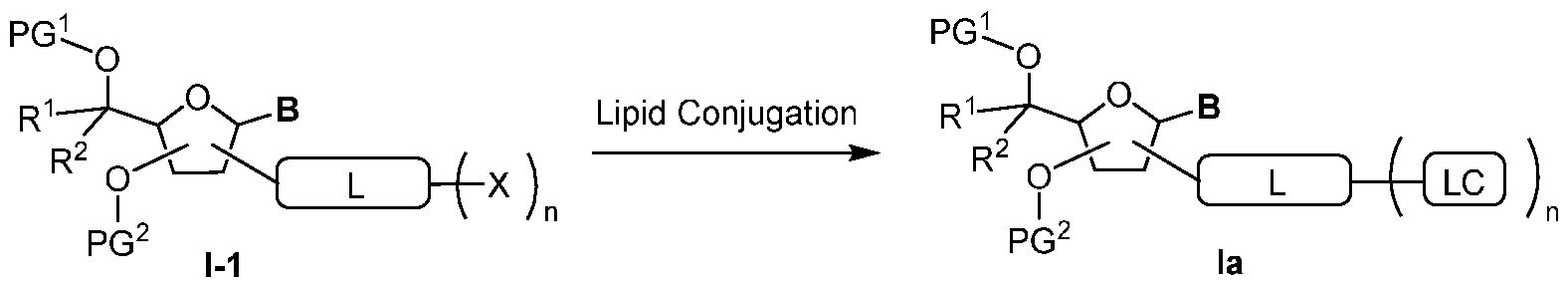
Attorney Docket: DCY-13025 RNAi OLIGONUCLEOTIDE CONJUGATES CROSS-REFERENCES TO RELATED APPLICATIONS This application claims the benefit of U.S. Provisional Patent Application No. 63/537,400 filed September 8, 2023, the contents of which is incorporated by reference herein in its entirety. TECHNICAL FIELD The disclosure relates to oligonucleotides linked to ligands useful in the inhibition of target genes in a variety of tissues (e.g., extrahepatic tissue). Specifically, the present disclosure relates to oligonucleotide-ligand conjugates, methods to prepare them, their chemical configuration, and methods to modulate (e.g., inhibit or reduce) the expression of a target gene using the conjugated nucleic acids and oligonucleotides according to the description provided herein. The disclosure also provides pharmaceutically acceptable compositions comprising the conjugates of the present description and methods of using said compositions in the treatment of various diseases or disorders. BACKGROUND OF THE DISCLOSURE Regulation of gene expression by modified nucleic acids shows great potential as both a research tool in the laboratory and a therapeutic approach in the clinic. Several classes of oligonucleotide or nucleic acid-based therapeutics have been under the clinical investigation, including antisense oligonucleotides (ASO), short interfering RNA (siRNA), double-stranded nucleic acids (dsNA), aptamers, ribozymes, exon-skipping and splice-altering oligonucleotides, immunomodulatory oligonucleotides, mRNAs, and CRISPR. Chemical modifications in the relevant molecules to allow functionality in various tissues, organs and/or cell types play a key role in overcoming challenges of oligonucleotide therapeutics, including improving nuclease stability, RNA-binding affinity, and pharmacokinetics. Various chemical modification strategies for oligonucleotides have been developed in the past three decades including modification of the sugars, nucleobases, and phosphodiester backbone to improve and optimize performance and therapeutic efficacy (Deleavey and Darma, CHEM. BIOL.2012, 19(8):937-54; Wan and Seth, J. MED. CHEM.2016, 59(21):9645-67; and Egli and Manoharan, ACC. CHEM. RES.2019, 54(4):1036-47). Dicer processed RNAi technologies utilize short double-stranded RNA (dsRNA) of approximately 21 base pair length with a two nucleotide (nt) 3’-overhang for the silencing of
FH12501620.1
Attorney Docket: DCY-13025 genes. These dsRNAs are generally called small interfering RNA (siRNA). siRNA 12 to 22 nucleotides in length are the active agent in RNAi. The siRNA duplex serves as a guide for mRNA degradation. Upon siRNA incorporation into the RNA-induced silencing complex (RISC) the complex interacts with a specific mRNA and ultimately suppresses the mRNA signal. The sense strand or passenger strand of siRNA is typically cleaved at the 9th nucleotide downstream from the 5’-end of the sense strand by Argonaute 2 (Ago2) endonuclease. The activated RISC complex containing the antisense strand or guide strand binds to the target mRNA through Watson–Crick base pairing causing degradation or translational blocking of the targeted RNA. However, the in vivo use of RNAi or siRNA molecules as pharmaceuticals has remained difficult due to obstacles encountered such as low biostability and unacceptable toxicity possibly caused by off-target effects. Various types of chemical modifications to improve the pharmacokinetics and to overcome bio-instability problems have been investigated over the years to improve the stability and specificity of the RNAi duplexes. In some cases, the chemical modification in siRNAs has improved the serum stability of siRNAs. However, often RNAi activity was lost, but the careful placement of some specific modified residues enables enhanced siRNA biostability without loss of siRNA potency. Some of these modifications have reduced siRNA side effects, such as the induction of recipient immune responses and inherent off-targeting effects and have even enhanced siRNA potency. Various chemically modified siRNAs have been investigated, among them were bridged nucleic acids (BNA’s) such as 2’,4’- methylene bridged nucleic acid 2’,4’-BNAs, also known as locked nucleic acid or LNA’s. Some of these modified siRNAs showed promising effects. Therapeutic gene silencing mediated by RNAi oligonucleotide-based therapeutics comprising siRNAs or double-stranded nucleic acids (dsNAs) offer the potential for considerable expansion of the druggable target space and the possibility for treating orphan diseases that may be therapeutically unapproachable by other drug modalities (e.g., antibodies and/or small molecules). RNAi oligonucleotide-based therapeutics that inhibit or reduce expression of specific target genes in the liver have been developed and are currently in clinical use (Sehgal et al., (2013) JOURNAL OF HEPATOLOGY 59:1354-59). Technological hurdles remain for the development and clinical use of RNAi oligonucleotides in extrahepatic cells, tissues, and organs. Thus, an ongoing need exists in the art for the successful development of new and effective RNAi oligonucleotides to modulate the expression of a target genes in extrahepatic cells, tissues, and/or organs. This is complicated by the variant nature of the cell types in extrahepatic as well as concerns about circulatory patterns and cell membrane
FH12501620.1
Attorney Docket: DCY-13025 constituents such as receptor types. Over the past decade, synthetic RNAi triggers such as double stranded RNAs have become ubiquitous tools in biological research, and extensive basic and clinical development efforts have recently culminated in the FDA approval of ONPATTRO
tm, the first RNAi drug. Despite a burgeoning drug development pipeline and an extensive compendium of excipients targeting ligands and delivery techniques, the difficulty of delivering RNAi agents to specific populations of disease related cells and or tissues, particularly outside the liver continues to limit the potential of RNAi therapy. Repeated attempts over the past several years to develop useful, active, and persistent RNAi agents and structures for use based on known liver delivery technology have not convincingly demonstrated the intended effects outside the liver. Thus, new dsRNA’s with variant structures have been developed to overcome the limitations in the field. BRIEF SUMMARY OF THE DISCLOSURE The present disclosure is based, in part, on the discovery of RNAi oligonucleotides capable of inhibiting expression of a target gene in extrahepatic tissues while having reduced inhibition in hepatocytes. As demonstrated herein, RNAi oligonucleotides having a ligand comprising a carboxyl conjugated to a nucleotide of the sense strand showed improved efficacy and duration in extrahepatic tissues, including adipose tissue, relative to hepatocytes. Specifically, in some embodiments, target gene expression was reduced in extrahepatic tissue by the RNAi oligonucleotides at a higher amount than reduction of expression of the same target gene in hepatocytes, e.g., reduction by greater than 50% in extrahepatic tissue compared to reduction by 25% in hepatocytes. Accordingly, in some aspects, the disclosure provides a double-stranded oligonucleotide comprising: (i) an antisense strand of 15 to 30 nucleotides in length, a sense strand of 13 to 40 nucleotides in length, wherein the antisense strand has a region of complementarity to a target sequence in a target mRNA in an extrahepatic tissue, and wherein the region of complementarity is at least 15 contiguous nucleotides in length; and (ii) one or more ligands, each ligand comprising one or more -L-Y-(CO2H)n groups, or a charged form thereof, wherein L is a linker, Y is alkylene, alkenylene, or alkynylene, and n is 1-6, wherein when Y is alkylene, L comprises -O(CH2)a(OCH2CH2)bNH-, wherein a is 1- 6 and b is 0-5, and wherein the ligand is conjugated to a nucleotide of the sense strand.
FH12501620.1
Attorney Docket: DCY-13025 In some aspects, the disclosure provides a double-stranded oligonucleotide comprising: (i) a double-stranded oligonucleotide comprising an antisense strand of 15 to 30 nucleotides in length, a sense strand of 15 to 40 nucleotides in length, wherein the antisense strand has a region of complementarity to a target sequence in a target mRNA in extrahepatic tissue, and wherein the region of complementarity is at least 15 contiguous nucleotides in length; and (ii) one or more ligands, each ligand comprising one or more C6-24 alkylene-(CO2H)n, or a charged form thereof, conjugated to a nucleotide of the sense strand, wherein n is 1-6, wherein knock down of the target mRNA in the extrahepatic tissue is greater than knock down of the target mRNA in liver tissue. In some embodiments, the disclosure provides a double-stranded oligonucleotide comprising: (i) an antisense strand of 15 to 30 nucleotides in length, a sense strand of 13 to 40 nucleotides in length, wherein the antisense strand has a region of complementarity to a target sequence in a target mRNA in an extrahepatic tissue, and wherein the region of complementarity is at least 15 contiguous nucleotides in length; and (ii) one or more ligands, each ligand comprising one or more -L-Y-W
n groups, or a charged form thereof, wherein L is a linker, Y is alkylene, alkenylene, or alkynylene, and n is 1-6, wherein when Y is alkylene, L comprises -O(CH2)a(OCH2CH2)bNH- or -O(CH2)a(OCH2CH2)b(NH)(CO)-, wherein a is 1-6 and b is 0-5, and wherein W is carboxyl, dicarboxyl, -SO2OH, tetrazolyl, -OH, -N(H)SO2(alkyl), -N(H)SO2(cycloalkyl), -N(H)SO2(aryl), -N(H)SO2(heterocycle), -N(H)SO2(heteroaryl), -O-Benzyl- S(O2NH2), -N(H)C(O)N(H)SO
2(alkyl), -N(H)C(O)(phenol), -N(H)(phenol), or heterocycle, and wherein the ligand is conjugated to a nucleotide of the sense strand. In some embodiments, the disclosure provides a double-stranded oligonucleotide comprising: (i) a double-stranded oligonucleotide comprising an antisense strand of 15 to 30 nucleotides in length, a sense strand of 15 to 40 nucleotides in length, wherein the antisense strand has a region of complementarity to a target sequence in a target mRNA in extrahepatic tissue, and wherein the region of complementarity is at least 15 contiguous nucleotides in length; and
FH12501620.1
Attorney Docket: DCY-13025 (ii) one or more ligands, each ligand comprising one or more C
6-24 alkylene-W
n, or a charged form thereof, conjugated to a nucleotide of the sense strand, wherein n is 1-6, wherein W is carboxyl, dicarboxyl, -SO
2OH, tetrazolyl, -OH, -N(H)SO
2(alkyl), -N(H)SO
2(cycloalkyl), -N(H)SO
2(aryl), -N(H)SO2(heterocycle), -N(H)SO2(heteroaryl), -O-Benzyl- S(O2NH2), -N(H)C(O)N(H)SO2(alkyl), -N(H)C(O)(phenol), -N(H)(phenol), heterocycle, and wherein knock down of the target mRNA in the extrahepatic tissue is greater than knock down of the target mRNA in liver tissue. In certain embodiments, n is 1. In certain such embodiments, W is preferably attached to the terminus of the Y group. For example, in certain such embodiments, W is preferably attached to the terminus of an alkylene group. In alternative embodiments, n is 2. In certain such embodiments, one occurrence of W is preferably attached to the terminus of the Y group. For example, in certain such embodiments, one occurrence of W is preferably attached to the terminus of an alkylene group. The other occurrence of W is attached elsewhere in the Y group, most preferably also at the terminus of the Y group, or one atom removed from the terminus of the Y group. In some or any of the foregoing or related embodiments, each ligand comprises a linker (L), wherein L is conjugated to the one or more C
6-24 alkylene-CO
2H. In some embodiments, L comprises a bivalent or trivalent C1-C50 alkylene, wherein 1-25 methylene groups are optionally and independently replaced by -N(H)-, -N(C1-C4 alkyl)-, - N(cycloalkyl)-, -O-, -C(O)-, -C(O)O-, -S-, -S(O)-, -S(O)2-, -S(O)2N(C1-C4 alkyl)-, - S(O)2N(cycloalkyl)-, -N(H)C(O)-, -N(C1-C4 alkyl)C(O)-, -N(cycloalkyl)C(O)-, -C(O)N(H)-, -C(O)N(C1-C4 alkyl), -C(O)N(cycloalkyl), aryl, heteroaryl, cycloalkyl, or cycloalkenyl. In some or any of the foregoing or related embodiments, 1-25 methylene groups are optionally and independently replaced by -N(H)-, -N(H)C(O)-, -N(C
1-C
4 alkyl)C(O)-, -O-, or heteroaryl. In some embodiments, the heteroaryl is a triazolyl. In some embodiments, the triazolyl
In some or any of the foregoing or related embodiments, Y is an C
6-24 alkylene, C
6-24 alkenylene, or C
6-24 alkynylene. In some embodiments, Y is an C
6-24 alkylene or C
6-24 alkenylene. In some embodiments, Y is a C
6-24 alkenylene. In some embodiments, Y is a C
6-20 alkylene.
FH12501620.1
Attorney Docket: DCY-13025 In some or any of the foregoing or related embodiments, the alkenylene comprises from 1-6 olefinic bonds. In some or any of the foregoing or related embodiments, the alkynylene comprises from 1-6 acetylenic bonds. In some or any of the foregoing or related embodiments, a is 1. In some or any of the foregoing or related embodiments, b is 0 or 1. In some or any of the foregoing or related embodiments, the -L-Y-(CO2H)n group is:
, wherein: M is absent, -NRC(O)-, or heteroaryl, wherein R is H or alkyl. c is 0, 1, 2, 3, 4, 5, or 6; d is 0, 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, or 20; and e is 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, or 23. In some embodiments, M is -NRC(O)-. In some embodiments, R is H or methyl. In some embodiments, R is H. In some embodiments, M is heteroaryl. In some embodiments, the

heteroaryl is triazolyl. In some embodiments, the triazolyl is . In some embodiments, M is absent. In some embodiments, c is 1. In some or any of the foregoing or related embodiments, d is 0 to 12. In some embodiments, d is 4 to 12. In some embodiments, d is 4 or 12. In some embodiments, d is 0. In some or any of the foregoing or related embodiments, e is 12 to 23. In some embodiments, e is 11, 14, 15, 20, or 21. In some or any of the foregoing or related embodiments, the extrahepatic tissue is adipose tissue, heart tissue, skeletal muscle, or adrenal gland tissue. In some embodiments, the extrahepatic tissue is adipose tissue. In some or any of the foregoing or related embodiments, the antisense strand is 22 nucleotides. In some or any of the foregoing or related embodiments, the antisense strand comprises a 3’ overhang of 2 to 6 nucleotides. In some or any of the foregoing or related embodiments, the sense strand and/or antisense strand comprises at least one modified internucleotide linkage. In some embodiments, the at least one modified internucleotide linkage is a phosphorothioate linkage.
FH12501620.1
Attorney Docket: DCY-13025 In some embodiments, the antisense strand comprises a phosphorothioate linkage (i) between positions 1 and 2, and between positions 2 and 3; or (ii) between positions 1 and 2, between positions 2 and 3, and between positions 3 and 4, wherein positions are numbered 1-4 from 5’ to 3’. In some embodiments, the antisense strand is 22 nucleotides in length, and wherein the antisense strand comprises a phosphorothioate linkage between positions 20 and 21 and between positions 21 and 22, wherein positions are numbered 1-22 from 5^ to 3^. In some embodiments, the sense strand comprises a phosphorothioate linkage (i) between positions 1 and 2; (ii) between positions 2 and 3; (iii) between positions 1 and 2, between positions 18 and 19, and between positions 19 and 20; or, (iv) between positions 2 and 3, between positions 18 and 19, and between positions 19 and 20, wherein positions are numbered 1-20 from 5^ to 3^. In some embodiments, the sense strand is 36 nucleotides in length, and wherein the sense strand comprises a phosphorothioate linkage between positions 1 and 2, wherein positions are numbered 1-36 from 5^ to 3^. In some embodiments, the sense strand is 20 nucleotides in length, and wherein the sense strand comprises a phosphorothioate linkage between positions 1 and 2, between positions 18 and 19, and between positions 19 and 20, wherein positions are numbered 1-20 from 5^ to 3^. In some or any of the foregoing or related embodiments, the oligonucleotide comprises a stem-loop comprising a tetraloop. In some embodiments, the tetraloop comprises a 5’-GAAA-3’ sequence. In some or any of the foregoing or related embodiments, the sense strand and/or antisense strand comprises one or more modified nucleotides. In some or any of the foregoing or related embodiments, the modified nucleotide comprises a modified sugar. In some embodiments, the modified sugar comprises a 2’-OMe group. In some embodiments, the modified sugar comprises a 2’-F substituent. In some or any of the foregoing or related embodiments, the antisense strand comprises up to 4 contiguous nucleotides with a 2’-F modified sugar. In some embodiments, the antisense strand comprises 22 nucleotides, wherein nucleotides at each of positions 2, 3, 4, 5, 7, 10, and 14, numbered 5^ to 3^ comprise a 2’-F modification. In some embodiments, the antisense strand comprises 22 nucleotides, wherein nucleotides at each of positions 2, 3, 4, 5, 7, 10, 14, 16, and 19, numbered 5^ to 3^, comprise a 2’-F modification. In some or any of the foregoing or related embodiments, the sense strand comprises 20 nucleotides, wherein nucleotides at each of positions 8, 9, 10, and 11 numbered 5^ to 3^, comprise a 2’-F modification. In some embodiments, the sense strand comprises 36
FH12501620.1
Attorney Docket: DCY-13025 nucleotides, wherein nucleotides at each of positions 3, 5, 8, 10, 12, 13, 15, and 17, numbered 5^ to 3^, comprise a 2’-F modification. In some or any of the foregoing or related embodiments, the antisense strand comprises a 4’-O-monomethylphosphonate-2’-O-methyl modified nucleotide. In some or any of the foregoing or related embodiments, the antisense strand comprises a phosphorylated nucleotide at the 5’ terminus, and wherein the phosphorylated nucleotide is selected from uridine and adenosine. In some or any of the foregoing or related embodiments, the 4^-carbon of the sugar of the 5^-nucleotide of the antisense strand comprises a phosphate analog. In some embodiments, the phosphate analog is oxymethyl phosphonate, vinyl phosphonate or malonyl phosphonate. In some embodiments, the phosphorylated nucleotide is 4’-O- monomethylphosphonate-2’-O-methyl uridine. In some or any of the foregoing or related embodiments, the one or more ligands is conjugated to a ribose of the sense strand at the 3’- or 5’-position. In some embodiments, the one or more ligands is conjugated to a ribose of the sense strand at the 2’-position. In some or any of the foregoing or related embodiments, one ligand is conjugated to position one of the sense strand, numbered 5^ to 3^. In some or any of the foregoing or related embodiments, the double-stranded oligonucleotide comprises at least two ligands. In some embodiments, the at least two ligands are conjugated to different nucleotides of the sense strand. In some embodiments, the at least two ligands are the same ligand. In some embodiments, the at least two ligands are different ligands. In some embodiments, the at least two ligands are conjugated to position 1, numbered 5^ to 3^, and a nucleotide within a tetraloop. In some or any of the foregoing or related embodiments, (a) at least one ligand is conjugated to position 1 and at least one ligand is conjugated to position 29; (b) at least one ligand is conjugated to position 1 and at least one ligand is conjugated to position 28; (c) at least one ligand is conjugated to position 1 and at least one ligand is conjugated to position 2; or (d) at least one ligand is conjugated to position 1 and at least one ligand is conjugated to position 20, numbered 5^ to 3^, of the sense strand.
FH12501620.1
Attorney Docket: DCY-13025 In some or any of the foregoing or related embodiments, the region of complementarity is fully complementary to the mRNA target sequence. In some embodiments, the region of complementarity is partially complementary to the mRNA target sequence. In some or any of the foregoing or related embodiments, the region of complementarity comprises no more than four mismatches to the mRNA target sequence. In some aspects, the disclosure provides an oligonucleotide-ligand conjugate of Formula (AI):

(AI), or a pharmaceutically acceptable salt or charged form thereof, wherein: A and A’ are each independently H or one or more nucleotides; B is a nucleobase; Z is O or S; M is absent, NRC(O), or heteroaryl, wherein R is H or alkyl. a is 1, 2, 3, 4, 5, or 6; b is 1, 2, 3, 4, 5, or 6; c is 0, 1, 2, 3, 4, 5, or 6; d is 0, 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, or 20; e is 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, or 23; and f is 0, 1, 2, 3, 4, 5, or 6. In some or any of the foregoing or related embodiments, M is -NRC(O)-. In some or any of the foregoing or related embodiments, R is H or C1-5 alkyl. In some embodiments, R is H or methyl. In some embodiments, R is H. In some or any of the foregoing or related embodiments, c is 1.
FH12501620.1
Attorney Docket: DCY-13025 In some or any of the foregoing or related embodiments, the oligonucleotide-ligand conjugate has a structure of Formula (AII)
(AII), or a pharmaceutically acceptable salt or charged form thereof. In some or any of the foregoing or related embodiments, M is heteroaryl. In some
embodiments, the heteroaryl is triazolyl. In some embodiments, the triazolyl is . In some or any of the foregoing or related embodiments, the oligonucleotide-ligand conjugate has a structure of Formula (AIII)
(AIII), or a pharmaceutically acceptable salt or charged form thereof. In some or any of the foregoing or related embodiments, d is 0 to 12. In some embodiments d is 4 to 12. In some embodiments, d is 4 or 12. In some embodiments, d is 0. In some or any of the foregoing or related embodiments, the oligonucleotide-ligand conjugate has a structure of Formula (AIV)
FH12501620.1
Attorney Docket: DCY-13025
(AIV), or a pharmaceutically acceptable salt or a charged form thereof. In some or any of the foregoing or related embodiments, M is absent. In some or any of the foregoing or related embodiments, the oligonucleotide-ligand conjugate has a structure of Formula (AV)
(AV), or a pharmaceutically acceptable salt or a charged form thereof, wherein e is 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, or 23. In some or any of the foregoing or related embodiments, a is 1 or 2. In some embodiments, a is 1. In some or any of the foregoing or related embodiments, b is 1 or 2. In some embodiments, b is 1. In some or any of the foregoing or related embodiments, e is 11 to 22. In some embodiments, e is 11, 15, or 21. In some or any of the foregoing or related embodiments, f is 0, 1, or 2. In some embodiments, f is 0. In some or any of the foregoing or related embodiments, B is
FH12501620.1
Attorney Docket: DCY-13025
embodiments,

In some or any of the foregoing or related embodiments, A is H. In some or any of the foregoing or related embodiments, Z is S. In some aspects, the disclosure provides a double-stranded oligonucleotide comprising a sense strand and an antisense strand, wherein the sense strand comprises the oligonucleotide-ligand conjugate of some or any of the foregoing or related embodiments, and wherein the sense strand and antisense strand form a duplex region. In some or any of the foregoing or related embodiments, the antisense strand is 15 to 30 nucleotides in length. In some or any of the foregoing or related embodiments, the sense strand and/or antisense strand comprises at least one modified internucleotide linkage. In some embodiments, the at least one modified internucleotide linkage is a phosphorothioate linkage. In some or any of the foregoing or related embodiments, A is H and A^ is 12 to 40 nucleotides. In some or any of the foregoing or related embodiments, the nucleotides of A^ comprise at least one modified internucleotide linkage. In some or any of the foregoing or related embodiments, A is 12-29 nucleotides and A^ is 1-10 nucleotides. In some or any of the foregoing or related embodiments, the nucleotides of A comprise at least one modified internucleotide linkage. In some or any of the foregoing or related embodiments, the oligonucleotide-ligand conjugate and at least 14 nucleotides of A’ form the duplex region with the antisense strand. In some or any of the foregoing or related embodiments, the antisense strand comprises a phosphorothioate linkage (i) between positions 1 and 2, and between positions 2 and 3; or (ii) between positions 1 and 2, between positions 2 and 3, and between positions 3 and 4, wherein positions are numbered 1-4 from 5’ to 3’. In some embodiments, the antisense strand is 22 nucleotides in length, and wherein the antisense strand comprises a
FH12501620.1
Attorney Docket: DCY-13025 phosphorothioate linkage between positions 20 and 21 and between positions 21 and 22, wherein positions are numbered 1-22 from 5^ to 3^. In some embodiments, the sense strand comprises a phosphorothioate linkage (i) between positions 1 and 2; (ii) between positions 2 and 3; (iii) between positions 1 and 2, between positions 18 and 19, and between positions 19 and 20; or, (iv) between positions 2 and 3, between positions 18 and 19, and between positions 19 and 20, wherein positions are numbered 1-20 from 5^ to 3^. In some embodiments, the sense strand is 36 nucleotides in length, and wherein the sense strand comprises a phosphorothioate linkage between positions 1 and 2, wherein positions are numbered 1-36 from 5^ to 3^. In some embodiments, the sense strand is 20 nucleotides in length, and wherein the sense strand comprises a phosphorothioate linkage between positions 1 and 2, between positions 18 and 19, and between positions 19 and 20, wherein positions are numbered 1-20 from 5^ to 3^. In some or any of the foregoing or related embodiments, the duplex region comprises 20 to 30 base pairs. In some or any of the foregoing or related embodiments, the sense strand comprises a stem-loop comprising a tetraloop. In some embodiments, the tetraloop comprises a 5’- GAAA-3’ sequence. In some embodiments, the oligonucleotide-ligand conjugate is a nucleotide of the tetraloop. In some or any of the foregoing or related embodiments, the sense and antisense strand comprise one or more modified nucleotides. In some embodiments, the modified nucleotide comprises a modified sugar. In some embodiments, the modified sugar comprises a 2’-OMe group. In some embodiments, the modified sugar comprises a 2’-F substituent. In some or any of the foregoing or related embodiments, the sense strand comprises 20 nucleotides, wherein nucleotides at each of positions 8, 9, 10, and 11 comprise a 2’-F modification. In some embodiments, the sense strand comprises 36 nucleotides, wherein nucleotides at each of positions 3, 5, 8, 10, 12, 13, 15, and 17 comprise a 2’-F modification. In some embodiments, the antisense strand comprises 22 nucleotides, wherein nucleotides at each of positions 2, 3, 4, 5, 7, 10, and 14 comprise a 2’-F modification. In some embodiments, the antisense strand comprises 22 nucleotides, wherein nucleotides at each of positions 2, 3, 4, 5, 7, 10, 14, 16, and 19 comprise a 2’-F modification. In some or any of the foregoing or related embodiments, the modified nucleotide comprises a modified nucleobase.
FH12501620.1
Attorney Docket: DCY-13025 In some or any of the foregoing or related embodiments, the double-stranded oligonucleotide comprises a 4’-O-monomethylphosphonate-2’-O-methyl modified nucleotide. In some or any of the foregoing or related embodiments, the antisense strand comprises a phosphorylated nucleotide at the 5’ terminus, and wherein the phosphorylated nucleotide is selected from uridine and adenosine. In some or any of the foregoing or related embodiments, the 4^-carbon of the sugar of the 5^-nucleotide of the antisense strand comprises a phosphate analog. In some embodiments, the phosphate analog is oxymethyl phosphonate, vinyl phosphonate or malonyl phosphonate. In some or any of the foregoing or related embodiments, wherein the phosphorylated nucleotide is 4’-O-monomethylphosphonate-2’-O-methyl uridine. In some aspects, the disclosure provides an oligonucleotide-ligand conjugate of Formula (BI)

(BI), or a pharmaceutically acceptable salt or charged form thereof, wherein: B is a nucleobase; A is a one or more nucleotides; Z
1 and Z
2 are each independently O or S; M is absent, NRC(O), or heteroaryl, wherein R is H or alkyl; a is 1, 2, 3, 4, or 5; c is 0, 1, 2, 3, 4, 5, or 6; d is 0, 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, or 20; e is 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, or 23; and f is 0, 1, 2, 3, 4, 5, or 6.
FH12501620.1
Attorney Docket: DCY-13025 In some aspects, the disclosure provides an oligonucleotide-ligand conjugate of Formula (BII)
(BII), or a salt or charged form thereof, wherein: B is a nucleobase; A is a one or more nucleotides ; Z is O or S; M is absent, NRC(O), or heteroaryl, wherein R is H or alkyl; a is 1, 2, 3, 4, or 5; c is 0, 1, 2, 3, 4, 5, or 6; d is 0, 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, or 20; e is 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, or 23; and f is 0, 1, 2, 3, 4, 5, or 6. In some aspects, the disclosure provides an oligonucleotide-ligand conjugate of Formula (CI)
(CI), or a pharmaceutically acceptable salt or charged form thereof, wherein: B is a nucleobase; A is one or more nucleotides; Z
1 and Z
2 are each independently O or S; M is absent, NRC(O), or heteroaryl, wherein R is H or alkyl;
FH12501620.1
Attorney Docket: DCY-13025 a is 1, 2, 3, 4, or 5; c is 0, 1, 2, 3, 4, 5, or 6; d is 0, 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, or 20; e is 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, or 23; and f is 0, 1, 2, 3, 4, 5, or 6. In some aspects, the disclosure provides an oligonucleotide-ligand conjugate of Formula (CII)

(CII), or a salt or charged form thereof, wherein: B is a nucleobase; A is one or more nucleotides; Z is O or S; M is absent, NRC(O), or heteroaryl, wherein R is H or alkyl; a is 1, 2, 3, 4, or 5; c is 0, 1, 2, 3, 4, 5, or 6; d is 0, 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, or 20; e is 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, or 23; and f is 0, 1, 2, 3, 4, 5, or 6. In some aspects, the disclosure provides a double-stranded oligonucleotide comprising a sense strand and an antisense strand, wherein the sense strand comprises an oligonucleotide-ligand conjugate of some or any of the foregoing or related embodiments, and wherein the sense strand and antisense strand form a duplex region. In some or any of the foregoing or related embodiments, the antisense strand is 15 to 30 nucleotides in length. In some or any of the foregoing or related embodiments, A is 12 to 40 nucleotides. In some or any of the foregoing or related embodiments, the sense strand and/or antisense strand comprises at least one modified internucleotide linkage. In some embodiments, the at least one modified internucleotide linkage is a phosphorothioate linkage.
FH12501620.1
Attorney Docket: DCY-13025 In some embodiments, the duplex region includes one or more phosphorothioate linkages. In some embodiments, two phosphorothioate linkages are adjacent to each other. In some embodiments, the antisense strand comprises a phosphorothioate linkage (i) between positions 1 and 2, and between positions 2 and 3; or (ii) between positions 1 and 2, between positions 2 and 3, and between positions 3 and 4, wherein positions are numbered 1-4 from 5’ to 3’. In some embodiments, the antisense strand is 22 nucleotides in length, and wherein the antisense strand comprises a phosphorothioate linkage between positions 20 and 21 and between positions 21 and 22, wherein positions are numbered 1-22 from 5^ to 3^. In some embodiments, the sense strand comprises a phosphorothioate linkage (i) between positions 1 and 2; (ii) between positions 2 and 3; (iii) between positions 1 and 2, between positions 18 and 19, and between positions 19 and 20; or, (iv) between positions 2 and 3, between positions 18 and 19, and between positiosn 19 and 20, wherein positions are numbered 1-20 from 5^ to 3^. In some embodiments, the sense strand is 36 nucleotides in length, and wherein the sense strand comprises a phosphorothioate linkage between positions 1 and 2, wherein positions are numbered 1-36 from 5^ to 3^. In some embodiments, the sense strand is 20 nucleotides in length, and wherein the sense strand comprises a phosphorothioate linkage between positions 1 and 2, between positions 18 and 19, and between positions 19 and 20, wherein positions are numbered 1-20 from 5^ to 3^. In some or any of the foregoing or related embodiments, the duplex region comprises 20 to 30 base pairs. In some or any of the foregoing or related embodiments, the sense strand comprises a loop region that includes a tetraloop region. In some embodiments, the tetraloop region comprises a 5’-GAAA-3’ sequence. In some or any of the foregoing or related embodiments, the sense and antisense strand comprise one or more modified nucleotides. In some embodiments, the modified nucleotide comprises a modified sugar. In some embodiments, the modified sugar comprises a 2’-OMe group. In some embodiments, the modified sugar comprises a 2’-F substituent. In some or any of the foregoing or related embodiments, the sense strand comprises 20 nucleotides, wherein nucleotides at each of positions 8, 9, 10, and 11 comprise a 2’-F modification. In some embodiments, the sense strand comprises 36 nucleotides, wherein nucleotides at each of positions 3, 5, 8, 10, 12, 13, 15, and 17 comprise a 2’-F modification. In some embodiments, the antisense strand comprises 22 nucleotides, wherein nucleotides at each of positions 2, 3, 4, 5, 7, 10, and 14 comprise a 2’-F modification. In some
FH12501620.1
Attorney Docket: DCY-13025 embodiments, the antisense strand comprises 22 nucleotides, wherein nucleotides at each of positions 2, 3, 4, 5, 7, 10, 14, 16, and 19 comprise a 2’-F modification. In some or any of the foregoing or related embodiments, the modified nucleotide comprises a modified nucleobase. In some or any of the foregoing or related embodiments, the double-stranded oligonucleotide comprises a 4’-O-monomethylphosphonate-2’-O-methyl modified nucleotide. In some or any of the foregoing or related embodiments, the antisense strand comprises a phosphorylated nucleotide at the 5’ terminus, and wherein the phosphorylated nucleotide is selected from uridine and adenosine. In some or any of the foregoing or related embodiments, the 4^-carbon of the sugar of the 5^-nucleotide of the antisense strand comprises a phosphate analog. In some embodiments, the phosphate analog is oxymethyl phosphonate, vinyl phosphonate or malonyl phosphonate. In some embodiments, the phosphorylated nucleotide is 4’-O- monomethylphosphonate-2’-O-methyl uridine. In some or any of the foregoing or related embodiments, the sense strand comprises a second oligonucleotide-ligand conjugate. In some embodiments, the second oligonucleotide- ligand conjugate is selected from the oligonucleotide-ligand conjugate of some or any of the foregoing or related embodiments. In some or any of the foregoing or related embodiments, the antisense strand has a region of complementarity to a target sequence in a target mRNA in extrahepatic tissue, wherein the region of complementarity is at least 15 contiguous nucleotides in length. In some embodiments, the region of complementarity is fully complementary to the mRNA target sequence. In some embodiments, the region of complementarity is partially complementary to the mRNA target sequence. In some embodiments, the region of complementarity comprises no more than four mismatches to the mRNA target sequence. In some or any of the foregoing or related embodiments, the extrahepatic tissue is adipose tissue, heart tissue, skeletal muscle, or adrenal gland tissue. In some embodiments, the extrahepatic tissue is adipose tissue. In some aspects, the disclosure provides a double-stranded oligonucleotide comprising a sense strand and an antisense strand, wherein the sense strand comprises two or more oligonucleotide-ligand conjugates of (i), (ii), or (iii): (i) Formula (AI), (AII), (AIII), (AIV), or (AV); (ii) Formula (BI) or (BII); and
FH12501620.1
Attorney Docket: DCY-13025 (iii) Formula (CI) or (CII). In some or any of the foregoing or related embodiments, the two or more oligonucleotide ligand conjugates are conjugated to different nucleotides of the sense strand. In some embodiments, the two or more oligonucleotide ligand conjugates are the same. In some embodiments, the two or more oligonucleotide ligand conjugates are different. In some or any of the foregoing or related embodiments, the sense strand comprises a first oligonucleotide-ligand conjugate and a second oligonucleotide-ligand conjugate. In some or any of the foregoing or related embodiments, the nucleobase (B) of the first oligonucleotide-ligand conjugate is the nucleobase at position 1 of the sense strand and the nucleobase (B) of the second oligonucleotide-ligand conjugate is a nucleobase within a tetraloop of the sense strand. In some or any of the foregoing or related embodiments, (a) the nucleobase (B) of the first oligonucleotide-ligand conjugate is the nucleobase at position 1 of the sense strand, and the nucleobase (B) of the second oligonucleotide-ligand conjugate is the nucleobase at position 29 of the sense strand; (b) the nucleobase (B) of the first oligonucleotide-ligand conjugate is the nucleobase at position 1 of the sense strand, and the nucleobase (B) of the second oligonucleotide-ligand conjugate is the nucleobase at position 28 of the sense strand; (c) the nucleobase (B) of the first oligonucleotide-ligand conjugate is the nucleobase at position 1 of the sense strand, and the nucleobase (B) of the second oligonucleotide-ligand conjugate is the nucleobase at position 20 of the sense strand; or (d) the nucleobase (B) of the first oligonucleotide-ligand conjugate is the nucleobase at position 1 of the sense strand, and the nucleobase (B) of the second oligonucleotide-ligand conjugate is the nucleobase at position 2 of the sense strand. In some or any of the foregoing or related embodiments, the oligonucleotide-ligand conjugate reduces expression of the target mRNA in an extrahepatic tissue, provided the oligonucleotide-ligand conjugate does not reduce expression of the mRNA target in the liver. In some aspects, the disclosure provides a pharmaceutical composition comprising the double-stranded oligonucleotide of some or any of the foregoing or related embodiments, and a pharmaceutically acceptable carrier, delivery agent, or excipient. In some aspects, the disclosure provides a method of inhibiting target mRNA expression in an extrahepatic cell or tissue in a subject, comprising administering to the subject the double-stranded oligonucleotide or the pharmaceutical composition of some or
FH12501620.1
Attorney Docket: DCY-13025 any of the foregoing or related embodiments, thereby inhibiting target mRNA expression in the cell of the extrahepatic tissue. In some or any of the foregoing or related embodiments, the extrahepatic cell or tissue is selected from skeletal muscle, adipose tissue, adrenal tissue, heart tissue, and any combination thereof. In some or any of the foregoing or related embodiments, reduction of the target mRNA in the extrahepatic cell or tissue is increased compared to reduction in liver cells or tissue, optionally wherein reduction of the target mRNA is increased by at least 10%. In some or any of the foregoing or related embodiments, reduction of the target mRNA is increased by at least 15%, at least 20%, at least 25%, at least 30%, at least 35%, at least 40%, at least 45% or at least 50%. In some aspects, the disclosure provides the double-stranded oligonucleotide of some or any of the foregoing or related embodiments in the manufacture of a medicament for inhibiting target mRNA expression in an extrahepatic cell or tissue in a subject. In some aspects, the disclosure provides for use of the double-stranded oligonucleotide of some or any of the foregoing or related embodiments for inhibiting target mRNA expression in a cell of an extrahepatic tissue in a subject. In some aspects, the disclosure provides a kit comprising a container comprising the double-stranded oligonucleotide of some or any of the foregoing or related embodiments, and optionally a pharmaceutically acceptable carrier, and instructions for administering the oligonucleotide-ligand conjugate to a subject in need thereof, wherein the oligonucleotide- ligand conjugate inhibits target mRNA expression in an extrahepatic cell or tissue in the subject. In some or any of the foregoing or related embodiments, the extrahepatic cell or tissue is selected from skeletal muscle, adipose tissue, adrenal tissue, heart tissue, and any combination thereof. In some embodiments, the cell of the extrahepatic cell or tissue is selected from a cardiomyocyte, a cell of skeletal muscle, a cell of adipose tissue, a cell of adrenal tissue, and any combination thereof. In some or any of the foregoing or related embodiments, reduction of the target mRNA in the extrahepatic cell or tissue is increased compared to reduction in a cell of the liver, optionally wherein reduction of the target mRNA is increased by at least 10%. In some or any of the foregoing or related embodiments, reduction of the target mRNA is increased by at least 15%, at least 20%, at least 25%, at least 30%, at least 35%, at least 40%, at least 45% or at least 50%.
FH12501620.1
Attorney Docket: DCY-13025 In some aspects, the disclosure provides a method for treating a subject having a disease, disorder or condition associated with expression of an mRNA in an extrahepatic cell or tissue, the method comprising administering to the subject a therapeutically effective amount of the double-stranded oligonucleotide or pharmaceutical composition of any or some of the foregoing or related aspects. In some aspects, the disclosure provides a method of delivering a double-stranded oligonucleotide to a cell or population of cells in extrahepatic tissue, the method comprising administering the pharmaceutical composition of any or some of the foregoing or related aspects. In some aspects, the disclosure provides a double-stranded oligonucleotide comprising: (i) an antisense strand of 15 to 30 nucleotides in length, a sense strand of 13 to 40 nucleotides in length, wherein the antisense strand has a region of complementarity to a target sequence in a target mRNA in an extrahepatic tissue, and wherein the region of complementarity is at least 15 contiguous nucleotides in length; and (ii) one or more ligands, each ligand comprising one or more -L-Y-W
n groups, or a charged form thereof, wherein: L is a linker selected from -O(CH
2)
a(OCH
2CH
2)
bNH- , -O(CH2)a(OCH2CH2)b(NH)(CO)-, -O(CH2)a(OCH2CH2)b(NH)(CO)(CH2)c(OCH2CH2)dM-, -OP(=O)(OH)O(CH2)a(OCH2CH2)bNH-, -OP(=O)(OH)O(CH2)a(OCH2CH2)b(NH)(CO)-, -OP(=O)(OH)O(CH2)aM, - OP(=O)(OH)O(CH2)a(OCH2CH2)b(NH)(CO)(CH2)c(OCH2CH2)dM-, Y is alkylene, alkenylene, alkynylene, or (CH2)e(OCH2CH2)f; each W is independently -C(O)OH, -CR^(C(O)OH)2, -CR^(C(O)O(alkyl))2, -SO2OH, tetrazolyl, -OH, -N(H)SO
2(alkyl), -N(H)SO
2(cycloalkyl), -N(H)SO
2(optionally substituted aryl), -N(H)SO
2(heterocyclyl), -N(H)SO
2(optionally substituted heteroaryl), -O(optionally substituted aryl), -N(H)C(O)N(H)SO
2(alkyl), -C(O)NH(hydroxyphenyl), -N(H)C(O)(hydroxyphenyl), -N (H)(hydroxyphenyl), hydroxyphenyl, or heterocyclyl; M is absent, -NHC(O)-, -N(alkyl)C(O)-, or heteroarylene; R^ is H or methyl; n is an integer from 1-6; a is an integer from 1-6;
FH12501620.1
Attorney Docket: DCY-13025 b is an integer from 0-8; c is an integer from 0-6; d is an integer from 0-20; e is an integer from 1-6; f is an integer from 1-20; wherein when Y is alkylene, L comprises -O(CH2)a(OCH2CH2)bNH- or -O(CH2)a(OCH2CH2)b(NH)(CO)-; and wherein the ligand is conjugated to a nucleotide of the sense strand. In some aspects, the disclosure provides a double-stranded oligonucleotide comprising: (i) an antisense strand of 15 to 30 nucleotides in length, a sense strand of 13 to 40 nucleotides in length, wherein the antisense strand has a region of complementarity to a target sequence in a target mRNA in an extrahepatic tissue, and wherein the region of complementarity is at least 15 contiguous nucleotides in length; and (ii) one or more ligands, each ligand comprising one or more -L-Y-W
n groups, or a charged form thereof, wherein: L is a linker selected from -O(CH2)a(OCH2CH2)bNH- , -O(CH
2)
a(OCH
2CH
2)
b(NH)(CO)-, -O(CH
2)
a(OCH
2CH
2)
b(NH)(CO)(CH
2)
c(OCH
2CH
2)
dM-, -OP(=O)(OH)O(CH2)a(OCH2CH2)bNH-, -OP(=O)(OH)O(CH2)a(OCH2CH2)b(NH)(CO)-, -OP(=O)(OH)O(CH2)aM, - OP(=O)(OH)O(CH2)a(OCH2CH2)b(NH)(CO)(CH2)c(OCH2CH2)dM-, Y is alkylene, alkenylene, alkynylene, or (CH2)e(OCH2CH2)f; each W is independently -C(O)OH, -CR^(C(O)OH)2, -CR^(C(O)O(alkyl))2, -SO2OH, tetrazolyl, -OH, -N(H)SO2(alkyl), -N(H)SO2(cycloalkyl), -N(H)SO2(optionally substituted aryl), -N(H)SO
2(heterocyclyl), -N(H)SO
2(optionally substituted heteroaryl), -O(optionally substituted aryl), -N(H)C(O)N(H)SO
2(alkyl), -C(O)NH(hydroxyphenyl), -N(H)C(O)(hydroxyphenyl), -N (H)(hydroxyphenyl), hydroxyphenyl, or heterocyclyl; M is absent, -NHC(O)-, -N(alkyl)C(O)-, or heteroarylene; R^ is H or methyl; n is an integer from 1-6; a is an integer from 1-6; b is an integer from 0-8;
FH12501620.1
Attorney Docket: DCY-13025 c is an integer from 0-6; d is an integer from 0-20; e is an integer from 1-6; f is an integer from 1-20; and wherein the ligand is conjugated to a nucleotide of the sense strand. In some aspects, the disclosure provides double-stranded oligonucleotide comprising: (i) a double-stranded oligonucleotide comprising an antisense strand of 15 to 30 nucleotides in length, a sense strand of 15 to 40 nucleotides in length, wherein the antisense strand has a region of complementarity to a target sequence in a target mRNA in extrahepatic tissue, and wherein the region of complementarity is at least 15 contiguous nucleotides in length; and (ii) one or more ligands, each ligand comprising one or more -L-(C6-24 alkylene)-Wn, or a charged form thereof, conjugated to a nucleotide of the sense strand, wherein: L is a linker selected from -O(CH
2)
a(OCH
2CH
2)
bNH- , -O(CH
2)
a(OCH
2CH
2)
b(NH)(CO)-, -O(CH
2)
a(OCH
2CH
2)
b(NH)(CO)(CH
2)
c(OCH
2CH
2)
dM-, -OP(=O)(OH)O(CH
2)
a(OCH
2CH
2)
bNH-, -OP(=O)(OH)O(CH
2)
a(OCH
2CH
2)
b(NH)(CO)-, -OP(=O)(OH)O(CH2)aM, and -OP(=O)(OH)O(CH
2)
a(OCH
2CH
2)
b(NH)(CO)(CH
2)
c(OCH
2CH
2)
dM-, each W is independently -C(O)OH, -CR^(C(O)OH)2, -CR^(C(O)O(alkyl))2, -SO2OH, tetrazolyl, -OH, -N(H)SO2(alkyl), -N(H)SO2(cycloalkyl), -N(H)SO2(optionally substituted aryl), -N(H)SO2(heterocyclyl), -N(H)SO2(optionally substituted heteroaryl), -O(optionally substituted aryl), -N(H)C(O)N(H)SO2(alkyl), -C(O)NH(hydroxyphenyl), -N(H)C(O)(hydroxyphenyl), -N(H)(hydroxyphenyl), hydroxyphenyl, or heterocyclyl; M is absent, -NHC(O)-, -N(alkyl)C(O)-, or heteroarylene; R^ is H or methyl; n is an integer from 1-6; a is an integer from 1-6; b is an integer from 0-8; c is an integer from 0-6; d is an integer from 0-20; wherein knock down of the target mRNA in the extrahepatic tissue is greater than knock down of the target mRNA in liver tissue.
FH12501620.1
Attorney Docket: DCY-13025 In some or any of the foregoing or related embodiments, L is a linker selected from -O(CH2)a(OCH2CH2)bNH-, -O(CH2)a(OCH2CH2)b(NH)(CO)-, and -O(CH
2)
a(OCH
2CH
2)
b(NH)(CO)(CH
2)
c(OCH
2CH
2)
dM-. In some or any of the foregoing or related embodiments, L is -O(CH
2)
a(OCH
2CH
2)
b(NH)(CO)(CH
2)
c(OCH
2CH
2)
dM-. In some or any of the foregoing or related embodiments, when Y is alkylene, L comprises -O(CH2)a(OCH2CH2)bNH- or -O(CH2)a(OCH2CH2)b(NH)(CO)-. In some or any of the foregoing or related embodiments, M is heteroarylene. In some ebodiments, the heteroarylene is a triazolylene. In some or any of the foregoing or related embodiments, the triazolylene is
. In some or any of the foregoing or related embodiments, M is -NHC(O)-. In some or any of the foregoing or related embodiments, Y is an C
6-24 alkylene, C
6-24 alkenylene, or C6-24 alkynylene. In some or any of the foregoing or related embodiments, Y is an C6-24 alkylene or C6-24 alkenylene. In some embodiments, Y is a C6-24 alkenylene. In some ebodiments, Y is a C6-20 alkylene. In some embodiments, the alkenylene comprises from 1-6 olefinic bonds. In some embodiments, the alkynylene comprises from 1-6 acetylenic bonds. In some embodiments, the alkenylene comprises from 1-6 olefinic bonds; and the alkynylene comprises from 1-6 acetylenic bonds In some or any of the foregoing or related embodiments, n is 1. In some or any of the foregoing or related embodiments, W is -CO
2H,
, , , - OH, -N(H)SO2(methyl), -N(H)SO2(ethyl), -N(H)SO2(n-propyl), -N(H)SO2(i-propyl), -N(H)S O2(sec-propyl), -N(H)SO2(n-butyl), -N(H)SO2(i-butyl), -N(H)SO2(sec-butyl), -N(H)SO2(t- butyl), -N(H)SO2(n-pentyl), -N(H)SO2(cyclopropyl), -N(H)SO2(cyclobutyl), -N(H)SO2(cyclopentyl), -N(H)SO2(cyclohexyl), -N(H)SO2(cycloheptyl),
FH12501620.1
Attorney Docket: DCY-13025 ,
In some or any of the foregoing or related embodiments, W is piperazinyl or piperidinyl. In some or any of the foregoing or related embodiments, a is 1. In some or any of the foregoing or related embodiments, b is 0 or 1. In some or any of the foregoing or related embodiments, b is 2. In some or any of the foregoing or related embodiments, c is 2. In some or any of the foregoing or related embodiments, d is 4. In some or any of the foregoing or related embodiments, the -L-Y-(W)
n group is:
wherein g is 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, or 23. In some or any of the foregoing or related embodiments, M is -NHC(O)-. In some embodiments, M is -N(methyl)C(O). In some embodiments, M is heteroarylene. In some or any of the foregoing or related embodiments, the heteroaryl is triazolylene. In some embodiments, the triazolylene is
. In some or any of the foregoing or related embodiments, M is absent.
FH12501620.1
Attorney Docket: DCY-13025 In some or any of the foregoing or related embodiments, W is -CO2H,
, , , - OH, -N(H)SO
2(methyl), -N(H)SO
2(ethyl), -N(H)SO
2(n-propyl), -N(H)SO
2(i-propyl), -N( H)SO
2(sec-propyl), -N(H)SO
2(n-butyl), -N(H)SO
2(i-butyl), -N(H)SO
2(sec- butyl), -N(H)SO2(t-butyl), -N(H)SO2(n- pentyl), -N(H)SO2(cyclopropyl), -N(H)SO2(cyclobutyl), -N(H)SO2(cyclopentyl), -N(H)SO2(cyclohexyl), -N(H)SO2(cycloheptyl),
-N(H)SO
2(cyclooctyl), -N(H)SO
2([1.1.1]propellane), ,
In some or any of the foregoing or related embodiments, wherein W is piperazinyl or piperidinyl. In some or any of the foregoing or related embodiments, c is 1. In some or any of the foregoing or related embodiments, c is 2. In some or any of the foregoing or related embodiments, d is 0 to 12. In some embodiments, d is 4 to 12. In some embodiments, d is 4. In some or any of the foregoing or related embodiments, g is 12 to 23. In some embodiments, g is 11, 14, 15, 20, or 21.
FH12501620.1
Attorney Docket: DCY-13025 In some or any of the foregoing or related embodiments, the extrahepatic tissue is adipose tissue, heart tissue, skeletal muscle, or adrenal gland tissue. In some embodiments, the extrahepatic tissue is adipose tissue. In some or any of the foregoing or related embodiments, the antisense strand is 22 nucleotides. In some or any of the foregoing or related embodiments, the antisense strand comprises a 3’ overhang of 2 to 6 nucleotides. In some or any of the foregoing or related embodiments, the sense strand and/or antisense strand comprises at least one modified internucleotide linkage. In some embodiments, the at least one modified internucleotide linkage is a phosphorothioate linkage. In some embodiments, the antisense strand comprises a phosphorothioate linkage (i) between positions 1 and 2, and between positions 2 and 3; or (ii) between positions 1 and 2, between positions 2 and 3, and between positions 3 and 4, wherein positions are numbered 1-4 from 5’ to 3’. In some embodiments, antisense strand is 22 nucleotides in length, and wherein the antisense strand comprises a phosphorothioate linkage between positions 20 and 21 and between positions 21 and 22, wherein positions are numbered 1-22 from 5^ to 3^. In some embodiments, the sense strand comprises a phosphorothioate linkage (i) between positions 1 and 2; (ii) between positions 2 and 3; (iii) between positions 1 and 2, between positions 18 and 19, and between positions 19 and 20; or, (iv) between positions 2 and 3, between positions 18 and 19, and between positions 19 and 20, wherein positions are numbered 1-20 from 5^ to 3^. In some embodiments, the sense strand is 36 nucleotides in length, and wherein the sense strand comprises a phosphorothioate linkage between positions 1 and 2, wherein positions are numbered 1-36 from 5^ to 3^. In some embodiments, the sense strand is 20 nucleotides in length, and wherein the sense strand comprises a phosphorothioate linkage between positions 1 and 2, between positions 18 and 19, and between positions 19 and 20, wherein positions are numbered 1-20 from 5^ to 3^. In some or any of the foregoing or related embodiments, the oligonucleotide comprises a stem-loop comprising a tetraloop. In some embodiments, the tetraloop comprises a 5’-GAAA-3’ sequence. In some or any of the foregoing or related embodiments, the sense strand and/or antisense strand comprises one or more modified nucleotides. In some embodiments, the modified nucleotide comprises a modified sugar. In some embodiments, the modified sugar comprises a 2’-OMe group. In some embodiments, the modified sugar comprises a 2’-F substituent.
FH12501620.1
Attorney Docket: DCY-13025 In some or any of the foregoing or related embodiments, the antisense strand comprises up to 4 contiguous nucleotides with a 2’-F modified sugar. In some embodiments, the antisense strand comprises 22 nucleotides, wherein nucleotides at each of positions 2, 3, 4, 5, 7, 10, and 14, numbered 5^ to 3^ comprise a 2’-F modification. In some embodiments, wherein the antisense strand comprises 22 nucleotides, wherein nucleotides at each of positions 2, 3, 4, 5, 7, 10, 14, 16, and 19, numbered 5^ to 3^, comprise a 2’-F modification. In some or any of the foregoing or related embodiments, the sense strand comprises 20 nucleotides, wherein nucleotides at each of positions 8, 9, 10, and 11 numbered 5^ to 3^, comprise a 2’-F modification. In some embodiments, the sense strand comprises 36 nucleotides, wherein nucleotides at each of positions 3, 5, 8, 10, 12, 13, 15, and 17, numbered 5^ to 3^, comprise a 2’-F modification. In some or any of the foregoing or related embodiments, the antisense strand comprises a 4’-O-monomethylphosphonate-2’-O-methyl modified nucleotide. In some or any of the foregoing or related embodiments, the antisense strand comprises a phosphorylated nucleotide at the 5’ terminus, and wherein the phosphorylated nucleotide is selected from uridine and adenosine. In some or any of the foregoing or related embodiments, the 4^-carbon of the sugar of the 5^-nucleotide of the antisense strand comprises a phosphate analog. In some embodiments, phosphate analog is oxymethyl phosphonate, vinyl phosphonate or malonyl phosphonate. In some or any of the foregoing or related embodiments, the phosphorylated nucleotide is 4’-O-monomethylphosphonate-2’-O-methyl uridine. In some or any of the foregoing or related embodiments, the one or more ligands is conjugated to a ribose of the sense strand at the 3’- or 5’-position. In some or any of the foregoing or related embodiments, the one or more ligands is conjugated to a ribose of the sense strand at the 2’-position. In some or any of the foregoing or related embodiments, one ligand is conjugated to position one of the sense strand, numbered 5^ to 3^. In some or any of the foregoing or related embodiments, the double-stranded oligonucleotide comprises at least two ligands. In some embodiments, the at least two ligands are conjugated to different nucleotides of the sense strand. In some embodiments, the at least two ligands are the same ligand. In some embodiments, the at least two ligands are different ligands. In some embodiments, the at least two ligands are conjugated to position 1, numbered 5^ to 3^, and a nucleotide within a tetraloop.
FH12501620.1
Attorney Docket: DCY-13025 In some or any of the foregoing or related embodiments, (a) at least one ligand is conjugated to position 1 and at least one ligand is conjugated to position 29; (b) at least one ligand is conjugated to position 1 and at least one ligand is conjugated to position 28; (c) at least one ligand is conjugated to position 1 and at least one ligand is conjugated to position 2; or (d) at least one ligand is conjugated to position 1 and at least one ligand is conjugated to position 20, numbered 5^ to 3^, of the sense strand. In some or any of the foregoing or related embodiments, the region of complementarity is fully complementary to the mRNA target sequence. In some or any of the foregoing or related embodiments, the region of complementarity is partially complementary to the mRNA target sequence. In some or any of the foregoing or related embodiments, the region of complementarity comprises no more than four mismatches to the mRNA target sequence. In some aspects, the disclosure provides an oligonucleotide-ligand conjugate of Formula (AI):
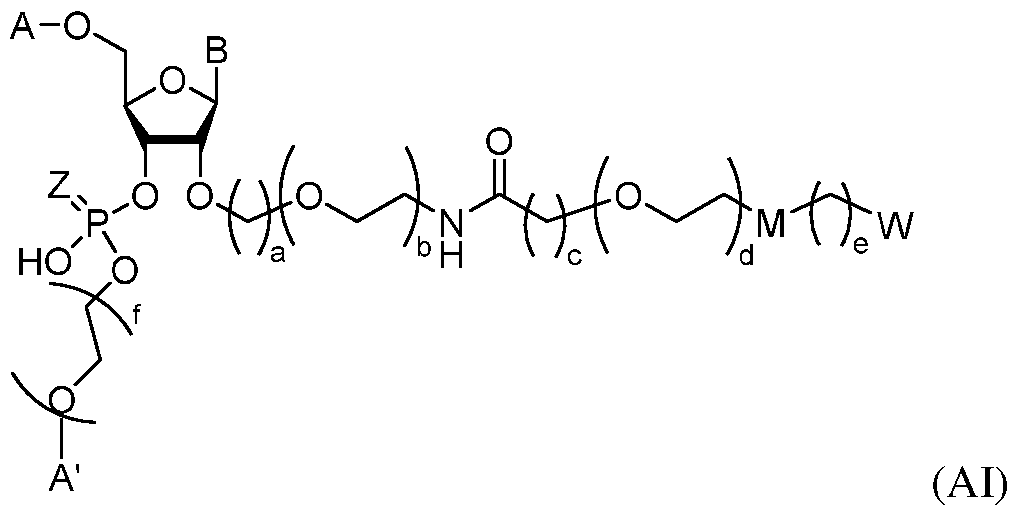
or a pharmaceutically acceptable salt or charged form thereof; wherein: A and A’ are each independently H or one or more nucleotides; B is a nucleobase; Z is O or S; M is absent, -NRC(O)-, or heteroarylene, wherein R is H or alkyl; each W is independently -C(O)OH, -CR^(C(O)OH)
2, -CR^(C(O)O(alkyl))
2, -SO
2OH, tetrazolyl, -OH, -N(H)SO
2(alkyl), -N(H)SO
2(cycloalkyl), -N(H)SO
2(optionally substituted aryl),
FH12501620.1
Attorney Docket: DCY-13025 -N(H)SO2(heterocyclyl), -N(H)SO2(optionally substituted heteroaryl), -O(optionally substituted aryl), -N(H)C(O)N(H)SO
2(alkyl), -C(O)NH(hydroxyphenyl), -N(H)C(O)(hydroxyphenyl), -N(H)(hydroxyphenyl), hydroxyphenyl, or heterocyclyl; a is 1, 2, 3, 4, 5, or 6; b is 1, 2, 3, 4, 5, 6, 7, or 8; c is 0, 1, 2, 3, 4, 5, or 6; d is 0, 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, or 20; e is 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, or 23; and f is 0, 1, 2, 3, 4, 5, or 6. In some or any of the foregoing or related embodiments, wherein M is -NRC(O)-. In some or any of the foregoing or related embodiments, wherein R is H or C1-5 alkyl. In some embodiments, R is H or methyl. In some embodiments, R is H. In some or any of the foregoing or related embodiments, W is -CO
2H,
, , , - OH, -N(H)SO2(methyl), -N(H)SO2(ethyl), -N(H)SO2(n-propyl), -N(H)SO2(i-propyl), -N( H)SO2(sec-propyl), -N(H)SO2(n-butyl), -N(H)SO2(i-butyl), -N(H)SO2(sec- butyl), -N(H)SO2(t-butyl), -N(H)SO2(n- pentyl), -N(H)SO2(cyclopropyl), -N(H)SO2(cyclobutyl), -N(H)SO2(cyclopentyl), -N(H)SO2(cyclohexyl), -N(H)SO2(cycloheptyl),
-N(H)SO
2(cyclooctyl), -N(H)SO
2([1.1.1]propellane), ,
FH12501620.1
Attorney Docket: DCY-13025
In some or any of the foregoing or related embodiments, W is piperazinyl or piperidinyl. In some or any of the foregoing or related embodiments, c is 1 or 2. In some or any of the foregoing or related embodiments, the oligonucleotide-ligand conjugate has a structure of Formula (AII)
or a pharmaceutically acceptable salt or charged form thereof. In some or any of the foregoing or related embodiments, M is heteroarylene. In some or any of the foregoing or related embodiments, the heteroarylene is
triazolylene. In some embodiments, the triazolylene is . In some or any of the foregoing or related embodiments, the oligonucleotide-ligand conjugate has a structure of Formula (AIII)
FH12501620.1
Attorney Docket: DCY-13025
or a pharmaceutically acceptable salt or charged form thereof. In some or any of the foregoing or related embodiments, d is 0 to 12. In some embodiments, d is 4 to 12. In some embodiments, d is 4. In some or any of the foregoing or related embodiments, the oligonucleotide-ligand has a structure of Formula (AIV)
or a pharmaceutically acceptable salt or a charged form thereof. In some or any of the foregoing or related embodiments, M is absent. In some or any of the foregoing or related embodiments, the oligonucleotide-ligand conjugate has a structure of Formula (AV)
or a pharmaceutically acceptable salt or a charged form thereof. In some or any of the foregoing or related embodiments, a is 1 or 2. In some or any of the foregoing or related embodiments, a is 1.
FH12501620.1
Attorney Docket: DCY-13025 In some or any of the foregoing or related embodiments, b is 1 or 2. In some embodiments, b is 2. In some or any of the foregoing or related embodiments, e is 11 to 22. In some embodiments, e is 11, 15, or 21. In some or any of the foregoing or related embodiments, f is 0, 1, or 2. In some embodiments, f is 0.

. In some or any of the foregoing or related embodiments, A is H. In some or any of the foregoing or related embodiments, Z is S. In some aspects, the disclosure provides a double-stranded oligonucleotide comprising a sense strand and an antisense strand, wherein the sense strand comprises the oligonucleotide-ligand conjugate of some or any of the foregoing or related embodiments, and wherein the sense strand and antisense strand form a duplex region. In some or any of the foregoing or related embodiments, the antisense strand is 15 to 30 nucleotides in length. In some or any of the foregoing or related embodiments, the sense strand and/or antisense strand comprises at least one modified internucleotide linkage. In some embodiments, the at least one modified internucleotide linkage is a phosphorothioate linkage. In some or any of the foregoing or related embodiments, A is H and A^ is 12 to 40 nucleotides. In some or any of the foregoing or related embodiments, the nucleotides of A^ comprise at least one modified internucleotide linkage. In some or any of the foregoing or related embodiments, A is 12-29 nucleotides and A^ is 1-10 nucleotides.
FH12501620.1
Attorney Docket: DCY-13025 In some or any of the foregoing or related embodiments, the nucleotides of A comprise at least one modified internucleotide linkage. In some or any of the foregoing or related embodiments, wherein the oligonucleotide- ligand conjugate and at least 14 nucleotides of A’ form the duplex region with the antisense strand. In some or any of the foregoing or related embodiments, the antisense strand comprises a phosphorothioate linkage (i) between positions 1 and 2, and between positions 2 and 3; or (ii) between positions 1 and 2, between positions 2 and 3, and between positions 3 and 4, wherein positions are numbered 1-4 from 5’ to 3’. In some or any of the foregoing or related embodiments, the antisense strand is 22 nucleotides in length, and wherein the antisense strand comprises a phosphorothioate linkage between positions 20 and 21 and between positions 21 and 22, wherein positions are numbered 1-22 from 5^ to 3^. In some or any of the foregoing or related embodiments, the sense strand comprises a phosphorothioate linkage (i) between positions 1 and 2; (ii) between positions 2 and 3; (iii) between positions 1 and 2, between positions 18 and 19, and between positions 19 and 20; or, (iv) between positions 2 and 3, between positions 18 and 19, and between positions 19 and 20, wherein positions are numbered 1-20 from 5^ to 3^. In some or any of the foregoing or related embodiments, the sense strand is 36 nucleotides in length, and wherein the sense strand comprises a phosphorothioate linkage between positions 1 and 2, wherein positions are numbered 1-36 from 5^ to 3^. In some or any of the foregoing or related embodiments, the sense strand is 20 nucleotides in length, and wherein the sense strand comprises a phosphorothioate linkage between positions 1 and 2, between positions 18 and 19, and between positions 19 and 20, wherein positions are numbered 1-20 from 5^ to 3^. In some or any of the foregoing or related embodiments, the duplex region comprises 20 to 30 base pairs. In some or any of the foregoing or related embodiments, the sense strand comprises a stem-loop comprising a tetraloop. In some embodiments, the tetraloop comprises a 5’- GAAA-3’ sequence. In some or any of the foregoing or related embodiments, the oligonucleotide-ligand conjugate is a nucleotide of the tetraloop. In some or any of the foregoing or related embodiments, the sense and antisense strand comprise one or more modified nucleotides. In some embodiments, the modified
FH12501620.1
Attorney Docket: DCY-13025 nucleotide comprises a modified sugar. In some embodiments, the modified sugar comprises a 2’-OMe group. In some embodiments, the modified sugar comprises a 2’-F substituent. In some or any of the foregoing or related embodiments, the sense strand comprises 20 nucleotides, wherein nucleotides at each of positions 8, 9, 10, and 11 comprise a 2’-F modification. In some or any of the foregoing or related embodiments, the sense strand comprises 36 nucleotides, wherein nucleotides at each of positions 3, 5, 8, 10, 12, 13, 15, and 17 comprise a 2’-F modification. In some or any of the foregoing or related embodiments, the antisense strand comprises 22 nucleotides, wherein nucleotides at each of positions 2, 3, 4, 5, 7, 10, and 14 comprise a 2’-F modification. In some or any of the foregoing or related embodiments, antisense strand comprises 22 nucleotides, wherein nucleotides at each of positions 2, 3, 4, 5, 7, 10, 14, 16, and 19 comprise a 2’-F modification. In some or any of the foregoing or related embodiments, the modified nucleotide comprises a modified nucleobase. In some or any of the foregoing or related embodiments, the double-stranded oligonucleotide comprises a 4’-O-monomethylphosphonate-2’-O-methyl modified nucleotide. In some or any of the foregoing or related embodiments, the antisense strand comprises a phosphorylated nucleotide at the 5’ terminus, and wherein the phosphorylated nucleotide is selected from uridine and adenosine. In some or any of the foregoing or related embodiments, the 4^-carbon of the sugar of the 5^-nucleotide of the antisense strand comprises a phosphate analog. In some embodiments, the phosphate analog is oxymethyl phosphonate, vinyl phosphonate or malonyl phosphonate. In some or any of the foregoing or related embodiments, the phosphorylated nucleotide is 4’-O-monomethylphosphonate-2’-O-methyl uridine. In some aspects, the disclosure provides an oligonucleotide-ligand conjugate of Formula (BI):
FH12501620.1
Attorney Docket: DCY-13025
or a pharmaceutically acceptable salt or charged form thereof, wherein: B is a nucleobase; A is a one or more nucleotides; Z
1 and Z
2 are each independently O or S; M is absent, -NRC(O)-, or heteroarylene, wherein R is H or alkyl; W is independently -C(O)OH, -CR^(C(O)OH)
2, -CR^(C(O)O(alkyl))
2, -SO
2OH, tetrazolyl, -OH, -N(H)SO
2(alkyl), -N(H)SO
2(cycloalkyl), -N(H)SO
2(optionally substituted aryl), -N(H)SO
2(heterocyclyl), -N(H)SO
2(optionally substituted heteroaryl), -O(optionally substituted aryl), -N(H)C(O)N(H)SO2(alkyl), -C(O)NH(hydroxyphenyl), -N(H)C(O)(hydroxyphenyl), -N(H)(hydroxyphenyl), hydroxyphenyl, or heterocyclyl; a is 1, 2, 3, 4, or 5; c is 0, 1, 2, 3, 4, 5, or 6; d is 0, 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, or 20; e is 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, or 23; and f is 0, 1, 2, 3, 4, 5, or 6. In some aspects, the disclosure provides an oligonucleotide-ligand conjugate of Formula (BII):
FH12501620.1
Attorney Docket: DCY-13025
or a salt or charged form thereof, wherein: B is a nucleobase; A is a one or more nucleotides ; Z is O or S; M is absent, NRC(O), or heteroarylene, wherein R is H or alkyl; W is independently -C(O)OH, -CR^(C(O)OH)2, -CR^(C(O)O(alkyl))2, -SO2OH, tetrazolyl, -OH, -N(H)SO2(alkyl), -N(H)SO2(cycloalkyl), -N(H)SO2(optionally substituted aryl), -N(H)SO
2(heterocyclyl), -N(H)SO
2(optionally substituted heteroaryl), -O(optionally substituted aryl), -N(H)C(O)N(H)SO
2(alkyl), -C(O)NH(hydroxyphenyl), -N(H)C(O)(hydroxyphenyl), -N(H)(hydroxyphenyl), hydroxyphenyl, or heterocyclyl; a is 1, 2, 3, 4, or 5; c is 0, 1, 2, 3, 4, 5, or 6; d is 0, 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, or 20; e is 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, or 23; and f is 0, 1, 2, 3, 4, 5, or 6. In some aspects, the disclosure provides an oligonucleotide-ligand conjugate of Formula (CI)
or a pharmaceutically acceptable salt or charged form thereof, wherein:
FH12501620.1
Attorney Docket: DCY-13025 B is a nucleobase; A is one or more nucleotides; Z
1 and Z
2 are each independently O or S; M is absent, NRC(O), or heteroarylene, wherein R is H or alkyl; W is independently -C(O)OH, -CR^(C(O)OH)2, -CR^(C(O)O(alkyl))2, -SO2OH, tetrazolyl, -OH, -N(H)SO2(alkyl), -N(H)SO2(cycloalkyl), -N(H)SO2(optionally substituted aryl), -N(H)SO2(heterocyclyl), -N(H)SO2(optionally substituted heteroaryl), -O(optionally substituted aryl), -N(H)C(O)N(H)SO2(alkyl), -C(O)NH(hydroxyphenyl), -N(H)C(O)(hydroxyphenyl), -N(H)(hydroxyphenyl), hydroxyphenyl, or heterocyclyl; a is 1, 2, 3, 4, or 5; c is 0, 1, 2, 3, 4, 5, or 6; d is 0, 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, or 20; e is 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, or 23; and f is 0, 1, 2, 3, 4, 5, or 6. In some aspects, the disclosure provides an oligonucleotide-ligand conjugate of Formula (CII)

