WO2025042711A1 - Engineered transferrin receptor binding peptides as well as methods of making and using the same - Google Patents
Engineered transferrin receptor binding peptides as well as methods of making and using the same Download PDFInfo
- Publication number
- WO2025042711A1 WO2025042711A1 PCT/US2024/042617 US2024042617W WO2025042711A1 WO 2025042711 A1 WO2025042711 A1 WO 2025042711A1 US 2024042617 W US2024042617 W US 2024042617W WO 2025042711 A1 WO2025042711 A1 WO 2025042711A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- peptide
- amino acid
- injection
- seq
- engineered
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K7/00—Peptides having 5 to 20 amino acids in a fully defined sequence; Derivatives thereof
- C07K7/04—Linear peptides containing only normal peptide links
- C07K7/08—Linear peptides containing only normal peptide links having 12 to 20 amino acids
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K38/00—Medicinal preparations containing peptides
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2319/00—Fusion polypeptide
- C07K2319/01—Fusion polypeptide containing a localisation/targetting motif
- C07K2319/10—Fusion polypeptide containing a localisation/targetting motif containing a tag for extracellular membrane crossing, e.g. TAT or VP22
Definitions
- the disclosure relates generally to biology and protein engineering and optimizing, and more particularly it relates to engineered transferrin receptor (TfR; or CD71) binding peptides that can be used to deliver a therapeutic agent to a cell, tissue, organ or system of an individual, as well as relates to methods of using the same for treating central nervous system (CNS) diseases, disorders or conditions or for treating TfR-associated diseases, disorders or conditions.
- CNS central nervous system
- BACKGROUND [003]
- Successful, active transport of therapeutic agents and/or carriers of such therapeutic agents to intracellular targets requires entry and/or passage through a cell membrane. Despite its selective permeability to compounds and molecules essential to cell function and survival, the cell membrane is a particularly daunting barrier to entry of therapeutic agents.
- a therapeutic agent can be conjugated to a peptide (e.g., RGD peptides or TAT peptides) or to an antibody ⁇ or a fragment thereof (e.g., antibody-drug (ADC)) that binds to a cell surface receptor, which is internalized and enters a cell's cytoplasm.
- ADC antibody-drug
- receptor-mediated internalization offers intracellular activity with the added benefit of specificity.
- TfR One target of interest for exploiting receptor-mediated internalization is the TfR. See, e.g., McQuiad et al. (2021) PLoS ONE 16:e0252341; and Intl. Patent Application Publication Nos.
- TfR binding agents are needed, however, especially TfR binding peptides that have rapid internalization kinetics, that have improved endosomal escape rates, and that can prevent receptor degradation by inducing recycling.
- the disclosure first describes an engineered peptide that can bind to the TfR (i.e., a TfR binding agent) and that can be used to deliver a therapeutic agent into (i.e., endocytosis) or across/through (i.e., transcytosis) a cell, tissue, organ or system of an individual, where the engineered peptide includes an amino acid sequence having at least about 95% sequence identity to any one of SEQ ID NOS:3 to 16, 18, 29 to 35 and 44 to 46.
- the engineered peptide is SEQ ID NO:3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 18, 29, 30, 31, 32, 33, 34, 35, 44, 45 or 46.
- the engineered peptide can be a monomer.
- the engineered peptide can be in the form of a multimer (e.g., a dimer, trimer, tetramer, etc.).
- the multimer can include the same monomer (e.g., a homodimer, homotrimer, homotetramer, etc.) or can include distinct monomers (e.g., a heterodimer, heterotrimer, heterotetramer, etc.).
- the disclosure describes an engineered peptide that can bind to the TfR and that can be used to deliver a therapeutic agent into or across/through (i.e., transcytosis) a cell, tissue, organ or system of an individual, where the engineered peptide includes an amino acid sequence of: X 1 X 2 X 3 CRX 6 FYPX 10 X 11 YCX 14 AX 16 (Formula I; SEQ ID NO:17), wherein X 1 , X 2 , X 3 , X 6 , X 10 , X 11 , X 14 and X 16 can be any naturally occurring amino acid or can be a variant thereof.
- X 1 , X 2 X 10 each and independently can be an aromatic amino acid.
- X 3 , X 11 and X 14 each and independently can be an acidic amino acid.
- X 1 can be F, I, L, M, W or Y.
- X 2 can be F, I, L, M, W or Y.
- X 3 can be D, E, H, K, N, R, Q or S.
- ⁇ X 6 can be S or Aib.
- X 10 be f, F, I, L, M, W or Y.
- X 11 can be D, E, H, K, N, R, Q or S.
- X 14 can be D, E, H, K, N, R, Q or S.
- X 16 is M, Nle or Hsm.
- X 1 is F
- X 2 is W
- X 3 is D
- X 6 is S
- X 10 is F
- X 11 is D
- X 1 is F
- X 2 is W
- X 3 is D
- X 6 is Aib
- X 10 is f
- X 11 is D
- X 1 is F
- X 2 is W
- X 3 is D
- X 6 is Aib
- X 10 is f
- X 11 is D
- X 1 is F
- X 2 is W
- X 3 is D
- X 6 is Aib
- X 10 is f
- X 11 is D
- the disclosure describes conjugates and fusions including an engineered peptide herein and a therapeutic agent linked thereto.
- the therapeutic agent can be an oligonucleotide, another peptide or protein, or a small molecule.
- the therapeutic agent is indirectly linked to the engineered peptide via, for example, a linker (i.e., a conjugate).
- the therapeutic agent is directly linked to the engineered peptide (i.e., a fusion).
- the therapeutic agent can be conjugated or fused to an amino (N)- terminal end of the engineered peptide.
- the therapeutic agent can be conjugated or fused to a carboxy (C)-terminal end of the engineered peptide.
- the engineered peptide can be conjugated or fused at least to an N-terminal end of the therapeutic agent. In other instances, the engineered peptide can be conjugated or fused at least to a C- terminal end of the therapeutic agent. In yet other instances, the engineered peptide can be conjugated or fused at least to one or more internal amino acids of the therapeutic agent. [0015] Third, the disclosure describes delivery vehicles including an engineered peptide herein, a conjugate herein or a fusion herein.
- the delivery vehicle can be a nanomedicine such as, for example, an enveloped nanocomplex (eNCP), a lipid nanoparticle ⁇ (LNP), a liposome, a nanocapsule (NC), nanocrystal (NCR), a nanoemulsion (NE), a proteolipid vehicle (PLV), a RNA nanoparticle (RNA NP) or a vector such as a recombinant adeno-associated virus (rAAV).
- eNCP enveloped nanocomplex
- LNP lipid nanoparticle ⁇
- NCR nanocrystal
- NE nanoemulsion
- PLV proteolipid vehicle
- RNA NP RNA nanoparticle
- rAAV recombinant adeno-associated virus
- the pharmaceutical compositions further can include other carriers, diluents and/or excipients. In other instances, the pharmaceutical compositions can include one or more additional therapeutic agents.
- the disclosure describes methods of transporting a therapeutic agent across a blood-brain barrier (BBB) of an individual, where the methods can include at least a step of administering to the individual an effective amount of a conjugate herein, a fusion herein, a delivery vehicle herein and/or a pharmaceutical composition herein.
- BBB blood-brain barrier
- the disclosure describes methods of treating an individual having a CNS disease, disorder or condition, where the methods can include at least a step of administering to the individual an effective amount of a conjugate herein, a fusion herein, a delivery vehicle herein and/or a pharmaceutical composition herein.
- the administering can be via direct injection into a tissue or an organ such as the CNS of the individual, which can be an ICV injection, an ICM injection, an IPM injection, or a combination thereof.
- the administering can be via a peripheral injection.
- the peripheral injection can be via IV injection or SC injection.
- the administering can be by a combination of direct injection and peripheral injection.
- the disclosure describes methods of treating an individual having a TfR- associated disease, disorder or condition, where the methods can include at least a step of administering to the individual an effective amount of a conjugate herein, a fusion herein, a delivery vehicle herein and/or a pharmaceutical composition herein.
- the ⁇ administering can be via direct injection a tissue or an organ such as the CNS of the individual, which can be an ICV injection, an ICM injection, an IPM injection, an IT injection or a combination thereof.
- the administering can be via a peripheral injection.
- the peripheral injection can be via IV injection or SC injection.
- the administering can be by a combination of direct injection and peripheral injection.
- the disclosure describes use of a conjugate herein, a fusion herein, a delivery vehicle herein or a pharmaceutical composition herein in the manufacture of a medicament for treatment of CNS disease, disorder or condition or for treatment of a TfR-associated disease, disorder or condition.
- the disclosure describes a conjugate herein, a fusion herein, a delivery vehicle herein or a pharmaceutical composition herein for use in the treatment of a CNS disease, disorder or condition or for use in the treatment of a TfR-associated disease, disorder or condition.
- An advantage of the engineered peptides herein is that, due to their small size, they can cross a physiological barrier such as the BBB and thus can be used to deliver therapeutic agents to the CNS when treating CNS diseases, disorders and/or conditions.
- An advantage of the engineered peptides herein is that they bind not only human TfR but also non-human TfR and therefore can be used for pharmacodynamic, pharmacokinetic and toxicology studies in animal models, especially non-transgenic animal models, that more readily translate from these species to humans. Likewise, and due to their cross-reactivity, the engineered peptides herein can be used not only for treating humans but also for treating animals.
- An advantage of the engineered peptides herein is that they are small (i.e., 20 residues or less) and thus can be efficiently transported into and/or across a cell. Likewise, and due to their size, the engineered peptides herein can be chemically synthesized in high quantities and at high purity with lower cost. [0025] An advantage of the engineered peptides herein is that they provide high yield and purity of conjugates and fusions, lower administered dose as compared to large molecules, higher tissue penetration and overall simplicity. BRIEF DESCRIPTION OF THE DRAWINGS ⁇ [0026]
- FIGS. 1A and 1B show binding and internalization of an exemplary TfR binding peptide displayed on phage (i.e., Peptide 9) using a hCMEC/D3 cell line, where FIG.1A shows binding at 4oC, and where FIG.1B shows internalizing at 37oC.
- FIGS. 1A and 1B show binding and internalization of an exemplary TfR binding peptide displayed on phage (i.e., Peptide 9) using a hCMEC/D3 cell line, where FIG.1A shows binding at 4oC, and where FIG.1B shows internalizing at 37oC.
- FIGS. 1A and 1B show binding and internalization of an exemplary TfR binding peptide displayed on phage (i.e., Peptide 9) using a hCMEC/D3 cell line, where FIG.1A shows binding at 4oC, and where FIG.1B shows internalizing at 37oC.
- FIGS. 1A and 1B show binding and
- FIG. 2A and 2B show binding of exemplary biotin-labeled, pure TfR binding peptides (i.e., Peptide 8 and Peptide 15) to EFO-21 (human TfR (hTfR)) or MDCK-c6 (cynomolgus TfR (cTfR)) cells as determined by flow cytometry, where FIG. 2A shows binding to EFO-21 cells, and where FIG.1B shows binding to MDCK-c6 cells.
- FIG. 3 shows internalizing of exemplary pure TfR binding peptides (i.e., Peptide 15 and Peptide 19) in EFO-21 cells.
- FIGS.4A and 4B show that an exemplary TfR binding peptide (i.e., Peptide 15) binds to hTfR at a distinct epitope from the transferrin (Tf) binding site, where FIG.4A is a heat map comparing deuterium (D 2 O) uptake between His-hTfR complexed with and without the exemplary TfR binding peptide, and where FIG.4B shows a putative epitope mapped on hTfR.
- FIGS.4A is a heat map comparing deuterium (D 2 O) uptake between His-hTfR complexed with and without the exemplary TfR binding peptide
- FIG.4B shows a putative epitope mapped on hTfR.
- FIG. 5A and 5B show cellular activity of exemplary TfR binding peptide (i.e., Peptide 15 and Peptide 17)-siRNA conjugates in a TfR-expressing cell line (EFO-21), where FIG.5A shows conjugates targeting the SNCA gene in EFO-21 cells, and where FIG.5B shows conjugates targeting the HPRT gene in EFO-21 cells.
- FIG.5A shows conjugates targeting the SNCA gene in EFO-21 cells
- FIG.5B shows conjugates targeting the HPRT gene in EFO-21 cells.
- FIGS. 7A and 7B show in vivo imaging of biodistribution of an exemplary TfR binding peptide (i.e., Peptide 15)-antibody fusion throughout the CNS, where FIG.
- FIG.8 shows in vivo activity of exemplary TfR binding peptides (i.e., Peptide 15 and Peptide 19)-siRNA conjugates targeting the HPRT gene in a mouse model.
- TfR is a transmembrane glycoprotein for Tf that functions to import iron by internalizing the Tf-iron complex through receptor-mediated endocytosis.
- TfR1 and TfR2 are two TfR isoforms, TfR1 and TfR2, where TfR1 is a high-affinity, ubiquitously expressed receptor but where TfR2 expression is restricted to certain cell types and is unaffected by intracellular iron concentrations.
- TfR1 is expressed in proliferative cells such as, for example, the basal epidermis, intestinal epithelium and certain activated immune cells. TfR1 also is expressed in non-proliferative cells such as, for example, orthochromatic normoblasts, reticulocytes, hepatocytes, placental trophoblasts, endothelial cells of the BBB and Sertoli cells of the blood- testis barrier (BTB). [0038] Of particular interest herein is TfR1. Exemplary nucleotide sequences for TfR1 can be found in NCBI Ref. Seq. Nos.
- NM_003234.4 human
- NM_011638.4 mouse
- NM_001257303.1 non-human primate
- XM_003310190.3 non-human primate
- NM_022712.1 rat
- exemplary amino acid sequences for TfR1 monomers can be found in NCBI Ref. Seq. Nos. NP_003225.2 (human; SEQ ID NO:1), NP_035768.1 (mouse), NP_001244232.1 (non-human primate), XP_003310238.1 (non-human primate) and NP_073203.1 (rat).
- TfR1 sequences are readily available using publicly available databases such as, for example, GenBank and UniProt.
- Engineered TfR binding peptides, conjugates and fusions including the same, delivery vehicles incorporating the same and compositions thereof are of interest for delivering a ⁇ therapeutic agent to, for example, a cell, organ or system of an individual via the TfR, especially the CNS and especially via TfR1.
- engineered peptides, conjugates, fusions, delivery vehicles and pharmaceutical compositions are of interest for treating individuals having a CNS disease, disorder or condition or having a TfR-associated disease, disorder or condition.
- AAV refers to adeno-associated virus
- Ab refers to antibody
- AD refers to Alzheimer’s disease
- ADC refers to antibody-drug conjugate
- ADHD attention deficit hyperactivity disorder
- Aib refers to 2-aminoisobutyric acid
- ALL refers to acute lymphoblastic leukemia
- aRNA refers to activating RNA
- ASO refers to antisense oligonucleotide
- BBB refers to blood-brain barrier
- BBTB refers to blood-testis barrier
- CLL refers to chronic lymphocytic leukemia
- CNS refers to central nervous system
- cTfR refers to cynomolgus transferrin receptor
- D 2 O refers to deuterium
- Da refers to dalton(s);
- DBCO refer
- activating RNA means a nucleic acid that contains RNA and that mediates the targeted activation of a promoter or other non-coding transcript of a RNA transcript via a RNA-induced transcriptional activation (RITA) complex pathway.
- aRNAs ⁇ typically are ds. aRNA activates, or upregulates expression of a target nucleotide sequence in a cell.
- administer means providing a substance (e.g., an engineered peptide herein, a conjugate herein, a fusion herein, a delivery vehicle herein or a pharmaceutical composition herein) to an individual in a manner that is pharmacologically useful (e.g., to treat a disease, disorder or condition in the individual).
- amino acid means a molecule that, from a chemical standpoint, is characterized by the presence of one or more amine groups and one or more carboxylic acid groups and may contain other functional groups.
- amino acid with a functional group available for conjugation means any natural or unnatural amino acid with a functional group that may be conjugated to a fatty acid by way of, for example, a linker.
- functional groups include, but are not limited to, alkynyl, alkenyl, amino, azido, bromo, carboxyl, chloro, iodo and thiol groups.
- central nervous system disease, disorder or condition or “CNS disease, disorder or condition” means a disease, disorder or condition that affects the brain (e.g., frontal lobe, occipital lobe, parietal lobe and/or temporal lobe including regions such as, for example, the amygdala, basal ganglia, Broca’s area, cerebellum, corpus callosum, hypothalamus, medulla oblongata and thalamus), brain stem, glial cells (e.g., astrocytes, ependymal cells, microglia and oligodendrocytes), neurons and/or spinal cord.
- glial cells e.g., astrocytes, ependymal cells, microglia and oligodendrocytes
- CNS diseases, disorders or conditions include, but are not limited to, attention deficit hyperactivity disorder (ADHD), autism, Alzheimer’s disease (AD), cancer, epilepsy, frontotemporal dementia (FTD), Huntington’s disease (HD), infection/inflammation, migraine, multiple sclerosis (MS), Parkinson’s disease (PD), seizure and stroke.
- ADHD attention deficit hyperactivity disorder
- AD Alzheimer’s disease
- cancer cancer
- epilepsy frontotemporal dementia
- HD Huntington’s disease
- MS multiple sclerosis
- PD Parkinson’s disease
- seizure and stroke seizure and stroke.
- “comparison window” means a contiguous and specified segment of a nucleotide sequence or amino acid sequence, where the sequence in the comparison window may include additions and/or deletions (i.e., gaps) compared to a reference sequence (which ⁇ does not include the additions and/or deletions) for optimal alignment of the two sequences.
- the comparison window is at least 5 contiguous nucleotides/amino acids in length, and optionally can be 10, 20, 30, 40, 50, 60, 70, 80, 90 or 100 nucleotides/amino acids, or longer.
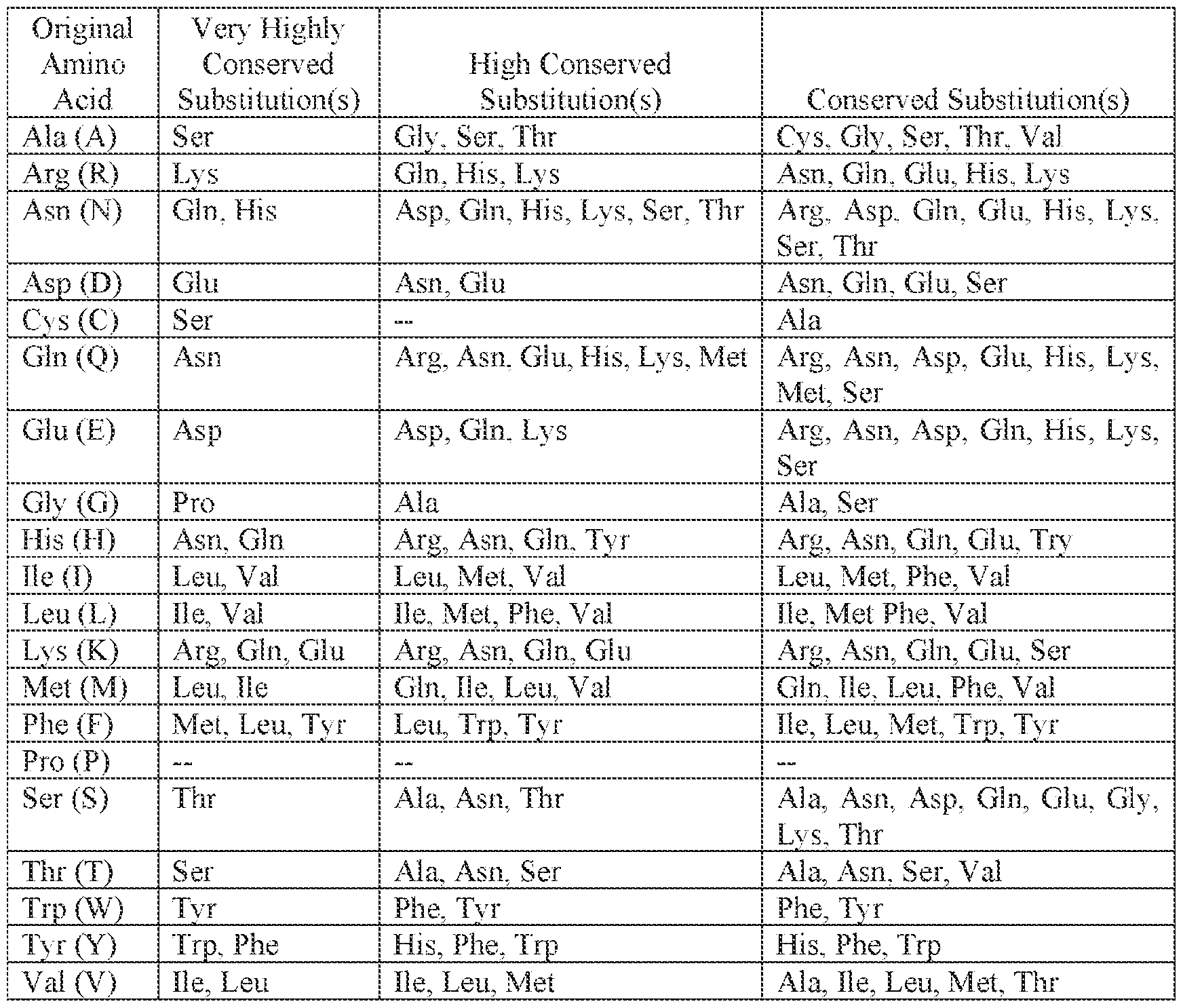
- “conservative substitution,” “conservative replacement” or “conservative mutation,” with regard to an amino acid sequence mean a change of one amino acid for another amino acid having a similar biochemical property (e.g., charge, hydrophobicity, size, etc.). Exemplary conservative substitutions are shown in Table 1. [0055] Table 1: Exemplary Conservative Amino Acid Substitution(s).
- a modified deoxyribonucleotide has one or more modifications or substitutions of atoms other ⁇ than hydroxyl at the 2' position, including or substitutions in or of the nucleobase, sugar, or phosphate group.
- an effective amount means an amount, concentration or dose of a therapeutic agent, or a pharmaceutical composition thereof, upon single or multiple dose administration to an individual in need thereof, provides a desired effect in such an individual under diagnosis or treatment (i.e., may produce a clinically measurable difference in a condition of the individual).
- An effective amount can be readily determined by one of skill in the art by using known techniques and by observing results obtained under analogous circumstances.
- RNA As used herein, “editing RNA” or “eRNA” means a nucleic acid that contains RNA and that mediates inserting, deleting and even base substituting of nucleotides within a target nucleotide sequence.
- RNA editing has been observed in a number of different types of RNA such as, for example, mRNA, microRNA (miRNA), transfer RNA (tRNA) and ribosomal RNA (rRNA).
- RNA editing is enzymatically mediated either by exogenously supplying adenosine deaminase acting on RNA enzyme (ADAR) or by directing an endogenous ADAR to a specific site in a target RNA nucleotide sequence, and typically involves editing at a single nucleotide site by directing the ADAR to that site through complimentary oligonucleotides.
- eRNAs typically are ss.
- engineered with respect to an amino acid sequence, means having an altered amino acid sequence obtained from genetic engineering techniques to nucleic acids that encode the protein and cells or organisms that express the protein.
- engineered means having an altered nucleotide sequence obtained from genetic engineering techniques. Genetic engineering techniques include, but are not limited to, PCR and DNA cloning technologies; transfection, transformation and other gene transfer technologies; homologous recombination; site-directed mutagenesis; and gene fusion. In this manner, a peptide or protein having an amino acid sequence identical to a naturally ⁇ occurring protein but produced by expression in a heterologous host, is not considered engineered.
- “engineered” means modified, especially with respect to a wild-type nucleotide sequence or a wild-type amino acid sequence.
- “engineered” may refer to a change, such as an addition, deletion and/or substitution of a nucleic acid residue or amino acid residue with respect to a given wild-type nucleotide or amino acid sequence.
- enveloped nanocomplex or “eNCP” means a NP that includes a lipid and therapeutic agent (e.g., a therapeutic oligonucleotide) complexed together by charge, which is further encapsulated by a coating. See, e.g., Niu et al. (2016) J. Contol.
- “individual” means any mammal, including cats, dogs, mice, rats and primates, especially humans. Moreover, “subject,” “participant” or “patient” may be used interchangeably with “individual.” [0062] As used herein, “individual in need thereof” means a mammal, such as a human, with a disease, disorder or condition requiring treatment or therapy including, for example, those listed herein. In particular, the preferred individual to be treated is a human.
- RNA means a nucleic acid that contains RNA and that mediates the targeted cleavage of a RNA transcript via RNA interference, for example, through a RNA-induced silencing complex (RISC) pathway.
- RISC RNA-induced silencing complex
- Some iRNAs are ss and other iRNAs are ds and have a sense strand and an antisense strand, where the sense strand and the antisense strand form a duplex.
- RISC RNA-induced silencing complex
- Some iRNAs are ss and other iRNAs are ds and have a sense strand and an antisense strand, where the sense strand and the antisense strand form a duplex.
- iRNA directs sequence-specific degradation of mRNA via RNA interference.
- iRNA attenuates, inhibits, modulates or reduces expression of a target nucleotide sequence in a cell.
- iRNA examples include, but are not limited to, an antisense oligonucleotide (ASO), Dicer substrate interfering RNA (DsiRNA), miRNA, short hairpin RNA (shRNA) and small interfering RNA (siRNA).
- ASO antisense oligonucleotide
- DsiRNA Dicer substrate interfering RNA
- miRNA miRNA
- shRNA short hairpin RNA
- shRNA small interfering RNA
- siRNA small interfering RNA
- linker means a structure used to conjugate one compound (e.g., an engineered peptide herein) to another compound (e.g., another engineered peptide or a therapeutic agent).
- a linker can be “labile” or “cleavable,” meaning the linker can be cleaved (e.g., by acidic pH or enzyme).
- lipid nanoparticle or “LNP” means a NP that includes a solid lipid core having a therapeutic agent (e.g., a therapeutic oligonucleotide) encapsulated within the ⁇ core, where the lipids can be cationic or lipids, as well as cholesterol and a helper lipid. See, e.g., Hou et al. (2021) Nat. Rev. Mater. 6:1078-1094; Mashima & Takada (2022) Curr. Issues Mol.
- a therapeutic agent e.g., a therapeutic oligonucleotide
- liposome means a NP that includes a closed lipid bilayer having a therapeutic agent (e.g., a therapeutic oligonucleotide) encapsulated therein. See, e.g., Hou et al. (2021), supra; Tenchov et al. (2021), supra; and Xue et al. (2015) Curr. Pharm. Des.21:3140- 3147.
- a therapeutic agent e.g., a therapeutic oligonucleotide
- nanomedicine means a material having a size from about 1 nm to about 100 nm for treating a disease, disorder or condition.
- examples of nanomedicines include, but are not limited to, liposomes, LNPs, eNCP, NCs, NCRs, NEs, PLVs, vectors (especially viral vectors), RNA NPs and recombinant viruses.
- nanocapsule or “NC” means a NP that includes a liquid lipid core and a therapeutic agent (e.g., a therapeutic oligonucleotide) on an outside membrane of the core, which is further encapsulated by a coating.
- nanoemulsion or “NE” means a NP that includes a liquid lipid core having a therapeutic agent (e.g., a therapeutic oligonucleotide) on an outside membrane of the core.
- a therapeutic agent e.g., a therapeutic oligonucleotide
- Chircov & Grumezescu “Nanoemulsion Preparation, Characterization, and Application in the Field of Biomedicine, Nanoarchitectonics in Biomedicine, 169-188 (Grumezescu ed., 2019).
- nanoparticle or “NP” means a natural or synthetic material or a combination thereof having lengths in two or three dimension and having a diameter between about 1 nm to about 100 nm that can be used to transport, for example, a therapeutic agent.
- NPs typically possess unique physical and chemical properties due to their high surface area and nanoscale size and typically include three layers: (1) a surface layer that may be functionalized, (2) a shell layer and (3) a core that is chemically distinct from the shell layer.
- NPs for use herein include, but not limited to, a dendrimer, a liposome, a LNP, an eNCP, a NC, a NCR, a NE, a PLV and a RNA NP.
- nucleic acid means a polymer of nucleotides. Although it may comprise any type of nucleotide units, the term generally applies to nucleotide polymers of DNA, cDNA and/or RNA.
- Polynucleotide is used to include single-stranded (ss) nucleic acids, double-stranded (ds) nucleic acids, and DNA, cDNA and/or RNA made from nucleotide or nucleoside analogues that may be identified by their sequences, which are generally presented in the 5' to 3' direction (as the coding strand), where the 5' and 3' indicate the linkages formed between the 5' hydroxyl group of one nucleotide and the 3'-hydroxyl group of the next nucleotide.
