5 or 7. [0104] In embodiments, d is 2. In embodiments, d is 3. In embodiments, d is 4. [0105] In embodiments, one of R1, R2, R3, and R4 is selected from option option (v) . In embodiments, two of R1, R2, R3, and R4 are independently selected from option (iv) and/or option (v) .
- R1, R2, R3, and R4 are independently selected from option (iv) r option (v) . In embodiments, all of R 1 , R 2 , R 3, and R4 are independently selected from option r option (v) . [0106] In embodiments, R1 and R2 are independently selected from option and/or option (v) , and R3 and R4 are not selected from option (iv) . In embodiments, R 3 and R 4 are independently selected from option r option (v) [0107] In embodiments, R1 and R2 are not identical to one another and R3 and R4 are identical to one another. [0108] In embodiments, R1 and R2 are identical to one another and R3 and R4 are not identical to one another.
- R 1 and R 2 are not identical to one another and R 3 and R 4 are not identical to one another. [0110] In embodiments, none of R1, R2, R3, and R4 are identical. [0111] In embodiments, one of R1, R2, R3, and R4 is selected from optionally substituted C5-C30 alkyl. In embodiments, two of R1, R2, R3, and R4 are independently selected from optionally substituted C5- C30 alkyl. In embodiments, three of R1, R2, R3, and R4 are independently selected from optionally substituted C 5 -C 30 alkyl. In embodiments, R 1 and R 2 are independently selected from optionally substituted C 5 -C 30 alkyl.
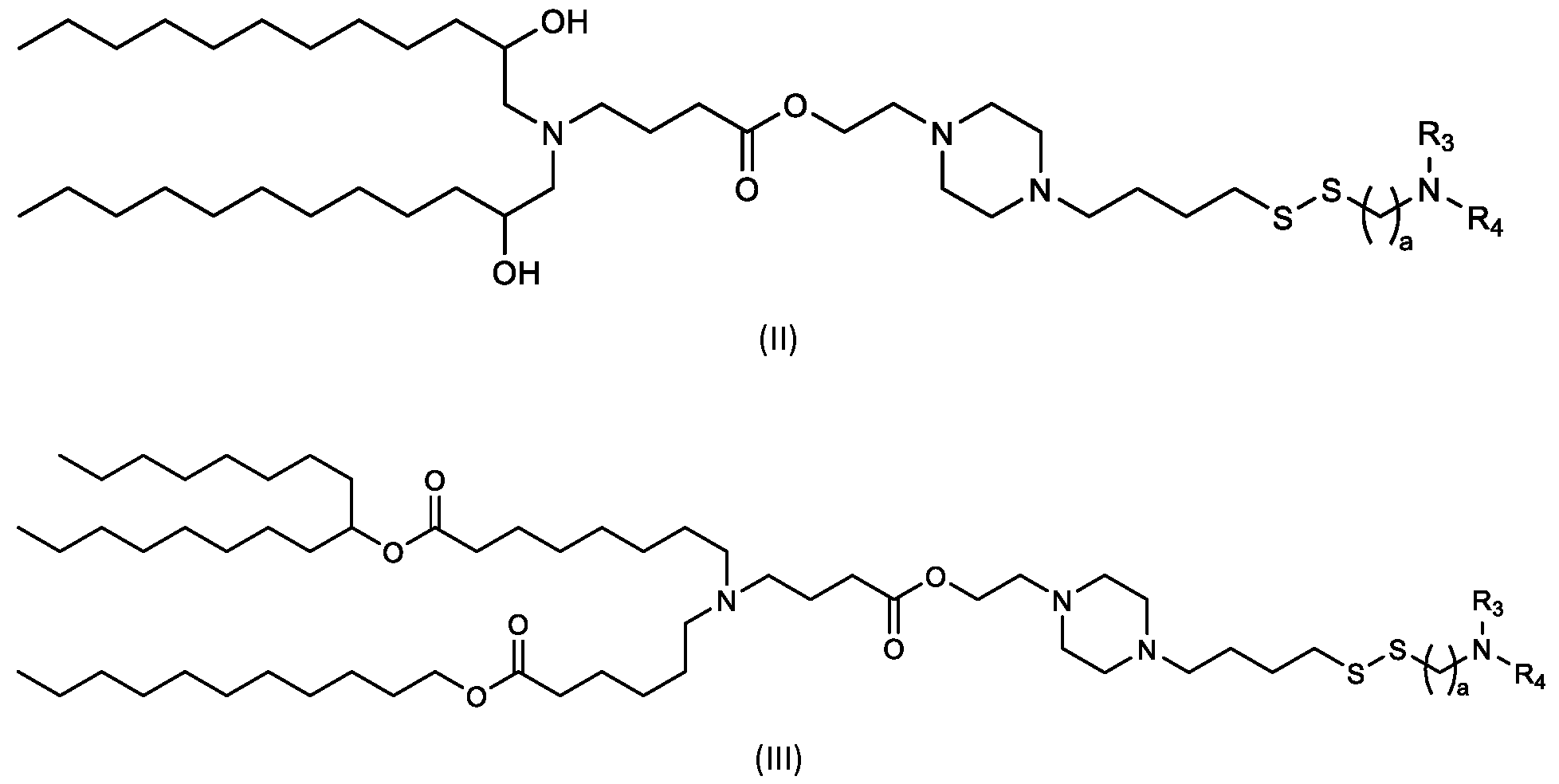
- the cationic lipids include compounds having a structure according to Formula (II’): or a pharmaceutically acceptable salt thereof, wherein a, d, R 3 and R 4 are as defined herein, for example wherein a is 4.
- the cationic lipids of the present invention include compounds having a structure according to Formula (II): (II) or a pharmaceutically acceptable salt thereof, wherein a, R 3 and R 4 are as defined herein, for example wherein a is 4.
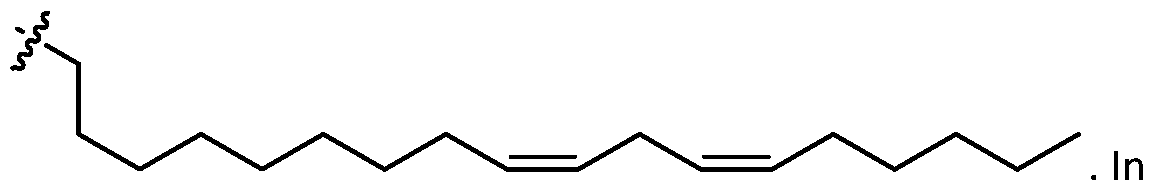
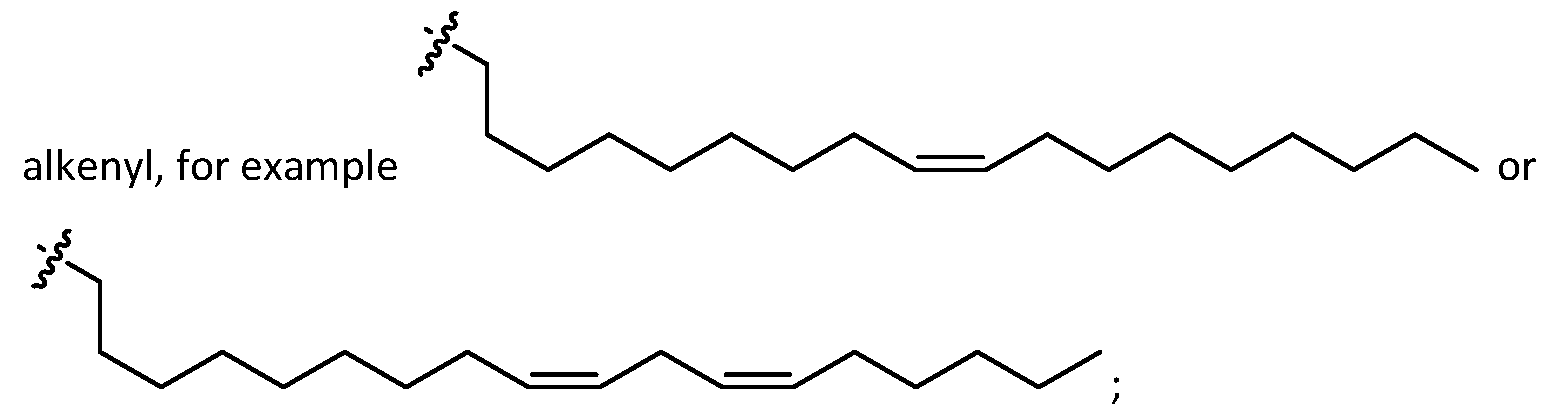
- one of R1, R2, R3, and R4 is selected from optionally substituted C5-C30 alkenyl.
- R1, R2, R3, and R4 are independently selected from optionally substituted C5-C30 alkenyl. In embodiments, three of R1, R2, R3, and R4 are independently selected from optionally substituted C 5 -C 30 alkenyl. [0117] In embodiments, one of R 3 and R 4 is selected from optionally substituted C 5 -C 30 alkenyl. In embodiments, one of R3 and R4 is selected from optionally substituted C5-C30 alkenyl and the other of R 3 and R 4 is selected from option r option (v) . [0118] In embodiments, one of R 1 , R 2 , R 3 , and R 4 is optionally substituted C 5 -C 18 alkenyl.
- two of R 1 , R 2 , R 3 , and R 4 are independently selected from optionally substituted C 5 -C 18 alkenyl. In embodiments, three of R 1 , R 2 , R 3 , and R 4 are independently selected from optionally substituted C5-C18 alkenyl. In embodiments, one of R3 and R4 is optionally substituted C5-C30 alkenyl. In embodiments, one of R3 and R4 is optionally substituted C5-C18 alkenyl and the other of R3 and R4 is selected from option r option (v) .
- one of R1, R2, R3, and R4 is [0120] In embodiments, one of R 1 , R 2 , R 3 , and R 4 is a e . e o e s, ee o 1 , 2 , 3 , and R4 are . embodiments, one of R3 [0121] In embodiments, two of R1, R2, R3, and R4 are independently selected from optionally substituted C5-C30 alkyl and one of R1, R2, R3, and R4 is selected from optionally substituted C5-C30 alkenyl.
- R1 and R2 are independently selected from optionally substituted C5-C30 alkyl and one of R 3 and R 4 is selected from optionally substituted C 5 -C 30 alkenyl.
- two of R 1 , R 2 , R 3 , and R 4 are independently selected from optionally substituted C 5 -C 12 alkyl and one of R 1 , R2, R3, and R4 is selected from optionally substituted C5-C18 alkenyl.
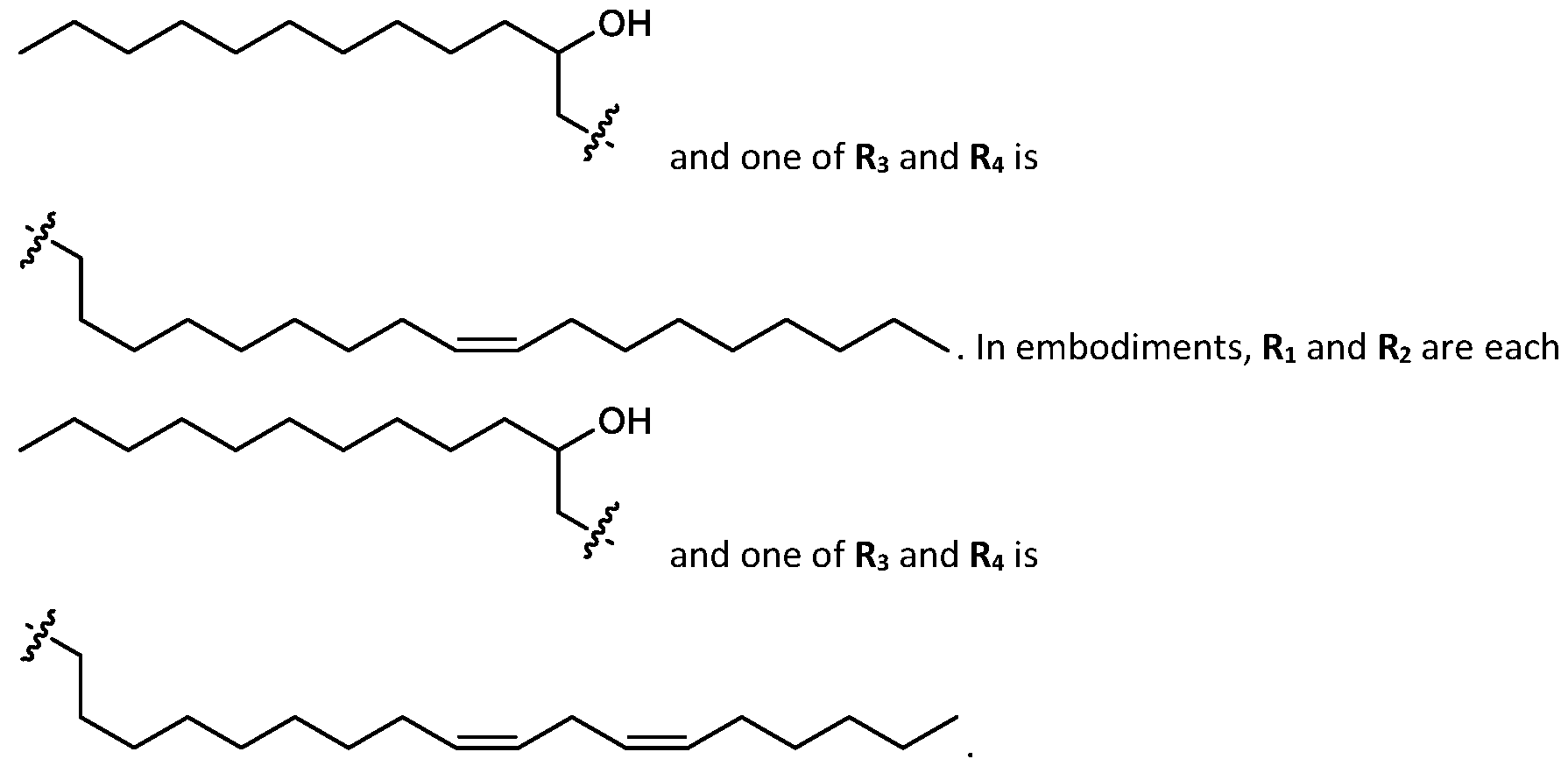
- R1 and R2 are independently selected from optionally substituted C5-C12 alkyl and one of R3 and R4 is selected from optionally substituted C5-C18 alkenyl. [0122]
- R1 and R2 are each and one of R3 and R4 is selected from optionally substituted C5-C18 alkenyl.
- R1 and R2 are each and one of R3 and R4 is selected from optionally substituted C5-C18 alkenyl.
- R1 and R2 are each and one of R3 and R4 is selected from
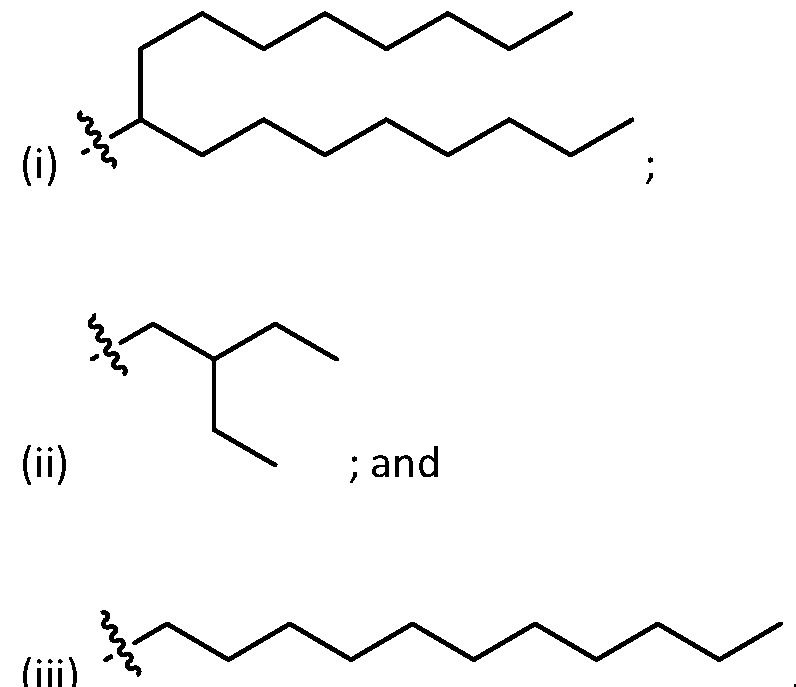
- R 1 , R 2 , R 3 , and R 4 are independently selected from , wherein each RA is independently selected from optionally substituted C3-C28 alkyl, optionally substituted C3-C28 alkenyl, and -W 1 -C(O)-O-X 1 , wherein each W 1 is independently selected from optionally substituted C3-C5 alkylene and optionally substituted C3-C5 alkenylene, and each X 1 is independently selected from optionally substituted C5-C30 alkyl and optionally substituted C5-C30 alkenyl.
- R1, R2, R3, and R4 is selected from as defined above.
- R , R , and R are independently 1 2 3 4 selected from as defined above. In embodiments, three of R1, R2, R3, and R4 are independently selected from as defined above. In embodiments, R 1 and R2 are independently selected from as defined above. In embodiments, R and R are independently selec 1 2 ted from as defined above and one of R3 and R4 is selected from optionally substituted C5-C30 alkenyl. In embodiments, R1 and R2 are independently selected from as defined above and one of R3 and R4 is selected from optionally substituted C5-C18 alkenyl. In embodiments, R1 and R2 are independently selected .
- R1 and R2 are independently selected from as defined above and one of R3 and R4 is . In embodiments, R3 and R4 are independently selected from . [0125] In embodiments, one R A is selected from optionally substituted C 3 -C 28 alkyl. In embodiments, two R A are independently selected from optionally substituted C 3 -C 28 alkyl. In embodiments, each R A is independently selected from optionally substituted C3-C28 alkyl. In embodiments, one RA is selected from optionally substituted C3-C10 alkyl. In embodiments, two RA are independently selected from optionally substituted C3-C10 alkyl. In embodiments, each RA is independently selected from optionally substituted C3-C10 alkyl.
- one RA is . In . In embodiments, each R A is . [0126] In embodiments, one R A is selected from optionally substituted C 3 -C 28 alkenyl. In embodiments, two R A are independently selected from optionally substituted C 3 -C 28 alkenyl. In embodiments, each R A is independently selected from optionally substituted C 3 -C 28 alkenyl. In embodiments, one RA is selected from optionally substituted C3-C16 alkenyl. In embodiments, two RA are independently selected from optionally substituted C3-C16 alkyl. In embodiments, each RA is independently selected from optionally substituted C3-C16 alkyl. In embodiments, one RA is .
- two RA are .
- one R A is selected from -W 1 -C(O)-O-X 1 .
- two R A are independently selected from -W 1 -C(O)-O-X 1 .
- each R A is independently selected from -W 1 -C(O)-O-X 1 .
- one W 1 is selected from optionally substituted C3-C5 alkylene.
- two W 1 are independently selected from optionally substituted C3-C5 alkylene.
- each W 1 is independently selected from optionally substituted C3-C5 alkylene.
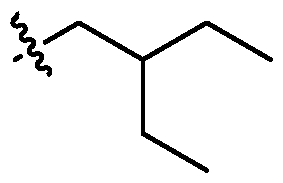
- one W 1 is selected from optionally substituted C3-C5 alkenylene. In embodiments, two W 1 are independently selected from optionally substituted C 3 -C 5 alkenylene. In embodiments, each W 1 is independently selected from optionally substituted C 3 -C 5 alkenylene. [0129] In embodiments, one X 1 is selected from optionally substituted C 5 -C 30 alkyl. In embodiments, two X 1 are independently selected from optionally substituted C5-C30 alkyl. In embodiments, each X 1 is independently selected from optionally substituted C5-C30 alkyl. In embodiments, one X 1 is selected from optionally substituted C6-C17 alkyl.
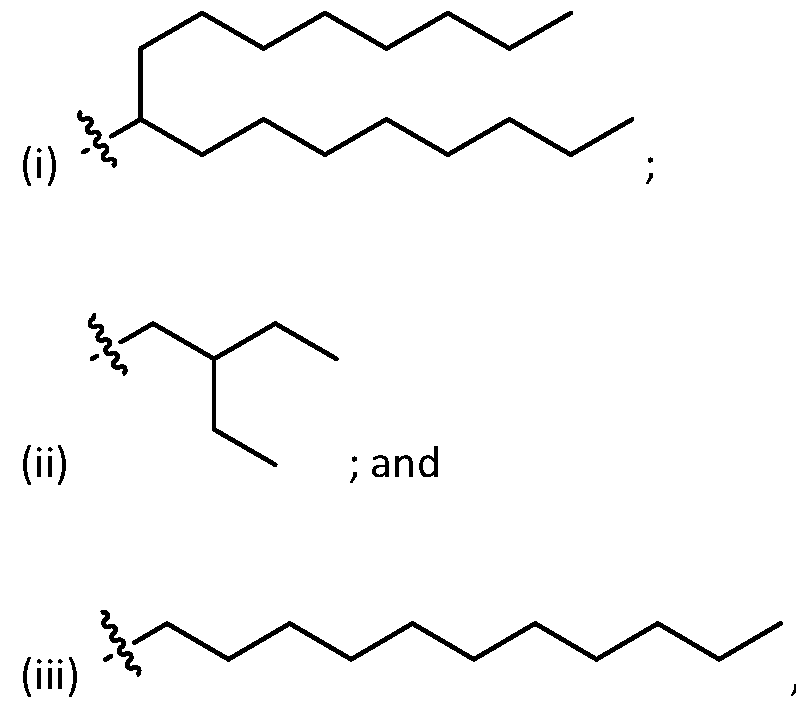
- two X 1 are independently selected from optionally substituted C6-C17 alkyl. In embodiments, each X 1 is independently selected from optionally substituted C 6 -C 17 alkyl. In embodiments, one X 1 is selected from optionally substituted branched C 6 -C 17 alkyl. In embodiments, two X 1 are independently selected from optionally substituted branched C6-C17 alkyl. In embodiments, each X 1 is independently selected from optionally substituted branched C6-C17 alkyl. [0130] In embodiments, one X 1 is . In embodiments, two X 1 are , . [0131] In embodiments, one X 1 is . In embodiments, two X 1 are .
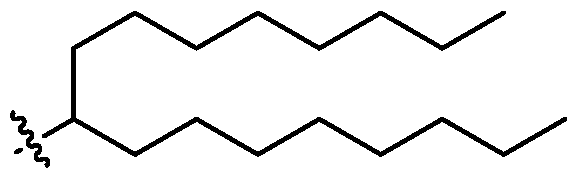
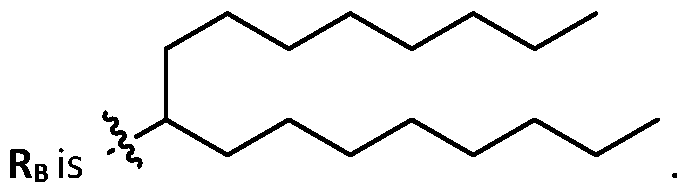
- each X 1 is independently selected from optionally substituted C5-C9 alkenyl. In embodiments, one X 1 is . , . embodiments, each . [0137] In embodiments, one or more of R1, R2, R3, and R4 are independently selected from , wherein each R B is independently selected from optionally substituted C 5 -C 30 alkyl and optionally substituted C 5 -C 30 alkenyl. In embodiments, one of R 1 , R 2 , R 3 , and R 4 is selected from as defined above. In embodiments, two of R1, R2, R3, and R4 are independently selected from as defined above. In embodiments, three of R1, R2, R3, and R4 are independently selected from as defined above.
- each of R1, R2, R3, and R4 are independently selected from as defined above. In embodiments, R1 and R2 are independently selected from as defined above. In embodiments, one of R3 and R4 is selected from as defined above. In embodiments, R3 and R4 are independently selected from as defined above. [0138] In embodiments, one R B is selected from optionally substituted C 5 -C 30 alkyl. In embodiments, two R B are independently selected from optionally substituted C 5 -C 30 alkyl. In embodiments, three R B are independently selected from optionally substituted C 5 -C 30 alkyl. In embodiments, each RB is independently selected from optionally substituted C5-C30 alkyl.
- one RB is selected from optionally substituted C6-C17 alkyl. In embodiments, two RB are independently selected from optionally substituted C6-C17 alkyl. In embodiments, three R B are independently selected from optionally substituted C 6 -C 17 alkyl. In embodiments, each R B is independently selected from optionally substituted C 6 -C 17 alkyl. [0140] In embodiments, one RB is selected from optionally substituted branched C6-C17 alkyl. In embodiments, two RB are independently selected from optionally substituted branched C6-C17 alkyl. In embodiments, three RB are independently selected from optionally substituted branched C6-C17 alkyl.
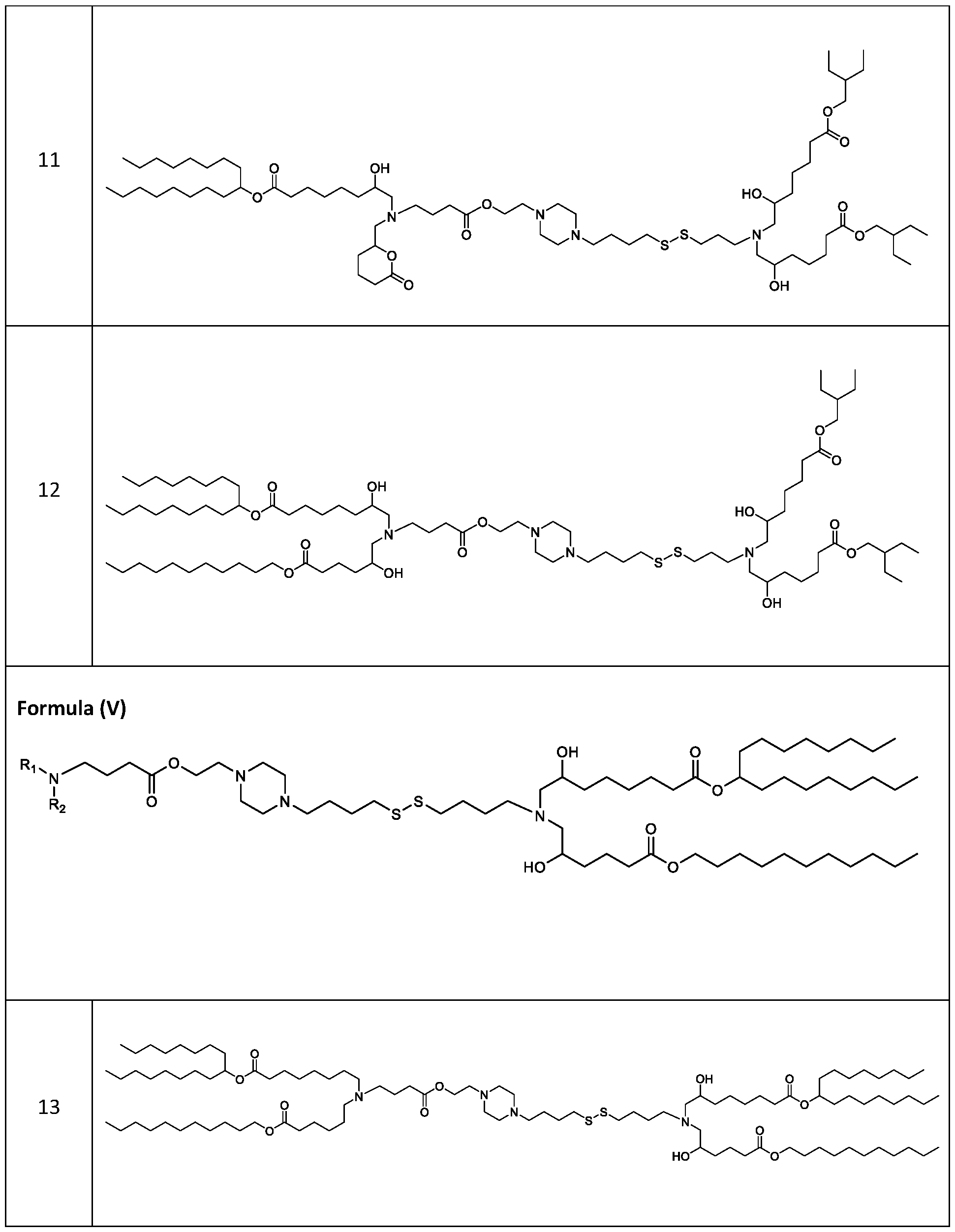
- each RB is independently selected from optionally substituted branched C6- C 17 alkyl. [0141] In embodiments, one RB is . In embodiments, two RB are embodiments, each . [0142] In embodiments, one RB is . In embodiments, two RB are . In embodiments, three RB are . In embodiments, each RB is . [0143] In embodiments, one R B is . In embodiments, two R B . [0144] In embodiments, the cationic lipids include compounds having a structure according to Formula (III’): or a pharmaceutically ac ed herein.
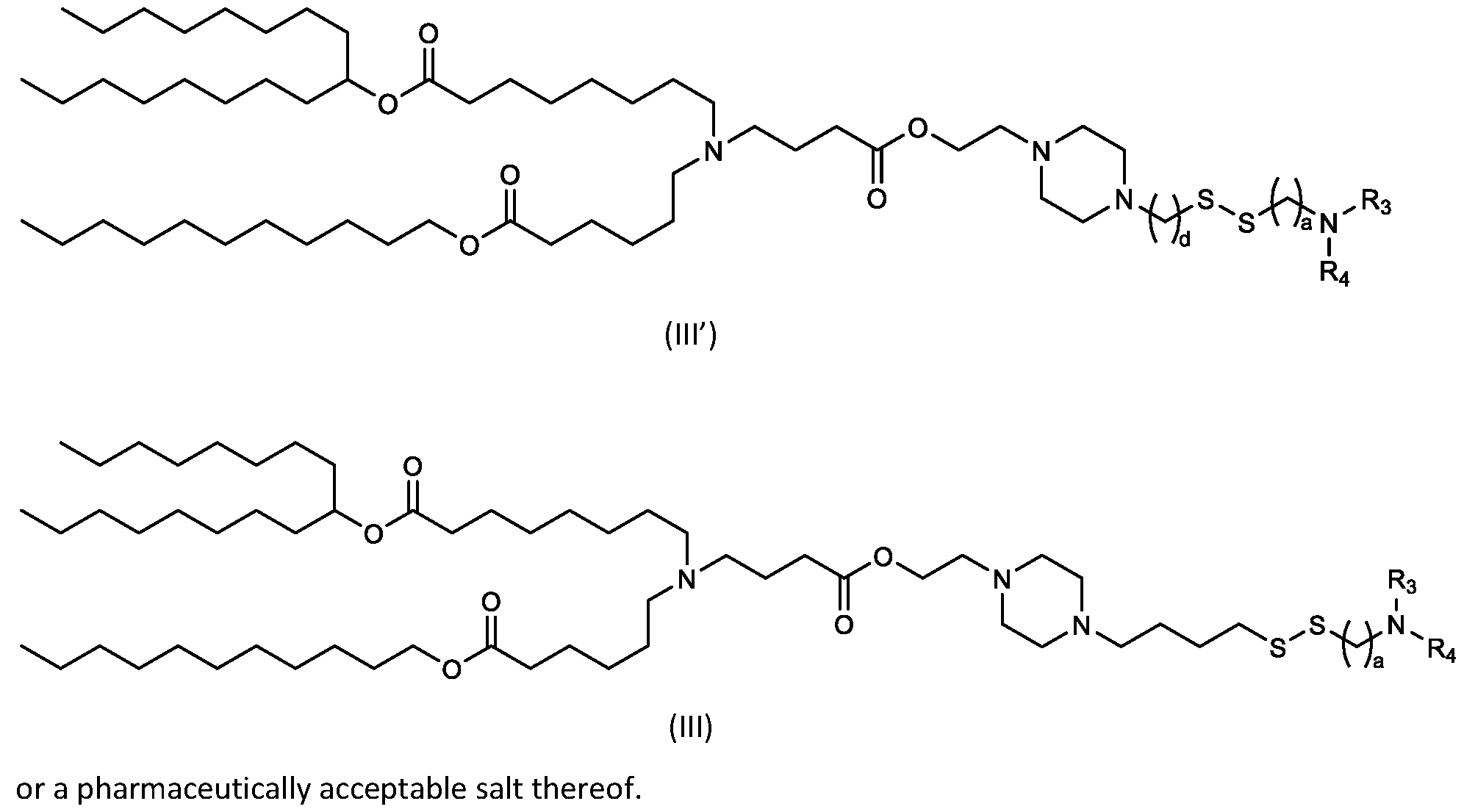
- the cationic lipids include compounds having a structure according to Formula (III): or a pharmaceutically acceptable salt thereof, wherein a, R3 and R4 are as defined herein.
- one R B is selected from optionally substituted C 5 -C 30 alkenyl.
- two R B are independently selected from optionally substituted C 5 -C 30 alkenyl.
- three R B are independently selected from optionally substituted C 5 -C 30 alkenyl.
- each RB is independently selected from optionally substituted C5-C30 alkenyl.
- one RB is selected from optionally substituted C5-C9 alkenyl.
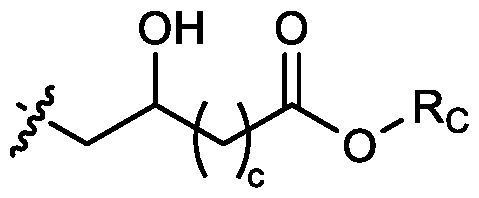
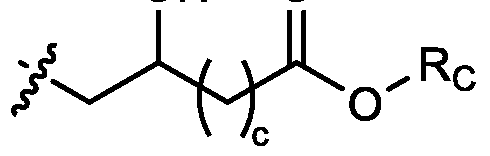
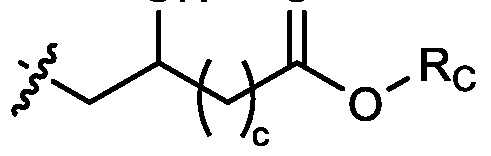
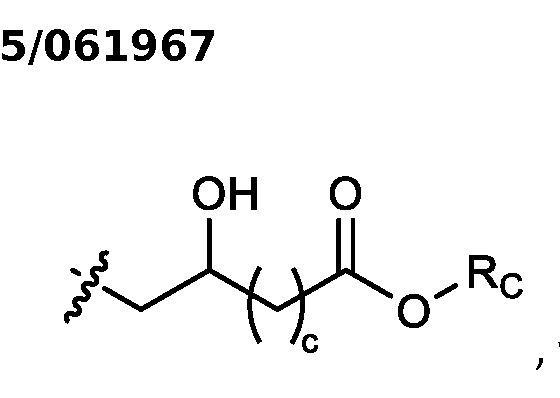
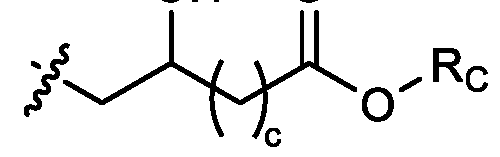
- two RB are independently selected from optionally substituted C5-C9 alkenyl. In embodiments, three RB are independently selected from optionally substituted C5-C9 alkenyl. In embodiments, each R B is independently selected from optionally substituted C 5 -C 9 alkenyl. In embodiments, one R B is . In embodiments, two R B are [0148] In embodiments, one or more of R1, R2, R3, and R4 are independently selected from , wherein each RC independently selected from optionally substituted C5- C30 alkyl and optionally substituted C5-C30 alkenyl. In embodiments, one of R1, R2, R3, and R4 is selected from , as defined above.
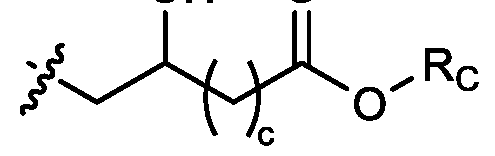
- R1, R2, R3, and R4 are independently selected from , as defined above. In embodiments, three of R1, R2, R3, and R4 are independently selected from , as defined above. In embodiments, each of R 1 , R 2 , R 3 , and R 4 are independently selected from defined above. [0149] In embodiments, R1 and R2 are independently selected from as defined above. [0150] In embodiments, one of R3 and R4 is selected from as defined above. In embodiments, R3 and R4 are independently selected from as defined above. [0151] In embodiments, one or more c is selected from 3 or 5. In embodiments, one c is selected from 3 or 5. In embodiments, two c are selected from 3 or 5.
- three c are selected from 3 or 5. In embodiments, each c is selected from 3 or 5. In embodiments, each c is 3. In embodiments, each c is 5. [0152] In embodiments, one or more c is 4. In embodiments, one c is 4. In embodiments, two c are 4. In embodiments, three c are 4. In embodiments, each c is 4. [0153] In embodiments, one R C is selected from optionally substituted C 5 -C 30 alkyl. In embodiments, two R C are independently selected from optionally substituted C 5 -C 30 alkyl. In embodiments, three R C are independently selected from optionally substituted C 5 -C 30 alkyl.
- each RC is independently selected from optionally substituted C5-C30 alkyl.
- one RC is selected from optionally substituted C6-C17 alkyl.
- two RC are independently selected from optionally substituted C6-C17 alkyl.
- three RC are independently selected from optionally substituted C6-C17 alkyl.
- each R C is independently selected from optionally substituted C 6 -C 17 alkyl.
- one R C is selected from optionally substituted branched C 6 -C 17 alkyl.
- two R C are independently selected from optionally substituted branched C 6 -C 17 alkyl.
- three RC are independently selected from optionally substituted branched C6-C17 alkyl.
- each RC is independently selected from optionally substituted branched C6- C17 alkyl.
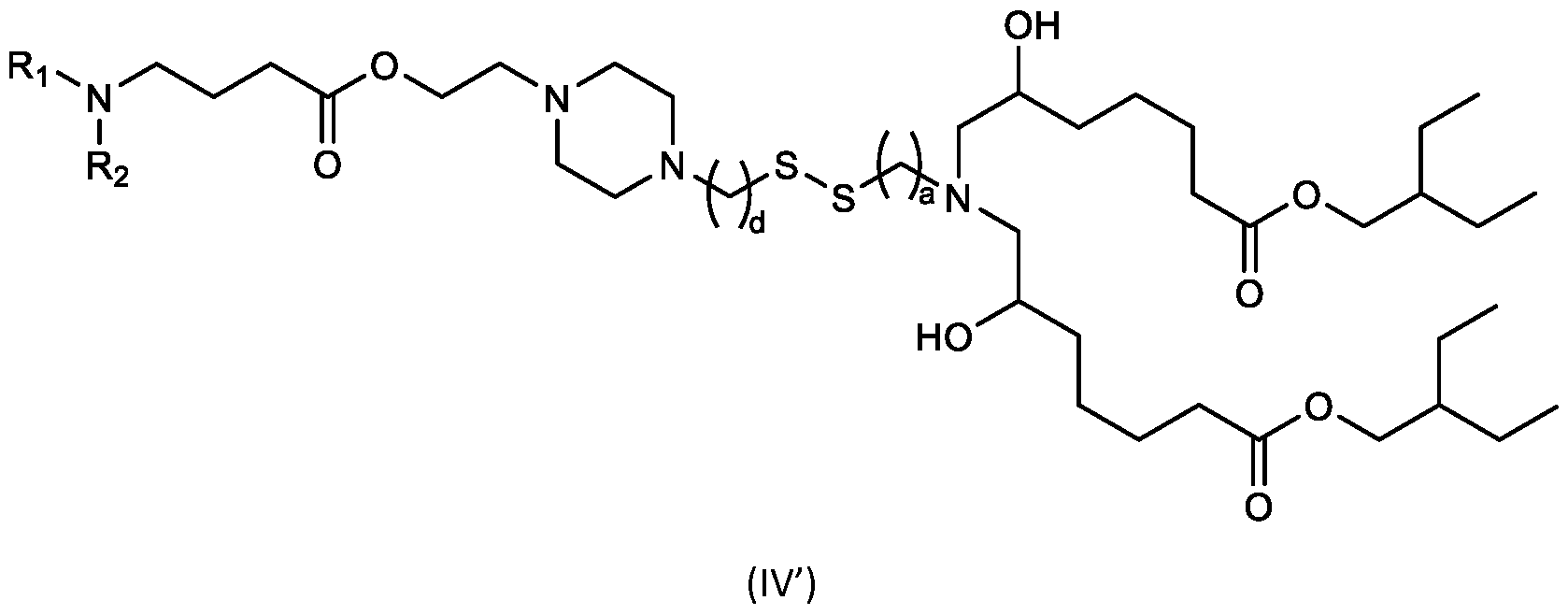
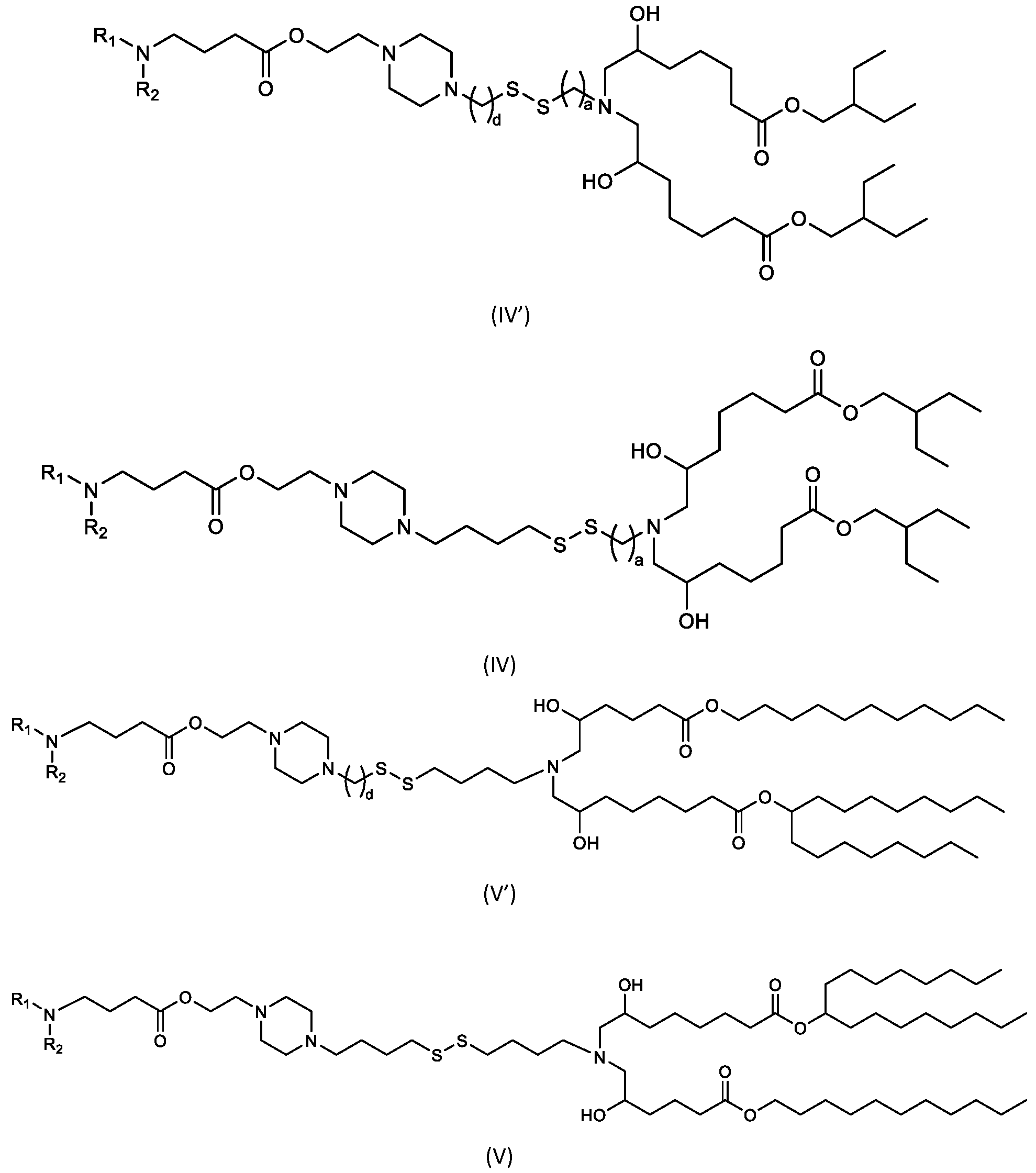
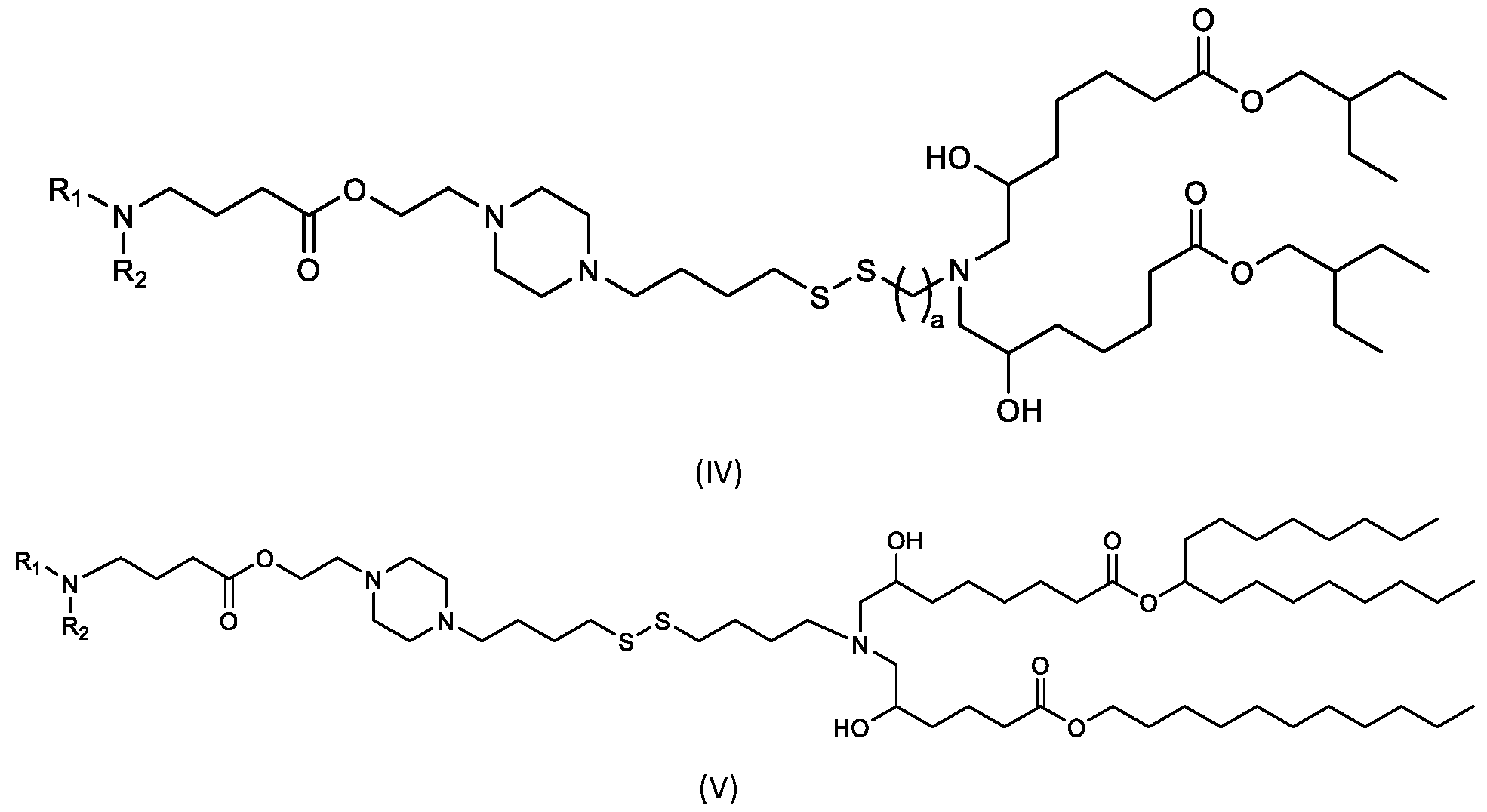
- the cationic lipids include compounds having a structure according to Formula (IV’): or a pharmaceutically acceptable salt thereof, wherein a, d, R 1 and R 2 are as defined herein, for example wherein a is 3.
