WO2025046387A1 - Dichroic polarizer film - Google Patents
Dichroic polarizer film Download PDFInfo
- Publication number
- WO2025046387A1 WO2025046387A1 PCT/IB2024/058059 IB2024058059W WO2025046387A1 WO 2025046387 A1 WO2025046387 A1 WO 2025046387A1 IB 2024058059 W IB2024058059 W IB 2024058059W WO 2025046387 A1 WO2025046387 A1 WO 2025046387A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- film
- polyvinyl alcohol
- composite construction
- crosslinked
- combination
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Classifications
-
- G—PHYSICS
- G02—OPTICS
- G02B—OPTICAL ELEMENTS, SYSTEMS OR APPARATUS
- G02B5/00—Optical elements other than lenses
- G02B5/30—Polarising elements
- G02B5/3025—Polarisers, i.e. arrangements capable of producing a definite output polarisation state from an unpolarised input state
- G02B5/3033—Polarisers, i.e. arrangements capable of producing a definite output polarisation state from an unpolarised input state in the form of a thin sheet or foil, e.g. Polaroid
-
- G—PHYSICS
- G02—OPTICS
- G02B—OPTICAL ELEMENTS, SYSTEMS OR APPARATUS
- G02B5/00—Optical elements other than lenses
- G02B5/30—Polarising elements
- G02B5/3025—Polarisers, i.e. arrangements capable of producing a definite output polarisation state from an unpolarised input state
- G02B5/3033—Polarisers, i.e. arrangements capable of producing a definite output polarisation state from an unpolarised input state in the form of a thin sheet or foil, e.g. Polaroid
- G02B5/3041—Polarisers, i.e. arrangements capable of producing a definite output polarisation state from an unpolarised input state in the form of a thin sheet or foil, e.g. Polaroid comprising multiple thin layers, e.g. multilayer stacks
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B29—WORKING OF PLASTICS; WORKING OF SUBSTANCES IN A PLASTIC STATE IN GENERAL
- B29D—PRODUCING PARTICULAR ARTICLES FROM PLASTICS OR FROM SUBSTANCES IN A PLASTIC STATE
- B29D11/00—Producing optical elements, e.g. lenses or prisms
- B29D11/00634—Production of filters
- B29D11/00644—Production of filters polarizing
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B29—WORKING OF PLASTICS; WORKING OF SUBSTANCES IN A PLASTIC STATE IN GENERAL
- B29D—PRODUCING PARTICULAR ARTICLES FROM PLASTICS OR FROM SUBSTANCES IN A PLASTIC STATE
- B29D11/00—Producing optical elements, e.g. lenses or prisms
- B29D11/0074—Production of other optical elements not provided for in B29D11/00009- B29D11/0073
- B29D11/00788—Producing optical films
-
- G—PHYSICS
- G02—OPTICS
- G02B—OPTICAL ELEMENTS, SYSTEMS OR APPARATUS
- G02B5/00—Optical elements other than lenses
- G02B5/30—Polarising elements
- G02B5/3025—Polarisers, i.e. arrangements capable of producing a definite output polarisation state from an unpolarised input state
- G02B5/3033—Polarisers, i.e. arrangements capable of producing a definite output polarisation state from an unpolarised input state in the form of a thin sheet or foil, e.g. Polaroid
- G02B5/3041—Polarisers, i.e. arrangements capable of producing a definite output polarisation state from an unpolarised input state in the form of a thin sheet or foil, e.g. Polaroid comprising multiple thin layers, e.g. multilayer stacks
- G02B5/305—Polarisers, i.e. arrangements capable of producing a definite output polarisation state from an unpolarised input state in the form of a thin sheet or foil, e.g. Polaroid comprising multiple thin layers, e.g. multilayer stacks including organic materials, e.g. polymeric layers
Definitions
- modified films are absorptive polarizers.
- the modified film comprises an oriented polyvinyl alcohol film containing a dichroic material, wherein the oriented polyvinyl alcohol film has been crosslinked, end capped or both with a polyphenol compound, a nano-platelet material, a hydrogen bonding polymer, a non-phenolic polyacid, or a combination thereof.
- the multi-layer film articles comprise at least one film; and an absorptive polarizer film disposed on the at least one film.
- the absorptive polarizer film comprises a modified film as described above.
- Also disclosed are methods of forming multi-layer film articles comprising providing at least one film, providing a film-forming solution, where the film-forming solution comprises at least one solvent and polyvinyl alcohol, disposing the film-forming solution on the at least one film, drying the film-forming solution to form a composite construction.
- the composite construction comprises a polyvinyl alcohol film disposed on the film.
- the composite construction is oriented, a staining composition is applied to the composite construction, where the staining composition comprises at least one dichroic material and at least one solvent.
- the composite construction with staining composition disposed on it is dried and a crosslinker/end-capper composition is applied to the polyvinyl alcohol film of the composite construction.
- the crosslinker/end-capper composition comprises a crosslinker, an end-capper or both, where the crosslinker or endcapper comprises a polyphenol compound, a nano-platelet material a hydrogen bonding polymer, a non-phenolic polyacid, or a combination thereof, and at least one solvent.
- the composite construction with crosslinker/end-capper composition disposed on it is dried.
- Figure 1 is a cross sectional view of an article of the current disclosure.
- Figure 2 is a cross sectional view of another article of the current disclosure.
- Optical polarizing films are widely used in optical applications. Optical polarizing films are used for example to reduce glare in sunglasses and for increasing optical contrast in Liquid Crystal Displays (LCD).
- LCD Liquid Crystal Displays
- polarizers There are generally two types of polarizers that are used.
- One type of polarizer is a dichroic polarizer, also known as an absorbing or absorptive polarizer.
- Dichroic polarizers absorb light of one polarization and transmit light of the orthogonal polarization.
- Dichroic polarizers can be prepared by incorporating a dichroic material such as a dye into a polymer sheet that is stretched in at least one direction.
- Another type of polarizer is a reflective polarizer. Reflective polarizers reflect light of one polarization and transmit light of the orthogonal polarization.
- One type of reflective polarizer is made by forming a stack of alternating polymer layers. The two types of polarizers may be combined to make a single optical polarizer, thereby incorporating the useful characteristics of both types of polarizers.
- the need remains for dichroic polarizer fdms, methods for preparing these fdms, as well as methods of forming single optical polarizers that contain a reflective polarizer and a dichroic polarizer.
- the desirable goals are the simplification of the process to form the dichroic polarizer films and the single optical polarizers.
- typically these films are prepared from polyvinyl alcohol that is crosslinked with boric acid. Boric acid is a hazardous material and requires special handling, so it is desirable to develop other crosslinking and/or end-capping agents for use with polyvinyl alcohol.
- Tg glass transition temperature
- DSC Differential Scanning Calorimetry
- adjacent as used herein when referring to two layers means that the two layers are in proximity with one another with no intervening open space between them. They may be in direct contact with one another (e.g., laminated together) or there may be intervening layers.
- crosslinking and end-capping of polyvinyl alcohol polymers are used according to their commonly understood meanings in the polymer art. Crosslinking and end-capping are carried out by crosslinking and end-capping agents. These agents can covalently bond with the hydroxyl groups of a polyvinyl alcohol polymer and/or interact through secondary/non-covalent interactions (e.g., hydrogen bonding) with the hydroxyl groups. Polyvinyl alcohol polymers contain multiple chains of polyvinyl alcohol within the polymeric mixture. If the agent reacts with a single hydroxyl group on the polyvinyl alcohol polymer, or hydroxyl groups on a single polyvinyl alcohol polymer strand, it is described as end-capping.
- agent reacts with more than one hydroxyl group and the hydroxyl groups are on different polyvinyl alcohol polymer strands, it is described as crosslinking.
- An agent may function as an end-capper, as a crosslinker, or both as an endcapper and a crosslinker.
- end-cappers whether they are monofunctional or polyfunctional, reside on the surface of the polymer layer, whereas crosslinkers typically migrate into the polymer layer to form crosslinks.
- Hydrogen bonding is used herein according to its well understood meaning. Hydrogen bonding is not covalent or ionic bonding but rather is an interaction involving a weak physical bond between two molecules resulting from an electrostatic attraction between a proton in one molecule and an electronegative atom (such as oxygen) in the other.
- dichroic material refers to materials that are dichroic and include dichroic dyes as well as dichroic materials such as iodine/iodide.
- optically transparent refers to an article, film or adhesive that has a high light transmittance over at least a portion of the visible light spectrum (about 400 to about 700 nm).
- optically transparent articles have a visible light transmittance of at least 90% and a haze of less than 10%.
- optically clear refers to an adhesive or article that has a high light transmittance over at least a portion of the visible light spectrum (about 400 to about 700 nm), and that exhibits low haze, typically less than about 5%, or even less than about 2%.
- optically clear articles exhibit a haze of less than 1% at a thickness of 50 micrometers or even 0.5% at a thickness of 50 micrometers.
- optically clear articles have a visible light transmittance of at least 95%, often higher such as 97%, 98% or even 99% or higher.
- the multilayer film articles comprise at least one film, and an absorptive polarizer film disposed on the at least one film.
- the absorptive polymer film comprises an oriented polyvinyl alcohol film containing a dichroic material, where the oriented polyvinyl alcohol film has been crosslinked, end capped or both by a polyphenol compound, a nano-platelet material, a hydrogen bonding polymer, a non-phenolic polyacid, or a combination thereof. Each of these materials is described in detail below.
- the multi-layer film articles are polarizers.
- the polyvinyl alcohol film can be an absorptive polarizer.
- the at least one film layer is a reflective polarizer.
