WO2025038600A1 - Methods for reducing yellow color - Google Patents
Methods for reducing yellow color Download PDFInfo
- Publication number
- WO2025038600A1 WO2025038600A1 PCT/US2024/042037 US2024042037W WO2025038600A1 WO 2025038600 A1 WO2025038600 A1 WO 2025038600A1 US 2024042037 W US2024042037 W US 2024042037W WO 2025038600 A1 WO2025038600 A1 WO 2025038600A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- wash
- sodium
- yellow color
- containing molecule
- sample
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K1/00—General methods for the preparation of peptides, i.e. processes for the organic chemical preparation of peptides or proteins of any length
- C07K1/14—Extraction; Separation; Purification
- C07K1/16—Extraction; Separation; Purification by chromatography
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D15/00—Separating processes involving the treatment of liquids with solid sorbents; Apparatus therefor
- B01D15/08—Selective adsorption, e.g. chromatography
- B01D15/10—Selective adsorption, e.g. chromatography characterised by constructional or operational features
- B01D15/16—Selective adsorption, e.g. chromatography characterised by constructional or operational features relating to the conditioning of the fluid carrier
- B01D15/166—Fluid composition conditioning, e.g. gradient
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D15/00—Separating processes involving the treatment of liquids with solid sorbents; Apparatus therefor
- B01D15/08—Selective adsorption, e.g. chromatography
- B01D15/10—Selective adsorption, e.g. chromatography characterised by constructional or operational features
- B01D15/20—Selective adsorption, e.g. chromatography characterised by constructional or operational features relating to the conditioning of the sorbent material
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D15/00—Separating processes involving the treatment of liquids with solid sorbents; Apparatus therefor
- B01D15/08—Selective adsorption, e.g. chromatography
- B01D15/26—Selective adsorption, e.g. chromatography characterised by the separation mechanism
- B01D15/36—Selective adsorption, e.g. chromatography characterised by the separation mechanism involving ionic interaction, e.g. ion-exchange, ion-pair, ion-suppression or ion-exclusion
- B01D15/361—Ion-exchange
- B01D15/362—Cation-exchange
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D15/00—Separating processes involving the treatment of liquids with solid sorbents; Apparatus therefor
- B01D15/08—Selective adsorption, e.g. chromatography
- B01D15/26—Selective adsorption, e.g. chromatography characterised by the separation mechanism
- B01D15/38—Selective adsorption, e.g. chromatography characterised by the separation mechanism involving specific interaction not covered by one or more of groups B01D15/265 and B01D15/30 - B01D15/36, e.g. affinity, ligand exchange or chiral chromatography
- B01D15/3804—Affinity chromatography
- B01D15/3809—Affinity chromatography of the antigen-antibody type, e.g. protein A, G or L chromatography
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K1/00—General methods for the preparation of peptides, i.e. processes for the organic chemical preparation of peptides or proteins of any length
- C07K1/14—Extraction; Separation; Purification
- C07K1/16—Extraction; Separation; Purification by chromatography
- C07K1/22—Affinity chromatography or related techniques based upon selective absorption processes
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
- C07K16/06—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies from serum
- C07K16/065—Purification, fragmentation
Definitions
- the present invention relates to methods of reducing yellow color and/or impurities during purification of constructs containing an Fc.
- the methods of the present invention relate to purification of Fc-containing molecules, such as antibodies.
- Color is an important quality attribute for therapeutic drug substance.
- the color of a monoclonal antibody (mAb) drug substance can vary due to multiple reasons, such as association with cell culture media component or stress-induced chemical modification of amino acids in the mAb sequence (Ambrogelly, Antibodies 2021, 10(2), 21).
- the ability to control and/or reduce color-causing species, without losing potency, during downstream purification is important to manufacturing drug substance for patients.
- the present invention provides a method of reducing yellow color in an Fc- containing molecule sample.
- the method comprises capture affinity, wherein the capture affinity comprises a detergent wash that is sodium deoxycholate and/or sodium caprylate.
- the capture affinity comprises sodium deoxycholate.
- the capture affinity comprises sodium caprylate.
- the capture affinity comprises a sodium deoxycholate step and a sodium caprylate step.
- the deoxycholate wash comprises about 10 mM to about 30 mM sodium deoxycholate.
- the deoxy cholate wash comprises about 15 mM to about 25 mM sodium deoxycholate.
- the deoxycholate wash comprises about 10 mM, 11 mM, 12 mM, 13 mM, 14 mM, 15 mM, 16 mM, 17 mM, 18 mM, 19 mM, 20 mM, 21 mM, 22 mM, 23 mM, 24 mM, 25 mM, 26 mM, 27 mM, 28 mM, 29 mM, or 30 mM.
- the deoxycholate wash comprises about 20 mM sodium deoxycholate.
- the deoxycholate wash comprises 20 mM sodium deoxycholate.
- the deoxycholate wash comprises about 15 to about 35 mM Tris, about 10 to about 30 mM sodium deoxy cholate, and about 90 to about 110 mM NaCl. In an embodiment, the deoxy cholate wash comprises about 25 mM Tris, about 20 mM sodium deoxycholate, and about 100 mM NaCl. In an embodiment, the deoxycholate wash comprises 25 mM Tris, 20 mM sodium deoxycholate, and 100 mM NaCl. In an embodiment, the deoxycholate wash is at about pH 8.0-8.4. In an embodiment, the deoxycholate wash is at pH 8.2.