- the cationic lipids of the present invention include compounds having a structure according to Formula (IV):
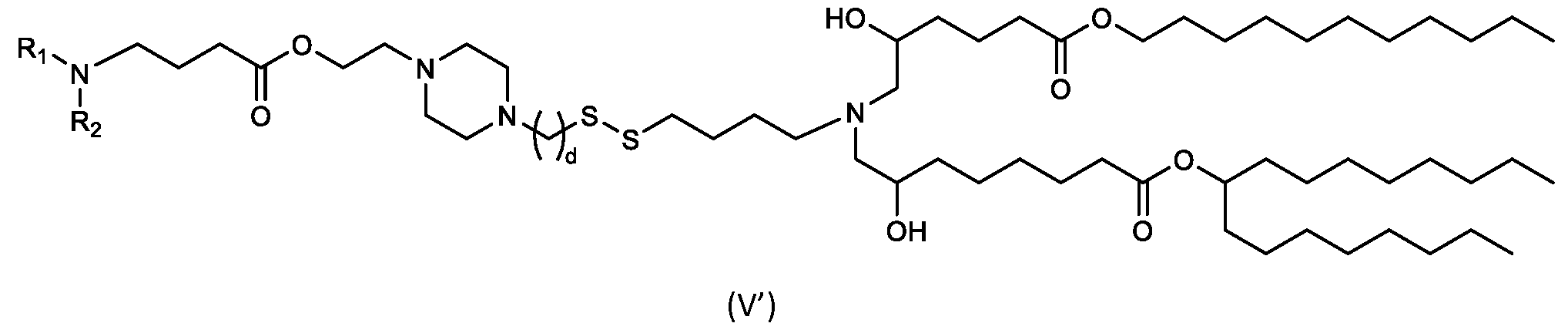
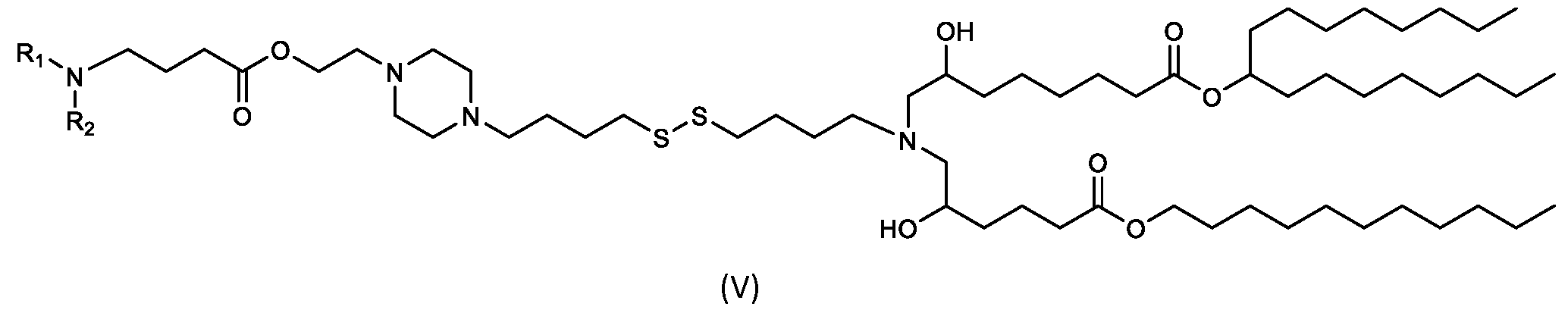
- the cationic lipids of the present invention include compounds having a structure according to Formula (V’): or a pharmaceutically acceptable salt thereof, wherein d, R 1 and R 2 are as defined herein.
- the cationic lipids of the present invention include compounds having a structure according to Formula (V): or a pharmaceutically acceptable salt thereof, wherein R1 and R2 are as defined herein.
- one R C is selected from optionally substituted C 5 -C 30 alkenyl.
- two R C are independently selected from optionally substituted C 5 -C 30 alkenyl. In embodiments, three RC are independently selected from optionally substituted C5-C30 alkenyl. In embodiments, each RC is independently selected from optionally substituted C5-C30 alkenyl. [0164] In embodiments, one RC is selected from optionally substituted C5-C9 alkenyl. In embodiments, two RC are independently selected from optionally substituted C5-C9 alkenyl. In embodiments, three RC are independently selected from optionally substituted C5-C9 alkenyl. In embodiments, each R C is independently selected from optionally substituted C 5 C 9 alkenyl. [0165] In embodiments, one R C is .
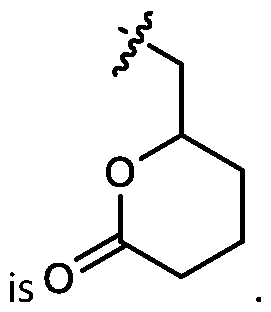
- R1, R2, R3, and R4 is independently selected from optionally substituted C1-C 5 alkyl which is substituted with an optionally substituted heterocyclyl moiety.
- one of R 1 , R 2 , R 3 , and R 4 is selected from optionally substituted C 1 -C 5 alkyl which is substituted with an optionally substituted heterocyclyl moiety.
- two of R 1 , R 2 , R 3 , and R 4 are independently selected from optionally substituted C 1 -C 5 alkyl which is substituted with an optionally substituted heterocyclyl moiety.
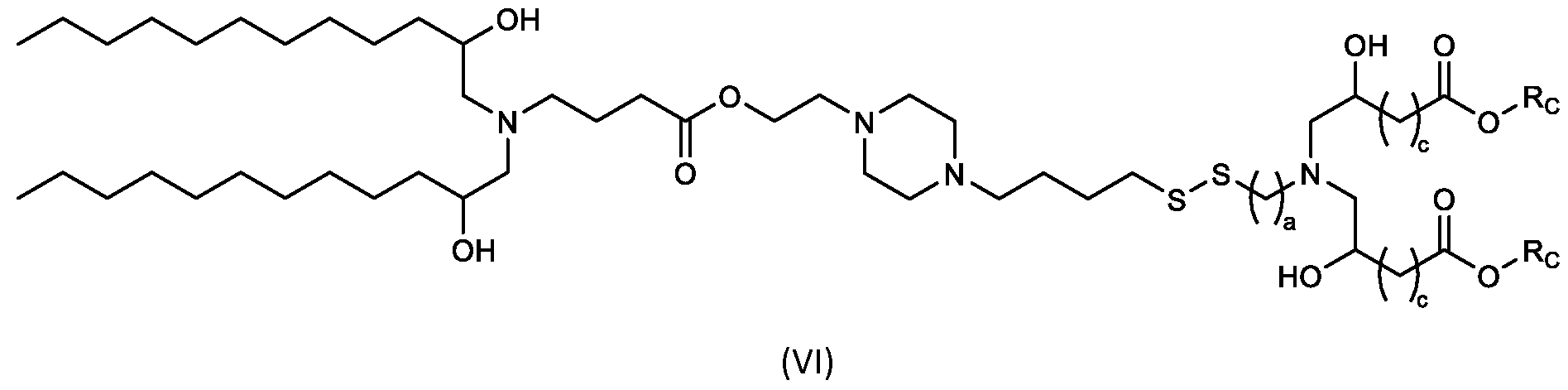
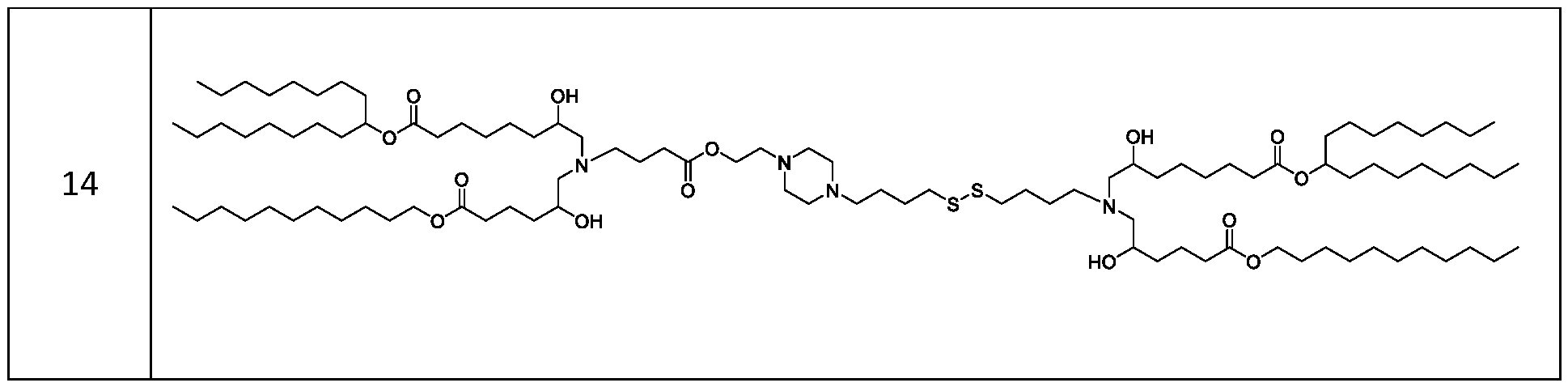
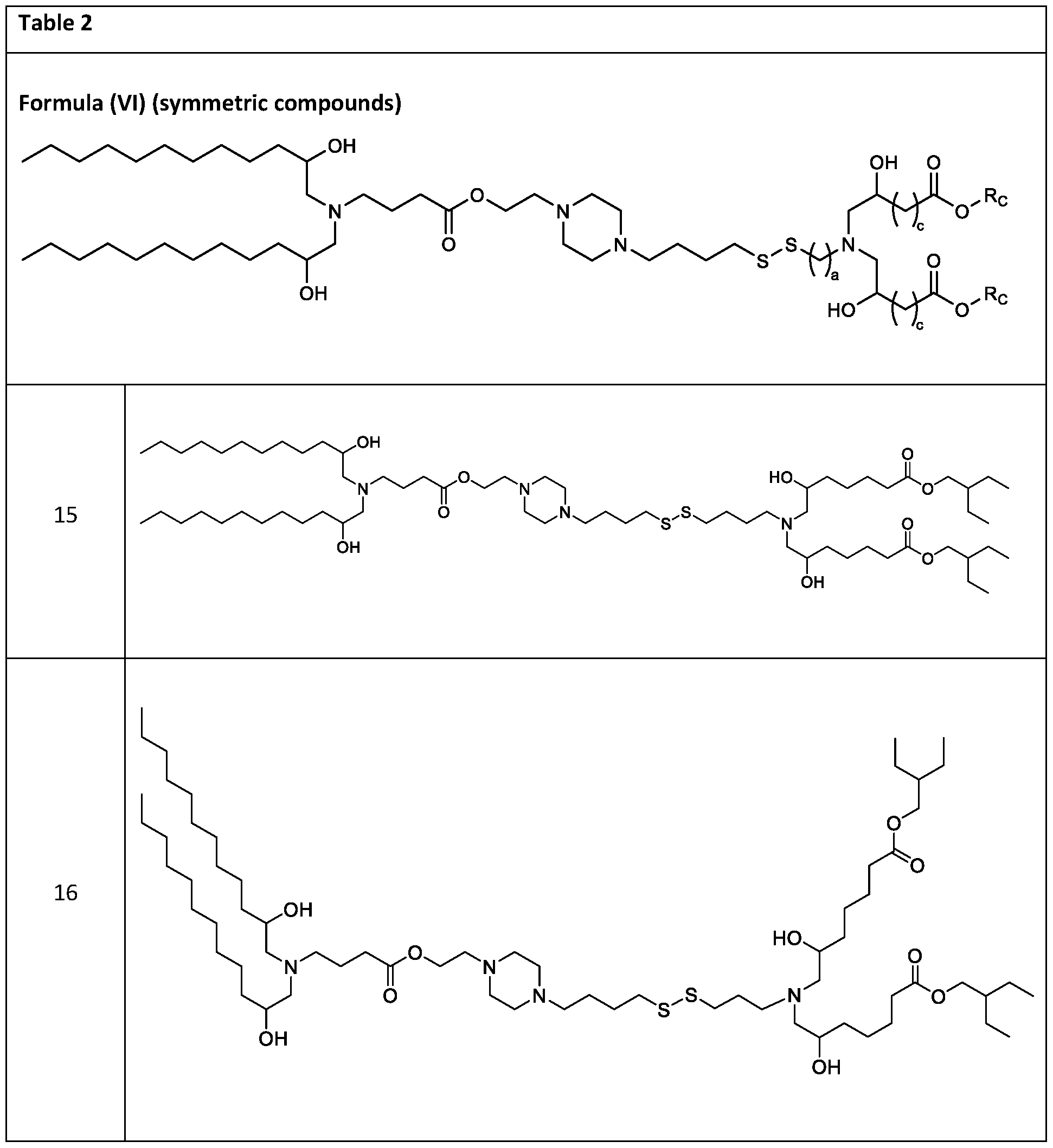
- the cationic lipids include compounds having a structure according to Formula (VI’): or a pharmaceutically acceptable salt thereof, wherein: a is 3 or 4; d is 2, 3, or 4; each c is independently selected from 3 and 4; and each RC is independently selected from .
- this composition is a lipid nanoparticle, optionally a liposome.
- the one or more cationic lipid(s) constitute(s) about 30 mol %-60 mol % of the lipid nanoparticle. In embodiments, the one or more cationic lipid(s) constitute(s) about 35 mol %-55 mol % of the lipid nanoparticle. In embodiments, the one or more cationic lipid(s) constitute(s) about 40 mol %-50 mol % of the lipid nanoparticle. [0177] In embodiments, the one or more non-cationic lipid(s) constitute(s) about 10 mol%-50 mol% of the lipid nanoparticle.
- the lipid nanoparticles have an encapsulation percentage for mRNA of at least 50%. In embodiments, the lipid nanoparticles have an encapsulation percentage for mRNA of at least 55%. In embodiments, the lipid nanoparticles have an encapsulation percentage for mRNA of at least 60%. In embodiments, the lipid nanoparticles have an encapsulation percentage for mRNA of at least 65%. In embodiments, the lipid nanoparticles have an encapsulation percentage for mRNA of at least 70%.
- the composition of the present invention is for use in therapy.
- the composition of the present invention is for use in a method of treating or preventing a disease amenable to treatment or prevention by the peptide or protein encoded by the mRNA encapsulated within said composition.
- the disease or disorder is caused by a viral infection.
- the disease or disorder is influenza.
- the disease or disorder is caused by a bacterial infection.
- the disease or disorder is chlamydia.
- the disease or disorder is induced by infection with P. gingivalis.
- the disease or disorder is acne.
- any of the compounds (1-18) identified in Table 1 and Table 2 above may be provided in the form of a pharmaceutically acceptable salt and such salts are intended to be encompassed by the present invention.
- the compounds of the invention as described herein can be prepared according to methods known in the art, including the exemplary syntheses of the Examples provided herein.
- Nucleic Acids [0194] The compounds of the invention as described herein can be used to prepare compositions useful for the delivery of nucleic acids. Synthesis of Nucleic Acids [0195] Nucleic acids according to the present invention may be synthesized according to any known methods. For example, mRNAs according to the present invention may be synthesized via in vitro transcription (IVT).
- IVT is typically performed with a linear or circular DNA template containing a promoter, a pool of ribonucleotide triphosphates, a buffer system that may include DTT and magnesium ions, and an appropriate RNA polymerase (e.g., T3, T7, mutated T7 or SP6 RNA polymerase), DNAse I, pyrophosphatase, and/or RNAse inhibitor.
- RNA polymerase e.g., T3, T7, mutated T7 or SP6 RNA polymerase
- a suitable DNA template typically has a promoter, for example a T3, T7, mutated T7 or SP6 promoter, for in vitro transcription, followed by desired nucleotide sequence for desired mRNA and a termination signal.
- Desired mRNA sequence(s) according to the invention may be determined and incorporated into a DNA template using standard methods. For example, starting from a desired amino acid sequence (e.g., an enzyme sequence), a virtual reverse translation is carried out based on the degenerated genetic code. Optimization algorithms may then be used for selection of suitable codons. Typically, the G/C content can be optimized to achieve the highest possible G/C content on one hand, taking into the best possible account the frequency of the tRNAs according to codon usage on the other hand.
- RNA sequence can be established and displayed, for example, with the aid of an appropriate display device and compared with the original (wild-type) sequence.
- a secondary structure can also be analyzed to calculate stabilizing and destabilizing properties or, respectively, regions of the RNA.
- Modified mRNA [0198]
- mRNA according to the present invention may be synthesized as unmodified or modified mRNA.
- Modified mRNA comprises nucleotide modifications in the RNA.
- a modified mRNA according to the invention can thus include nucleotide modification that are, for example, backbone modifications, sugar modifications or base modifications.
- compositions comprising such lipids, can be used in formulations to facilitate the delivery of encapsulated materials (e.g., one or more polynucleotides such as mRNA) to, and subsequent transfection of one or more target cells.
- encapsulated materials e.g., one or more polynucleotides such as mRNA
- cationic lipids described herein are characterized as resulting in one or more of receptor-mediated endocytosis, clathrin- mediated and caveolae-mediated endocytosis, phagocytosis and macropinocytosis, fusogenicity, endosomal or lysosomal disruption and/or releasable properties that afford such compounds advantages relative other similarly classified lipids.
- a nucleic acid e.g., mRNA encoding a protein (e.g., a full length, fragment or portion of a protein) as described herein may be delivered via a delivery vehicle comprising a compound of the invention as described herein.
- delivery vehicle comprising a compound of the invention as described herein.
- delivery vehicle comprising a compound of the invention as described herein.
- delivery vehicle comprising a compound of the invention as described herein.
- the terms “delivery vehicle,” “transfer vehicle,” “nanoparticle,” or grammatical equivalents thereof, are used interchangeably.
- the present invention provides a composition (e.g., a pharmaceutical composition) comprising a cationic lipids as described herein and one or more polynucleotides.
- a composition may further comprise (i) one or more additional cationic lipids, (ii) one or more non-cationic lipids, (iii) one or more cholesterol-based lipids and/or (iv) one or more PEG-modified lipids.
- a composition exhibits an enhanced (e.g., increased) ability to transfect one or more target cells. Accordingly, also provided herein are methods of transfecting one or more target cells.
- Such methods generally comprise the step of contacting the one or more target cells with the cationic lipids and/or pharmaceutical compositions disclosed herein (e.g., a liposomal formulation comprising a compound described herein encapsulating one or more polynucleotides) such that the one or more target cells are transfected with the materials encapsulated therein (e.g., one or more polynucleotides).
- the terms “transfect” or “transfection” refer to the intracellular introduction of one or more encapsulated materials (e.g., nucleic acids and/or polynucleotides) into a cell (e.g., into a target cell).
- the introduced polynucleotide may be stably or transiently maintained in the target cell.
- transfection efficiency refers to the relative amount of such encapsulated material (e.g., polynucleotides) up- taken by, introduced into, and/or expressed by the target cell which is subject to transfection. In practice, transfection efficiency may be estimated by the amount of a reporter polynucleotide product produced by the target cells following transfection.
- the compounds and pharmaceutical compositions described herein demonstrate high transfection efficiencies thereby improving the likelihood that appropriate dosages of the encapsulated materials (e.g., one or more polynucleotides) will be delivered to the site of pathology and subsequently expressed, while at the same time minimizing potential systemic adverse effects or toxicity associated with the compound or their encapsulated contents.
- the encapsulated materials e.g., one or more polynucleotides
- the production of the product e.g., a polypeptide or protein
- the production of the product may be stimulated and the capability of such target cells to express the polynucleotide and produce, for example, a polypeptide or protein of interest is enhanced.
- transfection of a target cell by one or more compounds or pharmaceutical compositions encapsulating mRNA will enhance (i.e., increase) the production of the protein or enzyme encoded by such mRNA.
- delivery vehicles described herein may be prepared to preferentially distribute to other target tissues, cells or organs, such as the heart, lungs, kidneys, spleen.
- the delivery vehicles described herein e.g., liposomal delivery vehicles
- the lipid nanoparticles of the present invention may be prepared to achieve enhanced delivery to the target cells and tissues.
- polynucleotides e.g., mRNA
- encapsulated in one or more of the compounds or pharmaceutical and liposomal compositions described herein can be delivered to and/or transfect targeted cells or tissues.
- the encapsulated polynucleotides are capable of being expressed and functional polypeptide products produced (and in some instances excreted) by the target cell, thereby conferring a beneficial property to, for example the target cells or tissues.
- Such encapsulated polynucleotides may encode, for example, an antigen hormone, enzyme, receptor, polypeptide, peptide or other protein of interest.
- Liposomal Delivery Vehicles [0206]
- a composition is a suitable delivery vehicle.
- a composition is a liposomal delivery vehicle, e.g., a lipid nanoparticle.
- liposomal delivery vehicle and “liposomal composition” are used interchangeably.
- Enriching liposomal compositions with one or more of the cationic lipids disclosed herein may be used as a means of improving the safety profile or otherwise conferring one or more desired properties to such enriched liposomal composition (e.g., improved delivery of the encapsulated polynucleotides to one or more target cells and/or reduced in vivo toxicity of a liposomal composition).
- the compounds of the invention as described herein may be used as a component of a liposomal composition to facilitate or enhance the delivery and release of encapsulated materials (e.g., one or more therapeutic agents) to one or more target cells (e.g., by permeating or fusing with the lipid membranes of such target cells).
- encapsulated materials e.g., one or more therapeutic agents
- target cells e.g., by permeating or fusing with the lipid membranes of such target cells.
- liposomal delivery vehicles e.g., lipid nanoparticles, are usually characterized as microscopic vesicles having an interior aqua space sequestered from an outer medium by a membrane of one or more bilayers.
- Bilayer membranes of liposomes are typically formed by amphiphilic molecules, such as lipids of synthetic or natural origin that comprise spatially separated hydrophilic and hydrophobic domains (Lasic, Trends Biotechnol., 16: 307-321, 1998). Bilayer membranes of the liposomes can also be formed by amphophilic polymers and surfactants (e.g., polymerosomes, niosomes, etc.). In the context of the present invention, a liposomal delivery vehicle typically serves to transport a desired mRNA to a target cell or tissue.
- compositions e.g., liposomal compositions
- encapsulate materials such as for example, one or more biologically-active polynucleotides (e.g., mRNA).
- a composition e.g., a pharmaceutical composition
- a liposome comprises: (i) one or more cationic lipids, (ii) one or more non-cationic lipids, (iii) one or more cholesterol-based lipids and (iv) one or more PEG-modified lipids, wherein at least one cationic lipid is a compound of the invention as described herein.
- a composition comprises an mRNA encoding for a peptide or protein (e.g., any peptide or protein described herein).
- a composition comprises an mRNA encoding for a peptide (e.g., any peptide described herein).
- a composition comprises an mRNA encoding for a protein (e.g., any protein described herein).
- a composition e.g., a pharmaceutical composition
- a nucleic acid is an mRNA encoding a peptide or protein.
- an mRNA encodes a peptide or protein for use in the delivery to or treatment of the human body. Exemplary mRNAs are described herein.
- a liposomal delivery vehicle e.g., a lipid nanoparticle
- a liposomal delivery vehicle can have a net positive charge.
- a liposomal delivery vehicle e.g., a lipid nanoparticle
- a liposomal delivery vehicle can have a net negative charge.
- a liposomal delivery vehicle e.g., a lipid nanoparticle
- a net neutral charge e.
- a lipid nanoparticle that encapsulates a nucleic acid e.g., mRNA encoding a peptide or protein
- the amount of a compound of the invention as described herein in a composition can be described as a percentage (“wt%”) of the combined dry weight of all lipids of a composition (e.g., the combined dry weight of all lipids present in a liposomal composition).
- wt% percentage of the combined dry weight of all lipids of a composition
- a compound of the invention as described herein is present in an amount that is about 0.5 wt% to about 30 wt% (e.g., about 0.5 wt% to about 20 wt%) of the combined dry weight of all lipids present in a composition (e.g., a liposomal composition).
- a compound of the invention as described herein is present in an amount that is about 1 wt% to about 30 wt%, about 1 wt% to about 20 wt%, about 1 wt% to about 15 wt%, about 1 wt% to about 10 wt%, or about 5 wt% to about 25 wt% of the combined dry weight of all lipids present in a composition (e.g., a liposomal composition).
- a compound of the invention as described herein is present in an amount that is about 0.5 wt% to about 5 wt%, about 1 wt% to about 10 wt%, about 5 wt% to about 20 wt%, or about 10 wt% to about 20 wt% of the combined dry weight of all lipids present in a composition such as a liposomal delivery vehicle.
- the amount of a compound of the invention as described herein is present in an amount that is at least about 5 wt%, about 10 wt%, about 15 wt%, about 20 wt%, about 25 wt%, about 30 wt%, about 35 wt%, about 40 wt%, about 45 wt%, about 50 wt%, about 55 wt%, about 60 wt%, about 65 wt%, about 70 wt%, about 75 wt%, about 80 wt%, about 85 wt%, about 90 wt%, about 95 wt%, about 96 wt%, about 97 wt%, about 98 wt%, or about 99 wt% of the combined dry weight of total lipids in a composition (e.g., a liposomal composition).
- a composition e.g., a liposomal composition
- a composition e.g., a liposomal delivery vehicle such as a lipid nanoparticle
- a delivery vehicle comprises about 0.5 wt%, about 1 wt%, about 3 wt%, about 5 wt%, or about 10 wt% of a compound described herein.
- a delivery vehicle (e.g., a liposomal delivery vehicle such as a lipid nanoparticle) comprises up to about 0.5 wt%, about 1 wt%, about 3 wt%, about 5 wt%, about 10 wt%, about 15 wt%, or about 20 wt% of a compound described herein. In embodiments, the percentage results in an improved beneficial effect (e.g., improved delivery to targeted tissues).
- the amount of a compound of the invention as described herein in a composition also can be described as a percentage (“mol%”) of the combined molar amounts of total lipids of a composition (e.g., the combined molar amounts of all lipids present in a liposomal delivery vehicle).
- mol% a percentage of the combined molar amounts of total lipids of a composition
- a compound of the invention as described herein is present in an amount that is about 0.5 mol% to about 50 mol% (e.g., about 0.5 mol% to about 20 mol%) of the combined molar amounts of all lipids present in a composition such as a liposomal delivery vehicle.
- a compound of the invention as described herein is present in an amount that is about 0.5 mol% to about 5 mol%, about 1 mol% to about 10 mol%, about 5 mol% to about 20 mol%, about 10 mol% to about 20 mol%, about 15 mol% to about 30 mol%, about 20 mol% to about 35 mol%, about 25 mol% to about 40 mol%, about 30 mol% to about 45 mol%, about 35 mol% to about 50 mol%, about 40 mol% to about 55 mol %, or about 45 mol% to about 60 mol% of the combined molar amounts of all lipids present in a composition such as a liposomal delivery vehicle.
- a compound of the invention as described herein is present in an amount that is about 1 mol% to about 60 mol%, 1 mol% to about 50 mol%, 1 mol% to about 40 mol%, 1 mol% to about 30 mol%, about 1 mol% to about 20 mol%, about 1 mol% to about 15 mol%, about 1 mol% to about 10 mol%, about 5 mol% to about 55 mol%, about 5 mol% to about 45 mol%, about 5 mol% to about 35 mol% or about 5 mol% to about 25 mol% of the combined molar amounts of all lipids present in a composition such as a liposomal delivery vehicle.
- a compound of the invention as described herein can comprise from about 0.1 mol% to about 50 mol%, or from 0.5 mol% to about 50 mol%, or from about 1 mol% to about 50 mol%, or from about 5 mol% to about 50 mol%, or from about 10 mol% to about 50 mol%, or from about 15 mol% to about 50 mol%, or from about 20 mol% to about 50 mol%, or from about 25 mol% to about 50 mol%, or from about 30 mol% to about 50 mol%, of the total amount of lipids in a composition (e.g., a liposomal delivery vehicle).
- a composition e.g., a liposomal delivery vehicle
- a compound of the invention as described herein can comprise greater than about 0.1 mol%, or greater than about 0.5 mol%, or greater than about 1 mol%, greater than about 5 mol%, greater than about 10 mol%, greater than about 20 mol%, greater than about 30 mol%, or greater than about 40 mol% of the total amount of lipids in the lipid nanoparticle.
- the amount of a compound of the invention as described herein is present in an amount that is at least about 5 mol%, about 10 mol%, about 15 mol%, about 20 mol%, about 25 mol%, about 30 mol%, about 35 mol%, about 40 mol%, about 45 mol%, about 50 mol%, about 55 mol%, about 60 mol%, about 65 mol%, about 70 mol%, about 75 mol%, about 80 mol%, about 85 mol%, about 90 mol%, about 95 mol%, about 96 mol%, about 97 mol%, about 98 mol%, or about 99 mol% of the combined molar amounts of total lipids in a composition (e.g., a liposomal composition).
- a composition e.g., a liposomal composition
- the amount of a compound of the invention as described herein is present in an amount that is no more than about 5 mol%, about 10 mol%, about 15 mol%, about 20 mol%, about 25 mol%, about 30 mol%, about 35 mol%, about 40 mol%, about 45 mol%, about 50 mol%, about 55 mol%, about 60 mol%, about 65 mol%, about 70 mol%, about 75 mol%, about 80 mol%, about 85 mol%, about 90 mol%, about 95 mol%, about 96 mol%, about 97 mol%, about 98 mol%, or about 99 mol% of the combined molar amounts of total lipids in a composition (e.g., a liposomal composition).
- a composition e.g., a liposomal composition
- such pharmaceutical (e.g., liposomal) compositions comprise: one or more PEG-modified lipids; one or more non-cationic lipids; and one or more cholesterol lipids.
- such pharmaceutical (e.g., liposomal) compositions comprise: one or more PEG-modified lipids and one or more cholesterol lipids.
- such a composition has four lipid components comprising a compound of the invention as described herein as the cationic lipid component, and further comprising: (i) a non-cationic lipid (e.g., DOPE), (ii) a cholesterol-based lipid (e.g., cholesterol) and (iii) a PEG-modified lipid (e.g., DMG-PEG2K).
- a non-cationic lipid e.g., DOPE
- a cholesterol-based lipid e.g., cholesterol
- PEG-modified lipid e.g., DMG-PEG2K
- a lipid nanoparticle that encapsulates a nucleic acid comprises one or more compounds of the invention as described herein, as well as one or more lipids selected from the group consisting of: (i) a cationic lipid, (ii) a non-cationic lipid, (iii) a PEGylated lipid, and (iv) a cholesterol-based lipid.
- the selection of cationic lipids, non-cationic lipids and/or PEG-modified lipids which comprise the lipid nanoparticle, as well as the relative molar ratio of such lipids to each other is based upon the characteristics of the selected lipid(s), the nature of the intended target cells, the characteristics of the mRNA to be delivered. Additional considerations include, for example, the saturation of the alkyl chain, as well as the size, charge, pH, pKa, fusogenicity and toxicity of the selected lipid(s). Thus, the molar ratios may be adjusted accordingly.
- a composition may comprise one or more additional cationic lipids.
- liposomes may comprise one or more additional cationic lipids.
- cationic lipid refers to any of a number of lipid species that have a net positive charge at a selected pH, such as physiological pH. Several cationic lipids have been described in the literature, many of which are commercially available. [0245] Suitable additional cationic lipids for use in the compositions include the cationic lipids as described in the literature.
- compositions may also comprise one or more helper lipids.
- helper lipids include non-cationic lipids.
- non-cationic lipid refers to any neutral, zwitterionic or anionic lipid.
- anionic lipid refers to any of a number of lipid species that carry a net negative charge at a selected pH, such as physiological pH.
- Non-cationic lipids include, but are not limited to, distearoylphosphatidylcholine (DSPC), dioleoylphosphatidylcholine (DOPC), dipalmitoylphosphatidylcholine (DPPC), dioleoylphosphatidylglycerol (DOPG), dipalmitoylphosphatidylglycerol (DPPG), dioleoylphosphatidylethanolamine (DOPE), 1,2-Dierucoyl-sn-glycero-3-phosphoethanolamine (DEPE), palmitoyloleoylphosphatidylcholine (POPC), palmitoyloleoyl-phosphatidylethanolamine (POPE), dioleoyl-phosphatidylethanolamine 4-(N-maleimidomethyl)-cyclohexane-1-carboxylate (DOPE-mal), dipalmitoyl phosphatidyl ethanolamine (DPPE), dimyristoyl
- a non- cationic or helper lipid suitable for practicing the invention is dioleoylphosphatidylethanolamine (DOPE).
- DOPE dioleoylphosphatidylethanolamine
- DEPE 1,2-Dierucoyl-sn-glycero-3-phosphoethanolamine
- a non-cationic lipid is a neutral lipid, i.e., a lipid that does not carry a net charge in the conditions under which the composition is formulated and/or administered.
- a non-cationic lipid may be present in a molar ratio (mol%) of about 5% to about 90%, about 5% to about 70%, about 5% to about 50%, about 5% to about 40%, about 5% to about 30%, about 10% to about 70%, about 10% to about 50%, or about 10% to about 40% of the total lipids present in a composition.
- total non-cationic lipids may be present in a molar ratio (mol%) of about 5% to about 90%, about 5% to about 70%, about 5% to about 50%, about 5% to about 40%, about 5% to about 30%, about 10 % to about 70%, about 10% to about 50%, or about 10% to about 40% of the total lipids present in a composition.
- the percentage of non-cationic lipid in a liposome may be greater than about 5 mol%, greater than about 10 mol%, greater than about 20 mol%, greater than about 30 mol%, or greater than about 40 mol%. In some embodiments, the percentage total non-cationic lipids in a liposome may be greater than about 5 mol%, greater than about 10 mol%, greater than about 20 mol%, greater than about 30 mol%, or greater than about 40 mol%.
- the percentage of non-cationic lipid in a liposome is no more than about 5 mol%, no more than about 10 mol%, no more than about 20 mol%, no more than about 30 mol%, or no more than about 40 mol%. In some embodiments, the percentage total non-cationic lipids in a liposome may be no more than about 5 mol%, no more than about 10 mol%, no more than about 20 mol%, no more than about 30 mol%, or no more than about 40 mol%.
- a non-cationic lipid may be present in a weight ratio (wt%) of about 5% to about 90%, about 5% to about 70%, about 5% to about 50%, about 5% to about 40%, about 5% to about 30%, about 10 % to about 70%, about 10% to about 50%, or about 10% to about 40% of the total lipids present in a composition.
- total non-cationic lipids may be present in a weight ratio (wt%) of about 5% to about 90%, about 5% to about 70%, about 5% to about 50%, about 5% to about 40%, about 5% to about 30%, about 10 % to about 70%, about 10% to about 50%, or about 10% to about 40% of the total lipids present in a composition.
- the percentage of non-cationic lipid in a liposome is no more than about 5 wt%, no more than about 10 wt%, no more than about 20 wt%, no more than about 30 wt%, or no more than about 40 wt%.
- the percentage total non-cationic lipids in a liposome may be no more than about 5 wt%, no more than about 10 wt%, no more than about 20 wt%, no more than about 30 wt%, or no more than about 40 wt%.
- a cholesterol-based lipid may be present in a molar ratio (mol%) of about 1% to about 30%, or about 5% to about 20% of the total lipids present in a liposome.
- the percentage of cholesterol-based lipid in the lipid nanoparticle may be greater than about 5 mol%, greater than about 10 mol%, greater than about 20 mol%, greater than about 30 mol%, or greater than about 40 mol%.
- the percentage of cholesterol-based lipid in the lipid nanoparticle may be no more than about 5 mol%, no more than about 10 mol%, no more than about 20 mol%, no more than about 30 mol%, or no more than about 40 mol%.
- a cholesterol-based lipid may be present in a weight ratio (wt%) of about 1% to about 30%, or about 5% to about 20% of the total lipids present in a liposome.
- the percentage of cholesterol-based lipid in the lipid nanoparticle may be greater than about 5 wt%, greater than about 10 wt%, greater than about 20 wt%, greater than about 30 wt%, or greater than about 40 wt%. In some embodiments, the percentage of cholesterol-based lipid in the lipid nanoparticle may be no more than about 5 wt%, no more than about 10 wt%, no more than about 20 wt%, no more than about 30 wt%, or no more than about 40 wt%.
- a composition e.g., a liposomal composition
- a suitable PEG-modified or PEGylated lipid for practicing the invention is 1,2-dimyristoyl-rac-glycero-3-methoxypolyethylene glycol-2000 (DMG-PEG2K).
- DMG-PEG2K 1,2-dimyristoyl-rac-glycero-3-methoxypolyethylene glycol-2000
- PEG-CER derivatized ceramides
- C8 PEG-2000 ceramide N-octanoyl-sphingosine-1- [succinyl(methoxy polyethylene glycol)-2000]
- particularly useful exchangeable lipids are PEG-ceramides having shorter acyl chains (e.g., (C 14 ) or (C 18 )).
- Contemplated further PEG-modified lipids include, but are not limited to, a polyethylene glycol chain of up to 5 kDa in length covalently attached to a lipid with alkyl chain(s) of (C6-C20) length.
- a PEG-modified or PEGylated lipid is PEGylated cholesterol or PEG-2K.
- Such components may prevent complex aggregation and may also provide a means for increasing circulation lifetime and increasing the delivery of the lipid-nucleic acid composition to the target cell, (Klibanov et al. (1990) FEBS Letters, 268 (1): 235-237), or they may be selected to rapidly exchange out of the formulation in vivo (see U.S. Pat. No.5,885,613).
- PEG-modified phospholipid and derivatized lipids of the present invention may be present in a molar ratio (mol%) from about 0% to about 10%, about 0.5% to about 10%, about 1% to about 10%, about 2% to about 10%, about 3% to about 5%, about 1% to about 5%, or about 1.5% to about 3% of the total lipid present in the composition (e.g., a liposomal composition).
- compositions e.g., to construct liposomal compositions
- encapsulated materials e.g., one or more therapeutic polynucleotides
- target cells e.g., by permeating or fusing with the lipid membranes of such target cells
- a liposomal composition e.g., a lipid nanoparticle
- the phase transition in the lipid bilayer of the one or more target cells may facilitate the delivery of the encapsulated materials (e.g., one or more therapeutic polynucleotides encapsulated in a lipid nanoparticle) into the one or more target cells.
- the encapsulated materials e.g., one or more therapeutic polynucleotides encapsulated in a lipid nanoparticle
- compounds of the invention as described herein may be used to prepare liposomal vehicles that are characterized by their reduced toxicity in vivo.
- the reduced toxicity is a function of the high transfection efficiencies associated with the compositions disclosed herein, such that a reduced quantity of such composition may be administered to the subject to achieve a desired therapeutic response or outcome.
- compounds of the invention as described herein may be used to prepare liposomal vehicles that are characterized by effective intranasal delivery of mRNA. In certain embodiments, compounds of the invention as described herein may be used to prepare liposomal vehicles that are characterized by effective pulmonary delivery of mRNA.
- compounds of the invention as described herein may be used to prepare liposomal vehicles that are characterized by achieving high levels of peptide or protein expression when delivering mRNA encoding for said peptide or protein by intravenous, intrathecal, intramuscular, intranasal, sublingual, or by pulmonary delivery, optionally through nebulization.
- compounds of the invention as described herein may be used to prepare liposomal vehicles that are characterized by achieving high levels of peptide or protein expression when delivering mRNA encoding for said peptide or protein by intramuscular delivery.
- pharmaceutical formulations comprising a compound described and nucleic acids provided by the present invention may be used for various therapeutic disease and/or disease prevention purposes.
- a compound described herein and nucleic acids can be formulated in combination with one or more additional pharmaceutical carriers, targeting ligands or stabilizing reagents.
- a compound described herein can be formulated via pre-mixed lipid solution.
- a composition comprising a compound described herein can be formulated using post-insertion techniques into the lipid membrane of the nanoparticles. Techniques for formulation and administration of drugs may be found in “Remington’s Pharmaceutical Sciences,” Mack Publishing Co., Easton, Pa., latest edition.
- Suitable routes of administration include, for example, oral, rectal, vaginal, transmucosal, pulmonary including intratracheal or inhaled, or intestinal administration; parenteral delivery, including intradermal, transdermal (topical), intramuscular, subcutaneous, intramedullary injections, as well as intrathecal, direct intraventricular, intravenous, intraperitoneal, or intranasal.
- the route of administration is selected from intravenous, intrathecal, intramuscular, intranasal, sublingual, or by pulmonary delivery, optionally through nebulization.
- the route of administration can be intramuscular.
- the intramuscular administration is to a muscle selected from the group consisting of skeletal muscle, smooth muscle and cardiac muscle.
- the administration results in delivery of the nucleic acids to a muscle cell.
- the administration results in delivery of the nucleic acids to a hepatocyte (i.e., liver cell).
- One route for administering a liposomal composition of the invention may be intravenous delivery, in particular when treating metabolic disorders, especially those affecting the liver (e.g., ornithine transcarbamylase (OTC) deficiency).
- OTC ornithine transcarbamylase
- the liposomal composition may be administered via pulmonary delivery (e.g., for the treatment of cystic fibrosis).
- a liposomal composition of the invention is typically administered intramuscularly.
- a liposomal composition of the invention may be administered intranasally for vaccination.
- Diseases or disorders affecting the eye may be treated by administering a liposomal composition of the invention intravitreally.
- pharmaceutical formulations of the invention may be administered in a local rather than systemic manner, for example, via injection of the pharmaceutical formulation directly into a targeted tissue (e.g., in a sustained release formulation). Local delivery can be affected in various ways, depending on the tissue to be targeted.
- Exemplary tissues in which mRNA may be delivered and/or expressed include, but are not limited to the liver, kidney, heart, spleen, serum, brain, skeletal muscle, lymph nodes, skin, and/or cerebrospinal fluid.
- aerosols containing compositions of the present invention can be inhaled (for nasal, tracheal, or bronchial delivery); compositions of the present invention can be injected into the site of injury, disease manifestation, or pain, for example; compositions can be provided in lozenges for oral, tracheal, or esophageal application; can be supplied in liquid, tablet or capsule form for administration to the stomach or intestines, can be supplied in suppository form for rectal or vaginal application; or can even be delivered to the eye by use of creams, drops, or even injection.