- the reflective polarizer can be used for polarization recycling in a liquid crystal display application, for example, and an absorbing polarizer can be added to the reflective polarizer to improve extinction efficiency. Integrating an absorbing polarizer with a reflective polarizer has been described in U.S. Pat. Nos. 6,096,375 (Ouderkirk et al.), 6,697,195 (Weber et al.), 7,826,009 (Weber et al.), and 6,111,697 (Kausch et al.).
- the multi-layer film articles of this disclosure comprise at least one film.
- the at least one film comprises a multi-layer film construction.
- An example of a particularly suitable multi-layer film construction is a multi-layer reflective polarizer film.
- the multilayer reflective polarizer film is a polymeric multilayer optical film comprising alternating polymeric layers configured to provide reflection for light polarized along a block axis and to transmit light polarized along a pass axis orthogonal to the block axis.
- Such films can be prepared by extruding a stack of alternating first and second types of polymeric layers and uniaxially or approximately uniaxially stretching the extruded stack to orient at least one of the first and second types of polymeric layers as generally described in U.S. Pat. No.
- the multi-layer film articles of this disclosure also comprise an absorptive polarizer film disposed on the at least one film.
- Absorbing polarizers are conventionally prepared by stretching a polyvinyl alcohol (PVA or PVOH) layer in order to align the PVA molecules in the layer and staining the aligned layer with a dichroic material such as iodine/iodide.
- the iodine/iodide molecules align with the oriented PVA molecules. Incident light polarized along the alignment direction (i.e., polarized along a block axis of the polarizer) is absorbed, or partially absorbed, by the iodine/iodide and incident light polarized along the orthogonal direction (i.e., polarized along a pass axis of the polarizer) is transmitted, or partially transmitted, through the polarizer.
- the alignment direction i.e., polarized along a block axis of the polarizer
- incident light polarized along the orthogonal direction i.e., polarized along a pass axis of the polarizer
- a polarizer which includes a modified PVA layer made from including a suitable crosslinker in PVA gives improved optical properties if the modified PVA layer is oriented with a higher degree of uniaxial orientation than that achievable using a conventional linear tenter.
- the combination of including suitable crosslinker in the PVA layer and drawing the layer by moving opposing edge portions along diverging non-linear paths (for example, using a parabolic tenter to substantially uniaxially orient the layer) to form an absorbing polarizer has been found to simultaneously achieve a low minimum transmittance of normally incident light in a wavelength range of interest and polarized along the block axis (e.g., less than 0.
- the absorbing polarizer has a polarization efficiency (PE) of at least 99.8 percent or at least 99.9 percent.
- PE polarization efficiency
- polyvinyl alcohol films are formed by coating a solution of polyvinyl alcohol onto a cast film, drying to form a coated layer, and then orienting in a tenter.
- the oriented PVA film is then stained with at least one dichroic material.
- Dichroic materials are described below.
- fully hydrolyzed (e.g. >98% hydrolyzed) grades of PVA are particularly suitable.
- An example of a suitable PVA is POVAL 28-99 from Kuraray, Houston, TX.
- the absorptive polarizer film also comprises at least one dichroic material.
- the dichroic material may include dyes, pigments, and the like.
- dichroic materials are described in E. H. Land, Colloid Chemistry (1946). Still other dichroic materials, and methods of making them, are discussed in the Kirk Othmer Encyclopedia of Chemical Technology, Vol. 8, pp. 652-661 (4 th Ed. 1993), and in the references cited therein.
- a particularly suitable dichroic material is iodine/iodide.
- polyvinyl alcohol film be crosslinked.
- crosslinked and end-capped with polyvinyl alcohol polymers are used according to their commonly understood meanings in the polymer art.
- Crosslinking and end-capping are carried out by crosslinking and end-capping agents. These agents can covalently bond with the hydroxyl groups of a polyvinyl alcohol polymer and/or interact through secondary/non-covalent interactions (e.g., hydrogen bonding) with the hydroxyl groups.
- Polyvinyl alcohol polymers contain multiple chains of polyvinyl alcohol within the polymeric mixture.
- agent reacts with a single hydroxyl group on the polyvinyl alcohol polymer, or hydroxyl groups on a single polyvinyl alcohol polymer strand, it is described as end-capping. If the agent reacts with more than one hydroxyl group and the hydroxyl groups are on different polyvinyl alcohol polymer strands, it is described as crosslinking.
- An agent may function as an end-capper, as a crosslinker, or both as an endcapper and a crosslinker. Typically, end-cappers, whether they are monofunctional or polyfunctional, reside on the surface of the polymer layer, whereas crosslinkers typically migrate into the polymer layer to form crosslinks.
- crosslinking and/or end-capping agents examples include polyphenol compounds and nano-platelet materials, a hydrogen bonding polymer, a non-phenolic polyacid, or a combination thereof.
- the materials may function as a crosslinking agent, an end-capping agent or both.
- polyphenol compounds are suitable.
- Polyphenols are compounds containing more than one phenolic hydroxyl group.
- the polyphenolic compound may be a flavonoid, a tannin, a lignan, a phenolic acid, or a combination thereof.
- the polyphenolic compound may be pyrogallol (PG), epigallocatechin-3 -gallate (EGCG), epigallocatechin (EGC), epicatechin gallate (ECG), theflavin-3 -gallate, tannic acid (TA), hydroxyhydroquinone (HHQ), catechin, morin, quercetin, naringenin, rutin, phloroglucinol, catechol, resorcinol, hydroquinone, gallic acid, ellagic acid, and/or stereoisomers thereof. Because of its availability and relatively low cost, tannic acid is a particularly suitable polyphenol.
- Nano-platelet materials are also suitable crosslinking and/or end-capping agents.
- the nano-platelet material comprises a clay platelet material.
- Clay platelet materials are layered silicates and include, for example, smectite, vermiculite, kaolin, sepiolite, glauconite, attapulgite, palygorskite, illite, chlorite, and serpentine.
- a particularly suitable clay platelet material is a synthetic smectite, such as LAPONITE, a lithium sodium magnesium silicate.
- Another suitable clay platelet material is a naturally occurring smectite, such as montmorillonite or bentonite. Another suitable clay platelet material is vermiculite.
- the nano-platelet material can be a layered double hydroxide (LDH).
- the nano-platelet material can be a 2-D material such as a transition metal dichalcogenide, hexagonal boron nitride, graphene oxide, or an Mxene.
- the nano-platelets may non-exfoliated, partially exfoliated, or fully exfoliated.
- the thickness of the individual platelets ranges from approximately 1 nm to 100 nm, and the width (or lateral dimension) of the platelets ranges from approximately 20 nm to 10,000 nm.
- Suitable hydrogen-bonding polymers include polymeric acids, especially those containing carboxylic acid groups. Examples include polymers containing acrylic acid or (meth)acrylic acid repeat units. Particularly suitable are polyacrylic acid, polymethacrylic acid, or a combination thereof.
- Suitable non-phenolic polyacids may be alpha-hydroxy acids. Examples of suitable non-phenolic polyacids include citric acid, succinic acid, tartaric acid, or a combination thereof.
- the modified film comprises an oriented polyvinyl alcohol film containing a dichroic material, where the oriented polyvinyl alcohol film has been crosslinked, end capped or both with a polyphenol compound, a nano-platelet material, a hydrogen bonding polymer, a non- phenolic polyacid, or a combination thereof.
- the modified film comprises an absorptive polarizer film as described above.
- the modified films are oriented polyvinyl alcohol films as described above.
- the films also include a dichroic material, crosslinkers and/or end-cappers as described above.
- the method of forming a multi-layer film article comprises providing at least one film, providing a film-forming solution, disposing the film-forming solution on the at least one film, drying the film-forming solution to form a composite construction comprising a polyvinyl alcohol film disposed on the at least one film, orienting the composite construction, disposing on the polyvinyl alcohol film of the composite construction a staining composition, drying the composite construction with staining composition disposed on it, disposing a crosslinker/end-capper composition on the polyvinyl alcohol film of the composite construction, drying the composite construction with crosslinker/end-capper composition disposed on it.
- the film-forming solution comprises at least one solvent; and polyvinyl alcohol.
- Suitable solvents include water, and water miscible solvents such as alcohols.
- Suitable polyvinyl alcohols are described above.
- the staining composition comprises at least one dichroic material and at least one solvent.
- Dichroic materials are described above.
- a particularly suitable dichroic material is iodine/iodide.
- suitable solvents include water, and water miscible solvents such as alcohols.
- the crosslinker/end-capper composition comprises a crosslinker, an end-capper or both, and at least one solvent.
- Suitable crosslinker or end-capper agents are polyphenol compounds, nano-platelet materials or a combination thereof.
- the crosslinker/end-capper composition may also comprise the optional crosslinker/end-capper agents, of hydrogen- bonding polymers and non-phenolic polyacids as described above.
- suitable solvents include water, and water miscible solvents such as alcohols.
- the method further comprises one or more rinsing steps.
- the method may comprise one of these rinsing steps, some of these rinsing steps, or may comprise all of the rinsing steps.
- the method further comprises rinsing the composite construction after disposing the staining composition on the polyvinyl alcohol film of the composite construction.
- the rinsing solution used for this rinsing step involves a solvent.
- suitable solvents include water, and water miscible solvents such as alcohols.
- the method further comprises rinsing the composite construction after disposing the crosslinker/end-capper composition on the polyvinyl alcohol film of the composite construction.
- the rinsing solution used for this rinsing step involves a solvent.
- suitable solvents include water, and water miscible solvents such as alcohols.