- the sodium caprylate wash comprises about 80 mM to about 120 mM sodium caprylate. In an embodiment, the sodium caprylate wash comprises about 90 mM to about 110 mM sodium caprylate. In an embodiment, the sodium caprylate wash comprises about 95 mM to about 105 mM sodium caprylate. In an embodiment, the sodium caprylate wash comprises about 100 mM sodium caprylate. In an embodiment, the sodium caprylate wash comprises 100 mM sodium caprylate. In an embodiment, the sodium caprylate wash comprises about 15 to about 35 mM Tris, about 90 to about 110 mM sodium caprylate and about 2 to about 3 M NaCl.
- the sodium caprylate wash comprises about 25 mM Tris, about 100 mM sodium caprylate and about 2.5 M NaCl. In an embodiment, the sodium caprylate wash comprises 25 mM Tris, 100 mM sodium caprylate and 2.5 M NaCl. In a particular embodiment, the sodium caprylate wash is at about pH 7.2 to about 7.6. In a particular embodiment, the sodium caprylate wash is at pH 7.4. In an embodiment, the deoxycholate wash is performed prior to the caprylate wash. In an embodiment, the caprylate wash is performed prior to the deoxycholate wash.
- the present invention provides a method of detecting yellow color in an Fc- containing molecule sample, wherein said method is a cation exchange (CEX) method comprising a strong cation exchange resin and detecting UV absorbance at 400 nm and 420 nm.
- the CEX method comprises a gradient elution.
- the gradient elution is from about 0% of 100 mM sodium acetate, pH 5.0, 500 mM sodium chloride, and about 100 % of 100 mM sodium acetate, pH 5.0; to about 65% of 100 mM sodium acetate, pH 5.0, 500 mM sodium chloride, and about 100 % of 100 mM sodium acetate, pH 5.0.
- the gradient elution is from 0% of 100 mM sodium acetate, pH 5.0, 500 mM sodium chloride, and 100 % of 100 mM sodium acetate, pH 5.0; to 65% of 100 mM sodium acetate, pH 5.0, 500 mM sodium chloride, and 100 % of 100 mM sodium acetate, pH 5.0.
- the gradient elution is performed at a rate of 8 mM/CV.
- the present invention provides a method of reducing yellow color in an Fc- containing molecule sample, wherein said method is a CEX method comprising a strong cation exchange resin and a sodium chloride salt wash.
- the sodium chloride salt wash is about 60 mM to about 90 mM sodium chloride.
- the sodium chloride salt wash is about 65 mM to about 85 mM sodium chloride.
- the sodium chloride salt wash is about 75 mM sodium chloride.
- the sodium chloride salt wash is 75 mM sodium chloride.
- the sodium chloride salt wash is 100 mM sodium acetate, pH 5.0, and 75 mM sodium chloride.
- the method comprises a gradient elution.
- the gradient elution is from: 0% of 100 mM sodium acetate, pH 5.0, 500 mM sodium chloride, and 100 % of 100 mM sodium acetate, pH 5.0; to 65% of 100 mM sodium acetate, pH 5.0, 500 mM sodium chloride, and 100 % of 100 mM sodium acetate, pH 5.0.
- the present invention provides a method of reducing yellow color in an Fc- containing molecule sample, comprising a combination of any of the methods described herein.
- the method comprises a method comprising capture affinity, wherein the capture affinity comprises a detergent wash that is sodium deoxycholate and/or sodium caprylate, and a method comprising a CEX method comprising a strong cation exchange resin and a sodium chloride salt wash.
- the capture affinity comprises sodium deoxycholate.
- the capture affinity comprises sodium caprylate.
- the capture affinity comprises a sodium deoxycholate step and a sodium caprylate step.
- the deoxycholate wash comprises 20 mM sodium deoxycholate.
- the deoxycholate wash comprises 25 mM Tris, 20 mM sodium deoxycholate, and 100 mM NaCl. In a particular embodiment, the deoxycholate wash is at about pH 8.2. In an embodiment, the sodium caprylate wash comprises 100 mM sodium caprylate. In an embodiment, the sodium caprylate wash comprises 25 mM Tris, 100 mM sodium caprylate and 2.5 M NaCl. In a particular embodiment, the sodium caprylate wash is at about pH 7.4. In an embodiment, the deoxycholate wash is performed prior to the caprylate wash. In an embodiment, the caprylate wash is performed prior to the deoxycholate wash. In an embodiment, the sodium chloride salt wash is 75 mM sodium chloride.
- the sodium chloride salt wash is 100 mM sodium acetate, pH 5.0, and 75 mM sodium chloride.
- the method comprises a gradient elution.
- the gradient elution is from: 0% of 100 mM sodium acetate, pH 5.0, 500 mM sodium chloride, and 100 % of 100 mM sodium acetate, pH 5.0; to 65% of 100 mM sodium acetate, pH 5.0, 500 mM sodium chloride, and 100 % of 100 mM sodium acetate, pH 5.0.
- the present invention also provides a method of reducing an impurity in an Fc- containing molecule sample, wherein said method comprises a method of the present invention.
- the impurity is detected by liquid chromatography with ultraviolet, fluorescence, and mass spectrometric detection.
- the impurity is a 303 Da species.
- the Fc-containing molecule sample is a drug substance intermediate.
- the method is performed during purification of the Fc- containing molecule.
- the Fc-containing molecule is an antibody.
- the Fc-containing molecule is a heterodimeric antibody that has an Fc.
- the yellow color or the impurity in a sample is reduced compared to yellow color or impurity in a sample that has been subjected to another wash or has not been subjected to any wash.
- the yellow color or impurity is reduced by at least 3%.
- the yellow color or impurity is reduced by at least 5%.
- the yellow color or impurity is reduced by at least 10%.
- the yellow color or impurity is reduced by at least 15%.
- the yellow color or impurity is reduced by at least 20%.