or a salt or charged form thereof, wherein: B is a nucleobase; A is one or more nucleotides; Z is O or S; M is absent, NRC(O), or heteroarylene, wherein R is H or alkyl; W is independently -C(O)OH, -CR^(C(O)OH)
2, -CR^(C(O)O(alkyl))
2, -SO
2OH, tetrazolyl, -OH, -N(H)SO
2(alkyl), -N(H)SO
2(cycloalkyl), -N(H)SO
2(optionally substituted aryl), -N(H)SO2(heterocyclyl), -N(H)SO2(optionally substituted heteroaryl), -O(optionally substituted
FH12501620.1
Attorney Docket: DCY-13025 aryl), -N(H)C(O)N(H)SO
2(alkyl), -C(O)NH(hydroxyphenyl), -N(H)C(O)(hydroxyphenyl), -N(H)(hydroxyphenyl), hydroxyphenyl, or heterocyclyl; a is 1, 2, 3, 4, or 5; c is 0, 1, 2, 3, 4, 5, or 6; d is 0, 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, or 20; e is 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, or 23; and f is 0, 1, 2, 3, 4, 5, or 6. In some aspects, the disclosure provides a double-stranded oligonucleotide comprising a sense strand and an antisense strand, wherein the sense strand comprises an oligonucleotide-ligand conjugate of some or any of the foregoing or related embodiments, and wherein the sense strand and antisense strand form a duplex region. In some or any of the foregoing or related embodiments, the antisense strand is 15 to 30 nucleotides in length. In some or any of the foregoing or related embodiments, A is 12 to 40 nucleotides. In some or any of the foregoing or related embodiments, the sense strand and/or antisense strand comprises at least one modified internucleotide linkage. In some embodiments, the at least one modified internucleotide linkage is a phosphorothioate linkage. In some embodiments, the duplex region includes one or more phosphorothioate linkages. In some embodiments, two phosphorothioate linkages are adjacent to each other. In some or any of the foregoing or related embodiments, the antisense strand comprises a phosphorothioate linkage (i) between positions 1 and 2, and between positions 2 and 3; or (ii) between positions 1 and 2, between positions 2 and 3, and between positions 3 and 4, wherein positions are numbered 1-4 from 5’ to 3’. In some or any of the foregoing or related embodiments, the antisense strand is 22 nucleotides in length, and wherein the antisense strand comprises a phosphorothioate linkage between positions 20 and 21 and between positions 21 and 22, wherein positions are numbered 1-22 from 5^ to 3^. In some or any of the foregoing or related embodiments, the sense strand comprises a phosphorothioate linkage (i) between positions 1 and 2; (ii) between positions 2 and 3; (iii) between positions 1 and 2, between positions 18 and 19, and between positions 19 and 20; or, (iv) between positions 2 and 3, between positions 18 and 19, and between positiosn 19 and 20, wherein positions are numbered 1-20 from 5^ to 3^.
FH12501620.1
Attorney Docket: DCY-13025 In some or any of the foregoing or related embodiments, wherein the sense strand is 36 nucleotides in length, and wherein the sense strand comprises a phosphorothioate linkage between positions 1 and 2, wherein positions are numbered 1-36 from 5^ to 3^. In some or any of the foregoing or related embodiments, the sense strand is 20 nucleotides in length, and wherein the sense strand comprises a phosphorothioate linkage between positions 1 and 2, between positions 18 and 19, and between positions 19 and 20, wherein positions are numbered 1-20 from 5^ to 3^. In some or any of the foregoing or related embodiments, the duplex region comprises 20 to 30 base pairs. In some or any of the foregoing or related embodiments, the sense strand comprises a loop region that includes a tetraloop region. In some embodiments, the tetraloop region comprises a 5’-GAAA-3’ sequence. In some or any of the foregoing or related embodiments, the sense and antisense strand comprise one or more modified nucleotides. In some embodiments, the modified nucleotide comprises a modified sugar. In some embodiments, the modified sugar comprises a 2’-OMe group. In some embodiments, the modified sugar comprises a 2’-F substituent. In some or any of the foregoing or related embodiments, the sense strand comprises 20 nucleotides, wherein nucleotides at each of positions 8, 9, 10, and 11 comprise a 2’-F modification. In some or any of the foregoing or related embodiments, the sense strand comprises 36 nucleotides, wherein nucleotides at each of positions 3, 5, 8, 10, 12, 13, 15, and 17 comprise a 2’-F modification. In some or any of the foregoing or related embodiments, the antisense strand comprises 22 nucleotides, wherein nucleotides at each of positions 2, 3, 4, 5, 7, 10, and 14 comprise a 2’-F modification. In some or any of the foregoing or related embodiments, the antisense strand comprises 22 nucleotides, wherein nucleotides at each of positions 2, 3, 4, 5, 7, 10, 14, 16, and 19 comprise a 2’-F modification. In some or any of the foregoing or related embodiments, the modified nucleotide comprises a modified nucleobase. In some or any of the foregoing or related embodiments, the double-stranded oligonucleotide of comprises a 4’-O-monomethylphosphonate-2’-O-methyl modified nucleotide.
FH12501620.1
Attorney Docket: DCY-13025 In some or any of the foregoing or related embodiments, the antisense strand comprises a phosphorylated nucleotide at the 5’ terminus, and wherein the phosphorylated nucleotide is selected from uridine and adenosine. In some or any of the foregoing or related embodiments, the 4^-carbon of the sugar of the 5^-nucleotide of the antisense strand comprises a phosphate analog. In some embodiments, the phosphate analog is oxymethyl phosphonate, vinyl phosphonate or malonyl phosphonate. In some or any of the foregoing or related embodiments, the phosphorylated nucleotide is 4’-O-monomethylphosphonate-2’-O-methyl uridine. In some or any of the foregoing or related embodiments, the sense strand comprises a second oligonucleotide-ligand conjugate. In some embodiments, the second oligonucleotide- ligand conjugate is selected from the oligonucleotide-ligand conjugate of any some or any of the foregoing or related aspects. In some or any of the foregoing or related embodiments, the antisense strand has a region of complementarity to a target sequence in a target mRNA in extrahepatic tissue, wherein the region of complementarity is at least 15 contiguous nucleotides in length. In some or any of the foregoing or related embodiments, the region of complementarity is fully complementary to the mRNA target sequence. In some or any of the foregoing or related embodiments, the region of complementarity is partially complementary to the mRNA target sequence. In some or any of the foregoing or related embodiments, the region of complementarity comprises no more than four mismatches to the mRNA target sequence. In some or any of the foregoing or related embodiments, the extrahepatic tissue is adipose tissue, heart tissue, skeletal muscle, or adrenal gland tissue. In some embodiments, the extrahepatic tissue is adipose tissue. In some aspects, the disclosure provides a double-stranded oligonucleotide comprising a sense strand and an antisense strand, wherein the sense strand comprises two or more oligonucleotide-ligand conjugates of (i), (ii), or (iii): (i) Formula (AI), (AII), (AIII), (AIV), or (AV); (ii) Formula (BI) or (BII); and (iii) Formula (CI) or (CII). In some or any of the foregoing or related embodiments, the two or more oligonucleotide ligand conjugates are conjugated to different nucleotides of the sense strand.
FH12501620.1
Attorney Docket: DCY-13025 In some or any of the foregoing or related embodiments, the two or more oligonucleotide ligand conjugates are the same. In some or any of the foregoing or related embodiments, the two or more oligonucleotide ligand conjugates are different. In some or any of the foregoing or related embodiments, the sense strand comprises a first oligonucleotide-ligand conjugate and a second oligonucleotide-ligand conjugate. In some or any of the foregoing or related embodiments, nucleobase (B) of the first oligonucleotide-ligand conjugate is the nucleobase at position 1 of the sense strand and the nucleobase (B) of the second oligonucleotide-ligand conjugate is a nucleobase within a tetraloop of the sense strand. In some or any of the foregoing or related embodiments, (a) the nucleobase (B) of the first oligonucleotide-ligand conjugate is the nucleobase at position 1 of the sense strand, and the nucleobase (B) of the second oligonucleotide-ligand conjugate is the nucleobase at position 29 of the sense strand; (b) the nucleobase (B) of the first oligonucleotide-ligand conjugate is the nucleobase at position 1 of the sense strand, and the nucleobase (B) of the second oligonucleotide-ligand conjugate is the nucleobase at position 28 of the sense strand; (c) the nucleobase (B) of the first oligonucleotide-ligand conjugate is the nucleobase at position 1 of the sense strand, and the nucleobase (B) of the second oligonucleotide-ligand conjugate is the nucleobase at position 20 of the sense strand; or (d) the nucleobase (B) of the first oligonucleotide-ligand conjugate is the nucleobase at position 1 of the sense strand, and the nucleobase (B) of the second oligonucleotide-ligand conjugate is the nucleobase at position 2 of the sense strand. In some or any of the foregoing or related embodiments, the oligonucleotide-ligand conjugate reduces expression of the target mRNA in an extrahepatic tissue, provided the oligonucleotide-ligand conjugate does not reduce expression of the mRNA target in the liver. In some aspect, the disclosure provides a pharmaceutical composition comprising the double-stranded oligonucleotide of some or any of the foregoing or related embodiments, and a pharmaceutically acceptable carrier, delivery agent, or excipient. In some aspects, the disclosure provides a method of inhibiting target mRNA expression in an extrahepatic cell or tissue in a subject, comprising administering to the subject the double-stranded oligonucleotide of some or any of the foregoing or related embodiments, or the pharmaceutical composition of any of the foregoing or related
FH12501620.1
Attorney Docket: DCY-13025 embodiments, thereby inhibiting target mRNA expression in the cell of the extrahepatic tissue. In some or any of the foregoing or related embodiments, the extrahepatic cell or tissue is selected from skeletal muscle, adipose tissue, adrenal tissue, heart tissue, and any combination thereof. In some or any of the foregoing or related embodiments, reduction of the target mRNA in the extrahepatic cell or tissue is increased compared to reduction in liver cells or tissue, optionally wherein reduction of the target mRNA is increased by at least 10%. In some or any of the foregoing or related embodiments, reduction of the target mRNA is increased by at least 15%, at least 20%, at least 25%, at least 30%, at least 35%, at least 40%, at least 45% or at least 50%. In some aspects, the disclosure provides the double-stranded oligonucleotide of any one of the foregoing or related embodiments in the manufacture of a medicament for inhibiting target mRNA expression in an extrahepatic cell or tissue in a subject. In some aspects, the disclosure provides for use of the double-stranded oligonucleotide of any one of the foregoing or related embodiments for inhibiting target mRNA expression in a cell of an extrahepatic tissue in a subject. In some aspects, the disclosure provides a kit comprising a container comprising the double-stranded oligonucleotide of some or any of the foregoing or related embodiments, and optionally a pharmaceutically acceptable carrier, and instructions for administering the oligonucleotide-ligand conjugate to a subject in need thereof, wherein the oligonucleotide- ligand conjugate inhibits target mRNA expression in an extrahepatic cell or tissue in the subject. In some or any of the foregoing or related embodiments, the extrahepatic cell or tissue is selected from skeletal muscle, adipose tissue, adrenal tissue, heart tissue, and any combination thereof. In some or any of the foregoing or related embodiments, the cell of the extrahepatic cell or tissue is selected from a cardiomyocyte, a cell of skeletal muscle, a cell of adipose tissue, a cell of adrenal tissue, and any combination thereof. In some or any of the foregoing or related embodiments, reduction of the target mRNA in the extrahepatic cell or tissue is increased compared to reduction in a cell of the liver, optionally wherein reduction of the target mRNA is increased by at least 10%.
FH12501620.1
Attorney Docket: DCY-13025 In some or any of the foregoing or related embodiments, reduction of the target mRNA is increased by at least 15%, at least 20%, at least 25%, at least 30%, at least 35%, at least 40%, at least 45% or at least 50%. In some aspects, the disclosure provides a method for treating a subject having a disease, disorder or condition associated with expression of an mRNA in an extrahepatic cell or tissue, the method comprising administering to the subject a therapeutically effective amount of the double-stranded oligonucleotide of some or any of the foregoing or related embodiments, or the pharmaceutical composition of any of the foregoing or related embodiments. In some aspects, the disclosure provides a method of delivering a double-stranded oligonucleotide to a cell or population of cells in extrahepatic tissue, the method comprising administering the pharmaceutical composition of some or any of the foregoing or related embodiments. BRIEF DESCRIPTION OF FIGURES FIG.1A provides structures of alkyl and alkylene-COOH groups suitable for conjugation to RNAi oligonucleotides. FIG.1B provides the structure of RNAi oligonucleotides having chemical modifications with a C22 conjugate linked to a nucleotide in a stem loop (Duplex A); a C16- COOH conjugate linked to a nucleotide in a stem loop (Duplex B); a C16-COOH conjugate linked to position 1 (P1) of the sense strand (Duplex C); or, a C16 conjugate linked to P1 of the sense strand and a C22 conjugate linked to a nucleotide in a the stem loop (Duplex D). FIGs.2A-2B are graphs demonstrating remaining mouse Aldh2 mRNA levels in the liver (FIG.2A) or adipose tissue (FIG.2B) of mice following intravenous treatment with a single dose (15mg/kg) of the corresponding oligonucleotide in FIG.1B. Tissue was collected 7 days post administration. Control mice were administered PBS. FIG.3 provides the structure of RNAi oligonucleotide molecules comprising a C22 conjugate (Duplex A) or a C16-COOH conjugate (Duplex B) linked to the stem loop of the sense strand. FIGs.4A-4D are graphs demonstrating remaining mouse Aldh2 mRNA levels in the liver (FIG.4A), gonadal white adipose tissue (gWAT) (FIG.4B), quadricep (FIG.4C), and subcutaneous white adipose tissue (scWAT) (FIG.4D) of mice following intravenous treatment with a single dose (15 mg/kg) of a corresponding oligonucleotide in FIG.3. Tissue was collected 7 days post administration. Control mice were administered PBS.
FH12501620.1
Attorney Docket: DCY-13025 FIG.5 provides structures of RNAi oligonucleotide conjugates. The key for the depicted molecules is the same as that provided in FIG.1B. FIGs.6A-6F are graphs demonstrating remaining mouse Aldh2 mRNA levels in the liver (FIG.6A), scWAT (FIG.6B), gWAT (FIG.6C), heart (FIG.6D), quadricep (FIG. 6E), and adrenal tissue (FIG.6F) of mice following intravenous treatment with a single dose (15 mg/kg) of oligonucleotides comprising a modified sense strand of SEQ ID NO: 2 or 4 and a modified antisense strand of SEQ ID NO: 9 linked to conjugates as depicted in FIG.5 and the parent C22 conjugate shown in FIG.1B. Tissue was collected 7 days post administration. Control mice were administered PBS. FIG.7 provides structures of RNAi oligonucleotide conjugates. The key for the depicted molecules is the same as that provided in FIG.1B. FIGs.8A-8D are graphs demonstrating remaining mouse Aldh2 mRNA levels in the liver (FIG.8A), adipose tissue (FIG.8B), quadricep (FIG.8C), and heart (FIG.8D) of mice following subcutaneous treatment with a single dose (10 mg/kg) of oligonucleotides comprising a modified sense strand of SEQ ID NO: 10 and a modified antisense strand of SEQ ID NO: 5 or 6 linked to conjugates as depicted in FIG.7 and the parent C22 conjugate shown in FIG.1B. Tissue was collected 7 days post administration. Control mice were administered PBS. FIG.9 provides structures of RNAi oligonucleotide conjugates. The key for the depicted molecules is the same as that provided in FIG.1B. FIGs.10A-10D are graphs demonstrating remaining mouse Aldh2 mRNA levels in the liver (FIG.10A), skeletal muscle (FIG.10B), heart (FIG.10C), and gWAT (FIG.10D) of mice following subcutaneous treatment with a single dose (10 mg/kg) of oligonucleotides comprising a modified sense strand of SEQ ID NO: 5 and a modified antisense strand of SEQ ID NO: 10 linked to conjugates as depicted in FIG.9. Tissue was collected 7 days post administration. Control mice were administered PBS. FIGs.11A-11D are graphs demonstrating remaining mouse Aldh2 mRNA levels in the liver (FIG.11A), skeletal muscle (FIG.11B), heart (FIG.11C), and gWAT (FIG.11D) of mice following subcutaneous treatment with a single dose (10 mg/kg) of oligonucleotides comprising a modified sense strand of SEQ ID NO: 5 and a modified antisense strand of SEQ ID NO: 10 linked to conjugates as depicted in FIG.9. Tissue was collected 21 days post administration. Control mice were administered PBS.
FH12501620.1
Attorney Docket: DCY-13025 FIG.12 provides structures of RNAi oligonucleotide molecules comprising a C16 conjugate and/or a C16-COOH conjugate at different positions of the sense strand. The key for the depicted molecules is the same as that provided in FIG.1B. FIGs.13A-13D are graphs demonstrating remaining mouse Aldh2 mRNA levels in the liver (FIG.13A), quadricep (FIG.13B), heart (FIG.13C), and adipose tissue (FIG. 13D) of mice following subcutaneous treatment with a single dose (10 mg/kg) of oligonucleotides comprising a modified sense strand of SEQ ID NO: 5, 7, or 8 and a modified antisense strand of SEQ ID NO: 10 linked to conjugates as depicted in FIG.12. Tissue was collected 7 days post administration. Control mice were administered PBS. FIG.14 provides structures of RNAi oligonucleotide conjugates. The key for the depicted molecules is the same as that provided in FIG.1B. FIGs.15A-15B are graphs demonstrating remaining monkey Aldh2 mRNA levels in liver (FIG.15A) and muscle (FIG.15B) of cynomolgus monkeys following subcutaneous treatment with a single dose 10 mg/kg of oligonucleotides comprising a modified sense strand of SEQ ID NO: 5 and a modified antisense strand of SEQ ID NO: 10 linked to conjugates as depicted in FIG.14. Tissue was collected 28 days post administration. Control animals were administered PBS. FIG.16 provides structures of RNAi oligonucleotide conjugates. The key for the depicted molecules is the same as that provided in FIG.1B. FIGs.17A-17D are graphs demonstrating remaining monkey Aldh2 mRNA levels in liver (FIG.17A), skeletal muscle (FIG.17B), heart (FIG.17C), and gWAT (FIG.17D) of cynomolgus monkeys following subcutaneous treatment with a single dose 10 mg/kg of oligonucleotides comprising a modified sense strand of SEQ ID NO: 5 and a modified antisense strand of SEQ ID NO: 10 linked to conjugates as depicted in FIG.16. Tissue was collected 28 days post administration. Control animals were administered PBS. FIG.18 provides structures of RNAi oligonucleotide conjugates. The key for the depicted molecules is the same as that provided in FIG.1B. FIGs.19A-19D are graphs demonstrating remaining monkey Aldh2 mRNA levels in liver (FIG.19A), skeletal muscle (FIG.19B), heart (FIG.19C), and gWAT (FIG.19D) of cynomolgus monkeys following subcutaneous treatment with a single dose 10 mg/kg of oligonucleotides comprising a modified sense strand of SEQ ID NO: 5 and a modified antisense strand of SEQ ID NO: 10 linked to conjugates as depicted in FIG.18. Tissue was collected 28 days post administration. Control animals were administered PBS.
FH12501620.1
Attorney Docket: DCY-13025 FIG.20 provides structures of RNAi oligonucleotide conjugates. The key for the depicted molecules is the same as that provided in FIG.1B. FIGs.21A-21D are graphs demonstrating remaining monkey Aldh2 mRNA levels in liver (FIG.21A), skeletal muscle (FIG.21B), heart (FIG.21C), and gWAT (FIG.21D) of cynomolgus monkeys following subcutaneous treatment with a single dose 10 mg/kg of oligonucleotides comprising a modified sense strand of SEQ ID NO: 5 and a modified antisense strand of SEQ ID NO: 10 linked to conjugates as depicted in FIG.20. Tissue was collected 28 days post administration. Control animals were administered PBS. FIG.22 provides structures of RNAi oligonucleotide conjugates. The key for the depicted molecules is the same as that provided in FIG.1B. FIGs.23A-23D are graphs demonstrating remaining monkey Aldh2 mRNA levels in liver (FIG.23A), skeletal muscle (FIG.23B), heart (FIG.23C), and gWAT (FIG.23D) of cynomolgus monkeys following subcutaneous treatment with a single dose 10 mg/kg of oligonucleotides comprising a modified sense strand of SEQ ID NO: 5 and a modified antisense strand of SEQ ID NO: 10 linked to conjugates as depicted in FIG.22. Tissue was collected 28 days post administration. Control animals were administered PBS. FIG.24 provides structures of RNAi oligonucleotide conjugates. The key for the depicted molecules is the same as that provided in FIG.1B. FIGs.25A-25D are graphs demonstrating remaining monkey Aldh2 mRNA levels in liver (FIG.25A), skeletal muscle (FIG.25B), heart (FIG.25C), and gWAT (FIG.25D) of cynomolgus monkeys following subcutaneous treatment with a single dose 10 mg/kg of oligonucleotides comprising a modified sense strand of SEQ ID NO: 5 or 7 and a modified antisense strand of SEQ ID NO: 10 linked to conjugates as depicted in FIG.24. Tissue was collected 28 days post administration. Control animals were administered PBS. DETAILED DESCRIPTION In some aspects, the disclosure provides oligonucleotide-conjugates (e.g., RNAi oligonucleotides comprising an oligonucleotide-ligand conjugate) that reduce expression of a target gene in extrahepatic tissue. In other aspects, the disclosure provides methods of treating a disease or disorder associated with expression of a target gene. In other aspects, the disclosure provides methods of treating a disease or disorder associated with expression of a target gene using the oligonucleotide-conjugates, or pharmaceutically acceptable compositions thereof, described herein. In other aspects, the disclosure provides methods of using the
FH12501620.1
Attorney Docket: DCY-13025 oligonucleotide-conjugates described herein in the manufacture of a medicament for treating a disease or disorder associated with expression of a target gene. The oligonucleotide-conjugates described herein comprise one or more ligands bearing carboxyl groups. The ligands comprise alkylene, alkenylene, or alkynylene groups, and one or more carboxyl groups are located on the alkylene, alkenylene, or alkynylene (referred to interchangeably herein as a “hydrocarbon chain bearing one or more carboxyl groups’). It was surprisingly discovered that such ligands containing hydrocarbon chains bearing one or more carboxyl groups target extrahepatic tissue, and thus are capable of modulating the expression of a target genes in extrahepatic cells, tissues, and/or organs. Without being bound by theory, the size of the alkylene, alkenylene or alkynylene chain bearing the carboxyl group was discovered to be important in targeting extrahepatic tissue. More specifically, when carboxylic acids were located on hydrocarbon chains having from 6 to 24 carbons (not including the number of carbons in the linker as described here), the conjugates effectively modified expression of a target gene in extrahepatic tissue at a higher amount than reduction of expression of the same target gene in hepatocytes. This unexpected discovery allows for oligonucleotide-conjugates to be used in treating disease with specificity for extrahepatic tissue. In nucleic acid chemistry, many different artificial nucleic acids have been developed to alter the behavior of siRNAs under physiological conditions. In particular, phosphorothioate (PS), 2^-methoxy (2^-OMe), and 2^-fluoro nucleic acid have often been used to modify the siRNA its behavior, toxicity and thermostability. Oligonucleotide-Ligand Conjugates The disclosure provides, inter alia, RNAi oligonucleotides comprising at least one oligonucleotide-ligand conjugate that reduce expression of a target gene in extrahepatic tissue. In some embodiments, an RNAi oligonucleotide provided by the disclosure targets an mRNA encoding the target gene. Messenger RNA (mRNA) that encodes a target gene and is targeted by an RNAi oligonucleotide of the disclosure is referred to herein as “target mRNA”. In some embodiments, the ligand comprises one or more Y-(CO
2H)
n groups, or a charged form thereof, wherein Y is alkylene, alkenylene, or alkynylene, and n is 1-6. In some embodiments, the alkylene, alkenylene, or alkynylene comprises at least 5, at least 6, at least 7 at least 8, at least 9, at least 10, at least 11, at least 12, at least 13, at least 14, at least 15, at least 16, at least 17, at least 18, at least 19, at least 20, at least 21, at least 22, at least 23, at least 24, at least 25, or at least 26 carbon atoms. In some embodiments, the alkylene, alkenylene, or
FH12501620.1
Attorney Docket: DCY-13025 alkynylene comprises from 6-26 carbon atoms (e.g., 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, or 26, including all values and ranges therein), or from 10-22 carbon atoms (e.g., 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, or 22, including all values and ranges therein). In some embodiments, the alkylene, alkenylene, or alkynylene comprises at least 6 (e.g., at least 6, at least 7, at least 8, at least 9, at least 10, at least 11, at least 12, at least 13, at least 14, at least 15, at least 16, at least 17, at least 18, at least 19, at least 20, at least 21, at least 22, at least 23, at least 24, at least 25, or at least 26) carbon atoms. In some embodiments, the alkylene, alkenylene, or alkynylene comprises less than or equal to 30, less than or equal to 29, less than or equal to 28, less than or equal to 27, less than or equal to 26, or less than equal to 25 carbon atoms. In some embodiments, n is 1, 2, 3, 4, 5 or 6. In some embodiments, n is 1 or 2. In some embodiments, the ligand comprises 1, 2, 3, 4, 5, 6 or more Y-(CO2H)n groups, or a charged form thereof. In some embodiments, the oligonucleotide comprises 1, 2, 3, 4, 5, or more ligands. In some embodiments, the ligand further comprises a linker moiety, L. In some embodiments, L comprises a bivalent or trivalent C
1-C
50 alkylene, wherein 1-25 methylene groups are optionally and independently replaced by -N(H)-, -N(C
1-C
4 alkyl)-, -N(cycloalkyl)- , -O-, -C(O)-, -C(O)O-, -S-, -S(O)-, -S(O)2-, -S(O)2N(C1-C4 alkyl)-, -S(O)2N(cycloalkyl)-, - N(H)C(O)-, -N(C
1-C
4 alkyl)C(O)-, -N(cycloalkyl)C(O)-, -C(O)N(H)-, -C(O)N(C
1-C
4 alkyl), - C(O)N(cycloalkyl), aryl, heteroaryl, cycloalkyl, or cycloalkenyl. In some embodiments, b is 0, 1, 2, 3, 4 or 5. When, Y is alkylene and L comprises -O(CH2)a(OCH2CH2)bNH-, L may further comprises a total of 50 carbon atoms in which up to 25 methylene groups are optionally and independently replaced as described above. In some embodiments when Y is alkylene, L comprises -O(CH2)a(OCH2CH2)bNH-, wherein a is 1-6 and b is 0-5, and optionally the methylene groups and substitutions mentioned above, such that L comprises up to C1-C50 alkylene, wherein 1-25 methylene groups are optionally and independently replaced as described above. In some embodiments, a is 1, 2, 3, 4, 5 or 6. In some embodiments, a is 2. In some embodiments, b is 1, 2, 3, 4, 5 or 6. In some embodiments, provided herein is an RNAi oligonucleotide comprising: (i) an antisense strand of 15 to 30 nucleotides in length and a sense strand of 13 to 40 nucleotides in length, wherein the antisense strand has a region of complementarity to a target sequence in a target mRNA in an extrahepatic tissue, and wherein the region of complementarity is at least 15 contiguous nucleotides in length; and (ii) one or more ligands, each ligand comprising one or more -L-Y-(CO2H)n groups, or a charged form thereof, wherein L is a linker, Y is alkylene, alkenylene, or alkynylene, and n
FH12501620.1
Attorney Docket: DCY-13025 is 1-6, wherein when Y is alkylene, L comprises -O(CH
2)
a(OCH
2CH
2)
bNH-, wherein a is 1-6, and b is 0-5, and wherein the ligand is conjugated to a nucleotide of the sense strand. In some embodiments, the RNAi oligonucleotide comprises: (i) an antisense strand of 15 to 30 nucleotides in length and a sense strand of 15 to 40 nucleotides in length, wherein the antisense strand has a region of complementarity to a target sequence in a target mRNA in extrahepatic tissue, and wherein the region of complementarity is at least 15 contiguous nucleotides in length; and (ii) one or more ligands, each ligand comprising one or more C6-24 alkylene-(CO2H)n, or a charged form thereof, conjugated to a nucleotide of the sense strand, wherein n is 1-6, wherein knock down of the target mRNA in the extrahepatic tissue is greater than knock down of the target mRNA in liver tissue. In some embodiments, the extra-hepatic tissue is adipose, heart, skeletal muscle, the central nervous system, or adrenal gland tissue. In some embodiments, the extrahepatic tissue is adipose tissue. In some embodiments, the extrahepatic tissue is heart tissue. In some embodiments, the extrahepatic tissue is skeletal muscle. In some embodiments, the extrahepatic tissue is adrenal gland tissue. In some embodiments, the extrahepatic tissue is central nervous system tissue. In some embodiments, the RNAi oligonucleotide comprises an antisense strand of about 15 to 30 nucleotides in length, for example, about 15 to 30 nucleotides in length, about 15 to 29 nucleotides in length, about 15 to 28 nucleotides in length, about 15 to 27 nucleotides in length, about 15 to 26 nucleotides in length, about 15 to 25 nucleotides in length, about 15 to 24 nucleotides in length, about 15 to 23 nucleotides in length, about 15 to 22 nucleotides in length, about 15 to 21 nucleotides in length, about 15 to 20 nucleotides in length, including any values or ranges therebetween. In some embodiments, the antisense strand comprises 22 nucleotides. In some embodiments, the RNAi oligonucleotide comprises a sense strand of about 13 to 40 nucleotides in length, for example, about 13 to 40 nucleotides in length, about 13 to 38 nucleotides in length, about 13 to 36 nucleotides in length, about 13 to 34 nucleotides in length, about 13 to 32 nucleotides in length, about 13 to 30 nucleotides in length, about 13 to 28 nucleotides in length, about 15 to 40 nucleotides in length, about 15 to 38 nucleotides in length, about 15 to 36 nucleotides in length, about 15 to 34 nucleotides in length, about 15 to 32 nucleotides in length, about 15 to 30 nucleotides in length, about 15 to 28 nucleotides in length, about 20 to 40 nucleotides in length, about 20 to 38 nucleotides in length, about 20 to 36 nucleotides in length, about 20 to 34 nucleotides in length, about 20 to 32 nucleotides in length,
FH12501620.1
Attorney Docket: DCY-13025 about 20 to 30 nucleotides in length, about 20 to 28 nucleotides in length, including any values or ranges therebetween. In some embodiments, the sense strand comprises 28 nucleotides. In some embodiments, each of the one or more ligands comprises one or more -L-Y- CO
2H group of a charged form thereof. In some embodiments, each of the one or more ligands comprises two or more -L-Y-CO2H group of a charged form thereof. In some embodiments, each of the three or more ligands comprises one or more -L-Y-CO2H group of a charged form thereof. In some embodiments, Y is alkylene. In some embodiments, Y is C6-C30 alkylene, for example, C6-C28 alkylene, C6-C26 alkylene, C6-C24 alkylene, C6-C22 alkylene, C6-C20 alkylene, C8-C30 alkylene, C10-C30 alkylene, C10-C28 alkylene, C12-C28 alkylene, C14-C28 alkylene, C16- C28 alkylene, or C16-C22 alkylene. In some embodiments, Y is C16-C22 alkylene. In some embodiments, when Y is alkylene, then L comprises -O(CH2)a(OCH2CH2)bNH-, wherein a is 1-6, and b is 0-5. In some embodiments, a is 1 and b is 2. In some embodiments, a is 1 and b is 0. In some embodiments, a is 1 and b is 1. In some embodiments, Y is alkenylene. In some embodiments, Y is C
6-C
30 alkenylene, for example, C
6-C
28 alkenylene, C
6-C
26 alkenylene, C
6-C
24 alkenylene, C
6-C
22 alkenylene, C
6- C20 alkenylene, C8-C30 alkenylene, C10-C30 alkenylene, C10-C28 alkenylene, C12-C28 alkenylene, C
14-C
28 alkenylene, C
16-C
28 alkenylene, or C
16-C
22 alkenylene. In some embodiments, the alkenylene comprises from 1-6 olefinic bonds, including 1-5 olefinic bonds, 1-4 olefinic bonds, 1-3 olefinic bonds, or 1-2 olefinic bonds. In some embodiments, Y is alkynylene. In some embodiments, Y is C6-C30 alkynylene, for example, C6-C28 alkynylene, C6-C26 alkynylene, C6-C24 alkynylene, C6-C22 alkynylene, C6- C20 alkynylene, C8-C30 alkynylene, C10-C30 alkynylene, C10-C28 alkynylene, C12-C28 alkynylene, C14-C28 alkynylene, C16-C28 alkynylene, or C16-C22 alkynylene. In some embodiments, the alkynylene comprises from 1-6 olefinic bonds, including 1-5 olefinic bonds, 1-4 olefinic bonds, 1-3 olefinic bonds, or 1-2 olefinic bonds. In some embodiments, each ligand comprises a linker (L), wherein L is conjugated the one or more alkylene-CO
2H, alkenylene-CO
2H, or alkynylene-CO
2H, or a charged form thereof. In some embodiments, the alkylene-CO
2H, alkenylene-CO
2H, or alkynylene-CO
2H is C
5-24 alkylene-CO
2H, C
5-24 alkenylene-CO
2H, or C
5-24 alkynylene-CO
2, or a charged form thereof. In some embodiments, the alkylene-CO2H, alkenylene-CO2H, or alkynylene-CO2H is C9-24 alkylene-CO2H, C9-24 alkenylene-CO2H, or C9-24 alkynylene-CO2, or a charged form thereof. In some embodiments, the alkylene-CO2H, alkenylene-CO2H, or alkynylene-CO2H is
FH12501620.1
Attorney Docket: DCY-13025 C
15-22 alkylene-CO
2H, C
15-22 alkenylene-CO
2H, or C
15-22 or alkynylene-CO
2H, or a charged form thereof. In some embodiments, L comprises a bivalent or trivalent C
1-C
50 alkylene, wherein 1- 25 methylene groups are optionally and independently replaced by -N(H)-, -N(C
1-C
4 alkyl)-, -N(cycloalkyl)-, -O-, -C(O)-, -C(O)O-, -S-, -S(O)-, -S(O)2-, -S(O)2N(C1-C4 alkyl)-, - S(O)2N(cycloalkyl)-, -N(H)C(O)-, -N(C1-C4 alkyl)C(O)-, -N(cycloalkyl)C(O)-, -C(O)N(H)-, -C(O)N(C1-C4 alkyl), -C(O)N(cycloalkyl), aryl, heteroaryl, cycloalkyl, or cycloalkenyl. In some embodiments, the 1-25 methylene groups are optionally and independently replaced by -C(O)-, -N(H)-, -N(H)C(O)-, -N(C1-C4 alkyl)C(O)-, -O-, or heteroaryl, or combinations thereof. In some embodiments, at least one methylene group is replaced by a 5-12 membered heteroaryl. In some embodiments, the 5-12 membered heteroaryl comprises 1-5 heteroatoms (e.g., 1, 2, 3, 4, or 5, including any values or ranges therebetween) selected from N, O, or S. In some embodiments, the 1-25 methylene groups are optionally and independently replaced by a 5-8 membered heteroaryl with up to 1-3 heteroatoms (e.g., 1, 2, or 3, including any values or ranges therebetween) selected from N, O, or S. In some embodiments, the heteroaryl is a 5 membered heteroaryl with up to 1-3 heteroatoms selected from N, O, or S. In some embodiments, heteroaryl is triazolyl, pyrrolyl, pyrazolyl, imidazolyl, isozazolyl, oxazolyl, isothiazolyl, thiazolyl, pyridinyl, pyrimidinyl, or pyrazinyl. In some embodiments, the heteroaryl is a triazolyl. In some embodiments, the triazolyl is

. In some embodiments, the ligand is L-Y-CO
2H. In some embodiments, the ligand
M is absent, or a bonding group (as described herein) such as NRC(O), or heteroaryl, wherein R is H or alkyl. a is 1, 2, 3, 4, 5, or 6; b is 1, 2, 3, 4, 5, 6, 7, or 8; c is 0, 1, 2, 3, 4, 5, or 6; d is 0, 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, or 20; and e is 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, or 23.
FH12501620.1
Attorney Docket: DCY-13025 In some embodiments, -L-Y-CO
2H is
, wherein: M is absent, or a bonding group (as described herein) such as -NRC(O)-, or heteroaryl, wherein R is H or alkyl. c is 0, 1, 2, 3, 4, 5, or 6; d is 0, 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, or 20; and e is 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, or 23. In some embodiments, M is -NRC(O)-. In some embodiments, R is H or methyl. In some embodiments, R is H. In some embodiments, M is heteroaryl. In some embodiments, the heteroaryl is triazolyl. In some embodiments, the triazolyl is
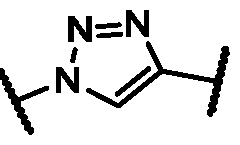
. In some embodiments, M is absent. In some embodiments, c is 1. In some embodiments, d is 0 to 12. In some embodiments, d is 4 to 12. In some embodiments, d is 0. In some embodiments, e is 12 to 23. In some embodiments, e is 11, 15, or 21. In some embodiments, the antisense strand includes an overhang at the 3’ end of 2 to 6 nucleotides (e.g., 2, 3, 4, 5, or 6 nucleotides). In some embodiments, the antisense strand comprises at least one phosphorothioate linkage. In some embodiments, the antisense strand comprises one or more modified nucleotides. In some embodiments, modified nucleotide comprises a modified sugar. In some embodiments, the modified sugar comprises a 2’-OMe group. In some embodiments, the modified sugar comprises a 2’-F substituent. In some embodiments, the antisense strand comprises up to 4 contiguous nucleotides with a 2’-F modified sugar. In some embodiments, the antisense strand comprises a 4’-O-monomethylphosphonate-2’-O-methyl modified nucleotide. In some embodiments, the ligand is conjugated to a ribose of the sense strand at the 3’- or 5’-position. In some embodiments, at least one ligand is conjugated to a ribose of the sense strand at the 2’-position.
FH12501620.1
Attorney Docket: DCY-13025 Formulas In some embodiments, the present disclosure provides an oligonucleotide-ligand conjugate of Formula (AI):
(AI) or a pharmaceutically acceptable salt or charged form thereof, wherein: A and A’ are each independently H or one or more nucleotide; B is a nucleobase; Z is O or S; M is absent, or a moiety formed by conjugating the carboxyl containing component to the remainder of the ligand, such as NRC(O), or heteroaryl, wherein R is H or alkyl. a is 1, 2, 3, 4, 5, or 6; b is 1, 2, 3, 4, 5, 6, 7, or 8; c is 0, 1, 2, 3, 4, 5, or 6; d is 0, 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, or 20; e is 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, or 23; and f is 0, 1, 2, 3, 4, 5, or 6. In some embodiments, the oligonucleotide-ligand conjugate of Formula (AI) has a structure of Formula (AII):
FH12501620.1
Attorney Docket: DCY-13025 (AII), or a pharmaceutically acceptable salt or charged form thereof, wherein A, A’, B, Z, a, b, d, e, and f are defined above in Formula (AI). In some embodiments, the oligonucleotide-ligand conjugate of Formula (AI) has a structure of Formula (AIII):
(AIII), or a pharmaceutically acceptable salt or charged form thereof, wherein A, A’, B, Z, a, b, d, e, and f are defined above in Formula (AI). In some embodiments, the oligonucleotide-ligand conjugate of Formula (AI) or (AIII) has a structure of Formula (AIV):
(AIV), or a pharmaceutically acceptable salt or charged form thereof, wherein A, A’, B, Z, a, b, e, and f are defined above in Formula (AI). In some embodiments, the oligonucleotide-ligand conjugate of Formula (AI) has a structure of Formula (AV):
FH12501620.1
Attorney Docket: DCY-13025 (AV), or a pharmaceutically acceptable salt or charged form thereof, wherein A, A’, B, Z, a, b, d, and f are defined above in Formula (AI), and wherein e is 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, or 23. In some embodiments, the present disclosure provides an oligonucleotide-ligand conjugate of Formula (BI)
(BI), or a pharmaceutically acceptable salt or charged form thereof, wherein: A is one or more nucleotide; B is a nucleobase; Z
1 and Z
2 are each independently O or S; M is absent, NRC(O), or heteroaryl, wherein R is H or alkyl. a is 1, 2, 3, 4, or 5; c is 0, 1, 2, 3, 4, 5, or 6; d1 and d2 is 0, 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, or e is 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, or 23; and f is 0, 1, 2, 3, 4, 5, or 6. In some embodiments, the present disclosure provides an oligonucleotide-ligand conjugate of Formula (BII):
FH12501620.1
Attorney Docket: DCY-13025
(BII), or a salt or charged form thereof, wherein B, A, M, a, c, d1, d2, e, and f are defined above in Formula (BI), and wherein Z is O or S. In some embodiments, the present disclosure provides an oligonucleotide-ligand conjugate of Formula (CI):
(CI), or a pharmaceutically acceptable salt or charged form thereof, wherein: A is one or more nucleotide; B is a nucleobase; Z
1 and Z
2 are each independently O or S; M is absent, NRC(O), or heteroaryl, wherein R is H or alkyl. a is 1, 2, 3, 4, or 5; c is 0, 1, 2, 3, 4, 5, or 6; d1 and d2 are each independently 0, 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, or 20; e is 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, or 23; and f is 0, 1, 2, 3, 4, 5, or 6. In some embodiments, the present disclosure provides an oligonucleotide-ligand conjugate of Formula (CII):
FH12501620.1
Attorney Docket: DCY-13025

(CII), or a salt or charged form thereof, wherein B, A, M, a, c, d1, d2, e, and f are defined above in Formula (CI), and wherein Z is O or S. M In some embodiments of the oligonucleotide-ligand conjugate of Formula (AI), (AII), (BI), (BII), (CI), or (CII), M is any suitable group (e.g., a reaction product) that couples the hydrocarbon bearing one or more carboxyl groups to the linker (L). In some embodiments of the oligonucleotide-ligand conjugate of Formula (AI), (AII), (BI), (BII), (CI), or (CII), M is -NRC(O)-. In some embodiments of the oligonucleotide-ligand conjugate of Formula (AI), (AII), (BI), (BII), (CI), or (CII), R is H. In some embodiments of the oligonucleotide-ligand conjugate of Formula (AI), (BI), (BII), (CI), or (CII), R is alkyl. In some embodiments, R is C1-6 alkyl. In some embodiments, R is methyl. In some embodiments, R is ethyl. In some embodiments of the oligonucleotide-ligand conjugate of Formula (AI), (AIII) (BI), (BII), (CI), or (CII), M is heteroaryl. In some embodiments M is a 5-12 membered heteroaryl. In some embodiments, M is a 5-12 membered heteroaryl containing 1-5 heteroatoms (e.g., 1, 2, 3, 4, or 5, including any ranges therebetween) selected from N, O, or S. In some embodiments, M is a 5-8 membered heteroaryl containing 1-3 heteroatoms (e.g., 1, 2, or 3, including any values or ranges therebetween) selected from N, O, or S. In some embodiments, M is a 5-7 membered heteroaryl containing 1-3 heteroatoms selected from N, O, or S. In some embodiments, M is triazolyl, pyrrolyl, pyrazolyl, imidazolyl, isozazolyl, oxazolyl, isothiazolyl, thiazolyl, pyridinyl, pyrimidinyl, or pyrazinyl. In some embodiments, M is triazolyl. In some embodiments of the oligonucleotide-ligand conjugate of Formula

In some embodiments of the oligonucleotide-ligand conjugate of Formula (AI), (AV), (BI), (BII), (CI), or (CII), M is absent.
FH12501620.1
Attorney Docket: DCY-13025 Additional examples of M groups include:
, , ,
B In some embodiments of the oligonucleotide-ligand conjugate of Formulas (AI), (AII), (AIII), (AIV), (AV), (BI), (BII), (CI), or (CII), B is a natural or non-natural base. In some embodiments, the base is
FH12501620.1
Attorney Docket: DCY-13025
some embodiments, B is . In some embodiments, B is . In some embodiments,

Z In some embodiments of the oligonucleotide-ligand conjugate of Formulas (AI), (AII), (AIII), (AIV), (AV), (BI), (BII), (CI), or (CII), Z is O. In some embodiments, Z is S. a, b, c, d, e, f In some embodiments of the oligonucleotide-ligand conjugate of Formulas (AI), (AII), (AIII), (AIV), (AV), (BI), (BII), (CI), or (CII), a is 1. In some embodiments, a is 2. In some embodiments, a is 3. In some embodiments, a is 4. In some embodiments, a is 5. In some embodiments, a is 6. In some embodiments, a is 1 or 2. In some embodiments of the oligonucleotide-ligand conjugate of Formulas (AI), (AII), (AIII), (AIV), or (AV), b is 1. In some embodiments, b is 2. In some embodiments, b is 3. In some embodiments, b is 4. In some embodiments, b is 5. In some embodiments, b is 6. In some embodiments, b is 1 or 2. In some embodiments, c is 0. In some embodiments, c is 1. In some embodiments, c is 2. In some embodiments, c is 3. In some embodiments, c is 4. In some embodiments, c is 5. In some embodiments, c is 6. In some embodiments of the oligonucleotide-ligand conjugate of Formulas (AI), (AII), (AIII), (AIV), or (AV), d is 0. In some embodiments, d is 1. In some embodiments, d is 2. In some embodiments, d is 3. In some embodiments, d is 4. In some embodiments, d is 5. In some embodiments, d is 6. In some embodiments, d is 7. In some embodiments, d is 8. In some embodiments, d is 9. In some embodiments, d is 10. In some embodiments, d is 11. In
FH12501620.1
Attorney Docket: DCY-13025 some embodiments, d is 12. In some embodiments, d is 13. In some embodiments, d is 14. In some embodiments, d is 15. In some embodiments, d is 16. In some embodiments, d is 17. In some embodiments, d is 18. In some embodiments, d is 19. In some embodiments, d is 20. In some embodiments, d is 0, 4, or 12. In some embodiments, d is 0 or 12. In some embodiments of the oligonucleotide-ligand conjugate of Formulas (BI), (BII), (CI), or (CII), d1 is 0. In some embodiments, d1 is 1. In some embodiments, d1 is 2. In some embodiments, d1 is 3. In some embodiments, d1 is 4. In some embodiments, d1 is 5. In some embodiments, d1 is 6. In some embodiments, d1 is 7. In some embodiments, d1 is 8. In some embodiments, d1 is 9. In some embodiments, d1 is 10. In some embodiments, d1 is 11. In some embodiments, d1 is 12. In some embodiments, d1 is 13. In some embodiments, d1 is 14. In some embodiments, d1 is 15. In some embodiments, d1 is 16. In some embodiments, d1 is 17. In some embodiments, d1 is 18. In some embodiments, d1 is 19. In some embodiments, d1 is 20. In some embodiments of the oligonucleotide-ligand conjugate of Formulas (BI), (BII), (CI), or (CII), d
2 is 0. In some embodiments, d
2 is 1. In some embodiments, d
2 is 2. In some embodiments, d
2 is 3. In some embodiments, d
2 is 4. In some embodiments, d
2 is 5. In some embodiments, d2 is 6. In some embodiments, d2 is 7. In some embodiments, d2 is 8. In some embodiments, d
2 is 9. In some embodiments, d
2 is 10. In some embodiments, d
2 is 11. In some embodiments, d2 is 12. In some embodiments, d2 is 13. In some embodiments, d2 is 14. In some embodiments, d2 is 15. In some embodiments, d2 is 16. In some embodiments, d2 is 17. In some embodiments, d2 is 18. In some embodiments, d2 is 19. In some embodiments, d2 is 20. In some embodiments of the oligonucleotide-ligand conjugate of Formulas (AI), (AII), (AIII), (AIV), (AV), (BI), (BII), (CI), or (CII), e is 5. In some embodiments, e is 6. In some embodiments, e is 7. In some embodiments, e is 8. In some embodiments, e is 9. In some embodiments, e is 10. In some embodiments, e is 11. In some embodiments, e is 12. In some embodiments, e is 13. In some embodiments, e is 14. In some embodiments, e is 15. In some embodiments, e is 16. In some embodiments, e is 17. In some embodiments, e is 18. In some embodiments, e is 19. In some embodiments, e is 20. In some embodiments, e is 21. In some embodiments, e is 22. In some embodiments, e is 23. In some embodiments, e is 11 or 22. In some embodiments of the oligonucleotide-ligand, e is 7, 15, or 21. In some embodiments of the oligonucleotide-ligand conjugate of Formulas (AI), (AII), (AIII), (AIV), (AV), (BI), (BII), (CI), or (CII), e is 11, 15, or 21.
FH12501620.1
Attorney Docket: DCY-13025 In some embodiments of the oligonucleotide-ligand conjugate of Formulas (AI), (AII), (AIII), (AIV), (AV), (BI), (BII), (CI), or (CII), f is 0. In some embodiments, f is 1. In some embodiments, f is 2. In some embodiments, f is 3. In some embodiments, f is 4. In some embodiments, f is 5. In some embodiments, f is 6. In some embodiments f is 0, 1, or 2. A and A^ In some embodiments of the oligonucleotide-ligand conjugate of Formulas (AI), (AII), (AIII), (AIV), (AV), (BI), (BII), (CI), or (CII), A is H. In some embodiments of the oligonucleotide-ligand conjugate of Formulas (AI), (AII), (AIII), (AIV), or (AV) A^ is H. In some embodiments, A is 13 to 40 nucleotides in length. In some embodiments, A is 13 to 36 nucleotides in length. In some embodiments, A is 12 to 29 nucleotides in length. In some embodiments, A is 15 to 20 nucleotides in length. In some embodiments, A is 13 nucleotides in length. In some embodiments, A is 14 nucleotides in length. In some embodiments, A is 15 nucleotides in length. In some embodiments, A is 16 nucleotides in length. In some embodiments, A is 17 nucleotides in length. In some embodiments, A is 18 nucleotides in length. In some embodiments, A is 19 nucleotides in length. In some embodiments, A is 20 nucleotides in length. In some embodiments, A is 21 nucleotides in length. In some embodiments, A is 22 nucleotides in length. In some embodiments, A is 14 nucleotides in length. In some embodiments, A is 23 nucleotides in length. In some embodiments, A is 24 nucleotides in length. In some embodiments, A is 25 nucleotides in length. In some embodiments, A is 26 nucleotides in length. In some embodiments, A is 27 nucleotides in length. In some embodiments, A is 28 nucleotides in length. In some embodiments, A is 29 nucleotides in length. In some embodiments, A is 30 nucleotides in length. In some embodiments, A is 31 nucleotides in length. In some embodiments, A is 32 nucleotides in length. In some embodiments, A is 33 nucleotides in length. In some embodiments, A is 34 nucleotides in length. In some embodiments, A is 35 nucleotides in length. In some embodiments, A is 36 nucleotides in length. In some embodiments, A is 37 nucleotides in length. In some embodiments, A is 38 nucleotides in length. In some embodiments, A is 39 nucleotides in length. In some embodiments, A is 40 nucleotides in length. In some embodiments, A^ is 13 to 40 nucleotides in length. In some embodiments, A^ is 13 to 36 nucleotides in length. In some embodiments, A^ is 15 to 20 nucleotides in length. In some embodiments, A^ is 13 nucleotides in length. In some embodiments, A^ is 14 nucleotides in length. In some embodiments, A^ is 15 nucleotides in length. In some embodiments, A^ is
FH12501620.1
Attorney Docket: DCY-13025 16 nucleotides in length. In some embodiments, A^ is 17 nucleotides in length. In some embodiments, A^ is 18 nucleotides in length. In some embodiments, A^ is 19 nucleotides in length. In some embodiments, A is 20 nucleotides in length. In some embodiments, A^ is 21 nucleotides in length. In some embodiments, A^ is 22 nucleotides in length. In some embodiments, A^ is 14 nucleotides in length. In some embodiments, A^ is 23 nucleotides in length. In some embodiments, A^ is 24 nucleotides in length. In some embodiments, A^ is 25 nucleotides in length. In some embodiments, A^ is 26 nucleotides in length. In some embodiments, A^ is 27 nucleotides in length. In some embodiments, A^ is 28 nucleotides in length. In some embodiments, A^ is 29 nucleotides in length. In some embodiments, A^ is 30 nucleotides in length. In some embodiments, A^ is 31 nucleotides in length. In some embodiments, A^ is 32 nucleotides in length. In some embodiments, A^ is 33 nucleotides in length. In some embodiments, A^ is 34 nucleotides in length. In some embodiments, A^ is 35 nucleotides in length. In some embodiments, A^ is 36 nucleotides in length. In some embodiments, A^ is 37 nucleotides in length. In some embodiments, A^ is 38 nucleotides in length. In some embodiments, A^ is 39 nucleotides in length. In some embodiments, A^ is 40 nucleotides in length. In some embodiments, A is 12-36 nucleotides in length and A^ is 1-10 nucleotides in length. In some embodiments, A is 12-29 nucleotides in length and A^ is 1-10 nucleotides in length. In some embodiments, A is 1-10 nucleotides in length and A^ is 12-29 nucleotides in length. In some embodiments, A is 13 to 40 nucleotides in length and A^ is H. In some embodiments, A is 13 to 20 nucleotides in length and A^ is H. In some embodiments, A is 18 to 30 nucleotides in length and A^ is H. In some embodiments, A is 27 nucleotides in length and A^ is 8 nucleotides in length. In some embodiments, A is 28 nucleotides in length and A^ is 8 nucleotides in length. In some embodiments, A is H and A^ is 12 to 40 nucleotides in length. In some embodiments, A is H and A^ is 36 nucleotides in length. In some embodiments, A is H and A^ is 19 nucleotides in length. In some embodiments, an RNAi oligonucleotide comprising a sense strand and an antisense strand comprises an oligonucleotide-ligand conjugate of any one Formulas (AI), (AII), (AIII), (AIV), (AV), (BI), (BII), (CI), or (CII). In some embodiments, an RNAi oligonucleotide comprising a sense strand and an antisense strand comprises at least one oligonucleotide-ligand conjugate of any one of Formulas (AI), (AII), (AIII), (AIV), (AV), (BI), (BII), (CI), or (CII). In some embodiments, an RNAi oligonucleotide comprising a sense strand and an antisense strand comprises at least two oligonucleotide-ligand conjugates of any one of Formulas (AI), (AII), (AIII), (AIV), (AV), (BI), (BII), (CI), or (CII).
FH12501620.1
Attorney Docket: DCY-13025 In some embodiments, an RNAi oligonucleotide comprises a sense and antisense strand, wherein the sense strand comprises an oligonucleotide-ligand conjugate of Formula (AI), wherein A is H and A^ is 19 nucleotides, and wherein the antisense strand is 15 to 30 nucleotides in length. In some embodiments, an RNAi oligonucleotide comprises a sense and antisense strand, wherein the sense strand comprises an oligonucleotide-ligand conjugate of Formula (AI), wherein A is H and A^ is 19 nucleotides, and wherein the antisense strand is 22 nucleotides in length. In some embodiments, an RNAi oligonucleotide comprises a sense and antisense strand, wherein the sense strand comprises an oligonucleotide-ligand conjugate of Formula (AI), wherein A is 27 nucleotides and A^ is 8 nucleotides, and wherein the antisense strand is 15 to 30 nucleotides in length. In some embodiments, an RNAi oligonucleotide comprises a sense and antisense strand, wherein the sense strand comprises an oligonucleotide-ligand conjugate of Formula (AI), wherein A is 27 nucleotides and A^ is 8 nucleotides, and wherein the antisense strand is 22 nucleotides in length. In some embodiments, an RNAi oligonucleotide comprises a sense and antisense strand, wherein the sense strand comprises an oligonucleotide-ligand conjugate of Formula (AI), wherein A is 28 nucleotides and A^ is 8 nucleotides, and wherein the antisense strand is 15 to 30 nucleotides in length. In some embodiments, an RNAi oligonucleotide comprises a sense and antisense strand, wherein the sense strand comprises an oligonucleotide-ligand conjugate of Formula (AI), wherein A is 28 nucleotides and A^ is 8 nucleotides, and wherein the antisense strand is 22 nucleotides in length. In some embodiments, an RNAi oligonucleotide comprises a sense and antisense strand, wherein the sense strand comprises an oligonucleotide-ligand conjugate of Formula (AII), wherein A is H and A^ is 19 nucleotides, and wherein the antisense strand is 22 nucleotides in length. In some embodiments, an RNAi oligonucleotide comprises a sense and antisense strand, wherein the sense strand comprises an oligonucleotide-ligand conjugate of Formula (AII), wherein A is 27 nucleotides and A^ is 8 nucleotides, and wherein the antisense strand is 22 nucleotides in length. In some embodiments, an RNAi oligonucleotide comprises a sense and antisense strand, wherein the sense strand comprises an oligonucleotide-ligand conjugate of Formula (AII), wherein A is 28 nucleotides and A^ is 8 nucleotides, and wherein the antisense strand is 22 nucleotides in length. In some embodiments, an RNAi oligonucleotide comprises a sense and antisense strand, wherein the sense strand comprises an oligonucleotide-ligand conjugate of Formula (AIII), wherein A is H and A^ is 19 nucleotides, and wherein the antisense strand is 22 nucleotides in length. In some embodiments, an RNAi oligonucleotide comprises a sense and
FH12501620.1
Attorney Docket: DCY-13025 antisense strand, wherein the sense strand comprises an oligonucleotide-ligand conjugate of Formula (AIII), wherein A is 27 nucleotides and A^ is 8 nucleotides, and wherein the antisense strand is 22 nucleotides in length. In some embodiments, an RNAi oligonucleotide comprises a sense and antisense strand, wherein the sense strand comprises an oligonucleotide-ligand conjugate of Formula (AIII), wherein A is 28 nucleotides and A^ is 8 nucleotides, and wherein the antisense strand is 22 nucleotides in length. In some embodiments, an RNAi oligonucleotide comprises a sense and antisense strand, wherein the sense strand comprises an oligonucleotide-ligand conjugate of Formula (AIV), wherein A is H and A^ is 19 nucleotides, and wherein the antisense strand is 22 nucleotides in length. In some embodiments, an RNAi oligonucleotide comprises a sense and antisense strand, wherein the sense strand comprises an oligonucleotide-ligand conjugate of Formula (AIV), wherein A is 27 nucleotides and A^ is 8 nucleotides, and wherein the antisense strand is 22 nucleotides in length. In some embodiments, an RNAi oligonucleotide comprises a sense and antisense strand, wherein the sense strand comprises an oligonucleotide-ligand conjugate of Formula (AIV), wherein A is 28 nucleotides and A^ is 8 nucleotides, and wherein the antisense strand is 22 nucleotides in length. In some embodiments, an RNAi oligonucleotide comprises a sense and antisense strand, wherein the sense strand comprises an oligonucleotide-ligand conjugate of Formula (AV), wherein A is H and A^ is 19 nucleotides, and wherein the antisense strand is 22 nucleotides in length. In some embodiments, an RNAi oligonucleotide comprises a sense and antisense strand, wherein the sense strand comprises an oligonucleotide-ligand conjugate of Formula (AV), wherein A is 27 nucleotides and A^ is 8 nucleotides, and wherein the antisense strand is 22 nucleotides in length. In some embodiments, an RNAi oligonucleotide comprises a sense and antisense strand, wherein the sense strand comprises an oligonucleotide-ligand conjugate of Formula (AV), wherein A is 28 nucleotides and A^ is 8 nucleotides, and wherein the antisense strand is 22 nucleotides in length. In some embodiments, the RNAi oligonucleotide comprises a duplex region. In some embodiments, the duplex region includes one or more phosphorothioate linkages (e.g., 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, or more phosphorothioate linkages). In some embodiments, the duplex region includes two phosphorothioate linkages. In some embodiments, the duplex region includes three phosphorothioate linkages. In some embodiments, the duplex region includes four phosphorothioate linkages. In some embodiments, the duplex region includes two or more phosphorothioate linkages, and two phosphorothioate linkages are adjacent to each other. In some embodiments, the duplex region includes two phosphorothioate linkages, and
FH12501620.1
Attorney Docket: DCY-13025 the two phosphorothioate linkages are adjacent to each other. In some embodiments, the duplex region comprises 20 to 30 base pairs, including 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, or 30 base pairs, including any values or ranges therebetween. In some embodiments, the RNAi oligonucleotide comprises a sense strand and an antisense strand, wherein the sense strand comprises an oligonucleotide-ligand conjugate of any of Formula (AI), (AII), (AIII), (AIV), and (AV), wherein A and A^ comprise at least 14 nucleotides, and wherein the oligonucleotide-ligand conjugate and the at least 14 nucleotides of A and A^ form a duplex region with the antisense strand. In some embodiments, the RNAi oligonucleotide comprises a sense strand and an antisense strand, wherein the sense strand comprises an oligonucleotide-ligand conjugate of any of Formula (AI), (AII), (AIII), (AIV), and (AV), wherein A comprises at least 14 nucleotides, and wherein the oligonucleotide-ligand conjugate and the at least 14 nucleotides of A form a duplex region with the antisense strand. In some embodiments, the RNAi oligonucleotide comprises a sense strand and an antisense strand, wherein the sense strand comprises an oligonucleotide-ligand conjugate of any of Formula (AI), (AII), (AIII), (AIV), and (AV), wherein A^ comprises at least 14 nucleotides, and wherein the oligonucleotide-ligand conjugate and the at least 14 nucleotides of A^ form a duplex region with the antisense strand. In some embodiments, the RNAi oligonucleotide comprises a sense strand and an antisense strand, wherein the sense strand comprises an oligonucleotide-ligand conjugate of any of Formula (AI), (AII), (AIII), (AIV), and (AV), wherein A and A^ comprise at least 15 nucleotides, and wherein the oligonucleotide-ligand conjugate and the at least 15 nucleotides of A and A^ form a duplex region with the antisense strand. In some embodiments, the RNAi oligonucleotide comprises a sense strand and an antisense strand, wherein the sense strand comprises an oligonucleotide-ligand conjugate of any of Formula (AI), (AII), (AIII), (AIV), and (AV), wherein A comprises at least 15 nucleotides, and wherein the oligonucleotide-ligand conjugate and the at least 15 nucleotides of A form a duplex region with the antisense strand. In some embodiments, the RNAi oligonucleotide comprises a sense strand and an antisense strand, wherein the sense strand comprises an oligonucleotide-ligand conjugate of any of Formula (AI), (AII), (AIII), (AIV), and (AV), wherein A^ comprises at least 15 nucleotides, and wherein the oligonucleotide-ligand conjugate and the at least 15 nucleotides of A^ form a duplex region with the antisense strand. In some embodiments, the RNAi oligonucleotide comprises a sense strand and an antisense strand, wherein the sense strand comprises an oligonucleotide-ligand conjugate of any of Formula (AI), (AII), (AIII), (AIV), and (AV), wherein A and A^ comprise at least 16 nucleotides, and wherein the oligonucleotide-ligand conjugate and the at least 16 nucleotides
FH12501620.1
Attorney Docket: DCY-13025 of A and A^ form a duplex region with the antisense strand. In some embodiments, the RNAi oligonucleotide comprises a sense strand and an antisense strand, wherein the sense strand comprises an oligonucleotide-ligand conjugate of any of Formula (AI), (AII), (AIII), (AIV), and (AV), wherein A comprises at least 16 nucleotides, and wherein the oligonucleotide-ligand conjugate and the at least 16 nucleotides of A form a duplex region with the antisense strand. In some embodiments, the RNAi oligonucleotide comprises a sense strand and an antisense strand, wherein the sense strand comprises an oligonucleotide-ligand conjugate of any of Formula (AI), (AII), (AIII), (AIV), and (AV), wherein A^ comprises at least 16 nucleotides, and wherein the oligonucleotide-ligand conjugate and the at least 16 nucleotides of A^ form a duplex region with the antisense strand. In some embodiments, the RNAi oligonucleotide comprises a sense strand and an antisense strand, wherein the sense strand comprises an oligonucleotide-ligand conjugate of any of Formula (AI), (AII), (AIII), (AIV), and (AV), wherein A and A^ comprise at least 17 nucleotides, and wherein the oligonucleotide-ligand conjugate and the at least 17 nucleotides of A and A^ form a duplex region with the antisense strand. In some embodiments, the RNAi oligonucleotide comprises a sense strand and an antisense strand, wherein the sense strand comprises an oligonucleotide-ligand conjugate of any of Formula (AI), (AII), (AIII), (AIV), and (AV), wherein A comprises at least 17 nucleotides, and wherein the oligonucleotide-ligand conjugate and the at least 17 nucleotides of A form a duplex region with the antisense strand. In some embodiments, the RNAi oligonucleotide comprises a sense strand and an antisense strand, wherein the sense strand comprises an oligonucleotide-ligand conjugate of any of Formula (AI), (AII), (AIII), (AIV), and (AV), wherein A^ comprises at least 17 nucleotides, and wherein the oligonucleotide-ligand conjugate and the at least 17 nucleotides of A^ form a duplex region with the antisense strand. In some embodiments, the RNAi oligonucleotide comprises a sense strand and an antisense strand, wherein the sense strand comprises an oligonucleotide-ligand conjugate of any of Formula (AI), (AII), (AIII), (AIV), and (AV), wherein A and A^ comprise at least 18 nucleotides, and wherein the oligonucleotide-ligand conjugate and the at least 18 nucleotides of A and A^ form a duplex region with the antisense strand. In some embodiments, the RNAi oligonucleotide comprises a sense strand and an antisense strand, wherein the sense strand comprises an oligonucleotide-ligand conjugate of any of Formula (AI), (AII), (AIII), (AIV), and (AV), wherein A comprises at least 18 nucleotides, and wherein the oligonucleotide-ligand conjugate and the at least 18 nucleotides of A form a duplex region with the antisense strand. In some embodiments, the RNAi oligonucleotide comprises a sense strand and an antisense strand, wherein the sense strand comprises an oligonucleotide-ligand conjugate of any of
FH12501620.1
Attorney Docket: DCY-13025 Formula (AI), (AII), (AIII), (AIV), and (AV), wherein A^ comprises at least 18 nucleotides, and wherein the oligonucleotide-ligand conjugate and the at least 18 nucleotides of A^ form a duplex region with the antisense strand. In some embodiments, the RNAi oligonucleotide comprises a sense strand and an antisense strand, wherein the sense strand comprises an oligonucleotide-ligand conjugate of any of Formula (AI), (AII), (AIII), (AIV), and (AV), wherein A and A^ comprise at least 19 nucleotides, and wherein the oligonucleotide-ligand conjugate and the at least 19 nucleotides of A and A^ form a duplex region with the antisense strand. In some embodiments, the RNAi oligonucleotide comprises a sense strand and an antisense strand, wherein the sense strand comprises an oligonucleotide-ligand conjugate of any of Formula (AI), (AII), (AIII), (AIV), and (AV), wherein A comprises at least 19 nucleotides, and wherein the oligonucleotide-ligand conjugate and the at least 19 nucleotides of A form a duplex region with the antisense strand. In some embodiments, the RNAi oligonucleotide comprises a sense strand and an antisense strand, wherein the sense strand comprises an oligonucleotide-ligand conjugate of any of Formula (AI), (AII), (AIII), (AIV), and (AV), wherein A^ comprises at least 19 nucleotides, and wherein the oligonucleotide-ligand conjugate and the at least 19 nucleotides of A^ form a duplex region with the antisense strand. In some embodiments, the RNAi oligonucleotide comprises a sense strand and an antisense strand, wherein the sense strand comprises an oligonucleotide-ligand conjugate of any of Formula (BI), (BII), (CI), and (CII), wherein A comprises at least 14 nucleotides, and wherein the oligonucleotide-ligand conjugate and the at least 14 nucleotides of A form a duplex region with the antisense strand. In some embodiments, the RNAi oligonucleotide comprises a sense strand and an antisense strand, wherein the sense strand comprises an oligonucleotide- ligand conjugate of any of Formula (BI), (BII), (CI), and (CII), wherein A comprises at least 15 nucleotides, and wherein the oligonucleotide-ligand conjugate and the at least 15 nucleotides of A form a duplex region with the antisense strand. In some embodiments, the RNAi oligonucleotide comprises a sense strand and an antisense strand, wherein the sense strand comprises an oligonucleotide-ligand conjugate of any of Formula (BI), (BII), (CI), and (CII), wherein A comprises at least 16 nucleotides, and wherein the oligonucleotide-ligand conjugate and the at least 16 nucleotides of A form a duplex region with the antisense strand. In some embodiments, the RNAi oligonucleotide comprises a sense strand and an antisense strand, wherein the sense strand comprises an oligonucleotide-ligand conjugate of any of Formula (BI), (BII), (CI), and (CII), wherein A comprises at least 17 nucleotides, and wherein the oligonucleotide-ligand conjugate and the at least 17 nucleotides of A form a duplex region with the antisense strand. In some embodiments, the RNAi oligonucleotide comprises a sense
FH12501620.1
Attorney Docket: DCY-13025 strand and an antisense strand, wherein the sense strand comprises an oligonucleotide-ligand conjugate of any of Formula (BI), (BII), (CI), and (CII), wherein A comprises at least 18 nucleotides, and wherein the oligonucleotide-ligand conjugate and the at least 18 nucleotides of A form a duplex region with the antisense strand. In some embodiments, the RNAi oligonucleotide comprises a sense strand and an antisense strand, wherein the sense strand comprises an oligonucleotide-ligand conjugate of any of Formula (BI), (BII), (CI), and (CII), wherein A comprises at least 19 nucleotides, and wherein the oligonucleotide-ligand conjugate and the at least 19 nucleotides of A form a duplex region with the antisense strand. In some embodiments the RNAi oligonucleotide comprises a loop region. In some embodiments, the RNAi oligonucleotide comprises a loop region, wherein the loop comprises an oligonucleotide-ligand conjugate of any of Formulas (AI), (AII), (AIII), (AIV), (AV), (BI), (BII), (CI), or (CII). In some embodiments, the RNAi oligonucleotide comprises a loop region, wherein the loop is a tetraloop comprising an oligonucleotide-ligand conjugate of any of Formulas (AI), (AII), (AIII), (AIV), (AV), (BI), (BII), (CI), or (CII).In some embodiments, an oligonucleotide-ligand conjugate of any of Formulas (AI), (AII), (AIII), (AIV), (AV), (BI), (BII), (CI), or (CII) is position 1 of the tetraloop. In some embodiments, an oligonucleotide-ligand conjugate of any of Formulas (AI), (AII), (AIII), (AIV), (AV), (BI), (BII), (CI), or (CII) is position 2 of the tetraloop. In some embodiments, an oligonucleotide- ligand conjugate of any of Formulas (AI), (AII), (AIII), (AIV), (AV), (BI), (BII), (CI), or (CII) is position 3 of the tetraloop. In some embodiments, an oligonucleotide-ligand conjugate of any of Formulas (AI), (AII), (AIII), (AIV), (AV), (BI), (BII), (CI), or (CII) is position 4 of the tetraloop. In some embodiments, the tetraloop region comprises a 5’- GAAA-3’ sequence. In some embodiments, an RNAi oligonucleotide comprising an oligonucleotide- ligand conjugate of Formulas (AI), (AII), (AIII), (AIV), (AV), (BI), (BII), (CI), or (CII) comprises one or more modified nucleotides, e.g., as described herein. In some embodiments, the modified nucleotide comprises a modified sugar. In some embodiments, the modified sugar comprises a 2’-OMe substituent. In some embodiments, the modified sugar comprises a 2’-F substituent. In some embodiments, the modified nucleotide comprises a modified nucleobase. In some embodiments, the modified nucleotide is a 4’-O- monomethylphosphonate-2’-O-methyl modified nucleotide. In some embodiments of the oligonucleotide-ligand conjugate of Formulas (AI), (AII), (AIII), (AIV), (AV), (BI), (BII), (CI), or (CII), wherein A is at least one nucleotide, one or more nucleotides of A is a modified nucleotide. In some embodiments of the
FH12501620.1
Attorney Docket: DCY-13025 oligonucleotide-ligand conjugate of Formulas (AI), (AII), (AIII), (AIV), (AV), (BI), (BII), (CI), or (CII), wherein A is 13 to 40 nucleotides, one or more nucleotides of A is a modified nucleotide. In some embodiments of the oligonucleotide-ligand conjugate of Formulas (AI), (AII), (AIII), (AIV), (AV), (BI), (BII), (CI), or (CII), wherein A is 13 to 40 nucleotides, one or more nucleotides of A comprises a modified sugar. In some embodiments of the oligonucleotide-ligand conjugate of Formulas (AI), (AII), (AIII), (AIV), (AV), (BI), (BII), (CI), or (CII), wherein A is 13 to 40 nucleotides, one or more nucleotides of A comprises a 2’-OMe substituent. In some embodiments of the oligonucleotide-ligand conjugate of Formulas (AI), (AII), (AIII), (AIV), (AV), (BI), (BII), (CI), or (CII), wherein A is 13 to 40 nucleotides, one or more nucleotides of A comprises a 2’-F substituent. In some embodiments of the oligonucleotide-ligand conjugate of Formulas (AI), (AII), (AIII), (AIV), (AV), (BI), (BII), (CI), or (CII), wherein A is 13 to 40 nucleotides, every nucleotide of A is a modified nucleotide. In some embodiments of the oligonucleotide-ligand conjugate of Formulas (AI), (AII), or (AIII), wherein A^ is at least one nucleotide, one or more nucleotides of A^ is a modified nucleotide. In some embodiments of the oligonucleotide-ligand conjugate of Formulas (AI), (AII), or (AIII), wherein A is 13 to 40 nucleotides, one or more nucleotides of A^ is a modified nucleotide. In some embodiments of the oligonucleotide-ligand conjugate of Formulas (AI), (AII), or (AIII), wherein A is 13 to 40 nucleotides, one or more nucleotides of A^ comprises a modified sugar. In some embodiments of the oligonucleotide-ligand conjugate of Formulas (AI), (AII), or (AIII), wherein A is 13 to 40 nucleotides, one or more nucleotides of A^ comprises a 2’-OMe substituent. In some embodiments of the oligonucleotide-ligand conjugate of Formulas (AI), (AII), or (AIII), wherein A is 13 to 40 nucleotides, one or more nucleotides of A^ comprises a 2’-F substituent. In some embodiments of the oligonucleotide-ligand conjugate of Formulas (AI), (AII), or (AIII), wherein A^ is 13 to 40 nucleotides, every nucleotide of A^ is a modified nucleotide. In some embodiments of the oligonucleotide-ligand conjugate of Formulas (AI), (AII), (AIII), (AIV), (AV), (BI), (BII), (CI), or (CII), the conjugate further comprises one or more ligands having the structure -L-Y-(CO
2H)
n as described herein. In some embodiments of the oligonucleotide-ligand conjugate of Formulas (AI), (AII), (AIII), (AIV), (AV), (BI), (BII), (CI), or (CII), the ligand is conjugated to a ribose of the sense strand at the 2’-position. In some embodiments, ligand is conjugated to a ribose of the sense strand at the 3’- or 5’-position.
FH12501620.1
Attorney Docket: DCY-13025 In some embodiments, the present disclosure provides an oligonucleotide-ligand conjugate comprises two or more of (i), (ii), or (iii): (i) Formula (AI), (AII), (AIII), (AIV), or (AV); (ii) Formula (BI) or (BII); and (iii) Formula (CI) or (CII). RNAi Oligonucleotide Targeting Sequences In some embodiments, the RNAi oligonucleotides provided by the disclosure comprise a targeting sequence. As used herein, the term “targeting sequence” refers to a nucleotide sequence having a region of complementarity to a specific nucleotide sequence comprising an mRNA. In some embodiments, the RNAi oligonucleotides provided by the disclosure comprise a gene targeting sequence having a region of complementarity to a nucleotide sequence comprising a target sequence of a target mRNA. The targeting sequence imparts the RNAi oligonucleotides with the ability to specifically target an mRNA by binding or annealing to a target sequence comprising a target mRNA by complementary (Watson-Crick) base pairing. In some embodiments, the RNAi oligonucleotides herein (or a strand thereof, e.g., an antisense strand or a guide strand of a double-stranded oligonucleotide) comprise a targeting sequence having a region of complementarity that binds or anneals to a target sequence comprising a target mRNA by complementary (Watson-Crick) base pairing. In some embodiments, the RNAi oligonucleotides herein (or a strand thereof, e.g., an antisense strand or a guide strand of a double-stranded oligonucleotide) comprise a targeting sequence having a region of complementarity that binds or anneals to a target sequence within a target mRNA by complementary (Watson-Crick) base pairing. The targeting sequence is generally of suitable length and base content to enable binding or annealing of the RNAi oligonucleotide (or a strand thereof) to a specific target mRNA for purposes of inhibiting target gene expression. In some embodiments, the targeting sequence is at least about 12, at least about 13, at least about 14, at least about 15, at least about 16, at least about 17, at least about 18, at least about 19, at least about 20, at least about 21, at least about 22, at least about 23, at least about 24, at least about 25, at least about 26, at least about 27, at least about 28, at least about 29 or at least about 30 nucleotides in length. In some embodiments, the targeting sequence is at least 12, at least 13, at least 14, at least 15, at least 16, at least 17, at least 18, at least 19 or at least 20 nucleotides. In some embodiments, the targeting sequence is about 12 to about 30 (e.g., 12 to 30, 12 to 22, 15 to 25, 17 to 21, 18 to 27, 19 to 27, or 15 to 30) nucleotides in length. In some embodiments, the targeting sequence is about 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29 or 30 nucleotides in length. In some embodiments, the targeting sequence is 18 nucleotides
FH12501620.1
Attorney Docket: DCY-13025 in length. In some embodiments, the targeting sequence is 19 nucleotides in length. In some embodiments, the targeting sequence is 20 nucleotides in length. In some embodiments, the targeting sequence is 21 nucleotides in length. In some embodiments, the targeting sequence is 22 nucleotides in length. In some embodiments, the targeting sequence is 23 nucleotides in length. In some embodiments, the targeting sequence is 24 nucleotides in length. In some embodiments, the RNAi oligonucleotides herein comprise a targeting sequence that is fully complementary to a target sequence comprising a target mRNA. In some embodiments, the RNAi oligonucleotides herein comprise a targeting sequence that is fully complementary to a target sequence within a target mRNA. In some embodiments, the targeting sequence is partially complementary to a target sequence comprising a target mRNA. In some embodiments, the targeting sequence is partially complementary to a target sequence within a target mRNA. In some embodiments, the targeting sequence comprises a region of contiguous nucleotides comprising the antisense strand. In some embodiments, the RNAi oligonucleotides herein comprise a targeting sequence that is complementary to a contiguous sequence of nucleotides comprising a target mRNA, wherein the contiguous sequence of nucleotides is about 12 to about 30 nucleotides in length (e.g., 12 to 30, 12 to 28, 12 to 26, 12 to 24, 12 to 20, 12 to 18, 12 to 16, 14 to 22, 16 to 20, 18 to 20 or 18 to 19 nucleotides in length). In some embodiments, the RNAi oligonucleotides comprise a targeting sequence that is complementary to a contiguous sequence of nucleotides comprising a target mRNA, wherein the contiguous sequence of nucleotides is 10, 11, 12, 13, 14, 15, 16, 17, 18, 19 or 20 nucleotides in length. In some embodiments, the RNAi oligonucleotides comprise a targeting sequence that is complementary to a contiguous sequence of nucleotides comprising a target mRNA, wherein the contiguous sequence of nucleotides is 15 nucleotides in length. In some embodiments, the RNAi oligonucleotides comprise a targeting sequence that is complementary to a contiguous sequence of nucleotides comprising a target mRNA, wherein the contiguous sequence of nucleotides is 19 nucleotides in length. In some embodiments, the RNAi oligonucleotide comprises a targeting sequence that is complementary to a contiguous sequence of nucleotides comprising a target mRNA, wherein the contiguous sequence of nucleotides is 15 nucleotides in length. In some embodiments, the RNAi oligonucleotide comprises a targeting sequence that is complementary to a contiguous sequence of nucleotides comprising a target mRNA, wherein the contiguous sequence of nucleotides is 19 nucleotides in length.
FH12501620.1
Attorney Docket: DCY-13025 In some embodiments, a targeting sequence of an RNAi oligonucleotide herein is fully complementary (e.g., having no mismatches) to a target sequence comprising a target mRNA and comprises the entire length of an antisense strand. In some embodiments, a targeting sequence of an RNAi oligonucleotide herein is fully complementary (e.g., having no mismatches) to a target sequence comprising a target mRNA and comprises a portion of the entire length of an antisense strand. In some embodiments, a targeting sequence of an RNAi oligonucleotide herein is fully complementary (e.g., having no mismatches) to a target sequence comprising a target mRNA and comprises 10 to 20 nucleotides of the antisense strand. In some embodiments, a targeting sequence of an RNAi oligonucleotide herein is fully complementary (e.g., having no mismatches) to a target sequence comprising a target mRNA and comprises 15 to 19 nucleotides of the antisense strand. In some embodiments, a targeting sequence of an RNAi oligonucleotide herein is fully complementary (e.g., having no mismatches) to a target sequence comprising a target mRNA and comprises 12 nucleotides, 13 nucleotides, 14 nucleotides, 15 nucleotides, 16 nucleotides, 17 nucleotides, 18 nucleotides, 19 nucleotides, 20 nucleotides, 21 nucleotides, or 22 nucleotides of the antisense strand. In some embodiments, a targeting sequence of an RNAi oligonucleotide herein is fully complementary (e.g., having no mismatches) to a target sequence comprising a target mRNA and comprises 19 nucleotides of the antisense strand. In some embodiments, an RNAi oligonucleotide herein comprises a targeting sequence having one or more base pair (bp) mismatches with the corresponding target sequence comprising a target mRNA. In some embodiments, the targeting sequence has a 1 bp mismatch, a 2 bp mismatch, a 3 bp mismatch, a 4 bp mismatch, or a 5 bp mismatch with the corresponding target sequence comprising a target mRNA provided that the ability of the targeting sequence to bind or anneal to the target sequence under appropriate hybridization conditions and/or the ability of the RNAi oligonucleotide to inhibit or reduce target gene expression is maintained (e.g., under physiological conditions). Alternatively, in some embodiments, the targeting sequence comprises no more than 1, no more than 2, no more than 3, no more than 4, or no more than 5 bp mismatches with the corresponding target sequence comprising a target mRNA provided that the ability of the targeting sequence to bind or anneal to the target sequence under appropriate hybridization conditions and/or the ability of the RNAi oligonucleotide to inhibit or reduce target gene expression is maintained. In some embodiments, the RNAi oligonucleotide comprises a targeting sequence having 1 mismatch with the corresponding target sequence. In some embodiments, the RNAi oligonucleotide comprises a targeting sequence having 2 mismatches with the corresponding target sequence.
FH12501620.1
Attorney Docket: DCY-13025 In some embodiments, the RNAi oligonucleotide comprises a targeting sequence having 3 mismatches with the corresponding target sequence. In some embodiments, the RNAi oligonucleotide comprises a targeting sequence having 4 mismatches with the corresponding target sequence. In some embodiments, the RNAi oligonucleotide comprises a targeting sequence having 5 mismatches with the corresponding target sequence. In some embodiments, the RNAi oligonucleotide comprises a targeting sequence having more than one mismatch (e.g., 2, 3, 4, 5 or more mismatches) with the corresponding target sequence, wherein at least 2 (e.g., all) of the mismatches are positioned consecutively (e.g., 2, 3, 4, 5 or more mismatches in a row), or wherein the mismatches are interspersed in any position throughout the targeting sequence. In some embodiments, the RNAi oligonucleotide comprises a targeting sequence having more than one mismatch (e.g., 2, 3, 4, 5 or more mismatches) with the corresponding target sequence, wherein at least 2 (e.g., all) of the mismatches are positioned consecutively (e.g., 2, 3, 4, 5 or more mismatches in a row), or wherein at least one or more non-mismatched base pair is located between the mismatches, or a combination thereof. Types of Oligonucleotides A variety of RNAi oligonucleotide types and/or structures are useful for reducing target gene expression in the methods herein. Any of the RNAi oligonucleotide types described herein or elsewhere are contemplated for use as a framework to incorporate a targeting sequence herein for the purposes of inhibiting or reducing corresponding target gene expression. In some embodiments, the RNAi oligonucleotides herein inhibit target gene expression by engaging with RNA interference (RNAi) pathways upstream or downstream of Dicer involvement. For example, RNAi oligonucleotides have been developed with each strand having sizes of about 19-25 nucleotides with at least one 3^ overhang of 1 to 5 nucleotides (see, e.g., US Patent No. 8,372,968). Longer oligonucleotides also have been developed that are processed by Dicer to generate active RNAi products (see, e.g., US Patent No. 8,883,996). Further work produced extended double-stranded oligonucleotides where at least one end of at least one strand is extended beyond a duplex targeting region, including structures where one of the strands includes a thermodynamically stabilizing tetraloop structure (see, e.g., US Patent Nos. 8,513,207 and 8,927,705, as well as Intl. Patent Application Publication No. WO 2010/033225). Such structures may include single-stranded extensions (on one or both sides of the molecule) as well as double-stranded extensions. In some embodiments, the RNAi oligonucleotides conjugates herein engage with the RNAi pathway downstream of the involvement of Dicer (e.g., Dicer cleavage). In some
FH12501620.1
Attorney Docket: DCY-13025 embodiments, the oligonucleotides described herein are Dicer substrates. In some embodiments, upon endogenous Dicer processing, double-stranded nucleic acids of 19-23 nucleotides in length capable of reducing expression of a target mRNA are produced. In some embodiments, the RNAi oligonucleotide has an overhang (e.g., of 1, 2, or 3 nucleotides in length) in the 3^ end of the sense strand. In some embodiments, the RNAi oligonucleotide (e.g., siRNA conjugate) comprises a 21-nucleotide guide strand that is antisense to a target mRNA and a complementary passenger strand, in which both strands anneal to form a 19-bp duplex and 2 nucleotide overhangs at either or both 3^ ends. Longer oligonucleotide designs also are contemplated including oligonucleotides having a guide strand of 23 nucleotides and a passenger strand of 21 nucleotides, where there is a blunt end on the right side of the molecule (3^ end of passenger strand/5^ end of guide strand) and a two nucleotide 3^-guide strand overhang on the left side of the molecule (5^ end of the passenger strand/3^ end of the guide strand). In such molecules, there is a 21 bp duplex region. See, e.g., US Patent Nos.9,012,138; 9,012,621; 9,193,753; 8,420,391; and, 8,552,171 all to Tuschl et al. Such patents also indicate a lack of activity with regard to double overhang constructs. In some embodiments, the RNAi oligonucleotides conjugates disclosed herein comprise sense and antisense strands that are both in the range of about 17 to 26 (e.g., 17 to 26, 20 to 25 or 21-23) nucleotides in length. In some embodiments, the RNAi oligonucleotides disclosed herein comprise a sense and antisense strand that are both in the range of about 19- 22 nucleotides in length. In some embodiments, the sense and antisense strands are of equal length. In some embodiments, the RNAi oligonucleotides disclosed herein comprise sense and antisense strands, such that there is a 3^-overhang on either the sense strand or the antisense strand, or both the sense and antisense strand. In some embodiments, for RNAi oligonucleotides that have sense and antisense strands that are both in the range of about 21- 23 nucleotides in length, a 3^ overhang on the sense, antisense, or both sense and antisense strands is 1 or 2 nucleotides in length. In some embodiments, an RNAi oligonucleotide has a guide strand of 22 nucleotides and a passenger strand of 20 nucleotides, where there is a blunt end on the right side of the molecule (3^ end of passenger strand/5^ end of guide strand) and a 2 nucleotide 3^-guide strand overhang on the left side of the molecule (5^ end of the passenger strand/3^ end of the guide strand). In such molecules, there is a 20 bp duplex region. Other RNAi oligonucleotide designs for use with the compositions and methods herein include: 16-mer siRNAs (see, e.g., Nucleic Acids in Chemistry and Biology, Blackburn (ed.), R
OYAL S
OCIETY OF C
HEMISTRY, 2006), shRNAs (e.g., having 19 bp or shorter stems; see, e.g., Moore et al. (2010) M
ETHODS M
OL. B
IOL.629:141-58), blunt siRNAs (e.g., of 19 bps in
FH12501620.1
Attorney Docket: DCY-13025 length; see, e.g., Kraynack & Baker (2006) RNA 12:163-76), asymmetrical siRNAs (aiRNA; see, e.g., Sun et al. (2008) NAT. BIOTECHNOL.26:1379-82), asymmetric shorter-duplex siRNA (see, e.g., Chang et al. (2009) MOL. THER.17:725-32), fork siRNAs (see, e.g., Hohjoh (2004) FEBS Lett. 557:193-98), and small internally segmented interfering RNA (siRNA; see, e.g., Bramsen et al. (2007) NUCLEIC ACIDS RES.35:5886-97). Further non-limiting examples of an oligonucleotide structure that may be used in some embodiments to reduce or inhibit the expression of a target gene are microRNA (miRNA), short hairpin RNA (shRNA) and short siRNA (see, e.g., Hamilton et al. (2002) EMBO J.21:4671-79; see also, US Patent Application Publication No.2009/0099115). Antisense Strands In some embodiments, an antisense strand of an RNAi oligonucleotide is referred to as a “guide strand.” For example, an antisense strand that engages with RNA-induced silencing complex (RISC) and binds to an Argonaute protein such as Ago2, or engages with or binds to one or more similar factors, and directs silencing of a target gene, the antisense strand is referred to as a guide strand. In some embodiments, a sense strand complementary to a guide strand is referred to as a “passenger strand.” In some embodiments, an RNAi oligonucleotide herein comprises an antisense strand of up to about 50 nucleotides in length (e.g., up to 50, up to 40, up to 35, up to 30, up to 27, up to 25, up to 21, up to 19, up to 17, up to 15, or up to 8 nucleotides in length). In some embodiments, an RNAi oligonucleotide herein comprises an antisense strand of at least about 12 nucleotides in length (e.g., at least 12, at least 15, at least 19, at least 21, at least 22, at least 25, at least 27, at least 30, at least 35 or at least 38 nucleotides in length). In some embodiments, a herein comprises an antisense strand in a range of about 8 to about 40 (e.g., 8 to 40, 8 to 36, 8 to 32, 8 to 28, 15 to 40, 15 to 36, 15 to 32, 15 to 30, 15 to 28, 17 to 22, 17 to 25, 19 to 27, 19 to 30, 20 to 40, 22 to 40, 25 to 40 or 32 to 40) nucleotides in length. In some embodiments, an RNAi oligonucleotide herein comprises an antisense strand of 15 to 30 nucleotides in length. In some embodiments, an antisense strand of any one of the RNAi oligonucleotide disclosed herein is of 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 38, 39 or 40 nucleotides in length. In some embodiments, an RNAi oligonucleotide comprises an antisense strand of 19-23 nucleotides in length. In some embodiments, an RNAi oligonucleotide comprises an antisense strand of 19 nucleotides in length. In some embodiments, an RNAi oligonucleotide comprises an antisense strand of 20 nucleotides in length. In some embodiments, an RNAi oligonucleotide comprises an antisense 76
FH12501620.1
Attorney Docket: DCY-13025 strand of 21 nucleotides in length. In some embodiments, an RNAi oligonucleotide comprises an antisense strand of 22 nucleotides in length. In some embodiments, an RNAi oligonucleotide comprises an antisense strand of 23 nucleotides in length. Sense Strands In some embodiments, an RNAi oligonucleotide disclosed herein comprises a sense strand (or passenger strand) of up to about 50 nucleotides in length (e.g., up to 50, up to 40, up to 36, up to 30, up to 27, up to 25, up to 21, up to 19, up to 17 or up to 12 nucleotides in length). In some embodiments, an RNAi oligonucleotide herein comprises a sense strand of at least about 12 nucleotides in length (e.g., at least 12, at least 15, at least 19, at least 21, at least 25, at least 27, at least 30, at least 36 or at least 38 nucleotides in length). In some embodiments, an RNAi oligonucleotide herein comprises a sense strand in a range of about 12 to about 50 (e.g., 12 to 50, 12 to 40, 12 to 36, 12 to 32, 12 to 28, 15 to 40, 15 to 36, 15 to 32, 15 to 28, 17 to 21, 17 to 25, 19 to 27, 19 to 30, 20 to 40, 22 to 40, 25 to 40 or 32 to 40) nucleotides in length. In some embodiments, an RNAi oligonucleotide herein comprises a sense strand 15 to 50 nucleotides in length. In some embodiments, an RNAi oligonucleotide herein comprises a sense strand 18 to 38 nucleotides in length. In some embodiments, an RNAi oligonucleotide herein comprises a sense strand of 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 38, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, or 50 nucleotides in length. In some embodiments, an RNAi oligonucleotide herein comprises a sense strand of 12-21 nucleotides in length. In some embodiments, an RNAi oligonucleotide herein comprises a sense strand of 12 nucleotides in length. In some embodiments, an RNAi oligonucleotide herein comprises a sense strand of 13 nucleotides in length. In some embodiments, an RNAi oligonucleotide herein comprises a sense strand of 14 nucleotides in length. In some embodiments, an RNAi oligonucleotide herein comprises a sense strand of 15 nucleotides in length. In some embodiments, an RNAi oligonucleotide herein comprises a sense strand of 16 nucleotides in length. In some embodiments, an RNAi oligonucleotide herein comprises a sense strand of 17 nucleotides in length. In some embodiments, an RNAi oligonucleotide herein comprises a sense strand of 18 nucleotides in length. In some embodiments, an RNAi oligonucleotide herein comprises a sense strand of 19 nucleotides in length. In some embodiments, an RNAi oligonucleotide herein comprises a sense strand of 20 nucleotides in length. In some embodiments, an RNAi oligonucleotide herein comprises a sense strand of 21 nucleotides in length. In some embodiments, an RNAi oligonucleotide herein comprises a sense strand of 22 nucleotides in length. In some embodiments, an RNAi
FH12501620.1
Attorney Docket: DCY-13025 oligonucleotide herein comprises a sense strand of 23 nucleotides in length. In some embodiments, an RNAi oligonucleotide herein comprises a sense strand of 24 nucleotides in length. In some embodiments, an RNAi oligonucleotide herein comprises a sense strand of 25 nucleotides in length. In some embodiments, an RNAi oligonucleotide herein comprises a sense strand of 26 nucleotides in length. In some embodiments, an RNAi oligonucleotide herein comprises a sense strand of 27 nucleotides in length. In some embodiments, an RNAi oligonucleotide herein comprises a sense strand of 28 nucleotides in length. In some embodiments, an RNAi oligonucleotide herein comprises a sense strand of 29 nucleotides in length. In some embodiments, an RNAi oligonucleotide herein comprises a sense strand of 30 nucleotides in length. In some embodiments, an RNAi oligonucleotide herein comprises a sense strand of 31 nucleotides in length. In some embodiments, an RNAi oligonucleotide herein comprises a sense strand of 32 nucleotides in length. In some embodiments, an RNAi oligonucleotide herein comprises a sense strand of 33 nucleotides in length. In some embodiments, an RNAi oligonucleotide herein comprises a sense strand of 34 nucleotides in length. In some embodiments, an RNAi oligonucleotide herein comprises a sense strand of 35 nucleotides in length. In some embodiments, an RNAi oligonucleotide herein comprises a sense strand of 36 nucleotides in length. In some embodiments, an RNAi oligonucleotide herein comprises a sense strand of 37 nucleotides in length. In some embodiments, an RNAi oligonucleotide herein comprises a sense strand of 38 nucleotides in length. In some embodiments, a sense strand comprises a stem-loop structure at its 3^ end. In some embodiments, a sense strand comprises a stem-loop structure at its 5^ end. In some embodiments, the stem-loop is formed by intrastrand base pairing. In some embodiments, a sense strand comprises a stem-loop structure at its 5^ end. In some embodiments, a stem is a duplex of 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13 or 14 nucleotides in length. In some embodiments, the stem of the stem-loop comprises a duplex of 1 nucleotide in length. In some embodiments, the stem of the stem-loop comprises a duplex of 2 nucleotides in length. In some embodiments, the stem of the stem-loop comprises a duplex of 3 nucleotides in length. In some embodiments, the stem of the stem-loop comprises a duplex of 4 nucleotides in length. In some embodiments, the stem of the stem-loop comprises a duplex of 5 nucleotides in length. In some embodiments, the stem of the stem-loop comprises a duplex of 6 nucleotides in length. In some embodiments, the stem of the stem-loop comprises a duplex of 7 nucleotides in length. In some embodiments, the stem of the stem-loop comprises a duplex of 8 nucleotides in length. In some embodiments, the stem of the stem-loop comprises a duplex of 9 nucleotides in length. In some embodiments, the stem of the stem-loop comprises a duplex of 10 nucleotides in length. In
FH12501620.1
Attorney Docket: DCY-13025 some embodiments, the stem of the stem-loop comprises a duplex of 11 nucleotides in length. In some embodiments, the stem of the stem-loop comprises a duplex of 12 nucleotides in length. In some embodiments, the stem of the stem-loop comprises a duplex of 13 nucleotides in length. In some embodiments, the stem of the stem-loop comprises a duplex of 14 nucleotides in length. In some embodiments, a stem-loop provides the RNAi oligonucleotide protection against degradation (e.g., enzymatic degradation), facilitates or improves targeting and/or delivery to a target cell, tissue, or organ, or both. For example, in some embodiments, the loop of a stem-loop provides nucleotides comprising one or more modifications that facilitate, improve, or increase targeting to a target mRNA (e.g., a target mRNA expressed in extrahepatic tissue), inhibition of target gene expression, and/or delivery to a target cell, tissue, or organ (e.g., extrahepatic tissue), or a combination thereof. In some embodiments, the stem-loop itself or modification(s) to the stem-loop do not substantially affect the inherent gene expression inhibition activity of the RNAi oligonucleotide, but facilitates, improves, or increases stability (e.g., provides protection against degradation) and/or delivery of the RNAi oligonucleotide to a target cell, tissue, or organ (e.g., extrahepatic tissue ). In some embodiments, an RNAi oligonucleotide herein comprises a sense strand comprising (e.g., at its 3^ end) a stem-loop set forth as: S1-L-S2, in which S1 is complementary to S2, and in which L forms a single-stranded loop between S1 and S2 of up to about 10 nucleotides in length (e.g., 3, 4, 5, 6, 7, 8, 9 or 10 nucleotides in length). In some embodiments, the loop (L) is 3 nucleotides in length. In some embodiments, the loop (L) is 4 nucleotides in length. In some embodiments, the tetraloop comprises the sequence 5’-GAAA-3’. In some embodiments, the tetraloop comprises the sequence 5’-UNCG-3’. In some embodiments, the tetraloop comprises the sequence 5’-UACG-3’. In some embodiments, the stem loop comprises the sequence 5’-GCAGCCGAAAGGCUGC-3’ (SEQ ID NO: 11). In some embodiments, a loop (L) of a stem-loop having the structure S1-L-S2 as described above is a triloop. In some embodiments, the triloop comprises ribonucleotides, deoxyribonucleotides, modified nucleotides, delivery ligands, and combinations thereof. In some embodiments, a loop (L) of a stem-loop having the structure S1-L-S2 as described above is a tetraloop (e.g., within a nicked tetraloop structure). In some embodiments, the tetraloop comprises ribonucleotides, deoxyribonucleotides, modified nucleotides, delivery ligands, and combinations thereof.
FH12501620.1
Attorney Docket: DCY-13025 In some embodiments, a loop (L) of a stem-loop having the structure S1-L-S2 as described above is a tetraloop as described in US Patent No. 10,131,912, incorporated herein by reference (e.g., within a nicked tetraloop structure). Duplex Length In some embodiments, a duplex formed between a sense and antisense strand is at least 8 (e.g., at least 10, at least 11, at least 12, at least 13, at least 14, at least 15, or at least 16) nucleotides in length. In some embodiments, a duplex formed between a sense and antisense strand is at least 12 (e.g., at least 15, at least 16, at least 17, at least 18, at least 19, at least 20, or at least 21) nucleotides in length. In some embodiments, a duplex formed between a sense and antisense strand is in the range of 12-30 nucleotides in length (e.g., 12 to 30, 12 to 27, 12 to 22, 15 to 25, 18 to 30, 18 to 22, 18 to 25, 18 to 27, 18 to 30, 19 to 30 or 21 to 30 nucleotides in length). In some embodiments, a duplex formed between a sense and antisense strand is 12, 13, 14, 15, 16, 17, 18, 19, 29, 21, 22, 23, 24, 25, 26, 27, 28, 29 or 30 nucleotides in length. In some embodiments, a duplex formed between a sense and antisense strand is 10-18 base pairs in length. In some embodiments, a duplex formed between a sense and antisense strand is 15- 30 base pairs in length. In some embodiments, a duplex formed between a sense and antisense strand is 17-21 base pairs in length. In some embodiments, a duplex formed between a sense and antisense strand is 12 base pairs in length. In some embodiments, a duplex formed between a sense and antisense strand is 13 base pairs in length. In some embodiments, a duplex formed between a sense and antisense strand is 14 base pairs in length. In some embodiments, a duplex formed between a sense and antisense strand is 15 base pairs in length. In some embodiments, a duplex formed between a sense and antisense strand is 16 base pairs in length. In some embodiments, a duplex formed between a sense and antisense strand is 17 base pairs in length. In some embodiments, a duplex formed between a sense and antisense strand is 18 base pairs in length. In some embodiments, a duplex formed between a sense and antisense strand is 19 base pairs in length. In some embodiments, a duplex formed between a sense and antisense strand is 20 base pairs in length. In some embodiments, a duplex formed between a sense and antisense strand is 21 base pairs in length. In some embodiments, a duplex formed between a sense and antisense strand does not span the entire length of the sense strand and/or antisense strand. In some embodiments, a duplex between a sense and antisense strand spans the entire length of either the sense or antisense strands. In some embodiments, a duplex between a sense and antisense strand spans the entire length of both the sense strand and the antisense strand.
FH12501620.1
Attorney Docket: DCY-13025 Oligonucleotide Ends In some embodiments, an RNAi oligonucleotide disclosed herein comprises sense and antisense strands, such that there is a 3’-overhang on either the sense strand or the antisense strand, or both the sense and antisense strand. In some embodiments, an RNAi oligonucleotide herein has one 5’end that is thermodynamically less stable compared to the other 5’ end. In some embodiments, an asymmetric RNAi oligonucleotide is provided that includes a blunt end at the 3’end of a sense strand and overhang at the 3’ end of the antisense strand. In some embodiments, a 3’ overhang on an antisense strand is 1-4 nucleotides in length (e.g., 1, 2, 3, or 4 nucleotides in length). In some embodiments, the 3’-overhang is about one to twenty nucleotides in length (e.g., about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, or about 20 nucleotides in length). In some embodiments, the 3’ overhang is about one to nineteen, one to eighteen, one to seventeen, one to sixteen, one to fifteen, one to fourteen, one to thirteen, one to twelve, one to eleven, one to ten, one to nine, one to eight, one to seven, one to six, one to five, one to four, one to three, or about one to two nucleotides in length. In some embodiments, the 3’-overhang is nucleotide in length. In some embodiments, the 3’-overhang is two nucleotides in length. In some embodiments, the 3’-overhang is three nucleotides in length. In some embodiments, the 3’-overhang is four nucleotides in length. In some embodiments, the 3’-overhang is five nucleotides in length. In some embodiments, the 3’- overhang is six nucleotides in length. In some embodiments, the 3’-overhang is seven nucleotides in length. In some embodiments, the 3’-overhang is eight nucleotides in length. In some embodiments, the 3’-overhang is nine nucleotides in length. In some embodiments, the 3’-overhang is ten nucleotides in length. In some embodiments, the 3’-overhang is eleven nucleotides in length. In some embodiments, the 3’-overhang is twelve nucleotides in length. In some embodiments, the 3’-overhang is thirteen nucleotides in length. In some embodiments, the 3’-overhang is fourteen nucleotides in length. In some embodiments, the 3’-overhang is fifteen nucleotides in length. In some embodiments, the 3’-overhang is sixteen nucleotides in length. In some embodiments, the 3’-overhang is seventeen nucleotides in length. In some embodiments, the 3’-overhang is eighteen nucleotides in length. In some embodiments, the 3’-overhang is nineteen nucleotides in length. In some embodiments, the 3’-overhang is twenty nucleotides in length. Typically, an oligonucleotide for RNAi has a two nucleotide overhang on the 3’ end of the antisense (guide) strand. However, other overhangs are possible. In some embodiments, an overhang is a 3’ overhang comprising a length of between one and four nucleotides,
FH12501620.1
Attorney Docket: DCY-13025 optionally one to four, one to three, one to two, two to four, two to three, or one, two, three, or four nucleotides. In some embodiments, the overhang is a 5’ overhang comprising a length of between one and four nucleotides, optionally one to four, one to three, one to two, two to four, two to three, or one, two, three, or four nucleotides. In some embodiments, an oligonucleotide herein comprises a sense strand and an antisense strand, wherein the 5’ terminus of either or both strands comprise a 5’-overhang comprising one or more nucleotides. In some embodiments, an oligonucleotide herein comprises a sense strand and an antisense strand, wherein the sense strand comprises a 5’- overhang comprising one or more nucleotides. In some embodiments, an oligonucleotide herein comprises a sense strand and an antisense strand, wherein the antisense strand comprises a 5’-overhang comprising one or more nucleotides. In some embodiments, an oligonucleotide herein comprises a sense strand and an antisense strand, wherein both the sense strand and the antisense strand comprises a 5’-overhang comprising one or more nucleotides. In some embodiments, the 5’-overhang is about one to twenty nucleotides in length (e.g., about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, or about 20 nucleotides in length). In some embodiments, the 5’ overhang is about one to nineteen, one to eighteen, one to seventeen, one to sixteen, one to fifteen, one to fourteen, one to thirteen, one to twelve, one to eleven, one to ten, one to nine, one to eight, one to seven, one to six, one to five, one to four, one to three, or about one to two nucleotides in length. In some embodiments, the 5’-overhang is nucleotide in length. In some embodiments, the 5’-overhang is two nucleotides in length. In some embodiments, the 5’-overhang is three nucleotides in length. In some embodiments, the 5’-overhang is four nucleotides in length. In some embodiments, the 5’-overhang is five nucleotides in length. In some embodiments, the 5’- overhang is six nucleotides in length. In some embodiments, the 5’-overhang is seven nucleotides in length. In some embodiments, the 5’-overhang is eight nucleotides in length. In some embodiments, the 5’-overhang is nine nucleotides in length. In some embodiments, the 5’-overhang is ten nucleotides in length. In some embodiments, the 5’-overhang is eleven nucleotides in length. In some embodiments, the 5’-overhang is twelve nucleotides in length. In some embodiments, the 5’-overhang is thirteen nucleotides in length. In some embodiments, the 5’-overhang is fourteen nucleotides in length. In some embodiments, the 5’-overhang is fifteen nucleotides in length. In some embodiments, the 5’-overhang is sixteen nucleotides in length. In some embodiments, the 5’-overhang is seventeen nucleotides in length. In some embodiments, the 5’-overhang is eighteen nucleotides in length. In some
FH12501620.1
Attorney Docket: DCY-13025 embodiments, the 5’-overhang is nineteen nucleotides in length. In some embodiments, the 5’-overhang is twenty nucleotides in length. In some embodiments, one or more (e.g., 2, 3, or 4) terminal nucleotides of the 3’ end or 5’ end of a sense and/or antisense strand are modified. For example, in some embodiments, one or two terminal nucleotides of the 3’ end of the antisense strand are modified. In some embodiments, the last nucleotide at the 3’ end of an antisense strand is modified, e.g., comprises 2’ modification, e.g., a 2’-O-methoxyethyl. In some embodiments, the last one or two terminal nucleotides at the 3’ end of an antisense strand are complementary with the target. In some embodiments, the last one or two nucleotides at the 3’ end of the antisense strand are not complementary with the target. In some embodiments, an RNAi oligonucleotide disclosed herein comprises a stem- loop structure at the 3’ end of the sense strand and comprises two terminal overhang nucleotides at the 3’ end of the antisense strand. In some embodiments, an RNAi oligonucleotide herein comprises a nicked tetraloop structure, wherein the 3’ end of the sense strand comprises a stem- tetraloop structure and comprises two terminal overhang nucleotides at the 3’ end of the antisense strand. In some embodiments, an RNAi oligonucleotide disclosed herein comprises a stem- loop structure at the 5’ end of the sense strand and comprises an overhang nucleotides at the 3’ end of the antisense strand. In some embodiments, an RNAi oligonucleotide herein comprises a nicked tetraloop structure, wherein the 5’ end of the sense strand comprises a stem-tetraloop structure and comprises two terminal overhang nucleotides at the 3’ end of the antisense strand. In some embodiments, an RNAi oligonucleotide disclosed herein comprises a stem- loop structure at the 5’ end of the sense strand and comprises a blunt end at the 5’ end of the antisense strand. In some embodiments, an RNAi oligonucleotide disclosed herein comprises an overhang of 1-8 nucleotides at the 5’ end of the sense strand and comprises an overhang of 1- 8 nucleotides at the 5’ end of the antisense strand. In some embodiments, the overhang is selected from AA, GG, AG, and GA. In some embodiments, the overhang is AA. In some embodiments, the overhang is AG. In some embodiments, the overhang is GA. In some embodiments, the two terminal overhang nucleotides are GG. Typically, one or both of the two terminal GG nucleotides of the antisense strand are not complementary with the target. In some embodiments, the 5’ end and/or the 3’end of a sense or antisense strand has an inverted cap nucleotide.
FH12501620.1
Attorney Docket: DCY-13025 In some embodiments, one or more (e.g., 2, 3, 4, 5, 6) modified internucleotide linkages are provided between terminal nucleotides of the 3’ end or 5’ end of a sense and/or antisense strand. In some embodiments, modified internucleotide linkages are provided between overhang nucleotides at the 3’ end or 5’ end of a sense and/or antisense strand. Oligonucleotide Modifications In some embodiments, an RNAi oligonucleotide disclosed herein comprises one or more modifications. Oligonucleotides (e.g., RNAi oligonucleotides) may be modified in various ways to improve or control specificity, stability, delivery, bioavailability, resistance from nuclease degradation, immunogenicity, base-pairing properties, RNA distribution and cellular uptake and other features relevant to therapeutic research use. In some embodiments, the modification is a modified sugar. In some embodiments, the modification is a 5’-terminal phosphate group. In some embodiments, the modification is a modified internucleoside linkage. In some embodiments, the modification is a modified base. In some embodiments, an oligonucleotide described herein can comprise any one of the modifications described herein or any combination thereof. For example, in some embodiments, an oligonucleotide described herein comprises at least one modified sugar, a 5’- terminal phosphate group, at least one modified internucleoside linkage, and at least one modified base. The number of modifications on an oligonucleotide (e.g., an RNAi oligonucleotide) and the position of those nucleotide modifications may influence the properties of an oligonucleotide. For example, oligonucleotides may be delivered in vivo by conjugating them to or encompassing them in a lipid nanoparticle (LNP) or similar carrier. However, when an oligonucleotide is not protected by an LNP or similar carrier, it may be advantageous for at least some of the nucleotides to be modified. Accordingly, in some embodiments, all or substantially all of the nucleotides of an oligonucleotides are modified. In some embodiments, more than half of the nucleotides are modified. In some embodiments, less than half of the nucleotides are modified. In some embodiments, the sugar moiety of all nucleotides comprising the oligonucleotide is modified at the 2’ position. In some embodiments, the sugar moiety of all nucleotides comprising the oligonucleotide is modified at the 2’ position, except for the nucleotide conjugated to a ligand (e.g., the 5’-terminal nucleotide of the sense strand). The modifications may be reversible or irreversible. In some embodiments, an oligonucleotide as disclosed herein has a number and type of modified nucleotides sufficient to cause the desired
FH12501620.1
Attorney Docket: DCY-13025 characteristics (e.g., protection from enzymatic degradation, capacity to target a desired cell after in vivo administration, and/or thermodynamic stability). Sugar Modifications In some embodiments, a nucleotide modification in a sugar comprises a 2^- modification. In some embodiments, a 2^-modification may be 2^-O-propargyl, 2^-O- propylamin, 2^-amino, 2^-ethyl, 2^-fluoro (2^-F), 2^-aminoethyl (EA), 2^-O-methyl (2^-OMe), 2^- O-methoxyethyl (2^-MOE), 2^-O-[2-(methylamino)-2-oxoethyl] (2^-O-NMA) or 2^-deoxy-2^- fluoro-^-d-arabinonucleic acid (2^-FANA). In some embodiments, the modification is 2^-F, 2^- OMe or 2^-MOE. In some embodiments, a modification in a sugar comprises a modification of the sugar ring, which may comprise modification of one or more carbons of the sugar ring. For example, a modification of a sugar of a nucleotide may comprise a 2^-oxygen of a sugar is linked to a 1^-carbon or 4^-carbon of the sugar, or a 2^-oxygen is linked to the 1^-carbon or 4^- carbon via an ethylene or methylene bridge. In some embodiments, a modified nucleotide has an acyclic sugar that lacks a 2^-carbon to 3^-carbon bond. In some embodiments, a modified nucleotide has a thiol group, e.g., in the 4^ position of the sugar. In some embodiments, an RNAi oligonucleotide described herein comprises at least about 1 modified nucleotide (e.g., at least 1, at least 5, at least 10, at least 15, at least 20, at least 25, at least 30, at least 35, at least 40, at least 45, at least 50, at least 55, at least 60, or more). In some embodiments, the sense strand of the RNAi oligonucleotide comprises at least about 1 modified nucleotide (e.g., at least 1, at least 5, at least 10, at least 15, at least 20, at least 25, at least 30, at least 35, or more). In some embodiments, the antisense strand of the RNAi oligonucleotide comprises at least about 1 modified nucleotide (e.g., at least 1, at least 5, at least 10, at least 15, at least 20, or more). In some embodiments, all the nucleotides of the sense strand of the RNAi oligonucleotide are modified. In some embodiments, all the nucleotides of the antisense strand of the RNAi oligonucleotide are modified. In some embodiments, all the nucleotides of the RNAi oligonucleotide, both the sense strand and the antisense strand) are modified. In some embodiments, the modified nucleotide comprises a 2^-modification (e.g., a 2^-F or 2^-OMe, 2^- MOE, and 2^-deoxy-2^-fluoro-^-d-arabinonucleic acid). In some embodiments, the disclosure provides RNAi oligonucleotides having different modification patterns. In some embodiments, the modified RNAi oligonucleotides comprise a sense strand sequence having a modification pattern as set forth in the Examples and Sequence
FH12501620.1
Attorney Docket: DCY-13025 Listing and an antisense strand having a modification pattern as set forth in the Examples and Sequence Listing. In some embodiments, an RNAi oligonucleotide disclosed herein comprises an antisense strand having nucleotides that are modified with 2^-F. In some embodiments, an RNAi oligonucleotide disclosed herein comprises an antisense strand comprises nucleotides that are modified with 2^-F and 2^-OMe. In some embodiments, an RNAi oligonucleotide disclosed herein comprises a sense strand having nucleotides that are modified with 2^-F. In some embodiments, an RNAi oligonucleotide disclosed herein comprises a sense strand comprising nucleotides that are modified with 2^-F and 2^-OMe. In some embodiments, an RNAi oligonucleotide disclosed herein comprises a sense strand comprising nucleotides that are modified with 2^-F and 2^-OMe, provided that a nucleotide conjugated to a ligand is not modified with 2’-F or 2’-OMe. In some embodiments, an oligonucleotide described herein comprises a sense strand with about 10-25%, 10%, 11%, 12%, 13%, 14% 15%, 16%, 17%, 18%, 19% or 20% of the nucleotides of the sense strand comprising a 2’-fluoro modification. In some embodiments, about 11% of the nucleotides of the sense strand comprise a 2-fluoro modification. In some embodiments, about 40% of the nucleotides of the sense strand comprise a 2-fluoro modification. In some embodiments, about 20% of the nucleotides of the sense strand comprise a 2-fluoro modification. In some embodiments, an oligonucleotide described herein comprises an antisense strand with about 25-45%, 25%, 26%, 27%, 28%, 29%, 30%, 31%, 32%, 33%, 34% 35%, 36%, 37%, 38%, 39%, 40%, 41%, 42%, 43%, 44%, or 45% of the nucleotides of the antisense strand comprising a 2’-fluoro modification. In some embodiments, an oligonucleotide described herein comprises an antisense strand with about 25-35%, 25%, 26%, 27%, 28%, 29%, 30%, 31%, 32%, 33%, 34% or 35% of the nucleotides of the antisense strand comprising a 2’-fluoro modification. In some embodiments, about 32% of the nucleotides of the antisense strand comprise a 2’-fluoro modification. In some embodiments, about 40% of the nucleotides of the antisense strand comprise a 2’-fluoro modification. In some embodiments, the oligonucleotide has about 15-30%, 15%, 16%, 17%, 18%, 19%, 20%, 21%, 22%, 23%, 24%, 25%, 26%, 27%, 28%, 29%, or 30% of its nucleotides comprising a 2’-fluoro modification. In some embodiments, the oligonucleotide has about 15-25%, 15%, 16%, 17%, 18%, 19%, 20%, 21%, 22%, 23%, 24%, or 25% of its nucleotides comprising a 2’-fluoro modification. In some embodiments, about 19% of the nucleotides in the oligonucleotide comprise a 2’-fluoro modification. In some embodiments, about 26% of the nucleotides in the
FH12501620.1
Attorney Docket: DCY-13025 oligonucleotide comprise a 2’-fluoro modification. In some embodiments, about 29% of the nucleotides in the oligonucleotide comprise a 2’-fluoro modification. In some embodiments, one or more of positions 8, 9, 10 or 11 of the sense strand is modified with a 2^-F group. In some embodiments, one or more nucleotides forming a base pair with a nucleotide at one or more of positions 10-13 of the antisense strand, is modified with a 2^-F group. In some embodiments, the sugar moiety at each of nucleotides not modified with a 2’-F group or conjugated to a ligand in the sense strand is modified with a 2^-OMe. In some embodiments, the sugar moiety at each of nucleotides at positions 1-7 and 12-20 in the sense strand is modified with a 2^-OMe. In some embodiments, an RNAi oligonucleotide provided herein comprises an antisense strand 22 nucleotides in length, with positions 1-22 numbered 5’ to 3’, and a sense strand having a 2’-fluoro modification at each of the nucleotides forming a base pair with nucleotides at one or more of positions 10, 11, 12, and 13 of the antisense strand. In some embodiments, an RNAi oligonucleotide provided herein comprises an antisense strand 22 nucleotides in length, with positions 1-22 numbered 5’ to 3’, and a sense strand having a 2’- fluoro modification at each of the nucleotides forming a base pair with nucleotides at positions 10, 11, 12, 13, or any combination thereof, of the antisense strand. In some embodiments, an RNAi oligonucleotide provided herein comprises an antisense strand 22 nucleotides in length, with positions 1-22 numbered 5’ to 3’, and a sense strand having a 2’-fluoro modification at each of the nucleotides forming a base pair with nucleotides at positions 10, 11, 12, and 13 of the antisense strand. In some embodiments, one or more of positions 4, 6, 9, 11, 13, 14, 16, and 18 of the sense strand is modified with a 2^-F group. In some embodiments, one or more nucleotides forming a base pair with a nucleotide at one or more of positions 4, 6, 8, 9, 11, 13, 16, or 18 of the antisense strand, is modified with a 2^-F group. In some embodiments, the sugar moiety at each of nucleotides not modified with a 2’-F group or conjugated to a ligand in the sense strand is modified with a 2^-OMe. In some embodiments, the sugar moiety at each of nucleotides at positions 2, 3, 5, 7, 8, 10, 12, 15, 17, 19, 20, and 21 in the sense strand is modified with a 2^- OMe. In some embodiments, one or more of positions 3, 5, 8, 10, 12, 13, 15, and 17 of the sense strand is modified with a 2^-F group. In some embodiments, one or more nucleotides forming a base pair with a nucleotide at one or more of positions 4, 6, 8, 9, 11, 13, 16, or 18 of the antisense strand, is modified with a 2^-F group. In some embodiments, the sugar moiety at each of nucleotides not modified with a 2’-F group or conjugated to a ligand in the sense strand
FH12501620.1
Attorney Docket: DCY-13025 is modified with a 2^-OMe. In some embodiments, the sugar moiety at each of nucleotides at positions 1, 2, 4, 6, 7, 9, 11, 14, 16, 18, 19, and 20 in the sense strand is modified with a 2^- OMe. In some embodiments, the sense strand comprises at least one 2’-F modified nucleotide wherein the remaining nucleotides not modified with a 2’-F group or conjugated to a ligand are modified with a 2’-OMe. In some embodiments, at least one internal position (e.g., a position other than the 5’ and 3’ terminal nucleotides) of the sense strand is modified with a 2^-F group. In some embodiments, there is no 2^-F group at a terminal nucleotide of the sense strand. In some embodiments, at least one internal position (e.g., a position other than the 5’ and 3’ terminal nucleotides) of the antisense strand is modified with a 2^-F group. In some embodiments, there is no 2^-F group at a terminal nucleotide of the antisense strand. In some embodiments, the antisense strand has 7 nucleotides that are modified at the 2’ position of the sugar moiety with a 2’-F. In some embodiments, the sugar moiety at positions 2, 3, 4, 5, 7, 10, and 14 of the antisense strand are modified with a 2’-F. In some embodiments, the antisense strand has 14 nucleotides that are modified at the 2’ position of the sugar moiety with a 2’-OMe. In some embodiments, the sugar moiety at positions 6, 8, 9, 11, 12, 13, 15, 16, 17, 18, 19, 20, 21, and 22 of the antisense strand are modified with a 2’-OMe. In some embodiments, antisense strand has 9 nucleotides that are modified at the 2’ position of the sugar moiety with a 2’-F. In some embodiments, the sugar moiety at positions 2, 3, 4, 5, 7, 10, 14, 16, and 19 of the antisense strand are modified with a 2’-F. In some embodiments, the antisense strand has 12 nucleotides that are modified at the 2’ position of the sugar moiety with a 2’-OMe. In some embodiments, the sugar moiety at positions 6, 8, 9, 11, 12, 13, 15, 17, 18, 20, 21, and 22 of the antisense strand are modified with a 2’-OMe. In some embodiments, an RNAi oligonucleotide provided herein comprises an antisense strand having the sugar moiety of each of the nucleotides at positions 2, 3, 4, 5, 10, and 14 of the antisense strand modified with 2^-F and the sugar moiety of each of the remaining nucleotides of the antisense strand modified with a modification selected from the group consisting of 2^-O-propargyl, 2^-O-propylamin, 2^-amino, 2^-ethyl, 2^-aminoethyl (EA), 2’-O- methyl (2^-OMe), 2’-O-methoxyethyl (2^-MOE), 2^-O-[2-(methylamino)-2-oxoethyl] (2^-O- NMA), and 2’-deoxy-2’-fluoro-^-d-arabinonucleic acid (2^-FANA). In some embodiments, an RNAi oligonucleotide provided herein comprises an antisense strand having the sugar moiety of each of the nucleotides at positions 2, 3, 4, 5, 7, 10,
FH12501620.1
Attorney Docket: DCY-13025 14, 16 and 19 of the antisense strand modified with 2^-F and the sugar moiety of each of the remaining nucleotides of the antisense strand modified with a modification selected from the group consisting of 2^-O-propargyl, 2^-O-propylamin, 2^-amino, 2^-ethyl, 2^-aminoethyl (EA), 2’-O-methyl (2^-OMe), 2’-O-methoxyethyl (2^-MOE), 2^-O-[2-(methylamino)-2-oxoethyl] (2^- O-NMA), and 2’-deoxy-2’-fluoro-^-d-arabinonucleic acid (2^-FANA). In some embodiments, an RNAi oligonucleotide provided herein comprises an antisense strand having the sugar moiety at position 1, position 2, position 3, position 4, position 5, position 6, position 7, position 8, position 9, position 10, position 11, position 12, position 13, position 14, position 15, position 16, position 17, position 18, position 19, position 20, position 21, or position 22 modified with 2^-F. In some embodiments, an RNAi oligonucleotide provided herein comprises an antisense strand having the sugar moiety at position 1, position 2, position 3, position 4, position 5, position 6, position 7, position 8, position 9, position 10, position 11, position 12, position 13, position 14, position 15, position 16, position 17, position 18, position 19, position 20, position 21, or position 22 modified with 2^-OMe. In some embodiments, an RNAi oligonucleotide provided herein comprises an antisense strand having the sugar moiety at position 1, position 2, position 3, position 4, position 5, position 6, position 7, position 8, position 9, position 10, position 11, position 12, position 13, position 14, position 15, position 16, position 17, position 18, position 19, position 20, position 21, or position 22 modified with a modification selected from the group consisting of 2^-O-propargyl, 2^-O-propylamin, 2^-amino, 2^-ethyl, 2’-aminoethyl (EA), 2^-O-methyl (2^- OMe), 2^-O-methoxyethyl (2^-MOE), 2^-O-[2-(methylamino)-2-oxoethyl] (2^-O-NMA), and 2^-deoxy-2^-fluoro-^-d-arabinonucleic acid (2^-FANA). In some embodiments, an RNAi oligonucleotide provided herein comprises a sense strand having the sugar moiety at positions 8-11 modified with 2^-F. In some embodiments, an RNAi oligonucleotide provided herein comprises a sense strand having the sugar moiety at positions 4, 6, 9, 11, 13, 14, 16, and 18 modified with 2^-F. In some embodiments, an RNAi oligonucleotide provided herein comprises a sense strand having the sugar moiety at positions 3, 5, 8, 10, 12, 13, 15, and 17 modified with 2^-F. In some embodiments, an RNAi oligonucleotide provided herein comprises a sense strand having the sugar moiety at positions 1-7 and 12-20 modified with 2’OMe. In some embodiments, an RNAi oligonucleotide provided herein comprises a sense strand having the sugar moiety at positions 2-7 and 12-20 modified with 2’OMe. In some embodiments, an RNAi oligonucleotide provided herein comprises a sense strand having the sugar moiety at positions 2-7 and 12-19 modified with 2’OMe. In some
FH12501620.1
Attorney Docket: DCY-13025 embodiments, an RNAi oligonucleotide provided herein comprises a sense strand having the sugar moiety at positions 3-7 and 12-20 modified with 2’OMe. In some embodiments, an RNAi oligonucleotide provided herein comprises a sense strand having the sugar moiety at positions 1, 2, 4, 6, 7, 9, 11, 14, 16, 18, 19, and 20 modified with 2’OMe. In some embodiments, an RNAi oligonucleotide provided herein comprises a sense strand having the sugar moiety at positions 2, 3, 5, 7, 8, 10, 12, 15, 17, 19, 20, and 21 modified with 2’OMe. In some embodiments, an RNAi oligonucleotide provided herein comprises a sense strand having the sugar moiety of each of the nucleotides at positions 1-7 and 12-20 of the sense strand modified with a modification selected from the group consisting of 2^-O- propargyl, 2^-O-propylamin, 2^-amino, 2^-ethyl, 2’-aminoethyl (EA), 2^-O-methyl (2^-OMe), 2^- O-methoxyethyl (2^-MOE), 2^-O-[2-(methylamino)-2-oxoethyl] (2^-O-NMA), and 2^-deoxy-2^- fluoro-^-d-arabinonucleic acid (2^-FANA). In some embodiments, an RNAi oligonucleotide provided herein comprises a sense strand having the sugar moiety of each of the nucleotides at positions 2-7 and 12-20 of the sense strand modified with a modification selected from the group consisting of 2^-O- propargyl, 2^-O-propylamin, 2^-amino, 2^-ethyl, 2’-aminoethyl (EA), 2^-O-methyl (2^-OMe), 2^- O-methoxyethyl (2^-MOE), 2^-O-[2-(methylamino)-2-oxoethyl] (2^-O-NMA), and 2^-deoxy-2^- fluoro-^-d-arabinonucleic acid (2^-FANA). In some embodiments, an RNAi oligonucleotide provided herein comprises a sense strand having the sugar moiety of each of the nucleotides at positions 2-7 and 12-19 of the sense strand modified with a modification selected from the group consisting of 2^-O- propargyl, 2^-O-propylamin, 2^-amino, 2^-ethyl, 2’-aminoethyl (EA), 2^-O-methyl (2^-OMe), 2^- O-methoxyethyl (2^-MOE), 2^-O-[2-(methylamino)-2-oxoethyl] (2^-O-NMA), and 2^-deoxy-2^- fluoro-^-d-arabinonucleic acid (2^-FANA). In some embodiments, an RNAi oligonucleotide provided herein comprises a sense strand having the sugar moiety of each of the nucleotides at positions 3-7 and 12-20 of the sense strand modified with a modification selected from the group consisting of 2^-O- propargyl, 2^-O-propylamin, 2^-amino, 2^-ethyl, 2’-aminoethyl (EA), 2^-O-methyl (2^-OMe), 2^- O-methoxyethyl (2^-MOE), 2^-O-[2-(methylamino)-2-oxoethyl] (2^-O-NMA), and 2^-deoxy-2^- fluoro-^-d-arabinonucleic acid (2^-FANA). In some embodiments, an RNAi oligonucleotide provided herein comprises a sense strand having the sugar moiety of each of the nucleotides at positions 2, 3, 5, 7, 8, 10, 12, 15, 17, 19, 20, and 12-17 or 12-20 of the sense strand modified with a modification selected from the group consisting of 2^-O-propargyl, 2^-O-propylamin, 2^-amino, 2^-ethyl, 2’-aminoethyl
FH12501620.1
Attorney Docket: DCY-13025 (EA), 2^-O-methyl (2^-OMe), 2^-O-methoxyethyl (2^-MOE), 2^-O-[2-(methylamino)-2- oxoethyl] (2^-O-NMA), and 2^-deoxy-2^-fluoro-^-d-arabinonucleic acid (2^-FANA). In some embodiments, an RNAi oligonucleotide provided herein comprises a sense strand having the sugar moiety at positions 1, 2, 4, 6, 7, 9, 11, 14, 16, 18, 19, and 20of the sense strand modified with a modification selected from the group consisting of 2^-O-propargyl, 2^- O-propylamin, 2^-amino, 2^-ethyl, 2’-aminoethyl (EA), 2^-O-methyl (2^-OMe), 2^-O- methoxyethyl (2^-MOE), 2^-O-[2-(methylamino)-2-oxoethyl] (2^-O-NMA), and 2^-deoxy-2^- fluoro-^-d-arabinonucleic acid (2^-FANA). In some embodiments, an RNAi oligonucleotide provided herein comprises a sense strand having the sugar moiety at position 1, position 2, position 3, position 4, position 5, position 6, position 7, position 8, position 9, position 10, position 11, position 12, position 13, position 14, position 15, position 16, position 17, position 18, position 19, position 20, position 21, position 22, position 23, position 24, position 25, position 26, position 27, position 28, position 29, position 30, position 31, position 32, position 33, position 34, position 35, or position 36 modified with 2^-F. In some embodiments, an RNAi oligonucleotide provided herein comprises a sense strand having the sugar moiety at position 1, position 2, position 3, position 4, position 5, position 6, position 7, position 8, position 9, position 10, position 11, position 12, position 13, position 14, position 15, position 16, position 17, position 18, position 19, position 20, position 21, position 22, position 23, position 24, position 25, position 26, position 27, position 28, position 29, position 30, position 31, position 32, position 33, position 34, position 35, position 36, position 37 or position 38 modified with 2^-F. In some embodiments, an RNAi oligonucleotide provided herein comprises a sense strand having the sugar moiety at position 1, position 2, position 3, position 4, position 5, position 6, position 7, position 8, position 9, position 10, position 11, position 12, position 13, position 14, position 15, position 16, position 17, position 18, position 19, position 20, position 21, position 22, position 23, position 24, position 25, position 26, position 27, position 28, position 29, position 30, position 31, position 32, position 33, position 34, position 35, or position 36 modified with 2^-OMe. In some embodiments, an RNAi oligonucleotide provided herein comprises a sense strand having the sugar moiety at position 1, position 2, position 3, position 4, position 5, position 6, position 7, position 8, position 9, position 10, position 11, position 12, position 13, position 14, position 15, position 16, position 17, position 18, position 19, position 20, position 21, position 22, position 23, position 24, position 25, position 26, position 27, position 28,
FH12501620.1
Attorney Docket: DCY-13025 position 29, position 30, position 31, position 32, position 33, position 34, position 35, position 36, position 37 or position 38 modified with 2^-OMe. In some embodiments, an RNAi oligonucleotide provided herein comprises a sense strand having the sugar moiety at position 1, position 2, position 3, position 4, position 5, position 6, position 7, position 8, position 9, position 10, position 11, position 12, position 13, position 14, position 15, position 16, position 17, position 18, position 19, position 20, position 21, position 22, position 23, position 24, position 25, position 26, position 27, position 28, position 29, position 30, position 31, position 32, position 33, position 34, position 35, or position 36 modified with a modification selected from the group consisting of 2^-O-propargyl, 2^-O-propylamin, 2^-amino, 2^-ethyl, 2’-aminoethyl (EA), 2^-O-methyl (2^-OMe), 2^-O- methoxyethyl (2^-MOE), 2^-O-[2-(methylamino)-2-oxoethyl] (2^-O-NMA), and 2^-deoxy-2^- fluoro-^-d-arabinonucleic acid (2^-FANA). In some embodiments, an RNAi oligonucleotide provided herein comprises a sense strand having the sugar moiety at position 1, position 2, position 3, position 4, position 5, position 6, position 7, position 8, position 9, position 10, position 11, position 12, position 13, position 14, position 15, position 16, position 17, position 18, position 19, position 20, position 21, position 22, position 23, position 24, position 25, position 26, position 27, position 28, position 29, position 30, position 31, position 32, position 33, position 34, position 35, position 36, position 37 or position 38 modified with a modification selected from the group consisting of 2^-O-propargyl, 2^-O-propylamin, 2^-amino, 2^-ethyl, 2’-aminoethyl (EA), 2^-O-methyl (2^- OMe), 2^-O-methoxyethyl (2^-MOE), 2^-O-[2-(methylamino)-2-oxoethyl] (2^-O-NMA), and 2^-deoxy-2^-fluoro-^-d-arabinonucleic acid (2^-FANA). 5’-Terminal Phosphate In some embodiments, an RNAi oligonucleotide described herein comprises a 5’- terminal phosphate. In some embodiments, the 5^-terminal phosphate groups of the RNAi oligonucleotide enhance the interaction with Ago2. However, oligonucleotides comprising a 5^-phosphate group may be susceptible to degradation via phosphatases or other enzymes, which can limit their bioavailability in vivo. In some embodiments, an RNAi oligonucleotide herein comprises analogs of 5^ phosphates that are resistant to such degradation. In some embodiments, the phosphate analog is oxymethyl phosphonate, vinyl phosphonate or malonyl phosphonate, or a combination thereof. In some embodiments, the 5^ end of an RNAi oligonucleotide strand is attached to chemical moiety that mimics the electrostatic and steric properties of a natural 5^-phosphate group (“phosphate mimic”).
FH12501620.1
Attorney Docket: DCY-13025 In some embodiments, an RNAi oligonucleotide herein has a phosphate analog at a 4^- carbon position of the sugar (referred to as a “4^-phosphate analog”). See, e.g., Intl. Patent Application Publication No. WO 2018/045317. In some embodiments, an RNAi oligonucleotide herein comprises a 4^-phosphate analog at a 5^-terminal nucleotide. In some embodiments, a phosphate analog is an oxymethyl phosphonate, in which the oxygen atom of the oxymethyl group is bound to the sugar moiety (e.g., at its 4^-carbon) or analog thereof. In other embodiments, a 4^-phosphate analog is a thiomethyl phosphonate or an aminomethyl phosphonate, in which the sulfur atom of the thiomethyl group or the nitrogen atom of the amino methyl group is bound to the 4^-carbon of the sugar moiety or analog thereof. In some embodiments, a 4^-phosphate analog is an oxymethyl phosphonate. In some embodiments, an oxymethyl phosphonate is represented by the formula –O–CH2–PO(OH)2,–O–CH2–PO(OR)2, or -O-CH2-POOH(R), in which R is independently selected from H, CH3, an alkyl group, CH
2CH
2CN, CH
2OCOC(CH
3)
3, CH
2OCH
2CH
2Si (CH
3)
3 or a protecting group. In some embodiments, the alkyl group is CH
2CH
3. More typically, R is independently selected from H, CH
3 or CH
2CH
3. In some embodiment, R is CH3. In some embodiments, the 4’-phosphate analog is 5’-methoxyphosphonate-4’-oxy. In some embodiments, the 4’-phosphate analog is 4’-oxymethyl phosphonate. In some embodiments, an RNAi oligonucleotide provided herein comprises an antisense strand comprising a phosphorylated nucleotide at the 5’ terminus. In some embodiments, an RNAi oligonucleotide provided herein comprises an antisense strand comprising a phosphorylated nucleotide at the 5’ terminus, wherein the phosphorylated nucleotide is selected from uridine and adenosine. In some embodiments, an RNAi oligonucleotide provided herein comprises an antisense strand comprising a phosphorylated nucleotide at the 5’ terminus, wherein the phosphorylated nucleotide is uridine. In some embodiments, an RNAi oligonucleotide provided herein comprises an antisense strand comprising a phosphorylated nucleotide at the 5’ terminus, wherein the phosphorylated nucleotide is adenosine. In some embodiments, an RNAi oligonucleotide provided herein comprises an antisense strand comprising a 4^-phosphate analog at the 5^-terminal nucleotide. In some embodiments, an RNAi oligonucleotide provided herein comprises an antisense strand comprising a 4^-phosphate analog at the 5^-terminal nucleotide, wherein the phosphorylated nucleotide is selected from uridine and adenosine. In some embodiments, an RNAi oligonucleotide provided herein comprises an antisense strand comprising a 4^-phosphate analog at the 5^-terminal nucleotide, wherein the phosphorylated nucleotide is uridine. In some
FH12501620.1
Attorney Docket: DCY-13025 embodiments, an RNAi oligonucleotide provided herein comprises an antisense strand comprising a 4^-phosphate analog at the 5^-terminal nucleotide, wherein the phosphorylated nucleotide is adenosine. In some embodiments, an RNAi oligonucleotide provided herein comprises an antisense strand comprising a 4^-phosphate analog at the 5^-terminal nucleotide, wherein 5’-terminal nucleotide comprises the following structure (Chem 1):