- the method further comprises rinsing the composite construction after disposing the staining composition on the polyvinyl alcohol film of the composite construction, and rinsing the composite construction after disposing the crosslinker/end-capper composition on the polyvinyl alcohol film of the composite construction.
- the rinsing solution used for this rinsing step involves a solvent.
- suitable solvents include water, and water miscible solvents such as alcohols.
- modified film layer 120 is an absorptive polarizer comprising an oriented polyvinyl alcohol film containing a dichroic material, wherein the oriented polyvinyl alcohol film has been crosslinked, end capped or both as described above.
- Figure 2 shows another article 200, comprising multilayer film 210, wherein multilayer film 210 is a reflective polarizer, and modified film layer 220, where modified film layer 220 is an absorptive polarizer comprising an oriented polyvinyl alcohol film containing a dichroic material, wherein the oriented polyvinyl alcohol film has been crosslinked, end capped or both as described above.
- multilayer film 210 is a reflective polarizer
- modified film layer 220 is an absorptive polarizer comprising an oriented polyvinyl alcohol film containing a dichroic material, wherein the oriented polyvinyl alcohol film has been crosslinked, end capped or both as described above.
- Visible light transmission was measured with a Haze-Gard Plus instrument (BYK, Geretsried, Germany). Samples were measured with the reflective side of the film facing the light source.
- Edge ingress was measured using a radiant imaging PROMETRIC 18 Colorimeter (Radiant Vision Systems, Redmon, WA) to image the samples under cross polarization. Image J software (National Institutes of Health) was then used to calculate the % area loss of the iodine/iodide stain for each sample image.
- PREPARATORY EXAMPLE 1 PREPARATION OF ORIENTED POLYVINYL ALCOHOL ON A MULTI-LAYER REFLECTIVE POLARIZER
- a 10% solids solution of POVAL 28-99 grade polyvinyl alcohol (PVOH) was prepared in water by first charging a temperature-controlled kettle with water at room temperature. Under agitation, the PVOH resin was added. The mixture was heated to 90- 105°C and maintained at this temperature for 3 hours under constant agitation. The solution was allowed to cool and drained from the kettle. To the cooled solution, surfactant was added at 0.1% of solution. Formaldehyde-adduct type crosslinker was added at 5-40% concentration, based upon PVOH resin solids, with mixing. Optionally isopropyl alcohol is incorporated into the PVOH dissolution process at a concentration up to 15%.
- PVOH polyvinyl alcohol
- a birefringent reflective polarizer was prepared as follows. A single multilayer optical packet was co-extruded as described in US Patent Application 2011/0102891, titled ‘Low Layer Count Reflective Polarizer with Optimized Gain’. Two polymers were used for the optical layers.
- the first polymer (first optical layers) was polyethylene naphthalate (PEN) homopolymer (100 mol % naphthalene dicarboxylate with 100 mol % ethylene glycol) having a Tg of 121-123°C.
- PEN polyethylene naphthalate
- the second polymer was a blend of a first polyethylene naphthalate copolymer (coPEN) having 90 mol % naphthalate and 10 mol % of the copolyester polyethylene terephthalate glycol (PETG) at a ratio of approximately 45 mol % 90/10 PEN to 55 mol% PETG where the second optical layer material Tg is approximately 97-100°C.
- the polymer used for the skin layers was the same as that used for the second polymer layers.
- These polyesters can be formed, for example, as described in U.S. Pat. No. 6,352,761.
- the materials were fed from separate extruders to a multilayer coextrusion feedblock, in which they were assembled into a packet of 305 alternating optical layers, plus a thicker protective boundary layer of the second optical layers, on each side, for a total of 307 layers.
- the skin layers of the second optical layer material were added to the construction in a manifold specific to that purpose, resulting in a final construction having 307 layers.
- the multilayer melt was then cast through a film die onto a chill roll, in the conventional manner for polyester films, upon which it was quenched.
- a polyvinyl alcohol (PVOH) coating was adhered. Prior to coating, the cast film was corona treated. The solvent was removed at elevated temperatures (85 °C for 45 seconds). Following coating, the coated cast web was stretched in a standard tenter as described in U.S. Pat. No. 5,882,774 (Jonza et al.).
- the resultant integrated polarizer precursor film had a physical thickness, as measured by a capacitance gauge Model PR2000 by SolveTech (Wilmington, DE), of approximately 40 micrometers including a PVOH layer thickness of approximately 1 micrometer.
- PREPARATORY EXAMPLE 3 PREPARATION OF BORATE/BORIC ACID CROSSLINKER SOLUTIONS
- PREPARATORY EXAMPLE 4 PREPARATION OF CROSSLINKER/END- CAPPER SOLUTIONS
- TA, GA, and CA solutions were prepared by dissolving 10 g of TA, 10 g of GA, and 10 g of CA, each in 950 g of de-ionized (DI) water in plastic bottles. The plastic bottles were shaken vigorously by hand and placed on a roller overnight.
- DI de-ionized
- a PAA solution was made by adding 40 g of ACUMER 1510 to 960 g of DI water in a plastic bottle. The plastic bottle was shaken vigorously by hand and placed on a roller overnight.
- a LapRD clay suspension was made by adding 10 g of LAPONITE RD powder to a plastic bottle with 950 g of DI water. The plastic bottle was shaken vigorously by hand and placed on a roller overnight.
- a VMT clay suspension was made by adding 118 g of MICROLITE 963++ to 882 g of DI water in a plastic bottle. The plastic bottle was shaken vigorously by hand.
- the substrate was then dried with an airknife at a speed of 10 mm/sec. This stain solution spray and airknife dry sequence was repeated five times.
- DI water was sprayed onto the substrate while the stage moved vertically upward at 102 mm/sec.
- the substrate was then dried with an airknife at a speed of 10 mm/sec.
- a crosslinker/end-capper solution was sprayed onto the substrate while the stage moved vertically downward at 76 millimeter/second (mm/sec).
- DI water was sprayed onto the substrate while the stage moved vertically upward at 102 mm/sec.
- the substrate was then dried with an airknife at a speed of 10 mm/sec. The film was stripped off of the glass prior to subsequent processing.
- Sheets of film made in PE-1 were cut to a size of 12 inch x 12 inch (30 cm x 30 cm) and then sequentially stained and crosslinked/end-capped using the “METHOD FOR APPLYING STAINING SOLUTION AND CROSSLINKER/END-CAPPER SOLUTIONS”. The corresponding crosslinkers/end-cappers are listed in Table 3.
- COMPARATIVE EXAMPLES 1-4 BORATE/BORIC ACID CROSSLINKING
- Sheets of fdm made in PE-1 were cut to a size of 12 inch x 12 inch (30 cm x 30 cm) and then stained using the “METHOD FOR APPLYING STAINING SOLUTION AND CROSSLINKER/END-CAPPER SOLUTIONS”.
- Crosslinker solutions were prepared as described in PE-3.
- the stained fdms were exposed to crosslinker solution at conditions shown in Table 2.
- the crosslinker solution was poured into a glass crystallization dish. A section of fdm was manually immersed in the solution with tweezers for the 42 sec, rinsed with copious amounts of DI water, and dried with an air gun.
- Sheets of fdm made in PE-1 were cut to a size of 12 inch x 12 inch (30 cm x 30 cm) and then stained using the “METHOD FOR APPLYING STAINING SOLUTION AND CROSSLINKER/END-CAPPER SOLUTIONS”. No crosslinking was carried out.
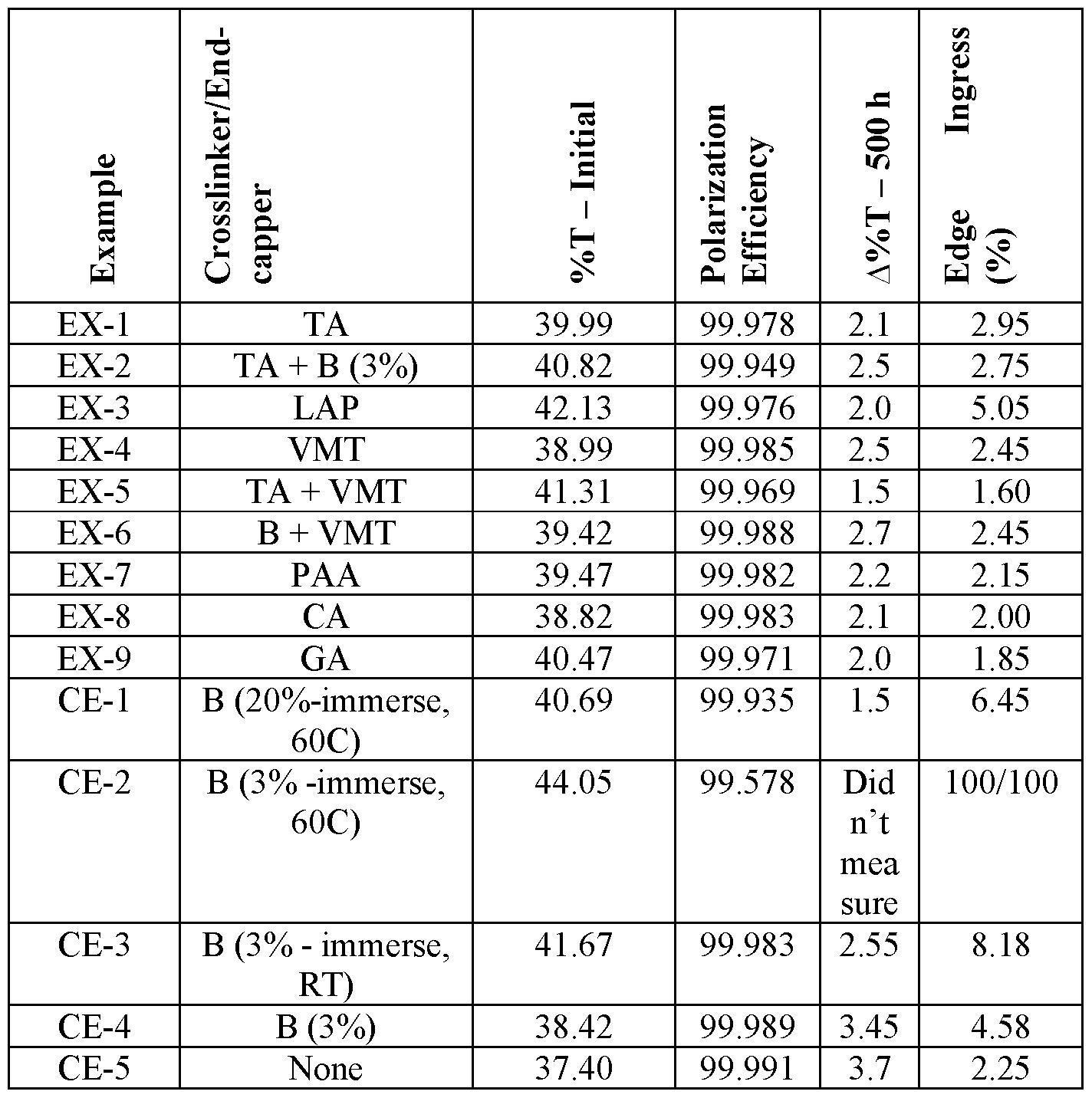
- TABLE 3 Visible light transmission (%T), change in visible light transmission after 500 hours environmental aging (A%T-500 h)), and edge ingress (%) for EX-1 to EX-9 and CE- 1 to CE-5.