- its complement or non-coding strand
- nucleoside means a nucleobase-sugar combination, where the nucleobase portion is normally a heterocyclic base.
- the two most common classes of such heterocyclic bases are purines and pyrimidines.
- the sugar is normally a pentose sugar such as a ribose or a deoxyribose (e.g., 2'-deoxyribose).
- nucleotide means an organic molecule having a nucleoside (a nucleobase such as, for example, adenine, cytosine, guanine, thymine or uracil; and a pentose sugar such as, e.g., ribose or 2'-deoxyribose) and a phosphate group, which can serve as a monomeric unit of nucleic acid polymers such as deoxyribonucleic acid (DNA) and ribonucleic acid (RNA).
- a nucleoside such as, for example, adenine, cytosine, guanine, thymine or uracil
- pentose sugar such as, e.g., ribose or 2'-deoxyribose
- phosphate group a monomeric unit of nucleic acid polymers such as deoxyribonucleic acid (DNA) and ribonucleic acid (RNA).
- oligonucleotide means a short nucleic acid compound (e.g., a polymer of less than about 100 nucleotides in length) that may include deoxyribonucleotides (or modified deoxyribonucleotides), ribonucleotides (or modified ribonucleotides) or both.
- an oligonucleotide may be ss or ds and thus may or may not have duplex regions.
- “pharmaceutically acceptable,” when referring to a material such as a carrier or diluent, means that it does not abrogate the biological activity or properties of a therapeutic agent and is relatively non-toxic (i.e., the material may be administered to an individual without causing undesirable biological effects or interacting in a deleterious manner with any of the components of the composition in which it is contained.
- acceptable carrier means a pharmaceutically acceptable material, composition or carrier, such as a liquid or solid filler, stabilizer, dispersing agent, suspending agent, diluent, excipient, thickening agent, solvent or encapsulating material, involved in carrying or transporting a therapeutic agent within or to an individual such that it may perform its intended function. Additional ingredients that may be included in the pharmaceutical compositions used in the practice of the invention are known in the art and described, for example in Remington’s Pharmaceutical Sciences, 21 st Edition, University of the Sciences in Philadelphia, PA (2006).
- “pharmaceutical composition” means a composition or therapeutic agent, mixed with at least one pharmaceutically acceptable chemical component, such as, but not limited to carriers, stabilizers, diluents, dispersing agents, suspending agents, thickening agents, excipients and the like.
- “proteolipid vehicle” or “PLV” means a NP that includes a combination of proteins, such as fusion-associated small transmembrane (FAST) proteins, and neutral or ionizable lipids having a therapeutic agent (e.g., a therapeutic oligonucleotide) encapsulated therein. See, e.g., Brown et al.
- recombinant adeno-associated virus vector means an engineered polynucleotide vector comprising one or more heterologous sequences (i.e., nucleic acid sequence not of an AAV origin) that are flanked by at least one AAV ITR sequence.
- heterologous sequences i.e., nucleic acid sequence not of an AAV origin
- Such rAAV vectors can be replicated and packaged into infectious viral particles when present in a host cell that has been infected with a suitable helper virus (or that is expressing suitable helper functions) that expresses AAV rep and cap gene products (i.e., AAV Rep and Cap proteins).
- ribonucleotide means a nucleotide having a ribose as its pentose sugar, which contains a hydroxyl group at its 2' position.
- a modified ribonucleotide is a ⁇ ribonucleotide having one or more or substitutions of atoms other than hydrogen at the 2' position, including modifications or substitutions in or of the nucleobase, sugar, or phosphate group.
- RNA nanoparticle or “RNA NP” means a NP that includes a plurality of oligonucleotide oligomers and a therapeutic agent (e.g., a therapeutic oligonucleotide).
- sequence identity in the context of two nucleotide sequences or two amino acid sequences, means that residues in the two sequences that are the same when aligned for maximum correspondence over a specified comparison window.
- transferrin receptor-associated disease, disorder or condition or “TfR-associated disease, disorder or condition” means a disease, disorder or condition in which TfR is an etiology thereof, especially TfR1 upregulation and/or dysregulation.
- TfR-associated diseases, disorders and/or conditions include, but are not limited to, anemia, cancer (e.g., solid cancers such as adrenal cortical carcinoma, bladder cancer, breast cancer, cervical cancer, cholangiocarcinoma, colon cancer, esophageal squamous cell carcinoma, hepatocellular carcinoma, liver cancer, lung cancer, osteosarcoma, ovarian cancer, pancreatic cancer, renal cell carcinoma, and cancers of the nervous system, as well as hematopoietic malignancies such as acute lymphoblastic leukemia (ALL), chronic lymphocytic leukemia (CLL) and non-Hodgkin lymphoma (NHL) and neurodegenerative disease.
- ALL acute lymphoblastic leukemia
- CLL chronic lymphocytic leukemia
- NHL non-Hodgkin lymphoma
- therapeutic oligonucleotide means a ss or ds nucleic acid that has a therapeutic application (i.e., application in diagnosing and/or treating a disease, disorder or condition). Such a nucleic acid typically contains one or more modified nucleotide residues or linkages and also can include a targeting ligand and/or delivery vehicle. Examples of therapeutic oligonucleotides include, but are not limited to, aRNA, eRNA, iRNA, mRNA and tRNA.
- therapeutic oligonucleotides include, but are not limited to, ASOs, aptamers, mRNAs, miRNAs, short activating RNAs (saRNAs), siRNAs, suppressor tRNAs (sup-tRNAs) and decoys.
- ASOs aptamers
- mRNAs miRNAs
- miRNAs miRNAs
- siRNAs short activating RNAs
- suppressor tRNAs suppressor tRNAs
- decoys decoys.
- “treat,” “to treat,” “treatment” or “treating” means an act of providing care to an individual in need thereof, for example, by administering a therapeutic agent to the individual for purposes of improving the health and/or well-being of the individual with respect ⁇ to an existing a disease, disorder or or to prevent or decrease the likelihood of the occurrence of a disease, disorder or condition.
- Treating also can involve reducing the frequency or severity of at least one sign, symptom or contributing factor of a disease, disorder or condition experienced by the individual.
- vector means a nucleic acid molecule capable of transporting another nucleic acid sequence (or multiple nucleic acid sequences) to which it has been ligated into a host cell or genome.
- plasmid refers to a circular DNA loop, typically double-stranded (ds), into which additional DNA segments may be ligated.
- a viral vector wherein additional DNA segments may be ligated into the viral genome.
- Certain vectors are capable of autonomous replication in a host cell into which they are introduced (e.g., bacterial vectors having a bacterial origin of replication). Moreover, certain vectors are capable of directing the expression of genes (e.g., genes encoding an exogenous peptide or protein of interest) to which they are operatively linked when combined with appropriate control sequences such as promoter and operator sequences and replication initiation sites. Such vectors are commonly referred to as “expression vectors” and may also include a multiple cloning site for insertion of the gene encoding the protein of interest. Alternatively, the gene encoding the peptide or protein of interest may be introduced by site-directed mutagenesis techniques such as Kunkel mutagenesis.
- viral vector means a vector that is derived from a naturally occurring or modified virus, especially a rAAV vector or a Baculovirus vector (e.g., Autographa californica nuclear polyhedrosis (AcNPV) vector)).
- the engineered peptides herein can bind to the TfR and can have from about 14 amino acids to about 20 amino acids, about 15 to about 19 amino acids, about 16 to about 18 amino acids or about 17 amino acids. In other instances, the engineered peptides can have about 14 amino acids, about 15 amino acids, about 16 amino acids, about 17 amino acids, about 18 amino acids, about 19 amino acids or about 20 amino acids.
- the engineered peptides can be ⁇ used to deliver, for example, a to a cell, tissue, organ or system of an individual.
- L leucine
- variants thereof e.g., inverted Phe (f), homoserine methyl ether (Hsm) and norleucine (Nle)
- 2-aminoisobutyric acid e.g., 2-aminoisobutyric acid
- amino acid sequence can have at least about 95% sequence identity to SEQ ID NO:3.
- amino acid sequence is SEQ ID NO:3.
- Another such engineered peptide includes an amino acid sequence having at least about 80% sequence identity to SEQ ID NO:4.
- the amino acid sequence can have at least 80%, about 81%, about 82%, about 83%, about 84%, about 85%, about 86%, about 87%, about 88%, about 89%, about 90%, about 91%, about 92%, about 93%, about 94%, about 95%, about 96%, about 97%, about 98%, about 99% or about 100% sequence identity to SEQ ID NO:4.
- the amino acid sequence can have at least about 95% sequence identity to SEQ ID NO:4.
- the amino acid sequence is SEQ ID NO:4.
- Another engineered peptide includes an amino acid sequence having at least about 80% sequence identity to SEQ ID NO:5.
- the amino acid sequence can have at least about 80%, about 81%, about 82%, about 83%, about 84%, about 85%, about 86%, ⁇ about 87%, about 88%, about 89%, about about 91%, about 92%, about 93%, about 94%, about 95%, about 96%, about 97%, about 98%, about 99% or about 100% sequence identity to SEQ ID NO:5.
- the amino acid sequence can have at least about 95% sequence identity to SEQ ID NO:5.
- the amino acid sequence is SEQ ID NO:5.
- Another engineered peptide includes an amino acid sequence having at least about 80% sequence identity to SEQ ID NO:6.
- the amino acid sequence can have at least about 80%, about 81%, about 82%, about 83%, about 84%, about 85%, about 86%, about 87%, about 88%, about 89%, about 90%, about 91%, about 92%, about 93%, about 94%, about 95%, about 96%, about 97%, about 98%, about 99% or about 100% sequence identity to SEQ ID NO:6.
- the amino acid sequence can have at least about 95% sequence identity to SEQ ID NO:6.
- the amino acid sequence is SEQ ID NO:6.
- Another engineered peptide includes an amino acid sequence having at least about 80% sequence identity to SEQ ID NO:7.
- the amino acid sequence can have at least about 80%, about 81%, about 82%, about 83%, about 84%, about 85%, about 86%, about 87%, about 88%, about 89%, about 90%, about 91%, about 92%, about 93%, about 94%, about 95%, about 96%, about 97%, about 98%, about 99% or about 100% sequence identity to SEQ ID NO:7.
- the amino acid sequence can have at least about 95% sequence identity to SEQ ID NO:7.
- the amino acid sequence is SEQ ID NO:7.
- Another engineered peptide includes an amino acid sequence having at least about 80% sequence identity to SEQ ID NO:8.
- the amino acid sequence can have at least about 80%, about 81%, about 82%, about 83%, about 84%, about 85%, about 86%, about 87%, about 88%, about 89%, about 90%, about 91%, about 92%, about 93%, about 94%, about 95%, about 96%, about 97%, about 98%, about 99% or about 100% sequence identity to SEQ ID NO:8.
- the amino acid sequence can have at least about 95% sequence identity to SEQ ID NO:8.
- the amino acid sequence is SEQ ID NO:8.
- Another engineered peptide includes an amino acid sequence having at least about 80% sequence identity to SEQ ID NO:9.
- the amino acid sequence can have at least about 80%, about 81%, about 82%, about 83%, about 84%, about 85%, about 86%, ⁇ about 87%, about 88%, about 89%, about about 91%, about 92%, about 93%, about 94%, about 95%, about 96%, about 97%, about 98%, about 99% or about 100% sequence identity to SEQ ID NO:9.
- the amino acid sequence can have at least about 95% sequence identity to SEQ ID NO:9.
- the amino acid sequence is SEQ ID NO:9.
- Another engineered peptide includes an amino acid sequence having at least about 80% sequence identity to SEQ ID NO:10.
- the amino acid sequence can have at least about 80%, about 81%, about 82%, about 83%, about 84%, about 85%, about 86%, about 87%, about 88%, about 89%, about 90%, about 91%, about 92%, about 93%, about 94%, about 95%, about 96%, about 97%, about 98%, about 99% or about 100% sequence identity to SEQ ID NO:10.
- the amino acid sequence can have at least about 95% sequence identity to SEQ ID NO:10.
- the amino acid sequence is SEQ ID NO:10.
- Another engineered peptide includes an amino acid sequence having at least about 80% sequence identity to SEQ ID NO:11.
- the amino acid sequence can be at least about 80%, about 81%, about 82%, about 83%, about 84%, about 85%, about 86%, about 87%, about 88%, about 89%, about 90%, about 91%, about 92%, about 93%, about 94%, about 95%, about 96%, about 97%, about 98%, about 99% or about 100% sequence identity to SEQ ID NO:11.
- the amino acid sequence can have at least about 95% sequence identity to SEQ ID NO:11.
- the amino acid sequence is SEQ ID NO:11.
- Another engineered peptide includes an amino acid sequence having at least about 80% sequence identity to SEQ ID NO:12.
- the amino acid sequence can have at least about 80%, about 81%, about 82%, about 83%, about 84%, about 85%, about 86%, about 87%, about 88%, about 89%, about 90%, about 91%, about 92%, about 93%, about 94%, about 95%, about 96%, about 97%, about 98%, about 99% or about 100% sequence identity to SEQ ID NO:12.
- the amino acid sequence can have at least about 95% sequence identity to SEQ ID NO:12.
- the amino acid sequence is SEQ ID NO:12.
- Another engineered peptide includes an amino acid sequence having at least about 80% sequence identity to SEQ ID NO:13.
- the amino acid sequence can have at least about 80%, about 81%, about 82%, about 83%, about 84%, about 85%, about 86%, ⁇ about 87%, about 88%, about 89%, about about 91%, about 92%, about 93%, about 94%, about 95%, about 96%, about 97%, about 98%, about 99% or about 100% sequence identity to SEQ ID NO:13.
- the amino acid sequence can have at least about 95% sequence identity to SEQ ID NO:13.
- the amino acid sequence is SEQ ID NO:13.
- Another engineered peptide includes an amino acid sequence having at least about 80% sequence identity to SEQ ID NO:14.
- the amino acid sequence can have at least about 80%, about 81%, about 82%, about 83%, about 84%, about 85%, about 86%, about 87%, about 88%, about 89%, about 90%, about 91%, about 92%, about 93%, about 94%, about 95%, about 96%, about 97%, about 98%, about 99% or about 100% sequence identity to SEQ ID NO:14.
- the amino acid sequence can have at least about 95% sequence identity to SEQ ID NO:14.
- the amino acid sequence is SEQ ID NO:14.
- Another engineered peptide includes an amino acid sequence having at least about 80% sequence identity to SEQ ID NO:15.
- the amino acid sequence can have at least about 80%, about 81%, about 82%, about 83%, about 84%, about 85%, about 86%, about 87%, about 88%, about 89%, about 90%, about 91%, about 92%, about 93%, about 94%, about 95%, about 96%, about 97%, about 98%, about 99% or about 100% sequence identity to SEQ ID NO:15.
- the amino acid sequence can have at least about 95% sequence identity to SEQ ID NO:15.
- the amino acid sequence is SEQ ID NO:15.
- Another engineered peptide includes an amino acid sequence having at least about 80% sequence identity to SEQ ID NO:16.
- the amino acid sequence can have at least about 80%, about 81%, about 82%, about 83%, about 84%, about 85%, about 86%, about 87%, about 88%, about 89%, about 90%, about 91%, about 92%, about 93%, about 94%, about 95%, about 96%, about 97%, about 98%, about 99% or about 100% sequence identity to SEQ ID NO:16.
- the amino acid sequence can have at least about 95% sequence identity to SEQ ID NO:16.
- the amino acid sequence is SEQ ID NO:16.
- Another engineered peptide includes an amino acid sequence having at least about 80% sequence identity to SEQ ID NO:18.
- the amino acid sequence can have at least about 80%, about 81%, about 82%, about 83%, about 84%, about 85%, about 86%, ⁇ about 87%, about 88%, about 89%, about about 91%, about 92%, about 93%, about 94%, about 95%, about 96%, about 97%, about 98%, about 99% or about 100% sequence identity to SEQ ID NO:18.
- the amino acid sequence can have at least about 95% sequence identity to SEQ ID NO:18.
- the amino acid sequence is SEQ ID NO:18.
- Another engineered peptide includes an amino acid sequence having at least about 80% sequence identity to SEQ ID NO:29.
- the amino acid sequence can have at least about 80%, about 81%, about 82%, about 83%, about 84%, about 85%, about 86%, about 87%, about 88%, about 89%, about 90%, about 91%, about 92%, about 93%, about 94%, about 95%, about 96%, about 97%, about 98%, about 99% or about 100% sequence identity to SEQ ID NO:29.
- the amino acid sequence can have at least about 95% sequence identity to SEQ ID NO:29.
- the amino acid sequence is SEQ ID NO:29.
- Another engineered peptide includes an amino acid sequence having at least about 80% sequence identity to SEQ ID NO:30.
- the amino acid sequence can have at least about 80%, about 81%, about 82%, about 83%, about 84%, about 85%, about 86%, about 87%, about 88%, about 89%, about 90%, about 91%, about 92%, about 93%, about 94%, about 95%, about 96%, about 97%, about 98%, about 99% or about 100% sequence identity to SEQ ID NO:30.
- the amino acid sequence can have at least about 95% sequence identity to SEQ ID NO:30.
- the amino acid sequence is SEQ ID NO:30. [00112]
- Another engineered peptide includes an amino acid sequence having at least about 80% sequence identity to SEQ ID NO:31.
- the amino acid sequence can have at least about 80%, about 81%, about 82%, about 83%, about 84%, about 85%, about 86%, about 87%, about 88%, about 89%, about 90%, about 91%, about 92%, about 93%, about 94%, about 95%, about 96%, about 97%, about 98%, about 99% or about 100% sequence identity to SEQ ID NO:31.
- the amino acid sequence can have at least about 95% sequence identity to SEQ ID NO:31.
- the amino acid sequence is SEQ ID NO:31.
- Another engineered peptide includes an amino acid sequence having at least about 80% sequence identity to SEQ ID NO:32.
- the amino acid sequence can have at least about 80%, about 81%, about 82%, about 83%, about 84%, about 85%, about 86%, ⁇ about 87%, about 88%, about 89%, about about 91%, about 92%, about 93%, about 94%, about 95%, about 96%, about 97%, about 98%, about 99% or about 100% sequence identity to SEQ ID NO:32.
- the amino acid sequence can have at least about 95% sequence identity to SEQ ID NO:32.
- the amino acid sequence can be SEQ ID NO:32.
- Another engineered peptide includes an amino acid sequence having at least about 80% sequence identity to SEQ ID NO:33.
- the amino acid sequence can have at least about 80%, about 81%, about 82%, about 83%, about 84%, about 85%, about 86%, about 87%, about 88%, about 89%, about 90%, about 91%, about 92%, about 93%, about 94%, about 95%, about 96%, about 97%, about 98%, about 99% or about 100% sequence identity to SEQ ID NO:33.
- the amino acid sequence can have at least about 95% sequence identity to SEQ ID NO:33.
- the amino acid sequence can be SEQ ID NO:33.
- Another engineered peptide includes an amino acid sequence having at least about 80% sequence identity to SEQ ID NO:34.
- the amino acid sequence can have at least about 80%, about 81%, about 82%, about 83%, about 84%, about 85%, about 86%, about 87%, about 88%, about 89%, about 90%, about 91%, about 92%, about 93%, about 94%, about 95%, about 96%, about 97%, about 98%, about 99% or about 100% sequence identity to SEQ ID NO:34.
- the amino acid sequence can have at least about 95% sequence identity to SEQ ID NO:34.
- the amino acid sequence can be SEQ ID NO:34.
- Another engineered peptide includes an amino acid sequence having at least about 80% sequence identity to SEQ ID NO:35.
- the amino acid sequence can have at least about 80%, about 81%, about 82%, about 83%, about 84%, about 85%, about 86%, about 87%, about 88%, about 89%, about 90%, about 91%, about 92%, about 93%, about 94%, about 95%, about 96%, about 97%, about 98%, about 99% or about 100% sequence identity to SEQ ID NO:35.
- the amino acid sequence can have at least about 95% sequence identity to SEQ ID NO:35.
- the amino acid sequence can be SEQ ID NO:35.
- Another engineered peptide includes an amino acid sequence having at least about 80% sequence identity to SEQ ID NO:44.
- the amino acid sequence can have at least about 80%, about 81%, about 82%, about 83%, about 84%, about 85%, about 86%, ⁇ about 87%, about 88%, about 89%, about about 91%, about 92%, about 93%, about 94%, about 95%, about 96%, about 97%, about 98%, about 99% or about 100% sequence identity to SEQ ID NO:44.
- the amino acid sequence can have at least about 95% sequence identity to SEQ ID NO:44.
- the amino acid sequence can be SEQ ID NO:44.
- Another engineered peptide includes an amino acid sequence having at least about 80% sequence identity to SEQ ID NO:45.
- the amino acid sequence can have at least about 80%, about 81%, about 82%, about 83%, about 84%, about 85%, about 86%, about 87%, about 88%, about 89%, about 90%, about 91%, about 92%, about 93%, about 94%, about 95%, about 96%, about 97%, about 98%, about 99% or about 100% sequence identity to SEQ ID NO:45.
- the amino acid sequence can have at least about 95% sequence identity to SEQ ID NO:45.
- the amino acid sequence can be SEQ ID NO:45.
- Another engineered peptide includes an amino acid sequence having at least about 80% sequence identity to SEQ ID NO:46.
- the amino acid sequence can have at least about 80%, about 81%, about 82%, about 83%, about 84%, about 85%, about 86%, about 87%, about 88%, about 89%, about 90%, about 91%, about 92%, about 93%, about 94%, about 95%, about 96%, about 97%, about 98%, about 99% or about 100% sequence identity to SEQ ID NO:46. In other instances, the amino acid sequence can have at least about 95% sequence identity to SEQ ID NO:46. In certain instances, the amino acid sequence can be SEQ ID NO:46.
- the engineered peptide includes an amino acid sequence of: X 1 X 2 X 3 CRX 6 FYPX 10 X 11 YCX 14 AX 16 (Formula I; SEQ ID NO:17), wherein X 1 can be any naturally occurring amino acid or a variant thereof, especially an aromatic amino acid, wherein X 2 can be any naturally occurring amino acid or a variant thereof, especially an aromatic amino acid, wherein X 3 can be any naturally occurring amino acid or a variant thereof, especially an acidic amino acid, wherein X 6 can be any naturally occurring amino acid or a variant thereof, especially an alpha-methylated amino acid, wherein X 10 can be any naturally occurring amino acid or a variant thereof, especially an aromatic amino acid, wherein X 11 can be any naturally occurring amino acid or a variant thereof, especially an acidic amino acid, wherein X 14 can be any naturally occurring amino acid or a variant thereof, ⁇ especially an acidic amino acid, wherein can be any naturally occurring amino acid or a variant thereof, especially an
- X 1 can be F, I, L, M, W, Y or a conservative substitution thereof.
- X 2 can be F, I, L, M, W, Y or a conservative substitution thereof.
- X 3 can be D, E, S, Q or a conservative substitution thereof.
- X 6 can be S, Aib or a conservative substitution thereof.
- X 10 can be f, F, I, L, M, W, Y or a conservative substitution thereof.
- X 11 can be D, E, S, Q or a conservative substitution thereof.
- X 14 can be D, E, H, K, N, R, Q, S or a conservative substitution thereof.
- X 16 can be M, Nle, Hsm or a conservative substitution thereof.
- X 1 can be F, I, L, M, W, Y or a conservative substitution thereof;
- X2 can be F, I, L, M, W, Y or a conservative substitution thereof;
- X3 can be D, E, S, Q or a conservative substitution thereof;
- X 6 can be S, Aib or a conservative substitution thereof;
- X10 can be f, F, I, L, M, W, Y or a conservative substitution thereof;
- X11 can be D, E, S, Q or a conservative substitution thereof;
- X 14 can be D, E, H, K, N, R, Q , S or a conservative substitution thereof; and
- X 16 can be M, Nle, Hsm or a conservative substitution thereof.
- X 1 is F or a conservative substitution thereof.
- X 2 is W or a conservative substitution thereof.
- X 3 is D or a conservative substitution thereof.
- X 6 is S or a conservative substitution thereof.
- X 10 is F or a conservative substitution thereof.
- X 11 is D or a conservative substitution thereof.
- X 14 is E or a conservative substitution thereof.
- X 16 is M or a conservative substitution thereof.
- X 1 is F or a conservative substitution thereof
- X 2 is W or a conservative substitution thereof
- X 3 is D or a conservative substitution thereof
- X 6 is S or a conservative substitution thereof
- X 10 is F or a conservative substitution thereof
- X 11 is D or a conservative substitution thereof
- X 14 is E or a conservative substitution thereof
- X 16 is M or a conservative substitution thereof (SEQ ID NO:18).
- X 1 is F
- X 2 is W
- X 3 is D
- X 6 is S
- X 10 is F
- X 11 is D
- X 14 is E
- X 16 is M or a conservative substitution thereof (SEQ ID NO:18).
- X 1 is F or a conservative substitution thereof.
- X 2 is W or a conservative substitution thereof.
- X 3 is D or a conservative substitution thereof.
- X 6 is Aib.
- X 10 is f.
- X 11 is D or a conservative substitution thereof.
- X 14 is E or a conservative substitution thereof.
- X 16 is M or a conservative substitution ⁇ thereof.
- X 1 is F conservative substitution thereof
- X 2 is W or a conservative substitution thereof
- X 3 is D or a conservative substitution thereof
- X 6 is Aib
- X 10 is f
- X 11 is D or a conservative substitution thereof
- X 14 is E or a conservative substitution thereof
- X 16 is M or a conservative substitution thereof (SEQ ID NO:44).
- X 1 is F
- X 2 is W
- X 3 is D
- X 6 is Aib
- X 10 is f
- X 11 is D
- X 14 is E
- X 16 is M or a conservative substitution thereof (SEQ ID NO:44).
- X 1 is F or a conservative substitution thereof.
- X 2 is W or a conservative substitution thereof.
- X 3 is D or a conservative substitution thereof.
- X 6 is Aib.
- X 10 is f.
- X 11 is D or a conservative substitution thereof.
- X 14 is E or a conservative substitution thereof.
- X16 is Nle.
- X1 is F or a conservative substitution thereof
- X 2 is W or a conservative substitution thereof
- X 3 is D or a conservative substitution thereof
- X6 is Aib
- X10 is f
- X11 is D or a conservative substitution thereof
- X 14 is E or a conservative substitution thereof
- X 16 is Nle (SEQ ID NO:45).
- X 1 is F
- X 2 is W
- X 3 is D
- X 6 is Aib
- X 10 is f
- X 11 is D
- X 14 is E
- X 16 is Nle (SEQ ID NO:45).
- X 1 is F or a conservative substitution thereof.
- X 2 is W or a conservative substitution thereof.
- X 3 is D or a conservative substitution thereof.
- X 6 is Aib.
- X 10 is f.
- X 11 is D or a conservative substitution thereof.
- X 14 is E or a conservative substitution thereof.
- X 16 is Hsm.
- X 1 is F or a conservative substitution thereof
- X 2 is W or a conservative substitution thereof
- X 3 is D or a conservative substitution thereof
- X 6 is Aib
- X 10 is f
- X 11 is D or a conservative substitution thereof
- X 14 is E or a conservative substitution thereof
- X 16 is Hsm (SEQ ID NO:46).
- X 1 is F
- X 2 is W
- X 3 is D
- X 6 is Aib
- X 10 is f
- X 11 is D
- the above engineered peptides are described as monomers, in some instances they can be in the form of a multimer including, but not limited to, a dimer, trimer, tetramer, pentamer, hexamer, etc.
- the multimer can include the same monomer (e.g., as in a homodimer, homotrimer, etc.).
- the multimer can include distinct monomers (e.g., as in a heterodimer, heterotrimer, etc.).
- the monomers multimer can be associated with one another via a covalent bond, a hydrogen bond and/or an ionic bond.
- the monomers of the multimer can be associated with one another via a linker.
- the engineered peptides can include a disulfide bond between C residues, such as those at positions 4 and 13. In other instances, the engineered peptides can include a C-terminal amide.
- Conjugates and Fusions [00130] The engineered peptides herein can be used to deliver a therapeutic agent to a cell, tissue, organ or system of an individual. In this manner, an engineered peptide herein can be conjugated to the therapeutic agent (i.e., a conjugate where the parts are linked to one another via one or more linkers).
- an engineered peptide herein can be fused to the therapeutic agent (i.e., a fusion where the parts are directly linked to one another).
- the engineered peptide includes an amino acid sequence having at least about 90% sequence identity to any one of SEQ ID NOS:3 to 16, 18, 29 to 35 and 44 to 46.