4’-O-monomethylphosphonate-2’-O-methyluridine phosphorothioate [MePhosphonate-4O-mUs, alternatively referred to as “MeMOP”] Chem 1 Modified Internucleotide Linkage In some embodiments, an RNAi oligonucleotide herein comprises a modified internucleoside linkage. In some embodiments, phosphate modifications or substitutions result in an oligonucleotide that comprises at least about 1 (e.g., at least 1, at least 2, at least 3 or at least 5) modified internucleotide linkage. In some embodiments, any one of the oligonucleotides disclosed herein comprises about 1 to about 10 (e.g., 1 to 10, 2 to 8, 4 to 6, 3 to 10, 5 to 10, 1 to 5, 1 to 3 or 1 to 2) modified internucleotide linkages. In some embodiments, any one of the oligonucleotides disclosed herein comprises 1, 2, 3, 4, 5, 6, 7, 8, 9 or 10 modified internucleotide linkages. A modified internucleotide linkage may be a phosphorodithioate linkage, a phosphorothioate linkage, a phosphotriester linkage, a thionoalkylphosphonate linkage, a thionalkylphosphotriester linkage, a phosphoramidite linkage, a phosphonate linkage or a boranophosphate linkage. In some embodiments, at least one modified internucleotide linkage of any one of the oligonucleotides as disclosed herein is a phosphorothioate linkage.
FH12501620.1
Attorney Docket: DCY-13025 In some embodiments, an RNAi oligonucleotide provided herein has a phosphorothioate linkage between one or more of positions 1 and 2 of the sense strand, positions 1 and 2 of the antisense strand, positions 2 and 3 of the antisense strand, positions 3 and 4 of the antisense strand, positions 20 and 21 of the antisense strand, and positions 21 and 22 of the antisense strand. In some embodiments, the oligonucleotide described herein has a phosphorothioate linkage between each of positions 1 and 2 of the sense strand, positions 1 and 2 of the antisense strand, positions 2 and 3 of the antisense strand, positions 20 and 21 of the antisense strand, and positions 21 and 22 of the antisense strand. In some embodiments, the oligonucleotide described herein has a phosphorothioate linkage between each of positions 1 and 2 of the sense strand, positions 1 and 2 of the antisense strand, positions 2 and 3 of the antisense strand, positions 3 and 4 of the antisense strand, positions 20 and 21 of the antisense strand, and positions 21 and 22 of the antisense strand. In some embodiments, the oligonucleotide described herein has a phosphorothioate linkage between each of positions 1 and 2 of the sense strand, positions 18 and 19 of the sense strand, positions 19 and 20 of the sense strand, positions 1 and 2 of the antisense strand, positions 2 and 3 of the antisense strand, positions 3 and 4 of the antisense strand, positions 20 and 21 of the antisense strand, and positions 21 and 22 of the antisense strand. In some embodiments, the oligonucleotide described herein has a phosphorothioate linkage between each of positions 1 and 2 of the sense strand, the third to last position and penultimate position of the sense strand, and the penultimate position and ultimate position of the sense strand. In some embodiments, the oligonucleotide comprises phosphodiester linkages between nucleotides that do not comprise a phosphorothioate linkage. In some embodiments, an oligonucleotide conjugate described herein comprises a peptide nucleic acid (PNA). PNAs are oligonucleotide mimics in which the sugar-phosphate backbone has been replaced by a pseudopeptide skeleton, composed of N-(2- aminoethyl)glycine units. Nucleobases are linked to this skeleton through a two-atom carboxymethyl spacer. In some embodiments, an oligonucleotide conjugate described herein comprises a morpholino oligomer (PMO) comprising an internucleotide linkage backbone of methylene morpholine rings linked through phosphorodiamidate groups. Base Modifications In some embodiments, an RNAi oligonucleotide herein comprises one or more modified nucleobases. In some embodiments, modified nucleobases (also referred to herein as
FH12501620.1
Attorney Docket: DCY-13025 base analogs) are linked at the 1^ position of a nucleotide sugar moiety. In some embodiments, a modified nucleobase is a nitrogenous base. In some embodiments, a modified nucleobase does not contain nitrogen atom. See, e.g., US Patent Application Publication No. 2008/0274462. In some embodiments, a modified nucleotide comprises a universal base. In some embodiments, a modified nucleotide does not contain a nucleobase (abasic). In some embodiments, a universal base is a heterocyclic moiety located at the 1^ position of a nucleotide sugar moiety in a modified nucleotide, or the equivalent position in a nucleotide sugar moiety substitution, that, when present in a duplex, can be positioned opposite more than one type of base without substantially altering structure of the duplex. In some embodiments, compared to a reference single-stranded nucleic acid (e.g., oligonucleotide) that is fully complementary to a target nucleic acid, a single-stranded nucleic acid containing a universal base forms a duplex with the target nucleic acid that has a lower Tm than a duplex formed with the complementary nucleic acid. In some embodiments, when compared to a reference single-stranded nucleic acid in which the universal base has been replaced with a base to generate a single mismatch, the single-stranded nucleic acid containing the universal base forms a duplex with the target nucleic acid that has a higher T
m than a duplex formed with the nucleic acid comprising the mismatched base. Non-limiting examples of universal-binding nucleotides include, but are not limited to, inosine, 1-^-D-ribofuranosyl-5-nitroindole and/or 1-^-D-ribofuranosyl-3-nitropyrrole (see, US Patent Application Publication No.2007/0254362; Van Aerschot et al. (1995) NUCLEIC ACIDS R
ES.23:4363-70; Loakes et al. (1995) N
UCLEIC A
CIDS R
ES.23:2361-66; and Loakes & Brown (1994) N
UCLEIC A
CIDS R
ES.22:4039-43). Tm-Increasing Nucleotides In some embodiments, the oligonucleotide described herein comprises at least one Tm- increasing nucleotide in the sense strand. In some embodiments, the oligonucleotide has one Tm-increasing nucleotide in the sense strand. In some embodiments, the oligonucleotide has up to two Tm-increasing nucleotides in the sense strand. In some embodiments, the oligonucleotide has up to three Tm-increasing nucleotides in the sense strand. In some embodiments, the oligonucleotide has up to four Tm-increasing nucleotides in the sense strand. In some embodiments, the oligonucleotide has up to five Tm-increasing nucleotides in the sense strand. In some embodiments, the oligonucleotide has up to six Tm-increasing nucleotides in the sense strand. In some embodiments, the oligonucleotide has up to seven Tm- increasing nucleotides in the sense strand. In some embodiments, the oligonucleotide has up to
FH12501620.1
Attorney Docket: DCY-13025 eight Tm-increasing nucleotides in the sense strand. In some embodiments, the oligonucleotide has up to nine Tm-increasing nucleotides in the sense strand. In some embodiments, the oligonucleotide has up to ten Tm-increasing nucleotides in the sense strand. In some embodiments, the oligonucleotide has 1 to 2 Tm-increasing nucleotides in the sense strand. In some embodiments, the oligonucleotide has 1 to 3 Tm-increasing nucleotides in the sense strand. In some embodiments, the oligonucleotide has 1 to 4 Tm-increasing nucleotides in the sense strand. In some embodiments, the oligonucleotide has 1 to 5 Tm- increasing nucleotides in the sense strand. In some embodiments, an oligonucleotide comprising a stem-loop comprises a Tm- increasing nucleotide in the stem. In some embodiments, an oligonucleotide comprising a stem-loop comprises Tm-increasing nucleotides in at least one base pair of the stem. In some embodiments, an oligonucleotide comprising a stem-loop comprises Tm-increasing nucleotides in one base pair of the stem. In some embodiments, an oligonucleotide comprising a stem-loop comprises Tm-increasing nucleotides in two base pairs of the stem. In some embodiments, an oligonucleotide comprising a stem-loop comprises Tm-increasing nucleotides in three base pairs of the stem. In some embodiments, an oligonucleotide comprising a stem-loop comprises Tm-increasing nucleotides in four base pairs of the stem. In some embodiments, an oligonucleotide comprising a stem-loop comprises Tm-increasing nucleotides in five base pairs of the stem. In some embodiments, an oligonucleotide comprising a stem-loop comprises Tm-increasing nucleotides in six base pairs of the stem. Tm-increasing nucleotides include, but are not limited to, bicyclic nucleotides, tricyclic nucleotides, a G-clamp, and analogues thereof, hexitol nucleotides, or a modified nucleotide. In some embodiments, the Tm-increasing nucleotide is a bicyclic nucleotide. In some embodiments, the Tm-increasing nucleotide is a locked nucleic acid (LNA). In some embodiments, the disclosure provides an RNAi oligonucleotide for reducing target gene expression by the RNAi pathway comprising a combination of one or more Tm- increasing nucleotides and one or more nucleotides (e.g., a modified nucleotide) having a lower binding affinity, wherein the duplex region comprising the RNAi oligonucleotide is maintained under physiological conditions and the ability of the RNAi oligonucleotide to inhibit or reduce target gene expression is maintained. Bicyclic Nucleotides In some embodiments, the oligonucleotide-ligand conjugates of the disclosure (e.g., Formula (AI), (AII), (BI), (BII), (CI), or (CII)) comprise are bicyclic nucleotides. For example,
FH12501620.1
Attorney Docket: DCY-13025 the monocyclic sugar moiety shown in Formula (AI), (AII), (BI), (BII), (CI), or (CII) may be replaced by a bicyclic sugar as discussed below. Bicyclic nucleotides typically have a sugar moiety with a 4 to 7 membered ring (including but not limited to a furanosyl) comprising a bridge connecting two atoms of the 4 to 7 membered ring to form a second ring, resulting in a bicyclic structure. Such bicyclic nucleotides have various names including BNA's and LNA's for bicyclic nucleic acids and locked nucleic acids, respectively. The synthesis of bicyclic nucleotides and their incorporation into nucleic acid compounds has also been reported in the literature, including, for example, Singh et al., Chem. Commun., 1998, 4, 455-56; Koshkin et al., T
ETRAHEDRON, 1998, 54, 3607- 30; Wahlestedt et al., P
ROC. N
ATL. A
CAD. S
CI. U.S.A., 2000, 97, 5633-38; Kumar et al., B
IOORG. M
ED. C
HEM. L
ETT., 1998, 8, 2219-22; Singh et al., J. O
RG. C
HEM., 1998, 63, 10035- 039; U.S. Patent Nos. 7,427,672, 7,053,207, 6,794,499, 6,770,748, 6,268,490 and 6,794,499; and published U.S. applications 20040219565, 20040014959, 20030207841, 20040192918, 20030224377, 20040143114 and 20030082807; each of which is incorporated by reference herein, in its entirety. In some embodiments, the T
m-increasing nucleotide is a bicyclic nucleotide that comprises a bicyclic sugar moiety. In certain embodiments, the bicyclic sugar moiety comprises a first ring of 4 to 7 members and a bridge forming a North-type sugar confirmation that connects any two atoms of the first ring of the sugar moiety to form a second ring. In some embodiments, the bridge connects the 2^-carbon and the 4^-carbon of the first ring to form a second ring. Typically, the bridge contains 2 to 8 atoms. In some embodiments, the bridge contains 3 atoms. In some embodiments, the bridge contains 4 atoms. In some embodiments, the bridge contains 5 atoms. In some embodiments, the bridge contains 6 atoms. In some embodiments, the bridge contains 7 atoms. In some embodiments, the bridge contains 8 atoms. In some embodiments, the bridge contains more than 8 atoms. In some embodiments, the bicyclic sugar moiety is a substituted furanosyl comprising a bridge that connects the 2^-carbon and the 4^-carbon of the furanosyl to form the second ring. In some embodiments, the bicyclic nucleotide has the structure of Formula I:
FH12501620.1
Attorney Docket: DCY-13025
wherein B is a nucleobase; wherein G is H, OH, NH
2, C
1-C
6 alkyl, C
2-C
6 alkenyl, C
2-C
6 alkynyl, substituted C
1-C
6 alkyl, substituted C
2-C
6 alkenyl, substituted C
2-C
6 alkynyl, acyl, substituted acyl, substituted amide, thiol, or substituted thio; wherein X is O, S, or NR
1, wherein R
1 is H, C
1-C
6 alkyl, C
1-C
6 alkoxy, benzene or pyrene; and wherein Wa and Wb are each independently, H, OH, a hydroxyl protecting group, a phosphorous moiety, or an internucleotide linking group attaching the nucleotide represented by Formula I to another nucleotide or to an oligonucleotide and wherein at least one of War or Wb is an internucleotide linking group attaching the nucleotide represented by Formula I to an oligonucleotide. In some embodiments of Formula I, G is H and X is NR1, wherein R1 is benzene or pyrene. In some embodiments, of Formula I, G is H and X is S. In some embodiments of Formula I, G is H and X is O:
Formula Ia In some embodiments of Formula I, G is H and X is NR
1, wherein R
1 is H, CH
3, or OCH
3: FH12501620.1
Attorney Docket: DCY-13025
Formula Ib In some embodiments of Formula I, G is OH or NH2 and X is O. In some embodiments of Formula I, G is OH and X is O:
In some embodiments of Formula I, G is NH
2 and X is O:
In some embodiments, of Formula I, G is CH
3 or CH
2OCH
3 and X is O. In some embodiments, of Formula I, G is CH
3 and X is O
:
In some embodiments, of Formula I, G is CH
2OCH
3 and X is O:
FH12501620.1
Attorney Docket: DCY-13025
In some embodiments, the bicyclic nucleotide has the structure of Formula II:
Formula II wherein B is a nucleobase; wherein Q
1 is CH
2 or O; wherein X is CH
2, O, S, or NR
1, wherein R
1 is H, C
1-C
6 alkyl, C
1-C
6 alkoxy, benzene or pyrene; wherein if Q1 is O, X is CH2; wherein if Q1 is CH2, X is CH2, O, S, or NR1, wherein R1 is H, C1-C6 alkyl, C1-C6 alkoxy, benzene or pyrene; wherein Wa and Wb are each independently, H, OH, a hydroxyl protecting group, a phosphorous moiety, or an internucleotide linking group attaching the nucleotide represented by Formula II to another nucleotide or to an oligonucleotide and wherein at least one of Wa or Wb is an internucleotide linking group attaching the nucleotide represented by Formula II to an oligonucleotide. In some embodiments of Formula II, Q
1 is O and X is CH
2:
Formula IIa In some embodiments of Formula II, Q
1 is CH
2 and X is O:
FH12501620.1
Attorney Docket: DCY-13025
Formula IIb In some embodiments of Formula II, Q1 is CH2 and X is NR1, wherein R1 is H, CH3 or OCH
3:
In some embodiments of Formula II, Q
1 is CH
2 and X is NH:
Formula IId In some embodiments, the bicyclic nucleotide has the structure of Formula III:
Formula III wherein B is a nucleobase; wherein Q2 is O or NR1, wherein R1 is H, C1-C6 alkyl, C1-C6 alkoxy, benzene or pyrene;
FH12501620.1
Attorney Docket: DCY-13025 wherein X is CH
2, O, S, or NR
1, wherein R
1 is H, C
1-C
6 alkyl, C
1-C
6 alkoxy, benzene or pyrene; wherein if Q
2 is O, X is NR
1; wherein if Q
2 is NR
1, X is O or S; wherein Wa and Wb are each independently, H, OH, a hydroxyl protecting group, a phosphorous moiety, or an internucleotide linking group attaching the nucleotide represented by Formula III to another nucleotide or to an oligonucleotide and wherein at least one of Wa or Wb is an internucleotide linking group attaching the nucleotide represented by Formula III to an oligonucleotide. In some embodiments of Formula III, Q2 is O and X is NR1. In some embodiments of Formula III, Q2 is O and X is NR1, wherein R1 is C1-C6 alkyl. In some embodiments of Formula III, Q2 is O and X is NR1 and R1 is H or CH3. In some embodiments of Formula III, Q
2 is O and X is NR
1 and R
1 is CH
3:
Formula IIIa In some embodiments of Formula III, Q
2 is NR
1 and X is O. In some embodiments of Formula III, Q
2 is NR
1, wherein R
1 is C
1-C
6 alkyl and X is O. In some embodiments of Formula III, Q
2 is NCH
3 and X is O:
Formula IIIb In some embodiments, the bicyclic nucleotide has the structure of Formula IV:
FH12501620.1
Attorney Docket: DCY-13025
Formula IV wherein B is a nucleobase; wherein P
1 and P
3 are CH
2, P
2 is CH
2 or O and P
4 is O; and wherein W
a and W
b are each independently, H, OH, a hydroxyl protecting group, a phosphorous moiety, or an internucleotide linking group attaching the nucleotide represented by Formula IV to another nucleotide or to an oligonucleotide and wherein at least one of W
a or Wb is an internucleotide linking group attaching the nucleotide represented by Formula IV to an oligonucleotide. In some embodiments of Formula IV, P1, P2, and P3 are CH2, and P4 is O:
In some embodiments of Formula IV, P1 and P3 are CH2, P2 is O and P4 is O:
Formula IVb In some embodiments, the bicyclic nucleotide has the structure of Formula Va or Vb:
FH12501620.1
Attorney Docket: DCY-13025
wherein B is a nucleobase; wherein r1, r2, r3, and r4 are each independently H, halogen, C1-C12 alkyl, substituted C1-C12 alkyl, C2-C12 alkenyl, substituted C2-C12 alkenyl, C2-C12 alkynyl; substituted C2-C12 alkynyl; C1-C12 alkoxy; substituted C1-C12 alkoxy, OT1, ST1, SOT1, SO2T1, NT1T2, N3, CN, C(^O)OT
1, C(^O)NT
1T
2, C(^O)T
1, O^C(^O)NT
1T2, N(H)C(^NH)NT
1T
2, N(H)C(^O)NT
1T
2 or N(H)C(^S)NT
1T
2, wherein each of T1 and T2 is independently H, C
1- C
6 alkyl, or substituted C
1-C
16 alkyl; or r1 and r2 or r3 and r4 together are ^C(r5)(r6), wherein r5 and r6 are each independently H, halogen, C
1-C
12 alkyl, or substituted C
1-C
12 alkyl; and wherein Wa and Wb are each independently, H, OH, a hydroxyl protecting group, a phosphorous moiety, or an internucleotide linking group attaching the nucleotide represented by Formula V to another nucleotide or to an oligonucleotide and wherein at least one of Wa or Wb is an internucleotide linking group attaching the nucleotide represented by Formula V to an oligonucleotide. In some embodiments, the bicyclic sugar moiety is a substituted furanosyl comprising a bridge that connects the 2^-carbon and the 4^-carbon of the furanosyl to form the second ring, wherein the bridge that connects the 2^-carbon and the 4^-carbon of the furanosyl includes, but is not limited to: a) 4^-CH
2-O-N(R)-2^ and 4^-CH
2-N(R)-O-2^, wherein R is H, C
1-C
12 alkyl, or a protecting group, including, for example, 4^-CH
2-NH-O-2^ (also known as BNA
NC), 4^-CH
2-N(CH
3)-O-2^ (also known as BNA
NC[NMe]), (as described in U.S. Patent No. 7,427,672, which is hereby incorporated by reference in its entirety); b) 4^-CH2-2^; 4^-(CH2)2-2^; 4^-(CH2)3-2^; 4^-(CH2)-O-2^ (also known as LNA); 4^-(CH2)-S-2^; 4^-(CH2)2-O-2^ (also known as ENA); 4^-CH(CH3)-O-2^
FH12501620.1
Attorney Docket: DCY-13025 (also known as cEt); and 4^-CH(CH
2OCH
3)-O-2^ (also known as cMOE), and analogs thereof (as described in U.S. Patent No. 7,399,845, which is hereby incorporated by reference in its entirety); c) 4^-C(CH
3)(CH
3)-O-2^ and analogs thereof (as described in U.S. Patent No. 8,278,283, which is hereby incorporated by reference in its entirety); d) 4^-CH2-N(OCH3)-2^ and analogs thereof (as described in U.S. Patent No. 8,278,425, which is hereby incorporated by reference in its entirety); e) 4^-CH2-O-N(CH3)-2^ and analogs thereof (as described in U.S. Patent Publication No.2004/0171570, which is hereby incorporated by reference in its entirety); f) 4^-CH2-C(H)(CH3)-2^ and analogs thereof (as described in Chattopadhyaya et al., J. O
RG. C
HEM., 2009, 74, 118-34, which is hereby incorporated by reference in its entirety); and g) 4^-CH
2-C(^CH
2)-2^ and analogs thereof as described in U.S. Patent No. 8,278,426, which is hereby incorporated by reference in its entirety). In some embodiments, the bicyclic nucleotide (BN) is one or more of the following: (a) methyleneoxy BN, (b) ethyleneoxy BN, (c) aminooxy BN; (d) oxyamino BN, (e) methyl(methyleneoxy) BN (also known as constrained ethyl or cET), (f) methylene-thio BN, (g) methylene amino BN, (h) methyl carbocyclic BN, and (i) propylene carbocyclic BN, as shown below.
FH12501620.1
Attorney Docket: DCY-13025
FH12501620.1
Attorney Docket: DCY-13025
In the bicyclic nucleotides of (a) to (i) above, B is a nucleobase, R
2 is H or CH
3 and W
a and W
b are each independently, H, OH, a hydroxyl protecting group, a phosphorous moiety, or an internucleotide linking group attaching the bicyclic nucleotide to another nucleotide or to an oligonucleotide and wherein at least one of Wa or Wb is an internucleotide linking group attaching the bicyclic nucleotide to an oligonucleotide. In one embodiment of the oxyamino BN (d), R2 is CH3, as follows (also known as BNA
NC[NMe]):
. In some embodiments, bicyclic sugar moieties and bicyclic nucleotides incorporating such bicyclic sugar moieties are further defined by isomeric configuration. In some embodiments, the bicyclic sugar moiety or nucleotide is in the ^-L configuration. In some embodiments, the bicyclic sugar moiety or nucleotide is in the ^-D configuration. For example, in some embodiments, the bicyclic sugar moiety or nucleotide comprises a 2^O,4^-C-methylene bridge (2^-O-CH
2-4^) in the ^-L configuration (^-L LNA). In some embodiments, the bicyclic sugar moiety or nucleotide is in the R configuration. In some embodiments, the bicyclic sugar
FH12501620.1
Attorney Docket: DCY-13025 moiety or nucleotide is in the S configuration. For example, in some embodiments, the bicyclic sugar moiety or nucleotide comprises a 4^-CH(CH
3)-O-2^ bridge (i.e., cEt) in the S- configuration. Tricyclic Nucleotides In some embodiments, the Tm-increasing nucleotide is a tricyclic nucleotide. The synthesis of tricyclic nucleotides and their incorporation into nucleic acid compounds has also been reported in the literature, including, for example, Steffens et al., J. AM. CHEM. SOC. 1997;119:11548-549; Steffens et al., J. O
RG. C
HEM. 1999;121(14):3249-55; Renneberg et al., J. A
M. C
HEM. S
OC. 2002;124:5993-6002; Ittig et al., N
UCLEIC A
CIDS R
ES.2004;32(1):346-53; Scheidegger et al., C
HEMISTRY 2006;12:8014-23; Ivanova et al., O
LIGONUCLEOTIDES 2007;17:54-65; each of which is each hereby incorporated by reference in its entirety. In some embodiments, the tricyclic nucleotide is a tricyclo nucleotide (also called tricyclo DNA) in which the 3^-carbon and 5^-carbon centers are connected by an ethylene that is fused to a cyclopropane ring, as discussed for example in Leumann CJ, BIOORG. MED. CHEM. 2002;10:841-54 and published U.S. Applications 2015/0259681 and 2018/0162897, which are each hereby incorporated by reference. In some embodiments, the tricyclic nucleotide comprises a substituted furanosyl ring comprising a bridge that connects the 2^-carbon and the 4^-carbon of the furanosyl to form a second ring, and a third fused ring resulting from a group connecting the 5^-carbon to the methylene group of the bridge that connects the 2^-carbon and the 4^-carbon of the furanosyl, as discussed, for example, in published U.S. Application 2015/0112055, which is hereby incorporated by reference. Other Tm-increasing nucleotides In addition to bicyclic and tricyclic nucleotides, other T
m-increasing nucleotides can be used in the RNAi oligonucleotides described herein. For example, in some embodiments, the T
m-increasing nucleotide is a G-clamp, guanidine G-clamp or analogue thereof (Wilds et al., CHEM, 2002;114:123 and Wilds et al., CHIM ACTA 2003;114:123), a hexitol nucleotide (Herdewijn, CHEM. BIODIVERSITY 2010;7:1-59), or a modified nucleotide. The modified nucleotide can have a modified nucleobase, as described herein, including for example, 5- bromo-uracil, 5-iodo-uracil, 5-propynyl-modified pyrimidines, or 2-amino adenine (also called 2,6-diaminopurine) (Deleavey et al., CHEM. & BIOL.2012;19:937-54) or 2-thio uridine, 5 Me- thio uridine, and pseudo uridine. The modified nucleotide can also have a modified sugar moiety, as described for example, in U.S. Patent No. 8,975,389, which is hereby incorporated
FH12501620.1
Attorney Docket: DCY-13025 by reference, or as described herein, except that the T
m-increasing nucleotide is not modified at the 2^-carbon of the sugar moiety with a 2^-F or a 2^-OMe. In some embodiments, the T
m-increasing nucleotide is a bicyclic nucleotide. In some embodiments, the T
m-increasing nucleotide is a tricyclic nucleotide. In some embodiments, the Tm-increasing nucleotide a G-clamp, guanidine G-clamp or analogue thereof. In some embodiments, the Tm-increasing nucleotide is a hexitol nucleotide. In some embodiments, the Tm-increasing nucleotide is a bicyclic or tricyclic nucleotide. In some embodiments, the Tm- increasing nucleotide is a bicyclic nucleotide, a tricyclic nucleotide, or a G-clamp, guanidine G-clamp or analogue thereof. In some embodiments, the Tm-increasing nucleotide is a bicyclic nucleotide, a tricyclic nucleotide, a G-clamp, guanidine G-clamp or analogue thereof, or a hexitol nucleotide. In some embodiments, the Tm-increasing nucleotide increases the Tm of the nucleic acid inhibitor molecule by at least 2 °C per incorporation. In some embodiments, the T
m-increasing nucleotide increases the T
m of nucleic acid inhibitor molecule by at least 3 °C per incorporation. In some embodiments, the T
m-increasing nucleotide increases the T
m of nucleic acid inhibitor molecule by at least 4 °C per incorporation. In some embodiments, the T
m-increasing nucleotide increases the Tm of nucleic acid inhibitor molecule by at least 5 °C per incorporation. mRNA Target Sequences In some embodiments, the RNAi oligonucleotide is targeted to a target sequence comprising a target mRNA. In some embodiments, the RNAi oligonucleotide is targeted to a target sequence within a target mRNA. In some embodiments, the RNAi oligonucleotide, or a portion, fragment, or strand thereof (e.g., an antisense strand or a guide strand of a double- stranded oligonucleotide) binds or anneals to a target sequence comprising a target mRNA, thereby reducing target gene expression. In some embodiments, the RNAi oligonucleotide is targeted to a target sequence comprising target mRNA for the purpose of reducing expression of a target gene in vivo. In some embodiments, the amount or extent of reduction of target gene expression by an RNAi oligonucleotide targeted to a specific target sequence correlates with the potency of the RNAi oligonucleotide. In some embodiments, the amount or extent of reduction of target gene expression by an RNAi oligonucleotide targeted to a specific target sequence correlates with the amount or extent of therapeutic benefit in a subject or patient having a disease, disorder or condition associated with target gene expression treated with the RNAi oligonucleotide.
FH12501620.1
Attorney Docket: DCY-13025 Through examination of the nucleotide sequence of mRNAs encoding target genes, including mRNAs of multiple different species (e.g., human, cynomolgus monkey, mouse, and rat) and as a result of in vitro and in vivo testing, it has been discovered that certain nucleotide sequences and certain systemic modifications to those oligonucleotides are more amenable than others to RNAi oligonucleotide-mediated reduction and are thus useful as part of oligonucleotides that are otherwise targeted to specific gene target sequences. In some embodiments, a sense strand of an RNAi oligonucleotide, or a portion or fragment thereof, described herein, comprises a nucleotide sequence that is similar (e.g., having no more than 4 mismatches) or is identical to a target sequence comprising a target mRNA. In some embodiments, a portion or region of the sense strand of a double-stranded oligonucleotide described herein comprises a target sequence comprising a target mRNA. In some embodiments, the target mRNA is expressed in hepatic tissue, adipose tissue, cardiac tissue, adrenal tissue, or skeletal muscle tissue. In some embodiments, the target mRNA is expressed in extrahepatic tissue. In some embodiments, the target mRNA is expressed in cardiac tissue, adipose tissue, adrenal tissue, the central nervous system, or skeletal muscle tissue. In some embodiments, the target mRNA is expressed in cardiac tissue. In some embodiments, the target mRNA is expressed in adipose tissue. In some embodiments, the target mRNA is expressed in gonadal white adipose tissue. In some embodiments, the target mRNA is expressed in subcutaneous white adipose tissue. In some embodiments, the target mRNA is expressed in skeletal muscle tissue. In some embodiments, the target mRNA is expressed in adrenal tissue. In some embodiments, the target mRNA is expressed in the central nervous system. Targeting Ligands In some embodiments, it is desirable to target the oligonucleotides of the disclosure (e.g., RNAi oligonucleotide) to one or more cells or tissues of extrahepatic tissue. In some embodiments, it is desirable to target the oligonucleotides of the disclosure (e.g., RNAi oligonucleotide) to one or more cells or tissues of the liver. In some embodiments, it is desirable to target the oligonucleotides of the disclosure (e.g., RNAi oligonucleotide) to one or more cells or tissues of extrahepatic tissue. In some embodiments, it is desirable to target the oligonucleotides of the disclosure (e.g., RNAi oligonucleotide) to one or more cells or tissues of the heart. In some embodiments, it is desirable to target the oligonucleotides of the disclosure (e.g., RNAi oligonucleotide) to one or more cells or tissues of the skeletal muscle. In some embodiments, it is desirable to target the oligonucleotides of the disclosure (e.g., RNAi 111
FH12501620.1
Attorney Docket: DCY-13025 oligonucleotide) to one or more cells or tissues of the adrenal gland. In some embodiments, it is desirable to target the oligonucleotides of the disclosure (e.g., RNAi oligonucleotide) to one or more cells of adipose tissue. In some embodiments, it is desirable to target the oligonucleotides of the disclosure (e.g., RNAi oligonucleotide) to one or more cells of gonadal white adipose tissue. In some embodiments, it is desirable to target the oligonucleotides of the disclosure (e.g., RNAi oligonucleotide) to one or more cells of subcutaneous white adipose tissue. In some embodiments, it is desirable to target the oligonucleotides of the disclosure (e.g., RNAi oligonucleotide) to one or more cells or tissues of the central nervous system. Such a strategy can help to avoid undesirable effects in other organs or avoid undue loss of the oligonucleotide to cells, tissue or organs that would not benefit from the oligonucleotide. Accordingly, in some embodiments, an RNAi oligonucleotide disclosed herein is modified to facilitate targeting and/or delivery to a particular tissue, cell, or organ (e.g., to facilitate delivery of the conjugate to extrahepatic tissue). In some embodiments, an RNAi oligonucleotide comprises at least one nucleotide (e.g., 1, 2, 3, 4, 5, 6 or more nucleotides) conjugated to one or more targeting ligand(s). In some embodiments, an RNAi oligonucleotide herein does not have a GalNAc conjugated thereto. In some embodiments, 1 or more (e.g., 1, 2, 3, 4, 5 or 6) nucleotides of an RNAi oligonucleotide disclosed herein are each conjugated to a separate targeting ligand. In some embodiments, 1 nucleotide of an RNAi oligonucleotide herein is conjugated to a separate targeting ligand. In some embodiments, 2 to 4 nucleotides of an RNAi oligonucleotide herein are each conjugated to a separate targeting ligand. In some embodiments, 2 nucleotides of an RNAi oligonucleotide herein are each conjugated to a separate targeting ligand. In some embodiments, the 5’ terminal nucleotide of the sense strand and the 3’ terminal nucleotide of the sense strand are conjugated to a targeting ligand described herein. In some embodiments, the 5’ terminal nucleotide of the sense strand and the 3’ terminal nucleotide of the sense strand are conjugated to the same targeting ligand. In some embodiments, the 5’ terminal nucleotide of the sense strand and the 3’ terminal nucleotide of the sense strand are conjugated to different targeting ligands. In some embodiments, nucleotides at position 1 and position 2 of the sense strand are conjugated to the same targeting ligand. In some embodiments, nucleotides at position 1 and position 2 of the sense strand are conjugated to different targeting ligands. In some embodiments, targeting ligands are conjugated to 2 to 4 nucleotides at either ends of the sense or antisense strand (e.g., targeting ligands are conjugated to a 2 to 4 nucleotide overhang or extension on the 5^ or 3^ end of the sense or antisense strand) such that the targeting ligands resemble bristles of a toothbrush and the RNAi oligonucleotide resembles a toothbrush. For
FH12501620.1
Attorney Docket: DCY-13025 example, an RNAi oligonucleotide may comprise a stem-loop at either the 5^ or 3^ end of the sense strand and 1, 2, 3 or 4 nucleotides of the loop of the stem may be individually conjugated to a targeting ligand. In some embodiments, an RNAi oligonucleotide provided by the disclosure comprises a stem-loop at the 3^ end of the sense strand, wherein the loop of the stem- loop comprises a triloop or a tetraloop, and wherein the 3 or 4 nucleotides comprising the triloop or tetraloop, respectfully, are individually conjugated to a targeting ligand. Appropriate methods or chemistry (e.g., click chemistry) can be used to link a targeting ligand to a nucleotide. In some embodiments, a targeting ligand is conjugated to a nucleotide using a click linker. In some embodiments, an acetal-based linker is used to conjugate a targeting ligand to a nucleotide of any one of the oligonucleotides described herein. Acetal- based linkers are disclosed, for example, in Intl. Patent Application Publication No. WO 2016/100401. In some embodiments, the linker is a labile linker. However, in other embodiments, the linker is stable. As mentioned, various appropriate methods or chemistry synthetic techniques (e.g., click chemistry) can be used to link a targeting ligand to a nucleotide. In some embodiments, a targeting ligand is conjugated to a nucleotide using a click linker. In some embodiments, an acetal-based linker is used to conjugate a targeting ligand to a nucleotide of any one of the oligonucleotides described herein. Acetal-based linkers are disclosed, for example, in Intl. Patent Application Publication No. WO 2016/100401. In some embodiments, the linker is a labile linker. However, in other embodiments, the linker is a stable linker.

In some embodiments, the linker is
In some embodiments, the linker is In some embodiments, a duplex extension (e.g., of up to 3, 4, 5 or 6 bp in length) is provided between a targeting ligand and an RNAi oligonucleotide. Ligand Conjugates
FH12501620.1
Attorney Docket: DCY-13025 In some embodiments, any of the ligands described herein are conjugated to a nucleotide of the sense strand of the oligonucleotide. In some embodiments, a ligand is conjugated to a terminal position of the oligonucleotide (e.g., as shown in Formula (BI), (BII), (CI) or (CII)). In some embodiments, the ligand is conjugated to the 5’ terminal nucleotide of the sense strand (e.g., as shown in Formula (BI) or (BII)). In some embodiments, the ligand is conjugated to the 3’ terminal nucleotide of the sense strand (e.g., as shown in Formula (CI) or (CII)). In some embodiments, the ligand is conjugated to an internal nucleotide on the sense strand (e.g., as shown in Formula (AI)-(AV)). An internal position is any nucleotide position other than the two terminal positions from each end of the sense strand. In some embodiments, the ligand is conjugated to one or more internal positions of the sense strand. In some embodiments, the ligand is conjugated to position 1, position 2, position 3, position 4, position 5, position 6, position 7, position 8, position 9, position 10, position 11, position 12, position 13, position 14, position 15, position 16, position 17, position 18, position 19, position 20, position 21, position 22, position 23, position 24, position 25, position 26, position 27, position 28, position 29, position 30, position 31, position 32, position 33, position 34, position 35, position 36, position 37 or position 38 of a sense strand. In some embodiments, the ligand is conjugated to position 1 of the sense strand. In some embodiments, the ligand is conjugated to position 28 of the sense strand. In some embodiments, two ligands are conjugated to the sense strand of the oligonucleotide. In some embodiments, the two ligands conjugated to the sense strand of the oligonucleotide are the same ligand. In some embodiments, the two ligands conjugated to the sense strand of the oligonucleotide are different ligands. In some embodiments, the two ligands conjugated to the sense strand are conjugated to position 1 and to a nucleotide in the stem loop. In some embodiments, the two ligands conjugated the sense strand are conjugated to position 1 and position and position 28. In some embodiments, the two ligands conjugated to the sense strand are conjugated at position 2 and position 29. In some embodiments, the two ligands conjugated to the sense strand are conjugated at the 5’ terminal nucleotide and the 3’ terminal nucleotide. In some embodiments, an RNAi oligonucleotide described herein comprises at least one nucleotide conjugated with one or more ligands. In some embodiments, the one or more ligands are conjugated to the same nucleotide. In some embodiments, the one or more ligands are conjugated to different nucleotides. In some embodiments, 1, 2, 3, 4, 5, 6, or more ligands are conjugated to the oligonucleotide. In some embodiments, one or more ligands are conjugated to an adenine nucleotide. In some embodiments, one or more ligands are conjugated
FH12501620.1
Attorney Docket: DCY-13025 to a guanine nucleotide. In some embodiments, one or more ligands are conjugated to a cytosine nucleotide. In some embodiments, one or more ligands are conjugated to a thymine nucleotide. In some embodiments, one or more ligands are conjugated to a uracil nucleotide. In some embodiments, the ligand comprises one or more -COOH groups and a hydrocarbon chain. In some embodiments, the hydrocarbon chain is saturated. In some embodiments, the hydrocarbon chain is unsaturated. In some embodiments, the hydrocarbon chain comprises at least 6 (e.g., at least 6, at least 7, at least 8, at least 9, at least 10, at least 11, at least 12, at least 13, at least 14, at least 15, at least 16, at least 17, at least 18, at least 19, at least 20, at least 21, at least 22, at least 23, at least 24, at least 25, or at least 26) carbon atoms. In some embodiments, the hydrocarbon chain comprises from 6-26 carbon atoms (e.g., 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, or 26). In some embodiments, the ligand comprises a C8:0, C10:0, C11:0, C12:0, C14:0, C16:0, C17:0, C18:0, C18:1, C18:2, C22:5, C22:0, C24:0, C26:0, C22:6, C24:1, diacyl C16:0 or diacyl C18:1 hydrocarbon chain. In some embodiments of the RNAi oligonucleotide of the present disclosure, Y-COOH is:
FH12501620.1
Attorney Docket: DCY-13025 ,
FH12501620.1
Attorney Docket: DCY-13025
FH12501620.1
Attorney Docket: DCY-13025

. In some embodiments, the oligonucleotide of the oligonucleotide-ligand conjugate is a double-stranded molecule. In some embodiments, the oligonucleotide is an RNAi molecule. In some embodiments, the double stranded oligonucleotide comprises a stem loop. In some embodiments, the stem loop is set forth as S1-L-S2, wherein S1 is complementary to S2, and wherein L forms a loop between S1 and S2. In some embodiments, the ligand is conjugated to any of the nucleotides in the loop of the stem loop. In some embodiments, the ligand is conjugated to any of the nucleotides in the stem of the stem loop. In some embodiments, the ligand is conjugated to the first nucleotide from 5’ to 3’ in the loop. In some embodiments, the ligand is conjugated to the second nucleotide from 5’ to 3’ in the loop. In some embodiments, the ligand is conjugated to the third nucleotide from 5’ to 3’ in the loop. In some embodiments, the ligand is conjugated to the fourth nucleotide from 5’ to 3’ in the loop. In some embodiments, the ligand is conjugated to one, two, three, or four of the nucleotides in the loop. In some embodiments, the ligand is conjugated to three of the nucleotides in the stem loop. In some embodiments, the stem loop is 16 nucleotides in length. In some embodiments, the ligand is conjugated to the third nucleotide from 5’ to 3’ in the stem loop. In some embodiments, the ligand is conjugated to the eighth nucleotide from 5’ to 3’ in the stem loop. In some embodiments, the ligand is conjugated to the ninth nucleotide from 5’ to 3’ in the stem loop. In some embodiments, the ligand is conjugated to the tenth nucleotide from 5’ to 3’ in the stem loop. Exemplary Oligonucleotides In some embodiments, RNAi oligonucleotides comprise a nucleotide conjugated with a ligand comprising Y-(CO
2H)
n groups, or a charged form thereof, wherein Y is alkylene, alkenylene, or alkynylene, and q is 1, 2, 3, 4, 5, or 6. In some embodiments, the oligonucleotide of the RNAi oligonucleotide is conjugated to a ligand comprising:
FH12501620.1
Attorney Docket: DCY-13025

. In some embodiments, the 3’ end of the sense strand is a blunt end. In some embodiments, the 5’ end of the antisense strand is a blunt end. In some embodiments, the 3’ end of the antisense strand comprises an overhang. In some embodiments, the 5’ end of the antisense strand comprises an overhang. In some embodiments, the 5’ and 3’ ends of the antisense strand each comprise an overhang. In some embodiments, the RNAi oligonucleotide comprises one or more 2’ modifications. In some embodiments, the 2’ modifications are selected from 2’-fluoro and 2’- methyl. In some embodiments, a lipid-conjugated RNAi oligonucleotide for reducing expression of a target gene comprises one or more of the following sequences: STEM-LOOP Sense Strand: 5’-[G][G][U][G][G][A][U][G][A][A][A][C][U][C] [A][G][U][U][U][A][G][C][A][G][C] [C][G][A][A][A][G][G][C][U][G][C]-3’ (SEQ ID NO: 12) Antisense Strand A: 5’-[U][A][A][A][C][U][G][A][G][U][U][U][C][A][U][C][C][A][C][C][G][G]-3’ (SEQ ID NO: 13) BLUNT Sense Strand 5’- [G][G][U][G][G][A][U][G][A][A][A][C][U][C][A][G][U][U][U][A]- 3’ (SEQ ID NO: 14) ^ Antisense B: 5’- [U][A][A][A][C][U][G][A][G][U][U][U][C][A][U][C][C][A][C][C][G][G]- 3’ (SEQ ID NO: 13)
FH12501620.1
Attorney Docket: DCY-13025 In some embodiments, an RNAi oligonucleotide comprises: (i) an antisense strand of 15 to 30 nucleotides in length, a sense strand of 13 to 40 nucleotides in length, wherein the antisense strand has a region of complementarity to a target sequence in a target mRNA in an extra-hepatic tissue; and (ii) one or more ligands. In some embodiments, an RNAi oligonucleotide comprises: (i) an antisense strand of 15 to 30 nucleotides in length, a sense strand of 13 to 40 nucleotides in length, wherein the antisense strand has a region of complementarity to a target sequence in a target mRNA in an extra-hepatic tissue, and wherein the region of complementarity is at least 15 contiguous nucleotides in length; and (ii) one or more ligands, each ligand comprising one or more -L-Y-(CO2H)n groups, or a charged form thereof, wherein L is a linker, Y is alkylene, alkenylene, or alkynylene, and n is 1-6, wherein when Y is alkylene, L comprises -O(CH2)a(OCH2CH2)bNH-, wherein a is 1- 6 and b is 0-5, and wherein the ligand is conjugated to a nucleotide of the sense strand. In some embodiments, an RNAi oligonucleotide comprises: (i) a double-stranded oligonucleotide comprising an antisense strand of 15 to 30 nucleotides in length, a sense strand of 15 to 40 nucleotides in length, wherein the antisense strand has a region of complementarity to a target sequence in a target mRNA in extra- hepatic tissue, and wherein the region of complementarity is at least 15 contiguous nucleotides in length; and (ii) one or more ligands, each ligand comprising one or more C6-24 alkylene-(CO2H)n, or a charged form thereof, conjugated to a nucleotide of the sense strand, wherein n is 1-6, wherein knock down of the target mRNA in the extra-hepatic tissue is greater than knock down of the target mRNA in liver tissue. In some embodiments, an RNAi oligonucleotide comprises: (i) an antisense strand of 15 to 30 nucleotides in length, a sense strand of 13 to 40 nucleotides in length, wherein the antisense strand has a region of complementarity to a target sequence in a target mRNA in an extra-hepatic tissue, and wherein the region of complementarity is at least 15 contiguous nucleotides in length; and (ii) one or more ligands, wherein the ligand is

, and wherein the ligand is conjugated to a nucleotide of the sense strand.
FH12501620.1
Attorney Docket: DCY-13025 In some embodiments, an RNAi oligonucleotide comprises: (i) a double-stranded oligonucleotide comprising an antisense strand of 15 to 30 nucleotides in length, a sense strand of 15 to 40 nucleotides in length, wherein the antisense strand has a region of complementarity to a target sequence in a target mRNA in extra- hepatic tissue, and wherein the region of complementarity is at least 15 contiguous nucleotides in length; and (ii) one or more ligands, wherein the ligand is

, and wherein knock down of the target mRNA in the extra-hepatic tissue is greater than knock down of the target mRNA in liver tissue. In some embodiments, an RNAi oligonucleotide comprises: (i) an antisense strand of 15 to 30 nucleotides in length, a sense strand of 13 to 40 nucleotides in length, wherein the antisense strand has a region of complementarity to a target sequence in a target mRNA in an extra-hepatic tissue, and wherein the region of complementarity is at least 15 contiguous nucleotides in length; and (ii) one or more ligands, wherein the ligand is

, and wherein the ligand is conjugated to a nucleotide of the sense strand. In some embodiments, an RNAi oligonucleotide comprises: (i) a double-stranded oligonucleotide comprising an antisense strand of 15 to 30 nucleotides in length, a sense strand of 15 to 40 nucleotides in length, wherein the antisense strand has a region of complementarity to a target sequence in a target mRNA in extra- hepatic tissue, and wherein the region of complementarity is at least 15 contiguous nucleotides in length; and (ii) one or more ligands, wherein the ligand is
FH12501620.1
Attorney Docket: DCY-13025 wherein knock down of the target mRNA in the extra-hepatic tissue is greater than knock down of the target mRNA in liver tissue. In some embodiments, an RNAi oligonucleotide comprises: (i) an antisense strand of 15 to 30 nucleotides in length, a sense strand of 13 to 40 nucleotides in length, wherein the antisense strand has a region of complementarity to a target sequence in a target mRNA in an extra-hepatic tissue, and wherein the region of complementarity is at least 15 contiguous nucleotides in length; and (ii) one or more ligands, wherein the ligand is selected from

, and wherein the ligand is conjugated to a nucleotide of the sense strand. In some embodiments, an RNAi oligonucleotide comprises: (i) a double-stranded oligonucleotide comprising an antisense strand of 15 to 30 nucleotides in length, a sense strand of 15 to 40 nucleotides in length, wherein the antisense strand has a region of complementarity to a target sequence in a target mRNA in extra- hepatic tissue, and wherein the region of complementarity is at least 15 contiguous nucleotides in length; and
FH12501620.1
Attorney Docket: DCY-13025 (ii) one or more ligands, wherein the ligand is selected from

wherein knock down of the target mRNA in the extra-hepatic tissue is greater than knock down of the target mRNA in liver tissue. In some embodiments, an RNAi oligonucleotide comprises: (i) an antisense strand of 22 nucleotides in length, a sense strand of 36 nucleotides in length, wherein the antisense strand has a region of complementarity to a target sequence in a target mRNA in an extra-hepatic tissue, and wherein the region of complementarity is at least 15 contiguous nucleotides in length; and (ii) one or more ligands, each ligand comprising one or more -L-Y-(CO2H)n groups, or a charged form thereof, wherein L is a linker, Y is alkylene, alkenylene, or alkynylene, and n is 1-6, wherein when Y is alkylene, L comprises -O(CH2)a(OCH2CH2)bNH-, wherein a is 1- 6 and b is 0-5, and wherein the ligand is conjugated to a nucleotide of the sense strand. In some embodiments, an RNAi oligonucleotide comprises: (i) a double-stranded oligonucleotide comprising an antisense strand of 22 nucleotides in length, a sense strand of 36 nucleotides in length, wherein the antisense strand has a region of complementarity to a target sequence in a target mRNA in extra-hepatic tissue, and wherein the region of complementarity is at least 15 contiguous nucleotides in length; and
FH12501620.1
Attorney Docket: DCY-13025 (ii) one or more ligands, each ligand comprising one or more C6-24 alkylene-(CO2H)n, or a charged form thereof, conjugated to a nucleotide of the sense strand, wherein n is 1-6, wherein knock down of the target mRNA in the extra-hepatic tissue is greater than knock down of the target mRNA in liver tissue. In some embodiments, an RNAi oligonucleotide comprises: (i) an antisense strand of 22 nucleotides in length, a sense strand of 36 nucleotides in length, wherein the antisense strand has a region of complementarity to a target sequence in a target mRNA in an extra-hepatic tissue, and wherein the region of complementarity is at least 15 contiguous nucleotides in length; and (ii) one or more ligands, wherein the ligand is

, and wherein the ligand is conjugated to a nucleotide of the sense strand. In some embodiments, an RNAi oligonucleotide comprises: (i) a double-stranded oligonucleotide comprising an antisense strand of 22 nucleotides in length, a sense strand of 36 nucleotides in length, wherein the antisense strand has a region of complementarity to a target sequence in a target mRNA in extra-hepatic tissue, and wherein the region of complementarity is at least 15 contiguous nucleotides in length; and (ii) one or more ligands, wherein the ligand is

, wherein knock down of the target mRNA in the extra-hepatic tissue is greater than knock down of the target mRNA in liver tissue. In some embodiments, an RNAi oligonucleotide comprises: (i) an antisense strand of 22 nucleotides in length, a sense strand of 36 nucleotides in length, wherein the antisense strand has a region of complementarity to a target sequence in a target mRNA in an extra-hepatic tissue, and wherein the region of complementarity is at least 15 contiguous nucleotides in length; and (ii) one or more ligands, wherein the ligand is
FH12501620.1
Attorney Docket: DCY-13025
, and wherein the ligand is conjugated to a nucleotide of the sense strand. In some embodiments, an RNAi oligonucleotide comprises: (i) a double-stranded oligonucleotide comprising an antisense strand of 22 nucleotides in length, a sense strand of 36 nucleotides in length, wherein the antisense strand has a region of complementarity to a target sequence in a target mRNA in extra-hepatic tissue, and wherein the region of complementarity is at least 15 contiguous nucleotides in length; and (ii) one or more ligands, wherein the ligand is

, wherein knock down of the target mRNA in the extra-hepatic tissue is greater than knock down of the target mRNA in liver tissue. In some embodiments, an RNAi oligonucleotide comprises: (i) an antisense strand of 22 nucleotides in length, a sense strand of 36 nucleotides in length, wherein the antisense strand has a region of complementarity to a target sequence in a target mRNA in an extra-hepatic tissue, and wherein the region of complementarity is at least 15 contiguous nucleotides in length; and (ii) one or more ligands, wherein the ligand is selected from
FH12501620.1
Attorney Docket: DCY-13025
, and wherein the ligand is conjugated to a nucleotide of the sense strand. In some embodiments, an RNAi oligonucleotide comprises: (i) a double-stranded oligonucleotide comprising an antisense strand of 22 nucleotides in length, a sense strand of 36 nucleotides in length, wherein the antisense strand has a region of complementarity to a target sequence in a target mRNA in extra-hepatic tissue, and wherein the region of complementarity is at least 15 contiguous nucleotides in length; and
FH12501620.1
Attorney Docket: DCY-13025 (ii) one or more ligands, wherein the ligand is selected from

, wherein knock down of the target mRNA in the extra-hepatic tissue is greater than knock down of the target mRNA in liver tissue. In some embodiments, an RNAi oligonucleotide comprises: (i) an antisense strand of 22 nucleotides in length, a sense strand of 37 nucleotides in length, wherein the antisense strand has a region of complementarity to a target sequence in a target mRNA in an extra-hepatic tissue, and wherein the region of complementarity is at least 15 contiguous nucleotides in length; and (ii) one or more ligands, each ligand comprising one or more -L-Y-(CO2H)n groups, or a charged form thereof, wherein L is a linker, Y is alkylene, alkenylene, or alkynylene, and n is 1-6, wherein when Y is alkylene, L comprises -O(CH2)a(OCH2CH2)bNH-, wherein a is 1- 6 and b is 0-5, and wherein the ligand is conjugated to a nucleotide of the sense strand. In some embodiments, an RNAi oligonucleotide comprises: (i) a double-stranded oligonucleotide comprising an antisense strand of 22 nucleotides in length, a sense strand of 37 nucleotides in length, wherein the antisense strand has a region of complementarity to a target sequence in a target mRNA in extra-hepatic tissue, and wherein the region of complementarity is at least 15 contiguous nucleotides in length; and
FH12501620.1
Attorney Docket: DCY-13025 (ii) one or more ligands, each ligand comprising one or more C6-24 alkylene-(CO2H)n, or a charged form thereof, conjugated to a nucleotide of the sense strand, wherein n is 1-6, wherein knock down of the target mRNA in the extra-hepatic tissue is greater than knock down of the target mRNA in liver tissue. In some embodiments, an RNAi oligonucleotide comprises: (i) an antisense strand of 22 nucleotides in length, a sense strand of 37 nucleotides in length, wherein the antisense strand has a region of complementarity to a target sequence in a target mRNA in an extra-hepatic tissue, and wherein the region of complementarity is at least 15 contiguous nucleotides in length; and (ii) one or more ligands, wherein the ligand is

