WO2025046513A1 - Methods of manufacturing myeloid-derived cells from hematopoietic stem cells and compositions and uses thereof - Google Patents
Methods of manufacturing myeloid-derived cells from hematopoietic stem cells and compositions and uses thereof Download PDFInfo
- Publication number
- WO2025046513A1 WO2025046513A1 PCT/IB2024/058426 IB2024058426W WO2025046513A1 WO 2025046513 A1 WO2025046513 A1 WO 2025046513A1 IB 2024058426 W IB2024058426 W IB 2024058426W WO 2025046513 A1 WO2025046513 A1 WO 2025046513A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- cell
- cells
- precursor
- population
- myeloid
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N5/00—Undifferentiated human, animal or plant cells, e.g. cell lines; Tissues; Cultivation or maintenance thereof; Culture media therefor
- C12N5/06—Animal cells or tissues; Human cells or tissues
- C12N5/0602—Vertebrate cells
- C12N5/0634—Cells from the blood or the immune system
- C12N5/0636—T lymphocytes
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K40/00—Cellular immunotherapy
- A61K40/10—Cellular immunotherapy characterised by the cell type used
- A61K40/11—T-cells, e.g. tumour infiltrating lymphocytes [TIL] or regulatory T [Treg] cells; Lymphokine-activated killer [LAK] cells
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K40/00—Cellular immunotherapy
- A61K40/10—Cellular immunotherapy characterised by the cell type used
- A61K40/17—Monocytes; Macrophages
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K40/00—Cellular immunotherapy
- A61K40/30—Cellular immunotherapy characterised by the recombinant expression of specific molecules in the cells of the immune system
- A61K40/31—Chimeric antigen receptors [CAR]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K40/00—Cellular immunotherapy
- A61K40/40—Cellular immunotherapy characterised by antigens that are targeted or presented by cells of the immune system
- A61K40/41—Vertebrate antigens
- A61K40/42—Cancer antigens
- A61K40/4202—Receptors, cell surface antigens or cell surface determinants
- A61K40/421—Immunoglobulin superfamily
- A61K40/4211—CD19 or B4
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K40/00—Cellular immunotherapy
- A61K40/40—Cellular immunotherapy characterised by antigens that are targeted or presented by cells of the immune system
- A61K40/41—Vertebrate antigens
- A61K40/42—Cancer antigens
- A61K40/428—Undefined tumor antigens, e.g. tumor lysate or antigens targeted by cells isolated from tumor
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2501/00—Active agents used in cell culture processes, e.g. differentation
- C12N2501/10—Growth factors
- C12N2501/125—Stem cell factor [SCF], c-kit ligand [KL]
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2501/00—Active agents used in cell culture processes, e.g. differentation
- C12N2501/10—Growth factors
- C12N2501/145—Thrombopoietin [TPO]
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2501/00—Active agents used in cell culture processes, e.g. differentation
- C12N2501/20—Cytokines; Chemokines
- C12N2501/22—Colony stimulating factors (G-CSF, GM-CSF)
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2501/00—Active agents used in cell culture processes, e.g. differentation
- C12N2501/20—Cytokines; Chemokines
- C12N2501/26—Flt-3 ligand (CD135L, flk-2 ligand)
Definitions
- the present disclosure relates to myeloid cells and genetically modified myeloid cells, precursors thereof, and improved methods of manufacturing the same. More specifically, the present disclosure relates to methods of manufacturing myeloid lineage cells from stem cells, such as hematopoietic stem cells (HSCs).
- stem cells such as hematopoietic stem cells (HSCs).
- the stem cells can be genetically modified with a chimeric antigen receptor (CAR) to generate genetically modified myeloid-derived CAR cells, whereby the HSCs are genetically modified with a chimeric antigen receptor (CAR) prior to expansion and differentiation into myeloid lineage cells.
- CAR chimeric antigen receptor
- Chimeric antigen receptors are engineered receptors that can combine a desired specificity with the functionality of an immune cell to target cancer cells. Typically, these receptors graft the specificity of a monoclonal antibody onto a T cell. The receptors are called chimeric because they are fusions of parts from different sources.
- CAR-T cell therapy refers to a treatment that uses such transformed cells primarily for cancer therapy.
- the general premise of CAR-engineered immune cells is to endow such cells with the ability to target markers found on diseased cells, e.g., cancer cells. In the case of CAR-T cells, scientists can remove T cells from a person, genetically alter them to express a CAR, and put them back into the patient for them to attack the diseased cells.
- CAR-M CAR-macrophages
- Macrophages are well recognized as effector cells that eliminate cancer cells due to their phagocytic ability and are therefore drawing attention in the field of immunotherapy of cancers.
- CAR-M therapy has shown its effective anti-tumor ability in animal experiments. Compared with CAR-T and CAR-NK, CAR-M has its unique advantages as a new cell immunotherapy, but it also has many shortcomings that must be overcome. Peripheral and mobilized monocytes present significant challenges as sources for CAR-M. For example, monocytes do not proliferate, have a short half-life, and must extravasate through vessels and into tissues to differentiate into long-lived macrophages. Engineering monocytes is a significant challenge that results in loss of viability and migratory capacity.
- compositions and methods that enable the production of myeloid cells, including myeloid cells expressing a CAR, such as CAR-M cells, that can be effectively used to treat disease and disorders such as cancers.
- the present application is directed to improved methods of generating myeloid-derived cells, and myeloid-derived CAR cells, such as CAR-M cells, from precursor cells, such as stem cells, and associated compositions and methods for use of the same.
- a myeloid-derived cell comprising the step of: contacting a precursor cell with a precursor media comprising a precursor cytokine mixture, whereby the number of precursor cell(s) increases to generate a cell population comprising a plurality of precursor cells.
- the precursor cell is a stem cell.
- the method comprises the step of: contacting said precursor cell with a nucleic acid molecule comprising a polynucleotide sequence encoding a chimeric antigen receptor (CAR), whereby said nucleic acid is introduced into said precursor cell to generate a precursor CAR cell comprising a chimeric antigen receptor (CAR).
- said precursor cell is contacted with said nucleic acid molecule prior to, or concurrently with, being contacted with said precursor media.
- the method comprises the steps of: a) contacting said precursor cell with a nucleic acid molecule comprising a polynucleotide sequence encoding a chimeric antigen receptor (CAR), whereby said nucleic acid is introduced into said precursor cell to generate a precursor CAR cell comprising a chimeric antigen receptor (CAR), and b) contacting said precursor CAR cell with a precursor media comprising a precursor cytokine mixture, whereby the number of precursor CAR cell(s) increases to generate a cell population comprising a plurality of precursor CAR cells, wherein step a) occurs prior to step b).
- a nucleic acid molecule comprising a polynucleotide sequence encoding a chimeric antigen receptor (CAR)
- CAR chimeric antigen receptor
- the method further comprises the step of: c) contacting said cell population comprising a plurality of precursor cells and/or said cell population comprising a plurality of precursor CAR cells with a myeloid media comprising a myeloid cytokine mixture, whereby said precursor cell(s) differentiates into a myeloid-lineage cell to generate a myeloid-derived cell and/or a mixed cell population comprising a plurality of myeloid-derived cells and/or a plurality of precursor cells, and/or said precursor CAR cell(s) differentiates into a myeloid-lineage cell to generate a myeloid- derived CAR cell and/or a mixed cell population comprising a plurality of myeloid-derived CAR cells and/or a plurality of said precursor CAR cells.
- the method further comprises the step of: c) contacting said mixed cell population with a myeloid media comprising a myeloid cytokine mixture, whereby upon contact with said myeloid media: a. the ratio of said myeloid-derived cells to said precursor cells in said mixed cell population is increased; and/or b. the ratio of myeloid-derived CAR cells relative to precursor CAR cells in said mixed cell population is increased.
- the precursor media is removed prior to addition of the myeloid media.
- the stem cell is a bone-marrow-derived stem cell.
- said stem cell is a hematopoietic stem cell (HSC).
- said stem cell is CD34+.
- said stem cell is a pluripotent stem cell.
- said stem cell is isolated from a subject.
- said subject is a human subject.
- said isolated stem cell is cryopreserved prior to contacting said cell with a precursor media and/or contacting said cell with a nucleic acid molecule.
- said cryopreserved stem cell is thawed prior to contacting said cell with a precursor media and/or contacting said cell with a nucleic acid molecule.
- said stem cell is present within a population of stem cells.
- said population comprises a plurality of stem cell(s).
- between 80%-99.9% of said population is CD34+ cells.
- at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% of said population is CD34+ cells.
- said stem cell is maintained in a culture medium for between 2-144 hours following contact with said nucleic acid molecule.
- said polynucleotide encoding is integrated into the stem cell chromosome.
- a selection agent is added to said culture medium.
- said precursor cytokine mixture comprises at least one cytokine selected from the following: stem cells factor (SCF), Flt3 ligand, thrombopoietin (TPO), Interleukin-3 (IL-3); Granulocyte colonystimulating factor (G-CSF), Interleukin-6 (IL-6), Granulocyte-macrophage colony-stimulating factor (GM-CSF), and Macrophage colony-stimulating factor (MCSF).
- said precursor media comprises G-CSF at a concentration between about 100 ng/mL and 200 ng/mL.
- precursor media comprises G- CSF at a concentration between about 100 ng/mL and 150 ng/mL.
- precursor cytokine mixture comprises G-CSF, GM-CSF, SCF, and TPO.
- said precursor media comprises GM-CSF at a concentration between about 75 ng/mL and 125 ng/mL.
- said precursor media comprises SCF at a concentration between about 75 ng/mL and 125 ng/mL.
- precursor media comprises TPO at a concentration between about 75 ng/mL and 125 ng/mL.
- the precursor media comprises G-CSF at a concentration of 150 ng/mL, GM-CSF at a concentration of 100 ng/mL, SCF at a concentration of 100 ng/mL, and/or TPO at a concentration of 100 ng/mL.
- said myeloid media comprises a myeloid cytokine mixture comprising Macrophage colony-stimulating factor (MCSF), Interleukin-3 (IL-3), and/or Interleukin-6 (IL-6), and/or a combination thereof.
- MCSF Macrophage colony-stimulating factor
- IL-3 Interleukin-3
- IL-6 Interleukin-6
- said myeloid media comprises stem cells factor (SCF), Flt3 ligand, thrombopoietin (TPO), Granulocyte colony-stimulating factor (G-CSF), and/or Granulocyte-macrophage colony-stimulating factor (GM-CSF), at a concentration of at most 50 ng/mL, at most 40 ng/mL, at most 30 ng/mL, at most 20 ng/mL, at most 10 ng/mL, at most 5 ng/mL, or below 5 ng/mL.
- SCF stem cells factor
- Flt3 ligand thrombopoietin
- TPO thrombopoietin
- G-CSF Granulocyte colony-stimulating factor
- GM-CSF Granulocyte-macrophage colony-stimulating factor
- said precursor cell(s) and/or said population comprising a plurality of precursor cells said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population, is maintained in said precursor media until the number of precursor cell(s), precursor CAR cells, myeloid-derived cells and/or myeloid-derived CAR cells is increased by at least 20-fold, at least 50-fold, at least 75-fold, at least 100-fold, at least 200-fold, at least 300-fold, at least 400-fold, at least 500-fold, or more than 500-fold.
- said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population is maintained in said precursor media until the number of precursor cell(s), precursor CAR cells, myeloid-derived cells and/or myeloid-derived CAR cells is at least 10 4 , at least 10 5 , at least 10 6 , at least 10 7 , at least 10 8 , at least 10 9 , at least 10 10 , at least 10 11 , at least 10 12 , at least 10 13 , at least 10 14 , at least 10 15 , at least 10 16 , at least 10 17 , at least 10 18 , at least 10 19 , at least IO 20 , or more than IO 20 .
- said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population is maintained in said precursor media for at least 2, at least 3, at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, at least 10, at least 11, or at least 12 days.
- said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population is maintained in precursor media for between 9-11 days.
- said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population is maintained in precursor media for 10 days.
- At least 20%, at least 30%, at least 40%, at least 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95% of cells within said mixed cell population are CD15+; d. between about 60%-85% of cells within said mixed cell population areCD15+; e. at least 2%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, at least 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of cells within said mixed cell population are CD1 lb+; f. between about 30%- 80% of cells within said mixed cell population are CD1 lb+; g.
- said mixed population is contacted with said myeloid media after contact with said precursor media.
- said mixed population is maintained in said myeloid media until: a. no more than 5%, no more than 4%, no more than 3%, no more than 2%, no more than 1%, no more than 0.9%, no more than 0.8%, no more than 0.7%, no more than 0.6%, no more than 0.5%, no more than 0.4%, no more than 0.3%, no more than 0.2%, no more than 0.1%, or less than 0.1% of cells within said mixed cell population are CD34+; b. at least 70%, at least 75%, at least 80%, at least 90%, or at least 95% of cells within said mixed cell population are CD15+; c. between about 50%-95% of cells within said mixed cell population are CD15+; d.
- At least 70%, least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or at least 99.5% of cells within said mixed cell population are CD1 lb+; e. between about 75%-99.5% of cells within said mixed cell population are CD1 lb+; f. at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, at least 50%, at least 60%, at least 70%, at least 80%, or at least 90% of cells within said mixed cell population are CD14+; g. between about 5%-90% of cells within said mixed cell population are CD14+; h. at least 85%, at least 90%, at least 95%, or at least 99% of cells within said mixed cell population are CD13+; and/or i. between about 85%-99% of cells within said mixed cell population are CD13+.
- said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population is maintained in a myeloid media for at least 1, at least 2, at least 3, at least 4, at least 5, at least 6, at least 7 or at least 8 days.
- said mixed cell population comprises at least 20%, at least 40%, at least 60%, least 80%, at least 85%, or at least 90% CD14+ cells after 7 days of being maintained in said myeloid media.
- contacting said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population with said precursor cytokine mixture reduces the ratio of CD34+ cells within said population comprising a plurality of precursor cells, said population comprising a plurality of precursor CAR cells, and/or said mixed cell population in a shorter amount of time compared to a control that is contacted with and/or maintained in a precursor media lacking said precursor cytokine mixture.
- contacting said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population with said precursor media increases: a. the number of CD1 lb+ cells; b. the number of CD13+ cells; c. the number of CD15+ cells; and/or d. the number of CD14+ cells within said population comprising a plurality of precursor cells, said population comprising a plurality of precursor CAR cells, and/or said mixed cell population in a shorter amount of time compared to a control that is contacted in and/or maintained in a precursor media lacking said precursor cytokine mixture
- contacting said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population with said precursor media increases: a. the ratio of CD1 lb+ to CD1 lb- cells; b. the ratio of CD13+ to CD13- cells; c. the ratio of CD15+ to CD15- cells; and/or d.
- CD14+ to CD14- cells within said population comprising a plurality of precursor cells, said population comprising a plurality of precursor CAR cells, and/or said mixed cell population in a shorter amount of time compared to a control that is contacted with and/or maintained in a precursor media lacking said precursor cytokine mixture.
- said myeloid-derived cell is a phagocytic cell.
- said phagocytic cell is a monocyte, macrophage, dendritic cell, neutrophil, or a precursor thereof.
- said phagocytic cell is a monocyte.
- said myeloid-derived cell is a myeloid progenitor cell.
- said CAR comprises: an antigen binding domain; a transmembrane domain; and an intracellular signaling domain. In some embodiments of the above methods, said CAR further comprises an intracellular co-stimulatory signaling domain. In some embodiments of the above methods, said CAR comprises an intracellular co-stimulatory signaling domain having an amino acid sequence having at least 90% sequence identity to a cytoplasmic domain of a herpes virus entry mediator (HVEM) protein set forth as any one of SEQ ID NOs: 1, 2, or 3, or a functional fragment or variant thereof that retains co-stimulatory activity.
- HVEM herpes virus entry mediator
- a genetically modified cell comprising a chimeric antigen receptor (CAR), wherein the genetically modified cell is a precursor cell, a myeloid-derived cell, a myeloid- lineage cell, a myeloid progenitor cell, and/or a precursor thereof.
- CAR chimeric antigen receptor
- the precursor cell comprises a bone marrow- derived cell, a blood-derived cell, or a stem cell.
- the stem cell is a hematopoietic stem cell (HSC).
- the cell is a myeloid precursor cell, such as a myeloid progenitor cell, granulo-monocyte progenitor cell, monocyte-dendritic cell progenitor/monoblasts, promonocyte, or myeloblast.
- said CAR comprises an intracellular co-stimulatory signaling domain having an amino acid sequence having at least 90% sequence identity to a cytoplasmic domain of a herpes virus entry mediator (HVEM) protein set forth as any one of SEQ ID NOs: 1, 2, or 3, or a functional fragment or variant thereof that retains co-stimulatory activity.
- HVEM herpes virus entry mediator
- a cell population comprising any of the genetically modified CAR cells disclosed herein.
- the cell population is generated by any of the methods of manufacturing a myeloid-derived cell disclosed herein.
- At least 40%, at least 50%, at least 60%, at least 70%, at least 80%, at least 90%, at least 95%, or at least 99% of cells within said population expresses said CAR on the cell surface.
- a. no more than 5%, no more than 4%, no more than 3%, no more than 2%, no more than 1%, no more than 0.5%, or no more than 0.1% of cells within said population are CD34+; b.
- At least 50%, at least 55%, at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 86%, at least 87%, at least 88%, at least 89%, or at least 90% of cells within said population are CD14+; c. at least 70%, at least 80%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or at least 99.5%% of cells within said population are CD1 lb+; d. at least 90%, at least 91%, at least 92%, at least 94%, or at least 95% of cells within said population are CD13+; and/or e. at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, or at least 95% of cells within said population are CD15+.
- composition comprising any of the genetically modified cells disclosed herein, or any of the cell populations disclosed herein, and a pharmaceutically acceptable carrier.
- FIGS. 1A-1B show schematics depicting exemplary workflow for generating myeloid-derived cells (FIG. 1A) and genetically modified myeloid-derived CAR cells (FIG. IB) hematopoietic stem cells (HSCs).
- FIG. 1A shows that mobilized HSCs can be expanded and differentiated into myeloid- lineage cells.
- FIG. IB shows that HSCs can be engineered with a CAR prior to expansion and differentiation into myeloid-derived CAR cells.
- FIGS. 2A-2N show the duration and stability of transduced chimeric antigen receptor (CAR- M83-GFP) integration in a starting population of precursor CAR cells subsequently expanded and differentiated into myeloid-derived cells using the methods disclosed herein.
- CAR surface expression and GFP expression were analyzed by flow cytometry and presented as expression intensity (x-axis) against percentage of cell population normalized to mode for the indicated population (y-axis).
- FIG. 2A shows a histogram depicting CAR expression in transduced cells (“CAR”) compared to control cells (untransduced; “UTD”) in precursor media 6 days, and 72-hours post-transduction of a polynucleotide encoding a CAR.
- CAR transduced cells
- UTD untransduced
- FIG. 2B shows a histogram depicting CAR expression in transduced cells (“CAR”) compared to control cells (untransduced; “UTD”) in precursor media 8 days.
- FIG. 2C shows a histogram depicting CAR expression in transduced cells (“CAR”) compared to control cells (untransduced; “UTD”) in precursor media 10 days.
- FIG. 2D shows a histogram depicting CAR expression in transduced cells (“CAR”) compared to control cells (untransduced; “UTD”) in precursor media 14 days.
- FIG. 2E shows a histogram depicting GFP expression in transduced cells (“GFP”) compared to control cells (untransduced; “UTD”) in precursor media 6 days.
- FIG. 2F shows a histogram depicting GFP expression in transduced cells (“GFP”) compared to control cells (untransduced; “UTD”) in precursor media 8 days.
- FIG. 2G shows a histogram depicting GFP expression in transduced cells (“GFP”) compared to control cells (untransduced; “UTD”) in precursor media 10 days.
- FIG. 2H shows a histogram depicting GFP expression in transduced cells (“GFP”) compared to control cells (untransduced; “UTD”) in precursor media 14 days.
- FIG. 21 shows a histogram depicting CAR expression in transduced cells (“CAR”) compared to control cells (untransduced; “UTD”) after 14 days in precursor media, followed by 8 days in Ml macrophage activation media.
- CAR histogram depicting CAR expression in transduced cells
- FIG. 2J shows a histogram depicting GFP expression in transduced cells (“GFP”) compared to control cells (untransduced; “UTD”) after 14 days in precursor media, followed by 8 days in Ml macrophage activation media.
- FIG. 2K shows a flow dot plot depicting CD 13 surface marker expression in the cell population of FIGS. 2D and 2H (i.e, maintained 14 days in precursor media). The number in the box indicates the percentage of the population that is CD13+.
- FIG. 2L shows a flow dot plot depicting CD 15 surface marker expression in the cell population of FIGS. 2D and 2H. The number in the box indicates the percentage of the population that is CD15+.
- FIG. 1 shows a histogram depicting GFP expression in transduced cells (“GFP”) compared to control cells (untransduced; “UTD”) after 14 days in precursor media, followed by 8 days in Ml macrophage activation media.
- FIG. 2K shows a flow dot plot depicting CD 13 surface marker expression
- FIG. 2M shows a flow dot plot depicting CD1 lb+ surface marker expression in the cell population of FIGS. 2D and 2H. The number in the box indicates the percentage of the population that is CD1 lb+.
- FIG. 2N shows a flow dot plot depicting CD14 surface marker expression in the cell population of FIGS. 2D and 2H. The number in the box indicates the percentage of the population that is CD14+.
- FIGS. 3A-3B show the efficacy of CAR transduction and differentiation of hematopoietic stem cells transduced with a CAR using the methods disclosed herein. Flow cytometry analysis was used to determine CAR and CD14 surface expression.
- FIG 3A is a flow plot depicting percentage of a cell population expressing a CAR (GFP+).
- FIG. 3B is a flow plot depicting percentage of the population of CAR-expressing cells from FIG. 3A that are CD14+.
- FIGS. 4A-4C show three graphs demonstrating the expansion of hematopoietic stem cells (HSCs) in precursor media. HSCs were maintained in precursor media for up to 12 days, and cell number was analyzed over time. Increase in cell number and expansion of the cell population in each precursor media at the indicated day is depicted as cumulative fold-expansion relative to day 0.
- HSCs hematopoietic stem cells
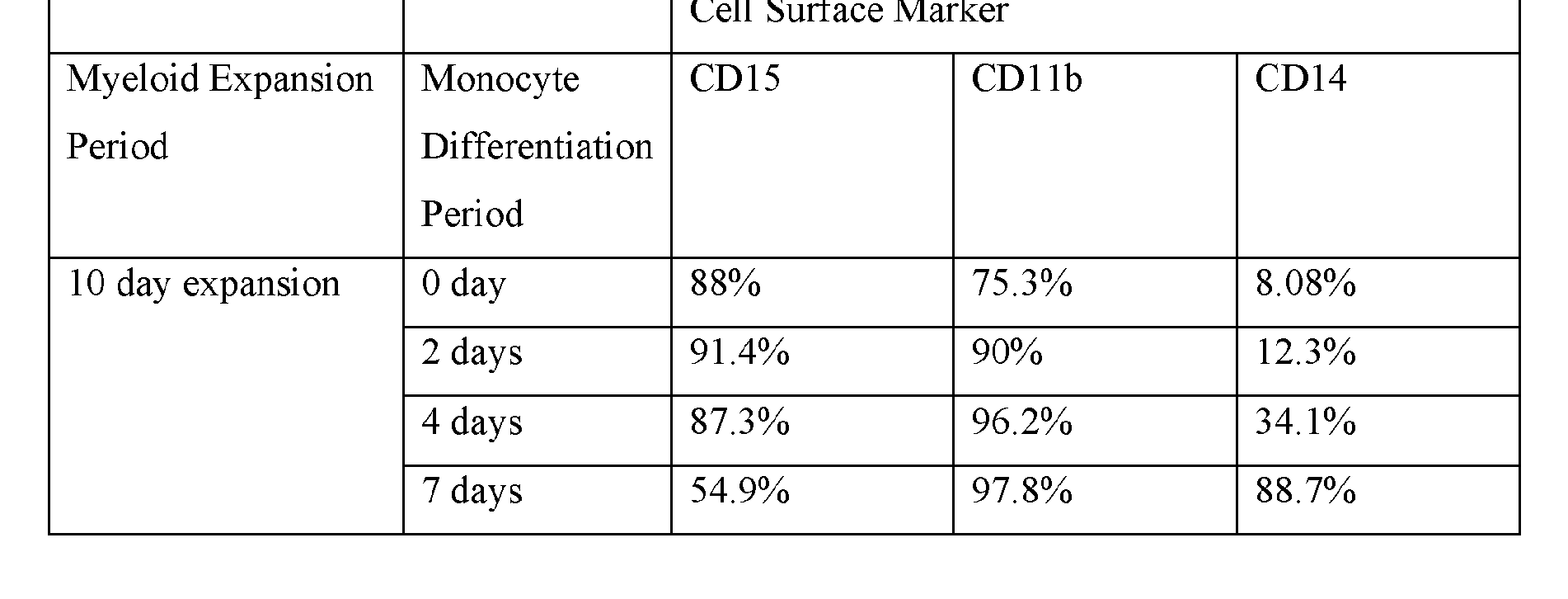
- FIG. 5 is a flow dot plot depicting CD14 and CD1 lb surface marker expression in a cell population of hemopoietic stem cells (HSCs) maintained in My el 4.1 precursor media for 7 days, followed by myeloid media for 9 days.
- HSCs hemopoietic stem cells
- the number depicted in each box represents the percentage of the cell population presenting the indicated marker(s).
- FIG. 6 is a graph depicting CD14 surface marker expression in populations of hemopoietic stem cells (HSCs) maintained in three different precursor medias (My el 1,; My el 2; and My el 3) for 8 days, and then maintained in myeloid media for out tothree days. “%CD14+” represents the percentage of the cell population expressing CD14 in the indicated media at each day.
- FIG. 7 shows chemoattractant-dependent (C5a) migratory capacity through human umbilical venular endothelial cells (HUVEC) endothelial monolayer of monocytes derived from HSCs (“HSC- Monocytes”; see “1” and “2”) compared to stimulated monocytes (“Stim. Monocytes”; see “3” and “4”).
- HSC- Monocytes human umbilical venular endothelial cells
- Stim. Monocytes see “3” and “4”.
- the number of migratory cells in each well were analyzed and are presented every 1 hour over 48 hours.
- FIGS. 8A-8B depict phagocytic efficacy of monocytes generated from hemopoietic stem cells (HSCs) using the expansion and differentiation methods disclosed herein.
- FIG. 8A is a schematic depicting a synthetic bead uptake assay used to analyze phagocytic capacity of the tested cells.
- FIG. 8B is a graph showing uptake by monocytes during co-culture with beads over 6 hours. Individual beads phagocytosed by monocytes detected (“red object count / well) are shown at the indicated time. A negative control is presented where cells were treated with cytochalasin D (“UTD + CytoD”).
- FIG. 8A is a schematic depicting a synthetic bead uptake assay used to analyze phagocytic capacity of the tested cells.
- FIG. 8B is a graph showing uptake by monocytes during co-culture with beads over 6 hours. Individual beads phagocytosed by monocytes detected (“red object count / well) are shown at the indicated time. A negative
- HSCs hemopoietic stem cells
- a can mean one or more than one.
- a cell can mean a single cell or a multiplicity of cells.
- ranges such as from 1-10 should be considered to have specifically disclosed subranges such as from 1 to 3, from 1 to 4, from 1 to 5, from 1 to 6, from 1 to 7, from 1 to 8, from 1 to 9, from 2 to 4, from 2 to 6, from 2 to 8, from 2 to 10, from 3 to 6, etc., as well as individual numbers within that range, for example, 1, 2, 3, 4, 5, 6, 7, 8, 9 and 10. This applies regardless of the breadth of the range.
- a numerical range is indicated herein, it is meant to include any cited numeral (fractional or integral) within the indicated range.
- the phrases “ranging/ranges between” a first indicate number and a second indicate number and “ranging/ranges from” a first indicate number “to” a second indicate number are used herein interchangeably and are meant to include the first and second indicated numbers and all the fractional and integral numerals there between.
- the recitation of a numerical range for a variable is intended to convey that the present disclosure may be practiced with the variable equal to any of the values within that range.
- the variable can be equal to any integer value within the numerical range, including the endpoints of the range.
- variable can be equal to any real value within the numerical range, including the end-points of the range.
- the term “gene” or “coding sequence”, herein used interchangeably, refers to a functional nucleic acid unit encoding a protein, polypeptide, or peptide.
- this functional term includes genomic sequences, cDNA sequences, and smaller engineered gene segments that express, or may be adapted to express proteins, polypeptides, domains, peptides, fusion proteins, and mutants.
- nucleic acid As used herein, “nucleic acid,” “nucleotide sequence,” and “polynucleotide” are used interchangeably and encompass both RNA and DNA, including cDNA, genomic DNA, mRNA, synthetic (e.g, chemically synthesized) DNA or RNA and chimeras of RNA and DNA.
- polynucleotide, nucleotide sequence, or nucleic acid refers to a chain of nucleotides without regard to length of the chain.
- fragment will be understood to mean a nucleotide sequence of reduced length relative to a reference nucleic acid or nucleotide sequence and comprising, consisting essentially of and/or consisting of a nucleotide sequence of contiguous nucleotides identical to the reference nucleic acid or nucleotide sequence.
- a nucleic acid fragment according to the invention may be, where appropriate, included in a larger polynucleotide of which it is a constituent.
- such fragments can comprise, consist essentially or and/or consist of, oligonucleotides having a length of at least about 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, or 25 consecutive nucleotides of a nucleic acid or nucleotide sequence according to the invention.
- a “mutation” is any change in a nucleic acid sequence. Nonlimiting examples comprise insertions, deletions, duplications, substitutions, inversions, and translocations of any nucleic acid sequence, regardless of how the mutation is brought about and regardless of how or whether the mutation alters the functions or interactions of the nucleic acid.
- a mutation may produce altered enzymatic activity of a ribozyme, altered base pairing between nucleic acids (e.g. RNA interference interactions, DNA-RNA binding, etc.), altered mRNA folding stability, and/or how a nucleic acid interacts with polypeptides (e.g. DNA-transcription factor interactions, RNA-ribosome interactions, gRNA-endonuclease reactions, etc.).
- a mutation might result in the production of proteins with altered amino acid sequences (e.g. missense mutations, nonsense mutations, frameshift mutations, etc.) and/or the production of proteins with the same amino acid sequence (e.g. silent mutations).
- Mutations may create no observed change in a cell while others that encode for an identical protein sequence nevertheless result in an altered cell phenotype (e.g. due to codon usage bias, altered secondary protein structures, etc.). Mutations may occur within coding regions (e.g., open reading frames) or outside of coding regions (e.g., within promoters, terminators, untranslated elements, or enhancers), and may affect, for example and without limitation, gene expression levels, gene expression profiles, protein sequences, and/or sequences encoding RNA elements such as tRNAs, ribozymes, ribosome components, and microRNAs.
- RNA elements such as tRNAs, ribozymes, ribosome components, and microRNAs.
- the terms “engineered” or “recombinant” in reference to a phagocyte, gene, nucleic acid and/or protein as used herein, refer to a phagocyte, gene, nucleic acid and/or protein that has been altered through human intervention.
- the term “naturally occurring” as used herein in reference to a phagocyte, gene, nucleic acid and/or protein as used herein, refer to a phagocyte, gene, nucleic acid and/or protein existing in nature and without any human intervention.
- Exemplary human interventions comprise transfection with a heterologous polynucleotide, molecular cloning resulting in a deletion, insertion, modification and/or rearrangement with respect to a naturally occurring sequence such as a naturally occurring sequence in a phagocyte, gene, nucleic acid and/or protein herein described.
- expression or “expressing” refers to the transcription and/or translation of a particular nucleic acid sequence driven by a promoter.
- An expression construct or expression vector can permit transcription of a particular nucleic acid sequence in a cell (e.g., a phagocytic cell).
- An expression cassette may be part of a plasmid, viral genome, or nucleic acid fragment.
- An expression cassette typically comprises at least three components: a promoter sequence, an open reading frame encoding gene(s) of interest, and a 3' untranslated region that, in eukaryotes, usually contains a polyadenylation site.
- An expression cassette can be formed by manipulable fragment of DNA carrying and capable of expressing, one or more genes of interest optionally located between one or more sets of restriction sites.
- Expression cassettes typically comprise further regulatory sequences additional to the promoter to regulate the expression of the gene or genes within the open reading frame herein also indicated as a coding region of the expression cassette.
- "Operably linked" is intended to mean a functional linkage between two or more elements.
- an operable linkage between a promoter of the present invention and a heterologous nucleotide is a functional link that allows for expression of the heterologous nucleic acid molecule.
- Operably linked elements may be contiguous or non-contiguous. When used to refer to the joining of two protein coding regions, by operably linked is intended that the coding regions are in the same reading frame.
- the cassette may additionally contain at least one additional gene to be co-transformed into a cell. Alternatively, the additional gene(s) can be provided on multiple expression cassettes or DNA constructs.
- the expression cassette may additionally contain selectable marker genes.
- an expression cassette Other elements that may be present in an expression cassette include those that enhance transcription (e.g., enhancers) and terminate transcription (e.g., terminators), as well as those that confer certain binding affinity or antigenicity to the recombinant protein produced from the expression cassette.
- enhancers e.g., enhancers
- terminate transcription e.g., terminators
- contact refers to placing the components of a desired interaction together under conditions suitable for enabling and carrying out the interaction.
- a precursor cell and/or a population comprising a plurality of precursor cells can be contacted with a precursor media. Any means of contacting a cell or cell population with a media can be used.
- a cell or cell population can be inoculated, suspended, or maintained in a culture media, such as a liquid culture media in order to contact said cell with said media.
- said cell or cell population can be present within a solution, such as a buffered and/or sterile solution, that can be mixed into a culture media such that the cell(s) come into contact with the precursor media.
- the precursor cells disclosed herein can be contacted with a nucleic acid molecule.
- the nucleic acid molecule can be purified from a biological sample, cell lysate, or culture medium, produced via in vitro transcription, or chemically synthesized.
- the cell can be purified from a biological sample, such as a G-CSF mobilized leukopack from a healthy donor, or a frozen vial.
- the cell and/or nucleic acid molecule can be brought into contact in any solution (e.g., buffered saline solution) or culture medium to allow for introduction of the nucleic acid molecule into the cell. Methods of introducing nucleic acid molecules into cells are readily known in the art.
- polypeptide indicates an organic linear, circular, or branched polymer composed of two or more amino acid monomers and/or analogs thereof.
- polypeptide includes amino acid polymers of any length including full-length proteins and peptides, as well as analogs and fragments thereof.
- a polypeptide of three or more amino acids is also called a protein oligomer, peptide, or oligopeptide.
- peptide and oligopeptide usually indicate a polypeptide with less than 100 amino acid monomers.
- the polypeptide provides the primary structure of the protein, wherein the term “primary structure” of a protein refers to the sequence of amino acids in the polypeptide chain covalently linked to form the polypeptide polymer.
- a protein “sequence” indicates the order of the amino acids that form the primary structure.
- Covalent bonds between amino acids within the primary structure can include peptide bonds or disulfide bonds, and additional bonds identifiable by a skilled person.
- Polypeptides in the sense of the present disclosure are usually composed of a linear chain of alpha-amino acid residues covalently linked by peptide bond or a synthetic covalent linkage.
- the two ends of the linear polypeptide chain encompassing the terminal residues and the adjacent segment are referred to as the carboxyl terminus (C-terminus) and the amino terminus (N-terminus) based on the nature of the free group on each extremity. Unless otherwise indicated, counting of residues in a polypeptide is performed from the N-terminal end (NHz-group), which is the end where the amino group is not involved in a peptide bond to the C-terminal end (-COOH group) which is the end where a COOH group is not involved in a peptide bond.
- NHz-group N-terminal end
- -COOH group the C-terminal end
- Proteins and polypeptides can be identified by x-ray crystallography, direct sequencing, immunoprecipitation, and a variety of other methods as understood by a person skilled in the art.
- Proteins can be provided in vitro or in vivo by several methods identifiable by a skilled person. In some instances where the proteins are synthetic proteins in at least a portion of the polymer two or more amino acid monomers and/or analogs thereof are joined through chemically-mediated condensation of an organic acid (-COOH) and an amine (-NH2) to form an amide bond or a “peptide” bond.
- -COOH organic acid
- -NH2 amine
- amino acid refers to a compound having a free carboxyl group and a free unsubstituted amino group on the a carbon, which may be joined by peptide bonds to form a peptide active agent as described herein.
- a “basic amino acid” refers to any amino acid that is positively charged at a pH of 6.0, including but not limited to R, K, and H.
- An “aromatic amino acid” refers to any amino acid that has an aromatic group in the side-chain coupled to the alpha carbon, including but not limited to F, Y, W, and H.
- function of a gene, a peptide, a protein, or a molecule refers to activity of a gene, a peptide, a protein, or a molecule.
- “Introducing,” “introduce,” and “introduced” in the context of a polynucleotide and/or polypeptide of interest means presenting a nucleotide sequence of interest (e.g., polynucleotide, a nucleic acid construct, and/or a guide nucleic acid) and/or polypeptide of interest to a host organism or cell of said organism (e.g., a mammalian cell) in such a manner that the nucleotide sequence and/or polypeptide gains access to the interior of a cell.
- a nucleotide sequence of interest e.g., polynucleotide, a nucleic acid construct, and/or a guide nucleic acid
- a host organism or cell of said organism e.g., a mammalian cell
- “introducing” includes inserting a nucleic acid molecule (e.g., a recombinant DNA construct) into a cell, by means of transformation, transfection, or transduction.
- the nucleic acid molecule may be incorporated into the genome of the cell (e.g., nuclear chromosome or mitochondrial chromosome), converted into an autonomous replicon, or transiently expressed (e.g., transfected mRNA).
- a “subject” that may be treated by methods of the present disclosure include both human subjects for medical and/or therapeutic purposes and animal subjects for veterinary and drug screening and development purposes.
- Other suitable animal subjects are, in general, mammalian subjects such as primates, bovines, ovines, caprines, porcines, equines, felines, canines, lagomorphs, rodents ( e.g ., rats and mice), etc.
- Human subjects are the most preferred. Human subjects include fetal, neonatal, infant, juvenile, adult and geriatric subjects.
- anti -turn or effect refers to a biological effect which can be manifested by a decrease in tumor volume, a decrease in the number of tumor cells, a decrease in the proliferation rate, a decrease in the number of metastases, an increase in life expectancy, and/or amelioration of various physiological symptoms associated with the cancerous condition.
- An “antitumor effect” can also be manifested by the ability of the peptides, polynucleotides, cells and antibodies of the invention to delay the occurrence of tumor in the first place.
- autologous is meant to refer to any material derived from the same individual to whom it is later to be re-introduced.
- Allogeneic refers to a graft derived from a different animal of the same species.
- Xenogeneic refers to a graft derived from an animal of a different species.
- antibody refers to full-length immunoglobulins as well as to fragments thereof. Such full-length immunoglobulins may be monoclonal, polyclonal, chimeric, humanized, veneered or human antibodies.
- Humanized forms of non-human (e.g., murine) antibodies are chimeric immunoglobulins, immunoglobulin chains or fragments thereof (such as Fv, Fab, Fab 1 , F(ab')2 or other antigen-binding subsequences of antibodies) which contain minimal sequence derived from non-human immunoglobulin.
- humanized antibodies are human immunoglobulins (recipient antibody) in which residues from a complementary-determining region (CDR) of the recipient are replaced by residues from a CDR of a non-human species (donor antibody) such as mouse, rat or rabbit having the desired specificity, affinity, and capacity.
- CDR complementary-determining region
- donor antibody such as mouse, rat or rabbit having the desired specificity, affinity, and capacity.
- Fv framework region (FR) residues of the human immunoglobulin are replaced by corresponding non-human residues.
- Fully human refers to an immunoglobulin, such as an antibody, where the whole molecule is of human origin or consists of an amino acid sequence identical to a human form of the antibody.
- the numbering system to identify amino acid residue positions in the VH and VL of an antibody can follow a system known to one of skill in the art, including Kabat (Wu and Kabat (1970) J Exp Med. 132(2):211-50; Borden and Kabat (1987) PNAS, 84:2440-2443; Kabat et al. U.S. Department of Health and Human Services, 1991), Chothia (Chothia and Lesk (1987) J Mol. Biol., 196(4): 901-917; Chothia et al. (1989) Nature 342:877-883), and the "AHo" system described by Honegger & Pluckthun (2001) Journal of Molecular Biology 309:657-670.
- antigen-binding portion or "antigen-binding fragment” of an antibody (or simply “antibody portion” or “antibody fragment”), as used herein, refers to one or more fragments, portions or domains of an antibody that retain the ability to specifically bind to an antigen. It has been shown that fragments of a full-length antibody can perform the antigen binding function of an antibody.
- binding fragments encompassed within the term "antigen-binding portion" of an antibody include (i) an Fab fragment, a monovalent fragment consisting of the VL, VH, CL1 and CHI domains; (ii) an F(ab')2 fragment, a bivalent fragment comprising two F(ab)' fragments linked by a disulfide bridge at the hinge region; (iii) an Fd fragment consisting of the VH and CHI domains; (iv) an Fv fragment consisting of the VL and VH domains of a single arm of an antibody; (v) a dAb fragment (Ward et al.
- VL and VH are coded for by separate genes, they can be joined, using recombinant methods, by a synthetic linker that enables them to be made as a single contiguous chain in which the VL and VH regions pair to form monovalent molecules (known as single chain Fv (scFv); see e.g., Bird et al. (1988) Science 242:423-426; and Huston et al. (1988) Proc. Natl. Acad. Sci. USA 85:5879- 5883).
- scFv single chain Fv
- single chain antibodies are also intended to be encompassed within the term "antigenbinding portion" of an antibody.
- Other forms of single chain antibodies, such as diabodies, are also encompassed (see e.g., Holliger et al. (1993) Proc. Natl. Acad Sci. USA 90:6444-6448).
- epitope refers to an antigenic determinant that interacts with a specific antigen binding site in the variable region of an antibody molecule known as a paratope.
- a single antigen may have more than one epitope.
- Epitopes may be either conformational or linear.
- a conformational epitope is produced by spatially juxtaposed amino acids from different segments of one (or more) linear polypeptide chain(s).
- a linear epitope is an epitope produced by adjacent amino acid residues in a polypeptide chain.
- an epitope may include other moieties, such as saccharides, phosphoryl groups, or sulfonyl groups on the antigen.
- antibody heavy chain refers to the larger of the two types of polypeptide chains present in all antibody molecules in their naturally occurring conformations.
- an “antibody light chain,” as used herein, refers to the smaller of the two types of polypeptide chains present in all antibody molecules in their naturally occurring conformations, K and 1 light chains refer to the two major antibody light chain isotypes.
- antigen or "Ag” as used herein is defined as a molecule that provokes an immune response. This immune response may involve either antibody production, or the activation of specific immunologically-competent cells, or both.
- antigens can be derived from recombinant or genomic DNA. A skilled artisan will understand that any DNA, which comprises a nucleotide sequence or a partial nucleotide sequence encoding a protein that elicits an immune response therefore encodes an "antigen" as that term is used herein.
- an antigen need not be encoded solely by a full length nucleotide sequence of a gene. It is readily apparent that the present invention includes, but is not limited to, the use of partial nucleotide sequences of more than one gene and that these nucleotide sequences are arranged in various combinations to elicit the desired immune response. Moreover, a skilled artisan will understand that an antigen need not be encoded by a "gene” at all. It is readily apparent that an antigen can be synthesized or can be derived from a biological sample. Such a biological sample can include, but is not limited to a tissue sample, a tumor sample, a cell or a biological fluid.
- an antibody or antigen binding domain which recognizes a specific antigen, but does not substantially recognize or bind other molecules in a sample.
- an antibody or antigen binding domain that specifically binds to an antigen from one species may also bind to that antigen from one or more species. But, such cross-species reactivity does not itself alter the classification of an antibody or antigen binding domain as specific.
- an antibody or antigen binding domain that specifically binds to an antigen may also bind to different allelic forms of the antigen. However, such cross reactivity does not itself alter the classification of an antibody or antigen binding domain as specific.
- the terms “specific binding” or “specifically binding,” can be used in reference to the interaction of an antibody, antigen binding domain, a protein, or a peptide with a second chemical species, to mean that the interaction is dependent upon the presence of a particular structure (e.g., an antigenic determinant or epitope) on the chemical species; for example, an antibody or antigen binding domain recognizes and binds to a specific protein structure rather than to proteins generally. If an antibody is specific for epitope "A”, the presence of a molecule containing epitope A (or free, unlabeled A), in a reaction containing labeled "A” and the antibody, will reduce the amount of labeled A bound to the antibody.
- a particular structure e.g., an antigenic determinant or epitope
- an antibody or antigen binding domain recognizes and binds to a specific protein structure rather than to proteins generally.
- an “immune response” refers to the reaction of a subject to the presence of an antigen, which may include at least one of the following: making antibodies, developing immunity, developing hypersensitivity to the antigen, and developing tolerance.
- the term “enhance an immune response” as used herein implies that the reaction of a subject to the presence of an antigen is increased and/or amplified in the presence of a CAR-modified phagocytic cell of the disclosure as compared to the reaction of a subject to the presence of an antigen in the absence of a CAR-modified phagocytic cell of the disclosure.
- treat indicates that the severity of the subject's condition is reduced or at least partially improved or modified and that some alleviation, mitigation or decrease in at least one clinical symptom is achieved.
- terapéutica as used herein means a treatment and/or prophylaxis.
- a therapeutic effect is obtained by suppression, remission, or eradication of a disease state.
- an “effective” amount as used herein is an amount that provides a desired effect.
- a “therapeutically effective” amount as used herein is an amount that provides some improvement or benefit to the subject.
- a “therapeutically effective” amount is an amount that will provide some alleviation, mitigation, or decrease in at least one clinical symptom in the subject.
- the therapeutic effects need not be complete or curative, as long as some benefit is provided to the subject.
- moduleating mediating a detectable increase or decrease in the level of a response in a subject compared with the level of a response in the subject in the absence of a treatment or compound, and/or compared with the level of a response in an otherwise identical but untreated subject.
- the term encompasses perturbing and/or affecting a native signal or response thereby mediating a beneficial therapeutic response in a subject, preferably, a human.
- target site or “target sequence” refers to a genomic nucleic acid sequence that defines a portion of a nucleic acid to which a binding molecule may specifically bind under conditions sufficient for binding to occur.
- target is meant a cell, organ, or site within the body that is in need of treatment.
- detectable moiety includes any suitable detectable group, such as radiolabels (e.g. 35 S, 125 I, 131 I, etc.), enzyme labels (e.g, horseradish peroxidase, alkaline phosphatase, etc.), fluorescence labels (e.g., fluorescein, green fluorescent protein, etc.), etc., as are well known in the art and used in accordance with known techniques.
- detectable group such as radiolabels (e.g. 35 S, 125 I, 131 I, etc.), enzyme labels (e.g, horseradish peroxidase, alkaline phosphatase, etc.), fluorescence labels (e.g., fluorescein, green fluorescent protein, etc.), etc., as are well known in the art and used in accordance with known techniques.
- agent or “biological agent” or “therapeutic agent” as used herein, refers to a molecule that may be expressed, released, secreted or delivered to a target by the modified cell described herein
- the agent includes, but is not limited to, a nucleic acid, an antibiotic, an antiinflammatory agent, an antibody or antibody fragments thereof, a growth factor, a cytokine, an enzyme, a protein, a peptide, a fusion protein, a synthetic molecule, an organic molecule (e.g., a small molecule), a carbohydrate or the like, a lipid, a hormone, a microsome, a derivative or a variation thereof, and any combination thereof.
- the agent may bind any cell moiety, such as a receptor, an antigenic determinant, or other binding site present on a target or target cell. The agent may diffuse or be transported into the cell, where it may act intracellularly.
- “Mobilization” refers to the process of forced emigration of hematopoietic stem cells (HSPCs) from the bone marrow (BM) into the peripheral blood. Mobilization of HSPCs can occur in response to a wide variety of stimuli including strenuous physical exercise, myelosuppressive chemotherapy, polyanions, chemokines, and hematopoietic growth factors.
- expand refers to increasing in number, as in an increase in the number of stem cells or cells derived therefrom.
- the stem cells, or cells derived therefrom, that are expanded ex vivo increase in number relative to the number originally present in the culture (e.g., in precursor media).
- the stem phagocytic cells, or cells derived therefrom that are expanded ex vivo increase in number relative to other cell types in the culture.
- ex vivo refers to cells that have been removed from a living organism, (e.g., a human) and propagated outside the organism (e.g., in a culture dish, test tube, or bioreactor).
- the term “expand” can refer to a cell, or a population comprising a plurality of cells. When used to refer to a cell population, the term “expand” refers to an increase in the total number of cells within the population. Expansion of a cell population can occur when at least one individual cell within the population undergoes expansion. In some instances, more than one cell within the population undergoes expansion. In some instances, all cells within a population can expand.
- a specific subset of cells within a population can expand, wherein cells not within said subset do not expand. Expansion can be temporal. For instance, a cell, a population, or a subset within a population can expand for a period of time and then cease to expand. In some instances, one subset of cells within a population can undergo expansion and then cease, after which a second subset can undergo expansion.
- Disclosed herein are mixed populations of cells. In some instances, any of the cells within the population can undergo expansion. The mixed populations of cells disclosed herein can undergo expansion. In some embodiments, the mixed population of cells disclosed herein comprises at least one precursor cell and/or at least one myeloid-derived cell.
- the mixed population of cells disclosed herein comprises at least one precursor CAR cell and/or at least one myeloid-derived CAR cell. In some embodiments, the mixed population of cells disclosed herein comprises a plurality of precursor CAR cells and/or a plurality of myeloid-derived CAR cells.
- any subset or plurality of cells within the population can undergo expansion. In some instances, all cells within the population undergo expansion. In some instances, the precursor CAR cells undergo expansion. In some instances, the myeloid-derived CAR cells undergo expansion.
- a cell, or population thereof has expanded, the number of cells within the population has increased. During expansion, or when expanding, the number of cells within the population that is expanding is increasing.
- Cell or cell population expansion
- the precursor mediums disclosed herein are particularly effective at enhancing the expansion of stem cells over a defined period of time compared to expansion of stem cells in a medium that is not said precursor medium.
- the specific components within the disclosed precursor mediums are thought to enhance stem cell expansion.
- the precursor medium disclosed herein enhance stem cell expansion greater than a medium that does not comprise the precursor cytokine mixture of the precursor mediums disclosed herein.
- a population of cells that is undergoing expansion is also undergoing differentiation. In such instances, a first subset of cells within the population can be expanding, and a second subset of cells within a population can be differentiating.
- differentiate refers to a process by which young, immature (unspecialized) cells (e.g, stem cells) take on individual characteristics and reach their mature (specialized) form (e.g., myeloid cells) and function. During this process, cells can lose their developmental potential and gain specialized functions and phenotypes.
- the specialized cell type can be characterized by its physiological function and its role as part of a tissue or organ.
- the differentiation of a cell is typically associated with a change in at least one cell surface marker. In some instances, differentiation of a cell is associated with a change in multiple cell surface markers.
- an immature cell such as a stem cell
- a stem cell can either gain or lose, or both, specific cell surface markers.
- the presence and or absence of individual cell surface markers can be used to categorized cells and/or to identifying a cell that has undergone differentiation.
- differentiation is associated with a change in function and role of a cell, it is understood that the cell type and/or cell name will change.
- a stem cell can differentiate into a myeloid cell.
- the hematopoietic stem cells (HSCs) disclosed herein undergo differentiation.
- the HSCs can differentiation into a cell within the myeloid lineage (i.e., myeloid-lineage cell).
- a stem cell differentiates into a myeloid-lineage cell the cell may acquire specific cell surface markers associated with the myeloid-lineage.
- the cell surface markers that are expressed by a differentiated cell with be dependent upon which cell it differentiations into.
- the precursor cells disclosed herein can differentiate into a myeloid-lineage or myeloid-derived cell.
- cell differentiation is associated with an increase in the amount or number of surface markers on at least one cell (e.g., a precursor cell), wherein said surface markers are myeloid- associated markers.
- a “myeloid-associated marker” can be any surface marker that is known and available in the art for identifying and/or isolating a cell within the myeloid lineage.
- myeloid-associated markers can include: CDl lb, CD14, CD13, and CD15.
- differentiation of a precursor stem cell is associated with the increase in the presence of CD1 lb, CD14, CD13, and CD15 on the cell surface.
- differentiation of a precursor stem cell is associated with the increase in the levels of CDl lb, CD14, CD13, and CD15 on the cell surface.
- differentiation of a precursor stem cell is associated with the decrease in the presence of CD34 on the cell surface.
- differentiation of a precursor stem cell is associated with the decrease in the level of CD34 on the cell surface.
- the levels of more than one myeloid-associated marker can increase on the surface of a cell that is differentiating into a myeloid-lineage cell.
- the hematopoietic stem cells (HSCs) disclosed herein undergo differentiation.
- the HSCs can differentiation into a cell within the myeloid lineage (i.e., myeloid-lineage cell). Differentiation of a cell population can occur when at least one individual cell within the population undergoes differentiation. In some instances, more than one cell within the population undergoes differentiation.
- all cells within a population can differentiate.
- a specific subset of cells within a population can differentiate, wherein cells not within said subset do not differentiate.
- Differentiation can be temporal. For instance, a cell, a population, or a subset within a population can undergo differentiation for a period of time and then cease to differentiate. In some instances, one subset of cells within a population can undergo differentiation and then cease, after which a second subset can undergo differentiation.
- Disclosed herein are mixed populations of cells. In some instances, any of the cells within the population can undergo differentiation.
- the mixed populations of cells disclosed herein can undergo differentiation.
- the mixed population of cells disclosed herein comprises at least one precursor cell and/or at least one myeloid-derived cell.
- the mixed population of cells that undergoes differentiation comprises at least one precursor cell and/or at least one myeloid-derived cell. In some embodiments, the mixed population of cells that undergoes differentiation comprises at least one precursor CAR cell and/or at least one myeloid-derived CAR cell. In some embodiments, the mixed population of cells that undergoes differentiation comprises a plurality of precursor CAR cells and/or a plurality of myeloid-derived CAR cells.
- any number of cells within the population, and any subset or plurality of cells within the population can differentiate. In some instances, all cells, or most of the cells, within the population undergo differentiation. In such instances, a population can go from comprising mostly precursor cells to mostly myeloid-lineage cells.
- the precursor CAR cells undergo differentiation.
- the myeloid- derived CAR cells undergo differentiation.
- the number of precursor cells within the population has decreased, while the number of differentiated cells within the population has increased.
- the type of cells within the population that is differentiating can be changing.
- a population of cells that is undergoing differentiation is also expanding. In such instances, a first subset of cells within the population can be expanding, and a second subset of cells within a population can be differentiating.
- Myeloid medium or “myeloid media”, used interchangeably herein, refer to a culture medium that promotes mature myeloid lineage development of any of the cells disclosed herein.
- the myeloid medium disclosed herein comprise a myeloid cytokine mixture.
- the specific myeloid cytokine mixture(s) within the myeloid medium is thought to promote myeloid lineage differentiation.
- the myeloid medium disclosed herein enhance stem cell differentiation greater than a medium that is similar to the disclosed myeloid media but does not comprise the myeloid cytokine mixture.
- Myeloid lineage refers to the group of cells that are derived from a common myeloid progenitor (CMP) in the bone marrow.
- CMP myeloid progenitor
- the cells within the myeloid lineage which include monocytes, granulocytes, erythrocytes, and platelets, serve as a primary component of the innate immune system.
- myeloid cells The cells within the myeloid lineage are referred to, for the purposes of the present disclosure, as “myeloid cells.”
- the common myeloid progenitor cell, and thus all myeloid cells, are derived from hematopoietic stem cells (HSCs), which are multipotent cells that can give rise to all blood lineages, including the myeloid lineage, of an adult organism in a process referred to a hematopoiesis.
- HSCs hematopoietic stem cells
- cytokines and growth factors enhance the direction.
- Myeloid cells, as well as many aspects of their development from HSCs, and differentiation of cells within the myeloid lineage are readily known in the art (Weiskopf, Kipp et al.
- Precursor media or “precursor medium”, used interchangeably herein, refer to a culture medium that promotes expansion (i.e., an increase in the number of cells) of any of the cells disclosed herein.
- the precursor media disclosed herein comprises a precursor cytokine mixture.
- the specific precursor cytokine mixture(s) present within the precursor media is thought to promote an increase in the number of at least one cell that is contacted with, or maintained in, the precursor media.
- the precursor medium disclosed herein enhances stem cell expansion (e.g., expansion of a single stem cell, or a plurality of stem cells present within a cell population) greater than a medium that is similar to the disclosed precursor media but does not comprise the precursor cytokine mixture.
- the precursor media can increase the number of cells of any cell type disclosed herein.
- a ’’precursor cytokine mixture refers to a composition comprising at least one cytokine that is present within a precursor media disclosed herein. Any cytokine disclosed herein can be present within the precursor cytokine mixture at any concentration.
- a ’’myeloid cytokine mixture refers to a composition comprising at least one cytokine that is present within a myeloid media disclosed herein. Any cytokine disclosed herein can be present within the myeloid cytokine mixture at any concentration.
- the precursor cell refers to any cell that can differentiate into another cell type.
- the precursor cell is a stem cell. Any type of stem cell is envisaged as being used as the precursor cell, so long as it can be differentiated into a myeloid-lineage cell.
- the precursor cell is a bone-marrow derived stem cell.
- the precursor cell is a hematopoietic stem cell (HSC). Suitable precursor cells can be identified and isolated by specific cell surface markers.
- the precursor cell is a CD34+ cell.
- the precursor cell can be isolated from a subject, such as a mammalian subject by any suitable means available in the art.
- the precursor cell may be frozen following isolation and prior to use in the disclosed methods.
- the precursor cell may be a cryopreserved stem cell.
- the precursor cell may be necessary to thaw the cell prior to use in the disclosed methods.
- modified is meant a changed state or structure of a molecule or cell of the disclosure.
- Molecules may be modified in many ways, including chemically, structurally, and functionally.
- Cells may be modified through the introduction of nucleic acids.
- the term “decreased” or “decreasing” or “decrease” or “reduced” or “reducing” or “reduce” or “lower” or “loss” refers to a detectable (e.g., at least about 5%, 10%, 15%, 20%, 25%, 30%, 35%, 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 96%, 97%, 98%, 99%, or 100%) negative change in the parameter from a comparison control, e.g., an established normal or reference level of the parameter, or an established standard control. Accordingly, the terms “decreased”, “reduced”, and the like encompass both a partial reduction and a complete reduction compared to a control.
- the term “increased” or “increasing” or “increase” refers to a detectable (e.g., at least about 5%, 10%, 15%, 20%, 25%, 30%, 35%, 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 96%, 97%, 98%, 99%, 100%, 120%, 150%, 200%, 300%, 400%, 500%, or more) positive change in the parameter from a comparison control, e.g., an established normal or reference level of the parameter, or an established standard control. Accordingly, the terms “increased”, “increase”, and the like encompass both a partial reduction and a significant increase compared to a control.
- fold change indicates a measure describing how much a quantity changes between an original and a subsequent measurement.
- fold change is defined as the ratio between two quantities. For example, for quantities A and B, the fold change of B with respect to A is B/A. For example, a change from 30 to 60 is defined as a fold-change of 2.
- sequences that substantially correspond to its complementary sequence as including minor sequence variations, resulting from, e.g., sequencing errors, cloning errors, or other alterations resulting in base substitution, base deletion or base addition, provided that the frequency of such variations is less than 1 in 50 nucleotides, alternatively, less than 1 in 100 nucleotides, alternatively, less than 1 in 200 nucleotides, alternatively, less than 1 in 500 nucleotides, alternatively, less than 1 in 1000 nucleotides, alternatively, less than 1 in 5,000 nucleotides, alternatively, less than 1 in 10,000 nucleotides.
- isolated refers to at least partially separated from the natural environment e.g., from a cell.
- a nucleic acid or a peptide naturally present in a living animal is not “isolated,” but the same nucleic acid or peptide partially or completely separated from the coexisting materials of its natural state is "isolated.”
- An isolated nucleic acid or protein can exist in substantially purified form, or can exist in a non-native environment such as, for example, a host cell.
- heterologous nucleic acid sequence in reference to a nucleic acid sequence or amino acid sequence are intended to mean a sequence that is purely synthetic, that originates from a foreign species, or, if from the same species, is substantially modified from its native form in composition and/or genomic locus by deliberate human intervention.
- a heterologous nucleic acid sequence may not be naturally expressed within a cell or may have altered expression when compared to the corresponding wild type cell.
- a heterologous polynucleotide encoding a CAR described herein can be a nucleic acid sequence that is not naturally present in a phagocytic cell in which it is present.
- exogenous polynucleotide may be introduced into the cell in a stable or transient manner, so as to produce a ribonucleic acid (RNA) molecule and/or a polypeptide molecule. It should be noted that the exogenous polynucleotide may comprise a nucleic acid sequence which is identical or partially homologous to an endogenous nucleic acid sequence of the cell.
- endogenous in reference to a gene or nucleic acid sequence or protein is intended a gene or nucleic acid sequence or protein that is naturally comprised within or expressed by a cell.
- Endogenous genes can include genes that naturally occur in a cell (e.g., phagocytic cell), but that have been modified in the genome of the cell without insertion or replacement of a heterologous gene that is from another species or another location within the genome of the modified cell.
- “Homolog” or “homologous sequence” may refer to both orthologous and paralogous sequences.
- Paralogous sequence relates to gene-duplications within the genome of a species.
- Orthologous sequence relates to homologous genes in different organisms due to ancestral relationship.
- orthologs are evolutionary counterparts derived from a single ancestral gene in the last common ancestor of given two species and therefore have great likelihood of having the same function.
- the term “homolog” as used herein refers to functional homologs of genes.
- a functional homolog is a gene encoding a polypeptide that has sequence similarity to a polypeptide encoded by a reference gene, and the polypeptide encoded by the homolog carries out one or more of the biochemical or physiological function(s) of the polypeptide encoded by the reference gene.
- Homology can be determined using any homology comparison software computing a pairwise sequence alignment.
- "Homologous” can refer to the subunit sequence identity between two polymeric molecules, e.g., between two nucleic acid molecules, such as, two DNA molecules or two RNA molecules, or between two polypeptide molecules. When a subunit position in both of the two molecules is occupied by the same monomeric subunit; e.g., if a position in each of two DNA molecules is occupied by adenine, then they are homologous at that position.
- the homology between two sequences is a direct function of the number of matching or homologous positions; e.g., if half (e.g., five positions in a polymer ten subunits in length) of the positions in two sequences are homologous, the two sequences are 50% homologous; if 90% of the positions (e.g., 9 of 10), are matched or homologous, the two sequences are 90% homologous.
- the term “homology” or “homologous” refers to identity of two or more nucleic acid sequences; or identity of two or more amino acid sequences; or the identity of an amino acid sequence to one or more nucleic acid sequence.
- the homology is a global homology, e.g., a homology over the entire amino acid or nucleic acid sequences of the invention and not over portions thereof.
- the degree of homology or identity between two or more sequences can be determined using various known sequence comparison tools which are described in WO2014/102774.
- "homologous" as used herein refers to a sequence that has about 50% sequence identity. More preferably, the homologous sequence has about 75% sequence identity, even more preferably, has at least about 80%, 85%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99% sequence identity.
- sequence identity As used herein, “sequence identity,” “identity,” “percent identity,” “percentage similarity,” “sequence similarity” and the like refer to a measure of the degree of similarity of two sequences based upon an alignment of the sequences that maximizes similarity between aligned amino acid residues or nucleotides, and which is a function of the number of identical or similar residues or nucleotides, the number of total residues or nucleotides, and the presence and length of gaps in the sequence alignment.
- a variety of algorithms and computer programs are available for determining sequence similarity using standard parameters.
- sequence similarity is measured using the BLAST®p program for amino acid sequences and the BLAST®n program for nucleic acid sequences, both of which are available through the National Center for Biotechnology Information, and are described in, for example, Altschul et al. (1990), J. Mol. Biol. 215:403-410; Gish and States (1993), Nature Genet. 3:266-272; Madden et al. (1996), Meth. Enzymol.266: 131-141; Altschul et al. (1997), Nucleic Acids Res. 25:3389-3402); Zhang et al. (2000), J. Comput. Biol. 7(l-2):203-14.
- sequence similarity or “similarity”.
- Means for making this adjustment are well-known to those of skill in the art. Typically this involves scoring a conservative substitution as a partial rather than a full mismatch, thereby increasing the percentage sequence identity. Thus, for example, where an identical amino acid is given a score of 1 and a non-conservative substitution is given a score of zero, a conservative substitution is given a score between zero and 1.
- the term “method” refers to manners, means, techniques and procedures for accomplishing a given task including, but not limited to, those manners, means, techniques and procedures either known to, or readily developed from known manners, means, techniques and procedures by practitioners of the chemical, pharmacological, biological, biochemical and medical arts.
- the methods can be used to generate myeloid-derived CAR cells from stem cells.
- the stem cells are isolated stem cells.
- the stem cells can be isolated from a subject.
- the subject can be a mammalian subject, such as a human.
- the stem cell can be a hematopoietic stem cell and/or a CD34+ cell.
- the stem cell can be isolated from the bone-marrow of a subject.
- the methods and compositions disclosed herein provide an effective and efficient process for generating a myeloid lineage cell (i.e., myeloid-derived cells), and populations comprising myeloid-derived cells, ex vivo.
- the methods of generating myeloid-derived cells ex vivo can be performed in a shorter amount of time, and can generate a cleaner (i.e., higher percentage of myeloid cells compared to non-myeloid cells) myeloid-derived cell population compared to standard methods known in the art.
- One aspect that contributes to the enhanced efficiency of the disclosed method is the superior ability of the precursor media disclosed herein to increase the number of precursor cells to generate a cell population in a shorter amount of time compared to known expansion medias.
- the precursor cytokine mixture present within the disclosed precursor media are thought to promote faster expansion of precursor cells compared to similar expansion media not comprising the disclosed precursor cytokine mixtures.
- Another aspect contributing to the enhanced efficiency of the disclosed method to generate a myeloid- derived cell or myeloid-derived CAR cell population is the superior technical capability of the precursor media to promote expansion and differentiation of precursor cells, in particular hematopoietic stem cells and/or CD34+ cells.
- the precursor media disclosed herein can promote differentiation of precursor cells simultaneously. This combined process of expansion and differentiation in a precursor media significantly reduces the amount of time required to generate a myeloid-derived cell population and/or a myeloid-derived CAR cell population.
- the precursor media disclosed herein are also particularly effective at promoting efficient differentiation of precursor cells into myeloid-derived cells.
- the precursor media can generate a cell population comprising a plurality of myeloid-derived cells, wherein said population comprises a higher percentage of myeloid-derived cells that have been sufficiently differentiated compared to myeloid-derived cell populations that are generated from standard methods. For example, standard methods may generate a cell population with a low, or sub-optimal, percentage of myeloid-derived cells.
- Standard methods for generating a myeloid-derived cell, or population comprising the same generally require a step wise process, which involves expansion of a cell population as a first step, followed by differentiation of the cells within the population into the desired myeloid cell population. This step wise process involving separate steps and medias can be time-consuming and require a significant amount of manufacturing and financial resources.
- a CAR can be introduced into a stem cell prior to expansion and differentiation of said stem cell into a myeloid lineage cell using the compositions and methods disclosed herein.
- the methods of the present disclosure provide conditions that allow for enhanced stability of transduced genes, such as a CAR, when introduced into stem cells prior to the expansion and/or differentiation process, resulting in a myeloid-derived CAR cell population.
- the compositions and methods disclosed promote the expansion and differentiation of stem cells into myeloid-derived cells in a shorter time compared to known expansion and differentiation methods.
- the precursor media disclosed herein promote expansion of precursor cells, such as stem cells, into myeloid cells in a shorter time compared to known expansion mediums.
- the precursor media of the present disclosure allow for the efficient and effective production of precursor cell populations in a shorter period of time compared to known media.
- the precursor media disclosed herein in addition to promoting expansion of precursor cell and precursor CAR cell populations, also promote the differentiation of said precursor cells and precursor CAR cells into myeloid lineage cells.
- the precursor media of the present disclosure are effective at promoting expansion and differentiation of precursor cells into myeloid-derived cells.
- the precursor media can also efficiently promote the differentiation of precursor CAR cells into myeloid-derived CAR cells.
- the precursor media promotes the expansion and differentiation of a precursor cell and/or a precursor CAR cell at the same time.
- a precursor cell when incubated in a precursor media can increase in number to generate a cell population comprising a plurality of precursor cells.
- the precursor cell can be a precursor CAR cell.
- said precursor cells within the cell population can be undergoing differentiation in the precursor media, thereby generating a cell population, wherein said cell population is a mixed cell population comprising a plurality of precursor cells and/or a plurality of myeloid-derived cells.
- the mixed cell population comprises a plurality of precursor cells and a plurality of myeloid-derived cells.
- the ability of the precursor media to both promote expansion of a precursor cell and/or a population comprising a plurality of precursor cells, as well as differentiation of said precursor cell and/or said population provides for enhanced methods over the art for generating a population of myeloid-derived cells, such as myeloid-derived CAR cells, in a shorter amount of time.
- the methods and compositions disclosed herein provide for more efficient and effective means of generating a population of myeloid- derived cells and/or myeloid-derived CAR cells, wherein said population comprises a higher percentage of myeloid cells within the population.
- the cell populations generated by the methods disclosed herein comprise a higher percentage of myeloid-derived cells compared to cell populations generated by known methods.
- a differentiation media can increase the ratio of myeloid-derived cells to precursor cells within a cell population.
- methods involving use of a myeloid media to differentiate cells into a myeloid-derived cell are particularly effective at promoting the differentiation of a precursor cell into a myeloid lineage cell.
- the myeloid media disclosed herein can increase the ratio of myeloid-derived cells to precursor cells within a population in a short amount of time compared to standard differentiation medias, saving time and costs.
- a mixed population comprising a plurality of precursor cells and/or a plurality of myeloid-derived cells can be incubated in a myeloid media to increase the ratio of myeloid-derived cells to precursor cells.
- the mixed population was generated by incubating a precursor cell, or a population comprising a plurality of precursor cells in a precursor media, whereby said mixed population is subsequently incubated in a myeloid media to further promote an increase in myeloid-derived cell number within the mixed population.
- the methods disclosed herein involving contacting a precursor cell, such as a precursor CAR cell, with a precursor media and, optionally, a myeloid media, provide for a shorter overall process for generating a myeloid-derived cell population compared to a similar method involving a media that is generally similar to the precursor media disclosed herein, but does not comprise the precursor cytokine mixture of the presently disclosed precursor media, and as such, is not capable of expanding and differentiation a precursor cell, or population comprising a precursor cell, in the same shortened time interval as the disclosed methods.
- the methods disclosed herein provide easier, stable engineering of myeloid-derived cells compared to standard, known methods.
- the transduced genes e.g., a polynucleotide encoding a CAR
- the methods and compositions disclosed herein provide a shorter process for generating a stable myeloid-derived CAR cell.
- the methods and compositions disclosed herein can be used to generate a population of myeloid-derived cells.
- the methods can generate a myeloid-derived CAR cell, or a population comprising a plurality of myeloid-derived CAR cells.
- the compositions and methods can generate a cell population comprising a high percentage of myeloid-derived cells.
- the methods and compositions can be used to generate a cell population wherein at least 70%, at least 71%, at least 72%, at least 73%, at least 74%, at least 75%, at least 76%, at least 77%, at least 78%, at least 79%, at least 80%, at least 81%, at least 82%, at least 83%, or greater than 84% at least 85%, at least 86%, at least 87%, at least 88%, at least 89%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or greater than 99% of the cells within the population are positive for a myeloid cell surface marker (e.g., CD14+, CD13+, CD15+, CD1 lb+).
- a myeloid cell surface marker e.g., CD14+, CD13+, CD15+, CD1 lb+.
- the methods and compositions can be used to generate a cell population wherein at least 70%, at least 71%, at least 72%, at least 73%, at least 74%, at least 75%, at least 76%, at least 77%, at least 78%, at least 79%, at least 80%, at least 81%, at least 82%, at least 83%, or greater than 84% at least 85%, at least 86%, at least 87%, at least 88%, at least 89%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or greater than 99% of the cells within the population are positive for more than one myeloid cell surface marker (e.g., CD14+, CD13+, CD15+, CD1 lb+).
- myeloid cell surface marker e.g., CD14+, CD13+, CD15+, CD1 lb+.
- the methods and compositions can be used to generate a cell population wherein at least 70%, at least 71%, at least 72%, at least 73%, at least 74%, at least 75%, at least 76%, at least 77%, at least 78%, at least 79%, at least 80%, at least 81%, at least 82%, at least 83%, or greater than 84% at least 85%, at least 86%, at least 87%, at least 88%, at least 89%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or greater than 99% of the cells within the population are positive for two myeloid cell surface marker (e.g., CD14+, CD13+, CD15+, CD1 lb+).
- two myeloid cell surface marker e.g., CD14+, CD13+, CD15+, CD1 lb+.
- the methods and compositions can be used to generate a cell population wherein at least 70%, at least 71%, at least 72%, at least 73%, at least 74%, at least 75%, at least 76%, at least 77%, at least 78%, at least 79%, at least 80%, at least 81%, at least 82%, at least 83%, or greater than 84% at least 85%, at least 86%, at least 87%, at least 88%, at least 89%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or greater than 99% of the cells within the population are positive for three myeloid cell surface marker (e.g., CD14+, CD13+, CD15+, CD1 lb+).
- myeloid cell surface marker e.g., CD14+, CD13+, CD15+, CD1 lb+.
- the methods and compositions can be used to generate a cell population wherein at least 70%, at least 71%, at least 72%, at least 73%, at least 74%, at least 75%, at least 76%, at least 77%, at least 78%, at least 79%, at least 80%, at least 81%, at least 82%, at least 83%, or greater than 84% at least 85%, at least 86%, at least 87%, at least 88%, at least 89%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or greater than 99% of the cells within the population are positive for four myeloid cell surface marker (e.g., CD14+, CD13+, CD15+, CDl lb+).
- myeloid cell surface marker e.g., CD14+, CD13+, CD15+, CDl lb+.
- the methods disclosed herein are particularly effective at generating cell populations comprising a plurality of myeloid-derived cells, wherein said population comprises a high percentage of myeloid-derived cells.
- the myeloid-derived cells can be identified by expression of at least one myeloid-associated marker.
- the cell populations generated by the compositions and methods comprise a low percentage of non-myeloid cells (e.g., lymphocytes).
- the cell populations generated by the compositions and methods comprise a low percentage of lymphocytes.
- lymphocytes For instance, less than 40%, 39%, 38%, 37%, 36%, 35%, 34%, 33%, 32%, 31%, 30%, 29%, 28%, 27%, 26%, 25%, 24%, 23%, 22%, 21%, 20%, 19%, 18%, 17%, 16%, 15%, 14%, 13%, 12%, 11%, 10%, 9%, 8%, 7%, 6%, 5%, 4%, 3%, 2%, 1%, or less than 1% of the cells within the disclosed cell populations generated by the methods of the present application are lymphocytes.
- the methods and compositions can generate myeloid-derived cell populations comprising a higher percentage of myeloid-derived cells, and a lower percentage of lymphocytes compared to standard methods and compositions known in the art.
- the disclosed methods can generate said populations in a shorter amount of time.
- the methods and compositions of the present disclosure provide for significant time and financial savings in the overall manufacturing process of myeloid-derived cells.
- the myeloid-derived cells produced by the disclosed methods can be myeloid- derived CAR cells. Any myeloid lineage cell comprising a CAR is envisaged as being produced by the methods disclosed herein.
- the methods provide for a significantly more effective and efficient manufacturing process for monocyte and macrophage cells expressing a CAR (i.e., CAR-M cells).
- the methods disclosed herein can generate a myeloid progenitor cell comprising a CAR. Such efficiency is envisaged for any myeloid cells produced using the methods and compositions disclosed herein.
- the stem cells have been genetically engineered with a chimeric antigen receptor (CAR), which is stably maintained in the cell throughout the expansion and differentiation process to generate a population of myeloid-derived CAR cells.
- CAR chimeric antigen receptor
- media and cytokine mixtures that are particularly effective at promoting the expansion and/or differentiation of stem cells into myeloid-derived CAR cells.
- the methods disclosed herein involve contacting isolated stem cells with the media disclosed herein to promote the expansion and differentiation of stem cells, and populations comprising the same. Over the course of expansion and differentiation, stem cells will begin to upregulate myeloid markers as they differentiate into myeloid- derived cells.
- the media disclosed herein can effectively promote the expansion and the differentiation of stem cells.
- the stem cells within the media are increasing in number through cell division while concurrently differentiating into myeloid cells through the upregulation of myeloid markers.
- Cell populations comprising such cells are also disclosed.
- the present disclosure provides cell populations comprising stem cells engineered with a CAR and myeloid-derived CAR cells derived therefrom.
- the present disclosure provides genetically modified myeloid cells and any precursor thereof expressing a chimeric antigen receptor (CAR), and populations comprising the same.
- the genetically modified myeloid CAR cells can be generated from any precursor cell (e.g., a stem cell) according to the methods disclosed herein.
- a nucleic acid molecule encoding a CAR can be introduced into a precursor cell, such as a hematopoietic stem cell, whereby a genetically modified stem cell is generated, which can be further differentiated into a myeloid lineage cell.
- a precursor cell such as a hematopoietic stem cell
- a genetically modified stem cell is generated, which can be further differentiated into a myeloid lineage cell.
- myeloid lineage cell As the cells of the present disclosure are differentiated toward the myeloid lineage (and away from the lymphoid lineage) from a pluripotent stem cell, any cell within the myeloid lineage is envisaged within the scope of the present disclosure as being produced by the methods disclosed herein.
- the present disclosure relates to methods of manufacturing genetically modified myeloid-derived CAR cells, such as a CAR myeloid progenitor cell or CAR-M cell, from hematopoietic stem cells (HSCs).
- HSCs hematopoietic stem cells
- stem cells comprising a CAR (i.e., CAR stem cells) as well as myeloid-derived CAR cells are also disclosed, as well as populations comprising the same.
- the HSCs that are used in the disclosed methods can be isolated from a subject.
- the isolated HSCs are first genetically modified with a CAR and then expanded and differentiated to generate a CAR-M cell or myeloid-derived lineage precursor cell thereof.
- the present disclosure provides media compositions within which the HSCs can be expanded and differentiated.
- the media disclosed herein comprise cytokine mixtures that enable superior and improved expansion and differentiation of stem cells into myeloid cells.
- the expansion and differentiation of the HSCs can occur within a single media (e.g., a precursor media), which contributes to a dramatically improved expansion and differentiation of the stem cells (e.g., hematopoietic stem cells) toward a myeloid lineage cell, for example, a myeloid progenitor cells or differentiated cell thereof.
- the methods disclosed herein, wherein a HSC is genetically modified with a CAR, prior to expansion and differentiation into a myeloid lineage cell provide a more stable CAR cell that can be differentiated into a myeloid lineage cell.
- any myeloid lineage cell downstream of a HSC is envisaged within the genetically modified myeloid-derived CAR cells of the present disclosure.
- a myeloid precursor cell such as a myeloid progenitor cell, granulo- monocyte progenitor cell, monocyte-dendritic cell progenitor/monoblasts, promonocyte, or myeloblast, in addition to a monocyte, macrophage, dendritic cell and/or neutrophil.
- the genetically modified myeloid-derived CAR cells disclosed herein, when the cell is a precursor cell maintain the ability to differentiate into a mature cell both in vitro and in vivo.
- Compositions and methods, related to the CAR-modified myeloid-derived cells and use of the same for treatment in a subject in need thereof are also provided.
- HSCs Hematopoietic Stem Cells
- the present disclosure provides a method of manufacturing a myeloid cell (i.e., myeloid- derived cell) from a precursor cell, such as a stem cell.
- a method of manufacturing a myeloid-derived cell comprising the step of contacting a precursor cell with a precursor media comprising a precursor cytokine mixture, whereby the number of precursor cell(s) increases to generate a cell population comprising a plurality of precursor cells.
- the precursor cell is a stem cell.
- cell populations comprising a plurality of precursor cells.
- the myeloid-derived cells disclosed herein do not require a chimeric antigen receptor (CAR).
- CAR chimeric antigen receptor
- the methods and compositions disclosed herein can effectively produce a myeloid-derived cell, and a cell population comprising a plurality of myeloid-derived cells, from a precursor cell that does not comprise a CAR.
- a hematopoietic stem cell can be contacted with, and maintained in, (i.e., cultured in) a precursor medium and, optionally, a myeloid medium disclosed herein.
- a method for manufacturing a myeloid-derived cell comprising the step of contacting a precursor cell with a precursor media comprising a precursor cytokine mixture, whereby the number of precursor cell(s) increases to generate a cell population comprising a plurality of precursor cells, wherein the method further comprises the step of contacting said cell population comprising a plurality of precursor cells with a myeloid media comprising a myeloid cytokine mixture, whereby said precursor cell(s) differentiates into a myeloid-lineage cell to generate a myeloid-derived cell and/or a mixed cell population comprising a plurality of myeloid-derived cells and/or a plurality of precursor cells.
- the precursor cell can be a stem cell and/or a CD34+ cell.
- the hematopoietic stem cells can be expanded (i.e., to generate a cell population comprising a plurality of HSCs) and differentiated (i.e., to generate a myeloid-derived cell and/or a mixed population comprising a plurality of myeloid-derived cells and/or a plurality of HSCs) in the precursor media disclosed herein.
- the HSCs in the precursor media are within a cell population, wherein a plurality of HSCs within the population are undergoing expansion, and/or wherein a plurality of HSCs within the population are undergoing differentiation.
- an HSC that is maintained in a precursor media will generate a cell population comprising a plurality of HSCs and a plurality of myeloid-derived cells.
- the hematopoietic stem cell is maintained in a precursor media, whereby the number of HSCs increases to generate a cell population comprising a plurality of HSCs, and whereby upon contact of said HSCs with said precursor media, said HSCs differentiates into a myeloid-lineage cell(s) to generate a myeloid-derived cell(s) and/or a mixed cell population comprising a plurality of myeloid-derived cells and a plurality of HSC cells.
- the method can further comprise the step of contacting said mixed cell population with a myeloid media comprising a myeloid cytokine mixture, whereby upon contact with said myeloid media: a. the ratio of said myeloid-derived cells to HSCs in said mixed cell population is increased.
- the precursor media can be removed prior to addition of the myeloid media.
- the methods and compositions disclosed herein can also effectively generate a myeloid- derived CAR cell from a precursor CAR cell. Any of the methods and/or compositions disclosed herein are envisaged as to be as effective for expansion and differentiation of an HSC CAR cell as with an HSC. The presence of a CAR in the HSC are not envisaged as impacting the effectiveness or the efficacy of the methods disclosed herein to expand and differentiate a precursor cell and/or a precursor CAR cell.
- the present disclosure provides a method of manufacturing a genetically modified myeloid cell (i.e., myeloid-derived cell) comprising a chimeric antigen receptor (CAR) (i.e., myeloid-derived CAR cell), wherein the genetically modified CAR cell has been generated from a precursor according to the methods described herein.
- the precursor cells can comprise a bone marrow-derived cell or a stem cell.
- the bone-marrow derived cell is a stem cell.
- the precursor cell is a stem cell.
- One type of stem cell that can be used is a hematopoietic stem cell (HSC).
- HSC hematopoietic stem cell
- the precursor cell is a hematopoietic stem cell (HSC).
- the methods and compositions disclosed herein are useful for expanding hematopoietic stem cells.
- the hematopoietic stem cells have been engineered with a CAR prior to expansion.
- the methods and compositions disclosed herein are also particularly useful for differentiating an HSC, or a population of cells comprising a plurality of HSCs, into a myeloid lineage cell.
- a method for manufacturing a myeloid-derived CAR cell comprising the step of contacting a precursor CAR cell with a precursor media comprising a precursor cytokine mixture, whereby the number of precursor CAR cell(s) increases to generate a cell population comprising a plurality of precursor CAR cells, wherein the method further comprises the step of contacting said cell population comprising a plurality of precursor CAR cells with a myeloid media comprising a myeloid cytokine mixture, whereby said precursor CAR cell(s) differentiates into a myeloid-lineage cell to generate a myeloid-derived CAR cell and/or a mixed cell population comprising a plurality of myeloid-derived CAR cells and/or a plurality of precursor CAR cells.
- the precursor CAR cell can be a stem cell and/or a CD34+ cell.
- the hematopoietic stem cells (HSCs) comprising a CAR can be expanded (i.e., to generate a cell population comprising a plurality of HSCs comprising a CAR) and differentiated (i.e., to generate a myeloid- derived CAR cell and/or a mixed population comprising a plurality of myeloid-derived CAR cells and/or a plurality of HSCs comprising a CAR) in the precursor media disclosed herein.
- HSCs hematopoietic stem cells
- the HSCs comprising a CAR in the precursor media are within a cell population, wherein a plurality of HSCs comprising a CAR within the population are undergoing expansion, and/or wherein a plurality of HSCs comprising a CAR within the population are undergoing differentiation.
- an HSC comprising a CAR that is maintained in a precursor media will generate a cell population comprising a plurality of HSCs comprising a CAR and a plurality of myeloid-derived CAR cells.
- the hematopoietic stem cell (HSC) comprising a CAR is maintained in a precursor media, whereby the number of HSCs comprising a CAR increases to generate a cell population comprising a plurality of HSCs comprising a CAR, and whereby upon contact of said HSCs with said precursor media, said HSCs differentiates into a myeloid-lineage cell(s) to generate a myeloid-derived CAR cell(s) and/or a mixed cell population comprising a plurality of myeloid-derived CAR cells and a plurality of HSC cells comprising a CAR.
- HSC hematopoietic stem cell
- the method can further comprise the step of contacting said mixed cell population with a myeloid media comprising a myeloid cytokine mixture, whereby upon contact with said myeloid media: a. the ratio of said myeloid-derived CAR cells to HSCs comprising a CAR in said mixed cell population is increased.
- the precursor media can be removed prior to addition of the myeloid media.
- the HSCs can be differentiated into any myeloid lineage cell. Exemplary myeloid cells into which the HSCs can be differentiated using the disclosed methods and compositions are described herein.
- the myeloid-derived CAR cells that can be generated from the disclosed methods can be used in the treatment of a disease or disorder, such as cancer, or in the manufacture of a medicament for use in treating a disease or a disorder. Any treatment, disease or disorder disclosed herein is envisaged.
- the HSCs disclosed herein can be isolated from a subject, expanded, and differentiated into myeloid-derived CAR cell, or a population of myeloid-derived CAR cells, whereby said myeloid-derived CAR cell is administered to said subject for treatment.
- the myeloid-derived CAR cells disclosed herein are administered to the same subject from which the HSCs used to generated said myeloid-derived CAR cells were isolated. That is, in some embodiments, the HSCs disclosed herein can be used to generate an autologous myeloid-derived cell. In some embodiments, said HSCs are engineered with a CAR prior to generating an autologous myeloid cell, generating a myeloid-derived CAR cell. Accordingly, the methods and compositions disclosed herein are particularly effective at generating myeloid-derived CAR cells from HSCs ex vivo. Any myeloid cell disclosed herein can be generated from the isolated HSCs disclosed herein ex vivo.
- HSCs Hematopoietic stem cells
- HSCs are multipotent precursors that have a unique ability to self-renew. HSCs produce hematopoietic progenitor cells that differentiate into every type of mature blood cell within a well-defined hierarchy (Bonnet, 2002; McCulloch and Till, 2005). HSCs can be found in various tissue, including bone marrow, mobilized peripheral blood, peripheral blood, and umbilical cord blood. HSCs and other blood cell progenitors can be isolated from fetal and embryonic tissues. Specifically, umbilical cord blood (UCB) and placenta are rich sources of HSCs (Abdulrazzak, Hassan et al.
- UMB umbilical cord blood
- placenta are rich sources of HSCs (Abdulrazzak, Hassan et al.
- Somatic cells can also be a source of HSCs, by conversion of these cells into induced pluripotent stem cells (IPSCs) (Vo, Linda T, and George Q Daley. “De novo generation of HSCs from somatic and pluripotent stem cell sources.” Blood vol. 125,17 (2015): 2641-8).
- ISCs induced pluripotent stem cells
- myeloid cells include, but are not limited to, monocytes, macrophages, neutrophils, basophils, eosinophils, erythrocytes, and megakaryocytes to platelets.
- lymphoid cells include T cells, B cells, natural killer cells, and innate lymphoid cells.
- CD34 is a type I transmembrane glycophosphoprotein predominantly regarded as a marker of hematopoietic stem cells (HSC) and hematopoietic progenitor cells (Civin et al., 1996b; Shizuru et al., 2005; Shpall et al., 1994). CD34 is expressed on -0.2-3% of the nucleated cells in cord blood, bone marrow and mobilized peripheral blood (Civin et al., 1984; Krause et al., 1996; Sutherland et al., 1996). CD34 has commonly been used to identify and select for populations of HSCs.
- CD34 expression is evaluated to ensure rapid engraftment in BM transplants; CD34 can also be used as a selective marker in cell sorting to enrich a population of immature hematopoietic cells (Berardi, A C et al. “Functional isolation and characterization of human hematopoietic stem cells.” Science (New York, N.Y.) vol. 267,5194 (1995): 104-8; Berenson, R J et al. “Engraftment after infusion of CD34+ marrow cells in patients with breast cancer or neuroblastoma.” Blood vol. 77,8 (1991): 1717-22.).
- the successful culture and maintenance of hematopoietic stem cells in an undifferentiated state in vitro requires additional considerations to ensure that cells maintain their key characteristics of self-renewal and pluripotency.
- precise media compositions can be required to properly balance aspects of HSC physiology and control self-renewal, expansion and lineage commitment (e.g., differentiation) in vitro.
- culturing HSCs requires a highly controlled environment where slight differences in media composition (e.g., basal media, added proteins) and environmental stimuli (e.g., temperature, time) can impact the expansion and differentiation of HSCs.
- cytokines are large family of extracellular ligands that stimulate hematopoietic cells to differentiate into eight principle types of blood cells. Numerous cytokines are involved in the regulation of hematopoiesis within a complex network of positive and negative regulators. Some cytokines have very narrow lineage specificities of their actions, while many others have rather broad and overlapping specificity ranges (Metcalf D. 2008. Hematopoietic cytokines. 111(2):485-491).
- cytokines seem to be more involved in processes involving the stimulation or regulation of hematopoietic cells. These include, for example: granulocyte-macrophage colony stimulating factor (GM-CSF), granulocyte-colony stimulating factor (G-CSF), macrophage-colony stimulating factor (M-CSF), interleukins, EPO and TPO.
- GM-CSF granulocyte-macrophage colony stimulating factor
- G-CSF granulocyte-colony stimulating factor
- M-CSF macrophage-colony stimulating factor
- interleukins EPO and TPO.
- EPO granulocyte-macrophage colony stimulating factor
- SCF stem cell factor
- FL flt-3/flk-2 ligand
- LIF leukemia inhibitory factor
- cytokines or ligands such as jagged-1, transforming growth factor-P (TGF-P) and tumor necrosis factor-a (TNF-a) also play significant roles in modulating hematopoiesis.
- TGF-P transforming growth factor-P
- TNF-a tumor necrosis factor-a
- a cytokine would be understood to be a type of protein that is made by certain immune and non-immune cells and has an effect on the immune system. Some cytokines stimulate the immune system and others slow it down. They can also be made in the laboratory and used to help the body fight cancer, infections, and other diseases.
- Nonlimiting, examples of cytokines include: interleukins, interferons, and colony-stimulating factors (i.e., a substance that stimulates the production of blood cells, including, for example, granulocyte colony-stimulating factor (G-CSF), granulocyte-macrophage colony-stimulating factor (GM-CSF), and promegapoietin).
- G-CSF granulocyte colony-stimulating factor
- GM-CSF granulocyte-macrophage colony-stimulating factor
- promegapoietin promegapoietin
- HSCs respond to multiple cytokines and that the fate of an HSC - self renewal, apoptosis, mobilization from the niche, formation of differentiated progeny cells - depends on these cytokines, adhesion proteins, and other signals produced by stromal cells and likely other cells in the body (Zhang, Cheng C, and Harvey F Lodish. “Cytokines regulating hematopoietic stem cell function.” Current opinion in hematology vol. 15,4 (2008): 307-11).
- cytokines within a culture media e.g., expansion and/or differentiation media
- a culture media e.g., expansion and/or differentiation media
- stem cell factor Journal of leukocyte biology vol. 58,1 (1995): 14-22; Ding, Lei et al. “Endothelial and perivascular cells maintain haematopoietic stem cells.” Nature vol. 481,7382 457-62. 25 Jan. 2012).
- hematopoietic stem cells can be produced through in vitro stem cell expansion, which is a long-term culture of HSCs with very limited or reduced amounts of cell exhaustion or differentiation of the cells. Cell exhaustion and differentiation can commonly occur during culture. Appropriate culture conditions, such as an expansion media containing the necessary supplements are required to control HSC proliferation, differentiation and viability. A variety of media and culturing conditions has been used to successfully expand HSCs. These aspects of HSC culturing and expansion are widely available and known within the art. Many commonly used modified basal media and corresponding recipes are well establish and are commercially available. Hematopoietic stem cells differentiate into various lineages.
- HSCs differentiation process is regulated by intracellular signaling pathways, which can be tightly regulated and controlled by growth factors and cytokines.
- cytokines and growth factors can impact differentiation efficacy (Yadav, Pooja et al. “Hematopoietic Stem Cells Culture, Expansion and Differentiation: An Insight into Variable and Available Media.” International journal of stem cells vol. 13,3 (2020): 326-334).
- compositions and methods for expanding and differentiating hematopoietic stem cells can be isolated from a subject, such as a human subject. HSCs isolated from a human subject can be identified and quantified according to any methods known in the art prior to expanding the HSCs. In the methods of generating a genetically modified cell disclosed herein, the HSCs can be expanded and differentiated after the cells are contacted with an agent comprising a nucleic acid molecule comprising a polynucleotide sequence encoding the CAR.
- the HSCs that are expanded and differentiated are HSCs, or a derivative therefrom, that comprise a nucleic acid molecule comprising a polynucleotide sequence.
- the polynucleotide sequence encodes a CAR.
- the nucleic acid molecule comprising the polynucleotide sequence is maintained within the HSCs and population of HSCs during and throughout expansion and differentiation. It is not anticipated that the nucleic acid molecule comprising the polynucleotide sequence is removed or lost from the HSCs, or the population thereof, or that the nucleic acid molecule is otherwise reduced within the population.
- the number of HSCs increases in number, whereby a population of HSCs is generated.
- the methods disclosed herein comprise a method of manufacturing a genetically modified cell comprising a chimeric antigen receptor (CAR), the method comprising: (a) contacting a precursor with an agent comprising a nucleic acid molecule comprising a polynucleotide sequence encoding the CAR to generate a precursor-CAR cell; and (b) inducing differentiation of said precursor-CAR cell into a phagocytic cell to generate a genetically modified phagocytic cell.
- the methods can further comprise: the step of (c) expanding the genetically modified phagocytic cell to generate a cell population comprising a plurality of genetically modified phagocytic cells.
- the cell population of HSCs can be expanded and/or differentiated for any number of days required to achieve the number of HSCs desired.
- the HSCs can be expanded and/or differentiated for at least one day, at least 2 days, at least 3 days, at least 4 days, at least 5 days, at least 6 days, at least 7 days, at least 8 days, at least 9 days, at least 10 days, or more than 10 days.
- the expansion and/or differentiation step can last for 1 day, 2 days, or 3 days. In some instances, the expansion step and/or differentiation step can last for 3 days.
- the expansion step may be maintained for as long as necessary to achieve the desired number of HSCs within the population.
- the desired number of cells would be understood and determined by the skilled artisan at the time of performing the method.
- the desired number of cells may be determined by the initial number of starting cells. Specifically, the desired number of cells may be a multiple of the initial number of starting cells.
- a skilled artisan would understand how to calculate the number of starting cells, as well as the number of current cells at any point throughout the expansion step and would be able to determine the amount of time to maintain the expansion phase. Any method of calculating the number of cells within the population that is available to one of skill in the art is envisaged within the present disclosure.
- the number of cells is decided based on a desired concentration.
- the expansion step can be maintained until the number of HSCs within a population has increased a specific amount relative to the starting number of cells.
- the HSCs are expanded to a number 100-fold, 200-fold, 300-fold, 400-fold, 500-fold, or greater than 500-fold of the initial starting HSC number.
- the HSCs are expanded to a number 100-fold the initial starting HSC number.
- the HSCs are expanded to a number 200-fold the initial starting HSC number.
- the HSCs are expanded to a number 300-fold the initial starting HSC number.
- the HSCs are expanded to a number 400-fold the initial starting HSC number. In some instances, the HSCs are expanded to a number 500-fold the initial starting HSC number. In some instances, wherein the HSCs are expanded to a number 300-fold the initial starting HSC number, the expansion lasts for 8 days.
- the present disclosure provides a myeloid-derived cell that has been generated from a stem cell using the methods and compositions disclosed herein. Also disclosed are cell populations comprising a plurality of the myeloid-derived cells disclosed herein. Importantly, the methods disclosed herein are envisaged as being effective at generating both myeloid-derived cells, as well as myeloid-derived CAR cells. As such, it would be understood that any myeloid-derived cell, or population comprising a myeloid-derived cell, generated from the disclosed methods can include, and encompass, for the purposes of the present disclosure, a myeloid-derived CAR cell and/or a myeloid-derived cell that does not comprise a CAR (i.e., a myeloid-derived cell). For instance, any cell population described in the present application can be generated to comprise myeloid-derived cells and/or myeloid-derived CAR cells.
- the present disclosure provides a myeloid-derived cell genetically modified to express a CAR.
- the myeloid-derived cells disclosed herein have been generated by expanding and differentiating a precursor CAR cell (e.g., a stem cell CAR cell) using the precursor and, optionally, the myeloid media disclosed herein.
- the precursor CAR cell can be differentiated into any myeloid-derived CAR cell known in the art.
- Myeloid cells i.e., cells within the myeloid lineage that are derived from a common myeloid progenitor cell
- the myeloid-derived CAR cell is a partially differentiated myeloid-derived cell.
- a partially differentiated cell is a cell that has undergone differentiation, and thus is no longer a stem cell, and has the capacity to undergo additional differentiation into a more specialized myeloid cell within the myeloid lineage.
- the myeloid-derived CAR cell is fully differentiated myeloid cell.
- the organization of the myeloid lineage, and, for example, the differentiation capacity of myeloid cells is known in the art, as described for example, in Weiskopf, Kipp et al. “Myeloid Cell Origins, Differentiation, and Clinical Implications.” Microbiology spectrum vol.
- Exemplary, non-limiting, cells within the myeloid lineage include: common myeloid progenitor (CMP), granulo-monocyte progenitors, monocyte-dendritic cell progenitors, common dendritic cell progenitors, myeoblasts, monoblasts, pre-conventional dendritic cells, promocytes, granulocytes, polymorphonuclear leukocytes (PMNs), myeloid-derived suppressor cells (MDSCs), mast cells, basophils, eosinophils, megakaryocytes, neutrophils, dendritic cells, monocytes, and macrophages.
- CMP common myeloid progenitor
- granulo-monocyte progenitors granulo-monocyte progenitors
- monocyte-dendritic cell progenitors common dendritic cell progenitors
- myeoblasts monoblasts
- pre-conventional dendritic cells promocytes
- a myeloid-derived cell can be identified and isolated based on cell surface markers, using any method available in the art.
- the myeloid-derived CAR cell is a granulocyte.
- the myeloid-derived CAR cell is a phagocytic cell.
- the myeloid-derived CAR cell is a monocyte, macrophage, dendritic cell, neutrophil, or a precursor thereof.
- cell populations comprising the genetically modified precursor CAR cells and/or myeloid-derived CAR cells generated by the methods described herein.
- the precursor CAR cell(s) disclosed herein can be expanded to generate a population comprising a plurality of genetically modified precursor CAR cells or expanded and differentiated to generate a population comprising a plurality of genetically modified myeloid-derived CAR cells.
- the genetically modified myeloid-derived cells can include any cell within the myeloid lineage that has been derived from a precursor as described herein.
- the precursor can comprise a bone marrow-derived cell, a blood-derived cell, or a stem cell.
- the precursor can be a hematopoietic stem cell.
- cell populations comprising a plurality of precursor CAR cells.
- the cell population comprises a plurality of precursor CAR cells
- said precursor cell is a stem cell.
- the stem cell is a hematopoietic stem cell.
- the cell population comprises a plurality of precursor CAR cells, wherein said precursor cell is a hematopoietic stem cell.
- the cell populations disclosed herein can be mixed populations.
- the cell population can comprise a plurality of precursor CAR cells and a plurality of myeloid-derived CAR cells. Any ratio of precursor CAR cells to myeloid-derived CAR cells within the cell populations disclosed herein is envisaged within the scope of the present application.
- the ratio of myeloid-derived cells to precursor cells will be expected to increase (i.e., the number of myeloid-derived cells in the population increases compared to the number of precursor cells).
- the number of myeloid-derived cells within the population can increase and/or the number of precursor cells within the population can decrease.
- the cell population can undergo expansion (i.e., the number of cells within the population increases).
- the cell population can undergo differentiation (i.e., a plurality of precursor cells within the population differentiates into myeloid cells).
- the myeloid-derived cell that is generated from the precursor cell can include any myeloid lineage cell disclosed herein.
- the myeloid-derived cell that is generated from the precursor cell can include any myeloid cell disclosed herein.
- the myeloid-derived cell that is generated from the precursor can include, for example, phagocytic cells, such as a macrophage, monocyte, dendritic cell, neutrophil, and/or precursor thereof.
- the myeloid-derived cell that is generated from the precursor can be a precursor of a phagocytic cells, such as any cell within the myeloid lineage, such as a myeloid precursor cell, such as a myeloid progenitor cell, granulo-monocyte progenitor cell, monocyte- dendritic cell progenitor/monoblasts, promonocyte, or myeloblast.
- a myeloid precursor cell such as a myeloid progenitor cell, granulo-monocyte progenitor cell, monocyte- dendritic cell progenitor/monoblasts, promonocyte, or myeloblast.
- the phagocytic cell, or precursor thereof, comprising a CAR recognizes and binds to an antigen present on the surface of a target cell, such as a cancer cell and/or viral particle.
- the CAR includes a co-stimulatory domain having at least 90% sequence identity to an HVEM co-stimulatory protein set forth as any one of SEQ ID NOs: 7, 1, or 2, or a functional fragment or variant thereof that retains co-stimulatory activity.
- Populations of cells comprising a plurality wherein at least 40%, at least 50%, at least 60%, at least 70%, at least 80%, at least 90%, at least 95%, or at least 99% of said plurality expresses a CAR on the cell surface are also disclosed.
- cell populations comprising a plurality of any of the genetically modified myeloid-derived CAR cells generated by the method described herein. Also disclosed are cell populations comprising a plurality of any of the genetically modified precursor CAR cells described herein.
- the cell populations disclosed herein can comprise any of the precursor CAR cells and/or myeloid-derived CAR cells disclosed herein.
- the cell populations comprise a precursor CAR cell and myeloid-derived CAR cell, any ratio of the number of precursor CAR cells to the number of myeloid-derived CAR cells is envisaged as within the scope of the cell populations of the present application.
- the myeloid-derived CAR cells are differentiated from a stem cell, at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of said plurality can be CD1 lb+ or CD14+ or Cl lb+CD14+.
- the cell populations disclosed herein can comprise a plurality of cells with any of the surface marker expression profiles disclosed herein.
- the cells within the cell populations disclosed herein can comprise any of the surface marker expression profiles disclosed herein.
- disclosed herein is a cell population comprising a plurality of precursor CAR cells and a plurality of myeloid-derived CAR cells, wherein between 90%-99.9% of said population is CD34+ cells.
- a cell population wherein said population is a mixed population comprising a plurality of precursor CAR cells and a plurality of myeloid-derived CAR cells, wherein at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% of said population is CD34+ cells.
- a cell population wherein said population is a mixed population comprising a plurality of precursor CAR cells and a plurality of myeloid-derived CAR cells, or a plurality of precursor cells and a plurality of myeloid-derived cells, wherein: a. no more than 99%, no more than 98%, no more than 97%, no more than 96%, no more than 95%, no more than 90%, no more than 80%, no more than 70%, no more than 60%, no more than 50%, no more than 40%, no more than 30%, no more than 20%, no more than 10%, no more than 5%, no more than 1%, no more than 0.5%, or no more than 0.1% of said mixed population of cells is CD34+; b.
- CD1 lb+ between about 30%-80% of said mixed population of cells is CD1 lb+; g. at least 0.1%, at least 0.5%, at least 1%, at least 2%, at least 3%, at least 4%, at least 5%, at least 6%, at least 7%, at least 8%, at least 9%, or at least 10% of said mixed population of cells is CD14+; h. between about 0.5%-10% of said mixed population of cells is CD14+; i. at least 20%, at least 30%, at least 40%, at least 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95% of said mixed population is CD13+; and/or j. between about 85%-95% of said mixed population of cells is CD13+.
- the myeloid-derived cell or myeloid-derived CAR cell can be a common myeloid progenitor (CMP), granulo-monocyte progenitor, monocyte- dendritic cell progenitor, common dendritic cell progenitor, myeoblast, monoblast, pre- conventional dendritic cell, promocyte, granulocyte, polymorphonuclear leukocyte (PMN), myeloid-derived suppressor cell (MDSC), mast cell, basophil, eosinophils, megakaryocyte, neutrophil, dendritic cell, monocyte, and/or a macrophage.
- the precursor cell or precursor CAR cell can be a hematopoietic stem cell (HSC).
- a cell population wherein said population is a mixed population comprising a plurality of precursor CAR cells and a plurality of myeloid-derived CAR cells , or a plurality of precursor cells and a plurality of myeloid-derived cells, wherein: a. no more than 99%, no more than 98%, no more than 97%, no more than 96%, no more than 95%, no more than 90%, no more than 80%, no more than 70%, no more than 60%, no more than 50%, no more than 40%, no more than 30%, no more than 20%, no more than 10%, no more than 5%, no more than 1%, no more than 0.5%, or no more than 0.1% of said mixed population of cells is CD34+; b.
- CD1 lb+ between about 30%-80% of said mixed population of cells is CD1 lb+; g. at least 0.1%, at least 0.5%, at least 1%, at least 2%, at least 3%, at least 4%, at least 5%, at least 6%, at least 7%, at least 8%, at least 9%, or at least 10% of said mixed population of cells is CD14+; h. between about 0.5%-10% of said mixed population of cells is CD14+; i. at least 20%, at least 30%, at least 40%, at least 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95% of said mixed population is CD13+; and/or j.
- the myeloid-derived cell or myeloid-derived CAR cell is a common myeloid progenitor (CMP), granulo-monocyte progenitor, monocyte-dendritic cell progenitor, common dendritic cell progenitor, myeoblast, monoblast, pre-conventional dendritic cell, promocyte, granulocyte, polymorphonuclear leukocyte (PMN), myeloid-derived suppressor cell (MDSC), mast cell, basophil, eosinophils, megakaryocyte, neutrophil, dendritic cell, monocyte, and/or a macrophage.
- CMP myeloid progenitor
- PMN polymorphonuclear leukocyte
- MDSC myeloid-derived suppressor cell
- mast cell basophil, eosinophils, megakaryocyte, neutrophil, dendritic cell, monocyte, and/or a macrophage.
- a cell population wherein said population is a mixed population comprising a plurality of precursor CAR cells and a plurality of myeloid-derived CAR cells, or a plurality of precursor cells and a plurality of myeloid-derived cells, wherein a. no more than 1%, no more than 0.9%, no more than 0.8%, no more than 0.7%, no more than 0.6%, no more than 0.5%, no more than 0.4%, no more than 0.3%, no more than 0.2%, no more than 0.1%, or less than 0.1% of said population of cells is CD34+; b.
- At least 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95% of said population is CD15+; c. between about 50%-95% of said population of cells is CD15+; d. least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of said population is CD1 lb+; e. between about 75%-99% of said population of cells is CD1 lb+; f. at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, at least 50%, at least 60%, at least 70%, at least 80%, or at least 90% of said population of cells is CD14+; g.
- CD14+ between about 5%-90% of said population of cells is CD14+; h. at least 85%, at least 90%, at least 95%, or at least 99% of said population is CD13+; and/or i. between about 85%-99% of said population of cells is CD13+.
- the myeloid-derived cell or myeloid- derived CAR cell can be a common myeloid progenitor (CMP), granulo-monocyte progenitor, monocyte-dendritic cell progenitor, common dendritic cell progenitor, myeoblast, monoblast, pre- conventional dendritic cell, promocyte, granulocyte, polymorphonuclear leukocyte (PMN), myeloid-derived suppressor cell (MDSC), mast cell, basophil, eosinophils, megakaryocyte, neutrophil, dendritic cell, monocyte, and/or a macrophage.
- the precursor cell can be a hematopoietic stem cell (HSC).
- a cell population wherein said population is a mixed population comprising a plurality of precursor CAR cells and a plurality of myeloid-derived CAR cells, or a plurality of precursor cells and a plurality of myeloid-derived cells, wherein a. no more than 1%, no more than 0.9%, no more than 0.8%, no more than 0.7%, no more than 0.6%, no more than 0.5%, no more than 0.4%, no more than 0.3%, no more than 0.2%, no more than 0.1%, or less than 0.1% of said population of cells is CD34+; b.
- At least 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95% of said population is CD15+; c. between about 50%-95% of said population of cells is CD15+; d. least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of said population is CD1 lb+; e. between about 75%-99% of said population of cells is CD1 lb+; f. at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, at least 50%, at least 60%, at least 70%, at least 80%, or at least 90% of said population of cells is CD14+; g.
- the myeloid-derived cell or myeloid-derived CAR cell is a common myeloid progenitor (CMP), granulo-monocyte progenitor, monocyte-dendritic cell progenitor, common dendritic cell progenitor, myeoblast, monoblast, pre-conventional dendritic cell, promocyte, granulocyte, polymorphonuclear leukocyte (PMN), myeloid-derived suppressor cell (MDSC), mast cell, basophil, eosinophils, megakaryocyte, neutrophil, dendritic cell, monocyte, and/or a macrophage.
- CMP myeloid progenitor
- PMN polymorphonuclear leukocyte
- MDSC myeloid-derived suppressor cell
- the cell population can comprise at least 10 4 , at least 10 5 , at least 10 6 , at least 10 7 , at least 10 8 , at least 10 9 , at least 10 10 , at least 10 11 , at least 10 12 , at least 10 13 , at least 10 14 , at least 10 15 , at least 10 16 , at least 10 17 , at least 10 18 , at least 10 19 , at least IO 20 , or more than IO 20 cells.
- the cell population comprises at least 10 4 , at least 10 5 , at least 10 6 , at least 10 7 , at least 10 8 , at least 10 9 , at least IO 10 , at least IO 11 , at least 10 12 , at least IO 13 , at least 10 14 , at least IO 15 , at least 10 16 , at least 10 17 , at least 10 18 , at least 10 19 , at least IO 20 , or more than IO 20 cells, wherein said population comprises a plurality of myeloid-derived cells and/or myeloid-derived CAR cells, and wherein: a.
- At least 20%, at least 30%, at least 40%, at least 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 95%, or at least 90% of said population is CD15+; d. between about 60%-90% of said mixed population of cells is CD15+; e. at least 20%, at least 30%, at least 40%, at least 50%, at least 60%, at least 70%, at least 75%, at least 80%, or at least 85%, of said population is CD1 lb+; f. between about 30%-80% of said population of cells is CD1 lb+; g.
- At least 0.1%, at least 0.5%, at least 1%, at least 2%, at least 3%, at least 4%, at least 5%, at least 6%, at least 7%, at least 8%, at least 9%, or at least 10% of said population of cells is CD14+; h. between about 0.5%-10% of said population of cells is CD14+; i. at least 20%, at least 30%, at least 40%, at least 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95% of said population is CD13+; and/or j. between about 85%-95% of said population of cells is CD13+.
- the myeloid-derived cell and/or myeloid-derived CAR cell can be a common myeloid progenitor (CMP), granulo-monocyte progenitor, monocyte-dendritic cell progenitor, common dendritic cell progenitor, myeoblast, monoblast, pre-conventional dendritic cell, promocyte, granulocyte, polymorphonuclear leukocyte (PMN), myeloid-derived suppressor cell (MDSC), mast cell, basophil, eosinophils, megakaryocyte, neutrophil, dendritic cell, monocyte, and/or a macrophage.
- CMP myeloid progenitor
- PMN polymorphonuclear leukocyte
- MDSC myeloid-derived suppressor cell
- mast cell basophil, eosinophils, megakaryocyte, neutrophil, dendritic cell, monocyte, and/or a macrophage.
- the cell population can comprise at least 10 4 , at least 10 5 , at least 10 6 , at least 10 7 , at least 10 8 , at least 10 9 , at least 10 10 , at least 10 11 , at least 10 12 , at least 10 13 , at least 10 14 , at least 10 15 , at least 10 16 , at least 10 17 , at least 10 18 , at least 10 19 , at least IO 20 , or more than IO 20 cells.
- the cell population comprises at least 10 4 , at least 10 5 , at least 10 6 , at least 10 7 , at least 10 8 , at least 10 9 , at least IO 10 , at least 10 11 , at least 10 12 , at least 10 13 , at least 10 14 , at least 10 15 , at least 10 16 , at least 10 17 , at least 10 18 , at least 10 19 , at least IO 20 , or more than IO 20 cells
- said population comprises a plurality of myeloid-derived cells and/or myeloid-derived CAR cells, and wherein: a.
- At least 20%, at least 30%, at least 40%, at least 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 95%, or at least 90% of said population is CD15+; d. between about 60%-90% of said mixed population of cells is CD15+; e. at least 20%, at least 30%, at least 40%, at least 50%, at least 60%, at least 70%, at least 75%, at least 80%, or at least 85%, of said population is CD1 lb+; f. between about 30%-80% of said population of cells is CD1 lb+; g.
- At least 0.1%, at least 0.5%, at least 1%, at least 2%, at least 3%, at least 4%, at least 5%, at least 6%, at least 7%, at least 8%, at least 9%, or at least 10% of said population of cells is CD14+; h. between about 0.5%-10% of said population of cells is CD14+; i. at least 20%, at least 30%, at least 40%, at least 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95% of said population is CD13+; and/or j.
- the myeloid- derived cell and/or myeloid-derived CAR cell is a common myeloid progenitor (CMP), granulo- monocyte progenitor, monocyte-dendritic cell progenitor, common dendritic cell progenitor, myeoblast, monoblast, pre-conventional dendritic cell, promocyte, granulocyte, polymorphonuclear leukocyte (PMN), myeloid-derived suppressor cell (MDSC), mast cell, basophil, eosinophils, megakaryocyte, neutrophil, dendritic cell, monocyte, and/or a macrophage.
- CMP myeloid progenitor
- PMN polymorphonuclear leukocyte
- MDSC myeloid-derived suppressor cell
- mast cell basophil, eosinophils, megakaryocyte, neutrophil, dendritic cell, monocyte, and/or a macrophage.
- the cell population can comprise at least 10 4 , at least 10 5 , at least 10 6 , at least 10 7 , at least 10 8 , at least 10 9 , at least 10 10 , at least 10 11 , at least 10 12 , at least 10 13 , at least 10 14 , at least 10 15 , at least 10 16 , at least 10 17 , at least 10 18 , at least 10 19 , at least IO 20 , or more than IO 20 cells.
- the cell population comprises at least 10 4 , at least 10 5 , at least 10 6 , at least 10 7 , at least 10 8 , at least 10 9 , at least IO 10 , at least 10 11 , at least 10 12 , at least 10 13 , at least 10 14 , at least 10 15 , at least 10 16 , at least 10 17 , at least 10 18 , at least 10 19 , at least IO 20 , or more than IO 20 cells
- said population comprises a plurality of myeloid-derived cells and/or myeloid-derived CAR cells, and wherein: a.
- no more than 1%, no more than 0.9%, no more than 0.8%, no more than 0.7%, no more than 0.6%, no more than 0.5%, no more than 0.4%, no more than 0.3%, no more than 0.2%, no more than 0.1%, or less than 0.1% of said population of cells is CD34+; b. at least 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95% of said population is CD15+; c. between about 50%-95% of said population of cells is CD15+; d. least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of said population is CD1 lb+; e.
- CD1 lb+ between about 75%-99% of said population of cells is CD1 lb+; f. at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, at least 50%, at least 60%, at least 70%, at least 80%, or at least 90% of said population of cells is CD14+; g. between about 5%-90% of said population of cells is CD14+; h. at least 85%, at least 90%, at least 95%, or at least 99% of said population is CD13+; and/or i. between about 85%-99% of said population of cells is CD13+.
- the myeloid- derived cell and/or myeloid-derived CAR cell can be a common myeloid progenitor (CMP), granulo-monocyte progenitor, monocyte-dendritic cell progenitor, common dendritic cell progenitor, myeoblast, monoblast, pre-conventional dendritic cell, promocyte, granulocyte, polymorphonuclear leukocyte (PMN), myeloid-derived suppressor cell (MDSC), mast cell, basophil, eosinophils, megakaryocyte, neutrophil, dendritic cell, monocyte, and/or a macrophage.
- CMP myeloid progenitor
- PMN polymorphonuclear leukocyte
- MDSC myeloid-derived suppressor cell
- mast cell basophil, eosinophils, megakaryocyte, neutrophil, dendritic cell, monocyte, and/or a macrophage.
- the cell population can comprise at least 10 4 , at least 10 5 , at least 10 6 , at least 10 7 , at least 10 8 , at least 10 9 , at least IO 10 , at least 10 11 , at least 10 12 , at least 10 13 , at least 10 14 , at least 10 15 , at least 10 16 , at least 10 17 , at least 10 18 , at least 10 19 , at least IO 20 , or more than IO 20 cells.
- the cell population comprises at least 10 4 , at least 10 5 , at least 10 6 , at least 10 7 , at least 10 8 , at least 10 9 , at least IO 10 , at least 10 11 , at least 10 12 , at least 10 13 , at least 10 14 , at least 10 15 , at least 10 16 , at least 10 17 , at least 10 18 , at least 10 19 , at least IO 20 , or more than IO 20 cells
- said population comprises a plurality of myeloid-derived CAR cells, and wherein: a.
- no more than 1%, no more than 0.9%, no more than 0.8%, no more than 0.7%, no more than 0.6%, no more than 0.5%, no more than 0.4%, no more than 0.3%, no more than 0.2%, no more than 0.1%, or less than 0.1% of said population of cells is CD34+; b. at least 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95% of said population is CD15+; c. between about 50%-95% of said population of cells is CD15+; d. least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of said population is CD1 lb+; e.
- CD1 lb+ between about 75%-99% of said population of cells is CD1 lb+; f. at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, at least 50%, at least 60%, at least 70%, at least 80%, or at least 90% of said population of cells is CD14+; g. between about 5%-90% of said population of cells is CD14+; h. at least 85%, at least 90%, at least 95%, or at least 99% of said population is CD13+; and/or i.
- the myeloid-derived cell and/or myeloid-derived CAR cell is a common myeloid progenitor (CMP), granulo-monocyte progenitor, monocyte-dendritic cell progenitor, common dendritic cell progenitor, myeoblast, monoblast, pre-conventional dendritic cell, promocyte, granulocyte, polymorphonuclear leukocyte (PMN), myeloid-derived suppressor cell (MDSC), mast cell, basophil, eosinophils, megakaryocyte, neutrophil, dendritic cell, monocyte, and/or a macrophage.
- CMP myeloid progenitor
- PMN polymorphonuclear leukocyte
- MDSC myeloid-derived suppressor cell
- mast cell basophil, eosinophils, megakaryocyte, neutrophil, dendritic cell, monocyte, and/or a macrophage.
- a cell population wherein said population is a mixed population comprising plurality of myeloid-derived cells, or a plurality of myeloid-derived CAR cells, wherein: a. no more than 5%, no more than 4%, no more than 3%, no more than 2%, no more than 1%, no more than 0.5%, or no more than 0.1% of cells within said population are CD34+; b. at least 50%, at least 55%, at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 86%, at least 87%, at least 88%, at least 89%, or at least 90% of cells within said population are CD14+; c.
- At least 70%, at least 80%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or at least 99.5%% of cells within said population are CD1 lb+; d. at least 90%, at least 91%, at least 92%, at least 94%, or at least 95% of cells within said population are CD13+; and/or e. at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, or at least 95% of cells within said population are CD15+.
- the myeloid- derived cell and/or myeloid-derived CAR cell can be a common myeloid progenitor (CMP), granulo-monocyte progenitor, monocyte-dendritic cell progenitor, common dendritic cell progenitor, myeoblast, monoblast, pre-conventional dendritic cell, promocyte, granulocyte, polymorphonuclear leukocyte (PMN), myeloid-derived suppressor cell (MDSC), mast cell, basophil, eosinophils, megakaryocyte, neutrophil, dendritic cell, monocyte, and/or a macrophage.
- the precursor cell can be a hematopoietic stem cell (HSC).
- a cell population wherein said population is a mixed population comprising plurality of myeloid-derived cells and/or myeloid-derived CAR cells, wherein: a. no more than 5%, no more than 4%, no more than 3%, no more than 2%, no more than 1%, no more than 0.5%, or no more than 0.1% of cells within said population are CD34+; b. at least 50%, at least 55%, at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 86%, at least 87%, at least 88%, at least 89%, or at least 90% of cells within said population are CD14+; c.
- At least 70%, at least 80%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or at least 99.5%% of cells within said population are CD1 lb+; d. at least 90%, at least 91%, at least 92%, at least 94%, or at least 95% of cells within said population are CD13+; and/or e. at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, or at least 95% of cells within said population are CD15+.
- the myeloid-derived cell and/or myeloid-derived CAR cell can be a common myeloid progenitor (CMP), granulo-monocyte progenitor, monocyte-dendritic cell progenitor, common dendritic cell progenitor, myeoblast, monoblast, pre-conventional dendritic cell, promocyte, granulocyte, polymorphonuclear leukocyte (PMN), myeloid-derived suppressor cell (MDSC), mast cell, basophil, eosinophils, megakaryocyte, neutrophil, dendritic cell, monocyte, and/or a macrophage.
- the precursor cell can be a hematopoietic stem cell (HSC).
- Any cell population described herein can comprise at least 10 4 , at least 10 5 , at least 10 6 , at least 10 7 , at least 10 8 , at least 10 9 , at least 10 10 , at least 10 11 , at least 10 12 , at least 10 13 , at least 10 14 , at least 10 15 , at least 10 16 , at least 10 17 , at least 10 18 , at least 10 19 , at least IO 20 , or more than IO 20 cells.
- the cell population comprises at least 10 4 , at least 10 5 , at least 10 6 , at least 10 7 , at least 10 8 , at least 10 9 , at least IO 10 , at least 10 11 , at least 10 12 , at least 10 13 , at least 10 14 , at least 10 15 , at least 10 16 , at least 10 17 , at least 10 18 , at least 10 19 , at least IO 20 , or more than IO 20 cells
- said population comprises a plurality of myeloid-derived cells and/or myeloid- derived CAR cells, wherein: a.