- the yellow color or impurity is reduced by at least 25%.
- the yellow color or impurity is reduced by at least 30%.
- the yellow color or impurity is reduced by at least 35%. In an embodiment, the yellow color or impurity is reduced by at least 40%. In an embodiment, the yellow color or impurity is reduced by at least 45%. In an embodiment, the yellow color or impurity is reduced by at least 50%. In an embodiment, the yellow color is reduced. In an embodiment, the impurity is reduced. In an embodiment, the yellow color is determined by the calculated b* value of the Fc-containing molecule sample.
- the yellow color is determined by calculating the b* value of the Fc-containing molecule sample and comparing the b* value with a b* value of a sample of an Fc-containing molecule that has not been subjected to a wash (method) of the present invention. In an embodiment, the yellow color is determined by visual observation of the sample. In an embodiment, the yellow color is determined by visual observation of the Fc- containing molecule sample to obtain a visually observed yellow color, and comparing the visually observed yellow color with a visually observed yellow color of a sample of an Fc- containing molecule that has not been subjected to a wash (method) of the present invention.
- the sample is an elution pool.
- Figure 1 HunterLab Assay yellow color measurement on samples with and without detergent washes during affinity capture step.
- Figure 1 shows the result of HunterLab assay on up-concentrated neutralized ProA Pool samples or controls.
- Figure 2 LC-UV-FD-MS measurement of 303 Da species on samples with and without detergent washes during affinity capture step.
- C and D detergent wash step and elution pool of proposed method.
- Figure 2 shows measurement of 303 Da species on samples with and without detergent washes during affinity capture step.
- FIG. 3 UV absorbance at 280 nm, 400 nm, and 420 nm during CEX elution step.
- the dotted boxes denote fractions.
- the elution profile revealed higher A400 and A420 absorbances during the first two fractions, which correlates to visibly yellower fractions.
- Figure 4 Hunterlab assay result from various salt wash samples as compared to yellow color standards (Y7-T4).
- FIG. 5 LC-UV-FD-MS results for detection of 303 Da species.
- a and B are 1st elution fraction and elution pool, respectively, using a CEX method without the sodium chloride salt wash step.
- C and D are wash sample and elution pool, respectively, of the CEX method with a sodium chloride salt wash step.
- E is the result from elution pool from an additional column step.
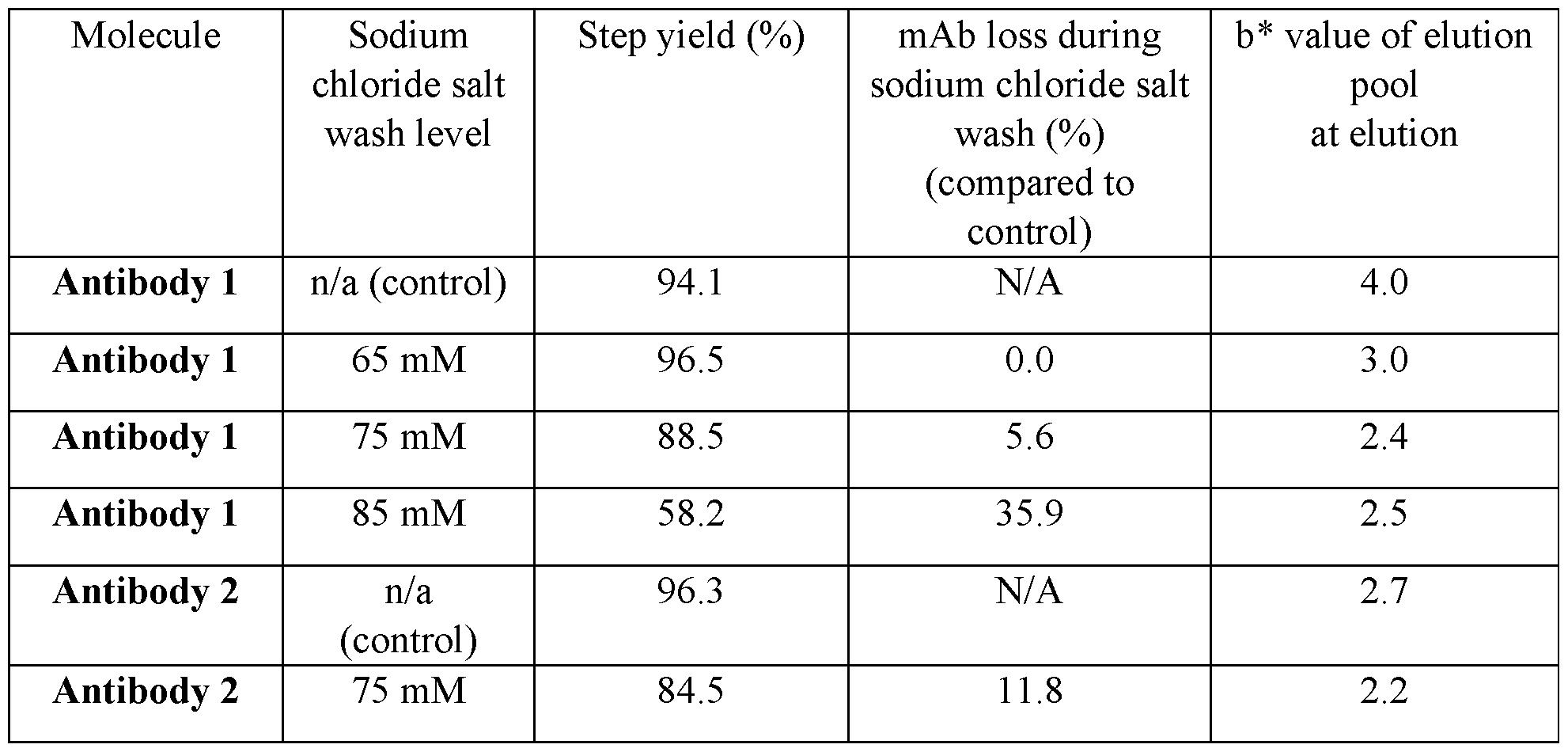
- FIG. 6 Level of 303 Da species in various sodium chloride salt wash samples of Antibody 1 and Antibody 2.
