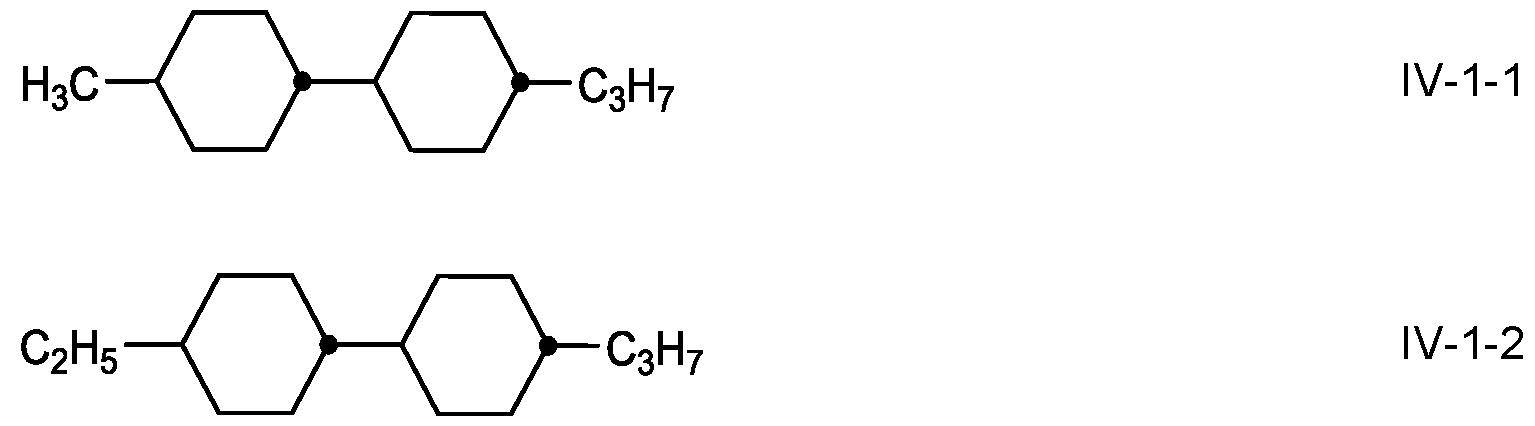WO2025061649A1 - Liquid crystal medium - Google Patents
Liquid crystal medium Download PDFInfo
- Publication number
- WO2025061649A1 WO2025061649A1 PCT/EP2024/075846 EP2024075846W WO2025061649A1 WO 2025061649 A1 WO2025061649 A1 WO 2025061649A1 EP 2024075846 W EP2024075846 W EP 2024075846W WO 2025061649 A1 WO2025061649 A1 WO 2025061649A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- atoms
- compounds
- denotes
- formula
- alkyl
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K19/00—Liquid crystal materials
- C09K19/04—Liquid crystal materials characterised by the chemical structure of the liquid crystal components, e.g. by a specific unit
- C09K19/06—Non-steroidal liquid crystal compounds
- C09K19/08—Non-steroidal liquid crystal compounds containing at least two non-condensed rings
- C09K19/30—Non-steroidal liquid crystal compounds containing at least two non-condensed rings containing saturated or unsaturated non-aromatic rings, e.g. cyclohexane rings
- C09K19/3098—Unsaturated non-aromatic rings, e.g. cyclohexene rings
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K19/00—Liquid crystal materials
- C09K19/04—Liquid crystal materials characterised by the chemical structure of the liquid crystal components, e.g. by a specific unit
- C09K19/06—Non-steroidal liquid crystal compounds
- C09K19/08—Non-steroidal liquid crystal compounds containing at least two non-condensed rings
- C09K19/30—Non-steroidal liquid crystal compounds containing at least two non-condensed rings containing saturated or unsaturated non-aromatic rings, e.g. cyclohexane rings
- C09K19/3001—Cyclohexane rings
- C09K19/3066—Cyclohexane rings in which the rings are linked by a chain containing carbon and oxygen atoms, e.g. esters or ethers
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K19/00—Liquid crystal materials
- C09K19/52—Liquid crystal materials characterised by components which are not liquid crystals, e.g. additives with special physical aspect: solvents, solid particles
- C09K19/60—Pleochroic dyes
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K19/00—Liquid crystal materials
- C09K19/04—Liquid crystal materials characterised by the chemical structure of the liquid crystal components, e.g. by a specific unit
- C09K19/06—Non-steroidal liquid crystal compounds
- C09K19/062—Non-steroidal liquid crystal compounds containing one non-condensed benzene ring
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K19/00—Liquid crystal materials
- C09K19/04—Liquid crystal materials characterised by the chemical structure of the liquid crystal components, e.g. by a specific unit
- C09K19/06—Non-steroidal liquid crystal compounds
- C09K19/08—Non-steroidal liquid crystal compounds containing at least two non-condensed rings
- C09K19/30—Non-steroidal liquid crystal compounds containing at least two non-condensed rings containing saturated or unsaturated non-aromatic rings, e.g. cyclohexane rings
- C09K19/3001—Cyclohexane rings
- C09K19/3028—Cyclohexane rings in which at least two rings are linked by a carbon chain containing carbon to carbon single bonds
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K19/00—Liquid crystal materials
- C09K19/04—Liquid crystal materials characterised by the chemical structure of the liquid crystal components, e.g. by a specific unit
- C09K19/06—Non-steroidal liquid crystal compounds
- C09K19/34—Non-steroidal liquid crystal compounds containing at least one heterocyclic ring
- C09K19/3491—Non-steroidal liquid crystal compounds containing at least one heterocyclic ring having sulfur as hetero atom
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K19/00—Liquid crystal materials
- C09K19/04—Liquid crystal materials characterised by the chemical structure of the liquid crystal components, e.g. by a specific unit
- C09K19/06—Non-steroidal liquid crystal compounds
- C09K19/34—Non-steroidal liquid crystal compounds containing at least one heterocyclic ring
- C09K19/3491—Non-steroidal liquid crystal compounds containing at least one heterocyclic ring having sulfur as hetero atom
- C09K19/3497—Non-steroidal liquid crystal compounds containing at least one heterocyclic ring having sulfur as hetero atom the heterocyclic ring containing sulfur and nitrogen atoms
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K19/00—Liquid crystal materials
- C09K19/04—Liquid crystal materials characterised by the chemical structure of the liquid crystal components, e.g. by a specific unit
- C09K2019/0444—Liquid crystal materials characterised by the chemical structure of the liquid crystal components, e.g. by a specific unit characterized by a linking chain between rings or ring systems, a bridging chain between extensive mesogenic moieties or an end chain group
- C09K2019/0466—Liquid crystal materials characterised by the chemical structure of the liquid crystal components, e.g. by a specific unit characterized by a linking chain between rings or ring systems, a bridging chain between extensive mesogenic moieties or an end chain group the linking chain being a -CF2O- chain
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K19/00—Liquid crystal materials
- C09K19/04—Liquid crystal materials characterised by the chemical structure of the liquid crystal components, e.g. by a specific unit
- C09K19/06—Non-steroidal liquid crystal compounds
- C09K19/08—Non-steroidal liquid crystal compounds containing at least two non-condensed rings
- C09K19/30—Non-steroidal liquid crystal compounds containing at least two non-condensed rings containing saturated or unsaturated non-aromatic rings, e.g. cyclohexane rings
- C09K19/3001—Cyclohexane rings
- C09K19/3003—Compounds containing at least two rings in which the different rings are directly linked (covalent bond)
- C09K2019/3004—Cy-Cy
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K19/00—Liquid crystal materials
- C09K19/04—Liquid crystal materials characterised by the chemical structure of the liquid crystal components, e.g. by a specific unit
- C09K19/06—Non-steroidal liquid crystal compounds
- C09K19/08—Non-steroidal liquid crystal compounds containing at least two non-condensed rings
- C09K19/30—Non-steroidal liquid crystal compounds containing at least two non-condensed rings containing saturated or unsaturated non-aromatic rings, e.g. cyclohexane rings
- C09K19/3001—Cyclohexane rings
- C09K19/3003—Compounds containing at least two rings in which the different rings are directly linked (covalent bond)
- C09K2019/3009—Cy-Ph
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K19/00—Liquid crystal materials
- C09K19/04—Liquid crystal materials characterised by the chemical structure of the liquid crystal components, e.g. by a specific unit
- C09K19/06—Non-steroidal liquid crystal compounds
- C09K19/08—Non-steroidal liquid crystal compounds containing at least two non-condensed rings
- C09K19/30—Non-steroidal liquid crystal compounds containing at least two non-condensed rings containing saturated or unsaturated non-aromatic rings, e.g. cyclohexane rings
- C09K19/3001—Cyclohexane rings
- C09K19/3003—Compounds containing at least two rings in which the different rings are directly linked (covalent bond)
- C09K2019/301—Cy-Cy-Ph
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K19/00—Liquid crystal materials
- C09K19/04—Liquid crystal materials characterised by the chemical structure of the liquid crystal components, e.g. by a specific unit
- C09K19/06—Non-steroidal liquid crystal compounds
- C09K19/08—Non-steroidal liquid crystal compounds containing at least two non-condensed rings
- C09K19/30—Non-steroidal liquid crystal compounds containing at least two non-condensed rings containing saturated or unsaturated non-aromatic rings, e.g. cyclohexane rings
- C09K19/3001—Cyclohexane rings
- C09K19/3003—Compounds containing at least two rings in which the different rings are directly linked (covalent bond)
- C09K2019/3016—Cy-Ph-Ph
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K19/00—Liquid crystal materials
- C09K19/04—Liquid crystal materials characterised by the chemical structure of the liquid crystal components, e.g. by a specific unit
- C09K19/06—Non-steroidal liquid crystal compounds
- C09K19/08—Non-steroidal liquid crystal compounds containing at least two non-condensed rings
- C09K19/30—Non-steroidal liquid crystal compounds containing at least two non-condensed rings containing saturated or unsaturated non-aromatic rings, e.g. cyclohexane rings
- C09K19/3001—Cyclohexane rings
- C09K19/3003—Compounds containing at least two rings in which the different rings are directly linked (covalent bond)
- C09K2019/3027—Compounds comprising 1,4-cyclohexylene and 2,3-difluoro-1,4-phenylene
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K19/00—Liquid crystal materials
- C09K19/04—Liquid crystal materials characterised by the chemical structure of the liquid crystal components, e.g. by a specific unit
- C09K19/06—Non-steroidal liquid crystal compounds
- C09K19/08—Non-steroidal liquid crystal compounds containing at least two non-condensed rings
- C09K19/30—Non-steroidal liquid crystal compounds containing at least two non-condensed rings containing saturated or unsaturated non-aromatic rings, e.g. cyclohexane rings
- C09K19/3001—Cyclohexane rings
- C09K19/3066—Cyclohexane rings in which the rings are linked by a chain containing carbon and oxygen atoms, e.g. esters or ethers
- C09K19/3068—Cyclohexane rings in which the rings are linked by a chain containing carbon and oxygen atoms, e.g. esters or ethers chain containing -COO- or -OCO- groups
- C09K2019/3071—Cy-Cy-COO-Cy
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K19/00—Liquid crystal materials
- C09K19/04—Liquid crystal materials characterised by the chemical structure of the liquid crystal components, e.g. by a specific unit
- C09K19/06—Non-steroidal liquid crystal compounds
- C09K19/34—Non-steroidal liquid crystal compounds containing at least one heterocyclic ring
- C09K19/3402—Non-steroidal liquid crystal compounds containing at least one heterocyclic ring having oxygen as hetero atom
- C09K2019/3422—Non-steroidal liquid crystal compounds containing at least one heterocyclic ring having oxygen as hetero atom the heterocyclic ring being a six-membered ring
Definitions
- the present invention relates to a liquid crystal (LC) medium and to the use thereof for optical, electro-optical and electronic purposes, in particular in LC displays, especially in IPS, FFS, VA or PS-VA displays.
- LC liquid crystal
- Liquid crystals have found widespread use since the first commercially usable liquid-crystalline compounds were found.
- Liquid crystal displays are used in many areas for the display of information. LCDs are used both for direct-view displays and for projection-type displays.
- TN LCDs have the disadvantage of a strong viewing-angle dependence of the contrast.
- FFS far-field switching
- FFS displays have been reported, see inter alia S.H. Jung et al., Jpn. J. Appl. Phys., Volume 43, No. 3, 2004, 1028, which contain two electrodes on the same substrate, one of which is structured in a comb- shaped manner and the other is unstructured.
- a strong, so-called “fringe field” is thereby generated, i.e. a strong electric field close to the edge of the electrodes, and, throughout the cell, an electric field which has both a strong vertical component and also a strong horizontal component.
- FFS displays have a low viewing-angle dependence of the contrast.
- FFS displays usually contain an LC medium with positive dielectric anisotropy, and an alignment layer, usually of polyimide, which provides planar alignment to the molecules of the LC medium.
- FFS displays can be operated as active-matrix or passive-matrix displays.
- active-matrix displays individual pixels are usually addressed by integrated, non- linear active elements, such as for example transistors, for example thin-film transistors ("TFTs"), while in the case of passive-matrix displays individual pixels are usually addressed by the multiplex method.
- TFTs thin-film transistors
- IPS in-plane switching
- FFS fringe-field switching
- HB-FFS High Brightness FFS
- FFS displays have been described in S.H. Lee et al., Appl. Phys. Lett. 73(20), 1998, 2882-2883 and S.H. Lee et al., Liquid Crystals 39(9), 2012, 1141-1148, which have similar electrode design and layer thickness as FFS displays, but comprise a layer of an LC medium with negative dielectric anisotropy instead of an LC medium with positive dielectric anisotropy.
- the LC medium with negative dielectric anisotropy can show a more favourable director orientation that has less tilt and more twist orientation compared to the LC medium with positive dielectric anisotropy, as a result of which these displays may have a higher transmission.
- the displays further comprise an alignment layer, preferably of polyimide provided on at least one of the substrates that is in contact with the LC medium and induces planar alignment of the LC molecules of the LC medium.
- These displays are also known as "Ultra Brightness FFS” (UB-FFS) mode displays. These displays require an LC medium with high reliability. Especially for monitor, mobile, video gaming and TV applications it is desirable to obtain favourably short response times, a wide viewing angle, low power consumption, a high transmittance and a suitably high contrast ratio of the LC displays.
- the retardation may be lowered by reducing the cell gap and/or by decreasing the optical anisotropy of the LC medium to improve the dark state quality.
- the elastic constants of the LC medium may however affect the operating voltage and the response time of the device.
- An object of the present invention is therefore to provide improved liquid crystalline media for use in electro-optical devices and in particular displays with a favourable electro-optical performance, in particular with a high contrast ratio and a good black state while at the same time enabling high brightness or a high transmittance in one optical state, a low threshold voltage, fast addressing times and a favourable reliability and stability, in particular at low temperatures and at high temperatures. Further objects of the present invention are immediately evident to the person skilled in the art from the following detailed description. Further, it is an object of the present invention to provide alternative media in addition to existing media known to the skilled person in order to broaden the range of available materials what allows for a more specific optimisation of a particular display.
- the present invention in particular provides the following items including main aspects, preferred embodiments and particular features, which respectively alone and in combination contribute to solving the above objects and eventually provide additional advantages.
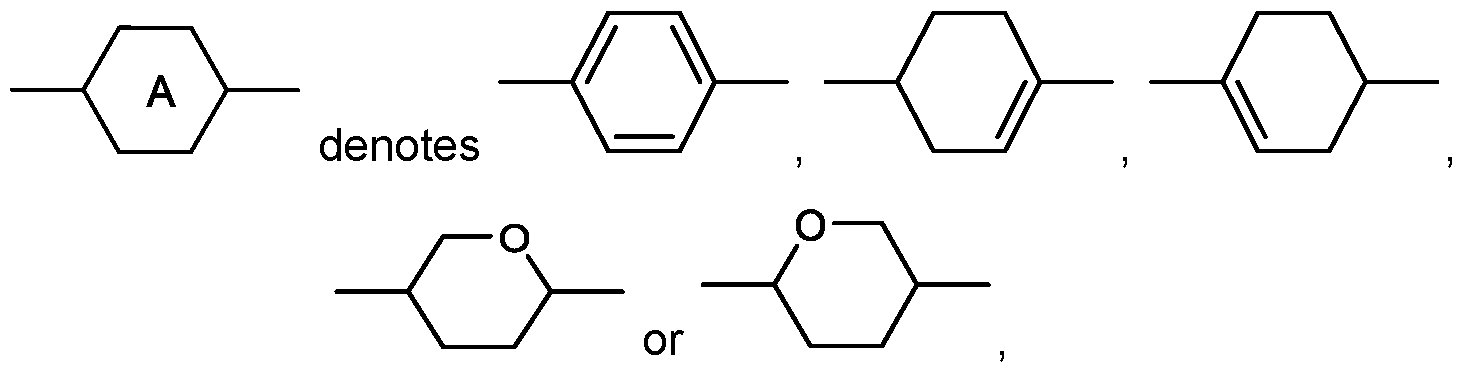
- the present invention relates to a liquid crystal medium comprising a) one or more compounds of formula I in which
- R 1 denotes H, a straight chain alkyl or alkoxy radical having 1 to 15 C atoms, or a branched alkyl or alkoxy radical having 3 to 15 C atoms, where one or more CH2 groups in these radicals may each be replaced, independently of one another, by
- -CH CH-, -OC-, -CF2O-, -OCF2-, -O-, -CO-O- or -O-CO- in such a way that O atoms are not linked directly to one another, and in which, in addition, one or more H atoms may be replaced by halogen, preferably alkyl or alkenyl having up to 7 C atoms,
- Y 1 denotes H or CH3, preferably H, n is 0 or 1 , preferably 1 v is 1 , 2, 3, 4, 5, or 6, and b) one or more dichroic dyes.
- a medium according to the invention in a display can favourably enhance the obtainable contrast ratio of an electro-optical devices, while it has been further found that at the same time a suitably high transmittance or brightness, fast response times, a low operating voltage and low power consumption and a wide viewing angle of the devices may be maintained.
- the one or more dichroic dyes are provided in relatively small and limited yet effective amounts in the liquid-crystalline media to efficiently improve the dark state and the overall contrast of the devices and in particular displays, e.g. FFS, HB-FFS and especially UB-FFS displays, without however affecting the desired device transmittance or brightness or only reducing the device transmittance or brightness to a minimal or even negligible extent.
- displays e.g. FFS, HB-FFS and especially UB-FFS displays
- the liquid-crystalline medium comprises one or more dichroic dyes, preferably in a total amount of 2.0% by weight or less.
- the total amount of the one or more dichroic dyes in the liquid-crystalline medium is 1.0% by weight or less. It is further preferred that the total amount of the one or more dichroic dyes in the liquid-crystalline medium is 0.5% by weight or less, more preferably 0.2% by weight or less.
- a dichroic dye is taken to mean a light-absorbing compound in which the absorption properties are dependent on the orientation of the compound relative to the direction of polarization of the light.
- a dichroic dye compound in accordance with the present invention typically has an elongated shape, i.e. the compound is significantly longer in one spatial direction, i.e. along the longitudinal axis, than in the other two spatial directions.
- the dichroic dye absorbs, or respectively preferentially absorbs, light in one orientation so that light transmission may be modulated by changing the orientation of the dichroic dye.
- liquid crystal host is doped with dichroic dye molecules in relatively small, limited amounts.
- dye-doped LC media are used particularly preferably for FFS and UB-FFS modes, however also other display modes as described above and below can be employed.
- the dichroic dye compounds are present in the liquid-crystalline medium in solution.
- the dichroic dye molecules as used according to the invention therefore preferably exhibit suitable and sufficient solubility in the liquid crystal host.
- the dye compounds preferably also favourably contribute to the stability and reliability of the media.
- Each of the one or more dichroic dyes is preferably present in the liquid-crystalline medium in a proportion of 0.002% by weight to 2.0% by weight, more preferably 0.003% by weight to 1.0% by weight, still more preferably 0.005% by weight to 0.5% by weight, even more preferably 0.01% by weight to 0.3% by weight, and particularly preferably 0.02% by weight to 0.2% by weight, based on the overall weight of the entire medium.
- the one or more dichroic dyes are present in the liquid-crystalline medium overall in a total concentration which is in the range of from 0.003% by weight to 2.0% by weight, more preferably 0.004% by weight to 1 .5% by weight, still more preferably 0.005% by weight to 1.0% by weight, yet more preferably 0.01% by weight to 0.5% by weight, even more preferably 0.02% by weight to 0.3% by weight, and particularly preferably 0.05% by weight to 0.2% by weight.
- the concentration of the dye(s) is favourably chosen such that the proper performance of the obtained liquid-crystalline material is ensured, in particular in terms of the desired reduction of the light leakage in the dark state and the improved black image quality and overall contrast, while suitably maintaining other device properties such as response times, operating voltage and brightness.
- the upper limit of the dye concentration is provided in particular in view of maintaining the bright state performance of the device such that transmittance is not affected or only to a minimal extent.
- Dichroic dyes may preferably be selected from for example azo dyes, anthraquinones, thiophenolanthraquinones, methine compounds, azomethine compounds, merocyanine compounds, naphthoquinones, tetrazines, pyrromethene dyes, malononitrile dyes, nickel dithiolenes, (metal) phthalocyanines, (metal) naphthalocyanines and (metal) porphyrins, rylenes, in particular perylenes and terylenes, thiadiazole dyes, thienothiadiazole dyes, benzothiadiazoles, thiadiazoloquinoxalines, and diketopyrrolopyrroles.
- azo dyes anthraquinones, thiophenolanthraquinones, methine compounds, azomethine compounds, merocyanine compounds, naphthoquinones, tetrazines, pyrrometh
- azo compounds anthraquinones, thiophenolanthraquinones, benzothiadiazoles, in particular as described in WO 2014/187529 and in WO 2020/104563 A1, diketopyrrolopyrroles, in particular as described in WO 2015/090497, thiadiazoloquinoxalines, in particular as described in WO 2016/177449 and in WO 2020/104563 A1 , and rylenes, in particular as described in WO 2014/090373.
- the one or more dichroic dyes are selected from azo compounds, benzothiadiazoles and thiadiazoloquinoxalines.
- the liquid-crystalline medium preferably comprises one, two, three, four, five, six, seven, eight, nine or ten different dichroic dyes, particularly preferably two, three, four, five or six dichroic dyes.
- the medium contains at least three different dichroic dyes.
- the absorption spectra of the dichroic dyes contained in the medium or respectively the switchable layer preferably complement one another in such a way that the impression of a black colour, or respectively a colour-neutral appearance, arises for the eye.
- two or more, more preferably three or more dichroic dyes are used in the liquid-crystalline medium to preferably cover a large part of the visible spectrum.
- the precise way in which a mixture of dyes which appears black or grey to the eye can be prepared is known in the art and is described, for example, in M. Richter, Einbowung in die Farbmetrik [Introduction to Colorimetry], 2nd Edition, 1981 , ISBN 3 11-008209-8, Walter de Gruyter & Co.
- the setting of the colour location of a mixture of dyes is described in the area of colorimetry.
- the spectra of the individual dyes are calculated taking into account the Lambert-Beer law to give an overall spectrum and converted into the corresponding colour locations and luminance values under the associated illumination, for example illuminant D65 for daylight, in accordance with the rules of colorimetry.
- the position of the white point is fixed by the respective illuminant, for example D65, and is quoted in tables, for example in the reference above.
- Different colour locations can be set by changing the proportions of the various dyes.
- the medium and the switchable layer comprise one or more dichroic dyes which absorb light in the visible spectrum, which herein is defined as light having a wavelength of from 380 nm to 780 nm.
- the dichroic dyes provided in the medium and the switchable layer are preferably selected from the dye classes indicated in B. Bahadur, Liquid Crystals - Applications and Uses, Vol. 3, 1992, World Scientific Publishing, Section 11.2.1, and particularly preferably from the explicit compounds given in the table present therein.
- Said dyes belong to the classes of dichroic dyes which are known in the art and have been described in the literature.
- anthraquinone dyes are described in EP 34832, EP 44893, EP 48583, EP 54217, EP 56492, EP 59036, GB 2065158, GB 2065695, GB 2081736, GB 2082196, GB 2094822, GB 2094825, JP A 55- 123673, DE 3017877, DE 3040102, DE 3115147, DE 3115762, DE 3150803 and DE 3201120, naphthoquinone dyes are described in DE 3126108 and DE 3202761 , azo dyes in EP 43904, DE 3123519, WO 82/2054, GB 2079770, JP-A 56-57850, JP-A 56- 104984, US 4308161, US 4308162, US 4340973, T.
- R denotes straight chain alkyl having 1 to 20 C atoms, or branched alkyl having 3 to 20 C atoms, or straight chain alkenyl having 2 to 20 C atoms, or branched alkenyl having 3 to 20 C atoms, and (O) denotes O or a single bond.
- Preferred compounds of formula I are selected from the following sub-formulae:
- Very preferred compounds of formula I are the compounds 1-1 to 1-16.
- the medium comprises one or more compounds of formula IA, in which the occurring groups and parameters have the meanings given above under formula I, and
- R 2 denotes , in which r is 0, 1 , 2, 3, 4, 5 or 6, and s is 1 , 2 or 3, preferably 1 or 3.
- Preferred compounds of formula IA are the compounds IA-1 to I A- 14.
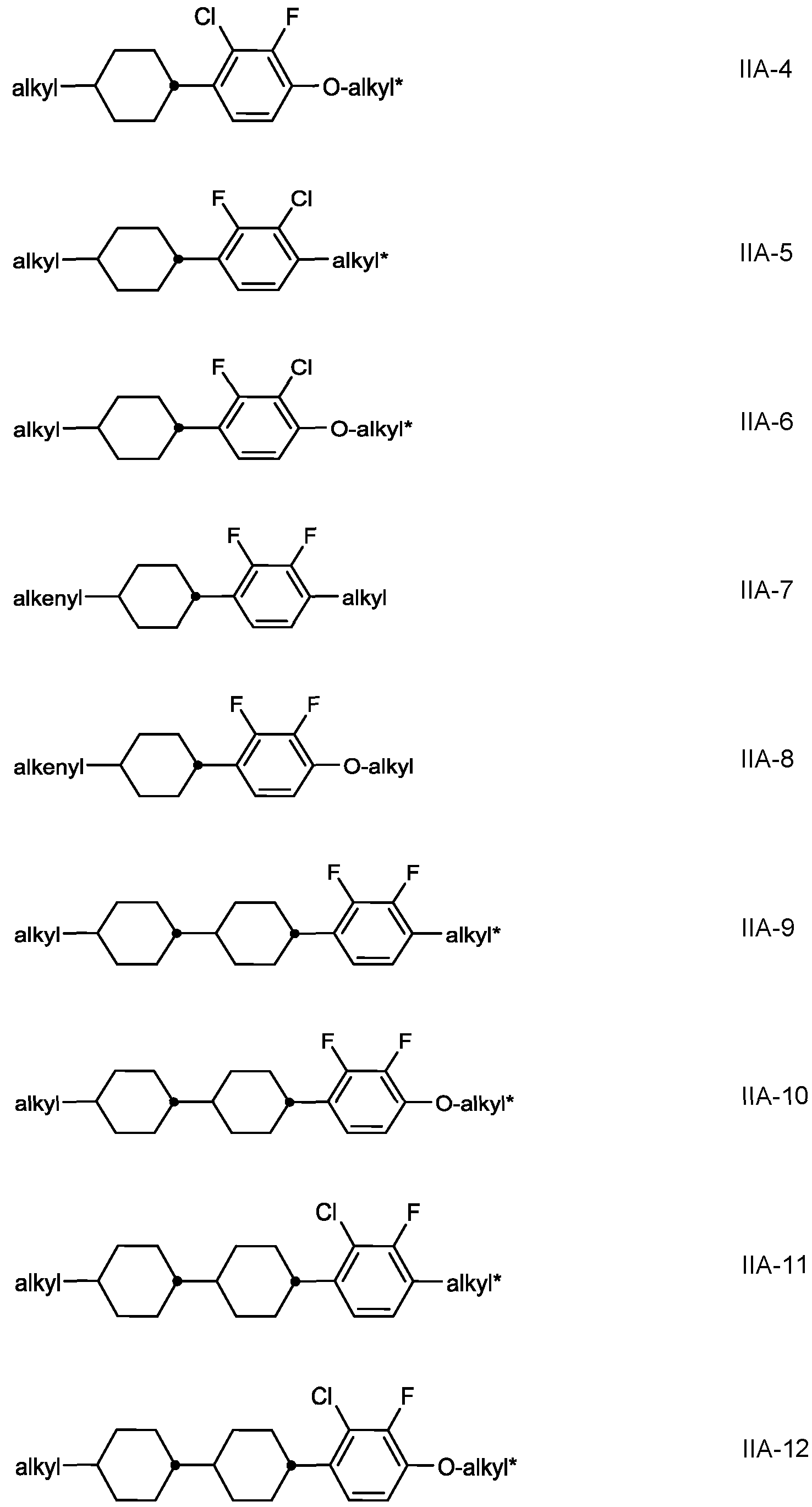
- the medium further comprises one or more compounds selected from the group of compounds of the formulae IIA, IIB, IIC and IID, in which
- R 2A , R 2B , R 2C and R 2D identically or differently, denote H, a straight chain alkyl or alkoxy radical having 1 to 15 C atoms, or a branched alkyl or alkoxy radical having 3 to 15 C atoms, where one or more CH2 groups in these radicals may each be replaced, independently of one another, by
- -CH CH-, -C ⁇ C-, -CF2O-, -OCF2-, -O-, -CO-O- or -O-CO- in such a way that O atoms are not linked directly to one another, and in which, in addition, one or more H atoms may be replaced by halogen, preferably alkyl or alkenyl having up to 7 C atoms,
- L 1 to L 4 each, independently of one another, denote F, Cl, CF3 or CHF2,
- Y denotes H, F, Cl, CF3, CHF2 or CH3, preferably H or CH3, particularly preferably H,
- (O) denotes O or a single bond
- p denotes 0, 1 or 2
- q denotes 0 or 1
- v denotes an integer from 1 to 6.
- Z 2 may have identical or different meanings.
- Z 2 and Z 2B may have identical or different meanings.
- Z 2 and Z 2D may have identical or different meanings.
- R 2A , R 2B , R 2C and R 2D each preferably denote alkyl having 1 to 6 C atoms, in particular CH 3 , C 2 H 5 , n-C 3 H 7 , n-C 4 H 9 , n-C 5 H 11 .
- Z 2 and Z 2B in the formulae IIA and IIB preferably each, independently of one another, denote a single bond, furthermore a -C 2 H 4 - bridge.
- Z 2D is preferably a single bond.
- (O)C v H 2v+1 preferably denotes OC v H 2v+1 .
- (O)C v H 2v+1 preferably denotes C v H 2v+1 .
- L 3 and L 4 preferably each denote F.
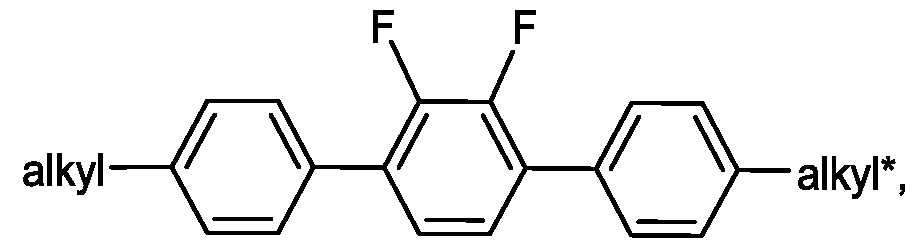
- Preferred compounds of the formulae IIA IIB IIC and IID are indicated below:
- alkyl and alkyl* each, independently of one another, denote a straight-chain alkyl radical having 1-6 C atoms
- alkenyl denotes a straight-chain alkenyl radical having 2-6 C atoms
- (O) denotes an oxygen atom or a single bond.
- Particularly preferred mixtures according to the invention comprise one or more compounds of the formulae IIA-2, IIA-8, IIA-10, IIA-16, 11-18, IIA-40, IIA-41 , IIA-42, IIA- 43, IIB-2, IIB-10, IIB-16, IIC-1 , and IID-4.
- the proportion of compounds of the formulae IIA and/or IIB in the mixture as a whole is preferably at least 20 % by weight.
- Particularly preferred media according to the invention comprise at least one compound of the formula IIC-1 , in which alkyl and alkyl* have the meanings indicated above, preferably in amounts of > 3 % by weight, in particular > 5 % by weight and particularly preferably 5 to 25 % by weight.
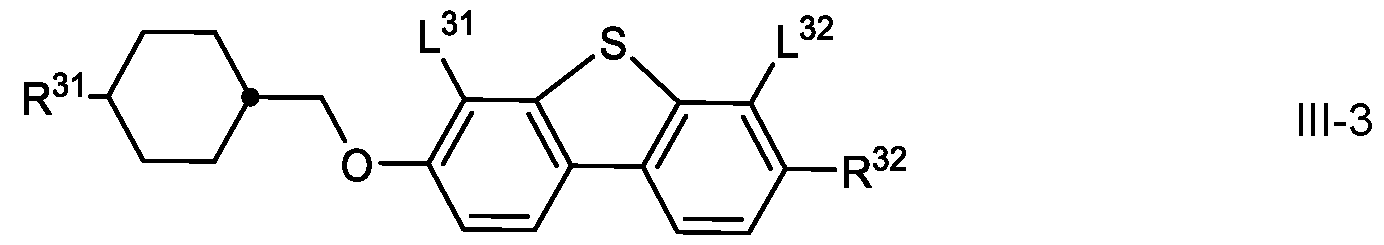
- the medium according to the invention preferably comprises one or more compounds of formula III in which
- R 31 and R 32 each, independently of one another, denote H, an alkyl or alkoxy radical having 1 to 15 C atoms, where one or more CH 2 groups in these radicals may each be replaced, independently of one another, by
- L 31 and L 32 each, independently of one another, denote H, F, Cl, CF3 or CHF2, preferably H or F, most preferably F, and
- W denotes O or S, preferably S.
- the compounds of formula III are preferably selected from the compounds of the formula 111-1 and/or III-2 in which the occurring groups have the same meanings as given under formula III above and preferably
- R 31 and R 32 each, independently of one another, an alkyl, alkenyl or alkoxy radical having up to 15 C atoms, more preferably one or both of them denote an alkoxy radical having 1 to 7 C atoms, and
- L 31 and L 32 each preferably denote F.
- the compounds of the formula 111-1 are selected from the group of compounds of the formulae 111-1-1 to 111-1-11 , preferably of formula 111-1-6, alkyl and alkyl* each, independently of one another, denote a straight-chain alkyl radical having 1-6 C atoms, alkenyl and alkenyl* each, independently of one another, denote a straight-chain alkenyl radical having 2-6 C atoms, alkoxy and alkoxy* each, independently of one another, denote a straight-chain alkoxy radical having 1-6 C atoms, and L 31 and L 32 each, independently of one another, denote F or Cl, preferably both F.
- the compounds of the formula III-2 are selected from the group of compounds of the formulae 111-2-1 to 111-2-10, preferably of formula III-2-6, alkyl and alkyl* each, independently of one another, denote a straight-chain alkyl radical having 1-6 C atoms, alkenyl and alkenyl* each, independently of one another, denote a straight-chain alkenyl radical having 2-6 C atoms, alkoxy and alkoxy* each, independently of one another, denote a straight-chain alkoxy radical having 1-6 C atoms, and L 31 and L 32 each, independently of one another, denote F or Cl, preferably both F.