- the myeloid-derived cell and/or myeloid-derived CAR cell is a common myeloid progenitor (CMP), granulo-monocyte progenitor, monocyte-dendritic cell progenitor, common dendritic cell progenitor, myeoblast, monoblast, pre-conventional dendritic cell, promocyte, granulocyte, polymorphonuclear leukocyte (PMN), myeloid-derived suppressor cell (MDSC), mast cell, basophil, eosinophils, megakaryocyte, neutrophil, dendritic cell, monocyte, and/or a macrophage.
- CMP myeloid progenitor
- PMN polymorphonuclear leukocyte
- MDSC myeloid-derived suppressor cell
- any myeloid-derived cell(s) and/or populations comprising a plurality of myeloid-derived cell(s) disclosed herein are intended to encompass myeloid-derived cells that: a. comprise a CAR (i.e., myeloid-derived CAR cells); or b. do not comprise a CAR. Any of the cell populations described herein can be generated wherein the myeloid-derived cells do not comprise a CAR.
- the present disclosure provides myeloid-derived cells that are generated from a precursor cell using the methods and compositions disclosed herein.
- the precursor cell can be a stem cell, such as a hematopoietic stem cell, and/or a CD34+ cell.
- the stem cell can be isolated from a subject.
- the subject can be a subject in need of a treatment, for which administration of the myeloid-derived cells that are generated from said isolated stem cells can be used as a treatment or a manufacture of a medicament for a treatment.
- cell populations comprising a plurality of the myeloid-derived cells generated from HSCs.
- the myeloid-derived cells, and populations comprising the same, disclosed herein do not require the presence of a CAR to effectively generate a myeloid-derived cell.
- the method of manufacturing a myeloid-derived cell disclosed herein comprises the step of contacting a precursor cell with a precursor media comprising a precursor cytokine mixture, whereby the number of precursor cell(s) increases to generate a cell population comprising a plurality of precursor cells, wherein said precursor cell is a stem cell, wherein the method further comprises the step of contacting said cell population comprising a plurality of precursor cells with a myeloid media comprising a myeloid cytokine mixture, whereby, said precursor cell(s) differentiates into a myeloid-lineage cell to generate a myeloid-derived cell and/or a mixed cell population comprising a plurality of myeloid-derived cells and/or a plurality of precursor cells.
- the myeloid-derived cell can be any cell within the myeloid lineage.
- myeloid lineage cells i.e, myeloid cells
- cell surface marker expression i.e, myeloid cells
- Known cell surface markers associated with myeloid cells are known in the art.
- the myeloid-derived cell is CD14+, CDl lb+, CD13+, and/or CD15+.
- a CAR is not envisaged as impacting the ability of the methods disclosed herein to effectively generate a myeloid-derived cell from a hematopoietic stem cell.
- myeloid-derived cells genetically modified to express a CAR.
- the CAR is introduced into the precursor cell prior to expansion and differentiation of said precursor CAR cell.
- a precursor CAR cell is generated, said cell can be contacted with, and maintained in the precursor media and, optionally, the myeloid media, disclosed herein to generated a myeloid-derived CAR cell.
- the compositions and methods disclosed herein provide for a more stable process for generating a myeloid-derived CAR cell.
- the precursor CAR cell maintains the CAR throughout the expansion and differentiation process, providing a more efficient and effective process for generating myeloid-derived CAR cells compared to known methods.
- the method of manufacturing a myeloid-derived cell disclosed herein comprises the step of contacting a precursor cell, such as an HSC, with a nucleic acid molecule comprising a polynucleotide sequence encoding a chimeric antigen receptor (CAR), whereby said nucleic acid is introduced into said precursor cell to generate a precursor CAR cell comprising a chimeric antigen receptor (CAR), wherein the method further comprises the step of contacting the precursor CAR cell with a precursor media comprising a precursor cytokine mixture, whereby the number of precursor CAR cell(s) increases to generate a cell population comprising a plurality of precursor CAR cells, wherein said precursor CAR cell is a stem cell.
- a precursor cell such as an HSC
- a nucleic acid molecule comprising a polynucleotide sequence encoding a chimeric antigen receptor (CAR)
- CAR chimeric antigen receptor
- the method further comprises the step of contacting the precursor CAR cell with
- the method can further comprise the step of contacting said cell population comprising a plurality of precursor CAR cells with a myeloid media comprising a myeloid cytokine mixture, whereby, said precursor CAR cell(s) differentiates into a myeloid-lineage cell to generate a myeloid-derived CAR cell and/or a mixed cell population comprising a plurality of myeloid-derived CAR cells and/or a plurality of precursor CAR cells.
- the myeloid-derived CAR cell can be any cell within the myeloid lineage.
- the myeloid-derived CAR cell is CD14+, CDl lb+, CD13+, and/or CD15+.
- a genetically modified myeloid-derived cell comprising a chimeric antigen receptor (CAR)
- CAR chimeric antigen receptor
- methods of manufacturing a genetically modified myeloid-derived cell comprising a chimeric antigen receptor (CAR) comprising: (a) contacting a precursor with a nucleic acid molecule comprising a polynucleotide sequence encoding the CAR to generate a genetically modified precursor CAR cell; and (b) expanding said genetically modified precursor CAR cell in a precursor media comprising a precursor cytokine mixture to generate a population comprising a plurality of genetically modified precursor CAR cells, wherein the precursor comprises a bone marrow-derived cell or a stem cell.
- CAR chimeric antigen receptor
- the methods can further comprise differentiating said genetically modified precursor CAR cell(s) in a myeloid media comprising a myeloid cytokine mixture to generate a myeloid-derived CAR cell or a population comprising a plurality of genetically modified myeloid-derived CAR cells.
- the precursor media can be removed prior to addition of the myeloid media.
- the precursor e.g., stem cells
- the precursor can be isolated from a human subject.
- the methods disclosed herein can be used to manufacture a genetically modified myeloid- derived cell from a precursor.
- the term “precursor” when used in connection with the myeloid- derived cells indicate parent cells in a cellular lineage resulting in any cell within the myeloid lineage herein described.
- Exemplary precursor cells include bone marrow-derived cells, stem cells, and other precursor cells identifiable by a person skilled in the art.
- Precursor cells can also indicate cells that have the potential to differentiate resulting in any cell within the myeloid lineage herein described.
- Exemplary precursor cells can include bone marrow-derived cells, stem cells, and other precursor cells identifiable by a person skilled in the art.
- the precursor is isolated from a source selected from one of the following: bone marrow, mobilized peripheral blood, peripheral blood, and umbilical cord blood. In some instances, the precursor is isolated from a source selected from one of the following: bone marrow, mobilized peripheral blood, peripheral blood, and umbilical cord blood.
- the stem cell is isolated from a source selected from one of the following: bone marrow, mobilized peripheral blood, peripheral blood, and umbilical cord blood.
- the bone marrow-derived cell or stem cell used for the disclosed method can be isolated from a subject, such as a human subject. In some instances, when the bone marrow- derived cell or stem cell is isolated from a subject, the cell is isolated from peripheral blood.
- the peripheral blood is mobilized peripheral blood.
- the peripheral blood is mobilized with a cytokine.
- the at least one cytokine comprises granulocyte-colony stimulating factor (G-CSF) or granulocyte-macrophage colony stimulating factor (GM-CSF), or a combination thereof.
- G-CSF granulocyte-colony stimulating factor
- GM-CSF granulocyte-macrophage colony stimulating factor
- the method disclosed herein comprises expanding and differentiation a precursor cell into a genetically modified myeloid-derived CAR cell.
- the precursor cell disclosed herein comprises a bone marrow-derived cell or a stem cell.
- the precursor cell can be a stem cell, wherein the stem cell is a pluripotent stem cell, such as a hematoiepoietic stem cell (HSC).
- the stem cell is a hematoiepoietic stem cell (HSC).
- the stem cells Prior to being expanded in a precursor media, are genetically modified with a CAR.
- Genetic modification with a CAR comprises contacting the precursor cell with a nucleic acid molecule comprising a polynucleotide sequence encoding the CAR.
- a “myeloid-derived” cell is a differentiated descendant from common progenitors derived from hematopoietic stem cells in, or isolated from, tissue such as the bone marrow or other source of blood tissue.
- a myeloid-derived cell can be any cell within the myeloid lineage, for example, a common myeloid progenitor (CMP) cell, or any cell derived therefrom, such as any granulocyte or monocyte cell.
- CMP common myeloid progenitor
- the myeloid-derived cells described herein are genetically modified to express a CAR (i.e., genetically modified myeloid-derived CAR cell).
- the genetically modified myeloid-derived cells can include any cell within the myeloid lineage that has been derived from a precursor as described herein.
- the myeloid-derived cell can include, for example, phagocytic cells, such as a macrophage, monocyte, dendritic cell, neutrophil, and/or precursor thereof within the myeloid lineage.
- the genetically modified myeloid-derived cell is a phagocytic cell.
- a “precursor” as described herein comprises a bone marrow-derived cell or a stem cell.
- the precursor can be a pluripotent stem cell, such as a hematopoietic stem cell (HSC).
- HSC hematopoietic stem cell
- a “stem cell” would be understood to be a cell that can self-renew and a cell from which other types of cells develop.
- a stem cell can be a hematopoietic stem cell.
- a hematopoietic stem cell would be understood to be an immature cell that can develop into various types of blood cells, including, for example, white blood cells, red blood cells, and platelets. Hematopoietic stem cells can be found in the peripheral blood and the bone marrow.
- the hematopoietic stem cell is a multipotent stem cell that resides in tissues such as cord blood, peripheral blood, and the bone marrow.
- Hematopoietic stem cells lay the foundation of hematopoiesis to generate all functional hematopoietic lineages, including erythrocytes, leukocytes (neutrophils, basophils, eosinophils, lymphocytes, monocytes and macrophages) and platelets. HSCs can self-replicate and differentiate into progeny of multiple lineages.
- the hematopoietic lineage is organized such that HSCs sit atop the hierarchy and give rise to committed progenitor cells, which in turn give rise to mature, differentiated cells.
- Mature HSCs generate multipotent progenitor (MPP) cells which have the potential to further differentiate into two major lineages: common myeloid progenitor (CMP) and common lymphoid progenitor (CLP).
- MPP multipotent progenitor
- CMP common myeloid progenitor
- CLP common lymphoid progenitor
- Downstream of MPPs are committed progenitors that are oligopotent and have limited ability to self-renew.
- CMP common myeloid progenitor
- CLP common lymphoid progenitor
- a myeloid-derived cell can be generated from a precursor.
- the precursor is a bone marrow-derived cell, a cord blood-derived cell, or a peripheral blood-derived cell.
- the precursor is a stem cell.
- a stem cell can be a hematopoietic stem cell that has been isolated from a subject using any know method in the art.
- the precursor is in a cell population comprising a plurality of precursors.
- the population comprising the plurality of precursors is a homogenous cell population.
- the cell population can also be a heterogenous cell population.
- myeloid precursor cells include cells such as a myeloid progenitor cell, granulo-monocyte progenitor cell, monocyte-dendritic cell progenitor/monoblasts, promonocyte, or myeloblast.
- the present disclosure comprises expanding a population of HSCs comprising a CAR, and expanding and differentiating them into myeloid progenitor cells, granulo-monocyte progenitor cells, monocyte-dendritic cell progenitor/monoblasts, promonocytes, myeloblasts, monocytes, macrophages, dendritic cells, and/or neutrophils, as described herein.
- the genetically modified myeloid- derived cell is a myeloid progenitor cell. In some embodiments, the genetically modified myeloid- derived cell is a granulo-monocyte progenitor cell. In some embodiments, the genetically modified myeloid-derived cell is a monocyte-dendritic cell progenitor/monoblast. In some embodiments, the genetically modified myeloid-derived cell is a promonocyte. In some embodiments, the genetically modified myeloid-derived cell is a myeloblasts. In some embodiments, the genetically modified myeloid-derived cell is a monocyte. In some embodiments, the genetically modified myeloid- derived cell is a macrophage. In some embodiments, the genetically modified myeloid-derived cell is a dendritic cell. In some embodiments, the genetically modified myeloid-derived cell is a neutrophil.
- Phagocytic cells are used in the compositions and methods described herein.
- the genetically modified myeloid-derived cell can be a phagocytic cell.
- phagocytic cell or “phagocyte” or plural forms thereof as used herein indicate a cell that is capable of phagocytosis, which is the process by which a cell uses its plasma membrane to engulf a large particle (>0.5 pm), giving rise to an internal compartment called the phagosome.
- Phagocytosis is one type of endocytosis as will be understood by a skilled person. Phagocytes of an individual typically use their plasma membrane to engulf and remove cellular debris, foreign substances, microbes, and cells to protect the body of an individual.
- Phagocytes in the sense of the disclosure can typically also perform trogocytosis, a process whereby one cell contacts and quickly nibbles another cell.
- trogocytosis occurs when lymphocytes (e.g., B, T, NK cells) conjugated to antigen-presenting cells (APCs) extract surface molecules from the APCs and express them on their own surface.
- lymphocytes e.g., B, T, NK cells
- APCs antigen-presenting cells
- Exemplary phagocytic cells herein described include macrophages, monocytes, neutrophils, dendritic cells and precursors thereof as a person skilled in the art would understand, though singled celled organisms such as Dictyostelium amoebae are also phagocytes.
- Phagocytic cells of the disclosure also include tissue-resident macrophages that not only sense and respond to invading pathogens but are important in tissue development, remodeling, and homeostasis.
- Tissue-resident macrophages originate from embryonic precursors and can self-renew.
- tissue-resident macrophages can include Kupffer macrophages (forms the lining of sinusoids of the liver), alveolar macrophages (lung), microglia macrophages (brain), red pulp macrophages (spleen), and macrophages in the heart.
- tissue-resident macrophages enter tissue during non-inflammatory conditions.
- Macrophages are differentiated from monocytes and function to regulate both innate and adaptive immunity to combat foreign particles including pathogens by stimulating the response of immune cells, such as lymphocytes.
- the putative surface markers for macrophages are CD1 lb (Mac- 1), CD14 (ligand receptor of lipopolysaccharide), CD115 (colony-stimulating factor 1 receptor) and F4/80 (a highly glycosylated proteoglycan extracellular antigen) (Lim, W.F., Inoue-Yokoo, T., Tan, K.S. et al. Hematopoietic cell differentiation from embryonic and induced pluripotent stem cells. Stem Cell Res Ther 4, 71 (2013)).
- a monocyte is a type of white blood cell of the immune system that is capable of phagocytosis.
- Macrophages are derived from blood monocytes that migrate into tissue. One of macrophages’ main functions is to phagocytose microbes and clear cellular debris. Macrophages also play an important role in both the initiation and resolution of inflammation. Macrophages comprise cells typically diffusely scattered in the connective tissue and in liver (Kupffer cells), spleen and lymph nodes (sinus histiocytes), lungs (alveolar macrophages), and central nervous system (microglia) as will be understood by a skilled person.
- Macrophages can also display different responses, ranging from pro-inflammatory to antiinflammatory, depending on the type of stimuli they receive from the surrounding microenvironment.
- Ml and M2 are two major macrophage phenotypes that have been proposed to correlate with extreme macrophage responses.
- Ml pro-inflammatory macrophages are activated upon contact with certain molecules such as lipopolysaccharide (LPS), interferon-gamma (IFN-y), interleukin (IL)-ip, tumor necrosis factor (TNF)-a, and Toll-like receptor engagement.
- LPS lipopolysaccharide
- IFN-y interferon-gamma
- IL interleukin
- TNF tumor necrosis factor
- Toll-like receptor engagement Ml macrophages constitute a potent arm of the immune system deployed to fight infections.
- Ml macrophages secrete pro-inflammatory cytokines and chemokines attracting other types of immune cells and integrating/orchestrating the immune response. Ml activation is induced by IFN-y, TNFa, granulocyte macrophage colony-stimulating factor (GM-CSF), LPS, and other toll-like receptors (TLR) ligands.
- ROS reactive oxygen species
- M2 anti-inflammatory macrophages also known as alternatively activated macrophages, are activated by anti-inflammatory molecules such as IL-4, IL- 13, and IL- 10.
- M2 macrophages exhibit immunomodulatory, tissue repair, and angiogenesis properties which allow them to recruit regulatory T cells to sites of inflammation.
- M2 macrophages do not constitute a uniform population and often are further subdivided into M2a, M2b and M2c categories. The common denominator of all three subpopulations is high IL- 10 production accompanied by low production of IL-12.
- One of their signatures is production of enzyme arginase-1 that depletes L-arginine, thereby suppressing T cell responses and depriving iNOS of its substrate.
- a monocyte is a type of leukocyte or white blood cell capable of phagocytosis and can differentiate into macrophages and myeloid lineage dendritic cells. As a part of the vertebrate innate immune system, monocytes also influence the process of adaptive immunity. There are at least three subclasses of monocytes in human blood based on their phenotypic receptors including CD14 ++ CD 16“ monocyte, CD14 + CD16 ++ monocyte and CD14 ++ CD16 + monocytes, as will be understood by a person skilled in the art. Monocytes serve as precursors for various tissue macrophage and dendritic cell populations and contribute to both protective and pathological immune responses.
- Dendritic cells are specialized antigen-presenting cells capable of phagocytosis that have long outgrowths called dendrites that help to engulf microbes and other invaders. Dendritic cells are present in the tissues that are in contact with the external environment, mainly the skin, the inner lining of the nose, the lungs, the stomach, and the intestines. Once activated, they mature and migrate to the lymphoid tissues where they interact with T cells and B cells to initiate and orchestrate the adaptive immune response. Mature dendritic cells activate T helper cells and cytotoxic T cells. The activated helper T cells interact with macrophages and B cells to activate them in turn. In addition, dendritic cells can influence the type of immune response produced; when they travel to the lymphoid areas where T cells are held, they can activate T cells, which then differentiate into cytotoxic T cells or helper T cells.
- Neutrophils are phagocytes that form the most abundant type of granulocytes and the most abundant type of white blood cells in most mammals as known to a person skilled in the art. Neutrophils are formed from stem cells in the bone marrow and differentiated into subpopulations of neutrophil-killers and neutrophil-eagers.
- a phagocytic cell includes a precursor cell that can develop into and/or be differentiated into a phagocyte.
- precursors or “precursor cells” when used in connection with macrophages, monocytes, dendritic cells, and/or neutrophils indicate parent cells in a cellular lineage resulting in phagocytic cells herein described.
- Exemplary precursor cells include bone marrow-derived cells, stem cells, hematopoietic stem cells, pluripotent stem cells, and other precursor cells identifiable by a person skilled in the art.
- precursor cells can be differentiated by culturing the precursor cells under conditions that promote development of phagocytic cells.
- cytokines that are typically used for differentiation of bone-marrow derived monocytes or hematopoietic stem cells into macrophages include granulocyte-macrophage colony-stimulating factor (GM-CSF) and macrophage colony-stimulating factor (M-CSF).
- GM-CSF granulocyte-macrophage colony-stimulating factor
- M-CSF macrophage colony-stimulating factor
- a source of phagocytic cells such as macrophages, monocytes, dendritic cells, neutrophils, and/or precursors thereof is obtained from a subject.
- the cells can be obtained from a number of sources, including peripheral blood mononuclear cells, bone marrow, lymph node tissue, spleen tissue, umbilical cord, and tumors.
- any number of macrophage, monocyte, dendritic cell, neutrophil, or precursor cell lines available in the art may be used.
- the cells can be obtained from a unit of blood collected from a subject using any number of techniques known to the skilled artisan, such as Ficoll separation.
- Cells can be isolated from peripheral blood by lysing the red blood cells and depleting the lymphocytes and red blood cells, for example, by centrifugation through a PERCOLLTM gradient. Alternatively, cells can be isolated from umbilical cord.
- Cells from the circulating blood of an individual can be obtained by apheresis or leukapheresis.
- the apheresis product typically contains lymphocytes, including T cells, monocytes, granulocytes, B cells, other nucleated white blood cells, red blood cells, and platelets.
- the cells collected by apheresis may be washed to remove the plasma fraction and to place the cells in an appropriate buffer or media, such as phosphate buffered saline (PBS) or a wash solution that lacks calcium and may lack magnesium or may lack many if not all divalent cations, for subsequent processing steps.
- PBS phosphate buffered saline
- wash solution that lacks calcium and may lack magnesium or may lack many if not all divalent cations, for subsequent processing steps.
- the cells may be resuspended in a variety of biocompatible buffers, such as, for example, Ca- free, Mg-free PBS.
- a variety of biocompatible buffers such as, for example, Ca- free, Mg-free PBS.
- the undesirable components of the apheresis sample may be removed and the cells directly resuspended in culture media.
- the concentration of cells and surface can be varied. In some embodiments, it may be desirable to significantly decrease the volume in which beads and cells are mixed together (i.e., increase the concentration of cells), to ensure maximum contact of cells and beads. For example, a concentration of 2 billion cells/ml, 1 billion cells/ml, or greater than 100 million cells/ml can be used. In some embodiments, a concentration of cells of 10, 15, 20, 25, 30, 35, 40, 45, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, or 100 million cells/ml can be used. In some embodiments, concentrations of 125 to 150 million cells/ml can be used. The use of high concentrations of cells can result in increased cell yield, cell activation, and cell expansion.
- a population of cells of the present disclosure can include macrophages, monocytes, dendritic cells, neutrophils, and/or precursors thereof.
- the stem cell When the precursor is a stem cell, such as a hematopoietic stem cell, the stem cell can be contacted with a nucleic acid molecule comprising a polynucleotide sequence encoding the CAR to generate a genetically modified precursor CAR cell. Contacting enables the nucleic acid to be introduced into the precursor. Once introduced into the cell, the nucleic acid comprising a polynucleotide sequence encoding the CAR, can be stably expressed by the cell.
- the precursor that is contacted with the nucleic acid is a hematopoietic stem cell that has been isolated from a human subject.
- the isolated HSCs can be frozen and preserved following isolation by any means known in the art, such as cryopreservation. The cryopreserved cells are then thawed prior to being contacted with a nucleic acid.
- the HSC Prior to being contacted with a nucleic acid comprising a polynucleotide encoding a CAR, the HSC can be within an isolated cell population, such as a population of cells isolated from a subject.
- the isolated cell population can be freshly isolated or can be an isolated population that was previously isolated and has been frozen to preserve the viability of the cells, such as cryopreserved.
- the isolated population can comprise a plurality of HSCs wherein at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or more than 99% CD34+ cells. Upon thawing, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or more than 99% of viable recovered cells are CD34+ cells.
- the nucleic acid can be contacted and introduced comprises introducing a viral vector comprising the polynucleotide into the precursor.
- the viral vector can be a lentiviral vector, a retroviral vector, or an adenoviral vector.
- contacting the precursor with a nucleic acid comprises introducing the nucleic acid into the precursor by transposition or mRNA transfection. In some instances, contacting the precursor with a nucleic acid comprises electroporating the nucleic acid into the precursor.
- the nucleic acid can be contacted and introduced into the precursor or plurality of precursor cells via transduction. Following transduction, at least 40%, at least 50%, at least 60%, at least 70%, at least 80%, at least 90%, or at least 95% of said plurality of genetically modified precursor CAR cells or said plurality of myeloid-derived CAR cells expresses said CAR on the cell surface. Expression of the CAR in the precursor cell(s) can be analyzed at any point in time following transduction. Following CAR transduction, the precursor cell(s) can be expanded. Expansion of the precursor can occur at any point in time that maximizes the recovery of genetically modified CAR cells. For example, expansion can occur 1 day, 2 days, 3 days, 4 days, or 5 days following transduction.
- Expansion can also occur is a shorter amount of time following transduction, such as 2 hours, 4 hours, 6 hours, 8 hours, 10 hours, 12 hours, 14 hours, 16 hours, 18 hours, or 20 hours following CAR transduction.
- contacting a precursor with a nucleic acid molecule comprising a polynucleotide sequence encoding the CAR occurs within a precursor media described herein.
- the precursor media comprises a precursor cytokine mixture.
- the precursor media do not comprise a precursor cytokine mixture as disclosed herein, when contacting a precursor with a nucleic acid molecule comprising a polynucleotide sequence encoding the CAR occurs within a precursor media, the precursor media does not comprise a precursor cytokine mixture.
- the precursor media can be removed.
- precursor media is removed using a centrifugation process.
- multiple rounds of washing and centrifugation are utilized to remove the precursor media following CAR transduction.
- a viral vector is used to introduce a CAR into the precursor cell
- multiple rounds of washing and centrifugation can be utilized to remove the viral vector and precursor media.
- the genetically modified precursor CAR cells which have been removed from contact with precursor media, are suspended in precursor media comprising a precursor cytokine mixture to expand the cells.
- the precursor is expanded in a precursor media comprising a precursor cytokine mixture as disclosed herein.
- the precursor can be expanded to generate a population wherein at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of said plurality of genetically modified myeloid-derived CAR cells expressing said CAR are CD1 lb+ or CD14+ or Cl lb+CD14+.
- a vector may be used to introduce a CAR described herein into a precursor cell (e.g., a hematopoietic stem cell or other bone marrow-derived cell).
- a vector comprising a nucleic acid sequence encoding a CAR as described herein.
- the vector comprises a plasmid vector, viral vector, retrotransposon (e.g. piggyback, sleeping beauty), site directed insertion vector (e.g. CRISPR, Zn finger nucleases, TALEN), suicide expression vector, or other known vector in the art.
- nucleic acid typically achieved by operably linking a nucleic acid or portions thereof to a promoter, and incorporating the construct into an expression vector.
- the vector is one generally capable of replication in a mammalian cell, and/or also capable of integration into the cellular genome of the mammal. Typical vectors contain transcription and translation terminators, initiation sequences, and promoters useful for regulation of the expression of the desired nucleic acid sequence.
- the nucleic acid can be cloned into any number of different types of vectors.
- the nucleic acid can be cloned into a vector including, but not limited to a plasmid, a phagemid, a phage derivative, an animal virus, and a cosmid.
- Vectors of particular interest include expression vectors, replication vectors, probe generation vectors, and sequencing vectors.
- a vector is used as a gene delivery vehicle to transfer a gene into a cell.
- Expression vectors can also include gene delivery nanomaterial such as polymeric nanoparticles or liposomes, and others identifiable by a person skilled in the art.
- the expression vector may be provided to a cell in the form of a viral vector.
- Viral vector technology is well known in the art and is described, for example, in Sambrook et al., 2012, MOLECULAR CLONING: A LABORATORY MANUAL, volumes 1 -4, Cold Spring Harbor Press, NY), and in other virology and molecular biology manuals.
- Viruses, which are useful as vectors include, but are not limited to, retroviruses, adenoviruses, adeno- associated viruses, herpes viruses, and lentiviruses.
- Vectors including those derived from retroviruses such as lentivirus, are suitable tools to achieve long-term gene transfer since they allow long-term, stable integration of a transgene and its propagation in daughter cells.
- lentiviral vector refers to a vector derived from at least a portion of a lentivirus genome, including especially a self-inactivating lentiviral vector as described in, e.g., Milone et al. (2009) Molecular therapy 17(8): 1453-1464.
- Other examples of lentivirus vectors include the LENTIVECTOR® gene delivery technology from Oxford BioMedica, the LENTIMAXTM vector system from Lentigen and the like.
- Lentiviral vectors have the added advantage over vectors derived from onco-retroviruses, such as murine leukemia viruses, in that they can transduce non-proliferating cells, such as hepatocytes. They also have the added advantage of resulting in low immunogenicity in the subject into which they are introduced.
- adeno-associated viral vectors can be used to deliver a nucleic acid (e.g., encoding a CAR of the disclosure) to a stem cell.
- AAVs are nonenveloped, single-stranded DNA viruses of the Dependoparvovirus genus of the Parvoviridae family. AAVs are innately nonpathogenic, poorly immunogenic, and broadly tropic, making them attractive gene delivery candidates.
- AAV vectors have shown to stably transfect mammalian cells without integration into the target genome.
- Exemplary suitable AAVs comprise AAVs of various serotypes that can be used as vectors for carrying genes.
- AAV serotypes are identified based on their interacting glycan moieties that mediate the initial attachment of AAVs to the cell surface.
- AAV serotypes include AAV serotype 1 (“AAV1”), AAV2, AAV3, AAV5, AAV6, AAV9 and other serotypes identifiable to a person skilled in the art such as AAV7, AAV8, AAV11, and AAV-DJ.
- a suitable vector contains an origin of replication functional in at least one organism, a promoter sequence, convenient restriction endonuclease sites, and one or more selectable markers, (e.g., WO 01/96584; WO 01/29058; and U.S. Pat. No. 6,326,193). Additional regulatory regions, e.g., enhancers, regulate the frequency of transcriptional initiation. Typically, these are located in the region 30-110 bp upstream of the start site, although a number of promoters have recently been shown to contain functional elements downstream of the start site as well. The spacing between promoter elements frequently is flexible, so that promoter function is preserved when elements are inverted or moved relative to one another.
- selectable markers e.g., WO 01/96584; WO 01/29058; and U.S. Pat. No. 6,326,193
- Additional regulatory regions e.g., enhancers, regulate the frequency of transcriptional initiation. Typically, these are located in the region 30-110 bp upstream
- regulatory sequence or “regulatory regions” as described herein indicate a segment of a nucleic acid molecule which is capable of increasing or decreasing transcription or translation of a gene within an organism either in vitro or in vivo.
- Regulatory regions of a gene herein described comprise promoters, transcription factor binding sites, binding site operators, activator binding sites, protein-protein binding domains, RNA binding domains, DNA binding domains, repressors, enhancers, insulators, silencers and additional regulatory regions that can alter gene expression in response to developmental and/or external stimuli as will be recognized by a person skilled in the art.
- nucleic acid molecules of the present disclosure includes phagocyte regulatory regions that control expression of a gene in a phagocyte.
- a phagocyte promoter includes a nucleotide sequence that drives or regulates expression in phagocytes. Promoters specific to the mononuclear phagocyte system (MPS) including macrophages, neutrophils, dendritic cells, and osteoclasts will constitute phagocyte promoters. Examples of such promoters include CSF-1 promoter, CD68, CDl lc, DC-SIGN, DC-STAMP, langerin, human neutrophil elastase, and any synthetic promoter containing elements of the phagocyte system designed to achieve high level of expression in phagocytic cells.
- MPS mononuclear phagocyte system
- a constitutive promoter includes an unregulated promoter that allows for continual transcription of its associated genes.
- An example of a strong constitutive promoter is the immediate early cytomegalovirus (CMV) promoter sequence. This promoter is capable of driving high levels of expression of any polynucleotide sequence operatively linked thereto.
- CMV immediate early cytomegalovirus
- constitutive promoter sequences may also be used, including a simian virus 40 (SV40) early promoter, a mouse mammary tumor virus (MMTV) promoter, a human immunodeficiency virus (HIV) long terminal repeat (LTR) promoter, a MoMuLV promoter, an avian leukemia virus promoter, an Epstein-Barr virus immediate early promoter, a Rous sarcoma virus promoter, an elongation factor- la promoter, PGK1 promoter from a phosphoglycerate kinase gene, as well as human gene promoters including an actin promoter, a ubiquitin promoter, a myosin promoter, a hemoglobin promoter, and a creatine kinase promoter.
- Other constitutive promoters are identifiable to one of skill in the art.
- conditional or inducible promoters can also be suitable in the present disclosure.
- a conditional or inducible promoter includes a promoter with activity regulatable or controlled by endogenous transcription factors or exogenous inputs such as chemical compounds, thermal inducers, or optical induction.
- inducible promoters include a metallothionine promoter, a glucocorticoid promoter, a progesterone promoter, a tetracycline promoter, and a Lac promoter.
- the expression vector to be introduced into a precursor cell or phagocytic cell can also contain either a selectable marker gene or a reporter gene or both to facilitate identification and selection of cells expressing a nucleic acid sequence of interest (e.g., a CAR described herein) from the population of phagocytic cells transfected or infected through viral vectors.
- the selectable marker may be carried on a separate piece of DNA and used in a co-transfection procedure. Both selectable marker genes and reporter genes may be flanked with appropriate regulatory sequences to enable expression in the host cells.
- Useful selectable markers include, for example, antibiotic-resistance genes, such as neomycin and the like.
- Reporter genes are used for identifying potentially transfected cells and for evaluating the functionality of regulatory sequences.
- a reporter gene is a gene that is not present in or expressed by the recipient organism or tissue and that encodes a polypeptide whose expression is manifested by some easily detectable property, e.g., enzymatic activity. Expression of the reporter gene is assessed at a suitable time after the DNA has been introduced into the recipient cells.
- Suitable reporter genes may include genes encoding luciferase, beta-galactosidase, chloramphenicol acetyl transferase, secreted alkaline phosphatase, or the green fluorescent protein gene (e.g., Ui-Tei et al., 2000 FEBS Letters 479: 79-82).
- Suitable expression systems are well known and may be prepared using known techniques or obtained commercially.
- the construct with the minimal 5' flanking region showing the highest level of expression of reporter gene is identified as the promoter.
- Such promoter regions may be linked to a reporter gene and used to evaluate agents for the ability to modulate promoter-driven transcription.
- an expression vector of the disclosure comprises a gene encoding a CAR as described herein and appropriate regulatory elements such as promoters, enhancers, and post- transcriptional and post-translational regulatory sequences that are compatible with the phagocytic cell expressing the gene encoding a CAR as would be understood by a skilled person.
- the CAR expression vector is configured for genomic insertion allowing long-term overexpression of the CAR in a precursor cell (e.g., a hematopoietic stem cell or bone marrow-derived cell) .
- the genomic insertion can be achieved by stable transfection.
- lentiviral transduction is preferred over physical or chemical transfections or adenoviral transduction.
- lentiviral transduction is expected to be used effectively in vivo for delivery of a gene encoding a CAR of the disclosure and will permit stable expression in dividing and non-dividing cells.
- a heterologous nucleic acid comprising a polynucleotide sequence encoding a CAR of the disclosure can be introduced into the genome of a phagocytic cell using transposases or targeted nucleases (e.g., Zinc finger nucleases (ZFN), meganucleases, or transcription activator-like effector (TALE) nucleases (TALENs), clustered regularly-interspaced short palindromic repeats associated nucleases (CRISPR/Cas)).
- transposases or targeted nucleases e.g., Zinc finger nucleases (ZFN), meganucleases, or transcription activator-like effector (TALE) nucleases (TALENs), clustered regularly-interspaced short palindromic repeats associated nucleases (CRISPR/Cas)
- the CRISPR/Cas system is a genome editing tool discovered in prokaryotic cells.
- the system includes a CRISPR associated (Cas) protein (e.g., Cas9) capable of modify nucleic acids when complexed with a guide RNA, a guide RNA comprising a CRISPR RNA (crRNA) that binds and directs the Cas protein to the target nucleic acid, optionally a trans-activating CRISPR RNA (tracrRNA, that hybridizes to the crRNA and forms an active complex with the Cas protein), and optionally a homology dependent repair (HDR) template that guides the cellular repair process, allowing insertion of a specific nucleic acid sequence.
- Cas CRISPR associated protein
- crRNA CRISPR RNA
- tracrRNA trans-activating CRISPR RNA
- HDR homology dependent repair
- an HDR template comprising a polynucleotide sequence encoding a CAR of the disclosure is included.
- a CRISPR/Cas system is typically transfected into a target cell (e.g., a phagocytic cell to be genetically modified) by means of one or more plasmids.
- CRISPR/Cas systems are described in, e.g., US8697359, US8771945, US8795965, US8865406, US8871445, US8889356, US8889418, US8895308, US8906616, US8932814, US8945839, US8993233 and US8999641 and applications related thereto; and WO2014/018423, WO2014/093595, WO2014/093622, WO2014/093635, WO2014/093655, WO20 14/093661, WO2014/093694, WO2014/093701, WO2014/093709, WO2014/093712, WO2014/093718, WO2014/145599, WO2014/204723, WO2014/204724, WO2014/204725, WO20 14/204726, WO2014/204727, WO2014/204728, WO2014/204729, WO2015/065964, WO2015/089351,
- a ZFN is an artificial restriction enzyme which is generated by combining a zinc finger DNA- binding domain with a DNA-cleavage domain.
- a zinc finger domain can be engineered to target specific DNA sequences which allows a zinc-finger nuclease to target desired sequences within genomes.
- the DNA-binding domains of individual ZFNs typically contain a plurality of individual zinc finger repeats and can each recognize a plurality of base pairs.
- the most common method to generate new zinc-finger domains is to combine smaller zinc-finger "modules" of known specificity.
- the most common cleavage domain in ZFNs is the non-specific cleavage domain from the type II restriction endonuclease Fokl.
- a cell e.g., a phagocytic cell
- ZFNs can be used to insert the CAR expression cassette into the cell genome.
- the HR machinery searches for homology between the damaged chromosome and the DNA template, and then copies the sequence of the template between the two broken ends of the chromosome, whereby the DNA template is integrated at the target nucleic acid sequence.
- ZFNs are described in, e.g., US 6,534,261; US 6,607,882; US 6,746,838; US 6,794,136; US 6,824,978; 6,866,997; US 6,933,113; 6,979,539; US 7,013,219; US 7,030,215; US 7,220,719; US 7,241,573; US 7,241,574; US 7,585,849; US 7,595,376; US 6,903,185; US 6,479,626; US 2003/0232410 and US 2009/0203140 as well as Gaj et al., Nat Methods, 2012, 9(8):805-7; Ramirez et al., Nucl Acids Res, 2012, 40(12):5560-8; Kim et al., Genome Res, 2012, 22(7): 1327-33; Urnov etal., Nature Reviews Genetics, 2010, 11 :636-646; Miller, et al.
- TALENs are restriction enzymes that can be engineered to cut specific sequences of DNA. TALEN system operates on almost the same principle as ZFNs. They are generated by combining a TALE DNA-binding domain with a DNA cleavage domain.
- TALEs are composed of 33-34 amino acid repeating motifs with two variable positions that have a strong recognition for specific nucleotides. By assembling arrays of these TALEs, the TALE DNA-binding domain can be engineered to bind desired DNA sequence, and thereby guide the nuclease to cut at specific locations in genome.
- TALENs are described in, e.g., US 8,440,431; US 8,440,432; US 8,450,471; US 8,586,363; and US 8,697,853; as well as Joung and Sander, Nat Rev Mol Cell Biol, 2013, 14(1) :49-55 ; Beurdeley et al., Nat Commun, 2013, 4: 1762; Scharenberg et al., Curr Gene Ther, 2013, 13(4):291-303; Gaj et al., Nat Methods, 2012, 9(8):805-7; Miller, et al. Nature biotechnology 29, 143-148 (2011); Christian, et al. Genetics 186, 757-761 (2010); Boch, et al. Science 326, 1509-1512 (2009); and Moscou, & Bogdanove, Science 326, 1501 (2009).
- Meganucleases are rare-cutting endonucleases that generate double-strand breaks (DSB) in a nucleic acid and recognize sequences larger than 12 base pairs. In the wild, such endonucleases are essentially represented by homing endonucleases (Chevalier and Stoddard (2001) Nucleic Acids Research 29:3757-3774). Homing endonucleases are found in fungi, algae, eubacteria and archae, and are often encoded in mobile genetic elements. Their cleavage activities initiate the spreading of these mobile elements by homologous recombination.
- HO and I-Scel have been used to induce homologous gene targeting in yeast, in cultured mammalian cells, and in plants. See, e.g., US 5,792,632; US 6,238,924; US 5,792,632; US 5,830,729; US 6,238,924; US 5,792,632; US 6,238,924.
- Meganucleases are further described in, e.g., W02004067753; W02008010009; WO2008149176; W02009013559; W02009059195; WO2017112859; WO2017062439; US 8,119,381; US 8,338,157; and US 8,927,247.
- the disclosure includes a method of modifying a precursor, the method comprising: introducing a CAR into the precursor, wherein the CAR comprises: an antigen binding domain; a transmembrane domain; an intracellular signaling domain; and an intracellular co-stimulatory signaling domain having an amino acid sequence having at least 90% sequence identity to a cytoplasmic domain of a herpes virus entry mediator (HVEM) protein set forth as any one of SEQ ID NOs: 7, 1, or 2, or a functional fragment or variant thereof that retains co-stimulatory activity, wherein the modified precursor cell expresses the CAR.
- HVEM herpes virus entry mediator
- introducing the CAR into the precursor cell comprises introducing a nucleic acid sequence comprising a polynucleotide sequence encoding the CAR.
- introducing the nucleic acid molecule comprises transducing the precursor cell with a viral vector comprising the nucleic acid sequence encoding the CAR.
- transformation or “transfection” may be used interchangeably and refer to the introduction of a nucleic acid into a cell. Transformation of a cell may be stable or transient.
- a host cell or host organism may be stably transformed with a polynucleotide/nucleic acid molecule of the disclosure.
- a host cell or host organism may be transiently transformed with a nucleic acid construct of the disclosure.
- Transient transformation in the context of a polynucleotide means that a polynucleotide is introduced into the cell and does not integrate into the genome of the cell.
- Stably introducing a polynucleotide introduced into a cell means that the introduced polynucleotide is stably incorporated into the genome of the cell, and thus the cell is stably transformed with the polynucleotide.
- Stable transformation of a cell means that a nucleic acid molecule is introduced into a cell and integrates into the genome of the cell.
- the integrated nucleic acid molecule is capable of being inherited by the progeny thereof, more particularly, by the progeny of multiple successive generations.
- Stable transformation can also refer to a transgene that is maintained extrachromosomally, for example, as a minichromosome or a plasmid.
- the vector can be readily introduced into a host cell, e.g., mammalian, bacterial, yeast, or insect cell by any method in the art.
- the expression vector can be transferred into a host cell by physical, chemical, or biological means.
- Physical methods for introducing a polynucleotide into a host cell include calcium phosphate precipitation, lipofection, particle bombardment, microinjection, electroporation, and the like.
- Methods for producing cells comprising vectors and/or exogenous nucleic acids are well-known in the art. See, for example, Sambrook et al., 2012, MOLECULAR CLONING: A LABORATORY MANUAL, volumes 1 -4, Cold Spring Harbor Press, NY.
- Nucleic acids can be introduced into target cells using commercially available methods which include electroporation (e.g., Amaxa Nucleofector-II (Amaxa Biosystems, Cologne, Germany), ECM 830 (BTX) (Harvard Instruments, Boston, Mass.), the Gene Pulser II (BioRad, Denver, Colo.), or Multiporator (Eppendort, Hamburg Germany)). Nucleic acids can also be introduced into cells using cationic liposome mediated transfection using lipofection, using polymer encapsulation, using peptide mediated transfection, or using biolistic particle delivery systems such as "gene guns” (see, for example, Nishikawa, et al. Hum Gene Then, 12(8): 861 -70 (2001).
- electroporation e.g., Amaxa Nucleofector-II (Amaxa Biosystems, Cologne, Germany), ECM 830 (BTX) (Harvard Instruments, Boston, Mass.), the Gene Pul
- RNA vectors include vectors having a RNA promoter and/or other relevant domains for production of a RNA transcript.
- Viral vectors, and especially retroviral vectors have become the most widely used method for inserting genes into mammalian, e.g., human cells.
- Other viral vectors may be derived from lentivirus, poxviruses, herpes simplex virus, adenoviruses and adeno-associated viruses, and the like. See, for example, U.S. Pat. Nos. 5,350,674 and 5,585,362.
- Chemical means for introducing a polynucleotide into a host cell include colloidal dispersion systems, such as macromolecule complexes, nanocapsules, microspheres, beads, and lipid-based systems including oil-in-water emulsions, micelles, mixed micelles, and liposomes.
- colloidal dispersion systems such as macromolecule complexes, nanocapsules, microspheres, beads, and lipid-based systems including oil-in-water emulsions, micelles, mixed micelles, and liposomes.
- An exemplary colloidal system for use as a delivery vehicle in vitro and in vivo is a liposome (e.g. , an artificial membrane vesicle).
- an exemplary delivery vehicle is a liposome.
- lipid formulations is contemplated for the introduction of the nucleic acids into a host cell (in vitro, ex vivo or in vivo).
- the nucleic acid may be associated with a lipid.
- the nucleic acid associated with a lipid may be encapsulated in the aqueous interior of a liposome, interspersed within the lipid bilayer of a liposome, attached to a liposome via a linking molecule that is associated with both the liposome and the nucleic acid, entrapped in a liposome, complexed with a liposome, dispersed in a solution containing a lipid, mixed with a lipid, combined with a lipid, contained as a suspension in a lipid, contained or complexed with a micelle, or otherwise associated with a lipid.
- Lipid, lipid/DNA or lipid/expression vector associated compositions are not limited to any particular structure in solution.
- Lipids are fatty substances which may be naturally occurring or synthetic lipids.
- lipids include the fatty droplets that naturally occur in the cytoplasm as well as the class of compounds which contain long-chain aliphatic hydrocarbons and their derivatives, such as fatty acids, alcohols, amines, amino alcohols, and aldehydes.
- Lipids suitable for use can be obtained from commercial sources.
- DMPC dimyristyl phosphatidylcholine
- DCP dicetyl phosphate
- Choi cholesterol
- DMPG dimyristyl phosphatidylglycerol
- Stock solutions of lipids in chloroform or chloroform/methanol can be stored at about -20°C. Chloroform is used as the only solvent since it is more readily evaporated than methanol.
- Liposome is a generic term encompassing a variety of single and multilamellar lipid vehicles formed by the generation of enclosed lipid bilayers or aggregates. Liposomes can be characterized as having vesicular structures with a phospholipid bilayer membrane and an inner aqueous medium.
- Multilamellar liposomes have multiple lipid layers separated by aqueous medium. They form spontaneously when phospholipids are suspended in an excess of aqueous solution. The lipid components undergo self-rearrangement before the formation of closed structures and entrap water and dissolved solutes between the lipid bilayers (Ghosh et al., 1991 Glycobiology 5: 505-10). However, compositions that have different structures in solution than the normal vesicular structure are also encompassed. For example, the lipids may assume a micellar structure or merely exist as nonuniform aggregates of lipid molecules. Also contemplated are lipofectamine-nucleic acid complexes.
- the nucleic acids introduced into a precursor cell can be RNA.
- the RNA is mRNA that comprises in vitro transcribed RNA or synthetic RNA.
- the RNA is produced by in vitro transcription using a PCR-generated template.
- DNA of interest from any source can be directly converted by PCR into a template for in vitro mRNA synthesis using appropriate primers and RNA polymerase.
- the source of the DNA can be, for example, genomic DNA, plasmid DNA, phage DNA, cDNA, synthetic DNA sequence or any other appropriate source of DNA.
- the desired template for in vitro transcription is a polynucleotide sequence encoding a CAR having an intracellular co-stimulatory signaling domain having an amino acid sequence having at least 90% sequence identity to a cytoplasmic domain of a herpes virus entry mediator (HVEM) protein set forth as any one of SEQ ID NOs: 1, 2, or 3, or a functional fragment or variant thereof that retains co-stimulatory activity.
- HVEM herpes virus entry mediator
- PCR can be used to generate a template for in vitro transcription of mRNA which is then introduced into cells.
- Methods for performing PCR are well known in the art.
- Primers for use in PCR are designed to have regions that are substantially complementary to regions of the DNA to be used as a template for the PCR.
- “Substantially complementary”, as used herein, refers to sequences of nucleotides where a majority or all of the bases in the primer sequence are complementary, or one or more bases are non-complementary, or mismatched. Substantially complementary sequences are able to anneal or hybridize with the intended DNA target under annealing conditions used for PCR.
- the primers can be designed to be substantially complementary to any portion of the DNA template.
- the primers can be designed to amplify the portion of a gene that is normally transcribed in cells (the open reading frame), including 5' and 3' UTRs.
- the primers can also be designed to amplify a portion of a gene that encodes a particular domain of interest.
- the primers are designed to amplify the coding region of a human cDNA, including all or portions of the 5' and 3' UTRs.
- Primers useful for PCR are generated by synthetic methods that are well known in the art.
- "Forward primers" are primers that contain a region of nucleotides that are substantially complementary to nucleotides on the DNA template that are upstream of the DNA sequence that is to be amplified.
- Upstream is used herein to refer to a location 5' to the DNA sequence to be amplified relative to the coding strand.
- reverse primers are primers that contain a region of nucleotides that are substantially complementary to a double-stranded DNA template that are downstream of the DNA sequence that is to be amplified.
- Downstream is used herein to refer to a location 3' to the DNA sequence to be amplified relative to the coding strand.
- the RNA preferably has 5' and 3' UTRs.
- the 5' UTR is between zero and 3000 nucleotides in length.
- the length of 5' and 3' UTR sequences to be added to the coding region can be altered by different methods, including designing primers for PCR that anneal to different regions of the UTRs. Using this approach, one of ordinary skill in the art can modify the 5' and 3' UTR lengths required to achieve optimal translation efficiency following transfection of the transcribed RNA.
- the 5' and 3' UTRs can be the naturally occurring, endogenous 5' and 3' UTRs for the gene of interest.
- UTR sequences that are not endogenous to the gene of interest can be added by incorporating the UTR sequences into the forward and reverse primers or by any other modifications of the template.
- the use of UTR sequences that are not endogenous to the gene of interest can be useful for modifying the stability and/or translation efficiency of the RNA. For example, it is known that AU-rich elements in 3' UTR sequences can decrease the stability of mRNA. Therefore, 3' UTRs can be selected or designed to increase the stability of the transcribed RNA based on properties of UTRs that are well known in the art.
- the 5' UTR can contain the Kozak sequence of the endogenous gene.
- a consensus Kozak sequence can be redesigned by adding the 5' UTR sequence.
- Kozak sequences can increase the efficiency of translation of some RNA transcripts, but does not appear to be required for all RNAs to enable efficient translation. The requirement for Kozak sequences for many mRNAs is known in the art.
- the 5' UTR can be derived from an RNA virus whose RNA genome is stable in cells. Various nucleotide analogues can be used in the 3' or 5' UTR to impede exonuclease degradation of the mRNA.
- a promoter of transcription should be attached to the DNA template upstream of the sequence to be transcribed.
- the RNA polymerase promoter becomes incorporated into the PCR product upstream of the open reading frame that is to be transcribed.
- the promoter is a T7 polymerase promoter.
- Other useful promoters include T3 and SP6 RNA polymerase promoters. Consensus nucleotide sequences for T7, T3, and SP6 promoters are known in the art.
- the mRNA has both a cap on the 5' end and a 3' poly(A) tail which determine ribosome binding, initiation of translation and stability mRNA in the cell.
- phage T7 RNA polymerase can extend the 3' end of the transcript beyond the last base of the template (Schenborn and Mierendorf, Nuc Acids Res., 13 :6223-36 (1985); Nacheva and Berzal- Herranz, Eur. J. Biochem., 270: 1485-65 (2003)).
- the poly A/T segment of the transcriptional DNA template can be produced during PCR by using a reverse primer containing a polyT tail, such as 100T tail (size can be 50-5000 T), or after PCR by any other method, including DNA ligation or in vitro recombination.
- Poly(A) tails also provide stability to RNAs and reduce their degradation. Generally, the length of a poly(A) tail positively correlates with the stability of the transcribed RNA. In some embodiments, the poly(A) tail is between 100 and 5000 adenosines.
- Poly(A) tails of RNAs can be further extended following in vitro transcription with the use of a poly(A) polymerase, such as E. coli poly A polymerase (E-PAP).
- E-PAP E. coli poly A polymerase
- increasing the length of a poly(A) tail from 100 nucleotides to between 300 and 400 nucleotides results in about a two-fold increase in the translation efficiency of the RNA.
- the attachment of different chemical groups to the 3' end can increase mRNA stability. Such attachment can contain modified/artificial nucleotides, aptamers and other compounds.
- ATP analogs can be incorporated into the poly(A) tail using poly(A) polymerase. ATP analogs can further increase the stability of the RNA.
- 5' caps also provide stability to RNA molecules.
- the 5' cap is provided using techniques known in the art and described in, e.g., Cougot et al. Trends in Biochem. Sci. 29:436-444 (2001); Stepinski et al. RNA 7: 1468-95 (2001); and Elango et al. Biochim.
- the RNAs produced by the methods disclosed herein can also contain an internal ribosome entry site (IRES) sequence.
- IRES sequence may be any viral, chromosomal or artificially designed sequence which initiates cap-independent ribosome binding to mRNA and facilitates the initiation of translation. Any solutes suitable for cell electroporation, which can contain factors facilitating cellular permeability and viability such as sugars, peptides, lipids, proteins, antioxidants, and surfactants can be included.
- IVT-RNA vectors are known in the literature which are utilized in a standardized manner as template for in vitro transcription and which have been genetically modified in such a way that stabilized RNA transcripts are produced.
- protocols used in the art are based on a plasmid vector with the following structure: a 5' RNA polymerase promoter enabling RNA transcription, followed by a gene of interest which is flanked either 3' and/or 5' by untranslated regions (UTR), and a 3' polyadenyl cassette containing 50-70 A nucleotides.
- UTR untranslated regions
- the circular plasmid Prior to in vitro transcription, the circular plasmid is linearized downstream of the polyadenyl cassette by type II restriction enzymes (recognition sequence corresponds to cleavage site).
- Nucleic acids can be delivered into cells by electroporation.
- the nucleic acid includes an RNA. See, e.g., the formulations and methodology of electroporation of nucleic acid constructs into mammalian cells as taught in, e.g., US 2004/0014645, US 2005/0052630A1, US 2005/0070841, US 2004/0059285, and US 2004/0092907.
- the various parameters including electric field strength required for electroporation of any known cell type are generally known in the relevant research literature as well as numerous patents and applications in the field. See e.g., U.S. Pat. No. 6,678,556, U.S. Pat. No. 7,171,264, and U.S. Pat. No.
- Electroporation may also be utilized to deliver nucleic acids into cells in vitro utilizing any of the many available devices and electroporation systems known to those of skill in the art. Electroporation of cells in vitro is described, e.g., in US 2007/0128708.
- a chimeric antigen receptor to be expressed in a precursor, such as a bone marrow-derived cell or a stem cell, wherein the CAR comprises an antigen binding domain; a transmembrane domain; an intracellular signaling domain; and an intracellular costimulatory signaling domain.
- the CAR comprises an intracellular co-stimulatory signaling domain having an amino acid sequence having at least 90% sequence identity to a cytoplasmic domain of a herpes virus entry mediator (HVEM) protein set forth as any one of SEQ ID NOs: 1, 2, or 3, or a functional fragment or variant thereof that retains co-stimulatory activity.
- HVEM herpes virus entry mediator
- a chimeric antigen receptor is an artificial cell surface receptor that is engineered to be expressed on an immune effector cell and specifically bind an antigen.
- a CAR typically comprises an antigen binding domain and a signal transduction domain capable of mimicking the T cell receptor-mediated signaling pathway.
- a “signaling pathway” or “signal transduction pathway” refers to the biochemical relationship between a variety of signal transduction molecules that play a role in the transmission of a signal from one portion of a cell to another portion of a cell.
- the phrase "cell surface receptor” includes molecules and complexes of molecules capable of receiving a signal and transmitting the signal across the plasma membrane of a cell.
- Antigen binding domains that are used in CARs include a natural ligand and the single-chain variable region of an antibody to a target molecule.
- the advantage of a CAR is that it can recognize a defined target without requiring antigen-processing or the major histocompatibility complex (MHC)-restricted antigen presentation, allowing the use of T cells expressing CAR (CAR-T cells) for adoptive immunotherapy in a wide range of patients (Doth et al, Immunol. Rev. 257(1): 107 (2014)). T cells expressing the first generation CAR having CD3z as the signal transduction domain often become anergic and fail to elicit a potent immune response (Kershaw et al, Clin. Cancer Res.
- the CAR can help immune cells such as macrophages find and kill cancer cells that have the specific protein the receptor is designed to bind.
- a precursor cell such as a stem cell can be removed from the bone-marrow or blood of a patient and modified to express a CAR.
- hematopoietic cells can be removed from the bone marrow and genetically modified to express a CAR specific to a particular form of antigen on tumor cells, and then expanded and differentiated into a myeloid-derived CAR (e.g., a monocyte or macrophage) using the methods described herein.
- the precursor cell e.g., hematopoietic stem cell
- expressing the CAR can then be differentiated into a cell within a specific cell lineage.
- the hematopoietic stem cell (HSC) expressing a CAR can be differentiated into a cell within the myeloid lineage, such as, for example, a monocyte or macrophage.
- the HSC CAR cell can also be differentiated into a parent cell of a monocyte, such as a myeloid progenitor cell.
- the stem cell i.e., hematopoietic stem cell
- the CAR is retained on the surface of the newly differentiated cell.
- CARs can comprise an antigen binding domain, a transmembrane domain, and an intracellular domain comprising a co-stimulatory signaling domain of a herpes virus entry mediator (HVEM) protein as described herein.
- HVEM herpes virus entry mediator
- a CAR expressed by a modified stem cell, or myeloid cell derived from a stem cell (e.g., a HSC differentiated into a modified phagocytic cell) of the disclosure can comprise an antigen binding domain that binds to an antigen on a target cell.
- cell surface markers that may act as an antigen that binds to the antigen binding domain of the CAR include those associated with viral, bacterial and parasitic infections, autoimmune disease, degenerative disease, and cancer cells.
- the antigen binding domain can include any domain that binds to the antigen and may include, but is not limited to, a monoclonal antibody, a polyclonal antibody, a synthetic antibody, a human antibody, a humanized antibody, a non-human antibody, and any fragment thereof.
- the antigen binding domain portion comprises a mammalian antibody or a fragment thereof.
- the antigen binding domain comprises a monovalent antibody fragment.
- the monovalent antibody fragment can comprise a single chain variable fragment (scFv) or a Fab fragment.
- the monovalent antibody fragment has a molecular weight of about 25 to about 30 kDa (or about 25 kDa, about 26 kDa, about 27 kDa, about 28 kDa, about 29 kDa, or 30 kDa).
- the monovalent antibody fragment can have a VH and VL domain connected in either orientation by a flexible linker (e.g., VL-linker-VH or VH-linker-VL).
- the flexible linker typically comprises 10 to about 25 amino acids (e.g., glycine to confer flexibility and/or serines and/or threonines for improved solubility).
- a Gly-Ser linker may be used.
- Further suitable linkers are described, e.g., in Alfthan, K. Properties of a single-chain antibody containing different linker peptides. Protein Engineering 1995, vol. 8, no. 7, p. 725-731, which is incorporated by reference in its entirety.
- the antigen binding domain is derived from the same species in which the CAR will ultimately be used in.
- the antigen binding domain of the CAR can comprise a human antibody, a humanized antibody, or a fragment thereof.
- the antigen binding domain is operably linked to another domain of the CAR, such as the transmembrane domain or the intracellular domain, for expression in the cell.
- a polynucleotide sequence encoding the antigen binding domain is operably linked at its 3’ end to a polynucleotide sequence encoding a transmembrane domain and a polynucleotide sequence encoding an intracellular domain.
- a nucleic acid molecule comprising a polynucleotide sequence encoding a CAR can be contacted with a precursor, such as a stem cell, to introduce the CAR into the cell, generating a stem cell modified to express a CAR.
- the nucleic acid molecule introduced into the stem cell can comprise multiple polynucleotide sequences encoding multiple domains of a CAR.
- antigen binding domain depends upon the type and number of antigens that are present on the surface of a target cell.
- the antigen binding domain may be chosen to recognize an antigen that acts as a cell surface marker on a target cell associated with a particular disease state.
- the antigen binding domain binds to a tumor antigen, such as an antigen that is specific for a tumor or cancer of interest.
- the tumor antigen can comprise one or more antigenic cancer epitopes.
- the antigen binding domain targets an antigen present on the surface of a viral particle.
- viral particles include influenza virus, equine infectious anemia virus, simian immunodeficiency virus (SIV), human immunodeficiency virus (HIV), lassa fever virus, herpes simplex virus, varicella zoster virus, cytomegalovirus, epstein-barr virus, variola virus, adeno virus, papilloma virus, parvo virus, measles virus, mumps virus, respiratory syncytial virus, para influenza virus, corona virus, rubella virus, rabies virus, human T-cell lymphotropic virus, picoma virus, hepa DNA virus, flavivirus, deltavirus, calicivirus, polio virus, zika virus, west nile virus, SARS, rubella, norovirus, human papillomavirus, malaria, human T-lymphotropic virus, and/or helicobacter pylori.
- Exemplary viral antigens include any surface protein and/or polypeptide present on the surface of the above listed viral particles.
- surface proteins and/or polypeptides include: Zika capsid protein (C), Zika envelope protein (E), Zika precursor membrane protein (PrM), WNV glycoprotein E, WNV small membrane protein M, VZV glycoprotein E (gE), VZV gB, VZV gH, CoV nucleocapsid (N), CoV envelope (E), CoV membrane (M), Rubella El, Rubella E2, Norovirus Group VP1, HPV 11, HPV capsid protein LI, HPV capsid protein L2, Lassa Fever Virus GP1, Lassa Fever Virus GP2, influenza neuraminidase (NA, N1 to N11), influenza hemagglutinin (HA, Hl to Hl 8), HTLV-1 Envelope, HTLV-1 gp21, HTLV-1 mosaic, HIV gpl20, HIV gp41, he
- a myeloid-derived cell of the disclosure can also be modified to express additional targeting ligands on the surface of the myeloid-derived cell in addition to the antigen binding domain of a CAR described herein.
- Additional targeting ligands comprise molecules configured to associate with any molecule presented on a target cell of interest, such as a target associated with an organ, a tissue, or an extracellular matrix of a target cell of interest.
- additional targeting ligands bind to molecules associated with a particular state of a target cell, such as a cancerous condition.
- An additional targeting ligand can be specific to one target or be configured to bind multiple target molecules.
- Suitable target molecules recognized by an additional target ligand can include a protein (e.g., a receptor, a tumor marker, a transmembrane protein), a nucleic acid (e.g., DNA, RNA), and/or a carbohydrate (e.g., a monosaccharide, disaccharide, or polysaccharide) that is present on the surface of a target cell.
- exemplary targeting ligands include an RGD-containing peptide, a small molecule (e.g., a peptide) mimetic ligand, and an antibody or antibody fragment specific for a particular target.
- a CAR expressed by a modified myeloid-derived cell of the disclosure can comprise a transmembrane domain that connects the antigen binding domain of the CAR to the intracellular domain.
- the transmembrane domain can function to stabilize the CAR as a whole.
- the transmembrane domain can be selected or modified by amino acid substitution to avoid binding of such domains to the transmembrane domains of the same or different surface membrane proteins to minimize interactions with other molecules.
- the transmembrane domain may be derived either from a natural or from a synthetic source. Where the source is natural, the domain may be derived from any membrane-bound or transmembrane protein. Transmembrane regions of particular use in this invention may be derived from (i.e.
- the transmembrane domain can be from any type I transmembrane protein such as CD4, CD28 or HVEM or a functional fragment or variant thereof that retains co-stimulatory activity.
- the transmembrane domain is a CD8 transmembrane domain or a functional fragment or variant thereof. In some embodiments, the transmembrane domain is a CD28 transmembrane domain or a functional fragment or variant thereof.
- a variety of human hinges can be employed as well including the human Ig (immunoglobulin) hinge.
- the transmembrane domain may be a hydrophobic alpha helix that spans across the membrane of the cell (e.g., macrophage).
- the transmembrane domain can be naturally associated with one or more of the domains in the CAR.
- the transmembrane domain may be synthetic, in which case it can comprise predominantly hydrophobic residues such as leucine and valine. A triplet of phenylalanine, tryptophan and valine can be found at each end of a synthetic transmembrane domain.
- a CAR expressed by a modified myeloid-derived cell of the disclosure can comprise an intracellular domain.
- the intracellular (z.e., cytoplasmic) domain of a CAR is a signaling domain that transduces the event of receptor antigen binding to an intracellular signal that contributes to activation and/or transduction of signals in a cell in which the CAR is expressed. In some embodiments, absent appropriate co-stimulatory signals, this event is insufficient for useful cell activation and proliferation.
- Cell activation can include cytokine production, clonal proliferation, differentiation, and survival.
- an intracellular domain for use in the disclosure include the cytoplasmic portion of a surface receptor, co-stimulatory molecule, and any molecule that acts in concert to initiate signal transduction in a myeloid-derived cell, as well as any derivative or variant of these elements and any synthetic sequence that has the same functional capability.
- an intracellular domain examples include a fragment or domain from one or more molecules or receptors including: T cell receptor (TCR), CD3 zeta, CD3 gamma, CD3 delta, CD3 epsilon, CD86, common FcR gamma, FcR beta (Fc Epsilon Rib), CD79a, CD79b, Fcgamma Rlla, DAP10, DAP12, CD27, CD28, 4-1BB (CD137), 0X40, CD30, CD40, PD-1, ICOS, lymphocyte function-associated antigen-1 (LFA-1), CD2, CD7, LIGHT, NKG2C, B7-H3, a ligand that specifically binds with CD83, CDS, ICAM-1, GITR, BAFFR, HVEM (LIGHTR), SLAMF7, NKp80 (KLRF1), CD127, CD160, CD 19, CD4, CD8alpha, CD8beta, IL2R beta, IL2R gam
- the intracellular signaling domain of the CAR includes at least 1, at least 2, at least 3, at least 4, or at least 5 immunoreceptor tyrosine-based activation motifs (IT AMs).
- ITAM immunoreceptor tyrosine-based activation motifs
- ITAM is a conserved protein motif that is generally present in the tail portion of signaling molecules expressed in many immune cells.
- the motif may include two repeats of the amino acid sequence YXXL/I separated by 6-8 amino acids, wherein each X is independently any amino acid, producing the conserved motifs: YXXL/IXXXXXXYXXL/I (SEQ ID NO: 7); YXXL/IXXXXXXYXXL/I (SEQ ID NO: 8); or YXXL/IXXXXXXXXYXXL/I (SEQ ID NO: 9).
- ITAMs within signaling molecules are important for signal transduction within the cell, which is mediated at least in part by phosphorylation of tyrosine residues in the ITAM following activation of the signaling molecule.
- ITAMs may also function as docking sites for other proteins involved in signaling pathways.
- the intracellular signaling domain includes at least 1, at least 2, at least 3, at least 4, or at least 5 IT AMs derived from CD3z, FcRy, Megfl 0, FcRy, and combinations thereof.
- the intracellular domain of a CAR useful in a modified myeloid-derived cell of the disclosure includes any portion of one or more co-stimulatory molecules.
- Co-stimulation is a key event for T cells to exhibit effective effector functions and is mediated by co-stimulatory molecules.
- the co-stimulatory domain of a CAR is essential for promoting the intracellular signal of the T-cell receptor domain to initiate T cell activation and proliferation. Thus, promotion of such a signal can depend upon the selected co-stimulatory signaling domain and/or combinations thereof.
- Second generation CARs incorporate an intracellular co-stimulatory signaling domain in addition to an intracellular signaling domain (e.g., CD3z) and can enhance CAR T cell activation.
- an intracellular signaling domain e.g., CD3z
- Co-stimulatory molecules are divided into two major families; the CD28 family which includes CD28 and ICOS, and the tumor necrosis factor receptor superfamily (TNFRSF) which includes 4- IBB (TNFRSF9), CD27, CD30, DR3, GITR, 0X40, TNFR2 and herpes virus entry mediator (HVEM, TNFRSF14). So far, co-stimulatory domains derived from CD28 or 4-1BB have commonly been used to construct CARs.
- CD28 which includes CD28 and ICOS
- TNFRSF tumor necrosis factor receptor superfamily
- HVEM herpes virus entry mediator
- HVEM another member of the TNFRSF, is the main receptor targeted by the Herpes simplex virus to enter host cells (Sedy et al. (2005) Nature Immunology 6( 1 ) : 90-98). It was previously described as AT AR (Another TRAF- Associated Receptor) (Hsu et al. (1997) Journal of Biological Chemistry 272(21): 13471-13474) and is also known as tumor necrosis factor receptor superfamily member 14 (TNFRSF14).
- AT AR Another TRAF- Associated Receptor
- TNFRSF14 tumor necrosis factor receptor superfamily member 14
- T lymphocyte attenuator a ligand of HVEM interaction with HVEM expressed on CD8 + T cells was also reported to promote survival and memory generation in response to a bacterial infection (Steinberg et al., PLoS One 8(10):e77992 (2013)). Additionally, tumor cells which express anti-HVEM single chain antibody induce a potent proliferation and cytokine production of co-cultured T cells (Park et al, Cancer Immunol. Immunother. 61(2):203 (2012)), suggesting HVEM might serve as a potent co- stimulatory signaling entity in T cells.
- HVEM is expressed in peripheral T and B cells, and in resting T and B cells (Ning et al. (2021) Frontiers in Immunology 12:654960). HVEM is constitutively expressed in naive T cells.
- the HVEM receptor has both stimulatory and inhibitory effects depending on its ligand, and at least five different ligands have been described (Cai & Freeman (2009) Immunological Reviews 229(l):244-258; Del Rio et al.
- HVEM binding to the LIGHT ligand of the TNF family and lymphotoxin alpha (LTa) produces a co- stimulatory signal, leading to increased T cell proliferation in vitro and formation of effector and memory T cells (Del Rio et al. (2010) Journal of Leukocyte Biology 87(2):223-235).
- HVEM can generate an inhibitory signal when bound to BTLA or CD 160 (Pasero & Olive (2013) Immunology Letters 15 l(l-2):71-75).
- the overexpression of BTLA/HVEM on T cells can contribute to T cell exhaustion (Ning et al. (2021) Frontiers in Immunology 12:654960; Shui et al. (2011) Journal of Leukocyte Biology, 89(4), 517-523).
- the cytoplasmic domain of HVEM is 60 amino acids in length and includes an a-helix, a TRAF domain, and a tail (amino acid residues 224-283 of SEQ ID NO: 4; Hennecke, Derek. 2022.
- M83 interacts with adapter proteins, including TRAFs (TRAF5 and TRAF2) (Hsu et al.
- the co-stimulatory signaling domain from an HVEM protein can include all or part of the transmembrane domain along with all or part of the intracellular region or can include only all or part of the intracellular region of the HVEM protein.
- a nucleic acid sequence disclosed herein can encode a HVEM co-stimulatory signaling domain, or functional fragment or variant thereof, of the disclosure, can have at least 80%, 81%, 82%, 83%, 84%, 85%, 86%, 87%, 88%, 89%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or 100% sequence identity to the nucleic acid sequence encoding SEQ ID NO: 3, wherein the encoded domain retains co-stimulatory activity.
- a nucleic acid sequence encoding a HVEM co-stimulatory signaling domain, or functional fragment or variant thereof, of the disclosure has at least 90% sequence identity to the nucleic acid sequence encoding SEQ ID NO: 3, wherein the encoded domain retains co-stimulatory activity. In some embodiments, a nucleic acid sequence encoding a HVEM co-stimulatory signaling domain, or functional fragment or variant thereof, of the disclosure, has at least 95% sequence identity to the nucleic acid sequence encoding SEQ ID NO: 3, wherein the encoded domain retains co-stimulatory activity. In some embodiments, a nucleic acid sequence encoding a HVEM co-stimulatory signaling domain of the disclosure has 100% sequence identity to the nucleic acid sequence encoding SEQ ID NO: 3.
- a HVEM co-stimulatory signaling domain, or functional fragment or variant thereof, of the disclosure can have an amino acid sequence having at least 80%, 81%, 82%, 83%, 84%, 85%, 86%, 87%, 88%, 89%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or 100% sequence identity to the amino acid sequence set forth as SEQ ID NO: 3 and retains co-stimulatory activity.
- a HVEM co-stimulatory signaling domain, or functional fragment or variant thereof, of the disclosure has an amino acid sequence having at least 90% sequence identity to the amino acid sequence set forth as SEQ ID NO: 3 and retains co-stimulatory activity.
- a HVEM co-stimulatory signaling domain, or functional fragment or variant thereof, of the disclosure has an amino acid sequence having at least 95% sequence identity to the amino acid sequence set forth as SEQ ID NO: 3 and retains co-stimulatory activity.
- a HVEM co-stimulatory signaling domain of the disclosure has an amino acid sequence set forth as SEQ ID NO: 3.
- a HVEM co-stimulatory signaling domain can include amino acid residues 224-283 of SEQ ID NO: 4.
- a nucleic acid sequence encoding a HVEM co-stimulatory signaling domain, or functional fragment or variant thereof, of the disclosure has at least 80%, 81%, 82%, 83%, 84%, 85%, 86%, 87%, 88%, 89%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or 100% sequence identity to the nucleic acid sequence encoding SEQ ID NO: 1, wherein the encoded domain retains co-stimulatory activity.
- a nucleic acid sequence encoding a HVEM co- stimulatory signaling domain, or functional fragment or variant thereof, of the disclosure has at least 90% sequence identity to the nucleic acid sequence encoding SEQ ID NO: 1, wherein the encoded domain retains co-stimulatory activity. In some embodiments, a nucleic acid sequence encoding a HVEM co-stimulatory signaling domain, or functional fragment or variant thereof, of the disclosure, has at least 95% sequence identity to the nucleic acid sequence encoding SEQ ID NO: 1, wherein the encoded domain retains co-stimulatory activity. In some embodiments, a nucleic acid sequence encoding a HVEM co-stimulatory signaling domain of the disclosure has 100% sequence identity to the nucleic acid sequence encoding SEQ ID NO: 1.
- a HVEM co-stimulatory signaling domain, or functional fragment or variant thereof, of the disclosure can have an amino acid sequence having at least 80%, 81%, 82%, 83%, 84%, 85%, 86%, 87%, 88%, 89%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or 100% sequence identity to the amino acid sequence set forth as SEQ ID NO: 1 and retains co-stimulatory activity.
- a HVEM co-stimulatory signaling domain, or functional fragment or variant thereof, of the disclosure has an amino acid sequence having at least 90% sequence identity to the amino acid sequence set forth as SEQ ID NO: 1 and retains co-stimulatory activity.
- a HVEM co-stimulatory signaling domain, or functional fragment or variant thereof, of the disclosure has an amino acid sequence having at least 95% sequence identity to the amino acid sequence set forth as SEQ ID NO: 1 and retains co-stimulatory activity.
- a HVEM co-stimulatory signaling domain of the disclosure has an amino acid sequence set forth as SEQ ID NO: 1.
- a HVEM co-stimulatory signaling domain can include amino acid residues 201-283 of SEQ ID NO: 4.
- a nucleic acid sequence encoding a HVEM co-stimulatory signaling domain, or functional fragment or variant thereof, of the disclosure can have at least 80%, 81%, 82%, 83%, 84%, 85%, 86%, 87%, 88%, 89%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or 100% sequence identity to the nucleic acid sequence encoding SEQ ID NO: 2, wherein the encoded domain retains co- stimulatory activity.
- a nucleic acid sequence encoding a HVEM co-stimulatory signaling domain, or functional fragment or variant thereof, of the disclosure has at least 90% sequence identity to the nucleic acid sequence encoding SEQ ID NO: 2, wherein the encoded domain retains co-stimulatory activity.
- a nucleic acid sequence encoding a HVEM co- stimulatory signaling domain, or functional fragment or variant thereof, of the disclosure has at least 95% sequence identity to the nucleic acid sequence encoding SEQ ID NO: 2, wherein the encoded domain retains co-stimulatory activity.
- a nucleic acid sequence encoding a HVEM co-stimulatory signaling domain of the disclosure has 100% sequence identity to the nucleic acid sequence encoding SEQ ID NO: 2.
- a HVEM co-stimulatory signaling domain, or functional fragment or variant thereof, of the disclosure can have an amino acid sequence having at least 80%, 81%, 82%, 83%, 84%, 85%, 86%, 87%, 88%, 89%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or 100% sequence identity to the amino acid sequence set forth as SEQ ID NO: 2 and retains co-stimulatory activity.
- a HVEM co-stimulatory signaling domain, or functional fragment or variant thereof, of the disclosure has an amino acid sequence having at least 90% sequence identity to the amino acid sequence set forth as SEQ ID NO: 2 and retains co-stimulatory activity.
- a HVEM co-stimulatory signaling domain, or functional fragment or variant thereof, of the disclosure has an amino acid sequence having at least 95% sequence identity to the amino acid sequence set forth as SEQ ID NO: 2 and retains co-stimulatory activity.
- a HVEM co-stimulatory signaling domain of the disclosure has an amino acid sequence set forth as SEQ ID NO: 2.
- a HVEM co-stimulatory signaling domain can include amino acid residues 210-277 of SEQ ID NO: 4.
- a HVEM co-stimulatory signaling domain having co-stimulatory activity can bind a TRAF1, a TRAF2, a TRAF3, and/or a TRAF5 intracellular signaling adapters. In some embodiments, a HVEM co-stimulatory signaling domain having co-stimulatory activity does not bind a TRAF6 intracellular signaling adapter.
- the HVEM co-stimulatory signaling domain can promote functional activity in the genetically modified myeloid-derived CAR cells disclosed herein.
- An intracellular signaling domain of the present disclosure can include a CD3 zeta intracellular signaling domain and an HVEM co-stimulatory signaling domain as described herein.
- spacer generally means any oligo- or polypeptide that functions to link the transmembrane domain to, either the antigen binding domain or, the intracellular domain in the CAR polypeptide.
- the spacer domain may comprise up to 300 amino acids, 10 to 100 amino acids, or 25 to 50 amino acids.
- the spacer is a short spacer which comprises less than 100, less than 90, less than 80, less than 70, less than 60, less than 50, less than 40, less than 30, less than 20, or less than 10 amino acids.
- the spacer may include a hinge region.
- the hinge region may be located between the antigen binding domain and the transmembrane domain of the CAR.
- a hinge region can include at least a portion of a Fc region, for example, a hinge portion of a human Fc region of a CH3 domain or variants thereof.
- the spacer includes all or part of an immunoglobulin (e.g., IgGl, IgG2, IgG3, IgG4) hinge region, i.e. the sequence that falls between the CHI and CH2 domains of an immunoglobulin, e.g., IgG4 Fc hinge or a CD8 hinge region.
- Examples include CD8 hinge, CD28 hinge, IgG4 (HL-CH3), or IgG4 (L235E, N297Q).
- cysteines in the hinge region may be replaced with serines.
- Other examples of hinge regions are well known in the art.
- a short oligo- or polypeptide linker may connect two different domains of a CAR.
- the linker may be located between the antigen binding domain and the transmembrane domain of the CAR, or between the transmembrane domain and the intracellular domain of the CAR.
- the linker may be a Gly linker, a Gly-Ser linker, a EAAAK (SEQ ID NO: 5) linker, a PAPAP (SEQ ID NO: 6) linker, or an (Ala- Pro)! linker.
- the length and amino acid composition of the linker peptide sequence can be optimized to vary the orientation and/or proximity of the polypeptide domains to one another to achieve a desired activity of the chimeric polypeptide.
- the orientation and/or proximity of the polypeptide domains to one another can be varied as a “tuning” tool to achieve a tuning effect that would enhance or reduce the biological activity of the chimeric polypeptide.
- the linker is a synthetic compound linker such as, for example, a chemical cross-linking agent.
- suitable cross-linking agents include N-hydroxysuccinimide (NHS), disuccinimidylsuberate (DSS), bis(sulfosuccinimidyl)suberate (BS3), dithiobis(succinimidylpropionate) (DSP), dithiobis(sulfosuccinimidylpropionate) (DTSSP), ethyleneglycol bis(succinimidylsuccinate) (EGS), ethyleneglycol bis(sulfosuccinimidyl)uccinate) (sulfo-EGS), disuccinimidyl tartrate (DST), disulfosuccinimidyl tartrate (sulfo-DST), bis [2 (succinimidooxycarbonyloxy)ethyl]sulfone (B
- linkers are well known in the art.
- a CAR useful for expression in modified myeloid-derived cells of the disclosure can further comprise a detectable moiety as would be known in the art and/or an effector molecule, nonlimiting examples of which include a drug, a toxin, a small molecule, an antibody, and/or an antibody fragment, singly or in any combination.
- a CAR may be glycosylated, pegylated, and/or otherwise post- translationally modified. Glycosylation, pegylation, and/or other post-translational modifications may occur in vivo or in vitro and/or may be performed using chemical techniques. In some embodiments, any glycosylation, pegylation and/or other post-translational modifications may be N-linked or O- linked.
- a myeloid-derived cell from a precursor cell, such as a stem cell and/or a CD34+ cell.
- the stem cell is a hematopoietic stem cell.
- the methods of manufacturing a myeloid-derived cell disclosed herein can comprise the step of contacting a precursor cell with a precursor media comprising a precursor cytokine mixture, whereby the number of precursor cell(s) increases to generate a cell population comprising a plurality of precursor cells, wherein said precursor cell is a stem cell.
- the methods of manufacturing a myeloid-derived cell disclosed herein can comprise the step of contacting a precursor cell with a precursor media comprising a precursor cytokine mixture, whereby the number of precursor cell(s) increases to generate a cell population comprising a plurality of precursor cells.
- Also disclosed herein are methods of manufacturing a myeloid-derived cell disclosed herein comprising the step of contacting a precursor cell with a precursor media comprising a precursor cytokine mixture, whereby the number of precursor cell(s) increases to generate a cell population comprising a plurality of precursor cells, whereby upon contact of said precursor cell(s) with said precursor media, said precursor cell(s) differentiates into a myeloid-lineage cell to generate a myeloid-derived cell and/or a mixed cell population comprising a plurality of myeloid-derived cells and a plurality of precursor cells.
- Any cell and/or population comprising a plurality of cells can be expected to expand (i.e., increase in cell number) when contacted with and maintained in any of the precursor media described herein.
- a myeloid-derived cell and/or a mixed cell population comprising a plurality of myeloid-derived cells and a plurality of precursor cells would expand in the precursor media.
- the number of myeloid-derived cells within the precursor media would be expected to increase.
- Also disclosed herein is a method of manufacturing a genetically modified myeloid-derived cell comprising a chimeric antigen receptor (CAR), wherein the method comprises: (a) contacting a precursor cell with a nucleic acid molecule comprising a polynucleotide sequence encoding the CAR to generate a genetically modified precursor CAR cell; and (b) expanding said genetically modified precursor CAR cell in a precursor media comprising a precursor cytokine mixture to generate a population comprising a plurality of genetically modified precursor CAR cells.
- the process of expanding the precursor CAR cell(s) can involve contacting said precursor CAR cell(s) with said precursor media and/or said precursor cytokine mixture.
- Also disclosed herein is a method of manufacturing a genetically modified myeloid-derived cell comprising a chimeric antigen receptor (CAR), the method comprising the steps of: (a) contacting a precursor cell with a nucleic acid molecule comprising a polynucleotide sequence encoding a CAR, whereby said nucleic acid is introduced into said precursor cell, and (b) contacting said precursor cell with a precursor media comprising a precursor cytokine mixture, whereby the number of precursor cell(s) increases to generate a population of cells comprising a plurality of precursor CAR cells, wherein said precursor cell is a stem cell.
- CAR chimeric antigen receptor
- precursor media and precursor cytokine mixtures that are utilized to expand a precursor cell.
- the process of expanding the precursor cell results in a cell population comprising a plurality of precursor cells.
- the precursor cell that undergoes expansion can be a genetically modified precursor CAR cell.
- the genetically modified precursor CAR cell undergoes genetic modification to introduce a CAR into the cell prior to expansion.
- the precursor cell is a bone marrow-derived cell or a stem cell.
- the precursor cell is a hematopoietic stem cell.
- the precursor cell that undergoes expansion is a genetically modified HSC CAR cell, and the genetically modified HSC CAR cell undergoes genetic modification to introduce a CAR into the cell prior to expansion.
- the precursor (e.g., HSC) cell is genetically modified to express the CAR
- the genetically modified precursor CAR cell is expanded in a precursor media comprising a precursor cytokine mixture. Expansion of the HSC CAR cell can occur at any time following genetic modification of the HSC cell that will result in expansion of the genetically modified HSC CAR cell.
- expansion can occur 1 day, 2 days, 3 days, 4 days, 5 days, 6 days, 7 days, 8 days, 9 days, 10 days, 11 days, 12 days, 13 days, 14 days, 15 days, 16 days, 17 days, 18 days, 19 days, 20 days, or more than 20 days following transduction.
- the number of days after transduction in which expansion should ideally occur will depend on the integration of the CAR into the precursor cell. For instance, it may be beneficial for the expansion process to occur once the CAR is suitable integrated into the precursor CAR cell. Expansion can also occur is a shorter amount of time following transduction, such as 2 hours, 4 hours, 6 hours, 8 hours, 10 hours, 12 hours, 14 hours, 16 hours, 18 hours, or 20 hours following CAR transduction.
- the step of expanding the genetically modified precursor CAR cell disclosed herein is particularly effective at generating a population comprising a plurality of genetically modified precursor CAR cells.
- the step of expanding the genetically modified HSC CAR cell disclosed herein results in a population comprising a plurality of genetically modified HSC CAR cells.
- the expansion step disclosed herein is also particularly effective at increasing the number of precursor cells. In some instances, the number of precursor cells within the precursor media is increased at a faster rate compared to precursor cells that are expanded in the absence of the precursor media disclosed herein.
- the precursor cells expanded in the precursor medium comprising the precursor cytokine mixture increase in number of cells over a short amount of time compared to precursor cells expanded in a medium lacking the precursor medium.
- a precursor cytokine mixture that can be added to a precursor media.
- the precursor cytokine mixture contributes to increased expansion of the precursor cell(s).
- the number of precursor cells is expanded at a faster rate in a precursor media comprising the precursor cytokine mixture described herein, compared to a precursor media lacking the precursor cytokine mixture of the present disclosure.
- it is thought that the presence of the precursor cytokine mixture contributes to the superior technical effects of the precursor media (i.e., increased expansion rate and differentiation toward a myeloid lineage).
- the genetically modified precursor CAR cell(s) in the precursor media begin differentiating at the same time the precursor CAR cell(s) are expanding.
- the step of expanding the genetically modified precursor CAR cell(s) in a precursor media also promotes the differentiation of the genetically modified precursor CAR cell(s) toward the myeloid lineage.
- the methods disclosed herein comprise the step of contacting said precursor cell with a precursor media comprising a precursor cytokine mixture, whereby the number of precursor cell(s) increases to generate a population of cells comprising a plurality of precursor CAR cells. Expansion of at least one precursor cell can involve maintaining said at least one precursor cell in said precursor media.
- the precursor cell can be maintained in said precursor media for any suitable amount of time to generate a population of cells comprising a plurality of precursor CAR cells. Once a population of cells comprising a plurality of precursor CAR cells is produced within a precursor media, said population can be maintained in the precursor media for any suitable amount of time necessary to generate population comprising the specific characteristics that are desired.
- characteristics include: total cell number with the population or total number of a specific type of cell within the population (e.g., precursor cell, or myeloid-derived cell)).
- the precursor CAR cell is maintained in said precursor media to increase the number of precursor CAR cells. In some instances, the precursor CAR cell is maintained in said precursor media to increase the number of precursor CAR cells or myeloid-derived CAR cells within the population.
- the precursor media disclosed herein upon contact with a precursor cell, can promote expansion and differentiation of said precursor cell.
- the precursor media disclosed herein possess a superior effect of promoting differentiation of a precursor cell toward a myeloid lineage cell.
- the precursor media described herein is particularly effective at promoting differentiation of a stem cell into a myeloid cell.
- the precursor media is particularly effective at promoting differentiation of a stem cell CAR cell into a myeloid-derived CAR cell. Accordingly, as a precursor cell is contacted with, and maintained in, a precursor media the number of precursor cells and myeloid-derived cells increases within the precursor media to generate a population of cells comprising a plurality of precursor cells and myeloid-derived cells.
- the precursor cells and myeloid-derived cells can be CAR cells.
- the precursor media are effective at promoting expansion of a cell population comprising a plurality of precursor cells and myeloid-derived cells.
- the precursor cells and myeloid-derived cells can be expanded.
- the precursor cells and myeloid-derived cells can be expanded.
- Expansion of genetically modified precursor CAR cells can increase the amount of cells by about 10 fold, 20 fold, 30 fold, 40 fold, 50 fold, 60 fold, 70 fold, 80 fold, 90 fold, 100 fold, 200 fold, 300 fold, 400 fold, 500 fold, 600 fold, 700 fold, 800 fold, 900 fold, 1000 fold, 2000 fold, 3000 fold, 4000 fold, 5000 fold, 6000 fold, 7000 fold, 8000 fold, 9000 fold, 10,000 fold, 100,000 fold, 1,000,000 fold, 10,000,000 fold, or greater.
- the cells are expanded in the range of about 20 fold to about 300 fold, about 20 fold to about 250 fold, about 20 fold to about 200 fold, about 20 fold to about 150 fold, about 20 fold to about 100 fold, about 20 fold to about 50 fold.
- expansion of genetically modified precursor CAR cells can increase the amount of precursor CAR cells between 10-fold to 100-fold, between 20-fold to 200-fold, between 30-fold to 300-fold, between 40-fold to 400-fold, between 50-fold to 500-fold, between 60-fold to 600-fold, between 70-fold to 700-fold, between 80-fold to 800-fold, between 90-fold to 900-fold, between 100-fold to 1000-fold, or greater than 1000-fold.
- the expansion step can be very short, for example less than 24 hours such as 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, or 23 hours.
- the expansion step can be longer, for example 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, or more days.
- the cells may be expanded for several hours (about 3 hours) to about 14 days or any hourly integer value in between.
- the genetically modified precursor CAR cell(s) is expanded for at least 1, at least 2, at least 3, at least 4, at least 5, at least 6 days, at least 7 days, at least 8 days, at least 9 days, at least 10 days, at least 11 days, at least 12 days, at least 13 days, at least 14 days, at least 15 days, at least 16 days, at least 17 days, at least 18 days, at least 19 days, at least 20 days, or more than 20 days.
- the genetically modified precursor CAR cell(s) is expanded for 1 day, for 2 days, for 3 days, for 4 days, for 5 days, for 6 days, for 7 days, for 8 days, for 9 days, for 10 days, for 11 days, for 12 days, for 13 days, for 14 days, for 15 days, or for more than 15 days.
- the precursor CAR cell, or population comprising the same is maintained in precursor media for 1 day.
- the precursor CAR cell, or population comprising the same is maintained in precursor media for 2 days.
- the precursor CAR cell, or population comprising the same is maintained in precursor media for 3 days.
- the precursor CAR cell, or population comprising the same is maintained in precursor media for 4 days. In some embodiments, the precursor CAR cell, or population comprising the same, is maintained in precursor media for 5 days. In some embodiments, the precursor CAR cell, or population comprising the same, is maintained in precursor media for 6 days. In some embodiments, the precursor CAR cell, or population comprising the same, is maintained in precursor media for 7 days. In some embodiments, the precursor CAR cell, or population comprising the same, is maintained in precursor media for 8 days. In some embodiments, the precursor CAR cell, or population comprising the same, is maintained in precursor media for 9 days. In some embodiments, the precursor CAR cell, or population comprising the same, is maintained in precursor media for 10 days.
- the precursor CAR cell, or population comprising the same is maintained in precursor media for 11 days. In some embodiments, the precursor CAR cell, or population comprising the same, is maintained in precursor media for 12 days. In some embodiments, the precursor CAR cell, or population comprising the same, is maintained in precursor media for 13 days. In some embodiments, the precursor CAR cell, or population comprising the same, is maintained in precursor media for 14 days. In some embodiments, the precursor CAR cell, or population comprising the same, is maintained in precursor media for 15 days. In some embodiments, the precursor CAR cell, or population comprising the same, is maintained in precursor media for 16 days. In some embodiments, the precursor CAR cell, or population comprising the same, is maintained in precursor media for 17 days.
- the precursor CAR cell, or population comprising the same is maintained in precursor media for 18 days. In some embodiments, the precursor CAR cell, or population comprising the same, is maintained in precursor media for 19 days. In some embodiments, the precursor CAR cell, or population comprising the same, is maintained in precursor media for 20 days.
- the expansion step of the methods disclosed herein can increase the amount of the differentiated cells by about 10 fold, 20 fold, 30 fold, 40 fold, 50 fold, 60 fold, 70 fold, 80 fold, 90 fold, 100 fold, 200 fold, 300 fold, 400 fold, 500 fold, 600 fold, 700 fold, 800 fold, 900 fold, 1000 fold, 2000 fold, 3000 fold, 4000 fold, 5000 fold, 6000 fold, 7000 fold, 8000 fold, 9000 fold, 10,000 fold, 100,000 fold, 1,000,000 fold, 10,000,000 fold, or greater.
- expansion can increase the amount of myeloid-derived CAR cells between 10-fold to 100-fold, between 20-fold to 200-fold, between 30-fold to 300-fold, between 40-fold to 400-fold, between 50-fold to 500-fold, between 60-fold to 600-fold, between 70-fold to 700-fold, between 80- fold to 800-fold, between 90-fold to 900-fold, between 100-fold to 1000-fold, or greater than 1000- fold.
- myeloid-derived CAR cells between 10-fold to 100-fold, between 20-fold to 200-fold, between 30-fold to 300-fold, between 40-fold to 400-fold, between 50-fold to 500-fold, between 60-fold to 600-fold, between 70-fold to 700-fold, between 80- fold to 800-fold, between 90-fold to 900-fold, between 100-fold to 1000-fold, or greater than 1000- fold.
- genetically modified precursor CAR cell(s) that are differentiated in a precursor media increases, the number of genetically modified precursor CAR cell(s) that are undifferentiated in the precursor media decreases.
- Differentiation of genetically modified precursor CAR cells can increase the ratio of the differentiated cells (i.e., myeloid-derived cells) to undifferentiated cells (i.e., stem cells) within a population by a factor of 10 fold, 20 fold, 30 fold, 40 fold, 50 fold, 60 fold, 70 fold, 80 fold, 90 fold, 100 fold, 200 fold, 300 fold, 400 fold, 500 fold, 600 fold, 700 fold, 800 fold, 900 fold, 1000 fold, 2000 fold, 3000 fold, 4000 fold, 5000 fold, 6000 fold, 7000 fold, 8000 fold, 9000 fold, 10,000 fold, 100,000 fold, 1,000,000 fold, 10,000,000 fold, or greater.
- the precursor CAR cell(s) is maintained in said precursor medium until the number of cells is at least 10 4 , at least 10 5 , at least 10 6 , at least 10 7 , at least 10 8 , at least 10 9 , at least IO 10 , at least IO 11 , at least 10 12 , at least IO 13 , at least 10 14 , at least IO 15 , at least 10 16 , at least 10 17 , at least 10 18 , at least 10 19 , at least IO 20 , or greater than IO 20 .
- the precursor CAR cell(s) is maintained in said precursor medium until the number of cells within the generated population is at least 10 4 , at least 10 5 , at least 10 6 , at least 10 7 , at least 10 8 , at least 10 9 , at least IO 10 , at least IO 11 , at least 10 12 , at least IO 13 , at least 10 14 , at least IO 15 , at least 10 16 , at least 10 17 , at least 10 18 , at least 10 19 , at least IO 20 , or greater than IO 20 .
- the precursor CAR cell(s) is maintained in said precursor medium to generate a population of cells comprising a plurality of precursor CAR cells and/or a plurality of myeloid-derived CAR cells, wherein the number of cells within the population is at least 10 4 , at least 10 5 , at least 10 6 , at least 10 7 , at least 10 8 , at least 10 9 , at least IO 10 , at least 10 11 , at least 10 12 , at least 10 13 , at least 10 14 , at least 10 15 , at least 10 16 , at least 10 17 , at least 10 18 , at least 10 19 , at least IO 20 , or greater than IO 20 .
- the precursor CAR cell(s) is maintained in the precursor medium for at least 2, at least 3, at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, at least 10, at least 11, or at least 12 days. In some embodiments, the precursor CAR cell(s) are maintained in precursor media for between 9-11 days. In some embodiments, the precursor CAR cell(s) are maintained in precursor media for 10 days. In some embodiments, said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population, is maintained in said precursor media for 1 day.
- said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population is maintained in said precursor media for 2 days. In some embodiments, said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population, is maintained in said precursor media for 3 days.
- said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population is maintained in said precursor media for 4 days. In some embodiments, said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population, is maintained in said precursor media for 5 days.
- said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population is maintained in said precursor media for 6 days. In some embodiments, said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population, is maintained in said precursor media for 7 days.
- said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population is maintained in said precursor media for 8 days. In some embodiments, said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population, is maintained in said precursor media for 9 days.
- said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population is maintained in said precursor media for 10 days. In some embodiments, said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population, is maintained in said precursor media for 11 days.
- said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population is maintained in said precursor media for 12 days. In some embodiments, said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population, is maintained in said precursor media for 13 days.
- said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population is maintained in said precursor media for 14 days. In some embodiments, said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population, is maintained in said precursor media for 15 days.
- said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population is maintained in said precursor media for 16 days. In some embodiments, said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population, is maintained in said precursor media for more than 16 days.
- said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population is maintained in precursor media for between 12-48 hours. In some embodiments, said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population, is maintained in precursor media for 24-72 hours.
- said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population is maintained in precursor media for between 48-96 hours. In some embodiments, said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population, is maintained in precursor media for between 72-120 hours.
- said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population is maintained in precursor media for between 96-144 hours. In some embodiments, said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population, is maintained in precursor media for between 120-168 hours.
- said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population is maintained in precursor media for between 144-192 hours. In some embodiments, said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population, is maintained in precursor media for between 168-216 hours.
- said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population is maintained in precursor media for between 192-240 hours. In some embodiments said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population, is maintained in precursor media for between 216-264 hours.
- said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population is maintained in precursor media for between 240-288 hours. In some embodiments, said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population, is maintained in precursor media for between 264-312 hours.
- said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population is maintained in precursor media for between 288-336 hours. In some embodiments, said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population, is maintained in precursor media for between 312-360 hours.
- said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population is maintained in precursor media for between 336-384 hours. In some embodiments, said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population, is maintained in precursor media for between 360-408 hours.
- said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population is maintained in precursor media for between 384-432 hours. In some embodiments, said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population, is maintained in precursor media for between 408-456 hours.
- said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population is maintained in precursor media for more than 456 hours.
- the precursor media and myeloid media disclosed herein can comprise a mixed population of cells.
- the precursor media and myeloid media disclosed herein are particularly effective at generating a mixed population of cells. For instance, when a precursor cell and/or a precursor CAR cell, for example, are contacted with, and maintained in a precursor media, said precursor cell and/or said precursor CAR cell, can expand to generate a cell population comprising a plurality of said precursor cells and/or said precursor CAR cells, and/or said differentiate into a myeloid- derived cell and/or myeloid-derived CAR cell.
- said precursor CAR cell expands to generate a cell population comprising a plurality of said precursor CAR cells
- said precursor CAR cells within said plurality of precursor CAR cells can expand to increase the number of precursor CAR cells within said population and/or differentiate into a myeloid-derived CAR cell(s).
- a plurality of precursor CAR cells can be undergoing expansion and/or differentiation, such that the number of precursor CAR cells is increasing and the number of myeloid-derived CAR cells generated from said precursor CAR cells is increasing, thereby generating a mixed population of cells comprising a plurality of precursor CAR cells and myeloid-derived CAR cells.
- Any precursor CAR cell within a precursor media can be expected to expand and/or differentiate.
- Any myeloid- derived CAR cell within said precursor media can be expected to expand and/or differentiate.
- the number of precursor CAR cells within a population present in a precursor media increases, the number of precursor CAR cells that can and/or will differentiate into myeloid-derived CAR cells increases accordingly.
- the rate at which the number of myeloid-derived CAR cells within the population increases can increase.
- the term “mixed population” as used herein refers to any cell population comprising a plurality of myeloid-derived cells and/or a plurality of precursor cells, and/or a plurality of myeloid-derived CAR cells and/or a plurality of precursor CAR cells.
- the mixed population can comprise a plurality of myeloid-derived cells and/or a plurality of precursor cells.
- the mixed population comprises a plurality of myeloid-derived cells and a plurality of precursor cells.
- the mixed population can comprise a plurality of myeloid-derived CAR cells and/or a plurality of precursor CAR cells.
- the mixed population comprises a plurality of myeloid-derived CAR cells and a plurality of precursor CAR cells.
- the mixed population comprises comprises a plurality of myeloid-derived CAR cells and/or a plurality of precursor CAR cells
- the mixed population comprises a plurality of myeloid-derived CAR cells and/or a plurality of precursor CAR cells
- myeloid-derived cell and/or precursor cell within the population would comprise a CAR (i.e., at least one cell within said population will not comprise a CAR).
- the mixed population comprises a plurality of myeloid-derived CAR cells and a plurality of precursor CAR cells
- At least 70%, at least 71%, at least 72%, at least 73%, at least 74%, at least 75%, at least 76%, at least 77%, at least 78%, at least 79%, at least 80%, at least 81%, at least 82%, at least 83%, at least 84%, at least 85%, at least 86%, at least 87%, at least 88%, at least 89%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or greater than 99% of cells within said plurality of myeloid-derived CAR cells within said population comprises a CAR.
- At least 70%, at least 71%, at least 72%, at least 73%, at least 74%, at least 75%, at least 76%, at least 77%, at least 78%, at least 79%, at least 80%, at least 81%, at least 82%, at least 83%, at least 84%, at least 85%, at least 86%, at least 87%, at least 88%, at least 89%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or greater than 99% of cells within said plurality of precursor CAR cells within said population comprises a CAR.
- the differentiated cells of the present disclosure are preferential differentiated toward the myeloid cell lineage.
- the step of differentiating a genetically modified precursor CAR cell in a precursor media generates a myeloid-derived CAR cell.
- the step of differentiating a genetically modified precursor CAR cell(s) in a precursor media generates a plurality of genetically modified myeloid-derived CAR cells.
- the ratio of precursor (e.g., stem cells) within the plurality is decreased relative to myeloid-derived cells.
- the ratio of CD34+ cells decreases as the plurality of precursor CAR cells differentiates. Differentiation can decrease the amount of CD34+ cells within a plurality of genetically modified precursor CAR cells or a plurality of genetically modified myeloid-derived cells.
- Differentiation of genetically modified precursor CAR cells can decrease the amount of CD34+ cells within a plurality of genetically modified precursor CAR cells or a plurality of genetically modified myeloid-derived cells by about 10 fold, 20 fold, 30 fold, 40 fold, 50 fold, 60 fold, 70 fold, 80 fold, 90 fold, 100 fold, 200 fold, 300 fold, 400 fold, 500 fold, 600 fold, 700 fold, 800 fold, 900 fold, 1000 fold, 2000 fold, 3000 fold, 4000 fold, 5000 fold, 6000 fold, 7000 fold, 8000 fold, 9000 fold, 10,000 fold, 100,000 fold, 1,000,000 fold, 10,000,000 fold, or greater.
- Myeloid cells can be identified and/or isolated based on cell surface markers.
- myeloid cells within the myeloid lineage can be identified based on specific cell surface markers.
- exemplary, non-limiting, surface markers for myeloid cells include: CD13, CD14, CD15 and CD1 lb. Accordingly, during the expansion step, the number of cells expression myeloid-associated cell surface markers can be expected to increase, while the number of cells expressing a common stem cell surface marker (i.e., CD34) can be expected to decrease as differentiation occurs.
- the expansion step of the methods disclosed herein can increase the amount of CD1 lb+ cells within a plurality of genetically modified precursor CAR cells or a plurality of genetically modified myeloid-derived cells.
- Differentiation of genetically modified precursor CAR cells can increase the amount of CD1 lb+ cells within a plurality of genetically modified precursor CAR cells or a plurality of genetically modified myeloid-derived cells by about 10 fold, 20 fold, 30 fold, 40 fold, 50 fold, 60 fold, 70 fold, 80 fold, 90 fold, 100 fold, 200 fold, 300 fold, 400 fold, 500 fold, 600 fold, 700 fold, 800 fold, 900 fold, 1000 fold, 2000 fold, 3000 fold, 4000 fold, 5000 fold, 6000 fold, 7000 fold, 8000 fold, 9000 fold, 10,000 fold, 100,000 fold, 1,000,000 fold, 10,000,000 fold, or greater.
- a precursor CAR cell, or population comprising a plurality of precursor CAR cells, or a population comprising a plurality of precursor CAR cells and a plurality of myeloid-derived CAR cells is maintained in a precursor media until the number of CD1 lb+ cells within the population is increased between 10 fold to 100 fold, between 20 fold and 200 fold, between 30 fold and 300 fold, between 40 fold and 400 fold, between 50 fold and 500 fold, between 60 fold and 600 fold, between 70 fold and 700 fold, between 80 fold and 800 fold, between 90 fold and 900 fold, between 100 fold and 10,000 fold, between 1000 fold and 100,000 fold, between 10,000 fold and 1,000,000 fold, between 100,000 fold and 10,000,000 fold , between 10 5 fold and 10 7 fold, between 10 6 fold and 10 8 fold, between 10 7 fold and 10 9 fold, 10 8 fold and 10 10 fold, 10 9 fold and 10 11 fold, 10 10 fold and 10 12 fold, 10 11 fold and 10 13 fold, 10 12 fold and 10 14 fold, 10 13 fold and 10 15 fold, 10 14 fold and 10 16 fold
- the expansion step of the methods disclosed herein can increase the amount of CD14+ cells within a plurality of genetically modified precursor CAR cells or a plurality of genetically modified myeloid-derived cells.
- Differentiation of genetically modified precursor CAR cells can increase the amount of CD14+ cells within a plurality of genetically modified precursor CAR cells or a plurality of genetically modified myeloid-derived cells by about 10 fold, 20 fold, 30 fold, 40 fold, 50 fold, 60 fold, 70 fold, 80 fold, 90 fold, 100 fold, 200 fold, 300 fold, 400 fold, 500 fold, 600 fold, 700 fold, 800 fold, 900 fold, 1000 fold, 2000 fold, 3000 fold, 4000 fold, 5000 fold, 6000 fold, 7000 fold, 8000 fold, 9000 fold, 10,000 fold, 100,000 fold, 1,000,000 fold, 10,000,000 fold, or greater.
- a precursor CAR cell, or population comprising a plurality of precursor CAR cells, or a population comprising a plurality of precursor CAR cells and a plurality of myeloid-derived CAR cells is maintained in a precursor media until the number of CD14+ cells within the population is increased between 10 fold to 100 fold, between 20 fold and 200 fold, between 30 fold and 300 fold, between 40 fold and 400 fold, between 50 fold and 500 fold, between 60 fold and 600 fold, between 70 fold and 700 fold, between 80 fold and 800 fold, between 90 fold and 900 fold, between 100 fold and 10,000 fold, between 1000 fold and 100,000 fold, between 10,000 fold and 1,000,000 fold, between 100,000 fold and 10,000,000 fold , between 10 5 fold and 10 7 fold, between 10 6 fold and 10 8 fold, between 10 7 fold and 10 9 fold, 10 8 fold and 10 10 fold, 10 9 fold and 10 11 fold, 10 10 fold and 10 12 fold, 10 11 fold and 10 13 fold, 10 12 fold and 10 14 fold, 10 13 fold and 10 15 fold, 10 14 fold and 10 16 fold, or greater
- the expansion step of the methods disclosed herein can increase the amount of CD 13+ cells within a plurality of genetically modified precursor CAR cells or a plurality of genetically modified myeloid-derived cells.
- Differentiation of genetically modified precursor CAR cells can increase the amount of CD 13+ cells within a plurality of genetically modified precursor CAR cells or a plurality of genetically modified myeloid-derived cells by about 10 fold, 20 fold, 30 fold, 40 fold, 50 fold, 60 fold, 70 fold, 80 fold, 90 fold, 100 fold, 200 fold, 300 fold, 400 fold, 500 fold, 600 fold, 700 fold, 800 fold, 900 fold, 1000 fold, 2000 fold, 3000 fold, 4000 fold, 5000 fold, 6000 fold, 7000 fold, 8000 fold, 9000 fold, 10,000 fold, 100,000 fold, 1,000,000 fold, 10,000,000 fold, or greater.
- a precursor CAR cell, or population comprising a plurality of precursor CAR cells, or a population comprising a plurality of precursor CAR cells and a plurality of myeloid-derived CAR cells is maintained in a precursor media until the number of CD 13+ cells within the population is increased between 10 fold to 100 fold, between 20 fold and 200 fold, between 30 fold and 300 fold, between 40 fold and 400 fold, between 50 fold and 500 fold, between 60 fold and 600 fold, between 70 fold and 700 fold, between 80 fold and 800 fold, between 90 fold and 900 fold, between 100 fold and 10,000 fold, between 1000 fold and 100,000 fold, between 10,000 fold and 1,000,000 fold, between 100,000 fold and 10,000,000 fold , between 10 5 fold and 10 7 fold, between 10 6 fold and 10 8 fold, between 10 7 fold and 10 9 fold, 10 8 fold and 10 10 fold, 10 9 fold and 10 11 fold, 10 10 fold and 10 12 fold, 10 11 fold and 10 13 fold, 10 12 fold and 10 14 fold, 10 13 fold and 10 15 fold, 10 14 fold and 10 16 fold, or greater
- the expansion step of the methods disclosed herein can increase the amount of CD 15+ cells within a plurality of genetically modified precursor CAR cells or a plurality of genetically modified myeloid-derived cells.
- Differentiation of genetically modified precursor CAR cells can increase the amount of CD 15+ cells within a plurality of genetically modified precursor CAR cells or a plurality of genetically modified myeloid-derived cells by about 10 fold, 20 fold, 30 fold, 40 fold, 50 fold, 60 fold, 70 fold, 80 fold, 90 fold, 100 fold, 200 fold, 300 fold, 400 fold, 500 fold, 600 fold, 700 fold, 800 fold, 900 fold, 1000 fold, 2000 fold, 3000 fold, 4000 fold, 5000 fold, 6000 fold, 7000 fold, 8000 fold, 9000 fold, 10,000 fold, 100,000 fold, 1,000,000 fold, 10,000,000 fold, or greater.
- a precursor CAR cell, or population comprising a plurality of precursor CAR cells, or a population comprising a plurality of precursor CAR cells and a plurality of myeloid- derived CAR cells is maintained in a precursor media until the number of CD 15+ cells within the population is increased between 10 fold to 100 fold, between 20 fold and 200 fold, between 30 fold and 300 fold, between 40 fold and 400 fold, between 50 fold and 500 fold, between 60 fold and 600 fold, between 70 fold and 700 fold, between 80 fold and 800 fold, between 90 fold and 900 fold, between 100 fold and 10,000 fold, between 1000 fold and 100,000 fold, between 10,000 fold and 1,000,000 fold, between 100,000 fold and 10,000,000 fold , between 10 5 fold and 10 7 fold, between 10 6 fold and 10 8 fold, between 10 7 fold and 10 9 fold, 10 8 fold and 10 10 fold, 10 9 fold and 10 11 fold, 10 10 fold and 10 12 fold, 10 11 fold and 10 13 fold, 10 12 fold and 10 14 fold, 10 13 fold and 10 15 fold, 10 14 fold and 10 16 fold, or
- the amount of cell surface marker expression within a plurality of genetically modified precursor CAR cells or plurality of plurality of genetically modified myeloid-derived CAR cells can be used to determine how long to maintain the expanding step of the method of manufacturing a genetically modified myeloid-derived CAR cells disclosed herein.
- the expanding step is maintained until a certain percentage of the population comprising a plurality of genetically modified precursor CAR cells and/or genetically modified myeloid-derived CAR cells expresses a specific marker.
- the expanding step is maintained until a certain percentage of the population comprising a plurality of genetically modified precursor CAR cells and/or plurality of genetically modified myeloid-derived cells expresses at least a specific marker.
- a genetically modified myeloid-derived CAR cell from a precursor cell, such as a hematopoietic stem cell, wherein the precursor cell comprising a CAR (i.e., precursor CAR cell) is expanded by contacting said precursor CAR cell with a precursor media comprising a precursor cytokine mixture, whereby the number of precursor CAR cell(s) increases to generate a population of precursor cells (e.g., stem cells) comprising a plurality of precursor CAR cells, wherein following contact of said precursor CAR cell(s) within said population with said precursor media, said precursor CAR cell(s) differentiates into a myeloid-lineage cell(s) to generate a myeloid-derived CAR cell(s) or a population comprising a plurality of myeloid-derived CAR cell(s).
- precursor cells i.e., precursor CAR cell
- a precursor media comprising a precursor cytokine mixture
- a cell population would be understood to have been generated from a precursor cell if, as described above, said population comprises at least one cell that was generated from a cell division and/or a cell differentiation of said precursor cell. In some cases, the population is a mixed population, as described herein. In some embodiments, the population is exclusively precursor cells. In some embodiments, the population is exclusively myeloid-derived cells.
- the expanding step is maintained until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population comprising a plurality of genetically modified precursor CAR cells and/or a plurality of genetically modified myeloid-derived CAR cells is CD1 lb+.
- the precursor CAR cell(s) and/or population comprising a plurality of precursor CAR cells is maintained in said precursor media until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population is CD1 lb+.
- the population comprising a plurality of myeloid-derived CAR cells is maintained in said precursor media until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population is CD1 lb+.
- the precursor CAR cell(s) and/or population comprising a plurality of precursor CAR cells and/or plurality of myeloid-derived CAR cells is maintained in said precursor media until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population is CD1 lb+CD13+CD14+.
- the precursor CAR cell(s) and/or population comprising a plurality of precursor CAR cells and/or plurality of myeloid-derived CAR cells is maintained in said precursor media until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population is CDl lb+CD13+CD15+.
- the precursor CAR cell(s) and/or population comprising a plurality of precursor CAR cells and/or plurality of myeloid-derived CAR cells is maintained in said precursor media until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population is CD1 lb+CD14+.
- the precursor CAR cell(s) and/or population comprising a plurality of precursor CAR cells and/or plurality of myeloid-derived CAR cells is maintained in said precursor media until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population is CD1 lb+CD14+CD15+.
- the expanding step is maintained until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population comprising a plurality of myeloid-derived CAR cells is CD14+.
- the expanding step is maintained until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population comprising a plurality of genetically modified precursor CAR cells and/or plurality of genetically modified myeloid-derived CAR cells is CD14+.
- the precursor CAR cell(s) and/or population comprising a plurality of precursor CAR cells is maintained in said precursor media until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population is CD14+.
- the precursor CAR cell(s) and/or population comprising a plurality of precursor CAR cells and/or plurality of myeloid-derived CAR cells is maintained in said precursor media until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population is CD14+.
- the population comprising a plurality of myeloid-derived CAR cells is maintained in said precursor media until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population is CD14+.
- the precursor CAR cell(s) and/or population comprising a plurality of precursor CAR cells and/or plurality of myeloid-derived CAR cells is maintained in said precursor media until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population is CD14+CD13+CD15+.
- the expanding step is maintained until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population comprising a plurality of myeloid- derived CAR cells is CD13+.
- the expanding step is maintained until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population comprising a plurality of genetically modified precursor CAR cells and/or plurality of genetically modified myeloid- derived CAR cells is CD13+.
- the precursor CAR cell(s) and/or population comprising a plurality of precursor CAR cells and/or plurality of myeloid-derived CAR cells is maintained in said precursor media until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population is CD13+.
- the precursor CAR cell(s) and/or population comprising a plurality of precursor CAR cells and/or plurality of myeloid-derived CAR cells is maintained in said precursor media until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population is CD 13+CD 15+
- the expanding step is maintained until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population comprising a plurality of myeloid-derived CAR cells is CD15+.
- the expanding step is maintained until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population comprising a plurality of genetically modified precursor CAR cells and/or plurality of genetically modified myeloid-derived CAR cells is CD15+.
- the precursor CAR cell(s) and/or population comprising a plurality of precursor CAR cells is maintained in said precursor media until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population is CD15+.
- the precursor CAR cell(s) and/or population comprising a plurality of precursor CAR cells and/or plurality of myeloid-derived CAR cells is maintained in said precursor media until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population is CD15+.
- the population comprising a plurality of myeloid-derived CAR cells is maintained in said precursor media until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population is CD15+.
- the expanding step is maintained until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the population comprising a plurality of genetically modified precursor CAR cells or the population comprising a plurality of genetically modified myeloid-derived CAR cells is CD13+CD15+.
- the cells can be incubated in cell medium in a culture apparatus for a period of time or until the cells reach confluency or high cell density for optimal passage before passing the cells to another culture apparatus.
- the culturing apparatus can be any culture apparatus commonly used for culturing cells in vitro.
- the level of confluence is 70% or greater before passing the cells to another culture apparatus.
- the level of confluence is 90% or greater.
- a period of time can be any time suitable for the culture of cells in vitro.
- the culture medium may be replaced during the culture of the cells at any time. In some embodiments, the culture medium is replaced about every 2 to 3 days.
- the cells are then harvested from the culture apparatus whereupon the cells can be used immediately or stored for use at a later time.
- Examples of cell markers that can be used for myeloid-derived cells include, but are not limited to CD1 lb, CD13, CD14 and CD15.
- the myeloid-derived cells can express at least one surface marker selected from the following: CD1 lb, CD13, CD14, and CD15.
- the myeloid-derived cells can express at least two surface markers selected from the following: CD1 lb, CD13, CD14, and CD15.
- the myeloid-derived cells can express at least three surface markers selected from the following: CDl lb, CD13, CD14, and CD15.
- the myeloid-derived cells express the following surface markers: CD1 lb, CD13, CD14, and CD15.
- the expanding step can be about 1 day. In some embodiments, the expanding step is maintained for about 2 days. In some embodiments, the expanding step is maintained for about 3 days. In some embodiments, the expanding step is maintained for about 4 days. In some embodiments, the expanding step is maintained for about 5 days. In some embodiments, the expanding step is maintained for about 6 days. In some embodiments, the expanding step is maintained for about 7 days. In some embodiments, the expanding step is maintained for about 8 days. In some embodiments, the expanding step is maintained for about 9 days. In some embodiments, the expanding step is maintained for about 10 days. In some embodiments, the expanding step is maintained for about 11 days. In some embodiments, the expanding step is maintained for about 12 days.
- the expanding step is maintained for about 13 days. In some embodiments, the expanding step is maintained for about 14 days. In some embodiments, the expanding step is maintained for about 15 days. In some embodiments, the expanding step is maintained for at least 2, at least 3, at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, at least 10, at least 11, or at least 12 days. In some embodiments, the expanding step is maintained for between 9-11 days. In some embodiments, the expanding step is maintained for 10 days.
- the expanding step is maintained for about 192 hours. In some embodiments, the expanding step is maintained for about 216 hours. In some embodiments, the expanding step is maintained for about 240 hours. In some embodiments, the expanding step is maintained for about 264 hours. In some embodiments, the expanding step is maintained for about 288 hours. In some embodiments, the expanding step is maintained for about 312 hours. In some embodiments, the expanding step is maintained for about 336 hours. In some embodiments, the expanding step is maintained for about 360 hours. In some embodiments, the expanding step is maintained for about 384 hours. In some embodiments, the expanding step is maintained for about 408 hours. In some embodiments, the expanding step is maintained for about 432 hours. In some embodiments, the expanding step is maintained for about 456 hours. In some embodiments, the expanding step is maintained for more than 456 hours.
- the expanding step is maintained between 12-48 hours. In some embodiments, the expanding step is maintained between 24-72 hours. In some embodiments, the expanding step is maintained between 48-96 hours. In some embodiments, the expanding step is maintained between 72-120 hours. In some embodiments, the expanding step is maintained between 96-144 hours. In some embodiments, the expanding step is maintained between 120-168 hours. In some embodiments, the expanding step is maintained between 144-192 hours. In some embodiments, the expanding step is maintained between 168-216 hours. In some embodiments, the expanding step is maintained between 192-240 hours. In some embodiments, the expanding step is maintained between 216-264 hours. In some embodiments, the expanding step is maintained between 240-288 hours.
- the expanding step is maintained between 264-312 hours. In some embodiments, the expanding step is maintained between 288-336 hours. In some embodiments, the expanding step is maintained between 312-360 hours. In some embodiments, the expanding step is maintained between 336-384 hours. In some embodiments, the expanding step is maintained between 360-408 hours. In some embodiments, the expanding step is maintained between 384-432 hours. In some embodiments, the expanding step is maintained between 408-456 hours. In some embodiments, the expanding step is maintained for more than 456 hours.
- the expansion step can be maintained for any length of time indicated in Table 1.
- the precursor CAR cell and/or population comprising a plurality of precursor CAR cells and/or a plurality of myeloid-derived CAR cells can be maintained in precursor media for any length of time indicated in Table 1.
- the cells can be incubated in cell medium in a culture apparatus for a period of time or until the cells reach confluency or high cell density for optimal passage before passing the cells to another culture apparatus.
- the culturing apparatus can be any culture apparatus commonly used for culturing cells in vitro.
- the level of confluence is 70% or greater before passing the cells to another culture apparatus.
- the level of confluence is 90% or greater.
- a period of time can be any time suitable for the culture of cells in vitro.
- the culture medium may be replaced during the culture of the cells at any time. In some embodiments, the culture medium is replaced about every 2 to 3 days.
- the cells are then harvested from the culture apparatus whereupon the cells can be used immediately or stored for use at a later time.
- the step of expanding the genetically modified precursor CAR cell in a precursor media can involve culturing the cell in a precursor media to increase the number of cells. Culturing of precursor CAR cells as disclosed herein can occur in multiple types of culture media. The culture media used for culturing cells is dependent upon the desired stage (e.g., expansion and/or differentiation). Examples of media that can be used for culturing the genetically modified precursor CAR cells include basal growth media, precursor media, or myeloid media, as disclosed herein, or any media that achieves similar and desired results of expansion and/or differentiation. As described herein, culture mediums can be defined by the constituents within the medium. For example, precursor mediums can be defined by the precursor cytokine mixtures contained within.
- the precursor media, as disclosed herein, comprising a precursor cytokine mixture is particularly effective at promoting the expansion and differentiation of stem cells toward the myeloid lineage.
- genetically modified precursor CAR cells which are capable of differentiation into myeloid-derived cells, can be expanded in a precursor media comprising a precursor cytokine mixture to generate a population comprising a plurality of genetically modified precursor CAR cells, wherein a significant proportion of the plurality of genetically modified precursor CAR cells comprises a myeloid cell line surface marker.
- the culturing step can be very short, for example less than 24 hours such as 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, or 23 hours.
- the culturing step can be longer, for example 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, or more days.
- the cells may be cultured for several hours (about 3 hours) to about 14 days or any hourly integer value in between.
- Conditions appropriate for cell culture include an appropriate media (e.g., macrophage complete medium, DMEM/F12, DMEM/F 12-10 (Invitrogen)) that may contain factors necessary for proliferation and viability, including serum (e.g., fetal bovine or human serum), L-glutamine, insulin, M-CSF, GM-CSF, IL-10, IL-12, IL-15, TGF-beta, and TNFa, or any other additives for the growth of cells known to the skilled artisan.
- Other additives for the growth of cells include, but are not limited to, surfactant, plasmanate, and reducing agents such as N-acetyl-cysteine and 2-mercaptoethanol.
- culture media can include Stemline II, RPMI 1640, AEVI-V, DMEM, MEM, a-MEM, F-12, X-Vivo 15, X-Vivo 20, and Optimizer, with added amino acids, sodium pyruvate, and vitamins, either serum-free or supplemented with an appropriate amount of serum (or plasma) or a defined set of hormones, and/or an amount of cytokine(s) sufficient for the growth, expansion and/or differentiation of the cells.
