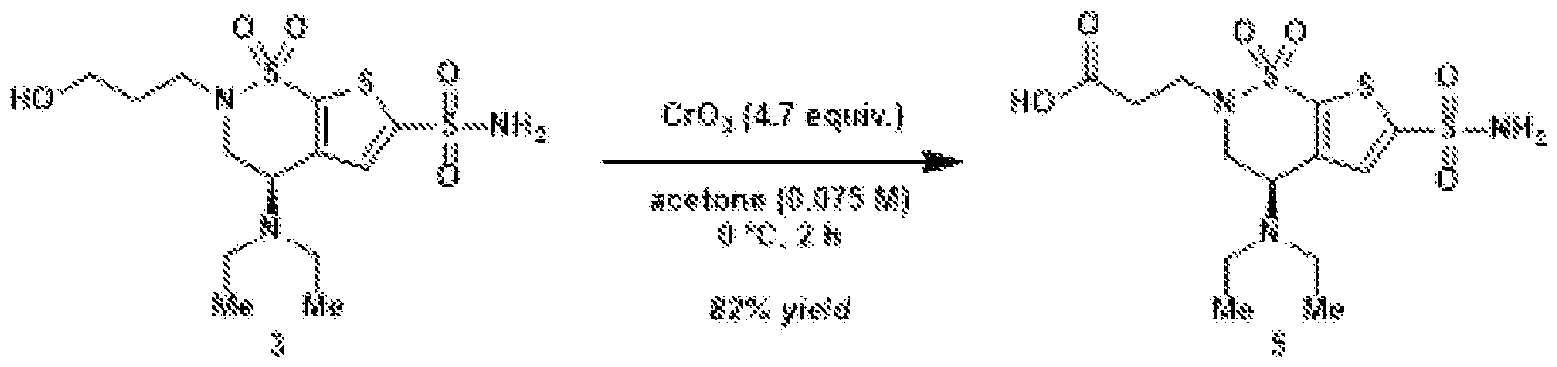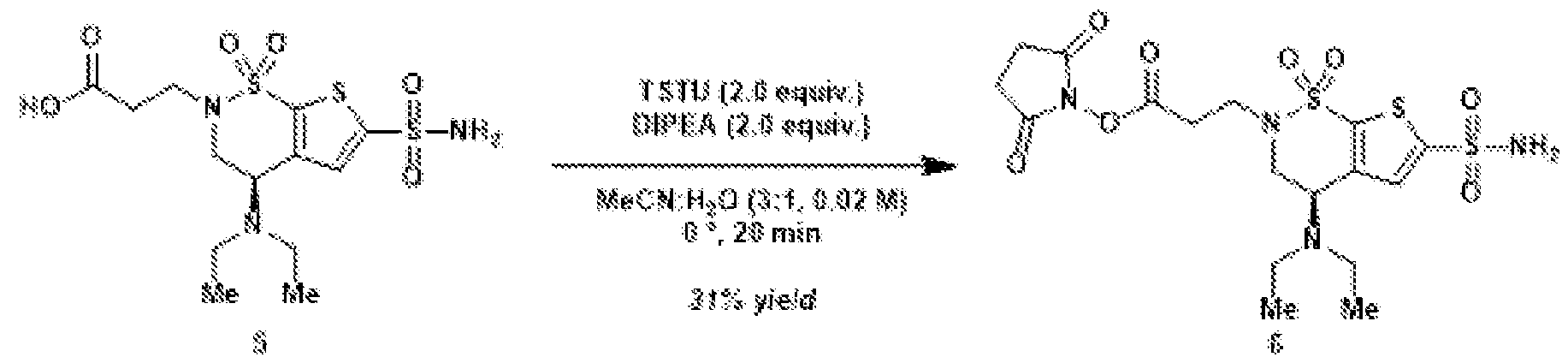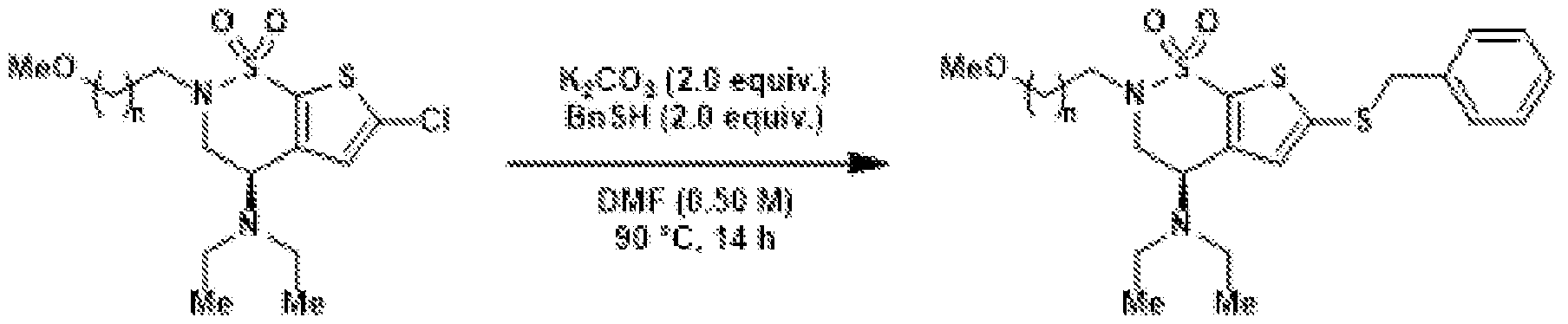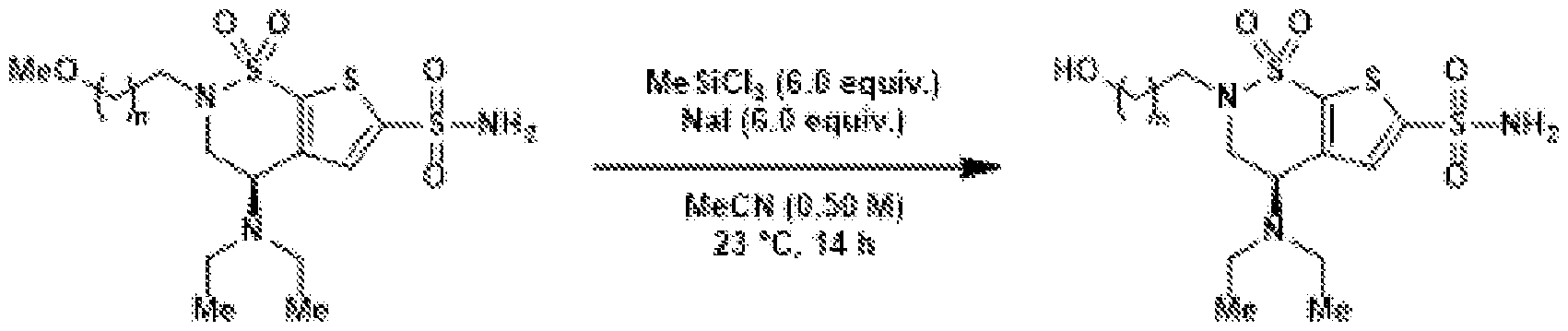WO2025054461A1 - Ester derivatives of binders targeting ca-iv - Google Patents
Ester derivatives of binders targeting ca-iv Download PDFInfo
- Publication number
- WO2025054461A1 WO2025054461A1 PCT/US2024/045602 US2024045602W WO2025054461A1 WO 2025054461 A1 WO2025054461 A1 WO 2025054461A1 US 2024045602 W US2024045602 W US 2024045602W WO 2025054461 A1 WO2025054461 A1 WO 2025054461A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- bza
- brinzolamide
- conjugated
- compound
- conjugate
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/51—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent
- A61K47/54—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an organic compound
- A61K47/545—Heterocyclic compounds
Definitions
- the invention relates to methods and shuttles for crossing the blood brain barrier.
- BBB blood brain barrier
- CNS central nervous system
- This structure comprising mainly of brain endothelial cells, requires large molecules to be delivered via invasive intracranial injections, technically challenging focused ultrasound, or receptor-mediated transcytosis.
- the rational design of BBB-crossing large molecules has long been hampered by the imperfect understanding of the mechanisms involved in transcytosis, with only a handful of targets, such as the transferrin receptor, validated for research and therapies. Thus, the identification of BBB-crossing targets, mechanisms, molecules and methods is needed to improve the efficiencies of research tools and therapies for CNS. Summary
- the present invention provides compositions and methods comprising brinzolamide and brinzolamide derivates as shuttles for the BBB-crossing, for example through the receptor carbonic anhydrase IV (CA-IV).
- CA-IV receptor carbonic anhydrase IV
- the present invention provides rationally designed reactive small-molecule binders based on brinzolamide, that can serve as a shuttle to facilitate CA-IV-mediated brain delivery.
- the reactive small-molecule shuttles may be conjugated to different therapeutic cargo modalities, including nanobodies, therapeutic IgG antibodies, small interfering RNAs (siRNAs), or antisense oligonucleotides (ASOs), for example through a single-step N-hydroxysuccinimide (NHS) ester reaction.
- the present invention provides a robust and adaptable approach for transporting therapeutic substances through the blood-brain barrier using CA-IV-mediated delivery introduced by linking therapeutic cargo to brinzolamide derivative CA-IV binders through bioconjugation.
- Aspects of the invention provide a conjugate comprising a blood brain barrier (BBB) shuttle selected from brinzolamide or a derivative thereof and a therapeutic cargo conjugated to the shuttle.
- BBB blood brain barrier
- the brinzolamide derivative may be, for example a brinzolamide ester.
- the brinzolamide derivative may advantageously have greater specificity for CA-IV in comparison to brinzolamide.
- the brinzolamide derivative may have decreased specificity for CA II in comparison to brinzolamide.
- the brinzolamide derivative may comprise an N-hydroxysuccinimide (NHS) ester.
- the brinzolamide derivative comprising an NHS ester may be formed from a single-step reaction ester reaction.
- the brinzolamide derivative may be formed from the steps of alkylation, tosylation, amination, oxidation, deprotection, Jones oxidation, and esterification of brinzolamide.
- the brinzolamide derivative may be covalently bonded to an NHS ester via a 1-8 carbon alkyl linker.
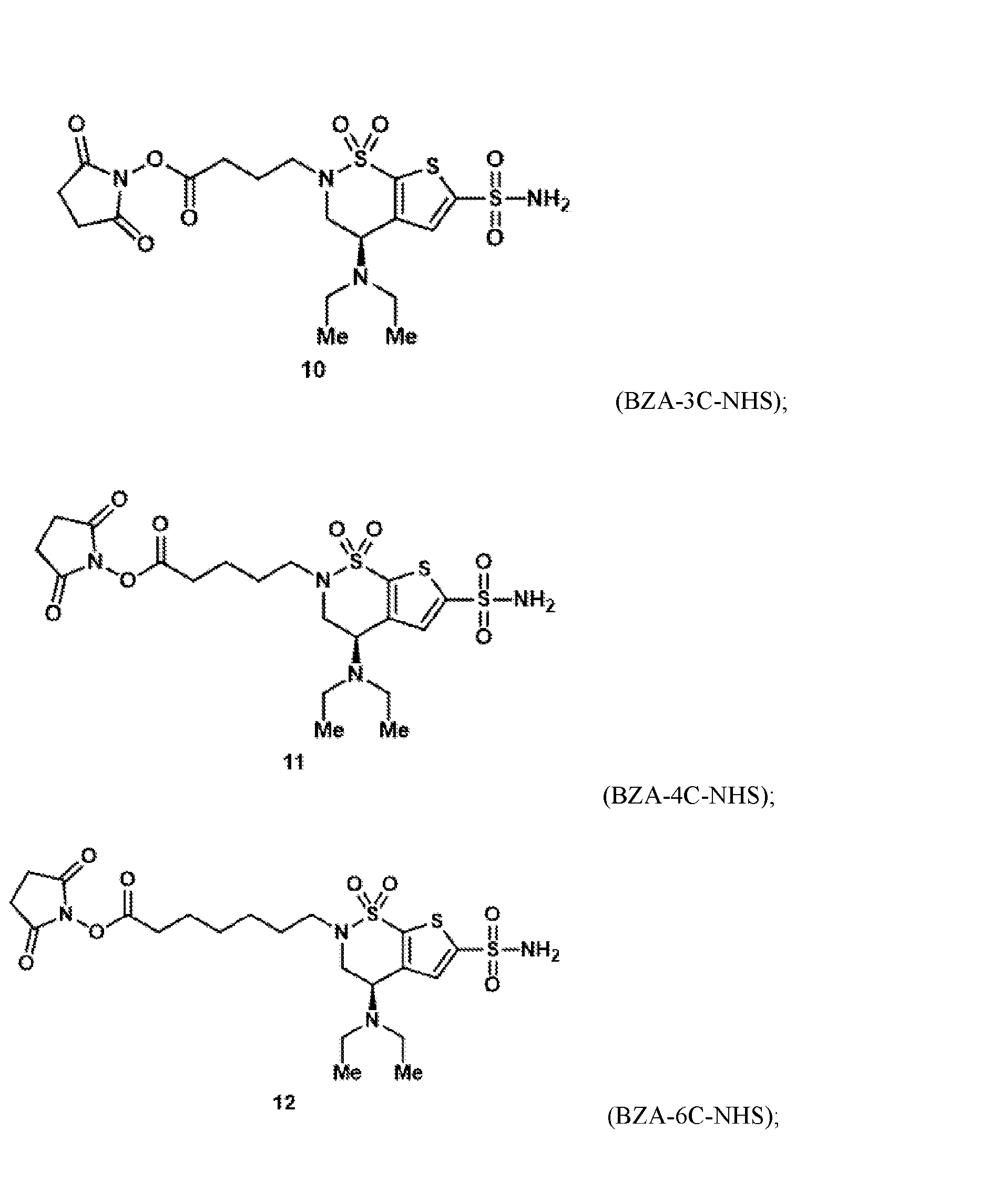
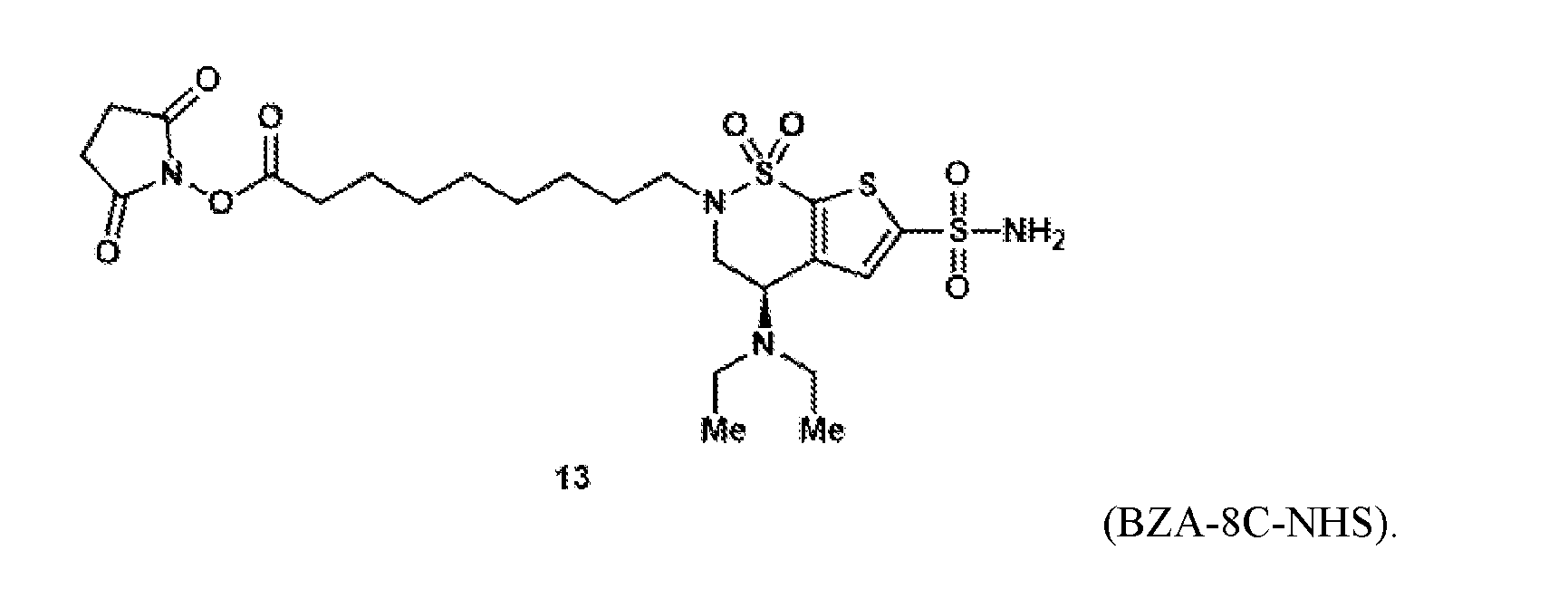
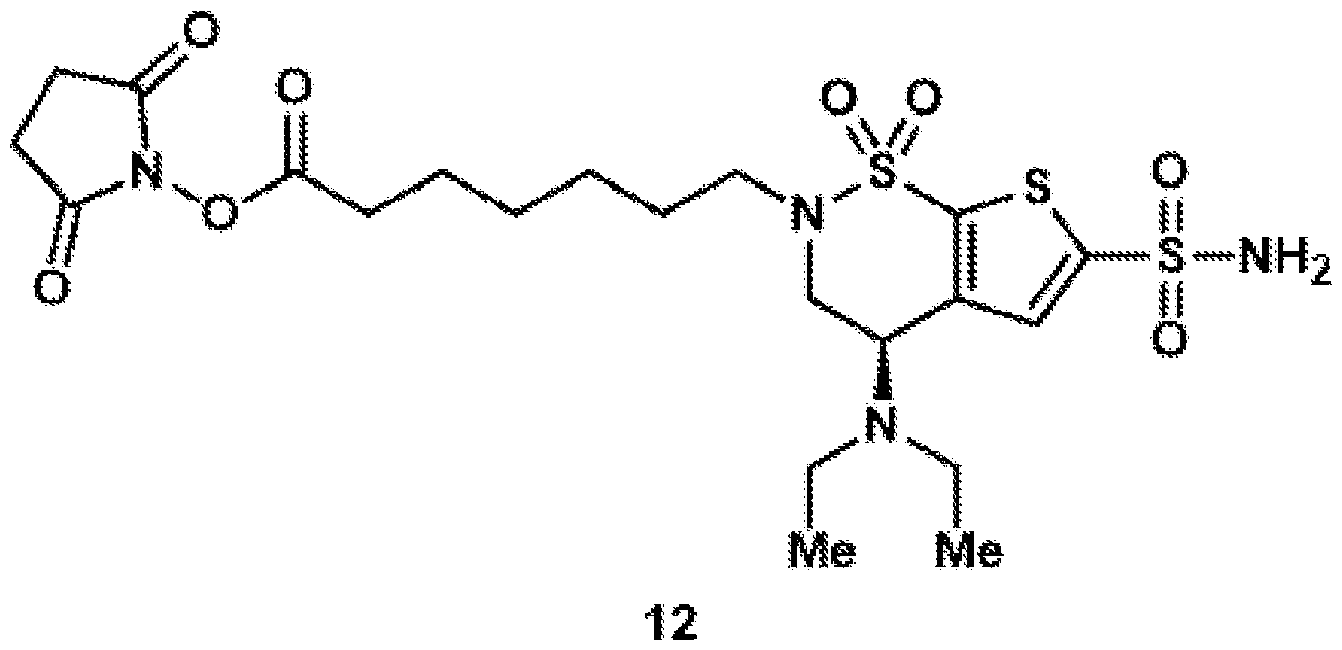
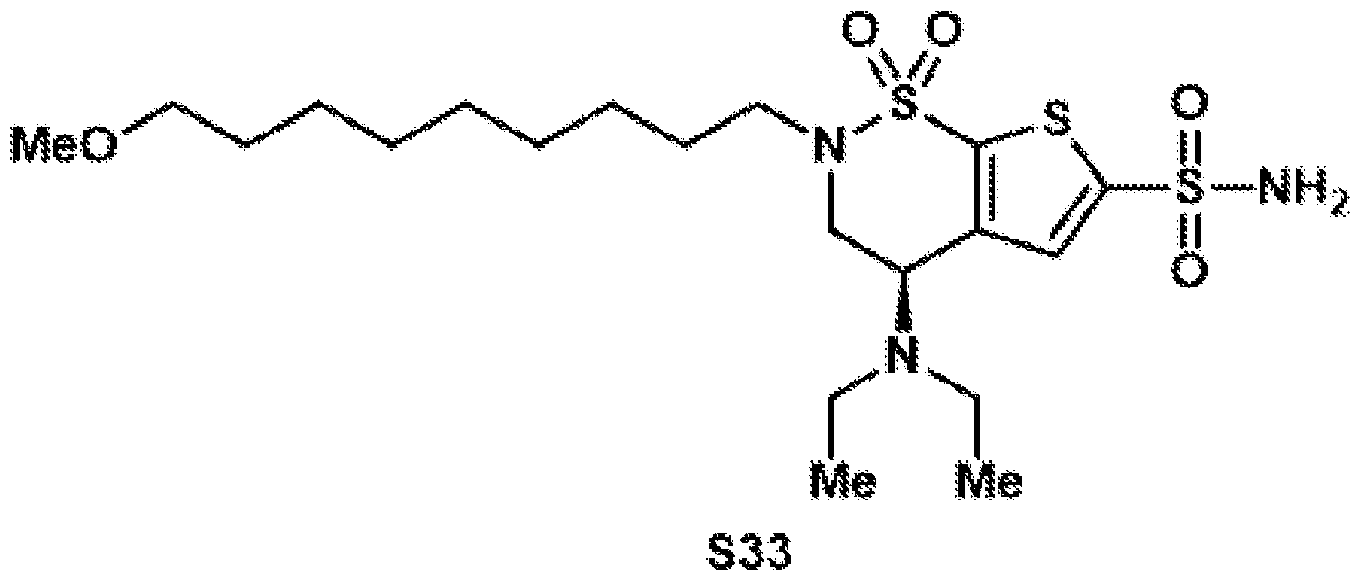
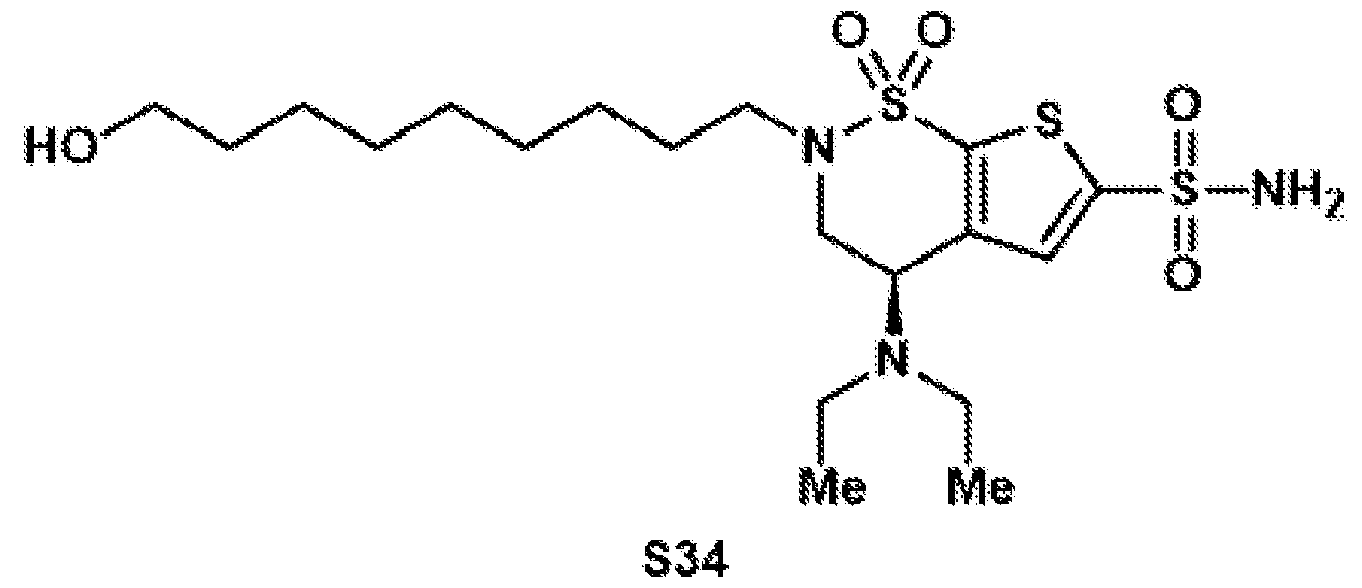
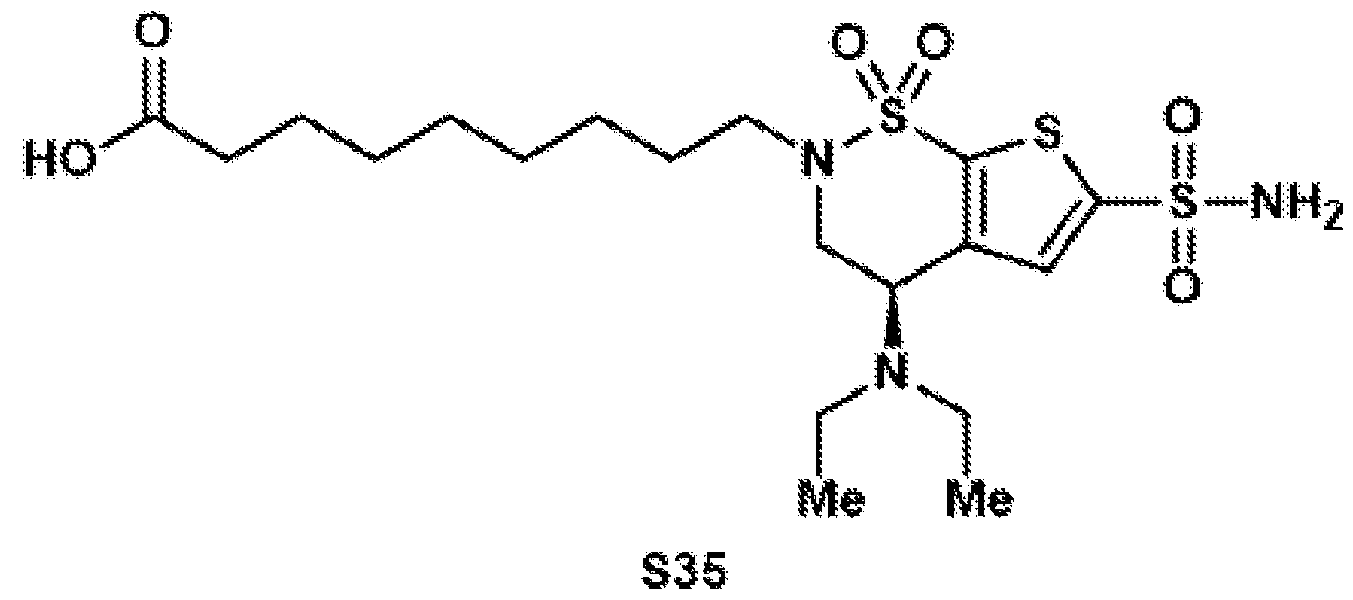
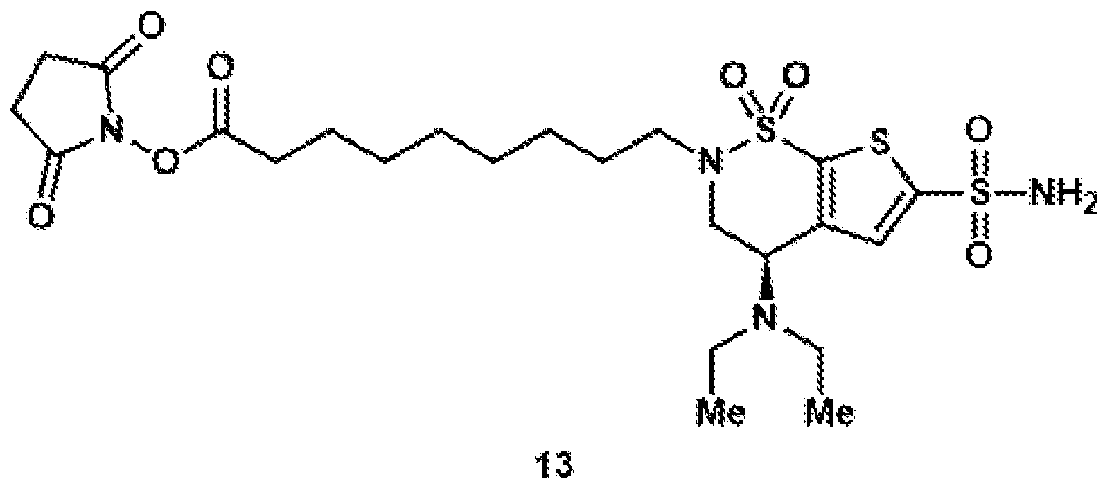
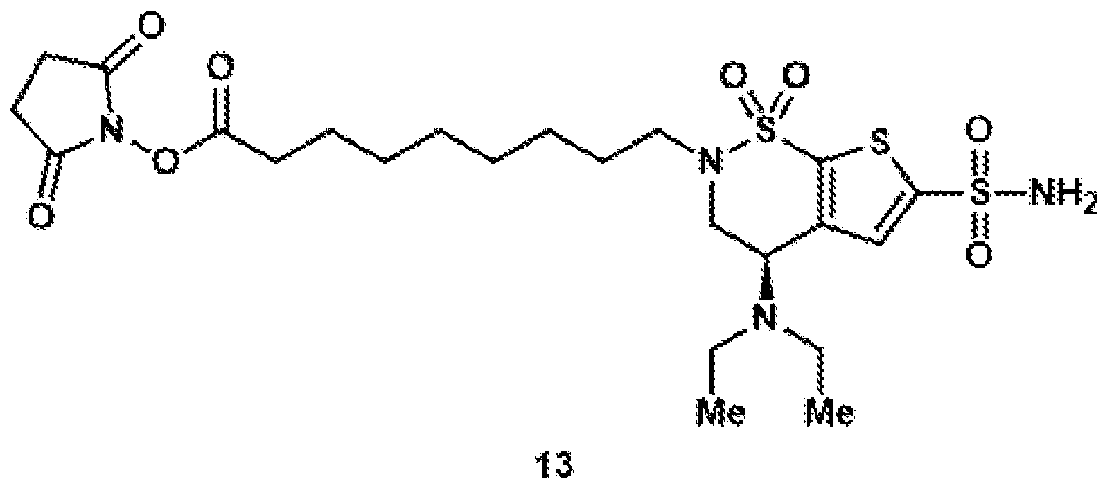
- the brinzolamide ester has the formula: (BZA-nc-NHS), wherein n is 1-8, preferably n is 1, 3, 4, 6 or 8.
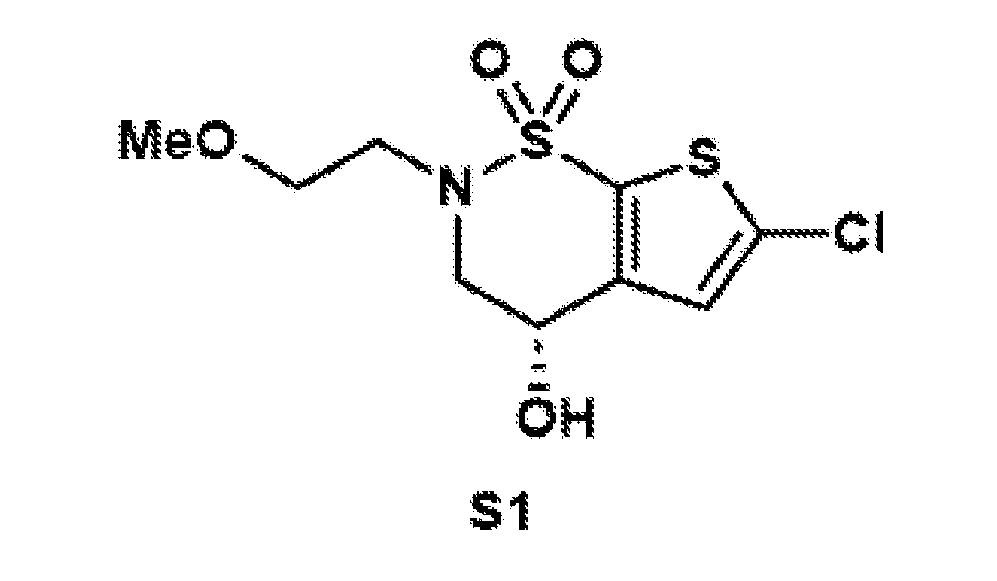
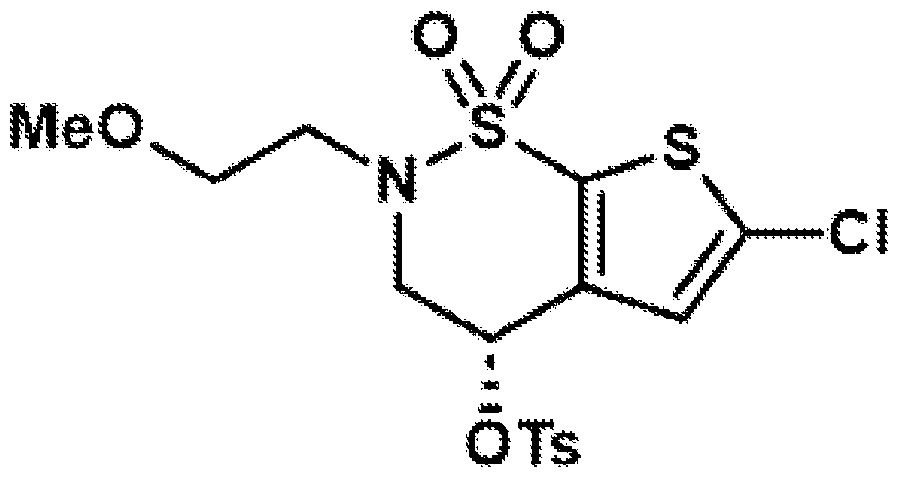
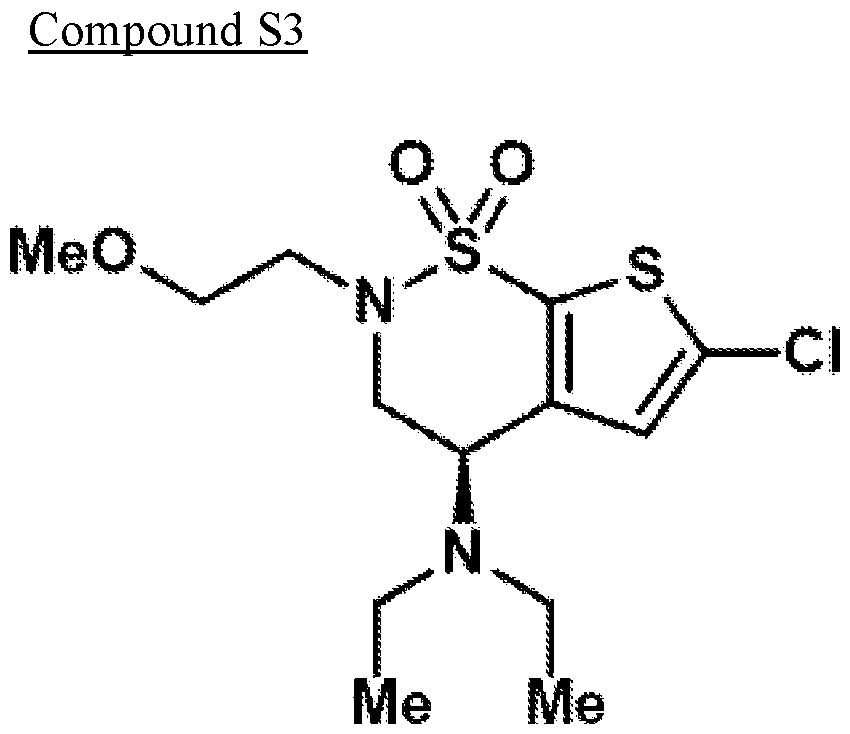
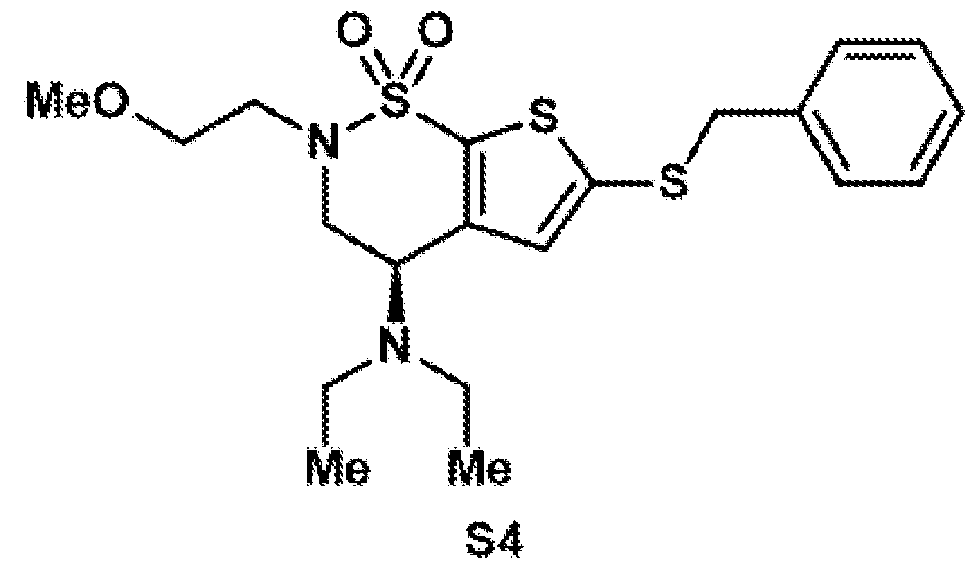
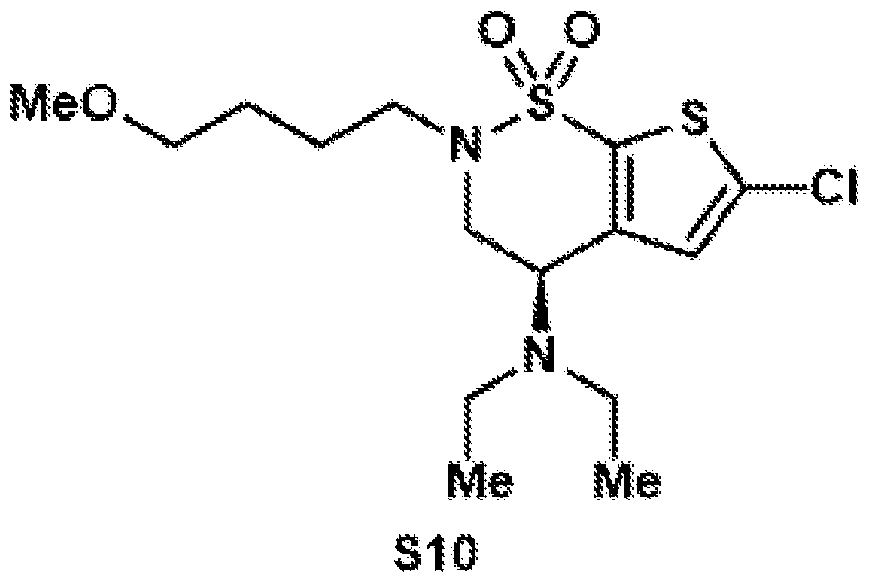
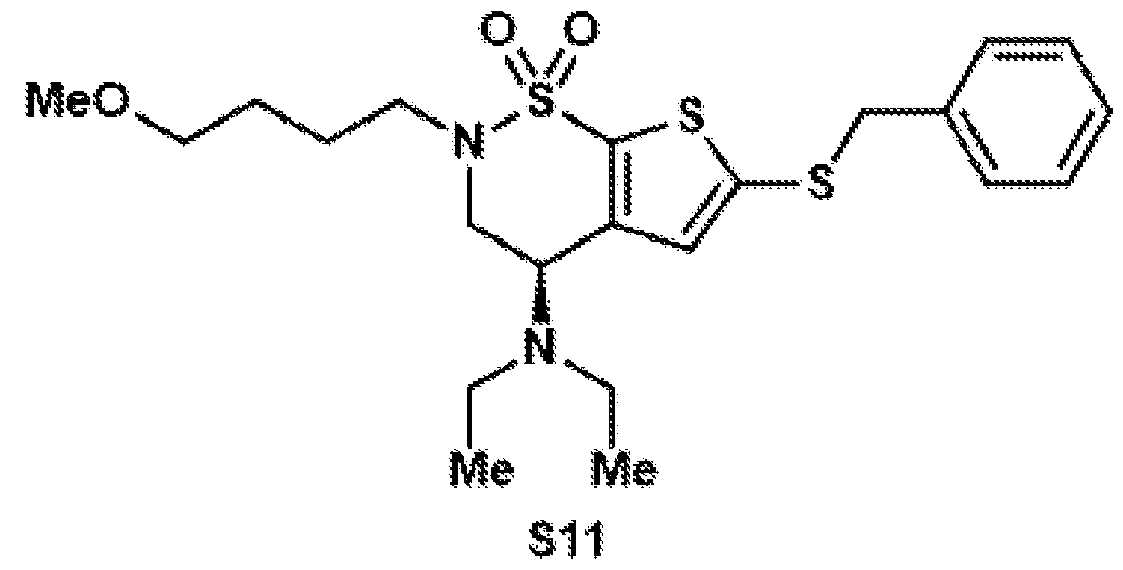
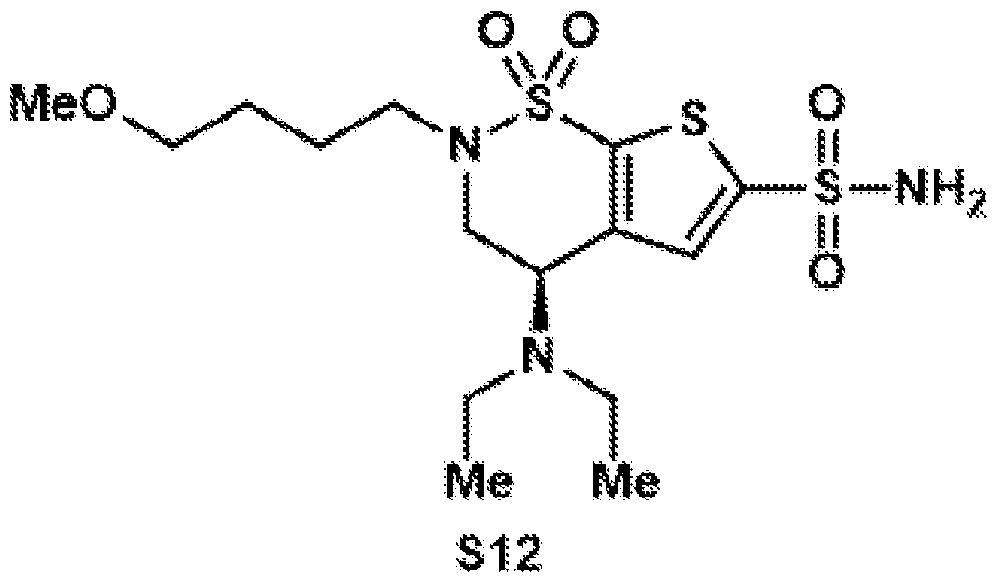
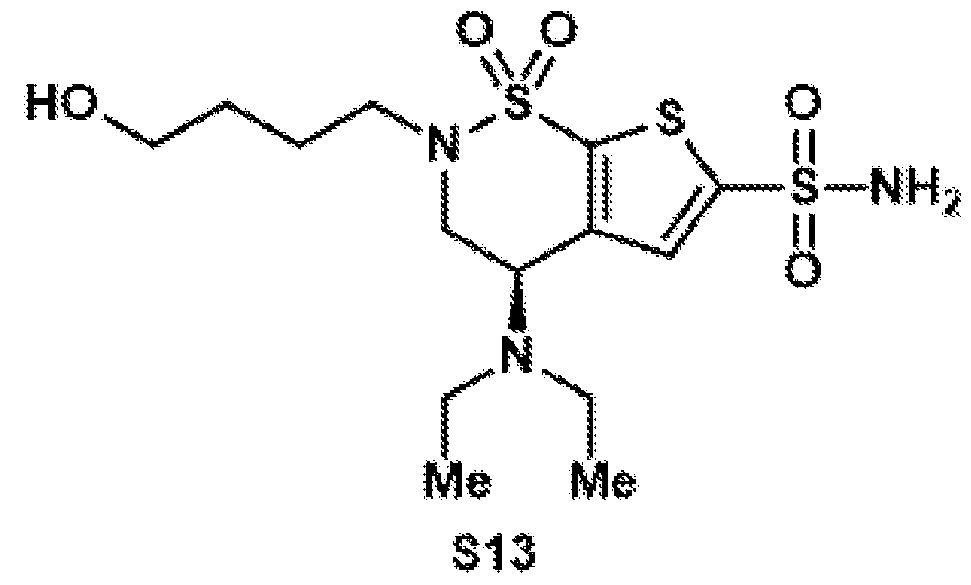
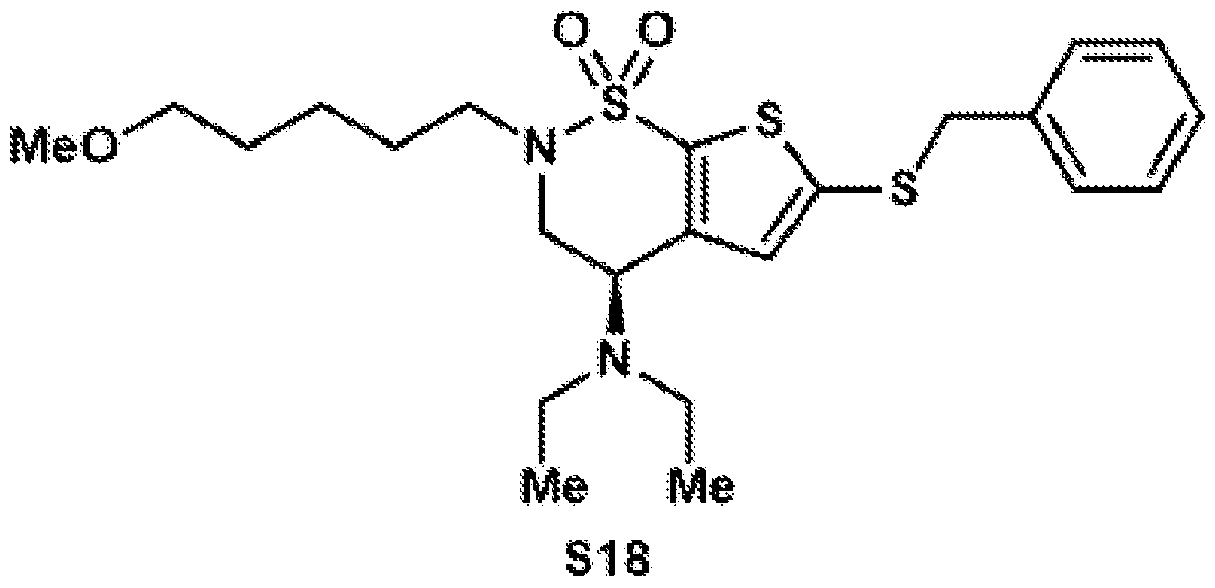
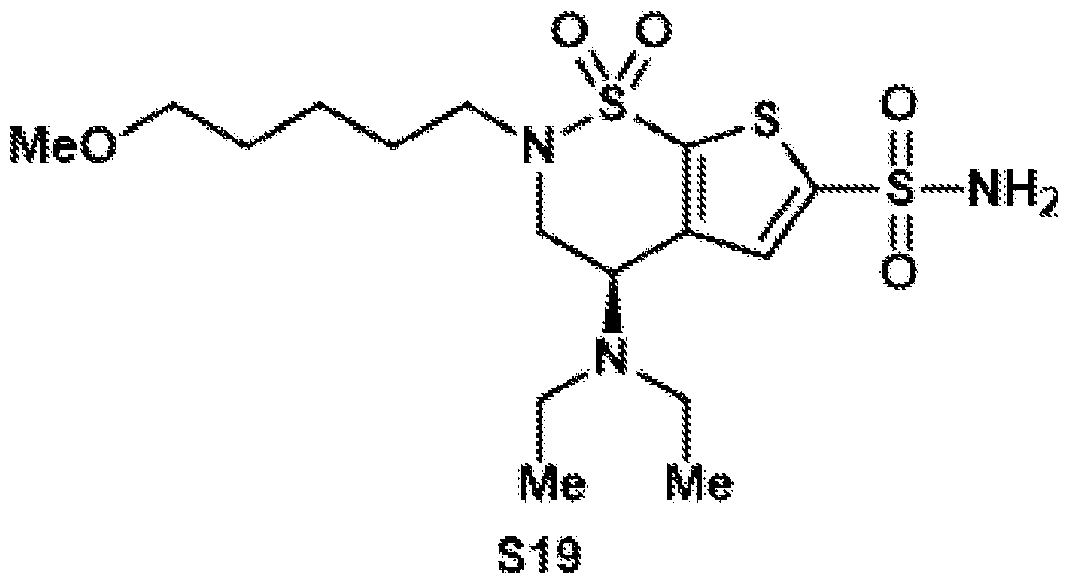
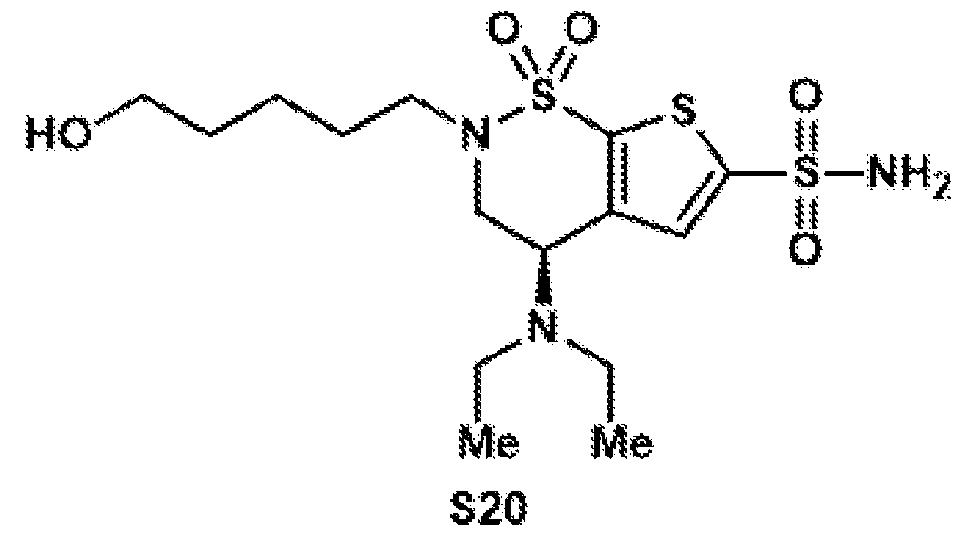
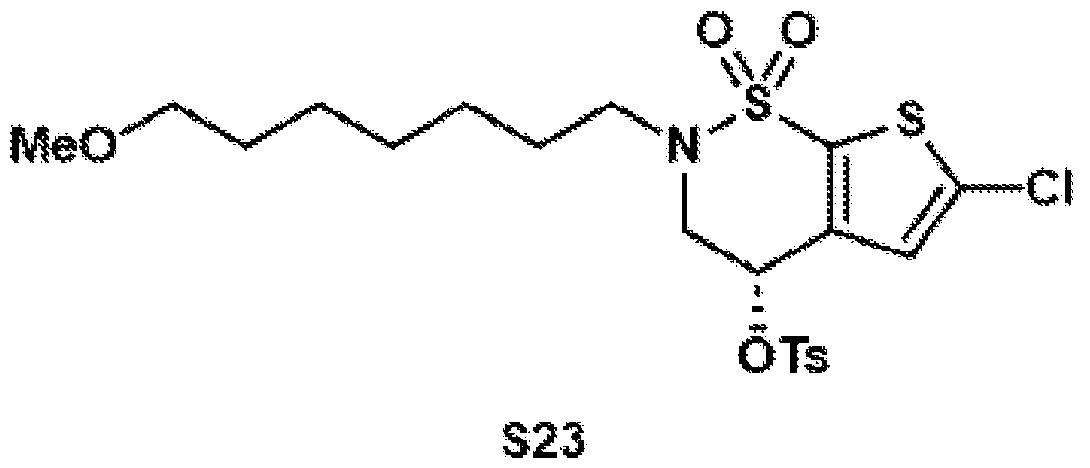
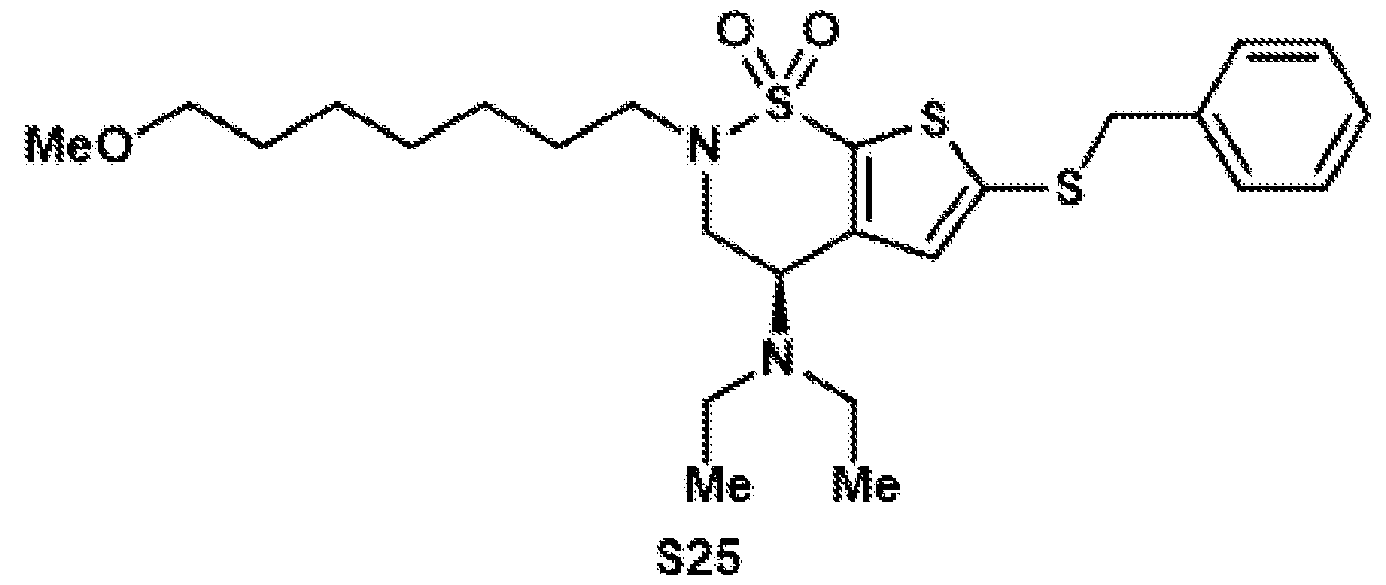
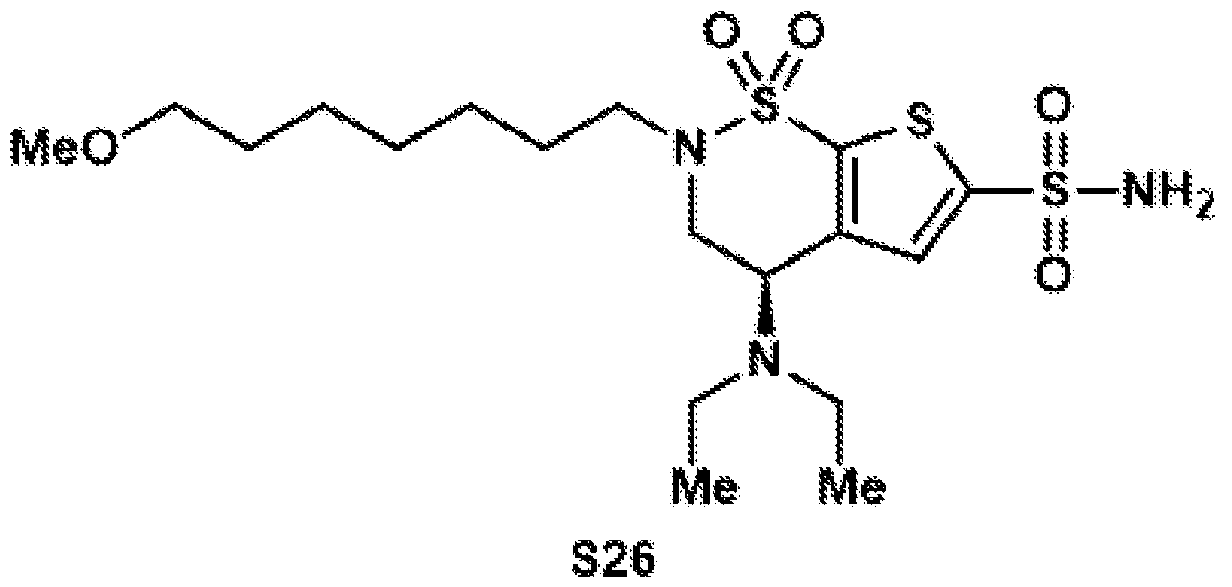
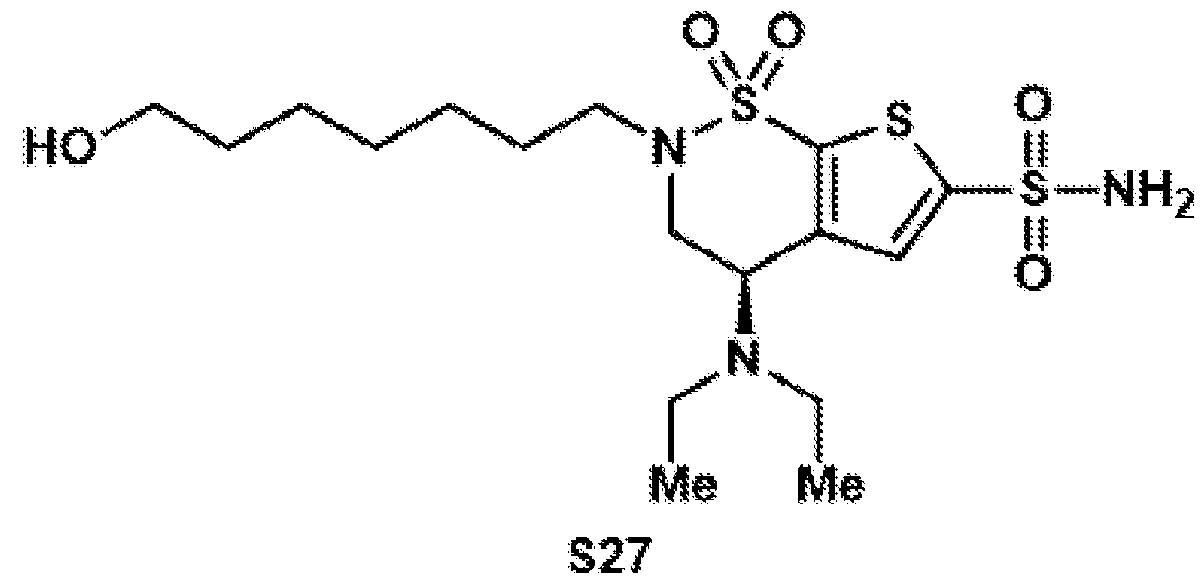
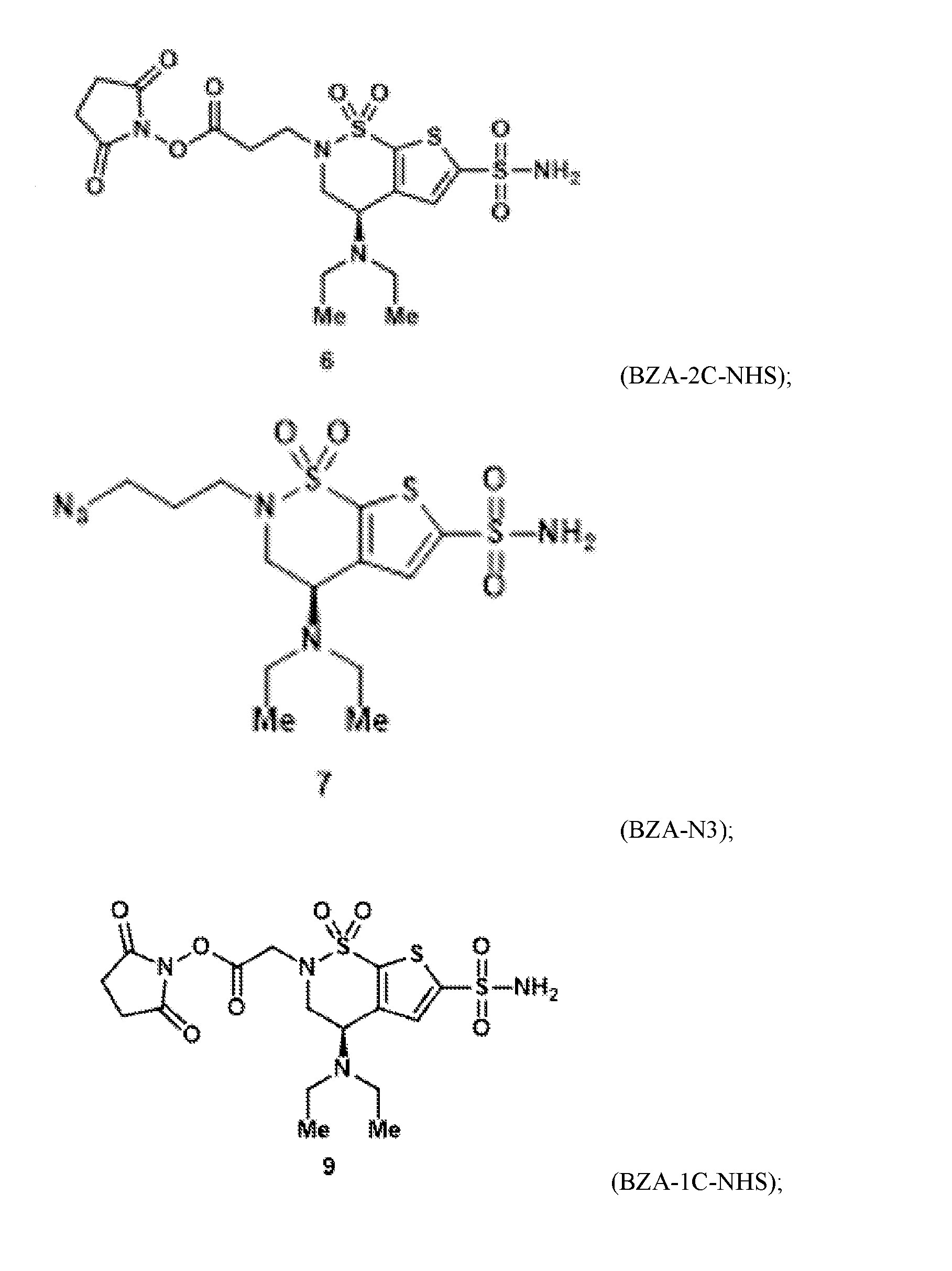
- the brinzolamide derivative may be selected from among the compounds:
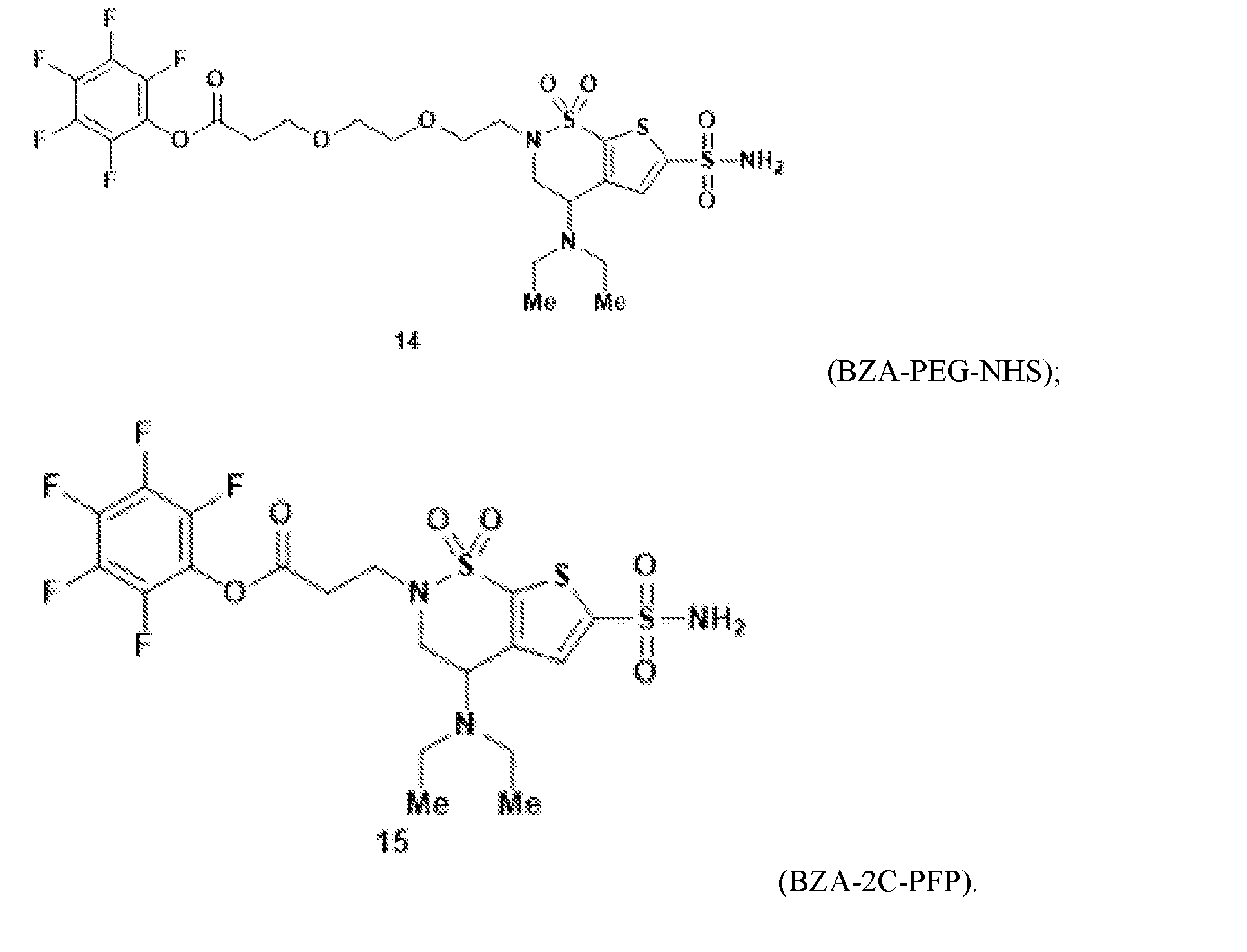
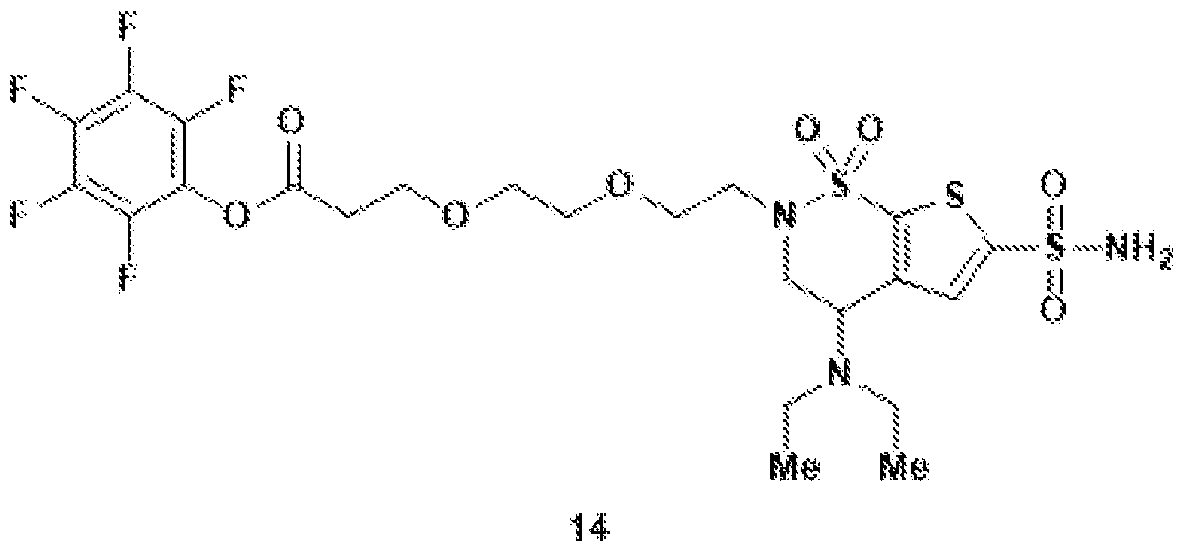
- the brinzolamide derivative may comprise a pentafluorophenyl (PFP) ester.
- PFP pentafluorophenyl
- brinzolamide may be covalently bonded to the PFP ester via polyethylene glycol.
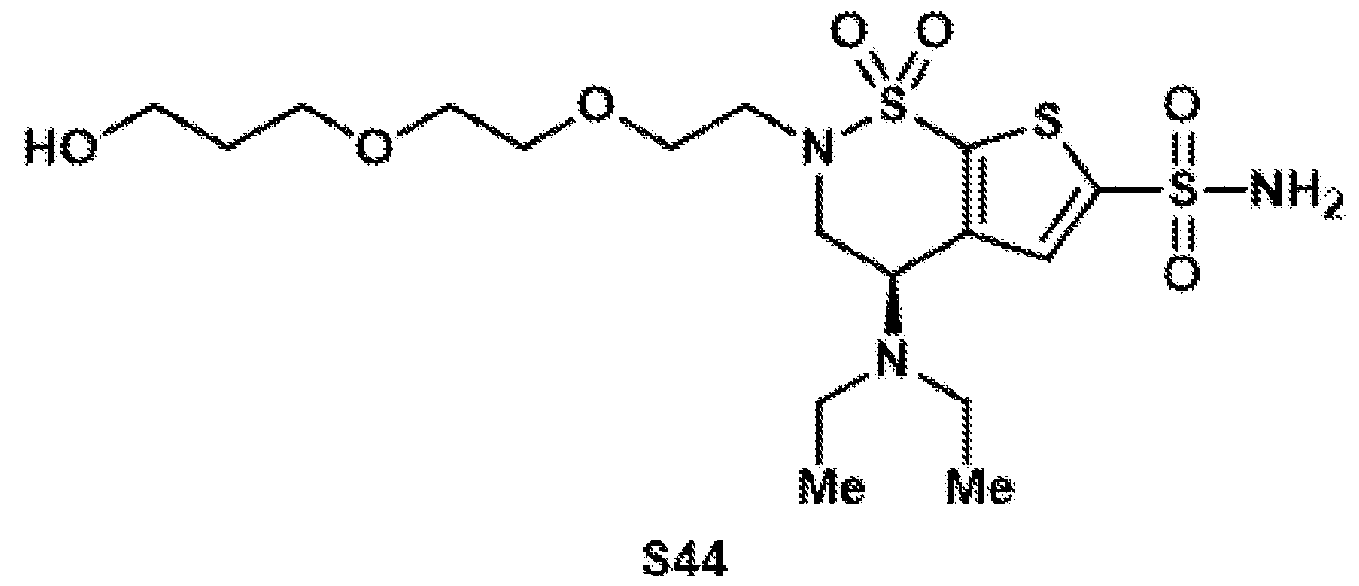
- the brinzolamide derivative may be selected from among the compounds:
- the conjugate may comprise a plurality of small molecule shuttles conjugated to the therapeutic cargo.
- conjugate may comprise 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, or greater than 14 brinzolamide or brinzolamide derivatives conjugated to a single therapeutic cargo.
- the therapeutic cargo may be a biological molecule, for example a nucleic acid (for example, RNA, siRNA, DNA, or an ASO), a protein (for example, an enzyme), a peptide, an antibody, a nanobody, a lipid, a polysaccharide, and a combination thereof.
- the therapeutic cargo may be a non-biological molecule, for example a small molecule or a dye.
- the therapeutic cargo may be conjugated to brinzolamide or a brinzolamide derivative via N-hydroxysuccinimide (NHS) ester coupling or copper(I)-catalyzed azide-alkyne cycloaddition (CuAAC).
- the shuttle may be a carbonic anhydrase IV (CA-IV) shuttle.
- CA-IV carbonic anhydrase IV
- the therapeutic cargo may be a therapeutic cargo for the treatment of a disorder affecting the central nervous system.
- the CA-IV shuttle may be a shuttle for human CA-IV.
- Aspects of the invention further provide methods of delivering therapeutic cargo across the BBB of a subject. Methods of the invention comprise providing to a subject a conjugate comprising brinzolamide or a brinzolamide derivative and a therapeutic cargo conjugated to the shuttle.
- FIG.1 is a schematic of CA-IV mediated transcytosis and antibody conjugation of the invention.
- FIG.2A is a structural analysis of the murine CA-IV binding pocket and its interaction with designed AAVs, BZA, and activated BZA (BZA-2C-NHS).
- FIG.2B is a structural alignment of BZA across CA-IV homologues.
- FIG.3 shows an 1 H-NMR verification of chemical synthesis of NHS-ester-brinzolamide.
- FIG.4A is a structural diagram showing Ate conjugation to BZA using linker 2C.
- FIG.4B-G are LC-MS graphs of BZA-2C-Ate conjugation.
- FIG.5A-D are graphs of results from a Surface Plasmon Resonance (SPR) assays of CA- IV and BZA-2C-Ate.
- FIG.5E shows immunofluorescent images of live CA-IV expressing cells incubated with either unconjugated or BZA-conjugated Ate.
- FIG.5 F-G show the average and counted puncta, respectively, intensity of CA-IV- expressing HeLa cells incubated with either unconjugated or BZA-2C-Ate conjugates.
- FIG.6A shows the structure of select BZA-Ate linker variants.
- FIG.6B-M shows LC-MS graphs of unmodified Ate and BZA-Ate linker variant conjugates.
- FIG.7A-C are graphs of results from SPR assays of CA-IV and BZA-Ate linker variant binding.
- FIG.7D shows immunofluorescent images of live CA-IV expressing cells incubated with either unconjugated or BZA-conjugated Ate linker variants.
- FIG.7E-F show representative live cell images showing CA-IV-expressing HeLa cells incubated with either unconjugated or BZA-conjugated antibodies after 3 hours and 6 hours of incubation with antibodies, respectively.
- FIG.7G-4H show the average and counted puncta, respectively, intensity of CA-IV- expressing Hela cells incubated with either unconjugated or BZA-conjugated linker variants after 3 hours.
- FIG.7I-4J show the average and counted puncta, respectively, intensity of CA-IV- expressing Hela cells incubated with either unconjugated or BZA-conjugated linker variants after 6 hours.
- FIG.8A shows the structures of NHS-ester variants of two alternative CA-IV binders, acetazolamide (AZA) and dorzolamide (DZA).
- FIG.8B-E show LC-MS graphs of NHS ester variants of two alternative CA-IV binders, AZA and DZA respectively.
- FIG.8F-G is a graph of results from an SPR assay of CA-IV and NHS ester variants of two alternative CA-IV binders, AZA and DZA respectively.
- FIG.8H-I show live cell images showing CA-IV-expressing Hela cells incubated with either AZA-conjugated or DZA-conjugated Ate, respectively.
- FIG.8J-K show the average and counted puncta intensity, respectively, of CA-IV- expressing Hela cells incubated with either unconjugated, AZA-conjugated, or DZA-conjugated Ate.
- FIG.9A shows the structures of an IgG antibody BZA variant conjugates.
- FIG.9B-K show LC-MS graphs of BZA-IgG antibody conjugates including Don.
- FIG.10A-E show graphs of the results from an SPR assay of CA-IV and BZA- conjugated Don, unconjugated Don, and BZA-conjugated higG1 isotype, respectively.
- FIG.10F shows representative immunofluorescent and live cell images showing CA-IV- expressing Hela cells incubated with either unconjugated-IgG of BZA-conjugated variant antibodies including Don.
- FIG.10G-H show the average and counted puncta intensity, respectively, of CA-IV- expressing Hela cells incubated with either unconjugated or BZA conjugated antibody variants.
- FIG.11A-D show SPR of modified and unmodified therapeutic IgG antibodies tested against purified human CA-IV and mouse CA-IV, including atezolizumab and Don.
- FIG.11E-F show internalization assays in cultured Hela cells with the BZA-modified IgG antibodies and unmodified IgG antibodies, including atezolizumab and Don.
- FIG.12A-B show the structure of a BZA-nanobody variants.
- FIG.12C-D show the LC-MS analysis of unmodified nanobodies and nanobodies BZA- conjugated nanobodies.
- FIG.12E-H show graphs of the results from an SPR assay of the binding interaction between an unconjugated or BZA-conjugated nanobody and Fc-tagged CA-IV proteins.
- FIG.12I show representative immunofluorescent images showing CA-IV-expressing HeLa cells incubated with either unconjugated or BZA-conjugated anti-GFP VHH nanobody.
- FIG.13A-B shows graphs of the results from an SPR assay of modified and unmodified nanobodies tested against purified human CA-IV and mouse CA-IV.
- FIG.13C-E show representative immunofluorescent images showing CA-IV-expressing Hela cells incubated with either anti-GFP VHH unconjugated nanobodies, BZA-2C conjugated nanobodies, and BZA-3C conjugated nanobodies.
- FIG.14A shows the structure of a BZA shuttle-siRNA variant.
- FIG.14B-D shows graphs of the results from an SPR assay of the binding interaction between an unconjugated or BZA-conjugated siRNA duplex and Fc-tagged CA-IV proteins.
- FIG.14E shows representative immunofluorescent images showing CA-IV-expressing Hela cells incubated with either unconjugated fluorescently labeled siRNA or BZA-2C conjugated siRNA.
- FIG.15A shows the structure of a BZA shuttle-siRNA variant.
- FIG.15B-C show graphs of the results from an SPR assay of the binding interaction between an unconjugated or BZA-conjugated siRNA duplex and Fc-tagged CA-IV proteins.
- FIG.16A shows the structure of a BZA shuttle-small molecule cargo variant.
- FIG.16B show representative immunofluorescent images showing CA-IV-expressing Hela cells incubated with either unconjugated small molecule dye or BZA-conjugated small molecules dye.
- FIG.16C shows the structure of a BZA shuttle-small molecule cargo variant.
- the schematic shows the process of conjugating a fluorescent molecule (Alexa Fluor 647) to brinzolamide using a CuAAC reaction.
- FIG.16D show images from an internalization assay using the HEK cells overexpressing CA-IV receptors. The bottom row shows zoom-in images of representative fields of views in the top-row images.
- FIG.16E shows representative images of brain slices and liver slices from mice injected with BZA-conjugated fluorophore or unconjugated fluorophores.