- pharmaceutical formulations of the invention may be administered intranasally.
- the pharmaceutical formulations of the invention may be administered via nasal spray.
- Exemplary tissues in which mRNA may be delivered and/or expressed include, but are not limited to the lungs, heart, liver, spleen and muscle.
- the tissue to be targeted is in the lungs.
- the tissue to be targeted is in muscle.
- pharmaceutical formulations of the invention may be administered by pulmonary delivery, optionally through nebulization or dry powder inhalation.
- the pharmaceutical formulations of the invention are administered by pulmonary delivery through nebulization.
- the pharmaceutical formulations of the invention are administered by pulmonary delivery through dry powder inhalation .
- tissue in which mRNA may be delivered and/or expressed include, but are not limited to the lungs, heart, liver, spleen and muscle.
- the tissue to be targeted is in the lungs.
- the tissue to be targeted is in muscle.
- Compositions described herein can comprise mRNA encoding peptides including those described herein (e.g., a polypeptide such as a protein).
- a mRNA encodes a polypeptide.
- a mRNA encodes a peptide.
- the peptide is an antigen.
- a mRNA encodes a peptide for treating or preventing Flu. In embodiments, a mRNA encodes a peptide for treating or preventing influenza A virus. In embodiments, a mRNA encodes a peptide for treating or preventing influenza B virus. In embodiments, a mRNA encodes a peptide for treating or preventing chlamydia. In embodiments, a mRNA encodes a peptide for treating or preventing a disease or disorder induced by infection with P. gingivalis. In embodiments, a mRNA encodes a peptide for treating or preventing acne. [0269] In embodiments, a mRNA encodes a protein.
- a mRNA encodes a protein for treating or preventing Flu. In embodiments, a mRNA encodes a protein for treating or preventing influenza A virus. In embodiments, a mRNA encodes a protein for treating or preventing influenza B virus. In embodiments, a mRNA encodes a protein for treating or preventing chlamydia. In embodiments, a mRNA encodes a protein for treating or preventing a disease or disorder induced by infection with P. gingivalis. In embodiments, a mRNA encodes a protein for treating or preventing acne. [0270] In embodiments, the mRNA encodes an influenza antigen. In embodiments, the mRNA encodes an antigen of influenza A virus.
- the mRNA encodes an antigen of influenza B virus. In embodiments, the mRNA encodes an antigen of Chlamydia sp. bacteria. In embodiments, the mRNA encodes an antigen of C.trachomatis. In embodiments, the mRNA encodes an antigen of P. gingivalis. In embodiments, the mRNA encodes an acne antigen. In embodiments, the mRNA encodes an antigen of C.acnes.
- compositions comprising a cationic lipid described herein are provided for use in treating or preventing Flu in a subject (e.g. human).
- compositions comprising a cationic lipid described herein are provided for use in treating or preventing a disease or disorder caused by influenza A virus in a subject (e.g. human).
- compositions comprising a cationic lipid described herein are provided for use in treating or preventing a disease or disorder caused by influenza B virus in a subject (e.g. human).
- compositions comprising a cationic lipid described herein are provided for use in treating or preventing a chlamydia in a subject (e.g. human).
- compositions comprising a cationic lipid described herein are provided for use in treating or preventing C.trachomatis infection in a subject (e.g. human).
- compositions comprising a cationic lipid described herein are provided for use in treating or preventing acne in a subject (e.g. human).
- compositions comprising a cationic lipid described herein are provided for use in treating or preventing C.acnes infection in a subject (e.g. human).
- compositions comprising a cationic lipid described herein are provided for use in treating or preventing a disease or disorder induced by infection with P. gingivalis.
- said composition further comprises mRNA encoding a polypeptide, peptide or antigen suitable for the treatment or prevention of said disease or disorder or infection, such as those described herein.
- a method of treating or preventing a disease or disorder caused by influenza A virus in a subject e.g. human
- the method comprising administering a composition of the invention comprising a cationic lipid described herein to the subject.
- a method of treating or preventing a disease or disorder caused by influenza B virus in a subject e.g. human
- the method comprising administering a composition of the invention comprising a cationic lipid described herein to the subject.
- a method of treating or preventing chlamydia in a subject e.g.
- a method of treating or preventing C.trachomatis infection in a subject e.g. human
- the method comprising administering a composition of the invention comprising a cationic lipid described herein to the subject e.g. a method of treating or preventing C.trachomatis infection in a subject (e.g. human)
- the method comprising administering a composition of the invention comprising a cationic lipid described herein to the subject a method of treating or preventing acne in a subject (e.g. human), the method comprising administering a composition of the invention comprising a cationic lipid described herein to the subject.
- a method of treating or preventing C.acnes infection in a subject e.g.
- composition of the invention comprising a cationic lipid described herein to the subject.
- a method of treating or preventing a disease or disorder induced by infection with P. gingivalis in a subject e.g. human
- the method comprising administering a composition of the invention comprising a cationic lipid described herein to the subject.
- said composition further comprises mRNA encoding a polypeptide, peptide or antigen suitable for the treatment or prevention of said disease or disorder or infection, such as those described herein.
- Delivery Methods [0274] The route of delivery used in the methods of the invention allows for non-invasive, self- administration of the compounds of the invention.
- the methods involve intranasal, intratracheal or pulmonary administration by aerosolization, nebulization, or instillation of a composition comprising mRNA encoding a therapeutic peptide or protein in a suitable transfection or lipid carrier vehicles as described above.
- the methods involve intranasal, intratracheal or pulmonary administration by intravenous, intrathecal, intramuscular, intranasal, sublingual, or by pulmonary delivery, optionally through nebulization of a composition comprising mRNA encoding a therapeutic peptide or protein in a suitable transfection or lipid carrier vehicles as described above.
- the administration is intramuscular.
- compositions of the invention are provided for use in skin injection, e.g., in the epidermis, the dermis or the hypodermis of the skin.
- the compositions are provided in a device suitable for skin injection, such as a needle (e.g., an epidermic, dermic or hypodermic needle), a needle free device, a microneedle device or a microprojection array device.
- microneedle or microprojection array devices suitable for the skin injection as described in US20230270842A1, US20220339416A1, US20210085598A1, US20200246450A1, US20220143376A1, US20180264244A1, US20180263641A1, US20110245776A1.
- the peptide or protein product encoded by the mRNA e.g., a functional protein or enzyme
- the amount of peptide or protein product necessary to achieve a therapeutic effect will vary depending on the condition being treated, the peptide or protein encoded, and the condition of the patient.
- the peptide or protein product may be detectable in the peripheral target tissues at a concentration (e.g., a therapeutic concentration) of at least 0.025-1.5 ⁇ g/ml (e.g., at least 0.050 ⁇ g/ml, at least 0.075 ⁇ g/ml, at least 0.1 ⁇ g/ml, at least 0.2 ⁇ g/ml, at least 0.3 ⁇ g/ml, at least 0.4 ⁇ g/ml, at least 0.5 ⁇ g/ml, at least 0.6 ⁇ g/ml, at least 0.7 ⁇ g/ml, at least 0.8 ⁇ g/ml, at least 0.9 ⁇ g/ml, at least 1.0 ⁇ g/ml, at least 1.1 ⁇ g/ml, at least 1.2 ⁇ g/ml, at least 1.3 ⁇ g/ml, at least 1.4 ⁇ g/ml, or at least 1.5 ⁇ g/ml), for at least about 1, 2, 3, 4, 5, 6, 7, 8,
- nucleic acids can be delivered to the lungs by intratracheal administration of a liquid suspension of the compound and inhalation of an aerosol mist produced by a liquid nebulizer or the use of a dry powder apparatus such as that described in U.S. patent 5,780,014, incorporated herein by reference.
- the compounds of the invention may be formulated such that they may be aerosolized or otherwise delivered as a particulate liquid or solid prior to or upon administration to the subject.
- Such compounds may be administered with the assistance of one or more suitable devices for administering such solid or liquid particulate compositions (such as, e.g., an aerosolized aqueous solution or suspension) to generate particles that are easily respirable or inhalable by the subject.
- suitable devices e.g., a metered dose inhaler, jet- nebulizer, ultrasonic nebulizer, dry-powder-inhalers, propellant-based inhaler or an insufflator
- a predetermined mass, volume or dose of the compositions e.g., about 0.5 mg/kg of mRNA per dose
- the compounds of the invention are administered to a subject using a metered dose inhaler containing a suspension or solution comprising the compound and a suitable propellant.
- the compounds of the invention may be formulated as a particulate powder (e.g., respirable dry particles) intended for inhalation.
- compositions of the invention formulated as respirable particles are appropriately sized such that they may be respirable by the subject or delivered using a suitable device (e.g., a mean D50 or D90 particle size less than about 500 ⁇ m, 400 ⁇ m, 300 ⁇ m, 250 ⁇ m, 200 ⁇ m, 150 ⁇ m, 100 ⁇ m, 75 ⁇ m, 50 ⁇ m, 25 ⁇ m, 20 ⁇ m, 15 ⁇ m, 12.5 ⁇ m, 10 ⁇ m, 5 ⁇ m, 2.5 ⁇ m or smaller).
- the compounds of the invention are formulated to include one or more pulmonary surfactants (e.g., lamellar bodies).
- the compounds of the invention are administered to a subject such that a concentration of at least 0.05 mg/kg, at least 0.1 mg/kg, at least 0.5 mg/kg, at least 1.0 mg/kg, at least 2.0 mg/kg, at least 3.0 mg/kg, at least 4.0 mg/kg, at least 5.0 mg/kg, at least 6.0 mg/kg, at least 7.0 mg/kg, at least 8.0 mg/kg, at least 9.0 mg/kg, at least 10 mg/kg, at least 15 mg/kg, at least 20 mg/kg, at least 25 mg/kg, at least 30 mg/kg, at least 35 mg/kg, at least 40 mg/kg, at least 45 mg/kg, at least 50 mg/kg, at least 55 mg/kg, at least 60 mg/kg, at least 65 mg/kg, at least 70 mg/kg, at least 75 mg/kg, at least 80 mg/kg, at least 85 mg/kg, at least 90 mg/kg, at least 95 mg/kg, or at least 100 mg/kg body weight is administered in
- the compounds of the invention are administered to a subject such that a total amount of at least 0.1 mg, at least 0.5 mg, at least 1.0 mg, at least 2.0 mg, at least 3.0 mg, at least 4.0 mg, at least 5.0 mg, at least 6.0 mg, at least 7.0 mg, at least 8.0 mg, at least 9.0 mg, at least 10 mg, at least 15 mg, at least 20 mg, at least 25 mg, at least 30 mg, at least 35 mg, at least 40 mg, at least 45 mg, at least 50 mg, at least 55 mg, at least 60 mg, at least 65 mg, at least 70 mg, at least 75 mg, at least 80 mg, at least 85 mg, at least 90 mg, at least 95 mg or at least 100 mg mRNA is administered in one or more doses.
- siRNA see WO2010/144740.
- the synthesis of this lipid involves a six-step process and requires handling of a Grignard reagent.
- the present invention provides cationic lipids that can be prepared from readily available starting reagents, such as "Good's" buffers (see Table A below). These starting reagents can be coupled to cationic headgroups and lipid tails using coupling reactions, such as sulfonylation, acetylation and alkylation (see for example, Tables B1 and B2 below). Table A: Examples of “Good” buffers
- Table B1 Examples of lipid chains that are suitable for the present invention at positions R 1 and R 2
- Table B2 Examples of lipid chains that are suitable for the present invention at positions R 3 and R 4
- Process A (“A”) relates to a conventional method of encapsulating mRNA by mixing mRNA with a mixture of lipids, without first pre-forming the lipids into lipid nanoparticles.
- an ethanolic solution of a mixture of lipids (cationic lipid, phosphatidylethanolamine, cholesterol, and polyethylene glycol-lipid) at a fixed lipid to mRNA ratio were combined with an aqueous buffered solution of target mRNA at an acidic pH under controlled conditions to yield a suspension of uniform LNPs.
- the resulting nanoparticle suspensions were diluted to final concentration, filtered, and stored frozen at ⁇ 80°C until use.
- Lipid nanoparticle formulations of Table C were prepared by Process A. All of the lipid nanoparticle formulations comprised hEPO mRNA and the different lipids (Cationic Lipid: DMG- PEG2000: Cholesterol: DOPE/DSPC) in the mol % ratios specified in Table C. Table C: Exemplary lipid nanoparticle characterizations * The N/P ratio is defined as the ratio of the number of nitrogen in cationic lipid to the number of phosphate in nucleic acid. [0284] The cationic lipids of the present invention and lipid OF-02 were evaluated with lipid nanoparticle formulation 1.
- MC3 and SM-102 were evaluated with lipid nanoparticle formulation 2, which is a typical formulation for those lipids
- Example 3 Delivery of hEPO mRNA by intramuscular administration Mouse Studies [0285] In summary, lipid screening studies were conducted with female BALB/cJ mice 6-8 weeks of age (Jax #000651). Mice were dosed with 0.1 ⁇ g in 30 ⁇ L of LNPs by a single intramuscular (IM) injection into the gastrocnemius leg muscle. Blood samples were taken 6 and 24 hours post injection and hEPO levels were measured in the blood serum of the mice using an ELISA assay according to the manufacture’s protocol (R&D systems, Cat# DEP-00).
- WO2022/099003 A1 also describes an in vivo assay for intramuscular administration (e.g. on page 46, paragraph [00206]). Measured serum hEPO expression data are presented in Table D below. This data demonstrates that the disclosed lipids can induce expression of delivered mRNAs. Table D: Results of hEPO mRNA delivery studies - intramuscular administration of hEPO mRNA lipid formulations comprising the disclosed cationic lipids
- Example 4 Influenza titer [0286] Groups of Balb/c mice (Mus musculus) as per the treatment group were immunized under isoflurane anesthesia with a dose of 0.4 ⁇ g per mouse in 0.05 mL of Modified Kenya H3 mRNA-lipid nanoparticles via the IM route in the quadriceps, on day 0 in one hind leg and day 21 in the contralateral leg. Mice were evaluated for a minimum of 3 days post-administration and any animal that displayed severe clinical signs after the veterinarian’s assessment was euthanized by administration of 5 mg/kg of meloxicam by subcutaneous injection.
- Enzyme was inactivated by a 30-minute incubation period at 56°C followed by addition of six parts PBS for a final dilution of 1/10.
- HAI assays were performed in V-bottom 96-well plates using four hemagglutinating units (HAU) of virus and 0.5% turkey RBC.
- the reference serum for each strain was included as a positive control on every assay plate.
- Each plate also included a back-titration to confirm the antigen dose (4 HAU/25 ⁇ l) as well as a negative control sample (PBS or naive control serum).
- the HAI titer was determined as the highest dilution of serum resulting in complete inhibition of hemagglutination.
- Results were only considered valid for plates with the appropriate back-titration result (verifying 4 HAU/25 ⁇ l added) and a reference serum titer within 2-fold of the expected titer.
- the majority of tested lipids induced HAI titers not significantly different from that of clinical lipid OF-02 (see Fig.1). This example demonstrates that the disclosed lipids can deliver antigenic sequences that induce significant immune responses.
- NUMBERED EMBODIMENTS 1.
- 14. The compound of any one of embodiments 1-7, 9, or 11-13, wherein the compound has a structure according to Formula (II’), optionally Formula (II): or a pharmaceutically acceptable salt thereof, optionally wherein a is 4. 15.
- each RA is independently selected from optionally substituted C3-C28 alkyl, optionally substituted C3-C28 alkenyl, and -W 1 -C(O)-O-X 1 , wherein each W 1 is independently selected from optionally substituted C3-C5 alkylene and optionally substituted C3-C5 alkenylene, and each X 1 is independently selected from optionally substituted C5-C30 alkyl and optionally substituted C5-C30 alkenyl. 19.
- the method of embodiment 85, wherein the disease or disorder is a disease induced by infection with P. gingivalis. 88.
- the method of embodiment 85, wherein the disease or disorder is acne.
- the composition for use according to embodiment 89, or the method of embodiment 89, wherein the composition is administered intramuscularly.
- each of R 1, R 2, R 3, and R 4 is independently selected from: (i) optionally substituted C 5 -C 30 alkyl, and optionally substituted C 5 -C 30 alkenyl; (ii) optionally substituted C1-5 alkyl which is substituted with an optionally substituted heterocyclyl moiety; (iii) , wherein each R A is independently selected from optionally substituted C 3 -C 28 alkyl, optionally substituted C 3 -C 28 alkenyl, and -W 1 -C(O)-O-X 1 , wherein each W 1 is independently selected from optionally substituted C 3 -C 5 alkylene and optionally substituted C3-C5 alkenylene, and each X 1 is independently selected from optionally substituted C5-C30 alkyl and optionally substituted C5-C30 alkenyl; (iv) , wherein each R B is independently selected from optionally substituted C
- the compound is not one of the following structures: , , . 101.
- each RB is independently selected from options (i), (ii), and (iii) above; (d) one or more RB is independently selected from optionally substituted C5-C30 alkenyl, optionally wherein each RB is independently selected from optionally substituted C5-C30 alkenyl; and/or (e) one or more R B is independently selected from optionally substituted C 5 -C 9 alkenyl, for example , optionally wherein each R B is independently selected from optionally substituted C5-C9 alkenyl, for example . 105.
- each RC independently selected from optionally substituted C5- C30 alkyl and optionally substituted C5-C30 alkenyl, optionally wherein: (a) one or more c is selected from 3 or 5, optionally wherein each c is selected from 3 or 5; (b) one or more c is 4, optionally wherein each c is 4; (c) one or more R C is independently selected from optionally substituted C 5 -C 30 alkyl, optionally wherein each R C is independently selected from optionally substituted C 5 -C 30 alkyl; (d) one or more R C is independently selected from optionally substituted C 6 -C 17 alkyl, for example optionally substituted branched C 6 -C 17 alkyl, optionally wherein each R C is independently selected from optionally substituted C6-C17 alkyl, for example optionally substituted
- a composition comprising the compound of any one of embodiments 99-109 or a pharmaceutically acceptable salt thereof, one or more non-cationic lipids, one or more cholesterol- based lipids and one or more PEG-modified lipids, optionally wherein: (a) the composition is a lipid nanoparticle, optionally a liposome; (b) the composition is a lipid nanoparticle, optionally a liposome and the one or more cationic lipid(s) constitute(s) about 30 mol %-60 mol % of the lipid nanoparticle; (c) the composition is a lipid nanoparticle, optionally a liposome and the one or more non-cationic lipid(s) constitute(s) 10 mol %-50 mol % of the lipid nanoparticle; (d) the composition is a lipid nanoparticle, optionally a liposome and the one or more PEG-modified lipid(s) constitute(s) 1 mol %-10 mol % of the lipid nano
- composition of embodiment 110 wherein the composition is a lipid nanoparticle, optionally a liposome, and wherein said lipid nanoparticle encapsulates: (a) a nucleic acid; (b) a mRNA encoding a peptide or protein; (c) a mRNA encoding an antigen; (d) a mRNA encoding an influenza antigen, for example an antigen of influenza A virus or an antigen of influenza B virus; (e) a mRNA encoding an antigen of Chlamydia sp. bacteria, for example an antigen of C.trachomatis; (f) a mRNA encoding an antigen of P.
- composition of embodiment 111 encapsulating a mRNA encoding a peptide, protein or antigen for use in therapy optionally wherein said composition is for use in: (a) a method of treating or preventing a disease or disorder amenable to treatment or prevention by the peptide, protein or antigen encoded by the mRNA; (b) a method of treating or preventing a disease or disorder amenable to treatment or prevention by the peptide, protein or antigen encoded by the mRNA, wherein the disease or disorder is caused by a viral infection; (c) a method of treating or preventing a disease or disorder amenable to treatment or prevention by the peptide, protein or antigen encoded by the mRNA, wherein the disease or disorder is influenza; (d) a method of treating or preventing a disease or disorder amenable to treatment or prevention by the peptide, protein
- gingivalis or (g) a method of treating or preventing a disease or disorder amenable to treatment or prevention by the peptide, protein or antigen encoded by the mRNA, wherein the disease or disorder is acne.
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Organic Chemistry (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Engineering & Computer Science (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Epidemiology (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Physics & Mathematics (AREA)
- Dispersion Chemistry (AREA)
- Molecular Biology (AREA)
- Biomedical Technology (AREA)
- Nanotechnology (AREA)
- Optics & Photonics (AREA)
- Biophysics (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
The present invention provides, in part, asymmetric "good" buffer-based cationic lipids of Formula (I'), and sub-formulas thereof: or a pharmaceutically acceptable salt thereof. The compounds provided herein can be useful for delivery and expression of mRNA and encoded protein, e.g., as a component of liposomal delivery vehicle, and accordingly can be useful for treating various diseases, disorders and conditions, such as those caused by viral or bacterial infection.
Description
GOOD" BUFFER-BASED CATIONIC LIPIDS FOR NUCLEIC ACID DELIVERY
RELATED APPLICATIONS
[001] This application claims priority to European application no. 23306558.0 filed on 20th September 2023, the entire disclosure of which is hereby incorporated by reference.
BACKGROUND
[002] Delivery of nucleic acids has been explored extensively as a potential therapeutic option for certain disease states. In particular, messenger RNA (mRNA) therapy has become an increasingly important option for the prevention and treatment of various diseases (e.g. in the use of vaccines). [003] Efficient delivery of liposome-encapsulated nucleic acids remains an active area of research. Liposome-encapsulated nucleic acids can be administered intramuscularly (IM).
[004] The cationic lipid component of a liposome plays an important role in facilitating effective encapsulation of the nucleic acid during the loading of liposomes. In addition, cationic lipids may play an important role in the efficient release of the nucleic acid cargo from the liposome into the cytoplasm of a target cell. Various cationic lipids suitable for in vivo use have been discovered. However, there remains a need to identify cationic lipids that are effective for e.g., intramuscular delivery of mRNA (e.g., in vaccines). There also remains a need to identify cationic lipids that can be synthesized efficiently and cheaply without the formation of potentially toxic by-products.
[005] "Good" buffers (or Good's buffers) are buffering agents for biochemical and biological research that were first selected and described by Norman Good and his colleagues (Good, N.E., et al. (1966) Hydrogen Ion Buffers for Biological Research. Biochemistry 5(2), 467-477). Most biological reactions take place near-neutral pH between 6 and 8. Good therefore reasoned that an ideal buffer for biochemical or biological applications would have a pKa value in this region to provide maximum buffering capacity. Additional selection criteria included high solubility, lack of toxicity, limited interference with biochemical reactions, very low absorbance between 240 nm and 700 nm, enzymatic and hydrolytic stability, minimal changes due to temperature and concentration, limited effects due to ionic or salt composition of the solution, limited interaction with mineral cations, and limited permeability of biological membranes.
[006] The foregoing characteristics make "Good" buffers exceptionally good starting points for the synthesis of cationic lipids for use in in vivo settings. Many "Good" buffers remain crucial tools in modern laboratories and are therefore readily available at low cost.
SUMMARY OF THE INVENTION
[007] The present invention provides, among other things, a novel class of cationic lipid compounds for in vivo delivery of therapeutic agents, such as nucleic acids. The "Good" HEPES, HEPPS, and HEPBS buffers form the cores of the cationic lipids of the invention and were used to synthesize unique ionizable lipids containing different degradable moieties and carbon tails. The core structure with a hydroxyl and sulfonic acid group on either side allows for the ionizable lipids to contain both ester and disulfide degradable moieties. The inventors of the present invention have surprisingly found that certain lipid nanoparticles comprising a cationic lipid derived from HEPES, HEPPS, and HEPBS buffers, for example which possesses at least one pair of asymmetric lipid tails on either arm of the lipid molecule, exhibit improved properties relative to lipid nanoparticles comprising other cationic lipids derived from "Good" buffers, such as those in WO 2022/221688 Al, incorporated herein by reference and are very effective for the intramuscular delivery of mRNA encapsulated in said lipid nanoparticles. Indeed, lipid nanoparticles comprising the cationic lipids of the present invention have demonstrated high levels of peptide or protein expression when delivering mRNA encoding for said peptide or protein by intramuscular delivery. For example, lipid nanoparticles comprising cationic lipids of the present invention and encapsulating human erythropoietin (hEPO) mRNA achieved improved expression of hEPO mRNA when administered to mice by intramuscular delivery to lipid nanoparticles comprising MC3, which is currently used for in vivo delivery of e.g. siRNA (see W02010/144740).
[008] The cationic lipids of the present invention are also more straightforward to synthesize than other cationic lipids, such as MC3. Indeed, the synthesis of MC3 involves a six-step process and requires handling of a Grignard reagent. In contrast, the cationic lipids of the present invention can be synthesized from readily available starting reagents, such as "Good's" buffers (see Table A).
[009] The cationic lipids of the present invention also comprise cleavable groups (e.g., esters, thioesters, disulphides, carbonates, carbamates and thiocarbamates) that are contemplated to improve biodegradability and thus contribute to their favorable safety profile.
[010] It is contemplated that these compounds are capable of highly effective in vivo intramuscular delivery of various therapeutic agents and vaccines (e.g., therapeutic agents and/or vaccines for use in Flu, acne, chlamydia, or a disease or disorder induced by infection with P. gingivalis). It is also contemplated that lipid nanoparticles comprising these cationic lipid compounds are capable of highly effective in vivo delivery while maintaining a favorable safety profile. It is also contemplated that lipid nanoparticles comprising these cationic lipid compounds may exhibit improved degradation in vivo.
[Oil] In an aspect, provided herein are cationic lipids having a structure according to Formula (I'), optionally Formula (I):
or a pharmaceutically acceptable salt thereof, wherein: a is 3 or 4; each of Ri, R2, R3, and R4 is independently selected from:
(i) optionally substituted C5-C30 alkyl, and optionally substituted C5-C30 alkenyl;
(ii) optionally substituted C1-5 alkyl which is substituted with an optionally substituted heterocyclyl moiety;
OH
(iii)
, wherein each RA is independently selected from optionally substituted C3-C28 alkyl, optionally substituted C3-C28 alkenyl, and -W1-C(O)-O-X1, wherein each W1 is independently selected from optionally substituted C3-C5 alkylene and optionally substituted C3-C5 alkenylene, and each X1 is independently selected from optionally substituted C5-C30 alkyl and optionally substituted C5-C30 alkenyl; o
(iv)
, wherein each RB is independently selected from optionally substituted C5-C30 alkyl, optionally substituted C5-C30 alkenyl; and
(v)
, wherein each RC independently selected from optionally substituted C5-C30 alkyl, optionally substituted C5-C30 alkenyl; each b is independently selected from 5, 6, and 7; each c is independently selected from 3, 4, and 5; d is 2, 3, or 4; and wherein R1 and R2 are not identical to one another and/or R3 and R4 are not identical to one another; and wherein at least one of R1, R2, R3, and R4, is independently selected from option
r option
above. [012] In an aspect, provided herein are cationic lipids having a structure according to Formula (VI’),
or a pharmaceutically acceptable salt thereof, wherein: a is 3 or 4; d is 2, 3, or 4;
each c is independently selected from 3 and 4; and each Rc is independently selected from
[013] In another aspect provided herein is a compound selected from those listed in Tables 1 or 2, or a pharmaceutically acceptable salt thereof.
[014] In another aspect provided herein is a composition comprising one or more cationic lipids of the present invention or pharmaceutically acceptable salts thereof, one or more non-cationic lipids, one or more cholesterol-based lipids and one or more PEG-modified lipids.
[015] Also provided herein are compositions of the invention for use in methods of treating or preventing a disease or disorder amenable to treatment or prevention by a peptide or protein encoded by mRNA that is encapsulated within said composition. Methods of treating or preventing a disease or disorder wherein said method comprises administering to a subject in need thereof a composition of the invention and wherein the disease is amenable to treatment or prevention by a peptide or protein encoded by mRNA that is encapsulated within said composition are also provided.
BRIEF DESCRIPTION OF DRAWINGS
[016] Figure 1 shows HAI Titers after 35 days of post intramuscular injection (0.4 pg/50 pL) in mice of mRNA encoded influenza antigen encapsulated within lipid nanoparticles comprising cationic lipids of the present invention.
DETAILED DESCRIPTION OF CERTAIN EMBODIMENTS
Definitions
[017] In order for the present invention to be more readily understood, certain terms are first defined below. Additional definitions for the following terms and other terms are set forth throughout the specification. The publications and other reference materials cited herein to describe the background of the invention and to provide additional detail regarding its practice are hereby incorporated by reference.
[018] Amino acid: As used herein, the term "amino acid," in its broadest sense, refers to any compound and/or substance that can be incorporated into a polypeptide chain. In some embodiments, an amino acid has the general structure H2N-C(H)(R)-COOH. In some embodiments, an amino acid is a naturally occurring amino acid. In some embodiments, an amino acid is a synthetic
amino acid; in some embodiments, an amino acid is a d-amino acid; in some embodiments, an amino acid is an l-amino acid. "Standard amino acid" refers to any of the twenty standard l-amino acids commonly found in naturally occurring peptides. "Nonstandard amino acid" refers to any amino acid, other than the standard amino acids, regardless of whether it is prepared synthetically or obtained from a natural source. As used herein, "synthetic amino acid" encompasses chemically modified amino acids, including but not limited to salts, amino acid derivatives (such as amides), and/or substitutions. Amino acids, including carboxy- and/or amino-terminal amino acids in peptides, can be modified by methylation, amidation, acetylation, protecting groups, and/or substitution with other chemical groups that can change the peptide's circulating half-life without adversely affecting their activity. Amino acids may participate in a disulfide bond. Amino acids may comprise one or post-translational modifications, such as association with one or more chemical entities (e.g., methyl groups, acetate groups, acetyl groups, phosphate groups, formyl moieties, isoprenoid groups, sulfate groups, polyethylene glycol moieties, lipid moieties, carbohydrate moieties, biotin moieties, etc.). The term "amino acid" is used interchangeably with "amino acid residue," and may refer to a free amino acid and/or to an amino acid residue of a peptide. It will be apparent from the context in which the term is used whether it refers to a free amino acid or a residue of a peptide.
[019] Animal: As used herein, the term "animal" refers to any member of the animal kingdom. In some embodiments, "animal" refers to humans, at any stage of development. In some embodiments, "animal" refers to non-human animals, at any stage of development. In certain embodiments, the non-human animal is a mammal (e.g., a rodent, a mouse, a rat, a rabbit, a monkey, a dog, a cat, a sheep, a bovine, a primate, and/or a pig). In some embodiments, animals include, but are not limited to, mammals, birds, reptiles, amphibians, fish, insects, and/or worms. In some embodiments, an animal may be a transgenic animal, genetically engineered animal, and/or a clone.
[020] Approximately or about: As used herein, the term "approximately" or "about," as applied to one or more values of interest, refers to a value that is similar to a stated reference value. In certain embodiments, the term "approximately" or "about" refers to a range of values that fall within 25%, 20%, 19%, 18%, 17%, 16%, 15%, 14%, 13%, 12%, 11%, 10%, 9%, 8%, 7%, 6%, 5%, 4%, 3%, 2%, 1%, or less in either direction (greater than or less than) of the stated reference value unless otherwise stated or otherwise evident from the context (except where such number would exceed 100% of a possible value).
[021] Biologically active: As used herein, the term "biologically active" refers to a characteristic of any agent that has activity in a biological system, and particularly in an organism. For instance, an
agent that, when administered to an organism, has a biological effect on that organism, is considered to be biologically active.
[022] Delivery: As used herein, the term "delivery" encompasses both local and systemic delivery. For example, delivery of mRNA encompasses situations in which an mRNA is delivered to a target tissue and the encoded protein is expressed and retained within the target tissue (also referred to as "local distribution" or "local delivery"), and situations in which an mRNA is delivered to a target tissue and the encoded protein is expressed and secreted into patient's circulation system (e.g., serum) and systematically distributed and taken up by other tissues (also referred to as "systemic distribution" or "systemic delivery").
[023] Expression: As used herein, "expression" of a nucleic acid sequence refers to translation of an mRNA into a polypeptide, assemble multiple polypeptides into an intact protein (e.g., enzyme) and/or post-translational modification of a polypeptide or fully assembled protein (e.g., enzyme). In this application, the terms "expression" and "production," and grammatical equivalents thereof, are used interchangeably.
[024] Functional: As used herein, a "functional" biological molecule is a biological molecule in a form in which it exhibits a property and/or activity by which it is characterized.
[025] Half-life: As used herein, the term "half-life" is the time required for a quantity such as nucleic acid or protein concentration or activity to fall to half of its value as measured at the beginning of a time period.
[026] Helper lipid: The term "helper lipid" as used herein refers to any neutral or zwitterionic lipid material including cholesterol. Without wishing to be held to a particular theory, helper lipids may add stability, rigidity, and/or fluidity within lipid bilayers/nanoparticles.
[027] Improve, increase, or reduce: As used herein, the terms "improve," "increase," or "reduce," or grammatical equivalents, indicate values that are relative to a baseline measurement, such as a measurement in the same individual prior to initiation of the treatment described herein, or a measurement in a control subject (or multiple control subject) in the absence of the treatment described herein. A "control subject" is a subject afflicted with the same form of disease as the subject being treated, who is about the same age as the subject being treated.
[028] In Vitro: As used herein, the term "in vitro” refers to events that occur in an artificial environment, e.g., in a test tube or reaction vessel, in cell culture, etc., rather than within a multicellular organism.
[029] In Vivo: As used herein, the term "in vivo” refers to events that occur within a multi-cellular organism, such as a human and a non-human animal. In the context of cell-based systems, the term
may be used to refer to events that occur within a living cell (as opposed to, for example, in vitro systems).
[030] Liposome: As used herein, the term "liposome" refers to any lamellar, multilamellar, or solid nanoparticle vesicle. Typically, a liposome as used herein can be formed by mixing one or more lipids or by mixing one or more lipids and polymer(s). In some embodiments, a liposome suitable for the present invention contains a cationic lipid(s) and optionally further comprises:
(i) non-cationic lipid(s),
(ii) cholesterol-based lipid(s), and/or
(iii) PEG-modified lipid(s).
[031] messenger RNA (mRNA): As used herein, the term "messenger RNA (mRNA)" or "mRNA" refers to a polynucleotide that encodes at least one polypeptide. mRNA as used herein encompasses both modified and unmodified RNA. The term "modified mRNA" related to mRNA comprising at least one chemically modified nucleotide. mRNA may contain one or more coding and non-coding regions. mRNA can be purified from natural sources, produced using recombinant expression systems and optionally purified, chemically synthesized, etc. Where appropriate, e.g., in the case of chemically synthesized molecules, mRNA can comprise nucleoside analogs such as analogs having chemically modified bases or sugars, backbone modifications, etc. An mRNA sequence is presented in the 5' to 3' direction unless otherwise indicated. In some embodiments, an mRNA is or comprises natural nucleosides (e.g., adenosine, guanosine, cytidine, uridine); nucleoside analogs (e.g., 2-aminoadenosine, 2-thiothymidine, inosine, pyrrolo-pyrimidine, 3-methyl adenosine, 5-methylcytidine, C5-propynyl-cytidine, C5-propynyl-uridine, 2-aminoadenosine, C5-bromouridine, C5-fluorouridine, C5-iodouridine, C5-propynyl-uridine, C5-propynyl-cytidine, C5-methylcytidine, 2- aminoadenosine, 7-deazaadenosine, 7-deazaguanosine, 8-oxoadenosine, 8-oxoguanosine, 0(6)- methylguanine, and 2-thiocytidine); chemically modified bases; biologically modified bases (e.g., methylated bases); intercalated bases; modified sugars (e.g., 2'-fluororibose, ribose, 2' -deoxyribose, arabinose, and hexose); and/or modified phosphate groups (e.g., phosphorothioates and S'-N- phosphoramidite linkages).
[032] Nucleic acid: As used herein, the term "nucleic acid," in its broadest sense, refers to any compound and/or substance that is or can be incorporated into a polynucleotide chain. In some embodiments, a nucleic acid is a compound and/or substance that is or can be incorporated into a polynucleotide chain via a phosphodiester linkage. In some embodiments, "nucleic acid" refers to individual nucleic acid residues (e.g., nucleotides and/or nucleosides). In some embodiments, "nucleic acid" refers to a polynucleotide chain comprising individual nucleic acid residues. In some embodiments, "nucleic acid" encompasses RNA as well as single and/or double-stranded DNA
and/or cDNA. In some embodiments, "nucleic acid" encompasses ribonucleic acids (RNA), including but not limited to any one or more of interference RNAs (RNAi), small interfering RNA (siRNA), short hairpin RNA (shRNA), antisense RNA (aRNA), messenger RNA (mRNA), modified messenger RNA (mmRNA), long non-coding RNA (IncRNA), micro-RNA (miRNA) multimeric coding nucleic acid (MCNA), polymeric coding nucleic acid (PCNA), guide RNA (gRNA) and CRISPR RNA (crRNA). In some embodiments, "nucleic acid" encompasses deoxyribonucleic acid (DNA), including but not limited to any one or more of single-stranded DNA (ssDNA), double-stranded DNA (dsDNA) and complementary DNA (cDNA). In some embodiments, "nucleic acid" encompasses both RNA and DNA. In embodiments, DNA may be in the form of antisense DNA, plasmid DNA, parts of a plasmid DNA, pre-condensed DNA, a product of a polymerase chain reaction (PCR), vectors (e.g., Pl, PAC, BAC, YAC, artificial chromosomes), expression cassettes, chimeric sequences, chromosomal DNA, or derivatives of these groups. In embodiments, RNA may be in the form of messenger RNA (mRNA), ribosomal RNA (rRNA), signal recognition particle RNA (7 SL RNA or SRP RNA), transfer RNA (tRNA), transfer-messenger RNA (tmRNA), small nuclear RNA (snRNA), small nucleolar RNA (snoRNA), SmY RNA, small Cajal body-specific RNA (scaRNA), guide RNA (gRNA), ribonuclease P (RNase P), Y RNA, telomerase RNA component (TERC), spliced leader RNA (SL RNA), antisense RNA (aRNA or asRNA), cis-natural antisense transcript (cis-NAT), CRISPR RNA (crRNA), long noncoding RNA (IncRNA), micro- RNA (miRNA), piwi-interacting RNA (piRNA), small interfering RNA (siRNA), transacting siRNA (tasiRNA), repeat associated siRNA (rasiRNA), 73K RNA, retrotransposons, a viral genome, a viroid, satellite RNA, or derivatives of these groups. In some embodiments, a nucleic acid is a mRNA encoding a protein such as an enzyme.
[033] Patient: As used herein, the term "patient" or "subject" refers to any organism to which a provided composition may be administered, e.g., for experimental, diagnostic, prophylactic, cosmetic, and/or therapeutic purposes. Typical patients include animals (e.g., mammals such as mice, rats, rabbits, non-human primates, and/or humans). In some embodiments, a patient is a human. A human includes pre- and post-natal forms.
[034] Pharmaceutically acceptable: The term "pharmaceutically acceptable," as used herein, refers to substances that, within the scope of sound medical judgment, are suitable for use in contact with the tissues of human beings and animals without excessive toxicity, irritation, allergic response, or other problem or complication, commensurate with a reasonable benefit/risk ratio.
[035] Pharmaceutically acceptable salt: Pharmaceutically acceptable salts are well known in the art. For example, S. M. Berge et al., describes pharmaceutically acceptable salts in detail in J. Pharmaceutical Sciences (1977) 66:1-19. Pharmaceutically acceptable salts of the compounds of this invention include those derived from suitable inorganic and organic acids and bases. Examples of
pharmaceutically acceptable, non-toxic acid addition salts are salts of an amino group formed with inorganic acids such as hydrochloric acid, hydrobromic acid, phosphoric acid, sulfuric acid, and perchloric acid or with organic acids such as acetic acid, oxalic acid, maleic acid, tartaric acid, citric acid, succinic acid, or malonic acid, or by using other methods used in the art such as ion exchange. Other pharmaceutically acceptable salts include adipate, alginate, ascorbate, aspartate, benzenesulfonate, benzoate, bisulfate, borate, butyrate, camphorate, camphorsulfonate, citrate, cyclopentanepropionate, digluconate, dodecylsulfate, ethanesulfonate, formate, fumarate, glucoheptonate, glycerophosphate, gluconate, hemisulfate, heptanoate, hexanoate, hydroiodide, 2- hydroxy-ethanesulfonate, lactobionate, lactate, laurate, lauryl sulfate, malate, maleate, malonate, methanesulfonate, 2-naphthalenesulfonate, nicotinate, nitrate, oleate, oxalate, palmitate, pamoate, pectinate, persulfate, 3-phenylpropionate, phosphate, picrate, pivalate, propionate, stearate, succinate, sulfate, tartrate, thiocyanate, p-toluenesulfonate, undecanoate, valerate salts, and the like. Salts derived from appropriate bases include alkali metal, alkaline earth metal, ammonium and N+(CI-4 alkyl)4 salts. Representative alkali or alkaline earth metal salts include sodium, lithium, potassium, calcium, magnesium, and the like. Further pharmaceutically acceptable salts include, when appropriate, nontoxic ammonium. Quaternary ammonium, and amine cations formed using counterions such as halide, hydroxide, carboxylate, sulfate, phosphate, nitrate, sulfonate, and aryl sulfonate. Further pharmaceutically acceptable salts include salts formed from the quarternization of an amine using an appropriate electrophile, e.g., an alkyl halide, to form a quarternized alkylated amino salt.
[036] Systemic distribution or delivery: As used herein, the terms "systemic distribution" or "systemic delivery," or grammatical equivalents thereof, refer to a delivery or distribution mechanism or approach that affect the entire body or an entire organism. Typically, systemic distribution or delivery is accomplished via body's circulation system, e.g., blood stream. Compared to the definition of "local distribution or delivery."
[037] Subject: As used herein, the term "subject" refers to a human or any non-human animal (e.g., mouse, rat, rabbit, dog, cat, cattle, swine, sheep, horse or primate). A human includes pre- and post-natal forms. In many embodiments, a subject is a human being. A subject can be a patient, which refers to a human presenting to a medical provider for diagnosis or treatment of a disease. The term "subject" is used herein interchangeably with "individual" or "patient." A subject can be afflicted with or is susceptible to a disease or disorder but may or may not display symptoms of the disease or disorder.
[038] Substantially: As used herein, the term "substantially" refers to the qualitative condition of exhibiting total or near-total extent or degree of a characteristic or property of interest. One of
ordinary skill in the biological arts will understand that biological and chemical phenomena rarely, if ever, go to completion and/or proceed to completeness or achieve or avoid an absolute result. The term "substantially" is therefore used herein to capture the potential lack of completeness inherent in many biological and chemical phenomena.
[039] Target tissues: As used herein, the term "target tissues" refers to any tissue that is affected by a disease to be treated. In some embodiments, target tissues include those tissues that display disease-associated pathology, symptom, or feature.
[040] Therapeutically effective amount: As used herein, the term "therapeutically effective amount" of a therapeutic agent means an amount that is sufficient, when administered to a subject suffering from or susceptible to a disease, disorder, and/or condition, to treat, diagnose, prevent, and/or delay the onset of the symptom(s) of the disease, disorder, and/or condition. It will be appreciated by those of ordinary skill in the art that a therapeutically effective amount is typically administered via a dosing regimen comprising at least one unit dose.
[041] Treating: As used herein, the term "treat," "treatment," or "treating" refers to any method used to partially or completely alleviate, ameliorate, relieve, inhibit, prevent, delay onset of, reduce severity of and/or reduce incidence of one or more symptoms or features of a particular disease, disorder, and/or condition. Treatment may be administered to a subject who does not exhibit signs of a disease and/or exhibits only early signs of the disease for the purpose of decreasing the risk of developing pathology associated with the disease.
Chemical definitions
[042] Acyl: As used herein, the term "acyl" refers to RZ-(C=O)-, wherein Rz is, for example, any alkyl, alkenyl, alkynyl, heteroalkyl or heteroalkylene.
[043] Aliphatic: As used herein, the term aliphatic refers to (C1-C50) hydrocarbons and includes both saturated and unsaturated hydrocarbons. An aliphatic may be linear, branched, or cyclic. For example, (C1-C20) aliphatics can include (C1-C20) alkyls (e.g., linear or branched (C1-C20) saturated alkyls), (C2-C20) alkenyls (e.g., linear or branched (C4-C20) dienyls, linear or branched (C6-C20) trienyls, and the like), and (C2-C2o)alkynyls (e.g., linear or branched (C2-C20) alkynyls). (C1-C20) aliphatics can include (C3-C20) cyclic aliphatics (e.g., (C3-C2o)cycloalkyls, (C4-C20) cycloalkenyls, or (C8-C20) cycloalkynyls). In certain embodiments, the aliphatic may comprise one or more cyclic aliphatic and/or one or more heteroatoms such as oxygen, nitrogen, or sulfur and may optionally be substituted with one or more substituents such as alkyl, halo, alkoxyl, hydroxy, amino, aryl, ether, ester or amide. An aliphatic group is unsubstituted or substituted with one or more substituent groups as described herein. For example, an aliphatic may be substituted with one or more (e.g., 1,
2, 3, 4, 5, or 6 independently selected substituents) of halogen, -COR", -CO2H, -CO2R", -CN, -OH, - OR", -OCOR", -OCO2R", -NH2, -NHR", -N(R")2, -SR" or -SO2R", wherein each instance of R" independently is (C1-C20) aliphatic (e.g., (C1-C20) alkyl, (C1-C15) alkyl, (C1-C10) alkyl, or (C1-C3) alkyl). In embodiments, R" independently is an unsubstituted alkyl (e.g., unsubstituted (C1-C20) alkyl, (C1-C15) alkyl, (C1-C10) alkyl, or (C1-C3) alkyl). In embodiments, R" independently is unsubstituted (C1-C3) alkyl. In embodiments, the aliphatic is unsubstituted. In embodiments, the aliphatic does not include any heteroatoms.