- Antibiotics e.g., penicillin and streptomycin, are included only in experimental cultures, not in cultures of cells that are to be infused into a subject.
- the target cells are maintained under conditions necessary to support growth, for example, an appropriate temperature (e.g., 37°C) and atmosphere (e.g., air plus 5% CO2).
- the medium used to culture the cells may include an agent that can activate the cells.
- an agent that is known in the art to activate a macrophage, monocyte, dendritic cell, and/or a neutrophil is included in the medium.
- a precursor media comprising a precursor cytokine mixture for expanding genetically modified precursor CAR cell(s).
- the precursor media are particularly useful in their capacity to enable the expansion and/or differentiation of precursor cells, including hematopoietic stem cells, ex vivo.
- a hematopoietic stem cell can be expanded and differentiated into a myeloid-derived cell in the precursor medias disclosed herein ex vivo.
- the precursor media disclosed herein can promote the ex vivo differentiation of a stem cell into any myeloid-derived cell.
- the precursor medias disclosed herein are particularly effective for expanding a precursor CAR cell, and/or population comprising the same ex vivo.
- the methods are also particularly effective at expanding a population comprising the precursor CAR cell and/or a
- the precursor media comprising a precursor cytokine mixture are also particularly effective at promoting differentiation of a genetically modified precursor CAR cell.
- the genetically modified precursor CAR cells are genetically modified stem cells expressing a CAR.
- the stem cells expressing a CAR are hematopoietic stem cells (HSCs).
- the HSCs can be CD34+ cells.
- Expanding the HSCs modified to express a CAR generates a population comprising a plurality of genetically modified HSC CAR cells.
- expanding the genetically modified HSC CAR cell increases the number of a genetically modified myeloid-derived CAR cell(s).
- the precursor media disclosed herein is particularly effective at promoting the expansion of HSCs.
- the precursor media is particularly effective at expanding a CD34+ cell.
- the precursor media is particularly effective at differentiating a CD34+ cell in a myeloid lineage cell.
- the precursor media can be particularly effective at differentiating a CD34+ cell into a CD14+ cell, a CDl lb+ cell, a CD 13+ cell, a CD 15+ cell, a CDl lb+CD13+ cell, a
- CD1 lb+CD13+CD14+ cell a CD1 lb+CD13+CD15+ cell, a CDl lb+CD13+CD14+CD15+ cell, a CDl lb+CD14+ cell, a CDl lb+CD15+ cell, a CDl lb+CD14+CD15+ cell, a CD14+CD13+ cell, a CD14+CD15+ cell, a CD14+CD13+CD15+ cell, and/or a CD13+CD15+ cell.
- the precursor media disclosed herein can promote both expansion of CD34+ cells and the differentiation of CD34+ cells into myeloid lineage cells. In some instances, the precursor media is particularly effective at differentiation a CD34+ cell into a CD1 lb+ cell. In some instances, the precursor media is particularly effective at differentiation a CD34+ cell into a CD14+CD1 lb+ cell.
- the cytokine mixture within the precursor media i.e., the precursor cytokine mixture
- the precursor cytokine mixture is thought to contribute to the effective expansion and differentiation seen with the precursor media disclosed herein.
- precursor cytokine mixtures that are particularly effective at promoting expansion of CD34+ cells (e.g., HSCs) and differentiation of CD34+ stem cells into myeloid-derived cells.
- myeloid-derived cells are CD14+ cells.
- myeloid-derived cells are CD1 lb+ cells.
- the precursor media comprising the precursor cytokine mixtures disclosed herein are particularly effective at promoting differentiation of HSCs into CD14+ cells.
- the precursor media comprising the precursor cytokine mixtures disclosed herein are particularly effective at promoting differentiation of HSCs into CDl lb+ cells.
- any cytokines at any concentrations can be used in the precursor media to support expansion and/or differentiation of the genetically modified precursor CAR cells.
- the precursor cytokine mixture within the precursor media can contribute to the combined expansion of CD34+ and differentiation of CD34+ cells into myeloid-derived cells.
- the precursor cytokine mixture comprises Flt3, SCF, TPO, M-CSF, and GM-CSF.
- the precursor media comprises Flt3 at a concentration of 100 ng/mL, SCF at a concentration of 100 ng/mL, TPO at a concentration of 100 ng/mL, M-CSF at a concentration of 150 ng/mL and GM-CSF at a concentration of 100 ng/mL.
- the precursor media comprises Flt3 at a concentration of 100 ng/mL, SCF at a concentration of 100 ng/mL, TPO at a concentration of 100 ng/mL, M-CSF at a concentration of 150 ng/mL and GM-CSF at a concentration of 100 ng/mL, the precursor media further comprises GlutaMAXTM Supplement at a concentration of 2mM, and a basal growth media comprising VIVOTM- 15 medium and Stemline® II Hematopoietic Stem Cell Expansion Media mixed at a ratio of 1 : 1.
- the precursor cytokine mixture comprises G-CSF at a concentration of between about 100 ng/mL and 150 ng/mL.
- the precursor cytokine mixture comprises G-CSF at a concentration of between about 100 ng/mL and 150 ng/mL
- the precursor cytokine mixture comprises G-CSF, SCF, and TPO.
- the precursor media comprises G-CSF at a concentration of 100 ng/mL, SCF at a concentration of 100 ng/mL, and TPO at a concentration of 100 ng/mL.
- the precursor media further comprises a basal growth media comprising VIVOTM- 15 medium.
- the precursor media comprises granulocyte colony-stimulating factor (G-CSF).
- G-CSF granulocyte colony-stimulating factor
- the granulocyte colony-stimulating factor (G-CSF) can be present in the precursor media at a concentration of at least 20 ng/mL, at least 30 ng/mL, at least 40 ng/mL, at least 50 ng/mL, at least 60 ng/mL, of at least 70 ng/mL, at least 80 ng/mL, at least 90 ng/mL, at least 100 ng/mL, at least 110 ng/mL, at least 120 ng/mL, at least 130 ng/mL, at least 140 ng/mL, at least 150 ng/mL, at least 160 ng/mL, at least 170 ng/mL, at least 180 ng/mL, at least 190 ng/mL, at least 200 ng/mL, at least 210 ng/mL,
- the granulocyte colony-stimulating factor can be present in the precursor media at a concentration of 20-40 ng/mL, 30-50 ng/mL, 40-60 ng/mL, 50-70 ng/mL, 60-80 ng/mL, 70-90 ng/mL, 80-100 ng/mL, 90-110 ng/mL, 100-120 ng/mL, 110-130 ng/mL, 120-140 ng/mL, 130-150 ng/mL, 140-160 ng/mL, 150-170 ng/mL, 160-180 ng/mL, 170-190 ng/mL, 180-200 ng/mL, 190-210 ng/mL, 200-220 ng/mL, 210-230 ng/mL, 220- 240 ng/mL, 230-250 ng/mL, or greater than 250 ng/mL.
- G-CSF granulocyte colony-stimulating factor
- said G-CSF when the precursor media comprises G-CSF, said G-CSF is present at a concentration of between about 100 ng/mL and 200 ng/mL. In some embodiments, when the precursor media comprises G-CSF, said G-CSF is present at a concentration of between 100-105 ng/mL, 105-110 ng/mL, 110-115 ng/mL, 115-120 ng/mL, 120-125 ng/mL, 125-130 ng/mL, 130- 135 ng/mL, 135-140 ng/mL, 140-145 ng/mL, 145-150 ng/mL, 150-155 ng/mL, 155-160 ng/mL, 160-165 ng/mL, 165-170 ng/mL, 170-175 ng/mL, 175-180 ng/mL, 180-185 ng/mL, 185-190 ng/mL, 190-195 ng/mL,
- the precursor media comprises more than one cytokine (i.e., a precursor cytokine mixture).
- a precursor cytokine mixture comprises G-CSF at a concentration of between about 100 ng/mL and 150 ng/mL in said precursor media
- the precursor cytokine mixture comprises G-CSF, GM-CSF, SCF, and TPO.
- the precursor media can comprise any of the following cytokines: GM-CSF, SCF, and TPO.
- the precursor media can comprise at lease one of the following cytokines: GM-CSF, SCF, and TPO.
- said precursor media can comprise all of the following cytokines: GM-CSF, SCF, and TPO.
- GM-CSF can be present at a concentration between about 75 ng/mL and 125 ng/mL; SCF can be present at a concentration between about 75 ng/mL and 125 ng/mL; and/or TPO can be present at a concentration between about 75 ng/mL and 125 ng/mL.
- the precursor media comprises G-CSF at a concentration of 150 ng/mL, GM-CSF at a concentration of 100 ng/mL, SCF at a concentration of 100 ng/mL, and TPO at a concentration of 100 ng/mL.
- the precursor media when the precursor media comprises granulocyte colony-stimulating factor (G-CSF) at a concentration of between about 100 ng/mL and 200 ng/mL, said precursor media further comprises: a. granulocyte-macrophage colony-stimulating factor (GM-CSF) at a concentration of 10-30 ng/mL, 20-40 ng/mL, 30-50 ng/mL, 40-60 ng/mL, 50-70 ng/mL, 60-80 ng/mL, 70-90 ng/mL, 80-100 ng/mL, 90-110 ng/mL, 100-120 ng/mL, 110-130 ng/mL, 120-140 ng/mL, 130-150 ng/mL, 140-160 ng/mL, 150-170 ng/mL, 160-180 ng/mL, 170-190 ng/mL, 180- 200 ng/mL, 190-210 ng/mL, 200-2
- SCF stem cells factor
- TPO thrombopoietin
- concentration 10-30 ng/mL, 20-40 ng/mL, 30-50 ng/mL, 40-60 ng/mL, 50-70 ng/mL, 60-80 ng/mL, 70-90 ng/mL, 80-100 ng/mL, 90-110 ng/mL, 100-120 ng/mL, 110-130 ng/mL, 120- 140 ng/mL, 130-150 ng/mL, 140-160 ng/mL, 150-170 ng/mL, 160-180 ng/mL, 170-190 ng/mL, 180-200 ng/mL, 190-210 ng/mL, 200-220 ng/mL, or greater than 220 ng/mL.
- TPO thrombopoietin
- the precursor media when the precursor media comprises granulocyte colony-stimulating factor (G-CSF) at a concentration of about 140 ng/mL. In some embodiments, when the precursor media comprises granulocyte colony-stimulating factor (G-CSF) at a concentration of about 140 ng/mL, said precursor media further comprises: a.
- GM-CSF granulocyte-macrophage colony-stimulating factor
- concentration 10-30 ng/mL, 20-40 ng/mL, 30-50 ng/mL, 40-60 ng/mL, 50- 70 ng/mL, 60-80 ng/mL, 70-90 ng/mL, 80-100 ng/mL, 90-110 ng/mL, 100-120 ng/mL, 110-130 ng/mL, 120-140 ng/mL, 130-150 ng/mL, 140-160 ng/mL, 150-170 ng/mL, 160-180 ng/mL, 170- 190 ng/mL, 180-200 ng/mL, 190-210 ng/mL, 200-220 ng/mL, or greater than 220 ng/mL; b.
- GM-CSF granulocyte-macrophage colony-stimulating factor
- SCF stem cells factor
- TPO thrombopoietin
- concentration 10-30 ng/mL, 20-40 ng/mL, 30-50 ng/mL, 40-60 ng/mL, 50-70 ng/mL, 60-80 ng/mL, 70-90 ng/mL, 80-100 ng/mL, 90-110 ng/mL, 100-120 ng/mL, 110-130 ng/mL, 120-140 ng/mL, 130-150 ng/mL, 140-160 ng/mL, 150-170 ng/mL, 160-180 ng/mL, 170-190 ng/mL, 180-200 ng/mL, 190-210 ng/mL, 200-220 ng/mL, or greater than 220 ng/mL.
- TPO thrombopoietin
- the precursor media when the precursor media comprises granulocyte colony-stimulating factor (G-CSF) at a concentration of about 145 ng/mL. In some embodiments, when the precursor media comprises granulocyte colony-stimulating factor (G-CSF) at a concentration of about 145 ng/mL, said precursor media further comprises: a.
- G-CSF granulocyte colony-stimulating factor
- GM-CSF granulocyte-macrophage colony-stimulating factor
- concentration 10-30 ng/mL, 20-40 ng/mL, 30-50 ng/mL, 40-60 ng/mL, 50- 70 ng/mL, 60-80 ng/mL, 70-90 ng/mL, 80-100 ng/mL, 90-110 ng/mL, 100-120 ng/mL, 110-130 ng/mL, 120-140 ng/mL, 130-150 ng/mL, 140-160 ng/mL, 150-170 ng/mL, 160-180 ng/mL, 170- 190 ng/mL, 180-200 ng/mL, 190-210 ng/mL, 200-220 ng/mL, or greater than 220 ng/mL; b.
- GM-CSF granulocyte-macrophage colony-stimulating factor
- SCF stem cells factor
- TPO thrombopoietin
- concentration 10-30 ng/mL, 20-40 ng/mL, 30-50 ng/mL, 40-60 ng/mL, 50-70 ng/mL, 60-80 ng/mL, 70-90 ng/mL, 80-100 ng/mL, 90-110 ng/mL, 100-120 ng/mL, 110-130 ng/mL, 120-140 ng/mL, 130-150 ng/mL, 140-160 ng/mL, 150-170 ng/mL, 160-180 ng/mL, 170-190 ng/mL, 180-200 ng/mL, 190-210 ng/mL, 200-220 ng/mL, or greater than 220 ng/mL.
- TPO thrombopoietin
- the precursor media when the precursor media comprises granulocyte colony-stimulating factor (G-CSF) at a concentration of about 150 ng/mL. In some embodiments, when the precursor media comprises granulocyte colony-stimulating factor (G-CSF) at a concentration of about 150 ng/mL, said precursor media further comprises: a.
- SCF stem cells factor
- GM-CSF granulocyte-macrophage colony-stimulating factor
- concentration 10-30 ng/mL, 20-40 ng/mL, 30-50 ng/mL, 40-60 ng/mL, 50- 70 ng/mL, 60-80 ng/mL, 70-90 ng/mL, 80-100 ng/mL, 90-110 ng/mL, 100-120 ng/mL, 110-130 ng/mL, 120-140 ng/mL, 130-150 ng/mL, 140-160 ng/mL, 150-170 ng/mL, 160-180 ng/mL, 170- 190 ng/mL, 180-200 ng/mL, 190-210 ng/mL, 200-220 ng/mL, or greater than 220 ng/mL; b.
- GM-CSF granulocyte-macrophage colony-stimulating factor
- the precursor media comprises G-CSF at a concentration of 150 ng/mL.
- said precursor media further comprises: a. granulocyte-macrophage colony-stimulating factor (GM- CSF) at a concentration between about 75 ng/mL and 125 ng/mL; b. stem cells factor (SCF) at a concentration between about 75 ng/mL and 125 ng/mL; and/or c. thrombopoietin (TPO) at a concentration between about 75 ng/mL and 125 ng/mL.
- GM- CSF granulocyte-macrophage colony-stimulating factor
- SCF stem cells factor
- TPO thrombopoietin
- the precursor media comprises G-CSF at a concentration of about 150ng/mL
- said precursor media further comprises: a. granulocyte-macrophage colony-stimulating factor (GM-CSF) at a concentration between about 70 ng/mL, about 75 ng/mL, about 80 ng/mL, about 85 ng/mL, about 90 ng/mL, about 95 ng/mL, about 100 ng/mL, about 105 ng/mL, about 110 ng/mL, about 115 ng/mL, about 120 ng/mL, or about 125 ng/mL; b.
- GM-CSF granulocyte-macrophage colony-stimulating factor
- TPO thrombopoietin
- the precursor media can further comprise GlutaMAXTM Supplement, a basal growth media, and/or a basal growth media comprising VIVOTM- 15 medium and Stemline® II Hematopoietic Stem Cell Expansion Media.
- the precursor media comprises G-CSF at a concentration of 150 ng/mL, GM-CSF at a concentration of 100 ng/mL, SCF at a concentration of 100 ng/mL, and TPO at a concentration of 100 ng/mL
- the precursor media further comprises GlutaMAXTM Supplement at a concentration of 2mM
- a basal growth media comprising VIVOTM-15 medium and Stemline® II Hematopoietic Stem Cell Expansion Media mixed at a ratio of 1 : 1.
- the precursor media comprises G-CSF at a concentration of about 150ng/mL
- said precursor media further comprises: a. granulocyte-macrophage colony-stimulating factor (GM-CSF) at a concentration between about 70 ng/mL, about 75 ng/mL, about 80 ng/mL, about 85 ng/mL, about 90 ng/mL, about 95 ng/mL, about 100 ng/mL, about 105 ng/mL, about 110 ng/mL, about 115 ng/mL, about 120 ng/mL, or about 125 ng/mL; b.
- GM-CSF granulocyte-macrophage colony-stimulating factor
- SCF stem cells factor
- thrombopoietin at a concentration between about 70 ng/mL, about 75 ng/mL, about 80 ng/mL, about 85 ng/mL, about 90 ng/mL, about 95 ng/mL, about 100 ng/mL, about 105 ng/mL, about 110 ng/mL, about 115 ng/mL, about 120 ng/mL, or about 125 ng/mL; and/or d.
- GlutaMAXTM Supplement at a concentration of between ImM and 3mM. Gibco GlutaMAX SupplementTM is an alternative to L-glutamine, with increased stability that improves cell health.
- GlutaMAX SupplementTM is suitable for both adherent and suspension culture of mammalian cells, with no adaptation required.
- GlutaMAX SupplementTM is offered as a 200 mM L-alanyl-L-glutamine dipeptide in 0.85% NaCl. It would be understood by a skilled artisan that any formulation of L-alanyl-L-glutamine dipeptide (e.g., a generic brand or generic formulation) matching that of GlutaMAX SupplementTM, which functions in about the same manner as GlutaMAX SupplementTM can be used in the precursor media.
- a generic formulation will function in about the same manner if it promotes expansion and/or differentiation of stem cells within precursor media as GlutaMAX SupplementTM.
- GlutaMAX SupplementTM is also included in a variety of media formulations.
- any media formulation can be used, so long as it is present within the indicated concentration in the precursor media.
- any formulation of L-alanyl-L- glutamine dipeptide present in the precursor media at any concentration disclosed herein for GlutaMAX Supplement TM e.g., between ImM and 3mM, preferably about 2mM
- the L-alanyl-L-glutamine dipeptide can be in a NaCl solution, of about 0.85% NaCl (e.g, about 0.80% to about 0.90%).
- the L-alanyl-L-glutamine dipeptide can be in a NaCl solution at any concentration of NaCl that enables the L-alanyl-L- glutamine dipeptide to function in about the same manner as GlutaMAX SupplmentTM.
- the precursor media comprises G-CSF at a concentration of about 150ng/mL
- said precursor media further comprises: a. granulocyte-macrophage colony-stimulating factor (GM-CSF) at a concentration between about 70 ng/mL, about 75 ng/mL, about 80 ng/mL, about 85 ng/mL, about 90 ng/mL, about 95 ng/mL, about 100 ng/mL, about 105 ng/mL, about 110 ng/mL, about 115 ng/mL, about 120 ng/mL, or about 125 ng/mL; b.
- GM-CSF granulocyte-macrophage colony-stimulating factor
- SCF stem cells factor
- TPO thrombopoietin
- TPO thrombopoietin
- GlutaMAXTM Supplement at a concentration of about 2 mM.
- the precursor media comprises G-CSF at a concentration of about 150ng/mL
- said precursor media further comprises: a. granulocyte-macrophage colony-stimulating factor (GM-CSF) at a concentration between about 70 ng/mL, about 75 ng/mL, about 80 ng/mL, about 85 ng/mL, about 90 ng/mL, about 95 ng/mL, about 100 ng/mL, about 105 ng/mL, about 110 ng/mL, about 115 ng/mL, about 120 ng/mL, or about 125 ng/mL; b.
- GM-CSF granulocyte-macrophage colony-stimulating factor
- SCF stem cells factor
- TPO thrombopoietin
- TPO thrombopoietin
- GlutaMAXTM Supplement at a concentration of 2 mM.
- the precursor media comprises G-CSF at a concentration of about 150ng/mL
- said precursor media further comprises: a. granulocyte-macrophage colony-stimulating factor (GM-CSF) at a concentration between about 70 ng/mL, about 75 ng/mL, about 80 ng/mL, about 85 ng/mL, about 90 ng/mL, about 95 ng/mL, about 100 ng/mL, about 105 ng/mL, about 110 ng/mL, about 115 ng/mL, about 120 ng/mL, or about 125 ng/mL; b.
- GM-CSF granulocyte-macrophage colony-stimulating factor
- SCF stem cells factor
- TPO thrombopoietin
- TPO thrombopoietin
- concentration between about 70 ng/mL, about 75 ng/mL, about 80 ng/mL, about 85 ng/mL, about 90 ng/mL, about 95 ng/mL, about 100 ng/mL, about 105 ng/mL, about 110 ng/mL, about 115 ng/mL, about 120 ng/mL, or about 125 ng/mL; and/or d.
- L-alanyl-L-glutamine dipeptide at a concentration of about 2 mM.
- the precursor media comprises G-CSF at a concentration of about 150ng/mL
- said precursor media further comprises: a. granulocyte-macrophage colony-stimulating factor (GM-CSF) at a concentration between about 70 ng/mL, about 75 ng/mL, about 80 ng/mL, about 85 ng/mL, about 90 ng/mL, about 95 ng/mL, about 100 ng/mL, about 105 ng/mL, about 110 ng/mL, about 115 ng/mL, about 120 ng/mL, or about 125 ng/mL; b.
- GM-CSF granulocyte-macrophage colony-stimulating factor
- SCF stem cells factor
- the precursor cytokine mixture comprises G-CSF at a concentration of between about 100 ng/mL and 150 ng/mL
- the precursor cytokine mixture comprises G-CSF, M-CSF, SCF, and TPO.
- the precursor media comprises G-CSF at a concentration of 150 ng/mL, M-CSF at a concentration of 150 ng/mL, SCF at a concentration of 100 ng/mL, and TPO at a concentration of 100 ng/mL.
- the precursor media comprises G-CSF at a concentration of 150 ng/mL, M-CSF at a concentration of 150 ng/mL, SCF at a concentration of 100 ng/mL, and TPO at a concentration of 100 ng/mL
- the precursor media further comprises GlutaMAXTM Supplement at a concentration of 2mM
- a basal growth media comprising VIVOTM-15 medium and Stemline® II Hematopoietic Stem Cell Expansion Media mixed at a ratio of 1 : 1.
- the precursor cytokine mixture comprises G-CSF at a concentration of between about 100 ng/mL and 150 ng/mL
- the precursor cytokine mixture comprises G-CSF, M-CSF, SCF, GM- CSF, and TPO.
- the precursor media comprises G-CSF at a concentration of 150 ng/mL, M-CSF at a concentration of 150 ng/mL, SCF at a concentration of 100 ng/mL, GM-CSF at a concentration of 100 ng/mL and TPO at a concentration of 100 ng/mL.
- the precursor media comprises G-CSF at a concentration of 150 ng/mL, M-CSF at a concentration of 150 ng/mL, SCF at a concentration of 100 ng/mL, and TPO at a concentration of 100 ng/mL
- the precursor media further comprises GlutaMAXTM Supplement at a concentration of 2mM
- a basal growth media comprising VIVOTM-15 medium and Stemline® II Hematopoietic Stem Cell Expansion Media mixed at a ratio of 1 : 1.
- the precursor cytokine mixture comprises G-CSF at a concentration of between about 100 ng/mL and 150 ng/mL
- the precursor cytokine mixture comprises G-CSF, M-CSF, IL-6, IL-3, SCF, and TPO.
- the precursor media comprises G-CSF at a concentration of 150 ng/mL, M-CSF at a concentration of 150 ng/mL, IL-3 at a concentration of 50 ng/mL, IL-6 at a concentration of 100 ng/mL, SCF at a concentration of 100 ng/mL, and TPO at a concentration of 100 ng/mL.
- the precursor media comprises G-CSF at a concentration of 150 ng/mL, M-CSF at a concentration of 150 ng/mL, IL-3 at a concentration of 50 ng/mL, IL-6 at a concentration of 100 ng/mL, SCF at a concentration of 100 ng/mL, and TPO at a concentration of 100 ng/mL
- the precursor media further comprises GlutaMAXTM Supplement at a concentration of 2 mM
- a basal growth media comprising VIVOTM- 15 medium and Stemline® II Hematopoietic Stem Cell Expansion Media mixed at a ratio of 1 : 1.
- the precursor cytokine mixture comprises G-CSF at a concentration of between about 100 ng/mL and 150 ng/mL
- the precursor cytokine mixture comprises G-CSF, M-CSF, IL-3, IL-6, SCF, GM-SCF, and TPO.
- the precursor media comprises G-CSF at a concentration of 150 ng/mL, M-CSF at a concentration of 150 ng/mL, IL-3 at a concentration of 50 ng/mL, IL-6 at a concentration of 100 ng/mL, SCF at a concentration of 100 ng/mL, GM-SCF at a concentration of 100 ng/mL, and TPO at a concentration of 100 ng/mL.
- the precursor media comprises G-CSF at a concentration of 150 ng/mL, M-CSF at a concentration of 150 ng/mL, IL-3 at a concentration of 50 ng/mL, IL-6 at a concentration of 100 ng/mL, SCF at a concentration of 100 ng/mL, GM-SCF at a concentration of 100 ng/mL, and TPO at a concentration of 100 ng/mL
- the precursor media further comprises GlutaMAXTM Supplement at a concentration of 2mM, and a basal growth media comprising VIVOTM- 15 medium and Stemline® II Hematopoietic Stem Cell Expansion Media mixed at a ratio of 1 : 1.
- the concentration of the SCF in the precursor media can be at least 50 ng/mL, at least 100 ng/mL, at least 150 ng/mL, or at least 200 ng/mL.
- the precursor media comprises SCF at a concentration of 100 ng/mL.
- the concentration of the Flt3 ligand in the precursor media can be at least 50 ng/mL, at least 100 ng/mL, at least 150 ng/mL, or at least 200 ng/mL.
- the concentration of the G-CSF in the precursor media can be at least 50 ng/mL, at least 100 ng/mL, at least 150 ng/mL, or at least 200 ng/mL.
- the precursor media comprises G-CSF at a concentration of 150 ng/mL.
- the concentration of the IL-6 in the precursor media can be at least 50 ng/mL, at least 100 ng/mL, at least 150 ng/mL, or at least 200 ng/mL. In a preferred embodiment, the precursor media comprises IL-6 at a concentration of 100 ng/mL.
- the precursor cytokine mixture comprises Granulocyte-macrophage colonystimulating factor (GM-CSF)
- the concentration of the GM-CSF in the precursor media can be at least 50 ng/mL, at least 100 ng/mL, at least 150 ng/mL, or at least 200 ng/mL.
- the precursor media comprises GM-CSF at a concentration of 100 ng/mL.
- the concentration of the MCSF in the precursor media can be at least 50 ng/mL, at least 100 ng/mL, at least 150 ng/mL, or at least 200 ng/mL.
- the precursor media comprises macrophage colony-stimulating factor (MCSF) at a concentration of 150 ng/mL.
- the precursor media comprises stem cells factor (SCF), Flt3 ligand, thrombopoietin (TPO), Granulocyte colony-stimulating factor (G-CSF), and Granulocytemacrophage colony-stimulating factor (GM-CSF).
- SCF stem cells factor
- Flt3 ligand thrombopoietin
- TPO Granulocyte colony-stimulating factor
- GM-CSF Granulocytemacrophage colony-stimulating factor
- a useful concentration can be defined as a concentration of a specific cytokine that contributes to a desired effect on expansion and/or differentiation in a precursor media. For example, a cytokine at a specific concentration that is associated with an increased expansion rate of genetically modified CAR HSCs.
- the myeloid media comprises stem cells factor (SCF), Flt3 ligand, thrombopoietin (TPO), Granulocyte colony-stimulating factor (G-CSF), and Granulocytemacrophage colony-stimulating factor (GM-CSF).
- SCF stem cells factor
- Flt3 ligand thrombopoietin
- G-CSF Granulocyte colony-stimulating factor
- GM-CSF Granulocytemacrophage colony-stimulating factor
- the stem cells factor (SCF), Flt3 ligand, thrombopoietin (TPO), Granulocyte colony-stimulating factor (G-CSF), and Granulocytemacrophage colony-stimulating factor (GM-CSF) can be present within the myeloid media at any useful concentration.
- a useful concentration can be defined as a concentration of a specific cytokine that contributes to a desired effect on expansion and/or differentiation in a precursor media. For example, a cytokine at a specific concentration that is associated with an increased expansion rate of genetically modified CAR HSCs.
- Flt3 ligand can be within the precursor mixture at a concentration of at most 50 ng/mL, at most 40 ng/mL, at most 30 ng/mL, at most 20 ng/mL, at most 10 ng/mL, at most 5 ng/mL, or below 5 ng/mL.
- the precursor media comprises Flt3 ligand at a concentration of below 50 ng/mL.
- the precursor media comprises Flt3 ligand at a concentration of below 40 ng/mL.
- the precursor media comprises Flt3 ligand at a concentration of below 30 ng/mL.
- the precursor media comprises Flt3 ligand at a concentration of below 20 ng/mL.
- the precursor media comprises Flt3 ligand at a concentration of below 10 ng/mL. In some instances, the precursor media comprises Flt3 ligand at a concentration of below 5 ng/mL. In a preferred embodiment, the precursor mixture does not comprise Flt3 ligand.
- Interleukin 3 can be within the precursor mixture at a concentration of at most 50 ng/mL, at most 40 ng/mL, at most 30 ng/mL, at most 20 ng/mL, at most 10 ng/mL, at most 5 ng/mL, or below 5 ng/mL.
- the precursor media comprises IL-3 at a concentration of below 50 ng/mL.
- the precursor media comprises IL-3 at a concentration of below 40 ng/mL.
- the precursor media comprises IL-3 at a concentration of below 30 ng/mL.
- the precursor media comprises IL-3 at a concentration of below 20 ng/mL.
- the precursor media comprises IL-3 at a concentration of below 10 ng/mL. In some instances, the precursor media comprises IL-3 at a concentration of below 5 ng/mL. In a preferred embodiment, the precursor mixture does not comprise IL-3.
- Interleukin 3 can be within the precursor mixture at a concentration of at most 50 ng/mL, at most 40 ng/mL, at most 30 ng/mL, at most 20 ng/mL, at most 10 ng/mL, at most 5 ng/mL, or below 5 ng/mL.
- the precursor media comprises IL-6 at a concentration of below 50 ng/mL.
- the precursor media comprises IL-6 at a concentration of below 40 ng/mL.
- the precursor media comprises IL-6 at a concentration of below 30 ng/mL.
- the precursor media comprises IL-6 at a concentration of below 20 ng/mL.
- the precursor media comprises IL-6 at a concentration of below 10 ng/mL. In some instances, the precursor media comprises IL-6 at a concentration of below 5 ng/mL. In a preferred embodiment, the precursor mixture does not comprise IL-6.
- Granulocyte-macrophage colony-stimulating factor can be within the precursor mixture at a concentration of at most 50 ng/mL, at most 40 ng/mL, at most 30 ng/mL, at most 20 ng/mL, at most 10 ng/mL, at most 5 ng/mL, or below 5 ng/mL.
- the precursor media comprises GM-CSF at a concentration of below 50 ng/mL.
- the precursor media comprises GM-CSF at a concentration of below 40 ng/mL.
- the precursor media comprises GM-CSF at a concentration of below 30 ng/mL.
- the precursor media comprises GM-CSF at a concentration of below 20 ng/mL. In some instances, the precursor media comprises GM-CSF at a concentration of below 10 ng/mL. In some instances, the precursor media comprises GM-CSF at a concentration of below 5 ng/mL. In a preferred embodiment, the precursor mixture does not comprise any GM-CSF.
- Macrophage colony-stimulating factor can be within the precursor mixture at a concentration of at most 50 ng/mL, at most 40 ng/mL, at most 30 ng/mL, at most 20 ng/mL, at most 10 ng/mL, at most 5 ng/mL, or below 5 ng/mL.
- the precursor media comprises M-CSF at a concentration of below 50 ng/mL.
- the precursor media comprises M-CSF at a concentration of below 40 ng/mL.
- the precursor media comprises M-CSF at a concentration of below 30 ng/mL.
- the precursor media comprises M-CSF at a concentration of below 20 ng/mL.
- the precursor media comprises M-CSF at a concentration of below 10 ng/mL. In some instances, the precursor media comprises M-CSF at a concentration of below 5 ng/mL. In a preferred embodiment, the precursor mixture does not comprise any M-CSF.
- the precursor media disclosed herein can comprise at least one cytokine selected from the following: stem cells factor (SCF), Flt3 ligand, thrombopoietin (TPO), Interleukin-3 (IL-3); Granulocyte colony-stimulating factor (G-CSF), Interleukin-6 (IL-6), Granulocyte-macrophage colony-stimulating factor (GM-CSF), and Macrophage colony-stimulating factor (MCSF).
- SCF stem cells factor
- Flt3 ligand Flt3 ligand
- TPO thrombopoietin
- IL-3 Interleukin-3
- G-CSF Granulocyte colony-stimulating factor
- IL-6 Interleukin-6
- GM-CSF Granulocyte-macrophage colony-stimulating factor
- MCSF Macrophage colony-stimulating factor
- the precursor media comprises at least two cytokines selected from the following cytokines: stem cells factor (SCF), Flt3 ligand, thrombopoietin (TPO), Interleukin-3 (IL-3); Granulocyte colony-stimulating factor (G-CSF), Interleukin-6 (IL-6), Granulocyte-macrophage colony-stimulating factor (GM-CSF), and Macrophage colony-stimulating factor (MCSF).
- SCF stem cells factor
- Flt3 ligand Flt3 ligand
- TPO thrombopoietin
- IL-3 Interleukin-3
- G-CSF Granulocyte colony-stimulating factor
- IL-6 Interleukin-6
- GM-CSF Granulocyte-macrophage colony-stimulating factor
- MCSF Macrophage colony-stimulating factor
- the precursor media comprises at least three cytokines selected from the following cytokines: stem cells factor (SCF), Flt3 ligand, thrombopoietin (TPO), Interleukin-3 (IL-3); Granulocyte colony-stimulating factor (G-CSF), Interleukin-6 (IL-6), Granulocyte-macrophage colony-stimulating factor (GM-CSF), and Macrophage colony-stimulating factor (MCSF).
- SCF stem cells factor
- Flt3 ligand Flt3 ligand
- TPO thrombopoietin
- IL-3 Interleukin-3
- G-CSF Granulocyte colony-stimulating factor
- IL-6 Interleukin-6
- GM-CSF Granulocyte-macrophage colony-stimulating factor
- MCSF Macrophage colony-stimulating factor
- the precursor media comprises at least four cytokines selected from the following cytokines: stem cells factor (SCF), Flt3 ligand, thrombopoietin (TPO), Interleukin-3 (IL-3); Granulocyte colony-stimulating factor (G-CSF), Interleukin-6 (IL-6), Granulocyte-macrophage colony-stimulating factor (GM-CSF), and Macrophage colony-stimulating factor (MCSF).
- SCF stem cells factor
- Flt3 ligand Flt3 ligand
- TPO thrombopoietin
- IL-3 Interleukin-3
- G-CSF Granulocyte colony-stimulating factor
- IL-6 Interleukin-6
- GM-CSF Granulocyte-macrophage colony-stimulating factor
- MCSF Macrophage colony-stimulating factor
- the precursor media comprises four cytokines selected from the following cytokines: stem cells factor (SCF), Flt3 ligand, thrombopoietin (TPO), Interleukin-3 (IL-3); Granulocyte colony-stimulating factor (G-CSF), Interleukin-6 (IL-6), Granulocyte-macrophage colonystimulating factor (GM-CSF), and Macrophage colony-stimulating factor (MCSF).
- the precursor cytokine mixture comprises G-CSF, GM-CSF, SCF, and TPO.
- the G-CSF can be present at a concentration between about 100 ng/mL and 200 ng/mL. In some embodiments, G-CSF is present within the precursor media at a concentration between about 100 ng/mL and 150 ng/mL. In such instances, the precursor media can further comprise at least one cytokine selected from the following: GM-CSF, SCF, and TPO. When the precursor media comprises G-CSF at a concentration between about 100 ng/mL and 150 ng/mL, said precursor media can further comprise GM-CSF, SCF, TPO, and any combination thereof.
- the precursor media can comprise GM-CSF at a concentration between about 75 ng/mL and 125 ng/mL.
- the precursor media can comprise SCF at a concentration between about 75 ng/mL and 125 ng/mL.
- the precursor media can comprise TPO at a concentration between about 75 ng/mL and 125 ng/mL.
- the precursor media comprises G-CSF, GM-CSF, SCF, and TPO.
- the precursor media comprises G-CSF at a concentration of 150 ng/mL, GM-CSF at a concentration of 100 ng/mL, SCF at a concentration of 100 ng/mL, and TPO at a concentration of 100 ng/mL.
- the precursor media disclosed herein can comprise a basal growth medium.
- the basal growth medium can be any medium that supports growth, expansion and/or differentiation of precursor cells.
- the basal growth medium can comprise glucose, salts, amino acids, and vitamins.
- the basal growth medium can contain a Roswell Park Memorial Institute (RPMI) 1640 medium, X-VIVOTM-15 medium, CTSTM OPTMIZERTM Serum Free Medium, CTSTM OPTMIZERTM Pro Serum Free Medium, and/or IMMUNOCULTTM-XF T Cell Expansion Medium.
- the basal growth medium comprises the RPMI 1640 medium with ATCC modification (Gibco A1049101).
- the basal growth media comprises VIVOTM- 15 medium.
- the basal growth media comprises Stemline® II Hematopoietic Stem Cell Expansion Media.
- the VIVOTM-15 medium can be combined or mixed with any other basal growth media at any ratio that is particularly effective.
- the basal growth media can comprise VIVOTM-15 medium and Stemline® II Hematopoietic Stem Cell Expansion Media mixed at a ratio.
- the precursor media can comprise a basal growth media comprising VIVOTM-15 medium and Stemline® II Hematopoietic Stem Cell Expansion Media mixed at a ratio of 1 : 1.
- the precursor media can comprise a culture medium.
- Any culture media known in the art that can support the growth, proliferation, expansion and/or differentiation of precursor cells within the precursor media is envisaged as being used with the precursor media disclosed herein.
- Cell culture methods including hematopoietic stem cell culture methods, are widely available in the art, as described, for example, in Yadav, Pooja et al. “Hematopoietic Stem Cells Culture, Expansion and Differentiation: An Insight into Variable and Available Media.” International journal of stem cells vol. 13,3 (2020): 326-334, the contents of which are incorporated herein by reference in their entirety.
- the myeloid media can further comprise GlutaMAXTM Supplement.
- the myeloid media can comprise a myeloid cytokine mixture, a basal growth medium and GlutaMAXTM Supplement.
- the GlutaMAXTM Supplement can be present in the myeloid media at a concentration of between 0.5 mM, 1 mM, 1.5 mM, 2 mM, 2.5 mM or 3 mM of the myeloid media.
- a myeloid-derived cell from a precursor cell, such as a stem cell and/or a CD34+ cell.
- the stem cell is a hematopoietic stem cell.
- the methods of manufacturing a myeloid-derived cell disclosed herein can comprise the step of contacting a precursor cell with a precursor media comprising a precursor cytokine mixture, whereby the number of precursor cell(s) increases to generate a cell population comprising a plurality of precursor cells.
- Also disclosed herein are methods of manufacturing a myeloid-derived cell disclosed herein comprising the step of contacting a precursor cell with a precursor media comprising a precursor cytokine mixture, whereby the number of precursor cell(s) increases to generate a cell population comprising a plurality of precursor cells, wherein said precursor cell is a HSC, whereby upon contact of said HSC with said precursor media, said HSC differentiates into a myeloid-lineage cell to generate a myeloid-derived cell and/or a mixed cell population comprising a plurality of myeloid-derived cells and a plurality of HSC cells.
- Also disclosed herein are methods of manufacturing a myeloid-derived cell disclosed herein comprising the step of contacting a precursor cell with a precursor media comprising a precursor cytokine mixture, whereby the number of precursor cell(s) increases to generate a cell population comprising a plurality of precursor cells, wherein said precursor cell is a stem cell, wherein the method further comprises the step of contacting said cell population comprising a plurality of precursor cells with a myeloid media comprising a myeloid cytokine mixture, whereby said precursor cell(s) differentiates into a myeloid-lineage cell to generate a myeloid-derived cell and/or a mixed cell population comprising a plurality of myeloid-derived cells and/or a plurality of precursor cells.
- Also disclosed herein are methods of manufacturing a myeloid-derived cell disclosed herein comprising the step of contacting a precursor cell with a precursor media comprising a precursor cytokine mixture, whereby the number of precursor cell(s) increases to generate a cell population comprising a plurality of precursor cells, wherein said precursor cell is a HSC, whereby upon contact of said HSC with said precursor media, said HSC differentiates into a myeloid-lineage cell to generate a myeloid-derived cell and/or a mixed cell population comprising a plurality of myeloid-derived cells and a plurality of HSC cells, wherein the method further comprises the step of: contacting said mixed cell population with a myeloid media comprising a myeloid cytokine mixture, whereby upon contact with said myeloid media: a. the ratio of said myeloid-derived cells to said precursor cells in said mixed cell population is increased.
- any of the above methods described herein can involve contacting a stem cell comprising a CAR (i.e., a precursor CAR cell) with a precursor media to increase to generate a cell population comprising a plurality of precursor cells.
- the precursor cell can be an HSC.
- Such methods can further comprise differentiating said precursor CAR cell into a myeloid-derived CAR cell using any of the methods and/or steps described herein to generate a cell population comprising a plurality of myeloid-derived CAR cells.
- the methods of manufacturing a myeloid-derived cell disclosed herein can comprise the step of contacting a precursor cell with a precursor media comprising a precursor cytokine mixture, whereby the number of precursor cell(s) increases to generate a cell population comprising a plurality of precursor cells, wherein said precursor cell is a stem cell, whereby upon contact of said precursor cell(s) with said precursor media, said precursor cell(s) differentiates into a myeloid- lineage cell to generate a myeloid-derived cell and/or a mixed cell population comprising a plurality of myeloid-derived cells and a plurality of precursor cells.
- the method can further comprise the step of contacting said mixed cell population with a myeloid media comprising a myeloid cytokine mixture, whereby upon contact with said myeloid media: a. the ratio of said myeloid-derived cells to said precursor cells in said mixed cell population is increased.
- compositions and methods that are particularly effective at promoting the differentiation of precursor cells, such as stem cells.
- the stem cells can be CD34+ and/or hematopoietic stem cells.
- the methods and compositions disclosed herein are effective at promoting the differentiation of stem cells into myeloid cells.
- the stem cells comprise a CAR and are differentiated into a myeloid-derived CAR cell.
- the stem cells are hematopoietic stem cells.
- the disclosed compositions and methods can effectively promote the differentiation of a hematopoietic stem cell into a myeloid lineage cell.
- the precursor cell can comprise any CAR available in the art.
- the precursor cell can be comprised within a cell population comprising at least one precursor cell.
- the cell population comprises a plurality of precursor cells.
- the plurality of precursor cells can comprise precursor cells comprising a CAR disclosed herein.
- a genetically modified myeloid-derived CAR cell from a precursor cell, such as a hematopoietic stem cell, wherein the precursor cell comprising a CAR (i.e., precursor CAR cell) is expanded by contacting said precursor CAR cell with a precursor media comprising a precursor cytokine mixture, whereby the number of precursor CAR cell(s) increases to generate a population of precursor cells (e.g., stem cells) comprising a plurality of precursor CAR cells.
- precursor cell such as a hematopoietic stem cell
- the method further comprises the step of contacting said precursor CAR cell(s) with a myeloid media comprising a myeloid cytokine mixture, whereby said precursor cell differentiates into a myeloid-lineage cell to generate a myeloid-derived CAR cell or a cell population comprising a plurality of myeloid-derived CAR cells, wherein said cell population is a mixed population comprising said precursor CAR cells and said myeloid-derived CAR cells.
- a genetically modified myeloid-derived CAR cell from a precursor cell, such as a hematopoietic stem cell, wherein the precursor cell comprising a CAR (i.e., precursor CAR cell) is expanded by contacting said precursor CAR cell with a precursor media comprising a precursor cytokine mixture, whereby the number of precursor CAR cell(s) increases to generate a population of precursor cells (e.g., stem cells) comprising a plurality of precursor CAR cells, further comprising the step of contacting said precursor CAR cell(s) with a myeloid media comprising a myeloid cytokine mixture, whereby said precursor cell differentiates into a myeloid-lineage cell to generate a myeloid-derived CAR cell or a cell population comprising a plurality of myeloid-derived CAR cells, wherein said cell population is a mixed population comprising said precursor CAR cells and said myeloid-derived CAR cells.
- a precursor cell such as a hematopoietic
- precursor cell such as a hematopoietic stem cell
- the cell population is a mixed population comprising said myeloid- derived CAR cells and a plurality of precursor CAR cells.
- the method further comprises the step of contacting said precursor CAR cell, myeloid-derived CAR cell, and/or said population comprising precursor CAR cells and said myeloid-derived CAR cells with a myeloid media comprising a myeloid cytokine mixture, whereby said ratio of myeloid-derived CAR cells relative to precursor CAR cells within said population is increased.
- precursor cell such as a hem
- precursor cell such as a hematopoietic stem cell
- the cell population is a mixed population comprising said myeloid- derived CAR cells and a plurality of precursor CAR cells
- the method further comprises the step of contacting said precursor CAR cell, myeloid-derived CAR cell, and/or said population comprising precursor CAR cells and said myeloid-derived CAR cells with a myeloid media comprising a myeloid cytokine mixture, whereby said ratio of myeloid-derived CAR cells relative to precursor CAR cells within said population is increased.
- the methods disclosed herein comprise differentiation of at least one precursor cell.
- Said at least one precursor cell can be comprised with in a cell population.
- the cell population disclosed herein can comprise a plurality of precursor cell.
- the methods disclosed herein can comprise differentiation of a plurality of precursor cells with a cell population.
- a control can include a method that does not use the media disclosed herein.
- the method of manufacturing a genetically modified myeloid-derived CAR cell disclosed herein can further comprise the step of differentiating genetically modified precursor CAR cell(s) in a myeloid media comprising a myeloid cytokine mixture to generate a myeloid-derived CAR cell or a population comprising a plurality of genetically modified myeloid-derived CAR cells.
- the differentiating step disclosed herein is particularly effective at promoting the differentiation of a genetically modified precursor CAR cell, or a plurality of genetically modified precursor CAR cells, toward a myeloid-derived CAR cell, or a plurality of genetically modified myeloid-derived CAR cells, respectively.
- the step of differentiating the genetically modified precursor CAR cell(s) in a myeloid media occurs after the precursor CAR cell(s) has started expanding. In some instances, the step of differentiating the genetically modified precursor CAR cell(s) in a myeloid media occurs after the precursor CAR cell(s) has started differentiating.
- the precursor CAR cell(s) has started differentiating in a precursor media comprising a precursor cytokine mixture.
- the step of differentiating the genetically modified precursor CAR cell(s) in a myeloid media further promotes the differentiation of the genetically modified precursor CAR cell(s) toward the myeloid lineage.
- the step of differentiating the genetically modified precursor CAR cell(s) in a myeloid media is particularly effective at promoting the differentiation of the genetically modified precursor CAR cell(s) toward the myeloid lineage.
- the precursor media can be removed prior to addition of the myeloid media.
- the removal of the precursor media serves to remove all contents of the precursor media, including the precursor cytokine mixture.
- Any method of removing the precursor media comprising the precursor cytokine mixture, prior to the addition of the myeloid media is envisaged within the method.
- removing the precursor media comprising the precursor cytokine mixture can for example, involve washing the cells. In some instances, the cells are washed with a sterile solution. When the cells are washed with a sterile solution, centrifugation can then be utilized to separate the cells from the sterile solution.
- the cells can be suspended at a desired concentration in for example, a myeloid media comprising a myeloid cytokine mixture. Therefore, in some methods disclosed herein, after the genetically modified precursor CAR cells are expanded in a precursor media comprising a cytokine mixture, the cells are removed from the precursor media and suspended in a myeloid media comprising a myeloid cytokine mixture. Within the myeloid media, the cells can be mixed to promote differentiation.
- Differentiation of genetically modified precursor CAR cells can increase the amount of the differentiated cells by about 10 fold, 20 fold, 30 fold, 40 fold, 50 fold, 60 fold, 70 fold, 80 fold, 90 fold, 100 fold, 200 fold, 300 fold, 400 fold, 500 fold, 600 fold, 700 fold, 800 fold, 900 fold, 1000 fold, 2000 fold, 3000 fold, 4000 fold, 5000 fold, 6000 fold, 7000 fold, 8000 fold, 9000 fold, 10,000 fold, 100,000 fold, 1,000,000 fold, 10,000,000 fold, or greater.
- the number of differentiated cells within a population increases, the number of undifferentiated cells within the population decreases.
- genetically modified precursor CAR cell(s) that are differentiated in a myeloid media increases, the number of genetically modified precursor CAR cell(s) that are undifferentiated in the myeloid media decreases.
- Differentiation of genetically modified precursor CAR cells can increase the ratio of the differentiated cells to undifferentiated cells within a population by a factor of 10 fold, 20 fold, 30 fold, 40 fold, 50 fold, 60 fold, 70 fold, 80 fold, 90 fold, 100 fold, 200 fold, 300 fold, 400 fold, 500 fold, 600 fold, 700 fold, 800 fold, 900 fold, 1000 fold, 2000 fold, 3000 fold, 4000 fold, 5000 fold, 6000 fold, 7000 fold, 8000 fold, 9000 fold, 10,000 fold, 100,000 fold, 1,000,000 fold, 10,000,000 fold, or greater.
- the differentiated cells of the present disclosure are preferential differentiated toward the myeloid cell lineage.
- the step of differentiating a genetically modified precursor CAR cell in a myeloid media generates a myeloid-derived CAR cell.
- the step of differentiating a genetically modified precursor CAR cell(s) in a myeloid media generates a plurality of genetically modified myeloid-derived CAR cells.
- the ratio of precursor (e.g., stem cells) within the plurality is decreased relative to myeloid-derived cells.
- the ratio of CD34+ cells decreases as the plurality of precursor CAR cells differentiates. Differentiation can decrease the amount of CD34+ cells within a plurality of genetically modified precursor CAR cells or a plurality of genetically modified myeloid-derived cells.
- Differentiation of genetically modified precursor CAR cells can decrease the amount of CD34+ cells within a plurality of genetically modified precursor CAR cells or a plurality of genetically modified myeloid-derived cells by about 10 fold, 20 fold, 30 fold, 40 fold, 50 fold, 60 fold, 70 fold, 80 fold, 90 fold, 100 fold, 200 fold, 300 fold, 400 fold, 500 fold, 600 fold, 700 fold, 800 fold, 900 fold, 1000 fold, 2000 fold, 3000 fold, 4000 fold, 5000 fold, 6000 fold, 7000 fold, 8000 fold, 9000 fold, 10,000 fold, 100,000 fold, 1,000,000 fold, 10,000,000 fold, or greater.
- the term “differentiation” when used in the present disclosure refers to an increase in the amount or number of surface markers on at least one cell (e.g., a precursor cell), wherein said surface markers are myeloid-associated markers.
- a “myeloid-associated marker” can be any surface marker that is known and available in the art for identifying and/or isolating a cell within the myeloid lineage. A person having skill in the art would understand how to determine if a cell was in the myeloid lineage and to identify a surface marker specific for said cell.
- Exemplary, non-limiting, myeloid-associated markers can include: CD1 lb, CD14, CD13, and CD15.
- a myeloid cell can have more than one myeloid-associated marker.
- One key aspect of the differentiation step is to increase the number of myeloid-derived cells within a myeloid media.
- the myeloid-derived cells are present within a population, such as a mixed population comprising a plurality of precursor CAR cells and myeloid-derived CAR cells.
- the differentiation step increases the number of myeloid-derived cells in a population comprising a plurality of precursor CAR cells.
- the differentiation step of the methods disclosed herein can increase the amount of the differentiated cells by about 10 fold, 20 fold, 30 fold, 40 fold, 50 fold, 60 fold, 70 fold, 80 fold, 90 fold, 100 fold, 200 fold, 300 fold, 400 fold, 500 fold, 600 fold, 700 fold, 800 fold, 900 fold, 1000 fold, 2000 fold, 3000 fold, 4000 fold, 5000 fold, 6000 fold, 7000 fold, 8000 fold, 9000 fold, 10,000 fold, 100,000 fold, 1,000,000 fold, 10,000,000 fold, or greater.
- differentiation can increase the amount of myeloid-derived CAR cells between 10-fold to 100-fold, between 20-fold to 200-fold, between 30-fold to 300-fold, between 40-fold to 400- fold, between 50-fold to 500-fold, between 60-fold to 600-fold, between 70-fold to 700-fold, between 80-fold to 800-fold, between 90-fold to 900-fold, between 100-fold to 1000-fold, or greater than 1000- fold.
- differentiated cells e.g., myeloid cells
- undifferentiated cells e.g., stem cells
- genetically modified precursor CAR cell(s) that are differentiated in a myeloid media increases, the number of genetically modified precursor CAR cell(s) that are undifferentiated in the myeloid media decreases.
- Differentiation of genetically modified precursor CAR cells can increase the ratio of the differentiated cells (i.e., myeloid-derived cells) to undifferentiated cells (i.e., stem cells) within a population by a factor of 10 fold, 20 fold, 30 fold, 40 fold, 50 fold, 60 fold, 70 fold, 80 fold, 90 fold, 100 fold, 200 fold, 300 fold, 400 fold, 500 fold, 600 fold, 700 fold, 800 fold, 900 fold, 1000 fold, 2000 fold, 3000 fold, 4000 fold, 5000 fold, 6000 fold, 7000 fold, 8000 fold, 9000 fold, 10,000 fold, 100,000 fold, 1,000,000 fold, 10,000,000 fold, or greater.
- the precursor CAR cell(s) is maintained in said myeloid medium until the number of cells is at least 10 4 , at least 10 5 , at least 10 6 , at least 10 7 , at least 10 8 , at least 10 9 , at least 10 10 , at least 10 11 , at least 10 12 , at least 10 13 , at least 10 14 , at least 10 15 , at least 10 16 , at least 10 17 , at least 10 18 , at least 10 19 , at least IO 20 , or greater than IO 20 .
- the precursor CAR cell(s) is maintained in said myeloid medium until the number of cells within the generated population is at least 10 4 , at least 10 5 , at least 10 6 , at least 10 7 , at least 10 8 , at least 10 9 , at least 10 10 , at least 10 11 , at least 10 12 , at least 10 13 , at least 10 14 , at least 10 15 , at least 10 16 , at least 10 17 , at least 10 18 , at least 10 19 , at least IO 20 , or greater than IO 20 .
- the precursor CAR cell(s) is maintained in said myeloid medium to generate a population of cells comprising a plurality of precursor CAR cells and/or a plurality of myeloid-derived CAR cells, wherein the number of cells within the population is at least 10 4 , at least 10 5 , at least 10 6 , at least 10 7 , at least 10 8 , at least 10 9 , at least 10 10 , at least 10 11 , at least 10 12 , at least IO 13 , at least 10 14 , at least IO 15 , at least 10 16 , at least 10 17 , at least 10 18 , at least 10 19 , at least IO 20 , or greater than IO 20 .
- the precursor CAR cell(s) is maintained in said myeloid medium to generate a population of cells comprising a plurality of precursor CAR cells and/or a plurality of myeloid-derived CAR cells, wherein the number of myeloid-derived CAR cells within the population is at least 10, at least 10 2 , at least 10 3 , at least 10 4 , at least 10 5 , at least 10 6 , at least 10 7 , at least 10 8 , at least 10 9 , at least IO 10 , at least 10 11 , at least 10 12 , at least 10 13 , at least 10 14 , at least 10 15 , at least 10 16 , at least 10 17 , at least 10 18 , at least 10 19 , at least IO 20 , or more than IO 20 higher than the number of precursor CAR cells.
- the precursor CAR cell(s) is maintained in said myeloid medium to generate a population of cells comprising a plurality of precursor CAR cells and/or a plurality of myeloid-derived CAR cells, wherein the ratio of precursor CAR cells to myeloid-derived CAR cells within the population is at least 1 : 10 (1 precursor CAR cell to 10 myeloid-derived CAR cells), at least 1 : 10 2 , at least 1 : 10 3 , at least l: 10 4 , at least 1 : 10 5 , at least 1 : 10 6 , at least l: 10 7 , at least l :10 8 , at least 1 : 10 9 , at least 1 : IO 10 , at least lilO 11 , at least 1 : 10 12 , at least 1 : 10 13 , at least l: 10 14 , at least 1 : 10 15 , at least 1 : 10 16 , at least 1 : 10 17 , at least 1 : 10 18 ,
- the precursor CAR cell(s) is maintained in the myelid medium for at least 2, at least 3, at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, at least 10, at least 11, or at least 12 days. In some embodiments, the precursor CAR cell(s), and/or population comprising a plurality of precursor CAR cells and/or plurality of myeloid-derived CAR cells, are maintained in myeloid media for between 9-11 days. In some embodiments, the precursor CAR cell(s), and/or population comprising a plurality of precursor CAR cells and/or plurality of myeloid-derived CAR cells, are maintained in precursor media for 10 days.
- the differentiated cells of the present disclosure are preferentially differentiated toward the myeloid cell lineage.
- the step of differentiating a genetically modified precursor CAR cell in a myeloid media generates a myeloid-derived CAR cell.
- the step of differentiating a genetically modified precursor CAR cell(s) in a myeloid media generates a plurality of genetically modified myeloid-derived CAR cells.
- the ratio of precursor (e.g., stem cells) within the plurality is decreased relative to myeloid-derived cells.
- the ratio of CD34+ cells decreases as the plurality of precursor CAR cells differentiates. Differentiation can decrease the amount of CD34+ cells within a plurality of genetically modified precursor CAR cells or a plurality of genetically modified myeloid-derived cells.
- Differentiation of genetically modified precursor CAR cells can decrease the amount of CD34+ cells within a plurality of genetically modified precursor CAR cells or a plurality of genetically modified myeloid-derived cells by about 10 fold, 20 fold, 30 fold, 40 fold, 50 fold, 60 fold, 70 fold, 80 fold, 90 fold, 100 fold, 200 fold, 300 fold, 400 fold, 500 fold, 600 fold, 700 fold, 800 fold, 900 fold, 1000 fold, 2000 fold, 3000 fold, 4000 fold, 5000 fold, 6000 fold, 7000 fold, 8000 fold, 9000 fold, 10,000 fold, 100,000 fold, 1,000,000 fold, 10,000,000 fold, or greater.
- Myeloid cells can be identified and/or isolated based on cell surface markers.
- myeloid cells within the myeloid lineage can be identified based on specific cell surface markers.
- exemplary, non-limiting, surface markers for myeloid cells include: CD13, CD14, CD15 and CD1 lb. Accordingly, during the expansion step, the number of cells expression myeloid-associated cell surface markers can be expected to increase, while the number of cells expressing a common stem cell surface marker (i.e., CD34) can be expected to decrease as differentiation occurs.
- the differentiation step of the methods disclosed herein can increase the amount of CD1 lb+ cells within a population comprising a plurality of precursor CAR cells or a plurality of myeloid- derived cells.
- Differentiation of genetically modified precursor CAR cells can increase the amount of CD1 lb+ cells within a population comprising a plurality of precursor CAR cells and/or a plurality of myeloid-derived CAR cells by about 10 fold, 20 fold, 30 fold, 40 fold, 50 fold, 60 fold, 70 fold, 80 fold, 90 fold, 100 fold, 200 fold, 300 fold, 400 fold, 500 fold, 600 fold, 700 fold, 800 fold, 900 fold, 1000 fold, 2000 fold, 3000 fold, 4000 fold, 5000 fold, 6000 fold, 7000 fold, 8000 fold, 9000 fold, 10,000 fold, 100,000 fold, 1,000,000 fold, 10,000,000 fold, or greater.
- a precursor CAR cell, or population comprising a plurality of precursor CAR cells, or a population comprising a plurality of precursor CAR cells and a plurality of myeloid-derived CAR cells is maintained in a myeloid media until the number of CD1 lb+ cells within the population is increased between 10 fold to 100 fold, between 20 fold and 200 fold, between 30 fold and 300 fold, between 40 fold and 400 fold, between 50 fold and 500 fold, between 60 fold and 600 fold, between 70 fold and 700 fold, between 80 fold and 800 fold, between 90 fold and 900 fold, between 100 fold and 10,000 fold, between 1000 fold and 100,000 fold, between 10,000 fold and 1,000,000 fold, between 100,000 fold and 10,000,000 fold , between 10 5 fold and 10 7 fold, between 10 6 fold and 10 8 fold, between 10 7 fold and 10 9 fold, 10 8 fold and 10 10 fold, 10 9 fold and 10 11 fold, 10 10 fold and 10 12 fold, 10 11 fold and 10 13 fold, 10 12 fold and 10 14 fold, 10 13 fold and 10 15 fold, 10 14 fold and 10
- the differentiation step of the methods disclosed herein can increase the amount of CD14+ cells within a population comprising a plurality of precursor CAR cells or a plurality of myeloid- derived CAR cells.
- Differentiation of precursor CAR cells can increase the amount of CD14+ cells within a population comprising a plurality of precursor CAR cells and/or a plurality of myeloid- derived CAR cells by about 10 fold, 20 fold, 30 fold, 40 fold, 50 fold, 60 fold, 70 fold, 80 fold, 90 fold, 100 fold, 200 fold, 300 fold, 400 fold, 500 fold, 600 fold, 700 fold, 800 fold, 900 fold, 1000 fold, 2000 fold, 3000 fold, 4000 fold, 5000 fold, 6000 fold, 7000 fold, 8000 fold, 9000 fold, 10,000 fold, 100,000 fold, 1,000,000 fold, 10,000,000 fold, or greater.
- a precursor CAR cell, or population comprising a plurality of precursor CAR cells, or a population comprising a plurality of precursor CAR cells and a plurality of myeloid-derived CAR cells is maintained in a myeloid media until the number of CD14+ cells within the population is increased between 10 fold to 100 fold, between 20 fold and 200 fold, between 30 fold and 300 fold, between 40 fold and 400 fold, between 50 fold and 500 fold, between 60 fold and 600 fold, between 70 fold and 700 fold, between 80 fold and 800 fold, between 90 fold and 900 fold, between 100 fold and 10,000 fold, between 1000 fold and 100,000 fold, between 10,000 fold and 1,000,000 fold, between 100,000 fold and 10,000,000 fold , between 10 5 fold and 10 7 fold, between 10 6 fold and 10 8 fold, between 10 7 fold and 10 9 fold, 10 8 fold and 10 10 fold, 10 9 fold and 10 11 fold, 10 10 fold and 10 12 fold, 10 11 fold and 10 13 fold, 10 12 fold and 10 14 fold, 10 13 fold and 10 15 fold, 10 14 fold and 10 16 fold,
- the differentiation step of the methods disclosed herein can increase the amount of CD 13+ cells within a population comprising a plurality of precursor CAR cells and/or a plurality of myeloid- derived CAR cells.
- Differentiation of precursor CAR cells can increase the amount of CD 13+ cells within a population comprising a plurality of precursor CAR cells and/or a plurality of myeloid- derived CAR cells by about 10 fold, 20 fold, 30 fold, 40 fold, 50 fold, 60 fold, 70 fold, 80 fold, 90 fold, 100 fold, 200 fold, 300 fold, 400 fold, 500 fold, 600 fold, 700 fold, 800 fold, 900 fold, 1000 fold, 2000 fold, 3000 fold, 4000 fold, 5000 fold, 6000 fold, 7000 fold, 8000 fold, 9000 fold, 10,000 fold, 100,000 fold, 1,000,000 fold, 10,000,000 fold, or greater.
- a precursor CAR cell, or population comprising a plurality of precursor CAR cells, or a population comprising a plurality of precursor CAR cells and a plurality of myeloid-derived CAR cells is maintained in a myeloid media until the number of CD 13+ cells within the population is increased between 10 fold to 100 fold, between 20 fold and 200 fold, between 30 fold and 300 fold, between 40 fold and 400 fold, between 50 fold and 500 fold, between 60 fold and 600 fold, between 70 fold and 700 fold, between 80 fold and 800 fold, between 90 fold and 900 fold, between 100 fold and 10,000 fold, between 1000 fold and 100,000 fold, between 10,000 fold and 1,000,000 fold, between 100,000 fold and 10,000,000 fold , between 10 5 fold and 10 7 fold, between 10 6 fold and 10 8 fold, between 10 7 fold and 10 9 fold, 10 8 fold and 10 10 fold, 10 9 fold and 10 11 fold, 10 10 fold and 10 12 fold, 10 11 fold and 10 13 fold, 10 12 fold and 10 14 fold, 10 13 fold and 10 15 fold, 10 14 fold and 10 16 fold,
- the differentiation step of the methods disclosed herein can increase the amount of CD 15+ cells within a population comprising a plurality of precursor CAR cells and/or a plurality of myeloid-derived CAR cells.
- Differentiation of genetically modified precursor CAR cells can increase the amount of CD 15+ cells within a plurality of genetically modified precursor CAR cells or a plurality of genetically modified myeloid-derived cells by about 10 fold, 20 fold, 30 fold, 40 fold, 50 fold, 60 fold, 70 fold, 80 fold, 90 fold, 100 fold, 200 fold, 300 fold, 400 fold, 500 fold, 600 fold, 700 fold, 800 fold, 900 fold, 1000 fold, 2000 fold, 3000 fold, 4000 fold, 5000 fold, 6000 fold, 7000 fold, 8000 fold, 9000 fold, 10,000 fold, 100,000 fold, 1,000,000 fold, 10,000,000 fold, or greater.
- a precursor CAR cell, or population comprising a plurality of precursor CAR cells, or a population comprising a plurality of precursor CAR cells and a plurality of myeloid-derived CAR cells is maintained in a myeloid media until the number of CD 15+ cells within the population is increased between 10 fold to 100 fold, between 20 fold and 200 fold, between 30 fold and 300 fold, between 40 fold and 400 fold, between 50 fold and 500 fold, between 60 fold and 600 fold, between 70 fold and 700 fold, between 80 fold and 800 fold, between 90 fold and 900 fold, between 100 fold and 10,000 fold, between 1000 fold and 100,000 fold, between 10,000 fold and 1,000,000 fold, between 100,000 fold and 10,000,000 fold , between 10 5 fold and 10 7 fold, between 10 6 fold and 10 8 fold, between 10 7 fold and 10 9 fold, 10 8 fold and 10 10 fold, 10 9 fold and 10 11 fold, 10 10 fold and 10 12 fold, 10 11 fold and 10 13 fold, 10 12 fold and 10 14 fold, 10 13 fold and 10 15 fold, 10 14 fold and 10 16 fold,
- the amount of cell surface marker expression within a population comprising a plurality of precursor CAR cells and/or a plurality of myeloid-derived CAR cells can be used to determine how long to maintain the differentiation step of the method of manufacturing a genetically modified myeloid-derived CAR cells disclosed herein.
- the differentiation step is maintained until a certain percentage of the population comprising a plurality of genetically modified precursor CAR cells and/or genetically modified myeloid-derived CAR cells expresses a specific marker.
- the differentiation step is maintained until a certain percentage of the population comprising a plurality of genetically modified precursor CAR cells and/or plurality of genetically modified myeloid-derived cells expresses at least a specific marker.
- the differentiation step is maintained until a certain percentage of the population comprising a plurality of genetically modified precursor CAR cells and/or plurality of genetically modified myeloid-derived CAR cells expresses at least two specific markers. In some instances, the differentiation step is maintained until a certain percentage of the population comprising a plurality of genetically modified precursor CAR cells and/or plurality of genetically modified myeloid-derived CAR cells expresses at least three specific markers. In some instances, the differentiation step is maintained until a certain percentage of the population comprising a plurality of genetically modified precursor CAR cells and/or plurality of genetically modified myeloid-derived CAR cells expresses at least more than three different specific markers.
- any reference to “a precursor cell” that is, or has been, maintained in a myeloid media for a period of time after which said precursor cell that was initially contacted with said myeloid media would reasonably be expected to have differentiated into a myeloid-derived cell is intended to encompass any cell population that arises as a result of said precursor cell being maintained in myeloid.
- a cell population would be understood to arise as a result of the precursor cell if, as described above, said population comprises at least one cell that was generated from, for example, at least one cell differentiation event of said precursor cell that was contacted with myeloid media.
- the precursor CAR cell, and/or population comprising a plurality of myeloid-derived CAR cells and/or a plurality of precursor CAR cells can be contacted with, and maintained in a myeloid media (i.e., differentiation step) for a short period of time, for example less than 24 hours, such as 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, or 23 hours.
- the cell(s) and/or population can be maintained in myeloid media for a longer period of time, for example, for 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, or more days.
- the differentiation step may be maintained for several hours (about 3 hours) to about 14 days or any hourly integer value in between.
- the differentiation step is maintained for 1 day, for 2 days, for 3 days, for 4 days, or for 5 days. In some embodiments, the differentiation step can be about 1 day. In some embodiments, the differentiation step is maintained for about 2 days. In some embodiments, the differentiation step is maintained for about 3 days. In some embodiments, the differentiation step is maintained for about 4 days. In some embodiments, the differentiation step is maintained for about 5 days. In some embodiments, the differentiation step is maintained for about 6 days. In some embodiments, the differentiation step is maintained for about 7 days. In some embodiments, the differentiation step is maintained for about 8 days. In some embodiments, the differentiation step is maintained for about 9 days. In some embodiments, the differentiation step is maintained for about 10 days.
- the differentiation step is maintained between 12-48 hours. In some embodiments, the differentiation step is maintained between 24-72 hours. In some embodiments, the differentiation step is maintained between 48-96 hours. In some embodiments, the differentiation step is maintained between 72-120 hours. In some embodiments, the differentiation step is maintained between 96-144 hours. In some embodiments, the differentiation step is maintained between 120-168 hours. In some embodiments, the differentiation step is maintained between 144-192 hours. In some embodiments, the differentiation step is maintained between 168-216 hours. In some embodiments, the differentiation step is maintained between 192-240 hours. In some embodiments, the differentiation step is maintained between 216-264 hours. In some embodiments, the differentiation step is maintained between 240-288 hours.
- the precursor CAR cell(s) and/or population comprising a plurality of precursor CAR cells and/or plurality of myeloid-derived CAR cells is maintained in said myeloid media until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population is CD1 lb+CD13+CD14+.
- the population comprising a plurality of myeloid-derived CAR cells is maintained in said myeloid media until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population is CD14+.
- the precursor CAR cell(s) and/or population comprising a plurality of precursor CAR cells and/or plurality of myeloid-derived CAR cells is maintained in said myeloid media until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population is CD14+CD13+.
- the precursor CAR cell(s) and/or population comprising a plurality of precursor CAR cells and/or plurality of myeloid-derived CAR cells is maintained in said myeloid media until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population is CD14+CD15+.
- the precursor CAR cell(s) and/or population comprising a plurality of precursor CAR cells and/or plurality of myeloid-derived CAR cells is maintained in said myeloid media until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population is CD14+CD13+CD15+.
- the differentiation step is maintained until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population comprising a plurality of myeloid-derived CAR cells is CD13+.
- the differentiation step is maintained until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population comprising a plurality of genetically modified precursor CAR cells and/or plurality of genetically modified myeloid-derived CAR cells is CD13+.
- the population comprising a plurality of myeloid-derived CAR cells is maintained in said myeloid media until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population is CD13+.
- the precursor CAR cell(s) and/or population comprising a plurality of precursor CAR cells and/or plurality of myeloid-derived CAR cells is maintained in said myeloid media until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population is CD 13+CD 15+
- the differentiation step is maintained until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population comprising a plurality of myeloid-derived CAR cells is CD15+.
- the differentiation step is maintained until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population comprising a plurality of genetically modified precursor CAR cells and/or plurality of genetically modified myeloid-derived CAR cells is CD15+.
- the precursor CAR cell(s) and/or population comprising a plurality of precursor CAR cells is maintained in said myeloid media until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population is CD15+.
- the precursor CAR cell(s) and/or population comprising a plurality of precursor CAR cells and/or plurality of myeloid- derived CAR cells is maintained in said myeloid media until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population is CD15+.
- the population comprising a plurality of myeloid-derived CAR cells is maintained in said myeloid media until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population is CD15+.
- the differentiation step is maintained until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the population comprising a plurality of genetically modified precursor CAR cells or the population comprising a plurality of genetically modified myeloid-derived CAR cells is CD13+CD15+.
- the ratio of differentiated cells to undifferentiated cells within the population increases during and after the differentiation step of the methods disclosed herein. For instance, during the differentiation step, the ratio of myeloid-derived CAR cells to precursor CAR cells with a population can change. In such instances, the ratio of myeloid-derived CAR cells to precursor CAR cells is expected to increase. In some embodiments, after the differentiation step, the number of myeloid-derived CAR cells compared to CD34+ CAR cells within the population increases. In some embodiments, after the differentiation step, the ratio of myeloid-derived CAR cells to CD34+ CAR cells within the population increases. Table 3. Myeloid Expansion and Differentiation from Hematopoietic Stem Cells
- Differentiation of genetically modified precursor CAR cells can increase the amount of CD1 lb+ cells within a plurality of genetically modified precursor CAR cells or a plurality of genetically modified myeloid-derived cells.
- Differentiation of genetically modified precursor CAR cells can increase the amount of CD1 lb+ cells within a plurality of genetically modified precursor CAR cells or a plurality of genetically modified myeloid-derived cells by about 10 fold, 20 fold, 30 fold, 40 fold, 50 fold, 60 fold, 70 fold, 80 fold, 90 fold, 100 fold, 200 fold, 300 fold, 400 fold, 500 fold, 600 fold, 700 fold, 800 fold, 900 fold, 1000 fold, 2000 fold, 3000 fold, 4000 fold, 5000 fold, 6000 fold, 7000 fold, 8000 fold, 9000 fold, 10,000 fold, 100,000 fold, 1,000,000 fold, 10,000,000 fold, or greater.
- Differentiation of genetically modified precursor CAR cells can increase the amount of CD 14+ cells within a plurality of genetically modified precursor CAR cells or a plurality of genetically modified myeloid-derived cells.
- Differentiation of genetically modified precursor CAR cells can increase the amount of CD14+ cells within a plurality of genetically modified precursor CAR cells or a plurality of genetically modified myeloid-derived cells by about 10 fold, 20 fold, 30 fold, 40 fold, 50 fold, 60 fold, 70 fold, 80 fold, 90 fold, 100 fold, 200 fold, 300 fold, 400 fold, 500 fold, 600 fold, 700 fold, 800 fold, 900 fold, 1000 fold, 2000 fold, 3000 fold, 4000 fold, 5000 fold, 6000 fold, 7000 fold, 8000 fold, 9000 fold, 10,000 fold, 100,000 fold, 1,000,000 fold, 10,000,000 fold, or greater.
- the amount of cell surface marker expression within a plurality of genetically modified precursor CAR cells or plurality of plurality of genetically modified myeloid-derived CAR cells can be used to determine how long to maintain the differentiating step of the method of manufacturing a genetically modified myeloid-derived CAR cells disclosed herein. In some instances, the differentiating step is maintained until a certain ratio of the population of genetically modified precursor cells or genetically modified myeloid-derived cells expresses a specific marker. In some instances, the differentiating step is maintained until a certain ratio of the population of genetically modified precursor cells or genetically modified myeloid-derived cells expresses at least a specific marker.
- the differentiating step is maintained until a certain ratio of the population of genetically modified precursor cells or genetically modified myeloid-derived cells expresses at least two specific markers. In some instances, the differentiating step is maintained until a certain ratio of the population of genetically modified precursor cells or genetically modified myeloid-derived cells expresses at least three specific markers. In some instances, the differentiating step is maintained until a certain ratio of the population of genetically modified precursor cells or genetically modified myeloid-derived cells expresses at least more than three different specific markers.
- the differentiating step is maintained until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the population comprising a plurality of genetically modified precursor CAR cells or the population comprising a plurality of genetically modified myeloid-derived CAR cells is CD1 lb+.
- the differentiating step is maintained until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the population comprising a plurality of genetically modified precursor CAR cells or the population comprising a plurality of genetically modified myeloid-derived CAR cells is CD14+.
- the differentiating step is maintained until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the population comprising a plurality of genetically modified precursor CAR cells or the population comprising a plurality of genetically modified myeloid-derived CAR cells is CD1 lb+CD14+.
- the expanding step is maintained until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the population comprising a plurality of genetically modified precursor CAR cells or the population comprising a plurality of genetically modified myeloid-derived CAR cells is CD13+.
- the expanding step is maintained until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the population comprising a plurality of genetically modified precursor CAR cells or the population comprising a plurality of genetically modified myeloid-derived CAR cells is CD15+.
- the expanding step is maintained until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the population comprising a plurality of genetically modified precursor CAR cells or the population comprising a plurality of genetically modified myeloid-derived CAR cells is CD13+CD15+.
- the ratio of differentiated cells to undifferentiated cells within the population increases during and after differentiation.
- Differentiation of genetically modified precursor CAR cells can increase the amount of CD1 lb+ cells within a plurality of genetically modified precursor CAR cells or a plurality of genetically modified myeloid-derived cells.
- the cells can be incubated in cell medium in a culture apparatus for a period of time or until the cells reach confluency or high cell density for optimal passage before passing the cells to another culture apparatus.
- the culturing apparatus can be any culture apparatus commonly used for culturing cells in vitro.
- the level of confluence is 70% or greater before passing the cells to another culture apparatus.
- the level of confluence is 90% or greater.
- a period of time can be any time suitable for the culture of cells in vitro.
- the culture medium may be replaced during the culture of the cells at any time. In some embodiments, the culture medium is replaced about every 2 to 3 days.
- the cells are then harvested from the culture apparatus whereupon the cells can be used immediately or stored for use at a later time.
- the differentiating step can be very short, for example less than 24 hours such as 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, or 23 hours.
- the differentiating step can be longer, for example 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, or more days.
- the cells may be differentiated for several hours (about 3 hours) to about 14 days or any hourly integer value in between.
- the genetically modified precursor CAR cell(s) is differentiated for at least 1, at least 2, at least 3, at least 4, or at least 5 days. In some embodiments, the genetically modified precursor CAR cell(s) is differentiated for 1 day, for 2 days, for 3 days, for 4 days, or for 5 days.
- Conditions appropriate for differentiation include an appropriate media
- DMEM/F12 fetal bovine or human serum
- serum e.g., fetal bovine or human serum
- L-glutamine insulin
- insulin M-CSF
- GM-CSF GM-CSF
- IL-10 IL-12
- IL-15 TGF-beta
- TNFa TNFa
- Other additives for the growth of cells include, but are not limited to, surfactant, plasmanate, and reducing agents such as N-acetyl-cysteine and 2-mercaptoethanol.
- culture media can include Stemline II, RPMI 1640, AEVI-V, DMEM, MEM, a-MEM, F-12, X-Vivo 15, X-Vivo 20, and Optimizer, with added amino acids, sodium pyruvate, and vitamins, either serum-free or supplemented with an appropriate amount of serum (or plasma) or a defined set of hormones, and/or an amount of cytokine(s) sufficient for the growth, expansion and/or differentiation of the cells.
- Antibiotics e.g., penicillin and streptomycin, are included only in experimental cultures, not in cultures of cells that are to be infused into a subject.
- the target cells are maintained under conditions necessary to support growth, for example, an appropriate temperature (e.g., 37°C) and atmosphere (e.g., air plus 5% CO2).
- the myeloid medium used to culture the cells may include an agent that can activate the cells.
- an agent that is known in the art to activate a macrophage, monocyte, dendritic cell, and/or a neutrophil is included in the medium.
- the step of differentiating the genetically modified precursor CAR cells can be performed until a number or ratio of precursor CAR cells have been differentiated into myeloid-derived CAR cells.
- cell surface markers can be examined; cell surface marker expression within a population can be examined over a series of hours or days to monitor the increase in desired surface marker.
- the population comprising a plurality of precursor CAR cells can be examined for decreases in expression of CD34+, a common stem cell surface marker.
- CD1 lb+ and/or CD14+ can be utilized to monitor myeloid lineage cells, including monocytes and macrophages.
- the precursor CAR cell is expanded and differentiated until the population comprising the plurality of genetically modified precursor myeloid-derived CAR cells comprises at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% CD1 lb+ or CD14+ or CD1 lb+CD14+ cells.
- the population of genetically modified precursor myeloid-derived CAR cells comprises at least 20%, at least 40%, at least 60%, or at least 80% CD 14+ after 3 days of differentiation.Jn some instances, at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of said plurality of genetically modified myeloid-derived CAR cells expressing said CAR are CD1 lb+ or CD14+ or Cl lb+CD14+.
- a myeloid media comprising a myeloid cytokine mixture useful for differentiating genetically modified precursor CAR cell(s).
- the myeloid media and myeloid cytokine mixture can be used in the methods disclosed herein to generate a myeloid-derived cell from a hematopoietic stem cell with increased efficiency and efficacy over known methods.
- a precursor cell is contacted with a precursor media to generate a cell population comprising a plurality of precursor cells, prior to contacting said precursor cell(s) with myeloid media.
- the precursor cell can be an HSC Following contact with a precursor media for a suitable time period, it can be expected that a precursor cell is expanded (i.e., the number of precursor cells increases) and differentiated (e.g., into a myeloid-derived cell) in said precursor media to generate a population comprising a plurality of myeloid-derived cells.
- the population comprises a plurality of myeloid-derived cells and a plurality of precursor cells.
- the precursor cells and myeloid-derived cells can be present at any ratio within the population after being maintained in the precursor media.
- the myeloid media and myeloid cytokine mixture can be used in the methods disclosed herein to generate a myeloid-derived CAR cell from a hematopoietic stem cell with increased efficiency and efficacy over known methods.
- a precursor CAR cell is contacted with a precursor media to generate a cell population comprising a plurality of precursor CAR cells, prior to contacting said precursor CAR cell(s) with myeloid media.
- the disclosed precursor media are particularly effective at both expanding a precursor cell to generate a cell population, as well as promoting the differentiation of a precursor cell, specifically into a myeloid cell.
- a precursor CAR cell is expanded (i.e., the number of precursor CAR cells increases) and differentiated (e.g., into a myeloid-derived CAR cell) in said precursor media to generate a population comprising a plurality of myeloid-derived CAR cells.
- the population comprises a plurality of myeloid-derived CAR cells and a plurality of precursor CAR cells.
- the precursor CAR cells and myeloid-derived CAR cells can be present at any ratio within the population after being maintained in the precursor media.
- the myeloid media disclosed herein is particularly effective and efficient, when compared to known differentiation mediums, at increasing the ratio of myeloid-derived CAR cells to precursor CAR cells within a cell population.
- a cell population is generated by maintaining a precursor CAR cell in a precursor media, wherein said cell population comprises a plurality of precursor CAR cells and a plurality of myeloid-derived CAR cells, said cell population is subsequently contacted with a myeloid media disclosed herein to increase the number of myeloid-derived CAR cells within the cell population.
- the total number of cells within the population is relatively unchanged. In some embodiments, when the number of myeloid-derived CAR cells increases within a population, the total number of cells within the population increases. In some embodiments, when the number of myeloid-derived CAR cells increases within a population, the total number of cells within the population decreases.
- a cell population when a cell population is generated by maintaining a precursor CAR cell in a precursor media, wherein said cell population comprises a plurality of precursor CAR cells and a plurality of myeloid-derived CAR cells, said cell population is subsequently contacted with a myeloid media disclosed herein to decrease the number of precursor CAR cells within the cell population.
- the number of precursor CAR cells decreases within a population, the total number of cells within the population is relatively unchanged.
- the number of precursor CAR cells decreases within a population, the total number of cells within the population increases.
- the total number of cells within the population decreases.
- a cell population when a cell population is generated by maintaining a precursor CAR cell in a precursor media, wherein said cell population comprises a plurality of precursor CAR cells and a plurality of myeloid-derived CAR cells, said cell population is subsequently contacted with a myeloid media disclosed herein to increase the number of myeloid-derived CAR cells compared to precursor CAR cells within the population.
- the total number of cells within the population when the number of myeloid-derived CAR cells compared to precursor CAR cells within the population, the total number of cells within the population is relatively unchanged.
- the total number of myeloid-derived CAR cells compared to precursor CAR cells within the population the total number of cells within the population increases.
- the number of myeloid-derived CAR cells compared to precursor CAR cells within the population the total number of cells within the population decreases.
- a cell population when a cell population is generated by maintaining a precursor CAR cell in a precursor media, wherein said cell population comprises a plurality of precursor CAR cells and a plurality of myeloid-derived CAR cells, said cell population is subsequently contacted with a myeloid media disclosed herein to increase the ratio of myeloid-derived CAR cells to precursor CAR cells within the population.
- a myeloid media disclosed herein to increase the ratio of myeloid-derived CAR cells to precursor CAR cells within the population.
- the ratio of myeloid-derived CAR cells to precursor CAR cells within the population when the ratio of myeloid-derived CAR cells to precursor CAR cells within the population is increased, the total number of cells within the population is relatively unchanged.
- the ratio of myeloid-derived CAR cells to precursor CAR cells within the population when the ratio of myeloid-derived CAR cells to precursor CAR cells within the population is increased, the total number of cells within the population decreases.
- the genetically modified precursor CAR cells are genetically modified stem cells expressing a CAR.
- the stem cells expressing a CAR are hematopoietic stem cells (HSCs).
- HSCs hematopoietic stem cells
- the HSCs can be CD34+ cells. Differentiating the HSCs modified to express a CAR promotes their differentiation in a genetically modified myeloid-derived CAR cell(s).
- the myeloid media disclosed herein is particularly effective at promoting the differentiation of HSCs to myeloid-derived cells.
- the myeloid media is particularly effective at differentiation a CD34+ cell into a CD14+ cell.
- the myeloid media is particularly effective at differentiation a CD34+ cell into a CD1 lb+ cell.
- the myeloid media is particularly effective at differentiation a CD34+ cell into a CD14+CD1 lb+ cell.
- the cytokine mixture within the myeloid media i.e., the myeloid cytokine mixture
- myeloid cytokine mixtures that are particularly effective at promoting differentiation of stem cells into myeloid-derived cells.
- myeloid-derived cells are CD14+ cells.
- the myeloid media comprising the myeloid cytokine mixtures disclosed herein are particularly effective at promoting differentiation of HSCs into CD 14+ cells.
- the myeloid media comprising the myeloid cytokine mixtures disclosed herein are particularly effective at promoting differentiation of HSCs into CD1 lb+ cells.
- the myeloid media comprising the myeloid cytokine mixtures disclosed herein are particularly effective at promoting differentiation of HSCs into any one of the following cells: CD13+ cell, a CD15+ cell, a CDl lb+CD13+ cell, a CDl lb+CD13+CD14+ cell, a CDl lb+CD13+CD15+ cell, a CD1 lb+CD13+CD14+CD15+ cell, a CDl lb+CD14+ cell, a CDl lb+CD15+ cell, a
- CD1 lb+CD14+CD15+ cell a CD14+CD13+ cell, a CD14+CD15+ cell, a CD14+CD13+CD15+ cell, and/or a CD13+CD15+ cell.
- the myeloid cytokine mixture can comprise Macrophage colony-stimulating factor (MCSF), Interleukin-3 (IL-3), or Interleukin-6 (IL-6), or a combination thereof.
- the myeloid cytokine mixture comprises Macrophage colony-stimulating factor (MCSF), Interleukin-3 (IL-3), and Interleukin-6 (IL-6).
- the concentration of the MCSF in the myeloid media can be at least 50 ng/mL, at least 100 ng/mL, at least 150 ng/mL, or at least 200 ng/mL.
- the myeloid media comprises macrophage colony-stimulating factor (MCSF) at a concentration of 150 ng/mL.
- the concentration of the IL-3 in the myeloid media can be at least 50 ng/mL, at least 100 ng/mL, at least 150 ng/mL, or at least 200 ng/mL. In a preferred embodiment, the myeloid media comprises IL-3 at a concentration of 150 ng/mL.
- the myeloid cytokine mixture comprises Interleukin-6 (IL-6)
- the concentration of the IL-6 in the myeloid media can be at least 50 ng/mL, at least 100 ng/mL, at least 150 ng/mL, or at least 200 ng/mL.
- the myeloid media comprises IL-6 at a concentration of 150 ng/mL.
- the myeloid cytokine mixture can comprise Macrophage colony-stimulating factor (MCSF), Interleukin-3 (IL-3), and Interleukin-6 (IL-6).
- MCSF Macrophage colony-stimulating factor
- IL-3 Interleukin-3
- IL-6 Interleukin-6
- the myeloid media can comprise MCSF at a concentration of 150 ng/mL, IL-3 at a concentration of 50 ng/mL and IL-6 at a concentration of 100 ng/mL.
- the myeloid media comprises MCSF at a concentration of 150 ng/mL, IL-3 at a concentration of 50 ng/mL, and IL-6 at a concentration of 100 ng/mL.
- the myeloid media comprises MCSF at a concentration of 150 ng/mL, IL-3 at a concentration of 50 ng/mL, and IL-6 at a concentration of 100 ng/mL, the myeloid media further comprises a basal growth media comprising VIVOTM- 15 medium.
- the myeloid media comprises macrophage colony-stimulating factor (MCSF).
- the macrophage colony-stimulating factor (MCSF) can be present in the myeloid media at a concentration of at least 20 ng/mL, at least 30 ng/mL, at least 40 ng/mL, at least 50 ng/mL, at least 60 ng/mL, of at least 70 ng/mL, at least 80 ng/mL, at least 90 ng/mL, at least 100 ng/mL, at least 110 ng/mL, at least 120 ng/mL, at least 130 ng/mL, at least 140 ng/mL, at least 150 ng/mL, at least 160 ng/mL, at least 170 ng/mL, at least 180 ng/mL, at least 190 ng/mL, at least 200 ng/mL, at least 210 ng/mL, of at least 220 ng/mL, at least 230 ng
- the macrophage colony-stimulating factor can be present in the myeloid media at a concentration of 20-40 ng/mL, 30-50 ng/mL, 40-60 ng/mL, 50-70 ng/mL, 60-80 ng/mL, 70-90 ng/mL, 80-100 ng/mL, 90-110 ng/mL, 100-120 ng/mL, 110-130 ng/mL, 120-140 ng/mL, 130-150 ng/mL, 140-160 ng/mL, 150-170 ng/mL, 160-180 ng/mL, 170-190 ng/mL, 180-200 ng/mL, 190-210 ng/mL, 200-220 ng/mL, 210-230 ng/mL, 220- 240 ng/mL, 230-250 ng/mL, or greater than 250 ng/mL.
- MCSF macrophage colony-stimulating factor
- the myeloid media comprises MCSF
- said MCSF is present at a concentration of between about 100 ng/mL and 200 ng/mL. In some embodiments, when the myeloid media comprises MCSF, said MCSF is present at a concentration of between 100-105 ng/mL, 105-110 ng/mL, 110-115 ng/mL, 115-120 ng/mL, 120-125 ng/mL, 125-130 ng/mL, BODS ng/mL, 135-140 ng/mL, 140-145 ng/mL, 145-150 ng/mL, 150-155 ng/mL, 155-160 ng/mL, 160-165 ng/mL, 165-170 ng/mL, 170-175 ng/mL, 175-180 ng/mL, 180-185 ng/mL, 185-190 ng/mL, 190-195 ng/mL, or 195
- the myeloid media comprises more than one cytokine (i.e., a myeloid cytokine mixture).
- a myeloid cytokine mixture comprises MCSF at a concentration of between about 125 ng/mL and 175 ng/mL in said myeloid media
- the myeloid cytokine mixture comprises MCSF, IL-3, and IL-6.
- said myeloid media can comprise any of the following cytokines: MCSF, IL-3, and IL-6.
- the myeloid media can comprise at least one of the following additional cytokines: IL-3 and/or IL-6.
- IL-3 can be present at a concentration between about 25 ng/mL and 75 ng/mL
- IL-6 can be present at a concentration between about 75 ng/mL and 125 ng/mL.
- the myeloid media comprises MCSF at a concentration of about 150 ng/mL, IL-3 at a concentration of about 50 ng/mL, and IL-6 at a concentration of about 100 ng/mL. In a preferred embodiment, the myeloid media comprises MCSF at a concentration of 150 ng/mL, IL-3 at a concentration of 50 ng/mL, and IL-6 at a concentration of 100 ng/mL.
- the myeloid media when the myeloid media comprises macrophage colony-stimulating factor (MCSF) at a concentration of between about 100 ng/mL and 200 ng/mL, said myeloid media further comprises: a. IL-3 at a concentration of 10-30 ng/mL, 20-40 ng/mL, 30-50 ng/mL, 40-60 ng/mL, 50-70 ng/mL, 60-80 ng/mL, 70-90 ng/mL, 80-100 ng/mL, 90-110 ng/mL, 100-120 ng/mL, 110-130 ng/mL, 120-140 ng/mL, 130-150 ng/mL, 140-160 ng/mL, 150-170 ng/mL, 160-180 ng/mL, 170-190 ng/mL, 180-200 ng/mL, 190-210 ng/mL, 200-220 ng/mL, or greater than 220 ng/mL;
- IL-6 at a concentration of 10-30 ng/mL, 20-40 ng/mL, 30-50 ng/mL, 40-60 ng/mL, 50-70 ng/mL, 60-80 ng/mL, 70-90 ng/mL, 80-100 ng/mL, 90-110 ng/mL, 100-120 ng/mL, 110-130 ng/mL, 120-140 ng/mL, 130-150 ng/mL, 140-160 ng/mL, 150-170 ng/mL, 160-180 ng/mL, 170-190 ng/mL, 180-200 ng/mL, 190-210 ng/mL, 200-220 ng/mL, or greater than 220 ng/mL.
- myeloid media when the myeloid media comprises macrophage colony-stimulating factor (MCSF) at a concentration of about 140 ng/mL. In some embodiments, when the myeloid media comprises macrophage colony-stimulating factor (MCSF) at a concentration of about 140 ng/mL, said myeloid media further comprises: a.
- MCSF macrophage colony-stimulating factor
- IL-3 at a concentration of 10-30 ng/mL, 20-40 ng/mL, 30-50 ng/mL, 40-60 ng/mL, 50-70 ng/mL, 60-80 ng/mL, 70-90 ng/mL, 80-100 ng/mL, 90- 110 ng/mL, 100-120 ng/mL, 110-130 ng/mL, 120-140 ng/mL, 130-150 ng/mL, 140-160 ng/mL, 150-170 ng/mL, 160-180 ng/mL, 170-190 ng/mL, 180-200 ng/mL, 190-210 ng/mL, 200-220 ng/mL, or greater than 220 ng/mL; and/or b.
- IL-6 at a concentration of 10-30 ng/mL, 20-40 ng/mL, 30-50 ng/mL, 40-60 ng/mL, 50-70 ng/mL, 60-80 ng/mL, 70-90 ng/mL, 80-100 ng/mL, 90-110 ng/mL, 100-120 ng/mL, 110-130 ng/mL, 120-140 ng/mL, 130-150 ng/mL, 140-160 ng/mL, 150- 170 ng/mL, 160-180 ng/mL, 170-190 ng/mL, 180-200 ng/mL, 190-210 ng/mL, 200-220 ng/mL, or greater than 220 ng/mL.
- myeloid media when the myeloid media comprises macrophage colony-stimulating factor (MCSF) at a concentration of about 145 ng/mL. In some embodiments, when the myeloid media comprises macrophage colony-stimulating factor (MCSF) at a concentration of about 145 ng/mL, said precursor media further comprises: a.
- MCSF macrophage colony-stimulating factor
- IL-3 at a concentration of 10-30 ng/mL, 20-40 ng/mL, 30-50 ng/mL, 40-60 ng/mL, 50-70 ng/mL, 60-80 ng/mL, 70-90 ng/mL, 80-100 ng/mL, 90- 110 ng/mL, 100-120 ng/mL, 110-130 ng/mL, 120-140 ng/mL, 130-150 ng/mL, 140-160 ng/mL, 150-170 ng/mL, 160-180 ng/mL, 170-190 ng/mL, 180-200 ng/mL, 190-210 ng/mL, 200-220 ng/mL, or greater than 220 ng/mL; and/or b.
- IL-6 at a concentration of 10-30 ng/mL, 20-40 ng/mL, 30-50 ng/mL, 40-60 ng/mL, 50-70 ng/mL, 60-80 ng/mL, 70-90 ng/mL, 80-100 ng/mL, 90-110 ng/mL, 100-120 ng/mL, 110-130 ng/mL, 120-140 ng/mL, 130-150 ng/mL, 140-160 ng/mL, 150- 170 ng/mL, 160-180 ng/mL, 170-190 ng/mL, 180-200 ng/mL, 190-210 ng/mL, 200-220 ng/mL, or greater than 220 ng/mL.
- myeloid media when the myeloid media comprises macrophage colony-stimulating factor (MCSF) at a concentration of about 150 ng/mL. In some embodiments, when the myeloid media comprises macrophage colony-stimulating factor (MCSF) at a concentration of about 150 ng/mL, said precursor media further comprises: a.
- IL-3 at a concentration of 10-30 ng/mL, 20-40 ng/mL, 30-50 ng/mL, 40-60 ng/mL, 50-70 ng/mL, 60-80 ng/mL, 70-90 ng/mL, 80-100 ng/mL, 90- 110 ng/mL, 100-120 ng/mL, 110-130 ng/mL, 120-140 ng/mL, 130-150 ng/mL, 140-160 ng/mL, 150-170 ng/mL, 160-180 ng/mL, 170-190 ng/mL, 180-200 ng/mL, 190-210 ng/mL, 200-220 ng/mL, or greater than 220 ng/mL; and/or b.
- IL-6 at a concentration of 10-30 ng/mL, 20-40 ng/mL, 30-50 ng/mL, 40-60 ng/mL, 50-70 ng/mL, 60-80 ng/mL, 70-90 ng/mL, 80-100 ng/mL, 90-110 ng/mL, 100-120 ng/mL, 110-130 ng/mL, 120-140 ng/mL, 130-150 ng/mL, 140-160 ng/mL, 150- 170 ng/mL, 160-180 ng/mL, 170-190 ng/mL, 180-200 ng/mL, 190-210 ng/mL, 200-220 ng/mL, or greater than 220 ng/mL.
- myeloid media when the myeloid media comprises macrophage colony-stimulating factor (MCSF) at a concentration of about 155 ng/mL. In some embodiments, when the myeloid media comprises macrophage colony-stimulating factor (MCSF) at a concentration of about 155 ng/mL, said precursor media further comprises: a.
- MCSF macrophage colony-stimulating factor
- IL-3 at a concentration of 10-30 ng/mL, 20-40 ng/mL, 30-50 ng/mL, 40-60 ng/mL, 50-70 ng/mL, 60-80 ng/mL, 70-90 ng/mL, 80-100 ng/mL, 90- 110 ng/mL, 100-120 ng/mL, 110-130 ng/mL, 120-140 ng/mL, 130-150 ng/mL, 140-160 ng/mL, 150-170 ng/mL, 160-180 ng/mL, 170-190 ng/mL, 180-200 ng/mL, 190-210 ng/mL, 200-220 ng/mL, or greater than 220 ng/mL; and/or b.
- IL-6 at a concentration of 10-30 ng/mL, 20-40 ng/mL, 30-50 ng/mL, 40-60 ng/mL, 50-70 ng/mL, 60-80 ng/mL, 70-90 ng/mL, 80-100 ng/mL, 90-110 ng/mL, 100-120 ng/mL, 110-130 ng/mL, 120-140 ng/mL, 130-150 ng/mL, 140-160 ng/mL, 150- 170 ng/mL, 160-180 ng/mL, 170-190 ng/mL, 180-200 ng/mL, 190-210 ng/mL, 200-220 ng/mL, or greater than 220 ng/mL.
- myeloid media when the myeloid media comprises macrophage colony-stimulating factor (MCSF) at a concentration of about 160 ng/mL. In some embodiments, when the myeloid media comprises macrophage colony-stimulating factor (MCSF) at a concentration of about 160 ng/mL, said precursor media further comprises: a.
- MCSF macrophage colony-stimulating factor
- IL-3 at a concentration of 10-30 ng/mL, 20-40 ng/mL, 30-50 ng/mL, 40-60 ng/mL, 50-70 ng/mL, 60-80 ng/mL, 70-90 ng/mL, 80-100 ng/mL, 90- 110 ng/mL, 100-120 ng/mL, 110-130 ng/mL, 120-140 ng/mL, 130-150 ng/mL, 140-160 ng/mL, 150-170 ng/mL, 160-180 ng/mL, 170-190 ng/mL, 180-200 ng/mL, 190-210 ng/mL, 200-220 ng/mL, or greater than 220 ng/mL; and/or b.
- IL-6 at a concentration of 10-30 ng/mL, 20-40 ng/mL, 30-50 ng/mL, 40-60 ng/mL, 50-70 ng/mL, 60-80 ng/mL, 70-90 ng/mL, 80-100 ng/mL, 90-110 ng/mL, 100-120 ng/mL, 110-130 ng/mL, 120-140 ng/mL, 130-150 ng/mL, 140-160 ng/mL, 150- 170 ng/mL, 160-180 ng/mL, 170-190 ng/mL, 180-200 ng/mL, 190-210 ng/mL, 200-220 ng/mL, or greater than 220 ng/mL.
- the myeloid media comprises MCSF at a concentration of 150 ng/mL.
- said myeloid media further comprises: a. IL-3 at a concentration between about 25 ng/mL and 75 ng/mL; and/or b. IL-6 at a concentration between about 75 ng/mL and 125 ng/mL.
- the myeloid media comprises MCSF at a concentration of about 150ng/mL
- said myeloid media further comprises: a. IL-3 at a concentration between about 20 ng/mL, about 25 ng/mL, about 30 ng/mL, about 35 ng/mL, about 40 ng/mL, about 45 ng/mL, about 50 ng/mL, about 555 ng/mL, about 60 ng/mL, about 65 ng/mL, about 70 ng/mL, or about 75 ng/mL; and/or b.
- IL-6 at a concentration between about 70 ng/mL, about 75 ng/mL, about 80 ng/mL, about 85 ng/mL, about 90 ng/mL, about 95 ng/mL, about 100 ng/mL, about 105 ng/mL, about 110 ng/mL, about 115 ng/mL, about 120 ng/mL, or about 125 ng/mL.
- the myeloid media can comprise stem cells factor (SCF), Flt3 ligand, thrombopoietin (TPO), Granulocyte colony-stimulating factor (G-CSF), and Granulocyte-macrophage colony-stimulating factor (GM-CSF).
- SCF stem cells factor
- Flt3 ligand thrombopoietin
- TPO Granulocyte colony-stimulating factor
- GM-CSF Granulocyte-macrophage colony-stimulating factor
- a useful concentration is defined as any concentration of an individual cytokine within the myeloid media that promotes the differentiation of precursor CAR cells or myeloid-derived CAR cells as described herein.
- Stem cells factor can be within the myeloid mixture at a concentration of at most 50 ng/mL, at most 40 ng/mL, at most 30 ng/mL, at most 20 ng/mL, at most 10 ng/mL, at most 5 ng/mL, at most 4 ng/mL, at most 3 ng/mL, at most 2 ng/mL, at most 1 ng/mL, at most 0.5 ng/mL, at most 0.1 ng/mL, at most 0.01 ng/mL, at most 0.0001 ng/mL, or less than 0.0001 ng/L.
- SCF Stem cells factor
- the myeloid media comprises stem cells factor (SCF) at a concentration of below 50 ng/mL. In some instances, the myeloid media comprises stem cells factor (SCF) at a concentration of below 40 ng/mL. In some instances, the myeloid media comprises stem cells factor (SCF) at a concentration of below 30 ng/mL. In some instances, the myeloid media comprises stem cells factor (SCF) at a concentration of below 20 ng/mL. In some instances, the myeloid media comprises stem cells factor (SCF) at a concentration of below 10 ng/mL. In some instances, the myeloid media comprises stem cells factor (SCF) at a concentration of below 5 ng/mL. In a preferred embodiment, the myeloid mixture does not comprise stem cells factor (SCF).
- SCF stem cells factor
- Flt3 ligand can be within the myeloid mixture at a concentration of at most 50 ng/mL, at most 40 ng/mL, at most 30 ng/mL, at most 20 ng/mL, at most 10 ng/mL, at most 5 ng/mL, at most 4 ng/mL, at most 3 ng/mL, at most 2 ng/mL, at most 1 ng/mL, at most 0.5 ng/mL, at most 0.1 ng/mL, at most 0.01 ng/mL, at most 0.0001 ng/mL, or less than 0.0001 ng/L.
- the myeloid media comprises Flt3 ligand at a concentration of below 50 ng/mL.
- the myeloid media comprises Flt3 ligand at a concentration of below 40 ng/mL. In some instances, the myeloid media comprises Flt3 ligand at a concentration of below 30 ng/mL. In some instances, the myeloid media comprises Flt3 ligand at a concentration of below 20 ng/mL. In some instances, the myeloid media comprises Flt3 ligand at a concentration of below 10 ng/mL. In some instances, the myeloid media comprises Flt3 ligand at a concentration of below 5 ng/mL. In a preferred embodiment, the myeloid mixture does not comprise Flt3 ligand.
- Thrombopoietin can be within the myeloid mixture at a concentration of at most 50 ng/mL, at most 40 ng/mL, at most 30 ng/mL, at most 20 ng/mL, at most 10 ng/mL, at most 5 ng/mL, at most 4 ng/mL, at most 3 ng/mL, at most 2 ng/mL, at most 1 ng/mL, at most 0.5 ng/mL, at most 0.1 ng/mL, at most 0.01 ng/mL, at most 0.0001 ng/mL, or less than 0.0001 ng/L.
- the myeloid media comprises TPO at a concentration of below 50 ng/mL. In some instances, the myeloid media comprises TPO at a concentration of below 40 ng/mL. In some instances, the myeloid media comprises TPO at a concentration of below 30 ng/mL. In some instances, the myeloid media comprises TPO at a concentration of below 20 ng/mL. In some instances, the myeloid media comprises TPO at a concentration of below 10 ng/mL. In some instances, the myeloid media comprises TPO at a concentration of below 5 ng/mL. In a preferred embodiment, the myeloid mixture does not comprise TPO.
- Granulocyte colony-stimulating factor can be within the myeloid mixture at a concentration of at most 50 ng/mL, at most 40 ng/mL, at most 30 ng/mL, at most 20 ng/mL, at most 10 ng/mL, at most 5 ng/mL, at most 4 ng/mL, at most 3 ng/mL, at most 2 ng/mL, at most 1 ng/mL, at most 0.5 ng/mL, at most 0.1 ng/mL, at most 0.01 ng/mL, at most 0.0001 ng/mL, or less than 0.0001 ng/L.
- G-CSF Granulocyte colony-stimulating factor
- the myeloid media comprises G-CSF at a concentration of below 50 ng/mL. In some instances, the myeloid media comprises G-CSF at a concentration of below 40 ng/mL. In some instances, the myeloid media comprises G-CSF at a concentration of below 30 ng/mL. In some instances, the myeloid media comprises G-CSF at a concentration of below 20 ng/mL. In some instances, the myeloid media comprises G-CSF at a concentration of below 10 ng/mL. In some instances, the myeloid media comprises G-CSF at a concentration of below 5 ng/mL. In a preferred embodiment, the myeloid mixture does not comprise any G-CSF.
- Granulocyte-macrophage colony-stimulating factor can be within the myeloid mixture at a concentration of at most 50 ng/mL, at most 40 ng/mL, at most 30 ng/mL, at most 20 ng/mL, at most 10 ng/mL, at most 5 ng/mL, at most 4 ng/mL, at most 3 ng/mL, at most 2 ng/mL, at most 1 ng/mL, at most 0.5 ng/mL, at most 0.1 ng/mL, at most 0.01 ng/mL, at most 0.0001 ng/mL, or less than 0.0001 ng/L.
- GM-CSF Granulocyte-macrophage colony-stimulating factor
- the myeloid media comprises GM-CSF at a concentration of below 50 ng/mL. In some instances, the myeloid media comprises GM-CSF at a concentration of below 40 ng/mL. In some instances, the myeloid media comprises GM-CSF at a concentration of below 30 ng/mL. In some instances, the myeloid media comprises GM-CSF at a concentration of below 20 ng/mL. In some instances, the myeloid media comprises GM-CSF at a concentration of below 10 ng/mL. In some instances, the myeloid media comprises GM-CSF at a concentration of below 5 ng/mL. In a preferred embodiment, the myeloid mixture does not comprise any GM-CSF.
- the myeloid media comprises MCSF at a concentration of between 125 ng/mL and 175 ng/mL, and said myeloid media further comprises: a. IL-3 at a concentration between about 20 ng/mL, about 25 ng/mL, about 30 ng/mL, about 35 ng/mL, about 40 ng/mL, about 45 ng/mL, about 50 ng/mL, about 555 ng/mL, about 60 ng/mL, about 65 ng/mL, about 70 ng/mL, or about 75 ng/mL; b.
- IL-3 at a concentration between about 20 ng/mL, about 25 ng/mL, about 30 ng/mL, about 35 ng/mL, about 40 ng/mL, about 45 ng/mL, about 50 ng/mL, about 555 ng/mL, about 60 ng/mL, about 65 ng/mL, about 70 ng/mL, or about 75
- IL-6 at a concentration between about 70 ng/mL, about 75 ng/mL, about 80 ng/mL, about 85 ng/mL, about 90 ng/mL, about 95 ng/mL, about 100 ng/mL, about 105 ng/mL, about 110 ng/mL, about 115 ng/mL, about 120 ng/mL, or about 125 ng/mL; c.
- stem cells factor at a concentration of at most 50 ng/mL, at most 40 ng/mL, at most 30 ng/mL, at most 20 ng/mL, at most 10 ng/mL, at most 5 ng/mL, at most 4 ng/mL, at most 3 ng/mL, at most 2 ng/mL, at most 1 ng/mL, at most 0.5 ng/mL, at most 0.1 ng/mL, at most 0.01 ng/mL, at most 0.0001 ng/mL, or less than 0.0001 ng/L; d.
- SCF stem cells factor
- Flt3 ligand at a concentration of at most 50 ng/mL, at most 40 ng/mL, at most 30 ng/mL, at most 20 ng/mL, at most 10 ng/mL, at most 5 ng/mL, at most 4 ng/mL, at most 3 ng/mL, at most 2 ng/mL, at most 1 ng/mL, at most 0.5 ng/mL, at most 0.1 ng/mL, at most 0.01 ng/mL, at most 0.0001 ng/mL, or less than 0.0001 ng/L; e.
- TPO thrombopoietin
- TPO thrombopoietin
- G-CSF granulocyte colony-stimulating factor
- GM-CSF granulocyte-macrophage colony-stimulating factor
- the myeloid media comprises MCSF at a concentration of about 150ng/mL
- said myeloid media further comprises: a. IL-3 at a concentration between about 20 ng/mL, about 25 ng/mL, about 30 ng/mL, about 35 ng/mL, about 40 ng/mL, about 45 ng/mL, about 50 ng/mL, about 555 ng/mL, about 60 ng/mL, about 65 ng/mL, about 70 ng/mL, or about 75 ng/mL; b.
- IL-6 at a concentration between about 70 ng/mL, about 75 ng/mL, about 80 ng/mL, about 85 ng/mL, about 90 ng/mL, about 95 ng/mL, about 100 ng/mL, about 105 ng/mL, about 110 ng/mL, about 115 ng/mL, about 120 ng/mL, or about 125 ng/mL; c.
- stem cells factor at a concentration of at most 50 ng/mL, at most 40 ng/mL, at most 30 ng/mL, at most 20 ng/mL, at most 10 ng/mL, at most 5 ng/mL, at most 4 ng/mL, at most 3 ng/mL, at most 2 ng/mL, at most 1 ng/mL, at most 0.5 ng/mL, at most 0.1 ng/mL, at most 0.01 ng/mL, at most 0.0001 ng/mL, or less than 0.0001 ng/L; d.
- SCF stem cells factor
- Flt3 ligand at a concentration of at most 50 ng/mL, at most 40 ng/mL, at most 30 ng/mL, at most 20 ng/mL, at most 10 ng/mL, at most 5 ng/mL, at most 4 ng/mL, at most 3 ng/mL, at most 2 ng/mL, at most 1 ng/mL, at most 0.5 ng/mL, at most 0.1 ng/mL, at most 0.01 ng/mL, at most 0.0001 ng/mL, or less than 0.0001 ng/L; e.
- TPO thrombopoietin
- TPO thrombopoietin
- G-CSF granulocyte colony-stimulating factor
- the term “growth” when as used herein in relation to cell refers to an increase in the total mass or a cell and/or an increase in the total number of cells within a population of cells. Importantly, cell growth is not to be confused with cell division, which is a distinct process generally associated with cell proliferation and/or cell division.
- the basal growth medium can comprise glucose, salts, amino acids, and vitamins.
- the basal growth medium can contain a Roswell Park Memorial Institute (RPMI) 1640 medium, X-VIVOTM-15 medium, CTSTM OPTMIZERTM Serum Free Medium, CTSTM OPTMIZERTM Pro Serum Free Medium, and/or IMMUNOCULTTM-XF T Cell Expansion Medium.
- the basal growth medium comprises the RPMI 1640 medium with ATCC modification (Gibco A1049101).
- the basal growth media comprises VIVOTM- 15 medium.
- the VIVOTM- 15 medium can be combined or mixed with any other basal growth media at any ratio that is particularly effective. Any volume of basal growth media is envisaged within the method disclosed herein.
- the myeloid media can further comprise GlutaMAXTM Supplement.
- the myeloid media can comprise a myeloid cytokine mixture, a basal growth medium and GlutaMAXTM Supplement.
- the GlutaMAXTM Supplement can be present in the myeloid media at a concentration of between 0.5 mM, 1 mM, 1.5 mM, 2 mM, 2.5 mM or 3 mM of the myeloid media.
- a genetically modified myeloid-derived cell comprising a CAR
- the method comprises the steps of: (a) contacting a precursor cell with a nucleic acid molecule comprising a polynucleotide sequence encoding a CAR, whereby said nucleic acid is introduced into said precursor cell, and (b) contacting said precursor cell with a precursor media comprising a precursor cytokine mixture, whereby the number of precursor cell(s) increases to generate a population of cells comprising a plurality of precursor CAR cells, wherein said precursor cell is a stem cell.
- the method comprises maintaining the precursor cell in the precursor medium of until a desired cell population is achieved.
- a cell population can be analyzed and classified according to certain cell surface markers. For instance, a skilled artisan would be capable of determining the percentage of cells within a given cell population expressing a cell surface marker.
- the number of cell surface markers that can be is no limiting. Any number of cell surface markers can be analyzed during the methods disclosed herein to determine if a desired cell population has been achieved.
- Exemplary, non-limiting, cell surface markers that can be analyzed for the disclosed cell populations include: CD34, CD13, CD15, CD1 lb, and CD14. Any cell surface marker that can distinguish a stem cell and/or a myeloid- lineage cell from other known cell types are envisaged as being used.
- the methods disclosed herein are particularly effective at generating myeloid-derived CAR cells from hematopoietic stem cells (HSCs) due, at least in part, to the particular precursor and myeloid mediums disclosed herein.
- the precursor mediums disclosed herein are particularly effective at expanding an HSC, or a population of HSCs.
- the HSC is present within a population of HSCs.
- a population of HSCs can be expanded (i.e., increased in number of cells) when at least one HSC within the population expands.
- a genetically modified myeloid-derived CAR cell from a stem cell
- the stem cell comprising a CAR i.e., stem cell CAR cell
- a precursor media comprising a precursor cytokine mixture
- the number of stem cell CAR cell(s) increases to generate a population of stem cells comprising a plurality of stem cell CAR cells
- said stem cell CAR cell differentiates into a myeloid- lineage cell to generate a myeloid-derived CAR cell or a population comprising a plurality of myeloid-derived CAR cells
- said cell population is a mixed population comprising said stem cell CAR cells and said myeloid-derived CAR cells
- said precursor media comprises a precursor cytokine mixture comprising G-CSF, GM-CSF, SCF, and/or TPO
- the stem cell CAR cell is
- the precursor cytokine media can comprise: a. G-CSF at a concentration between about 100 ng/mL and 200 ng/mL; b. GM-CSF at a concentration between about 75 ng/mL and 125 ng/mL; c. SCF at a concentration between about 75 ng/mL and 125 ng/mL; and/or d. TPO at a concentration between about 75 ng/mL and 125 ng/mL.
- the stem cell can be a hematopoietic stem cell.
- the method can further comprise the step of contacting said stem cell CAR cell, myeloid-derived CAR cell, and/or said population comprising stem cell CAR cells and said myeloid-derived CAR cells with a myeloid media comprising a myeloid cytokine mixture, whereby said ratio of myeloid-derived CAR cells relative to stem cell CAR cells within the population is increased.
- said cell population comprising said stem cell CAR cells and said myeloid-derived CAR cells is maintained in said myeloid media until no more than 1%, 0.9%, 0,8%, 0,7%, 0.6%, 0.5%, 0.4%, 0.3%, 0.2%, 0.1%, or less than 0.1% of said population of cells is CD34+.
- the precursor cytokine media can comprise: a. G-CSF at a concentration between about 100 ng/mL and 200 ng/mL; b. GM-CSF at a concentration between about 75 ng/mL and 125 ng/mL; c. SCF at a concentration between about 75 ng/mL and 125 ng/mL; and/or d. TPO at a concentration between about 75 ng/mL and 125 ng/mL.
- the stem cell can be a hematopoietic stem cell.
- Such instances can involve a step wherein the precursor media is removed prior to the addition of said myeloid media.
- the cell population can be washed at least once prior to contact with a myeloid medium.
- At least one wash step between contact with a precursor medium and myeloid medium may be beneficial to ensure cytokines and molecule components from the precursor medium are removed.
- the myeloid media can comprise a myeloid cytokine mixture comprising Macrophage colony-stimulating factor (MCSF), Interleukin-3 (IL-3), or Interleukin-6 (IL-6), or a combination thereof.
- MCSF Macrophage colony-stimulating factor
- IL-3 Interleukin-3
- IL-6 Interleukin-6
- the myeloid media comprises a myeloid cytokine mixture comprising Macrophage colonystimulating factor (MCSF), Interleukin-3 (IL-3), or Interleukin-6 (IL-6), or a combination thereof
- said myeloid media can comprise stem cells factor (SCF), Flt3 ligand, thrombopoietin (TPO), Granulocyte colony-stimulating factor (G-CSF), and Granulocyte-macrophage colony-stimulating factor (GM-CSF), at a concentration of at most 50 ng/mL, at most 40 ng/mL, at most 30 ng/mL, at most 20 ng/mL, at most 10 ng/mL, at most 5 ng/mL, or below 5 ng/mL.
- SCF stem cells factor
- Flt3 ligand Flt3 ligand
- TPO thrombopoietin
- G-CSF Granulocyte colony-stimulating factor
- the stem cell comprising a CAR is expanded by contacting said stem cell CAR cell with a precursor media comprising a precursor cytokine mixture, whereby the number of stem cell CAR cell(s) increases to generate a population of stem cells comprising a plurality of stem cell CAR cells, wherein following contact of said stem cell CAR cell with said precursor media, said stem cell CAR cell differentiates into a myeloid-lineage cell to generate a myeloid-derived CAR cell or a population comprising a plurality of myeloid- derived CAR cells, wherein said cell population is a mixed population comprising said stem cell CAR cells and said myeloid-derived CAR cells, wherein said precursor media comprises a precursor cytokine mixture comprising G-CSF, GM-CSF, SCF, and/or TPO, and wherein the stem cell CAR cell is maintained
- the precursor cytokine media can comprise: a. G-CSF at a concentration between about 100 ng/mL and 200 ng/mL; b. GM-CSF at a concentration between about 75 ng/mL and 125 ng/mL; c. SCF at a concentration between about 75 ng/mL and 125 ng/mL; and/or d. TPO at a concentration between about 75 ng/mL and 125 ng/mL.
- the stem cell can be a hematopoietic stem cell.
- the method can further comprise the step of contacting said stem cell CAR cell, myeloid-derived CAR cell, and/or said population comprising stem cell CAR cells and said myeloid-derived CAR cells with a myeloid media comprising a myeloid cytokine mixture, whereby said ratio of myeloid-derived CAR cells relative to stem cell CAR cells within the population is increased.
- said cell population comprising said stem cell CAR cells and said myeloid-derived CAR cells is maintained in said myeloid media until no more than 1%, 0.9%, 0,8%, 0,7%, 0.6%, 0.5%, 0.4%, 0.3%, 0.2%, 0.1%, or less than 0.1% of said population of cells is CD34+.
- Such instances can involve a step wherein the precursor media is removed prior to the addition of said myeloid media.
- the cell population can be washed at least once prior to contact with a myeloid medium.
- At least one wash step between contact with a precursor medium and myeloid medium may be beneficial to ensure cytokines and molecule components from the precursor medium are removed.
- the myeloid media can comprise a myeloid cytokine mixture comprising Macrophage colony-stimulating factor (MCSF), Interleukin-3 (IL-3), or Interleukin-6 (IL-6), or a combination thereof.
- MCSF Macrophage colony-stimulating factor
- IL-3 Interleukin-3
- IL-6 Interleukin-6
- the myeloid media comprises a myeloid cytokine mixture comprising Macrophage colony-stimulating factor (MCSF), Interleukin-3 (IL-3), or Interleukin-6 (IL-6), or a combination thereof
- said myeloid media can comprise stem cells factor (SCF), Flt3 ligand, thrombopoietin (TPO), Granulocyte colonystimulating factor (G-CSF), and Granulocyte-macrophage colony-stimulating factor (GM-CSF), at a concentration of at most 50 ng/mL, at most 40 ng/mL, at most 30 ng/mL, at most 20 ng/mL, at most 10 ng/mL, at most 5 ng/mL, or below 5 ng/mL.
- SCF stem cells factor
- Flt3 ligand Flt3 ligand
- TPO thrombopoietin
- G-CSF Granulocyte colonystimulating factor
- the stem cell CAR cell is expanded by contacting said stem cell CAR cell with a precursor media comprising a precursor cytokine mixture, whereby the number of stem cell CAR cell(s) increases to generate a population of stem cells comprising a plurality of stem cell CAR cells, wherein following contact of said stem cell CAR cell with said precursor media, said stem cell CAR cell differentiates into a myeloid-lineage cell to generate a myeloid-derived CAR cell or a population comprising a plurality of myeloid-derived CAR cells, wherein said cell population is a mixed population comprising said stem cell CAR cells and said myeloid-derived CAR cells, wherein said precursor media comprises a precursor cytokine mixture comprising G- CSF, GM-CSF, SCF, and/or TPO, and wherein the stem cell CAR cell is maintained in said precursor media until no more than 5%, no more than 4%, no
- the precursor cytokine media can comprise: a. G-CSF at a concentration between about 100 ng/mL and 200 ng/mL; b. GM-CSF at a concentration between about 75 ng/mL and 125 ng/mL; c. SCF at a concentration between about 75 ng/mL and 125 ng/mL; and/or d. TPO at a concentration between about 75 ng/mL and 125 ng/mL.