- FIG.16F shows a fluorescence reading of lysed tissues of animals injected with BZA- conjugated fluorophore or unconjugated fluorophores using a plate reader.
- FIG.18A-B show immunofluorescence images of whole livers sections in mice systemically administered with unconjugated and BZA-conjugated Ate antibodies.
- FIG.18C shows quantification of BZA-conjugated and unconjugated Ate antibodies in peripheral organs at day 7 in mice systemically administered with unconjugated and BZA- conjugated Ate antibodies.
- the present invention provides compositions and methods comprising brinzolamide and brinzolamide derivates as shuttles for the BBB-crossing, for example through the receptor carbonic anhydrase IV (CA-IV).
- CA-IV receptor carbonic anhydrase IV
- Receptors for Enhanced Blood-Brain Barrier Crossing Blood-brain barrier (BBB) has emerged as a complex, dynamic, adaptable interface that controls the exchange of substances between the central nervous system (CNS) and the blood, to prevent the uncontrolled leakage of substances from the blood into the brain.
- the cells that make up the structure of the BBB include mostly brain endothelial cells, which constantly communicate with the other cells of the CNS (e.g., astrocytes, microglia, neurons, mast cells and pericytes, as well as circulating immune cells), adapting their behaviors to serve the needs of the CNS, responding to pathological conditions, and in some cases participating in the onset, maintenance or progression of disease.
- the complexity of BBB functions explains much of the difficulty in developing drugs that can cross the BBB. Utilizing receptors on the BBB interface can offer a method of crossing BBB.
- the present invention provides shuttles for receptors on the BBB interface and methods of using the same to enhance BBB crossing and CNS potency, such as increasing the permeability of the BBB and delivering a therapeutic agent across the BBB to a nervous system, specifically carbonic anhydrase IV.
- the novel target receptors disclosed herein may facilitate enhanced BBB receptor-mediated transcytosis across various species, including mammals such as human.
- a method of increasing permeability of the BBB comprises providing a shuttle capable of binding to a BBB crossing receptor (e.g., carbonic anhydrase IV), thereby increasing permeability of the BBB (e.g., through transcytosis).
- At least one activity of the BBB-crossing receptor can be reduced through binding to a small molecule.
- a method of increasing permeability of the BBB comprises reducing the activity of carbonic anhydrase IV, thereby increasing permeability of the BBB.
- a shuttle binds to one or more of the zinc binding site (e.g., a catalytic pocket) and substrate binding site of the carbonic anhydrase IV.
- the carbonic anhydrase IV can be a vertebrate carbonic anhydrase IV including non-human primates and humans.
- the carbonic anhydrase IV is a mouse carbonic anhydrase IV (Car4), a human carbonic anhydrase IV (CA4), or a variant or a homolog thereof. It is understood that CA-IV as used herein refers to any variant.
- Carbonic Anhydrase IV The carbonic anhydrases (or carbonate dehydratases) (CAs) are a family of enzymes that catalyze the interconversion between carbon dioxide and water and the dissociated ions of carbonic acid. CAs participate in a variety of biological processes, including respiration, calcification, acid-base balance, bone resorption, and the formation of aqueous humor, cerebrospinal fluid, saliva, and gastric acid.
- the present invention provides shuttles for the BBB-crossing using the receptor carbonic anhydrase IV, capable of facilitating the delivery of a pharmaceutical agent across the BBB (CA- IV shuttles).
- Carbonic anhydrase IV is an isozyme that belongs to the carbonic anhydrase family, a family of zinc metalloenzymes, which catalyzes the reversible reaction of hydration of CO 2 (H 2 O+CO 2 ⁇ HCO 3 ⁇ +H+), allowing the enzyme to regulate intra- and extra-cellular concentrations of CO2, H+, and HCO3 ⁇ .
- the carbonic anhydrases participate in a variety of biological processes, including respiration, calcification, acid-base balance, bone resorption, and the formation of aqueous humor, cerebrospinal fluid, saliva, and gastric acid.
- the carbonic anhydrases show extensive diversity in tissue distribution and in their subcellular localization.
- I-VII mammalian carbonic anhydrase
- Physiological functions that are regulated by carbonic anhydrase comprise, for example, removal of HCO3 ⁇ in lung by respiration, reutilization of HCO3 ⁇ in kidney, production of aqueous humor in eyes, cerebrospinal fluids in brain, gastric juice production in stomach, pancreatic juice, and bone resorption by osteoclasts.
- Carbonic anhydrase family members also play important roles in metabolic processes that include ureagenesis, gluconeogenesis, and lipogenesis.
- carbonic anhydrase IV is a glycosylphosphatidyl- inositol-anchored membrane isozyme.
- Carbonic anhydrase IV is broadly conserved across vertebrates and has similar CNS expression profiles in humans, with a recent single cell analysis of human brain vasculature confirming CA-IV's expression in the human BBB. Carbonic anhydrase IV has been shown to regulate pH, which is associated with neural discharge and can influence neuronal function through ion-gated channels. In some embodiments, the carbonic anhydrase IV disclosed herein is a human carbonic anhydrase IV.
- CA-IV is known to localize on the luminal surface of brain endothelial cells throughout the cortex and cerebellum where it enzymatically modulates carbon dioxide- bicarbonate balance.
- Human CA-IV has been previously characterized as a 35-kDa protein with a “high activity” in CO2 hydration and a higher activity than other isozymes in catalyzing the dehydration of HCO3 ⁇ .
- human CA-IV contains an 18-amino acid signal sequence at the N-terminal of the protein for endoplasmic reticulum (ER) translocation and a 260-amino acid “CA domain” containing active site amino acid residues that shows 30-36% homology with cytoplasmic CAs.
- ER endoplasmic reticulum
- CA domain 260-amino acid “CA domain” containing active site amino acid residues that shows 30-36% homology with cytoplasmic CAs.
- an additional 27 amino acid residues containing the hydrophobic sequences of 21 amino acids sufficient to span the membrane are preceded by the 6- amino acid signal sequence for GPI-anchoring.
- human CA-IV contains no classical consensus sites (Asn-Xxx-Ser/Thr) for N-glycosylation. Human CA-IV also contains no oligosaccharide chains, while other mammalian carbonic anhydrase IV (e.g. mouse carbonic anhydrases IV) are glycoproteins with one to several oligosaccharide side chains.
- the carbonic anhydrase IV disclosed herein is a mouse carbonic anhydrase IV.
- CA-IV has recently been found to be among the mouse proteins most strongly positively correlated with plasma-protein uptake in the brain (slightly stronger than the often- targeted transferrin receptor). This property is useful for identifying receptors for enhanced BBB crossing.
- CA-IV is also expressed in the GI tract, kidney, and lung, as well as taste receptor cells where it allows the sensing of carbonation.
- Mouse and human CA-IV are highly homologous, containing the same amino acids at positions crucial for enzyme activity (e.g., histidine residue 64 (His 64)), with several differences including, for example, that mouse CA-IV is an N-linked glycoprotein and the CO2 hydration rate catalyzed by mouse CA-IV is much lower than human CA-IV. Without being bound by any theory, the lower enzyme activity of mouse CA-IV may be associated with the replacement of Gly 63 in human CA-IV with Gln 63, among several other amino acid replacements.
- Another difference between mouse and human CA-IV is the Val-131- Asp-136 segment (130's segment) that forms an ⁇ -helix in mouse and an extended loop in human CA-IV.
- a carbonic anhydrase IV disclosed herein as a receptor for enhancing BBB crossing can be any carbonic anhydrase IV, such as a mouse CA-IV, a humanCA-IV, or a homology or a variant thereof.
- Carbonic anhydrase IV homologs and/or variants can be derived from a vertebrate species including, but not limited to, mouse, rat, human, bovine, rabbit, monkey, pig, horse, rainbow trout, chimpanzee, squirrel, chicken, goat, and sheep.
- Carbonic anhydrase IV homologs from various species can be found in public databases identifiable to a person skilled in the art, including for example UniProt, NCBI, and Swiss-Prot.
- a small molecule can interact with a carbonic anhydrase IV disclosed herein (e.g., mouse CA-IV, human CA_IV or a homology or a variant thereof), thereby increasing permeability of the BBB (e.g., through transcytosis).
- the increase in the permeability of the BBB is achieved by altering (e.g., increasing or decreasing) the carbonic anhydrase IV activity, such as reducing its activity.
- the alteration of carbonic anhydrase IV activity is achieved by a shuttle interacting to one or more active sites of the carbonic anhydrase IV including the zinc binding site and the hydrophobic substrate binding pocket.
- the shuttle can interacting with the zinc binding site, the hydrophobic substrate binding pocket, or both.
- the zinc binding site in carbonic anhydrase IV has a conserved structure dominated by a ⁇ -sheet super-structure with a metal binding site formed by at least three His residues. Without being bound by any theory, it is believed that the zinc binding site is on one face of the ⁇ -sheet at the bottom of a 15- ⁇ -deep, conical active site cleft in which zinc is liganded by three His residues and hydroxide ion with tetrahedral geometry.
- the hydrophobic substrate binding pocket is adjacent to zinc-bound hydroxide, formed in large part by bulky residues such as Val at its base and Val, Trp and Leu at its neck.
- This pocket is highly conserved among all active isozymes on the basis of phylogenetic comparisons. Without being bound by any theory, it is believed that the hydrophobic pocket has a minimum width and depth for efficient catalysis, and linear free energy relationships indicate that the volume of the amino acid residue at the base of the pocket and the hydrophobicity of residues at the neck of the pocket are critical for activity. Both the zinc binding site and the hydrophobic substrate binding pocket are highly conserved among carbonic anhydrase isozymes.
- Brinzolamide and brinzolamide derivatives
- Brinzolamide is a highly specific, non-competitive, reversible carbonic anhydrase II (CA II) inhibitor indicated to reduce ocular pressure in patients with ocular hypertension or open-angle glaucoma.
- Brinzolamide was approved by the FDA in 1998 as a topical product under the trade name AZOPT and later as combination products with timolol under the trade name AZARGA and brimonidine tartrate under the trade name SIMBRINZA.
- N-Desethylbrinzolamide is an active metabolite of brinzolamide, exhibiting CA 1 inhibitory activity, when in the presence of Brinzolamide, and also accumulates in the erythrocytes.
- Brinzolamide's other known metabolites either have no activity or their activity is currently unknown.
- the structure for Brizolamide is: with a molecular formula of C12H21N3O5S3 and IUPAC name (4R)-4-(ethylamino)-2-(3- methoxypropyl)-1,1-dioxo-3,4-dihydrothieno[3,2-e]thiazine-6-sulfonamide.
- brinzolamide and particularly brinzolamide derivates, may act as shuttles for receptors on the BBB interface and methods of using the same to enhance BBB crossing and CNS potency, such as increasing the permeability of the BBB and delivering a therapeutic agent across the BBB to a nervous system, specifically carbonic anhydrase IV.
- This invention proposes an innovative method for surmounting this challenge using receptor-mediated transcytosis (RMT), capitalizing on Carbonic Anhydrase IVCA-IV as a BBB the first time that brinzolamide, a carbonic anhydrase inhibitor, binds to the mouse CA-IV at the same catalytic site as some BBB-crossing capsids (including 9P31 and 9P36), implying a potential mechanism for transporting therapeutic agents across the BBB.
- Brinzolamide showcases binding affinity to CA-IV, with a half maximal inhibitory concentration (IC50) of 45 nM.
- brinzolamide (BZA) derivatives may be used in the instant invention to conjugate brinzolamide (BZA) derivatives with therapeutic cargoes, including nanobodies, therapeutic IgG antibodies, and small molecules.
- BZA brinzolamide
- therapeutic cargoes including nanobodies, therapeutic IgG antibodies, and small molecules.