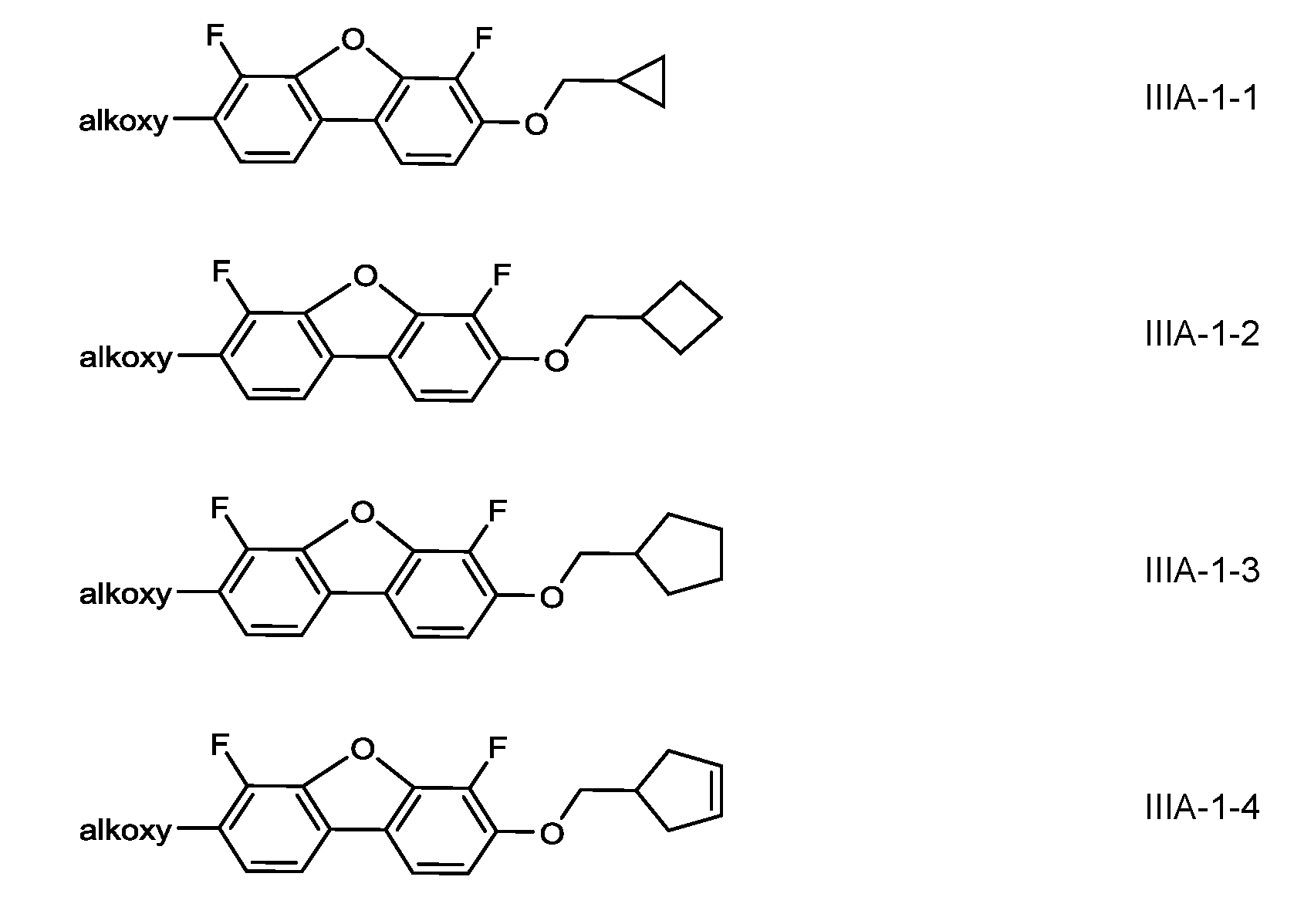
- the medium comprises one or more compounds of the formula IIIA-1 and/or IIIA-2
- R IIIA denotes alkyl or alkenyl having up to 7 C atoms or a group Cy-C m H 2m+1 -, m and n are, identically or differently, 0, 1 , 2, 3, 4, 5 or 6, preferably 1 , 2 or 3, very preferably 1 ,
- Cy denotes a cycloaliphatic group having 3, 4 or 5 ring atoms, which is optionally substituted with alkyl or alkenyl each having up to 3 C atoms, or with halogen or CN, and preferably denotes cyclopropyl, cyclobutyl or cyclopentyl
- the compounds of formula IIIA-1 and/or IIIA-2 are contained in the medium either alternatively or additionally to the compounds of formula III, preferably additionally.
- Very preferred compounds of the formulae IIIA-1 and IIIA-2 are the following: in which alkoxy denotes a straight-chain alkoxy radical having 1-6 C atoms or alternatively -(CH2) n F in which n is 2,3,4, or 5, preferably C2H4F.
- L 31 and L 32 each, independently of one another, denote F or Cl, preferably both F.
- the compounds of formula III-3 are preferably selected from the group of compounds of the formulae 111-3-1 to 111-3-10: in which R 32 denotes alkyl having 1 to 7 C-atoms, preferably ethyl, n-propyl or n-butyl, or alternatively cyclopropylmethyl, cyclobutylmethyl or cyclopentyl methyl or alternatively -(CH2) n F in which n is 2,3,4, or 5, preferably C2H4F.
- the medium comprises one or more compounds of the formulae III-4 to III-6, preferably of formula II I-5, in which the parameters have the meanings given above, R 31 preferably denotes straight-chain alkyl and R 32 preferably denotes alkoxy, each having 1 to 7 C atoms.
- the medium comprises one or more compounds of the formula I selected from the group of compounds of formulae 111-7 to 111-9, preferably of formula 111-8, in which the parameters have the meanings given above, R 31 preferably denotes straight-chain alkyl and R 32 preferably denotes alkoxy each having 1 to 7 C atoms.
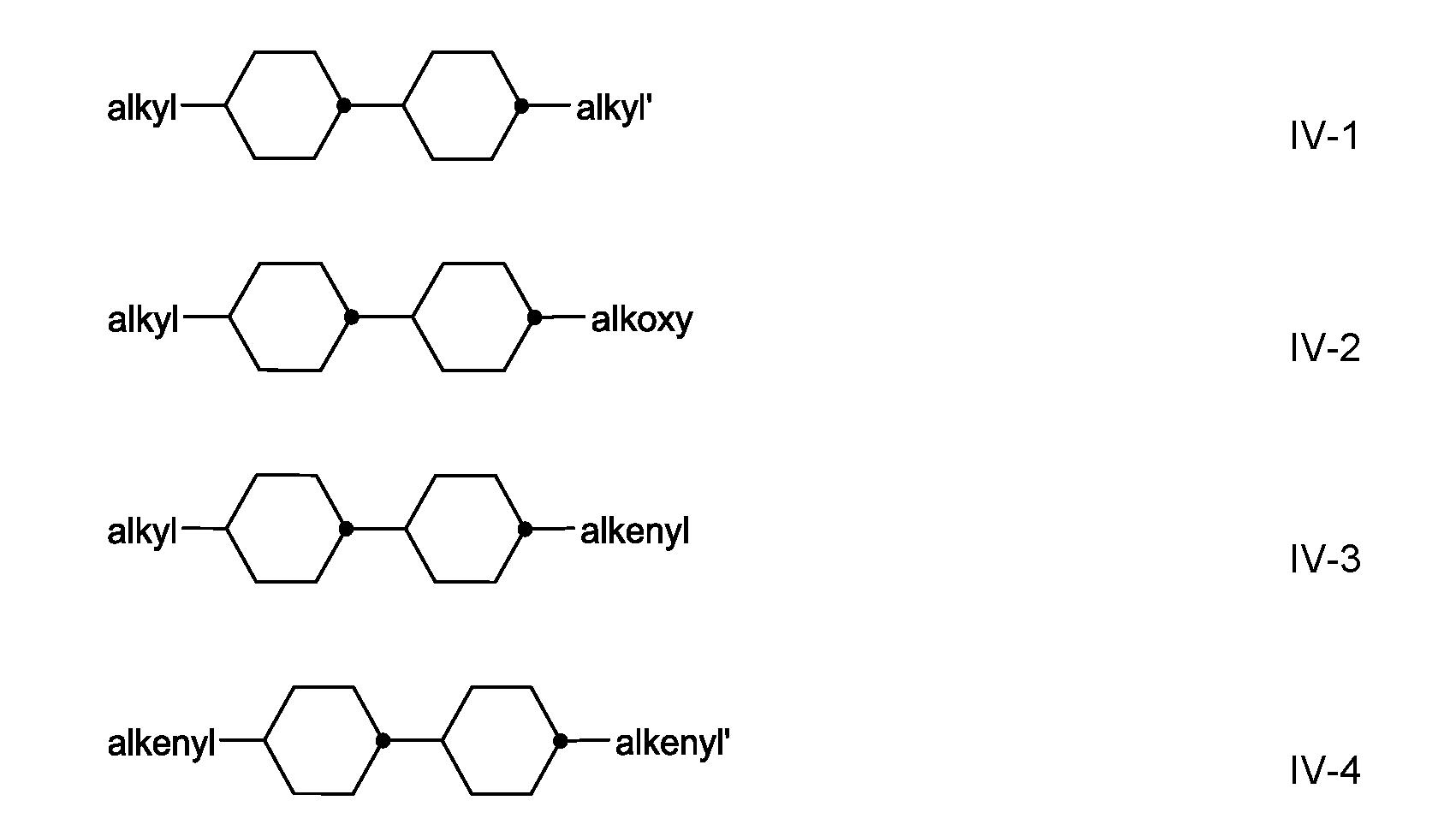
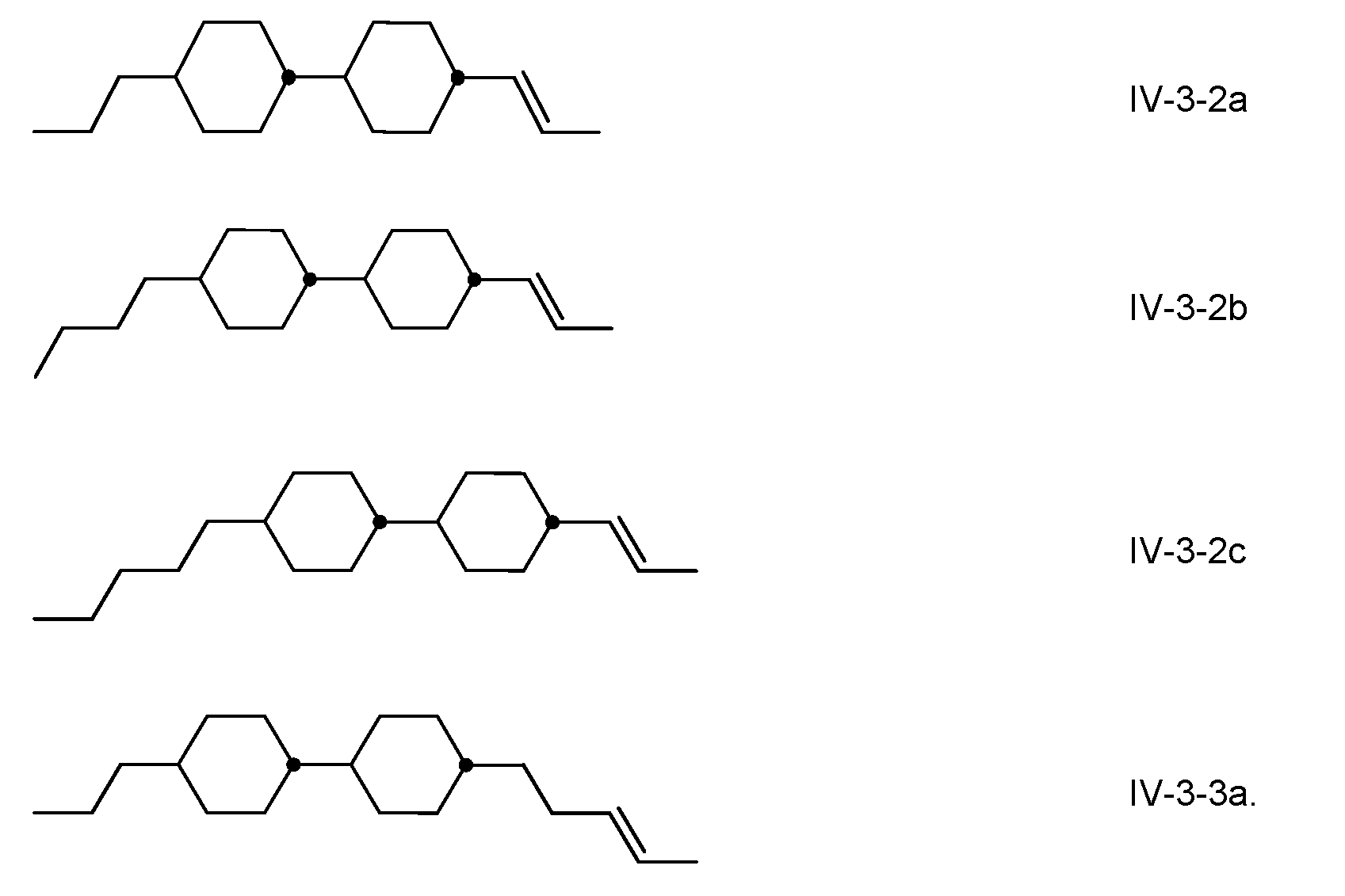
- the medium comprises one or more compounds of the formula IV, in which
- R 42 denotes an unsubstituted alkyl radical having 1 to 7 C atoms or an unsubstituted alkoxy radical having 1 to 6 C atoms, both preferably having 2 to 5 C atoms, an unsubstituted alkenyl radical having 2 to 7 C atoms, preferably having 2, 3 or 4 C atoms, more preferably a vinyl radical or a 1 -propenyl radical and in particular a vinyl radical.
- the compounds of the formula IV are preferably selected from the group of the compounds of the formulae I V-1 to IV-4, in which alkyl and alkyl’, independently of one another, denote alkyl having 1 to 7 C atoms, preferably having 2 to 5 C atoms, alkenyl denotes an alkenyl radical having 2 to 5 C atoms, preferably having
- alkenyl denotes an alkenyl radical having 2 to 5 C atoms, preferably having
- alkoxy denotes alkoxy having 1 to 5 C atoms, preferably having 2 to 4 C atoms.
- the medium comprises one or more compounds selected from the compounds of the formulae IV-1-1 to IV-1-6
- the medium according to the invention comprises one or more compounds of the formulae I -2-1 and/or I -2-2
- the medium according to the invention comprises a compound of formula IV-4, in particular selected from the compounds of the formulae IV-4-1 and IV-4-2
- the liquid-crystalline medium preferably additionally comprises one or more compounds of the formula IVa,
- R 41 and R 42 each, independently of one another, denote a straight-chain alkyl, alkenyl, alkoxyalkyl or alkoxy radical having up to 12 C atoms, and
- alkyl and alkyl* each, independently of one another, denote a straight-chain alkyl radical having 1 to 6 C atoms.
- the medium according to the invention preferably comprises at least one compound of the formula IVa-1 and/or formula IVa-2.
- the proportion of compounds of the formula IVa in the mixture as a whole is preferably at least 5 % by weight
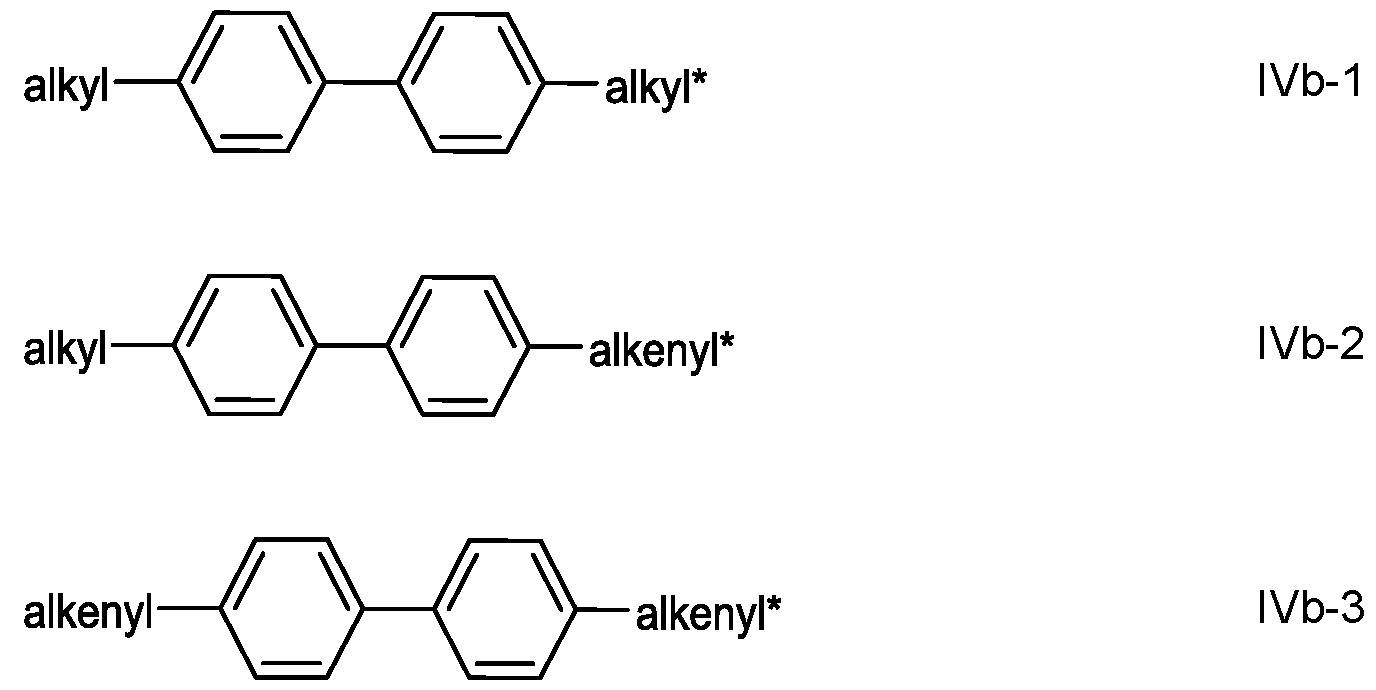
- the medium comprises one or more compounds of formula IVb-1 to IVb-3 in which alkyl and alkyl* each, independently of one another, denote a straight- chain alkyl radical having 1 to 6 C atoms, and alkenyl and alkenyl* each, independently of one another, denote a straight- chain alkenyl radical having 2 to 6 C atoms.
- the compounds of the formula IVb-1 to IVb-3 are particularly preferred.
- biphenyls of the formulae IVb-1 to IVb-3 are in which alkyl* denotes an alkyl radical having 1 to 6 C atoms and preferably denotes n-propyl.
- the medium according to the invention particularly preferably comprises one or more compounds of the formulae IVb-1-1 and/or IVb-2-3.
- the medium according to the invention comprises one or more compounds of formula V in which
- R 51 , R 52 denote alkyl having 1 to 7 C atoms, alkoxy having 1 to 7 C atoms, or alkoxyalkyl, alkenyl or alkenyloxy having 2 to 7 C atoms,
- R 51 and R 52 preferably each, independently of one another, denote straight-chain alkyl having 1 to 7 C atoms or alkenyl having 2 to 7 C atoms.
- the medium according to the invention comprises one or more compounds of the formula CL in which
- X L denotes F, Cl, CN, CHF2, CF3, OCF3, or, identically or differently, has one of the meanings of R L ,
- Y L denotes H, F, Cl or CH3.
- the compounds of formula CL are preferably selected form the group of compounds of the formulae CL-1 , CL-2 and CL-3: in which
- R L1 and R L2 identically or differently, have the meanings given above for R L in formula CL and, preferably denote alkyl or alkenyl having 1 to 7 C atoms or 2 to 7 C atoms, respectively, in which a CH2 group may be replaced by cyclopropane- 1 , 2-diyl.
- Very preferred compounds of the formula I are selected from the compounds of the formulae CL-3-1 to CL-3-12:
- the medium according to the invention comprises the compound CL-3-1.
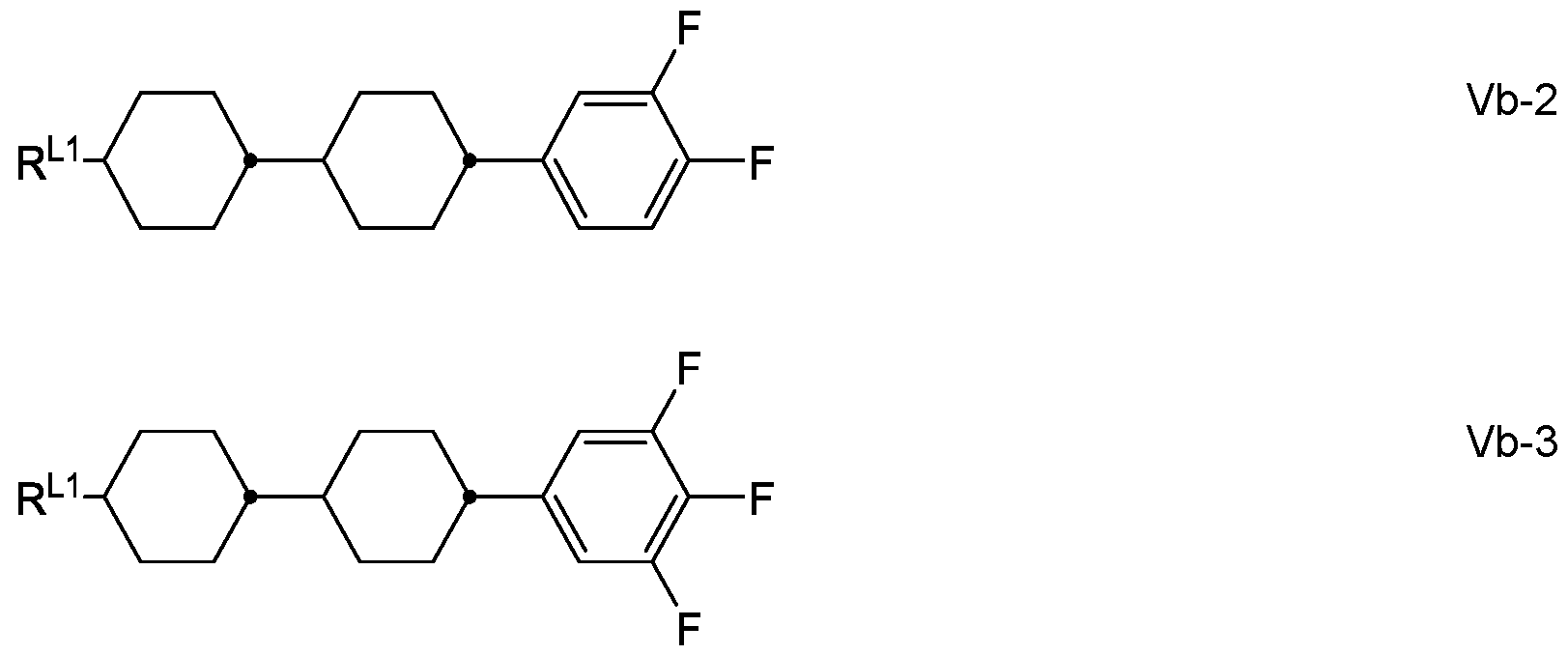
- the medium comprises one or more compounds of formula Vb selected from the following sub-formulae Vb-1 in which R L1 has the meaning given above for formula Va.
- the medium comprises the compound Vb-2, in which R L1 denotes alkyl or alkenyl having up to 7 C atoms, in particular vinyl.
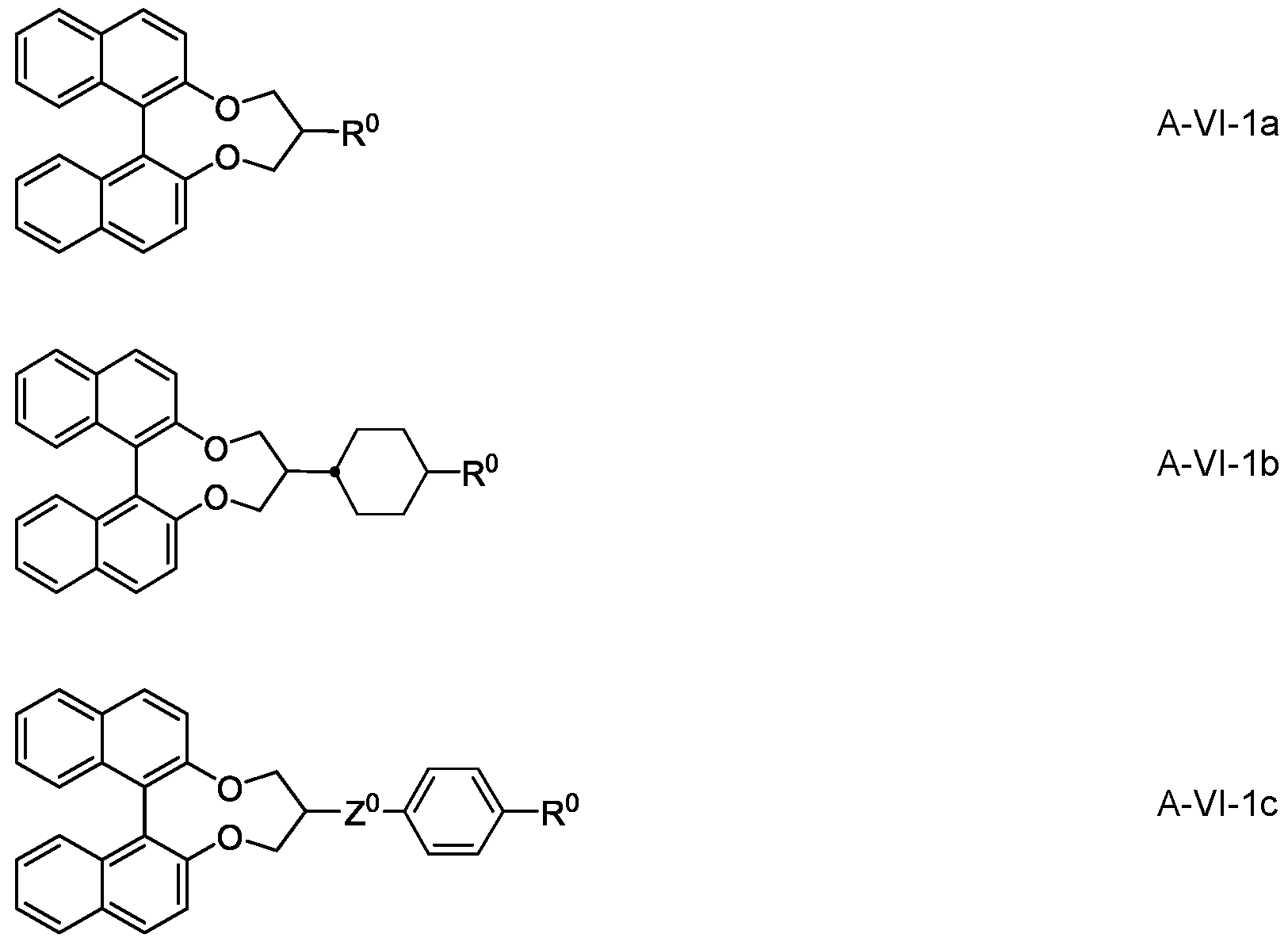
- the medium additionally comprises one or more compounds of the formulae VI-1 to VI-9
- R 7 each, independently of one another, have one of the meanings indicated for R 2A in Claim 5, and w and x each, independently of one another, denote 1 to 6.
- mixtures comprising at least one compound of the formula V-9.
- the medium additionally comprises one or more compounds of the formulae VII-1 to VII-21 ,
- the medium according to the invention preferably comprises the terphenyls of the formulae VII-1 to VII-21 in amounts of 2 to 30 % by weight, in particular 5 to 20 % by weight.
- R preferably denotes alkyl, furthermore alkoxy, each having 1 to 5 C atoms.
- R preferably denotes alkyl or alkenyl, in particular alkyl.
- R preferably denotes alkyl.
- the terphenyls are preferably employed in the mixtures according to the invention if the An value of the mixture is to be > 0.1.
- Preferred mixtures comprise 2 to 20 % by weight of one or more terphenyl compounds selected from the group of the compounds VII-1 to VII-21.
- Liquid-crystalline medium comprising at least one compound of the formulae
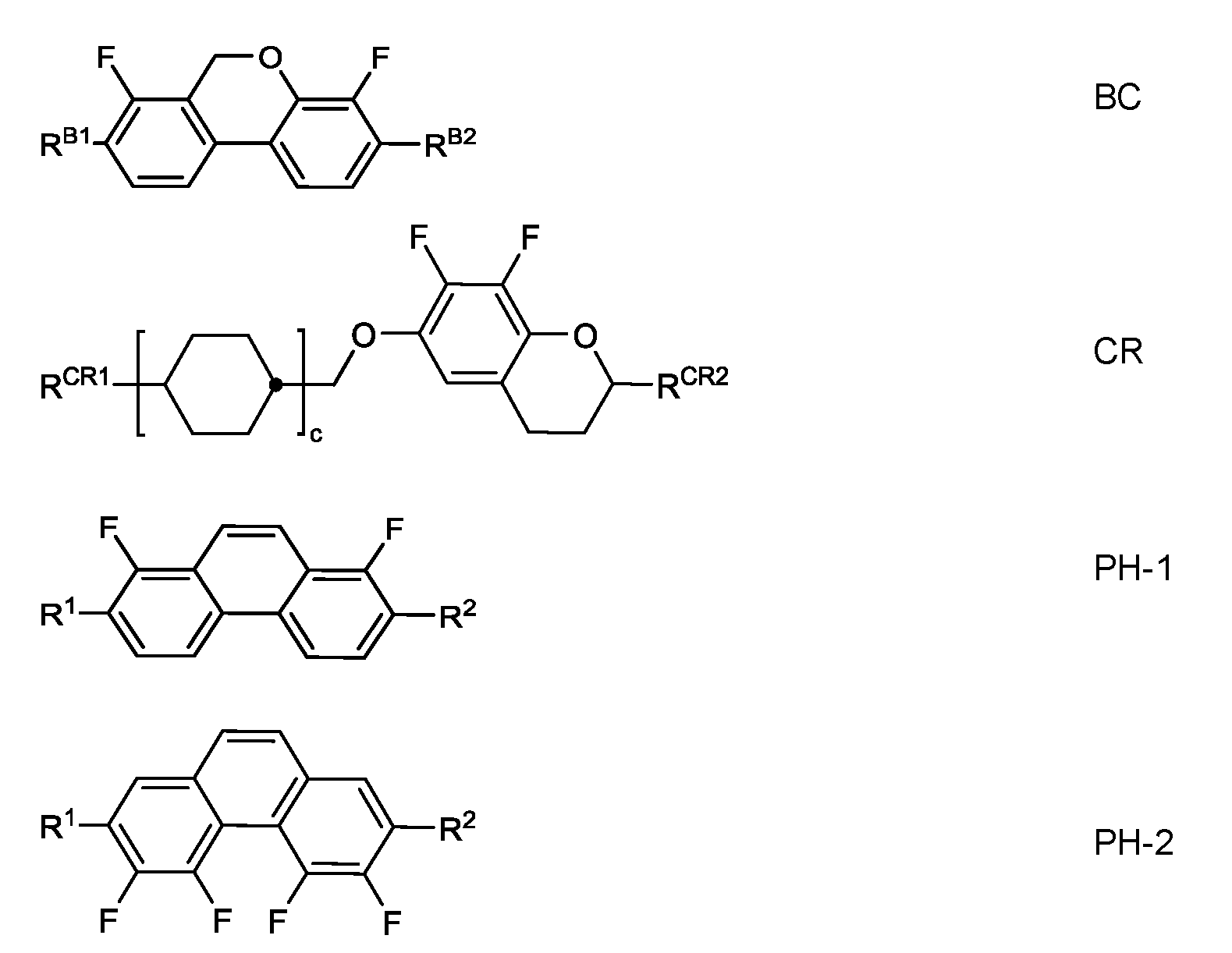
- R has the meanings indicated for R 1 of formula I, preferably denotes straight-chain alkyl or alkoxy having 1 to 7 C atoms or straight-chain alkenyl having 2 to 7 C atoms, (O) denotes an oxygen atom or a single bond, and alkyl denotes n-alkyl having 1 to 6 C atoms.
- Preferred mixtures comprise one or more compounds selected from the group of the difluorodibenzochroman compounds of the formula BC, chromans of the formula CR, fluorinated phenanthrenes of the formulae PH-1 and PH-2, fluorinated dibenzofurans of the formula BF-1 and BF-2, in which
- R B1 , R B2 , R CR1 , R CR2 , R 1 , R 2 each, independently of one another, have the meaning of R 2A .
- c is 0, 1 or 2 and d denotes 1 or 2.
- R 1 and R 2 preferably, independently of one another, denote alkyl or alkoxy having 1 to 6 C atoms.
- the mixtures according to the invention preferably comprise the compounds of the formulae BC, CR, PH-1 , PH-2 in amounts of 3 to 20 % by weight, in particular in amounts of 3 to 15 % by weight.
- Particularly preferred compounds of the formulae BC and CR are the compounds BC-1 to BC-7 and CR-1 to CR-5,
- alkyl and alkyl* each, independently of one another denote a straight-chain alkyl radical having 1 to 6 C atoms
- alkenyl and alkenyl* each, independently of one another denote a straight-chain alkenyl radical having 2 to 6 C atoms.
- mixtures comprising one, two or three compounds of the formula BC-2.
- Preferred mixtures comprise one or more indane compounds of the formula In, in which
- R 11 , R 12 , R 13 each, independently of one another, denote a straight-chain alkyl, alkoxy, alkoxyalkyl or alkenyl radical having 1 to 6 C atoms,
- R 12 and R 13 additionally denote halogen, preferably F, i denotes 0, 1 or 2.
- Preferred compounds of the formula In are the compounds of the formulae ln-1 to In-16 indicated below:
- the compounds of the formula In and the sub-formulae ln-1 to In-16 are preferably employed in the mixtures according to the invention in concentrations
- Preferred mixtures additionally comprise one or more compounds of the formulae L-1 to L-5, in which
- R and R 1 each, independently of one another, have the meanings indicated for R 2A in formula IIA above, and alkyl denotes an alkyl radical having 1 to 6 C atoms.
- the parameter s denotes 1 or 2.
- the compounds of the formulae L-1 to L-5 are preferably employed in concentrations of 5 to 50 % by weight, in particular 5 to 40 % by weight and very particularly preferably 10 to 40 % by weight.
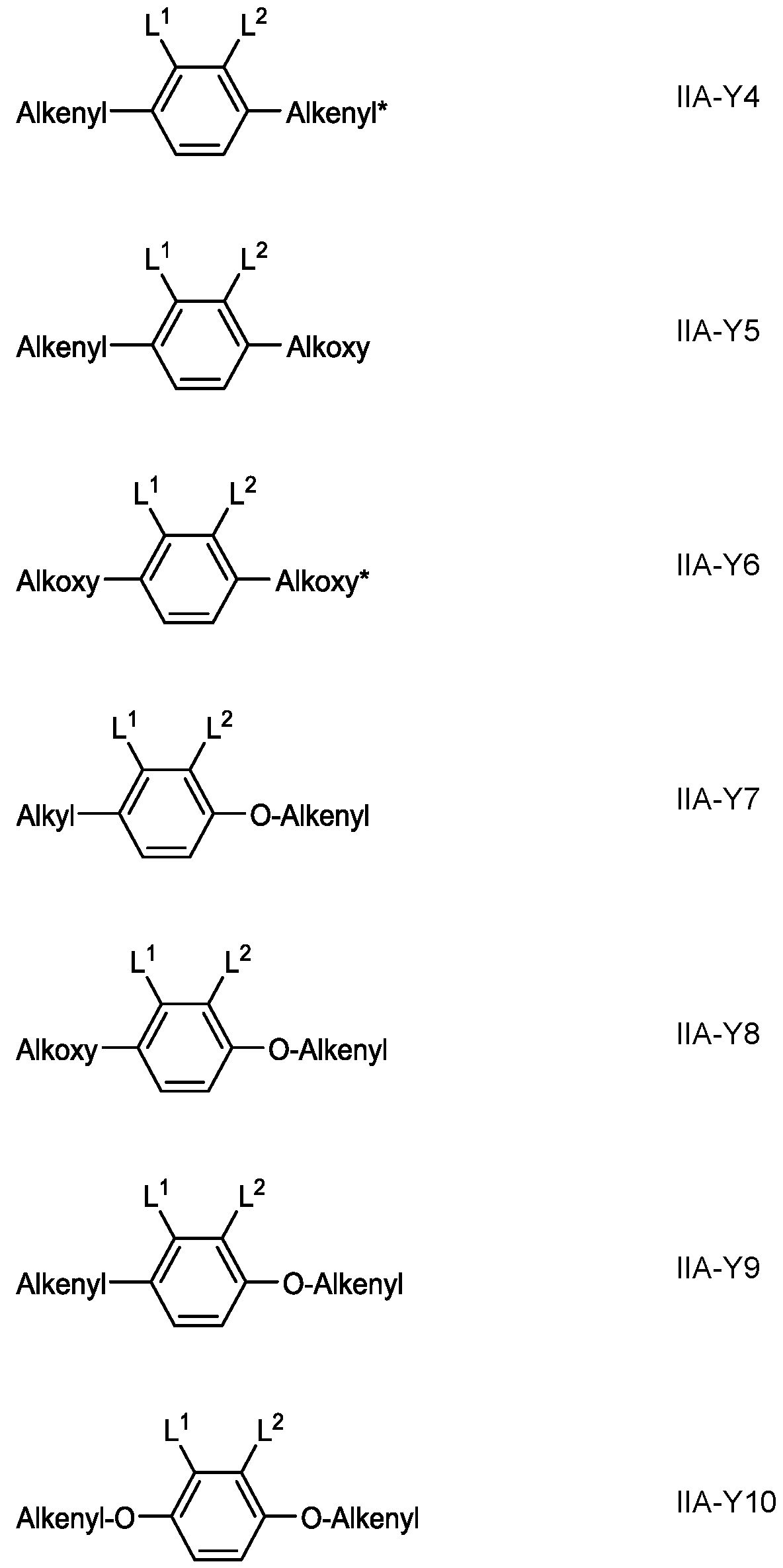
- Preferred mixtures additionally comprise one or more compounds of formula IIA-Y in which R 11 and R 12 have one of the meanings given in formula I above, and L 1 and L 2 , identically or differently, denote F or Cl.