- the amino acid sequence can have at least about 90%, about 91%, about 92%, about 93%, about 94%, about 95%, about 96%, about 97%, about 98%, about 99% or about 100% sequence identity to any one of SEQ ID NOS:3 to 16, 18, 29 to 35 and 44 to 46.
- the amino acid sequence can have at least about 95% sequence identity to any one of SEQ ID NOS:3 to 16, 18, 29 to 35 and 44 to 46. In certain instances, the amino acid sequence can be any one of SEQ ID NO:3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 18, 29, 30, 31, 32, 33, 34, 35, 44, 45 or 46.
- the engineered peptide can be a monomer. In other instances, the engineered peptide can be in the form of a multimer including, but not limited to, dimers, trimers, tetramers, pentamers, hexamers, etc.
- the multimer can include the same monomer (e.g., a homodimer) or can include distinct monomers (e.g., a heterodimer).
- the therapeutic agent can be an oligonucleotide, another peptide or protein (including antibodies or fragments thereof) or a small molecule.
- the therapeutic agent when it is an oligonucleotide, it can be an aRNA agent, an eRNA agent, an iRNA agent, a mRNA agent or a tRNA agent.
- oligonucleotides ⁇ include, but are not limited to, ASOs, mRNAs, miRNAs, short activating RNAs (saRNAs), siRNAs, sup-tRNA and decoys.
- the engineered peptide can be at a 3'-end, both a 5'-end, a 3'-end and a 5'-end, and/or an internal nucleotide.
- the therapeutic agent when it is another peptide or a protein (i.e., distinct from the engineered peptide herein), it can be an Ab or fragment thereof, an anti-infective agent, an anti- rejection agent, an antioxidant, a chemotherapeutic, a growth factor, a hormone, a neurotransmitter, a receptor agonist or antagonist, a toxin, etc.
- the engineered peptide can be at an N-terminal end of the peptide or protein, a C-terminal end of the peptide or protein, both a N-terminal end and a C-terminal end of the peptide or protein, and/or an internal amino acid.
- the therapeutic agent when it is a small molecule (i.e., an organic compound of about ⁇ 1000 Da), it can be a small molecule that intervenes with a biological function and can be, for example, a chemotherapeutic or a toxin.
- the therapeutic agent can be conjugated or fused to an N-terminal end of the engineered peptide.
- the therapeutic agent can be conjugated or fused to a C-terminal end of the engineered peptide.
- one therapeutic agent can be conjugated or fused to the N-terminal end and an identical therapeutic agent can be conjugated to the C-terminal end.
- one therapeutic agent can be conjugated or fused to the N-terminal end and a distinct therapeutic agent can be conjugated to the C-terminal end.
- the therapeutic agent can be conjugated to an internal amino acid of the engineered peptide with a functional group available for conjugation (e.g., a side chain of the amino acid. Examples of amino acids with a functional group available for conjugation include, but are not limited to C (thiol), D (carboxyl), E (carboxyl), K (amino) and Q (amide).
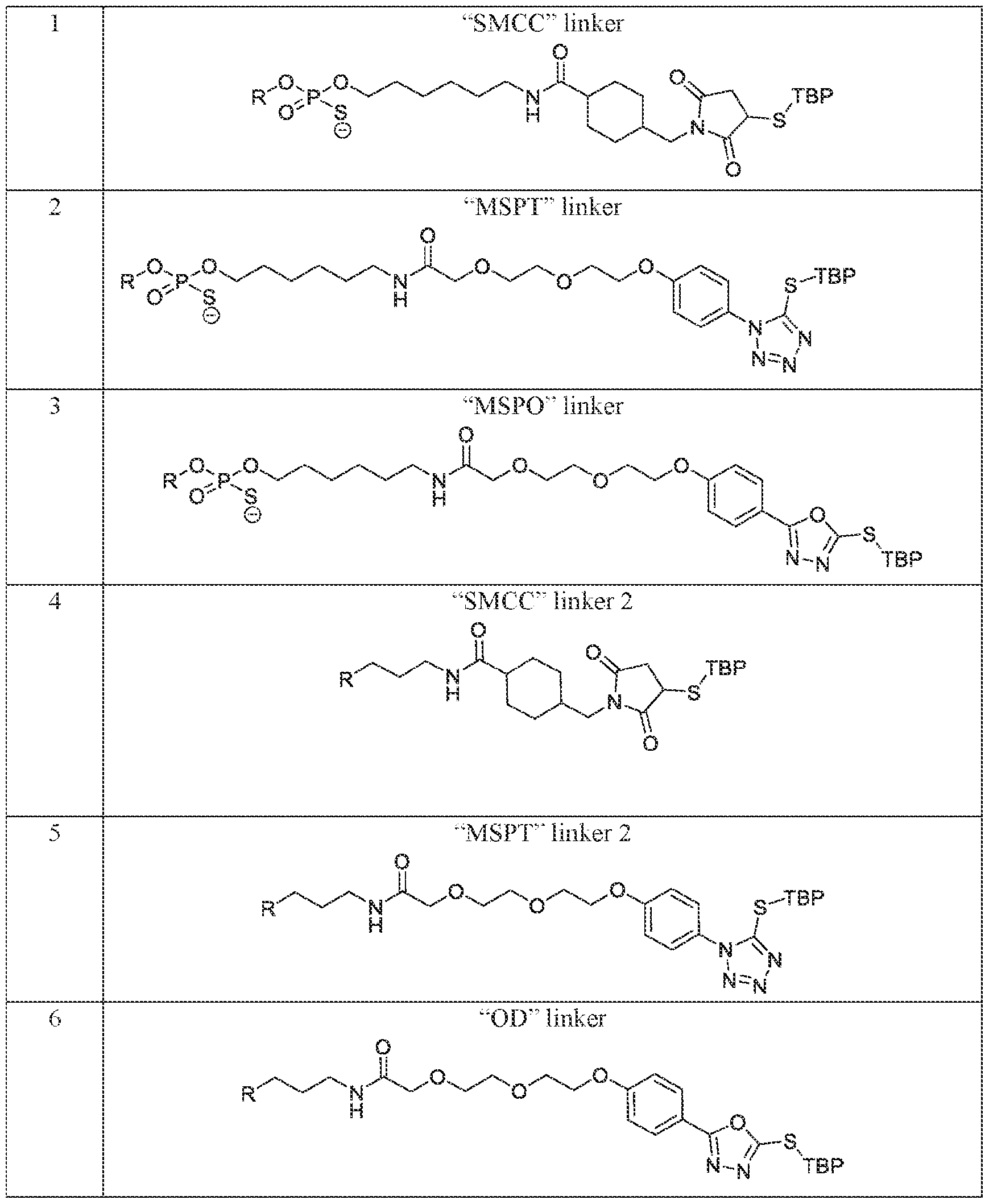
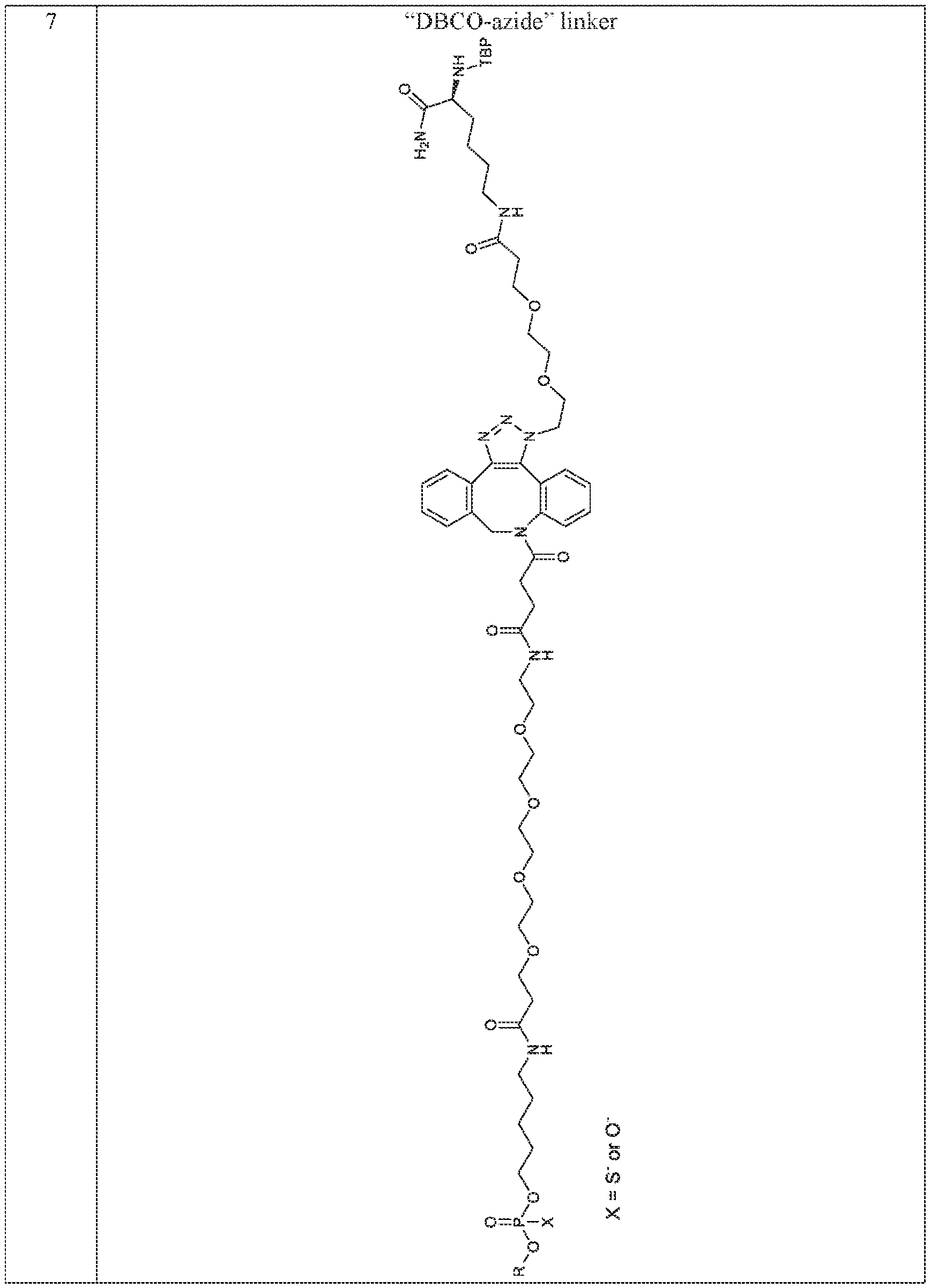
- the therapeutic agent i.e., “R” can be linked to the engineered peptide (i.e., “TBP”) via a linker.
- Exemplary linkers include, but are not limited to, the linkers depicted in Table 3 below.
- Table 3 Exemplary TBP and Therapeutic Agent Linkers.
- Linker Structure ⁇ 1 linker ⁇ 7 “DBCO-azide” linker
- Delivery Vehicles ⁇ [00144] The engineered peptides herein incorporated into a delivery vehicle to deliver a therapeutic agent to a cell, tissue, organ or system of an individual.
- an engineered peptide herein can be incorporated into, for example, a nanomedicine such as a NP (e.g., a dendrimer, liposome, LNP, eNCP, NC, NCR, NE, PLV or RNA NP).
- a NP e.g., a dendrimer, liposome, LNP, eNCP, NC, NCR, NE, PLV or RNA NP.
- an engineered peptide herein can be incorporated into a nanomedicine such as, for example, a capsid of a recombinant virus such as a rAAV.
- the engineered peptide includes an amino acid sequence having at least about 90% sequence identity to any one of SEQ ID NOS:3 to 16, 18, 29 to 35 and 44 to 46.
- the amino acid sequence can have at least about 90%, about 91%, about 92%, about 93%, about 94%, about 95%, about 96%, about 97%, about 98%, about 99% or about 100% sequence identity to any one of SEQ ID NOS:3 to 16, 18, 29 to 35 and 44 to 46.
- the amino acid sequence can have at least about 95% sequence identity to any one of SEQ ID NOS:3 to 16, 18, 29 to 35 and 44 to 46. In certain instances, the amino acid sequence is any one of SEQ ID NO:3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 18, 29, 30, 31, 32, 33, 34, 35, 44, 45 or 46.
- the delivery vehicle is a NP, it can be a dendrimer, an eNCP, a liposome, a LNP, a NC, a NCR, a NE, a PLV or a RNA NP.
- the delivery vehicle is a recombinant virus, it can be a rAAV.
- AAV capsid protein serotypes include, but are not limited to, AAV1, AAV2, AAV3, AAV4, AAV5, AAV6, AAV7, AAV8 and AAV9 or variants thereof. See, e.g., Intl. Patent Application Publication No. WO 2005/033321.
- Pharmaceutical Compositions [00149] The conjugates herein, fusions herein and delivery vehicles herein can be formulated as pharmaceutical compositions that include the conjugate, fusion or delivery vehicle and a pharmaceutically acceptable carrier. Such pharmaceutical compositions and techniques for preparing the same are well known in the art. See, e.g., Remington, “The Science and Practice of Pharmacy” (D.B.
- the conjugates, fusions or delivery vehicles may be reacted with any of a number of inorganic and organic acids/bases to form pharmaceutically acceptable acid/base addition salts.
- Pharmaceutically acceptable salts and common methodologies for preparing them are well ⁇ known in the art (see, e.g., Stahl et al., of Pharmaceutical Salts: Properties, Selection and Use,” 2 nd Revised Edition (Wiley-VCH, 2011)).
- Pharmaceutically acceptable salts for use herein include sodium, trifluoroacetate, hydrochloride and acetate salts.
- the pharmaceutical compositions further include an excipient, which can confer to a composition improved stability, improved absorption, improved solubility and/or therapeutic enhancement of the active ingredient.
- the excipient can be a buffering agent (e.g., sodium citrate, sodium phosphate, a tris base or sodium hydroxide) or a vehicle (e.g., a buffered solution, petrolatum, dimethyl sulfoxide or mineral oil).
- the conjugates, fusions or delivery vehicles can be lyophilized for extending shelf-life and then made into a solution before use (e.g., administration to an individual).
- the excipient in the pharmaceutical compositions including the conjugates, fusions or delivery vehicles can be a lyoprotectant (e.g., mannitol, lactose, polyethylene glycol, or polyvinylpyrrolidone) or a collapse temperature modifier (e.g., dextran, FicollTM or gelatin).
- a lyoprotectant e.g., mannitol, lactose, polyethylene glycol, or polyvinylpyrrolidone
- a collapse temperature modifier e.g., dextran, FicollTM or gelatin.
- the pharmaceutical compositions can include one or more additional therapeutic agents.
- the pharmaceutical composition can be administered by any route, including enteral (e.g., oral), parenteral, intravenous, intramuscular, intra-arterial, intramedullary, intrathecal, subcutaneous, intraventricular, transdermal, interdermal, rectal, intravaginal, intraperitoneal, topical (as by powders, ointments, creams and/or drops), mucosal, nasal, buccal, sublingual; by intratracheal instillation, bronchial instillation and/or inhalation; and/or as an oral spray, nasal spray, and/or aerosol.
- enteral e.g., oral
- parenteral intravenous, intramuscular, intra-arterial, intramedullary
- intrathecal subcutaneous, intraventricular, transdermal, interdermal, rectal, intravaginal, intraperitoneal
- topical as by powders, ointments, creams and/or drops
- mucosal nasal, buccal, sublingual
- Specifically contemplated routes are oral administration, IV injection (e.g., systemic intravenous injection), SC injection, ICV injection, ICM injection, IPM injection, IT injection or a combination thereof.
- IV injection e.g., systemic intravenous injection
- SC injection e.g., SC injection
- ICV injection e.g., SC injection
- ICV injection e.g., SC injection
- ICM injection e.g., styline injection
- IPM injection e.g., IT injection or a combination thereof.
- the most appropriate route of administration will depend upon a variety of factors including, but not limited to, the nature of the agent (e.g., its stability in the environment of its administration and/or intended target) and/or the condition of the individual (e.g., whether the subject is able to tolerate oral administration).
- the engineered nucleic acids, rAAV or pharmaceutical compositions are suitable for administration to, for example, the CNS of an individual.
- kits ⁇ [00156]
- an engineered herein, conjugate herein, fusion herein, delivery vehicle herein and/or pharmaceutical composition herein can be included in a kit that includes the same and instructions for its use.
- the kit includes an engineered peptide, conjugate, fusion, delivery vehicle and/or pharmaceutical composition and a package insert containing instructions for use of the kit and/or any component thereof.
- the kit comprises, in a suitable container or other means for containing, the engineered peptide, conjugate, fusion, delivery vehicle and/or pharmaceutical composition, one or more controls, and various buffers, reagents, enzymes and other standard ingredients well known in the art.
- the container comprises at least one vial, well, test tube, flask, bottle, syringe or other container means, into which the engineered peptide, conjugate, fusion, delivery vehicle and/or pharmaceutical composition is placed, and in some instances, suitably aliquoted.
- the kit includes additional containers into which this component is placed.
- the kits also can include a means for containing the engineered peptide, conjugate, fusion, delivery vehicle and/or pharmaceutical composition and any other reagent in close confinement for commercial sale.
- Such containers may include injection or blow-molded plastic containers into which the desired vials are retained.
- Containers and/or kits can include labeling with instructions for use and/or warnings.
- the engineered peptides herein can be synthesized via any number of peptide synthesis methods known in the art using standard manual or automated solid-phase synthesis procedures. Automated peptide synthesizers are commercially available from, for example, Applied Biosystems (Foster City, CA) and Protein Technologies Inc. (Tucson, AZ). Reagents for solid-phase synthesis are readily available from commercial sources. Solid-phase synthesizers can be used according to the manufacturer’s instructions for blocking interfering groups, protecting amino acids during reaction, coupling, deprotecting and capping of unreacted amino acids.
- the peptides can also be expressed recombinantly, or by using in vitro transcription translation machinery, or by using a combination of the said methods.
- an N- ⁇ -carbamoyl-protected amino acid and the N-terminal amino acid on the growing peptide chain attached to a resin are coupled at room temperature in an inert solvent ⁇ such as DMF, N-methylpyrrolidone or chloride in the presence of coupling agents such as diisopropy1-carbodiimide and 1-hydroxybenzotriazole.
- N- ⁇ -carbamoyl protecting group is removed from the resulting peptide resin using a reagent such TFA or piperidine, and the coupling reaction is repeated with the next desired N- ⁇ -protected amino acid to be added to the peptide chain.
- Suitable amine protecting groups are well known in the art and are described, for example, in Green & Wuts, “Protecting Groups in Organic Synthesis,” (John Wiley and Sons, 1991). The most commonly used examples include tBoc and Fmoc. After completion of synthesis, peptides are cleaved from the solid-phase support with simultaneous side chain deprotection using standard treatment methods under acidic conditions.
- Peptides can be solubilized in aqueous buffers over a wide pH range.
- Methods of Transporting Therapeutic Agents Across the BBB can include the steps described herein, and these maybe be, but not necessarily, carried out in the sequence as described. Other sequences, however, also are conceivable. Moreover, individual or multiple steps bay be carried out either in parallel and/or overlapping in time and/or individually or in multiply repeated steps. Furthermore, the methods may include additional, unspecified steps.
- the conjugates herein, fusions herein, delivery vehicles herein or pharmaceutical compositions herein can be used for transporting a therapeutic agent across the BBB.
- the methods can include at least a step of administering to the individual an effective amount of a conjugate, a fusion, a delivery vehicle and/or a pharmaceutical composition herein.
- the administering can be via direct (i.e., central) injection into the CNS of the individual, which can be an ICV injection, an ICM injection, an IPM injection, an IT injection or a combination thereof.
- the administering can be via a peripheral injection.
- the peripheral injection can be via IV injection or SC injection.
- the administering can be by a of direct injection and peripheral injection.
- the methods can include a step of administering an effective amount of one or more additional therapeutic agents other than those discussed above.
- the methods below can include the steps described herein, and these may be, but not necessarily, carried out in the sequence as described. Other sequences, however, also are conceivable. Moreover, individual or multiple steps may be carried out either in parallel and/or overlapping in time and/or individually or in multiply repeated steps. Furthermore, the methods may include additional, unspecified steps.
- the conjugates herein, fusion proteins herein, delivery vehicles herein or pharmaceutical compositions herein can be used for treating individuals having a CNS disease, disorder or condition.
- the methods can include at least a step of administering to the individual an effective amount of a conjugate, a fusion, a delivery vehicle and/or a pharmaceutical composition.
- the administering can be via direct injection into the CNS of the individual, which can be an ICV injection, an ICM injection, an IPM injection, an IT injection or a combination thereof.
- the administering can be via a peripheral injection.
- the peripheral injection can be via IV injection or SC injection.
- the administering can be by a combination of direct injection and peripheral injection.
- the methods can include a step of administering an effective amount of one or more additional therapeutic agents other than those discussed above.
- the conjugates herein, fusion proteins herein, delivery vehicles herein or pharmaceutical compositions herein can be used for treating individuals having a TfR- associated disease, disorder or condition.
- the methods can include at least a step of administering to the individual an effective amount of a conjugate, a fusion, a delivery vehicle and/or a pharmaceutical composition.
- the administering can be via direct injection into the CNS of the individual, which can be an ICV injection, an ICM injection, an IPM injection, an IT injection or a combination thereof.
- the administering can be via a peripheral injection.
- the peripheral injection can be via IV ⁇ injection or SC injection.
- the administering can be by a combination of direct injection and peripheral injection.
- the methods can include a step of administering an effective amount of one or more additional therapeutic agents other than those discussed above.
- a conjugate herein, fusion herein, delivery vehicle herein and/or pharmaceutical composition herein can be used, or adapted for use, to transport a therapeutic agent across the BBB of an individual.
- the conjugate, fusion, delivery vehicle and/or pharmaceutical composition is provided for use, or adapted for use, to transport a therapeutic agent across the BBB.
- the conjugate, fusion, delivery vehicle and/or pharmaceutical composition is provided for use, or adaptable for use, in the manufacture of a medicament or a pharmaceutical composition for transporting a therapeutic agent across the BBB.
- a conjugate herein, fusion herein, delivery vehicle herein and/or pharmaceutical composition herein can be used, or adapted for use, to treat an individual (e.g., a human) having a CNS disease, disorder or condition.
- the conjugate, fusion, delivery vehicle and/or pharmaceutical composition is provided for use, or adapted for use, to treat an individual having a CNS disease, disorder or condition.
- the conjugate, fusion, delivery vehicle and/or pharmaceutical composition is provided for use, or adaptable for use, in the manufacture of a medicament or a pharmaceutical composition for treating a CNS disease, disorder or condition.
- a conjugate herein, fusion herein, delivery vehicle herein and/or pharmaceutical composition herein can be used, or adapted for use, to treat an individual (e.g., a human) having a TfR-associated disease, disorder or condition.
- the conjugate, fusion, delivery vehicle and/or pharmaceutical composition is provided for use, or adapted for use, to treat an individual having a TfR-associated disease, disorder or condition.
- the conjugate, fusion, delivery vehicle and/or pharmaceutical composition is provided for use, or adaptable for use, in the manufacture of a medicament or a pharmaceutical composition for treating a TfR-associated disease, disorder or condition.
- conjugates, fusions, delivery vehicles and/or pharmaceutical compositions for use in therapy. Furthermore, conjugates, fusions, delivery vehicles and/or pharmaceutical compositions are described herein for use in transporting a therapeutic agent ⁇ across the BBB or in the treatment of a disorder or condition or in the treatment of a TfR-associated disease, disorder or condition. [00178] Also described are use of conjugates, fusions, delivery vehicles and/or pharmaceutical compositions in the manufacture of a medicament for transporting a therapeutic agent across the BBB or for the treatment of a CNS disease, disorder or condition or for the treatment of a TfR-associated disease, disorder or condition. EXAMPLES [00179] The following non-limiting examples are offered for purposes of illustration, not limitation.
- TfR binding peptides using phage libraries Two peptide libraries – 282 and 585 – were selected against the hTfR extracellular domain (SEQ ID NO:2). These peptide libraries are 14 and 20 residues long, respectively, and contain 8 random amino acids within 2 engineered Cys residues. The libraries differ by having either 2 (282 library) or 5 (585 library) random sequences flanking each engineered Cys residue. [00184] Both libraries were constructed using a backbone phage vector as described previously (Chang et al.
- Phage displaying these peptides such as Peptide 9, internalized in hCMEC/D3 cells; whereas naked phage that lacked any peptide did not show binding at 4oC or internalizing at 37oC (see, FIGS.1A and 1B).
- a known TfR Ab was used as positive control that showed significant level of internalization (MEM189; see, Sade et al. ⁇ (2014) PLoS One 9:e96340).
- Anti-phage DL650 was used in both the binding and internalizing assays (commercially available from abcam; Waltham, MA). [00186] Table 4: Phage Library and Peptide Sequences.
- peptides with fast off-rate dissociate bio-hTfR and bind to unlabeled TfR present in excess amount.
- peptides with slower off-rate remain bound to the bio-hTfR.
- streptavidin-coated beads separated peptides with slower off-rate from the mix.
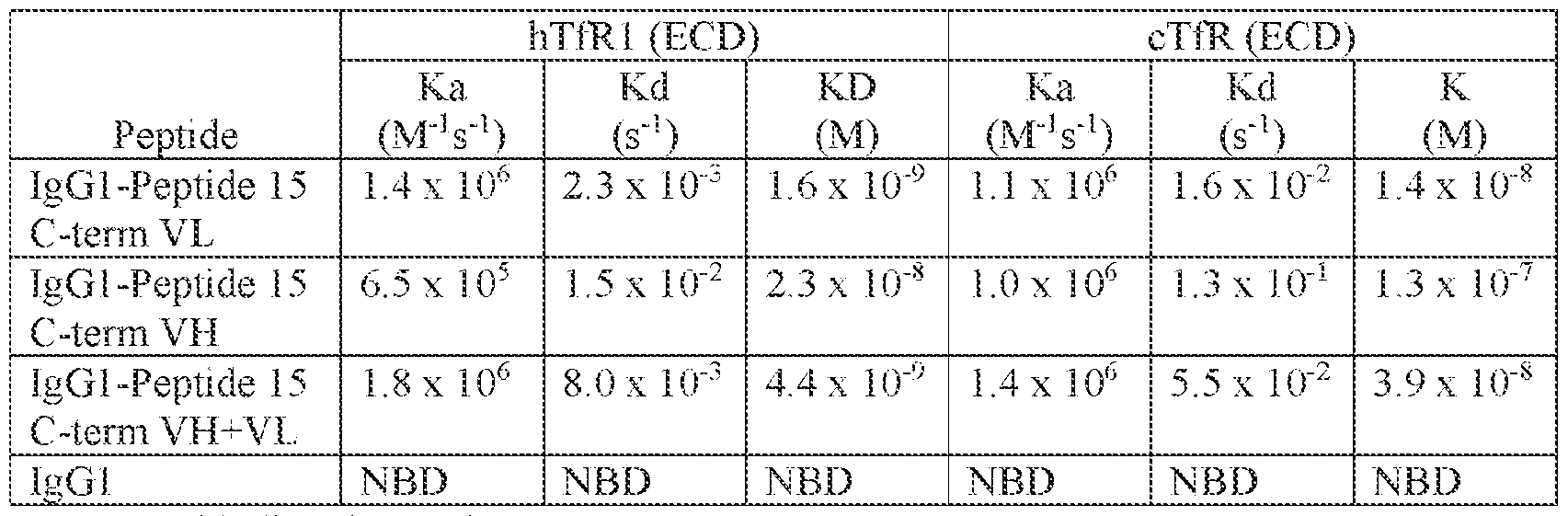
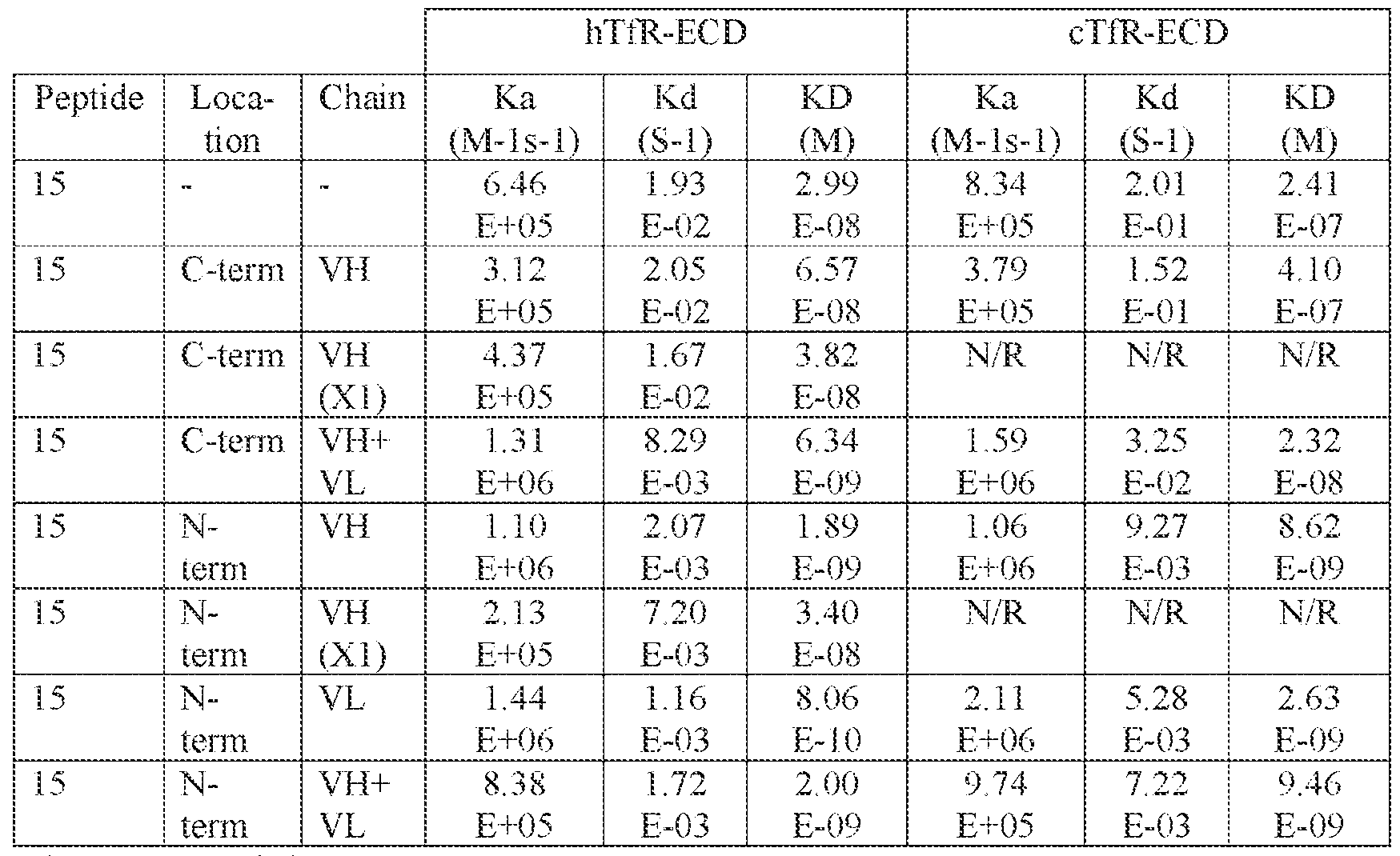
- Peptide 15 bound hTfR-expressing cells (i.e., EFO- 21) with EC 50 of 17 nM and to cTfR-expressing cells (i.e., MDCK-c6) at EC 50 of 730 nM.