[044] Alkyl: As used herein, the term "alkyl" means acyclic linear and branched hydrocarbon groups, e.g. "(C1-C30) alkyl" refers to alkyl groups having 1-30 carbons. An alkyl group may be linear or branched. Examples of alkyl groups include, but are not limited to, methyl, ethyl, n-propyl, isopropyl, butyl, isobutyl, sec-butyl, tert-butyl, pentyl, isopentyl tert-pentylhexyl, isohexyl, etc. The term "lower alkyl" means an alkyl group straight chain or branched alkyl having 1 to 6 carbon atoms. Other alkyl groups will be readily apparent to those of skill in the art given the benefit of the present disclosure. An alkyl group may be unsubstituted or substituted with one or more substituent groups as described herein. For example, an alkyl group may be substituted with one or more (e.g., 1, 2, 3, 4, 5, or 6 independently selected substituents) of halogen, -COR", -CO2H, -CO2R", -CN, -OH, -OR", - OCOR", -OCO2R", -NH2, -NHR", -N(R")2, -SR" or -SO2R", wherein each instance of R" independently is (C1-C20) aliphatic (e.g., (C1-C20) alkyl, (C1-C15) alkyl, (C1-C10) alkyl, or (C1-C3) alkyl). In embodiments, R" independently is an unsubstituted alkyl (e.g., unsubstituted (C1-C20) alkyl, (C1-C15) alkyl, (C1-C10) alkyl, or (C1-C3) alkyl). In embodiments, R" independently is unsubstituted (C1-C3) alkyl. In embodiments, the alkyl is substituted (e.g., with 1, 2, 3, 4, 5, or 6 substituent groups as described herein). In embodiments, an alkyl group is substituted with a -OH group and may also be referred to herein as a "hydroxyalkyl" group, where the prefix denotes the -OH group and "alkyl" is as described herein.
[045] As used herein, "alkyl" also refers to a radical of a straight-chain or branched saturated hydrocarbon group having from 1 to 50 carbon atoms ("(C1-C50) alkyl"). In some embodiments, an alkyl group has 1 to 40 carbon atoms ("(C1-C40) alkyl"). In some embodiments, an alkyl group has 1 to 30 carbon atoms ("(C1-C30) alkyl"). In some embodiments, an alkyl group has 1 to 20 carbon atoms ("(C1-C20) alkyl"). In some embodiments, an alkyl group has 1 to 10 carbon atoms ("(C1-C10) alkyl"). In some embodiments, an alkyl group has 1 to 9 carbon atoms ("(C1-C9) alkyl"). In some embodiments, an alkyl group has 1 to 8 carbon atoms ("(Ci-Cg) alkyl"). In some embodiments, an alkyl group has 1 to 7 carbon atoms ("(C1-C7) alkyl"). In some embodiments, an alkyl group has 1 to 6 carbon atoms ("(Ci-Cg) alkyl"). In some embodiments, an alkyl group has 1 to 5 carbon atoms ("(C1-C5) alkyl"). In some embodiments, an alkyl group has 1 to 4 carbon atoms ("(C1-C4) alkyl"). In some embodiments,
an alkyl group has 1 to 3 carbon atoms ("(C1-C3) alkyl"). In some embodiments, an alkyl group has 1 to 2 carbon atoms ("(C1-C2) alkyl"). In some embodiments, an alkyl group has 1 carbon atom ("Ci alkyl"). In some embodiments, an alkyl group has 2 to 6 carbon atoms ("(Cz-Cg) alkyl"). Examples of (Ci-Cg) alkyl groups include, without limitation, methyl (Ci), ethyl (C2), n-propyl (C3), isopropyl (C3), n- butyl (C4), tert-butyl (C4), sec-butyl (C4), iso-butyl (C4), n-pentyl (C5), 3-pentanyl (C5), amyl (C5), neopentyl (C5), 3-methyl-2-butanyl (C5), tertiary amyl (C5), and n-hexyl (Cg). Additional examples of alkyl groups include n-heptyl (C7), n-octyl (Cg) and the like. Unless otherwise specified, each instance of an alkyl group is independently unsubstituted (an "unsubstituted alkyl") or substituted (a "substituted alkyl") with one or more substituents. In certain embodiments, the alkyl group is an unsubstituted (C1-C50) alkyl. In certain embodiments, the alkyl group is a substituted (C1-C50) alkyl. [046] Affixing the suffix "-ene" to a group indicates the group is a divalent moiety, e.g., arylene is the divalent moiety of aryl, and heteroarylene is the divalent moiety of heteroaryl.
[047] Alkylene: The term "alkylene," as used herein, represents a saturated divalent straight or branched chain hydrocarbon group and is exemplified by methylene, ethylene, isopropylene and the like. Likewise, the term "alkenylene" as used herein represents an unsaturated divalent straight or branched chain hydrocarbon group having one or more unsaturated carbon-carbon double bonds that may occur in any stable point along the chain, and the term "alkynylene" herein represents an unsaturated divalent straight or branched chain hydrocarbon group having one or more unsaturated carbon-carbon triple bonds that may occur in any stable point along the chain. In certain embodiments, an alkylene, alkenylene, or alkynylene group may comprise one or more cyclic aliphatic and/or one or more heteroatoms such as oxygen, nitrogen, or sulfur and may optionally be substituted with one or more substituents such as alkyl, halo, alkoxyl, hydroxy, amino, aryl, ether, ester or amide. For example, an alkylene, alkenylene, or alkynylene may be substituted with one or more (e.g., 1, 2, 3, 4, 5, or 6 independently selected substituents) of halogen, -COR", -CO2H, -CO2R", -CN, -OH, -OR", -OCOR", -OCO2R", -NH2, -NHR", -N(R")2, -SR" or -SO2R", wherein each instance of R" independently is (C1-C20) aliphatic (e.g., (C1-C20) alkyl, (C1-C15) alkyl, (C1-C10) alkyl, or (C1-C3) alkyl). In embodiments, R" independently is an unsubstituted alkyl (e.g., unsubstituted (C1-C20) alkyl, (C1-C15) alkyl, (C1-C10) alkyl, or (C1-C3) alkyl). In embodiments, R" independently is unsubstituted (C1-C3) alkyl. In certain embodiments, an alkylene, alkenylene, or alkynylene is unsubstituted. In certain embodiments, an alkylene, alkenylene, or alkynylene does not include any heteroatoms.
[048] Alkenyl: As used herein, "alkenyl" means any linear or branched hydrocarbon chains having one or more unsaturated carbon-carbon double bonds that may occur in any stable point along the chain, e.g. "(C2-C30) alkenyl" refers to an alkenyl group having 2-30 carbons. For example, an alkenyl group includes prop-2-enyl, but-2-enyl, but-3-enyl, 2-methylprop-2-enyl, hex-2-enyl, hex-5-enyl, 2,3-
dimethylbut-2-enyl, and the like. In embodiments, the alkenyl comprises 1, 2, or 3 carbon-carbon double bond. In embodiments, the alkenyl comprises a single carbon-carbon double bond. In embodiments, multiple double bonds (e.g., 2 or 3) are conjugated. An alkenyl group may be unsubstituted or substituted with one or more substituent groups as described herein. For example, an alkenyl group may be substituted with one or more (e.g., 1, 2, 3, 4, 5, or 6 independently selected substituents) of halogen, -COR’’, -CO2H, -CO2R’’, -CN, -OH, -OR’’, -OCOR’’, -OCO2R’’, -NH2, -NHR’’, - N(R’’)2, -SR’’ or -SO2R’’, wherein each instance of R’’ independently is (C1-C20) aliphatic (e.g., (C1-C20) alkyl, (C1-C15) alkyl, (C1-C10) alkyl, or (C1-C3) alkyl). In embodiments, R’’ independently is an unsubstituted alkyl (e.g., unsubstituted (C1-C20) alkyl, (C1-C15) alkyl, (C1-C10) alkyl, or (C1-C3) alkyl). In embodiments, R’’ independently is unsubstituted (C1-C3) alkyl. In embodiments, the alkenyl is unsubstituted. In embodiments, the alkenyl is substituted (e.g., with 1, 2, 3, 4, 5, or 6 substituent groups as described herein). In embodiments, an alkenyl group is substituted with a–OH group and may also be referred to herein as a “hydroxyalkenyl” group, where the prefix denotes the –OH group and “alkenyl” is as described herein. [049] As used herein, “alkenyl” also refers to a radical of a straight-chain or branched hydrocarbon group having from 2 to 50 carbon atoms and one or more carbon-carbon double bonds (e.g., 1, 2, 3, or 4 double bonds) (“(C2-C50) alkenyl”). In some embodiments, an alkenyl group has 2 to 40 carbon atoms (“(C2-C40) alkenyl”). In some embodiments, an alkenyl group has 2 to 30 carbon atoms (“(C2- C30) alkenyl”). In some embodiments, an alkenyl group has 2 to 20 carbon atoms (“(C2-C20) alkenyl”). In some embodiments, an alkenyl group has 2 to 10 carbon atoms (“(C2-C10) alkenyl”). In some embodiments, an alkenyl group has 2 to 9 carbon atoms (“(C2-C9) alkenyl”). In some embodiments, an alkenyl group has 2 to 8 carbon atoms (“(C2-C8) alkenyl”). In some embodiments, an alkenyl group has 2 to 7 carbon atoms (“(C2-C7) alkenyl”). In some embodiments, an alkenyl group has 2 to 6 carbon atoms (“(C2-C6) alkenyl”). In some embodiments, an alkenyl group has 2 to 5 carbon atoms (“(C2-C5) alkenyl”). In some embodiments, an alkenyl group has 2 to 4 carbon atoms (“(C2-C4) alkenyl”). In some embodiments, an alkenyl group has 2 to 3 carbon atoms (“(C2-C3) alkenyl”). In some embodiments, an alkenyl group has 2 carbon atoms (“(C2) alkenyl”). The one or more carbon- carbon double bonds can be internal (such as in 2-butenyl) or terminal (such as in 1-butenyl). Examples of (C2-C4) alkenyl groups include, without limitation, ethenyl (C2), 1-propenyl (C3), 2- propenyl (C3), 1-butenyl (C4), 2-butenyl (C4), butadienyl (C4), and the like. Examples of (C2-C6) alkenyl groups include the aforementioned (C2-C4) alkenyl groups as well as pentenyl (C5), pentadienyl (C5), hexenyl (C6), and the like. Additional examples of alkenyl include heptenyl (C7), octenyl (C8), octatrienyl (C8), and the like. Unless otherwise specified, each instance of an alkenyl group is independently unsubstituted (an “unsubstituted alkenyl”) or substituted (a “substituted alkenyl”)
with one or more substituents. In certain embodiments, the alkenyl group is an unsubstituted (C2- C50) alkenyl. In certain embodiments, the alkenyl group is a substituted (C2-C50) alkenyl.
[050] Alkynyl: As used herein, "alkynyl" means any hydrocarbon chain of either linear or branched configuration, having one or more carbon-carbon triple bonds occurring in any stable point along the chain, e.g., "(C2-C30) alkynyl", refers to an alkynyl group having 2-30 carbons. Examples of an alkynyl group include prop-2-ynyl, but-2-ynyl, but-3-ynyl, pent-2-ynyl, 3-methylpent-4-ynyl, hex-2-ynyl, hex- 5-ynyl, etc. In embodiments, an alkynyl comprises one carbon-carbon triple bond. An alkynyl group may be unsubstituted or substituted with one or more substituent groups as described herein. For example, an alkynyl group may be substituted with one or more (e.g., 1, 2, 3, 4, 5, or 6 independently selected substituents) of halogen, -COR", -CO2H, -CO2R", -CN, -OH, -OR", -OCOR", - OCO2R", -NH2, -NHR", -N(R")2, -SR" or -SO2R", wherein each instance of R" independently is (C1-C20) aliphatic (e.g., (C1-C20) alkyl, (C1-C15) alkyl, (C1-C10) alkyl, or (C1-C3) alkyl). In embodiments, R" independently is an unsubstituted alkyl (e.g., unsubstituted (C1-C20) alkyl, (C1-C15) alkyl, (C1-C10) alkyl, or (C1-C3) alkyl). In embodiments, R" independently is unsubstituted (C1-C3) alkyl. In embodiments, the alkynyl is unsubstituted. In embodiments, the alkynyl is substituted (e.g., with 1, 2, 3, 4, 5, or 6 substituent groups as described herein).
[051] As used herein, "alkynyl" also refers to a radical of a straight-chain or branched hydrocarbon group having from 2 to 50 carbon atoms and one or more carbon-carbon triple bonds (e.g., 1, 2, 3, or 4 triple bonds) and optionally one or more double bonds (e.g., 1, 2, 3, or 4 double bonds) ("(C2-C50) alkynyl"). An alkynyl group that has one or more triple bonds, and one or more double bonds is also referred to as an "ene-yne". In some embodiments, an alkynyl group has 2 to 40 carbon atoms ("(C2- C40) alkynyl"). In some embodiments, an alkynyl group has 2 to 30 carbon atoms ("(C2-C30) alkynyl"). In some embodiments, an alkynyl group has 2 to 20 carbon atoms ("(C2-C20) alkynyl"). In some embodiments, an alkynyl group has 2 to 10 carbon atoms ("(C2-C10) alkynyl"). In some embodiments, an alkynyl group has 2 to 9 carbon atoms ("(C2-C9) alkynyl"). In some embodiments, an alkynyl group has 2 to 8 carbon atoms ("(C2-Cg) alkynyl"). In some embodiments, an alkynyl group has 2 to 7 carbon atoms ("(C2-C7) alkynyl"). In some embodiments, an alkynyl group has 2 to 6 carbon atoms ("(C2-C6) alkynyl"). In some embodiments, an alkynyl group has 2 to 5 carbon atoms ("(C2-C5) alkynyl"). In some embodiments, an alkynyl group has 2 to 4 carbon atoms ("(C2-C4) alkynyl"). In some embodiments, an alkynyl group has 2 to 3 carbon atoms ("(C2-C3) alkynyl"). In some embodiments, an alkynyl group has 2 carbon atoms ("(C2) alkynyl"). The one or more carbon- carbon triple bonds can be internal (such as in 2-butynyl) or terminal (such as in 1-butynyl).
Examples of (C2-C4) alkynyl groups include, without limitation, ethynyl (C2), 1-propynyl (C3), 2- propynyl (C3), 1-butynyl (C4), 2-butynyl (C4), and the like. Examples of (C2-Cg) alkenyl groups include
the aforementioned (C2-C4) alkynyl groups as well as pentynyl (C5), hexynyl (C6), and the like. Additional examples of alkynyl include heptynyl (C7), octynyl (C8), and the like. Unless otherwise specified, each instance of an alkynyl group is independently unsubstituted (an “unsubstituted alkynyl”) or substituted (a “substituted alkynyl”) with one or more substituents. In certain embodiments, the alkynyl group is an unsubstituted (C2-C50) alkynyl. In certain embodiments, the alkynyl group is a substituted (C2-C50) alkynyl. [052] Aryl: The term “aryl” used alone or as part of a larger moiety as in “aralkyl,” refers to a monocyclic, bicyclic, or tricyclic carbocyclic ring system having a total of six to fourteen ring members, wherein said ring system has a single point of attachment to the rest of the molecule, at least one ring in the system is aromatic and wherein each ring in the system contains 4 to 7 ring members. In embodiments, an aryl group has 6 ring carbon atoms (“(C6) aryl,” e.g., phenyl). In some embodiments, an aryl group has 10 ring carbon atoms (“(C10) aryl,” e.g., naphthyl such as 1-naphthyl and 2-naphthyl). In some embodiments, an aryl group has 14 ring carbon atoms (“(C14) aryl,” e.g., anthracyl). “Aryl” also includes ring systems wherein the aryl ring, as defined above, is fused with one or more carbocyclyl or heterocyclyl groups wherein the radical or point of attachment is on the aryl ring, and in such instances, the number of carbon atoms continue to designate the number of carbon atoms in the aryl ring system. Exemplary aryls include phenyl, naphthyl, and anthracene. [053] As used herein, “aryl” also refers to a radical of a monocyclic or polycyclic (e.g., bicyclic or tricyclic) 4n+2 aromatic ring system (e.g., having 6, 10, or 14 π electrons shared in a cyclic array) having 6-14 ring carbon atoms and zero heteroatoms provided in the aromatic ring system (“(C6-C14) aryl”). In some embodiments, an aryl group has 6 ring carbon atoms (“(C6) aryl”; e.g., phenyl). In some embodiments, an aryl group has 10 ring carbon atoms (“(C10) aryl”; e.g., naphthyl such as 1- naphthyl and 2-naphthyl). In some embodiments, an aryl group has 14 ring carbon atoms (“(C14) aryl”; e.g., anthracyl). “Aryl” also includes ring systems wherein the aryl ring, as defined above, is fused with one or more carbocyclyl or heterocyclyl groups wherein the radical or point of attachment is on the aryl ring, and in such instances, the number of carbon atoms continue to designate the number of carbon atoms in the aryl ring system. Unless otherwise specified, each instance of an aryl group is independently unsubstituted (an “unsubstituted aryl”) or substituted (a “substituted aryl”) with one or more substituents. In certain embodiments, the aryl group is an unsubstituted (C6-C14) aryl. In certain embodiments, the aryl group is a substituted (C6-C14) aryl. [054] Arylene: The term “arylene” as used herein refers to an aryl group that is divalent (that is, having two points of attachment to the molecule). Exemplary arylenes include phenylene (e.g., unsubstituted phenylene or substituted phenylene).
[055] Carbocyclyl: As used herein, “carbocyclyl” or “carbocyclic” refers to a radical of a non- aromatic cyclic hydrocarbon group having from 3 to 10 ring carbon atoms (“(C3-C10) carbocyclyl”) and zero heteroatoms in the non-aromatic ring system. In some embodiments, a carbocyclyl group has 3 to 8 ring carbon atoms (“(C3-C8) carbocyclyl”). In some embodiments, a carbocyclyl group has 3 to 7 ring carbon atoms (“(C3-C7) carbocyclyl”). In some embodiments, a carbocyclyl group has 3 to 6 ring carbon atoms (“(C3-C6) carbocyclyl”). In some embodiments, a carbocyclyl group has 4 to 6 ring carbon atoms (“(C4-C6) carbocyclyl”). In some embodiments, a carbocyclyl group has 5 to 6 ring carbon atoms (“(C5-C6) carbocyclyl”). In some embodiments, a carbocyclyl group has 5 to 10 ring carbon atoms (“(C5-C10) carbocyclyl”). Exemplary (C3-C6) carbocyclyl groups include, without limitation, cyclopropyl (C3), cyclopropenyl (C3), cyclobutyl (C4), cyclobutenyl (C4), cyclopentyl (C5), cyclopentenyl (C5), cyclohexyl (C6), cyclohexenyl (C6), cyclohexadienyl (C6), and the like. Exemplary (C3-C8) carbocyclyl groups include, without limitation, the aforementioned (C3-C6) carbocyclyl groups as well as cycloheptyl (C7), cycloheptenyl (C7), cycloheptadienyl (C7), cycloheptatrienyl (C7), cyclooctyl (C8), cyclooctenyl (C8), bicyclo[2.2.1]heptanyl (C7), bicyclo[2.2.2]octanyl (C8), and the like. Exemplary (C3-C10) carbocyclyl groups include, without limitation, the aforementioned (C3-C8) carbocyclyl groups as well as cyclononyl (C9), cyclononenyl (C9), cyclodecyl (C10), cyclodecenyl (C10), octahydro-1H-indenyl (C9), decahydronaphthalenyl (C10), spiro[4.5]decanyl (C10), and the like. As the foregoing examples illustrate, in certain embodiments, the carbocyclyl group is either monocyclic (“monocyclic carbocyclyl”) or polycyclic (e.g., containing a fused, bridged or spiro ring system such as a bicyclic system (“bicyclic carbocyclyl”) or tricyclic system (“tricyclic carbocyclyl”)) and can be saturated or can contain one or more carbon-carbon double or triple bonds. “Carbocyclyl” also includes ring systems wherein the carbocyclyl ring, as defined above, is fused with one or more aryl or heteroaryl groups wherein the point of attachment is on the carbocyclyl ring, and in such instances, the number of carbons continue to designate the number of carbons in the carbocyclic ring system. Unless otherwise specified, each instance of a carbocyclyl group is independently unsubstituted (an “unsubstituted carbocyclyl”) or substituted (a “substituted carbocyclyl”) with one or more substituents. In certain embodiments, the carbocyclyl group is an unsubstituted C3-C10 carbocyclyl. In certain embodiments, the carbocyclyl group is a substituted (C3-C10) carbocyclyl. [056] In some embodiments, “carbocyclyl” or “carbocyclic” is referred to as a “cycloalkyl”, i.e., a monocyclic, saturated carbocyclyl group having from 3 to 10 ring carbon atoms (“(C3-C10) cycloalkyl”). In some embodiments, a cycloalkyl group has 3 to 8 ring carbon atoms (“(C3-C8) cycloalkyl”). In some embodiments, a cycloalkyl group has 3 to 6 ring carbon atoms (“(C3-C6), cycloalkyl”). In some embodiments, a cycloalkyl group has 4 to 6 ring carbon atoms (“(C4-C6)
cycloalkyl”). In some embodiments, a cycloalkyl group has 5 to 6 ring carbon atoms (“(C5-C6) cycloalkyl”). In some embodiments, a cycloalkyl group has 5 to 10 ring carbon atoms (“(C5-C10) cycloalkyl”). Examples of (C5-C6) cycloalkyl groups include cyclopentyl (C5) and cyclohexyl (C5). Examples of (C3-C6) cycloalkyl groups include the aforementioned (C5-C6) cycloalkyl groups as well as cyclopropyl (C3) and cyclobutyl (C4). Examples of (C3-C8) cycloalkyl groups include the aforementioned (C3-C6) cycloalkyl groups as well as cycloheptyl (C7) and cyclooctyl (C8). Unless otherwise specified, each instance of a cycloalkyl group is independently unsubstituted (an “unsubstituted cycloalkyl”) or substituted (a “substituted cycloalkyl”) with one or more substituents. In certain embodiments, the cycloalkyl group is an unsubstituted (C3-C10) cycloalkyl. In certain embodiments, the cycloalkyl group is a substituted (C3-C10) cycloalkyl. [057] Heteroalkyl: The term “heteroalkyl” is meant a branched or unbranched alkyl, alkenyl, or alkynyl group having from 1 to 14 carbon atoms in addition to 1, 2, 3 or 4 heteroatoms independently selected from the group consisting of N, O, S, and P. Heteroalkyls include tertiary amines, secondary amines, ethers, thioethers, amides, thioamides, carbamates, thiocarbamates, hydrazones, imines, phosphodiesters, phosphoramidates, sulfonamides, and disulfides. A heteroalkyl group may optionally include monocyclic, bicyclic, or tricyclic rings, in which each ring desirably has three to six members. Examples of heteroalkyls include polyethers, such as methoxymethyl and ethoxyethyl. [058] Heteroalkylene: The term “heteroalkylene,” as used herein, represents a divalent form of a heteroalkyl group as described herein. [059] Heteroaryl: The term “heteroaryl,” as used herein, represents a fully unsaturated heteroatom-containing ring wherein at least one ring atom is a heteroatom such as, but not limited to, nitrogen and oxygen. [060] As used herein, “heteroaryl” also refers to a radical of a 5-14 membered monocyclic or polycyclic (e.g., bicyclic or tricyclic) 4n+2 aromatic ring system (e.g., having 6, 10, or 14 π electrons shared in a cyclic array) having ring carbon atoms and 1 or more (e.g., 1, 2, 3, or 4 ring heteroatoms) ring heteroatoms provided in the aromatic ring system, wherein each heteroatom is independently selected from oxygen, sulfur, nitrogen, boron, silicon, and phosphorus (“5-14 membered heteroaryl”). In heteroaryl groups that contain one or more nitrogen atoms, the point of attachment can be a carbon or nitrogen atom, as valency permits. Heteroaryl polycyclic ring systems can include one or more heteroatoms in one or both rings. “Heteroaryl” includes ring systems wherein the heteroaryl ring, as defined above, is fused with one or more carbocyclyl or heterocyclyl groups wherein the point of attachment is on the heteroaryl ring, and in such instances, the number of ring members continue to designate the number of ring members in the heteroaryl ring system.
“Heteroaryl” also includes ring systems wherein the heteroaryl ring, as defined above, is fused with one or more aryl groups wherein the point of attachment is either on the aryl or heteroaryl ring, and in such instances, the number of ring members designates the number of ring members in the fused polycyclic (aryl/heteroaryl) ring system. Polycyclic heteroaryl groups wherein one ring does not contain a heteroatom (e.g., indolyl, quinolinyl, carbazolyl, and the like) the point of attachment can be on either ring, i.e., either the ring bearing a heteroatom (e.g., 2-indolyl) or the ring that does not contain a heteroatom (e.g., 5-indolyl). [061] In some embodiments, a heteroaryl group is a 5-10 membered aromatic ring system having ring carbon atoms and 1 or more (e.g., 1, 2, 3, or 4) ring heteroatoms provided in the aromatic ring system, wherein each heteroatom is independently selected from oxygen, sulfur, nitrogen, boron, silicon, and phosphorus (“5-10 membered heteroaryl”). In some embodiments, a heteroaryl group is a 5-8 membered aromatic ring system having ring carbon atoms and 1 or more (e.g., 1, 2, 3, or 4) ring heteroatoms provided in the aromatic ring system, wherein each heteroatom is independently selected from oxygen, sulfur, nitrogen, boron, silicon, and phosphorus (“5-8 membered heteroaryl”). In some embodiments, a heteroaryl group is a 5-6 membered aromatic ring system having ring carbon atoms and 1 or more (e.g., 1, 2, 3, or 4) ring heteroatoms provided in the aromatic ring system, wherein each heteroatom is independently selected from oxygen, sulfur, nitrogen, boron, silicon, and phosphorus (“5-6 membered heteroaryl”). In some embodiments, the 5-6 membered heteroaryl has 1 or more (e.g., 1, 2, or 3) ring heteroatoms selected from oxygen, sulfur, nitrogen, boron, silicon, and phosphorus. In some embodiments, the 5-6 membered heteroaryl has 1 or 2 ring heteroatoms selected from oxygen, sulfur, nitrogen, boron, silicon, and phosphorus. In some embodiments, the 5-6 membered heteroaryl has 1 ring heteroatom selected from oxygen, sulfur, nitrogen, boron, silicon, and phosphorus. Unless otherwise specified, each instance of a heteroaryl group is independently unsubstituted (an “unsubstituted heteroaryl”) or substituted (a “substituted heteroaryl”) with one or more substituents. In certain embodiments, the heteroaryl group is an unsubstituted 5-14 membered heteroaryl. In certain embodiments, the heteroaryl group is a substituted 5-14 membered heteroaryl. [062] Exemplary 5-membered heteroaryl groups containing 1 heteroatom include, without limitation, pyrrolyl, furanyl and thiophenyl. Exemplary 5-membered heteroaryl groups containing 2 heteroatoms include, without limitation, imidazolyl, pyrazolyl, oxazolyl, isoxazolyl, thiazolyl, and isothiazolyl. Exemplary 5-membered heteroaryl groups containing 3 heteroatoms include, without limitation, triazolyl, oxadiazolyl, and thiadiazolyl. Exemplary 5-membered heteroaryl groups containing 4 heteroatoms include, without limitation, tetrazolyl. Exemplary 6-membered heteroaryl groups containing 1 heteroatom include, without limitation, pyridinyl. Exemplary 6-membered
heteroaryl groups containing 2 heteroatoms include, without limitation, pyridazinyl, pyrimidinyl, and pyrazinyl. Exemplary 6-membered heteroaryl groups containing 3 or 4 heteroatoms include, without limitation, triazinyl and tetrazinyl, respectively. Exemplary 7-membered heteroaryl groups containing 1 heteroatom include, without limitation, azepinyl, oxepinyl, and thiepinyl. Exemplary 5,6-bicyclic heteroaryl groups include, without limitation, indolyl, isoindolyl, indazolyl, benzotriazolyl, benzothiophenyl, isobenzothiophenyl, benzofuranyl, benzoisofuranyl, benzimidazolyl, benzoxazolyl, benzisoxazolyl, benzoxadiazolyl, benzthiazolyl, benzisothiazolyl, benzthiadiazolyl, indolizinyl, and purinyl. Exemplary 6,6-bicyclic heteroaryl groups include, without limitation, naphthyridinyl, pteridinyl, quinolinyl, isoquinolinyl, cinnolinyl, quinoxalinyl, phthalazinyl, and quinazolinyl. Exemplary tricyclic heteroaryl groups include, without limitation, phenanthridinyl, dibenzofuranyl, carbazolyl, acridinyl, phenothiazinyl, phenoxazinyl and phenazinyl. [063] As used herein, “heterocyclyl” or “heterocyclic” refers to a radical of a 3- to 14-membered non-aromatic ring system having ring carbon atoms and 1 or more (e.g., 1, 2, 3, or 4) ring heteroatoms, wherein each heteroatom is independently selected from oxygen, sulfur, nitrogen, boron, silicon, and phosphorus (“3-14 membered heterocyclyl”). In heterocyclyl groups that contain one or more nitrogen atoms, the point of attachment can be a carbon or nitrogen atom, as valency permits. A heterocyclyl group can either be monocyclic (“monocyclic heterocyclyl”) or polycyclic (e.g., a fused, bridged or spiro ring system such as a bicyclic system (“bicyclic heterocyclyl”) or tricyclic system (“tricyclic heterocyclyl”)) and can be saturated or can contain one or more carbon- carbon double or triple bonds. Heterocyclyl polycyclic ring systems can include one or more heteroatoms in one or both rings. “Heterocyclyl” also includes ring systems wherein the heterocyclyl ring, as defined above, is fused with one or more carbocyclyl groups wherein the point of attachment is either on the carbocyclyl or heterocyclyl ring, or ring systems wherein the heterocyclyl ring, as defined above, is fused with one or more aryl or heteroaryl groups, wherein the point of attachment is on the heterocyclyl ring, and in such instances, the number of ring members continue to designate the number of ring members in the heterocyclyl ring system. Unless otherwise specified, each instance of heterocyclyl is independently unsubstituted (an “unsubstituted heterocyclyl”) or substituted (a “substituted heterocyclyl”) with one or more substituents. In certain embodiments, the heterocyclyl group is an unsubstituted 3-14 membered heterocyclyl. In certain embodiments, the heterocyclyl group is a substituted 3-14 membered heterocyclyl. [064] In some embodiments, a heterocyclyl group is a 5-10 membered non-aromatic ring system having ring carbon atoms and 1 or more (e.g., 1, 2, 3, or 4) ring heteroatoms, wherein each heteroatom is independently selected from oxygen, sulfur, nitrogen, boron, silicon, and phosphorus (“5-10 membered heterocyclyl”). In some embodiments, a heterocyclyl group is a 5-8 membered
non-aromatic ring system having ring carbon atoms and 1 or more (e.g., 1, 2, 3, or 4) ring heteroatoms, wherein each heteroatom is independently selected from oxygen, sulfur, nitrogen, boron, silicon, and phosphorus (“5-8 membered heterocyclyl”). In some embodiments, a heterocyclyl group is a 5-6 membered non-aromatic ring system having ring carbon atoms and 1 or more (e.g., 1, 2, 3, or 4) ring heteroatoms, wherein each heteroatom is independently selected from oxygen, sulfur, nitrogen, boron, silicon, and phosphorus (“5-6 membered heterocyclyl”). In some embodiments, the 5-6 membered heterocyclyl has 1 or more (e.g., 1, 2, or 3) ring heteroatoms selected from oxygen, sulfur, nitrogen, boron, silicon, and phosphorus. In some embodiments, the 5- 6 membered heterocyclyl has 1 or 2 ring heteroatoms selected from oxygen, sulfur, nitrogen, boron, silicon, and phosphorus. In some embodiments, the 5-6 membered heterocyclyl has 1 ring heteroatom selected from oxygen, sulfur, nitrogen, boron, silicon, and phosphorus. [065] Exemplary 3-membered heterocyclyl groups containing 1 heteroatom include, without limitation, azirdinyl, oxiranyl, thiorenyl. Exemplary 4-membered heterocyclyl groups containing 1 heteroatom include, without limitation, azetidinyl, oxetanyl and thietanyl. Exemplary 5-membered heterocyclyl groups containing 1 heteroatom include, without limitation, tetrahydrofuranyl, dihydrofuranyl, tetrahydrothiophenyl, dihydrothiophenyl, pyrrolidinyl, dihydropyrrolyl and pyrrolyl- 2,5-dione. Exemplary 5- membered heterocyclyl groups containing 2 heteroatoms include, without limitation, dioxolanyl, oxathiolanyl and dithiolanyl. Exemplary 5-membered heterocyclyl groups containing 3 heteroatoms include, without limitation, triazolinyl, oxadiazolinyl, and thiadiazolinyl. Exemplary 6-membered heterocyclyl groups containing 1 heteroatom include, without limitation, piperidinyl, tetrahydropyranyl, dihydropyridinyl, and thianyl. Exemplary 6-membered heterocyclyl groups containing 2 heteroatoms include, without limitation, piperazinyl, morpholinyl, dithianyl, dioxanyl. Exemplary 6-membered heterocyclyl groups containing 2 heteroatoms include, without limitation, triazinanyl. Exemplary 7-membered heterocyclyl groups containing 1 heteroatom include, without limitation, azepanyl, oxepanyl and thiepanyl. Exemplary 8-membered heterocyclyl groups containing 1 heteroatom include, without limitation, azocanyl, oxecanyl and thiocanyl. Exemplary bicyclic heterocyclyl groups include, without limitation, indolinyl, isoindolinyl, dihydrobenzofuranyl, dihydrobenzothienyl, tetrahydrobenzothienyl, tetrahydrobenzofuranyl, tetrahydroindolyl, tetrahydroquinolinyl, tetrahydroisoquinolinyl, decahydroquinolinyl, decahydroisoquinolinyl, octahydrochromenyl, octahydroisochromenyl, decahydronaphthyridinyl, decahydro-1,8- naphthyridinyl, octahydropyrrolo[3,2-b]pyrrole, indolinyl, phthalimidyl, naphthalimidyl, chromanyl, chromenyl, 1H-benzo[e][1,4]diazepinyl, 1,4,5,7-tetrahydropyrano[3,4-b] pyrrolyl, 5,6-dihydro-4H- furo[3,2-b]pyrrolyl, 6,7-dihydro-5H-furo[3,2-b]pyranyl, 5,7-dihydro-4H-thieno[2,3-c]pyranyl, 2,3-dihydro- 1H-pyrrolo[2,3-b ]pyridinyl, 2,3-dihydrofuro[2,3-b]pyridinyl, 4,5,6,7-tetrahydro-1H-pyrrolo-[2,3-
b]pyridinyl, 4,5,6,7-tetrahydrofuro[3,2-c]pyridinyl, 4,5,6,7-tetrahydrothieno [3,2- b]pyridinyl, 1,2,3,4- tetrahydro-1,6-naphthyridinyl, and the like. [066] Heterocycloalkyl: The term “heterocycloalkyl,” as used herein, is a non-aromatic ring wherein at least one atom is a heteroatom such as, but not limited to, nitrogen, oxygen, sulfur, or phosphorus, and the remaining atoms are carbon. The heterocycloalkyl group can be substituted or unsubstituted. [067] As understood from the above, alkyl, alkenyl, alkynyl, acyl, carbocyclyl, heterocyclyl, aryl, and heteroaryl groups, as defined herein, are, in certain embodiments, optionally substituted. Optionally substituted refers to a group which may be substituted or unsubstituted (e.g., “substituted” or “unsubstituted” alkyl, “substituted” or “unsubstituted” alkenyl, “substituted” or “unsubstituted” alkynyl, “substituted” or “unsubstituted” heteroalkyl, “substituted” or “unsubstituted” heteroalkenyl, “substituted” or ’unsubstituted” heteroalkynyl, “substituted” or “unsubstituted” carbocyclyl, “substituted” or “unsubstituted” heterocyclyl, “substituted” or “unsubstituted” aryl or “substituted” or “unsubstituted” heteroaryl group. In general, the term “substituted” means that at least one hydrogen present on a group is replaced with a permissible substituent, e.g., a substituent which upon substitution results in a stable compound, e.g., a compound which does not spontaneously undergo transformation such as by rearrangement, cyclization, elimination, or other reaction. Unless otherwise indicated, a “substituted” group has a substituent at one or more substitutable positions of the group, and when more than one position in any given structure is substituted, the substituent is either the same or different at each position. The term “substituted” is contemplated to include substitution with all permissible substituents of organic compounds, any of the substituents described herein that results in the formation of a stable compound. The present invention contemplates any and all such combinations in order to arrive at a stable compound. For purposes of this invention, heteroatoms such as nitrogen may have hydrogen substituents and/or any suitable substituent as described herein which satisfy the valences of the heteroatoms and results in the formation of a stable moiety. [068] Exemplary carbon atom substituents include, but are not limited to, halogen, -CN, - NO2, -N3, -SO2, -SO3H, -OH, -ORaa, -ON(Rbb)2, -N(Rbb)2, -N(Rbb)3+X-, -N(ORcc)Rbb, -SeH, -SeRaa, -SH, -SRaa, -SSRcc, -C(=O)Raa, -CO2H, -CHO, -C(ORcc)2, -CO2Raa, -OC(=O)Raa, -OCO2Raa, -C(=O)N(Rbb)2, - OC(=O)N(Rbb)2, -NRbbC(=O)Raa, -NRbbCO2Raa, -NRbbC(=O)N(Rbb)2, -C(=NRbb)Raa, -C(=NRbb)ORaa, - OC(=NRbb)Raa, - OC(=NRbb)ORaa, -C(=NRbb)N(Rbb)2, -OC(=NRbb)N(Rbb)2, -NRbbC(=NRbb)N(Rbb)2, - C(=O)NRbbSO2Raa, -NRbbSO2Raa, -SO2N(Rbb)2, -SO2Raa, -SO2ORaa, -OSO2Raa, -S(=O)Raa, -OS(=O)Raa, - Si(Raa)3 -Osi(Raa)3 -C(=S)N(Rbb)2, -C(=O)SRaa, -C(=S)SRaa, - SC(=S)SRaa, -SC(=O)SRaa, -OC(=O)SRaa, -
SC(=O)ORaa, -SC(=O)Raa, -P(=O)2Raa, -OP(=O)2Raa, -P(=O)(Raa)2, -OP(=O)(Raa)2, -OP(=O)(ORcc)2, - P(=O)2N(Rbb)2, -OP(=O)2N(Rbb)2, - P(=O)(NRbb)2, -OP(=O)(NRbb)2, -NRbbP(=O)(ORcc)2, - NRbbP(=O)(NRbb)2, -P(Rcc)2, - P(Rcc)3, -OP(Rcc)2, -OP(Rcc)3, -B(Raa)2, -B(ORcc)2, -BRaa(ORcc), (C1-C50) alkyl, (C2-C50) alkenyl, (C2-C50) alkynyl, (C3-C14) carbocyclyl, 3-14 membered heterocyclyl, (C6- C14) aryl, and 5-14 membered heteroaryl, wherein each alkyl, alkenyl, alkynyl, carbocyclyl, heterocyclyl, aryl, and heteroaryl is independently substituted with 0, 1, 2, 3, 4, or 5 R dd groups; or two geminal hydrogens on a carbon atom are replaced with the group =O, =S, =NN(Rbb)2, =NNRbbC(=O)Raa, =NNRbbC(=O)ORaa, =NNRbbS(=O)2Raa, =NRbb, or =NORcc; [069] each instance of Raa is, independently, selected from (C1-C50) alkyl, (C2-C50) alkenyl, (C2-C50) alkynyl, (C3-C10) carbocyclyl, 3-14 membered heterocyclyl, (C6-C14) aryl, and 5-14 membered heteroaryl, or two Raa groups are joined to form a 3-14 membered heterocyclyl or 5-14 membered heteroaryl ring, wherein each alkyl, alkenyl, alkynyl, carbocyclyl, heterocyclyl, aryl, and heteroaryl is independently substituted with 0, 1, 2, 3, 4, or 5 Rdd groups; [070] each instance of Rbb is, independently, selected from hydrogen, -OH, -ORaa, - N(Rcc)2, -CN, - C(=O)Raa, -C(=O)N(Rcc)2, -CO2Raa, -SO2Raa, -C(=NRcc)ORaa, - C(=NRcc)N(Rcc)2, -SO2N(Rcc)2, -SO2Rcc, - SO2ORcc, -SORaa, -C(=S)N(Rcc)2, -C(=O)SRcc, - C(=S)SRcc, -P(=O)2Raa, -P(=O)(Raa)2, -P(=O)2N(Rcc)2, - P(=O)(NRcc)2, (C1-C50) alkyl, (C2-C50) alkenyl, (C2-C50) alkynyl, (C3-C10) carbocyclyl, 3-14 membered heterocyclyl, (C6-C14) aryl, and 5-14 membered heteroaryl, or two Rbb groups, together with the heteroatom to which they are attached, form a 3-14 membered heterocyclyl or 5-14 membered heteroaryl ring, wherein each alkyl, alkenyl, alkynyl, carbocyclyl, heterocyclyl, aryl, and heteroaryl is independently substituted with 0, 1, 2, 3, 4, or 5 Rdd groups; [071] each instance of Rcc is, independently, selected from hydrogen, (C1-C50) alkyl, (C2-C50) alkenyl, (C2-C50) alkynyl, (C3-C10) carbocyclyl, 3-14 membered heterocyclyl, (C6-C14) aryl, and 5-14 membered heteroaryl, or two Rcc groups, together with the heteroatom to which they are attached, form a 3-14 membered heterocyclyl or 5-14 membered heteroaryl ring, wherein each alkyl, alkenyl, alkynyl, carbocyclyl, heterocyclyl, aryl, and heteroaryl is independently substituted with 0, 1, 2, 3, 4, or 5 Rdd groups; [072] each instance of Rdd is, independently, selected from halogen, -CN, -NO2, -N3, - SO2H, -SO3H, - OH, -ORee, -ON(Rff)2, -N(Rff)2, -N(Rff)3+X-, -N(ORee)Rff, -SH, -SRee, - SSRee, -C(=O)Ree, -CO2H, -CO2Ree, - OC(=O)Ree, -OCO2Ree, -C(=O)N(Rff)2, - OC(=O)N(Rff)2, -NRffC(=O)Ree, -NRffCO2Ree, -NRffC(=O)N(Rff)2, - C(=NRff)ORee, - OC(=NRff)Ree, -OC(=NRff)ORee, -C(=NRff)N(Rff)2, -OC(=NRff)N(Rff)2, -NRffC(=NRff)N(Rff)2, -NRffSO2Ree, -SO2N(Rff)2, -SO2Ree, -SO2ORee, -OSO2Ree, -S(=O)Ree, -Si(Ree)3, -Osi(Ree)3, - C(=S)N(Rff)2, -C(=O)SRee, -C(=S)SRee, -SC(=S)SRee, -P(=O)2Ree, - P(=O)(Ree)2, -OP(=O)(Ree)2, -
OP(=O)(ORee)2, (C1-C50) alkyl, (C2-C50) alkenyl, (C2-C50) alkynyl, (C3-C10) carbocyclyl, 3-10 membered heterocyclyl, (C6-C10) aryl, 5-10 membered heteroaryl, wherein each alkyl, alkenyl, alkynyl, carbocyclyl, heterocyclyl, aryl, and heteroaryl is independently substituted with 0, 1, 2, 3, 4, or 5 Rgg groups, or two geminal Rdd substituents can be joined to form =O or =S; [073] each instance of Ree is, independently, selected from (C1-C50) alkyl, (C2-C50) alkenyl, (C2-C50) alkynyl, (C3-C10) carbocyclyl, (C6-C10) aryl, 3-10 membered heterocyclyl, and 3-10 membered heteroaryl, wherein each alkyl, alkenyl, alkynyl, carbocyclyl, heterocyclyl, aryl, and heteroaryl is independently substituted with 0, 1, 2, 3, 4, or 5 Rgg groups; [074] each instance of Rff is, independently, selected from hydrogen, (C1-C50) alkyl, (C2-C50) alkenyl, (C2-C50) alkynyl, (C3-C10) carbocyclyl, 3-10 membered heterocyclyl, (C6-C10) aryl and 5-10 membered heteroaryl, or two Rff groups, together with the heteroatom to which they are attached, form a 3-14 membered heterocyclyl or 5-14 membered heteroaryl ring, wherein each alkyl, alkenyl, alkynyl, carbocyclyl, heterocyclyl, aryl, and heteroaryl is independently substituted with 0, 1, 2, 3, 4, or 5 Rgg groups; and [075] each instance of Rgg is, independently, halogen, -CN, -NO2, -N3, -SO2H, -SO3H, -OH, - O(C1-C50) alkyl, -ON((C1-C50) alkyl)2, -N((C1-C50) alkyl)2, -N((C1-C50) alkyl)3+X-, -NH((C1-C50) alkyl)2+X-, - NH2((C1-C50) alkyl) +X-, -NH3+X-, -N(O(C1-C50) alkyl)((C1-C50) alkyl), -N(OH)((C1-C50) alkyl), -NH(OH), -SH, -S(C1-C50) alkyl, -SS((C1-C50) alkyl), -C(=O)((C1-C50) alkyl), -CO2H, -CO2((C1-C50) alkyl), -OC(=O)((C1-C50) alkyl), -OCO2((C1-C50) alkyl), -C(=O)NH2, -C(=O)N((C1-C50) alkyl)2, -OC(=O)NH((C1-C50) alkyl), - NHC(=O)((C1-C50) alkyl), -N((C1-C50) alkyl)C(=O)((C1-C50) alkyl), -NHCO2((C1-C50) alkyl), - NHC(=O)N((C1-C50) alkyl)2, -NHC(=O)NH((C1-C50) alkyl), -NHC(=O)NH2, -C(=NH)O((C1-C50) alkyl),- OC(=NH)((C1-C50) alkyl), -OC(=NH)O(C1-C50) alkyl, - C(=NH)N((C1-C50) alkyl)2, -C(=NH)NH((C1-C50) alkyl), -C(=NH)NH2, -OC(=NH)N((C1-C50)alkyl)2, -OC(NH)NH((C1-C50) alkyl), -OC(NH)NH2, -NHC(NH)N((C1-C50) alkyl)2, -NHC(=NH)NH2, -NHSO2((C1-C50) alkyl), -SO2N((C1-C50) alkyl)2, -SO2NH((C1-C50) alkyl), - SO2NH2,- SO2((C1-C50) alkyl), -SO2O((C1-C50) alkyl), -OSO2((C1-C6) alkyl), -SO((C1-C6) alkyl), -Si((C1-C50) alkyl)3, - Osi((C1-C6) alkyl)3, -C(=S)N((C1-C50) alkyl)2, C(=S)NH((C1-C50) alkyl), C(=S)NH2, -C(=O)S((C1-C6) alkyl), - C(=S)S((C1-C6) alkyl), -SC(=S)S((C1-C6) alkyl), -P(=O)2((C1-C50) alkyl), -P(=O)((C1-C50) alkyl)2, -OP(=O)((C1- C50) alkyl)2, -OP(=O)(O(C1-C50) alkyl)2, (C1-C50) alkyl, (C2-C50) alkenyl, (C2-C50) alkynyl, (C3-C10) carbocyclyl, (C6-C10) aryl, 3-10 membered heterocyclyl, 5-10 membered heteroaryl; or two geminal Rgg substituents can be joined to form =O or =S; wherein X- is a counterion. [076] As used herein, the term “halo” or “halogen” refers to fluorine (fluoro, -F), chlorine (chloro, - Cl), bromine (bromo, -Br), or iodine (iodo, -I). [077] As used herein, a “counterion” is a negatively charged group associated with a positively charged quaternary amine in order to maintain electronic neutrality. Exemplary counterions include