- Crosslinking/end-capping were carried out at room temperature (RT) unless otherwise specified.
- the Comparative Examples all have one or more of lower polarization efficiency, greater A%T-500 h, and greater edge ingress compared to the Examples.
Landscapes
- Physics & Mathematics (AREA)
- General Physics & Mathematics (AREA)
- Optics & Photonics (AREA)
- Laminated Bodies (AREA)
Abstract
Dichroic polarizer films include at least one film that may be a multi-layer reflective polarizer film and an absorptive polarizer film. The absorptive polarizer film is a modified film. The modified film is an oriented polyvinyl alcohol film containing a dichroic material, where the oriented polyvinyl alcohol film has been crosslinked, end capped or both with a polyphenol compound, a nano-platelet material, a hydrogen bonding polymer, a non-phenolic polyacid, or a combination thereof.
Description
DICHROIC POLARIZER FILM
Summary
Disclosed herein are modified films, multi-layer film articles that include the modified films, and methods for forming the multi-layer film articles. The modified films are absorptive polarizers. In some embodiments, the modified film comprises an oriented polyvinyl alcohol film containing a dichroic material, wherein the oriented polyvinyl alcohol film has been crosslinked, end capped or both with a polyphenol compound, a nano-platelet material, a hydrogen bonding polymer, a non-phenolic polyacid, or a combination thereof.
In some embodiments, the multi-layer film articles comprise at least one film; and an absorptive polarizer film disposed on the at least one film. The absorptive polarizer film comprises a modified film as described above.
Also disclosed are methods of forming multi-layer film articles comprising providing at least one film, providing a film-forming solution, where the film-forming solution comprises at least one solvent and polyvinyl alcohol, disposing the film-forming solution on the at least one film, drying the film-forming solution to form a composite construction. The composite construction comprises a polyvinyl alcohol film disposed on the film. The composite construction is oriented, a staining composition is applied to the composite construction, where the staining composition comprises at least one dichroic material and at least one solvent. The composite construction with staining composition disposed on it is dried and a crosslinker/end-capper composition is applied to the polyvinyl alcohol film of the composite construction. The crosslinker/end-capper composition comprises a crosslinker, an end-capper or both, where the crosslinker or endcapper comprises a polyphenol compound, a nano-platelet material a hydrogen bonding polymer, a non-phenolic polyacid, or a combination thereof, and at least one solvent. The composite construction with crosslinker/end-capper composition disposed on it is dried.
Brief Description of the Drawings
The present application may be more completely understood in consideration of the following detailed description of various embodiments of the disclosure in connection with the accompanying drawings.
Figure 1 is a cross sectional view of an article of the current disclosure.
Figure 2 is a cross sectional view of another article of the current disclosure.
In the following description of the illustrated embodiments, reference is made to the accompanying drawings, in which is shown by way of illustration, various embodiments in which the disclosure may be practiced. It is to be understood that the embodiments may be utilized and structural changes may be made without departing from the scope of the present disclosure. The figures are not necessarily to scale. Like numbers used in the figures refer to like components. However, it will be understood that the use of a number to refer to a component in a given figure is not intended to limit the component in another figure labeled with the same number.
Detailed Description
Optical polarizing films are widely used in optical applications. Optical polarizing films are used for example to reduce glare in sunglasses and for increasing optical contrast in Liquid Crystal Displays (LCD). There are generally two types of polarizers that are used. One type of polarizer is a dichroic polarizer, also known as an absorbing or absorptive polarizer. Dichroic polarizers absorb light of one polarization and transmit light of the orthogonal polarization. Dichroic polarizers can be prepared by incorporating a dichroic material such as a dye into a polymer sheet that is stretched in at least one direction. Dichroic polarizers can also be made by uniaxially stretching a semicrystalline polymer such a polyvinyl alcohol, then staining the polymer with a dichroic material. Alternatively, dichroic polarizers can be prepared by coating a polymer with oriented dichroic material. Dichroic materials include anthraquinone and azo dyes as well as iodine/iodide.
Another type of polarizer is a reflective polarizer. Reflective polarizers reflect light of one polarization and transmit light of the orthogonal polarization. One type of reflective polarizer is made by forming a stack of alternating polymer layers.
The two types of polarizers may be combined to make a single optical polarizer, thereby incorporating the useful characteristics of both types of polarizers. The need remains for dichroic polarizer fdms, methods for preparing these fdms, as well as methods of forming single optical polarizers that contain a reflective polarizer and a dichroic polarizer. Among the desirable goals are the simplification of the process to form the dichroic polarizer films and the single optical polarizers. Also, typically these films are prepared from polyvinyl alcohol that is crosslinked with boric acid. Boric acid is a hazardous material and requires special handling, so it is desirable to develop other crosslinking and/or end-capping agents for use with polyvinyl alcohol.
In this disclosure, multi-layer film articles are described as well as methods for preparing multi-layer film articles. Among the multi-layer film articles are ones that include a reflective polarizer construction in combination with a dichroic absorptive polarizer. Also disclosed are modified films, where the modified films contain a dichroic material and can be absorptive polarizers.
The terms "room temperature” and “ambient temperature” are used interchangeably to mean temperatures in the range of 20°C to 25°C.
The terms “Tg” and “glass transition temperature” are used interchangeably. If measured, Tg values are determined by Differential Scanning Calorimetry (DSC) at a scan rate of 10°C/minute, unless otherwise indicated. Typically, Tg values for copolymers are not measured but are calculated using the well-known Fox Equation, using the monomer Tg values provided by the monomer supplier, as is understood by one of skill in the art.
The term “adjacent” as used herein when referring to two layers means that the two layers are in proximity with one another with no intervening open space between them. They may be in direct contact with one another (e.g., laminated together) or there may be intervening layers.
The terms “polymer” and “macromolecule” are used herein consistent with their common usage in chemistry. Polymers and macromolecules are composed of many repeated subunits. As used herein, the term “macromolecule” is used to describe a group attached to a monomer that has multiple repeating units. The term “polymer” is used to describe the resultant material formed from a polymerization reaction.
The terms “crosslinking” and “end-capping” of polyvinyl alcohol polymers are used according to their commonly understood meanings in the polymer art. Crosslinking
and end-capping are carried out by crosslinking and end-capping agents. These agents can covalently bond with the hydroxyl groups of a polyvinyl alcohol polymer and/or interact through secondary/non-covalent interactions (e.g., hydrogen bonding) with the hydroxyl groups. Polyvinyl alcohol polymers contain multiple chains of polyvinyl alcohol within the polymeric mixture. If the agent reacts with a single hydroxyl group on the polyvinyl alcohol polymer, or hydroxyl groups on a single polyvinyl alcohol polymer strand, it is described as end-capping. If the agent reacts with more than one hydroxyl group and the hydroxyl groups are on different polyvinyl alcohol polymer strands, it is described as crosslinking. An agent may function as an end-capper, as a crosslinker, or both as an endcapper and a crosslinker. Typically, end-cappers, whether they are monofunctional or polyfunctional, reside on the surface of the polymer layer, whereas crosslinkers typically migrate into the polymer layer to form crosslinks.
The term “hydrogen bonding” is used herein according to its well understood meaning. Hydrogen bonding is not covalent or ionic bonding but rather is an interaction involving a weak physical bond between two molecules resulting from an electrostatic attraction between a proton in one molecule and an electronegative atom (such as oxygen) in the other.
The term “dichroic material” as used herein refers to materials that are dichroic and include dichroic dyes as well as dichroic materials such as iodine/iodide.
The terms “iodine” and “iodine/iodide” are used interchangeably when referring to dichroic staining solutions, as is understood by one of skill in the optical arts.