- Parental peptide, Peptide 8 SEQ ID NO:10
- Peptide 15 did not bind to murine TfR (mTfR)-expressing cells or recombinant protein.
- Table 5 Peptide 8 and Peptide well as Peptide 15 Variants, Binding to hTfR and cTfR.
- siRNA conjugates Peptides 15 and 17 were conjugated to siRNA targeting synuclein (SNCA; SEQ ID NO:42) or hypoxanthine-guanine phosphoribosyltransferase (HPRT; SEQ ID NO:43) with phosphorothioate (PS) linkage(s) at 3' end and a vinylphosphonate (VP) cap at 5' of the sense and anti-sense strands, respectively.
- PS phosphorothioate
- VP vinylphosphonate
- a PEG12 linker and DBCO click chemistry was used to prepare the conjugates. For all the conjugates prepared, more than 60% yield and 94% purity were obtained.
- the sense strand was conjugated to the peptide C- terminus via azide click chemistry.
- a cholesterol-conjugated control siRNA also was synthesized.
- Knockdown assay The siRNA conjugates were tested for gene knockdown in the EFO-21 cell line.
- Plasma stability The siRNA conjugates were tested for plasma stability in human and cynomolgus plasma for 24 hr.
- Results With regard to SNCA, Peptide 15-conjugated siRNA targeting SNCA remained fully intact in the presence of human and non-human (i.e., cynomolgus) plasma for up to 24 hr.
- Peptide 15-conjugated siRNA showed comparable knockdown efficacy to cholesterol-conjugated siRNA with nearly 3-fold stronger IC 50 at 9 nM.
- Peptide 17-conjugated siRNA did not induce any gene silencing, indicating that the observed activity was mediated via interaction of Peptide 15 with TfR (see, FIG. 5A).
- Peptide 17- conjugated siRNA did not induce any SNCA gene silencing (see also, FIG.5A).
- Fusion proteins Fusion proteins were generated by cloning Peptide 15 (SEQ ID NO:18) at the C-terminus of either heavy (VH), light (VL) or both (VH + VL) chains of an IgG1. The fusion proteins were evaluated for binding to recombinant and cell-expressed TfR- 1 using SPR (as described above) or flow cytometry, respectively.
- Flow cytometry The EFO-21 cell line was used to evaluate binding of the fusion proteins to hTfR-1-expressing cells.
- EFO-21 cells were grown to confluency prior to detachment from plates using TrypleE. Cell viability and count was next determined by a Vi- CELLTM cell and particle counter. Cells were transferred to 96-well plates at density of 5 x 10 5 cells per well, washed with FACS buffer (1% FBS/PBS, 0.1% NaN 3 , plus 2 mM EDTA) and stained with LIVE/DEAD dye for 30 min while on ice. Subsequently, cells were washed two times with FACS buffer and then incubated with Ab-peptide fusions for 1 hr on ice. Cells were washed again two times with FACS buffer and incubated with anti-human IgG PE for 1 hr on ice.
- FACS buffer 1% FBS/PBS, 0.1% NaN 3 , plus 2 mM EDTA
- AZDye 647 was conjugated to fusion proteins using a site-specific conjugation technology. Briefly, glycans were removed by EndoS followed by adding azidosugar. AZDye 647-DBCO was conjugated with the azidosugar on the Ab via a click chemistry reaction.
- Fluorescently labeled fusion proteins were injected intravenously at 10 mg/kg to humanized-TfR transgenic mice. 48 hr post dose, mice were euthanized, and brain and gastrocnemius were isolated. Subsequently, isolated brain and gastrocnemius were imaged using an inverted LSM 980 Confocal Microscope (Carl Zeiss). [00216] Biodistribution via biochemical analysis: CNS exposure of fusion proteins was evaluated in humanized-TfR transgenic mice by administering a single 5 mg/kg IV injection of either a mAb fused with a single TfR peptide (2:1) or two TfR binding peptides (2:2) fused to the C-terminal of VH.
- Table 7 shows binding of various fusion proteins (Peptide 15 fused to IgG1 at the specified site) to hTfR in EFO-21 cells, which is a human ovarian tumor cell line with high hTfR expression by flow cytometry. Again, naked IgG1 was used as negative control and did not show any binding to EFO-21 by flow cytometry.
- Table 6 Fusion Protein Binding to hTfR and cTfR Using SPR.
- Table 7 Fusion Protein Binding to hTfR and cTfR Using Flow Cytometry.
- Example 5 Peptide-Directed siRNA Targeting In Vivo
- Methods [00225] Animal model: hTfR transgenic mice were injected 1 mg/kg or 10 mg/kg of Peptide 15-conjugated siRNA or Peptide 19-conjugated siRNA (fluorophore labeled) targeting HPRT that were made according to Example 3. Images were taken at 1 hr or 24 hr post injection. Live images were taken from liver, gastric and adipose tissue, and ex vivo images were taken from hypothalamus and DRG neurons. [00226] The siRNA conjugates were intravenously (IV) dosed in mice at 2.5 mg/kg, 5.0 mg/kg and 10 mg/kg.
- IV intravenously
- the following nucleotide and/or amino acid sequences are referred to in the disclosure above and are provided below for reference.
- SEQ ID NO:1 – human TfR1 (760 aa) MMDQARSAFSNLFGGEPLSYTRFSLARQVDGDNSHVEMKLAVDEEENADNNTKAN VTKPKRCSGSICYGTIAVIVFFLIGFMIGYLGYCKGVEPKTECERLAGTESPVREEPGE DFPAARRLYWDDLKRKLSEKLDSTDFTGTIKLLNENSYVPREAGSQKDENLALYVE NQFREFKLSKVWRDQHFVKIQVKDSAQNSVIIVDKNGRLVYLVENPGGYVAYSKAA TVTGKLVHANFGTKKDFEDLYTPVNGSIVRAGKITFAEKVANAESLNAIGVLIYM DQTKFPIVNAELSFFGHAHLGTGDPYTPGFPSFNHTQFPPSRSSGLPNIPVQTISRAAAE KLFGNMEGDCPSDWKTDSTCRMVTSESKNVKLTVSNVLKEIKILNIFGVIKGFVEPD HYV
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Biochemistry (AREA)
- Biophysics (AREA)
- General Health & Medical Sciences (AREA)
- Genetics & Genomics (AREA)
- Medicinal Chemistry (AREA)
- Molecular Biology (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
Abstract
Engineered transferrin receptor (TfR) binding peptides are disclosed that can be used to deliver a therapeutic agent into a cell, tissue, organ or system of an individual. Conjugates, fusions, delivery vehicles and pharmaceutical compositions including the same also are disclosed, as well as methods of using the same for treating central nervous system (CNS) diseases, disorders or conditions or for treating TfR-associated diseases, disorders or conditions.
Description
ENGINEERED TRANSFERRIN RECEPTOR BINDING PEPTIDES AS WELL AS METHODS OF MAKING AND USING THE SAME REFERENCE TO SEQUENCE LISTING SUBMITTED ELECTRONICALLY [001] The disclosure is being filed along with a Sequence Listing in ST.26 XML format. The Sequence Listing is provided as a file titled “txt_30429A_US_PRI” created 30 May 2024 and is 50 kilobytes (kb) in size. The Sequence Listing information in the ST.26 XML format is incorporated herein by reference in its entirety. TECHNICAL FIELD [002] The disclosure relates generally to biology and protein engineering and optimizing, and more particularly it relates to engineered transferrin receptor (TfR; or CD71) binding peptides that can be used to deliver a therapeutic agent to a cell, tissue, organ or system of an individual, as well as relates to methods of using the same for treating central nervous system (CNS) diseases, disorders or conditions or for treating TfR-associated diseases, disorders or conditions. BACKGROUND [003] Successful, active transport of therapeutic agents and/or carriers of such therapeutic agents to intracellular targets requires entry and/or passage through a cell membrane. Despite its selective permeability to compounds and molecules essential to cell function and survival, the cell membrane is a particularly daunting barrier to entry of therapeutic agents. As there are three to four times more intracellular targets than cell surface targets for therapeutic agents, many delivery systems have been developed to facilitate passage of therapeutic agents across the cell membrane to reach their intracellular target. [004] Cell-penetrating peptides or nanoparticles can be used for delivering therapeutic agents across a cell membrane. Likewise, lipids or liposomes can be used for delivering therapeutic agents across a cell membrane. These approaches, however, may lack cellular specificity and may induce toxicity associated with broad exposure to therapeutic agents. [005] In contrast, receptor-mediated internalization is a selective approach for delivering therapeutic agents across a cell membrane to intracellular targets. For example, a therapeutic agent can be conjugated to a peptide (e.g., RGD peptides or TAT peptides) or to an antibody ^
or a fragment thereof (e.g., antibody-drug (ADC)) that binds to a cell surface receptor, which is internalized and enters a cell's cytoplasm. Hence, receptor-mediated internalization offers intracellular activity with the added benefit of specificity. [006] One target of interest for exploiting receptor-mediated internalization is the TfR. See, e.g., McQuiad et al. (2021) PLoS ONE 16:e0252341; and Intl. Patent Application Publication Nos. WO 2020/144233 and 2022/026555. Additional TfR binding agents are needed, however, especially TfR binding peptides that have rapid internalization kinetics, that have improved endosomal escape rates, and that can prevent receptor degradation by inducing recycling. BRIEF SUMMARY [007] To address this need, the disclosure first describes an engineered peptide that can bind to the TfR (i.e., a TfR binding agent) and that can be used to deliver a therapeutic agent into (i.e., endocytosis) or across/through (i.e., transcytosis) a cell, tissue, organ or system of an individual, where the engineered peptide includes an amino acid sequence having at least about 95% sequence identity to any one of SEQ ID NOS:3 to 16, 18, 29 to 35 and 44 to 46. In certain instances, the engineered peptide is SEQ ID NO:3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 18, 29, 30, 31, 32, 33, 34, 35, 44, 45 or 46. [008] In some instances, the engineered peptide can be a monomer. In other instances, the engineered peptide can be in the form of a multimer (e.g., a dimer, trimer, tetramer, etc.). Moreover, the multimer can include the same monomer (e.g., a homodimer, homotrimer, homotetramer, etc.) or can include distinct monomers (e.g., a heterodimer, heterotrimer, heterotetramer, etc.). [009] Alternatively, the disclosure describes an engineered peptide that can bind to the TfR and that can be used to deliver a therapeutic agent into or across/through (i.e., transcytosis) a cell, tissue, organ or system of an individual, where the engineered peptide includes an amino acid sequence of: X1X2X3CRX6FYPX10X11YCX14AX16 (Formula I; SEQ ID NO:17), wherein X1, X2, X3, X6, X10, X11, X14 and X16 can be any naturally occurring amino acid or can be a variant thereof. In some instances, X1, X2 X10 each and independently can be an aromatic amino acid. In some instances, X3, X11 and X14 each and independently can be an acidic amino acid. In other instances, X1 can be F, I, L, M, W or Y. In other instances, X2 can be F, I, L, M, W or Y. In other instances, X3 can be D, E, H, K, N, R, Q or S. In other instances, ^
X6 can be S or Aib. In other instances, X10 be f, F, I, L, M, W or Y. In other instances, X11 can be D, E, H, K, N, R, Q or S. In other instances, X14 can be D, E, H, K, N, R, Q or S. In other instances, X16 is M, Nle or Hsm. In certain instances, X1 is F, X2 is W, X3 is D, X6 is S, X10 is F, X11 is D, X14 is E and X16 is M (SEQ ID NO:18). In certain instances, X1 is F, X2 is W, X3 is D, X6 is Aib, X10 is f, X11 is D, X14 is E and X16 is M (SEQ ID NO:44). In certain instances, X1 is F, X2 is W, X3 is D, X6 is Aib, X10 is f, X11 is D, X14 is E and X16 is Nle (SEQ ID NO:45). In certain instances, X1 is F, X2 is W, X3 is D, X6 is Aib, X10 is f, X11 is D, X14 is E and X16 is Hsm (SEQ ID NO:46). [0010] Second, the disclosure describes conjugates and fusions including an engineered peptide herein and a therapeutic agent linked thereto. [0011] In some instances, the therapeutic agent can be an oligonucleotide, another peptide or protein, or a small molecule. In some instances, the therapeutic agent is indirectly linked to the engineered peptide via, for example, a linker (i.e., a conjugate). In other instances, the therapeutic agent is directly linked to the engineered peptide (i.e., a fusion). [0012] In some instances, the therapeutic agent can be conjugated or fused to an amino (N)- terminal end of the engineered peptide. In other instances, the therapeutic agent can be conjugated or fused to a carboxy (C)-terminal end of the engineered peptide. In yet other instances, the therapeutic agent can be conjugated or fused to an internal amino acid of the engineered peptide. [0013] In some instances, such as when the therapeutic agent is an oligonucleotide, the engineered peptide can be conjugated or fused at least to a 5'-end of the therapeutic agent. In other instances, the engineered peptide can be conjugated or fused to at least to a 3'-end of the therapeutic agent. In yet other instances, the engineered peptide can be conjugated or fused to at least one or more internal nucleotides of the therapeutic agent. [0014] In some instances, such as when the therapeutic agent is a peptide or protein, the engineered peptide can be conjugated or fused at least to an N-terminal end of the therapeutic agent. In other instances, the engineered peptide can be conjugated or fused at least to a C- terminal end of the therapeutic agent. In yet other instances, the engineered peptide can be conjugated or fused at least to one or more internal amino acids of the therapeutic agent. [0015] Third, the disclosure describes delivery vehicles including an engineered peptide herein, a conjugate herein or a fusion herein. In some instances, the delivery vehicle can be a nanomedicine such as, for example, an enveloped nanocomplex (eNCP), a lipid nanoparticle ^
(LNP), a liposome, a nanocapsule (NC), nanocrystal (NCR), a nanoemulsion (NE), a proteolipid vehicle (PLV), a RNA nanoparticle (RNA NP) or a vector such as a recombinant adeno-associated virus (rAAV). [0016] Fourth, the disclosure describes pharmaceutical compositions including an engineered peptide herein, a conjugate herein, a fusion herein and/or a delivery vehicle herein and a pharmaceutically acceptable carrier. In some instances, the pharmaceutical compositions further can include other carriers, diluents and/or excipients. In other instances, the pharmaceutical compositions can include one or more additional therapeutic agents. [0017] Fifth, the disclosure describes methods of transporting a therapeutic agent across a blood-brain barrier (BBB) of an individual, where the methods can include at least a step of administering to the individual an effective amount of a conjugate herein, a fusion herein, a delivery vehicle herein and/or a pharmaceutical composition herein. In some instances, the administering can be via direct (i.e., central) injection into a tissue or an organ such as the CNS of the individual, which can be an intracerebroventricular (ICV) injection, an intracisterna magna (ICM) injection, an intraparenchymal (IPM) injection, an intrathecal (IT) injection or a combination thereof. In other instances, the administering can be via a peripheral injection. In certain instances, the peripheral injection can be via intravenous (IV) injection or subcutaneous (SC) injection. In yet other instances, the administering can be by a combination of direct injection and peripheral injection. [0018] Sixth, the disclosure describes methods of treating an individual having a CNS disease, disorder or condition, where the methods can include at least a step of administering to the individual an effective amount of a conjugate herein, a fusion herein, a delivery vehicle herein and/or a pharmaceutical composition herein. In some instances, the administering can be via direct injection into a tissue or an organ such as the CNS of the individual, which can be an ICV injection, an ICM injection, an IPM injection, or a combination thereof. In other instances, the administering can be via a peripheral injection. In certain instances, the peripheral injection can be via IV injection or SC injection. In yet other instances, the administering can be by a combination of direct injection and peripheral injection. [0019] Seventh, the disclosure describes methods of treating an individual having a TfR- associated disease, disorder or condition, where the methods can include at least a step of administering to the individual an effective amount of a conjugate herein, a fusion herein, a delivery vehicle herein and/or a pharmaceutical composition herein. In some instances, the ^
administering can be via direct injection a tissue or an organ such as the CNS of the individual, which can be an ICV injection, an ICM injection, an IPM injection, an IT injection or a combination thereof. In other instances, the administering can be via a peripheral injection. In certain instances, the peripheral injection can be via IV injection or SC injection. In yet other instances, the administering can be by a combination of direct injection and peripheral injection. [0020] Eighth, the disclosure describes use of a conjugate herein, a fusion herein, a delivery vehicle herein or a pharmaceutical composition herein in the manufacture of a medicament for treatment of CNS disease, disorder or condition or for treatment of a TfR-associated disease, disorder or condition. [0021] Ninth, the disclosure describes a conjugate herein, a fusion herein, a delivery vehicle herein or a pharmaceutical composition herein for use in the treatment of a CNS disease, disorder or condition or for use in the treatment of a TfR-associated disease, disorder or condition. [0022] An advantage of the engineered peptides herein is that, due to their small size, they can cross a physiological barrier such as the BBB and thus can be used to deliver therapeutic agents to the CNS when treating CNS diseases, disorders and/or conditions. [0023] An advantage of the engineered peptides herein is that they bind not only human TfR but also non-human TfR and therefore can be used for pharmacodynamic, pharmacokinetic and toxicology studies in animal models, especially non-transgenic animal models, that more readily translate from these species to humans. Likewise, and due to their cross-reactivity, the engineered peptides herein can be used not only for treating humans but also for treating animals. [0024] An advantage of the engineered peptides herein is that they are small (i.e., 20 residues or less) and thus can be efficiently transported into and/or across a cell. Likewise, and due to their size, the engineered peptides herein can be chemically synthesized in high quantities and at high purity with lower cost. [0025] An advantage of the engineered peptides herein is that they provide high yield and purity of conjugates and fusions, lower administered dose as compared to large molecules, higher tissue penetration and overall simplicity. BRIEF DESCRIPTION OF THE DRAWINGS ^
[0026] The advantages, effects, features objects other than those set forth above will become more readily apparent when consideration is given to the detailed description below. Such detailed description refers to the following drawings, where: [0027] FIGS. 1A and 1B show binding and internalization of an exemplary TfR binding peptide displayed on phage (i.e., Peptide 9) using a hCMEC/D3 cell line, where FIG.1A shows binding at 4ºC, and where FIG.1B shows internalizing at 37ºC. [0028] FIGS. 2A and 2B show binding of exemplary biotin-labeled, pure TfR binding peptides (i.e., Peptide 8 and Peptide 15) to EFO-21 (human TfR (hTfR)) or MDCK-c6 (cynomolgus TfR (cTfR)) cells as determined by flow cytometry, where FIG. 2A shows binding to EFO-21 cells, and where FIG.1B shows binding to MDCK-c6 cells. [0029] FIG. 3 shows internalizing of exemplary pure TfR binding peptides (i.e., Peptide 15 and Peptide 19) in EFO-21 cells. [0030] FIGS.4A and 4B show that an exemplary TfR binding peptide (i.e., Peptide 15) binds to hTfR at a distinct epitope from the transferrin (Tf) binding site, where FIG.4A is a heat map comparing deuterium (D2O) uptake between His-hTfR complexed with and without the exemplary TfR binding peptide, and where FIG.4B shows a putative epitope mapped on hTfR. [0031] FIGS. 5A and 5B show cellular activity of exemplary TfR binding peptide (i.e., Peptide 15 and Peptide 17)-siRNA conjugates in a TfR-expressing cell line (EFO-21), where FIG.5A shows conjugates targeting the SNCA gene in EFO-21 cells, and where FIG.5B shows conjugates targeting the HPRT gene in EFO-21 cells. [0032] FIG. 6 shows in vitro proteolytic stability of exemplary TfR binding peptide (i.e., Peptide 15 and Peptide 17)-siRNA conjugates targeting the SNCA gene in the presence of human or cynomolgus monkey serum, where GLP-1 peptide was used as control for serum activity (cyno = cynomolgus serum; hu = human serum). [0033] FIGS. 7A and 7B show in vivo imaging of biodistribution of an exemplary TfR binding peptide (i.e., Peptide 15)-antibody fusion throughout the CNS, where FIG. 7A shows distribution in the whole brain (top panel is IgG1 VL + VH-Peptide 15 C-terminal fusion, middle panel is a IgG1 VH-Peptide 15 C-terminal fusion and the lower panel is a IgG1 VL- Peptide 15 C-terminal fusion; tile-scan image of brain, and scale bar = 1000 μm), and where FIG.7B shows distribution in specific regions of the brain (top panel is IgG1 VL + VH-Peptide 15 C-terminal fusion, middle panel is a IgG1 VH-Peptide 15 C-terminal fusion and the lower panel is a IgG1 VL-Peptide 15 C-terminal fusion; first column is cortex, second column is ^
hippocampus, third column is cerebellum fourth column is thalamus; high magnification image from FIG.7A; and scale bar = 100 μm). [0034] FIG.8 shows in vivo activity of exemplary TfR binding peptides (i.e., Peptide 15 and Peptide 19)-siRNA conjugates targeting the HPRT gene in a mouse model. [0035] FIGS. 9A and 9B show in vivo imaging of biodistribution of an exemplary TfR binding peptide (i.e., Peptide 15)-siRNA conjugate in a mouse model, where FIG. 9A shows the distribution in the gastrocnemius muscle (scale bar = 50 μm), and where FIG. 9B shows the distribution in white adipose tissue, pancreatic islet, dorsal root ganglion (DRG) neurons and hypothalamus in the brain (scale bar = 50 μm, or 500 μm for hypothalamus tile scan). DETAILED DESCRIPTION [0036] Overview [0037] TfR is a transmembrane glycoprotein for Tf that functions to import iron by internalizing the Tf-iron complex through receptor-mediated endocytosis. There are two TfR isoforms, TfR1 and TfR2, where TfR1 is a high-affinity, ubiquitously expressed receptor but where TfR2 expression is restricted to certain cell types and is unaffected by intracellular iron concentrations. TfR1 is expressed in proliferative cells such as, for example, the basal epidermis, intestinal epithelium and certain activated immune cells. TfR1 also is expressed in non-proliferative cells such as, for example, orthochromatic normoblasts, reticulocytes, hepatocytes, placental trophoblasts, endothelial cells of the BBB and Sertoli cells of the blood- testis barrier (BTB). [0038] Of particular interest herein is TfR1. Exemplary nucleotide sequences for TfR1 can be found in NCBI Ref. Seq. Nos. NM_003234.4 (human), NM_011638.4 (mouse), NM_001257303.1 (non-human primate), XM_003310190.3 (non-human primate) and NM_022712.1 (rat). Likewise, exemplary amino acid sequences for TfR1 monomers can be found in NCBI Ref. Seq. Nos. NP_003225.2 (human; SEQ ID NO:1), NP_035768.1 (mouse), NP_001244232.1 (non-human primate), XP_003310238.1 (non-human primate) and NP_073203.1 (rat). One of skill in the art, however, understands that additional examples of TfR1 sequences are readily available using publicly available databases such as, for example, GenBank and UniProt. [0039] Engineered TfR binding peptides, conjugates and fusions including the same, delivery vehicles incorporating the same and compositions thereof are of interest for delivering a ^
therapeutic agent to, for example, a cell, organ or system of an individual via the TfR, especially the CNS and especially via TfR1. Likewise, such engineered peptides, conjugates, fusions, delivery vehicles and pharmaceutical compositions are of interest for treating individuals having a CNS disease, disorder or condition or having a TfR-associated disease, disorder or condition. [0040] Abbreviations and Definitions [0041] Unless defined otherwise, all technical and scientific terms used herein have the same meaning as commonly understood by one of skill in the art to which the disclosure pertains. Although any methods and materials similar to or equivalent to those described herein can be used in the practice or testing of the methods herein, the preferred methods and materials are described herein. [0042] Additionally, reference to an element by the indefinite article “a” or “an” does not exclude the possibility that more than one element is present, unless the context clearly requires that there be one and only one element. The indefinite article “a” or “an” thus usually means “at least one.” [0043] Moreover, use of “including,” as well as other forms, such as “including but not limited, “include,” “includes” and “included,” is not limiting. [0044] Certain abbreviations used herein are as follows: [0045] As used herein, “AAV” refers to adeno-associated virus; “Ab” refers to antibody; “AD” refers to Alzheimer’s disease; “ADC” refers to antibody-drug conjugate; “ADHD” refers to attention deficit hyperactivity disorder; “Aib” refers to 2-aminoisobutyric acid; “ALL” refers to acute lymphoblastic leukemia; “aRNA” refers to activating RNA; “ASO” refers to antisense oligonucleotide; “BBB” refers to blood-brain barrier; “BTB” refers to blood-testis barrier; “CLL” refers to chronic lymphocytic leukemia; “CNS” refers to central nervous system; “cTfR” refers to cynomolgus transferrin receptor; “D2O” refers to deuterium; “Da” refers to dalton(s); “DBCO” refers to dibenzocyclooctyne; “DNA” refers to deoxyribonucleic acid; “DRG” refers to dorsal root ganglion; “ds” refers to double-stranded; “DsiRNA” refers to Dicer substrate interfering RNA; “ELISA” refers to enzyme-linked immunosorbent assay; “eNCP” refers to enveloped nanocomplex; “eRNA” refers to editing RNA; “f” refers to inverted phenylalanine; “FTD” refers to frontotemporal dementia; “HD” refers to Huntington’s disease; “hr” refers to hour(s); “Hsm” refers to homoserine methyl ether; “hTfR” refers to human ^
transferrin receptor; “ICM” refers to an intracisterna magna; “ICV” refers to intracerebroventricular; “IPM” refers to intraparenchymal; “iRNA” refers to inhibiting RNA; “IT” refers to intrathecal; “IV” refers to intravenous or intravenously; “kg” refers to kilogram; “LNP” refers to lipid nanoparticle; “mg” refers to milligram; “min” refers to minute(s); refers to messenger RNA; “miRNA” refers to microRNA; “MS” refers to multiple sclerosis; “MSPT” refers to 4-(5-methylsulfonyl-1H-tetrazole-1yl)phenol; “mTfR” refers to murine transferrin receptor; “NC” refers to nanocapsule; “NCR” refers to nanocrystal; “NE” refers to nanoemulsion; “NHL” refers to non-Hodgkin lymphoma; “nm” refers to nanometer(s); “Nle” refers to norleucine; “NP” refers to nanoparticle; “OD” refers to 4-(5- (methylsulfonyl)-1,3,4-oxadiazol-2-yl)phenol; “PD” refers to Parkinson’s disease; “PLV” refers to proteolipid vehicle; “rAAV” refers to recombinant adeno-associated virus; “RISC” refers to RNA-induced silencing complex; “RITA” refers to RNA-induced transcriptional activation; “RNA” refers to ribonucleic acid; “RNA NP” refers to RNA nanoparticle; “rRNA” refers to ribosomal RNA; “SAR” refers to structure-activity relationship; “SC” refers to subcutaneous or subcutaneously; “shRNA” refers to short hairpin RNA; “siRNA” refers to small interfering RNA; “SMCC” refers to succinimidyl-4-(N-maleimidomethyl)cyclohexane- 1-carboxylate; “ss” refers to single-stranded; “sup-tRNA” refers to suppressor transfer RNA; “TBP” refers to transferrin receptor binding protein; “tRNA” refers to transfer RNA; “TfR” refers to transferrin receptor; “Tf” refers to transferrin; “VH” refers to variable heavy chain; and “VL refers to variable light chain. [0046] Certain definitions used herein are defined as follows: [0047] As used herein, “about” means within a statistically meaningful range of a value or values such as, for example, a stated concentration, length, molecular weight, pH, sequence similarity, time frame, temperature, volume, etc. Such a value or range can be within an order of magnitude typically within 20%, more typically within 10%, and even more typically within 5% of a given value or range. The allowable variation encompassed by “about” will depend upon the particular system under study, and can be readily appreciated by one of skill in the art. [0048] As used herein, “activating RNA” or “aRNA” means a nucleic acid that contains RNA and that mediates the targeted activation of a promoter or other non-coding transcript of a RNA transcript via a RNA-induced transcriptional activation (RITA) complex pathway. aRNAs ^