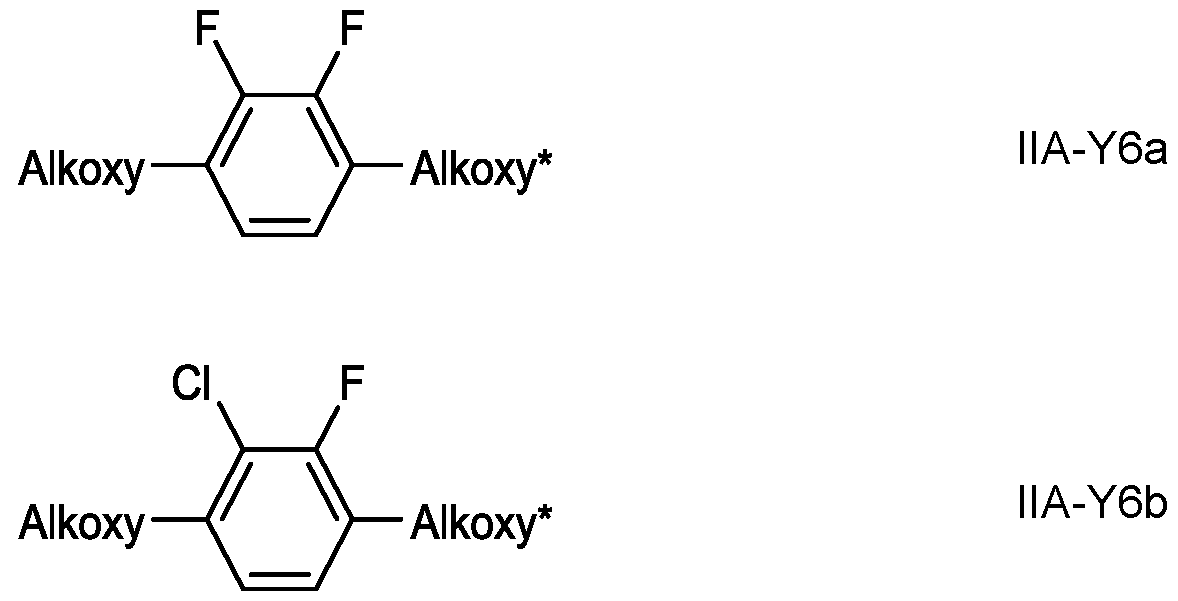
- Preferred compounds of the formula IIA-Y are selected from the group consisting of the following subformulae in which, Alkyl and Alkyl* each, independently of one another, denote a straight-chain alkyl radical having 1-6 C atoms, Alkoxy denotes a straight-chain alkoxy radical having 1-6 C atoms, Alkenyl and Alkenyl* each, independently of one another, denote a straight-chain alkenyl radical having 2-6 C atoms, and O denotes an oxygen atom or a single bond.
- Particularly preferred compounds of the formula IIA-Y are selected from the group consisting of following subformulae: in which Alkoxy and Alkoxy* have the meanings defined above and preferably denote methoxy, ethoxy, n- propyloxy, n-butyloxy or n-pentyloxy.
- liquid crystal medium according to the invention herein also referred to as liquid crystal host mixture, is suitable for the use in polymer stabilized displays.
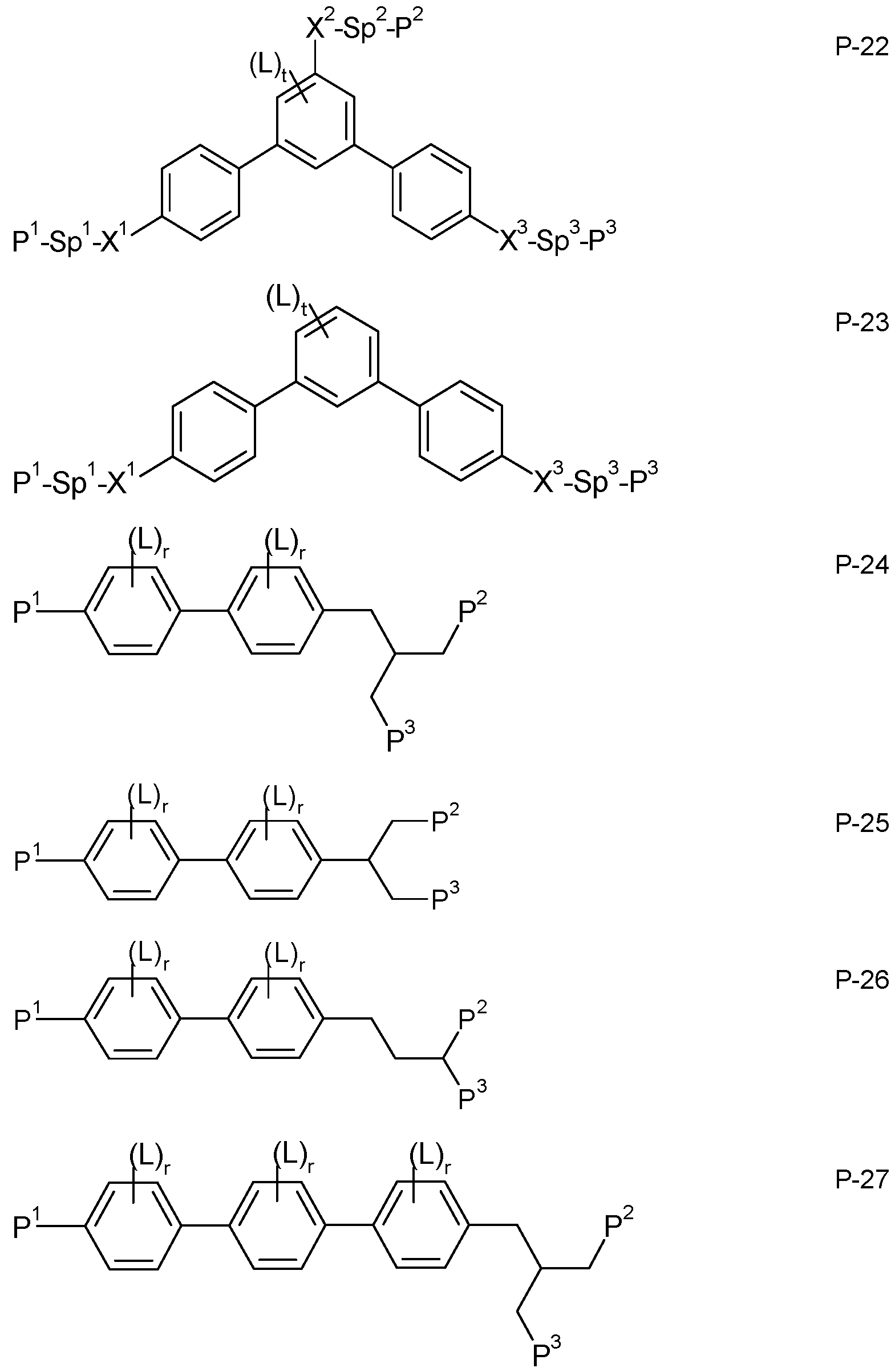
- the medium according to the invention optionally comprises one or more polymerizable compounds of formula P
- the terms “active layer” and “switchable layer” mean a layer in an electrooptical display, for example an LC display, that comprises one or more molecules having structural and optical anisotropy, like for example LC molecules, which change their orientation upon an external stimulus like an electric or magnetic field, resulting in a change of the transmission of the layer for polarized or unpolarized light.
- the terms “tilt” and “tilt angle” will be understood to mean a tilted alignment of the LC molecules of an LC medium relative to the surfaces of the cell in an LC display (here preferably a PSA display).
- the tilt angle here denotes the average angle ( ⁇ 90°) between the longitudinal molecular axes of the LC molecules (LC director) and the surface of the plane-parallel outer plates which form the LC cell.
- a low value for the tilt angle i.e. a large deviation from the 90° angle
- tilt angle values disclosed above and below relate to this measurement method.
- the terms “reactive mesogen” and “RM” will be understood to mean a compound containing a mesogenic or liquid crystalline skeleton, and one or more functional groups attached thereto which are suitable for polymerization and are also referred to as "polymerizable group” or "P".
- the term “polymerizable compound” as used herein will be understood to mean a polymerizable monomeric compound.
- low-molecular-weight compound will be understood to mean to a compound that is monomeric and/or is not prepared by a polymerization reaction, as opposed to a "polymeric compound” or a "polymer”.
- unpolymerizable compound will be understood to mean a compound that does not contain a functional group that is suitable for polymerization under the conditions usually applied for the polymerization of the RMs.
- mesogenic group as used herein is known to the person skilled in the art and described in the literature, and means a group which, due to the anisotropy of its attracting and repelling interactions, essentially contributes to causing a liquid-crystal (LC) phase in low-molecular-weight or polymeric substances.
- Compounds containing mesogenic groups do not necessarily have to have an LC phase themselves. It is also possible for mesogenic compounds to exhibit LC phase behaviour only after mixing with other compounds and/or after polymerization. Typical mesogenic groups are, for example, rigid rod- or disc-shaped units.
- optically active and “chiral” are synonyms for materials that are able to induce a helical pitch in a nematic host material, also referred to as “chiral dopants”.
- spacer group hereinafter also referred to as "Sp”, as used herein is known to the person skilled in the art and is described in the literature, see, for example, Pure Appl. Chem. 2001 , 73(5), 888 and C. Tschierske, G. Pelzl, S. Diele, Angew. Chem. 2004, 116, 6340-6368.
- spacer group or “spacer” mean a flexible group, for example an alkylene group, which connects the mesogenic group and the polymerizable group(s) in a polymerizable mesogenic compound.
- trans-1 ,4-cyclohexylene ring denotes a trans-1 ,4-cyclohexylene ring.
- the single bond shown between the two ring atoms can be attached to any free position of the benzene ring.
- organic group denotes a carbon or hydrocarbon group.
- Carbon group denotes a mono- or polyvalent organic group containing at least one carbon atom, where this either contains no further atoms (such as, for example, -C ⁇ C-) or optionally contains one or more further atoms, such as, for example, N, O, S, B, P, Si, Se, As, Te or Ge (for example carbonyl, etc.).
- hydrocarbon group denotes a carbon group which additionally contains one or more H atoms and optionally one or more heteroatoms, such as, for example, N, O, S, B, P, Si, Se, As, Te or Ge.
- Halogen denotes F, Cl, Br or I, preferably F or Cl.
- a carbon or hydrocarbon group can be a saturated or unsaturated group. Unsaturated groups are, for example, aryl, alkenyl or alkynyl groups.
- a carbon or hydrocarbon radical having more than 3 C atoms can be straight-chain, branched and/or cyclic and may also contain spiro links or condensed rings.
- carbon and hydrocarbon groups are C 1 -C 20 alkyl, C 2 -C 20 alkenyl, C 2 - C 20 alkynyl, C 3 -C 20 allyl, C 4 -C 20 alkyldienyl, C 4 -C 20 polyenyl, C 6 -C 20 cycloalkyl, C 4 -C 15 cycloalkenyl, C 6 -C 30 aryl, C 6 -C 30 alkylaryl, C 6 -C 30 arylalkyl, C 6 -C 30 alkylaryloxy, C 6 -C 30 arylalkyloxy, C 2 -C 30 heteroaryl, C 2 -C 30 heteroaryloxy.
- C 1 -C 12 alkyl C 2 -C 12 alkenyl, C 2 -C 12 alkynyl, C 6 -C 25 aryl and C 2 -C 25 heteroaryl.
- Preferred alkyl groups are, for example, methyl, ethyl, n-propyl, isopropyl, n-butyl, isobutyl, s-butyl, t-butyl, 2-methylbutyl, n-pentyl, s-pentyl, cyclopentyl, n-hexyl, cyclohexyl, 2-ethylhexyl, n-heptyl, cycloheptyl, n-octyl, cyclooctyl, n-nonyl, n-decyl, n-undecyl, n-dodecyl, dodecanyl, trifluoromethyl, perfluoro-n-butyl, 2,2,2-trifluoroethyl, perfluorooctyl, perfluorohexyl, etc.
- Preferred alkenyl groups are, for example, ethenyl, propenyl, butenyl, pentenyl, cyclopentenyl, hexenyl, cyclohexenyl, heptenyl, cycloheptenyl, octenyl, cyclooctenyl, etc.
- Preferred alkynyl groups are, for example, ethynyl, propynyl, butynyl, pentynyl, hexynyl, octynyl, etc.
- Aryl and heteroaryl groups can be monocyclic or polycyclic, i.e. they can contain one ring (such as, for example, phenyl) or two or more rings, which may also be fused (such as, for example, naphthyl) or covalently bonded (such as, for example, biphenyl), or contain a combination of fused and linked rings.
- Heteroaryl groups contain one or more heteroatoms, preferably selected from O, N, S and Se.
- Preferred aryl groups are, for example, phenyl, biphenyl, terphenyl, [1 ,1':3',1"]terphenyl-2'-yl, naphthyl, anthracene, binaphthyl, phenanthrene, 9,10- dihydro-phenanthrene, pyrene, dihydropyrene, chrysene, perylene, tetracene, pentacene, benzopyrene, fluorene, indene, indenofluorene, spirobifluorene, etc.
- Preferred heteroaryl groups are, for example, 5-membered rings, such as pyrrole, pyrazole, imidazole, 1 ,2,3-triazole, 1 ,2,4-triazole, tetrazole, furan, thiophene, selenophene, oxazole, isoxazole, 1,2-thiazole, 1 ,3-thiazole, 1,2,3-oxadiazole, 1 ,2,4-oxadiazole, 1,2,5-oxadiazole, 1 ,3,4-oxadiazole, 1 ,2,3-thiadiazole, 1,2,4- thiadiazole, 1,2,5-thiadiazole, 1 ,3,4-thiadiazole, 6-membered rings, such as pyridine, pyridazine, pyrimidine, pyrazine, 1 ,3,5-triazine, 1 ,2,4-triazine, 1 ,2,3-triazine, 1
- the (non-aromatic) alicyclic and heterocyclic groups encompass both saturated rings, i.e. those containing exclusively single bonds, and also partially unsaturated rings, i.e. those which may also contain multiple bonds.
- Heterocyclic rings contain one or more heteroatoms, preferably selected from Si, O, N, S and Se.
- Very preferred groups -A 1 -(Z-A 2 ) z - in formula P are selected from the following formulae wherein at least one benzene ring is substituted by at least one group L and the benzene rings are optionally further substituted by one or more groups L or P-Sp-.
- Preferred compounds of formula P and their subformulae are selected from the following preferred embodiments, including any combination thereof:
- the compounds contain exactly two polymerizable groups (represented by the groups P),
- the compounds contain exactly three polymerizable groups (represented by the groups P),
- - P is selected from the group consisting of acrylate, methacrylate and oxetane, very preferably acrylate or methacrylate,
- - Sp when being different from a single bond, is -(CH 2 ) P 2-, -(CH2) P 2-O-,
- - Sp is a single bond or denotes -(CH2) P 2-, -(CH2) P 2-O-, -(CH2) P 2-CO-O-, -(CH2) P 2-O- CO-, wherein p2 is 2, 3, 4, 5 or 6, and the O-atom or the CO-group, respectively, is connected to the benzene ring,
- - R does not denote or contain a polymerizable group and denotes straight chain, branched or cyclic alkyl having 1 to 25 C atoms, wherein one or more non-adjacent CH2-groups are optionally replaced by -O-,
- Suitable and preferred compounds of formula P are selected from the following formulae:
- P 1 , P 2 and P 3 each, independently of one another, denote an acrylate or methacrylate group
- Sp 1 , Sp 2 and Sp 3 each, independently of one another, denote a single bond or a spacer group having one of the meanings indicated above and below for Sp, and particularly preferably denote -(CH 2 ) p1 -, -(CH 2 ) p1 -O-, -(CH 2 ) p1 -CO-O-, -(CH 2 ) p1 -O-CO- or -(CH 2 ) p1 -O-CO-O-, in which p1 is an integer from 1 to 12, where, in addition, one or more of the radicals P 1 -Sp 1 -, P 2 -Sp 2 - and P 3 -Sp 3 - may denote R aa , with the proviso that at least one of the radicals
- P 1 -Sp 1 -, P 2 -Sp 2 - and P 3 -Sp 3 - present is different from R aa ,
- H atoms may be replaced by F, Cl, CN or P 1 -Sp 1 -, particularly preferably straight-chain or branched, optionally mono- or polyfluorinated alkyl, alkoxy, alkenyl, alkynyl, alkylcarbonyl, alkoxycarbonyl, alkylcarbonyloxy or alkoxycarbonyloxy having 1 to 12 C atoms (where the alkenyl and alkynyl radicals have at least two C atoms and the branched radicals have at least three C atoms),
- R 0 , R 00 each, independently of one another and identically or differently on each occurrence, denote H or alkyl having 1 to 12 C atoms,
- R y and R z each, independently of one another, denote H, F, CH3 or CF3,
- X 1 , X 2 and X 3 each, independently of one another, denote -CO-O-, -O-CO- or a single bond,
- L on each occurrence denotes F, Cl, CN or straight-chain or branched, optionally mono- or polyfluorinated alkyl, alkoxy, alkenyl, alkynyl, alkylcarbonyl, alkoxycarbonyl, alkylcarbonyloxy or alkoxycarbonyloxy having 1 to 12 C atoms, preferably F,
- L' and L" each, independently of one another, denote H, F or Cl, k denotes 0 or 1 , r denotes 0, 1 , 2, 3 or 4, s denotes 0, 1 , 2 or 3, t denotes 0, 1 or 2, x denotes 0 or 1.
- L on each occurrence identically or differently, has one of the meanings given above or below, and is preferably F, Cl, CN, NO 2 , CH 3 , C 2 H 5 , C(CH 3 ) 3 , CH(CH 3 ) 2 , CH 2 CH(CH 3 )C 2 H 5 , OCH 3 , OC 2 H 5 , COCH 3 , COC 2 H 5 , COOCH 3 , COOC 2 H 5 , CF 3 , OCF 3 , OCHF 2 , OC 2 F 5 or P-Sp-, very preferably F, Cl, CN, CH 3 , C 2 H 5 , OCH 3 , COCH 3 , OCF 3 or P-Sp-, more preferably F, Cl, CH 3 , OCH 3 , COCH 3 oder OCF 3 , especially F or CH 3 .
- the temperature dependence of the HTP of the individual compounds may be high or low.
- the temperature dependence of the pitch of the medium can be compensated by mixing compounds having different temperature dependencies of the HTP in corresponding ratios.
- optically active component a multitude of chiral dopants, some of which are commercially available, is available to the person skilled in the art, such as, for example, cholesteryl nonanoate, R- and S-811, R- and S-1011 , R- and S-2011, R- and S-3011, R- and S-4011, or CB15 (all Merck KGaA, Darmstadt).
- Particularly suitable dopants are compounds which contain one or more chiral groups and one or more mesogenic groups, or one or more aromatic or alicyclic groups which form a mesogenic group with the chiral group.
- Preferred chiral groups are sugar derivatives, such as glucose, mannose, galactose, fructose, arabinose and dextrose; sugar alcohols, such as, for example, sorbitol, mannitol, iditol, galactitol or anhydro derivatives thereof, in particular dianhydrohexitols, such as dianhydrosorbide (1 ,4:3,6-dianhydro-D-sorbide, isosorbide), dianhydromannitol (isosorbitol) or dianhydroiditol (isoiditol); sugar acids, such as, for example, gluconic acid, gulonic acid and ketogulonic acid; chiral substituted glycol radicals, such as, for example, mono- or oligoethylene or propylene glycols, in which one or more CH2 groups are substituted by alkyl or alkoxy; amino acids, such as, for example, alanine, valine, pheny
- the media according to the present invention preferably comprise chiral dopants which are selected from the group of known chiral dopants. Suitable chiral groups and mesogenic chiral compounds are described, for example, in DE 3425 503, DE 35 34 777, DE 35 34 778, DE 35 34 779 and DE 35 34 780, DE 4342 280, EP 01 038 941 and DE 19541 820. Examples are also compounds listed in Table F below.
- Chiral compounds preferably used according to the present invention are selected from the group consisting of the formulae shown below.
- chiral dopants selected from the group consisting of compounds of the following formulae A-l to A-lll and A-Ch: in which
- R z denotes H, CH 3 , F, Cl, or CN, preferably H or F
- R 8 has one of the meanings of R a11 given above, preferably alkyl, more preferably n-alkyl having 1 to 15 C atoms,
- Z 8 denotes- C(O)O-, CH2O, CF2O or a single bond, preferably -C(O)O-,
- a 11 is defined as A 12 below, or alternatively denotes in which
- L 12 on each occurrence denotes halogen, CN, or alkyl, alkenyl, alkoxy or alkenyloxy having up to 12 C atoms and in which one or more H atoms are optionally replaced with halogen, preferably methyl, ethyl, Cl or F, particularly preferably F,
- a 22 has the meanings given for A 12
- a 31 has the meanings given for A 11 , or alternatively denotes
- a 32 has the meanings given for A 12 .
- n3 is 1 , 2 or 3
- r is 0, 1 , 2, 3 or 4.
- dopants selected from the group consisting of the compounds of the following formulae: in which m is, on each occurrence, identically or differently, an integer from 1 to 9 and n is, on each occurrence, identically or differently, an integer from 2 to 9.
- Particularly preferred compounds of formula A are compounds of formula A-lll.
- dopants are derivatives of the isosorbide, isomannitol or isoiditol of the following formula A-IV:
- dianhydroiditol preferably dianhydrosorbitol
- chiral ethane diols such as, for example, diphenylethanediol (hydrobenzoin), in particular mesogenic hydrobenzoin derivatives of the following formula A-V: including the (S,S) enantiomers, which are not shown, in which are each, independently of one another, 1 ,4-phenylene, which may also be mono-, di- or trisubstituted by L, or 1,4-cyclo- hexylene,
- L is H, F, Cl, CN or optionally halogenated alkyl, alkoxy, alkylcarbonyl, alkoxycarbonyl or alkoxycarbonyloxy having 1-7 carbon atoms, c is O or 1,
- X is CH 2 or -C(O)-
- Z 0 is -COO-, -OCO-, -CH2CH2- or a single bond
- R 0 is alkyl, alkoxy, alkylcarbonyl, alkoxycarbonyl or alkylcarbonyloxy having
- the compounds of the formula A-IV are described in WO 98/00428.
- the compounds of the formula A-V are described in GB-A-2,328,207.
- Very particularly preferred dopants are chiral binaphthyl derivatives, as described in WO 02/94805, chiral binaphthol acetal derivatives, as described in WO 02/34739, chiral TADDOL derivatives, as described in WO 02/06265, and chiral dopants having at least one fluorinated bridging group and a terminal or central chiral group, as described in WO 02/06196 and WO 02/06195.
- B 1 and B 2 are each, independently of one another, an aromatic or partially or fully saturated aliphatic six-membered ring in which one or more CH groups may each be replaced by N and one or more non-adjacent CH2 groups may each be replaced by O or S,
- W 1 and W2 are each, independently of one another, -Z 1 -A 1 -(Z 2 -A 2 ) m -R, and one of the two is alternatively R 1 or A 3 , but both are not simultaneously H, or
- II 1 and II 2 are each, independently of one another, CH2, O, S, CO or CS,
- V 1 and V 2 are each, independently of one another, (CH2)n, in which from one to four non-adjacent CH2 groups may each be replaced by O or S, and one of V 1 and V 2 and, in the case where both are a single bond, n is 1 ,2 or 3
- Z 1 and Z 2 are each, independently of one another, -O-, -S-, -CO-, -COO-, -OCO-, -O-COO-, -CO-NR X -, -NR X - CO-, -O-CH2-, -CH2-O-,
- R x denotes alkyl having 1 to 6 C atoms
- a 1 , A 2 and A 3 are each, independently of one another, 1 ,4-phenylene, in which one or two non-adjacent CH groups may each be replaced by N, 1,4- cyclohexylene, in which one or two non-adjacent CH2 groups may each be replaced by O or S, 1,3-dioxolane-4,5-diyl, 1 ,4-cyclohexenylene, 1 ,4- bicyclo[2.2.2]octylene, piperidine- 1 ,4-diyl, naphthalene-2,6-diyl, decahydronaphthalene-2,6-diyl or 1 ,2,3,4-tetrahydronaphthalene-2,6-diyl, where each of these groups may be monosubstituted or polysubstituted by L, and in addition A 1 can be a single bond,
- L is a halogen atom, preferably F, CN, NO2, alkyl, alkoxy, alkylcarbonyl, alkoxycarbonyl or alkoxycarbonyloxy having 1-7 carbon atoms, in which one or more H atoms may each be replaced by F or Cl, m is in each case, independently, 0, 1, 2 or 3, and
- R 0 as defined for formula A-IV or H or alkyl having from 1 to 4 carbon atoms, Z 0 is, in particular, -OC(O)- or a single bond.
- the concentration of the one or more chiral dopant(s), in the LC medium is preferably in the range from 0.001 % to 20 %, preferably from 0.05 % to 5 %, more preferably from 0.1 % to 2 %, and, most preferably from 0.5 % to 1.5 %.
- concentration ranges apply in particular to the chiral dopant S-4011 or R-4011 (both from Merck KGaA) and for chiral dopants having the same or a similar HTP.
- S-4011 or R-4011 both from Merck KGaA
- these preferred concentrations have to be decreased, respectively increased proportionally according to the ratio of their HTP values relatively to that of S-4011.
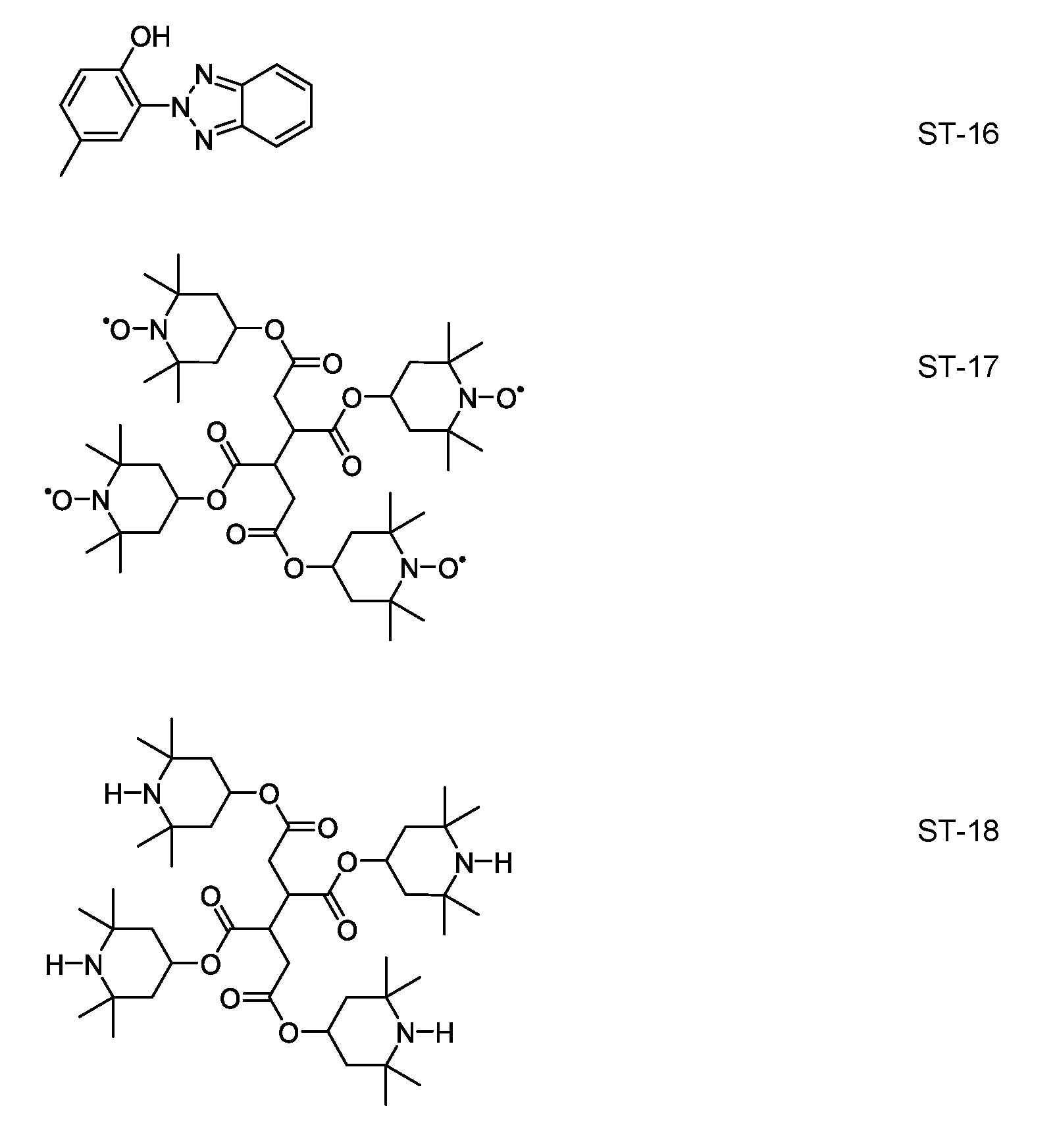
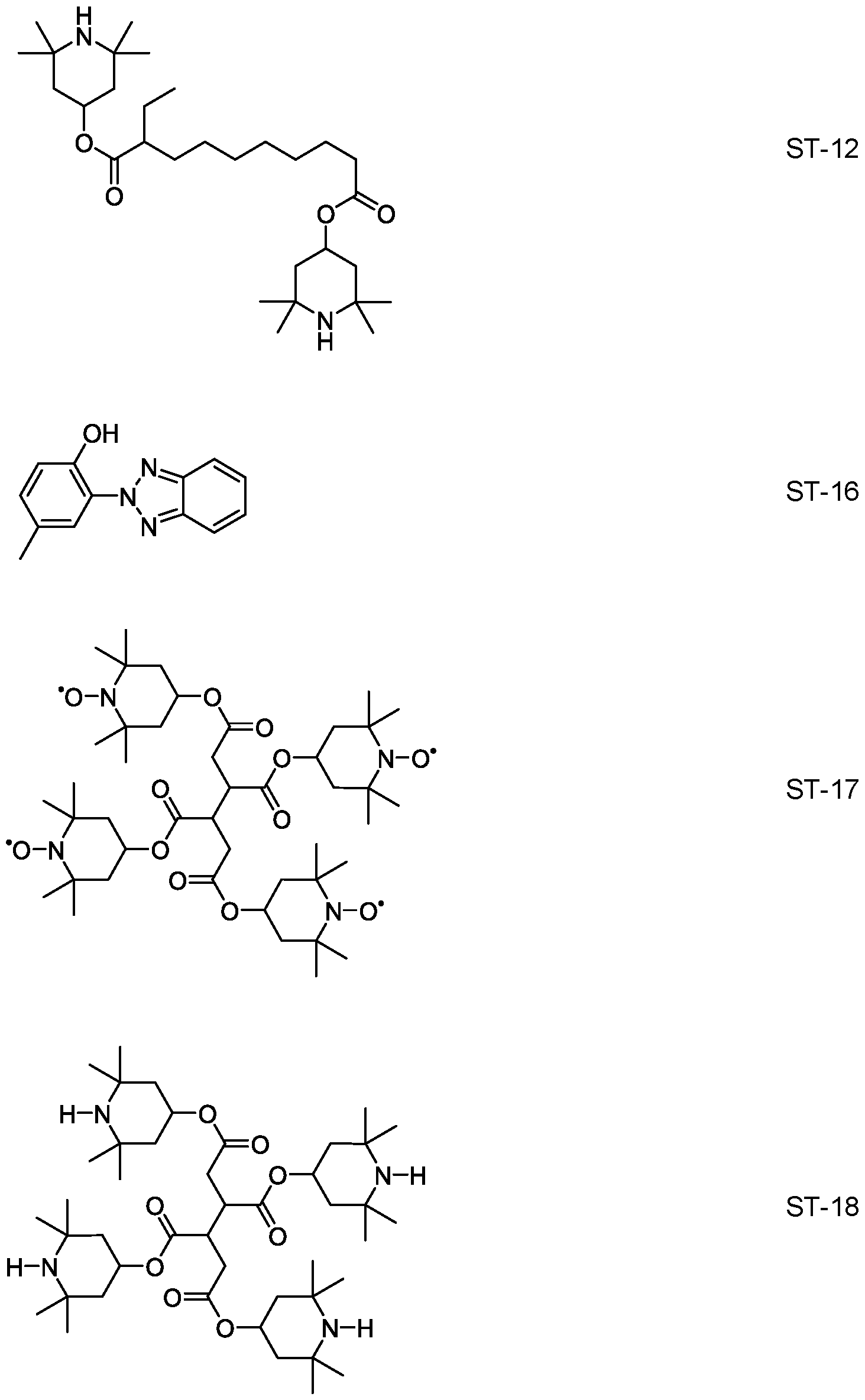
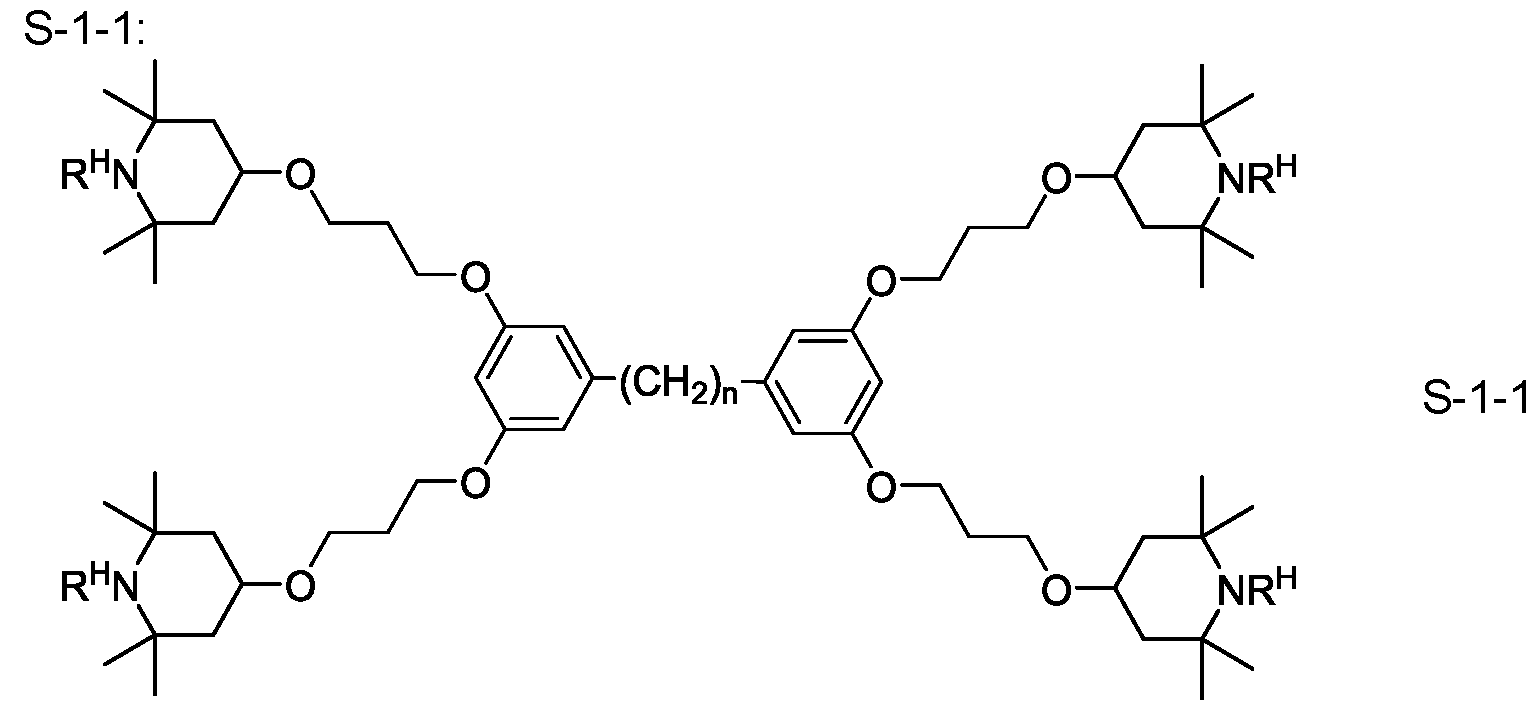
- the media according to the invention comprise a stabilizer selected from the group of compounds of the formulae ST-1 to ST- 18:
- R ST denotes H, an alkyl or alkoxy radical having 1 to 15 C atoms, where, in addition, one or more CH2 groups in these radicals may each be replaced, independently
- L 1 and L 2 each, independently of one another, denote F, Cl, CF3 or CHF2, p denotes 1 or 2, and q denotes 1 , 2, 3, 4, 5, 6, 7, 8, 9 or 10.