- the stem cell can be a hematopoietic stem cell.
- the method can further comprise the step of contacting said stem cell CAR cell, myeloid-derived CAR cell, and/or said population comprising stem cell CAR cells and said myeloid-derived CAR cells with a myeloid media comprising a myeloid cytokine mixture, whereby said ratio of myeloid-derived CAR cells relative to stem cell CAR cells within the population is increased.
- said cell population comprising said stem cell CAR cells and said myeloid-derived CAR cells is maintained in said myeloid media until no more than 1%, 0.9%, 0,8%, 0,7%, 0.6%, 0.5%, 0.4%, 0.3%, 0.2%, 0.1%, or less than 0.1% of said population of cells is CD34+.
- Such instances can involve a step wherein the precursor media is removed prior to the addition of said myeloid media.
- the cell population can be washed at least once prior to contact with a myeloid medium.
- At least one wash step between contact with a precursor medium and myeloid medium may be beneficial to ensure cytokines and molecule components from the precursor medium are removed.
- the myeloid media can comprise a myeloid cytokine mixture comprising Macrophage colony-stimulating factor (MCSF), Interleukin-3 (IL-3), or Interleukin-6 (IL-6), or a combination thereof.
- MCSF Macrophage colony-stimulating factor
- IL-3 Interleukin-3
- IL-6 Interleukin-6
- the myeloid media comprises a myeloid cytokine mixture comprising Macrophage colony-stimulating factor (MCSF), Interleukin-3 (IL-3), or Interleukin-6 (IL-6), or a combination thereof
- said myeloid media can comprise stem cells factor (SCF), Flt3 ligand, thrombopoietin (TPO), Granulocyte colonystimulating factor (G-CSF), and Granulocyte-macrophage colony-stimulating factor (GM-CSF), at a concentration of at most 50 ng/mL, at most 40 ng/mL, at most 30 ng/mL, at most 20 ng/mL, at most 10 ng/mL, at most 5 ng/mL, or below 5 ng/mL.
- SCF stem cells factor
- Flt3 ligand Flt3 ligand
- TPO thrombopoietin
- G-CSF Granulocyte colonystimulating factor
- the stem cell CAR cell is expanded by contacting said stem cell CAR cell with a precursor media comprising a precursor cytokine mixture, whereby the number of stem cell CAR cell(s) increases to generate a population of stem cells comprising a plurality of stem cell CAR cells, wherein following contact of said stem cell CAR cell with said precursor media, said stem cell CAR cell differentiates into a myeloid-lineage cell to generate a myeloid-derived CAR cell or a population comprising a plurality of myeloid-derived CAR cells, wherein said cell population is a mixed population comprising said stem cell CAR cells and said myeloid-derived CAR cells, wherein said precursor media comprises a precursor cytokine mixture comprising G- CSF, GM-CSF, SCF, and/or TPO, and wherein the stem cell CAR cell is maintained in said precursor media until at least 20%, at least 30%, at least 40%, at
- the precursor cytokine media can comprise: a. G-CSF at a concentration between about 100 ng/mL and 200 ng/mL; b. GM-CSF at a concentration between about 75 ng/mL and 125 ng/mL; c. SCF at a concentration between about 75 ng/mL and 125 ng/mL; and/or d. TPO at a concentration between about 75 ng/mL and 125 ng/mL.
- the stem cell can be a hematopoietic stem cell.
- the precursor cytokine media can comprise: a. G-CSF at a concentration between about 100 ng/mL and 200 ng/mL; b.
- the stem cell can be a hematopoietic stem cell.
- the method can further comprise the step of contacting said stem cell CAR cell, myeloid-derived CAR cell, and/or said population comprising stem cell CAR cells and said myeloid-derived CAR cells with a myeloid media comprising a myeloid cytokine mixture, whereby said ratio of myeloid-derived CAR cells relative to stem cell CAR cells within the population is increased.
- said cell population comprising said stem cell CAR cells and said myeloid-derived CAR cells is maintained in said myeloid media until no more than 1%, 0.9%, 0,8%, 0,7%, 0.6%, 0.5%, 0.4%, 0.3%, 0.2%, 0.1%, or less than 0.1% of said population of cells is CD34+.
- Such instances can involve a step wherein the precursor media is removed prior to the addition of said myeloid media.
- the cell population can be washed at least once prior to contact with a myeloid medium.
- At least one wash step between contact with a precursor medium and myeloid medium may be beneficial to ensure cytokines and molecule components from the precursor medium are removed.
- the myeloid media can comprise a myeloid cytokine mixture comprising Macrophage colony-stimulating factor (MCSF), Interleukin-3 (IL-3), or Interleukin-6 (IL-6), or a combination thereof.
- MCSF Macrophage colony-stimulating factor
- IL-3 Interleukin-3
- IL-6 Interleukin-6
- the myeloid media comprises a myeloid cytokine mixture comprising Macrophage colony-stimulating factor (MCSF), Interleukin-3 (IL-3), or Interleukin-6 (IL-6), or a combination thereof
- said myeloid media can comprise stem cells factor (SCF), Flt3 ligand, thrombopoietin (TPO), Granulocyte colony-stimulating factor (G-CSF), and Granulocytemacrophage colony-stimulating factor (GM-CSF), at a concentration of at most 50 ng/mL, at most 40 ng/mL, at most 30 ng/mL, at most 20 ng/mL, at most 10 ng/mL, at most 5 ng/mL, or below 5 ng/mL.
- SCF stem cells factor
- Flt3 ligand Flt3 ligand
- TPO thrombopoietin
- G-CSF Granulocyte colony-stimulating factor
- the stem cell CAR cell is expanded by contacting said stem cell CAR cell with a precursor media comprising a precursor cytokine mixture, whereby the number of stem cell CAR cell(s) increases to generate a population of stem cells comprising a plurality of stem cell CAR cells, wherein following contact of said stem cell CAR cell with said precursor media, said stem cell CAR cell differentiates into a myeloid-lineage cell to generate a myeloid-derived CAR cell or a population comprising a plurality of myeloid-derived CAR cells, wherein said cell population is a mixed population comprising said stem cell CAR cells and said myeloid-derived CAR cells, wherein said precursor media comprises a precursor cytokine mixture comprising G- CSF, GM-CSF, SCF, and/or TPO, and wherein the stem cell CAR cell is maintained in said precursor media until between about 60%-95% of said population of cells is CD15
- the precursor cytokine media can comprise: a. G-CSF at a concentration between about 100 ng/mL and 200 ng/mL; b. GM-CSF at a concentration between about 75 ng/mL and 125 ng/mL; c. SCF at a concentration between about 75 ng/mL and 125 ng/mL; and/or d. TPO at a concentration between about 75 ng/mL and 125 ng/mL.
- the stem cell can be a hematopoietic stem cell.
- the method can further comprise the step of contacting said stem cell CAR cell, myeloid-derived CAR cell, and/or said population comprising stem cell CAR cells and said myeloid-derived CAR cells with a myeloid media comprising a myeloid cytokine mixture, whereby said ratio of myeloid-derived CAR cells relative to stem cell CAR cells within the population is increased.
- said cell population comprising said stem cell CAR cells and said myeloid-derived CAR cells is maintained in said myeloid media until no more than 1%, 0.9%, 0,8%, 0,7%, 0.6%, 0.5%, 0.4%, 0.3%, 0.2%, 0.1%, or less than 0.1% of said population of cells is CD34+.
- Such instances can involve a step wherein the precursor media is removed prior to the addition of said myeloid media.
- the cell population can be washed at least once prior to contact with a myeloid medium.
- At least one wash step between contact with a precursor medium and myeloid medium may be beneficial to ensure cytokines and molecule components from the precursor medium are removed.
- the myeloid media can comprise a myeloid cytokine mixture comprising Macrophage colony-stimulating factor (MCSF), Interleukin-3 (IL-3), or Interleukin-6 (IL-6), or a combination thereof.
- MCSF Macrophage colony-stimulating factor
- IL-3 Interleukin-3
- IL-6 Interleukin-6
- the myeloid media comprises a myeloid cytokine mixture comprising Macrophage colony-stimulating factor (MCSF), Interleukin-3 (IL-3), or Interleukin-6 (IL-6), or a combination thereof
- said myeloid media can comprise stem cells factor (SCF), Flt3 ligand, thrombopoietin (TPO), Granulocyte colony-stimulating factor (G-CSF), and Granulocytemacrophage colony-stimulating factor (GM-CSF), at a concentration of at most 50 ng/mL, at most 40 ng/mL, at most 30 ng/mL, at most 20 ng/mL, at most 10 ng/mL, at most 5 ng/mL, or below 5 ng/mL.
- SCF stem cells factor
- Flt3 ligand Flt3 ligand
- TPO thrombopoietin
- G-CSF Granulocyte colony-stimulating factor
- the stem cell comprising a CAR is expanded by contacting said stem cell CAR cell with a precursor media comprising a precursor cytokine mixture, whereby the number of stem cell CAR cell(s) increases to generate a population of stem cells comprising a plurality of stem cell CAR cells, wherein following contact of said stem cell CAR cell with said precursor media, said stem cell CAR cell differentiates into a myeloid-lineage cell to generate a myeloid-derived CAR cell or a population comprising a plurality of myeloid- derived CAR cells, wherein said cell population is a mixed population comprising said stem cell CAR cells and said myeloid-derived CAR cells, wherein said precursor media comprises a precursor cytokine mixture comprising G-CSF, GM-CSF, SCF, and/or TPO, and wherein the stem cell CAR cell is maintained
- the precursor cytokine media can comprise: a. G-CSF at a concentration between about 100 ng/mL and 200 ng/mL; b. GM-CSF at a concentration between about 75 ng/mL and 125 ng/mL; c. SCF at a concentration between about 75 ng/mL and 125 ng/mL; and/or d. TPO at a concentration between about 75 ng/mL and 125 ng/mL.
- the stem cell can be a hematopoietic stem cell.
- the method can further comprise the step of contacting said stem cell CAR cell, myeloid-derived CAR cell, and/or said population comprising stem cell CAR cells and said myeloid-derived CAR cells with a myeloid media comprising a myeloid cytokine mixture, whereby said ratio of myeloid-derived CAR cells relative to stem cell CAR cells within the population is increased.
- said cell population comprising said stem cell CAR cells and said myeloid-derived CAR cells is maintained in said myeloid media until no more than 1%, 0.9%, 0,8%, 0,7%, 0.6%, 0.5%, 0.4%, 0.3%, 0.2%, 0.1%, or less than 0.1% of said population of cells is CD34+.
- Such instances can involve a step wherein the precursor media is removed prior to the addition of said myeloid media.
- the cell population can be washed at least once prior to contact with a myeloid medium.
- At least one wash step between contact with a precursor medium and myeloid medium may be beneficial to ensure cytokines and molecule components from the precursor medium are removed.
- the myeloid media can comprise a myeloid cytokine mixture comprising Macrophage colony-stimulating factor (MCSF), Interleukin-3 (IL-3), or Interleukin-6 (IL-6), or a combination thereof.
- MCSF Macrophage colony-stimulating factor
- IL-3 Interleukin-3
- IL-6 Interleukin-6
- the myeloid media comprises a myeloid cytokine mixture comprising Macrophage colonystimulating factor (MCSF), Interleukin-3 (IL-3), or Interleukin-6 (IL-6), or a combination thereof
- said myeloid media can comprise stem cells factor (SCF), Flt3 ligand, thrombopoietin (TPO), Granulocyte colony-stimulating factor (G-CSF), and Granulocyte-macrophage colony-stimulating factor (GM-CSF), at a concentration of at most 50 ng/mL, at most 40 ng/mL, at most 30 ng/mL, at most 20 ng/mL, at most 10 ng/mL, at most 5 ng/mL, or below 5 ng/mL.
- SCF stem cells factor
- Flt3 ligand Flt3 ligand
- TPO thrombopoietin
- G-CSF Granulocyte colony-stimulating factor
- the stem cell CAR cell is expanded by contacting said stem cell CAR cell with a precursor media comprising a precursor cytokine mixture, whereby the number of stem cell CAR cell(s) increases to generate a population of stem cells comprising a plurality of stem cell CAR cells, wherein following contact of said stem cell CAR cell with said precursor media, said stem cell CAR cell differentiates into a myeloid-lineage cell to generate a myeloid-derived CAR cell or a population comprising a plurality of myeloid-derived CAR cells, wherein said cell population is a mixed population comprising said stem cell CAR cells and said myeloid-derived CAR cells, wherein said precursor media comprises a precursor cytokine mixture comprising G- CSF, GM-CSF, SCF, and/or TPO, and wherein the stem cell CAR cell is maintained in said precursor media until between about 60%-95% of said population of cells is CD15
- the precursor cytokine media can comprise: a. G-CSF at a concentration between about 100 ng/mL and 200 ng/mL; b. GM-CSF at a concentration between about 75 ng/mL and 125 ng/mL; c. SCF at a concentration between about 75 ng/mL and 125 ng/mL; and/or d. TPO at a concentration between about 75 ng/mL and 125 ng/mL.
- the stem cell can be a hematopoietic stem cell.
- the method can further comprise the step of contacting said stem cell CAR cell, myeloid- derived CAR cell, and/or said population comprising stem cell CAR cells and said myeloid-derived CAR cells with a myeloid media comprising a myeloid cytokine mixture, whereby said ratio of myeloid-derived CAR cells relative to stem cell CAR cells within the population is increased.
- said cell population comprising said stem cell CAR cells and said myeloid-derived CAR cells is maintained in said myeloid media until no more than 1%, 0.9%, 0,8%, 0,7%, 0.6%, 0.5%, 0.4%, 0.3%, 0.2%, 0.1%, or less than 0.1% of said population of cells is CD34+.
- Such instances can involve a step wherein the precursor media is removed prior to the addition of said myeloid media.
- the cell population can be washed at least once prior to contact with a myeloid medium.
- At least one wash step between contact with a precursor medium and myeloid medium may be beneficial to ensure cytokines and molecule components from the precursor medium are removed.
- the myeloid media can comprise a myeloid cytokine mixture comprising Macrophage colony-stimulating factor (MCSF), Interleukin-3 (IL-3), or Interleukin-6 (IL-6), or a combination thereof.
- MCSF Macrophage colony-stimulating factor
- IL-3 Interleukin-3
- IL-6 Interleukin-6
- the myeloid media comprises a myeloid cytokine mixture comprising Macrophage colony-stimulating factor (MCSF), Interleukin-3 (IL-3), or Interleukin-6 (IL-6), or a combination thereof
- said myeloid media can comprise stem cells factor (SCF), Flt3 ligand, thrombopoietin (TPO), Granulocyte colony-stimulating factor (G-CSF), and Granulocyte-macrophage colony-stimulating factor (GM- CSF), at a concentration of at most 50 ng/mL, at most 40 ng/mL, at most 30 ng/mL, at most 20 ng/mL, at most 10 ng/mL, at most 5 ng/mL, or below 5 ng/mL.
- SCF stem cells factor
- Flt3 ligand Flt3 ligand
- TPO thrombopoietin
- G-CSF Granulocyte colony-stimulating factor
- the stem cell CAR cell is expanded by contacting said stem cell CAR cell with a precursor media comprising a precursor cytokine mixture, whereby the number of stem cell CAR cell(s) increases to generate a population of stem cells comprising a plurality of stem cell CAR cells, wherein following contact of said stem cell CAR cell with said precursor media, said stem cell CAR cell differentiates into a myeloid-lineage cell to generate a myeloid-derived CAR cell or a population comprising a plurality of myeloid-derived CAR cells, wherein said cell population is a mixed population comprising said stem cell CAR cells and said myeloid-derived CAR cells, wherein said precursor media comprises a precursor cytokine mixture comprising G- CSF, GM-CSF, SCF, and/or TPO, and wherein the stem cell CAR cell is maintained in said precursor media until at least 20%, at least 30%, at least 40%, at
- the precursor cytokine media can comprise: a. G-CSF at a concentration between about 100 ng/mL and 200 ng/mL; b. GM-CSF at a concentration between about 75 ng/mL and 125 ng/mL; c. SCF at a concentration between about 75 ng/mL and 125 ng/mL; and/or d. TPO at a concentration between about 75 ng/mL and 125 ng/mL.
- the stem cell can be a hematopoietic stem cell.
- the method can further comprise the step of contacting said stem cell CAR cell, myeloid-derived CAR cell, and/or said population comprising stem cell CAR cells and said myeloid-derived CAR cells with a myeloid media comprising a myeloid cytokine mixture, whereby said ratio of myeloid-derived CAR cells relative to stem cell CAR cells within the population is increased.
- said cell population comprising said stem cell CAR cells and said myeloid-derived CAR cells is maintained in said myeloid media until no more than 1%, 0.9%, 0,8%, 0,7%, 0.6%, 0.5%, 0.4%, 0.3%, 0.2%, 0.1%, or less than 0.1% of said population of cells is CD34+.
- Such instances can involve a step wherein the precursor media is removed prior to the addition of said myeloid media.
- the cell population can be washed at least once prior to contact with a myeloid medium.
- At least one wash step between contact with a precursor medium and myeloid medium may be beneficial to ensure cytokines and molecule components from the precursor medium are removed.
- the myeloid media can comprise a myeloid cytokine mixture comprising Macrophage colony-stimulating factor (MCSF), Interleukin-3 (IL-3), or Interleukin-6 (IL-6), or a combination thereof.
- MCSF Macrophage colony-stimulating factor
- IL-3 Interleukin-3
- IL-6 Interleukin-6
- the myeloid media comprises a myeloid cytokine mixture comprising Macrophage colonystimulating factor (MCSF), Interleukin-3 (IL-3), or Interleukin-6 (IL-6), or a combination thereof
- said myeloid media can comprise stem cells factor (SCF), Flt3 ligand, thrombopoietin (TPO), Granulocyte colony-stimulating factor (G-CSF), and Granulocyte-macrophage colony-stimulating factor (GM-CSF), at a concentration of at most 50 ng/mL, at most 40 ng/mL, at most 30 ng/mL, at most 20 ng/mL, at most 10 ng/mL, at most 5 ng/mL, or below 5 ng/mL.
- SCF stem cells factor
- Flt3 ligand Flt3 ligand
- TPO thrombopoietin
- G-CSF Granulocyte colony-stimulating factor
- the myeloid media comprises a myeloid cytokine mixture comprising Macrophage colonystimulating factor (MCSF), Interleukin-3 (IL-3), or Interleukin-6 (IL-6), or a combination thereof
- said myeloid media can comprise stem cells factor (SCF), Flt3 ligand, thrombopoietin (TPO), Granulocyte colony-stimulating factor (G-CSF), and Granulocyte-macrophage colony-stimulating factor (GM-CSF), at a concentration of at most 50 ng/mL, at most 40 ng/mL, at most 30 ng/mL, at most 20 ng/mL, at most 10 ng/mL, at most 5 ng/mL, or below 5 ng/mL.
- SCF stem cells factor
- Flt3 ligand Flt3 ligand
- TPO thrombopoietin
- G-CSF Granulocyte colony-stimulating factor
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Epidemiology (AREA)
- Engineering & Computer Science (AREA)
- Organic Chemistry (AREA)
- Chemical & Material Sciences (AREA)
- Biomedical Technology (AREA)
- Immunology (AREA)
- Zoology (AREA)
- Wood Science & Technology (AREA)
- Genetics & Genomics (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Biotechnology (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Cell Biology (AREA)
- Hematology (AREA)
- Medicinal Chemistry (AREA)
- Microbiology (AREA)
- Pharmacology & Pharmacy (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Biochemistry (AREA)
- General Engineering & Computer Science (AREA)
- Micro-Organisms Or Cultivation Processes Thereof (AREA)
- Medicines Containing Material From Animals Or Micro-Organisms (AREA)
Abstract
Provided herein are compositions and methods for manufacturing myeloid-derived cells, and populations comprising the same, from hematopoietic stem cells (HSCs). The precursor and myeloid media compositions disclosed herein are particularly effective at expanding and differentiating HSCs into myeloid-derived cells. The HSCs can be genetically modified with a chimeric antigen receptor (CAR) and then expanded and differentiated to generate a myeloid-derived lineage cell expressing a CAR. Also disclosed are genetically modified stem cells and myeloid-derived cells comprising a CAR and compositions and methods for using the same for treatment of a disease and/or disorder in a subject.
Description
METHODS OF MANUFACTURING MYELOID-DERIVED CELLS FROM HEMATOPOIETIC STEM CELLS AND COMPOSITIONS AND USES THEREOF
CROSS-REFERENCE TO RELATED APPLICATIONS
This application claims priority to U.S. Provisional Application No. 63/579,469, filed August 29, 2023, the contents of which are incorporated by reference herein in their entirety.
STATEMENT REGARDING THE SEQUENCE LISTING
The instant application contains an electronic Sequence Listing which has been submitted in xml (ST.26) format concurrently herewith, herein incorporated by reference in its entirety. Said xml copy named “1115814 1030WO.xml” is 14,753 bytes in size, and was created on August 28, 2024.
FIELD OF THE DISCLOSURE
The present disclosure relates to myeloid cells and genetically modified myeloid cells, precursors thereof, and improved methods of manufacturing the same. More specifically, the present disclosure relates to methods of manufacturing myeloid lineage cells from stem cells, such as hematopoietic stem cells (HSCs). The stem cells can be genetically modified with a chimeric antigen receptor (CAR) to generate genetically modified myeloid-derived CAR cells, whereby the HSCs are genetically modified with a chimeric antigen receptor (CAR) prior to expansion and differentiation into myeloid lineage cells.
BACKGROUND OF THE DISCLOSURE
Chimeric antigen receptors (CARs) are engineered receptors that can combine a desired specificity with the functionality of an immune cell to target cancer cells. Typically, these receptors graft the specificity of a monoclonal antibody onto a T cell. The receptors are called chimeric because they are fusions of parts from different sources. CAR-T cell therapy refers to a treatment that uses such transformed cells primarily for cancer therapy. The general premise of CAR-engineered immune cells is to endow such cells with the ability to target markers found on diseased cells, e.g., cancer cells. In the case of CAR-T cells, scientists can remove T cells from a person, genetically alter them to express a CAR, and put them back into the patient for them to attack the diseased cells. Currently, tumor immunotherapy has mainly focused on adaptive immune systems, such as T cells and B cells. Innate immune cells, such as macrophages, have not been widely studied in cancer treatment. Given the
current success of CAR-T cell therapy and the potential for development of CAR-natural killer cells, a developing interest exists in developing CAR-macrophages (CAR-M) for tumor immunotherapy.
Taking into account their capabilities of phagocytosis, antigen presentation and penetration in the tumor microenvironment, macrophages present a promising cell for use in cancer therapy. Macrophages are well recognized as effector cells that eliminate cancer cells due to their phagocytic ability and are therefore drawing attention in the field of immunotherapy of cancers.
CAR-M therapy has shown its effective anti-tumor ability in animal experiments. Compared with CAR-T and CAR-NK, CAR-M has its unique advantages as a new cell immunotherapy, but it also has many shortcomings that must be overcome. Peripheral and mobilized monocytes present significant challenges as sources for CAR-M. For example, monocytes do not proliferate, have a short half-life, and must extravasate through vessels and into tissues to differentiate into long-lived macrophages. Engineering monocytes is a significant challenge that results in loss of viability and migratory capacity.
Thus, a need exists in the art for compositions and methods that enable the production of myeloid cells, including myeloid cells expressing a CAR, such as CAR-M cells, that can be effectively used to treat disease and disorders such as cancers.
The present application is directed to improved methods of generating myeloid-derived cells, and myeloid-derived CAR cells, such as CAR-M cells, from precursor cells, such as stem cells, and associated compositions and methods for use of the same.
BRIEF SUMMARY OF THE DISCLOSURE
Provided herein are methods of manufacturing a myeloid-derived cell, said method comprising the step of: contacting a precursor cell with a precursor media comprising a precursor cytokine mixture, whereby the number of precursor cell(s) increases to generate a cell population comprising a plurality of precursor cells. In some embodiments of the methods of manufacturing a myeloid-derived cell, the precursor cell is a stem cell.
In some embodiments of the above methods of manufacturing a myeloid-derived cell, the method comprises the step of: contacting said precursor cell with a nucleic acid molecule comprising a polynucleotide sequence encoding a chimeric antigen receptor (CAR), whereby said nucleic acid is introduced into said precursor cell to generate a precursor CAR cell comprising a chimeric antigen receptor (CAR). In some embodiments of the above methods, said precursor cell is contacted with said nucleic acid molecule prior to, or concurrently with, being contacted with said precursor media.
In some embodiments of the above methods of manufacturing a myeloid-derived cell, the method comprises the steps of: a) contacting said precursor cell with a nucleic acid molecule comprising a polynucleotide sequence encoding a chimeric antigen receptor (CAR), whereby said nucleic acid is introduced into said precursor cell to generate a precursor CAR cell comprising a chimeric antigen receptor (CAR), and b) contacting said precursor CAR cell with a precursor media comprising a precursor cytokine mixture, whereby the number of precursor CAR cell(s) increases to generate a cell population comprising a plurality of precursor CAR cells, wherein step a) occurs prior to step b).
In some embodiments of the above methods of manufacturing a myeloid-derived cell, the method further comprises the step of: c) contacting said cell population comprising a plurality of precursor cells and/or said cell population comprising a plurality of precursor CAR cells with a myeloid media comprising a myeloid cytokine mixture, whereby said precursor cell(s) differentiates into a myeloid-lineage cell to generate a myeloid-derived cell and/or a mixed cell population comprising a plurality of myeloid-derived cells and/or a plurality of precursor cells, and/or said precursor CAR cell(s) differentiates into a myeloid-lineage cell to generate a myeloid- derived CAR cell and/or a mixed cell population comprising a plurality of myeloid-derived CAR cells and/or a plurality of said precursor CAR cells.
In some embodiments of the above methods of manufacturing a myeloid-derived cell, upon contact of said precursor cell(s) with said precursor media, said precursor cell(s) differentiates into a myeloid-lineage cell to generate a myeloid-derived cell and/or a mixed cell population comprising a plurality of myeloid-derived cells and a plurality of precursor cells, or upon contact of said precursor CAR cell(s) with said precursor media, said precursor CAR cell(s) differentiates into a myeloid-lineage cell to generate a myeloid-derived CAR cell and/or a mixed cell population comprising a plurality of myeloid-derived CAR cells and a plurality of precursor CAR cells.
In some embodiments of the above methods of manufacturing a myeloid-derived cell, the method further comprises the step of: c) contacting said mixed cell population with a myeloid media comprising a myeloid cytokine mixture, whereby upon contact with said myeloid media: a. the ratio of said myeloid-derived cells to said precursor cells in said mixed cell population is increased; and/or b. the ratio of myeloid-derived CAR cells relative to precursor CAR cells in said mixed cell population is increased.
In some embodiments of the above methods of manufacturing a myeloid-derived cell, the precursor media is removed prior to addition of the myeloid media.
In some embodiments of the above methods of manufacturing a myeloid-derived cell, the stem cell is a bone-marrow-derived stem cell. In some embodiments of the above methods, said
stem cell is a hematopoietic stem cell (HSC). In some embodiments of the above methods, said stem cell is CD34+. In some embodiments of the above methods, said stem cell is a pluripotent stem cell. In some embodiments of the above methods, said stem cell is isolated from a subject. In some embodiments of the above methods, said subject is a human subject. In some embodiments of the above methods, said isolated stem cell is cryopreserved prior to contacting said cell with a precursor media and/or contacting said cell with a nucleic acid molecule. In some embodiments of the above methods, said cryopreserved stem cell is thawed prior to contacting said cell with a precursor media and/or contacting said cell with a nucleic acid molecule. In some embodiments of the above methods, said stem cell is present within a population of stem cells. In some embodiments of the above methods, said population comprises a plurality of stem cell(s). In some embodiments of the above methods, between 80%-99.9% of said population is CD34+ cells. In some embodiments of the above methods, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% of said population is CD34+ cells.
In some embodiments of the above methods of manufacturing a myeloid-derived cell, said stem cell is maintained in a culture medium for between 2-144 hours following contact with said nucleic acid molecule. In some embodiments of the above methods, while said stem cell is maintained within said culture medium, said polynucleotide encoding is integrated into the stem cell chromosome. In some embodiments of the above methods, a selection agent is added to said culture medium.
In some embodiments of the above methods of manufacturing a myeloid-derived cell, said precursor cytokine mixture comprises at least one cytokine selected from the following: stem cells factor (SCF), Flt3 ligand, thrombopoietin (TPO), Interleukin-3 (IL-3); Granulocyte colonystimulating factor (G-CSF), Interleukin-6 (IL-6), Granulocyte-macrophage colony-stimulating factor (GM-CSF), and Macrophage colony-stimulating factor (MCSF). In some embodiments of the above methods, said precursor media comprises G-CSF at a concentration between about 100 ng/mL and 200 ng/mL. In some embodiments of the above methods, precursor media comprises G- CSF at a concentration between about 100 ng/mL and 150 ng/mL. In some embodiments of the above methods, precursor cytokine mixture comprises G-CSF, GM-CSF, SCF, and TPO. In some embodiments of the above methods, said precursor media comprises GM-CSF at a concentration between about 75 ng/mL and 125 ng/mL. In some embodiments of the above methods, said precursor media comprises SCF at a concentration between about 75 ng/mL and 125 ng/mL. In some embodiments of the above methods, precursor media comprises TPO at a concentration between about 75 ng/mL and 125 ng/mL. In some embodiments of the above methods, the
precursor media comprises G-CSF at a concentration of 150 ng/mL, GM-CSF at a concentration of 100 ng/mL, SCF at a concentration of 100 ng/mL, and/or TPO at a concentration of 100 ng/mL.
In some embodiments of the above methods of manufacturing a myeloid-derived cell, said myeloid media comprises a myeloid cytokine mixture comprising Macrophage colony-stimulating factor (MCSF), Interleukin-3 (IL-3), and/or Interleukin-6 (IL-6), and/or a combination thereof. In some embodiments of the above methods, said myeloid media comprises stem cells factor (SCF), Flt3 ligand, thrombopoietin (TPO), Granulocyte colony-stimulating factor (G-CSF), and/or Granulocyte-macrophage colony-stimulating factor (GM-CSF), at a concentration of at most 50 ng/mL, at most 40 ng/mL, at most 30 ng/mL, at most 20 ng/mL, at most 10 ng/mL, at most 5 ng/mL, or below 5 ng/mL.
In some embodiments of the above methods of manufacturing a myeloid-derived cell, said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population, is maintained in said precursor media until the number of precursor cell(s), precursor CAR cells, myeloid-derived cells and/or myeloid-derived CAR cells is increased by at least 20-fold, at least 50-fold, at least 75-fold, at least 100-fold, at least 200-fold, at least 300-fold, at least 400-fold, at least 500-fold, or more than 500-fold. In some embodiments of the above methods, said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population, is maintained in said precursor media until the number of precursor cell(s), precursor CAR cells, myeloid-derived cells and/or myeloid-derived CAR cells is at least 104, at least 105, at least 106, at least 107, at least 108, at least 109, at least 1010, at least 1011, at least 1012, at least 1013, at least 1014, at least 1015, at least 1016, at least 1017, at least 1018, at least 1019, at least IO20, or more than IO20. In some embodiments of the above methods, said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population, is maintained in said precursor media for at least 2, at least 3, at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, at least 10, at least 11, or at least 12 days. In some embodiments of the above methods, said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population, is maintained in precursor media for between 9-11 days. In some embodiments of the above methods, said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a
plurality of precursor CAR cells, and/or said mixed cell population, is maintained in precursor media for 10 days.
In some embodiments of the above methods of manufacturing a myeloid-derived cell, said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population, is maintained in said precursor media until: a. no more than 99%, no more than 98%, no more than 97%, no more than 96%, no more than 95%, no more than 90%, no more than 80%, no more than 70%, no more than 60%, no more than 50%, no more than 40%, no more than 30%, no more than 20%, no more than 10%, no more than 5%, no more than 1%, or no more than 0.1% of cells within said mixed cell population are CD34+; b. no more than 5%, no more than 4%, no more than 3%, no more than 2%, no more than 1%, or no more than 0.1% of cells within said mixed cell population are CD34+; c. at least 20%, at least 30%, at least 40%, at least 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95% of cells within said mixed cell population are CD15+; d. between about 60%-85% of cells within said mixed cell population areCD15+; e. at least 2%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, at least 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of cells within said mixed cell population are CD1 lb+; f. between about 30%- 80% of cells within said mixed cell population are CD1 lb+; g. at least 0.1%, at least 0.5%, at least 1%, at least 2%, at least 3%, at least 4%, at least 5%, at least 6%, at least 7%, at least 8%, at least 9%, or at least 10% of cells within said mixed cell population are CD14+; h. between about 1%- 10% of cells within said mixed cell population are CD14+; i.at least 20%, at least 30%, at least 40%, at least 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95% of cells within said mixed cell population are CD13+; and/or j. between about 85%-95% of cells within said mixed cell population are CD13+. In some embodiments of the above methods, said mixed population is contacted with said myeloid media after contact with said precursor media.
In some embodiments of the above methods, said mixed population is maintained in said myeloid media until: a. no more than 5%, no more than 4%, no more than 3%, no more than 2%, no more than 1%, no more than 0.9%, no more than 0.8%, no more than 0.7%, no more than 0.6%, no more than 0.5%, no more than 0.4%, no more than 0.3%, no more than 0.2%, no more than 0.1%, or less than 0.1% of cells within said mixed cell population are CD34+; b. at least 70%, at least 75%, at least 80%, at least 90%, or at least 95% of cells within said mixed cell population are CD15+; c. between about 50%-95% of cells within said mixed cell population are CD15+; d. at
least 70%, least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or at least 99.5% of cells within said mixed cell population are CD1 lb+; e. between about 75%-99.5% of cells within said mixed cell population are CD1 lb+; f. at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, at least 50%, at least 60%, at least 70%, at least 80%, or at least 90% of cells within said mixed cell population are CD14+; g. between about 5%-90% of cells within said mixed cell population are CD14+; h. at least 85%, at least 90%, at least 95%, or at least 99% of cells within said mixed cell population are CD13+; and/or i. between about 85%-99% of cells within said mixed cell population are CD13+.
In some embodiments of the above methods, said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population, is maintained in a myeloid media for at least 1, at least 2, at least 3, at least 4, at least 5, at least 6, at least 7 or at least 8 days. In some embodiments of the above methods, said mixed cell population comprises at least 20%, at least 40%, at least 60%, least 80%, at least 85%, or at least 90% CD14+ cells after 7 days of being maintained in said myeloid media.
In some embodiments of the above methods, contacting said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population with said precursor cytokine mixture reduces the ratio of CD34+ cells within said population comprising a plurality of precursor cells, said population comprising a plurality of precursor CAR cells, and/or said mixed cell population in a shorter amount of time compared to a control that is contacted with and/or maintained in a precursor media lacking said precursor cytokine mixture.
In some embodiments of the above methods, contacting said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population with said precursor media increases: a. the number of CD1 lb+ cells; b. the number of CD13+ cells; c. the number of CD15+ cells; and/or d. the number of CD14+ cells within said population comprising a plurality of precursor cells, said population comprising a plurality of precursor CAR cells, and/or said mixed cell population in a shorter amount of time compared to a control that is contacted in and/or maintained in a precursor media lacking said precursor cytokine mixture
In some embodiments of the above methods, contacting said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population with said precursor media increases: a. the ratio of CD1 lb+ to CD1 lb- cells; b. the ratio of CD13+ to
CD13- cells; c. the ratio of CD15+ to CD15- cells; and/or d. the ratio of CD14+ to CD14- cells within said population comprising a plurality of precursor cells, said population comprising a plurality of precursor CAR cells, and/or said mixed cell population in a shorter amount of time compared to a control that is contacted with and/or maintained in a precursor media lacking said precursor cytokine mixture.
In some embodiments of the above methods, at least 40%, at least 50%, at least 60%, at least 70%, at least 80%, at least 90%, or at least 95% of cells within said population comprising a plurality of precursor cells, said population comprising a plurality of precursor CAR cells, and/or said mixed cell population comprises said CAR, or a functional fragment thereof, on the cell surface.
In some embodiments of the above methods, said myeloid-derived cell is a phagocytic cell. In some embodiments of the above methods, said phagocytic cell is a monocyte, macrophage, dendritic cell, neutrophil, or a precursor thereof. In some embodiments of the above methods, said phagocytic cell is a monocyte. In some embodiments of the above methods, said myeloid-derived cell is a myeloid progenitor cell.
In some embodiments of the above methods, said CAR comprises: an antigen binding domain; a transmembrane domain; and an intracellular signaling domain. In some embodiments of the above methods, said CAR further comprises an intracellular co-stimulatory signaling domain. In some embodiments of the above methods, said CAR comprises an intracellular co-stimulatory signaling domain having an amino acid sequence having at least 90% sequence identity to a cytoplasmic domain of a herpes virus entry mediator (HVEM) protein set forth as any one of SEQ ID NOs: 1, 2, or 3, or a functional fragment or variant thereof that retains co-stimulatory activity.
Also provided herein is a genetically modified cell comprising a chimeric antigen receptor (CAR), wherein the genetically modified cell is a precursor cell, a myeloid-derived cell, a myeloid- lineage cell, a myeloid progenitor cell, and/or a precursor thereof.
In some embodiments of the above CAR cell, the precursor cell comprises a bone marrow- derived cell, a blood-derived cell, or a stem cell. In some embodiments of the above CAR cell, the stem cell is a hematopoietic stem cell (HSC). In some embodiments of the above CAR cell, the cell is a myeloid precursor cell, such as a myeloid progenitor cell, granulo-monocyte progenitor cell, monocyte-dendritic cell progenitor/monoblasts, promonocyte, or myeloblast. In some embodiments of the above CAR cell, said CAR comprises an intracellular co-stimulatory signaling domain having an amino acid sequence having at least 90% sequence identity to a cytoplasmic domain of a herpes virus entry mediator (HVEM) protein set forth as any one of SEQ ID NOs: 1, 2, or 3, or a functional fragment or variant thereof that retains co-stimulatory activity.
Also provided herein is a cell population comprising any of the genetically modified CAR cells disclosed herein. In some embodiments, the cell population is generated by any of the methods of manufacturing a myeloid-derived cell disclosed herein. In some embodiments of the above cell populations, at least 40%, at least 50%, at least 60%, at least 70%, at least 80%, at least 90%, at least 95%, or at least 99% of cells within said population expresses said CAR on the cell surface. In some embodiments of the above cell populations: a. no more than 5%, no more than 4%, no more than 3%, no more than 2%, no more than 1%, no more than 0.5%, or no more than 0.1% of cells within said population are CD34+; b. at least 50%, at least 55%, at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 86%, at least 87%, at least 88%, at least 89%, or at least 90% of cells within said population are CD14+; c. at least 70%, at least 80%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or at least 99.5%% of cells within said population are CD1 lb+; d. at least 90%, at least 91%, at least 92%, at least 94%, or at least 95% of cells within said population are CD13+; and/or e. at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, or at least 95% of cells within said population are CD15+.
Also provided herein is a pharmaceutical composition comprising any of the genetically modified cells disclosed herein, or any of the cell populations disclosed herein, and a pharmaceutically acceptable carrier.
Also provided herein are methods of treating a disease or condition associated with a tumor or cancer in a subject in need thereof, comprising administering to the subject a therapeutically effective amount of any of the pharmaceutical compositions disclosed herein.
Also provided herein are methods of treating a tumor in a subject in need thereof, comprising administering to the subject a therapeutically effective amount of any of the pharmaceutical compositions disclosed herein.
Also provided herein are methods of treating an infection in a subject in need thereof, comprising administering to the subject a therapeutically effective amount of any of the pharmaceutical compositions disclosed herein.
Also provided herein are methods for stimulating an immune response to a target tumor cell or tumor tissue in a subject in need thereof, comprising administering to the subject a therapeutically effective amount of any of the pharmaceutical compositions disclosed herein.
Also provided herein are methods of treating a subject by engulfment and/or trogocytosis of a target cell in the subject, comprising administering to the subject a therapeutically effective amount of any of the pharmaceutical compositions disclosed herein.
BRIEF DESCRIPTION OF THE DRAWINGS
FIGS. 1A-1B show schematics depicting exemplary workflow for generating myeloid-derived cells (FIG. 1A) and genetically modified myeloid-derived CAR cells (FIG. IB) hematopoietic stem cells (HSCs). FIG. 1A shows that mobilized HSCs can be expanded and differentiated into myeloid- lineage cells. FIG. IB shows that HSCs can be engineered with a CAR prior to expansion and differentiation into myeloid-derived CAR cells.
FIGS. 2A-2N show the duration and stability of transduced chimeric antigen receptor (CAR- M83-GFP) integration in a starting population of precursor CAR cells subsequently expanded and differentiated into myeloid-derived cells using the methods disclosed herein. CAR surface expression and GFP expression were analyzed by flow cytometry and presented as expression intensity (x-axis) against percentage of cell population normalized to mode for the indicated population (y-axis). FIG. 2A shows a histogram depicting CAR expression in transduced cells (“CAR”) compared to control cells (untransduced; “UTD”) in precursor media 6 days, and 72-hours post-transduction of a polynucleotide encoding a CAR. FIG. 2B shows a histogram depicting CAR expression in transduced cells (“CAR”) compared to control cells (untransduced; “UTD”) in precursor media 8 days. FIG. 2C shows a histogram depicting CAR expression in transduced cells (“CAR”) compared to control cells (untransduced; “UTD”) in precursor media 10 days. FIG. 2D shows a histogram depicting CAR expression in transduced cells (“CAR”) compared to control cells (untransduced; “UTD”) in precursor media 14 days. FIG. 2E shows a histogram depicting GFP expression in transduced cells (“GFP”) compared to control cells (untransduced; “UTD”) in precursor media 6 days. FIG. 2F shows a histogram depicting GFP expression in transduced cells (“GFP”) compared to control cells (untransduced; “UTD”) in precursor media 8 days. FIG. 2G shows a histogram depicting GFP expression in transduced cells (“GFP”) compared to control cells (untransduced; “UTD”) in precursor media 10 days. FIG. 2H shows a histogram depicting GFP expression in transduced cells (“GFP”) compared to control cells (untransduced; “UTD”) in precursor media 14 days. FIG. 21 shows a histogram depicting CAR expression in transduced cells (“CAR”) compared to control cells (untransduced; “UTD”) after 14 days in precursor media, followed by 8 days in Ml macrophage activation media. FIG. 2J shows a histogram depicting GFP expression in transduced cells (“GFP”) compared to control cells (untransduced; “UTD”) after 14 days in precursor media, followed by 8 days in Ml macrophage activation media. FIG. 2K shows a flow dot plot depicting CD 13 surface marker expression in the cell population of FIGS. 2D and 2H (i.e, maintained 14 days in precursor media). The number in the box indicates the percentage of the population that is CD13+. FIG. 2L shows a flow dot plot depicting CD 15 surface marker expression in the cell population of FIGS. 2D and 2H. The
number in the box indicates the percentage of the population that is CD15+. FIG. 2M shows a flow dot plot depicting CD1 lb+ surface marker expression in the cell population of FIGS. 2D and 2H. The number in the box indicates the percentage of the population that is CD1 lb+. FIG. 2N shows a flow dot plot depicting CD14 surface marker expression in the cell population of FIGS. 2D and 2H. The number in the box indicates the percentage of the population that is CD14+.
FIGS. 3A-3B show the efficacy of CAR transduction and differentiation of hematopoietic stem cells transduced with a CAR using the methods disclosed herein. Flow cytometry analysis was used to determine CAR and CD14 surface expression. FIG 3A is a flow plot depicting percentage of a cell population expressing a CAR (GFP+). FIG. 3B is a flow plot depicting percentage of the population of CAR-expressing cells from FIG. 3A that are CD14+.
FIGS. 4A-4C show three graphs demonstrating the expansion of hematopoietic stem cells (HSCs) in precursor media. HSCs were maintained in precursor media for up to 12 days, and cell number was analyzed over time. Increase in cell number and expansion of the cell population in each precursor media at the indicated day is depicted as cumulative fold-expansion relative to day 0.
FIG. 5 is a flow dot plot depicting CD14 and CD1 lb surface marker expression in a cell population of hemopoietic stem cells (HSCs) maintained in My el 4.1 precursor media for 7 days, followed by myeloid media for 9 days. The number depicted in each box represents the percentage of the cell population presenting the indicated marker(s).
FIG. 6 is a graph depicting CD14 surface marker expression in populations of hemopoietic stem cells (HSCs) maintained in three different precursor medias (My el 1,; My el 2; and My el 3) for 8 days, and then maintained in myeloid media for out tothree days. “%CD14+” represents the percentage of the cell population expressing CD14 in the indicated media at each day.
FIG. 7 shows chemoattractant-dependent (C5a) migratory capacity through human umbilical venular endothelial cells (HUVEC) endothelial monolayer of monocytes derived from HSCs (“HSC- Monocytes”; see “1” and “2”) compared to stimulated monocytes (“Stim. Monocytes”; see “3” and “4”). The number of migratory cells in each well were analyzed and are presented every 1 hour over 48 hours.
FIGS. 8A-8B depict phagocytic efficacy of monocytes generated from hemopoietic stem cells (HSCs) using the expansion and differentiation methods disclosed herein. FIG. 8A is a schematic depicting a synthetic bead uptake assay used to analyze phagocytic capacity of the tested cells. FIG. 8B is a graph showing uptake by monocytes during co-culture with beads over 6 hours. Individual beads phagocytosed by monocytes detected (“red object count / well) are shown at the indicated time. A negative control is presented where cells were treated with cytochalasin D (“UTD + CytoD”).
FIG. 9 is a graph showing the potent phagocytic and killing capacity of MO and Ml macrophages generated from hemopoietic stem cells (HSCs) obtained from three donors (“Donor 1”, “1”; “Donor 2”, “2”; and “Donor 3”, “3”). Cells were co-cultured with H1299 tumor cells following differentiation into MO or Ml polarized cells and monitored over 72 hours by live imaging. Tumor burden was normalized to a tumor cell only control (H1229). Reduction in tumor burden during coculture compared to the tumor only control was assessed over the hours as indicated for each cell type from each donor.
DETAILED DESCRIPTION
The present disclosure now will be described more fully hereinafter. The disclosure may be embodied in many different forms and should not be construed as limited to the aspects set forth herein; rather, these aspects are provided so that this disclosure will satisfy applicable legal requirements.
I. Definitions
Unless otherwise defined, all technical and scientific terms used herein have the same meaning as commonly understood by one of ordinary skill in the art to which this invention belongs. The terminology used in the description of the invention herein is for the purpose of describing particular embodiments only and is not intended to be limiting of the invention.
As used herein, “a,” “an,” or “the” can mean one or more than one. For example, “a” cell can mean a single cell or a multiplicity of cells.
As used herein, unless specifically indicated otherwise, the word “or” is used in the inclusive sense of “and/or” and not the exclusive sense of “either/or.”
The term “about” or “approximately” usually means within 5%, or more preferably within 1%, of a given value or range.
The terms “comprises”, “comprising”, “includes”, “including”, “having” and their conjugates mean “including but not limited to”.
Various embodiments of this disclosure may be presented in a range format. It should be noted that whenever a value or range of values of a parameter are recited, it is intended that values and ranges intermediate to the recited values are also part of this disclosure. It should be understood that the description in range format is merely for convenience and brevity and should not be construed as an inflexible limitation on the scope of the disclosure. Accordingly, the description of a range should be considered to have specifically disclosed all the possible subranges as well as individual numerical values within that range. For example, description of a range such as from 1-10 should be considered
to have specifically disclosed subranges such as from 1 to 3, from 1 to 4, from 1 to 5, from 1 to 6, from 1 to 7, from 1 to 8, from 1 to 9, from 2 to 4, from 2 to 6, from 2 to 8, from 2 to 10, from 3 to 6, etc., as well as individual numbers within that range, for example, 1, 2, 3, 4, 5, 6, 7, 8, 9 and 10. This applies regardless of the breadth of the range.
Whenever a numerical range is indicated herein, it is meant to include any cited numeral (fractional or integral) within the indicated range. The phrases “ranging/ranges between” a first indicate number and a second indicate number and “ranging/ranges from” a first indicate number “to” a second indicate number are used herein interchangeably and are meant to include the first and second indicated numbers and all the fractional and integral numerals there between. The recitation of a numerical range for a variable is intended to convey that the present disclosure may be practiced with the variable equal to any of the values within that range. Thus, for a variable which is inherently discrete, the variable can be equal to any integer value within the numerical range, including the endpoints of the range. Similarly, for a variable which is inherently continuous, the variable can be equal to any real value within the numerical range, including the end-points of the range. As an example, and without limitation, a variable which is described as having values between 0 and 2 can take the values 0, 1 or 2 if the variable is inherently discrete, and can take the values 0.0, 0.1, 0.01, 0.001, or any other real values ^0 and =2 if the variable is inherently continuous.
As used herein, the term “gene” or “coding sequence”, herein used interchangeably, refers to a functional nucleic acid unit encoding a protein, polypeptide, or peptide. As will be understood by those in the art, this functional term includes genomic sequences, cDNA sequences, and smaller engineered gene segments that express, or may be adapted to express proteins, polypeptides, domains, peptides, fusion proteins, and mutants.
As used herein, “nucleic acid,” “nucleotide sequence,” and “polynucleotide" are used interchangeably and encompass both RNA and DNA, including cDNA, genomic DNA, mRNA, synthetic (e.g, chemically synthesized) DNA or RNA and chimeras of RNA and DNA. The term polynucleotide, nucleotide sequence, or nucleic acid refers to a chain of nucleotides without regard to length of the chain. The term "fragment" will be understood to mean a nucleotide sequence of reduced length relative to a reference nucleic acid or nucleotide sequence and comprising, consisting essentially of and/or consisting of a nucleotide sequence of contiguous nucleotides identical to the reference nucleic acid or nucleotide sequence. Such a nucleic acid fragment according to the invention may be, where appropriate, included in a larger polynucleotide of which it is a constituent. In some embodiments, such fragments can comprise, consist essentially or and/or consist of, oligonucleotides having a length of at least about 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, or 25 consecutive nucleotides of a nucleic acid or nucleotide sequence according to the invention.
As used herein, a “mutation” is any change in a nucleic acid sequence. Nonlimiting examples comprise insertions, deletions, duplications, substitutions, inversions, and translocations of any nucleic acid sequence, regardless of how the mutation is brought about and regardless of how or whether the mutation alters the functions or interactions of the nucleic acid. For example and without limitation, a mutation may produce altered enzymatic activity of a ribozyme, altered base pairing between nucleic acids (e.g. RNA interference interactions, DNA-RNA binding, etc.), altered mRNA folding stability, and/or how a nucleic acid interacts with polypeptides (e.g. DNA-transcription factor interactions, RNA-ribosome interactions, gRNA-endonuclease reactions, etc.). A mutation might result in the production of proteins with altered amino acid sequences (e.g. missense mutations, nonsense mutations, frameshift mutations, etc.) and/or the production of proteins with the same amino acid sequence (e.g. silent mutations). Certain synonymous mutations may create no observed change in a cell while others that encode for an identical protein sequence nevertheless result in an altered cell phenotype (e.g. due to codon usage bias, altered secondary protein structures, etc.). Mutations may occur within coding regions (e.g., open reading frames) or outside of coding regions (e.g., within promoters, terminators, untranslated elements, or enhancers), and may affect, for example and without limitation, gene expression levels, gene expression profiles, protein sequences, and/or sequences encoding RNA elements such as tRNAs, ribozymes, ribosome components, and microRNAs.
As used herein, the term “recombinant DNA construct,” “recombinant construct,” “expression cassette,” “expression construct,” “chimeric construct,” “construct,” and “recombinant DNA fragment” are used interchangeably herein and are single or double-stranded polynucleotides. A recombinant construct comprises an artificial combination of nucleic acid fragments, including, without limitation, regulatory and coding sequences that are not found together in nature. For example, a recombinant DNA construct may comprise regulatory sequences and coding sequences that are derived from different sources, or regulatory sequences and coding sequences derived from the same source and arranged in a manner different than that found in nature. Such a construct may be used by itself or may be used in conjunction with a vector. Similarly, the terms “engineered” or “recombinant” in reference to a phagocyte, gene, nucleic acid and/or protein as used herein, refer to a phagocyte, gene, nucleic acid and/or protein that has been altered through human intervention. Accordingly, the term “naturally occurring” as used herein in reference to a phagocyte, gene, nucleic acid and/or protein as used herein, refer to a phagocyte, gene, nucleic acid and/or protein existing in nature and without any human intervention. Exemplary human interventions comprise transfection with a heterologous polynucleotide, molecular cloning resulting in a deletion, insertion, modification and/or rearrangement with respect to a naturally occurring sequence such as a naturally occurring sequence in a phagocyte, gene, nucleic acid and/or protein herein described.
As used herein, the term “expression” or “expressing” refers to the transcription and/or translation of a particular nucleic acid sequence driven by a promoter. An expression construct or expression vector can permit transcription of a particular nucleic acid sequence in a cell (e.g., a phagocytic cell). An expression cassette may be part of a plasmid, viral genome, or nucleic acid fragment. An expression cassette typically comprises at least three components: a promoter sequence, an open reading frame encoding gene(s) of interest, and a 3' untranslated region that, in eukaryotes, usually contains a polyadenylation site. An expression cassette can be formed by manipulable fragment of DNA carrying and capable of expressing, one or more genes of interest optionally located between one or more sets of restriction sites. Expression cassettes typically comprise further regulatory sequences additional to the promoter to regulate the expression of the gene or genes within the open reading frame herein also indicated as a coding region of the expression cassette. "Operably linked" is intended to mean a functional linkage between two or more elements. For example, an operable linkage between a promoter of the present invention and a heterologous nucleotide is a functional link that allows for expression of the heterologous nucleic acid molecule. Operably linked elements may be contiguous or non-contiguous. When used to refer to the joining of two protein coding regions, by operably linked is intended that the coding regions are in the same reading frame. The cassette may additionally contain at least one additional gene to be co-transformed into a cell. Alternatively, the additional gene(s) can be provided on multiple expression cassettes or DNA constructs. The expression cassette may additionally contain selectable marker genes. Other elements that may be present in an expression cassette include those that enhance transcription (e.g., enhancers) and terminate transcription (e.g., terminators), as well as those that confer certain binding affinity or antigenicity to the recombinant protein produced from the expression cassette.
As used herein, "contact", contacting", "contacted," refer to placing the components of a desired interaction together under conditions suitable for enabling and carrying out the interaction. For instance, a precursor cell and/or a population comprising a plurality of precursor cells can be contacted with a precursor media. Any means of contacting a cell or cell population with a media can be used. A skilled artisan would understand that a cell or cell population can be inoculated, suspended, or maintained in a culture media, such as a liquid culture media in order to contact said cell with said media. In some instances, said cell or cell population can be present within a solution, such as a buffered and/or sterile solution, that can be mixed into a culture media such that the cell(s) come into contact with the precursor media. For instance, the precursor cells disclosed herein can be contacted with a nucleic acid molecule. The nucleic acid molecule can be purified from a biological sample, cell lysate, or culture medium, produced via in vitro transcription, or chemically synthesized. The cell can be purified from a biological sample, such as a G-CSF mobilized leukopack from a healthy donor, or a
frozen vial. The cell and/or nucleic acid molecule can be brought into contact in any solution (e.g., buffered saline solution) or culture medium to allow for introduction of the nucleic acid molecule into the cell. Methods of introducing nucleic acid molecules into cells are readily known in the art. Any method for introducing a nucleic acid molecule is envisaged within the scope of the present application. The term “polypeptide” as used herein indicates an organic linear, circular, or branched polymer composed of two or more amino acid monomers and/or analogs thereof. The term “polypeptide” includes amino acid polymers of any length including full-length proteins and peptides, as well as analogs and fragments thereof. A polypeptide of three or more amino acids is also called a protein oligomer, peptide, or oligopeptide. In particular, the terms “peptide” and “oligopeptide” usually indicate a polypeptide with less than 100 amino acid monomers. In particular, in a protein, the polypeptide provides the primary structure of the protein, wherein the term “primary structure” of a protein refers to the sequence of amino acids in the polypeptide chain covalently linked to form the polypeptide polymer. A protein “sequence” indicates the order of the amino acids that form the primary structure. Covalent bonds between amino acids within the primary structure can include peptide bonds or disulfide bonds, and additional bonds identifiable by a skilled person. Polypeptides in the sense of the present disclosure are usually composed of a linear chain of alpha-amino acid residues covalently linked by peptide bond or a synthetic covalent linkage. The two ends of the linear polypeptide chain encompassing the terminal residues and the adjacent segment are referred to as the carboxyl terminus (C-terminus) and the amino terminus (N-terminus) based on the nature of the free group on each extremity. Unless otherwise indicated, counting of residues in a polypeptide is performed from the N-terminal end (NHz-group), which is the end where the amino group is not involved in a peptide bond to the C-terminal end (-COOH group) which is the end where a COOH group is not involved in a peptide bond. Proteins and polypeptides can be identified by x-ray crystallography, direct sequencing, immunoprecipitation, and a variety of other methods as understood by a person skilled in the art. Proteins can be provided in vitro or in vivo by several methods identifiable by a skilled person. In some instances where the proteins are synthetic proteins in at least a portion of the polymer two or more amino acid monomers and/or analogs thereof are joined through chemically-mediated condensation of an organic acid (-COOH) and an amine (-NH2) to form an amide bond or a “peptide” bond.
"Amino acid" as used herein refers to a compound having a free carboxyl group and a free unsubstituted amino group on the a carbon, which may be joined by peptide bonds to form a peptide active agent as described herein. Amino acids may be standard or non-standard, natural or synthetic, with examples (and their abbreviations) including but not limited to: Asp=D= Aspartic Acid; Ala=A=Alanine; Arg=R=Arginine; Asn=N=Asparagine; Cys=C=Cysteine; Gly=G=Glycine;
Glu=E=Glutamic Acid; Gln=Q=Glutamine; His=H=Histidine; Ile=I=Isoleucine; Leu=L=Leucine; Lys=K=Lysine; Met=M=Methionine; Phe=F=Phenylalanine; Pro=P=Proline; Ser=S=Serine Thr=T=Threonine; Trp=W=Tryptophan; Tyr=Y=Tyrosine; Val=V= Valine; Orn=Ornithine; Nal=2- napthylalanine; Nva=Norvaline; Nle=Norleucine; Thi=2 -thienylalanine; Pcp=4-chlorophenylalanine; Bth=3-benzothienyalanine; Bip=4,4'-biphenylalanine; Tic=tetrahydroisoquinoline-3-carboxylic acid; Aib=aminoisobutyric acid; Anb=a-aminonormalbutyric acid; Dip=2, 2-diphenylalanine; Thz=4- Thiazolylalanine. A "basic amino acid" refers to any amino acid that is positively charged at a pH of 6.0, including but not limited to R, K, and H. An "aromatic amino acid" refers to any amino acid that has an aromatic group in the side-chain coupled to the alpha carbon, including but not limited to F, Y, W, and H.
As used herein, “function” of a gene, a peptide, a protein, or a molecule refers to activity of a gene, a peptide, a protein, or a molecule.
“Introducing,” “introduce,” and “introduced” (and grammatical variations thereof) in the context of a polynucleotide and/or polypeptide of interest means presenting a nucleotide sequence of interest (e.g., polynucleotide, a nucleic acid construct, and/or a guide nucleic acid) and/or polypeptide of interest to a host organism or cell of said organism (e.g., a mammalian cell) in such a manner that the nucleotide sequence and/or polypeptide gains access to the interior of a cell. In some embodiments, “introducing” includes inserting a nucleic acid molecule (e.g., a recombinant DNA construct) into a cell, by means of transformation, transfection, or transduction. The nucleic acid molecule may be incorporated into the genome of the cell (e.g., nuclear chromosome or mitochondrial chromosome), converted into an autonomous replicon, or transiently expressed (e.g., transfected mRNA).
As used therein, a “subject” that may be treated by methods of the present disclosure include both human subjects for medical and/or therapeutic purposes and animal subjects for veterinary and drug screening and development purposes. Other suitable animal subjects are, in general, mammalian subjects such as primates, bovines, ovines, caprines, porcines, equines, felines, canines, lagomorphs, rodents ( e.g ., rats and mice), etc. Human subjects are the most preferred. Human subjects include fetal, neonatal, infant, juvenile, adult and geriatric subjects.
The term "anti -turn or effect" as used herein, refers to a biological effect which can be manifested by a decrease in tumor volume, a decrease in the number of tumor cells, a decrease in the proliferation rate, a decrease in the number of metastases, an increase in life expectancy, and/or amelioration of various physiological symptoms associated with the cancerous condition. An "antitumor effect" can also be manifested by the ability of the peptides, polynucleotides, cells and antibodies of the invention to delay the occurrence of tumor in the first place.
As used herein, the term "autologous" is meant to refer to any material derived from the same individual to whom it is later to be re-introduced.
"Allogeneic" refers to a graft derived from a different animal of the same species.
"Xenogeneic" refers to a graft derived from an animal of a different species.
The term "antibody" refers to full-length immunoglobulins as well as to fragments thereof. Such full-length immunoglobulins may be monoclonal, polyclonal, chimeric, humanized, veneered or human antibodies.
The term "antibody fragments" comprises portions of a full-length immunoglobulin retaining the targeting specificity of said immunoglobulin. Many but not all antibody fragments lack at least partially the constant region (Fc region) of the full-length immunoglobulin. In some embodiments, antibody fragments are produced by digestion of the full-length immunoglobulin. An antibody fragment may also be a synthetic or recombinant construct comprising parts of the immunoglobulin or immunoglobulin chains (see e.g. Holliger, P. and Hudson, J. Engineered antibody fragments and the rise of single domains. Nature Biotechnology 2005, vol. 23, no. 9, p. 1126-1136). Examples of antibody fragments include, without being limited to, include scFv, Fab, Fv, Fab', F(ab')2 fragments, dAb, VHH, nanobodies, V(NAR) or minimal recognition units. “Single chain variable fragments” or “single chain antibodies” or “scFv” are one type of antibody fragment. scFv are fusion proteins comprising the variable heavy (VH) and variable light (VL) of immunoglobulins connected by a linker. They thus lack the constant Fc region present in full-length immunoglobulins, but retain the specificity of the original immunoglobulin.
"Humanized" forms of non-human (e.g., murine) antibodies are chimeric immunoglobulins, immunoglobulin chains or fragments thereof (such as Fv, Fab, Fab1, F(ab')2 or other antigen-binding subsequences of antibodies) which contain minimal sequence derived from non-human immunoglobulin. For the most part, humanized antibodies are human immunoglobulins (recipient antibody) in which residues from a complementary-determining region (CDR) of the recipient are replaced by residues from a CDR of a non-human species (donor antibody) such as mouse, rat or rabbit having the desired specificity, affinity, and capacity. In some instances, Fv framework region (FR) residues of the human immunoglobulin are replaced by corresponding non-human residues.
Furthermore, humanized antibodies can comprise residues which are found neither in the recipient antibody nor in the imported CDR or framework sequences. These modifications are made to further refine and optimize antibody performance. In general, the humanized antibody will comprise substantially all of at least one, and typically two, variable domains, in which all or substantially all of the CDR regions correspond to those of a non-human immunoglobulin and all or substantially all of the FR regions are those of a human immunoglobulin sequence. The humanized antibody optimally
also will comprise at least a portion of an immunoglobulin constant region (Fc), typically that of a human immunoglobulin. For further details, see Jones et al. (1986) Nature 321 :522-525; Reichmann et al. (1988) Nature 332:323-329; Presta (1992) Curr. Op. Struct. Biol. 2:593-596.
"Fully human" refers to an immunoglobulin, such as an antibody, where the whole molecule is of human origin or consists of an amino acid sequence identical to a human form of the antibody.
The numbering system to identify amino acid residue positions in the VH and VL of an antibody can follow a system known to one of skill in the art, including Kabat (Wu and Kabat (1970) J Exp Med. 132(2):211-50; Borden and Kabat (1987) PNAS, 84:2440-2443; Kabat et al. U.S. Department of Health and Human Services, 1991), Chothia (Chothia and Lesk (1987) J Mol. Biol., 196(4): 901-917; Chothia et al. (1989) Nature 342:877-883), and the "AHo" system described by Honegger & Pluckthun (2001) Journal of Molecular Biology 309:657-670.
The term "antigen-binding portion" or "antigen-binding fragment" of an antibody (or simply "antibody portion" or "antibody fragment"), as used herein, refers to one or more fragments, portions or domains of an antibody that retain the ability to specifically bind to an antigen. It has been shown that fragments of a full-length antibody can perform the antigen binding function of an antibody. Examples of binding fragments encompassed within the term "antigen-binding portion" of an antibody include (i) an Fab fragment, a monovalent fragment consisting of the VL, VH, CL1 and CHI domains; (ii) an F(ab')2 fragment, a bivalent fragment comprising two F(ab)' fragments linked by a disulfide bridge at the hinge region; (iii) an Fd fragment consisting of the VH and CHI domains; (iv) an Fv fragment consisting of the VL and VH domains of a single arm of an antibody; (v) a dAb fragment (Ward et al. (1989) Nature 241 :544-546), which consists of a VH domain; and (vi) an isolated complementary determining region (CDR). Furthermore, although the two domains of the Fv fragment, VL and VH, are coded for by separate genes, they can be joined, using recombinant methods, by a synthetic linker that enables them to be made as a single contiguous chain in which the VL and VH regions pair to form monovalent molecules (known as single chain Fv (scFv); see e.g., Bird et al. (1988) Science 242:423-426; and Huston et al. (1988) Proc. Natl. Acad. Sci. USA 85:5879- 5883). Such single chain antibodies are also intended to be encompassed within the term "antigenbinding portion" of an antibody. Other forms of single chain antibodies, such as diabodies, are also encompassed (see e.g., Holliger et al. (1993) Proc. Natl. Acad Sci. USA 90:6444-6448).
The term "epitope" refers to an antigenic determinant that interacts with a specific antigen binding site in the variable region of an antibody molecule known as a paratope. A single antigen may have more than one epitope. Epitopes may be either conformational or linear. A conformational epitope is produced by spatially juxtaposed amino acids from different segments of one (or more) linear polypeptide chain(s). A linear epitope is an epitope produced by adjacent amino acid residues in
a polypeptide chain. In certain embodiments, an epitope may include other moieties, such as saccharides, phosphoryl groups, or sulfonyl groups on the antigen.
An "antibody heavy chain," as used herein, refers to the larger of the two types of polypeptide chains present in all antibody molecules in their naturally occurring conformations.
An "antibody light chain," as used herein, refers to the smaller of the two types of polypeptide chains present in all antibody molecules in their naturally occurring conformations, K and 1 light chains refer to the two major antibody light chain isotypes.
The term "antigen" or "Ag" as used herein is defined as a molecule that provokes an immune response. This immune response may involve either antibody production, or the activation of specific immunologically-competent cells, or both. The skilled artisan will understand that any macromolecule, including virtually all proteins or peptides, can serve as an antigen. Furthermore, antigens can be derived from recombinant or genomic DNA. A skilled artisan will understand that any DNA, which comprises a nucleotide sequence or a partial nucleotide sequence encoding a protein that elicits an immune response therefore encodes an "antigen" as that term is used herein. Furthermore, one skilled in the art will understand that an antigen need not be encoded solely by a full length nucleotide sequence of a gene. It is readily apparent that the present invention includes, but is not limited to, the use of partial nucleotide sequences of more than one gene and that these nucleotide sequences are arranged in various combinations to elicit the desired immune response. Moreover, a skilled artisan will understand that an antigen need not be encoded by a "gene" at all. It is readily apparent that an antigen can be synthesized or can be derived from a biological sample. Such a biological sample can include, but is not limited to a tissue sample, a tumor sample, a cell or a biological fluid.
By the term "specifically binds," as used herein with respect to an antibody or antigen binding domain, is meant an antibody or antigen binding domain which recognizes a specific antigen, but does not substantially recognize or bind other molecules in a sample. For example, an antibody or antigen binding domain that specifically binds to an antigen from one species may also bind to that antigen from one or more species. But, such cross-species reactivity does not itself alter the classification of an antibody or antigen binding domain as specific. In another example, an antibody or antigen binding domain that specifically binds to an antigen may also bind to different allelic forms of the antigen. However, such cross reactivity does not itself alter the classification of an antibody or antigen binding domain as specific. In some instances, the terms "specific binding" or "specifically binding," can be used in reference to the interaction of an antibody, antigen binding domain, a protein, or a peptide with a second chemical species, to mean that the interaction is dependent upon the presence of a particular structure (e.g., an antigenic determinant or epitope) on the chemical species; for example, an antibody or antigen binding domain recognizes and binds to a specific protein structure
rather than to proteins generally. If an antibody is specific for epitope "A", the presence of a molecule containing epitope A (or free, unlabeled A), in a reaction containing labeled "A" and the antibody, will reduce the amount of labeled A bound to the antibody.
An "immune response" refers to the reaction of a subject to the presence of an antigen, which may include at least one of the following: making antibodies, developing immunity, developing hypersensitivity to the antigen, and developing tolerance. The term “enhance an immune response” as used herein implies that the reaction of a subject to the presence of an antigen is increased and/or amplified in the presence of a CAR-modified phagocytic cell of the disclosure as compared to the reaction of a subject to the presence of an antigen in the absence of a CAR-modified phagocytic cell of the disclosure.
The terms "treat", "treating", or "treatment of indicates that the severity of the subject's condition is reduced or at least partially improved or modified and that some alleviation, mitigation or decrease in at least one clinical symptom is achieved.
The term "therapeutic" as used herein means a treatment and/or prophylaxis. A therapeutic effect is obtained by suppression, remission, or eradication of a disease state.
An "effective" amount as used herein is an amount that provides a desired effect.
A "therapeutically effective" amount as used herein is an amount that provides some improvement or benefit to the subject. Alternatively stated, a "therapeutically effective" amount is an amount that will provide some alleviation, mitigation, or decrease in at least one clinical symptom in the subject. Those skilled in the art will appreciate that the therapeutic effects need not be complete or curative, as long as some benefit is provided to the subject.
By the term "modulating," as used herein, is meant mediating a detectable increase or decrease in the level of a response in a subject compared with the level of a response in the subject in the absence of a treatment or compound, and/or compared with the level of a response in an otherwise identical but untreated subject. The term encompasses perturbing and/or affecting a native signal or response thereby mediating a beneficial therapeutic response in a subject, preferably, a human.
A "target site" or "target sequence" refers to a genomic nucleic acid sequence that defines a portion of a nucleic acid to which a binding molecule may specifically bind under conditions sufficient for binding to occur.
By "target" is meant a cell, organ, or site within the body that is in need of treatment.
The term "detectable moiety" as used herein includes any suitable detectable group, such as radiolabels (e.g. 35S, 125I, 131I, etc.), enzyme labels (e.g, horseradish peroxidase, alkaline phosphatase, etc.), fluorescence labels (e.g., fluorescein, green fluorescent protein, etc.), etc., as are well known in the art and used in accordance with known techniques.
The term "agent," or "biological agent" or "therapeutic agent" as used herein, refers to a molecule that may be expressed, released, secreted or delivered to a target by the modified cell described herein. The agent includes, but is not limited to, a nucleic acid, an antibiotic, an antiinflammatory agent, an antibody or antibody fragments thereof, a growth factor, a cytokine, an enzyme, a protein, a peptide, a fusion protein, a synthetic molecule, an organic molecule (e.g., a small molecule), a carbohydrate or the like, a lipid, a hormone, a microsome, a derivative or a variation thereof, and any combination thereof. The agent may bind any cell moiety, such as a receptor, an antigenic determinant, or other binding site present on a target or target cell. The agent may diffuse or be transported into the cell, where it may act intracellularly.
“Mobilization” refers to the process of forced emigration of hematopoietic stem cells (HSPCs) from the bone marrow (BM) into the peripheral blood. Mobilization of HSPCs can occur in response to a wide variety of stimuli including strenuous physical exercise, myelosuppressive chemotherapy, polyanions, chemokines, and hematopoietic growth factors.
The term(s) "expand", “expanding”, and “expansion” as used herein refers to increasing in number, as in an increase in the number of stem cells or cells derived therefrom. In some embodiments, the stem cells, or cells derived therefrom, that are expanded ex vivo increase in number relative to the number originally present in the culture (e.g., in precursor media). In some embodiments, the stem phagocytic cells, or cells derived therefrom that are expanded ex vivo increase in number relative to other cell types in the culture. The term "ex vivo" refers to cells that have been removed from a living organism, (e.g., a human) and propagated outside the organism (e.g., in a culture dish, test tube, or bioreactor). The term “expand” can refer to a cell, or a population comprising a plurality of cells. When used to refer to a cell population, the term “expand” refers to an increase in the total number of cells within the population. Expansion of a cell population can occur when at least one individual cell within the population undergoes expansion. In some instances, more than one cell within the population undergoes expansion. In some instances, all cells within a population can expand. In some instances, a specific subset of cells within a population can expand, wherein cells not within said subset do not expand. Expansion can be temporal. For instance, a cell, a population, or a subset within a population can expand for a period of time and then cease to expand. In some instances, one subset of cells within a population can undergo expansion and then cease, after which a second subset can undergo expansion. Disclosed herein are mixed populations of cells. In some instances, any of the cells within the population can undergo expansion. The mixed populations of cells disclosed herein can undergo expansion. In some embodiments, the mixed population of cells disclosed herein comprises at least one precursor cell and/or at least one myeloid-derived cell. In some embodiments, the mixed population of cells disclosed herein comprises at least one precursor CAR cell
and/or at least one myeloid-derived CAR cell. In some embodiments, the mixed population of cells disclosed herein comprises a plurality of precursor CAR cells and/or a plurality of myeloid-derived CAR cells. When the mixed populations disclosed herein undergo expansion, any subset or plurality of cells within the population can undergo expansion. In some instances, all cells within the population undergo expansion. In some instances, the precursor CAR cells undergo expansion. In some instances, the myeloid-derived CAR cells undergo expansion. When a cell, or population thereof, has expanded, the number of cells within the population has increased. During expansion, or when expanding, the number of cells within the population that is expanding is increasing. Cell, or cell population expansion, can occur at different rates. A skilled artisan would understand how to determine the expansion rate by calculating the increase in cell number over a defined time. That is, the rate at which the number of cells increases can differ between different medium (e.g., precursor mediums). The precursor mediums disclosed herein are particularly effective at enhancing the expansion of stem cells over a defined period of time compared to expansion of stem cells in a medium that is not said precursor medium. Without wishing to be bound by theory, the specific components within the disclosed precursor mediums are thought to enhance stem cell expansion. In some instances, the precursor medium disclosed herein enhance stem cell expansion greater than a medium that does not comprise the precursor cytokine mixture of the precursor mediums disclosed herein. In some instances, a population of cells that is undergoing expansion is also undergoing differentiation. In such instances, a first subset of cells within the population can be expanding, and a second subset of cells within a population can be differentiating.
The terms “differentiate”, “differentiating”, and “differentiation”, as used herein refer to a process by which young, immature (unspecialized) cells (e.g, stem cells) take on individual characteristics and reach their mature (specialized) form (e.g., myeloid cells) and function. During this process, cells can lose their developmental potential and gain specialized functions and phenotypes. The specialized cell type can be characterized by its physiological function and its role as part of a tissue or organ. The differentiation of a cell is typically associated with a change in at least one cell surface marker. In some instances, differentiation of a cell is associated with a change in multiple cell surface markers. For instances, after undergoing differentiation, an immature cell, such as a stem cell, can either gain or lose, or both, specific cell surface markers. The presence and or absence of individual cell surface markers can be used to categorized cells and/or to identifying a cell that has undergone differentiation. As differentiation is associated with a change in function and role of a cell, it is understood that the cell type and/or cell name will change. For instance, a stem cell can differentiate into a myeloid cell. In some instances, the hematopoietic stem cells (HSCs) disclosed herein undergo differentiation. In such instances, the HSCs can differentiation into a cell within the
myeloid lineage (i.e., myeloid-lineage cell). When a stem cell differentiates into a myeloid-lineage cell, the cell may acquire specific cell surface markers associated with the myeloid-lineage. The cell surface markers that are expressed by a differentiated cell with be dependent upon which cell it differentiations into. The precursor cells disclosed herein can differentiate into a myeloid-lineage or myeloid-derived cell. In some instances, cell differentiation is associated with an increase in the amount or number of surface markers on at least one cell (e.g., a precursor cell), wherein said surface markers are myeloid- associated markers. A “myeloid-associated marker” can be any surface marker that is known and available in the art for identifying and/or isolating a cell within the myeloid lineage. A person having skill in the art would understand how to determine if a cell was in the myeloid lineage and to identify a surface marker specific for said cell. Exemplary, non-limiting, myeloid-associated markers can include: CDl lb, CD14, CD13, and CD15. In some instances, differentiation of a precursor stem cell is associated with the increase in the presence of CD1 lb, CD14, CD13, and CD15 on the cell surface. In some instances, differentiation of a precursor stem cell is associated with the increase in the levels of CDl lb, CD14, CD13, and CD15 on the cell surface. In some instances, differentiation of a precursor stem cell is associated with the decrease in the presence of CD34 on the cell surface. In some instances, differentiation of a precursor stem cell is associated with the decrease in the level of CD34 on the cell surface. As there are multiple myeloid-associated markers that are presence on different cell types within the myeloid lineage, the levels of more than one myeloid-associated marker can increase on the surface of a cell that is differentiating into a myeloid-lineage cell. In some instances, the hematopoietic stem cells (HSCs) disclosed herein undergo differentiation. In such instances, the HSCs can differentiation into a cell within the myeloid lineage (i.e., myeloid-lineage cell). Differentiation of a cell population can occur when at least one individual cell within the population undergoes differentiation. In some instances, more than one cell within the population undergoes differentiation. In some instances, all cells within a population can differentiate. In some instances, a specific subset of cells within a population can differentiate, wherein cells not within said subset do not differentiate. Differentiation can be temporal. For instance, a cell, a population, or a subset within a population can undergo differentiation for a period of time and then cease to differentiate. In some instances, one subset of cells within a population can undergo differentiation and then cease, after which a second subset can undergo differentiation. Disclosed herein are mixed populations of cells. In some instances, any of the cells within the population can undergo differentiation. The mixed populations of cells disclosed herein can undergo differentiation. In some embodiments, the mixed population of cells disclosed herein comprises at least one precursor cell and/or at least one myeloid-derived cell. In some embodiments, the mixed population of cells that undergoes differentiation comprises at least one precursor cell and/or at least one myeloid-derived cell. In some embodiments, the mixed population of
cells that undergoes differentiation comprises at least one precursor CAR cell and/or at least one myeloid-derived CAR cell. In some embodiments, the mixed population of cells that undergoes differentiation comprises a plurality of precursor CAR cells and/or a plurality of myeloid-derived CAR cells. When the mixed populations disclosed herein undergo differentiation, any number of cells within the population, and any subset or plurality of cells within the population, can differentiate. In some instances, all cells, or most of the cells, within the population undergo differentiation. In such instances, a population can go from comprising mostly precursor cells to mostly myeloid-lineage cells. In some instances, the precursor CAR cells undergo differentiation. In some instances, the myeloid- derived CAR cells undergo differentiation. In some instances, when a population of cells has differentiated, the number of precursor cells within the population has decreased, while the number of differentiated cells within the population has increased. During differentiation, or when differentiating, the type of cells within the population that is differentiating can be changing. In some instances, a population of cells that is undergoing differentiation is also expanding. In such instances, a first subset of cells within the population can be expanding, and a second subset of cells within a population can be differentiating.
“Myeloid medium” or “myeloid media”, used interchangeably herein, refer to a culture medium that promotes mature myeloid lineage development of any of the cells disclosed herein. The myeloid medium disclosed herein comprise a myeloid cytokine mixture. Without wishing to be bound by theory, the specific myeloid cytokine mixture(s) within the myeloid medium is thought to promote myeloid lineage differentiation. In some instances, the myeloid medium disclosed herein enhance stem cell differentiation greater than a medium that is similar to the disclosed myeloid media but does not comprise the myeloid cytokine mixture.
“Myeloid lineage” refers to the group of cells that are derived from a common myeloid progenitor (CMP) in the bone marrow. The cells within the myeloid lineage, which include monocytes, granulocytes, erythrocytes, and platelets, serve as a primary component of the innate immune system. The cells within the myeloid lineage are referred to, for the purposes of the present disclosure, as “myeloid cells.” The common myeloid progenitor cell, and thus all myeloid cells, are derived from hematopoietic stem cells (HSCs), which are multipotent cells that can give rise to all blood lineages, including the myeloid lineage, of an adult organism in a process referred to a hematopoiesis. Without wishing to be bound by any theory, it is thought that cytokines and growth factors enhance the direction. Myeloid cells, as well as many aspects of their development from HSCs, and differentiation of cells within the myeloid lineage are readily known in the art (Weiskopf, Kipp et al. “Myeloid Cell Origins, Differentiation, and Clinical Implications.” Microbiology spectrum vol. 4,5 (2016): 10.1128/microbiolspec.MCHD-0031-2016, incorporated herein by reference in its entirety).
“Precursor media” or “precursor medium”, used interchangeably herein, refer to a culture medium that promotes expansion (i.e., an increase in the number of cells) of any of the cells disclosed herein. The precursor media disclosed herein comprises a precursor cytokine mixture. Without wishing to be bound by theory, the specific precursor cytokine mixture(s) present within the precursor media is thought to promote an increase in the number of at least one cell that is contacted with, or maintained in, the precursor media. In some instances, the precursor medium disclosed herein enhances stem cell expansion (e.g., expansion of a single stem cell, or a plurality of stem cells present within a cell population) greater than a medium that is similar to the disclosed precursor media but does not comprise the precursor cytokine mixture. The precursor media can increase the number of cells of any cell type disclosed herein.
A ’’precursor cytokine mixture” refers to a composition comprising at least one cytokine that is present within a precursor media disclosed herein. Any cytokine disclosed herein can be present within the precursor cytokine mixture at any concentration.
A ’’myeloid cytokine mixture” refers to a composition comprising at least one cytokine that is present within a myeloid media disclosed herein. Any cytokine disclosed herein can be present within the myeloid cytokine mixture at any concentration.
“Precursor” or “precursor cell”, used interchangeably herein, refers to any cell that can differentiate into another cell type. In preferred embodiments, the precursor cell is a stem cell. Any type of stem cell is envisaged as being used as the precursor cell, so long as it can be differentiated into a myeloid-lineage cell. In some embodiments, the precursor cell is a bone-marrow derived stem cell. In another preferred embodiment, the precursor cell is a hematopoietic stem cell (HSC). Suitable precursor cells can be identified and isolated by specific cell surface markers. In some embodiments, the precursor cell is a CD34+ cell. The precursor cell can be isolated from a subject, such as a mammalian subject by any suitable means available in the art. In such instances, the precursor cell may be frozen following isolation and prior to use in the disclosed methods. For example, the precursor cell may be a cryopreserved stem cell. When the precursor cell has been cryopreserved, it may be necessary to thaw the cell prior to use in the disclosed methods.
By the term "modified" as used herein, is meant a changed state or structure of a molecule or cell of the disclosure. Molecules may be modified in many ways, including chemically, structurally, and functionally. Cells may be modified through the introduction of nucleic acids.
As used herein with respect to a parameter, the term “decreased” or “decreasing” or “decrease” or “reduced” or “reducing” or “reduce” or “lower” or “loss” refers to a detectable (e.g., at least about 5%, 10%, 15%, 20%, 25%, 30%, 35%, 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 96%, 97%, 98%, 99%, or 100%) negative change in the parameter from a comparison control,
e.g., an established normal or reference level of the parameter, or an established standard control. Accordingly, the terms “decreased”, “reduced”, and the like encompass both a partial reduction and a complete reduction compared to a control.
As used herein with respect to a parameter, the term “increased” or “increasing” or “increase” refers to a detectable (e.g., at least about 5%, 10%, 15%, 20%, 25%, 30%, 35%, 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 96%, 97%, 98%, 99%, 100%, 120%, 150%, 200%, 300%, 400%, 500%, or more) positive change in the parameter from a comparison control, e.g., an established normal or reference level of the parameter, or an established standard control. Accordingly, the terms “increased”, “increase”, and the like encompass both a partial reduction and a significant increase compared to a control.
The wording “fold change” as used herein indicates a measure describing how much a quantity changes between an original and a subsequent measurement. In particular, fold change is defined as the ratio between two quantities. For example, for quantities A and B, the fold change of B with respect to A is B/A. For example, a change from 30 to 60 is defined as a fold-change of 2.
When reference is made to particular sequence listings, such reference is to be understood to also encompass sequences that substantially correspond to its complementary sequence as including minor sequence variations, resulting from, e.g., sequencing errors, cloning errors, or other alterations resulting in base substitution, base deletion or base addition, provided that the frequency of such variations is less than 1 in 50 nucleotides, alternatively, less than 1 in 100 nucleotides, alternatively, less than 1 in 200 nucleotides, alternatively, less than 1 in 500 nucleotides, alternatively, less than 1 in 1000 nucleotides, alternatively, less than 1 in 5,000 nucleotides, alternatively, less than 1 in 10,000 nucleotides.
The term “isolated” refers to at least partially separated from the natural environment e.g., from a cell. For example, a nucleic acid or a peptide naturally present in a living animal is not "isolated," but the same nucleic acid or peptide partially or completely separated from the coexisting materials of its natural state is "isolated." An isolated nucleic acid or protein can exist in substantially purified form, or can exist in a non-native environment such as, for example, a host cell.
As used herein, the terms “exogenous” or “heterologous” in reference to a nucleic acid sequence or amino acid sequence are intended to mean a sequence that is purely synthetic, that originates from a foreign species, or, if from the same species, is substantially modified from its native form in composition and/or genomic locus by deliberate human intervention. Thus, a heterologous nucleic acid sequence may not be naturally expressed within a cell or may have altered expression when compared to the corresponding wild type cell. For example, a heterologous polynucleotide encoding a CAR described herein can be a nucleic acid sequence that is not naturally present in a
phagocytic cell in which it is present. An exogenous polynucleotide may be introduced into the cell in a stable or transient manner, so as to produce a ribonucleic acid (RNA) molecule and/or a polypeptide molecule. It should be noted that the exogenous polynucleotide may comprise a nucleic acid sequence which is identical or partially homologous to an endogenous nucleic acid sequence of the cell.
As used herein, by “endogenous” in reference to a gene or nucleic acid sequence or protein is intended a gene or nucleic acid sequence or protein that is naturally comprised within or expressed by a cell. Endogenous genes can include genes that naturally occur in a cell (e.g., phagocytic cell), but that have been modified in the genome of the cell without insertion or replacement of a heterologous gene that is from another species or another location within the genome of the modified cell.
“Homolog” or “homologous sequence” may refer to both orthologous and paralogous sequences. Paralogous sequence relates to gene-duplications within the genome of a species. Orthologous sequence relates to homologous genes in different organisms due to ancestral relationship. Thus, orthologs are evolutionary counterparts derived from a single ancestral gene in the last common ancestor of given two species and therefore have great likelihood of having the same function.
In some embodiments, the term “homolog” as used herein, refers to functional homologs of genes. A functional homolog is a gene encoding a polypeptide that has sequence similarity to a polypeptide encoded by a reference gene, and the polypeptide encoded by the homolog carries out one or more of the biochemical or physiological function(s) of the polypeptide encoded by the reference gene. In general, it is preferred that functional homologs and/or polypeptides encoded by functional homologs share at least some degree of sequence identity with the reference gene or polypeptide encoded by the reference gene.
Homology (e.g., percent homology, sequence identity+sequence similarity) can be determined using any homology comparison software computing a pairwise sequence alignment. "Homologous" can refer to the subunit sequence identity between two polymeric molecules, e.g., between two nucleic acid molecules, such as, two DNA molecules or two RNA molecules, or between two polypeptide molecules. When a subunit position in both of the two molecules is occupied by the same monomeric subunit; e.g., if a position in each of two DNA molecules is occupied by adenine, then they are homologous at that position. The homology between two sequences is a direct function of the number of matching or homologous positions; e.g., if half (e.g., five positions in a polymer ten subunits in length) of the positions in two sequences are homologous, the two sequences are 50% homologous; if 90% of the positions (e.g., 9 of 10), are matched or homologous, the two sequences are 90% homologous. According to some embodiments, the term “homology” or “homologous” refers to identity of two or more nucleic acid sequences; or identity of two or more amino acid sequences; or the identity of an amino acid sequence to one or more nucleic acid sequence. According to some
embodiments, the homology is a global homology, e.g., a homology over the entire amino acid or nucleic acid sequences of the invention and not over portions thereof. The degree of homology or identity between two or more sequences can be determined using various known sequence comparison tools which are described in WO2014/102774. As applied to the nucleic acid or protein, "homologous" as used herein refers to a sequence that has about 50% sequence identity. More preferably, the homologous sequence has about 75% sequence identity, even more preferably, has at least about 80%, 85%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99% sequence identity.
As used herein, “sequence identity,” “identity,” “percent identity,” “percentage similarity,” “sequence similarity” and the like refer to a measure of the degree of similarity of two sequences based upon an alignment of the sequences that maximizes similarity between aligned amino acid residues or nucleotides, and which is a function of the number of identical or similar residues or nucleotides, the number of total residues or nucleotides, and the presence and length of gaps in the sequence alignment. A variety of algorithms and computer programs are available for determining sequence similarity using standard parameters. As used herein, sequence similarity is measured using the BLAST®p program for amino acid sequences and the BLAST®n program for nucleic acid sequences, both of which are available through the National Center for Biotechnology Information, and are described in, for example, Altschul et al. (1990), J. Mol. Biol. 215:403-410; Gish and States (1993), Nature Genet. 3:266-272; Madden et al. (1996), Meth. Enzymol.266: 131-141; Altschul et al. (1997), Nucleic Acids Res. 25:3389-3402); Zhang et al. (2000), J. Comput. Biol. 7(l-2):203-14. As used herein, percent similarity of two amino acid sequences is the score based upon the following parameters for the BLAST®p algorithm: word size=3; gap opening penalty=-l l; gap extension penalty=-l; and scoring matrix=BLOSUM62. As used herein, percent similarity of two nucleic acid sequences is the score based upon the following parameters for the BLAST®n algorithm: word size=l 1; gap opening penalty=-5; gap extension penalty=-2; match reward=l; and mismatch penalty=-3. When percentage of sequence identity is used in reference to proteins it is recognized that residue positions which are not identical often differ by conservative amino acid substitutions, where amino acid residues are substituted for other amino acid residues with similar chemical properties (e.g. charge or hydrophobicity) and therefore do not change the functional properties of the molecule. Where sequences differ in conservative substitutions, the percent sequence identity may be adjusted upwards to correct for the conservative nature of the substitution. Sequences which differ by such conservative substitutions are considered to have “sequence similarity” or “similarity”. Means for making this adjustment are well-known to those of skill in the art. Typically this involves scoring a conservative substitution as a partial rather than a full mismatch, thereby increasing the percentage sequence identity. Thus, for example, where an identical amino acid is given a score of 1 and a non-conservative
substitution is given a score of zero, a conservative substitution is given a score between zero and 1. The scoring of conservative substitutions is calculated, e.g., according to the algorithm of Henikoff S and Henikoff J G. (Proc Natl Acad Sci 89: 10915-9 (1992)). Identity (e.g., percent homology) can be determined using any homology comparison software, including for example, the Blast®N software of the National Center of Biotechnology Information (NCBI) such as by using default parameters.
As used herein, the term “method” refers to manners, means, techniques and procedures for accomplishing a given task including, but not limited to, those manners, means, techniques and procedures either known to, or readily developed from known manners, means, techniques and procedures by practitioners of the chemical, pharmacological, biological, biochemical and medical arts.
The patent and scientific literature referred to herein establishes knowledge that is available to those of skill in the art. The issued US patents, allowed applications, published foreign applications, and references, including GenBank database sequences, which are cited herein are hereby incorporated by reference to the same extent as if each was specifically and individually indicated to be incorporated by reference.
All publications, patent applications, patents, and other references mentioned herein are incorporated by reference herein in their entirety.
II. Overview
Disclosed herein are compositions and methods for efficiently and effectively generating myeloid-derived cells from stem cells. In some embodiments, the methods can be used to generate myeloid-derived CAR cells from stem cells. In preferred embodiments, the stem cells are isolated stem cells. For instance, the stem cells can be isolated from a subject. The subject can be a mammalian subject, such as a human. In some embodiments, the stem cell can be a hematopoietic stem cell and/or a CD34+ cell. The stem cell can be isolated from the bone-marrow of a subject. As such, the methods and compositions disclosed herein provide an effective and efficient process for generating a myeloid lineage cell (i.e., myeloid-derived cells), and populations comprising myeloid-derived cells, ex vivo. The methods of generating myeloid-derived cells ex vivo can be performed in a shorter amount of time, and can generate a cleaner (i.e., higher percentage of myeloid cells compared to non-myeloid cells) myeloid-derived cell population compared to standard methods known in the art. One aspect that contributes to the enhanced efficiency of the disclosed method (e.g., reduced time to generate a myeloid cell population) is the superior ability of the precursor media disclosed herein to increase the number of precursor cells to generate a cell population in a shorter amount of time compared to known expansion medias. Without wishing to be bound by theory, the precursor cytokine mixture present within the disclosed precursor media are thought to promote faster expansion of precursor cells
compared to similar expansion media not comprising the disclosed precursor cytokine mixtures. Another aspect contributing to the enhanced efficiency of the disclosed method to generate a myeloid- derived cell or myeloid-derived CAR cell population is the superior technical capability of the precursor media to promote expansion and differentiation of precursor cells, in particular hematopoietic stem cells and/or CD34+ cells. In particular, the precursor media disclosed herein can promote differentiation of precursor cells simultaneously. This combined process of expansion and differentiation in a precursor media significantly reduces the amount of time required to generate a myeloid-derived cell population and/or a myeloid-derived CAR cell population. The precursor media disclosed herein are also particularly effective at promoting efficient differentiation of precursor cells into myeloid-derived cells. For instance, the precursor media can generate a cell population comprising a plurality of myeloid-derived cells, wherein said population comprises a higher percentage of myeloid-derived cells that have been sufficiently differentiated compared to myeloid-derived cell populations that are generated from standard methods. For example, standard methods may generate a cell population with a low, or sub-optimal, percentage of myeloid-derived cells. Such instances, require additional differentiation steps of said population in an effort to increase the percentage of myeloid cells within the population, which requires additional resources and time. Standard methods for generating a myeloid-derived cell, or population comprising the same, generally require a step wise process, which involves expansion of a cell population as a first step, followed by differentiation of the cells within the population into the desired myeloid cell population. This step wise process involving separate steps and medias can be time-consuming and require a significant amount of manufacturing and financial resources.
It is envisaged that the methods disclosed herein can effectively generate a myeloid-derived CAR cell, and/or cell populations comprising a plurality of myeloid-derived CAR cells. In such embodiments of the disclosed methods, a CAR can be introduced into a stem cell prior to expansion and differentiation of said stem cell into a myeloid lineage cell using the compositions and methods disclosed herein. The methods of the present disclosure provide conditions that allow for enhanced stability of transduced genes, such as a CAR, when introduced into stem cells prior to the expansion and/or differentiation process, resulting in a myeloid-derived CAR cell population. The compositions and methods disclosed promote the expansion and differentiation of stem cells into myeloid-derived cells in a shorter time compared to known expansion and differentiation methods. For instance, the precursor media disclosed herein promote expansion of precursor cells, such as stem cells, into myeloid cells in a shorter time compared to known expansion mediums. As such, the precursor media of the present disclosure allow for the efficient and effective production of precursor cell populations in a shorter period of time compared to known media. The precursor media disclosed herein, in
addition to promoting expansion of precursor cell and precursor CAR cell populations, also promote the differentiation of said precursor cells and precursor CAR cells into myeloid lineage cells. Thus, the precursor media of the present disclosure are effective at promoting expansion and differentiation of precursor cells into myeloid-derived cells. The precursor media can also efficiently promote the differentiation of precursor CAR cells into myeloid-derived CAR cells. In some embodiments, the precursor media promotes the expansion and differentiation of a precursor cell and/or a precursor CAR cell at the same time. In such instances, a precursor cell, when incubated in a precursor media can increase in number to generate a cell population comprising a plurality of precursor cells. In such instances, the precursor cell can be a precursor CAR cell. At the same time, said precursor cells within the cell population can be undergoing differentiation in the precursor media, thereby generating a cell population, wherein said cell population is a mixed cell population comprising a plurality of precursor cells and/or a plurality of myeloid-derived cells. In some embodiments, the mixed cell population comprises a plurality of precursor cells and a plurality of myeloid-derived cells. The ability of the precursor media to both promote expansion of a precursor cell and/or a population comprising a plurality of precursor cells, as well as differentiation of said precursor cell and/or said population provides for enhanced methods over the art for generating a population of myeloid-derived cells, such as myeloid-derived CAR cells, in a shorter amount of time. Thus, the methods and compositions disclosed herein provide for more efficient and effective means of generating a population of myeloid- derived cells and/or myeloid-derived CAR cells, wherein said population comprises a higher percentage of myeloid cells within the population. In particular, the cell populations generated by the methods disclosed herein comprise a higher percentage of myeloid-derived cells compared to cell populations generated by known methods. The methods disclosed herein are capable of producing a cell population with a higher percentage of myeloid-derived cells (i.e., myeloid lineage cells) in a shorter amount of time, making the overall manufacturing process more efficient, and saving resources, compared to standard, known methods. Also disclosed herein are precursor cytokine mixtures that contribute to the superior technical effect demonstrated by the precursor media disclosed herein. The precursor media of the present disclosure are particularly effective, at least in part, due to the precursor cytokine mixture comprised within the precursor media. Without wishing to be bound by any theory, the presence of the disclosed precursor cytokine mixtures within the precursor media are thought to contribute to the enhance expansion and differentiation of the precursor cells.
In some situations, it may be beneficial to use a separate differentiation media following an expansion step. For instance, use of a differentiation media can increase the ratio of myeloid-derived cells to precursor cells within a cell population. Disclosed herein are methods involving use of a myeloid media to differentiate cells into a myeloid-derived cell. The myeloid media, like the precursor
media, are particularly effective at promoting the differentiation of a precursor cell into a myeloid lineage cell. In particular, the myeloid media disclosed herein can increase the ratio of myeloid-derived cells to precursor cells within a population in a short amount of time compared to standard differentiation medias, saving time and costs. In some instances, a mixed population comprising a plurality of precursor cells and/or a plurality of myeloid-derived cells can be incubated in a myeloid media to increase the ratio of myeloid-derived cells to precursor cells. In some instances, the mixed population was generated by incubating a precursor cell, or a population comprising a plurality of precursor cells in a precursor media, whereby said mixed population is subsequently incubated in a myeloid media to further promote an increase in myeloid-derived cell number within the mixed population. The methods disclosed herein, involving contacting a precursor cell, such as a precursor CAR cell, with a precursor media and, optionally, a myeloid media, provide for a shorter overall process for generating a myeloid-derived cell population compared to a similar method involving a media that is generally similar to the precursor media disclosed herein, but does not comprise the precursor cytokine mixture of the presently disclosed precursor media, and as such, is not capable of expanding and differentiation a precursor cell, or population comprising a precursor cell, in the same shortened time interval as the disclosed methods. The methods disclosed herein provide easier, stable engineering of myeloid-derived cells compared to standard, known methods. For instance, once introduced into a precursor cell, such as a stem cell, the transduced genes (e.g., a polynucleotide encoding a CAR) are stably integrated and thus maintained in said precursor CAR cell, and all cells derived therefrom. The methods and compositions disclosed herein provide a shorter process for generating a stable myeloid-derived CAR cell.
The methods and compositions disclosed herein can be used to generate a population of myeloid-derived cells. In some instances, the methods can generate a myeloid-derived CAR cell, or a population comprising a plurality of myeloid-derived CAR cells. The compositions and methods can generate a cell population comprising a high percentage of myeloid-derived cells. In some instances, the methods and compositions can be used to generate a cell population wherein at least 70%, at least 71%, at least 72%, at least 73%, at least 74%, at least 75%, at least 76%, at least 77%, at least 78%, at least 79%, at least 80%, at least 81%, at least 82%, at least 83%, or greater than 84% at least 85%, at least 86%, at least 87%, at least 88%, at least 89%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or greater than 99% of the cells within the population are positive for a myeloid cell surface marker (e.g., CD14+, CD13+, CD15+, CD1 lb+). In some instances, the methods and compositions can be used to generate a cell population wherein at least 70%, at least 71%, at least 72%, at least 73%, at least 74%, at least 75%, at least 76%, at least 77%, at least 78%, at least 79%, at least 80%, at least 81%, at least 82%, at
least 83%, or greater than 84% at least 85%, at least 86%, at least 87%, at least 88%, at least 89%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or greater than 99% of the cells within the population are positive for more than one myeloid cell surface marker (e.g., CD14+, CD13+, CD15+, CD1 lb+). In some instances, the methods and compositions can be used to generate a cell population wherein at least 70%, at least 71%, at least 72%, at least 73%, at least 74%, at least 75%, at least 76%, at least 77%, at least 78%, at least 79%, at least 80%, at least 81%, at least 82%, at least 83%, or greater than 84% at least 85%, at least 86%, at least 87%, at least 88%, at least 89%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or greater than 99% of the cells within the population are positive for two myeloid cell surface marker (e.g., CD14+, CD13+, CD15+, CD1 lb+). In some instances, the methods and compositions can be used to generate a cell population wherein at least 70%, at least 71%, at least 72%, at least 73%, at least 74%, at least 75%, at least 76%, at least 77%, at least 78%, at least 79%, at least 80%, at least 81%, at least 82%, at least 83%, or greater than 84% at least 85%, at least 86%, at least 87%, at least 88%, at least 89%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or greater than 99% of the cells within the population are positive for three myeloid cell surface marker (e.g., CD14+, CD13+, CD15+, CD1 lb+). In some instances, the methods and compositions can be used to generate a cell population wherein at least 70%, at least 71%, at least 72%, at least 73%, at least 74%, at least 75%, at least 76%, at least 77%, at least 78%, at least 79%, at least 80%, at least 81%, at least 82%, at least 83%, or greater than 84% at least 85%, at least 86%, at least 87%, at least 88%, at least 89%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or greater than 99% of the cells within the population are positive for four myeloid cell surface marker (e.g., CD14+, CD13+, CD15+, CDl lb+). The methods disclosed herein are particularly effective at generating cell populations comprising a plurality of myeloid-derived cells, wherein said population comprises a high percentage of myeloid-derived cells. The myeloid-derived cells can be identified by expression of at least one myeloid-associated marker. Accordingly, the cell populations generated by the compositions and methods comprise a low percentage of non-myeloid cells (e.g., lymphocytes). The cell populations generated by the compositions and methods comprise a low percentage of lymphocytes. For instance, less than 40%, 39%, 38%, 37%, 36%, 35%, 34%, 33%, 32%, 31%, 30%, 29%, 28%, 27%, 26%, 25%, 24%, 23%, 22%, 21%, 20%, 19%, 18%, 17%, 16%, 15%, 14%, 13%, 12%, 11%, 10%, 9%, 8%, 7%, 6%, 5%, 4%, 3%, 2%, 1%, or less than 1% of the cells within the disclosed cell populations generated by the methods of the present application are lymphocytes. The methods and compositions can generate myeloid-derived cell populations
comprising a higher percentage of myeloid-derived cells, and a lower percentage of lymphocytes compared to standard methods and compositions known in the art. The disclosed methods can generate said populations in a shorter amount of time.
For at least the reasons described above, the methods and compositions of the present disclosure provide for significant time and financial savings in the overall manufacturing process of myeloid-derived cells. The myeloid-derived cells produced by the disclosed methods can be myeloid- derived CAR cells. Any myeloid lineage cell comprising a CAR is envisaged as being produced by the methods disclosed herein. As such, the methods provide for a significantly more effective and efficient manufacturing process for monocyte and macrophage cells expressing a CAR (i.e., CAR-M cells). In some instances, the methods disclosed herein can generate a myeloid progenitor cell comprising a CAR. Such efficiency is envisaged for any myeloid cells produced using the methods and compositions disclosed herein.
In some embodiments of the methods disclosed herein, the stem cells have been genetically engineered with a chimeric antigen receptor (CAR), which is stably maintained in the cell throughout the expansion and differentiation process to generate a population of myeloid-derived CAR cells. Also disclosed are media and cytokine mixtures that are particularly effective at promoting the expansion and/or differentiation of stem cells into myeloid-derived CAR cells. The methods disclosed herein involve contacting isolated stem cells with the media disclosed herein to promote the expansion and differentiation of stem cells, and populations comprising the same. Over the course of expansion and differentiation, stem cells will begin to upregulate myeloid markers as they differentiate into myeloid- derived cells. In some embodiments, the media disclosed herein can effectively promote the expansion and the differentiation of stem cells. Importantly, in such instances, the stem cells within the media are increasing in number through cell division while concurrently differentiating into myeloid cells through the upregulation of myeloid markers. Cell populations comprising such cells are also disclosed. In particular, the present disclosure provides cell populations comprising stem cells engineered with a CAR and myeloid-derived CAR cells derived therefrom. The present disclosure provides genetically modified myeloid cells and any precursor thereof expressing a chimeric antigen receptor (CAR), and populations comprising the same. Also disclosed are improved methods of manufacturing a genetically modified myeloid-derived cell comprising a chimeric antigen receptor (CAR). The genetically modified myeloid CAR cells can be generated from any precursor cell (e.g., a stem cell) according to the methods disclosed herein. For instance, a nucleic acid molecule encoding a CAR can be introduced into a precursor cell, such as a hematopoietic stem cell, whereby a genetically modified stem cell is generated, which can be further differentiated into a myeloid lineage cell. As the cells of the present disclosure are differentiated toward the myeloid lineage (and away from the
lymphoid lineage) from a pluripotent stem cell, any cell within the myeloid lineage is envisaged within the scope of the present disclosure as being produced by the methods disclosed herein. For example, the present disclosure relates to methods of manufacturing genetically modified myeloid-derived CAR cells, such as a CAR myeloid progenitor cell or CAR-M cell, from hematopoietic stem cells (HSCs). Accordingly, stem cells comprising a CAR (i.e., CAR stem cells) as well as myeloid-derived CAR cells are also disclosed, as well as populations comprising the same. The HSCs that are used in the disclosed methods can be isolated from a subject. The isolated HSCs are first genetically modified with a CAR and then expanded and differentiated to generate a CAR-M cell or myeloid-derived lineage precursor cell thereof. The present disclosure provides media compositions within which the HSCs can be expanded and differentiated. The media disclosed herein comprise cytokine mixtures that enable superior and improved expansion and differentiation of stem cells into myeloid cells. The expansion and differentiation of the HSCs can occur within a single media (e.g., a precursor media), which contributes to a dramatically improved expansion and differentiation of the stem cells (e.g., hematopoietic stem cells) toward a myeloid lineage cell, for example, a myeloid progenitor cells or differentiated cell thereof. The methods disclosed herein, wherein a HSC is genetically modified with a CAR, prior to expansion and differentiation into a myeloid lineage cell, provide a more stable CAR cell that can be differentiated into a myeloid lineage cell. Any myeloid lineage cell downstream of a HSC is envisaged within the genetically modified myeloid-derived CAR cells of the present disclosure. This includes for example, a myeloid precursor cell, such as a myeloid progenitor cell, granulo- monocyte progenitor cell, monocyte-dendritic cell progenitor/monoblasts, promonocyte, or myeloblast, in addition to a monocyte, macrophage, dendritic cell and/or neutrophil. The genetically modified myeloid-derived CAR cells disclosed herein, when the cell is a precursor cell maintain the ability to differentiate into a mature cell both in vitro and in vivo. Compositions and methods, related to the CAR-modified myeloid-derived cells and use of the same for treatment in a subject in need thereof are also provided.
III. Hematopoietic Stem Cells (HSCs)
The present disclosure provides a method of manufacturing a myeloid cell (i.e., myeloid- derived cell) from a precursor cell, such as a stem cell. In one aspect, disclosed herein is a method of manufacturing a myeloid-derived cell, comprising the step of contacting a precursor cell with a precursor media comprising a precursor cytokine mixture, whereby the number of precursor cell(s) increases to generate a cell population comprising a plurality of precursor cells. In a preferred embodiment, the precursor cell is a stem cell. Also disclosed herein are cell populations comprising a plurality of precursor cells. The myeloid-derived cells disclosed herein do not require a chimeric antigen receptor (CAR). The methods and
compositions disclosed herein can effectively produce a myeloid-derived cell, and a cell population comprising a plurality of myeloid-derived cells, from a precursor cell that does not comprise a CAR. To generate a myeloid-derived cell and/or a cell population comprising a plurality of the same, a hematopoietic stem cell can be contacted with, and maintained in, (i.e., cultured in) a precursor medium and, optionally, a myeloid medium disclosed herein.
In some aspects, disclosed herein is a method for manufacturing a myeloid-derived cell, comprising the step of contacting a precursor cell with a precursor media comprising a precursor cytokine mixture, whereby the number of precursor cell(s) increases to generate a cell population comprising a plurality of precursor cells, wherein the method further comprises the step of contacting said cell population comprising a plurality of precursor cells with a myeloid media comprising a myeloid cytokine mixture, whereby said precursor cell(s) differentiates into a myeloid-lineage cell to generate a myeloid-derived cell and/or a mixed cell population comprising a plurality of myeloid-derived cells and/or a plurality of precursor cells. The precursor cell can be a stem cell and/or a CD34+ cell. The hematopoietic stem cells (HSCs) can be expanded (i.e., to generate a cell population comprising a plurality of HSCs) and differentiated (i.e., to generate a myeloid-derived cell and/or a mixed population comprising a plurality of myeloid-derived cells and/or a plurality of HSCs) in the precursor media disclosed herein. In some instances, the HSCs in the precursor media are within a cell population, wherein a plurality of HSCs within the population are undergoing expansion, and/or wherein a plurality of HSCs within the population are undergoing differentiation. A skilled artisan would understand the processes of expansion and differentiation can occur simultaneously within the cell populations that are maintained in precursor media. As such, it is possible that an HSC that is maintained in a precursor media will generate a cell population comprising a plurality of HSCs and a plurality of myeloid-derived cells. In some aspects of the method disclosed herein, the hematopoietic stem cell (HSC) is maintained in a precursor media, whereby the number of HSCs increases to generate a cell population comprising a plurality of HSCs, and whereby upon contact of said HSCs with said precursor media, said HSCs differentiates into a myeloid-lineage cell(s) to generate a myeloid-derived cell(s) and/or a mixed cell population comprising a plurality of myeloid-derived cells and a plurality of HSC cells. In such instances, the method can further comprise the step of contacting said mixed cell population with a myeloid media comprising a myeloid cytokine mixture, whereby upon contact with said myeloid media: a. the ratio of said myeloid-derived cells to HSCs in said mixed cell population is increased. In such instances, the precursor media can be removed prior to addition of the myeloid media.
The methods and compositions disclosed herein can also effectively generate a myeloid- derived CAR cell from a precursor CAR cell. Any of the methods and/or compositions disclosed
herein are envisaged as to be as effective for expansion and differentiation of an HSC CAR cell as with an HSC. The presence of a CAR in the HSC are not envisaged as impacting the effectiveness or the efficacy of the methods disclosed herein to expand and differentiate a precursor cell and/or a precursor CAR cell. The present disclosure provides a method of manufacturing a genetically modified myeloid cell (i.e., myeloid-derived cell) comprising a chimeric antigen receptor (CAR) (i.e., myeloid-derived CAR cell), wherein the genetically modified CAR cell has been generated from a precursor according to the methods described herein. The precursor cells can comprise a bone marrow-derived cell or a stem cell. In some instances, the bone-marrow derived cell is a stem cell. In a preferred embodiment, the precursor cell is a stem cell. One type of stem cell that can be used is a hematopoietic stem cell (HSC). In another preferred embodiment, the precursor cell is a hematopoietic stem cell (HSC). The methods and compositions disclosed herein are useful for expanding hematopoietic stem cells. In some embodiments, the hematopoietic stem cells have been engineered with a CAR prior to expansion. The methods and compositions disclosed herein are also particularly useful for differentiating an HSC, or a population of cells comprising a plurality of HSCs, into a myeloid lineage cell.
In some aspects, disclosed herein is a method for manufacturing a myeloid-derived CAR cell, comprising the step of contacting a precursor CAR cell with a precursor media comprising a precursor cytokine mixture, whereby the number of precursor CAR cell(s) increases to generate a cell population comprising a plurality of precursor CAR cells, wherein the method further comprises the step of contacting said cell population comprising a plurality of precursor CAR cells with a myeloid media comprising a myeloid cytokine mixture, whereby said precursor CAR cell(s) differentiates into a myeloid-lineage cell to generate a myeloid-derived CAR cell and/or a mixed cell population comprising a plurality of myeloid-derived CAR cells and/or a plurality of precursor CAR cells. The precursor CAR cell can be a stem cell and/or a CD34+ cell. The hematopoietic stem cells (HSCs) comprising a CAR can be expanded (i.e., to generate a cell population comprising a plurality of HSCs comprising a CAR) and differentiated (i.e., to generate a myeloid- derived CAR cell and/or a mixed population comprising a plurality of myeloid-derived CAR cells and/or a plurality of HSCs comprising a CAR) in the precursor media disclosed herein. In some instances, the HSCs comprising a CAR in the precursor media are within a cell population, wherein a plurality of HSCs comprising a CAR within the population are undergoing expansion, and/or wherein a plurality of HSCs comprising a CAR within the population are undergoing differentiation. As described above, it is possible that an HSC comprising a CAR that is maintained in a precursor media will generate a cell population comprising a plurality of HSCs comprising a CAR and a plurality of myeloid-derived CAR cells. In some aspects of the method disclosed
herein, the hematopoietic stem cell (HSC) comprising a CAR is maintained in a precursor media, whereby the number of HSCs comprising a CAR increases to generate a cell population comprising a plurality of HSCs comprising a CAR, and whereby upon contact of said HSCs with said precursor media, said HSCs differentiates into a myeloid-lineage cell(s) to generate a myeloid-derived CAR cell(s) and/or a mixed cell population comprising a plurality of myeloid-derived CAR cells and a plurality of HSC cells comprising a CAR. In such instances, the method can further comprise the step of contacting said mixed cell population with a myeloid media comprising a myeloid cytokine mixture, whereby upon contact with said myeloid media: a. the ratio of said myeloid-derived CAR cells to HSCs comprising a CAR in said mixed cell population is increased. In such instances, the precursor media can be removed prior to addition of the myeloid media.
The HSCs can be differentiated into any myeloid lineage cell. Exemplary myeloid cells into which the HSCs can be differentiated using the disclosed methods and compositions are described herein. The myeloid-derived CAR cells that can be generated from the disclosed methods can be used in the treatment of a disease or disorder, such as cancer, or in the manufacture of a medicament for use in treating a disease or a disorder. Any treatment, disease or disorder disclosed herein is envisaged. The HSCs disclosed herein can be isolated from a subject, expanded, and differentiated into myeloid-derived CAR cell, or a population of myeloid-derived CAR cells, whereby said myeloid-derived CAR cell is administered to said subject for treatment. Accordingly, the myeloid-derived CAR cells disclosed herein are administered to the same subject from which the HSCs used to generated said myeloid-derived CAR cells were isolated. That is, in some embodiments, the HSCs disclosed herein can be used to generate an autologous myeloid-derived cell. In some embodiments, said HSCs are engineered with a CAR prior to generating an autologous myeloid cell, generating a myeloid-derived CAR cell. Accordingly, the methods and compositions disclosed herein are particularly effective at generating myeloid-derived CAR cells from HSCs ex vivo. Any myeloid cell disclosed herein can be generated from the isolated HSCs disclosed herein ex vivo.
Hematopoietic stem cells (HSCs) are multipotent precursors that have a unique ability to self-renew. HSCs produce hematopoietic progenitor cells that differentiate into every type of mature blood cell within a well-defined hierarchy (Bonnet, 2002; McCulloch and Till, 2005). HSCs can be found in various tissue, including bone marrow, mobilized peripheral blood, peripheral blood, and umbilical cord blood. HSCs and other blood cell progenitors can be isolated from fetal and embryonic tissues. Specifically, umbilical cord blood (UCB) and placenta are rich sources of HSCs (Abdulrazzak, Hassan et al. “Biological characteristics of stem cells from foetal, cord blood and extraembryonic tissues.” Journal of the Royal Society, Interface vol. 7 Suppl 6, Suppl 6 (2010):
S689-706). Somatic cells can also be a source of HSCs, by conversion of these cells into induced pluripotent stem cells (IPSCs) (Vo, Linda T, and George Q Daley. “De novo generation of HSCs from somatic and pluripotent stem cell sources.” Blood vol. 125,17 (2015): 2641-8). Through the process of differentiation, HSCs give rise to two different lines of blood cells called myeloid and lymphoid. Examples of myeloid cells include, but are not limited to, monocytes, macrophages, neutrophils, basophils, eosinophils, erythrocytes, and megakaryocytes to platelets. Non-limiting examples of lymphoid cells include T cells, B cells, natural killer cells, and innate lymphoid cells.
CD34 is a type I transmembrane glycophosphoprotein predominantly regarded as a marker of hematopoietic stem cells (HSC) and hematopoietic progenitor cells (Civin et al., 1996b; Shizuru et al., 2005; Shpall et al., 1994). CD34 is expressed on -0.2-3% of the nucleated cells in cord blood, bone marrow and mobilized peripheral blood (Civin et al., 1984; Krause et al., 1996; Sutherland et al., 1996). CD34 has commonly been used to identify and select for populations of HSCs. For example, in clinical practice, CD34 expression is evaluated to ensure rapid engraftment in BM transplants; CD34 can also be used as a selective marker in cell sorting to enrich a population of immature hematopoietic cells (Berardi, A C et al. “Functional isolation and characterization of human hematopoietic stem cells.” Science (New York, N.Y.) vol. 267,5194 (1995): 104-8; Berenson, R J et al. “Engraftment after infusion of CD34+ marrow cells in patients with breast cancer or neuroblastoma.” Blood vol. 77,8 (1991): 1717-22.).
In addition to the requirement for essential components such as glucose, vitamins, amino acids, etc., which are vital for stem cell metabolism, the successful culture and maintenance of hematopoietic stem cells in an undifferentiated state in vitro requires additional considerations to ensure that cells maintain their key characteristics of self-renewal and pluripotency. For example, precise media compositions can be required to properly balance aspects of HSC physiology and control self-renewal, expansion and lineage commitment (e.g., differentiation) in vitro. Thus, culturing HSCs requires a highly controlled environment where slight differences in media composition (e.g., basal media, added proteins) and environmental stimuli (e.g., temperature, time) can impact the expansion and differentiation of HSCs.
Various types of basal media are available for use in culturing stem cells. While serum-free media can be used, the addition of serum and other growth factors, is commonly employed to achieve a desired proliferation and/or differentiation of cells. One example of factors that are commonly added to media include cytokines. Hematopoietic cytokines are large family of extracellular ligands that stimulate hematopoietic cells to differentiate into eight principle types of blood cells. Numerous cytokines are involved in the regulation of hematopoiesis within a complex network of positive and negative regulators. Some cytokines have very narrow lineage specificities
of their actions, while many others have rather broad and overlapping specificity ranges (Metcalf D. 2008. Hematopoietic cytokines. 111(2):485-491).
Some cytokines seem to be more involved in processes involving the stimulation or regulation of hematopoietic cells. These include, for example: granulocyte-macrophage colony stimulating factor (GM-CSF), granulocyte-colony stimulating factor (G-CSF), macrophage-colony stimulating factor (M-CSF), interleukins, EPO and TPO. There are a number of cytokines that exert profound effects on the formation and maturation of hematopoietic cells, including stem cell factor (SCF), flt-3/flk-2 ligand (FL) and leukemia inhibitory factor (LIF). Other cytokines or ligands such as jagged-1, transforming growth factor-P (TGF-P) and tumor necrosis factor-a (TNF-a) also play significant roles in modulating hematopoiesis. A cytokine would be understood to be a type of protein that is made by certain immune and non-immune cells and has an effect on the immune system. Some cytokines stimulate the immune system and others slow it down. They can also be made in the laboratory and used to help the body fight cancer, infections, and other diseases. Nonlimiting, examples of cytokines include: interleukins, interferons, and colony-stimulating factors (i.e., a substance that stimulates the production of blood cells, including, for example, granulocyte colony-stimulating factor (G-CSF), granulocyte-macrophage colony-stimulating factor (GM-CSF), and promegapoietin). Cytokines have been shown to bind directly to receptors on hematopoietic stem cells (HSCs) to regulate HSC functions, including quiescence, self-renewal, differentiation, apoptosis, and mobility. Increasing evidence suggests that HSCs respond to multiple cytokines and that the fate of an HSC - self renewal, apoptosis, mobilization from the niche, formation of differentiated progeny cells - depends on these cytokines, adhesion proteins, and other signals produced by stromal cells and likely other cells in the body (Zhang, Cheng C, and Harvey F Lodish. “Cytokines regulating hematopoietic stem cell function.” Current opinion in hematology vol. 15,4 (2008): 307-11). Thus, the presence of cytokines within a culture media (e.g., expansion and/or differentiation media) is an important aspect to consider for maintenance, expansion and differentiation of HSCs in vitro (McNiece, I K, and R A Briddell. “Stem cell factor.” Journal of leukocyte biology vol. 58,1 (1995): 14-22; Ding, Lei et al. “Endothelial and perivascular cells maintain haematopoietic stem cells.” Nature vol. 481,7382 457-62. 25 Jan. 2012).
Expansion and Differentiation of HSCs
Large numbers of hematopoietic stem cells can be produced through in vitro stem cell expansion, which is a long-term culture of HSCs with very limited or reduced amounts of cell exhaustion or differentiation of the cells. Cell exhaustion and differentiation can commonly occur during culture. Appropriate culture conditions, such as an expansion media containing the necessary supplements are required to control HSC proliferation, differentiation and viability. A
variety of media and culturing conditions has been used to successfully expand HSCs. These aspects of HSC culturing and expansion are widely available and known within the art. Many commonly used modified basal media and corresponding recipes are well establish and are commercially available. Hematopoietic stem cells differentiate into various lineages. Each type of stem cell can require a culture medium supplemented with specific growth factors to sustain cell proliferation and differentiation into precise lineages. HSCs differentiation process is regulated by intracellular signaling pathways, which can be tightly regulated and controlled by growth factors and cytokines. Various studies examining in vitro HSCs differentiation indicate the media composition, including the addition of cytokines and growth factors, and culture conditions can impact differentiation efficacy (Yadav, Pooja et al. “Hematopoietic Stem Cells Culture, Expansion and Differentiation: An Insight into Variable and Available Media.” International journal of stem cells vol. 13,3 (2020): 326-334).