halide ions (e.g., F-, Cl-, Br-, I-), NO3-, ClO4-, OH-, H2PO4-, HSO4-, sulfonate ions (e.g., methansulfonate, trifluoromethanesulfonate, p-toluenesulfonate, benzenesulfonate, 10-camphor sulfonate, naphthalene-2-sulfonate, naphthalene-l-sulfonic acid-5-sulfonate, ethan-1-sulfonic acid-2-sulfonate, and the like), and carboxylate ions (e.g., acetate, ethanoate, propanoate, benzoate, glycerate, lactate, tartrate, glycolate, and the like). [078] Nitrogen atoms can be substituted or unsubstituted as valency permits, and include primary, secondary, tertiary, and quaternary nitrogen atoms. Exemplary nitrogen atom substituents include, but are not limited to, hydrogen, -OH, -ORaa, -N(Rcc)2, -CN, - C(=O)Raa, -C(=O)N(Rcc)2, -CO2Raa, -SO2Raa, - C(=NRbb)Raa, -C(=NRcc)ORaa, - C(=NRcc)N(Rcc)2, -SO2N(Rcc)2, -SO2Rcc, -SO2ORcc, -SORaa, -C(=S)N(Rcc)2, - C(=O)SRcc, -C(=S)SRcc, -P(=O)2Raa, -P(=O)(Raa)2, -P(=O)2N(Rcc)2, -P(=O)(NRcc)2, (C1-C50) alkyl, (C2-C50) alkenyl, (C2-C50) alkynyl, (C3-C10) carbocyclyl, 3-14 membered heterocyclyl, (C6-C14) aryl, and 5-14 membered heteroaryl, or two Rcc groups, together with the N atom to which they are attached, form a 3-14 membered heterocyclyl or 5-14 membered heteroaryl ring, wherein each alkyl, alkenyl, alkynyl, carbocyclyl, heterocyclyl, aryl, and heteroaryl is independently substituted with 0, 1, 2, 3, 4, or 5 Rdd groups, and wherein Raa, Rbb, Rcc and Rdd are as defined above. [079] In certain embodiments, the substituent present on a nitrogen atom is a nitrogen protecting group (also referred to as an amino protecting group). Nitrogen protecting groups are well known in the art and include those described in detail in Protecting Groups in Organic Synthesis, T. W. Greene and P. G. M. Wuts, 3rd edition, John Wiley & Sons, 1999, incorporated herein by reference. [080] For example, nitrogen protecting groups such as amide groups (e.g., - C(=O)Raa) include, but are not limited to, formamide, acetamide, chloroacetamide, trichloroacetamide, trifluoroacetamide, phenylacetamide, 3-phenylpropanamide, picolinamide, 3-pyridylcarboxamide, N- benzoylphenylalanyl derivative, benzamide, p-phenylbenzamide, o-nitophenylacetamide, o- nitrophenoxyacetamide, acetoacetamide, (N’-dithiobenzyloxyacylamino)acetamide, 3-(p- hydroxyphenyl)propanamide, 3-(o-nitrophenyl)propanamide, 2-methyl-2-(o- nitrophenoxy)propanamide, 2-methyl-2-(o-phenylazophenoxy)propanamide, 4-chlorobutanamide, 3-methyl-3-nitrobutanamide, o-nitrocinnamide, N-acetylmethionine derivative, o-nitrobenzamide and o-(benzoyloxymethyl)benzamide. [081] Nitrogen protecting groups such as carbamate groups (e.g., -C(=O)ORaa) include, but are not limited to, methyl carbamate, ethyl carbamante, 9-fluorenylmethyl carbamate (Fmoc), 9-(2- sulfo)fluorenylmethyl carbamate, 9-(2,7-dibromo)fluoroenylmethyl carbamate, 2,7-di-t-butyl-[9- (10,10-dioxo-10,10,10,10-tetrahydrothioxanthyl)]methyl carbamate (DBD-Tmoc), 4- methoxyphenacyl carbamate (Phenoc), 2,2,2-trichloroethyl carbamate (Troc), 2-trimethylsilylethyl carbamate (Teoc), 2-phenylethyl carbamate (hZ), 1-(1-adamantyl)-1-methylethyl carbamate (Adpoc),
1,1-dimethyl-2-haloethyl carbamate, 1,1-dimethyl-2,2-dibromoethyl carbamate (DB-t-BOC), 1,1- dimethyl-2,2,2-trichloroethyl carbamate (TCBOC), 1-methyl-1-(4-biphenylyl)ethyl carbamate (Bpoc), 1-(3,5-di-t-butylphenyl)-1-methylethyl carbamate (t-Bumeoc), 2-(2’-and 4’-pyridyl)ethyl carbamate (Pyoc), 2-(N,N-dicyclohexylcarboxamido)ethyl carbamate, t-butyl carbamate (BOC), 1-adamantyl carbamate (Adoc), vinyl carbamate (Voc), allyl carbamate (Alloc), 1-isopropylallyl carbamate (Ipaoc), cinnamyl carbamate (Coc), 4-nitrocinnamyl carbamate (Noc), 8-quinolyl carbamate, N- hydroxypiperidinyl carbamate, alkyldithio carbamate, benzyl carbamate (Cbz), p-methoxybenzyl carbamate (Moz), p-nitobenzyl carbamate, p-bromobenzyl carbamate, p-chlorobenzyl carbamate, 2,4-dichlorobenzyl carbamate, 4-methylsulfinylbenzyl carbamate (Msz), 9-anthrylmethyl carbamate, diphenylmethyl carbamate, 2-methylthioethyl carbamate, 2-methylsulfonylethyl carbamate, 2-(p- toluenesulfonyl)ethyl carbamate, [2-(1,3-dithianyl)]methyl carbamate (Dmoc), 4- methylthiophenyl carbamate (Mtpc), 2,4-dimethylthiophenyl carbamate (Bmpc), 2-phosphonioethyl carbamate (Peoc), 2-triphenylphosphonioisopropyl carbamate (Ppoc), 1,1-dimethyl-2-cyanoethyl carbamate, m-chloro- p-acyloxybenzyl carbamate, p-(dihydroxyboryl)benzyl carbamate, 5-benzisoxazolylmethyl carbamate, 2-(trifluoromethyl)-6-chromonylmethyl carbamate (Tcroc), m-nitrophenyl carbamate, 3,5-dimethoxybenzyl carbamate, o-nitrobenzyl carbamate, 3,4-dimethoxy-6-nitrobenzyl carbamate, phenyl(o-nitrophenyl)methyl carbamate, t-amyl carbamate, S-benzyl thiocarbamate, p-cyanobenzyl carbamate, cyclobutyl carbamate, cyclohexyl carbamate, cyclopentyl carbamate, cyclopropylmethyl carbamate, p-decyloxybenzyl carbamate, 2,2-dimethoxyacylvinyl carbamate, o-(N,N- dimethylcarboxamido)benzyl carbamate, 1,1-dimethyl-3-(N,N-dimethylcarboxamido)propyl carbamate, 1,1-dimethylpropynyl carbamate, di(2-pyridyl)methyl carbamate, 2-furanylmethyl carbamate, 2-iodoethyl carbamate, isoborynl carbamate, isobutyl carbamate, isonicotinyl carbamate, p-(p’-methoxyphenylazo)benzyl carbamate, 1-methylcyclobutyl carbamate, 1- methylcyclohexyl carbamate, 1-methyl-l-cyclopropylmethyl carbamate, 1-methyl-1(3,5- dimethoxyphenyl)ethyl carbamate, 1-methyl-1-(p-phenylazophenyl)ethyl carbamate, 1-methyl-l- phenylethyl carbamate, 1- methyl-1-(4-pyridyl)ethyl carbamate, phenyl carbamate, p- (phenylazo)benzyl carbamate, 2,4,6-tri-t-butylphenyl carbamate, 4-(trimethylammonium)benzyl carbamate, and 2,4,6-trimethylbenzyl carbamate. [082] Nitrogen protecting groups such as sulfonamide groups (e.g., -S(=O)2Raa) include, but are not limited to, p-toluenesulfonamide (Ts), benzenesulfonamide, 2,3,6,-trimethyl-4- methoxybenzenesulfonamide (Mtr), 2,4,6-trimethoxybenzenesulfonamide (Mtb), 2,6-dimethyl-4- methoxybenzenesulfonamide (Pme), 2,3,5,6-tetramethyl-4-methoxybenzenesulfonamide (Mte), 4- methoxybenzenesulfonamide (Mbs), 2,4,6- trimethylbenzenesulfonamide (Mts), 2,6-dimethoxy-4- methylbenzenesulfonamide (iMds), 2,2,5,7,8-pentamethylchroman-6-sulfonamide (Pmc),
methanesulfonamide (Ms), β-trimethylsilylethanesulfonamide (SES), 9-anthracenesulfonamide, 4- (4’,8’-dimethoxynaphthylmethyl)benzenesulfonamide (DNMBS), benzylsulfonamide, trifluoromethylsulfonamide, and phenacylsulfonamide. [083] Other nitrogen protecting groups include, but are not limited to, phenothiazinyl-(10)-acyl derivative, N’-p-toluenesulfonylaminoacyl derivative, N’ -phenylaminothioacyl derivative, N- benzoylphenylalanyl derivative, N-acetylmethionine derivative, 4,5-diphenyl-3-oxazolin-2-one, N- phthalimide, N-dithiasuccinimide (Dts), N-2,3-diphenylmaleimide, N-2,5-dimethylpyrrole, N-1,1,4,4- tetramethyldisilylazacyclopentane adduct (STABASE), 5-substituted 1,3-dimethyl-1,3,5- triazacyclohexan-2-one, 5-substituted 1,3-dibenzyl-1,3,5-triazacyclohexan-2-one, 1- substituted 3,5- dinitro-4-pyridone, N-methylamine, N-allylamine, N-[2- (trimethylsilyl)ethoxy]methylamine (SEM), N- 3-acetoxypropylamine, N-(1-isopropyl-4-nitro-2-oxo-3-pyroolin-3-yl)amine, quaternary ammonium salts, N-benzylamine, N-di(4-methoxyphenyl)methylamine, N-5-dibenzosuberylamine, N- triphenylmethylamine (Tr), N-[(4-methoxyphenyl)diphenylmethyl]amine (MMTr), N-9- phenylfluorenylamine (PhF), N-2,7 -dichloro-9-fluorenylmethyleneamine, N-ferrocenylmethylamino (Fcm), N-2- picolylamino N’-oxide, N-1,1-dimethylthiomethyleneamine, N-benzylideneamine, N-p- methoxybenzylideneamine, N-diphenylmethyleneamine, N-[(2-pyridyl)mesityl]methyleneamine, N- (N’ ,N’-dimethylaminomethylene)amine, N,N’ -isopropylidenediamine, N-p-nitrobenzylideneamine, N-salicylideneamine, N-5- chlorosalicylideneamine, N-(5-chloro-2-hydroxyphenyl) phenylmethyleneamine, N-cyclohexylideneamine, N-(5,5-dimethyl-3-oxo-l-cyclohexenyl)amine, N- borane derivative, N-diphenylborinic acid derivative, N-[phenyl(pentaacylchromium- or tungsten)acyl]amine, N-copper chelate, N-zinc chelate, N-nitroamine, N-nitrosoamine, amine N- oxide, diphenylphosphinamide (Dpp), dimethylthiophosphinamide (Mpt), diphenylthiophosphinamide (Ppt), dialkyl phosphoramidates, dibenzyl phosphoramidate, diphenyl phosphoramidate, benzenesulfenamide, o-nitrobenzenesulfenamide (Nps), 2,4- dinitrobenzenesulfenamide, pentachlorobenzenesulfenamide, 2-nitro-4- methoxybenzenesulfenamide, triphenylmethylsulfenamide, and 3-nitropyridinesulfenamide (Npys). [084] In certain embodiments, the substituent present on an oxygen atom is an oxygen protecting group (also referred to as a hydroxyl protecting group). Oxygen protecting groups are well known in the art and include those described in detail in Protecting Groups in Organic Synthesis, T. W. Greene and P. G. M. Wuts, 3rd edition, John Wiley & Sons, 1999, incorporated herein by reference. [085] Exemplary oxygen protecting groups include, but are not limited to, methyl, methoxylmethyl (MOM), methylthiomethyl (MTM), t-butylthiomethyl, (phenyldimethylsilyl)methoxymethyl (SMOM), benzyloxymethyl (BOM), p-methoxybenzyloxymethyl (PMBM), (4-methoxyphenoxy)methyl (p-AOM), guaiacolmethyl (GUM), t-butoxymethyl, 4-pentenyloxymethyl (POM), siloxymethyl, 2-
methoxyethoxymethyl (MEM), 2,2,2-trichloroethoxymethyl, bis(2-chloroethoxy)methyl, 2- (trimethylsilyl)ethoxymethyl (SEMOR), tetrahydropyranyl (THP), 3-bromotetrahydropyranyl, tetrahydrothiopyranyl, 1-methoxycyclohexyl, 4- methoxytetrahydropyranyl (MTHP), 4- methoxytetrahydrothiopyranyl, 4- methoxytetrahydrothiopyranyl S,S-dioxide, 1-[(2-chloro-4- methyl)phenyl]-4-methoxypiperidin-4-yl (CTMP), 1,4-dioxan-2-yl, tetrahydrofuranyl, tetrahydrothiofuranyl, 2,3,3a,4,5,6,7,7a-octahydro-7,8,8-trimethyl-4,7-methanobenzofuran-2-yl, 1- ethoxyethyl, 1-(2-chloroethoxy)ethyl, 1-methyl-l-methoxyethyl, 1-methyl-1-benzyloxyethyl, 1- methyl-1-benzyloxy-2-fluoroethyl, 2,2,2-trichloroethyl, 2-trimethylsilylethyl, 2- (phenylselenyl)ethyl, t-butyl, allyl, p-chlorophenyl, p-methoxyphenyl, 2,4-dinitrophenyl, benzyl (Bn), p-methoxybenzyl, 3,4-dimethoxybenzyl, o-nitrobenzyl, p-nitrobenzyl, p-halobenzyl, 2,6-dichlorobenzyl, p-cyanobenzyl, p-phenylbenzyl, 2-picolyl, 4-picolyl, 3- methyl-2-picolyl N-oxido, diphenylmethyl, p,p’- dinitrobenzhydryl, 5-dibenzosuberyl, triphenylmethyl, α-naphthyldiphenylmethyl, p- methoxyphenyldiphenylmethyl, di(p-methoxyphenyl)phenylmethyl, tri(p-methoxyphenyl)methyl, 4- (4’- bromophenacyloxyphenyl)diphenylmethyl, 4,4’,4”-tris(4,5-dichlorophthalimidophenyl)methyl, 4,4’,4”-tris(levulinoyloxyphenyl)methyl, 4,4’,4”-tris(benzoyloxyphenyl)methyl, 3-(imidazol-1- yl)bis(4’,4”-dimethoxyphenyl)methyl, 1,1-bis(4-methoxyphenyl)-1’-pyrenylmethyl, 9-anthryl, 9-(9- phenyl)xanthenyl, 9-(9-phenyl-10-oxo)anthryl, 1,3-benzodisulfuran-2-yl, benzisothiazolyl S,S-dioxido, trimethylsilyl (TMS), triethylsilyl (TES), triisopropylsilyl (TIPS), dimethylisopropylsilyl (IPDMS), diethylisopropylsilyl (DEIPS), dimethylthexylsilyl, t-butyldimethylsilyl (TBDMS), t-butyldiphenylsilyl (TBDPS), tribenzylsilyl, tri-p-xylylsilyl, triphenylsilyl, diphenylmethylsilyl (DPMS), t- butylmethoxyphenylsilyl (TBMPS), formate, benzoylformate, acetate, chloroacetate, dichloroacetate, trichloroacetate, trifluoroacetate, methoxyacetate, triphenylmethoxyacetate, phenoxyacetate, p-chlorophenoxyacetate, 3- phenylpropionate, 4-oxopentanoate (levulinate), 4,4- (ethylenedithio)pentanoate (levulinoyldithioacetal), pivaloate, adamantoate, crotonate, 4- methoxycrotonate, benzoate, p-phenylbenzoate, 2,4,6-trimethylbenzoate (mesitoate), alkyl methyl carbonate, 9- fluorenylmethyl carbonate (Fmoc), alkyl ethyl carbonate, alkyl 2,2,2-trichloroethyl carbonate (Troc), 2-(trimethylsilyl)ethyl carbonate (TMSEC), 2-(phenylsulfonyl) ethyl carbonate (Psec), 2-(triphenylphosphonio) ethyl carbonate (Peoc), alkyl isobutyl carbonate, alkyl vinyl carbonate alkyl allyl carbonate, alkyl p-nitrophenyl carbonate, alkyl benzyl carbonate, alkyl p- methoxybenzyl carbonate, alkyl 3,4-dimethoxybenzyl carbonate, alkyl o-nitrobenzyl carbonate, alkyl p-nitrobenzyl carbonate, alkyl S-benzyl thiocarbonate, 4-ethoxy-1-napththyl carbonate, methyl dithiocarbonate, 2-iodobenzoate, 4-azidobutyrate, 4-nitro-4-methylpentanoate, o- (dibromomethyl)benzoate, 2-formylbenzenesulfonate, 2-(methylthiomethoxy)ethyl, 4- (methylthiomethoxy)butyrate, 2- (methylthiomethoxymethyl)benzoate, 2,6-dichloro-4-
methylphenoxyacetate, 2,6-dichloro-4-(1,1,3,3-tetramethylbutyl)phenoxyacetate, 2,4-bis(1,1- dimethylpropyl)phenoxyacetate, chlorodiphenylacetate, isobutyrate, monosuccinoate, (E)-2-methyl- 2-butenoate, o-(methoxyacyl)benzoate, α-naphthoate, nitrate, alkyl N,N,N’,N’- tetramethylphosphorodiamidate, alkyl N-phenylcarbamate, borate, dimethylphosphinothioyl, alkyl 2,4-dinitrophenylsulfenate, sulfate, methanesulfonate (mesylate), benzylsulfonate, and tosylate (Ts). [086] In certain embodiments, the substituent present on a sulfur atom is a sulfur protecting group (also referred to as a thiol protecting group). Sulfur protecting groups are well known in the art and include those described in detail in Protecting Groups in Organic Synthesis, T. W. Greene and P. G. M. Wuts, 3rd edition, John Wiley & Sons, 1999, incorporated herein by reference. [087] Exemplary sulfur protecting groups include, but are not limited to, alkyl, benzyl, p- methoxybenzyl, 2,4,6-trimethylbenzyl, 2,4,6-trimethoxybenzyl, o-hydroxybenzyl, p-hydroxybenzyl, o- acetoxybenzyl, p-acetoxybenzyl, p-nitrobenzyl, 4-picolyl, 2-quinolinylmethyl, 2-picolyl N-oxido, 9- anthrylmethyl, 9-fluorenylmethyl, xanthenyl, ferrocenylmethyl, diphenylmethyl, bis(4- methoxyphenyl)methyl, 5-dibenzosuberyl, triphenylmethyl, diphenyl-4-pyridylmethyl, phenyl, 2,4- dinitrophenyl, t-butyl, 1-adamantyl, methoxymethyl (MOM), isobutoxymethyl, benzyloxymethyl, 2- tetrahydropyranyl, benzylthiomethyl, phenylthiomethyl, thiazolidino, acetamidomethyl, trimethylacetamidomethyl, benzamidomethyl, allyloxycarbonylaminomethyl, phenylacetamidomethyl, phthalimidomethyl, acetylmethyl, carboxymethyl, cyanomethyl, (2-nitro-1- phenyl)ethyl, 2-(2,4-dinitrophenyl)ethyl, 2-cyanoethyl, 2-(Trimethylsilyl)ethyl, 2,2- bis(carboethoxy)ethyl, (1-m-nitrophenyl-2-benzoyl)othyl, 2-phenylsulfonylethyl, 2-(4- methylphenylsulfonyl)-2-methylprop-2-yl, acetyl, benzoyl, trifluoroacetyl, N-[[(p- biphenylyl)isopropoxy]carbonyl]-N-methyl]- γ-aminothiobutyrate, 2,2,2-trichloroethoxycarbonyl, t- butoxycarbonyl, benzyloxycarbonyl, p-methoxybenzyloxycarbonyl, N-ethyl, N-methoxymethyl, sulfonate, sulfenylthiocarbonate, 3-nitro-2-pyridinesulfenyl sulfide, oxathiolone. Compounds of the Invention [088] Liposomal-based vehicles are considered as an attractive carrier for therapeutic agents and remain subject to continued development efforts. While liposomal-based vehicles that comprise certain lipid components have shown promising results with regard to encapsulation, stability and site localization, there remains a great need for improvement of liposomal-based delivery systems. For example, a significant drawback of liposomal delivery systems relates to the construction of liposomes that have sufficient cell culture or in vivo stability to reach desired target cells and/or intracellular compartments, and the ability of such liposomal delivery systems to efficiently release their encapsulated materials to such target cells.
[089] In particular, there remains a need for cationic lipids that are effective for intramuscular delivery of mRNA (e.g., for preventing or treating Flu, acne, chlamydia, or a disease or disorder induced by infection with P. gingivalis). There also remains a need for improved lipids compounds that demonstrate improved pharmacokinetic properties, and which are capable of delivering macromolecules, such as nucleic acids, to a wide variety cell types and tissues with enhanced efficiency. Importantly, there also remains a particular need for novel lipid compounds that are characterized as having improved safety profiles and are capable of efficiently delivering encapsulated nucleic acids and polynucleotides to targeted cells, tissues and organs. [090] Described herein is a novel class of cationic lipid compounds for improved in vivo delivery of therapeutic agents, such as nucleic acids (e.g., for treating or preventing Flu, acne, chlamydia, or a disease or disorder induced by infection with P. gingivalis). In particular, a cationic lipid described herein may be used, optionally with other lipids, to formulate a lipid-based nanoparticle (e.g., liposome) for encapsulating therapeutic agents, such as nucleic acids (e.g., DNA, siRNA, mRNA, microRNA) for therapeutic use, such as disease treatment and prevention (e.g for use as a vaccine e.g., for Flu, or for the prevention or treatment of acne, chlamydia, or a disease or disorder induced by infection with P. gingivalis) purposes. [091] In embodiments, compounds of the invention as described herein can provide one or more desired characteristics or properties. That is, in certain embodiments, compounds of the invention as described herein can be characterized as having one or more properties that afford such compounds advantages relative to other similarly classified lipids. For example, compounds disclosed herein can allow for the control and tailoring of the properties of liposomal compositions (e.g., lipid nanoparticles) of which they are a component. In particular, compounds disclosed herein can be characterized by enhanced transfection efficiencies and their ability to provoke specific biological outcomes. Such outcomes can include, for example enhanced cellular uptake, endosomal/lysosomal disruption capabilities and/or promoting the release of encapsulated materials (e.g., polynucleotides) intracellularly. The compounds disclosed herein can also be characterized by achieving high levels of peptide or protein expression when delivering mRNA encoding for said peptide or protein by intravenous, intrathecal, intramuscular, intranasal, sublingual, or by pulmonary delivery, optionally through nebulization. The compounds disclosed herein can also be characterized by achieving high Hemagglutination Inhibition (HAI) when delivering mRNA encoding an influenza antigen by intramuscular delivery. Additionally, the compounds disclosed herein have advantageous pharmacokinetic properties, biodistribution, and efficiency. [092] Additionally, the cationic lipids of the present invention have cleavable groups such as ester groups. These cleavable groups (e.g. esters, thioesters, disulphides, carbonates, carbamates and
thiocarbamates) are contemplated to improve biodegradability and thus contribute to the lipids’ favorable safety profiles. [093] It is contemplated that the cationic lipids of the present invention are capable of highly effective in vivo intramuscular delivery of therapeutic agents and vaccines (e.g., for treating or preventing Flu, acne, chlamydia, or a disease or disorder induced by infection with P. gingivalis). It is also contemplated that lipid nanoparticles comprising the cationic lipids of the present invention are capable of highly effective in vivo delivery while maintaining a favorable safety profile. It is also contemplated that lipid nanoparticles comprising the cationic lipids of the present invention may exhibit improved degradation in vivo. [094] Provided herein are compounds which are cationic lipids. In embodiments, the cationic lipids include compounds having a structure according to Formula (I’):
or a pharmaceutically acceptable salt thereof, wherein: a is 3 or 4; each of R1, R2, R3, and R4 is independently selected from: (i) optionally substituted C5-C30 alkyl, and optionally substituted C5-C30 alkenyl; (ii) optionally substituted C1-5 alkyl which is substituted with an optionally substituted heterocyclyl moiety;
(iii) , wherein each RA is independently selected from optionally substituted C3-C28 alkyl, optionally substituted C3-C28 alkenyl, and -W1-C(O)-O-X1, wherein each W1 is independently selected from optionally substituted C3-C5 alkylene and optionally substituted C3-C5 alkenylene, and each X1 is independently selected from optionally substituted C5-C30 alkyl and optionally substituted C5-C30 alkenyl; (iv)
, wherein each RB is independently selected from optionally substituted C5-C30 alkyl, optionally substituted C5-C30 alkenyl; and
(v) , wherein each RC independently selected from optionally substituted C5-C30 alkyl, optionally substituted C5-C30 alkenyl; each b is independently selected from 5, 6, and 7; each c is independently selected from 3, 4, and 5; d is 2, 3, or 4; and wherein R1 and R2 are not identical to one another and/or R3 and R4 are not identical to one another; and wherein at least one of R1, R2, R3, and R4, is independently selected from option (
r option
above. [095] In embodiments, the cationic lipids include compounds having a structure according to Formula (I):
or a pharmaceutically acceptable salt thereof, wherein: a is 3 or 4; each of R1, R2, R3, and R4 is independently selected from: (i) optionally substituted C5-C30 alkyl, and optionally substituted C5-C30 alkenyl; (ii) optionally substituted C1-5 alkyl which is substituted with an optionally substituted heterocyclyl moiety;
, wherein each RA is independently selected from optionally substituted C3-C28 alkyl, optionally substituted C3-C28 alkenyl, and -W1-C(O)-O-X1,
wherein each W1 is independently selected from optionally substituted C3-C5 alkylene and optionally substituted C3-C5 alkenylene, and each X1 is independently selected from optionally substituted C5-C30 alkyl and optionally substituted C5-C30 alkenyl; (iv)
, wherein each RB is independently selected from optionally substituted C5-C30 alkyl, optionally substituted C5-C30 alkenyl; and (v)
, wherein each RC independently selected from optionally substituted C5-C30 alkyl, optionally substituted C5-C30 alkenyl; each b is independently selected from 5, 6, and 7; each c is independently selected from 3, 4, and 5; and wherein R1 and R2 are not identical to one another and/or R3 and R4 are not identical to one another; and wherein at least one of R1, R2, R3, and R4, is independently selected from option
r option (
above. [096] In embodiments, if R1 and R2 are both
then the other of R3 and R4 is not selected from
,
. [097] In embodiments, if R1 and R2 are both
. [098] In embodiments, the cationic lipids do not include the following compound:
.
[099] In embodiments, the cationic lipids do not include the following compound:
. [0100] In embodiments, the cationic lipids do not include the following compound:
. [0101] In embodiments, the cationic lipids do not include the following compounds:
,
. [0102] In embodiments, a is 3. In embodiments, a is 4. [0103] In embodiments, b is 5. In embodiments, b is 6. In embodiments, b is 7. In embodiments, each b is independently selected from 5 or 6. In embodiments, each b is independently selected from 6 or 7. In one embodiment, each b is independently selected from 5 or 7. [0104] In embodiments, d is 2. In embodiments, d is 3. In embodiments, d is 4. [0105] In embodiments, one of R1, R2, R3, and R4 is selected from option
option (v)
. In embodiments, two of R1, R2, R3, and R4 are independently selected from option (iv) and/or option (v) . In embodiments, three of R1, R2, R3, and R4 are independently selected from option (iv)
r option (v) . In embodiments, all of R1, R2, R3, and
R4 are independently selected from option
r option (v)
. [0106] In embodiments, R1 and R2 are independently selected from option
and/or option (v)
, and R3 and R4 are not selected from option (iv)
. In embodiments, R3 and R4 are independently selected from option
r option (v)
[0107] In embodiments, R1 and R2 are not identical to one another and R3 and R4 are identical to one another. [0108] In embodiments, R1 and R2 are identical to one another and R3 and R4 are not identical to one another. [0109] In embodiments, R1 and R2 are not identical to one another and R3 and R4 are not identical to one another. [0110] In embodiments, none of R1, R2, R3, and R4 are identical. [0111] In embodiments, one of R1, R2, R3, and R4 is selected from optionally substituted C5-C30 alkyl. In embodiments, two of R1, R2, R3, and R4 are independently selected from optionally substituted C5- C30 alkyl. In embodiments, three of R1, R2, R3, and R4 are independently selected from optionally
substituted C5-C30 alkyl. In embodiments, R1 and R2 are independently selected from optionally substituted C5-C30 alkyl. [0112] In embodiments, one of R1, R2, R3, and R4 is selected from optionally substituted C5-C12 alkyl. In embodiments, two of R1, R2, R3, and R4 are independently selected from optionally substituted C5- C12 alkyl. In embodiments, three of R1, R2, R3, and R4 are independently selected from optionally substituted C5-C12 alkyl. In embodiments, R1 and R2 are independently selected from optionally substituted C5-C12 alkyl.
a e . [0114] In embodiments, the cationic lipids include compounds having a structure according to Formula (II’):
or a pharmaceutically acceptable salt thereof, wherein a, d, R3 and R4 are as defined herein, for example wherein a is 4. [0115] In embodiments, the cationic lipids of the present invention include compounds having a structure according to Formula (II):
(II) or a pharmaceutically acceptable salt thereof, wherein a, R3 and R4 are as defined herein, for example wherein a is 4. [0116] In embodiments, one of R1, R2, R3, and R4 is selected from optionally substituted C5-C30 alkenyl. In embodiments, two of R1, R2, R3, and R4 are independently selected from optionally substituted C5-C30 alkenyl. In embodiments, three of R1, R2, R3, and R4 are independently selected from optionally substituted C5-C30 alkenyl. [0117] In embodiments, one of R3 and R4 is selected from optionally substituted C5-C30 alkenyl. In embodiments, one of R3 and R4 is selected from optionally substituted C5-C30 alkenyl and the other of R3 and R4 is selected from option
r option (v)
. [0118] In embodiments, one of R1, R2, R3, and R4 is optionally substituted C5-C18 alkenyl. In embodiments, two of R1, R2, R3, and R4 are independently selected from optionally substituted C5-C18 alkenyl. In embodiments, three of R1, R2, R3, and R4 are independently selected from optionally substituted C5-C18 alkenyl. In embodiments, one of R3 and R4 is optionally substituted C5-C30 alkenyl. In embodiments, one of R3 and R4 is optionally substituted C5-C18 alkenyl and the other of R3 and R4 is selected from option
r option (v)
. [0119] In embodiments, one of R1, R2, R3, and R4 is
[0120] In embodiments, one of R1, R2, R3, and R4 is a
e . e o e s, ee o 1, 2, 3, and R4 are
. embodiments, one of R3
[0121] In embodiments, two of R1, R2, R3, and R4 are independently selected from optionally substituted C5-C30 alkyl and one of R1, R2, R3, and R4 is selected from optionally substituted C5-C30 alkenyl. In embodiments, R1 and R2 are independently selected from optionally substituted C5-C30 alkyl and one of R3 and R4 is selected from optionally substituted C5-C30 alkenyl. In embodiments, two of R1, R2, R3, and R4 are independently selected from optionally substituted C5-C12 alkyl and one of R1, R2, R3, and R4 is selected from optionally substituted C5-C18 alkenyl. In embodiments, R1 and R2 are independently selected from optionally substituted C5-C12 alkyl and one of R3 and R4 is selected from optionally substituted C5-C18 alkenyl. [0122] In embodiments, R1 and R2 are each
and one of R3 and R4 is selected from optionally substituted C5-C18 alkenyl. In embodiments, R1 and R2 are each
. [0123] In embodiments, one or more of R1, R2, R3, and R4 are independently selected from
, wherein each RA is independently selected from optionally substituted C3-C28 alkyl, optionally substituted C3-C28 alkenyl, and -W1-C(O)-O-X1, wherein each W1 is independently selected from optionally substituted C3-C5 alkylene and optionally substituted C3-C5 alkenylene, and each X1 is independently selected from optionally substituted C5-C30 alkyl and optionally substituted C5-C30 alkenyl. [0124] In embodiments, one of R1, R2, R3, and R4 is selected from
as defined above. In embodiments, two of R , R , R , and R are independently
1 2 3 4 selected from as defined above. In embodiments, three of R1, R2, R3, and R4 are independently selected from
as defined above. In embodiments, R
1 and R2 are independently selected from as defined above. In embodiments, R and R are independently selec
1 2 ted from as defined above and one of R3 and R4 is selected from optionally substituted C5-C30 alkenyl. In embodiments, R1 and R2
are independently selected from
as defined above and one of R3 and R4 is selected from optionally substituted C5-C18 alkenyl. In embodiments, R1 and R2 are independently selected
. n embodiments, R1 and R2 are independently selected from
as defined above and one of R3 and R4 is
. In embodiments, R3 and R4 are independently selected from
. [0125] In embodiments, one RA is selected from optionally substituted C3-C28 alkyl. In embodiments, two RA are independently selected from optionally substituted C3-C28 alkyl. In embodiments, each RA is independently selected from optionally substituted C3-C28 alkyl. In embodiments, one RA is selected from optionally substituted C3-C10 alkyl. In embodiments, two RA are independently selected from optionally substituted C3-C10 alkyl. In embodiments, each RA is independently selected from optionally substituted C3-C10 alkyl. In embodiments, one RA is
. In . In embodiments, each RA is
. [0126] In embodiments, one RA is selected from optionally substituted C3-C28 alkenyl. In embodiments, two RA are independently selected from optionally substituted C3-C28 alkenyl. In embodiments, each RA is independently selected from optionally substituted C3-C28 alkenyl. In embodiments, one RA is selected from optionally substituted C3-C16 alkenyl. In embodiments, two RA are independently selected from optionally substituted C3-C16 alkyl. In embodiments, each RA is independently selected from optionally substituted C3-C16 alkyl. In embodiments, one RA is
. embodiments, two RA are
. [0127] In embodiments, one RA is selected from -W1-C(O)-O-X1. In embodiments, two RA are independently selected from -W1-C(O)-O-X1. In embodiments, each RA is independently selected from -W1-C(O)-O-X1 . [0128] In embodiments, one W1 is selected from optionally substituted C3-C5 alkylene. In embodiments, two W1 are independently selected from optionally substituted C3-C5 alkylene. In embodiments, each W1 is independently selected from optionally substituted C3-C5 alkylene. In embodiments, one W1 is selected from optionally substituted C3-C5 alkenylene. In embodiments, two W1 are independently selected from optionally substituted C3-C5 alkenylene. In embodiments, each W1 is independently selected from optionally substituted C3-C5 alkenylene. [0129] In embodiments, one X1 is selected from optionally substituted C5-C30 alkyl. In embodiments, two X1 are independently selected from optionally substituted C5-C30 alkyl. In embodiments, each X1 is independently selected from optionally substituted C5-C30 alkyl. In embodiments, one X1 is selected from optionally substituted C6-C17 alkyl. In embodiments, two X1 are independently selected from optionally substituted C6-C17 alkyl. In embodiments, each X1 is independently selected from optionally substituted C6-C17 alkyl. In embodiments, one X1 is selected from optionally substituted branched C6-C17 alkyl. In embodiments, two X1 are independently selected from optionally substituted branched C6-C17 alkyl. In embodiments, each X1 is independently selected from optionally substituted branched C6-C17 alkyl. [0130] In embodiments, one X1 is
. In embodiments, two X1 are
, .
[0131] In embodiments, one X1 is
. In embodiments, two X1 are
. In embodiments, each
. [0132] In embodiments, one X1 is
. In embodiments, two X1 embodiments, each X1 is
.
. [0136] In embodiments, one X1 is selected from optionally substituted C5-C30 alkenyl. In embodiments, two X1 are independently selected from optionally substituted C5-C30 alkenyl. In embodiments, each X1 is independently selected from optionally substituted C5-C30 alkenyl. In embodiments, one X1 is selected from optionally substituted C5-C9 alkenyl. In embodiments, two X1 are independently selected from optionally substituted C5-C9 alkenyl. In embodiments, each X1 is independently selected from optionally substituted C5-C9 alkenyl. In embodiments, one X1 is
. , . embodiments, each
. [0137] In embodiments, one or more of R1, R2, R3, and R4 are independently selected from , wherein each RB is independently selected from optionally substituted C5-C30 alkyl and optionally substituted C5-C30 alkenyl. In embodiments, one of R1, R2, R3, and R4 is selected from as defined above. In embodiments, two of R1, R2, R3, and R4 are independently selected from
as defined above. In embodiments, three of R1, R2, R3, and R4 are independently selected from
as defined above. In embodiments, each of R1, R2, R3, and R4 are independently selected from as defined above. In embodiments, R1 and R2 are independently selected from
as defined above. In embodiments, one of R3 and R4 is selected from
as defined above. In embodiments, R3 and R4 are independently selected from as defined above. [0138] In embodiments, one RB is selected from optionally substituted C5-C30 alkyl. In embodiments, two RB are independently selected from optionally substituted C5-C30 alkyl. In embodiments, three RB are independently selected from optionally substituted C5-C30 alkyl. In embodiments, each RB is independently selected from optionally substituted C5-C30 alkyl. [0139] In embodiments, one RB is selected from optionally substituted C6-C17 alkyl. In embodiments, two RB are independently selected from optionally substituted C6-C17 alkyl. In
embodiments, three RB are independently selected from optionally substituted C6-C17 alkyl. In embodiments, each RB is independently selected from optionally substituted C6-C17 alkyl. [0140] In embodiments, one RB is selected from optionally substituted branched C6-C17 alkyl. In embodiments, two RB are independently selected from optionally substituted branched C6-C17 alkyl. In embodiments, three RB are independently selected from optionally substituted branched C6-C17 alkyl. In embodiments, each RB is independently selected from optionally substituted branched C6- C17 alkyl. [0141] In embodiments, one RB is
. In embodiments, two RB are
embodiments, each
. [0142] In embodiments, one RB is
. In embodiments, two RB are
. In embodiments, three RB are
. In embodiments, each RB is
. [0143] In embodiments, one RB is
. In embodiments, two RB
. [0144] In embodiments, the cationic lipids include compounds having a structure according to Formula (III’):
or a pharmaceutically ac ed herein.
[0145] In embodiments, the cationic lipids include compounds having a structure according to Formula (III):
or a pharmaceutically acceptable salt thereof, wherein a, R3 and R4 are as defined herein. [0146] In embodiments, one RB is selected from optionally substituted C5-C30 alkenyl. In embodiments, two RB are independently selected from optionally substituted C5-C30 alkenyl. In embodiments, three RB are independently selected from optionally substituted C5-C30 alkenyl. In embodiments, each RB is independently selected from optionally substituted C5-C30 alkenyl. [0147] In embodiments, one RB is selected from optionally substituted C5-C9 alkenyl. In embodiments, two RB are independently selected from optionally substituted C5-C9 alkenyl. In embodiments, three RB are independently selected from optionally substituted C5-C9 alkenyl. In embodiments, each RB is independently selected from optionally substituted C5-C9 alkenyl. In embodiments, one RB is
. In embodiments, two RB are
[0148] In embodiments, one or more of R1, R2, R3, and R4 are independently selected from
, wherein each RC independently selected from optionally substituted C5- C30 alkyl and optionally substituted C5-C30 alkenyl. In embodiments, one of R1, R2, R3, and R4 is selected from
, as defined above. In embodiments, two of R1, R2, R3, and R4 are independently selected from
, as defined above. In embodiments, three of R1, R2, R3, and R4 are independently selected from
, as defined
above. In embodiments, each of R1, R2, R3, and R4 are independently selected from
defined above. [0149] In embodiments, R1 and R2 are independently selected from
as defined above. [0150] In embodiments, one of R3 and R4 is selected from
as defined above. In embodiments, R3 and R4 are independently selected from
as defined above. [0151] In embodiments, one or more c is selected from 3 or 5. In embodiments, one c is selected from 3 or 5. In embodiments, two c are selected from 3 or 5. In embodiments, three c are selected from 3 or 5. In embodiments, each c is selected from 3 or 5. In embodiments, each c is 3. In embodiments, each c is 5. [0152] In embodiments, one or more c is 4. In embodiments, one c is 4. In embodiments, two c are 4. In embodiments, three c are 4. In embodiments, each c is 4. [0153] In embodiments, one RC is selected from optionally substituted C5-C30 alkyl. In embodiments, two RC are independently selected from optionally substituted C5-C30 alkyl. In embodiments, three RC are independently selected from optionally substituted C5-C30 alkyl. In embodiments, each RC is independently selected from optionally substituted C5-C30 alkyl. [0154] In embodiments, one RC is selected from optionally substituted C6-C17 alkyl. In embodiments, two RC are independently selected from optionally substituted C6-C17 alkyl. In embodiments, three RC are independently selected from optionally substituted C6-C17 alkyl. In embodiments, each RC is independently selected from optionally substituted C6-C17 alkyl. [0155] In embodiments, one RC is selected from optionally substituted branched C6-C17 alkyl. In embodiments, two RC are independently selected from optionally substituted branched C6-C17 alkyl. In embodiments, three RC are independently selected from optionally substituted branched C6-C17 alkyl. In embodiments, each RC is independently selected from optionally substituted branched C6- C17 alkyl.