, and wherein the ligand is conjugated to a nucleotide of the sense strand. In some embodiments, an RNAi oligonucleotide comprises: (i) a double-stranded oligonucleotide comprising an antisense strand of 22 nucleotides in length, a sense strand of 37 nucleotides in length, wherein the antisense strand has a region of complementarity to a target sequence in a target mRNA in extra-hepatic tissue, and wherein the region of complementarity is at least 15 contiguous nucleotides in length; and (ii) one or more ligands, wherein the ligand is

,wherein knock down of the target mRNA in the extra-hepatic tissue is greater than knock down of the target mRNA in liver tissue. In some embodiments, an RNAi oligonucleotide comprises: (i) an antisense strand of 22 nucleotides in length, a sense strand of 37 nucleotides in length, wherein the antisense strand has a region of complementarity to a target sequence in a target mRNA in an extra-hepatic tissue, and wherein the region of complementarity is at least 15 contiguous nucleotides in length; and (ii) one or more ligands, wherein the ligand is
FH12501620.1
Attorney Docket: DCY-13025 , and wherein the ligand is conjugated to a nucleotide of the sense strand. In some embodiments, an RNAi oligonucleotide comprises: (i) a double-stranded oligonucleotide comprising an antisense strand of 22 nucleotides in length, a sense strand of 37 nucleotides in length, wherein the antisense strand has a region of complementarity to a target sequence in a target mRNA in extra-hepatic tissue, and wherein the region of complementarity is at least 15 contiguous nucleotides in length; and (ii) one or more ligands, wherein the ligand is

,wherein knock down of the target mRNA in the extra-hepatic tissue is greater than knock down of the target mRNA in liver tissue. In some embodiments, an RNAi oligonucleotide comprises: (i) an antisense strand of 22 nucleotides in length, a sense strand of 37 nucleotides in length, wherein the antisense strand has a region of complementarity to a target sequence in a target mRNA in an extra-hepatic tissue, and wherein the region of complementarity is at least 15 contiguous nucleotides in length; and (ii) one or more ligands, wherein the ligand is selected from
FH12501620.1
Attorney Docket: DCY-13025
, and wherein the ligand is conjugated to a nucleotide of the sense strand. In some embodiments, an RNAi oligonucleotide comprises: (i) a double-stranded oligonucleotide comprising an antisense strand of 22 nucleotides in length, a sense strand of 37 nucleotides in length, wherein the antisense strand has a region of complementarity to a target sequence in a target mRNA in extra-hepatic tissue, and wherein the region of complementarity is at least 15 contiguous nucleotides in length; and (ii) one or more ligands, wherein the ligand is selected from
FH12501620.1
Attorney Docket: DCY-13025
,wherein knock down of the target mRNA in the extra-hepatic tissue is greater than knock down of the target mRNA in liver tissue. In some embodiments, an RNAi oligonucleotide comprises: (i) an antisense strand of 22 nucleotides in length, a sense strand of 20 nucleotides in length, wherein the antisense strand has a region of complementarity to a target sequence in a target mRNA in an extra-hepatic tissue, and wherein the region of complementarity is at least 15 contiguous nucleotides in length; and (ii) one or more ligands, each ligand comprising one or more -L-Y-(CO2H)n groups, or a charged form thereof, wherein L is a linker, Y is alkylene, alkenylene, or alkynylene, and n is 1-6, wherein when Y is alkylene, L comprises -O(CH
2)
a(OCH
2CH
2)
bNH-, wherein a is 1- 6 and b is 0-5, and wherein the ligand is conjugated to a nucleotide of the sense strand. In some embodiments, an RNAi oligonucleotide comprises: (i) a double-stranded oligonucleotide comprising an antisense strand of 20 nucleotides in length, a sense strand of 36 nucleotides in length, wherein the antisense strand has a region of complementarity to a target sequence in a target mRNA in extra-hepatic tissue, and wherein the region of complementarity is at least 15 contiguous nucleotides in length; and
FH12501620.1
Attorney Docket: DCY-13025 (ii) one or more ligands, each ligand comprising one or more C6-24 alkylene-(CO2H)n, or a charged form thereof, conjugated to a nucleotide of the sense strand, wherein n is 1-6, wherein knock down of the target mRNA in the extra-hepatic tissue is greater than knock down of the target mRNA in liver tissue. In some embodiments, an RNAi oligonucleotide comprises: (i) an antisense strand of 22 nucleotides in length, a sense strand of 20 nucleotides in length, wherein the antisense strand has a region of complementarity to a target sequence in a target mRNA in an extra-hepatic tissue, and wherein the region of complementarity is at least 15 contiguous nucleotides in length; and (ii) one or more ligands, wherein the ligand is

, and wherein the ligand is conjugated to a nucleotide of the sense strand. In some embodiments, an RNAi oligonucleotide comprises: (i) a double-stranded oligonucleotide comprising an antisense strand of 20 nucleotides in length, a sense strand of 36 nucleotides in length, wherein the antisense strand has a region of complementarity to a target sequence in a target mRNA in extra-hepatic tissue, and wherein the region of complementarity is at least 15 contiguous nucleotides in length; and (ii) one or more ligands, wherein the ligand is

,wherein knock down of the target mRNA in the extra-hepatic tissue is greater than knock down of the target mRNA in liver tissue. In some embodiments, an RNAi oligonucleotide comprises: (i) an antisense strand of 22 nucleotides in length, a sense strand of 20 nucleotides in length, wherein the antisense strand has a region of complementarity to a target sequence in a target mRNA in an extra-hepatic tissue, and wherein the region of complementarity is at least 15 contiguous nucleotides in length; and (ii) one or more ligands, wherein the ligand is
FH12501620.1
Attorney Docket: DCY-13025 , and wherein the ligand is conjugated to a nucleotide of the sense strand. In some embodiments, an RNAi oligonucleotide comprises: (i) a double-stranded oligonucleotide comprising an antisense strand of 20 nucleotides in length, a sense strand of 36 nucleotides in length, wherein the antisense strand has a region of complementarity to a target sequence in a target mRNA in extra-hepatic tissue, and wherein the region of complementarity is at least 15 contiguous nucleotides in length; and (ii) one or more ligands, wherein the ligand is

,wherein knock down of the target mRNA in the extra-hepatic tissue is greater than knock down of the target mRNA in liver tissue. In some embodiments, an RNAi oligonucleotide comprises: (i) an antisense strand of 22 nucleotides in length, a sense strand of 20 nucleotides in length, wherein the antisense strand has a region of complementarity to a target sequence in a target mRNA in an extra-hepatic tissue, and wherein the region of complementarity is at least 15 contiguous nucleotides in length; and
FH12501620.1
Attorney Docket: DCY-13025 (ii) one or more ligands, wherein the ligand is selected from
, and wherein the ligand is conjugated to a nucleotide of the sense strand. In some embodiments, an RNAi oligonucleotide comprises: (i) a double-stranded oligonucleotide comprising an antisense strand of 20 nucleotides in length, a sense strand of 36 nucleotides in length, wherein the antisense strand has a region of complementarity to a target sequence in a target mRNA in extra-hepatic tissue, and wherein the region of complementarity is at least 15 contiguous nucleotides in length; and
FH12501620.1
Attorney Docket: DCY-13025 (ii) one or more ligands, wherein the ligand is selected from

,wherein knock down of the target mRNA in the extra-hepatic tissue is greater than knock down of the target mRNA in liver tissue. In some embodiments, an RNAi oligonucleotide comprises a sense and antisense strand, wherein the sense strand is 20 nucleotides in length, and the antisense strand is 22 nucleotides in length, wherein the sense strand comprises at least one oligonucleotide-ligand conjugate, and wherein the oligonucleotide-ligand conjugate is selected from Formulas (AI), (AII), (AIII), (AIV), (AV), (BI), (BII), (CI), or (CII). In some embodiments, an RNAi oligonucleotide comprises a sense and antisense strand, wherein the sense strand is 36 nucleotides in length, and the antisense strand is 22 nucleotides in length, wherein the sense strand comprises at least one oligonucleotide-ligand conjugate, and wherein the oligonucleotide-ligand conjugate is selected from Formulas (AI), (AII), (AIII), (AIV), (AV), (BI), (BII), (CI), or (CII). In some embodiments, an RNAi oligonucleotide comprises a sense and antisense strand, wherein the sense strand is 37 nucleotides in length, and the antisense strand is 22 nucleotides in length, wherein the sense strand comprises at least one
FH12501620.1
Attorney Docket: DCY-13025 oligonucleotide-ligand conjugate, and wherein the oligonucleotide-ligand conjugate is selected from Formulas (AI), (AII), (AIII), (AIV), (AV), (BI), (BII), (CI), or (CII). In some embodiments, an RNAi oligonucleotide comprises a sense and antisense strand, wherein the sense strand is 20 nucleotides in length, and the antisense strand is 22 nucleotides in length, wherein the sense strand comprises at least one oligonucleotide-ligand conjugate, wherein the oligonucleotide-ligand conjugate is selected from Formulas (AI), (AII), (AIII), (AIV), (AV), (BI), (BII), (CI), or (CII), and wherein the nucleobase (B) is the nucleobase at position 1 of the sense strand. In some embodiments, an RNAi oligonucleotide comprises a sense and antisense strand, wherein the sense strand is 20 nucleotides in length, and the antisense strand is 22 nucleotides in length, wherein the sense strand comprises at least one oligonucleotide-ligand conjugate, wherein the oligonucleotide-ligand conjugate is selected from Formulas (AI), (AII), (AIII), (AIV), (AV), (BI), (BII), (CI), or (CII), and wherein the nucleobase (B) is the nucleobase at position 20 of the sense strand. In some embodiments, an RNAi oligonucleotide comprises a sense and antisense strand, wherein the sense strand is 36 nucleotides in length, and the antisense strand is 22 nucleotides in length, wherein the sense strand comprises at least one oligonucleotide-ligand conjugate, wherein the oligonucleotide- ligand conjugate is selected from Formulas (AI), (AII), (AIII), (AIV), (AV), (BI), (BII), (CI), or (CII), and wherein the nucleobase (B) is the nucleobase at position 28 of the sense strand. In some embodiments, an RNAi oligonucleotide comprises a sense and antisense strand, wherein the sense strand is 37 nucleotides in length, and the antisense strand is 22 nucleotides in length, wherein the sense strand comprises at least one oligonucleotide-ligand conjugate, wherein the oligonucleotide-ligand conjugate is selected from Formulas (AI), (AII), (AIII), (AIV), (AV), (BI), (BII), (CI), or (CII), and wherein the nucleobase (B) is the nucleobase at position 29 of the sense strand. In some embodiments, an RNAi oligonucleotide comprises a sense and antisense strand, wherein the sense strand is 36 nucleotides in length, and the antisense strand is 22 nucleotides in length, wherein the sense strand comprises at least one oligonucleotide-ligand conjugate, wherein the oligonucleotide-ligand conjugate is selected from Formulas (AI), (AII), (AIII), (AIV), (AV), (BI), (BII), (CI), or (CII), and wherein the nucleobase (B) is the nucleobase in the tetraloop of the sense strand. In some embodiments, an RNAi oligonucleotide comprises an oligonucleotide-ligand conjugate of any one of Formulas (AI), (AII), (AIII), (AIV), (AV), (BI), (BII), (CI), or (CII). In some embodiments, an RNAi oligonucleotide comprises at least one oligonucleotide-ligand conjugate of any of Formulas (AI), (AII), (AIII), (AIV), (AV), (BI), (BII), (CI), or (CII). In some embodiments, an RNAi oligonucleotide comprises at least two oligonucleotide-ligand
FH12501620.1
Attorney Docket: DCY-13025 conjugates selected from Formulas (AI), (AII), (AIII), (AIV), (AV), (BI), (BII), (CI), or (CII). In some embodiments, an RNAi oligonucleotide comprises at least two oligonucleotide-ligand conjugates selected from Formulas (AI), (AII), (AIII), (AIV), (AV), (BI), (BII), (CI), or (CII), wherein the at least two oligonucleotide-ligand conjugates are selected from the same Formula. In some embodiments, an RNAi oligonucleotide comprises at least two oligonucleotide-ligand conjugates selected from Formulas (AI), (AII), (AIII), (AIV), (AV), (BI), (BII), (CI), or (CII), wherein the at least two oligonucleotide-ligand conjugates are selected from a different Formula. In some embodiments, an RNAi oligonucleotide comprises a sense and antisense strand, wherein the sense strand comprises an oligonucleotide-ligand conjugate of any one of Formulas (AI), (AII), (AIII), (AIV), (AV), (BI), (BII), (CI), or (CII), wherein the nucleobase (B) is the nucleobase at position 1 of the sense strand. In some embodiments, an RNAi oligonucleotide comprises a sense and antisense strand, wherein the sense strand comprises an oligonucleotide-ligand conjugate of any one of Formulas (AI), (AII), (AIII), (AIV), or (AV), wherein the nucleobase (B) is the nucleobase at position 2 of the sense strand. In some embodiments, an RNAi oligonucleotide comprises a sense and antisense strand, wherein the sense strand comprises an oligonucleotide-ligand conjugate of any one of Formulas (AI), (AII), (AIII), (AIV), (AV), (BI), (BII), (CI), or (CII), wherein the nucleobase (B) is the nucleobase at position 20 of the sense strand. In some embodiments, an RNAi oligonucleotide comprises a sense and antisense strand, wherein the sense strand comprises an oligonucleotide-ligand conjugate of any one of Formulas (AI), (AII), (AIII), (AIV), (AV), (BI), (BII), (CI), or (CII), wherein the nucleobase (B) is the nucleobase at position 28 of the sense strand. In some embodiments, an RNAi oligonucleotide comprises a sense and antisense strand, wherein the sense strand comprises an oligonucleotide-ligand conjugate of any one of Formulas (AI), (AII), (AIII), (AIV), (AV), (BI), (BII), (CI), or (CII), wherein the nucleobase (B) is the nucleobase at position 29 of the sense strand. In some embodiments, an RNAi oligonucleotide comprises a sense and antisense strand, wherein the sense strand comprises an oligonucleotide-ligand conjugate of any one of Formulas (AI), (AII), (AIII), (AIV), or (AV), wherein the nucleobase (B) is a nucleobase in the tetraloop of the sense strand. In some embodiments, an RNAi oligonucleotide comprises a sense and antisense strand, wherein the sense strand comprises a first and a second oligonucleotide-ligand conjugate, wherein:
FH12501620.1
Attorney Docket: DCY-13025 (i) the first oligonucleotide-ligand conjugate is selected from Formulas (AI), (AII), (AIII), (AIV), (AV), (BI), (BII), (CI), or (CII), wherein the nucleobase (B) is the nucleobase at position 1 of the sense strand; and (ii) the second oligonucleotide-ligand conjugate is selected from Formulas (AI), (AII), (AIII), (AIV), or (AV), wherein the nucleobase (B) is the nucleobase at position 2 of the sense strand. In some embodiments, an RNAi oligonucleotide comprises a sense and antisense strand, wherein the sense strand comprises a first and a second oligonucleotide-ligand conjugate, wherein: (i) the first oligonucleotide-ligand conjugate is selected from Formulas (AI), (AII), (AIII), (AIV), (AV), (BI), (BII), (CI), or (CII), wherein the nucleobase (B) is the nucleobase at position 1 of the sense strand; and (ii) the second oligonucleotide-ligand conjugate is selected from Formulas (AI), (AII), (AIII), (AIV), or (AV), wherein the nucleobase (B) is the nucleobase at position 20 of the sense strand. In some embodiments, an RNAi oligonucleotide comprises a sense and antisense strand, wherein the sense strand comprises a first and a second oligonucleotide-ligand conjugate, wherein: (i) the first oligonucleotide-ligand conjugate is selected from Formulas (AI), (AII), (AIII), (AIV), (AV), (BI), (BII), (CI), or (CII), wherein the nucleobase (B) is the nucleobase at position 1 of the sense strand; and (ii) the second oligonucleotide-ligand conjugate is selected from Formulas (AI), (AII), (AIII), (AIV), (AV), (BI), (BII), (CI), or (CII), wherein the nucleobase (B) is a nucleobase in the tetraloop of the sense strand. In some embodiments, an RNAi oligonucleotide comprises a sense and antisense strand, wherein the sense strand comprises a first and a second oligonucleotide-ligand conjugate, wherein: (i) the first oligonucleotide-ligand conjugate is selected from Formulas (AI), (AII), (AIII), (AIV), (AV), (BI), (BII), (CI), or (CII), wherein the nucleobase (B) is the nucleobase at position 1 of the sense strand; and (ii) the second oligonucleotide-ligand conjugate is selected from Formulas (AI), (AII), (AIII), (AIV), (AV), (BI), (BII), (CI), or (CII), wherein the nucleobase (B) is the nucleobase at position 28 of the sense strand.
FH12501620.1
Attorney Docket: DCY-13025 In some embodiments, an RNAi oligonucleotide comprises a sense and antisense strand, wherein the sense strand comprises a first and a second oligonucleotide-ligand conjugate, wherein: (i) the first oligonucleotide-ligand conjugate is selected from Formulas (AI), (AII), (AIII), (AIV), (AV), (BI), (BII), (CI), or (CII), wherein the nucleobase (B) is the nucleobase at position 1 of the sense strand; and (ii) the second oligonucleotide-ligand conjugate is selected from Formulas (AI), (AII), (AIII), (AIV), (AV), (BI), (BII), (CI), or (CII), wherein the nucleobase (B) is the nucleobase at position 29 of the sense strand. In some embodiments, an RNAi oligonucleotide comprises a sense and antisense strand, wherein the sense strand comprises a first and a second oligonucleotide-ligand conjugate, wherein: (i) the first oligonucleotide-ligand conjugate is selected from Formulas (AI), (AII), (AIII), (AIV), (AV), (BI), (BII), (CI), or (CII), wherein the nucleobase (B) is the nucleobase at position 1 of the sense strand; and (ii) the second oligonucleotide-ligand conjugate is selected from Formulas (AI), (AII), (AIII), (AIV), or (AV), wherein the nucleobase (B) is the nucleobase at position 2 of the sense strand. In some embodiments, an RNAi oligonucleotide comprises a sense and antisense strand, wherein the sense strand comprises a first and a second oligonucleotide-ligand conjugate, wherein: (i) the first oligonucleotide-ligand conjugate is selected from Formulas (AI), (AII), (AIII), (AIV), (AV), (BI), (BII), (CI), or (CII), wherein the nucleobase (B) is the nucleobase at position 1 of the sense strand; and (ii) the second oligonucleotide-ligand conjugate is selected from Formulas (AI), (AII), (AIII), (AIV), (AV), (BI), (BII), (CI), or (CII), wherein the nucleobase (B) is the nucleobase at position 20 of the sense strand. In some embodiments, an RNAi oligonucleotide comprises a sense and antisense strand, wherein the sense strand is 20 nucleotides in length and the antisense strand is 22 nucleotides in length, wherein the sense strand comprises a first and a second oligonucleotide- ligand conjugate, and wherein: (i) the first oligonucleotide-ligand conjugate is selected from Formulas (AI), (AII), (AIII), (AIV), (AV), (BI), (BII), (CI), or (CII), wherein the nucleobase (B) is the nucleobase at position 1 of the sense strand; and
FH12501620.1
Attorney Docket: DCY-13025 (ii) the second oligonucleotide-ligand conjugate is selected from Formulas (AI), (AII), (AIII), (AIV), or (AV), wherein the nucleobase (B) is the nucleobase at position 2 of the sense strand. In some embodiments, an RNAi oligonucleotide comprises a sense and antisense strand, wherein the sense strand is 20 nucleotides in length and the antisense strand is 22 nucleotides in length, wherein the sense strand comprises a first and a second oligonucleotide- ligand conjugate, and wherein: (i) the first oligonucleotide-ligand conjugate is selected from Formulas (AI), (AII), (AIII), (AIV), (AV), (BI), (BII), (CI), or (CII), wherein the nucleobase (B) is the nucleobase at position 1 of the sense strand; and (ii) the second oligonucleotide-ligand conjugate is selected from Formulas (AI), (AII), (AIII), (AIV), (AV), (BI), (BII), (CI), or (CII), wherein the nucleobase (B) is the nucleobase at position 20 of the sense strand. In some embodiments, an RNAi oligonucleotide comprises a sense and antisense strand, wherein the sense strand is 36 nucleotides in length and the antisense strand is 22 nucleotides in length, wherein the sense strand comprises a first and a second oligonucleotide- ligand conjugate, and wherein: (i) the first oligonucleotide-ligand conjugate is selected from Formulas (AI), (AII), (AIII), (AIV), (AV), (BI), (BII), (CI), or (CII), wherein the nucleobase (B) is the nucleobase at position 1 of the sense strand; and (ii) the second oligonucleotide-ligand conjugate is selected from Formulas (AI), (AII), (AIII), (AIV), or (AV), wherein the nucleobase (B) is a nucleobase in the tetraloop of the sense strand. In some embodiments, an RNAi oligonucleotide comprises a sense and antisense strand, wherein the sense strand is 36 nucleotides in length and the antisense strand is 22 nucleotides in length, wherein the sense strand comprises a first and a second oligonucleotide- ligand conjugate, and wherein: (i) the first oligonucleotide-ligand conjugate is selected from Formulas (AI), (AII), (AIII), (AIV), (AV), (BI), (BII), (CI), or (CII), wherein the nucleobase (B) is the nucleobase at position 1 of the sense strand; and (ii) the second oligonucleotide-ligand conjugate is selected from Formulas (AI), (AII), (AIII), (AIV), or (AV), wherein the nucleobase (B) is the nucleobase at position 28 of the sense strand.
FH12501620.1
Attorney Docket: DCY-13025 In some embodiments, an RNAi oligonucleotide comprises a sense and antisense strand, wherein the sense strand is 37 nucleotides in length and the antisense strand is 22 nucleotides in length, wherein the sense strand comprises a first and a second oligonucleotide- ligand conjugate, and wherein: (i) the first oligonucleotide-ligand conjugate is selected from Formulas (AI), (AII), (AIII), (AIV), (AV), (BI), (BII), (CI), or (CII), wherein the nucleobase (B) is the nucleobase at position 1 of the sense strand; and (ii) the second oligonucleotide-ligand conjugate is selected from Formulas (AI), (AII), (AIII), (AIV), or (AV), wherein the nucleobase (B) is the nucleobase at position 29 of the sense strand. General Methods of Providing the Nucleic Acids and Analogues Thereof The nucleic acids and analogues thereof comprising an oligonucleotide-ligand conjugate described herein can be made using a variety of synthetic methods known in the art, including standard phosphoramidite methods. Any phosphoramidite synthesis method can be used to synthesize the provided nucleic acids of this disclosure. In some embodiments, phosphoramidites are used in a solid phase synthesis method to yield reactive intermediate phosphite compounds, which are subsequently oxidized using known methods to produce phosphonate-modified oligonucleotides, typically with a phosphodiester or phosphorothioate internucleotide linkages. The oligonucleotide synthesis of the present disclosure can be performed in either direction: from 5^ to 3^ or from 3^ to 5^ using art known methods. In some embodiments, the method for synthesizing a provided nucleic acid comprises (a) attaching a nucleoside or analogue thereof to a solid support via a covalent linkage; (b) coupling a nucleoside phosphoramidite or analogue thereof to a reactive hydroxyl group on the nucleoside or analogue thereof of step (a) to form an internucleotide bond there between, wherein any uncoupled nucleoside or analogue thereof on the solid support is capped with a capping reagent; (c) oxidizing said internucleotide bond with an oxidizing agent; and (d) repeating steps (b) to (c) iteratively with subsequent nucleoside phosphoramidites or analogue thereof to form a nucleic acid or analogue thereof, wherein at least the nucleoside or analogue thereof of step (a), the nucleoside phosphoramidite or analogue thereof of step (b) or at least one of the subsequent nucleoside phosphoramidites or analogues thereof of step (d) comprises a hydrocarbon chain bearing one or more carboxyl groupsas described herein. Typically, the coupling, capping/oxidizing steps and optionally, the deprotecting steps, are repeated until the oligonucleotide reaches the desired length and/or sequence, after which it is cleaved from the
FH12501620.1
Attorney Docket: DCY-13025 solid support. In some embodiments, an oligonucleotide is prepared comprising 1-3 nucleic acid or analogues thereof comprising hydrocarbon chain bearing one or more carboxyl groups on a tetraloop. In Scheme A below, where a particular protecting group, leaving group, or transformation condition is depicted, one of ordinary skill in the art will appreciate that other protecting groups, leaving groups, and transformation conditions are also suitable and are contemplated. Certain reactive functional groups (e.g., -N(H)-, -OH, etc.) envisioned in the genera in Scheme A requiring additional protection group strategies are also contemplated and is appreciated by those having ordinary skill in the art. Such groups and transformations are described in detail in M
ARCH'
S A
DVANCED O
RGANIC C
HEMISTRY: R
EACTIONS, M
ECHANISMS,
AND S
TRUCTURE, M. B. Smith and J. March, 5
th Edition, John Wiley & Sons, 2001, C
OMPREHENSIVE O
RGANIC T
RANSFORMATIONS, (R. C. Larock, 2
nd Edition, John Wiley & Sons, 1999), and PROTECTING GROUPS IN ORGANIC SYNTHESIS, (T. W. Greene and P. G. M. Wuts, 3
rd edition, John Wiley & Sons, 1999), the entirety of each of which is hereby incorporated herein by reference. In some embodiments, nucleic acids, and analogues thereof of the present disclosure are generally prepared according to Scheme A, Scheme A1 and Scheme B set forth below:
FH12501620.1
Attorney Docket: DCY-13025 Scheme A: Synthesis of Ligand Conjugated Oligonucleotides of the Disclosure
Scheme A1: Synthesis of Conjugated Oligonucleotides of the Disclosure
FH12501620.1
Attorney Docket: DCY-13025
FH12501620.1
Attorney Docket: DCY-13025 As depicted in Scheme A and Scheme A1 above, a nucleic acid or analogue thereof of formula I-1 is conjugated with one or more ligand/lipophilic compound to form a compound of formula I or Ia comprising one more ligand conjugates. Typically, conjugation is performed through an esterification or amidation reaction between a nucleic acid or analogue thereof of formula I-1 or I-1a and one or more hydrocarbon bearing carboxylic acid in series or in parallel by known techniques in the art. Nucleic acid or analogue thereof of formula I or Ia can then be deprotected to form a compound of formula I-2 or I-2a and protected with a suitable hydroxyl protecting group (e.g., DMTr) to form a compound of formula I-3 or I-3a. In one aspect, nucleic acid-ligand conjugates of formula I-3 or I-3a can be covalently attached to a solid support (e.g., through a succinic acid linking group) to form a solid support nucleic acid- ligand conjugate or analogue thereof of formula I-4 or I-4a comprising one or more hydrocarbon with carboxylic acid. In another aspect, a nucleic acid- ligand conjugates of formula I-3 or I-3a can react with a P(III) forming reagent (e.g., 2-cyanoethyl N,N-di- isopropylchlorophosphoramidite) to form a nucleic acid or analogue thereof of formula I-5 or I-5a comprising a P(III) group. A nucleic acid-ligand conjugate or analogue thereof of formula I-5 or I-5a can then be subjected to oligomerization forming conditions preformed using known and commonly applied processes to prepare oligonucleotides in the art. For example, the compound of formula I-5 or I-5a is coupled to a solid supported nucleic acid-ligand conjugate or analogue thereof bearing a 5’-hydroxyl group. Further steps can comprise one or more deprotections, couplings, phosphite oxidation, and/or cleavage from the solid support to provide an oligonucleotide of various nucleotide lengths, including one or more conjugate nucleotide units represented by a compound of formula II-1 or II-Ia. Each of B, L, R
1, R
2, and Z is as defined above and in Formula (AI), (BI), or (CI) or as described herein. X, as provided in formula I-1, is a reactive functional group or leaving group. In some embodiments, PG
1 and PG
2 comprise silyl ethers or cyclic silylene derivatives that can be removed under acidic conditions or with fluoride anion. In some embodiments, PG
1 and PG
2 are taken together with their intervening atoms to form a cyclic diol protecting group, such as a cyclic acetal or ketal. PG
3 used for protection of the 5’-hydroxyl group includes an acid labile protecting group such as trityl, 4-methyoxytrityl, 4,4’-dimethyoxytrityl, 4,4’,4’’-trimethyoxytrityl, 9-phenyl-xanthen- 9-yl, 9-(p-tolyl)-xanthen-9-yl, pixyl, 2,7-dimethylpixyl, and the like. E is 2-cyanoethyl when 2-cyanoethyl N,N-diisopropylchlorophosphoramidite or 2-cyanoethyl phosphorodichloridate is used as a P(III) forming reagent. X
3 is O-, -S-, or a covalent bond. R
3 is hydrogen or a suitable protecting group. n is 1, 2, or 3.
FH12501620.1
Attorney Docket: DCY-13025 Scheme B: Post-Synthetic Conjugation of Oligonucleotides of the Disclosure

As depicted in Scheme B above, a nucleic acid or analogue thereof of formula I-1 can be deprotected to form a compound of formula I-6, protected with a suitable hydroxyl protecting group (e.g., DMTr) to form a compound of formula I-7, and reacted with a P(III) forming reagent (e.g., 2-cyanoethyl N,N-di-isopropylchlorophosphoramidite) to form a nucleic acid or analogue thereof of formula I-8 comprising a P(III) group. Next, a nucleic acid or analogue thereof of formula I-8 is subjected to oligomerization forming conditions preformed using known and commonly applied processes to prepare oligonucleotides in the art. For example, the compound of formula I-8 is coupled to a solid supported nucleic acid or analogue thereof bearing a 5’-hydroxyl group. Further steps can comprise one or more deprotections, couplings, phosphite oxidation, and/or cleavage from the solid support to provide an oligonucleotide of various nucleotide lengths represented by a compound of formula II-2. An oligonucleotide of formula II-2 can then be conjugated with one or more ligands e.g., hydrocarbon bearing carboxylic acid to form a compound of formula II-1 comprising one ligand conjugate (n=1). Typically, conjugation is performed through an esterification or amidation reaction between a nucleic acid or analogue thereof of formula II-2 and one or more hydrocarbon bearing carboxylic acid in series or in parallel by known techniques in the art. Each of B, E, L, ligand, LC, n, PG
1, PG
2, PG
3, R
1, R
2, R
3, X
3, and Z is as defined above. In some embodiments, nucleic acids, and analogues thereof of the present disclosure
FH12501620.1
Attorney Docket: DCY-13025 are prepared according to Scheme C and Scheme D set forth below: Scheme C: Synthesis of Conjugated Oligonucleotides of the Disclosure
As depicted in Scheme C above, a nucleic acid or analogue thereof of formula C1 is protected to form a compound of formula C2. Nucleic acid or analogue thereof of formula C2 is then alkylated (e.g., using DMSO and acetic acid via the Pummerer rearrangement) to form a monothioacetal compound of formula C3. Next, nucleic acid or analogue thereof of formula C3 is coupled with C4 under appropriate conditions (e.g., mild oxidizing conditions) to form a nucleic acid or analogue thereof of formula C5. Nucleic acid or analogue thereof of formula C5 can then be deprotected to form a compound of formula C6 and coupled with a ligand (adamantyl or lipophilic compound (e.g., a fatty acid)) of formula C7 under appropriate amide forming conditions (e.g., HATU, DIPEA), to form a nucleic acid-ligand conjugate or analogue thereof of formula I-b comprising a hydrocarbon chain bearing one or more carboxyl groups
FH12501620.1
Attorney Docket: DCY-13025 of the disclosure. Nucleic acid-ligand conjugate or analogue thereof of formula I-b can then be deprotected to form a compound of formula C8 and protected with a suitable hydroxyl protecting group (e.g., DMTr) to form a compound of formula C9. In one aspect, nucleic acid, or analogue thereof of formula C9 can be covalently attached to a solid support (e.g., through a succinic acid linking group) to form a solid support nucleic acid-ligand conjugate or analogue thereof of formula C10 comprising a ligand conjugate of the disclosure. In another aspect, a nucleic acid-ligand conjugate or analogue thereof of formula C9 can reacted with a P(III) forming reagent (e.g., 2-cyanoethyl N,N-di-isopropylchlorophosphoramidite) to form a nucleic acid-ligand conjugate or analogue thereof of formula C11 comprising a P(III) group. A nucleic acid-ligand conjugate or analogue thereof of formula C11 can then be subjected to oligomerization forming conditions preformed using known and commonly applied processes to prepare oligonucleotides in the art. For example, the compound of formula C11 is coupled to a solid supported nucleic acid-ligand conjugate or analogue thereof bearing a 5’-hydroxyl group. Further steps can comprise one or more deprotections, couplings, phosphite oxidation, and/or cleavage from the solid support to provide an oligonucleotide of various nucleotide lengths, including one or more conjugate nucleotide units represented by a compound of formula II-b-3. Each of B, E, PG
1, PG
2, PG
3, R
1, R
2, R
3, R
4, a, b, X
1, X
2, X
3, Y, and Z is as defined above. W is -(CH
2)
a-. L
2 is -(OCH
2CH
2)
b-. PG
3 is an appropriate protecting group. Scheme D: Post-Synthetic Conjugation of Oligonucleotides of the Disclosure
FH12501620.1
Attorney Docket: DCY-13025
Each of B, E, L
2, PG
1, PG
2, PG
3, PG
4, R
1, R
2, R
3, R
4, X
3, W, Y, and Z is as defined above and described herein. As depicted in Scheme D above, a nucleic acid or analogue thereof of formula C5 can be selectively deprotected to form a compound of formula D1, protected with a suitable hydroxyl protecting group (e.g., DMTr) to form a compound of formula D2, and reacted with a P(III) forming reagent (e.g., 2-cyanoethyl N,N-di- isopropylchlorophosphoramidite) to form a nucleic acid or analogue thereof of formula D3. Next, a nucleic acid or analogue thereof of formula D3 is subjected to oligomerization forming conditions preformed using known and commonly applied processes to prepare oligonucleotides in the art. For example, the compound of formula D3 is coupled to a solid supported nucleic acid or analogue thereof bearing a 5’-hydroxyl group. Further steps can comprise one or more deprotections, couplings, phosphite oxidation, and/or cleavage from the solid support to provide an oligonucleotide of various nucleotide lengths, represented by a compound of formula D4. An oligonucleotide of formula D4 can then be deprotected to form a compound of formula D5 and coupled with a hydrophobic ligand (e.g., hydrocarbon chain moiety) to form a compound of formula C7 (e.g., hydrocarbon chain moeity) under appropriate amide forming conditions (e.g., HATU, DIPEA), to form an oligonucleotide of formula II-b-3 comprising a ligand (e.g., a fatty acid) conjugate of the disclosure. One of skill in the art will appreciate that various functional groups present in the nucleic acid or analogues thereof of the disclosure such as aliphatic groups, alcohols,
FH12501620.1
Attorney Docket: DCY-13025 carboxylic acids, esters, amides, aldehydes, halogens, and nitriles can be interconverted by techniques well known in the art including, but not limited to reduction, oxidation, esterification, hydrolysis, partial oxidation, partial reduction, halogenation, dehydration, partial hydration, and hydration. See for example, “MARCH’S ADVANCED ORGANIC CHEMISTRY”, (5
th Ed., Ed.: Smith, M.B. and March, J., John Wiley & Sons, New York: 2001), the entirety of each of which is herein incorporated by reference. Such interconversions may require one or more of the aforementioned techniques, and certain methods for synthesizing the provided nucleic acids of the disclosure are described below in the Exemplification. In some embodiments, the present disclosure provides a method for preparing an oligonucleotide comprising one or more conjugate, said conjugate unit represent by formula II-a-1:
II-a-1 or a pharmaceutically acceptable salt thereof, comprising the steps of: (a) providing a nucleic acid or analogue thereof of formula I-5a:
or salt thereof, and (b) oligomerizing said compound of formula I-5a to form a compound of formula II-1a, wherein each of B, E, L, LC, n, PG
3, R
1, R
2, R
3, X
3, and Z is as defined above and described herein. In step (b) above, oligomerizing refers to preforming oligomerization forming conditions using known and commonly applied processes to prepare oligonucleotides in the art. For example, the compound of formula I-5a is coupled to a solid supported nucleic acid or analogue thereof bearing a 5’-hydroxyl group. Further steps can comprise one or more deprotections, couplings, phosphite oxidation, and cleavage from the solid support to provide an oligonucleotide of various nucleotide lengths, represented by a compound of formula II-1a
FH12501620.1
Attorney Docket: DCY-13025 comprising a conjugate of the disclosure. In some embodiments, the present disclosure provides a method for preparing an oligonucleotide comprising one or more conjugate, further comprising preparing a nucleic acid or analogue thereof of formula I-5a:
I-5a or a salt thereof, comprising the steps of: (a) providing a nucleic acid or analogue thereof of formula Ia:
or salt thereof, (b) deprotecting said nucleic acid or analogue thereof of formula Ia to form a compound of formula I-2a:
or salt thereof, (c) protecting said nucleic acid or analogue thereof of formula I-2 to form a compound of formula I-3a:
I-3a or salt thereof, and
FH12501620.1
Attorney Docket: DCY-13025 (d) treating said nucleic acid or analogue thereof of formula I-3a with a P(III) forming reagent to form a nucleic acid or analogue thereof of formula I-5a, wherein each of B, E, L, LC, n, PG
3, R
1, R
2, R
3, X, X
1, X
2, X
3, E, and Z is as defined above and described herein. In step (b) above, PG
1 and PG
2 of a compound of formula Ia comprise silyl ethers or cyclic silylene derivatives that can be removed under acidic conditions or with fluoride anion. Examples of reagents providing fluoride anion for the removal of silicon-based protecting groups include hydrofluoric acid, hydrogen fluoride pyridine, triethylamine trihydrofluoride, tetra-N-butylammonium fluoride, and the like. In step (c) above, a compound of formula I-2a is protected with a suitable hydroxyl protecting group. In some embodiments, the protecting group PG
3 used for protection of the 5’-hydroxyl group of a compound of formula I-2a includes an acid labile protecting group such as trityl, 4-methyoxytrityl, 4,4’-dimethyoxytrityl, 4,4’,4’’-trimethyoxytrityl, 9-phenyl- xanthen-9-yl, 9-(p-tolyl)-xanthen-9-yl, pixyl, 2,7-dimethylpixyl, and the like. In some embodiments, the acid labile protecting group is suitable for deprotection during both solution- phase and solid-phase synthesis of acid-sensitive nucleic acids or analogues thereof using for example, dichloroacetic acid or trichloroacetic acid. In step (d) above, a compound of formula I-3a is treated with a P(III) forming reagent to afford a compound of formula I-5a. In the context of the present disclosure, a P(III) forming reagent is a phosphorus reagent that is reacted to for a phosphorus (III) compound. In some embodiments, the P(III) forming reagent is 2-cyanoethyl N,N- diisopropylchlorophosphoramidite or 2-cyanoethyl phosphorodichloridate. In some embodiments, the P(III) forming reagent is 2-cyanoethyl N,N- diisopropylchlorophosphoramidite. One of ordinary skill would recognize that the displacement of a leaving group in a P(III) forming reagent by -OH of a compound of formula I-3a is achieved either with or without the presence of a suitable base. Such suitable bases are well known in the art and include organic and inorganic bases. In some embodiments, the base is a tertiary amine such as triethylamine or diisopropylethylamine. In other embodiments, step (d) above is preformed using N,N-dimethylphosphoramic dichloride as a P(V) forming reagent. In some embodiments, the present disclosure provides a method for preparing an oligonucleotide comprising one or more conjugates, further comprising preparing a nucleic acid-conjugate or analogue thereof of formula Ia:
FH12501620.1
Attorney Docket: DCY-13025
or a salt thereof, comprising the steps of: (a) providing a nucleic acid or analogue thereof of formula I-1:
or salt thereof, and, (b) conjugating one or more lipophilic compounds to a nucleic acid or analogue thereof of formula I-1 to form a nucleic acid or analogue thereof of formula Ia comprising one or more lipid conjugates, wherein: each of B, E, L, LC, n, PG
1, PG
2, R1, R
2, X, and Z is as defined above and described herein. In step (b) above, a nucleic acid or analogue thereof of formula I-1a is conjugated with one or more lipophilic compounds to form a compound of formula Ia comprising one more lipid conjugates of the disclosure. Typically, conjugation is performed through an esterification or amidation reaction between a nucleic acid or analogue thereof of formula I-1a and one or more fatty acids in series or in parallel by known techniques in the art. In some embodiments, conjugation is performed under suitable amide forming conditions to afford a compound of formula I comprising one or more conjugates. Suitable amide forming conditions can include the use of an amide coupling reagent known in the art such as, but not limited to HATU, PyBOP, DCC, DIC, EDC, HBTU, HCTU, PyAOP, PyBrOP, BOP, BOP-Cl, DEPBT, T3P, TATU, TBTU, TNTU, TOTU, TPTU, TSTU, or TDBTU. In some embodiments, the present disclosure provides a method for preparing an oligonucleotide comprising one or more conjugates, said conjugate unit represent by formula II-1:
FH12501620.1
Attorney Docket: DCY-13025
II-1 or a pharmaceutically acceptable salt thereof, comprising the steps of: (a) providing an oligonucleotide of formula II-2:

II-2 or salt thereof, and, (b) conjugating one or more lipophilic compounds to an oligonucleotide of formula II-2 to form an oligonucleotide of formula II-1 comprising one or more conjugates. In step (b) above, an oligonucleotide of formula II-2 is conjugated with one or more lipophilic compounds to form an oligonucleotide of formula II-1 comprising one more conjugates of the disclosure. Typically, conjugation is performed through an esterification or amidation reaction between an oligonucleotide of formula II-2 and one or more fatty acids in series or in parallel by known techniques in the art. In some embodiments, conjugation is performed under suitable amide forming conditions to afford an oligonucleotide of formula II-1 comprising one more conjugates. Suitable amide forming conditions can include the use of an amide coupling reagent known in the art such as, but not limited to HATU, PyBOP, DCC, DIC, EDC, HBTU, HCTU, PyAOP, PyBrOP, BOP, BOP-Cl, DEPBT, T3P, TATU, TBTU, TNTU, TOTU, TPTU, TSTU, or TDBTU. In some embodiments, the present disclosure provides a method for preparing an oligonucleotide comprising a unit represent by formula II-2:

Attorney Docket: DCY-13025 or a pharmaceutically acceptable salt thereof, comprising the steps of: (a) providing a nucleic acid or analogue thereof of formula I-8:

I-8 or salt thereof, and (b) oligomerizing said compound of formula I-8 to form a compound of formula II-2. In step (b) above, oligomerizing refers to preforming oligomerization forming conditions using known and commonly applied processes to prepare oligonucleotides in the art. For example, the compound of formula I-8 is coupled to a solid supported nucleic acid or analogue thereof bearing a 5’-hydroxyl group. Further steps can comprise one or more deprotections, couplings, phosphite oxidation, and cleavage from the solid support to provide an oligonucleotide of various nucleotide lengths, represented by a compound of formula II-2. In some embodiments, the present disclosure provides a method for preparing a nucleic acid or analogue thereof comprising one or more conjugates, further comprising preparing a nucleic acid or analogue thereof of formula I-8:
I-8 or a salt thereof, comprising the steps of: (a) providing a nucleic acid or analogue thereof of formula I-1:
I-1 or salt thereof, (b) deprotecting said nucleic acid or analogue thereof of formula I-1 to form a compound of formula I-6:
FH12501620.1
Attorney Docket: DCY-13025
I-6 or salt thereof, (c) protecting said nucleic acid or analogue thereof of formula I-6 to form a compound of formula I-7:
I-7 or salt thereof, and (d) treating said nucleic acid or analogue thereof of formula I-7 with a P(III) forming reagent to form a nucleic acid or analogue thereof of formula I-8, In step (b) above, PG
1 and PG
2 of a compound of formula I-1 comprise silyl ethers or cyclic silylene derivatives that can be removed under acidic conditions or with fluoride anion. Examples of reagents providing fluoride anion for the removal of silicon-based protecting groups include hydrofluoric acid, hydrogen fluoride pyridine, triethylamine trihydrofluoride, tetra-N-butylammonium fluoride, and the like. In step (c) above, a compound of formula I-6 is protected with a suitable hydroxyl protecting group. In some embodiments, the protecting group PG
3 used for protection of the 5’-hydroxyl group of a compound of formula I-6 includes an acid labile protecting group such as trityl, 4-methyoxytrityl, 4,4’-dimethyoxytrityl, 4,4’,4’’-trimethyoxytrityl, 9-phenyl- xanthen-9-yl, 9-(p-tolyl)-xanthen-9-yl, pixyl, 2,7-dimethylpixyl, and the like. In some embodiments, the acid labile protecting group is suitable for deprotection during both solution- phase and solid-phase synthesis of acid-sensitive nucleic acids or analogues thereof using for example, dichloroacetic acid or trichloroacetic acid. In step (d) above, a compound of formula I-7 is treated with a P(III) forming reagent to afford a compound of formula I-8. In the context of the present disclosure, a P(III) forming reagent is a phosphorus reagent that is reacted to for a phosphorus (III) compound. In some embodiments, the P(III) forming reagent is 2-cyanoethyl N,N- diisopropylchlorophosphoramidite or 2-cyanoethyl phosphorodichloridate. In some
FH12501620.1
Attorney Docket: DCY-13025 embodiments, the P(III) forming reagent is 2-cyanoethyl N,N- diisopropylchlorophosphoramidite. One of ordinary skill would recognize that the displacement of a leaving group in a P(III) forming reagent by -OH of a compound of formula I-7 is achieved either with or without the presence of a suitable base. Such suitable bases are well known in the art and include organic and inorganic bases. In some embodiments, the base is a tertiary amine such as triethylamine or diisopropylethylamine. In other embodiments, step (d) above is preformed using N,N-dimethylphosphoramic dichloride as a P(V) forming reagent. In some embodiments, the present disclosure provides a method for preparing an oligonucleotide-ligand conjugate comprising one or more Y-COOH groups, said conjugate unit represented by formula II-b-3:
II-b-3 or a pharmaceutically acceptable salt thereof, comprising the steps of: (a) providing a nucleic acid-ligand conjugate or analogue thereof of formula C11:
or salt thereof, and (b) oligomerizing said compound of formula C11 to form a compound of formula II-b-3, In step (b) above, oligomerizing refers to preforming oligomerization forming conditions using known and commonly applied processes to prepare oligonucleotides in the art. For example, the compound of formula C11 is coupled to a solid supported nucleic acid or analogue thereof bearing a 5’-hydroxyl group. Further steps can comprise one or more deprotections, couplings, phosphite oxidation, and cleavage from the solid support to provide an oligonucleotide-ligand conjugate of various nucleotide lengths, with one or more nucleic acid-ligand conjugate units, wherein each unit is represented by a compound of formula II-b-3 comprising an adamantyl or lipid moiety of the disclosure.
FH12501620.1
Attorney Docket: DCY-13025 In some embodiments, the method for preparing an oligonucleotide of formula II-b-3 comprising one or more lipid conjugate, further comprises preparing a nucleic acid-ligand conjugate or analogue thereof of formula C11:
or a salt thereof, comprising the steps of: (a) providing a nucleic acid-ligand conjugate or analogue thereof of formula I-b:
I-b or salt thereof, (b) deprotecting said nucleic acid-ligand conjugate or analogue thereof of formula I-b to form a compound of formula C8:
C8 or salt thereof, (c) protecting said nucleic acid-ligand conjugate or analogue thereof of formula C8 to form a compound of formula C9:
or salt thereof, and (d) treating said nucleic acid-ligand conjugate or analogue thereof of formula C9 with a
FH12501620.1
Attorney Docket: DCY-13025 P(III) forming reagent to form a nucleic acid or analogue thereof of formula C11. In step (b) above, PG
1 and PG
2 of a compound of formula I-b comprise silyl ethers or cyclic silylene derivatives that can be removed under acidic conditions or with fluoride anion. Examples of reagents providing fluoride anion for the removal of silicon-based protecting groups include hydrofluoric acid, hydrogen fluoride pyridine, triethylamine trihydrofluoride, tetra-N- butylammonium fluoride, and the like. In step (c) above, a compound of formula C8 is protected with a suitable hydroxyl protecting group. In some embodiments, the protecting group PG
4 used for protection of the 5’-hydroxyl group of a compound of formula C8 includes an acid labile protecting group such as trityl, 4-methyoxytrityl, 4,4’-dimethyoxytrityl, 4,4’,4’’-trimethyoxytrityl, 9-phenyl- xanthen-9-yl, 9-(p-tolyl)-xanthen-9-yl, pixyl, 2,7-dimethylpixyl, and the like. In some embodiments, the acid labile protecting group is suitable for deprotection during both solution- phase and solid-phase synthesis of acid-sensitive nucleic acids or analogues thereof using for example, dichloroacetic acid or trichloroacetic acid. In step (d) above, a compound of formula C9 is treated with a P(III) forming reagent to afford a compound of formula C11. In the context of the present disclosure, a P(III) forming reagent is a phosphorus reagent that is reacted to for a phosphorus (III) compound. In some embodiments, the P(III) forming reagent is 2-cyanoethyl N,N- diisopropylchlorophosphoramidite or 2-cyanoethyl phosphorodichloridate. In some embodiments, the P(III) forming reagent is 2-cyanoethyl N,N- diisopropylchlorophosphoramidite. One of ordinary skill would recognize that the displacement of a leaving group in a P(III) forming reagent by -OH of a compound of formula C9 is achieved either with or without the presence of a suitable base. Such suitable bases are well known in the art and include organic and inorganic bases. In some embodiments, the base is a tertiary amine such as triethylamine or diisopropylethylamine. In other embodiments, step (d) above is preformed using N,N-dimethylphosphoramic dichloride as a P(V) forming reagent. In some embodiments, the present disclosure provides a method for preparing an oligonucleotide-ligand conjugate of formula II-b-3 comprising one or more nucleic acid-ligand conjugate units each comprising one or more adamantyl or lipid moieties, further comprising preparing a nucleic acid-ligand conjugate or analogue thereof of formula I-b:
FH12501620.1
Attorney Docket: DCY-13025
or a salt thereof, comprising the steps of: (a) providing a nucleic acid-ligand conjugate or analogue thereof of formula C6:

or salt thereof, and, (b) conjugating a lipophilic compound to a nucleic acid or analogue thereof of formula C6 to form a nucleic acid-ligand conjugate or analogue thereof of formula I-b comprising one or more adamantyl and/or lipid conjugates. In step (b) above, conjugation is performed under suitable amide forming conditions to afford a compound of formula I-b comprising an adamantyl and/or lipid conjugate. Suitable amide forming conditions can include the use of an amide coupling reagent known in the art such as, but not limited to HATU, PyBOP, DCC, DIC, EDC, HBTU, HCTU, PyAOP, PyBrOP, BOP, BOP-Cl, DEPBT, T3P, TATU, TBTU, TNTU, TOTU, TPTU, TSTU, or TDBTU. In some embodiments, the amide forming conditions comprise HATU and DIPEA or TEA. In some embodiments, a nucleic acid-ligand conjugate or analogue thereof of formula C6 is provided in salt form (e.g., a fumarate salt) and is first converted to the free base (e.g., using sodium bicarbonate) before preforming the conjugation step. In some embodiments, the present disclosure provides a method for preparing an oligonucleotide-ligand conjugate of formula II-b-3 comprising one or more nucleic acid-ligand conjugate units, further comprises preparing a nucleic acid-ligand conjugate or analogue thereof of formula C6:

Attorney Docket: DCY-13025 C6 or a salt thereof, comprising the steps of: (a) providing a nucleic acid or analogue thereof of formula C1:
C1 or salt thereof, and, (b) protecting said nucleic acid or analogue thereof of formula C1 to form a compound of formula C2:
C2 or salt thereof, (c) alkylating said nucleic acid or analogue thereof of formula C2 to form a compound of formula C3:
C3 or salt thereof, (d) substituting said nucleic acid or analogue thereof of formula C3 with a compound of formula C4:
C4 or salt thereof, to form a compound of formula C5:
FH12501620.1
Attorney Docket: DCY-13025 C5 or salt thereof, (e) deprotecting said nucleic acid or analogue thereof of formula C5 to form a nucleic acid- ligand conjugate or analogue thereof of formula C6. In step (b) above, PG
1 and PG
2 groups of formula C2 are taken together with their intervening atoms to form a cyclic diol protecting group, such as a cyclic acetal or ketal. Such groups include methylene, ethylidene, benzylidene, isopropylidene, cyclohexylidene, and cyclopentylidene, silylene derivatives such as di-t-butylsilylene and 1,1,3,3-tetraisopropylidisiloxanylidene, a cyclic carbonate, a cyclic boronate, and cyclic monophosphate derivatives based on cyclic adenosine monophosphate (i.e., cAMP). In some embodiments, the cyclic diol protection group is 1,1,3,3- tetraisopropylidisiloxanylidene prepared from the reaction of a diol of formula C1 and 1,3- dichloro-1,1,3,3-tetraisopropyldisiloxane under basic conditions. In step (c) above, a nucleic acid or analogue thereof of formula C2 is alkylated with a mixture of DMSO and acetic anhydride under acidic conditions. In some embodiments, when -V-H is a hydroxyl group, the mixture of DMSO and acetic anhydride in the presence of acetic acid forms (methylthio)methyl acetate in situ via the Pummerer rearrangement which then reacts with the hydroxyl group of the nucleic acid or analogue thereof of formula C2 to provide a monothioacetal functionalized fragment nucleic acid or analogue thereof of formula C3. In step (d) above, substitution of the thiomethyl group of a nucleic acid or analogue thereof of formula C3 using a nucleic acid or analogue thereof of formula C4 affords a nucleic acid or analogue thereof of formula C4. In some embodiments, substitution occurs under mild oxidizing and/or acidic conditions. In some embodiments, the mild oxidation reagent includes a mixture of elemental iodine and hydrogen peroxide, urea hydrogen peroxide complex, silver nitrate/silver sulfate, sodium bromate, ammonium peroxodisulfate, tetrabutylammonium peroxydisulfate, Oxone®, Chloramine T, Selectfluor®, Selectfluor® II, sodium hypochlorite, or potassium iodate/sodium periodiate. In some embodiments, the mild oxidizing agent includes N-iodosuccinimide, N-bromosuccinimide, N-chlorosuccinimide, 1,3-diiodo-5,5- dimethylhydantion, pyridinium tribromide, iodine monochloride or complexes thereof, etc. Acids that are typically used under mild oxidizing condition include sulfuric acid, p- toluenesulfonic acid, trifluoromethanesulfonic acid, methanesulfonic acid, and trifluoroacetic acid. In some embodiments, the mild oxidation reagent includes a mixture of N- iodosuccinimide and trifluoromethanesulfonic acid. In step (e) above, removal of PG
4 and optionally R
4 (when R
4 is a suitable amine protecting group) of a nucleic acid-ligand conjugate or analogue thereof of formula C5 affords
FH12501620.1
Attorney Docket: DCY-13025 a nucleic acid-ligand conjugate or analogue thereof of formula C6 or a salt thereof. In some embodiments, PG
4 and/or R
4 comprise carbamate derivatives that can be removed under acidic or basic conditions. In some embodiments, the protecting groups (e.g., both PG
4 and R
4 or either of PG
4 or R
4 independently) of a nucleic acid-ligand conjugate or analogue thereof of formula C5 are removed by acid hydrolysis. It will be appreciated that upon acid hydrolysis of the protecting groups of a nucleic acid-ligand conjugate or analogue thereof of formula C5, a salt of formula C6 thereof is formed. For example, when an acid-labile protecting group of a nucleic acid-ligand conjugate or analogue thereof of formula C5 is removed by treatment with an acid such as hydrochloric acid, then the resulting amine compound would be formed as its hydrochloride salt. One of ordinary skill in the art would recognize that a wide variety of acids are useful for removing amino protecting groups that are acid-labile and therefore a wide variety of salt forms of a nucleic acid or analogue thereof of formula C6 are contemplated. In other embodiments, the protecting groups (e.g., both PG
4 and R
4 or either of PG
4 or R
4 independently) of a nucleic acid or analogue thereof of formula C5 are removed by base hydrolysis. For example, Fmoc and trifluoroacetyl protecting groups can be removed by treatment with base. One of ordinary skill in the art would recognize that a wide variety of bases are useful for removing amino protecting groups that are base-labile. In some embodiments, a base is piperidine. In some embodiments, a base is 1,8- diazabicyclo[5.4.0]undec-7-ene (DBU). In some embodiments, a nucleic acid-ligand conjugate or analogue thereof of formula C5 is deprotected under basic conditions followed by treating with an acid to form a salt of formula C6. In some embodiments, the acid is fumaric acid the salt of formula C6 is the fumarate. In some embodiments, the present disclosure provides a method for preparing an oligonucleotide-ligand conjugate comprising one or more nucleic acid-ligand conjugate, said nucleic acid-ligand conjugate unit represented by formula II-b-3:

II-b-3 or a pharmaceutically acceptable salt thereof, comprising the steps of: (a) providing an oligonucleotide of formula D5:
FH12501620.1
Attorney Docket: DCY-13025
or salt thereof, and, (b) conjugating one or more adamantyl or lipophilic compounds to an oligonucleotide of formula D5 to form an oligonucleotide-ligand conjugate of formula II-b-3 comprising one or more nucleic acid-ligand conjugate units. In step (b) above, conjugation is performed under suitable amide forming conditions to afford a compound of formula D5 comprising an adamantyl or lipid conjugate. Suitable amide forming conditions can include the use of an amide coupling reagent known in the art such as, but not limited to HATU, PyBOP, DCC, DIC, EDC, HBTU, HCTU, PyAOP, PyBrOP, BOP, BOP-Cl, DEPBT, T3P, TATU, TBTU, TNTU, TOTU, TPTU, TSTU, or TDBTU. In some embodiments, the amide forming conditions comprise HATU and DIPEA or TEA. In some embodiments, the present disclosure provides a method for preparing an oligonucleotide-ligand conjugate comprising a unit represent by formula D5:

D5 or a salt thereof, comprising the steps of: (a) providing a nucleic acid-ligand conjugate or analogue thereof of formula D4:
D4 or salt thereof, and (b) deprotecting said compound of formula D4 to form a compound of formula D5. In step (b) above, removal of PG
4 and optionally R
4 (when R
4 is a suitable amine protecting group) of an oligonucleotide of formula D4 affords an oligonucleotide-ligand conjugate of formula D5
FH12501620.1
Attorney Docket: DCY-13025 or a salt thereof. In some embodiments, PG
4 and/or R
4 comprise carbamate derivatives that can be removed under acidic or basic conditions. In some embodiments, the protecting groups (e.g., both PG
4 and R
4 or either of PG
4 or R
4 independently) of an oligonucleotide-ligand conjugate of formula D4 are removed by acid hydrolysis. It will be appreciated that upon acid hydrolysis of the protecting groups of an oligonucleotide-ligand conjugate of formula D4, a salt of formula D5 thereof is formed. For example, when an acid-labile protecting group of an oligonucleotide of formula D4 is removed by treatment with an acid such as hydrochloric acid, then the resulting amine compound would be formed as its hydrochloride salt. One of ordinary skill in the art would recognize that a wide variety of acids are useful for removing amino protecting groups that are acid-labile and therefore a wide variety of salt forms of a nucleic acid-ligand conjugate unit or analogue thereof of formula D5 are contemplated. In other embodiments, the protecting groups (e.g., both PG
4 and R
4 or either of PG
4 or R
4 independently) of an oligonucleotide-ligand conjugate of formula D4 are removed by base hydrolysis. For example, Fmoc and trifluoroacetyl protecting groups can be removed by treatment with base. One of ordinary skill in the art would recognize that a wide variety of bases are useful for removing amino protecting groups that are base-labile. In some embodiments, a base is piperidine. In some embodiments, a base is 1,8- diazabicyclo[5.4.0]undec-7-ene (DBU). In some embodiments, the present disclosure provides a method for preparing an oligonucleotide-ligand conjugate comprising one or more nucleic acid-ligand conjugate unit with one or more adamantyl and/or lipid moiety, said conjugate unit represented by formula D4:
or a pharmaceutically acceptable salt thereof, comprising the steps of: (a) providing a nucleic acid or analogue thereof of formula D3:
FH12501620.1
Attorney Docket: DCY-13025 or salt thereof, and (b) oligomerizing said compound of formula D3 to form a compound of formula D4, In step (b) above, oligomerizing refers to preforming oligomerization forming conditions using known and commonly applied processes to prepare oligonucleotides in the art. For example, the nucleic acid or analogue thereof of formula D3 is coupled to a solid supported nucleic acid or analogue thereof bearing a 5’-hydroxyl group. Further steps can comprise one or more deprotections, couplings, phosphite oxidation, and cleavage from the solid support to provide an oligonucleotide of various nucleotide lengths, represented by a compound of formula D4 comprising an adamantyl or lipid conjugate of the disclosure. In some embodiments, the present disclosure provides a method for preparing a nucleic acid or analogue thereof comprising one or more lipid conjugate, further comprising preparing a nucleic acid or analogue thereof of formula D3:

D3 or a salt thereof, comprising the steps of: (a) providing a nucleic acid or analogue thereof of formula C5:
C5 or salt thereof, (b) deprotecting said nucleic acid or analogue thereof of formula C5 to form a compound of formula D1:
D1 or salt thereof, (c) protecting said nucleic acid or analogue thereof of formula D1 to form a nucleic acid or analogue thereof of formula D2:
FH12501620.1
Attorney Docket: DCY-13025
D2 or salt thereof, and (d) treating said nucleic acid or analogue thereof of formula D2 with a P(III) forming reagent to form a nucleic acid or analogue thereof of formula D3. In step (b) above, PG
1 and PG
2 of a nucleic acid or analogue thereof of formula C5 comprise silyl ethers or cyclic silylene derivatives that can be removed under acidic conditions or with fluoride anion. Examples of reagents providing fluoride anion for the removal of silicon-based protecting groups include hydrofluoric acid, hydrogen fluoride pyridine, triethylamine trihydrofluoride, tetra-N- butylammonium fluoride, and the like. In step (c) above, a nucleic acid or analogue thereof of formula D1 is protected with a suitable hydroxyl protecting group. In some embodiments, the protecting group PG
3 used for protection of the 5’-hydroxyl group of a compound of formula D1 includes an acid labile protecting group such as trityl, 4-methyoxytrityl, 4,4’-dimethyoxytrityl, 4,4’,4’’- trimethyoxytrityl, 9-phenyl-xanthen-9-yl, 9-(p-tolyl)-xanthen-9-yl, pixyl, 2,7-dimethylpixyl, and the like. In some embodiments, the acid labile protecting group is suitable for deprotection during both solution-phase and solid-phase synthesis of acid-sensitive nucleic acids or analogues thereof using for example, dichloroacetic acid or trichloroacetic acid. In step (d) above, a nucleic acid or analogue thereof of formula D2 is treated with a P(III) forming reagent to afford a compound of formula D3. In the context of the present disclosure, a P(III) forming reagent is a phosphorus reagent that is reacted to for a phosphorus (III) compound. In some embodiments, the P(III) forming reagent is 2-cyanoethyl N,N- diisopropylchlorophosphoramidite or 2-cyanoethyl phosphorodichloridate. In some embodiments, the P(III) forming reagent is 2-cyanoethyl N,N- diisopropylchlorophosphoramidite. One of ordinary skill would recognize that the displacement of a leaving group in a P(III) forming reagent by -OH of a compound of formula D2 is achieved either with or without the presence of a suitable base. Such suitable bases are well known in the art and include organic and inorganic bases. In some embodiments, the base is a tertiary amine such as triethylamine or diisopropylethylamine. In other embodiments, step (d) above is preformed using N,N-dimethylphosphoramic dichloride as a P(V) forming reagent.
FH12501620.1
Attorney Docket: DCY-13025 Formulations Various formulations have been developed to facilitate oligonucleotide use. For example, oligonucleotides (e.g., RNAi oligonucleotides) can be delivered to a subject or a cellular environment using a formulation that minimizes degradation, facilitates delivery and/or uptake, or provides another beneficial property to the oligonucleotides in the formulation. In some embodiments, provided herein are compositions comprising oligonucleotides (e.g., RNAi oligonucleotide) reduce the expression of a target mRNA (e.g., a target mRNA expressed in adipose tissue). Such compositions can be suitably formulated such that when administered to a subject, either into the immediate environment of a target cell or systemically, a sufficient portion of the oligonucleotides enter the cell to reduce target gene expression. Any variety of suitable oligonucleotide formulations can be used to deliver oligonucleotides for the reduction of target gene expression as disclosed herein. In some embodiments, an oligonucleotide is formulated in buffer solutions such as phosphate buffered saline solutions, liposomes, micellar structures, and capsids. In some embodiments, the formulations herein comprise an excipient. In some embodiments, an excipient confers to a composition improved stability, improved absorption, improved solubility and/or therapeutic enhancement of the active ingredient. In some embodiments, an excipient is a buffering agent (e.g., sodium citrate, sodium phosphate, a tris base, or sodium hydroxide) or a vehicle (e.g., a buffered solution, petrolatum, dimethyl sulfoxide, or mineral oil). In some embodiments, an oligonucleotide is lyophilized for extending its shelf-life and then made into a solution before use (e.g., administration to a subject). Accordingly, an excipient in a composition comprising any one of the oligonucleotides described herein may be a lyoprotectant (e.g., mannitol, lactose, polyethylene glycol or polyvinylpyrrolidone) or a collapse temperature modifier (e.g., dextran, Ficoll™ or gelatin). Likewise, the oligonucleotides herein may be provided in the form of their free acids. In some embodiments, a pharmaceutical composition is formulated to be compatible with its intended route of administration. Examples of routes of administration include parenteral (e.g., intravenous, intramuscular, intraperitoneal, intradermal, subcutaneous, intrathecal), oral (e.g., inhalation), transdermal (e.g., topical), transmucosal and rectal administration. Pharmaceutical compositions suitable for injectable use include sterile aqueous solutions (where water soluble) or dispersions and sterile powders for the extemporaneous preparation of sterile injectable solutions or dispersions. For intravenous administration, suitable carriers include physiological saline, bacteriostatic water, Cremophor EL™ (BASF,
FH12501620.1
Attorney Docket: DCY-13025 Parsippany, N.J.) or phosphate buffered saline (PBS). The carrier can be a solvent or dispersion medium containing, for example, water, ethanol, polyol (e.g., glycerol, propylene glycol, and liquid polyethylene glycol, and the like), and suitable mixtures thereof. In many cases, it will be preferable to include isotonic agents, for example, sugars, polyalcohols such as mannitol, sorbitol, sodium chloride in the composition. Sterile injectable solutions can be prepared by incorporating the oligonucleotides in a required amount in a selected solvent with one or a combination of ingredients enumerated above, as required, followed by filtered sterilization. In some embodiments, a composition may contain at least about 0.1% of the therapeutic agent (e.g., an lipid-conjugated RNAi oligonucleotide herein) or more, although the percentage of the active ingredient(s) may be between about 1% to about 80% or more of the weight or volume of the total composition. Factors such as solubility, bioavailability, biological half- life, route of administration, product shelf life, as well as other pharmacological considerations will be contemplated by one skilled in the art of preparing such pharmaceutical formulations, and as such, a variety of dosages and treatment regimens may be desirable. Structural Modifications As nucleic acids are polymers of subunits or compounds, many of the modifications described below occur at a position which is repeated within a nucleic acid (e.g., a modification of a base, or a phosphate moiety, or the non-bridging oxygen of a phosphate moiety). In some cases the modification will occur at all of the subject positions in the nucleic acid but in many, and in fact in most cases it will not. By way of example, a modification may only occur at a 3' or 5' terminal position, may only occur in the internal unpaired region, may only occur in a terminal regions, e.g. at a position on a terminal nucleotide or in the last 2, 3, 4, 5, or 10 nucleotides of a strand. In some embodiments, a modification occurs at all of the subject positions in the nucleic acid. A modification may occur in a double strand region, a single strand region, or in both. A modification may occur only in the double strand region of an RNA agent or may only occur in a single strand region of an RNA agent. (e.g., a phosphorothioate modification at a non-bridging oxygen position may only occur at one or both termini, may only occur in a terminal regions or at a position on a terminal nucleotide or in the last 2, 3, 4, 5, or 10 nucleotides of a strand, or may occur in double strand and single strand regions, particularly at termini. The 5' end or ends can be phosphorylated. Many studies in the art have indicated that modified oligonucleotides and oligonucleotide analogs may be less readily internalized than their natural counterparts. As a
FH12501620.1
Attorney Docket: DCY-13025 result, the activity of many previously available RNAi trigger molecules has not been sufficient for practical therapeutic, research or diagnostic purposes. Modifications to enhance the effectiveness of the RNAi trigger molecule oligonucleotides and overcome these problems have taken many forms. These modifications include base ring modifications, sugar moiety modifications, and sugar-phosphate backbone modifications, many exemplified herein and used in the current disclosure. Prior sugar- phosphate backbone modifications, particularly on the phosphorus atom, have affected various levels of resistance to nucleases. However, while the ability of an RNAi trigger molecule oligonucleotide to load into the RISC and direct the location of relevant mRNA sequences is fundamental to RNAi trigger molecule methodology, many modifications work at cross purposes with each other to optimize the behavior of the RNAi trigger. It is this balancing act which must be taken into account relative to the development of superior and effective RNAi molecules. Another key factor is the stereochemical effect that arises in oligomers having P-chiral centers. In general, an oligomer with a length of n nucleosides will constitute a mixture of chirality in successive non-stereospecific chain synthesis. It has been observed that Rp and Sp homochiral chains, whose absolute configuration at all internucleotide methane phosphonate phosphorus atoms are either Rp or Sp, and non-stereoregular chains show different physicochemical properties as well as different capabilities of forming adducts with oligonucleotides of complementary sequence. In addition, phosphorothioate analogs of nucleotides have shown substantial stereoselectivity differences between Oligo-Rp and Oligo- Sp oligonucleotides in resistance to nucleases activity (Potter, B
IOCHEMISTRY, 22:1369, (1983); Bryant et al., B
IOCHEMISTRY, 18:2825, (1979)). Lesnikowski (N
UCL. A
CIDS R
ES., 18:2109, (1990)) observed that diastereomerically pure octathymidine methylphosphonates, in which six out of seven methylphosphonate bonds have defined configuration at the phosphorus atom when complexed with the matrix showed substantial differences in melting temperatures. According to the current disclosure chirally pure nucleotide analogs, or portions thereof, are expected to provide trigger structures with improved characteristics allowing the development of more potent and longer lasting RNAi triggers. In some embodiments of the current disclosure it is particularly preferred to enhance stability, to include particular bases in overhangs, or to include modified nucleotides or nucleotide surrogates, in single strand overhangs, e.g., in a 5' or 3' overhang, or in both. Likewise, it can be desirable to include purine nucleotides in overhangs as they are more resistant to nuclease activity. In some embodiments all or some of the bases in a 3' or 5'
FH12501620.1
Attorney Docket: DCY-13025 overhang will be modified, with a modification described herein. Modifications can include the use of modifications at the 2' OH group of the ribose sugar, deoxythymidine, instead of ribonucleotides, and modifications in the phosphate group, that is, phosphothioate modifications. Overhangs need not be homologous with the target sequence. Methods of Use Reducing Target Gene Expression In some embodiments, the disclosure provides methods for contacting or delivering to a cell or population of cells an effective amount of any of the RNAi oligonucleotides herein to reduce expression of a target gene. In some embodiments, expression of a target gene is reduced in extrahepatic tissue. In some embodiments, expression of a target gene is reduced in cardiac tissue. In some embodiments, expression of a target gene is reduced in the liver. In some embodiments, expression of a target gene is reduced in adipose tissue. In some embodiments, expression of a target gene is reduced in adrenal tissue. In some embodiments, expression of a target gene is reduced in skeletal muscle tissue. In some embodiments, expression of a target gene is reduced in the central nervous system. In some embodiments, a reduction of target gene expression is determined by measuring a reduction in the amount or level of target mRNA, protein encoded by the target mRNA, or target gene (mRNA or protein) activity in a cell. The methods include those described herein and known to one of ordinary skill in the art. Methods provided herein are useful in any appropriate cell type. In some embodiments, a cell is any cell that expresses the target mRNA. In some embodiments, the cell is a primary cell obtained from a subject. In some embodiments, the primary cell has undergone a limited number of passages such that the cell substantially maintains is natural phenotypic properties. In some embodiments, a cell to which the oligonucleotide is delivered is ex vivo or in vitro (i.e., can be delivered to a cell in culture or to an organism in which the cell resides). In some embodiments, the RNAi oligonucleotides disclosed herein are delivered to a cell or population of cells using a nucleic acid delivery method known in the art including, but not limited to, injection of a solution or pharmaceutical composition containing the RNAi oligonucleotide, bombardment by particles covered by the RNAi oligonucleotide, exposing the cell or population of cells to a solution containing the RNAi oligonucleotide, or electroporation of cell membranes in the presence of the RNAi oligonucleotide. Other methods known in the art for delivering oligonucleotides to cells may be used, such as lipid-mediated carrier transport,
FH12501620.1
Attorney Docket: DCY-13025 chemical-mediated transport, and cationic liposome transfection such as calcium phosphate, and others. In some embodiments, reduction of target gene expression is determined by an assay or technique that evaluates one or more molecules, properties or characteristics of a cell or population of cells associated with target gene expression, or by an assay or technique that evaluates molecules that are directly indicative of target gene expression in a cell or population of cells (e.g., target mRNA or protein). In some embodiments, the extent to which an RNAi oligonucleotide provided herein reduces target gene expression in a cell is evaluated by comparing target gene expression in a cell or population of cells contacted with the RNAi oligonucleotide to a control cell or population of cells (e.g., a cell or population of cells not contacted with the RNAi oligonucleotide or contacted with a control RNAi oligonucleotide). In some embodiments, a control amount or level of target gene expression in a control cell or population of cells is predetermined, such that the control amount or level need not be measured in every instance the assay or technique is performed. The predetermined level or value can take a variety of forms. In some embodiments, a predetermined level or value can be single cut-off value, such as a median or mean. In some embodiments, contacting or delivering an RNAi oligonucleotide described herein to a cell or a population of cells results in a reduction in expression of a target gene. In some embodiments, the reduction in target gene expression is relative to a control amount or level of target gene expression in cell or population of cells not contacted with the RNAi oligonucleotide or contacted with a control RNAi oligonucleotide. In some embodiments, the reduction in target gene expression is about 1% or lower, about 5% or lower, about 10% or lower, about 15% or lower, about 20% or lower, about 25% or lower, about 30% or lower, about 35% or lower, about 40% or lower, about 45% or lower, about 50% or lower, about 55% or lower, about 60% or lower, about 70% or lower, about 80% or lower, or about 90% or lower relative to a control amount or level of target gene expression. In some embodiments, the control amount or level of target gene expression is an amount or level of target mRNA and/or protein in a cell or population of cells that has not been contacted with an RNAi oligonucleotide herein. In some embodiments, the effect of delivery of an RNAi oligonucleotide to a cell or population of cells according to a method herein is assessed after any finite period or amount of time (e.g., minutes, hours, days, weeks, months). For example, in some embodiments, target gene expression is determined in a cell or population of cells at least about 4 hours, about 8 hours, about 12 hours, about 18 hours, about 24 hours; or at least about 1 day, about 2 days, about 3 days, about 4 days, about 5 days, about 6 days, about 7 days, about 8 days, about 9
FH12501620.1
Attorney Docket: DCY-13025 days, about 10 days, about 11 days, about 12 days, about 13 days, about 14 days, about 21 days, about 28 days, about 35 days, about 42 days, about 49 days, about 56 days, about 63 days, about 70 days, about 77 days, or about 84 days or more after contacting or delivering the RNAi oligonucleotide to the cell or population of cells. In some embodiments, target gene expression is determined in a cell or population of cells at least about 1 month, about 2 months, about 3 months, about 4 months, about 5 months, or about 6 months or more after contacting or delivering the RNAi oligonucleotide to the cell or population of cells. Reducing Target Gene Expression in Extrahepatic Tissue In some embodiments, expression of a target gene is reduced in extrahepatic tissue. In some embodiments, expression of a target gene is reduced in a cell of an extrahepatic tissue. In some embodiments, expression of a target gene is reduced in at least one extrahepatic tissue. In some embodiments, expression of a target gene is reduced in one or more extrahepatic tissues. In some embodiments, extrahepatic tissue is one or more of adipose tissue, adrenal gland, cardiac tissue, or skeletal muscle. In some embodiments, expression of a target gene is reduced in at least one extrahepatic tissue, selected from adipose tissue, adrenal gland, cardiac tissue, skeletal muscle, the central nervous system, and any combination thereof. In some embodiments, expression of a target gene in the extrahepatic tissue of a subject is reduced by at least about 30%, about 35%, about 40%, about 45%, about 50%, about 55%, about 60%, about 65%, about 70%, about 75%, about 80%, about 85%, about 90%, about 95%, about 99% or greater than 99% when compared to target gene expression in a control tissue. In some embodiments, expression of a target gene in the extrahepatic tissue of a subject is reduced by at least about 30%, about 35%, about 40%, about 45%, about 50%, about 55%, about 60%, about 65%, about 70%, about 75%, about 80%, about 85%, about 90%, about 95%, about 99% or greater than 99% when compared to target gene expression in liver tissue. In some embodiments, contacting or delivering an RNAi oligonucleotide described herein to a cell or a population of cells results in a reduction in expression of a target gene in extrahepatic tissue. In some embodiments, the reduction in expression of a target gene in extrahepatic tissue is relative to an amount or level of target gene expression in liver tissue contacted with the RNAi oligonucleotide. In some embodiments, reduction in expression of a target gene in the extrahepatic tissue of a subject is increased by at least 1%, at least 5%, at least 10%, at least 15%, at least 20%, at least 25%, at least 30%, at least 30%, at least 35%, at
FH12501620.1
Attorney Docket: DCY-13025 least 40%, at least 45%, at least 50%, at least 55%, at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, or at least 95% relative to reduction in expression of the target gene in liver tissue. In some embodiments, reduction in expression of a target gene in liver tissue is about 1% or lower, about 5% or lower, about 10% or lower, about 15% or lower, about 20% or lower, about 25% or lower, about 30% or lower, about 35% or lower, about 40% or lower, about 45% or lower, about 50% or lower, about 55% or lower, about 60% or lower, about 70% or lower, about 80% or lower, or about 90% or lower relative to the reduction in expression of the target gene in extrahepatic tissue. In some embodiments, differences in target mRNA expression between cell types or tissue types is measured using methods known in the art. In some embodiments, differences in target mRNA expression between cell types or tissue types measures the reduction of the target mRNA in a first cell/tissue type compared to the reduction of target mRNA in a second cell/tissue type. For example, differences in target mRNA expression between cell types or tissue types is measured using polymerase chain reaction methods (e.g., RT-PCR) comparing relative expression between different tissue or cell types. In some embodiments, differences in target mRNA expression between cell types or tissue types is measured using Northern blot analysis, in situ hybridization, RT-PCR, RNA sequencing, or other methods known in the art. In some embodiments, a relative amount of target mRNA expression is compared between cell or tissue types. In some embodiments, an absolute amount of target mRNA expression is compared between cell or tissue types. Reducing Target Gene Expression in Cardiac Tissue In some embodiments, expression of a target gene is reduced in a region of cardiac tissue. In some embodiments, expression of a target gene in the cardiac tissue of a subject is reduced by at least about 30%, about 35%, about 40%, about 45%, about 50%, about 55%, about 60%, about 65%, about 70%, about 75%, about 80%, about 85%, about 90%, about 95%, about 99% or greater than 99% when compared to target gene expression in other control tissue. In some embodiments, expression of a target gene in the cardiac tissue of a subject is reduced by at least about 30%, about 35%, about 40%, about 45%, about 50%, about 55%, about 60%, about 65%, about 70%, about 75%, about 80%, about 85%, about 90%, about 95%, about 99% or greater than 99% when compared to target gene expression in liver tissue.
FH12501620.1
Attorney Docket: DCY-13025 In some embodiments, contacting or delivering an RNAi oligonucleotide described herein to a cell or a population of cells results in a reduction in expression of a target gene in cardiac tissue. In some embodiments, the reduction in expression of a target gene in cardiac tissue is relative to an amount or level of target gene expression in liver tissue contacted with the RNAi oligonucleotide. In some embodiments, reduction in expression of a target gene in the cardiac tissue of a subject is increased by at least 1%, at least 5%, at least 10%, at least 15%, at least 20%, at least 25%, at least 30%, at least 30%, at least 35%, at least 40%, at least 45%, at least 50%, at least 55%, at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, or at least 95% relative to reduction in expression of the target gene in liver tissue. In some embodiments, reduction in expression of a target gene in liver tissue is about 1% or lower, about 5% or lower, about 10% or lower, about 15% or lower, about 20% or lower, about 25% or lower, about 30% or lower, about 35% or lower, about 40% or lower, about 45% or lower, about 50% or lower, about 55% or lower, about 60% or lower, about 70% or lower, about 80% or lower, or about 90% or lower relative to the reduction in expression of the target gene in cardiac tissue. Reducing Target Gene Expression in Skeletal Muscle In some embodiments, expression of a target gene is reduced in skeletal muscle. In some embodiments, expression of a target gene in the skeletal muscle of a subject is reduced by at least about 30%, about 35%, about 40%, about 45%, about 50%, about 55%, about 60%, about 65%, about 70%, about 75%, about 80%, about 85%, about 90%, about 95%, about 99% or greater than 99% when compared to target gene expression in other control tissue. In some embodiments, expression of a target gene in the skeletal muscle of a subject is reduced by at least about 30%, about 35%, about 40%, about 45%, about 50%, about 55%, about 60%, about 65%, about 70%, about 75%, about 80%, about 85%, about 90%, about 95%, about 99% or greater than 99% when compared to target gene expression in liver tissue. In some embodiments, contacting or delivering an RNAi oligonucleotide described herein to a cell or a population of cells results in a reduction in expression of a target gene in skeletal muscle. In some embodiments, the reduction in expression of a target gene in skeletal muscle is relative to an amount or level of target gene expression in liver tissue contacted with the RNAi oligonucleotide. In some embodiments, reduction in expression of a target gene in the skeletal muscle of a subject is increased by at least 1%, at least 5%, at least 10%, at least 15%, at least 20%, at least 25%, at least 30%, at least 30%, at least 35%, at least 40%,
FH12501620.1
Attorney Docket: DCY-13025 at least 45%, at least 50%, at least 55%, at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, or at least 95% relative to reduction in expression of the target gene in liver tissue. In some embodiments, reduction in expression of a target gene in liver tissue is about 1% or lower, about 5% or lower, about 10% or lower, about 15% or lower, about 20% or lower, about 25% or lower, about 30% or lower, about 35% or lower, about 40% or lower, about 45% or lower, about 50% or lower, about 55% or lower, about 60% or lower, about 70% or lower, about 80% or lower, or about 90% or lower relative to the reduction in expression of the target gene in skeletal muscle. Reducing Target Gene Expression in Adrenal Gland In some embodiments, expression of a target gene is reduced in a region of the adrenal gland. In some embodiments, expression of a target gene in the adrenal gland of a subject is reduced by at least about 30%, about 35%, about 40%, about 45%, about 50%, about 55%, about 60%, about 65%, about 70%, about 75%, about 80%, about 85%, about 90%, about 95%, about 99% or greater than 99% when compared to target gene expression in other control tissue. In some embodiments, expression of a target gene in the adrenal gland of a subject is reduced by at least about 30%, about 35%, about 40%, about 45%, about 50%, about 55%, about 60%, about 65%, about 70%, about 75%, about 80%, about 85%, about 90%, about 95%, about 99% or greater than 99% when compared to target gene expression in liver tissue. In some embodiments, contacting or delivering an RNAi oligonucleotide described herein to a cell or a population of cells results in a reduction in expression of a target gene in adrenal gland. In some embodiments, the reduction in expression of a target gene in adrenal gland is relative to an amount or level of target gene expression in liver tissue contacted with the RNAi oligonucleotide. In some embodiments, reduction in expression of a target gene in the adrenal gland of a subject is increased by at least 1%, at least 5%, at least 10%, at least 15%, at least 20%, at least 25%, at least 30%, at least 30%, at least 35%, at least 40%, at least 45%, at least 50%, at least 55%, at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, or at least 95% relative to reduction in expression of the target gene in liver tissue. In some embodiments, reduction in expression of a target gene in liver tissue is about 1% or lower, about 5% or lower, about 10% or lower, about 15% or lower, about 20% or lower, about 25% or lower, about 30% or lower, about 35% or lower, about 40% or lower, about 45% or lower, about 50% or lower, about 55% or lower, about
FH12501620.1
Attorney Docket: DCY-13025 60% or lower, about 70% or lower, about 80% or lower, or about 90% or lower relative to the reduction in expression of the target gene in adrenal gland. Reducing Target Gene Expression in Adipose Tissue In some embodiments, expression of a target gene is reduced in adipose tissue. In some embodiments, expression of a target gene in adipose tissue of a subject is reduced by at least about 30%, about 35%, about 40%, about 45%, about 50%, about 55%, about 60%, about 65%, about 70%, about 75%, about 80%, about 85%, about 90%, about 95%, about 99% or greater than 99% when compared to target gene expression in control tissue. In some embodiments, expression of a target gene in gonadal white adipose tissue of a subject is reduced by at least about 30%, about 35%, about 40%, about 45%, about 50%, about 55%, about 60%, about 65%, about 70%, about 75%, about 80%, about 85%, about 90%, about 95%, about 99% or greater than 99% when compared to target gene expression in control tissue. In some embodiments, expression of a target gene in subcutaneous white adipose tissue of a subject is reduced by at least about 30%, about 35%, about 40%, about 45%, about 50%, about 55%, about 60%, about 65%, about 70%, about 75%, about 80%, about 85%, about 90%, about 95%, about 99% or greater than 99% when compared to target gene expression in liver tissue. In some embodiments, expression of a target gene in adipose tissue of a subject is reduced by at least about 30%, about 35%, about 40%, about 45%, about 50%, about 55%, about 60%, about 65%, about 70%, about 75%, about 80%, about 85%, about 90%, about 95%, about 99% or greater than 99% when compared to target gene expression in control tissue. In some embodiments, expression of a target gene in gonadal white adipose tissue of a subject is reduced by at least about 30%, about 35%, about 40%, about 45%, about 50%, about 55%, about 60%, about 65%, about 70%, about 75%, about 80%, about 85%, about 90%, about 95%, about 99% or greater than 99% when compared to target gene expression in liver tissue. In some embodiments, expression of a target gene in subcutaneous white adipose tissue of a subject is reduced by at least about 30%, about 35%, about 40%, about 45%, about 50%, about 55%, about 60%, about 65%, about 70%, about 75%, about 80%, about 85%,
FH12501620.1
Attorney Docket: DCY-13025 about 90%, about 95%, about 99% or greater than 99% when compared to target gene expression in liver tissue. In some embodiments, contacting or delivering an RNAi oligonucleotide described herein to a cell or a population of cells results in a reduction in expression of a target gene in adipose tissue (e.g., gWAT and/or scWAT). In some embodiments, the reduction in expression of a target gene in adipose tissue (e.g., gWAT and/or scWAT) is relative to an amount or level of target gene expression in liver tissue contacted with the RNAi oligonucleotide. In some embodiments, reduction in expression of a target gene in the adipose tissue (e.g., gWAT and/or scWAT) of a subject is increased by at least 1%, at least 5%, at least 10%, at least 15%, at least 20%, at least 25%, at least 30%, at least 30%, at least 35%, at least 40%, at least 45%, at least 50%, at least 55%, at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, or at least 95% relative to reduction in expression of the target gene in liver tissue. In some embodiments, reduction in expression of a target gene in liver tissue is about 1% or lower, about 5% or lower, about 10% or lower, about 15% or lower, about 20% or lower, about 25% or lower, about 30% or lower, about 35% or lower, about 40% or lower, about 45% or lower, about 50% or lower, about 55% or lower, about 60% or lower, about 70% or lower, about 80% or lower, or about 90% or lower relative to the reduction in expression of the target gene in adipose tissue (e.g., gWAT and/or scWAT). Reducing Target Gene Expression in Central Nervous Tissue In some embodiments, expression of a target gene is reduced in a region of the central nervous system. In some embodiments, expression of a target gene in the central nervous system of a subject is reduced by at least about 30%, about 35%, about 40%, about 45%, about 50%, about 55%, about 60%, about 65%, about 70%, about 75%, about 80%, about 85%, about 90%, about 95%, about 99% or greater than 99% when compared to target gene expression in other control tissue. In some embodiments, expression of a target gene in the central nervous system of a subject is reduced by at least about 30%, about 35%, about 40%, about 45%, about 50%, about 55%, about 60%, about 65%, about 70%, about 75%, about 80%, about 85%, about 90%, about 95%, about 99% or greater than 99% when compared to target gene expression in liver tissue.
FH12501620.1
Attorney Docket: DCY-13025 In some embodiments, contacting or delivering an RNAi oligonucleotide described herein to a cell or a population of cells results in a reduction in expression of a target gene in the central nervous system. In some embodiments, the reduction in expression of a target gene in the central nervous system is relative to an amount or level of target gene expression in liver tissue contacted with the RNAi oligonucleotide. In some embodiments, reduction in expression of a target gene in the central nervous system of a subject is increased by at least 1%, at least 5%, at least 10%, at least 15%, at least 20%, at least 25%, at least 30%, at least 30%, at least 35%, at least 40%, at least 45%, at least 50%, at least 55%, at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, or at least 95% relative to reduction in expression of the target gene in liver tissue. In some embodiments, reduction in expression of a target gene in liver tissue is about 1% or lower, about 5% or lower, about 10% or lower, about 15% or lower, about 20% or lower, about 25% or lower, about 30% or lower, about 35% or lower, about 40% or lower, about 45% or lower, about 50% or lower, about 55% or lower, about 60% or lower, about 70% or lower, about 80% or lower, or about 90% or lower relative to the reduction in expression of the target gene in the central nervous system. Treatment Methods In some embodiments, the disclosure provides methods for treating a disease, disorder, or condition associated with expression of a target gene in extrahepatic tissue. In some embodiments, the disclosure provides methods for treating a disease, disorder, or condition associated with expression of a target gene in cardiac tissue. In some embodiments, the disclosure provides methods for treating a disease, disorder, or condition associated with expression of a target gene in adipose tissue. In some embodiments, the disclosure provides methods for treating a disease, disorder, or condition associated with expression of a target gene in adrenal tissue. In some embodiments, the disclosure provides methods for treating a disease, disorder, or condition associated with expression of a target gene in skeletal muscle tissue. In some embodiments, the disclosure provides methods for treating a disease, disorder, or condition associated with expression of a target gene in the central nervous system. Methods described herein are typically involve administering to a subject a therapeutically effective amount of an RNAi oligonucleotide herein, that is, an amount capable of producing a desirable therapeutic result. A therapeutically acceptable amount may be an amount that can therapeutically treat a disease or disorder. The appropriate dosage for any one subject will depend on certain factors, including the subject^s size, body surface area, age, the
FH12501620.1
Attorney Docket: DCY-13025 composition to be administered, the active ingredient(s) in the composition, time and route of administration, general health, and other drugs being administered concurrently. In some embodiments, a subject is administered any one of the compositions herein either enterally (e.g., orally, by gastric feeding tube, by duodenal feeding tube, via gastrostomy or rectally), parenterally (e.g., subcutaneous injection, intravenous injection or infusion, intra- arterial injection or infusion, intraosseous infusion, intramuscular injection, intracerebral injection, intracerebroventricular injection, intrathecal), topically (e.g., epicutaneous, inhalational, via eye drops, or through a mucous membrane), or by direct injection into a target organ (e.g., the brain of a subject). In some embodiments, an RNAi oligonucleotide herein, or a composition thereof, is administered once every year, once every 6 months, once every 4 months, quarterly (once every three months), bi-monthly (once every two months), monthly or weekly. In some embodiments, an RNAi oligonucleotide herein, or a composition thereof, is administered every week or at intervals of two, or three weeks. In some embodiments, an RNAi oligonucleotide herein, or a composition thereof, is administered daily. In some embodiments, a subject is administered one or more loading doses of an RNAi oligonucleotide herein, or a composition thereof, followed by one or more maintenance doses of the RNAi oligonucleotide, or a composition thereof. In some embodiments, the subject to be treated is a human or non-human primate or other mammalian subject. Other exemplary subjects include domesticated animals such as dogs and cats; livestock such as horses, cattle, pigs, sheep, goats, and chickens; and animals such as mice, rats, guinea pigs, and hamsters. Treatment Methods in the Extrahepatic Tissue The disclosure provides oligonucleotides for use as a medicament, in particular for use in a method for the treatment of diseases, disorders, and conditions associated with extrahepatic tissue. The disclosure also provides RNAi oligonucleotides for use, or adaptable for use, to treat a subject (e.g., a human) having a disease, disorder or condition associated with expression of a target gene that would benefit from reducing expression of the target gene. In some embodiments, the disclosure provides RNAi oligonucleotides for use, or adapted for use, to treat a subject having a disease, disorder or condition associated with expression of a target gene in extrahepatic tissue. The disclosure also provides RNAi oligonucleotides for use, or adaptable for use, in the manufacture of a medicament or pharmaceutical composition for treating a disease, disorder or condition associated with expression of a target gene in
FH12501620.1
Attorney Docket: DCY-13025 extrahepatic tissue. In some embodiments, the RNAi oligonucleotides for use, or adaptable for use, target mRNA and reduce expression of a target gene in extrahepatic tissue (e.g., via the RNAi pathway). In some embodiments, the RNAi oligonucleotides for use, or adaptable for use, target mRNA and reduce the amount or level of target mRNA, protein and/or activity. In addition, in some embodiments of the methods herein, a subject having a disease, disorder or condition associated with expression of a target gene in extrahepatic tissue or is predisposed to the same is selected for treatment with an RNAi oligonucleotide herein. In some embodiments, the method comprises selecting an individual having a marker (e.g., a biomarker) for a disease, disorder or condition associated with expression of a target gene in extrahepatic tissue, or predisposed to the same, such as, but not limited to, target mRNA, protein, or a combination thereof. Likewise, and as detailed below, some embodiments of the methods provided by the disclosure include steps such as measuring or obtaining a baseline value for a marker of expression of a target gene in extrahepatic tissue, and then comparing such obtained value to one or more other baseline values or values obtained after the subject is administered the RNAi oligonucleotide to assess the effectiveness of treatment. The disclosure also provides methods of treating a subject having, suspected of having, or at risk of developing a disease, disorder or condition associated with expression of a target gene in extrahepatic tissue with an RNAi oligonucleotide provided herein. In some embodiments, the disclosure provides methods of treating or attenuating the onset or progression of a disease, disorder or condition associated with expression of a target gene in extrahepatic tissue using the RNAi oligonucleotides provided herein. In some embodiments, the disclosure provides methods to achieve one or more therapeutic benefits in a subject having a disease, disorder or condition associated with expression of a target gene in extrahepatic tissue using the RNAi oligonucleotides provided herein. In some embodiments of the methods herein, the subject is treated by administering a therapeutically effective amount of any one or more of the RNAi oligonucleotides provided herein. In some embodiments, treatment comprises reducing expression of a target gene in extrahepatic tissue. In some embodiments, the subject is treated therapeutically. In some embodiments, the subject is treated prophylactically. In some embodiments of the methods herein, an RNAi oligonucleotide provided herein, or a pharmaceutical composition comprising the RNAi oligonucleotide, is administered to a subject having a disease, disorder or condition associated with expression of a target gene in extrahepatic tissue such that target gene expression is reduced in the subject, thereby treating
FH12501620.1
Attorney Docket: DCY-13025 the subject. In some embodiments, an amount or level of target mRNA is reduced in the subject. In some embodiments, an amount or level of protein encoded by the target mRNA is reduced in the subject. In some embodiments of the methods herein, an RNAi oligonucleotide provided herein, or a pharmaceutical composition comprising the RNAi oligonucleotide, is administered to a subject having a disease, disorder or condition associated with expression of a target gene in extrahepatic tissue such that target gene expression is reduced in the subject by at least about 30%, about 35%, about 40%, about 45%, about 50%, about 55%, about 60%, about 65%, about 70%, about 75%, about 80%, about 85%, about 90%, about 95%, about 99% or greater than 99% when compared to target gene expression prior to administration of the RNAi oligonucleotide or pharmaceutical composition. In some embodiments, expression of a target gene in extrahepatic tissue is reduced in the subject by at least about 30%, about 35%, about 40%, about 45%, about 50%, about 55%, about 60%, about 65%, about 70%, about 75%, about 80%, about 85%, about 90%, about 95%, about 99% or greater than 99% when compared to target gene expression in a subject (e.g., a reference or control subject) not receiving RNAi oligonucleotide or pharmaceutical composition or receiving a control oligonucleotide, pharmaceutical composition or treatment. In some embodiments of the methods herein, an RNAi oligonucleotide herein, or a pharmaceutical composition comprising the RNAi oligonucleotide, is administered to a subject having a disease, disorder or condition associated with expression of a target gene in extrahepatic tissue such that an amount or level of target mRNA is reduced in the subject by at least about 30%, about 35%, about 40%, about 45%, about 50%, about 55%, about 60%, about 65%, about 70%, about 75%, about 80%, about 85%, about 90%, about 95%, about 99% or greater than 99% when compared to the amount or level of target mRNA prior to administration of the RNAi oligonucleotide or pharmaceutical composition. In some embodiments, an amount or level of target mRNA is reduced in the subject by at least about 30%, about 35%, about 40%, about 45%, about 50%, about 55%, about 60%, about 65%, about 70%, about 75%, about 80%, about 85%, about 90%, about 95%, about 99% or greater than 99% when compared to an amount or level of target mRNA in a subject (e.g., a reference or control subject) not receiving the RNAi oligonucleotide or pharmaceutical composition or receiving a control oligonucleotide, pharmaceutical composition or treatment. In some embodiments of the methods herein, an RNAi oligonucleotide herein, or a pharmaceutical composition comprising the RNAi oligonucleotide, is administered to a subject having a disease, disorder or condition associated with expression of a target gene in
FH12501620.1
Attorney Docket: DCY-13025 extrahepatic tissue such that an amount or level of protein encoded by the target gene is reduced in the subject by at least about 30%, about 35%, about 40%, about 45%, about 50%, about 55%, about 60%, about 65%, about 70%, about 75%, about 80%, about 85%, about 90%, about 95%, about 99% or greater than 99% when compared to the amount or level of protein encoded by the target gene prior to administration of the RNAi oligonucleotide or pharmaceutical composition. In some embodiments, an amount or level of protein encoded by a target gene in extrahepatic tissue is reduced in the subject by at least about 30%, about 35%, about 40%, about 45%, about 50%, about 55%, about 60%, about 65%, about 70%, about 75%, about 80%, about 85%, about 90%, about 95%, about 99% or greater than 99% when compared to an amount or level of protein encoded by the target gene in a subject (e.g., a reference or control subject) not receiving the RNAi oligonucleotide or pharmaceutical composition or receiving a control oligonucleotide, pharmaceutical composition or treatment. In some embodiments of the methods herein, an RNAi oligonucleotide herein, or a pharmaceutical composition comprising the RNAi oligonucleotide, is administered to a subject having a disease, disorder or condition associated with expression of a target gene in extrahepatic tissue such that an amount or level of target gene activity is reduced in the subject by at least about 30%, about 35%, about 40%, about 45%, about 50%, about 55%, about 60%, about 65%, about 70%, about 75%, about 80%, about 85%, about 90%, about 95%, about 99% or greater than 99% when compared to the amount or level of target gene activity prior to administration of the RNAi oligonucleotide or pharmaceutical composition. In some embodiments, an amount or level of target gene activity is reduced in the subject by at least about 30%, about 35%, about 40%, about 45%, about 50%, about 55%, about 60%, about 65%, about 70%, about 75%, about 80%, about 85%, about 90%, about 95%, about 99% or greater than 99% when compared to an amount or level of target gene activity in a subject (e.g., a reference or control subject) not receiving the RNAi oligonucleotide or pharmaceutical composition or receiving a control oligonucleotide, pharmaceutical composition or treatment. In some embodiments, an RNAi oligonucleotide comprising a C16-COOH conjugated to a nucleotide on the sense strand reduces target mRNA expression by at least 10%, at least 20%, at least 30%, at least 40%, at least 50%, at least 60%, at least 70%, or at least 80% more in adipose tissue relative to target gene knockdown in liver tissue. In some embodiments, an RNAi oligonucleotide comprising a C16-COOH conjugated to a nucleotide at position 1 of the sense strand reduces target mRNA expression by at least 10%, at least 20%, at least 30%, at least 40%, at least 50%, at least 60%, at least 70%, or at least 80% more in adipose tissue relative to target gene knockdown in liver tissue. In some embodiments, an RNAi
FH12501620.1
Attorney Docket: DCY-13025 oligonucleotide comprising a C16-COOH conjugated to a nucleotide at position 28 of the sense strand reduces target mRNA expression by at least 10%, at least 20%, at least 30%, at least 40%, at least 50%, at least 60%, at least 70%, or at least 80% more in adipose tissue relative to target gene knockdown in liver tissue. In some embodiments, an RNAi oligonucleotide comprising a C16-COOH conjugated to a nucleotide in the tetraloop of the sense strand reduces target mRNA expression by at least 10%, at least 20%, at least 30%, at least 40%, at least 50%, at least 60%, at least 70%, or at least 80% more in adipose tissue relative to target gene knockdown in liver tissue. In some embodiments, an RNAi oligonucleotide comprising a C16-COOH conjugated to a nucleotide on the sense strand reduces target mRNA expression by at least 10%, at least 20%, at least 30%, at least 40%, at least 50%, at least 60%, at least 70%, or at least 80% more in adipose tissue relative to target gene knockdown in skeletal muscle tissue. In some embodiments, an RNAi oligonucleotide comprising a C16-COOH conjugated to a nucleotide at position 1 of the sense strand reduces target mRNA expression by at least 10%, at least 20%, at least 30%, at least 40%, at least 50%, at least 60%, at least 70%, or at least 80% more in adipose tissue relative to target gene knockdown in skeletal muscle tissue. In some embodiments, an RNAi oligonucleotide comprising a PEG4-triz-C16-COOH conjugated to a nucleotide on the sense strand reduces target mRNA expression by at least 10%, at least 20%, at least 30%, at least 40%, at least 50%, at least 60%, at least 70%, or at least 80% more in adipose tissue relative to target gene knockdown in liver tissue. In some embodiments, an RNAi oligonucleotide comprising a PEG4-triz-C16-COOH conjugated to a nucleotide at position 1 of the sense strand reduces target mRNA expression by at least 10%, at least 20%, at least 30%, at least 40%, at least 50%, at least 60%, at least 70%, or at least 80% more in adipose tissue relative to target gene knockdown in liver tissue. In some embodiments, an RNAi oligonucleotide comprising a C2-triazole-C16-COOH conjugated to a nucleotide on the sense strand reduces target mRNA expression by at least 10%, at least 20%, at least 30%, at least 40%, at least 50%, at least 60%, at least 70%, or at least 80% more in adipose tissue relative to target gene knockdown in liver tissue. In some embodiments, an RNAi oligonucleotide comprising a C2-triazole-C16-COOH conjugated to a nucleotide at position 1 of the sense strand reduces target mRNA expression by at least 10%, at least 20%, at least 30%, at least 40%, at least 50%, at least 60%, at least 70%, or at least 80% more in adipose tissue relative to target gene knockdown in liver tissue. In some embodiments, an RNAi oligonucleotide comprising a C22-COOH conjugated to a nucleotide on the sense strand reduces target mRNA expression by at least 10%, at least 20%, at least 30%, at least
FH12501620.1
Attorney Docket: DCY-13025 40%, at least 50%, at least 60%, at least 70%, or at least 80% more in adipose tissue relative to target gene knockdown in liver tissue. In some embodiments, an RNAi oligonucleotide comprising a C22-COOH conjugated to a nucleotide at position 1 of the sense strand reduces target mRNA expression by at least 10%, at least 20%, at least 30%, at least 40%, at least 50%, at least 60%, at least 70%, or at least 80% more in adipose tissue relative to target gene knockdown in liver tissue. In some embodiments, an RNAi oligonucleotide comprising a PEG4-C16-COOH conjugated to a nucleotide on the sense strand reduces target mRNA expression by at least 10%, at least 20%, at least 30%, at least 40%, at least 50%, at least 60%, at least 70%, or at least 80% more in adipose tissue relative to target gene knockdown in liver tissue. In some embodiments, an RNAi oligonucleotide comprising a PEG4-C16-COOH conjugated to a nucleotide at position 1 of the sense strand reduces target mRNA expression by at least 10%, at least 20%, at least 30%, at least 40%, at least 50%, at least 60%, at least 70%, or at least 80% more in adipose tissue relative to target gene knockdown in liver tissue. In some embodiments, an RNAi oligonucleotide comprising a PEG
12-C16-COOH conjugated to a nucleotide on the sense strand reduces target mRNA expression by at least 10%, at least 20%, at least 30%, at least 40%, at least 50%, at least 60%, at least 70%, or at least 80% more in adipose tissue relative to target gene knockdown in liver tissue. In some embodiments, an RNAi oligonucleotide comprising a PEG
12-C16-COOH conjugated to a nucleotide at position 1 of the sense strand reduces target mRNA expression by at least 10%, at least 20%, at least 30%, at least 40%, at least 50%, at least 60%, at least 70%, or at least 80% more in adipose tissue relative to target gene knockdown in liver tissue. In some embodiments, an RNAi oligonucleotide comprising a Hexylamine-PEG4-C16-COOH conjugated to a nucleotide on the sense strand reduces target mRNA expression by at least 10%, at least 20%, at least 30%, at least 40%, at least 50%, at least 60%, at least 70%, or at least 80% more in adipose tissue relative to target gene knockdown in liver tissue. In some embodiments, an RNAi oligonucleotide comprising a Hexylamine-PEG
4-C16-COOH conjugated to a nucleotide at position 1 of the sense strand reduces target mRNA expression by at least 10%, at least 20%, at least 30%, at least 40%, at least 50%, at least 60%, at least 70%, or at least 80% more in adipose tissue relative to target gene knockdown in liver tissue. In some embodiments, an RNAi oligonucleotide comprising a PEG
2-C16-COOH conjugated to a nucleotide on the sense strand reduces target mRNA expression by at least 10%, at least 20%, at least 30%, at least 40%, at least 50%, at least 60%, at least 70%, or at least 80% more in adipose tissue relative to target gene knockdown in liver tissue. In some embodiments, an RNAi oligonucleotide comprising a PEG2-C16-COOH conjugated to a
FH12501620.1
Attorney Docket: DCY-13025 nucleotide on the sense strand reduces target mRNA expression by at least 10%, at least 20%, at least 30%, at least 40%, at least 50%, at least 60%, at least 70%, or at least 80% more in skeletal muscle tissue relative to target gene knockdown in liver tissue. In some embodiments, an RNAi oligonucleotide comprising a PEG
2-C16-COOH conjugated to a nucleotide on the sense strand reduces target mRNA expression by at least 10%, at least 20%, at least 30%, at least 40%, at least 50%, at least 60%, at least 70%, or at least 80% more in heart tissue relative to target gene knockdown in liver tissue. In some embodiments, an RNAi oligonucleotide comprising a PEG2-C16-COOH conjugated to a nucleotide on the sense strand at position 1 and position 20 reduces target mRNA expression by at least 10%, at least 20%, at least 30%, at least 40%, at least 50%, at least 60%, at least 70%, or at least 80% more in adipose tissue relative to target gene knockdown in liver tissue. In some embodiments, an RNAi oligonucleotide comprising a PEG2-C16-COOH conjugated to a nucleotide on the sense strand at position 1 and position 20 reduces target mRNA expression by at least 10%, at least 20%, at least 30%, at least 40%, at least 50%, at least 60%, at least 70%, or at least 80% more in skeletal muscle tissue relative to target gene knockdown in liver tissue. In some embodiments, an RNAi oligonucleotide comprising a PEG
2-C16-COOH conjugated to a nucleotide on the sense strand at position 1 and position 20 reduces target mRNA expression by at least 10%, at least 20%, at least 30%, at least 40%, at least 50%, at least 60%, at least 70%, or at least 80% more in heart tissue relative to target gene knockdown in liver tissue. Suitable methods for determining target gene expression, an amount or level of target mRNA, an amount or level of protein encoded by the target gene, and/or an amount or level of target gene activity, in the subject, or in a sample from the subject, are known in the art. Further, the Examples set forth herein illustrate exemplary methods for determining target gene expression. In some embodiments, target gene expression, an amount or level of target gene mRNA, an amount or level of protein encoded by a target gene, an amount or level of target gene activity, or any combination thereof, is reduced in a cell, a population or a group of cells (e.g., an organoid), an organ, blood or a fraction thereof (e.g., plasma), a tissue, a sample (e.g., a biopsy sample), or any other biological material obtained or isolated from the subject. In some embodiments, expression of a target gene in extrahepatic tissue, an amount or level of target gene mRNA, an amount or level of protein encoded by the target gene, an amount or level of target gene activity, or any combination thereof, is reduced in more than one type of cell, more than one groups of cells, more than one organ, more than one fraction of blood (e.g., plasma
FH12501620.1
Attorney Docket: DCY-13025 and one or more other blood fraction(s)), more than one type of tissue, more than one type of sample obtained or isolated from the subject. In some embodiments, expression of a target gene in extrahepatic tissue is reduced in one or more of skeletal muscle, cardiac tissue, adipose tissue, and adrenal tissue. In some embodiments, expression of a target gene in extrahepatic tissue is reduced in the skeletal muscle. In some embodiments, expression of a target gene in extrahepatic tissue is reduced in the cardiac tissue. In some embodiments, expression of a target gene in extrahepatic tissue is reduced in adipose tissue. In some embodiments, expression of a target gene in extrahepatic tissue is reduced in adrenal tissue. Examples of a disease, disorder or condition associated with expression of a target gene in extrahepatic tissue include, but are not limited to, myopathy, amyotrophic lateral sclerosis, spinal muscular atrophy, multiple sclerosis, muscular dystrophy, lipedema, lipodystrophy, lymphedema, lipomatosis, familial multiple lipomatosis, angiolipomatosis, Dercum disease, multiple symmetric lipomatosis, Proteus syndrome, Cowden Syndrom, Modeling disease, lymphatic leakage, de novo adipogenesis, obesity, and satiety. In some embodiments, the target gene in extrahepatic tissue may be a target gene from any mammal, such as a human. Any target gene in extrahepatic tissue may be silenced according to the method described herein. Kits In some embodiments, the disclosure provides a kit comprising an RNAi oligonucleotide herein, or a composition thereof, described herein, and instructions for use. In some embodiments, the kit comprises an RNAi oligonucleotide herein, or a composition thereof, described herein, and a package insert containing instructions for use of the kit and/or any component thereof. In some embodiments, the kit comprises, in a suitable container, a RNAi oligonucleotide herein, or a composition thereof, described herein, one or more controls, and various buffers, reagents, enzymes and other standard ingredients well known in the art. In some embodiments, the container comprises at least one vial, well, test tube, flask, bottle, syringe, or other container means, into which the RNAi oligonucleotide herein, or a composition thereof, is placed, and in some instances, suitably aliquoted. In some embodiments where an additional component is provided, the kit contains additional containers into which this component is placed. The kits can also include a means for containing a RNAi oligonucleotide herein, or a composition thereof, and any other reagent in close confinement for commercial sale. Such containers may include injection or blow-molded plastic containers
FH12501620.1
Attorney Docket: DCY-13025 into which the desired vials are retained. Containers and/or kits can include labeling with instructions for use and/or warnings. In some embodiments, a kit comprises an RNAi oligonucleotide herein, or a composition thereof, described herein, and a pharmaceutically acceptable carrier, or a pharmaceutical composition comprising the RNAi oligonucleotide and instructions for treating or delaying progression of a disease, disorder or condition associated with expression of a target gene expressed extrahepatic tissue in a subject in need thereof. In some embodiments, a kit comprises an RNAi oligonucleotide herein, or a composition thereof, described herein, and a pharmaceutically acceptable carrier, or a pharmaceutical composition comprising the RNAi oligonucleotide and instructions for treating or delaying progression of a disease, disorder or condition associated with expression of a target gene expressed in adipose tissue in a subject in need thereof. In some embodiments, a kit comprises an RNAi oligonucleotide herein, or a composition thereof, described herein, and a pharmaceutically acceptable carrier, or a pharmaceutical composition comprising the RNAi oligonucleotide and instructions for treating or delaying progression of a disease, disorder or condition associated with expression of a target gene expressed in the adrenal gland in a subject in need thereof. In some embodiments, a kit comprises an RNAi oligonucleotide herein, or a composition thereof, described herein, and a pharmaceutically acceptable carrier, or a pharmaceutical composition comprising the RNAi oligonucleotide and instructions for treating or delaying progression of a disease, disorder or condition associated with expression of a target gene expressed in the cardiac tissue in a subject in need thereof. In some embodiments, a kit comprises an RNAi oligonucleotide herein, or a composition thereof, described herein, and a pharmaceutically acceptable carrier, or a pharmaceutical composition comprising the RNAi oligonucleotide and instructions for treating or delaying progression of a disease, disorder or condition associated with expression of a target gene expressed in the skeletal muscle in a subject in need thereof. Definitions As used herein, the term "and/or" includes any and all combinations of one or more of the associated listed items. Further, the singular forms and the articles "a", "an" and "the" are intended to include the plural forms as well, unless expressly stated otherwise. It will be further understood that the terms: includes, comprises, including and/or comprising, when used in this specification, specify the presence of stated features, integers, steps, operations, elements, and/or components, but do not preclude the presence or addition of one or more other features, integers, steps, operations, elements, components, and/or groups thereof. Further, it will be
FH12501620.1
Attorney Docket: DCY-13025 understood that when an element, including component or subsystem, is referred to and/or shown as being connected or coupled to another element, it can be directly connected or coupled to the other element or intervening elements may be present. Unless defined otherwise, all technical and scientific terms used herein have the same meaning as commonly understood to one of ordinary skill in the art to which this disclosure belongs. Although methods and materials similar or equivalent to those described herein can be used in the practice of the disclosed methods and compositions, exemplary methods, and materials are described herein. General texts which describe molecular biological techniques useful herein, including the use of vectors, promoters and many other relevant topics, include Berger and Kimmel, G
UIDE TO M
OLECULAR C
LONING T
ECHNIQUES, METHODS IN ENZYMOLOGY, volume 152, (Academic Press, Inc., San Diego, Calif.) ("Berger"); Sambrook et al., MOLECULAR CLONING--A LABORATORY MANUAL, 2d ed., Vol.1-3, Cold Spring Harbor Laboratory, Cold Spring Harbor, 1989 ("Sambrook") and CURRENT PROTOCOLS IN MOLECULAR BIOLOGY, F.M. Ausubel et al., eds., CURRENT PROTOCOLS, A JOINT VENTURE BETWEEN GREENE PUBLISHING ASSOCIATES, INC. AND JOHN WILEY AND SONS, INC., (supplemented through 1999) ("Ausubel"). Examples of protocols sufficient to direct persons of skill through in vitro amplification methods, including the polymerase chain reaction (PCR), the ligase chain reaction(LCR), Q.beta.-replicase amplification and other RNA polymerase mediated techniques (e.g., NASBA), e.g., for the production of the homologous nucleic acids of the disclosure are found in Berger, Sambrook, and Ausubel, as well as in Mullis et al., (1987) U.S. Pat. No. 4,683,202; Innis et al., eds. (1990); PCR P
ROTOCOLS: A G
UIDE TO M
ETHODS AND A
PPLICATIONS (Academic Press Inc. San Diego, Calif.) ("Innis"); Arnheim and Levinson (Oct. 1, 1990) CandEN 36-47; J.NIH R
ES. (1991) 3:81-94; Kwoh et al., (1989) P
ROC. N
ATL. A
CAD. SCI. USA 86: 1173; Guatelliet al (1990) PROC. NAT'L. ACAD. SCI. USA 87: 1874; Lomell et al., (1989) J. CLIN. CHEM 35: 1826; Landegren et al., (1988) SCIENCE 241: 1077-80; Van Brunt (1990) BIOTECHNOLOGY 8: 291-94; Wu and Wallace (1989) GENE 4:560; Barringer et al., (1990) GENE 89:117; and, Sooknanan and Malek (1995) BIOTECHNOLOGY 13: 563-564. Improved methods for cloning in vitro amplified nucleic acids are described in Wallace et al., U.S. Pat. No.5,426,039. Improved methods for amplifying large nucleic acids by PCR are summarized in Cheng et al., (1994) NATURE 369: 684-85 and the references cited therein, in which PCR amplicons of up to 40 kb are generated. As used in the specification and the appended claims, the singular forms "a," "an" and "the" include plural referents unless the context clearly dictates otherwise. Thus, for example,
FH12501620.1
Attorney Docket: DCY-13025 reference to "a pharmaceutical carrier" includes mixtures of two or more such carriers, and the like. Ranges can be expressed herein as from "about" one value, and/or to "about" another value. When such a range is expressed, another embodiment includes from the one value and/or to the other value. Similarly, when values are expressed as approximations, by use of the antecedent "about," it will be understood that the value forms another embodiment. It will be further understood that the endpoints of each of the ranges are significant both in relation to the other endpoint, and independently of the other endpoint. It is also understood that there are several values disclosed herein, and that each value is also herein disclosed as "about" that value in addition to the value itself. For example, if the value "10" is disclosed, then "about 10" is also disclosed. It is also understood that when a value is disclosed that "less than or equal to" the value, "greater than or equal to the value" and possible ranges between values are also disclosed, as appropriately understood by the skilled artisan. For example, if the value "10" is disclosed the "less than or equal to 10" as well as "greater than or equal to 10" is also disclosed. It is also understood that the throughout the application, data is provided in several different formats, and that this data, represents endpoints and starting points, and ranges for any combination of the data points. For example, if a particular datapoint "10" and a particular data point 15 are disclosed, it is understood that greater than, greater than or equal to, less than, less than or equal to, and equal to 10 and 15 are considered disclosed as well as between 10 and 15. It is also understood that each unit between two particular units are also disclosed. For example, if 10 and 15 are disclosed, then 11, 12, 13, and 14 are also disclosed. As used here, the term “amount” refers to an absolute amount (e.g., an absolute amount of mRNA or protein), a relative amount (e.g., a relative amount of target mRNA as measured by PCR assay or protein), or a concentration (e.g. a concentration of ligand-conjugated oligonucleotide in a composition), whether the amount referred to in a given instance refers to an absolute amount, concentration, or both, will be clear to the skilled artisan based on the context provided herein. As used herein, “bicyclic nucleotide” refers to a nucleotide comprising a bicyclic sugar moiety. As used herein “bicyclic sugar moiety” refers to a modified sugar moiety comprising a 4 to 7 membered ring (including but not limited to a furanosyl) comprising a bridge connecting two atoms of the 4 to 7 membered ring to form a second ring, resulting in a bicyclic structure. Typically, the 4 to 7 membered ring is a sugar. In some embodiments, the 4-to-7-member ring
FH12501620.1
Attorney Docket: DCY-13025 is a furanosyl. In some embodiments, the bridge connects the 2^-carbon and the 4^-carbon of the furanosyl. As used herein, “complementary” refers to a structural relationship between two nucleotides (e.g., on two opposing nucleic acids or on opposing regions of a single nucleic acid strand) that permits the two nucleotides to form base pairs with one another. For example, a purine nucleotide of one nucleic acid that is complementary to a pyrimidine nucleotide of an opposing nucleic acid may base pair together by forming hydrogen bonds with one another. In some embodiments, complementary nucleotides can base pair in the Watson-Crick manner or in any other manner that allows for the formation of stable duplexes. In some embodiments, two nucleic acids may have regions of multiple nucleotides that are complementary with each other to form regions of complementarity, as described herein. As used herein, “deoxyribonucleotide” refers to a nucleotide having a hydrogen in place of a hydroxyl at the 2^ position of its pentose sugar when compared with a ribonucleotide. A modified deoxyribonucleotide is a deoxyribonucleotide having one or more modifications or substitutions of atoms other than at the 2^ position, including modifications or substitutions in or of the sugar, phosphate group or base. As used herein, “double-stranded RNA” or “dsRNA” refers to an RNA oligonucleotide that is substantially in a duplex form. In some embodiments, the complementary base-pairing of duplex region(s) of a dsRNA oligonucleotide is formed between antiparallel sequences of nucleotides of covalently separate nucleic acid strands. In some embodiments, complementary base-pairing of duplex region(s) of a dsRNA formed between antiparallel sequences of nucleotides of nucleic acid strands that are covalently linked. In some embodiments, complementary base-pairing of duplex region(s) of a dsRNA is formed from single nucleic acid strand that is folded (e.g., via a hairpin) to provide complementary antiparallel sequences of nucleotides that base pair together. In some embodiments, a dsRNA comprises two covalently separate nucleic acid strands that are fully duplexed with one another. However, in some embodiments, a dsRNA comprises two covalently separate nucleic acid strands that are partially duplexed (e.g., having overhangs at one or both ends). In some embodiments, a dsRNA comprises antiparallel sequence of nucleotides that are partially complementary, and thus, may have one or more mismatches, which may include internal mismatches or end mismatches. As used herein, “duplex,” in reference to nucleic acids (e.g., oligonucleotides), refers to a structure formed through complementary base pairing of two antiparallel sequences of nucleotides.
FH12501620.1
Attorney Docket: DCY-13025 As used herein, “excipient” refers to a non-therapeutic agent that may be included in a composition, for example, to provide or contribute to a desired consistency or stabilizing effect. As used herein, “loop” refers to an unpaired region of a nucleic acid (e.g., oligonucleotide) that is flanked by two antiparallel regions of the nucleic acid that are sufficiently complementary to one another, such that under appropriate hybridization conditions (e.g., in a phosphate buffer, in a cells), the two antiparallel regions, which flank the unpaired region, hybridize to form a duplex (referred to as a “stem”). As used herein, “melting temperature” or “Tm” means the temperature at which the two strands of a duplex nucleic acid separate. Tm is often used as a measure of duplex stability or the binding affinity of two strands of complementary nucleic acids or portions thereof. Tm can be measured by using the UV spectrum to determine the formation and breakdown (melting) of hybridization. Base stacking, which occurs during hybridization, is accompanied by a reduction in UV absorption (hypochromicity). Consequently, a reduction in UV absorption indicates a higher T
m. As used herein, “modified internucleotide linkage” refers to an internucleotide linkage having one or more chemical modifications when compared with a reference internucleotide linkage comprising a phosphodiester bond. In some embodiments, a modified nucleotide is a non-naturally occurring linkage. Typically, a modified internucleotide linkage confers one or more desirable properties to a nucleic acid in which the modified internucleotide linkage is present. For example, a modified nucleotide may improve thermal stability, resistance to degradation, nuclease resistance, solubility, bioavailability, bioactivity, reduced immunogenicity, etc. As used herein, “modified nucleotide” refers to a nucleotide having one or more chemical modifications when compared with a corresponding reference nucleotide selected from: adenine ribonucleotide, guanine ribonucleotide, cytosine ribonucleotide, uracil ribonucleotide, adenine deoxyribonucleotide, guanine deoxyribonucleotide, cytosine deoxyribonucleotide and thymidine deoxyribonucleotide. In some embodiments, a modified nucleotide is a non-naturally occurring nucleotide. In some embodiments, a modified nucleotide has one or more chemical modification in its sugar, nucleobase and/or phosphate group. In some embodiments, a modified nucleotide has one or more chemical moieties conjugated to a corresponding reference nucleotide. Typically, a modified nucleotide confers one or more desirable properties to a nucleic acid in which the modified nucleotide is present. For example, a modified nucleotide may improve thermal stability, resistance to degradation, nuclease resistance, solubility, bioavailability, bioactivity, reduced immunogenicity, etc.
FH12501620.1
Attorney Docket: DCY-13025 As used herein, “nicked tetraloop structure” refers to a structure of a RNAi oligonucleotide that is characterized by separate sense (passenger) and antisense (guide) strands, in which the sense strand has a region of complementarity with the antisense strand, and in which at least one of the strands, generally the sense strand, has a tetraloop configured to stabilize an adjacent stem region formed within the at least one strand. As used herein, “oligonucleotide” refers to a short nucleic acid (e.g., less than about 100 nucleotides in length). An oligonucleotide may be single stranded (ss) or ds. An oligonucleotide may or may not have duplex regions. As a set of non-limiting examples, an oligonucleotide may be, but is not limited to, a small interfering RNA (siRNA), microRNA (miRNA), short hairpin RNA (shRNA), dicer substrate interfering RNA (dsiRNA), antisense oligonucleotide, short siRNA or ss siRNA. In some embodiments, a double-stranded (dsRNA) is an RNAi oligonucleotide. As used herein, “overhang” refers to terminal non-base pairing nucleotide(s) resulting from one strand or region extending beyond the terminus of a complementary strand with which the one strand or region forms a duplex. In some embodiments, an overhang comprises one or more unpaired nucleotides extending from a duplex region at the 5^ terminus or 3^ terminus of a dsRNA. In some embodiments, the overhang is a 3^ or 5^ overhang on the antisense strand or sense strand of a dsRNA. As used herein, “phosphate analog” refers to a chemical moiety that mimics the electrostatic and/or steric properties of a phosphate group. In some embodiments, a phosphate analog is positioned at the 5^ terminal nucleotide of an oligonucleotide in place of a 5^- phosphate, which is often susceptible to enzymatic removal. In some embodiments, a 5^ phosphate analog contains a phosphatase-resistant linkage. Examples of phosphate analogs include, but are not limited to, 5^ phosphonates, such as 5^ methylene phosphonate (5^-MP) and 5^-(E)-vinylphosphonate (5^-VP). In some embodiments, an oligonucleotide has a phosphate analog at a 4^-carbon position of the sugar (referred to as a “4^-phosphate analog”) at a 5^- terminal nucleotide. An example of a 4^-phosphate analog is oxymethyl phosphonate, in which the oxygen atom of the oxymethyl group is bound to the sugar moiety (e.g., at its 4^-carbon) or analog thereof. See, e.g., US Provisional Patent Application Nos. 62/383,207 (filed on 2 September 2016) and 62/393,401 (filed on 12 September 2016). Other modifications have been developed for the 5^ end of oligonucleotides (see, e.g., Intl. Patent Application No. WO 2011/133871; US Patent No. 8,927,513; and Prakash et al., (2015) NUCLEIC ACIDS RES. 43:2993-3011).
FH12501620.1
Attorney Docket: DCY-13025 As used herein, “reduced expression” of a target gene refers to a decrease in the amount or level of RNA transcript (e.g., target mRNA) or protein encoded by the target gene and/or a decrease in the amount or level of activity of the gene in a cell, a population of cells, a sample, or a subject, when compared to an appropriate reference (e.g., a reference cell, population of cells, sample, or subject). For example, the act of contacting a cell with an oligonucleotide or conjugate herein (e.g., an RNAi oligonucleotide comprising an antisense strand having a nucleotide sequence that is complementary to a nucleotide sequence comprising a target mRNA) may result in a decrease in the amount or level of target mRNA, protein encoded by a target gene, and/or target gene activity (e.g., via inactivation and/or degradation of target mRNA by the RNAi pathway) when compared to a cell that is not treated with the double- stranded oligonucleotide. Similarly, and as used herein, “reducing expression” refers to an act that results in reduced expression of a target gene. As used herein, “region of complementarity” refers to a sequence of nucleotides of a nucleic acid (e.g., a dsRNA) that is sufficiently complementary to an antiparallel sequence of nucleotides to permit hybridization between the two sequences of nucleotides under appropriate hybridization conditions (e.g., in a phosphate buffer, in a cell, etc.). In some embodiments, an oligonucleotide herein comprises a targeting sequence having a region of complementary to a mRNA target sequence. As used herein, “ribonucleotide” refers to a nucleotide having a ribose as its pentose sugar, which contains a hydroxyl group at its 2^ position. A modified ribonucleotide is a ribonucleotide having one or more modifications or substitutions of atoms other than at the 2^ position, including modifications or substitutions in or of the ribose, phosphate group or base. As used herein, “RNAi oligonucleotide” refers to either (a) a dsRNA having a sense strand (passenger) and antisense strand (guide), in which the antisense strand or part of the antisense strand is used by the Argonaute 2 (Ago2) endonuclease in the cleavage of a target mRNA or (b) a ss oligonucleotide having a single antisense strand, where that antisense strand (or part of that antisense strand) is used by the Ago2 endonuclease in the cleavage of a target mRNA. As used herein, “strand” refers to a single, contiguous sequence of nucleotides linked together through internucleotide linkages (e.g., phosphodiester linkages or phosphorothioate linkages). In some embodiments, a strand has two free ends (e.g., a 5^ end and a 3^ end). As used herein, “subject” means any mammal, including mice, rabbits, and humans. In one embodiment, the subject is a human or NHP. Moreover, “individual” or “patient” may be used interchangeably with “subject.”
FH12501620.1
Attorney Docket: DCY-13025 As used herein, “synthetic” refers to a nucleic acid or other molecule that is artificially synthesized (e.g., using a machine (e.g., a solid-state nucleic acid synthesizer)) or that is otherwise not derived from a natural source (e.g., a cell or organism) that normally produces the molecule. As used herein, “targeting ligand” refers to a molecule or “moiety” (e.g., a carboxylate, carbohydrate, amino sugar, cholesterol, polypeptide, or lipid) that selectively binds to a cognate molecule (e.g., a receptor) of a tissue or cell of interest and/or that is conjugatable to another substance for purposes of targeting the other substance to the tissue or cell of interest. For example, in some embodiments, a targeting ligand may be conjugated to an oligonucleotide for purposes of targeting the oligonucleotide to a specific tissue (e.g., extrahepatic tissue) or cell of interest. In some embodiments, a targeting ligand selectively binds to a cell surface receptor, such as a fatty acid binding proteins. Accordingly, in some embodiments, a targeting ligand when conjugated to an oligonucleotide facilitates delivery of the oligonucleotide into a particular cell through selective binding to a receptor expressed on the surface of the cell and endosomal internalization by the cell of the complex comprising the oligonucleotide, targeting ligand and receptor. In some embodiments, a targeting ligand is conjugated to an oligonucleotide via a linker that is cleaved following or during cellular internalization such that the oligonucleotide is released from the targeting ligand in the cell. As used herein, “tetraloop” refers to a loop that increases stability of an adjacent duplex formed by hybridization of flanking sequences of nucleotides. The increase in stability is detectable as an increase in melting temperature (Tm) of an adjacent stem duplex that is higher than the Tm of the adjacent stem duplex expected, on average, from a set of loops of comparable length consisting of randomly selected sequences of nucleotides. For example, a tetraloop can confer a Tm of at least about 50°C, at least about 55°C, at least about 56°C, at least about 58°C, at least about 60°C, at least about 65°C or at least about 75°C in 10 mM NaHPO
4 to a hairpin comprising a duplex of at least 2 base pairs (bp) in length. In some embodiments, a tetraloop may stabilize a bp in an adjacent stem duplex by stacking interactions. In addition, interactions among the nucleotides in a tetraloop include, but are not limited to, non-Watson-Crick base pairing, stacking interactions, hydrogen bonding and contact interactions (Cheong et al., (1990) NATURE 346:680-82; Heus and Pardi (1991) SCIENCE 253:191-94). In some embodiments, a tetraloop comprises or consists of 3 to 6 nucleotides and is typically 4 to 5 nucleotides. In some embodiments, a tetraloop comprises or consists of 3, 4, 5 or 6 nucleotides, which may or may not be modified (e.g., which may or may not be conjugated to a targeting moiety). In one embodiment, a tetraloop consists of 4 nucleotides. Any nucleotide may be used in the tetraloop
FH12501620.1
Attorney Docket: DCY-13025 and standard IUPAC-IUB symbols for such nucleotides may be used as described in Cornish- Bowden ((1985) NUCLEIC ACIDS RES.13:3021-3030). For example, the letter “N” may be used to mean that any base may be in that position, the letter “R” may be used to show that A (adenine) or G (guanine) may be in that position, and “B” may be used to show that C (cytosine), G (guanine), or T (thymine) may be in that position. Examples of tetraloops include the UNCG family of tetraloops (e.g., UUCG), the GNRA family of tetraloops (e.g., GAAA), and the CUUG tetraloop (Woese et al., (1990) PROC. NATL. ACAD. SCI. USA 87:8467-71; Antao et al., (1991) NUCLEIC ACIDS RES. 19:5901-05). Examples of DNA tetraloops include the d(GNNA) family of tetraloops (e.g., d(GTTA), the d(GNRA)) family of tetraloops, the d(GNAB) family of tetraloops, the d(CNNG) family of tetraloops, and the d(TNCG) family of tetraloops (e.g., d(TTCG)). (See, e.g., Nakano et al., (2002) B
IOCHEM. 41:4281-92; Shinji et al., (2000) N
IPPON K
AGAKKAI K
OEN Y
OKOSHU 78:731). In some embodiments, the tetraloop is contained within a nicked tetraloop structure. As used herein, “Tm-increasing nucleotide” refers to a nucleotide that increases the melting temperature (T
m) of an oligonucleotide duplex as compared to the oligonucleotide duplex without the T
m-increasing nucleotide. T
m-increasing nucleotides include, but are not limited to, bicyclic nucleotides, tricyclic nucleotides, a G-clamp, and analogues thereof, and hexitol nucleotides. Certain modified nucleotides having a modified sugar moiety, or a modified nucleobase can also be used to increase the Tm of an oligonucleotide duplex. As used herein, the term “Tm-increasing nucleotide” specifically excludes nucleotides modified at the 2^-position of the sugar moiety with 2^-OMe or 2^-F. As used herein, “treat” or “treating” refers to the act of providing care to a subject in need thereof, for example, by administering a therapeutic agent (e.g., an oligonucleotide herein) to the subject, for purposes of improving the health and/or well-being of the subject with respect to an existing condition (e.g., a disease, disorder) or to prevent or decrease the likelihood of the occurrence of a condition. In some embodiments, treatment involves reducing the frequency or severity of at least one sign, symptom or contributing factor of a condition (e.g., disease, disorder) experienced by a subject. As used herein, “alkyl” or “alkyl group” refers to a fully saturated, straight or branched hydrocarbon chain group, which is attached to the rest of the molecule by a single bond. Alkyls comprising any number of carbon atoms, including but not limited to from 1 to 12 are included. An alkyl comprising up to 12 carbon atoms is a C1-C12 alkyl, an alkyl comprising up to 10 carbon atoms is a C1-C10 alkyl, an alkyl comprising up to 6 carbon atoms is a C1-C6 alkyl and an alkyl comprising up to 5 carbon atoms is a C1-C5 alkyl. A C1-C5 alkyl includes C5 alkyls,
FH12501620.1
Attorney Docket: DCY-13025 C
4 alkyls, C
3 alkyls, C
2 alkyls and C
1 alkyl (i.e., methyl). A C
1-C
6 alkyl includes all moieties described above for C
1-C
5 alkyls but also includes C
6 alkyls. A C
1-C
10 alkyl includes all moieties described above for C
1-C
5 alkyls and C
1-C
6 alkyls, but also includes C
7, C
8, C
9 and C
10 alkyls. Similarly, a C
1-C
12 alkyl includes all the foregoing moieties, but also includes C
11 and C12 alkyls. Non-limiting examples of C1-C12 alkyl include methyl, ethyl, n-propyl, i-propyl, sec-propyl, n-butyl, i-butyl, sec-butyl, t-butyl, n-pentyl, t-amyl, n-hexyl, n-heptyl, n-octyl, n– nonyl, n-decyl, n-undecyl, and n-dodecyl. Unless stated otherwise, an alkyl group can be optionally substituted. “Alkylene” or “alkylene chain” refers to a fully saturated, straight or branched divalent hydrocarbon chain radical, and having from one to twelve carbon atoms. Non- limiting examples of C1-C12 alkylene include methylene, ethylene, propylene, n-butylene, and the like. The alkylene chain is attached to the rest of the molecule through a single bond and to a radical group (e.g., those described herein) through a single bond. The points of attachment of the alkylene chain to the rest of the molecule and to the radical group can be through one carbon or any two carbons within the chain. Unless stated otherwise, an alkylene chain can be optionally substituted. “Alkenyl” or “alkenyl group” refers to a straight or branched hydrocarbon chain having from two to twelve carbon atoms and having one or more carbon-carbon double bonds. Each alkenyl group is attached to the rest of the molecule by a single bond. Alkenyl group comprising any number of carbon atoms from 2 to 12 are included. An alkenyl group comprising up to 12 carbon atoms is a C2-C12 alkenyl, an alkenyl comprising up to 10 carbon atoms is a C2-C10 alkenyl, an alkenyl group comprising up to 6 carbon atoms is a C2-C6 alkenyl and an alkenyl comprising up to 5 carbon atoms is a C2-C5 alkenyl. A C2-C5 alkenyl includes C5 alkenyls, C4 alkenyls, C3 alkenyls, and C2 alkenyls. A C2-C6 alkenyl includes all moieties described above for C
2-C
5 alkenyls but also includes C
6 alkenyls. A C
2-C
10 alkenyl includes all moieties described above for C
2-C
5 alkenyls and C
2-C
6 alkenyls, but also includes C
7, C
8, C
9 and C
10 alkenyls. Similarly, a C
2-C
12 alkenyl includes all the foregoing moieties, but also includes C
11 and C
12 alkenyls. Non-limiting examples of C
2-C
12 alkenyl include ethenyl (vinyl), 1-propenyl, 2-propenyl (allyl), iso-propenyl, 2-methyl-1-propenyl, 1-butenyl, 2-butenyl, 3-butenyl, 1- pentenyl, 2-pentenyl, 3-pentenyl, 4-pentenyl, 1-hexenyl, 2-hexenyl, 3-hexenyl, 4-hexenyl, 5- hexenyl, 1-heptenyl, 2-heptenyl, 3-heptenyl, 4-heptenyl, 5-heptenyl, 6-heptenyl, 1-octenyl, 2- octenyl, 3-octenyl, 4-octenyl, 5-octenyl, 6-octenyl, 7-octenyl, 1-nonenyl, 2-nonenyl, 3- nonenyl, 4-nonenyl, 5-nonenyl, 6-nonenyl, 7-nonenyl, 8-nonenyl, 1-decenyl, 2-decenyl, 3- decenyl, 4-decenyl, 5-decenyl, 6-decenyl, 7-decenyl, 8-decenyl, 9-decenyl, 1-undecenyl, 2-
FH12501620.1
Attorney Docket: DCY-13025 undecenyl, 3-undecenyl, 4-undecenyl, 5-undecenyl, 6-undecenyl, 7-undecenyl, 8-undecenyl, 9-undecenyl, 10-undecenyl, 1-dodecenyl, 2-dodecenyl, 3-dodecenyl, 4-dodecenyl, 5- dodecenyl, 6-dodecenyl, 7-dodecenyl, 8-dodecenyl, 9-dodecenyl, 10-dodecenyl, and 11- dodecenyl. Unless stated otherwise, an alkyl group can be optionally substituted. “Alkenylene” or “alkenylene chain” refers to an unsaturated, straight or branched divalent hydrocarbon chain radical having one or more olefins and from two to twelve carbon atoms. Non-limiting examples of C2-C12 alkenylene include ethenylene, propenylene, n-butenylene, and the like. The alkenylene chain is attached to the rest of the molecule through a single bond and to a radical group (e.g., those described herein) through a single bond. The points of attachment of the alkenylene chain to the rest of the molecule and to the radical group can be through one carbon or any two carbons within the chain. Unless stated otherwise, an alkenylene chain can be optionally substituted. “Alkynyl” or “alkynyl group” refers to a straight or branched hydrocarbon chain having from two to twelve carbon atoms and having one or more carbon-carbon triple bonds. Each alkynyl group is attached to the rest of the molecule by a single bond. Alkynyl group comprising any number of carbon atoms from 2 to 12 are included. An alkynyl group comprising up to 12 carbon atoms is a C2-C12 alkynyl, an alkynyl comprising up to 10 carbon atoms is a C
2-C
10 alkynyl, an alkynyl group comprising up to 6 carbon atoms is a C
2-C
6 alkynyl and an alkynyl comprising up to 5 carbon atoms is a C2-C5 alkynyl. A C2-C5 alkynyl includes C5 alkynyls, C4 alkynyls, C3 alkynyls, and C2 alkynyls. A C2-C6 alkynyl includes all moieties described above for C2-C5 alkynyls but also includes C6 alkynyls. A C2-C10 alkynyl includes all moieties described above for C2-C5 alkynyls and C2-C6 alkynyls, but also includes C7, C8, C9 and C10 alkynyls. Similarly, a C2-C12 alkynyl includes all the foregoing moieties, but also includes C11 and C12 alkynyls. Non-limiting examples of C2-C12 alkenyl include ethynyl, propynyl, butynyl, pentynyl and the like. Unless stated otherwise, an alkyl group can be optionally substituted. “Alkynylene” or “alkynylene chain” refers to an unsaturated, straight or branched divalent hydrocarbon chain radical having one or more alkynes and from two to twelve carbon atoms. Non-limiting examples of C
2-C
12 alkynylene include ethynylene, propynylene, n-butynylene, and the like. The alkynylene chain is attached to the rest of the molecule through a single bond and to a radical group (e.g., those described herein) through a single bond. The points of attachment of the alkynylene chain to the rest of the molecule and to the radical group can be through any two carbons within the chain having a suitable valency. Unless stated otherwise, an alkynylene chain can be optionally substituted.
FH12501620.1
Attorney Docket: DCY-13025 “Alkoxy” refers to a group of the formula -OR
a where R
a is an alkyl, alkenyl or alknyl as defined above containing one to twelve carbon atoms. Unless stated otherwise, an alkoxy group can be optionally substituted. “Cycloalkyl” refers to a stable non-aromatic monocyclic or polycyclic fully saturated hydrocarbon group consisting solely of carbon and hydrogen atoms, which can include fused or bridged ring systems, having from three to twenty carbon atoms, preferably having from three to ten carbon atoms, and which is attached to the rest of the molecule by a single bond. Monocyclic cycloalkyl groups include, for example, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, cycloheptyl, and cyclooctyl. Polycyclic cycloalkyl groups include, for example, adamantyl, norbornyl, decalinyl, 7,7-dimethyl-bicyclo[2.2.1]heptanyl, and the like. Unless otherwise stated specifically in the specification, a cycloalkyl group can be optionally substituted. “Aryl” refers to a hydrocarbon ring system group comprising hydrogen, 6 to 18 carbon atoms and at least one aromatic ring. For purposes of this disclosure, the aryl group can be a monocyclic, bicyclic, tricyclic or tetracyclic ring system, which can include fused or bridged ring systems. Aryl groups include, but are not limited to, aryl groups derived from aceanthrylene, acenaphthylene, acephenanthrylene, anthracene, azulene, benzene, chrysene, fluoranthene, fluorene, as-indacene, s-indacene, indane, indene, naphthalene, phenalene, phenanthrene, pleiadene, pyrene, and triphenylene. Unless stated otherwise, the term “aryl” is meant to include aryl groups that are optionally substituted. “Heterocyclyl,” “heterocyclic ring” or “heterocycle” refers to a stable 3- to 20-membered ring group which consists of two to twelve carbon atoms and from one to six heteroatoms selected from the group consisting of nitrogen, oxygen and sulfur. The heterocyclyl group can be a monocyclic, bicyclic, tricyclic or tetracyclic ring system, which can include fused or bridged ring systems. The nitrogen, carbon or sulfur atoms in the heterocyclyl group can be optionally oxidized, the nitrogen atom can be optionally quaternized. The heterocyclyl group can be partially or fully saturated. Examples of such heterocyclyl groups include, but are not limited to, dioxolanyl, decahydroisoquinolyl, imidazolinyl, imidazolidinyl, isoxazolidinyl, morpholinyl, octahydroindolyl, octahydroisoindolyl, 2-oxopiperazinyl, 2-oxopiperidinyl, 2-oxopyrrolidinyl, oxazolidinyl, piperidinyl, piperazinyl, 4-piperidonyl, pyrrolidinyl, pyrazolidinyl, quinuclidinyl, thiazolidinyl, tetrahydrofuryl, trithianyl, tetrahydropyranyl, thiomorpholinyl, thiamorpholinyl, 1-oxo-thiomorpholinyl, and 1,1-dioxo-thiomorpholinyl. Unless stated otherwise, a heterocyclyl group can be optionally substituted.
FH12501620.1
Attorney Docket: DCY-13025 “Heteroaryl” refers to a 5- to 20-membered ring system group comprising hydrogen atoms, one to thirteen carbon atoms, one to six heteroatoms selected from the group consisting of nitrogen, oxygen and sulfur, and at least one aromatic ring. For purposes of this disclosure, the heteroaryl group can be a monocyclic, bicyclic, tricyclic or tetracyclic ring system, which can include fused or bridged ring systems; and the nitrogen, carbon or sulfur atoms in the heteroaryl group can be optionally oxidized; the nitrogen atom can be optionally quaternized. Examples include, but are not limited to, azepinyl, acridinyl, benzimidazolyl, benzothiazolyl, benzindolyl, benzodioxolyl, benzofuranyl, benzooxazolyl, benzothiazolyl, benzothiadiazolyl, benzo[b][1,4]dioxepinyl, 1,4-benzodioxanyl, benzonaphthofuranyl, benzoxazolyl, benzodioxolyl, benzodioxinyl, benzopyranyl, benzopyranonyl, benzofuranyl, benzofuranonyl, benzothienyl (benzothiophenyl), benzotriazolyl, benzo[4,6]imidazo[1,2-a]pyridinyl, carbazolyl, cinnolinyl, dibenzofuranyl, dibenzothiophenyl, furanyl, furanonyl, isothiazolyl, imidazolyl, indazolyl, indolyl, indazolyl, isoindolyl, indolinyl, isoindolinyl, isoquinolyl, indolizinyl, isoxazolyl, naphthyridinyl, oxadiazolyl, 2-oxoazepinyl, oxazolyl, oxiranyl, 1- oxidopyridinyl, 1-oxidopyrimidinyl, 1-oxidopyrazinyl, 1-oxidopyridazinyl, 1-phenyl-1H-pyrrolyl, phenazinyl, phenothiazinyl, phenoxazinyl, phthalazinyl, pteridinyl, purinyl, pyrrolyl, pyrazolyl, pyridinyl, pyrazinyl, pyrimidinyl, pyridazinyl, quinazolinyl, quinoxalinyl, quinolinyl, quinuclidinyl, isoquinolinyl, tetrahydroquinolinyl, thiazolyl, thiadiazolyl, triazolyl, tetrazolyl, triazinyl, and thiophenyl (i.e. thienyl). Unless stated otherwise, a heteroaryl group can be optionally substituted. The term “carboxyl” refers to radical group -COOH, or a charged form thereof, including carboxylate (e.g., -COO-). Therefore, any reference herein to a carboxyl group, such as a carboxylated ligand, also include the changed forms of said group. The term “substituted” used herein means any of the above groups wherein at least one hydrogen atom is replaced by a bond to a non-hydrogen atoms such as, but not limited to: a halogen atom such as F, Cl, Br, and I; an oxygen atom in groups such as hydroxyl groups, alkoxy groups, and ester groups; a sulfur atom in groups such as thiol groups, thioalkyl groups, sulfone groups, sulfonyl groups, and sulfoxide groups; a nitrogen atom in groups such as amines, amides, alkylamines, dialkylamines, arylamines, alkylarylamines, diarylamines, N- oxides, imides, and enamines; a silicon atom in groups such as trialkylsilyl groups, dialkylarylsilyl groups, alkyldiarylsilyl groups, and triarylsilyl groups; and other heteroatoms in various other groups. “Substituted” also means any of the above groups in which one or more hydrogen atoms are replaced by a higher-order bond (e.g., a double- or triple-bond) to a heteroatom such as oxygen in oxo, carbonyl, carboxyl, and ester groups; and nitrogen in groups
FH12501620.1
Attorney Docket: DCY-13025 such as imines, oximes, hydrazones, and nitriles. For example, “substituted” includes any of the above groups in which one or more hydrogen atoms are replaced with –NR
gR
h, – NR
gC(=O)R
h, –NR
gC(=O)NR
gR
h, –NR
gC(=O)OR
h, – NR
gSO
2R
h, -OC(=O)NR
gR
h, -OR
g, -SR
g, -SOR
g, -SO
2R
g, -OSO
2R
g, -SO
2OR
g, =NSO
2R
g, and -SO2NRgRh. “Substituted also means any of the above groups in which one or more hydrogen atoms are replaced with -C(=O)Rg, -C(=O)ORg, -C(=O)NRgRh, –CH2SO2Rg, – CH2SO2NRgRh. In the foregoing, Rg and Rh are the same or different and independently hydrogen, alkyl, alkenyl, alkynyl, alkoxy, alkylamino, thioalkyl, aryl, aralkyl, cycloalkyl, cycloalkenyl, cycloalkynyl, cycloalkylalkyl, haloalkyl, haloalkenyl, haloalkynyl, heterocyclyl, N-heterocyclyl, heterocyclylalkyl, heteroaryl, N-heteroaryl and/or heteroarylalkyl. “Substituted” further means any of the above groups in which one or more hydrogen atoms are replaced by a bond to an amino, cyano, hydroxyl, imino, nitro, oxo, thioxo, halo, alkyl, alkenyl, alkynyl, alkoxy, alkylamino, thioalkyl, aryl, aralkyl, cycloalkyl, cycloalkenyl, cycloalkynyl, cycloalkylalkyl, haloalkyl, haloalkenyl, haloalkynyl, heterocyclyl, N-heterocyclyl, heterocyclylalkyl, heteroaryl, N-heteroaryl and/or heteroarylalkyl group. In addition, each of the foregoing groups can also be optionally substituted with one or more of the above groups. EXEMPLARY EMBODIMENTS Embodiment I-1. A double-stranded oligonucleotide comprising: (i) an antisense strand of 15 to 30 nucleotides in length, a sense strand of 13 to 40 nucleotides in length, wherein the antisense strand has a region of complementarity to a target sequence in a target mRNA in an extrahepatic tissue, and wherein the region of complementarity is at least 15 contiguous nucleotides in length; and (ii) one or more ligands, each ligand comprising one or more -L-Y-(CO2H)n groups, or a charged form thereof, wherein L is a linker, Y is alkylene, alkenylene, or alkynylene, and n is 1-6, wherein when Y is alkylene, L comprises -O(CH
2)
a(OCH
2CH
2)
bNH-, wherein a is 1- 6 and b is 0-5, and wherein the ligand is conjugated to a nucleotide of the sense strand. Embodiment I-2. A double-stranded oligonucleotide comprising: (i) a double-stranded oligonucleotide comprising an antisense strand of 15 to 30 nucleotides in length, a sense strand of 15 to 40 nucleotides in length, wherein the antisense strand has a region of complementarity to a target sequence in a target mRNA in extrahepatic tissue, and wherein the region of complementarity is at least 15 contiguous nucleotides in length; and
FH12501620.1
Attorney Docket: DCY-13025 (ii) one or more ligands, each ligand comprising one or more C
6-24 alkylene-(CO
2H)
n, or a charged form thereof, conjugated to a nucleotide of the sense strand, wherein n is 1-6, wherein knock down of the target mRNA in the extrahepatic tissue is greater than knock down of the target mRNA in liver tissue. Embodiment I-3. The double-stranded oligonucleotide of Embodiment I-2, wherein each ligand comprises a linker (L), wherein L is conjugated to the one or more C6-24 alkylene- CO2H. Embodiment I-4. The double-stranded oligonucleotide of any one of Embodiments I-1-3, wherein L comprises a bivalent or trivalent C1-C50 alkylene, wherein 1-25 methylene groups are optionally and independently replaced by -N(H)-, -N(C1-C4 alkyl)-, -N(cycloalkyl)-, -O-, -C(O)-, -C(O)O-, -S-, -S(O)-, -S(O)2-, -S(O)2N(C1-C4 alkyl)-, -S(O)2N(cycloalkyl)-, - N(H)C(O)-, -N(C1-C4 alkyl)C(O)-, -N(cycloalkyl)C(O)-, -C(O)N(H)-, -C(O)N(C1-C4 alkyl), - C(O)N(cycloalkyl), aryl, heteroaryl, cycloalkyl, or cycloalkenyl. Embodiment I-5. The double-stranded oligonucleotide of Embodiment I-4, wherein 1-25 methylene groups are optionally and independently replaced by -N(H)-, -N(H)C(O)-, -N(C
1- C
4 alkyl)C(O)-, -O-, or heteroaryl. Embodiment I-6. The double-stranded oligonucleotide of Embodiment I-4 or 5, wherein the heteroaryl is a triazolyl. Embodiment I-7. The double-stranded oligonucleotide of Embodiment I-6, wherein the triazolyl
Embodiment I-8. The double-stranded oligonucleotide of any one of Embodiments I-1-7, wherein Y is an C
6-24 alkylene, C
6-24 alkenylene, or C
6-24 alkynylene. Embodiment I-9. The double-stranded oligonucleotide of any one of Embodiments I-1-8, wherein Y is an C6-24 alkylene or C6-24 alkenylene. Embodiment I-10. The double-stranded oligonucleotide of any one of Embodiments I-1-9, wherein Y is a C6-24 alkenylene. Embodiment I-11. The double-stranded oligonucleotide of any one of Embodiments I-1-9, wherein Y is a C
6-20 alkylene. Embodiment I-12. The double-stranded oligonucleotide of any one of Embodiments I-1- 10, wherein the alkenylene comprises from 1-6 olefinic bonds. Embodiment I-13. The double-stranded oligonucleotide of any one of Embodiments I-1- 10, wherein the alkynylene comprises from 1-6 acetylenic bonds.
FH12501620.1
Attorney Docket: DCY-13025 Embodiment I-14. The double-stranded oligonucleotide of any one of Embodiments I-1- 13, wherein a is 1. Embodiment I-15. The double-stranded oligonucleotide of any one of EmbodimentsI-1- 14, wherein b is 0 or 1. Embodiment I-16. The double-stranded oligonucleotide of Embodiment I-1 or 2, wherein the -L-Y-(CO2H)n group is:

, wherein: M is absent, -NRC(O)-, or heteroaryl, wherein R is H or alkyl. c is 0, 1, 2, 3, 4, 5, or 6; d is 0, 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, or 20; and e is 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, or 23. Embodiment I-17. The double-stranded oligonucleotide of Embodiment I-16, wherein M is -NRC(O)-. Embodiment I-18. The double-stranded oligonucleotide of Embodiment I-16 or 17, wherein R is H or methyl. Embodiment I-19. The double-stranded oligonucleotide of any one of Embodiments I-16- 18, wherein R is H. Embodiment I-20. The double-stranded oligonucleotide of Embodiment I-16, wherein M is heteroaryl. Embodiment I-21. The double-stranded oligonucleotide of Embodiment I-20, wherein the heteroaryl is triazolyl. Embodiment I-22. The double-stranded oligonucleotide of Embodiment I-21, wherein the triazolyl

Embodiment I-23. The double-stranded oligonucleotide of Embodiment I-16, wherein M is absent. Embodiment I-24. The double-stranded oligonucleotide of any one of Embodiments I-16- 23, wherein c is 1. Embodiment I-25. The double-stranded oligonucleotide of any one of Embodiments I-16- 24, wherein d is 0 to 12.
FH12501620.1
Attorney Docket: DCY-13025 Embodiment I-26. The double-stranded oligonucleotide of any one of Embodiments I-16- 25, wherein d is 4 to 12. Embodiment I-27. The double-stranded oligonucleotide of any one of Embodiments I-16- 26, wherein d is 4 or 12. Embodiment I-28. The double-stranded oligonucleotide of any one of Embodiments I-16- 25, wherein d is 0. Embodiment I-29. The double-stranded oligonucleotide of any one of Embodiments I-16- 28, wherein e is 12 to 23. Embodiment I-30. The double-stranded oligonucleotide of any one of Embodiments I-16- 28, wherein e is 11, 14, 15, 20, or 21. Embodiment I-31. The double-stranded oligonucleotide of any one of Embodiments I-1- 30, wherein the extrahepatic tissue is adipose tissue, heart tissue, skeletal muscle, or adrenal gland tissue. Embodiment I-32. The double-stranded oligonucleotide of any one of Embodiments I-1- 31, wherein the extrahepatic tissue is adipose tissue. Embodiment I-33. The double-stranded oligonucleotide of any one of Embodiments I-1- 32, wherein the antisense strand is 22 nucleotides. Embodiment I-34. The double-stranded oligonucleotide of any one of Embodiments I-1- 33, wherein the antisense strand comprises a 3’ overhang of 2 to 6 nucleotides. Embodiment I-35. The double-stranded oligonucleotide of any one of Embodiments I-1- 34, wherein the sense strand and/or antisense strand comprises at least one modified internucleotide linkage. Embodiment I-36. The double-stranded oligonucleotide of Embodiment I-35, wherein the at least one modified internucleotide linkage is a phosphorothioate linkage. Embodiment I-37. The double-stranded oligonucleotide of any one of Embodiments I-1- 36, wherein the antisense strand comprises a phosphorothioate linkage (i) between positions 1 and 2, and between positions 2 and 3; or (ii) between positions 1 and 2, between positions 2 and 3, and between positions 3 and 4, wherein positions are numbered 1-4 from 5’ to 3’. Embodiment I-38. The double-stranded oligonucleotide of any one of Embodiments I-35- 37, wherein the antisense strand is 22 nucleotides in length, and wherein the antisense strand comprises a phosphorothioate linkage between positions 20 and 21 and between positions 21 and 22, wherein positions are numbered 1-22 from 5^ to 3^. Embodiment I-39. The double-stranded oligonucleotide of any one of Embodiments I-35- 39, wherein the sense strand comprises a phosphorothioate linkage (i) between positions 1
FH12501620.1
Attorney Docket: DCY-13025 and 2; (ii) between positions 2 and 3; (iii) between positions 1 and 2, between positions 18 and 19, and between positions 19 and 20; or, (iv) between positions 2 and 3, between positions 18 and 19, and between positions 19 and 20, wherein positions are numbered 1-20 from 5^ to 3^. Embodiment I-40. The double-stranded oligonucleotide of any one of Embodiments I-35- 39, wherein the sense strand is 36 nucleotides in length, and wherein the sense strand comprises a phosphorothioate linkage between positions 1 and 2, wherein positions are numbered 1-36 from 5^ to 3^. Embodiment I-41. The double-stranded oligonucleotide of any one of Embodiments I-35- 39, wherein the sense strand is 20 nucleotides in length, and wherein the sense strand comprises a phosphorothioate linkage between positions 1 and 2, between positions 18 and 19, and between positions 19 and 20, wherein positions are numbered 1-20 from 5^ to 3^. Embodiment I-42. The double-stranded oligonucleotide of any one of Embodiments I-1- 41, wherein the oligonucleotide comprises a stem-loop comprising a tetraloop. Embodiment I-43. The double-stranded oligonucleotide of Embodiment I-42, wherein the tetraloop comprises a 5’-GAAA-3’ sequence. Embodiment I-44. The double-stranded oligonucleotide of any one of Embodiments I-1- 43, wherein the sense strand and/or antisense strand comprises one or more modified nucleotides. Embodiment I-45. The double-stranded oligonucleotide of Embodiment I-44, wherein the modified nucleotide comprises a modified sugar. Embodiment I-46. The double-stranded oligonucleotide of Embodiment I-45, wherein the modified sugar comprises a 2’-OMe group. Embodiment I-47. The double-stranded oligonucleotide of Embodiment I-45, wherein the modified sugar comprises a 2’-F substituent. Embodiment I-48. The double-stranded oligonucleotide of any one of Embodiments I-1- 47, wherein the antisense strand comprises up to 4 contiguous nucleotides with a 2’-F modified sugar. Embodiment I-49. The double-stranded oligonucleotide of any one of Embodiments I-1- 48, wherein the antisense strand comprises 22 nucleotides, wherein nucleotides at each of positions 2, 3, 4, 5, 7, 10, and 14, numbered 5^ to 3^ comprise a 2’-F modification. Embodiment I-50. The double-stranded oligonucleotide of any one of Embodiments I-1- 48, wherein the antisense strand comprises 22 nucleotides, wherein nucleotides at each of positions 2, 3, 4, 5, 7, 10, 14, 16, and 19, numbered 5^ to 3^, comprise a 2’-F modification.
FH12501620.1
Attorney Docket: DCY-13025 Embodiment I-51. The double-stranded oligonucleotide of any one of Embodiments I-1- 49, wherein the sense strand comprises 20 nucleotides, wherein nucleotides at each of positions 8, 9, 10, and 11 numbered 5^ to 3^, comprise a 2’-F modification. Embodiment I-52. The double-stranded oligonucleotide of any one of Embodiments I-1- 50, wherein the sense strand comprises 36 nucleotides, wherein nucleotides at each of positions 3, 5, 8, 10, 12, 13, 15, and 17, numbered 5^ to 3^, comprise a 2’-F modification. Embodiment I-53. The double-stranded oligonucleotide of any one of Embodiments I-1- 52, wherein the antisense strand comprises a 4’-O-monomethylphosphonate-2’-O-methyl modified nucleotide. Embodiment I-54. The double-stranded oligonucleotide of any one of Embodiments I-1- 52, wherein the antisense strand comprises a phosphorylated nucleotide at the 5’ terminus, and wherein the phosphorylated nucleotide is selected from uridine and adenosine. Embodiment I-55. The double-stranded oligonucleotide of any one of Embodiments I-1- 52, wherein the 4^-carbon of the sugar of the 5^-nucleotide of the antisense strand comprises a phosphate analog. Embodiment I-56. The double-stranded oligonucleotide of Embodiment I-55, wherein the phosphate analog is oxymethyl phosphonate, vinyl phosphonate or malonyl phosphonate. Embodiment I-57. The double-stranded oligonucleotide of any one of Embodiments I-54- 56, wherein the phosphorylated nucleotide is 4’-O-monomethylphosphonate-2’-O-methyl uridine. Embodiment I-58. The double-stranded oligonucleotide of any one of Embodiments I-1- 57, wherein the one or more ligands is conjugated to a ribose of the sense strand at the 3’- or 5’-position. Embodiment I-59. The double-stranded oligonucleotide of any one of Embodiments I-1- 57, wherein the one or more ligands is conjugated to a ribose of the sense strand at the 2’- position. Embodiment I-60. The double-stranded oligonucleotide of any one of Embodiments I-1- 59, wherein one ligand is conjugated to position one of the sense strand, numbered 5^ to 3^. Embodiment I-61. The double-stranded oligonucleotide of any one of Embodiments I-1- 59, comprising at least two ligands. Embodiment I-62. The double-stranded oligonucleotide of Embodiment I-61, wherein the at least two ligands are conjugated to different nucleotides of the sense strand. Embodiment I-63. The double-stranded oligonucleotide of Embodiment I-62, wherein the at least two ligands are the same ligand.
FH12501620.1
Attorney Docket: DCY-13025 Embodiment I-64. The double-stranded oligonucleotide of Embodiment I-62, wherein the at least two ligands are different ligands. Embodiment I-65. The double-stranded oligonucleotide of Embodiment I-63 or 64, wherein the at least two ligands are conjugated to position 1, numbered 5^ to 3^, and a nucleotide within a tetraloop. Embodiment I-66. The double-stranded oligonucleotide of Embodiment I-63 or 64, wherein (a) at least one ligand is conjugated to position 1 and at least one ligand is conjugated to position 29; (b) at least one ligand is conjugated to position 1 and at least one ligand is conjugated to position 28; (c) at least one ligand is conjugated to position 1 and at least one ligand is conjugated to position 2; or (d) at least one ligand is conjugated to position 1 and at least one ligand is conjugated to position 20, numbered 5^ to 3^, of the sense strand. Embodiment I-67. The double-stranded oligonucleotide of any one of Embodiments I-1- 66, wherein the region of complementarity is fully complementary to the mRNA target sequence. Embodiment I-68. The double-stranded oligonucleotide of any one of Embodiments I-1- 66, wherein the region of complementarity is partially complementary to the mRNA target sequence. Embodiment I-69. The double-stranded oligonucleotide of Embodiment I-68, wherein the region of complementarity comprises no more than four mismatches to the mRNA target sequence. Embodiment I-70. An oligonucleotide-ligand conjugate of Formula (AI):
FH12501620.1
Attorney Docket: DCY-13025

(AI), or a pharmaceutically acceptable salt or charged form thereof, wherein: A and A’ are each independently H or one or more nucleotides; B is a nucleobase; Z is O or S; M is absent, NRC(O), or heteroaryl, wherein R is H or alkyl. a is 1, 2, 3, 4, 5, or 6; b is 1, 2, 3, 4, 5, or 6; c is 0, 1, 2, 3, 4, 5, or 6; d is 0, 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, or 20; e is 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, or 23; and f is 0, 1, 2, 3, 4, 5, or 6. Embodiment I-71. The oligonucleotide-ligand conjugate of Embodiment I-70, wherein M is -NRC(O)-. Embodiment I-72. The oligonucleotide-ligand conjugate of Embodiment I-70 or 71, wherein R is H or C1-5 alkyl. Embodiment I-73. The oligonucleotide-ligand conjugate of any one of Embodiments I-70- 72, wherein R is H or methyl. Embodiment I-74. The oligonucleotide-ligand conjugate of any one of Embodiments I-70- 73, wherein R is H. Embodiment I-75. The oligonucleotide-ligand conjugate of any one of Embodiments I-70- 74, wherein c is 1. Embodiment I-76. The oligonucleotide-ligand conjugate of any one of Embodiments I-70- 75, having a structure of Formula (AII)
FH12501620.1
Attorney Docket: DCY-13025
(AII), or a pharmaceutically acceptable salt or charged form thereof. Embodiment I-77. The oligonucleotide-ligand conjugate of Embodiment I-70, wherein M is heteroaryl. Embodiment I-78. The oligonucleotide-ligand conjugate of Embodiment I-77, wherein the heteroaryl is triazolyl. Embodiment I-79. The oligonucleotide-ligand conjugate of Embodiment I-78, wherein the triazolyl
Embodiment I-80. The oligonucleotide-ligand conjugate of any one of Embodiments I-70 and 77-79, having a structure of Formula (AIII)
(AIII), or a pharmaceutically acceptable salt or charged form thereof. Embodiment I-81. The oligonucleotide-ligand conjugate of any one of Embodiments I-70- 80, wherein d is 0 to 12. Embodiment I-82. The oligonucleotide-ligand conjugate of any one of Embodiments I-70- 81, wherein d is 4 to 12. Embodiment I-83. The oligonucleotide-ligand conjugate of any one of Embodiments I-70- 82, wherein d is 4 or 12. Embodiment I-84. The oligonucleotide-ligand conjugate of any one of Embodiments I-70- 81, wherein d is 0.
FH12501620.1
Attorney Docket: DCY-13025 Embodiment I-85. The oligonucleotide-ligand conjugate of any one of Embodiments I-70, 77-79, and 84, having a structure of Formula (AIV)
(AIV), or a pharmaceutically acceptable salt or a charged form thereof. Embodiment I-86. The oligonucleotide-ligand conjugate of Embodiment I-70, wherein M is absent. Embodiment I-87. The oligonucleotide-ligand conjugate of Embodiment I-70 or 86, having a structure of Formula (AV)
(AV), or a pharmaceutically acceptable salt or a charged form thereof, wherein e is 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, or 23. Embodiment I-88. The oligonucleotide-ligand conjugate of any one of Embodiments I-70- 87, wherein a is 1 or 2. Embodiment I-89. The oligonucleotide-ligand conjugate of any one of Embodiments I-70- 88, wherein a is 1. Embodiment I-90. The oligonucleotide-ligand conjugate of any one of Embodiments I-70- 89, wherein b is 1 or 2. Embodiment I-91. The oligonucleotide-ligand conjugate of any one of Embodiments I-70- 90, wherein b is 1.
FH12501620.1
Attorney Docket: DCY-13025 Embodiment I-92. The oligonucleotide-ligand conjugate of any one of Embodiments I-70- 91, wherein e is 11 to 22. Embodiment I-93. The oligonucleotide-ligand conjugate of any one of Embodiments I-70- 92, wherein e is 11, 15, or 21. Embodiment I-94. The oligonucleotide-ligand conjugate of any one of Embodiments I-70- 93, wherein f is 0, 1, or 2. Embodiment I-95. The oligonucleotide-ligand conjugate of any one of Embodiments I-70- 94, wherein f is 0. Embodiment I-96. The oligonucleotide-ligand conjugate of any one of Embodiments I-70-
97. The oligonucleotide-ligand conjugate of any one of Embodiments I-70-96, wherein B

Embodiment I-98. The oligonucleotide-ligand conjugate of any one of Embodiments I-70- 97, wherein A is H. Embodiment I-99. The oligonucleotide-ligand conjugate of any one of Embodiments I-70- 98, wherein Z is S. Embodiment I-100. A double-stranded oligonucleotide comprising a sense strand and an antisense strand, wherein the sense strand comprises the oligonucleotide-ligand conjugate of any one of Embodiments I-70-99, and wherein the sense strand and antisense strand form a duplex region. Embodiment I-101. The double-stranded oligonucleotide of Embodiment I-100, wherein the antisense strand is 15 to 30 nucleotides in length. Embodiment I-102. The double-stranded oligonucleotide of any one of Embodiments I- 100-101, wherein the sense strand and/or antisense strand comprises at least one modified internucleotide linkage. Embodiment I-103. The oligonucleotide-ligand conjugate of Embodiment I-102, wherein the at least one modified internucleotide linkage is a phosphorothioate linkage.
FH12501620.1
Attorney Docket: DCY-13025 Embodiment I-104. The double-stranded oligonucleotide of any one of Embodiments I- 100-103, wherein A is H and A^ is 12 to 40 nucleotides. Embodiment I-105. The double-stranded oligonucleotide of any one of Embodiments I- 100-104, wherein the nucleotides of A^ comprise at least one modified internucleotide linkage. Embodiment I-106. The double-stranded oligonucleotide of any one of Embodiments I-100- 103, wherein A is 12-29 nucleotides and A^ is 1-10 nucleotides. Embodiment I-107. The double stranded oligonucleotide of Embodiment I-106, wherein the nucleotides of A comprise at least one modified internucleotide linkage. Embodiment I-108. The double-stranded oligonucleotide of any one of Embodiments I- 100-107, wherein the oligonucleotide-ligand conjugate and at least 14 nucleotides of A’ form the duplex region with the antisense strand. Embodiment I-109. The double-stranded oligonucleotide of any one of Embodiments I- 100-108, wherein the antisense strand comprises a phosphorothioate linkage (i) between positions 1 and 2, and between positions 2 and 3; or (ii) between positions 1 and 2, between positions 2 and 3, and between positions 3 and 4, wherein positions are numbered 1-4 from 5’ to 3’. Embodiment I-110. The double-stranded oligonucleotide of any one of Embodiments I- 100-108, wherein the antisense strand is 22 nucleotides in length, and wherein the antisense strand comprises a phosphorothioate linkage between positions 20 and 21 and between positions 21 and 22, wherein positions are numbered 1-22 from 5^ to 3^. Embodiment I-111. The double-stranded oligonucleotide of any one of Embodiments I- 100-110, wherein the sense strand comprises a phosphorothioate linkage (i) between positions 1 and 2; (ii) between positions 2 and 3; (iii) between positions 1 and 2, between positions 18 and 19, and between positions 19 and 20; or, (iv) between positions 2 and 3, between positions 18 and 19, and between positions 19 and 20, wherein positions are numbered 1-20 from 5^ to 3^. Embodiment I-112. The double-stranded oligonucleotide of any one of Embodiments I- 100-110, wherein the sense strand is 36 nucleotides in length, and wherein the sense strand comprises a phosphorothioate linkage between positions 1 and 2, wherein positions are numbered 1-36 from 5^ to 3^. Embodiment I-113. The double-stranded oligonucleotide of any one of Embodiments I- 100-110, wherein the sense strand is 20 nucleotides in length, and wherein the sense strand
FH12501620.1
Attorney Docket: DCY-13025 comprises a phosphorothioate linkage between positions 1 and 2, between positions 18 and 19, and between positions 19 and 20, wherein positions are numbered 1-20 from 5^ to 3^. Embodiment I-114. The double-stranded oligonucleotide of any one of Embodiments I- 100-113, wherein the duplex region comprises 20 to 30 base pairs. Embodiment I-115. The double-stranded oligonucleotide of any one of Embodiments I- 100-114, wherein the sense strand comprises a stem-loop comprising a tetraloop. Embodiment I-116. The double-stranded oligonucleotide of Embodiment I-115, wherein the tetraloop comprises a 5’-GAAA-3’ sequence. Embodiment I-117. The double-stranded oligonucleotide of Embodiment I-115, wherein the oligonucleotide-ligand conjugate is a nucleotide of the tetraloop. Embodiment I-118. The double-stranded oligonucleotide of any one of Embodiments I- 100-117, wherein the sense and antisense strand comprise one or more modified nucleotides. Embodiment I-119. The double-stranded oligonucleotide of Embodiment I-118, wherein the modified nucleotide comprises a modified sugar. Embodiment I-120. The double-stranded oligonucleotide of Embodiment I-119, wherein the modified sugar comprises a 2’-OMe group. Embodiment I-121. The double-stranded oligonucleotide of Embodiment I-119, wherein the modified sugar comprises a 2’-F substituent. Embodiment I-122. The double-stranded oligonucleotide of any one of Embodiments I- 100-121, wherein the sense strand comprises 20 nucleotides, wherein nucleotides at each of positions 8, 9, 10, and 11 comprise a 2’-F modification. Embodiment I-123. The double-stranded oligonucleotide of any one of Embodiments I- 100-121, wherein the sense strand comprises 36 nucleotides, wherein nucleotides at each of positions 3, 5, 8, 10, 12, 13, 15, and 17 comprise a 2’-F modification. Embodiment I-124. The double-stranded oligonucleotide of any one of Embodiments I- 100-121, wherein the antisense strand comprises 22 nucleotides, wherein nucleotides at each of positions 2, 3, 4, 5, 7, 10, and 14 comprise a 2’-F modification. Embodiment I-125. The double-stranded oligonucleotide of any one of Embodiments I- 100-121, wherein the antisense strand comprises 22 nucleotides, wherein nucleotides at each of positions 2, 3, 4, 5, 7, 10, 14, 16, and 19 comprise a 2’-F modification. Embodiment I-126. The double-stranded oligonucleotide of any one of Embodiments I- 100-125, wherein the modified nucleotide comprises a modified nucleobase. Embodiment I-127. The double-stranded oligonucleotide of any one of Embodiments I- 100-126 comprising a 4’-O-monomethylphosphonate-2’-O-methyl modified nucleotide.
FH12501620.1
Attorney Docket: DCY-13025 Embodiment I-128. The double-stranded oligonucleotide of any one of Embodiments I- 100-127, wherein the antisense strand comprises a phosphorylated nucleotide at the 5’ terminus, and wherein the phosphorylated nucleotide is selected from uridine and adenosine. Embodiment I-129. The double-stranded oligonucleotide of any one of Embodiments I- 100-127, wherein the 4^-carbon of the sugar of the 5^-nucleotide of the antisense strand comprises a phosphate analog. Embodiment I-130. The double-stranded oligonucleotide of Embodiment I-129, wherein the phosphate analog is oxymethyl phosphonate, vinyl phosphonate or malonyl phosphonate. Embodiment I-131. The double-stranded oligonucleotide of any one of Embodiments I- 128-130, wherein the phosphorylated nucleotide is 4’-O-monomethylphosphonate-2’-O- methyl uridine. Embodiment I-132. An oligonucleotide-ligand conjugate of Formula (BI)