Disclosed herein are compositions and methods for expanding and differentiating hematopoietic stem cells. The HSCs that are expanded and differentiated in the present disclosure can be isolated from a subject, such as a human subject. HSCs isolated from a human subject can be identified and quantified according to any methods known in the art prior to expanding the HSCs. In the methods of generating a genetically modified cell disclosed herein, the HSCs can be expanded and differentiated after the cells are contacted with an agent comprising a nucleic acid molecule comprising a polynucleotide sequence encoding the CAR. In some instances, the HSCs that are expanded and differentiated are HSCs, or a derivative therefrom, that comprise a nucleic acid molecule comprising a polynucleotide sequence. In some instances, the polynucleotide sequence encodes a CAR. The nucleic acid molecule comprising the polynucleotide sequence is maintained within the HSCs and population of HSCs during and throughout expansion and differentiation. It is not anticipated that the nucleic acid molecule comprising the polynucleotide sequence is removed or lost from the HSCs, or the population thereof, or that the nucleic acid molecule is otherwise reduced within the population. As the HSC expands, the number of HSCs increases in number, whereby a population of HSCs is generated.
The methods disclosed herein comprise a method of manufacturing a genetically modified cell comprising a chimeric antigen receptor (CAR), the method comprising: (a) contacting a precursor with an agent comprising a nucleic acid molecule comprising a polynucleotide sequence encoding the CAR to generate a precursor-CAR cell; and (b) inducing differentiation of said precursor-CAR cell into a phagocytic cell to generate a genetically modified phagocytic cell. The methods can further comprise: the step of (c) expanding the genetically modified phagocytic cell to generate a cell population comprising a plurality of genetically modified phagocytic cells. Without
wishing to be bound by theory, as the expansion time increases, the number of HSCs within the population should increase. The cell population of HSCs can be expanded and/or differentiated for any number of days required to achieve the number of HSCs desired. In some instances, the HSCs can be expanded and/or differentiated for at least one day, at least 2 days, at least 3 days, at least 4 days, at least 5 days, at least 6 days, at least 7 days, at least 8 days, at least 9 days, at least 10 days, or more than 10 days. In some instances, the expansion and/or differentiation step can last for 1 day, 2 days, or 3 days. In some instances, the expansion step and/or differentiation step can last for 3 days. One purpose for the expansion step is to increase the number of HSCs within a population, therefore, the expansion step may be maintained for as long as necessary to achieve the desired number of HSCs within the population. The desired number of cells would be understood and determined by the skilled artisan at the time of performing the method. The desired number of cells may be determined by the initial number of starting cells. Specifically, the desired number of cells may be a multiple of the initial number of starting cells. A skilled artisan would understand how to calculate the number of starting cells, as well as the number of current cells at any point throughout the expansion step and would be able to determine the amount of time to maintain the expansion phase. Any method of calculating the number of cells within the population that is available to one of skill in the art is envisaged within the present disclosure. In some instances, the number of cells is decided based on a desired concentration. In some instances, the expansion step can be maintained until the number of HSCs within a population has increased a specific amount relative to the starting number of cells. In some instances, the HSCs are expanded to a number 100-fold, 200-fold, 300-fold, 400-fold, 500-fold, or greater than 500-fold of the initial starting HSC number. In some instances, the HSCs are expanded to a number 100-fold the initial starting HSC number. In some instances, the HSCs are expanded to a number 200-fold the initial starting HSC number. In some instances, the HSCs are expanded to a number 300-fold the initial starting HSC number. In some instances, the HSCs are expanded to a number 400-fold the initial starting HSC number. In some instances, the HSCs are expanded to a number 500-fold the initial starting HSC number. In some instances, wherein the HSCs are expanded to a number 300-fold the initial starting HSC number, the expansion lasts for 8 days.
IV. Myeloid-Derived Cells and Myeloid-Derived Cells Genetically Modified to Express a Chimeric Antigen Receptors (CAR)
The present disclosure provides a myeloid-derived cell that has been generated from a stem cell using the methods and compositions disclosed herein. Also disclosed are cell populations comprising a plurality of the myeloid-derived cells disclosed herein. Importantly, the methods disclosed herein are envisaged as being effective at generating both myeloid-derived cells, as well as myeloid-derived CAR
cells. As such, it would be understood that any myeloid-derived cell, or population comprising a myeloid-derived cell, generated from the disclosed methods can include, and encompass, for the purposes of the present disclosure, a myeloid-derived CAR cell and/or a myeloid-derived cell that does not comprise a CAR (i.e., a myeloid-derived cell). For instance, any cell population described in the present application can be generated to comprise myeloid-derived cells and/or myeloid-derived CAR cells.
The present disclosure provides a myeloid-derived cell genetically modified to express a CAR. The myeloid-derived cells disclosed herein have been generated by expanding and differentiating a precursor CAR cell (e.g., a stem cell CAR cell) using the precursor and, optionally, the myeloid media disclosed herein. The precursor CAR cell can be differentiated into any myeloid-derived CAR cell known in the art. Myeloid cells (i.e., cells within the myeloid lineage that are derived from a common myeloid progenitor cell) would be readily known in the art. In some instances, the myeloid-derived CAR cell is a partially differentiated myeloid-derived cell. A partially differentiated cell is a cell that has undergone differentiation, and thus is no longer a stem cell, and has the capacity to undergo additional differentiation into a more specialized myeloid cell within the myeloid lineage. In some instances, the myeloid-derived CAR cell is fully differentiated myeloid cell. A skilled artisan would readily understand how to identity a myeloid cell that was partially or fully differentiated. The organization of the myeloid lineage, and, for example, the differentiation capacity of myeloid cells, is known in the art, as described for example, in Weiskopf, Kipp et al. “Myeloid Cell Origins, Differentiation, and Clinical Implications.” Microbiology spectrum vol. 4,5 (2016): 10.1128/microbiolspec.MCHD-0031-2016; Haas, Lisa, and Anna C Obenauf. “Allies or Enemies-The Multifaceted Role of Myeloid Cells in the Tumor Microenvironment.” Frontiers in immunology vol. 10 2746. 28 Nov. 2019; and Lambert, Claude et al. “Monocytes and macrophages in flow: an ESCCA initiative on advanced analyses of monocyte lineage using flow cytometry.” Cytometry. PartB, Clinical cytometry vol. 92,3 (2017): 180-188, the contents of which are all incorporated herein in their entirety. Exemplary, non-limiting, cells within the myeloid lineage include: common myeloid progenitor (CMP), granulo-monocyte progenitors, monocyte-dendritic cell progenitors, common dendritic cell progenitors, myeoblasts, monoblasts, pre-conventional dendritic cells, promocytes, granulocytes, polymorphonuclear leukocytes (PMNs), myeloid-derived suppressor cells (MDSCs), mast cells, basophils, eosinophils, megakaryocytes, neutrophils, dendritic cells, monocytes, and macrophages. Cell surface markers can be used to distinguish myeloid cells. A skilled artisan would readily understand that a myeloid-derived cell can be identified and isolated based on cell surface markers, using any method available in the art. In some embodiments, the myeloid-derived CAR cell is a granulocyte. In some embodiments, the myeloid-derived CAR cell is a phagocytic cell. In some
embodiments, the myeloid-derived CAR cell is a monocyte, macrophage, dendritic cell, neutrophil, or a precursor thereof.
Also disclosed herein are cell populations comprising the genetically modified precursor CAR cells and/or myeloid-derived CAR cells generated by the methods described herein. The precursor CAR cell(s) disclosed herein can be expanded to generate a population comprising a plurality of genetically modified precursor CAR cells or expanded and differentiated to generate a population comprising a plurality of genetically modified myeloid-derived CAR cells. The genetically modified myeloid-derived cells can include any cell within the myeloid lineage that has been derived from a precursor as described herein. The precursor can comprise a bone marrow-derived cell, a blood-derived cell, or a stem cell. For example, the precursor can be a hematopoietic stem cell. Disclosed herein are cell populations comprising a plurality of precursor CAR cells. In some embodiments, wherein the cell population comprises a plurality of precursor CAR cells, said precursor cell is a stem cell. In some embodiments, the stem cell is a hematopoietic stem cell. In some embodiments of the cell populations disclosed herein, the cell population comprises a plurality of precursor CAR cells, wherein said precursor cell is a hematopoietic stem cell. The cell populations disclosed herein can be mixed populations. For instance, the cell population can comprise a plurality of precursor CAR cells and a plurality of myeloid-derived CAR cells. Any ratio of precursor CAR cells to myeloid-derived CAR cells within the cell populations disclosed herein is envisaged within the scope of the present application. The cell populations disclosed herein can be contacted with the precursor and myeloid medias disclosed herein. In some instances, the cell populations are maintained in the precursor and/or myeloid medias until the cells within the population have expanded and/or differentiated to a desirable amount. Without wishing to be bound by theory, it is likely that the longer than a cell population is contacted with a precursor media and/or myeloid media, the more the cell population will undergo differentiation, when maintained under conditions suitable for differentiation of cells within the population. When the cell populations are contacted with the precursor medias and/or myeloid medias of the present disclosure, any of the precursor cells within the cell population can undergo differentiation into a myeloid-derived cell. As such, when a cell population is contacted with and/or maintained in any of the precursor medias disclosed herein, the ratio of myeloid-derived cells to precursor cells will be expected to increase (i.e., the number of myeloid-derived cells in the population increases compared to the number of precursor cells). When a cell population is contacted with and/or maintained in any of the precursor medias disclosed herein, the number of myeloid-derived cells within the population can increase and/or the number of precursor cells within the population can decrease. In some instances, when the cell population is contacted with a precursor media disclosed herein, the cell population can undergo expansion (i.e., the number of cells within the population
increases). In some instances, when the cell population is contacted with a precursor media disclosed herein, the cell population can undergo differentiation (i.e., a plurality of precursor cells within the population differentiates into myeloid cells).
The myeloid-derived cell that is generated from the precursor cell can include any myeloid lineage cell disclosed herein. The myeloid-derived cell that is generated from the precursor cell can include any myeloid cell disclosed herein. The myeloid-derived cell that is generated from the precursor can include, for example, phagocytic cells, such as a macrophage, monocyte, dendritic cell, neutrophil, and/or precursor thereof. The myeloid-derived cell that is generated from the precursor can be a precursor of a phagocytic cells, such as any cell within the myeloid lineage, such as a myeloid precursor cell, such as a myeloid progenitor cell, granulo-monocyte progenitor cell, monocyte- dendritic cell progenitor/monoblasts, promonocyte, or myeloblast. The phagocytic cell, or precursor thereof, comprising a CAR recognizes and binds to an antigen present on the surface of a target cell, such as a cancer cell and/or viral particle. The CAR includes a co-stimulatory domain having at least 90% sequence identity to an HVEM co-stimulatory protein set forth as any one of SEQ ID NOs: 7, 1, or 2, or a functional fragment or variant thereof that retains co-stimulatory activity. Populations of cells comprising a plurality wherein at least 40%, at least 50%, at least 60%, at least 70%, at least 80%, at least 90%, at least 95%, or at least 99% of said plurality expresses a CAR on the cell surface are also disclosed.
Disclosed herein are cell populations comprising a plurality of any of the genetically modified myeloid-derived CAR cells generated by the method described herein. Also disclosed are cell populations comprising a plurality of any of the genetically modified precursor CAR cells described herein. The cell populations disclosed herein can comprise any of the precursor CAR cells and/or myeloid-derived CAR cells disclosed herein. When the cell populations comprise a precursor CAR cell and myeloid-derived CAR cell, any ratio of the number of precursor CAR cells to the number of myeloid-derived CAR cells is envisaged as within the scope of the cell populations of the present application.
When the myeloid-derived CAR cells are differentiated from a stem cell, at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of said plurality can be CD1 lb+ or CD14+ or Cl lb+CD14+.
The cell populations disclosed herein can comprise a plurality of cells with any of the surface marker expression profiles disclosed herein. The cells within the cell populations disclosed herein can comprise any of the surface marker expression profiles disclosed herein.
In some embodiments, disclosed herein is a cell population comprising a plurality of precursor CAR cells and a plurality of myeloid-derived CAR cells, wherein between 90%-99.9% of said population is CD34+ cells.
In some embodiments, disclosed herein is a cell population wherein said population is a mixed population comprising a plurality of precursor CAR cells and a plurality of myeloid-derived CAR cells, wherein at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% of said population is CD34+ cells.
In some embodiments, disclosed herein is a cell population wherein said population is a mixed population comprising a plurality of precursor CAR cells and a plurality of myeloid-derived CAR cells, or a plurality of precursor cells and a plurality of myeloid-derived cells, wherein: a. no more than 99%, no more than 98%, no more than 97%, no more than 96%, no more than 95%, no more than 90%, no more than 80%, no more than 70%, no more than 60%, no more than 50%, no more than 40%, no more than 30%, no more than 20%, no more than 10%, no more than 5%, no more than 1%, no more than 0.5%, or no more than 0.1% of said mixed population of cells is CD34+; b. no more than 5%, no more than 4%, no more than 3%, no more than 2%, no more than 1%, no more than 0.5%, or no more than 0.1% of said mixed population of cells is CD34+; c. at least 20%, at least 30%, at least 40%, at least 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 95%, or at least 90% of said population is CD15+; d. between about 60%-90% of said mixed population of cells is CD15+; e. at least 20%, at least 30%, at least 40%, at least 50%, at least 60%, at least 70%, at least 75%, at least 80%, or at least 85%, of said mixed population is CD1 lb+; f. between about 30%-80% of said mixed population of cells is CD1 lb+; g. at least 0.1%, at least 0.5%, at least 1%, at least 2%, at least 3%, at least 4%, at least 5%, at least 6%, at least 7%, at least 8%, at least 9%, or at least 10% of said mixed population of cells is CD14+; h. between about 0.5%-10% of said mixed population of cells is CD14+; i. at least 20%, at least 30%, at least 40%, at least 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95% of said mixed population is CD13+; and/or j. between about 85%-95% of said mixed population of cells is CD13+. In such instances, the myeloid-derived cell or myeloid-derived CAR cell can be a common myeloid progenitor (CMP), granulo-monocyte progenitor, monocyte- dendritic cell progenitor, common dendritic cell progenitor, myeoblast, monoblast, pre- conventional dendritic cell, promocyte, granulocyte, polymorphonuclear leukocyte (PMN), myeloid-derived suppressor cell (MDSC), mast cell, basophil, eosinophils, megakaryocyte, neutrophil, dendritic cell, monocyte, and/or a macrophage. In such instances, the precursor cell or precursor CAR cell can be a hematopoietic stem cell (HSC).
In some embodiments, disclosed herein is a cell population wherein said population is a mixed population comprising a plurality of precursor CAR cells and a plurality of myeloid-derived CAR cells , or a plurality of precursor cells and a plurality of myeloid-derived cells, wherein: a. no more than 99%, no more than 98%, no more than 97%, no more than 96%, no more than 95%, no more than 90%, no more than 80%, no more than 70%, no more than 60%, no more than 50%, no more than 40%, no more than 30%, no more than 20%, no more than 10%, no more than 5%, no more than 1%, no more than 0.5%, or no more than 0.1% of said mixed population of cells is CD34+; b. no more than 5%, no more than 4%, no more than 3%, no more than 2%, no more than 1%, no more than 0.5%, or no more than 0.1% of said mixed population of cells is CD34+; c. at least 20%, at least 30%, at least 40%, at least 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 95%, or at least 90% of said population is CD15+; d. between about 60%-90% of said mixed population of cells is CD15+; e. at least 20%, at least 30%, at least 40%, at least 50%, at least 60%, at least 70%, at least 75%, at least 80%, or at least 85%, of said mixed population is CD1 lb+; f. between about 30%-80% of said mixed population of cells is CD1 lb+; g. at least 0.1%, at least 0.5%, at least 1%, at least 2%, at least 3%, at least 4%, at least 5%, at least 6%, at least 7%, at least 8%, at least 9%, or at least 10% of said mixed population of cells is CD14+; h. between about 0.5%-10% of said mixed population of cells is CD14+; i. at least 20%, at least 30%, at least 40%, at least 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95% of said mixed population is CD13+; and/or j. between about 85%-95% of said mixed population of cells is CD13+, wherein the myeloid-derived cell or myeloid-derived CAR cell is a common myeloid progenitor (CMP), granulo-monocyte progenitor, monocyte-dendritic cell progenitor, common dendritic cell progenitor, myeoblast, monoblast, pre-conventional dendritic cell, promocyte, granulocyte, polymorphonuclear leukocyte (PMN), myeloid-derived suppressor cell (MDSC), mast cell, basophil, eosinophils, megakaryocyte, neutrophil, dendritic cell, monocyte, and/or a macrophage.
In some embodiments, disclosed herein is a cell population wherein said population is a mixed population comprising a plurality of precursor CAR cells and a plurality of myeloid-derived CAR cells, or a plurality of precursor cells and a plurality of myeloid-derived cells, wherein a. no more than 1%, no more than 0.9%, no more than 0.8%, no more than 0.7%, no more than 0.6%, no more than 0.5%, no more than 0.4%, no more than 0.3%, no more than 0.2%, no more than 0.1%, or less than 0.1% of said population of cells is CD34+; b. at least 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95% of said population is CD15+; c. between about 50%-95% of said population of cells is CD15+; d. least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of said population is CD1 lb+; e. between about 75%-99% of said
population of cells is CD1 lb+; f. at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, at least 50%, at least 60%, at least 70%, at least 80%, or at least 90% of said population of cells is CD14+; g. between about 5%-90% of said population of cells is CD14+; h. at least 85%, at least 90%, at least 95%, or at least 99% of said population is CD13+; and/or i. between about 85%-99% of said population of cells is CD13+. In such instances, the myeloid-derived cell or myeloid- derived CAR cell can be a common myeloid progenitor (CMP), granulo-monocyte progenitor, monocyte-dendritic cell progenitor, common dendritic cell progenitor, myeoblast, monoblast, pre- conventional dendritic cell, promocyte, granulocyte, polymorphonuclear leukocyte (PMN), myeloid-derived suppressor cell (MDSC), mast cell, basophil, eosinophils, megakaryocyte, neutrophil, dendritic cell, monocyte, and/or a macrophage. In such instances, the precursor cell can be a hematopoietic stem cell (HSC).
In some embodiments, disclosed herein is a cell population wherein said population is a mixed population comprising a plurality of precursor CAR cells and a plurality of myeloid-derived CAR cells, or a plurality of precursor cells and a plurality of myeloid-derived cells, wherein a. no more than 1%, no more than 0.9%, no more than 0.8%, no more than 0.7%, no more than 0.6%, no more than 0.5%, no more than 0.4%, no more than 0.3%, no more than 0.2%, no more than 0.1%, or less than 0.1% of said population of cells is CD34+; b. at least 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95% of said population is CD15+; c. between about 50%-95% of said population of cells is CD15+; d. least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of said population is CD1 lb+; e. between about 75%-99% of said population of cells is CD1 lb+; f. at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, at least 50%, at least 60%, at least 70%, at least 80%, or at least 90% of said population of cells is CD14+; g. between about 5%-90% of said population of cells is CD14+; h. at least 85%, at least 90%, at least 95%, or at least 99% of said population is CD13+; and/or i. between about 85%-99% of said population of cells is CD13+, wherein the myeloid-derived cell or myeloid-derived CAR cell is a common myeloid progenitor (CMP), granulo-monocyte progenitor, monocyte-dendritic cell progenitor, common dendritic cell progenitor, myeoblast, monoblast, pre-conventional dendritic cell, promocyte, granulocyte, polymorphonuclear leukocyte (PMN), myeloid-derived suppressor cell (MDSC), mast cell, basophil, eosinophils, megakaryocyte, neutrophil, dendritic cell, monocyte, and/or a macrophage.
Any of the cell populations disclosed above can comprise any number of cells disclosed herein. The cell population can comprise at least 104, at least 105, at least 106, at least 107, at least 108, at least 109, at least 1010, at least 1011, at least 1012, at least 1013, at least 1014, at least 1015, at least 1016, at least 1017, at least 1018, at least 1019, at least IO20, or more than IO20 cells. In such
instances, when the cell population comprises at least 104, at least 105, at least 106, at least 107, at least 108, at least 109, at least IO10, at least IO11, at least 1012, at least IO13, at least 1014, at least IO15, at least 1016, at least 1017, at least 1018, at least 1019, at least IO20, or more than IO20 cells, wherein said population comprises a plurality of myeloid-derived cells and/or myeloid-derived CAR cells, and wherein: a. no more than 99%, no more than 98%, no more than 97%, no more than 96%, no more than 95%, no more than 90%, no more than 80%, no more than 70%, no more than 60%, no more than 50%, no more than 40%, no more than 30%, no more than 20%, no more than 10%, no more than 5%, no more than 1%, no more than 0.5%, or no more than 0.1% of said population of cells is CD34+; b. no more than 5%, no more than 4%, no more than 3%, no more than 2%, no more than 1%, no more than 0.5%, or no more than 0.1% of said population of cells is CD34+; c. at least 20%, at least 30%, at least 40%, at least 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 95%, or at least 90% of said population is CD15+; d. between about 60%-90% of said mixed population of cells is CD15+; e. at least 20%, at least 30%, at least 40%, at least 50%, at least 60%, at least 70%, at least 75%, at least 80%, or at least 85%, of said population is CD1 lb+; f. between about 30%-80% of said population of cells is CD1 lb+; g. at least 0.1%, at least 0.5%, at least 1%, at least 2%, at least 3%, at least 4%, at least 5%, at least 6%, at least 7%, at least 8%, at least 9%, or at least 10% of said population of cells is CD14+; h. between about 0.5%-10% of said population of cells is CD14+; i. at least 20%, at least 30%, at least 40%, at least 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95% of said population is CD13+; and/or j. between about 85%-95% of said population of cells is CD13+. In such instances, the myeloid-derived cell and/or myeloid-derived CAR cell can be a common myeloid progenitor (CMP), granulo-monocyte progenitor, monocyte-dendritic cell progenitor, common dendritic cell progenitor, myeoblast, monoblast, pre-conventional dendritic cell, promocyte, granulocyte, polymorphonuclear leukocyte (PMN), myeloid-derived suppressor cell (MDSC), mast cell, basophil, eosinophils, megakaryocyte, neutrophil, dendritic cell, monocyte, and/or a macrophage.
The cell population can comprise at least 104, at least 105, at least 106, at least 107, at least 108, at least 109, at least 1010, at least 1011, at least 1012, at least 1013, at least 1014, at least 1015, at least 1016, at least 1017, at least 1018, at least 1019, at least IO20, or more than IO20 cells. In such instances, when the cell population comprises at least 104, at least 105, at least 106, at least 107, at least 108, at least 109, at least IO10, at least 1011, at least 1012, at least 1013, at least 1014, at least 1015, at least 1016, at least 1017, at least 1018, at least 1019, at least IO20, or more than IO20 cells, wherein said population comprises a plurality of myeloid-derived cells and/or myeloid-derived CAR cells, and wherein: a. no more than 99%, no more than 98%, no more than 97%, no more than 96%, no more than 95%, no more than 90%, no more than 80%, no more than 70%, no more than 60%, no
more than 50%, no more than 40%, no more than 30%, no more than 20%, no more than 10%, no more than 5%, no more than 1%, no more than 0.5%, or no more than 0.1% of said population of cells is CD34+; b. no more than 5%, no more than 4%, no more than 3%, no more than 2%, no more than 1%, no more than 0.5%, or no more than 0.1% of said population of cells is CD34+; c. at least 20%, at least 30%, at least 40%, at least 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 95%, or at least 90% of said population is CD15+; d. between about 60%-90% of said mixed population of cells is CD15+; e. at least 20%, at least 30%, at least 40%, at least 50%, at least 60%, at least 70%, at least 75%, at least 80%, or at least 85%, of said population is CD1 lb+; f. between about 30%-80% of said population of cells is CD1 lb+; g. at least 0.1%, at least 0.5%, at least 1%, at least 2%, at least 3%, at least 4%, at least 5%, at least 6%, at least 7%, at least 8%, at least 9%, or at least 10% of said population of cells is CD14+; h. between about 0.5%-10% of said population of cells is CD14+; i. at least 20%, at least 30%, at least 40%, at least 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95% of said population is CD13+; and/or j. between about 85%-95% of said population of cells is CD13+, wherein the myeloid- derived cell and/or myeloid-derived CAR cell is a common myeloid progenitor (CMP), granulo- monocyte progenitor, monocyte-dendritic cell progenitor, common dendritic cell progenitor, myeoblast, monoblast, pre-conventional dendritic cell, promocyte, granulocyte, polymorphonuclear leukocyte (PMN), myeloid-derived suppressor cell (MDSC), mast cell, basophil, eosinophils, megakaryocyte, neutrophil, dendritic cell, monocyte, and/or a macrophage.
The cell population can comprise at least 104, at least 105, at least 106, at least 107, at least 108, at least 109, at least 1010, at least 1011, at least 1012, at least 1013, at least 1014, at least 1015, at least 1016, at least 1017, at least 1018, at least 1019, at least IO20, or more than IO20 cells. In such instances, when the cell population comprises at least 104, at least 105, at least 106, at least 107, at least 108, at least 109, at least IO10, at least 1011, at least 1012, at least 1013, at least 1014, at least 1015, at least 1016, at least 1017, at least 1018, at least 1019, at least IO20, or more than IO20 cells, wherein said population comprises a plurality of myeloid-derived cells and/or myeloid-derived CAR cells, and wherein: a. no more than 1%, no more than 0.9%, no more than 0.8%, no more than 0.7%, no more than 0.6%, no more than 0.5%, no more than 0.4%, no more than 0.3%, no more than 0.2%, no more than 0.1%, or less than 0.1% of said population of cells is CD34+; b. at least 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95% of said population is CD15+; c. between about 50%-95% of said population of cells is CD15+; d. least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of said population is CD1 lb+; e. between about 75%-99% of said population of cells is CD1 lb+; f. at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, at least 50%, at least 60%, at least 70%, at least 80%, or at least 90% of said
population of cells is CD14+; g. between about 5%-90% of said population of cells is CD14+; h. at least 85%, at least 90%, at least 95%, or at least 99% of said population is CD13+; and/or i. between about 85%-99% of said population of cells is CD13+. In such instances, the myeloid- derived cell and/or myeloid-derived CAR cell can be a common myeloid progenitor (CMP), granulo-monocyte progenitor, monocyte-dendritic cell progenitor, common dendritic cell progenitor, myeoblast, monoblast, pre-conventional dendritic cell, promocyte, granulocyte, polymorphonuclear leukocyte (PMN), myeloid-derived suppressor cell (MDSC), mast cell, basophil, eosinophils, megakaryocyte, neutrophil, dendritic cell, monocyte, and/or a macrophage.
The cell population can comprise at least 104, at least 105, at least 106, at least 107, at least 108, at least 109, at least IO10, at least 1011, at least 1012, at least 1013, at least 1014, at least 1015, at least 1016, at least 1017, at least 1018, at least 1019, at least IO20, or more than IO20 cells. In such instances, when the cell population comprises at least 104, at least 105, at least 106, at least 107, at least 108, at least 109, at least IO10, at least 1011, at least 1012, at least 1013, at least 1014, at least 1015, at least 1016, at least 1017, at least 1018, at least 1019, at least IO20, or more than IO20 cells, wherein said population comprises a plurality of myeloid-derived CAR cells, and wherein: a. no more than 1%, no more than 0.9%, no more than 0.8%, no more than 0.7%, no more than 0.6%, no more than 0.5%, no more than 0.4%, no more than 0.3%, no more than 0.2%, no more than 0.1%, or less than 0.1% of said population of cells is CD34+; b. at least 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95% of said population is CD15+; c. between about 50%-95% of said population of cells is CD15+; d. least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of said population is CD1 lb+; e. between about 75%-99% of said population of cells is CD1 lb+; f. at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, at least 50%, at least 60%, at least 70%, at least 80%, or at least 90% of said population of cells is CD14+; g. between about 5%-90% of said population of cells is CD14+; h. at least 85%, at least 90%, at least 95%, or at least 99% of said population is CD13+; and/or i. between about 85%-99% of said population of cells is CD13+, wherein the myeloid-derived cell and/or myeloid-derived CAR cell is a common myeloid progenitor (CMP), granulo-monocyte progenitor, monocyte-dendritic cell progenitor, common dendritic cell progenitor, myeoblast, monoblast, pre-conventional dendritic cell, promocyte, granulocyte, polymorphonuclear leukocyte (PMN), myeloid-derived suppressor cell (MDSC), mast cell, basophil, eosinophils, megakaryocyte, neutrophil, dendritic cell, monocyte, and/or a macrophage.
In some embodiments, disclosed herein is a cell population wherein said population is a mixed population comprising plurality of myeloid-derived cells, or a plurality of myeloid-derived CAR cells, wherein: a. no more than 5%, no more than 4%, no more than 3%, no more than 2%, no
more than 1%, no more than 0.5%, or no more than 0.1% of cells within said population are CD34+; b. at least 50%, at least 55%, at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 86%, at least 87%, at least 88%, at least 89%, or at least 90% of cells within said population are CD14+; c. at least 70%, at least 80%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or at least 99.5%% of cells within said population are CD1 lb+; d. at least 90%, at least 91%, at least 92%, at least 94%, or at least 95% of cells within said population are CD13+; and/or e. at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, or at least 95% of cells within said population are CD15+. In such instances, the myeloid- derived cell and/or myeloid-derived CAR cell can be a common myeloid progenitor (CMP), granulo-monocyte progenitor, monocyte-dendritic cell progenitor, common dendritic cell progenitor, myeoblast, monoblast, pre-conventional dendritic cell, promocyte, granulocyte, polymorphonuclear leukocyte (PMN), myeloid-derived suppressor cell (MDSC), mast cell, basophil, eosinophils, megakaryocyte, neutrophil, dendritic cell, monocyte, and/or a macrophage. In such instances, the precursor cell can be a hematopoietic stem cell (HSC).
In some embodiments, disclosed herein is a cell population wherein said population is a mixed population comprising plurality of myeloid-derived cells and/or myeloid-derived CAR cells, wherein: a. no more than 5%, no more than 4%, no more than 3%, no more than 2%, no more than 1%, no more than 0.5%, or no more than 0.1% of cells within said population are CD34+; b. at least 50%, at least 55%, at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 86%, at least 87%, at least 88%, at least 89%, or at least 90% of cells within said population are CD14+; c. at least 70%, at least 80%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or at least 99.5%% of cells within said population are CD1 lb+; d. at least 90%, at least 91%, at least 92%, at least 94%, or at least 95% of cells within said population are CD13+; and/or e. at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, or at least 95% of cells within said population are CD15+. In such instances, the myeloid-derived cell and/or myeloid-derived CAR cell can be a common myeloid progenitor (CMP), granulo-monocyte progenitor, monocyte-dendritic cell progenitor, common dendritic cell progenitor, myeoblast, monoblast, pre-conventional dendritic cell, promocyte, granulocyte, polymorphonuclear leukocyte (PMN), myeloid-derived suppressor cell (MDSC), mast cell, basophil, eosinophils, megakaryocyte, neutrophil, dendritic cell, monocyte, and/or a macrophage. In such instances, the precursor cell can be a hematopoietic stem cell (HSC).
Any cell population described herein can comprise at least 104, at least 105, at least 106, at least 107, at least 108, at least 109, at least 1010, at least 1011, at least 1012, at least 1013, at least 1014, at least 1015, at least 1016, at least 1017, at least 1018, at least 1019, at least IO20, or more than IO20
cells. In such instances, when the cell population comprises at least 104, at least 105, at least 106, at least 107, at least 108, at least 109, at least IO10, at least 1011, at least 1012, at least 1013, at least 1014, at least 1015, at least 1016, at least 1017, at least 1018, at least 1019, at least IO20, or more than IO20 cells, wherein said population comprises a plurality of myeloid-derived cells and/or myeloid- derived CAR cells, wherein: a. no more than 5%, no more than 4%, no more than 3%, no more than 2%, no more than 1%, no more than 0.5%, or no more than 0.1% of cells within said population are CD34+; b. at least 50%, at least 55%, at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 86%, at least 87%, at least 88%, at least 89%, or at least 90% of cells within said population are CD14+; c. at least 70%, at least 80%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or at least 99.5%% of cells within said population are CD1 lb+; d. at least 90%, at least 91%, at least 92%, at least 94%, or at least 95% of cells within said population are CD13+; and/or e. at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, or at least 95% of cells within said population are CD15+, wherein the myeloid-derived cell and/or myeloid-derived CAR cell is a common myeloid progenitor (CMP), granulo-monocyte progenitor, monocyte-dendritic cell progenitor, common dendritic cell progenitor, myeoblast, monoblast, pre-conventional dendritic cell, promocyte, granulocyte, polymorphonuclear leukocyte (PMN), myeloid-derived suppressor cell (MDSC), mast cell, basophil, eosinophils, megakaryocyte, neutrophil, dendritic cell, monocyte, and/or a macrophage.
It is envisaged that the methods disclosed herein can effectively generate a myeloid-derived cell from a precursor cell, such as a hematopoietic stem cell, wherein said precursor cell either does or does not express a CAR. For instance, the presence of a CAR expressed in and on the surface of a precursor cell is not envisaged as impacting the expansion and/or differentiation of said precursor cell and/or said precursor CAR cell, as described herein. As such, it should be understood that any myeloid-derived cell(s) and/or populations comprising a plurality of myeloid-derived cell(s) disclosed herein are intended to encompass myeloid-derived cells that: a. comprise a CAR (i.e., myeloid-derived CAR cells); or b. do not comprise a CAR. Any of the cell populations described herein can be generated wherein the myeloid-derived cells do not comprise a CAR.
V. Methods of Manufacturing Myeloid-Derived Cells and Genetically modified Myeloid- Derived CAR Cells
The present disclosure provides myeloid-derived cells that are generated from a precursor cell using the methods and compositions disclosed herein. The precursor cell can be a stem cell, such as a hematopoietic stem cell, and/or a CD34+ cell. The stem cell can be isolated from a subject. In such instances, the subject can be a subject in need of a treatment, for which administration of the myeloid-derived cells that are generated from said isolated stem cells can be
used as a treatment or a manufacture of a medicament for a treatment. Also disclosed herein are cell populations comprising a plurality of the myeloid-derived cells generated from HSCs. The myeloid-derived cells, and populations comprising the same, disclosed herein do not require the presence of a CAR to effectively generate a myeloid-derived cell.
In some aspects, the method of manufacturing a myeloid-derived cell disclosed herein comprises the step of contacting a precursor cell with a precursor media comprising a precursor cytokine mixture, whereby the number of precursor cell(s) increases to generate a cell population comprising a plurality of precursor cells, wherein said precursor cell is a stem cell, , wherein the method further comprises the step of contacting said cell population comprising a plurality of precursor cells with a myeloid media comprising a myeloid cytokine mixture, whereby, said precursor cell(s) differentiates into a myeloid-lineage cell to generate a myeloid-derived cell and/or a mixed cell population comprising a plurality of myeloid-derived cells and/or a plurality of precursor cells. In such instances, the myeloid-derived cell can be any cell within the myeloid lineage. A skilled artisan would understand that myeloid lineage cells (i.e, myeloid cells) can be identified by cell surface marker expression. Known cell surface markers associated with myeloid cells are known in the art. In some instances of the methods disclosed herein, the myeloid-derived cell is CD14+, CDl lb+, CD13+, and/or CD15+.
The presence of a CAR is not envisaged as impacting the ability of the methods disclosed herein to effectively generate a myeloid-derived cell from a hematopoietic stem cell. Accordingly, also disclosed herein are myeloid-derived cells genetically modified to express a CAR. In such instances, it is envisaged that the CAR is introduced into the precursor cell prior to expansion and differentiation of said precursor CAR cell. Once a precursor CAR cell is generated, said cell can be contacted with, and maintained in the precursor media and, optionally, the myeloid media, disclosed herein to generated a myeloid-derived CAR cell. The compositions and methods disclosed herein provide for a more stable process for generating a myeloid-derived CAR cell. In particular, the precursor CAR cell maintains the CAR throughout the expansion and differentiation process, providing a more efficient and effective process for generating myeloid-derived CAR cells compared to known methods.
Disclosed herein are methods of manufacturing a myeloid-derived CAR cell from a hematopoietic stem cell comprising a chimeric antigen receptor (CAR). In some aspects, the method of manufacturing a myeloid-derived cell disclosed herein comprises the step of contacting a precursor cell, such as an HSC, with a nucleic acid molecule comprising a polynucleotide sequence encoding a chimeric antigen receptor (CAR), whereby said nucleic acid is introduced into said precursor cell to generate a precursor CAR cell comprising a chimeric antigen receptor (CAR),
wherein the method further comprises the step of contacting the precursor CAR cell with a precursor media comprising a precursor cytokine mixture, whereby the number of precursor CAR cell(s) increases to generate a cell population comprising a plurality of precursor CAR cells, wherein said precursor CAR cell is a stem cell. In such instances, the method can further comprise the step of contacting said cell population comprising a plurality of precursor CAR cells with a myeloid media comprising a myeloid cytokine mixture, whereby, said precursor CAR cell(s) differentiates into a myeloid-lineage cell to generate a myeloid-derived CAR cell and/or a mixed cell population comprising a plurality of myeloid-derived CAR cells and/or a plurality of precursor CAR cells. In such instances, the myeloid-derived CAR cell can be any cell within the myeloid lineage. In some instances of the methods disclosed herein, the myeloid-derived CAR cell is CD14+, CDl lb+, CD13+, and/or CD15+.
Disclosed herein are methods of manufacturing a genetically modified myeloid-derived cell comprising a chimeric antigen receptor (CAR), comprising: (a) contacting a precursor with a nucleic acid molecule comprising a polynucleotide sequence encoding the CAR to generate a genetically modified precursor CAR cell; and (b) expanding said genetically modified precursor CAR cell in a precursor media comprising a precursor cytokine mixture to generate a population comprising a plurality of genetically modified precursor CAR cells, wherein the precursor comprises a bone marrow-derived cell or a stem cell. The methods can further comprise differentiating said genetically modified precursor CAR cell(s) in a myeloid media comprising a myeloid cytokine mixture to generate a myeloid-derived CAR cell or a population comprising a plurality of genetically modified myeloid-derived CAR cells.
When the method comprises differentiating said genetically modified precursor CAR cell(s), prior to the differentiation step, the precursor media can be removed prior to addition of the myeloid media.
The precursor (e.g., stem cells) can be isolated from a human subject.
A. Precursor
The methods disclosed herein can be used to manufacture a genetically modified myeloid- derived cell from a precursor. The term “precursor” when used in connection with the myeloid- derived cells indicate parent cells in a cellular lineage resulting in any cell within the myeloid lineage herein described. Exemplary precursor cells include bone marrow-derived cells, stem cells, and other precursor cells identifiable by a person skilled in the art. Precursor cells can also indicate cells that have the potential to differentiate resulting in any cell within the myeloid lineage herein described. Exemplary precursor cells can include bone marrow-derived cells, stem cells, and other precursor cells identifiable by a person skilled in the art. In some instances, the precursor is isolated
from a source selected from one of the following: bone marrow, mobilized peripheral blood, peripheral blood, and umbilical cord blood. In some instances, the precursor is isolated from a source selected from one of the following: bone marrow, mobilized peripheral blood, peripheral blood, and umbilical cord blood. In some instances, the stem cell is isolated from a source selected from one of the following: bone marrow, mobilized peripheral blood, peripheral blood, and umbilical cord blood. The bone marrow-derived cell or stem cell used for the disclosed method can be isolated from a subject, such as a human subject. In some instances, when the bone marrow- derived cell or stem cell is isolated from a subject, the cell is isolated from peripheral blood. In some instances, the peripheral blood is mobilized peripheral blood. In some instances, the peripheral blood is mobilized with a cytokine. In some embodiments, wherein the peripheral blood is mobilized with at least one cytokine, the at least one cytokine comprises granulocyte-colony stimulating factor (G-CSF) or granulocyte-macrophage colony stimulating factor (GM-CSF), or a combination thereof.
Any method of isolating and identifying a HSC (or population comprising the same) is envisaged within the scope of the present disclosure. A skilled artisan would understand how to isolate and identify a HSC for use in the disclosed method. The method disclosed herein comprises expanding and differentiation a precursor cell into a genetically modified myeloid-derived CAR cell. The precursor cell disclosed herein comprises a bone marrow-derived cell or a stem cell. The precursor cell can be a stem cell, wherein the stem cell is a pluripotent stem cell, such as a hematoiepoietic stem cell (HSC). In a preferred embodiment, the stem cell is a hematoiepoietic stem cell (HSC). Prior to being expanded in a precursor media, the stem cells are genetically modified with a CAR. Genetic modification with a CAR comprises contacting the precursor cell with a nucleic acid molecule comprising a polynucleotide sequence encoding the CAR.
Myeloid-Derived Cell
Also disclosed herein are genetically modified HSCs and myeloid-derived cells produced by the described methods. A “myeloid-derived” cell is a differentiated descendant from common progenitors derived from hematopoietic stem cells in, or isolated from, tissue such as the bone marrow or other source of blood tissue. For example, a myeloid-derived cell can be any cell within the myeloid lineage, for example, a common myeloid progenitor (CMP) cell, or any cell derived therefrom, such as any granulocyte or monocyte cell. As the CAR is introduced into a bone marrow derived, or stem cell (i.e., precursor cell), prior to expansion and differentiation of the precursor cell, the myeloid-derived cells described herein are genetically modified to express a CAR (i.e., genetically modified myeloid-derived CAR cell). The genetically modified myeloid-derived cells can include any cell within the myeloid lineage that has been derived from a precursor as described
herein. The myeloid-derived cell can include, for example, phagocytic cells, such as a macrophage, monocyte, dendritic cell, neutrophil, and/or precursor thereof within the myeloid lineage. In some instances, the genetically modified myeloid-derived cell is a phagocytic cell.
A “precursor” as described herein comprises a bone marrow-derived cell or a stem cell. For example, the precursor can be a pluripotent stem cell, such as a hematopoietic stem cell (HSC). A “stem cell” would be understood to be a cell that can self-renew and a cell from which other types of cells develop. A stem cell can be a hematopoietic stem cell. A hematopoietic stem cell would be understood to be an immature cell that can develop into various types of blood cells, including, for example, white blood cells, red blood cells, and platelets. Hematopoietic stem cells can be found in the peripheral blood and the bone marrow. The hematopoietic stem cell (HSC) is a multipotent stem cell that resides in tissues such as cord blood, peripheral blood, and the bone marrow. Hematopoietic stem cells (HSCs) lay the foundation of hematopoiesis to generate all functional hematopoietic lineages, including erythrocytes, leukocytes (neutrophils, basophils, eosinophils, lymphocytes, monocytes and macrophages) and platelets. HSCs can self-replicate and differentiate into progeny of multiple lineages. In general, the hematopoietic lineage is organized such that HSCs sit atop the hierarchy and give rise to committed progenitor cells, which in turn give rise to mature, differentiated cells. Mature HSCs generate multipotent progenitor (MPP) cells which have the potential to further differentiate into two major lineages: common myeloid progenitor (CMP) and common lymphoid progenitor (CLP). Downstream of MPPs are committed progenitors that are oligopotent and have limited ability to self-renew. As general principle, when cells progress through hematopoiesis they become more differentiated and more frequent in number. They also lose their capacity to self-renew, become more restricted in their differentiation potential, and gain expression of molecules required for functional specialization. Differentiation is thought to occur in one direction with restriction towards a particular lineage (e.g., myleoid or lymphoid), with no significant evidence of transdifferentiation between hematopoietic lineages under normal conditions.
Using the methods disclosed herein, a myeloid-derived cell can be generated from a precursor. In some instances, the precursor is a bone marrow-derived cell, a cord blood-derived cell, or a peripheral blood-derived cell. In some instances, the precursor is a stem cell. A stem cell can be a hematopoietic stem cell that has been isolated from a subject using any know method in the art. In some instances, the precursor is in a cell population comprising a plurality of precursors. In some embodiments, the population comprising the plurality of precursors is a homogenous cell population. The cell population can also be a heterogenous cell population. The cells or population of cells comprising a precursor of a macrophage, monocyte, dendritic cell, neutrophil can be
cultured for expansion after genetic modification to express a CAR. In some embodiments, the cells or population of cells comprising precursor cells are cultured for differentiation and expansion of macrophages, monocytes, dendritic cells, and/or neutrophils. The cells or population of cells comprising precursor cells, can include precursor CAR cells that are cultured for differentiation and expansion into any cell within the myeloid lineage. Examples of cells within the myeloid lineage can include parent cells of macrophages, monocytes, dendritic cells, and/or neutrophils, such as a myeloid lineage precursor cell. Examples of myeloid precursor cells include cells such as a myeloid progenitor cell, granulo-monocyte progenitor cell, monocyte-dendritic cell progenitor/monoblasts, promonocyte, or myeloblast. In some embodiments, the present disclosure comprises expanding a population of HSCs comprising a CAR, and expanding and differentiating them into myeloid progenitor cells, granulo-monocyte progenitor cells, monocyte-dendritic cell progenitor/monoblasts, promonocytes, myeloblasts, monocytes, macrophages, dendritic cells, and/or neutrophils, as described herein. In some embodiments, the genetically modified myeloid- derived cell is a myeloid progenitor cell. In some embodiments, the genetically modified myeloid- derived cell is a granulo-monocyte progenitor cell. In some embodiments, the genetically modified myeloid-derived cell is a monocyte-dendritic cell progenitor/monoblast. In some embodiments, the genetically modified myeloid-derived cell is a promonocyte. In some embodiments, the genetically modified myeloid-derived cell is a myeloblasts. In some embodiments, the genetically modified myeloid-derived cell is a monocyte. In some embodiments, the genetically modified myeloid- derived cell is a macrophage. In some embodiments, the genetically modified myeloid-derived cell is a dendritic cell. In some embodiments, the genetically modified myeloid-derived cell is a neutrophil.
Phagocytic cells
Phagocytic cells are used in the compositions and methods described herein. The genetically modified myeloid-derived cell can be a phagocytic cell. The terms “phagocytic cell” or “phagocyte” or plural forms thereof as used herein indicate a cell that is capable of phagocytosis, which is the process by which a cell uses its plasma membrane to engulf a large particle (>0.5 pm), giving rise to an internal compartment called the phagosome. Phagocytosis is one type of endocytosis as will be understood by a skilled person. Phagocytes of an individual typically use their plasma membrane to engulf and remove cellular debris, foreign substances, microbes, and cells to protect the body of an individual. Phagocytes in the sense of the disclosure can typically also perform trogocytosis, a process whereby one cell contacts and quickly nibbles another cell. For example, trogocytosis occurs when lymphocytes (e.g., B, T, NK cells) conjugated to antigen-presenting cells (APCs) extract surface molecules from the APCs and express them on their own surface. Exemplary phagocytic cells herein
described include macrophages, monocytes, neutrophils, dendritic cells and precursors thereof as a person skilled in the art would understand, though singled celled organisms such as Dictyostelium amoebae are also phagocytes. Phagocytic cells of the disclosure also include tissue-resident macrophages that not only sense and respond to invading pathogens but are important in tissue development, remodeling, and homeostasis. Tissue-resident macrophages originate from embryonic precursors and can self-renew. In some embodiments, tissue-resident macrophages can include Kupffer macrophages (forms the lining of sinusoids of the liver), alveolar macrophages (lung), microglia macrophages (brain), red pulp macrophages (spleen), and macrophages in the heart. In some embodiments, tissue-resident macrophages enter tissue during non-inflammatory conditions.
Macrophages are differentiated from monocytes and function to regulate both innate and adaptive immunity to combat foreign particles including pathogens by stimulating the response of immune cells, such as lymphocytes. The putative surface markers for macrophages are CD1 lb (Mac- 1), CD14 (ligand receptor of lipopolysaccharide), CD115 (colony-stimulating factor 1 receptor) and F4/80 (a highly glycosylated proteoglycan extracellular antigen) (Lim, W.F., Inoue-Yokoo, T., Tan, K.S. et al. Hematopoietic cell differentiation from embryonic and induced pluripotent stem cells. Stem Cell Res Ther 4, 71 (2013)).
A monocyte is a type of white blood cell of the immune system that is capable of phagocytosis. Macrophages are derived from blood monocytes that migrate into tissue. One of macrophages’ main functions is to phagocytose microbes and clear cellular debris. Macrophages also play an important role in both the initiation and resolution of inflammation. Macrophages comprise cells typically diffusely scattered in the connective tissue and in liver (Kupffer cells), spleen and lymph nodes (sinus histiocytes), lungs (alveolar macrophages), and central nervous system (microglia) as will be understood by a skilled person.
Macrophages can also display different responses, ranging from pro-inflammatory to antiinflammatory, depending on the type of stimuli they receive from the surrounding microenvironment. Ml and M2 are two major macrophage phenotypes that have been proposed to correlate with extreme macrophage responses. Ml pro-inflammatory macrophages are activated upon contact with certain molecules such as lipopolysaccharide (LPS), interferon-gamma (IFN-y), interleukin (IL)-ip, tumor necrosis factor (TNF)-a, and Toll-like receptor engagement. Ml macrophages constitute a potent arm of the immune system deployed to fight infections. They are capable of either direct (pathogen pattern recognition receptors) or indirect (Fc receptors, complement receptors) recognition of the pathogen. They are also armed in their ability to produce reactive oxygen species (ROS) as means to help kill pathogens. In addition, Ml macrophages secrete pro-inflammatory cytokines and chemokines attracting other types of immune cells and integrating/orchestrating the immune response. Ml
activation is induced by IFN-y, TNFa, granulocyte macrophage colony-stimulating factor (GM-CSF), LPS, and other toll-like receptors (TLR) ligands.
In contrast, M2 anti-inflammatory macrophages, also known as alternatively activated macrophages, are activated by anti-inflammatory molecules such as IL-4, IL- 13, and IL- 10. M2 macrophages exhibit immunomodulatory, tissue repair, and angiogenesis properties which allow them to recruit regulatory T cells to sites of inflammation. M2 macrophages do not constitute a uniform population and often are further subdivided into M2a, M2b and M2c categories. The common denominator of all three subpopulations is high IL- 10 production accompanied by low production of IL-12. One of their signatures is production of enzyme arginase-1 that depletes L-arginine, thereby suppressing T cell responses and depriving iNOS of its substrate.
The in vivo molecular mechanisms of macrophage polarization to Ml or M2 phenotype are poorly characterized because of the variety of signals macrophages experience in the cellular microenvironment. In recent years, progress has been made in identifying in vivo macrophage polarization under physiological conditions such as ontogenesis, pregnancy, and pathological conditions such as allergies, chronic inflammation, and cancer. In vitro macrophage polarization is plastic and macrophages, with the help of cytokines, can be polarized back and forth to either phenotype. IFN-y and IL-4 are two cytokines that can polarize macrophages to Ml and M2 phenotypes, respectively.
A monocyte is a type of leukocyte or white blood cell capable of phagocytosis and can differentiate into macrophages and myeloid lineage dendritic cells. As a part of the vertebrate innate immune system, monocytes also influence the process of adaptive immunity. There are at least three subclasses of monocytes in human blood based on their phenotypic receptors including CD14++ CD 16“ monocyte, CD14+CD16++ monocyte and CD14++CD16+ monocytes, as will be understood by a person skilled in the art. Monocytes serve as precursors for various tissue macrophage and dendritic cell populations and contribute to both protective and pathological immune responses.
Dendritic cells are specialized antigen-presenting cells capable of phagocytosis that have long outgrowths called dendrites that help to engulf microbes and other invaders. Dendritic cells are present in the tissues that are in contact with the external environment, mainly the skin, the inner lining of the nose, the lungs, the stomach, and the intestines. Once activated, they mature and migrate to the lymphoid tissues where they interact with T cells and B cells to initiate and orchestrate the adaptive immune response. Mature dendritic cells activate T helper cells and cytotoxic T cells. The activated helper T cells interact with macrophages and B cells to activate them in turn. In addition, dendritic cells can influence the type of immune response produced; when they travel to the lymphoid areas
where T cells are held, they can activate T cells, which then differentiate into cytotoxic T cells or helper T cells.
Neutrophils are phagocytes that form the most abundant type of granulocytes and the most abundant type of white blood cells in most mammals as known to a person skilled in the art. Neutrophils are formed from stem cells in the bone marrow and differentiated into subpopulations of neutrophil-killers and neutrophil-eagers.
A phagocytic cell includes a precursor cell that can develop into and/or be differentiated into a phagocyte. The terms “precursors” or “precursor cells” when used in connection with macrophages, monocytes, dendritic cells, and/or neutrophils indicate parent cells in a cellular lineage resulting in phagocytic cells herein described. Exemplary precursor cells include bone marrow-derived cells, stem cells, hematopoietic stem cells, pluripotent stem cells, and other precursor cells identifiable by a person skilled in the art. In some embodiments, precursor cells can be differentiated by culturing the precursor cells under conditions that promote development of phagocytic cells. For example, cytokines that are typically used for differentiation of bone-marrow derived monocytes or hematopoietic stem cells into macrophages include granulocyte-macrophage colony-stimulating factor (GM-CSF) and macrophage colony-stimulating factor (M-CSF). Macrophage differentiation protocols are described in, e.g., van Furth et al. (1972) Bulletin of the World Health Organization, 46(6), 845-852; Mosser and Edwards (2008) Nature Reviews Immunology, 8(12), 958-969; and Lutz et al. (1999) Journal of Immunological Methods, 223(1), 77-92.
A source of phagocytic cells, such as macrophages, monocytes, dendritic cells, neutrophils, and/or precursors thereof is obtained from a subject. The cells can be obtained from a number of sources, including peripheral blood mononuclear cells, bone marrow, lymph node tissue, spleen tissue, umbilical cord, and tumors. In some embodiments, any number of macrophage, monocyte, dendritic cell, neutrophil, or precursor cell lines available in the art, may be used. In certain embodiments, the cells can be obtained from a unit of blood collected from a subject using any number of techniques known to the skilled artisan, such as Ficoll separation.
Cells can be isolated from peripheral blood by lysing the red blood cells and depleting the lymphocytes and red blood cells, for example, by centrifugation through a PERCOLL™ gradient. Alternatively, cells can be isolated from umbilical cord.
Cells from the circulating blood of an individual can be obtained by apheresis or leukapheresis. The apheresis product typically contains lymphocytes, including T cells, monocytes, granulocytes, B cells, other nucleated white blood cells, red blood cells, and platelets. The cells collected by apheresis may be washed to remove the plasma fraction and to place the cells in an appropriate buffer or media, such as phosphate buffered saline (PBS) or a wash solution that lacks calcium and may lack
magnesium or may lack many if not all divalent cations, for subsequent processing steps. After washing, the cells may be resuspended in a variety of biocompatible buffers, such as, for example, Ca- free, Mg-free PBS. Alternatively, the undesirable components of the apheresis sample may be removed and the cells directly resuspended in culture media.
During isolation of a desired population of cells by positive or negative selection, the concentration of cells and surface (e.g., particles such as beads) can be varied. In some embodiments, it may be desirable to significantly decrease the volume in which beads and cells are mixed together (i.e., increase the concentration of cells), to ensure maximum contact of cells and beads. For example, a concentration of 2 billion cells/ml, 1 billion cells/ml, or greater than 100 million cells/ml can be used. In some embodiments, a concentration of cells of 10, 15, 20, 25, 30, 35, 40, 45, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, or 100 million cells/ml can be used. In some embodiments, concentrations of 125 to 150 million cells/ml can be used. The use of high concentrations of cells can result in increased cell yield, cell activation, and cell expansion.
A population of cells of the present disclosure can include macrophages, monocytes, dendritic cells, neutrophils, and/or precursors thereof.
B. Chimeric Antigen Receptor (CAR) Transduction
When the precursor is a stem cell, such as a hematopoietic stem cell, the stem cell can be contacted with a nucleic acid molecule comprising a polynucleotide sequence encoding the CAR to generate a genetically modified precursor CAR cell. Contacting enables the nucleic acid to be introduced into the precursor. Once introduced into the cell, the nucleic acid comprising a polynucleotide sequence encoding the CAR, can be stably expressed by the cell. In some instances, the precursor that is contacted with the nucleic acid is a hematopoietic stem cell that has been isolated from a human subject. The isolated HSCs can be frozen and preserved following isolation by any means known in the art, such as cryopreservation. The cryopreserved cells are then thawed prior to being contacted with a nucleic acid.
Prior to being contacted with a nucleic acid comprising a polynucleotide encoding a CAR, the HSC can be within an isolated cell population, such as a population of cells isolated from a subject. The isolated cell population can be freshly isolated or can be an isolated population that was previously isolated and has been frozen to preserve the viability of the cells, such as cryopreserved. The isolated population can comprise a plurality of HSCs wherein at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or more than 99% CD34+ cells. Upon thawing, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or more than 99% of viable recovered cells are CD34+ cells.
Contacting a precursor with a nucleic acid comprising a polynucleotide encoding a CAR can result in the introduction of the polynucleotide encoding a CAR into the precursor cell(s). The nucleic acid can be contacted and introduced comprises introducing a viral vector comprising the polynucleotide into the precursor. The viral vector can be a lentiviral vector, a retroviral vector, or an adenoviral vector.
In some instances, contacting the precursor with a nucleic acid comprises introducing the nucleic acid into the precursor by transposition or mRNA transfection. In some instances, contacting the precursor with a nucleic acid comprises electroporating the nucleic acid into the precursor.
In some instances, the nucleic acid can be contacted and introduced into the precursor or plurality of precursor cells via transduction. Following transduction, at least 40%, at least 50%, at least 60%, at least 70%, at least 80%, at least 90%, or at least 95% of said plurality of genetically modified precursor CAR cells or said plurality of myeloid-derived CAR cells expresses said CAR on the cell surface. Expression of the CAR in the precursor cell(s) can be analyzed at any point in time following transduction. Following CAR transduction, the precursor cell(s) can be expanded. Expansion of the precursor can occur at any point in time that maximizes the recovery of genetically modified CAR cells. For example, expansion can occur 1 day, 2 days, 3 days, 4 days, or 5 days following transduction. Expansion can also occur is a shorter amount of time following transduction, such as 2 hours, 4 hours, 6 hours, 8 hours, 10 hours, 12 hours, 14 hours, 16 hours, 18 hours, or 20 hours following CAR transduction. In some instances, contacting a precursor with a nucleic acid molecule comprising a polynucleotide sequence encoding the CAR occurs within a precursor media described herein. In some instances, the precursor media comprises a precursor cytokine mixture. In some instances, the precursor media do not comprise a precursor cytokine mixture as disclosed herein, when contacting a precursor with a nucleic acid molecule comprising a polynucleotide sequence encoding the CAR occurs within a precursor media, the precursor media does not comprise a precursor cytokine mixture.
Following the step of contacting the precursor cell with the nucleic acid in precursor media, the precursor media can be removed. In some instances, precursor media is removed using a centrifugation process. In some instances, multiple rounds of washing and centrifugation are utilized to remove the precursor media following CAR transduction. When a viral vector is used to introduce a CAR into the precursor cell, multiple rounds of washing and centrifugation can be utilized to remove the viral vector and precursor media. Following removal of precursor media, the genetically modified precursor CAR cells, which have been removed from contact with precursor
media, are suspended in precursor media comprising a precursor cytokine mixture to expand the cells.
The precursor is expanded in a precursor media comprising a precursor cytokine mixture as disclosed herein. The precursor can be expanded to generate a population wherein at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of said plurality of genetically modified myeloid-derived CAR cells expressing said CAR are CD1 lb+ or CD14+ or Cl lb+CD14+.
A vector may be used to introduce a CAR described herein into a precursor cell (e.g., a hematopoietic stem cell or other bone marrow-derived cell). The present disclosure provides a vector comprising a nucleic acid sequence encoding a CAR as described herein. In some embodiments, the vector comprises a plasmid vector, viral vector, retrotransposon (e.g. piggyback, sleeping beauty), site directed insertion vector (e.g. CRISPR, Zn finger nucleases, TALEN), suicide expression vector, or other known vector in the art.
The expression of natural or synthetic nucleic acids is typically achieved by operably linking a nucleic acid or portions thereof to a promoter, and incorporating the construct into an expression vector. The vector is one generally capable of replication in a mammalian cell, and/or also capable of integration into the cellular genome of the mammal. Typical vectors contain transcription and translation terminators, initiation sequences, and promoters useful for regulation of the expression of the desired nucleic acid sequence. The nucleic acid can be cloned into any number of different types of vectors. For example, the nucleic acid can be cloned into a vector including, but not limited to a plasmid, a phagemid, a phage derivative, an animal virus, and a cosmid. Vectors of particular interest include expression vectors, replication vectors, probe generation vectors, and sequencing vectors. In some embodiments, a vector is used as a gene delivery vehicle to transfer a gene into a cell. Expression vectors can also include gene delivery nanomaterial such as polymeric nanoparticles or liposomes, and others identifiable by a person skilled in the art.
The expression vector may be provided to a cell in the form of a viral vector. Viral vector technology is well known in the art and is described, for example, in Sambrook et al., 2012, MOLECULAR CLONING: A LABORATORY MANUAL, volumes 1 -4, Cold Spring Harbor Press, NY), and in other virology and molecular biology manuals. Viruses, which are useful as vectors include, but are not limited to, retroviruses, adenoviruses, adeno- associated viruses, herpes viruses, and lentiviruses. Vectors, including those derived from retroviruses such as lentivirus, are suitable tools to achieve long-term gene transfer since they allow long-term, stable integration of a transgene and its propagation in daughter cells. The term “lentiviral vector” refers to a vector derived from at
least a portion of a lentivirus genome, including especially a self-inactivating lentiviral vector as described in, e.g., Milone et al. (2009) Molecular therapy 17(8): 1453-1464. Other examples of lentivirus vectors include the LENTIVECTOR® gene delivery technology from Oxford BioMedica, the LENTIMAX™ vector system from Lentigen and the like. Lentiviral vectors have the added advantage over vectors derived from onco-retroviruses, such as murine leukemia viruses, in that they can transduce non-proliferating cells, such as hepatocytes. They also have the added advantage of resulting in low immunogenicity in the subject into which they are introduced.
In some embodiments, adeno-associated viral vectors (AAV) can be used to deliver a nucleic acid (e.g., encoding a CAR of the disclosure) to a stem cell. AAVs are nonenveloped, single-stranded DNA viruses of the Dependoparvovirus genus of the Parvoviridae family. AAVs are innately nonpathogenic, poorly immunogenic, and broadly tropic, making them attractive gene delivery candidates. AAV vectors have shown to stably transfect mammalian cells without integration into the target genome. Exemplary suitable AAVs comprise AAVs of various serotypes that can be used as vectors for carrying genes. AAV serotypes are identified based on their interacting glycan moieties that mediate the initial attachment of AAVs to the cell surface. Examples of AAV serotypes include AAV serotype 1 (“AAV1”), AAV2, AAV3, AAV5, AAV6, AAV9 and other serotypes identifiable to a person skilled in the art such as AAV7, AAV8, AAV11, and AAV-DJ.
In general, a suitable vector contains an origin of replication functional in at least one organism, a promoter sequence, convenient restriction endonuclease sites, and one or more selectable markers, (e.g., WO 01/96584; WO 01/29058; and U.S. Pat. No. 6,326,193). Additional regulatory regions, e.g., enhancers, regulate the frequency of transcriptional initiation. Typically, these are located in the region 30-110 bp upstream of the start site, although a number of promoters have recently been shown to contain functional elements downstream of the start site as well. The spacing between promoter elements frequently is flexible, so that promoter function is preserved when elements are inverted or moved relative to one another. The term “regulatory sequence” or “regulatory regions” as described herein indicate a segment of a nucleic acid molecule which is capable of increasing or decreasing transcription or translation of a gene within an organism either in vitro or in vivo. Regulatory regions of a gene herein described comprise promoters, transcription factor binding sites, binding site operators, activator binding sites, protein-protein binding domains, RNA binding domains, DNA binding domains, repressors, enhancers, insulators, silencers and additional regulatory regions that can alter gene expression in response to developmental and/or external stimuli as will be recognized by a person skilled in the art. In some embodiments, nucleic acid molecules of the present disclosure includes phagocyte regulatory regions that control expression of a gene in a phagocyte.
A phagocyte promoter includes a nucleotide sequence that drives or regulates expression in phagocytes. Promoters specific to the mononuclear phagocyte system (MPS) including macrophages, neutrophils, dendritic cells, and osteoclasts will constitute phagocyte promoters. Examples of such promoters include CSF-1 promoter, CD68, CDl lc, DC-SIGN, DC-STAMP, langerin, human neutrophil elastase, and any synthetic promoter containing elements of the phagocyte system designed to achieve high level of expression in phagocytic cells.
A constitutive promoter includes an unregulated promoter that allows for continual transcription of its associated genes. An example of a strong constitutive promoter is the immediate early cytomegalovirus (CMV) promoter sequence. This promoter is capable of driving high levels of expression of any polynucleotide sequence operatively linked thereto. However, other constitutive promoter sequences may also be used, including a simian virus 40 (SV40) early promoter, a mouse mammary tumor virus (MMTV) promoter, a human immunodeficiency virus (HIV) long terminal repeat (LTR) promoter, a MoMuLV promoter, an avian leukemia virus promoter, an Epstein-Barr virus immediate early promoter, a Rous sarcoma virus promoter, an elongation factor- la promoter, PGK1 promoter from a phosphoglycerate kinase gene, as well as human gene promoters including an actin promoter, a ubiquitin promoter, a myosin promoter, a hemoglobin promoter, and a creatine kinase promoter. Other constitutive promoters are identifiable to one of skill in the art.
Further, the use of conditional or inducible promoters can also be suitable in the present disclosure. A conditional or inducible promoter includes a promoter with activity regulatable or controlled by endogenous transcription factors or exogenous inputs such as chemical compounds, thermal inducers, or optical induction. Examples of inducible promoters include a metallothionine promoter, a glucocorticoid promoter, a progesterone promoter, a tetracycline promoter, and a Lac promoter.
To assess expression of a polypeptide or portions thereof, the expression vector to be introduced into a precursor cell or phagocytic cell can also contain either a selectable marker gene or a reporter gene or both to facilitate identification and selection of cells expressing a nucleic acid sequence of interest (e.g., a CAR described herein) from the population of phagocytic cells transfected or infected through viral vectors. The selectable marker may be carried on a separate piece of DNA and used in a co-transfection procedure. Both selectable marker genes and reporter genes may be flanked with appropriate regulatory sequences to enable expression in the host cells. Useful selectable markers include, for example, antibiotic-resistance genes, such as neomycin and the like. Reporter genes are used for identifying potentially transfected cells and for evaluating the functionality of regulatory sequences. In general, a reporter gene is a gene that is not present in or expressed by the recipient organism or tissue and that encodes a polypeptide whose expression is manifested by some
easily detectable property, e.g., enzymatic activity. Expression of the reporter gene is assessed at a suitable time after the DNA has been introduced into the recipient cells. Suitable reporter genes may include genes encoding luciferase, beta-galactosidase, chloramphenicol acetyl transferase, secreted alkaline phosphatase, or the green fluorescent protein gene (e.g., Ui-Tei et al., 2000 FEBS Letters 479: 79-82). Suitable expression systems are well known and may be prepared using known techniques or obtained commercially. In general, the construct with the minimal 5' flanking region showing the highest level of expression of reporter gene is identified as the promoter. Such promoter regions may be linked to a reporter gene and used to evaluate agents for the ability to modulate promoter-driven transcription.
In some embodiments, an expression vector of the disclosure comprises a gene encoding a CAR as described herein and appropriate regulatory elements such as promoters, enhancers, and post- transcriptional and post-translational regulatory sequences that are compatible with the phagocytic cell expressing the gene encoding a CAR as would be understood by a skilled person. In some embodiments, the CAR expression vector is configured for genomic insertion allowing long-term overexpression of the CAR in a precursor cell (e.g., a hematopoietic stem cell or bone marrow-derived cell) . The genomic insertion can be achieved by stable transfection. In some embodiments, lentiviral transduction is preferred over physical or chemical transfections or adenoviral transduction. In some embodiments, lentiviral transduction is expected to be used effectively in vivo for delivery of a gene encoding a CAR of the disclosure and will permit stable expression in dividing and non-dividing cells.
A heterologous nucleic acid comprising a polynucleotide sequence encoding a CAR of the disclosure can be introduced into the genome of a phagocytic cell using transposases or targeted nucleases (e.g., Zinc finger nucleases (ZFN), meganucleases, or transcription activator-like effector (TALE) nucleases (TALENs), clustered regularly-interspaced short palindromic repeats associated nucleases (CRISPR/Cas)).
The CRISPR/Cas system is a genome editing tool discovered in prokaryotic cells. When utilized for genome editing, the system includes a CRISPR associated (Cas) protein (e.g., Cas9) capable of modify nucleic acids when complexed with a guide RNA, a guide RNA comprising a CRISPR RNA (crRNA) that binds and directs the Cas protein to the target nucleic acid, optionally a trans-activating CRISPR RNA (tracrRNA, that hybridizes to the crRNA and forms an active complex with the Cas protein), and optionally a homology dependent repair (HDR) template that guides the cellular repair process, allowing insertion of a specific nucleic acid sequence. In embodiments of the disclosure where a CRISPR/Cas system is used to introduce a CAR into a phagocytic cell, an HDR template comprising a polynucleotide sequence encoding a CAR of the disclosure is included. A CRISPR/Cas system is typically transfected into a target cell (e.g., a phagocytic cell to be genetically
modified) by means of one or more plasmids. CRISPR/Cas systems are described in, e.g., US8697359, US8771945, US8795965, US8865406, US8871445, US8889356, US8889418, US8895308, US8906616, US8932814, US8945839, US8993233 and US8999641 and applications related thereto; and WO2014/018423, WO2014/093595, WO2014/093622, WO2014/093635, WO2014/093655, WO20 14/093661, WO2014/093694, WO2014/093701, WO2014/093709, WO2014/093712, WO2014/093718, WO2014/145599, WO2014/204723, WO2014/204724, WO2014/204725, WO20 14/204726, WO2014/204727, WO2014/204728, WO2014/204729, WO2015/065964, WO2015/089351, WO2015/089354, WO2015/089364, WO2015/089419, WO2015/089427, WO20 15/089462, WO2015/089465, WO2015/089473 and WO2015/089486, W02016205711, WO2017/106657, WO2017/127807 and applications related thereto.
A ZFN is an artificial restriction enzyme which is generated by combining a zinc finger DNA- binding domain with a DNA-cleavage domain. A zinc finger domain can be engineered to target specific DNA sequences which allows a zinc-finger nuclease to target desired sequences within genomes. The DNA-binding domains of individual ZFNs typically contain a plurality of individual zinc finger repeats and can each recognize a plurality of base pairs. The most common method to generate new zinc-finger domains is to combine smaller zinc-finger "modules" of known specificity. The most common cleavage domain in ZFNs is the non-specific cleavage domain from the type II restriction endonuclease Fokl. Using the endogenous homologous recombination (HR) machinery and a DNA template comprising a CAR expression cassette and sequences homologous to a target nucleic acid sequence in a cell (e.g., a phagocytic cell), ZFNs can be used to insert the CAR expression cassette into the cell genome. When the target nucleic acid sequence is cleaved by ZFNs, the HR machinery searches for homology between the damaged chromosome and the DNA template, and then copies the sequence of the template between the two broken ends of the chromosome, whereby the DNA template is integrated at the target nucleic acid sequence. ZFNs are described in, e.g., US 6,534,261; US 6,607,882; US 6,746,838; US 6,794,136; US 6,824,978; 6,866,997; US 6,933,113; 6,979,539; US 7,013,219; US 7,030,215; US 7,220,719; US 7,241,573; US 7,241,574; US 7,585,849; US 7,595,376; US 6,903,185; US 6,479,626; US 2003/0232410 and US 2009/0203140 as well as Gaj et al., Nat Methods, 2012, 9(8):805-7; Ramirez et al., Nucl Acids Res, 2012, 40(12):5560-8; Kim et al., Genome Res, 2012, 22(7): 1327-33; Urnov etal., Nature Reviews Genetics, 2010, 11 :636-646; Miller, et al. Nature biotechnology 25, 778-785 (2007); Bibikova, et al. Science 300, 764 (2003); Bibikova, et al. Genetics 161, 1169-1175 (2002); Wolfe, et al. Annual review of biophysics and biomolecular structure 29, 183-212 (2000); Kim, et al. Proceedings of the National Academy of Sciences of the United States of America 93, 1156-1160 (1996); and Miller, et al. The EMBO journal 4, 1609-1614 (1985).
TALENs are restriction enzymes that can be engineered to cut specific sequences of DNA. TALEN system operates on almost the same principle as ZFNs. They are generated by combining a TALE DNA-binding domain with a DNA cleavage domain. TALEs are composed of 33-34 amino acid repeating motifs with two variable positions that have a strong recognition for specific nucleotides. By assembling arrays of these TALEs, the TALE DNA-binding domain can be engineered to bind desired DNA sequence, and thereby guide the nuclease to cut at specific locations in genome. TALENs are described in, e.g., US 8,440,431; US 8,440,432; US 8,450,471; US 8,586,363; and US 8,697,853; as well as Joung and Sander, Nat Rev Mol Cell Biol, 2013, 14(1) :49-55 ; Beurdeley et al., Nat Commun, 2013, 4: 1762; Scharenberg et al., Curr Gene Ther, 2013, 13(4):291-303; Gaj et al., Nat Methods, 2012, 9(8):805-7; Miller, et al. Nature biotechnology 29, 143-148 (2011); Christian, et al. Genetics 186, 757-761 (2010); Boch, et al. Science 326, 1509-1512 (2009); and Moscou, & Bogdanove, Science 326, 1501 (2009).
Meganucleases are rare-cutting endonucleases that generate double-strand breaks (DSB) in a nucleic acid and recognize sequences larger than 12 base pairs. In the wild, such endonucleases are essentially represented by homing endonucleases (Chevalier and Stoddard (2001) Nucleic Acids Research 29:3757-3774). Homing endonucleases are found in fungi, algae, eubacteria and archae, and are often encoded in mobile genetic elements. Their cleavage activities initiate the spreading of these mobile elements by homologous recombination. The biology of HO, I-Scel, and I-Eevl endonucleases are among the many paradigms for such DSB-induced recombination events. HO and I-Scel have been used to induce homologous gene targeting in yeast, in cultured mammalian cells, and in plants. See, e.g., US 5,792,632; US 6,238,924; US 5,792,632; US 5,830,729; US 6,238,924; US 5,792,632; US 6,238,924. Meganucleases are further described in, e.g., W02004067753; W02008010009; WO2008149176; W02009013559; W02009059195; WO2017112859; WO2017062439; US 8,119,381; US 8,338,157; and US 8,927,247.
The disclosure includes a method of modifying a precursor, the method comprising: introducing a CAR into the precursor, wherein the CAR comprises: an antigen binding domain; a transmembrane domain; an intracellular signaling domain; and an intracellular co-stimulatory signaling domain having an amino acid sequence having at least 90% sequence identity to a cytoplasmic domain of a herpes virus entry mediator (HVEM) protein set forth as any one of SEQ ID NOs: 7, 1, or 2, or a functional fragment or variant thereof that retains co-stimulatory activity, wherein the modified precursor cell expresses the CAR. In some embodiments, introducing the CAR into the precursor cell comprises introducing a nucleic acid sequence comprising a polynucleotide sequence encoding the CAR. In some embodiments, introducing the nucleic acid molecule comprises transducing the precursor cell with a viral vector comprising the nucleic acid sequence encoding the CAR.
The terms “transformation” or “transfection” may be used interchangeably and refer to the introduction of a nucleic acid into a cell. Transformation of a cell may be stable or transient. Thus, in some embodiments, a host cell or host organism may be stably transformed with a polynucleotide/nucleic acid molecule of the disclosure. In some embodiments, a host cell or host organism may be transiently transformed with a nucleic acid construct of the disclosure. Transient transformation in the context of a polynucleotide means that a polynucleotide is introduced into the cell and does not integrate into the genome of the cell. Stably introducing a polynucleotide introduced into a cell means that the introduced polynucleotide is stably incorporated into the genome of the cell, and thus the cell is stably transformed with the polynucleotide. Stable transformation of a cell means that a nucleic acid molecule is introduced into a cell and integrates into the genome of the cell. As such, the integrated nucleic acid molecule is capable of being inherited by the progeny thereof, more particularly, by the progeny of multiple successive generations. Stable transformation can also refer to a transgene that is maintained extrachromosomally, for example, as a minichromosome or a plasmid.
Methods of introducing and expressing genes, such as a CAR of the disclosure, into a cell are known in the art. In the context of an expression vector, the vector can be readily introduced into a host cell, e.g., mammalian, bacterial, yeast, or insect cell by any method in the art. For example, the expression vector can be transferred into a host cell by physical, chemical, or biological means.
Physical methods for introducing a polynucleotide into a host cell include calcium phosphate precipitation, lipofection, particle bombardment, microinjection, electroporation, and the like. Methods for producing cells comprising vectors and/or exogenous nucleic acids are well-known in the art. See, for example, Sambrook et al., 2012, MOLECULAR CLONING: A LABORATORY MANUAL, volumes 1 -4, Cold Spring Harbor Press, NY. Nucleic acids can be introduced into target cells using commercially available methods which include electroporation (e.g., Amaxa Nucleofector-II (Amaxa Biosystems, Cologne, Germany), ECM 830 (BTX) (Harvard Instruments, Boston, Mass.), the Gene Pulser II (BioRad, Denver, Colo.), or Multiporator (Eppendort, Hamburg Germany)). Nucleic acids can also be introduced into cells using cationic liposome mediated transfection using lipofection, using polymer encapsulation, using peptide mediated transfection, or using biolistic particle delivery systems such as "gene guns" (see, for example, Nishikawa, et al. Hum Gene Then, 12(8): 861 -70 (2001).
Biological methods for introducing a polynucleotide of interest into a host cell include the use of DNA and RNA vectors. RNA vectors include vectors having a RNA promoter and/or other relevant domains for production of a RNA transcript. Viral vectors, and especially retroviral vectors, have become the most widely used method for inserting genes into mammalian, e.g., human cells. Other
viral vectors may be derived from lentivirus, poxviruses, herpes simplex virus, adenoviruses and adeno-associated viruses, and the like. See, for example, U.S. Pat. Nos. 5,350,674 and 5,585,362.
Chemical means for introducing a polynucleotide into a host cell include colloidal dispersion systems, such as macromolecule complexes, nanocapsules, microspheres, beads, and lipid-based systems including oil-in-water emulsions, micelles, mixed micelles, and liposomes. An exemplary colloidal system for use as a delivery vehicle in vitro and in vivo is a liposome (e.g. , an artificial membrane vesicle).
In the case where a non-viral delivery system is utilized, an exemplary delivery vehicle is a liposome. The use of lipid formulations is contemplated for the introduction of the nucleic acids into a host cell (in vitro, ex vivo or in vivo). The nucleic acid may be associated with a lipid. The nucleic acid associated with a lipid may be encapsulated in the aqueous interior of a liposome, interspersed within the lipid bilayer of a liposome, attached to a liposome via a linking molecule that is associated with both the liposome and the nucleic acid, entrapped in a liposome, complexed with a liposome, dispersed in a solution containing a lipid, mixed with a lipid, combined with a lipid, contained as a suspension in a lipid, contained or complexed with a micelle, or otherwise associated with a lipid. Lipid, lipid/DNA or lipid/expression vector associated compositions are not limited to any particular structure in solution. For example, they may be present in a bilayer structure, as micelles, or with a "collapsed" structure. They may also simply be interspersed in a solution, possibly forming aggregates that are not uniform in size or shape. Lipids are fatty substances which may be naturally occurring or synthetic lipids. For example, lipids include the fatty droplets that naturally occur in the cytoplasm as well as the class of compounds which contain long-chain aliphatic hydrocarbons and their derivatives, such as fatty acids, alcohols, amines, amino alcohols, and aldehydes.
Lipids suitable for use can be obtained from commercial sources. For example, dimyristyl phosphatidylcholine ("DMPC") can be obtained from Sigma, St. Louis, MO; dicetyl phosphate ("DCP") can be obtained from K & K Laboratories (Plainview, NY); cholesterol ("Choi") can be obtained from Calbiochem-Behring; dimyristyl phosphatidylglycerol ("DMPG") and other lipids may be obtained from Avanti Polar Lipids, Inc. (Birmingham, AL.). Stock solutions of lipids in chloroform or chloroform/methanol can be stored at about -20°C. Chloroform is used as the only solvent since it is more readily evaporated than methanol. "Liposome" is a generic term encompassing a variety of single and multilamellar lipid vehicles formed by the generation of enclosed lipid bilayers or aggregates. Liposomes can be characterized as having vesicular structures with a phospholipid bilayer membrane and an inner aqueous medium.
Multilamellar liposomes have multiple lipid layers separated by aqueous medium. They form spontaneously when phospholipids are suspended in an excess of aqueous solution. The lipid
components undergo self-rearrangement before the formation of closed structures and entrap water and dissolved solutes between the lipid bilayers (Ghosh et al., 1991 Glycobiology 5: 505-10). However, compositions that have different structures in solution than the normal vesicular structure are also encompassed. For example, the lipids may assume a micellar structure or merely exist as nonuniform aggregates of lipid molecules. Also contemplated are lipofectamine-nucleic acid complexes.
The nucleic acids introduced into a precursor cell can be RNA. In some embodiments, the RNA is mRNA that comprises in vitro transcribed RNA or synthetic RNA. The RNA is produced by in vitro transcription using a PCR-generated template. DNA of interest from any source can be directly converted by PCR into a template for in vitro mRNA synthesis using appropriate primers and RNA polymerase. The source of the DNA can be, for example, genomic DNA, plasmid DNA, phage DNA, cDNA, synthetic DNA sequence or any other appropriate source of DNA. The desired template for in vitro transcription is a polynucleotide sequence encoding a CAR having an intracellular co-stimulatory signaling domain having an amino acid sequence having at least 90% sequence identity to a cytoplasmic domain of a herpes virus entry mediator (HVEM) protein set forth as any one of SEQ ID NOs: 1, 2, or 3, or a functional fragment or variant thereof that retains co-stimulatory activity.
PCR can be used to generate a template for in vitro transcription of mRNA which is then introduced into cells. Methods for performing PCR are well known in the art. Primers for use in PCR are designed to have regions that are substantially complementary to regions of the DNA to be used as a template for the PCR. "Substantially complementary", as used herein, refers to sequences of nucleotides where a majority or all of the bases in the primer sequence are complementary, or one or more bases are non-complementary, or mismatched. Substantially complementary sequences are able to anneal or hybridize with the intended DNA target under annealing conditions used for PCR. The primers can be designed to be substantially complementary to any portion of the DNA template. For example, the primers can be designed to amplify the portion of a gene that is normally transcribed in cells (the open reading frame), including 5' and 3' UTRs. The primers can also be designed to amplify a portion of a gene that encodes a particular domain of interest. In some embodiments, the primers are designed to amplify the coding region of a human cDNA, including all or portions of the 5' and 3' UTRs. Primers useful for PCR are generated by synthetic methods that are well known in the art. "Forward primers" are primers that contain a region of nucleotides that are substantially complementary to nucleotides on the DNA template that are upstream of the DNA sequence that is to be amplified. "Upstream" is used herein to refer to a location 5' to the DNA sequence to be amplified relative to the coding strand. "Reverse primers" are primers that contain a region of nucleotides that are substantially complementary to a double-stranded DNA template that are downstream of the DNA
sequence that is to be amplified. "Downstream" is used herein to refer to a location 3' to the DNA sequence to be amplified relative to the coding strand.
Chemical structures that have the ability to promote stability and/or translation efficiency of the RNA may also be used. The RNA preferably has 5' and 3' UTRs. In one embodiment, the 5' UTR is between zero and 3000 nucleotides in length. The length of 5' and 3' UTR sequences to be added to the coding region can be altered by different methods, including designing primers for PCR that anneal to different regions of the UTRs. Using this approach, one of ordinary skill in the art can modify the 5' and 3' UTR lengths required to achieve optimal translation efficiency following transfection of the transcribed RNA.
The 5' and 3' UTRs can be the naturally occurring, endogenous 5' and 3' UTRs for the gene of interest. Alternatively, UTR sequences that are not endogenous to the gene of interest can be added by incorporating the UTR sequences into the forward and reverse primers or by any other modifications of the template. The use of UTR sequences that are not endogenous to the gene of interest can be useful for modifying the stability and/or translation efficiency of the RNA. For example, it is known that AU-rich elements in 3' UTR sequences can decrease the stability of mRNA. Therefore, 3' UTRs can be selected or designed to increase the stability of the transcribed RNA based on properties of UTRs that are well known in the art.
In some embodiments, the 5' UTR can contain the Kozak sequence of the endogenous gene. Alternatively, when a 5' UTR that is not endogenous to the gene of interest is being added by PCR as described above, a consensus Kozak sequence can be redesigned by adding the 5' UTR sequence. Kozak sequences can increase the efficiency of translation of some RNA transcripts, but does not appear to be required for all RNAs to enable efficient translation. The requirement for Kozak sequences for many mRNAs is known in the art. In some embodiments, the 5' UTR can be derived from an RNA virus whose RNA genome is stable in cells. Various nucleotide analogues can be used in the 3' or 5' UTR to impede exonuclease degradation of the mRNA.
To enable synthesis of RNA from a DNA template without the need for gene cloning, a promoter of transcription should be attached to the DNA template upstream of the sequence to be transcribed. When a sequence that functions as a promoter for an RNA polymerase is added to the 5' end of the forward primer, the RNA polymerase promoter becomes incorporated into the PCR product upstream of the open reading frame that is to be transcribed. In some embodiments, the promoter is a T7 polymerase promoter. Other useful promoters include T3 and SP6 RNA polymerase promoters. Consensus nucleotide sequences for T7, T3, and SP6 promoters are known in the art.
In some embodiments, the mRNA has both a cap on the 5' end and a 3' poly(A) tail which determine ribosome binding, initiation of translation and stability mRNA in the cell. On a linear DNA
template, phage T7 RNA polymerase can extend the 3' end of the transcript beyond the last base of the template (Schenborn and Mierendorf, Nuc Acids Res., 13 :6223-36 (1985); Nacheva and Berzal- Herranz, Eur. J. Biochem., 270: 1485-65 (2003)).
The conventional method of integration of poly A/T stretches into a DNA template is molecular cloning. However poly A/T sequence integrated into plasmid DNA can cause plasmid instability, which is why plasmid DNA templates obtained from bacterial cells are often highly contaminated with deletions and other aberrations. This makes cloning procedures not only laborious and time consuming but often not reliable. That is why a method which allows construction of DNA templates with poly A/T 3' stretch without cloning is highly desirable.
The poly A/T segment of the transcriptional DNA template can be produced during PCR by using a reverse primer containing a polyT tail, such as 100T tail (size can be 50-5000 T), or after PCR by any other method, including DNA ligation or in vitro recombination. Poly(A) tails also provide stability to RNAs and reduce their degradation. Generally, the length of a poly(A) tail positively correlates with the stability of the transcribed RNA. In some embodiments, the poly(A) tail is between 100 and 5000 adenosines. Poly(A) tails of RNAs can be further extended following in vitro transcription with the use of a poly(A) polymerase, such as E. coli poly A polymerase (E-PAP). In some embodiments, increasing the length of a poly(A) tail from 100 nucleotides to between 300 and 400 nucleotides results in about a two-fold increase in the translation efficiency of the RNA. Additionally, the attachment of different chemical groups to the 3' end can increase mRNA stability. Such attachment can contain modified/artificial nucleotides, aptamers and other compounds. For example, ATP analogs can be incorporated into the poly(A) tail using poly(A) polymerase. ATP analogs can further increase the stability of the RNA.
5' caps also provide stability to RNA molecules. The 5' cap is provided using techniques known in the art and described in, e.g., Cougot et al. Trends in Biochem. Sci. 29:436-444 (2001); Stepinski et al. RNA 7: 1468-95 (2001); and Elango et al. Biochim.
Biophys. Res. Commun, 330:958-966 (2005). The RNAs produced by the methods disclosed herein can also contain an internal ribosome entry site (IRES) sequence. The IRES sequence may be any viral, chromosomal or artificially designed sequence which initiates cap-independent ribosome binding to mRNA and facilitates the initiation of translation. Any solutes suitable for cell electroporation, which can contain factors facilitating cellular permeability and viability such as sugars, peptides, lipids, proteins, antioxidants, and surfactants can be included.
Some in iv/m-transcribed RNA (IVT-RNA) vectors are known in the literature which are utilized in a standardized manner as template for in vitro transcription and which have been genetically modified in such a way that stabilized RNA transcripts are produced.
Currently protocols used in the art are based on a plasmid vector with the following structure: a 5' RNA polymerase promoter enabling RNA transcription, followed by a gene of interest which is flanked either 3' and/or 5' by untranslated regions (UTR), and a 3' polyadenyl cassette containing 50-70 A nucleotides. Prior to in vitro transcription, the circular plasmid is linearized downstream of the polyadenyl cassette by type II restriction enzymes (recognition sequence corresponds to cleavage site).
Nucleic acids can be delivered into cells by electroporation. In some embodiments, the nucleic acid includes an RNA. See, e.g., the formulations and methodology of electroporation of nucleic acid constructs into mammalian cells as taught in, e.g., US 2004/0014645, US 2005/0052630A1, US 2005/0070841, US 2004/0059285, and US 2004/0092907. The various parameters including electric field strength required for electroporation of any known cell type are generally known in the relevant research literature as well as numerous patents and applications in the field. See e.g., U.S. Pat. No. 6,678,556, U.S. Pat. No. 7,171,264, and U.S. Pat. No. 7,173,116. Apparatus for therapeutic application of electroporation are available commercially, e.g., the MedPulser™ DNA Electroporation Therapy System (Inovio/Genetronics, San Diego, Calif), and are described in patents such as U.S. Pat. No. 6,567,694; U.S. Pat. No. 6,516,223, U.S. Pat. No. 5,993,434, U.S. Pat. No. 6, 181,964, U.S. Pat. No. 6,241,701, and U.S. Pat. No. 6,233,482. Electroporation may also be utilized to deliver nucleic acids into cells in vitro utilizing any of the many available devices and electroporation systems known to those of skill in the art. Electroporation of cells in vitro is described, e.g., in US 2007/0128708.
C. Chimeric Antigen Receptor (CAR)
Also disclosed herein is a chimeric antigen receptor (CAR) to be expressed in a precursor, such as a bone marrow-derived cell or a stem cell, wherein the CAR comprises an antigen binding domain; a transmembrane domain; an intracellular signaling domain; and an intracellular costimulatory signaling domain. The CAR comprises an intracellular co-stimulatory signaling domain having an amino acid sequence having at least 90% sequence identity to a cytoplasmic domain of a herpes virus entry mediator (HVEM) protein set forth as any one of SEQ ID NOs: 1, 2, or 3, or a functional fragment or variant thereof that retains co-stimulatory activity.
The present disclosure provides a myeloid-derived cell genetically modified to express a chimeric antigen receptor. A chimeric antigen receptor (CAR) is an artificial cell surface receptor that is engineered to be expressed on an immune effector cell and specifically bind an antigen. A CAR typically comprises an antigen binding domain and a signal transduction domain capable of mimicking the T cell receptor-mediated signaling pathway. A "signaling pathway" or "signal transduction pathway" refers to the biochemical relationship between a variety of signal transduction molecules that play a role in the transmission of a signal from one portion of a cell to another portion of a cell. The phrase "cell surface receptor" includes molecules and complexes of molecules capable of receiving a
signal and transmitting the signal across the plasma membrane of a cell. Antigen binding domains that are used in CARs include a natural ligand and the single-chain variable region of an antibody to a target molecule. The advantage of a CAR is that it can recognize a defined target without requiring antigen-processing or the major histocompatibility complex (MHC)-restricted antigen presentation, allowing the use of T cells expressing CAR (CAR-T cells) for adoptive immunotherapy in a wide range of patients (Doth et al, Immunol. Rev. 257(1): 107 (2014)). T cells expressing the first generation CAR having CD3z as the signal transduction domain often become anergic and fail to elicit a potent immune response (Kershaw et al, Clin. Cancer Res. 12(20 Pt 1): 6106 (2006)). To solve this problem, second and third generation CARs that have one and two co-stimulatory signaling domains derived from CD28, 4-1BB or ICOS have been developed (Doth et al., Immunol. Rev. 257(1): 107 (2014)). These CARs have been shown to successfully mimic T cell receptor-mediated signal transduction upon antigen stimulation, leading to proliferation and activation of CAR-T cells (Maus et al, Blood 123(17):2625 (2014)).
The CAR can help immune cells such as macrophages find and kill cancer cells that have the specific protein the receptor is designed to bind. In some instances, a precursor cell, such as a stem cell can be removed from the bone-marrow or blood of a patient and modified to express a CAR. For example, hematopoietic cells can be removed from the bone marrow and genetically modified to express a CAR specific to a particular form of antigen on tumor cells, and then expanded and differentiated into a myeloid-derived CAR (e.g., a monocyte or macrophage) using the methods described herein. The precursor cell (e.g., hematopoietic stem cell) expressing the CAR can then be differentiated into a cell within a specific cell lineage. In some instances, the hematopoietic stem cell (HSC) expressing a CAR can be differentiated into a cell within the myeloid lineage, such as, for example, a monocyte or macrophage. The HSC CAR cell can also be differentiated into a parent cell of a monocyte, such as a myeloid progenitor cell. When the stem cell (i.e., hematopoietic stem cell) expressing the CAR is differentiated, the CAR is retained on the surface of the newly differentiated cell. Therefore, when a HSC CAR cell is differentiated to generate a CAR-expressing phagocytic cell, the CAR can target cancers by redirecting a phagocyte such as a monocyte or macrophage expressing the CAR specific for tumor associated antigens. In some embodiments of a genetically modified phagocytic cell of the present disclosure and related compositions and methods herein described, CARs can comprise an antigen binding domain, a transmembrane domain, and an intracellular domain comprising a co-stimulatory signaling domain of a herpes virus entry mediator (HVEM) protein as described herein.
Antigen Binding Domain
A CAR expressed by a modified stem cell, or myeloid cell derived from a stem cell (e.g., a HSC differentiated into a modified phagocytic cell) of the disclosure can comprise an antigen binding domain that binds to an antigen on a target cell. Examples of cell surface markers that may act as an antigen that binds to the antigen binding domain of the CAR include those associated with viral, bacterial and parasitic infections, autoimmune disease, degenerative disease, and cancer cells.
The antigen binding domain can include any domain that binds to the antigen and may include, but is not limited to, a monoclonal antibody, a polyclonal antibody, a synthetic antibody, a human antibody, a humanized antibody, a non-human antibody, and any fragment thereof. Thus, in some embodiments, the antigen binding domain portion comprises a mammalian antibody or a fragment thereof.
In some embodiments, the antigen binding domain comprises a monovalent antibody fragment. The monovalent antibody fragment can comprise a single chain variable fragment (scFv) or a Fab fragment. In some embodiments, the monovalent antibody fragment has a molecular weight of about 25 to about 30 kDa (or about 25 kDa, about 26 kDa, about 27 kDa, about 28 kDa, about 29 kDa, or 30 kDa). The monovalent antibody fragment can have a VH and VL domain connected in either orientation by a flexible linker (e.g., VL-linker-VH or VH-linker-VL). The flexible linker typically comprises 10 to about 25 amino acids (e.g., glycine to confer flexibility and/or serines and/or threonines for improved solubility). For example, a Gly-Ser linker may be used. Further suitable linkers are described, e.g., in Alfthan, K. Properties of a single-chain antibody containing different linker peptides. Protein Engineering 1995, vol. 8, no. 7, p. 725-731, which is incorporated by reference in its entirety.
In some embodiments, the antigen binding domain is derived from the same species in which the CAR will ultimately be used in. For example, for use in humans, the antigen binding domain of the CAR can comprise a human antibody, a humanized antibody, or a fragment thereof.
In some embodiments, the antigen binding domain is operably linked to another domain of the CAR, such as the transmembrane domain or the intracellular domain, for expression in the cell. For example, a polynucleotide sequence encoding the antigen binding domain is operably linked at its 3’ end to a polynucleotide sequence encoding a transmembrane domain and a polynucleotide sequence encoding an intracellular domain. A nucleic acid molecule comprising a polynucleotide sequence encoding a CAR can be contacted with a precursor, such as a stem cell, to introduce the CAR into the cell, generating a stem cell modified to express a CAR. The nucleic acid molecule introduced into the stem cell can comprise multiple polynucleotide sequences encoding multiple domains of a CAR.
The choice of antigen binding domain depends upon the type and number of antigens that are present on the surface of a target cell. For example, the antigen binding domain may be chosen to
recognize an antigen that acts as a cell surface marker on a target cell associated with a particular disease state.
In some embodiments, the antigen binding domain binds to a tumor antigen, such as an antigen that is specific for a tumor or cancer of interest. The tumor antigen can comprise one or more antigenic cancer epitopes.
In some embodiments, the antigen binding domain targets an antigen present on the surface of a viral particle. Examples of viral particles include influenza virus, equine infectious anemia virus, simian immunodeficiency virus (SIV), human immunodeficiency virus (HIV), lassa fever virus, herpes simplex virus, varicella zoster virus, cytomegalovirus, epstein-barr virus, variola virus, adeno virus, papilloma virus, parvo virus, measles virus, mumps virus, respiratory syncytial virus, para influenza virus, corona virus, rubella virus, rabies virus, human T-cell lymphotropic virus, picoma virus, hepa DNA virus, flavivirus, deltavirus, calicivirus, polio virus, zika virus, west nile virus, SARS, rubella, norovirus, human papillomavirus, malaria, human T-lymphotropic virus, and/or helicobacter pylori.
Exemplary viral antigens include any surface protein and/or polypeptide present on the surface of the above listed viral particles. Examples of such surface proteins and/or polypeptides include: Zika capsid protein (C), Zika envelope protein (E), Zika precursor membrane protein (PrM), WNV glycoprotein E, WNV small membrane protein M, VZV glycoprotein E (gE), VZV gB, VZV gH, CoV nucleocapsid (N), CoV envelope (E), CoV membrane (M), Rubella El, Rubella E2, Norovirus Group VP1, HPV 11, HPV capsid protein LI, HPV capsid protein L2, Lassa Fever Virus GP1, Lassa Fever Virus GP2, influenza neuraminidase (NA, N1 to N11), influenza hemagglutinin (HA, Hl to Hl 8), HTLV-1 Envelope, HTLV-1 gp21, HTLV-1 mosaic, HIV gpl20, HIV gp41, hepatitis A virus (HAV) capsid protein VP1, HAV capsid protein VP2, HAV capsid protein VP3, hepatitis B surface antigen (HbsAg), hepatitis B core antigen (HbcAg), herpes simplex virus glycoprotein (gB), herpes simplex virus glycoprotein (gC), and/or herpes simplex virus glycoprotein (gD).
A myeloid-derived cell of the disclosure can also be modified to express additional targeting ligands on the surface of the myeloid-derived cell in addition to the antigen binding domain of a CAR described herein. Additional targeting ligands comprise molecules configured to associate with any molecule presented on a target cell of interest, such as a target associated with an organ, a tissue, or an extracellular matrix of a target cell of interest. In some embodiments, additional targeting ligands bind to molecules associated with a particular state of a target cell, such as a cancerous condition. An additional targeting ligand can be specific to one target or be configured to bind multiple target molecules. Suitable target molecules recognized by an additional target ligand can include a protein (e.g., a receptor, a tumor marker, a transmembrane protein), a nucleic acid (e.g., DNA, RNA), and/or a
carbohydrate (e.g., a monosaccharide, disaccharide, or polysaccharide) that is present on the surface of a target cell. Exemplary targeting ligands include an RGD-containing peptide, a small molecule (e.g., a peptide) mimetic ligand, and an antibody or antibody fragment specific for a particular target.
Transmembrane Domain
A CAR expressed by a modified myeloid-derived cell of the disclosure can comprise a transmembrane domain that connects the antigen binding domain of the CAR to the intracellular domain. The transmembrane domain can function to stabilize the CAR as a whole. The transmembrane domain can be selected or modified by amino acid substitution to avoid binding of such domains to the transmembrane domains of the same or different surface membrane proteins to minimize interactions with other molecules.
The transmembrane domain may be derived either from a natural or from a synthetic source. Where the source is natural, the domain may be derived from any membrane-bound or transmembrane protein. Transmembrane regions of particular use in this invention may be derived from (i.e. comprise at least the transmembrane region(s) of) the alpha, beta or zeta chain of the T-cell receptor (TCR), CD28, CD3 epsilon, CD45, CD4, CD5, CD8, CD9, CD16, CD22, CD33, CD37, CD64, CD80, CD86, CD134, CD137, CD154, Toll-like receptor 1 (TLR1), TLR2, TLR3, TLR4, TLR5, TLR6, TLR7, TLR8, and TLR9. In some embodiments, the transmembrane domain can be from any type I transmembrane protein such as CD4, CD28 or HVEM or a functional fragment or variant thereof that retains co-stimulatory activity. In some embodiments, the transmembrane domain is a CD8 transmembrane domain or a functional fragment or variant thereof. In some embodiments, the transmembrane domain is a CD28 transmembrane domain or a functional fragment or variant thereof. In some embodiments, a variety of human hinges can be employed as well including the human Ig (immunoglobulin) hinge.
In some embodiments, the transmembrane domain may be a hydrophobic alpha helix that spans across the membrane of the cell (e.g., macrophage). The transmembrane domain can be naturally associated with one or more of the domains in the CAR.
In some embodiments, the transmembrane domain may be synthetic, in which case it can comprise predominantly hydrophobic residues such as leucine and valine. A triplet of phenylalanine, tryptophan and valine can be found at each end of a synthetic transmembrane domain.
Intracellular Domain
A CAR expressed by a modified myeloid-derived cell of the disclosure can comprise an intracellular domain. The intracellular (z.e., cytoplasmic) domain of a CAR is a signaling domain that transduces the event of receptor antigen binding to an intracellular signal that contributes to activation and/or transduction of signals in a cell in which the CAR is expressed. In some embodiments, absent
appropriate co-stimulatory signals, this event is insufficient for useful cell activation and proliferation. Cell activation can include cytokine production, clonal proliferation, differentiation, and survival.
Examples of an intracellular domain for use in the disclosure include the cytoplasmic portion of a surface receptor, co-stimulatory molecule, and any molecule that acts in concert to initiate signal transduction in a myeloid-derived cell, as well as any derivative or variant of these elements and any synthetic sequence that has the same functional capability.
Examples of an intracellular domain include a fragment or domain from one or more molecules or receptors including: T cell receptor (TCR), CD3 zeta, CD3 gamma, CD3 delta, CD3 epsilon, CD86, common FcR gamma, FcR beta (Fc Epsilon Rib), CD79a, CD79b, Fcgamma Rlla, DAP10, DAP12, CD27, CD28, 4-1BB (CD137), 0X40, CD30, CD40, PD-1, ICOS, lymphocyte function-associated antigen-1 (LFA-1), CD2, CD7, LIGHT, NKG2C, B7-H3, a ligand that specifically binds with CD83, CDS, ICAM-1, GITR, BAFFR, HVEM (LIGHTR), SLAMF7, NKp80 (KLRF1), CD127, CD160, CD 19, CD4, CD8alpha, CD8beta, IL2R beta, IL2R gamma, IL7R alpha, ITGA4, VLA1, CD49a, ITGA4, IA4, CD49D, ITGA6, VLA-6, CD49f, ITGAD, CD 1 id, ITGAE, CD103, ITGAL, CDl la, LFA-1, ITGAM, CDl lb, ITGAX, CDl lc, ITGB1, CD29, ITGB2, CD18, LFA-1, ITGB7, TNFR2, TRANCE/RANKL, DNAM1 (CD226), SLAMF4 (CD244, 2B4), CD84, CD96 (Tactile), CEACAM1, CRTAM, Ly9 (CD229), CD160 (BY55), PSGL1, CD100 (SEMA4D), CD69, SLAMF6 (NTB-A, Lyl08), SLAM (SLAMF1, CD 150, IPO-3), BLAME (SLAMF8), SELPLG (CD 162), LTBR, LAT, GADS, SLP-76, PAG/Cbp, NKp44, NKp30, NKp46, NKG2D, Toll-like receptor 1 (TLR1), TLR2, TLR3, TLR4, TLR5, TLR6, TLR7, TLR8, TLR9, other co-stimulatory molecules described herein, any derivative, variant, or fragment thereof, any synthetic sequence of a co-stimulatory molecule that has the same functional capability, and any combination thereof.
In some embodiments of the disclosure, the intracellular signaling domain of the CAR includes at least 1, at least 2, at least 3, at least 4, or at least 5 immunoreceptor tyrosine-based activation motifs (IT AMs). Generally, any intracellular signaling domain including an ITAM can be suitably used for the construction of CAR. An “ITAM,” as used herein, is a conserved protein motif that is generally present in the tail portion of signaling molecules expressed in many immune cells. The motif may include two repeats of the amino acid sequence YXXL/I separated by 6-8 amino acids, wherein each X is independently any amino acid, producing the conserved motifs: YXXL/IXXXXXXYXXL/I (SEQ ID NO: 7); YXXL/IXXXXXXXYXXL/I (SEQ ID NO: 8); or YXXL/IXXXXXXXXYXXL/I (SEQ ID NO: 9). ITAMs within signaling molecules are important for signal transduction within the cell, which is mediated at least in part by phosphorylation of tyrosine residues in the ITAM following activation of the signaling molecule. ITAMs may also function as docking sites for other proteins involved in signaling pathways. In some embodiments, the intracellular signaling domain includes at least 1, at
least 2, at least 3, at least 4, or at least 5 IT AMs derived from CD3z, FcRy, Megfl 0, FcRy, and combinations thereof.
In some embodiments, the intracellular domain of a CAR useful in a modified myeloid-derived cell of the disclosure includes any portion of one or more co-stimulatory molecules. Co-stimulation is a key event for T cells to exhibit effective effector functions and is mediated by co-stimulatory molecules. In some embodiments, the co-stimulatory domain of a CAR is essential for promoting the intracellular signal of the T-cell receptor domain to initiate T cell activation and proliferation. Thus, promotion of such a signal can depend upon the selected co-stimulatory signaling domain and/or combinations thereof. Second generation CARs incorporate an intracellular co-stimulatory signaling domain in addition to an intracellular signaling domain (e.g., CD3z) and can enhance CAR T cell activation. Co-stimulatory molecules are divided into two major families; the CD28 family which includes CD28 and ICOS, and the tumor necrosis factor receptor superfamily (TNFRSF) which includes 4- IBB (TNFRSF9), CD27, CD30, DR3, GITR, 0X40, TNFR2 and herpes virus entry mediator (HVEM, TNFRSF14). So far, co-stimulatory domains derived from CD28 or 4-1BB have commonly been used to construct CARs. A previous study has shown that T cells expressing the second generation CAR with the 4-lBB-derived co-stimulatory signaling domain persist for more than 6 months in the blood of most patients, whereas CAR-T cells with the CD28-derived co-stimulatory signaling domain become mostly undetectable after 3 months (Zhang et al, Oncotarget 6(32):33961 (2015)). In addition, 4-lBB-mediated co-stimulation selectively induced mitochondrial biogenesis and oxidative metabolism for energy production, resulting in enhanced differentiation and increased in vitro persistence of central memory T cells (Kawalekar et al, Immunity 44(2):380 (2016)). Moreover, 4-lBB-mediated co-stimulation averts T cell exhaustion induced by tonic signaling (Long et al, Nat. Med. 21 (6):581 (2015)). Therefore, the co-stimulatory signaling domain derived from the TNFRSF appears to function better than the one from the CD28 family in the context of second generation CAR. Third generation CARs incorporate two co-stimulatory domains and can promote CAR-T cell proliferation, reduce CAR-T cell apoptosis, and/or increase the the NF- K B pathway (Dai et al. (2020) Frontiers in Immunology 11 : 539654).
HVEM, another member of the TNFRSF, is the main receptor targeted by the Herpes simplex virus to enter host cells (Sedy et al. (2005) Nature Immunology 6( 1 ) : 90-98). It was previously described as AT AR (Another TRAF- Associated Receptor) (Hsu et al. (1997) Journal of Biological Chemistry 272(21): 13471-13474) and is also known as tumor necrosis factor receptor superfamily member 14 (TNFRSF14). A human HVEM is 283 amino acids in length (UniProt ID Q92956-1; SEQ ID NO: 4), comprising: an extracellular region that includes amino acid residues 39-202; a transmembrane region that includes amino acid residues 203-223; and a cytoplasmic region that
includes amino acid residues 224-283. HVEM plays a role in effector CD8+ T cell effector function and memory T cell development. HVEM deficiency in CD8+ T cells is shown to profoundly impair effector CD8+ T cell survival and development of protective immune memory (Flynn et al, PLoS One 8(10):e77991 (2013)). B and T lymphocyte attenuator (a ligand of HVEM) interaction with HVEM expressed on CD8+ T cells was also reported to promote survival and memory generation in response to a bacterial infection (Steinberg et al., PLoS One 8(10):e77992 (2013)). Additionally, tumor cells which express anti-HVEM single chain antibody induce a potent proliferation and cytokine production of co-cultured T cells (Park et al, Cancer Immunol. Immunother. 61(2):203 (2012)), suggesting HVEM might serve as a potent co- stimulatory signaling entity in T cells.
HVEM is expressed in peripheral T and B cells, and in resting T and B cells (Ning et al. (2021) Frontiers in Immunology 12:654960). HVEM is constitutively expressed in naive T cells. The HVEM receptor has both stimulatory and inhibitory effects depending on its ligand, and at least five different ligands have been described (Cai & Freeman (2009) Immunological Reviews 229(l):244-258; Del Rio et al. (2010) Journal of Leukocyte Biology 87(2):223-235; Granger & Rickert (2003) Cytokine Growth Factor Rev 14(3-4):289-296; Pasero & Olive (2013) Immunology Letters 15 l(l-2):71-75; Sorobetea & Brodsky (2018) Cell Host & Microbe 24(2): 187-188). For example, in an inflammatory disease model setting, HVEM binding to the LIGHT ligand of the TNF family and lymphotoxin alpha (LTa) produces a co- stimulatory signal, leading to increased T cell proliferation in vitro and formation of effector and memory T cells (Del Rio et al. (2010) Journal of Leukocyte Biology 87(2):223-235). However, HVEM can generate an inhibitory signal when bound to BTLA or CD 160 (Pasero & Olive (2013) Immunology Letters 15 l(l-2):71-75). The overexpression of BTLA/HVEM on T cells can contribute to T cell exhaustion (Ning et al. (2021) Frontiers in Immunology 12:654960; Shui et al. (2011) Journal of Leukocyte Biology, 89(4), 517-523).
The cytoplasmic domain of HVEM is 60 amino acids in length and includes an a-helix, a TRAF domain, and a tail (amino acid residues 224-283 of SEQ ID NO: 4; Hennecke, Derek. 2022. A Comparison of Co-stimulatory HVEM Domains in Second Generation CAR-T Cells. Master's thesis, Harvard University Division of Continuing Education). M83 interacts with adapter proteins, including TRAFs (TRAF5 and TRAF2) (Hsu et al. (1997) Journal of Biological Chemistry 272(21): 13471- 13474), which are important signaling molecules downstream of TNF receptors, connecting receptor signaling with kinase (e.g., IKB kinase) activation, which leads to transcription factor (e.g., AP-1, NF AT, NF-KB) activation (Zarnegar et al. (2008) Nature Immunology 9(12): 1371-1378). Transcription factor activation is important, for example, in T cell priming and control of tumors in vivo (Barnes et al. (2015) J for Immunotherapy of Cancer 3(1): 1-11).
The co-stimulatory signaling domain from an HVEM protein can include all or part of the transmembrane domain along with all or part of the intracellular region or can include only all or part of the intracellular region of the HVEM protein.
A nucleic acid sequence disclosed herein can encode a HVEM co-stimulatory signaling domain, or functional fragment or variant thereof, of the disclosure, can have at least 80%, 81%, 82%, 83%, 84%, 85%, 86%, 87%, 88%, 89%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or 100% sequence identity to the nucleic acid sequence encoding SEQ ID NO: 3, wherein the encoded domain retains co-stimulatory activity. In some embodiments, a nucleic acid sequence encoding a HVEM co-stimulatory signaling domain, or functional fragment or variant thereof, of the disclosure, has at least 90% sequence identity to the nucleic acid sequence encoding SEQ ID NO: 3, wherein the encoded domain retains co-stimulatory activity. In some embodiments, a nucleic acid sequence encoding a HVEM co-stimulatory signaling domain, or functional fragment or variant thereof, of the disclosure, has at least 95% sequence identity to the nucleic acid sequence encoding SEQ ID NO: 3, wherein the encoded domain retains co-stimulatory activity. In some embodiments, a nucleic acid sequence encoding a HVEM co-stimulatory signaling domain of the disclosure has 100% sequence identity to the nucleic acid sequence encoding SEQ ID NO: 3.
A HVEM co-stimulatory signaling domain, or functional fragment or variant thereof, of the disclosure, can have an amino acid sequence having at least 80%, 81%, 82%, 83%, 84%, 85%, 86%, 87%, 88%, 89%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or 100% sequence identity to the amino acid sequence set forth as SEQ ID NO: 3 and retains co-stimulatory activity. In some embodiments, a HVEM co-stimulatory signaling domain, or functional fragment or variant thereof, of the disclosure, has an amino acid sequence having at least 90% sequence identity to the amino acid sequence set forth as SEQ ID NO: 3 and retains co-stimulatory activity. In some embodiments, a HVEM co-stimulatory signaling domain, or functional fragment or variant thereof, of the disclosure, has an amino acid sequence having at least 95% sequence identity to the amino acid sequence set forth as SEQ ID NO: 3 and retains co-stimulatory activity. In some embodiments, a HVEM co-stimulatory signaling domain of the disclosure has an amino acid sequence set forth as SEQ ID NO: 3. A HVEM co-stimulatory signaling domain can include amino acid residues 224-283 of SEQ ID NO: 4.
In some embodiments, a nucleic acid sequence encoding a HVEM co-stimulatory signaling domain, or functional fragment or variant thereof, of the disclosure, has at least 80%, 81%, 82%, 83%, 84%, 85%, 86%, 87%, 88%, 89%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or 100% sequence identity to the nucleic acid sequence encoding SEQ ID NO: 1, wherein the encoded domain retains co-stimulatory activity. In some embodiments, a nucleic acid sequence encoding a HVEM co- stimulatory signaling domain, or functional fragment or variant thereof, of the disclosure, has at least
90% sequence identity to the nucleic acid sequence encoding SEQ ID NO: 1, wherein the encoded domain retains co-stimulatory activity. In some embodiments, a nucleic acid sequence encoding a HVEM co-stimulatory signaling domain, or functional fragment or variant thereof, of the disclosure, has at least 95% sequence identity to the nucleic acid sequence encoding SEQ ID NO: 1, wherein the encoded domain retains co-stimulatory activity. In some embodiments, a nucleic acid sequence encoding a HVEM co-stimulatory signaling domain of the disclosure has 100% sequence identity to the nucleic acid sequence encoding SEQ ID NO: 1.
A HVEM co-stimulatory signaling domain, or functional fragment or variant thereof, of the disclosure, can have an amino acid sequence having at least 80%, 81%, 82%, 83%, 84%, 85%, 86%, 87%, 88%, 89%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or 100% sequence identity to the amino acid sequence set forth as SEQ ID NO: 1 and retains co-stimulatory activity. In some embodiments, a HVEM co-stimulatory signaling domain, or functional fragment or variant thereof, of the disclosure, has an amino acid sequence having at least 90% sequence identity to the amino acid sequence set forth as SEQ ID NO: 1 and retains co-stimulatory activity. In some embodiments, a HVEM co-stimulatory signaling domain, or functional fragment or variant thereof, of the disclosure, has an amino acid sequence having at least 95% sequence identity to the amino acid sequence set forth as SEQ ID NO: 1 and retains co-stimulatory activity. In some embodiments, a HVEM co-stimulatory signaling domain of the disclosure has an amino acid sequence set forth as SEQ ID NO: 1. A HVEM co-stimulatory signaling domain can include amino acid residues 201-283 of SEQ ID NO: 4.
A nucleic acid sequence encoding a HVEM co-stimulatory signaling domain, or functional fragment or variant thereof, of the disclosure, can have at least 80%, 81%, 82%, 83%, 84%, 85%, 86%, 87%, 88%, 89%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or 100% sequence identity to the nucleic acid sequence encoding SEQ ID NO: 2, wherein the encoded domain retains co- stimulatory activity. In some embodiments, a nucleic acid sequence encoding a HVEM co-stimulatory signaling domain, or functional fragment or variant thereof, of the disclosure, has at least 90% sequence identity to the nucleic acid sequence encoding SEQ ID NO: 2, wherein the encoded domain retains co-stimulatory activity. In some embodiments, a nucleic acid sequence encoding a HVEM co- stimulatory signaling domain, or functional fragment or variant thereof, of the disclosure, has at least 95% sequence identity to the nucleic acid sequence encoding SEQ ID NO: 2, wherein the encoded domain retains co-stimulatory activity. In some embodiments, a nucleic acid sequence encoding a HVEM co-stimulatory signaling domain of the disclosure has 100% sequence identity to the nucleic acid sequence encoding SEQ ID NO: 2.
A HVEM co-stimulatory signaling domain, or functional fragment or variant thereof, of the disclosure, can have an amino acid sequence having at least 80%, 81%, 82%, 83%, 84%, 85%, 86%,
87%, 88%, 89%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or 100% sequence identity to the amino acid sequence set forth as SEQ ID NO: 2 and retains co-stimulatory activity. In some embodiments, a HVEM co-stimulatory signaling domain, or functional fragment or variant thereof, of the disclosure, has an amino acid sequence having at least 90% sequence identity to the amino acid sequence set forth as SEQ ID NO: 2 and retains co-stimulatory activity. In some embodiments, a HVEM co-stimulatory signaling domain, or functional fragment or variant thereof, of the disclosure, has an amino acid sequence having at least 95% sequence identity to the amino acid sequence set forth as SEQ ID NO: 2 and retains co-stimulatory activity. In some embodiments, a HVEM co-stimulatory signaling domain of the disclosure has an amino acid sequence set forth as SEQ ID NO: 2. A HVEM co-stimulatory signaling domain can include amino acid residues 210-277 of SEQ ID NO: 4.
In some embodiments, a HVEM co-stimulatory signaling domain having co-stimulatory activity can bind a TRAF1, a TRAF2, a TRAF3, and/or a TRAF5 intracellular signaling adapters. In some embodiments, a HVEM co-stimulatory signaling domain having co-stimulatory activity does not bind a TRAF6 intracellular signaling adapter.
The HVEM co-stimulatory signaling domain can promote functional activity in the genetically modified myeloid-derived CAR cells disclosed herein.
In particular embodiments, the co-stimulatory signaling domain further comprises one or more additional co-stimulatory signaling domains (e.g., 2, 3, 4, 5, 6, 7, 8, 9, 10, etc.), or functional fragments or variants thereof. Non-limiting examples include a CD28 co-stimulatory signaling domain, a 4- IBB co-stimulatory signaling domain, an OX-40 co-stimulatory signaling domain, an ICOS co-stimulatory signaling domain, or any other co-stimulatory signaling domain and/or functional fragment or variant thereof now known or later identified. In some embodiments, a linker may be present between two or more of the domains, e.g., a 3-12 amino acid linker, or a 5-8 amino acid linker.
An intracellular signaling domain of the present disclosure can include a CD3 zeta intracellular signaling domain and an HVEM co-stimulatory signaling domain as described herein.
Spacers and linkers
Between the antigen binding domain and the transmembrane domain of the CAR, or between the intracellular domain and the transmembrane domain of the CAR, a spacer may be incorporated. As used herein, the term "spacer" generally means any oligo- or polypeptide that functions to link the transmembrane domain to, either the antigen binding domain or, the intracellular domain in the CAR polypeptide. In some embodiments, the spacer domain may comprise up to 300 amino acids, 10 to 100 amino acids, or 25 to 50 amino acids. In some embodiments, the spacer is a short spacer which comprises less than 100, less than 90, less than 80, less than 70, less than 60, less than 50, less than 40, less than 30, less than 20, or less than 10 amino acids.
The spacer may include a hinge region. In some embodiments, the hinge region may be located between the antigen binding domain and the transmembrane domain of the CAR. A hinge region can include at least a portion of a Fc region, for example, a hinge portion of a human Fc region of a CH3 domain or variants thereof. In some embodiments, the spacer includes all or part of an immunoglobulin (e.g., IgGl, IgG2, IgG3, IgG4) hinge region, i.e. the sequence that falls between the CHI and CH2 domains of an immunoglobulin, e.g., IgG4 Fc hinge or a CD8 hinge region. Examples include CD8 hinge, CD28 hinge, IgG4 (HL-CH3), or IgG4 (L235E, N297Q). In some embodiments, cysteines in the hinge region may be replaced with serines. Other examples of hinge regions are well known in the art.
In some embodiments, a short oligo- or polypeptide linker, between 2 and 10 amino acids in length, may connect two different domains of a CAR. For example, the linker may be located between the antigen binding domain and the transmembrane domain of the CAR, or between the transmembrane domain and the intracellular domain of the CAR. The linker may be a Gly linker, a Gly-Ser linker, a EAAAK (SEQ ID NO: 5) linker, a PAPAP (SEQ ID NO: 6) linker, or an (Ala- Pro)!! linker.
The length and amino acid composition of the linker peptide sequence can be optimized to vary the orientation and/or proximity of the polypeptide domains to one another to achieve a desired activity of the chimeric polypeptide. In some embodiments, the orientation and/or proximity of the polypeptide domains to one another can be varied as a “tuning” tool to achieve a tuning effect that would enhance or reduce the biological activity of the chimeric polypeptide.
In some embodiments, the linker is a synthetic compound linker such as, for example, a chemical cross-linking agent. Non-limiting examples of suitable cross-linking agents that are available on the market include N-hydroxysuccinimide (NHS), disuccinimidylsuberate (DSS), bis(sulfosuccinimidyl)suberate (BS3), dithiobis(succinimidylpropionate) (DSP), dithiobis(sulfosuccinimidylpropionate) (DTSSP), ethyleneglycol bis(succinimidylsuccinate) (EGS), ethyleneglycol bis(sulfosuccinimidyl)uccinate) (sulfo-EGS), disuccinimidyl tartrate (DST), disulfosuccinimidyl tartrate (sulfo-DST), bis [2 (succinimidooxycarbonyloxy)ethyl]sulfone (BSOCOES), and bis[2-(sulfosuccinimidooxycarbonyloxy)ethyl]sulfone (sulfo-BSOCOES).
Other examples of linkers are well known in the art.
A CAR useful for expression in modified myeloid-derived cells of the disclosure can further comprise a detectable moiety as would be known in the art and/or an effector molecule, nonlimiting examples of which include a drug, a toxin, a small molecule, an antibody, and/or an antibody fragment, singly or in any combination.
In some embodiments, a CAR may be glycosylated, pegylated, and/or otherwise post- translationally modified. Glycosylation, pegylation, and/or other post-translational modifications may
occur in vivo or in vitro and/or may be performed using chemical techniques. In some embodiments, any glycosylation, pegylation and/or other post-translational modifications may be N-linked or O- linked.