[0158] In embodiments, one RC is
. In embodiments, two RC
. [0159] In embodiments, the cationic lipids include compounds having a structure according to Formula (IV’):
or a pharmaceutically acceptable salt thereof, wherein a, d, R1 and R2 are as defined herein, for example wherein a is 3. [0160] In embodiments, the cationic lipids of the present invention include compounds having a structure according to Formula (IV):
or a pharmaceutically acceptable salt thereof, wherein a, R1 and R2 are as defined herein, for example wherein a is 3. [0161] In embodiments, the cationic lipids of the present invention include compounds having a structure according to Formula (V’):
or a pharmaceutically acceptable salt thereof, wherein d, R1 and R2 are as defined herein. [0162] In embodiments, the cationic lipids of the present invention include compounds having a structure according to Formula (V):
or a pharmaceutically acceptable salt thereof, wherein R1 and R2 are as defined herein. [0163] In embodiments, one RC is selected from optionally substituted C5-C30 alkenyl. In embodiments, two RC are independently selected from optionally substituted C5-C30 alkenyl. In embodiments, three RC are independently selected from optionally substituted C5-C30 alkenyl. In embodiments, each RC is independently selected from optionally substituted C5-C30 alkenyl. [0164] In embodiments, one RC is selected from optionally substituted C5-C9 alkenyl. In embodiments, two RC are independently selected from optionally substituted C5-C9 alkenyl. In embodiments, three RC are independently selected from optionally substituted C5-C9 alkenyl. In embodiments, each RC is independently selected from optionally substituted C5 C9 alkenyl.
[0165] In embodiments, one RC is
. In embodiments, two RC are
. In embodiments, three RC are
. In embodiments, each
. [0166] In embodiments one or more of R1, R2, R3, and R4 is independently selected from optionally substituted C1-C 5 alkyl which is substituted with an optionally substituted heterocyclyl moiety. In embodiments, one of R1, R2, R3, and R4 is selected from optionally substituted C1-C5 alkyl which is substituted with an optionally substituted heterocyclyl moiety. In embodiments, two of R1, R2, R3, and R4 are independently selected from optionally substituted C1-C5 alkyl which is substituted with an optionally substituted heterocyclyl moiety. In embodiments, three of R1, R2, R3, and R4 are independently selected from optionally substituted C1-C5 alkyl which is substituted with an optionally substituted heterocyclyl moiety. In embodiments, the heterocyclyl moiety is substituted. In one embodiment, said optionally substituted C1-C5 alkyl which is substituted with an optionally substituted heterocyclyl moiety
. [0167] In embodiments, the cationic lipids include compounds having a structure according to Formula (VI’):
or a pharmaceutically acceptable salt thereof, wherein: a is 3 or 4; d is 2, 3, or 4; each c is independently selected from 3 and 4; and each RC is independently selected from
. [0168] In embodiments, the cationic lipids of the present invention include compounds having a structure according to Formula (VI):
or a pharmaceutically acceptable salt thereof, wherein: a is 3 or 4; each c is independently selected from 3 and 4; and each RC is independently selected from
. [0169] In embodiments, a is 3. In embodiments, a is 4. [0170] In embodiments, each c is 3. In embodiments, one c is 3 and one c is 4. In embodiments, each c is 4. [0171] In embodiments, each RC is
. In embodiments, each RC is
. In embodiments, one RC is
and the other RC is
. [0172] In embodiments, the substituents are not optionally substituted. In embodiments, the R1 and R2 substituents are optionally substituted and the R3 and R4 substituents are not optionally substituted. In embodiments, the R3 and R4 substituents are optionally substituted and the R1 and R2 substituents are not optionally substituted. [0173] In embodiments, the cationic lipids of the present invention have any one of the structures in Table 1 or Table 2, or a pharmaceutically acceptable salt thereof. [0174] In embodiments, provided herein is a composition comprising a cationic lipid of the present invention, and further comprising: (i) one or more non-cationic lipids, (ii) one or more cholesterol-based lipids and (iii) one or more PEG-modified lipids. [0175] In embodiments, this composition is a lipid nanoparticle, optionally a liposome. [0176] In embodiments, the one or more cationic lipid(s) constitute(s) about 30 mol %-60 mol % of the lipid nanoparticle. In embodiments, the one or more cationic lipid(s) constitute(s) about 35 mol
%-55 mol % of the lipid nanoparticle. In embodiments, the one or more cationic lipid(s) constitute(s) about 40 mol %-50 mol % of the lipid nanoparticle. [0177] In embodiments, the one or more non-cationic lipid(s) constitute(s) about 10 mol%-50 mol% of the lipid nanoparticle. In embodiments, the one or more non-cationic lipid(s) constitute(s) about 15 mol%-45 mol% of the lipid nanoparticle. In embodiments, the one or more non-cationic lipid(s) constitute(s) about 20 mol%-40 mol% of the lipid nanoparticle. In embodiments, the one or more non-cationic lipid(s) constitute(s) about 25 mol%-35 mol% of the lipid nanoparticle. [0178] In embodiments, the one or more PEG-modified lipid(s) constitute(s) about 1 mol%-10 mol% of the lipid nanoparticle. In embodiments, the one or more PEG-modified lipid(s) constitute(s) about 2 mol%-9 mol% of the lipid nanoparticle. In embodiments, the one or more PEG-modified lipid(s) constitute(s) about 3 mol%-8 mol% of the lipid nanoparticle. In embodiments, the one or more PEG- modified lipid(s) constitute(s) about 4 mol%-7 mol% of the lipid nanoparticle. In embodiments, the one or more PEG-modified lipid(s) constitute(s) about 5 mol%-6 mol% of the lipid nanoparticle. [0179] In embodiments, the cholesterol-based lipid constitutes about 10 mol%-50 mol% of the lipid nanoparticle. In embodiments, the cholesterol-based lipid constitutes about 15 mol%-45 mol% of the lipid nanoparticle. In embodiments, the cholesterol-based lipid constitutes about 20 mol%-40 mol% of the lipid nanoparticle. In embodiments, the cholesterol-based lipid constitutes about 25 mol%-35 mol% of the lipid nanoparticle. [0180] In embodiments, the lipid nanoparticle encapsulates a nucleic acid, optionally an mRNA encoding a peptide or protein. In embodiments, the lipid nanoparticle encapsulates an mRNA encoding a peptide or protein, optionally for use in a vaccine. In embodiments, the peptide encoded by the mRNA is an antigen. In embodiments, the mRNA encodes an influenza antigen. In embodiments, the mRNA encodes an antigen of influenza A virus. In embodiments, the mRNA encodes an antigen of influenza B virus. In embodiments, the mRNA encodes an antigen of Chlamydia sp. bacteria. In embodiments, the mRNA encodes an antigen of C.trachomatis. In embodiments, the mRNA encodes an antigen of P. gingivalis. In embodiments, the mRNA encodes an acne antigen. In embodiments, the mRNA encodes an antigen of C.acnes. [0181] As used herein, the phrase “encapsulation percentage” refers to the fraction of therapeutic agent (e.g. mRNA) that is effectively encapsulated within a liposomal-based vehicle (e.g. a lipid nanoparticle) relative to the initial fraction of therapeutic agent present in the lipid phase. In embodiments, the lipid nanoparticles have an encapsulation percentage for mRNA of at least 50%. In embodiments, the lipid nanoparticles have an encapsulation percentage for mRNA of at least 55%. In embodiments, the lipid nanoparticles have an encapsulation percentage for mRNA of at least 60%. In embodiments, the lipid nanoparticles have an encapsulation percentage for mRNA of
at least 65%. In embodiments, the lipid nanoparticles have an encapsulation percentage for mRNA of at least 70%. In embodiments, the lipid nanoparticles have an encapsulation percentage for mRNA of at least 75%. In embodiments, the lipid nanoparticles have an encapsulation percentage for mRNA of at least 80%. In embodiments, the lipid nanoparticles have an encapsulation percentage for mRNA of at least 85%. In embodiments, the lipid nanoparticles have an encapsulation percentage for mRNA of at least 90%. In embodiments, the lipid nanoparticles have an encapsulation percentage for mRNA of at least 95%. In embodiments, the encapsulation percentage is calculated by performing the Ribogreen assay (Invitrogen) with and without the presence of 0.1% Triton-X 100. [0182] In embodiments, the composition of the present invention is for use in therapy. [0183] In embodiments, the composition of the present invention is for use in a method of treating or preventing a disease amenable to treatment or prevention by the peptide or protein encoded by the mRNA encapsulated within said composition. [0184] In embodiments, the disease or disorder is caused by a viral infection. In embodiments, the disease or disorder is influenza. [0185] In embodiments, the disease or disorder is caused by a bacterial infection. In embodiments, the disease or disorder is chlamydia. [0186] In embodiments, the disease or disorder is induced by infection with P. gingivalis. [0187] In embodiments, the disease or disorder is acne. [0188] In embodiments, a method for treating or preventing a disease is provided, wherein said method comprises administering to a subject in need thereof a composition of the present invention and wherein the disease is amenable to treatment or prevention by the peptide or protein encoded by the mRNA. [0189] In embodiments, provided herein is the use of a composition of the present invention for the manufacture of a medicament for the treatment of a disease, wherein the disease is amenable to treatment or prevention by the peptide or protein encoded by the mRNA. [0190] In embodiments, the composition is administered intravenously, intrathecally, intramuscularly, intranasally, sublingually, or by pulmonary delivery, optionally through nebulization. In one embodiment, the composition is administered intramuscularly. Exemplary Compounds [0191] In embodiments, the cationic lipids of the present invention include compounds selected from those depicted in Table 1 or Table 2, or a pharmaceutically acceptable salt thereof.
Table 1
[0192] Any of the compounds (1-18) identified in Table 1 and Table 2 above may be provided in the form of a pharmaceutically acceptable salt and such salts are intended to be encompassed by the present invention. [0193] The compounds of the invention as described herein can be prepared according to methods known in the art, including the exemplary syntheses of the Examples provided herein. Nucleic Acids [0194] The compounds of the invention as described herein can be used to prepare compositions useful for the delivery of nucleic acids. Synthesis of Nucleic Acids [0195] Nucleic acids according to the present invention may be synthesized according to any known methods. For example, mRNAs according to the present invention may be synthesized via in vitro transcription (IVT). Briefly, IVT is typically performed with a linear or circular DNA template containing a promoter, a pool of ribonucleotide triphosphates, a buffer system that may include DTT and magnesium ions, and an appropriate RNA polymerase (e.g., T3, T7, mutated T7 or SP6 RNA polymerase), DNAse I, pyrophosphatase, and/or RNAse inhibitor. The exact conditions will vary according to the specific application. [0196] In some embodiments, for the preparation of mRNA according to the invention, a DNA template is transcribed in vitro. A suitable DNA template typically has a promoter, for example a T3, T7, mutated T7 or SP6 promoter, for in vitro transcription, followed by desired nucleotide sequence for desired mRNA and a termination signal. [0197] Desired mRNA sequence(s) according to the invention may be determined and incorporated into a DNA template using standard methods. For example, starting from a desired amino acid sequence (e.g., an enzyme sequence), a virtual reverse translation is carried out based on the degenerated genetic code. Optimization algorithms may then be used for selection of suitable codons. Typically, the G/C content can be optimized to achieve the highest possible G/C content on one hand, taking into the best possible account the frequency of the tRNAs according to codon usage on the other hand. The optimized RNA sequence can be established and displayed, for example, with the aid of an appropriate display device and compared with the original (wild-type) sequence. A secondary structure can also be analyzed to calculate stabilizing and destabilizing properties or, respectively, regions of the RNA.
Modified mRNA [0198] In some embodiments, mRNA according to the present invention may be synthesized as unmodified or modified mRNA. Modified mRNA comprises nucleotide modifications in the RNA. A modified mRNA according to the invention can thus include nucleotide modification that are, for example, backbone modifications, sugar modifications or base modifications. In some embodiments, mRNAs may be synthesized from naturally occurring nucleotides and/or nucleotide analogues (modified nucleotides) including, but not limited to, purines (adenine (A), guanine (G)) or pyrimidines (thymine (T), cytosine (C), uracil (U)), and as modified nucleotides analogues or derivatives of purines and pyrimidines, such as e.g., 1-methyl-adenine, 2-methyl-adenine, 2- methylthio-N-6-isopentenyl-adenine, N-6-methyl-adenine, N-6-isopentenyl-adenine, 2-thio-cytosine, 3-methyl-cytosine, 4-acetyl-cytosine, 5-methyl-cytosine, 2,6-diaminopurine, 1-methyl-guanine, 2- methyl-guanine, 2,2-dimethyl-guanine, 7-methyl-guanine, inosine, 1-methyl-inosine, pseudouracil (5-uracil), dihydro-uracil, 2-thio-uracil, 4-thio-uracil, 5-carboxymethylaminomethyl-2-thio-uracil, 5- (carboxyhydroxymethyl)-uracil, 5-fluoro-uracil, 5-bromo-uracil, 5-carboxymethylaminomethyl-uracil, 5-methyl-2-thio-uracil, 5-methyl-uracil, N-uracil-5-oxyacetic acid methyl ester, 5- methylaminomethyl-uracil, 5-methoxyaminomethyl-2-thio-uracil, 5'-methoxycarbonylmethyl-uracil, 5-methoxy-uracil, uracil-5-oxyacetic acid methyl ester, uracil-5-oxyacetic acid (v), 1-methyl- pseudouracil, queuosine, beta-D-mannosyl-queuosine, wybutoxosine, and phosphoramidates, phosphorothioates, peptide nucleotides, methylphosphonates, 7-deazaguanosine, 5-methylcytosine and inosine. The preparation of such analogues is known to a person skilled in the art e.g., from the U.S. Pat. No.4,373,071, U.S. Pat. No.4,401,796, U.S. Pat. No.4,415,732, U.S. Pat. No.4,458,066, U.S. Pat. No.4,500,707, U.S. Pat. No.4,668,777, U.S. Pat. No.4,973,679, U.S. Pat. No.5,047,524, U.S. Pat. No.5,132,418, U.S. Pat. No.5,153,319, U.S. Pat. Nos.5,262,530 and 5,700,642, the disclosures of which are incorporated by reference in their entirety. Pharmaceutical Formulations of Cationic Lipids and Nucleic Acids [0199] In certain embodiments, the compounds of the invention as described herein, as well as pharmaceutical and liposomal compositions comprising such lipids, can be used in formulations to facilitate the delivery of encapsulated materials (e.g., one or more polynucleotides such as mRNA) to, and subsequent transfection of one or more target cells. For example, in certain embodiments cationic lipids described herein (and compositions such as liposomal compositions comprising such lipids) are characterized as resulting in one or more of receptor-mediated endocytosis, clathrin- mediated and caveolae-mediated endocytosis, phagocytosis and macropinocytosis, fusogenicity,
endosomal or lysosomal disruption and/or releasable properties that afford such compounds advantages relative other similarly classified lipids. [0200] According to the present invention, a nucleic acid, e.g., mRNA encoding a protein (e.g., a full length, fragment or portion of a protein) as described herein may be delivered via a delivery vehicle comprising a compound of the invention as described herein. [0201] As used herein, the terms “delivery vehicle,” “transfer vehicle,” “nanoparticle,” or grammatical equivalents thereof, are used interchangeably. [0202] For example, the present invention provides a composition (e.g., a pharmaceutical composition) comprising a cationic lipids as described herein and one or more polynucleotides. A composition (e.g., a pharmaceutical composition) may further comprise (i) one or more additional cationic lipids, (ii) one or more non-cationic lipids, (iii) one or more cholesterol-based lipids and/or (iv) one or more PEG-modified lipids. [0203] In certain embodiments a composition exhibits an enhanced (e.g., increased) ability to transfect one or more target cells. Accordingly, also provided herein are methods of transfecting one or more target cells. Such methods generally comprise the step of contacting the one or more target cells with the cationic lipids and/or pharmaceutical compositions disclosed herein (e.g., a liposomal formulation comprising a compound described herein encapsulating one or more polynucleotides) such that the one or more target cells are transfected with the materials encapsulated therein (e.g., one or more polynucleotides). As used herein, the terms “transfect” or “transfection” refer to the intracellular introduction of one or more encapsulated materials (e.g., nucleic acids and/or polynucleotides) into a cell (e.g., into a target cell). The introduced polynucleotide may be stably or transiently maintained in the target cell. The term “transfection efficiency” refers to the relative amount of such encapsulated material (e.g., polynucleotides) up- taken by, introduced into, and/or expressed by the target cell which is subject to transfection. In practice, transfection efficiency may be estimated by the amount of a reporter polynucleotide product produced by the target cells following transfection. In certain embodiments, the compounds and pharmaceutical compositions described herein demonstrate high transfection efficiencies thereby improving the likelihood that appropriate dosages of the encapsulated materials (e.g., one or more polynucleotides) will be delivered to the site of pathology and subsequently expressed, while at the same time minimizing potential systemic adverse effects or toxicity associated with the compound or their encapsulated contents.
[0204] Following transfection of one or more target cells by, for example, the polynucleotides encapsulated in the one or more lipid nanoparticles comprising the pharmaceutical or liposomal compositions disclosed herein, the production of the product (e.g., a polypeptide or protein) encoded by such polynucleotide may be stimulated and the capability of such target cells to express the polynucleotide and produce, for example, a polypeptide or protein of interest is enhanced. For example, transfection of a target cell by one or more compounds or pharmaceutical compositions encapsulating mRNA will enhance (i.e., increase) the production of the protein or enzyme encoded by such mRNA. [0205] Further, delivery vehicles described herein (e.g., liposomal delivery vehicles) may be prepared to preferentially distribute to other target tissues, cells or organs, such as the heart, lungs, kidneys, spleen. In embodiments, the delivery vehicles described herein (e.g., liposomal delivery vehicles) may be prepared to preferentially distribute to the lungs. In embodiments, the lipid nanoparticles of the present invention may be prepared to achieve enhanced delivery to the target cells and tissues. For example, polynucleotides (e.g., mRNA) encapsulated in one or more of the compounds or pharmaceutical and liposomal compositions described herein can be delivered to and/or transfect targeted cells or tissues. In some embodiments, the encapsulated polynucleotides (e.g., mRNA) are capable of being expressed and functional polypeptide products produced (and in some instances excreted) by the target cell, thereby conferring a beneficial property to, for example the target cells or tissues. Such encapsulated polynucleotides (e.g., mRNA) may encode, for example, an antigen hormone, enzyme, receptor, polypeptide, peptide or other protein of interest. Liposomal Delivery Vehicles [0206] In some embodiments, a composition is a suitable delivery vehicle. In embodiments, a composition is a liposomal delivery vehicle, e.g., a lipid nanoparticle. [0207] The terms “liposomal delivery vehicle” and “liposomal composition” are used interchangeably. [0208] Enriching liposomal compositions with one or more of the cationic lipids disclosed herein may be used as a means of improving the safety profile or otherwise conferring one or more desired properties to such enriched liposomal composition (e.g., improved delivery of the encapsulated polynucleotides to one or more target cells and/or reduced in vivo toxicity of a liposomal composition). Accordingly, also contemplated are pharmaceutical compositions, and in particular liposomal compositions, that comprise one or more of the cationic lipids disclosed herein. [0209] Thus, in certain embodiments, the compounds of the invention as described herein may be used as a component of a liposomal composition to facilitate or enhance the delivery and release of
encapsulated materials (e.g., one or more therapeutic agents) to one or more target cells (e.g., by permeating or fusing with the lipid membranes of such target cells). [0210] As used herein, liposomal delivery vehicles, e.g., lipid nanoparticles, are usually characterized as microscopic vesicles having an interior aqua space sequestered from an outer medium by a membrane of one or more bilayers. Bilayer membranes of liposomes are typically formed by amphiphilic molecules, such as lipids of synthetic or natural origin that comprise spatially separated hydrophilic and hydrophobic domains (Lasic, Trends Biotechnol., 16: 307-321, 1998). Bilayer membranes of the liposomes can also be formed by amphophilic polymers and surfactants (e.g., polymerosomes, niosomes, etc.). In the context of the present invention, a liposomal delivery vehicle typically serves to transport a desired mRNA to a target cell or tissue. [0211] In certain embodiments, such compositions (e.g., liposomal compositions) are loaded with or otherwise encapsulate materials, such as for example, one or more biologically-active polynucleotides (e.g., mRNA). [0212] In embodiments, a composition (e.g., a pharmaceutical composition) comprises an mRNA encoding a peptide or protein, encapsulated within a liposome. In embodiments, a liposome comprises: (i) one or more cationic lipids, (ii) one or more non-cationic lipids, (iii) one or more cholesterol-based lipids and (iv) one or more PEG-modified lipids, wherein at least one cationic lipid is a compound of the invention as described herein. [0213] In embodiments, a composition comprises an mRNA encoding for a peptide or protein (e.g., any peptide or protein described herein). In embodiments, a composition comprises an mRNA encoding for a peptide (e.g., any peptide described herein). In embodiments, a composition comprises an mRNA encoding for a protein (e.g., any protein described herein). [0214] In embodiments, a composition (e.g., a pharmaceutical composition) comprises a nucleic acid encapsulated within a liposome, wherein the liposome comprises a compound described herein. [0215] In embodiments, a nucleic acid is an mRNA encoding a peptide or protein. In embodiments, an mRNA encodes a peptide or protein for use in the delivery to or treatment of the human body. Exemplary mRNAs are described herein. [0216] In embodiments, a liposomal delivery vehicle (e.g., a lipid nanoparticle) can have a net positive charge.
[0217] In embodiments, a liposomal delivery vehicle (e.g., a lipid nanoparticle) can have a net negative charge. [0218] In embodiments, a liposomal delivery vehicle (e.g., a lipid nanoparticle) can have a net neutral charge. [0219] In embodiments, a lipid nanoparticle that encapsulates a nucleic acid (e.g., mRNA encoding a peptide or protein) comprises one or more compounds of the invention as described herein. [0220] For example, the amount of a compound of the invention as described herein in a composition can be described as a percentage (“wt%”) of the combined dry weight of all lipids of a composition (e.g., the combined dry weight of all lipids present in a liposomal composition). [0221] In embodiments of the pharmaceutical compositions described herein, a compound of the invention as described herein is present in an amount that is about 0.5 wt% to about 30 wt% (e.g., about 0.5 wt% to about 20 wt%) of the combined dry weight of all lipids present in a composition (e.g., a liposomal composition). [0222] In embodiments, a compound of the invention as described herein is present in an amount that is about 1 wt% to about 30 wt%, about 1 wt% to about 20 wt%, about 1 wt% to about 15 wt%, about 1 wt% to about 10 wt%, or about 5 wt% to about 25 wt% of the combined dry weight of all lipids present in a composition (e.g., a liposomal composition). In embodiments, a compound of the invention as described herein is present in an amount that is about 0.5 wt% to about 5 wt%, about 1 wt% to about 10 wt%, about 5 wt% to about 20 wt%, or about 10 wt% to about 20 wt% of the combined dry weight of all lipids present in a composition such as a liposomal delivery vehicle. [0223] In embodiments, the amount of a compound of the invention as described herein is present in an amount that is at least about 5 wt%, about 10 wt%, about 15 wt%, about 20 wt%, about 25 wt%, about 30 wt%, about 35 wt%, about 40 wt%, about 45 wt%, about 50 wt%, about 55 wt%, about 60 wt%, about 65 wt%, about 70 wt%, about 75 wt%, about 80 wt%, about 85 wt%, about 90 wt%, about 95 wt%, about 96 wt%, about 97 wt%, about 98 wt%, or about 99 wt% of the combined dry weight of total lipids in a composition (e.g., a liposomal composition). [0224] In embodiments, the amount of a compound of the invention as described herein is present in an amount that is no more than about 5 wt%, about 10 wt%, about 15 wt%, about 20 wt%, about 25 wt%, about 30 wt%, about 35 wt%, about 40 wt%, about 45 wt%, about 50 wt%, about 55 wt%, about 60 wt%, about 65 wt%, about 70 wt%, about 75 wt%, about 80 wt%, about 85 wt%, about 90 wt%, about 95 wt%, about 96 wt%, about 97 wt%, about 98 wt%, or about 99 wt% of the combined dry weight of total lipids in a composition (e.g., a liposomal composition). [0225] In embodiments, a composition (e.g., a liposomal delivery vehicle such as a lipid nanoparticle) comprises about 0.1 wt% to about 20 wt% (e.g., about 0.1 wt% to about 15 wt%) of a
compound described herein. In embodiments, a delivery vehicle (e.g., a liposomal delivery vehicle such as a lipid nanoparticle) comprises about 0.5 wt%, about 1 wt%, about 3 wt%, about 5 wt%, or about 10 wt% of a compound described herein. In embodiments, a delivery vehicle (e.g., a liposomal delivery vehicle such as a lipid nanoparticle) comprises up to about 0.5 wt%, about 1 wt%, about 3 wt%, about 5 wt%, about 10 wt%, about 15 wt%, or about 20 wt% of a compound described herein. In embodiments, the percentage results in an improved beneficial effect (e.g., improved delivery to targeted tissues). [0226] The amount of a compound of the invention as described herein in a composition also can be described as a percentage (“mol%”) of the combined molar amounts of total lipids of a composition (e.g., the combined molar amounts of all lipids present in a liposomal delivery vehicle). [0227] In embodiments of pharmaceutical compositions described herein, a compound of the invention as described herein is present in an amount that is about 0.5 mol% to about 50 mol% (e.g., about 0.5 mol% to about 20 mol%) of the combined molar amounts of all lipids present in a composition such as a liposomal delivery vehicle. [0228] In embodiments, a compound of the invention as described herein is present in an amount that is about 0.5 mol% to about 5 mol%, about 1 mol% to about 10 mol%, about 5 mol% to about 20 mol%, about 10 mol% to about 20 mol%, about 15 mol% to about 30 mol%, about 20 mol% to about 35 mol%, about 25 mol% to about 40 mol%, about 30 mol% to about 45 mol%, about 35 mol% to about 50 mol%, about 40 mol% to about 55 mol %, or about 45 mol% to about 60 mol% of the combined molar amounts of all lipids present in a composition such as a liposomal delivery vehicle. In embodiments, a compound of the invention as described herein is present in an amount that is about 1 mol% to about 60 mol%, 1 mol% to about 50 mol%, 1 mol% to about 40 mol%, 1 mol% to about 30 mol%, about 1 mol% to about 20 mol%, about 1 mol% to about 15 mol%, about 1 mol% to about 10 mol%, about 5 mol% to about 55 mol%, about 5 mol% to about 45 mol%, about 5 mol% to about 35 mol% or about 5 mol% to about 25 mol% of the combined molar amounts of all lipids present in a composition such as a liposomal delivery vehicle. [0229] In certain embodiments, a compound of the invention as described herein can comprise from about 0.1 mol% to about 50 mol%, or from 0.5 mol% to about 50 mol%, or from about 1 mol% to about 50 mol%, or from about 5 mol% to about 50 mol%, or from about 10 mol% to about 50 mol%, or from about 15 mol% to about 50 mol%, or from about 20 mol% to about 50 mol%, or from about 25 mol% to about 50 mol%, or from about 30 mol% to about 50 mol%, of the total amount of lipids in a composition (e.g., a liposomal delivery vehicle). [0230] In certain embodiments, a compound of the invention as described herein can comprise greater than about 0.1 mol%, or greater than about 0.5 mol%, or greater than about 1 mol%, greater
than about 5 mol%, greater than about 10 mol%, greater than about 20 mol%, greater than about 30 mol%, or greater than about 40 mol% of the total amount of lipids in the lipid nanoparticle. [0231] In certain embodiments, a compound as described can comprise less than about 60 mol%, or less than about 55 mol%, or less than about 50 mol%, or less than about 45 mol%, or less than about 40 mol%, or less than about 35 mol %, less than about 30 mol%, or less than about 25 mol%, or less than about 10 mol%, or less than about 5 mol%, or less than about 1 mol% of the total amount of lipids in a composition (e.g., a liposomal delivery vehicle). [0232] In embodiments, the amount of a compound of the invention as described herein is present in an amount that is at least about 5 mol%, about 10 mol%, about 15 mol%, about 20 mol%, about 25 mol%, about 30 mol%, about 35 mol%, about 40 mol%, about 45 mol%, about 50 mol%, about 55 mol%, about 60 mol%, about 65 mol%, about 70 mol%, about 75 mol%, about 80 mol%, about 85 mol%, about 90 mol%, about 95 mol%, about 96 mol%, about 97 mol%, about 98 mol%, or about 99 mol% of the combined molar amounts of total lipids in a composition (e.g., a liposomal composition). [0233] In embodiments, the amount of a compound of the invention as described herein is present in an amount that is no more than about 5 mol%, about 10 mol%, about 15 mol%, about 20 mol%, about 25 mol%, about 30 mol%, about 35 mol%, about 40 mol%, about 45 mol%, about 50 mol%, about 55 mol%, about 60 mol%, about 65 mol%, about 70 mol%, about 75 mol%, about 80 mol%, about 85 mol%, about 90 mol%, about 95 mol%, about 96 mol%, about 97 mol%, about 98 mol%, or about 99 mol% of the combined molar amounts of total lipids in a composition (e.g., a liposomal composition). [0234] In embodiments, the percentage results in an improved beneficial effect (e.g., improved delivery to targeted tissues). [0235] In a typical embodiment, a composition of the invention (e.g., a liposomal composition) comprises: (i) one or more cationic lipids, (ii) one or more non-cationic lipids, (iii) one or more cholesterol-based lipids, and (iv) one or more PEG-modified lipids, wherein at least one cationic lipid is a compound of the invention as described herein. [0236] For example, a composition suitable for practicing the invention has four lipid components comprising a compound of the invention as described herein as the cationic lipid component, and further comprising: a non-cationic lipid,
(ii) a cholesterol-based lipid and (iii) a PEG-modified lipid. [0237] The non-cationic lipid may be DOPE or DEPE. The cholesterol-based lipid may be cholesterol. The PEG-modified lipid may be DMG-PEG2K. [0238] In further embodiments, pharmaceutical (e.g., liposomal) compositions comprise one or more of a PEG-modified lipid, a non-cationic lipid and a cholesterol lipid. In other embodiments, such pharmaceutical (e.g., liposomal) compositions comprise: one or more PEG-modified lipids; one or more non-cationic lipids; and one or more cholesterol lipids. In yet further embodiments, such pharmaceutical (e.g., liposomal) compositions comprise: one or more PEG-modified lipids and one or more cholesterol lipids. [0239] In embodiments, a composition (e.g., lipid nanoparticle) that encapsulates a nucleic acid (e.g., mRNA encoding a peptide or protein) comprises one or more compounds of the invention as described herein, and one or more lipids selected from the group consisting of a cationic lipid, a non- cationic lipid, and a PEGylated lipid. [0240] In embodiments, a composition (e.g., lipid nanoparticle) that encapsulates a nucleic acid (e.g., mRNA encoding a peptide or protein) comprises one or more compound of the invention as described herein; one or more lipids selected from the group consisting of a cationic lipid, a non- cationic lipid, and a PEGylated lipid; and further comprises a cholesterol-based lipid. Typically, such a composition has four lipid components comprising a compound of the invention as described herein as the cationic lipid component, and further comprising: (i) a non-cationic lipid (e.g., DOPE), (ii) a cholesterol-based lipid (e.g., cholesterol) and (iii) a PEG-modified lipid (e.g., DMG-PEG2K). [0241] In embodiments, a lipid nanoparticle that encapsulates a nucleic acid (e.g., mRNA encoding a peptide or protein) comprises one or more compounds of the invention as described herein, as well as one or more lipids selected from the group consisting of: (i) a cationic lipid, (ii) a non-cationic lipid, (iii) a PEGylated lipid, and (iv) a cholesterol-based lipid. [0242] According to various embodiments, the selection of cationic lipids, non-cationic lipids and/or PEG-modified lipids which comprise the lipid nanoparticle, as well as the relative molar ratio of such lipids to each other, is based upon the characteristics of the selected lipid(s), the nature of the intended target cells, the characteristics of the mRNA to be delivered. Additional considerations
include, for example, the saturation of the alkyl chain, as well as the size, charge, pH, pKa, fusogenicity and toxicity of the selected lipid(s). Thus, the molar ratios may be adjusted accordingly. Cationic Lipids [0243] In addition to any of the compounds of the invention as described herein, a composition may comprise one or more additional cationic lipids. [0244] In some embodiments, liposomes may comprise one or more additional cationic lipids. As used herein, the phrase “cationic lipid” refers to any of a number of lipid species that have a net positive charge at a selected pH, such as physiological pH. Several cationic lipids have been described in the literature, many of which are commercially available. [0245] Suitable additional cationic lipids for use in the compositions include the cationic lipids as described in the literature. Helper Lipids [0246] Compositions (e.g., liposomal compositions) may also comprise one or more helper lipids. Such helper lipids include non-cationic lipids. As used herein, the phrase “non-cationic lipid” refers to any neutral, zwitterionic or anionic lipid. As used herein, the phrase “anionic lipid” refers to any of a number of lipid species that carry a net negative charge at a selected pH, such as physiological pH. Non-cationic lipids include, but are not limited to, distearoylphosphatidylcholine (DSPC), dioleoylphosphatidylcholine (DOPC), dipalmitoylphosphatidylcholine (DPPC), dioleoylphosphatidylglycerol (DOPG), dipalmitoylphosphatidylglycerol (DPPG), dioleoylphosphatidylethanolamine (DOPE), 1,2-Dierucoyl-sn-glycero-3-phosphoethanolamine (DEPE), palmitoyloleoylphosphatidylcholine (POPC), palmitoyloleoyl-phosphatidylethanolamine (POPE), dioleoyl-phosphatidylethanolamine 4-(N-maleimidomethyl)-cyclohexane-1-carboxylate (DOPE-mal), dipalmitoyl phosphatidyl ethanolamine (DPPE), dimyristoylphosphoethanolamine (DMPE), distearoyl-phosphatidyl-ethanolamine (DSPE), 16-O-monomethyl PE, 16-O-dimethyl PE, 18- 1-trans PE, 1-stearoyl-2-oleoyl-phosphatidyethanolamine (SOPE), or a mixture thereof. A non- cationic or helper lipid suitable for practicing the invention is dioleoylphosphatidylethanolamine (DOPE). Alternatively, 1,2-Dierucoyl-sn-glycero-3-phosphoethanolamine (DEPE) can be used as a non-cationic or helper lipid. [0247] In some embodiments, a non-cationic lipid is a neutral lipid, i.e., a lipid that does not carry a net charge in the conditions under which the composition is formulated and/or administered. [0248] In some embodiments, a non-cationic lipid may be present in a molar ratio (mol%) of about 5% to about 90%, about 5% to about 70%, about 5% to about 50%, about 5% to about 40%, about
5% to about 30%, about 10% to about 70%, about 10% to about 50%, or about 10% to about 40% of the total lipids present in a composition. In some embodiments, total non-cationic lipids may be present in a molar ratio (mol%) of about 5% to about 90%, about 5% to about 70%, about 5% to about 50%, about 5% to about 40%, about 5% to about 30%, about 10 % to about 70%, about 10% to about 50%, or about 10% to about 40% of the total lipids present in a composition. In some embodiments, the percentage of non-cationic lipid in a liposome may be greater than about 5 mol%, greater than about 10 mol%, greater than about 20 mol%, greater than about 30 mol%, or greater than about 40 mol%. In some embodiments, the percentage total non-cationic lipids in a liposome may be greater than about 5 mol%, greater than about 10 mol%, greater than about 20 mol%, greater than about 30 mol%, or greater than about 40 mol%. In some embodiments, the percentage of non-cationic lipid in a liposome is no more than about 5 mol%, no more than about 10 mol%, no more than about 20 mol%, no more than about 30 mol%, or no more than about 40 mol%. In some embodiments, the percentage total non-cationic lipids in a liposome may be no more than about 5 mol%, no more than about 10 mol%, no more than about 20 mol%, no more than about 30 mol%, or no more than about 40 mol%. [0249] In some embodiments, a non-cationic lipid may be present in a weight ratio (wt%) of about 5% to about 90%, about 5% to about 70%, about 5% to about 50%, about 5% to about 40%, about 5% to about 30%, about 10 % to about 70%, about 10% to about 50%, or about 10% to about 40% of the total lipids present in a composition. In some embodiments, total non-cationic lipids may be present in a weight ratio (wt%) of about 5% to about 90%, about 5% to about 70%, about 5% to about 50%, about 5% to about 40%, about 5% to about 30%, about 10 % to about 70%, about 10% to about 50%, or about 10% to about 40% of the total lipids present in a composition. In some embodiments, the percentage of non-cationic lipid in a liposome may be greater than about 5 wt%, greater than about 10 wt%, greater than about 20 wt%, greater than about 30 wt%, or greater than about 40 wt%. In some embodiments, the percentage total non-cationic lipids in a liposome may be greater than about 5 wt%, greater than about 10 wt%, greater than about 20 wt%, greater than about 30 wt%, or greater than about 40 wt%. In some embodiments, the percentage of non-cationic lipid in a liposome is no more than about 5 wt%, no more than about 10 wt%, no more than about 20 wt%, no more than about 30 wt%, or no more than about 40 wt%. In some embodiments, the percentage total non-cationic lipids in a liposome may be no more than about 5 wt%, no more than about 10 wt%, no more than about 20 wt%, no more than about 30 wt%, or no more than about 40 wt%.
Cholesterol-based Lipids [0250] In some embodiments, a composition (e.g., a liposomal composition) comprising a cationic lipid of the present invention further comprises one or more cholesterol-based lipids. For example, a suitable cholesterol-based lipid for practicing the invention is cholesterol. Other suitable cholesterol- based lipids include, for example, DC-Chol (N,N-dimethyl-N-ethylcarboxamidocholesterol), 1,4-bis(3- N-oleylamino-propyl)piperazine (Gao, et al. Biochem. Biophys. Res. Comm.179, 280 (1991); Wolf et al. BioTechniques 23, 139 (1997); U.S. Pat. No.5,744,335), beta-sitosterol, or imidazole cholesterol ester (ICE), which has the following structure,
[0251] In some embodiments, a cholesterol-based lipid may be present in a molar ratio (mol%) of about 1% to about 30%, or about 5% to about 20% of the total lipids present in a liposome. In some embodiments, the percentage of cholesterol-based lipid in the lipid nanoparticle may be greater than about 5 mol%, greater than about 10 mol%, greater than about 20 mol%, greater than about 30 mol%, or greater than about 40 mol%. In some embodiments, the percentage of cholesterol-based lipid in the lipid nanoparticle may be no more than about 5 mol%, no more than about 10 mol%, no more than about 20 mol%, no more than about 30 mol%, or no more than about 40 mol%. [0252] In some embodiments, a cholesterol-based lipid may be present in a weight ratio (wt%) of about 1% to about 30%, or about 5% to about 20% of the total lipids present in a liposome. In some embodiments, the percentage of cholesterol-based lipid in the lipid nanoparticle may be greater than about 5 wt%, greater than about 10 wt%, greater than about 20 wt%, greater than about 30 wt%, or greater than about 40 wt%. In some embodiments, the percentage of cholesterol-based lipid in the lipid nanoparticle may be no more than about 5 wt%, no more than about 10 wt%, no more than about 20 wt%, no more than about 30 wt%, or no more than about 40 wt%. PEGylated Lipids [0253] In some embodiments, a composition (e.g., a liposomal composition) comprises one or more further PEGylated lipids. A suitable PEG-modified or PEGylated lipid for practicing the invention is 1,2-dimyristoyl-rac-glycero-3-methoxypolyethylene glycol-2000 (DMG-PEG2K).
[0254] For example, the use of polyethylene glycol (PEG)-modified phospholipids and derivatized lipids such as derivatized ceramides (PEG-CER), including N-octanoyl-sphingosine-1- [succinyl(methoxy polyethylene glycol)-2000] (C8 PEG-2000 ceramide) is also contemplated by the present invention in combination with one or more of compounds of the invention as described herein and, in some embodiments, other lipids together which comprise the liposome. In some embodiments, particularly useful exchangeable lipids are PEG-ceramides having shorter acyl chains (e.g., (C14) or (C18)). [0255] Contemplated further PEG-modified lipids (also referred to herein as a PEGylated lipid, which term is interchangeable with PEG-modified lipid) include, but are not limited to, a polyethylene glycol chain of up to 5 kDa in length covalently attached to a lipid with alkyl chain(s) of (C6-C20) length. In some embodiments, a PEG-modified or PEGylated lipid is PEGylated cholesterol or PEG-2K. The addition of such components may prevent complex aggregation and may also provide a means for increasing circulation lifetime and increasing the delivery of the lipid-nucleic acid composition to the target cell, (Klibanov et al. (1990) FEBS Letters, 268 (1): 235-237), or they may be selected to rapidly exchange out of the formulation in vivo (see U.S. Pat. No.5,885,613). [0256] Further PEG-modified phospholipid and derivatized lipids of the present invention may be present in a molar ratio (mol%) from about 0% to about 10%, about 0.5% to about 10%, about 1% to about 10%, about 2% to about 10%, about 3% to about 5%, about 1% to about 5%, or about 1.5% to about 3% of the total lipid present in the composition (e.g., a liposomal composition). Pharmaceutical Formulations and Therapeutic Uses [0257] Compounds of the invention as described herein may be used in the preparation of compositions (e.g., to construct liposomal compositions) that facilitate or enhance the delivery and release of encapsulated materials (e.g., one or more therapeutic polynucleotides) to one or more target cells (e.g., by permeating or fusing with the lipid membranes of such target cells). [0258] For example, when a liposomal composition (e.g., a lipid nanoparticle) comprises or is otherwise enriched with one or more of the compounds disclosed herein, the phase transition in the lipid bilayer of the one or more target cells may facilitate the delivery of the encapsulated materials (e.g., one or more therapeutic polynucleotides encapsulated in a lipid nanoparticle) into the one or more target cells. [0259] Similarly, in certain embodiments compounds of the invention as described herein may be used to prepare liposomal vehicles that are characterized by their reduced toxicity in vivo. In certain embodiments, the reduced toxicity is a function of the high transfection efficiencies associated with
the compositions disclosed herein, such that a reduced quantity of such composition may be administered to the subject to achieve a desired therapeutic response or outcome. [0260] In certain embodiments, compounds of the invention as described herein may be used to prepare liposomal vehicles that are characterized by effective intranasal delivery of mRNA. In certain embodiments, compounds of the invention as described herein may be used to prepare liposomal vehicles that are characterized by effective pulmonary delivery of mRNA. In certain embodiments, compounds of the invention as described herein may be used to prepare liposomal vehicles that are characterized by achieving high levels of peptide or protein expression when delivering mRNA encoding for said peptide or protein by intravenous, intrathecal, intramuscular, intranasal, sublingual, or by pulmonary delivery, optionally through nebulization. In certain embodiments, compounds of the invention as described herein may be used to prepare liposomal vehicles that are characterized by achieving high levels of peptide or protein expression when delivering mRNA encoding for said peptide or protein by intramuscular delivery. [0261] Thus, pharmaceutical formulations comprising a compound described and nucleic acids provided by the present invention may be used for various therapeutic disease and/or disease prevention purposes. To facilitate delivery of nucleic acids in vivo, a compound described herein and nucleic acids can be formulated in combination with one or more additional pharmaceutical carriers, targeting ligands or stabilizing reagents. In some embodiments, a compound described herein can be formulated via pre-mixed lipid solution. In other embodiments, a composition comprising a compound described herein can be formulated using post-insertion techniques into the lipid membrane of the nanoparticles. Techniques for formulation and administration of drugs may be found in “Remington’s Pharmaceutical Sciences,” Mack Publishing Co., Easton, Pa., latest edition. [0262] Suitable routes of administration include, for example, oral, rectal, vaginal, transmucosal, pulmonary including intratracheal or inhaled, or intestinal administration; parenteral delivery, including intradermal, transdermal (topical), intramuscular, subcutaneous, intramedullary injections, as well as intrathecal, direct intraventricular, intravenous, intraperitoneal, or intranasal. In embodiments, the route of administration is selected from intravenous, intrathecal, intramuscular, intranasal, sublingual, or by pulmonary delivery, optionally through nebulization. In embodiments, the route of administration can be intramuscular. In particular embodiments, the intramuscular administration is to a muscle selected from the group consisting of skeletal muscle, smooth muscle and cardiac muscle. In some embodiments the administration results in delivery of the nucleic acids to a muscle cell. In some embodiments the administration results in delivery of the nucleic acids to a hepatocyte (i.e., liver cell).