- BZA brinzolamide
- CHS lysine–N- hydroxysuccinimide
- CuAAC Copper(I)-catalyzed azide-alkyne cycloaddition
- BZA brinzolamide
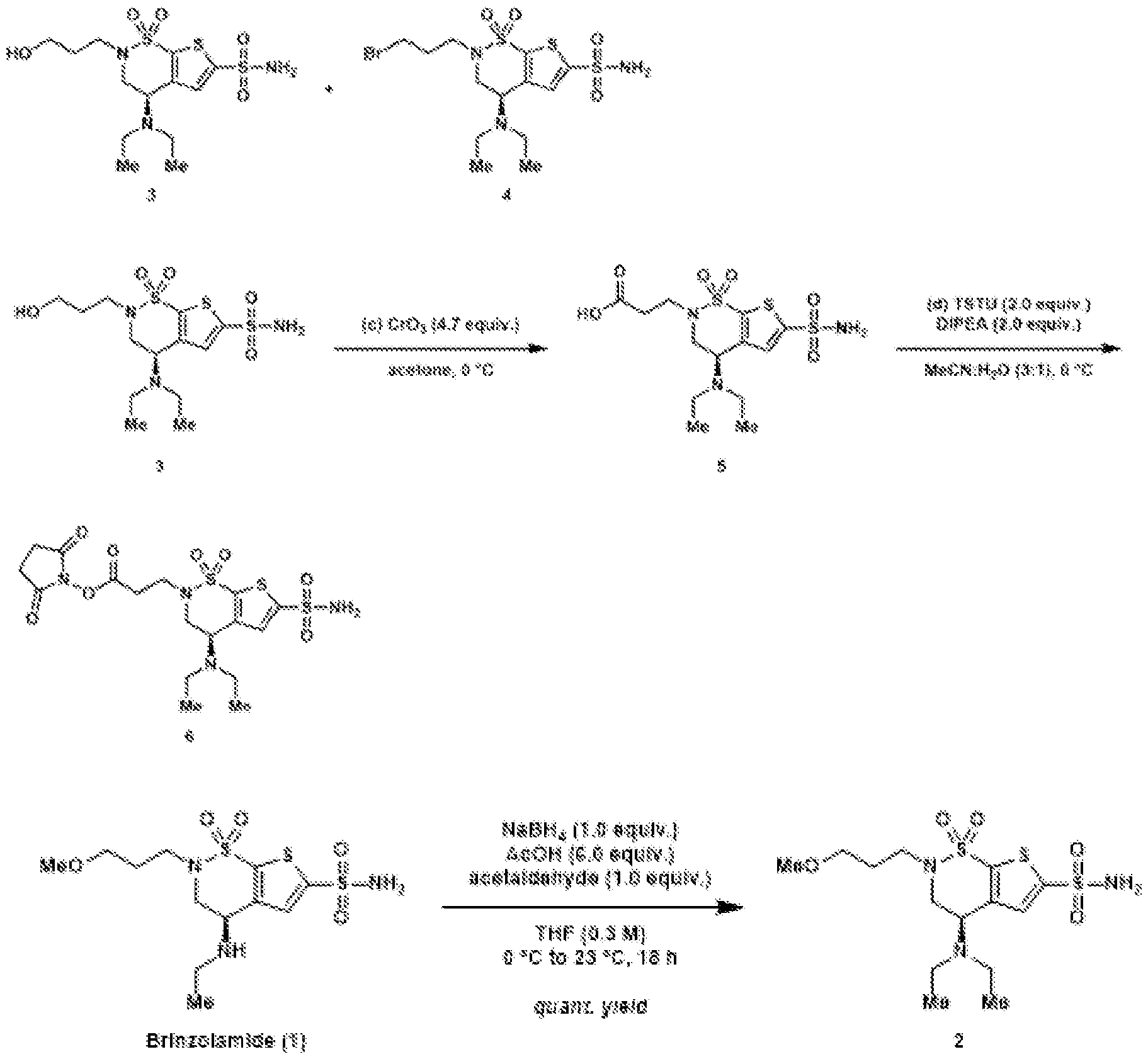
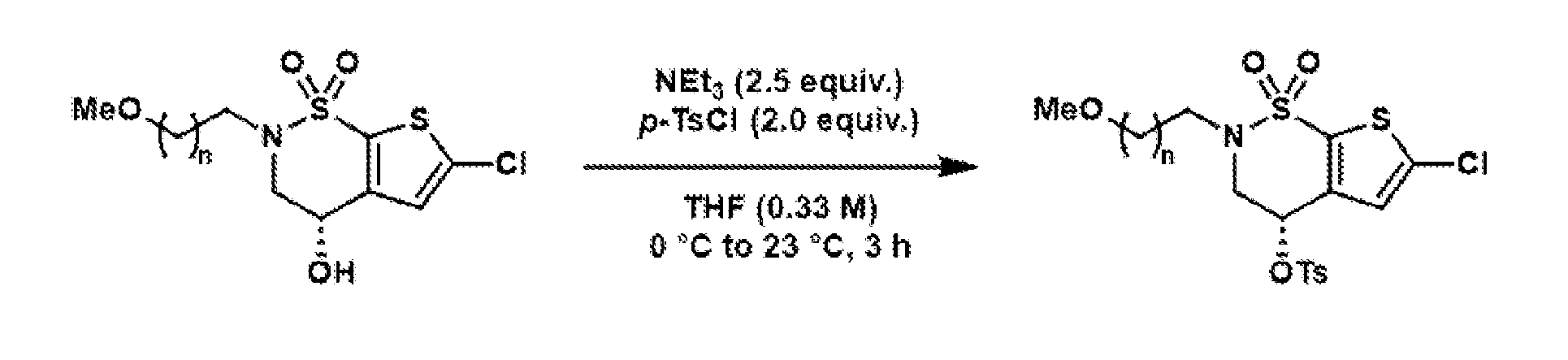
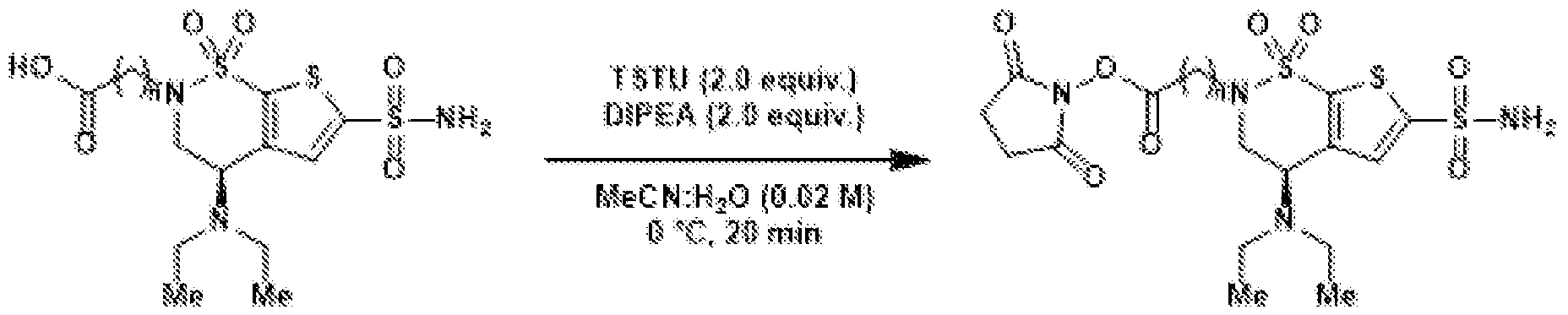
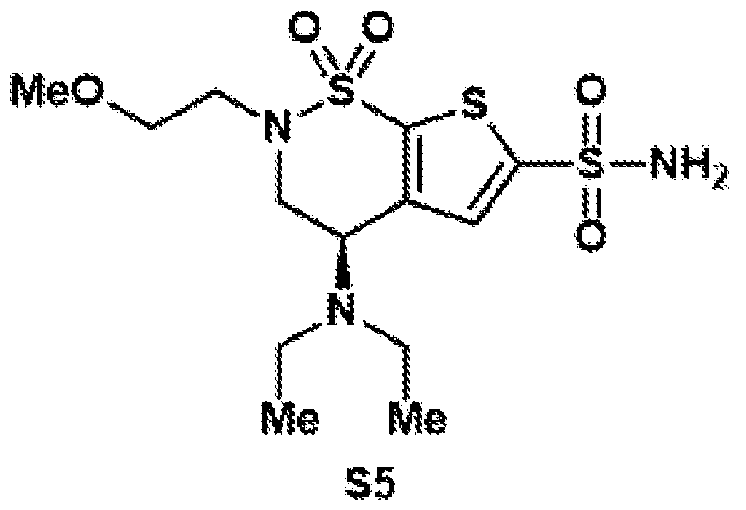
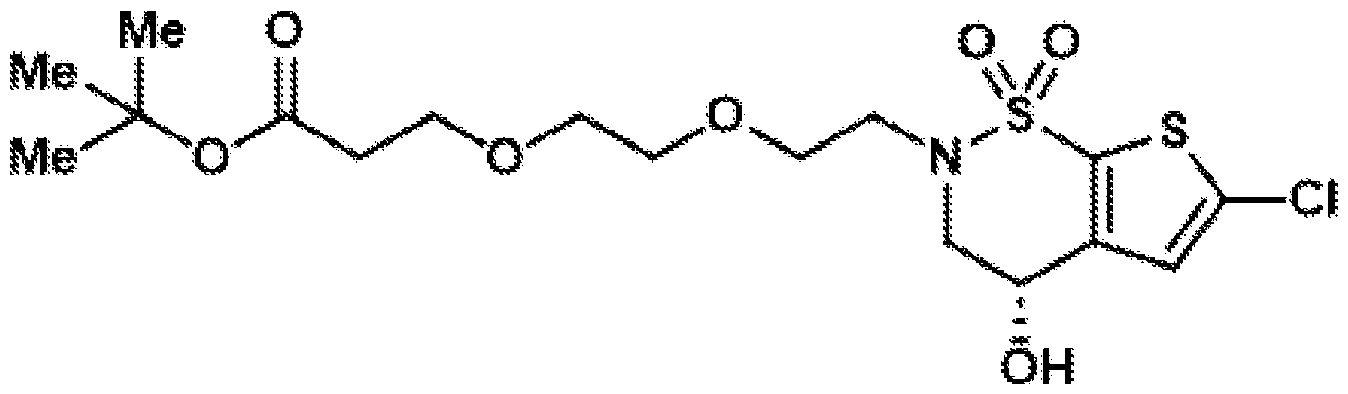
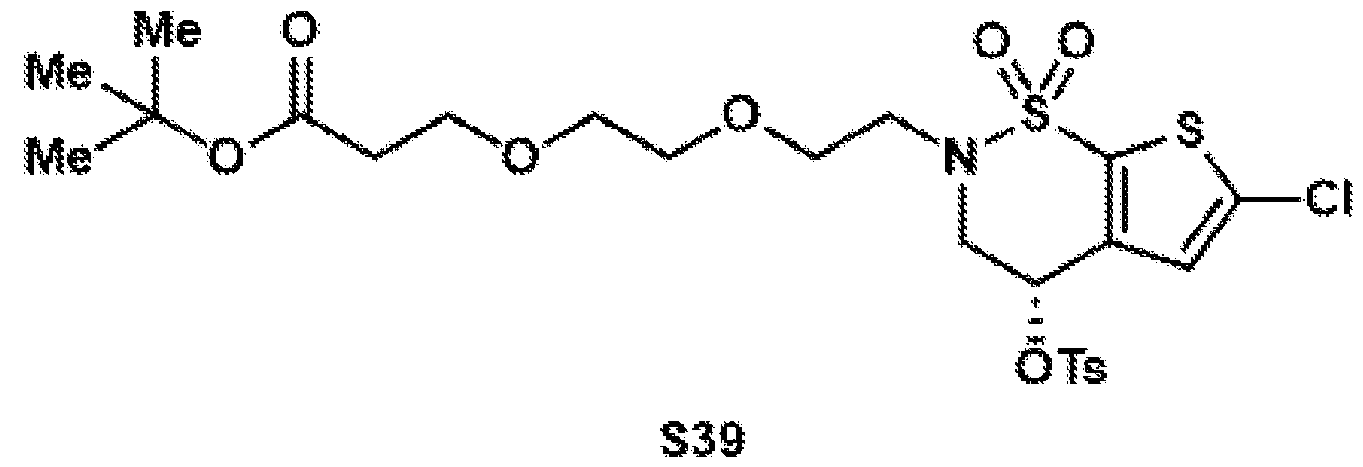
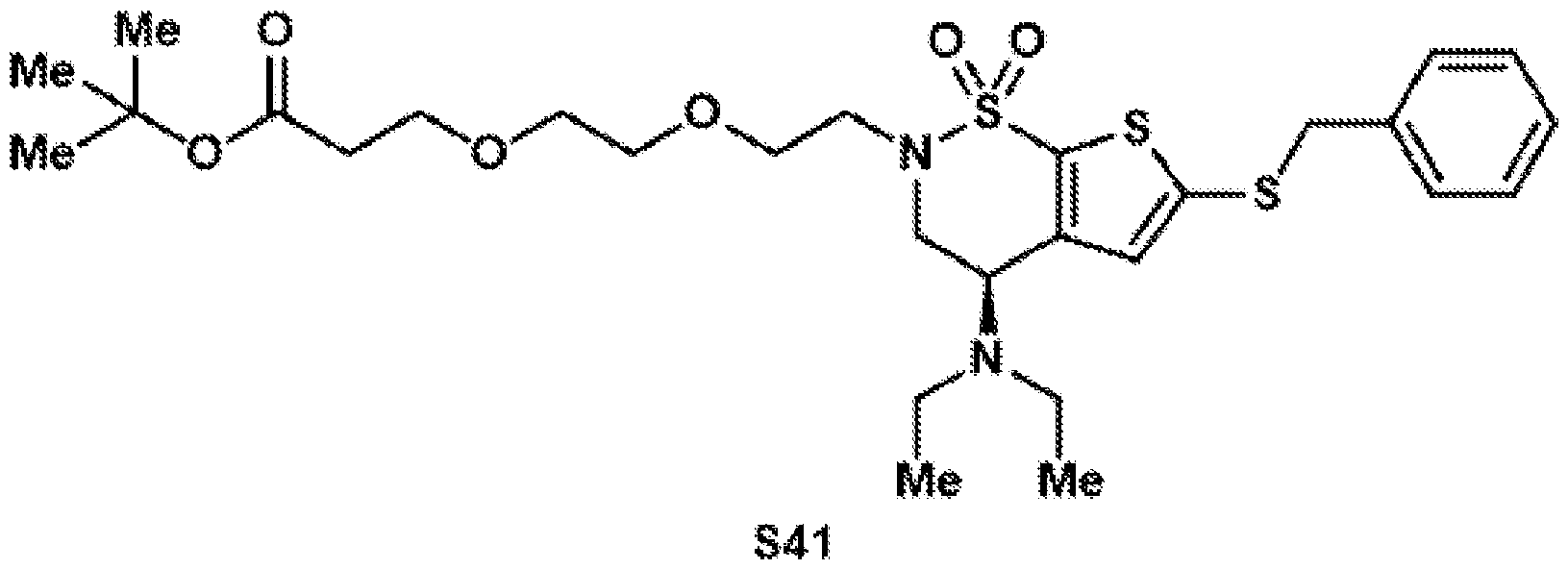
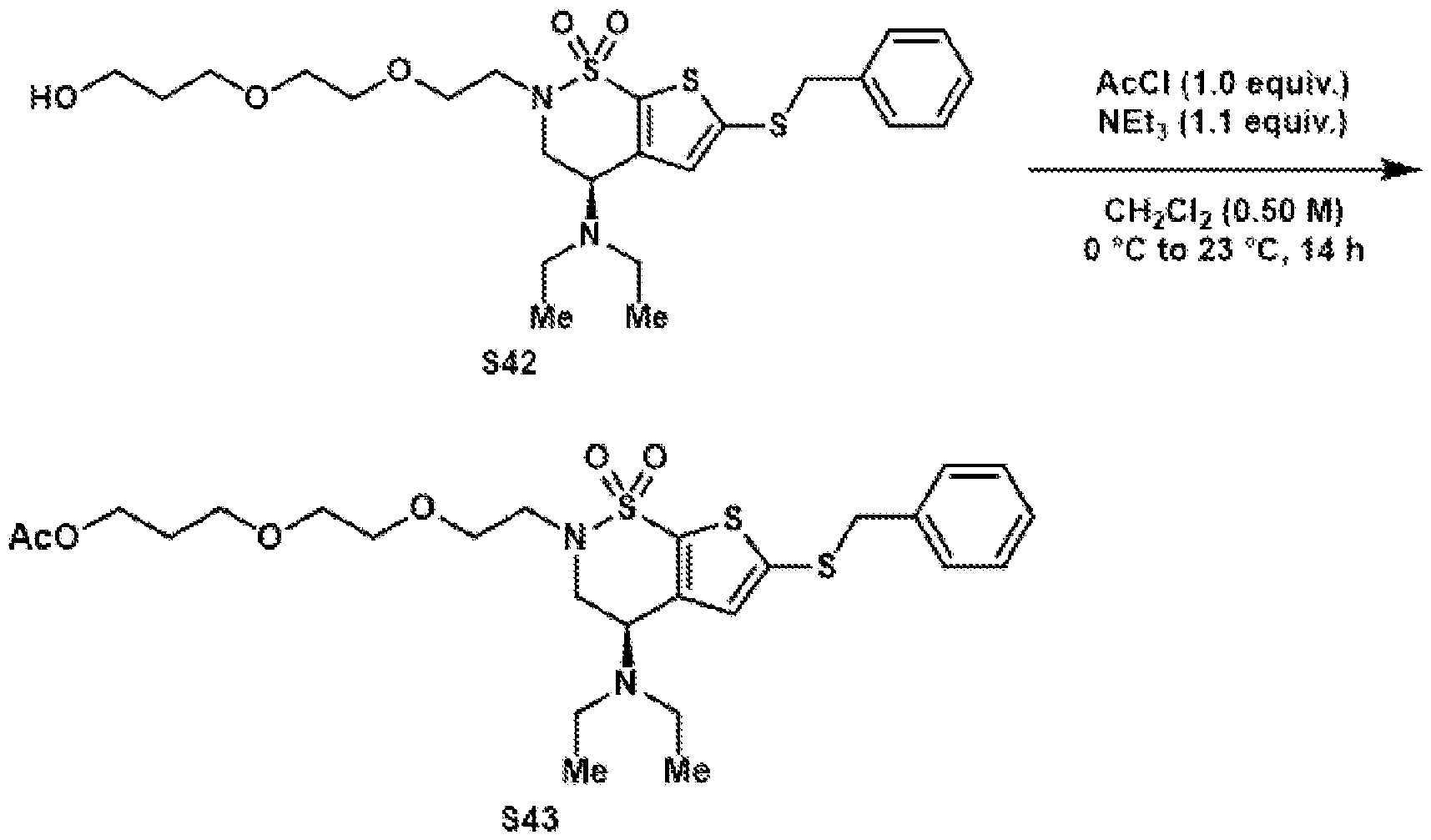
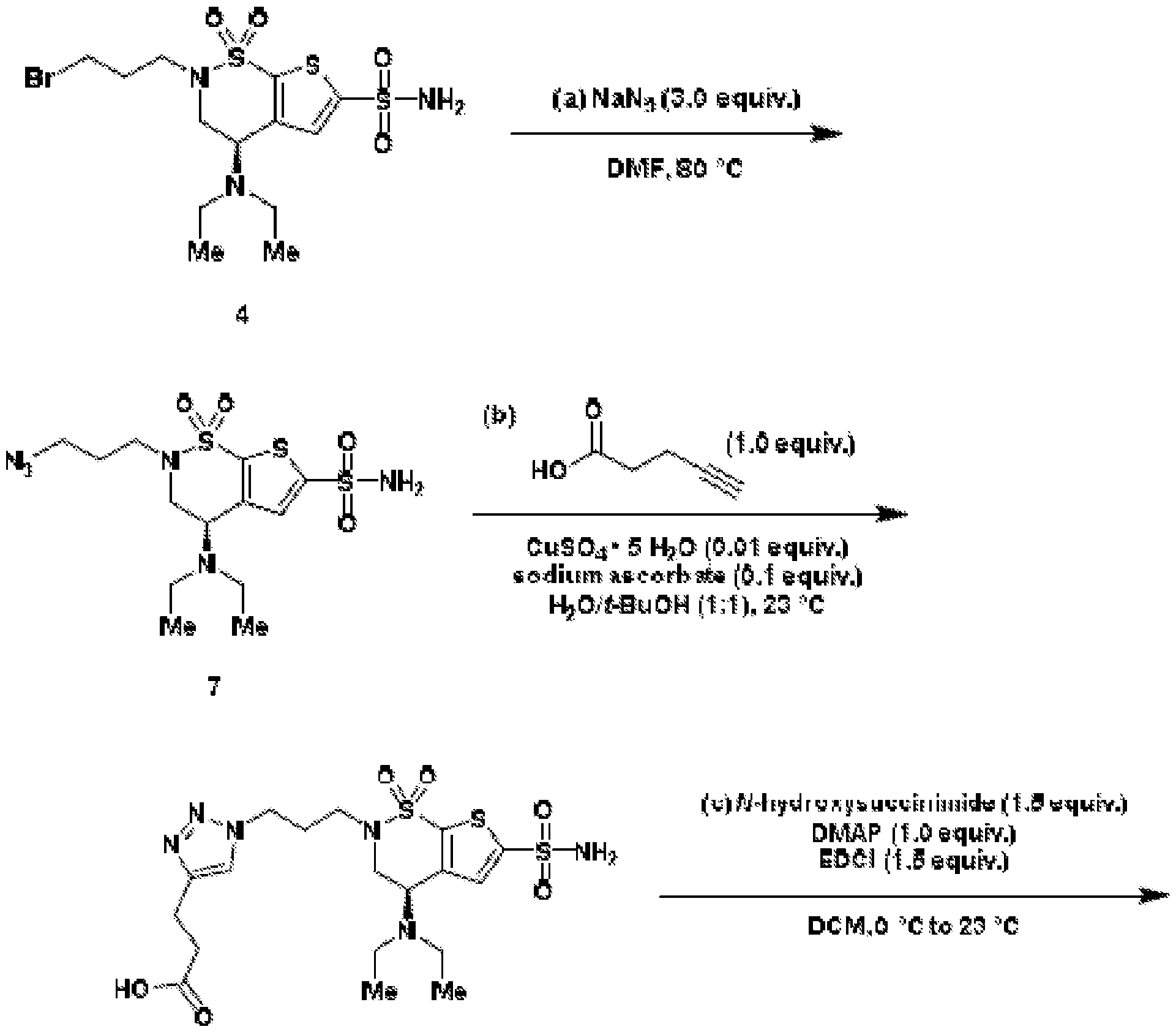
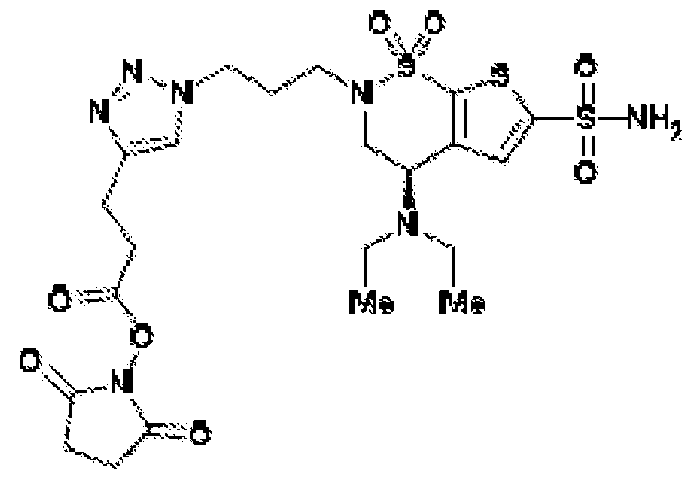
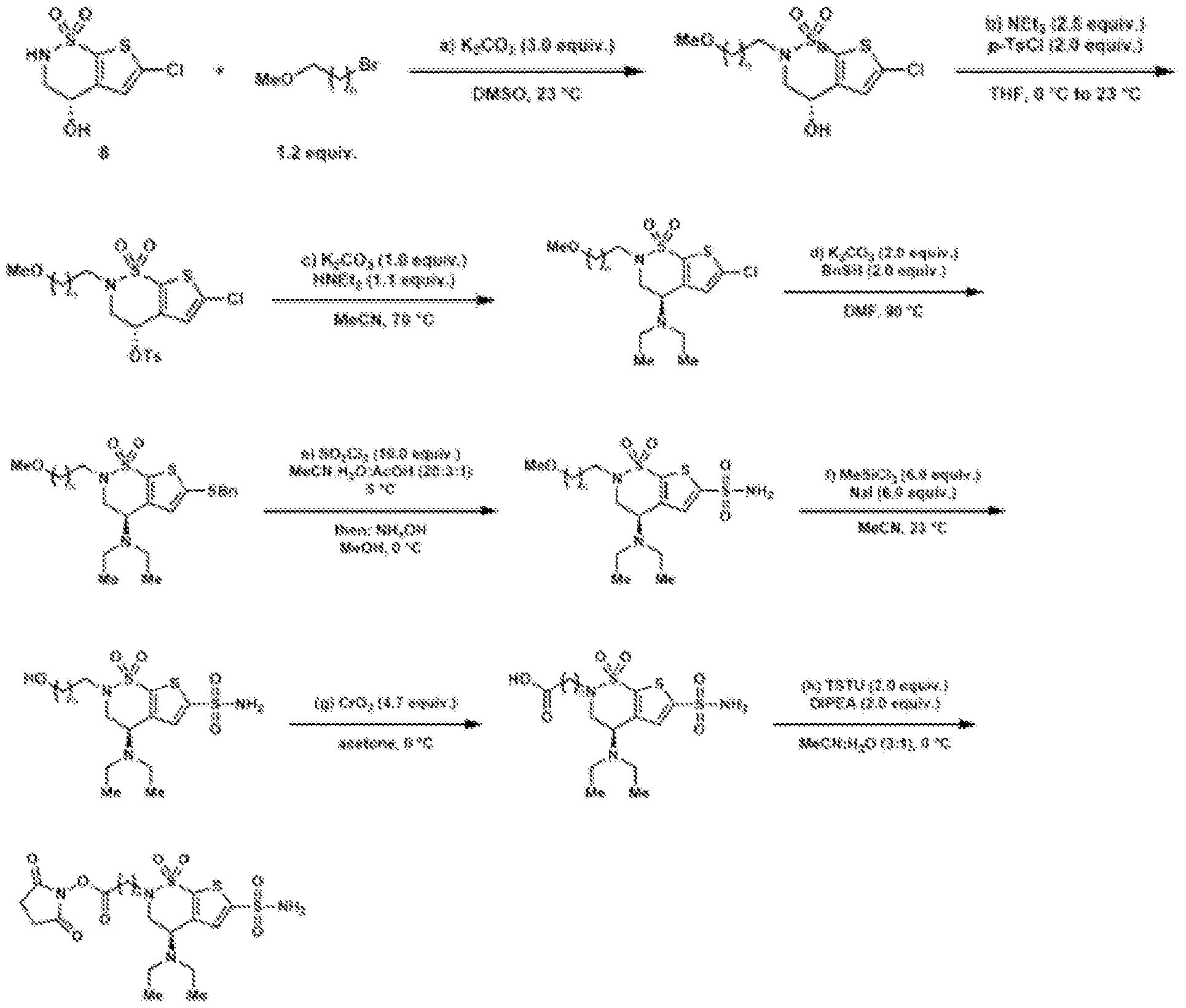
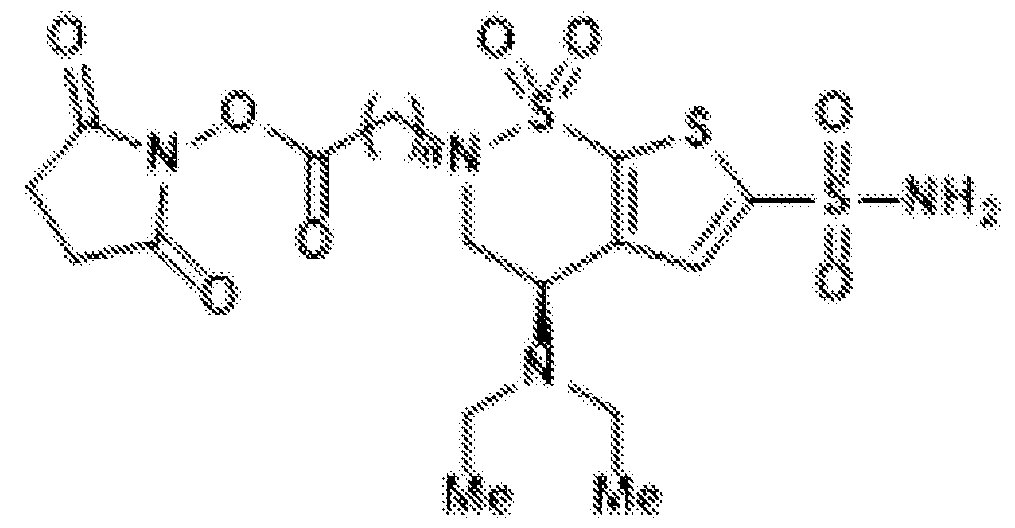
- aspects of the invention provide brinzolamide derivates synthesized using the linkers and esters as described below: BZA-2C-NHS (1-6)
- N,N,N′,N′-tetramethyl-O-(N-succinimidyl)uronium tetrafluoroborate (TSTU) (36.6 mg, 122 ⁇ mol, 2.0 equiv.) and N,N-diisopropylethylamine (21.2 ⁇ L, 15.7 mg, 122 ⁇ mol, 2.0 equiv.) were added. After 20 minutes at 0 ⁇ C, the volatile materials were evaporated under reduced pressure.
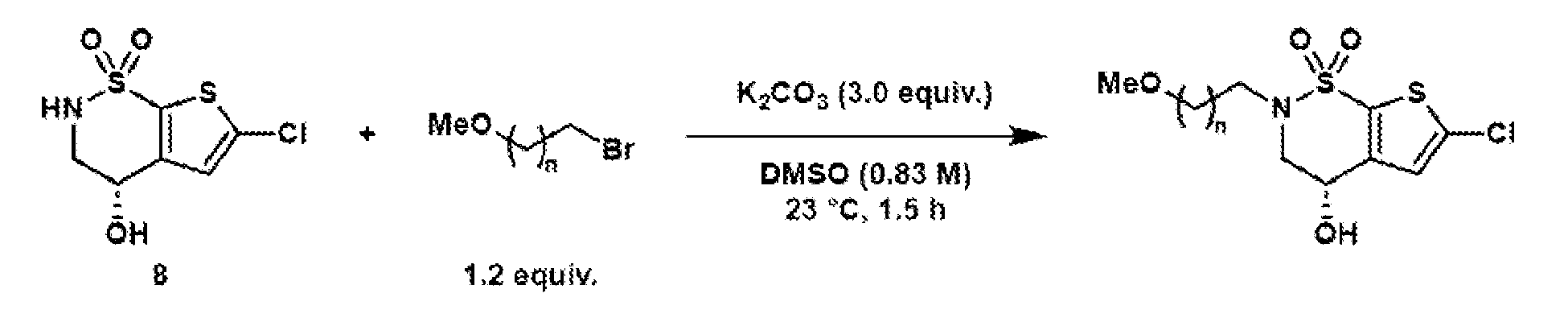
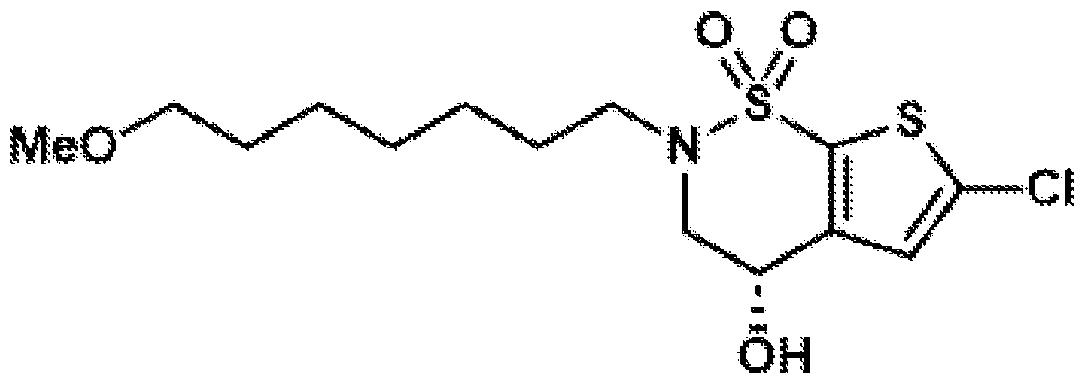
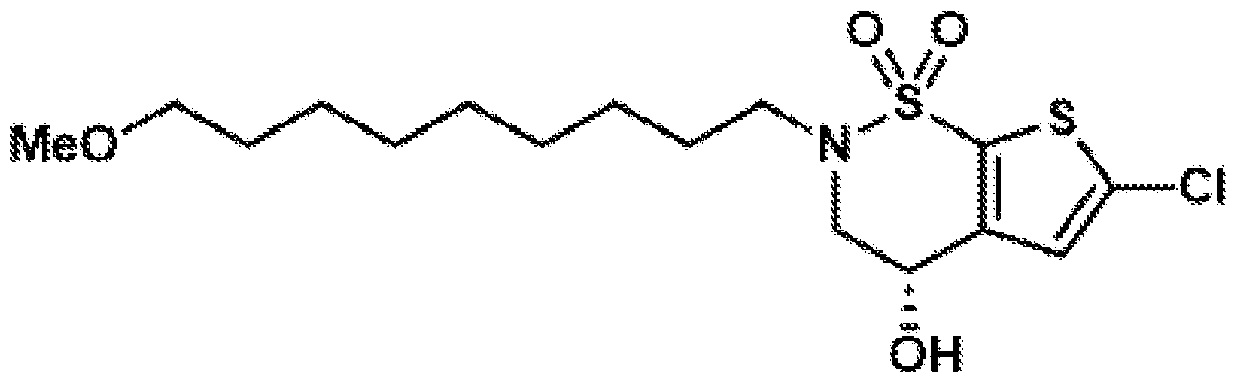
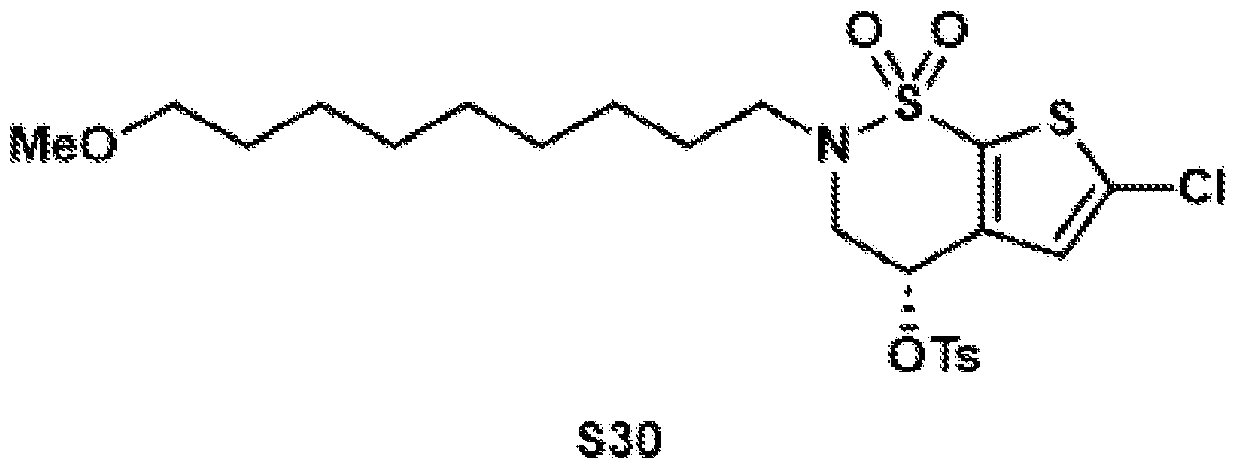
- General procedure 1 Alkylation of the BZA core (8)
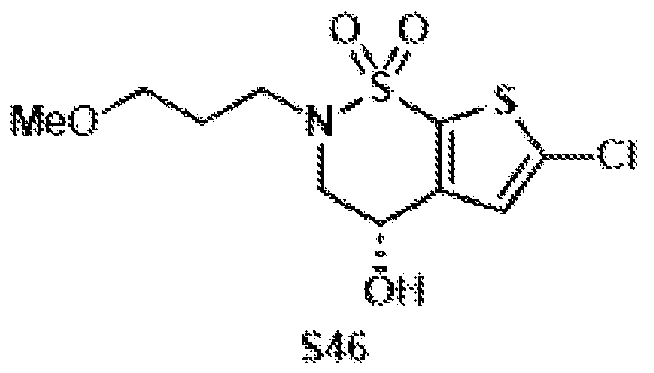
- (S)-6-chloro-4-hydroxy-3,4- dihydro-2H-thieno[3,2-e][1,2]thiazine 1,1-dioxide (8) (2.40 g, 10.0 mmol, 1.0 equiv.) was dissolved in DMSO (12.0 mL), and potassium carbonate (4.15 g, 30.0 mmol, 3.0 equiv.) was added.
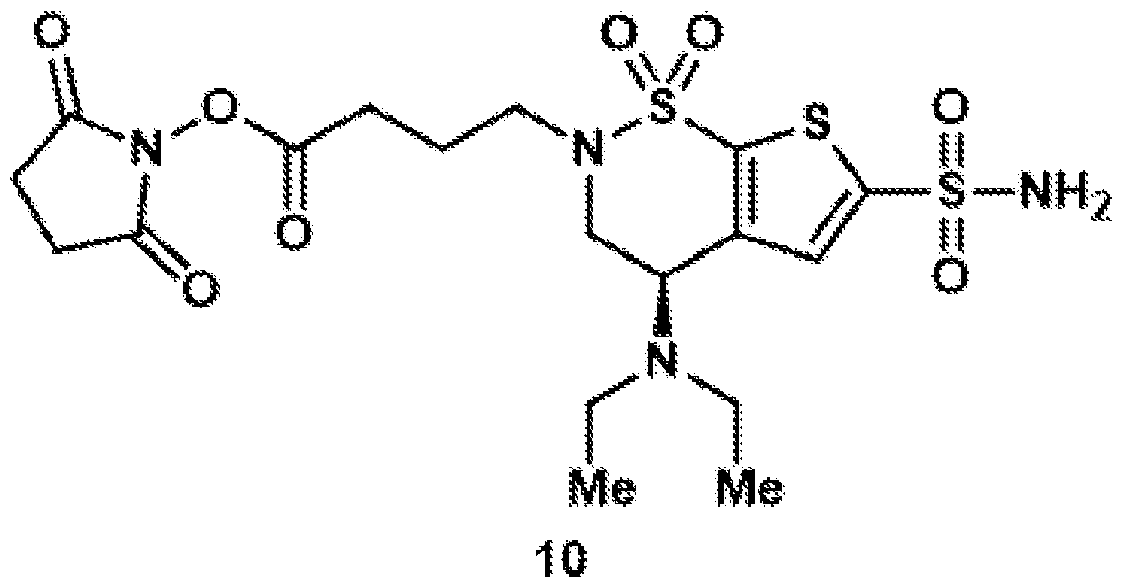
- Compound 10 was synthesized according to general procedure 8 on a 117 ⁇ mol scale. After purification by preparative HPLC (C18 column, 9.4 x 250 mm), the target compound was isolated as a colorless oil (22.2 mg, 42.5 ⁇ mol, 36%).
- Compound S21 was synthesized according to general procedure 7 on a 557 ⁇ mol scale. Since the compound is not stable on silica, the crude reaction mixture was directly used for the next step after filtration through a cotton plug and evaporation. Compound 11 was synthesized according to general procedure 8 on a 114 ⁇ mol scale. After purification by preparative HPLC (C18 column, 9.4 x 250 mm), the target compound was isolated as a colorless oil (6.20 mg, 11.6 ⁇ mol, 10%).
- Compound 13 was synthesized according to general procedure 8 on a 101 ⁇ mol scale. After purification by preparative HPLC (C18 column, 9.4 x 250 mm), the target compound was isolated as a colorless oil (25.2 mg, 42.5 ⁇ mol, 42%).
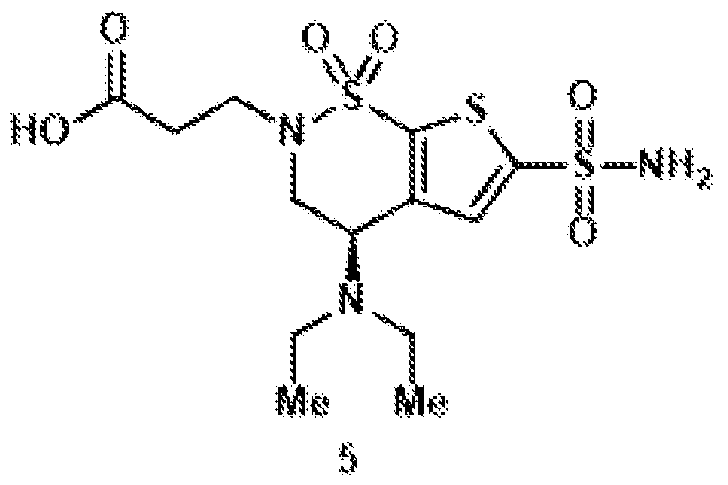
- Compound 5 Compound 5 was synthesized according to general procedure 7 on a 153 ⁇ mol scale.
- N-(3- Dimethylaminopropyl)-N′-ethylcarbodiimide hydrochloride 5.12 mg, 26.7 ⁇ mol, 1.1 equiv.
- pentafluorophenol 4.92 mg, 26.7 ⁇ mol, 1.1 equiv.
- DMAP 0.3 mg, 2.43 ⁇ mol, 0.1 equiv.
- the reaction was stirred overnight at room temperature. Afterwards, the volatile materials were evaporated under reduced pressure.
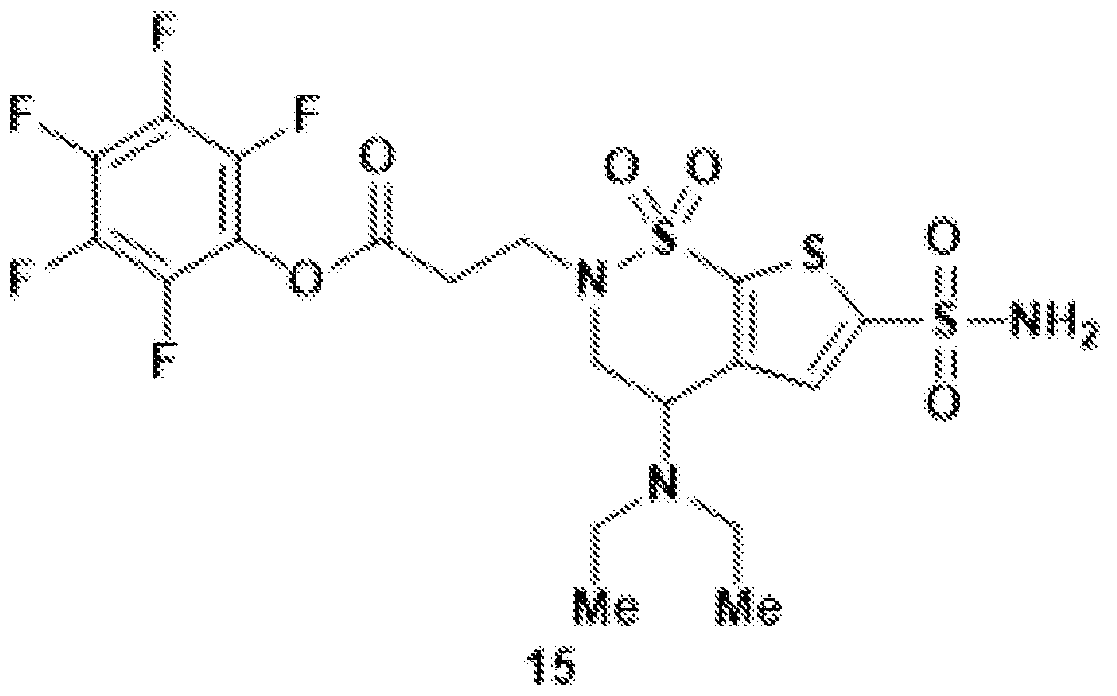
- the crude material was purified by preparative HPLC (C18 column, 9.4 x 250 mm) to obtain the target compound 15 (8.2 mg, 14.2 ⁇ mol, 58%) as a colorless oil.