D. Expansion of Precursor Cells, Precursor CAR cells, Myeloid-Derived Cells, and/or Myeloid-Derived CAR Cells
Disclosed herein are methods of manufacturing a myeloid-derived cell from a precursor cell, such as a stem cell and/or a CD34+ cell. In some instances, the stem cell is a hematopoietic stem cell. The methods of manufacturing a myeloid-derived cell disclosed herein can comprise the step of contacting a precursor cell with a precursor media comprising a precursor cytokine mixture, whereby the number of precursor cell(s) increases to generate a cell population comprising a plurality of precursor cells, wherein said precursor cell is a stem cell.
In some embodiments, the methods of manufacturing a myeloid-derived cell disclosed herein can comprise the step of contacting a precursor cell with a precursor media comprising a precursor cytokine mixture, whereby the number of precursor cell(s) increases to generate a cell population comprising a plurality of precursor cells.
Also disclosed herein are methods of manufacturing a myeloid-derived cell disclosed herein comprising the step of contacting a precursor cell with a precursor media comprising a precursor cytokine mixture, whereby the number of precursor cell(s) increases to generate a cell population comprising a plurality of precursor cells, whereby upon contact of said precursor cell(s) with said precursor media, said precursor cell(s) differentiates into a myeloid-lineage cell to generate a myeloid-derived cell and/or a mixed cell population comprising a plurality of myeloid-derived cells and a plurality of precursor cells. Any cell and/or population comprising a plurality of cells can be expected to expand (i.e., increase in cell number) when contacted with and maintained in any of the precursor media described herein. As such, it would be expected that a myeloid-derived cell and/or a mixed cell population comprising a plurality of myeloid-derived cells and a plurality of precursor cells would expand in the precursor media. In such instances, the number of myeloid-derived cells within the precursor media would be expected to increase.
Also disclosed herein is a method of manufacturing a genetically modified myeloid-derived cell comprising a chimeric antigen receptor (CAR), wherein the method comprises: (a) contacting a precursor cell with a nucleic acid molecule comprising a polynucleotide sequence encoding the CAR to generate a genetically modified precursor CAR cell; and (b) expanding said genetically modified precursor CAR cell in a precursor media comprising a precursor cytokine mixture to generate a population comprising a plurality of genetically modified precursor CAR cells. The process of expanding the precursor CAR cell(s) can involve contacting said precursor CAR cell(s)
with said precursor media and/or said precursor cytokine mixture. The process of expanding the precursor CAR cell(s) can involve maintaining said precursor CAR cell(s) in a precursor media comprising a precursor cytokine mixture. The precursor CAR(s) can be contacted with said precursor media for any amount of time necessary to generate a population comprising a plurality of genetically modified precursor CAR cells. Any precursor media disclosed herein can be used.
Also disclosed herein is a method of manufacturing a genetically modified myeloid-derived cell comprising a chimeric antigen receptor (CAR), the method comprising the steps of: (a) contacting a precursor cell with a nucleic acid molecule comprising a polynucleotide sequence encoding a CAR, whereby said nucleic acid is introduced into said precursor cell, and (b) contacting said precursor cell with a precursor media comprising a precursor cytokine mixture, whereby the number of precursor cell(s) increases to generate a population of cells comprising a plurality of precursor CAR cells, wherein said precursor cell is a stem cell.
Also disclosed herein is precursor media and precursor cytokine mixtures that are utilized to expand a precursor cell. The process of expanding the precursor cell results in a cell population comprising a plurality of precursor cells. The precursor cell that undergoes expansion can be a genetically modified precursor CAR cell. The genetically modified precursor CAR cell undergoes genetic modification to introduce a CAR into the cell prior to expansion. As described herein, the precursor cell is a bone marrow-derived cell or a stem cell. In a preferred embodiment of the method disclosed herein, the precursor cell is a hematopoietic stem cell. Thus, the precursor cell that undergoes expansion is a genetically modified HSC CAR cell, and the genetically modified HSC CAR cell undergoes genetic modification to introduce a CAR into the cell prior to expansion. Once the precursor (e.g., HSC) cell is genetically modified to express the CAR, the genetically modified precursor CAR cell is expanded in a precursor media comprising a precursor cytokine mixture. Expansion of the HSC CAR cell can occur at any time following genetic modification of the HSC cell that will result in expansion of the genetically modified HSC CAR cell. For example, expansion can occur 1 day, 2 days, 3 days, 4 days, 5 days, 6 days, 7 days, 8 days, 9 days, 10 days, 11 days, 12 days, 13 days, 14 days, 15 days, 16 days, 17 days, 18 days, 19 days, 20 days, or more than 20 days following transduction. The number of days after transduction in which expansion should ideally occur will depend on the integration of the CAR into the precursor cell. For instance, it may be beneficial for the expansion process to occur once the CAR is suitable integrated into the precursor CAR cell. Expansion can also occur is a shorter amount of time following transduction, such as 2 hours, 4 hours, 6 hours, 8 hours, 10 hours, 12 hours, 14 hours, 16 hours, 18 hours, or 20 hours following CAR transduction.
The step of expanding the genetically modified precursor CAR cell disclosed herein is particularly effective at generating a population comprising a plurality of genetically modified precursor CAR cells. For example, the step of expanding the genetically modified HSC CAR cell disclosed herein results in a population comprising a plurality of genetically modified HSC CAR cells. The expansion step disclosed herein is also particularly effective at increasing the number of precursor cells. In some instances, the number of precursor cells within the precursor media is increased at a faster rate compared to precursor cells that are expanded in the absence of the precursor media disclosed herein. The precursor cells expanded in the precursor medium comprising the precursor cytokine mixture increase in number of cells over a short amount of time compared to precursor cells expanded in a medium lacking the precursor medium. Also disclosed herein is a precursor cytokine mixture that can be added to a precursor media. The precursor cytokine mixture contributes to increased expansion of the precursor cell(s). In some instances, the number of precursor cells is expanded at a faster rate in a precursor media comprising the precursor cytokine mixture described herein, compared to a precursor media lacking the precursor cytokine mixture of the present disclosure. Without wishing to be bound by theory, it is thought that the presence of the precursor cytokine mixture contributes to the superior technical effects of the precursor media (i.e., increased expansion rate and differentiation toward a myeloid lineage).
The precursor media comprising the precursor cytokine mixture is also particularly effective at promoting the differentiation of a genetically modified precursor CAR cell, or a plurality of genetically modified precursor CAR cells. Specifically, the precursor media promotes the differentiation of a precursor CAR cell, toward a myeloid-derived CAR cell, or a plurality of genetically modified myeloid-derived CAR cells, respectively. Contributing to the unexpected technical effect is the result that the precursor CAR cells can be expanded and differentiated in the precursor media comprising the precursor cytokine mixture. In some instances, the genetically modified precursor CAR cell(s) in the precursor media begin differentiating after the precursor CAR cell(s) have started expanding. In some instances, the genetically modified precursor CAR cell(s) in the precursor media begin differentiating at the same time the precursor CAR cell(s) are expanding. Thus, in addition to increasing the number of stem cells, for instance, hematopoietic stem cells, the step of expanding the genetically modified precursor CAR cell(s) in a precursor media also promotes the differentiation of the genetically modified precursor CAR cell(s) toward the myeloid lineage. The methods disclosed herein comprise the step of contacting said precursor cell with a precursor media comprising a precursor cytokine mixture, whereby the number of precursor cell(s) increases to generate a population of cells comprising a plurality of precursor CAR cells.
Expansion of at least one precursor cell can involve maintaining said at least one precursor cell in said precursor media. The precursor cell can be maintained in said precursor media for any suitable amount of time to generate a population of cells comprising a plurality of precursor CAR cells. Once a population of cells comprising a plurality of precursor CAR cells is produced within a precursor media, said population can be maintained in the precursor media for any suitable amount of time necessary to generate population comprising the specific characteristics that are desired. A skilled artisan would understand specific characteristics that are desired in a population. For instance, exemplary, nonlimiting, characteristics include: total cell number with the population or total number of a specific type of cell within the population (e.g., precursor cell, or myeloid-derived cell)). In some instances, the precursor CAR cell is maintained in said precursor media to increase the number of precursor CAR cells. In some instances, the precursor CAR cell is maintained in said precursor media to increase the number of precursor CAR cells or myeloid-derived CAR cells within the population.
As described herein, the precursor media disclosed herein, upon contact with a precursor cell, can promote expansion and differentiation of said precursor cell. In particular, the precursor media disclosed herein possess a superior effect of promoting differentiation of a precursor cell toward a myeloid lineage cell. Specifically, the precursor media described herein is particularly effective at promoting differentiation of a stem cell into a myeloid cell. The precursor media is particularly effective at promoting differentiation of a stem cell CAR cell into a myeloid-derived CAR cell. Accordingly, as a precursor cell is contacted with, and maintained in, a precursor media the number of precursor cells and myeloid-derived cells increases within the precursor media to generate a population of cells comprising a plurality of precursor cells and myeloid-derived cells. The precursor cells and myeloid-derived cells can be CAR cells. As such, the precursor media are effective at promoting expansion of a cell population comprising a plurality of precursor cells and myeloid-derived cells. When a cell population comprising a plurality of precursor cells and myeloid-derived cells is maintained in a precursor media, the precursor cells and myeloid-derived cells can be expanded. When a cell population comprising a plurality of precursor CAR cells and myeloid-derived CAR cells is maintained in a precursor media, the precursor cells and myeloid-derived cells can be expanded. Expansion of genetically modified precursor CAR cells can increase the amount of cells by about 10 fold, 20 fold, 30 fold, 40 fold, 50 fold, 60 fold, 70 fold, 80 fold, 90 fold, 100 fold, 200 fold, 300 fold, 400 fold, 500 fold, 600 fold, 700 fold, 800 fold, 900 fold, 1000 fold, 2000 fold, 3000 fold, 4000 fold, 5000 fold, 6000 fold, 7000 fold, 8000 fold, 9000 fold, 10,000 fold, 100,000 fold, 1,000,000 fold, 10,000,000 fold, or greater. In some embodiments, the cells are expanded in the range of about 20 fold to about 300 fold, about 20 fold to about 250 fold, about 20 fold to about 200 fold, about 20 fold to about 150 fold, about 20 fold to about 100 fold, about 20 fold to about 50 fold. In some embodiments,
expansion of genetically modified precursor CAR cells can increase the amount of precursor CAR cells between 10-fold to 100-fold, between 20-fold to 200-fold, between 30-fold to 300-fold, between 40-fold to 400-fold, between 50-fold to 500-fold, between 60-fold to 600-fold, between 70-fold to 700-fold, between 80-fold to 800-fold, between 90-fold to 900-fold, between 100-fold to 1000-fold, or greater than 1000-fold.
The expansion step can be very short, for example less than 24 hours such as 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, or 23 hours. The expansion step can be longer, for example 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, or more days. In some embodiments, the cells may be expanded for several hours (about 3 hours) to about 14 days or any hourly integer value in between. In some embodiments, the genetically modified precursor CAR cell(s) is expanded for at least 1, at least 2, at least 3, at least 4, at least 5, at least 6 days, at least 7 days, at least 8 days, at least 9 days, at least 10 days, at least 11 days, at least 12 days, at least 13 days, at least 14 days, at least 15 days, at least 16 days, at least 17 days, at least 18 days, at least 19 days, at least 20 days, or more than 20 days. In some embodiments, the genetically modified precursor CAR cell(s) is expanded for 1 day, for 2 days, for 3 days, for 4 days, for 5 days, for 6 days, for 7 days, for 8 days, for 9 days, for 10 days, for 11 days, for 12 days, for 13 days, for 14 days, for 15 days, or for more than 15 days. In some embodiments, the precursor CAR cell, or population comprising the same, is maintained in precursor media for 1 day. In some embodiments, the precursor CAR cell, or population comprising the same, is maintained in precursor media for 2 days. In some embodiments, the precursor CAR cell, or population comprising the same, is maintained in precursor media for 3 days. In some embodiments, the precursor CAR cell, or population comprising the same, is maintained in precursor media for 4 days. In some embodiments, the precursor CAR cell, or population comprising the same, is maintained in precursor media for 5 days. In some embodiments, the precursor CAR cell, or population comprising the same, is maintained in precursor media for 6 days. In some embodiments, the precursor CAR cell, or population comprising the same, is maintained in precursor media for 7 days. In some embodiments, the precursor CAR cell, or population comprising the same, is maintained in precursor media for 8 days. In some embodiments, the precursor CAR cell, or population comprising the same, is maintained in precursor media for 9 days. In some embodiments, the precursor CAR cell, or population comprising the same, is maintained in precursor media for 10 days. In some embodiments, the precursor CAR cell, or population comprising the same, is maintained in precursor media for 11 days. In some embodiments, the precursor CAR cell, or population comprising the same, is maintained in precursor media for 12 days. In some embodiments, the precursor CAR cell, or population comprising the same, is maintained in
precursor media for 13 days. In some embodiments, the precursor CAR cell, or population comprising the same, is maintained in precursor media for 14 days. In some embodiments, the precursor CAR cell, or population comprising the same, is maintained in precursor media for 15 days. In some embodiments, the precursor CAR cell, or population comprising the same, is maintained in precursor media for 16 days. In some embodiments, the precursor CAR cell, or population comprising the same, is maintained in precursor media for 17 days. In some embodiments, the precursor CAR cell, or population comprising the same, is maintained in precursor media for 18 days. In some embodiments, the precursor CAR cell, or population comprising the same, is maintained in precursor media for 19 days. In some embodiments, the precursor CAR cell, or population comprising the same, is maintained in precursor media for 20 days.
One key aspect of the expansion step is to increase the number of precursor cell(s), and/or myeloid-derived cells derived therefrom, the number of myeloid-derived cells within a precursor media can be expected to increase as well. The expansion step of the methods disclosed herein can increase the amount of the differentiated cells by about 10 fold, 20 fold, 30 fold, 40 fold, 50 fold, 60 fold, 70 fold, 80 fold, 90 fold, 100 fold, 200 fold, 300 fold, 400 fold, 500 fold, 600 fold, 700 fold, 800 fold, 900 fold, 1000 fold, 2000 fold, 3000 fold, 4000 fold, 5000 fold, 6000 fold, 7000 fold, 8000 fold, 9000 fold, 10,000 fold, 100,000 fold, 1,000,000 fold, 10,000,000 fold, or greater. In some embodiments, expansion can increase the amount of myeloid-derived CAR cells between 10-fold to 100-fold, between 20-fold to 200-fold, between 30-fold to 300-fold, between 40-fold to 400-fold, between 50-fold to 500-fold, between 60-fold to 600-fold, between 70-fold to 700-fold, between 80- fold to 800-fold, between 90-fold to 900-fold, between 100-fold to 1000-fold, or greater than 1000- fold. Without wishing to be bound by theory, as the number of differentiated cells within a population, increases, the number of undifferentiated cells within the population decreases. For example, as the number of genetically modified precursor CAR cell(s) that are differentiated in a precursor media increases, the number of genetically modified precursor CAR cell(s) that are undifferentiated in the precursor media decreases. Differentiation of genetically modified precursor CAR cells can increase the ratio of the differentiated cells (i.e., myeloid-derived cells) to undifferentiated cells (i.e., stem cells) within a population by a factor of 10 fold, 20 fold, 30 fold, 40 fold, 50 fold, 60 fold, 70 fold, 80 fold, 90 fold, 100 fold, 200 fold, 300 fold, 400 fold, 500 fold, 600 fold, 700 fold, 800 fold, 900 fold, 1000 fold, 2000 fold, 3000 fold, 4000 fold, 5000 fold, 6000 fold, 7000 fold, 8000 fold, 9000 fold, 10,000 fold, 100,000 fold, 1,000,000 fold, 10,000,000 fold, or greater.
In some embodiments, the precursor CAR cell(s) is maintained in said precursor medium until the number of cells is at least 104, at least 105, at least 106, at least 107, at least 108, at least 109, at least
IO10, at least IO11, at least 1012, at least IO13, at least 1014, at least IO15, at least 1016, at least 1017, at least 1018, at least 1019, at least IO20, or greater than IO20. In some embodiments, the precursor CAR cell(s) is maintained in said precursor medium until the number of cells within the generated population is at least 104, at least 105, at least 106, at least 107, at least 108, at least 109, at least IO10, at least IO11, at least 1012, at least IO13, at least 1014, at least IO15, at least 1016, at least 1017, at least 1018, at least 1019, at least IO20, or greater than IO20. In some embodiments, the precursor CAR cell(s) is maintained in said precursor medium to generate a population of cells comprising a plurality of precursor CAR cells and/or a plurality of myeloid-derived CAR cells, wherein the number of cells within the population is at least 104, at least 105, at least 106, at least 107, at least 108, at least 109, at least IO10, at least 1011, at least 1012, at least 1013, at least 1014, at least 1015, at least 1016, at least 1017, at least 1018, at least 1019, at least IO20, or greater than IO20. In some embodiments, the precursor CAR cell(s) is maintained in the precursor medium for at least 2, at least 3, at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, at least 10, at least 11, or at least 12 days. In some embodiments, the precursor CAR cell(s) are maintained in precursor media for between 9-11 days. In some embodiments, the precursor CAR cell(s) are maintained in precursor media for 10 days. In some embodiments, said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population, is maintained in said precursor media for 1 day. In some embodiments, said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population, is maintained in said precursor media for 2 days. In some embodiments, said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population, is maintained in said precursor media for 3 days. In some embodiments, said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population, is maintained in said precursor media for 4 days. In some embodiments, said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population, is maintained in said precursor media for 5 days. In some embodiments, said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population, is maintained in said precursor media for 6 days. In some embodiments, said precursor cell(s) and/or said population comprising a plurality of precursor cells,
said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population, is maintained in said precursor media for 7 days. In some embodiments, said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population, is maintained in said precursor media for 8 days. In some embodiments, said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population, is maintained in said precursor media for 9 days. In some embodiments, said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population, is maintained in said precursor media for 10 days. In some embodiments, said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population, is maintained in said precursor media for 11 days. In some embodiments, said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population, is maintained in said precursor media for 12 days. In some embodiments, said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population, is maintained in said precursor media for 13 days. In some embodiments, said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population, is maintained in said precursor media for 14 days. In some embodiments, said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population, is maintained in said precursor media for 15 days. In some embodiments, said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population, is maintained in said precursor media for 16 days. In some embodiments, said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population, is maintained in said precursor media for more than 16 days.
In some embodiments, said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of
precursor CAR cells, and/or said mixed cell population, is maintained in precursor media for between 12-48 hours. In some embodiments, said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population, is maintained in precursor media for 24-72 hours. In some embodiments, said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population, is maintained in precursor media for between 48-96 hours. In some embodiments, said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population, is maintained in precursor media for between 72-120 hours. In some embodiments, said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population, is maintained in precursor media for between 96-144 hours. In some embodiments, said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population, is maintained in precursor media for between 120-168 hours. In some embodiments, said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population, is maintained in precursor media for between 144-192 hours. In some embodiments, said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population, is maintained in precursor media for between 168-216 hours. In some embodiments, said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population, is maintained in precursor media for between 192-240 hours. In some embodiments said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population, is maintained in precursor media for between 216-264 hours. In some embodiments, said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population, is maintained in precursor media for between 240-288 hours.
In some embodiments, said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population, is maintained in precursor media for between 264-312 hours. In some embodiments, said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population, is maintained in precursor media for between 288-336 hours. In some embodiments, said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population, is maintained in precursor media for between 312-360 hours. In some embodiments, said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population, is maintained in precursor media for between 336-384 hours. In some embodiments, said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population, is maintained in precursor media for between 360-408 hours. In some embodiments, said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population, is maintained in precursor media for between 384-432 hours. In some embodiments, said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population, is maintained in precursor media for between 408-456 hours. In some embodiments, said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population, is maintained in precursor media for more than 456 hours.
The precursor media and myeloid media disclosed herein can comprise a mixed population of cells. The precursor media and myeloid media disclosed herein are particularly effective at generating a mixed population of cells. For instance, when a precursor cell and/or a precursor CAR cell, for example, are contacted with, and maintained in a precursor media, said precursor cell and/or said precursor CAR cell, can expand to generate a cell population comprising a plurality of said precursor cells and/or said precursor CAR cells, and/or said differentiate into a myeloid- derived cell and/or myeloid-derived CAR cell. In some embodiments, wherein said precursor CAR cell expands to generate a cell population comprising a plurality of said precursor CAR cells, said
precursor CAR cells within said plurality of precursor CAR cells can expand to increase the number of precursor CAR cells within said population and/or differentiate into a myeloid-derived CAR cell(s). A skilled artisan would readily understand that within a precursor media, a plurality of precursor CAR cells (or precursor cells) can be undergoing expansion and/or differentiation, such that the number of precursor CAR cells is increasing and the number of myeloid-derived CAR cells generated from said precursor CAR cells is increasing, thereby generating a mixed population of cells comprising a plurality of precursor CAR cells and myeloid-derived CAR cells. Any precursor CAR cell within a precursor media can be expected to expand and/or differentiate. Any myeloid- derived CAR cell within said precursor media can be expected to expand and/or differentiate. Without wishing to be bound by any theory, as the number of precursor CAR cells within a population present in a precursor media increases, the number of precursor CAR cells that can and/or will differentiate into myeloid-derived CAR cells increases accordingly. As such, as the number of precursor CAR cells within a precursor media increases, the rate at which the number of myeloid-derived CAR cells within the population increases can increase.
The term “mixed population” as used herein refers to any cell population comprising a plurality of myeloid-derived cells and/or a plurality of precursor cells, and/or a plurality of myeloid-derived CAR cells and/or a plurality of precursor CAR cells. The mixed population can comprise a plurality of myeloid-derived cells and/or a plurality of precursor cells. In some embodiments, the mixed population comprises a plurality of myeloid-derived cells and a plurality of precursor cells. The mixed population can comprise a plurality of myeloid-derived CAR cells and/or a plurality of precursor CAR cells. In some embodiments, the mixed population comprises a plurality of myeloid-derived CAR cells and a plurality of precursor CAR cells. When the mixed population comprises comprises a plurality of myeloid-derived CAR cells and/or a plurality of precursor CAR cells, it would be understood that not every myeloid-derived cell and/or precursor cell within the population would comprise a CAR (i.e., at least one cell within said population will not comprise a CAR). In a preferred embodiment, when the mixed population comprises a plurality of myeloid-derived CAR cells and a plurality of precursor CAR cells, at least 70%, at least 71%, at least 72%, at least 73%, at least 74%, at least 75%, at least 76%, at least 77%, at least 78%, at least 79%, at least 80%, at least 81%, at least 82%, at least 83%, at least 84%, at least 85%, at least 86%, at least 87%, at least 88%, at least 89%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or greater than 99% of cells within said population comprises a CAR. In such instances, at least 70%, at least 71%, at least 72%, at least 73%, at least 74%, at least 75%, at least 76%, at least 77%, at least 78%, at least 79%, at least 80%, at least 81%, at least 82%, at least 83%, at least 84%, at least 85%, at least 86%,
at least 87%, at least 88%, at least 89%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or greater than 99% of cells within said plurality of myeloid-derived CAR cells within said population comprises a CAR. In such instances, at least 70%, at least 71%, at least 72%, at least 73%, at least 74%, at least 75%, at least 76%, at least 77%, at least 78%, at least 79%, at least 80%, at least 81%, at least 82%, at least 83%, at least 84%, at least 85%, at least 86%, at least 87%, at least 88%, at least 89%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or greater than 99% of cells within said plurality of precursor CAR cells within said population comprises a CAR.
The differentiated cells of the present disclosure are preferential differentiated toward the myeloid cell lineage. In some instances, the step of differentiating a genetically modified precursor CAR cell in a precursor media generates a myeloid-derived CAR cell. In some instances, the step of differentiating a genetically modified precursor CAR cell(s) in a precursor media generates a plurality of genetically modified myeloid-derived CAR cells. As the plurality of genetically modified precursor CAR cells is differentiated toward the myeloid cell lineage (i.e., myeloid-derived cell), the ratio of precursor (e.g., stem cells) within the plurality is decreased relative to myeloid-derived cells. For example, the ratio of CD34+ cells decreases as the plurality of precursor CAR cells differentiates. Differentiation can decrease the amount of CD34+ cells within a plurality of genetically modified precursor CAR cells or a plurality of genetically modified myeloid-derived cells. Differentiation of genetically modified precursor CAR cells can decrease the amount of CD34+ cells within a plurality of genetically modified precursor CAR cells or a plurality of genetically modified myeloid-derived cells by about 10 fold, 20 fold, 30 fold, 40 fold, 50 fold, 60 fold, 70 fold, 80 fold, 90 fold, 100 fold, 200 fold, 300 fold, 400 fold, 500 fold, 600 fold, 700 fold, 800 fold, 900 fold, 1000 fold, 2000 fold, 3000 fold, 4000 fold, 5000 fold, 6000 fold, 7000 fold, 8000 fold, 9000 fold, 10,000 fold, 100,000 fold, 1,000,000 fold, 10,000,000 fold, or greater. Myeloid cells can be identified and/or isolated based on cell surface markers. A skilled artisan would understand that specific myeloid cells within the myeloid lineage can be identified based on specific cell surface markers. Exemplary, non-limiting, surface markers for myeloid cells include: CD13, CD14, CD15 and CD1 lb. Accordingly, during the expansion step, the number of cells expression myeloid-associated cell surface markers can be expected to increase, while the number of cells expressing a common stem cell surface marker (i.e., CD34) can be expected to decrease as differentiation occurs.
The expansion step of the methods disclosed herein can increase the amount of CD1 lb+ cells within a plurality of genetically modified precursor CAR cells or a plurality of genetically modified myeloid-derived cells. Differentiation of genetically modified precursor CAR cells can increase the
amount of CD1 lb+ cells within a plurality of genetically modified precursor CAR cells or a plurality of genetically modified myeloid-derived cells by about 10 fold, 20 fold, 30 fold, 40 fold, 50 fold, 60 fold, 70 fold, 80 fold, 90 fold, 100 fold, 200 fold, 300 fold, 400 fold, 500 fold, 600 fold, 700 fold, 800 fold, 900 fold, 1000 fold, 2000 fold, 3000 fold, 4000 fold, 5000 fold, 6000 fold, 7000 fold, 8000 fold, 9000 fold, 10,000 fold, 100,000 fold, 1,000,000 fold, 10,000,000 fold, or greater. In some embodiments, a precursor CAR cell, or population comprising a plurality of precursor CAR cells, or a population comprising a plurality of precursor CAR cells and a plurality of myeloid-derived CAR cells is maintained in a precursor media until the number of CD1 lb+ cells within the population is increased between 10 fold to 100 fold, between 20 fold and 200 fold, between 30 fold and 300 fold, between 40 fold and 400 fold, between 50 fold and 500 fold, between 60 fold and 600 fold, between 70 fold and 700 fold, between 80 fold and 800 fold, between 90 fold and 900 fold, between 100 fold and 10,000 fold, between 1000 fold and 100,000 fold, between 10,000 fold and 1,000,000 fold, between 100,000 fold and 10,000,000 fold , between 105 fold and 107 fold, between 106 fold and 108 fold, between 107 fold and 109 fold, 108 fold and 1010 fold, 109 fold and 1011 fold, 1010 fold and 1012 fold, 1011 fold and 1013 fold, 1012 fold and 1014 fold, 1013 fold and 1015 fold, 1014 fold and 1016 fold, or greater than 1016 fold.
The expansion step of the methods disclosed herein can increase the amount of CD14+ cells within a plurality of genetically modified precursor CAR cells or a plurality of genetically modified myeloid-derived cells. Differentiation of genetically modified precursor CAR cells can increase the amount of CD14+ cells within a plurality of genetically modified precursor CAR cells or a plurality of genetically modified myeloid-derived cells by about 10 fold, 20 fold, 30 fold, 40 fold, 50 fold, 60 fold, 70 fold, 80 fold, 90 fold, 100 fold, 200 fold, 300 fold, 400 fold, 500 fold, 600 fold, 700 fold, 800 fold, 900 fold, 1000 fold, 2000 fold, 3000 fold, 4000 fold, 5000 fold, 6000 fold, 7000 fold, 8000 fold, 9000 fold, 10,000 fold, 100,000 fold, 1,000,000 fold, 10,000,000 fold, or greater. In some embodiments, a precursor CAR cell, or population comprising a plurality of precursor CAR cells, or a population comprising a plurality of precursor CAR cells and a plurality of myeloid-derived CAR cells is maintained in a precursor media until the number of CD14+ cells within the population is increased between 10 fold to 100 fold, between 20 fold and 200 fold, between 30 fold and 300 fold, between 40 fold and 400 fold, between 50 fold and 500 fold, between 60 fold and 600 fold, between 70 fold and 700 fold, between 80 fold and 800 fold, between 90 fold and 900 fold, between 100 fold and 10,000 fold, between 1000 fold and 100,000 fold, between 10,000 fold and 1,000,000 fold, between 100,000 fold and 10,000,000 fold , between 105 fold and 107 fold, between 106 fold and 108 fold, between 107 fold and 109 fold, 108 fold and 1010 fold, 109 fold and 1011 fold, 1010 fold and 1012 fold, 1011 fold and
1013 fold, 1012 fold and 1014 fold, 1013 fold and 1015 fold, 1014 fold and 1016 fold, or greater than 1016 fold.
The expansion step of the methods disclosed herein can increase the amount of CD 13+ cells within a plurality of genetically modified precursor CAR cells or a plurality of genetically modified myeloid-derived cells. Differentiation of genetically modified precursor CAR cells can increase the amount of CD 13+ cells within a plurality of genetically modified precursor CAR cells or a plurality of genetically modified myeloid-derived cells by about 10 fold, 20 fold, 30 fold, 40 fold, 50 fold, 60 fold, 70 fold, 80 fold, 90 fold, 100 fold, 200 fold, 300 fold, 400 fold, 500 fold, 600 fold, 700 fold, 800 fold, 900 fold, 1000 fold, 2000 fold, 3000 fold, 4000 fold, 5000 fold, 6000 fold, 7000 fold, 8000 fold, 9000 fold, 10,000 fold, 100,000 fold, 1,000,000 fold, 10,000,000 fold, or greater. In some embodiments, a precursor CAR cell, or population comprising a plurality of precursor CAR cells, or a population comprising a plurality of precursor CAR cells and a plurality of myeloid-derived CAR cells is maintained in a precursor media until the number of CD 13+ cells within the population is increased between 10 fold to 100 fold, between 20 fold and 200 fold, between 30 fold and 300 fold, between 40 fold and 400 fold, between 50 fold and 500 fold, between 60 fold and 600 fold, between 70 fold and 700 fold, between 80 fold and 800 fold, between 90 fold and 900 fold, between 100 fold and 10,000 fold, between 1000 fold and 100,000 fold, between 10,000 fold and 1,000,000 fold, between 100,000 fold and 10,000,000 fold , between 105 fold and 107 fold, between 106 fold and 108 fold, between 107 fold and 109 fold, 108 fold and 1010 fold, 109 fold and 1011 fold, 1010 fold and 1012 fold, 1011 fold and 1013 fold, 1012 fold and 1014 fold, 1013 fold and 1015 fold, 1014 fold and 1016 fold, or greater than 1016 fold.
The expansion step of the methods disclosed herein can increase the amount of CD 15+ cells within a plurality of genetically modified precursor CAR cells or a plurality of genetically modified myeloid-derived cells. Differentiation of genetically modified precursor CAR cells can increase the amount of CD 15+ cells within a plurality of genetically modified precursor CAR cells or a plurality of genetically modified myeloid-derived cells by about 10 fold, 20 fold, 30 fold, 40 fold, 50 fold, 60 fold, 70 fold, 80 fold, 90 fold, 100 fold, 200 fold, 300 fold, 400 fold, 500 fold, 600 fold, 700 fold, 800 fold, 900 fold, 1000 fold, 2000 fold, 3000 fold, 4000 fold, 5000 fold, 6000 fold, 7000 fold, 8000 fold, 9000 fold, 10,000 fold, 100,000 fold, 1,000,000 fold, 10,000,000 fold, or greater. In some embodiments, a precursor CAR cell, or population comprising a plurality of precursor CAR cells, or a population comprising a plurality of precursor CAR cells and a plurality of myeloid- derived CAR cells is maintained in a precursor media until the number of CD 15+ cells within the population is increased between 10 fold to 100 fold, between 20 fold and 200 fold, between 30 fold and 300 fold, between 40 fold and 400 fold, between 50 fold and 500 fold, between 60 fold and 600
fold, between 70 fold and 700 fold, between 80 fold and 800 fold, between 90 fold and 900 fold, between 100 fold and 10,000 fold, between 1000 fold and 100,000 fold, between 10,000 fold and 1,000,000 fold, between 100,000 fold and 10,000,000 fold , between 105 fold and 107 fold, between 106 fold and 108 fold, between 107 fold and 109 fold, 108 fold and 1010 fold, 109 fold and 1011 fold, 1010 fold and 1012 fold, 1011 fold and 1013 fold, 1012 fold and 1014 fold, 1013 fold and 1015 fold, 1014 fold and 1016 fold, or greater than 1016 fold.
The amount of cell surface marker expression within a plurality of genetically modified precursor CAR cells or plurality of plurality of genetically modified myeloid-derived CAR cells can be used to determine how long to maintain the expanding step of the method of manufacturing a genetically modified myeloid-derived CAR cells disclosed herein. In some instances, the expanding step is maintained until a certain percentage of the population comprising a plurality of genetically modified precursor CAR cells and/or genetically modified myeloid-derived CAR cells expresses a specific marker. In some instances, the expanding step is maintained until a certain percentage of the population comprising a plurality of genetically modified precursor CAR cells and/or plurality of genetically modified myeloid-derived cells expresses at least a specific marker. In some instances, the expanding step is maintained until a certain percentage of the population comprising a plurality of genetically modified precursor CAR cells and/or plurality of genetically modified myeloid-derived CAR cells expresses at least two specific markers. In some instances, the expanding step is maintained until a certain percentage of the population comprising a plurality of genetically modified precursor CAR cells and/or plurality of genetically modified myeloid-derived CAR cells expresses at least three specific markers. In some instances, the expanding step is maintained until a certain percentage of the population comprising a plurality of genetically modified precursor CAR cells and/or plurality of genetically modified myeloid-derived CAR cells expresses at least more than three different specific markers.
Disclosed herein are methods of generating a genetically modified myeloid-derived CAR cell from a precursor cell, such as a hematopoietic stem cell, wherein the precursor cell comprising a CAR (i.e., precursor CAR cell) is expanded by contacting said precursor CAR cell with a precursor media comprising a precursor cytokine mixture, whereby the number of precursor CAR cell(s) increases to generate a population of precursor cells (e.g., stem cells) comprising a plurality of precursor CAR cells, wherein following contact of said precursor CAR cell(s) within said population with said precursor media, said precursor CAR cell(s) differentiates into a myeloid-lineage cell(s) to generate a myeloid-derived CAR cell(s) or a population comprising a plurality of myeloid-derived CAR cell(s). In some instances, the cell population is a mixed population comprising said myeloid-derived CAR cells and a plurality of precursor CAR cells. Importantly, a skilled artisan would understand that as a
precursor CAR cell is maintained in a precursor media, said cell will expand to generate a population comprising a plurality of precursor CAR cells through the process of cell division (i.e., the process by which a single parent cell splits to form two new cells) and/or differentiate into a myeloid-derived CAR cell and/or both. Accordingly, for the purposes of the present application, any reference to “a precursor cell” that after being maintained in a precursor media has generated a cell population is intended to encompass the cell population generated. A cell population would be understood to have been generated from a precursor cell if, as described above, said population comprises at least one cell that was generated from a cell division and/or a cell differentiation of said precursor cell. In some cases, the population is a mixed population, as described herein. In some embodiments, the population is exclusively precursor cells. In some embodiments, the population is exclusively myeloid-derived cells.
In some instances, the expanding step is maintained until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population comprising a plurality of myeloid-derived CAR cells is CD1 lb+. In some instances, the expanding step is maintained until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population comprising a plurality of genetically modified precursor CAR cells and/or a plurality of genetically modified myeloid-derived CAR cells is CD1 lb+. In some instances, the precursor CAR cell(s) and/or population comprising a plurality of precursor CAR cells is maintained in said precursor media until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population is CD1 lb+. In some instances, the precursor CAR cell(s) and/or population comprising a plurality of precursor CAR cells and/or plurality of myeloid-derived CAR cells is maintained in said precursor media until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population is CD1 lb+. In some instances, the population comprising a plurality of myeloid-derived CAR cells is maintained in said precursor media until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population is CD1 lb+.
In some instances, the precursor CAR cell(s) and/or population comprising a plurality of precursor CAR cells and/or plurality of myeloid-derived CAR cells is maintained in said precursor media until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at
least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population is CD1 lb+CD13+.
In some instances, the precursor CAR cell(s) and/or population comprising a plurality of precursor CAR cells and/or plurality of myeloid-derived CAR cells is maintained in said precursor media until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population is CD1 lb+CD13+CD14+.
In some instances, the precursor CAR cell(s) and/or population comprising a plurality of precursor CAR cells and/or plurality of myeloid-derived CAR cells is maintained in said precursor media until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population is CDl lb+CD13+CD15+.
In some instances, the precursor CAR cell(s) and/or population comprising a plurality of precursor CAR cells and/or plurality of myeloid-derived CAR cells is maintained in said precursor media until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population is CDl lb+CD13+CD14+CD15+.
In some instances, the precursor CAR cell(s) and/or population comprising a plurality of precursor CAR cells and/or plurality of myeloid-derived CAR cells is maintained in said precursor media until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population is CD1 lb+CD14+.
In some instances, the precursor CAR cell(s) and/or population comprising a plurality of precursor CAR cells and/or plurality of myeloid-derived CAR cells is maintained in said precursor media until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population is CD1 lb+CD15+.
In some instances, the precursor CAR cell(s) and/or population comprising a plurality of precursor CAR cells and/or plurality of myeloid-derived CAR cells is maintained in said precursor media until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population is CD1 lb+CD14+CD15+.
In some instances, the expanding step is maintained until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least
80%, at least 90%, or at least 95%, or at least 99% of the cell population comprising a plurality of myeloid-derived CAR cells is CD14+. In some instances, the expanding step is maintained until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population comprising a plurality of genetically modified precursor CAR cells and/or plurality of genetically modified myeloid-derived CAR cells is CD14+. In some instances, the precursor CAR cell(s) and/or population comprising a plurality of precursor CAR cells is maintained in said precursor media until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population is CD14+. In some instances, the precursor CAR cell(s) and/or population comprising a plurality of precursor CAR cells and/or plurality of myeloid-derived CAR cells is maintained in said precursor media until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population is CD14+. In some instances, the population comprising a plurality of myeloid-derived CAR cells is maintained in said precursor media until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population is CD14+.
In some instances, the precursor CAR cell(s) and/or population comprising a plurality of precursor CAR cells and/or plurality of myeloid-derived CAR cells is maintained in said precursor media until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population is CD14+CD13+.
In some instances, the precursor CAR cell(s) and/or population comprising a plurality of precursor CAR cells and/or plurality of myeloid-derived CAR cells is maintained in said precursor media until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population is CD14+CD15+.
In some instances, the precursor CAR cell(s) and/or population comprising a plurality of precursor CAR cells and/or plurality of myeloid-derived CAR cells is maintained in said precursor media until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population is CD14+CD13+CD15+.
In some instances, the expanding step is maintained until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population comprising a plurality of myeloid- derived CAR cells is CD13+. In some instances, the expanding step is maintained until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population comprising a plurality of genetically modified precursor CAR cells and/or plurality of genetically modified myeloid- derived CAR cells is CD13+. In some instances, the precursor CAR cell(s) and/or population comprising a plurality of precursor CAR cells is maintained in said precursor media until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population is CD13+. In some instances, the precursor CAR cell(s) and/or population comprising a plurality of precursor CAR cells and/or plurality of myeloid-derived CAR cells is maintained in said precursor media until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population is CD13+. In some instances, the population comprising a plurality of myeloid-derived CAR cells is maintained in said precursor media until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population is CD13+. In some instances, the precursor CAR cell(s) and/or population comprising a plurality of precursor CAR cells and/or plurality of myeloid-derived CAR cells is maintained in said precursor media until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population is CD 13+CD 15+
In some instances, the expanding step is maintained until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population comprising a plurality of myeloid-derived CAR cells is CD15+. In some instances, the expanding step is maintained until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population comprising a plurality of genetically modified precursor CAR cells and/or plurality of genetically modified myeloid-derived CAR cells is CD15+. In some instances, the precursor CAR cell(s) and/or population comprising a plurality of precursor CAR cells is maintained in said precursor media until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least
99% of the cell population is CD15+. In some instances, the precursor CAR cell(s) and/or population comprising a plurality of precursor CAR cells and/or plurality of myeloid-derived CAR cells is maintained in said precursor media until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population is CD15+. In some instances, the population comprising a plurality of myeloid-derived CAR cells is maintained in said precursor media until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population is CD15+.
In some instances, the expanding step is maintained until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the population comprising a plurality of genetically modified precursor CAR cells or the population comprising a plurality of genetically modified myeloid-derived CAR cells is CD13+CD15+.
In some instances, the ratio of differentiated cells to undifferentiated cells within the population increases during and after the expansion step of the methods disclosed herein. The expansion step can increase the amount of CD1 lb+ cells within a plurality of genetically modified precursor CAR cells or a plurality of genetically modified myeloid-derived cells.
Following the expansion step, the cells can be incubated in cell medium in a culture apparatus for a period of time or until the cells reach confluency or high cell density for optimal passage before passing the cells to another culture apparatus. The culturing apparatus can be any culture apparatus commonly used for culturing cells in vitro. In some embodiments, the level of confluence is 70% or greater before passing the cells to another culture apparatus. In some embodiments, the level of confluence is 90% or greater. A period of time can be any time suitable for the culture of cells in vitro. The culture medium may be replaced during the culture of the cells at any time. In some embodiments, the culture medium is replaced about every 2 to 3 days. The cells are then harvested from the culture apparatus whereupon the cells can be used immediately or stored for use at a later time.
Examples of cell markers that can be used for myeloid-derived cells include, but are not limited to CD1 lb, CD13, CD14 and CD15. In some instances, the myeloid-derived cells can express at least one surface marker selected from the following: CD1 lb, CD13, CD14, and CD15. In some instances, the myeloid-derived cells can express at least two surface markers selected from the following: CD1 lb, CD13, CD14, and CD15. In some instances, the myeloid-derived cells can express at least three surface markers selected from the following: CDl lb, CD13, CD14, and CD15. In some
instances, the myeloid-derived cells express the following surface markers: CD1 lb, CD13, CD14, and CD15.
The expanding step can be very short, for example less than 24 hours such as 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, or 23 hours. The expanding step can be longer, for example 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, or more days. In some embodiments, the expansion step may be maintained for several hours (about 3 hours) to about 14 days or any hourly integer value in between. During the expanding step, the precursor CAR cell is maintained in the precursor medium. In some embodiments, the expansion step is maintained for at least 1, at least 2, at least 3, at least 4, or at least 5 days. In some embodiments, the expansion step is maintained for 1 day, for 2 days, for 3 days, for 4 days, or for 5 days. In some embodiments, the expanding step can be about 1 day. In some embodiments, the expanding step is maintained for about 2 days. In some embodiments, the expanding step is maintained for about 3 days. In some embodiments, the expanding step is maintained for about 4 days. In some embodiments, the expanding step is maintained for about 5 days. In some embodiments, the expanding step is maintained for about 6 days. In some embodiments, the expanding step is maintained for about 7 days. In some embodiments, the expanding step is maintained for about 8 days. In some embodiments, the expanding step is maintained for about 9 days. In some embodiments, the expanding step is maintained for about 10 days. In some embodiments, the expanding step is maintained for about 11 days. In some embodiments, the expanding step is maintained for about 12 days. In some embodiments, the expanding step is maintained for about 13 days. In some embodiments, the expanding step is maintained for about 14 days. In some embodiments, the expanding step is maintained for about 15 days. In some embodiments, the expanding step is maintained for at least 2, at least 3, at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, at least 10, at least 11, or at least 12 days. In some embodiments, the expanding step is maintained for between 9-11 days. In some embodiments, the expanding step is maintained for 10 days.
The expanding step (i.e., maintaining a precursor CAR cell, and/or a population comprising a plurality of precursor CAR cells and/or a plurality of myeloid-derived cells in precursor media) can also be maintained for a set number of hours. In some embodiments, the expanding step is maintained for about 12 hours. In some embodiments, the expanding step is maintained for about 24 hours. In some embodiments, the expanding step is maintained for about 48 hours. In some embodiments, the expanding step is maintained for about 72 hours. In some embodiments, the expanding step is maintained for about 96 hours. In some embodiments, the expanding step is maintained for about 120 hours. In some embodiments, the expanding step is maintained for about 144 hours. In some embodiments, the expanding step is maintained for about 168 hours. In some embodiments, the
expanding step is maintained for about 192 hours. In some embodiments, the expanding step is maintained for about 216 hours. In some embodiments, the expanding step is maintained for about 240 hours. In some embodiments, the expanding step is maintained for about 264 hours. In some embodiments, the expanding step is maintained for about 288 hours. In some embodiments, the expanding step is maintained for about 312 hours. In some embodiments, the expanding step is maintained for about 336 hours. In some embodiments, the expanding step is maintained for about 360 hours. In some embodiments, the expanding step is maintained for about 384 hours. In some embodiments, the expanding step is maintained for about 408 hours. In some embodiments, the expanding step is maintained for about 432 hours. In some embodiments, the expanding step is maintained for about 456 hours. In some embodiments, the expanding step is maintained for more than 456 hours.
In some embodiments, the expanding step is maintained between 12-48 hours. In some embodiments, the expanding step is maintained between 24-72 hours. In some embodiments, the expanding step is maintained between 48-96 hours. In some embodiments, the expanding step is maintained between 72-120 hours. In some embodiments, the expanding step is maintained between 96-144 hours. In some embodiments, the expanding step is maintained between 120-168 hours. In some embodiments, the expanding step is maintained between 144-192 hours. In some embodiments, the expanding step is maintained between 168-216 hours. In some embodiments, the expanding step is maintained between 192-240 hours. In some embodiments, the expanding step is maintained between 216-264 hours. In some embodiments, the expanding step is maintained between 240-288 hours.
In some embodiments, the expanding step is maintained between 264-312 hours. In some embodiments, the expanding step is maintained between 288-336 hours. In some embodiments, the expanding step is maintained between 312-360 hours. In some embodiments, the expanding step is maintained between 336-384 hours. In some embodiments, the expanding step is maintained between 360-408 hours. In some embodiments, the expanding step is maintained between 384-432 hours. In some embodiments, the expanding step is maintained between 408-456 hours. In some embodiments, the expanding step is maintained for more than 456 hours.
The expansion step can be maintained for any length of time indicated in Table 1. The precursor CAR cell and/or population comprising a plurality of precursor CAR cells and/or a plurality of myeloid-derived CAR cells can be maintained in precursor media for any length of time indicated in Table 1.
Table 1. Optimal Timeline of Myeloid Differentiation from hematopoietic stem cells in Myel 1
Following expanding, the cells can be incubated in cell medium in a culture apparatus for a period of time or until the cells reach confluency or high cell density for optimal passage before passing the cells to another culture apparatus. The culturing apparatus can be any culture apparatus commonly used for culturing cells in vitro. In some embodiments, the level of confluence is 70% or greater before passing the cells to another culture apparatus. In some embodiments, the level of confluence is 90% or greater. A period of time can be any time suitable for the culture of cells in vitro. The culture medium may be replaced during the culture of the cells at any time. In some embodiments, the culture medium is replaced about every 2 to 3 days. The cells are then harvested from the culture apparatus whereupon the cells can be used immediately or stored for use at a later time.
The step of expanding the genetically modified precursor CAR cell in a precursor media can involve culturing the cell in a precursor media to increase the number of cells. Culturing of precursor CAR cells as disclosed herein can occur in multiple types of culture media. The culture media used for culturing cells is dependent upon the desired stage (e.g., expansion and/or differentiation). Examples of media that can be used for culturing the genetically modified precursor CAR cells include basal growth media, precursor media, or myeloid media, as disclosed herein, or any media that achieves similar and desired results of expansion and/or differentiation. As described herein, culture mediums can be defined by the constituents within the medium. For example, precursor mediums can be defined by the precursor cytokine mixtures contained within. Various constituents within the mediums can have a specific desired effect on the cells being cultured, such as promoting expansion or differentiation of a cell or plurality of cells. For example, the precursor media, as disclosed herein, comprising a precursor cytokine mixture is particularly effective at promoting the expansion and differentiation of stem cells toward the myeloid lineage. As such, genetically modified precursor CAR cells, which are capable of differentiation into myeloid-derived cells, can be expanded in a precursor media comprising a
precursor cytokine mixture to generate a population comprising a plurality of genetically modified precursor CAR cells, wherein a significant proportion of the plurality of genetically modified precursor CAR cells comprises a myeloid cell line surface marker.
The culturing step can be very short, for example less than 24 hours such as 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, or 23 hours. The culturing step can be longer, for example 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, or more days. In some embodiments, the cells may be cultured for several hours (about 3 hours) to about 14 days or any hourly integer value in between. Conditions appropriate for cell culture include an appropriate media (e.g., macrophage complete medium, DMEM/F12, DMEM/F 12-10 (Invitrogen)) that may contain factors necessary for proliferation and viability, including serum (e.g., fetal bovine or human serum), L-glutamine, insulin, M-CSF, GM-CSF, IL-10, IL-12, IL-15, TGF-beta, and TNFa, or any other additives for the growth of cells known to the skilled artisan. Other additives for the growth of cells include, but are not limited to, surfactant, plasmanate, and reducing agents such as N-acetyl-cysteine and 2-mercaptoethanol. In some embodiments, culture media can include Stemline II, RPMI 1640, AEVI-V, DMEM, MEM, a-MEM, F-12, X-Vivo 15, X-Vivo 20, and Optimizer, with added amino acids, sodium pyruvate, and vitamins, either serum-free or supplemented with an appropriate amount of serum (or plasma) or a defined set of hormones, and/or an amount of cytokine(s) sufficient for the growth, expansion and/or differentiation of the cells. Antibiotics, e.g., penicillin and streptomycin, are included only in experimental cultures, not in cultures of cells that are to be infused into a subject. The target cells are maintained under conditions necessary to support growth, for example, an appropriate temperature (e.g., 37°C) and atmosphere (e.g., air plus 5% CO2).
The medium used to culture the cells may include an agent that can activate the cells. For example, an agent that is known in the art to activate a macrophage, monocyte, dendritic cell, and/or a neutrophil is included in the medium.
Precursor Media
Disclosed herein is a precursor media comprising a precursor cytokine mixture for expanding genetically modified precursor CAR cell(s). The precursor media are particularly useful in their capacity to enable the expansion and/or differentiation of precursor cells, including hematopoietic stem cells, ex vivo. For instance, a hematopoietic stem cell can be expanded and differentiated into a myeloid-derived cell in the precursor medias disclosed herein ex vivo. The precursor media disclosed herein can promote the ex vivo differentiation of a stem cell into any myeloid-derived cell. The precursor medias disclosed herein are particularly effective for expanding a precursor CAR cell, and/or population comprising the same ex vivo. The methods are also particularly effective at expanding a population comprising the precursor CAR cell and/or a
I l l
plurality of said precursor CAR cells and/or a plurality of myeloid-derived CAR cells, wherein said myeloid-derived CAR cells are derived from said precursor cell. In addition to promoting expansion of a genetically modified precursor CAR cell, the precursor media comprising a precursor cytokine mixture are also particularly effective at promoting differentiation of a genetically modified precursor CAR cell. In some instances, the genetically modified precursor CAR cells are genetically modified stem cells expressing a CAR. In some instances, the stem cells expressing a CAR are hematopoietic stem cells (HSCs). The HSCs can be CD34+ cells. Expanding the HSCs modified to express a CAR generates a population comprising a plurality of genetically modified HSC CAR cells. For example, expanding the genetically modified HSC CAR cell increases the number of a genetically modified myeloid-derived CAR cell(s). The precursor media disclosed herein is particularly effective at promoting the expansion of HSCs. In some instances, the precursor media is particularly effective at expanding a CD34+ cell. In some instances, the precursor media is particularly effective at differentiating a CD34+ cell in a myeloid lineage cell. For example, the precursor media can be particularly effective at differentiating a CD34+ cell into a CD14+ cell, a CDl lb+ cell, a CD 13+ cell, a CD 15+ cell, a CDl lb+CD13+ cell, a
CD1 lb+CD13+CD14+ cell, a CD1 lb+CD13+CD15+ cell, a CDl lb+CD13+CD14+CD15+ cell, a CDl lb+CD14+ cell, a CDl lb+CD15+ cell, a CDl lb+CD14+CD15+ cell, a CD14+CD13+ cell, a CD14+CD15+ cell, a CD14+CD13+CD15+ cell, and/or a CD13+CD15+ cell.
. The precursor media disclosed herein can promote both expansion of CD34+ cells and the differentiation of CD34+ cells into myeloid lineage cells. In some instances, the precursor media is particularly effective at differentiation a CD34+ cell into a CD1 lb+ cell. In some instances, the precursor media is particularly effective at differentiation a CD34+ cell into a CD14+CD1 lb+ cell. Without wishing to be bound by theory, the cytokine mixture within the precursor media (i.e., the precursor cytokine mixture) is thought to contribute to the effective expansion and differentiation seen with the precursor media disclosed herein. Also disclosed herein are precursor cytokine mixtures that are particularly effective at promoting expansion of CD34+ cells (e.g., HSCs) and differentiation of CD34+ stem cells into myeloid-derived cells. In some instances, myeloid-derived cells are CD14+ cells. In some instances, myeloid-derived cells are CD1 lb+ cells. The precursor media comprising the precursor cytokine mixtures disclosed herein are particularly effective at promoting differentiation of HSCs into CD14+ cells. The precursor media comprising the precursor cytokine mixtures disclosed herein are particularly effective at promoting differentiation of HSCs into CDl lb+ cells.
Any cytokines at any concentrations can be used in the precursor media to support expansion and/or differentiation of the genetically modified precursor CAR cells. Specifically, the
precursor cytokine mixture within the precursor media can contribute to the combined expansion of CD34+ and differentiation of CD34+ cells into myeloid-derived cells. For example, the precursor cytokine mixture can comprise at least one cytokine selected from the following: stem cells factor (SCF), Flt3 ligand, thrombopoietin (TPO), Interleukin-3 (IL-3); Granulocyte colony-stimulating factor (G-CSF), Interleukin-6 (IL-6), Granulocyte-macrophage colony-stimulating factor (GM- CSF), and Macrophage colony-stimulating factor (MCSF).
Precursor Media Myel 2
In some instances, the precursor cytokine mixture comprises Flt3, SCF, TPO, M-CSF, and GM-CSF. In a preferred embodiment, the precursor media comprises Flt3 at a concentration of 100 ng/mL, SCF at a concentration of 100 ng/mL, TPO at a concentration of 100 ng/mL, M-CSF at a concentration of 150 ng/mL and GM-CSF at a concentration of 100 ng/mL.
In a preferred embodiment, the precursor media comprises Flt3 at a concentration of 100 ng/mL, SCF at a concentration of 100 ng/mL, TPO at a concentration of 100 ng/mL, M-CSF at a concentration of 150 ng/mL and GM-CSF at a concentration of 100 ng/mL, the precursor media further comprises GlutaMAX™ Supplement at a concentration of 2mM, and a basal growth media comprising VIVO™- 15 medium and Stemline® II Hematopoietic Stem Cell Expansion Media mixed at a ratio of 1 : 1.
In some instances, the precursor cytokine mixture comprises G-CSF at a concentration of between about 100 ng/mL and 150 ng/mL.
Precursor Media HSC 1
When the precursor cytokine mixture comprises G-CSF at a concentration of between about 100 ng/mL and 150 ng/mL, the precursor cytokine mixture comprises G-CSF, SCF, and TPO. In a preferred embodiment, the precursor media comprises G-CSF at a concentration of 100 ng/mL, SCF at a concentration of 100 ng/mL, and TPO at a concentration of 100 ng/mL. In a preferred embodiment, wherein the precursor media that comprises G-CSF at a concentration of 100 ng/mL, SCF at a concentration of 100 ng/mL, and TPO at a concentration of 100 ng/mL, the precursor media further comprises a basal growth media comprising VIVO™- 15 medium.
Precursor Media Myel 1
In some embodiments, the precursor media comprises granulocyte colony-stimulating factor (G-CSF). The granulocyte colony-stimulating factor (G-CSF) can be present in the precursor media at a concentration of at least 20 ng/mL, at least 30 ng/mL, at least 40 ng/mL, at least 50 ng/mL, at least 60 ng/mL, of at least 70 ng/mL, at least 80 ng/mL, at least 90 ng/mL, at least 100 ng/mL, at least 110 ng/mL, at least 120 ng/mL, at least 130 ng/mL, at least 140 ng/mL, at least 150 ng/mL, at least 160 ng/mL, at least 170 ng/mL, at least 180 ng/mL, at least 190 ng/mL, at least 200 ng/mL, at
least 210 ng/mL, of at least 220 ng/mL, at least 230 ng/mL, at least 240 ng/mL, at least 250 ng/mL, or more than 250 ng/mL. In some embodiments, the granulocyte colony-stimulating factor (G-CSF) can be present in the precursor media at a concentration of 20-40 ng/mL, 30-50 ng/mL, 40-60 ng/mL, 50-70 ng/mL, 60-80 ng/mL, 70-90 ng/mL, 80-100 ng/mL, 90-110 ng/mL, 100-120 ng/mL, 110-130 ng/mL, 120-140 ng/mL, 130-150 ng/mL, 140-160 ng/mL, 150-170 ng/mL, 160-180 ng/mL, 170-190 ng/mL, 180-200 ng/mL, 190-210 ng/mL, 200-220 ng/mL, 210-230 ng/mL, 220- 240 ng/mL, 230-250 ng/mL, or greater than 250 ng/mL.
In some embodiments, when the precursor media comprises G-CSF, said G-CSF is present at a concentration of between about 100 ng/mL and 200 ng/mL. In some embodiments, when the precursor media comprises G-CSF, said G-CSF is present at a concentration of between 100-105 ng/mL, 105-110 ng/mL, 110-115 ng/mL, 115-120 ng/mL, 120-125 ng/mL, 125-130 ng/mL, 130- 135 ng/mL, 135-140 ng/mL, 140-145 ng/mL, 145-150 ng/mL, 150-155 ng/mL, 155-160 ng/mL, 160-165 ng/mL, 165-170 ng/mL, 170-175 ng/mL, 175-180 ng/mL, 180-185 ng/mL, 185-190 ng/mL, 190-195 ng/mL, or 195-200 ng/mL. In some embodiments, when the precursor media comprises G-CSF, said G-CSF is present at a concentration of between about 100 ng/mL and 150 ng/mL.
In some embodiments, the precursor media comprises more than one cytokine (i.e., a precursor cytokine mixture). When the precursor cytokine mixture comprises G-CSF at a concentration of between about 100 ng/mL and 150 ng/mL in said precursor media, the precursor cytokine mixture comprises G-CSF, GM-CSF, SCF, and TPO. When the precursor media comprises G-CSF at a concentration of between 100 ng/mL and 150 ng/mL, said precursor media can comprise any of the following cytokines: GM-CSF, SCF, and TPO. When the precursor media comprises G-CSF at a concentration of between 100 ng/mL and 150 ng/mL, said precursor media can comprise at lease one of the following cytokines: GM-CSF, SCF, and TPO. When the precursor media comprises G-CSF at a concentration of between 100 ng/mL and 150 ng/mL, said precursor media can comprise all of the following cytokines: GM-CSF, SCF, and TPO. In such instances, GM-CSF can be present at a concentration between about 75 ng/mL and 125 ng/mL; SCF can be present at a concentration between about 75 ng/mL and 125 ng/mL; and/or TPO can be present at a concentration between about 75 ng/mL and 125 ng/mL.
In a preferred embodiment, the precursor media comprises G-CSF at a concentration of 150 ng/mL, GM-CSF at a concentration of 100 ng/mL, SCF at a concentration of 100 ng/mL, and TPO at a concentration of 100 ng/mL.
In some embodiments, when the precursor media comprises granulocyte colony-stimulating factor (G-CSF) at a concentration of between about 100 ng/mL and 200 ng/mL, said precursor media
further comprises: a. granulocyte-macrophage colony-stimulating factor (GM-CSF) at a concentration of 10-30 ng/mL, 20-40 ng/mL, 30-50 ng/mL, 40-60 ng/mL, 50-70 ng/mL, 60-80 ng/mL, 70-90 ng/mL, 80-100 ng/mL, 90-110 ng/mL, 100-120 ng/mL, 110-130 ng/mL, 120-140 ng/mL, 130-150 ng/mL, 140-160 ng/mL, 150-170 ng/mL, 160-180 ng/mL, 170-190 ng/mL, 180- 200 ng/mL, 190-210 ng/mL, 200-220 ng/mL, or greater than 220 ng/mL; b. stem cells factor (SCF) at a concentration of 10-30 ng/mL, 20-40 ng/mL, 30-50 ng/mL, 40-60 ng/mL, 50-70 ng/mL, 60-80 ng/mL, 70-90 ng/mL, 80-100 ng/mL, 90-110 ng/mL, 100-120 ng/mL, 110-130 ng/mL, 120-140 ng/mL, 130-150 ng/mL, 140-160 ng/mL, 150-170 ng/mL, 160-180 ng/mL, 170-190 ng/mL, 180- 200 ng/mL, 190-210 ng/mL, 200-220 ng/mL, or greater than 220 ng/mL; and/or c. thrombopoietin (TPO) at a concentration of 10-30 ng/mL, 20-40 ng/mL, 30-50 ng/mL, 40-60 ng/mL, 50-70 ng/mL, 60-80 ng/mL, 70-90 ng/mL, 80-100 ng/mL, 90-110 ng/mL, 100-120 ng/mL, 110-130 ng/mL, 120- 140 ng/mL, 130-150 ng/mL, 140-160 ng/mL, 150-170 ng/mL, 160-180 ng/mL, 170-190 ng/mL, 180-200 ng/mL, 190-210 ng/mL, 200-220 ng/mL, or greater than 220 ng/mL.
In some embodiments, when the precursor media comprises granulocyte colony-stimulating factor (G-CSF) at a concentration of about 140 ng/mL. In some embodiments, when the precursor media comprises granulocyte colony-stimulating factor (G-CSF) at a concentration of about 140 ng/mL, said precursor media further comprises: a. granulocyte-macrophage colony-stimulating factor (GM-CSF) at a concentration of 10-30 ng/mL, 20-40 ng/mL, 30-50 ng/mL, 40-60 ng/mL, 50- 70 ng/mL, 60-80 ng/mL, 70-90 ng/mL, 80-100 ng/mL, 90-110 ng/mL, 100-120 ng/mL, 110-130 ng/mL, 120-140 ng/mL, 130-150 ng/mL, 140-160 ng/mL, 150-170 ng/mL, 160-180 ng/mL, 170- 190 ng/mL, 180-200 ng/mL, 190-210 ng/mL, 200-220 ng/mL, or greater than 220 ng/mL; b. stem cells factor (SCF) at a concentration of 10-30 ng/mL, 20-40 ng/mL, 30-50 ng/mL, 40-60 ng/mL, 50-70 ng/mL, 60-80 ng/mL, 70-90 ng/mL, 80-100 ng/mL, 90-110 ng/mL, 100-120 ng/mL, 110-130 ng/mL, 120-140 ng/mL, 130-150 ng/mL, 140-160 ng/mL, 150-170 ng/mL, 160-180 ng/mL, 170- 190 ng/mL, 180-200 ng/mL, 190-210 ng/mL, 200-220 ng/mL, or greater than 220 ng/mL; and/or c. thrombopoietin (TPO) at a concentration of 10-30 ng/mL, 20-40 ng/mL, 30-50 ng/mL, 40-60 ng/mL, 50-70 ng/mL, 60-80 ng/mL, 70-90 ng/mL, 80-100 ng/mL, 90-110 ng/mL, 100-120 ng/mL, 110-130 ng/mL, 120-140 ng/mL, 130-150 ng/mL, 140-160 ng/mL, 150-170 ng/mL, 160-180 ng/mL, 170-190 ng/mL, 180-200 ng/mL, 190-210 ng/mL, 200-220 ng/mL, or greater than 220 ng/mL.
In some embodiments, when the precursor media comprises granulocyte colony-stimulating factor (G-CSF) at a concentration of about 145 ng/mL. In some embodiments, when the precursor media comprises granulocyte colony-stimulating factor (G-CSF) at a concentration of about 145 ng/mL, said precursor media further comprises: a. granulocyte-macrophage colony-stimulating
factor (GM-CSF) at a concentration of 10-30 ng/mL, 20-40 ng/mL, 30-50 ng/mL, 40-60 ng/mL, 50- 70 ng/mL, 60-80 ng/mL, 70-90 ng/mL, 80-100 ng/mL, 90-110 ng/mL, 100-120 ng/mL, 110-130 ng/mL, 120-140 ng/mL, 130-150 ng/mL, 140-160 ng/mL, 150-170 ng/mL, 160-180 ng/mL, 170- 190 ng/mL, 180-200 ng/mL, 190-210 ng/mL, 200-220 ng/mL, or greater than 220 ng/mL; b. stem cells factor (SCF) at a concentration of 10-30 ng/mL, 20-40 ng/mL, 30-50 ng/mL, 40-60 ng/mL, 50-70 ng/mL, 60-80 ng/mL, 70-90 ng/mL, 80-100 ng/mL, 90-110 ng/mL, 100-120 ng/mL, 110-130 ng/mL, 120-140 ng/mL, 130-150 ng/mL, 140-160 ng/mL, 150-170 ng/mL, 160-180 ng/mL, 170- 190 ng/mL, 180-200 ng/mL, 190-210 ng/mL, 200-220 ng/mL, or greater than 220 ng/mL; and/or c. thrombopoietin (TPO) at a concentration of 10-30 ng/mL, 20-40 ng/mL, 30-50 ng/mL, 40-60 ng/mL, 50-70 ng/mL, 60-80 ng/mL, 70-90 ng/mL, 80-100 ng/mL, 90-110 ng/mL, 100-120 ng/mL, 110-130 ng/mL, 120-140 ng/mL, 130-150 ng/mL, 140-160 ng/mL, 150-170 ng/mL, 160-180 ng/mL, 170-190 ng/mL, 180-200 ng/mL, 190-210 ng/mL, 200-220 ng/mL, or greater than 220 ng/mL.
In some embodiments, when the precursor media comprises granulocyte colony-stimulating factor (G-CSF) at a concentration of about 150 ng/mL. In some embodiments, when the precursor media comprises granulocyte colony-stimulating factor (G-CSF) at a concentration of about 150 ng/mL, said precursor media further comprises: a. granulocyte-macrophage colony-stimulating factor (GM-CSF) at a concentration of 10-30 ng/mL, 20-40 ng/mL, 30-50 ng/mL, 40-60 ng/mL, 50- 70 ng/mL, 60-80 ng/mL, 70-90 ng/mL, 80-100 ng/mL, 90-110 ng/mL, 100-120 ng/mL, 110-130 ng/mL, 120-140 ng/mL, 130-150 ng/mL, 140-160 ng/mL, 150-170 ng/mL, 160-180 ng/mL, 170- 190 ng/mL, 180-200 ng/mL, 190-210 ng/mL, 200-220 ng/mL, or greater than 220 ng/mL; b. stem cells factor (SCF) at a concentration of 10-30 ng/mL, 20-40 ng/mL, 30-50 ng/mL, 40-60 ng/mL, 50-70 ng/mL, 60-80 ng/mL, 70-90 ng/mL, 80-100 ng/mL, 90-110 ng/mL, 100-120 ng/mL, 110-130 ng/mL, 120-140 ng/mL, 130-150 ng/mL, 140-160 ng/mL, 150-170 ng/mL, 160-180 ng/mL, 170- 190 ng/mL, 180-200 ng/mL, 190-210 ng/mL, 200-220 ng/mL, or greater than 220 ng/mL; and/or c. thrombopoietin (TPO) at a concentration of 10-30 ng/mL, 20-40 ng/mL, 30-50 ng/mL, 40-60 ng/mL, 50-70 ng/mL, 60-80 ng/mL, 70-90 ng/mL, 80-100 ng/mL, 90-110 ng/mL, 100-120 ng/mL, 110-130 ng/mL, 120-140 ng/mL, 130-150 ng/mL, 140-160 ng/mL, 150-170 ng/mL, 160-180 ng/mL, 170-190 ng/mL, 180-200 ng/mL, 190-210 ng/mL, 200-220 ng/mL, or greater than 220 ng/mL.
In some embodiments, when the precursor media comprises granulocyte colony-stimulating factor (G-CSF) at a concentration of about 155 ng/mL. In some embodiments, when the precursor media comprises granulocyte colony-stimulating factor (G-CSF) at a concentration of about 155 ng/mL, said precursor media further comprises: a. granulocyte-macrophage colony-stimulating
factor (GM-CSF) at a concentration of 10-30 ng/mL, 20-40 ng/mL, 30-50 ng/mL, 40-60 ng/mL, 50- 70 ng/mL, 60-80 ng/mL, 70-90 ng/mL, 80-100 ng/mL, 90-110 ng/mL, 100-120 ng/mL, 110-130 ng/mL, 120-140 ng/mL, 130-150 ng/mL, 140-160 ng/mL, 150-170 ng/mL, 160-180 ng/mL, 170- 190 ng/mL, 180-200 ng/mL, 190-210 ng/mL, 200-220 ng/mL, or greater than 220 ng/mL; b. stem cells factor (SCF) at a concentration of 10-30 ng/mL, 20-40 ng/mL, 30-50 ng/mL, 40-60 ng/mL, 50-70 ng/mL, 60-80 ng/mL, 70-90 ng/mL, 80-100 ng/mL, 90-110 ng/mL, 100-120 ng/mL, 110-130 ng/mL, 120-140 ng/mL, 130-150 ng/mL, 140-160 ng/mL, 150-170 ng/mL, 160-180 ng/mL, 170- 190 ng/mL, 180-200 ng/mL, 190-210 ng/mL, 200-220 ng/mL, or greater than 220 ng/mL; and/or c. thrombopoietin (TPO) at a concentration of 10-30 ng/mL, 20-40 ng/mL, 30-50 ng/mL, 40-60 ng/mL, 50-70 ng/mL, 60-80 ng/mL, 70-90 ng/mL, 80-100 ng/mL, 90-110 ng/mL, 100-120 ng/mL, 110-130 ng/mL, 120-140 ng/mL, 130-150 ng/mL, 140-160 ng/mL, 150-170 ng/mL, 160-180 ng/mL, 170-190 ng/mL, 180-200 ng/mL, 190-210 ng/mL, 200-220 ng/mL, or greater than 220 ng/mL.
In some embodiments, when the precursor media comprises granulocyte colony-stimulating factor (G-CSF) at a concentration of about 160 ng/mL. In some embodiments, when the precursor media comprises granulocyte colony-stimulating factor (G-CSF) at a concentration of about 160 ng/mL, said precursor media further comprises: a. granulocyte-macrophage colony-stimulating factor (GM-CSF) at a concentration of 10-30 ng/mL, 20-40 ng/mL, 30-50 ng/mL, 40-60 ng/mL, 50- 70 ng/mL, 60-80 ng/mL, 70-90 ng/mL, 80-100 ng/mL, 90-110 ng/mL, 100-120 ng/mL, 110-130 ng/mL, 120-140 ng/mL, 130-150 ng/mL, 140-160 ng/mL, 150-170 ng/mL, 160-180 ng/mL, 170- 190 ng/mL, 180-200 ng/mL, 190-210 ng/mL, 200-220 ng/mL, or greater than 220 ng/mL; b. stem cells factor (SCF) at a concentration of 10-30 ng/mL, 20-40 ng/mL, 30-50 ng/mL, 40-60 ng/mL, 50-70 ng/mL, 60-80 ng/mL, 70-90 ng/mL, 80-100 ng/mL, 90-110 ng/mL, 100-120 ng/mL, 110-130 ng/mL, 120-140 ng/mL, 130-150 ng/mL, 140-160 ng/mL, 150-170 ng/mL, 160-180 ng/mL, 170- 190 ng/mL, 180-200 ng/mL, 190-210 ng/mL, 200-220 ng/mL, or greater than 220 ng/mL; and/or c. thrombopoietin (TPO) at a concentration of 10-30 ng/mL, 20-40 ng/mL, 30-50 ng/mL, 40-60 ng/mL, 50-70 ng/mL, 60-80 ng/mL, 70-90 ng/mL, 80-100 ng/mL, 90-110 ng/mL, 100-120 ng/mL, 110-130 ng/mL, 120-140 ng/mL, 130-150 ng/mL, 140-160 ng/mL, 150-170 ng/mL, 160-180 ng/mL, 170-190 ng/mL, 180-200 ng/mL, 190-210 ng/mL, 200-220 ng/mL, or greater than 220 ng/mL.
In a preferred embodiment, the precursor media comprises G-CSF at a concentration of 150 ng/mL. When the precursor media comprises G-CSF at a concentration of about 150ng/mL, said precursor media further comprises: a. granulocyte-macrophage colony-stimulating factor (GM- CSF) at a concentration between about 75 ng/mL and 125 ng/mL; b. stem cells factor (SCF) at a
concentration between about 75 ng/mL and 125 ng/mL; and/or c. thrombopoietin (TPO) at a concentration between about 75 ng/mL and 125 ng/mL.
In another preferred embodiment, the precursor media comprises G-CSF at a concentration of about 150ng/mL, and said precursor media further comprises: a. granulocyte-macrophage colony-stimulating factor (GM-CSF) at a concentration between about 70 ng/mL, about 75 ng/mL, about 80 ng/mL, about 85 ng/mL, about 90 ng/mL, about 95 ng/mL, about 100 ng/mL, about 105 ng/mL, about 110 ng/mL, about 115 ng/mL, about 120 ng/mL, or about 125 ng/mL; b. stem cells factor (SCF) at a concentration between about 70 ng/mL, about 75 ng/mL, about 80 ng/mL, about 85 ng/mL, about 90 ng/mL, about 95 ng/mL, about 100 ng/mL, about 105 ng/mL, about 110 ng/mL, about 115 ng/mL, about 120 ng/mL, or about 125 ng/mL; and/or c. thrombopoietin (TPO) at a concentration between about 70 ng/mL, about 75 ng/mL, about 80 ng/mL, about 85 ng/mL, about 90 ng/mL, about 95 ng/mL, about 100 ng/mL, about 105 ng/mL, about 110 ng/mL, about 115 ng/mL, about 120 ng/mL, or about 125 ng/mL.
In any of the above embodiments, the precursor media can further comprise GlutaMAX™ Supplement, a basal growth media, and/or a basal growth media comprising VIVO™- 15 medium and Stemline® II Hematopoietic Stem Cell Expansion Media.
In a preferred embodiment, wherein the precursor media comprises G-CSF at a concentration of 150 ng/mL, GM-CSF at a concentration of 100 ng/mL, SCF at a concentration of 100 ng/mL, and TPO at a concentration of 100 ng/mL, the precursor media further comprises GlutaMAX™ Supplement at a concentration of 2mM, and a basal growth media comprising VIVO™-15 medium and Stemline® II Hematopoietic Stem Cell Expansion Media mixed at a ratio of 1 : 1.
In another preferred embodiment, the precursor media comprises G-CSF at a concentration of about 150ng/mL, and said precursor media further comprises: a. granulocyte-macrophage colony-stimulating factor (GM-CSF) at a concentration between about 70 ng/mL, about 75 ng/mL, about 80 ng/mL, about 85 ng/mL, about 90 ng/mL, about 95 ng/mL, about 100 ng/mL, about 105 ng/mL, about 110 ng/mL, about 115 ng/mL, about 120 ng/mL, or about 125 ng/mL; b. stem cells factor (SCF) at a concentration between about 70 ng/mL, about 75 ng/mL, about 80 ng/mL, about 85 ng/mL, about 90 ng/mL, about 95 ng/mL, about 100 ng/mL, about 105 ng/mL, about 110 ng/mL, about 115 ng/mL, about 120 ng/mL, or about 125 ng/mL; c. thrombopoietin (TPO) at a concentration between about 70 ng/mL, about 75 ng/mL, about 80 ng/mL, about 85 ng/mL, about 90 ng/mL, about 95 ng/mL, about 100 ng/mL, about 105 ng/mL, about 110 ng/mL, about 115 ng/mL, about 120 ng/mL, or about 125 ng/mL; and/or d. GlutaMAX™ Supplement at a concentration of between ImM and 3mM.
Gibco GlutaMAX Supplement™ is an alternative to L-glutamine, with increased stability that improves cell health. GlutaMAX Supplement™ is suitable for both adherent and suspension culture of mammalian cells, with no adaptation required. GlutaMAX Supplement™ is offered as a 200 mM L-alanyl-L-glutamine dipeptide in 0.85% NaCl. It would be understood by a skilled artisan that any formulation of L-alanyl-L-glutamine dipeptide (e.g., a generic brand or generic formulation) matching that of GlutaMAX Supplement™, which functions in about the same manner as GlutaMAX Supplement™ can be used in the precursor media. A generic formulation will function in about the same manner if it promotes expansion and/or differentiation of stem cells within precursor media as GlutaMAX Supplement™. GlutaMAX Supplement™ is also included in a variety of media formulations. Any media formulation can be used, so long as it is present within the indicated concentration in the precursor media. As such, any formulation of L-alanyl-L- glutamine dipeptide present in the precursor media at any concentration disclosed herein for GlutaMAX Supplement ™ (e.g., between ImM and 3mM, preferably about 2mM), can be used in the precursor media disclosed herein. The L-alanyl-L-glutamine dipeptide can be in a NaCl solution, of about 0.85% NaCl (e.g, about 0.80% to about 0.90%). The L-alanyl-L-glutamine dipeptide can be in a NaCl solution at any concentration of NaCl that enables the L-alanyl-L- glutamine dipeptide to function in about the same manner as GlutaMAX Supplment™.
In another preferred embodiment, the precursor media comprises G-CSF at a concentration of about 150ng/mL, and said precursor media further comprises: a. granulocyte-macrophage colony-stimulating factor (GM-CSF) at a concentration between about 70 ng/mL, about 75 ng/mL, about 80 ng/mL, about 85 ng/mL, about 90 ng/mL, about 95 ng/mL, about 100 ng/mL, about 105 ng/mL, about 110 ng/mL, about 115 ng/mL, about 120 ng/mL, or about 125 ng/mL; b. stem cells factor (SCF) at a concentration between about 70 ng/mL, about 75 ng/mL, about 80 ng/mL, about 85 ng/mL, about 90 ng/mL, about 95 ng/mL, about 100 ng/mL, about 105 ng/mL, about 110 ng/mL, about 115 ng/mL, about 120 ng/mL, or about 125 ng/mL; c. thrombopoietin (TPO) at a concentration between about 70 ng/mL, about 75 ng/mL, about 80 ng/mL, about 85 ng/mL, about 90 ng/mL, about 95 ng/mL, about 100 ng/mL, about 105 ng/mL, about 110 ng/mL, about 115 ng/mL, about 120 ng/mL, or about 125 ng/mL; and/or d. GlutaMAX™ Supplement at a concentration of about 2 mM.
In another preferred embodiment, the precursor media comprises G-CSF at a concentration of about 150ng/mL, and said precursor media further comprises: a. granulocyte-macrophage colony-stimulating factor (GM-CSF) at a concentration between about 70 ng/mL, about 75 ng/mL, about 80 ng/mL, about 85 ng/mL, about 90 ng/mL, about 95 ng/mL, about 100 ng/mL, about 105 ng/mL, about 110 ng/mL, about 115 ng/mL, about 120 ng/mL, or about 125 ng/mL; b. stem cells
factor (SCF) at a concentration between about 70 ng/mL, about 75 ng/mL, about 80 ng/mL, about 85 ng/mL, about 90 ng/mL, about 95 ng/mL, about 100 ng/mL, about 105 ng/mL, about 110 ng/mL, about 115 ng/mL, about 120 ng/mL, or about 125 ng/mL; c. thrombopoietin (TPO) at a concentration between about 70 ng/mL, about 75 ng/mL, about 80 ng/mL, about 85 ng/mL, about 90 ng/mL, about 95 ng/mL, about 100 ng/mL, about 105 ng/mL, about 110 ng/mL, about 115 ng/mL, about 120 ng/mL, or about 125 ng/mL; and/or d. GlutaMAX™ Supplement at a concentration of 2 mM.
In another preferred embodiment, the precursor media comprises G-CSF at a concentration of about 150ng/mL, and said precursor media further comprises: a. granulocyte-macrophage colony-stimulating factor (GM-CSF) at a concentration between about 70 ng/mL, about 75 ng/mL, about 80 ng/mL, about 85 ng/mL, about 90 ng/mL, about 95 ng/mL, about 100 ng/mL, about 105 ng/mL, about 110 ng/mL, about 115 ng/mL, about 120 ng/mL, or about 125 ng/mL; b. stem cells factor (SCF) at a concentration between about 70 ng/mL, about 75 ng/mL, about 80 ng/mL, about 85 ng/mL, about 90 ng/mL, about 95 ng/mL, about 100 ng/mL, about 105 ng/mL, about 110 ng/mL, about 115 ng/mL, about 120 ng/mL, or about 125 ng/mL; c. thrombopoietin (TPO) at a concentration between about 70 ng/mL, about 75 ng/mL, about 80 ng/mL, about 85 ng/mL, about 90 ng/mL, about 95 ng/mL, about 100 ng/mL, about 105 ng/mL, about 110 ng/mL, about 115 ng/mL, about 120 ng/mL, or about 125 ng/mL; and/or d. L-alanyl-L-glutamine dipeptide at a concentration of about 2 mM.
In another preferred embodiment, the precursor media comprises G-CSF at a concentration of about 150ng/mL, and said precursor media further comprises: a. granulocyte-macrophage colony-stimulating factor (GM-CSF) at a concentration between about 70 ng/mL, about 75 ng/mL, about 80 ng/mL, about 85 ng/mL, about 90 ng/mL, about 95 ng/mL, about 100 ng/mL, about 105 ng/mL, about 110 ng/mL, about 115 ng/mL, about 120 ng/mL, or about 125 ng/mL; b. stem cells factor (SCF) at a concentration between about 70 ng/mL, about 75 ng/mL, about 80 ng/mL, about 85 ng/mL, about 90 ng/mL, about 95 ng/mL, about 100 ng/mL, about 105 ng/mL, about 110 ng/mL, about 115 ng/mL, about 120 ng/mL, or about 125 ng/mL; c. thrombopoietin (TPO) at a concentration between about 70 ng/mL, about 75 ng/mL, about 80 ng/mL, about 85 ng/mL, about 90 ng/mL, about 95 ng/mL, about 100 ng/mL, about 105 ng/mL, about 110 ng/mL, about 115 ng/mL, about 120 ng/mL, or about 125 ng/mL; d. L-alanyl-L-glutamine dipeptide at a concentration of about 2 mM; and/or e. a basal growth media.
In another preferred embodiment, the precursor media comprises G-CSF at a concentration of about 150ng/mL, and said precursor media further comprises: a. granulocyte-macrophage colony-stimulating factor (GM-CSF) at a concentration between about 70 ng/mL, about 75 ng/mL,
about 80 ng/mL, about 85 ng/mL, about 90 ng/mL, about 95 ng/mL, about 100 ng/mL, about 105 ng/mL, about 110 ng/mL, about 115 ng/mL, about 120 ng/mL, or about 125 ng/mL; b. stem cells factor (SCF) at a concentration between about 70 ng/mL, about 75 ng/mL, about 80 ng/mL, about 85 ng/mL, about 90 ng/mL, about 95 ng/mL, about 100 ng/mL, about 105 ng/mL, about 110 ng/mL, about 115 ng/mL, about 120 ng/mL, or about 125 ng/mL; c. thrombopoietin (TPO) at a concentration between about 70 ng/mL, about 75 ng/mL, about 80 ng/mL, about 85 ng/mL, about 90 ng/mL, about 95 ng/mL, about 100 ng/mL, about 105 ng/mL, about 110 ng/mL, about 115 ng/mL, about 120 ng/mL, or about 125 ng/mL; d. GlutaMAX™ Supplement; and/or e. a basal growth media, wherein said basal growth media comprises: i. VIV0™-15 medium and/or ii. Stemline® II Hematopoietic Stem Cell Expansion Media.
In another preferred embodiment, the precursor media comprises G-CSF at a concentration of about 150ng/mL, and said precursor media further comprises: a. granulocyte-macrophage colony-stimulating factor (GM-CSF) at a concentration between about 70 ng/mL, about 75 ng/mL, about 80 ng/mL, about 85 ng/mL, about 90 ng/mL, about 95 ng/mL, about 100 ng/mL, about 105 ng/mL, about 110 ng/mL, about 115 ng/mL, about 120 ng/mL, or about 125 ng/mL; b. stem cells factor (SCF) at a concentration between about 70 ng/mL, about 75 ng/mL, about 80 ng/mL, about 85 ng/mL, about 90 ng/mL, about 95 ng/mL, about 100 ng/mL, about 105 ng/mL, about 110 ng/mL, about 115 ng/mL, about 120 ng/mL, or about 125 ng/mL; c. thrombopoietin (TPO) at a concentration between about 70 ng/mL, about 75 ng/mL, about 80 ng/mL, about 85 ng/mL, about 90 ng/mL, about 95 ng/mL, about 100 ng/mL, about 105 ng/mL, about 110 ng/mL, about 115 ng/mL, about 120 ng/mL, or about 125 ng/mL; d. GlutaMAX™ Supplement at a concentration of about 2 mM; and/or e. a basal growth media, wherein said basal growth media comprises: i. VIV0™-15 medium and ii. Stemline® II Hematopoietic Stem Cell Expansion Media, wherein said VIV0™-15 medium and Stemline® II Hematopoietic Stem Cell Expansion Media are present at a 1 : 1 ratio.
Any basal growth media known in the art is envisaged as being used in the precursor media disclosed herein.
Precursor Media My el 1.1
When the precursor cytokine mixture comprises G-CSF at a concentration of between about 100 ng/mL and 150 ng/mL, the precursor cytokine mixture comprises G-CSF, M-CSF, SCF, and TPO. In a preferred embodiment, the precursor media comprises G-CSF at a concentration of 150 ng/mL, M-CSF at a concentration of 150 ng/mL, SCF at a concentration of 100 ng/mL, and TPO at a concentration of 100 ng/mL.
In a preferred embodiment, wherein the precursor media comprises G-CSF at a concentration of 150 ng/mL, M-CSF at a concentration of 150 ng/mL, SCF at a concentration of 100 ng/mL, and TPO at a concentration of 100 ng/mL, the precursor media further comprises GlutaMAX™ Supplement at a concentration of 2mM, and a basal growth media comprising VIVO™-15 medium and Stemline® II Hematopoietic Stem Cell Expansion Media mixed at a ratio of 1 : 1.
Precursor Media Myel 3
When the precursor cytokine mixture comprises G-CSF at a concentration of between about 100 ng/mL and 150 ng/mL, the precursor cytokine mixture comprises G-CSF, M-CSF, SCF, GM- CSF, and TPO. In a preferred embodiment, the precursor media comprises G-CSF at a concentration of 150 ng/mL, M-CSF at a concentration of 150 ng/mL, SCF at a concentration of 100 ng/mL, GM-CSF at a concentration of 100 ng/mL and TPO at a concentration of 100 ng/mL.
In a preferred embodiment, wherein the precursor media comprises G-CSF at a concentration of 150 ng/mL, M-CSF at a concentration of 150 ng/mL, SCF at a concentration of 100 ng/mL, and TPO at a concentration of 100 ng/mL, the precursor media further comprises GlutaMAX™ Supplement at a concentration of 2mM, and a basal growth media comprising VIVO™-15 medium and Stemline® II Hematopoietic Stem Cell Expansion Media mixed at a ratio of 1 : 1.
Precursor Media Myel 4
When the precursor cytokine mixture comprises G-CSF at a concentration of between about 100 ng/mL and 150 ng/mL, the precursor cytokine mixture comprises G-CSF, M-CSF, IL-6, IL-3, SCF, and TPO. In a preferred embodiment, the precursor media comprises G-CSF at a concentration of 150 ng/mL, M-CSF at a concentration of 150 ng/mL, IL-3 at a concentration of 50 ng/mL, IL-6 at a concentration of 100 ng/mL, SCF at a concentration of 100 ng/mL, and TPO at a concentration of 100 ng/mL.
In a preferred embodiment, wherein the precursor media comprises G-CSF at a concentration of 150 ng/mL, M-CSF at a concentration of 150 ng/mL, IL-3 at a concentration of 50 ng/mL, IL-6 at a concentration of 100 ng/mL, SCF at a concentration of 100 ng/mL, and TPO at a concentration of 100 ng/mL, the precursor media further comprises GlutaMAX™ Supplement at a concentration of 2 mM, and a basal growth media comprising VIVO™- 15 medium and Stemline® II Hematopoietic Stem Cell Expansion Media mixed at a ratio of 1 : 1.
Precursor Media Myel 4.1
When the precursor cytokine mixture comprises G-CSF at a concentration of between about 100 ng/mL and 150 ng/mL, the precursor cytokine mixture comprises G-CSF, M-CSF, IL-3, IL-6,
SCF, GM-SCF, and TPO. In a preferred embodiment, the precursor media comprises G-CSF at a concentration of 150 ng/mL, M-CSF at a concentration of 150 ng/mL, IL-3 at a concentration of 50 ng/mL, IL-6 at a concentration of 100 ng/mL, SCF at a concentration of 100 ng/mL, GM-SCF at a concentration of 100 ng/mL, and TPO at a concentration of 100 ng/mL.
In a preferred embodiment, wherein the precursor media comprises G-CSF at a concentration of 150 ng/mL, M-CSF at a concentration of 150 ng/mL, IL-3 at a concentration of 50 ng/mL, IL-6 at a concentration of 100 ng/mL, SCF at a concentration of 100 ng/mL, GM-SCF at a concentration of 100 ng/mL, and TPO at a concentration of 100 ng/mL, the precursor media further comprises GlutaMAX™ Supplement at a concentration of 2mM, and a basal growth media comprising VIVO™- 15 medium and Stemline® II Hematopoietic Stem Cell Expansion Media mixed at a ratio of 1 : 1.
When the precursor cytokine mixture comprises stem cells factor (SCF), the concentration of the SCF in the precursor media can be at least 50 ng/mL, at least 100 ng/mL, at least 150 ng/mL, or at least 200 ng/mL. In a preferred embodiment, the precursor media comprises SCF at a concentration of 100 ng/mL.
When the precursor cytokine mixture comprises Flt3 ligand, the concentration of the Flt3 ligand in the precursor media can be at least 50 ng/mL, at least 100 ng/mL, at least 150 ng/mL, or at least 200 ng/mL.
When the precursor cytokine mixture comprises thrombopoietin (TPO), the concentration of the TPO in the precursor media can be at least 50 ng/mL, at least 100 ng/mL, at least 150 ng/mL, or at least 200 ng/mL. In a preferred embodiment, the precursor media comprises TPO at a concentration of 100 ng/mL.
When the precursor cytokine mixture comprises Interleukin-3 (IL-3), the concentration of the IL-3 in the precursor media can be at least 50 ng/mL, at least 100 ng/mL, at least 150 ng/mL, or at least 200 ng/mL. In a preferred embodiment, the precursor media comprises IL-3 at a concentration of 50 ng/mL.
When the precursor cytokine mixture comprises Granulocyte colony-stimulating factor (G- CSF), the concentration of the G-CSF in the precursor media can be at least 50 ng/mL, at least 100 ng/mL, at least 150 ng/mL, or at least 200 ng/mL. In a preferred embodiment, the precursor media comprises G-CSF at a concentration of 150 ng/mL.
When the precursor cytokine mixture comprises Interleukin-6 (IL-6), the concentration of the IL-6 in the precursor media can be at least 50 ng/mL, at least 100 ng/mL, at least 150 ng/mL, or at least 200 ng/mL. In a preferred embodiment, the precursor media comprises IL-6 at a concentration of 100 ng/mL.
When the precursor cytokine mixture comprises Granulocyte-macrophage colonystimulating factor (GM-CSF), the concentration of the GM-CSF in the precursor media can be at least 50 ng/mL, at least 100 ng/mL, at least 150 ng/mL, or at least 200 ng/mL. In a preferred embodiment, the precursor media comprises GM-CSF at a concentration of 100 ng/mL.
When the precursor cytokine mixture comprises macrophage colony-stimulating factor (MCSF), the concentration of the MCSF in the precursor media can be at least 50 ng/mL, at least 100 ng/mL, at least 150 ng/mL, or at least 200 ng/mL. In a preferred embodiment, the precursor media comprises macrophage colony-stimulating factor (MCSF) at a concentration of 150 ng/mL.
In some instances, the precursor media comprises stem cells factor (SCF), Flt3 ligand, thrombopoietin (TPO), Granulocyte colony-stimulating factor (G-CSF), and Granulocytemacrophage colony-stimulating factor (GM-CSF). The stem cells factor (SCF), Flt3 ligand, thrombopoietin (TPO), Granulocyte colony-stimulating factor (G-CSF), and Granulocytemacrophage colony-stimulating factor (GM-CSF) can be present within the precursor media at any useful concentration. A useful concentration can be defined as a concentration of a specific cytokine that contributes to a desired effect on expansion and/or differentiation in a precursor media. For example, a cytokine at a specific concentration that is associated with an increased expansion rate of genetically modified CAR HSCs.
In some instances, the myeloid media comprises stem cells factor (SCF), Flt3 ligand, thrombopoietin (TPO), Granulocyte colony-stimulating factor (G-CSF), and Granulocytemacrophage colony-stimulating factor (GM-CSF). The stem cells factor (SCF), Flt3 ligand, thrombopoietin (TPO), Granulocyte colony-stimulating factor (G-CSF), and Granulocytemacrophage colony-stimulating factor (GM-CSF) can be present within the myeloid media at any useful concentration. A useful concentration can be defined as a concentration of a specific cytokine that contributes to a desired effect on expansion and/or differentiation in a precursor media. For example, a cytokine at a specific concentration that is associated with an increased expansion rate of genetically modified CAR HSCs.
Flt3 ligand can be within the precursor mixture at a concentration of at most 50 ng/mL, at most 40 ng/mL, at most 30 ng/mL, at most 20 ng/mL, at most 10 ng/mL, at most 5 ng/mL, or below 5 ng/mL. In some instances, the precursor media comprises Flt3 ligand at a concentration of below 50 ng/mL. In some instances, the precursor media comprises Flt3 ligand at a concentration of below 40 ng/mL. In some instances, the precursor media comprises Flt3 ligand at a concentration of below 30 ng/mL. In some instances, the precursor media comprises Flt3 ligand at a concentration of below 20 ng/mL. In some instances, the precursor media comprises Flt3 ligand at a concentration of below 10 ng/mL. In some instances, the precursor media comprises Flt3 ligand
at a concentration of below 5 ng/mL. In a preferred embodiment, the precursor mixture does not comprise Flt3 ligand.
Interleukin 3 (IL-3) can be within the precursor mixture at a concentration of at most 50 ng/mL, at most 40 ng/mL, at most 30 ng/mL, at most 20 ng/mL, at most 10 ng/mL, at most 5 ng/mL, or below 5 ng/mL. In some instances, the precursor media comprises IL-3 at a concentration of below 50 ng/mL. In some instances, the precursor media comprises IL-3 at a concentration of below 40 ng/mL. In some instances, the precursor media comprises IL-3 at a concentration of below 30 ng/mL. In some instances, the precursor media comprises IL-3 at a concentration of below 20 ng/mL. In some instances, the precursor media comprises IL-3 at a concentration of below 10 ng/mL. In some instances, the precursor media comprises IL-3 at a concentration of below 5 ng/mL. In a preferred embodiment, the precursor mixture does not comprise IL-3.
Interleukin 3 (IL-6) can be within the precursor mixture at a concentration of at most 50 ng/mL, at most 40 ng/mL, at most 30 ng/mL, at most 20 ng/mL, at most 10 ng/mL, at most 5 ng/mL, or below 5 ng/mL. In some instances, the precursor media comprises IL-6 at a concentration of below 50 ng/mL. In some instances, the precursor media comprises IL-6 at a concentration of below 40 ng/mL. In some instances, the precursor media comprises IL-6 at a concentration of below 30 ng/mL. In some instances, the precursor media comprises IL-6 at a concentration of below 20 ng/mL. In some instances, the precursor media comprises IL-6 at a concentration of below 10 ng/mL. In some instances, the precursor media comprises IL-6 at a concentration of below 5 ng/mL. In a preferred embodiment, the precursor mixture does not comprise IL-6.
Granulocyte-macrophage colony-stimulating factor (GM-CSF) can be within the precursor mixture at a concentration of at most 50 ng/mL, at most 40 ng/mL, at most 30 ng/mL, at most 20 ng/mL, at most 10 ng/mL, at most 5 ng/mL, or below 5 ng/mL. In some instances, the precursor media comprises GM-CSF at a concentration of below 50 ng/mL. In some instances, the precursor media comprises GM-CSF at a concentration of below 40 ng/mL. In some instances, the precursor media comprises GM-CSF at a concentration of below 30 ng/mL. In some instances, the precursor media comprises GM-CSF at a concentration of below 20 ng/mL. In some instances, the precursor media comprises GM-CSF at a concentration of below 10 ng/mL. In some instances, the precursor media comprises GM-CSF at a concentration of below 5 ng/mL. In a preferred embodiment, the precursor mixture does not comprise any GM-CSF.
Macrophage colony-stimulating factor (M-CSF) can be within the precursor mixture at a concentration of at most 50 ng/mL, at most 40 ng/mL, at most 30 ng/mL, at most 20 ng/mL, at
most 10 ng/mL, at most 5 ng/mL, or below 5 ng/mL. In some instances, the precursor media comprises M-CSF at a concentration of below 50 ng/mL. In some instances, the precursor media comprises M-CSF at a concentration of below 40 ng/mL. In some instances, the precursor media comprises M-CSF at a concentration of below 30 ng/mL. In some instances, the precursor media comprises M-CSF at a concentration of below 20 ng/mL. In some instances, the precursor media comprises M-CSF at a concentration of below 10 ng/mL. In some instances, the precursor media comprises M-CSF at a concentration of below 5 ng/mL. In a preferred embodiment, the precursor mixture does not comprise any M-CSF.
The precursor media disclosed herein can comprise at least one cytokine selected from the following: stem cells factor (SCF), Flt3 ligand, thrombopoietin (TPO), Interleukin-3 (IL-3); Granulocyte colony-stimulating factor (G-CSF), Interleukin-6 (IL-6), Granulocyte-macrophage colony-stimulating factor (GM-CSF), and Macrophage colony-stimulating factor (MCSF). In some embodiments, the precursor media comprises at least two cytokines selected from the following cytokines: stem cells factor (SCF), Flt3 ligand, thrombopoietin (TPO), Interleukin-3 (IL-3); Granulocyte colony-stimulating factor (G-CSF), Interleukin-6 (IL-6), Granulocyte-macrophage colony-stimulating factor (GM-CSF), and Macrophage colony-stimulating factor (MCSF). In some embodiments, the precursor media comprises at least three cytokines selected from the following cytokines: stem cells factor (SCF), Flt3 ligand, thrombopoietin (TPO), Interleukin-3 (IL-3); Granulocyte colony-stimulating factor (G-CSF), Interleukin-6 (IL-6), Granulocyte-macrophage colony-stimulating factor (GM-CSF), and Macrophage colony-stimulating factor (MCSF). In some embodiments, the precursor media comprises at least four cytokines selected from the following cytokines: stem cells factor (SCF), Flt3 ligand, thrombopoietin (TPO), Interleukin-3 (IL-3); Granulocyte colony-stimulating factor (G-CSF), Interleukin-6 (IL-6), Granulocyte-macrophage colony-stimulating factor (GM-CSF), and Macrophage colony-stimulating factor (MCSF). In some embodiments, the precursor media comprises four cytokines selected from the following cytokines: stem cells factor (SCF), Flt3 ligand, thrombopoietin (TPO), Interleukin-3 (IL-3); Granulocyte colony-stimulating factor (G-CSF), Interleukin-6 (IL-6), Granulocyte-macrophage colonystimulating factor (GM-CSF), and Macrophage colony-stimulating factor (MCSF). In some embodiments, the precursor cytokine mixture comprises G-CSF, GM-CSF, SCF, and TPO.
When the precursor media comprises G-CSF, the G-CSF can be present at a concentration between about 100 ng/mL and 200 ng/mL. In some embodiments, G-CSF is present within the precursor media at a concentration between about 100 ng/mL and 150 ng/mL. In such instances, the precursor media can further comprise at least one cytokine selected from the following: GM-CSF, SCF, and TPO. When the precursor media comprises G-CSF at a concentration between about 100
ng/mL and 150 ng/mL, said precursor media can further comprise GM-CSF, SCF, TPO, and any combination thereof. In some instances, the precursor media can comprise GM-CSF at a concentration between about 75 ng/mL and 125 ng/mL. In some embodiments, the precursor media can comprise SCF at a concentration between about 75 ng/mL and 125 ng/mL. In some instances, the precursor media can comprise TPO at a concentration between about 75 ng/mL and 125 ng/mL. In a preferred embodiment, the precursor media comprises G-CSF, GM-CSF, SCF, and TPO. In such instances, the precursor media comprises G-CSF at a concentration of 150 ng/mL, GM-CSF at a concentration of 100 ng/mL, SCF at a concentration of 100 ng/mL, and TPO at a concentration of 100 ng/mL.
In addition to a precursor cytokine mixture, the precursor media disclosed herein can comprise a basal growth medium. The basal growth medium can be any medium that supports growth, expansion and/or differentiation of precursor cells. The basal growth medium can comprise glucose, salts, amino acids, and vitamins. For example, the basal growth medium can contain a Roswell Park Memorial Institute (RPMI) 1640 medium, X-VIVO™-15 medium, CTS™ OPTMIZER™ Serum Free Medium, CTS™ OPTMIZER™ Pro Serum Free Medium, and/or IMMUNOCULT™-XF T Cell Expansion Medium. In some embodiments, the basal growth medium comprises the RPMI 1640 medium with ATCC modification (Gibco A1049101). In some embodiments, the basal growth media comprises VIVO™- 15 medium. In some embodiments, the basal growth media comprises Stemline® II Hematopoietic Stem Cell Expansion Media. When the basal growth media comprises X-Vivo, the VIVO™-15 medium can be combined or mixed with any other basal growth media at any ratio that is particularly effective. For example, the basal growth media can comprise VIVO™-15 medium and Stemline® II Hematopoietic Stem Cell Expansion Media mixed at a ratio. For example, the precursor media can comprise a basal growth media comprising VIVO™-15 medium and Stemline® II Hematopoietic Stem Cell Expansion Media mixed at a ratio of 1 : 1. Any volume of basal growth media is envisaged within the method disclosed herein. In some embodiments, the precursor media can comprise a culture medium. Any culture media known in the art that can support the growth, proliferation, expansion and/or differentiation of precursor cells within the precursor media is envisaged as being used with the precursor media disclosed herein. Cell culture methods, including hematopoietic stem cell culture methods, are widely available in the art, as described, for example, in Yadav, Pooja et al. “Hematopoietic Stem Cells Culture, Expansion and Differentiation: An Insight into Variable and Available Media.” International journal of stem cells vol. 13,3 (2020): 326-334, the contents of which are incorporated herein by reference in their entirety.
In some instances, the myeloid media can further comprise GlutaMAX™ Supplement. The myeloid media can comprise a myeloid cytokine mixture, a basal growth medium and GlutaMAX™ Supplement. The GlutaMAX™ Supplement can be present in the myeloid media at a concentration of between 0.5 mM, 1 mM, 1.5 mM, 2 mM, 2.5 mM or 3 mM of the myeloid media.
E. Differentiation of Precursor Cells, Precursor CAR cells, Myeloid-Derived Cells, and/or Myeloid-Derived CAR Cells
Disclosed herein are methods of manufacturing a myeloid-derived cell from a precursor cell, such as a stem cell and/or a CD34+ cell. In some instances, the stem cell is a hematopoietic stem cell. The methods of manufacturing a myeloid-derived cell disclosed herein can comprise the step of contacting a precursor cell with a precursor media comprising a precursor cytokine mixture, whereby the number of precursor cell(s) increases to generate a cell population comprising a plurality of precursor cells.
Also disclosed herein are methods of manufacturing a myeloid-derived cell disclosed herein comprising the step of contacting a precursor cell with a precursor media comprising a precursor cytokine mixture, whereby the number of precursor cell(s) increases to generate a cell population comprising a plurality of precursor cells, wherein said precursor cell is a HSC, whereby upon contact of said HSC with said precursor media, said HSC differentiates into a myeloid-lineage cell to generate a myeloid-derived cell and/or a mixed cell population comprising a plurality of myeloid-derived cells and a plurality of HSC cells.
Also disclosed herein are methods of manufacturing a myeloid-derived cell disclosed herein comprising the step of contacting a precursor cell with a precursor media comprising a precursor cytokine mixture, whereby the number of precursor cell(s) increases to generate a cell population comprising a plurality of precursor cells, wherein said precursor cell is a stem cell, wherein the method further comprises the step of contacting said cell population comprising a plurality of precursor cells with a myeloid media comprising a myeloid cytokine mixture, whereby said precursor cell(s) differentiates into a myeloid-lineage cell to generate a myeloid-derived cell and/or a mixed cell population comprising a plurality of myeloid-derived cells and/or a plurality of precursor cells.
Also disclosed herein are methods of manufacturing a myeloid-derived cell disclosed herein comprising the step of contacting a precursor cell with a precursor media comprising a precursor cytokine mixture, whereby the number of precursor cell(s) increases to generate a cell population comprising a plurality of precursor cells, wherein said precursor cell is a HSC, whereby upon contact of said HSC with said precursor media, said HSC differentiates into a myeloid-lineage cell to generate a myeloid-derived cell and/or a mixed cell population comprising a plurality of
myeloid-derived cells and a plurality of HSC cells, wherein the method further comprises the step of: contacting said mixed cell population with a myeloid media comprising a myeloid cytokine mixture, whereby upon contact with said myeloid media: a. the ratio of said myeloid-derived cells to said precursor cells in said mixed cell population is increased.
Any of the above methods described herein can involve contacting a stem cell comprising a CAR (i.e., a precursor CAR cell) with a precursor media to increase to generate a cell population comprising a plurality of precursor cells. In such instances, the precursor cell can be an HSC. Such methods can further comprise differentiating said precursor CAR cell into a myeloid-derived CAR cell using any of the methods and/or steps described herein to generate a cell population comprising a plurality of myeloid-derived CAR cells.
The methods of manufacturing a myeloid-derived cell disclosed herein can comprise the step of contacting a precursor cell with a precursor media comprising a precursor cytokine mixture, whereby the number of precursor cell(s) increases to generate a cell population comprising a plurality of precursor cells, wherein said precursor cell is a stem cell, whereby upon contact of said precursor cell(s) with said precursor media, said precursor cell(s) differentiates into a myeloid- lineage cell to generate a myeloid-derived cell and/or a mixed cell population comprising a plurality of myeloid-derived cells and a plurality of precursor cells. In such instances, the method can further comprise the step of contacting said mixed cell population with a myeloid media comprising a myeloid cytokine mixture, whereby upon contact with said myeloid media: a. the ratio of said myeloid-derived cells to said precursor cells in said mixed cell population is increased.
Disclosed herein are compositions and methods that are particularly effective at promoting the differentiation of precursor cells, such as stem cells. The stem cells can be CD34+ and/or hematopoietic stem cells. In particular, the methods and compositions disclosed herein are effective at promoting the differentiation of stem cells into myeloid cells. In some embodiments, the stem cells comprise a CAR and are differentiated into a myeloid-derived CAR cell. In some embodiments, the stem cells are hematopoietic stem cells. For instance, the disclosed compositions and methods can effectively promote the differentiation of a hematopoietic stem cell into a myeloid lineage cell. The precursor cell can comprise any CAR available in the art. The precursor cell can be comprised within a cell population comprising at least one precursor cell. In some embodiments, the cell population comprises a plurality of precursor cells. In such embodiments, the plurality of precursor cells can comprise precursor cells comprising a CAR disclosed herein.
Disclosed herein are methods of generating a genetically modified myeloid-derived CAR cell from a precursor cell, such as a hematopoietic stem cell, wherein the precursor cell comprising a CAR (i.e., precursor CAR cell) is expanded by contacting said precursor CAR cell with a precursor media
comprising a precursor cytokine mixture, whereby the number of precursor CAR cell(s) increases to generate a population of precursor cells (e.g., stem cells) comprising a plurality of precursor CAR cells. In some instances, the method further comprises the step of contacting said precursor CAR cell(s) with a myeloid media comprising a myeloid cytokine mixture, whereby said precursor cell differentiates into a myeloid-lineage cell to generate a myeloid-derived CAR cell or a cell population comprising a plurality of myeloid-derived CAR cells, wherein said cell population is a mixed population comprising said precursor CAR cells and said myeloid-derived CAR cells.
Disclosed herein are methods of generating a genetically modified myeloid-derived CAR cell from a precursor cell, such as a hematopoietic stem cell, wherein the precursor cell comprising a CAR (i.e., precursor CAR cell) is expanded by contacting said precursor CAR cell with a precursor media comprising a precursor cytokine mixture, whereby the number of precursor CAR cell(s) increases to generate a population of precursor cells (e.g., stem cells) comprising a plurality of precursor CAR cells, further comprising the step of contacting said precursor CAR cell(s) with a myeloid media comprising a myeloid cytokine mixture, whereby said precursor cell differentiates into a myeloid-lineage cell to generate a myeloid-derived CAR cell or a cell population comprising a plurality of myeloid-derived CAR cells, wherein said cell population is a mixed population comprising said precursor CAR cells and said myeloid-derived CAR cells.
Also disclosed herein are methods of generating a genetically modified myeloid-derived CAR cell from a precursor cell, such as a hematopoietic stem cell, wherein the precursor cell comprising a CAR (i.e., precursor CAR cell) is expanded by contacting said precursor CAR cell with a precursor media comprising a precursor cytokine mixture, whereby the number of precursor CAR cell(s) increases to generate a population of precursor cells (e.g., stem cells) comprising a plurality of precursor CAR cells, wherein following contact of said precursor CAR cell(s) within said population with said precursor media, said precursor CAR cell(s) differentiates into a myeloid-lineage cell(s) to generate a myeloid-derived CAR cell(s) or a population comprising a plurality of myeloid-derived CAR cell(s). In some instances, the cell population is a mixed population comprising said myeloid- derived CAR cells and a plurality of precursor CAR cells. In some instances, the method further comprises the step of contacting said precursor CAR cell, myeloid-derived CAR cell, and/or said population comprising precursor CAR cells and said myeloid-derived CAR cells with a myeloid media comprising a myeloid cytokine mixture, whereby said ratio of myeloid-derived CAR cells relative to precursor CAR cells within said population is increased.
Also disclosed herein are methods of generating a genetically modified myeloid-derived CAR cell from a precursor cell, such as a hematopoietic stem cell, wherein the precursor cell comprising a CAR (i.e., precursor CAR cell) is expanded by contacting said precursor CAR cell with a precursor
media comprising a precursor cytokine mixture, whereby the number of precursor CAR cell(s) increases to generate a population of precursor cells (e.g., stem cells) comprising a plurality of precursor CAR cells, wherein following contact of said precursor CAR cell(s) within said population with said precursor media, said precursor CAR cell(s) differentiates into a myeloid-lineage cell(s) to generate a myeloid-derived CAR cell(s) or a population comprising a plurality of myeloid-derived CAR cell(s), wherein the cell population is a mixed population comprising said myeloid-derived CAR cells and a plurality of precursor CAR cells.
Also disclosed herein are methods of generating a genetically modified myeloid-derived CAR cell from a precursor cell, such as a hematopoietic stem cell, wherein the precursor cell comprising a CAR (i.e., precursor CAR cell) is expanded by contacting said precursor CAR cell with a precursor media comprising a precursor cytokine mixture, whereby the number of precursor CAR cell(s) increases to generate a population of precursor cells (e.g., stem cells) comprising a plurality of precursor CAR cells, wherein following contact of said precursor CAR cell(s) within said population with said precursor media, said precursor CAR cell(s) differentiates into a myeloid-lineage cell(s) to generate a myeloid-derived CAR cell(s) or a population comprising a plurality of myeloid-derived CAR cell(s). In some instances, the cell population is a mixed population comprising said myeloid- derived CAR cells and a plurality of precursor CAR cells, wherein the method further comprises the step of contacting said precursor CAR cell, myeloid-derived CAR cell, and/or said population comprising precursor CAR cells and said myeloid-derived CAR cells with a myeloid media comprising a myeloid cytokine mixture, whereby said ratio of myeloid-derived CAR cells relative to precursor CAR cells within said population is increased.
In some embodiments, the methods disclosed herein comprise differentiation of at least one precursor cell. Said at least one precursor cell can be comprised with in a cell population. The cell population disclosed herein can comprise a plurality of precursor cell. In such instances, the methods disclosed herein can comprise differentiation of a plurality of precursor cells with a cell population.
The methods disclosed herein are particularly effective in their ability to promote differentiation of a high percentage of a plurality of cells within a cell population compared to a control. A control can include a method that does not use the media disclosed herein.
The method of manufacturing a genetically modified myeloid-derived CAR cell disclosed herein can further comprise the step of differentiating genetically modified precursor CAR cell(s) in a myeloid media comprising a myeloid cytokine mixture to generate a myeloid-derived CAR cell or a population comprising a plurality of genetically modified myeloid-derived CAR cells. The differentiating step disclosed herein is particularly effective at promoting the differentiation of a
genetically modified precursor CAR cell, or a plurality of genetically modified precursor CAR cells, toward a myeloid-derived CAR cell, or a plurality of genetically modified myeloid-derived CAR cells, respectively. In some instances, the step of differentiating the genetically modified precursor CAR cell(s) in a myeloid media occurs after the precursor CAR cell(s) has started expanding. In some instances, the step of differentiating the genetically modified precursor CAR cell(s) in a myeloid media occurs after the precursor CAR cell(s) has started differentiating. When the step of differentiating the genetically modified precursor CAR cell(s) in a myeloid media occurs after the precursor CAR cell(s) has started differentiating, the precursor CAR cell(s) has started differentiating in a precursor media comprising a precursor cytokine mixture. The step of differentiating the genetically modified precursor CAR cell(s) in a myeloid media further promotes the differentiation of the genetically modified precursor CAR cell(s) toward the myeloid lineage. The step of differentiating the genetically modified precursor CAR cell(s) in a myeloid media is particularly effective at promoting the differentiation of the genetically modified precursor CAR cell(s) toward the myeloid lineage.
When the genetically modified precursor CAR cells are differentiated in a myeloid media comprising a myeloid cytokine mixture, the precursor media can be removed prior to addition of the myeloid media. The removal of the precursor media serves to remove all contents of the precursor media, including the precursor cytokine mixture. Any method of removing the precursor media comprising the precursor cytokine mixture, prior to the addition of the myeloid media is envisaged within the method. A skilled artisan would understand that removing the precursor media comprising the precursor cytokine mixture can for example, involve washing the cells. In some instances, the cells are washed with a sterile solution. When the cells are washed with a sterile solution, centrifugation can then be utilized to separate the cells from the sterile solution. Once the cells are separated, they can be suspended at a desired concentration in for example, a myeloid media comprising a myeloid cytokine mixture. Therefore, in some methods disclosed herein, after the genetically modified precursor CAR cells are expanded in a precursor media comprising a cytokine mixture, the cells are removed from the precursor media and suspended in a myeloid media comprising a myeloid cytokine mixture. Within the myeloid media, the cells can be mixed to promote differentiation.
Differentiation of genetically modified precursor CAR cells can increase the amount of the differentiated cells by about 10 fold, 20 fold, 30 fold, 40 fold, 50 fold, 60 fold, 70 fold, 80 fold, 90 fold, 100 fold, 200 fold, 300 fold, 400 fold, 500 fold, 600 fold, 700 fold, 800 fold, 900 fold, 1000 fold, 2000 fold, 3000 fold, 4000 fold, 5000 fold, 6000 fold, 7000 fold, 8000 fold, 9000 fold, 10,000 fold, 100,000 fold, 1,000,000 fold, 10,000,000 fold, or greater. Without wishing to be bound by theory, as
the number of differentiated cells within a population, increases, the number of undifferentiated cells within the population decreases. For example, as the number of genetically modified precursor CAR cell(s) that are differentiated in a myeloid media increases, the number of genetically modified precursor CAR cell(s) that are undifferentiated in the myeloid media decreases. Differentiation of genetically modified precursor CAR cells can increase the ratio of the differentiated cells to undifferentiated cells within a population by a factor of 10 fold, 20 fold, 30 fold, 40 fold, 50 fold, 60 fold, 70 fold, 80 fold, 90 fold, 100 fold, 200 fold, 300 fold, 400 fold, 500 fold, 600 fold, 700 fold, 800 fold, 900 fold, 1000 fold, 2000 fold, 3000 fold, 4000 fold, 5000 fold, 6000 fold, 7000 fold, 8000 fold, 9000 fold, 10,000 fold, 100,000 fold, 1,000,000 fold, 10,000,000 fold, or greater.
The differentiated cells of the present disclosure are preferential differentiated toward the myeloid cell lineage. In some instances, the step of differentiating a genetically modified precursor CAR cell in a myeloid media generates a myeloid-derived CAR cell. In some instances, the step of differentiating a genetically modified precursor CAR cell(s) in a myeloid media generates a plurality of genetically modified myeloid-derived CAR cells. As the plurality of genetically modified precursor CAR cells is differentiated toward the myeloid cell lineage (i.e., myeloid-derived cell), the ratio of precursor (e.g., stem cells) within the plurality is decreased relative to myeloid-derived cells. For example, the ratio of CD34+ cells decreases as the plurality of precursor CAR cells differentiates. Differentiation can decrease the amount of CD34+ cells within a plurality of genetically modified precursor CAR cells or a plurality of genetically modified myeloid-derived cells. Differentiation of genetically modified precursor CAR cells can decrease the amount of CD34+ cells within a plurality of genetically modified precursor CAR cells or a plurality of genetically modified myeloid-derived cells by about 10 fold, 20 fold, 30 fold, 40 fold, 50 fold, 60 fold, 70 fold, 80 fold, 90 fold, 100 fold, 200 fold, 300 fold, 400 fold, 500 fold, 600 fold, 700 fold, 800 fold, 900 fold, 1000 fold, 2000 fold, 3000 fold, 4000 fold, 5000 fold, 6000 fold, 7000 fold, 8000 fold, 9000 fold, 10,000 fold, 100,000 fold, 1,000,000 fold, 10,000,000 fold, or greater.
The term “differentiation” when used in the present disclosure refers to an increase in the amount or number of surface markers on at least one cell (e.g., a precursor cell), wherein said surface markers are myeloid-associated markers. A “myeloid-associated marker” can be any surface marker that is known and available in the art for identifying and/or isolating a cell within the myeloid lineage. A person having skill in the art would understand how to determine if a cell was in the myeloid lineage and to identify a surface marker specific for said cell. Exemplary, non-limiting, myeloid-associated markers can include: CD1 lb, CD14, CD13, and CD15. A myeloid cell can have more than one myeloid-associated marker.
One key aspect of the differentiation step is to increase the number of myeloid-derived cells within a myeloid media. In some instances, the myeloid-derived cells are present within a population, such as a mixed population comprising a plurality of precursor CAR cells and myeloid-derived CAR cells. In some embodiments, the differentiation step increases the number of myeloid-derived cells in a population comprising a plurality of precursor CAR cells. The differentiation step of the methods disclosed herein can increase the amount of the differentiated cells by about 10 fold, 20 fold, 30 fold, 40 fold, 50 fold, 60 fold, 70 fold, 80 fold, 90 fold, 100 fold, 200 fold, 300 fold, 400 fold, 500 fold, 600 fold, 700 fold, 800 fold, 900 fold, 1000 fold, 2000 fold, 3000 fold, 4000 fold, 5000 fold, 6000 fold, 7000 fold, 8000 fold, 9000 fold, 10,000 fold, 100,000 fold, 1,000,000 fold, 10,000,000 fold, or greater. In some embodiments, differentiation can increase the amount of myeloid-derived CAR cells between 10-fold to 100-fold, between 20-fold to 200-fold, between 30-fold to 300-fold, between 40-fold to 400- fold, between 50-fold to 500-fold, between 60-fold to 600-fold, between 70-fold to 700-fold, between 80-fold to 800-fold, between 90-fold to 900-fold, between 100-fold to 1000-fold, or greater than 1000- fold. Without wishing to be bound by theory, as the number of differentiated cells (e.g., myeloid cells) within a population increases, the number of undifferentiated cells (e.g., stem cells) within the population decreases. For example, as the number of genetically modified precursor CAR cell(s) that are differentiated in a myeloid media increases, the number of genetically modified precursor CAR cell(s) that are undifferentiated in the myeloid media decreases. Differentiation of genetically modified precursor CAR cells can increase the ratio of the differentiated cells (i.e., myeloid-derived cells) to undifferentiated cells (i.e., stem cells) within a population by a factor of 10 fold, 20 fold, 30 fold, 40 fold, 50 fold, 60 fold, 70 fold, 80 fold, 90 fold, 100 fold, 200 fold, 300 fold, 400 fold, 500 fold, 600 fold, 700 fold, 800 fold, 900 fold, 1000 fold, 2000 fold, 3000 fold, 4000 fold, 5000 fold, 6000 fold, 7000 fold, 8000 fold, 9000 fold, 10,000 fold, 100,000 fold, 1,000,000 fold, 10,000,000 fold, or greater.