(BI), or a pharmaceutically acceptable salt or charged form thereof, wherein: B is a nucleobase; A is a one or more nucleotides; Z
1 and Z
2 are each independently O or S; M is absent, NRC(O), or heteroaryl, wherein R is H or alkyl; a is 1, 2, 3, 4, or 5; c is 0, 1, 2, 3, 4, 5, or 6; d is 0, 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, or 20; e is 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, or 23; and f is 0, 1, 2, 3, 4, 5, or 6. Embodiment I-133. An oligonucleotide-ligand conjugate of Formula (BII)
FH12501620.1
Attorney Docket: DCY-13025
(BII), or a salt or charged form thereof, wherein: B is a nucleobase; A is a one or more nucleotides ; Z is O or S; M is absent, NRC(O), or heteroaryl, wherein R is H or alkyl; a is 1, 2, 3, 4, or 5; c is 0, 1, 2, 3, 4, 5, or 6; d is 0, 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, or 20; e is 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, or 23; and f is 0, 1, 2, 3, 4, 5, or 6. Embodiment I-134. An oligonucleotide-ligand conjugate of Formula (CI)
(CI), or a pharmaceutically acceptable salt or charged form thereof, wherein: B is a nucleobase; A is one or more nucleotides; Z
1 and Z
2 are each independently O or S; M is absent, NRC(O), or heteroaryl, wherein R is H or alkyl; a is 1, 2, 3, 4, or 5; c is 0, 1, 2, 3, 4, 5, or 6; d is 0, 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, or 20; e is 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, or 23; and
FH12501620.1
Attorney Docket: DCY-13025 f is 0, 1, 2, 3, 4, 5, or 6. Embodiment I-135. An oligonucleotide-ligand conjugate of Formula (CII)

(CII), or a salt or charged form thereof, wherein: B is a nucleobase; A is one or more nucleotides; Z is O or S; M is absent, NRC(O), or heteroaryl, wherein R is H or alkyl; a is 1, 2, 3, 4, or 5; c is 0, 1, 2, 3, 4, 5, or 6; d is 0, 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, or 20; e is 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, or 23; and f is 0, 1, 2, 3, 4, 5, or 6. Embodiment I-136. A double-stranded oligonucleotide comprising a sense strand and an antisense strand, wherein the sense strand comprises an oligonucleotide-ligand conjugate of any one of Embodiments I-132-135, and wherein the sense strand and antisense strand form a duplex region. Embodiment I-137. The double-stranded oligonucleotide of Embodiment I-136, wherein the antisense strand is 15 to 30 nucleotides in length. Embodiment I-138. The double-stranded oligonucleotide of Embodiment I-137, wherein A is 12 to 40 nucleotides. Embodiment I-139. The double-stranded oligonucleotide of any one of Embodiments I- 136-138, wherein the sense strand and/or antisense strand comprises at least one modified internucleotide linkage. Embodiment I-140. The double-stranded oligonucleotide of Embodiment I-139, wherein the at least one modified internucleotide linkage is a phosphorothioate linkage. Embodiment I-141. The double-stranded oligonucleotide of any one of Embodiments I- 136-140, wherein the duplex region includes one or more phosphorothioate linkages.
FH12501620.1
Attorney Docket: DCY-13025 Embodiment I-142. The double-stranded oligonucleotide of Embodiment I-141, wherein two phosphorothioate linkages are adjacent to each other. Embodiment I-143. The double-stranded oligonucleotide of any one of Embodiments I- 136-142, wherein the antisense strand comprises a phosphorothioate linkage (i) between positions 1 and 2, and between positions 2 and 3; or (ii) between positions 1 and 2, between positions 2 and 3, and between positions 3 and 4, wherein positions are numbered 1-4 from 5’ to 3’. Embodiment I-144. The double-stranded oligonucleotide of any one of Embodiments I- 136-143, wherein the antisense strand is 22 nucleotides in length, and wherein the antisense strand comprises a phosphorothioate linkage between positions 20 and 21 and between positions 21 and 22, wherein positions are numbered 1-22 from 5^ to 3^. Embodiment I-145. The double-stranded oligonucleotide of any one of Embodiments I- 136-144, wherein the sense strand comprises a phosphorothioate linkage (i) between positions 1 and 2; (ii) between positions 2 and 3; (iii) between positions 1 and 2, between positions 18 and 19, and between positions 19 and 20; or, (iv) between positions 2 and 3, between positions 18 and 19, and between positiosn 19 and 20, wherein positions are numbered 1-20 from 5^ to 3^. Embodiment I-146. The double-stranded oligonucleotide of any one of Embodiments I- 136-144, wherein the sense strand is 36 nucleotides in length, and wherein the sense strand comprises a phosphorothioate linkage between positions 1 and 2, wherein positions are numbered 1-36 from 5^ to 3^. Embodiment I-147. The double-stranded oligonucleotide of any one of Embodiments I- 136-144, wherein the sense strand is 20 nucleotides in length, and wherein the sense strand comprises a phosphorothioate linkage between positions 1 and 2, between positions 18 and 19, and between positions 19 and 20, wherein positions are numbered 1-20 from 5^ to 3^. Embodiment I-148. The double-stranded oligonucleotide of any one of Embodiments I- 136-148, wherein the duplex region comprises 20 to 30 base pairs. Embodiment I-149. The double-stranded oligonucleotide of any one of Embodiments I- 136-148, wherein the sense strand comprises a loop region that includes a tetraloop region. Embodiment I-150. The double-stranded oligonucleotide of Embodiment I-149, wherein the tetraloop region comprises a 5’-GAAA-3’ sequence. Embodiment I-151. The double-stranded oligonucleotide of any one of Embodiments I- 136-150, wherein the sense and antisense strand comprise one or more modified nucleotides.
FH12501620.1
Attorney Docket: DCY-13025 Embodiment I-152. The double-stranded oligonucleotide of Embodiment I-151, wherein the modified nucleotide comprises a modified sugar. Embodiment I-153. The double-stranded oligonucleotide of Embodiment I-152, wherein the modified sugar comprises a 2’-OMe group. Embodiment I-154. The double-stranded oligonucleotide of Embodiment I-152, wherein the modified sugar comprises a 2’-F substituent. Embodiment I-155. The double-stranded oligonucleotide of any one of Embodiments I- 136-154, wherein the sense strand comprises 20 nucleotides, wherein nucleotides at each of positions 8, 9, 10, and 11 comprise a 2’-F modification. Embodiment I-156. The double-stranded oligonucleotide of any one of Embodiments I- 136-154, wherein the sense strand comprises 36 nucleotides, wherein nucleotides at each of positions 3, 5, 8, 10, 12, 13, 15, and 17 comprise a 2’-F modification. Embodiment I-157. The double-stranded oligonucleotide of any one of Embodiments I- 136-154, wherein the antisense strand comprises 22 nucleotides, wherein nucleotides at each of positions 2, 3, 4, 5, 7, 10, and 14 comprise a 2’-F modification. Embodiment I-158. The double-stranded oligonucleotide of any one of Embodiments I- 136-154, wherein the antisense strand comprises 22 nucleotides, wherein nucleotides at each of positions 2, 3, 4, 5, 7, 10, 14, 16, and 19 comprise a 2’-F modification. Embodiment I-159. The double-stranded oligonucleotide of Embodiment I-151-158, wherein the modified nucleotide comprises a modified nucleobase. Embodiment I-160. The double-stranded oligonucleotide of any one of Embodiments I- 136-159 comprising a 4’-O-monomethylphosphonate-2’-O-methyl modified nucleotide. Embodiment I-161. The double-stranded oligonucleotide of any one of Embodiments I- 136-160, wherein the antisense strand comprises a phosphorylated nucleotide at the 5’ terminus, and wherein the phosphorylated nucleotide is selected from uridine and adenosine. Embodiment I-162. The double-stranded oligonucleotide of any one of Embodiments I- 136-160, wherein the 4^-carbon of the sugar of the 5^-nucleotide of the antisense strand comprises a phosphate analog. Embodiment I-163. The double-stranded oligonucleotide of Embodiment I-162, wherein the phosphate analog is oxymethyl phosphonate, vinyl phosphonate or malonyl phosphonate. Embodiment I-164. The double-stranded oligonucleotide of any one of Embodiments I- 161-163, wherein the phosphorylated nucleotide is 4’-O-monomethylphosphonate-2’-O- methyl uridine.
FH12501620.1
Attorney Docket: DCY-13025 Embodiment I-165. The double-stranded oligonucleotide of any one of Embodiments I- 100-164, wherein the sense strand comprises a second oligonucleotide-ligand conjugate. Embodiment I-166. The double-stranded oligonucleotide of Embodiment I-165, wherein the second oligonucleotide-ligand conjugate is selected from the oligonucleotide-ligand conjugate of any one of Embodiments I-100 and 132-135. Embodiment I-167. The double-stranded oligonucleotide of any one of Embodiments I- 100-131 and 136-166, wherein the antisense strand has a region of complementarity to a target sequence in a target mRNA in extrahepatic tissue, wherein the region of complementarity is at least 15 contiguous nucleotides in length. Embodiment I-168. The double-stranded oligonucleotide of Embodiment I-167, wherein the region of complementarity is fully complementary to the mRNA target sequence. Embodiment I-169. The double-stranded oligonucleotide of Embodiment I-167, wherein the region of complementarity is partially complementary to the mRNA target sequence. Embodiment I-170. The double-stranded oligonucleotide of Embodiment I-169, wherein the region of complementarity comprises no more than four mismatches to the mRNA target sequence. Embodiment I-171. The double-stranded oligonucleotide of any one of Embodiments I- 167-170, wherein the extrahepatic tissue is adipose tissue, heart tissue, skeletal muscle, or adrenal gland tissue. Embodiment I-172. The double-stranded oligonucleotide of any one of Embodiments I- 167-171, wherein the extrahepatic tissue is adipose tissue. Embodiment I-173. A double-stranded oligonucleotide comprising a sense strand and an antisense strand, wherein the sense strand comprises two or more oligonucleotide-ligand conjugates of (i), (ii), or (iii): (i) Formula (AI), (AII), (AIII), (AIV), or (AV); (ii) Formula (BI) or (BII); and (iii) Formula (CI) or (CII). Embodiment I-174. The double-stranded oligonucleotide of Embodiment I-173, wherein the two or more oligonucleotide ligand conjugates are conjugated to different nucleotides of the sense strand. Embodiment I-175. The double-stranded oligonucleotide of Embodiment I-173 or 174, wherein the two or more oligonucleotide ligand conjugates are the same. Embodiment I-176. The double-stranded oligonucleotide of Embodiment I-173 or 174, wherein the two or more oligonucleotide ligand conjugates are different.
FH12501620.1
Attorney Docket: DCY-13025 Embodiment I-177. The double-stranded oligonucleotide of Embodiment I-173, wherein the sense strand comprises a first oligonucleotide-ligand conjugate and a second oligonucleotide- ligand conjugate. Embodiment I-178. The double-stranded oligonucleotide of Embodiment I-177, wherein the nucleobase (B) of the first oligonucleotide-ligand conjugate is the nucleobase at position 1 of the sense strand and the nucleobase (B) of the second oligonucleotide-ligand conjugate is a nucleobase within a tetraloop of the sense strand. Embodiment I-179. The double-stranded oligonucleotide of Embodiment I-177, wherein (a) the nucleobase (B) of the first oligonucleotide-ligand conjugate is the nucleobase at position 1 of the sense strand, and the nucleobase (B) of the second oligonucleotide-ligand conjugate is the nucleobase at position 29 of the sense strand; (b) the nucleobase (B) of the first oligonucleotide-ligand conjugate is the nucleobase at position 1 of the sense strand, and the nucleobase (B) of the second oligonucleotide-ligand conjugate is the nucleobase at position 28 of the sense strand; (c) the nucleobase (B) of the first oligonucleotide-ligand conjugate is the nucleobase at position 1 of the sense strand, and the nucleobase (B) of the second oligonucleotide-ligand conjugate is the nucleobase at position 20 of the sense strand; or (d) the nucleobase (B) of the first oligonucleotide-ligand conjugate is the nucleobase at position 1 of the sense strand, and the nucleobase (B) of the second oligonucleotide-ligand conjugate is the nucleobase at position 2 of the sense strand. Embodiment I-180. The double-stranded oligonucleotide of any one of the proceeding Embodiments I, wherein the oligonucleotide-ligand conjugate reduces expression of the target mRNA in an extrahepatic tissue, provided the oligonucleotide-ligand conjugate does not reduce expression of the mRNA target in the liver. Embodiment I-181. A pharmaceutical composition comprising the double-stranded oligonucleotide of any one of Embodiments I-1-69, 100-131, and 136-166, and a pharmaceutically acceptable carrier, delivery agent, or excipient. Embodiment I-182. A method of inhibiting target mRNA expression in an extrahepatic cell or tissue in a subject, comprising administering to the subject the double-stranded oligonucleotide of any one of Embodiments I-1-69, 100-131, and 136-166, or the pharmaceutical composition of Embodiment I-181, thereby inhibiting target mRNA expression in the cell of the extrahepatic tissue.
FH12501620.1
Attorney Docket: DCY-13025 Embodiment I-183. The method of Embodiment I-182, wherein the extrahepatic cell or tissue is selected from skeletal muscle, adipose tissue, adrenal tissue, heart tissue, and any combination thereof. Embodiment I-184. The method of Embodiment I-182 or 183, wherein reduction of the target mRNA in the extrahepatic cell or tissue is increased compared to reduction in liver cells or tissue, optionally wherein reduction of the target mRNA is increased by at least 10%. Embodiment I-185. The method of Embodiment I-184, wherein reduction of the target mRNA is increased by at least 15%, at least 20%, at least 25%, at least 30%, at least 35%, at least 40%, at least 45% or at least 50%. Embodiment I-186. The double-stranded oligonucleotide of any one of Embodiments I-1-180 in the manufacture of a medicament for inhibiting target mRNA expression in an extrahepatic cell or tissue in a subject. Embodiment I-187. Use of the double-stranded oligonucleotide of any one of Embodiments I-1-180 for inhibiting target mRNA expression in a cell of an extrahepatic tissue in a subject. Embodiment I-188. A kit comprising a container comprising the double-stranded oligonucleotide of any one of Embodiments I-1-69, 100-131, and 136-166, and optionally a pharmaceutically acceptable carrier, and instructions for administering the oligonucleotide- ligand conjugate to a subject in need thereof, wherein the oligonucleotide-ligand conjugate inhibits target mRNA expression in an extrahepatic cell or tissue in the subject. Embodiment I-189. The double-stranded oligonucleotide of Embodiment I-186, the use of Embodiment I-187, or the kit of Embodiment I-188, wherein the extrahepatic cell or tissue is selected from skeletal muscle, adipose tissue, adrenal tissue, heart tissue, and any combination thereof. Embodiment I-190. The double-stranded oligonucleotide of Embodiment I-186, the use of Embodiment I-187, or the kit of Embodiment I-188, wherein the cell of the extrahepatic cell or tissue is selected from a cardiomyocyte, a cell of skeletal muscle, a cell of adipose tissue, a cell of adrenal tissue, and any combination thereof. Embodiment I-191. The double-stranded oligonucleotide, use, or kit of any one of Embodiments I-186-189, wherein reduction of the target mRNA in the extrahepatic cell or tissue is increased compared to reduction in a cell of the liver, optionally wherein reduction of the target mRNA is increased by at least 10%.
FH12501620.1
Attorney Docket: DCY-13025 Embodiment I-192. The double-stranded oligonucleotide, use, or kit of Embodiment I-191, wherein reduction of the target mRNA is increased by at least 15%, at least 20%, at least 25%, at least 30%, at least 35%, at least 40%, at least 45% or at least 50%. Embodiment I-193. A method for treating a subject having a disease, disorder or condition associated with expression of an mRNA in an extrahepatic cell or tissue, the method comprising administering to the subject a therapeutically effective amount of the double- stranded oligonucleotide of any one of Embodiments I-1-69, 100-131, and 136-166, or the pharmaceutical composition of Embodiment I-181. Embodiment I-194. A method of delivering a double-stranded oligonucleotide to a cell or population of cells in extrahepatic tissue, the method comprising administering the pharmaceutical composition of Embodiment I-181. Embodiment II-1. A double-stranded oligonucleotide comprising: (i) an antisense strand of 15 to 30 nucleotides in length, a sense strand of 13 to 40 nucleotides in length, wherein the antisense strand has a region of complementarity to a target sequence in a target mRNA in an extrahepatic tissue, and wherein the region of complementarity is at least 15 contiguous nucleotides in length; and (ii) one or more ligands, each ligand comprising one or more -L-C6-24 alkylene- (CO
2H)
n groups, or a charged form thereof, wherein L is a linker and comprises -O(CH2)a(OCH2CH2)bNH-, -O(CH2)a(OCH2CH2)b(NH)(CO)-, or -O(CH2)a(OCH2CH2)b(NH)(CO)(CH2)c(OCH2CH2)dM-, wherein M is absent, -NRC(O)-, or heteroarylene; R is H or alkyl; n is 1-6; a is 1-6; b is 0-5; c is 0, 1, 2, 3, 4, 5, or 6; and d is 0, 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, or 20; wherein the ligand is conjugated to a nucleotide of the sense strand. Embodiment II-2. The double-stranded oligonucleotide of Embodiment II- 0, wherein n is 1. Embodiment II-3. The double-stranded oligonucleotide of Embodiment II- 0 or 2, wherein at least one ligand comprises -L-C16 alkylene-(CO2H)n. Embodiment II-4. The double-stranded oligonucleotide of Embodiment II-0 or 2, wherein at least one ligand comprises -L-C22 alkylene-(CO2H)n.
FH12501620.1
Attorney Docket: DCY-13025 Embodiment II-5. The double-stranded oligonucleotide of any one of Embodiments II-1-4, wherein L is -O(CH
2)
a(OCH
2CH
2)
b(NH)(CO)(CH
2)
c(OCH
2CH
2)
dM-. Embodiment II-6. The double-stranded oligonucleotide of Embodiment II-5, wherein M is heteroarylene. Embodiment II-7. The double-stranded oligonucleotide of Embodiment II- 6, wherein the heteroarylene is triazolylene. Embodiment II-8. The double-stranded oligonucleotide of Embodiment II-7, wherein triazolylene is

. Embodiment II-9. The double-stranded oligonucleotide of any one of Embodiments II-1-8, wherein a is 1. Embodiment II-10. The double-stranded oligonucleotide of any one of Embodiments II-1- 9, wherein b is 2. Embodiment II-11. The double-stranded oligonucleotide of any one of Embodiments II-1- 10, wherein knock down of the target mRNA in the extrahepatic tissue is greater than knock down of the target mRNA in liver tissue. Embodiment II-12. The double-stranded oligonucleotide of Embodiment II-11, wherein the extrahepatic tissue is adipose tissue, heart tissue, skeletal muscle, or adrenal gland tissue. Embodiment III-1. A double-stranded oligonucleotide comprising: (i) an antisense strand of 15 to 30 nucleotides in length, a sense strand of 13 to 40 nucleotides in length, wherein the antisense strand has a region of complementarity to a target sequence in a target mRNA in an extrahepatic tissue, and wherein the region of complementarity is at least 15 contiguous nucleotides in length; and (ii) one or more ligands, each ligand comprising one or more -L-C6-24 alkylene-Wn groups, or a charged form thereof, wherein L is a linker and comprises -O(CH2)a(OCH2CH2)bNH-, -O(CH2)a(OCH2CH2)b(NH)(CO)-, or -O(CH2)a(OCH2CH2)b(NH)(CO)(CH2)c(OCH2CH2)dM-, wherein: W is -C(O)OH, -CR^(C(O)OH)2, -CR^(C(O)O(alkyl))2, -SO2OH, tetrazolyl, -OH, -N(H)SO2(alkyl), -N(H)SO2(cycloalkyl), -N(H)SO2(optionally substituted aryl), -N(H)SO
2(heterocyclyl), -N(H)SO
2(optionally substituted heteroaryl), - O(optionally substituted aryl), -N(H)C(O)N(H)SO
2(alkyl), -C(O)NH(hydroxyphenyl), -N(H)
FH12501620.1
Attorney Docket: DCY-13025 C(O)(hydroxyphenyl), -N(H)(hydroxyphenyl), hydroxyphenyl, or heterocyclyl, M is absent, -NRC(O)-, or heteroarylene; R is H or alkyl; R^ is H or methyl; n is 1-6; a is 1-6; b is 0-5; c is 0, 1, 2, 3, 4, 5, or 6; and d is 0, 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, or 20; and wherein the ligand is conjugated to a nucleotide of the sense strand. Embodiment III-2. The double-stranded oligonucleotide of Embodiment III-0, wherein n is 1. Embodiment III-3. The double-stranded oligonucleotide of Embodiment III-0 or 2, wherein at least one ligand comprises -L-C16 alkylene-Wn. Embodiment III-4. The double-stranded oligonucleotide of Embodiment III-0 or 2, wherein at least one ligand comprises -L-C
22 alkylene-W
n. Embodiment III-5. The double-stranded oligonucleotide of any one of Embodiments III-0-4, wherein W is
, , , - OH, -N(H)SO2(methyl), -N(H)SO2(ethyl), -N(H)SO2(n-propyl), -N(H)SO2(i-propyl), -N( H)SO2(sec-propyl), -N(H)SO2(n-butyl), -N(H)SO2(i-butyl), -N(H)SO2(sec- butyl), -N(H)SO2(t-butyl), -N(H)SO2(n- pentyl), -N(H)SO2(cyclopropyl), -N(H)SO2(cyclobutyl), -N(H)SO2(cyclopentyl), -N(H)SO2(cyclohexyl), -N(H)SO2(cycloheptyl),
-N(H)SO2(cyclooctyl), -N(H)SO2([1.1.1]propellane), ,
FH12501620.1
Attorney Docket: DCY-13025
Embodiment III-6. The double-stranded oligonucleotide of Embodiment III-0, wherein W is
. Embodiment III-7. The double-stranded oligonucleotide of Embodiment III-0, wherein W is
. Embodiment III-8. The double-stranded oligonucleotide of Embodiment III-0, wherein W is -N(H)C(O)N(H)SO2(CH3). Embodiment III-9. The double-stranded oligonucleotide of Embodiment III-0, wherein W is
. Embodiment III-10. The double-stranded oligonucleotide of any one of Embodiments III-0-4, wherein W is piperazinyl. Embodiment III-11. The double-stranded oligonucleotide of any one of Embodiments III-0-4, wherein W is piperidinyl. Embodiment III-12. The double-stranded oligonucleotide of any one of Embodiments III-0- 11, wherein L is -O(CH2)a(OCH2CH2)b(NH)(CO)(CH2)c(OCH2CH2)dM-.
FH12501620.1
Attorney Docket: DCY-13025 Embodiment III-13. The double-stranded oligonucleotide of Embodiment III-12, wherein M is heteroarylene. Embodiment III-14. The double-stranded oligonucleotide of Embodiment III-0, wherein the heteroarylene is triazolylene. Embodiment III-15. The double-stranded oligonucleotide of Embodiment III-0, wherein triazolylene is

. Embodiment III-16. The double-stranded oligonucleotide of any one of Embodiments III-1- 15, wherein a is 1. Embodiment III-17. The double-stranded oligonucleotide of any one of Embodiments III-1- 16, wherein b is 2. Embodiment III-18. The double-stranded oligonucleotide of any one of Embodiments III-1-0, wherein knock down of the target mRNA in the extrahepatic tissue is greater than knock down of the target mRNA in liver tissue. Embodiment III-19. The double-stranded oligonucleotide of Embodiment III-0, wherein the extrahepatic tissue is adipose tissue, heart tissue, skeletal muscle, or adrenal gland tissue. EXAMPLES Preparation of Double-Stranded RNAi Oligonucleotides General Synthetic Methods The following examples are intended to illustrate the disclosure and are not to be construed as being limitations thereon. Temperatures are given in degrees centigrade (^C). If not mentioned otherwise, all evaporations are performed under reduced pressure, preferably between about 15 mm Hg and 100 mm Hg (= 20-133 mbar). The structure of final products, intermediates and starting materials was confirmed by standard analytical methods, e.g., microanalysis and spectroscopic characteristics, e.g., MS, IR, NMR. Abbreviations used are those conventional in the art. All starting materials, building blocks, reagents, acids, bases, dehydrating agents, solvents, and catalysts utilized to synthesis the nucleic acid or analogues thereof of the present disclosure are either commercially available or can be produced by organic synthesis methods known to one of ordinary skill in the art (Methods of Organic Synthesis, Thieme, Volume
FH12501620.1
Attorney Docket: DCY-13025 21 (Houben-Weyl 4th Ed.1952)). Further, the nucleic acid or analogues thereof of the present disclosure can be produced by organic synthesis methods known to one of ordinary skill in the art as shown in the following examples. All reactions are carried out under nitrogen or argon unless otherwise stated. Proton NMR (
1H NMR) was conducted in deuterated solvent. In certain nucleic acid or analogues thereof disclosed herein, one or more
1H shifts overlap with residual proteo solvent signals; these signals have not been reported in the experimental provided hereinafter. As depicted in the Examples below, in certain exemplary embodiments, the nucleic acid or analogues thereof were prepared according to the following general procedures. It will be appreciated that, although the general methods depict the synthesis of certain nucleic acid or analogues thereof of the present disclosure, the following general methods, and other methods known to one of ordinary skill in the art, can be applied to all nucleic acid or analogues thereof and subclasses and species of each of these nucleic acid or analogues thereof, as described herein. Example 1a: Synthesis of 2-(2-((((6aR,8R,9R,9aR)-8-(6-benzamido-9H-purin-9-yl)- 2,2,4,4-tetraisopropyltetrahydro-6H-furo[3,2-f][1,3,5,2,4]trioxadisilocin-9- yl)oxy)methoxy)ethoxy) ethan-1-ammonium formate (1-6)
FH12501620.1
Attorney Docket: DCY-13025 A solution of compound 1-1 (25.00 g, 67.38 mmol) in 20 mL of DMF was treated with pyridine (11 mL, 134.67 mmol) and tetraisopropyldisiloxane dichloride (22.63 mL, 70.75 mmol) at 10 °C. The resulting mixture was stirred at 25 °C for 3 h and quenched with 20% citric acid (50 mL). The aqueous layer was extracted with EtOAc (3X50 mL), and the combined organic layers were concentrated in vacuo. The crude residue was recrystallized from a mixture of MTBE and n-heptane (1:15, 320 mL) to afford compound 1-2 (37.20 g, 90%) as a white oily solid. A solution of compound 1-2 (37.00 g, 60.33 mmol) in 20 mL of DMSO was treated with AcOH (20 mL, 317.20 mmol) and Ac2O (15 mL, 156.68 mmol). The mixture was stirred at 25 °C for 15 h. The reaction was diluted with EtOAc (100 mL) and quenched with sat. K2CO3 (50 mL). The aqueous layer was extracted with EtOAc (3X50 mL). The combined organic layers were concentrated and recrystallized with ACN (30 mL) to afford compound 1-3 (15.65 g, 38.4%) as a white solid. A solution of compound 1-3 (20.00 g, 29.72 mmol) in 120 mL of DCM was treated with Fmoc-amino-ethoxy ethanol (11.67 g, 35.66 mmol) at 25 °C. The mixture was stirred to afford a clear solution and then treated with 4Å molecular sieves (20.0 g), N-iodosuccinimide (8.02 g, 35.66 mmol), and TfOH (5.25 mL, 59.44 mmol). The mixture was stirred at 30 °C until the HPLC analysis indicated >95% consumption of compound 1-3. The reaction was quenched with TEA (6 mL) and filtered. The filtrate was diluted with EtOAc, washed with sat. NaHCO3 (2X100 mL), sat. Na2SO3 (2X100 mL), and water (2X100 mL) and concentrated in vacuo to afford crude compound 1-4 (26.34 g, 93.9%) as a yellow solid, which was used directly for the next step without further purification. A solution of compound 1-4 (26.34 g, 27.62 mmol) in a mixture of DCM/water (10:7, 170 mL) was treated with DBU (7.00 mL, 45.08 mmol) at 5 °C. The mixture was stirred at 5-25 °C for 1 h. The organic layer was then separated, washed with water (100 mL), and diluted with DCM (130 mL). The solution was treated with fumaric acid (7.05 g, 60.76 mmol) and 4Å molecular sieves (26.34 g) in four portions. The mixture was stirred for 1 h, concentrated, and recrystallized from a mixture of MTBE and DCM (5:1) to afford compound 1-6 (14.74 g, 62.9%) as a white solid:
1H NMR (400 MHz, d
6-DMSO) 8.73 (s, 1H), 8.58 (s, 1H), 8.15-8.02 (m, 2H), 7.65-7.60 (m, 1H), 7.59-7.51 (m, 2H), 6.52 (s, 2H), 6.15(s, 1H), 5.08-4.90 (m, 3H), 4.83-4.78 (m, 1H), 4.15-3.90 (m, 3H), 3.79-3.65 (m, 2H), 2.98-2.85 (m, 6H), 1.20-0.95 (m, 28H).
FH12501620.1
Attorney Docket: DCY-13025 Example 1b: Synthesis of (2R,3R,4R,5R)-5-(6-benzamido-9H-purin-9-yl)-2-((bis(4- methoxyphenyl)(phenyl)methoxy)methyl)-4-((2-(2-[lipid]-amidoethoxy)ethoxy)methoxy)
A solution of compound 1-6 (50.00 g, 59.01 mmol) in 150 mL of 2-methyltetrahydrofuran was washed with ice cold aqueous K
2HPO
4 (6%, 100 mL) and brine (20%, 2X100 mL). The organic layer was separated and treated with hexanoic acid (10.33 mL, 82.61 mmol), HATU (33.66 g, 88.52 mmol), and DMAP (10.81 g, 147.52 mmol) at 0 °C. The resulting mixture was warmed to 25 °C and stirred for 1 h. The solution was washed with water (2X100 mL), brine (100 mL), and concentrated in vacuo to afford a crude residue. Flash chromatography on silica gel (1:1 hexanes/acetone) gave compound 2-1a (34.95 g, 71.5%) as a white solid. A mixture of compound 2-1a (34.95 g, 42.19 mmol) and TEA (9.28 mL, 126.58 mmol) in 80 mL of THF was treated with triethylamine trihydrofluoride (20.61 mL, 126.58 mmol) dropwise at 10 °C. The mixture was warmed to 25 °C and stirred for 2 h. The reaction was concentrated, dissolved in DCM (100 mL), and washed with sat. NaHCO3 (5X20 mL) and
FH12501620.1
Attorney Docket: DCY-13025 brine (50 mL). The organic layer was concentrated in vacuo to afford crude compound 2-2a (24.72 g, 99%), which was used directly for the next step without further purification. A solution of compound 2-2a (24.72 g, 42.18 mmol) in 50 mL of DCM was treated with N- methylmorpholine (18.54 mL, 168.67 mmol) and DMTr-Cl (15.69 g, 46.38 mmol). The mixture was stirred at 25 °C for 2 h and quenched with sat. NaHCO3 (50 mL). The organic layer was separated, washed with water, concentrated to afford a slurry crude. Flash chromatography on silica gel (1:1 hexanes/acetone) gave compound 2-3a (30.05 g, 33.8 mmol, 79.9%) as a white solid. A solution of compound 2-3a (25.00 g, 28.17 mmol) in 50 mL of DCM was treated with N- methylmorpholine (3.10 mL, 28.17 mmol) and tetrazole (0.67 mL, 14.09 mmol) under nitrogen atmosphere. Bis(diisopropylamino) chlorophosphine (9.02 g, 33.80 mmol) was added to the solution dropwise and the resulting mixture was stirred at 25 °C for 4 h. The reaction was quenched with water (15 mL), and the aqueous layer was extracted with DCM (3X50 mL). The combined organic layers were washed with sat. NaHCO
3 (50 mL), concentrated to afford a crude solid that was recrystallized from a mixture of DCM/MTBE/n- hexane (1:4:40) to afford compound 2-4a (25.52 g, 83.4%) as a white solid:
1H NMR (400 MHz, d6-DMSO) 11.25 (s, 1H), 8.65-8.60 (m, 2 H), 8.09-8.02 (m, 2H), 7.71 (s, 1H), 7.67- 7.60 (m, 1H), 7.59-7.51 (m, 2H), 7.38-7.34 (m, 2H), 7.30-7.25 (m, 7H), 6.85-6.79 (m, 4H), 6.23-6.20 (m, 1H), 5.23-5.14 (m, 1H), 4.80-4.69 (m, 3H), 4.33-4.23 (m, 2H), 3.90-3.78 (m, 1H), 3.75 (s, 6H), 3.74-3.52 (m, 3H), 3.50-3.20 (m, 6H), 3.14-3.09 (m, 2H), 3.09 (s, 1H), 2.82-2.80 (m, 1H), 2.65-2.60 (m, 1H), 2.05-1.96 (m, 2H), 1.50-1.39 (m, 2H), 1.31-1.10 (m, 14H), 1.08-1.05 (m, 2 H), 0.85-0.79 (m, 3H);
31P NMR (162 MHz, d6-DMSO) 149.43, 149.18. Compound 2-4b, 2-4c, 2-4d, and 2-4e were prepared using similar procedures described above for compound 2-4a. Compound 2-4b was obtained (25.50 g, 85.4%) as a white solid:
1H NMR (400 MHz, d
6-DMSO) 11.23 (s, 1H), 8.65-8.60 (m, 2 H), 8.05-8.02 (m, 2H), 7.73- 7.70 (m, 1H), 7.67-7.60 (m, 1H), 7.59-7.51 (m, 2H), 7.38-7.34 (m, 2H), 7.30-7.25 (m, 7H), 6.89-6.80 (m, 4H), 6.21-6.15 (m, 1H), 5.23-5.17 (m, 1H), 4.80-4.69 (m, 3H), 4.40-4.21 (m, 2H), 3.91-3.80 (m, 1H), 3.74 (s, 6H), 3.74-3.52 (m, 3H), 3.50-3.20 (m, 6H), 3.14-3.09 (m, 2H), 3.09 (s, 1H), 2.83-2.79 (m, 1H), 2.68-2.62 (m, 1H), 2.05-1.97 (m, 2H), 1.50-1.38 (m, 2H), 1.31-1.10 (m, 18H), 1.08-1.05 (m, 2H), 0.85-0.78 (m, 3H);
31P NMR (162 MHz, d6- DMSO) 149.43, 149.19. Compound 2-4c was obtained (36.60 g, 66.3%) as an off-white solid:
1H NMR (400 MHz, d6- DMSO) 11.22 (s, 1H), 8.64-8.59 (m, 2H), 8.05-8.00 (m, 2H), 7.73-7.70 (m, 1H), 7.67-7.60
FH12501620.1
Attorney Docket: DCY-13025 (m, 1H), 7.59-7.51 (m, 2H), 7.38-7.34 (m, 2H), 7.30-7.25 (m, 7H), 6.89-6.80 (m, 4H), 6.21- 6.15 (m, 1H), 5.25-5.17 (m, 1H), 4.80-4.69 (m, 3H), 4.40-4.21 (m, 2H), 3.91-3.80 (m, 1H), 3.74 (s, 6H), 3.74-3.50 (m, 3H), 3.50-3.20 (m, 6H), 3.14-3.09 (m, 2H), 3.09 (s, 1H), 2.83- 2.79 (m, 1H), 2.68-2.62 (m, 1H), 2.05-1.99 (m, 2H), 1.50-1.38 (m, 2H), 1.33-1.12 (m, 38H), 1.08-1.05 (m, 2 H), 0.86-0.80 (m, 3H);
31P NMR (162 MHz, d6-DMSO) 149.42, 149.17. Compound 2-4d was obtained (26.60 g, 72.9%) as an off-white solid:
1H NMR (400 MHz, d6-DMSO) 11.22 (s, 1H), 8.64-8.59 (m, 2H), 8.05-8.00 (m, 2H), 7.73-7.70 (m, 1H), 7.67-7.60 (m, 1H), 7.59-7.51 (m, 2H), 7.38-7.33 (m, 2H), 7.30-7.25 (m, 7H), 6.89-6.80 (m, 4H), 6.21- 6.15 (m, 1H), 5.22-5.17 (m, 1H), 4.80-4.69 (m, 3H), 4.40-4.21 (m, 2H), 3.91-3.80 (m, 1H), 3.74 (s, 6H), 3.74-3.52 (m, 3H), 3.50-3.20 (m, 6H), 3.14-3.09 (m, 2H), 3.09 (s, 1H), 2.83- 2.79 (m, 1H), 2.68-2.62 (m, 1H), 2.05-1.99 (m, 2H), 1.50-1.38 (m, 2H), 1.35-1.08 (m, 38H), 1.08-1.05 (m, 2 H), 0.85-0.79 (m, 3H);
31P NMR (162 MHz, d6-DMSO) 149.47, 149.22. Compound 2-4e was obtained (38.10 g, 54.0%) as a white solid:
1H NMR (400 MHz, d
6- DMSO) 11.21 (s, 1H), 8.64-8.59 (m, 2H), 8.05-8.00 (m, 2H), 7.73-7.70 (m, 1H), 7.67-7.60 (m, 1H), 7.59-7.51 (m, 2H), 7.38-7.34 (m, 2H), 7.30-7.25 (m, 7H), 6.89-6.80 (m, 4H), 6.21- 6.15 (m, 1H), 5.23-5.17 (m, 1H), 4.80-4.69 (m, 3H), 4.40-4.21 (m, 2H), 3.91-3.80 (m, 1H), 3.73 (s, 6H), 3.74-3.52 (m, 3H), 3.47-3.22 (m, 6H), 3.14-3.09 (m, 2H), 3.09 (s, 1H), 2.83- 2.79 (m, 1H), 2.68-2.62 (m, 1H), 2.05-1.99 (m, 2H), 1.50-1.38 (m, 2H), 1.35-1.06 (m, 46H), 1.08-1.06 (m, 2 H), 0.85-0.77 (m, 3H);
31P NMR (162 MHz, d6-DMSO) 149.41, 149.15. Lipid amidites with other bases (U, G, C) were prepared via similar procedures. Example 2. Synthesis of GalXC RNAi Oligonucleotide-Lipid-COOH Conjugates Scheme 1. Synthesis of GalXC RNAi oligonucleotide-lipid-COOH conjugates with mono- lipid (linear and branched) or di-lipid (linear and branched) conjugates were realized through post-solid phase synthetic conjugation Synthesis of Duplex A:
FH12501620.1
Attorney Docket: DCY-13025

Sense A was prepared by solid-phase synthesis using a commercial oligo synthesizer. The oligonucleotides were synthesized using 2’-modified nucleoside phosphoramidites, such as 2'-F or 2'-OMe, and 2'-diethoxymethanol linked Docosanoic acid (C22) fatty acid amide nucleoside phosphoramidites. Oligonucleotide synthesis was conducted on a solid support in the 3' to 5' direction using a standard oligonucleotide synthesis protocol. In these efforts, 5- ethylthio-1H-tetrazole (ETT) was used as an activator for the coupling reaction. Iodine solution was used for phosphite triester oxidation.3-(Dimethylaminomethylidene)amino-3H- 1,2,4-dithiazole-3-thione (DDTT) was used for the formation of phosphorothioate linkages. Synthesized oligonucleotides were treated with concentrated aqueous ammonium for 10 h. The ammonia was removed from the suspension and the solid support residues were removed by filtration. The crude oligonucleotide was treated with TEAA, analyzed, and purified by strong anion exchange high performance liquid chromatography (SAX- HPLC). The fractions were combined and dialyzed against water (3 X), saline (1 X), and water (3 X) using Amicon® Ultra-15 Centrifugal (3K). The remaining solvent was then lyophilized to afford the desired Conjugated Sense A (LCMS found mass 12502). Annealing of Duplex A. Conjugated Sense A (5 mg, measured by weight) was dissolved in 500 µL deionized water to prepare a 10 mg/mL solution. Antisense A (10 mg, measured by OD) was dissolved in 1 mL deionized water to prepare a 10 mg/mL solution, which was used for the titration of the conjugated sense and quantification of the duplex amount. Based on the calculation of molar amounts of both conjugated sense and antisense, a proportion of required Antisense A was added to the Conjugated Sense A solution. The resulting mixture was stirred at 95 °C for 5
FH12501620.1
Attorney Docket: DCY-13025 min and allowed to cool down to room temperature. The annealing progress was monitored by ion-exchange HPLC. Based on the annealing progress, several proportions of Antisense A were further added to complete the annealing with >95% purity. The solution was lyophilized to afford Duplex A (C22) and its amount was calculated based on the molar amount of the antisense consumed in the annealing. Antisense A was prepared by solid-phase synthesis (LCMS found mass ^^^^). Synthesis of Duplex B:

Conjugated Sense B was synthesized through a two-step post solid phase conjugation approach. Step 1- Conjugation of Azido-PEG4 Handle: In Eppendorf tube 1, a solution of Azido-PEG4-NHS ester (4.25 mg, 0.011 mmol) in 562 ^L DMF was prepared at rt. In Eppendorf tube 2, oligo Sense B1 (22.5 mg, 0.0018 mmol) was dissolved in 562 ^L water and treated with DIPEA (1.95 ^L, 0.011 mmol). Next, the solution in Eppendorf tube 1 was added to the Eppendorf tube 2 and mixed using Thermomixer at 35 ˚C. After 1 hour, the reaction was indicated complete by LC-MS analysis. The reaction
FH12501620.1
Attorney Docket: DCY-13025 mixture was diluted with 10 mL of water and then dialyzed against water (2X) using Amicon® Ultra-15 Centrifugal (10K). The Amicon membrane was washed with water (3 X 2 mL) and the combined solvents were then lyophilized to afford an amorphous white solid of Conjugated Sense B2 (22 mg, yield: 98%). Step 2- Conjugation to Azido-PEG4 Handle: In Eppendorf tube 1, a 1:1 mixture of water and DMA (360 ^L) was degassed under nitrogen gas for 10 minutes. Next 20 mg (1 eq, 0.0016 mmol) of Conjugated Sense B2 and 1.7 ^L DIPEA (6eq, 0.0096 mmol) were added to the mixture of water and DMA in Eppendorf tube 1, followed by addition of 2.682 mg 17-Octadecynoic acid (6 eq, 0.0096 mmol) to Eppendorf tube 1. The resulting mixture was kept under Nitrogen gas for another 10 minutes to remove any dissolved oxygen. In Eppendorf tube 2, 2.631 mg CuBr.SCH3 (8 eq, 0.0128) was dissolved in 240 ^L of previously degassed acetonitrile. Next, the solution in Eppendorf tube 2 was quickly added to Eppendorf tube 1 and mixed using Thermomixer at 35 °C for 3 hours. After the reaction was indicated complete by LC-MS analysis, the reaction mixture was diluted with 15 mL EDTA 0.5 M (pH 8) in a 50mL falcon tube and stirred for 15 minutes at room temperature. Mixture was then dialyzed against water (2X) using Amicon® Ultra-15 Centrifugal (3K). The combined solvents were purified by revers phase XBridge C18 column using a 5-95% gradient of 100 mM TEAA in ACN and H
2O. The product fractions were concentrated under reduced pressure using Genevac. The combined residual solvent was dialyzed against water (1 X), saline (1 X), and water (3 X) using Amicon® Ultra-15 Centrifugal (3K). The Amicon membrane was washed with water (3 X 2 mL) and the combined solvents were then lyophilized to afford an amorphous white solid of Conjugated Sense B (7.28 mg, yield: 36.4%) (LCMS found mass 12734). Sense B1 and Antisense A were prepared by solid-phase synthesis. Duplex B was prepared using the same procedures as described for the annealing of Duplex A. Synthesis of Duplex C:
FH12501620.1
Attorney Docket: DCY-13025

Conjugated Sense C was synthesized through a two-step post solid phase conjugation approach similar to Conjugated Sense B. Step 1- Attachment of Azido-PEG4 Handle: In Eppendorf tube 1, a solution of Azido-PEG4-NHS ester (8.5 mg, 0.022 mmol) in 1125 ^L DMF was prepared at rt. In Eppendorf tube 2, oligo Sense C1 (45 mg, 0.0036 mmol) was dissolved in 1125 ^L water and treated with DIPEA (3.9 ^L, 0.022 mmol). Next, the solution in Eppendorf tube 1 was added to the Eppendorf tube 2 and mixed using Thermomixer at 35 ˚C. After 1 hour, the reaction was indicated complete by LC-MS analysis. The reaction mixture was diluted with 10 mL of water and then dialyzed against water (2X) using Amicon® Ultra-15 Centrifugal (10K). The Amicon membrane was washed with water (3 X 2 mL) and the combined solvents were then lyophilized to afford an amorphous white solid of Conjugated Sense C2 (44.55 mg, yield: 99%). Step 2- Conjugation to Azido-PEG4 Handle: In Eppendorf tube 1, a 1:1 mixture of water and DMA (360 ^L) was degassed under nitrogen gas for 10 minutes. Next 20 mg (1 eq, 0.0016 mmol) of Conjugated Sense C2 and 1.7 ^L DIPEA (6eq, 0.0096 mmol) were added to the mixture of water and DMA in Eppendorf tube
FH12501620.1
Attorney Docket: DCY-13025 1, followed by addition of 2.682 mg 17-Octadecynoic acid (6 eq, 0.0096 mmol) to Eppendorf tube 1. The resulting mixture was kept under Nitrogen gas for another 10 minutes to remove any dissolved oxygen. In Eppendorf tube 2, 2.631 mg CuBr.SCH
3 (8 eq, 0.0128) was dissolved in 240 ^L of previously degassed acetonitrile. Next, the solution in Eppendorf tube 2 was quickly added to Eppendorf tube 1 and mixed using Thermomixer at 35 °C for 3 hours. After the reaction was indicated complete by LC-MS analysis, the reaction mixture was diluted with 15 mL EDTA 0.5 M (pH 8) in a 50mL falcon tube and stirred for 15 minutes at room temperature. Mixture was then dialyzed against water (2X) using Amicon® Ultra-15 Centrifugal (3K). The combined solvents were purified by reverse phase XBridge C18 column using a 5-95% gradient of 100 mM TEAA in ACN and H2O. The product fractions were concentrated under reduced pressure using Genevac. The combined residual solvent was dialyzed against water (1 X), saline (1 X), and water (3 X) using Amicon® Ultra-15 Centrifugal (3K). The Amicon membrane was washed with water (3 X 2 mL) and the combined solvents were then lyophilized to afford an amorphous white solid of Conjugated Sense C (5.28 mg, 26.4% yield) (LCMS found mass 12809). Sense C1 and Antisense A were prepared by solid-phase synthesis. Duplex C was prepared using the same procedures as described for the annealing of Duplex A. Synthesis of Duplex D:

Conjugated Sense D was synthesized through a two-step post solid phase conjugation approach similar to Conjugated Sense B and C.
FH12501620.1
Attorney Docket: DCY-13025 Step 1- Attachment of Azido-PEG4 Handle: In Eppendorf tube 1, a solution of Azido- PEG4-NHS ester (8.5 mg, 0.022 mmol) in 1125 ^L DMF was prepared at rt. In Eppendorf tube 2, oligo Sense D1 (45 mg, 0.0036 mmol) was dissolved in 1125 ^L water and treated with DIPEA (3.9 ^L, 0.022 mmol). Next, the solution in Eppendorf tube 1 was added to the Eppendorf tube 2 and mixed using Thermomixer at 35 ˚C. After 1 hour, the reaction was indicated complete by LC-MS analysis. The reaction mixture was diluted with 10 mL of water and then dialyzed against water (2X) using Amicon® Ultra-15 Centrifugal (10K). The Amicon membrane was washed with water (3 X 2 mL) and the combined solvents were then lyophilized to afford an amorphous white solid of Conjugated Sense D2 (44.55 mg, yield: 99%). Step 2- Conjugation to Azido-PEG4 Handle: In Eppendorf tube 1, a 1:1 mixture of water and DMA (360 ^L) was degassed under nitrogen gas for 10 minutes. Next 20 mg (1 eq, 0.0016 mmol) of Conjugated Sense D2 and 1.7 ^L DIPEA (6eq, 0.0096 mmol) were added to the mixture of water and DMA in Eppendorf tube 1, followed by addition of 2.682 mg 17-Octadecynoic acid (6 eq, 0.0096 mmol) to Eppendorf tube 1. The resulting mixture was kept under Nitrogen gas for another 10 minutes to remove any dissolved oxygen. In Eppendorf tube 2, 2.631 mg CuBr.SCH
3 (8 eq, 0.0128) was dissolved in 240 ^L of previously degassed acetonitrile. Next, the solution in Eppendorf tube 2 was quickly added to Eppendorf tube 1 and mixed using Thermomixer at 35 °C for 3 hours. After the reaction was indicated complete by LC-MS analysis, the reaction mixture was diluted with 15 mL EDTA 0.5 M (pH 8) in a 50mL falcon tube and stirred for 15 minutes at room temperature. Mixture was then dialyzed against water (2X) using Amicon® Ultra-15 Centrifugal (3K). The combined solvents were purified by revers phase XBridge C18 column using a 5-95% gradient of 100 mM TEAA in ACN and H
2O. The product fractions were concentrated under reduced pressure using Genevac. The combined residual solvent was dialyzed against water (1 X), saline (1 X), and water (3 X) using Amicon® Ultra-15 Centrifugal (3K). The Amicon membrane was washed with water (3 X 2 mL) and the combined solvents were then lyophilized to afford an amorphous white solid of Conjugated Sense D (7.41 mg, yield: 37%) (LCMS found mass 13235). Sense D1 and Antisense A were prepared by solid-phase synthesis. Duplex D was prepared using the same procedures as described for the annealing of Duplex A.
FH12501620.1
Attorney Docket: DCY-13025
FH12501620.1
Attorney Docket: DCY-13025 Synthesis of Duplex E:
Conjugated Sense E was synthesized through a two-step post solid phase conjugation approach similar to Conjugated Sense C. Step 1- Attachment of Azido-PEG4 Handle: In Eppendorf tube 1, a solution of Azido- PEG4-NHS ester (8.5 mg, 0.022 mmol) in 1125 ^L DMF was prepared at rt. In Eppendorf tube 2, oligo Sense C1 (45 mg, 0.0036 mmol) was dissolved in 1125 ^L water and treated with DIPEA (3.9 ^L, 0.022 mmol). Next, the solution in Eppendorf tube 1 was added to the Eppendorf tube 2 and mixed using Thermomixer at 35 ˚C. After 1 hour, the reaction was indicated complete by LC-MS analysis. The reaction mixture was diluted with 10 mL of water and then dialyzed against water (2X) using Amicon® Ultra-15 Centrifugal (10K). The Amicon membrane was washed with water (3 X 2 mL) and the combined solvents were then lyophilized to afford an amorphous white solid of Conjugated Sense C2 (44.55 mg, yield: 99%). Step 2- Conjugation to Azido-PEG4 Handle:
FH12501620.1
Attorney Docket: DCY-13025 In Eppendorf tube 1, a 1:1 mixture of water and DMA (360 ^L) was degassed under nitrogen gas for 10 minutes. Next 20 mg (1 eq, 0.0016 mmol) of Conjugated Sense C2 and 1.7 ^L DIPEA (6eq, 0.0096 mmol) were added to the mixture of water and DMA in Eppendorf tube 1, followed by addition of 2.682 mg Propargyl-PEG3-acid (6 eq, 0.0096 mmol) to Eppendorf tube 1. The resulting mixture was kept under Nitrogen gas for another 10 minutes to remove any dissolved oxygen. In Eppendorf tube 2, 2.631 mg CuBr.SCH3 (8 eq, 0.0128) was dissolved in 240 ^L of previously degassed acetonitrile. Next, the solution in Eppendorf tube 2 was quickly added to Eppendorf tube 1 and mixed using Thermomixer at 35 °C for 3 hours. After the reaction was indicated complete by LC-MS analysis, the reaction mixture was diluted with 15 mL EDTA 0.5 M (pH 8) in a 50mL falcon tube and stirred for 15 minutes at room temperature. Mixture was then dialyzed against water (2X) using Amicon® Ultra-15 Centrifugal (3K). The combined solvents were purified by revers phase XBridge C18 column using a 5-95% gradient of 100 mM TEAA in ACN and H
2O. The product fractions were concentrated under reduced pressure using Genevac. The combined residual solvent was dialyzed against water (1 X), saline (1 X), and water (3 X) using Amicon® Ultra-15 Centrifugal (3K). The Amicon membrane was washed with water (3 X 2 mL) and the combined solvents were then lyophilized to afford an amorphous white solid of Conjugated Sense E (7.41 mg, yield: 37%) (LCMS found mass 12789). Sense C1 and Antisense A were prepared by solid-phase synthesis. Duplex E was prepared using the same procedures as described for the annealing of Duplex A. Synthesis of Duplex F and Duplex G:
FH12501620.1
Attorney Docket: DCY-13025 Conjugated Sense F and G were synthesized through post solid-phase conjugation approach substantially similar to Conjugated Sense E. Post-synthetic conjugation was realized through two step Amide reaction followed by Cu-catalyzed alkyne-azide cycloaddition reaction. 6.32 mg of Conjugated Sense F was obtained in 31.6% yield (LCMS found mass 12627). 12.56 mg of Conjugated Sense G was obtained in 62.8% yield (LCMS found mass 12779). Duplex F and Duplex G were prepared using the same procedures as described for the annealing of Duplex A. Synthesis of Duplex H:

Conjugated Sense H was synthesized through a one-step post-syntenic conjugation approach. In Eppendorf tube 1, a solution of Palmitic acid (1.5 mg, 0.0053 mmol) in DMA (495 µL) was treated with HATU ( 2.01 mg, 0.0053 mmol) at rt. In Eppendorf tube 2, a solution of oligo Sense C1 (13 mg, 0.00106 mmol) in H2O (55 µL) was treated with DIPEA (1.11 µL, 0.00636 mmol). The solution in Eppendorf tube 1 was added to the Eppendorf tube 2 and mixed using Thermomixer at rt. After the reaction was completed indicated by LC-MS analysis, the reaction mixture was diluted with 5 mL of water and purified by revers phase XBridge C18 column using a 5-95% gradient of 100 mM TEAA in ACN and H2O. The product fractions were concentrated under reduced pressure using Genevac. The combined residual solvent was dialyzed against water (1 X), saline (1 X), and water (3 X) using
FH12501620.1
Attorney Docket: DCY-13025 Amicon® Ultra-15 Centrifugal (3K). The Amicon membrane was washed with water (3 X 2 mL) and the combined solvents were then lyophilized to afford an amorphous white solid of Conjugated Sense H (5.047 mg, %39 yield) (LCMS found mass 12494). Sense C1 and Antisense A were prepared by solid-phase synthesis. Duplex H was annealed prepared using the same procedures as described for the annealing of Duplex A. Synthesis of Duplex I:

Conjugated Sense I was synthesized through a one-step post-syntenic Cu-catalyzed alkyne- azide cycloaddition reaction. In Eppendorf tube 1, a 1:1 mixture of water and DMA (360 ^L) was degassed under nitrogen gas for 10 minutes. Next 20 mg (1 eq, 0.0016 mmol) of Sense I1 and 1.7 ^L DIPEA (6eq, 0.0096 mmol) were added to the mixture of water and DMA in Eppendorf tube 1, followed by addition of 3.12 mg 18-Azido-stearic acid (6 eq, 0.0096 mmol) to Eppendorf tube 1. The resulting mixture was kept under Nitrogen gas for another 10 minutes to remove any dissolved oxygen. In Eppendorf tube 2, 2.631 mg CuBr.SCH3 (8 eq, 0.0128) was dissolved in 240 ^L of previously degassed acetonitrile. Next, the solution in Eppendorf tube 2 was quickly added to Eppendorf tube 1 and mixed using Thermomixer at 35 °C for 3 hours.
FH12501620.1
Attorney Docket: DCY-13025 After the reaction was indicated complete by LC-MS analysis, the reaction mixture was diluted with 15 mL EDTA 0.5 M (pH 8) in a 50mL falcon tube and stirred for 15 minutes at room temperature. Mixture was then dialyzed against water (2X) using Amicon® Ultra-15 Centrifugal (3K). The combined solvents were purified by revers phase XBridge C18 column using a 5-95% gradient of 100 mM TEAA in ACN and H2O. The product fractions were concentrated under reduced pressure using Genevac. The combined residual solvent was dialyzed against water (1 X), saline (1 X), and water (3 X) using Amicon® Ultra-15 Centrifugal (3K). The Amicon membrane was washed with water (3 X 2 mL) and the combined solvents were then lyophilized to afford an amorphous white solid of Conjugated Sense I (9.11 mg, yield: 45.5%) (LCMS found mass 12563). Sense I1 and Antisense A were prepared by solid-phase synthesis. Duplex H was annealed prepared using the same procedures as described for the annealing of Duplex A. Synthesis of Duplex J:

Conjugated Sense J was prepared by solid-phase synthesis using a commercial oligo synthesizer. The oligonucleotides were synthesized using 2’-modified nucleoside phosphoramidites, such as 2'-F or 2'-OMe, and 2'-diethoxymethanol linked Docosanoic acid (C22) fatty acid amide nucleoside phosphoramidites. Oligonucleotide synthesis was conducted on a solid support in the 3' to 5' direction using a standard oligonucleotide synthesis protocol. In these efforts, 5-ethylthio-1H-tetrazole (ETT) was used as an activator for the coupling reaction. Iodine solution was used for phosphite triester oxidation.3- (Dimethylaminomethylidene)amino-3H-1,2,4-dithiazole-3-thione (DDTT) was used for the F
H12501620.1
Attorney Docket: DCY-13025 formation of phosphorothioate linkages. Synthesized oligonucleotides were treated with concentrated aqueous ammonium for 10 h. The ammonia was removed from the suspension and the solid support residues were removed by filtration. The crude oligonucleotide was treated with TEAA, analyzed, and purified by strong anion exchange high performance liquid chromatography (SAX-HPLC). The fractions were combined and dialyzed against water (3 X), saline (1 X), and water (3 X) using Amicon® Ultra-15 Centrifugal (3K). The remaining solvent was then lyophilized to afford the desired Conjugated Sense J (LCMS found mass 7138). Sense J was annealed to Antisense B to yield Duplex J using the same procedures as described for the annealing of Duplex A. Antisense B was prepared by solid-phase synthesis (LCMS found mass ^^^^). Synthesis of Duplex K:

Conjugated Sense K was synthesized through a one-step post-syntenic conjugation approach. In Eppendorf tube 1, a solution of hexadecanedioic acid (2.51 mg, 0.00879 mmol) in DMA
FH12501620.1
Attorney Docket: DCY-13025 (800 µL) was treated with HATU (5.6 mg, 0.01465 mmol) at rt. In Eppendorf tube 2, a solution of oligo Sense K (20 mg, 0.00293 mmol) in 200 µL H
2O was treated with DIPEA (3 µL, 0.01758 mmol). The solution in Eppendorf tube 1 was added to the Eppendorf tube 2 and mixed using Thermomixer at rt. After the reaction was completed indicated by LC-MS analysis, the reaction mixture was diluted with 5 mL of water and purified by revers phase XBridge C18 column using a 5-95% gradient of 100 mM TEAA in ACN and H2O. The product fractions were concentrated under reduced pressure using Genevac. The combined residual solvent was dialyzed against water (1 X), saline (1 X), and water (3 X) using Amicon® Ultra-15 Centrifugal (3K). The Amicon membrane was washed with water (3 X 2 mL) and the combined solvents were then lyophilized to afford an amorphous white solid of Conjugated Sense K (15.14 mg, 75.7% yield) (LCMS found mass 7084). Sense K was annealed to Antisense B to yield Duplex K using the same procedures as described for the annealing of Duplex A. Synthesis of Duplex L:

Conjugated Sense L was synthesized through a one-step post-syntenic conjugation approach substantially similar to Conjugated Sense K. 5.02 mg of Conjugated Sense L was obtained in 24% yield (LCMS found mass 7168). Sense L was annealed to Antisense B to yield Duplex L using the same procedures as described for the annealing of Duplex A. Synthesis of Duplex M:
FH12501620.1
Attorney Docket: DCY-13025

Conjugated Sense M was synthesized through a two-step post-syntenic conjugation approach. Step 1- Attachment of Amino-PEG4 Handle: In Eppendorf tube 1, a solution of Fmoc-N-amido-PEG4-NHS Ester (5.144 mg, 0.00880 mmol) was dissolved in DMA (500µL) and treated with DIPEA (1.531 µL, 0.00879 mmol). In Eppendorf tube 2, a solution of oligo Sense K1 (20 mg, 0.00293 mmol) in 500 µL H2O was treated with DIPEA (1.531 µL, 0.00879 mmol). The solution in Eppendorf tube 1 was added to the Eppendorf tube 2 and mixed for 1 hour using Thermomixer at 40°C. After the reaction was completed indicated by LC-MS analysis, the crude was then dialyzed against water (2 X) using Pierce Thermo Fisher 3k filters to remove excess of Fmoc-N-amido-PEG4-NHS Ester. The crude was then removed from filters and deprotected using water and Piperidine mixture (4:1) (800 µL water: 200 µL Piperidine) and mixed for 20 minutes using Thermomixer at rt. The reaction was then diluted with 5 mL of water and purified by reverse phase XBridge C18 column using a 5-95% gradient of 100 mM TEAA in ACN and H
2O. The product fractions were concentrated under reduced pressure using Genevac. The combined residual solvent was dialyzed against water (2 X), saline (2 X), and water (2 X) using Pierce Thermo Fisher 3k filters. The filter membrane was washed with water (3 X 2 mL) and with a last wash of 20%
FH12501620.1
Attorney Docket: DCY-13025 ethanol in water (20 µL). The combined solvents were then lyophilized to afford an amorphous white solid of Conjugated
Sense M1 (15.14 mg, 75.7% yield). Step 2- Conjugation to Amino-PEG4 Handle In Eppendorf tube 1, a solution of hexadecanedioic acid (6.137 mg, 0.02143 mmol) was dissolved in DMA (450 µL) and treated with HATU (8.148 mg, 0.002143 mmol) and DIPEA (1.9 µL, 0.00107 mmol) at rt. In Eppendorf tube 2, a solution of Conjugated Sense M1 (15.14mg, 0.002143 mmol) in 50 µL H2O was treated with DIPEA (1.9 µL, 0.00107 mmol). The solution in Eppendorf tube 1 was then added to the Eppendorf tube 2 and mixed for 30 minutes using Thermomixer at 40°C. After the reaction was completed indicated by LC-MS analysis, the reaction mixture was diluted with 5 mL of water and purified by revers phase XBridge C18 column using a 5-95% gradient of 100 mM TEAA in ACN and H2O. The product fractions were concentrated under reduced pressure using Genevac. The combined residual solvent was dialyzed against water (2 X), saline (2 X), and water (2 X) using Pierce Thermo Fisher 3k filters. The Amicon membrane was washed with water (3 X 2 mL) and with a last wash of 20% ethanol in water (20 µL). The combined solvents were then lyophilized to afford an amorphous white solid of Conjugated Sense M (5.3 mg, 35% yield) (LCMS found mass 7331). Sense M was annealed to Antisense B to yield Duplex M using the same procedures as described for the annealing of Duplex A. Synthesis of Duplex N:

Conjugated Sense N was synthesized through a two-step post-syntenic conjugation approach substantially similar to Conjugated Sense M. 2 mg of Conjugated Sense N was obtained in 10% yield (LCMS found mass 7683). Sense N was annealed to Antisense B to yield Duplex N using the same procedures as described for the annealing of Duplex A.
FH12501620.1
Attorney Docket: DCY-13025 Synthesis of Duplex O:
Conjugated Sense O was synthesized through a two-step post-syntenic conjugation approach. Step 1- Attachment of Azide Handle: In Eppendorf tube 1, a solution of 3-azidopropanoic acid (1.4 mg, 0.015 mmol) in DMA (500 µL) was treated with HATU ( 5.6 mg, 0.015 mmol) at rt. In Eppendorf tube 2, oligo Sense K1 (20 mg, 0.003 mmol) was dissolved in 500 ^L water and treated with DIPEA (3 ^L, 0.018 mmol). Next, the solution in Eppendorf tube 1 was added to the Eppendorf tube 2 and
FH12501620.1
Attorney Docket: DCY-13025 mixed using Thermomixer at 35 ˚C. After 1 hour, the reaction was indicated complete by LC- MS analysis. The reaction mixture was diluted with 10 mL of water and then dialyzed against water (2X) using Amicon® Ultra-15 Centrifugal (10K). The Amicon membrane was washed with water (3 X 2 mL) and the combined solvents were then lyophilized to afford an amorphous white solid of Conjugated Sense O1 (14 mg, yield: 70%). Step 2- Conjugation to Azide Handle: In Eppendorf tube 1, a 1:1 mixture of water and DMA (200 ^L) was degassed under nitrogen gas for 10 minutes. Next 14 mg (1 eq, 0.0020 mmol) of Conjugated Sense O1 and 2 ^L DIPEA (0.012mmol) were added to the mixture of water and DMA in Eppendorf tube 1, followed by addition of 3.6 mg 17-Octadecynoic acid (6 eq, 0.0096 mmol) to Eppendorf tube 1. The resulting mixture was kept under Nitrogen gas for another 10 minutes to remove any dissolved oxygen. In Eppendorf tube 2, 2.631 mg CuBr.SCH3 (8 eq, 0.0122 mmol) was dissolved in 200 ^L of previously degassed acetonitrile. Next, the solution in Eppendorf tube 2 was quickly added to Eppendorf tube 1 and mixed using Thermomixer at 35 °C for 3 hours. After the reaction was indicated complete by LC-MS analysis, the reaction mixture was diluted with 15 mL EDTA 0.5 M (pH 8) in a 50mL falcon tube and stirred for 15 minutes at room temperature. Mixture was then dialyzed against water (2X) using Amicon® Ultra-15 Centrifugal (3K). The combined solvents were purified by revers phase XBridge C18 column using a 5-95% gradient of 100 mM TEAA in ACN and H2O. The product fractions were concentrated under reduced pressure using Genevac. The combined residual solvent was dialyzed against water (1 X), saline (1 X), and water (3 X) using Amicon® Ultra-15 Centrifugal (3K). The Amicon membrane was washed with water (3 X 2 mL) and the combined solvents were then lyophilized to afford an amorphous white solid of Conjugated Sense O (1.01 mg, yield: 4.8%) (LCMS found mass 7193). Sense O was annealed to Antisense B to yield Duplex O using the same procedures as described for the annealing of Duplex A.
FH12501620.1
Attorney Docket: DCY-13025 Synthesis of Duplex P:
Conjugated Sense P was synthesized through a two-step post-syntenic conjugation approach. Step 1- Attachment of Azido-PEG4 Handle: In Eppendorf tube 1, a solution of Azido-PEG4-NHS ester (6.8 mg, 0.017604 mmol) in 500 ^L DMF was prepared at rt. In Eppendorf tube 2, oligo Sense K1 (20 mg, 0.002934 mmol) was dissolved in 500 ^L water and treated with DIPEA (3 ^L, 0.017604 mmol). Next, the solution in Eppendorf tube 1 was added to the Eppendorf tube 2 and mixed using Thermomixer at 35 ˚C. After 1 hour, the reaction was indicated complete by LC-MS analysis. The reaction mixture was diluted with 10 mL of water and then dialyzed against water (2X) using Amicon® Ultra-15 Centrifugal (10K). The Amicon membrane was washed with water (3 X 2 mL) and the combined solvents were then lyophilized to afford an amorphous white solid of Conjugated Sense P1 (20 mg, yield: 100%). Step 2- Conjugation to Azido-PEG4 Handle:
FH12501620.1
Attorney Docket: DCY-13025 In Eppendorf tube 1, a 1:1 mixture of water and DMA (360 ^L) was degassed under nitrogen gas for 10 minutes. Next 20 mg (1 eq, 0.00282 mmol) of Conjugated Sense P1 and 3 ^L DIPEA (6eq, 0.0169 mmol) were added to the mixture of water and DMA in Eppendorf tube 1, followed by addition of 4.7 mg 17-Octadecynoic acid (6 eq, 0.0096 mmol) to Eppendorf tube 1. The resulting mixture was kept under Nitrogen gas for another 10 minutes to remove any dissolved oxygen. In Eppendorf tube 2, 4.6 mg CuBr.SCH3 (8 eq, 0.02257 mmol) was dissolved in 240 ^L of previously degassed acetonitrile. Next, the solution in Eppendorf tube 2 was quickly added to Eppendorf tube 1 and mixed using Thermomixer at 35 °C for 3 hours. After the reaction was indicated complete by LC-MS analysis, the reaction mixture was diluted with 15 mL EDTA 0.5 M (pH 8) in a 50mL falcon tube and stirred for 15 minutes at room temperature. Mixture was then dialyzed against water (2X) using Amicon® Ultra-15 Centrifugal (3K). The combined solvents were purified by revers phase XBridge C18 column using a 5-95% gradient of 100 mM TEAA in ACN and H
2O. The product fractions were concentrated under reduced pressure using Genevac. The combined residual solvent was dialyzed against water (1 X), saline (1 X), and water (3 X) using Amicon® Ultra-15 Centrifugal (3K). The Amicon membrane was washed with water (3 X 2 mL) and the combined solvents were then lyophilized to afford an amorphous white solid of Conjugated Sense P (2.2 mg, yield: 10%) (LCMS found mass 7369). Sense P was annealed to Antisense B to yield Duplex P using the same procedures as described for the annealing of Duplex A.
FH12501620.1
Attorney Docket: DCY-13025 Synthesis of Duplex Q:
Conjugated Sense Q was synthesized through a two-step post-syntenic conjugation approach. Step 1- Attachment of Azido-PEG4 Handle: In Eppendorf tube 1, a solution of Azido-PEG4-NHS ester (6.8 mg, 0.017604 mmol) in 500 ^L DMF was prepared at rt. In Eppendorf tube 2, oligo Sense Q1 (20 mg, 0.002934 mmol) was dissolved in 500 ^L water and treated with DIPEA (3 ^L, 0.017604 mmol). Next, the solution in Eppendorf tube 1 was added to the Eppendorf tube 2 and mixed using Thermomixer at 35 ˚C. After 1 hour, the reaction was indicated complete by LC-MS analysis. The reaction mixture was diluted with 10 mL of water and then dialyzed against water (2X) using Amicon® Ultra-15 Centrifugal (10K). The Amicon membrane was washed with water (3 X 2 mL) and the combined solvents were then lyophilized to afford an amorphous white solid of Conjugated Sense Q2 (20 mg, yield: 99%). Step 2- Conjugation to Azido-PEG4 Handle:
FH12501620.1
Attorney Docket: DCY-13025 In Eppendorf tube 1, a 1:1 mixture of water and DMA (360 ^L) was degassed under nitrogen gas for 10 minutes. Next 20 mg (1 eq, 0.00282 mmol) of Conjugated Sense Q2 and 3 ^L DIPEA (6eq, 0.0169 mmol) were added to the mixture of water and DMA in Eppendorf tube 1, followed by addition of 4.7 mg 17-Octadecynoic acid (6 eq, 0.0096 mmol) to Eppendorf tube 1. The resulting mixture was kept under Nitrogen gas for another 10 minutes to remove any dissolved oxygen. In Eppendorf tube 2, 4.6 mg CuBr.SCH3 (8 eq, 0.02257 mmol) was dissolved in 240 ^L of previously degassed acetonitrile. Next, the solution in Eppendorf tube 2 was quickly added to Eppendorf tube 1 and mixed using Thermomixer at 35 °C for 3 hours. After the reaction was indicated complete by LC-MS analysis, the reaction mixture was diluted with 15 mL EDTA 0.5 M (pH 8) in a 50mL falcon tube and stirred for 15 minutes at room temperature. Mixture was then dialyzed against water (2X) using Amicon® Ultra-15 Centrifugal (3K). The combined solvents were purified by revers phase XBridge C18 column using a 5-95% gradient of 100 mM TEAA in ACN and H
2O. The product fractions were concentrated under reduced pressure using Genevac. The combined residual solvent was dialyzed against water (1 X), saline (1 X), and water (3 X) using Amicon® Ultra-15 Centrifugal (3K). The Amicon membrane was washed with water (3 X 2 mL) and the combined solvents were then lyophilized to afford an amorphous white solid of Conjugated Sense Q (1 mg, yield: 5%) (LCMS found mass 7445). Sense Q was annealed to Antisense B to yield Duplex Q using the same procedures as described for the annealing of Duplex A.
FH12501620.1
Attorney Docket: DCY-13025 Synthesis of Duplex R:
Conjugated Sense R was synthesized through a two-step post-syntenic conjugation approach. Step 1- Attachment of Azido-PEG4 Handle: In Eppendorf tube 1, a solution of Azido-PEG4-NHS ester (6.8 mg, 0.017604 mmol) in 500 ^L DMF was prepared at rt. In Eppendorf tube 2, Sense K1 (20 mg, 0.002934 mmol) was dissolved in 500 ^L water and treated with DIPEA (3 ^L, 0.017604 mmol). Next, the solution in Eppendorf tube 1 was added to the Eppendorf tube 2 and mixed using Thermomixer at 35 ˚C. After 1 hour, the reaction was indicated complete by LC-MS analysis. The reaction mixture was diluted with 10 mL of water and then dialyzed against water (2X) using Amicon® Ultra- 15 Centrifugal (10K). The Amicon membrane was washed with water (3 X 2 mL) and the combined solvents were then lyophilized to afford an amorphous white solid of Conjugated Sense P1 (20 mg, yield: 100%). Step 2- Conjugation to Azido-PEG4 Handle:
FH12501620.1
Attorney Docket: DCY-13025 In Eppendorf tube 1, a 1:1 mixture of water and DMA (360 ^L) was degassed under nitrogen gas for 10 minutes. Next 20 mg (1 eq, 0.00282 mmol) of Conjugated Sense P1 and 3 ^L DIPEA (6eq, 0.0169 mmol) were added to the mixture of water and DMA in Eppendorf tube 1, followed by addition of 4.7 mg Octadec-1-yne (6 eq, 0.0096 mmol) to Eppendorf tube 1. The resulting mixture was kept under Nitrogen gas for another 10 minutes to remove any dissolved oxygen. In Eppendorf tube 2, 4.6 mg CuBr.SCH3 (8 eq, 0.02257 mmol) was dissolved in 240 ^L of previously degassed acetonitrile. Next, the solution in Eppendorf tube 2 was quickly added to Eppendorf tube 1 and mixed using Thermomixer at 35 °C for 3 hours. After the reaction was indicated complete by LC-MS analysis, the reaction mixture was diluted with 15 mL EDTA 0.5 M (pH 8) in a 50mL falcon tube and stirred for 15 minutes at room temperature. Mixture was then dialyzed against water (2X) using Amicon® Ultra-15 Centrifugal (3K). The combined solvents were purified by revers phase XBridge C18 column using a 5-95% gradient of 100 mM TEAA in ACN and H
2O. The product fractions were concentrated under reduced pressure using Genevac. The combined residual solvent was dialyzed against water (1 X), saline (1 X), and water (3 X) using Amicon® Ultra-15 Centrifugal (3K). The Amicon membrane was washed with water (3 X 2 mL) and the combined solvents were then lyophilized to afford an amorphous white solid of Conjugated Sense R (2.2 mg, yield: 10%) (LCMS found mass 7331). Sense R was annealed to Antisense B to yield Duplex R using the same procedures as described for the annealing of Duplex A. Synthesis of Duplex S:
FH12501620.1
Attorney Docket: DCY-13025

Conjugated Sense S was synthesized through a two-step post-syntenic conjugation approach. Step 1- Attachment of Azido-PEG4 Handle: In Eppendorf tube 1, a solution of Azido-PEG4-NHS ester (12.7 mg, 0.03252 mmol) in 375 ^L DMF was prepared at rt. In Eppendorf tube 2, Sense S1 (15 mg, 0.002168 mmol) was dissolved in 375 ^L water and treated with DIPEA (6 ^L, 0.0325 mmol). Next, the solution in Eppendorf tube 1 was added to the Eppendorf tube 2 and mixed using Thermomixer at 35 ˚C. After 1 hour, the reaction was indicated complete by LC-MS analysis. The reaction mixture was diluted with 10 mL of water and then dialyzed against water (2X) using Amicon® Ultra- 15 Centrifugal (10K). The Amicon membrane was washed with water (3 X 2 mL) and the combined solvents were then lyophilized to afford an amorphous white solid of Conjugated Sense S2 (12.5 mg, yield: 84%). Step 2- Conjugation to Azido-PEG4 Handle: In Eppendorf tube 1, a 1:4 mixture of water and DMA (750 ^L) was degassed under nitrogen gas for 10 minutes. Next 12.5 mg (1 eq, 0.001674mmol) of Conjugated Sense S2 and 4.4 ^L
FH12501620.1
Attorney Docket: DCY-13025 DIPEA (6eq, 0.02512 mmol) were added to the mixture of water and DMA in Eppendorf tube 1, followed by addition of 7 mg Octadec-1-yne (6 eq, 0.02512 mmol) to Eppendorf tube 1. The resulting mixture was kept under Nitrogen gas for another 10 minutes to remove any dissolved oxygen. In Eppendorf tube 2, 6.8 mg CuBr.SCH3 (8 eq, 0.03348 mmol) was dissolved in 600 ^L of previously degassed acetonitrile. Next, the solution in Eppendorf tube 2 was quickly added to Eppendorf tube 1 and mixed using Thermomixer at 35 °C for 3 hours. After the reaction was indicated complete by LC-MS analysis, the reaction mixture was diluted with 15 mL EDTA 0.5 M (pH 8) in a 50mL falcon tube and stirred for 15 minutes at room temperature. Mixture was then dialyzed against water (2X) using Amicon® Ultra-15 Centrifugal (3K). The combined solvents were purified by revers phase XBridge C18 column using a 5-95% gradient of 100 mM TEAA in ACN and H2O. The product fractions were concentrated under reduced pressure using Genevac. The combined residual solvent was dialyzed against water (1 X), saline (1 X), and water (3 X) using Amicon® Ultra-15 Centrifugal (3K). The Amicon membrane was washed with water (3 X 2 mL) and the combined solvents were then lyophilized to afford an amorphous white solid of Conjugated Sense S (4.49 mg, 25.8% yield) (LCMS found mass 7966). Sense S was annealed to Antisense B to yield Duplex S using the same procedures as described for the annealing of Duplex A. Synthesis of Duplex T:

Conjugated Sense T was synthesized through a two-step post-syntenic conjugation approach substantially similar to Conjugated Sense S. 8.41 mg of Conjugated Sense T was obtained in 48.4% yield (LCMS found mass 8026). Sense T was annealed to Antisense B to yield Duplex T using the same procedures as described for the annealing of Duplex A. Synthesis of Duplex U:
FH12501620.1
Attorney Docket: DCY-13025
Conjugated Sense U was synthesized utilizing a two-step conjugation approach. Step 1- Attachment of Azido-PEG4 Handle: In Eppendorf tube 1, a solution of Azido-PEG4-NHS ester (12.7 mg, 0.03252 mmol) in 375 ^L DMF was prepared at rt. In Eppendorf tube 2, Sense U1 (15 mg, 0.002168 mmol) was dissolved in 375 ^L water and treated with DIPEA (6 ^L, 0.0325 mmol). Next, the solution in Eppendorf tube 1 was added to the Eppendorf tube 2 and mixed using Thermomixer at 35 ˚C. After 1 hour, the reaction was indicated complete by LC-MS analysis. The reaction mixture was diluted with 10 mL of water and then dialyzed against water (2X) using Amicon® Ultra- 15 Centrifugal (10K). The Amicon membrane was washed with water (3 X 2 mL) and the
FH12501620.1
Attorney Docket: DCY-13025 combined solvents were then lyophilized to afford an amorphous white solid of Conjugated Sense U2 (12.5 mg, yield: 84%). Step 2- Conjugation to Azido-PEG4 Handle: In Eppendorf tube 1, a 1:4 mixture of water and DMA (750 ^L) was degassed under nitrogen gas for 10 minutes. Next 12.5 mg (1 eq, 0.001674mmol) of Conjugated Sense U2 and 4.4 ^L DIPEA (6eq, 0.02512 mmol) were added to the mixture of water and DMA in Eppendorf tube 1, followed by addition of 7.31 mg Octadec-1-yne (0.0292 mmol) to Eppendorf tube 1. The resulting mixture was kept under Nitrogen gas for another 10 minutes to remove any dissolved oxygen. In Eppendorf tube 2, 6.8 mg CuBr.SCH3 (8 eq, 0.03348 mmol) was dissolved in 600 ^L of previously degassed acetonitrile. Next, the solution in Eppendorf tube 2 was quickly added to Eppendorf tube 1 and mixed using Thermomixer at 35 °C for 3 hours. After the reaction was indicated complete by LC-MS analysis, the reaction mixture was diluted with 15 mL EDTA 0.5 M (pH 8) in a 50mL falcon tube and stirred for 15 minutes at room temperature. Mixture was then dialyzed against water (2X) using Amicon® Ultra-15 Centrifugal (3K). The combined solvents were purified by revers phase XBridge C18 column using a 5-95% gradient of 100 mM TEAA in ACN and H
2O. The product fractions were concentrated under reduced pressure using Genevac. The combined residual solvent was dialyzed against water (1 X), saline (1 X), and water (3 X) using Amicon® Ultra-15 Centrifugal (3K). The Amicon membrane was washed with water (3 X 2 mL) and the combined solvents were then lyophilized to afford an amorphous white solid of Conjugated Sense U (2.72 mg, yield: 23%) (LCMS found mass 7966). Sense U was annealed to Antisense B to yield Duplex U using the same procedures as described for the annealing of Duplex A. Synthesis of Duplex V:
FH12501620.1
Attorney Docket: DCY-13025 Conjugated Sense V was synthesized through a two-step post-synthetic conjugation approach substantially similar to Conjugated Sense U. 1.47 mg of Conjugated Sense V was obtained in 7.5% yield (LCMS found mass 8026). Sense V was annealed to Antisense B to yield Duplex V using the same procedures as described for the annealing of Duplex A.
FH12501620.1
Attorney Docket: DCY-13025 Synthesis of Duplex W:

Conjugated Sense W was prepared by solid-phase synthesis using a commercial oligo synthesizer. The oligonucleotides were synthesized using 2’-modified nucleoside phosphoramidites, such as 2'-F or 2'-OMe, and 2'-diethoxymethanol linked hexadecanoic acid (C16) fatty acid amide nucleoside phosphoramidites. Oligonucleotide synthesis was conducted on a solid support in the 3' to 5' direction using a standard oligonucleotide synthesis protocol. In these efforts, 5-ethylthio-1H-tetrazole (ETT) was used as an activator for the coupling reaction. Iodine solution was used for phosphite triester oxidation.3- (Dimethylaminomethylidene)amino-3H-1,2,4-dithiazole-3-thione (DDTT) was used for the formation of phosphorothioate linkages. Synthesized oligonucleotides were treated with concentrated aqueous ammonium for 10 h. The ammonia was removed from the suspension and the solid support residues were removed by filtration. The crude oligonucleotide was treated with TEAA, analyzed, and purified by strong anion exchange high performance liquid chromatography (SAX-HPLC). The fractions were combined and dialyzed against water (3 X), saline (1 X), and water (3 X) using Amicon® Ultra-15 Centrifugal (3K). The remaining solvent was then lyophilized to afford the desired Conjugated Sense W (LCMS found mass 7054). Sense W was annealed to Antisense B to yield Duplex W using the same procedures as described for the annealing of Duplex A. Synthesis of Duplex X:
FH12501620.1
Attorney Docket: DCY-13025
Conjugated Sense X was synthesized through a two-step post-syntenic conjugation approach. Step 1- Attachment of Amino-PEG4 Handle: In Eppendorf tube 1, a solution of Fmoc-N-amido-PEG4-NHS Ester (285 mg, 0.488 mmol, 6.0 equiv) was dissolved in DMF (20 mL). In Eppendorf tube 2, a solution of oligo
Sense X1 (500 mg, 0.0813 mmol, 1 equiv) in 5 mL H
2O was treated with DIPEA (142 µL, 0.8127 mmol, 10 equiv). The solution in Eppendorf tube 1 was added to the Eppendorf tube 2 and mixed for 1 hour using Thermomixer at 35°C. After the reaction was completed indicated by LC-MS analysis, the crude was then dialyzed against water (2 X) using Pierce Thermo Fisher 3k filters to remove excess of Fmoc-N-amido-PEG4-NHS Ester. The crude was then removed from filters and deprotected using water and Piperidine mixture (20 mL water: 3 mL Piperidine) and mixed for 1 h using Thermomixer at 35°C. The reaction was then diluted with 10 mL of water and purified by reverse phase XBridge C18 column using a 5-95% gradient of 100 mM TEAA in ACN and H2O. The product fractions were concentrated under reduced pressure using Genevac. The combined residual solvent was dialyzed against water (2 X), saline (2 X), and water (2 X) using Pierce Thermo Fisher 3k filters. The filter membrane was washed with water (3 X 2 mL) and with a last wash of 20% ethanol in water (2000 µL). The combined solvents were then lyophilized to afford an amorphous white solid of Conjugated Sense X2 (550 mg, 100% yield). Step 2- Conjugation to Amino-PEG4 Handle
FH12501620.1
Attorney Docket: DCY-13025 In Eppendorf tube 1, a solution of docosanedioic acid (290 mg, 0.7813 mmol, 10 equiv) was dissolved in DMA (17.5 mL) and treated with HATU (298 mg, 0.7813 mmol, 10 equiv) and DIPEA (140 µL, 0.7813 mmol, 10 euqiv) at rt. In Eppendorf tube 2, a solution of Conjugated
Sense X2 (550 mg, 0.07813 mmol, 1 equiv) in 2.5 mL H
2O and 5 mL DMA was treated with DIPEA (140 µL, 0.7813 mmol, 10 equiv). The solution in Eppendorf tube 1 was then added to the Eppendorf tube 2 and mixed for 30 minutes using Thermomixer at 40°C. After the reaction was completed indicated by LC-MS analysis, the reaction mixture was diluted with 5 mL of water and purified by revers phase XBridge C18 column using a 5-95% gradient of 100 mM TEAA in ACN and H2O. The product fractions were concentrated under reduced pressure using Genevac. The combined residual solvent was dialyzed against water (2 X), saline (2 X), and water (2 X) using Pierce Thermo Fisher 3k filters. The Amicon membrane was washed with water (3 X 2 mL) and with a last wash of 20% ethanol in water (20 µL). The combined solvents were then lyophilized to afford an amorphous white solid of Conjugated Sense X (222 mg, 40% yield) (LCMS found mass 6751). Sense X was annealed to Antisense B to yield Duplex X using the same procedures as described for the annealing of Duplex A. Synthesis of Duplex Y:
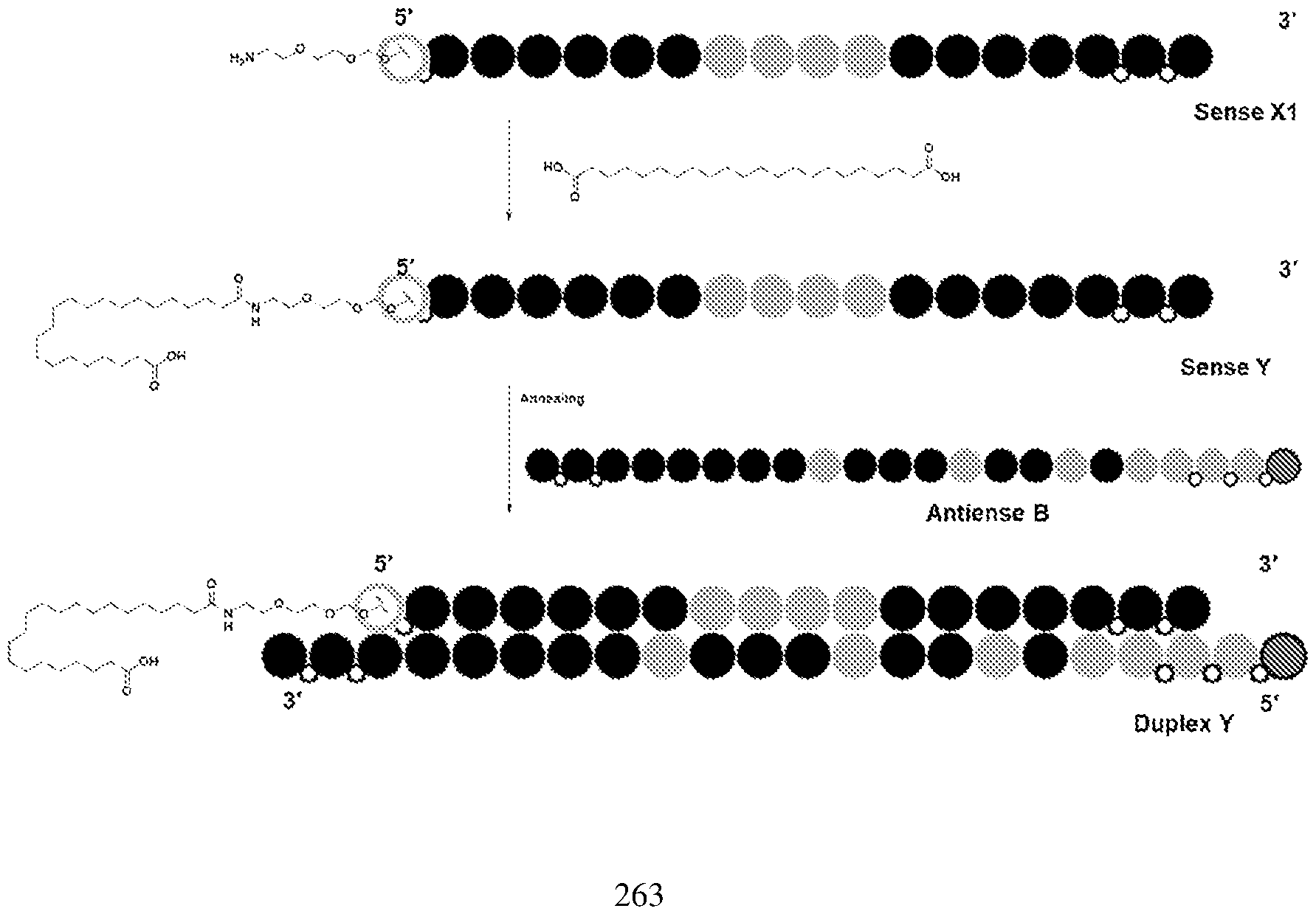
Attorney Docket: DCY-13025 Conjugated Sense Y was synthesized through a one-step post-syntenic conjugation approach. In Eppendorf tube 1, a solution of docosanedioic acid (310 mg, 0.8127 mmol, 10 equiv) in DMA (17.5 mL) was treated with HATU (310 mg, 0.8127 mmol, 10 equiv) at rt. In Eppendorf tube 2, a solution of oligo Sense X1 (500 mg, 0.0813 mmol, 1 equiv) in 2.5 mL H
2O and 5 mL DMA was treated with DIPEA (142 µL, 0.8127 mmol, 10 equiv). The solution in Eppendorf tube 1 was added to the Eppendorf tube 2 and mixed using Thermomixer at rt. After the reaction was completed indicated by LC-MS analysis, the reaction mixture was diluted with 5 mL of water and purified by revers phase XBridge C18 column using a 5-95% gradient of 100 mM TEAA in ACN and H2O. The product fractions were concentrated under reduced pressure using Genevac. The combined residual solvent was dialyzed against water (1 X), saline (1 X), and water (3 X) using Amicon® Ultra-15 Centrifugal (3K). The Amicon membrane was washed with water (3 X 2 mL) and the combined solvents were then lyophilized to afford an amorphous white solid of Conjugated Sense Y (438 mg, 82% yield) (LCMS found mass 6504). Sense Y was annealed to Antisense B to yield Duplex Y using the same procedures as described for the annealing of Duplex A. Synthesis of Duplex Z:
FH12501620.1
Attorney Docket: DCY-13025 Conjugated Sense Z was synthesized through a one-step post-syntenic conjugation approach. In Eppendorf tube 1, a solution of hexanedioic acid (233 mg, 0.8127 mmol, 10 equiv) in DMA (17.5 mL) was treated with HATU (310 mg, 0.8127 mmol, 10 equiv) at rt. In Eppendorf tube 2, a solution of oligo Sense X1 (500 mg, 0.0813 mmol, 1 equiv) in 2.5 mL H
2O and 5 mL DMA was treated with DIPEA (142 µL, 0.8127 mmol, 10 equiv). The solution in Eppendorf tube 1 was added to the Eppendorf tube 2 and mixed using Thermomixer at rt. After the reaction was completed indicated by LC-MS analysis, the reaction mixture was diluted with 5 mL of water and purified by revers phase XBridge C18 column using a 5-95% gradient of 100 mM TEAA in ACN and H2O. The product fractions were concentrated under reduced pressure using Genevac. The combined residual solvent was dialyzed against water (1 X), saline (1 X), and water (3 X) using Amicon® Ultra-15 Centrifugal (3K). The Amicon membrane was washed with water (3 X 2 mL) and the combined solvents were then lyophilized to afford an amorphous white solid of Conjugated Sense Z (454 mg, 87% yield) (LCMS found mass 6420). Sense Z was annealed to Antisense B to yield Duplex Z using the same procedures as described for the annealing of Duplex A. Synthesis of Duplex AA:

Conjugated Sense X was synthesized through a two-step post-syntenic conjugation approach.
FH12501620.1
Attorney Docket: DCY-13025 Step 1- Attachment of Amino-PEG4 Handle:
Sense X2 was synthesized with the method above. Step 2- Conjugation to Amino-PEG4 Handle In Eppendorf tube 1, a solution of hexadecanedioic acid (95 mg, 0.3313 mmol, 10 equiv) was dissolved in DMA (17.5 mL) and treated with HATU (126 mg, 0.3313 mmol, 10 equiv) and DIPEA (70 µL, 0.3313 mmol, 10 euqiv) at rt. In Eppendorf tube 2, a solution of Conjugated Sense X2 (212 mg, 0.03313 mmol, 1 equiv) in 1.37 mL H2O and 5 mL DMA was treated with DIPEA (70 µL, 0.3313 mmol, 10 equiv). The solution in Eppendorf tube 1 was then added to the Eppendorf tube 2 and mixed for 30 minutes using Thermomixer at 40°C. After the reaction was completed indicated by LC-MS analysis, the reaction mixture was diluted with 5 mL of water and purified by revers phase XBridge C18 column using a 5-95% gradient of 100 mM TEAA in ACN and H2O. The product fractions were concentrated under reduced pressure using Genevac. The combined residual solvent was dialyzed against water (2 X), saline (2 X), and water (2 X) using Pierce Thermo Fisher 3k filters. The Amicon membrane was washed with water (3 X 2 mL) and with a last wash of 20% ethanol in water (20 µL). The combined solvents were then lyophilized to afford an amorphous white solid of Conjugated Sense AA (222 mg, 40% yield) (LCMS found mass 6751). Sense AA was annealed to Antisense B to yield Duplex AA using the same procedures as described for the annealing of Duplex A. Synthesis of Duplex AB:
FH12501620.1
Attorney Docket: DCY-13025
Conjugated Sense AB was synthesized through a two-step post-syntenic conjugation approach. Step 1- Attachment of Amino-PEG4 Handle: Sense M1 was synthesized with the method above. Step 2- Conjugation to Amino-PEG4 Handle In Eppendorf tube 1,
ABa (29 mg, 0.061 mmol, 43 equiv) was dissolved in DMA (450 µL). In Eppendorf tube 2, a solution of Conjugated Sense M1 (10.0 mg, 0.00142 mmol, 1 equiv) in 100 µL H
2O was treated with DIPEA (1.9 µL, 0.00107 mmol). The solution in Eppendorf tube 1 was then added to the Eppendorf tube 2 and mixed for 30 minutes using Thermomixer at 25°C. After the reaction was completed indicated by LC-MS analysis, the reaction mixture was diluted with 5 mL of water and purified by revers phase XBridge C18 column using a 5-95% gradient of 100 mM TEAA in ACN and H2O. The product fractions were concentrated under reduced pressure using Genevac. The combined residual solvent was dialyzed against water (2 X), saline (2 X), and water (2 X) using Pierce Thermo Fisher 3k filters. The Amicon membrane was washed with water (3 X 2 mL) and with a last wash of 20% ethanol in water (20 µL). The combined solvents were then lyophilized to afford an amorphous white solid of Conjugated
Sense AB (5.0 mg, 0.00068 mmol, 48% yield) (LCMS found mass 7417). Sense AB was annealed to Antisense B to yield Duplex AB using the same procedures as described for the annealing of Duplex A. 267
FH12501620.1
Attorney Docket: DCY-13025 Synthesis of the small molecule: 2-(16-((2,5-dioxopyrrolidin-1-yl)oxy)-16-oxohexadecyl)- 2-methylmalonic acid (ABa):

Step 1: 1,1-di-tert-butyl 16-methyl hexadecane-1,1,16-tricarboxylate (ABc) To a 6-dram vial were added NaH (229 mg, 60 w.%, 4.0 equiv). The vial was evacuated with vacuum and back filled with nitrogen three times, then cooled in ice/water bath. THF (10 mL, anhydrous) was added, followed by di-tert-butyl malonate (464 mg, 1.5 equiv) dropwise via syringe. The mixture was then removed from ice/water bath and stirred at r.t. for 30 min. After 30 min, the mixture was cooled in ice/water bath, methyl 16- bromohexadecanoate (ABb, 500 mg, 1.0 equiv, dissolved in 3 mL THF) was added dropwise via syringe. The mixture was then stirred at r.t. overngiht. After overnight the mixture was cooled in ice/water bath, quenched with sat. ammonium chloride, then extracted with DCM. The org layer was washed with brine, dried over sodium sulfate, and concentrated in vacuo. The resulting crude was purified with normal phase chromatography (0-5% EtOAc in Hexanes). Fractions containing product were combined and concentrated in vacuo to give 1,1-di-tert-butyl 16-methyl hexadecane-1,1,16- tricarboxylate (ABc) as a white powder (69 mg, 10%). LCMS [M-tBu+Na]
+ calculated 507.4, found 508.0.
1H NMR (500 MHz, CDCl
3) ^ 3.67 (d, J = 0.9 Hz, 3H), 3.16 – 3.06 (m, 1H), 2.30 (t, J = 7.5 Hz, 2H), 1.79 (q, J = 7.8, 6.9 Hz, 2H), 1.66 – 1.56 (m, 2H), 1.54 (d, J = 0.9 Hz, 9H), 1.46 (d, J = 0.9 Hz, 12H), 1.27 (d, J = 25.4 Hz, 21H).
13C NMR (101 MHz, CDCl3) ^ 174.53, 169.23, 81.29, 54.17, 51.60, 34.28, 29.81, 29.79, 29.77, 29.75, 29.69, 29.61, 29.51, 29.44, 29.41, 29.31, 28.74, 28.09, 27.35, 25.12. Step 2: 16,16-di-tert-butyl 1-methyl heptadecane-1,16,16-tricarboxylate (ABd) To a 1-dram vial was added NaH (4 mg, 1.1 equiv), The vial was evacuated with vacuum and back filled with nitrogen three times, then cooled in ice/water bath. THF (1 mL, anhydrous) was added, followed by 1,1-di-tert-butyl 16-methyl hexadecane-1,1,16- tricarboxylate (69 mg, 1 equiv, dissolved in 1 mL THF). The mixture was stirred at r.t. for
FH12501620.1
Attorney Docket: DCY-13025 10 min, then cooled in ice/water bath. MeI (20 mg, 1 equiv) was added dropwise via syringe, and the mixture was stirred at r.t. for 30 min. After 30 min, the mixture was quenched with sat. ammonium chloride, and extracted with DCM (10 mL x 3). The org. layers were combined and washed with brine, dried over sodium sulfate and concentrated in vacuo to give 16,16-di-tert-butyl 1-methyl heptadecane-1,16,16-tricarboxylate (ABd) as a faint yellow liquid (66 mg, 93%). LCMS [M-tBu+H]
+ calculated 443.3, found 443.9. Used directly for next step without further purifiation. Step 3: 18-(tert-butoxy)-17-(tert-butoxycarbonyl)-17-methyl-18-oxooctadecanoic acid (ABe) To a 6-dram vial containing 16,16-di-tert-butyl 1-methyl heptadecane-1,16,16- tricarboxylate (66 mg, 1.0 equiv) were added THF (1.5 mL), MeOH (0.5 mL), water (0.5 mL), and LiOH (32 mg, 10 equiv). The mixture was stirred at r.t. for 3 h. After 3 h, the mixture was diluted with DCM, acidified with citric acid until pH<2, and washed with sat. ammonium chloride. The aq. layer was extracted twice with DCM. All org layers combined, washed three times with water, then washed with brine, dried over sodium sulfate, and concentrated in vacuo gave 18-(tert-butoxy)-17-(tert-butoxycarbonyl)-17- methyl-18-oxooctadecanoic acid (ABe) (61 mg, 95%) as a slightly yellow viscous liquid. LCMS [M-tBu+H]
+ calculated 429.3, found 429.0.
1H NMR (500 MHz, CDCl
3) ^ 2.35 (t, J = 7.5 Hz, 2H), 1.80 – 1.72 (m, 2H), 1.68 – 1.50 (m, 8H), 1.44 (s, 18H), 1.37 – 1.16 (m, 21H).
13C NMR (101 MHz, CDCl3) ^ 177.33, 172.12, 80.92, 54.78, 28.08, 27.98. Step 4&5: 2-(16-((2,5-dioxopyrrolidin-1-yl)oxy)-16-oxohexadecyl)-2-methylmalonic acid (ABa) To a 1-dram vial were added 18-(tert-butoxy)-17-(tert-butoxycarbonyl)-17-methyl-18- oxooctadecanoic acid (30 mg, 1.0 equiv) and TSTU (38 mg, 2.0 equiv). The vial was evacuated with vacuum and back filled with nitrogen three times, then added THF (1 mL) and DIPEA (20 uL, 2.0 equiv). The mixture was stirred at r.t. for 16 h. After 16 h, the mixture was separated between EtOAc (10 mL) and sat. NH
4Cl (10 mL). The organic layer was washed with water, brine, dried over sodium sulfate, and concentrated in vacuo. The crude was then stirred in neat TFA (0.5 mL) for 1 h, then concentrated in vacuo, stripped with toluene in vacuo 3 times and dissolved in 400 µL DMA. The obtained 2- (16-((2,5-dioxopyrrolidin-1-yl)oxy)-16-oxohexadecyl)-2-methylmalonic acid (ABa) was used without further purification. Synthesis of Duplex AC:
FH12501620.1
Attorney Docket: DCY-13025
Conjugated Sense AB was synthesized through a two-step post-syntenic conjugation approach. Step 1- Attachment of Amino-PEG4 Handle: Sense M1 was synthesized with the method above. Step 2- Conjugation to Amino-PEG4 Handle In Eppendorf tube 1, a solution of 16-sulfohexadecanoic acid (4.77 mg, 0.0142 mmol, 10 equiv) was dissolved in DMF (500 µL) and treated with HATU (4.29 mg, 0.0113 mmol, 8 equiv) and DIPEA (2.5 µL, 0.0142 mmol, 10 equiv) at rt and mixed for 15 minutes. In Eppendorf tube 2, a solution of Conjugated Sense M1 (10.0 mg, 0.00142 mmol, 1 equiv) in 100 µL H2O was treated with DIPEA (2.5 µL, 0.0142 mmol, 10 equiv). The solution in Eppendorf tube 1 was then added to the Eppendorf tube 2 and mixed for 30 minutes using Thermomixer at 40°C. After the reaction was completed indicated by LC-MS analysis, the reaction mixture was diluted with 5 mL of water and purified by reverse phase XBridge C18 column using a 5-95% gradient of 100 mM TEAA in ACN and H
2O. The product fractions were concentrated under reduced pressure using GeneVac. The combined residual solvent was dialyzed against water (2 X), saline (2 X), and water (2 X) using Pierce Thermo Fisher 3k filters. The Amicon membrane was washed with water (3 X 2 mL) and with a last wash of 20% ethanol in water (20 µL). The combined solvents were then lyophilized to afford an amorphous white solid of Conjugated Sense AC (5.0 mg, 0.00068 mmol, 48% yield). Post-synthetic conjugation was
FH12501620.1
Attorney Docket: DCY-13025 realized through an amide coupling reaction. The Conjugated Sense AC was characterized by LCMS. Observed LCMS: 7341. Sense AC was annealed to Antisense B to yield Duplex AC using the same procedures as described for the annealing of Duplex A. Synthesis of Duplex AD:
Conjugated Sense AD was synthesized through a two-step post-syntenic conjugation approach. Step 1- Attachment of Amino-PEG4 Handle: Sense M1 was synthesized with the method above. Step 2- Conjugation to Amino-PEG4 Handle In Eppendorf tube 1, a solution of 16-(2H-tetrazol-5-yl)hexadecanoic acid (ADa) (20 mg, 0.0617 mmol, 4 equiv) was dissolved in DMF (500 µL) and treated with HATU (4.29 mg, 0.0113 mmol, 8 equiv) and DIPEA (2.5 µL, 0.0142 mmol, 10 equiv) at rt and mixed for 15 minutes. In Eppendorf tube 2, a solution of Conjugated Sense M1 (10.0 mg, 0.00142 mmol, 1 equiv) in 100 µL H
2O was treated with DIPEA (2.5 µL, 0.0142 mmol, 10 equiv). The solution in Eppendorf tube 1 was then added to the Eppendorf tube 2 and mixed for 30 minutes using Thermomixer at 40°C. After the reaction was completed indicated by LC-MS analysis, the reaction mixture was diluted with 5 mL of water and purified by reverse phase XBridge C18 column using a 5-95% gradient of 100 mM TEAA in ACN and H2O. The product fractions
FH12501620.1
Attorney Docket: DCY-13025 were concentrated under reduced pressure using GeneVac. The combined residual solvent was dialyzed against water (2 X), saline (2 X), and water (2 X) using Pierce Thermo Fisher 3k filters. The Amicon membrane was washed with water (3 X 2 mL) and with a last wash of 20% ethanol in water (20 µL). The combined solvents were then lyophilized to afford an amorphous white solid of Conjugated Sense AD (1.0 mg, 0.00014 mmol, 10% yield). Post-synthetic conjugation was realized through an amide coupling reaction. The Conjugated Sense AC was characterized by LCMS. Observed LCMS: 7369. Sense AD was annealed to Antisense B to yield Duplex AD using the same procedures as described for the annealing of Duplex A. Synthesis of the small molecule: 16-(2H-tetrazol-5-yl)hexadecanoic acid (ADa)

To a 2-dram vial were added 16-cyanohexadecanoic acid (60 mg, 0.22 mmol, 1.0 equiv), NaN3 (55 mg, 4.0 equiv, freshly crushed), triethylamine hydrochloride (59 mg, 2.0 equiv). The vial was evacuated with vacuum and back filled with nitrogen three times, then added NMP (0.5 mL). The mixture was heated to 150 °C for 24 h. The crude was diluted with 1 mL DMF and 2 mL water, loaded on C18 cartridge, and purified with reverse phase chromatography, MeCN in water 0-100% gradient. Fractions containing product were combined and concentrated in vacuo at 50 °C, then lyophilized to give 16- (2H-tetrazol-5-yl)hexadecanoic acid (ADa) as an off-white powder (51 mg, 74%). LCMS [M+H]
+ calculated 325.3, found 325.0.
1H NMR (500 MHz, DMSO) ^ 2.57 (t, 2H), 1.98 (t, J = 7.4 Hz, 2H), 1.56 (t, J = 7.5 Hz, 2H), 1.47 – 1.40 (m, 2H), 1.23 (d, J = 3.4 Hz, 22H).
13C NMR (126 MHz, DMSO) ^ 175.98, 160.59, 36.47, 29.16, 29.05, 29.02, 28.96, 28.94, 28.92, 25.69, 25.43. Synthesis of Duplex AE:
FH12501620.1
Attorney Docket: DCY-13025
Conjugated Sense AE was synthesized through a two-step post-syntenic conjugation approach. Step 1- Attachment of Amino-PEG4 Handle: Sense M1 was synthesized with the method above. Step 2- Conjugation to Amino-PEG4 Handle In Eppendorf tube 1, a solution of 16-hydroxyhexadecanoic acid (3.8 mg, 0.014 mmol, 10 equiv) was dissolved in DMF (500 µL) and treated with HATU (4.29 mg, 0.0113 mmol, 8 equiv) and DIPEA (2.5 µL, 0.0142 mmol, 10 equiv) at rt and mixed for 15 minutes. In Eppendorf tube 2, a solution of Conjugated Sense M1 (10.0 mg, 0.00142 mmol, 1 equiv) in 100 µL H2O was treated with DIPEA (2.5 µL, 0.0142 mmol, 10 equiv). The solution in Eppendorf tube 1 was then added to the Eppendorf tube 2 and mixed for 30 minutes using Thermomixer at 40°C. After the reaction was completed indicated by LC-MS analysis, the reaction mixture was diluted with 5 mL of water and purified by reverse phase XBridge C18 column using a 5-95% gradient of 100 mM TEAA in ACN and H
2O. The product fractions were concentrated under reduced pressure using GeneVac. The combined residual solvent was dialyzed against water (2 X), saline (2 X), and water (2 X) using Pierce Thermo Fisher 3k filters. The Amicon membrane was washed with water (3 X 2 mL) and with a last wash of 20% ethanol in water (20 µL). The combined solvents were then lyophilized to afford an amorphous white solid of Conjugated Sense AE (1.44 mg, 0.00019 mmol, 14% yield). Post-synthetic
FH12501620.1
Attorney Docket: DCY-13025 conjugation was realized through an amide coupling reaction. The Conjugated Sense AC was characterized by LCMS. Observed LCMS: 7317. Sense AE was annealed to Antisense B to yield Duplex AE using the same procedures as described for the annealing of Duplex A. Synthesis of Duplex AF:
Conjugated Sense AF was synthesized through a two-step post-syntenic conjugation approach. Step 1- Attachment of Amino-PEG4 Handle: Sense M1 was synthesized with the method above. Step 2- Conjugation to Amino-PEG4 Handle In Eppendorf tube 1, a solution of 16-(cyclopropanesulfonamido)hexadecanoic acid (AFa) (5 mg, 0.0142 mmol, 10 equiv) was dissolved in DMF (500 µL) and treated with HATU (4.29 mg, 0.0113 mmol, 8 equiv) and DIPEA (2.5 µL, 0.0142 mmol, 10 equiv) at rt and mixed for 15 minutes. In Eppendorf tube 2, a solution of
Conjugated Sense M1 (10.0 mg, 0.00142 mmol, 1 equiv) in 100 µL H
2O was treated with DIPEA (2.5 µL, 0.0142 mmol, 10 equiv). The solution in Eppendorf tube 1 was then added to the Eppendorf tube 2 and mixed for 30 minutes using Thermomixer at 40°C. After the reaction was completed indicated by LC-MS analysis, the reaction mixture was diluted with 5 mL of water and purified by reverse phase XBridge C18
FH12501620.1
Attorney Docket: DCY-13025 column using a 5-95% gradient of 100 mM TEAA in ACN and H
2O. The product fractions were concentrated under reduced pressure using GeneVac. The combined residual solvent was dialyzed against water (2 X), saline (2 X), and water (2 X) using Pierce Thermo Fisher 3k filters. The Amicon membrane was washed with water (3 X 2 mL) and with a last wash of 20% ethanol in water (20 µL). The combined solvents were then lyophilized to afford an amorphous white solid of Conjugated Sense AF (6.2 mg, 0.00088 mmol, 57% yield). Post-synthetic conjugation was realized through an amide coupling reaction. The Conjugated Sense AF was characterized by LCMS. Observed LCMS: 7421. Sense AF was annealed to Antisense B to yield Duplex AF using the same procedures as described for the annealing of Duplex A. Synthesis of the small molecule: 16-(cyclopropanesulfonamido)hexadecanoic acid
To a stirred solution of cyclopropane sulfonamide (156.6 mg, 1.289 mmol, 1.5 equiv) in anhydrous DMF (4 mL) at 0
oC, sodium hydride (55% in mineral oil, 52.5 mg, 1.201 mmol, 1.4 equiv) was added. The resulting mixture was stirred at the same temperature for 30 min. A solution of methyl 16-bromohexadecanoate (300 mg, 0.859 mmol, 1.0 equiv) in anhydrous DMF (2 mL) was added to the reaction mixture and the resulting mixture was stirred at 25 °C for 3 h. The reaction mixture was cooled to 0
oC and carefully quenched by adding saturated aqueous NH4Cl solution (10 mL). The aqueous layer was extracted with EtOAc (3 × 10 mL) and the combined organic layer was dried over anhydrous Na2SO4, filtered, and concentrated under reduced pressure. The obtained residue was purified by flash column chromatography (silica gel, 5
20% ethyl acetate in hexanes) to afford pure 3) Methyl 16- (cyclopropanesulfonamido)hexadecanoate (AFb) (207.5 mg, 0.532 mmol, 62% yield) as a white solid.
1H NMR (400 MHz, CDCl3) ^ 4.48 (t, J = 7.8 Hz, 1H), 3.64 (s, 3H),
FH12501620.1
Attorney Docket: DCY-13025 3.17 (t, J = 7.8 Hz, 2H), 2.28 (t, J = 7.8 Hz, 2H), 1.69 – 1.48 (m, 6H), 1.36 – 1.19 (m, 21H), 1.17 – 1.11 (m, 2H), 0.97 (dt, J = 7.8, 1.6 Hz, 1H), 0.92 (dd, J = 7.8, 2.4 Hz, 1H) ppm;
13C NMR (101 MHz, CDCl
3) ^ 174.5, 51.6, 47.6, 43.6, 34.3, 29.9, 29.8, 29.7, 29.6, 29.5, 29.46, 29.40, 29.3, 29.2, 28.9, 26.9, 26.7, 25.1, 5.2 ppm; To a stirred solution of Methyl 16-(cyclopropanesulfonamido)hexadecanoate (AFb) (120 mg, 0.308 mmol, 1.0 equiv) in THF:MeOH (3:1 v/v, 4 mL) was added a solution of LiOH (73.8 mg, 3.080 mmol, 10 equiv) in H2O (xx mL) at 25
oC. The resulting mixture was stirred at the same temperature for 4 h. The pH of the reaction mixture was adjusted to 3-4 using 1N HCl solution. The aqueous layer was extracted with EtOAc (3 × 10 mL) and the combined organic layer was dried over anhydrous Na2SO4, filtered, and concentrated under reduced pressure. The obtained residue was purified by flash column chromatography (silica gel, 20 → 80% ethyl acetate in hexanes) to afford pure 16-(cyclopropanesulfonamido)hexadecanoic acid (AFa) (69.4 mg, 0.185 mmol, 60% yield) as a white solid.
1H NMR (500 MHz, CDCl3) ^ 3.24 (t, J = 7.7 Hz, 2H), 2.39 (t, J = 7.7 Hz, 2H), 1.70 – 1.58 (m, 6H), 1.39 – 1.30 (m, 21H), 1.23 – 1.17 (m, 2H), 1.06 – 1.01 (m, 1H), 1.00 – 0.95 (m, 1H) ppm;
13C NMR (126 MHz, CDCl3) ^ 180.1, 47.9, 43.6, 34.2, 29.8, 29.76, 29.70, 29.65, 29.59, 29.51, 29.42, 29.36, 29.2, 29.0, 27.0, 26.8, 24.9, 5.5, 5.3 ppm; Synthesis of Duplex AG:
Conjugated Sense AG was synthesized through a two-step post-syntenic conjugation approach.
FH12501620.1
Attorney Docket: DCY-13025 Step 1- Attachment of Amino-PEG4 Handle:
Sense M1 was synthesized with the method above. Step 2- Conjugation to Amino-PEG4 Handle In Eppendorf tube 1, a solution of 16-(methylsulfonamido)hexadecanoic acid (AGa) (5 mg, 0.0142 mmol, 10 equiv) was dissolved in DMF (500 µL) and treated with HATU (4.29 mg, 0.0113 mmol, 8 equiv) and DIPEA (2.5 µL, 0.0142 mmol, 10 equiv) at rt and mixed for 15 minutes. In Eppendorf tube 2, a solution of Conjugated Sense M1 (10.0 mg, 0.00142 mmol, 1 equiv) in 100 µL H2O was treated with DIPEA (2.5 µL, 0.0142 mmol, 10 equiv). The solution in Eppendorf tube 1 was then added to the Eppendorf tube 2 and mixed for 30 minutes using Thermomixer at 40°C. After the reaction was completed indicated by LC-MS analysis, the reaction mixture was diluted with 5 mL of water and purified by reverse phase XBridge C18 column using a 5-95% gradient of 100 mM TEAA in ACN and H2O. The product fractions were concentrated under reduced pressure using GeneVac. The combined residual solvent was dialyzed against water (2 X), saline (2 X), and water (2 X) using Pierce Thermo Fisher 3k filters. The Amicon membrane was washed with water (3 X 2 mL) and with a last wash of 20% ethanol in water (20 µL). The combined solvents were then lyophilized to afford an amorphous white solid of Conjugated Sense AG (4.0 mg, 0.00061 mmol, 43% yield). Post-synthetic conjugation was realized through an amide coupling reaction. The Conjugated Sense AG was characterized by LCMS. Observed LCMS: 7390. Sense AG was annealed to Antisense B to yield Duplex AG using the same procedures as described for the annealing of Duplex A. Synthesis of the small molecule: 16-(methylsulfonamido)hexadecanoic acid (AGa):

To a stirred solution of methane sulfonamide (81.7 mg, 0.859 mmol, 1.5 equiv) in anhydrous DMF (3 mL) at 0
oC, sodium hydride (55% in mineral oil, 34.0 mg, 0.801 mmol, 1.4 equiv) was added. The resulting mixture was stirred at the same temperature for 30 min. A solution of methyl 16-bromohexadecanoate (200 mg, 0.572 mmol, 1.0 equiv) in anhydrous DMF (2
FH12501620.1
Attorney Docket: DCY-13025 mL) was added to the reaction mixture and the resulting mixture was stirred at 25 °C for 3 h. The reaction mixture was cooled to 0
oC and carefully quenched by the addition of saturated aqueous NH
4Cl solution (10 mL). The aqueous layer was extracted with EtOAc (3 × 10 mL) and the combined organic layer was dried over anhydrous Na
2SO
4, filtered, and concentrated under reduced pressure. The obtained residue was purified by flash column chromatography (silica gel, 2
15% ethyl acetate in hexanes) to afford pure Methyl 16- (methylsulfonamido)hexadecanoate (AGb) (149.7 mg, 0.412 mmol, 72% yield) as a white solid.
1H NMR (CDCl
3, 600 MHz) ^ 4.19 (t, J = 6.2 Hz, 1H), 3.66 (s, 3H), 3.18 – 3.08 (m, 2H), 2.95 (s, 3H), 2.30 (t, J = 7.6 Hz, 2H), 1.67 – 1.51 (m, 6H), 1.27 (d, J = 15.4 Hz, 20H) ppm;
13C NMR (CDCl3, 151 MHz) ^ = 174.5, 51.6, 43.5, 40.5, 34.3, 30.3, 29.8, 29.74, 29.73, 29.71, 29.67, 29.61, 29.5, 29.4, 29.3, 26.7, 25.1 ppm; To a stirred solution of Methyl 16-(methylsulfonamido)hexadecanoate (AGb) (100 mg, 0.275 mmol, 1.0 equiv) in THF:MeOH (3:1 v/v, 4 mL) was added a solution of LiOH (65.9 mg, 2.750 mmol, 10 equiv) in H2O (1 mL) at 25
oC. The resulting mixture was stirred at the same temperature for 4h. The pH of the reaction mixture was adjusted to 3-4 using 1N HCl solution. The aqueous layer was extracted with EtOAc (3 × 10 mL) and the combined organic layer was dried over anhydrous Na
2SO
4, filtered, and concentrated under reduced pressure. The obtained residue was purified by flash column chromatography (silica gel, 20 → 80% ethyl acetate in hexanes) to afford pure 16-(methylsulfonamido)hexadecanoic acid (AGa) (62.5 mg, 0.179 mmol, 65% yield) as a white solid.
1H NMR (400 MHz, MeOD) ^ 3.17 – 3.13 (m, 1H), 3.04 (t, J = 7.1 Hz, 2H), 2.91 (s, 3H), 2.28 (t, J = 7.4 Hz, 2H), 1.57 (dt, J = 20.8, 7.3 Hz, 6H), 1.39 – 1.26 (m, 20H) ppm;
13C NMR (126 MHz, MeOD) ^ 177.7, 44.1, 39.6, 35.0, 31.2, 30.8, 30.7, 30.6, 30.6, 30.5, 30.4, 30.3, 30.2, 30.1, 27.8, 26.1 ppm; Synthesis of Duplex AH:
FH12501620.1
Attorney Docket: DCY-13025
Conjugated Sense AH was synthesized through a two-step post-syntenic conjugation approach. Step 1- Attachment of Amino-PEG4 Handle: Sense M1 was synthesized with the method above. Step 2- Conjugation to Amino-PEG4 Handle In Eppendorf tube 1, a solution of palmitic acid (5 mg, 0.0142 mmol, 10 equiv) was dissolved in DMF (500 µL) and treated with HATU (4.29 mg, 0.0113 mmol, 8 equiv) and DIPEA (2.5 µL, 0.0142 mmol, 10 equiv) at rt and mixed for 15 minutes. In Eppendorf tube 2, a solution of Conjugated Sense M1 (10.0 mg, 0.00142 mmol, 1 equiv) in 100 µL H2O was treated with DIPEA (2.5 µL, 0.0142 mmol, 10 equiv). The solution in Eppendorf tube 1 was then added to the Eppendorf tube 2 and mixed for 30 minutes using Thermomixer at 40°C. After the reaction was completed indicated by LC-MS analysis, the reaction mixture was diluted with 5 mL of water and purified by reverse phase XBridge C18 column using a 5-95% gradient of 100 mM TEAA in ACN and H
2O. The product fractions were concentrated under reduced pressure using GeneVac. The combined residual solvent was dialyzed against water (2 X), saline (2 X), and water (2 X) using Pierce Thermo Fisher 3k filters. The Amicon membrane was washed with water (3 X 2 mL) and with a last wash of 20% ethanol in water (20 µL). The combined solvents were then lyophilized to afford an amorphous white solid of Conjugated Sense AH (10 mg, 0.0014 mmol, 100% yield). Post-synthetic conjugation was realized through an amide coupling reaction. The Conjugated Sense AH was characterized by LCMS. Observed LCMS: 7301.
FH12501620.1
Attorney Docket: DCY-13025 Sense AH was annealed to Antisense B to yield Duplex AH using the same procedures as described for the annealing of Duplex A. Synthesis of Duplex AI:
Conjugated Sense AI was synthesized through a two-step post-syntenic conjugation approach. Step 1- Attachment of Amino-PEG4 Handle: Sense M1 was synthesized with the method above. Step 2- Conjugation to Amino-PEG4 Handle
In Eppendorf tube 1, a solution of 16-(3-(methylsulfonyl)ureido)hexadecanoic acid (AIa) (5 mg, 0.0142 mmol, 10 equiv) was dissolved in DMF (500 µL) and treated with HATU (4.29 mg, 0.0113 mmol, 8 equiv) and DIPEA (2.5 µL, 0.0142 mmol, 10 equiv) at rt and mixed for 15 minutes. In Eppendorf tube 2, a solution of Conjugated Sense M1 (10.0 mg, 0.00142 mmol, 1 equiv) in 100 µL H
2O was treated with DIPEA (2.5 µL, 0.0142 mmol, 10 equiv). The solution in Eppendorf tube 1 was then added to the Eppendorf tube 2 and mixed for 30 minutes using Thermomixer at 40°C. After the reaction was completed indicated by LC-MS analysis, the reaction mixture was diluted with 5 mL of water and purified by reverse phase XBridge C18 column using a 5-95% gradient of 100 mM TEAA in ACN and H2O. The product fractions were concentrated under reduced pressure using GeneVac. The combined residual solvent was dialyzed against water (2 X), saline (2 X), and water (2 X) using Pierce Thermo Fisher 3k
FH12501620.1
Attorney Docket: DCY-13025 filters. The Amicon membrane was washed with water (3 X 2 mL) and with a last wash of 20% ethanol in water (20 µL). The combined solvents were then lyophilized to afford an amorphous white solid of Conjugated Sense AI (3.0 mg, 0.00051 mmol, 36% yield). Post-synthetic conjugation was realized through an amide coupling reaction. The Conjugated Sense AI was characterized by LCMS. Observed LCMS: 7439. Sense AI was annealed to Antisense B to yield Duplex AI using the same procedures as described for the annealing of Duplex A. Synthesis of the small molecule: 16-(3-(methylsulfonyl)ureido)hexadecanoic acid (AIa):
To a stirred solution of 17-(tert-butoxy)-17-oxoheptadecanoic acid (200 mg, 0.561 mmol, 1.0 equiv) in 1,4-dioxane (4 mL) was added P(Ph)2N3 (180.9 µL, 0.841 mmol, 1.5 equiv) followed by K2CO3 (155.3 mg, 1.122 mmol, 2.0 equiv) at 23
oC, and a reaction mixture was stirred at the same temperature for 10 minutes. Solid methane sulfonamide (77.5 mg, 0.841 mmol, 1.5 equiv) was added to the reaction mixture and further stirred for 5 h. The progress was monitored by TLC. The reaction mixture was carefully quenched with saturated aqueous NH
4Cl solution (10 mL). The aqueous layer was extracted with EtOAc (3 x 10 mL), dried, and evaporated to dryness under reduced pressure. The obtained residue was purified by flash column chromatography (silica gel, 10 → 40% ethyl acetate in hexanes) to furnish the tert-butyl 16-(3- (methylsulfonyl)ureido)hexadecanoate (AIb) (130.9 mg, 0.292 mmol, 52% yield) along with phosphine oxide impurity as a white solid.
1H NMR (500 MHz, CDCl
3) ^ 8.46 (s, 1H), 6.30 (t, J = 5.7 Hz, 1H), 3.26 – 3.18 (m, 5H), 2.20 (t, J = 7.5 Hz, 2H), 1.60 – 1.47 (m, 6H), 1.44 (s, 9H), 1.28 – 1.24 (m, 20H) ppm;
13C NMR (101 MHz, CDCl3) ^ 173.8, 152.1, 80.2, 42.1, 35.9, 29.8, 29.78, 29.75, 29.73, 29.71, 29.66, 29.50, 29.44, 29.30, 28.32, 26.99, 25.33 ppm; To a round bottom flask containing tert-butyl 16-(3- (methylsulfonyl)ureido)hexadecanoate (AIb) (70 mg, 0.156 mmol, 1.0 equiv) was added a solution of TFA:CH2Cl2 (1.5mL:2.5mL) at 23
oC, and a reaction mixture was stirred for 2 h. After completion of the reaction, CH2Cl2 and TFA were removed
FH12501620.1
Attorney Docket: DCY-13025 under reduced pressure, and the residue was co-evaporated twice with toluene (3 mL) to remove excess TFA. The obtained residue was purified by flash column chromatography (silica gel, 30 → 100% ethyl acetate in hexanes) to furnish the 16-(3- (methylsulfonyl)ureido)hexadecanoic acid (AIa) (48.9 mg, 0.125 mmol, 80% yield) as a foamy white solid.
1H NMR (CDCl3, 600 MHz) ^ =
1H NMR (500 MHz, MeOD) ^ 3.22 (s, 3H), 3.16 (t, J = 7.1 Hz, 2H), 2.28 (t, J = 7.4 Hz, 2H), 1.60 (p, J = 7.1 Hz, 2H), 1.50 (q, J = 6.9 Hz, 2H), 1.31 (d, J = 10.3 Hz, 24H) ppm; Synthesis of Duplex AJ:
Conjugated Sense AJ was synthesized through a two-step post-syntenic conjugation approach. Step 1- Attachment of Amino-PEG4 Handle:
Sense M1 was synthesized with the method above. Step 2- Conjugation to Amino-PEG4 Handle In Eppendorf tube 1, a solution of 17-((4-hydroxyphenyl)amino)-17-oxoheptadecanoic acid (AJa) (5 mg, 0.0142 mmol, 10 equiv) was dissolved in DMF (500 µL) and treated with HATU (4.29 mg, 0.0113 mmol, 8 equiv) and DIPEA (2.5 µL, 0.0142 mmol, 10 equiv) at rt and mixed for 15 minutes. In Eppendorf tube 2, a solution of Conjugated Sense M1 (10.0 mg, 0.00142 mmol, 1 equiv) in 100 µL H2O was treated with DIPEA (2.5 µL, 0.0142 mmol, 10 equiv). The
FH12501620.1
Attorney Docket: DCY-13025 solution in Eppendorf tube 1 was then added to the Eppendorf tube 2 and mixed for 30 minutes using Thermomixer at 40°C. After the reaction was completed indicated by LC-MS analysis, the reaction mixture was diluted with 5 mL of water and purified by reverse phase XBridge C18 column using a 5-95% gradient of 100 mM TEAA in ACN and H
2O. The product fractions were concentrated under reduced pressure using GeneVac. The combined residual solvent was dialyzed against water (2 X), saline (2 X), and water (2 X) using Pierce Thermo Fisher 3k filters. The Amicon membrane was washed with water (3 X 2 mL) and with a last wash of 20% ethanol in water (20 µL). The combined solvents were then lyophilized to afford an amorphous white solid of Conjugated Sense AJ (6.0 mg, 0.00081 mmol, 57% yield). Post-synthetic conjugation was realized through an amide coupling reaction. The Conjugated Sense AJ was characterized by LCMS. Observed LCMS: 7436. Sense AJ was annealed to Antisense B to yield Duplex AJ using the same procedures as described for the annealing of Duplex A. Synthesis of the small molecule: 17-((4-hydroxyphenyl)amino)-17- oxoheptadecanoic acid (AJa):

To a stirred solution of 4-amino phenol (55.6 mg, 0.509 mmol, 1 equiv) and 17-(tert-butoxy)- 17-oxoheptadecanoic acid (200 mg, 0.560 mmol, 1.1 equiv) in CH2Cl2:CH3CN (4 mL) was added DCC (115.7 mg, 0.560 mmol, 1.1 equiv) followed by DMAP (62 mg, 0.560 mmol, 1.1 equiv) at 23
oC. The reaction mixture was stirred at the same temperature for 5 h. The TLC indicated the completion of the starting material acid. The reaction was quenched with water (10 mL) and extracted with EtOAc (3 x 10 mL). The combined organic layer was dried and evaporated, and the obtained residue was purified by flash column chromatography (silica gel, 10 → 40% ethyl acetate in hexanes) to furnish the tert-butyl 17-((4-hydroxyphenyl)amino)-17- oxoheptadecanoate (AJb) (91.1 mg, 0.204 mmol, 40% yield) and 1-(4-aminophenyl) 17-(tert- butyl) heptadecanedioate (AJc) (86.5 mg, 0.193 mmol, 38% yield) as white solids. Their structures were assigned using COSY and HMBC NMR experiments. Phenol (AJb)
1H NMR (500 MHz, CDCl3) ^ 7.28 (d, J = 8.7 Hz, 2H), 7.23 (s, 1H), 6.76 (d, J = 8.5 Hz, 2H), 2.32 (t, J = 7.6 Hz, 2H), 2.20 (t, J = 7.5 Hz, 2H), 1.75 – 1.66 (m, 3H), 1.63 – 1.54 (m, 3H), 1.44 (s, 9H),
FH12501620.1
Attorney Docket: DCY-13025 1.40 – 1.21 (m, 20H) ppm;
13C NMR (101 MHz, CDCl3) ^ 173.9, 172.1, 153.5, 130.4, 122.7, 116.0, 80.3, 37.9, 35.9, 34.0, 29.8, 29.79, 29.74, 29.6, 29.5, 29.49, 29.44, 29.3, 29.2, 28.3, 26.0, 25.3 ppm; Aniline (AJc)
1H NMR (500 MHz, CDCl
3) ^ 6.90 – 6.74 (m, 2H), 6.72 – 6.57 (m, 2H), 3.64 (bs, 2H), 2.50 (t, J = 7.5 Hz, 2H), 2.20 (t, J = 7.5 Hz, 2H), 1.73 (p, J = 7.5 Hz, 2H), 1.57 (p, J = 7.2 Hz, 2H), 1.44 (s, 9H), 1.40 – 1.13 (m, 22H) ppm;
13C NMR (101 MHz, CDCl
3) ^ 173.5, 173.0, 144.2, 143.1, 122.3, 115.8, 35.8, 34.5, 29.9, 29.8, 29.75, 29.74, 29.63, 29.61, 29.5, 29.4, 29.3, 29.2, 28.3, 25.3, 25.2 ppm; To a round bottom flask containing tert-butyl 17-((4-hydroxyphenyl)amino)-17- oxoheptadecanoate (AJb) (70 mg, 0.156 mmol, 1.0 equiv) was added a solution of TFA:CH2Cl2 (1.5mL:2.5mL) at 23
oC, and a reaction mixture was stirred for 2 h. After completion of the reaction, CH2Cl2 and TFA were removed under reduced pressure, and the residue was co-evaporated twice with toluene (3 mL) to remove excess TFA. The obtained residue was purified by flash column chromatography (silica gel, 30 → 100% ethyl acetate in hexanes) to furnish the 17-((4- hydroxyphenyl)amino)-17-oxoheptadecanoic acid (AJa) (48.9 mg, 0.125 mmol, 80% yield) as a foamy white solid. Synthesis of Duplex AK:
FH12501620.1
Attorney Docket: DCY-13025 Conjugated Sense AK was synthesized through a two-step post-syntenic conjugation approach. Step 1- Attachment of Amino-PEG4 Handle: Sense M1 was synthesized with the method above. Step 2- Conjugation to Amino-PEG4 Handle In Eppendorf tube 1, a solution of 16-(4-hydroxyphenyl)hexadecanoic acid (AKa) (5 mg, 0.0142 mmol, 10 equiv) was dissolved in DMF (500 µL) and treated with HATU (4.29 mg, 0.0113 mmol, 8 equiv) and DIPEA (2.5 µL, 0.0142 mmol, 10 equiv) at rt and mixed for 15 minutes. In Eppendorf tube 2, a solution of Conjugated Sense M1 (10.0 mg, 0.00142 mmol, 1 equiv) in 100 µL H2O was treated with DIPEA (2.5 µL, 0.0142 mmol, 10 equiv). The solution in Eppendorf tube 1 was then added to the Eppendorf tube 2 and mixed for 30 minutes using Thermomixer at 40°C. After the reaction was completed indicated by LC-MS analysis, the reaction mixture was diluted with 5 mL of water and purified by reverse phase XBridge C18 column using a 5-95% gradient of 100 mM TEAA in ACN and H
2O. The product fractions were concentrated under reduced pressure using GeneVac. The combined residual solvent was dialyzed against water (2 X), saline (2 X), and water (2 X) using Pierce Thermo Fisher 3k filters. The Amicon membrane was washed with water (3 X 2 mL) and with a last wash of 20% ethanol in water (20 µL). The combined solvents were then lyophilized to afford an amorphous white solid of Conjugated Sense AD (3.0 mg, 0.00051 mmol, 36% yield). Post-synthetic conjugation was realized through an amide coupling reaction. The Conjugated Sense AC was characterized by LCMS. Observed LCMS: 7393. Sense AD was annealed to Antisense B to yield Duplex AD using the same procedures as described for the annealing of Duplex A. a) Synthesis of the small molecule: 16-(4-hydroxyphenyl)hexadecanoic acid
NiI2.xH2O (32 mg, 0.085 mmol, 0.1 equiv) 4,4’-dimethoxy-2, 2’-bipyridine (18 mg, 0.085 mmol, 0.1 equiv), sodium iodide (32 mg, 0.214 mmol, 0.250 equiv), 4-bromo
FH12501620.1
Attorney Docket: DCY-13025 phenol (148 mg, 0.858 mmol, 1.00 equiv), and methyl 16-bromohexadecanoate (300 mg, 0.858 mmol, 1.00 equiv) were weighed on weigh paper and transferred to a dram vial equipped with a magnetic stir bar. Solvent, DMPU (1,3-Dimethyl-3,4,5,6- tetrahydro-2(1H)-pyrimidinone (3.0 mL), pyridine (7 µL, 0.085 mmol, 0.1 equiv), and zinc dust (>10 µm, 112 mg, 1.71 mmol, 2.00 equiv) were added. The reaction vials were capped with a PTFE-faced silicone septum, and the generally green solution was stirred at room temperature for approximately 5 minutes before heating to 80 °C in a reaction block for 12 h. The color of the reaction changes from green to black upon completion. The reaction mixture was filtered and evaporated, and the crude was purified by flash column chromatography (silica gel, 10 → 25% ethyl acetate in hexanes) to furnish the methyl 16-(4-hydroxyphenyl)hexadecanoate (AKb) (124.4 mg, 0.343 mmol, 40% yield) as a white solid.
1H NMR (500 MHz, CDCl3) ^ 7.03 (d, J = 8.4 Hz, 2H), 6.75 (d, J = 8.4 Hz, 2H), 5.23 (s, 1H), 3.67 (s, 3H), 2.52 (t, J = 7.5 Hz, 2H), 2.31 (t, J = 7.5 Hz, 2H), 1.65 – 1.53 (m, 6H), 1.31 – 1.25 (m, 20H) ppm;
13C NMR (126 MHz, CDCl3) ^ 174.9, 153.8, 135.2, 129.6, 115.3, 51.7, 35.2, 34.4, 31.9, 29.9, 29.83, 29.82, 29.81, 29.80, 29.7, 29.68, 29.64, 29.5, 29.4, 29.3, 25.2 ppm; To a stirred solution of methyl 16-(4-hydroxyphenyl)hexadecanoate (AKb) (52 mg, 0.143 mmol, 1.0 equiv) in THF:MeOH (3:1 v/v, 4 mL) was added a solution of LiOH (34.4 mg, 1.434 mmol, 10 equiv) in H
2O (1 mL) at 25
oC. The resulting mixture was stirred at the same temperature for overnight. The pH of the reaction mixture was adjusted to 3-4 using 1N HCl solution. The aqueous layer was extracted with EtOAc (3 × 10 mL) and the combined organic layer was dried over anhydrous Na2SO4, filtered, and concentrated under reduced pressure. The obtained residue was purified by flash column chromatography (silica gel, 30 → 90% ethyl acetate in hexanes) to afford pure 16-(4-hydroxyphenyl)hexadecanoic acid (AKa) (36.9 mg, 0.105 mmol, 74% yield) as a white solid.
1H NMR (400 MHz, CDCl3) ^ 7.06 – 7.01 (m, 2H), 6.74 (d, J = 8.4 Hz, 2H), 2.52 (t, J = 7.5 Hz, 2H), 2.35 (t, J = 7.5 Hz, 2H), 1.67 – 1.49 (m, 6H), 1.36 – 1.19 (m, 20H) ppm;
13C NMR (101 MHz, CDCl3) ^ 176.9, 154.4, 134.4, 129.4, 115.1, 35.2, 34.2, 31.9, 29.74, 29.71, 29.69, 29.62, 29.5, 29.4, 29.3, 29.2, 25.0 ppm; Synthesis of Duplex AL:
FH12501620.1
Attorney Docket: DCY-13025
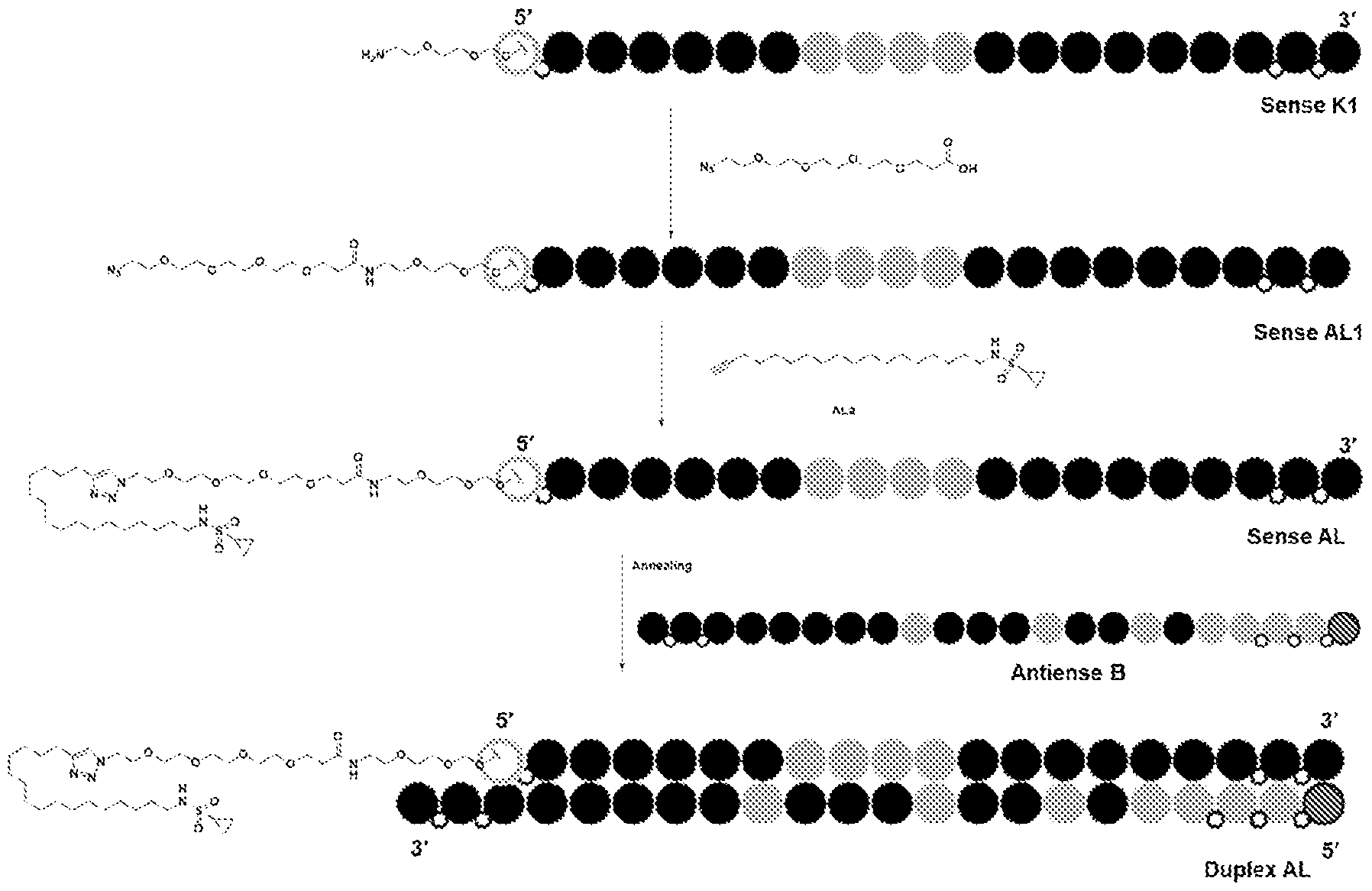
Conjugated Sense AL was synthesized through a two-step post-syntenic conjugation approach. Step 1- Attachment of Azido-PEG4 Handle: In Eppendorf tube 1, a solution of Azido-PEG4-NHS ester (11.4 mg, 0.02934 mmol, 10 equiv) in 500 ^L DMF was prepared at rt. In Eppendorf tube 2, Sense K1 (20 mg, 0.002934 mmol, 1 equiv) was dissolved in 100 ^L water and treated with DIPEA (5 ^L, 0.02934 mmol, 10 equiv). Next, the solution in Eppendorf tube 1 was added to Eppendorf tube 2 and mixed using Thermomixer at 35 ˚C. After 1 hour, the reaction was indicated complete by LC-MS analysis. The reaction mixture was diluted with 10 mL of water and then dialyzed against water (2X) using Amicon® Ultra-15 Centrifugal (10K). The Amicon membrane was washed with water (3 X 2 mL) and the combined solvents were then lyophilized to afford an amorphous white solid of Conjugated Sense AL1 (19.6 mg, 0.00278 mmol, yield: 95%). Observed LCMS: 7068. Step 2- Conjugation to Amino-PEG4 Handle In Eppendorf tube 1, a 1:5 mixture of water and DMA (1000 ^L) was degassed under nitrogen gas for 5 minutes. Next of Sense AL120 mg (20 mg, 0.00283 mmol, 1 equiv) and DIPEA (100 ^L, excess) were added to the mixture of water and DMA in Eppendorf tube 1, followed by addition of N-(octadec-17-yn-1-yl)cyclopropanesulfonamide (ALa) (10.4 mg,
FH12501620.1
Attorney Docket: DCY-13025 0.0283 mmol, 10 equiv) to Eppendorf tube 1. The resulting mixture was kept under Nitrogen gas for another 10 minutes to remove any dissolved oxygen. In Eppendorf tube 2, CuBr.SCH
3 (0.58 mg, 0.00283 mmol, 1 equiv) was dissolved in 200 ^L of previously degassed acetonitrile. Next, the solution in Eppendorf tube 2 was quickly added to Eppendorf tube 1 and mixed using Thermomixer at 40 °C for 3 hours. After the reaction was indicated complete by LC-MS analysis, the reaction mixture was diluted with 15 mL EDTA 0.5 M (pH 8) in a 50mL falcon tube and stirred for 15 minutes at room temperature. The mixture was then dialyzed against water (2X) using Amicon® Ultra- 15 Centrifugal (3K). The combined solvents were purified by revers phase XBridge C18 column using a 5-95% gradient of 100 mM TEAA in ACN and H2O. The product fractions were concentrated under reduced pressure using Genevac. The combined residual solvent was dialyzed against water (1 X), saline (1 X), and water (3 X) using Amicon® Ultra-15 Centrifugal (3K). The Amicon membrane was washed with water (3 X 2 mL) and the combined solvents were then lyophilized to afford an amorphous white solid of Conjugated Sense AL (7.1 mg, 0.000952 mmol, yield: 34%). Observed LCMS: 7458. Sense AL was annealed to Antisense B to yield Duplex AL using the same procedures as described for the annealing of Duplex A. Synthesis of the small molecule: N-(octadec-17-yn-1-yl)cyclopropanesulfonamide (ALa):

To a stirred solution of octadec-17-yn-1-ol (100 mg, 0.375 mmol, 1 equiv) in anhydrous CH2Cl2 (4 mL) at room temperature was added PPh3 (147.5 mg, 0.563 mmol, 1.5 equiv) followed by CBr4 (186.7 mg, 0.563 mmol, 1.5 equiv), and the reaction mixture was stirred at the same temperature for 30 minutes. TLC indicated the complete consumption of starting alcohol. The reaction mixture was quenched with saturated aqueous NaHCO3 solution (10 mL). The aqueous layer was extracted with EtOAc (3 × 10 mL) and the combined organic layer was dried over anhydrous Na
2SO
4, filtered, and concentrated under reduced pressure. The obtained residue was purified by flash column chromatography (silica gel, 2
10%
FH12501620.1
Attorney Docket: DCY-13025 ethyl acetate in hexanes) to afford pure 18-bromooctadec-1-yne (ALb) (113.6 mg, 0.345 mmol, 92% yield) as a white solid.
1H NMR (500 MHz, CDCl
3) ^ 3.40 (t, J = 6.9 Hz, 2H), 2.18 (td, J = 7.1, 2.7 Hz, 2H), 1.93 (t, J = 2.6 Hz, 1H), 1.85 (dt, J = 14.4, 7.0 Hz, 2H), 1.55 – 1.49 (m, 2H), 1.40 (ddd, J = 9.5, 7.1, 4.4 Hz, 2H), 1.27 (d, J = 11.6 Hz, 22H) ppm; To a stirred solution of cyclopropane sulfonamide (22.0 mg, 0.182 mmol, 1.5 equiv) in anhydrous DMF (3 mL) at 0
oC, sodium hydride (55% in mineral oil, 7.4 mg, 0.170 mmol, 1.4 equiv) was added. The resulting mixture was stirred at the same temperature for 30 min. A solution of 18-bromooctadec-1-yne (ALb, 40 mg, 0.121 mmol, 1.0 equiv) in anhydrous DMF (1.5 mL) was added to the reaction mixture and the resulting mixture was stirred at 25 °C for 3 h. The reaction mixture was cooled to 0
oC and carefully quenched by the addition of saturated aqueous NH4Cl solution (10 mL). The aqueous layer was extracted with EtOAc (3 × 10 mL) and the combined organic layer was dried over anhydrous Na
2SO
4, filtered, and concentrated under reduced pressure. The obtained residue was purified by flash column chromatography (silica gel, 2
10% ethyl acetate in hexanes) to afford pure N-(octadec-17- yn-1-yl)cyclopropanesulfonamide (ALa) (29.5 mg, 0.0799 mmol, 66% yield) as a white solid.
1H NMR (400 MHz, CDCl
3) ^ 4.15 (t, J = 6.2 Hz, 1H), 3.15 (q, J = 6.8 Hz, 2H), 2.40 (tt, J = 7.9, 4.8 Hz, 1H), 2.18 (td, J = 7.1, 2.6 Hz, 2H), 1.93 (t, J = 2.7 Hz, 1H), 1.61 – 1.47 (m, 6H), 1.41 – 1.22 (m, 22H), 1.20 – 1.13 (m, 2H), 1.06 – 0.91 (m, 2H) ppm;
13C NMR (101 MHz, CDCl3) ^ 85.1, 68.2, 43.7, 30.5, 30.2, 29.86, 29.83, 29.81, 29.76, 29.71, 29.4, 29.3, 29.0, 28.7, 26.8, 18.6, 5.5 ppm; Synthesis of Duplex AM:
FH12501620.1
Attorney Docket: DCY-13025
Conjugated Sense AG was synthesized through a two-step post-syntenic conjugation approach. Step 1- Attachment of Amino-PEG4 Handle: Sense M1 was synthesized with the method above. Step 2- Conjugation to Amino-PEG4 Handle In Eppendorf tube 1, a solution of 16-(phenylsulfonamido)hexadecanoic acid (AMa) (5 mg, 0.0142 mmol, 10 equiv) was dissolved in DMF (500 µL) and treated with HATU (4.29 mg, 0.0113 mmol, 8 equiv) and DIPEA (2.5 µL, 0.0142 mmol, 10 equiv) at rt and mixed for 15 minutes. In Eppendorf tube 2, a solution of Conjugated Sense M1 (10.0 mg, 0.00142 mmol, 1 equiv) in 100 µL H
2O was treated with DIPEA (2.5 µL, 0.0142 mmol, 10 equiv). The solution in Eppendorf tube 1 was then added to the Eppendorf tube 2 and mixed for 30 minutes using Thermomixer at 40°C. After the reaction was completed indicated by LC-MS analysis, the reaction mixture was diluted with 5 mL of water and purified by reverse phase XBridge C18 column using a 5-95% gradient of 100 mM TEAA in ACN and H
2O. The product fractions were concentrated under reduced pressure using GeneVac. The combined residual solvent was dialyzed against water (2 X), saline (2 X), and water (2 X) using Pierce Thermo Fisher 3k filters. The Amicon membrane was washed with water (3 X 2 mL) and with a last wash of 20% ethanol in water (20 µL). The combined solvents were then lyophilized to afford an amorphous white solid of Conjugated Sense AM (4.2 mg, 0.00054 mmol, 38% yield). Post-synthetic
FH12501620.1
Attorney Docket: DCY-13025 conjugation was realized through an amide coupling reaction. The
Conjugated Sense AM was characterized by LCMS. Observed LCMS: 7456. Sense AM was annealed to Antisense B to yield Duplex AM using the same procedures as described for the annealing of Duplex A. Synthesis of the small molecule: 16-(phenylsulfonamido)hexadecanoic acid
To a stirred solution of benzenesulfonamide (67.4 mg, 0.429 mmol, 1.5 equiv) in anhydrous DMF (3 mL) at 0
oC, sodium hydride (55% in mineral oil, 17.5 mg, 0.401 mmol, 1.4 equiv) was added. The resulting mixture was stirred at the same temperature for 30 min. A solution of methyl 16-bromohexadecanoate (100 mg, 0.286 mmol, 1.0 equiv) in anhydrous DMF (1.5 mL) was added to the reaction mixture and the resulting mixture was stirred at 25 °C for 3 h. The reaction mixture was cooled to 0
oC and carefully quenched by the addition of saturated aqueous NH
4Cl solution (10 mL). The aqueous layer was extracted with EtOAc (3 × 10 mL) and the combined organic layer was dried over anhydrous Na
2SO
4, filtered, and concentrated under reduced pressure. The obtained residue was purified by flash column chromatography (silica gel, 5
15% ethyl acetate in hexanes) to afford pure methyl 16- (phenylsulfonamido)hexadecanoate (AMb) (65.7 mg, 0.154 mmol, 54% yield) as a white solid.
1H NMR (400 MHz, CDCl
3) ^ 7.95 – 7.68 (m, 2H), 7.62 – 7.38 (m, 3H), 4.31 (t, J = 6.3 Hz, 1H), 3.66 (s, 3H), 2.96 (td, J = 7.1, 6.2 Hz, 2H), 2.30 (t, J = 7.5 Hz, 2H), 1.65 – 1.40 (m, 4H), 1.35 – 1.15 (m, 22H) ppm;
13C NMR (101 MHz, CDCl
3) ^ 174.5, 140.2, 132.7, 129.2, 127.2, 51.6, 43.4, 34.3, 29.9, 29.73, 29.70, 29.6, 29.59, 29.56, 29.54, 29.4, 29.3, 29.2, 26.6, 25.1 ppm; To a stirred solution of methyl 16-(phenylsulfonamido)hexadecanoate (AMb) (60 mg, 0.140 mmol, 1.0 equiv) in THF:MeOH (3:1 v/v, 4 mL) was added a solution of LiOH (33.8 mg, 1.409 mmol, 10 equiv) in H2O (1 mL) at 25
oC. The resulting mixture was stirred at the same temperature for 4 h. The pH of the reaction mixture was adjusted to 3-4 using 1N HCl
FH12501620.1
Attorney Docket: DCY-13025 solution. The aqueous layer was extracted with EtOAc (3 × 10 mL) and the combined organic layer was dried over anhydrous Na2SO4, filtered, and concentrated under reduced pressure. The obtained residue was purified by flash column chromatography (silica gel, 20 → 80% ethyl acetate in hexanes) to afford pure 16-(phenylsulfonamido)hexadecanoic acid (AMa) (41.6 mg, 0.101 mmol, 72% yield) as a white solid.
1H NMR (400 MHz, CDCl
3) ^ 7.98 – 7.75 (m, 2H), 7.60 – 7.41 (m, 3H), 4.60 (t, J = 6.1 Hz, 1H), 2.95 (q, J = 6.8 Hz, 2H), 2.35 (t, J = 7.5 Hz, 2H), 1.63 (q, J = 7.3 Hz, 2H), 1.45 (p, J = 7.1 Hz, 2H), 1.35 – 1.15 (m, 22H) ppm;
13C NMR (101 MHz, CDCl
3) ^ 179.4, 140.2, 132.8, 129.3, 127.2, 43.4, 34.1, 29.8, 29.7, 29.67, 29.63, 29.55, 29.51, 29.4, 29.3, 29.2, 26.7, 24.9 ppm Synthesis of Duplex AN:
Conjugated Sense AN was synthesized through a two-step post-syntenic conjugation approach. Step 1- Attachment of Amino-PEG4 Handle: Sense M1 was synthesized with the method above. Step 2- Conjugation to Amino-PEG4 Handle In Eppendorf tube 1, a solution of 16-((4-aminophenyl)sulfonamido)hexadecanoic acid (ANa) (5 mg, 0.0142 mmol, 10 equiv) was dissolved in DMF (500 µL) and treated with HATU (4.29 mg, 0.0113 mmol, 8 equiv) and DIPEA (2.5 µL, 0.0142 mmol, 10 equiv) at rt and mixed
FH12501620.1
Attorney Docket: DCY-13025 for 15 minutes. In Eppendorf tube 2, a solution of
Conjugated Sense M1 (10.0 mg, 0.00142 mmol, 1 equiv) in 100 µL H
2O was treated with DIPEA (2.5 µL, 0.0142 mmol, 10 equiv). The solution in Eppendorf tube 1 was then added to the Eppendorf tube 2 and mixed for 30 minutes using Thermomixer at 40°C. After the reaction was completed indicated by LC-MS analysis, the reaction mixture was diluted with 5 mL of water and purified by reverse phase XBridge C18 column using a 5-95% gradient of 100 mM TEAA in ACN and H2O. The product fractions were concentrated under reduced pressure using GeneVac. The combined residual solvent was dialyzed against water (2 X), saline (2 X), and water (2 X) using Pierce Thermo Fisher 3k filters. The Amicon membrane was washed with water (3 X 2 mL) and with a last wash of 20% ethanol in water (20 µL). The combined solvents were then lyophilized to afford an amorphous white solid of Conjugated Sense AN (5.4 mg, 0.00077 mmol, 51% yield). Post-synthetic conjugation was realized through an amide coupling reaction. The Conjugated Sense AN was characterized by LCMS. Observed LCMS: 7471. Sense AN was annealed to Antisense B to yield Duplex AN using the same procedures as described for the annealing of Duplex A. a) Synthesis of the small molecule: 16-((4-aminophenyl)sulfonamido)hexadecanoic

To a stirred solution of 4-aminobenzenesulfonamide (58.9 mg, 0.342 mmol, 1.5 equiv) in anhydrous DMF (3 mL) at 0
oC, sodium hydride (55% in mineral oil, 14.0 mg, 0.320 mmol, 1.4 equiv) was added. The resulting mixture was stirred at the same temperature for 30 min. A solution of methyl 16-bromohexadecanoate (80 mg, 0.228 mmol, 1.0 equiv) in anhydrous DMF (1.5 mL) was added to the reaction mixture and the resulting mixture was stirred at 25 °C for 3 h. The reaction mixture was cooled to 0
oC and carefully quenched by the addition of saturated aqueous NH4Cl solution (10 mL). The aqueous layer was extracted with EtOAc (3 × 10 mL) and the combined organic layer was dried over anhydrous Na2SO4, filtered, and concentrated under reduced pressure. The obtained residue was purified by flash column
FH12501620.1
Attorney Docket: DCY-13025 chromatography (silica gel, 5 → 20% ethyl acetate in hexanes) to afford pure methyl 16-((4- aminophenyl)sulfonamido)hexadecanoate (ANb) (42.2 mg, 0.095 mmol, 42% yield) as a white solid.
1H NMR (500 MHz, CDCl
3) ^ 7.55 (d, J = 7.6 Hz, 2H), 6.62 (d, J = 7.6 Hz, 2H), 4.73 (t, J = 6.2 Hz, 1H), 4.28 (brs, 2H), 3.61 (s, 3H), 2.85 – 2.79 (m, 2H), 2.25 (t, J = 7.5 Hz, 2H), 1.56 (p, J = 7.3 Hz, 2H), 1.40 (q, J = 7.1 Hz, 2H), 1.32 – 1.09 (m, 22H) ppm;
13C NMR (126 MHz, CDCl
3) ^ 174.4, 150.8, 129.2, 128.0, 114.1, 51.5, 43.3, 34.2, 31.5, 29.69, 29.65, 29.63, 29.60, 29.54, 29.51, 29.3, 29.22, 29.20, 26.7, 25.0 ppm; To a stirred solution of methyl 16-((4-aminophenyl)sulfonamido)hexadecanoate (ANb) (55 mg, 0.124 mmol, 1.0 equiv) in THF:MeOH (3:1 v/v, 4 mL) was added a solution of LiOH (29.9 mg, 1.248 mmol, 10 equiv) in H2O (1 mL) at 25
oC. The resulting mixture was stirred at the same temperature for 4h. The pH of the reaction mixture was adjusted to 3-4 using 1N HCl solution. The aqueous layer was extracted with EtOAc (3 × 50 mL) and the combined organic layer was dried over anhydrous Na2SO4, filtered, and concentrated under reduced pressure. The obtained residue was purified by flash column chromatography (silica gel, 30 100% ethyl acetate in hexanes) to afford pure 16-((4- aminophenyl)sulfonamido)hexadecanoic acid (ANa) (45.0 mg, 0.105 mmol, 85% yield) as a white solid.
1H NMR (400 MHz, MeOD) ^ 7.51 (d, J = 7.6 Hz, 2H), 6.69 (d, J = 7.6 Hz, 2H), 2.77 (t, J = 7.0 Hz, 2H), 2.28 (t, J = 7.4 Hz, 2H), 1.59 (q, J = 7.2 Hz, 2H), 1.41 (t, J = 6.7 Hz, 2H), 1.35 – 1.18 (m, 22H) ppm;
13C NMR (101 MHz, MeOD) ^ 177.9, 154.1, 130.1, 127.8, 114.6, 44.2, 35.1, 30.89, 30.85, 30.83, 30.7, 30.6, 30.5, 30.4, 30.3, 27.9, 26.3 ppm; Synthesis of Duplex AO:
FH12501620.1
Attorney Docket: DCY-13025

Conjugated Sense AO was synthesized through a two-step post-syntenic conjugation approach. Step 1- Attachment of Amino-PEG4 Handle: Sense M1 was synthesized with the method above. Step 2- Conjugation to Amino-PEG4 Handle In Eppendorf tube 1, a solution of 16-((4-methylpyridine)-2-sulfonamido)hexadecanoic acid (AOa) (5 mg, 0.0142 mmol, 10 equiv) was dissolved in DMF (500 µL) and treated with HATU (4.29 mg, 0.0113 mmol, 8 equiv) and DIPEA (2.5 µL, 0.0142 mmol, 10 equiv) at rt and mixed for 15 minutes. In Eppendorf tube 2, a solution of Conjugated Sense M1 (10.0 mg, 0.00142 mmol, 1 equiv) in 100 µL H2O was treated with DIPEA (2.5 µL, 0.0142 mmol, 10 equiv). The solution in Eppendorf tube 1 was then added to the Eppendorf tube 2 and mixed for 30 minutes using Thermomixer at 40°C. After the reaction was completed indicated by LC-MS analysis, the reaction mixture was diluted with 5 mL of water and purified by reverse phase XBridge C18 column using a 5-95% gradient of 100 mM TEAA in ACN and H
2O. The product fractions were concentrated under reduced pressure using GeneVac. The combined residual solvent was dialyzed against water (2 X), saline (2 X), and water (2 X) using Pierce Thermo Fisher 3k filters. The Amicon membrane was washed with water (3 X 2 mL) and with a last wash of 20% ethanol in water (20 µL). The combined solvents were then lyophilized to afford an amorphous white solid of Conjugated Sense AO (4.0 mg, 0.00054 mmol, 38% yield). Post-synthetic
FH12501620.1
Attorney Docket: DCY-13025 conjugation was realized through an amide coupling reaction. The
Conjugated Sense AO was characterized by LCMS. Observed LCMS: 7471. Sense AO was annealed to Antisense B to yield Duplex AO using the same procedures as described for the annealing of Duplex A. Synthesis of the small molecule: 16-((4-methylpyridine)-2-
To a stirred solution of 4-methylpyridine-2-sulfonamide (57.1 mg, 0.322 mmol, 1.5 equiv) in anhydrous DMF (2 mL) at 0
oC, sodium hydride (55% in mineral oil, 13.1 mg, 0.299 mmol, 1.4 equiv) was added. The resulting mixture was stirred at the same temperature for 30 min. A solution of methyl 16-bromohexadecanoate (75 mg, 0.214 mmol, 1.0 equiv) in anhydrous DMF (1 mL) was added to the reaction mixture and the resulting mixture was stirred at 25 °C for 3 h. The reaction mixture was cooled to 0
oC and carefully quenched by the addition of saturated aqueous NH4Cl solution (10 mL). The aqueous layer was extracted with EtOAc (3 × 10 mL) and the combined organic layer was dried over anhydrous Na2SO4, filtered, and concentrated under reduced pressure. The obtained residue was purified by flash column chromatography (silica gel, 5
20% ethyl acetate in hexanes) to afford pure methyl 16-((4- methylpyridine)-2-sulfonamido)hexadecanoate (AOb) (61.3 mg, 0.160 mmol, 65% yield) as a white solid.
1H NMR (500 MHz, CDCl3) ^ 8.51 (d, J = 2.1 Hz, 1H), 7.89 (d, J = 7.9 Hz, 1H), 7.68 (dd, J = 8.0, 2.2 Hz, 1H), 5.09 (t, J = 6.1 Hz, 1H), 3.65 (s, 3H), 2.98 (q, J = 6.8 Hz, 2H), 2.43 (s, 3H), 2.29 (t, J = 7.5 Hz, 2H), 1.60 (p, J = 7.3 Hz, 2H), 1.45 (p, J = 7.1 Hz, 2H), 1.34 – 1.18 (m, 22H) ppm;
13C NMR (126 MHz, CDCl3) ^ 174.5, 154.9, 150.6, 138.2, 137.2, 122.1, 51.5, 43.8, 34.2, 29.9, 29.8, 29.7, 29.68, 29.63, 29.56, 29.54, 29.4, 29.3, 29.2, 26.6, 25.1, 18.6 ppm; To a stirred solution of methyl 16-((4-methylpyridine)-2-sulfonamido)hexadecanoate (AOb) (40 mg, 0.091 mmol, 1.0 equiv) in THF:MeOH (3:1 v/v, 3 mL) was added a solution of LiOH
FH12501620.1
Attorney Docket: DCY-13025 (21.7 mg, 0.908 mmol, 10 equiv) in H2O (0.8 mL) at 25
oC. The resulting mixture was stirred at the same temperature for 3 h. The pH of the reaction mixture was adjusted to 3-4 using 1N HCl solution. The aqueous layer was extracted with EtOAc (3 × 8 mL) and the combined organic layer was dried over anhydrous Na
2SO
4, filtered, and concentrated under reduced pressure. The obtained residue was purified by flash column chromatography (silica gel, 40 → 90% ethyl acetate in hexanes) to afford pure 16-((4-methylpyridine)-2- sulfonamido)hexadecanoic acid (AOa) (30.3 g, 0.071 mmol, 78% yield) as a white solid.
1H NMR (400 MHz, CDCl
3) ^ 8.51 (d, J = 2.1 Hz, 1H), 7.92 (d, J = 7.9 Hz, 1H), 7.72 (ddd, J = 7.9, 2.2, 0.9 Hz, 1H), 5.50 (t, J = 6.1 Hz, 1H), 2.97 (q, J = 6.8 Hz, 2H), 2.45 (s, 3H), 2.36 (t, J = 7.4 Hz, 2H), 1.63 (q, J = 7.4 Hz, 2H), 1.46 (p, J = 7.1 Hz, 2H), 1.39 – 1.16 (m, 22H) ppm; Synthesis of Duplex AP:
Conjugated Sense AP was synthesized through a two-step post-syntenic conjugation approach. Step 1- Attachment of Amino-PEG4 Handle: Sense M1 was synthesized with the method above. Step 2- Conjugation to Amino-PEG4 Handle In Eppendorf tube 1, a solution of 16-((2,5-dichlorothiophene)-3-sulfonamido)hexadecanoic acid (APa) (5 mg, 0.0142 mmol, 10 equiv) was dissolved in DMF (500 µL) and treated with
FH12501620.1
Attorney Docket: DCY-13025 HATU (4.29 mg, 0.0113 mmol, 8 equiv) and DIPEA (2.5 µL, 0.0142 mmol, 10 equiv) at rt and mixed for 15 minutes. In Eppendorf tube 2, a solution of
Conjugated Sense M1 (10.0 mg, 0.00142 mmol, 1 equiv) in 100 µL H
2O was treated with DIPEA (2.5 µL, 0.0142 mmol, 10 equiv). The solution in Eppendorf tube 1 was then added to the Eppendorf tube 2 and mixed for 30 minutes using Thermomixer at 40°C. After the reaction was completed indicated by LC- MS analysis, the reaction mixture was diluted with 5 mL of water and purified by reverse phase XBridge C18 column using a 5-95% gradient of 100 mM TEAA in ACN and H2O. The product fractions were concentrated under reduced pressure using GeneVac. The combined residual solvent was dialyzed against water (2 X), saline (2 X), and water (2 X) using Pierce Thermo Fisher 3k filters. The Amicon membrane was washed with water (3 X 2 mL) and with a last wash of 20% ethanol in water (20 µL). The combined solvents were then lyophilized to afford an amorphous white solid of Conjugated Sense AP (1.1 mg, 0.00014 mmol, 10% yield). Post- synthetic conjugation was realized through an amide coupling reaction. The
Conjugated Sense AP was characterized by LCMS. Observed LCMS: 7531. Sense AP was annealed to Antisense B to yield Duplex AP using the same procedures as described for the annealing of Duplex A. Synthesis of the small molecule: 16-((2,5-dichlorothiophene)-3-
To a stirred solution of 2,5-dichlorothiophene-3-sulfonamide (139.6 mg, 0.601 mmol, 1.5 equiv) in anhydrous DMF (3 mL) at 0
oC, sodium hydride (55% in mineral oil, 24.5 mg, 0.561 mmol, 1.4 equiv) was added. The resulting mixture was stirred at the same temperature for 30 min. A solution of methyl 16-bromohexadecanoate (140 mg, 0.401 mmol, 1.0 equiv) in anhydrous DMF (2 mL) was added to the reaction mixture and the resulting mixture was stirred at 25 °C for 3 h. The reaction mixture was cooled to 0
oC and carefully quenched by the addition of saturated aqueous NH4Cl solution (10 mL). The aqueous layer was extracted
FH12501620.1
Attorney Docket: DCY-13025 with EtOAc (3 × 10 mL) and the combined organic layer was dried over anhydrous Na
2SO
4, filtered, and concentrated under reduced pressure. The obtained residue was purified by flash column chromatography (silica gel, 8
20% ethyl acetate in hexanes) to afford pure methyl 16-((2,5-dichlorothiophene)-3-sulfonamido)hexadecanoate (APb) (136.5 g, 0.272 mmol, 68% yield) as a white solid:
1H NMR (400 MHz, CDCl
3) ^ 7.14 (s, 1H), 4.65 (t, J = 6.1 Hz, 1H), 3.67 (s, 3H), 3.04 (q, J = 6.8 Hz, 2H), 2.30 (t, J = 7.6 Hz, 2H), 1.63 (q, J = 7.4 Hz, 2H), 1.50 (q, J = 7.1 Hz, 2H), 1.36 – 1.18 (m, 22H) ppm;
13C NMR (101 MHz, CDCl
3) ^ 174.5, 136.7, 129.3, 127.5, 126.9, 51.6, 43.4, 34.3, 29.8, 29.71, 29.70, 29.6, 29.5, 29.4, 29.3, 29.2, 26.6, 25.1 ppm; To a stirred solution of methyl 16-((2,5-dichlorothiophene)-3-sulfonamido)hexadecanoate (APb) (60 mg, 0.119 mmol, 1.0 equiv) in THF:MeOH (3:1 v/v, 4 mL) was added a solution of LiOH (28.7 mg, 1.198 mmol, 10 equiv) in H2O (1 mL) at 25
oC. The resulting mixture was stirred at the same temperature for 4h. The pH of the reaction mixture was adjusted to 3-4 using 1N HCl solution. The aqueous layer was extracted with EtOAc (3 × 10 mL) and the combined organic layer was dried over anhydrous Na2SO4, filtered, and concentrated under reduced pressure. The obtained residue was purified by flash column chromatography (silica gel, 20 80% ethyl acetate in hexanes) to afford pure 16-((2,5-dichlorothiophene)-3- sulfonamido)hexadecanoic acid (APa) (40.5 mg, 0.083 mmol, 70% yield) as a white solid:
1H NMR (400 MHz, CDCl
3) ^ 7.13 (s, 1H), 4.87 (t, J = 6.1 Hz, 1H), 3.04 (q, J = 6.8 Hz, 2H), 2.35 (t, J = 7.5 Hz, 2H), 1.63 (p, J = 7.4 Hz, 2H), 1.51 (p, J = 7.1 Hz, 2H), 1.35 – 1.20 (m, 22H) ppm;
13C NMR (101 MHz, CDCl
3) ^ 178.4, 136.8, 129.4, 127.6, 127.0, 43.5, 33.9, 29.9, 29.8, 29.69, 29.65, 29.57, 29.50, 29.4, 29.3, 29.2, 26.7, 24.9 ppm; Synthesis of Duplex AQ:
FH12501620.1
Attorney Docket: DCY-13025