- the medium according to the invention preferably comprises one or more compounds of the formula ST-3.
- n preferably denotes 3.
- n preferably denotes 7.
- Very particularly preferred mixtures according to the invention comprise one or more stabilizers from the group of the compounds of the formulae ST-2a-1 , ST-3a-1 , ST-
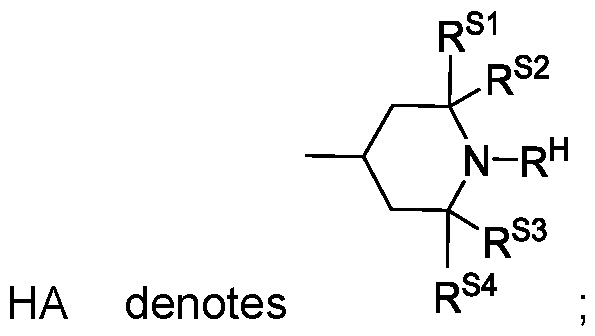
- the medium comprises one or more compounds of the formula S in which
- Ar denotes a methylene group or an aromatic hydrocarbon group having 6 to 40
- Sp denotes a spacer group
- R s denotes H, alkyl having 1 to 12 C atoms or alkenyl having 2 to 12 C atoms;
- Z s denotes -O-, -C(O)O-, -(CH2) Z - or— (CH2) Z O-, or a single bond;
- R H denotes H, O’, CH 3 , OH or OR S ;
- R S1 , R S2 , R S3 and R S4 identically or differently, denote alkyl having 1 to 6 C atoms, preferably having 1 to 3 C atoms, very preferably CH 3 ;
- G denotes H or R s or a group Z S -HA; z is an integer from 1 to 6, and q is 2, 3 or 4, preferably 3 or 4.
- Preferred aryl groups are benzene, naphthalene, anthracene, biphenyl, m-terphenyl, p-terphenyl, and (phenylalkyl)benzene in which alkyl is straight chain alkyl having 1 to 12 C atoms.
- the medium according to the invention comprises a compound of the formula S in which the parameter q is 3 and G denotes a group Z S -HA.
- the medium according to the invention comprises a compound of the formula S in which the parameter q is 4 and G denotes H or R S .
- the compounds of formula S are preferably selected from the compounds of the formulae S-1 S-2 and S-3: in which R H has the meanings given above and preferably denotes H or O, Sp on each occurrence, identically or differently, denotes a spacer group, and W denotes linear or branched, optionally unsaturated alkylene having 1 to 12 C atoms, in which one or more non-adjacent -CH 2 - groups may each be replaced with - O-.
- Preferred compounds of formula S-1 are selected from the compounds of the formula in which R H has the meanings given above and preferably denotes H or O, and n is an integer from 0 to 12, preferably 5, 6, 7, 8 or 9, very preferably 7.
- the medium comprises a compound of the formula S-1-1a
- Preferred compounds of formula S-2 are selected from the compounds of the formula in which R H has the meanings given above and preferably denotes H or O, and n2, on each occurrence identically or differently, preferably identically, is an integer from 1 to 12, preferably 2, 3, 4, 5, or 6, very preferably 3, and R s on each occurrence identically or differently, preferably identically, denotes alkyl having 1 to 6 C atoms, preferably n-butyl.
- the medium comprises a compound of the formula S-2-1a
- the total proportion of the compounds of the formulae ST-1 to ST-18, based on the mixture according to the invention, should not exceed 2%.
- CY-n-Om in particular CY-3-04, CY-5-04 and/or CY-3- 02, preferably in a total concentration in the range of from 0.5% to 10%, preferably 1% to 8%, very preferably 2 to 5%; and/or
- CLY-n-Om preferably CLY-2-04, CLY-3-02 and/or CLY-3-03, preferably in concentrations > 3 %, very preferably in total 5 to 20 %, very preferably 7 to 15% and/or
- the expression "have a nematic phase” here means on the one hand that no smectic phase and no crystallisation are observed at low temperatures at the corresponding temperature and on the other hand that clearing still does not occur on heating from the nematic phase.
- the investigation at low temperatures is carried out in a flow viscometer at the corresponding temperature and checked by storage in test cells having a layer thickness corresponding to the electro-optical use for at least 100 hours. If the storage stability at a temperature of -20°C in a corresponding test cell is 1000 h or more, the medium is referred to as stable at this temperature. At tempera- tures of -30°C and -40°C, the corresponding times are 500 h and 250 h respectively. At high temperatures, the clearing point is measured by conventional methods in capillaries.
- the medium has a birefringence in the range of from 0.075 to 0.110, preferably from 0.080 to 0.108, more preferably from 0.085 to 0.105, in particular from 0.088 to 0.100.
- the medium according to the invention has a value ⁇ 1 /K 1 of 8.0 or 7.9 or 7.8 or 7.7 or less, preferably 7.6 or less.
- liquid-crystal media according to the invention have high values for the voltage holding ratio in liquid-crystal cells.
- the proportion of component A is preferably between 45 and 100 %, in particular between 60 and 85 %.
- one (or more) individual compound(s) which has (have) a value of ⁇ ⁇ -0.8 is (are) preferably selected. This value must be more negative, the smaller the proportion A in the mixture as a whole.
- Particularly preferred individual compounds in component B are extremely low- viscosity nematic liquid crystals having a flow viscosity of not greater than 18 mm 2 • s' 1 , preferably not greater than 12 mm 2 • s -1 , at 20°C.
- Q denotes halogen, preferably chlorine, or -CN
- R 20 and R 21 each denote alkyl, alkenyl, alkoxy, alkoxyalkyl or alkoxycarbonyloxy having up to 18, preferably up to 8, carbon atoms, or one of these radicals alternatively denotes CN, NC, NO 2 , NCS, CF 3 , SF 5 , OCF 3 , F, Cl or Br.
- the VA, IPS or FFS mixture according to the invention may also comprise compounds in which, for example, H, N, O, Cl and F have been replaced by the corresponding isotopes.
- the compounds of formula P are optionally added to the mixtures according to the invention in concentrations of preferably 0.01 to 5 % by weight, particularly preferably 0.2 to 2 % by weight, based on the mixture.
- These mixtures may optionally also comprise an initiator, as described, for example, in U.S. 6,781,665.
- the initiator for example lrganox-1076 from BASF, is preferably added to the mixture comprising polymerizable compounds in amounts of 0 to 1 %.
- PS-VA polymer-stabilized VA modes
- PSA polymer sustained VA
- the liquid-crystalline compounds of the LC host do not react under the polymerization conditions of the reactive mesogens, i.e. generally on exposure to UV in the wavelength range from 320 to 360 nm.
- Liquid-crystalline compounds containing an alkenyl side chain, such as, for example, CC-3-V exhibit no reaction under the polymerization conditions (UV polymerization) for the RMs, hence, herein, such compounds are not to be considered as RMs.
- the compounds according to the present invention can be synthesized by or in analogy to known methods described in the literature (for example in the standard works such as Houben-Weyl, Methoden der Organischen Chemie [Methods of Organic Chemistry], Georg-Thieme-Verlag, Stuttgart), under reaction conditions which are known and suitable for said reactions. Use may also be made here of variants which are known per se, but are not mentioned here. In particular, they can be prepared as described in or in analogy to the following reaction schemes. Further methods for preparing the inventive compounds can be taken from the examples.
- n, m and I are independently of each other 1 , 2, 3, 4, 5, 6, or 7.
- Table A shows the codes for the ring elements of the nuclei of the compound
- Table B lists the bridging units
- Table C lists the meanings of the symbols for the left- and right- hand end groups of the molecules.
- the acronyms are composed of the codes for the ring elements with optional linking groups, followed by a first hyphen and the codes for the left-hand end group, and a second hyphen and the codes for the right-hand end group.
- Table D shows illustrative structures of compounds together with their respective abbreviations.
- Table C End groups in which n and m are each integers, and the three dots are placeholders for other abbreviations from this table.
- the mixtures according to the invention preferably comprise one or more compounds of the compounds mentioned below.
- n, m, k and I are, independently of one another, each an integer, preferably 1 to 9 preferably 1 to 7, k and I possibly may be also 0 and preferably are 0 to 4, more preferably 0 or 2 and most preferably 2, n preferably is 1 , 2, 3, 4 or 5, in the combination “-nO-” it preferably is 1 , 2, 3 or 4, preferably 2 or 4, m preferably is 1 , 2, 3, 4 or 5, in the combination “-Om” it preferably is 1 , 2, 3 or 4, more preferably 2 or 4.
- the combination “-IVm” preferably is “2V1”.
- Table E shows chiral dopants which can be added to the LC media according to the invention.
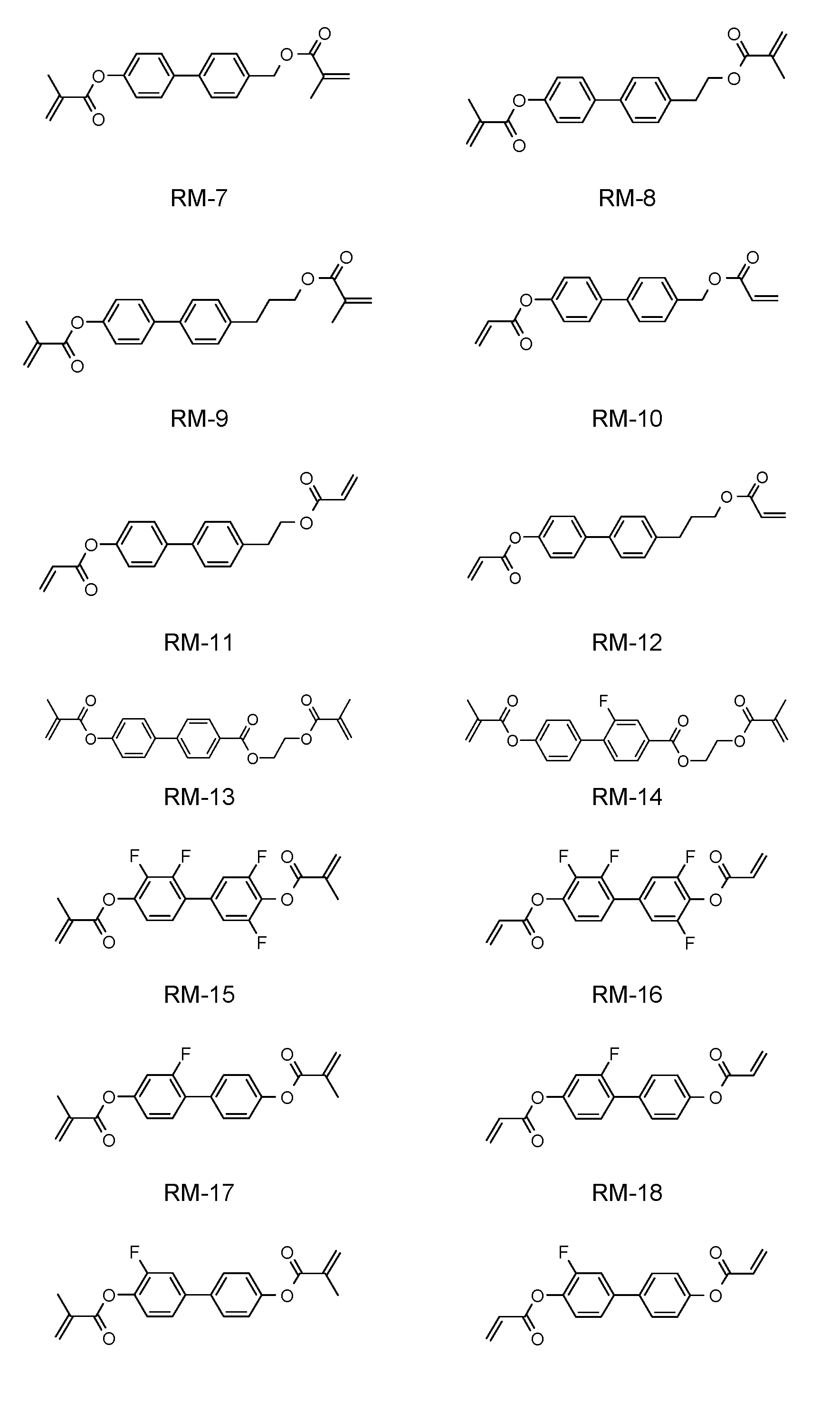
- Table F shows preferred reactive mesogenic compounds which can be used in the LC media in accordance with the present invention.
- the mixtures according to the invention comprise one or more polymerizable compounds, preferably selected from the polymerizable compounds of the formulae RM-1 to RM-182.
- m.p. denotes the melting point and C denotes the clearing point of a liquid- crystalline substance in degrees Celsius; boiling temperatures are denoted by m.p.
- C denotes crystalline solid state
- S denotes smectic phase (the index denotes the phase type)
- N denotes nematic state
- Ch denotes cholesteric phase
- I denotes isotropic phase
- T g denotes glass-transition temperature. The number between two symbols indicates the conversion temperature in degrees Celsius.
- the host mixture used for determination of the optical anisotropy An of single com- pounds is the commercial mixture ZLI-4792 (Merck KGaA).
- the dielectric anisotropy ⁇ is determined using commercial mixture ZLI-2857.
- the physical data of the compound to be investigated are obtained from the change in the dielectric constants of the host mixture after addition of the compound to be investigated and extrapolation to 100 % of the compound employed. In general, 10 % of the compound to be investigated are dissolved in the host mixture, depending on the solubility.
- parts or per cent data denote parts by weight or per cent by weight.
- V o denotes threshold voltage
- capacitive [V] at 20°C n e denotes extraordinary refractive index at 20°C and 589 nm
- n 0 denotes ordinary refractive index at 20°C and 589 nm
- ⁇ n denotes optical anisotropy at 20°C and 589 nm, denotes dielectric permittivity perpendicular to the director at 20°C and
- K2 denotes elastic constant, "twist" deformation at 20°C [pN],
- K3 denotes elastic constant, "bend” deformation at 20°C [pN],
- LTS denotes low-temperature stability (nematic phase), determined in test cells or in the bulk, as specified.
- temperatures such as, for example, the melting point T(C,N), the transition from the smectic (S) to the nematic (N) phase T(S,N) and the clearing point T(N , I) or cl.p., are indicated in degrees Celsius (°C).
- M.p. denotes melting point .
- Tg glass state
- C crystalline state
- N nematic phase
- S smectic phase
- I isotropic phase.
- the numbers between these symbols represent the transition tem- peratures.
- threshold voltage for the present invention relates to the capacitive threshold (Vo), also called the Freedericksz threshold, unless explicitly indicated otherwise.
- the optical threshold can also be indicated for 10 % relative contrast (V10).
- the display used for measurement of the capacitive threshold voltage consists of two plane-parallel glass outer plates at a separation of 20 ⁇ m, which each have on the insides an electrode layer and an unrubbed polyimide alignment layer on top, which cause a homeotropic edge alignment of the liquid-crystal molecules.
- the display or test cell used for measurement of the tilt angle consists of two plane- parallel glass outer plates at a separation of 4 ⁇ m, which each have on the insides an electrode layer and a polyimide alignment layer on top, where the two polyimide layers are rubbed antiparallel to one another and cause a homeotropic edge alignment of the liquid-crystal molecules.
- VHR is determined at 20°C (VHR20) and after
- VHR100 5 minutes in an oven at 100°C (VHR100) in a commercially available instrument Model LCM-1 (00004) from TOYO Corporation, Japan.
- the voltage used has a frequency of in a range from 1 Hz to 60 Hz, unless indicated more precisely.
- the stability to UV irradiation is investigated in a "Suntest CPS+", a commercial instrument from Heraeus, Germany, using a Xenon lamp NXE1500B.
- the sealed test cells are irradiated for 2.0 h, unless explicitly indicated, without additional heating.
- the irradiation power in the wavelength range from 300 nm to 800 nm is 765 W/ m 2 V.
- a UV "cut-off" filter having an edge wavelength of 310 nm is used in order to simulate the so-called window glass mode.
- at least four test cells are investigated for each condition, and the respective results are indicated as averages of the corresponding individual measurements.
- AVHR voltage holding ratio
- LTS low-temperature stability
- LC mixture in the bulk against spontaneous crystallisation of individual components at low temperatures or the occurrence of smectic phases, as the case may be
- several sealed bottles each containing about 1 g of the material, are stored at one or more given temperatures, typically of -10°C, -20°C, -30°C and/or -40°C and it is inspected at regular intervals visually, whether a phase transition is observed or not.
- the first one of the samples at a given temperature shows a change time is noted.
- the time until the last inspection, at which no change has been observed is noted as the respective LTS.
- the ion density from which the resistivity is calculated is measured using the commercially available LC Material Characteristics Measurement System Model 6254 from Toyo Corporation, Japan, using VHR test cells with AL16301 Polyimide (JSR Corp., Japan) having a 3.2 ⁇ m cell gap. The measurement is performed after 5 min of storage in an oven at 60 °C or 100 °C.
- HTP denotes the helical twisting power of an optically active or chiral substance in an LC medium (in ⁇ m). Unless indicated otherwise, the HTP is measured in the commercially available nematic LC host mixture MLD-6260 (Merck KGaA) at a temperature of 20°C.
- the Clearing point is measured using the Mettler Thermosystem FP900.
- the optical anisotropy (An) is measured using an Abbe Refractometer H005 (Natrium-spectral lamp Na10 at 589nm, 20 °C).
- the dielectric anisotropy ( ⁇ ) is measured using an LCR-Meter E4980A/Agilent (G005) at 20°C (s-parallel-cells with JALS 2096-R1).
- the turn on voltage (Vo) is measured using an LCR-Meter E4980A/Agilent (G005) at 20°C (s-parallel-cells with JALS 2096-R1).
- the rotational viscosity ( ⁇ 1 ) is measured using a TOYO LCM-2 (0002) at 20°C (gamma 1 negative cells with JALS-2096-R 1 ).
- the elastic constant (K 1 , splay) is measured using an LCR-Meter E4980A/Agilent (G005) at 20°C (s parallel-cells with JALS 2096-R1).
- K 3 The elastic constant (K3, bend) is measured using an LCR-Meter E4980A/Agilent (G005) at 20°C (s-parallel-cells with JALS 2096-R1).
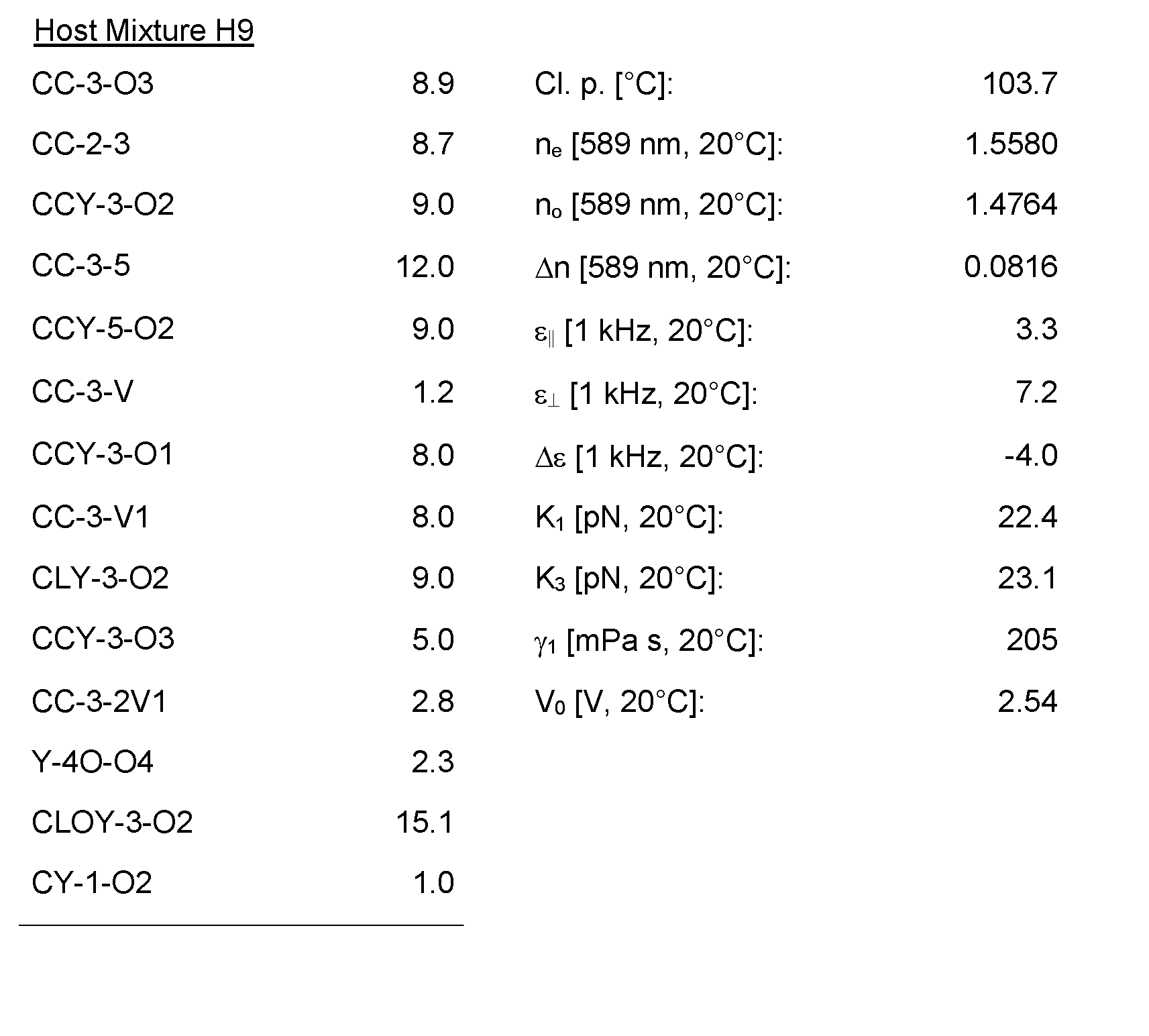
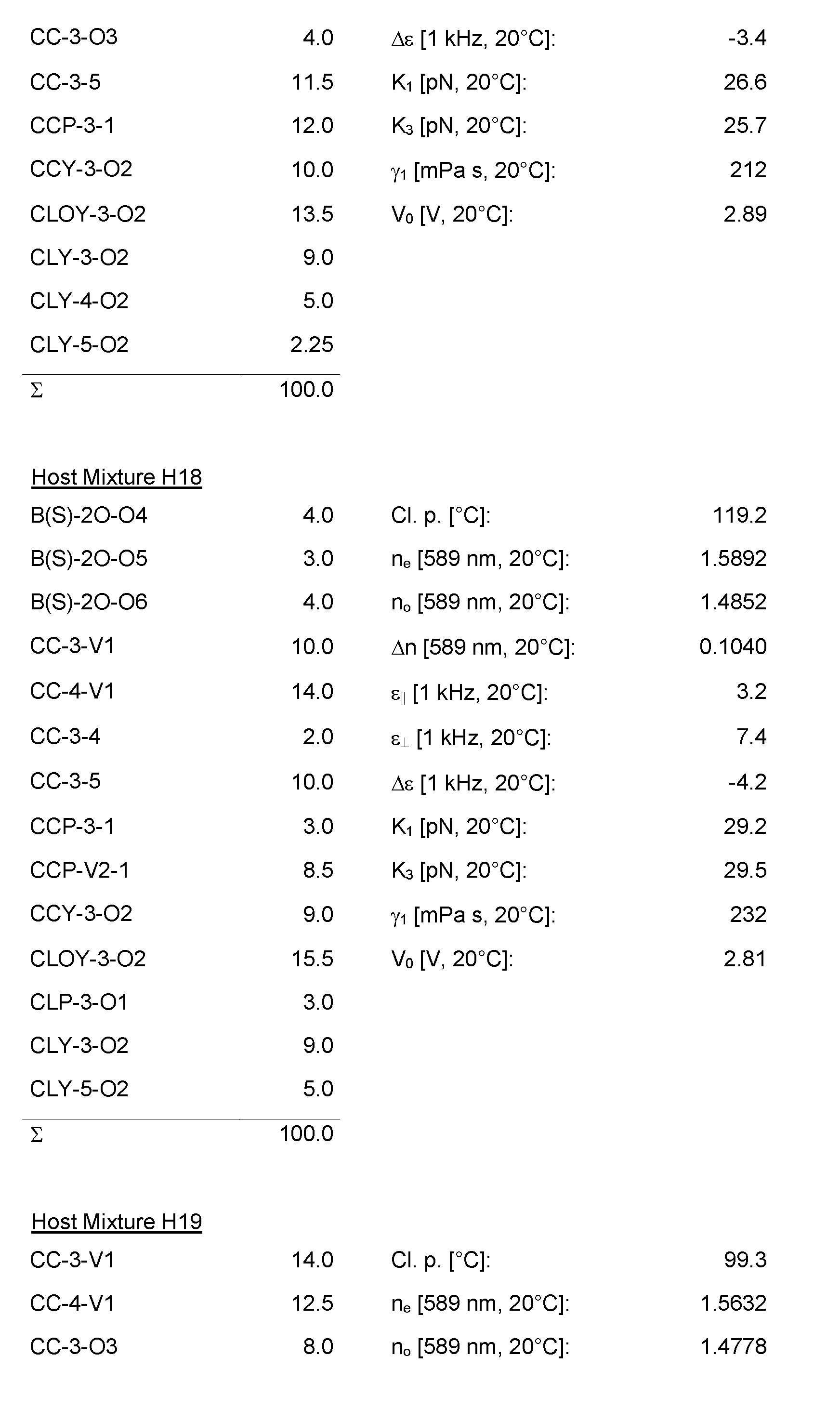
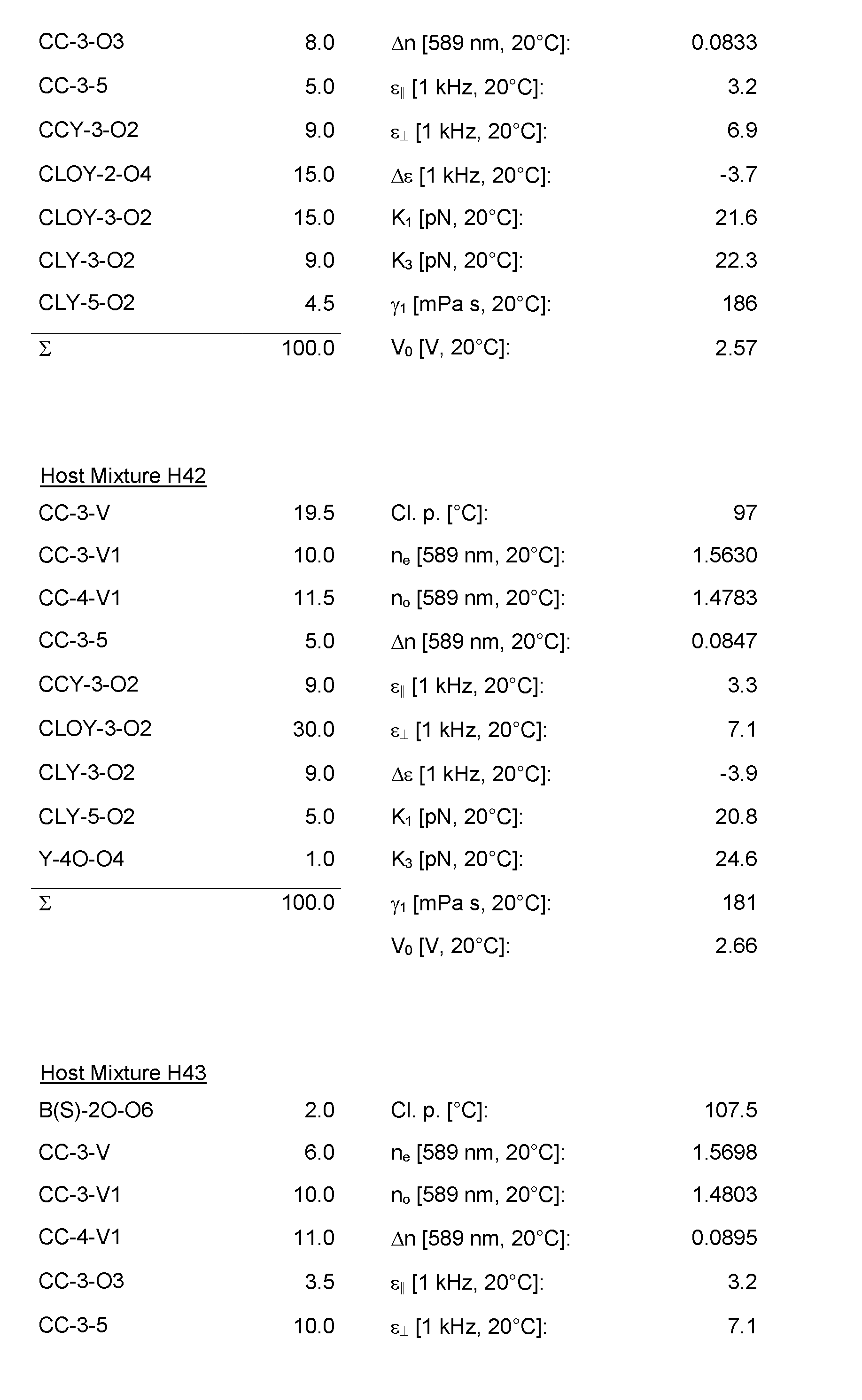
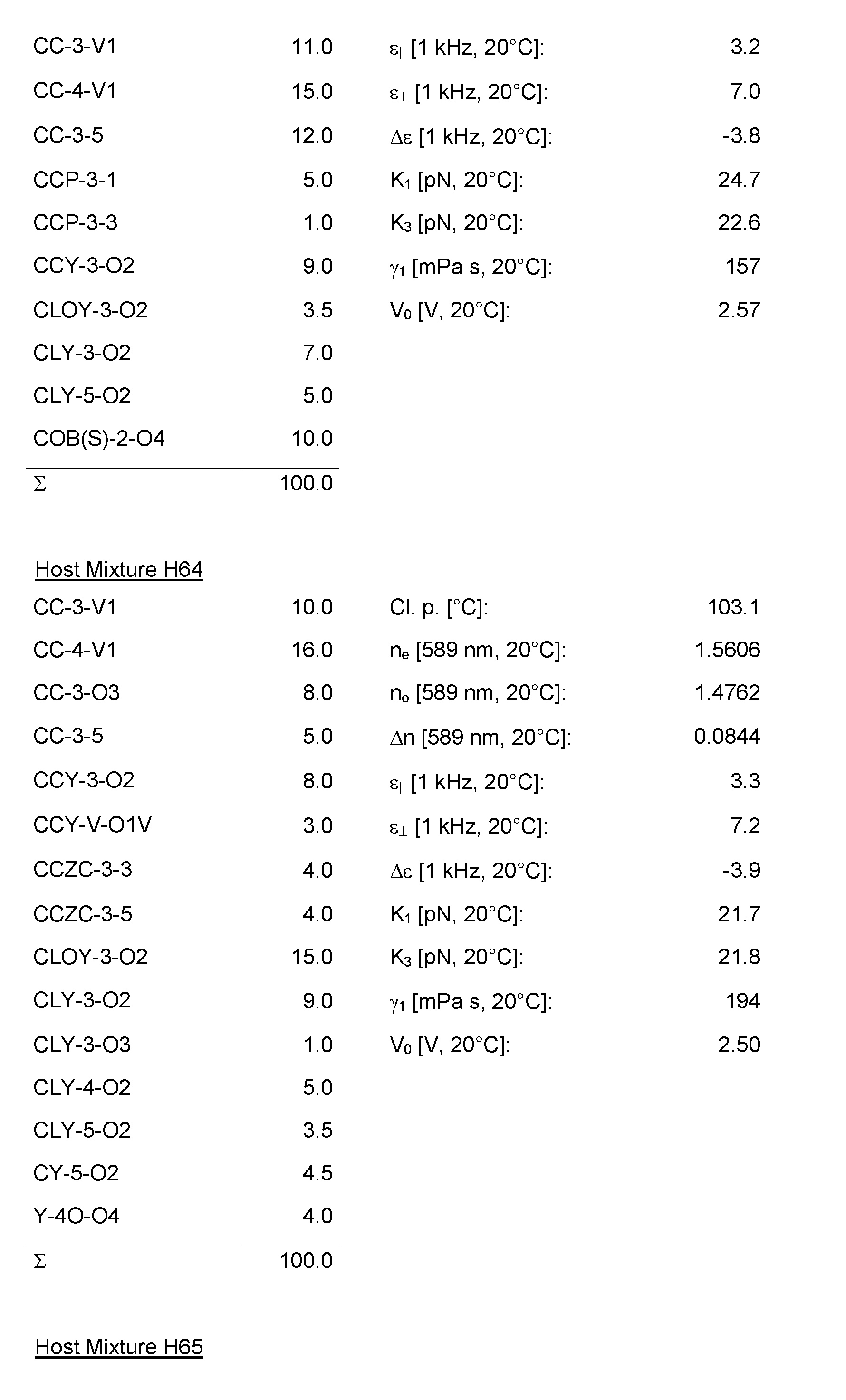
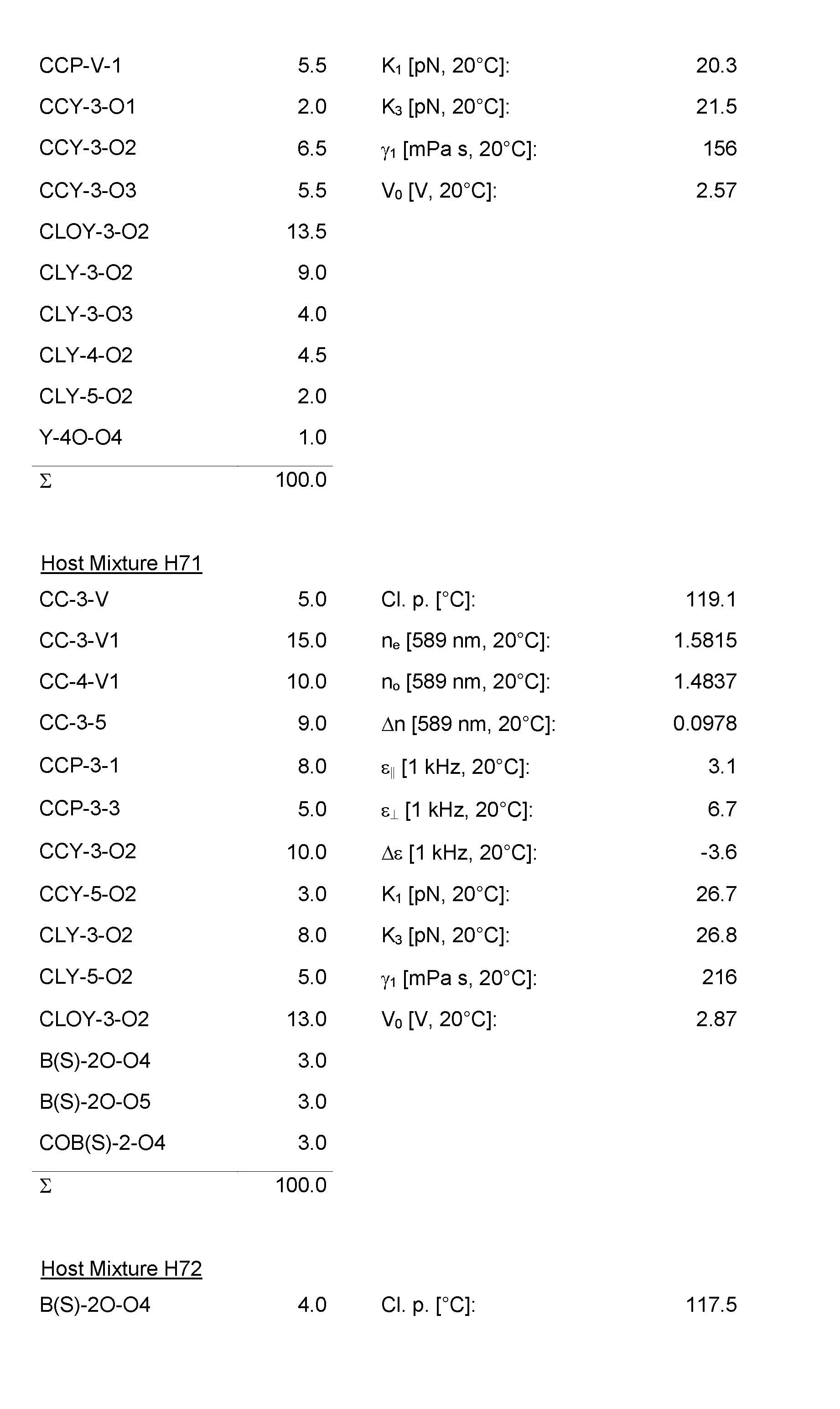
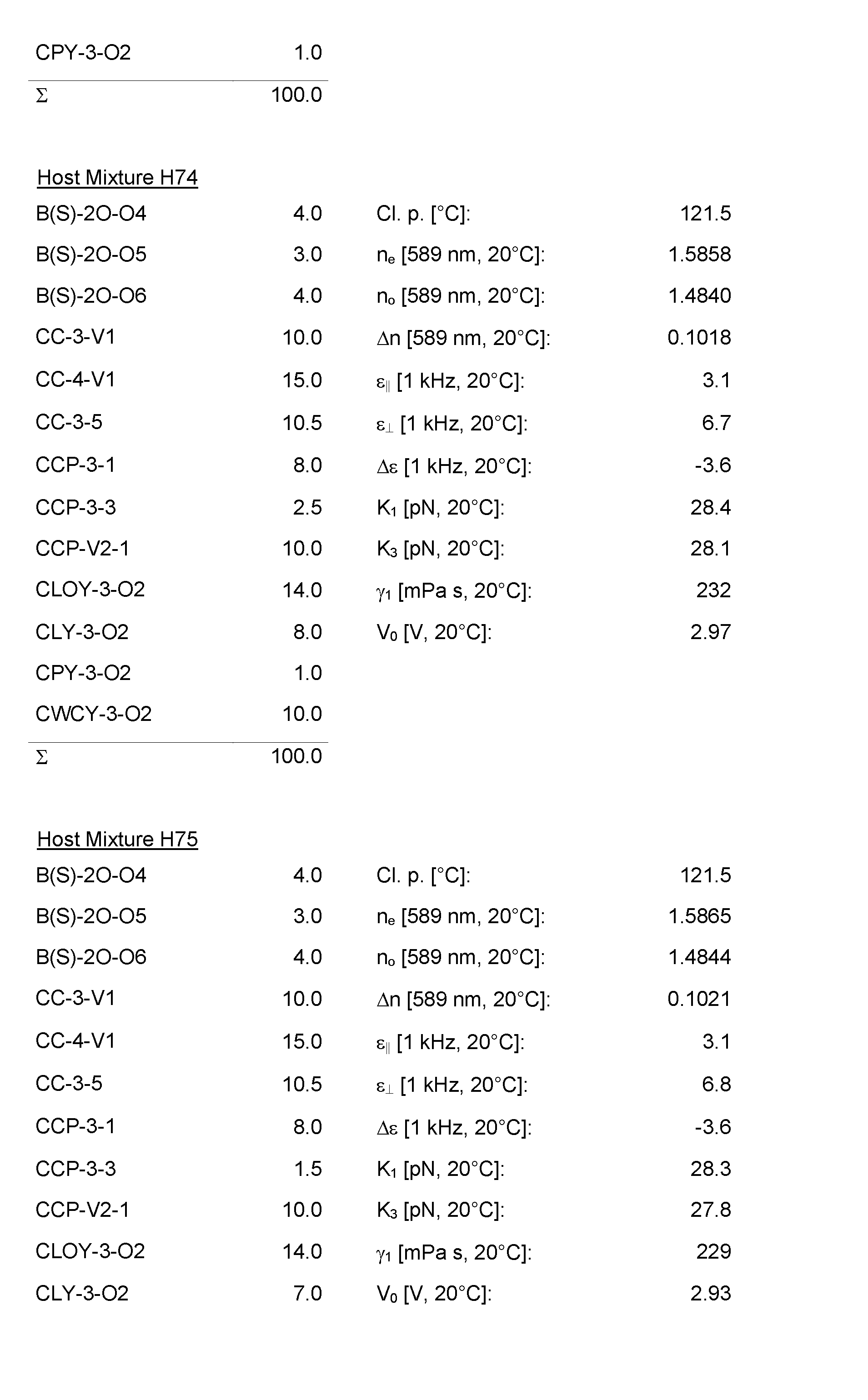
- the Host Mixtures H1 to H116 and the Mixture Examples M1 to M9 have the compositions and properties given in the following tables. The quantities in the tables are given in percent by weight.