In some embodiments, the precursor CAR cell(s) is maintained in said myeloid medium until the number of cells is at least 104, at least 105, at least 106, at least 107, at least 108, at least 109, at least 1010, at least 1011, at least 1012, at least 1013, at least 1014, at least 1015, at least 1016, at least 1017, at least 1018, at least 1019, at least IO20, or greater than IO20. In some embodiments, the precursor CAR cell(s) is maintained in said myeloid medium until the number of cells within the generated population is at least 104, at least 105, at least 106, at least 107, at least 108, at least 109, at least 1010, at least 1011, at least 1012, at least 1013, at least 1014, at least 1015, at least 1016, at least 1017, at least 1018, at least 1019, at least IO20, or greater than IO20. In some embodiments, the precursor CAR cell(s) is maintained in said myeloid medium to generate a population of cells comprising a plurality of precursor CAR cells and/or a plurality of myeloid-derived CAR cells, wherein the number of cells within the population is at least 104, at least 105, at least 106, at least 107, at least 108, at least 109, at least 1010, at least 1011, at
least 1012, at least IO13, at least 1014, at least IO15, at least 1016, at least 1017, at least 1018, at least 1019, at least IO20, or greater than IO20. In some embodiments, the precursor CAR cell(s) is maintained in said myeloid medium to generate a population of cells comprising a plurality of precursor CAR cells and/or a plurality of myeloid-derived CAR cells, wherein the number of myeloid-derived CAR cells within the population is at least 10, at least 102, at least 103, at least 104, at least 105, at least 106, at least 107, at least 108, at least 109, at least IO10, at least 1011, at least 1012, at least 1013, at least 1014, at least 1015, at least 1016, at least 1017, at least 1018, at least 1019, at least IO20, or more than IO20 higher than the number of precursor CAR cells. In some embodiments, the precursor CAR cell(s) is maintained in said myeloid medium to generate a population of cells comprising a plurality of precursor CAR cells and/or a plurality of myeloid-derived CAR cells, wherein the ratio of precursor CAR cells to myeloid-derived CAR cells within the population is at least 1 : 10 (1 precursor CAR cell to 10 myeloid-derived CAR cells), at least 1 : 102, at least 1 : 103, at least l: 104, at least 1 : 105, at least 1 : 106, at least l: 107, at least l :108, at least 1 : 109, at least 1 : IO10, at least lilO11, at least 1 : 1012, at least 1 : 1013, at least l: 1014, at least 1 : 1015, at least 1 : 1016, at least 1 : 1017, at least 1 : 1018, at least 1 : 1019, at least 1 : IO20, or more than 1 : IO20. In some embodiments, the precursor CAR cell(s) is maintained in the myelid medium for at least 2, at least 3, at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, at least 10, at least 11, or at least 12 days. In some embodiments, the precursor CAR cell(s), and/or population comprising a plurality of precursor CAR cells and/or plurality of myeloid-derived CAR cells, are maintained in myeloid media for between 9-11 days. In some embodiments, the precursor CAR cell(s), and/or population comprising a plurality of precursor CAR cells and/or plurality of myeloid-derived CAR cells, are maintained in precursor media for 10 days.
Table 2. Optimal Timeline of Myeloid Expansion and Differentiation of Hematopoietic Stem Cells
The differentiated cells of the present disclosure are preferentially differentiated toward the myeloid cell lineage. In some instances, the step of differentiating a genetically modified precursor CAR cell in a myeloid media generates a myeloid-derived CAR cell. In some instances, the step of differentiating a genetically modified precursor CAR cell(s) in a myeloid media generates a plurality of genetically modified myeloid-derived CAR cells. As the plurality of genetically modified precursor CAR cells is differentiated toward the myeloid cell lineage (i.e., myeloid-derived cell), the ratio of precursor (e.g., stem cells) within the plurality is decreased relative to myeloid-derived cells. For example, the ratio of CD34+ cells decreases as the plurality of precursor CAR cells differentiates. Differentiation can decrease the amount of CD34+ cells within a plurality of genetically modified precursor CAR cells or a plurality of genetically modified myeloid-derived cells. Differentiation of genetically modified precursor CAR cells can decrease the amount of CD34+ cells within a plurality of genetically modified precursor CAR cells or a plurality of genetically modified myeloid-derived cells by about 10 fold, 20 fold, 30 fold, 40 fold, 50 fold, 60 fold, 70 fold, 80 fold, 90 fold, 100 fold, 200 fold, 300 fold, 400 fold, 500 fold, 600 fold, 700 fold, 800 fold, 900 fold, 1000 fold, 2000 fold, 3000 fold, 4000 fold, 5000 fold, 6000 fold, 7000 fold, 8000 fold, 9000 fold, 10,000 fold, 100,000 fold, 1,000,000 fold, 10,000,000 fold, or greater. Myeloid cells can be identified and/or isolated based on cell surface markers. A skilled artisan would understand that specific myeloid cells within the myeloid lineage can be identified based on specific cell surface markers. Exemplary, non-limiting, surface markers for myeloid cells include: CD13, CD14, CD15 and CD1 lb. Accordingly, during the expansion step, the number of cells expression myeloid-associated cell surface markers can be expected to increase, while the number of cells expressing a common stem cell surface marker (i.e., CD34) can be expected to decrease as differentiation occurs.
The differentiation step of the methods disclosed herein can increase the amount of CD1 lb+ cells within a population comprising a plurality of precursor CAR cells or a plurality of myeloid- derived cells. Differentiation of genetically modified precursor CAR cells can increase the amount of CD1 lb+ cells within a population comprising a plurality of precursor CAR cells and/or a plurality of myeloid-derived CAR cells by about 10 fold, 20 fold, 30 fold, 40 fold, 50 fold, 60 fold, 70 fold, 80 fold, 90 fold, 100 fold, 200 fold, 300 fold, 400 fold, 500 fold, 600 fold, 700 fold, 800 fold, 900 fold, 1000 fold, 2000 fold, 3000 fold, 4000 fold, 5000 fold, 6000 fold, 7000 fold, 8000 fold, 9000 fold, 10,000 fold, 100,000 fold, 1,000,000 fold, 10,000,000 fold, or greater. In some embodiments, a precursor CAR cell, or population comprising a plurality of precursor CAR cells, or a population comprising a plurality of precursor CAR cells and a plurality of myeloid-derived CAR cells is maintained in a myeloid media until the number of CD1 lb+ cells within the population is increased between 10 fold to 100 fold, between 20 fold and 200 fold, between 30 fold and 300 fold, between 40
fold and 400 fold, between 50 fold and 500 fold, between 60 fold and 600 fold, between 70 fold and 700 fold, between 80 fold and 800 fold, between 90 fold and 900 fold, between 100 fold and 10,000 fold, between 1000 fold and 100,000 fold, between 10,000 fold and 1,000,000 fold, between 100,000 fold and 10,000,000 fold , between 105 fold and 107 fold, between 106 fold and 108 fold, between 107 fold and 109 fold, 108 fold and 1010 fold, 109 fold and 1011 fold, 1010 fold and 1012 fold, 1011 fold and 1013 fold, 1012 fold and 1014 fold, 1013 fold and 1015 fold, 1014 fold and 1016 fold, or greater than 1016 fold.
The differentiation step of the methods disclosed herein can increase the amount of CD14+ cells within a population comprising a plurality of precursor CAR cells or a plurality of myeloid- derived CAR cells. Differentiation of precursor CAR cells can increase the amount of CD14+ cells within a population comprising a plurality of precursor CAR cells and/or a plurality of myeloid- derived CAR cells by about 10 fold, 20 fold, 30 fold, 40 fold, 50 fold, 60 fold, 70 fold, 80 fold, 90 fold, 100 fold, 200 fold, 300 fold, 400 fold, 500 fold, 600 fold, 700 fold, 800 fold, 900 fold, 1000 fold, 2000 fold, 3000 fold, 4000 fold, 5000 fold, 6000 fold, 7000 fold, 8000 fold, 9000 fold, 10,000 fold, 100,000 fold, 1,000,000 fold, 10,000,000 fold, or greater. In some embodiments, a precursor CAR cell, or population comprising a plurality of precursor CAR cells, or a population comprising a plurality of precursor CAR cells and a plurality of myeloid-derived CAR cells is maintained in a myeloid media until the number of CD14+ cells within the population is increased between 10 fold to 100 fold, between 20 fold and 200 fold, between 30 fold and 300 fold, between 40 fold and 400 fold, between 50 fold and 500 fold, between 60 fold and 600 fold, between 70 fold and 700 fold, between 80 fold and 800 fold, between 90 fold and 900 fold, between 100 fold and 10,000 fold, between 1000 fold and 100,000 fold, between 10,000 fold and 1,000,000 fold, between 100,000 fold and 10,000,000 fold , between 105 fold and 107 fold, between 106 fold and 108 fold, between 107 fold and 109 fold, 108 fold and 1010 fold, 109 fold and 1011 fold, 1010 fold and 1012 fold, 1011 fold and 1013 fold, 1012 fold and 1014 fold, 1013 fold and 1015 fold, 1014 fold and 1016 fold, or greater than 1016 fold.
The differentiation step of the methods disclosed herein can increase the amount of CD 13+ cells within a population comprising a plurality of precursor CAR cells and/or a plurality of myeloid- derived CAR cells. Differentiation of precursor CAR cells can increase the amount of CD 13+ cells within a population comprising a plurality of precursor CAR cells and/or a plurality of myeloid- derived CAR cells by about 10 fold, 20 fold, 30 fold, 40 fold, 50 fold, 60 fold, 70 fold, 80 fold, 90 fold, 100 fold, 200 fold, 300 fold, 400 fold, 500 fold, 600 fold, 700 fold, 800 fold, 900 fold, 1000 fold, 2000 fold, 3000 fold, 4000 fold, 5000 fold, 6000 fold, 7000 fold, 8000 fold, 9000 fold, 10,000 fold, 100,000 fold, 1,000,000 fold, 10,000,000 fold, or greater. In some embodiments, a precursor CAR cell, or population comprising a plurality of precursor CAR cells, or a population comprising a plurality of
precursor CAR cells and a plurality of myeloid-derived CAR cells is maintained in a myeloid media until the number of CD 13+ cells within the population is increased between 10 fold to 100 fold, between 20 fold and 200 fold, between 30 fold and 300 fold, between 40 fold and 400 fold, between 50 fold and 500 fold, between 60 fold and 600 fold, between 70 fold and 700 fold, between 80 fold and 800 fold, between 90 fold and 900 fold, between 100 fold and 10,000 fold, between 1000 fold and 100,000 fold, between 10,000 fold and 1,000,000 fold, between 100,000 fold and 10,000,000 fold , between 105 fold and 107 fold, between 106 fold and 108 fold, between 107 fold and 109 fold, 108 fold and 1010 fold, 109 fold and 1011 fold, 1010 fold and 1012 fold, 1011 fold and 1013 fold, 1012 fold and 1014 fold, 1013 fold and 1015 fold, 1014 fold and 1016 fold, or greater than 1016 fold.
The differentiation step of the methods disclosed herein can increase the amount of CD 15+ cells within a population comprising a plurality of precursor CAR cells and/or a plurality of myeloid-derived CAR cells. Differentiation of genetically modified precursor CAR cells can increase the amount of CD 15+ cells within a plurality of genetically modified precursor CAR cells or a plurality of genetically modified myeloid-derived cells by about 10 fold, 20 fold, 30 fold, 40 fold, 50 fold, 60 fold, 70 fold, 80 fold, 90 fold, 100 fold, 200 fold, 300 fold, 400 fold, 500 fold, 600 fold, 700 fold, 800 fold, 900 fold, 1000 fold, 2000 fold, 3000 fold, 4000 fold, 5000 fold, 6000 fold, 7000 fold, 8000 fold, 9000 fold, 10,000 fold, 100,000 fold, 1,000,000 fold, 10,000,000 fold, or greater. In some embodiments, a precursor CAR cell, or population comprising a plurality of precursor CAR cells, or a population comprising a plurality of precursor CAR cells and a plurality of myeloid-derived CAR cells is maintained in a myeloid media until the number of CD 15+ cells within the population is increased between 10 fold to 100 fold, between 20 fold and 200 fold, between 30 fold and 300 fold, between 40 fold and 400 fold, between 50 fold and 500 fold, between 60 fold and 600 fold, between 70 fold and 700 fold, between 80 fold and 800 fold, between 90 fold and 900 fold, between 100 fold and 10,000 fold, between 1000 fold and 100,000 fold, between 10,000 fold and 1,000,000 fold, between 100,000 fold and 10,000,000 fold , between 105 fold and 107 fold, between 106 fold and 108 fold, between 107 fold and 109 fold, 108 fold and 1010 fold, 109 fold and 1011 fold, 1010 fold and 1012 fold, 1011 fold and 1013 fold, 1012 fold and 1014 fold, 1013 fold and 1015 fold, 1014 fold and 1016 fold, or greater than 1016 fold.
The amount of cell surface marker expression within a population comprising a plurality of precursor CAR cells and/or a plurality of myeloid-derived CAR cells can be used to determine how long to maintain the differentiation step of the method of manufacturing a genetically modified myeloid-derived CAR cells disclosed herein. In some instances, the differentiation step is maintained until a certain percentage of the population comprising a plurality of genetically modified precursor CAR cells and/or genetically modified myeloid-derived CAR cells expresses a specific marker. In
some instances, the differentiation step is maintained until a certain percentage of the population comprising a plurality of genetically modified precursor CAR cells and/or plurality of genetically modified myeloid-derived cells expresses at least a specific marker. In some instances, the differentiation step is maintained until a certain percentage of the population comprising a plurality of genetically modified precursor CAR cells and/or plurality of genetically modified myeloid-derived CAR cells expresses at least two specific markers. In some instances, the differentiation step is maintained until a certain percentage of the population comprising a plurality of genetically modified precursor CAR cells and/or plurality of genetically modified myeloid-derived CAR cells expresses at least three specific markers. In some instances, the differentiation step is maintained until a certain percentage of the population comprising a plurality of genetically modified precursor CAR cells and/or plurality of genetically modified myeloid-derived CAR cells expresses at least more than three different specific markers.
Importantly, a skilled artisan would understand that as a precursor CAR cell is maintained in a myeloid media, said cell will differentiate into a myeloid-derived CAR cell. Accordingly, for the purposes of the present application, any reference to “a precursor cell” that is, or has been, maintained in a myeloid media for a period of time after which said precursor cell that was initially contacted with said myeloid media would reasonably be expected to have differentiated into a myeloid-derived cell, is intended to encompass any cell population that arises as a result of said precursor cell being maintained in myeloid. A cell population would be understood to arise as a result of the precursor cell if, as described above, said population comprises at least one cell that was generated from, for example, at least one cell differentiation event of said precursor cell that was contacted with myeloid media.
The precursor CAR cell, and/or population comprising a plurality of myeloid-derived CAR cells and/or a plurality of precursor CAR cells, can be contacted with, and maintained in a myeloid media (i.e., differentiation step) for a short period of time, for example less than 24 hours, such as 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, or 23 hours. The cell(s) and/or population can be maintained in myeloid media for a longer period of time, for example, for 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, or more days. In some embodiments, the differentiation step may be maintained for several hours (about 3 hours) to about 14 days or any hourly integer value in between. During the differentiation step, the precursor CAR cell, and/or population comprising a plurality of myeloid-derived CAR cells and/or a plurality of precursor CAR cells, can be contacted with, and maintained in a myeloid media. The differentiation step can occur after an expansion step. Accordingly, the precursor CAR cell, and/or population comprising a plurality of myeloid-derived CAR cells and/or a plurality of precursor CAR cells, can be contacted with, and maintained in a myeloid media after being contacted with and maintained in a precursor media. In
some embodiments, the differentiation step is maintained for at least 1, at least 2, at least 3, at least 4, or at least 5 days. In some embodiments, the differentiation step is maintained for 1 day, for 2 days, for 3 days, for 4 days, or for 5 days. In some embodiments, the differentiation step can be about 1 day. In some embodiments, the differentiation step is maintained for about 2 days. In some embodiments, the differentiation step is maintained for about 3 days. In some embodiments, the differentiation step is maintained for about 4 days. In some embodiments, the differentiation step is maintained for about 5 days. In some embodiments, the differentiation step is maintained for about 6 days. In some embodiments, the differentiation step is maintained for about 7 days. In some embodiments, the differentiation step is maintained for about 8 days. In some embodiments, the differentiation step is maintained for about 9 days. In some embodiments, the differentiation step is maintained for about 10 days. In some embodiments, the differentiation step is maintained for about 11 days. In some embodiments, the differentiation step is maintained for about 12 days. In some embodiments, the differentiation step is maintained for about 13 days. In some embodiments, the differentiation step is maintained for about 14 days. In some embodiments, the differentiation step is maintained for about 15 days. In some embodiments, the differentiation step is maintained for at least 2, at least 3, at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, at least 10, at least 11, or at least 12 days. In some embodiments, the differentiation step is maintained for between 9-11 days. In some embodiments, the expanding step is maintained for 10 days.
The differentiation step (i.e., maintaining a precursor CAR cell, and/or a population comprising a plurality of precursor CAR cells and/or a plurality of myeloid-derived cells in myeloid media) can also be maintained for a set number of hours. In some embodiments, the differentiation step is maintained for about 12 hours. In some embodiments, the differentiation step is maintained for about 24 hours. In some embodiments, the differentiation step is maintained for about 48 hours. In some embodiments, the differentiation step is maintained for about 72 hours. In some embodiments, the differentiation step is maintained for about 96 hours. In some embodiments, the differentiation step is maintained for about 120 hours. In some embodiments, the differentiation step is maintained for about 144 hours. In some embodiments, the differentiation step is maintained for about 168 hours. In some embodiments, the differentiation step is maintained for about 192 hours. In some embodiments, the differentiation step is maintained for about 216 hours. In some embodiments, the differentiation step is maintained for about 240 hours. In some embodiments, the differentiation step is maintained for about 264 hours. In some embodiments, the differentiation step is maintained for about 288 hours. In some embodiments, the differentiation step is maintained for about 312 hours. In some embodiments, the differentiation step is maintained for about 336 hours. In some embodiments, the differentiation step is maintained for about 360
hours. In some embodiments, the differentiation step is maintained for about 384 hours. In some embodiments, the differentiation step is maintained for about 408 hours. In some embodiments, the differentiation step is maintained for about 432 hours. In some embodiments, the differentiation step is maintained for about 456 hours. In some embodiments, the differentiation step is maintained for more than 456 hours.
In some embodiments, the differentiation step is maintained between 12-48 hours. In some embodiments, the differentiation step is maintained between 24-72 hours. In some embodiments, the differentiation step is maintained between 48-96 hours. In some embodiments, the differentiation step is maintained between 72-120 hours. In some embodiments, the differentiation step is maintained between 96-144 hours. In some embodiments, the differentiation step is maintained between 120-168 hours. In some embodiments, the differentiation step is maintained between 144-192 hours. In some embodiments, the differentiation step is maintained between 168-216 hours. In some embodiments, the differentiation step is maintained between 192-240 hours. In some embodiments, the differentiation step is maintained between 216-264 hours. In some embodiments, the differentiation step is maintained between 240-288 hours.
In some embodiments, the differentiation step is maintained between 264-312 hours. In some embodiments, the differentiation step is maintained between 288-336 hours. In some embodiments, the differentiation step is maintained between 312-360 hours. In some embodiments, the differentiation step is maintained between 336-384 hours. In some embodiments, the differentiation step is maintained between 360-408 hours. In some embodiments, the differentiation step is maintained between 384-432 hours. In some embodiments, the differentiation step is maintained between 408-456 hours.
In some instances, the differentiation step is maintained until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population comprising a plurality of myeloid-derived CAR cells is CD1 lb+. In some instances, the differentiation step is maintained until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population comprising a plurality of genetically modified precursor CAR cells and/or a plurality of genetically modified myeloid-derived CAR cells is CD1 lb+. In some instances, the precursor CAR cell(s) and/or population comprising a plurality of precursor CAR cells is maintained in said myeloid media until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population is CD1 lb+. In some instances, the precursor CAR cell(s) and/or population comprising a plurality of precursor CAR cells and/or plurality of
myeloid-derived CAR cells is maintained in said myeloid media until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population is CD1 lb+. In some instances, the population comprising a plurality of myeloid-derived CAR cells is maintained in said myeloid media until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population is CD1 lb+.
In some instances, the precursor CAR cell(s) and/or population comprising a plurality of precursor CAR cells and/or plurality of myeloid-derived CAR cells is maintained in said myeloid media until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population is CD1 lb+CD13+.
In some instances, the precursor CAR cell(s) and/or population comprising a plurality of precursor CAR cells and/or plurality of myeloid-derived CAR cells is maintained in said myeloid media until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population is CD1 lb+CD13+CD14+.
In some instances, the precursor CAR cell(s) and/or population comprising a plurality of precursor CAR cells and/or plurality of myeloid-derived CAR cells is maintained in said myeloid media until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population is CDl lb+CD13+CD15+.
In some instances, the precursor CAR cell(s) and/or population comprising a plurality of precursor CAR cells and/or plurality of myeloid-derived CAR cells is maintained in said myeloid media until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population is CDl lb+CD13+CD14+CD15+.
In some instances, the precursor CAR cell(s) and/or population comprising a plurality of precursor CAR cells and/or plurality of myeloid-derived CAR cells is maintained in said myeloid media until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population is CD1 lb+CD14+.
In some instances, the precursor CAR cell(s) and/or population comprising a plurality of precursor CAR cells and/or plurality of myeloid-derived CAR cells is maintained in said myeloid
media until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population is CD1 lb+CD15+.
In some instances, the precursor CAR cell(s) and/or population comprising a plurality of precursor CAR cells and/or plurality of myeloid-derived CAR cells is maintained in said myeloid media until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population is CD1 lb+CD14+CD15+.
In some instances, the differentiation step is maintained until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population comprising a plurality of myeloid-derived CAR cells is CD14+. In some instances, the differentiation step is maintained until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population comprising a plurality of genetically modified precursor CAR cells and/or plurality of genetically modified myeloid-derived CAR cells is CD14+. In some instances, the precursor CAR cell(s) and/or population comprising a plurality of precursor CAR cells is maintained in said myeloid media until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population is CD14+. In some instances, the precursor CAR cell(s) and/or population comprising a plurality of precursor CAR cells and/or plurality of myeloid- derived CAR cells is maintained in said myeloid media until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population is CD14+. In some instances, the population comprising a plurality of myeloid-derived CAR cells is maintained in said myeloid media until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population is CD14+.
In some instances, the precursor CAR cell(s) and/or population comprising a plurality of precursor CAR cells and/or plurality of myeloid-derived CAR cells is maintained in said myeloid media until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population is CD14+CD13+.
In some instances, the precursor CAR cell(s) and/or population comprising a plurality of precursor CAR cells and/or plurality of myeloid-derived CAR cells is maintained in said myeloid media until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population is CD14+CD15+.
In some instances, the precursor CAR cell(s) and/or population comprising a plurality of precursor CAR cells and/or plurality of myeloid-derived CAR cells is maintained in said myeloid media until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population is CD14+CD13+CD15+.
In some instances, the differentiation step is maintained until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population comprising a plurality of myeloid-derived CAR cells is CD13+. In some instances, the differentiation step is maintained until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population comprising a plurality of genetically modified precursor CAR cells and/or plurality of genetically modified myeloid-derived CAR cells is CD13+. In some instances, the precursor CAR cell(s) and/or population comprising a plurality of precursor CAR cells is maintained in said myeloid media until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population is CD13+. In some instances, the precursor CAR cell(s) and/or population comprising a plurality of precursor CAR cells and/or plurality of myeloid-derived CAR cells is maintained in said myeloid media until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population is CD13+. In some instances, the population comprising a plurality of myeloid-derived CAR cells is maintained in said myeloid media until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population is CD13+. In some instances, the precursor CAR cell(s) and/or population comprising a plurality of precursor CAR cells and/or plurality of myeloid-derived CAR cells is maintained in said myeloid media until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population is CD 13+CD 15+
In some instances, the differentiation step is maintained until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population comprising a plurality of myeloid-derived CAR cells is CD15+. In some instances, the differentiation step is maintained until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population comprising a plurality of genetically modified precursor CAR cells and/or plurality of genetically modified myeloid-derived CAR cells is CD15+. In some instances, the precursor CAR cell(s) and/or population comprising a plurality of precursor CAR cells is maintained in said myeloid media until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population is CD15+. In some instances, the precursor CAR cell(s) and/or population comprising a plurality of precursor CAR cells and/or plurality of myeloid- derived CAR cells is maintained in said myeloid media until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population is CD15+. In some instances, the population comprising a plurality of myeloid-derived CAR cells is maintained in said myeloid media until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the cell population is CD15+.
In some instances, the differentiation step is maintained until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the population comprising a plurality of genetically modified precursor CAR cells or the population comprising a plurality of genetically modified myeloid-derived CAR cells is CD13+CD15+.
In some instances, the ratio of differentiated cells to undifferentiated cells within the population increases during and after the differentiation step of the methods disclosed herein. For instance, during the differentiation step, the ratio of myeloid-derived CAR cells to precursor CAR cells with a population can change. In such instances, the ratio of myeloid-derived CAR cells to precursor CAR cells is expected to increase. In some embodiments, after the differentiation step, the number of myeloid-derived CAR cells compared to CD34+ CAR cells within the population increases. In some embodiments, after the differentiation step, the ratio of myeloid-derived CAR cells to CD34+ CAR cells within the population increases.
Table 3. Myeloid Expansion and Differentiation from Hematopoietic Stem Cells
Differentiation of genetically modified precursor CAR cells can increase the amount of CD1 lb+ cells within a plurality of genetically modified precursor CAR cells or a plurality of genetically modified myeloid-derived cells. Differentiation of genetically modified precursor CAR cells can increase the amount of CD1 lb+ cells within a plurality of genetically modified precursor CAR cells or a plurality of genetically modified myeloid-derived cells by about 10 fold, 20 fold, 30 fold, 40 fold, 50 fold, 60 fold, 70 fold, 80 fold, 90 fold, 100 fold, 200 fold, 300 fold, 400 fold, 500 fold, 600 fold, 700 fold, 800 fold, 900 fold, 1000 fold, 2000 fold, 3000 fold, 4000 fold, 5000 fold, 6000 fold, 7000 fold, 8000 fold, 9000 fold, 10,000 fold, 100,000 fold, 1,000,000 fold, 10,000,000 fold, or greater.
Differentiation of genetically modified precursor CAR cells can increase the amount of CD 14+ cells within a plurality of genetically modified precursor CAR cells or a plurality of genetically modified myeloid-derived cells. Differentiation of genetically modified precursor CAR cells can increase the amount of CD14+ cells within a plurality of genetically modified precursor CAR cells or a plurality of genetically modified myeloid-derived cells by about 10 fold, 20 fold, 30 fold, 40 fold, 50 fold, 60 fold, 70 fold, 80 fold, 90 fold, 100 fold, 200 fold, 300 fold, 400 fold, 500 fold, 600 fold, 700 fold, 800 fold, 900 fold, 1000 fold, 2000 fold, 3000 fold, 4000 fold, 5000 fold, 6000 fold, 7000 fold, 8000 fold, 9000 fold, 10,000 fold, 100,000 fold, 1,000,000 fold, 10,000,000 fold, or greater.
The amount of cell surface marker expression within a plurality of genetically modified precursor CAR cells or plurality of plurality of genetically modified myeloid-derived CAR cells can be used to determine how long to maintain the differentiating step of the method of manufacturing a genetically modified myeloid-derived CAR cells disclosed herein. In some instances, the differentiating step is maintained until a certain ratio of the population of genetically modified precursor cells or genetically modified myeloid-derived cells expresses a specific marker. In some instances, the differentiating step is maintained until a certain ratio of the population of genetically modified precursor cells or genetically modified myeloid-derived cells expresses at least a specific marker. In some instances, the differentiating step is maintained until a certain ratio of the population
of genetically modified precursor cells or genetically modified myeloid-derived cells expresses at least two specific markers. In some instances, the differentiating step is maintained until a certain ratio of the population of genetically modified precursor cells or genetically modified myeloid-derived cells expresses at least three specific markers. In some instances, the differentiating step is maintained until a certain ratio of the population of genetically modified precursor cells or genetically modified myeloid-derived cells expresses at least more than three different specific markers.
In some instances, the differentiating step is maintained until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the population comprising a plurality of genetically modified precursor CAR cells or the population comprising a plurality of genetically modified myeloid-derived CAR cells is CD1 lb+.
In some instances, the differentiating step is maintained until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the population comprising a plurality of genetically modified precursor CAR cells or the population comprising a plurality of genetically modified myeloid-derived CAR cells is CD14+.
In some instances, the differentiating step is maintained until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the population comprising a plurality of genetically modified precursor CAR cells or the population comprising a plurality of genetically modified myeloid-derived CAR cells is CD1 lb+CD14+.
In some instances, the expanding step is maintained until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the population comprising a plurality of genetically modified precursor CAR cells or the population comprising a plurality of genetically modified myeloid-derived CAR cells is CD13+.
In some instances, the expanding step is maintained until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the population comprising a plurality of genetically modified precursor CAR cells or the population comprising a plurality of genetically modified myeloid-derived CAR cells is CD15+.
In some instances, the expanding step is maintained until at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of the population comprising a plurality of
genetically modified precursor CAR cells or the population comprising a plurality of genetically modified myeloid-derived CAR cells is CD13+CD15+.
In some instances, the ratio of differentiated cells to undifferentiated cells within the population increases during and after differentiation. Differentiation of genetically modified precursor CAR cells can increase the amount of CD1 lb+ cells within a plurality of genetically modified precursor CAR cells or a plurality of genetically modified myeloid-derived cells.
Following differentiation, the cells can be incubated in cell medium in a culture apparatus for a period of time or until the cells reach confluency or high cell density for optimal passage before passing the cells to another culture apparatus. The culturing apparatus can be any culture apparatus commonly used for culturing cells in vitro. In some embodiments, the level of confluence is 70% or greater before passing the cells to another culture apparatus. In some embodiments, the level of confluence is 90% or greater. A period of time can be any time suitable for the culture of cells in vitro. The culture medium may be replaced during the culture of the cells at any time. In some embodiments, the culture medium is replaced about every 2 to 3 days. The cells are then harvested from the culture apparatus whereupon the cells can be used immediately or stored for use at a later time.
The differentiating step can be very short, for example less than 24 hours such as 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, or 23 hours. The differentiating step can be longer, for example 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, or more days. In some embodiments, the cells may be differentiated for several hours (about 3 hours) to about 14 days or any hourly integer value in between. In some embodiments, the genetically modified precursor CAR cell(s) is differentiated for at least 1, at least 2, at least 3, at least 4, or at least 5 days. In some embodiments, the genetically modified precursor CAR cell(s) is differentiated for 1 day, for 2 days, for 3 days, for 4 days, or for 5 days.
Conditions appropriate for differentiation include an appropriate media
(e.g., macrophage complete medium, DMEM/F12, DMEM/F 12-10 (Invitrogen)) that may contain factors necessary for proliferation and viability, including serum (e.g., fetal bovine or human serum), L-glutamine, insulin, M-CSF, GM-CSF, IL-10, IL-12, IL-15, TGF-beta, and TNFa, or any other additives for the growth of cells known to the skilled artisan. Other additives for the growth of cells include, but are not limited to, surfactant, plasmanate, and reducing agents such as N-acetyl-cysteine and 2-mercaptoethanol. In some embodiments, culture media can include Stemline II, RPMI 1640, AEVI-V, DMEM, MEM, a-MEM, F-12, X-Vivo 15, X-Vivo 20, and Optimizer, with added amino acids, sodium pyruvate, and vitamins, either serum-free or supplemented with an appropriate amount of serum (or plasma) or a defined set of hormones, and/or an amount of cytokine(s) sufficient for the growth, expansion and/or differentiation of the cells. Antibiotics, e.g., penicillin and streptomycin, are
included only in experimental cultures, not in cultures of cells that are to be infused into a subject. The target cells are maintained under conditions necessary to support growth, for example, an appropriate temperature (e.g., 37°C) and atmosphere (e.g., air plus 5% CO2).
The myeloid medium used to culture the cells may include an agent that can activate the cells. For example, an agent that is known in the art to activate a macrophage, monocyte, dendritic cell, and/or a neutrophil is included in the medium.
The step of differentiating the genetically modified precursor CAR cells can be performed until a number or ratio of precursor CAR cells have been differentiated into myeloid-derived CAR cells. To determine the number or ratio of precursor CAR cells, cell surface markers can be examined; cell surface marker expression within a population can be examined over a series of hours or days to monitor the increase in desired surface marker. The population comprising a plurality of precursor CAR cells can be examined for decreases in expression of CD34+, a common stem cell surface marker. Conversely, CD1 lb+ and/or CD14+ can be utilized to monitor myeloid lineage cells, including monocytes and macrophages. In some instances, the precursor CAR cell is expanded and differentiated until the population comprising the plurality of genetically modified precursor myeloid-derived CAR cells comprises at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% CD1 lb+ or CD14+ or CD1 lb+CD14+ cells. In some instances, the population of genetically modified precursor myeloid-derived CAR cells comprises at least 20%, at least 40%, at least 60%, or at least 80% CD 14+ after 3 days of differentiation.Jn some instances, at least 1%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of said plurality of genetically modified myeloid-derived CAR cells expressing said CAR are CD1 lb+ or CD14+ or Cl lb+CD14+.
Myeloid Media
Disclosed herein is a myeloid media comprising a myeloid cytokine mixture useful for differentiating genetically modified precursor CAR cell(s). The myeloid media and myeloid cytokine mixture can be used in the methods disclosed herein to generate a myeloid-derived cell from a hematopoietic stem cell with increased efficiency and efficacy over known methods. For instance, in some embodiments, a precursor cell is contacted with a precursor media to generate a cell population comprising a plurality of precursor cells, prior to contacting said precursor cell(s) with myeloid media. The precursor cell can be an HSC Following contact with a precursor media for a suitable time period, it can be expected that a precursor cell is expanded (i.e., the number of precursor cells increases) and differentiated (e.g., into a myeloid-derived cell) in said precursor
media to generate a population comprising a plurality of myeloid-derived cells. In some embodiments, the population comprises a plurality of myeloid-derived cells and a plurality of precursor cells. The precursor cells and myeloid-derived cells can be present at any ratio within the population after being maintained in the precursor media.
The myeloid media and myeloid cytokine mixture can be used in the methods disclosed herein to generate a myeloid-derived CAR cell from a hematopoietic stem cell with increased efficiency and efficacy over known methods. For instance, in some embodiments, a precursor CAR cell is contacted with a precursor media to generate a cell population comprising a plurality of precursor CAR cells, prior to contacting said precursor CAR cell(s) with myeloid media. As described herein, the disclosed precursor media are particularly effective at both expanding a precursor cell to generate a cell population, as well as promoting the differentiation of a precursor cell, specifically into a myeloid cell. As such, following contact with a precursor media for a suitable time period, it can be expected that a precursor CAR cell is expanded (i.e., the number of precursor CAR cells increases) and differentiated (e.g., into a myeloid-derived CAR cell) in said precursor media to generate a population comprising a plurality of myeloid-derived CAR cells. In some embodiments, the population comprises a plurality of myeloid-derived CAR cells and a plurality of precursor CAR cells. The precursor CAR cells and myeloid-derived CAR cells can be present at any ratio within the population after being maintained in the precursor media.
For the purposes of the present disclosure, it may be particularly useful to generate a population of cells comprising a higher ratio of myeloid-derived CAR cells compared to precursor CAR cells. The myeloid media disclosed herein is particularly effective and efficient, when compared to known differentiation mediums, at increasing the ratio of myeloid-derived CAR cells to precursor CAR cells within a cell population. For instance, in some embodiments, when a cell population is generated by maintaining a precursor CAR cell in a precursor media, wherein said cell population comprises a plurality of precursor CAR cells and a plurality of myeloid-derived CAR cells, said cell population is subsequently contacted with a myeloid media disclosed herein to increase the number of myeloid-derived CAR cells within the cell population. In some embodiments, when the number of myeloid-derived CAR cells increases within a population, the total number of cells within the population is relatively unchanged. In some embodiments, when the number of myeloid-derived CAR cells increases within a population, the total number of cells within the population increases. In some embodiments, when the number of myeloid-derived CAR cells increases within a population, the total number of cells within the population decreases.
In some embodiments, when a cell population is generated by maintaining a precursor CAR cell in a precursor media, wherein said cell population comprises a plurality of precursor CAR cells
and a plurality of myeloid-derived CAR cells, said cell population is subsequently contacted with a myeloid media disclosed herein to decrease the number of precursor CAR cells within the cell population. In some embodiments, when the number of precursor CAR cells decreases within a population, the total number of cells within the population is relatively unchanged. In some embodiments, when the number of precursor CAR cells decreases within a population, the total number of cells within the population increases. In some embodiments, when the number of precursor CAR cells decreases within a population, the total number of cells within the population decreases.
In some embodiments, when a cell population is generated by maintaining a precursor CAR cell in a precursor media, wherein said cell population comprises a plurality of precursor CAR cells and a plurality of myeloid-derived CAR cells, said cell population is subsequently contacted with a myeloid media disclosed herein to increase the number of myeloid-derived CAR cells compared to precursor CAR cells within the population. In some embodiments, when the number of myeloid- derived CAR cells compared to precursor CAR cells within the population, the total number of cells within the population is relatively unchanged. In some embodiments, when the number of myeloid-derived CAR cells compared to precursor CAR cells within the population, the total number of cells within the population increases. In some embodiments, when the number of myeloid-derived CAR cells compared to precursor CAR cells within the population, the total number of cells within the population decreases.
In some embodiments, when a cell population is generated by maintaining a precursor CAR cell in a precursor media, wherein said cell population comprises a plurality of precursor CAR cells and a plurality of myeloid-derived CAR cells, said cell population is subsequently contacted with a myeloid media disclosed herein to increase the ratio of myeloid-derived CAR cells to precursor CAR cells within the population. In some embodiments, when the ratio of myeloid-derived CAR cells to precursor CAR cells within the population is increased, the total number of cells within the population is relatively unchanged. In some embodiments, when the ratio of myeloid-derived CAR cells to precursor CAR cells within the population is increased, the total number of cells within the population increases. In some embodiments, when the ratio of myeloid-derived CAR cells to precursor CAR cells within the population is increased, the total number of cells within the population decreases.
In some instances, the genetically modified precursor CAR cells are genetically modified stem cells expressing a CAR. In some instances, the stem cells expressing a CAR are hematopoietic stem cells (HSCs). The HSCs can be CD34+ cells. Differentiating the HSCs modified to express a CAR promotes their differentiation in a genetically modified myeloid-derived CAR cell(s). The
myeloid media disclosed herein is particularly effective at promoting the differentiation of HSCs to myeloid-derived cells. In some instances, the myeloid media is particularly effective at differentiation a CD34+ cell into a CD14+ cell. In some instances, the myeloid media is particularly effective at differentiation a CD34+ cell into a CD1 lb+ cell. In some instances, the myeloid media is particularly effective at differentiation a CD34+ cell into a CD14+CD1 lb+ cell. Without wishing to be bound by theory, the cytokine mixture within the myeloid media (i.e., the myeloid cytokine mixture) is thought to contribute to the effective differentiation seen with the myeloid media disclosed herein. Also disclosed herein are myeloid cytokine mixtures that are particularly effective at promoting differentiation of stem cells into myeloid-derived cells. In some instances, myeloid-derived cells are CD14+ cells. The myeloid media comprising the myeloid cytokine mixtures disclosed herein are particularly effective at promoting differentiation of HSCs into CD 14+ cells. The myeloid media comprising the myeloid cytokine mixtures disclosed herein are particularly effective at promoting differentiation of HSCs into CD1 lb+ cells. The myeloid media comprising the myeloid cytokine mixtures disclosed herein are particularly effective at promoting differentiation of HSCs into any one of the following cells: CD13+ cell, a CD15+ cell, a CDl lb+CD13+ cell, a CDl lb+CD13+CD14+ cell, a CDl lb+CD13+CD15+ cell, a CD1 lb+CD13+CD14+CD15+ cell, a CDl lb+CD14+ cell, a CDl lb+CD15+ cell, a
CD1 lb+CD14+CD15+ cell, a CD14+CD13+ cell, a CD14+CD15+ cell, a CD14+CD13+CD15+ cell, and/or a CD13+CD15+ cell.
Any cytokines at any concentrations can be used in the myeloid media to support differentiation of the genetically modified precursor CAR cells. For example, the myeloid cytokine mixture can comprise Macrophage colony-stimulating factor (MCSF), Interleukin-3 (IL-3), or Interleukin-6 (IL-6), or a combination thereof. In some instances, the myeloid cytokine mixture comprises Macrophage colony-stimulating factor (MCSF), Interleukin-3 (IL-3), and Interleukin-6 (IL-6).
When the myeloid cytokine mixture comprises macrophage colony-stimulating factor (MCSF), the concentration of the MCSF in the myeloid media can be at least 50 ng/mL, at least 100 ng/mL, at least 150 ng/mL, or at least 200 ng/mL. In a preferred embodiment, the myeloid media comprises macrophage colony-stimulating factor (MCSF) at a concentration of 150 ng/mL.
When the myeloid cytokine mixture comprises Interleukin-3 (IL-3), the concentration of the IL-3 in the myeloid media can be at least 50 ng/mL, at least 100 ng/mL, at least 150 ng/mL, or at least 200 ng/mL. In a preferred embodiment, the myeloid media comprises IL-3 at a concentration of 150 ng/mL.
When the myeloid cytokine mixture comprises Interleukin-6 (IL-6), the concentration of the IL-6 in the myeloid media can be at least 50 ng/mL, at least 100 ng/mL, at least 150 ng/mL, or at least 200 ng/mL. In a preferred embodiment, the myeloid media comprises IL-6 at a concentration of 150 ng/mL.
The myeloid cytokine mixture can comprise Macrophage colony-stimulating factor (MCSF), Interleukin-3 (IL-3), and Interleukin-6 (IL-6).
When the myeloid cytokine mixture comprises MCSF, IL-3 and IL-6, the myeloid media can comprise MCSF at a concentration of 150 ng/mL, IL-3 at a concentration of 50 ng/mL and IL-6 at a concentration of 100 ng/mL. In a preferred embodiment, the myeloid media comprises MCSF at a concentration of 150 ng/mL, IL-3 at a concentration of 50 ng/mL, and IL-6 at a concentration of 100 ng/mL. In a preferred embodiment, the myeloid media comprises MCSF at a concentration of 150 ng/mL, IL-3 at a concentration of 50 ng/mL, and IL-6 at a concentration of 100 ng/mL, the myeloid media further comprises a basal growth media comprising VIVO™- 15 medium.
In some embodiments, the myeloid media comprises macrophage colony-stimulating factor (MCSF). The macrophage colony-stimulating factor (MCSF) can be present in the myeloid media at a concentration of at least 20 ng/mL, at least 30 ng/mL, at least 40 ng/mL, at least 50 ng/mL, at least 60 ng/mL, of at least 70 ng/mL, at least 80 ng/mL, at least 90 ng/mL, at least 100 ng/mL, at least 110 ng/mL, at least 120 ng/mL, at least 130 ng/mL, at least 140 ng/mL, at least 150 ng/mL, at least 160 ng/mL, at least 170 ng/mL, at least 180 ng/mL, at least 190 ng/mL, at least 200 ng/mL, at least 210 ng/mL, of at least 220 ng/mL, at least 230 ng/mL, at least 240 ng/mL, at least 250 ng/mL, or more than 250 ng/mL. In some embodiments, the macrophage colony-stimulating factor (MCSF) can be present in the myeloid media at a concentration of 20-40 ng/mL, 30-50 ng/mL, 40-60 ng/mL, 50-70 ng/mL, 60-80 ng/mL, 70-90 ng/mL, 80-100 ng/mL, 90-110 ng/mL, 100-120 ng/mL, 110-130 ng/mL, 120-140 ng/mL, 130-150 ng/mL, 140-160 ng/mL, 150-170 ng/mL, 160-180 ng/mL, 170-190 ng/mL, 180-200 ng/mL, 190-210 ng/mL, 200-220 ng/mL, 210-230 ng/mL, 220- 240 ng/mL, 230-250 ng/mL, or greater than 250 ng/mL.
In some embodiments, when the myeloid media comprises MCSF, said MCSF is present at a concentration of between about 100 ng/mL and 200 ng/mL. In some embodiments, when the myeloid media comprises MCSF, said MCSF is present at a concentration of between 100-105 ng/mL, 105-110 ng/mL, 110-115 ng/mL, 115-120 ng/mL, 120-125 ng/mL, 125-130 ng/mL, BODS ng/mL, 135-140 ng/mL, 140-145 ng/mL, 145-150 ng/mL, 150-155 ng/mL, 155-160 ng/mL, 160-165 ng/mL, 165-170 ng/mL, 170-175 ng/mL, 175-180 ng/mL, 180-185 ng/mL, 185-190 ng/mL, 190-195 ng/mL, or 195-200 ng/mL. In some embodiments, when the myeloid media
comprises MCSF, said MCSF is present at a concentration of between about 100 ng/mL and 150 ng/mL.
In some embodiments, the myeloid media comprises more than one cytokine (i.e., a myeloid cytokine mixture). When the myeloid cytokine mixture comprises MCSF at a concentration of between about 125 ng/mL and 175 ng/mL in said myeloid media, the myeloid cytokine mixture comprises MCSF, IL-3, and IL-6. When the myeloid media comprises MCSF at a concentration of between 125 ng/mL and 175 ng/mL, said myeloid media can comprise any of the following cytokines: MCSF, IL-3, and IL-6. When the myeloid media comprises MCSF at a concentration of between 125 ng/mL and 175 ng/mL, said myeloid media can comprise at least one of the following additional cytokines: IL-3 and/or IL-6. When the myeloid media comprises MCSF at a concentration of between 125 ng/mL and 175 ng/mL, said myeloid media can comprise all of the following cytokines: IL_3 and IL-6. In such instances, IL-3 can be present at a concentration between about 25 ng/mL and 75 ng/mL; and/or IL-6 can be present at a concentration between about 75 ng/mL and 125 ng/mL.
In a preferred embodiment, the myeloid media comprises MCSF at a concentration of about 150 ng/mL, IL-3 at a concentration of about 50 ng/mL, and IL-6 at a concentration of about 100 ng/mL. In a preferred embodiment, the myeloid media comprises MCSF at a concentration of 150 ng/mL, IL-3 at a concentration of 50 ng/mL, and IL-6 at a concentration of 100 ng/mL.
In some embodiments, when the myeloid media comprises macrophage colony-stimulating factor (MCSF) at a concentration of between about 100 ng/mL and 200 ng/mL, said myeloid media further comprises: a. IL-3 at a concentration of 10-30 ng/mL, 20-40 ng/mL, 30-50 ng/mL, 40-60 ng/mL, 50-70 ng/mL, 60-80 ng/mL, 70-90 ng/mL, 80-100 ng/mL, 90-110 ng/mL, 100-120 ng/mL, 110-130 ng/mL, 120-140 ng/mL, 130-150 ng/mL, 140-160 ng/mL, 150-170 ng/mL, 160-180 ng/mL, 170-190 ng/mL, 180-200 ng/mL, 190-210 ng/mL, 200-220 ng/mL, or greater than 220 ng/mL; and/or b. IL-6 at a concentration of 10-30 ng/mL, 20-40 ng/mL, 30-50 ng/mL, 40-60 ng/mL, 50-70 ng/mL, 60-80 ng/mL, 70-90 ng/mL, 80-100 ng/mL, 90-110 ng/mL, 100-120 ng/mL, 110-130 ng/mL, 120-140 ng/mL, 130-150 ng/mL, 140-160 ng/mL, 150-170 ng/mL, 160-180 ng/mL, 170-190 ng/mL, 180-200 ng/mL, 190-210 ng/mL, 200-220 ng/mL, or greater than 220 ng/mL.
In some embodiments, when the myeloid media comprises macrophage colony-stimulating factor (MCSF) at a concentration of about 140 ng/mL. In some embodiments, when the myeloid media comprises macrophage colony-stimulating factor (MCSF) at a concentration of about 140 ng/mL, said myeloid media further comprises: a. IL-3 at a concentration of 10-30 ng/mL, 20-40 ng/mL, 30-50 ng/mL, 40-60 ng/mL, 50-70 ng/mL, 60-80 ng/mL, 70-90 ng/mL, 80-100 ng/mL, 90-
110 ng/mL, 100-120 ng/mL, 110-130 ng/mL, 120-140 ng/mL, 130-150 ng/mL, 140-160 ng/mL, 150-170 ng/mL, 160-180 ng/mL, 170-190 ng/mL, 180-200 ng/mL, 190-210 ng/mL, 200-220 ng/mL, or greater than 220 ng/mL; and/or b. IL-6 at a concentration of 10-30 ng/mL, 20-40 ng/mL, 30-50 ng/mL, 40-60 ng/mL, 50-70 ng/mL, 60-80 ng/mL, 70-90 ng/mL, 80-100 ng/mL, 90-110 ng/mL, 100-120 ng/mL, 110-130 ng/mL, 120-140 ng/mL, 130-150 ng/mL, 140-160 ng/mL, 150- 170 ng/mL, 160-180 ng/mL, 170-190 ng/mL, 180-200 ng/mL, 190-210 ng/mL, 200-220 ng/mL, or greater than 220 ng/mL.
In some embodiments, when the myeloid media comprises macrophage colony-stimulating factor (MCSF) at a concentration of about 145 ng/mL. In some embodiments, when the myeloid media comprises macrophage colony-stimulating factor (MCSF) at a concentration of about 145 ng/mL, said precursor media further comprises: a. IL-3 at a concentration of 10-30 ng/mL, 20-40 ng/mL, 30-50 ng/mL, 40-60 ng/mL, 50-70 ng/mL, 60-80 ng/mL, 70-90 ng/mL, 80-100 ng/mL, 90- 110 ng/mL, 100-120 ng/mL, 110-130 ng/mL, 120-140 ng/mL, 130-150 ng/mL, 140-160 ng/mL, 150-170 ng/mL, 160-180 ng/mL, 170-190 ng/mL, 180-200 ng/mL, 190-210 ng/mL, 200-220 ng/mL, or greater than 220 ng/mL; and/or b. IL-6 at a concentration of 10-30 ng/mL, 20-40 ng/mL, 30-50 ng/mL, 40-60 ng/mL, 50-70 ng/mL, 60-80 ng/mL, 70-90 ng/mL, 80-100 ng/mL, 90-110 ng/mL, 100-120 ng/mL, 110-130 ng/mL, 120-140 ng/mL, 130-150 ng/mL, 140-160 ng/mL, 150- 170 ng/mL, 160-180 ng/mL, 170-190 ng/mL, 180-200 ng/mL, 190-210 ng/mL, 200-220 ng/mL, or greater than 220 ng/mL.
In some embodiments, when the myeloid media comprises macrophage colony-stimulating factor (MCSF) at a concentration of about 150 ng/mL. In some embodiments, when the myeloid media comprises macrophage colony-stimulating factor (MCSF) at a concentration of about 150 ng/mL, said precursor media further comprises: a. IL-3 at a concentration of 10-30 ng/mL, 20-40 ng/mL, 30-50 ng/mL, 40-60 ng/mL, 50-70 ng/mL, 60-80 ng/mL, 70-90 ng/mL, 80-100 ng/mL, 90- 110 ng/mL, 100-120 ng/mL, 110-130 ng/mL, 120-140 ng/mL, 130-150 ng/mL, 140-160 ng/mL, 150-170 ng/mL, 160-180 ng/mL, 170-190 ng/mL, 180-200 ng/mL, 190-210 ng/mL, 200-220 ng/mL, or greater than 220 ng/mL; and/or b. IL-6 at a concentration of 10-30 ng/mL, 20-40 ng/mL, 30-50 ng/mL, 40-60 ng/mL, 50-70 ng/mL, 60-80 ng/mL, 70-90 ng/mL, 80-100 ng/mL, 90-110 ng/mL, 100-120 ng/mL, 110-130 ng/mL, 120-140 ng/mL, 130-150 ng/mL, 140-160 ng/mL, 150- 170 ng/mL, 160-180 ng/mL, 170-190 ng/mL, 180-200 ng/mL, 190-210 ng/mL, 200-220 ng/mL, or greater than 220 ng/mL.
In some embodiments, when the myeloid media comprises macrophage colony-stimulating factor (MCSF) at a concentration of about 155 ng/mL. In some embodiments, when the myeloid media comprises macrophage colony-stimulating factor (MCSF) at a concentration of about 155
ng/mL, said precursor media further comprises: a. IL-3 at a concentration of 10-30 ng/mL, 20-40 ng/mL, 30-50 ng/mL, 40-60 ng/mL, 50-70 ng/mL, 60-80 ng/mL, 70-90 ng/mL, 80-100 ng/mL, 90- 110 ng/mL, 100-120 ng/mL, 110-130 ng/mL, 120-140 ng/mL, 130-150 ng/mL, 140-160 ng/mL, 150-170 ng/mL, 160-180 ng/mL, 170-190 ng/mL, 180-200 ng/mL, 190-210 ng/mL, 200-220 ng/mL, or greater than 220 ng/mL; and/or b. IL-6 at a concentration of 10-30 ng/mL, 20-40 ng/mL, 30-50 ng/mL, 40-60 ng/mL, 50-70 ng/mL, 60-80 ng/mL, 70-90 ng/mL, 80-100 ng/mL, 90-110 ng/mL, 100-120 ng/mL, 110-130 ng/mL, 120-140 ng/mL, 130-150 ng/mL, 140-160 ng/mL, 150- 170 ng/mL, 160-180 ng/mL, 170-190 ng/mL, 180-200 ng/mL, 190-210 ng/mL, 200-220 ng/mL, or greater than 220 ng/mL.
In some embodiments, when the myeloid media comprises macrophage colony-stimulating factor (MCSF) at a concentration of about 160 ng/mL. In some embodiments, when the myeloid media comprises macrophage colony-stimulating factor (MCSF) at a concentration of about 160 ng/mL, said precursor media further comprises: a. IL-3 at a concentration of 10-30 ng/mL, 20-40 ng/mL, 30-50 ng/mL, 40-60 ng/mL, 50-70 ng/mL, 60-80 ng/mL, 70-90 ng/mL, 80-100 ng/mL, 90- 110 ng/mL, 100-120 ng/mL, 110-130 ng/mL, 120-140 ng/mL, 130-150 ng/mL, 140-160 ng/mL, 150-170 ng/mL, 160-180 ng/mL, 170-190 ng/mL, 180-200 ng/mL, 190-210 ng/mL, 200-220 ng/mL, or greater than 220 ng/mL; and/or b. IL-6 at a concentration of 10-30 ng/mL, 20-40 ng/mL, 30-50 ng/mL, 40-60 ng/mL, 50-70 ng/mL, 60-80 ng/mL, 70-90 ng/mL, 80-100 ng/mL, 90-110 ng/mL, 100-120 ng/mL, 110-130 ng/mL, 120-140 ng/mL, 130-150 ng/mL, 140-160 ng/mL, 150- 170 ng/mL, 160-180 ng/mL, 170-190 ng/mL, 180-200 ng/mL, 190-210 ng/mL, 200-220 ng/mL, or greater than 220 ng/mL.
In a preferred embodiment, the myeloid media comprises MCSF at a concentration of 150 ng/mL. When the myeloid media comprises MCSF at a concentration of about 150ng/mL, said myeloid media further comprises: a. IL-3 at a concentration between about 25 ng/mL and 75 ng/mL; and/or b. IL-6 at a concentration between about 75 ng/mL and 125 ng/mL.
In another preferred embodiment, the myeloid media comprises MCSF at a concentration of about 150ng/mL, and said myeloid media further comprises: a. IL-3 at a concentration between about 20 ng/mL, about 25 ng/mL, about 30 ng/mL, about 35 ng/mL, about 40 ng/mL, about 45 ng/mL, about 50 ng/mL, about 555 ng/mL, about 60 ng/mL, about 65 ng/mL, about 70 ng/mL, or about 75 ng/mL; and/or b. IL-6 at a concentration between about 70 ng/mL, about 75 ng/mL, about 80 ng/mL, about 85 ng/mL, about 90 ng/mL, about 95 ng/mL, about 100 ng/mL, about 105 ng/mL, about 110 ng/mL, about 115 ng/mL, about 120 ng/mL, or about 125 ng/mL.
In any of the embodiments above, the myeloid media can comprise stem cells factor (SCF), Flt3 ligand, thrombopoietin (TPO), Granulocyte colony-stimulating factor (G-CSF), and
Granulocyte-macrophage colony-stimulating factor (GM-CSF). The stem cells factor (SCF), Flt3 ligand, thrombopoietin (TPO), Granulocyte colony-stimulating factor (G-CSF), and Granulocytemacrophage colony-stimulating factor (GM-CSF) can be present within the myeloid media at any useful concentration. A useful concentration is defined as any concentration of an individual cytokine within the myeloid media that promotes the differentiation of precursor CAR cells or myeloid-derived CAR cells as described herein.
Stem cells factor (SCF) can be within the myeloid mixture at a concentration of at most 50 ng/mL, at most 40 ng/mL, at most 30 ng/mL, at most 20 ng/mL, at most 10 ng/mL, at most 5 ng/mL, at most 4 ng/mL, at most 3 ng/mL, at most 2 ng/mL, at most 1 ng/mL, at most 0.5 ng/mL, at most 0.1 ng/mL, at most 0.01 ng/mL, at most 0.0001 ng/mL, or less than 0.0001 ng/L. In some instances, the myeloid media comprises stem cells factor (SCF) at a concentration of below 50 ng/mL. In some instances, the myeloid media comprises stem cells factor (SCF) at a concentration of below 40 ng/mL. In some instances, the myeloid media comprises stem cells factor (SCF) at a concentration of below 30 ng/mL. In some instances, the myeloid media comprises stem cells factor (SCF) at a concentration of below 20 ng/mL. In some instances, the myeloid media comprises stem cells factor (SCF) at a concentration of below 10 ng/mL. In some instances, the myeloid media comprises stem cells factor (SCF) at a concentration of below 5 ng/mL. In a preferred embodiment, the myeloid mixture does not comprise stem cells factor (SCF).
Flt3 ligand can be within the myeloid mixture at a concentration of at most 50 ng/mL, at most 40 ng/mL, at most 30 ng/mL, at most 20 ng/mL, at most 10 ng/mL, at most 5 ng/mL, at most 4 ng/mL, at most 3 ng/mL, at most 2 ng/mL, at most 1 ng/mL, at most 0.5 ng/mL, at most 0.1 ng/mL, at most 0.01 ng/mL, at most 0.0001 ng/mL, or less than 0.0001 ng/L. In some instances, the myeloid media comprises Flt3 ligand at a concentration of below 50 ng/mL. In some instances, the myeloid media comprises Flt3 ligand at a concentration of below 40 ng/mL. In some instances, the myeloid media comprises Flt3 ligand at a concentration of below 30 ng/mL. In some instances, the myeloid media comprises Flt3 ligand at a concentration of below 20 ng/mL. In some instances, the myeloid media comprises Flt3 ligand at a concentration of below 10 ng/mL. In some instances, the myeloid media comprises Flt3 ligand at a concentration of below 5 ng/mL. In a preferred embodiment, the myeloid mixture does not comprise Flt3 ligand.
Thrombopoietin (TPO) can be within the myeloid mixture at a concentration of at most 50 ng/mL, at most 40 ng/mL, at most 30 ng/mL, at most 20 ng/mL, at most 10 ng/mL, at most 5 ng/mL, at most 4 ng/mL, at most 3 ng/mL, at most 2 ng/mL, at most 1 ng/mL, at most 0.5 ng/mL, at most 0.1 ng/mL, at most 0.01 ng/mL, at most 0.0001 ng/mL, or less than 0.0001 ng/L. In some instances, the myeloid media comprises TPO at a concentration of below 50 ng/mL. In some
instances, the myeloid media comprises TPO at a concentration of below 40 ng/mL. In some instances, the myeloid media comprises TPO at a concentration of below 30 ng/mL. In some instances, the myeloid media comprises TPO at a concentration of below 20 ng/mL. In some instances, the myeloid media comprises TPO at a concentration of below 10 ng/mL. In some instances, the myeloid media comprises TPO at a concentration of below 5 ng/mL. In a preferred embodiment, the myeloid mixture does not comprise TPO.
Granulocyte colony-stimulating factor (G-CSF) can be within the myeloid mixture at a concentration of at most 50 ng/mL, at most 40 ng/mL, at most 30 ng/mL, at most 20 ng/mL, at most 10 ng/mL, at most 5 ng/mL, at most 4 ng/mL, at most 3 ng/mL, at most 2 ng/mL, at most 1 ng/mL, at most 0.5 ng/mL, at most 0.1 ng/mL, at most 0.01 ng/mL, at most 0.0001 ng/mL, or less than 0.0001 ng/L. In some instances, the myeloid media comprises G-CSF at a concentration of below 50 ng/mL. In some instances, the myeloid media comprises G-CSF at a concentration of below 40 ng/mL. In some instances, the myeloid media comprises G-CSF at a concentration of below 30 ng/mL. In some instances, the myeloid media comprises G-CSF at a concentration of below 20 ng/mL. In some instances, the myeloid media comprises G-CSF at a concentration of below 10 ng/mL. In some instances, the myeloid media comprises G-CSF at a concentration of below 5 ng/mL. In a preferred embodiment, the myeloid mixture does not comprise any G-CSF.
Granulocyte-macrophage colony-stimulating factor (GM-CSF) can be within the myeloid mixture at a concentration of at most 50 ng/mL, at most 40 ng/mL, at most 30 ng/mL, at most 20 ng/mL, at most 10 ng/mL, at most 5 ng/mL, at most 4 ng/mL, at most 3 ng/mL, at most 2 ng/mL, at most 1 ng/mL, at most 0.5 ng/mL, at most 0.1 ng/mL, at most 0.01 ng/mL, at most 0.0001 ng/mL, or less than 0.0001 ng/L. In some instances, the myeloid media comprises GM-CSF at a concentration of below 50 ng/mL. In some instances, the myeloid media comprises GM-CSF at a concentration of below 40 ng/mL. In some instances, the myeloid media comprises GM-CSF at a concentration of below 30 ng/mL. In some instances, the myeloid media comprises GM-CSF at a concentration of below 20 ng/mL. In some instances, the myeloid media comprises GM-CSF at a concentration of below 10 ng/mL. In some instances, the myeloid media comprises GM-CSF at a concentration of below 5 ng/mL. In a preferred embodiment, the myeloid mixture does not comprise any GM-CSF.
In a preferred embodiment, the myeloid media comprises MCSF at a concentration of between 125 ng/mL and 175 ng/mL, and said myeloid media further comprises: a. IL-3 at a concentration between about 20 ng/mL, about 25 ng/mL, about 30 ng/mL, about 35 ng/mL, about 40 ng/mL, about 45 ng/mL, about 50 ng/mL, about 555 ng/mL, about 60 ng/mL, about 65 ng/mL, about 70 ng/mL, or about 75 ng/mL; b. IL-6 at a concentration between about 70 ng/mL, about 75
ng/mL, about 80 ng/mL, about 85 ng/mL, about 90 ng/mL, about 95 ng/mL, about 100 ng/mL, about 105 ng/mL, about 110 ng/mL, about 115 ng/mL, about 120 ng/mL, or about 125 ng/mL; c. stem cells factor (SCF) at a concentration of at most 50 ng/mL, at most 40 ng/mL, at most 30 ng/mL, at most 20 ng/mL, at most 10 ng/mL, at most 5 ng/mL, at most 4 ng/mL, at most 3 ng/mL, at most 2 ng/mL, at most 1 ng/mL, at most 0.5 ng/mL, at most 0.1 ng/mL, at most 0.01 ng/mL, at most 0.0001 ng/mL, or less than 0.0001 ng/L; d. Flt3 ligand at a concentration of at most 50 ng/mL, at most 40 ng/mL, at most 30 ng/mL, at most 20 ng/mL, at most 10 ng/mL, at most 5 ng/mL, at most 4 ng/mL, at most 3 ng/mL, at most 2 ng/mL, at most 1 ng/mL, at most 0.5 ng/mL, at most 0.1 ng/mL, at most 0.01 ng/mL, at most 0.0001 ng/mL, or less than 0.0001 ng/L; e. thrombopoietin (TPO) at a concentration of at most 50 ng/mL, at most 40 ng/mL, at most 30 ng/mL, at most 20 ng/mL, at most 10 ng/mL, at most 5 ng/mL, at most 4 ng/mL, at most 3 ng/mL, at most 2 ng/mL, at most 1 ng/mL, at most 0.5 ng/mL, at most 0.1 ng/mL, at most 0.01 ng/mL, at most 0.0001 ng/mL, or less than 0.0001 ng/L; f. granulocyte colony-stimulating factor (G-CSF) at a concentration of at most 50 ng/mL, at most 40 ng/mL, at most 30 ng/mL, at most 20 ng/mL, at most 10 ng/mL, at most 5 ng/mL, at most 4 ng/mL, at most 3 ng/mL, at most 2 ng/mL, at most 1 ng/mL, at most 0.5 ng/mL, at most 0.1 ng/mL, at most 0.01 ng/mL, at most 0.0001 ng/mL, or less than 0.0001 ng/L; and/or g. granulocyte-macrophage colony-stimulating factor (GM-CSF) at a concentration of at most 50 ng/mL, at most 40 ng/mL, at most 30 ng/mL, at most 20 ng/mL, at most 10 ng/mL, at most 5 ng/mL, at most 4 ng/mL, at most 3 ng/mL, at most 2 ng/mL, at most 1 ng/mL, at most 0.5 ng/mL, at most 0.1 ng/mL, at most 0.01 ng/mL, at most 0.0001 ng/mL, or less than 0.0001 ng/L.
In a preferred embodiment, the myeloid media comprises MCSF at a concentration of about 150ng/mL, and said myeloid media further comprises: a. IL-3 at a concentration between about 20 ng/mL, about 25 ng/mL, about 30 ng/mL, about 35 ng/mL, about 40 ng/mL, about 45 ng/mL, about 50 ng/mL, about 555 ng/mL, about 60 ng/mL, about 65 ng/mL, about 70 ng/mL, or about 75 ng/mL; b. IL-6 at a concentration between about 70 ng/mL, about 75 ng/mL, about 80 ng/mL, about 85 ng/mL, about 90 ng/mL, about 95 ng/mL, about 100 ng/mL, about 105 ng/mL, about 110 ng/mL, about 115 ng/mL, about 120 ng/mL, or about 125 ng/mL; c. stem cells factor (SCF) at a concentration of at most 50 ng/mL, at most 40 ng/mL, at most 30 ng/mL, at most 20 ng/mL, at most 10 ng/mL, at most 5 ng/mL, at most 4 ng/mL, at most 3 ng/mL, at most 2 ng/mL, at most 1 ng/mL, at most 0.5 ng/mL, at most 0.1 ng/mL, at most 0.01 ng/mL, at most 0.0001 ng/mL, or less than 0.0001 ng/L; d. Flt3 ligand at a concentration of at most 50 ng/mL, at most 40 ng/mL, at most 30 ng/mL, at most 20 ng/mL, at most 10 ng/mL, at most 5 ng/mL, at most 4 ng/mL, at most 3 ng/mL, at most 2 ng/mL, at most 1 ng/mL, at most 0.5 ng/mL, at most 0.1 ng/mL, at most 0.01 ng/mL, at most 0.0001 ng/mL, or less than 0.0001 ng/L; e. thrombopoietin (TPO) at a concentration
of at most 50 ng/mL, at most 40 ng/mL, at most 30 ng/mL, at most 20 ng/mL, at most 10 ng/mL, at most 5 ng/mL, at most 4 ng/mL, at most 3 ng/mL, at most 2 ng/mL, at most 1 ng/mL, at most 0.5 ng/mL, at most 0.1 ng/mL, at most 0.01 ng/mL, at most 0.0001 ng/mL, or less than 0.0001 ng/L; f. granulocyte colony-stimulating factor (G-CSF) at a concentration of at most 50 ng/mL, at most 40 ng/mL, at most 30 ng/mL, at most 20 ng/mL, at most 10 ng/mL, at most 5 ng/mL, at most 4 ng/mL, at most 3 ng/mL, at most 2 ng/mL, at most 1 ng/mL, at most 0.5 ng/mL, at most 0.1 ng/mL, at most 0.01 ng/mL, at most 0.0001 ng/mL, or less than 0.0001 ng/L; and/or g. granulocyte-macrophage colony-stimulating factor (GM-CSF) at a concentration of at most 50 ng/mL, at most 40 ng/mL, at most 30 ng/mL, at most 20 ng/mL, at most 10 ng/mL, at most 5 ng/mL, at most 4 ng/mL, at most 3 ng/mL, at most 2 ng/mL, at most 1 ng/mL, at most 0.5 ng/mL, at most 0.1 ng/mL, at most 0.01 ng/mL, at most 0.0001 ng/mL, or less than 0.0001 ng/L.
In addition to a myeloid cytokine mixture, the myleoid media disclosed herein can comprise a basal growth medium. A cell “growth medium” or “culture medium” refers to any solid, semisolid, or liquid composition comprising components necessary to support growth and function of cells. A person having skill in the art would understand that a basal growth medium is a growth medium comprising a minimal amount of components such as amino acids, vitamins, inorganic salts, and a carbon source, such as glucose, to support cell growth and function. The basal growth medium can be any medium that supports growth, expansion and/or differentiation of precursor cells. The term “growth” when as used herein in relation to cell refers to an increase in the total mass or a cell and/or an increase in the total number of cells within a population of cells. Importantly, cell growth is not to be confused with cell division, which is a distinct process generally associated with cell proliferation and/or cell division. The basal growth medium can comprise glucose, salts, amino acids, and vitamins. For example, the basal growth medium can contain a Roswell Park Memorial Institute (RPMI) 1640 medium, X-VIVO™-15 medium, CTS™ OPTMIZER™ Serum Free Medium, CTS™ OPTMIZER™ Pro Serum Free Medium, and/or IMMUNOCULT™-XF T Cell Expansion Medium. In some embodiments, the basal growth medium comprises the RPMI 1640 medium with ATCC modification (Gibco A1049101). In some embodiments, the basal growth media comprises VIVO™- 15 medium. When the basal growth media comprises VIVO™- 15 medium, the VIVO™- 15 medium can be combined or mixed with any other basal growth media at any ratio that is particularly effective. Any volume of basal growth media is envisaged within the method disclosed herein.
In some instances, the myeloid media can further comprise GlutaMAX™ Supplement. The myeloid media can comprise a myeloid cytokine mixture, a basal growth medium and
GlutaMAX™ Supplement. The GlutaMAX™ Supplement can be present in the myeloid media at a concentration of between 0.5 mM, 1 mM, 1.5 mM, 2 mM, 2.5 mM or 3 mM of the myeloid media.
Disclosed herein are methods of manufacturing a genetically modified myeloid-derived cell comprising a CAR, wherein the method the method comprises the steps of: (a) contacting a precursor cell with a nucleic acid molecule comprising a polynucleotide sequence encoding a CAR, whereby said nucleic acid is introduced into said precursor cell, and (b) contacting said precursor cell with a precursor media comprising a precursor cytokine mixture, whereby the number of precursor cell(s) increases to generate a population of cells comprising a plurality of precursor CAR cells, wherein said precursor cell is a stem cell.
In some embodiments, the method comprises maintaining the precursor cell in the precursor medium of until a desired cell population is achieved. A skilled artisan would understand that a cell population can be analyzed and classified according to certain cell surface markers. For instance, a skilled artisan would be capable of determining the percentage of cells within a given cell population expressing a cell surface marker. The number of cell surface markers that can be is no limiting. Any number of cell surface markers can be analyzed during the methods disclosed herein to determine if a desired cell population has been achieved. Exemplary, non-limiting, cell surface markers that can be analyzed for the disclosed cell populations include: CD34, CD13, CD15, CD1 lb, and CD14. Any cell surface marker that can distinguish a stem cell and/or a myeloid- lineage cell from other known cell types are envisaged as being used.
The methods disclosed herein are particularly effective at generating myeloid-derived CAR cells from hematopoietic stem cells (HSCs) due, at least in part, to the particular precursor and myeloid mediums disclosed herein. For instance, the precursor mediums disclosed herein are particularly effective at expanding an HSC, or a population of HSCs. In some embodiments, the HSC is present within a population of HSCs. As described herein, a population of HSCs can be expanded (i.e., increased in number of cells) when at least one HSC within the population expands.
Disclosed herein are methods of generating a genetically modified myeloid-derived CAR cell from a stem cell, wherein the stem cell comprising a CAR (i.e., stem cell CAR cell) is expanded by contacting said stem cell CAR cell with a precursor media comprising a precursor cytokine mixture, whereby the number of stem cell CAR cell(s) increases to generate a population of stem cells comprising a plurality of stem cell CAR cells, wherein following contact of said stem cell CAR cell with said precursor media, said stem cell CAR cell differentiates into a myeloid- lineage cell to generate a myeloid-derived CAR cell or a population comprising a plurality of myeloid-derived CAR cells, wherein said cell population is a mixed population comprising said stem cell CAR cells and said myeloid-derived CAR cells, wherein said precursor media comprises
a precursor cytokine mixture comprising G-CSF, GM-CSF, SCF, and/or TPO, and wherein the stem cell CAR cell is maintained in said precursor media until no more than 98%, no more than 95%, no more than 90%, no more than 80%, no more than 70%, no more than 60%, no more than 50%, no more than 40%, no more than 30%, no more than 20%, no more than 10%, no more than 5%, no more than 1%, or no more than 0.1% of said population of cells is CD34+. In such instances, the precursor cytokine media can comprise: a. G-CSF at a concentration between about 100 ng/mL and 200 ng/mL; b. GM-CSF at a concentration between about 75 ng/mL and 125 ng/mL; c. SCF at a concentration between about 75 ng/mL and 125 ng/mL; and/or d. TPO at a concentration between about 75 ng/mL and 125 ng/mL. In such instances, the stem cell can be a hematopoietic stem cell. In such instances, the method can further comprise the step of contacting said stem cell CAR cell, myeloid-derived CAR cell, and/or said population comprising stem cell CAR cells and said myeloid-derived CAR cells with a myeloid media comprising a myeloid cytokine mixture, whereby said ratio of myeloid-derived CAR cells relative to stem cell CAR cells within the population is increased. In such instances, said cell population comprising said stem cell CAR cells and said myeloid-derived CAR cells is maintained in said myeloid media until no more than 1%, 0.9%, 0,8%, 0,7%, 0.6%, 0.5%, 0.4%, 0.3%, 0.2%, 0.1%, or less than 0.1% of said population of cells is CD34+. Such instances can involve a step wherein the precursor media is removed prior to the addition of said myeloid media. When the precursor media is removed, the cell population can be washed at least once prior to contact with a myeloid medium. At least one wash step between contact with a precursor medium and myeloid medium may be beneficial to ensure cytokines and molecule components from the precursor medium are removed. In such instances, the myeloid media can comprise a myeloid cytokine mixture comprising Macrophage colony-stimulating factor (MCSF), Interleukin-3 (IL-3), or Interleukin-6 (IL-6), or a combination thereof. In such instances, when the myeloid media comprises a myeloid cytokine mixture comprising Macrophage colony-stimulating factor (MCSF), Interleukin-3 (IL-3), or Interleukin-6 (IL-6), or a combination thereof, said myeloid media can comprise stem cells factor (SCF), Flt3 ligand, thrombopoietin (TPO), Granulocyte colony-stimulating factor (G-CSF), and Granulocytemacrophage colony-stimulating factor (GM-CSF), at a concentration of at most 50 ng/mL, at most 40 ng/mL, at most 30 ng/mL, at most 20 ng/mL, at most 10 ng/mL, at most 5 ng/mL, or below 5 ng/mL.
In some embodiments of the methods of generating a genetically modified myeloid-derived CAR cell disclosed herein, the stem cell CAR cell is expanded by contacting said stem cell CAR cell with a precursor media comprising a precursor cytokine mixture, whereby the number of stem cell CAR cell(s) increases to generate a population of stem cells comprising a plurality of stem cell
CAR cells, wherein following contact of said stem cell CAR cell with said precursor media, said stem cell CAR cell differentiates into a myeloid-lineage cell to generate a myeloid-derived CAR cell or a population comprising a plurality of myeloid-derived CAR cells, wherein said cell population is a mixed population comprising said stem cell CAR cells and said myeloid-derived CAR cells, wherein said precursor media comprises a precursor cytokine mixture comprising G- CSF, GM-CSF, SCF, and/or TPO, and wherein the stem cell CAR cell is maintained in said precursor media until no more than 5%, no more than 4%, no more than 3%, no more than 2%, no more than 1%, or no more than 0.1% of said population of cells is CD34+. In such instances, the precursor cytokine media can comprise: a. G-CSF at a concentration between about 100 ng/mL and 200 ng/mL; b. GM-CSF at a concentration between about 75 ng/mL and 125 ng/mL; c. SCF at a concentration between about 75 ng/mL and 125 ng/mL; and/or d. TPO at a concentration between about 75 ng/mL and 125 ng/mL. In such instances, the stem cell can be a hematopoietic stem cell. In such instances, the method can further comprise the step of contacting said stem cell CAR cell, myeloid-derived CAR cell, and/or said population comprising stem cell CAR cells and said myeloid-derived CAR cells with a myeloid media comprising a myeloid cytokine mixture, whereby said ratio of myeloid-derived CAR cells relative to stem cell CAR cells within the population is increased. In such instances, said cell population comprising said stem cell CAR cells and said myeloid-derived CAR cells is maintained in said myeloid media until no more than 1%, 0.9%, 0,8%, 0,7%, 0.6%, 0.5%, 0.4%, 0.3%, 0.2%, 0.1%, or less than 0.1% of said population of cells is CD34+. Such instances can involve a step wherein the precursor media is removed prior to the addition of said myeloid media. When the precursor media is removed, the cell population can be washed at least once prior to contact with a myeloid medium. At least one wash step between contact with a precursor medium and myeloid medium may be beneficial to ensure cytokines and molecule components from the precursor medium are removed. In such instances, the myeloid media can comprise a myeloid cytokine mixture comprising Macrophage colony-stimulating factor (MCSF), Interleukin-3 (IL-3), or Interleukin-6 (IL-6), or a combination thereof. In such instances, when the myeloid media comprises a myeloid cytokine mixture comprising Macrophage colonystimulating factor (MCSF), Interleukin-3 (IL-3), or Interleukin-6 (IL-6), or a combination thereof, said myeloid media can comprise stem cells factor (SCF), Flt3 ligand, thrombopoietin (TPO), Granulocyte colony-stimulating factor (G-CSF), and Granulocyte-macrophage colony-stimulating factor (GM-CSF), at a concentration of at most 50 ng/mL, at most 40 ng/mL, at most 30 ng/mL, at most 20 ng/mL, at most 10 ng/mL, at most 5 ng/mL, or below 5 ng/mL.
In some embodiments of the methods of generating a genetically modified myeloid-derived CAR cell from a stem cell, the stem cell comprising a CAR (i.e., stem cell CAR cell) is expanded
by contacting said stem cell CAR cell with a precursor media comprising a precursor cytokine mixture, whereby the number of stem cell CAR cell(s) increases to generate a population of stem cells comprising a plurality of stem cell CAR cells, wherein following contact of said stem cell CAR cell with said precursor media, said stem cell CAR cell differentiates into a myeloid-lineage cell to generate a myeloid-derived CAR cell or a population comprising a plurality of myeloid- derived CAR cells, wherein said cell population is a mixed population comprising said stem cell CAR cells and said myeloid-derived CAR cells, wherein said precursor media comprises a precursor cytokine mixture comprising G-CSF, GM-CSF, SCF, and/or TPO, and wherein the stem cell CAR cell is maintained in said precursor media until no more than 98%, no more than 95%, no more than 90%, no more than 80%, no more than 70%, no more than 60%, no more than 50%, no more than 40%, no more than 30%, no more than 20%, no more than 10%, no more than 5%, no more than 1%, or no more than 0.1% of said population of cells is CD34+, and until the number of genetically modified precursor CAR cells is increased by at least 20-fold, at least 50-fold, at least 75-fold, at least 100-fold, at least 200-fold, at least 300-fold, at least 400-fold, at least 500-fold, at least 1000-fold, or more than 1000-fold. In such instances, the precursor cytokine media can comprise: a. G-CSF at a concentration between about 100 ng/mL and 200 ng/mL; b. GM-CSF at a concentration between about 75 ng/mL and 125 ng/mL; c. SCF at a concentration between about 75 ng/mL and 125 ng/mL; and/or d. TPO at a concentration between about 75 ng/mL and 125 ng/mL. In such instances, the stem cell can be a hematopoietic stem cell. In such instances, the method can further comprise the step of contacting said stem cell CAR cell, myeloid-derived CAR cell, and/or said population comprising stem cell CAR cells and said myeloid-derived CAR cells with a myeloid media comprising a myeloid cytokine mixture, whereby said ratio of myeloid-derived CAR cells relative to stem cell CAR cells within the population is increased. In such instances, said cell population comprising said stem cell CAR cells and said myeloid-derived CAR cells is maintained in said myeloid media until no more than 1%, 0.9%, 0,8%, 0,7%, 0.6%, 0.5%, 0.4%, 0.3%, 0.2%, 0.1%, or less than 0.1% of said population of cells is CD34+. Such instances can involve a step wherein the precursor media is removed prior to the addition of said myeloid media. When the precursor media is removed, the cell population can be washed at least once prior to contact with a myeloid medium. At least one wash step between contact with a precursor medium and myeloid medium may be beneficial to ensure cytokines and molecule components from the precursor medium are removed. In such instances, the myeloid media can comprise a myeloid cytokine mixture comprising Macrophage colony-stimulating factor (MCSF), Interleukin-3 (IL-3), or Interleukin-6 (IL-6), or a combination thereof. In such instances, when the myeloid media comprises a myeloid cytokine mixture comprising Macrophage colony-stimulating factor (MCSF),
Interleukin-3 (IL-3), or Interleukin-6 (IL-6), or a combination thereof, said myeloid media can comprise stem cells factor (SCF), Flt3 ligand, thrombopoietin (TPO), Granulocyte colonystimulating factor (G-CSF), and Granulocyte-macrophage colony-stimulating factor (GM-CSF), at a concentration of at most 50 ng/mL, at most 40 ng/mL, at most 30 ng/mL, at most 20 ng/mL, at most 10 ng/mL, at most 5 ng/mL, or below 5 ng/mL.
In some embodiments of the methods of generating a genetically modified myeloid-derived CAR cell disclosed herein, the stem cell CAR cell is expanded by contacting said stem cell CAR cell with a precursor media comprising a precursor cytokine mixture, whereby the number of stem cell CAR cell(s) increases to generate a population of stem cells comprising a plurality of stem cell CAR cells, wherein following contact of said stem cell CAR cell with said precursor media, said stem cell CAR cell differentiates into a myeloid-lineage cell to generate a myeloid-derived CAR cell or a population comprising a plurality of myeloid-derived CAR cells, wherein said cell population is a mixed population comprising said stem cell CAR cells and said myeloid-derived CAR cells, wherein said precursor media comprises a precursor cytokine mixture comprising G- CSF, GM-CSF, SCF, and/or TPO, and wherein the stem cell CAR cell is maintained in said precursor media until no more than 5%, no more than 4%, no more than 3%, no more than 2%, no more than 1%, or no more than 0.1% of said population of cells is CD34+, and until the number of genetically modified precursor CAR cells is increased by at least 20-fold, at least 50-fold, at least 75-fold, at least 100-fold, at least 200-fold, at least 300-fold, at least 400-fold, at least 500-fold, at least 1000-fold, or more than 1000-fold. In such instances, the precursor cytokine media can comprise: a. G-CSF at a concentration between about 100 ng/mL and 200 ng/mL; b. GM-CSF at a concentration between about 75 ng/mL and 125 ng/mL; c. SCF at a concentration between about 75 ng/mL and 125 ng/mL; and/or d. TPO at a concentration between about 75 ng/mL and 125 ng/mL. In such instances, the stem cell can be a hematopoietic stem cell. In such instances, the method can further comprise the step of contacting said stem cell CAR cell, myeloid-derived CAR cell, and/or said population comprising stem cell CAR cells and said myeloid-derived CAR cells with a myeloid media comprising a myeloid cytokine mixture, whereby said ratio of myeloid-derived CAR cells relative to stem cell CAR cells within the population is increased. In such instances, said cell population comprising said stem cell CAR cells and said myeloid-derived CAR cells is maintained in said myeloid media until no more than 1%, 0.9%, 0,8%, 0,7%, 0.6%, 0.5%, 0.4%, 0.3%, 0.2%, 0.1%, or less than 0.1% of said population of cells is CD34+. Such instances can involve a step wherein the precursor media is removed prior to the addition of said myeloid media. When the precursor media is removed, the cell population can be washed at least once prior to contact with a myeloid medium. At least one wash step between contact with a precursor medium
and myeloid medium may be beneficial to ensure cytokines and molecule components from the precursor medium are removed. In such instances, the myeloid media can comprise a myeloid cytokine mixture comprising Macrophage colony-stimulating factor (MCSF), Interleukin-3 (IL-3), or Interleukin-6 (IL-6), or a combination thereof. In such instances, when the myeloid media comprises a myeloid cytokine mixture comprising Macrophage colony-stimulating factor (MCSF), Interleukin-3 (IL-3), or Interleukin-6 (IL-6), or a combination thereof, said myeloid media can comprise stem cells factor (SCF), Flt3 ligand, thrombopoietin (TPO), Granulocyte colonystimulating factor (G-CSF), and Granulocyte-macrophage colony-stimulating factor (GM-CSF), at a concentration of at most 50 ng/mL, at most 40 ng/mL, at most 30 ng/mL, at most 20 ng/mL, at most 10 ng/mL, at most 5 ng/mL, or below 5 ng/mL.
In some embodiments of the methods of generating a genetically modified myeloid-derived CAR cell disclosed herein, the stem cell CAR cell is expanded by contacting said stem cell CAR cell with a precursor media comprising a precursor cytokine mixture, whereby the number of stem cell CAR cell(s) increases to generate a population of stem cells comprising a plurality of stem cell CAR cells, wherein following contact of said stem cell CAR cell with said precursor media, said stem cell CAR cell differentiates into a myeloid-lineage cell to generate a myeloid-derived CAR cell or a population comprising a plurality of myeloid-derived CAR cells, wherein said cell population is a mixed population comprising said stem cell CAR cells and said myeloid-derived CAR cells, wherein said precursor media comprises a precursor cytokine mixture comprising G- CSF, GM-CSF, SCF, and/or TPO, and wherein the stem cell CAR cell is maintained in said precursor media until at least 20%, at least 30%, at least 40%, at least 50%, at least 60%, at least 70%, at least 80%, or at least 90% of said population of cells is CD15+. In such instances, the precursor cytokine media can comprise: a. G-CSF at a concentration between about 100 ng/mL and 200 ng/mL; b. GM-CSF at a concentration between about 75 ng/mL and 125 ng/mL; c. SCF at a concentration between about 75 ng/mL and 125 ng/mL; and/or d. TPO at a concentration between about 75 ng/mL and 125 ng/mL. In such instances, the stem cell can be a hematopoietic stem cell. In such instances, the precursor cytokine media can comprise: a. G-CSF at a concentration between about 100 ng/mL and 200 ng/mL; b. GM-CSF at a concentration between about 75 ng/mL and 125 ng/mL; c. SCF at a concentration between about 75 ng/mL and 125 ng/mL; and/or d. TPO at a concentration between about 75 ng/mL and 125 ng/mL. In such instances, the stem cell can be a hematopoietic stem cell. In such instances, the method can further comprise the step of contacting said stem cell CAR cell, myeloid-derived CAR cell, and/or said population comprising stem cell CAR cells and said myeloid-derived CAR cells with a myeloid media comprising a myeloid cytokine mixture, whereby said ratio of myeloid-derived CAR cells relative to stem cell CAR cells
within the population is increased. In such instances, said cell population comprising said stem cell CAR cells and said myeloid-derived CAR cells is maintained in said myeloid media until no more than 1%, 0.9%, 0,8%, 0,7%, 0.6%, 0.5%, 0.4%, 0.3%, 0.2%, 0.1%, or less than 0.1% of said population of cells is CD34+. Such instances can involve a step wherein the precursor media is removed prior to the addition of said myeloid media. When the precursor media is removed, the cell population can be washed at least once prior to contact with a myeloid medium. At least one wash step between contact with a precursor medium and myeloid medium may be beneficial to ensure cytokines and molecule components from the precursor medium are removed. In such instances, the myeloid media can comprise a myeloid cytokine mixture comprising Macrophage colony-stimulating factor (MCSF), Interleukin-3 (IL-3), or Interleukin-6 (IL-6), or a combination thereof. In such instances, when the myeloid media comprises a myeloid cytokine mixture comprising Macrophage colony-stimulating factor (MCSF), Interleukin-3 (IL-3), or Interleukin-6 (IL-6), or a combination thereof, said myeloid media can comprise stem cells factor (SCF), Flt3 ligand, thrombopoietin (TPO), Granulocyte colony-stimulating factor (G-CSF), and Granulocytemacrophage colony-stimulating factor (GM-CSF), at a concentration of at most 50 ng/mL, at most 40 ng/mL, at most 30 ng/mL, at most 20 ng/mL, at most 10 ng/mL, at most 5 ng/mL, or below 5 ng/mL.
In some embodiments of the methods of generating a genetically modified myeloid-derived CAR cell disclosed herein, the stem cell CAR cell is expanded by contacting said stem cell CAR cell with a precursor media comprising a precursor cytokine mixture, whereby the number of stem cell CAR cell(s) increases to generate a population of stem cells comprising a plurality of stem cell CAR cells, wherein following contact of said stem cell CAR cell with said precursor media, said stem cell CAR cell differentiates into a myeloid-lineage cell to generate a myeloid-derived CAR cell or a population comprising a plurality of myeloid-derived CAR cells, wherein said cell population is a mixed population comprising said stem cell CAR cells and said myeloid-derived CAR cells, wherein said precursor media comprises a precursor cytokine mixture comprising G- CSF, GM-CSF, SCF, and/or TPO, and wherein the stem cell CAR cell is maintained in said precursor media until between about 60%-95% of said population of cells is CD15+. In such instances, the precursor cytokine media can comprise: a. G-CSF at a concentration between about 100 ng/mL and 200 ng/mL; b. GM-CSF at a concentration between about 75 ng/mL and 125 ng/mL; c. SCF at a concentration between about 75 ng/mL and 125 ng/mL; and/or d. TPO at a concentration between about 75 ng/mL and 125 ng/mL. In such instances, the stem cell can be a hematopoietic stem cell. In such instances, the method can further comprise the step of contacting said stem cell CAR cell, myeloid-derived CAR cell, and/or said population comprising stem cell
CAR cells and said myeloid-derived CAR cells with a myeloid media comprising a myeloid cytokine mixture, whereby said ratio of myeloid-derived CAR cells relative to stem cell CAR cells within the population is increased. In such instances, said cell population comprising said stem cell CAR cells and said myeloid-derived CAR cells is maintained in said myeloid media until no more than 1%, 0.9%, 0,8%, 0,7%, 0.6%, 0.5%, 0.4%, 0.3%, 0.2%, 0.1%, or less than 0.1% of said population of cells is CD34+. Such instances can involve a step wherein the precursor media is removed prior to the addition of said myeloid media. When the precursor media is removed, the cell population can be washed at least once prior to contact with a myeloid medium. At least one wash step between contact with a precursor medium and myeloid medium may be beneficial to ensure cytokines and molecule components from the precursor medium are removed. In such instances, the myeloid media can comprise a myeloid cytokine mixture comprising Macrophage colony-stimulating factor (MCSF), Interleukin-3 (IL-3), or Interleukin-6 (IL-6), or a combination thereof. In such instances, when the myeloid media comprises a myeloid cytokine mixture comprising Macrophage colony-stimulating factor (MCSF), Interleukin-3 (IL-3), or Interleukin-6 (IL-6), or a combination thereof, said myeloid media can comprise stem cells factor (SCF), Flt3 ligand, thrombopoietin (TPO), Granulocyte colony-stimulating factor (G-CSF), and Granulocytemacrophage colony-stimulating factor (GM-CSF), at a concentration of at most 50 ng/mL, at most 40 ng/mL, at most 30 ng/mL, at most 20 ng/mL, at most 10 ng/mL, at most 5 ng/mL, or below 5 ng/mL.
In some embodiments of the methods of generating a genetically modified myeloid-derived CAR cell from a stem cell, the stem cell comprising a CAR (i.e., stem cell CAR cell) is expanded by contacting said stem cell CAR cell with a precursor media comprising a precursor cytokine mixture, whereby the number of stem cell CAR cell(s) increases to generate a population of stem cells comprising a plurality of stem cell CAR cells, wherein following contact of said stem cell CAR cell with said precursor media, said stem cell CAR cell differentiates into a myeloid-lineage cell to generate a myeloid-derived CAR cell or a population comprising a plurality of myeloid- derived CAR cells, wherein said cell population is a mixed population comprising said stem cell CAR cells and said myeloid-derived CAR cells, wherein said precursor media comprises a precursor cytokine mixture comprising G-CSF, GM-CSF, SCF, and/or TPO, and wherein the stem cell CAR cell is maintained in said precursor media until at least 20%, at least 30%, at least 40%, at least 50%, at least 60%, at least 70%, at least 80%, or at least 90% of said population of cells is CD15+, and until the number of genetically modified precursor CAR cells is increased by at least 20-fold, at least 50-fold, at least 75-fold, at least 100-fold, at least 200-fold, at least 300-fold, at least 400-fold, at least 500-fold, at least 1000-fold, or more than 1000-fold. In such instances, the
precursor cytokine media can comprise: a. G-CSF at a concentration between about 100 ng/mL and 200 ng/mL; b. GM-CSF at a concentration between about 75 ng/mL and 125 ng/mL; c. SCF at a concentration between about 75 ng/mL and 125 ng/mL; and/or d. TPO at a concentration between about 75 ng/mL and 125 ng/mL. In such instances, the stem cell can be a hematopoietic stem cell. In such instances, the method can further comprise the step of contacting said stem cell CAR cell, myeloid-derived CAR cell, and/or said population comprising stem cell CAR cells and said myeloid-derived CAR cells with a myeloid media comprising a myeloid cytokine mixture, whereby said ratio of myeloid-derived CAR cells relative to stem cell CAR cells within the population is increased. In such instances, said cell population comprising said stem cell CAR cells and said myeloid-derived CAR cells is maintained in said myeloid media until no more than 1%, 0.9%, 0,8%, 0,7%, 0.6%, 0.5%, 0.4%, 0.3%, 0.2%, 0.1%, or less than 0.1% of said population of cells is CD34+. Such instances can involve a step wherein the precursor media is removed prior to the addition of said myeloid media. When the precursor media is removed, the cell population can be washed at least once prior to contact with a myeloid medium. At least one wash step between contact with a precursor medium and myeloid medium may be beneficial to ensure cytokines and molecule components from the precursor medium are removed. In such instances, the myeloid media can comprise a myeloid cytokine mixture comprising Macrophage colony-stimulating factor (MCSF), Interleukin-3 (IL-3), or Interleukin-6 (IL-6), or a combination thereof. In such instances, when the myeloid media comprises a myeloid cytokine mixture comprising Macrophage colonystimulating factor (MCSF), Interleukin-3 (IL-3), or Interleukin-6 (IL-6), or a combination thereof, said myeloid media can comprise stem cells factor (SCF), Flt3 ligand, thrombopoietin (TPO), Granulocyte colony-stimulating factor (G-CSF), and Granulocyte-macrophage colony-stimulating factor (GM-CSF), at a concentration of at most 50 ng/mL, at most 40 ng/mL, at most 30 ng/mL, at most 20 ng/mL, at most 10 ng/mL, at most 5 ng/mL, or below 5 ng/mL.
In some embodiments of the methods of generating a genetically modified myeloid-derived CAR cell disclosed herein, the stem cell CAR cell is expanded by contacting said stem cell CAR cell with a precursor media comprising a precursor cytokine mixture, whereby the number of stem cell CAR cell(s) increases to generate a population of stem cells comprising a plurality of stem cell CAR cells, wherein following contact of said stem cell CAR cell with said precursor media, said stem cell CAR cell differentiates into a myeloid-lineage cell to generate a myeloid-derived CAR cell or a population comprising a plurality of myeloid-derived CAR cells, wherein said cell population is a mixed population comprising said stem cell CAR cells and said myeloid-derived CAR cells, wherein said precursor media comprises a precursor cytokine mixture comprising G- CSF, GM-CSF, SCF, and/or TPO, and wherein the stem cell CAR cell is maintained in said
precursor media until between about 60%-95% of said population of cells is CD15+, and until the number of genetically modified precursor CAR cells is increased by at least 20-fold, at least 50- fold, at least 75-fold, at least 100-fold, at least 200-fold, at least 300-fold, at least 400-fold, at least 500-fold, at least 1000-fold, or more than 1000-fold. In such instances, the precursor cytokine media can comprise: a. G-CSF at a concentration between about 100 ng/mL and 200 ng/mL; b. GM-CSF at a concentration between about 75 ng/mL and 125 ng/mL; c. SCF at a concentration between about 75 ng/mL and 125 ng/mL; and/or d. TPO at a concentration between about 75 ng/mL and 125 ng/mL. In such instances, the stem cell can be a hematopoietic stem cell. In such instances, the method can further comprise the step of contacting said stem cell CAR cell, myeloid- derived CAR cell, and/or said population comprising stem cell CAR cells and said myeloid-derived CAR cells with a myeloid media comprising a myeloid cytokine mixture, whereby said ratio of myeloid-derived CAR cells relative to stem cell CAR cells within the population is increased. In such instances, said cell population comprising said stem cell CAR cells and said myeloid-derived CAR cells is maintained in said myeloid media until no more than 1%, 0.9%, 0,8%, 0,7%, 0.6%, 0.5%, 0.4%, 0.3%, 0.2%, 0.1%, or less than 0.1% of said population of cells is CD34+. Such instances can involve a step wherein the precursor media is removed prior to the addition of said myeloid media. When the precursor media is removed, the cell population can be washed at least once prior to contact with a myeloid medium. At least one wash step between contact with a precursor medium and myeloid medium may be beneficial to ensure cytokines and molecule components from the precursor medium are removed. In such instances, the myeloid media can comprise a myeloid cytokine mixture comprising Macrophage colony-stimulating factor (MCSF), Interleukin-3 (IL-3), or Interleukin-6 (IL-6), or a combination thereof. In such instances, when the myeloid media comprises a myeloid cytokine mixture comprising Macrophage colony-stimulating factor (MCSF), Interleukin-3 (IL-3), or Interleukin-6 (IL-6), or a combination thereof, said myeloid media can comprise stem cells factor (SCF), Flt3 ligand, thrombopoietin (TPO), Granulocyte colony-stimulating factor (G-CSF), and Granulocyte-macrophage colony-stimulating factor (GM- CSF), at a concentration of at most 50 ng/mL, at most 40 ng/mL, at most 30 ng/mL, at most 20 ng/mL, at most 10 ng/mL, at most 5 ng/mL, or below 5 ng/mL.
In some embodiments of the methods of generating a genetically modified myeloid-derived CAR cell disclosed herein, the stem cell CAR cell is expanded by contacting said stem cell CAR cell with a precursor media comprising a precursor cytokine mixture, whereby the number of stem cell CAR cell(s) increases to generate a population of stem cells comprising a plurality of stem cell CAR cells, wherein following contact of said stem cell CAR cell with said precursor media, said stem cell CAR cell differentiates into a myeloid-lineage cell to generate a myeloid-derived CAR
cell or a population comprising a plurality of myeloid-derived CAR cells, wherein said cell population is a mixed population comprising said stem cell CAR cells and said myeloid-derived CAR cells, wherein said precursor media comprises a precursor cytokine mixture comprising G- CSF, GM-CSF, SCF, and/or TPO, and wherein the stem cell CAR cell is maintained in said precursor media until at least 20%, at least 30%, at least 40%, at least 50%, at least 60%, at least 70%, at least 80%, or at least 90% of said population of cells is CD1 lb+. In such instances, the precursor cytokine media can comprise: a. G-CSF at a concentration between about 100 ng/mL and 200 ng/mL; b. GM-CSF at a concentration between about 75 ng/mL and 125 ng/mL; c. SCF at a concentration between about 75 ng/mL and 125 ng/mL; and/or d. TPO at a concentration between about 75 ng/mL and 125 ng/mL. In such instances, the stem cell can be a hematopoietic stem cell. In such instances, the method can further comprise the step of contacting said stem cell CAR cell, myeloid-derived CAR cell, and/or said population comprising stem cell CAR cells and said myeloid-derived CAR cells with a myeloid media comprising a myeloid cytokine mixture, whereby said ratio of myeloid-derived CAR cells relative to stem cell CAR cells within the population is increased. In such instances, said cell population comprising said stem cell CAR cells and said myeloid-derived CAR cells is maintained in said myeloid media until no more than 1%, 0.9%, 0,8%, 0,7%, 0.6%, 0.5%, 0.4%, 0.3%, 0.2%, 0.1%, or less than 0.1% of said population of cells is CD34+. Such instances can involve a step wherein the precursor media is removed prior to the addition of said myeloid media. When the precursor media is removed, the cell population can be washed at least once prior to contact with a myeloid medium. At least one wash step between contact with a precursor medium and myeloid medium may be beneficial to ensure cytokines and molecule components from the precursor medium are removed. In such instances, the myeloid media can comprise a myeloid cytokine mixture comprising Macrophage colony-stimulating factor (MCSF), Interleukin-3 (IL-3), or Interleukin-6 (IL-6), or a combination thereof. In such instances, when the myeloid media comprises a myeloid cytokine mixture comprising Macrophage colonystimulating factor (MCSF), Interleukin-3 (IL-3), or Interleukin-6 (IL-6), or a combination thereof, said myeloid media can comprise stem cells factor (SCF), Flt3 ligand, thrombopoietin (TPO), Granulocyte colony-stimulating factor (G-CSF), and Granulocyte-macrophage colony-stimulating factor (GM-CSF), at a concentration of at most 50 ng/mL, at most 40 ng/mL, at most 30 ng/mL, at most 20 ng/mL, at most 10 ng/mL, at most 5 ng/mL, or below 5 ng/mL.
In some embodiments of the methods of generating a genetically modified myeloid-derived CAR cell disclosed herein, the stem cell CAR cell is expanded by contacting said stem cell CAR cell with a precursor media comprising a precursor cytokine mixture, whereby the number of stem cell CAR cell(s) increases to generate a population of stem cells comprising a plurality of stem cell
CAR cells, wherein following contact of said stem cell CAR cell with said precursor media, said stem cell CAR cell differentiates into a myeloid-lineage cell to generate a myeloid-derived CAR cell or a population comprising a plurality of myeloid-derived CAR cells, wherein said cell population is a mixed population comprising said stem cell CAR cells and said myeloid-derived CAR cells, wherein said precursor media comprises a precursor cytokine mixture comprising G- CSF, GM-CSF, SCF, and/or TPO, and wherein the stem cell CAR cell is maintained in said precursor media between about 30%-85% of said population of cells is CD1 lb+. In such instances, the precursor cytokine media can comprise: a. G-CSF at a concentration between about 100 ng/mL and 200 ng/mL; b. GM-CSF at a concentration between about 75 ng/mL and 125 ng/mL; c. SCF at a concentration between about 75 ng/mL and 125 ng/mL; and/or d. TPO at a concentration between about 75 ng/mL and 125 ng/mL. In such instances, the stem cell can be a hematopoietic stem cell. In such instances, the method can further comprise the step of contacting said stem cell CAR cell, myeloid-derived CAR cell, and/or said population comprising stem cell CAR cells and said myeloid-derived CAR cells with a myeloid media comprising a myeloid cytokine mixture, whereby said ratio of myeloid-derived CAR cells relative to stem cell CAR cells within the population is increased. In such instances, said cell population comprising said stem cell CAR cells and said myeloid-derived CAR cells is maintained in said myeloid media until no more than 1%, 0.9%, 0,8%, 0,7%, 0.6%, 0.5%, 0.4%, 0.3%, 0.2%, 0.1%, or less than 0.1% of said population of cells is CD34+. Such instances can involve a step wherein the precursor media is removed prior to the addition of said myeloid media. When the precursor media is removed, the cell population can be washed at least once prior to contact with a myeloid medium. At least one wash step between contact with a precursor medium and myeloid medium may be beneficial to ensure cytokines and molecule components from the precursor medium are removed. In such instances, the myeloid media can comprise a myeloid cytokine mixture comprising Macrophage colony-stimulating factor (MCSF), Interleukin-3 (IL-3), or Interleukin-6 (IL-6), or a combination thereof. In such instances, when the myeloid media comprises a myeloid cytokine mixture comprising Macrophage colonystimulating factor (MCSF), Interleukin-3 (IL-3), or Interleukin-6 (IL-6), or a combination thereof, said myeloid media can comprise stem cells factor (SCF), Flt3 ligand, thrombopoietin (TPO), Granulocyte colony-stimulating factor (G-CSF), and Granulocyte-macrophage colony-stimulating factor (GM-CSF), at a concentration of at most 50 ng/mL, at most 40 ng/mL, at most 30 ng/mL, at most 20 ng/mL, at most 10 ng/mL, at most 5 ng/mL, or below 5 ng/mL.
In some embodiments of the methods of generating a genetically modified myeloid-derived CAR cell from a stem cell, the stem cell comprising a CAR (i.e., stem cell CAR cell) is expanded by contacting said stem cell CAR cell with a precursor media comprising a precursor cytokine
mixture, whereby the number of stem cell CAR cell(s) increases to generate a population of stem cells comprising a plurality of stem cell CAR cells, wherein following contact of said stem cell CAR cell with said precursor media, said stem cell CAR cell differentiates into a myeloid-lineage cell to generate a myeloid-derived CAR cell or a population comprising a plurality of myeloid- derived CAR cells, wherein said cell population is a mixed population comprising said stem cell CAR cells and said myeloid-derived CAR cells, wherein said precursor media comprises a precursor cytokine mixture comprising G-CSF, GM-CSF, SCF, and/or TPO, and wherein the stem cell CAR cell is maintained in said precursor media until at least 20%, at least 30%, at least 40%, at least 50%, at least 60%, at least 70%, at least 80%, or at least 90% of said population of cells is CD1 lb+, and until the number of genetically modified precursor CAR cells is increased by at least 20-fold, at least 50-fold, at least 75-fold, at least 100-fold, at least 200-fold, at least 300-fold, at least 400-fold, at least 500-fold, at least 1000-fold, or more than 1000-fold. In such instances, the precursor cytokine media can comprise: a. G-CSF at a concentration between about 100 ng/mL and 200 ng/mL; b. GM-CSF at a concentration between about 75 ng/mL and 125 ng/mL; c. SCF at a concentration between about 75 ng/mL and 125 ng/mL; and/or d. TPO at a concentration between about 75 ng/mL and 125 ng/mL. In such instances, the stem cell can be a hematopoietic stem cell. In such instances, the method can further comprise the step of contacting said stem cell CAR cell, myeloid-derived CAR cell, and/or said population comprising stem cell CAR cells and said myeloid-derived CAR cells with a myeloid media comprising a myeloid cytokine mixture, whereby said ratio of myeloid-derived CAR cells relative to stem cell CAR cells within the population is increased. In such instances, said cell population comprising said stem cell CAR cells and said myeloid-derived CAR cells is maintained in said myeloid media until no more than 1%, 0.9%, 0,8%, 0,7%, 0.6%, 0.5%, 0.4%, 0.3%, 0.2%, 0.1%, or less than 0.1% of said population of cells is CD34+. Such instances can involve a step wherein the precursor media is removed prior to the addition of said myeloid media. When the precursor media is removed, the cell population can be washed at least once prior to contact with a myeloid medium. At least one wash step between contact with a precursor medium and myeloid medium may be beneficial to ensure cytokines and molecule components from the precursor medium are removed. In such instances, the myeloid media can comprise a myeloid cytokine mixture comprising Macrophage colony-stimulating factor (MCSF), Interleukin-3 (IL-3), or Interleukin-6 (IL-6), or a combination thereof. In such instances, when the myeloid media comprises a myeloid cytokine mixture comprising Macrophage colonystimulating factor (MCSF), Interleukin-3 (IL-3), or Interleukin-6 (IL-6), or a combination thereof, said myeloid media can comprise stem cells factor (SCF), Flt3 ligand, thrombopoietin (TPO), Granulocyte colony-stimulating factor (G-CSF), and Granulocyte-macrophage colony-stimulating
factor (GM-CSF), at a concentration of at most 50 ng/mL, at most 40 ng/mL, at most 30 ng/mL, at most 20 ng/mL, at most 10 ng/mL, at most 5 ng/mL, or below 5 ng/mL.
In some embodiments of the methods of generating a genetically modified myeloid-derived CAR cell disclosed herein, the stem cell CAR cell is expanded by contacting said stem cell CAR cell with a precursor media comprising a precursor cytokine mixture, whereby the number of stem cell CAR cell(s) increases to generate a population of stem cells comprising a plurality of stem cell CAR cells, wherein following contact of said stem cell CAR cell with said precursor media, said stem cell CAR cell differentiates into a myeloid-lineage cell to generate a myeloid-derived CAR cell or a population comprising a plurality of myeloid-derived CAR cells, wherein said cell population is a mixed population comprising said stem cell CAR cells and said myeloid-derived CAR cells, wherein said precursor media comprises a precursor cytokine mixture comprising G- CSF, GM-CSF, SCF, and/or TPO, and wherein the stem cell CAR cell is maintained in said precursor media between about 30%-85% of said population of cells is CD1 lb+, and until the number of genetically modified precursor CAR cells is increased by at least 20-fold, at least 50- fold, at least 75-fold, at least 100-fold, at least 200-fold, at least 300-fold, at least 400-fold, at least 500-fold, at least 1000-fold, or more than 1000-fold. In such instances, the precursor cytokine media can comprise: a. G-CSF at a concentration between about 100 ng/mL and 200 ng/mL; b. GM-CSF at a concentration between about 75 ng/mL and 125 ng/mL; c. SCF at a concentration between about 75 ng/mL and 125 ng/mL; and/or d. TPO at a concentration between about 75 ng/mL and 125 ng/mL. In such instances, the stem cell can be a hematopoietic stem cell. In such instances, the method can further comprise the step of contacting said stem cell CAR cell, myeloid- derived CAR cell, and/or said population comprising stem cell CAR cells and said myeloid-derived CAR cells with a myeloid media comprising a myeloid cytokine mixture, whereby said ratio of myeloid-derived CAR cells relative to stem cell CAR cells within the population is increased. In such instances, said cell population comprising said stem cell CAR cells and said myeloid-derived CAR cells is maintained in said myeloid media until no more than 1%, 0.9%, 0,8%, 0,7%, 0.6%, 0.5%, 0.4%, 0.3%, 0.2%, 0.1%, or less than 0.1% of said population of cells is CD34+. Such instances can involve a step wherein the precursor media is removed prior to the addition of said myeloid media. When the precursor media is removed, the cell population can be washed at least once prior to contact with a myeloid medium. At least one wash step between contact with a precursor medium and myeloid medium may be beneficial to ensure cytokines and molecule components from the precursor medium are removed. In such instances, the myeloid media can comprise a myeloid cytokine mixture comprising Macrophage colony-stimulating factor (MCSF), Interleukin-3 (IL-3), or Interleukin-6 (IL-6), or a combination thereof. In such instances, when the
myeloid media comprises a myeloid cytokine mixture comprising Macrophage colony-stimulating factor (MCSF), Interleukin-3 (IL-3), or Interleukin-6 (IL-6), or a combination thereof, said myeloid media can comprise stem cells factor (SCF), Flt3 ligand, thrombopoietin (TPO), Granulocyte colony-stimulating factor (G-CSF), and Granulocyte-macrophage colony-stimulating factor (GM- CSF), at a concentration of at most 50 ng/mL, at most 40 ng/mL, at most 30 ng/mL, at most 20 ng/mL, at most 10 ng/mL, at most 5 ng/mL, or below 5 ng/mL.
In some embodiments of the methods of generating a genetically modified myeloid-derived CAR cell disclosed herein, the stem cell CAR cell is expanded by contacting said stem cell CAR cell with a precursor media comprising a precursor cytokine mixture, whereby the number of stem cell CAR cell(s) increases to generate a population of stem cells comprising a plurality of stem cell CAR cells, wherein following contact of said stem cell CAR cell with said precursor media, said stem cell CAR cell differentiates into a myeloid-lineage cell to generate a myeloid-derived CAR cell or a population comprising a plurality of myeloid-derived CAR cells, wherein said cell population is a mixed population comprising said stem cell CAR cells and said myeloid-derived CAR cells, wherein said precursor media comprises a precursor cytokine mixture comprising G- CSF, GM-CSF, SCF, and/or TPO, and wherein the stem cell CAR cell is maintained in said precursor media until at least 0.1%, at least 0.5%, at least 1%, at least 2%, at least 3%, at least 4%, at least 5%, at least 6%, at least 7%, at least 8%, at least 9%, or at least 10% of said population of cells is CD14+. In such instances, the precursor cytokine media can comprise: a. G-CSF at a concentration between about 100 ng/mL and 200 ng/mL; b. GM-CSF at a concentration between about 75 ng/mL and 125 ng/mL; c. SCF at a concentration between about 75 ng/mL and 125 ng/mL; and/or d. TPO at a concentration between about 75 ng/mL and 125 ng/mL. In such instances, the stem cell can be a hematopoietic stem cell. In such instances, the method can further comprise the step of contacting said stem cell CAR cell, myeloid-derived CAR cell, and/or said population comprising stem cell CAR cells and said myeloid-derived CAR cells with a myeloid media comprising a myeloid cytokine mixture, whereby said ratio of myeloid-derived CAR cells relative to stem cell CAR cells within the population is increased. In such instances, said cell population comprising said stem cell CAR cells and said myeloid-derived CAR cells is maintained in said myeloid media until no more than 1%, 0.9%, 0,8%, 0,7%, 0.6%, 0.5%, 0.4%, 0.3%, 0.2%, 0.1%, or less than 0.1% of said population of cells is CD34+. Such instances can involve a step wherein the precursor media is removed prior to the addition of said myeloid media. When the precursor media is removed, the cell population can be washed at least once prior to contact with a myeloid medium. At least one wash step between contact with a precursor medium and myeloid medium may be beneficial to ensure cytokines and molecule components from the precursor
medium are removed. In such instances, the myeloid media can comprise a myeloid cytokine mixture comprising Macrophage colony-stimulating factor (MCSF), Interleukin-3 (IL-3), or Interleukin-6 (IL-6), or a combination thereof. In such instances, when the myeloid media comprises a myeloid cytokine mixture comprising Macrophage colony-stimulating factor (MCSF), Interleukin-3 (IL-3), or Interleukin-6 (IL-6), or a combination thereof, said myeloid media can comprise stem cells factor (SCF), Flt3 ligand, thrombopoietin (TPO), Granulocyte colonystimulating factor (G-CSF), and Granulocyte-macrophage colony-stimulating factor (GM-CSF), at a concentration of at most 50 ng/mL, at most 40 ng/mL, at most 30 ng/mL, at most 20 ng/mL, at most 10 ng/mL, at most 5 ng/mL, or below 5 ng/mL.
In some embodiments of the methods of generating a genetically modified myeloid-derived CAR cell disclosed herein, the stem cell CAR cell is expanded by contacting said stem cell CAR cell with a precursor media comprising a precursor cytokine mixture, whereby the number of stem cell CAR cell(s) increases to generate a population of stem cells comprising a plurality of stem cell CAR cells, wherein following contact of said stem cell CAR cell with said precursor media, said stem cell CAR cell differentiates into a myeloid-lineage cell to generate a myeloid-derived CAR cell or a population comprising a plurality of myeloid-derived CAR cells, wherein said cell population is a mixed population comprising said stem cell CAR cells and said myeloid-derived CAR cells, wherein said precursor media comprises a precursor cytokine mixture comprising G- CSF, GM-CSF, SCF, and/or TPO, and wherein the stem cell CAR cell is maintained in said precursor media until between about 0.5%-10% of said population of cells is CD14+. In such instances, the precursor cytokine media can comprise: a. G-CSF at a concentration between about 100 ng/mL and 200 ng/mL; b. GM-CSF at a concentration between about 75 ng/mL and 125 ng/mL; c. SCF at a concentration between about 75 ng/mL and 125 ng/mL; and/or d. TPO at a concentration between about 75 ng/mL and 125 ng/mL. In such instances, the stem cell can be a hematopoietic stem cell. In such instances, the method can further comprise the step of contacting said stem cell CAR cell, myeloid-derived CAR cell, and/or said population comprising stem cell CAR cells and said myeloid-derived CAR cells with a myeloid media comprising a myeloid cytokine mixture, whereby said ratio of myeloid-derived CAR cells relative to stem cell CAR cells within the population is increased. In such instances, said cell population comprising said stem cell CAR cells and said myeloid-derived CAR cells is maintained in said myeloid media until no more than 1%, 0.9%, 0,8%, 0,7%, 0.6%, 0.5%, 0.4%, 0.3%, 0.2%, 0.1%, or less than 0.1% of said population of cells is CD34+. Such instances can involve a step wherein the precursor media is removed prior to the addition of said myeloid media. When the precursor media is removed, the cell population can be washed at least once prior to contact with a myeloid medium. At least one
wash step between contact with a precursor medium and myeloid medium may be beneficial to ensure cytokines and molecule components from the precursor medium are removed. In such instances, the myeloid media can comprise a myeloid cytokine mixture comprising Macrophage colony-stimulating factor (MCSF), Interleukin-3 (IL-3), or Interleukin-6 (IL-6), or a combination thereof. In such instances, when the myeloid media comprises a myeloid cytokine mixture comprising Macrophage colony-stimulating factor (MCSF), Interleukin-3 (IL-3), or Interleukin-6 (IL-6), or a combination thereof, said myeloid media can comprise stem cells factor (SCF), Flt3 ligand, thrombopoietin (TPO), Granulocyte colony-stimulating factor (G-CSF), and Granulocytemacrophage colony-stimulating factor (GM-CSF), at a concentration of at most 50 ng/mL, at most 40 ng/mL, at most 30 ng/mL, at most 20 ng/mL, at most 10 ng/mL, at most 5 ng/mL, or below 5 ng/mL.
In some embodiments of the methods of generating a genetically modified myeloid-derived CAR cell from a stem cell, the stem cell CAR cell is expanded by contacting said stem cell CAR cell with a precursor media comprising a precursor cytokine mixture, whereby the number of stem cell CAR cell(s) increases to generate a population of stem cells comprising a plurality of stem cell CAR cells, wherein following contact of said stem cell CAR cell with said precursor media, said stem cell CAR cell differentiates into a myeloid-lineage cell to generate a myeloid-derived CAR cell or a population comprising a plurality of myeloid-derived CAR cells, wherein said cell population is a mixed population comprising said stem cell CAR cells and said myeloid-derived CAR cells, wherein said precursor media comprises a precursor cytokine mixture comprising G- CSF, GM-CSF, SCF, and/or TPO, and wherein the stem cell CAR cell is maintained in said precursor media until at least 0.1%, at least 0.5%, at least 1%, at least 2%, at least 3%, at least 4%, at least 5%, at least 6%, at least 7%, at least 8%, at least 9%, or at least 10% of said population of cells is CD14+, and until the number of genetically modified precursor CAR cells is increased by at least 20-fold, at least 50-fold, at least 75-fold, at least 100-fold, at least 200-fold, at least 300-fold, at least 400-fold, at least 500-fold, at least 1000-fold, or more than 1000-fold. In such instances, the precursor cytokine media can comprise: a. G-CSF at a concentration between about 100 ng/mL and 200 ng/mL; b. GM-CSF at a concentration between about 75 ng/mL and 125 ng/mL; c. SCF at a concentration between about 75 ng/mL and 125 ng/mL; and/or d. TPO at a concentration between about 75 ng/mL and 125 ng/mL. In such instances, the stem cell can be a hematopoietic stem cell. In such instances, the method can further comprise the step of contacting said stem cell CAR cell, myeloid-derived CAR cell, and/or said population comprising stem cell CAR cells and said myeloid-derived CAR cells with a myeloid media comprising a myeloid cytokine mixture, whereby said ratio of myeloid-derived CAR cells relative to stem cell CAR cells within the population is
\T1
increased. In such instances, said cell population comprising said stem cell CAR cells and said myeloid-derived CAR cells is maintained in said myeloid media until no more than 1%, 0.9%, 0,8%, 0,7%, 0.6%, 0.5%, 0.4%, 0.3%, 0.2%, 0.1%, or less than 0.1% of said population of cells is CD34+. Such instances can involve a step wherein the precursor media is removed prior to the addition of said myeloid media. When the precursor media is removed, the cell population can be washed at least once prior to contact with a myeloid medium. At least one wash step between contact with a precursor medium and myeloid medium may be beneficial to ensure cytokines and molecule components from the precursor medium are removed. In such instances, the myeloid media can comprise a myeloid cytokine mixture comprising Macrophage colony-stimulating factor (MCSF), Interleukin-3 (IL-3), or Interleukin-6 (IL-6), or a combination thereof. In such instances, when the myeloid media comprises a myeloid cytokine mixture comprising Macrophage colonystimulating factor (MCSF), Interleukin-3 (IL-3), or Interleukin-6 (IL-6), or a combination thereof, said myeloid media can comprise stem cells factor (SCF), Flt3 ligand, thrombopoietin (TPO), Granulocyte colony-stimulating factor (G-CSF), and Granulocyte-macrophage colony-stimulating factor (GM-CSF), at a concentration of at most 50 ng/mL, at most 40 ng/mL, at most 30 ng/mL, at most 20 ng/mL, at most 10 ng/mL, at most 5 ng/mL, or below 5 ng/mL.
In some embodiments of the methods of generating a genetically modified myeloid-derived CAR cell disclosed herein, the stem cell CAR cell is expanded by contacting said stem cell CAR cell with a precursor media comprising a precursor cytokine mixture, whereby the number of stem cell CAR cell(s) increases to generate a population of stem cells comprising a plurality of stem cell CAR cells, wherein following contact of said stem cell CAR cell with said precursor media, said stem cell CAR cell differentiates into a myeloid-lineage cell to generate a myeloid-derived CAR cell or a population comprising a plurality of myeloid-derived CAR cells, wherein said cell population is a mixed population comprising said stem cell CAR cells and said myeloid-derived CAR cells, wherein said precursor media comprises a precursor cytokine mixture comprising G- CSF, GM-CSF, SCF, and/or TPO, and wherein the stem cell CAR cell is maintained in said precursor media until between about 0.5%-10% of said population of cells is CD14+, and until the number of genetically modified precursor CAR cells is increased by at least 20-fold, at least 50- fold, at least 75-fold, at least 100-fold, at least 200-fold, at least 300-fold, at least 400-fold, at least 500-fold, at least 1000-fold, or more than 1000-fold. In such instances, the precursor cytokine media can comprise: a. G-CSF at a concentration between about 100 ng/mL and 200 ng/mL; b. GM-CSF at a concentration between about 75 ng/mL and 125 ng/mL; c. SCF at a concentration between about 75 ng/mL and 125 ng/mL; and/or d. TPO at a concentration between about 75 ng/mL and 125 ng/mL. In such instances, the stem cell can be a hematopoietic stem cell. In such
instances, the method can further comprise the step of contacting said stem cell CAR cell, myeloid- derived CAR cell, and/or said population comprising stem cell CAR cells and said myeloid-derived CAR cells with a myeloid media comprising a myeloid cytokine mixture, whereby said ratio of myeloid-derived CAR cells relative to stem cell CAR cells within the population is increased. In such instances, said cell population comprising said stem cell CAR cells and said myeloid-derived CAR cells is maintained in said myeloid media until no more than 1%, 0.9%, 0,8%, 0,7%, 0.6%, 0.5%, 0.4%, 0.3%, 0.2%, 0.1%, or less than 0.1% of said population of cells is CD34+. Such instances can involve a step wherein the precursor media is removed prior to the addition of said myeloid media. When the precursor media is removed, the cell population can be washed at least once prior to contact with a myeloid medium. At least one wash step between contact with a precursor medium and myeloid medium may be beneficial to ensure cytokines and molecule components from the precursor medium are removed. In such instances, the myeloid media can comprise a myeloid cytokine mixture comprising Macrophage colony-stimulating factor (MCSF), Interleukin-3 (IL-3), or Interleukin-6 (IL-6), or a combination thereof. In such instances, when the myeloid media comprises a myeloid cytokine mixture comprising Macrophage colony-stimulating factor (MCSF), Interleukin-3 (IL-3), or Interleukin-6 (IL-6), or a combination thereof, said myeloid media can comprise stem cells factor (SCF), Flt3 ligand, thrombopoietin (TPO), Granulocyte colony-stimulating factor (G-CSF), and Granulocyte-macrophage colony-stimulating factor (GM- CSF), at a concentration of at most 50 ng/mL, at most 40 ng/mL, at most 30 ng/mL, at most 20 ng/mL, at most 10 ng/mL, at most 5 ng/mL, or below 5 ng/mL.