typically are ds. aRNA activates, or upregulates expression of a target nucleotide sequence in a cell. [0049] As used herein, “administer,” “administering,” “administration” and the like mean providing a substance (e.g., an engineered peptide herein, a conjugate herein, a fusion herein, a delivery vehicle herein or a pharmaceutical composition herein) to an individual in a manner that is pharmacologically useful (e.g., to treat a disease, disorder or condition in the individual). [0050] As used herein, “amino acid” means a molecule that, from a chemical standpoint, is characterized by the presence of one or more amine groups and one or more carboxylic acid groups and may contain other functional groups. There is a set of twenty naturally occurring amino acids that are designated as standard amino acids and that are used as building blocks for most of the peptides/proteins produced by any living being. Amino acid, however, also can include non-canonical analogs and derivatives thereof that function in a similar manner as naturally occurring amino acids. [0051] As used herein, “amino acid with a functional group available for conjugation” means any natural or unnatural amino acid with a functional group that may be conjugated to a fatty acid by way of, for example, a linker. Examples of such functional groups include, but are not limited to, alkynyl, alkenyl, amino, azido, bromo, carboxyl, chloro, iodo and thiol groups. Additionally, examples of natural amino acids including such functional groups include C (thiol), D (carboxyl), E (carboxyl), K (amino) and Q (amide). [0052] As used herein, “central nervous system disease, disorder or condition” or “CNS disease, disorder or condition” means a disease, disorder or condition that affects the brain (e.g., frontal lobe, occipital lobe, parietal lobe and/or temporal lobe including regions such as, for example, the amygdala, basal ganglia, Broca’s area, cerebellum, corpus callosum, hypothalamus, medulla oblongata and thalamus), brain stem, glial cells (e.g., astrocytes, ependymal cells, microglia and oligodendrocytes), neurons and/or spinal cord. Examples of CNS diseases, disorders or conditions include, but are not limited to, attention deficit hyperactivity disorder (ADHD), autism, Alzheimer’s disease (AD), cancer, epilepsy, frontotemporal dementia (FTD), Huntington’s disease (HD), infection/inflammation, migraine, multiple sclerosis (MS), Parkinson’s disease (PD), seizure and stroke. [0053] As used herein, “comparison window” means a contiguous and specified segment of a nucleotide sequence or amino acid sequence, where the sequence in the comparison window may include additions and/or deletions (i.e., gaps) compared to a reference sequence (which ^
does not include the additions and/or deletions) for optimal alignment of the two sequences. Generally, the comparison window is at least 5 contiguous nucleotides/amino acids in length, and optionally can be 10, 20, 30, 40, 50, 60, 70, 80, 90 or 100 nucleotides/amino acids, or longer. [0054] As used herein, “conservative substitution,” “conservative replacement” or “conservative mutation,” with regard to an amino acid sequence, mean a change of one amino acid for another amino acid having a similar biochemical property (e.g., charge, hydrophobicity, size, etc.). Exemplary conservative substitutions are shown in Table 1. [0055] Table 1: Exemplary Conservative Amino Acid Substitution(s). Original Very Highly Amino Conserved High Conserved s, s, s, y,
ace of a hydroxyl at the 2' position of its pentose sugar when compared with a ribonucleotide. A modified deoxyribonucleotide has one or more modifications or substitutions of atoms other ^
than hydroxyl at the 2' position, including or substitutions in or of the nucleobase, sugar, or phosphate group. [0057] As used herein, “effective amount” means an amount, concentration or dose of a therapeutic agent, or a pharmaceutical composition thereof, upon single or multiple dose administration to an individual in need thereof, provides a desired effect in such an individual under diagnosis or treatment (i.e., may produce a clinically measurable difference in a condition of the individual). An effective amount can be readily determined by one of skill in the art by using known techniques and by observing results obtained under analogous circumstances. In determining the effective amount for an individual, a number of factors are considered, including, but not limited to, the species of mammal, its size, age and general health, the specific disease, disorder or condition involved, the degree of or involvement or the severity of the disease, disorder or condition, the response of the individual, the therapeutic agent administered, the mode of administration, the bioavailability characteristics of the preparation administered, the dose regimen selected, the use of concomitant medication, and other relevant circumstances. [0058] As used herein, “editing RNA” or “eRNA” means a nucleic acid that contains RNA and that mediates inserting, deleting and even base substituting of nucleotides within a target nucleotide sequence. RNA editing has been observed in a number of different types of RNA such as, for example, mRNA, microRNA (miRNA), transfer RNA (tRNA) and ribosomal RNA (rRNA). RNA editing is enzymatically mediated either by exogenously supplying adenosine deaminase acting on RNA enzyme (ADAR) or by directing an endogenous ADAR to a specific site in a target RNA nucleotide sequence, and typically involves editing at a single nucleotide site by directing the ADAR to that site through complimentary oligonucleotides. eRNAs typically are ss. [0059] As used herein, “engineered,” with respect to an amino acid sequence, means having an altered amino acid sequence obtained from genetic engineering techniques to nucleic acids that encode the protein and cells or organisms that express the protein. Likewise, and with respect to a nucleotide sequence, “engineered” means having an altered nucleotide sequence obtained from genetic engineering techniques. Genetic engineering techniques include, but are not limited to, PCR and DNA cloning technologies; transfection, transformation and other gene transfer technologies; homologous recombination; site-directed mutagenesis; and gene fusion. In this manner, a peptide or protein having an amino acid sequence identical to a naturally ^
occurring protein but produced by expression in a heterologous host, is not considered engineered. Alternatively, “engineered” means modified, especially with respect to a wild-type nucleotide sequence or a wild-type amino acid sequence. For example, “engineered” may refer to a change, such as an addition, deletion and/or substitution of a nucleic acid residue or amino acid residue with respect to a given wild-type nucleotide or amino acid sequence. [0060] As used herein, “enveloped nanocomplex” or “eNCP” means a NP that includes a lipid and therapeutic agent (e.g., a therapeutic oligonucleotide) complexed together by charge, which is further encapsulated by a coating. See, e.g., Niu et al. (2018) J. Contol. Release 276:125-139 and Samaridou et al. (2020) Biomaterials 230:119657. [0061] As used herein, “individual” means any mammal, including cats, dogs, mice, rats and primates, especially humans. Moreover, “subject,” “participant” or “patient” may be used interchangeably with “individual.” [0062] As used herein, “individual in need thereof” means a mammal, such as a human, with a disease, disorder or condition requiring treatment or therapy including, for example, those listed herein. In particular, the preferred individual to be treated is a human. [0063] As used herein, “inhibiting RNA” or “iRNA” means a nucleic acid that contains RNA and that mediates the targeted cleavage of a RNA transcript via RNA interference, for example, through a RNA-induced silencing complex (RISC) pathway. Some iRNAs are ss and other iRNAs are ds and have a sense strand and an antisense strand, where the sense strand and the antisense strand form a duplex. iRNA directs sequence-specific degradation of mRNA via RNA interference. iRNA attenuates, inhibits, modulates or reduces expression of a target nucleotide sequence in a cell. Examples of iRNA include, but are not limited to, an antisense oligonucleotide (ASO), Dicer substrate interfering RNA (DsiRNA), miRNA, short hairpin RNA (shRNA) and small interfering RNA (siRNA). [0064] As used herein, “linker” means a structure used to conjugate one compound (e.g., an engineered peptide herein) to another compound (e.g., another engineered peptide or a therapeutic agent). A linker can be “labile” or “cleavable,” meaning the linker can be cleaved (e.g., by acidic pH or enzyme). Alternatively, a linker can be “stable” or “non-cleavable,” meaning the linker cannot be cleaved in physiological conditions. [0065] As used herein, “lipid nanoparticle” or “LNP” means a NP that includes a solid lipid core having a therapeutic agent (e.g., a therapeutic oligonucleotide) encapsulated within the ^
core, where the lipids can be cationic or lipids, as well as cholesterol and a helper lipid. See, e.g., Hou et al. (2021) Nat. Rev. Mater. 6:1078-1094; Mashima & Takada (2022) Curr. Issues Mol. Biol.44:5013-5027; and Tenchov et al. (2021) ACS Nano.11:16982-17015. [0066] As used herein, “liposome” means a NP that includes a closed lipid bilayer having a therapeutic agent (e.g., a therapeutic oligonucleotide) encapsulated therein. See, e.g., Hou et al. (2021), supra; Tenchov et al. (2021), supra; and Xue et al. (2015) Curr. Pharm. Des.21:3140- 3147. [0067] As used herein, “nanomedicine” means a material having a size from about 1 nm to about 100 nm for treating a disease, disorder or condition. Examples of nanomedicines include, but are not limited to, liposomes, LNPs, eNCP, NCs, NCRs, NEs, PLVs, vectors (especially viral vectors), RNA NPs and recombinant viruses. [0068] As used herein, “nanocapsule” or “NC” means a NP that includes a liquid lipid core and a therapeutic agent (e.g., a therapeutic oligonucleotide) on an outside membrane of the core, which is further encapsulated by a coating. See, e.g., Jiang et al. (2018) J. Control Release 283:235-240; Kim et al. (2021) ACS Nano.15:4576-4593; and Yan et al. (2012) J. Am. Chem. Soc.134:13542-13545. [0069] As used herein, “nanocrystal” or “NCR” means a NP that includes a crystalized therapeutic agent (e.g., a therapeutic oligonucleotide) and a polymer or stabilizer. See, e.g., Jahangir et al. (2020) J. Pharm. Innov. 17:237-248; Pardhi et al. (2018) Curr. Pharm. Des. 24:5129-5146. [0070] As used herein, “nanoemulsion” or “NE” means a NP that includes a liquid lipid core having a therapeutic agent (e.g., a therapeutic oligonucleotide) on an outside membrane of the core. See, e.g., Brito et al. (2014) Mol. Ther. 22:2118-2129; Chircov & Grumezescu, “Nanoemulsion Preparation, Characterization, and Application in the Field of Biomedicine, Nanoarchitectonics in Biomedicine, 169-188 (Grumezescu ed., 2019). [0071] As used herein, “nanoparticle” or “NP” means a natural or synthetic material or a combination thereof having lengths in two or three dimension and having a diameter between about 1 nm to about 100 nm that can be used to transport, for example, a therapeutic agent. NPs typically possess unique physical and chemical properties due to their high surface area and nanoscale size and typically include three layers: (1) a surface layer that may be functionalized, (2) a shell layer and (3) a core that is chemically distinct from the shell layer. ^
Examples of NPs for use herein include, but not limited to, a dendrimer, a liposome, a LNP, an eNCP, a NC, a NCR, a NE, a PLV and a RNA NP. [0072] As used herein, “nucleic acid” means a polymer of nucleotides. Although it may comprise any type of nucleotide units, the term generally applies to nucleotide polymers of DNA, cDNA and/or RNA. Polynucleotide is used to include single-stranded (ss) nucleic acids, double-stranded (ds) nucleic acids, and DNA, cDNA and/or RNA made from nucleotide or nucleoside analogues that may be identified by their sequences, which are generally presented in the 5' to 3' direction (as the coding strand), where the 5' and 3' indicate the linkages formed between the 5' hydroxyl group of one nucleotide and the 3'-hydroxyl group of the next nucleotide. For a coding strand presented in the 5'-3' direction, its complement (or non-coding strand) is the strand that hybridizes to that sequence according to Watson-Crick base pairing. Thus, as used herein, the complement of a nucleic acid such as a polynucleotide is the same as the “reverse complement” and describes the nucleic acid that in its natural form, would be based paired with the nucleic acid in question. [0073] As used herein, “nucleoside” means a nucleobase-sugar combination, where the nucleobase portion is normally a heterocyclic base. The two most common classes of such heterocyclic bases are purines and pyrimidines. The sugar is normally a pentose sugar such as a ribose or a deoxyribose (e.g., 2'-deoxyribose). [0074] As used herein, “nucleotide” means an organic molecule having a nucleoside (a nucleobase such as, for example, adenine, cytosine, guanine, thymine or uracil; and a pentose sugar such as, e.g., ribose or 2'-deoxyribose) and a phosphate group, which can serve as a monomeric unit of nucleic acid polymers such as deoxyribonucleic acid (DNA) and ribonucleic acid (RNA). [0075] As used herein, “oligonucleotide” means a short nucleic acid compound (e.g., a polymer of less than about 100 nucleotides in length) that may include deoxyribonucleotides (or modified deoxyribonucleotides), ribonucleotides (or modified ribonucleotides) or both. Likewise, an oligonucleotide may be ss or ds and thus may or may not have duplex regions. [0076] As used herein, “pharmaceutically acceptable,” when referring to a material such as a carrier or diluent, means that it does not abrogate the biological activity or properties of a therapeutic agent and is relatively non-toxic (i.e., the material may be administered to an individual without causing undesirable biological effects or interacting in a deleterious manner with any of the components of the composition in which it is contained. ^
[0077] As used herein, acceptable carrier” means a pharmaceutically acceptable material, composition or carrier, such as a liquid or solid filler, stabilizer, dispersing agent, suspending agent, diluent, excipient, thickening agent, solvent or encapsulating material, involved in carrying or transporting a therapeutic agent within or to an individual such that it may perform its intended function. Additional ingredients that may be included in the pharmaceutical compositions used in the practice of the invention are known in the art and described, for example in Remington’s Pharmaceutical Sciences, 21st Edition, University of the Sciences in Philadelphia, PA (2006). [0078] As used herein, “pharmaceutical composition” means a composition or therapeutic agent, mixed with at least one pharmaceutically acceptable chemical component, such as, but not limited to carriers, stabilizers, diluents, dispersing agents, suspending agents, thickening agents, excipients and the like. [0079] As used herein, “proteolipid vehicle” or “PLV” means a NP that includes a combination of proteins, such as fusion-associated small transmembrane (FAST) proteins, and neutral or ionizable lipids having a therapeutic agent (e.g., a therapeutic oligonucleotide) encapsulated therein. See, e.g., Brown et al. (2022), “Safe and Effective Delivery of Nucleic Acids Using Proteolipid Vehicles Formulated with Fusion-Associated Small Transmembrane Proteins” (under review); and Raturi et al. (2022), “Rapid Prototyping and Immunogenicity of SARS-CoV-2 DNA Vaccine Candidates Formulated with the Fusion-Associated Small Transmembrane Protein Proteolipid Vehicle Delivery System,” (under review); see also, Intl. Patent Application Publication No. WO 2022/067446. [0080] As used herein, “recombinant adeno-associated virus,” “recombinant AAV” and “rAAV” mean viral particles comprising a rAAV vector encapsidated by AAV capsid protein. [0081] As used herein, “recombinant adeno-associated virus vector,” “recombinant AAV vector” and “rAAV vector” mean an engineered polynucleotide vector comprising one or more heterologous sequences (i.e., nucleic acid sequence not of an AAV origin) that are flanked by at least one AAV ITR sequence. Such rAAV vectors can be replicated and packaged into infectious viral particles when present in a host cell that has been infected with a suitable helper virus (or that is expressing suitable helper functions) that expresses AAV rep and cap gene products (i.e., AAV Rep and Cap proteins). [0082] As used herein, “ribonucleotide” means a nucleotide having a ribose as its pentose sugar, which contains a hydroxyl group at its 2' position. A modified ribonucleotide is a ^
ribonucleotide having one or more or substitutions of atoms other than hydrogen at the 2' position, including modifications or substitutions in or of the nucleobase, sugar, or phosphate group. [0083] As used herein, “RNA nanoparticle” or “RNA NP” means a NP that includes a plurality of oligonucleotide oligomers and a therapeutic agent (e.g., a therapeutic oligonucleotide). See, e.g., Guo et al. (2020) Nat. Comm. 11:972; Jasinski et al. (2017) ACS Nano. 11:1142-1164; Pi et al. (2018) Nat. Nanotechnol. 13:89-89; and Shu et al. (2015) ACS Nano 9:9731-9740. [0084] As used herein, “sequence identity,” in the context of two nucleotide sequences or two amino acid sequences, means that residues in the two sequences that are the same when aligned for maximum correspondence over a specified comparison window. [0085] As used herein, “transferrin receptor-associated disease, disorder or condition” or “TfR-associated disease, disorder or condition” means a disease, disorder or condition in which TfR is an etiology thereof, especially TfR1 upregulation and/or dysregulation. Examples of TfR-associated diseases, disorders and/or conditions include, but are not limited to, anemia, cancer (e.g., solid cancers such as adrenal cortical carcinoma, bladder cancer, breast cancer, cervical cancer, cholangiocarcinoma, colon cancer, esophageal squamous cell carcinoma, hepatocellular carcinoma, liver cancer, lung cancer, osteosarcoma, ovarian cancer, pancreatic cancer, renal cell carcinoma, and cancers of the nervous system, as well as hematopoietic malignancies such as acute lymphoblastic leukemia (ALL), chronic lymphocytic leukemia (CLL) and non-Hodgkin lymphoma (NHL) and neurodegenerative disease. [0086] As used herein, “therapeutic oligonucleotide” means a ss or ds nucleic acid that has a therapeutic application (i.e., application in diagnosing and/or treating a disease, disorder or condition). Such a nucleic acid typically contains one or more modified nucleotide residues or linkages and also can include a targeting ligand and/or delivery vehicle. Examples of therapeutic oligonucleotides include, but are not limited to, aRNA, eRNA, iRNA, mRNA and tRNA. Specific examples of therapeutic oligonucleotides include, but are not limited to, ASOs, aptamers, mRNAs, miRNAs, short activating RNAs (saRNAs), siRNAs, suppressor tRNAs (sup-tRNAs) and decoys. [0087] As used herein, “treat,” “to treat,” “treatment” or “treating” means an act of providing care to an individual in need thereof, for example, by administering a therapeutic agent to the individual for purposes of improving the health and/or well-being of the individual with respect ^
to an existing a disease, disorder or or to prevent or decrease the likelihood of the occurrence of a disease, disorder or condition. Treating also can involve reducing the frequency or severity of at least one sign, symptom or contributing factor of a disease, disorder or condition experienced by the individual. [0088] As used herein, “vector” means a nucleic acid molecule capable of transporting another nucleic acid sequence (or multiple nucleic acid sequences) to which it has been ligated into a host cell or genome. One type of vector is a “plasmid,” which refers to a circular DNA loop, typically double-stranded (ds), into which additional DNA segments may be ligated. Another type of vector is a viral vector, wherein additional DNA segments may be ligated into the viral genome. Certain vectors are capable of autonomous replication in a host cell into which they are introduced (e.g., bacterial vectors having a bacterial origin of replication). Moreover, certain vectors are capable of directing the expression of genes (e.g., genes encoding an exogenous peptide or protein of interest) to which they are operatively linked when combined with appropriate control sequences such as promoter and operator sequences and replication initiation sites. Such vectors are commonly referred to as “expression vectors” and may also include a multiple cloning site for insertion of the gene encoding the protein of interest. Alternatively, the gene encoding the peptide or protein of interest may be introduced by site-directed mutagenesis techniques such as Kunkel mutagenesis. See, e.g., Handa et al., Rapid and Reliable Site-Directed Mutagenesis Using Kunkel’s Approach, Methods in Molecular Biology, vol 182: In Vitro: Mutagenesis Protocols, 2nd Ed.). [0089] As used herein, “viral vector” means a vector that is derived from a naturally occurring or modified virus, especially a rAAV vector or a Baculovirus vector (e.g., Autographa californica nuclear polyhedrosis (AcNPV) vector)). [0090] Compositions [0091] Engineered Peptides: [0092] The engineered peptides herein can bind to the TfR and can have from about 14 amino acids to about 20 amino acids, about 15 to about 19 amino acids, about 16 to about 18 amino acids or about 17 amino acids. In other instances, the engineered peptides can have about 14 amino acids, about 15 amino acids, about 16 amino acids, about 17 amino acids, about 18 amino acids, about 19 amino acids or about 20 amino acids. The engineered peptides can be ^
used to deliver, for example, a to a cell, tissue, organ or system of an individual. [0093] The amino acid sequences of incretin analogs described herein incorporate naturally occurring amino acids, typically depicted herein using standard one letter codes (e.g., L = leucine), as well as variants thereof (e.g., inverted Phe (f), homoserine methyl ether (Hsm) and norleucine (Nle)), and certain other unnatural amino acids, such as 2-aminoisobutyric acid (Aib). The structures of these amino acids are depicted in Table 2 below. [0094] Table 2: Exemplary Amino Acid Structures. f
at least about 80%, about 81%, about 82%, about 83%, about 84%, about 85%, about 86%, about 87%, about 88%, about 89%, about 90%, about 91%, about 92%, about 93%, about 94%, about 95%, about 96%, about 97%, about 98%, about 99% or about 100% sequence identity to SEQ ID NO:3. In other instances, the amino acid sequence can have at least about 95% sequence identity to SEQ ID NO:3. In certain instances, the amino acid sequence is SEQ ID NO:3. [0096] Another such engineered peptide includes an amino acid sequence having at least about 80% sequence identity to SEQ ID NO:4. In some instances, the amino acid sequence can have at least 80%, about 81%, about 82%, about 83%, about 84%, about 85%, about 86%, about 87%, about 88%, about 89%, about 90%, about 91%, about 92%, about 93%, about 94%, about 95%, about 96%, about 97%, about 98%, about 99% or about 100% sequence identity to SEQ ID NO:4. In other instances, the amino acid sequence can have at least about 95% sequence identity to SEQ ID NO:4. In certain instances, the amino acid sequence is SEQ ID NO:4. [0097] Another engineered peptide includes an amino acid sequence having at least about 80% sequence identity to SEQ ID NO:5. In some instances, the amino acid sequence can have at least about 80%, about 81%, about 82%, about 83%, about 84%, about 85%, about 86%, ^
about 87%, about 88%, about 89%, about about 91%, about 92%, about 93%, about 94%, about 95%, about 96%, about 97%, about 98%, about 99% or about 100% sequence identity to SEQ ID NO:5. In other instances, the amino acid sequence can have at least about 95% sequence identity to SEQ ID NO:5. In certain instances, the amino acid sequence is SEQ ID NO:5. [0098] Another engineered peptide includes an amino acid sequence having at least about 80% sequence identity to SEQ ID NO:6. In some instances, the amino acid sequence can have at least about 80%, about 81%, about 82%, about 83%, about 84%, about 85%, about 86%, about 87%, about 88%, about 89%, about 90%, about 91%, about 92%, about 93%, about 94%, about 95%, about 96%, about 97%, about 98%, about 99% or about 100% sequence identity to SEQ ID NO:6. In other instances, the amino acid sequence can have at least about 95% sequence identity to SEQ ID NO:6. In certain instances, the amino acid sequence is SEQ ID NO:6. [0099] Another engineered peptide includes an amino acid sequence having at least about 80% sequence identity to SEQ ID NO:7. In some instances, the amino acid sequence can have at least about 80%, about 81%, about 82%, about 83%, about 84%, about 85%, about 86%, about 87%, about 88%, about 89%, about 90%, about 91%, about 92%, about 93%, about 94%, about 95%, about 96%, about 97%, about 98%, about 99% or about 100% sequence identity to SEQ ID NO:7. In other instances, the amino acid sequence can have at least about 95% sequence identity to SEQ ID NO:7. In certain instances, the amino acid sequence is SEQ ID NO:7. [00100] Another engineered peptide includes an amino acid sequence having at least about 80% sequence identity to SEQ ID NO:8. In some instances, the amino acid sequence can have at least about 80%, about 81%, about 82%, about 83%, about 84%, about 85%, about 86%, about 87%, about 88%, about 89%, about 90%, about 91%, about 92%, about 93%, about 94%, about 95%, about 96%, about 97%, about 98%, about 99% or about 100% sequence identity to SEQ ID NO:8. In other instances, the amino acid sequence can have at least about 95% sequence identity to SEQ ID NO:8. In certain instances, the amino acid sequence is SEQ ID NO:8. [00101] Another engineered peptide includes an amino acid sequence having at least about 80% sequence identity to SEQ ID NO:9. In some instances, the amino acid sequence can have at least about 80%, about 81%, about 82%, about 83%, about 84%, about 85%, about 86%, ^