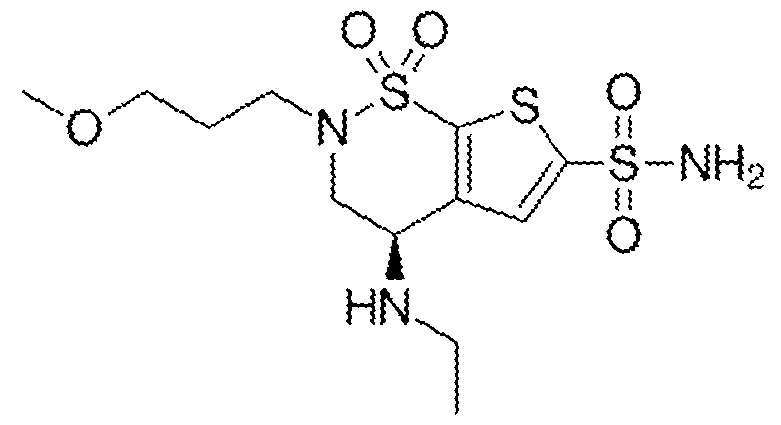
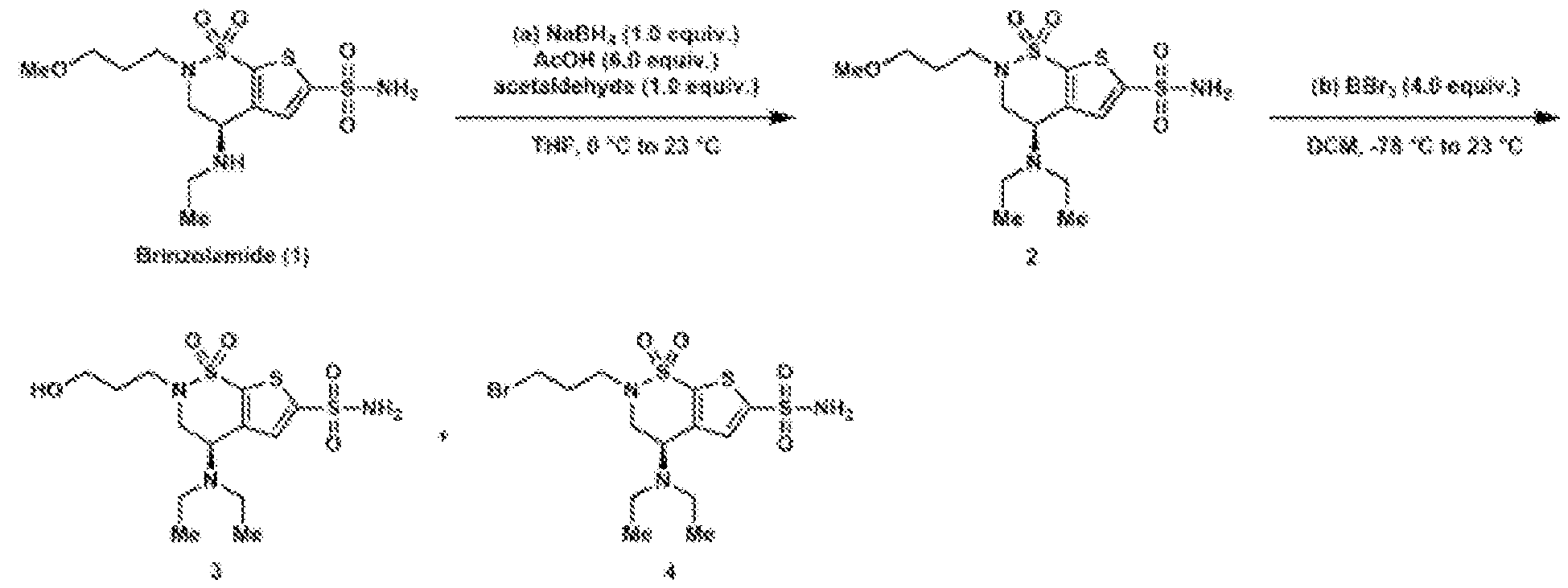
- a reactive handle may be installed to brinzolamide (BZA).
- BZA brinzolamide
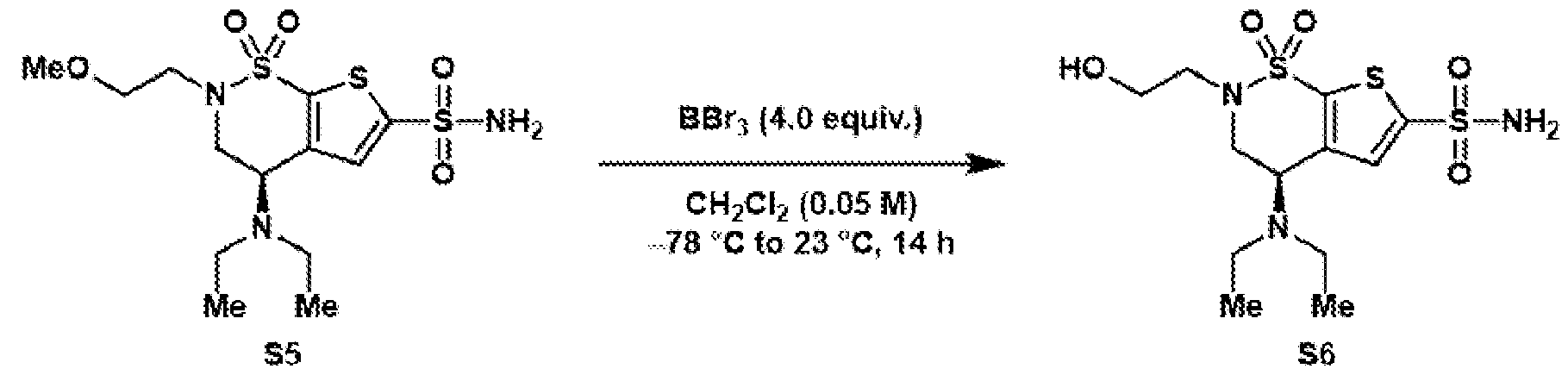
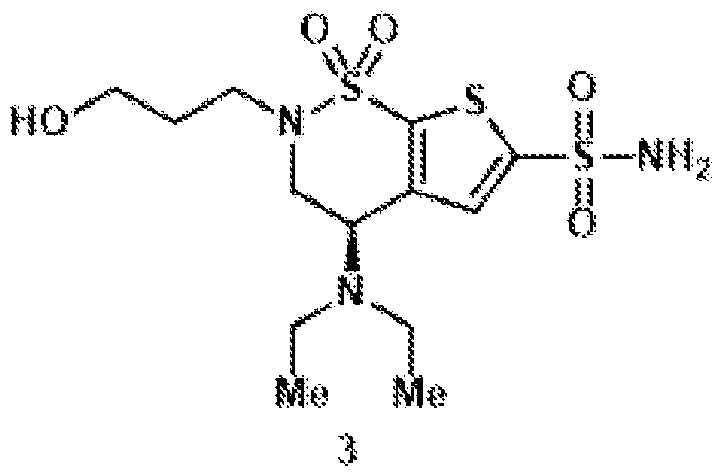
- Reductive amination of the secondary amine of brinzolamide (1) followed by treatment of the intermediary methoxy ether (2) with boron tribromide provides access to either the primary alcohol (3) or the primary alkyl bromide (4).
- These are both useful intermediates for further derivatization.
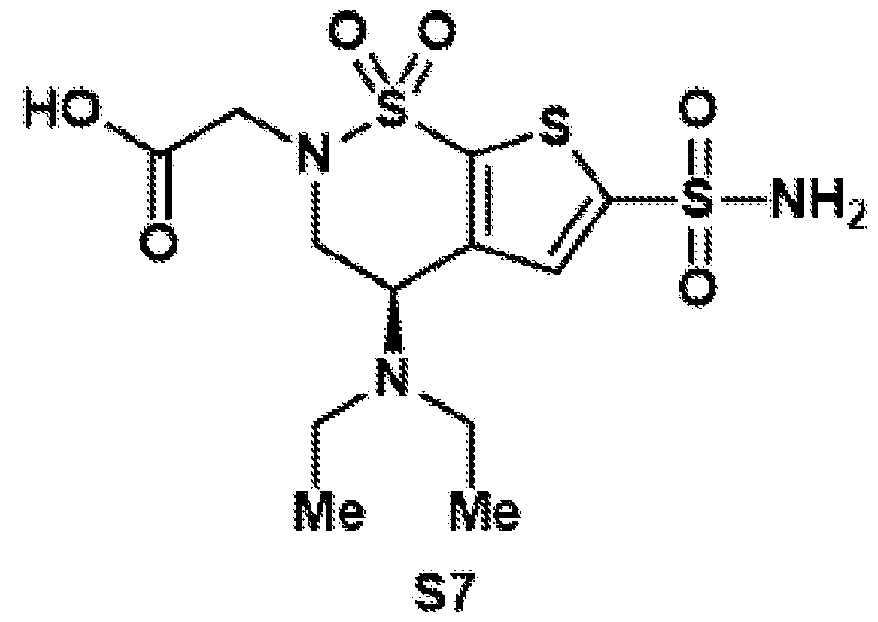
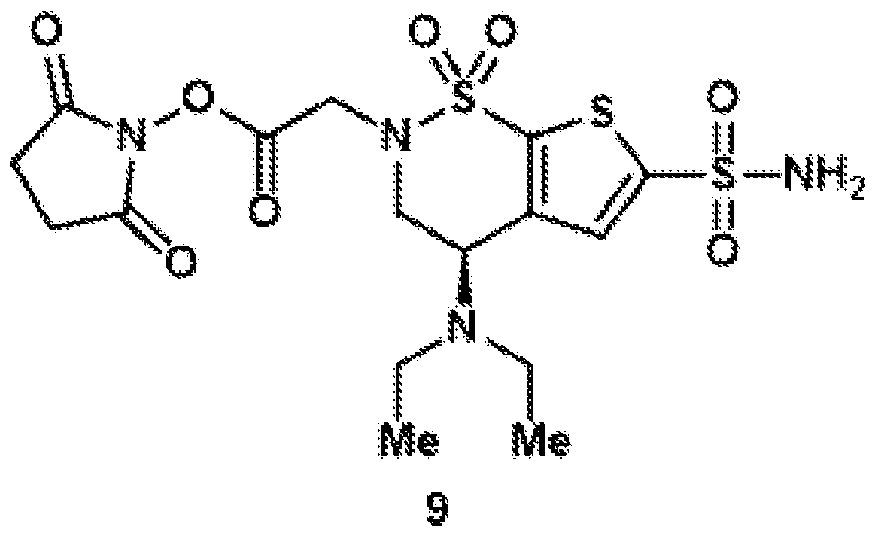
- Oxidation of the primary alcohol to the carboxylic acid (5) and EDC coupling with N-hydroxysuccinimide gives NHS ester (6).
- Other carboxylic acid derivatives for chemical conjugation can also be prepared by this method.
- the alkyl bromide (4) can be converted to the alkyl azide (7), which can be used in click cycloaddition reactions to make additional NHS derivatives.
- the alkyl azide can also be used to conjugate a small-molecule cargo, such as a fluorophore, to brinzolamide.
- Payload delivery across the BBB Disclosed herein include methods and delivery systems for delivering a payload (e.g., a therapeutic agent) to a nervous system.
- the method comprise providing a small molecule capable of interacting with a carbonic anhydrase IV or a derivative thereof.
- the small molecule can be part of a delivery system and the delivery system can comprise a payload to be delivered to a nervous system.
- the method can further comprise administering the delivery system to the subject.
- the delivery system comprises nanoparticles, nanotubes, nanowires, dendrimers, liposomes, ethosomes and aquasomes, polymersomes and niosomes, foams, hydrogels, cubosomes, quantum dots, exosomes, macrophages, and combinations thereof.
- the delivery system comprises a nanoparticle selected from lipid-based nanoparticles, polymeric nanoparticles, inorganic nanoparticles, surfactant-based emulsions, nanowires, silica nanoparticles, virus-like particles, peptide or protein-based particles, lipid- polymer particles, nanolipoprotein particles, and combinations thereof.
- the payload may include an antimicrobial agent, a therapeutic agent, a prodrug, a peptide, a protein, an enzyme, a lipid, a biological response modifier, a pharmaceutical agent, a lymphokine, a heterologous antibody or fragment thereof, a detectable label, a polyethylene glycol (PEG) molecule, or a combination of two or more of the agents.
- the payload can include a neuroactive polypeptide, for example, a neurotrophic factors, endocrine factors, growth factors, paracrine factors, hypothalamic release factors, neurotransmitter polypeptides, polypeptide agonists for a receptor expressed by a CNS cell, polypeptides involved in lysosomal storage disease or any combination thereof.
- the payload can include an IL-1 receptor antagonist (IL-1Ra), dalargin, an interferon- ⁇ , Glial-derived neurotrophic factor (GDNF), tumor necrosis factor receptor (TNFR), nerve growth factor (NGF), brain derived neurotrophic factor (BDNF), neurotrophin-4/5, neurotrophin (NT)-3, a neurturin, neuregulin, a netrin, ciliary neurotrophic factor (CNTF), stem cell factor (SCF), a semaphorin, hepatocyte growth factor (HGF), epidermal growth factor (EGF), transforming growth factor (TGF)-cx, TGF-B, vascular endothelial growth factor (VEGF), platelet-derived growth factor (PDGF), heregulin, artemin, persephin, interleukins, granulocyte-colony stimulating factor (CSF), granulocyte-macrophage-CSF, cardiotrophin-1, hedgehogs, leukemia inhibitory factor
- aspects of the invention also provide for delivery of the conjugate to a subject in order to transport a therapeutic agent across the BBB.
- delivery of the therapeutic payload may be for the treatment of a disease, disorder, or injury of the CNS.
- the therapeutic agent may be released from the conjugate following entry into the CNS.
- the disease, disorder, or injury of the CNS can be, without limitation, multiple sclerosis (MS), amylotrophic lateral sclerosis (ALS), Huntington's disease, Alzheimer's disease, Parkinson's disease, spinal cord injury, traumatic brain injury, stroke, neuropathic pain, neurodegeneration, neuroinflammation, progressive multifocal leukoencephalopathy (PML), encephalomyelitis (EPL), central pontine myelolysis (CPM), adrenoleukodystrophy, Alexander's disease, Pelizaeus Merzbacher disease (PMZ), Globoid cell Leucodystrophy (Krabbe's disease), Wallerian Degeneration, optic neuritis, transverse myelitis, post radiation injury, neurologic complications of chemotherapy, acute ischemic optic neuropathy, vitamin E deficiency, isolated vitamin E deficiency syndrome, Bassen-Kornzweig syndrome, Marchiafava-Bignami syndrome, metachromatic leukodystrophy,
- MS
- the blood-brain barrier serves as a highly selective semi-permeable membrane that separates the circulating blood from the brain and extracellular fluid in the central nervous system (CNS). Due to its selective nature, this barrier restricts the entry of numerous therapeutic agents, especially large molecules, into the brain.
- Mechanism-agnostic engineering strategies such as directed evolution of adeno-associated viral capsids, have succeeded in enabling cross- BBB delivery in animals.
- the brain shuttle molecules generated through this strategy have unpredictable translatability across species due to unclear mechanism.
- Receptor-mediated transport mechanisms have emerged as promising avenues to rationally engineer translatable molecular shuttles that facilitate the transport of therapeutic agents across the BBB.
- the present invention provides rationally designed reactive small-molecule binders based on carbonic anhydrase inhibitor brinzolamide, that can serve as a shuttle to facilitate CA-IV- mediated brain delivery. Design of activated CA-IV binders for bioconjugation One important factor considered in designing shuttles of the invention as the epitope that was be targeted.
- FIG.1 is a schematic of CA-IV mediated transcytosis and antibody conjugation of the invention.
- Carbonic anhydrase binders have been modified with reaction groups that are designed to facilitate a one-step bioconjugation to therapeutic cargo while minimizing any impact on CA-IV binding.
- the conjugate can attach to the CA-IV receptor that is present on brain endothelial cells, thus prompting transcytosis to occur across the BBB.
- existing binder molecules were unutilized that have been developed for other intracellular carbonic anhydrase family members. These carbonic anhydrase inhibitors may exhibit cross-reactivity with CA-IV and can serve as the parent compounds for our shuttle molecules.
- brinzolamide BZA
- BZA brinzolamide
- FIG.2A is a structural analysis of the murine CA-IV binding pocket and its interaction with designed AAVs, BZA, and activated BZA (BZA-2C-NHS).
- FIG.2B is a structural alignment of BZA across CA-IV homologues.
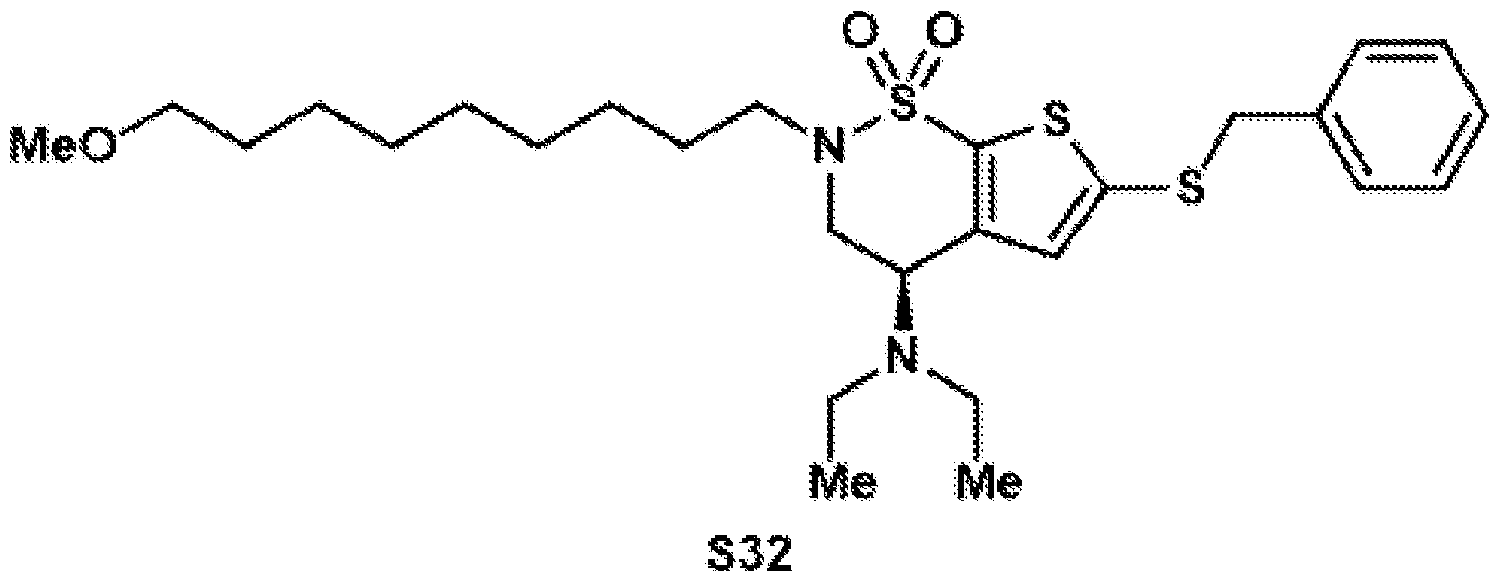
- an extra ethyl group was introduced to convert the secondary amine into a tertiary amine, thereby minimizing self- reaction.
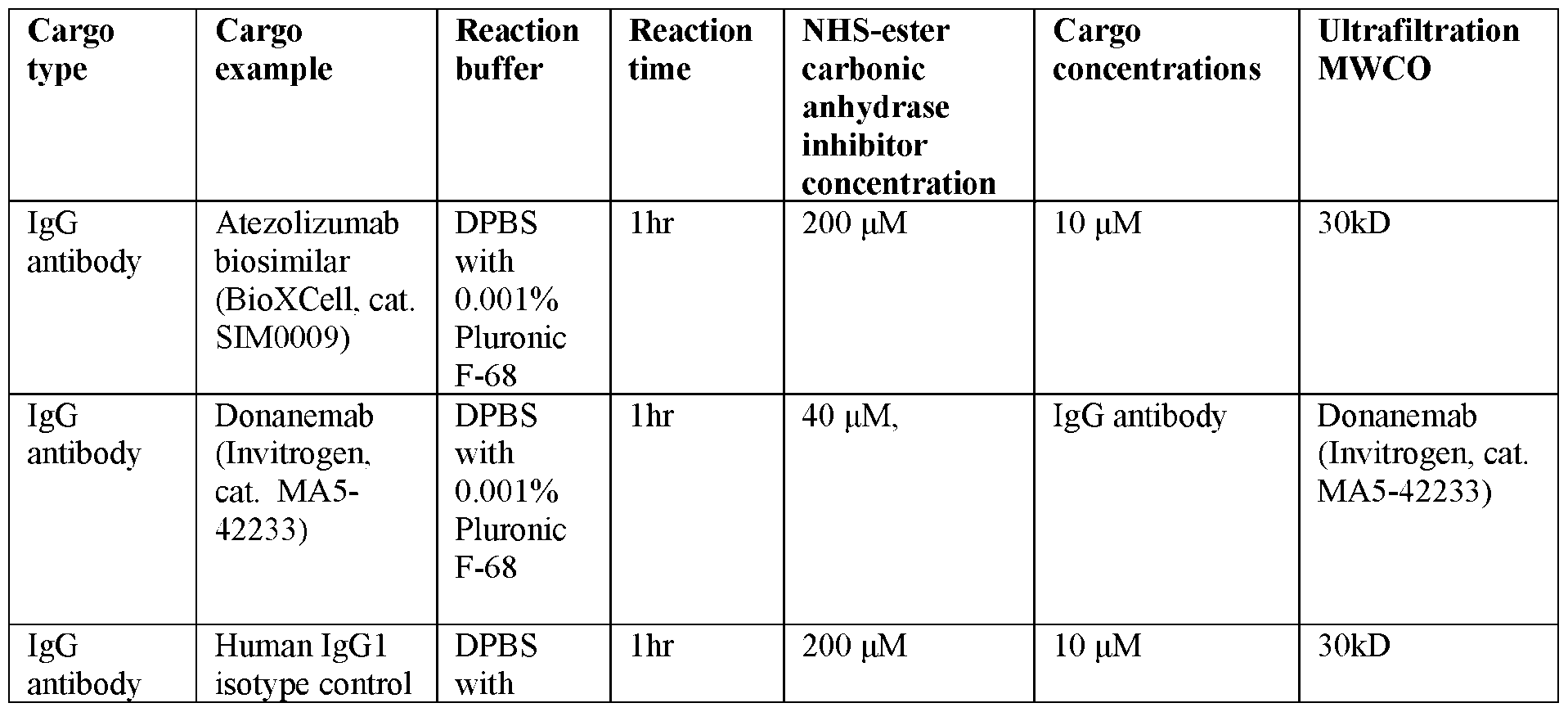
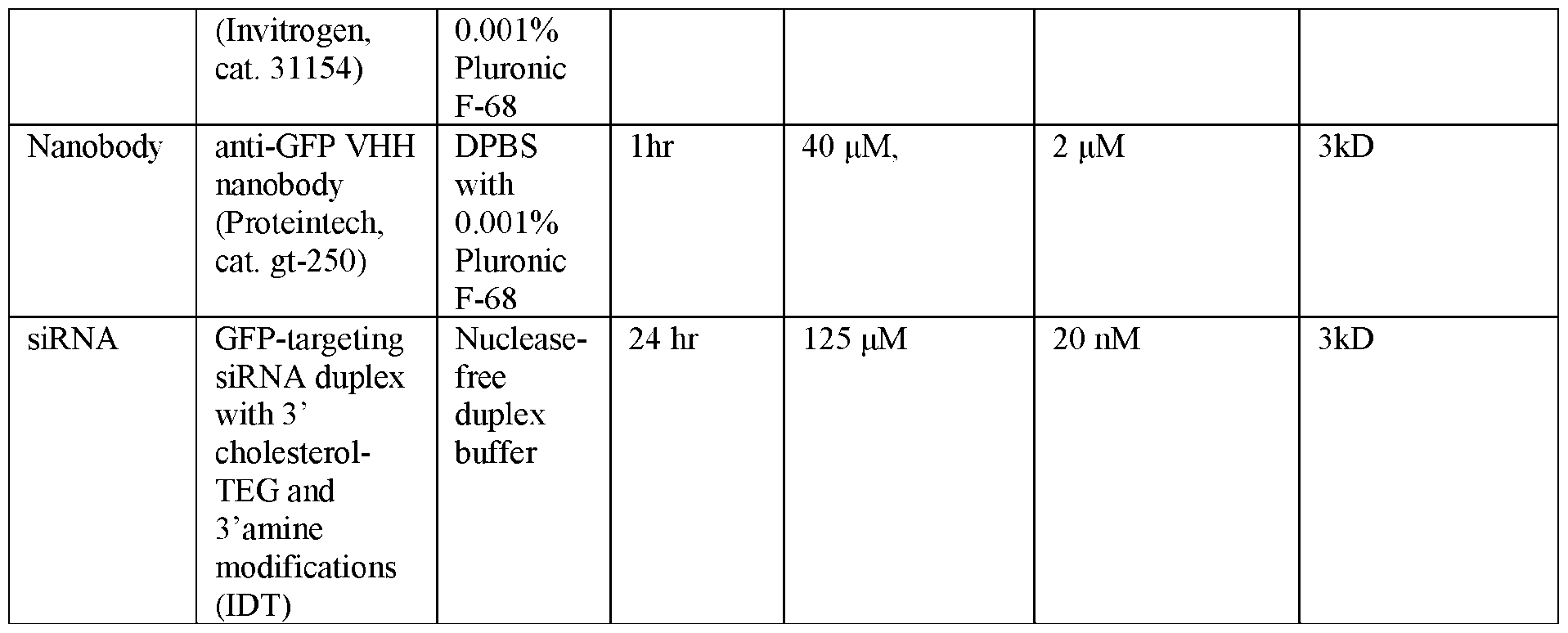
- Production of BZA-conjugated cargo through NHS-ester ligation The NHS-ester ligation was performed in a neutral pH buffer to maintain the stability of cargo molecules.
- the reaction buffer was DPBS with 0.001% Pluronic F-68 (2.7mM KCl, 1.5mM KH 2 PO 4 , 136.9 mM NaCl, 8.1mM Na 2 HPO 4 , pH 7.4; 0.001% Pluronic F- 68).
- the reaction buffer was nuclease-free duplex buffer (30 mM HEPES, pH 7.5; 100 mM potassium acetate) NHS-ester-brinzolamide powder was dissolved in DMSO at 50mM, aliquoted, and stored at -80°C. Right before the reaction, the stock NHS-ester-brinzolamide solution was dissolved to 100 ⁇ L of reaction buffer at 10x of the target final concentration. The 10x solution was mixed with purified cargo molecule diluted in 900 ⁇ L of the same reaction buffer in a 2 mL centrifuge tube. The tube was rotated and agitated in 25°C during the reaction.
- FIG.3 shows an 1 H-NMR verification of chemical synthesis of NHS-ester-brinzolamide.
- FIG.4A is a structural diagram showing Ate conjugation to BZA using linker 2C to generate BZA-2C-Ate.
- FIG.4B-G are LC-MS graphs of BZA-Ate conjugation.
- Cell-based binding assays with HEK293T cells and HeLa cells were conducted, as described below: Select Experimental Methods Cell-based binding assays – HEK293T HEK293T cells were seeded at 80% confluency in six-well plates and maintained in Dulbecco’s modified Eagle’s medium (DMEM) supplemented with 5% fetal bovine serum (FBS), 1% nonessential amino acids (NEAAs), and penicillin-streptomycin (100 U/ml) at 37°C in 5% CO2.
- DMEM Dulbecco’s modified Eagle’s medium
- FBS fetal bovine serum
- NEAAs nonessential amino acids
- penicillin-streptomycin 100 U/ml
- Membrane-associated human and mouse CA-IV were transfected by polyethylenimine (PolySciences, no.23966). Cells were seeded on Neuvitro poly-d-lysine– coated sterile German glass coverslips (Fisher Scientific, no. NC0343705) 24 hours after transfection in 24-well plates. Brinzolamide-conjugated nanobodies and antibodies were added to the media at 0.4 and 0.2 ⁇ M respectively and incubated for one hour at 37 ⁇ C in 5% CO2 then fixed in 4% PFA.
- Coverslips were blocked with 1 ⁇ tris-buffered saline (TBS) containing 3% bovine serum albumin (BSA) and 0.3% Triton-X100 for permeabilizing conditions for 30 min and incubated in secondary antibody (nanobodies, 1:1000 dilution; GenScript, A01994; antibodies, 1:1000 dilution; Invitrogen, A-21445) in 1 ⁇ TBS and 3% BSA with 0.05% Triton X- 100 for permeabilizing conditions for 60 min at ambient temperature. Coverslips were washed three times in 1 ⁇ TBS.
- TBS tris-buffered saline
- BSA bovine serum albumin
- Triton-X100 Triton-X100
- Membrane-associated human and mouse CA-IV were transfected by polyethylenimine (PolySciences, no.23966). Cells were seeded in black, 96-well glass-bottom plates coated with poly-L-ornithine (Cellvis, no. P96-1.5H-N; Sigma Aldrich, no. P4957) 24 hours after transfection. Brinzolamide-conjugated nanobodies and antibodies were added to the media at 0.4 and 0.2 ⁇ M respectively and incubated for one hour at 37 ⁇ C in 5% CO2 then fixed in 4% PFA.
- TBS tris-buffered saline
- BSA bovine serum albumin
- Triton-X100 Triton-X100 for permeabilizing conditions for 60 min at ambient temperature.
- Cells were washed three times in 1 ⁇ TBS. Fluorescent microscopic images were captured on a confocal laser scanning microscope (LSM 980, Carl Zeiss, USA).
- HeLa cells were seeded at 80% confluency in six-well plates and maintained in Dulbecco’s modified Eagle’s medium (DMEM) supplemented with 10% fetal bovine serum (FBS), 1% nonessential amino acids (NEAAs), and penicillin-streptomycin (100 U/ml) at 37°C in 5% CO2.
- DMEM Dulbecco’s modified Eagle’s medium
- FBS fetal bovine serum
- NEAAs 1% nonessential amino acids
- penicillin-streptomycin 100 U/ml
- antibodies were labeled by combining an 800 nM solution of the treatment antibody with a 960 nM solution of the labeling reagent at a 1:1 v/v ratio. After 10 minutes at room temperature, the labeled antibodies were added to cells at a 1:1 v/v ratio. Following three or six hours of incubation, the cells were stained with NucBlue LiveReady Probes (Invitrogen, R37065) and imaged on a high content confocal microscope with a 40x objective (Micro Confocal ImageXPress, Molecular Devices). Surface Plasmon Resonance (SPR) A Sierra SPR-32 (Bruker) loaded with a protein A sensor chip was used.
- SPR Surface Plasmon Resonance
- Fc-fusion CA-IV receptor proteins in HBS-EP+ buffer were immobilized at 200nM. Conjugated and unconjugated cargo molecules in the same buffer at labeled concentrations were injected.
- IgG antibody cargos the setup was reversed, where cargo proteins in HBS-EP+ buffer (GE Healthcare) were immobilized at 200nM, and the untagged CA-IV receptor proteins at labeled concentrations were injected.
- the analyte was injected at a flow rate of 10 uL per min for 240 s followed by a 600 s dissociation. Regeneration steps with 10 mM glycine pH 1.5 was performed between each cycle. All kinetic data were double reference subtracted.
- mice were intravenously injected with the 0.05mg IgG antibodies.
- the mice were anesthetized with Euthasol (pentobarbital sodium and phenytoin sodium solution, Virbac AH) and transcardially perfused using 30–50 mL of 0.1 M phosphate-buffered saline (PBS) (pH 7.4), followed by 30– 50 mL of 4% paraformaldehyde (PFA) in 0.1 M PBS.
- PBS phosphate-buffered saline
- PFA paraformaldehyde
- the tissues were washed with 0.1 M PBS twice and stored in fresh PBS-azide (0.1 M PBS containing 0.05% sodium azide) at 4 °C.
- PBS-azide 0.1 M PBS containing 0.05% sodium azide
- brains and livers were sectioned into 100um thick slice with vibratome.
- the slices were first incubated in the blocking buffer (10% normal donkey serum (NDS), 0.1% Triton X-100, and 0.01% sodium azide in 0.1 M PBS, pH 7.4) with antibody anti-human Igg-Alexa647 at 1:200.
- the slices were then washed 3 times in 0.1 M PBS over a total duration of 5–6 h.
- DAPI 6-diamidino-2-phenylindole
- Sigma-Aldrich 10236276001, 1:1000
- the DAPI and/or antibody-stained tissue sections were mounted with ProLong Diamond Antifade Mountant (Thermo Fisher Scientific, P36970) before imaging them under the microscope.
- ELISA Both Ate and BZA-Ate concentrations were quantified using an anti-human IgG ELISA Kit (abcam, #ab195215).
- PBS perfused organs from mice were collected in tubes with prefilled beads (Benchmark #D1032-30, #D1032-15, #D1033-28). These tubes were then filled up with 1 mL of cell extraction buffer from the ELISA’s kit following the protocol. Tubes were then placed into a Benchmark BeadBlaster 24 homogenization machine at 7 m/s speed, 2 total cycles with 15 second cycles and a 30 second pause interval between each cycle. After 2 cycles, a manual 1 minute pause was given. This homogenization and pause step were repeated two more times. Samples were centrifuged at 18,000g, 4°C for 30 minutes.700 ⁇ L of supernatant was collected into two wells, 350 ⁇ L each well without disturbing the debris near the beads.
- Supernatant was then diluted in DI water, and 50 ⁇ L of diluted sample was pipetted onto each well of the precoated ELISA plate from the kit.
- the diluted antibody solution and standard from the kit were prepared following its protocol. Standard concentrations ranged from 0.23 ng/mL to 15 ng/mL.50 ⁇ L of diluted antibody solution was then added to every well, and the plate was incubated for 50 minutes on a shaker. The plate was then washed 3x with wash buffer from the kit, and 100 ⁇ L of TMB solution was added to each well and incubated for 35 minutes on a shaker.100 ⁇ L of stop solution was then added to the wells, and the plate was sealed and read at 450 nm absorbance.
- FIG.5A-D are graphs of results from a SPR assays of CA-IV and BZA-2C-Ate.
- SPR assays evaluated the binding interaction between conjugated or unconjugated IgG antibody and purified CA-IV proteins.200 nM IgG antibody was immobilized on a Protein A- precoated capture sensor, and CA-IV proteins at different concentrations were subsequently introduced. Receptor concentrations are indicated in the inset. A concentration-dependent binding signal was observed between CA-IV and BZA-2C- Ate, with estimated dissociation constants ranging from 100nM to 200nM was shown. Following this, tests were conducted to determine if the conjugated antibodies can bind to cultured cells that have an over-expression of CA-IV.
- FIG.5E shows immunofluorescent images of live CA-IV expressing cells incubated with either unconjugated or BZA-conjugated Ate.
- the cells were fixed after 1 hr of incubation with 0.2 ⁇ M cargo antibody. Following fixation, the cells were washed with a detergent-containing buffer to permeabilize them. Next, the fixed cells were stained with an anti-human antibody conjugated to Alexa Fluor 647 and subsequently imaged.
- Panels labeled with IF (MIP) display a max- intensity projection of the cells, while panels labeled with IF (z slice) show a section close to the well bottom.
- the cargo antibodies were pre-labeled with a fluorescent pH indicator, pHrodo, which exhibits increased fluorescence intensity in acidic environments like endosomes before being applied at 0.4 ⁇ M to the cells.
- the treated cells were imaged 6 hr after incubation with the antibodies. Punctate structures were clearly visible inside cells that have been permeabilized before staining, suggesting the internalization of antibodies via endocytosis. Binding and internalization can be observed with CA-IV from various species such as mouse, human, and rhesus macaque.
- immunofluorescence analysis revealed the presence of BZA-2C-Ate signals on cells expressing CA-IV.
- pHrodo a pH-sensitive dye
- CA-IV-expressing cells were incubated with labeled antibody samples.
- the pHrodo dye exhibits a significant increase in fluorescence as the pH decreases, making it an indicator of internalization. After a 6-hour incubation period, a consistent presence of puncta structures was observed, which closely resembled the patterns observed in the immunofluorescence images.
- FIG.5 F-G show the average and counted puncta, respectively, intensity of CA-IV- expressing HeLa cells incubated with either unconjugated or BZA-2C-Ate conjugates.
- the in vitro findings demonstrated the consistent binding of BZA-2C-Ate to CA-IV and its subsequent internalization in CA-IV expressing cells across various species, including human, mouse, and macaque. This observation indicated the preservation of crucial residues for brinzolamide interaction, enabling in vivo assessments in rodent and non-human primate models.
- FIG.6A shows the structure of select BZA-Ate linker variants.
- FIG.6B-M shows LC-MS graphs of unmodified Ate and BZA-Ate linker variant conjugates. As shown, the efficiency of conjugation is dependent on both the length of the linker and the type of binder.
- FIG.7A-C are graphs of results from SPR assays of CA-IV and BZA-Ate linker variant binding.
- FIG.7D shows immunofluorescent images of live CA-IV expressing cells incubated with either unconjugated or BZA-conjugated Ate linker variants.
- FIG.7E-F show representative live cell images showing CA-IV-expressing HeLa cells incubated with either unconjugated or BZA-conjugated antibodies after 3 hours and 6 hours of incubation with antibodies, respectively.
- Puncta identified in the live cell images were segmented, and the intensities contained within each punctum are quantified and averaged by cells.
- FIG.7G-4H show the average and counted puncta, respectively, intensity of CA-IV- expressing Hela cells incubated with either unconjugated or BZA-conjugated linker variants after 3 hours.
- FIG.7I-4J show the average and counted puncta, respectively, intensity of CA-IV- expressing Hela cells incubated with either unconjugated or BZA-conjugated linker variants after 6 hours.
- the average puncta intensity of each cell was calculated and displayed as a box plot, with outliers highlighted.
- Statistical significance was determined using Student’s t-test.
- the number of puncta of each cell was counted and displayed as a box plot, with outliers highlighted.
- Statistical significance was determined using Poisson means test.
- BZA-2C-Ate BZA-3C-Ate
- BZA-4C-Ate there are clear puncta inside cells that are permeabilized before staining, indicating internalization of the antibodies.
- BZA-3C-NHS exhibited the highest drug-to-antibody ratio.
- BZA-2C-NHS facilitated highly efficient internalization and could be synthesized easily (4 steps compared to 8 steps for other linker variants) using commercially available starting materials.
- FIG.8A shows the structures of NHS-ester variants of two alternative CA-IV binders, acetazolamide (AZA) and dorzolamide (DZA).
- FIG.8B-E show LC-MS graphs of NHS ester variants of two alternative CA-IV binders, AZA and DZA respectively.
- FIG.8F-G is a graph of results from an SPR assay of CA-IV and NHS ester variants of two alternative CA-IV binders, AZA and DZA respectively.
- FIG.8H-I show live cell images showing CA-IV-expressing Hela cells incubated with either AZA-conjugated or DZA-conjugated Ate, respectively. In Puncta identified in the live cell images were segmented, and the intensities contained within each punctum are quantified and averaged by cells.
- FIG.8J-K show the average and counted puncta intensity, respectively, of CA-IV- expressing Hela cells incubated with either unconjugated, AZA-conjugated, or DZA-conjugated Ate.
- Ate antibody The functional binding between the Ate antibody and its target antigen is unaffected by the conjugation of these compounds to lysine residues, regardless of the type of linker and binding core employed.
- BZA and BZA-2C-IgG cargo variants In addition to conjugating the Ate antibody, the potential for extending this bioconjugation approach to other biological molecules and modalities to facilitate receptor binding was explored. Additional in vitro characterization of BZA-NHS conjugated to different therapeutic modalities was tested, including nanobodies, siRNA, and small molecules.
- IgG antibodies Don
- Immunoglobulin G (IgG) antibodies are crucial components of the immune response, capable of neutralizing pathogens and signaling immune cells.
- BZA-modified human IgG1 focusing on its interaction with CA-IV was explored.
- FIG.9A shows the structures of an IgG antibody BZA variant conjugates.
- FIG.9B-K show LC-MS graphs of BZA-IgG antibody conjugates including Don.
- FIG.10A-E show graphs of the results from an SPR assay of CA-IV and BZA- conjugated Don, unconjugated Don, and BZA-conjugated higG1 isotype, respectively.
- FIG.10F shows representative immunofluorescent and live cell images showing CA-IV- expressing Hela cells incubated with either unconjugated-IgG of BZA-conjugated variant antibodies including Don.
- Puncta identified in the live cell images were segmented, and the intensities contained within each punctum are quantified and averaged by cells.
- FIG.10G-H show the average and counted puncta intensity, respectively, of CA-IV- expressing Hela cells incubated with either unconjugated or BZA conjugated antibody variants.
- FIG.11A-D show SPR of modified and unmodified therapeutic IgG antibodies tested against purified human CA-IV and mouse CA-IV, including atezolizumab and Don.
- the IgG antibodies were immobilized on a Protein A chip, and CA-IV proteins at different concentrations are injected. Receptor concentrations were indicated in the inset.
- FIG.11E-F show internalization assays in cultured Hela cells with the BZA-modified IgG antibodies and unmodified IgG antibodies, including atezolizumab and Don. Hela cells are transfected with indicated receptors and incubated with indicated antibodies. After fixation, the cells are washed with buffer either with detergent (permeabilized) or without detergents (unpermeabilized).
- the cells were washed with a detergent-containing buffer to permeabilize them.
- the fixed cells were stained with an anti-human antibody conjugated to Alexa Fluor 647 and subsequently imaged.
- Panels labeled with IF (z slice) showed a section close to the well bottom.
- the cargo antibodies were pre-labeled with a fluorescent pH indicator, pHrodo, which exhibited increased fluorescence intensity in acidic environments like endosomes before being used to treat the cells.
- the treated cells were imaged 6 hr after incubation with the antibodies. Punctate structures were clearly visible inside cells that have been permeabilized before staining, suggesting the internalization of antibodies via endocytosis. Binding and internalization could be observed with CA-IV from various species.
- the BZA-modified therapeutic human IgG1 showed binding affinity to purified CA-IV with a Kd of 100-300nM. This is similar to the Kd of Denali's TfR-binding ATVs, suggesting comparable effectiveness. Similar to the case in modified nanobodies, products from high-label- density condition showed stronger binding to CA-IV proteins, indicating that controlling the label density can be a potential way to tune the binding affinity of modified proteins.
- the BZA- modified IgG molecules also show binding to cultured cells overexpressing CA-IV. Importantly, BZA-modified IgG can bind to not only human CA-IV protein, but also CA-IV proteins from mouse and rhesus macaque.
- Nanobodies Nanobodies, or camelid single-domain antibodies, have been highlighted for their high stability, high yield, and demonstrated therapeutic value with several FDA-approved drugs currently in use. Notably, these nanobodies, derived from Camelus dromedarius, have three surface- exposed lysine residues in the conserved region.
- FIG.12A-B show the structure of a BZA-nanobody variants.
- FIG.12C-D show the LC-MS analysis of unmodified nanobodies and nanobodies BZA- conjugated nanobodies. The results demonstrate that approximately half of the nanobodies conjugated with 1-2 BZA moieties ( ⁇ 390 Da).
- FIG.12E-H show graphs of the results from an SPR assay of the binding interaction between an unconjugated or BZA-conjugated nanobody and Fc-tagged CA-IV proteins. 200 nM Fc-tagged CA-IV protein were immobilized on a Protein A-precoated capture sensor, and nanobodies at different concentrations were subsequently introduced. Nanobody concentrations were indicated in the inset. The background binding signal observed in unconjugated nanobody were likely due to weak interaction between nanobody and Protein A.
- FIG.12I show representative immunofluorescent images showing CA-IV-expressing Hela cells incubated with either anti-GFP VHH unconjugated nanobodies, BZA-2C conjugated nanobodies, and BZA-3C conjugated nanobodies.
- the cells were fixed after 1 hr of incubation with 0.4 ⁇ M cargo nanobody. Following fixation, the cells were washed with a detergent-containing buffer to permeabilize them. Next, the fixed cells were stained with an anti-camelid antibody conjugated to Alexa Fluor 647 and subsequently imaged. Max-intensity projection images are shown. Scale bar, 100 ⁇ m.
- FIG.13A-B shows graphs of the results from an SPR assay of modified and unmodified nanobodies tested against purified human CA-IV and mouse CA-IV.
- the CA-IV proteins are fused to a human Fc tag and immobilized on a Protein A chip.
- Nanobodies are injected at different concentrations as indicated in the inset. The background signal seen in unmodified nanobody is likely due to the sporadic interaction between nanobody and protein A.
- FIG.13C-D show an internalization assay in cultured HEK293 cells with the BZA- modified nanobodies and unmodified nanobodies. After fixation, cells incubated with the nanobodies are washed with buffer either with detergent (permeabilized) or without detergents (unpermeabilized).
- the fixed cells were then stained with anti-camelid antibody conjugated to Alexa Fluor 647.
- the cells were fixed after 1 hr of incubation with 0.4 ⁇ M cargo nanobody. Following fixation, the cells were washed with a detergent-containing buffer to permeabilize them. Next, the fixed cells were stained with an anti-camelid antibody conjugated to Alexa Fluor 647 and subsequently imaged. Max-intensity projection images are shown. Scale bar, 100 ⁇ m.
- SPR Surface Plasmon Resonance
- cell-based binding assays suggests a direct binding interaction between BZA-modified nanobody and purified human sulfonic carbonic anhydrase IV (hCA-IV) protein.
- siRNA Small interfering RNA have emerged as a pivotal therapeutic modality in the world of molecular medicine. At its core, siRNA functions by specifically targeting and degrading messenger RNA (mRNA) sequences, thereby halting the production of targeted proteins. This provides a highly selective mechanism to downregulate disease-causing genes.
- siRNA provides precise gene silencing, ensuring a higher degree of specificity. Furthermore, the ability of siRNA to target traditionally 'undruggable' genes offers new hope for conditions that have been elusive to treat. Despite the promises, systemic delivery of siRNA to the brain has been a challenging problem.
- FIG.14A shows the structure of a BZA shuttle-siRNA variant.