- the peak area was obtained by integration of 303 Da peak on the LC-UV-FD-MS chromatogram and reflects the relative level of 303 Da species in each sample.
- the sodium chloride salt washes are effective in reducing the level of 303 Da species in CEX pool samples.
- Antibody 2 has lower level of 303 Da species compared to Antibody 1.
- Protein A affinity capture step is often the first critical step of purification, specifically designed to separate Fc-containing molecule from cell culture media components and other impurities present in the harvest material.
- the present invention describes the inclusion of a wash step using various salts, such as sodium caprylate and sodium deoxycholate, has been found to help with further removal of host cell protein (HCP) and DNA.
- the capture affinity purification is often the first critical step of therapeutic monoclonal antibody (mAb) purification process. It is designed to separate Fc-containing molecule from cell culture media components and other impurities present in the harvest material.
- mAb therapeutic monoclonal antibody
- the addition of detergent washes can disrupt non-covalent binding to further improve separation between Fc-containing molecules and impurities.
- Two detergents, sodium caprylate Hu et al., Protein Expr Purif.
- sodium deoxycholate can remove host cell proteins including endogenous protease(s).
- the present invention discovered that the implementation of detergent wash(es) during Protein A step (capture affinity) resulted in a sample (e.g. elution pool) with a reduction in yellow color compared to a sample that has been subjected to a different wash or has not been subjected to any wash.
- the detergent washes also resulted in a reduction of an impurity present in cell culture.
- Affinity chromatography e.g. Protein A
- Affinity chromatography is widely employed for the preparative purification of proteins possessing a heavy chain Fc region comprising the CH2 and CH3 domains of an immunoglobulin molecule.
- Gagnon Protein A affinity chromatography, In: Purification tools for monoclonal antibodies, 1996, Validated Biosystems, Arlington, Ariz., 1996; Aybay and Imir, J. Immunol. Methods 233(1-2): 77- 81, 2000; Ford et al., J. Chromatogr.
- Fc proteins include antibodies, particularly monoclonal antibodies, as well as Fc fusion proteins.
- Such proteins are typically produced by mammalian or bacterial cells engineered to express the desired recombinant protein, intracellularly or directly, into the culture medium or intracellularly. Purification of the expressed proteins typically begins with either collecting the cell culture medium to harvest extracellularly expressed protein or harvesting and lysing the host cells to release intracellularly expressed protein.
- This host cell “harvest medium” contains not only the protein of interest but also DNA, RNA, and protein contaminants from the host cell that must be separated from the desired protein. These host cell contaminants may be separated by various chromatographic methods based on their charge, size or hydrophobicity.
- Cation exchange chromatography is a form of ion exchange chromatography, which is used to separate molecules based on their net surface charge. Cation exchange chromatography, more specifically, uses a negatively charged ion exchange resin with an affinity for molecules having net positive surface charges.
- Another way to reduce yellow color was by introducing a sodium chloride salt wash during cation exchange (CEX) polishing step. Since visible yellow color is associated with light absorbance around 400 nm, the AKTA UV detector was used to measure light absorbance at 400nm and 420 nm during a CEX run. The resulting chromatogram revealed that the yellow color species was asymmetrically distributed during the CEX elution step, with higher concentrations found at the beginning of the elution. Interestingly, the earlier fractions were also visibly more yellow. Based on this finding, a sodium chloride salt wash step was then developed to separate early fractions from the rest of the elution peak, which was shown to successfully reduce yellow color in the CEX elution pool.
- CEX cation exchange
- a 303 Da species was identified to be elevated in earlier CEX elution fractions, which are also yellow in color. The 303 Da species and yellow color appear to co-elute during the earlier fractions of the CEX elution. This species was also reduced with the addition of the salt wash step.
- a neutral or basic loading buffer (such as pH 7 to 8) is used to bind the antigen-binding protein onto the Protein A resin.
- low pH is used to elute the antigen-binding protein from the Protein A resin, such as a pH between 3 and 5, such as 3 to 4, or 4 to 5. It is envisioned that other resins or columns, such as Protein G or Protein A/G, are also encompassed in the present invention.
- a chromatography column may be used for any of many different liquid chromatographic processes, including ion-exchange chromatography, size-exclusion chromatography, hydrophobic interaction chromatography, and affinity chromatography.
- ion- exchange chromatography an Fc-containing molecule is separated from a complex solution or mixture based on electrostatic forces between charged functional groups of Fc-containing molecules and charged functional groups of the chromatography-column matrix.
- Cationexchange resins have negatively charged functional groups that attract positively charged functional groups of Fc-containing molecules
- anion-exchange resins have positively charged functional groups that attract negatively charged functional groups of Fc-containing molecules.
- Molecules bound through electrostatic forces to the matrix can be eluted by increasing the ionic strength of the buffer solution within the chromatography column over time.
- size-exclusion chromatography an Fc-containing molecule is separated from a complex solution or mixture based on the Fc-containing molecule's size-related exclusion from the interior regions of spherical beads that make up the matrix. Progress through the chromatography column of smaller molecules that are capable of diffusing into the beads is slowed with respect to the Fc-containing molecule.