about 87%, about 88%, about 89%, about about 91%, about 92%, about 93%, about 94%, about 95%, about 96%, about 97%, about 98%, about 99% or about 100% sequence identity to SEQ ID NO:9. In other instances, the amino acid sequence can have at least about 95% sequence identity to SEQ ID NO:9. In certain instances, the amino acid sequence is SEQ ID NO:9. [00102] Another engineered peptide includes an amino acid sequence having at least about 80% sequence identity to SEQ ID NO:10. In some instances, the amino acid sequence can have at least about 80%, about 81%, about 82%, about 83%, about 84%, about 85%, about 86%, about 87%, about 88%, about 89%, about 90%, about 91%, about 92%, about 93%, about 94%, about 95%, about 96%, about 97%, about 98%, about 99% or about 100% sequence identity to SEQ ID NO:10. In other instances, the amino acid sequence can have at least about 95% sequence identity to SEQ ID NO:10. In certain instances, the amino acid sequence is SEQ ID NO:10. [00103] Another engineered peptide includes an amino acid sequence having at least about 80% sequence identity to SEQ ID NO:11. In some instances, the amino acid sequence can be at least about 80%, about 81%, about 82%, about 83%, about 84%, about 85%, about 86%, about 87%, about 88%, about 89%, about 90%, about 91%, about 92%, about 93%, about 94%, about 95%, about 96%, about 97%, about 98%, about 99% or about 100% sequence identity to SEQ ID NO:11. In other instances, the amino acid sequence can have at least about 95% sequence identity to SEQ ID NO:11. In certain instances, the amino acid sequence is SEQ ID NO:11. [00104] Another engineered peptide includes an amino acid sequence having at least about 80% sequence identity to SEQ ID NO:12. In some instances, the amino acid sequence can have at least about 80%, about 81%, about 82%, about 83%, about 84%, about 85%, about 86%, about 87%, about 88%, about 89%, about 90%, about 91%, about 92%, about 93%, about 94%, about 95%, about 96%, about 97%, about 98%, about 99% or about 100% sequence identity to SEQ ID NO:12. In other instances, the amino acid sequence can have at least about 95% sequence identity to SEQ ID NO:12. In certain instances, the amino acid sequence is SEQ ID NO:12. [00105] Another engineered peptide includes an amino acid sequence having at least about 80% sequence identity to SEQ ID NO:13. In some instances, the amino acid sequence can have at least about 80%, about 81%, about 82%, about 83%, about 84%, about 85%, about 86%, ^
about 87%, about 88%, about 89%, about about 91%, about 92%, about 93%, about 94%, about 95%, about 96%, about 97%, about 98%, about 99% or about 100% sequence identity to SEQ ID NO:13. In other instances, the amino acid sequence can have at least about 95% sequence identity to SEQ ID NO:13. In certain instances, the amino acid sequence is SEQ ID NO:13. [00106] Another engineered peptide includes an amino acid sequence having at least about 80% sequence identity to SEQ ID NO:14. In some instances, the amino acid sequence can have at least about 80%, about 81%, about 82%, about 83%, about 84%, about 85%, about 86%, about 87%, about 88%, about 89%, about 90%, about 91%, about 92%, about 93%, about 94%, about 95%, about 96%, about 97%, about 98%, about 99% or about 100% sequence identity to SEQ ID NO:14. In other instances, the amino acid sequence can have at least about 95% sequence identity to SEQ ID NO:14. In certain instances, the amino acid sequence is SEQ ID NO:14. [00107] Another engineered peptide includes an amino acid sequence having at least about 80% sequence identity to SEQ ID NO:15. In some instances, the amino acid sequence can have at least about 80%, about 81%, about 82%, about 83%, about 84%, about 85%, about 86%, about 87%, about 88%, about 89%, about 90%, about 91%, about 92%, about 93%, about 94%, about 95%, about 96%, about 97%, about 98%, about 99% or about 100% sequence identity to SEQ ID NO:15. In other instances, the amino acid sequence can have at least about 95% sequence identity to SEQ ID NO:15. In certain instances, the amino acid sequence is SEQ ID NO:15. [00108] Another engineered peptide includes an amino acid sequence having at least about 80% sequence identity to SEQ ID NO:16. In some instances, the amino acid sequence can have at least about 80%, about 81%, about 82%, about 83%, about 84%, about 85%, about 86%, about 87%, about 88%, about 89%, about 90%, about 91%, about 92%, about 93%, about 94%, about 95%, about 96%, about 97%, about 98%, about 99% or about 100% sequence identity to SEQ ID NO:16. In other instances, the amino acid sequence can have at least about 95% sequence identity to SEQ ID NO:16. In certain instances, the amino acid sequence is SEQ ID NO:16. [00109] Another engineered peptide includes an amino acid sequence having at least about 80% sequence identity to SEQ ID NO:18. In some instances, the amino acid sequence can have at least about 80%, about 81%, about 82%, about 83%, about 84%, about 85%, about 86%, ^
about 87%, about 88%, about 89%, about about 91%, about 92%, about 93%, about 94%, about 95%, about 96%, about 97%, about 98%, about 99% or about 100% sequence identity to SEQ ID NO:18. In other instances, the amino acid sequence can have at least about 95% sequence identity to SEQ ID NO:18. In certain instances, the amino acid sequence is SEQ ID NO:18. [00110] Another engineered peptide includes an amino acid sequence having at least about 80% sequence identity to SEQ ID NO:29. In some instances, the amino acid sequence can have at least about 80%, about 81%, about 82%, about 83%, about 84%, about 85%, about 86%, about 87%, about 88%, about 89%, about 90%, about 91%, about 92%, about 93%, about 94%, about 95%, about 96%, about 97%, about 98%, about 99% or about 100% sequence identity to SEQ ID NO:29. In other instances, the amino acid sequence can have at least about 95% sequence identity to SEQ ID NO:29. In certain instances, the amino acid sequence is SEQ ID NO:29. [00111] Another engineered peptide includes an amino acid sequence having at least about 80% sequence identity to SEQ ID NO:30. In some instances, the amino acid sequence can have at least about 80%, about 81%, about 82%, about 83%, about 84%, about 85%, about 86%, about 87%, about 88%, about 89%, about 90%, about 91%, about 92%, about 93%, about 94%, about 95%, about 96%, about 97%, about 98%, about 99% or about 100% sequence identity to SEQ ID NO:30. In other instances, the amino acid sequence can have at least about 95% sequence identity to SEQ ID NO:30. In certain instances, the amino acid sequence is SEQ ID NO:30. [00112] Another engineered peptide includes an amino acid sequence having at least about 80% sequence identity to SEQ ID NO:31. In some instances, the amino acid sequence can have at least about 80%, about 81%, about 82%, about 83%, about 84%, about 85%, about 86%, about 87%, about 88%, about 89%, about 90%, about 91%, about 92%, about 93%, about 94%, about 95%, about 96%, about 97%, about 98%, about 99% or about 100% sequence identity to SEQ ID NO:31. In other instances, the amino acid sequence can have at least about 95% sequence identity to SEQ ID NO:31. In certain instances, the amino acid sequence is SEQ ID NO:31. [00113] Another engineered peptide includes an amino acid sequence having at least about 80% sequence identity to SEQ ID NO:32. In some instances, the amino acid sequence can have at least about 80%, about 81%, about 82%, about 83%, about 84%, about 85%, about 86%, ^
about 87%, about 88%, about 89%, about about 91%, about 92%, about 93%, about 94%, about 95%, about 96%, about 97%, about 98%, about 99% or about 100% sequence identity to SEQ ID NO:32. In other instances, the amino acid sequence can have at least about 95% sequence identity to SEQ ID NO:32. In certain instances, the amino acid sequence can be SEQ ID NO:32. [00114] Another engineered peptide includes an amino acid sequence having at least about 80% sequence identity to SEQ ID NO:33. In some instances, the amino acid sequence can have at least about 80%, about 81%, about 82%, about 83%, about 84%, about 85%, about 86%, about 87%, about 88%, about 89%, about 90%, about 91%, about 92%, about 93%, about 94%, about 95%, about 96%, about 97%, about 98%, about 99% or about 100% sequence identity to SEQ ID NO:33. In other instances, the amino acid sequence can have at least about 95% sequence identity to SEQ ID NO:33. In certain instances, the amino acid sequence can be SEQ ID NO:33. [00115] Another engineered peptide includes an amino acid sequence having at least about 80% sequence identity to SEQ ID NO:34. In some instances, the amino acid sequence can have at least about 80%, about 81%, about 82%, about 83%, about 84%, about 85%, about 86%, about 87%, about 88%, about 89%, about 90%, about 91%, about 92%, about 93%, about 94%, about 95%, about 96%, about 97%, about 98%, about 99% or about 100% sequence identity to SEQ ID NO:34. In other instances, the amino acid sequence can have at least about 95% sequence identity to SEQ ID NO:34. In certain instances, the amino acid sequence can be SEQ ID NO:34. [00116] Another engineered peptide includes an amino acid sequence having at least about 80% sequence identity to SEQ ID NO:35. In some instances, the amino acid sequence can have at least about 80%, about 81%, about 82%, about 83%, about 84%, about 85%, about 86%, about 87%, about 88%, about 89%, about 90%, about 91%, about 92%, about 93%, about 94%, about 95%, about 96%, about 97%, about 98%, about 99% or about 100% sequence identity to SEQ ID NO:35. In other instances, the amino acid sequence can have at least about 95% sequence identity to SEQ ID NO:35. In certain instances, the amino acid sequence can be SEQ ID NO:35. [00117] Another engineered peptide includes an amino acid sequence having at least about 80% sequence identity to SEQ ID NO:44. In some instances, the amino acid sequence can have at least about 80%, about 81%, about 82%, about 83%, about 84%, about 85%, about 86%, ^
about 87%, about 88%, about 89%, about about 91%, about 92%, about 93%, about 94%, about 95%, about 96%, about 97%, about 98%, about 99% or about 100% sequence identity to SEQ ID NO:44. In other instances, the amino acid sequence can have at least about 95% sequence identity to SEQ ID NO:44. In certain instances, the amino acid sequence can be SEQ ID NO:44. [00118] Another engineered peptide includes an amino acid sequence having at least about 80% sequence identity to SEQ ID NO:45. In some instances, the amino acid sequence can have at least about 80%, about 81%, about 82%, about 83%, about 84%, about 85%, about 86%, about 87%, about 88%, about 89%, about 90%, about 91%, about 92%, about 93%, about 94%, about 95%, about 96%, about 97%, about 98%, about 99% or about 100% sequence identity to SEQ ID NO:45. In other instances, the amino acid sequence can have at least about 95% sequence identity to SEQ ID NO:45. In certain instances, the amino acid sequence can be SEQ ID NO:45. [00119] Another engineered peptide includes an amino acid sequence having at least about 80% sequence identity to SEQ ID NO:46. In some instances, the amino acid sequence can have at least about 80%, about 81%, about 82%, about 83%, about 84%, about 85%, about 86%, about 87%, about 88%, about 89%, about 90%, about 91%, about 92%, about 93%, about 94%, about 95%, about 96%, about 97%, about 98%, about 99% or about 100% sequence identity to SEQ ID NO:46. In other instances, the amino acid sequence can have at least about 95% sequence identity to SEQ ID NO:46. In certain instances, the amino acid sequence can be SEQ ID NO:46. [00120] Alternatively, the engineered peptide includes an amino acid sequence of: X1X2X3CRX6FYPX10X11YCX14AX16 (Formula I; SEQ ID NO:17), wherein X1 can be any naturally occurring amino acid or a variant thereof, especially an aromatic amino acid, wherein X2 can be any naturally occurring amino acid or a variant thereof, especially an aromatic amino acid, wherein X3 can be any naturally occurring amino acid or a variant thereof, especially an acidic amino acid, wherein X6 can be any naturally occurring amino acid or a variant thereof, especially an alpha-methylated amino acid, wherein X10 can be any naturally occurring amino acid or a variant thereof, especially an aromatic amino acid, wherein X11 can be any naturally occurring amino acid or a variant thereof, especially an acidic amino acid, wherein X14 can be any naturally occurring amino acid or a variant thereof, ^
especially an acidic amino acid, wherein can be any naturally occurring amino acid or a variant thereof, especially an aliphatic amino acid. [00121] In some instances, X1 can be F, I, L, M, W, Y or a conservative substitution thereof. In some instances, X2 can be F, I, L, M, W, Y or a conservative substitution thereof. In some instances, X3 can be D, E, S, Q or a conservative substitution thereof. In some instances, X6 can be S, Aib or a conservative substitution thereof. In some instances, X10 can be f, F, I, L, M, W, Y or a conservative substitution thereof. In some instances, X11 can be D, E, S, Q or a conservative substitution thereof. In some instances, X14 can be D, E, H, K, N, R, Q, S or a conservative substitution thereof. In some instances, X16 can be M, Nle, Hsm or a conservative substitution thereof. In other instances, X1 can be F, I, L, M, W, Y or a conservative substitution thereof; X2 can be F, I, L, M, W, Y or a conservative substitution thereof; X3 can be D, E, S, Q or a conservative substitution thereof; X6 can be S, Aib or a conservative substitution thereof; X10 can be f, F, I, L, M, W, Y or a conservative substitution thereof; X11 can be D, E, S, Q or a conservative substitution thereof; X14 can be D, E, H, K, N, R, Q , S or a conservative substitution thereof; and X16 can be M, Nle, Hsm or a conservative substitution thereof. [00122] In certain instances, X1 is F or a conservative substitution thereof. In certain instances, X2 is W or a conservative substitution thereof. In certain instances, X3 is D or a conservative substitution thereof. In certain instances, X6 is S or a conservative substitution thereof. In certain instances, X10 is F or a conservative substitution thereof. In certain instances, X11 is D or a conservative substitution thereof. In certain instances, X14 is E or a conservative substitution thereof. In certain instances, X16 is M or a conservative substitution thereof. In other certain instances, X1 is F or a conservative substitution thereof, X2 is W or a conservative substitution thereof, X3 is D or a conservative substitution thereof, X6 is S or a conservative substitution thereof, X10 is F or a conservative substitution thereof, X11 is D or a conservative substitution thereof, X14 is E or a conservative substitution thereof, and X16 is M or a conservative substitution thereof (SEQ ID NO:18). In particular instances, X1 is F, X2 is W, X3 is D, X6 is S, X10 is F, X11 is D, X14 is E and X16 is M (SEQ ID NO:18). [00123] In certain instances, X1 is F or a conservative substitution thereof. In certain instances, X2 is W or a conservative substitution thereof. In certain instances, X3 is D or a conservative substitution thereof. In certain instances, X6 is Aib. In certain instances, X10 is f. In certain instances, X11 is D or a conservative substitution thereof. In certain instances, X14 is E or a conservative substitution thereof. In certain instances, X16 is M or a conservative substitution ^
thereof. In other certain instances, X1 is F conservative substitution thereof, X2 is W or a conservative substitution thereof, X3 is D or a conservative substitution thereof, X6 is Aib, X10 is f, X11 is D or a conservative substitution thereof, X14 is E or a conservative substitution thereof, and X16 is M or a conservative substitution thereof (SEQ ID NO:44). In particular instances, X1 is F, X2 is W, X3 is D, X6 is Aib, X10 is f, X11 is D, X14 is E and X16 is M (SEQ ID NO:44). [00124] In certain instances, X1 is F or a conservative substitution thereof. In certain instances, X2 is W or a conservative substitution thereof. In certain instances, X3 is D or a conservative substitution thereof. In certain instances, X6 is Aib. In certain instances, X10 is f. In certain instances, X11 is D or a conservative substitution thereof. In certain instances, X14 is E or a conservative substitution thereof. In certain instances, X16 is Nle. In other certain instances, X1 is F or a conservative substitution thereof, X2 is W or a conservative substitution thereof, X3 is D or a conservative substitution thereof, X6 is Aib, X10 is f, X11 is D or a conservative substitution thereof, X14 is E or a conservative substitution thereof, and X16 is Nle (SEQ ID NO:45). In particular instances, X1 is F, X2 is W, X3 is D, X6 is Aib, X10 is f, X11 is D, X14 is E and X16 is Nle (SEQ ID NO:45). [00125] In certain instances, X1 is F or a conservative substitution thereof. In certain instances, X2 is W or a conservative substitution thereof. In certain instances, X3 is D or a conservative substitution thereof. In certain instances, X6 is Aib. In certain instances, X10 is f. In certain instances, X11 is D or a conservative substitution thereof. In certain instances, X14 is E or a conservative substitution thereof. In certain instances, X16 is Hsm. In other certain instances, X1 is F or a conservative substitution thereof, X2 is W or a conservative substitution thereof, X3 is D or a conservative substitution thereof, X6 is Aib, X10 is f, X11 is D or a conservative substitution thereof, X14 is E or a conservative substitution thereof, and X16 is Hsm (SEQ ID NO:46). In particular instances, X1 is F, X2 is W, X3 is D, X6 is Aib, X10 is f, X11 is D, X14 is E and X16 is Hsm (SEQ ID NO:46). [00126] While the above engineered peptides are described as monomers, in some instances they can be in the form of a multimer including, but not limited to, a dimer, trimer, tetramer, pentamer, hexamer, etc. In some instances, the multimer can include the same monomer (e.g., as in a homodimer, homotrimer, etc.). In other instances, the multimer can include distinct monomers (e.g., as in a heterodimer, heterotrimer, etc.). ^
[00127] In some instances, the monomers multimer can be associated with one another via a covalent bond, a hydrogen bond and/or an ionic bond. In other instances, the monomers of the multimer can be associated with one another via a linker. [00128] In some instances, the engineered peptides can include a disulfide bond between C residues, such as those at positions 4 and 13. In other instances, the engineered peptides can include a C-terminal amide. [00129] Conjugates and Fusions: [00130] The engineered peptides herein can be used to deliver a therapeutic agent to a cell, tissue, organ or system of an individual. In this manner, an engineered peptide herein can be conjugated to the therapeutic agent (i.e., a conjugate where the parts are linked to one another via one or more linkers). Alternatively, an engineered peptide herein can be fused to the therapeutic agent (i.e., a fusion where the parts are directly linked to one another). [00131] In some instances, the engineered peptide includes an amino acid sequence having at least about 90% sequence identity to any one of SEQ ID NOS:3 to 16, 18, 29 to 35 and 44 to 46. In other instances, the amino acid sequence can have at least about 90%, about 91%, about 92%, about 93%, about 94%, about 95%, about 96%, about 97%, about 98%, about 99% or about 100% sequence identity to any one of SEQ ID NOS:3 to 16, 18, 29 to 35 and 44 to 46. In yet other instances, the amino acid sequence can have at least about 95% sequence identity to any one of SEQ ID NOS:3 to 16, 18, 29 to 35 and 44 to 46. In certain instances, the amino acid sequence can be any one of SEQ ID NO:3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 18, 29, 30, 31, 32, 33, 34, 35, 44, 45 or 46. [00132] As above, in some instances, the engineered peptide can be a monomer. In other instances, the engineered peptide can be in the form of a multimer including, but not limited to, dimers, trimers, tetramers, pentamers, hexamers, etc. Moreover, the multimer can include the same monomer (e.g., a homodimer) or can include distinct monomers (e.g., a heterodimer). [00133] In some instances of the conjugates and fusions, the therapeutic agent can be an oligonucleotide, another peptide or protein (including antibodies or fragments thereof) or a small molecule. [00134] When the therapeutic agent is an oligonucleotide, it can be an aRNA agent, an eRNA agent, an iRNA agent, a mRNA agent or a tRNA agent. Examples of such oligonucleotides ^
include, but are not limited to, ASOs, mRNAs, miRNAs, short activating RNAs (saRNAs), siRNAs, sup-tRNA and decoys. [00135] In some instances when the therapeutic agent is an oligonucleotide, the engineered peptide can be at a 3'-end, both a 5'-end, a 3'-end and a 5'-end, and/or an internal nucleotide. [00136] When the therapeutic agent is another peptide or a protein (i.e., distinct from the engineered peptide herein), it can be an Ab or fragment thereof, an anti-infective agent, an anti- rejection agent, an antioxidant, a chemotherapeutic, a growth factor, a hormone, a neurotransmitter, a receptor agonist or antagonist, a toxin, etc. [00137] In some instances when the therapeutic agent is peptide or protein, the engineered peptide can be at an N-terminal end of the peptide or protein, a C-terminal end of the peptide or protein, both a N-terminal end and a C-terminal end of the peptide or protein, and/or an internal amino acid. [00138] When the therapeutic agent is a small molecule (i.e., an organic compound of about ≤ 1000 Da), it can be a small molecule that intervenes with a biological function and can be, for example, a chemotherapeutic or a toxin. [00139] Alternatively, and in other instances and regardless of the type of therapeutic agent, the therapeutic agent can be conjugated or fused to an N-terminal end of the engineered peptide. In other instances, the therapeutic agent can be conjugated or fused to a C-terminal end of the engineered peptide. In yet other instances, one therapeutic agent can be conjugated or fused to the N-terminal end and an identical therapeutic agent can be conjugated to the C-terminal end. In yet other instances, one therapeutic agent can be conjugated or fused to the N-terminal end and a distinct therapeutic agent can be conjugated to the C-terminal end. [00140] In some instances, the therapeutic agent can be conjugated to an internal amino acid of the engineered peptide with a functional group available for conjugation (e.g., a side chain of the amino acid. Examples of amino acids with a functional group available for conjugation include, but are not limited to C (thiol), D (carboxyl), E (carboxyl), K (amino) and Q (amide). [00141] In some instances, the therapeutic agent (i.e., “R”) can be linked to the engineered peptide (i.e., “TBP”) via a linker. Exemplary linkers include, but are not limited to, the linkers depicted in Table 3 below. [00142] Table 3: Exemplary TBP and Therapeutic Agent Linkers. Linker Structure ^
1 linker
^
7 “DBCO-azide” linker
[00143] Delivery Vehicles ^
[00144] The engineered peptides herein incorporated into a delivery vehicle to deliver a therapeutic agent to a cell, tissue, organ or system of an individual. In this manner, an engineered peptide herein can be incorporated into, for example, a nanomedicine such as a NP (e.g., a dendrimer, liposome, LNP, eNCP, NC, NCR, NE, PLV or RNA NP). Alternatively, an engineered peptide herein can be incorporated into a nanomedicine such as, for example, a capsid of a recombinant virus such as a rAAV. [00145] In some instances, the engineered peptide includes an amino acid sequence having at least about 90% sequence identity to any one of SEQ ID NOS:3 to 16, 18, 29 to 35 and 44 to 46. In other instances, the amino acid sequence can have at least about 90%, about 91%, about 92%, about 93%, about 94%, about 95%, about 96%, about 97%, about 98%, about 99% or about 100% sequence identity to any one of SEQ ID NOS:3 to 16, 18, 29 to 35 and 44 to 46. In yet other instances, the amino acid sequence can have at least about 95% sequence identity to any one of SEQ ID NOS:3 to 16, 18, 29 to 35 and 44 to 46. In certain instances, the amino acid sequence is any one of SEQ ID NO:3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 18, 29, 30, 31, 32, 33, 34, 35, 44, 45 or 46. [00146] When the delivery vehicle is a NP, it can be a dendrimer, an eNCP, a liposome, a LNP, a NC, a NCR, a NE, a PLV or a RNA NP. [00147] When the delivery vehicle is a recombinant virus, it can be a rAAV. AAV capsid protein serotypes include, but are not limited to, AAV1, AAV2, AAV3, AAV4, AAV5, AAV6, AAV7, AAV8 and AAV9 or variants thereof. See, e.g., Intl. Patent Application Publication No. WO 2005/033321. [00148] Pharmaceutical Compositions: [00149] The conjugates herein, fusions herein and delivery vehicles herein can be formulated as pharmaceutical compositions that include the conjugate, fusion or delivery vehicle and a pharmaceutically acceptable carrier. Such pharmaceutical compositions and techniques for preparing the same are well known in the art. See, e.g., Remington, “The Science and Practice of Pharmacy” (D.B. Troy ed., 21st Edition, Lippincott, Williams & Wilkins, 2006). [00150] In addition, and to improve their in vivo compatibility and effectiveness, the conjugates, fusions or delivery vehicles may be reacted with any of a number of inorganic and organic acids/bases to form pharmaceutically acceptable acid/base addition salts. Pharmaceutically acceptable salts and common methodologies for preparing them are well ^
known in the art (see, e.g., Stahl et al., of Pharmaceutical Salts: Properties, Selection and Use,” 2nd Revised Edition (Wiley-VCH, 2011)). Pharmaceutically acceptable salts for use herein include sodium, trifluoroacetate, hydrochloride and acetate salts. [00151] In some instances, the pharmaceutical compositions further include an excipient, which can confer to a composition improved stability, improved absorption, improved solubility and/or therapeutic enhancement of the active ingredient. In some instances, the excipient can be a buffering agent (e.g., sodium citrate, sodium phosphate, a tris base or sodium hydroxide) or a vehicle (e.g., a buffered solution, petrolatum, dimethyl sulfoxide or mineral oil). In some instances, the conjugates, fusions or delivery vehicles can be lyophilized for extending shelf-life and then made into a solution before use (e.g., administration to an individual). Accordingly, the excipient in the pharmaceutical compositions including the conjugates, fusions or delivery vehicles can be a lyoprotectant (e.g., mannitol, lactose, polyethylene glycol, or polyvinylpyrrolidone) or a collapse temperature modifier (e.g., dextran, Ficoll™ or gelatin). [00152] In some instances, the pharmaceutical compositions can include one or more additional therapeutic agents. [00153] The pharmaceutical composition can be administered by any route, including enteral (e.g., oral), parenteral, intravenous, intramuscular, intra-arterial, intramedullary, intrathecal, subcutaneous, intraventricular, transdermal, interdermal, rectal, intravaginal, intraperitoneal, topical (as by powders, ointments, creams and/or drops), mucosal, nasal, buccal, sublingual; by intratracheal instillation, bronchial instillation and/or inhalation; and/or as an oral spray, nasal spray, and/or aerosol. Specifically contemplated routes are oral administration, IV injection (e.g., systemic intravenous injection), SC injection, ICV injection, ICM injection, IPM injection, IT injection or a combination thereof. [00154] Generally, the most appropriate route of administration will depend upon a variety of factors including, but not limited to, the nature of the agent (e.g., its stability in the environment of its administration and/or intended target) and/or the condition of the individual (e.g., whether the subject is able to tolerate oral administration). In some instances, the engineered nucleic acids, rAAV or pharmaceutical compositions are suitable for administration to, for example, the CNS of an individual. [00155] Kits: ^