- FIG.14B-D shows graphs of the results from an SPR assay of the binding interaction between an unconjugated or BZA-conjugated siRNA duplex and Fc-tagged CA-IV proteins.
- 200 nM Fc-tagged CA-IV protein were immobilized on a Protein A-precoated capture sensor, and siRNA at different concentrations were subsequently introduced. siRNA concentrations were indicated in the inset.
- FIG.14E shows representative immunofluorescent images showing CA-IV-expressing Hela cells incubated with either unconjugated fluorescently labeled siRNA or BZA-2C conjugated siRNA.
- FIG.15A shows the structure of a BZA shuttle-siRNA variant. The sequence and design of a GFP-targeting siRNA duplex. The sense and antisense strand of the duplex is modified with cholesterol-TEG and a primary amine, respectively.
- FIG.15B-C show graphs of the results from an SPR assay of the binding interaction between an unconjugated or BZA-conjugated siRNA duplex and Fc-tagged CA-IV proteins.
- FIG.16A shows the structure of a BZA shuttle-small molecule cargo variant.
- FIG.16B show representative immunofluorescent images showing CA-IV-expressing Hela cells incubated with either unconjugated small molecule dye or BZA-conjugated small molecules dye.
- FIG.16C shows the structure of a BZA shuttle-small molecule cargo variant. The schematic shows the process of conjugating a fluorescent molecule (Alexa Fluor 647) to brinzolamide using a CuAAC reaction.
- FIG.16D show images from an internalization assay using the HEK cells overexpressing CA-IV receptors.
- FIG.16E shows representative images of brain slices and liver slices from mice injected with BZA-conjugated fluorophore or unconjugated fluorophores. Animals injected with BZA- conjugated fluorophore shows stained vasculature in both brain and liver.
- FIG.16F shows a fluorescence reading of lysed tissues of animals injected with BZA- conjugated fluorophore or unconjugated fluorophores using a plate reader.
- BZA-conjugated fluorophore molecules exhibited signal throughout the cytosol, suggesting their ability to penetrate the membrane and potentially interact with intracellular carbonic anhydrases.
- This variance in membrane permeability and access to carbonic anhydrase family members may lead to differing biodistributions of small versus large BZA-conjugated molecules.
- BZA-modified nanobodies, therapeutic IgG antibodies, and small molecules have strong potential for broad applications in targeted therapeutics.
- Their binding affinity to hCA-IV and their ability to mediate the binding and internalization show promising translational potentials.
- Biologics conjugated to carbonic anhydrase binders cross the BBB in mice BZA-conjugation technology in mice was analyzed in vivo.
- FIG.17A shows immunofluorescent images of mice systemically administered with unconjugated and BZA-conjugated Ate antibodies in sagittal brain sections. Scale bar, 1 mm.
- FIG.17B shows immunofluorescent images of mice systemically administered with unconjugated and BZA-conjugated Ate antibodies in brain cortex. Scale bar, 100 ⁇ m.
- FIG.17D shows a graph of biodistribution of BZA-conjugated Ate in comparison to unconjugated control at day 7. Left, fold change of BZA-conjugated Ate concentration over unconjugated Ate.
- FIG.17E-F show graphs of pharmacokinetics of BZA-conjugated and unconjugated Ate in whole brain and liver, respectively, over a period of 14 days.
- FIG.17G-H show graphs of pharmacokinetics of BZA-conjugated and unconjugated Ate in the brain vasculature and the brain parenchyma, respectively, over a period of 14 days.
- the BZA-conjugated Ate exhibited prolonged enrichment in the brain, including the cortex, compared to unconjugated Ate.
- Both immunofluorescent imaging and ELISA quantification indicated a peak in brain IgG signal between day 5 and day 7, followed by a decline within a 14-day period.
- the ELISA-based quantification on day 7 indicated that the concentration of BZA-conjugated Ate in the brain was one order of magnitude higher than the unconjugated control.
- the duration of brain exposure for the CA-IV-binding brain shuttle surpassed that of TfR-based shuttles.
- Further analysis indicated that BZA-conjugated Ate can efficiently traverse the BBB to reach the brain parenchyma.
- FIG.18C shows quantification of BZA-conjugated and unconjugated Ate antibodies in peripheral organs at day 7 in mice systemically administered with unconjugated and BZA- conjugated Ate antibodies.
- Both ELISA-based quantification and immunofluorescence images validated that the liver distribution of BZA-conjugated Ate did not significantly differ from unconjugated antibodies over a 14-day period.
- Brain access through Carbonic Anhydrase Binder bioconjugation “BrainCAB”) receptor binder can successfully bind to homologs of CA- IV across various species, such as rodents and non-human primates, due to the conserved binding pocket of CA-IV.
- This capability allows for the broad application and evaluation of this technology in diverse model organisms and disease models.
- the single-step bioconjugation method employed by the present invention exhibits versatility in accommodating various therapeutic agents, including nanobodies and oligonucleotides, underscoring its wide-ranging applicability.
- the straightforward nature of this bioconjugation process enables seamless upscaling of manufacturing.
- the BrainCAB technology facilitated by its single-step bioconjugation process, demonstrated considerable potential in both in vitro and in vivo studies. With its high BBB- crossing efficiency, enhanced brain specificity, and extended pharmacokinetics, along with its wide applicability and simple production process, this approach shows great potential for delivering therapeutics to conditions traditionally hindered by the intricacies of the blood-brain barrier.
- conjugated molecules’ of the invention were shown to exhibit successful binding to CA-IV proteins from different mammalian species and subsequent internalization were verified via surface plasmon resonance (SPR) and cell-based assays.
Landscapes
- Health & Medical Sciences (AREA)
- Epidemiology (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Engineering & Computer Science (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
The present invention provides compositions and methods comprising brinzolamide and brinzolamide derivates as shuttles for the BBB-crossing, for example through the receptor carbonic anhydrase IV (CA-IV).
Description
Ester Derivatives of Binders Targeting CA-IV Government License Rights Statement This invention was made with government support under Grant No(s). NS111369 & GM118191 awarded by the National Institutes of Health. The government has certain rights in the invention. Field of the Invention The invention relates to methods and shuttles for crossing the blood brain barrier. Background The blood brain barrier (BBB) presents a fundamental bottleneck to the development of effective research tools and therapeutics for the central nervous system (CNS). This structure, comprising mainly of brain endothelial cells, requires large molecules to be delivered via invasive intracranial injections, technically challenging focused ultrasound, or receptor-mediated transcytosis. The rational design of BBB-crossing large molecules has long been hampered by the imperfect understanding of the mechanisms involved in transcytosis, with only a handful of targets, such as the transferrin receptor, validated for research and therapies. Thus, the identification of BBB-crossing targets, mechanisms, molecules and methods is needed to improve the efficiencies of research tools and therapies for CNS. Summary The present invention provides compositions and methods comprising brinzolamide and brinzolamide derivates as shuttles for the BBB-crossing, for example through the receptor carbonic anhydrase IV (CA-IV). The present invention provides rationally designed reactive small-molecule binders based on brinzolamide, that can serve as a shuttle to facilitate CA-IV-mediated brain delivery. The reactive small-molecule shuttles may be conjugated to different therapeutic cargo modalities, including nanobodies, therapeutic IgG antibodies, small interfering RNAs (siRNAs), or antisense oligonucleotides (ASOs), for example through a single-step N-hydroxysuccinimide (NHS) ester reaction.
The present invention provides a robust and adaptable approach for transporting therapeutic substances through the blood-brain barrier using CA-IV-mediated delivery introduced by linking therapeutic cargo to brinzolamide derivative CA-IV binders through bioconjugation. Aspects of the invention provide a conjugate comprising a blood brain barrier (BBB) shuttle selected from brinzolamide or a derivative thereof and a therapeutic cargo conjugated to the shuttle. The brinzolamide derivative may be, for example a brinzolamide ester. The brinzolamide derivative may advantageously have greater specificity for CA-IV in comparison to brinzolamide. The brinzolamide derivative may have decreased specificity for CA II in comparison to brinzolamide. The brinzolamide derivative may comprise an N-hydroxysuccinimide (NHS) ester. The brinzolamide derivative comprising an NHS ester may be formed from a single-step reaction ester reaction. The brinzolamide derivative may be formed from the steps of alkylation, tosylation, amination, oxidation, deprotection, Jones oxidation, and esterification of brinzolamide. The brinzolamide derivative may be covalently bonded to an NHS ester via a 1-8 carbon alkyl linker. For example, the brinzolamide ester has the formula:
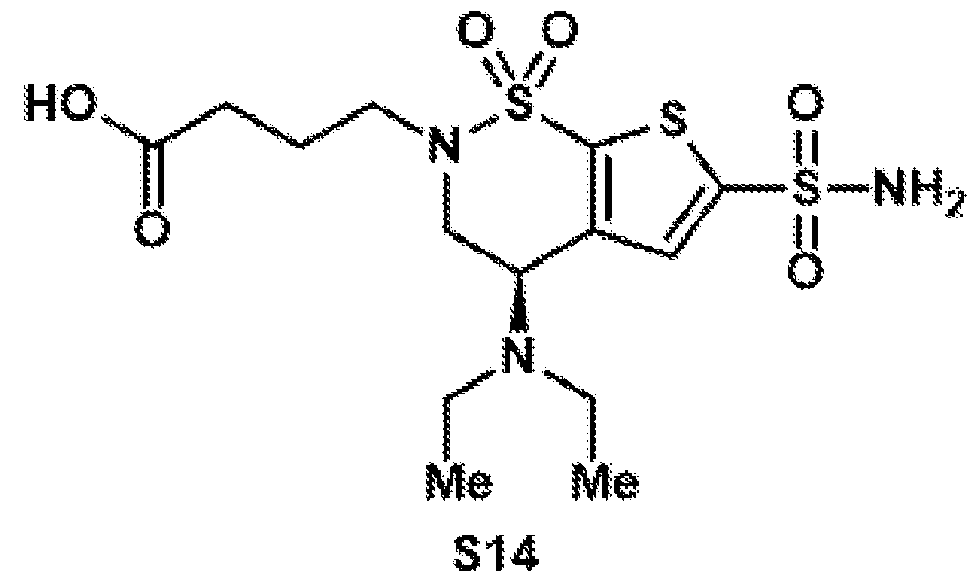
(BZA-nc-NHS), wherein n is 1-8, preferably n is 1, 3, 4, 6 or 8. The brinzolamide derivative may be selected from among the compounds:
In aspects of the invention, the brinzolamide derivative may comprise a pentafluorophenyl (PFP) ester. In the derivative, brinzolamide may be covalently bonded to the PFP ester via polyethylene glycol. The brinzolamide derivative may be selected from among the compounds:
The conjugate may comprise a plurality of small molecule shuttles conjugated to the therapeutic cargo. For example, conjugate may comprise 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, or greater than 14 brinzolamide or brinzolamide derivatives conjugated to a single therapeutic cargo. The therapeutic cargo may be a biological molecule, for example a nucleic acid (for example, RNA, siRNA, DNA, or an ASO), a protein (for example, an enzyme), a peptide, an antibody, a nanobody, a lipid, a polysaccharide, and a combination thereof.. The therapeutic cargo may be a non-biological molecule, for example a small molecule or a dye. The therapeutic cargo may be conjugated to brinzolamide or a brinzolamide derivative via N-hydroxysuccinimide (NHS) ester coupling or copper(I)-catalyzed azide-alkyne cycloaddition (CuAAC). Advantageously, the shuttle may be a carbonic anhydrase IV (CA-IV) shuttle. Thereby, when provided to a cell expressing CA-IV as a surface protein, binding of the shuttle to the CA- IV protein mediates transcytosis of the therapeutic cargo across the BBB. The therapeutic cargo may be a therapeutic cargo for the treatment of a disorder affecting the central nervous system. The CA-IV shuttle may be a shuttle for human CA-IV. Aspects of the invention further provide methods of delivering therapeutic cargo across the BBB of a subject. Methods of the invention comprise providing to a subject a conjugate comprising brinzolamide or a brinzolamide derivative and a therapeutic cargo conjugated to the shuttle. Brief Description of the Drawings FIG.1 is a schematic of CA-IV mediated transcytosis and antibody conjugation of the invention. FIG.2A is a structural analysis of the murine CA-IV binding pocket and its interaction with designed AAVs, BZA, and activated BZA (BZA-2C-NHS). FIG.2B is a structural alignment of BZA across CA-IV homologues. FIG.3 shows an 1H-NMR verification of chemical synthesis of NHS-ester-brinzolamide. FIG.4A is a structural diagram showing Ate conjugation to BZA using linker 2C. FIG.4B-G are LC-MS graphs of BZA-2C-Ate conjugation.
FIG.5A-D are graphs of results from a Surface Plasmon Resonance (SPR) assays of CA- IV and BZA-2C-Ate. FIG.5E shows immunofluorescent images of live CA-IV expressing cells incubated with either unconjugated or BZA-conjugated Ate. FIG.5 F-G show the average and counted puncta, respectively, intensity of CA-IV- expressing HeLa cells incubated with either unconjugated or BZA-2C-Ate conjugates. FIG.6A shows the structure of select BZA-Ate linker variants. FIG.6B-M shows LC-MS graphs of unmodified Ate and BZA-Ate linker variant conjugates. FIG.7A-C are graphs of results from SPR assays of CA-IV and BZA-Ate linker variant binding. FIG.7D shows immunofluorescent images of live CA-IV expressing cells incubated with either unconjugated or BZA-conjugated Ate linker variants. FIG.7E-F show representative live cell images showing CA-IV-expressing HeLa cells incubated with either unconjugated or BZA-conjugated antibodies after 3 hours and 6 hours of incubation with antibodies, respectively. FIG.7G-4H show the average and counted puncta, respectively, intensity of CA-IV- expressing Hela cells incubated with either unconjugated or BZA-conjugated linker variants after 3 hours. FIG.7I-4J show the average and counted puncta, respectively, intensity of CA-IV- expressing Hela cells incubated with either unconjugated or BZA-conjugated linker variants after 6 hours. FIG.8A shows the structures of NHS-ester variants of two alternative CA-IV binders, acetazolamide (AZA) and dorzolamide (DZA). FIG.8B-E show LC-MS graphs of NHS ester variants of two alternative CA-IV binders, AZA and DZA respectively. FIG.8F-G is a graph of results from an SPR assay of CA-IV and NHS ester variants of two alternative CA-IV binders, AZA and DZA respectively. FIG.8H-I show live cell images showing CA-IV-expressing Hela cells incubated with either AZA-conjugated or DZA-conjugated Ate, respectively.
FIG.8J-K show the average and counted puncta intensity, respectively, of CA-IV- expressing Hela cells incubated with either unconjugated, AZA-conjugated, or DZA-conjugated Ate. FIG.9A shows the structures of an IgG antibody BZA variant conjugates. FIG.9B-K show LC-MS graphs of BZA-IgG antibody conjugates including Don. FIG.10A-E show graphs of the results from an SPR assay of CA-IV and BZA- conjugated Don, unconjugated Don, and BZA-conjugated higG1 isotype, respectively. FIG.10F shows representative immunofluorescent and live cell images showing CA-IV- expressing Hela cells incubated with either unconjugated-IgG of BZA-conjugated variant antibodies including Don. FIG.10G-H show the average and counted puncta intensity, respectively, of CA-IV- expressing Hela cells incubated with either unconjugated or BZA conjugated antibody variants. Including Don FIG.11A-D show SPR of modified and unmodified therapeutic IgG antibodies tested against purified human CA-IV and mouse CA-IV, including atezolizumab and Don. FIG.11E-F show internalization assays in cultured Hela cells with the BZA-modified IgG antibodies and unmodified IgG antibodies, including atezolizumab and Don. FIG.12A-B show the structure of a BZA-nanobody variants. FIG.12C-D show the LC-MS analysis of unmodified nanobodies and nanobodies BZA- conjugated nanobodies. FIG.12E-H show graphs of the results from an SPR assay of the binding interaction between an unconjugated or BZA-conjugated nanobody and Fc-tagged CA-IV proteins. FIG.12I show representative immunofluorescent images showing CA-IV-expressing HeLa cells incubated with either unconjugated or BZA-conjugated anti-GFP VHH nanobody. FIG.13A-B shows graphs of the results from an SPR assay of modified and unmodified nanobodies tested against purified human CA-IV and mouse CA-IV. FIG.13C-E show representative immunofluorescent images showing CA-IV-expressing Hela cells incubated with either anti-GFP VHH unconjugated nanobodies, BZA-2C conjugated nanobodies, and BZA-3C conjugated nanobodies. FIG.14A shows the structure of a BZA shuttle-siRNA variant.
FIG.14B-D shows graphs of the results from an SPR assay of the binding interaction between an unconjugated or BZA-conjugated siRNA duplex and Fc-tagged CA-IV proteins. FIG.14E shows representative immunofluorescent images showing CA-IV-expressing Hela cells incubated with either unconjugated fluorescently labeled siRNA or BZA-2C conjugated siRNA. FIG.15A shows the structure of a BZA shuttle-siRNA variant. FIG.15B-C show graphs of the results from an SPR assay of the binding interaction between an unconjugated or BZA-conjugated siRNA duplex and Fc-tagged CA-IV proteins. FIG.16A shows the structure of a BZA shuttle-small molecule cargo variant. FIG.16B show representative immunofluorescent images showing CA-IV-expressing Hela cells incubated with either unconjugated small molecule dye or BZA-conjugated small molecules dye. FIG.16C shows the structure of a BZA shuttle-small molecule cargo variant. The schematic shows the process of conjugating a fluorescent molecule (Alexa Fluor 647) to brinzolamide using a CuAAC reaction. FIG.16D show images from an internalization assay using the HEK cells overexpressing CA-IV receptors. The bottom row shows zoom-in images of representative fields of views in the top-row images. FIG.16E shows representative images of brain slices and liver slices from mice injected with BZA-conjugated fluorophore or unconjugated fluorophores. FIG.16F shows a fluorescence reading of lysed tissues of animals injected with BZA- conjugated fluorophore or unconjugated fluorophores using a plate reader. FIG.18A-B show immunofluorescence images of whole livers sections in mice systemically administered with unconjugated and BZA-conjugated Ate antibodies. FIG.18C shows quantification of BZA-conjugated and unconjugated Ate antibodies in peripheral organs at day 7 in mice systemically administered with unconjugated and BZA- conjugated Ate antibodies. Detailed Description
The present invention provides compositions and methods comprising brinzolamide and brinzolamide derivates as shuttles for the BBB-crossing, for example through the receptor carbonic anhydrase IV (CA-IV). Receptors for Enhanced Blood-Brain Barrier Crossing Blood-brain barrier (BBB) has emerged as a complex, dynamic, adaptable interface that controls the exchange of substances between the central nervous system (CNS) and the blood, to prevent the uncontrolled leakage of substances from the blood into the brain. The cells that make up the structure of the BBB include mostly brain endothelial cells, which constantly communicate with the other cells of the CNS (e.g., astrocytes, microglia, neurons, mast cells and pericytes, as well as circulating immune cells), adapting their behaviors to serve the needs of the CNS, responding to pathological conditions, and in some cases participating in the onset, maintenance or progression of disease. The complexity of BBB functions explains much of the difficulty in developing drugs that can cross the BBB. Utilizing receptors on the BBB interface can offer a method of crossing BBB. The present invention provides shuttles for receptors on the BBB interface and methods of using the same to enhance BBB crossing and CNS potency, such as increasing the permeability of the BBB and delivering a therapeutic agent across the BBB to a nervous system, specifically carbonic anhydrase IV. Without being bound by any theory, the novel target receptors disclosed herein may facilitate enhanced BBB receptor-mediated transcytosis across various species, including mammals such as human. In some embodiments, a method of increasing permeability of the BBB comprises providing a shuttle capable of binding to a BBB crossing receptor (e.g., carbonic anhydrase IV), thereby increasing permeability of the BBB (e.g., through transcytosis). In some embodiments, at least one activity of the BBB-crossing receptor (e.g., carbonic anhydrase IV) can be reduced through binding to a small molecule. Accordingly, in some embodiments, a method of increasing permeability of the BBB comprises reducing the activity of carbonic anhydrase IV, thereby increasing permeability of the BBB. In some embodiments, a shuttle binds to one or more of the zinc binding site (e.g., a catalytic pocket) and substrate binding site of the carbonic anhydrase IV. The carbonic anhydrase IV can be a vertebrate carbonic anhydrase IV including non-human primates and humans. In some embodiments, the carbonic anhydrase IV is a mouse carbonic
anhydrase IV (Car4), a human carbonic anhydrase IV (CA4), or a variant or a homolog thereof. It is understood that CA-IV as used herein refers to any variant. Carbonic Anhydrase IV The carbonic anhydrases (or carbonate dehydratases) (CAs) are a family of enzymes that catalyze the interconversion between carbon dioxide and water and the dissociated ions of carbonic acid. CAs participate in a variety of biological processes, including respiration, calcification, acid-base balance, bone resorption, and the formation of aqueous humor, cerebrospinal fluid, saliva, and gastric acid. The present invention provides shuttles for the BBB-crossing using the receptor carbonic anhydrase IV, capable of facilitating the delivery of a pharmaceutical agent across the BBB (CA- IV shuttles). Carbonic anhydrase IV is an isozyme that belongs to the carbonic anhydrase family, a family of zinc metalloenzymes, which catalyzes the reversible reaction of hydration of CO2 (H2O+CO2⇄HCO3 −+H+), allowing the enzyme to regulate intra- and extra-cellular concentrations of CO2, H+, and HCO3 −. The carbonic anhydrases participate in a variety of biological processes, including respiration, calcification, acid-base balance, bone resorption, and the formation of aqueous humor, cerebrospinal fluid, saliva, and gastric acid. The carbonic anhydrases show extensive diversity in tissue distribution and in their subcellular localization. There are at least seven genetically distinct isozymes of mammalian carbonic anhydrase, designated I-VII, each of which catalyzes the reversible hydration of carbon dioxide by a zinc- hydroxide mechanism. Physiological functions that are regulated by carbonic anhydrase comprise, for example, removal of HCO3− in lung by respiration, reutilization of HCO3− in kidney, production of aqueous humor in eyes, cerebrospinal fluids in brain, gastric juice production in stomach, pancreatic juice, and bone resorption by osteoclasts. Carbonic anhydrase family members also play important roles in metabolic processes that include ureagenesis, gluconeogenesis, and lipogenesis. Different from other carbonic anhydrases that are either soluble or attached to the plasma membrane by a membrane-spanning domain, carbonic anhydrase IV is a glycosylphosphatidyl- inositol-anchored membrane isozyme. Carbonic anhydrase IV is broadly conserved across vertebrates and has similar CNS expression profiles in humans, with a recent single cell analysis of human brain vasculature confirming CA-IV's expression in the human BBB. Carbonic
anhydrase IV has been shown to regulate pH, which is associated with neural discharge and can influence neuronal function through ion-gated channels. In some embodiments, the carbonic anhydrase IV disclosed herein is a human carbonic anhydrase IV. CA-IV is known to localize on the luminal surface of brain endothelial cells throughout the cortex and cerebellum where it enzymatically modulates carbon dioxide- bicarbonate balance. Human CA-IV has been previously characterized as a 35-kDa protein with a “high activity” in CO2 hydration and a higher activity than other isozymes in catalyzing the dehydration of HCO3 −. In general, human CA-IV contains an 18-amino acid signal sequence at the N-terminal of the protein for endoplasmic reticulum (ER) translocation and a 260-amino acid “CA domain” containing active site amino acid residues that shows 30-36% homology with cytoplasmic CAs. At the C-terminal, an additional 27 amino acid residues containing the hydrophobic sequences of 21 amino acids sufficient to span the membrane are preceded by the 6- amino acid signal sequence for GPI-anchoring. The amino acid residue, Ser 266, was identified as the site for the attachment of the GPI anchor. The removal of C-terminal hydrophobic domain found in the CA-IV precursor has important impact on GPI-anchoring, cell surface expression, and realization of the enzyme activity. Based on amino acid sequences deduced from the nucleotide sequence, human CA-IV contains no classical consensus sites (Asn-Xxx-Ser/Thr) for N-glycosylation. Human CA-IV also contains no oligosaccharide chains, while other mammalian carbonic anhydrase IV (e.g. mouse carbonic anhydrases IV) are glycoproteins with one to several oligosaccharide side chains. In some embodiments, the carbonic anhydrase IV disclosed herein is a mouse carbonic anhydrase IV. CA-IV has recently been found to be among the mouse proteins most strongly positively correlated with plasma-protein uptake in the brain (slightly stronger than the often- targeted transferrin receptor). This property is useful for identifying receptors for enhanced BBB crossing. CA-IV is also expressed in the GI tract, kidney, and lung, as well as taste receptor cells where it allows the sensing of carbonation. Mouse and human CA-IV are highly homologous, containing the same amino acids at positions crucial for enzyme activity (e.g., histidine residue 64 (His 64)), with several differences including, for example, that mouse CA-IV is an N-linked glycoprotein and the CO2 hydration rate catalyzed by mouse CA-IV is much lower than human CA-IV. Without being bound by any theory, the lower enzyme activity of mouse CA-IV may be associated with the replacement of Gly 63 in human CA-IV with Gln 63, among several other
amino acid replacements. Another difference between mouse and human CA-IV is the Val-131- Asp-136 segment (130's segment) that forms an α-helix in mouse and an extended loop in human CA-IV. In some embodiments, a carbonic anhydrase IV disclosed herein as a receptor for enhancing BBB crossing can be any carbonic anhydrase IV, such as a mouse CA-IV, a humanCA-IV, or a homology or a variant thereof. Carbonic anhydrase IV homologs and/or variants can be derived from a vertebrate species including, but not limited to, mouse, rat, human, bovine, rabbit, monkey, pig, horse, rainbow trout, chimpanzee, squirrel, chicken, goat, and sheep. Carbonic anhydrase IV homologs from various species can be found in public databases identifiable to a person skilled in the art, including for example UniProt, NCBI, and Swiss-Prot. In some embodiments, a small molecule can interact with a carbonic anhydrase IV disclosed herein (e.g., mouse CA-IV, human CA_IV or a homology or a variant thereof), thereby increasing permeability of the BBB (e.g., through transcytosis). In some embodiments, the increase in the permeability of the BBB is achieved by altering (e.g., increasing or decreasing) the carbonic anhydrase IV activity, such as reducing its activity. In some embodiments, the alteration of carbonic anhydrase IV activity is achieved by a shuttle interacting to one or more active sites of the carbonic anhydrase IV including the zinc binding site and the hydrophobic substrate binding pocket. For example, the shuttle can interacting with the zinc binding site, the hydrophobic substrate binding pocket, or both. The zinc binding site in carbonic anhydrase IV has a conserved structure dominated by a β-sheet super-structure with a metal binding site formed by at least three His residues. Without being bound by any theory, it is believed that the zinc binding site is on one face of the β-sheet at the bottom of a 15-Å-deep, conical active site cleft in which zinc is liganded by three His residues and hydroxide ion with tetrahedral geometry. The hydrophobic substrate binding pocket is adjacent to zinc-bound hydroxide, formed in large part by bulky residues such as Val at its base and Val, Trp and Leu at its neck. This pocket is highly conserved among all active isozymes on the basis of phylogenetic comparisons. Without being bound by any theory, it is believed that the hydrophobic pocket has a minimum width and depth for efficient catalysis, and linear free energy relationships indicate that the volume of the amino acid residue at the base of the pocket and the hydrophobicity of residues at the neck of the pocket are critical for activity. Both the zinc
binding site and the hydrophobic substrate binding pocket are highly conserved among carbonic anhydrase isozymes. Brinzolamide and brinzolamide derivatives Brinzolamide (BZA) is a highly specific, non-competitive, reversible carbonic anhydrase II (CA II) inhibitor indicated to reduce ocular pressure in patients with ocular hypertension or open-angle glaucoma. Brinzolamide was approved by the FDA in 1998 as a topical product under the trade name AZOPT and later as combination products with timolol under the trade name AZARGA and brimonidine tartrate under the trade name SIMBRINZA. N-Desethylbrinzolamide is an active metabolite of brinzolamide, exhibiting CA 1 inhibitory activity, when in the presence of Brinzolamide, and also accumulates in the erythrocytes. However, Brinzolamide's other known metabolites (N- Desmethoxypropylbrinzolamide and O-Desmethylbrinzolamide) either have no activity or their activity is currently unknown. The structure for Brizolamide is:
with a molecular formula of C12H21N3O5S3 and IUPAC name (4R)-4-(ethylamino)-2-(3- methoxypropyl)-1,1-dioxo-3,4-dihydrothieno[3,2-e]thiazine-6-sulfonamide. By the present invention it was discovered the brinzolamide, and particularly brinzolamide derivates, may act as shuttles for receptors on the BBB interface and methods of using the same to enhance BBB crossing and CNS potency, such as increasing the permeability of the BBB and delivering a therapeutic agent across the BBB to a nervous system, specifically carbonic anhydrase IV. This invention proposes an innovative method for surmounting this challenge using receptor-mediated transcytosis (RMT), capitalizing on Carbonic Anhydrase IVCA-IV as a BBB
the first time that brinzolamide, a carbonic anhydrase inhibitor, binds to the mouse CA-IV at the same catalytic site as some BBB-crossing capsids (including 9P31 and 9P36), implying a potential mechanism for transporting therapeutic agents across the BBB. Brinzolamide showcases binding affinity to CA-IV, with a half maximal inhibitory concentration (IC50) of 45 nM. Its safety profile is well established, with an oral LD50 in rats between 1000 to 2000 mg/kg, and it has an extraordinarily long half-life in whole blood (111 days) due to its strong adherence to carbonic anhydrase-containing erythrocytes and minimal metabolism. Importantly, by the present invention it was shown that the brinzolamide binding pocket on CA-IV is conserved across species, promising that therapeutic molecules conjugated to brinzolamide can be used in across animals and animal models. As described further below, chemical ligation reactions like the lysine–N- hydroxysuccinimide (NHS) ester coupling or Copper(I)-catalyzed azide-alkyne cycloaddition (CuAAC) may be used in the instant invention to conjugate brinzolamide (BZA) derivatives with therapeutic cargoes, including nanobodies, therapeutic IgG antibodies, and small molecules. These derivatives are specifically designed to function as effective binders to CA-IV. As described herein, cell-based assays, surface plasmon resonance (SPR), and in vivo intravenous delivery have been utilized to verify the successful binding and subsequent internalization of these conjugated molecules to CA-IV. Accordingly, aspects of the invention provide of select brinzolamide derivates. For example, aspects of the invention provide brinzolamide derivates synthesized using the linkers and esters as described below: BZA-2C-NHS (1-6)
AcOH = acetic acid, THF = tetrahydrofuran, DCM = dichloromethane, DMAP = 4- dimethylaminopyridine, EDCI = N-(3-Dimethylaminopropyl)-N′-ethylcarbodiimide hydrochloride, TSTU = N,N,N′,N′-tetramethyl-O-(N-succinimidyl)uronium tetrafluoroborate, DIPEA = N,N- diisopropylethylamine Under nitrogen, to an oven-dried 1-dram vial equipped with a Teflon-coated stir bar was added NaBH4 (4.93 mg, 0.13 mmol, 1.0 equiv.). The solid was suspended in anhydrous THF (0.44 mL) and the suspension was cooled to 0 °C in an ice bath. Acetic acid (33.6 µL, 35.2 mg, 059 mmol 45 equiv) was slowly added (gas evolution) The resulting solution was stirred at 0
°C for 30 minutes, before it was warmed to room temperature (23 C) and stirred for further 30 minutes. Brinzolamide (1) (50.0 mg, 0.13 mmol, 1.0 equiv.) and acetic acid (11.2 µL, 11.7 mg, 0.20 mmol, 1.5 equiv.) were subsequently added. After Brinzolamide had fully dissolved (approx.5 minutes), acetaldehyde (5 M in THF, 26.1 µL, 0.13 mmol, 1.0 equiv.) was added dropwise. The reaction mixture was stirred at room temperature for 18 hours. Afterwards, saturated aqueous NaHCO3-solution (1.0 mL) was added. The aqueous phase was extracted with EtOAc (3 x 3 mL). The combined organic phases were dried over Na2SO4, filtered, and the volatile materials were evaporated under reduced pressure. The crude material was purified by column chromatography on silica (hexanes:EtOAc = 2:3) to afford the tertiary amine 2 as a colorless oil (53.7 mg, 0.13 mmol, quant.). 1H NMR (500 MHz, CDCl3): δ 7.63 (s, 1H), 5.50 (s, 2H), 4.27 (dd, J = 10.7, 5.3 Hz, 1H), 4.14– 4.03 (m, 1H), 3.61–3.48 (m, 3H), 3.44 (ddd, J = 9.6, 6.2, 5.3 Hz, 1H), 3.34 (s, 3H), 3.10 (dt, J = 13.9, 7.0 Hz, 1H), 2.57 (qq, J = 13.5, 6.9, 6.3 Hz, 4H), 1.92 (dqd, J = 12.1, 6.6, 6.2, 3.4 Hz, 2H), 1.06 (t, J = 7.1 Hz, 6H). 13C{1H} NMR (126 MHz, CDCl3): δ 146.7, 145.7, 139.3, 130.8, 69.3, 58.8, 52.8, 47.6, 46.0, 44.7, 29.6, 15.0. FTIR (NaCl, thin film): 3271, 2968, 2932, 1459, 1344, 1172, 1158, 1104, 1040, 1016, 653 cm-1. HRMS: (ESI-TOF) calc’d for C14H26N3O5S3 [M+H]+ 412.1029, found 412.1065. TLC (1:4 hexanes:EtOAc), Rf: 0.64 (UV).
Under nitrogen, in a 2-dram vial, a suspension of 2 (104 mg, 253 µmol, 1.0 equiv.) in CH2Cl2 (5.1 mL) was cooled to −78 °C in a dry ice-acetone bath. To the suspension was added boron tribromide (1.01 mL, 1.01 mmol, 4.0 equiv.) dropwise. The reaction mixture was allowed to stir for 10 min at −78 °C, before it was gradually warmed to room temperature (23 °C). After being stirred at room temperature for 20 hours, it was cooled to 0 °C in an ice bath and MeOH (2.5 mL) was carefully added. The resulting homogeneous mixture was stirred at room temperature for 30 min. Afterwards, the volatile materials were evaporated, and the crude
material was purified by preparative HPLC (C18 column, 9.4 x 250 mm) eluting with 20% MeCN over 2.5 minutes at a flow of 5 mL/min, then a gradient of 20 to 70% MeCN over 10 minutes at a flow rate of 5 mL/min, and then 100% MeCN over 2.5 minutes at a flow rate of 5 mL/min. Bromide 4 (14.8 mg, 32.1 µmol, 13%) was obtained as a colorless oil and alcohol 3 (42.4 mg, 107 µmol, 42%) was obtained as a colorless oil. Analytical data for alcohol 3: 1H NMR (400 MHz, CD3CN): δ 7.67 (s, 1H), 6.24 (s, 2H), 4.45 (s, 1H), 4.03 (dd, J = 15.0, 10.5 Hz, 1H), 3.77–3.69 (m, 1H), 3.57 (t, J = 6.1 Hz, 2H), 3.49 (dt, J = 14.3, 7.4 Hz, 1H), 3.09 (dt, J = 13.6, 6.8 Hz, 1H), 2.85–2.59 (m, 4H), 1.96 (s, 1H), 1.87–1.75 (m, 2H), 1.08 (t, J = 7.2 Hz, 6H). 13C{1H} NMR (101 MHz, CD3CN): δ 148.7, 139.2, 131.3, 59.3, 53.3, 47.6, 46.3, 45.6, 35.4, 32.6, 14.9. FTIR (NaCl, thin film): 3347, 2963, 2653, 2263, 1558, 1453, 1346, 1156, 918, 741 cm-1. HRMS: (ESI-TOF) calc’d for C13H24N3O5S3 [M+H]+ 398.0873, found 398.0881. TLC (95:5 CH2Cl2:MeOH), Rf: 0.28 (UV). Analytical data for bromide 4: 1H NMR (400 MHz, CD3CN): δ 7.58 (s, 1H), 6.14 (s, 2H), 4.39 (dd, J = 10.7, 5.5 Hz, 1H), 4.02 (ddd, J = 15.1, 10.8, 1.1 Hz, 1H), 3.67 (dd, J = 15.0, 5.5 Hz, 1H), 3.62 – 3.48 (m, 3H), 3.16 (dt, J = 13.9, 6.4 Hz, 1H), 2.57 (q, J = 7.1 Hz, 4H), 2.22–2.12 (m, 2H), 1.05 (t, J = 7.1 Hz, 6H). 13C{1H} NMR (101 MHz, CD3CN): δ 148.7, 147.4, 138.7, 131.2, 53.3, 48.2, 47.7, 45.3, 32.8, 31.6, 15.2. FTIR (NaCl, thin film): 3356, 3274, 3094, 2969, 1152, 1452, 1342, 1157, 911, 648 cm-1. HRMS: (ESI-TOF) calc’d for C13H23BrN3O4S3 [M+H]+ 460.0029, found 460.0034. TLC (3:7 hexanes:EtOAc), Rf: 0.77 (UV).
In a 2-dram vial equipped with a stir bar, alcohol 3 (100 mg, 252 µmol, 1.0 equiv.) was dissolved in acetone (3.4 mL). The solution was cooled to 0 °C in an ice bath. Jones reagent (2.5 M solution in 3:1 water/concentrated H2SO4, 473 µL, 1.18 mmol, 4.7 equiv.) was added dropwise. The reaction mixture was stirred at 0 °C for two hours, before neopentyl-alcohol (250 mg) was added in one portion. The mixture was stirred for one hour at room temperature (23 °C). Afterwards, the mixture was filtered through a cotton plug, washing with acetone. To the solution was then added silica, the suspension was concentrated, and subjected to purification by column chromatography (CH2Cl2:MeOH = 93:7 to 9:1). Carboxylic acid 5 (85.4 mg, 208 µmol, 82%) was isolated as a colorless oil. The carboxylic acid is not stable towards acidic conditions. Prolonged exposure to silica resulted in decomposition. 1H NMR (500 MHz, CD3OD): δ 7.58 (s, 1H), 4.44 (dd, J = 10.7, 5.5 Hz, 1H), 4.09–3.99 (m, 1H), 3.78 (dd, J = 14.9, 5.6 Hz, 1H), 3.65–3.56 (m, 1H), 3.37 (ddd, J = 13.9, 7.7, 6.1 Hz, 1H), 2.73–2.63 (m, 2H), 2.60 (q, J = 7.1 Hz, 4H), 2.02 (s, 1H), 1.08 (t, J = 7.1 Hz, 6H). 13C{1H} NMR (101 MHz, CD3CN): δ 173.0, 148.7, 147.5, 138.6, 131.2, 53.5, 48.8, 45.6, 45.3, 35.0, 15.2. HRMS: (ESI-TOF) calc’d for C13H22N3O6S3 [M+H]+ 412.0665, found 412.0699.
In a 20 mL scintillation vial equipped with a stir bar, carboxylic acid 5 (25.0 mg, 60.8 µmol, 1.0 equiv.) was dissolved in a mixture of MeCN (2.28 mL) and H2O (759 µL) and cooled to 0 ˚C. To this solution, N,N,N′,N′-tetramethyl-O-(N-succinimidyl)uronium tetrafluoroborate (TSTU) (36.6 mg, 122 µmol, 2.0 equiv.) and N,N-diisopropylethylamine (21.2 µL, 15.7 mg, 122 µmol, 2.0 equiv.) were added. After 20 minutes at 0 ˚C, the volatile materials were evaporated under reduced pressure. The crude reaction mixture was taken up in MeCN (approx.1.5 mL) and purified by preparative HPLC (C18 column, 9.4 x 250 mm) eluting with a gradient of 40 to 50%
MeCN over 11 minutes at a flow rate of 5 mL/min. NHS-ester 6 (9.5 mg, 18.7 μmol, 31%) was obtained as a colorless oil. 1H NMR (400 MHz, CD3CN) δ 7.58 (s, 1H), 6.14 (s, 2H), 4.39 (dd, J = 10.8, 5.5 Hz, 1H), 4.02 (dddd, J = 15.0, 10.3, 9.3, 0.9 Hz, 1H), 3.81–3.55 (m, 2H), 3.39 (ddt, J = 53.6, 14.0, 6.9 Hz, 1H), 3.10–2.95 (m, 1H), 2.78 (s, 2H), 2.67 (td, J = 6.7, 1.8 Hz, 1H), 2.62–2.48 (m, 4H), 1.96 (s, 2H), 1.04 (td, J = 7.1, 1.4 Hz, 6H). 13C{1H} NMR (101 MHz, CD3CN) δ 173.1, 172.8, 171.0, 168.2, 148.9, 147.6, 138.3, 131.2, 53.6, 49.2, 45.3, 34.9, 32.8, 26.4, 15.2. HRMS: (ESI-TOF) calc’d for C17H25N4O8S3 [M+H]+ 509.0829, found 509.0839. BZA-N3 (7)
In a 1-dram vial equipped with a stir bar, bromide 4 (14.8 mg, 32.6 µmol, 1.0 equiv.) and sodium azide (6.35 mg, 97.7 µmol, 3.0 equiv.) were dissolved in dimethylformamide (163 µL). The solution was heated to 80 °C overnight. Afterwards, the mixture was diluted with EtOAc (1.5 mL) and washed with brine (1.5 mL). The aqueous layer was extracted 3 times with EtOAc (3 mL), and the combined organic layers were dried over MgSO4. The dried organic layer was concentrated in vacuo and to the resulting residue was then added silica, and subjected to purification by column chromatography (EtOAc:Hexanes= 6:4). Azide 7 (6.20 mg, 14.7 µmol, 45%) was isolated as a colorless oil. 1H NMR (500 MHz, CD3Cl): δ 7.69 (s, 1H), 5.24 (s, 2H), 4.30 (s, 1H), 4.14 (dd, J = 14.7, 10.6 Hz, 1H), 3.67–3.36 (m, 4H), 3.07 (dt, J = 13.7, 6.7 Hz, 1H), 2.61 (tq, J = 13.1, 6.4 Hz, 4H), 2.00– 1.84 (m, 2H), 1.07 (t, J = 7.1 Hz, 6H). 13C{1H} NMR (101 MHz, CD3Cl): δ 146.9, 139.6, 130.9, 52.9, 48.5, 47.9, 46.3, 44.9, 32.1, 29.9, 28.9, 14.9. FTIR (NaCl, thin film): 3334, 2923, 2358, 2097, 1618, 1458, 1342, 1259, 1154, 845, 608 cm-1. HRMS: (ESI-TOF) calc’d for C13H23N6O4S3 [M+H]+ 423.0937, found 423.1033.
TLC (6:4 Hexanes:EtOAc), Rf: 0.67 (UV). BZA-nC-NHS (n = 1, 3, 4, 6, and 8) General procedure 1: Alkylation of the BZA core (8)
In a 50 mL round-bottom flask equipped with a stir bar, (S)-6-chloro-4-hydroxy-3,4- dihydro-2H-thieno[3,2-e][1,2]thiazine 1,1-dioxide (8) (2.40 g, 10.0 mmol, 1.0 equiv.) was dissolved in DMSO (12.0 mL), and potassium carbonate (4.15 g, 30.0 mmol, 3.0 equiv.) was added. The suspension was stirred at room temperature and a solution of the bromoalkyl methyl ether (12.0 mmol, 1.2 equiv.) in DMSO (2.0 mL) was added dropwise over the course of 1 hour. After completion of addition, the reaction mixture was stirred for a further 1.5 h. The mixture was then poured into brine (20 mL) and extracted with Et2O (3x). The combined extracts were washed with 1 M NaOH (30 mL), 1:1 bleach/water (40 mL), and dried over Na2SO4, filtered, and the volatile materials were evaporated under reduced pressure. The crude material was purified by column chromatography on silica to afford the alkylated BZA core. General procedure 2: Tosylation of the alcohol
In a 100 mL round-bottom flask equipped with a stir bar, the corresponding secondary alcohol (1.0 equiv.) and triethylamine (2.5 equiv.) were dissolved in THF(0.5 M with respect to alcohol) and the solution was cooled to 0 °C in an ice bath. To this solution was added dropwise a solution of p-toluenesulfonyl chloride (2.0 equiv.) in THF (2 M with respect to p-TsCl). After addition, the reaction mixture was warmed to room temperature and stirred for 3 hours. Water
was then added, and the aqueous phase was extracted with ethyl acetate (3x). The combined organic layers were dried over Na2SO4, filtered, and the volatile materials were evaporated under reduced pressure. The crude material was purified by column chromatography on silica to afford the tosyl-protected alcohol.
In a 100 mL round-bottom flask equipped with a stir bar, to a solution of the corresponding tosylate (1.0 equiv.) in MeCN (0.25 M) was added potassium carbonate (1.0 equiv.) and di ethylamine (1.1 equiv.) at room temperature. A reflux condenser was attached to the flask. The reaction mixture was warmed to 70 °C in an oil bath and stirred for 14 hours. After cooling the reaction mixture to room temperature, water was added. The aqueous phase was extracted with DCM (3x). The combined organic layers were dried over Na2SO4, filtered, and evaporated. The crude material was purified by column chromatography on silica to afford the tertiary amine.