Unless otherwise indicated, the terms “optically transparent”, and “visible light transmissive” are used interchangeably, and refer to an article, film or adhesive that has a high light transmittance over at least a portion of the visible light spectrum (about 400 to about 700 nm). Typically, optically transparent articles have a visible light transmittance of at least 90% and a haze of less than 10%.
Unless otherwise indicated, “optically clear” refers to an adhesive or article that has a high light transmittance over at least a portion of the visible light spectrum (about 400 to about 700 nm), and that exhibits low haze, typically less than about 5%, or even less than about 2%. In some embodiments, optically clear articles exhibit a haze of less than 1% at a thickness of 50 micrometers or even 0.5% at a thickness of 50 micrometers.
Typically, optically clear articles have a visible light transmittance of at least 95%, often higher such as 97%, 98% or even 99% or higher.
Disclosed herein are multi-layer film articles. In some embodiments, the multilayer film articles comprise at least one film, and an absorptive polarizer film disposed on the at least one film. In some embodiments, the absorptive polymer film comprises an oriented polyvinyl alcohol film containing a dichroic material, where the oriented polyvinyl alcohol film has been crosslinked, end capped or both by a polyphenol compound, a nano-platelet material, a hydrogen bonding polymer, a non-phenolic polyacid, or a combination thereof. Each of these materials is described in detail below.
In many embodiments, the multi-layer film articles are polarizers. As will be described in detail below, the polyvinyl alcohol film can be an absorptive polarizer. In some embodiments, the at least one film layer is a reflective polarizer. In some cases, it may be desirable to include both an absorbing polarizer and a reflective polarizer in a display application. The reflective polarizer can be used for polarization recycling in a liquid crystal display application, for example, and an absorbing polarizer can be added to the reflective polarizer to improve extinction efficiency. Integrating an absorbing polarizer with a reflective polarizer has been described in U.S. Pat. Nos. 6,096,375 (Ouderkirk et al.), 6,697,195 (Weber et al.), 7,826,009 (Weber et al.), and 6,111,697 (Kausch et al.).
The multi-layer film articles of this disclosure comprise at least one film. In many embodiments, the at least one film comprises a multi-layer film construction. An example of a particularly suitable multi-layer film construction is a multi-layer reflective polarizer film. The multilayer reflective polarizer film is a polymeric multilayer optical film comprising alternating polymeric layers configured to provide reflection for light polarized along a block axis and to transmit light polarized along a pass axis orthogonal to the block axis. Such films can be prepared by extruding a stack of alternating first and second types of polymeric layers and uniaxially or approximately uniaxially stretching the extruded stack to orient at least one of the first and second types of polymeric layers as generally described in U.S. Pat. No. 5,882,774 (Jonza et al.), for example. Parabolic stretchers or tenter machines such as those described in U.S. Pat. No. 6,916,440 (Jackson et al.), for example, have been used to improve the degree of uniaxial orientation of the birefringent layers in the reflective polarizer.
The multi-layer film articles of this disclosure also comprise an absorptive polarizer film disposed on the at least one film. Absorbing polarizers are conventionally prepared by stretching a polyvinyl alcohol (PVA or PVOH) layer in order to align the PVA molecules in the layer and staining the aligned layer with a dichroic material such as iodine/iodide. The iodine/iodide molecules align with the oriented PVA molecules. Incident light polarized along the alignment direction (i.e., polarized along a block axis of the polarizer) is absorbed, or partially absorbed, by the iodine/iodide and incident light polarized along the orthogonal direction (i.e., polarized along a pass axis of the polarizer) is transmitted, or partially transmitted, through the polarizer.
It has been found that a polarizer which includes a modified PVA layer made from including a suitable crosslinker in PVA gives improved optical properties if the modified PVA layer is oriented with a higher degree of uniaxial orientation than that achievable using a conventional linear tenter. In particular, the combination of including suitable crosslinker in the PVA layer and drawing the layer by moving opposing edge portions along diverging non-linear paths (for example, using a parabolic tenter to substantially uniaxially orient the layer) to form an absorbing polarizer has been found to simultaneously achieve a low minimum transmittance of normally incident light in a wavelength range of interest and polarized along the block axis (e.g., less than 0. 1 percent) and a high maximum transmittance of normally incident light in the wavelength range of interest and polarized along the pass axis (e.g., greater than 75 percent). The wavelength range of interest may be the visible range (400 nm to 700 nm) or may be 540 nm to 640 nm, for example. In some embodiments, the absorbing polarizer has a polarization efficiency (PE) of at least 99.8 percent or at least 99.9 percent. The polarization efficiency is given by Equation 1 :
(Equation 1) where MaxPass is the maximum pass state transmission at normal incidence over the wavelength range of interest and MinBklck is the minimum block state transmission at normal incidence over the wavelength range of interest.
In this disclosure, polyvinyl alcohol films are formed by coating a solution of polyvinyl alcohol onto a cast film, drying to form a coated layer, and then orienting in a tenter. The oriented PVA film is then stained with at least one dichroic material. Dichroic
materials are described below. For high polarization efficiency, fully hydrolyzed (e.g. >98% hydrolyzed) grades of PVA are particularly suitable. An example of a suitable PVA is POVAL 28-99 from Kuraray, Houston, TX.
The absorptive polarizer film also comprises at least one dichroic material. A wide range of dichroic materials are suitable. The dichroic material may include dyes, pigments, and the like. Suitable materials for use in the dichroic polarizer film include, for example, iodine/iodide, as well as anthraquinone and azo dyes, such as Congo Red (sodium diphenyl-bis-a-naphthylamine sulfonate), methylene blue, stilbene dye (Color Index (CI)=620), and 1,1 '-diethyl-2, 2'-cyanine chloride (374 (orange) or 518 (blue)). The properties of these materials, and methods of making them, are described in E. H. Land, Colloid Chemistry (1946). Still other dichroic materials, and methods of making them, are discussed in the Kirk Othmer Encyclopedia of Chemical Technology, Vol. 8, pp. 652-661 (4th Ed. 1993), and in the references cited therein. A particularly suitable dichroic material is iodine/iodide.
As mentioned above, it is desirable that the polyvinyl alcohol film be crosslinked. In this disclosure it has been discovered that it is desirable for the oriented polyvinyl alcohol film be crosslinked, end-capped or a combination thereof. The terms “crosslinked” and “end-capped” with polyvinyl alcohol polymers are used according to their commonly understood meanings in the polymer art. Crosslinking and end-capping are carried out by crosslinking and end-capping agents. These agents can covalently bond with the hydroxyl groups of a polyvinyl alcohol polymer and/or interact through secondary/non-covalent interactions (e.g., hydrogen bonding) with the hydroxyl groups. Polyvinyl alcohol polymers contain multiple chains of polyvinyl alcohol within the polymeric mixture. If the agent reacts with a single hydroxyl group on the polyvinyl alcohol polymer, or hydroxyl groups on a single polyvinyl alcohol polymer strand, it is described as end-capping. If the agent reacts with more than one hydroxyl group and the hydroxyl groups are on different polyvinyl alcohol polymer strands, it is described as crosslinking. An agent may function as an end-capper, as a crosslinker, or both as an endcapper and a crosslinker. Typically, end-cappers, whether they are monofunctional or polyfunctional, reside on the surface of the polymer layer, whereas crosslinkers typically migrate into the polymer layer to form crosslinks.
Examples of crosslinking and/or end-capping agents include polyphenol compounds and nano-platelet materials, a hydrogen bonding polymer, a non-phenolic polyacid, or a combination thereof. The materials may function as a crosslinking agent, an end-capping agent or both.
A number of polyphenol compounds are suitable. Polyphenols are compounds containing more than one phenolic hydroxyl group. In some embodiments, the polyphenolic compound may be a flavonoid, a tannin, a lignan, a phenolic acid, or a combination thereof. In some embodiments, the polyphenolic compound may be pyrogallol (PG), epigallocatechin-3 -gallate (EGCG), epigallocatechin (EGC), epicatechin gallate (ECG), theflavin-3 -gallate, tannic acid (TA), hydroxyhydroquinone (HHQ), catechin, morin, quercetin, naringenin, rutin, phloroglucinol, catechol, resorcinol, hydroquinone, gallic acid, ellagic acid, and/or stereoisomers thereof. Because of its availability and relatively low cost, tannic acid is a particularly suitable polyphenol.
Nano-platelet materials are also suitable crosslinking and/or end-capping agents. In some embodiments, the nano-platelet material comprises a clay platelet material. Clay platelet materials are layered silicates and include, for example, smectite, vermiculite, kaolin, sepiolite, glauconite, attapulgite, palygorskite, illite, chlorite, and serpentine. A particularly suitable clay platelet material is a synthetic smectite, such as LAPONITE, a lithium sodium magnesium silicate. Another suitable clay platelet material is a naturally occurring smectite, such as montmorillonite or bentonite. Another suitable clay platelet material is vermiculite. In some embodiments, the nano-platelet material can be a layered double hydroxide (LDH). In some embodiments, the nano-platelet material can be a 2-D material such as a transition metal dichalcogenide, hexagonal boron nitride, graphene oxide, or an Mxene. The nano-platelets may non-exfoliated, partially exfoliated, or fully exfoliated. In some embodiments, the thickness of the individual platelets ranges from approximately 1 nm to 100 nm, and the width (or lateral dimension) of the platelets ranges from approximately 20 nm to 10,000 nm.
Suitable hydrogen-bonding polymers include polymeric acids, especially those containing carboxylic acid groups. Examples include polymers containing acrylic acid or (meth)acrylic acid repeat units. Particularly suitable are polyacrylic acid, polymethacrylic acid, or a combination thereof.