- the Host mixture H1 and the mixture example M1 are filled in test cells with a cell gap of 3 ⁇ m and the dark state was investigated using a Konica Minolta CA-410 Display Analyser.
- the medium M1 shows an improvement of the dark state by 15% compared with the host mixture H1.
- the medium shows favourably high elastic constants K 1 , K3 and K aVg , which enables displays with improved contrast which in combination with the improved dark state results in an excellent picture quality of a display.
- Example M5 consists of the host H1 and the following additives:
- the host mixtures H2 to H8 and the mixture examples M2 to M8 are filled in test cells with a cell gap of 3 ⁇ m and the dark state was investigated using a Konica Minolta CA-410 Display Analyser.
- the medium M1 shows an improvement of the dark state by 15% compared with the host mixture H1.
- the medium shows favourably high elastic constants K 1 , K3 and K avg , which enables displays with improved contrast which in combination with the improved dark state results in an excellent picture quality of a display.
Landscapes
- Chemical & Material Sciences (AREA)
- Crystallography & Structural Chemistry (AREA)
- Engineering & Computer Science (AREA)
- Materials Engineering (AREA)
- Organic Chemistry (AREA)
- Liquid Crystal (AREA)
- Heterocyclic Carbon Compounds Containing A Hetero Ring Having Oxygen Or Sulfur (AREA)
- Liquid Crystal Substances (AREA)
Abstract
A liquid crystal material containing a compound of formula (I) in which the occurring groups and parameters have the meanings given in claim 1, and one ore more dichroic dyes, and the use thereof in an active matrix display, in particular in an IPS or FFS display.
Description
Liquid Crystal Medium
The present invention relates to a liquid crystal (LC) medium and to the use thereof for optical, electro-optical and electronic purposes, in particular in LC displays, especially in IPS, FFS, VA or PS-VA displays.
Liquid crystals (LCs) have found widespread use since the first commercially usable liquid-crystalline compounds were found. Liquid crystal displays (LCDs) are used in many areas for the display of information. LCDs are used both for direct-view displays and for projection-type displays.
One of the liquid crystal display modes used at present is the TN (“twisted nematic”) mode. However, TN LCDs have the disadvantage of a strong viewing-angle dependence of the contrast.
In addition, so-called VA (“vertically aligned”) displays are known which have a broader viewing angle. The LC cell of a VA display contains a layer of an LC medium between two transparent electrodes, where the LC medium usually has a negative dielectric anisotropy. In the switched-off state, the molecules of the LC layer are aligned perpendicular to the electrode surfaces (homeotropically) or have a tilted homeotropic alignment. On application of an electrical voltage to the two electrodes, a realignment of the LC molecules parallel to the electrode surfaces takes place.
Also known are so-called IPS (“in-plane switching”) displays, which contain an LC layer between two substrates, where the two electrodes are arranged on only one of the two substrates and preferably have intermeshed, comb-shaped structures. On application of a voltage to the electrodes, an electric field which has a significant component parallel to the LC layer is thereby generated between them. This causes realignment of the LC molecules in the layer plane.
Furthermore, so-called FFS (“fringe-field switching”) displays have been reported, see inter alia S.H. Jung et al., Jpn. J. Appl. Phys., Volume 43, No. 3, 2004, 1028, which contain two electrodes on the same substrate, one of which is structured in a comb- shaped manner and the other is unstructured. A strong, so-called "fringe field" is thereby generated, i.e. a strong electric field close to the edge of the electrodes, and, throughout the cell, an electric field which has both a strong vertical component and
also a strong horizontal component. FFS displays have a low viewing-angle dependence of the contrast. FFS displays usually contain an LC medium with positive dielectric anisotropy, and an alignment layer, usually of polyimide, which provides planar alignment to the molecules of the LC medium.
FFS displays can be operated as active-matrix or passive-matrix displays. In the case of active-matrix displays, individual pixels are usually addressed by integrated, non- linear active elements, such as for example transistors, for example thin-film transistors ("TFTs"), while in the case of passive-matrix displays individual pixels are usually addressed by the multiplex method.
Typical applications of the in-plane switching (IPS) and fringe-field switching (FFS) electro-optical modes are desktop monitors, notebooks, TVs, mobile telephones, tablet PCs, etc. and multimedia applications. In general, dielectrically positive liquid- crystalline media having rather lower values of the dielectric anisotropy are used in FFS displays, but in some cases liquid-crystalline media having a dielectric anisotropy of only about 3 or even less are also used in IPS displays.
A further improvement has been achieved by the so-called “High Brightness FFS” (HB-FFS) mode. One of the favourable features of the HB-FFS mode in contrast to the traditional FFS technology is that it enables higher transmittance which allows operation of the panel with lower energy consumption.
Furthermore, FFS displays have been described in S.H. Lee et al., Appl. Phys. Lett. 73(20), 1998, 2882-2883 and S.H. Lee et al., Liquid Crystals 39(9), 2012, 1141-1148, which have similar electrode design and layer thickness as FFS displays, but comprise a layer of an LC medium with negative dielectric anisotropy instead of an LC medium with positive dielectric anisotropy. The LC medium with negative dielectric anisotropy can show a more favourable director orientation that has less tilt and more twist orientation compared to the LC medium with positive dielectric anisotropy, as a result of which these displays may have a higher transmission. The displays further comprise an alignment layer, preferably of polyimide provided on at least one of the substrates that is in contact with the LC medium and induces planar alignment of the LC molecules of the LC medium. These displays are also known as "Ultra Brightness FFS” (UB-FFS) mode displays. These displays require an LC medium with high reliability.
Especially for monitor, mobile, video gaming and TV applications it is desirable to obtain favourably short response times, a wide viewing angle, low power consumption, a high transmittance and a suitably high contrast ratio of the LC displays.
It is inter alia desirable to provide displays with high brightness which at the same time can give favourable contrast, also under ambient light conditions. In this respect, it may in principle be considered to improve the image quality of the dark state in order to contribute to increasing the contrast ratio. For example, in general the retardation may be lowered by reducing the cell gap and/or by decreasing the optical anisotropy of the LC medium to improve the dark state quality. However, such a provision may negatively impact the device transmittance or brightness. It is in principle also possible to adjust the elastic constants of the LC medium to improve the dark state quality. Such an adjustment may however affect the operating voltage and the response time of the device.
There is still a need in the art for LC displays with a favourable electro-optical performance and reliability which offer broad applicability under various conditions, for example at different temperatures or different ambient light conditions or with reduced power consumption, and for liquid-crystalline media which can be suitably used in such LC displays.
It has presently been recognized that light leakage, in particular due to scattering, may detract from the dark or black state image quality and thus may also affect the obtainable contrast ratio.
An object of the present invention is therefore to provide improved liquid crystalline media for use in electro-optical devices and in particular displays with a favourable electro-optical performance, in particular with a high contrast ratio and a good black state while at the same time enabling high brightness or a high transmittance in one optical state, a low threshold voltage, fast addressing times and a favourable reliability and stability, in particular at low temperatures and at high temperatures. Further objects of the present invention are immediately evident to the person skilled in the art from the following detailed description.
Further, it is an object of the present invention to provide alternative media in addition to existing media known to the skilled person in order to broaden the range of available materials what allows for a more specific optimisation of a particular display.
The objects are solved by the subject-matter defined in the independent claims, while preferred embodiments are set forth in the respective dependent claims and are further described below.
The present invention in particular provides the following items including main aspects, preferred embodiments and particular features, which respectively alone and in combination contribute to solving the above objects and eventually provide additional advantages.
The present invention relates to a liquid crystal medium comprising a) one or more compounds of formula I
in which
R1 denotes H, a straight chain alkyl or alkoxy radical having 1 to 15 C atoms, or a branched alkyl or alkoxy radical having 3 to 15 C atoms, where one or more CH2 groups in these radicals may each be replaced, independently of one another, by
-CH=CH-, -OC-, -CF2O-, -OCF2-, -O-, -CO-O- or -O-CO-
in such a way that O atoms are not linked directly to one another, and in which, in addition, one or more H atoms may be replaced by halogen, preferably alkyl or alkenyl having up to 7 C atoms,
Y1 denotes H or CH3, preferably H, n is 0 or 1 , preferably 1 v is 1 , 2, 3, 4, 5, or 6, and b) one or more dichroic dyes.
It has surprisingly been found that the use of a medium according to the invention in a display can favourably enhance the obtainable contrast ratio of an electro-optical devices, while it has been further found that at the same time a suitably high transmittance or brightness, fast response times, a low operating voltage and low power consumption and a wide viewing angle of the devices may be maintained.
According to the invention the one or more dichroic dyes are provided in relatively small and limited yet effective amounts in the liquid-crystalline media to efficiently improve the dark state and the overall contrast of the devices and in particular displays, e.g. FFS, HB-FFS and especially UB-FFS displays, without however affecting the desired device transmittance or brightness or only reducing the device transmittance or brightness to a minimal or even negligible extent.
According to the invention the liquid-crystalline medium comprises one or more dichroic dyes, preferably in a total amount of 2.0% by weight or less. In a preferred embodiment the total amount of the one or more dichroic dyes in the liquid-crystalline medium is 1.0% by weight or less. It is further preferred that the total amount of the
one or more dichroic dyes in the liquid-crystalline medium is 0.5% by weight or less, more preferably 0.2% by weight or less.
Herein, a dichroic dye is taken to mean a light-absorbing compound in which the absorption properties are dependent on the orientation of the compound relative to the direction of polarization of the light. A dichroic dye compound in accordance with the present invention typically has an elongated shape, i.e. the compound is significantly longer in one spatial direction, i.e. along the longitudinal axis, than in the other two spatial directions. The dichroic dye absorbs, or respectively preferentially absorbs, light in one orientation so that light transmission may be modulated by changing the orientation of the dichroic dye.
According to the invention a so-called liquid crystal host is doped with dichroic dye molecules in relatively small, limited amounts. These dye-doped LC media are used particularly preferably for FFS and UB-FFS modes, however also other display modes as described above and below can be employed.
Preferably, the dichroic dye compounds are present in the liquid-crystalline medium in solution. The dichroic dye molecules as used according to the invention therefore preferably exhibit suitable and sufficient solubility in the liquid crystal host. In addition, the dye compounds preferably also favourably contribute to the stability and reliability of the media.
Each of the one or more dichroic dyes is preferably present in the liquid-crystalline medium in a proportion of 0.002% by weight to 2.0% by weight, more preferably 0.003% by weight to 1.0% by weight, still more preferably 0.005% by weight to 0.5% by weight, even more preferably 0.01% by weight to 0.3% by weight, and particularly preferably 0.02% by weight to 0.2% by weight, based on the overall weight of the entire medium.
Preferably, the one or more dichroic dyes are present in the liquid-crystalline medium overall in a total concentration which is in the range of from 0.003% by weight to 2.0% by weight, more preferably 0.004% by weight to 1 .5% by weight, still more preferably 0.005% by weight to 1.0% by weight, yet more preferably 0.01% by weight to 0.5% by weight, even more preferably 0.02% by weight to 0.3% by weight, and particularly preferably 0.05% by weight to 0.2% by weight.
The concentration of the dye(s) is favourably chosen such that the proper performance of the obtained liquid-crystalline material is ensured, in particular in terms of the desired reduction of the light leakage in the dark state and the improved black image quality and overall contrast, while suitably maintaining other device properties such as response times, operating voltage and brightness. The upper limit of the dye concentration is provided in particular in view of maintaining the bright state performance of the device such that transmittance is not affected or only to a minimal extent.
Dichroic dyes may preferably be selected from for example azo dyes, anthraquinones, thiophenolanthraquinones, methine compounds, azomethine compounds, merocyanine compounds, naphthoquinones, tetrazines, pyrromethene dyes, malononitrile dyes, nickel dithiolenes, (metal) phthalocyanines, (metal) naphthalocyanines and (metal) porphyrins, rylenes, in particular perylenes and terylenes, thiadiazole dyes, thienothiadiazole dyes, benzothiadiazoles, thiadiazoloquinoxalines, and diketopyrrolopyrroles. Particular preference is given to azo compounds, anthraquinones, thiophenolanthraquinones, benzothiadiazoles, in particular as described in WO 2014/187529 and in WO 2020/104563 A1, diketopyrrolopyrroles, in particular as described in WO 2015/090497, thiadiazoloquinoxalines, in particular as described in WO 2016/177449 and in WO 2020/104563 A1 , and rylenes, in particular as described in WO 2014/090373.
In a preferred embodiment the one or more dichroic dyes are selected from azo compounds, benzothiadiazoles and thiadiazoloquinoxalines.
The liquid-crystalline medium preferably comprises one, two, three, four, five, six, seven, eight, nine or ten different dichroic dyes, particularly preferably two, three, four, five or six dichroic dyes. In a preferred embodiment the medium contains at least three different dichroic dyes.
In an embodiment the absorption spectra of the dichroic dyes contained in the medium or respectively the switchable layer preferably complement one another in such a way that the impression of a black colour, or respectively a colour-neutral appearance, arises for the eye. Preferably two or more, more preferably three or more dichroic dyes are used in the liquid-crystalline medium to preferably cover a large part of the visible spectrum. The precise way in which a mixture of dyes which
appears black or grey to the eye can be prepared is known in the art and is described, for example, in M. Richter, Einfuhrung in die Farbmetrik [Introduction to Colorimetry], 2nd Edition, 1981 , ISBN 3 11-008209-8, Walter de Gruyter & Co.
The setting of the colour location of a mixture of dyes is described in the area of colorimetry. To this end, the spectra of the individual dyes are calculated taking into account the Lambert-Beer law to give an overall spectrum and converted into the corresponding colour locations and luminance values under the associated illumination, for example illuminant D65 for daylight, in accordance with the rules of colorimetry. The position of the white point is fixed by the respective illuminant, for example D65, and is quoted in tables, for example in the reference above. Different colour locations can be set by changing the proportions of the various dyes.
According to a preferred embodiment, the medium and the switchable layer comprise one or more dichroic dyes which absorb light in the visible spectrum, which herein is defined as light having a wavelength of from 380 nm to 780 nm.
In an embodiment the dichroic dyes provided in the medium and the switchable layer are preferably selected from the dye classes indicated in B. Bahadur, Liquid Crystals - Applications and Uses, Vol. 3, 1992, World Scientific Publishing, Section 11.2.1, and particularly preferably from the explicit compounds given in the table present therein.
Said dyes belong to the classes of dichroic dyes which are known in the art and have been described in the literature. Thus, for example, anthraquinone dyes are described in EP 34832, EP 44893, EP 48583, EP 54217, EP 56492, EP 59036, GB 2065158, GB 2065695, GB 2081736, GB 2082196, GB 2094822, GB 2094825, JP A 55- 123673, DE 3017877, DE 3040102, DE 3115147, DE 3115762, DE 3150803 and DE 3201120, naphthoquinone dyes are described in DE 3126108 and DE 3202761 , azo dyes in EP 43904, DE 3123519, WO 82/2054, GB 2079770, JP-A 56-57850, JP-A 56- 104984, US 4308161, US 4308162, US 4340973, T. Uchida, C. Shishido, H. Seki and M. Wada: Mol. Cryst. Liq. Cryst. 39, 39-52 (1977), and H. Seki, C. Shishido, S. Yasui and T. Uchida: Jpn. J. Appl. Phys. 21, 191-192 (1982), and perylenes are described in EP 60895, EP 68427 and WO 82/1191. Rylene dyes as described, for example, in EP 2166040, US 2011/0042651 , EP 68427, EP 47027, EP 60895, DE 3110960 and EP 698649.
in which R denotes straight chain alkyl having 1 to 20 C atoms, or branched alkyl having 3 to 20 C atoms, or straight chain alkenyl having 2 to 20 C atoms, or branched alkenyl having 3 to 20 C atoms, and (O) denotes O or a single bond. Preferred compounds of formula I are selected from the following sub-formulae:
Very preferred compounds of formula I are the compounds 1-1 to 1-16.
In a preferred embodiment, alternatively or in addition to the compounds of the formula I, the medium comprises one or more compounds of formula IA,
in which the occurring groups and parameters have the meanings given above under formula I, and
In a preferred embodiment, the medium further comprises one or more compounds selected from the group of compounds of the formulae IIA, IIB, IIC and IID,
in which
R2A, R2B, R2C and R2D identically or differently, denote H, a straight chain alkyl or alkoxy radical having 1 to 15 C atoms, or a branched alkyl or alkoxy radical having 3 to 15 C atoms, where one or more CH2 groups in these radicals may each be replaced, independently of one another, by
-CH=CH-, -C≡C-, -CF2O-, -OCF2-, -O-, -CO-O- or -O-CO-
in such a way that O atoms are not linked directly to one another, and in which, in addition, one or more H atoms may be replaced by halogen, preferably alkyl or alkenyl having up to 7 C atoms,
L1 to L4 each, independently of one another, denote F, Cl, CF3 or CHF2,
Y denotes H, F, Cl, CF3, CHF2 or CH3, preferably H or CH3, particularly preferably H,
Z2, Z2B and Z2D each, independently of one another, denote a single bond, -CH2CH2-, -CH=CH-, -CF2O-, -OCF2-, -CH2O-, -OCH2-, -COO-, -OC O-, -C2F4-, -CF=CF-, -CH=CHCH2O-,
(O) denotes O or a single bond, p denotes 0, 1 or 2, q denotes 0 or 1 , and v denotes an integer from 1 to 6.
In the compounds of the formulae IIA, IIB and IID, Z2 may have identical or different meanings. In the compounds of the formula IIB, Z2 and Z2B may have identical or
different meanings. In the compounds of the formula IID, Z2 and Z2D may have identical or different meanings. In the compounds of the formulae IIA, IIB, IIC and IID, R2A, R2B, R2C and R2D each preferably denote alkyl having 1 to 6 C atoms, in particular CH3, C2H5, n-C3H7, n-C4H9, n-C5H11. In the compounds of the formulae IIA, IIB and IID, L1, L2, L3 and L4 preferably denote L1 = L2 = F and L3 = L4 = F, furthermore L1 = F and L2 = Cl, L1 = Cl and L2 = F, L3 = F and L4 = Cl, L3 = Cl and L4 = F. Z2 and Z2B in the formulae IIA and IIB preferably each, independently of one another, denote a single bond, furthermore a -C2H4- bridge. If, in the formula IIB, if Z2 = -C2H4- or -CH2O-, Z2B is preferably a single bond or, if Z2B = -C2H4- or -CH2O-, Z2 is preferably a single bond. In formula IID, Z2D is preferably a single bond. In the compounds of the formulae IIA, IIB and IID, (O)CvH2v+1 preferably denotes OCvH2v+1. In the compounds of the formula IIC, (O)CvH2v+1 preferably denotes CvH2v+1. In the compounds of the formula IIC, L3 and L4 preferably each denote F. Preferred compounds of the formulae IIA IIB IIC and IID are indicated below:
in which the parameter a denotes 1 or 2, alkyl and alkyl* each, independently of one another, denote a straight-chain alkyl radical having 1-6 C atoms, and alkenyl denotes a straight-chain alkenyl radical having 2-6 C atoms, and (O) denotes an oxygen atom or a single bond. Alkenyl preferably denotes CH2=CH-,
CH2=CHCH2CH2-, CH3-CH=CH-, CH3-CH2-CH=CH-, CH3-(CH2)2-CH=CH-, CH3- (CH2)3-CH=CH- or CH3-CH=CH-(CH2)2-.
Particularly preferred mixtures according to the invention comprise one or more compounds of the formulae IIA-2, IIA-8, IIA-10, IIA-16, 11-18, IIA-40, IIA-41 , IIA-42, IIA- 43, IIB-2, IIB-10, IIB-16, IIC-1 , and IID-4.
The proportion of compounds of the formulae IIA and/or IIB in the mixture as a whole is preferably at least 20 % by weight.
Particularly preferred media according to the invention comprise at least one compound of the formula IIC-1 ,
in which alkyl and alkyl* have the meanings indicated above, preferably in amounts of > 3 % by weight, in particular > 5 % by weight and particularly preferably 5 to 25 % by weight.
The medium according to the invention preferably comprises one or more compounds of formula III
in which
R31 and R32 each, independently of one another, denote H, an alkyl or alkoxy radical having 1 to 15 C atoms, where one or more CH2 groups in these radicals may each be replaced, independently of one another, by
-CH=CH-, -O-, -CO-O-, or -O-CO- in such a way that O atoms are not linked directly to one another, and in which one or more H atoms may each be replaced by halogen,
A31 on each occurrence, independently of one another, denotes a) 1,4-cyclohexenylene or 1,4-cyclohexylene radical, in which one or two non- adjacent CH2 groups may each be replaced by -O- or -S-, b) a 1,4-phenylene radical, in which one or two CH groups may each be replaced by N, or c) a radical from the group spiro[3.3]heptane-2,6-diyl, 1,4-bicyclo[2.2.2]- octylene, naphthalene-2,6-diyl, decahydronaphthalene-2,6-diyl, 1, 2,3,4- tetrahydronaphthalene-2,6-diyl, phenanthrene-2,7-diyl and fluorene-2,7-diyl, where the radicals a), b) and c) may be mono- or polysubstituted by halogen atoms, n denotes 0, 1 or 2, preferably 0 or 1,
Z31 on each occurrence independently of one another denotes -CO-O-, -O-CO-, -CF2O- , -OCF2-, -CH2O-, -OCH2-, -CH2-, -CH2CH2-, -(CH2)4-, -CH=CH-CH2O-, -C2F4-, -CH2CF2-, -CF2CH2 -, -CF=CF-, -CH=CF-, -CF=CH-, -CH=CH-, -C=C- or a single bond, and
L31 and L32 each, independently of one another, denote H, F, Cl, CF3 or CHF2, preferably H or F, most preferably F, and
W denotes O or S, preferably S.
The compounds of formula III are preferably selected from the compounds of the formula 111-1 and/or III-2
in which the occurring groups have the same meanings as given under formula III above and preferably
R31 and R32 each, independently of one another, an alkyl, alkenyl or alkoxy radical having up to 15 C atoms, more preferably one or both of them denote an alkoxy radical having 1 to 7 C atoms, and
L31 and L32 each preferably denote F.
Preferably, the compounds of the formula 111-1 are selected from the group of compounds of the formulae 111-1-1 to 111-1-11 , preferably of formula 111-1-6,
alkyl and alkyl* each, independently of one another, denote a straight-chain alkyl radical having 1-6 C atoms, alkenyl and alkenyl* each, independently of one another, denote a straight-chain alkenyl radical having 2-6 C atoms, alkoxy and alkoxy* each, independently of one another, denote a straight-chain alkoxy radical having 1-6 C atoms, and L31 and L32 each, independently of one another, denote F or Cl, preferably both F.
Preferably, the compounds of the formula III-2 are selected from the group of compounds of the formulae 111-2-1 to 111-2-10, preferably of formula III-2-6,
alkyl and alkyl* each, independently of one another, denote a straight-chain alkyl radical having 1-6 C atoms, alkenyl and alkenyl* each, independently of one another, denote a straight-chain alkenyl radical having 2-6 C atoms, alkoxy and alkoxy* each, independently of one another, denote a straight-chain alkoxy radical having 1-6 C atoms, and L31 and L32 each, independently of one another, denote F or Cl, preferably both F.
Optionally the medium comprises one or more compounds of the formula IIIA-1 and/or IIIA-2
in which L31 and L32 have the same meanings as given under formula III, (O) denotes O or a single bond,
RIIIA denotes alkyl or alkenyl having up to 7 C atoms or a group Cy-CmH2m+1-, m and n are, identically or differently, 0, 1 , 2, 3, 4, 5 or 6, preferably 1 , 2 or 3, very preferably 1 ,
Cy denotes a cycloaliphatic group having 3, 4 or 5 ring atoms, which is optionally substituted with alkyl or alkenyl each having up to 3 C atoms, or with halogen or CN, and preferably denotes cyclopropyl, cyclobutyl or cyclopentyl The compounds of formula IIIA-1 and/or IIIA-2 are contained in the medium either alternatively or additionally to the compounds of formula III, preferably additionally.
Very preferred compounds of the formulae IIIA-1 and IIIA-2 are the following:
in which alkoxy denotes a straight-chain alkoxy radical having 1-6 C atoms or alternatively -(CH2)nF in which n is 2,3,4, or 5, preferably C2H4F.
In a preferred embodiment of the present invention, the medium comprises one or more compounds of formula III-3
in which
R31, R32 identically or differently, denote H, an alkyl or alkoxy radical having 1 to 15 C atoms, in which one or more CH2 groups in these radicals are optionally replaced, independently of one another, by -C=C-, -CF2O-, -OCF2-, -CH=CH-,
-CO-O- or -O-CO- in such a way that O atoms are not linked directly to one another, and in which, in addition, one or more H atoms may each be replaced by halogen,
R31 preferably denotes alkyl having 1 to 7 C atoms, in which one or more CH2 groups are optionally replaced by -CH=CH-
R31 preferably denotes alkoxy having 1 to 7 C atoms, in which one or more CH2 groups are optionally replaced by -CH=CH-,
and
L31 and L32 each, independently of one another, denote F or Cl, preferably both F.
The compounds of formula III-3 are preferably selected from the group of compounds of the formulae 111-3-1 to 111-3-10:
in which R32 denotes alkyl having 1 to 7 C-atoms, preferably ethyl, n-propyl or n-butyl, or alternatively cyclopropylmethyl, cyclobutylmethyl or cyclopentyl methyl or alternatively -(CH2)nF in which n is 2,3,4, or 5, preferably C2H4F.
In a preferred embodiment of the present invention, the medium comprises one or more compounds of the formulae III-4 to III-6, preferably of formula II I-5,
in which the parameters have the meanings given above, R31 preferably denotes straight-chain alkyl and R32 preferably denotes alkoxy, each having 1 to 7 C atoms.
In a preferred embodiment the medium comprises one or more compounds of the formula I selected from the group of compounds of formulae 111-7 to 111-9, preferably of formula 111-8,
in which the parameters have the meanings given above, R31 preferably denotes straight-chain alkyl and R32 preferably denotes alkoxy each having 1 to 7 C atoms.
R41 denotes an unsubstituted alkyl radical having 1 to 7 C atoms or an unsubstituted alkenyl radical having 2 to 7 C atoms, preferably an n-alkyl radical, particularly preferably having 2, 3, 4 or 5 C atoms, and
R42 denotes an unsubstituted alkyl radical having 1 to 7 C atoms or an unsubstituted alkoxy radical having 1 to 6 C atoms, both preferably having 2 to 5 C atoms, an unsubstituted alkenyl radical having 2 to 7
C atoms, preferably having 2, 3 or 4 C atoms, more preferably a vinyl radical or a 1 -propenyl radical and in particular a vinyl radical.
The compounds of the formula IV are preferably selected from the group of the compounds of the formulae I V-1 to IV-4,
in which alkyl and alkyl’, independently of one another, denote alkyl having 1 to 7 C atoms, preferably having 2 to 5 C atoms, alkenyl denotes an alkenyl radical having 2 to 5 C atoms, preferably having
2 to 4 C atoms, particularly preferably 2 C atoms, alkenyl’ denotes an alkenyl radical having 2 to 5 C atoms, preferably having
2 to 4 C atoms, particularly preferably having 2 to 3 C atoms, and alkoxy denotes alkoxy having 1 to 5 C atoms, preferably having 2 to 4 C atoms.
Preferably, the medium comprises one or more compounds selected from the compounds of the formulae IV-1-1 to IV-1-6
Very preferably, the medium according to the invention comprises one or more compounds of the formulae I -2-1 and/or I -2-2
Very preferably, the medium according to the invention comprises a compound of formula I V-3, in particular selected from the compounds of the formulae IV-3-1a to IV-3-1c, IV-3-2a to IV-3-2c and IV-3-3a, in particular selected from IV-3-2a to IV-3-2c and IV-3-3a:
Very preferably, the medium according to the invention comprises a compound of formula IV-4, in particular selected from the compounds of the formulae IV-4-1 and IV-4-2
The liquid-crystalline medium preferably additionally comprises one or more compounds of the formula IVa,
IVa
in which
R41 and R42 each, independently of one another, denote a straight-chain alkyl, alkenyl, alkoxyalkyl or alkoxy radical having up to 12 C atoms, and
Z4 denotes a single bond, -CH2CH2-, -CH=CH-, -CF2O-, -OCF2-, -CH2O-, -OCH2-, -C OO-, -OCO-, -C2F4-, -C4H8-, -CF=CF-.
Preferred compounds of the formula IVa are indicated below:
alkyl and alkyl* each, independently of one another, denote a straight-chain alkyl radical having 1 to 6 C atoms.
The medium according to the invention preferably comprises at least one compound of the formula IVa-1 and/or formula IVa-2.
The proportion of compounds of the formula IVa in the mixture as a whole is preferably at least 5 % by weight
Preferably, the medium comprises one or more compounds of formula IVb-1 to IVb-3
in which alkyl and alkyl* each, independently of one another, denote a straight- chain alkyl radical having 1 to 6 C atoms, and alkenyl and alkenyl* each, independently of one another, denote a straight- chain alkenyl radical having 2 to 6 C atoms.
The proportion of the biphenyls of the formulae IVb-1 to IVb-3 in the mixture as a whole is preferably 5 % by weight or less, in particular 3 % by weight or less.
Of the compounds of the formulae IVb-1 to IVb-3, the compounds of the formula IVb- 2 are particularly preferred.
Particularly preferred biphenyls of the formulae IVb-1 to IVb-3 are
in which alkyl* denotes an alkyl radical having 1 to 6 C atoms and preferably denotes n-propyl. The medium according to the invention particularly preferably comprises one or more compounds of the formulae IVb-1-1 and/or IVb-2-3.
In a preferred embodiment, the medium according to the invention comprises one or more compounds of formula V
in which
R51, R52 denote alkyl having 1 to 7 C atoms, alkoxy having 1 to 7 C atoms, or alkoxyalkyl, alkenyl or alkenyloxy having 2 to 7 C atoms,
Z51 , Z52 each, independently of one another, denote -CH2-CH2-, -CH2-O-, -CH=CH-,
-C=C-, -COO- or a single bond, and n is 1 or 2, where the compounds of the formula CL are excluded from formula V.
The compounds of formula V are preferably selected from the compounds of the formulae V-1 to V-16:
in which R51 and R52 have the meanings indicated for formula V above.
R51 and R52 preferably each, independently of one another, denote straight-chain alkyl having 1 to 7 C atoms or alkenyl having 2 to 7 C atoms.
Preferred media comprise one or more compounds of the formulae V-1, V-5, V-6, V-8, V-9, V-10, V-11 , V-12, V-14, V-15, and/or V-16, very preferably V-5 and/or V-6 and/or V-8.
Preferably, the medium according to the invention comprises one or more compounds of the formula CL
in which
RL denotes H, a straight chain or branched alkyl or alkoxy radical having 1 to 15 C atoms, or a straight chain or branched alkenyl radical having 2 to 15 C atoms, where one or more CH2 groups in these radicals may each be replaced, independently of one another, by
-C≡C-, -CF2O-, -OCF2-, -CH=CH-, -O-, -CO-O- or -O-CO- in such a
way that O atoms are not linked directly to one another, and in which, in addition, one or more H atoms may each be replaced by halogen,
XL denotes F, Cl, CN, CHF2, CF3, OCF3, or, identically or differently, has one of the meanings of RL,
YL denotes H, F, Cl or CH3.
The compounds of formula CL are preferably selected form the group of compounds of the formulae CL-1 , CL-2 and CL-3:
in which
RL1 and RL2, identically or differently, have the meanings given above for RL in formula CL and, preferably denote alkyl or alkenyl having 1 to 7 C atoms or 2 to 7 C atoms, respectively, in which a CH2 group may be replaced by cyclopropane- 1 , 2-diyl.
Very preferred compounds of the formula I are selected from the compounds of the formulae CL-3-1 to CL-3-12:
In a preferred embodiment, the medium according to the invention comprises the compound CL-3-1.