In a 25 mL round-bottom flask equipped with a stir bar, to a solution of the corresponding thiophene chloride (1.0 equiv.) in DMF (0.5 M) was added potassium carbonate (2.0 equiv.) and benzyl mercaptan (2.0 equiv.) at room temperature. The reaction mixture was heated to 90 °C and stirred for 14 hours. Afterwards, the reaction was cooled to room
temperature. Water was added, and the aqueous phase was extracted with Et2O (3x). The combined organic phases were dried over Na2SO4, filtered, and concentrated under reduced pressure. The crude material was purified by column chromatography on silica to afford the benzyl sulfide. General procedure 5: Oxidation of the benzyl sulfide
In a 100 mL round-bottom flask equipped with a stir bar, the corresponding benzyl sulfide (1.0 equiv.) was dissolved in a mixture of MeCN, acetic acid, and water (20:3:1, 0.05 M). The solution was cooled to 0 °C in an ice bath and sulfuryl chloride (10 equiv.) was added dropwise. The reaction mixture was stirred for 1 hour, while keeping the reaction temperature at approximately 5 °C. Afterwards, the solvent was evaporated under reduced pressure. The residue was dissolved in MeOH (0.05 M) and cooled to 0 °C in an ice bath. Ammonia (2.77 mL/mmol of benzyl sulfide starting material) was added and the resulting suspension was stirred for 3 hours at 0 °C. Water was added and the aqueous phase was extracted with EtOAc (3x). The combined organic phases were dried over Na2SO4, filtered, and the volatile materials were evaporated. The crude material was purified by column chromatography on silica to afford the corresponding sulfonamide. General procedure 6: Deprotection of the methoxy group
In a 20 mL scintillation vial equipped with a stir bar, to a solution of the corresponding alkyl methoxide (1.0 equiv.) in MeCN (0.50 M) at room temperature was added sodium iodide (6.0 equiv.) and methyltrichlorosilane (6.0 equiv.). The resulting mixture was stirred at room temperature for 14 hours. Afterwards, sat. aq. Na2S2O3 solution was added and the aqueous phase was extracted with DCM (3x). The combined organic phases were dried over Na2SO4, filtered, and the volatile materials were evaporated. Column chromatography on silica afforded the corresponding free alcohol. General procedure 7: Jones oxidation to the carboxylic acid
The corresponding alcohol (1.0 equiv.) was dissolved in acetone (0.075 M) and the solution was cooled to 0 °C in an ice bath. Jones reagent (2.5 M solution in 3:1 water/concentrated H2SO4, 4.7 equiv.) was added dropwise. The reaction mixture was stirred at 0 °C for two hours, before neopentyl-alcohol (1 mg/µmol of starting material) was added in one portion. The mixture was stirred for one hour at room temperature. Afterwards, the mixture was filtered through a cotton plug, washing with acetone. If not indicated otherwise, to the solution was added silica, the suspension was concentrated, and subjected to purification by column chromatography on silica to afford the corresponding carboxylic acid. General procedure 8: NHS-ester formation
In a 20 mL scintillation vial equipped with a stir bar, the corresponding carboxylic acid (1.0 equiv.) was dissolved in a mixture of MeCN and H2O (3:1, 0.02 M) and cooled to 0 ˚C. To this solution, N,N,N′,N′-tetramethyl-O-(N-succinimidyl)uronium tetrafluoroborate (TSTU) (2.0 equiv.) and N,N-diisopropylethylamine (2.0 equiv.) were added. After 20 minutes at 0 ˚C, the volatile materials were evaporated under reduced pressure. The crude reaction mixture was taken up in MeCN (approx.1.5 mL) and purified by preparative HPLC (C18 column, 9.4 x 250 mm) eluting with a mixture of water/acetonitrile to afford the corresponding NHS-ester. BZA-1C-NHS (9) Compound S1
Compound S1 was synthesized according to general procedure 1 on a 10.0 mmol scale, using 1-bromo-2-methoxyethane (1.13 mL, 1.67 g, 12.0 mmol, 1.2 equiv.). After column chromatography on silica (hex:EtOAc = 4:1 to 7:3), the target compound was isolated as a colorless oil (3.00 g, 10.0 mmol, quant.). 1H NMR (400 MHz, CDCl3): δ 6.98 (s, 1H), 4.56 (ddd, J = 8.6, 4.2, 2.8 Hz, 1H), 4.36 (dd, J = 15.8, 4.2 Hz, 1H), 4.11 (d, J = 8.8 Hz, 1H), 3.90–3.80 (m, 2H), 3.75 (ddd, J = 10.5, 7.6, 2.7 Hz, 1H), 3.60 (ddd, J = 10.8, 5.9, 2.9 Hz, 1H), 3.42 (ddd, J = 14.8, 7.5, 2.8 Hz, 1H), 3.30 (s, 3H), 1.60 (s, 1H). 13C{1H} NMR (101 MHz, CDCl3): δ 143.8, 135.9, 133.2, 126.4, 71.8, 62.0, 58.9, 55.2, 50.0. FTIR (NaCl, thin film): 3425, 2924, 2359, 1420, 1331, 1156, 1026, 833, 734, 668 cm-1. HRMS: (ESI-TOF) calc’d for C19H12ClNaNO4S2 [M+Na]+ 319.9788, found 319.9802. TLC (3:7 hexanes:EtOAc), Rf: 0.58 (UV).
Compound S2
Compound S2 was synthesized according to general procedure 2 on a 10.0 mmol scale. After column chromatography on silica (hex:EtOAc = 3:1), the target compound was isolated as a colorless oil (3.70 g, 8.17 mmol, 82%). 1H NMR (500 MHz, CDCl3): δ 7.90–7.76 (m, 2H), 7.46–7.38 (m, 2H), 6.64 (s, 1H), 5.37 (dd, J = 4.1, 3.1 Hz, 1H), 4.24 (dd, J = 16.2, 4.1 Hz, 1H), 4.11 (dd, J = 16.2, 3.1 Hz, 1H), 3.69–3.62 (m, 2H), 3.62–3.53 (m, 2H), 3.41–3.36 (m, 1H), 3.35 (s, 3H), 2.51 (s, 3H). 13C{1H} NMR (125 MHz, CDCl3): δ 146.1, 136.9, 136.4, 135.7, 133.0, 130.4, 128.1, 126.2, 72.2, 68.3, 59.1, 52.1, 49.7, 21.9. FTIR (NaCl, thin film): 2922, 2357, 1597, 1427, 1356, 1168, 1119 cm-1. HRMS: (ESI-TOF) calc’d for C16H18NNaClO6S3 [M+Na]+ 473.9877, found 473.9896. TLC (7:3 hexanes:EtOAc), Rf: 0.64 (UV).
Compound S3 was synthesized according to general procedure 3 on a 8.16 mmol scale. After column chromatography on silica (hex:EtOAc = 85:15), the target compound was isolated as a colorless oil (730 mg, 2.07 mmol, 25%). 1H NMR (500 MHz, CDCl3): δ 6.97 (s, 1H), 4.28 (dd, J = 10.8, 5.2 Hz, 1H), 4.12 (ddd, J = 14.7, 10.7, 0.7 Hz, 1H), 3.75 (dd, J = 14.6, 5.2 Hz, 1H), 3.66–3.56 (m, 3H), 3.36 (s, 3H), 3.28–3.17 (m, 1H), 2.65 – 2.45 (m, 4H), 1.06 (t, J = 7.1 Hz, 6H). 13C{1H} NMR (125 MHz, CDCl3): δ 146.0, 135.5, 132.8, 126.3, 72.5, 59.1, 52.1, 48.2, 47.7, 44.5, 14.8. FTIR (NaCl, thin film): 3646, 3331, 2924, 2359, 1715, 1557, 1531, 1455, 1418, 1356, 1169 cm-1. HRMS: (ESI-TOF) calc’d for C13H22ClN2O3S2 [M+H]+ 353.0755, found 353.0862. TLC (85:15 hexanes:EtOAc), Rf: 0.15 (UV). Compound S4
Compound S4 was synthesized according to general procedure 4 on a 2.05 mmol scale. After column chromatography on silica (hex:EtOAc = 85:15), the target compound was isolated as a yellow oil (634 mg, 1.44 mmol, 70%). 1H NMR (500 MHz, CDCl3): δ 7.35–7.15 (m, 5H), 6.89 (s, 1H), 4.23 (dd, J = 10.7, 5.3 Hz, 1H), 4.14–3.94 (m, 3H), 3.70 (dd, J = 14.6, 5.3 Hz, 1H), 3.66–3.49 (m, 3H), 3.36 (s, 3H), 3.25–3.15 (m, 1H), 2.48 (qd, J = 7.2, 5.2 Hz, 4H), 1.01 (t, J = 7.1 Hz, 6H). 13C{1H} NMR (125 MHz, CDCl3): δ 145.9, 140.0, 136.6, 136.0, 132.1, 128.9, 128.6, 127.7, 72.3, 58.9, 52.1, 48.0, 47.9, 44.4, 43.0, 14.8. FTIR (NaCl, thin film): 2923, 2550, 2331, 1724, 1333, 1142, 678 cm-1. HRMS: (ESI-TOF) calc’d for C20H28N2O3S3 [M+H]+ 440.1262, found 441.1323. TLC (85:15 hexanes:EtOAc), Rf: 0.13 (UV).
Compound S5
Compound S5 was synthesized according to general procedure 5 on a 1.37 mmol scale. After column chromatography on silica (DCM:MeOH = 99:1), the target compound was isolated as a brown oil (444 mg, 1.12 mmol, 81%). The compound was isolated with an aromatic impurity which could be separated in the subsequent step. 1H NMR (400 MHz, CDCl3): δ 7.64 (s, 1H), 5.33 (bs, 2H), 4.43 (d, J = 5.7 Hz, 1H), 4.34 (dd, J = 10.7, 5.3 Hz, 1H), 4.13 (dd, J = 14.7, 10.7 Hz, 1H), 3.79 (dd, J = 14.7, 5.3 Hz, 1H), 3.66–3.56 (m, 3H), 3.36 (s, 3H), 3.32–3.19 (m, 1H), 2.56 (dtt, J = 19.9, 12.9, 7.0 Hz, 4H), 1.05 (t, J = 7.1 Hz, 6H). 13C{1H} NMR (101 MHz, CDCl3): δ 146.6, 146.1, 139.6, 130.9, 72.3, 59.1, 52.7, 48.3, 47.7, 44.6, 44.0, 23.5, 14.8. FTIR (NaCl, thin film): 3330, 2925, 2850, 2356, 2336, 1714, 1458, 1270, 718, 615 cm-1. HRMS: (ESI-TOF) calc’d for C13H23N3O5S3 [M]+ 397.0800, found 397.0582. TLC (99:1 DCM:MeOH), Rf: 0.16 (UV). Compound S6
Under N2, in a 100 mL round-bottom flask equipped with a stir bar, a suspension of alkyl methoxide S5 (444 mg, 1.12 mmol, 1.0 equiv.) in DCM (22.3 mL) was cooled to −78 °C. Boron
tribromide (1.0 M solution in DCM, 4.47 mL, 4.47 mmol, 4.0 equiv.) was added dropwise. The reaction mixture was allowed to stir at −78 °C for 10 minutes and then gradually warmed to room temperature. After being stirred at room temperature for 14 hours, it was cooled to 0 °C and carefully quenched by addition of MeOH (~21 mL/mmol of starting material). The resulting colorless homogeneous mixture was then stirred at room temperature for 30 min. The solvent was evaporated and the residue was purified by column chromatography on silica (DCM:MeOH 100% to 8:2) to afford the target compound S6 as a brown oil (391 mg, 1.02 mmol, 91%). 1H NMR (400 MHz, acetone-d6): δ 9.00 (s, 1H), 5.45 (dd, J = 8.2, 6.3 Hz, 1H), 4.69 – 4.45 (m, 2H), 3.82 (qdd, J = 11.7, 6.3, 3.8 Hz, 2H), 3.67–3.51 (m, 2H), 3.43 (tdq, J = 14.2, 6.7, 3.7 Hz, 2H), 3.22 (dq, J = 13.2, 7.3 Hz, 1H), 2.09–2.00 (m, 4H), 1.64 (t, J = 7.2 Hz, 3H), 1.47 (t, J = 7.3 Hz, 3H). 13C{1H} NMR (101 MHz, acetone-d6): δ 150.5, 142.6, 136.2, 132.5, 61.2, 53.7, 51.2, 48.3, 47.5, 46.7, 11.1, 10.3. FTIR (NaCl, thin film): 3733, 3550, 2356, 2342, 1754, 1524, 1110, 741, 668 cm-1. HRMS: (ESI-TOF) calc’d for C13H22N3O5S3 [M+H]+ 384.0716, found 384.0762. TLC (8:2 DCM:MeOH), Rf: 0.2 (UV). Compound S7
Compound S7 was synthesized according to general procedure 7 on a 130 µmol scale. After column chromatography on silica (DCM:MeOH = 8:2), the target compound was isolated as a white foam (45.8 mg, 115 µmol, 88%).
Compound 9
Compound 9 was synthesized according to general procedure 8 on a 94.6 µmol scale. Since the compound was not stable towards concentration, the crude reaction mixture was directly used for the next step. BZA-3C-NHS (10) Compound S8
Compound S8 was synthesized according to general procedure 1 on a 10.0 mmol scale, using 1-bromo-4-methoxybutane (2.69 mL, 2.00 g, 12.0 mmol, 1.2 equiv.). After column chromatography on silica (hex:EtOAc = 4:1 to 7:3), the target compound was isolated as a colorless oil (3.61 g, 11.1 mmol, quant.). 1H NMR (500 MHz, CDCl3): δ 6.96 (s, 1H), 4.69 (dt, J = 7.8, 4.5 Hz, 1H), 4.03 (dd, J = 15.3, 4.3 Hz, 1H), 3.80 (dd, J = 15.3, 4.8 Hz, 1H), 3.49–3.36 (m, 3H), 3.34–3.26 (m, 4H), 2.86 (d, J = 7.4 Hz, 1H), 1.79–1.70 (m, 2H), 1.71–1.59 (m, 2H). 13C{1H} NMR (125 MHz, CDCl3): δ 143.1, 136.3, 132.9, 125.9, 72.0, 61.7, 58.7, 53.0, 49.0, 26.6, 25.2. FTIR (NaCl, thin film): 3363, 2934, 2358, 2356, 1419, 1337, 1163, 1027, 731 cm-1.
HRMS: (ESI-TOF) calc’d for C11H17ClNO4S2 [M+H]+ 326.0282, found 326.0303. TLC (7:3 hexanes:EtOAc), Rf: 0.33 (UV). Compound S9
Compound S9 was synthesized according to general procedure 2 on a 10.0 mmol scale. After column chromatography on silica (hex:EtOAc = 4:1 to 3:2), the target compound was isolated as a colorless oil (4.82 g, 10.0 mmol, quant.). 1H NMR (500 MHz, CDCl3): δ 7.92–7.71 (m, 2H), 7.50–7.35 (m, 2H), 6.59 (s, 1H), 5.32 (dd, J = 3.9, 2.4 Hz, 1H), 4.21 (ddd, J = 16.3, 3.9, 0.8 Hz, 1H), 3.90 (dd, J = 16.3, 2.5 Hz, 1H), 3.39 (t, J = 6.0 Hz, 2H), 3.34 (s, 3H), 3.16 (ddd, J = 13.3, 7.3, 5.6 Hz, 1H), 2.51 (s, 3H), 1.80–1.47 (m, 4H). 13C{1H} NMR (125 MHz, CDCl3): δ 146.3, 136.4, 136.4, 135.6, 132.9, 130.5, 128.0, 126.1, 72.0, 67.9, 58.8, 50.5, 49.6, 26.6, 25.0, 21.9. FTIR (NaCl, thin film): 2927, 2357, 1339, 1169, 1114, 921, 739 cm-1. HRMS: (ESI-TOF) calc’d for C18H26ClN2O6S3 [M+NH4]+ 497.0636, found 497.0645. TLC (7:3 hexanes:EtOAc), Rf: 0.58 (UV). Compound S10
Compound S10 was synthesized according to general procedure 3 on a 10.0 mmol scale. After column chromatography on silica (hex:EtOAc = 85:15), the target compound was isolated as a colorless oil (2.10 g, 5.51 mmol, 55%). 1H NMR (500 MHz, CDCl3): δ 6.95 (s, 1H), 4.18 (dd, J = 10.9, 5.1 Hz, 1H), 4.07 (dd, J = 14.7, 10.8 Hz, 1H), 3.52 (ddt, J = 20.5, 14.2, 4.3 Hz, 2H), 3.41 (t, J = 6.0 Hz, 2H), 3.33 (s, 3H), 2.98 (ddd, J = 13.2, 7.2, 5.5 Hz, 1H), 2.67–2.46 (m, 4H), 1.85–1.59 (m, 4H), 1.06 (t, J = 7.1 Hz, 6H). 13C{1H} NMR (125 MHz, CDCl3): δ 145.4, 135.5, 133.0, 126.2, 72.1, 58.8, 52.1, 48.0, 46.3, 44.7, 26.7, 25.6, 15.0. FTIR (NaCl, thin film): 29332355, 1417, 1339, 1165, 1115, 733 cm-1. HRMS: (ESI-TOF) calc’d for C15H26ClN2O3S2 [M+H]+ 381.1068, found 381.1289. TLC (8:2 hexanes:EtOAc), Rf: 0.46 (UV). Compound S11
Compound S11 was synthesized according to general procedure 4 on a 5.49 mmol scale. After column chromatography on silica (hex:EtOAc = 85:15), the target compound was isolated as a yellow oil (1.39 g, 2.96 mmol, 54%). 1H NMR (500 MHz, CDCl3): δ 7.32–7.19 (m, 5H), 6.86 (s, 1H), 4.13 (dd, J = 10.8, 5.2 Hz, 1H), 4.05 (s, 2H), 4.03–3.94 (m, 1H), 3.47 (ddd, J = 20.6, 14.6, 6.5 Hz, 2H), 3.41 (t, J = 5.9 Hz, 2H), 3.33 (s, 3H), 2.95 (ddd, J = 13.2, 7.3, 5.5 Hz, 1H), 2.49 (qd, J = 7.1, 3.8 Hz, 4H), 1.88–1.50 (m, 4H), 1.01 (t, J = 7.1 Hzi, 6H). 13C{1H} NMR (125 MHz, CDCl3): δ 145.4, 140.1, 136.7, 136.3, 132.1, 129.1, 128.8, 127.8, 72.1, 58.8, 52.2, 47.9, 46.7, 44.7, 43.2, 26.7, 25.6, 15.0. FTIR (NaCl, thin film): 2924, 2358, 2356, 1338, 1166, 1114, 737, 668 cm-1. HRMS: (ESI-TOF) calc’d for C22H33N2O3S3 [M+H]+ 469.1648, found 469.1647. TLC (8:2 hexanes:EtOAc), Rf: 0.56 (UV).
Compound S12
Compound S12 was synthesized according to general procedure 5 on a 2.94 mmol scale. After column chromatography on silica (DCM:MeOH = 99:1), the target compound was isolated as a brown oil (855 mg, 2.01 mmol, 68%). 1H NMR (400 MHz, CDCl3): δ 7.63 (s, 1H), 5.31 (bs, 2H), 4.24 (dd, J = 10.8, 5.2 Hz, 1H), 4.14– 3.99 (m, 1H), 3.58 (dd, J = 14.8, 5.3 Hz, 1H), 3.53–3.44 (m, 1H), 3.42 (t, J = 5.9 Hz, 2H), 3.33 (s, 3H), 3.00 (ddd, J = 13.3, 7.2, 5.5 Hz, 1H), 2.58 (dddd, J = 20.0, 13.1, 7.1, 5.9 Hz, 4H), 1.86–1.52 (m, 4H), 1.06 (t, J = 7.1 Hz, 6H). 13C{1H} NMR (101 MHz, CDCl3): δ 146.6, 139.8, 130.9, 128.9, 72.1, 58.8, 52.7, 48.2, 46.5, 44.8, 44.0, 26.6, 25.7, 23.5, 15.0. FTIR (NaCl, thin film): 3733, 3256, 2356, 2354, 1521, 1167, 831, 733, 673 cm-1. HRMS: (ESI-TOF) calc’d for C15H28N3O5S3 [M+H]+ 426.1186, found 426.1209. TLC (99:1 DCM:MeOH), Rf: 0.1 (UV). Compound S13
Compound S13 was synthesized according to general procedure 6 on a 2.01 mmol scale. After column chromatography on silica (DCM:MeOH = 97:3), the target compound was isolated as a yellow oil (759 mg, 1.84 mmol, 92%). 1H NMR (400 MHz, CD3CN): δ 7.57 (s, 1H), 6.14 (bs, 2H), 4.37 (dd, J = 10.7, 5.5 Hz, 1H), 3.99 (dd, J = 15.0, 10.7 Hz, 1H), 3.68 (dd, J = 15.0, 5.6 Hz, 1H), 3.52 (q, J = 6.0 Hz, 2H), 3.40 (dt, J = 13.3, 7.6 Hz, 1H), 3.06 (ddd, J = 13.4, 7.6, 5.7 Hz, 1H), 2.69–2.47 (m, 5H), 1.76–1.61 (m, 2H), 1.52 (ddt, J = 13.0, 8.6, 6.5 Hz, 2H), 1.05 (t, J = 7.1 Hz, 6H). 13C{1H} NMR (101 MHz, CD3CN): δ 148.5, 147.4, 131.2, 129.3, 128.3, 61.9, 53.3, 49.0, 47.4, 45.3, 30.8, 30.3, 26.0, 15.2. FTIR (NaCl, thin film): 3734, 3220, 2356, 1354, 1522, 1110, 739 cm-1. HRMS: (ESI-TOF) calc’d for C14H26N3O5S3 [M+H]+ 412.1029, found 412.0804. TLC (3:7 hexanes:EtOAc), Rf: 0.15 (UV). Compound S14
Compound S14 was synthesized according to general procedure 7 on a 1.83 mmol scale. Since the compound is not stable on silica, the crude reaction mixture was directly used for the next step after filtration through a cotton plug and evaporation.
Compound 10
Compound 10 was synthesized according to general procedure 8 on a 117 µmol scale. After purification by preparative HPLC (C18 column, 9.4 x 250 mm), the target compound was isolated as a colorless oil (22.2 mg, 42.5 µmol, 36%). 1H NMR (400 MHz, CD3CN): δ 7.58 (s, 1H), 6.13 (bs, 2H), 4.38 (dd, J = 10.7, 5.6 Hz, 1H), 4.02 (ddd, J = 15.0, 10.7, 0.9 Hz, 1H), 3.69 (dd, J = 15.0, 5.5 Hz, 1H), 3.55–3.42 (m, 1H), 3.13 (ddd, J = 13.7, 7.2, 6.1 Hz, 1H), 2.83–2.66 (m, 6H), 2.57 (qd, J = 7.1, 1.5 Hz, 4H), 2.08–1.98 (m, 2H), 1.04 (t, J = 7.1 Hz, 6H). 13C{1H} NMR (101 MHz, CD3CN): δ 171.0, 169.7, 148.6, 147.4, 138.7, 131.2, 53.3, 48.12, 48.08, 45.3, 28.4, 26.3, 25.0, 15.2. HRMS: (ESI-TOF) calc’d for C18H27N4O8S3 [M+H]+ 523.0986, found 523.1107. BZA-4C-NHS (11) Compound S15
Compound S15 was synthesized according to general procedure 1 on a 9.50 mmol scale, using 1 bromo 5 methoxypentane (140 mL 206 g 114 mmol 12 equiv) After column
chromatography on silica (hex:EtOAc = 4:1), the target compound was isolated as a colorless oil (2.47 g, 7.26 mmol, 76%). 1H NMR (500 MHz, CDCl3): δ 6.96 (s, 1H), 4.72 (d, J = 5.1 Hz, 1H), 4.02 (dd, J = 15.3, 4.4 Hz, 1H), 3.82 (dd, J = 15.3, 4.9 Hz, 1H), 3.46–3.35 (m, 3H), 3.35–3.25 (m, 4H), 2.50 (bs, 1H), 1.70 (p, J = 7.2 Hz, 2H), 1.65–1.56 (m, 2H), 1.50–1.39 (m, 2H). 13C{1H} NMR (125 MHz, CDCl3): δ 142.9, 136.4, 133.0, 125.8, 72.6, 61.8, 58.7, 53.0, 49.4, 29.1, 28.4, 23.3. FTIR (NaCl, thin film): 3375, 2929, 2358, 1424, 1335, 1120 cm-1. HRMS: (ESI-TOF) calc’d for C12H19ClNO4S2 [M+H]+ 340.0439, found 340.0459. TLC (7:3 hexanes:EtOAc), Rf: 0.66 (UV). Compound S16
Compound S16 was synthesized according to general procedure 2 on a 7.24 mmol scale. After column chromatography on silica (hex:EtOAc = 4:1 to 7:3), the target compound was isolated as a colorless oil (2.84 g, 5.76 mmol, 80%). 1H NMR (500 MHz, CDCl3): δ 7.89–7.72 (m, 2H), 7.52–7.33 (m, 2H), 6.57 (s, 1H), 5.32 (dd, J = 3.9, 2.4 Hz, 1H), 4.22 (ddd, J = 16.4, 3.9, 0.8 Hz, 1H), 3.90 (dd, J = 16.4, 2.5 Hz, 1H), 3.46 (dt, J = 13.4, 7.6 Hz, 1H), 3.37 (t, J = 6.4 Hz, 2H), 3.34 (s, 3H), 3.12 (ddd, J = 13.4, 7.3, 6.1 Hz, 1H), 2.51 (s, 3H), 1.68–1.49 (m, 4H), 1.46–1.31 (m, 2H). 13C{1H} NMR (125 MHz, CDCl3): δ 146.3, 136.40, 136.39, 135.6, 132.9, 130.5, 128.0, 126.1, 72.6, 67.9, 58.7, 50.5, 49.8, 29.2, 27.9, 23.2, 21.9. FTIR (NaCl, thin film): 2924, 2359, 1345, 1168, 823, 737, 668 cm-1. HRMS: (ESI-TOF) calc’d for C19H28ClN2O6S3 [M+NH4]+ 511.0793, found 511.0804. TLC (7:3 hexanes:EtOAc), Rf: 0.4 (UV).
Compound S17
Compound S17 was synthesized according to general procedure 3 on a 5.75 mmol scale. After column chromatography on silica (hex:EtOAc = 85:15), the target compound was isolated as a colorless oil (1.05 g, 2.67 mmol, 46%). 1H NMR (500 MHz, CDCl3): δ 6.95 (s, 1H), 4.17 (dd, J = 10.8, 5.1 Hz, 1H), 4.11–4.00 (m, 1H), 3.56–3.43 (m, 2H), 3.38 (t, J = 6.4 Hz, 2H), 3.33 (s, 3H), 2.94 (dt, J = 13.5, 6.7 Hz, 1H), 2.6 –2.42 (m, 4H), 1.77–1.54 (m, 4H), 1.52–1.37 (m, 2H), 1.06 (t, J = 7.1 Hz, 6H). 13C{1H} NMR (125 MHz, CDCl3): δ 145.3, 135.5, 133.0, 126.2, 72.6, 58.6, 52.1, 48.2, 46.5, 44.7, 29.3, 28.7, 23.4, 15.0. FTIR (NaCl, thin film): 2927, 2358, 1338, 1163, 1115, 739, 668 cm-1. HRMS: (ESI-TOF) calc’d for C16H28ClN2O3S2 [M+H]+ 395.1224, found 395.1268. TLC (81:15 hexanes:EtOAc), Rf: 0.5 (UV). Compound S18
Compound S18 was synthesized according to general procedure 4 on a 2.66 mmol scale. After column chromatography on silica (hex:EtOAc = 85:15), the target compound was isolated as a yellow oil (813 mg, 1.69 mmol, 63%). 1H NMR (500 MHz, CDCl3): δ 7.34–7.16 (m, 5H), 6.86 (s, 1H), 4.12 (dd, J = 10.8, 5.3 Hz, 1H), 4.05 (s, 2H), 4.03–3.95 (m, 1H), 3.53–3.41 (m, 2H), 3.38 (t, J = 6.4 Hz, 2H), 3.33 (s, 3H), 2.90 (dt, J = 13.5, 6.8 Hz, 1H), 2.49 (qd, J = 7.0, 3.6 Hz, 4H), 1.73–1.54 (m, 4H), 1.51–1.38 (m, 2H), 1.01 (t, J = 7.1 Hz, 6H). 13C{1H} NMR (125 MHz, CDCl3): δ 145.3, 140.1, 136.7, 136.4, 132.1, 129.1, 128.8, 127.8, 72.6, 58.7, 52.2, 48.1, 46.9, 44.8, 43.2, 29.3, 28.7, 23.4, 15.1. FTIR (NaCl, thin film): 2928, 2359, 1339, 1166, 1117, 742 cm-1. HRMS: (ESI-TOF) calc’d for C23H35N2O3S3 [M+H]+ 483.1804, found 483.2046. TLC (3:7 hexanes:EtOAc), Rf: 0.65 (UV). Compound S19
Compound S19 was synthesized according to general procedure 5 on a 1.20 mmol scale. After column chromatography on silica (DCM:MeOH = 99:1), the target compound was isolated as a brown oil (347 mg, 0.79 mmol, 66%). 1H NMR (400 MHz, CDCl3): δ 7.63 (s, 1H), 5.32 (bs, 2H), 4.24 (dd, J = 10.8, 5.2 Hz, 1H), 4.11– 4.04 (m, 1H), 3.55 (dd, J = 14.8, 5.3 Hz, 1H), 3.47 (dt, J = 13.3, 7.6 Hz, 1H), 3.38 (t, J = 6.3 Hz, 2H), 3.32 (s, 3H), 2.96 (dt, J = 13.5, 6.7 Hz, 1H), 2.67–2.48 (m, 4H), 1.73–1.55 (m, 4H), 1.50– 1.37 (m, 2H), 1.06 (t, J = 7.1 Hz, 6H). 13C{1H} NMR (101 MHz, CDCl3): δ 146.6, 145.5, 139.7, 130.8, 72.6, 58.7, 52.7, 48.4, 46.7, 44.8, 29.2, 28.7, 23.4, 15.0. FTIR (NaCl, thin film): 3733, 3220, 2927, 2359, 1339, 1157, 1101 cm-1. HRMS: (ESI-TOF) calc’d for C16H30N3O5S3 [M+H]+ 440.1342, found 440.1344. TLC (99:1 DCM:MeOH), Rf: 01 (UV)
Compound S20
Compound S20 was synthesized according to general procedure 6 on a 735 µmol scale. After column chromatography on silica (DCM:MeOH = 97:3), the target compound was isolated as a yellow oil (240 mg, 563 µmol, 77%). 1H NMR (400 MHz, CD3CN): δ 7.57 (s, 1H), 6.12 (bs, 2H), 4.37 (dd, J = 10.7, 5.5 Hz, 1H), 3.99 (dd, J = 15.0, 10.7 Hz, 1H), 3.67 (dd, J = 15.0, 5.5 Hz, 1H), 3.53–3.46 (m, 2H), 3.39 (dt, J = 13.1, 7.7 Hz, 1H), 3.08–2.97 (m, 1H), 2.66–2.44 (m, 5H), 1.71–1.59 (m, 2H), 1.57–1.46 (m, 2H), 1.44– 1.32 (m, 2H), 1.05 (t, J = 7.1 Hz, 6H). 13C{1H} NMR (101 MHz, CD3CN): δ 148.1, 147.0, 130.8, 61.9, 52.9, 48.7, 47.0, 44.9, 32.6, 30.5, 28.8, 23.2, 14.9 (one signal disappearing underneath solvent signal). FTIR (NaCl, thin film): 3734, 2947, 2356, 2347, 1507, 1165, 826, 739, 668 cm-1. HRMS: (ESI-TOF) calc’d for C15H28N3O5S3 [M+H]+ 426.1186, found 425.1194. TLC (97:3 DCM:MeOH), Rf: 0.1 (UV). Compound S21
Compound S21 was synthesized according to general procedure 7 on a 557 µmol scale. Since the compound is not stable on silica, the crude reaction mixture was directly used for the next step after filtration through a cotton plug and evaporation.
Compound 11 was synthesized according to general procedure 8 on a 114 µmol scale. After purification by preparative HPLC (C18 column, 9.4 x 250 mm), the target compound was isolated as a colorless oil (6.20 mg, 11.6 µmol, 10%). 1H NMR (400 MHz, CD3CN): δ 7.58 (s, 1H), 6.13 (bs, 2H), 4.38 (dd, J = 10.7, 5.6 Hz, 1H), 3.99 (ddd, J = 15.0, 10.8, 1.0 Hz, 1H), 3.68 (dd, J = 15.0, 5.6 Hz, 1H), 3.47–3.35 (m, 1H), 3.08 (dt, J = 13.4, 5.9 Hz, 1H), 2.77 (s, 3H), 2.71–2.52 (m, 6H), 2.35–2.06 (m, 2H), 1.83–1.59 (m, 4H), 1.05 (t, J = 7.1 Hz, 6H). 13C{1H} NMR (101 MHz, CD3CN): δ 171.1, 169.9, 148.5, 147.3, 138.9, 131.2, 53.2, 48.5, 47.4, 45.3, 30.9, 28.2, 26.3, 22.4, 15.2. HRMS: (ESI-TOF) calc’d for C19H29N4O8S3 [M+H]+ 537.1142, found 537.1162. BZA-6C-NHS (12)
Compound S22
Compound S22 was synthesized according to general procedure 1 on a 8.50 mmol scale, using 1-bromo-7-methoxypentane (1.44 mL, 2.13 g, 10.2 mmol, 1.2 equiv.). After column chromatography on silica (hex:EtOAc = 4:1), the target compound was isolated as a colorless oil (2.44 g, 6.64 mmol, 78%). 1H NMR (500 MHz, CDCl3): δ 6.96 (s, 1H), 4.72 (bs, 1H), 4.01 (dd, J = 15.3, 4.4 Hz, 1H), 3.82 (dd, J = 15.3, 4.4 Hz, 1H), 3.37 (dd, J = 7.4, 5.7 Hz, 3H), 3.32 (s, 3H), 3.28 (dd, J = 13.6, 7.1 Hz, 1H), 2.52 (s, 1H), 1.67 (p, J = 7.2 Hz, 2H), 1.55 (q, J = 6.8, Hz, 2H), 1.36 (dd, J = 6.0, 2.6 Hz, 6H). 13C{1H} NMR (125 MHz, CDCl3): δ 142.9, 136.4, 133.0, 125.8, 72.9, 61.7, 58.7, 53.0, 49.5, 29.6, 29.0, 28.5, 26.5, 26.1. FTIR (NaCl, thin film): 3362, 2928, 2361, 1422, 1334, 1169 cm-1. HRMS: (ESI-TOF) calc’d for C14H22ClNO4S2 [M]+ 367.0679, found 367.0770. TLC (8:2 hexanes:EtOAc), Rf: 0.5 (UV). Compound S23
Compound S23 was synthesized according to general procedure 2 on a 6.63 mmol scale. After column chromatography on silica (hex:EtOAc = 4:1 to 7:3), the target compound was isolated as a colorless oil (3.17 g, 6.63 mmol, 91%).
1H NMR (500 MHz, CDCl3): δ 7.88–7.78 (m, 2H), 7.47–7.39 (m, 2H), 6.57 (s, 1H), 5.32 (dd, J = 3.9, 2.4 Hz, 1H), 4.29–4.17 (m, 1H), 3.90 (dd, J = 16.4, 2.5 Hz, 1H), 3.44 (dt, J = 13.3, 7.6 Hz, 1H), 3.37 (t, J = 6.6 Hz, 2H), 3.34 (s, 3H), 3.10 (ddd, J = 13.4, 7.5, 6.1 Hz, 1H), 2.51 (s, 3H), 1.67– 1.51 (m, 4H), 1.39–1.28 (m, 6H). 13C{1H} NMR (125 MHz, CDCl3): δ 146.3, 136.39, 136.36, 135.6, 133.0, 130.5, 128.0, 126.1, 72.9, 67.9, 58.7, 50.4, 49.7, 29.7, 29.1, 28.0, 26.5, 26.2, 21.9. FTIR (NaCl, thin film): 2920, 2358, 1356, 1169 cm-1. HRMS: (ESI-TOF) calc’d for C21H32ClN2O6S3 [M+NH4]+ 539.1106, found 539.1121. TLC (7:3 hexanes:EtOAc), Rf: 0.83 (UV). Compound S24
Compound S24 was synthesized according to general procedure 3 on a 6.05 mmol scale. After column chromatography on silica (hex:EtOAc = 85:15), the target compound was isolated as a colorless oil (1.27 g, 3.00 mmol, 50%). 1H NMR (500 MHz, CDCl3): δ 6.95 (s, 1H), 4.17 (dd, J = 10.8, 5.1 Hz, 1H), 4.11–4.02 (m, 1H), 3.57–3.41 (m, 2H), 3.36 (t, J = 6.5 Hz, 2H), 3.33 (s, 3H), 2.92 (dt, J = 13.4, 6.8 Hz, 1H), 2.67– 2.46 (m, 4H), 1.69–1.49 (m, 5H), 1.46–1.30 (m, 6H), 1.06 (t, J = 7.1 Hz, 6H). 13C{1H} NMR (125 MHz, CDCl3): δ 145.3, 135.5, 133.1, 126.2, 72.9, 58.7, 52.1, 48.2, 46.3, 44.7, 29.7, 29.2, 28.7, 26.6, 26.2, 15.0. FTIR (NaCl, thin film): 2930, 2857, 1416, 1343, 1168, 1117 cm-1. HRMS: (ESI-TOF) calc’d for C18H31ClN2O3S2 [M+H]+ 423.1537, found 423.1776. TLC (85:15 hexanes:EtOAc), Rf: 0.6 (UV).
Compound S25
Compound S25 was synthesized according to general procedure 4 on a 2.98 mmol scale. After column chromatography on silica (hex:EtOAc = 85:15), the target compound was isolated as a yellow oil (1.12 g, 2.19 mmol, 74%). 1H NMR (500 MHz, CDCl3): 7.31–7.20 (m, 5H), 6.86 (s, 1H), 4.12 (dd, J = 10.8, 5.2 Hz, 1H), 4.04 (s, 2H), 4.02–3.94 (m, 1H), 3.45 (ddd, J = 22.8, 14.9, 6.5 Hz, 2H), 3.36 (t, J = 6.6 Hz, 2H), 3.33 (s, 3H), 2.89 (dt, J = 13.4, 6.7 Hz, 1H), 2.49 (qd, J = 6.7, 6.2, 3.1 Hz, 4H), 1.71–1.50 (m, 4H), 1.45–1.31 (m, 6H), 1.01 (t, J = 7.1 Hz, 5H). 13C{1H} NMR (125 MHz, CDCl3): δ 145.3, 140.1, 136.7, 136.4, 132.1, 129.1, 128.8, 127.8, 72.9, 58.7, 52.2, 48.1, 46.8, 44.8, 43.2, 29.7, 29.2, 28.7, 26.6, 26.2, 15.1. FTIR (NaCl, thin film): 1929, 1857, 1357, 1456, 1339, 1160, 1111, 732 cm-1. HRMS: (ESI-TOF) calc’d for C25H39N2O3S3 [M+H]+ 511.2117, found 511.2327. TLC (85:15 hexanes:EtOAc), Rf: 0.74 (UV). Compound S26
Compound S26 was synthesized according to general procedure 5 on a 2.17 mmol scale. After column chromatography on silica (DCM:MeOH = 99:1), the target compound was isolated as a brown oil (139 mg, 0.30 mmol, 14%). 1H NMR (500 MHz, CDCl3): δ 7.63 (s, 1H), 5.32 (bs, 2H), 4.24 (dd, J = 10.8, 5.2 Hz, 1H), 4.11– 4.04 (m, 1H), 3.55 (dd, J = 14.8, 5.3 Hz, 1H), 3.47 (dt, J = 13.3, 7.6 Hz, 1H), 3.38 (t, J = 6.3 Hz, 2H), 3.32 (s, 3H), 3.02 (dt, J = 13.5, 6.7 Hz, 1H), 2.71–2.47 (m, 4H), 1.71–1.61 (m, 2H), 1.59– 1.50 (m, 2H), 1.44–1.31 (m, 6H), 1.06 (t, J = 7.1 Hz, 6H). 13C{1H} NMR (101 MHz, CDCl3): δ 146.6, 145.5, 139.8, 130.8, 72.9, 58.7, 52.8, 48.4, 46.5, 44.8, 29.6, 29.1, 28.8, 26.6, 26.1, 15.0. FTIR (NaCl, thin film): 3296, 2854, 2360, 1454, 1346, 1170, 1161, 1101, 738 cm-1. HRMS: (ESI-TOF) calc’d for C18H34N3O5S3 [M+H]+ 468.1655, found 468.1665. TLC (99:1 DCM:MeOH), Rf: 0.13 (UV). Compound S27
Compound S27 was synthesized according to general procedure 6 on a 14.5 mmol scale. After column chromatography on silica (DCM:MeOH = 97:3), the target compound was isolated as a yellow oil (147 mg, 2.42 mmol, 13%). 1H NMR (400 MHz, acetone-d6): δ 7.60 (s, 1H), 7.22 (bs, 1H), 4.52 (dd, J = 10.8, 5.6 Hz, 1H), 4.05 (dd, J = 14.9, 10.8 Hz, 1H), 3.79 (dd, J = 14.9, 5.6 Hz, 1H), 3.52 (t, J = 6.5 Hz, 3H), 3.49– 3.31 (m, 2H), 3.15 (ddd, J = 13.4, 7.4, 5.9 Hz, 1H), 2.62 (qd, J = 6.9, 3.1 Hz, 4H), 1.79–1.60 (m, 3H), 1.57–1.45 (m, 2H), 1.43–1.33 (m, 7H), 1.07 (t, J = 7.1 Hz, 6H). 13C{1H} NMR (101 MHz, acetone-d6): δ 149.4, 146.9, 139.2, 130.7, 62.5, 53.4, 49.0, 47.4, 45.4, 33.8, 27.4, 26.7, 15.4. FTIR (NaCl, thin film): 3367, 2934, 2361, 29359, 1269, 1110, 778, 740, 668 cm-1. HRMS: (ESI-TOF) calc’d for C17H32N3O5S3 [M+H]+ 454.1499, found 454.1537. TLC (3:7 hexanes:EtOAc), Rf: 077 (UV)
Compound S28
Compound S28 was synthesized according to general procedure 7 on a 310 µmol scale. After purification by preparative HPLC (C18 column, 9.4 x 250 mm), the target compound was isolated as a colorless oil (107 mg, 229 µmol, 74%). 1H NMR (400 MHz, acetone-d6): δ 7.60 (s, 1H), 7.21 (bs, 2H), 4.52 (dd, J = 10.7, 5.5 Hz, 1H), 4.05 (dd, J = 14.9, 10.7 Hz, 1H), 3.80 (dd, J = 14.9, 5.5 Hz, 1H), 3.43 (dt, J = 13.4, 7.6 Hz, 1H), 3.15 (ddd, J = 13.5, 7.7, 5.8 Hz, 1H), 2.62 (qd, J = 6.8, 3.4 Hz, 4H), 2.29 (t, J = 7.4 Hz, 2H), 1.79– 1.54 (m, 4H), 1.47–1.33 (m, 4H), 1.06 (t, J = 7.1 Hz, 6H). 13C{1H} NMR (101 MHz, acetone-d6): δ 174.7, 149.3, 146.9, 139.1, 130.7, 53.4, 49.0, 47.4, 45.3, 34.1, 29.41, 29.39, 27.0, 25.6, 15.4. HRMS: (ESI-TOF) calc’d for C17H30N3O6S3 [M+H]+ 468.1291, found 468.1299. Compound 12
Compound 12 was synthesized according to general procedure 8 on a 45.3 µmol scale. After purification by preparative HPLC (C18 column, 9.4 x 250 mm), the target compound was isolated as a colorless oil (7.60 mg, 13.5 µmol, 30%). 1H NMR (400 MHz, CD3CN): δ 7.57 (s, 1H), 6.12 (bs, 2H), 4.37 (dd, J = 10.7, 5.6 Hz, 1H), 3.98 (ddd, J = 14.5, 10.8, 3.7 Hz, 1H), 3.72–3.62 (m, 1H), 3.43–3.28 (m, 1H), 3.03 (ddd, J = 13.6, 7.7, 5.9 Hz, 1H), 2.76 (s, 4H), 2.59 (dt, J = 20.2, 7.1 Hz, 6H), 1.78–1.55 (m, 4H), 1.49–1.32 (m, 4H), 1.05 (t, J = 7.0 Hz, 6H). 13C{1H} NMR (101 MHz, CD3CN): δ 171.1, 170.1, 148.5, 147.3, 139.0, 131.2, 53.3, 49.0, 47.5, 45.3, 31.3, 29.1, 28.8, 26.6, 26.3, 25.2, 15.2. HRMS: (ESI-TOF) calc’d for C21H33N4O8S3 [M+H]+ 565.1455, found 565.1451. BZA-8C-NHS (13) Compound S29
Compound S29 was synthesized according to general procedure 1 on a 10.0 mmol scale, using 1-bromo-9-methoxynonane (1.92 mL, 2.85 g, 12.0 mmol, 1.2 equiv.). After column chromatography on silica (hex:EtOAc = 4:1), the target compound was isolated as a colorless oil (4.20 g, 10.6 mmol, quant.). 1H NMR (400 MHz, CDCl3): δ 6.95 (s, 1H), 3.99 (dd, J = 15.4, 4.4 Hz, 1H), 3.82 (dd, J = 15.3, 5.2 Hz, 1H), 3.53–3.22 (m, 7H), 2.72–2.58 (m, 1H), 1.72–1.60 (m, 2H), 1.60–1.49 (m, 2H), 1.37– 1.27 (m, 10H). 13C{1H} NMR (101 MHz, CDCl3): δ 143.1, 136.3, 132.9, 125.8, 73.1, 61.7, 58.7, 52.9, 49.5, 29.7, 29.5, 29.4, 29.1, 28.6, 26.5, 26.2. FTIR (NaCl, thin film): 3422, 2927, 2855, 2357, 1339, 1154 cm-1. HRMS: (ESI-TOF) calc’d for C16H27ClNO4S2 [M+H]+ 396.1065, found 396.1139. TLC (8:2 hexanes:EtOAc), Rf: 0.53 (UV).