[0263] One route for administering a liposomal composition of the invention may be intravenous delivery, in particular when treating metabolic disorders, especially those affecting the liver (e.g., ornithine transcarbamylase (OTC) deficiency). Alternatively, depending on the disease or disorder to be treated, the liposomal composition may be administered via pulmonary delivery (e.g., for the treatment of cystic fibrosis). For vaccination, a liposomal composition of the invention is typically administered intramuscularly. Alternatively, a liposomal composition of the invention may be administered intranasally for vaccination. Diseases or disorders affecting the eye may be treated by administering a liposomal composition of the invention intravitreally. [0264] Alternatively or additionally, pharmaceutical formulations of the invention may be administered in a local rather than systemic manner, for example, via injection of the pharmaceutical formulation directly into a targeted tissue (e.g., in a sustained release formulation). Local delivery can be affected in various ways, depending on the tissue to be targeted. Exemplary tissues in which mRNA may be delivered and/or expressed include, but are not limited to the liver, kidney, heart, spleen, serum, brain, skeletal muscle, lymph nodes, skin, and/or cerebrospinal fluid. For example, aerosols containing compositions of the present invention can be inhaled (for nasal, tracheal, or bronchial delivery); compositions of the present invention can be injected into the site of injury, disease manifestation, or pain, for example; compositions can be provided in lozenges for oral, tracheal, or esophageal application; can be supplied in liquid, tablet or capsule form for administration to the stomach or intestines, can be supplied in suppository form for rectal or vaginal application; or can even be delivered to the eye by use of creams, drops, or even injection. [0265] Alternatively or additionally, pharmaceutical formulations of the invention may be administered intranasally. For example, the pharmaceutical formulations of the invention may be administered via nasal spray. Exemplary tissues in which mRNA may be delivered and/or expressed include, but are not limited to the lungs, heart, liver, spleen and muscle. In embodiments, the tissue to be targeted is in the lungs. In embodiments, the tissue to be targeted is in muscle. [0266] Alternatively or additionally, pharmaceutical formulations of the invention may be administered by pulmonary delivery, optionally through nebulization or dry powder inhalation. In embodiments, the pharmaceutical formulations of the invention are administered by pulmonary delivery through nebulization. In embodiments, the pharmaceutical formulations of the invention are administered by pulmonary delivery through dry powder inhalation. Exemplary tissues in which mRNA may be delivered and/or expressed include, but are not limited to the lungs, heart, liver, spleen and muscle. In embodiments, the tissue to be targeted is in the lungs. In embodiments, the tissue to be targeted is in muscle.
[0267] Compositions described herein can comprise mRNA encoding peptides including those described herein (e.g., a polypeptide such as a protein). [0268] In embodiments, a mRNA encodes a polypeptide. In embodiments, a mRNA encodes a peptide. In embodiments, the peptide is an antigen. In embodiments, a mRNA encodes a peptide for treating or preventing Flu. In embodiments, a mRNA encodes a peptide for treating or preventing influenza A virus. In embodiments, a mRNA encodes a peptide for treating or preventing influenza B virus. In embodiments, a mRNA encodes a peptide for treating or preventing chlamydia. In embodiments, a mRNA encodes a peptide for treating or preventing a disease or disorder induced by infection with P. gingivalis. In embodiments, a mRNA encodes a peptide for treating or preventing acne. [0269] In embodiments, a mRNA encodes a protein. In embodiments, a mRNA encodes a protein for treating or preventing Flu. In embodiments, a mRNA encodes a protein for treating or preventing influenza A virus. In embodiments, a mRNA encodes a protein for treating or preventing influenza B virus. In embodiments, a mRNA encodes a protein for treating or preventing chlamydia. In embodiments, a mRNA encodes a protein for treating or preventing a disease or disorder induced by infection with P. gingivalis. In embodiments, a mRNA encodes a protein for treating or preventing acne. [0270] In embodiments, the mRNA encodes an influenza antigen. In embodiments, the mRNA encodes an antigen of influenza A virus. In embodiments, the mRNA encodes an antigen of influenza B virus. In embodiments, the mRNA encodes an antigen of Chlamydia sp. bacteria. In embodiments, the mRNA encodes an antigen of C.trachomatis. In embodiments, the mRNA encodes an antigen of P. gingivalis. In embodiments, the mRNA encodes an acne antigen. In embodiments, the mRNA encodes an antigen of C.acnes. [0271] The present invention provides methods for delivering a composition having full-length mRNA molecules encoding a peptide or protein of interest for use in the treatment of a subject, e.g., a human subject or a cell of a human subject or a cell that is treated and delivered to a human subject. [0272] In embodiments, compositions comprising a cationic lipid described herein are provided for use in treating or preventing Flu in a subject (e.g. human). In embodiments, compositions comprising a cationic lipid described herein are provided for use in treating or preventing a disease or disorder caused by influenza A virus in a subject (e.g. human). In embodiments, compositions comprising a cationic lipid described herein are provided for use in treating or preventing a disease or disorder caused by influenza B virus in a subject (e.g. human). In embodiments, compositions comprising a cationic lipid described herein are provided for use in treating or preventing a chlamydia in a subject
(e.g. human). In embodiments, compositions comprising a cationic lipid described herein are provided for use in treating or preventing C.trachomatis infection in a subject (e.g. human). In embodiments, compositions comprising a cationic lipid described herein are provided for use in treating or preventing acne in a subject (e.g. human). In embodiments, compositions comprising a cationic lipid described herein are provided for use in treating or preventing C.acnes infection in a subject (e.g. human). In embodiments, compositions comprising a cationic lipid described herein are provided for use in treating or preventing a disease or disorder induced by infection with P. gingivalis. In any of these embodiments, said composition further comprises mRNA encoding a polypeptide, peptide or antigen suitable for the treatment or prevention of said disease or disorder or infection, such as those described herein. [0273] Provided herein is a method of treating or preventing Flu in a subject (e.g. human), the method comprising administering a composition of the invention comprising a cationic lipid described herein to the subject. Provided herein is a method of treating or preventing a disease or disorder caused by influenza A virus in a subject (e.g. human), the method comprising administering a composition of the invention comprising a cationic lipid described herein to the subject. Provided herein is a method of treating or preventing a disease or disorder caused by influenza B virus in a subject (e.g. human), the method comprising administering a composition of the invention comprising a cationic lipid described herein to the subject. Provided herein is a method of treating or preventing chlamydia in a subject (e.g. human), the method comprising administering a composition of the invention comprising a cationic lipid described herein to the subject. Provided herein is a method of treating or preventing C.trachomatis infection in a subject (e.g. human), the method comprising administering a composition of the invention comprising a cationic lipid described herein to the subject. Provided herein is a method of treating or preventing acne in a subject (e.g. human), the method comprising administering a composition of the invention comprising a cationic lipid described herein to the subject. Provided herein is a method of treating or preventing C.acnes infection in a subject (e.g. human), the method comprising administering a composition of the invention comprising a cationic lipid described herein to the subject. Provided herein is a method of treating or preventing a disease or disorder induced by infection with P. gingivalis in a subject (e.g. human), the method comprising administering a composition of the invention comprising a cationic lipid described herein to the subject. In any of these embodiments, said composition further comprises mRNA encoding a polypeptide, peptide or antigen suitable for the treatment or prevention of said disease or disorder or infection, such as those described herein.
Delivery Methods [0274] The route of delivery used in the methods of the invention allows for non-invasive, self- administration of the compounds of the invention. In some embodiments, the methods involve intranasal, intratracheal or pulmonary administration by aerosolization, nebulization, or instillation of a composition comprising mRNA encoding a therapeutic peptide or protein in a suitable transfection or lipid carrier vehicles as described above. In some embodiments, the methods involve intranasal, intratracheal or pulmonary administration by intravenous, intrathecal, intramuscular, intranasal, sublingual, or by pulmonary delivery, optionally through nebulization of a composition comprising mRNA encoding a therapeutic peptide or protein in a suitable transfection or lipid carrier vehicles as described above. In some embodiments, the administration is intramuscular. In some embodiments, the peptide or protein is encapsulated with a liposome. In some embodiments, the liposome comprises a lipid, which is a compound of the invention. As used herein below, administration of a compound of the invention includes administration of a composition comprising a compound of the invention. [0275] In certain embodiments compositions of the invention are provided for use in skin injection, e.g., in the epidermis, the dermis or the hypodermis of the skin. In some embodiments, the compositions are provided in a device suitable for skin injection, such as a needle (e.g., an epidermic, dermic or hypodermic needle), a needle free device, a microneedle device or a microprojection array device. Examples of microneedle or microprojection array devices suitable for the skin injection as described in US20230270842A1, US20220339416A1, US20210085598A1, US20200246450A1, US20220143376A1, US20180264244A1, US20180263641A1, US20110245776A1. [0276] Following administration of the composition to the subject, the peptide or protein product encoded by the mRNA (e.g., a functional protein or enzyme) is detectable in the peripheral target tissues for at least about one to seven days or longer following administration of the compound to the subject. The amount of peptide or protein product necessary to achieve a therapeutic effect will vary depending on the condition being treated, the peptide or protein encoded, and the condition of the patient. For example, the peptide or protein product may be detectable in the peripheral target tissues at a concentration (e.g., a therapeutic concentration) of at least 0.025-1.5 µg/ml (e.g., at least 0.050 µg/ml, at least 0.075 µg/ml, at least 0.1 µg/ml, at least 0.2 µg/ml, at least 0.3 µg/ml, at least 0.4 µg/ml, at least 0.5 µg/ml, at least 0.6 µg/ml, at least 0.7 µg/ml, at least 0.8 µg/ml, at least 0.9 µg/ml, at least 1.0 µg/ml, at least 1.1 µg/ml, at least 1.2 µg/ml, at least 1.3 µg/ml, at least 1.4 µg/ml, or at least 1.5 µg/ml), for at least about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11,12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 35, 40, 45 days or longer following administration of the compound to the subject.
[0277] It has been demonstrated that nucleic acids can be delivered to the lungs by intratracheal administration of a liquid suspension of the compound and inhalation of an aerosol mist produced by a liquid nebulizer or the use of a dry powder apparatus such as that described in U.S. patent 5,780,014, incorporated herein by reference. [0278] In certain embodiments, the compounds of the invention may be formulated such that they may be aerosolized or otherwise delivered as a particulate liquid or solid prior to or upon administration to the subject. Such compounds may be administered with the assistance of one or more suitable devices for administering such solid or liquid particulate compositions (such as, e.g., an aerosolized aqueous solution or suspension) to generate particles that are easily respirable or inhalable by the subject. In some embodiments, such devices (e.g., a metered dose inhaler, jet- nebulizer, ultrasonic nebulizer, dry-powder-inhalers, propellant-based inhaler or an insufflator) facilitate the administration of a predetermined mass, volume or dose of the compositions (e.g., about 0.5 mg/kg of mRNA per dose) to the subject. For example, in certain embodiments, the compounds of the invention are administered to a subject using a metered dose inhaler containing a suspension or solution comprising the compound and a suitable propellant. In certain embodiments, the compounds of the invention may be formulated as a particulate powder (e.g., respirable dry particles) intended for inhalation. In certain embodiments, compositions of the invention formulated as respirable particles are appropriately sized such that they may be respirable by the subject or delivered using a suitable device (e.g., a mean D50 or D90 particle size less than about 500μm, 400μm, 300μm, 250μm, 200μm, 150μm, 100μm, 75μm, 50μm, 25μm, 20μm, 15μm, 12.5μm, 10μm, 5μm, 2.5μm or smaller). In yet other embodiments, the compounds of the invention are formulated to include one or more pulmonary surfactants (e.g., lamellar bodies). In some embodiments, the compounds of the invention are administered to a subject such that a concentration of at least 0.05 mg/kg, at least 0.1 mg/kg, at least 0.5 mg/kg, at least 1.0 mg/kg, at least 2.0 mg/kg, at least 3.0 mg/kg, at least 4.0 mg/kg, at least 5.0 mg/kg, at least 6.0 mg/kg, at least 7.0 mg/kg, at least 8.0 mg/kg, at least 9.0 mg/kg, at least 10 mg/kg, at least 15 mg/kg, at least 20 mg/kg, at least 25 mg/kg, at least 30 mg/kg, at least 35 mg/kg, at least 40 mg/kg, at least 45 mg/kg, at least 50 mg/kg, at least 55 mg/kg, at least 60 mg/kg, at least 65 mg/kg, at least 70 mg/kg, at least 75 mg/kg, at least 80 mg/kg, at least 85 mg/kg, at least 90 mg/kg, at least 95 mg/kg, or at least 100 mg/kg body weight is administered in a single dose. In some embodiments, the compounds of the invention are administered to a subject such that a total amount of at least 0.1 mg, at least 0.5 mg, at least 1.0 mg, at least 2.0 mg, at least 3.0 mg, at least 4.0 mg, at least 5.0 mg, at least 6.0 mg, at least 7.0 mg, at least 8.0 mg, at least 9.0 mg, at least 10 mg, at least 15 mg, at least 20 mg, at least 25 mg, at least 30 mg, at least 35 mg, at least 40 mg, at least 45 mg, at least 50 mg, at least 55 mg, at
least 60 mg, at least 65 mg, at least 70 mg, at least 75 mg, at least 80 mg, at least 85 mg, at least 90 mg, at least 95 mg or at least 100 mg mRNA is administered in one or more doses. EXAMPLES [0279] While certain compounds, compositions and methods of the present invention have been described with specificity in accordance with certain embodiments, the following examples serve only to illustrate the compounds of the invention and are not intended to limit the same. List of abbreviations: DCM: Dichloromethane DIPEA: N,N-Diisopropylethylamine DMAP: 4-Dimethylaminopyridine EDC: 1-Ethyl-3-(3-dimethylaminopropyl)carbodiimide EtOAc: Ethyl acetate NaHCO3: Sodium hydrogencarbonate Py: Pyridine Na2SO4: Sodium Sulfate TEA: Triethylamine TFA: Trifluoroacetic Acid MS: Mass spectrometry ESI-MS: Electrospray ionization mass spectrometry TLC: Thin Layer Chromatography Example 1: Synthesis of Compounds of the Invention [0280] The cationic lipid MC3 is the currently used for in vivo delivery of e.g. siRNA (see WO2010/144740). However, the synthesis of this lipid involves a six-step process and requires handling of a Grignard reagent. In contrast, the present invention provides cationic lipids that can be prepared from readily available starting reagents, such as "Good's" buffers (see Table A below). These starting reagents can be coupled to cationic headgroups and lipid tails using coupling reactions, such as sulfonylation, acetylation and alkylation (see for example, Tables B1 and B2 below).
Table A: Examples of “Good” buffers
Table B1: Examples of lipid chains that are suitable for the present invention at positions R1 and R2
Table B2: Examples of lipid chains that are suitable for the present invention at positions R3 and R4
Compound 1
Intermediate [3]:
To a solution of triphenylmethanethiol (5.0 g, 18.08 mmol) in EtOH (40 mL) and water (40 mL) was added a solution (in 40 mL water) of NaOH (1.44 g, 36.16 mmol). The reaction mixture was stirred for 10 min and added a solution (in 40 ml EtOH) of 1,4-dibromobutane (3.65 g, 18.08 mmol) to reaction mixture. The reaction mixture was stirred for 4 hours at room temperature. The progress of reaction was monitored by TLC (5% EtOAc/hexanes). The reaction mixture was diluted with DCM and aqueous sodium bicarbonate solution, the organic layer was washed with brine. The organic layer was dried over sodium sulphate, concentrated under vacuum to give crude compound. To the crude was added MeOH (15 mL) and stirred for 15 min at 0-10 oC, the solid compound was filtered and dried under vacuum to give [3] (5.1 g, 69%) as a white solid. Results: 24 (t, 2H),
To a solution of [3] (5.0 g, 12.16 mmol) and [4] (3.16 g, 24.32 mmol) in ACN (75 mL) was added K2CO3 (6.72 g, 48.62 mmol). The reaction mixture was heated at 40 oC for 48 hours. The reaction progress was monitored by TLC (2.5% MeOH in DCM)). The reaction mixture was cooled to room temperature and filtered. The filtrate was concentrated under vacuum to give crude product. The crude was purified by flash chromatography (0 to 2.5 % MeOH in DCM) to give [5] (2.6 g, 46%) as a white solid. Results: 1H NMR (400 MHz, DMSO-d6): δ 7.41 (d, 6H), 7.28 (d, 6H), 7.20 (t, 3H), 3.59 (t, 2H), 2.73 (brs, 1H), 2.53- 2.39 (m, 10H), 2.20-2.14 (m, 4H), 1.41 (brs, 4H). LCMS: Purity 98 % ESI-MS analysis: Calculated C29H37N2OS, [M+H] = 461.26, Observed = 461.29 Intermediate [7]:
To a solution of [5] (0.5 g, 1.09 mmol) in DCM (8 mL) were added [6] (0.84 g, 1.19 mmol) in DCM (7 mL), EDC (0.31 g, 1.63 mmol), DMAP (27 mg, 0.22 mmol), DIPEA (0.38 mL, 2.17 mmol) and stirred at room temperature for 14 hours. After completion of the reaction as monitored by MS. The reaction mixture was diluted with DCM washed with NaHCO3 solution, water and brine. The organic layer was dried over anhydrous Na2SO4, concentrated, and the crude compound was purified (eluent: 20% EtOAc in hexanes) to obtain pure compound [7] as a color less oil (0.75 g, 60%). It was confirmed by MS analysis. Results: ESI-MS analysis: Calculated C69H120N3O4SSi2, [M+H] = 1142.85, Observed = 1142.8 Intermediate [8]:
To a solution of [7] (0.75 g, 0.66 mmol) in DCM (6 mL) was slowly added TFA (6 mL) at room temperature and stirred at room temperature for 0.5 hour. To that triethylsilane (0.13 mL, 0.82 mmol) was added slowly and stirred for 1 hour. After completion of the reaction as monitored by MS. The reaction mixture was concentrated to obtain crude product [8] (>0.59 g, quantitative). It was confirmed by MS analysis. Results: ESI-MS analysis: Calculated C50H106N3O4SSi2, [M+H] = 900.74, Observed = 900.7 Intermediate [10]:
To a solution of [8] (0.59 g, 0.65 mmol) in MeOH (10 mL) was added [9] (0.22 g, 0.98 mmol) at room temperature and stirred for 2 hours. After completion of the reaction as monitored by MS. The reaction mixture was concentrated, and the crude compound was purified (eluent: 8% MeOH in DCM) to obtain pure product [10] (0.45 g, 68%). It was confirmed by MS analysis. Results: ESI-MS analysis: Calculated for C55H109N4O4S2Si2, [M+H] = 1009.74; Observed = 1009.70 Intermediate [12]:
To a solution of [10] (0.500 g, 0.495 mmol) and [11] (0.583 g, 0.792 mmol) in chloroform was added triethylamine (0.35 ml, 2.47 mmol) and allowed to react at room temperature for 2.5 hours. After completion of the reaction, the reaction mixture was concentrated and purified to obtain [12] as colorless oil (0.355 g, 44% yield). ESI-MS analysis: Calculated for C97H196N4O6S2Si2, [M+H] = 1634.95; Observed = 1634.10 Compound 1 [13]:
To a 20 ml polypropylene scintillation vial was added [12] (0.355 g, 0.217 mmol, 1.0 eq) along with 4 mL of dry tetrahydrofuran. The vial was cooled to 0-5 oC and HF/pyridine (1.22 mL, 42.78 mmol) was added dropwise. After addition, the reaction vial was allowed to warm to room temperature and stirred for 18 hours. Afterwards, the reaction mixture was cooled back to 0 oC and neutralized with solid sodium bicarbonate solid, diluted with ethyl acetate, washed with NaHCO3 solution, water and brine. The organic layer was dried over anhydrous Na2SO4 and concentrated. The crude product was purified to obtain compound [13] (0.20 g, 65%). It was confirmed by 1H NMR and MS analysis. Results: NMR (400 MHz, CDCl3) 5.39 – 5.30 (m, 2H), 4.85 (quin, 1H), 4.19 (t, 2H), 3.62 (br, 2H), 3.03 – 2.18 (m, 29H), 2.07 – 1.89 (m, 4H), 1.88 – 1.54 (m, 16H), 1.53–1.40 (m, 8H), 1.39 – 1.14 (m, 90H), 0.86 (t, 15H). ESI-MS analysis: Calculated for C85H168N4O6S2, [M+H] = 1406.42; Observed = 1406.94
Synthetic Protocol for Compound 2
*Note: Intermediate 10 was synthesized using the same procedures as Compound 1.
Intermediate [12]:
To a solution of [10] (0.500 g, 0.495 mmol) and [11] (0.596 g, 0.792 mmol) in chloroform was added triethylamine (0.35 ml, 2.47 mmol) and allowed to react at room temperature for 2.5 hours. After completion of the reaction, the reaction mixture was concentrated and purified to obtain [12] as colorless oil (0.500 g, 61% yield). ESI-MS analysis: Calculated for C96H194N4O8S2Si2, [M+H] = 1652.92; Observed = 1653. Compound 2 [13]:
To a 20 ml polypropylene scintillation vial was added [12] (0.500 g, 0.302 mmol, 1.0 eq) along with 4 mL of dry tetrahydrofuran. The vial was cooled to 0-5 oC and HF/pyridine (1.55 mL, 59.59 mmol) was added dropwise. After addition, the reaction vial was allowed to warm to room temperature and stirred for 18 hours. Afterwards, the reaction mixture was cooled back to 0 oC and neutralized with solid sodium bicarbonate solid, diluted with ethyl acetate, washed with NaHCO3 solution, water and brine. The organic layer was dried over anhydrous Na2SO4 and concentrated. The crude product was purified to obtain compound [13] (0.30 g, 69%). It was confirmed by 1H NMR and MS analysis. Results:
1H NMR (400 MHz, CDCl3) 4.85 (quin, 1H), 4.20 (t, 2H), 4.04 (t, 2H), 3.63 (m, 2H), 3.04 – 2.49 (m, 20H), 2.48 – 2.18 (m, 12H), 1.95–1.55 (m, 18H), 1.54 – 1.40 (m, 8H), 1.39 – 1.16 (m, 86H), 0.86 (t, 15H). ESI-MS analysis: Calculated for C84H166N4O8S2, [M+H] = 1424.39; Observed = 1424.
Synthetic Protocol for Compound 3
*Note: Intermediate 10 was synthesized using the same procedures as Compound 1
Intermediate [12]:
To a solution of [10] (0.500 g, 0.495 mmol) and [11] (0.558 g, 0.743 mmol) in chloroform was added triethylamine (0.35 ml, 2.47 mmol) and allowed to react at room temperature for 2.5 hours. After completion of the reaction, the reaction mixture was concentrated and purified to obtain [12] as colorless oil (0.400 g, 49% yield). ESI-MS analysis: Calculated for C96H192N4O8S2Si2, [M+H] = 1650.90; Observed = 1650.91 Compound 3 [13]:
To a 20 ml polypropylene scintillation vial was added [12] (0.400 g, 0.242 mmol, 1.0 eq) along with 4 mL of dry tetrahydrofuran. The vial was cooled to 0-5 oC and HF/pyridine (1.24 mL, 47.73 mmol) was added dropwise. After addition, the reaction vial was allowed to warm to room temperature and stirred for 18 hours. Afterwards, the reaction mixture was cooled back to 0 oC and neutralized with solid sodium bicarbonate solid, diluted with ethyl acetate, washed with NaHCO3 solution, water and brine. The organic layer was dried over anhydrous Na2SO4 and concentrated. The crude product was purified to obtain compound [13] (0.220 g, 64%). It was confirmed by 1H NMR and MS analysis. Results: NMR (400 MHz, CDCl3) 5.67 – 5.46 (m, 2H), 4.84 (quin, 1H), 4.60 (d, 2H), 4.20 (t, 2H), 3.65 (br, 2H), 3.05 – 2.23 (m, 34H), 2.13 – 2.04 (m, 2H), 1.94–1.54 (m, 10H), 1.53 – 1.40 (m, 6H), 1.39 – 1.14 (m, 90H), 0.87 (t, 15H). ESI-MS analysis: Calculated for C84H166N4O8S2, [M+H] = 1422.38; Observed = 1422.42
Synthetic Protocol for Compound 4
*Note: Intermediate 10 was synthesized using the same procedures as Compound 1
Intermediate [12]:
To a solution of [10] (0.350 g, 0.346 mmol) and [11] (0.646 g, 0.420 mmol) in chloroform was added triethylamine (0.24 ml, 1.73 mmol) and allowed to react at room temperature for 2.5 hours. After completion of the reaction, the reaction mixture was concentrated and purified to obtain [12] as colorless oil (0.350 g, 62% yield). ESI-MS analysis: Calculated for C97H194N4O6S2Si2, [M+H] = 1632.93; Observed = 1632.01 Compound 4 [13]:
To a 20 ml polypropylene scintillation vial was added [12] (0.350 g, 0.214 mmol, 1.0 eq) along with 4 mL of dry tetrahydrofuran. The vial was cooled to 0-5 oC and HF/pyridine (1.1 mL, 42.22 mmol) was added dropwise. After addition, the reaction vial was allowed to warm to room temperature and stirred for 18 hours. Afterwards, the reaction mixture was cooled back to 0 oC and neutralized with solid sodium bicarbonate solid, diluted with ethyl acetate, washed with NaHCO3 solution, water and brine. The organic layer was dried over anhydrous Na2SO4 and concentrated. The crude product was purified to obtain compound [13] (0.219 g, 72%). It was confirmed by 1H NMR and MS analysis.
Results:
Following the very similar synthetic route described before, Compound 5 was obtained as a light- yellow oil (0.34 g, 30% over two steps). It was confirmed by 1H NMR and MS analysis. Results: 1H NMR (400 MHz, CDCl3) 5.67 – 5.46 (m, 2H), 4.84 (m, 1H), 4.51-4.48 (m, 2H), 4.20 (m, 2H), 3.65 (m, 4H), 2.67 – 2.23 (m, 32H), 2.13 – 1.81 (m, 2H), 1.80–1.51 (m, 14H), 1.50 – 1.13 (m, 90H), 0.87-0.75 (m, 15H). ESI-MS analysis: Calculated for C84H164N4O10S2, [M+H] = 1454.0; Observed = 1454.1
Synthetic Protocol for Compound 6
Intermediate 10 was synthesized using the same procedures as for Compound 1.
Intermediate [12]:
To a solution of [10] (0.500 g, 0.495 mmol) and [11] (0.646 g, 0.841 mmol) in chloroform was added triethylamine (0.35 ml, 2.47 mmol) and allowed to react at room temperature for 2.5 hours. After completion of the reaction, the reaction mixture was concentrated and purified to obtain [12] as colorless oil (0.370 g, 45% yield). ESI-MS analysis: Calculated for C97H196N4O8S2Si2, [M+H] = 1666.95; Observed = 1666. Compound 6:
To a 20 ml polypropylene scintillation vial was added [12] (0.370 g, 0.222 mmol, 1.0 eq) along with 4 mL of dry tetrahydrofuran. The vial was cooled to 0-5 oC and HF/pyridine (1.14 mL, 43.73 mmol) was added dropwise. After addition, the reaction vial was allowed to warm to room temperature and stirred for 18 hours. Afterwards, the reaction mixture was cooled back to 0 oC and neutralized with solid sodium bicarbonate solid, diluted with ethyl acetate, washed with NaHCO3 solution, water and brine. The organic layer was dried over anhydrous Na2SO4 and concentrated. The crude product was purified to obtain compound [13] (0.213 g, 66%). It was confirmed by 1H NMR and MS analysis. Results:
1H NMR (400 MHz, CDCl3) 5.42 – 5.29 (m, 2H), 4.84 (quin, 1H), 4.20 (t, 2H), 3.68 (br, 3H), 2,92 – 2.31 (m, 22H), 2.30 – 2.23 (t, 2H), 2.03 – 1.91 (m, 4H), 1.88–1.76 (m, 2H), 1,75 – 1.55 (m, 12H), 1.54 – 1.11 (m, 102H), 0.86 (t, 15H). ESI-MS analysis: Calculated for C85H168N4O8S2, [M+H] = 1438.42; Observed = 1438 Synthetic Protocol for Compound 7
Intermediate 10 was prepared as for Compound 1.
Intermediate [12]:
To a solution of [10] (0.390 g, 0.386 mmol) and [11] (0.518 g, 0.675 mmol) in chloroform was added triethylamine (0.322 ml, 2.32 mmol) and allowed to react at room temperature for 2.5 hours. After completion of the reaction, the reaction mixture was concentrated and purified to obtain [12] as colorless oil (0.320 g, 50% yield). ESI-MS analysis: Calculated for C97H194N4O8S2Si2, [M+H] = 1664.93; Observed = 1664. Compound 7:
To a 20 ml polypropylene scintillation vial was added [12] (0.320 g, 0.192 mmol, 1.0 eq) along with 4 mL of dry tetrahydrofuran. The vial was cooled to 0-5 oC and HF/pyridine (0.983 mL, 37.86 mmol) was added dropwise. After addition, the reaction vial was allowed to warm to room temperature and stirred for 18 hours. Afterwards, the reaction mixture was cooled back to 0 oC and neutralized with solid sodium bicarbonate solid, diluted with ethyl acetate, washed with NaHCO3 solution, water and brine. The organic layer was dried over anhydrous Na2SO4 and concentrated. The crude product was purified to obtain compound [13] (0.176 g, 64%). It was confirmed by 1H NMR and MS analysis. Results:
1H NMR (400 MHz, CDCl3) 5.44 – 5.26 (m, 4H), 4.84 (quin, 1H), 4.20 (t, 2H), 3.70 (br, 4H), 2.99 – 2.33 (m, 32H), 2.32 – 2.24 (t, 2H), 2.09–1.92 (m, 4H), 1.91 – 1.56 (m, 12H), 1.55 – 1.14 (m, 90H), 0.87 (t, 15H). ESI-MS analysis: Calculated for C85H166N4O8S2, [M+H] = 1436.40; Observed = 1436. Synthetic Protocol for Compound 8
Intermediate 10 was prepared as for Compound 1. Intermediate [12]:
To a solution of [10] (0.190 g, 0.188 mmol) and [11] (0.259 g, 0.329 mmol) in chloroform was added triethylamine (0.157 ml, 1.13 mmol) and allowed to react at room temperature for 2.5 hours. After completion of the reaction, the reaction mixture was concentrated and taken to the next step without purification (0.317 g Crude Material). ESI-MS analysis: Calculated for C96H194N4O10S2Si2, [M+H] = 1684.92; Observed = 1684. Compound 8
To a 20 ml polypropylene scintillation vial was added [12] (Crude Material, 0.317 g) along with 4 mL of dry tetrahydrofuran. The vial was cooled to 0-5 oC and HF/pyridine (1.0 mL, 37.06 mmol) was added dropwise. After addition, the reaction vial was allowed to warm to room temperature and stirred for 18 hours. Afterwards, the reaction mixture was cooled back to 0 oC and neutralized with solid sodium bicarbonate solid, diluted with ethyl acetate, washed with NaHCO3 solution, water and brine. The organic layer was dried over anhydrous Na2SO4 and concentrated. The crude product was purified to obtain compound [13] (0.090 g, 33% over two steps). It was confirmed by 1H NMR and MS analysis. Results: NMR (400 MHz, CDCl3) 4.84 (quin, 1H), 4.20 (t, 2H), 4.04 (t, 2H), 3.98 – 3.65 (br, 3H), 3.20 – 2.47 (m, 22H), 2.46 – 2.19 (m, 6H), 1.99 – 1.66 (m, 10H), 1.65–1.56 (m, 5H), 1.55 – 1.13 (m, 80H), 0.87 (t, 15H). ESI-MS analysis: Calculated for C84H N O S [M H] 145639 Ob ed = 1456.
Synthetic Protocol for Compound 9
Following the very similar synthetic route described before, Compound 9 was obtained as a dark- yellow oil 0.295 (64% yield). It was confirmed by the MS analysis. Results: ESI-MS analysis: Calculated for C85H165N4O12S2, [M+H] = 1498.19; Observed = 1498.1 Synthetic Protocol for Compound 10
Following the very similar synthetic route described before, Compound 10 was obtained as a light- brown oil (0.085 mg, 21%). It was confirmed by the MS analysis. Results: ESI-MS analysis: Calculated for C91H175N4O11S2, [M+H] = 1565.27; Observed = 1565.1 Synthetic Protocol for Compound 11
Following the very similar synthetic route described before, Compound 11 was obtained as a yellow oil (0.050 g, 13%). It was confirmed by the MS analysis. Results:
ESI-MS analysis: Calculated for C74H141N4O13S2, [M+H] = 1358.07; Observed = 1358.1 Synthetic Protocol for Compound 12
Following the very similar synthetic route described before, Asymmetric Good lipid (GL-Asymm-032) was obtained as a light-yellow oil (0.060 g, 15%). It was confirmed by the MS analysis. Results: ESI-MS analysis: Calculated for C85H165N4O14S2, [M+H] = 1530.18; Observed = 1530.0 Synthetic Protocol for Compound 13
Following the very similar synthetic route described before, Asymmetric Good lipid (GL-Asymm-031) was obtained as a light-yellow oil (0.197 g, 50%). It was confirmed by the MS analysis. Results: ESI-MS analysis: Calculated for C102H199N4O12S2, [M+H] = 1737.46; Observed = 1736.84 Synthetic Protocol for Compound 14
Following the very similar synthetic route described before, Compound 14 was obtained as a pale- yellow oil (0.15 g, 15% over two steps). It was confirmed by 1H NMR and MS analysis.
Results: 1H NMR (400 MHz, CDCl3) 4.80-4.77 (m, 2H), 4.10-3.96 (m, 6H), 3.65 – 3.98 (m, 4H), 3.30 – 2.47 (m, 24H), 2.23 – 2.19 (m, 12H), 1.99 – 1.66 (m, 12H), 1.65–1.56 (m, 30H), 1.22 – 1.18 (m, 90H), 0.82-0.79 (m, 18H). ESI-MS analysis: Calculated for C102H198N4O14S2, [M+H] = 1768.84; Observed = 1768.74 Synthetic Protocol for Compound 15
Following the very similar synthetic route described before, Compound 15 was obtained as a light- yellow oil (0.32 g, 53%). It was confirmed by the MS analysis. Results: ESI-MS analysis: Calculated for C68H135N4O10S2, [M+H] = 1231.96; Observed = 1231.10 Synthetic Protocol for Compound 16
Following the very similar synthetic route described before, Compound 16 was obtained as a pale- yellow oil (0.275 g, 35%). It was confirmed by the MS analysis. Results: ESI-MS analysis: Calculated for C67H133N4O10S2, [M+H] = 1217.95; Observed = 1217.10
Synthetic Protocol for Compound 17
Following the very similar synthetic route described before, Compound 17 was obtained as a light- yellow oil 0.150 g (38%). It was confirmed by the MS analysis. Results: ESI-MS analysis: Calculated for C64H127N4O10S2, [M+H] = 1175.90; Observed = 1175.10
Following the very similar synthetic route described before, Asymmetric Good lipid (GL-Asymm-029) was obtained as a light-yellow oil (0.208 g, 42%). It was confirmed by the MS analysis. Results: ESI-MS analysis: Calculated for C63H125N4O10S2, [M+H] = 1161.88; Observed = 1161.1
Example 2: Lipid Nanoparticle Formulation [0281] Cationic lipids described herein can be used in the preparation of lipid nanoparticles according to methods known in the art. For example, suitable methods include methods described in International Publication No. WO 2018/089801, which is hereby incorporated by reference in its entirety. [0282] The lipid nanoparticles in the examples of the present invention were formulated using Process A of WO 2018/089801 (see, e.g., Example 1 and Figure 1 of WO 2018/089801). Process A (“A”) relates to a conventional method of encapsulating mRNA by mixing mRNA with a mixture of lipids, without first pre-forming the lipids into lipid nanoparticles. In an exemplary process, an ethanolic solution of a mixture of lipids (cationic lipid, phosphatidylethanolamine, cholesterol, and polyethylene glycol-lipid) at a fixed lipid to mRNA ratio were combined with an aqueous buffered solution of target mRNA at an acidic pH under controlled conditions to yield a suspension of uniform LNPs. After ultrafiltration and diafiltration into a suitable diluent system, the resulting nanoparticle suspensions were diluted to final concentration, filtered, and stored frozen at −80°C until use. [0283] Lipid nanoparticle formulations of Table C were prepared by Process A. All of the lipid nanoparticle formulations comprised hEPO mRNA and the different lipids (Cationic Lipid: DMG- PEG2000: Cholesterol: DOPE/DSPC) in the mol % ratios specified in Table C. Table C: Exemplary lipid nanoparticle characterizations
* The N/P ratio is defined as the ratio of the number of nitrogen in cationic lipid to the number of phosphate in nucleic acid. [0284] The cationic lipids of the present invention and lipid OF-02 were evaluated with lipid nanoparticle formulation 1. MC3 and SM-102 were evaluated with lipid nanoparticle formulation 2, which is a typical formulation for those lipids
Example 3: Delivery of hEPO mRNA by intramuscular administration Mouse Studies [0285] In summary, lipid screening studies were conducted with female BALB/cJ mice 6-8 weeks of age (Jax #000651). Mice were dosed with 0.1 µg in 30 µL of LNPs by a single intramuscular (IM) injection into the gastrocnemius leg muscle. Blood samples were taken 6 and 24 hours post injection and hEPO levels were measured in the blood serum of the mice using an ELISA assay according to the manufacture’s protocol (R&D systems, Cat# DEP-00). WO2022/099003 A1 also describes an in vivo assay for intramuscular administration (e.g. on page 46, paragraph [00206]). Measured serum hEPO expression data are presented in Table D below. This data demonstrates that the disclosed lipids can induce expression of delivered mRNAs. Table D: Results of hEPO mRNA delivery studies - intramuscular administration of hEPO mRNA lipid formulations comprising the disclosed cationic lipids
Example 4: Influenza titer [0286] Groups of Balb/c mice (Mus musculus) as per the treatment group were immunized under isoflurane anesthesia with a dose of 0.4 µg per mouse in 0.05 mL of Modified Tasmania H3 mRNA-lipid nanoparticles via the IM route in the quadriceps, on day 0 in one hind leg and day 21 in the contralateral leg. Mice were evaluated for a minimum of 3 days post-administration and any animal that displayed severe clinical signs after the veterinarian’s assessment was euthanized by administration of 5 mg/kg of meloxicam by subcutaneous injection. [0287] Blood was collected via submandibular or orbital sinus bleeds (in-life bleeds were performed on day -1 and on day 20) and cardiac puncture (terminal bleed, day 35) from all animals under sedation. Mice were bled on pre-study to obtain a base-line pre-immune serum sample and for pre- screening purposes. [0288] HAI assays were performed using the A/Tasmania/503/2020 (H3N2) virus stocks (BIOQUAL, Inc.). Sera were treated with receptor-destroying enzyme (RDE) by diluting one part serum with three parts enzyme and incubated overnight in a 37°C water bath. Enzyme was inactivated by a 30-minute incubation period at 56°C followed by addition of six parts PBS for a final dilution of 1/10. HAI assays were performed in V-bottom 96-well plates using four hemagglutinating units (HAU) of virus and 0.5% turkey RBC. The reference serum for each strain was included as a positive control on every assay plate. Each plate also included a back-titration to confirm the antigen dose (4 HAU/25 µl) as well as a negative control sample (PBS or naive control serum). The HAI titer was determined as the highest dilution of serum resulting in complete inhibition of hemagglutination. Results were only considered valid for plates with the appropriate back-titration result (verifying 4 HAU/25 µl added) and a reference serum titer within 2-fold of the expected titer. The majority of tested lipids induced HAI
titers not significantly different from that of clinical lipid OF-02 (see Fig.1). This example demonstrates that the disclosed lipids can deliver antigenic sequences that induce significant immune responses.
NUMBERED EMBODIMENTS 1. A compound having a structure according to Formula (I’), optionally Formula (I):
or a pharmaceutically acceptable salt thereof, wherein: a is 3 or 4; each of R1, R2, R3, and R4 is independently selected from: (i) optionally substituted C5-C30 alkyl, and optionally substituted C5-C30 alkenyl; (ii) optionally substituted C1-5 alkyl which is substituted with a heterocycle moiety; (iii)
, wherein each RA is independently selected from optionally substituted C3-C28 alkyl, optionally substituted C3-C28 alkenyl, and -W1-C(O)-O-X1, wherein each W1 is independently selected from optionally substituted C3-C5 alkylene and optionally substituted C3-C5 alkenylene, and each X1 is independently selected from optionally substituted C5-C30 alkyl and optionally substituted C5-C30 alkenyl; (iv)
, wherein each RB is independently selected from optionally substituted C5-C30 alkyl, optionally substituted C5-C30 alkenyl; and
(v)
wherein each RC independently selected from optionally substituted C5-C30 alkyl, optionally substituted C5-C30 alkenyl; each b is independently selected from 5, 6, and 7; each c is independently selected from 3, 4, and 5; d is 2, 3, or 4; and wherein R1 and R2 are not identical to one another and/or R3 and R4 are not identical to one another; and wherein at least one of R1, R2, R3, and R4, is independently selected from option
option (v) above.
2. The compound of embodiment 1, or a pharmaceutically acceptable salt thereof, wherein
. 3. The compound of embodiments 1 or 2, or a pharmaceutically acceptable salt thereof, wherein the compound is not
. 4. The compound of any one of embodiments 1-3, or a pharmaceutically acceptable salt thereof, wherein the compound is not
.
5. The compound of any one of embodiments 1-4, or a pharmaceutically acceptable salt thereof, wherein the compound is not
. 6. The compound of any one of embodiments 1-5, or a pharmaceutically acceptable salt thereof, wherein: (i) a is 3; or (ii) a is 4. 7. The compound of any one of embodiments 1-6, or a pharmaceutically acceptable salt thereof, wherein: (i) b is 5; or (ii) b is 6; or (iii) b is 7; or (iv) each b is independently selected from 5 or 7. 8. The compound of any one of embodiments 1-7, or a pharmaceutically acceptable salt thereof, wherein R1 and R2 are not identical to one another and R3 and R4 are identical to one another. 9. The compound of any one of embodiments 1-7, or a pharmaceutically acceptable salt thereof, wherein R1 and R2 are identical to one another and R3 and R4 are not identical to one another. 10. The compound of any one of embodiments 1-7, or a pharmaceutically acceptable salt thereof, wherein: (i) R1 and R2 are not identical to one another and R3 and R4 are not identical to one another; or
(ii) none of R1, R2, R3, and R4 are identical. 11. The compound of any one of embodiments 1-10, or a pharmaceutically acceptable salt thereof, wherein one or more of R1, R2, R3, and R4 are independently selected from optionally substituted C5-C30 alkyl or optionally substituted C5-C30 alkenyl. 12. The compound of embodiment 11, or a pharmaceutically acceptable salt thereof, wherein one or more of R1, R2, R3, and R4 are independently selected from optionally substituted C5-C30 alkyl. 13. The compound of embodiment 12, or a pharmaceutically acceptable salt thereof, wherein one or more of R1, R2, R3, and R4 are independently selected from optionally substituted C5-C12 alkyl, for example
. 14. The compound of any one of embodiments 1-7, 9, or 11-13, wherein the compound has a structure according to Formula (II’), optionally Formula (II):
or a pharmaceutically acceptable salt thereof, optionally wherein a is 4. 15. The compound of any one of embodiments 11-14, or a pharmaceutically acceptable salt thereof, wherein one or more of R1, R2, R3, and R4 are independently selected from optionally substituted C5-C30 alkenyl.