[00156] In some instances, an engineered herein, conjugate herein, fusion herein, delivery vehicle herein and/or pharmaceutical composition herein can be included in a kit that includes the same and instructions for its use. In other instances, the kit includes an engineered peptide, conjugate, fusion, delivery vehicle and/or pharmaceutical composition and a package insert containing instructions for use of the kit and/or any component thereof. In yet other instances, the kit comprises, in a suitable container or other means for containing, the engineered peptide, conjugate, fusion, delivery vehicle and/or pharmaceutical composition, one or more controls, and various buffers, reagents, enzymes and other standard ingredients well known in the art. In some instances, the container comprises at least one vial, well, test tube, flask, bottle, syringe or other container means, into which the engineered peptide, conjugate, fusion, delivery vehicle and/or pharmaceutical composition is placed, and in some instances, suitably aliquoted. In those instances where an additional component is provided, the kit includes additional containers into which this component is placed. The kits also can include a means for containing the engineered peptide, conjugate, fusion, delivery vehicle and/or pharmaceutical composition and any other reagent in close confinement for commercial sale. Such containers may include injection or blow-molded plastic containers into which the desired vials are retained. Containers and/or kits can include labeling with instructions for use and/or warnings. [00157] Methods [00158] Methods of Making Engineered Peptides: [00159] The engineered peptides herein can be synthesized via any number of peptide synthesis methods known in the art using standard manual or automated solid-phase synthesis procedures. Automated peptide synthesizers are commercially available from, for example, Applied Biosystems (Foster City, CA) and Protein Technologies Inc. (Tucson, AZ). Reagents for solid-phase synthesis are readily available from commercial sources. Solid-phase synthesizers can be used according to the manufacturer’s instructions for blocking interfering groups, protecting amino acids during reaction, coupling, deprotecting and capping of unreacted amino acids. The peptides can also be expressed recombinantly, or by using in vitro transcription translation machinery, or by using a combination of the said methods. [00160] Typically, an N-α-carbamoyl-protected amino acid and the N-terminal amino acid on the growing peptide chain attached to a resin are coupled at room temperature in an inert solvent ^
such as DMF, N-methylpyrrolidone or chloride in the presence of coupling agents such as diisopropy1-carbodiimide and 1-hydroxybenzotriazole. The N-α-carbamoyl protecting group is removed from the resulting peptide resin using a reagent such TFA or piperidine, and the coupling reaction is repeated with the next desired N-α-protected amino acid to be added to the peptide chain. Suitable amine protecting groups are well known in the art and are described, for example, in Green & Wuts, “Protecting Groups in Organic Synthesis,” (John Wiley and Sons, 1991). The most commonly used examples include tBoc and Fmoc. After completion of synthesis, peptides are cleaved from the solid-phase support with simultaneous side chain deprotection using standard treatment methods under acidic conditions. [00161] One of skill in the art will appreciate that the peptide chains described herein are synthesized with a C-terminal carboxamide. For the synthesis of C-terminal amide peptides, resins incorporating Rink amide MBHA or Rink amide AM linkers typically are used with Fmoc synthesis, while MBHA resin is generally used with tBoc synthesis. [00162] Crude peptides typically are purified using RP-HPLC on C8 or C18 columns using water-acetonitrile gradients in 0.05 % to 0.1 % TFA. Purity can be verified by analytical RP- HPLC. Identity of peptides can be verified by mass spectrometry. Peptides can be solubilized in aqueous buffers over a wide pH range. [00163] Methods of Transporting Therapeutic Agents Across the BBB [00164] The methods below can include the steps described herein, and these maybe be, but not necessarily, carried out in the sequence as described. Other sequences, however, also are conceivable. Moreover, individual or multiple steps bay be carried out either in parallel and/or overlapping in time and/or individually or in multiply repeated steps. Furthermore, the methods may include additional, unspecified steps. [00165] The conjugates herein, fusions herein, delivery vehicles herein or pharmaceutical compositions herein can be used for transporting a therapeutic agent across the BBB. The methods can include at least a step of administering to the individual an effective amount of a conjugate, a fusion, a delivery vehicle and/or a pharmaceutical composition herein. In some instances, the administering can be via direct (i.e., central) injection into the CNS of the individual, which can be an ICV injection, an ICM injection, an IPM injection, an IT injection or a combination thereof. In other instances, the administering can be via a peripheral injection. In certain instances, the peripheral injection can be via IV injection or SC injection. In yet other ^
instances, the administering can be by a of direct injection and peripheral injection. [00166] It is further contemplated that the methods can include a step of administering an effective amount of one or more additional therapeutic agents other than those discussed above. [00167] Methods of Treating Individuals [00168] The methods below can include the steps described herein, and these may be, but not necessarily, carried out in the sequence as described. Other sequences, however, also are conceivable. Moreover, individual or multiple steps may be carried out either in parallel and/or overlapping in time and/or individually or in multiply repeated steps. Furthermore, the methods may include additional, unspecified steps. [00169] The conjugates herein, fusion proteins herein, delivery vehicles herein or pharmaceutical compositions herein can be used for treating individuals having a CNS disease, disorder or condition. The methods can include at least a step of administering to the individual an effective amount of a conjugate, a fusion, a delivery vehicle and/or a pharmaceutical composition. In some instances, the administering can be via direct injection into the CNS of the individual, which can be an ICV injection, an ICM injection, an IPM injection, an IT injection or a combination thereof. In other instances, the administering can be via a peripheral injection. In certain instances, the peripheral injection can be via IV injection or SC injection. In yet other instances, the administering can be by a combination of direct injection and peripheral injection. [00170] It is further contemplated that the methods can include a step of administering an effective amount of one or more additional therapeutic agents other than those discussed above. [00171] Alternatively, the conjugates herein, fusion proteins herein, delivery vehicles herein or pharmaceutical compositions herein can be used for treating individuals having a TfR- associated disease, disorder or condition. The methods can include at least a step of administering to the individual an effective amount of a conjugate, a fusion, a delivery vehicle and/or a pharmaceutical composition. In some instances, the administering can be via direct injection into the CNS of the individual, which can be an ICV injection, an ICM injection, an IPM injection, an IT injection or a combination thereof. In other instances, the administering can be via a peripheral injection. In certain instances, the peripheral injection can be via IV ^
injection or SC injection. In yet other the administering can be by a combination of direct injection and peripheral injection. [00172] It is further contemplated that the methods can include a step of administering an effective amount of one or more additional therapeutic agents other than those discussed above. [00173] Uses: [00174] A conjugate herein, fusion herein, delivery vehicle herein and/or pharmaceutical composition herein can be used, or adapted for use, to transport a therapeutic agent across the BBB of an individual. As such, the conjugate, fusion, delivery vehicle and/or pharmaceutical composition is provided for use, or adapted for use, to transport a therapeutic agent across the BBB. Also, the conjugate, fusion, delivery vehicle and/or pharmaceutical composition is provided for use, or adaptable for use, in the manufacture of a medicament or a pharmaceutical composition for transporting a therapeutic agent across the BBB. [00175] Likewise, a conjugate herein, fusion herein, delivery vehicle herein and/or pharmaceutical composition herein can be used, or adapted for use, to treat an individual (e.g., a human) having a CNS disease, disorder or condition. As such, the conjugate, fusion, delivery vehicle and/or pharmaceutical composition is provided for use, or adapted for use, to treat an individual having a CNS disease, disorder or condition. Also, the conjugate, fusion, delivery vehicle and/or pharmaceutical composition is provided for use, or adaptable for use, in the manufacture of a medicament or a pharmaceutical composition for treating a CNS disease, disorder or condition. [00176] Moreover, a conjugate herein, fusion herein, delivery vehicle herein and/or pharmaceutical composition herein can be used, or adapted for use, to treat an individual (e.g., a human) having a TfR-associated disease, disorder or condition. As such, the conjugate, fusion, delivery vehicle and/or pharmaceutical composition is provided for use, or adapted for use, to treat an individual having a TfR-associated disease, disorder or condition. Also, the conjugate, fusion, delivery vehicle and/or pharmaceutical composition is provided for use, or adaptable for use, in the manufacture of a medicament or a pharmaceutical composition for treating a TfR-associated disease, disorder or condition. [00177] Also described are conjugates, fusions, delivery vehicles and/or pharmaceutical compositions for use in therapy. Furthermore, conjugates, fusions, delivery vehicles and/or pharmaceutical compositions are described herein for use in transporting a therapeutic agent ^
across the BBB or in the treatment of a disorder or condition or in the treatment of a TfR-associated disease, disorder or condition. [00178] Also described are use of conjugates, fusions, delivery vehicles and/or pharmaceutical compositions in the manufacture of a medicament for transporting a therapeutic agent across the BBB or for the treatment of a CNS disease, disorder or condition or for the treatment of a TfR-associated disease, disorder or condition. EXAMPLES [00179] The following non-limiting examples are offered for purposes of illustration, not limitation. [00180] GENERATING ENGINEERED TRANSFERRIN RECEPTOR BINDING PEPTIDES [00181] Example 1: Engineering TfR Binding Peptides [00182] Methods: [00183] TfR binding peptides using phage libraries: Two peptide libraries – 282 and 585 – were selected against the hTfR extracellular domain (SEQ ID NO:2). These peptide libraries are 14 and 20 residues long, respectively, and contain 8 random amino acids within 2 engineered Cys residues. The libraries differ by having either 2 (282 library) or 5 (585 library) random sequences flanking each engineered Cys residue. [00184] Both libraries were constructed using a backbone phage vector as described previously (Chang et al. (2020) Protein Eng. Des. Sel. 33:1-8). Three rounds of selection against 100 nM biotinylated hTfR were completed, and the eluted phage from final round was screened in filter-lift assay against hTfR (SEQ ID NO:1). Positive hits were sequenced and further evaluated in single point and titer dependent ELISAs as previously described (Ting et al. (2018) PLoS One 13:e0190850). [00185] Binding of the selected peptides on phage to hTfR-expressing cells was confirmed using hCMEC/D3- and hTfR-expressing CHO cell lines by flow cytometry. A family of peptides were identified (see, Table 4). Phage displaying these peptides, such as Peptide 9, internalized in hCMEC/D3 cells; whereas naked phage that lacked any peptide did not show binding at 4ºC or internalizing at 37ºC (see, FIGS.1A and 1B). A known TfR Ab was used as positive control that showed significant level of internalization (MEM189; see, Sade et al. ^
(2014) PLoS One 9:e96340). Anti-phage DL650 was used in both the binding and internalizing assays (commercially available from abcam; Waltham, MA). [00186] Table 4: Phage Library and Peptide Sequences. Library Amino Acid Sequence SEQ ID NO: Diversity 282 X2CX8CX2 (14 aa total) 1.0 x 109
[00187] Three peptides, Peptides 8, 9 and 10, were selected for affinity optimization. [00188] Optimization of selected hits: First, a minimum peptide pharmacophore was determined as described in Ting et al. (2018), supra. Second, each amino acid of the pharmacophore was mutated to Ala (A) and binding to hTfR was assessed. [00189] This information then was used to design and construct a focused library on phage. Hotspot residues that drove binding of the peptide to the target were left untouched and the remaining residues were either subjected to full or soft randomization as described in Ting et al. (2018), supra. [00190] The focused library was selected against recombinant hTfR under stringent conditions that favored selection of peptides with slower off-rate as described in Ting et al. (2018), supra. In this selection, the library was incubated for a brief period with 10 nM biotinylated hTfR (bio-TfR). This was followed by adding an excess amount of unlabeled hTfR to release the phage from the bio-TfR at a rate that was proportional to the peptide dissociation rate. As a ^
result, peptides with fast off-rate dissociate bio-hTfR and bind to unlabeled TfR present in excess amount. In contrast, peptides with slower off-rate remain bound to the bio-hTfR. Subsequent pull-down using streptavidin-coated beads separated peptides with slower off-rate from the mix. To identify peptides that internalize and deliver their cargos inside the cell, binding of peptides on phage to hCMEC/D3 cells at RT and 37ºC was compared. [00191] Results: A family of peptides on phage was identified that binds to hTfR, Caco2 and hCMEC/D3 cells stronger than the parent and efficiently internalized in the hCMEC/D3 cell line. Multiple representatives of this series, including Peptide 15, were chemically synthesized for further evaluation. [00192] IN VITRO FUNCTION [00193] Example 2: Characterizing Peptide 15 [00194] Methods: [00195] Binding assay: Binding of Peptide 15 to recombinant hTfr and cTfR was determined by surface plasmon resonance (SPR). Flow cytometry also was used to assess interaction of Peptide 15 to hTfR- and cTfR-expressing cell lines. [00196] Control peptide development: In a systemic study, binding of Peptide 15 variants with or without disulfide bond linkage to hTfR and cTfR was assessed using SPR. A non- disulfide-linked peptide with Ala mutations at residues S6, F7, Y8 and P9, called Peptide 17 (SEQ ID NO:20), was used as a control peptide. [00197] Epitope mapping: Hydrogen-D2O Exchange-Mass Spectrometry (HDX-MS) was used to map the binding sites of Peptide 15 to hTfR as described in Ting et al. (2018), supra. [00198] Results: Binding of Peptide 15 to recombinant hTfr and cTfR was determined by surface plasmon resonance (SPR) to be 25 nM and 160 nM, respectively. This translated into more than 46-fold and 19-fold increase, respectively, in binding over the parent peptide, Peptide 8 (see, Table 5). [00199] With regard to the binding assay, Peptide 15 bound hTfR-expressing cells (i.e., EFO- 21) with EC50 of 17 nM and to cTfR-expressing cells (i.e., MDCK-c6) at EC50 of 730 nM. Parental peptide, Peptide 8 (SEQ ID NO:10), showed specific but weak binding to either of the cell lines. Moreover, Peptide 15 did not bind to murine TfR (mTfR)-expressing cells or recombinant protein. ^
[00200] Table 5: Peptide 8 and Peptide well as Peptide 15 Variants, Binding to hTfR and cTfR. hTfR cTfR Peptide Amino Acid Sequence ka kd KD ka kd KD 6 6 1 7 D D D D D D D D D D vs.
Peptide 15; NBD = no binding detected. [00201] With regard to control peptide development, disulfide bond breakage or mutation of Y8A and P9A in a non-disulfide linked peptide abrogated binding of Peptide 15 to TfR. Internalization of the biotinylated Peptides 8 and 15 (as well as a truncated Peptide 8, which lacks 2 resides at both the amino- and carboxy-termini) into engineered EFO-21 and MDCK cell lines was evaluated by high content microscopy using Operetta (see, FIGS. 2A-B). Streptavidin-pHast conjugate was used to detect internalization of the peptide. Peptide 15 showed strong internalization in EFO-21 cells; whereas no internalization was observed with Peptide 19 and streptavidin-pHast conjugates (see, FIG.3). [00202] With regard to the epitope mapping, Peptide 15 interacted with hTfR at sites distant from Tf binding, including regions 152-166 (i.e., YVPREAGSQKDENLA; SEQ ID NO:36), ^
189-200 (i.e., KIQVKDSAQNSV; SEQ ID and 376-383 (i.e., TVSNVLKE; SEQ ID NO:38) of, for example, SEQ ID NO:1. Minor binding sites or structural changes due to binding were at regions encompassing 213-221 (i.e., VENPGGYVA; SEQ ID NO:39), 285- 290 (i.e., QTKFPI; SEQ ID NO:40) and 338-344 (i.e., SRAAAEK; SEQ ID NO:41) of, for example, SEQ ID NO:1. TfR sequence coverage in the presence of Peptide 15 is shown in FIGS.4A and 4B. [00203] Example 3: Peptide-Directed siRNA Targeting In Vitro [00204] Methods: [00205] siRNA conjugates: Peptides 15 and 17 were conjugated to siRNA targeting synuclein (SNCA; SEQ ID NO:42) or hypoxanthine-guanine phosphoribosyltransferase (HPRT; SEQ ID NO:43) with phosphorothioate (PS) linkage(s) at 3' end and a vinylphosphonate (VP) cap at 5' of the sense and anti-sense strands, respectively. A PEG12 linker and DBCO click chemistry was used to prepare the conjugates. For all the conjugates prepared, more than 60% yield and 94% purity were obtained. In general, the sense strand was conjugated to the peptide C- terminus via azide click chemistry. A cholesterol-conjugated control siRNA also was synthesized. [00206] Knockdown assay: The siRNA conjugates were tested for gene knockdown in the EFO-21 cell line. [00207] Plasma stability: The siRNA conjugates were tested for plasma stability in human and cynomolgus plasma for 24 hr. [00208] Results: With regard to SNCA, Peptide 15-conjugated siRNA targeting SNCA remained fully intact in the presence of human and non-human (i.e., cynomolgus) plasma for up to 24 hr. In addition, Peptide 15-conjugated siRNA showed comparable knockdown efficacy to cholesterol-conjugated siRNA with nearly 3-fold stronger IC50 at 9 nM. Moreover, Peptide 17-conjugated siRNA did not induce any gene silencing, indicating that the observed activity was mediated via interaction of Peptide 15 with TfR (see, FIG. 5A). In contrast, Peptide 17- conjugated siRNA did not induce any SNCA gene silencing (see also, FIG.5A). [00209] With regard to HPRT, robust delivery of Peptide 15-conjugated siRNA and HPRT gene silencing was observed in EFO-21 cells (IC50 = 6 nM) (see, FIG.5B). No knockdown was observed with Peptide 17, suggesting that siRNA delivery was mediated by specific interaction of Peptide 15 with TfR expressed on EFO-21 cells. ^
[00210] With regard to plasma stability, 15- and Peptide 17-conjugated siRNA targeting SNCA remained fully intact in the presence of human and cynomolgus plasma for up to 24 hr (see, FIG.6). [00211] Example 4: Peptide-Directed Antibody Targeting In Vitro and In Vivo [00212] Methods: [00213] Fusion proteins: Fusion proteins were generated by cloning Peptide 15 (SEQ ID NO:18) at the C-terminus of either heavy (VH), light (VL) or both (VH + VL) chains of an IgG1. The fusion proteins were evaluated for binding to recombinant and cell-expressed TfR- 1 using SPR (as described above) or flow cytometry, respectively. [00214] Flow cytometry: The EFO-21 cell line was used to evaluate binding of the fusion proteins to hTfR-1-expressing cells. First, EFO-21 cells were grown to confluency prior to detachment from plates using TrypleE. Cell viability and count was next determined by a Vi- CELL™ cell and particle counter. Cells were transferred to 96-well plates at density of 5 x 105 cells per well, washed with FACS buffer (1% FBS/PBS, 0.1% NaN3, plus 2 mM EDTA) and stained with LIVE/DEAD dye for 30 min while on ice. Subsequently, cells were washed two times with FACS buffer and then incubated with Ab-peptide fusions for 1 hr on ice. Cells were washed again two times with FACS buffer and incubated with anti-human IgG PE for 1 hr on ice. Finally, cells were washed three times with FACS buffer and processed on a BD™ LSR II Benchtop Flow Cytometer (Becton Dickinson) to evaluate binding of the fusion proteins and the parental IgG1 to EFO-21 cells. Data is an average of 4 separate experiments. [00215] Biodistribution via confocal microscopy: AZDye 647 was conjugated to fusion proteins using a site-specific conjugation technology. Briefly, glycans were removed by EndoS followed by adding azidosugar. AZDye 647-DBCO was conjugated with the azidosugar on the Ab via a click chemistry reaction. Fluorescently labeled fusion proteins were injected intravenously at 10 mg/kg to humanized-TfR transgenic mice. 48 hr post dose, mice were euthanized, and brain and gastrocnemius were isolated. Subsequently, isolated brain and gastrocnemius were imaged using an inverted LSM 980 Confocal Microscope (Carl Zeiss). [00216] Biodistribution via biochemical analysis: CNS exposure of fusion proteins was evaluated in humanized-TfR transgenic mice by administering a single 5 mg/kg IV injection of either a mAb fused with a single TfR peptide (2:1) or two TfR binding peptides (2:2) fused to the C-terminal of VH. CNS exposures in multiple compartments were measured at 6, 24, 96 ^
and 192 hr post-dose (N=3 per treatment and . Brains were isolated and homogenized in RIPA buffer to generate brain extracts. Brain extracts, plasma and CSF levels of fusion proteins were determined by enzyme-linked immunoassay (ELISA). Compound levels in the CNS extracts are expressed as pg/mg of tissue wet weight. [00217] Results: With regard to binding of the fusion protein to Tfr, Table 6 shows binding of the fusion protein (Peptide 15 fused to IgG1 at the specified site) to hTfR and cTfR by SPR. Naked IgG1 was used as negative control and did not show any binding by SPR. Additionally, Table 7 shows binding of various fusion proteins (Peptide 15 fused to IgG1 at the specified site) to hTfR in EFO-21 cells, which is a human ovarian tumor cell line with high hTfR expression by flow cytometry. Again, naked IgG1 was used as negative control and did not show any binding to EFO-21 by flow cytometry. [00218] Table 6: Fusion Protein Binding to hTfR and cTfR Using SPR. hTfR1 (ECD) cTfR (ECD) Ka Kd KD Ka Kd K -8 -7 -8
[00219] Table 7: Fusion Protein Binding to hTfR and cTfR Using Flow Cytometry. EC50 Pe tide (M)
[00220] With regard to biodistribution, Ab-Peptide 15 fusions successfully crossed the BBB following IV infusion (see, FIGS. 7A and 7B). Moreover, Ab-Peptide 15 fusions resulted in remarkably increased exposure in both the brain tissue, CSF and plasma/CSF ratio, thereby ^
demonstrating Peptide 15 is capable of a therapeutic agent such as an Ab across the BBB (see, Table 8). [00221] Table 8: Biodistribution of Fusion Protein to hTfR Using Biochemistry. G
[00223] Example 5: Peptide-Directed siRNA Targeting In Vivo [00224] Methods: [00225] Animal model: hTfR transgenic mice were injected 1 mg/kg or 10 mg/kg of Peptide 15-conjugated siRNA or Peptide 19-conjugated siRNA (fluorophore labeled) targeting HPRT that were made according to Example 3. Images were taken at 1 hr or 24 hr post injection. Live images were taken from liver, gastric and adipose tissue, and ex vivo images were taken from hypothalamus and DRG neurons. [00226] The siRNA conjugates were intravenously (IV) dosed in mice at 2.5 mg/kg, 5.0 mg/kg and 10 mg/kg. After 7 days, muscle was isolated and HPRT gene expression was assessed by qPCR. In addition, 2.5 mg/kg and 10.0 mg/kg doses of Peptide 15-conjugated siRNA were tested via SC administration. [00227] Results: A robust knockdown was observed with Peptide 15-conjugated siRNA targeting HPRT as compared to Peptide 19-conjugated siRNA targeting HPRT at all doses administered (see, FIG.8). A dose response was not observed with Peptide 15-siRNA targeting HPRT in the dose-range tested (see, FIG.8). [00228] Knockdown levels with SC administration of Peptide 15-conjugated siRNA targeting HPRT was comparable with IV dosing at the same dose, indicating that SC dosing is feasible (see, FIG.8). ^
[00229] Example 6: Peptide-Directed Targeting In Vitro [00230] Methods: [00231] TfR binding peptide-Ab fusions: Peptide 15 was fused to N- or C-termini of heavy, light or both chains of an IgG1 Ab. The fusion proteins were tested for binding to TfR and the Ab target in a TrR-transgenic mouse model. Select fusion proteins were injected intravenously in a hTfR-transgenic mice, and biodistribution was determined using confocal microscopy. [00232] Results: C-terminus (C-term) fusion proteins bound to TfR with affinities comparable to free peptide; whereas N-terminal (N-term) fusion proteins resulted in improved affinity due to avidity (see, Table 9). Interestingly, the fusion protein with C-term fusion of Peptide 15 bound to N3pE42 peptide (antigen) at comparable levels to the parental Ab. [00233] Table 9: Peptide 15-Ab Fusion Affinity Data. hTfR-ECD cTfR-ECD P i L h i K K KD K K KD
[00234] With regard to the mouse model, strong localization was observed in muscle and adipose tissue, DRG neurons, hypothalamus and pancreatic beta cells, suggesting that Peptide 15 localization follows the expression pattern of TfR (see, FIGS.9A and 9B). [00235] GENERATING ADDITIONAL ENGINEERED TRANSFERRIN RECEPTOR BINDING PEPTIDES [00236] Example 7: Improving Potency of TfR Binding Peptide 15 ^
[00237] Methods: [00238] Structure-activity relationship (SAR): Peptide 15 was modified at individual or multiple residues via traditional solid-phase peptide synthesis, followed by HPLC purification. The modifications included, but were not limited to, natural and unnatural amino acids, alpha- methylated amino acids, D-amino acids and beta-amino acids. Additionally, truncations, extensions and acylation of N- and C-termini also were investigated. The engineered peptides were tested by SPR for binding to hTfR and cTfR, as described above. [00239] Table 10: Peptide 15 and Peptide 15 Variant Sequences. Peptide Amino Acid Sequence SEQ ID NO: 15 FWDCRSFYPFDYCEAM 18
[00240] Results: Truncation of either the N- or C-terminus beyond the first residue led to a significant loss in affinity. Extension of either terminus with additional amino acids was tolerated without significant change in binding affinity. Likewise, addition of a residue at position 17 to facilitate conjugation to a payload did not lead to loss of receptor binding (see, Tables 11-12). [00241] In contrast, improvements in binding affinity were observed with changes that reduced conformational flexibility (e.g., methylation, Aib) rather than with specific side chain mutations. Moreover, mutating S at position 6 to Aib (S6Aib) also gave unprecedented relative improvement in binding to cTfR receptor. Likewise, mutating F at position 10 to an inversion of stereocenter (F10f) led to significant improvement in hTfR binding affinity (see, Table 12). [00242] To create negative controls, enantiomeric versions of peptides 15, 33, 34 and 35 were synthesized and were confirmed to lack measurable binding to either human or cTfR (see, Table 12). ^