Compound S30
Compound S30 was synthesized according to general procedure 2 on a 10.0 mmol scale. After column chromatography on silica (hex:EtOAc = 3:1), the target compound was isolated as a colorless oil (5.44 g, 9.90 mmol, 99%). 1H NMR (500 MHz, CDCl3): δ 7.89–7.78 (m, 2H), 7.46–7.40 (m, 2H), 6.57 (s, 1H), 4.25–4.16 (m, 1H), 3.90 (dd, J = 16.3, 2.5 Hz, 1H), 3.47–3.35 (m, 3H), 3.34 (s, 3H), 3.10 (ddd, J = 13.4, 7.5, 6.1 Hz, 1H), 2.51 (s, 3H), 1.66–1.49 (m, 4H), 1.41–1.24 (m, 10H). 13C{1H} NMR (125 MHz, CDCl3): δ 146.2, 136.4, 136.4, 135.6, 133.0, 130.5, 128.0, 126.1, 73.0, 67.9, 58.7, 50.3, 49.8, 29.8, 29.6, 29.5, 29.2, 28.0, 26.5, 26.3, 21.9. FTIR (NaCl, thin film): 2926, 2854, 2359, 1716, 1454, 1360, 1166, 940, 732 cm-1. HRMS: (ESI-TOF) calc’d for C23H36ClN2O6S3 [M+NH4]+ 567.1419, found 567.1434. TLC (7:3 hexanes:EtOAc), Rf: 0.92 (UV). Compound S31
Compound S31 was synthesized according to general procedure 3 on a 9.89 mmol scale. After column chromatography on silica (hex:EtOAc = 85:15), the target compound was isolated as a colorless oil (2.15 g, 4.77 mmol, 48%). 1H NMR (500 MHz, CDCl3): δ 6.95 (s, 1H), 4.17 (dd, J = 10.8, 5.1 Hz, 1H), 4.06 (ddd, J = 14.7, 10.8, 0.9 Hz, 1H), 3.53–3.42 (m, 2H), 3.36 (t, J = 6.6 Hz, 2H), 3.33 (s, 3H), 2.92 (dt, J = 13.5, 6.8 Hz, 1H), 2.67–2.48 (m, 4H), 1.70–1.51 (m, 4H), 1.42–1.26 (m, 10H), 1.06 (t, J = 7.1 Hz, 6H). 13C{1H} NMR (125 MHz, CDCl3): δ 145.2, 135.3, 133.0, 126.1, 72.9, 58.6, 51.9, 48.1, 46.2, 44.6, 29.6, 29.4, 29.4, 29.1, 28.6, 26.5, 26.1, 14.9. FTIR (NaCl, thin film): 2924, 2853, 2358, 1539, 1455, 1348, 1166, 749 cm-1. HRMS: (ESI-TOF) calc’d for C20H36ClN2O3S2 [M+H]+ 451.1850, found 451.1867. TLC (85:15 hexanes:EtOAc), Rf: 0.66 (UV). Compound S32
Compound S32 was synthesized according to general procedure 4 on a 4.77 mmol scale. After column chromatography on silica (hex:EtOAc = 85:15), the target compound was isolated as a yellow oil (1.34 g, 2.49 mmol, 52%). 1H NMR (400 MHz, CDCl3): δ 7.34–7.17 (m, 5H), 6.86 (s, 1H), 4.12 (dd, J = 10.8, 5.1 Hz, 1H), 4.04 (s, 2H), 3.98 (dd, J = 14.7, 10.8 Hz, 1H), 3.52–3.40 (m, 2H), 3.36 (t, J = 6.6 Hz, 2H), 3.33 (s, 3H), 2.88 (dt, J = 13.5, 6.8 Hz, 1H), 2.49 (qd, J = 7.0, 2.0 Hz, 4H), 1.58 (dq, J = 20.8, 6.9 Hz, 4H), 1.39–1.25 (m, 10H), 1.00 (t, J = 7.1 Hz, 6H). 13C{1H} NMR (101 MHz, CDCl3): δ 145.3, 140.0, 136.7, 136.5, 132.1, 129.1, 128.8, 127.8, 73.1, 58.7, 52.3, 48.1, 46.8, 44.8, 43.2, 29.8, 29.6, 29.5, 29.3, 28.8, 26.7, 26.2. FTIR (NaCl, thin film): 2925, 2853, 2359, 1540, 1450, 1362, 1169, 770 cm-1. HRMS: (ESI-TOF) calc’d for C27H43N2O3S3 [M+H]+ 539.2430, found 539.2675. TLC (85:15 hexanes:EtOAc), Rf: 0.83 (UV).
Compound S33
Compound S33 was synthesized according to general procedure 5 on a 2.49 mmol scale. After column chromatography on silica (hex:EtOAc = 3:2), the target compound was isolated as a brown oil (833 mg, 1.68 mmol, 68%). 1H NMR (500 MHz, CDCl3): δ 7.63 (s, 1H), 5.40 (bs, 2H), 4.24 (dd, J = 10.8, 5.2 Hz, 1H), 4.12– 4.03 (m, 1H), 3.55 (dd, J = 14.9, 5.3 Hz, 1H), 3.45 (dt, J = 13.5, 7.6 Hz, 1H), 3.37 (t, J = 6.6 Hz, 2H), 3.33 (s, 3H), 2.96 (dt, J = 13.5, 6.8 Hz, 1H), 2.69–2.48 (m, 4H), 1.70–1.60 (m, 2H), 1.59– 1.51 (m, 2H), 1.41–1.27 (m, 10H), 1.07 (t, J = 7.1 Hz, 6H). 13C{1H} NMR (125 MHz, CDCl3): δ 146.6, 145.4, 139.8, 130.8, 73.1, 58.7, 52.7, 48.4, 46.5, 44.8, 29.7, 29.5, 29.5, 29.1, 28.7, 26.6, 26.2, 15.0. FTIR (NaCl, thin film): 3334, 2928, 2357, 1540, 1352, 1161, 780 cm-1. HRMS: (ESI-TOF) calc’d for C20H38N3O5S3 [M+H]+ 496.1968, found 495.2363. TLC (6:4 hexanes:EtOAc), Rf: 0.16(UV). Compound S34
Compound S34 was synthesized according to general procedure 6 on a 1.67 mmol scale. After column chromatography on silica (DCM:MeOH = 97:3), the target compound was isolated as a yellow oil (344 mg, 0.71 mmol, 43%). 1H NMR (400 MHz, acetone-d6): δ 7.60 (s, 1H), 7.22 (bs, 2H), 4.51 (dd, J = 10.7, 5.6 Hz, 1H), 4.05 (ddd, J = 14.9, 10.7, 1.0 Hz, 1H), 3.79 (dd, J = 15.0, 5.6 Hz, 1H), 3.52 (dt, J = 9.2, 4.4 Hz, 2H), 3.48–3.35 (m, 2H), 3.14 (ddd, J = 13.5, 7.5, 5.9 Hz, 1H), 2.62 (qd, J = 7.1, 3.1 Hz, 4H), 1.72– 1.64 (m, 2H), 1.54–1.45 (m, 2H), 1.41–1.26 (m, 10H), 1.06 (t, J = 7.1 Hz, 6H). 13C{1H} NMR (101 MHz, acetone-d6): δ 149.3, 146.8, 139.1, 130.6, 62.5, 53.3, 48.9, 47.3, 45.3, 33.8, 30.3, 30.2, 27.2, 26.7, 15.3. FTIR (NaCl, thin film): 3337, 2930, 2854, 1453, 1344, 1170, 1157, 681 cm-1. HRMS: (ESI-TOF) calc’d for C19H36N3O5S3 [M+H]+ 482.1812, found 482.1766. TLC (97:3 DCM:MeOH), Rf: 0.18 (UV). Compound S35
Compound S35 was synthesized according to general procedure 7 on a 688 µmol scale. Since the compound is not stable on silica, the crude reaction mixture was directly used for the next step after filtration through a cotton plug and evaporation.
Compound 13
Compound 13 was synthesized according to general procedure 8 on a 101 µmol scale. After purification by preparative HPLC (C18 column, 9.4 x 250 mm), the target compound was isolated as a colorless oil (25.2 mg, 42.5 µmol, 42%). 1H NMR (400 MHz, CD3CN): δ 7.57 (s, 1H), 6.12 (bs, 2H), 4.36 (dd, J = 10.7, 5.5 Hz, 1H), 3.98 (dd, J = 15.0, 10.7 Hz, 1H), 3.66 (dd, J = 15.0, 5.5 Hz, 1H), 3.37 (dt, J = 13.5, 7.7 Hz, 1H), 3.02 (ddd, J = 13.4, 7.4, 5.9 Hz, 1H), 2.76 (s, 4H), 2.68–2.51 (m, 6H), 1.75–1.55 (m, 4H), 1.46–1.28 (m, 8H), 1.04 (t, J = 7.1 Hz, 6H). 13C{1H} NMR (101 MHz, CD3CN): δ 171.1, 170.1, 148.4, 147.3, 139.0, 131.2, 53.2, 49.0, 47.4, 45.3, 31.4, 29.5, 29.5, 29.2, 29.2, 27.0, 26.3, 25.3, 15.2. HRMS: (ESI-TOF) calc’d for C23H37N4O8S3 [M+H]+ 593.1768, found 593.1794. BZA-PEG-PFP (14) Compound S36 and Compound S37
In a 250 mL round-bottom flask equipped with a stir bar, tert-butyl 3-(2-(2- hydroxyethoxy)ethoxy)propanoate (S36) (5.00 g, 21.3 mmol, 1.0 equiv.) was dissolved in DCM (61.0 mL). Carbon tetrabromide (8.85 g, 26.7 mmol, 1.25 equiv.) and triphenylphosphine (7.28 g, 27.7 mmol, 1.3 equiv.) were slowly added (exothermic reaction). The reaction mixture was stirred at room temperature for 2 hours, before the volatile materials were evaporated. The
residue was purified by column chromatography on silica (hex:EtOAc = 9:1) to afford alkyl bromide S37 (3.69 g, 12.4 mmol, 58%) as a colorless oil. 1H NMR (400 MHz, CDCl3): δ 3.80 (t, J = 6.3 Hz, 2H), 3.72 (t, J = 6.5 Hz, 2H), 3.68–3.58 (m, 4H), 3.46 (t, J = 6.4 Hz, 2H), 2.50 (t, J = 6.5 Hz, 2H), 1.44 (s, 9H). 13C{1H} NMR (101 MHz, CDCl3): δ 171.0, 80.7, 71.4, 70.6, 70.5, 67.1, 36.4, 30.4, 28.2. FTIR (NaCl, thin film): 2977, 2928, 2872, 1731, 1457, 1392, 1366, 1255, 1156, 848 cm-1. HRMS: (ESI-TOF) calc’d for C11H21BrNaO4 [M+Na]+ 319.0515, found 319.0513. TLC (4:1 hexanes:EtOAc), Rf: 0.40 (KMnO4). Compound S38
Compound S38 was synthesized according to general procedure 1 on a 10.0 mmol scale, using S37 (3.57 g, 12.0 mmol, 1.2 equiv.). After column chromatography on silica (hex:EtOAc = 3:2 to 2:3), the target compound was isolated as a colorless oil (4.78 g, 10.5 mmol, quant.). 1H NMR (400 MHz, CDCl3): δ 6.99 (s, 1H), 4.72 (q, J = 4.4 Hz, 1H), 4.34 (d, J = 5.9 Hz, 1H), 4.22 (dd, J = 15.5, 4.3 Hz, 1H), 4.07 (dd, J = 15.5, 3.9 Hz, 1H), 3.81–3.73 (m, 3H), 3.70 (t, J = 6.2 Hz, 2H), 3.64–3.40 (m, 5H), 2.50 (t, J = 6.2 Hz, 2H), 1.45 (s, 9H). 13C{1H} NMR (101 MHz, CDCl3): δ 171.5, 144.1, 135.7, 132.7, 126.5, 81.2, 71.0, 70.3, 70.0, 61.7, 54.2, 48.8, 36.4, 31.1, 28.2. FTIR (NaCl, thin film): 3445, 2977, 2875, 1725, 1423, 1367, 1341, 1167, 1027, 845 cm-1. HRMS: (ESI-TOF) calc’d for C17H26ClNNaO7S2 [M+Na]+ 478.0731, found 478.0758. TLC (2:3 hexanes:EtOAc), Rf: 0.46 (UV).
Compound S39
e 2 on a 10.0 mmol scale. After column chromatography on silica (hex:EtOAc = 3:1), the target compound was isolated as a colorless oil (6.05 g, 9.92 mmol, 99%). 1H NMR (400 MHz, CDCl3): δ 7.91–7.77 (m, 2H), 7.45–7.37 (m, 2H), 6.60 (s, 1H), 5.40 (t, J = 3.7 Hz, 1H), 4.26 (dd, J = 16.2, 4.1 Hz, 1H), 4.18–4.11 (m, 1H), 3.76–3.56 (m, 9H), 3.44–3.31 (m, 1H), 2.56–2.43 (m, 5H), 1.44 (s, 9H). 13C{1H} NMR (101 MHz, CDCl3): δ 171.0, 146.1, 137.0, 136.3, 135.8, 133.1, 130.4, 128.1, 126.2, 80.7, 70.8, 70.7, 70.4, 68.1, 67.1, 52.3, 49.8, 36.4, 28.2, 21.9. FTIR (NaCl, thin film): 3349, 3095, 2850, 2315, 1728, 1605, 1520, 1415, 1335, 1169 cm-1. HRMS: (ESI-TOF) calc’d for C24H33ClNO9S3 [M+H]+ 610.1000, found 610.1057. TLC (3:1 hexanes:EtOAc), Rf: 0.11 (UV). Compound S40
Compound S40 was synthesized according to general procedure 3 on a 9.92 mmol scale. After column chromatography on silica (hex:EtOAc = 75:25), the target compound was isolated as a colorless oil (2.51 g, 4.92 mmol, 50%).
1H NMR (400 MHz, CDCl3): δ 6.96 (s, 1H), 4.26 (dd, J = 10.7, 5.1 Hz, 1H), 4.11 (dd, J = 14.6, 10.8 Hz, 1H), 3.78 (dd, J = 14.7, 5.2 Hz, 1H), 3.73–3.66 (m, 4H), 3.66–3.54 (m, 5H), 3.22 (ddd, J = 14.3, 6.7, 4.8 Hz, 1H), 2.65–2.42 (m, 6H), 1.44 (s, 9H), 1.05 (t, J = 7.0 Hz, 6H). 13C{1H} NMR (101 MHz, CDCl3): δ 170.9, 146.0, 135.5, 132.9, 126.4, 80.7, 71.1, 70.7, 67.0, 52.2, 48.2, 47.9, 44.6, 36.4, 28.2, 15.0. FTIR (NaCl, thin film): 3096, 2969, 2873, 1737, 1731, 1454, 1416, 1338, 1160, 1005 cm-1. HRMS: (ESI-TOF) calc’d for C21H36ClN2O6S3 [M+H]+ 511.1698, found 511.1815. TLC (3:1 hexanes:EtOAc), Rf: 0.28 (UV). Compound S41
Compound S41 was synthesized according to general procedure 4 on a 4.91 mmol scale. After column chromatography on silica (hex:EtOAc = 85:15), the target compound was isolated as a yellow oil (1.78 g, 2.97 mmol, 60%). 1H NMR (400 MHz, CDCl3): 7.32–7.18 (m, 5H), 6.88 (s, 1H), 4.21 (dd, J = 10.7, 5.3 Hz, 1H), 4.10–3.96 (m, 3H), 3.79–3.66 (m, 5H), 3.64–3.53 (m, 5H), 3.19 (dt, J = 14.3, 5.5 Hz, 1H), 2.48 (dt, J = 12.2, 6.8 Hz, 6H), 1.44 (s, 9H), 0.99 (t, J = 7.1 Hz, 6H). 13C{1H} NMR (101 MHz, CDCl3): δ 170.9, 140.1, 136.7, 136.2, 132.3, 129.0, 128.8, 127.8, 80.7, 71.2, 70.7, 70.4, 67.0, 52.4, 48.3, 48.2, 44.6, 43.2, 36.4, 28.2, 15.0. FTIR (NaCl, thin film): 3061, 2970, 2925, 2869, 1730, 1453, 1364, 1136, 948, 848 cm-1. HRMS: (ESI-TOF) calc’d for C28H43N2O6S3 [M+H]+ 599.2278, found 599.2428. TLC (3:1 hexanes:EtOAc), Rf: 0.21 (UV).
Compound S42
In a 50 mL round-bottom flask equipped with a stir bar, tert-butyl ester S41 (1.16 g, 1.94 mmol, 1.0 equiv.) was dissolved in THF (9.69 mL). The flask was cooled to 0 °C and lithium aluminum hydride (110 mg, 2.91 mmol, 1.5 equiv.) was added portion wise. The ice bath was removed and the reaction was stirred for 4 hours at room temperature. After Fieser work-up, the crude material was purified by column chromatography on silica (hex:EtOAc = 2:3 to 100% EtOAc) to afford alcohol S42 (814 mg, 1.54 mmol, 79%) as a brown oil. 1H NMR (400 MHz, CDCl3): δ 7.33–7.18 (m, 5H), 6.88 (s, 1H), 4.20 (dd, J = 10.7, 5.3 Hz, 1H), 4.04 (s, 3H), 3.82–3.52 (m, 12H), 3.18 (dt, J = 14.4, 5.8 Hz, 1H), 2.48 (q, J = 7.1 Hz, 4H), 1.83 (p, J = 5.7 Hz, 2H), 1.00 (t, J = 7.0 Hz, 6H). 13C{1H} NMR (101 MHz, CDCl3): δ 145.9, 140.1, 136.7, 136.1, 132.3, 129.0, 128.8, 127.8, 71.2, 70.6, 70.5, 70.2, 61.9, 52.4, 48.5, 48.3, 44.6, 43.2, 32.0, 15.0. FTIR (NaCl, thin film): 3432, 3027, 2873, 2474, 1952, 1670, 1453, 1335, 1163, 1110 cm-1. HRMS: (ESI-TOF) calc’d for C24H37N2O5S3 [M+H]+ 529.1859, found 529.1980. TLC (1:4 hexanes:EtOAc), Rf: 0.36 (UV).
Compound S43
In a 2-dram vial equipped with a stir bar, alcohol S42 (264 mg, 0.50 mmol, 1.0 equiv.) was dissolved in DCM (1.0 mL). The solution was cooled to 0 °C and triethylamine (76.7 µL, 55.7 mg, 0.55 mmol, 1.1 equiv.) and acetyl chloride (35.6 µL, 39.2 mg, 0.50 mmol, 1.0 equiv.) were added. The ice bath was removed and the reaction was stirred for overnight at room temperature. Afterwards, water (2.0 mL) was added, and the aqueous phase was extracted with DCM (3x). The combined organic phases were dried over Na2SO4, filtered, and the volatile materials were evaporated under reduced pressure. The crude material was purified by column chromatography on silica (hex:EtOAc = 1:1) to afford acetate S43 (209 mg, 367 µmol, 73%) as a colorless oil. 1H NMR (400 MHz, CDCl3): δ 7.33–7.18 (m, 5H), 6.88 (s, 1H), 4.21 (dd, J = 10.7, 5.2 Hz, 1H), 4.15 (t, J = 6.5 Hz, 2H), 4.08–3.97 (m, 3H), 3.77–3.67 (m, 3H), 3.65–3.47 (m, 7H), 3.19 (dt, J = 14.3, 5.6 Hz, 1H), 2.54–2.39 (m, 4H), 2.04 (s, 3H), 1.90 (p, J = 6.4 Hz, 2H), 0.99 (t, J = 7.0 Hz, 6H). 13C{1H} NMR (101 MHz, CDCl3): δ 171.2, 145.9, 140.1, 136.7, 136.1, 132.2, 129.0, 128.8, 127.8, 71.1, 70.7, 70.3, 67.9, 61.8, 52.4, 48.3, 48.2, 44.6, 43.2, 29.1, 21.1, 15.0. FTIR (NaCl, thin film): 3449, 3029, 2967, 2468, 1737, 1437, 1341, 1245, 1140, 953, 829, 718 cm-1. HRMS: (ESI-TOF) calc’d for C26H39N2O6S3 [M+H]+ 571.1965, found 571.2076.
TLC (1:1 hexanes:EtOAc), Rf: 0.63 (UV). Compound S44
Compound S44 was synthesized according to general procedure 6 on a 345 µmol scale. It was observed that the acetate protecting group was hydrolyzed during the reaction. After column chromatography on silica (DCM:MeOH = 95:5), the free alcohol S44 was isolated as a colorless oil (44.1 mg, 90.8 µmol, 26%). 1H NMR (400 MHz, CD3CN): δ 7.58 (s, 1H), 6.28 (bs, 2H), 4.44 (dd, J = 10.7, 5.6 Hz, 1H), 4.03 (ddd, J = 14.9, 10.8, 1.0 Hz, 1H), 3.85 (dd, J = 14.9, 5.6 Hz, 1H), 3.69–3.62 (m, 2H), 3.60–3.46 (m, 9H), 3.28 (ddd, J = 14.4, 6.4, 4.4 Hz, 1H), 2.57 (qd, J = 7.0, 1.1 Hz, 4H), 1.71 (p, J = 6.2 Hz, 2H), 1.05 (t, J = 7.1 Hz, 6H). 13C{1H} NMR (101 MHz, CD3CN): δ 148.5, 147.6, 139.0, 131.2, 71.1, 70.7, 70.6, 69.3, 60.2, 53.4, 48.9, 48.2, 45.2, 33.4, 22.5, 15.2. TLC (19:1 DCM:MeOH), Rf: 0.25 (UV). Compound S45
Compound S45 was synthesized according to general procedure 7 on a 90.6 µmol scale. After purification by preparative HPLC (C18 column, 9.4 x 250 mm), the target compound was isolated as a colorless oil (26.7 mg, 53.4 µmol, 59%). 1H NMR (400 MHz, CD3CN): δ 7.58 (s, 1H), 6.15 (bs, 2H), 4.45 (dd, J = 10.6, 5.6 Hz, 1H), 4.04 (dd, J = 15.0, 10.6 Hz, 1H), 3.87 (dd, J = 15.0, 5.7 Hz, 1H), 3.72–3.62 (m, 4H), 3.59–3.48 (m, 5H), 3.25 (ddd, J = 14.5, 6.6, 4.3 Hz, 1H), 2.66–2.53 (m, 4H), 2.50 (t, J = 6.2 Hz, 2H), 1.05 (t, J = 7.1 Hz, 6H). 13C{1H} NMR (101 MHz, CD3CN): δ 173.3, 148.5, 147.2, 139.1, 131.2, 71.0, 70.8, 70.6, 67.3, 53.4, 48.9, 48.3, 45.3, 35.4, 15.0. HRMS: (ESI-TOF) calc’d for C17H30N3O8S3 [M+H]+ 500.1190, found 500.1209. Compound 14
In a 1-dram vial equipped with a stir bar, acid S45 (10.0 mg, 20.0 µmol, 1.0 equiv.) was dissolved in DCM:MeCN (9:1, 200 µL). To the solution were added N-(3- Dimethylaminopropyl)-N′-ethylcarbodiimide hydrochloride (4.20 mg, 22.0 µmol, 1.1 equiv.), pentafluorophenol (4.10 mg, 22.0 µmol, 1.1 equiv.), and DMAP (0.3 mg, 2.00 µmol, 0.1 equiv.). The reaction was stirred overnight at room temperature. Afterwards, the volatile materials were evaporated under reduced pressure. The crude material was purified by preparative HPLC (C18
column, 9.4 x 250 mm) to obtain the target compound 14 (7.9 mg, 11.9 µmol, 59%) as a colorless oil. 1H NMR (400 MHz, CD3CN): δ 7.57 (s, 1H), 6.16 (bs, 2H), 4.42 (dd, J = 10.7, 5.6 Hz, 1H), 4.02 (ddd, J = 15.0, 10.7, 1.0 Hz, 1H), 3.90–3.78 (m, 3H), 3.69–3.64 (m, 2H), 3.62–3.50 (m, 5H), 3.25 (ddd, J = 14.4, 6.4, 4.4 Hz, 1H), 2.94 (t, J = 6.0 Hz, 2H), 2.56 (q, J = 7.1 Hz, 4H), 1.04 (t, J = 7.1 Hz, 6H). 13C{1H} NMR (101 MHz, CD3CN): δ 168.9, 148.5, 147.5, 131.2, 71.0, 70.6, 66.7, 53.3, 48.80, 48.1, 45.2, 35.1, 15.1. Note: Phenol carbons not observed due to C–F coupling. 19F{1H} NMR (376 MHz, CD3CN): δ –154.67 (d, J = 16.6 Hz), –160.41 (t, J = 20.9 Hz), –164.58 (dd, J = 20.7, 16.8 Hz). HRMS: (ESI-TOF) calc’d for C23H29F5N3O8S3 [M+H]+ 666.1031, found 666.1042. BZA-2C-PFP (15) Compound S46
Compound S46 was synthesized according to general procedure 1 on a 10.0 mmol scale, using 1-bromo-3-methoxypropane (1.36 mL, 1.84 g, 12.0 mmol, 1.2 equiv.). After column chromatography on silica (hex:EtOAc = 4:1 to 7:3), the target compound was isolated as a colorless oil (3.25 g, 10.4 mmol, quant.). 1H NMR (400 MHz, CDCl3): δ 6.95 (s, 1H), 4.63 (dt, J = 8.3, 4.1 Hz, 1H), 4.13–3.98 (m, 1H), 3.76 (dd, J = 15.3, 4.2 Hz, 1H), 3.65 (ddd, J = 12.6, 7.8, 6.4 Hz, 1H), 3.51 (qdd, J = 9.6, 6.7, 4.5 Hz, 2H), 3.42–3.27 (m, 2H), 3.24 (s, 3H), 2.03–1.80 (m, 2H). 13C{1H} NMR (101 MHz, CDCl3): δ 143.1, 136.1, 133.2, 126.0, 70.2, 61.8, 58.7, 53.9, 47.1, 29.0. FTIR (NaCl, thin film): 3434, 2931, 2881, 2104, 1644, 1422, 1333, 1163, 1112, 1027, 684 cm-1. HRMS: (ESI-TOF) calc’d for C10H14ClNaNO4S2 [M+Na]+ 333.9945, found 333.9983. TLC (3:1 hexanes:EtOAc), Rf: 0.11 (UV).
Compound S47
Compound S47 was synthesized according to general procedure 2 on a 10.0 mmol scale. After column chromatography on silica (hex:EtOAc = 3:1), the target compound was isolated as a colorless oil (4.66 g, 10.0 mmol, quant.). 1H NMR (500 MHz, CDCl3): δ 7.90–7.76 (m, 2H), 7.47–7.40 (m, 2H), 6.61 (s, 1H), 5.34 (dd, J = 3.9, 2.6 Hz, 1H), 4.23 (ddd, J = 16.2, 3.9, 0.8 Hz, 1H), 3.92 (dd, J = 16.3, 2.6 Hz, 1H), 3.55 (dt, J = 13.8, 6.9 Hz, 1H), 3.47–3.38 (m, 2H), 3.32 (s, 3H), 3.26 (dt, J = 13.7, 6.8 Hz, 1H), 2.51 (s, 3H), 1.91–1.81 (m, 2H). 13C{1H} NMR (125 MHz, CDCl3): δ 146.2, 136.5, 136.4, 135.6, 133.0, 130.5, 128.1, 126.1, 69.2, 67.8, 58.8, 51.4, 47.5, 29.0, 21.9. FTIR (NaCl, thin film): 3439, 2111, 1659, 1645, 1423, 1346, 1190, 1175, 1116, 939 cm-1. HRMS: (ESI-TOF) calc’d for C17H21NClO6S3 [M+H]+ 466.0214, found 466.0268. TLC (3:1 hexanes:EtOAc), Rf: 0.38 (UV). Compound S48
Compound S48 was synthesized according to general procedure 3 on a 8.28 mmol scale. After column chromatography on silica (hex:EtOAc = 85:15), the target compound was isolated as a colorless oil (1.70 g, 4.63 mmol, 56%). 1H NMR (500 MHz, CDCl3): δ 6.96 (s, 1H), 4.20 (dd, J = 10.8, 5.1 Hz, 1H), 4.10 (ddd, J = 14.6, 10.8, 0.9 Hz, 1H), 3.62–3.48 (m, 3H), 3.43 (ddd, J = 9.5, 6.4, 5.3 Hz, 1H), 3.34 (s, 3H), 3.08 (dt, J = 139 70 Hz 1H) 268–245 (m 4H) 199–183 (m 2H) 106 (t J = 71 Hz 6H)
13C{1H} NMR (125 MHz, CDCl3): δ 145.5, 135.6, 132.9, 126.3, 69.4, 58.9, 52.2, 47.5, 45.9, 44.7, 29.7, 15.0. FTIR (NaCl, thin film): 3433, 2968, 2932, 2872, 2831, 2100, 1644, 1342, 1169, 1115 cm-1. HRMS: (ESI-TOF) calc’d for C14H24ClN2O3S2 [M+H]+ 367.0911, found 367.0940. TLC (3:1 hexanes:EtOAc), Rf: 0.43 (UV). Compound S49
Compound S49 was synthesized according to general procedure 4 on a 4.61 mmol scale. After column chromatography on silica (hex:EtOAc = 85:15), the target compound was isolated as a yellow oil (1.02 g, 2.25 mmol, 49%). 1H NMR (500 MHz, CDCl3): δ 7.33–7.17 (m, 5H), 6.86 (s, 1H), 4.14 (dd, J = 10.8, 5.2 Hz, 1H), 4.07–3.92 (m, 3H), 3.59–3.37 (m, 4H), 3.34 (s, 3H), 3.05 (dt, J = 13.8, 7.0 Hz, 1H), 2.48 (qd, J = 7.1, 1.5 Hz, 4H), 1.98–1.79 (m, 2H), 1.00 (t, J = 7.1 Hz, 6H). 13C{1H} NMR (125 MHz, CDCl3): δ 145.5, 140.2, 136.7, 136.2, 132.2, 129.1, 128.8, 127.8, 69.5, 58.9, 52.4, 47.9, 45.8, 44.7, 43.2, 29.7, 15.1. FTIR (NaCl, thin film): 3422, 2967, 2114, 1657, 1643, 1463, 1377, 1339, 1165, 1115 cm-1. HRMS: (ESI-TOF) calc’d for C21H31N2O3S3 [M+H]+ 455.1491, found 455.1520. TLC (3:1 hexanes:EtOAc), Rf: 0.36 (UV).
Compound 2
Compound 2 was synthesized according to general procedure 5 on a 2.24 mmol scale. After column chromatography on silica (DCM:MeOH = 99:1), the target compound was isolated as a brown oil (733 mg, 1.78 mmol, 79%). The spectroscopic data matched the data reported above. Compound 3
Compound 3 was synthesized according to general procedure 6 on a 1.77 mmol scale. After column chromatography on silica (DCM:MeOH = 97:3), the target compound was isolated as a yellow oil (64.0 mg, 161 µmol, 9.1%). The spectroscopic data matched the data reported above.
Compound 5
Compound 5 was synthesized according to general procedure 7 on a 153 µmol scale. After purification by column chromatography (CH2Cl2:MeOH = 93:7 to 9:1). Carboxylic acid (22.5 mg, 54.7 µmol, 36%) was isolated as a colorless oil. The spectroscopic data matched the data reported above. Compound 15
In a 1-dram vial equipped with a stir bar, acid 5 (10.0 mg, 24.3 µmol, 1.0 equiv.) was dissolved in DCM:MeCN (9:1, 243 µL). To the solution were added N-(3- Dimethylaminopropyl)-N′-ethylcarbodiimide hydrochloride (5.12 mg, 26.7 µmol, 1.1 equiv.), pentafluorophenol (4.92 mg, 26.7 µmol, 1.1 equiv.), and DMAP (0.3 mg, 2.43 µmol, 0.1 equiv.). The reaction was stirred overnight at room temperature. Afterwards, the volatile materials were evaporated under reduced pressure. The crude material was purified by preparative HPLC (C18 column, 9.4 x 250 mm) to obtain the target compound 15 (8.2 mg, 14.2 µmol, 58%) as a colorless oil. 1H NMR (400 MHz, CD3CN): δ 7.60 (s, 1H), 6.19 (bs, 2H), 4.42 (dd, J = 10.8, 5.5 Hz, 1H), 4.05 (ddd, J = 15.0, 10.8, 0.8 Hz, 1H), 3.86–3.65 (m, 2H), 3.46 (dt, J = 14.3, 6.7 Hz, 1H), 3.12 (td, J = 6.5, 1.6 Hz, 2H), 2.58 (qd, J = 7.1, 1.9 Hz, 4H), 1.05 (t, J = 7.1 Hz, 6H).
13C{1H} NMR (101 MHz, CD3CN): δ 168.6, 148.9, 147.4, 138.3, 131.2, 53.5, 49.0, 45.3, 45.3, 34.8, 15.2. Note: Phenol carbons not observed due to C–F coupling. 19F{1H} NMR (376 MHz, CD3CN): δ –154.33 – –154.53 (m), –160.16 (t, J = 21.0 Hz), –164.10 – –164.94 (m). HRMS: (ESI-TOF) calc’d for C19H21F5N3O6S3 [M+H]+ 578.0507, found 578.0510. Chemical synthesis of NHS-ester brinzolamide using click chemistry
General Summary A reactive handle may be installed to brinzolamide (BZA). Reductive amination of the secondary amine of brinzolamide (1) followed by treatment of the intermediary methoxy ether (2) with boron tribromide provides access to either the primary alcohol (3) or the primary alkyl bromide (4). These are both useful intermediates for further derivatization. Oxidation of the primary alcohol to the carboxylic acid (5) and EDC coupling with N-hydroxysuccinimide gives NHS ester (6). Other carboxylic acid derivatives for chemical conjugation can also be prepared by this method. Alternatively, the alkyl bromide (4) can be converted to the alkyl azide (7), which can be used in click cycloaddition reactions to make additional NHS derivatives. The alkyl azide can also be used to conjugate a small-molecule cargo, such as a fluorophore, to brinzolamide. Payload delivery across the BBB Disclosed herein include methods and delivery systems for delivering a payload (e.g., a therapeutic agent) to a nervous system. The method comprise providing a small molecule capable of interacting with a carbonic anhydrase IV or a derivative thereof. The small molecule can be part of a delivery system and the delivery system can comprise a payload to be delivered to a nervous system. The method can further comprise administering the delivery system to the subject. In some embodiments, the delivery system comprises nanoparticles, nanotubes, nanowires, dendrimers, liposomes, ethosomes and aquasomes, polymersomes and niosomes, foams, hydrogels, cubosomes, quantum dots, exosomes, macrophages, and combinations thereof. In some embodiments, the delivery system comprises a nanoparticle selected from lipid-based nanoparticles, polymeric nanoparticles, inorganic nanoparticles, surfactant-based emulsions,
nanowires, silica nanoparticles, virus-like particles, peptide or protein-based particles, lipid- polymer particles, nanolipoprotein particles, and combinations thereof. For example, the payload may include an antimicrobial agent, a therapeutic agent, a prodrug, a peptide, a protein, an enzyme, a lipid, a biological response modifier, a pharmaceutical agent, a lymphokine, a heterologous antibody or fragment thereof, a detectable label, a polyethylene glycol (PEG) molecule, or a combination of two or more of the agents. The payload can include a neuroactive polypeptide, for example, a neurotrophic factors, endocrine factors, growth factors, paracrine factors, hypothalamic release factors, neurotransmitter polypeptides, polypeptide agonists for a receptor expressed by a CNS cell, polypeptides involved in lysosomal storage disease or any combination thereof. In another example, the payload can include an IL-1 receptor antagonist (IL-1Ra), dalargin, an interferon-β, Glial-derived neurotrophic factor (GDNF), tumor necrosis factor receptor (TNFR), nerve growth factor (NGF), brain derived neurotrophic factor (BDNF), neurotrophin-4/5, neurotrophin (NT)-3, a neurturin, neuregulin, a netrin, ciliary neurotrophic factor (CNTF), stem cell factor (SCF), a semaphorin, hepatocyte growth factor (HGF), epidermal growth factor (EGF), transforming growth factor (TGF)-cx, TGF-B, vascular endothelial growth factor (VEGF), platelet-derived growth factor (PDGF), heregulin, artemin, persephin, interleukins, granulocyte-colony stimulating factor (CSF), granulocyte-macrophage-CSF, cardiotrophin-1, hedgehogs, leukemia inhibitory factor (LIF), midkine, pleiotrophin, erythropoietin (EPO), bone morphogenetic proteins (BMPs), netrins, saposins, any fragment thereof, or any combination thereof. Aspects of the invention also provide for delivery of the conjugate to a subject in order to transport a therapeutic agent across the BBB. In aspects of the invention, delivery of the therapeutic payload may be for the treatment of a disease, disorder, or injury of the CNS. In aspects of the invention, the therapeutic agent may be released from the conjugate following entry into the CNS. In certain aspects, the disease, disorder, or injury of the CNS can be, without limitation, multiple sclerosis (MS), amylotrophic lateral sclerosis (ALS), Huntington's disease, Alzheimer's disease, Parkinson's disease, spinal cord injury, traumatic brain injury, stroke, neuropathic pain, neurodegeneration, neuroinflammation, progressive multifocal leukoencephalopathy (PML), encephalomyelitis (EPL), central pontine myelolysis (CPM), adrenoleukodystrophy, Alexander's disease, Pelizaeus Merzbacher disease (PMZ), Globoid cell Leucodystrophy (Krabbe's disease), Wallerian Degeneration, optic neuritis, transverse myelitis,
post radiation injury, neurologic complications of chemotherapy, acute ischemic optic neuropathy, vitamin E deficiency, isolated vitamin E deficiency syndrome, Bassen-Kornzweig syndrome, Marchiafava-Bignami syndrome, metachromatic leukodystrophy, trigeminal neuralgia, Bell's palsy, primary tumors, secondary metastases, or any combination thereof. Experimental examples Study Design The blood-brain barrier (BBB) serves as a highly selective semi-permeable membrane that separates the circulating blood from the brain and extracellular fluid in the central nervous system (CNS). Due to its selective nature, this barrier restricts the entry of numerous therapeutic agents, especially large molecules, into the brain. Mechanism-agnostic engineering strategies, such as directed evolution of adeno-associated viral capsids, have succeeded in enabling cross- BBB delivery in animals. However, the brain shuttle molecules generated through this strategy have unpredictable translatability across species due to unclear mechanism. Receptor-mediated transport mechanisms have emerged as promising avenues to rationally engineer translatable molecular shuttles that facilitate the transport of therapeutic agents across the BBB. However, previously identified receptors all express ubiquitously in both CNS and peripheral tissues, causing delivery to off-target tissues and potential safety liabilities such as decreased number of reticulocytes. Moreover, most of these binder-cargo conjugates require individual optimization. There remains a need for a more specific and versatile method that can address the delivery of biologics to the brain. The present invention provides rationally designed reactive small-molecule binders based on carbonic anhydrase inhibitor brinzolamide, that can serve as a shuttle to facilitate CA-IV- mediated brain delivery. Design of activated CA-IV binders for bioconjugation One important factor considered in designing shuttles of the invention as the epitope that was be targeted. This is because the efficiency of receptor-mediated transcytosis can vary depending on the specific epitope on the BBB receptor that a binder is aiming for. AAV capsids proposed to rely on CA-IV to penetrate the blood-brain barrier bind to a deep pocket on their
surface that functions as a catalytic center for their enzymatic processes, implying that binding to the pocket may be sufficient to trigger efficient CA-IV-mediated transcytosis. FIG.1 is a schematic of CA-IV mediated transcytosis and antibody conjugation of the invention. Carbonic anhydrase binders have been modified with reaction groups that are designed to facilitate a one-step bioconjugation to therapeutic cargo while minimizing any impact on CA-IV binding. The conjugate can attach to the CA-IV receptor that is present on brain endothelial cells, thus prompting transcytosis to occur across the BBB. To effectively target the CA-IV catalytic pocket, existing binder molecules were unutilized that have been developed for other intracellular carbonic anhydrase family members. These carbonic anhydrase inhibitors may exhibit cross-reactivity with CA-IV and can serve as the parent compounds for our shuttle molecules. An example of this is brinzolamide (BZA), which is an FDA-approved generic inhibitor of intracellular CA-II with desirable physicochemical properties and safety profile. By the present invention, it was discovered that this compound binds to CA-IV using a possible overlap binding site with brain-transducing AAVs. To enable the bioconjugation of brinzolamide with therapeutic cargos, brinzolamide was modified by introducing an N-Hydroxysuccinimide (NHS)-ester group. NHS-ester was selected for its ability to react with a wide range of protein molecules that have exposed primary amines on their surface under mild buffer conditions and at room temperature. Structure analysis of brinzolamide in complex with a mouse CA-IV receptor revealed that the methyl ether moiety of brinzolamide is exposed on the receptor surface. FIG.2A is a structural analysis of the murine CA-IV binding pocket and its interaction with designed AAVs, BZA, and activated BZA (BZA-2C-NHS). FIG.2B is a structural alignment of BZA across CA-IV homologues. The alignment shows that all residues immediately next to the BZA binding pocket have same identities across all homologs. BZA-mediated receptor binding and targeted delivery may potentially be translated across species. Without being limited by a mechanism of action, this suggested that therapeutic cargo could be conjugated to the receptor without facing any steric hindrance. Examination of CA-IV
structures from diverse mammalian species verified the high conservation of this binding pocket, as detailed below by amino acid sequence alignment and residues forming the binding pocket in bold:
Without being bound by a mechanism of action, it was proposed that that brinzolamide binds to various CA-IV homologs in a similar manner. Following the structure analyses, brinzolamide derivatives were designed featuring a reactive NHS-ester to substitute the methyl ether group. To simplify the synthesis of an NHS-ester-brinzolamide, an extra ethyl group was
introduced to convert the secondary amine into a tertiary amine, thereby minimizing self- reaction. Production of BZA-conjugated cargo through NHS-ester ligation The NHS-ester ligation was performed in a neutral pH buffer to maintain the stability of cargo molecules. For protein cargos, the reaction buffer was DPBS with 0.001% Pluronic F-68 (2.7mM KCl, 1.5mM KH2PO4, 136.9 mM NaCl, 8.1mM Na2HPO4, pH 7.4; 0.001% Pluronic F- 68). For siRNA cargos (described below), the reaction buffer was nuclease-free duplex buffer (30 mM HEPES, pH 7.5; 100 mM potassium acetate) NHS-ester-brinzolamide powder was dissolved in DMSO at 50mM, aliquoted, and stored at -80℃. Right before the reaction, the stock NHS-ester-brinzolamide solution was dissolved to 100μL of reaction buffer at 10x of the target final concentration. The 10x solution was mixed with purified cargo molecule diluted in 900 μL of the same reaction buffer in a 2 mL centrifuge tube. The tube was rotated and agitated in 25℃ during the reaction. The reaction was then stopped by adding 1 μL Tris-based buffer with at least 500mM of amine groups (e.g., 1M Tris- HCl pH7.5). The resulting product was purified using a PD-10 column following the manufacturer’s protocol to exchange buffer to fresh DPBS with 0.001% Pluronic F-68. The product was concentrated to up to 10 mg/mL using a 30 kD MWCO protein concentrator. The final product was sterilized using a 0.22uM filter. The final concentration was measured using nanodrop.