Suitable non-phenolic polyacids may be alpha-hydroxy acids. Examples of suitable non-phenolic polyacids include citric acid, succinic acid, tartaric acid, or a combination thereof.
Also disclosed herein are modified films. In some embodiments, the modified film comprises an oriented polyvinyl alcohol film containing a dichroic material, where the oriented polyvinyl alcohol film has been crosslinked, end capped or both with a polyphenol compound, a nano-platelet material, a hydrogen bonding polymer, a non- phenolic polyacid, or a combination thereof. In some embodiments, the modified film comprises an absorptive polarizer film as described above.
The modified films are oriented polyvinyl alcohol films as described above. The films also include a dichroic material, crosslinkers and/or end-cappers as described above.
Also disclosed herein are methods for preparing multi-layer film articles. In some embodiments, the method of forming a multi-layer film article comprises providing at least one film, providing a film-forming solution, disposing the film-forming solution on the at least one film, drying the film-forming solution to form a composite construction comprising a polyvinyl alcohol film disposed on the at least one film, orienting the composite construction, disposing on the polyvinyl alcohol film of the composite construction a staining composition, drying the composite construction with staining composition disposed on it, disposing a crosslinker/end-capper composition on the polyvinyl alcohol film of the composite construction, drying the composite construction with crosslinker/end-capper composition disposed on it.
The film-forming solution comprises at least one solvent; and polyvinyl alcohol. Suitable solvents include water, and water miscible solvents such as alcohols. Suitable polyvinyl alcohols are described above.
The staining composition comprises at least one dichroic material and at least one solvent. Dichroic materials are described above. A particularly suitable dichroic material is iodine/iodide. Examples of suitable solvents include water, and water miscible solvents such as alcohols.
The crosslinker/end-capper composition comprises a crosslinker, an end-capper or both, and at least one solvent. Suitable crosslinker or end-capper agents are polyphenol compounds, nano-platelet materials or a combination thereof. The crosslinker/end-capper composition may also comprise the optional crosslinker/end-capper agents, of hydrogen-
bonding polymers and non-phenolic polyacids as described above. Examples of suitable solvents include water, and water miscible solvents such as alcohols.
In some embodiments, the method further comprises one or more rinsing steps. The method may comprise one of these rinsing steps, some of these rinsing steps, or may comprise all of the rinsing steps.
In some embodiments the method further comprises rinsing the composite construction after disposing the staining composition on the polyvinyl alcohol film of the composite construction. The rinsing solution used for this rinsing step involves a solvent. Examples of suitable solvents include water, and water miscible solvents such as alcohols.
In some embodiments, the method further comprises rinsing the composite construction after disposing the crosslinker/end-capper composition on the polyvinyl alcohol film of the composite construction. The rinsing solution used for this rinsing step involves a solvent. Examples of suitable solvents include water, and water miscible solvents such as alcohols.
In some embodiments, the method further comprises rinsing the composite construction after disposing the staining composition on the polyvinyl alcohol film of the composite construction, and rinsing the composite construction after disposing the crosslinker/end-capper composition on the polyvinyl alcohol film of the composite construction. The rinsing solution used for this rinsing step involves a solvent. Examples of suitable solvents include water, and water miscible solvents such as alcohols.
The Figures illustrate some embodiments of the current disclosure. In Figure 1, article 100 is shown comprising film 110 and modified film layer 120. In this embodiment, modified film layer 120 is an absorptive polarizer comprising an oriented polyvinyl alcohol film containing a dichroic material, wherein the oriented polyvinyl alcohol film has been crosslinked, end capped or both as described above.
Figure 2 shows another article 200, comprising multilayer film 210, wherein multilayer film 210 is a reflective polarizer, and modified film layer 220, where modified film layer 220 is an absorptive polarizer comprising an oriented polyvinyl alcohol film containing a dichroic material, wherein the oriented polyvinyl alcohol film has been crosslinked, end capped or both as described above.
Examples
Unless otherwise noted, all parts, percentages, ratios, etc. in the Examples and the rest of the specification are by weight. Unless otherwise indicated, all other reagents were obtained, or are available from fine chemical vendors such as Sigma-Aldrich Company, St. Eouis, Missouri, or may be synthesized by known methods. The following abbreviations are used: cm = centimeters; mm = millimeters; min = minutes; sec = seconds; h = hours; psi = pounds per square inch; MPa = megaPascals. The terms “weight %”, “% by weight”, and “wt%” are used interchangeably. Table 1 (below) lists materials used in the examples and their sources.
Test Methods
TEST METHOD 1: HEAT/HUMIDITY STABILITY TESTING
Film was laminated to CORNING EAGLE XG Display Glass with a barrier adhesive (described in US 8,232,350 (Fujita)) and put into a chamber at 60°C / 90% RH for durations of 24, 250, 500, and 1000 hours.
TEST METHOD 2: MEASURING THE VISIBLE LIGHT TRANSMISSION AND HAZE OF A COATED FILM
Visible light transmission (%T) was measured with a Haze-Gard Plus instrument (BYK, Geretsried, Germany). Samples were measured with the reflective side of the film facing the light source.
TEST METHOD 3: EDGE INGRESS
Edge ingress was measured using a radiant imaging PROMETRIC 18 Colorimeter (Radiant Vision Systems, Redmon, WA) to image the samples under cross polarization. Image J software (National Institutes of Health) was then used to calculate the % area loss of the iodine/iodide stain for each sample image.
Examples
PREPARATORY EXAMPLE 1 (PE-1): PREPARATION OF ORIENTED POLYVINYL ALCOHOL ON A MULTI-LAYER REFLECTIVE POLARIZER
A 10% solids solution of POVAL 28-99 grade polyvinyl alcohol (PVOH) was prepared in water by first charging a temperature-controlled kettle with water at room temperature. Under agitation, the PVOH resin was added. The mixture was heated to 90- 105°C and maintained at this temperature for 3 hours under constant agitation. The solution was allowed to cool and drained from the kettle. To the cooled solution, surfactant was added at 0.1% of solution. Formaldehyde-adduct type crosslinker was added at 5-40% concentration, based upon PVOH resin solids, with mixing. Optionally
isopropyl alcohol is incorporated into the PVOH dissolution process at a concentration up to 15%.
A birefringent reflective polarizer was prepared as follows. A single multilayer optical packet was co-extruded as described in US Patent Application 2011/0102891, titled ‘Low Layer Count Reflective Polarizer with Optimized Gain’. Two polymers were used for the optical layers. The first polymer (first optical layers) was polyethylene naphthalate (PEN) homopolymer (100 mol % naphthalene dicarboxylate with 100 mol % ethylene glycol) having a Tg of 121-123°C. The second polymer (second optical layers) was a blend of a first polyethylene naphthalate copolymer (coPEN) having 90 mol % naphthalate and 10 mol % of the copolyester polyethylene terephthalate glycol (PETG) at a ratio of approximately 45 mol % 90/10 PEN to 55 mol% PETG where the second optical layer material Tg is approximately 97-100°C. The polymer used for the skin layers was the same as that used for the second polymer layers. These polyesters can be formed, for example, as described in U.S. Pat. No. 6,352,761. The materials were fed from separate extruders to a multilayer coextrusion feedblock, in which they were assembled into a packet of 305 alternating optical layers, plus a thicker protective boundary layer of the second optical layers, on each side, for a total of 307 layers. The skin layers of the second optical layer material were added to the construction in a manifold specific to that purpose, resulting in a final construction having 307 layers. The multilayer melt was then cast through a film die onto a chill roll, in the conventional manner for polyester films, upon which it was quenched.
To the cast web, a polyvinyl alcohol (PVOH) coating was adhered. Prior to coating, the cast film was corona treated. The solvent was removed at elevated temperatures (85 °C for 45 seconds). Following coating, the coated cast web was stretched in a standard tenter as described in U.S. Pat. No. 5,882,774 (Jonza et al.). The resultant integrated polarizer precursor film had a physical thickness, as measured by a capacitance gauge Model PR2000 by SolveTech (Wilmington, DE), of approximately 40 micrometers including a PVOH layer thickness of approximately 1 micrometer.
PREPARATORY EXAMPLE 2 (PE-2): PREPARATION OF STAINING SOLUTIONS
The iodine/iodide staining solutions were prepared at 20% solids using a 60: 1 KI: h ratio. DI water was used and heated to 30°C to allow the potassium iodide and iodine/iodide to dissolve. It was then brought back to room temperature for use.
PREPARATORY EXAMPLE 3 (PE-3): PREPARATION OF BORATE/BORIC ACID CROSSLINKER SOLUTIONS
Two different boration solutions were prepared, 3% solids (abbreviated B(3%)) and 20% solids (abbreviated B(20%)). Both used a 2.33: 1 ratio of boric acid to borax. Both were heated to 60°C to dissolve solids in solution. The 3% solids solution remains in solution at room temperature, but the 20% solids solution requires heating for solids to stay in solution.
PREPARATORY EXAMPLE 4 (PE-4): PREPARATION OF CROSSLINKER/END- CAPPER SOLUTIONS
Separate TA, GA, and CA solutions were prepared by dissolving 10 g of TA, 10 g of GA, and 10 g of CA, each in 950 g of de-ionized (DI) water in plastic bottles. The plastic bottles were shaken vigorously by hand and placed on a roller overnight.
A PAA solution was made by adding 40 g of ACUMER 1510 to 960 g of DI water in a plastic bottle. The plastic bottle was shaken vigorously by hand and placed on a roller overnight.
A LapRD clay suspension was made by adding 10 g of LAPONITE RD powder to a plastic bottle with 950 g of DI water. The plastic bottle was shaken vigorously by hand and placed on a roller overnight.