16. The compound of embodiment 15, or a pharmaceutically acceptable salt thereof, wherein one or more of R1, R2, R3, and R4 are independently selected from optionally substituted C5-C18
. 17. The compound of any one of embodiments 11-16, or a pharmaceutically acceptable salt thereof, wherein two of R1, R2, R3, and R4 are independently selected from optionally substituted C5- C30 alkyl and one of R1, R2, R3, and R4 is selected from optionally substituted C5-C30 alkenyl. 18. The compound of any one of embodiments 1-17, or a pharmaceutically acceptable salt thereof, wherein one or more of R1, R2, R3, and R4 is independently selected from
, wherein each RA is independently selected from optionally substituted C3-C28 alkyl, optionally substituted C3-C28 alkenyl, and -W1-C(O)-O-X1, wherein each W1 is independently selected from optionally substituted C3-C5 alkylene and optionally substituted C3-C5 alkenylene, and each X1 is independently selected from optionally substituted C5-C30 alkyl and optionally substituted C5-C30 alkenyl. 19. The compound of embodiment 18, or a pharmaceutically acceptable salt thereof, wherein one or more RA is independently selected from optionally substituted C3-C28 alkyl, optionally wherein each RA is independently selected from optionally substituted C3-C28 alkyl. 20. The compound of embodiment 19, or a pharmaceutically acceptable salt thereof, wherein one or more RA is independently selected from optionally substituted C3-C10 alkyl, for example
, optionally wherein each RA is independently selected from optionally substituted C3-C10 alkyl, for example
. 21. The compound of any one of embodiments 18-20, or a pharmaceutically acceptable salt thereof, wherein one or more RA is i d d tl l t d f ti lly substituted C3-C28
alkenyl, for example optionally substituted C3-C16 alkenyl, optionally wherein each RA is independently selected from optionally substituted C3-C28 alkenyl, for example optionally substituted C3-C16 alkenyl. 22. The compound of any one of embodiments 18-21, or a pharmaceutically acceptable salt thereof, wherein one or more RA is independently selected from -W1-C(O)-O-X1, optionally wherein each RA is independently selected from -W1-C(O)-O-X1. 23. The compound of embodiment 22, or a pharmaceutically acceptable salt thereof, wherein one or more W1 is independently selected from optionally substituted C3-C5 alkylene, optionally wherein each W1 is independently selected from optionally substituted C3-C5 alkylene. 24. The compound of embodiment 22, or a pharmaceutically acceptable salt thereof, wherein one or more W1 is independently selected from optionally substituted C3-C5 alkenylene, optionally wherein each W1 is independently selected from optionally substituted C3-C5 alkenylene. 25. The compound of any one of embodiments 22-24, or a pharmaceutically acceptable salt thereof, wherein one or more X1 is independently selected from optionally substituted C5-C30 alkyl, optionally wherein each X1 is independently selected from optionally substituted C5-C30 alkyl. 26. The compound of embodiment 25, or a pharmaceutically acceptable salt thereof, wherein one or more X1 is independently selected from optionally substituted C6-C17 alkyl, for example optionally substituted branched C6-C17 alkyl, optionally wherein each X1 is independently selected from optionally substituted C6-C17 alkyl, for example optionally substituted branched C6-C17 alkyl. 27. The compound of embodiment 26, or a pharmaceutically acceptable salt thereof, wherein one or more X1 is independently selected from
(iii)
, optionally wherein each X1 is independently selected from options (i), (ii), and (iii) above. 28. The compound of any one of embodiments 22-27, or a pharmaceutically acceptable salt thereof, wherein one or more X1 is independently selected from optionally substituted C5-C30 alkenyl, optionally wherein each X1 is independently selected from optionally substituted C5-C30 alkenyl. 29. The compound of embodiment 28, or a pharmaceutically acceptable salt thereof, wherein one or more X1 is independently selected from optionally substituted C5-C9 alkenyl, for example
, optionally wherein each X1 is independently selected from optionally substituted C5-C9 alkenyl, for example
. 30. The compound of any one of embodiments 1-29, or a pharmaceutically acceptable salt thereof, wherein one or more of R1, R2, R3, and R4 is independently selected from
, wherein each RB is independently selected from optionally substituted C5-C30 alkyl and optionally substituted C5-C30 alkenyl. 31. The compound of embodiment 30, or a pharmaceutically acceptable salt thereof, wherein one or more RB is independently selected from optionally substituted C5-C30 alkyl, optionally wherein each RB is independently selected from optionally substituted C5-C30 alkyl. 32. The compound of embodiment 31, or a pharmaceutically acceptable salt thereof, wherein one or more RB is independently selected from optionally substituted C6-C17 alkyl, for example optionally substituted branched C6-C17 alkyl, optionally wherein each RB is independently selected from optionally substituted C6-C17 alkyl, for example optionally substituted branched C6-C17 alkyl. 33. The compound of embodiment 32, or a pharmaceutically acceptable salt thereof, wherein one or more RB is independently selected from
, optionally wherein each RB is independently selected from options (i), (ii), and (iii) above. 34. The compound of any one of embodiments 1-8, 10-13, or 15-33 wherein the compound has a structure according to Formula (III’), optionally Formula (III):
35. The compound of any one of embodiments 30-34, or a pharmaceutically acceptable salt thereof, wherein one or more RB is independently selected from optionally substituted C5-C30 alkenyl, optionally wherein each RB is independently selected from optionally substituted C5-C30 alkenyl. 36. The compound of embodiment 35, or a pharmaceutically acceptable salt thereof, wherein one or more RB is independently selected from optionally substituted C5-C9 alkenyl, for example
, optionally wherein each RB is independently selected from optionally substituted C5-C9 alkenyl, for example
.
37. The compound of any one of embodiments 1-36, or a pharmaceutically acceptable salt thereof, wherein one or more of R1, R2, R3, and R4 are independently selected from
, wherein each RC independently selected from optionally substituted C5- C30 alkyl and optionally substituted C5-C30 alkenyl. 38. The compound of embodiment 37, or a pharmaceutically acceptable salt thereof, wherein one or more c is selected from 3 or 5, optionally wherein each c is selected from 3 or 5. 39. The compound of embodiment 37, or a pharmaceutically acceptable salt thereof, wherein one or more c is 4, optionally wherein each c is 4. 40. The compound of any one of embodiments 37 to 39, or a pharmaceutically acceptable salt thereof, wherein one or more RC is independently selected from optionally substituted C5-C30 alkyl, optionally wherein each RC is independently selected from optionally substituted C5-C30 alkyl. 41. The compound of embodiment 40, or a pharmaceutically acceptable salt thereof, wherein one or more RC is independently selected from optionally substituted C6-C17 alkyl, for example optionally substituted branched C6-C17 alkyl, optionally wherein each RC is independently selected from optionally substituted C6-C17 alkyl, for example optionally substituted branched C6-C17 alkyl. 42. The compound of embodiment 41, or a pharmaceutically acceptable salt thereof, wherein one or more RC is independently selected from
, optionally wherein each RC is independently selected from options (i), (ii) and (iii) above.
43. The compound of any one of embodiments 1-8, 11-13, 15-33, or 35-42 wherein the compound has a structure according to Formula (IV’), optionally Formula (IV); or Formula (V’), optionally Formula (V):
or a pharmaceutically acceptable salt thereof, optionally wherein a is 3.
44. The compound of any one of embodiments 37-43, or a pharmaceutically acceptable salt thereof, wherein one or more RC is independently selected from optionally substituted C5-C30 alkenyl, optionally wherein each RC is independently selected from optionally substituted C5-C30 alkenyl. 45. The compound of embodiment 44, or a pharmaceutically acceptable salt thereof, wherein one or more RC are independently selected from optionally substituted C5-C9 alkenyl, for example
, optionally wherein each RC are independently selected from optionally substituted C5-C9 alkenyl, for example
. 46. The compound of any one of embodiments 1-45, or a pharmaceutically acceptable salt thereof, wherein one or more of R1, R2, R3, and R4 is independently selected from optionally substituted C1-5 alkyl which is substituted with an optionally substituted heterocyclyl moiety. 47. The compound of embodiment 46, or a pharmaceutically acceptable salt thereof, wherein said optionally substituted C1-5 alkyl which is substituted with an optionally substituted heterocyclyl
48. A compound having a structure according to Formula (VI’), optionally Formula (VI):
(VI) or a pharmaceutically acceptable salt thereof, wherein: a is 3 or 4; d is 2, 3, or 4; each c is independently selected from 3 and 4; and each RC is independently selected from
. 49. The compound of embodiment 48, or a pharmaceutically acceptable salt thereof, wherein a is 3. 50. The compound of embodiment 48, or a pharmaceutically acceptable salt thereof, wherein a is 4. 51. The compound of any one of embodiments 48-50, or a pharmaceutically acceptable salt thereof, wherein each c is 3. 52. The compound of any one of embodiments 48-50, or a pharmaceutically acceptable salt thereof, wherein each c is 4. 53. The compound of any one of embodiments 48-52, or a pharmaceutically acceptable salt thereof, wherein: (i) each
(ii) each
54. The compound of any one of embodiments 1-53, or a pharmaceutically acceptable salt thereof, wherein, in the compound of Formula (I’), (II’), (III'), (IV'), (V’), or (VI’): (i) d is 2; (ii) d is 3; or (iii) d is 4.
55. A compound selected from those listed in Tables 1 or 2, or a pharmaceutically acceptable salt thereof. 56. A compound of embodiment 1, wherein the compound is selected from:
, or a pharmaceutically acceptable salt thereof. 57. A compound of any one of embodiments 1-33, 35-42, 44-47, or 55-56, wherein the compound is selected from:
or a pharmaceutically acceptable salt thereof. 58. A compound of embodiment 1, wherein the compound is
, or a pharmaceutically acceptable salt thereof.
59. A compound of any one of embodiments 48-55, wherein the compound is
, or a pharmaceutically acceptable salt thereof. 60. A composition comprising the compound of any one of embodiments 1-59 or a pharmaceutically acceptable salt thereof, one or more non-cationic lipids, one or more cholesterol- based lipids and one or more PEG-modified lipids. 61. The composition of embodiment 60, wherein the composition is a lipid nanoparticle, optionally a liposome. 62. The composition of embodiment 61, wherein the one or more cationic lipid(s) constitute(s) about 30 mol %-60 mol % of the lipid nanoparticle. 63. The composition of any one of embodiments 61-62, wherein the one or more non-cationic lipid(s) constitute(s) 10 mol %-50 mol % of the lipid nanoparticle. 64. The composition of any one of embodiments 61-63, wherein the one or more PEG-modified lipid(s) constitute(s) 1 mol %-10 mol % of the lipid nanoparticle. 65. The composition of any one of embodiments 61-64, wherein the cholesterol-based lipid constitutes 10 mol %-50 mol% of the lipid nanoparticle. 66. The composition of any one of embodiments 61-65, wherein the lipid nanoparticle encapsulates a nucleic acid. 67. The composition of any one of embodiments 61-65, wherein the lipid nanoparticle encapsulates a mRNA encoding a peptide or protein. 68. The composition of embodiment 67, wherein the mRNA encodes an antigen.
69. The composition of embodiment 68, wherein the mRNA encodes an influenza antigen, for example an antigen of influenza A virus or an antigen of influenza B virus. 70. The composition of embodiment 68, wherein the mRNA encodes an antigen of Chlamydia sp. bacteria, for example an antigen of C.trachomatis. 71. The composition of embodiment 68, wherein the mRNA encodes an antigen of P. gingivalis. 72. The composition of embodiment 68, wherein the mRNA encodes an acne antigen, for example an antigen of C.acnes. 73. The composition of any one of embodiments 67-72, wherein the lipid nanoparticles have an encapsulation percentage for mRNA of (i) at least 60% (ii) at least 65% (iii) at least 70%; (iv) at least 75%; (v) at least 80%; (vi) at least 85%; (vii) at least 90%; or (viii) at least 95%. 74. The composition of any one of embodiments 66-73 for use in therapy. 75. The composition of any one of embodiments 67-73 for use in a method of treating or preventing a disease or disorder amenable to treatment or prevention by the peptide or protein encoded by the mRNA. 76. The composition for use according to embodiment 75, wherein the disease or disorder is caused by a viral infection. 77. The composition for use according to embodiment 76, wherein the disease or disorder is influenza.
78. The composition for use according to embodiment 75, wherein the disease or disorder is caused by a bacterial infection. 79. The composition for use according to embodiment 78, wherein the disease or disorder is chlamydia. 80. The composition for use according to embodiment 78, wherein the disease or disorder is induced by infection with P. gingivalis. 81. The composition for use according to embodiment 78 wherein the disease or disorder is acne. 82. A method of treating or preventing a disease or disorder wherein said method comprises administering to a subject in need thereof the composition of any one of embodiments 67-73 and wherein the disease is amenable to treatment or prevention by the peptide or protein encoded by the mRNA. 83. The method of embodiment 82 wherein the disease or disorder is caused by a viral infection. 84. The method of embodiment 83, wherein the disease or disorder is influenza. 85. The method of embodiment 82, wherein the disease or disorder is caused by a bacterial infection. 86. The method of embodiment 85, wherein the disease or disorder is chlamydia. 87. The method of embodiment 85, wherein the disease or disorder is a disease induced by infection with P. gingivalis. 88. The method of embodiment 85, wherein the disease or disorder is acne. 89. The composition for use according to any of embodiments 74-81, or the method of any one of embodiments 82-88, wherein the composition is administered intravenously, intrathecally or intramuscularly, or by pulmonary delivery, optionally through nebulization.
90. The composition for use according to embodiment 89, or the method of embodiment 89, wherein the composition is administered intramuscularly. 91. The composition of embodiment 69 for use in a method of treating or preventing influenza, wherein the composition is administered intramuscularly. 92. The composition of embodiment 70 for use in a method of treating or preventing chlamydia, wherein the composition is administered intramuscularly. 93. The composition of embodiment 71 for use in a method of treating or preventing a disease induced by infection with P. gingivalis, wherein the composition is administered intramuscularly. 94. The composition of embodiment 72 for use in a method of treating or preventing acne, wherein the composition is administered intramuscularly. 95. A method for treating or preventing influenza, wherein said method comprises administering to a subject in need thereof the composition of embodiment 69, wherein the composition is administered intramuscularly. 96. A method for treating or preventing chlamydia, wherein said method comprises administering to a subject in need thereof the composition of embodiment 70, wherein the composition is administered intramuscularly. 97. A method for treating or preventing a disease induced by infection with P. gingivalis, wherein said method comprises administering to a subject in need thereof the composition of embodiment 71, wherein the composition is administered intramuscularly. 98. A method for treating or preventing acne, wherein said method comprises administering to a subject in need thereof the composition of embodiment 72, wherein the composition is administered intramuscularly. 99. A compound having a structure according to Formula (I’), optionally Formula (I):
or a pharmaceutically acceptable salt thereof, wherein: a is 3 or 4; each of R1, R2, R3, and R4 is independently selected from: (i) optionally substituted C5-C30 alkyl, and optionally substituted C5-C30 alkenyl; (ii) optionally substituted C1-5 alkyl which is substituted with an optionally substituted heterocyclyl moiety; (iii)
, wherein each RA is independently selected from optionally substituted C3-C28 alkyl, optionally substituted C3-C28 alkenyl, and -W1-C(O)-O-X1, wherein each W1 is independently selected from optionally substituted C3-C5 alkylene and optionally substituted C3-C5 alkenylene, and each X1 is independently selected from optionally substituted C5-C30 alkyl and optionally substituted C5-C30 alkenyl; (iv)
, wherein each RB is independently selected from optionally substituted C5-C30 alkyl, optionally substituted C5-C30 alkenyl; and (v)
, wherein each RC independently selected from optionally substituted C5-C30 alkyl, optionally substituted C5-C30 alkenyl;
each b is independently selected from 5, 6, and 7; each c is independently selected from 3, 4, and 5; d is 2, 3, or 4; and wherein R1 and R2 are not identical to one another and/or R3 and R4 are not identical to one another; and wherein at least one of R1, R2, R3, and R4, is independently selected from option
option (
above. 100. The compound of embodiment 99, or a pharmaceutically acceptable salt thereof, wherein:
(c) the compound is not one of the following structures: , ,
.
101. The compound of any one of embodiments 99-100, or a pharmaceutically acceptable salt thereof, wherein: (a) R1 and R2 are not identical to one another and R3 and R4 are identical to one another; (b) R1 and R2 are identical to one another and R3 and R4 are not identical to one another; (c) R1 and R2 are not identical to one another and R3 and R4 are not identical to one another; or (d) none of R1, R2, R3, and R4 are identical. 102. The compound of any one of embodiments 99-101, or a pharmaceutically acceptable salt thereof, wherein: (a) one or more of R1, R2, R3, and R4 are independently selected from optionally substituted C5-C30 alkyl or optionally substituted C5-C30 alkenyl; (b) one or more of R1, R2, R3, and R4 are independently selected from optionally substituted C5-C30 alkyl; (c) one or more of R1, R2, R3, and R4 are independently selected from optionally substituted C5-C12 alkyl, for example
(d) one or more of R1, R2, R3, and R4 are independently selected from optionally substituted C5-C30 alkenyl; (e) one or more of R1, R2, R3, and R4 are independently selected from optionally substituted C5-C18
(f) two of R1, R2, R3, and R4 are independently selected from optionally substituted C5-C30 alkyl and one of R1, R2, R3, and R4 is selected from optionally substituted C5-C30 alkenyl; and/or (g) one or more of R1, R2, R3, and R4, or one of R1, R2, R3, and R4 is independently selected from optionally substituted C1-5 alkyl which is substituted with an optionally substituted heterocyclyl moiety, optionally wherein said optionally substituted C1-5 alkyl which is substituted with an optionally substituted heterocyclyl moiety
.
103. The compound of any one of embodiments 99-102, or a pharmaceutically acceptable salt thereof, wherein one or more of R1, R2, R3, and R4 is independently selected from
, wherein each RA is independently selected from optionally substituted C3-C28 alkyl, optionally substituted C3-C28 alkenyl, and -W1-C(O)-O-X1, wherein each W1 is independently selected from optionally substituted C3-C5 alkylene and optionally substituted C3-C5 alkenylene, and each X1 is independently selected from optionally substituted C5-C30 alkyl and optionally substituted C5-C30 alkenyl, optionally wherein: (a) one or more RA is independently selected from optionally substituted C3-C28 alkyl, optionally wherein each RA is independently selected from optionally substituted C3-C28 alkyl; (b) one or more RA is independently selected from optionally substituted C3-C10 alkyl, for example
, optionally wherein each RA is independently selected from optionally substituted C3-C10 alkyl, for example
; (c) one or more RA is independently selected from optionally substituted C3-C28 alkenyl, for example optionally substituted C3-C16 alkenyl, optionally wherein each RA is independently selected from optionally substituted C3-C28 alkenyl, for example optionally substituted C3-C16 alkenyl; and/or (d) one or more RA is independently selected from -W1-C(O)-O-X1, optionally wherein each RA is independently selected from -W1-C(O)-O-X1. 104. The compound of any one of embodiments 99-103, or a pharmaceutically acceptable salt thereof, wherein one or more of R1, R2, R3, and R4 is independently selected from
, wherein each RB is independently selected from optionally substituted C5-C30 alkyl and optionally substituted C5-C30 alkenyl, optionally wherein: (a) one or more RB is independently selected from optionally substituted C5-C30 alkyl, optionally wherein each RB is independently selected from optionally substituted C5-C30 alkyl; (b) one or more RB is independently selected from optionally substituted C6-C17 alkyl, for example optionally substituted branched C6-C17 alkyl, optionally wherein each RB is independently selected from optionally substituted C6-C17 alkyl, for example optionally substituted branched C6-C17 alkyl; (c) one or more RB is independently selected from
( ) , optionally wherein each RB is independently selected from options (i), (ii), and (iii) above; (d) one or more RB is independently selected from optionally substituted C5-C30 alkenyl, optionally wherein each RB is independently selected from optionally substituted C5-C30 alkenyl; and/or (e) one or more RB is independently selected from optionally substituted C5-C9 alkenyl, for example
, optionally wherein each RB is independently selected from optionally substituted C5-C9 alkenyl, for example
. 105. The compound of any one of embodiments 99-104, or a pharmaceutically acceptable salt thereof, wherein one or more of R1, R2, R3, and R4 are independently selected from
, wherein each RC independently selected from optionally substituted C5- C30 alkyl and optionally substituted C5-C30 alkenyl, optionally wherein: (a) one or more c is selected from 3 or 5, optionally wherein each c is selected from 3 or 5; (b) one or more c is 4, optionally wherein each c is 4; (c) one or more RC is independently selected from optionally substituted C5-C30 alkyl, optionally wherein each RC is independently selected from optionally substituted C5-C30 alkyl; (d) one or more RC is independently selected from optionally substituted C6-C17 alkyl, for example optionally substituted branched C6-C17 alkyl, optionally wherein each RC is independently selected from optionally substituted C6-C17 alkyl, for example optionally substituted branched C6-C17 alkyl; (e) one or more RC is independently selected from
, optionally wherein each RC is independently selected from options (i), (ii) and (iii) above; (f) one or more RC is independently selected from optionally substituted C5-C30 alkenyl, optionally wherein each RC is independently selected from optionally substituted C5-C30 alkenyl; and/or (g) one or more RC are independently selected from optionally substituted C5-C9 alkenyl, for example
, optionally wherein each RC are independently selected from optionally substituted C5-C9 alkenyl, for example
. 106. The compound of any one of embodiments 99-105, or a pharmaceutically acceptable salt thereof, wherein the compound has a structure according to Formula (II’), optionally Formula (II), Formula (III’), optionally Formula (III), Formula (IV’), optionally Formula (IV); or Formula (V’), optionally Formula (V):
or a pharmaceutically acceptable salt thereof. 107. A compound having a structure according to Formula (VI’), optionally Formula (VI):
or a pharmaceutically acceptable salt thereof, wherein: a is 3 or 4; d is 2, 3, or 4; each c is independently selected from 3 and 4; and each RC is independently selected from
. 108. The compound of embodiment 107, or a pharmaceutically acceptable salt thereof, wherein: (a) a is 3; or (b) a is 4; and/or (c) each c is 3; or (d) each c is 4; and/or
(e) each
(f) each
109. The compound of any one of embodiments 99-108, or a pharmaceutically acceptable salt thereof, wherein: (a) in the compound of Formula (I’), (II’), (III'), (IV'), (V’), or (VI’): (i) d is 2; (ii) d is 3; or (iii) d is 4; or (b) the compound is selected from those listed in Tables 1 or 2, or a pharmaceutically acceptable salt thereof. 110. A composition comprising the compound of any one of embodiments 99-109 or a pharmaceutically acceptable salt thereof, one or more non-cationic lipids, one or more cholesterol- based lipids and one or more PEG-modified lipids, optionally wherein: (a) the composition is a lipid nanoparticle, optionally a liposome; (b) the composition is a lipid nanoparticle, optionally a liposome and the one or more cationic lipid(s) constitute(s) about 30 mol %-60 mol % of the lipid nanoparticle; (c) the composition is a lipid nanoparticle, optionally a liposome and the one or more non-cationic lipid(s) constitute(s) 10 mol %-50 mol % of the lipid nanoparticle; (d) the composition is a lipid nanoparticle, optionally a liposome and the one or more PEG-modified lipid(s) constitute(s) 1 mol %-10 mol % of the lipid nanoparticle; and/or (e) the composition is a lipid nanoparticle, optionally a liposome and the cholesterol-based lipid constitutes 10 mol %-50 mol% of the lipid nanoparticle. 111. The composition of embodiment 110, wherein the composition is a lipid nanoparticle, optionally a liposome, and wherein said lipid nanoparticle encapsulates: (a) a nucleic acid; (b) a mRNA encoding a peptide or protein; (c) a mRNA encoding an antigen;
(d) a mRNA encoding an influenza antigen, for example an antigen of influenza A virus or an antigen of influenza B virus; (e) a mRNA encoding an antigen of Chlamydia sp. bacteria, for example an antigen of C.trachomatis; (f) a mRNA encoding an antigen of P. gingivalis; or (g) a mRNA encoding an acne antigen, for example an antigen of C.acnes. 112. The composition of embodiment 111 encapsulating a mRNA encoding a peptide, protein or antigen for use in therapy optionally wherein said composition is for use in: (a) a method of treating or preventing a disease or disorder amenable to treatment or prevention by the peptide, protein or antigen encoded by the mRNA; (b) a method of treating or preventing a disease or disorder amenable to treatment or prevention by the peptide, protein or antigen encoded by the mRNA, wherein the disease or disorder is caused by a viral infection; (c) a method of treating or preventing a disease or disorder amenable to treatment or prevention by the peptide, protein or antigen encoded by the mRNA, wherein the disease or disorder is influenza; (d) a method of treating or preventing a disease or disorder amenable to treatment or prevention by the peptide, protein or antigen encoded by the mRNA, wherein the disease or disorder is caused by a bacterial infection; (e) a method of treating or preventing a disease or disorder amenable to treatment or prevention by the peptide, protein or antigen encoded by the mRNA, wherein the disease or disorder is chlamydia; (f) a method of treating or preventing a disease or disorder amenable to treatment or prevention by the peptide, protein or antigen encoded by the mRNA, wherein the disease or disorder is induced by infection with P. gingivalis; or (g) a method of treating or preventing a disease or disorder amenable to treatment or prevention by the peptide, protein or antigen encoded by the mRNA, wherein the disease or disorder is acne. 113. The composition for use according to embodiment 112, wherein the composition is administered intravenously, intrathecally or intramuscularly, or by pulmonary delivery, optionally through nebulization, optionally wherein the composition is administered intramuscularly.
Claims
CLAIMS 1. A compound having a structure according to Formula (I):
or a pharmaceutically acceptable salt thereof, wherein: a is 3 or 4; each of R1, R2, R3, and R4 is independently selected from: (i) optionally substituted C5-C30 alkyl, and optionally substituted C5-C30 alkenyl; (ii) optionally substituted C1-5 alkyl which is substituted with an optionally substituted heterocyclyl moiety; (iii)
, wherein each RA is independently selected from optionally substituted C3-C28 alkyl, optionally substituted C3-C28 alkenyl, and -W1-C(O)-O-X1, wherein each W1 is independently selected from optionally substituted C3-C5 alkylene and optionally substituted C3-C5 alkenylene, and each X1 is independently selected from optionally substituted C5-C30 alkyl and optionally substituted C5-C30 alkenyl; (iv)
, wherein each RB is independently selected from optionally substituted C5-C30 alkyl, optionally substituted C5-C30 alkenyl; and (v)
, wherein each RC independently selected from optionally substituted C5-C30 alkyl, optionally substituted C5-C30 alkenyl; each b is independently selected from 5, 6, and 7; each c is independently selected from 3, 4, and 5;
d is 2, 3, or 4; and wherein R1 and R2 are not identical to one another and/or R3 and R4 are not identical to one another; and wherein at least one of R1, R2, R3, and R4, is independently selected from option
option (
above.
3. The compound of any one of claims 1-2, or a pharmaceutically acceptable salt thereof, wherein: (a) R1 and R2 are not identical to one another and R3 and R4 are identical to one another;
(b) R1 and R2 are identical to one another and R3 and R4 are not identical to one another; (c) R1 and R2 are not identical to one another and R3 and R4 are not identical to one another; or (d) none of R1, R2, R3, and R4 are identical.
4. The compound of any one of claims 1-3, or a pharmaceutically acceptable salt thereof, wherein: (a) one or more of R1, R2, R3, and R4 are independently selected from optionally substituted C5-C30 alkyl or optionally substituted C5-C30 alkenyl; (b) one or more of R1, R2, R3, and R4 are independently selected from optionally substituted C5-C30 alkyl; (c) one or more of R1, R2, R3, and R4 are independently selected from optionally substituted C5-C12 alkyl, for example
(d) one or more of R1, R2, R3, and R4 are independently selected from optionally substituted C5-C30 alkenyl; (e) one or more of R1, R2, R3, and R4 are independently selected from optionally substituted C5-C18
(f) two of R1, R2, R3, and R4 are independently selected from optionally substituted C5-C30 alkyl and one of R1, R2, R3, and R4 is selected from optionally substituted C5-C30 alkenyl; and/or (g) one or more of R1, R2, R3, and R4, or one of R1, R2, R3, and R4 is independently selected from optionally substituted C1-5 alkyl which is substituted with an optionally substituted heterocyclyl moiety, optionally wherein said optionally substituted C1-5 alkyl which is substituted with an optionally substituted heterocyclyl moiety
.
5. The compound of any one of claims 1-4, or a pharmaceutically acceptable salt thereof, wherein one or more of R1, R2, R3, and R4 is independently selected from
, wherein each RA is independently selected from optionally substituted C3-C28 alkyl, optionally substituted C3- C28 alkenyl, and -W1-C(O)-O-X1, wherein each W1 is independently selected from optionally substituted C3-C5 alkylene and optionally substituted C3-C5 alkenylene, and each X1 is independently selected from optionally substituted C5-C30 alkyl and optionally substituted C5-C30 alkenyl, optionally wherein: (a) one or more RA is independently selected from optionally substituted C3-C28 alkyl, optionally wherein each RA is independently selected from optionally substituted C3-C28 alkyl; (b) one or more RA is independently selected from optionally substituted C3-C10 alkyl, for example
, optionally wherein each RA is independently selected from optionally substituted C3-C10 alkyl, for example
; (c) one or more RA is independently selected from optionally substituted C3-C28 alkenyl, for example optionally substituted C3-C16 alkenyl, optionally wherein each RA is independently selected from optionally substituted C3-C28 alkenyl, for example optionally substituted C3-C16 alkenyl; and/or (d) one or more RA is independently selected from -W1-C(O)-O-X1, optionally wherein each RA is independently selected from -W1-C(O)-O-X1.
6. The compound of any one of claims 1-5, or a pharmaceutically acceptable salt thereof, wherein one or more of R1, R2, R3, and R4 is independently selected from , wherein each RB is independently selected from optionally substituted C5-C30 alkyl and optionally substituted C5-C30 alkenyl, optionally wherein: (a) one or more RB is independently selected from optionally substituted C5-C30 alkyl, optionally wherein each RB is independently selected from optionally substituted C5-C30 alkyl; (b) one or more RB is independently selected from optionally substituted C6-C17 alkyl, for example optionally substituted branched C6-C17 alkyl, optionally wherein each RB is independently selected from optionally substituted C6-C17 alkyl, for example optionally substituted branched C6-C17 alkyl; (c) one or more RB is independently selected from
( ) , optionally wherein each RB is independently selected from options (i), (ii), and (iii) above; (d) one or more RB is independently selected from optionally substituted C5-C30 alkenyl, optionally wherein each RB is independently selected from optionally substituted C5-C30 alkenyl; and/or (e) one or more RB is independently selected from optionally substituted C5-C9 alkenyl, for example
, optionally wherein each RB is independently selected from optionally substituted C5-C9 alkenyl, for example
.
7. The compound of any one of claims 1-6, or a pharmaceutically acceptable salt thereof, wherein one or more of R1, R2, R3, and R4 are independently selected from
, wherein each RC independently selected from optionally substituted C5-C30 alkyl and optionally substituted C5-C30 alkenyl, optionally wherein: (a) one or more c is selected from 3 or 5, optionally wherein each c is selected from 3 or 5; (b) one or more c is 4, optionally wherein each c is 4; (c) one or more RC is independently selected from optionally substituted C5-C30 alkyl, optionally wherein each RC is independently selected from optionally substituted C5-C30 alkyl; (d) one or more RC is independently selected from optionally substituted C6-C17 alkyl, for example optionally substituted branched C6-C17 alkyl, optionally wherein each RC is independently selected from optionally substituted C6-C17 alkyl, for example optionally substituted branched C6-C17 alkyl; (e) one or more RC is independently selected from
, optionally wherein each RC is independently selected from options (i), (ii) and (iii) above; (f) one or more RC is independently selected from optionally substituted C5-C30 alkenyl, optionally wherein each RC is independently selected from optionally substituted C5-C30 alkenyl; and/or (g) one or more RC are independently selected from optionally substituted C5-C9 alkenyl, for example
, optionally wherein each RC are independently selected from optionally substituted C5-C9 alkenyl, for example
.
8. The compound of any one of claims 1-7, or a pharmaceutically acceptable salt thereof, wherein the compound has a structure according to Formula (II), Formula (III), Formula (IV), or Formula (V):
11. The compound of any one of claims 1-10, or a pharmaceutically acceptable salt thereof, wherein the compound is selected from those listed in Tables 1 or 2, or a pharmaceutically acceptable salt thereof.
12. A composition comprising the compound of any one of claims 1-11 or a pharmaceutically acceptable salt thereof, one or more non-cationic lipids, one or more cholesterol-based lipids and one or more PEG-modified lipids, optionally wherein: (a) the composition is a lipid nanoparticle, optionally a liposome; (b) the composition is a lipid nanoparticle, optionally a liposome and the one or more cationic lipid(s) constitute(s) about 30 mol %-60 mol % of the lipid nanoparticle; (c) the composition is a lipid nanoparticle, optionally a liposome and the one or more non-cationic lipid(s) constitute(s) 10 mol %-50 mol % of the lipid nanoparticle; (d) the composition is a lipid nanoparticle, optionally a liposome and the one or more PEG-modified lipid(s) constitute(s) 1 mol %-10 mol % of the lipid nanoparticle; and/or (e) the composition is a lipid nanoparticle, optionally a liposome and the cholesterol-based lipid constitutes 10 mol %-50 mol% of the lipid nanoparticle.
13. The composition of claim 12, wherein the composition is a lipid nanoparticle, optionally a liposome, and wherein said lipid nanoparticle encapsulates: (a) a nucleic acid; (b) a mRNA encoding a peptide or protein; (c) a mRNA encoding an antigen; (d) a mRNA encoding an influenza antigen, for example an antigen of influenza A virus or an antigen of influenza B virus; (e) a mRNA encoding an antigen of Chlamydia sp. bacteria, for example an antigen of C.trachomatis; (f) a mRNA encoding an antigen of P. gingivalis; or (g) a mRNA encoding an acne antigen, for example an antigen of C.acnes.
14. The composition of claim 13 encapsulating a mRNA encoding a peptide, protein or antigen for use in therapy optionally wherein said composition is for use in: (a) a method of treating or preventing a disease or disorder amenable to treatment or prevention by the peptide, protein or antigen encoded by the mRNA; (b) a method of treating or preventing a disease or disorder amenable to treatment or prevention by the peptide, protein or antigen encoded by the mRNA, wherein the disease or disorder is caused by a viral infection; (c) a method of treating or preventing a disease or disorder amenable to treatment or prevention by the peptide, protein or antigen encoded by the mRNA, wherein the disease or disorder is influenza; (d) a method of treating or preventing a disease or disorder amenable to treatment or prevention by the peptide, protein or antigen encoded by the mRNA, wherein the disease or disorder is caused by a bacterial infection; (e) a method of treating or preventing a disease or disorder amenable to treatment or prevention by the peptide, protein or antigen encoded by the mRNA, wherein the disease or disorder is chlamydia; (f) a method of treating or preventing a disease or disorder amenable to treatment or prevention by the peptide, protein or antigen encoded by the mRNA, wherein the disease or disorder is induced by infection with P. gingivalis; or (g) a method of treating or preventing a disease or disorder amenable to treatment or prevention by the peptide, protein or antigen encoded by the mRNA, wherein the disease or disorder is acne.
15. The composition for use according to claim 14, wherein the composition is administered intravenously, intrathecally or intramuscularly, or by pulmonary delivery, optionally through nebulization, optionally wherein the composition is administered intramuscularly.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP23306558 | 2023-09-20 | ||
| EP23306558.0 | 2023-09-20 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2025061967A1 true WO2025061967A1 (en) | 2025-03-27 |
Family
ID=88295762
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2024/076467 Pending WO2025061967A1 (en) | 2023-09-20 | 2024-09-20 | "good" buffer-based cationic lipids for nucleic acid delivery |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2025061967A1 (en) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2026003582A2 (en) | 2024-06-27 | 2026-01-02 | Axelyf ehf. | Lipids and lipid nanoparticles |
Citations (27)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4373071A (en) | 1981-04-30 | 1983-02-08 | City Of Hope Research Institute | Solid-phase synthesis of polynucleotides |
| US4401796A (en) | 1981-04-30 | 1983-08-30 | City Of Hope Research Institute | Solid-phase synthesis of polynucleotides |
| US4415732A (en) | 1981-03-27 | 1983-11-15 | University Patents, Inc. | Phosphoramidite compounds and processes |
| US4458066A (en) | 1980-02-29 | 1984-07-03 | University Patents, Inc. | Process for preparing polynucleotides |
| US4500707A (en) | 1980-02-29 | 1985-02-19 | University Patents, Inc. | Nucleosides useful in the preparation of polynucleotides |
| US4668777A (en) | 1981-03-27 | 1987-05-26 | University Patents, Inc. | Phosphoramidite nucleoside compounds |
| US4973679A (en) | 1981-03-27 | 1990-11-27 | University Patents, Inc. | Process for oligonucleo tide synthesis using phosphormidite intermediates |
| US5047524A (en) | 1988-12-21 | 1991-09-10 | Applied Biosystems, Inc. | Automated system for polynucleotide synthesis and purification |
| US5132418A (en) | 1980-02-29 | 1992-07-21 | University Patents, Inc. | Process for preparing polynucleotides |
| US5153319A (en) | 1986-03-31 | 1992-10-06 | University Patents, Inc. | Process for preparing polynucleotides |
| US5262530A (en) | 1988-12-21 | 1993-11-16 | Applied Biosystems, Inc. | Automated system for polynucleotide synthesis and purification |
| US5700642A (en) | 1995-05-22 | 1997-12-23 | Sri International | Oligonucleotide sizing using immobilized cleavable primers |
| US5744335A (en) | 1995-09-19 | 1998-04-28 | Mirus Corporation | Process of transfecting a cell with a polynucleotide mixed with an amphipathic compound and a DNA-binding protein |
| US5780014A (en) | 1995-04-14 | 1998-07-14 | Inhale Therapeutic Systems | Method and apparatus for pulmonary administration of dry powder alpha 1-antitrypsin |
| US5885613A (en) | 1994-09-30 | 1999-03-23 | The University Of British Columbia | Bilayer stabilizing components and their use in forming programmable fusogenic liposomes |
| WO2010144740A1 (en) | 2009-06-10 | 2010-12-16 | Alnylam Pharmaceuticals, Inc. | Improved lipid formulation |
| US20110245776A1 (en) | 2004-01-30 | 2011-10-06 | Mark Anthony Fernance Kendall | Delivery device |
| WO2018089801A1 (en) | 2016-11-10 | 2018-05-17 | Translate Bio, Inc. | Improved process of preparing mrna-loaded lipid nanoparticles |
| US20180264244A1 (en) | 2015-09-28 | 2018-09-20 | Vaxxas Pty Limited | Microprojection arrays with enhanced skin penetrating properties and methods thereof |
| US20180263641A1 (en) | 2015-09-18 | 2018-09-20 | Vaxxas Pty Limited | Microprojection arrays with microprojections having large surface area profiles |
| US20200246450A1 (en) | 2017-08-10 | 2020-08-06 | Vaxxas Pty Limited | Differential coating of microprojections and microneedles on arrays |
| US20210085598A1 (en) | 2018-04-03 | 2021-03-25 | Vaxess Technologies, Inc. | Microneedle comprising silk fibroin applied to a dissolvable base |
| US20220143376A1 (en) | 2019-03-29 | 2022-05-12 | Angus FORSTER | Vaccination using high-density microprojection array patch |
| WO2022099003A1 (en) | 2020-11-06 | 2022-05-12 | Sanofi | Lipid nanoparticles for delivering mrna vaccines |
| WO2022221688A1 (en) | 2021-04-15 | 2022-10-20 | Translate Bio, Inc. | "good"buffer-based cationic lipids |
| US20220339416A1 (en) | 2019-10-09 | 2022-10-27 | Vaxess Technologies, Inc. | Silk fibroin-based microneedles and uses thereof |
| US20230270842A1 (en) | 2020-05-21 | 2023-08-31 | Vaxess Technologies, Inc. | Compositions and devices for vaccine release and uses thereof |
-
2024
- 2024-09-20 WO PCT/EP2024/076467 patent/WO2025061967A1/en active Pending
Patent Citations (27)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4458066A (en) | 1980-02-29 | 1984-07-03 | University Patents, Inc. | Process for preparing polynucleotides |
| US5132418A (en) | 1980-02-29 | 1992-07-21 | University Patents, Inc. | Process for preparing polynucleotides |
| US4500707A (en) | 1980-02-29 | 1985-02-19 | University Patents, Inc. | Nucleosides useful in the preparation of polynucleotides |
| US4973679A (en) | 1981-03-27 | 1990-11-27 | University Patents, Inc. | Process for oligonucleo tide synthesis using phosphormidite intermediates |
| US4415732A (en) | 1981-03-27 | 1983-11-15 | University Patents, Inc. | Phosphoramidite compounds and processes |
| US4668777A (en) | 1981-03-27 | 1987-05-26 | University Patents, Inc. | Phosphoramidite nucleoside compounds |
| US4401796A (en) | 1981-04-30 | 1983-08-30 | City Of Hope Research Institute | Solid-phase synthesis of polynucleotides |
| US4373071A (en) | 1981-04-30 | 1983-02-08 | City Of Hope Research Institute | Solid-phase synthesis of polynucleotides |
| US5153319A (en) | 1986-03-31 | 1992-10-06 | University Patents, Inc. | Process for preparing polynucleotides |
| US5047524A (en) | 1988-12-21 | 1991-09-10 | Applied Biosystems, Inc. | Automated system for polynucleotide synthesis and purification |
| US5262530A (en) | 1988-12-21 | 1993-11-16 | Applied Biosystems, Inc. | Automated system for polynucleotide synthesis and purification |
| US5885613A (en) | 1994-09-30 | 1999-03-23 | The University Of British Columbia | Bilayer stabilizing components and their use in forming programmable fusogenic liposomes |
| US5780014A (en) | 1995-04-14 | 1998-07-14 | Inhale Therapeutic Systems | Method and apparatus for pulmonary administration of dry powder alpha 1-antitrypsin |
| US5700642A (en) | 1995-05-22 | 1997-12-23 | Sri International | Oligonucleotide sizing using immobilized cleavable primers |
| US5744335A (en) | 1995-09-19 | 1998-04-28 | Mirus Corporation | Process of transfecting a cell with a polynucleotide mixed with an amphipathic compound and a DNA-binding protein |
| US20110245776A1 (en) | 2004-01-30 | 2011-10-06 | Mark Anthony Fernance Kendall | Delivery device |
| WO2010144740A1 (en) | 2009-06-10 | 2010-12-16 | Alnylam Pharmaceuticals, Inc. | Improved lipid formulation |
| US20180263641A1 (en) | 2015-09-18 | 2018-09-20 | Vaxxas Pty Limited | Microprojection arrays with microprojections having large surface area profiles |
| US20180264244A1 (en) | 2015-09-28 | 2018-09-20 | Vaxxas Pty Limited | Microprojection arrays with enhanced skin penetrating properties and methods thereof |
| WO2018089801A1 (en) | 2016-11-10 | 2018-05-17 | Translate Bio, Inc. | Improved process of preparing mrna-loaded lipid nanoparticles |
| US20200246450A1 (en) | 2017-08-10 | 2020-08-06 | Vaxxas Pty Limited | Differential coating of microprojections and microneedles on arrays |
| US20210085598A1 (en) | 2018-04-03 | 2021-03-25 | Vaxess Technologies, Inc. | Microneedle comprising silk fibroin applied to a dissolvable base |
| US20220143376A1 (en) | 2019-03-29 | 2022-05-12 | Angus FORSTER | Vaccination using high-density microprojection array patch |
| US20220339416A1 (en) | 2019-10-09 | 2022-10-27 | Vaxess Technologies, Inc. | Silk fibroin-based microneedles and uses thereof |
| US20230270842A1 (en) | 2020-05-21 | 2023-08-31 | Vaxess Technologies, Inc. | Compositions and devices for vaccine release and uses thereof |
| WO2022099003A1 (en) | 2020-11-06 | 2022-05-12 | Sanofi | Lipid nanoparticles for delivering mrna vaccines |
| WO2022221688A1 (en) | 2021-04-15 | 2022-10-20 | Translate Bio, Inc. | "good"buffer-based cationic lipids |
Non-Patent Citations (8)
| Title |
|---|
| "Remington's Pharmaceutical Sciences", MACK PUBLISHING CO. |
| GAO ET AL., BIOCHEM. BIOPHYS. RES. COMM., vol. 179, 1991, pages 280 |
| GOOD, N.E. ET AL.: "Hydrogen Ion Buffers for Biological Research", BIOCHEMISTRY, vol. 5, no. 2, 1966, pages 467 - 477, XP000881106, DOI: 10.1021/bi00866a011 |
| J. PHARMACEUTICAL SCIENCES, vol. 66, 1977, pages 1 - 19 |
| KLIBANOV ET AL., FEBS LETTERS, vol. 268, no. 1, 1990, pages 235 - 237 |
| LASIC, TRENDS BIOTECHNOL., vol. 16, 1998, pages 307 - 321 |
| T. W. GREENEP. G. M. WUTS: "Protecting Groups in Organic Synthesis,", 1999, JOHN WILEY & SONS |
| WOLF ET AL., BIOTECHNIQUES, vol. 23, 1997, pages 139 |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2026003582A2 (en) | 2024-06-27 | 2026-01-02 | Axelyf ehf. | Lipids and lipid nanoparticles |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP7791182B2 (en) | Piperazine-based cationic lipids | |
| JP7791103B2 (en) | Phenolic acid lipid-based cationic lipid | |
| WO2022221688A1 (en) | "good"buffer-based cationic lipids | |
| US20260021055A1 (en) | Asymmetric Piperazine-Based Cationic Lipids | |
| US20230357140A1 (en) | Tes-based cationic lipids | |
| WO2025061967A1 (en) | "good" buffer-based cationic lipids for nucleic acid delivery | |
| WO2024256453A1 (en) | Sterol-based cationic lipids with aromatic head groups | |
| WO2024256457A1 (en) | Tricine and citric acid-based cationic lipids with aromatic head groups | |
| WO2025003755A1 (en) | Helper lipids for nucleic acid delivery | |
| WO2025003759A9 (en) | Dianhydrohexitol based ionizable lipids for nucleic acid delivery | |
| US20250263385A1 (en) | "Good" buffer-based cationic lipids | |
| US20240270707A1 (en) | "Good" buffer-based cationic lipids | |
| AU2024306474A1 (en) | Helper lipids for nucleic acid delivery | |
| US20230150921A1 (en) | Phenolic acid lipid based cationic lipids | |
| HK40099554B (en) | Piperazine-based cationic lipids | |
| HK40099554A (en) | Piperazine-based cationic lipids | |
| WO2026006524A2 (en) | Bis-ester and amide cationic lipids | |
| AU2024310306A1 (en) | Dianhydrohexitol based ionizable lipids for nucleic acid delivery | |
| CN117136182A (en) | Cationic lipids based on "Goode's" buffer |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 24772384 Country of ref document: EP Kind code of ref document: A1 |