In some embodiments of the methods of generating a genetically modified myeloid-derived CAR cell disclosed herein, the stem cell CAR cell is expanded by contacting said stem cell CAR cell with a precursor media comprising a precursor cytokine mixture, whereby the number of stem cell CAR cell(s) increases to generate a population of stem cells comprising a plurality of stem cell CAR cells, wherein following contact of said stem cell CAR cell with said precursor media, said stem cell CAR cell differentiates into a myeloid-lineage cell to generate a myeloid-derived CAR cell or a population comprising a plurality of myeloid-derived CAR cells, wherein said cell population is a mixed population comprising said stem cell CAR cells and said myeloid-derived CAR cells, wherein said precursor media comprises a precursor cytokine mixture comprising G- CSF, GM-CSF, SCF, and/or TPO, and wherein the stem cell CAR cell is maintained in said precursor media until at least 20%, at least 30%, at least 40%, at least 50%, at least 60%, at least 70%, at least 80%, at least 90%, at least 95%, or at least 98% of said population of cells is CD13+. In such instances, the precursor cytokine media can comprise: a. G-CSF at a concentration between about 100 ng/mL and 200 ng/mL; b. GM-CSF at a concentration between about 75 ng/mL and 125
ng/mL; c. SCF at a concentration between about 75 ng/mL and 125 ng/mL; and/or d. TPO at a concentration between about 75 ng/mL and 125 ng/mL. In such instances, the stem cell can be a hematopoietic stem cell. In such instances, the method can further comprise the step of contacting said stem cell CAR cell, myeloid-derived CAR cell, and/or said population comprising stem cell CAR cells and said myeloid-derived CAR cells with a myeloid media comprising a myeloid cytokine mixture, whereby said ratio of myeloid-derived CAR cells relative to stem cell CAR cells within the population is increased. In such instances, said cell population comprising said stem cell CAR cells and said myeloid-derived CAR cells is maintained in said myeloid media until no more than 1%, 0.9%, 0,8%, 0,7%, 0.6%, 0.5%, 0.4%, 0.3%, 0.2%, 0.1%, or less than 0.1% of said population of cells is CD34+. Such instances can involve a step wherein the precursor media is removed prior to the addition of said myeloid media. When the precursor media is removed, the cell population can be washed at least once prior to contact with a myeloid medium. At least one wash step between contact with a precursor medium and myeloid medium may be beneficial to ensure cytokines and molecule components from the precursor medium are removed. In such instances, the myeloid media can comprise a myeloid cytokine mixture comprising Macrophage colony-stimulating factor (MCSF), Interleukin-3 (IL-3), or Interleukin-6 (IL-6), or a combination thereof. In such instances, when the myeloid media comprises a myeloid cytokine mixture comprising Macrophage colony-stimulating factor (MCSF), Interleukin-3 (IL-3), or Interleukin-6 (IL-6), or a combination thereof, said myeloid media can comprise stem cells factor (SCF), Flt3 ligand, thrombopoietin (TPO), Granulocyte colony-stimulating factor (G-CSF), and Granulocytemacrophage colony-stimulating factor (GM-CSF), at a concentration of at most 50 ng/mL, at most 40 ng/mL, at most 30 ng/mL, at most 20 ng/mL, at most 10 ng/mL, at most 5 ng/mL, or below 5 ng/mL.
In some embodiments of the methods of generating a genetically modified myeloid-derived CAR cell disclosed herein, the stem cell CAR cell is expanded by contacting said stem cell CAR cell with a precursor media comprising a precursor cytokine mixture, whereby the number of stem cell CAR cell(s) increases to generate a population of stem cells comprising a plurality of stem cell CAR cells, wherein following contact of said stem cell CAR cell with said precursor media, said stem cell CAR cell differentiates into a myeloid-lineage cell to generate a myeloid-derived CAR cell or a population comprising a plurality of myeloid-derived CAR cells, wherein said cell population is a mixed population comprising said stem cell CAR cells and said myeloid-derived CAR cells, wherein said precursor media comprises a precursor cytokine mixture comprising G- CSF, GM-CSF, SCF, and/or TPO, and wherein the stem cell CAR cell is maintained in said precursor media until between about 85%-98% of said population of cells is CD13+. In such
instances, the precursor cytokine media can comprise: a. G-CSF at a concentration between about 100 ng/mL and 200 ng/mL; b. GM-CSF at a concentration between about 75 ng/mL and 125 ng/mL; c. SCF at a concentration between about 75 ng/mL and 125 ng/mL; and/or d. TPO at a concentration between about 75 ng/mL and 125 ng/mL. In such instances, the stem cell can be a hematopoietic stem cell. In such instances, the method can further comprise the step of contacting said stem cell CAR cell, myeloid-derived CAR cell, and/or said population comprising stem cell CAR cells and said myeloid-derived CAR cells with a myeloid media comprising a myeloid cytokine mixture, whereby said ratio of myeloid-derived CAR cells relative to stem cell CAR cells within the population is increased. In such instances, said cell population comprising said stem cell CAR cells and said myeloid-derived CAR cells is maintained in said myeloid media until no more than 1%, 0.9%, 0,8%, 0,7%, 0.6%, 0.5%, 0.4%, 0.3%, 0.2%, 0.1%, or less than 0.1% of said population of cells is CD34+. Such instances can involve a step wherein the precursor media is removed prior to the addition of said myeloid media. When the precursor media is removed, the cell population can be washed at least once prior to contact with a myeloid medium. At least one wash step between contact with a precursor medium and myeloid medium may be beneficial to ensure cytokines and molecule components from the precursor medium are removed. In such instances, the myeloid media can comprise a myeloid cytokine mixture comprising Macrophage colony-stimulating factor (MCSF), Interleukin-3 (IL-3), or Interleukin-6 (IL-6), or a combination thereof. In such instances, when the myeloid media comprises a myeloid cytokine mixture comprising Macrophage colony-stimulating factor (MCSF), Interleukin-3 (IL-3), or Interleukin-6 (IL-6), or a combination thereof, said myeloid media can comprise stem cells factor (SCF), Flt3 ligand, thrombopoietin (TPO), Granulocyte colony-stimulating factor (G-CSF), and Granulocytemacrophage colony-stimulating factor (GM-CSF), at a concentration of at most 50 ng/mL, at most 40 ng/mL, at most 30 ng/mL, at most 20 ng/mL, at most 10 ng/mL, at most 5 ng/mL, or below 5 ng/mL.
In some embodiments of the methods of generating a genetically modified myeloid-derived CAR cell from a stem cell, the stem cell CAR cell is expanded by contacting said stem cell CAR cell with a precursor media comprising a precursor cytokine mixture, whereby the number of stem cell CAR cell(s) increases to generate a population of stem cells comprising a plurality of stem cell CAR cells, wherein following contact of said stem cell CAR cell with said precursor media, said stem cell CAR cell differentiates into a myeloid-lineage cell to generate a myeloid-derived CAR cell or a population comprising a plurality of myeloid-derived CAR cells, wherein said cell population is a mixed population comprising said stem cell CAR cells and said myeloid-derived CAR cells, wherein said precursor media comprises a precursor cytokine mixture comprising G-
CSF, GM-CSF, SCF, and/or TPO, and wherein the stem cell CAR cell is maintained in said precursor media until at least 20%, at least 30%, at least 40%, at least 50%, at least 60%, at least 70%, at least 80%, at least 90%, at least 95%, or at least 98% of said population of cells is CD13+, and until the number of genetically modified precursor CAR cells is increased by at least 20-fold, at least 50-fold, at least 75-fold, at least 100-fold, at least 200-fold, at least 300-fold, at least 400-fold, at least 500-fold, at least 1000-fold, or more than 1000-fold. In such instances, the precursor cytokine media can comprise: a. G-CSF at a concentration between about 100 ng/mL and 200 ng/mL; b. GM-CSF at a concentration between about 75 ng/mL and 125 ng/mL; c. SCF at a concentration between about 75 ng/mL and 125 ng/mL; and/or d. TPO at a concentration between about 75 ng/mL and 125 ng/mL. In such instances, the stem cell can be a hematopoietic stem cell. In such instances, the method can further comprise the step of contacting said stem cell CAR cell, myeloid-derived CAR cell, and/or said population comprising stem cell CAR cells and said myeloid-derived CAR cells with a myeloid media comprising a myeloid cytokine mixture, whereby said ratio of myeloid-derived CAR cells relative to stem cell CAR cells within the population is increased. In such instances, said cell population comprising said stem cell CAR cells and said myeloid-derived CAR cells is maintained in said myeloid media until no more than 1%, 0.9%, 0,8%, 0,7%, 0.6%, 0.5%, 0.4%, 0.3%, 0.2%, 0.1%, or less than 0.1% of said population of cells is CD34+. Such instances can involve a step wherein the precursor media is removed prior to the addition of said myeloid media. When the precursor media is removed, the cell population can be washed at least once prior to contact with a myeloid medium. At least one wash step between contact with a precursor medium and myeloid medium may be beneficial to ensure cytokines and molecule components from the precursor medium are removed. In such instances, the myeloid media can comprise a myeloid cytokine mixture comprising Macrophage colony-stimulating factor (MCSF), Interleukin-3 (IL-3), or Interleukin-6 (IL-6), or a combination thereof. In such instances, when the myeloid media comprises a myeloid cytokine mixture comprising Macrophage colonystimulating factor (MCSF), Interleukin-3 (IL-3), or Interleukin-6 (IL-6), or a combination thereof, said myeloid media can comprise stem cells factor (SCF), Flt3 ligand, thrombopoietin (TPO), Granulocyte colony-stimulating factor (G-CSF), and Granulocyte-macrophage colony-stimulating factor (GM-CSF), at a concentration of at most 50 ng/mL, at most 40 ng/mL, at most 30 ng/mL, at most 20 ng/mL, at most 10 ng/mL, at most 5 ng/mL, or below 5 ng/mL.
In some embodiments of the methods of generating a genetically modified myeloid-derived CAR cell disclosed herein, the stem cell CAR cell is expanded by contacting said stem cell CAR cell with a precursor media comprising a precursor cytokine mixture, whereby the number of stem cell CAR cell(s) increases to generate a population of stem cells comprising a plurality of stem cell
CAR cells, wherein following contact of said stem cell CAR cell with said precursor media, said stem cell CAR cell differentiates into a myeloid-lineage cell to generate a myeloid-derived CAR cell or a population comprising a plurality of myeloid-derived CAR cells, wherein said cell population is a mixed population comprising said stem cell CAR cells and said myeloid-derived CAR cells, wherein said precursor media comprises a precursor cytokine mixture comprising G- CSF, GM-CSF, SCF, and/or TPO, and wherein the stem cell CAR cell is maintained in said precursor media until between about 85%-98% of said population of cells is CD13+, and until the number of genetically modified precursor CAR cells is increased by at least 20-fold, at least 50- fold, at least 75-fold, at least 100-fold, at least 200-fold, at least 300-fold, at least 400-fold, at least 500-fold, at least 1000-fold, or more than 1000-fold. In such instances, the precursor cytokine media can comprise: a. G-CSF at a concentration between about 100 ng/mL and 200 ng/mL; b. GM-CSF at a concentration between about 75 ng/mL and 125 ng/mL; c. SCF at a concentration between about 75 ng/mL and 125 ng/mL; and/or d. TPO at a concentration between about 75 ng/mL and 125 ng/mL. In such instances, the stem cell can be a hematopoietic stem cell. In such instances, the method can further comprise the step of contacting said stem cell CAR cell, myeloid- derived CAR cell, and/or said population comprising stem cell CAR cells and said myeloid-derived CAR cells with a myeloid media comprising a myeloid cytokine mixture, whereby said ratio of myeloid-derived CAR cells relative to stem cell CAR cells within the population is increased. In such instances, said cell population comprising said stem cell CAR cells and said myeloid-derived CAR cells is maintained in said myeloid media until no more than 1%, 0.9%, 0,8%, 0,7%, 0.6%, 0.5%, 0.4%, 0.3%, 0.2%, 0.1%, or less than 0.1% of said population of cells is CD34+. Such instances can involve a step wherein the precursor media is removed prior to the addition of said myeloid media. When the precursor media is removed, the cell population can be washed at least once prior to contact with a myeloid medium. At least one wash step between contact with a precursor medium and myeloid medium may be beneficial to ensure cytokines and molecule components from the precursor medium are removed. In such instances, the myeloid media can comprise a myeloid cytokine mixture comprising Macrophage colony-stimulating factor (MCSF), Interleukin-3 (IL-3), or Interleukin-6 (IL-6), or a combination thereof. In such instances, when the myeloid media comprises a myeloid cytokine mixture comprising Macrophage colony-stimulating factor (MCSF), Interleukin-3 (IL-3), or Interleukin-6 (IL-6), or a combination thereof, said myeloid media can comprise stem cells factor (SCF), Flt3 ligand, thrombopoietin (TPO), Granulocyte colony-stimulating factor (G-CSF), and Granulocyte-macrophage colony-stimulating factor (GM- CSF), at a concentration of at most 50 ng/mL, at most 40 ng/mL, at most 30 ng/mL, at most 20 ng/mL, at most 10 ng/mL, at most 5 ng/mL, or below 5 ng/mL.
VI. Compositions
The myeloid-derived cells and/or genetically modified myeloid-derived cells expressing a CAR of the disclosure can be comprised in a composition together with a compatible vehicle. The term “vehicle” as used herein indicates any of various media acting usually as solvents, carriers, binders, or diluents for the modified phagocytic cells expressing a CAR as described herein that are comprised in the composition as an active ingredient. In some embodiments, the vehicle is a pharmaceutically acceptable vehicle and the composition is a pharmaceutically acceptable composition. Pharmaceutical compositions of the present disclosure can comprise modified myeloid-derived or phagocytic cells as described herein, in combination with one or more pharmaceutically or physiologically acceptable carriers, diluents, or excipients. The term "pharmaceutically acceptable" means not biologically or otherwise undesirable, in that it can be administered to a subject without excessive toxicity, irritation, or allergic response, and does not cause unacceptable biological effects or interact in a deleterious manner with any of the other components of the composition in which it is contained. Such compositions may comprise buffers such as neutral buffered saline, phosphate buffered saline and the like; carbohydrates such as glucose, mannose, sucrose or dextrans, mannitol; proteins; polypeptides or amino acids such as glycine; antioxidants; chelating agents such as EDTA or glutathione; adjuvants (e.g., aluminum hydroxide); and preservatives. Compositions of the present disclosure are preferably formulated for intravenous administration.
Suitable vehicles for an injectable composition comprise a solvent or dispersion medium containing, for example, water, ethanol, polyol (for example, glycerol, propylene glycol, and liquid polyethylene glycol, and the like), and suitable mixtures thereof. Suitable vehicles for oral composition comprise inert diluent or an edible carrier and excipients which can be combined with the active ingredients in the form of tablets, pills, troches, or capsules, e.g., gelatin capsules. Pharmaceutically compatible binding agents, and/or adjuvant materials can be included as part of the composition, such as microcrystalline cellulose, gum tragacanth or gelatin and additional binding agents and/or adjuvant identifiable by a skilled person. Suitable vehicles for aerosol spray used for inhalation from a pressured container or dispenser can contain a suitable propellant, e.g., a gas such as carbon dioxide, or a nebulizer and are expected to be formulated and/or administered with methods such as the ones described in US 6,468,798, incorporated herein by reference in its entirety. Suitable vehicles for transmucosal or transdermal administration comprising penetrants appropriate to the barrier to be permeated can be used in the formulation. Such penetrants are generally known in the art, and include, for example, for transmucosal administration, detergents, bile salts, and fusidic acid derivatives. Transmucosal administration can be accomplished through the use of nasal sprays or suppositories. For transdermal administration, the active compounds are formulated into ointments, salves, gels, or
creams as generally known in the art. Suitable vehicles for composition in the form of suppositories (e.g., with conventional suppository bases such as cocoa butter and other glycerides) or retention enemas for rectal delivery. In some embodiments, pharmaceutical composition carriers are included that will protect the modified phagocytic cells against rapid elimination from the body, such as a controlled release formulation, including implants and microencapsulated delivery systems.
Pharmaceutical compositions of the present disclosure may be administered in a manner appropriate to the disease to be treated (or prevented). The quantity and frequency of administration will be determined by such factors as the condition of the patient, and the type and severity of the patient's disease, although appropriate dosages may be determined by clinical trials.
When "an immunologically effective amount", "a therapeutically effective amount", "an effective amount", or "therapeutic amount" is indicated, the precise amount of the compositions of the present disclosure to be administered can be determined by a physician with consideration of individual differences in age, weight, immune response, and condition of the patient (subject). A pharmaceutical composition comprising modified myeloid-derived cells expressing a CAR as described herein may be administered at a dosage of about 103 to about IO10 cells/kg body weight, and in some embodiments, the dosage can be from about 105 to about 108 cells/kg body weight or from about 106 to about 108 cells/kg body weight, including all integer values (e.g., 104, 105, 106, 107, 108, 109) within those ranges. The cell compositions of the disclosure can be administered multiple times (e.g., hourly, four times daily, three times daily, two times daily, daily, twice weekly, three times weekly, weekly, monthly, bi-monthly, semi-annually, annually, etc.) at these dosages. The cells can be administered by using infusion techniques that are commonly known in immunotherapy (see, e.g., Rosenberg et al., New Eng. J. of Med. 319: 1676-1988). The optimal dosage and treatment regime for a particular patient can readily be determined by one skilled in the art of medicine by monitoring the patient for signs of disease and adjusting the treatment accordingly.
In some embodiments of the present disclosure, cells are modified using the methods described herein, or other methods known in the art where the cells are expanded to therapeutic levels, are administered to a patient in conjunction any number of relevant treatment modalities, including one or more of radiation therapy, genetically engineered cellular immunotherapy (e.g., T cell, natural killer cell, chimeric antigen receptor (CAR) therapy), antibody therapy, immune checkpoint molecule inhibitor therapy, or a pharmaceutical therapy, such as a chemotherapeutic, a therapeutic peptide, antibiotic, anti-viral agent, anti-fungal agent, anti-inflammatory agent, or a small molecule therapy. In embodiments where a composition comprising modified myeloid-derived cells expressing a CAR as described herein are administered in combination with one or more additional therapies, the one or more additional therapies may be administered at a sub therapeutic dose due to an additive or
synergistic effect of the combination with the composition comprising modified phagocytic cells. Combination therapy includes administration of a composition comprising modified myeloid-derived cells of the disclosure before an additional therapy (e.g., 1 day to 30 days or more before the additional therapy), concurrently with an additional therapy (on the same day), or after an additional therapy (e.g., 1 day to 30 days or more after the additional therapy). Where the one or more additional therapies involves multiple doses, a composition comprising modified phagocytic cells as described herein may be administered after the initial dose of the one or more additional therapies, after the final dose of the one or more additional therapies, or in between multiple doses of the one or more additional therapies. For example, subjects may undergo standard treatment with high dose chemotherapy followed by peripheral blood stem cell transplantation. In some embodiments, following the transplant, subjects receive an infusion of a composition comprising modified myeloid-derived cells of the present disclosure. In some embodiments, a composition comprising modified myeloid-derived cells of the present disclosure may be administered before or following surgery.
The dosage of one or more additional therapies to be administered to a subject will vary with the precise nature of the condition being treated and the recipient of the treatment. The scaling of dosages for human administration can be performed according to art-accepted practices.
Expression vectors comprising genes encoding CARs of the disclosure, along with myeloid- derived cells modified to express these CARs can be provided as part of a system to treat a disease or disorder (e.g., cancer). The systems herein described can be provided in the form of kits of parts. In kits of parts for performing any one of the methods herein described, the expression vectors, genes encoding CARs, genetically modified myeloid-derived cells expressing a CAR of the disclosure, and compositions comprising the CAR-modified myeloid-derived cells can be included in the kit alone or in the presence of additional labels for detection of cells as well as additional components identifiable by a skilled person. In a kit of parts, the expression vectors, genes encoding CARs, genetically modified myeloid-derived cells expressing a CAR of the disclosure, compositions comprising the CAR-modified myeloid-derived cells, and additional reagents identifiable by a skilled person are comprised in the kit independently and possibly includes suitable vehicle carriers or auxiliary agents. Additional components can include labels, reference standards, and additional components identifiable by a skilled person upon reading of the present disclosure. The terms “label” and “labeled molecule” as used herein refer to a molecule capable of detection, including radioactive isotopes, fluorophores, chemiluminescent dyes, chromophores, enzymes, enzymes substrates, enzyme cofactors, enzyme inhibitors, dyes, metal ions, nanoparticles, metal sols, ligands (such as biotin, avidin, streptavidin or haptens) and the like. The term “fluorophore” refers to a substance or a portion thereof which is capable of exhibiting fluorescence in a detectable image. As a consequence, the term “labeling signal”
as used herein indicates the signal emitted from the label that allows detection of the label, including radioactivity, fluorescence, chemoluminescence, production of a compound in an enzymatic reaction, and the like. The components of the kit can be provided, with suitable instructions and other necessary reagents, in order to perform the methods here disclosed. The kit will normally contain the compositions in separate containers. Instructions, for example written or audio instructions, on paper or electronic support such as tapes, CD-ROMs, flash drives, or by indication of a Uniform Resource Locator (URL), which contains a pdf copy of the instructions for carrying out the assay, will usually be included in the kit. The kit can also contain, depending on the particular method used, other packaged reagents and materials (e.g., wash buffers and the like).
VII. Methods of treatment
The myeloid-derived cells and/or myeloid-derived CAR cells described herein may be included in a composition for treatment of a disease or disorder in a subject. The composition may include a pharmaceutical composition and further include a pharmaceutically acceptable carrier. A therapeutically effective amount of the pharmaceutical composition comprising the modified phagocytic cells may be administered. A "disease" is a state of health of an animal wherein the animal cannot maintain homeostasis, and wherein if the disease is not ameliorated then the animal's health continues to deteriorate. A "disorder" in an animal is a state of health in which the animal is able to maintain homeostasis, but in which the animal's state of health is less favorable than it would be in the absence of the disorder. Left untreated, a disorder does not necessarily cause a further decrease in the animal's state of health.
The disclosure includes a method of treating a disease or condition associated with a tumor or cancer in a subject in need thereof comprising administering to the subject a therapeutically effective amount of a pharmaceutical composition comprising any genetically modified myeloid-derived CAR cell described herein. The disclosure further includes a method of treating a tumor in a subject in need thereof, comprising administering to the subject a therapeutically effective amount of a pharmaceutical composition comprising any genetically modified myeloid-derived CAR cell described herein. The disclosure further provides a method of treating a tumor in a subject in need thereof, comprising administering to the subject a therapeutically effective amount of a pharmaceutical composition comprising any genetically modified myeloid-derived CAR cell described herein. The disclosure further provides a method of treating an infection in a subject in need thereof, comprising administering to the subject a therapeutically effective amount of a pharmaceutical composition comprising any genetically modified myeloid-derived CAR cell described herein. The disclosure further provides a method for stimulating an immune response to a target tumor cell or tumor tissue in a subject in need thereof, comprising administering to the subject a therapeutically effective amount of
a pharmaceutical composition comprising any genetically modified myeloid-derived CAR cell described herein. The disclosure further provides a method of treating a subject by engulfment and/or trogocytosis of a target cell in a subject in need thereof, comprising administering to the subject a therapeutically effective amount of a pharmaceutical composition comprising any genetically modified myeloid-derived CAR cell described herein.
Also disclosed herein are uses of the myeloid-derived CAR cells of the present disclosure. For example, the myeloid-derived CAR cells can be used in the manufacture of a medicament for any treatment disclosed herein. The myeloid-derived CAR cells can be used in the manufacture of a medicament for the treatment of a disease or a condition associated with a tumor or cancer in a subject in need thereof. The myeloid-derived CAR cells can be used in the manufacture of a medicament of treating a tumor in a subject in need thereof. The myeloid-derived CAR cells can be used in the manufacture of a medicament for the treatment of treating an infection in a subject in need thereof. The myeloid-derived CAR cells can be used in the manufacture of a medicament for stimulating an immune response to a target tumor cell or tumor tissue in a subject in need thereof. The myeloid- derived CAR cells can be used in the manufacture of a medicament for the treating a subject by engulfment and/or trogocytosis of a target cell in a subject in need thereof.
The myeloid-derived CAR cells can be used for treatment of a disease or disorder in a subject. In some embodiments, the myeloid-derived CAR cells can be used for the treatment of cancer. In some embodiments, the myeloid-derived CAR cells can be used for the treatment of a disorder or symptom associated with cancer. In some embodiments, the myeloid-derived CAR cells can be used for the treatment of a disorder or symptom associated with a tumor.
The myeloid-derived CAR cells disclosed herein and pharmaceutical compositions comprising the same are particularly useful for the treatment of disease or disorder in a subject as the myeloid- derived CAR cell compositions are highly pure myeloid cell compositions. The high purity myeloid cell compositions disclosed herein provide for a more stable, safer product. In particular, the myeloid- derived CAR cell compositions disclosed herein comprise high levels of pure myeloid-derived cells due to the manufacturing methods and compositions disclosed herein.
In some instances, the myeloid-derived CAR cells disclosed herein are generated from a hematopoietic stem cell isolated from a subject, wherein said subject is in need of a treatment or medicament described herein. In such instances, the myeloid-derived CAR cells disclosed herein can be autologous. In some instances, the myeloid-derived CAR cells disclosed herein for use in a treatment or manufacture of a medicament are allogeneic. In some instances, the myeloid-derived CAR cells disclosed herein for use in a treatment or manufacture of a medicament are xenogeneic.
Cancers include tumors that are not vascularized, or not yet substantially vascularized, as well as vascularized tumors. The cancers may comprise non-solid tumors (such as hematological tumors, for example, leukemias and lymphomas) or may comprise solid tumors. Types of cancers to be treated with CAR-modified cells of the disclosure include carcinoma, blastoma, and sarcoma, and certain leukemia or lymphoid malignancies, benign and malignant tumors, and malignancies e.g., sarcomas, carcinomas, and melanomas. Adult tumors/cancers and pediatric tumors/cancers are also included.
Solid tumors are abnormal masses of tissue that usually do not contain cysts or liquid areas. Solid tumors can be benign or malignant. Different types of solid tumors are named for the type of cells that form them (such as sarcomas, carcinomas, and lymphomas).
Examples of solid tumors, such as sarcomas and carcinomas, include fibrosarcoma, myxosarcoma, liposarcoma, chondrosarcoma, osteosarcoma, and other sarcomas, synovioma, mesothelioma, Ewing's tumor, leiomyosarcoma, rhabdomyosarcoma, colon carcinoma, lymphoid malignancy, pancreatic cancer, breast cancer, lung cancers, ovarian cancer, prostate cancer, hepatocellular carcinoma, squamous cell carcinoma, basal cell carcinoma, adenocarcinoma, sweat gland carcinoma, medullary thyroid carcinoma, papillary thyroid carcinoma, pheochromocytomas sebaceous gland carcinoma, papillary carcinoma, papillary adenocarcinomas, medullary carcinoma, bronchogenic carcinoma, renal cell carcinoma, hepatoma, bile duct carcinoma, choriocarcinoma, Wilms' tumor, cervical cancer, testicular tumor, seminoma, bladder carcinoma, melanoma, and CNS tumors (such as a glioma (such as brainstem glioma and mixed gliomas), glioblastoma (also known as glioblastoma multiforme) astrocytoma, CNS lymphoma, germinoma, medulloblastoma, Schwannoma craniopharyogioma, ependymoma, pineal oma, hemangioblastoma, acoustic neuroma, oligodendroglioma, menangioma, neuroblastoma, retinoblastoma and brain metastases).
Hematologic cancers are cancers of the blood or bone marrow. Examples of hematological (or hematogenous) cancers include leukemias, including acute leukemias (such as acute lymphocytic leukemia, acute myelocytic leukemia, acute myelogenous leukemia and myeloblastic, promyelocytic, myelomonocytic, monocytic and erythroleukemia), chronic leukemias (such as chronic myelocytic (granulocytic) leukemia, chronic myelogenous leukemia, and chronic lymphocytic leukemia), polycythemia vera, lymphoma, Hodgkin's lymphoma, non-Hodgkin's lymphoma (indolent and high grade forms), multiple myeloma, Waldenstrom's macroglobulinemia, heavy chain disease, myelodysplastic syndrome, hairy cell leukemia and myelodysplasia.
Treatment can result in a reduction in tumor size. In some embodiments, the tumor size/volume in a subject treated with a composition comprising myeloid-derived CAR cells described herein is reduced by at least 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, or 90% relative to untreated tumors. In some embodiments, treatment results in an increased survival rate of the subject. In some
embodiments, the survival rate increases by at least 5%, 10%, 15%, 20%, 25%, 30%, 35%, 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, or 99% relative to untreated subjects.
Treatment can result in a reduction the number of cancer cells present in the subject. In some embodiments, the number of cancer cells in subjects treated with a composition comprising myeloid- derived CAR cells described herein is reduced by at least about 2-fold, 3-fold, 4-fold, 5-fold, 6-fold, 7- fold, 8-fold, 9-fold, or 10-fold relative to untreated subjects.
In some embodiments, the disclosure provides for treating an infection in a subject in need thereof, comprising administering to the subject an effective amount of a genetically modified myeloid-derived cell described herein. The pathogen recognized by the CAR may be essentially any kind of pathogen, but in some embodiments the pathogen is a fungus, bacteria, or virus. Exemplary viral pathogens include those of the families of Adenoviridae, Epstein-Barr virus (EB V), Cytomegalovirus (CMV), Respiratory Syncytial Virus (RSV), JC virus, BK virus, HSV, HHV family of viruses, Picornaviridae, Herpesviridae, Hepadnaviridae, Flaviviridae, Retroviridae, Orthomyxoviridae, Paramyxoviridae, Papovaviridae, Polyomavirus, Rhabdoviridae, and Togaviridae. Exemplary pathogenic viruses cause smallpox, influenza, mumps, measles, chicken pox, ebola, and rubella. Exemplary pathogenic fungi include Candida, Aspergillus, Cryptococcus, Histoplasma, Pneumocystis, and Stachybotrys. Exemplary pathogenic bacteria include Streptococcus, Pseudomonas, Shigella, Campylobacter, Staphylococcus, Helicobacter, E. coli, Rickettsia, Bacillus, Bordetella, Chlamydia, Spirochetes, and Salmonella. In some embodiments the pathogen receptor Dectin- 1 may be used to generate a CAR that recognizes the carbohydrate structure on the cell wall of fungi such as Aspergillus. In another embodiment, CARs can be made based on an antibody recognizing viral determinants (e.g., the glycoproteins from CMV and Ebola) to interrupt viral infections and pathology.
The disclosure provides a method of providing an immune response against a target in a subject in need thereof, the method comprising: transfecting a stem cell, such as a hematopoietic stem cell, obtained from the subject with a nucleic acid molecule comprising a polynucleotide sequence encoding a chimeric antigen receptor (CAR), wherein the CAR comprises: an antigen binding domain; a transmembrane domain; an intracellular signaling domain; and an intracellular co-stimulatory signaling domain having an amino acid sequence having at least at least 80%, 81%, 82%, 83%, 84%, 85%, 86%, 87%, 88%, 89%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or 100% sequence identity sequence identity to a cytoplasmic domain of a herpes virus entry mediator (HVEM) protein set forth as any one of SEQ ID NOs: 1, 2, or 3, or a functional fragment or variant thereof that retains costimulatory activity, to obtain genetically modified myeloid-derived cells expressing the CAR; culturing the genetically modified myeloid-derived cells expressing the CAR; and administering a composition comprising the cultured genetically modified myeloid-derived cells expressing the CAR
to the subject, thereby providing an immune response against the target in the subject. In some embodiments, the subject has cancer and/or an infection. In some embodiments, the target is a cancer cell or an infectious agent. The disclosure further includes use of the CAR-modified myeloid-derived cells described herein in the manufacture of a medicament for the treatment of an immune response in a subject in need thereof.
The disclosure further provides a method of treating a subject by engulfment and/or trogocytosis of a target cell in the subject, the method comprising administering to the subject a therapeutically effective amount of a pharmaceutical composition comprising a genetically modified myeloid-derived cell described herein. In particular, methods of the disclosure can be used to treat individuals who have, who are suspected of having, or who may be at high risk for developing one or more health conditions or disorders for which trogocytosis and/or engulfment of a target cell is known or expected to have a therapeutic effect. Examples of target cells to be recognized and eliminated by the CAR-modified myeloid-derived cells of the disclosure include tumor cells, bacteria, virus-infected cells, viral particles, senescent cells and other cells identifiable to a person skilled in the art. In some embodiments, target cells also include neurons that are non-functional or dying due to accumulation of abnormal forms of Tau or beta-amyloid.
Any chimeric antigen receptor (CAR) can be used with the myeloid-derived CAR cells disclosed herein.
The myeloid-derived CAR cells described herein possess targeted effector activity. In some embodiments, the myeloid-derived CAR cells have targeted effector activity directed against an antigen on a target cell, such as through specific binding to an antigen binding domain of a CAR. In another embodiment, the targeted effector activity includes, but is not limited to, phagocytosis, targeted cellular cytotoxicity, antigen presentation, and cytokine secretion.
In another embodiment, the myeloid-derived CAR cells described herein have the capacity to deliver an agent, a biological agent or a therapeutic agent to the target. The cell may be modified or engineered to deliver an agent to a target, wherein the agent is selected from the group consisting of a nucleic acid, an antibiotic, an anti -inflammatory agent, an antibody or antibody fragments thereof, a growth factor, a cytokine, an enzyme, a protein, a peptide, a fusion protein, a synthetic molecule, an organic molecule, a carbohydrate or the like, a lipid, a hormone, a microsome, a derivative or a variation thereof, and any combination thereof. As a non-limiting example, a macrophage modified with a CAR of the disclosure is capable of secreting an agent, such as a cytokine or antibody, to aid in macrophage function. Antibodies, such as anti-CD47/antiSIRPa monoclonal antibody, may also aid in macrophage function. In yet another example, the macrophage modified with a CAR of the disclosure is engineered to encode a siRNA that aids macrophage function by downregulating
inhibitory genes (e.g., SIRPa). As another example, the CAR-modified myeloid-derived cell is engineered to express a dominant negative (or otherwise mutated) version of a receptor or enzyme that aids in macrophage function.
The myeloid-derived cell (e.g., myeloid progenitor cell or derivative thereof) can be modified with multiple genes, wherein at least one gene encodes a CAR and at least one other gene comprises a genetic element that enhances myeloid-derived CAR cell function. In some embodiments, the myeloid- derived cell is modified with multiple genes, wherein at least one gene encodes a CAR and at least one other gene aids or reprograms the function of other immune cells (such as T cells within the tumor microenvironment). Further, the CAR-modified myeloid-derived cells can be administered to an animal, preferably a mammal, even more preferably a human, to suppress an immune reaction, such as those common to autoimmune diseases such as diabetes, psoriasis, rheumatoid arthritis, multiple sclerosis, graft versus host disease (GVHD), transplant rejection, and the like. In addition, the CAR- modified myeloid-derived cells of the present disclosure can be used for the treatment of any condition in which a diminished or otherwise inhibited immune response, especially a cell-mediated immune response, is desirable to treat or alleviate a disease. Therefore, in some embodiments, methods of the disclosure include treating a condition, such as an autoimmune disease, in a subject, comprising administering to the subject a therapeutically effective amount of a pharmaceutical composition comprising the CAR-modified myeloid-derived cells described herein.
Examples of autoimmune disease include Acquired Immunodeficiency Syndrome (AIDS, which is a viral disease with an autoimmune component), alopecia areata, ankylosing spondylitis, antiphospholipid syndrome, autoimmune Addison's disease, autoimmune hemolytic anemia, autoimmune hepatitis, autoimmune inner ear disease (AIED), autoimmune lymphoproliferative syndrome (ALPS), autoimmune thrombocytopenic purpura (ATP), Behcet's disease, cardiomyopathy, celiac sprue-dermatitis hepetiformis; chronic fatigue immune dysfunction syndrome (CFIDS), chronic inflammatory demyelinating polyneuropathy (CIPD), cicatricial pemphigoid, cold agglutinin disease, crest syndrome, Crohn's disease, Degos' disease, dermatomyositis- juvenile, discoid lupus, essential mixed cryoglobulinemia, fibromyalgia-fibromyositis, Graves' disease, Guillain-Barre syndrome, Hashimoto's thyroiditis, idiopathic pulmonary fibrosis, idiopathic thrombocytopenia purpura (ITP), IgA nephropathy, insulin-dependent diabetes mellitus, juvenile chronic arthritis (Still's disease), juvenile rheumatoid arthritis, Meniere's disease, mixed connective tissue disease, multiple sclerosis, myasthenia gravis, pernacious anemia, polyarteritis nodosa, polychondritis, polyglandular syndromes, polymyalgia rheumatica, polymyositis and dermatomyositis, primary agammaglobulinemia, primary biliary cirrhosis, psoriasis, psoriatic arthritis, Raynaud's phenomena, Reiter's syndrome, rheumatic fever, rheumatoid arthritis, sarcoidosis, scleroderma (progressive systemic sclerosis (PSS), also known
as systemic sclerosis (SS)), Sjogren's syndrome, stiff- man syndrome, systemic lupus erythematosus, Takayasu arteritis, temporal arteritis/giant cell arteritis, ulcerative colitis, uveitis, vitiligo, and Wegener's granulomatosis. A composition comprising the CAR-modified phagocytic cells can also be used to treat inflammatory disorders. Examples of inflammatory disorders include chronic and acute inflammatory disorders. Examples of inflammatory disorders include Alzheimer's disease, asthma, atopic allergy, allergy, atherosclerosis, bronchial asthma, eczema, glomerulonephritis, GVHD, hemolytic anemias, osteoarthritis, sepsis, stroke, transplantation of tissue and organs, vasculitis, diabetic retinopathy, and ventilator induced lung injury.
In embodiments of methods disclosed herein, the CAR comprises an intracellular costimulatory signaling domain having an amino acid sequence having at least at least 80%, 81%, 82%, 83%, 84%, 85%, 86%, 87%, 88%, 89%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or 100% sequence identity sequence identity to a cytoplasmic domain of a herpes virus entry mediator (HVEM) protein set forth as any one of SEQ ID NOs: 1, 2 or 3, or a functional fragment or variant thereof that retains co-stimulatory activity.
CAR-modified myeloid-derived cells of the disclosure can be administered in dosages and by routes and at times to be determined in appropriate pre-clinical and clinical experimentation and trials. Cell compositions may be administered multiple times at dosages within these ranges.
Administration of CAR-modified myeloid-derived cells of the disclosure may be combined with other methods useful to treat the desired disease or condition as determined by those of skill in the art.
CAR-modified myeloid-derived cells of the disclosure to be administered may be autologous, allogeneic or xenogeneic with respect to the subject undergoing therapy.
The administration of CAR-modified myeloid-derived cells of the disclosure to a subject may be carried out in any convenient manner known to those of skill in the art, including by aerosol inhalation, injection, ingestion, transfusion, implantation, and/or transplantation. The compositions described herein may be administered to a subject transarterially, subcutaneously, intradermally, intratum orally, intranodally, intramedullary, intramuscularly, by intravenous (iv) injection, or intraperitoneally. In some embodiments, modified myeloid-derived cells of the disclosure are injected directly into a target region, a local disease site in the subject, a site of inflammation, a site of infection, a lymph node, an organ and/or a tumor of the subject.
It will be readily apparent to those skilled in the art that other suitable modifications and adaptations of the methods of the invention described herein are obvious and may be made using suitable equivalents without departing from the scope of the invention or the embodiments disclosed herein. Having now described the invention in detail, the same will be more clearly understood by
reference to the following examples, which are included for purposes of illustration only and are not intended to be limiting.
EXAMPLES
EXAMPLE 1: Isolation and selection of CD34+ precursor cells
A G-CSF mobilized leukopack from a healthy donor is positively selected for CD34+ cells using CD34+ Miltenyi microbeads on the CliniMACs instrument. The resulting positive-beaded eluted fraction is cryopreserved in 10% DMSO CryoStor CS10 at a concentration of 5-10e6 cells/mL and stored in liquid nitrogen.
Vials of >98% CD34+ cells are removed from liquid nitrogen and thawed in water bath for 90 seconds until a small ice crystal remains. Pre-warmed Sigma Aldrich Stemline® II media is added dropwise to each vial, and the resulting cell solution is then added dropwise back into a pre-warmed aliquot of basal Stemline® II media. The cells are centrifuged to remove DMSO and resuspended in expansion media. The cells are plated on Ultra-low attachment cell culture vessels at 0.125e6 cells/mL and incubated at 37C 5% CO2 for approximately 24 hours.
EXAMPLE 2: Expansion and differentiation of CD34+ cells into myeloid-derived cells
Clinical experience with mobilization regimens in cancer patients is extensive. 50-500 x 106 CD34+ cells are regularly harvested from these collections. The methods described herein can expand these cells >200 fold and generate differentiated monocytes in <14 days. Following isolation of CD34+ cells, as described in Example 1, >95% purity of CD34+ cells from a mobilized leukopak can be achieved. Optimized expansion methods using precursor media as described herein can reach between about300- to 1,000-fold increases in total cell numbers, as shown in FIGS. 4A-4C. In particular, maintaining CD34+ precursor cells in Myel 1 precursor medium for up to 8 days results in an about 250-fold to 300-fold-increase in cell number. Thus, the precursor media described herein are particularly effective at generating a cell population when precursor cells are incubated (i.e., contacted with, and maintained in, precursor media). By day 12 in Myel 1 precursor media, the number of cells increased >350-fold.
FIGS. 4A-4C show the results of three experiments examining the cumulative fold expansion of a population of CD34+ hematopoietic stem cells maintained in five precursor medias described herein (HSC1; Myel 1; Myel 1.1; Myel 4; and Myel 4.1). The cumulative expansion of HSCs occurred at the fastest rate (up to 300-fold within 7-8 days) in “Myel 4.1” and “Myel 1” precursor media, compared to precursor media comprising different precursor cytokine mixtures (“HSC 1”, “Myel 1.1” and “Myel 4”). When cell expansion was maintained for out to 12 days (FIG. 4C), a
cumulative fold expansion of 1000 was observed in “Myel 4.1” precursor media, and cumulative fold expansion in “Myel 1” precursor media was greater than 350-fold. These results demonstrate the surprising and superior effect of the precursor media compositions disclosed herein to promoter expansion of precursor cells to generate a cell population comprising precursor cells.
Over the course of expansion and differentiation (e.g., in precursor media only or in precursor media followed by myeloid media), cells will upregulate myeloid markers. The impact on myeloid marker expression on a starting population of hematopoietic stem cells (HSCs) following incubation in precursor media (“Myel 4.1”), followed by subsequent incubation in myeloid media was examined. As shown in FIG. 5, about 76.7% of the cell population was CD14+CD1 lb+, as determined by flow cytometry analysis, after incubation of a starting population ofHSCs in precursor media (“Myel 4.1”) for 7 days followed by differentiation in myeloid media for 9 days.
In some instances, it may not be beneficial or necessary to utilize multiple medium compositions to generate a differentiated cell or to expand and/or differentiate a plurality of cells within a cell population. Accordingly, the effect on precursor cell differentiation that were maintained in precursor media was examined.
Additionally, CD14+expression levels were examined on a population of cells generated following expansion and differentiation of a population ofCD34+ cells in three precursor media. CD34+ cells were contacted with one of three different precursor media, and maintained in each respective media for 8 days to generate cell populations. Following incubation in precursor media, cell populations were contacted with myeloid media. Cells were maintained in myeloid media for up to 3 days. The amount of cells within each population expressing CD14 on the cell surface was analyzed on day 0 (prior to myeloid media incubation) and after 1 day, after 2 days, and after 3 days in myeloid media, as shown in FIG. 6. Following each day, the percentage of cells within the population that were CD14+ was examined. As shown in FIG. 6, about 20% of cells within the cell population were CD14+ after being maintained in Meyl 1 and Myel 3 media for 8 days. Additionally, 80% of the cell population was CD 14+ after three days in myeloid media, following 8 days in precursor media “Myel 1” and “Myel 3”. After 1 day of myeloid media incubation,, about 60% of cells within the population were CD14+ after incubation in Myel 1 precursor media. These results indicate that maintaining a population of CD34+ cells in Myel 1 precursor media promotes high levels of differentiation of such precursor cells (i.e., hematopoietic stem cells) after 8 days. Additionally, when these cells are then incubated in myeloid media, the cells continue to differentiate such that an increased number of cells within the population are CD14+ each day after being maintained in myeloid media.
These results demonstrate that the precursor media disclosed herein (e.g., Myel 1 media) promotes the differentiation of CD34+ precursor cells into myeloid-lineage cells (i.e., CD14+ cells). These data also demonstrate that incubation in myeloid media following precursor media increases the percentage of the cell population that are myeloid cells. The percentage of myeloid- derived cells within the population increases with increasing time in myeloid media.
As shown in FIG. 7, early HSC-monocytes retain capacity to quickly migrate across a HUVEC monolayer when compared to later monocytes. In particular, myeloid-derived cells generated from HSCs demonstrate chemoattractant-dependent migration through human umbilical venular endothelial cells (HUVEC) endothelial monolayer. HSC-M cell lines generated via HSC- to-Monocyte expansion and differentiation processes described herein (“HSC-Monocytes”; FIG. 7) were collected, counted, and re-plated at 10k cells per well in the top chamber of a Sartorius ClearView 96-well chemotaxis plate in 0.06 mL X-Vivo-15/GlutaMAX (Lonza) + 100 ng/mL GM- CSF + 0.5% HLFBS. During development, some HSC-M cell lines ('Stim. Monocytes'; FIG. 7) were conditioned with added pro-inflammatory cytokines to force macrophage-like phenotypes. After allowing cells to settle for Ih at 25C, 0.2 mL of the same media +/- C5a (Complement component 5a) (100 nM) was added to the bottom chamber of each well. Incucyte S3 imaging commenced immediately, imaging every Ih in the phase bottom imaging plane, with in-focus objects being classified as migrated cells via Incucyte imaging software. One day prior to migration, each chamber of the 96-well chemotaxis plate was treated with Cultrex matrigel (50 ug/mL + 10% HLFBS), incubating for 30 min at 37C, then 30 min at 25C. Once incubation was complete, all chambers were washed with 0.1 mL IX D-PBS and 10k HUVEC endothelial cells were plated into top chambers in 60 uL of VCBM + 2% HLFBS, with 0.2 mL IX D-PBS added to each bottom chamber. Incubation for 24h at 37C was then initiated. After overnight incubation, HUVEC endothelial monolayers were washed with 0.1 mL IX D-PBS, and HSC-M cell lines were plated for migration assessment.
EXAMPLE 3: The optimal timeline of myeloid expansion and monocyte differentiation in precursor media (Myel 1)
The expansion period is a crucial step to generate a high yield of myeloid cells derived from mobilized HSCs. Table 1 depicts the results of astudy investigating the optimal differentiation timeline for myeloid lineage specification derived from HSCs at various induction days (Day 0, 4, 6, 8, 11). HSCs were directly differentiated using specific cytokine combinations (precursor Myel 1 medium) to promote mature myeloid lineage development according to the induction days. Under the Myel 1 medium condition, HSC-derived differentiating cells showed a significant decrease in
CD34 HSC marker expression, from 98.3% to 0.8%, during differentiation. Concurrently, there was an increase in myeloid marker expression, with peak differentiation observed between days 9-11. This peak corresponded to the highest expression levels, with 84.6% of cells positive for CD 15+ and 76.2% positive for CD1 lb+ at day 11.
For the myeloid expansion step in Myel medium, a 9-10 day period of cell expansion is the most efficient for mass cell growth and shows the highest expression of myeloid markers, specifically 88% for CD15 and 75.3% for CD1 lb in Table 2. Following incubation in precursor media, cells are cultured in monocyte differentiation medium (i.e., myeloid media) at various induction timepoints to investigate the optimal period for monocyte generation following myeloid expansion. Table 2 shows that CD14 marker expression gradually increases, with the percentage of CD14 positive cells rising from day 0 to day 7 following induction of differentiation. After 7 days of differentiation, the CD 14 expression reaches its peak at 88.7%. The overall data indicates that a 9-10 day expansion period and a 5-7 day differentiation period are optimal for producing monocytes derived from mobilized HSCs.
Table 3 shows the results of an additional study that was conducted to examine the effect of Myel 1 precursor media and subsequent myeloid media incubation on precursor cell expansion (i.e., cumulative fold expansion of the cell population) and differentiation into myeloid lineage cells.
EXAMPLE 4: Generating and expanding a genetically modified precursor CAR cell in a precursor media
An ultra-low attachment plate is pre-treated with Retronectin (25ug/mL) and PBS solution and incubated >2 hours (the plate is blocked with 10% BSA and washed with DPBS prior to use). The appropriate amount of viral supernatant at an MOI of 10 per le6 cells is calculated and split in half. During the first round of transduction, half the viral supernatant is added to the Retronectin plate and diluted with precursor media. The plate is centrifuged 2000xg for 60 minutes at 32°C. After the initial spin, le6 cells are added to each transduction well and the plate is centrifuged for 10 minutes at 450xg and 32°C. Once the cells are settled, the remaining half of the viral stock is added to each appropriate virus well and centrifuged for 450xg for 60 minutes at 32°C. After the last spin, 300uL of HSC Media 1 is added on top of each transduction well and the plate is incubated for 24 hours at 37°C 5% CO2.
After 24 hours, the transduction plate is removed from the incubator and cells are collected and washed from each appropriate well. Centrifugation removes excess viral supernatant from the cell suspension, and cells are resuspended in fresh precursor media comprising a precursor cytokine mixture. Cells are then counted and re-seeded into ultra-low attachment vessels at 0.125e6 cells/mL. Cells are resuspended in precursor media comprising the same mixture of precursor
cytokines and mixed approximately every other day of expansion and counted every 2-3 days for re-seeding at approximately 0.125-0.15e6 cells/mL. In between count and resuspension, cells are incubated at 37°C 5% CO2.
72 hours post-transduction, cells are sampled for flow cytometry to measure % of GFP+ expression to approximate CAR integration. At this time, ImM puromycin is added to the HSC Media 1 in culture to remove cells not expressing CAR (and thus, GFP). Cells are placed back in incubator at 37°C 5% CO2.
Transduction and differentiation efficiency was examined following introduction of a chimeric antigen receptor (CAR) into hematopoietic stem cells (HSCs), followed by culture in myeloid media. HSCs were transduced with a CAR as described above, and then cultured in HSC 1 media for 2 weeks, followed by culture in myeloid media for 5 days. CAR (GFP+) and CD14 expression was analyzed by flow cytometry following incubation in myeloid media. As shown in FIG. 3A, following CAR transduction with a lentivirus, and incubation in precursor media and myeloid media, 52.6% of hematopoietic cells express the CAR. These results indicate very high transduction efficiency. Within the population of cells expressing a CAR, 85.9% are CD14+, as shown in FIG. 3B.
EXAMPLE 5: Differentiating stem cells and genetically modified precursor CAR cells in a myeloid media
After 7-9 days of expansion, cells are removed from precursor media and exchanged into myeloid media comprising a myeloid cytokine mixture. Cells are then plated at approximately l-2e6 cells/mL in a spinner flask bioreactor (speed 25rpm) or plated in a low-attachment vessel pre-treated with StemCell Technologies Anti -Adherence solution. Cells are manually mixed every 1-2 days to prevent monocytes from sticking to plastic and differentiating, and a full media exchange is performed every 4-5 days. Samples for flow cytometry are taken every 3 days to measure CD14+ expression to determine rate of HSC differentiation into monocytes. In between sample stages, cells are incubated at 37C 5% CO2.
EXAMPLE 6: Duration of transduced gene expression in HSC-derived monocytes
Astudy was conducted to investigate the duration of transduced gene expression in mobilized HSCs using a procedure outlined in the schematic of FIG. IB. A nucleic acid construct encoding for an FMC63 chimeric antigen receptor (CAR) comprising an HVEM co-stimulatory intracellular signaling domain (M83) (“CAR (M83)”; FIGS. 2A-2D and FIG. 21) and green fluorescence protein (GFP) gene were operably linked to an elongation factor-1 (EFl) promoter was transduced into mobilized HSC cells
using VSV-G pseudotyped lentiviral particles. Cells were incubated at 37°C 5% CO2 in HSC Media 1 for 72 hours. 72 hours post-transduction, cells were sampled for flow cytometry to measure % of GFP+ expression to approximate CAR integration (Day 6; FIG. 2A and FIG. 2E). At this time, ImM puromycin was added to the HSC Media 1 in culture to remove cells not expressing CAR (and thus, GFP). Cells were placed back in incubator at 37°C 5% CO2. After 2 days of selection with puromycin (Day 8), the majority of untransduced cells were eliminated and FACS analysis indicated high expression levels of > 82.2% FMC63 and > 84.05% GFP in the remaining cells as depicted in FIG. 2B and FIG. 2F, respectively. As depicted in FIGS. 2A-2H,, transduced cells consistently showed expression of FMC63 (FIGS. 2A-2D) and GFP (FIGS. 2E-2H) at multiple time points analyzed during expansion and differentiation in precursor media. In particular, as shown in FIGS. 2K-2N, by Day 14, HSC-derived monocytes exhibited high expression levels of myeloid and monocyte markers, with 96.9% positive for CD13 (FIG. 2K), 45.9% for CD15 (FIG. 2L), 93.3% for CD1 lb (FIG. 2M), and 77.2% for CD14 (FIG. 2N) . After differentiation of transduced HSC-derived monocytes into Ml macrophages, using a similar protocol as described in Example 8, the expression of transduced proteins was consistently maintained. As depicted in FIGS. 2I-2J, flow cytometry analysis showed that 92.9% of the cells were positive for FMC63 (FIG. 21) and 84.3% were positive for GFP (FIG. 2J) in Ml macrophages. These findings demonstrate successful gene transduction into HSCs, resulting in abundant expression of transduced proteins (i.e., a CAR) throughout myeloid expansion, monocyte differentiation, and polarization stages. This efficient gene transduction, in particular with a chimeric antigen receptor, enhances the potency of transduced monocytes against tumors.
EXAMPLE 7: HSC-derived monocytes exhibit the ability to phagocytose
To investigate the key functional phagocytic capacity of HSC-derived monocytes generated in Myel 1 media, 50,000 HSC-derived monocytes generated in Myel 1 media using the methods disclosed herein were seeded and co-cultured with pHrodo-labeled polystyrene beads (Spherotech, Protein G coupled, 7.2 pm diameter) in a NUNC 96-well TC plate (Thermo Scientific) at a 1 : 1 effector -to- target (E:T) ratio (50,000 HSC-derived monocytes: 50,000 beads) The polystyrene beads were first coupled to pHrodo-conjugated mAb clones (a-Smurf2, Cell Signaling Technology), modified via carbohydrate digestion (SiteClick Modification Kit, Invitrogen) and fluorophore click chemistry processes (SiteClick pHrodo Kit), 24 hours prior to the HSC-derived monocyte co-culture, following the manufacturer’s protocol. The co-culture was performed in 0.2 mL RPMI-1640 GlutaMAX + 10% HI-FBS media (Gibco). Negative control conditions were pre-treated with 10 pM Cytochalasin D (Sigma Aldrich) at 37°C for 10 minutes prior to bead co-culture. Upon co-culture and multi-channel mixing, the TC plates were centrifuged at 25 x g, 21 °C, for 1 minute, and whole-well images were taken immediately using
the IncuCyte S3 system (Sartorius) every hour in Phase/Red channels for 6 hours. Quantified red objects were analyzed using in-house analyses through IncuCyte S3 software (v208B, Sartorius). Identified red events represent individual polystyrene beads phagocytosed by HSC-derived monocytes, fluorescing red due to pHrodo alterations when contained in cytosol-localized acidic phagolysosomes in FIG. 8A. The results demonstrated rapid bead intake as early as 1 hour post-co-culture, plateauing after 5-6 hours of co-culture. HSC-monocytes pre-treated with a G-actin hydrolyzing agent (Cytochalasin D), which inhibits F-actin polymerization and membrane rearrangement essential for phagocytic function, showed a complete disruption of phagocytic efficacy across all timepoints in FIG. 8B. Together, this data verifies the key functional characteristic of phagocytosis in HSC-derived monocytes.
EXAMPLE 8: Macrophages derived from HSC-monocytes show potent phagocytic and killing capacity
To investigate the phenotypic, functional, and plasticity features of HSC-derived monocytes, 200,000 HSC-derived monocytes from 3 different donors were seeded in 48-well Corning cell-treated plates. The monocytes were pre-differentiated into M0 macrophages over 8 days using 500 pl per well of preconditioning medium (X-vivol5/5% FBS with 100 ng/ml M-CSF). After 8 days, the preconditioning medium was removed and replaced gently with 400 pl per well of a premixed medium (Ml activation medium: X-vivol5/5% FBS with 100 ng/ml GM-CSF, 50 ng/ml IFN-gamma, 25 ng/ml LPS) along with tumor cells to polarize the pre-macrophages (M0) into the Ml state (without H1299 cells) at an E:T ratio of 3: 1. The co-culture was monitored in real-time using an IncuCyte live imaging system for 3 days. In summary, the study defined in vitro conditions for differentiating HSC-derived monocytes into macrophages and subsequently polarizing them into Ml subsets. The results showed that HSC-derived macrophages exhibited robust phagocytic and killing capabilities compared to the tumor- only condition, highlighting distinct differences between the M0 and Ml states in FIG. 9.
The section headings used herein are for organizational purposes only and are not to be construed as limiting the subject matter described in any way.
It is appreciated that certain features of the disclosure, which are, for clarity, described in the context of separate embodiments, may also be provided in combination in a single embodiment. Conversely, various features of the disclosure, which are, for brevity, described in the context of a single embodiment, may also be provided separately or in any suitable sub-combination or as suitable in any other described embodiment of the disclosure. Certain features described in the context of various embodiments are not to be considered essential features of those embodiments, unless the embodiment is inoperative without those elements.
While various aspects of the invention are described herein, it is not intended that the invention be limited by any particular aspect. On the contrary, the invention encompasses various alternatives, modifications, and equivalents, as will be appreciated by those of skill in the art. Furthermore, where feasible, any of the aspects disclosed herein may be combined with each other (e.g., the feature according to one aspect may be added to the features of another aspect or replace an equivalent feature of another aspect) or with features that are well known in the art, unless indicated otherwise by context.
Claims
1. A method of manufacturing a myeloid-derived cell, said method comprising the step of: contacting a precursor cell with a precursor media comprising a precursor cytokine mixture, whereby the number of precursor cell(s) increases to generate a cell population comprising a plurality of precursor cells, wherein said precursor cell is a stem cell.
2. The method of claim 1, wherein said method comprises the step of: contacting said precursor cell with a nucleic acid molecule comprising a polynucleotide sequence encoding a chimeric antigen receptor (CAR), whereby said nucleic acid is introduced into said precursor cell to generate a precursor CAR cell comprising a chimeric antigen receptor (CAR).
3. The method of claim 2, wherein said precursor cell is contacted with said nucleic acid molecule prior to, or concurrently with, being contacted with said precursor media.
4. The method of claim 3, wherein said method comprises the steps of: a) contacting said precursor cell with a nucleic acid molecule comprising a polynucleotide sequence encoding a chimeric antigen receptor (CAR), whereby said nucleic acid is introduced into said precursor cell to generate a precursor CAR cell comprising a chimeric antigen receptor (CAR), and b) contacting said precursor CAR cell with a precursor media comprising a precursor cytokine mixture, whereby the number of precursor CAR cell(s) increases to generate a cell population comprising a plurality of precursor CAR cells, wherein step a) occurs prior to step b).
5. The method of any one of claims 1-4, wherein the method further comprises the step of: c) contacting said cell population comprising a plurality of precursor cells and/or said cell population comprising a plurality of precursor CAR cells with a myeloid media comprising a myeloid cytokine mixture, whereby
said precursor cell(s) differentiates into a myeloid-lineage cell to generate a myeloid- derived cell and/or a mixed cell population comprising a plurality of myeloid-derived cells and/or a plurality of precursor cells, and/or said precursor CAR cell(s) differentiates into a myeloid-lineage cell to generate a myeloid- derived CAR cell and/or a mixed cell population comprising a plurality of myeloid-derived CAR cells and/or a plurality of said precursor CAR cells.
6. The method of any one of claims 1-4, whereby upon contact of said precursor cell(s) with said precursor media, said precursor cell(s) differentiates into a myeloid-lineage cell to generate a myeloid-derived cell and/or a mixed cell population comprising a plurality of myeloid-derived cells and a plurality of precursor cells, or whereby upon contact of said precursor CAR cell(s) with said precursor media, said precursor CAR cell(s) differentiates into a myeloid-lineage cell to generate a myeloid-derived CAR cell and/or a mixed cell population comprising a plurality of myeloid-derived CAR cells and a plurality of precursor CAR cells.
7. The method of claim 6, wherein the method further comprises the step of: c) contacting said mixed cell population with a myeloid media comprising a myeloid cytokine mixture, whereby upon contact with said myeloid media: a. the ratio of said myeloid-derived cells to said precursor cells in said mixed cell population is increased; and/or b. the ratio of myeloid-derived CAR cells relative to precursor CAR cells in said mixed cell population is increased.
8. The method of claim 5 or 7, wherein the precursor media is removed prior to addition of the myeloid media.
9. The method of any one of claims 1-8, wherein said stem cell is a bone-marrow-derived stem cell.
10. The method of any one of claims 1-9, wherein said stem cell is a hematopoietic stem cell (HSC).
11. The method of any one of claims 1-10, wherein said stem cell is CD34+.
12. The method of any one of claims 1-11, wherein said stem cell is a pluripotent stem cell.
13. The method of any one of claims 1-12, wherein said stem cell is isolated from a subject.
14. The method of claim 13, wherein said subject is a human subject.
15. The method of claim 13 or 14, wherein said isolated stem cell is cryopreserved prior to contacting said cell with a precursor media and/or contacting said cell with a nucleic acid molecule.
16. The method of claim 15, wherein said cryopreserved stem cell is thawed prior to contacting said cell with a precursor media and/or contacting said cell with a nucleic acid molecule.
17. The method of any one of claims 1-16, wherein said stem cell is present within a population of stem cells.
18. The method of claim 17, wherein said population comprises a plurality of stem cell(s).
19. The method of claim 17 or 18, wherein between 80%-99.9% of said population is CD34+ cells.
20. The method of claim 19, wherein at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% of said population is CD34+ cells.
21. The method of any one of claims 2-20, wherein said stem cell is maintained in a culture medium for between 2-144 hours following contact with said nucleic acid molecule.
22. The method of claim 21, wherein while said stem cell is maintained within said culture medium, said polynucleotide encoding is integrated into the stem cell chromosome.
23. The method of claim 21 or 22, wherein a selection agent is added to said culture medium.
24. The method of any one of claims 1-23, wherein said precursor cytokine mixture comprises at least one cytokine selected from the following: stem cells factor (SCF), Flt3 ligand,
thrombopoietin (TPO), Interleukin-3 (IL-3); Granulocyte colony-stimulating factor (G-CSF), Interleukin-6 (IL-6), Granulocyte-macrophage colony-stimulating factor (GM-CSF), and Macrophage colony-stimulating factor (MCSF).
25. The method of any one of claims 1-24, wherein said precursor media comprises G-CSF at a concentration between about 100 ng/mL and 200 ng/mL.
26. The method of any one of claims 1-25, wherein said precursor media comprises G-CSF at a concentration between about 100 ng/mL and 150 ng/mL.
27. The method of any one of claims 1-26, wherein said precursor media comprises G-CSF, GM-CSF, SCF, and TPO.
28. The method of any one of claims 1-27, wherein said precursor media comprises GM-CSF at a concentration between about 75 ng/mL and 125 ng/mL.
29. The method of any one of claims 1-28, wherein said precursor media comprises SCF at a concentration between about 75 ng/mL and 125 ng/mL.
30. The method of any one of claims 1-29, wherein said precursor media comprises TPO at a concentration between about 75 ng/mL and 125 ng/mL.
31. The method of claim 30, wherein said precursor media comprises G-CSF at a concentration of 150 ng/mL, GM-CSF at a concentration of 100 ng/mL, SCF at a concentration of 100 ng/mL, and/or TPO at a concentration of 100 ng/mL.
32. The method of any one of claims 1-31, wherein said myeloid media comprises a myeloid cytokine mixture comprising Macrophage colony-stimulating factor (MCSF), Interleukin-3 (IL-3), and/or Interleukin-6 (IL-6), and/or a combination thereof.
33. The method of any one of claims 1-32, wherein said myeloid media comprises stem cells factor (SCF), Flt3 ligand, thrombopoietin (TPO), Granulocyte colony-stimulating factor (G-CSF), and/or Granulocyte-macrophage colony-stimulating factor (GM-CSF) at a concentration of at most
50 ng/mL, at most 40 ng/mL, at most 30 ng/mL, at most 20 ng/mL, at most 10 ng/mL, at most 5 ng/mL, or below 5 ng/mL.
34. The method of any one of claims 1-33, wherein said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population, is maintained in said precursor media until the number of precursor cell(s), precursor CAR cells, myeloid-derived cells and/or myeloid-derived CAR cells is increased by at least 20-fold, at least 50-fold, at least 75- fold, at least 100-fold, at least 200-fold, at least 300-fold, at least 400-fold, at least 500-fold, or more than 500-fold.
35. The method of any one of claims 1-34, wherein said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population, is maintained in said precursor media until the number of precursor cell(s), precursor CAR cells, myeloid-derived cells and/or myeloid-derived CAR cells is at least 104, at least 105, at least 106, at least 107, at least 108, at least 109, at least 1010, at least 1011, at least 1012, at least 1013, at least 1014, at least 1015, at least 1016, at least 1017, at least 1018, at least 1019, at least IO20, or more than IO20.
36. The method of any one of claims 1-35, wherein said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population, is maintained in said precursor media for at least 2, at least 3, at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, at least 10, at least 11, or at least 12 days.
37. The method of any one of claims 1-36, wherein said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population, is maintained in said precursor media for between 9-11 days.
38. The method of any one of claims 1-37, wherein said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population, is maintained in said precursor media for 10 days.
39. The method of any one of claims 1-38, wherein said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population, is maintained in said precursor media until: a. no more than 99%, no more than 98%, no more than 97%, no more than 96%, no more than 95%, no more than 90%, no more than 80%, no more than 70%, no more than 60%, no more than 50%, no more than 40%, no more than 30%, no more than 20%, no more than 10%, no more than 5%, no more than 1%, or no more than 0.1% of cells within said mixed cell population are CD34+; b. no more than 5%, no more than 4%, no more than 3%, no more than 2%, no more than 1%, or no more than 0.1% of cells within said mixed cell population are CD34+; c. at least 20%, at least 30%, at least 40%, at least 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95% of cells within said mixed cell population are CD15+; d. between about 60%-85% of cells within said mixed cell population are CD15+; e. at least 2%, at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, at least 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95%, or at least 99% of cells within said mixed cell population are CD1 lb+; f. between about 30%-80% of cells within said mixed cell population are CD1 lb+; g. at least 0.1%, at least 0.5%, at least 1%, at least 2%, at least 3%, at least 4%, at least 5%, at least 6%, at least 7%, at least 8%, at least 9%, or at least 10% of cells within said mixed cell population are CD14+; h. between about 1%- 10% of cells within said mixed cell population are CD14+; i. at least 20%, at least 30%, at least 40%, at least 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 90%, or at least 95% of cells within said mixed cell population are CD13+; and/or j. between about 85%-95% of cells within said cells within said mixed cell population are CD13+.
40. The method of any one of claims 1-39, wherein said mixed population is contacted with said myeloid media after contact with said precursor media.
41. The method of claim 40, wherein said mixed population is maintained in said myeloid media until: a. no more than 5%, no more than 4%, no more than 3%, no more than 2%, no more than 1%, no more than 0.9%, no more than 0.8%, no more than 0.7%, no more than 0.6%, no more than 0.5%, no more than 0.4%, no more than 0.3%, no more than 0.2%, no more than 0.1%, or less than 0.1% of cells within said mixed cell population are CD34+; b. at least 70%, at least 75%, at least 80%, at least 90%, or at least 95% of cells within said mixed cell population are CD15+; c. between about 50%-95% of cells within said mixed cell population are CD15+; d. at least 70%, least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or at least 99.5% of cells within said mixed cell population are CD1 lb+; e. between about 75%-99.5% of cells within said mixed cell population are CD1 lb+; f. at least 5%, at least 10%, at least 20%, at least 30%, at least 40%, at least 50%, at least 60%, at least 70%, at least 80%, or at least 90% of cells within said mixed cell population are CD14+; g. between about 5%-90% of cells within said mixed cell population are CD14+; h. at least 85%, at least 90%, at least 95%, or at least 99% of cells within said mixed cell population are CD13+; and/or i. between about 85%-99% of cells within said mixed cell population are CD13+.
42. The method of any one of claims 5 or 7-41, wherein said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population, is maintained in a myeloid media for at least 1, at least 2, at least 3, at least 4, at least 5, at least 6, at least 7, or at least 8 days.
43. The method of any one of claims 5 or 7-42, wherein said mixed cell population comprises at least 20%, at least 40%, at least 60%, least 80%, at least 85%, or at least 90% CD14+ cells after 7 days of being maintained in said myeloid media.
44. The method of any one of claims 1-43, wherein contacting said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population with
said precursor media reduces the ratio of CD34+ cells within said population comprising a plurality of precursor cells, said population comprising a plurality of precursor CAR cells, and/or said mixed cell population in a shorter amount of time compared to a control that is contacted with and/or maintained in a precursor media lacking said precursor cytokine mixture.
45. The method of any one of claims 1-44, wherein contacting said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population with said precursor media increases: a. the number of CD1 lb+ cells; b. the number of CD13+ cells; c. the number of CD 15+ cells; and/or d. the number of CD14+ cells, within said population comprising a plurality of precursor cells, said population comprising a plurality of precursor CAR cells, and/or said mixed cell population in a shorter amount of time compared to a control that is contacted in and/or maintained in a precursor media lacking said precursor cytokine mixture.
46. The method of any one of claims 1-45, wherein contacting said precursor cell(s) and/or said population comprising a plurality of precursor cells, said precursor CAR cell(s) and/or said population comprising a plurality of precursor CAR cells, and/or said mixed cell population with said precursor media increases: a. the ratio of CD1 lb+ to CD1 lb- cells; b. the ratio of CD13+ to CD13- cells; c. the ratio of CD 15+ to CD 15- cells; and/or d. the ratio of CD14+ to CD14- cells within said population comprising a plurality of precursor cells, said population comprising a plurality of precursor CAR cells, and/or said mixed cell population in a shorter amount of time compared to a control that is contacted with and/or maintained in a precursor media lacking said precursor cytokine mixture.
47. The method any one of claims 1-46, wherein at least 40%, at least 50%, at least 60%, at least 70%, at least 80%, at least 90%, or at least 95% of cells within said population comprising a plurality of precursor cells, said population comprising a plurality of precursor CAR cells, and/or
said mixed cell population comprises said CAR, or a functional fragment thereof, on the cell surface.
48. The method of any one of claims 1-47, wherein said myeloid-derived cell is a phagocytic cell.
49. The method of claim 48, wherein said phagocytic cell is a monocyte, macrophage, dendritic cell, neutrophil, or a precursor thereof.
50. The method of claim 48 or 49, wherein said phagocytic cell is a monocyte.
51. The method of any one of claims 1-50, wherein said myeloid-derived cell is a myeloid progenitor cell.
52. The method of any one of claims 1-51, wherein said CAR comprises: an antigen binding domain; a transmembrane domain; and an intracellular signaling domain.
53. The method of claim 52, wherein said CAR further comprises an intracellular costimulatory signaling domain.
54. The method of claim 52 or 53, wherein said CAR comprises an intracellular co-stimulatory signaling domain having an amino acid sequence having at least 90% sequence identity to a cytoplasmic domain of a herpes virus entry mediator (HVEM) protein set forth as any one of SEQ ID NOs: 1, 2, or 3, or a functional fragment or variant thereof that retains co-stimulatory activity.
55. A genetically modified cell comprising a chimeric antigen receptor (CAR), wherein the genetically modified cell is a precursor cell, a myeloid-derived cell, a myeloid-lineage cell, a myeloid progenitor cell, and/or a precursor thereof.
56. The genetically modified cell of claim 55, wherein the precursor cell comprises a bone marrow-derived cell, a blood-derived cell, or a stem cell.
57. The genetically modified cell of claim 56, wherein the stem cell is a hematopoietic stem cell (HSC).
58. The genetically modified cell of claim 55, wherein the cell is a myeloid precursor cell, such as a myeloid progenitor cell, granulo-monocyte progenitor cell, monocyte-dendritic cell progenitor/monoblasts, promonocyte, or myeloblast.
59. The genetically modified cell of any one of claims 55-58, wherein said CAR comprises an intracellular co-stimulatory signaling domain having an amino acid sequence having at least 90% sequence identity to a cytoplasmic domain of a herpes virus entry mediator (HVEM) protein set forth as any one of SEQ ID NOs: 1, 2, or 3, or a functional fragment or variant thereof that retains co-stimulatory activity.
60. A cell population comprising the genetically modified CAR cell of any one of claims 55-59.
61. A cell population generated by the method of any one of claims 1-54.
62. The cell population of claim 60 or 61, wherein at least 40%, at least 50%, at least 60%, at least 70%, at least 80%, at least 90%, at least 95%, or at least 99% of cells within said population expresses said CAR on the cell surface.
63. The cell population of any one of claims 60-62, wherein: a. no more than 5%, no more than 4%, no more than 3%, no more than 2%, no more than 1%, no more than 0.5%, or no more than 0.1% of cells within said population are CD34+; b. at least 50%, at least 55%, at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 86%, at least 87%, at least 88%, at least 89%, or at least 90% of cells within said population are CD14+; c. at least 70%, at least 80%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or at least 99.5%% of cells within said population are CD1 lb+; d. at least 90%, at least 91%, at least 92%, at least 94%, or at least 95% of cells within said population are CD13+; and/or e. at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, or at least 95% of cells within said population are CD15+.
64. A pharmaceutical composition comprising the genetically modified cell of any one of claims 55-59, or the cell population of any one of claims 60-63, and a pharmaceutically acceptable carrier.
65. A method of treating a disease or condition associated with a tumor or cancer in a subject in need thereof, comprising administering to the subject a therapeutically effective amount of the pharmaceutical composition of claim 64.
66. A method of treating a tumor in a subject in need thereof, comprising administering to the subject a therapeutically effective amount of the pharmaceutical composition of claim 64.
67. A method of treating an infection in a subject in need thereof, comprising administering to the subject a therapeutically effective amount of the pharmaceutical composition of claim 64.
68. A method for stimulating an immune response to a target tumor cell or tumor tissue in a subject in need thereof, comprising administering to the subject a therapeutically effective amount of the pharmaceutical composition of claim 64.
69. A method of treating a subject by engulfment and/or trogocytosis of a target cell in the subject, comprising administering to the subject a therapeutically effective amount of the pharmaceutical composition of claim 64.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US202363579469P | 2023-08-29 | 2023-08-29 | |
| US63/579,469 | 2023-08-29 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2025046513A1 true WO2025046513A1 (en) | 2025-03-06 |
Family
ID=92895628
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/IB2024/058426 Pending WO2025046513A1 (en) | 2023-08-29 | 2024-08-29 | Methods of manufacturing myeloid-derived cells from hematopoietic stem cells and compositions and uses thereof |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2025046513A1 (en) |
Citations (77)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5350674A (en) | 1992-09-04 | 1994-09-27 | Becton, Dickinson And Company | Intrinsic factor - horse peroxidase conjugates and a method for increasing the stability thereof |
| US5585362A (en) | 1989-08-22 | 1996-12-17 | The Regents Of The University Of Michigan | Adenovirus vectors for gene therapy |
| US5792632A (en) | 1992-05-05 | 1998-08-11 | Institut Pasteur | Nucleotide sequence encoding the enzyme I-SceI and the uses thereof |
| US5830729A (en) | 1996-04-18 | 1998-11-03 | Institut Pasteur | I Sce I-induced gene replacement and gene conversion in embryonic stem cells |
| US5993434A (en) | 1993-04-01 | 1999-11-30 | Genetronics, Inc. | Method of treatment using electroporation mediated delivery of drugs and genes |
| US6181964B1 (en) | 1997-08-01 | 2001-01-30 | Genetronics, Inc. | Minimally invasive apparatus and method to electroporate drugs and genes into tissue |
| WO2001029058A1 (en) | 1999-10-15 | 2001-04-26 | University Of Massachusetts | Rna interference pathway genes as tools for targeted genetic interference |
| US6238924B1 (en) | 1992-05-05 | 2001-05-29 | Institut Pasteur | Nucleotide sequence encoding the enzyme I-SceI and the uses thereof |
| US6241701B1 (en) | 1997-08-01 | 2001-06-05 | Genetronics, Inc. | Apparatus for electroporation mediated delivery of drugs and genes |
| US6326193B1 (en) | 1999-11-05 | 2001-12-04 | Cambria Biosciences, Llc | Insect control agent |
| WO2001096584A2 (en) | 2000-06-12 | 2001-12-20 | Akkadix Corporation | Materials and methods for the control of nematodes |
| US6468798B1 (en) | 1991-12-17 | 2002-10-22 | The Regents Of The University Of California | Expression of cloned genes in the lung by aerosol and liposome-based delivery |
| US6479626B1 (en) | 1998-03-02 | 2002-11-12 | Massachusetts Institute Of Technology | Poly zinc finger proteins with improved linkers |
| US6534261B1 (en) | 1999-01-12 | 2003-03-18 | Sangamo Biosciences, Inc. | Regulation of endogenous gene expression in cells using zinc finger proteins |
| US20030232410A1 (en) | 2002-03-21 | 2003-12-18 | Monika Liljedahl | Methods and compositions for using zinc finger endonucleases to enhance homologous recombination |
| US6678556B1 (en) | 1998-07-13 | 2004-01-13 | Genetronics, Inc. | Electrical field therapy with reduced histopathological change in muscle |
| US20040014645A1 (en) | 2002-05-28 | 2004-01-22 | Advisys, Inc. | Increased delivery of a nucleic acid construct in vivo by the poly-L-glutamate ("PLG") system |
| US20040059285A1 (en) | 2002-07-04 | 2004-03-25 | Inovio As | Electroporation device and injection apparatus |
| US20040092907A1 (en) | 1997-04-03 | 2004-05-13 | Inovio As | Method for muscle delivery of drugs, nucleic acids and other compounds |
| US6746838B1 (en) | 1997-05-23 | 2004-06-08 | Gendaq Limited | Nucleic acid binding proteins |
| WO2004067753A2 (en) | 2003-01-28 | 2004-08-12 | Cellectis | Use of meganucleases for inducing homologous recombination ex vivo and in toto in vertebrate somatic tissues and application thereof. |
| US6794136B1 (en) | 2000-11-20 | 2004-09-21 | Sangamo Biosciences, Inc. | Iterative optimization in the design of binding proteins |
| US20050052630A1 (en) | 2002-03-07 | 2005-03-10 | Advisys, Inc. | Constant current electroporation device and methods of use |
| US20050070841A1 (en) | 2002-07-04 | 2005-03-31 | Inovio As | Electroporation device and injection apparatus |
| US7013219B2 (en) | 1999-01-12 | 2006-03-14 | Sangamo Biosciences, Inc. | Regulation of endogenous gene expression in cells using zinc finger proteins |
| US7030215B2 (en) | 1999-03-24 | 2006-04-18 | Sangamo Biosciences, Inc. | Position dependent recognition of GNN nucleotide triplets by zinc fingers |
| US7171264B1 (en) | 1999-05-10 | 2007-01-30 | Genetronics, Inc. | Intradermal delivery of active agents by needle-free injection and electroporation |
| US7173116B2 (en) | 2000-03-03 | 2007-02-06 | Genetronics Biomedical Corporation | Nucleic acid formulations for gene delivery and methods of use |
| US20070128708A1 (en) | 2005-12-07 | 2007-06-07 | Genetronics, Inc. | Variable volume electroporation chamber and methods therefore |
| WO2008010009A1 (en) | 2006-07-18 | 2008-01-24 | Cellectis | Meganuclease variants cleaving a dna target sequence from a rag gene and uses thereof |
| WO2008149176A1 (en) | 2007-06-06 | 2008-12-11 | Cellectis | Meganuclease variants cleaving a dna target sequence from the mouse rosa26 locus and uses thereof |
| WO2009013559A1 (en) | 2007-07-23 | 2009-01-29 | Cellectis | Meganuclease variants cleaving a dna target sequence from the human hemoglobin beta gene and uses thereof |
| WO2009059195A2 (en) | 2007-10-31 | 2009-05-07 | Precision Biosciences | Rationally-designed single-chain meganucleases with non-palindromic recognition sequences |
| US20090203140A1 (en) | 2007-09-27 | 2009-08-13 | Sangamo Biosciences, Inc. | Genomic editing in zebrafish using zinc finger nucleases |
| US7585849B2 (en) | 1999-03-24 | 2009-09-08 | Sangamo Biosciences, Inc. | Position dependent recognition of GNN nucleotide triplets by zinc fingers |
| US8119381B2 (en) | 2005-10-18 | 2012-02-21 | Duke University | Rationally-designed meganucleases with altered sequence specificity and DNA-binding affinity |
| US8338157B2 (en) | 2008-03-11 | 2012-12-25 | Precision Biosciences, Inc. | Rationally-designed meganuclease variants of lig-34 and I-crei for maize genome engineering |
| US8440431B2 (en) | 2009-12-10 | 2013-05-14 | Regents Of The University Of Minnesota | TAL effector-mediated DNA modification |
| WO2014018423A2 (en) | 2012-07-25 | 2014-01-30 | The Broad Institute, Inc. | Inducible dna binding proteins and genome perturbation tools and applications thereof |
| US8697359B1 (en) | 2012-12-12 | 2014-04-15 | The Broad Institute, Inc. | CRISPR-Cas systems and methods for altering expression of gene products |
| WO2014093622A2 (en) | 2012-12-12 | 2014-06-19 | The Broad Institute, Inc. | Delivery, engineering and optimization of systems, methods and compositions for sequence manipulation and therapeutic applications |
| WO2014093718A1 (en) | 2012-12-12 | 2014-06-19 | The Broad Institute, Inc. | Methods, systems, and apparatus for identifying target sequences for cas enzymes or crispr-cas systems for target sequences and conveying results thereof |
| WO2014093709A1 (en) | 2012-12-12 | 2014-06-19 | The Broad Institute, Inc. | Methods, models, systems, and apparatus for identifying target sequences for cas enzymes or crispr-cas systems for target sequences and conveying results thereof |
| WO2014093694A1 (en) | 2012-12-12 | 2014-06-19 | The Broad Institute, Inc. | Crispr-cas nickase systems, methods and compositions for sequence manipulation in eukaryotes |
| WO2014093635A1 (en) | 2012-12-12 | 2014-06-19 | The Broad Institute, Inc. | Engineering and optimization of improved systems, methods and enzyme compositions for sequence manipulation |
| WO2014093712A1 (en) | 2012-12-12 | 2014-06-19 | The Broad Institute, Inc. | Engineering of systems, methods and optimized guide compositions for sequence manipulation |
| WO2014093595A1 (en) | 2012-12-12 | 2014-06-19 | The Broad Institute, Inc. | Crispr-cas component systems, methods and compositions for sequence manipulation |
| WO2014093701A1 (en) | 2012-12-12 | 2014-06-19 | The Broad Institute, Inc. | Functional genomics using crispr-cas systems, compositions, methods, knock out libraries and applications thereof |
| WO2014093655A2 (en) | 2012-12-12 | 2014-06-19 | The Broad Institute, Inc. | Engineering and optimization of systems, methods and compositions for sequence manipulation with functional domains |
| WO2014102774A1 (en) | 2012-12-26 | 2014-07-03 | Evogene Ltd. | Isolated polynucleotides and polypeptides, construct and plants comprising same and methods of using same for increasing nitrogen use efficiency of plants |
| WO2014145599A2 (en) | 2013-03-15 | 2014-09-18 | The Broad Institute, Inc. | Recombinant virus and preparations thereof |
| WO2014204723A1 (en) | 2013-06-17 | 2014-12-24 | The Broad Institute Inc. | Oncogenic models based on delivery and use of the crispr-cas systems, vectors and compositions |
| WO2014204726A1 (en) | 2013-06-17 | 2014-12-24 | The Broad Institute Inc. | Delivery and use of the crispr-cas systems, vectors and compositions for hepatic targeting and therapy |
| WO2014204727A1 (en) | 2013-06-17 | 2014-12-24 | The Broad Institute Inc. | Functional genomics using crispr-cas systems, compositions methods, screens and applications thereof |
| WO2014204729A1 (en) | 2013-06-17 | 2014-12-24 | The Broad Institute Inc. | Delivery, use and therapeutic applications of the crispr-cas systems and compositions for targeting disorders and diseases using viral components |
| WO2014204728A1 (en) | 2013-06-17 | 2014-12-24 | The Broad Institute Inc. | Delivery, engineering and optimization of systems, methods and compositions for targeting and modeling diseases and disorders of post mitotic cells |
| WO2014204725A1 (en) | 2013-06-17 | 2014-12-24 | The Broad Institute Inc. | Optimized crispr-cas double nickase systems, methods and compositions for sequence manipulation |
| WO2014204724A1 (en) | 2013-06-17 | 2014-12-24 | The Broad Institute Inc. | Delivery, engineering and optimization of tandem guide systems, methods and compositions for sequence manipulation |
| US8927247B2 (en) | 2008-01-31 | 2015-01-06 | Cellectis, S.A. | I-CreI derived single-chain meganuclease and uses thereof |
| WO2015065964A1 (en) | 2013-10-28 | 2015-05-07 | The Broad Institute Inc. | Functional genomics using crispr-cas systems, compositions, methods, screens and applications thereof |
| WO2015089364A1 (en) | 2013-12-12 | 2015-06-18 | The Broad Institute Inc. | Crystal structure of a crispr-cas system, and uses thereof |
| WO2015089462A1 (en) | 2013-12-12 | 2015-06-18 | The Broad Institute Inc. | Delivery, use and therapeutic applications of the crispr-cas systems and compositions for genome editing |
| WO2015089486A2 (en) | 2013-12-12 | 2015-06-18 | The Broad Institute Inc. | Systems, methods and compositions for sequence manipulation with optimized functional crispr-cas systems |
| WO2015089465A1 (en) | 2013-12-12 | 2015-06-18 | The Broad Institute Inc. | Delivery, use and therapeutic applications of the crispr-cas systems and compositions for hbv and viral diseases and disorders |
| WO2015089473A1 (en) | 2013-12-12 | 2015-06-18 | The Broad Institute Inc. | Engineering of systems, methods and optimized guide compositions with new architectures for sequence manipulation |
| WO2015089427A1 (en) | 2013-12-12 | 2015-06-18 | The Broad Institute Inc. | Crispr-cas systems and methods for altering expression of gene products, structural information and inducible modular cas enzymes |
| WO2015089354A1 (en) | 2013-12-12 | 2015-06-18 | The Broad Institute Inc. | Compositions and methods of use of crispr-cas systems in nucleotide repeat disorders |
| WO2015089419A2 (en) | 2013-12-12 | 2015-06-18 | The Broad Institute Inc. | Delivery, use and therapeutic applications of the crispr-cas systems and compositions for targeting disorders and diseases using particle delivery components |
| WO2016205711A1 (en) | 2015-06-18 | 2016-12-22 | The Broad Institute Inc. | Novel crispr enzymes and systems |
| WO2017062439A1 (en) | 2015-10-05 | 2017-04-13 | Precision Biosciences, Inc. | Engineered meganucleases with recognition sequences found in the human t cell receptor alpha constant region gene |
| WO2017106657A1 (en) | 2015-12-18 | 2017-06-22 | The Broad Institute Inc. | Novel crispr enzymes and systems |
| WO2017112859A1 (en) | 2015-12-23 | 2017-06-29 | Precision Biosciences, Inc. | Engineered meganucleases with recognition sequences found in the human beta-2 microglobulin gene |
| WO2017127807A1 (en) | 2016-01-22 | 2017-07-27 | The Broad Institute Inc. | Crystal structure of crispr cpf1 |
| CN114854692A (en) * | 2022-04-02 | 2022-08-05 | 北京默赛尔生物科技有限责任公司 | CAR-macrophage and preparation method thereof |
| WO2022246112A1 (en) * | 2021-05-19 | 2022-11-24 | University Of Southern California | Chimeric antigen receptor-modified granulocyte-macrophage progenitors for cancer immunotherapy |
| WO2023194607A1 (en) * | 2022-04-07 | 2023-10-12 | Institut Curie | Myeloid cells modified by chimeric antigen receptor with cd40 and uses thereof for anti-cancer therapy |
| WO2023194608A1 (en) * | 2022-04-07 | 2023-10-12 | Institut Curie | Myeloid cells modified by chimeric antigen receptor and uses thereof for anti-cancer therapy |
-
2024
- 2024-08-29 WO PCT/IB2024/058426 patent/WO2025046513A1/en active Pending
Patent Citations (108)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5585362A (en) | 1989-08-22 | 1996-12-17 | The Regents Of The University Of Michigan | Adenovirus vectors for gene therapy |
| US6468798B1 (en) | 1991-12-17 | 2002-10-22 | The Regents Of The University Of California | Expression of cloned genes in the lung by aerosol and liposome-based delivery |
| US6238924B1 (en) | 1992-05-05 | 2001-05-29 | Institut Pasteur | Nucleotide sequence encoding the enzyme I-SceI and the uses thereof |
| US5792632A (en) | 1992-05-05 | 1998-08-11 | Institut Pasteur | Nucleotide sequence encoding the enzyme I-SceI and the uses thereof |
| US5350674A (en) | 1992-09-04 | 1994-09-27 | Becton, Dickinson And Company | Intrinsic factor - horse peroxidase conjugates and a method for increasing the stability thereof |
| US6567694B2 (en) | 1993-04-01 | 2003-05-20 | Genetronics, Inc. | Needle electrodes for mediated delivery of drugs and genes |
| US5993434A (en) | 1993-04-01 | 1999-11-30 | Genetronics, Inc. | Method of treatment using electroporation mediated delivery of drugs and genes |
| US5830729A (en) | 1996-04-18 | 1998-11-03 | Institut Pasteur | I Sce I-induced gene replacement and gene conversion in embryonic stem cells |
| US20040092907A1 (en) | 1997-04-03 | 2004-05-13 | Inovio As | Method for muscle delivery of drugs, nucleic acids and other compounds |
| US6746838B1 (en) | 1997-05-23 | 2004-06-08 | Gendaq Limited | Nucleic acid binding proteins |
| US7241574B2 (en) | 1997-05-23 | 2007-07-10 | Gendaq Ltd. | Nucleic acid binding proteins |
| US7241573B2 (en) | 1997-05-23 | 2007-07-10 | Gendaq Ltd. | Nucleic acid binding proteins |
| US6866997B1 (en) | 1997-05-23 | 2005-03-15 | Gendaq Limited | Nucleic acid binding proteins |
| US6233482B1 (en) | 1997-08-01 | 2001-05-15 | Genetronics, Inc. | Method of electroporation mediated delivery of drugs and genes |
| US6241701B1 (en) | 1997-08-01 | 2001-06-05 | Genetronics, Inc. | Apparatus for electroporation mediated delivery of drugs and genes |
| US6181964B1 (en) | 1997-08-01 | 2001-01-30 | Genetronics, Inc. | Minimally invasive apparatus and method to electroporate drugs and genes into tissue |
| US6516223B2 (en) | 1997-08-01 | 2003-02-04 | Genetronics, Inc. | Apparatus for electroporation mediated delivery for drugs and genes |
| US6479626B1 (en) | 1998-03-02 | 2002-11-12 | Massachusetts Institute Of Technology | Poly zinc finger proteins with improved linkers |
| US7595376B2 (en) | 1998-03-02 | 2009-09-29 | Massachusetts Institute Of Technology | Poly zinc finger proteins with improved linkers |
| US6903185B2 (en) | 1998-03-02 | 2005-06-07 | Massachusetts Institute Of Technology | Poly zinc finger proteins with improved linkers |
| US6678556B1 (en) | 1998-07-13 | 2004-01-13 | Genetronics, Inc. | Electrical field therapy with reduced histopathological change in muscle |
| US6824978B1 (en) | 1999-01-12 | 2004-11-30 | Sangamo Biosciences, Inc. | Regulation of endogenous gene expression in cells using zinc finger proteins |
| US7220719B2 (en) | 1999-01-12 | 2007-05-22 | Sangamo Biosciences, Inc. | Modulation of endogenous gene expression in cells |
| US7013219B2 (en) | 1999-01-12 | 2006-03-14 | Sangamo Biosciences, Inc. | Regulation of endogenous gene expression in cells using zinc finger proteins |
| US6607882B1 (en) | 1999-01-12 | 2003-08-19 | Sangamo Biosciences, Inc. | Regulation of endogenous gene expression in cells using zinc finger proteins |
| US6979539B2 (en) | 1999-01-12 | 2005-12-27 | Sangamo Biosciences, Inc. | Regulation of endogenous gene expression in cells using zinc finger proteins |
| US6534261B1 (en) | 1999-01-12 | 2003-03-18 | Sangamo Biosciences, Inc. | Regulation of endogenous gene expression in cells using zinc finger proteins |
| US6933113B2 (en) | 1999-01-12 | 2005-08-23 | Sangamo Biosciences, Inc. | Modulation of endogenous gene expression in cells |
| US7585849B2 (en) | 1999-03-24 | 2009-09-08 | Sangamo Biosciences, Inc. | Position dependent recognition of GNN nucleotide triplets by zinc fingers |
| US7030215B2 (en) | 1999-03-24 | 2006-04-18 | Sangamo Biosciences, Inc. | Position dependent recognition of GNN nucleotide triplets by zinc fingers |
| US7171264B1 (en) | 1999-05-10 | 2007-01-30 | Genetronics, Inc. | Intradermal delivery of active agents by needle-free injection and electroporation |
| WO2001029058A1 (en) | 1999-10-15 | 2001-04-26 | University Of Massachusetts | Rna interference pathway genes as tools for targeted genetic interference |
| US6326193B1 (en) | 1999-11-05 | 2001-12-04 | Cambria Biosciences, Llc | Insect control agent |
| US7173116B2 (en) | 2000-03-03 | 2007-02-06 | Genetronics Biomedical Corporation | Nucleic acid formulations for gene delivery and methods of use |
| WO2001096584A2 (en) | 2000-06-12 | 2001-12-20 | Akkadix Corporation | Materials and methods for the control of nematodes |
| US6794136B1 (en) | 2000-11-20 | 2004-09-21 | Sangamo Biosciences, Inc. | Iterative optimization in the design of binding proteins |
| US20050052630A1 (en) | 2002-03-07 | 2005-03-10 | Advisys, Inc. | Constant current electroporation device and methods of use |
| US20030232410A1 (en) | 2002-03-21 | 2003-12-18 | Monika Liljedahl | Methods and compositions for using zinc finger endonucleases to enhance homologous recombination |
| US20040014645A1 (en) | 2002-05-28 | 2004-01-22 | Advisys, Inc. | Increased delivery of a nucleic acid construct in vivo by the poly-L-glutamate ("PLG") system |
| US20040059285A1 (en) | 2002-07-04 | 2004-03-25 | Inovio As | Electroporation device and injection apparatus |
| US20050070841A1 (en) | 2002-07-04 | 2005-03-31 | Inovio As | Electroporation device and injection apparatus |
| WO2004067753A2 (en) | 2003-01-28 | 2004-08-12 | Cellectis | Use of meganucleases for inducing homologous recombination ex vivo and in toto in vertebrate somatic tissues and application thereof. |
| US8119381B2 (en) | 2005-10-18 | 2012-02-21 | Duke University | Rationally-designed meganucleases with altered sequence specificity and DNA-binding affinity |
| US20070128708A1 (en) | 2005-12-07 | 2007-06-07 | Genetronics, Inc. | Variable volume electroporation chamber and methods therefore |
| WO2008010009A1 (en) | 2006-07-18 | 2008-01-24 | Cellectis | Meganuclease variants cleaving a dna target sequence from a rag gene and uses thereof |
| WO2008149176A1 (en) | 2007-06-06 | 2008-12-11 | Cellectis | Meganuclease variants cleaving a dna target sequence from the mouse rosa26 locus and uses thereof |
| WO2009013559A1 (en) | 2007-07-23 | 2009-01-29 | Cellectis | Meganuclease variants cleaving a dna target sequence from the human hemoglobin beta gene and uses thereof |
| US20090203140A1 (en) | 2007-09-27 | 2009-08-13 | Sangamo Biosciences, Inc. | Genomic editing in zebrafish using zinc finger nucleases |
| WO2009059195A2 (en) | 2007-10-31 | 2009-05-07 | Precision Biosciences | Rationally-designed single-chain meganucleases with non-palindromic recognition sequences |
| US8927247B2 (en) | 2008-01-31 | 2015-01-06 | Cellectis, S.A. | I-CreI derived single-chain meganuclease and uses thereof |
| US8338157B2 (en) | 2008-03-11 | 2012-12-25 | Precision Biosciences, Inc. | Rationally-designed meganuclease variants of lig-34 and I-crei for maize genome engineering |
| US8697853B2 (en) | 2009-12-10 | 2014-04-15 | Regents Of The University Of Minnesota | TAL effector-mediated DNA modification |
| US8440431B2 (en) | 2009-12-10 | 2013-05-14 | Regents Of The University Of Minnesota | TAL effector-mediated DNA modification |
| US8586363B2 (en) | 2009-12-10 | 2013-11-19 | Regents Of The University Of Minnesota | TAL effector-mediated DNA modification |
| US8440432B2 (en) | 2009-12-10 | 2013-05-14 | Regents Of The University Of Minnesota | Tal effector-mediated DNA modification |
| US8450471B2 (en) | 2009-12-10 | 2013-05-28 | Regents Of The University Of Minnesota | TAL effector-mediated DNA modification |
| WO2014018423A2 (en) | 2012-07-25 | 2014-01-30 | The Broad Institute, Inc. | Inducible dna binding proteins and genome perturbation tools and applications thereof |
| WO2014093655A2 (en) | 2012-12-12 | 2014-06-19 | The Broad Institute, Inc. | Engineering and optimization of systems, methods and compositions for sequence manipulation with functional domains |
| US8906616B2 (en) | 2012-12-12 | 2014-12-09 | The Broad Institute Inc. | Engineering of systems, methods and optimized guide compositions for sequence manipulation |
| WO2014093709A1 (en) | 2012-12-12 | 2014-06-19 | The Broad Institute, Inc. | Methods, models, systems, and apparatus for identifying target sequences for cas enzymes or crispr-cas systems for target sequences and conveying results thereof |
| WO2014093694A1 (en) | 2012-12-12 | 2014-06-19 | The Broad Institute, Inc. | Crispr-cas nickase systems, methods and compositions for sequence manipulation in eukaryotes |
| WO2014093635A1 (en) | 2012-12-12 | 2014-06-19 | The Broad Institute, Inc. | Engineering and optimization of improved systems, methods and enzyme compositions for sequence manipulation |
| WO2014093712A1 (en) | 2012-12-12 | 2014-06-19 | The Broad Institute, Inc. | Engineering of systems, methods and optimized guide compositions for sequence manipulation |
| WO2014093661A2 (en) | 2012-12-12 | 2014-06-19 | The Broad Institute, Inc. | Crispr-cas systems and methods for altering expression of gene products |
| WO2014093595A1 (en) | 2012-12-12 | 2014-06-19 | The Broad Institute, Inc. | Crispr-cas component systems, methods and compositions for sequence manipulation |
| WO2014093701A1 (en) | 2012-12-12 | 2014-06-19 | The Broad Institute, Inc. | Functional genomics using crispr-cas systems, compositions, methods, knock out libraries and applications thereof |
| WO2014093622A2 (en) | 2012-12-12 | 2014-06-19 | The Broad Institute, Inc. | Delivery, engineering and optimization of systems, methods and compositions for sequence manipulation and therapeutic applications |
| US8932814B2 (en) | 2012-12-12 | 2015-01-13 | The Broad Institute Inc. | CRISPR-Cas nickase systems, methods and compositions for sequence manipulation in eukaryotes |
| US8771945B1 (en) | 2012-12-12 | 2014-07-08 | The Broad Institute, Inc. | CRISPR-Cas systems and methods for altering expression of gene products |
| US8795965B2 (en) | 2012-12-12 | 2014-08-05 | The Broad Institute, Inc. | CRISPR-Cas component systems, methods and compositions for sequence manipulation |
| US8697359B1 (en) | 2012-12-12 | 2014-04-15 | The Broad Institute, Inc. | CRISPR-Cas systems and methods for altering expression of gene products |
| US8865406B2 (en) | 2012-12-12 | 2014-10-21 | The Broad Institute Inc. | Engineering and optimization of improved systems, methods and enzyme compositions for sequence manipulation |
| US8871445B2 (en) | 2012-12-12 | 2014-10-28 | The Broad Institute Inc. | CRISPR-Cas component systems, methods and compositions for sequence manipulation |
| US8889418B2 (en) | 2012-12-12 | 2014-11-18 | The Broad Institute Inc. | Engineering and optimization of improved systems, methods and enzyme compositions for sequence manipulation |
| US8889356B2 (en) | 2012-12-12 | 2014-11-18 | The Broad Institute Inc. | CRISPR-Cas nickase systems, methods and compositions for sequence manipulation in eukaryotes |
| US8895308B1 (en) | 2012-12-12 | 2014-11-25 | The Broad Institute Inc. | Engineering and optimization of improved systems, methods and enzyme compositions for sequence manipulation |
| WO2014093718A1 (en) | 2012-12-12 | 2014-06-19 | The Broad Institute, Inc. | Methods, systems, and apparatus for identifying target sequences for cas enzymes or crispr-cas systems for target sequences and conveying results thereof |
| US8999641B2 (en) | 2012-12-12 | 2015-04-07 | The Broad Institute Inc. | Engineering and optimization of systems, methods and compositions for sequence manipulation with functional domains |
| US8993233B2 (en) | 2012-12-12 | 2015-03-31 | The Broad Institute Inc. | Engineering and optimization of systems, methods and compositions for sequence manipulation with functional domains |
| US8945839B2 (en) | 2012-12-12 | 2015-02-03 | The Broad Institute Inc. | CRISPR-Cas systems and methods for altering expression of gene products |
| WO2014102774A1 (en) | 2012-12-26 | 2014-07-03 | Evogene Ltd. | Isolated polynucleotides and polypeptides, construct and plants comprising same and methods of using same for increasing nitrogen use efficiency of plants |
| WO2014145599A2 (en) | 2013-03-15 | 2014-09-18 | The Broad Institute, Inc. | Recombinant virus and preparations thereof |
| WO2014204729A1 (en) | 2013-06-17 | 2014-12-24 | The Broad Institute Inc. | Delivery, use and therapeutic applications of the crispr-cas systems and compositions for targeting disorders and diseases using viral components |
| WO2014204724A1 (en) | 2013-06-17 | 2014-12-24 | The Broad Institute Inc. | Delivery, engineering and optimization of tandem guide systems, methods and compositions for sequence manipulation |
| WO2014204725A1 (en) | 2013-06-17 | 2014-12-24 | The Broad Institute Inc. | Optimized crispr-cas double nickase systems, methods and compositions for sequence manipulation |
| WO2014204728A1 (en) | 2013-06-17 | 2014-12-24 | The Broad Institute Inc. | Delivery, engineering and optimization of systems, methods and compositions for targeting and modeling diseases and disorders of post mitotic cells |
| WO2014204727A1 (en) | 2013-06-17 | 2014-12-24 | The Broad Institute Inc. | Functional genomics using crispr-cas systems, compositions methods, screens and applications thereof |
| WO2014204726A1 (en) | 2013-06-17 | 2014-12-24 | The Broad Institute Inc. | Delivery and use of the crispr-cas systems, vectors and compositions for hepatic targeting and therapy |
| WO2014204723A1 (en) | 2013-06-17 | 2014-12-24 | The Broad Institute Inc. | Oncogenic models based on delivery and use of the crispr-cas systems, vectors and compositions |
| WO2015065964A1 (en) | 2013-10-28 | 2015-05-07 | The Broad Institute Inc. | Functional genomics using crispr-cas systems, compositions, methods, screens and applications thereof |
| WO2015089427A1 (en) | 2013-12-12 | 2015-06-18 | The Broad Institute Inc. | Crispr-cas systems and methods for altering expression of gene products, structural information and inducible modular cas enzymes |
| WO2015089462A1 (en) | 2013-12-12 | 2015-06-18 | The Broad Institute Inc. | Delivery, use and therapeutic applications of the crispr-cas systems and compositions for genome editing |
| WO2015089486A2 (en) | 2013-12-12 | 2015-06-18 | The Broad Institute Inc. | Systems, methods and compositions for sequence manipulation with optimized functional crispr-cas systems |
| WO2015089465A1 (en) | 2013-12-12 | 2015-06-18 | The Broad Institute Inc. | Delivery, use and therapeutic applications of the crispr-cas systems and compositions for hbv and viral diseases and disorders |
| WO2015089473A1 (en) | 2013-12-12 | 2015-06-18 | The Broad Institute Inc. | Engineering of systems, methods and optimized guide compositions with new architectures for sequence manipulation |
| WO2015089364A1 (en) | 2013-12-12 | 2015-06-18 | The Broad Institute Inc. | Crystal structure of a crispr-cas system, and uses thereof |
| WO2015089354A1 (en) | 2013-12-12 | 2015-06-18 | The Broad Institute Inc. | Compositions and methods of use of crispr-cas systems in nucleotide repeat disorders |
| WO2015089351A1 (en) | 2013-12-12 | 2015-06-18 | The Broad Institute Inc. | Compositions and methods of use of crispr-cas systems in nucleotide repeat disorders |
| WO2015089419A2 (en) | 2013-12-12 | 2015-06-18 | The Broad Institute Inc. | Delivery, use and therapeutic applications of the crispr-cas systems and compositions for targeting disorders and diseases using particle delivery components |
| WO2016205711A1 (en) | 2015-06-18 | 2016-12-22 | The Broad Institute Inc. | Novel crispr enzymes and systems |
| WO2017062439A1 (en) | 2015-10-05 | 2017-04-13 | Precision Biosciences, Inc. | Engineered meganucleases with recognition sequences found in the human t cell receptor alpha constant region gene |
| WO2017106657A1 (en) | 2015-12-18 | 2017-06-22 | The Broad Institute Inc. | Novel crispr enzymes and systems |
| WO2017112859A1 (en) | 2015-12-23 | 2017-06-29 | Precision Biosciences, Inc. | Engineered meganucleases with recognition sequences found in the human beta-2 microglobulin gene |
| WO2017127807A1 (en) | 2016-01-22 | 2017-07-27 | The Broad Institute Inc. | Crystal structure of crispr cpf1 |
| WO2022246112A1 (en) * | 2021-05-19 | 2022-11-24 | University Of Southern California | Chimeric antigen receptor-modified granulocyte-macrophage progenitors for cancer immunotherapy |
| CN114854692A (en) * | 2022-04-02 | 2022-08-05 | 北京默赛尔生物科技有限责任公司 | CAR-macrophage and preparation method thereof |
| WO2023194607A1 (en) * | 2022-04-07 | 2023-10-12 | Institut Curie | Myeloid cells modified by chimeric antigen receptor with cd40 and uses thereof for anti-cancer therapy |
| WO2023194608A1 (en) * | 2022-04-07 | 2023-10-12 | Institut Curie | Myeloid cells modified by chimeric antigen receptor and uses thereof for anti-cancer therapy |
Non-Patent Citations (84)
| Title |
|---|
| ABDIN SHIFAA M. ET AL: "Scalable generation of functional human iPSC-derived CAR-macrophages that efficiently eradicate CD19-positive leukemia", JOURNAL FOR IMMUNOTHERAPY OF CANCER, vol. 11, no. 12, 1 December 2023 (2023-12-01), GB, pages e007705, XP093242485, ISSN: 2051-1426, Retrieved from the Internet <URL:https://pmc.ncbi.nlm.nih.gov/articles/PMC10749073/> DOI: 10.1136/jitc-2023-007705 * |
| ABDULRAZZAK, HASSAN ET AL.: "Biological characteristics of stem cells from foetal, cord blood and extraembryonic tissues.", JOURNAL OF THE ROYAL SOCIETY, INTERFACE, vol. 7, 2010, pages 689 - 706 |
| ALFTHAN, K: "Properties of a single-chain antibody containing different linker peptides", PROTEIN ENGINEERING, vol. 8, no. 7, 1995, pages 725 - 731, XP002112077 |
| ALTSCHUL ET AL., J. MOL. BIOL., vol. 215, 1990, pages 403 - 410 |
| ALTSCHUL ET AL., NUCLEIC ACIDS RES., vol. 25, 1997, pages 3389 - 3402 |
| BARNES ET AL., J FOR IMMUNOTHERAPY OF CANCER, vol. 3, no. 1, 2015, pages 1 - 11 |
| BERARDI, A C ET AL.: "Functional isolation and characterization of human hematopoietic stem cells", SCIENCE, vol. 267, no. 5194, 1995, pages 104 - 8, XP002923338, DOI: 10.1126/science.7528940 |
| BERENSON, R: "ngraftment after infusion of CD34+ marrow cells in patients with breast cancer or neuroblastoma", BLOOD, vol. 77, no. 8, 1991, pages 1717 - 22 |
| BEURDELEY ET AL., NAT COMMUN, vol. 4, 2013, pages 1762 |
| BIBIKOVA ET AL., GENETICS, vol. 161, 2002, pages 1169 - 1175 |
| BIBIKOVA ET AL., SCIENCE, vol. 300, 2003, pages 764 |
| BIRD ET AL., SCIENCE, vol. 242, 1988, pages 423 - 426 |
| BORDENKABAT, PNAS, vol. 84, 1987, pages 2440 - 2443 |
| CAFREEMAN, IMMUNOLOGICAL REVIEWS, vol. 229, no. 1, 2009, pages 244 - 258 |
| CHEVALIERSTODDARD, NUCLEIC ACIDS RESEARCH, vol. 29, 2001, pages 3757 - 3774 |
| CHOTHIA ET AL., NATURE, vol. 342, 1989, pages 877 - 883 |
| CHOTHIALESK, J MOL. BIOL., vol. 196, no. 4, 1987, pages 901 - 917 |
| CHRISTIAN ET AL., GENETICS, vol. 186, 2010, pages 757 - 761 |
| COUGOT ET AL., TRENDS IN BIOCHEM. SCI., vol. 29, 2001, pages 436 - 444 |
| DAI ET AL., FRONTIERS IN IMMUNOLOGY, vol. 11, 2020, pages 539654 |
| DEL RIO ET AL., JOURNAL OF LEUKOCYTE BIOLOGY, vol. 87, no. 2, 2010, pages 223 - 235 |
| DING, LEI ET AL.: "Endothelial and perivascular cells maintain haematopoietic stem cells", NATURE, vol. 481, no. 7382, 25 January 2012 (2012-01-25), pages 457 - 546, XP055090112, DOI: 10.1038/nature10783 |
| DOTTI ET AL., IMMUNOL. REV., vol. 257, no. 1, 2014, pages 107 |
| ELGANO, BIOPHYS. RES. COMMUN, vol. 330, 2005, pages 958 - 966 |
| GAJ ET AL., NAT METHODS, vol. 1 -4, no. 8, 2012, pages 805 - 7 |
| GHOSH ET AL., GLYCOBIOLOGY, vol. 5, 1991, pages 505 - 10 |
| GISHSTATES, NATURE GENET., vol. 3, 1993, pages 266 - 272 |
| GRANGERRICKERT, CYTOKINE GROWTH FACTOR REV, vol. 14, no. 3-4, 2003, pages 289 - 296 |
| HAAS, LISAANNA C OBENAUF: "Allies or Enemies-The Multifaceted Role of Myeloid Cells in the Tumor Microenvironment.", FRONTIERS IN IMMUNOLOGY, vol. 10, 29 November 2019 (2019-11-29), pages 2746 |
| HENIKOFF SHENIKOFF J G., PROC NATL ACAD SCI, vol. 89, 1992, pages 10915 - 9 |
| HOLLIGER ET AL., PROC. NATL. ACAD SCI. USA, vol. 90, 1993, pages 6444 - 6448 |
| HOLLIGER, PHUDSON, J: "Engineered antibody fragments and the rise of single domains", NATURE BIOTECHNOLOGY, vol. 23, no. 9, 2005, pages 1126 - 1136, XP008076746, DOI: 10.1038/nbt1142 |
| HONEGGERPLUCKTHUN, JOURNAL OF MOLECULAR BIOLOGY, vol. 309, 2001, pages 657 - 670 |
| HSU ET AL., JOURNAL OF BIOLOGICAL CHEMISTRY, vol. 272, no. 21, 1997, pages 13471 - 13474 |
| HUSTON ET AL., PROC. NATL. ACAD. SCI. USA, vol. 85, 1988, pages 5879 - 5883 |
| JONES ET AL., NATURE, vol. 321, 1986, pages 522 - 525 |
| JOUNGSANDER, NAT REV MOL CELL BIOL, vol. 14, no. 1, 2013, pages 49 - 55 |
| KABAT ET AL., U.S. DEPARTMENT OF HEALTH AND HUMAN SERVICES, 1991 |
| KAWALEKAR ET AL., IMMUNITY, vol. 44, no. 2, 2016 |
| KERSHAW ET AL., CLIN. CANCER RES, vol. 12, no. 20, 2006, pages 6106 |
| KIM ET AL., GENOME RES, vol. 22, no. 7, 2012, pages 1327 - 33 |
| KIM ET AL., PROCEEDINGS OF THE NATIONAL ACADEMY OF SCIENCES OF THE UNITED STATES OF AMERICA, vol. 93, 1996, pages 1156 - 1160 |
| LAMBERT, CLAUDE ET AL.: "Monocytes and macrophages in flow: an ESCCA initiative on advanced analyses of monocyte lineage using flow cytometry", CYTOMETRY. PART B, CLINICAL CYTOMETRY, vol. 92, no. 3, 2017, pages 180 - 188, XP072330013, DOI: 10.1002/cyto.b.21280 |
| LIM, W.FNOUE-YOKOO, TTAN, K.S ET AL.: "Hematopoietic cell differentiation from embryonic and induced pluripotent stem cells", STEM CELL RES THER, vol. 4, 2013, pages 71 |
| LONG ET AL., NAT. MED., vol. 21, no. 6, 2015, pages 581 |
| LUTZ ET AL., JOURNAL OF IMMUNOLOGICAL METHODS, vol. 223, no. 1, 1999, pages 77 - 92 |
| MADDEN ET AL., METH. ENZYMOL., vol. 266, 1996, pages 131 - 141 |
| MAUS ET AL., BLOOD, vol. 123, no. 17, 2014, pages 2625 |
| MCNIECE, I KR A BRIDDELLSTEM CELL FACTOR, JOURNAL OF LEUKOCYTE BIOLOGY, vol. 58, no. 1, 1995, pages 14 - 22 |
| METCALF D., HEMATOPOIETIC CYTOKINES, vol. 111, no. 2, 2008, pages 485 - 491 |
| MILLER ET AL., NATURE BIOTECHNOLOGY, vol. 25, 2007, pages 778 - 785 |
| MILLER ET AL., NATURE BIOTECHNOLOGY, vol. 29, 2011, pages 143 - 148 |
| MILLER ET AL., THE EMBO JOURNAL, vol. 4, 1985, pages 1609 - 1614 |
| MILONE ET AL., MOLECULAR THERAPY, vol. 17, no. 8, 2009, pages 1453 - 1464 |
| MOSCOUBOGDANOVE, SCIENCE, vol. 326, 2009, pages 1501 - 1512 |
| MOSSEREDWARDS, NATURE REVIEWS IMMUNOLOGY, vol. 8, no. 12, 2008, pages 958 - 969 |
| NACHEVABERZAL-HERRANZ, EUR. J. BIOCHEM., vol. 270, 2003, pages 1485 - 65 |
| NING ET AL., FRONTIERS IN IMMUNOLOGY, vol. 12, 2021, pages 654960 |
| NISHIKAWA ET AL., HUM GENE THER, vol. 1 -4, no. 8, 2001, pages 861 - 70 |
| PAASCH DANIELA ET AL: "Ex Vivo Generation of CAR Macrophages from Hematopoietic Stem and Progenitor Cells for Use in Cancer Therapy", CELLS, vol. 11, no. 6, 15 March 2022 (2022-03-15), pages 994, XP093242491, ISSN: 2073-4409, DOI: 10.3390/cells11060994 * |
| PARK ET AL., CANCER IMMUNOL. IMMUNOTHER., vol. 61, no. 2, 2012, pages 203 |
| PASEROOLIVE, IMMUNOLOGY LETTERS, vol. 151, no. 1-2, 2013, pages 71 - 75 |
| PRESTA, CURR. OP. STRUCT. BIOL., vol. 2, 1992, pages 593 - 596 |
| RAMIREZ ET AL., NUCL ACIDS RES, vol. 40, no. 12, 2012, pages 5560 - 8 |
| REICHMANN ET AL., NATURE, vol. 332, 1988, pages 323 - 329 |
| ROSENBERG ET AL., NEW ENG. J. OF MED., vol. 319, pages 1676 - 1988 |
| SCHARENBERG ET AL., CURR GENE THER, vol. 13, no. 4, 2013, pages 291 - 303 |
| SCHENBORNMIERENDORF, NUC ACIDS RES., vol. 13, 1985, pages 6223 - 36 |
| SEDY ET AL., NATURE IMMUNOLOGY, vol. 6, no. 1, 2005, pages 90 - 98 |
| SHUI, JOURNAL OF LEUKOCYTE BIOLOGY, vol. 89, no. 4, 2011, pages 517 - 523 |
| SOROBETEABRODSKY, CELL HOST & MICROBE, vol. 24, no. 2, 2018, pages 187 - 188 |
| STEINBERG ET AL., PLOS ONE, vol. 8, no. 10, 2013, pages 77992 |
| STEPINSKI ET AL., RNA, vol. 7, 2001, pages 1468 - 95 |
| URNOV ET AL., NATURE REVIEWS GENETICS, vol. 11, 2010, pages 636 - 646 |
| VAN FURTH ET AL., BULLETIN OF THE WORLD HEALTH ORGANIZATION, vol. 46, no. 6, 1972, pages 845 - 852 |
| VO, LINDA TGEORGE Q DALEY.: "De novo generation of HSCs from somatic and pluripotent stem cell sources", BLOOD, vol. 125, no. 17, 2015, pages 2641 - 8, XP055323623, DOI: 10.1182/blood-2014-10-570234 |
| WEISKOPF, KIPP ET AL.: "Myeloid Cell Origins, Differentiation, and Clinical Implications", MICROBIOLOGY SPECTRUM, vol. 4, 2016, pages 5 |
| WOLFE ET AL., ANNUAL REVIEW OF BIOPHYSICS AND BIOMOLECULAR STRUCTURE, vol. 29, 2000, pages 183 - 212 |
| WUKABAT, J EXP MED., vol. 132, no. 2, 1970, pages 211 - 50 |
| YADAV, POOJA ET AL.: "Hematopoietic Stem Cells Culture, Expansion and Differentiation: An Insight into Variable and Available Media", INTERNATIONAL JOURNAL OF STEM CELLS, vol. 13, no. 3, 2020, pages 326 - 334 |
| ZARNEGAR ET AL., NATURE IMMUNOLOGY, vol. 9, no. 12, 2008, pages 1371 - 1378 |
| ZHANG ET AL., J. COMPUT. BIOL., vol. 7, no. 1-2, 2000, pages 203 - 14 |
| ZHANG ET AL., ONCOTARGET, vol. 6, no. 32, 2015, pages 33961 |
| ZHANG, CHENG CHARVEY F LODISH: "Cytokines regulating hematopoietic stem cell function.", CURRENT OPINION IN HEMATOLOGY, vol. 15, no. 4, 2008, pages 307 - 11, XP055731675, DOI: 10.1097/MOH.0b013e3283007db5 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US20240066064A1 (en) | Methods and compositions for gene editing in hematopoietic stem cells | |
| US20190175651A1 (en) | Immortalized car-t cells genetically modified to elminate t-cell receptor and beta 2-microglobulin expression | |
| JP2023529841A (en) | Novel constructs for chimeric antigen receptors | |
| JP2022524081A (en) | Human Carbonic Amhydrase 2 Compositions and Methods for Adjustable Control | |
| CN113454115B (en) | Chimeric antigen receptor targeting sialyl Lewis A and use thereof | |
| US20220133917A1 (en) | Small Molecules for Dual Function Positron Emission Tomography (PET) and Cell Suicide Switches | |
| US20220119476A1 (en) | Activation of Antigen Presenting Cells and Methods for Using the Same | |
| WO2019097083A1 (en) | Modified k562 cell | |
| TW202430629A (en) | New large scale car-t immune cell manufacturing method utilizing lentiviral vector transfection | |
| US20250281536A1 (en) | Cd4-specific antibody constructs and compositions and uses thereof | |
| WO2024030583A2 (en) | Novel constructs for chimeric antigen receptors and uses thereof | |
| US12473342B2 (en) | Chimeric antigen receptor T regulatory cells for the treatment of atherosclerosis | |
| WO2025046513A1 (en) | Methods of manufacturing myeloid-derived cells from hematopoietic stem cells and compositions and uses thereof | |
| WO2024236547A1 (en) | Modified phagocytic cells expressing chimeric antigen receptors comprising a herpes virus entry mediator (hvem) co-stimulatory domain and uses thereof | |
| KR20230131816A (en) | Hypoimmunogenic stem cells, hypoimmunogenic cells differentiated or derived from stem cells, and method for producing the same | |
| US20220041984A1 (en) | Mobilized peripheral blood as a source of modified immune cells | |
| US12258379B2 (en) | Compositions and methods for switchable CAR T cells using surface-bound sortase transpeptidase | |
| WO2025085744A1 (en) | Fusion polypeptides and uses thereof | |
| WO2025085721A1 (en) | Chimeric receptors and uses thereof | |
| WO2025222093A2 (en) | Engineered cells and uses thereof | |
| EP4486787A1 (en) | Anti-cd19 and anti-cd79b chimeric antigen receptors and methods of use thereof | |
| HK1259151B (en) | Methods and compositions for gene editing in hematopoietic stem cells |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 24776648 Country of ref document: EP Kind code of ref document: A1 |