A VMT clay suspension was made by adding 118 g of MICROLITE 963++ to 882 g of DI water in a plastic bottle. The plastic bottle was shaken vigorously by hand.
METHOD FOR APPLYING STAINING SOLUTION AND CROSSLINKER/END- CAPPER SOLUTIONS
Staining solution and crosslinker/end-capper solutions were applied to the oriented polyvinyl alcohol coated fdm (from PE-1) using an apparatus purchased from Svaya Nanotechnologies, Inc. (Sunnyvale, CA) and modeled after the system described in US
Pat. No. 8,234,998 (Krogman et al.) as well as Krogman et al. Automated Process for Improved Uniformity and Versatility of Layer-by-Layer Deposition, Langmuir 2007, 23, 3137-3141. The apparatus comprises pressure vessels loaded with the solutions. Spray nozzles with a flat spray pattern (from Spraying Systems, Inc., Wheaton, IL) were mounted to spray the coating solutions and rinse water at specified times, controlled by solenoid valves. The pressure vessels (Alloy Products Corp., Waukesha, WI) containing the solutions were pressurized with nitrogen to 30 pounds per square inch (psi) (0.21 MPa), while the pressure vessel containing DI water was pressurized with air to 30 psi (0.21 MPa). Flow rates from the solution nozzles were each 10 gallons per hour (38 Liters/hour), while flow rate from the DI water rinse nozzles were 40 gallons per hour (150 Liters/hour). The substrate to be coated (12 inch x 12 inch) (30 cm x 30 cm) was adhered at the edges with epoxy (Scotch-Weld epoxy adhesive, DP 100 Clear, 3M Company, St. Paul, MN) to a glass plate (12 inch x 12 inch x 1/8 inch thick) (30 cm x 30 cm x 0.3 cm) (Brin Northwestern Glass Co., Minneapolis, MN), which was mounted on a vertical translation stage and held in place with a vacuum chuck. In a typical staining and crosslinking/end-capping sequence, the staining solution was sprayed onto the substrate while the stage moved vertically downward at 76 millimeter/second (mm/sec). Next, after a dwell time of 12 sec, the substrate was then dried with an airknife at a speed of 10 mm/sec. This stain solution spray and airknife dry sequence was repeated five times. Next, DI water was sprayed onto the substrate while the stage moved vertically upward at 102 mm/sec. The substrate was then dried with an airknife at a speed of 10 mm/sec. Next, a crosslinker/end-capper solution was sprayed onto the substrate while the stage moved vertically downward at 76 millimeter/second (mm/sec). Next, after a dwell time of 12 sec, DI water was sprayed onto the substrate while the stage moved vertically upward at 102 mm/sec. Next, the substrate was then dried with an airknife at a speed of 10 mm/sec. The film was stripped off of the glass prior to subsequent processing.
EXAMPLES 1-9:
Sheets of film made in PE-1 were cut to a size of 12 inch x 12 inch (30 cm x 30 cm) and then sequentially stained and crosslinked/end-capped using the “METHOD FOR APPLYING STAINING SOLUTION AND CROSSLINKER/END-CAPPER SOLUTIONS”. The corresponding crosslinkers/end-cappers are listed in Table 3.
COMPARATIVE EXAMPLES 1-4: BORATE/BORIC ACID CROSSLINKING
Sheets of fdm made in PE-1 were cut to a size of 12 inch x 12 inch (30 cm x 30 cm) and then stained using the “METHOD FOR APPLYING STAINING SOLUTION AND CROSSLINKER/END-CAPPER SOLUTIONS”. Crosslinker solutions were prepared as described in PE-3. For CE-1 through CE-4, the stained fdms were exposed to crosslinker solution at conditions shown in Table 2. For the immersion method, the crosslinker solution was poured into a glass crystallization dish. A section of fdm was manually immersed in the solution with tweezers for the 42 sec, rinsed with copious amounts of DI water, and dried with an air gun. For samples crosslinked at elevated temperature, following drying with an air gun, they were dried in an oven at 70 C for 4 min. Crosslinking via the spray method was done as described in the “METHOD FOR APPLYING STAINING SOLUTION AND CROSSLINKER/END-CAPPER SOLUTIONS”.
COMPARATIVE EXAMPLE 5: NO CROSSLINKING
Sheets of fdm made in PE-1 were cut to a size of 12 inch x 12 inch (30 cm x 30 cm) and then stained using the “METHOD FOR APPLYING STAINING SOLUTION AND CROSSLINKER/END-CAPPER SOLUTIONS”. No crosslinking was carried out.
TABLE 3: Visible light transmission (%T), change in visible light transmission after 500 hours environmental aging (A%T-500 h)), and edge ingress (%) for EX-1 to EX-9 and CE- 1 to CE-5. Crosslinking/end-capping were carried out at room temperature (RT) unless otherwise specified.
The Comparative Examples all have one or more of lower polarization efficiency, greater A%T-500 h, and greater edge ingress compared to the Examples.
Claims
1. A multi-layer film article comprising: at least one film; and an absorptive polarizer film disposed on the at least one film, the absorptive polarizer film comprising: an oriented polyvinyl alcohol film containing a dichroic material, wherein the oriented polyvinyl alcohol film has been crosslinked, end capped or both with a polyphenol compound, a nano-platelet material, a hydrogen bonding polymer, a non-phenolic polyacid, or a combination thereof.
2. The multi-layer film article of claim 1, wherein the dichroic material comprises iodine/iodide.
3. The multi-layer film article of claim 1, wherein the at least one film comprises a multilayer reflective polarizer film.
4. The multi-layer film article of claim 1, wherein the oriented polyvinyl alcohol film has been crosslinked or end-capped with a polyphenol compound, wherein the polyphenol compound comprises a flavonoid, a tannin, a lignan, a phenolic acid, or a combination thereof.
5. The multi-layer film article of claim 1, wherein the oriented polyvinyl alcohol film has been crosslinked or end-capped with a nano-platelet material comprising a clay platelet material.
6. The multi-layer film article of claim 5, wherein the clay platelet material comprises smectite, vermiculite, kaolin, sepiolite, glauconite, attapulgite, palygorskite, illite, chlorite, serpentine, layered double hydroxide, or a combination thereof.
7. The multi-layer film article of claim 1, wherein the oriented polyvinyl alcohol film has been crosslinked or end-capped with a hydrogen-bonding polymer comprising polyacrylic acid, polymethacrylic acid, or a combination thereof.
8. The multi-layer film article of claim 1, wherein the oriented polyvinyl alcohol film has been crosslinked or end-capped with a non-phenolic polyacid comprising citric acid, succinic acid, tartaric acid, or a combination thereof.
9. A modified film comprising: an oriented polyvinyl alcohol film containing a dichroic material, wherein the oriented polyvinyl alcohol film has been crosslinked, end capped or both with a polyphenol compound, a nano-platelet material, a hydrogen bonding polymer, a non-phenolic polyacid, or a combination thereof.
10. The modified film of claim 9, wherein the modified film comprises an absorptive polarizer film.
11. The modified film of claim 9, wherein the dichroic material comprises iodine/iodide.
12. The modified film of claim 9, wherein the oriented polyvinyl alcohol film has been crosslinked or end-capped with a polyphenol compound, wherein the polyphenol compound comprises a flavonoid, a tannin, a lignan, a phenolic acid, or a combination thereof.
13. The modified film of claim 9, wherein the oriented polyvinyl alcohol film has been crosslinked or end-capped with a nano-platelet material comprises a clay platelet material.
14. The modified film of claim 13, wherein the clay platelet material comprises smectite, vermiculite, kaolin, sepiolite, glauconite, attapulgite, palygorskite, illite, chlorite, serpentine, layered double hydroxide, or a combination thereof.
15. The modified film of claim 9, wherein the oriented polyvinyl alcohol film has been crosslinked or end-capped with a hydrogen-bonding polymer comprising polyacrylic acid, polymethacrylic acid, or a combination thereof.
16. The modified film of claim 9, wherein the oriented polyvinyl alcohol has been crosslinked or end-capped with a non-phenolic polyacid comprising citric acid, succinic acid, tartaric acid, or a combination thereof.
17. A method of forming a multi-layer film article comprising; providing at least one film; providing a film-forming solution, wherein the film -forming solution comprises: at least one solvent; and polyvinyl alcohol; disposing the film-forming solution on the at least one film; drying the film-forming solution to form a composite construction comprising a polyvinyl alcohol film disposed on the at least one film; orienting the composite construction; disposing on the polyvinyl alcohol film of the composite construction a staining composition comprising: at least one dichroic material; and at least one solvent; drying the composite construction with staining composition disposed on it; disposing a crosslinker/end-capper composition to the polyvinyl alcohol film of the composite construction, the crosslinker/end-capper composition comprises: a crosslinker, an end-capper or both, wherein the crosslinker or end-capper comprises a polyphenol compound, a nano-platelet material, a hydrogen bonding polymer, a non-phenolic polyacid, or a combination thereof; and at least one solvent; drying the composite construction with crosslinker/end-capper composition disposed on it.
18. The method of claim 17, wherein dichroic material comprises iodine/iodide.
19. The method of claim 17, further comprising: rinsing the composite construction after disposing the staining composition on the polyvinyl alcohol film of the composite construction.
20. The method of claim 17, further comprising: rinsing the composite construction after disposing the crosslinker/end-capper composition on the polyvinyl alcohol film of the composite construction.