In a preferred embodiment the medium comprises one or more compounds of formula Vb selected from the following sub-formulae
Vb-1
in which RL1 has the meaning given above for formula Va.
Very preferably, the medium comprises the compound Vb-2, in which RL1 denotes alkyl or alkenyl having up to 7 C atoms, in particular vinyl.
In a preferred embodiment of the present invention the medium additionally comprises one or more compounds of the formulae VI-1 to VI-9
R7 each, independently of one another, have one of the meanings indicated for R2A in Claim 5, and w and x each, independently of one another, denote 1 to 6.
Particular preference is given to mixtures comprising at least one compound of the formula V-9.
In a preferred embodiment of the present invention the medium additionally comprises one or more compounds of the formulae VII-1 to VII-21 ,
R denotes a straight-chain alkyl or alkoxy radical having 1 to 6 C atoms, (O) denotes -O- or a single bond, and m is 0, 1 , 2, 3, 4, 5 or 6 and n is 0, 1 , 2, 3 or 4.
R preferably denotes methyl, ethyl, propyl, butyl, pentyl, hexyl, methoxy, ethoxy, propoxy, butoxy, pentoxy.
The medium according to the invention preferably comprises the terphenyls of the formulae VII-1 to VII-21 in amounts of 2 to 30 % by weight, in particular 5 to 20 % by weight.
Particular preference is given to compounds of the formulae VII-1, VII-2, VII-4, VII-20 and VII-21. In these compounds, R preferably denotes alkyl, furthermore alkoxy, each having 1 to 5 C atoms. In the compounds of the formula VII-20, R preferably denotes alkyl or alkenyl, in particular alkyl. In the compound of the formula VII-21, R preferably denotes alkyl.
The terphenyls are preferably employed in the mixtures according to the invention if the An value of the mixture is to be > 0.1. Preferred mixtures comprise 2 to 20 % by weight of one or more terphenyl compounds selected from the group of the compounds VII-1 to VII-21.
Further preferred embodiments are listed below: a) Liquid-crystalline medium comprising at least one compound of the formulae
Z-1 to Z-7,
in which R has the meanings indicated for R1 of formula I, preferably denotes straight-chain alkyl or alkoxy having 1 to 7 C atoms or straight-chain alkenyl having 2 to 7 C atoms, (O) denotes an oxygen atom or a single bond, and alkyl denotes n-alkyl having 1 to 6 C atoms.
b) Preferred liquid-crystalline media according to the invention comprise one or more substances which contain a tetrahydronaphthyl or naphthyl unit, such as, for example, the compounds of the formulae N-1 to N-5,
in which R1N and R2N each, independently of one another, have the meanings indicated for R2A (see formula IIA), preferably denote straight-chain alkyl, straight-chain alkoxy or straight-chain alkenyl, and Z1 and Z2 each, independently of one another, denote -C2H4-, -CH=CH-, -(CH2)4-, -(CH2)3O-, -O(CH2)3-, -CH=CHC H2CH2-, -CH2CH2CH=CH-, -CH2O-, -OCH2-, -COO-, -OCO-, -C2F4-, -CF=CF-, -CF=CH-, -CH=CF-, -CF2O-, -OCF2-, -CH2- or a single bond.
c) Preferred mixtures comprise one or more compounds selected from the group of the difluorodibenzochroman compounds of the formula BC, chromans of the formula CR, fluorinated phenanthrenes of the formulae PH-1 and PH-2, fluorinated dibenzofurans of the formula BF-1 and BF-2,
in which
RB1, RB2, RCR1, RCR2, R1, R2 each, independently of one another, have the meaning of R2A. c is 0, 1 or 2 and d denotes 1 or 2. R1 and R2 preferably, independently of one another, denote alkyl or alkoxy having 1 to 6 C atoms.
The mixtures according to the invention preferably comprise the compounds of the formulae BC, CR, PH-1 , PH-2 in amounts of 3 to 20 % by weight, in particular in amounts of 3 to 15 % by weight.
Particularly preferred compounds of the formulae BC and CR are the compounds BC-1 to BC-7 and CR-1 to CR-5,
BC-1
in which alkyl and alkyl* each, independently of one another, denote a straight-chain alkyl radical having 1 to 6 C atoms, and alkenyl and alkenyl* each, independently of one another, denote a straight-chain alkenyl radical having 2 to 6 C atoms.
Very particular preference is given to mixtures comprising one, two or three compounds of the formula BC-2. d) Preferred mixtures comprise one or more indane compounds of the formula In,
in which
R11, R12,
R13 each, independently of one another, denote a straight-chain alkyl, alkoxy, alkoxyalkyl or alkenyl radical having 1 to 6 C atoms,
Preferred compounds of the formula In are the compounds of the formulae ln-1 to In-16 indicated below:
Particular preference is given to the compounds of the formulae ln-1 , ln-2, ln-3 and ln-4.
The compounds of the formula In and the sub-formulae ln-1 to In-16 are preferably employed in the mixtures according to the invention in concentrations
≥ 5 % by weight, in particular 5 to 30 % by weight and very particularly preferably 5 to 25 % by weight. e) Preferred mixtures additionally comprise one or more compounds of the formulae L-1 to L-5,
in which
R and R1 each, independently of one another, have the meanings indicated for R2A in formula IIA above, and alkyl denotes an alkyl radical having 1 to 6 C atoms. The parameter s denotes 1 or 2.
The compounds of the formulae L-1 to L-5 are preferably employed in concentrations of 5 to 50 % by weight, in particular 5 to 40 % by weight and very particularly preferably 10 to 40 % by weight. f) Preferred mixtures additionally comprise one or more compounds of formula IIA-Y
in which R11 and R12 have one of the meanings given in formula I above, and L1 and L2, identically or differently, denote F or Cl.
Preferred compounds of the formula IIA-Y are selected from the group consisting of the following subformulae
in which, Alkyl and Alkyl* each, independently of one another, denote a straight-chain alkyl radical having 1-6 C atoms, Alkoxy denotes a straight-chain alkoxy radical having 1-6 C atoms, Alkenyl and Alkenyl* each, independently of one another, denote a straight-chain alkenyl radical having 2-6 C atoms, and O denotes an oxygen atom or a single bond. Alkenyl and Alkenyl* preferably denote CH2=CH-, CH2=CHCH2CH2-,
CH3-CH=CH-, CH3-CH2-CH=CH-, CH3-(CH2)2-CH=CH-, CH3-(CH2)3-CH=CH- or CH3- CH=CH-(CH2)2-.
Particularly preferred compounds of the formula IIA-Y are selected from the group consisting of following subformulae:
in which Alkoxy and Alkoxy* have the meanings defined above and preferably denote methoxy, ethoxy, n- propyloxy, n-butyloxy or n-pentyloxy.
The liquid crystal medium according to the invention, herein also referred to as liquid crystal host mixture, is suitable for the use in polymer stabilized displays. To this end, the medium according to the invention optionally comprises one or more polymerizable compounds of formula P
P-Sp-A1-(Z1-A2)z-R P in which the individual radicals, independently of each other and on each occurrence identically or differently, have the following meanings:
P a polymerizable group,
Sp a spacer group or a single bond,
A1, A2 an aromatic, heteroaromatic, alicyclic or heterocyclic group, preferably having 4 to 25 ring atoms, which may also contain fused rings, and which is unsubstituted, or mono- or polysubstituted by L,
Z1 -O-, -S-, -CO-, -CO-O-, -O-CO-, -O-CO-O-, -OCH2-, -CH2O-, -SCH2-, -CH2S-, -CF2O-, -OCF2-, -CF2S-, -SCF2-, -(CH2)n1-, -CF2CH2-, -CH2CF2-, -(CF2)n1-, -CH=CH-, -CF=CF-, -CH=CF-, -CF=CH-, -C ^C-, -CH=CH-CO-O-, -O-CO- CH=CH-, -CH2-CH2-CO-O-, -O-CO-CH2-CH2-, -CR0R00-, or a single bond, R0, R00 H or alkyl having 1 to 12 C atoms, R H, L, or P-Sp-, L F, Cl, -CN, P-Sp- or straight chain, branched or cyclic alkyl having 1 to 25 C atoms, wherein one or more non-adjacent CH2-groups are optionally replaced by -O-, -S-, -CO-, -CO-O-, -O-CO-, -O-CO-O- in such a manner that O- and/or S-atoms are not directly connected with each other, and wherein one or more H atoms are each optionally replaced by P-Sp-, F or Cl, z 0, 1, 2 or 3, n1 1, 2, 3 or 4. As used herein, the terms "active layer" and "switchable layer" mean a layer in an electrooptical display, for example an LC display, that comprises one or more molecules having structural and optical anisotropy, like for example LC molecules, which change their orientation upon an external stimulus like an electric or magnetic field, resulting in a change of the transmission of the layer for polarized or unpolarized light. As used herein, the terms "tilt" and "tilt angle" will be understood to mean a tilted alignment of the LC molecules of an LC medium relative to the surfaces of the cell in an LC display (here preferably a PSA display). The tilt angle here denotes the average angle (< 90°) between the longitudinal molecular axes of the LC molecules (LC director) and the surface of the plane-parallel outer plates which form the LC cell. A low value for the tilt angle (i.e. a large deviation from the 90° angle) corresponds to a large tilt here. A suitable method for measurement of the tilt angle is given in the examples. Unless indicated otherwise, tilt angle values disclosed above and below relate to this measurement method. As used herein, the terms "reactive mesogen" and "RM" will be understood to mean a compound containing a mesogenic or liquid crystalline skeleton, and one or more functional groups attached thereto which are suitable for polymerization and are also referred to as "polymerizable group" or "P".
Unless stated otherwise, the term "polymerizable compound" as used herein will be understood to mean a polymerizable monomeric compound.
As used herein, the term "low-molecular-weight compound" will be understood to mean to a compound that is monomeric and/or is not prepared by a polymerization reaction, as opposed to a "polymeric compound" or a "polymer".
As used herein, the term "unpolymerizable compound" will be understood to mean a compound that does not contain a functional group that is suitable for polymerization under the conditions usually applied for the polymerization of the RMs.
The term "mesogenic group" as used herein is known to the person skilled in the art and described in the literature, and means a group which, due to the anisotropy of its attracting and repelling interactions, essentially contributes to causing a liquid-crystal (LC) phase in low-molecular-weight or polymeric substances. Compounds containing mesogenic groups (mesogenic compounds) do not necessarily have to have an LC phase themselves. It is also possible for mesogenic compounds to exhibit LC phase behaviour only after mixing with other compounds and/or after polymerization. Typical mesogenic groups are, for example, rigid rod- or disc-shaped units. An overview of the terms and definitions used in connection with mesogenic or LC compounds is given in Pure Appl. Chem. 2001, 73(5), 888 and C. Tschierske, G. Pelzl, S. Diele, Angew. Chem. 2004, 116, 6340-6368.
As used herein, the terms “optically active” and “chiral” are synonyms for materials that are able to induce a helical pitch in a nematic host material, also referred to as “chiral dopants”.
The term "spacer group", hereinafter also referred to as "Sp", as used herein is known to the person skilled in the art and is described in the literature, see, for example, Pure Appl. Chem. 2001 , 73(5), 888 and C. Tschierske, G. Pelzl, S. Diele, Angew. Chem. 2004, 116, 6340-6368. As used herein, the terms "spacer group" or "spacer" mean a flexible group, for example an alkylene group, which connects the mesogenic group and the polymerizable group(s) in a polymerizable mesogenic compound.
Above and below,
denotes a trans-1 ,4-cyclohexylene ring.
In a group
the single bond shown between the two ring atoms can be attached to any free position of the benzene ring.
Above and below "organic group" denotes a carbon or hydrocarbon group.
"Carbon group" denotes a mono- or polyvalent organic group containing at least one carbon atom, where this either contains no further atoms (such as, for example, -C≡C-) or optionally contains one or more further atoms, such as, for example, N, O, S, B, P, Si, Se, As, Te or Ge (for example carbonyl, etc.). The term "hydrocarbon group" denotes a carbon group which additionally contains one or more H atoms and optionally one or more heteroatoms, such as, for example, N, O, S, B, P, Si, Se, As, Te or Ge.
"Halogen" denotes F, Cl, Br or I, preferably F or Cl.
A carbon or hydrocarbon group can be a saturated or unsaturated group. Unsaturated groups are, for example, aryl, alkenyl or alkynyl groups. A carbon or hydrocarbon radical having more than 3 C atoms can be straight-chain, branched and/or cyclic and may also contain spiro links or condensed rings.
The terms "alkyl", "aryl", "heteroaryl", etc., also encompass polyvalent groups, for example alkylene, arylene, heteroarylene, etc.
The term "aryl" denotes an aromatic carbon group or a group derived therefrom. The term "heteroaryl" denotes "aryl" as defined above, containing one or more heteroatoms, preferably selected from N, O, S, Se, Te, Si and Ge.
Preferred carbon and hydrocarbon groups are optionally substituted, straight-chain, branched or cyclic, alkyl, alkenyl, alkynyl, alkoxy, alkylcarbonyl, alkoxycarbonyl, alkylcarbonyloxy and alkoxycarbonyloxy having 1 to 40, preferably 1 to 20, very preferably 1 to 12, C atoms, optionally substituted aryl or aryloxy having 5 to 30,
preferably 6 to 25, C atoms, or optionally substituted alkylaryl, arylalkyl, alkylaryloxy, arylalkyloxy, arylcarbonyl, aryloxycarbonyl, arylcarbonyloxy and aryloxycarbonyloxy having 5 to 30, preferably 6 to 25, C atoms, wherein one or more C atoms may also be replaced by hetero atoms, preferably selected from N, O, S, Se, Te, Si and Ge. Further preferred carbon and hydrocarbon groups are C1-C20 alkyl, C2-C20 alkenyl, C2- C20 alkynyl, C3-C20 allyl, C4-C20 alkyldienyl, C4-C20 polyenyl, C6-C20 cycloalkyl, C4-C15 cycloalkenyl, C6-C30 aryl, C6-C30 alkylaryl, C6-C30 arylalkyl, C6-C30 alkylaryloxy, C6-C30 arylalkyloxy, C2-C30 heteroaryl, C2-C30 heteroaryloxy. Particular preference is given to C1-C12 alkyl, C2-C12 alkenyl, C2-C12 alkynyl, C6-C25 aryl and C2-C25 heteroaryl. Further preferred carbon and hydrocarbon groups are straight-chain, branched or cyclic alkyl having 1 to 20, preferably 1 to 12, C atoms, which are unsubstituted or mono- or polysubstituted by F, Cl, Br, I or CN and in which one or more non-adjacent CH2 groups may each be replaced, independently of one another, by -C(Rx)=C(Rx)-, - C≡-, -N(Rx)-, -O-, -S-, -CO-, -CO-O-, -O-CO-, -O-CO-O- in such a way that O and/or S atoms are not linked directly to one another. Rx preferably denotes H, F, Cl, CN, a straight-chain, branched or cyclic alkyl chain having 1 to 25 C atoms, in which, in addition, one or more non-adjacent C atoms may be replaced by -O-, -S-, -CO-, -CO-O-, -O-CO-, -O-CO-O- and in which one or more H atoms may be replaced by F or Cl, or denotes an optionally substituted aryl or aryloxy group with 6 to 30 C atoms, or an optionally substituted heteroaryl or heteroaryloxy group with 2 to 30 C atoms. Preferred alkyl groups are, for example, methyl, ethyl, n-propyl, isopropyl, n-butyl, isobutyl, s-butyl, t-butyl, 2-methylbutyl, n-pentyl, s-pentyl, cyclopentyl, n-hexyl, cyclohexyl, 2-ethylhexyl, n-heptyl, cycloheptyl, n-octyl, cyclooctyl, n-nonyl, n-decyl, n-undecyl, n-dodecyl, dodecanyl, trifluoromethyl, perfluoro-n-butyl, 2,2,2-trifluoroethyl, perfluorooctyl, perfluorohexyl, etc.
Preferred alkenyl groups are, for example, ethenyl, propenyl, butenyl, pentenyl, cyclopentenyl, hexenyl, cyclohexenyl, heptenyl, cycloheptenyl, octenyl, cyclooctenyl, etc.
Preferred alkynyl groups are, for example, ethynyl, propynyl, butynyl, pentynyl, hexynyl, octynyl, etc.
Preferred alkoxy groups are, for example, methoxy, ethoxy, 2-methoxyethoxy, n-propoxy, i-propoxy, n-butoxy, i-butoxy, s-butoxy, t-butoxy, 2-methylbutoxy, n-pentoxy, n-hexoxy, n-heptoxy, n-octoxy, n-nonoxy, n-decoxy, n-undecoxy, n-dodecoxy, etc.
Preferred amino groups are, for example, dimethylamino, methylamino, methylphenylamino, phenylamino, etc.
Aryl and heteroaryl groups can be monocyclic or polycyclic, i.e. they can contain one ring (such as, for example, phenyl) or two or more rings, which may also be fused (such as, for example, naphthyl) or covalently bonded (such as, for example, biphenyl), or contain a combination of fused and linked rings. Heteroaryl groups contain one or more heteroatoms, preferably selected from O, N, S and Se.
Particular preference is given to mono-, bi- or tricyclic aryl groups having 6 to 25 C atoms and mono-, bi- or tricyclic heteroaryl groups having 5 to 25 ring atoms, which optionally contain fused rings and are optionally substituted. Preference is furthermore given to 5-, 6- or 7-membered aryl and heteroaryl groups, in which, in addition, one or more CH groups may be replaced by N, S or O in such a way that O atoms and/or S atoms are not linked directly to one another.
Preferred aryl groups are, for example, phenyl, biphenyl, terphenyl, [1 ,1':3',1"]terphenyl-2'-yl, naphthyl, anthracene, binaphthyl, phenanthrene, 9,10- dihydro-phenanthrene, pyrene, dihydropyrene, chrysene, perylene, tetracene, pentacene, benzopyrene, fluorene, indene, indenofluorene, spirobifluorene, etc.
Preferred heteroaryl groups are, for example, 5-membered rings, such as pyrrole, pyrazole, imidazole, 1 ,2,3-triazole, 1 ,2,4-triazole, tetrazole, furan, thiophene, selenophene, oxazole, isoxazole, 1,2-thiazole, 1 ,3-thiazole, 1,2,3-oxadiazole,
1 ,2,4-oxadiazole, 1,2,5-oxadiazole, 1 ,3,4-oxadiazole, 1 ,2,3-thiadiazole, 1,2,4- thiadiazole, 1,2,5-thiadiazole, 1 ,3,4-thiadiazole, 6-membered rings, such as pyridine, pyridazine, pyrimidine, pyrazine, 1 ,3,5-triazine, 1 ,2,4-triazine, 1 ,2,3-triazine, 1 , 2,4,5- tetrazine, 1 ,2,3,4-tetrazine, 1 ,2,3,5-tetrazine, or condensed groups, such as indole, isoindole, indolizine, indazole, benzimidazole, benzotriazole, purine, naphthimidazole, phenanthrimidazole, pyridimidazole, pyrazinimidazole, quinoxalinimidazole, benzoxazole, naphthoxazole, anthroxazole, phenanthroxazole, isoxazole, benzothiazole, benzofuran, isobenzofuran, dibenzofuran, quinoline, isoquinoline, pteridine, benzo-5,6-quinoline, benzo-6,7-quinoline, benzo-7,8-quinoline, benzoisoquinoline, acridine, phenothiazine, phenoxazine, benzopyridazine, benzopyrimidine, quinoxaline, phenazine, naphthyridine, azacarbazole, benzocarboline, phenanthridine, phenanthroline, thieno[2,3b]thiophene, thieno[3,2b]thiophene, dithienothiophene, isobenzothiophene, dibenzothiophene, benzothiophene, benzothiadiazothiophene, or combinations of these groups.
The aryl and heteroaryl groups mentioned above and below may also be substituted by alkyl, alkoxy, thioalkyl, fluorine, fluoroalkyl or further aryl or heteroaryl groups.
The (non-aromatic) alicyclic and heterocyclic groups encompass both saturated rings, i.e. those containing exclusively single bonds, and also partially unsaturated rings, i.e. those which may also contain multiple bonds. Heterocyclic rings contain one or more heteroatoms, preferably selected from Si, O, N, S and Se.
The (non-aromatic) alicyclic and heterocyclic groups can be monocyclic, i.e. contain only one ring (such as, for example, cyclohexane), or polycyclic, i.e. contain a plurality of rings (such as, for example, decahydronaphthalene or bicyclooctane). Particular preference is given to saturated groups. Preference is furthermore given to mono-, bi- or tricyclic groups having 5 to 25 ring atoms, which optionally contain fused rings and are optionally substituted. Preference is furthermore given to 5-, 6-, 7- or 8-membered carbocyclic groups, in which, in addition, one or more C atoms may be replaced by Si and/or one or more CH groups may be replaced by N and/or one or more non- adjacent CH2 groups may be replaced by -O- and/or -S-.
Preferred alicyclic and heterocyclic groups are, for example, 5-membered groups, such as cyclopentane, tetrahydrofuran, tetrahydrothiofuran, pyrrolidine, 6-membered groups, such as cyclohexane, silinane, cyclohexene, tetrahydropyran,
tetrahydrothiopyran, 1 ,3-dioxane, 1 ,3-dithiane, piperidine, 7-membered groups, such as cycloheptane, and fused groups, such as tetrahydronaphthalene, decahydronaphthalene, indane, bicyclo[1.1.1]pentane-1 ,3-diyl, bicyclo[2.2.2]octane- 1 ,4-diyl, spiro[3.3]heptane-2,6-diyl, octahydro-4, 7-methanoindane-2,5-diyl.
Preferred substituents are, for example, solubility-promoting groups, such as alkyl or alkoxy, electron-withdrawing groups, such as fluorine, nitro or nitrile, or substituents for increasing the glass transition temperature (Tg) in the polymer, in particular bulky groups, such as, for example, t-butyl or optionally substituted aryl groups.
Preferred substituents, hereinafter also referred to as "Ls", are, for example, F, Cl, Br, I, -CN, -NO2, -NCO, -NCS, -OCN, -SCN, -C(=O)N(RX)2, -C(=O)Y1,
-C(=O)RX, -N(RX)2, straight-chain or branched alkyl, alkoxy, alkylcarbonyl, alkoxycarbonyl, alkylcarbonyloxy or alkoxycarbonyloxy each having 1 to 25 C atoms, in which one or more H atoms may optionally be replaced by F or Cl, optionally substituted silyl having 1 to 20 Si atoms, or optionally substituted aryl having 6 to 25, preferably 6 to 15, C atoms, wherein Rx denotes H, F, Cl, CN, or straight chain, branched or cyclic alkyl having 1 to 25 C atoms, wherein one or more non-adjacent CH2-groups are optionally replaced by -O-, -S-, -CO-, -CO-O-, -O-CO-, -O-CO-O- in such a manner that O- and/or S- atoms are not directly connected with each other, and wherein one or more H atoms are each optionally replaced by F, Cl, P- or P-Sp-, and
Y1 denotes halogen.
"Substituted silyl or aryl" preferably means substituted by halogen, -CN, R0, -OR0, -CO-R0, -CO-O-R0, -O-CO-R0 or -O-CO-O-R0, wherein R0 denotes H or alkyl with 1 to 20 C atoms.
Particularly preferred substituents L are, for example, F, Cl, CN, NO2, CH3, C2H5, OCH3, OC2H5, COCH3, COC2H5, COOCH3, COOC2H5, CF3, OCF3, OCHF2, OC2F5, furthermore phenyl.
A1 and A2 very preferably deno
te in which L has one of the meanings indicated above and r denotes 0, 1, 2, 3 or 4., in particular ,
or , The polymerizable group P is a group which is suitable for a polymerization reaction, such as, for example, free-radical or ionic chain polymerization, polyaddition or polycondensation, or for a polymer-analogous reaction, for example addition or condensation onto a main polymer chain. Particular preference is given to groups for chain polymerization, in particular those containing a C=C double bond or -C ^C- triple bond, and groups which are suitable for polymerization with ring opening, such as, for example, oxetane or epoxide groups. Preferred groups P are selected from the group consisting of CH2=CW 1 -CO-O-, CH2=CW 1 -C
, , CH2=CW2-(O)k3-, CW1=CH-CO-(O)k3-, CW1=CH-CO-NH-, CH2=CW1-CO-NH-, CH3-CH=CH-O-, (CH2=CH)2CH-OCO-, (CH2=CH-CH2)2CH-OCO-, (CH2=CH)2CH-O-, (CH2=CH- CH2)2N-, (CH2=CH-CH2)2N-CO-, HO-CW2W3-, HS-CW2W3-, HW2N-, HO-CW2W3-NH-, CH2=CW1-CO-NH-, CH2=CH-(COO)k1-Phe-(O)k2-, CH2=CH-(CO)k1-Phe-(O)k2-, Phe- CH=CH-, HOOC-, OCN- and W4W5W6Si-, in which W1 denotes H, F, Cl, CN, CF3, phenyl or alkyl having 1 to 5 C atoms, in particular H, F, Cl or CH3, W2 and W3 each, independently of one another, denote H or alkyl having 1 to 5 C atoms, in particular
H, methyl, ethyl or n-propyl, W4, W5 and W6 each, independently of one another, denote Cl, oxaalkyl or oxacarbonylalkyl having 1 to 5 C atoms, W7 and W8 each, independently of one another, denote H, Cl or alkyl having 1 to 5 C atoms, Phe denotes 1,4-phenylene, which is optionally substituted by one or more radicals L as defined above which are other than P-Sp-, k1, k2 and k3 each, independently of one another, denote 0 or 1, k3 preferably denotes 1, and k4 denotes an integer from 1 to 10. Very preferred groups P are selected from the group consisting of
C 2 C CO O , C 2 C CO , , ,
, CH2=CW2-O-, CH2=CW2-, CW1=CH-CO-(O)k3-, CW1=CH-CO-NH-, CH2=CW1-CO-NH-, (CH2=CH)2CH-OCO-, (CH2=CH-CH2)2CH-OCO-, (CH2=CH)2CH-O-, (CH2=CH-CH2)2N-, (CH2=CH-CH2)2N-CO-, CH2=CW1-CO-NH-, CH2=CH-(COO)k1-Phe-(O)k2-, CH2=CH-(CO)k1-Phe-(O)k2-, Phe-CH=CH- and W4W5W6Si-, in which W1 denotes H, F, Cl, CN, CF3, phenyl or alkyl having 1 to 5 C atoms, in particular H, F, Cl or CH3, W2 and W3 each, independently of one another, denote H or alkyl having 1 to 5 C atoms, in particular H, methyl, ethyl or n-propyl, W4, W5 and W6 each, independently of one another, denote Cl, oxaalkyl or oxa- carbonylalkyl having 1 to 5 C atoms, W7 and W8 each, independently of one another, denote H, Cl or alkyl having 1 to 5 C atoms, Phe denotes 1,4-phenylene, k1, k2 and k3 each, independently of one another, denote 0 or 1, k3 preferably denotes 1, and k4 denotes an integer from 1 to 10. Very particularly preferred groups P are selected from the group consisting of CH2=CW1-CO-O-, in particular CH2=CH-CO-O-, CH2=C(CH3)-CO-O- and CH2=CF- CO-O-, furthermore CH2=CH-O-, (CH2=CH)2CH-O-CO-, (CH2=CH)2CH-O-,
Further preferred polymerizable groups P are selected from the group consisting of vinyloxy, acrylate, methacrylate, fluoroacrylate, chloroacrylate, oxetane and epoxide, most preferably from acrylate and methacrylate. If the spacer group Sp is different from a single bond, it is preferably of the formula Sp"-X", so that the respective radical P-Sp- conforms to the formula R-Sp"-X"-, wherein Sp" denotes linear or branched alkylene having 1 to 20, preferably 1 to 12, C atoms, which is optionally mono- or polysubstituted by F, Cl, Br, I or CN and in which, in addition, one or more non-adjacent CH2 groups may each be replaced, independently of one another, by -O-, -S-, -NH-, -N(R0)-, -Si(R0R00)-, -CO-, -CO-O-, -O-CO-, -O-CO-O-, -S-CO-, -CO-S-, -N(R00)-CO-O-, -O-CO-N(R0)-, -N(R0)-CO-N(R00)-, -CH=CH- or -C ^C- in such a way that O and/or S atoms are not linked directly to one another, X" denotes -O-, -S-, -CO-, -CO-O-, -O-CO-, -O-CO-O-, -CO-N(R0)-, -N(R0)-CO-, -N(R0)-CO-N(R00)-, -OCH2-, -CH2O-, -SCH2-, -CH2S-, -CF2O-, -OCF2-, -CF2S-, -SCF2-, -CF2CH2-, -CH2CF2-, -CF2CF2-, -CH=N-, -N=CH-, -N=N-, -CH=CR0-, -CY2=CY3-, -C ^C-, -CH=CH-CO-O-, -O-CO-CH=CH- or a single bond, R0 and R00 each, independently of one another, denote H or alkyl having 1 to 20 C atoms, and Y2 and Y3 each, independently of one another, denote H, F, Cl or CN. X" is preferably -O-, -S-, -CO-, -COO-, -OCO-, -O-COO-, -CO-NR0-, -NR0-CO-, -NR0-CO-NR00- or a single bond. Typical spacer groups Sp and -Sp"-X"- are, for example, -(CH2)p1-, -(CH2)p1-O-, - (CH2)p1-O-CO-, -(CH2)p1-CO-O-, -(CH2)p1-O-CO-O-, -(CH2CH2O)q1-CH2CH2-, -CH2CH2- S-CH2CH2-, -CH2CH2-NH-CH2CH2- or -(SiR0R00-O)p1-, in which p1 is an integer from 1 to 12, q1 is an integer from 1 to 3, and R0 and R00 have the meanings indicated above.
Particularly preferred groups Sp and -Sp"-X"- are -(CH2)p1-, -(CH2)p1-O-, -(CH2)p1-O- CO-, -(CH2)p1-CO-O-, -(CH2)p1-O-CO-O-, in which p1 has the meanings indicated above. Particularly preferred groups Sp" are, in each case straight-chain, ethylene, propylene, butylene, pentylene, hexylene, heptylene, octylene, nonylene, decylene, undecylene, dodecylene, octadecylene, ethyleneoxyethylene, methyleneoxybutylene, ethylenethioethylene, ethylene-N-methyliminoethylene, 1-methylalkylene, ethenylene, propenylene and butenylene. In a preferred embodiment of the invention the compounds of formula P and its subformulae contain a spacer group Sp that is substituted by one or more polymerizable groups P, so that the group Sp-P corresponds to Sp(P)s, with s being ≥2 (branched polymerizable groups). Preferred compounds of formula P according to this preferred embodiment are those wherein s is 2, i.e. compounds which contain a group Sp(P)2. Very preferred compounds of formula P according to this preferred embodiment contain a group selected from the following formulae: -X-alkyl-CHPP S1 -X-alkyl-CH((CH2)aaP)((CH2)bbP) S2 -X-N((CH2)aaP)((CH2)bbP) S3 -X-alkyl-CHP-CH2-CH2P S4 -X-alkyl-C(CH2P)(CH2P)-CaaH2aa+1 S5 -X-alkyl-CHP-CH2P S6 -X-alkyl-CPP-CaaH2aa+1 S7 -X-alkyl-CHPCHP-CaaH2aa+1 S8 in which P is as defined in formula P, alkyl denotes a single bond or straight-chain or branched alkylene having 1 to 12 C atoms which is unsubstituted or mono- or polysubstituted by F, Cl or CN and in which one or more non-adjacent CH2 groups may each, independently of one another, be replaced by -C(R0)=C(R0)-, -C≡C-, -N(R0)-, -O-, -S-, -CO-, -CO-O-, -O- CO-, -O-CO-O- in such a way that O and/or S atoms are not linked directly to one another, where R0 has the meaning indicated above,
aa and bb each, independently of one another, denote 0, 1, 2, 3, 4, 5 or 6, X has one of the meanings indicated for X", and is preferably O, CO, SO2, O-CO-, CO-O or a single bond. Preferred spacer groups Sp(P)2 are selected from formulae S1, S2 and S3. Very preferred spacer groups Sp(P)2 are selected from the following subformulae: -CHPP S1a -O-CHPP S1b -CH2-CHPP S1c -OCH2-CHPP S1d -CH(CH2-P)(CH2-P) S2a -OCH(CH2-P)(CH2-P) S2b -CH2-CH(CH2-P)(CH2-P) S2c -OCH2-CH(CH2-P)(CH2-P) S2d -CO-NH((CH2)2P)((CH2)2P) S3a In the compounds of formula P and its subformulae as described above and below, P is preferably selected from the group consisting of vinyloxy, acrylate, methacrylate, fluoroacrylate, chloroacrylate, oxetane and epoxide, most preferably from acrylate and methacrylate. Further preferred are compounds of formula P and its subformulae as described above and below, wherein all polymerizable groups P that are present in the compound have the same meaning, and very preferably denote acrylate or methacrylate, most preferably methacrylate. In the compounds of formula P and its subformulae as described above and below, R preferably denotes P-Sp-. Further preferred are compounds of formula P and its subformulae as described above and below, wherein Sp denotes a single bond or -(CH2)p1-, -O-(CH2)p1-, -O-CO- (CH2)p1, or -CO-O-(CH2)p1, wherein p1 is 2, 3, 4, 5 or 6, and, if Sp is -O-(CH2)p1-, -O- CO-(CH2)p1 or -CO-O-(CH2)p1 the O-atom or CO-group, respectively, is linked to the benzene ring.
Further preferred are compounds of formula P and its subformulae as described above and below, wherein at least one group Sp is a single bond. Further preferred are compounds of formula P and its subformulae as described above and below, wherein at least one group Sp is different from a single bond, and is preferably selected from -(CH2)p1-, -O-(CH2)p1-, -O-CO-(CH2)p1, or -CO-O-(CH2)p1, wherein p1 is 2, 3, 4, 5 or 6, and, if Sp is -O-(CH2)p1-, -O-CO-(CH2)p1 or -CO-O-(CH2)p1 the O-atom or CO-group, respectively, is linked to the benzene ring. Very preferred groups -A1-(Z-A2)z- in formula P are selected from the following formulae
wherein at least one benzene ring is substituted by at least one group L and the benzene rings are optionally further substituted by one or more groups L or P-Sp-.