- hydrophobic interaction chromatography an Fc-containing molecule is separated from a complex solution or mixture based on the hydrophobicity of the Fc-containing molecule.
- a complex solution containing the Fc-containing molecule is applied to a chromatography column equilibrated with a high salt buffer that facilitates binding of the Fc-containing molecule to the resin.
- a salt-gradient mobile phase with decreasing ionic strength is then introduced into the chromatography column to release bound Fc-containing molecules from the matrix.
- hydrophobic interaction chromatography may separate a monomeric Fc-containing molecule from a complex solution or mixture by binding hydrophobic impurities, including inactive dimers and aggregates of the Fc-containing molecule, while permitting monomeric Fc- containing molecules to flow through the chromatography column relatively unimpeded.
- an Fc-containing molecule such as an antibody
- a complex solution based on the affinity of the Fc-containing molecule for a ligand or ligandbinding entity that is covalently bound to the matrix.
- Molecules in the complex solution or mixture with weak affinity, or lacking affinity, for the ligand or ligand-binding entity flow through the chromatography column unimpeded, leaving the Fc-containing molecule bound to the matrix.
- the Fc-containing molecule can then be eluted from the chromatography column by altering buffer conditions to decrease the affinity of the Fc-containing molecule for the ligand or ligand-binding entity.
- Protein A is a ⁇ 41 kDa protein from Staphylococcus aureas that binds with high affinity ( ⁇ 10— 8 M-10-12 M to human IgG) to the CH2/CH3 domain of the Fc regions of antibodies and is therefore commonly immobilized within an affinity-chromatography matrix for purifying target antibodies. Due to the biochemical properties of protein A, including a lack of disulfide bond linkages, protein A is very stable and can be used with high salt conditions and/or denaturants, such as 10 M urea, 6 M guanidine, and 80 mM dithiothreitol. Protein-A affinity chromatography is often used for purification of monoclonal antibodies and fusion proteins containing the antibody constant fragment Fc. About 98% of process impurities, including viral particles, can be removed by protein-A affinity column chromatography in a single step, with high product yields.
- protein-A affinity chromatography resins that may be used for antibody purification, including ProSep® controlled-pore glass resins produced by Millipore and Mab SelectTM, cross-linked agarose resin products produced by Amersham Biosciences, and other types of protein-A affinity chromatography resins, including gel-based resins and silica-based resins.
- the different types of protein-A affinity chromatography resins have different characteristics and properties, and chromatography columns prepared with a particular type of protein-A affinity chromatography resin may be more suitable than chromatography columns prepared with other types of protein-A affinity chromatography resins for purification of particular Fc-containing molecules under particular conditions.
- Protein-A affinity column chromatography may be used as a first step, sometimes referred to as an initial purification step, or “capture step,” of a multi-step purification process.
- An example of a multi-step purification process includes a capture step and one or more polishing steps.
- the capture step may include adsorbing the protein from a complex solution or complex mixture to protein A immobilized on a solid phase, removing contaminants by washing the solid phase with a wash buffer, and recovering the Fc-containing molecule from the solid phase with an elution buffer.
- the elution buffer is designed to introduce conditions that interfere with protein- A/Fc binding, such as lowering the pH within the column matrix to a point at which Fc no longer binds to protein A.
- the capture step may concentrate the Fc-containing molecule.
- the effectiveness of the capture step may be assessed based on the speed of the chromatography process, the degree of separation of the Fc-containing molecule from undesired solutes and suspended entities, and the load capacity of the chromatography column. Chromatography resins with high load capacities and good flow properties may be particularly well suited for capture steps.
- Suitable buffers for the equilibration and wash steps include tris and sodium chloride, pH 7.4. Acetate can be used for the elution step.
- a range such as, for example, a sodium caprylate wash comprising about 90 mM to about 110 mM sodium caprylate, is intended to include all values in between and including the lowest value (e.g. 90 mM) and the highest value (e.g. 110 mM).
- the polishing steps may separate the Fc-containing molecule that has been concentrated and partially purified in the capture step from remaining impurities.
- the polishing steps may be useful for removing impurities that have relatively closely related chromatographic properties and that eluted together with the Fc-containing molecule during the capture step. Flow rates and load volumes of the polishing steps may be decreased in order to improve resolution.
- the polishing steps may be useful for removing product-related contaminants, such as aggregates, and trace amounts of process-related contaminants, such as viral particles, from the Fc-containing molecule-containing eluate, as well as for adjusting buffer conditions in preparation for subsequent processing steps.
- Ion exchange including cation exchange chromatography (CEX)
- CEX cation exchange chromatography
- an ion exchanger is brought to a starting state, in terms of pH and ionic strength, which allows the binding of the desired solute molecules.
- the exchanger groups are associated at this time with exchangeable counter-ions (usually simple anions or cations, such as chloride or sodium).
- the solute molecules carrying the appropriate charge then displace counter-ions and bind reversibly to the gel. Unbound substances can be washed out from the exchanger bed using starting buffer. Substances are removed from the column by changing to elution conditions unfavorable for ionic bonding of the solute molecules. This may involve increasing the ionic strength of the eluting buffer or changing its pH.
- Yellow color of a sample can be determined according to methods known in the art (see e.g. Ambrogelly A. The Different Colors of mAbs in Solution. Antibodies. 2021; 10(2):21).
- the HunterLab UltraScan PRO spectrophotometer which measures color expressed in the three dimensions of the human color response (ie, red, green and blue) rather than spectral colors (ie, wavelengths) by a physical detector in the visible wavelength range, can be used for the yellow color measurement in antibody samples.
- the reduction in yellow color as part of the present invention is 1% through 100%.