Conjugated Sense AQ was synthesized through a two-step post-syntenic conjugation approach. Step 1- Attachment of Amino-PEG4 Handle: Sense M1 was synthesized with the method above. Step 2- Conjugation to Amino-PEG4 Handle In Eppendorf tube 1, a solution of 16-(4-sulfamoylphenoxy)hexadecanoic acid (AQa) (5 mg, 0.0142 mmol, 10 equiv) was dissolved in DMF (500 µL) and treated with HATU (4.29 mg, 0.0113 mmol, 8 equiv) and DIPEA (2.5 µL, 0.0142 mmol, 10 equiv) at rt and mixed for 15 minutes. In Eppendorf tube 2, a solution of Conjugated Sense M1 (10.0 mg, 0.00142 mmol, 1 equiv) in 100 µL H2O was treated with DIPEA (2.5 µL, 0.0142 mmol, 10 equiv). The solution in Eppendorf tube 1 was then added to the Eppendorf tube 2 and mixed for 30 minutes using Thermomixer at 40°C. After the reaction was completed indicated by LC-MS analysis, the reaction mixture was diluted with 5 mL of water and purified by reverse phase XBridge C18 column using a 5-95% gradient of 100 mM TEAA in ACN and H
2O. The product fractions were concentrated under reduced pressure using GeneVac. The combined residual solvent was dialyzed against water (2 X), saline (2 X), and water (2 X) using Pierce Thermo Fisher 3k filters. The Amicon membrane was washed with water (3 X 2 mL) and with a last wash of 20% ethanol in water (20 µL). The combined solvents were then lyophilized to afford an amorphous white solid of Conjugated Sense AQ (2.2 mg, 0.00030 mmol, 21% yield). Post-synthetic
FH12501620.1
Attorney Docket: DCY-13025 conjugation was realized through an amide coupling reaction. The
Conjugated Sense AQ was characterized by LCMS. Observed LCMS: 7472. Sense AQ was annealed to Antisense B to yield Duplex AQ using the same procedures as described for the annealing of Duplex A. Synthesis of the small molecule: 16-(4-sulfamoylphenoxy)hexadecanoic acid
To a stirred solution of 4-hydroxybenzenesulfonamide (89.1 mg, 0.514 mmol, 1.5 equiv) in anhydrous DMF (3 mL) at 0
oC, sodium hydride (55% in mineral oil, 21.0 mg, 0.481 mmol, 1.4 equiv) was added. The resulting mixture was stirred at the same temperature for 30 min. A solution of methyl 16-bromohexadecanoate (120 mg, 0.343 mmol, 1.0 equiv) in anhydrous DMF (1.5 mL) was added to the reaction mixture and the resulting mixture was stirred at 25 °C for 3 h. The reaction mixture was cooled to 0
oC and carefully quenched by the addition of saturated aqueous NH4Cl solution (10 mL). The aqueous layer was extracted with EtOAc (3 × 10 mL) and the combined organic layer was dried over anhydrous Na2SO4, filtered, and concentrated under reduced pressure. The obtained residue was purified by flash column chromatography (silica gel, 5
20% ethyl acetate in hexanes) to afford pure methyl 16-(4- sulfamoylphenoxy)hexadecanoate (AQb) (93.9 mg, 0.212 mmol, 62% yield) as a white solid:
1H NMR (400 MHz, CDCl3) ^ 7.84 (d, J = 8.9 Hz, 2H), 6.96 (d, J = 8.9 Hz, 2H), 4.78 (s, 2H), 4.01 (t, J = 6.6 Hz, 2H), 3.66 (s, 3H), 2.30 (t, J = 7.6 Hz, 2H), 1.85 – 1.73 (m, 2H), 1.60 (q, J = 7.1 Hz, 2H), 1.51 – 1.40 (m, 2H), 1.35 – 1.23 (m, 20H) ppm;
13C NMR (101 MHz, CDCl
3) ^ 174.6, 162.8, 133.5, 128.8, 114.9, 68.7, 51.7, 34.3, 29.8, 29.78, 29.74, 29.71, 29.6, 29.5, 29.4, 29.3, 29.2, 26.1, 25.2 ppm; To a stirred solution of methyl 16-(4-sulfamoylphenoxy)hexadecanoate (AQb) (40 mg, 0.0905 mmol, 1.0 equiv) in THF:MeOH (3:1 v/v, 4 mL) was added a solution of LiOH (21.8 mg, 0.905 mmol, 10 equiv) in H2O (1 mL) at 25
oC. The resulting mixture was stirred at the
FH12501620.1
Attorney Docket: DCY-13025 same temperature for 4 h. The pH of the reaction mixture was adjusted to 3-4 using 1N HCl solution. The aqueous layer was extracted with EtOAc (3 × 10 mL) and the combined organic layer was dried over anhydrous Na
2SO
4, filtered, and concentrated under reduced pressure. The obtained residue was purified by flash column chromatography (silica gel, 30 → 90% ethyl acetate in hexanes) to afford pure 16-(4-sulfamoylphenoxy)hexadecanoic acid (AQa) (25.5 mg, 0.0597 mmol, 66% yield) as a white solid:
1H NMR (500 MHz, MeOD) ^ 7.81 (d, J = 8.9 Hz, 2H), 7.02 (d, J = 8.9 Hz, 2H), 4.04 (t, J = 6.4 Hz, 2H), 2.27 (t, J = 7.4 Hz, 2H), 1.84 – 1.73 (m, 2H), 1.60 (q, J = 7.2 Hz, 2H), 1.53 – 1.42 (m, 2H), 1.40 – 1.26 (m, 20H) ppm; Synthesis of Duplex AR:
Conjugated Sense AR was synthesized through a two-step post-syntenic conjugation approach. Step 1- Attachment of Amino-PEG4 Handle:
Sense M1 was synthesized with the method above. Step 2- Conjugation to Amino-PEG4 Handle
In Eppendorf tube 1, a solution of 16-((1-methylethyl)sulfonamido)hexadecanoic acid (ARa) (5 mg, 0.0142 mmol, 10 equiv) was dissolved in DMF (500 µL) and treated with HATU (4.29 mg, 0.0113 mmol, 8 equiv) and DIPEA (2.5 µL, 0.0142 mmol, 10 equiv) at rt and mixed
FH12501620.1
Attorney Docket: DCY-13025 for 15 minutes. In Eppendorf tube 2, a solution of
Conjugated Sense M1 (10.0 mg, 0.00142 mmol, 1 equiv) in 100 µL H
2O was treated with DIPEA (2.5 µL, 0.0142 mmol, 10 equiv). The solution in Eppendorf tube 1 was then added to the Eppendorf tube 2 and mixed for 30 minutes using Thermomixer at 40°C. After the reaction was completed indicated by LC-MS analysis, the reaction mixture was diluted with 5 mL of water and purified by reverse phase XBridge C18 column using a 5-95% gradient of 100 mM TEAA in ACN and H2O. The product fractions were concentrated under reduced pressure using GeneVac. The combined residual solvent was dialyzed against water (2 X), saline (2 X), and water (2 X) using Pierce Thermo Fisher 3k filters. The Amicon membrane was washed with water (3 X 2 mL) and with a last wash of 20% ethanol in water (20 µL). The combined solvents were then lyophilized to afford an amorphous white solid of Conjugated Sense AR (3.2 mg, 0.00044 mmol, 31% yield). Post-synthetic conjugation was realized through an amide coupling reaction. The Conjugated Sense AR was characterized by LCMS. Observed LCMS: 7422. Sense AR was annealed to Antisense B to yield Duplex AR using the same procedures as described for the annealing of Duplex A. Synthesis of the small molecule: 16-((1-methylethyl)sulfonamido)hexadecanoic acid (ARa):
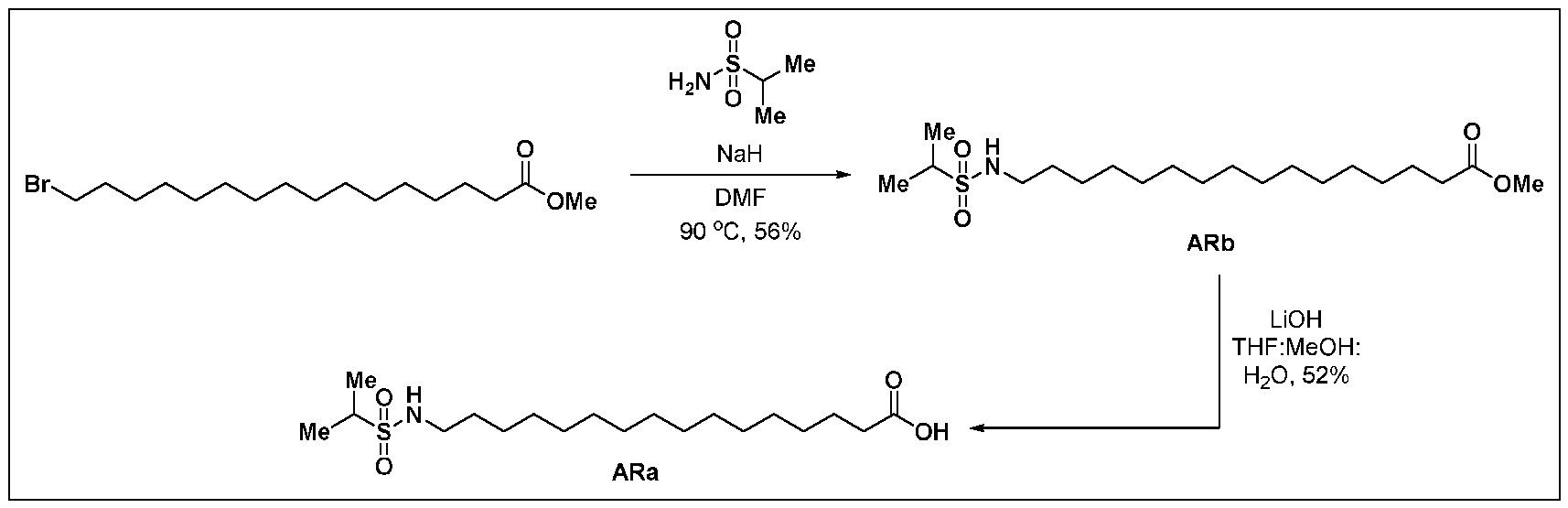
To a stirred solution of iso-propane sulfonamide (52.9 mg, 0.429 mmol, 1.5 equiv) in anhydrous DMF (4 mL) at 0
oC, sodium hydride (55% in mineral oil, 17.5 mg, 0.400 mmol, 1.4 equiv) was added. The resulting mixture was stirred at the same temperature for 30 min. A solution of methyl 16-bromohexadecanoate (100 mg, 0.286 mmol, 1.0 equiv) in anhydrous DMF (2 mL) was added to the reaction mixture and the resulting mixture was stirred at 25 °C for 3 h. The reaction mixture was cooled to 0
oC and carefully quenched by the addition of saturated aqueous NH
4Cl solution (10 mL). The aqueous layer was extracted with EtOAc (3 × 10 mL) and the combined organic layer was dried over anhydrous Na2SO4, filtered, and
FH12501620.1
Attorney Docket: DCY-13025 concentrated under reduced pressure. The obtained residue was purified by flash column chromatography (silica gel, 5
20% ethyl acetate in hexanes) to afford pure Methyl 16-((1- methylethyl)sulfonamido)hexadecanoate (ARb) (62.7 mg, 0.160 mmol, 56% yield) as a white solid.
1H NMR (400 MHz, CDCl
3) ^ 4.19 (t, J = 6.1 Hz, 1H), 3.65 (s, 3H), 3.22 – 3.04 (m, 3H), 2.29 (t, J = 7.6 Hz, 2H), 1.74 – 1.50 (m, 4H), 1.36 (d, J = 6.8 Hz, 6H), 1.33 – 1.21 (m, 22H) ppm;
13C NMR (126 MHz, CDCl
3) ^ 174.5, 53.4, 51.5, 43.9, 34.2, 30.8, 29.72, 29.71, 29.68, 29.65, 29.60, 29.5, 29.4, 29.3, 29.2, 26.7, 25.1, 16.8 ppm; To a stirred solution of Methyl 16-((1-methylethyl)sulfonamido)hexadecanoate (ARb) (40 mg, 0.102 mmol, 1.0 equiv) in THF:MeOH (3:1 v/v, 3 mL) was added a solution of LiOH (24.5 mg, 1.021 mmol, 10 equiv) in H2O (0.8 mL) at 25
oC. The resulting mixture was stirred at the same temperature for 4h. The pH of the reaction mixture was adjusted to 3-4 using 1N HCl solution. The aqueous layer was extracted with EtOAc (3 × 8 mL) and the combined organic layer was dried over anhydrous Na2SO4, filtered, and concentrated under reduced pressure. The obtained residue was purified by flash column chromatography (silica gel, 20 80% ethyl acetate in hexanes) to afford pure 16-((1- methylethyl)sulfonamido)hexadecanoic acid (ARa) (3.52 g, 2.002 mmol, 52% yield) as a white solid.
1H NMR (500 MHz, CDCl3) ^ 4.28 (t, J = 6.1 Hz, 1H), 3.25 – 3.01 (m, 3H), 2.35 (t, J = 7.5 Hz, 2H), 1.67 – 1.51 (m, 4H), 1.37 (d, J = 6.8 Hz, 6H), 1.35 – 1.23 (m, 22H) ppm;
13C NMR (126 MHz, CDCl
3) ^ 179.5, 53.5, 44.0, 34.2, 30.9, 29.7, 29.69, 29.68, 29.64, 29.5, 29.4, 29.3, 29.2, 26.8, 24.9, 16.9 ppm; Synthesis of Duplex AS:
FH12501620.1
Attorney Docket: DCY-13025
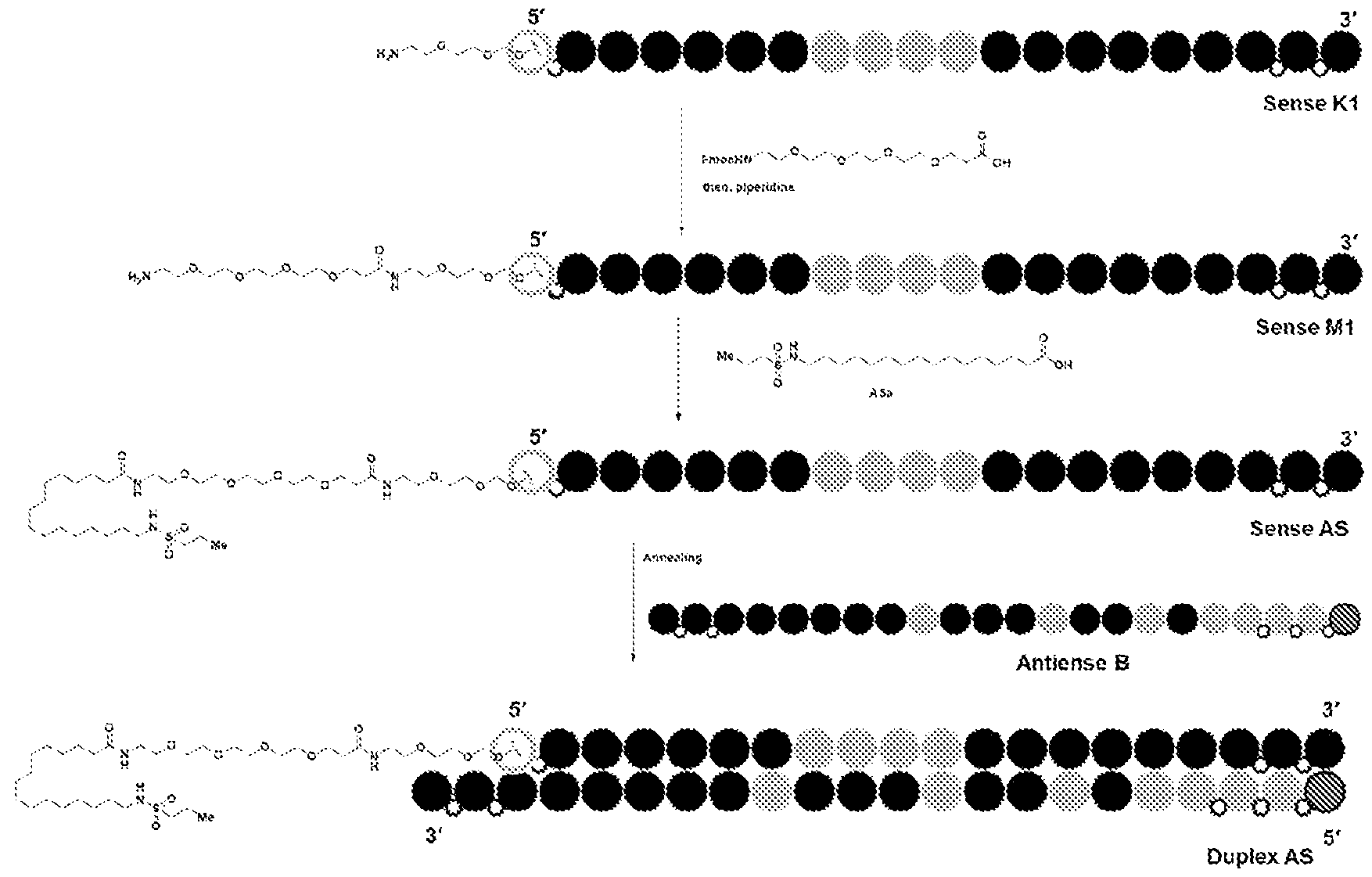
Conjugated Sense AS was synthesized through a two-step post-syntenic conjugation approach. Step 1- Attachment of Amino-PEG4 Handle: Sense M1 was synthesized with the method above. Step 2- Conjugation to Amino-PEG4 Handle In Eppendorf tube 1, a solution of 16-(propylsulfonamido)hexadecanoic acid (ASa) (5 mg, 0.0142 mmol, 10 equiv) was dissolved in DMF (500 µL) and treated with HATU (4.29 mg, 0.0113 mmol, 8 equiv) and DIPEA (2.5 µL, 0.0142 mmol, 10 equiv) at rt and mixed for 15 minutes. In Eppendorf tube 2, a solution of Conjugated Sense M1 (10.0 mg, 0.00142 mmol, 1 equiv) in 100 µL H2O was treated with DIPEA (2.5 µL, 0.0142 mmol, 10 equiv). The solution in Eppendorf tube 1 was then added to the Eppendorf tube 2 and mixed for 30 minutes using Thermomixer at 40°C. After the reaction was completed indicated by LC-MS analysis, the reaction mixture was diluted with 5 mL of water and purified by reverse phase XBridge C18 column using a 5-95% gradient of 100 mM TEAA in ACN and H
2O. The product fractions were concentrated under reduced pressure using GeneVac. The combined residual solvent was dialyzed against water (2 X), saline (2 X), and water (2 X) using Pierce Thermo Fisher 3k filters. The Amicon membrane was washed with water (3 X 2 mL) and with a last wash of 20% ethanol in water (20 µL). The combined solvents were then lyophilized to afford an amorphous white solid of Conjugated Sense AS (2.2 mg, 0.00030 mmol, 21% yield). Post-synthetic
FH12501620.1
Attorney Docket: DCY-13025 conjugation was realized through an amide coupling reaction. The
Conjugated Sense AS was characterized by LCMS. Observed LCMS: 7422. Sense AS was annealed to Antisense B to yield Duplex AS using the same procedures as described for the annealing of Duplex A. Synthesis of the small molecule: 16-(propylsulfonamido)hexadecanoic acid (ASa):
To a stirred solution of n-propane sulfonamide (79.3 mg, 0.644 mmol, 1.5 equiv) in anhydrous DMF (5 mL) at 0
oC, sodium hydride (55% in mineral oil, 26.2 mg, 0.601 mmol, 1.4 equiv) was added. The resulting mixture was stirred at the same temperature for 30 min. A solution of methyl 16-bromohexadecanoate (150 mg, 0.429 mmol, 1.0 equiv) in anhydrous DMF (5 mL) was added to the reaction mixture and the resulting mixture was stirred at 25 °C for 3 h. The reaction mixture was cooled to 0
oC and carefully quenched by the addition of saturated aqueous NH4Cl solution (10 mL). The aqueous layer was extracted with EtOAc (3 × 10 mL) and the combined organic layer was dried over anhydrous Na2SO4, filtered, and concentrated under reduced pressure. The obtained residue was purified by flash column chromatography (silica gel, 5
20% ethyl acetate in hexanes) to afford pure Methyl 16- (propylsulfonamido)hexadecanoate (ASb) (126 mg, 0.321 mmol, 75% yield) as a white solid.
1H NMR (500 MHz, CDCl3) ^ 4.03 (t, J = 6.8 Hz, 1H), 3.67 (s, 3H), 3.10 (q, J = 6.8 Hz, 2H), 3.01 – 2.95 (m, 2H), 2.30 (t, J = 7.6 Hz, 2H), 1.88 – 1.79 (m, 2H), 1.62 (t, J = 7.3 Hz, 2H), 1.58 –1.52 (m, 4H), 1.35 – 1.22 (m, 20H), 1.07 (t, J = 7.4 Hz, 3H) ppm;
13C NMR (126 MHz, CDCl3) ^ 174.6, 54.6, 51.7, 43.6, 34.3, 30.6, 29.9, 29.81, 29.80, 29.78, 29.74, 29.69, 29.64, 29.5, 29.4, 26.8, 25.2, 17.7, 13.2 ppm; To a stirred solution of Methyl 16-(propylsulfonamido)hexadecanoate (ASb) (80 mg, 0.204 mmol, 1.0 equiv) in THF:MeOH (3:1 v/v, 4 mL) was added a solution of LiOH (48.9 mg, 2.042 mmol, 10 equiv) in H
2O (1 mL) at 25
oC. The resulting mixture was stirred at the same temperature for 4 h. The pH of the reaction mixture was adjusted to 3-4 using 1N HCl solution. The aqueous layer was extracted with EtOAc (3 × 10 mL) and the combined organic
FH12501620.1
Attorney Docket: DCY-13025 layer was dried over anhydrous Na2SO4, filtered, and concentrated under reduced pressure. The obtained residue was purified by flash column chromatography (silica gel, 20 → 80% ethyl acetate in hexanes) to afford pure 16-(propylsulfonamido)hexadecanoic acid (ASa) (63.2 mg, 0.167 mmol, 82% yield) as a white solid.
1H NMR (500 MHz, MeOD) ^ 3.05 – 2.96 (m, 4H), 2.25 (dd, J = 8.2, 6.7 Hz, 2H), 1.85 – 1.74 (m, 2H), 1.63 – 1.49 (m, 4H), 1.35 – 1,27 (m, 22H), 1.06 (t, J = 7.5 Hz, 3H) ppm;
13C NMR (126 MHz, MeOD) ^ 178.92, 54.55, 43.94, 35.98, 31.43, 30.78, 30.76, 30.72, 30.70, 30.67, 30.64, 30.48, 30.39, 30.35, 27.73, 26.50, 18.41, 13.26 ppm; Synthesis of Duplex AT:
Conjugated Sense AT was synthesized through a two-step post-syntenic conjugation approach. Step 1- Attachment of Amino-PEG4 Handle: Sense M1 was synthesized with the method above. Step 2- Conjugation to Amino-PEG4 Handle In Eppendorf tube 1, a solution of 16-(butylsulfonamido)hexadecanoic acid (ATa) (5 mg, 0.0142 mmol, 10 equiv) was dissolved in DMF (500 µL) and treated with HATU (4.29 mg, 0.0113 mmol, 8 equiv) and DIPEA (2.5 µL, 0.0142 mmol, 10 equiv) at rt and mixed for 15
FH12501620.1
Attorney Docket: DCY-13025 minutes. In Eppendorf tube 2, a solution of
Conjugated Sense M1 (10.0 mg, 0.00142 mmol, 1 equiv) in 100 µL H
2O was treated with DIPEA (2.5 µL, 0.0142 mmol, 10 equiv). The solution in Eppendorf tube 1 was then added to the Eppendorf tube 2 and mixed for 30 minutes using Thermomixer at 40°C. After the reaction was completed indicated by LC-MS analysis, the reaction mixture was diluted with 5 mL of water and purified by reverse phase XBridge C18 column using a 5-95% gradient of 100 mM TEAA in ACN and H2O. The product fractions were concentrated under reduced pressure using GeneVac. The combined residual solvent was dialyzed against water (2 X), saline (2 X), and water (2 X) using Pierce Thermo Fisher 3k filters. The Amicon membrane was washed with water (3 X 2 mL) and with a last wash of 20% ethanol in water (20 µL). The combined solvents were then lyophilized to afford an amorphous white solid of Conjugated Sense AT (2.5 mg, 0.00057 mmol, 24% yield). Post-synthetic conjugation was realized through an amide coupling reaction. The Conjugated Sense AT was characterized by LCMS. Observed LCMS: 7436. Sense AT was annealed to Antisense B to yield Duplex AT using the same procedures as described for the annealing of Duplex A. Synthesis of the small molecule: 16-(butylsulfonamido)hexadecanoic acid (ATa):

To a stirred solution of n-butane sulfonamide (47.1 mg, 0.343 mmol, 1.5 equiv) in anhydrous DMF (5 mL) at 0
oC, sodium hydride (55% in mineral oil, 14.0 mg, 0.321 mmol, 1.4 equiv) was added. The resulting mixture was stirred at the same temperature for 30 min. A solution of methyl 16-bromohexadecanoate (80 mg, 0.229 mmol, 1.0 equiv) in anhydrous DMF (5 mL) was added to the reaction mixture and the resulting mixture was stirred at 25 °C for 3 h. The reaction mixture was cooled to 0
oC and carefully quenched by the addition of saturated aqueous NH
4Cl solution (10 mL). The aqueous layer was extracted with EtOAc (3 × 10 mL) and the combined organic layer was dried over anhydrous Na
2SO
4, filtered, and concentrated under reduced pressure. The obtained residue was purified by flash column chromatography (silica gel, 5 → 20% ethyl acetate in hexanes) to afford pure Methyl 16-
FH12501620.1
Attorney Docket: DCY-13025 (butylsulfonamido)hexadecanoate (ATb) (60.4 g, 0.148 mmol, 65% yield) as a white solid.
1H NMR (400 MHz, CDCl
3) ^ 4.22 (t, J = 6.1 Hz, 1H), 3.66 (s, 3H), 3.09 (td, J = 7.2, 6.2 Hz, 2H), 3.03 – 2.93 (m, 2H), 2.29 (t, J = 7.6 Hz, 2H), 1.81 – 1.70 (m, 2H), 1.63 – 1.38 (m, 6H), 1.36 – 1.19 (m, 22H), 0.95 (t, J = 7.4 Hz, 3H) ppm;
13C NMR (126 MHz, CDCl
3) ^ 174.4, 52.4, 51.5, 43.4, 34.1, 30.4, 29.63, 29.60, 29.56, 29.51, 29.4, 29.3, 29.2, 26.6, 25.7, 25.0, 21.6, 13.6 ppm; To a stirred solution of Methyl 16-(butylsulfonamido)hexadecanoate (ATb) (50 mg, 0.123 mmol, 1.0 equiv) in THF:MeOH (3:1 v/v, 4 mL) was added a solution of LiOH (29.5 mg, 1.233 mmol, 10 equiv) in H2O (1 mL) at 25
oC. The resulting mixture was stirred at the same temperature for 4 h. The pH of the reaction mixture was adjusted to 3-4 using 1N HCl solution. The aqueous layer was extracted with EtOAc (3 × 10 mL) and the combined organic layer was dried over anhydrous Na2SO4, filtered, and concentrated under reduced pressure. The obtained residue was purified by flash column chromatography (silica gel, 5 → 20% ethyl acetate in hexanes) to afford pure 116-(butylsulfonamido)hexadecanoic acid (ATa) (37.6 mg, 0.095 mmol, 78% yield) as a white solid.
1H NMR (400 MHz, MeOD) ^ 2.95 – 2.81 (m, 4H), 2.16 (t, J = 7.6 Hz, 2H), 1.73 – 1.58 (m, 2H), 1.55 – 1.28 (m, 4H), 1.26 – 1.10 (m, 24H), 0.83 (t, J = 7.4 Hz, 3H) ppm;
13C NMR (126 MHz, MeOD) ^ 176.7, 52.0, 43.1, 34.1, 30.2, 29.55, 29.52, 29.50, 29.46, 29.40, 29.2, 29.15, 29.10, 26.6, 25.5, 24.9, 21.5, 13.4 ppm; Synthesis of Duplex AU:
FH12501620.1
Attorney Docket: DCY-13025
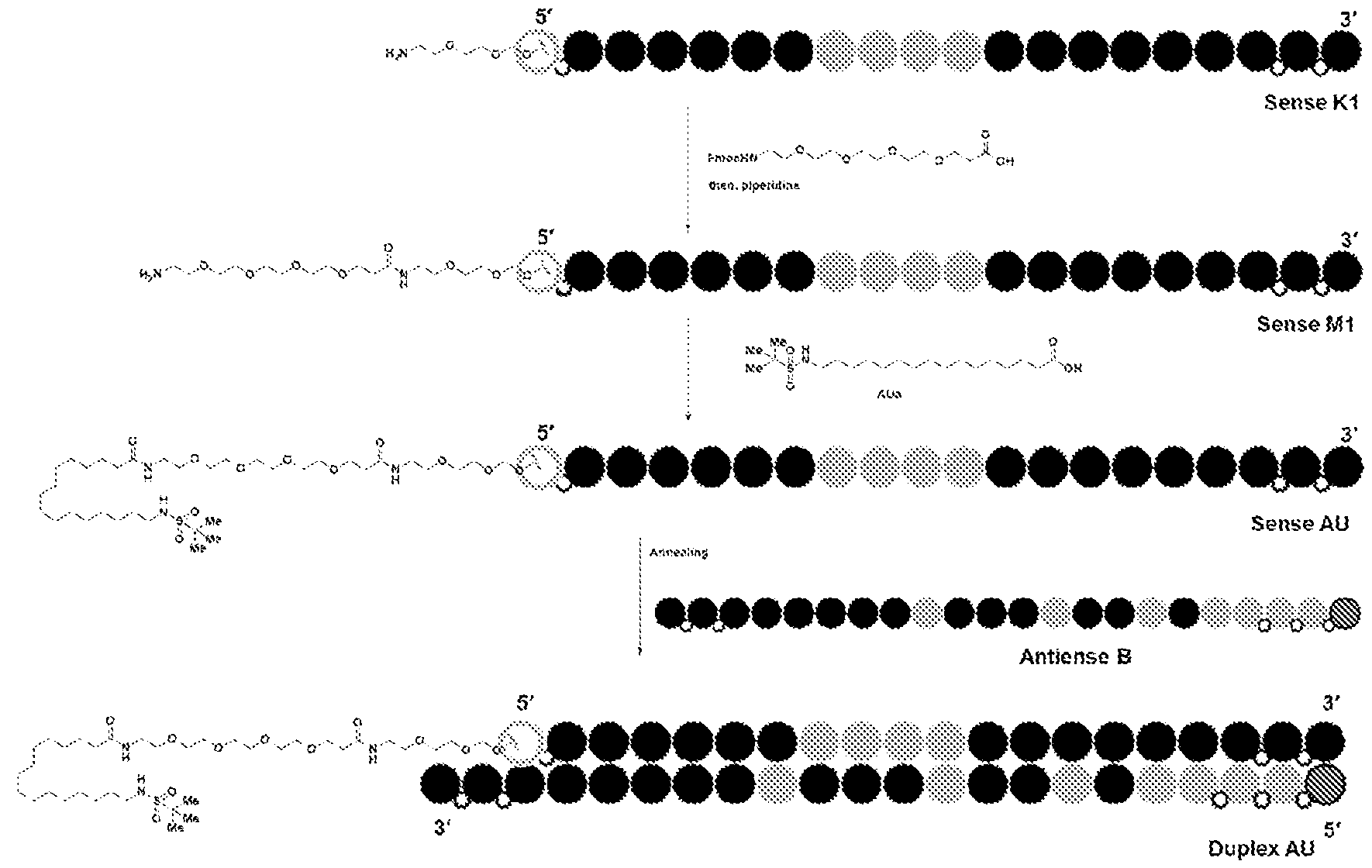
Conjugated Sense AU was synthesized through a two-step post-syntenic conjugation approach. Step 1- Attachment of Amino-PEG4 Handle: Sense M1 was synthesized with the method above. Step 2- Conjugation to Amino-PEG4 Handle In Eppendorf tube 1, a solution of 16-((1,1-dimethylethyl)sulfonamido)hexadecanoic acid (AUa) (5 mg, 0.0142 mmol, 10 equiv) was dissolved in DMF (500 µL) and treated with HATU (4.29 mg, 0.0113 mmol, 8 equiv) and DIPEA (2.5 µL, 0.0142 mmol, 10 equiv) at rt and mixed for 15 minutes. In Eppendorf tube 2, a solution of Conjugated Sense M1 (10.0 mg, 0.00142 mmol, 1 equiv) in 100 µL H2O was treated with DIPEA (2.5 µL, 0.0142 mmol, 10 equiv). The solution in Eppendorf tube 1 was then added to the Eppendorf tube 2 and mixed for 30 minutes using Thermomixer at 40°C. After the reaction was completed indicated by LC-MS analysis, the reaction mixture was diluted with 5 mL of water and purified by reverse phase XBridge C18 column using a 5-95% gradient of 100 mM TEAA in ACN and H
2O. The product fractions were concentrated under reduced pressure using GeneVac. The combined residual solvent was dialyzed against water (2 X), saline (2 X), and water (2 X) using Pierce Thermo Fisher 3k filters. The Amicon membrane was washed with water (3 X 2 mL) and with a last wash of 20% ethanol in water (20 µL). The combined solvents were then lyophilized to afford an amorphous white solid of Conjugated Sense AU (3.2 mg, 0.00043 mmol, 30% yield). Post-synthetic
FH12501620.1
Attorney Docket: DCY-13025 conjugation was realized through an amide coupling reaction. The
Conjugated Sense AU was characterized by LCMS. Observed LCMS: 7436. Sense AU was annealed to Antisense B to yield Duplex AU using the same procedures as described for the annealing of Duplex A. Synthesis of the small molecule: 16-((1,1-dimethylethyl)sulfonamido)hexadecanoic acid
To a stirred solution of tert-butyl sulfonamide (106.0 mg, 0.773 mmol, 1.5 equiv) in anhydrous DMF (3 mL) at 0
oC, sodium hydride (55% in mineral oil, 31.5 mg, 0.721 mmol, 1.4 equiv) was added. The resulting mixture was stirred at the same temperature for 30 min. A solution of methyl 16-bromohexadecanoate (180 mg, 0.515 mmol, 1.0 equiv) in anhydrous DMF (2 mL) was added to the reaction mixture and the resulting mixture was stirred at 25 °C for 3 h. The reaction mixture was cooled to 0
oC and carefully quenched by the addition of saturated aqueous NH4Cl solution (10 mL). The aqueous layer was extracted with EtOAc (3 × 10 mL) and the combined organic layer was dried over anhydrous Na2SO4, filtered, and concentrated under reduced pressure. The obtained residue was purified by flash column chromatography (silica gel, 5
20% ethyl acetate in hexanes) to afford pure Methyl 16- ((1,1-dimethylethyl)sulfonamido)hexadecanoate (AUb) (125.3 mg, 0.309 mmol, 60% yield) as a white solid.
1H NMR (400 MHz, CDCl
3) ^ 3.92 (t, J = 6.0 Hz, 1H), 3.66 (s, 3H), 3.16 (td, J = 7.2, 6.1 Hz, 2H), 2.29 (t, J = 7.5 Hz, 2H), 1.65 – 1.50 (m, 4H), 1.38 (s, 9H), 1.32 – 1.21 (m, 22H) ppm;
13C NMR (126 MHz, CDCl
3) ^ 174.6, 60.0, 51.6, 45.1, 34.3, 31.4, 29.8, 29.77, 29.75, 29.69, 29.64, 29.5, 29.4, 29.3, 26.8, 25.2, 24.6 ppm; To a stirred solution of Methyl 16-((1,1-dimethylethyl)sulfonamido)hexadecanoate (AUb) (75 mg, 0.184 mmol, 1.0 equiv) in THF:MeOH (3:1 v/v, 4 mL) was added a solution of LiOH (44.2 mg, 1.848 mmol, 10 equiv) in H2O (1 mL) at 25
oC. The resulting mixture was stirred at the same temperature for 4 h. The pH of the reaction mixture was adjusted to 3-4 using 1N HCl solution. The aqueous layer was extracted with EtOAc (3 × 10 mL) and the combined
FH12501620.1
Attorney Docket: DCY-13025 organic layer was dried over anhydrous Na2SO4, filtered, and concentrated under reduced pressure. The obtained residue was purified by flash column chromatography (silica gel, 5 → 20% ethyl acetate in hexanes) to afford pure 16-((1,1- dimethylethyl)sulfonamido)hexadecanoic acid (AUa) (49.0 mg, 0.125 mmol, 68% yield) as a white solid.
1H NMR (500 MHz, CDCl3) ^ 4.21 (t, J = 6.0 Hz, 1H), 3.16 (td, J = 7.3, 6.1 Hz, 2H), 2.34 (t, J = 7.5 Hz, 2H), 1.62 (p, J = 7.5 Hz, 2H), 1.55 (q, J = 7.5 Hz, 2H), 1.39 (s, 9H), 1.34 – 1.22 (m, 22H) ppm;
13C NMR (101 MHz, CDCl
3) ^ 179.6, 60.0, 45.1, 34.2, 31.4, 29.8, 29.70, 29.68, 29.66, 29.63, 29.5, 29.4, 29.2, 26.8, 24.9, 24.6 ppm; Synthesis of Duplex AV:
Conjugated Sense AV was synthesized through a two-step post-syntenic conjugation approach. Step 1- Attachment of Amino-PEG4 Handle:
Sense M1 was synthesized with the method above. Step 2- Conjugation to Amino-PEG4 Handle In Eppendorf tube 1, a solution of 16-(cyclobutanesulfonamido)hexadecanoic acid (AVa) (5 mg, 0.0142 mmol, 10 equiv) was dissolved in DMF (500 µL) and treated with HATU (4.29 mg, 0.0113 mmol, 8 equiv) and DIPEA (2.5 µL, 0.0142 mmol, 10 equiv) at rt and mixed for 15 minutes. In Eppendorf tube 2, a solution of Conjugated Sense M1 (10.0 mg, 0.00142 mmol, 1 equiv) in 100 µL H2O was treated with DIPEA (2.5 µL, 0.0142 mmol, 10 equiv). The solution
FH12501620.1
Attorney Docket: DCY-13025 in Eppendorf tube 1 was then added to the Eppendorf tube 2 and mixed for 30 minutes using Thermomixer at 40°C. After the reaction was completed indicated by LC-MS analysis, the reaction mixture was diluted with 5 mL of water and purified by reverse phase XBridge C18 column using a 5-95% gradient of 100 mM TEAA in ACN and H
2O. The product fractions were concentrated under reduced pressure using GeneVac. The combined residual solvent was dialyzed against water (2 X), saline (2 X), and water (2 X) using Pierce Thermo Fisher 3k filters. The Amicon membrane was washed with water (3 X 2 mL) and with a last wash of 20% ethanol in water (20 µL). The combined solvents were then lyophilized to afford an amorphous white solid of Conjugated Sense AV (1.3 mg, 0.00030 mmol, 21% yield). Post-synthetic conjugation was realized through an amide coupling reaction. The Conjugated Sense AV was characterized by LCMS. Observed LCMS: 7434. Sense AV was annealed to Antisense B to yield Duplex AV using the same procedures as described for the annealing of Duplex A. Synthesis of the small molecule: 16-(cyclobutanesulfonamido)hexadecanoic acid (AVa):
To a stirred solution of cyclobutane sulfonamide (52.2 mg, 0.386 mmol, 1.5 equiv) in anhydrous DMF (3 mL) at 0
oC, sodium hydride (55% in mineral oil, 15.7 mg, 0.359 mmol, 1.4 equiv) was added. The resulting mixture was stirred at the same temperature for 30 min. A solution of methyl 16-bromohexadecanoate (xx) (90 mg, 0.257 mmol, 1.0 equiv) in anhydrous DMF (2 mL) was added to the reaction mixture and the resulting mixture was stirred at 25 °C for 3 h. The reaction mixture was cooled to 0
oC and carefully quenched by the addition of saturated aqueous NH
4Cl solution (10 mL). The aqueous layer was extracted with EtOAc (3 × 10 mL) and the combined organic layer was dried over anhydrous Na
2SO
4, filtered, and concentrated under reduced pressure. The obtained residue was purified by flash column chromatography (silica gel, 5
20% ethyl acetate in hexanes) to afford pure methyl 16-(cyclobutanesulfonamido)hexadecanoate (AVb) (72.6 mg, 0.179 mmol, 70% yield) as a white solid.
1H NMR (500 MHz, CDCl
3) ^ 4.03 (t, J = 7.3 Hz, 1H), 3.79 (p, J = 8.4 Hz, 1H),
FH12501620.1
Attorney Docket: DCY-13025 3.66 (s, 3H), 3.09 (q, J = 6.9 Hz, 2H), 2.53 – 2.44 (m, 2H), 2.34 – 2.24 (m, 4H), 2.08 – 1.96 (m, 2H), 1.70 – 1.56 (m, 2H), 1.55 – 1.48 (m, 2H), 1.31 – 1.21 (m, 22H) ppm;
13C NMR (101 MHz, CDCl
3) ^ 174.6, 54.2, 51.6, 47.6, 34.3, 29.8, 29.79, 29.77, 29.65, 29.5, 29.4, 29.35, 29.34, 29.1, 26.9, 25.2, 24.6, 17.3 ppm; To a stirred solution of methyl 16-(cyclobutanesulfonamido)hexadecanoate (AVb) (55 mg, 0.136 mmol, 1.0 equiv) in THF:MeOH (3:1 v/v, 4 mL) was added a solution of LiOH (32.6 mg, 1.362 mmol, 10 equiv) in H2O (1 mL) at 25
oC. The resulting mixture was stirred at the same temperature for overnight. The pH of the reaction mixture was adjusted to 3-4 using 1N HCl solution. The aqueous layer was extracted with EtOAc (3 × 10 mL) and the combined organic layer was dried over anhydrous Na2SO4, filtered, and concentrated under reduced pressure. The obtained residue was purified by flash column chromatography (silica gel, 20 → 80% ethyl acetate in hexanes) to afford pure 16-(cyclobutanesulfonamido)hexadecanoic acid (AVa) (28.6 g, 0.073 mmol, 54% yield) as a white solid.
1H NMR (500 MHz, MeOD) ^ 3.77 (p, J = 8.2 Hz, 1H), 3.00 (t, J = 7.2 Hz, 2H), 2.49 – 2.34 (m, 2H), 2.30 – 2.13 (m, 4H), 2.06 – 1.84 (m, 2H), 1.57 (p, J = 7.4 Hz, 2H), 1.49 (q, J = 7.1 Hz, 2H), 1.34 – 1.19 (m, 22H) ppm;
13C NMR (126 MHz, MeOD) ^ 176.8, 54.3, 43.1, 34.2, 30.5, 29.54, 29.50, 29.47, 29.45, 29.41, 29.3, 29.2, 29.1, 29.0, 26.5, 24.9, 23.8, 16.6 ppm; Synthesis of Duplex AW:
FH12501620.1
Attorney Docket: DCY-13025 Conjugated Sense AG was synthesized through a two-step post-syntenic conjugation approach. Step 1- Attachment of Amino-PEG4 Handle: Sense M1 was synthesized with the method above. Step 2- Conjugation to Amino-PEG4 Handle In Eppendorf tube 1, a solution of 16-(methylsulfonamido)hexadecanoic acid (AWa) (5 mg, 0.0142 mmol, 10 equiv) was dissolved in DMF (500 µL) and treated with HATU (4.29 mg, 0.0113 mmol, 8 equiv) and DIPEA (2.5 µL, 0.0142 mmol, 10 equiv) at rt and mixed for 15 minutes. In Eppendorf tube 2, a solution of Conjugated Sense M1 (10.0 mg, 0.00142 mmol, 1 equiv) in 100 µL H2O was treated with DIPEA (2.5 µL, 0.0142 mmol, 10 equiv). The solution in Eppendorf tube 1 was then added to the Eppendorf tube 2 and mixed for 30 minutes using Thermomixer at 40°C. After the reaction was completed indicated by LC-MS analysis, the reaction mixture was diluted with 5 mL of water and purified by reverse phase XBridge C18 column using a 5-95% gradient of 100 mM TEAA in ACN and H
2O. The product fractions were concentrated under reduced pressure using GeneVac. The combined residual solvent was dialyzed against water (2 X), saline (2 X), and water (2 X) using Pierce Thermo Fisher 3k filters. The Amicon membrane was washed with water (3 X 2 mL) and with a last wash of 20% ethanol in water (20 µL). The combined solvents were then lyophilized to afford an amorphous white solid of Conjugated Sense AW (2.5 mg, 0.00068 mmol, 48% yield). Post-synthetic conjugation was realized through an amide coupling reaction. The Conjugated Sense AW was characterized by LCMS. Observed LCMS: 7448. Sense AW was annealed to Antisense B to yield Duplex AW using the same procedures as described for the annealing of Duplex A. Synthesis of the small molecule: 16-(cyclopentanesulfonamido)hexadecanoic acid
To a stirred solution of cyclopentane sulfonamide (76.8 mg, 0.515 mmol, 1.5 equiv) in anhydrous DMF (3 mL) at 0
oC, sodium hydride (55% in mineral oil, 21.0 mg, 0.480 mmol,
FH12501620.1
Attorney Docket: DCY-13025 1.4 equiv) was added. The resulting mixture was stirred at the same temperature for 30 min. A solution of methyl 16-bromohexadecanoate (120 mg, 0.343 mmol, 1.0 equiv) in anhydrous DMF (2 mL) was added to the reaction mixture and the resulting mixture was stirred at 25 °C for 6 h. The reaction mixture was cooled to 0
oC and carefully quenched by the addition of saturated aqueous NH4Cl solution (10 mL). The aqueous layer was extracted with EtOAc (3 × 10 mL) and the combined organic layer was dried over anhydrous Na2SO4, filtered, and concentrated under reduced pressure. The obtained residue was purified by flash column chromatography (silica gel, 5
20% ethyl acetate in hexanes) to afford pure methyl 16- (cyclopentanesulfonamido)hexadecanoate (AWb) (74.3 mg, 0.178 mmol, 52% yield) as a white solid.
1H NMR (400 MHz, CDCl3) ^ 4.18 (t, J = 6.1 Hz, 1H), 3.66 (s, 3H), 3.46 (p, J = 8.0 Hz, 1H), 3.11 (td, J = 7.1, 6.0 Hz, 2H), 2.29 (t, J = 7.6 Hz, 2H), 2.00 (tdd, J = 7.4, 5.2, 1.8 Hz, 4H), 1.87 – 1.72 (m, 2H), 1.67 – 1.50 (m, 6H), 1.34 – 1.21 (m, 22H) ppm;
13C NMR (101 MHz, CDCl3) ^ 174.6, 61.8, 51.6, 43.7, 34.3, 30.8, 29.8, 29.75, 29.72, 29.67, 29.61, 29.59, 29.4, 29.35, 29.33, 28.3, 26.9, 26.1, 25.1 ppm; To a stirred solution of methyl 16-(cyclopentanesulfonamido)hexadecanoate (AWb) (70 mg, 0.167 mmol, 1.0 equiv) in THF:MeOH (3:1 v/v, 4 mL) was added a solution of LiOH (40.1 mg, 1.676 mmol, 10 equiv) in H2O (1 mL) at 25
oC. The resulting mixture was stirred at the same temperatrure for 4 h. The pH of the reaction mixture was adjusted to 3-4 using 1N HCl solution. The aqueous layer was extracted with EtOAc (3 × 10 mL) and the combined organic layer was dried over anhydrous Na2SO4, filtered, and concentrated under reduced pressure. The obtained residue was purified by flash column chromatography (silica gel, 30 → 80% ethyl acetate in hexanes) to afford pure 16-(cyclopentanesulfonamido)hexadecanoic acid (AWa) (45.2 mg, 0.113 mmol, 68% yield) as a white solid.
1H NMR (500 MHz, MeOD) ^ 3.49 (p, J = 8.2 Hz, 1H), 3.04 (t, J = 7.2 Hz, 2H), 2.26 (t, J = 7.4 Hz, 2H), 2.02 – 1.92 (m, 4H), 1.86 – 1.72 (m, 2H), 1.69 – 1.49 (m, 6H), 1.36 – 1.23 (m, 22H) ppm;
13C NMR (101 MHz, MeOD) ^ 177.7, 62.1, 44.1, 35.0, 31.4, 30.54, 30.50, 30.46, 30.40, 30.2, 30.1, 30.0, 28.8, 27.6, 26.7, 25.9 ppm; Synthesis of Duplex AX:
FH12501620.1
Attorney Docket: DCY-13025

Conjugated Sense AX was synthesized through a two-step post-syntenic conjugation approach. Step 1- Attachment of Amino-PEG4 Handle: Sense M1 was synthesized with the method above. Step 2- Conjugation to Amino-PEG4 Handle In Eppendorf tube 1, a solution of 16-(cyclohexanesulfonamido)hexadecanoic acid (AXa) (5 mg, 0.0142 mmol, 10 equiv) was dissolved in DMF (500 µL) and treated with HATU (4.29 mg, 0.0113 mmol, 8 equiv) and DIPEA (2.5 µL, 0.0142 mmol, 10 equiv) at rt and mixed for 15 minutes. In Eppendorf tube 2, a solution of Conjugated Sense M1 (10.0 mg, 0.00142 mmol, 1 equiv) in 100 µL H2O was treated with DIPEA (2.5 µL, 0.0142 mmol, 10 equiv). The solution in Eppendorf tube 1 was then added to the Eppendorf tube 2 and mixed for 30 minutes using Thermomixer at 40°C. After the reaction was completed indicated by LC-MS analysis, the reaction mixture was diluted with 5 mL of water and purified by reverse phase XBridge C18 column using a 5-95% gradient of 100 mM TEAA in ACN and H
2O. The product fractions were concentrated under reduced pressure using GeneVac. The combined residual solvent was dialyzed against water (2 X), saline (2 X), and water (2 X) using Pierce Thermo Fisher 3k filters. The Amicon membrane was washed with water (3 X 2 mL) and with a last wash of 20% ethanol in water (20 µL). The combined solvents were then lyophilized to afford an amorphous white solid of Conjugated Sense AX (4.2 mg, 0.00057 mmol, 40% yield). Post-synthetic
FH12501620.1
Attorney Docket: DCY-13025 conjugation was realized through an amide coupling reaction. The
Conjugated Sense AX was characterized by LCMS. Observed LCMS: 7462. Sense AX was annealed to Antisense B to yield Duplex AX using the same procedures as described for the annealing of Duplex A. Synthesis of the small molecule: 16-(cyclohexanesulfonamido)hexadecanoic acid (AXa):
To a stirred solution of cyclohexane sulfonamide (210 mg, 1.288 mmol, 1.5 equiv) in anhydrous DMF (4 mL) at 0
oC, sodium hydride (55% in mineral oil, 52.5 mg, 1.203 mmol, 1.4 equiv) was added. The resulting mixture was stirred at the same temperature for 30 min. A solution of methyl 16-bromohexadecanoate (300 mg, 0.859 mmol, 1.0 equiv) in anhydrous DMF (3 mL) was added to the reaction mixture and the resulting mixture was stirred at 25 °C for 3 h. The reaction mixture was cooled to 0
oC and carefully quenched by the addition of saturated aqueous NH
4Cl solution (10 mL). The aqueous layer was extracted with EtOAc (3 × 10 mL) and the combined organic layer was dried over anhydrous Na
2SO
4, filtered, and concentrated under reduced pressure. The obtained residue was purified by flash column chromatography (silica gel, 5 → 15% ethyl acetate in hexanes) to afford pure Methyl 16- (cyclohexanesulfonamido)hexadecanoate (AXb) (274.4 mg, 0.636 mmol, 74% yield) as a white solid.
1H NMR (400 MHz, CDCl3) ^ 4.15 (t, J = 6.1 Hz, 1H), 3.65 (s, 3H), 3.09 (td, J = 7.2, 6.2 Hz, 2H), 2.94 – 2.74 (m, 1H), 2.29 (t, J = 7.6 Hz, 2H), 2.15 (ddt, J = 11.2, 3.6, 1.7 Hz, 2H), 1.88 (dt, J = 12.1, 2.9 Hz, 2H), 1.76 – 1.67 (m, 1H), 1.64 – 1.42 (m, 7H), 1.38 – 1.22 (m, 24H) ppm;
13C NMR (101 MHz, CDCl3) ^ 174.6, 61.4, 51.6, 43.8, 34.3, 30.9, 29.8, 29.76, 29.73, 29.69, 29.62, 29.4, 29.36, 29.33, 26.9, 26.7, 25.4, 25.3, 25.1 ppm; To a stirred solution of Methyl 16-(cyclohexanesulfonamido)hexadecanoate (AXb) (120 mg, 0.278 mmol, 9.0 equiv) in THF:MeOH (3:1 v/v, 4 mL) was added a solution of LiOH (66.6 mg, 2.779 mmol, 10 equiv) in H
2O (1 mL) at 25
oC. The resulting mixture was stirred at the same temperature for 4h. The pH of the reaction mixture was adjusted to 3-4 using 1N HCl solution. The aqueous layer was extracted with EtOAc (3 × 50 mL) and the combined organic
FH12501620.1
Attorney Docket: DCY-13025 layer was dried over anhydrous Na2SO4, filtered, and concentrated under reduced pressure. The obtained residue was purified by flash column chromatography (silica gel, 30 → 80% ethyl acetate in hexanes) to afford pure 16-(cyclohexanesulfonamido)hexadecanoic acid (AXa) (81.3 mg, 0.195 mmol, 70% yield) as a white solid.
1H NMR (400 MHz, CDCl
3) ^ 4.38 (t, J = 6.1 Hz, 1H), 3.09 (q, J = 6.8 Hz, 2H), 2.86 (tt, J = 12.1, 3.4 Hz, 1H), 2.34 (t, J = 7.5 Hz, 2H), 2.23 – 2.07 (m, 2H), 1.89 (ddd, J = 10.4, 5.2, 2.6 Hz, 2H), 1.76 – 1.66 (m, 1H), 1.65 – 1.43 (m, 7H), 1.36 – 1.17 (m, 24H) ppm;
13C NMR (101 MHz, CDCl
3) ^ 179.5, 61.4, 43.8, 34.1, 30.9, 29.7, 29.67, 29.63, 29.5, 29.38, 29.36, 29.2, 26.9, 26.7, 25.4, 25.3, 24.9 ppm; Synthesis of Duplex AY:
Conjugated Sense AY was synthesized through a two-step post-syntenic conjugation approach. Step 1- Attachment of Amino-PEG4 Handle:
Sense M1 was synthesized with the method above. Step 2- Conjugation to Amino-PEG4 Handle In Eppendorf tube 1, a solution of 16-(bicyclo[1.1.1]pentane-1-sulfonamido)hexadecanoic acid (AYa) (5 mg, 0.0142 mmol, 10 equiv) was dissolved in DMF (500 µL) and treated with HATU (4.29 mg, 0.0113 mmol, 8 equiv) and DIPEA (2.5 µL, 0.0142 mmol, 10 equiv) at rt and mixed for 15 minutes. In Eppendorf tube 2, a solution of Conjugated Sense M1 (10.0 mg, 0.00142 mmol, 1 equiv) in 100 µL H2O was treated with DIPEA (2.5 µL, 0.0142 mmol, 10
FH12501620.1
Attorney Docket: DCY-13025 equiv). The solution in Eppendorf tube 1 was then added to the Eppendorf tube 2 and mixed for 30 minutes using Thermomixer at 40°C. After the reaction was completed indicated by LC- MS analysis, the reaction mixture was diluted with 5 mL of water and purified by reverse phase XBridge C18 column using a 5-95% gradient of 100 mM TEAA in ACN and H
2O. The product fractions were concentrated under reduced pressure using GeneVac. The combined residual solvent was dialyzed against water (2 X), saline (2 X), and water (2 X) using Pierce Thermo Fisher 3k filters. The Amicon membrane was washed with water (3 X 2 mL) and with a last wash of 20% ethanol in water (20 µL). The combined solvents were then lyophilized to afford an amorphous white solid of Conjugated Sense AY (3.5 mg, 0.00053 mmol, 37% yield). Post- synthetic conjugation was realized through an amide coupling reaction. The Conjugated Sense AY was characterized by LCMS. Observed LCMS: 7446. Sense AY was annealed to Antisense B to yield Duplex AY using the same procedures as described for the annealing of Duplex A. Synthesis of the small molecule: 16-(bicyclo[1.1.1]pentane-1-sulfonamido)hexadecanoic
To a stirred solution of bicyclo[1.1.1]pentane-1-sulfonamide (50.5 mg, 0.343 mmol, 1.5 equiv) in anhydrous DMF (3 mL) at 0
oC, sodium hydride (55% in mineral oil, 13.9 mg, 0.319 mmol, 1.4 equiv) was added. The resulting mixture was stirred at the same temperature for 30 min. A solution of methyl 16-bromohexadecanoate (80 mg, 0.228 mmol, 1.0 equiv) in anhydrous DMF (1.5 mL) was added to the reaction mixture and the resulting mixture was stirred at 25 °C for 3 h. The reaction mixture was cooled to 0
oC and carefully quenched by the addition of saturated aqueous NH4Cl solution (10 mL). The aqueous layer was extracted with EtOAc (3 × 10 mL) and the combined organic layer was dried over anhydrous Na2SO4, filtered, and concentrated under reduced pressure. The obtained residue was purified by flash column chromatography (silica gel, 5 15% ethyl acetate in hexanes) to afford pure methyl 16-(bicyclo[1.1.1]pentane-1-sulfonamido)hexadecanoate (AYb) (64.4 mg, 0.155 mmol, 68%
FH12501620.1
Attorney Docket: DCY-13025 yield) as a white solid.
1H NMR (400 MHz, CDCl3) ^ 4.15 (t, J = 6.1 Hz, 1H), 3.66 (s, 3H), 3.11 (td, J = 7.1, 6.1 Hz, 2H), 2.70 (s, 1H), 2.29 (t, J = 7.5 Hz, 2H), 2.20 (s, 6H), 1.65 – 1.47 (m, 4H), 1.33 – 1.21 (m, 22H) ppm;
13C NMR (101 MHz, CDCl
3) ^ 174.6, 54.2, 51.6, 51.4, 43.9, 34.3, 31.0, 29.8, 29.77, 29.74, 29.69, 29.63, 29.5, 29.4, 29.3, 26.8, 26.7, 26.6, 25.2 ppm; To a stirred solution of methyl 16-(bicyclo[1.1.1]pentane-1-sulfonamido)hexadecanoate (AYb) (50 mg, 0.120 mmol, 1.0 equiv) in THF:MeOH (3:1 v/v, 3 mL) was added a solution of LiOH (28.8 mg, 1.203 mmol, 10 equiv) in H2O (0.8 mL) at 25
oC. The resulting mixture was stirred at the same temperature for 4h. The pH of the reaction mixture was adjusted to 3- 4 using 1N HCl solution. The aqueous layer was extracted with EtOAc (3 × 10 mL) and the combined organic layer was dried over anhydrous Na2SO4, filtered, and concentrated under reduced pressure. The obtained residue was purified by flash column chromatography (silica gel, 30 → 80% ethyl acetate in hexanes) to afford pure 16-(bicyclo[1.1.1]pentane-1- sulfonamido)hexadecanoic acid (AYa) (40.9 mg, 0.102 mmol, 85% yield) as a white solid.
1H NMR (500 MHz, MeOD) ^ 3.05 (t, J = 7.1 Hz, 2H), 2.68 (s, 1H), 2.27 (t, J = 7.4 Hz, 2H), 2.19 (s, 6H), 1.60 (p, J = 7.1 Hz, 2H), 1.55 – 1.46 (m, 2H), 1.40 – 1.26 (m, 22H) ppm;
13C NMR (101 MHz, MeOD) ^ 178.0, 55.2, 52.3, 44.6, 35.2, 32.1, 30.9, 30.86, 30.81, 30.7, 30.6, 30.5, 30.4, 27.8, 27.7, 27.6, 26.3 ppm; Synthesis of Duplex AZ:
Conjugated Sense AZ was synthesized through a two-step post-syntenic conjugation approach. Step 1- Attachment of Amino-PEG4 Handle:
FH12501620.1
Attorney Docket: DCY-13025 In Eppendorf tube 1, a solution of Fmoc-N-amido-PEG
4-TFP ester (18.3 mg, 0.0289 mmol, 10 equiv) was dissolved in DMF (1 mL) and treated with DIPEA (50 µL, excess). In Eppendorf tube 2, a solution of oligo Sense AZ1 (20 mg, 0.00289 mmol, 1 equiv) in 200 µL H
2O was treated with DIPEA (50 µL, excess). The solution in Eppendorf tube 1 was added to Eppendorf tube 2 and mixed for 1 hour using Thermomixer at 40°C. After the reaction was completed indicated by LC-MS analysis, the crude was then dialyzed against water (2 X) using Pierce Thermo Fisher 3k filters to remove excess Fmoc-N-amido-PEG4-TFP ester. The crude was then removed from filters and dissolved in DMF: H2O (5:1 v/v, 1.5 mL). Piperidine (100 µL) was added to the reaction mixture and mixed for 20 minutes using Thermomixer at rt. The reaction was then diluted with 5 mL of water and purified by reverse phase XBridge C18 column using a 5-95% gradient of 100 mM TEAA in ACN and H2O. The product fractions were concentrated under reduced pressure using Genevac. The combined residual solvent was dialyzed against water (2 X), saline (2 X), and water (2 X) using Pierce Thermo Fisher 3k filters. The filter membrane was washed with water (3 X 2 mL) and with a last wash of 20% ethanol in water (20 µL). The combined solvents were then lyophilized to afford an amorphous white solid of Conjugated Sense AZ2 (13.0 mg, 0.00175 mmol, 61% yield). Post-synthetic conjugation was realized through an amide coupling reaction. Observed LCMS: 7413. Step 2- Conjugation to Amino-PEG4 Handle In Eppendorf tube 1, a solution of 16-(cyclopropanesulfonamido)hexadecanoic acid (AFa, 6.58 mg, 0.0175 mmol, 10 equiv) was dissolved in DMF (500 µL) and treated with HATU (5.3 mg, 0.014 mmol, 8 equiv) and DIPEA (100 µL, excess) at rt and mixed for 15 minutes. In Eppendorf tube 2, a solution of Conjugated Sense AZ2 (13.0 mg, 0.00175 mmol, 1 equiv) in 100 µL H2O was treated with DIPEA (50 µL, excess). The solution in Eppendorf tube 1 was then added to Eppendorf tube 2 and mixed for 30 minutes using Thermomixer at 40°C. After the reaction was completed indicated by LC-MS analysis, the reaction mixture was diluted with 5 mL of water and purified by reverse phase XBridge C18 column using a 5-95% gradient of 100 mM TEAA in ACN and H
2O. The product fractions were concentrated under reduced pressure using GeneVac. The combined residual solvent was dialyzed against water (2 X), saline (2 X), and water (2 X) using Pierce Thermo Fisher 3k filters. The Amicon membrane was washed with water (3 X 2 mL) and with a last wash of 20% ethanol in water (20 µL). The combined solvents were then lyophilized to afford an amorphous white solid of Conjugated Sense AZ (5.0 mg, 0.000615 mmol, 35% yield). Post-synthetic conjugation was realized through an amide coupling reaction. The Conjugated Sense AZ was characterized by LCMS. LCMS: 8128.
FH12501620.1
Attorney Docket: DCY-13025 Sense AZ was annealed to Antisense B to yield Duplex AZ using the same procedures as described for the annealing of Duplex A. Example 3: Tissue Selectivity of RNAi Oligonucleotide with Terminal C16 Conjugate with Carboxylate Double-stranded RNAi oligonucleotides with a C16-carboxylate were designed with a conjugate linked to the 5’-terminal nucleotide of the sense strand or to a nucleotide of the tetraloop of the sense strand (as depicted in FIGs.1A and 1B). The oligonucleotides were compared to investigate whether inclusion of a C16-carboxylate conjugate alters target gene knockdown in different tissue types. Briefly, mice were intravenously (i.v.) administered oligonucleotides comprising a modified sense strand of SEQ ID NO: 1, 2, or 3, and a modified antisense strand of SEQ ID NO: 9 linked to the conjugates indicated in Table 1 to generate the corresponding duplex , at a dose of 15 mg/kg. Seven days after administration, tissue was collected, and total RNA was subjected to qRT-PCT analysis to determine Aldh2 mRNA levels. mRNA levels were measured for mouse mRNA. The values were normalized for transfection efficiency using the GAPDH housekeeping gene. Table 1. Oligonucleotide conjugates

C16 in Duplex C-D refers to the number of carbon atoms in the chain from triazole to the carboxylic acid. Although the carbon atom in the -COOH group is shown above, it is counted as parted of alkyl chain. The results in FIGs.2A-2B demonstrate that oligonucleotides comprising a C16- COOH on either a nucleotide of the tetraloop or P1 (position 1 of the sense strand based on 5’ to 3’ numbering) inhibit target mRNA in adipose tissue while decreasing overall target gene knockdown in the liver (see C16-COOH on Stem loop and C16-COOH on P1). However, inclusion of a conjugate without carboxylate (C22 and conjugate with C16 on P1 and C22 on loop) eliminates the adipose tissue selectivity and maintains target gene knockdown in the liver. Overall, the inclusion of a C16-COOH oligonucleotide increases the ability of the oligonucleotide to inhibit target mRNA with adipose tissue selectivity.
FH12501620.1
Attorney Docket: DCY-13025 Example 4: Adipose Tissue Selectivity of COOH Conjugated Oligonucleotides To validate if a COOH conjugate to an oligonucleotide was selective for adipose tissue, oligonucleotides comprising a C22 or a C16-COOH on the stem loop of the sense strand (as depicted in FIG.3) were compared. Briefly, mice were intravenously (i.v.) administered an oligonucleotide linked to a conjugate indicated in Table 2 to generate the corresponding oligonucleotide, at a dose of 15 mg/kg. Seven days after administration, tissue was collected, and total RNA was subjected to qRT-PCT analysis to determine Aldh2 mRNA levels as described in Example 3. Table 2. Oligonucleotide conjugates

C16 in Duplex B refers to the number of carbon atoms in the chain from triazole to the carboxylic acid. Although the carbon atom in the -COOH group is shown above, it is counted as parted of alkyl chain. The results in FIGs.4A-4D show target gene knockdown for C22 conjugation across liver, gWAT (gonadal white adipose tissue), quadricep, and scWAT (subcutaneous white adipose tissue). Similar knockdown is observed in adipose tissue (gWAT and scWAT) for C16- COOH, however, knockdown in the liver and quadricep is reduced demonstrating a tissue specific knockdown effect in adipose tissue. Overall, carboxylate conjugation (COOH) can target adipose tissue while reducing target gene knockdown in the liver. Example 5: Alkyl Chains and Terminal Carboxylate Produce Liver Sparing Effect on Target Gene Knockdown with RNAi Oligonucleotides To better understand the liver sparing effect of oligonucleotides comprising a carboxylate conjugation, ligands and their corresponding linkers were modified with different PEG4 extensions, triazole rings, alkyl chains, and terminal carboxylate (as depicted in FIG. 5). Briefly, mice were intravenously (i.v.) administered oligonucleotides comprising a modified sense strand of SEQ ID NO: 2 or 4 and a modified antisense strand of SEQ ID NO: 9 linked to the conjugations indicated in Table 3 to generate the corresponding duplex, at a dose of 15 mg/kg. Seven days after administration, tissue was collected, and total RNA was subjected to qRT-PCT analysis to determine Aldh2 mRNA levels as described in Example 3.
FH12501620.1
Attorney Docket: DCY-13025 Table 3. Oligonucleotide conjugates
C16 in Duplex C, E, F, G, and I refers to the number of carbon atoms in the chain from triazole to the carboxylic acid. Although the carbon atom in the -COOH group is shown above, it is counted as parted of the alkyl chain. The results in FIGs.6A-6F show the conjugate ademG-PEG
4-triazole-C16-COOH has liver sparing effects while maintaining target gene knockdown in scWAT and gWAT tissue. The results demonstrate the importance of a carboxylate (ademG-PEG
4-triazole-C16- COOH ) as compared to a terminal alkyl with a PEG4 and triazole (PEG4-triz-C16) which shows no liver sparing effect. The results further demonstrate the importance of an alkyl chain as activity is lost upon exclusion of an alkyl chain (e.g., PEG4-tri-PEG4-COOH and PEG4-triz-C3-COOH ). Additionally, inclusion of a PEG4 is not a necessary requirement for liver sparing effects (compare PEG4-triz-C16-COOH to C16). Together this data demonstrates the inclusion of an alkyl chain and terminal carboxylate generate a liver sparing effect with extrahepatic knockdown of a target gene. Example 6: Optimized Structure for AdemG Amidite Synthesis To identifying structures for incorporation of a carboxylate into a 2’Adem structure , conjugates including triazole, various PEG length modifications, and alkyl length (as depicted in FIG.7) were evaluated. Briefly, mice were subcutaneously (s.c.) administered oligonucleotides comprising a modified sense strand of SEQ ID NO: 5 or 6 and a modified antisense strand of SEQ ID NO: 10 linked to the conjugates indicated in Table 4 to generate the corresponding duplex, at a dose of 10 mg/kg. Seven days after administration, tissue was collected, and total RNA was subjected to qRT-PCT analysis to determine Aldh2 mRNA levels as described in Example 3.
FH12501620.1
Attorney Docket: DCY-13025 Table 4. Oligonucleotide conjugates
C16 and C22 in Duplexes K-Q refer to the number of carbon atoms in the chain from the triazole to the carboxylic acid or the number of carbons in the chain from the amide (not inclusive) to the carboxylic acid. Although the carbon atom in the -COOH group is shown above, it is counted as parted of alkyl chain. The results in FIGs.8A-8D show that all 2’Adem conjugated carboxylates were liver sparing as compared to the blunt C22 conjugate. Knockdown in adipose tissue was similar among the conjugates with a minor increase in knockdown observed in conjugates comprising triazole (see e.g., ademG-PEG
4-triazole-C16-COOH). Example 7: Alkyl Chain Length Does Not Alter Carboxylate Duration To better understand the duration of the carboxylate, C22 or C16 conjugates linked to the oligonucleotide via an alkyl chain or a 2’Adem linker were compared (as depicted in FIG.9). Briefly, mice were subcutaneously (s.c.) administered oligonucleotides comprising a modified sense strand of SEQ ID NO: 5 and a modified antisense strand of SEQ ID NO: 10 linked to the conjugates indicated in Table 5 to generate the corresponding oligonucleotide, at a dose of 10 mg/kg. Seven or twenty-one days after administration, tissue was collected, and total RNA was subjected to qRT-PCT analysis to determine Aldh2 mRNA levels as described in Example 3. Table 5. Oligonucleotide conjugates
FH12501620.1
Attorney Docket: DCY-13025
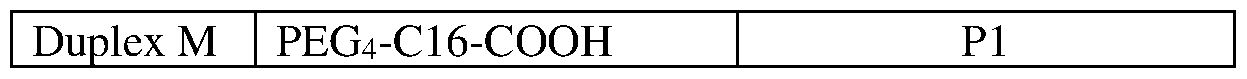
C16 and C22 in Duplex L and P refer to the number of carbon atoms in the chain from the triazole to the carboxylic acid or the number of carbons in the chain from the amide (not inclusive) to the carboxylic acid. Although the carbon atom in the -COOH group is shown above, it is counted as parted of alkyl chain. The results in FIGs.10A-10D and FIGs.11A-11D show similar liver sparing effects when comparing control conjugate (no carboxylate) to conjugates comprising a carboxylate (comparing C22 and C16 to AdemG-C22-COOH and AdemG-C16-COOH ). Similar knockdown was observed between alkyl chain lengths (AdemG-C22-COOH and AdemG- C16-COOH) at days 7 and 21 post-administration. Thus, alkyl group length does not appear to impact duration and knockdown efficacy of the RNAi oligonucleotides. Example 8: Distal Di-Carboxylate Increases Peripheral Tissue Activity To determine if the location or number of carboxylate conjugates impacts oligonucleotide activity, C16 conjugates were compared when conjugated at position 1 of the sense strand, position 1 and the 3’ terminal position of the sense strand, or at position 1 and position 2 of the sense strand (as depicted in FIG.12). Briefly, mice were subcutaneously (s.c.) administered oligonucleotides comprising a modified sense strand of SEQ ID NO: 5, 7, or 8 and a modified antisense strand of SEQ ID NO: 10 linked to the conjugates indicated in Table 6 to generate the corresponding oligonucleotide, at a dose of 10 mg/kg. Seven days after administration, tissue was collected, and total RNA was subjected to qRT-PCT analysis to determine Aldh2 mRNA levels as described in Example 3. Table 6. Oligonucleotide conjugates

C16 in Duplexes P, T, and V refer to the number of carbon atoms in the chain from the triazole to the carboxylic acid or the number of carbons in the chain from the amide (not inclusive) to the carboxylic acid. Although the carbon atom in the -COOH group is shown, it is counted as parted of alkyl chain (C16). The results in FIGs.13A-13D show similar liver sparing effects when comparing control conjugate (no carboxylate) to conjugates comprising a carboxylate (C16-COOH).
FH12501620.1
Attorney Docket: DCY-13025 Knockdown was observed in peripheral tissue for all oligonucleotides with an increase in knockdown observed for oligonucleotides comprising two conjugated nucleotides at the terminal positions of the sense strand (Duplex S and T). Thus, inclusion of acyl chains at the terminal positions of the sense strand increases target gene knockdown in peripheral tissue. Example 9: Alkyl Chains and Terminal Carboxylate Effect on Target Gene Knockdown with RNAi Oligonucleotides To understand the effect of oligonucleotides comprising a carboxylate conjugation in non-human primates, cynomolgus monkeys were administered C22 or C16 conjugates as depicted in FIG.14. Briefly, monkeys were subcutaneously (s.c..) administered oligonucleotides comprising a modified sense strand of SEQ ID NO: 5 and a modified antisense strand of SEQ ID NO: 10 linked to the conjugations indicated in Table 7 to generate the corresponding oligonucleotide, at a dose of 10 mg/kg. Twenty-eight days after administration, tissue was collected, and total RNA was subjected to qRT-PCT analysis to determine Aldh2 mRNA levels. Table 7. Oligonucleotide conjugates

C16 and C22 in Duplexes X, Y, Z, and AA refer to the number of carbon atoms in the chain from amide carbon (inclusive) to the carboxylic acid or the number of carbons in the chain from the amide (not inclusive) to the carboxylic acid. Although the carbon atom in the -COOH group is shown, it is counted as parted of alkyl chain (C16 or C22). The results in FIGs.15A-15B show knockdown of target gene expression in liver and muscle 28 days after administration of the oligonucleotides. Example 10: Terminal Carboxylate and their Bioisosteres Effects on Target Gene Knockdown with RNAi Oligonucleotides To understand the effect in non-human primates of oligonucleotides comprising various bioisosteric terminal group conjugation, cynomolgus monkeys were administered conjugates as depicted in FIG.16. Briefly, monkeys were subcutaneously (s.c.) administered oligonucleotides comprising a modified sense strand of SEQ ID NO: 5 and a modified
FH12501620.1
Attorney Docket: DCY-13025 antisense strand of SEQ ID NO: 10 linked to the conjugations indicated in Table 8 to generate the corresponding oligonucleotide, at a dose of 10 mg/kg. Twenty-eight days after administration, tissue was collected, and total RNA was subjected to qRT-PCT analysis to determine Aldh2 mRNA levels. Table 8. Oligonucleotide conjugates

The carbon chain lengths (e.g. C16 in Duplexes P, AC, AD, AE, AF, and AG refer to the number of carbon atoms in the chain from amide carbon to the carboxylic acid or bioisosteric equivalent. In Duplex P, although the carbon atom in the -COOH group is shown, it is counted as part of alkyl chain (C16). Similarly, in Duplexes AC, AD, AE, AF, and AG, although the carbon atom of the amide is shown, it is counted as part of the alkyl chain. In Duplex AB, the C19 chain length is inclusive of both the amide carbon, and both carboxylate carbons, but not the methyl at the terminal position. The results in FIGs.17A-17D show knockdown of target gene expression in liver, skeletal muscle, heart and gWAT 28 days after administration of the oligonucleotides. Example 11: Terminal Alkyl Chains, Terminal Carboxylate, and their Bioisosteres Effects on Target Gene Knockdown with RNAi Oligonucleotides To understand the effect in non-human primates of oligonucleotides comprising various bioisosteric terminal group conjugation, cynomolgus monkeys were administered conjugates as depicted in FIG.18. Briefly, monkeys were subcutaneously (s.c.) administered oligonucleotides comprising a modified sense strand of SEQ ID NO: 5 and a modified antisense strand of SEQ ID NO: 10 linked to the conjugations indicated in Table 9 to generate the corresponding oligonucleotide, at a dose of 10 mg/kg. Twenty-eight days after administration, tissue was collected, and total RNA was subjected to qRT-PCT analysis to determine Aldh2 mRNA levels. Table 9. Oligonucleotide conjugates
FH12501620.1
Attorney Docket: DCY-13025

C16 and C17 in Duplexes J, M, AI, AJ, and AK refer to the number of carbon atoms in the chain from amide carbon to the carboxylic acid or bioisosteric equivalent. In Duplex M, although the carbon atom in the -COOH group is shown, it is counted as parted of alkyl chain (C16 or C22). Similarly, in Duplexes AH, M, AI, AJ, and AK although the carbon atom of the amide is shown, it is counted as part of the alkyl chain. The results in FIGs.19A-19D show knockdown of target gene expression in liver, skeletal muscle, heart and gWAT 28 days after administration of the oligonucleotides. Example 12: Terminal Aromatic and Non-Aromatic Carboxylate Bioisosteres Effect on Target Gene Knockdown with RNAi Oligonucleotides To understand the effect in non-human primates of oligonucleotides comprising various aromatic, non-aromatic cyclic, and heterocyclic terminal group conjugation, cynomolgus monkeys were administered conjugates as depicted in FIG.20. Briefly, monkeys were subcutaneously (s.c.) administered oligonucleotides comprising a modified sense strand of SEQ ID NO: 5 and a modified antisense strand of SEQ ID NO: 10 linked to the conjugations indicated in Table 10 to generate the corresponding oligonucleotide, at a dose of 10 mg/kg. Twenty-eight days after administration, tissue was collected, and total RNA was subjected to qRT-PCT analysis to determine Aldh2 mRNA levels. Table 10. Oligonucleotide conjugates
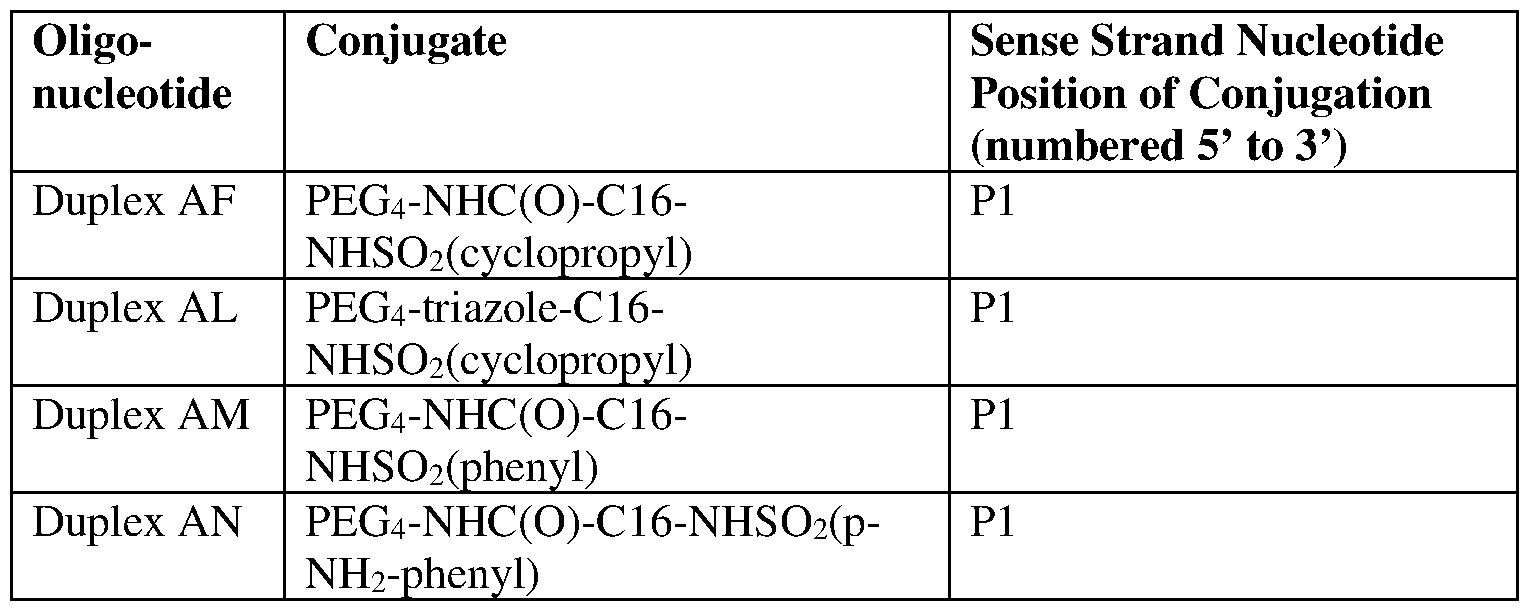
Attorney Docket: DCY-13025

C16 or C22 in Duplexes J, M, AI, AJ, and AK refer to the number of carbon atoms in the chain from amide carbon to the carboxylic acid or bioisosteric equivalent. In Duplex M, although the carbon atom in the -COOH group is shown, it is counted as parted of alkyl chain (C16). Similarly, in Duplexes AF,AL, AM, AN, AO, AP, AQ, and M, although the carbon atom of the amide is shown, it is counted as part of the alkyl chain. The results in FIGs.21A-21D show knockdown of target gene expression in liver, skeletal muscle, heart and gWAT 28 days after administration of the oligonucleotides. Example 13: Terminal Sulfonamides Effect on Target Gene Knockdown with RNAi Oligonucleotides To understand the effect in non-human primates of oligonucleotides comprising various sulfonamide terminal group conjugation, cynomolgus monkeys were administered conjugates as depicted in FIG.22. Briefly, monkeys were subcutaneously (s.c.) administered oligonucleotides comprising a modified sense strand of SEQ ID NO: 5 and a modified antisense strand of SEQ ID NO: 10 linked to the conjugations indicated in Table 11 to generate the corresponding oligonucleotide, at a dose of 10 mg/kg. Twenty-eight days after administration, tissue was collected, and total RNA was subjected to qRT-PCT analysis to determine Aldh2 mRNA levels. Table 11. Oligonucleotide conjugates
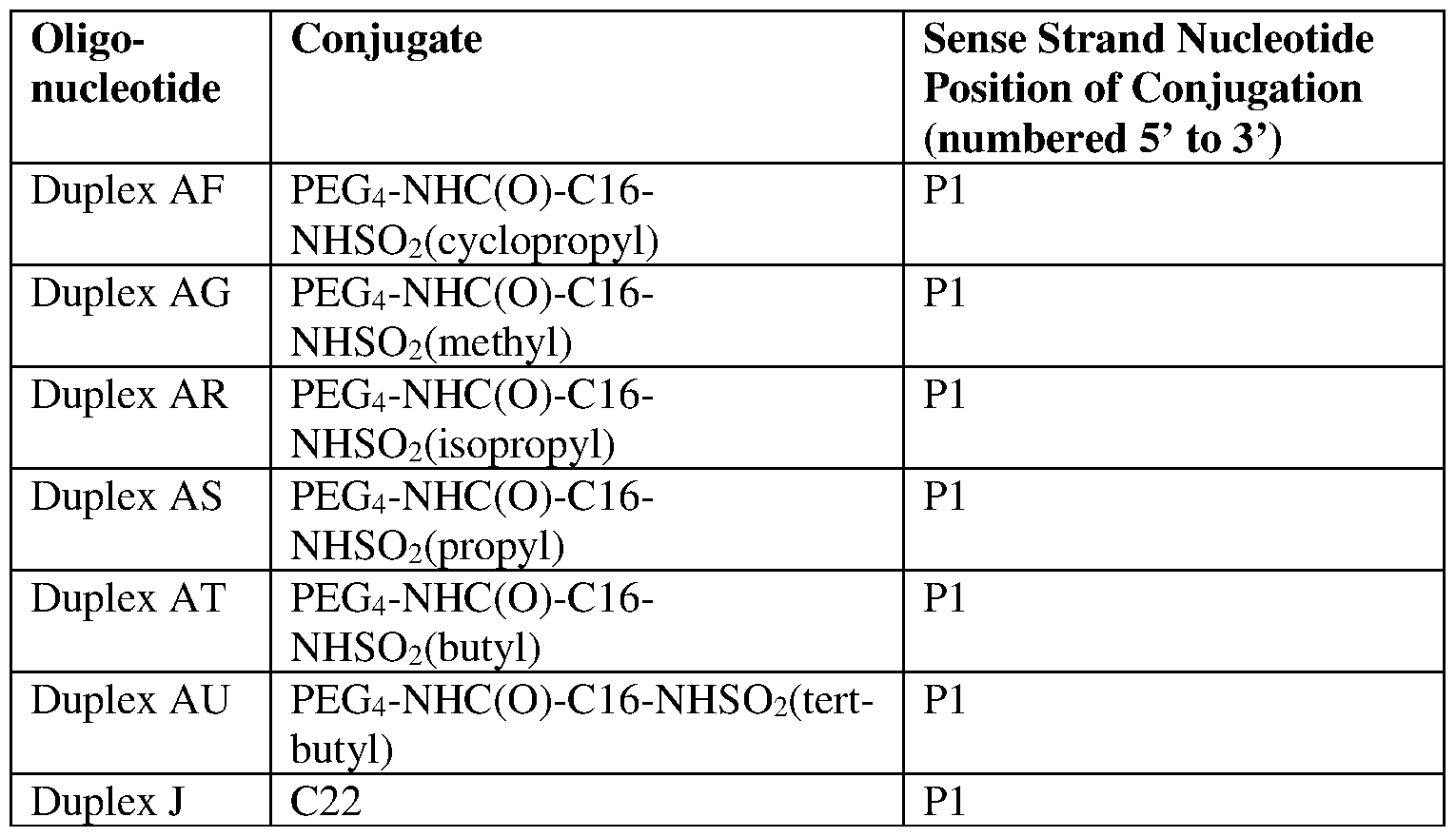
Attorney Docket: DCY-13025 C16 or C22 in Duplexes J, AF, AG, AR, AS, AT, and AU refer to the number of carbon atoms in the chain from amide carbon to the carboxylic acid or bioisosteric equivalent. In Duplexes AF, AG, AR, AS, AT, and AU, although the carbon atom of the amide is shown, it is counted as part of the alkyl chain. The results in FIGs.23A-23D show knockdown of target gene expression in liver, skeletal muscle, heart and gWAT 28 days after administration of the oligonucleotides. Example 14: Terminal Aromatic and Non-Aromatic Sulfonamides Effect on Target Gene Knockdown with RNAi Oligonucleotides To understand the effect in non-human primates of oligonucleotides comprising various aromatic and non-aromatic sulfonamide conjugation, cynomolgus monkeys were administered conjugates as depicted in FIG.24. Briefly, monkeys were subcutaneously (s.c.) administered oligonucleotides comprising a modified sense strand of SEQ ID NO: 5 or 7 and a modified antisense strand of SEQ ID NO: 10 linked to the conjugations indicated in Table 12 to generate the corresponding oligonucleotide, at a dose of 10 mg/kg. Twenty-eight days after administration, tissue was collected, and total RNA was subjected to qRT-PCT analysis to determine Aldh2 mRNA levels. Table 12. Oligonucleotide conjugates

C16 or C22 in Duplexes J, AF, AV, AW, AX, AY, and AZ refer to the number of carbon atoms in the chain from amide carbon to the carboxylic acid or bioisosteric equivalent. C16 in Duplex T refers to the number of carbon atoms in the chain from the triazole to the carboxylic acid. In Duplexes AF, AV, AW, AX, AY, and AZ, although the carbon atom of the amide is shown, it is counted as part of the alkyl chain The results in FIGs.25A-25D show knockdown of target gene expression in liver, skeletal muscle, heart and gWAT 28 days after administration of the oligonucleotides.
FH12501620.1
Attorney Docket: DCY-13025 SEQUENCE KEY [MePhosphonate-4O-mXs] – 4’-O-monomethylphosphonate-2’-O-methyl modified nucleotide [mX] – 2’-O- methyl modified nucleotide with phosphodiester linkages to neighboring nucleotides [fX] – 2’-fluoro modified nucleotide with phosphodiester linkages to neighboring nucleotides [mXs]- 2’-O-methyl modified nucleotide with a phosphorothioate linkage to the neighboring nucleotide [fXs] – 2’-fluoro modified nucleotide with a phosphorothioate linkage to the neighboring nucleotide [ademX]- nucleotide with a linker [5aminoC6]- six carbon amino linker [5C3] – linkage between a triazole and phosphate SEQUENCE LISTING
FH12501620.1
Attorney Docket: DCY-13025
FH12501620.1