Preferred compounds of formula P and their subformulae are selected from the following preferred embodiments, including any combination thereof:
- All groups P in the compound have the same meaning,
- -A1-(Z-A2)z- is selected from formulae A1, A2 and A5,
- the compounds contain exactly two polymerizable groups (represented by the groups P),
- the compounds contain exactly three polymerizable groups (represented by the groups P),
- P is selected from the group consisting of acrylate, methacrylate and oxetane, very preferably acrylate or methacrylate,
- P is methacrylate,
- all groups Sp are a single bond,
- at least one of the groups Sp is a single bond and at least one of the groups Sp is different from a single bond,
- Sp, when being different from a single bond, is -(CH2)P2-, -(CH2)P2-O-,
-(CH2)P2-CO-O-, -(CH2)P2-O-CO-, wherein p2 is 2, 3, 4, 5 or 6, and the O- atom or the CO-group, respectively, is connected to the benzene ring,
- Sp is a single bond or denotes -(CH2)P2-, -(CH2)P2-O-, -(CH2)P2-CO-O-, -(CH2)P2-O- CO-, wherein p2 is 2, 3, 4, 5 or 6, and the O-atom or the CO-group, respectively, is connected to the benzene ring,
- R denotes P-Sp-,
- R does not denote or contain a polymerizable group,
- R does not denote or contain a polymerizable group and denotes straight chain, branched or cyclic alkyl having 1 to 25 C atoms, wherein one or more non-adjacent CH2-groups are optionally replaced by -O-,
-S-, -CO-, -CO-O-, -O-CO-, -O-CO-O- in such a manner that O- and/or S- atoms are not directly connected with each other, and wherein one or more H atoms are each optionally replaced by F, Cl or L,
- L or L’ denote F, Cl or CN,
- L is F.
in which the individual radicals have the following meanings: P1, P2 and P3 each, independently of one another, denote an acrylate or methacrylate group, Sp1, Sp2 and Sp3 each, independently of one another, denote a single bond or a spacer group having one of the meanings indicated above and below for Sp, and particularly preferably denote -(CH2)p1-, -(CH2)p1-O-, -(CH2)p1-CO-O-, -(CH2)p1-O-CO- or -(CH2)p1-O-CO-O-, in which p1 is an integer from 1 to 12, where, in addition, one or more of
the radicals P1-Sp1-, P2-Sp2- and P3-Sp3- may denote Raa, with the proviso that at least one of the radicals
P1-Sp1-, P2-Sp2- and P3-Sp3- present is different from Raa,
Raa denotes H, F, Cl, CN or straight-chain or branched alkyl having 1 to 25 C atoms, in which, in addition, one or more non-adjacent CH2 groups may each be replaced, independently of one another, by C(R0)=C(R00)-, -C=C-, -N(R0)-, -O-, -S-, -CO-,
-CO-O-, -O-CO-, -O-CO-O- in such a way that O and/or S atoms are not linked directly to one another, and in which, in addition, one or more H atoms may be replaced by F, Cl, CN or P1-Sp1-, particularly preferably straight-chain or branched, optionally mono- or polyfluorinated alkyl, alkoxy, alkenyl, alkynyl, alkylcarbonyl, alkoxycarbonyl, alkylcarbonyloxy or alkoxycarbonyloxy having 1 to 12 C atoms (where the alkenyl and alkynyl radicals have at least two C atoms and the branched radicals have at least three C atoms),
R0, R00 each, independently of one another and identically or differently on each occurrence, denote H or alkyl having 1 to 12 C atoms,
Ry and Rz each, independently of one another, denote H, F, CH3 or CF3,
X1, X2 and X3 each, independently of one another, denote -CO-O-, -O-CO- or a single bond,
Z1 denotes -O-, -CO-, -C(RyRz)- or -CF2CF2-,
Z2 and Z3 each, independently of one another, denote -CO-O-, -O-CO-, -CH2O-, -OCH2-, -CF2O-, -OCF2- or -(CH2)n-, where n is 2, 3 or 4,
L on each occurrence, identically or differently, denotes F, Cl, CN or straight-chain or branched, optionally mono- or polyfluorinated alkyl, alkoxy, alkenyl, alkynyl, alkylcarbonyl, alkoxycarbonyl, alkylcarbonyloxy or alkoxycarbonyloxy having 1 to 12 C atoms, preferably F,
L' and L" each, independently of one another, denote H, F or Cl, k denotes 0 or 1 , r denotes 0, 1 , 2, 3 or 4, s denotes 0, 1 , 2 or 3,
t denotes 0, 1 or 2, x denotes 0 or 1. Especially preferred are compounds of formulae P2, P13, P17, P22, P23, P24, P30, P31 and P32. Further preferred are trireactive compounds P15 to P30, in particular P17, P18, P19, P22, P23, P24, P25, P26, P30, P31 and P32. In the compounds of formulae P1 to P32 the group ,
wherein L on each occurrence, identically or differently, has one of the meanings given above or below, and is preferably F, Cl, CN, NO2, CH3, C2H5, C(CH3)3, CH(CH3)2, CH2CH(CH3)C2H5, OCH3, OC2H5, COCH3, COC2H5, COOCH3, COOC2H5, CF3, OCF3, OCHF2, OC2F5 or P-Sp-, very preferably F, Cl, CN, CH3, C2H5, OCH3, COCH3, OCF3 or P-Sp-, more preferably F, Cl, CH3, OCH3, COCH3 oder OCF3 , especially F or CH3. The media according to the present invention comprise one or more chiral dopants. Preferably these chiral dopants have an absolute value of the helical twisting power (HTP) in the range of from 1 μm-1 to 150 μm-1, preferably in the range of from 10 μm-1 to 100 μm-1. In case the media comprise two or more chiral dopants, these may have opposite signs of their HTP-values. This condition is preferred for some specific embodiments, as it allows to compensate the chirality of the respective compounds to some degree and, thus, may be used to compensate various temperature dependent properties of the resulting media in the devices. Generally, however, it is preferred that
most, preferably all of the chiral compounds present in the media according to the present invention have the same sign of their HTP-values.
Preferably the chiral dopants present in the media according to the instant application are mesogenic compounds and most preferably they exhibit a mesophase on their own.
In a preferred embodiment of the present invention, the medium comprises two or more chiral compounds which all have the same algebraic sign of the HTP.
The temperature dependence of the HTP of the individual compounds may be high or low. The temperature dependence of the pitch of the medium can be compensated by mixing compounds having different temperature dependencies of the HTP in corresponding ratios.
For the optically active component, a multitude of chiral dopants, some of which are commercially available, is available to the person skilled in the art, such as, for example, cholesteryl nonanoate, R- and S-811, R- and S-1011 , R- and S-2011, R- and S-3011, R- and S-4011, or CB15 (all Merck KGaA, Darmstadt).
Particularly suitable dopants are compounds which contain one or more chiral groups and one or more mesogenic groups, or one or more aromatic or alicyclic groups which form a mesogenic group with the chiral group.
Suitable chiral groups are, for example, chiral branched hydrocarbon radicals, chiral ethane diols, binaphthols or dioxolanes, furthermore mono- or polyvalent chiral groups selected from the group consisting of sugar derivatives, sugar alcohols, sugar acids, lactic acids, chiral substituted glycols, steroid derivatives, terpene derivatives, amino acids or sequences of a few, preferably 1-5, amino acids.
Preferred chiral groups are sugar derivatives, such as glucose, mannose, galactose, fructose, arabinose and dextrose; sugar alcohols, such as, for example, sorbitol, mannitol, iditol, galactitol or anhydro derivatives thereof, in particular dianhydrohexitols, such as dianhydrosorbide (1 ,4:3,6-dianhydro-D-sorbide, isosorbide), dianhydromannitol (isosorbitol) or dianhydroiditol (isoiditol); sugar acids, such as, for example, gluconic acid, gulonic acid and ketogulonic acid; chiral substituted glycol radicals, such as, for example, mono- or oligoethylene or propylene
glycols, in which one or more CH2 groups are substituted by alkyl or alkoxy; amino acids, such as, for example, alanine, valine, phenylglycine or phenylalanine, or sequences of from 1 to 5 of these amino acids; steroid derivatives, such as, for example, cholesteryl or cholic acid radicals; terpene derivatives, such as, for example, menthyl, neomenthyl, campheyl, pineyl, terpineyl, isolongifolyl, fenchyl, carreyl, myrthenyl, nopyl, geraniyl, linaloyl, neryl, citronellyl or dihydrocitronel lyl .
The media according to the present invention preferably comprise chiral dopants which are selected from the group of known chiral dopants. Suitable chiral groups and mesogenic chiral compounds are described, for example, in DE 3425 503, DE 35 34 777, DE 35 34 778, DE 35 34 779 and DE 35 34 780, DE 4342 280, EP 01 038 941 and DE 19541 820. Examples are also compounds listed in Table F below.
Chiral compounds preferably used according to the present invention are selected from the group consisting of the formulae shown below.
Particular preference is given to chiral dopants selected from the group consisting of compounds of the following formulae A-l to A-lll and A-Ch:
in which
Ra11, Ra12 and Rb12, independently of one another, denote alkyl having 1 to 15 C atoms, in which, in addition, one or more non-adjacent CH2 groups may each be replaced, independently of one another, by -C(RZ)=C(RZ)-, -C=C-, -O-, -S-, -CO-, -CO-O-, -O-CO- or -O-CO-O- in such a way that O and/or S atoms are not linked directly to one another, and in which, in addition, one or more H atoms may each be replaced by F, Cl, Br, I or CN, preferably alkyl, more preferably n-alkyl, with the proviso that Ra12 is different from Rb12
Ra21 and Ra22, independently of one another, denote alkyl having 1 to 15 C atoms, in which, in addition, one or more non-adjacent CH2 groups may each be replaced, independently of one another, by -C(RZ)=C(RZ)-, -C≡C-, -O-, -
S-, -CO-, -CO-O-, -O-CO- or -O-CO-O- in such a way that O and/or S atoms are not linked directly to one another, and in which, in addition, one or more H atoms may be replaced by F, Cl, Br, I or CN, preferably both are alkyl, more preferably n-alkyl,
Ra31, Ra32and Rb32, independently of one another, denote straight-chain or branched alkyl having 1 to 15 C atoms, in which, in addition, one or more non- adjacent CH2 groups may each be replaced, independently of one another, by -C(RZ)=C(RZ)-, -C=C-, -O-, -S-, -CO-, -CO-O-, -O-CO- or -O-CO-O- in such a way that O and/or S atoms are not linked directly to one another, and in which, in addition, one or more H atoms may be replaced by F, Cl, Br, I or CN, preferably alkyl, more preferably n-alkyl, with the proviso that Ra32 is different from Rb32;
Rz denotes H, CH3, F, Cl, or CN, preferably H or F,
R8 has one of the meanings of Ra11 given above, preferably alkyl, more preferably n-alkyl having 1 to 15 C atoms,
Z8 denotes- C(O)O-, CH2O, CF2O or a single bond, preferably -C(O)O-,
L12 on each occurrence, independently of one another, denotes halogen, CN, or alkyl, alkenyl, alkoxy or alkenyloxy having up to 12 C atoms and in which one or more H atoms are optionally replaced with halogen, preferably methyl, ethyl, Cl or F, particularly preferably F,
A22 has the meanings given for A12
A32 has the meanings given for A12. n2 on each occurrence, identically or differently, is 0, 1 or 2, and n3 is 1 , 2 or 3, and r is 0, 1 , 2, 3 or 4.
Particular preference is given to dopants selected from the group consisting of the compounds of the following formulae:
in which m is, on each occurrence, identically or differently, an integer from 1 to 9 and n is, on each occurrence, identically or differently, an integer from 2 to 9.
Particularly preferred compounds of formula A are compounds of formula A-lll.
Further preferred dopants are derivatives of the isosorbide, isomannitol or isoiditol of the following formula A-IV:
(dianhydrosorbitol),
(dianhydromannitol), or
(dianhydroiditol),
preferably dianhydrosorbitol,
and chiral ethane diols, such as, for example, diphenylethanediol (hydrobenzoin), in particular mesogenic hydrobenzoin derivatives of the following formula A-V:
including the (S,S) enantiomers, which are not shown, in which
are each, independently of one another, 1 ,4-phenylene, which may also be mono-, di- or trisubstituted by L, or 1,4-cyclo- hexylene,
L is H, F, Cl, CN or optionally halogenated alkyl, alkoxy, alkylcarbonyl, alkoxycarbonyl or alkoxycarbonyloxy having 1-7 carbon atoms, c is O or 1,
X is CH2 or -C(O)-,
Z0 is -COO-, -OCO-, -CH2CH2- or a single bond, and
R0 is alkyl, alkoxy, alkylcarbonyl, alkoxycarbonyl or alkylcarbonyloxy having
1-12 carbon atoms.
The compounds of the formula A-IV are described in WO 98/00428. The compounds of the formula A-V are described in GB-A-2,328,207.
Very particularly preferred dopants are chiral binaphthyl derivatives, as described in WO 02/94805, chiral binaphthol acetal derivatives, as described in WO 02/34739, chiral TADDOL derivatives, as described in WO 02/06265, and chiral dopants having at least one fluorinated bridging group and a terminal or central chiral group, as described in WO 02/06196 and WO 02/06195.
X1, X2, Y1 and Y2 are each, independently of one another, F, Cl, Br, I, CN, SCN, SF5, straight-chain or branched alkyl having from 1 to 25 carbon atoms, which is unsubstituted or monosubstituted or polysubstituted by F, Cl, Br, I or CN and in which, in addition, one or more non-adjacent CH2 groups may each, independently of one another, be replaced by -O-, -S-, -NH-, NRX-, -CO-, -COO-, -OCO-, -OCOO-, -S-CO-, -CO-S-, -CH=CH- or -C^C- in such a way that O and/or S atoms are not bonded directly to one
another, a polymerizable group or cycloalkyl or aryl having up to 20 carbon atoms, which may optionally be monosubstituted or polysubstituted by halogen, preferably F, or by a polymerizable group, x1 and x2 are each, independently of one another, 0, 1 or 2, y1 and y2 are each, independently of one another, 0, 1 , 2, 3 or 4,
B1 and B2 are each, independently of one another, an aromatic or partially or fully saturated aliphatic six-membered ring in which one or more CH groups may each be replaced by N and one or more non-adjacent CH2 groups may each be replaced by O or S,
W1 and W2 are each, independently of one another, -Z1-A1-(Z2-A2)m-R, and one of the two is alternatively R1 or A3, but both are not simultaneously H, or
II1 and II2 are each, independently of one another, CH2, O, S, CO or CS,
V1 and V2 are each, independently of one another, (CH2)n, in which from one to four non-adjacent CH2 groups may each be replaced by O or S, and one of V1 and V2 and, in the case where
both are a single bond, n is 1 ,2 or 3
Z1 and Z2 are each, independently of one another, -O-, -S-, -CO-, -COO-, -OCO-, -O-COO-, -CO-NRX-, -NRX-
CO-, -O-CH2-, -CH2-O-,
-S-CH2-, -CH2-S-, -CF2-O-, -O-CF2-, -CF2-S-, -S-CF2-, -CH2-CH2-, -CF2-CH2-, -CH2-CF2-, -CF2-
CF2-, -CH=N-, -N=CH-, -N=N-, -CH=CH-, -CF=CH-, -CH=CF-, -CF=CF-, -C=C-, a combination of two of these groups, where no two O and/or S and/or N atoms are bonded directly to one another, preferably -CH=CH- COO-, or -COO-CH=CH-, or a single bond,
Rx denotes alkyl having 1 to 6 C atoms,
A1, A2 and A3 are each, independently of one another, 1 ,4-phenylene, in which one or two non-adjacent CH groups may each be replaced by N, 1,4- cyclohexylene, in which one or two non-adjacent CH2 groups may each be replaced by O or S, 1,3-dioxolane-4,5-diyl, 1 ,4-cyclohexenylene, 1 ,4- bicyclo[2.2.2]octylene, piperidine- 1 ,4-diyl, naphthalene-2,6-diyl, decahydronaphthalene-2,6-diyl or 1 ,2,3,4-tetrahydronaphthalene-2,6-diyl, where each of these groups may be monosubstituted or polysubstituted by L, and in addition A1 can be a single bond,
L is a halogen atom, preferably F, CN, NO2, alkyl, alkoxy, alkylcarbonyl, alkoxycarbonyl or alkoxycarbonyloxy having 1-7 carbon atoms, in which one or more H atoms may each be replaced by F or Cl, m is in each case, independently, 0, 1, 2 or 3, and
R and R1 are each, independently of one another, H, F, Cl, Br, I, CN, SCN, SF5, straight-chain or branched alkyl having from 1 or 3 to 25 carbon atoms respectively, which may optionally be monosubstituted or polysubstituted by F, Cl, Br, I or CN, and in which one or more non-adjacent CH2 groups may each be replaced by -O-, -S-, -NH-, -NR0-, -CO-, -COO-, -OCO-, -O- COO-, -S-CO-, -CO-S-, -CH=CH- or -C=C-, where no two O and/or S atoms are bonded directly to one another, or a polymerizable group.
Particular preference is given to chiral binaphthyl derivatives of the formula A-VI-1
in which ring B, R0 and Z0 are as defined for the formulae A-IV and A-V, and b is 0, 1 , or 2, in particular those selected from the following formulae A-VI-1a to A-VI-1c:
in which ring B, R0’ and Z0 are as defined for the formula A-VI-1 , and
R0 as defined for formula A-IV or H or alkyl having from 1 to 4 carbon atoms, Z0 is, in particular, -OC(O)- or a single bond.
The concentration of the one or more chiral dopant(s), in the LC medium is preferably in the range from 0.001 % to 20 %, preferably from 0.05 % to 5 %, more preferably from 0.1 % to 2 %, and, most preferably from 0.5 % to 1.5 %. These preferred concentration ranges apply in particular to the chiral dopant S-4011 or R-4011 (both from Merck KGaA) and for chiral dopants having the same or a similar HTP. For Chiral dopants having either a higher or a lower absolute value of the HTP compared to S-4011 these preferred concentrations have to be decreased, respectively
increased proportionally according to the ratio of their HTP values relatively to that of S-4011.
The pitch p of the LC media or host mixtures according to the invention is preferably in the range of from 5 to 50 .m, more preferably from 8 to 30 .m and particularly preferably from 10 to 20 .m.
Preferably, the media according to the invention, comprise a stabilizer selected from the group of compounds of the formulae ST-1 to ST- 18:
RST denotes H, an alkyl or alkoxy radical having 1 to 15 C atoms, where, in addition, one or more CH2 groups in these radicals may each be replaced, independently
-O-, -CO-O-, or -O-CO- in such a way that O atoms are not linked directly to one another, and in which, in addition, one or more H atoms may be replaced by halogen,
ZST each, independently of one another, denote -CO-O-, -O-CO-, -CF2O-, -OCF2-, -CH2O-, -OCH2-, -CH2-, -CH2CH2-, -(CH2)4-, -CH=CH-CH2O-, -C2F4-, -CH2CF2-, -CF2CH2-, -CF=CF-, -CH=CF-, -CF=CH-, -CH=CH-, -C=C- or a single bond,
L1 and L2 each, independently of one another, denote F, Cl, CF3 or CHF2, p denotes 1 or 2, and q denotes 1 , 2, 3, 4, 5, 6, 7, 8, 9 or 10.
The medium according to the invention preferably comprises one or more compounds of the formula ST-3.
Of the compounds of the formula ST, special preference is given to the compounds of the formulae
in which n = 1 , 2, 3, 4, 5, 6 or 7, preferably n = 3
In the compounds of the formulae ST-3a and ST-3b, n preferably denotes 3. In the compounds of the formula ST-2a, n preferably denotes 7.
Very particularly preferred mixtures according to the invention comprise one or more stabilizers from the group of the compounds of the formulae ST-2a-1 , ST-3a-1 , ST-
Ar denotes a methylene group or an aromatic hydrocarbon group having 6 to 40
C atoms or a heteroaromatic hydrocarbon group having 4 to 40 C atoms; preferably an aromatic hydrocarbon group having 6 to 40 C atoms;
Sp denotes a spacer group;
Rs denotes H, alkyl having 1 to 12 C atoms or alkenyl having 2 to 12 C atoms;
RH denotes H, O’, CH3, OH or ORS;
RS1, RS2, RS3 and RS4, identically or differently, denote alkyl having 1 to 6 C atoms, preferably having 1 to 3 C atoms, very preferably CH3;
G denotes H or Rs or a group ZS-HA; z is an integer from 1 to 6, and q is 2, 3 or 4, preferably 3 or 4.
In formula S, aryl denotes an aromatic or heteroaromatic hydrocarbon group having 4 to 40 C atoms, comprising one, two, three or four aromatic rings including condensed rings that may be linked directly or via an alkylene linking group having 1 to 12 C atoms, in which one or more H atoms are each optionally replaced with alkyl or alkoxy having 1 to 6 C atoms or alkenyl having 2 to 6 C atoms, or with CN, CF3 or halogen, and in which one or more CH2 groups may each, independently of one another, be
replaced by -O-, -S-, -NH-, -N(C1-C4-alkyl)-, -CO-, -CO-O-, -O-CO-, -O-CO-O-, -CH=CH- or -C ^C- in such a way that O or S atoms are not linked directly to one another. Preferred aryl groups are benzene, naphthalene, anthracene, biphenyl, m-terphenyl, p-terphenyl, and (phenylalkyl)benzene in which alkyl is straight chain alkyl having 1 to 12 C atoms. In a preferred embodiment, the medium according to the invention comprises a compound of the formula S in which the parameter q is 3 and G denotes a group ZS-HA. In another preferred embodiment, the medium according to the invention comprises a compound of the formula S in which the parameter q is 4 and G denotes H or RS. The compounds of formula S are preferably selected from the compounds of the formulae S-1 S-2 and S-3:
in which RH has the meanings given above and preferably denotes H or O, Sp on each occurrence, identically or differently, denotes a spacer group, and W denotes linear or branched, optionally unsaturated alkylene having 1 to 12 C atoms, in which one or more non-adjacent -CH2- groups may each be replaced with - O-.
Preferred compounds of formula S-1 are selected from the compounds of the formula
in which RH has the meanings given above and preferably denotes H or O, and n is an integer from 0 to 12, preferably 5, 6, 7, 8 or 9, very preferably 7.
Preferred compounds of formula S-2 are selected from the compounds of the formula
in which RH has the meanings given above and preferably denotes H or O, and n2, on each occurrence identically or differently, preferably identically, is an integer from 1 to 12, preferably 2, 3, 4, 5, or 6, very preferably 3, and Rs on each occurrence identically or differently, preferably identically, denotes alkyl having 1 to 6 C atoms, preferably n-butyl.
Preferred compounds of formula S-3 are selected from the compounds of the formula
in which RH has the meanings given above and preferably denotes H or O, and n is an integer from 0 to 12, preferably 5, 6, 7, 8 or 9, very preferably 7.
The compounds of the formulae ST-1 to ST-19 and S are preferably each present in the liquid-crystal mixtures according to the invention in amounts of 0.005 - 0.5%, based on the mixture.
If the mixtures according to the invention comprise two or more compounds from the group of the compounds of the formulae ST-1 to ST- 18, the concentration
correspondingly increases to 0.01 - 1 % in the case of two compounds, based on the mixtures.
However, the total proportion of the compounds of the formulae ST-1 to ST-18, based on the mixture according to the invention, should not exceed 2%.
The mixtures according to the invention preferably comprise
- one or more compounds of formula I, selected from the compounds of the formulae 1-1 to 1-16, very preferably 1-1 to I-5, preferably in a total concentration in the range of from 5% to 40%, more preferably, from 10 % to 39% still more preferably from 13% to 38%, particularly preferably from 15% to 38%, in particular from 21% to 35%; and/or
- one or more compounds of formula IIA, preferably in a total concentration in the range of from 5% to 30%, more preferably from 8% to 25%, particularly preferably from 10% to 18%; and/or
- one or more compounds of formula IID, preferably in a total concentration in the range of from 1% to 20%, more preferably from 3% to 18%, particularly preferably from 5% to 15%; and/or one or more compounds of the formula I and one or more compounds selected from the compounds of the formulae IIA, IIB, IIC and IID in a total concentration in the range of from 30% to 70%, more preferably from 35% to 55%, very preferably from 40% to 60%; and/or one or more compounds of the formulae I and IIA and IID in a total concentration in the range of from 30% to 70%, more preferably from 35% to 55%, very preferably from 40% to 60%; and/or
- one or more compounds of formula IV, preferably in a total concentration in the range of from 20% to 55%, more preferably from 25 % to 50%, particularly preferably from 30% to 40%; and/or
- one or more compounds of formula V, preferably in a total concentration in the range of from 1% to 30%, more preferably from 5 % to 25%, particularly preferably from 10% to 20%; and/or one or more compounds of the formula S and one or more compounds selected from the group of compounds of the formulae ST-1 to ST-18, preferably of the formula S-2- 1 and ST-3, very preferably of the formulae S-2-1a and ST-3b.
In particular, the medium comprises
- one or more compounds CY-n-Om, in particular CY-3-04, CY-5-04 and/or CY-3- 02, preferably in a total concentration in the range of from 0.5% to 10%, preferably 1% to 8%, very preferably 2 to 5%; and/or
- one or more compounds CCY-n-Om, preferably CCY-4-02, CCY-3-02, CCY-3-03, CCY-3-O1 and/or CCY-5-02, preferably in concentrations > 3 %, in particular in total 5 to 15 %, and/or
- CLY-n-Om, preferably CLY-2-04, CLY-3-02 and/or CLY-3-03, preferably in concentrations > 3 %, very preferably in total 5 to 20 %, very preferably 7 to 15% and/or
- CLY-n-Om and CY-n-Om, preferably in a total concentration of 5 to 25 %, more preferably from 8% to 20%; and/or
- CC-V-V, preferably in concentrations of 5 to 50 % and/or
- the compound(s) of the formula CC-3-V1 and/or CC-4-V1 , in a total concentration in the range of from 10 to 35%, more preferably from 15% to 32%, particularly preferably from 18% to 28%, and/or
- 0.1% to 3% of the compound PPGU-3-F and/or
- >0% to 10% of the compound PGIY-nO-Om.
Preferably, the medium according to the invention comprises one or more compounds of the formula I and one or more compounds selected from the compounds of the formulae IIA, IIB, IIC and IID, in which R1, R2A, R2B, R2C and R2D denote n-propyl, (O) denotes O, and v is 2, preferably in a total concentration in the range of from 30% to 70%, more preferably from 35% to 55%, very preferably from 40% to 60%.
The invention furthermore relates to an electro-optical display having active-matrix addressing, characterised in that it contains, as dielectric, a liquid-crystalline medium according to claim 1 and wherein the display is a VA, SA-VA, IPS, U-IPS, FFS, UB- FFS, SA-FFS, PS-VA, PS-OCB, PS-IPS, PS-FFS, PS-UB-FFS, PS-posi-VA, PS-TN, polymer stabilized SA-VA or polymer stabilized SA-FFS display.
It is advantageous for the liquid-crystalline medium according to the invention to preferably have a nematic phase from ≤ -20°C to ≥ 70°C, particularly preferably from ≤ -30°C to ≥ 80°C, very particularly preferably from ≤ -40°C to ≥ 90°C.
The medium according to the invention has a clearing temperature of 100°C or more, preferably of 105°C or more, more preferably of 110°C or more, very preferably of 115°C or more
The expression "have a nematic phase" here means on the one hand that no smectic phase and no crystallisation are observed at low temperatures at the corresponding temperature and on the other hand that clearing still does not occur on heating from the nematic phase. The investigation at low temperatures is carried out in a flow viscometer at the corresponding temperature and checked by storage in test cells having a layer thickness corresponding to the electro-optical use for at least 100
hours. If the storage stability at a temperature of -20°C in a corresponding test cell is 1000 h or more, the medium is referred to as stable at this temperature. At tempera- tures of -30°C and -40°C, the corresponding times are 500 h and 250 h respectively. At high temperatures, the clearing point is measured by conventional methods in capillaries.
The liquid-crystal mixture preferably has a nematic phase range of at least 60 K and a flow viscosity V20 of at most 30 mm2 • s-1 at 20°C.
The mixture is nematic at a temperature of -20°C or less, preferably at -30°C or less, very preferably at -40°C or less.
In a preferred embodiment of the present invention, the medium has a birefringence in the range of from 0.075 to 0.110, preferably from 0.080 to 0.108, more preferably from 0.085 to 0.105, in particular from 0.088 to 0.100.
The liquid-crystal mixture according to the invention has a dielectric anisotropy Δε of -1.5 to -8.0, preferably of -3.0 to -5.0, in particular -3.3 to -3.7.
The rotational viscosity γ1 at 20°C is preferably < 250 mPa-s, in particular ≤ 200 mPa-s.
The medium according to the invention has an average elastic constant Kavg of 20 or more, preferably 21 or more, very preferably 21.5 or more.
The medium according to the invention has a value γ1/K1 of 8.0 or 7.9 or 7.8 or 7.7 or less, preferably 7.6 or less.
In addition, the liquid-crystal media according to the invention have high values for the voltage holding ratio in liquid-crystal cells.
In general, liquid-crystal media having a low addressing voltage or threshold voltage exhibit a lower voltage holding ratio than those having a higher addressing voltage or threshold voltage and vice versa.
For the present invention, the term "dielectrically positive compounds" denotes compounds having a Δε > 1.5, the term "dielectrically neutral compounds" denotes those having -1 .5 < Δε < 1 .5 and the term "dielectrically negative compounds” denotes those having Δε < -1.5. The dielectric anisotropy of the compounds is determined here by dissolving 10 % of the compounds in a liquid-crystalline host and determining the capacitance of the resultant mixture in at least one test cell in each case having a layer thickness of 20 μm with homeotropic and with homogeneous surface alignment at 1 kHz. The measurement voltage is typically 0.5 V to 1 .0 V, but is always lower than the capacitive threshold of the respective liquid-crystal mixture investigated.
All temperature values indicated for the present invention are in °C.
The mixtures according to the invention are suitable for all VA-TFT applications, such as, for example, VAN, MVA, (S)-PVA, ASV, PSA (polymer sustained VA) and PS-VA (polymer stabilized VA). They are furthermore suitable for IPS (in-plane switching) and FFS (fringe field switching) applications having negative Δε.
The nematic liquid-crystal mixtures in the displays according to the invention generally comprise two components A and B, which themselves consist of one or more individual compounds.
Component A has significantly negative dielectric anisotropy and gives the nematic phase a dielectric anisotropy of < -0.5. Besides one or more compounds of the formula I, it preferably comprises the compounds of the formulae IIA, IIB and/or IIC, furthermore one or more compounds of the formula IV-1.
The proportion of component A is preferably between 45 and 100 %, in particular between 60 and 85 %.
For component A, one (or more) individual compound(s) which has (have) a value of Δε < -0.8 is (are) preferably selected. This value must be more negative, the smaller the proportion A in the mixture as a whole.
Component B has pronounced nematogeneity and a flow viscosity of not greater than 30 mm2 • s-1, preferably not greater than 25 mm2 • s-1, at 20°C.
A multiplicity of suitable materials is known to the person skilled in the art from the literature. Particular preference is given to compounds of the formula 0-17.
Particularly preferred individual compounds in component B are extremely low- viscosity nematic liquid crystals having a flow viscosity of not greater than 18 mm2 • s'1, preferably not greater than 12 mm2 • s-1, at 20°C.
Component B is monotropically or enantiotropically nematic, has no smectic phases and is able to prevent the occurrence of smectic phases down to very low temperatures in liquid-crystal mixtures. For example, if various materials of high nematogeneity are added to a smectic liquid-crystal mixture, the nematogeneity of these materials can be compared through the degree of suppression of smectic phases that is achieved.
The mixture may optionally also comprise a component C, comprising compounds having a dielectric anisotropy of Δε >1.5. These so-called positive compounds are generally present in a mixture of negative dielectric anisotropy in amounts of < 20 % by weight, based on the mixture as a whole.
Besides one or more compounds of the formula I, the medium preferably comprises 4 to 15, in particular 5 to 12, and particularly preferably < 10, compounds of the formulae IIA, IIB and/or IIC and optionally one or more compounds of the formula IV-1
Besides compounds of the formula IB and the compounds of the formulae IIA, IIB and/or IIC and optionally IV-1 , other constituents may also be present, for example in an amount of up to 45 % of the mixture as a whole, but preferably up to 35 %, in particular up to 10 %.
The other constituents are preferably selected from nematic or nematogenic substances, in particular known substances, from the classes of the azoxybenzenes, benzylideneanilines, biphenyls, terphenyls, phenyl or cyclohexyl benzoates, phenyl or cyclohexyl cyclohexanecarboxylates, phenylcyclohexanes, cyclohexylbiphenyls, cyclohexylcyclohexanes, cyclohexylnaphthalenes, 1 ,4-biscyclohexylbiphenyls or cyclohexylpyrimidines, phenyl- or cyclohexyldioxanes, optionally halogenated stilbenes, benzyl phenyl ethers, tolanes and substituted cinnamic acid esters.
The most important compounds which are suitable as constituents of liquid-crystal phases of this type can be characterised by the formula IV
R20-L-G-E-R21 IV in which L and E each denote a carbo- or heterocyclic ring system from the group formed by 1 ,4-disubstituted benzene and cyclohexane rings, 4,4’-disubstituted biphenyl, phenylcyclohexane and cyclohexylcyclohexane systems, 2,5-disubstituted pyrimidine and 1 ,3-dioxane rings, 2,6-disubstituted naphthalene, di- and tetrahydronaphthalene, quinazoline and tetrahydroquinazoline,
G denotes -CH=CH- -N(O)=N- -CH=CQ- -CH=N(O)- -≡=C- -CH2-CH2- -CO-O- -CH2-O- -CO-S- -CH2-S- -CH=N- -COO-Phe-COO- -CF2O- -CF=CF- -OCF2- -OCH2- -(CH2)4- -(CH2)3O- or a C-C single bond, Q denotes halogen, preferably chlorine, or -CN, and R20 and R21 each denote alkyl, alkenyl, alkoxy, alkoxyalkyl or alkoxycarbonyloxy having up to 18, preferably up to 8, carbon atoms, or one of these radicals alternatively denotes CN, NC, NO2, NCS, CF3, SF5, OCF3, F, Cl or Br.
In most of these compounds, R20 and R21 are different from one another, one of these radicals usually being an alkyl or alkoxy group. Other variants of the proposed substituents are also common. Many such substances or also mixtures thereof are commercially available. All these substances can be prepared by methods known from the literature.
It goes without saying for the person skilled in the art that the VA, IPS or FFS mixture according to the invention may also comprise compounds in which, for example, H, N, O, Cl and F have been replaced by the corresponding isotopes.
The compounds of formula P are optionally added to the mixtures according to the invention in concentrations of preferably 0.01 to 5 % by weight, particularly preferably 0.2 to 2 % by weight, based on the mixture. These mixtures may optionally also comprise an initiator, as described, for example, in U.S. 6,781,665. The initiator, for example lrganox-1076 from BASF, is preferably added to the mixture comprising polymerizable compounds in amounts of 0 to 1 %. Mixtures of this type can be used for so-called polymer-stabilized VA modes (PS-VA) or PSA (polymer sustained VA), in which polymerization of the reactive mesogens is intended to take place in the liquid-crystalline mixture after filling of a display panel. The prerequisite for this is that the liquid-crystalline compounds of the LC host do not react under the polymerization conditions of the reactive mesogens, i.e. generally on exposure to UV in the wavelength range from 320 to 360 nm. Liquid-crystalline compounds containing an alkenyl side chain, such as, for example, CC-3-V, exhibit no reaction under the polymerization conditions (UV polymerization) for the RMs, hence, herein, such compounds are not to be considered as RMs.
The compounds according to the present invention can be synthesized by or in analogy to known methods described in the literature (for example in the standard works such as Houben-Weyl, Methoden der Organischen Chemie [Methods of Organic Chemistry], Georg-Thieme-Verlag, Stuttgart), under reaction conditions which are known and suitable for said reactions. Use may also be made here of variants which are known per se, but are not mentioned here. In particular, they can be prepared as described in or in analogy to the following reaction schemes. Further methods for preparing the inventive compounds can be taken from the examples.
Other mesogenic compounds which are not explicitly mentioned above can optionally and advantageously also be used in the media in accordance with the present invention. Such compounds are known to the person skilled in the art.
The following examples explain the present invention without limiting it. However, they show the person skilled in the art preferred mixture concepts with compounds preferably to be employed and the respective concentrations thereof and combinations thereof with one another. In addition, the examples illustrate the properties and property combinations that are accessible.
For the present invention and in the following examples, the structures of the liquid- crystal compounds are indicated by means of acronyms, with the transformation into chemical formulae taking place in accordance with Tables A to C below. All radicals CnH2n+1. CmH2m+1 and C1H21+1 or CnH2n. CmH2m and ClH2l are straight-chain alkyl radicals or alkylene radicals, in each case having n, m and I C atoms respectively. Preferably n, m and I are independently of each other 1 , 2, 3, 4, 5, 6, or 7. Table A shows the codes for the ring elements of the nuclei of the compound, Table B lists the bridging units, and Table C lists the meanings of the symbols for the left- and right- hand end groups of the molecules. The acronyms are composed of the codes for the ring elements with optional linking groups, followed by a first hyphen and the codes for the left-hand end group, and a second hyphen and the codes for the right-hand end group. Table D shows illustrative structures of compounds together with their respective abbreviations.
Table C: End groups
in which n and m are each integers, and the three dots are placeholders for other abbreviations from this table.
Apart from the compounds of formula I, IIA, IIB, IIC and/or IID, the mixtures according to the invention preferably comprise one or more compounds of the compounds mentioned below.
The following abbreviations are used:
(n, m, k and I are, independently of one another, each an integer, preferably 1 to 9 preferably 1 to 7, k and I possibly may be also 0 and preferably are 0 to 4, more
preferably 0 or 2 and most preferably 2, n preferably is 1 , 2, 3, 4 or 5, in the combination “-nO-” it preferably is 1 , 2, 3 or 4, preferably 2 or 4, m preferably is 1 , 2, 3, 4 or 5, in the combination “-Om” it preferably is 1 , 2, 3 or 4, more preferably 2 or 4. The combination “-IVm” preferably is “2V1”.)
Table E
Table F
Table F shows preferred reactive mesogenic compounds which can be used in the LC media in accordance with the present invention.
In a preferred embodiment, the mixtures according to the invention comprise one or more polymerizable compounds, preferably selected from the polymerizable compounds of the formulae RM-1 to RM-182. Of these, compounds RM-1 , RM-4, RM-8, RM-17, RM-19, RM-35, RM-37, RM-39, RM-40, RM-41 , RM-48, RM-52, RM- 54, RM-57, RM-58, RM-64, RM-74, RM-76, RM-88, RM-91 , RM-102, RM-103, RM- 109, RM-116, RM-117, RM-120, RM-121 , RM-122, RM-139, RM-140, RM-142, RM- 143, RM-145, RM-146, RM-147, RM-149, RM-156 to RM-163, RM-169, RM-170 and RM-171 to RM-183 are particularly preferred.