- the reduction in yellow color is 1%, 5%, 10%, 15%, 20%, 25%, 30%, 35%, 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 98%, 99%, or 100%.
- the yellow color is determined by HunterLab assay.
- yellow color of a sample can be determined by visual observation of the sample (e.g. looking at the sample).
- Up-concentrated refers to increasing the concentration of the ProA pool to ensure all of the samples are comparable to each other. This is important since color can intensify with increased concentrations.
- a polishing step refers to column steps that are meant to clear out high molecular weights and clippings.
- a detergent is class of molecules that have a hydrophobic chain. Examples include deoxycholate and sodium caprylate.
- the sample can be taken based on absorbance, for example from 0.5 OD A280 to 0.5 OD A280.
- the sample can also be fractionated, which refers to cutting the peak into different fractions based on volume. For example, fractions may be collected every 100 mis.
- An elution pool refers to a solution containing the substance that is eluted from a column or resin.
- the affinity column capture step and the CEX step both have an elution step in which the Fc-containing molecule (e.g. antibody) is removed from the column or resin in an elution buffer.
- an elution pool can comprise an elution buffer and an Fc- containing molecule.
- An elution pool is a sample, as used herein.
- a gradient elution refers to eluting the Fc-containing molecule with a buffer gradient.
- the gradient can start at 0% of 100 mM sodium acetate, pH 5.0, 500 mM sodium chloride, and 100 % of 100 mM sodium acetate, pH 5.0; and end at 65% of 100 mM sodium acetate, pH 5.0, 500 mM sodium chloride, and 100 % of 100 mM sodium acetate, pH 5.0.
- the rate is indicated as mM/CV.
- An Fc-containing molecule is a construct (or polypeptide(s)) that has an Fc region that is capable of binding Protein A.
- Non-limiting examples of Fc-containing molecules are antibodies and heterodimeric antibodies.
- the Fc-containing molecule is an antibody.
- antibody refers to a protein having a conventional immunoglobulin format, comprising heavy and light chains, and comprising variable and constant regions.
- an antibody may be an IgG which is a “Y-shaped” structure of two identical pairs of polypeptide chains, each pair having one “light” (typically having a molecular weight of about 25 kDa) and one “heavy” chain (typically having a molecular weight of about 50-70 kDa).
- An antibody has a variable region and a constant region.
- variable region is generally about 100- 110 or more amino acids, comprises three complementarity determining regions (CDRs), is primarily responsible for antigen recognition, and substantially varies among other antibodies that bind to different antigens.
- the constant region allows the antibody to recruit cells and molecules of the immune system.
- the variable region is made of the N-terminal regions of each light chain and heavy chain, while the constant region is made of the C-terminal portions of each of the heavy and light chains.
- the fragment crystallizable region (“Fc region”) is the end of the antibody that binds Fc receptors and proteins of the complement system.
- the IgG Fc region for example, comprises two CH2 and two CH3 domains.
- the Fc region binds with high affinity to Protein A.
- CDRs of antibodies have been described in the art. Briefly, in an antibody scaffold, the CDRs are embedded within a framework in the heavy and light chain variable region where they constitute the regions largely responsible for antigen binding and recognition.
- a variable region comprises at least three heavy or light chain CDRs (Kabat et al., 1991, Sequences of Proteins of Immunological Interest, Public Health Service N.I.H., Bethesda, Md.; see also Chothia and Lesk, 1987, J. Mol. Biol.
- framework region designated framework regions 1-4, FR1, FR2, FR3, and FR4, by Kabat et al., 1991; see also Chothia and Lesk, 1987, supra).
- Human light chains are classified as kappa and lambda light chains. Heavy chains are classified as mu, delta, gamma, alpha, or epsilon, and define the antibody's isotype as IgM, IgD, IgG, IgA, and IgE, respectively.
- IgG has several subclasses, including, but not limited to IgGl, IgG2, IgG3, and IgG4.
- IgM has subclasses, including, but not limited to, IgMl and IgM2.
- Embodiments of the invention include all such classes or isotypes of antibodies.
- the light chain constant region can be, for example, a kappa- or lambda-type light chain constant region, e.g., a human kappa- or lambda-type light chain constant region.
- the heavy chain constant region can be, for example, an alpha-, delta-, epsilon-, gamma-, or mu-type heavy chain constant regions, e.g., a human alpha-, delta-, epsilon-, gamma-, or mu-type heavy chain constant region.
- the antibody is an antibody of isotype IgA, IgD, IgE, IgG, or IgM, including any one of IgGl, IgG2, IgG3 or IgG4.
- IgGl antibodies are particularly susceptible to reduction of di-sulfide bonds and, as a result, represent one preferred embodiment of the disclosure.
- the antibody may be a monoclonal antibody or a polyclonal antibody.
- the antibody comprises a sequence that is substantially similar to a naturally- occurring antibody produced by a mammal, e.g., mouse, rabbit, goat, horse, chicken, hamster, human, and the like.
- the antibody may be considered as a mammalian antibody, e.g., a mouse antibody, rabbit antibody, goat antibody, horse antibody, chicken antibody, hamster antibody, human antibody, and the like.
- the monoclonal antibody is a human antibody.
- the monoclonal antibody is a chimeric antibody or a humanized antibody.
- chimeric antibody is used herein to refer to an antibody containing constant domains from one species and the variable domains from a second, or more generally, containing stretches of amino acid sequence from at least two species.
- humanized when used in relation to antibodies refers to antibodies having at least CDR regions from a non-human source which are engineered to have a structure and immunological function more similar to true human antibodies than the original source antibodies. For example, humanizing can involve grafting CDR from a non-human antibody, such as a mouse antibody, into a human antibody.