Production of BZA-conjugated cargo through CuAAC ligation Both azide-brinzolamide and alkyne-Alexa647 were dissolved in 1:1 mixture of water + tert-butyl alcohol at 5 mM. Sodium ascorbate was dissolved in water at 12.5 mM (10x working concentration). Copper(II) sulfate pentahydrate was dissolved in water at 12.5 mM (100x working concentration). In 2 mL centrifuge tube, a reaction was made by mixing 50 μL of azide- brinzolamide, 50 μL of alkyne-Alexa647, 10 μL of sodium ascorbate, and 1 μL of copper(II) sulfate pentahydrate. The tube was rotated and agitated in 25℃ overnight during the reaction. The product was stored in -20℃ before being tested Synthesis of BZA-2C-NHS from a commercially available brinzolamide molecule
AcOH = acetic acid, THF = tetrahydrofuran, DCM = dichloromethane, DMAP = 4- dimethylaminopyridine, EDCI = N-(3-Dimethylaminopropyl)-N′-ethylcarbodiimide hydrochloride, TSTU = N,N,N′,N′-tetramethyl-O-(N-succinimidyl)uronium tetrafluoroborate, DIPEA = N,N- diisopropylethylamine FIG.3 shows an 1H-NMR verification of chemical synthesis of NHS-ester-brinzolamide. Biologics conjugated to carbonic anhydrase binder interactions with CA-IV in vitro To examine whether NHS-ester brinzolamide can conjugate to a biological cargo, BZA- 2C-NHS was used to react with a commercially accessible anti-human PD-L1 antibody, Atezolizumab biosimilar (Ate), a model for human antibody cargo. After purification, the conjugation product was analyzed using Liquid chromatography-mass spectrometry (LC-MS) and compared to the unconjugated Ate. FIG.4A is a structural diagram showing Ate conjugation to BZA using linker 2C to generate BZA-2C-Ate. FIG.4B-G are LC-MS graphs of BZA-Ate conjugation. Approximately half of the light chains and half of the heavy chains were found to contain at least one copy of BZA (~400 Da) conjugation after reacting with BZA-2C-NHS. This indicated that over 90% of IgG complexes, each made up of two light chains and two heavy chains are attached to at least one copy of the BZA compound
LC-MS with electrospray shows that more than half of both heavy and light chains of an IgG antibody are conjugated with 1-3 brinzolamide moieties (~390 Da) compared to the unmodified Atezolizumab control. Cell-based binding assays with HEK293T cells and HeLa cells, cell-based internalization assays with pHrodo dyes, SPR assays, in vivo biodistribution and pharmacokinetic characterization, and ELIZA assays were conducted, as described below: Select Experimental Methods Cell-based binding assays – HEK293T HEK293T cells were seeded at 80% confluency in six-well plates and maintained in Dulbecco’s modified Eagle’s medium (DMEM) supplemented with 5% fetal bovine serum (FBS), 1% nonessential amino acids (NEAAs), and penicillin-streptomycin (100 U/ml) at 37°C in 5% CO2. Membrane-associated human and mouse CA-IV were transfected by polyethylenimine (PolySciences, no.23966). Cells were seeded on Neuvitro poly-d-lysine– coated sterile German glass coverslips (Fisher Scientific, no. NC0343705) 24 hours after transfection in 24-well plates. Brinzolamide-conjugated nanobodies and antibodies were added to the media at 0.4 and 0.2 µM respectively and incubated for one hour at 37˚C in 5% CO2 then fixed in 4% PFA. Coverslips were blocked with 1× tris-buffered saline (TBS) containing 3% bovine serum albumin (BSA) and 0.3% Triton-X100 for permeabilizing conditions for 30 min and incubated in secondary antibody (nanobodies, 1:1000 dilution; GenScript, A01994; antibodies, 1:1000 dilution; Invitrogen, A-21445) in 1× TBS and 3% BSA with 0.05% Triton X- 100 for permeabilizing conditions for 60 min at ambient temperature. Coverslips were washed three times in 1× TBS. Coverslips were mounted on slides with Diamond antifade mounting media with 4′,6-diamidino-2-phenylindole (Invitrogen, P36931). Fluorescent microscopic images were captured on a confocal laser scanning microscope (LSM 880, Carl Zeiss, USA). Cell-based binding assays – HeLa HeLa cells were seeded at 80% confluency in six-well plates and maintained in Dulbecco’s modified Eagle’s medium (DMEM) supplemented with 10% fetal bovine serum
(FBS), 1% nonessential amino acids (NEAAs), and penicillin-streptomycin (100 U/ml) at 37°C in 5% CO2. Membrane-associated human and mouse CA-IV were transfected by polyethylenimine (PolySciences, no.23966). Cells were seeded in black, 96-well glass-bottom plates coated with poly-L-ornithine (Cellvis, no. P96-1.5H-N; Sigma Aldrich, no. P4957) 24 hours after transfection. Brinzolamide-conjugated nanobodies and antibodies were added to the media at 0.4 and 0.2 µM respectively and incubated for one hour at 37˚C in 5% CO2 then fixed in 4% PFA. Cells were blocked with 1× tris-buffered saline (TBS) containing 3% bovine serum albumin (BSA) and 0.3% Triton-X100 for permeabilizing conditions for 30 min then incubated in secondary antibody (nanobodies, 1:1000 dilution; GenScript, A01994; antibodies, 1:1000 dilution; Invitrogen, A-21445) in 1× TBS and 3% BSA with 0.05% Triton X-100 for permeabilizing conditions for 60 min at ambient temperature. Cells were washed three times in 1× TBS. Fluorescent microscopic images were captured on a confocal laser scanning microscope (LSM 980, Carl Zeiss, USA). Cell-based internalization assays with pHrodo dyes HeLa cells were seeded at 80% confluency in six-well plates and maintained in Dulbecco’s modified Eagle’s medium (DMEM) supplemented with 10% fetal bovine serum (FBS), 1% nonessential amino acids (NEAAs), and penicillin-streptomycin (100 U/ml) at 37°C in 5% CO2. Membrane-associated CA-IV of human, mouse, marmoset, or rhesus macaque origin were co-transfected with CAG-mNeonGreen at a 1:1 ratio by polyethylenimine (PolySciences, no.23966). The following day, cells were seeded in black, 96-well plates with an optically clear polystyrene bottom (Greiner Bio-One, no.655096) and maintained in FluoroBrite™ Dulbecco’s modified Eagle’s medium (DMEM) supplemented with 5% fetal bovine serum (FBS), 1% nonessential amino acids (NEAAs), penicillin-streptomycin (100 U/ml), 1x GlutaMAX, and 15 mM HEPES at 37°C in 5% CO2. Once cells adhered to the plate, antibodies conjugated to small molecules by various linkers were labeled with a pH sensing dye (Invitrogen, Z25612) following the manufacturer’s protocol with adjustments for dosing needs. Briefly, antibodies were labeled by combining an 800 nM solution of the treatment antibody with a 960 nM solution of the labeling reagent at a 1:1 v/v ratio. After 10 minutes at room temperature, the labeled antibodies were added to cells at a 1:1 v/v ratio. Following three or six hours of incubation, the cells were
stained with NucBlue LiveReady Probes (Invitrogen, R37065) and imaged on a high content confocal microscope with a 40x objective (Micro Confocal ImageXPress, Molecular Devices). Surface Plasmon Resonance (SPR) A Sierra SPR-32 (Bruker) loaded with a protein A sensor chip was used. For nanobodies and siRNA cargos, Fc-fusion CA-IV receptor proteins in HBS-EP+ buffer (GE Healthcare) were immobilized at 200nM. Conjugated and unconjugated cargo molecules in the same buffer at labeled concentrations were injected. For IgG antibody cargos, the setup was reversed, where cargo proteins in HBS-EP+ buffer (GE Healthcare) were immobilized at 200nM, and the untagged CA-IV receptor proteins at labeled concentrations were injected. The analyte was injected at a flow rate of 10 uL per min for 240 s followed by a 600 s dissociation. Regeneration steps with 10 mM glycine pH 1.5 was performed between each cycle. All kinetic data were double reference subtracted. In vivo biodistribution and pharmacokinetics characterization 8 week old C57BL/6J mice were intravenously injected with the 0.05mg IgG antibodies. At the indicated time point, to harvest the tissues of interest, the mice were anesthetized with Euthasol (pentobarbital sodium and phenytoin sodium solution, Virbac AH) and transcardially perfused using 30–50 mL of 0.1 M phosphate-buffered saline (PBS) (pH 7.4), followed by 30– 50 mL of 4% paraformaldehyde (PFA) in 0.1 M PBS. The organs were collected and post-fixed 24–48 h in 4% PFA at 4 °C. Following this, the tissues were washed with 0.1 M PBS twice and stored in fresh PBS-azide (0.1 M PBS containing 0.05% sodium azide) at 4 °C. For immunochemistry, brains and livers were sectioned into 100um thick slice with vibratome. The slices were first incubated in the blocking buffer (10% normal donkey serum (NDS), 0.1% Triton X-100, and 0.01% sodium azide in 0.1 M PBS, pH 7.4) with antibody anti-human Igg-Alexa647 at 1:200. The slices were then washed 3 times in 0.1 M PBS over a total duration of 5–6 h. When performing DNA staining, the tissues were incubated with 4’, 6-diamidino-2-phenylindole (DAPI) (Sigma-Aldrich, 10236276001, 1:1000) in 0.1 M PBS for 15 min followed by a single wash for 10 min in 0.1 M PBS. The DAPI and/or antibody-stained tissue sections were mounted with ProLong Diamond Antifade Mountant (Thermo Fisher Scientific, P36970) before imaging them under the microscope.
ELISA Both Ate and BZA-Ate concentrations were quantified using an anti-human IgG ELISA Kit (abcam, #ab195215). PBS perfused organs from mice were collected in tubes with prefilled beads (Benchmark #D1032-30, #D1032-15, #D1033-28). These tubes were then filled up with 1 mL of cell extraction buffer from the ELISA’s kit following the protocol. Tubes were then placed into a Benchmark BeadBlaster 24 homogenization machine at 7 m/s speed, 2 total cycles with 15 second cycles and a 30 second pause interval between each cycle. After 2 cycles, a manual 1 minute pause was given. This homogenization and pause step were repeated two more times. Samples were centrifuged at 18,000g, 4°C for 30 minutes.700 µL of supernatant was collected into two wells, 350 µL each well without disturbing the debris near the beads. Supernatant was then diluted in DI water, and 50 µL of diluted sample was pipetted onto each well of the precoated ELISA plate from the kit. The diluted antibody solution and standard from the kit were prepared following its protocol. Standard concentrations ranged from 0.23 ng/mL to 15 ng/mL.50 µL of diluted antibody solution was then added to every well, and the plate was incubated for 50 minutes on a shaker. The plate was then washed 3x with wash buffer from the kit, and 100 µL of TMB solution was added to each well and incubated for 35 minutes on a shaker.100 µL of stop solution was then added to the wells, and the plate was sealed and read at 450 nm absorbance. FIG.5A-D are graphs of results from a SPR assays of CA-IV and BZA-2C-Ate. SPR assays evaluated the binding interaction between conjugated or unconjugated IgG antibody and purified CA-IV proteins.200 nM IgG antibody was immobilized on a Protein A- precoated capture sensor, and CA-IV proteins at different concentrations were subsequently introduced. Receptor concentrations are indicated in the inset. A concentration-dependent binding signal was observed between CA-IV and BZA-2C- Ate, with estimated dissociation constants ranging from 100nM to 200nM was shown. Following this, tests were conducted to determine if the conjugated antibodies can bind to cultured cells that have an over-expression of CA-IV. FIG.5E shows immunofluorescent images of live CA-IV expressing cells incubated with either unconjugated or BZA-conjugated Ate.
In the immunofluorescent images, the cells were fixed after 1 hr of incubation with 0.2 µM cargo antibody. Following fixation, the cells were washed with a detergent-containing buffer to permeabilize them. Next, the fixed cells were stained with an anti-human antibody conjugated to Alexa Fluor 647 and subsequently imaged. Panels labeled with IF (MIP) display a max- intensity projection of the cells, while panels labeled with IF (z slice) show a section close to the well bottom. In the live cell images, the cargo antibodies were pre-labeled with a fluorescent pH indicator, pHrodo, which exhibits increased fluorescence intensity in acidic environments like endosomes before being applied at 0.4 µM to the cells. The treated cells were imaged 6 hr after incubation with the antibodies. Punctate structures were clearly visible inside cells that have been permeabilized before staining, suggesting the internalization of antibodies via endocytosis. Binding and internalization can be observed with CA-IV from various species such as mouse, human, and rhesus macaque. In line with the findings from SPR experiments, immunofluorescence analysis revealed the presence of BZA-2C-Ate signals on cells expressing CA-IV. These signals were observed either at cell boundaries or in the form of puncta within the cell boundaries. In order to provide additional confirmation that the puncta originate from the endocytosis of BZA-conjugated antibodies, pHrodo, a pH-sensitive dye, was employed to label the antibody samples. Subsequently, CA-IV-expressing cells were incubated with labeled antibody samples. The pHrodo dye exhibits a significant increase in fluorescence as the pH decreases, making it an indicator of internalization. After a 6-hour incubation period, a consistent presence of puncta structures was observed, which closely resembled the patterns observed in the immunofluorescence images. The quantified and intensities of puncta within each cell revealed that BZA-2C-Ate exhibited a significantly higher number of puncta compared to unconjugated Ate (p <0.0001), with notably greater average intensities (p <0.0001). FIG.5 F-G show the average and counted puncta, respectively, intensity of CA-IV- expressing HeLa cells incubated with either unconjugated or BZA-2C-Ate conjugates. The in vitro findings demonstrated the consistent binding of BZA-2C-Ate to CA-IV and its subsequent internalization in CA-IV expressing cells across various species, including human, mouse, and macaque. This observation indicated the preservation of crucial residues for brinzolamide interaction, enabling in vivo assessments in rodent and non-human primate models.
BZA-NHS linker variants
Synthesis methods were developed to synthesize the NHS-ester brinzolamide molecule with different linker variations. The identity of synthesis products were confirmed using LC-MS and NMR.
BZA derivates were synthesized and analyzed as follows:
De novo synthesis of NHS-ester-BZ A variants with hydrocarbon linkers.
The carbonic anhydrase inhibitor-based shuttles comprising linker variants of NHS-ester-
BZA were analyzed.
FIG.6A shows the structure of select BZA-Ate linker variants. FIG.6B-M shows LC-MS graphs of unmodified Ate and BZA-Ate linker variant conjugates. As shown, the efficiency of conjugation is dependent on both the length of the linker and the type of binder. FIG.7A-C are graphs of results from SPR assays of CA-IV and BZA-Ate linker variant binding. FIG.7D shows immunofluorescent images of live CA-IV expressing cells incubated with either unconjugated or BZA-conjugated Ate linker variants. Z-slice images of Hela cells incubated with 0.2 µM BZA-conjugated Ate stained for human IgG with BZA-2C-Ate, BZA-3C-Ate, and BZA-4C-Ate show that there are clear puncta inside cells that are permeabilized before staining, indicating internalization of the antibodies. FIG.7E-F show representative live cell images showing CA-IV-expressing HeLa cells incubated with either unconjugated or BZA-conjugated antibodies after 3 hours and 6 hours of incubation with antibodies, respectively. In Puncta identified in the live cell images were segmented, and the intensities contained within each punctum are quantified and averaged by cells. FIG.7G-4H show the average and counted puncta, respectively, intensity of CA-IV- expressing Hela cells incubated with either unconjugated or BZA-conjugated linker variants after 3 hours. FIG.7I-4J show the average and counted puncta, respectively, intensity of CA-IV- expressing Hela cells incubated with either unconjugated or BZA-conjugated linker variants after 6 hours. The average puncta intensity of each cell was calculated and displayed as a box plot, with outliers highlighted. Statistical significance was determined using Student’s t-test. The number of puncta of each cell was counted and displayed as a box plot, with outliers highlighted. Statistical significance was determined using Poisson means test. (Statistical significance, *: 0.01 < p <= 0.05, **: 0.001 < p <= 0.01, ***: 0.0001 < p <= 0.01, ****: p <= 0.0001)
With BZA-2C-Ate, BZA-3C-Ate, and BZA-4C-Ate, there are clear puncta inside cells that are permeabilized before staining, indicating internalization of the antibodies. Of the NHS-ester-BZA linker variants examined, BZA-3C-NHS exhibited the highest drug-to-antibody ratio. BZA-2C-NHS facilitated highly efficient internalization and could be synthesized easily (4 steps compared to 8 steps for other linker variants) using commercially available starting materials. NHS-ester derivates of other CA-IV binders In addition to examining BZA, a range of other carbonic anhydrase inhibitor-based shuttles were examined, comprising NHS-ester derivatives of other CA-IV binders, namely acetazolamide (AZA) and dorzolamide (DZA). AZA and DZA In vitro characterization of an IgG cargo conjugated to alternative CA-IV binders FIG.8A shows the structures of NHS-ester variants of two alternative CA-IV binders, acetazolamide (AZA) and dorzolamide (DZA). FIG.8B-E show LC-MS graphs of NHS ester variants of two alternative CA-IV binders, AZA and DZA respectively. FIG.8F-G is a graph of results from an SPR assay of CA-IV and NHS ester variants of two alternative CA-IV binders, AZA and DZA respectively. FIG.8H-I show live cell images showing CA-IV-expressing Hela cells incubated with either AZA-conjugated or DZA-conjugated Ate, respectively. In Puncta identified in the live cell images were segmented, and the intensities contained within each punctum are quantified and averaged by cells. FIG.8J-K show the average and counted puncta intensity, respectively, of CA-IV- expressing Hela cells incubated with either unconjugated, AZA-conjugated, or DZA-conjugated Ate. The average puncta intensity of each cell was calculated and displayed as a box plot, with outliers highlighted. Statistical significance was determined using Student’s t-test. The number
of puncta of each cell was counted and displayed as a box plot, with outliers highlighted. Statistical significance was determined using Poisson means test. (Statistical significance, *: 0.01 < p <= 0.05, **: 0.001 < p <= 0.01, ***: 0.0001 < p <= 0.01, ****: p <= 0.0001) When compared to BZA-based variants, compounds utilizing AZA and DZA as binding cores demonstrated inferior conjugation efficiency and binding signal strength. The functional binding between the Ate antibody and its target antigen is unaffected by the conjugation of these compounds to lysine residues, regardless of the type of linker and binding core employed. BZA and BZA-2C-IgG cargo variants In addition to conjugating the Ate antibody, the potential for extending this bioconjugation approach to other biological molecules and modalities to facilitate receptor binding was explored. Additional in vitro characterization of BZA-NHS conjugated to different therapeutic modalities was tested, including nanobodies, siRNA, and small molecules. IgG antibodies (Don) Immunoglobulin G (IgG) antibodies are crucial components of the immune response, capable of neutralizing pathogens and signaling immune cells. The potential for therapeutic application of a BZA-modified human IgG1, focusing on its interaction with CA-IV was explored. Multiple IgG antibodies, in addition to Ate, were shown to be able to be conjugated to BZA, facilitating binding to CA-IV in vitro, including Donanemab (Don). BZA-modified IgG antibodies, including Donanemab, were generated which target beta amyloid aggregates, and Atezolizumab, which targets PD-L1. For Donanemab, two reaction conditions were used, namely a high-label-density condition (reaction condition 1, 40μM NHS- ester-BZA: 0.2μM antibody) and a low-label-density condition (reaction condition 2, 4μM NHS- ester-BZA: 0.2μM antibody). For Atezolizumab, only one reaction condition was used (200μM NHS-ester-BZA: 10μM antibody). FIG.9A shows the structures of an IgG antibody BZA variant conjugates. FIG.9B-K show LC-MS graphs of BZA-IgG antibody conjugates including Don.
FIG.10A-E show graphs of the results from an SPR assay of CA-IV and BZA- conjugated Don, unconjugated Don, and BZA-conjugated higG1 isotype, respectively. FIG.10F shows representative immunofluorescent and live cell images showing CA-IV- expressing Hela cells incubated with either unconjugated-IgG of BZA-conjugated variant antibodies including Don. In Puncta identified in the live cell images were segmented, and the intensities contained within each punctum are quantified and averaged by cells. FIG.10G-H show the average and counted puncta intensity, respectively, of CA-IV- expressing Hela cells incubated with either unconjugated or BZA conjugated antibody variants. Including Don FIG.11A-D show SPR of modified and unmodified therapeutic IgG antibodies tested against purified human CA-IV and mouse CA-IV, including atezolizumab and Don. The IgG antibodies were immobilized on a Protein A chip, and CA-IV proteins at different concentrations are injected. Receptor concentrations were indicated in the inset. FIG.11E-F show internalization assays in cultured Hela cells with the BZA-modified IgG antibodies and unmodified IgG antibodies, including atezolizumab and Don. Hela cells are transfected with indicated receptors and incubated with indicated antibodies. After fixation, the cells are washed with buffer either with detergent (permeabilized) or without detergents (unpermeabilized). The fixed cells were then stained with anti-human antibody conjugated to Alexa Fluor 647 and imaged. C, Max-intensity projection images of cells incubated with BZA-modified and unmodified antibodies. Only BZA modified antibodies show evident binding to cells overexpressing CA-IV proteins. Z-slice images of Hela cells incubated with BZA-modified and unmodified Atezolizumab show clear puncta inside cells that are permeabilized before staining, indicating endocytosis of the antibodies. Binding and endocytosis can be observed with CA-IV from different species, including mouse, human, and rhesus macaque. In the immunofluorescent images, the cells were fixed after 1 hr of incubation with 0.2 µM cargo antibodies. Following fixation, the cells were washed with a detergent-containing
buffer to permeabilize them. Next, the fixed cells were stained with an anti-human antibody conjugated to Alexa Fluor 647 and subsequently imaged. Panels labeled with IF (z slice) showed a section close to the well bottom. In the live cell images, the cargo antibodies were pre-labeled with a fluorescent pH indicator, pHrodo, which exhibited increased fluorescence intensity in acidic environments like endosomes before being used to treat the cells. The treated cells were imaged 6 hr after incubation with the antibodies. Punctate structures were clearly visible inside cells that have been permeabilized before staining, suggesting the internalization of antibodies via endocytosis. Binding and internalization could be observed with CA-IV from various species. The BZA-modified therapeutic human IgG1 showed binding affinity to purified CA-IV with a Kd of 100-300nM. This is similar to the Kd of Denali's TfR-binding ATVs, suggesting comparable effectiveness. Similar to the case in modified nanobodies, products from high-label- density condition showed stronger binding to CA-IV proteins, indicating that controlling the label density can be a potential way to tune the binding affinity of modified proteins. The BZA- modified IgG molecules also show binding to cultured cells overexpressing CA-IV. Importantly, BZA-modified IgG can bind to not only human CA-IV protein, but also CA-IV proteins from mouse and rhesus macaque. This allows the modified IgG molecules to be tested in both rodent and non-human primate (NHP) models. The ability of BZA-modified IgG antibodies to penetrate and localize in the brain in mice was tested and found that the BZA-modified Atezolizumab displayed sustained and brain- enriched signals when compared to its unmodified counterpart. Sagittal sections of mouse brains that were intravenously injected with the antibodies showed an unequivocally stronger brain enrichment for BZA-modified Atezolizumab biosimilars compared to the unmodified version starting from 48 hrs post injection. Notably, certain sections of the brain cortex, which were costained with a neuronal marker, demonstrated that BZA-modified Atezolizumab biosimilar molecules were able to cross the blood-brain barrier and bind to neurons. To confirm that this enrichment is not universal to all organs, liver sections from mice intravenously injected with both types of Atezolizumab were examined, where no significant difference was observed.
Nanobodies Nanobodies, or camelid single-domain antibodies, have been highlighted for their high stability, high yield, and demonstrated therapeutic value with several FDA-approved drugs currently in use. Notably, these nanobodies, derived from Camelus dromedarius, have three surface- exposed lysine residues in the conserved region. This structure allows for chemical modification without obstructing target binding, making them ideal for further manipulation in therapeutic applications. A commercially available nanobody, anti-GFP VHH, was conjugated to brinzolamide using the NHS ester ligation. The protein concentration did not show notable change after the reaction, indicating the reaction product's stability and high yield potential. Two reaction conditions were used, namely a high-label-density condition (reaction condition 1, 40μM NHS- ester-BZA : 2μM nanobody) and a low-label-density condition (reaction condition 2, 4μM NHS- ester-BZA : 2μM nanobody). FIG.12A-B show the structure of a BZA-nanobody variants. FIG.12C-D show the LC-MS analysis of unmodified nanobodies and nanobodies BZA- conjugated nanobodies. The results demonstrate that approximately half of the nanobodies conjugated with 1-2 BZA moieties (~390 Da). FIG.12E-H show graphs of the results from an SPR assay of the binding interaction between an unconjugated or BZA-conjugated nanobody and Fc-tagged CA-IV proteins. 200 nM Fc-tagged CA-IV protein were immobilized on a Protein A-precoated capture sensor, and nanobodies at different concentrations were subsequently introduced. Nanobody concentrations were indicated in the inset. The background binding signal observed in unconjugated nanobody were likely due to weak interaction between nanobody and Protein A. FIG.12I show representative immunofluorescent images showing CA-IV-expressing Hela cells incubated with either anti-GFP VHH unconjugated nanobodies, BZA-2C conjugated nanobodies, and BZA-3C conjugated nanobodies. In the immunofluorescent images, the cells were fixed after 1 hr of incubation with 0.4 µM cargo nanobody. Following fixation, the cells were washed with a detergent-containing
buffer to permeabilize them. Next, the fixed cells were stained with an anti-camelid antibody conjugated to Alexa Fluor 647 and subsequently imaged. Max-intensity projection images are shown. Scale bar, 100 µm. FIG.13A-B shows graphs of the results from an SPR assay of modified and unmodified nanobodies tested against purified human CA-IV and mouse CA-IV. The CA-IV proteins are fused to a human Fc tag and immobilized on a Protein A chip. Nanobodies are injected at different concentrations as indicated in the inset. The background signal seen in unmodified nanobody is likely due to the sporadic interaction between nanobody and protein A. FIG.13C-D show an internalization assay in cultured HEK293 cells with the BZA- modified nanobodies and unmodified nanobodies. After fixation, cells incubated with the nanobodies are washed with buffer either with detergent (permeabilized) or without detergents (unpermeabilized). The fixed cells were then stained with anti-camelid antibody conjugated to Alexa Fluor 647. In the immunofluorescent images, the cells were fixed after 1 hr of incubation with 0.4 µM cargo nanobody. Following fixation, the cells were washed with a detergent-containing buffer to permeabilize them. Next, the fixed cells were stained with an anti-camelid antibody conjugated to Alexa Fluor 647 and subsequently imaged. Max-intensity projection images are shown. Scale bar, 100 µm. Evidence from Surface Plasmon Resonance (SPR) and cell-based binding assays suggests a direct binding interaction between BZA-modified nanobody and purified human sulfonic carbonic anhydrase IV (hCA-IV) protein. The products from high-label-density condition showed stronger binding to CA-IV proteins, indicating that controlling the label density can be a potential way to tune the binding affinity of modified proteins. Furthermore, the BZA-modified nanobody was also able to bind to purified mouse C -IV (mCA-IV). The results demonstrate that conjugation to nanobodies also enables binding to CA-IV. siRNA Small interfering RNA (siRNA) have emerged as a pivotal therapeutic modality in the world of molecular medicine. At its core, siRNA functions by specifically targeting and
degrading messenger RNA (mRNA) sequences, thereby halting the production of targeted proteins. This provides a highly selective mechanism to downregulate disease-causing genes. The importance of siRNA is underscored by its potential to treat a plethora of diseases, especially those where conventional therapies fall short. Unlike traditional therapeutic modalities, which can inadvertently affect multiple targets leading to off-target effects, siRNA provides precise gene silencing, ensuring a higher degree of specificity. Furthermore, the ability of siRNA to target traditionally 'undruggable' genes offers new hope for conditions that have been elusive to treat. Despite the promises, systemic delivery of siRNA to the brain has been a challenging problem. FIG.14A shows the structure of a BZA shuttle-siRNA variant. FIG.14B-D shows graphs of the results from an SPR assay of the binding interaction between an unconjugated or BZA-conjugated siRNA duplex and Fc-tagged CA-IV proteins. 200 nM Fc-tagged CA-IV protein were immobilized on a Protein A-precoated capture sensor, and siRNA at different concentrations were subsequently introduced. siRNA concentrations were indicated in the inset. FIG.14E shows representative immunofluorescent images showing CA-IV-expressing Hela cells incubated with either unconjugated fluorescently labeled siRNA or BZA-2C conjugated siRNA. Representative immunofluorescent images showing CA-IV-expressing Hela cells incubated with either unconjugated or BZA-conjugated fluorescently labeled siRNA molecules with a sequence targeting GFP at 1.0 µM. Scale bar, 100 µm. FIG.15A shows the structure of a BZA shuttle-siRNA variant. The sequence and design of a GFP-targeting siRNA duplex. The sense and antisense strand of the duplex is modified with cholesterol-TEG and a primary amine, respectively. FIG.15B-C show graphs of the results from an SPR assay of the binding interaction between an unconjugated or BZA-conjugated siRNA duplex and Fc-tagged CA-IV proteins.
200 nM Fc-tagged CA-IV protein were immobilized on a Protein A-precoated capture sensor, and siRNA at different concentrations were subsequently introduced. siRNA concentrations were indicated in the inset. The results demonstrate that conjugation of brinzolamide with an oligonucleotide molecule, such as a primary amine-modified siRNA molecule, facilitates CA-IV binding and internalization in CA-IV-expressing cells. Small molecule Small molecules have unique properties that make them desirable for use in drug development, including the ability to pass through cell membranes and be designed to bind specific targets. BZA derivates were conjugated to fluorophores, a type of small molecule, to assess their ability to bind and internalize into cells. To determine if BZA can mediate binding and internalization of small molecule cargos, a BZA-conjugated fluorophore was synthesized using CuAAC. FIG.16A shows the structure of a BZA shuttle-small molecule cargo variant. FIG.16B show representative immunofluorescent images showing CA-IV-expressing Hela cells incubated with either unconjugated small molecule dye or BZA-conjugated small molecules dye. FIG.16C shows the structure of a BZA shuttle-small molecule cargo variant. The schematic shows the process of conjugating a fluorescent molecule (Alexa Fluor 647) to brinzolamide using a CuAAC reaction. FIG.16D show images from an internalization assay using the HEK cells overexpressing CA-IV receptors. The bottom row shows zoom-in images of representative fields of views in the top-row images. The results indicate that BZA fluorophores bind to cells overexpressing either hCA-IV or mCA-IV and are internalized.
FIG.16E shows representative images of brain slices and liver slices from mice injected with BZA-conjugated fluorophore or unconjugated fluorophores. Animals injected with BZA- conjugated fluorophore shows stained vasculature in both brain and liver. FIG.16F shows a fluorescence reading of lysed tissues of animals injected with BZA- conjugated fluorophore or unconjugated fluorophores using a plate reader. Preliminary data suggested that this BZA-conjugated small-molecule fluorophore binds to the cell membrane and gets internalized. This was confirmed by the observations made on the cell contour in the absence of detergent and the presence of puncta in the presence of detergent. In vivo studies showed that BZA-modified fluorophores were localized on brain endothelial cells. Interestingly, this was not the case for fluorophores without BZA modification. Quantification of the fluorescence distribution in different organs indicated that BZA modification increased retention of the small molecule in multiple organs, including the brain. The results demonstrate that conjugation of brinzolamide to a small molecule compound, such as a fluorophore, can result in the localization of the fluorophore within the cell. Notably, BZA-conjugated fluorophore molecules exhibited signal throughout the cytosol, suggesting their ability to penetrate the membrane and potentially interact with intracellular carbonic anhydrases. This variance in membrane permeability and access to carbonic anhydrase family members may lead to differing biodistributions of small versus large BZA-conjugated molecules. The results suggests that BZA-modified nanobodies, therapeutic IgG antibodies, and small molecules have strong potential for broad applications in targeted therapeutics. Their binding affinity to hCA-IV and their ability to mediate the binding and internalization show promising translational potentials. Biologics conjugated to carbonic anhydrase binders cross the BBB in mice BZA-conjugation technology in mice was analyzed in vivo. Both unconjugated Ate and BZA-conjugated Ate were intravenously administered to mice. Animal tissues were harvested at various time intervals for analysis through immunofluorescence imaging. Following intravenous injection, the brain tissues were collected at different time points and then fixed and stained for human IgG. N=3 animals at each time point.
FIG.17A shows immunofluorescent images of mice systemically administered with unconjugated and BZA-conjugated Ate antibodies in sagittal brain sections. Scale bar, 1 mm. FIG.17B shows immunofluorescent images of mice systemically administered with unconjugated and BZA-conjugated Ate antibodies in brain cortex. Scale bar, 100 µm. FIG.17C shows an image of zoomed-in views of the brain cortex of mice systemically administered with unconjugated and BZA-conjugated Ate antibodies 120 hr post injection co- stained with a neuronal marker. Scale bar, 100 µm. Quantification of BZA-conjugated and unconjugated Ate antibodies was determined by IgG concentrations through anti-human IgG ELISA and then normalized based on the total protein concentration measured by BCA. N=3 animals at each time point. FIG.17D shows a graph of biodistribution of BZA-conjugated Ate in comparison to unconjugated control at day 7. Left, fold change of BZA-conjugated Ate concentration over unconjugated Ate. Right, normalized IgG concentrations in the whole brain, the brain vasculature fraction, and the brain parenchyma fraction. FIG.17E-F show graphs of pharmacokinetics of BZA-conjugated and unconjugated Ate in whole brain and liver, respectively, over a period of 14 days. FIG.17G-H show graphs of pharmacokinetics of BZA-conjugated and unconjugated Ate in the brain vasculature and the brain parenchyma, respectively, over a period of 14 days. The BZA-conjugated Ate exhibited prolonged enrichment in the brain, including the cortex, compared to unconjugated Ate. Both immunofluorescent imaging and ELISA quantification indicated a peak in brain IgG signal between day 5 and day 7, followed by a decline within a 14-day period. The ELISA-based quantification on day 7 indicated that the concentration of BZA-conjugated Ate in the brain was one order of magnitude higher than the unconjugated control. The duration of brain exposure for the CA-IV-binding brain shuttle surpassed that of TfR-based shuttles. Further analysis indicated that BZA-conjugated Ate can efficiently traverse the BBB to reach the brain parenchyma. This is evidenced by the co-localization of IgG signals with neurons in immunofluorescence images and ELISA outcomes following capillary depletion, affirming a notable rise in brain distribution within the brain parenchyma fraction.
Moreover, the enrichment induced by BZA displayed a strong preference towards the brain. FIG.18A-B show immunofluorescence images of whole livers sections in mice systemically administered with unconjugated and BZA-conjugated Ate antibodies, scale bar 1 mm and 100 µm, respectively. Following intravenous injection, the liver tissues were collected at different time points and then fixed and stained for human IgG. N=3 animals at each time point. FIG.18C shows quantification of BZA-conjugated and unconjugated Ate antibodies in peripheral organs at day 7 in mice systemically administered with unconjugated and BZA- conjugated Ate antibodies. The IgG concentrations were determined through anti-human IgG ELISA and then normalized based on the total protein concentration measured by BCA. N=3 animals at each time point. The data demonstrated that on day 7, there was no notable increase in distribution evident in major peripheral organs assessed. The distribution pattern aligned with the receptor's predominant expression in the brain and lung. Both ELISA-based quantification and immunofluorescence images validated that the liver distribution of BZA-conjugated Ate did not significantly differ from unconjugated antibodies over a 14-day period. Discussion By the present invention, for the first time, the transportation of an IgG cargo to the brain via the CA-IV receptor was demonstrated. Our results not only validate CA-IV as a BBB receptor for non-viral cargos but also highlight notable advantages over prevalent BBB receptors such as TfR and CD98hc. The elevated brain selectivity of CA-IV mediated delivery offers potential for mitigating toxicity associated with off-target delivery. One common challenge in brain delivery technology development is the failure of binders designed for a BBB receptor in one species to bind to the corresponding receptor in another species. Instances of this issue have been documented with TfR binders and CD98hc binders. However, the present invention’s (referred to as Brain access through Carbonic Anhydrase Binder bioconjugation “BrainCAB”) receptor binder can successfully bind to homologs of CA- IV across various species, such as rodents and non-human primates, due to the conserved binding
pocket of CA-IV. This capability allows for the broad application and evaluation of this technology in diverse model organisms and disease models. The single-step bioconjugation method employed by the present invention exhibits versatility in accommodating various therapeutic agents, including nanobodies and oligonucleotides, underscoring its wide-ranging applicability. Moreover, the straightforward nature of this bioconjugation process enables seamless upscaling of manufacturing. These attributes collectively position the present invention as a versatile technology with practical translational potential for developing therapeutics for neurological indications. The BrainCAB technology, facilitated by its single-step bioconjugation process, demonstrated considerable potential in both in vitro and in vivo studies. With its high BBB- crossing efficiency, enhanced brain specificity, and extended pharmacokinetics, along with its wide applicability and simple production process, this approach shows great potential for delivering therapeutics to conditions traditionally hindered by the intricacies of the blood-brain barrier. Overall, conjugated molecules’ of the invention were shown to exhibit successful binding to CA-IV proteins from different mammalian species and subsequent internalization were verified via surface plasmon resonance (SPR) and cell-based assays. In vivo mouse experiments showed that intravenously administered conjugated therapeutic IgG antibody efficiently penetrated the BBB. Antibody level in the brain parenchyma rose by 10-fold in 7 days compared to unconjugated control, with this substantial boost localized to the brain, matching the CA-IV receptor expression. The wide-reaching potential of this technology, sets the stage for pioneering therapeutic strategies across diverse CNS indications. Incorporation by Reference References and citations to other documents, such as patents, patent applications, patent publications, journals, books, papers, web contents, have been made throughout this disclosure. All such documents are hereby incorporated herein by reference in their entirety for all purposes. Equivalents
Various modifications of the invention and many further embodiments thereof, in addition to those shown and described herein, will become apparent to those skilled in the art from the full contents of this document, including references to the scientific and patent literature cited herein. The subject matter herein contains important information, exemplification and guidance that can be adapted to the practice of this invention in its various embodiments and equivalents thereof.
Claims
Claims 1. A conjugate comprising: a blood brain barrier (BBB) shuttle selected from brinzolamide or a derivative thereof; and a therapeutic cargo conjugated to the shuttle. 2. The conjugate of claim 1, wherein the shuttle is a brinzolamide ester. 3. The conjugate of claim 2, wherein the brinzolamide derivative comprises an N- hydroxysuccinimide (NHS) ester. 4. The conjugate of claim 3, wherein in the brinzolamide derivative, brinzolamide is covalently bonded to the NHS ester via a 1-8 carbon alkyl linker. 5. The conjugate of claim 4, wherein the brinzolamide ester has the formula:
(BZA-nc-NHS), wherein n is 1-8; 6. The conjugate of claim 5, wherein the brinzolamide derivative is selected from among the compounds:
(BZA-8C-NHS). 7. The conjugate of claim 2, wherein the brinzolamide derivative comprises a pentafluorophenyl (PFP) ester. 8. The conjugate of claim 7, wherein in the brinzolamide derivative, brinzolamide is covalently bonded to the PFP ester via polyethylene glycol. 9. The conjugate of claim 8, wherein the brinzolamide derivative is selected from among the compounds:
(BZA-PEG-NHS);
(BZA-2C-PFP). 10. The conjugate of claim 1, wherein the conjugate comprises a plurality of small molecule shuttles conjugated to the therapeutic cargo. 11. The conjugate of claim 1, wherein the therapeutic cargo is a biological molecule. 12. The conjugate of claim 11, wherein the biological molecule is selected from the group consisting of a nucleic acid, a protein, a peptide, an antibody, a nanobody, a lipid, a polysaccharide, and a combination thereof. 13. The conjugate of claim 1, wherein the therapeutic cargo is a small molecule. 14. The conjugate of claim 1, wherein the shuttle is a carbonic anhydrase IV (CA-IV) shuttle. 15. The conjugate of claim 14, wherein when provided to a cell expressing CA-IV as a surface protein, binding of the shuttle to the CA-IV protein mediates transcytosis of the therapeutic cargo across the BBB. 16. A method of delivering a therapeutic cargo across the BBB of a subject, the method comprising: providing to a subject a conjugate comprising:
a blood brain barrier (BBB) shuttle selected from brinzolamide or a derivative thereof; and a therapeutic cargo conjugated to the shuttle. 17. The method of claim 16, wherein the shuttle is a brinzolamide ester. 18. The method of claim 17, wherein the brinzolamide derivative comprises an N- hydroxysuccinimide (NHS) ester. 19. The method of claim 18, wherein in the brinzolamide derivative, brinzolamide is covalently bonded to the NHS ester via a 1-8 carbon alkyl linker. 20. The method of claim 19, wherein the brinzolamide derivative has the formula:
(BZA-nc-NHS), wherein n is 1-8; 21. The method of claim 20, wherein the brinzolamide derivative is selected from among the compounds:
(BZA-8C-NHS). 22. The method of claim 17, wherein the brinzolamide derivative comprises a pentafluorophenyl (PFP) ester. 23. The method of claim 22, wherein in the brinzolamide derivative, brinzolamide is covalently bonded to the PFP ester via polyethylene glycol. 24. The method of claim 23, wherein the brinzolamide derivative is selected from among the compounds:
(BZA-PEG-NHS);
(BZA-2C-PFP). 25. The method of claim 16, wherein the conjugate comprises a plurality of small molecule shuttles conjugated to the therapeutic cargo. 26. The method of claim 16, wherein the therapeutic cargo is a biological molecule. 27. The method of claim 26, wherein the biological molecule is selected from the group consisting of a nucleic acid, a protein, a peptide, an antibody, a nanobody, a lipid, a polysaccharide, and a combination thereof. 28. The method of claim 16, wherein the therapeutic cargo is a small molecule. 29. The method of claim 16, wherein the shuttle is a carbonic anhydrase IV (CA-IV) shuttle. 30. The method of claim 29, wherein when provided to a cell expressing CA-IV as a surface protein, binding of the shuttle to the CA-IV protein mediates transcytosis of the therapeutic cargo across the BBB.
Applications Claiming Priority (6)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US202363537422P | 2023-09-08 | 2023-09-08 | |
| US63/537,422 | 2023-09-08 | ||
| US202363542675P | 2023-10-05 | 2023-10-05 | |
| US63/542,675 | 2023-10-05 | ||
| US202463679469P | 2024-08-05 | 2024-08-05 | |
| US63/679,469 | 2024-08-05 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2025054461A1 true WO2025054461A1 (en) | 2025-03-13 |
Family
ID=94924336
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2024/045602 Pending WO2025054461A1 (en) | 2023-09-08 | 2024-09-06 | Ester derivatives of binders targeting ca-iv |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2025054461A1 (en) |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20190030160A1 (en) * | 2012-05-21 | 2019-01-31 | Genentech, Inc. | Methods for improving safety of blood-brain barrier transport |
| US20190276530A1 (en) * | 2012-08-29 | 2019-09-12 | Hoffmann-La Roche Inc. | Blood brain barrier shuttle |
| WO2023168333A1 (en) * | 2022-03-03 | 2023-09-07 | California Institute Of Technology | Compositions and methods for crossing blood brain barrier |
-
2024
- 2024-09-06 WO PCT/US2024/045602 patent/WO2025054461A1/en active Pending
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20190030160A1 (en) * | 2012-05-21 | 2019-01-31 | Genentech, Inc. | Methods for improving safety of blood-brain barrier transport |
| US20190276530A1 (en) * | 2012-08-29 | 2019-09-12 | Hoffmann-La Roche Inc. | Blood brain barrier shuttle |
| WO2023168333A1 (en) * | 2022-03-03 | 2023-09-07 | California Institute Of Technology | Compositions and methods for crossing blood brain barrier |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| Liu et al. | Dopamine receptor-mediated binding and cellular uptake of polydopamine-coated nanoparticles | |
| Acosta-Ruiz et al. | Branched photoswitchable tethered ligands enable ultra-efficient optical control and detection of G protein-coupled receptors in vivo | |
| Cho et al. | Development of a novel imaging agent using peptide-coated gold nanoparticles toward brain glioma stem cell marker CD133 | |
| López-Andarias et al. | Cell-penetrating streptavidin: a general tool for bifunctional delivery with spatiotemporal control, mediated by transport systems such as adaptive benzopolysulfane networks | |
| JP2022518392A (en) | Transferrin receptor binding molecule, its conjugate, and its use | |
| US11376331B2 (en) | Compositions and methods for transport across the blood brain barrier | |
| EP2152752B1 (en) | Antibody directed to g protein coupled receptors (gpcr) | |
| Haroon et al. | A designed recombinant fusion protein for targeted delivery of siRNA to the mouse brain | |
| Ruderisch et al. | Potent and selective BACE-1 peptide inhibitors lower brain Aβ levels mediated by brain shuttle transport | |
| Röder et al. | Intracellular Delivery of Nanobodies for Imaging of Target Proteins in Live Cells: Röder et al. | |
| Bi et al. | Tagging transferrin receptor with a disulfide FRET probe to gauge the redox state in endosomal compartments | |
| Gu et al. | mRNA delivery enabled by metal–organic nanoparticles | |
| Davis et al. | Protein binding and release by polymeric cell-penetrating peptide mimics | |
| Yeoh et al. | Efficient systemic CNS delivery of a therapeutic antisense oligonucleotide with a blood-brain barrier-penetrating ApoE-derived peptide | |
| Choi et al. | Efficient siRNA delivery into tumor cells by p19-YSA fusion protein | |
| Hoffmann et al. | PEGylation and folic-acid functionalization of cationic lipoplexes—improved nucleic acid transfer into cancer cells | |
| Kim et al. | Presumed LRP1-targeting transport peptide delivers β-secretase inhibitor to neurons in vitro with limited efficiency | |
| US20240053333A1 (en) | Chemical modification of antibodies and functional fragments thereof | |
| Wu et al. | Discovery of novel HER2 targeting peptide-camptothecin conjugates with effective suppression for selective cancer treatment | |
| CN112004826A (en) | Reverse agonistic anti-US 28 antibodies | |
| WO2025054461A1 (en) | Ester derivatives of binders targeting ca-iv | |
| Cordova et al. | Aminopeptidase P mediated targeting for breast tissue specific conjugate delivery | |
| Rogers et al. | Multivalent Display of Erythropoietin on Quantum Dots Enhances Aquaporin-4 Expression and Water Transport in Human Astrocytes In Vitro | |
| Jia et al. | Highly efficient method for intracellular delivery of proteins mediated by cholera toxin-induced protein internalization | |
| Gabashvili et al. | Internalization of transferrin-tagged Myxococcus xanthus encapsulins into mesenchymal stem cells |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 24863677 Country of ref document: EP Kind code of ref document: A1 |