21. The method of claim 17, further comprising: rinsing the composite construction after disposing the staining composition on the polyvinyl alcohol film of the composite construction; and rinsing the composite construction after disposing the crosslinker/end-capper composition on the polyvinyl alcohol film of the composite construction.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US202363535617P | 2023-08-31 | 2023-08-31 | |
| US63/535,617 | 2023-08-31 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2025046387A1 true WO2025046387A1 (en) | 2025-03-06 |
Family
ID=92627428
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/IB2024/058059 Pending WO2025046387A1 (en) | 2023-08-31 | 2024-08-19 | Dichroic polarizer film |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2025046387A1 (en) |
Citations (18)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE2835432C2 (en) * | 1978-08-12 | 1987-10-15 | Licentia Patent-Verwaltungs-Gmbh, 6000 Frankfurt, De | |
| US5882774A (en) | 1993-12-21 | 1999-03-16 | Minnesota Mining And Manufacturing Company | Optical film |
| US6096375A (en) | 1993-12-21 | 2000-08-01 | 3M Innovative Properties Company | Optical polarizer |
| US6111697A (en) | 1998-01-13 | 2000-08-29 | 3M Innovative Properties Company | Optical device with a dichroic polarizer and a multilayer optical film |
| US6352761B1 (en) | 1998-01-13 | 2002-03-05 | 3M Innovative Properties Company | Modified copolyesters and improved multilayer reflective films |
| US6697195B2 (en) | 2000-08-21 | 2004-02-24 | 3M Innovative Properties Company | Loss enhanced reflective optical filters |
| US6916440B2 (en) | 2001-05-31 | 2005-07-12 | 3M Innovative Properties Company | Processes and apparatus for making transversely drawn films with substantially uniaxial character |
| US20080088060A1 (en) * | 1999-11-22 | 2008-04-17 | Fujifilm Corporation | Sheet polarizer, optical film, liquid crystal display, and method of producing sheet polarizers |
| US7826009B2 (en) | 2006-12-21 | 2010-11-02 | 3M Innovative Properties Company | Hybrid polarizer |
| US20110102891A1 (en) | 2008-03-31 | 2011-05-05 | Derks Kristopher J | Low layer count reflective polarizer with optimized gain |
| US8232350B2 (en) | 2008-06-02 | 2012-07-31 | 3M Innovative Properties Company | Adhesive encapsulating composition and electronic devices made therewith |
| US8234998B2 (en) | 2006-09-08 | 2012-08-07 | Massachusetts Institute Of Technology | Automated layer by layer spray technology |
| US20120327512A1 (en) * | 2009-03-05 | 2012-12-27 | Nitto Denko Corporation | Thin high-performance polarizing film and method for manufacturing the same |
| TW201400540A (en) * | 2012-06-22 | 2014-01-01 | Univ Nat Kaohsiung Applied Sci | Composite solid state electrolyte membrane with crosslinked polyvinyl alcohol |
| US20170003426A1 (en) * | 2013-11-29 | 2017-01-05 | Sumitomo Chemical Company, Limited | Polarizer and polarizing plate including same |
| CN109244324A (en) * | 2018-09-19 | 2019-01-18 | 东华大学 | The bacteria cellulose composite diaphragm of heat cross-linking poly propenoic acid vinyl alcohol filling |
| KR20210058440A (en) * | 2019-11-14 | 2021-05-24 | (주)두와이즈켐 | Sustained antibacterial curable coating composition |
| CN114558558A (en) * | 2022-04-19 | 2022-05-31 | 成都理工大学 | Attapulgite-doped double-crosslinked hydrogel and preparation method and application thereof |
-
2024
- 2024-08-19 WO PCT/IB2024/058059 patent/WO2025046387A1/en active Pending
Patent Citations (18)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE2835432C2 (en) * | 1978-08-12 | 1987-10-15 | Licentia Patent-Verwaltungs-Gmbh, 6000 Frankfurt, De | |
| US5882774A (en) | 1993-12-21 | 1999-03-16 | Minnesota Mining And Manufacturing Company | Optical film |
| US6096375A (en) | 1993-12-21 | 2000-08-01 | 3M Innovative Properties Company | Optical polarizer |
| US6111697A (en) | 1998-01-13 | 2000-08-29 | 3M Innovative Properties Company | Optical device with a dichroic polarizer and a multilayer optical film |
| US6352761B1 (en) | 1998-01-13 | 2002-03-05 | 3M Innovative Properties Company | Modified copolyesters and improved multilayer reflective films |
| US20080088060A1 (en) * | 1999-11-22 | 2008-04-17 | Fujifilm Corporation | Sheet polarizer, optical film, liquid crystal display, and method of producing sheet polarizers |
| US6697195B2 (en) | 2000-08-21 | 2004-02-24 | 3M Innovative Properties Company | Loss enhanced reflective optical filters |
| US6916440B2 (en) | 2001-05-31 | 2005-07-12 | 3M Innovative Properties Company | Processes and apparatus for making transversely drawn films with substantially uniaxial character |
| US8234998B2 (en) | 2006-09-08 | 2012-08-07 | Massachusetts Institute Of Technology | Automated layer by layer spray technology |
| US7826009B2 (en) | 2006-12-21 | 2010-11-02 | 3M Innovative Properties Company | Hybrid polarizer |
| US20110102891A1 (en) | 2008-03-31 | 2011-05-05 | Derks Kristopher J | Low layer count reflective polarizer with optimized gain |
| US8232350B2 (en) | 2008-06-02 | 2012-07-31 | 3M Innovative Properties Company | Adhesive encapsulating composition and electronic devices made therewith |
| US20120327512A1 (en) * | 2009-03-05 | 2012-12-27 | Nitto Denko Corporation | Thin high-performance polarizing film and method for manufacturing the same |
| TW201400540A (en) * | 2012-06-22 | 2014-01-01 | Univ Nat Kaohsiung Applied Sci | Composite solid state electrolyte membrane with crosslinked polyvinyl alcohol |
| US20170003426A1 (en) * | 2013-11-29 | 2017-01-05 | Sumitomo Chemical Company, Limited | Polarizer and polarizing plate including same |
| CN109244324A (en) * | 2018-09-19 | 2019-01-18 | 东华大学 | The bacteria cellulose composite diaphragm of heat cross-linking poly propenoic acid vinyl alcohol filling |
| KR20210058440A (en) * | 2019-11-14 | 2021-05-24 | (주)두와이즈켐 | Sustained antibacterial curable coating composition |
| CN114558558A (en) * | 2022-04-19 | 2022-05-31 | 成都理工大学 | Attapulgite-doped double-crosslinked hydrogel and preparation method and application thereof |
Non-Patent Citations (4)
| Title |
|---|
| E. H. LAND, COLLOID CHEMISTRY, 1946 |
| KIRK OTHMER: "Encyclopedia of Chemical Technology", vol. 8, 1993, pages: 652 - 661 |
| KROGMAN ET AL., AUTOMATED PROCESS FOR IMPROVED UNIFORMITY AND VERSATILITY OF LAYER-BY-LAYER DEPOSITION, LANGMUIR, vol. 23, 2007, pages 3137 - 3141 |
| MA CHAO ET AL: "Oyster-inspired organic-inorganic hybrid system to improve cold-pressing adhesion, flame retardancy, and mildew resistance of soybean meal adhesive", COMPOSITES PART B, ELSEVIER, AMSTERDAM, NL, vol. 242, 9 June 2022 (2022-06-09), XP087139474, ISSN: 1359-8368, [retrieved on 20220609], DOI: 10.1016/J.COMPOSITESB.2022.110049 * |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US8557384B2 (en) | Silicate-containing antifog coatings | |
| KR20010034068A (en) | Optical Device with a Dichroic Polarizer and a Multilayer Optical Film | |
| JP6110596B2 (en) | Dye-iodine polarizing element film, polarizing plate, and liquid crystal display device | |
| CN106249335B (en) | Polarizing film and polarizing plate containing the same | |
| KR20230151987A (en) | Circular polarizer and image display device using the same | |
| KR20220104181A (en) | laminated film | |
| KR20010095231A (en) | Release film | |
| TW201940906A (en) | Electroluminescent display device | |
| KR20210002457A (en) | Polarizing plate and organic EL display device with retardation layer | |
| CN111480100B (en) | Laminate for polarizing plate, laminate film roll, and method for producing same | |
| JP6024271B2 (en) | Acrylic film for protecting polarizer, polarizing plate, and liquid crystal display device | |
| CN109254342A (en) | Polarizer | |
| WO2025046387A1 (en) | Dichroic polarizer film | |
| TWI857106B (en) | Easy-adhesion polyester film | |
| EP1360225A1 (en) | Tie layers for polyvinylalcohol coatings | |
| KR20160094364A (en) | Polarization element exhibiting uniform transmittance at respective wavelengths, and polarization plate | |
| TW202125005A (en) | Polarizing plate and manufacturing method thereof | |
| CN101278216B (en) | Optical functional film, retardation film, composition for forming optical functional layer, and method for producing optical functional film | |
| KR20090060467A (en) | Biaxially Oriented Polyester Laminated Film for Optics | |
| CN119907742A (en) | Highly adhesive polyester film | |
| CN113272114B (en) | Laminated body, polarizing plate, manufacturing method of laminated body, polarizing plate manufacturing method, and display device manufacturing method | |
| WO2022124104A1 (en) | Optical film with adhesive layer, and image display device including said optical film with adhesive layer | |
| KR20160092985A (en) | High-contrast polarization element exhibiting uniform transmittance at respective wavelengths, and polarization plate | |
| CN111480101B (en) | Laminate for polarizing plate, display device, and method for producing polarizing plate | |
| TWI912245B (en) | Optical laminate and image display device |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 24764909 Country of ref document: EP Kind code of ref document: A1 |