Working examples:
The following examples are intended to explain the invention without limiting it. In the examples, m.p. denotes the melting point and C denotes the clearing point of a liquid- crystalline substance in degrees Celsius; boiling temperatures are denoted by m.p. Furthermore: C denotes crystalline solid state, S denotes smectic phase (the index denotes the phase type), N denotes nematic state, Ch denotes cholesteric phase, I denotes isotropic phase, Tg denotes glass-transition temperature. The number between two symbols indicates the conversion temperature in degrees Celsius.
The host mixture used for determination of the optical anisotropy An of single com- pounds is the commercial mixture ZLI-4792 (Merck KGaA). The dielectric anisotropy Δε is determined using commercial mixture ZLI-2857. The physical data of the compound to be investigated are obtained from the change in the dielectric constants of the host mixture after addition of the compound to be investigated and extrapolation to 100 % of the compound employed. In general, 10 % of the compound to be investigated are dissolved in the host mixture, depending on the solubility.
Unless indicated otherwise, parts or per cent data denote parts by weight or per cent by weight.
Above and below:
Vo denotes threshold voltage, capacitive [V] at 20°C, ne denotes extraordinary refractive index at 20°C and 589 nm, n0 denotes ordinary refractive index at 20°C and 589 nm,
Δn denotes optical anisotropy at 20°C and 589 nm, denotes dielectric permittivity perpendicular to the director at 20°C and
1 kHz, denotes dielectric permittivity parallel to the director at 20°C and 1 kHz, Δε denotes dielectric anisotropy at 20°C and 1 kHz, cl. p., T(N, I) denotes clearing point [°C], γ1 denotes rotational viscosity measured at 20°C [mPa-s], K1 denotes elastic constant, "splay" deformation at 20°C [pN],
K2 denotes elastic constant, "twist" deformation at 20°C [pN],
K3 denotes elastic constant, "bend" deformation at 20°C [pN],
LTS denotes low-temperature stability (nematic phase), determined in test cells or in the bulk, as specified.
Unless explicitly noted otherwise, all values indicated in the present application for temperatures, such as, for example, the melting point T(C,N), the transition from the smectic (S) to the nematic (N) phase T(S,N) and the clearing point T(N , I) or cl.p., are indicated in degrees Celsius (°C). M.p. denotes melting point . Furthermore, Tg = glass state, C = crystalline state, N = nematic phase, S = smectic phase and I = isotropic phase. The numbers between these symbols represent the transition tem- peratures.
The term "threshold voltage" for the present invention relates to the capacitive threshold (Vo), also called the Freedericksz threshold, unless explicitly indicated otherwise. In the examples, as is generally usual, the optical threshold can also be indicated for 10 % relative contrast (V10).
The display used for measurement of the capacitive threshold voltage consists of two plane-parallel glass outer plates at a separation of 20 μm, which each have on the insides an electrode layer and an unrubbed polyimide alignment layer on top, which cause a homeotropic edge alignment of the liquid-crystal molecules.
The display or test cell used for measurement of the tilt angle consists of two plane- parallel glass outer plates at a separation of 4 μm, which each have on the insides an electrode layer and a polyimide alignment layer on top, where the two polyimide layers are rubbed antiparallel to one another and cause a homeotropic edge alignment of the liquid-crystal molecules.
Unless indicated otherwise, the VHR is determined at 20°C (VHR20) and after
5 minutes in an oven at 100°C (VHR100) in a commercially available instrument Model LCM-1 (00004) from TOYO Corporation, Japan. The voltage used has a frequency of in a range from 1 Hz to 60 Hz, unless indicated more precisely.
The stability to UV irradiation is investigated in a "Suntest CPS+", a commercial instrument from Heraeus, Germany, using a Xenon lamp NXE1500B. The sealed test cells are irradiated for 2.0 h, unless explicitly indicated, without additional heating.
The irradiation power in the wavelength range from 300 nm to 800 nm is 765 W/ m2 V. A UV "cut-off" filter having an edge wavelength of 310 nm is used in order to simulate the so-called window glass mode. In each series of experiments, at least four test cells are investigated for each condition, and the respective results are indicated as averages of the corresponding individual measurements.
The decrease in the voltage holding ratio (AVHR) usually caused by the exposure, for example by UV irradiation or by LCD backlighting, is
In order to investigate the low-temperature stability, also known as "LTS", i.e. the stability of the LC mixture in the bulk against spontaneous crystallisation of individual components at low temperatures or the occurrence of smectic phases, as the case may be, several sealed bottles, each containing about 1 g of the material, are stored at one or more given temperatures, typically of -10°C, -20°C, -30°C and/or -40°C and it is inspected at regular intervals visually, whether a phase transition is observed or not. As soon as the first one of the samples at a given temperature shows a change time is noted. The time until the last inspection, at which no change has been observed, is noted as the respective LTS.
The ion density from which the resistivity is calculated is measured using the commercially available LC Material Characteristics Measurement System Model 6254 from Toyo Corporation, Japan, using VHR test cells with AL16301 Polyimide (JSR
Corp., Japan) having a 3.2 μm cell gap. The measurement is performed after 5 min of storage in an oven at 60 °C or 100 °C.
The so-called "HTP" denotes the helical twisting power of an optically active or chiral substance in an LC medium (in μm). Unless indicated otherwise, the HTP is measured in the commercially available nematic LC host mixture MLD-6260 (Merck KGaA) at a temperature of 20°C.
The Clearing point is measured using the Mettler Thermosystem FP900. The optical anisotropy (An) is measured using an Abbe Refractometer H005 (Natrium-spectral lamp Na10 at 589nm, 20 °C). The dielectric anisotropy (Δε) is measured using an LCR-Meter E4980A/Agilent (G005) at 20°C (s-parallel-cells with JALS 2096-R1). The turn on voltage (Vo) is measured using an LCR-Meter E4980A/Agilent (G005) at 20°C (s-parallel-cells with JALS 2096-R1). The rotational viscosity (γ1) is measured using a TOYO LCM-2 (0002) at 20°C (gamma 1 negative cells with JALS-2096-R 1 ). The elastic constant (K1 , splay) is measured using an LCR-Meter E4980A/Agilent (G005) at 20°C (s parallel-cells with JALS 2096-R1). K3: The elastic constant (K3, bend) is measured using an LCR-Meter E4980A/Agilent (G005) at 20°C (s-parallel-cells with JALS 2096-R1).
Unless explicitly noted otherwise, all concentrations in the present application are indicated in per cent by weight and relate to the corresponding mixture as a whole, comprising all solid or liquid-crystalline components, without solvents. All physical properties are determined in accordance with "Merck Liquid Crystals, Physical Properties of Liquid Crystals", Status November 1997, Merck KGaA, Germany, and apply for a temperature of 20°C, unless explicitly indicated otherwise.
The entire disclosures of all applications, patents and publications, cited herein and of corresponding European Application No. EP20183843, filed July 3, 2020, are incorporated by reference herein.
The following mixture examples having negative dielectric anisotropy are suitable, in particular, for liquid-crystal displays which have at least one planar alignment layer, such as, for example, IPS and FFS displays, in particular UB-FFS ( = ultra-bright FFS), and for VA displays.
Mixture Examples
The Host Mixtures H1 to H116 and the Mixture Examples M1 to M9 have the compositions and properties given in the following tables. The quantities in the tables are given in percent by weight.
The Host mixture H1 and the mixture example M1 are filled in test cells with a cell gap of 3 μm and the dark state was investigated using a Konica Minolta CA-410 Display Analyser. The medium M1 shows an improvement of the dark state by 15% compared with the host mixture H1. In addition, the medium shows favourably high elastic constants K1 , K3 and KaVg, which enables displays with improved contrast which in combination with the improved dark state results in an excellent picture quality of a display.
Likewise, the host mixtures H2 to H8 and the mixture examples M2 to M8 are filled in test cells with a cell gap of 3 μm and the dark state was investigated using a Konica Minolta CA-410 Display Analyser. The medium M1 shows an improvement of the dark state by 15% compared with the host mixture H1. In addition, the medium shows favourably high elastic constants K1 , K3 and Kavg, which enables displays with improved contrast which in combination with the improved dark state results in an excellent picture quality of a display.
10
15
20
25
„n
10
15
20
25
_n
10
15
20
25
„n
10
15
20
25
„n
Claims
R1 denotes H, a straight chain alkyl or alkoxy radical having 1 to 15 C atoms, or a branched alkyl or alkoxy radical having 3 to 15 C atoms, where one or more CH2 groups in these radicals may each be replaced independently of one another, by
-CH=CH-, -C=C-, -CF2O-, -OCF2-, -0-, -CO-O- or -O-CO- in such a way that O atoms are not linked directly to one another, and in which, in addition, one or more H atoms may be replaced by halogen,
denotes H or CH3,
n is 0 or 1 , v is 1 , 2, 3, 4, 5, or 6; and b) one or more dichroic dyes.
2. The liquid crystal medium according to claim 1 , wherein the one or more dichroic dyes are selected from azo compounds, anthraquinones, thiophenolanthraquinones, methine compounds, azomethine compounds, merocyanine compounds, naphthoquinones, tetrazines, pyrromethene dyes, malononitrile dyes, nickel dithiolenes, phthalocyanines, naphthalocyanines, porphyrins, rylenes, thiadiazole dyes, thienothiadiazole dyes, benzothiadiazoles, thiadiazoloquinoxalines and diketopyrrolopyrroles.
4. The liquid crystal medium according to one or more of claims 1 to 3, wherein the birefringence of the medium is in the range of from 0.075 to 0.110, measured at 20°C and at a wavelength of 589nm.
5. The liquid crystal medium according to one or more of claims 1 to 4, wherein the medium comprises one or more compounds selected from the group of compounds of the formulae IIA, IIB, IIC and IID,
R2A, R2B, R2C and R2D identically or differently, denote H, a straight chain alkyl or alkoxy radical having 1 to 15 C atoms, or a branched alkyl or alkoxy radical having 3 to 15 C atoms, where one or more CH2 groups in these radicals may each be replaced, independently of one another, by
, -CH=CH-, -C=C-, -CF2O-, -OCF2-, -O-, -CO-O- or -O-CO- in such a way that O atoms are not linked directly to one another, and in which, in addition, one or more H atoms may be replaced by halogen,
L1 to L4 each, independently of one another, denote F, Cl, CF3 or CHF2, denotes H, F, Cl, CF3, CHF2 or CH3,
Z2, Z2B and Z2D each, independently of one another, denote a single bond, -CH2CH2-, -CH=CH-, -CF2O-, -OCF2-, -CH2O-, -OCH2-, -COO-,
-OCO-, -C2F4-, -CF=CF- or -CH=CHCH2O-,
(O) denotes O or a single bond,
P denotes 0, 1 or 2, q denotes 0 or 1 , and v denotes an integer from 1 to 6, wherein the compounds of formula I are excluded from formula IID.
6. The medium according to claim 5, wherein the medium comprises one or more compounds of the formula I and one or more compounds selected from the compounds of the formulae IIA, II B, IIC and II D, in which R1, R2A, R2B, R2C and R2D
denote n-propyl, (O) denotes O, and v is 2, in a total concentration in the range of from 30% to 70% by weight.
7. The medium according to one or more of claims 1 to 6, wherein the medium comprises one or more compounds of formula IV
in which
R41 denotes alkyl having 1 to 7 C atoms or alkenyl having 2 to 7 C atoms, and
R42 denotes alkyl having 1 to 7 C atoms or alkoxy having 1 to 6 C atoms or alkenyl having 2 to 7 C atoms.
10. The medium according one or more of claims 1 to 9, wherein the medium comprises one or more compounds of formula V
in which
11. The medium according one or more of claims 1 to 10, wherein the medium comprises one or more compounds of the formula S
in which
Ar denotes a methylene group or an aromatic hydrocarbon group having 6 to 40 C atoms or a heteroaromatic hydrocarbon group having 4 to 40 C atoms;
Sp denotes a spacer group;
Rs denotes H, alkyl having 1 to 12 C atoms or alkenyl having 2 to 12 C atoms;
Zs denotes -O-, -C(O)O-, -(CH2)Z- , -(CH2)ZO-, or a single bond;
RH denotes H, O• CH3, OH or ORS;
RS1, RS2, RS3 and RS4, identically or differently, denote alkyl having 1 to 6 C atoms;
G denotes H or Rs or a group ZS-HA; is an integer from 1 to 6, and q is 2, 3 or 4.
12. The medium according to one or more of claims 1 to 11 , wherein the medium comprises one or more compounds of the formula ST-3
in which
RST denotes H, an alkyl or alkoxy radical having 1 to 15 C atoms, where, in addition, one or more CH2 groups in these radicals may each be replaced, independently of one another, by
-CH=CH-, -C≡C-,
-CF2O-, -OCF2-, -0-, -C0-0-, or -O-CO- in such a way that O atoms are not linked directly to one another, and in which, in addition, one or more H atoms may be replaced by halogen,
ZST each, independently of one another, denote -CO-O-, -O-CO-, -CF2O-, -OCF2-, -CH2O-, -OCH2-, -CH2-, -CH2CH2-, -(CH2)4-, -CH=CH-CH2O-, -C2F4-, -CH2CF2-, -CF2CH2-, -CF=CF-, -CH=CF-, -CF=CH-, -CH=CH-, -C=C- or a single bond,
L1 and L2 each, independently of one another, denote F, Cl, CF3 or CHF2, and p denotes 1 or 2.
13. An LC display comprising the medium according to one or more of claims 1 to 12.
14. The LC display according to claim 13, wherein the display is a VA, IPS, FFS, UB- FFS, SA-FFS, SA-VA, PS-VA, PS-IPS, PS-FFS, PS-UB-FFS, polymer stabilized SA-VA or polymer stabilized SA-FFS display.
15. Use of a medium according to one or more of claims 1 to 11 in an LC display.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP23198445 | 2023-09-20 | ||
| EP23198445.1 | 2023-09-20 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2025061649A1 true WO2025061649A1 (en) | 2025-03-27 |
Family
ID=88097312
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2024/075846 Pending WO2025061649A1 (en) | 2023-09-20 | 2024-09-17 | Liquid crystal medium |
Country Status (2)
| Country | Link |
|---|---|
| TW (1) | TW202528517A (en) |
| WO (1) | WO2025061649A1 (en) |
Citations (55)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS55123673A (en) | 1979-03-16 | 1980-09-24 | Mitsui Toatsu Chem Inc | Liquid crystal color display device |
| DE3017877A1 (en) | 1979-05-14 | 1980-11-27 | Mitsubishi Chem Ind | ANTHRACHINONE COMPOUNDS, THEIR PRODUCTION AND USE |
| JPS5657850A (en) | 1979-10-18 | 1981-05-20 | Nippon Kanko Shikiso Kenkyusho:Kk | Azo compound and dichromatic dye for liquid crystal consisting of the same |
| GB2065158A (en) | 1979-12-13 | 1981-06-24 | Standard Telephones Cables Ltd | Anthraquinone Dichroic Dyes |
| GB2065695A (en) | 1979-10-12 | 1981-07-01 | Hitachi Ltd | Guest-host type liquid crystal composition and liquid crystal display device using the same |
| JPS56104984A (en) | 1980-01-24 | 1981-08-21 | Nippon Kanko Shikiso Kenkyusho:Kk | Bichromic pigment for yellow liquid crystal |
| EP0034832A1 (en) | 1980-02-26 | 1981-09-02 | Siemens Aktiengesellschaft | Liquid crystal with an incorporated pleochroic anthraquinone dyestuff and its use |
| US4308161A (en) | 1980-08-04 | 1981-12-29 | General Electric Company | Novel yellow azo dyes and dichroic liquid crystal compositions made therewith |
| US4308162A (en) | 1980-12-08 | 1981-12-29 | General Electric Company | Novel yellow azo dyes and dichroic liquid crystal compositions made therewith |
| EP0043904A2 (en) | 1980-07-10 | 1982-01-20 | F. HOFFMANN-LA ROCHE & CO. Aktiengesellschaft | Mixtures containing a dyestuff, dyestuffs and their use in liquid crystal mixtures |
| GB2079770A (en) | 1980-06-30 | 1982-01-27 | Gen Electric | Novel tris-azo dyes and liquid crystal compositions made therewith |
| EP0044893A1 (en) | 1980-07-29 | 1982-02-03 | Imperial Chemical Industries Plc | Anthraquinone compounds and process for their preparation |
| GB2081736A (en) | 1980-07-29 | 1982-02-24 | Secr Defence | Liquid Crystal Materials Containing Diamino-Dihydroxy Anthraquinone Pleochroic Dyes |
| GB2082196A (en) | 1980-07-29 | 1982-03-03 | Secr Defence | Liquid Crystal Materials Containing Anthraquinone Pleochroic Dyes |
| EP0047027A1 (en) | 1980-08-22 | 1982-03-10 | BBC Aktiengesellschaft Brown, Boveri & Cie. | Liquid crystals mixture |
| EP0048583A1 (en) | 1980-09-22 | 1982-03-31 | Hitachi, Ltd. | Liquid crystal compositions containing pleochroic anthraquinone dyes, liquid crystal devices containing the compositions, and the dyes |
| WO1982001191A1 (en) | 1980-10-03 | 1982-04-15 | Yamada Y | Composition for liquid crystal color display |
| DE3040102A1 (en) | 1980-10-24 | 1982-06-03 | Merck Patent Gmbh, 6100 Darmstadt | DICHROITIC ANTHRACHINONE DYES, THIS CONTAINING LIQUID CRYSTALINE DIELECTRICS AND ELECTRO-OPTICAL DISPLAY ELEMENT |
| EP0054217A1 (en) | 1980-12-12 | 1982-06-23 | Bayer Ag | Anthraquinone dyes, process for their preparation, their use, as well as liquid-crystal material containing anthraquinone dyes |
| US4340973A (en) | 1979-09-29 | 1982-07-20 | Nippon Electric Co., Ltd. | Selective calling receiver including a voltage converter with low power consumption |
| EP0056492A1 (en) | 1981-01-10 | 1982-07-28 | BASF Aktiengesellschaft | Dyes for liquid crystal mixtures |
| EP0059036A1 (en) | 1981-02-25 | 1982-09-01 | Imperial Chemical Industries Plc | Pleochroic anthraquinone dyes |
| DE3150803A1 (en) | 1981-01-12 | 1982-09-09 | Nippon Electric Co., Ltd., Tokyo | ANTRACHINONE DYES AND LIQUID CRYSTAL DISPLAYING CONTAINERS THEREOF |
| GB2094825A (en) | 1981-01-17 | 1982-09-22 | Mitsubishi Chem Ind | Liquid crystal compositions containing pleochroic anthraquinone dyes |
| GB2094822A (en) | 1981-02-25 | 1982-09-22 | Ici Plc | Anthraquinone dyes |
| DE3110960A1 (en) | 1981-03-20 | 1982-09-30 | Basf Ag, 6700 Ludwigshafen | ELECTROPHOTOGRAPHIC RECORDING MATERIAL |
| DE3115147A1 (en) | 1981-04-15 | 1982-11-04 | Basf Ag, 6700 Ludwigshafen | Dyes for liquid-crystal mixtures |
| DE3115762A1 (en) | 1981-04-18 | 1982-11-11 | Merck Patent Gmbh, 6100 Darmstadt | "DICHROITIC ANTHRACHINONE DYES, THIS CONTAINING LIQUID CRYSTAL DIELECTRICS AND ELECTROOPRIC DISPLAY ELEMENT" |
| DE3123519A1 (en) | 1981-06-13 | 1982-12-30 | Bayer Ag, 5090 Leverkusen | LIQUID CRYSTAL MATERIAL CONTAINING AZO COLORS |
| EP0068427A1 (en) | 1981-06-26 | 1983-01-05 | Mitsubishi Kasei Corporation | Liquid crystal composition |
| DE3126108A1 (en) | 1981-07-02 | 1983-01-20 | Merck Patent Gmbh, 6100 Darmstadt | "LIQUID CRYSTAL DIELECTRIC, NEW DICHROITIC NAPHTHOQUINONE DYES AND ELECTRO-OPTICAL DISPLAY ELEMENT" |
| DE3202761A1 (en) | 1982-01-28 | 1983-08-04 | Merck Patent Gmbh, 6100 Darmstadt | LIQUID CRYSTAL DIELECTRIC, NEW COLORS, METHOD FOR THEIR PRODUCTION AND ELECTRO-OPTICAL DISPLAY ELEMENT |
| DE3534777A1 (en) | 1985-09-30 | 1987-04-02 | Hoechst Ag | LIQUID CRYSTAL PHASE WITH DOPERATING EFFECT |
| DE3534778A1 (en) | 1985-09-30 | 1987-04-02 | Hoechst Ag | CHIRAL ESTER OF MESOGENIC CARBONIC ACIDS, A METHOD FOR THE PRODUCTION THEREOF AND THEIR USE AS A DOPER IN LIQUID CRYSTAL PHASES |
| DE3534780A1 (en) | 1985-09-30 | 1987-04-02 | Hoechst Ag | Chiral phenol esters of mesogenic carboxylic acids, a process for the preparation thereof and the use thereof as dopes in liquid-crystal phases |
| DE4342280A1 (en) | 1993-12-11 | 1995-06-14 | Basf Ag | Polymerizable chiral compounds and their use |
| EP0698649A1 (en) | 1994-08-26 | 1996-02-28 | Basf Aktiengesellschaft | Use of thermoplastically processible long time stable electroluminescent materials |
| DE19541820A1 (en) | 1995-11-09 | 1997-05-15 | Consortium Elektrochem Ind | Liquid crystalline organosiloxanes containing chiral dianhydrohexite derivatives |
| WO1998000428A1 (en) | 1996-07-01 | 1998-01-08 | Merck Patent Gmbh | Chiral dopants |
| GB2328207A (en) | 1997-08-13 | 1999-02-17 | Merck Patent Gmbh | Chiral hydrobenzoin derivatives for use as dopants in liquid crystalline mixtures |
| WO2002006196A1 (en) | 2000-07-13 | 2002-01-24 | Merck Patent Gmbh | Chiral compounds i |
| WO2002006195A1 (en) | 2000-07-13 | 2002-01-24 | Merck Patent Gmbh | Chiral compounds ii |
| WO2002006265A1 (en) | 2000-07-13 | 2002-01-24 | Merck Patent Gmbh | Chiral compounds iii |
| WO2002034739A1 (en) | 2000-10-20 | 2002-05-02 | Merck Patent Gmbh | Chiral binaphthol derivatives |
| WO2002094805A1 (en) | 2001-05-21 | 2002-11-28 | Merck Patent Gmbh | Chiral compounds |
| US6781665B2 (en) | 2002-02-04 | 2004-08-24 | Fujitsu Display Technologies Corporation | Liquid crystal display and method of manufacturing the same |
| US20090032771A1 (en) * | 2007-08-01 | 2009-02-05 | Chisso Corporation | Liquid crystal composition and liquid crystal display device |
| EP2166040A1 (en) | 2008-09-22 | 2010-03-24 | Radiant Color N.V. | Novel lipophilic fluorescent dyes and a process for their production |
| US20110042651A1 (en) | 2006-04-07 | 2011-02-24 | Basf Se | Liquid crystalline rylene tetracarboxylic acid derivatives and use thereof |
| WO2014090373A1 (en) | 2012-12-13 | 2014-06-19 | Merck Patent Gmbh | Liquid-crystalline medium |
| WO2014187529A1 (en) | 2013-05-24 | 2014-11-27 | Merck Patent Gmbh | Device for controlling the passage of energy, containing a dichroic dye compound |
| WO2015090497A1 (en) | 2013-12-19 | 2015-06-25 | Merck Patent Gmbh | Dye compounds |
| WO2016177449A1 (en) | 2015-05-06 | 2016-11-10 | Merck Patent Gmbh | Thiadiazoloquinoxaline derivatives |
| WO2020104563A1 (en) | 2018-11-23 | 2020-05-28 | Merck Patent Gmbh | Dichroic dye composition |
| EP4166628A1 (en) * | 2021-10-18 | 2023-04-19 | Merck Patent GmbH | Liquid crystal medium |
-
2024
- 2024-09-17 WO PCT/EP2024/075846 patent/WO2025061649A1/en active Pending
- 2024-09-19 TW TW113135531A patent/TW202528517A/en unknown
Patent Citations (58)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS55123673A (en) | 1979-03-16 | 1980-09-24 | Mitsui Toatsu Chem Inc | Liquid crystal color display device |
| DE3017877A1 (en) | 1979-05-14 | 1980-11-27 | Mitsubishi Chem Ind | ANTHRACHINONE COMPOUNDS, THEIR PRODUCTION AND USE |
| US4340973A (en) | 1979-09-29 | 1982-07-20 | Nippon Electric Co., Ltd. | Selective calling receiver including a voltage converter with low power consumption |
| GB2065695A (en) | 1979-10-12 | 1981-07-01 | Hitachi Ltd | Guest-host type liquid crystal composition and liquid crystal display device using the same |
| JPS5657850A (en) | 1979-10-18 | 1981-05-20 | Nippon Kanko Shikiso Kenkyusho:Kk | Azo compound and dichromatic dye for liquid crystal consisting of the same |
| GB2065158A (en) | 1979-12-13 | 1981-06-24 | Standard Telephones Cables Ltd | Anthraquinone Dichroic Dyes |
| JPS56104984A (en) | 1980-01-24 | 1981-08-21 | Nippon Kanko Shikiso Kenkyusho:Kk | Bichromic pigment for yellow liquid crystal |
| EP0034832A1 (en) | 1980-02-26 | 1981-09-02 | Siemens Aktiengesellschaft | Liquid crystal with an incorporated pleochroic anthraquinone dyestuff and its use |
| GB2079770A (en) | 1980-06-30 | 1982-01-27 | Gen Electric | Novel tris-azo dyes and liquid crystal compositions made therewith |
| EP0043904A2 (en) | 1980-07-10 | 1982-01-20 | F. HOFFMANN-LA ROCHE & CO. Aktiengesellschaft | Mixtures containing a dyestuff, dyestuffs and their use in liquid crystal mixtures |
| EP0044893A1 (en) | 1980-07-29 | 1982-02-03 | Imperial Chemical Industries Plc | Anthraquinone compounds and process for their preparation |
| GB2081736A (en) | 1980-07-29 | 1982-02-24 | Secr Defence | Liquid Crystal Materials Containing Diamino-Dihydroxy Anthraquinone Pleochroic Dyes |
| GB2082196A (en) | 1980-07-29 | 1982-03-03 | Secr Defence | Liquid Crystal Materials Containing Anthraquinone Pleochroic Dyes |
| US4308161A (en) | 1980-08-04 | 1981-12-29 | General Electric Company | Novel yellow azo dyes and dichroic liquid crystal compositions made therewith |
| EP0047027A1 (en) | 1980-08-22 | 1982-03-10 | BBC Aktiengesellschaft Brown, Boveri & Cie. | Liquid crystals mixture |
| EP0048583A1 (en) | 1980-09-22 | 1982-03-31 | Hitachi, Ltd. | Liquid crystal compositions containing pleochroic anthraquinone dyes, liquid crystal devices containing the compositions, and the dyes |
| EP0060895A1 (en) | 1980-10-03 | 1982-09-29 | MITSUI TOATSU CHEMICALS, Inc. | Composition for liquid crystal color display |
| WO1982001191A1 (en) | 1980-10-03 | 1982-04-15 | Yamada Y | Composition for liquid crystal color display |
| DE3040102A1 (en) | 1980-10-24 | 1982-06-03 | Merck Patent Gmbh, 6100 Darmstadt | DICHROITIC ANTHRACHINONE DYES, THIS CONTAINING LIQUID CRYSTALINE DIELECTRICS AND ELECTRO-OPTICAL DISPLAY ELEMENT |
| WO1982002054A1 (en) | 1980-12-08 | 1982-06-24 | Gen Electric | Novel yellow azo dyes and dichroic liquid crystal compositions made therefrom |
| US4308162A (en) | 1980-12-08 | 1981-12-29 | General Electric Company | Novel yellow azo dyes and dichroic liquid crystal compositions made therewith |
| EP0054217A1 (en) | 1980-12-12 | 1982-06-23 | Bayer Ag | Anthraquinone dyes, process for their preparation, their use, as well as liquid-crystal material containing anthraquinone dyes |
| EP0056492A1 (en) | 1981-01-10 | 1982-07-28 | BASF Aktiengesellschaft | Dyes for liquid crystal mixtures |
| DE3150803A1 (en) | 1981-01-12 | 1982-09-09 | Nippon Electric Co., Ltd., Tokyo | ANTRACHINONE DYES AND LIQUID CRYSTAL DISPLAYING CONTAINERS THEREOF |
| GB2094825A (en) | 1981-01-17 | 1982-09-22 | Mitsubishi Chem Ind | Liquid crystal compositions containing pleochroic anthraquinone dyes |
| DE3201120A1 (en) | 1981-01-17 | 1982-10-21 | Hitachi, Ltd., Tokyo | LIQUID CRYSTAL COMPOSITION |
| GB2094822A (en) | 1981-02-25 | 1982-09-22 | Ici Plc | Anthraquinone dyes |
| EP0059036A1 (en) | 1981-02-25 | 1982-09-01 | Imperial Chemical Industries Plc | Pleochroic anthraquinone dyes |
| DE3110960A1 (en) | 1981-03-20 | 1982-09-30 | Basf Ag, 6700 Ludwigshafen | ELECTROPHOTOGRAPHIC RECORDING MATERIAL |
| DE3115147A1 (en) | 1981-04-15 | 1982-11-04 | Basf Ag, 6700 Ludwigshafen | Dyes for liquid-crystal mixtures |
| DE3115762A1 (en) | 1981-04-18 | 1982-11-11 | Merck Patent Gmbh, 6100 Darmstadt | "DICHROITIC ANTHRACHINONE DYES, THIS CONTAINING LIQUID CRYSTAL DIELECTRICS AND ELECTROOPRIC DISPLAY ELEMENT" |
| DE3123519A1 (en) | 1981-06-13 | 1982-12-30 | Bayer Ag, 5090 Leverkusen | LIQUID CRYSTAL MATERIAL CONTAINING AZO COLORS |
| EP0068427A1 (en) | 1981-06-26 | 1983-01-05 | Mitsubishi Kasei Corporation | Liquid crystal composition |
| DE3126108A1 (en) | 1981-07-02 | 1983-01-20 | Merck Patent Gmbh, 6100 Darmstadt | "LIQUID CRYSTAL DIELECTRIC, NEW DICHROITIC NAPHTHOQUINONE DYES AND ELECTRO-OPTICAL DISPLAY ELEMENT" |
| DE3202761A1 (en) | 1982-01-28 | 1983-08-04 | Merck Patent Gmbh, 6100 Darmstadt | LIQUID CRYSTAL DIELECTRIC, NEW COLORS, METHOD FOR THEIR PRODUCTION AND ELECTRO-OPTICAL DISPLAY ELEMENT |
| DE3534777A1 (en) | 1985-09-30 | 1987-04-02 | Hoechst Ag | LIQUID CRYSTAL PHASE WITH DOPERATING EFFECT |
| DE3534778A1 (en) | 1985-09-30 | 1987-04-02 | Hoechst Ag | CHIRAL ESTER OF MESOGENIC CARBONIC ACIDS, A METHOD FOR THE PRODUCTION THEREOF AND THEIR USE AS A DOPER IN LIQUID CRYSTAL PHASES |
| DE3534780A1 (en) | 1985-09-30 | 1987-04-02 | Hoechst Ag | Chiral phenol esters of mesogenic carboxylic acids, a process for the preparation thereof and the use thereof as dopes in liquid-crystal phases |
| DE4342280A1 (en) | 1993-12-11 | 1995-06-14 | Basf Ag | Polymerizable chiral compounds and their use |
| EP0698649A1 (en) | 1994-08-26 | 1996-02-28 | Basf Aktiengesellschaft | Use of thermoplastically processible long time stable electroluminescent materials |
| DE19541820A1 (en) | 1995-11-09 | 1997-05-15 | Consortium Elektrochem Ind | Liquid crystalline organosiloxanes containing chiral dianhydrohexite derivatives |
| WO1998000428A1 (en) | 1996-07-01 | 1998-01-08 | Merck Patent Gmbh | Chiral dopants |
| GB2328207A (en) | 1997-08-13 | 1999-02-17 | Merck Patent Gmbh | Chiral hydrobenzoin derivatives for use as dopants in liquid crystalline mixtures |
| WO2002006196A1 (en) | 2000-07-13 | 2002-01-24 | Merck Patent Gmbh | Chiral compounds i |
| WO2002006265A1 (en) | 2000-07-13 | 2002-01-24 | Merck Patent Gmbh | Chiral compounds iii |
| WO2002006195A1 (en) | 2000-07-13 | 2002-01-24 | Merck Patent Gmbh | Chiral compounds ii |
| WO2002034739A1 (en) | 2000-10-20 | 2002-05-02 | Merck Patent Gmbh | Chiral binaphthol derivatives |
| WO2002094805A1 (en) | 2001-05-21 | 2002-11-28 | Merck Patent Gmbh | Chiral compounds |
| US6781665B2 (en) | 2002-02-04 | 2004-08-24 | Fujitsu Display Technologies Corporation | Liquid crystal display and method of manufacturing the same |
| US20110042651A1 (en) | 2006-04-07 | 2011-02-24 | Basf Se | Liquid crystalline rylene tetracarboxylic acid derivatives and use thereof |
| US20090032771A1 (en) * | 2007-08-01 | 2009-02-05 | Chisso Corporation | Liquid crystal composition and liquid crystal display device |
| EP2166040A1 (en) | 2008-09-22 | 2010-03-24 | Radiant Color N.V. | Novel lipophilic fluorescent dyes and a process for their production |
| WO2014090373A1 (en) | 2012-12-13 | 2014-06-19 | Merck Patent Gmbh | Liquid-crystalline medium |
| WO2014187529A1 (en) | 2013-05-24 | 2014-11-27 | Merck Patent Gmbh | Device for controlling the passage of energy, containing a dichroic dye compound |
| WO2015090497A1 (en) | 2013-12-19 | 2015-06-25 | Merck Patent Gmbh | Dye compounds |
| WO2016177449A1 (en) | 2015-05-06 | 2016-11-10 | Merck Patent Gmbh | Thiadiazoloquinoxaline derivatives |
| WO2020104563A1 (en) | 2018-11-23 | 2020-05-28 | Merck Patent Gmbh | Dichroic dye composition |
| EP4166628A1 (en) * | 2021-10-18 | 2023-04-19 | Merck Patent GmbH | Liquid crystal medium |
Non-Patent Citations (10)
| Title |
|---|
| B. BAHADUR: "Liquid Crystals - Applications and Uses", vol. 3, 1992, SCIENTIFIC PUBLISHING |
| C. TSCHIERSKEG. PELZLS. DIELE, ANGEW. CHEM., vol. 116, 2004, pages 6340 - 6368 |
| H. SEKIC. SHISHIDOS. YASUIT. UCHIDA, JPN. J. APPL. PHYS., vol. 21, 1982, pages 191 - 192 |
| M. RICHTER: "Einführung in die Farbmetrik", 1981, WALTER DE GRUYTER & CO |
| MERCK LIQUID CRYSTALS, PHYSICAL PROPERTIES OF LIQUID CRYSTALS, November 1997 (1997-11-01) |
| PURE APPL. CHEM, vol. 73, no. 5, 2001, pages 888 |
| S.H. JUNG ET AL., JPN. J. APPL. PHYS., vol. 43, no. 3, 2004, pages 1028 |
| S.H. LEE ET AL., APPL. PHYS. LETT., vol. 73, no. 20, 1998, pages 2882 - 2883 |
| S.H. LEE ET AL., LIQUID CRYSTALS, vol. 39, no. 9, 2012, pages 1141 - 1148 |
| T. UCHIDAC. SHISHIDOH. SEKIM. WADA, MOL. CRYST. LIQ. CRYST, vol. 39, 1977, pages 39 - 52 |
Also Published As
| Publication number | Publication date |
|---|---|
| TW202528517A (en) | 2025-07-16 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP3839008B1 (en) | Liquid crystal medium | |
| CN103459554B (en) | liquid crystal medium | |
| JP2025094008A (en) | liquid crystal medium | |
| EP3933009B1 (en) | Liquid crystal medium | |
| EP4176021B1 (en) | Liquid crystal medium | |
| EP4399258A1 (en) | Liquid-crystal medium | |
| EP3898892B1 (en) | Liquid-crystal medium | |
| JP7709473B2 (en) | liquid crystal medium | |
| KR20240008264A (en) | Liquid crystal medium | |
| WO2025061649A1 (en) | Liquid crystal medium |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 24768998 Country of ref document: EP Kind code of ref document: A1 |