6 1 M8 . 4 1 . * 1 5 . 2 - - 8 . 5
51 A- - 7 . 0 0 . 2 - - 1 . 6
1 Y2 2 . > 7 1 > 1 2 > 11 D0 . 9 4 . 7 1 . 1 6 . 9 1 . 3 3 01 F 1 . 3 3 1 2 . 1 . 0 . 7 1 . 2 2 9 P . 9 0 1 0 0 2 0 6 . 9 - - > 2 > 7 1 . s t 0 n 8 Y0 1 . 0 0 0 0 0 0 i 2 2 a r > 7 1 2 > 2 > 2 > a V 5 0 0 0 0 1 7 F 0 e 2 0 2 0 2 0 0 0 d > > > 2 > 2 > it p e 0 P 6 S . 6 1 . 0 0 0 2 2 . 0 0 > 2 2 d > n a 5 1 5 R 1 9 8 . 2 . 6 . 5 . . 7 e 2 1 2 0 0 di 1 t p e P - - 0 f 4 C - - 0 o 2 1 . 4 . > 1 1 1 y r . a 6 5 0 9 5 1 m 3 D. 5 . 7 . . 1 . e m 4 1 2 di u t S p e R A 2 W6 . 8 . 9 . - 7 . P . S 1 1 4 8 - 0 s v : e 1 1 g e 8 l 1 F . 3 7 5 6 n a b 0 . 1 . 2 . 1 . 0 h a 2 c T d l n e o ] 3 o i u o f 4 ti 2 s di i s t e : 0 o e a l b i n e a n M- M- E T 0 [ P R A A e α N O N ^
[00244] Table 12: Peptide 15, as well as 15 Variants, Binding to hTfR and cTfR. Peptide SEQ ID NO: hTfR h/T83 h/M16Nle h/M16Hsm cTfr (nM) (nM) 0
^
LISTING [00245] The following nucleotide and/or amino acid sequences are referred to in the disclosure above and are provided below for reference. [00246] SEQ ID NO:1 – human TfR1 (760 aa) MMDQARSAFSNLFGGEPLSYTRFSLARQVDGDNSHVEMKLAVDEEENADNNTKAN VTKPKRCSGSICYGTIAVIVFFLIGFMIGYLGYCKGVEPKTECERLAGTESPVREEPGE DFPAARRLYWDDLKRKLSEKLDSTDFTGTIKLLNENSYVPREAGSQKDENLALYVE NQFREFKLSKVWRDQHFVKIQVKDSAQNSVIIVDKNGRLVYLVENPGGYVAYSKAA TVTGKLVHANFGTKKDFEDLYTPVNGSIVIVRAGKITFAEKVANAESLNAIGVLIYM DQTKFPIVNAELSFFGHAHLGTGDPYTPGFPSFNHTQFPPSRSSGLPNIPVQTISRAAAE KLFGNMEGDCPSDWKTDSTCRMVTSESKNVKLTVSNVLKEIKILNIFGVIKGFVEPD HYVVVGAQRDAWGPGAAKSGVGTALLLKLAQMFSDMVLKDGFQPSRSIIFASWSA GDFGSVGATEWLEGYLSSLHLKAFTYINLDKAVLGTSNFKVSASPLLYTLIEKTMQN VKHPVTGQFLYQDSNWASKVEKLTLDNAAFPFLAYSGIPAVSFCFCEDTDYPYLGTT MDTYKELIERIPELNKVARAAAEVAGQFVIKLTHDVELNLDYERYNSQLLSFVRDLN QYRADIKEMGLSLQWLYSARGDFFRATSRLTTDFGNAEKTDRFVMKKLNDRVMRV EYHFLSPYVSPKESPFRHVFWGSGSHTLPALLENLKLRKQNNGAFNETLFRNQLALA TWTIQGAANALSGDVWDIDNEF [00247] SEQ ID NO:2 – human TfR1 extracellular domain (672 aa) CKGVEPKTECERLAGTESPVREEPGEDFPAARRLYWDDLKRKLSEKLDSTDFTGTIK LLNENSYVPREAGSQKDENLALYVENQFREFKLSKVWRDQHFVKIQVKDSAQNSVII VDKNGRLVYLVENPGGYVAYSKAATVTGKLVHANFGTKKDFEDLYTPVNGSIVIVR AGKITFAEKVANAESLNAIGVLIYMDQTKFPIVNAELSFFGHAHLGTGDPYTPGFPSF NHTQFPPSRSSGLPNIPVQTISRAAAEKLFGNMEGDCPSDWKTDSTCRMVTSESKNV KLTVSNVLKEIKILNIFGVIKGFVEPDHYVVVGAQRDAWGPGAAKSGVGTALLLKLA QMFSDMVLKDGFQPSRSIIFASWSAGDFGSVGATEWLEGYLSSLHLKAFTYINLDKA VLGTSNFKVSASPLLYTLIEKTMQNVKHPVTGQFLYQDSNWASKVEKLTLDNAAFPF LAYSGIPAVSFCFCEDTDYPYLGTTMDTYKELIERIPELNKVARAAAEVAGQFVIKLT HDVELNLDYERYNSQLLSFVRDLNQYRADIKEMGLSLQWLYSARGDFFRATSRLTT ^
NLKLRKQNNGAFNETLFRNQLALATWTIQGAANALSGDVWDIDNEF [00248] SEQ ID NO:3 – Artificial Sequence 1 (TfR Binding Peptide 1; 14 aa) RLCVPWMYPMSCTT [00249] SEQ ID NO:4 – Artificial Sequence 2 (TfR Binding Peptide 2; 14 aa) STCLQYYPFDYCQP [00250] SEQ ID NO:5 – Artificial Sequence 3 (TfR Binding Peptide 3; 14 aa) WQCDRYYPAEYCQP [00251] SEQ ID NO:6 – Artificial Sequence 4 (TfR Binding Peptide 4; 14 aa) NECYSFYWLIFCTT [00252] SEQ ID NO:7 – Artificial Sequence 5 (TfR Binding Peptide 5; 14 aa) STCLPYYPFDYCQP [00253] SEQ ID NO:8 – Artificial Sequence 6 (TfR Binding Peptide 6; 14 aa) STCLQYYPDDNCQR [00254] SEQ ID NO:9 – Artificial Sequence 7 (TfR Binding Peptide 7; 14 aa) WQCDRYYPAEYCVR [00255] SEQ ID NO:10 – Artificial Sequence 8 (TfR Binding Peptide 8; 20 aa) QSWEACRSFYPQDYCVAMKN [00256] SEQ ID NO:11 – Artificial Sequence 9 (TfR Binding Peptide 9; 20 aa) WTYMDCAMYYPQEYCQQIIA [00257] SEQ ID NO:12 – Artificial Sequence 10 (TfR Binding Peptide 10; 20 aa) QPTTDCLSFYPKTYCVHIQA ^
[00258] SEQ ID NO:13 – Artificial Sequence 11 (TfR Binding Peptide 11; 20 aa) DSDMECLQFYPKTFCTDINT [00259] SEQ ID NO:14 – Artificial Sequence 12 (TfR Binding Peptide 12; 20 aa) QPTTDCLSFYPKTYCVQIQA [00260] SEQ ID NO:15 – Artificial Sequence 13 (TfR Binding Peptide 13; 20 aa) WTYMDCAMYYPYEYCLQIIA [00261] SEQ ID NO:16 – Artificial Sequence 14 (TfR Binding Peptide 14; 20 aa) GKIFNCSEFYPVEFCHQFAA [00262] SEQ ID NO:17 – Artificial Sequence 15 (Formula I; 16 aa) X1X2X3CRSFYPX10X11YCX14AM [00263] SEQ ID NO:18 – Artificial Sequence 16 (TfR Binding Peptide 15; 16 aa) FWDCRSFYPFDYCEAM [00264] SEQ ID NO:19 – Artificial Sequence 17 (TfR Binding Peptide 16; 16 aa) FWDSRSFYPFDYSEAM [00265] SEQ ID NO:20 – Artificial Sequence 18 (TfR Binding Peptide 17; 16 aa) FWDCRAAAAFDYCEAM [00266] SEQ ID NO:21 – Artificial Sequence 19 (TfR Binding Peptide 18; 17 aa + biotin on the terminal K) FWDCRAAAAFDYCEAMK(biotin) [00267] SEQ ID NO:22 – Artificial Sequence 20 (TfR Binding Peptide 19; 16 aa) FWDSRAAAAFDYSEAM ^
[00268] SEQ ID NO:23 – Artificial 21 (TfR Binding Peptide 20; 16 aa) FWDSRSFAAFDYSEAM [00269] SEQ ID NO:24 – Artificial Sequence 22 (TfR Binding Peptide 21; 17 aa + biotin on the terminal K) FWDSRSFAAFDYSEAMK(biotin) [00270] SEQ ID NO:25 – Artificial Sequence 23 (TfR Binding Peptide 22; 16 aa) FWDCRSAAPFDACEAM [00271] SEQ ID NO:26 – Artificial Sequence 24 (TfR Binding Peptide 23; 16 aa) FWDSRSAAPFDASEAM [00272] SEQ ID NO:27 – Artificial Sequence 25 (TfR Binding Peptide 24; 16 aa) FWDCRSAAAFDACEAM [00273] SEQ ID NO:28 – Artificial Sequence 26 (TfR Binding Peptide 25; 16 aa) FWDSRSAAAFDASEAM [00274] SEQ ID NO:29 – Artificial Sequence 27 (TfR Binding Peptide 26; 17 aa) RLYCVHFSSWLQHCTWH [00275] SEQ ID NO:30 – Artificial Sequence 28 (TfR Binding Peptide 27; 17 aa) YTSCLAVNDYLSLCTIL [00276] SEQ ID NO:31 – Artificial Sequence 29 (TfR Binding Peptide 28; 20 aa) YVRSCKYIIPDILAICTSLE [00277] SEQ ID NO:32 – Artificial Sequence 30 (TfR Binding Peptide 29; 20 aa) QPTTDCLSFYPKTYCVHIQA [00278] SEQ ID NO:33 – Artificial Sequence 31 (TfR Binding Peptide 30; 17 aa) ^
SEFCIRIVEGLYICSPN [00279] SEQ ID NO:34 – Artificial Sequence 32 (TfR Binding Peptide 31; 20 aa) WTYMDCAMYYPQEYCQQIIA [00280] SEQ ID NO:35 – Artificial Sequence 33 (TfR Binding Peptide 32; 16 aa) FPDCFWYYPAEYCLGI [00281] SEQ ID NO:36 – Artificial Sequence 34 (hTfR fragment; 15 aa) YVPREAGSQKDENLA [00282] SEQ ID NO:37 – Artificial Sequence 35 (hTfR fragment; 12 aa) KIQVKDSAQNSV [00283] SEQ ID NO:38 – Artificial Sequence 36 (hTfR fragment; 8 aa) TVSNVLKE [00284] SEQ ID NO:39 – Artificial Sequence 37 (hTfR fragment; 9 aa) VENPGGYVA [00285] SEQ ID NO:40 – Artificial Sequence 38 (hTfR fragment; 6 aa) QTKFPI [00286] SEQ ID NO:41 – Artificial Sequence 39 (hTfR fragment; 7 aa) SRAAAEK [00287] SEQ ID NO:42 – Human SNCA mRNA sequence (3177 nt) ggcgacgaccagaaggggcccaagagagggggcgagcgaccgagcgccgcgacgcggaagtgaggtgcgtgcgggctgcag cgcagaccccggcccggcccctccgagagcgtcctgggcgctccctcacgccttgccttcaagccttctgcctttccaccctcgtgag cggagaactgggagtggccattcgacgacagtgtggtgtaaaggaattcattagccatggatgtattcatgaaaggactttcaaaggcc aaggagggagttgtggctgctgctgagaaaaccaaacagggtgtggcagaagcagcaggaaagacaaaagagggtgttctctatgt aggctccaaaaccaaggagggagtggtgcatggtgtggcaacagtggctgagaagaccaaagagcaagtgacaaatgttggagga ^
aggaccagttgggcaagaatgaagaaggagccccacaggaaggaattctggaagatatgcctgtggatcctgacaatgaggcttatg aaatgccttctgaggaagggtatcaagactacgaacctgaagcctaagaaatatctttgctcccagtttcttgagatctgctgacagatgt tccatcctgtacaagtgctcagttccaatgtgcccagtcatgacatttctcaaagtttttacagtgtatctcgaagtcttccatcagcagtgat tgaagtatctgtacctgcccccactcagcatttcggtgcttccctttcactgaagtgaatacatggtagcagggtctttgtgtgctgtggatt ttgtggcttcaatctacgatgttaaaacaaattaaaaacacctaagtgactaccacttatttctaaatcctcactatttttttgttgctgttgttca gaagttgttagtgatttgctatcatatattataagatttttaggtgtcttttaatgatactgtctaagaataatgacgtattgtgaaatttgttaata tatataatacttaaaaatatgtgagcatgaaactatgcacctataaatactaaatatgaaattttaccattttgcgatgtgttttattcacttgtgt ttgtatataaatggtgagaattaaaataaaacgttatctcattgcaaaaatattttatttttatcccatctcactttaataataaaaatcatgcttat aagcaacatgaattaagaactgacacaaaggacaaaaatataaagttattaatagccatttgaagaaggaggaattttagaagaggtag agaaaatggaacattaaccctacactcggaattccctgaagcaacactgccagaagtgtgttttggtatgcactggttccttaagtggctg tgattaattattgaaagtggggtgttgaagaccccaactactattgtagagtggtctatttctcccttcaatcctgtcaatgtttgctttacgtat tttggggaactgttgtttgatgtgtatgtgtttataattgttatacatttttaattgagccttttattaacatatattgttatttttgtctcgaaataattt tttagttaaaatctattttgtctgatattggtgtgaatgctgtacctttctgacaataaataatattcgaccatgaataaaaaaaaaaaaaaagt gggttcccgggaactaagcagtgtagaagatgattttgactacaccctccttagagagccataagacacattagcacatattagcacatt caaggctctgagagaatgtggttaactttgtttaactcagcattcctcactttttttttttaatcatcagaaattctctctctctctctctctttttctc tcgctctctttttttttttttttttacaggaaatgcctttaaacatcgttggaactaccagagtcaccttaaaggagatcaattctctagactgata aaaatttcatggcctcctttaaatgttgccaaatatatgaattctaggatttttccttaggaaaggtttttctctttcagggaagatctattaactc cccatgggtgctgaaaataaacttgatggtgaaaaactctgtataaattaatttaaaaattatttggtttctctttttaattattctggggcatag tcatttctaaaagtcactagtagaaagtataatttcaagacagaatattctagacatgctagcagtttatatgtattcatgagtaatgtgatata tattgggcgctggtgaggaaggaaggaggaatgagtgactataaggatggttaccatagaaacttccttttttacctaattgaagagaga ctactacagagtgctaagctgcatgtgtcatcttacactagagagaaatggtaagtttcttgttttatttaagttatgtttaagcaaggaaagg atttgttattgaacagtatatttcaggaaggttagaaagtggcggttaggatatattttaaatctacctaaagcagcatattttaaaaatttaaa
aatttaaggatttatgtggatacaaattctcctttaaagtgtttcttcccttaatatttatctgacggtaatttttgagcagtgaattactttatatat cttaatagtttatttgggaccaaacacttaaacaaaaagttctttaagtcatataagccttttcaggaagcttgtctcatattcactcccgaga cattcacctgccaagtggcctgaggatcaatccagtcctaggtttattttgcagacttacattctcccaagttattcagcctcatatgactcc acggtcggctttaccaaaacagttcagagtgcactttggcacacaattgggaacagaacaatctaatgtgtggtttggtattccaagtgg ggtctttttcagaatctctgcactagtgtgagatgcaaacatgtttcctcatctttctggcttatccagtatgtagctatttgtgacataataaat atatacatatatgaaaata^ [00288] SEQ ID NO:43 – Human HPRT mRNA sequence (1395 nt) ^
tgctccgccaccggcttcctcctcctgagcagtcagcccgcgcgccggccggctccgttatggcgacccgcagccctggcgtcgtga ttagtgatgatgaaccaggttatgaccttgatttattttgcatacctaatcattatgctgaggatttggaaagggtgtttattcctcatggacta attatggacaggactgaacgtcttgctcgagatgtgatgaaggagatgggaggccatcacattgtagccctctgtgtgctcaaggggg gctataaattctttgctgacctgctggattacatcaaagcactgaatagaaatagtgatagatccattcctatgactgtagattttatcagact gaagagctattgtaatgaccagtcaacaggggacataaaagtaattggtggagatgatctctcaactttaactggaaagaatgtcttgatt gtggaagatataattgacactggcaaaacaatgcagactttgctttccttggtcaggcagtataatccaaagatggtcaaggtcgcaagc ttgctggtgaaaaggaccccacgaagtgttggatataagccagactttgttggatttgaaattccagacaagtttgttgtaggatatgccct tgactataatgaatacttcagggatttgaatcatgtttgtgtcattagtgaaactggaaaagcaaaatacaaagcctaagatgagagttcaa gttgagtttggaaacatctggagtcctattgacatcgccagtaaaattatcaatgttctagttctgtggccatctgcttagtagagctttttgc atgtatcttctaagaattttatctgttttgtactttagaaatgtcagttgctgcattcctaaactgtttatttgcactatgagcctatagactatcag ttccctttgggcggattgttgtttaacttgtaaatgaaaaaattctcttaaaccacagcactattgagtgaaacattgaactcatatctgtaag aaataaagagaagatatattagttttttaattggtattttaatttttatatatgcaggaaagaatagaagtgattgaatattgttaattataccacc gtgtgttagaaaagtaagaagcagtcaattttcacatcaaagacagcatctaagaagttttgttctgtcctggaattattttagtagtgtttca gtaatgttgactgtattttccaacttgttcaaattattaccagtgaatctttgtcagcagttcccttttaaatgcaaatcaataaattcccaaaaa tttaa [00289] SEQ ID NO:44 – Artificial Sequence 40 (TfR Binding Peptide 33; 16 aa) FWDCR(Aib)FYP(f)DYCEAM [00290] SEQ ID NO:45 – Artificial Sequence 41 (TfR Binding Peptide 34; 16 aa) FWDCR(Aib)FYP(f)DYCEA(Nle) [00291] SEQ ID NO:46 – Artificial Sequence 42 (TfR Binding Peptide 35; 16 aa) FWDCR(Aib)FYP(f)DYCEA(Hsm) ^
Claims
The invention claimed is: 1. An engineered peptide comprising an amino acid sequence having at least about 95% sequence identity to any one of SEQ ID NOS:1 to 16, 18, 29 to 35 and 44 to 46, or a pharmaceutically acceptable salt thereof. 2. The engineered peptide of Claim 1, wherein the amino acid sequence is SEQ ID NO:1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 18, 29, 30, 31, 32, 33, 34, 35, 44, 45 or 46, or a pharmaceutically acceptable salt thereof. 3. A compound comprising an engineered peptide comprising an amino acid sequence having at least about 95% sequence identity to any of SEQ ID NOS:1 to 16, 18, 29 to 35 and 44 to 46; and a therapeutic agent linked thereto. 4. The compound of Claim 3, wherein the amino acid sequence of the engineered peptide is SEQ ID NO:1,
2,
3,
4,
5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 18, 29, 30, 31, 32, 33, 34, 35, 44, 45 or 46 or a pharmaceutically acceptable salt thereof 5. The compound of Claim 3 or 4, wherein the compound is a conjugate.
6. The compound of Claim 3 or 4, wherein the compound is a fusion.
7. The compound of any one of Claims 3 to 6, wherein the therapeutic agent is selected from the group consisting of an oligonucleotide, another peptide or protein, or a small molecule.
8. The compound of Claim 7, wherein the oligonucleotide is selected from the group consisting of an activating ribonucleic acid (aRNA) agent, an editing ribonucleic acid (eRNA) agent, an inhibiting ribonucleic acid (iRNA) agent, a messenger ribonucleic acid (mRNA) agent and transfer RNA (rRNA) agent. ^
9. The compound of Claim 7, wherein the another peptide or protein in an antibody.
10. A delivery vehicle comprising: an engineered peptide of Claim 1 or 2 or a compound of any one of Claims 3 to 9; and a nanomedicine.
11. The delivery vehicle of Claim 10, wherein the nanomedicine is a nanoparticle selected from the group consisting of an enveloped nanocomplex (eNCP), a lipid nanoparticle (LNP), a liposome, a nanocapsule (NC), a nanocrystal (NCR), a nanoemulsion (NE) and a proteolipid vehicle (PLV).
12. The delivery vehicle of Claim 10, wherein the nanomedicine is a recombinant adeno- associated virus (rAAV).
13. The delivery vehicle of Claim 12, wherein the rAAV comprises a capsid protein selected from the group consisting of AAV1, AAV2, AAV3, AAV4, AAV5, AAV6, AAV7, AAV8 and AAV9 or a variant thereof.
14. A pharmaceutical composition comprising: an engineered peptide of Claim 1 or 2, a compound of any one of Claims 3 to 9, or a delivery vehicle of any one of Claims 10 to 13; and a pharmaceutically acceptable carrier.
15. A method of targeting a therapeutic agent across the blood-brain barrier (BBB) of an individual, the method comprising the step of: administering to the individual an effective amount of a compound of any one of Claims 3 to 9, a delivery vehicle of any one of Claims 10 to 13, or a pharmaceutical composition of Claim 14. ^
16. The method of Claim 15, wherein administering is via a direct injection into the central nervous system (CNS) of the individual, a peripheral injection into the individual or a combination thereof.
17. The method of Claim 16, wherein the direct injection into the CNS is selected from the group consisting of an intracerebroventricular (ICV) injection, an intracisterna magna (ICM) injection, an intreparenchymal (IPM) injection, an intrathecal (IT) injection and a combination thereof.
18. The method of Claim 16 or 17, wherein the peripheral injection is selected from the group consisting of an intravenous (IV) injection or a subcutaneous (SC) injection.
19. A method of treating an individual having a CNS disease, disorder or condition, the method comprising the step of: administering to the individual an effective amount of a compound of any one of Claims 3 to 9, a delivery vehicle of any one of Claims 10 to 13, or a pharmaceutical composition of Claim 14.
20. The method of Claim 19, wherein the administering is via a direct injection into the central nervous system (CNS) of the individual, a peripheral injection into the individual or a combination thereof.
21. The method of Claim 20, wherein the direct injection into the CNS is selected from the group consisting of an intracerebroventricular (ICV) injection, an intracisterna magna (ICM) injection, an intreparenchymal (IPM) injection, an intrathecal (IT) injection and a combination thereof.
22. The method of Claim 20 or 21, wherein the peripheral injection is selected from the group consisting of an intravenous (IV) injection or a subcutaneous (SC) injection.
23. The method of any one of Claims 19 to 22, wherein the CNS disease, disorder or condition is selected from the group consisting of attention deficit hyperactivity disorder ^
(ADHD), autism, Alzheimer’s disease (AD) epilepsy, frontotemporal dementia (FTD), Huntington’s disease (HD), infection/inflammation, migraine, multiple sclerosis (MS), Parkinson’s disease (PD), seizure and stroke.
24. A method of treating an individual having a transferrin receptor (TfR)-associated disease, disorder or condition, the method comprising the step of: administering to the individual an effective amount of a compound of any one of Claims 3 to 9, a delivery vehicle of any one of Claims 10 to 13, or a pharmaceutical composition of Claim 15.
25. The method of Claim 24, wherein the administering is via a direct injection into the central nervous system (CNS) of the individual, a peripheral injection into the individual or a combination thereof.
26. The method of Claim 25, wherein the direct injection into the CNS is selected from the group consisting of an intracerebroventricular (ICV) injection, an intracisterna magna (ICM) injection, an intreparenchymal (IPM) injection, an intrathecal (IT) injection and a combination thereof.
27. The method of Claim 25 or 26, wherein the peripheral injection is selected from the group consisting of an intravenous (IV) injection or a subcutaneous (SC) injection.
28. The method of any one of Claims 24 to 27, wherein the TfR-associated disease, disorder or condition is selected from the group consisting of anemia, cancer and neurodegenerative disease.
29. Use of an engineered peptide of Claim 1 or 2, a compound of any one of Claims 3 to 9 or a delivery vehicle of any one of Claims 10 to 13 in the manufacture of a medicament for treatment of a central nervous system (CNS) disease, disorder or condition. ^
30. Use of an engineered peptide of 1 or 2, a compound of any one of Claims 3 to 9 or a delivery vehicle of any one of Claims 10 to 13 in the manufacture of a medicament for treatment of a transferrin receptor (TfR)-associated disease, disorder or condition.
31. An engineered peptide of Claim 1 or 2, a compound of any one of Claims 3 to 9 or a delivery vehicle of any one of Claims 10 to 13 for use in treating a central nervous system (CNS) disease, disorder or condition.
32. An engineered peptide of Claim 1 or 2, a compound of any one of Claims 3 to 9 or a delivery vehicle of any one of Claims 10 to 13 for use treating a transferrin receptor (TfR)- associated disease, disorder or condition.
33. An engineered transferrin receptor (TfR) binding peptide comprising an amino acid sequence of: X1X2X3CRX6FYPX10X11YCX14AX16 (Formula I; SEQ ID NO:17), wherein X1 is a naturally occurring amino acid or a variant thereof, wherein X2 is a naturally occurring amino acid or a variant thereof, wherein X3 is a naturally occurring amino acid or a variant thereof, wherein X6 is a naturally occurring amino acid or a variant thereof, wherein X10 is a naturally occurring amino acid or a variant thereof, wherein X11 is a naturally occurring amino acid or a variant thereof, wherein X14 is a naturally occurring amino acid or a variant thereof, and wherein X16 is a naturally occurring amino acid or a variant thereof.
34. The engineered TfR binding peptide of Claim 33, wherein X1, X2 and X10 each and independently are an aromatic amino acid.
35. The engineered TfR binding peptide of Claim 33 or 34, wherein X3, X11 and X14 each and independently are an acidic amino acid.^ 36. The engineered TfR binding peptide of Claim 33, wherein X1 is F, I, L, M, W or Y, ^
wherein X2 is F, I, L, M, W or Y, wherein X3 is D, E, H, K, N, R, Q or S, wherein X6 is S or Aib, wherein X10 is f, F, I, L, M, W or Y, wherein X11 is D, E, H, K, N, R, Q or S, wherein X14 is D, E, H, K, N, R, Q or S, and wherein X16 is M, Hsm or Nle. 37. The engineered TfR binding peptide of Claim 36, wherein X1 is F, wherein X2 is W, wherein X3 is D, wherein X6 is S, wherein X10 is F, wherein X11 is D, wherein X14 is E, and wherein X16 is M (SEQ ID NO:18). 38. The engineered TfR binding peptide of Claim 36, wherein X1 is F, wherein X2 is W, wherein X3 is D, wherein X6 is Aib, wherein X10 is f, wherein X11 is D, wherein X14 is E, and wherein X16 is M (SEQ ID NO:44). 39. The engineered TfR binding peptide of Claim 36, wherein X1 is F, wherein X2 is W, wherein X3 is D, ^
wherein X6 is Aib, wherein X10 is f, wherein X11 is D, wherein X14 is E, and wherein X16 is Nle (SEQ ID NO:45). 40. The engineered TfR binding peptide of Claim 36, wherein X1 is F, wherein X2 is W, wherein X3 is D, wherein X6 is Aib, wherein X10 is f, wherein X11 is D, wherein X14 is E, and wherein X16 is Hsm (SEQ ID NO:46). ^
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US202363520380P | 2023-08-18 | 2023-08-18 | |
| US63/520,380 | 2023-08-18 | ||
| US202463654192P | 2024-05-31 | 2024-05-31 | |
| US63/654,192 | 2024-05-31 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2025042711A1 true WO2025042711A1 (en) | 2025-02-27 |
Family
ID=92593344
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2024/042617 Pending WO2025042711A1 (en) | 2023-08-18 | 2024-08-16 | Engineered transferrin receptor binding peptides as well as methods of making and using the same |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2025042711A1 (en) |
Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2005033321A2 (en) | 2003-09-30 | 2005-04-14 | The Trustees Of The University Of Pennsylvania | Adeno-associated virus (aav) clades, sequences, vectors containing same, and uses therefor |
| WO2020028836A1 (en) * | 2018-08-02 | 2020-02-06 | Dyne Therapeutics, Inc. | Muscle-targeting complexes and uses thereof in treating muscle atrophy |
| WO2020144233A1 (en) | 2019-01-09 | 2020-07-16 | Vect-Horus | Transferrin receptor-binding molecules, conjugates thereof and their uses |
| WO2022026555A1 (en) | 2020-07-30 | 2022-02-03 | University Of Washington | Transferrin receptor binding proteins |
| US20220048961A1 (en) * | 2018-12-14 | 2022-02-17 | Fred Hutchinson Cancer Research Center | Transferrin receptor targeting peptides |
| WO2022067446A1 (en) | 2020-10-01 | 2022-04-07 | Entos Pharmaceuticals Inc. | Proteolipid vesicles formulated with fusion associated small transmembrane proteins |
-
2024
- 2024-08-16 WO PCT/US2024/042617 patent/WO2025042711A1/en active Pending
Patent Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2005033321A2 (en) | 2003-09-30 | 2005-04-14 | The Trustees Of The University Of Pennsylvania | Adeno-associated virus (aav) clades, sequences, vectors containing same, and uses therefor |
| WO2020028836A1 (en) * | 2018-08-02 | 2020-02-06 | Dyne Therapeutics, Inc. | Muscle-targeting complexes and uses thereof in treating muscle atrophy |
| US20220048961A1 (en) * | 2018-12-14 | 2022-02-17 | Fred Hutchinson Cancer Research Center | Transferrin receptor targeting peptides |
| WO2020144233A1 (en) | 2019-01-09 | 2020-07-16 | Vect-Horus | Transferrin receptor-binding molecules, conjugates thereof and their uses |
| WO2022026555A1 (en) | 2020-07-30 | 2022-02-03 | University Of Washington | Transferrin receptor binding proteins |
| WO2022067446A1 (en) | 2020-10-01 | 2022-04-07 | Entos Pharmaceuticals Inc. | Proteolipid vesicles formulated with fusion associated small transmembrane proteins |
Non-Patent Citations (25)
| Title |
|---|
| "Remington, ''The Science and Practice of Pharmacy", 2006, UNIVERSITY OF THE SCIENCES IN PHILADELPHIA |
| BRITO ET AL., MOL. THER., vol. 22, 2014, pages 2118 - 2129 |
| BROWN ET AL., SAFE AND EFFECTIVE DELIVERY OF NUCLEIC ACIDS USING PROTEOLIPID VEHICLES FORMULATED WITH FUSION-ASSOCIATED SMALL TRANSMEMBRANE PROTEINS, 2022 |
| CHANG ET AL., PROTEIN ENG. DES. SEL., vol. 33, 2020, pages 1 - 8 |
| CHIRCOVGRUMEZESCU: "Nanoemulsion Preparation. Characterization, and Application in the Field of Biomedicine", NANOARCHITECTONICS IN BIOMEDICINE, pages 169 - 188 |
| GUO ET AL., NAT. COMM., vol. 11, 2020, pages 972 |
| HOU ET AL., NAT. REV. MATER., vol. 6, 2021, pages 1078 - 1094 |
| JAHANGIR ET AL., J. PHARM. INNOV., vol. 17, 2020, pages 237 - 248 |
| JASINSKI ET AL., ACS NANO., vol. 11, 2017, pages 1142 - 1164 |
| JIANG ET AL., J. CONTROL RELEASE, vol. 283, 2018, pages 235 - 240 |
| KIM ET AL., ACSNANO., vol. 15, 2021, pages 4576 - 4593 |
| MASHIMATAKADA, CURR. ISSUES MOL. BIOL., vol. 44, 2022, pages 5013 - 5027 |
| MCQUIAD ET AL., PLOS ONE, vol. 16, 2021, pages e0252341 |
| PARDHI ET AL., CURR. PHARM. DES., vol. 24, 2018, pages 5129 - 5146 |
| PI ET AL., NAT. NANOTECHNOL., vol. 13, 2018, pages 89 - 89 |
| RATURI ET AL., RAPID PROTO!) PING AND IMMUNOGENICITY OF SARS-COV-2 DNA VACCINE CANDIDATES FORMULATED WITH THE FUSION-ASSOCIATED SMALL TRANSMEMBRANE PROTEIN PROTEOLIPID VEHICLE DELIVERY SYSTEM, 2022 |
| SADE, PLOS ONE, vol. 9, 2014, pages e96340 |
| SAMARIDOU ET AL., BIOMATERIALS, vol. 230, 2020, pages 119657 |
| SHU ET AL., ACS NANO, vol. 9, 2015, pages 9731 - 9740 |
| STAHL ET AL.: "Handbook of Pharmaceutical Salts: Properties, Selection and Use", 2011, WILEY-VCH |
| TENCHOV ET AL., ACS NANO, vol. 1 1, 2021, pages 16982 - 17015 |
| TING ET AL., YI,«S ONE, vol. 13, 2018, pages 190950 |
| XUE ET AL., CURR. PHARM. DES., vol. 21, 2015, pages 3140 - 3147 |
| YAN ET AL., J. AM. CHEM. SOC., vol. 134, 2012, pages 13542 - 13545 |
| Σ-LANDA ET AL.: "Rapid and Reliable Site-Directed Mutagenesis Using Kunkel's Approach", METHODS IN MOLECULAR BIOLOGY, vol. 182 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| Stanimirovic et al. | Emerging Technologies for Delivery of Biotherapeutics and Gene Therapy Across the Blood–Brain Barrier: DB Stanimirovic et al. | |
| KR101317100B1 (en) | Peptides useful as cell-penetrating peptides | |
| US8680045B2 (en) | Compositions of a peptide targeting system for treating cancer | |
| CN114127089A (en) | Recombinant adeno-associated virus and use thereof | |
| JP6077543B2 (en) | peptide | |
| US20250302963A1 (en) | Peptides and nanoparticles for intracellular delivery of molecules | |
| JP7014857B2 (en) | Compositions and Methods for Transporting Through the Blood-Brain Barrier | |
| JP2014504150A5 (en) | ||
| WO2013036889A1 (en) | Retrograde transport peptide and use of same for delivery to central nervous system | |
| JP2024029097A (en) | Mininucleosome core protein and use in nucleic acid delivery | |
| AU2014267044A1 (en) | Bacteriophage | |
| AU2024329222A1 (en) | Engineered transferrin receptor binding peptides as well as methods of making and using the same | |
| WO2025042711A1 (en) | Engineered transferrin receptor binding peptides as well as methods of making and using the same | |
| Ranka et al. | Fibronectin-binding nanoparticles for intracellular targeting addressed by B. burgdorferi BBK32 protein fragments | |
| KR20230034334A (en) | Extracellular vesicles with improved half-life | |
| US20230128981A1 (en) | Cd24-loaded vesicles for treatment of cytokine storm and other conditions | |
| KR20220117091A (en) | An Expression cassette encoding a polypeptide of interest synthesized and secreted from cells in vivo and penetrating into cells, and uses thereof | |
| WO2025226642A1 (en) | Lipid nano delivery particles for targeted transport of nucleic acids and methods of making and using the same | |
| WO2024216244A2 (en) | Targeting aav capsids, methods of manufacturing and using same | |
| WO2025084336A1 (en) | Molecule capable of simultaneously binding to aav capsid and target receptor | |
| WO2023014724A2 (en) | Scaffold matrix attachment regions for gene therapy | |
| SAXENA | In-Silico Designing, Synthesis and Modification of Papilloma based CPPs in Drug Delivery Thesis | |
| Vivès et al. | Peptide-Mediated Delivery of Antisense Oligonucleotides and Related Material | |
| Begum | Development of peptide-based multicomponent systems for the targeted delivery of oligonucleotides | |
| Liew et al. | 994. Comparative studies of oral and intramuscular immunization with mite allergen gene encapsulated with chitosan |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 24762805 Country of ref document: EP Kind code of ref document: A1 |
|
| REG | Reference to national code |
Ref country code: BR Ref legal event code: B01A Ref document number: 112026000582 Country of ref document: BR |
|
| WWE | Wipo information: entry into national phase |
Ref document number: AU2024329222 Country of ref document: AU |