- Humanizing also can involve select amino acid substitutions to make a non-human sequence look more like a human sequence.
- the methods are not limited to the antigen-specificity of the antibody. Accordingly, the antibody has any binding specificity for virtually any antigen. In exemplary aspects, the antibody binds to a hormone, growth factor, cytokine, a cell-surface receptor, or any ligand thereof.
- the Fc-containing molecule is a heterodimeric antibody, which refer to antibodies comprising two different light chains and two different heavy chains.
- a hetero Ig comprises two Fabs and an Fc region.
- the two Fabs are each N-terminal to the Fc region.
- the two Fabs are each C-terminal to the Fc region.
- the heterodimeric antibodies can comprise any immunoglobulin constant region.
- the term “constant region” as used herein refers to all domains of an antibody other than the variable region.
- the constant region is not involved directly in binding of an antigen, but exhibits various effector functions.
- antibodies are divided into particular isotypes (IgA, IgD, IgE, IgG, and IgM) and subtypes (IgGl, IgG2, IgG3, IgG4, IgAl IgA2) depending on the amino acid sequence of the constant region of their heavy chains.
- the light chain constant region can be, for example, a kappa- or lambda-type light chain constant region, e.g., a human kappa- or lambda-type light chain constant region, which are found in all five antibody isotypes.
- the heavy chain constant region of the heterodimeric antibodies can be, for example, an alpha-, delta-, epsilon-, gamma-, or mu-type heavy chain constant region, e.g., a human alpha-, delta-, epsilon-, gamma-, or mu-type heavy chain constant region.
- the heterodimeric antibodies comprise a heavy chain constant region from an IgGl, IgG2, IgG3, or IgG4 immunoglobulin.
- DuoBody® An example of a heterodimeric antibody is a DuoBody®.
- DuoBodies can be made by the DuoBody® technology platform (Genmab A/S) as described, e.g., in International Publication Nos. WO 2008/119353, WO 2011/131746, WO 2011/147986, and WO 2013/060867, Labrijn A F et al., PNAS, 110(13): 5145-5150 (2013), Gramer et al., mAbs, 5(6): 962-973 (2013), and Labrijn et al., Nature Protocols, 9(10): 2450-2463 (2014).
- This technology can be used to combine one half of a first monospecific antibody containing two heavy and two light chains with one half of a second monospecific antibody containing two heavy and two light chains.
- the resultant heterodimer contains one heavy chain and one light chain from the first antibody paired with one heavy chain and one light chain from the second antibody.
- both of the monospecific antibodies recognize different epitopes on different antigens, the resultant heterodimer is a multispecific antibody.
- Another exemplary method of generating multispecific antibodies is by the knobs- into-holes technology (Ridgway et al., Protein Eng., 9:617-621 (1996); WO 2006/028936).
- the mispairing problem of Ig heavy chains that is a chief drawback for making multispecific antibodies is reduced in this technology by mutating selected amino acids forming the interface of the heavy chains in IgG.
- an amino acid with a small side chain (hole) is introduced into the sequence of one heavy chain and an amino acid with a large side chain (knob) into the counterpart interacting residue location on the other heavy chain.
- antibodies of the disclosure have immunoglobulin chains in which the heavy chains have been modified by mutating selected amino acids that interact at the interface between two polypeptides so as to preferentially form a multispecific antibody.
- the multispecific antibodies can be composed of immunoglobulin chains of the same subclass or different subclasses.
- CrossMab are chimeric antibodies constituted by the halves of two full-length antibodies. For correct chain pairing, it combines two technologies: (i) the knob-into-hole which favors a correct pairing between the two heavy chains; and (ii) an exchange between the heavy and light chains of one of the two Fabs to introduce an asymmetry which avoids light-chain mispairing. See, Ridgway et al., Protein Eng., 9:617-621 (1996); Schaefer et al., PNAS, 108: 11187-11192 (2011).
- CrossMabs can combine two or more antigen-binding domains for targeting two or more targets or for introducing bivalency towards one target such as the 2: 1 format.
- the Fc-containing molecule is a T cell engager molecule that has an Fc.
- a T cell engager (“TCE”) molecule comprises a polypeptide(s) that can bind to two different antigens, one of which is preferentially CD3.
- An example of a TCE molecule with an Fc is a BiTE® molecule with an Fc. Said Fc can be used to extend the half-life of the molecule.
- the antigen binding protein, antibody or therapeutic protein is one of those described in U.S. Pat. No. 7,947,809 and U.S. Patent Application Publication No.
- 20100255538 IGF-1 receptor
- U.S. Pat. No. 7,868,140 B7RP1
- U.S. Pat. No. 7,807,159 and U.S. Patent Application Publication No. 20110091455 myostatin
- U.S. Pat. Nos. 7,736,644, 7,628,986, 7,524,496, and U.S. Patent Application Publication No. 20100111979 dotion mutants of epidermal growth factor receptor
- U.S. Pat. No. 7,728,110 SARS coronavirus
- U.S. Pat. No. 7,718,776 and U.S. Patent Application Publication No. 20100209435 OPGL
- 20110040076 (NGF), U.S. Pat. No. 7,579,186 (TGF-P type II receptor), U.S. Pat. No. 7,541,438 (connective tissue growth factor), U.S. Pat. No. 7,438,910 (IL1-R1), U.S. Pat. No. 7,423,128 (properdin), U.S. Pat. Nos. 7,411,057, 7,824,679, 7,109,003, 6,682,736, 7,132,281, and
- 20080182976 (erythropoietin receptor), U.S. Patent Application Publication No. 20080166352 and U.S. Pat. No. 7,435,796 (B7RP1), U.S. Pat. No. 7,423,128 (properdin), U.S. Pat. Nos. 7,422,742 and 7,141,653 (interleukin-5), U.S. Pat. Nos. 6,740,522 and 7,411,050 (RANKL), U.S. Pat. No. 7,378,091 (carbonic anhydrase IX (CA IX) tumor antigen), U.S. Pat. Nos. 7,318,925 and 7,288,253 (parathyroid hormone), U.S. Pat. No.
- variable domain polypeptides variable domain encoding nucleic acids
- host cells vectors
- methods of making polypeptides encoding said variable domains pharmaceutical compositions, and methods of treating diseases associated with the respective target of the variable domain-containing antigen binding protein or antibody.
- the antibody is selected from the group consisting of Murom onab-CD3 (product marketed with the brand name Orthoclone Okt3®), Abciximab (product marketed with the brand name Reopro®.), Rituximab (product marketed with the brand name Mab Thera®, Rituxan®) (U.S. Pat. No.
- Basiliximab product marketed with the brand name Simulect®
- Daclizumab product marketed with the brand name Zenapax®
- Palivizumab product marketed with the brand name Synagis®
- Infliximab product marketed with the brand name Remicade®
- Trastuzumab product marketed with the brand name Herceptin®
- Alemtuzumab product marketed with the brand name MabCampath®, Campath- 1H®
- Adalimumab product marketed with the brand name Humira®
- Tositumomab-1131 product marketed with the brand name Bexxar®
- Efalizumab product marketed with the brand name Raptiva®
- Cetuximab product marketed with the brand name Erbitux®
- Flbritumomab tiuxetan product marketed with the brand name Zevalin®
- I'Omalizumab product marketed with the brand name Xol
- the antibody is one of anti-TNF alpha antibodies such as adalimumab, infliximab, etanercept, golimumab, and certolizumab pegol; anti-ILl.beta. antibodies such as canakinumab; anti-IL12/23 (p40) antibodies such as ustekinumab and briakinumab; and anti-IL2R antibodies, such as daclizumab.
- anti-TNF alpha antibodies such as adalimumab, infliximab, etanercept, golimumab, and certolizumab pegol
- anti-ILl.beta. antibodies such as canakinumab
- anti-IL12/23 (p40) antibodies such as ustekinumab and briakinumab
- anti-IL2R antibodies such as daclizumab.
- anti-cancer antibodies include, but are not limited to, anti- CCR8 antibodies, anti-BAFF antibodies such as belimumab; anti-CD20 antibodies such as rituximab; anti-CD22 antibodies such as epratuzumab; anti-CD25 antibodies such as daclizumab; anti-CD30 antibodies such as iratumumab, anti-CD33 antibodies such as gemtuzumab, anti- CD52 antibodies such as alemtuzumab; anti-CD152 antibodies such as ipilimumab; anti-EGFR antibodies such as cetuximab; anti-HER2 antibodies such as trastuzumab and pertuzumab; anti- IL6 antibodies such as siltuximab; and anti-VEGF antibodies such as bevacizumab; anti-IL6 receptor antibodies such as tocilizumab.
- anti-BAFF antibodies such as belimumab
- anti-CD20 antibodies such as rituximab
- anti-CD22 antibodies such
- a reduction in yellow color can be determined for a sample if the b* value or the amount of yellow color visually observed (by way of examples) in the sample is less using a wash of the present invention compared to a sample using another wash (e.g. a wash not of the present invention) or a sample which has not been subjected to any wash.
- a reduction in the amount of impurity for a sample can be determined by comparing the amount of impurity in the sample using a wash of the present invention compared to the amount of impurity in a sample using another wash (e.g. a wash not of the present invention) or a sample which has not been subjected to any wash.
- HCCF Antibody 1 harvested cell culture fluid
- a deoxycholate wash was step (25 mM Tris, 20 mM Sodium Deoxycholate, 100 mM NaCl, pH 8.2; 10 CV volume; 250 cm/hr flow rate) or a sodium caprylate step (25 mM Tris, 100 mM Sodium Caprylate, 2.5 M NaCl, pH 7.4; 10 CV volume; 250 cm/hr flow rate) was performed.
- a deoxycholate or caprylate wash was performed, subsequent washes, elution, cleaning, and flush step(s) were performed prior to storage of the Fc- containing molecule.
- Control samples are generated from the same steps described below except there is no deoxycholate or caprylate wash step or the following wash step of 25 mM tris, lOOmM sodium chloride, pH 7.4.
- the protein A pool is collected from 0.5 OD A280 to 0.5 OD A280.
- the HunterLab UltraScan PRO spectrophotometer was used for the yellow color measurement in various Antibody 1 samples.
- the three color primaries measured across the visible wavelength range in a sample is mathematically converted by the instrument to the Opponent color scale values (CIELAB) of L*, a*, and b*.
- the L* is the degree of lightness to darkness
- a* is the degree of redness to greenness
- b* is the degree of yellowness to blueness. The higher value of b* suggests that the sample is more yellow.
- HCCF Antibody 1 harvested cell culture fluid
- the purification method utilized Cytiva MabSelect SuRe resin. Protein A pools were collected and neutralized to pH 5.0, and samples were taken for analysis of 303 Da species. The procedure was performed essentially as described in Example 1, however both a deoxy cholate wash step and a caprylate wash step were used.
- a deoxycholate wash was step (25 mM Tris, 20 mM Sodium Deoxycholate, 100 mM NaCl, pH 8.2; 10 CV volume; 250 cm/hr flow rate) and a sodium caprylate wash step (25 mM Tris, 100 mM Sodium Caprylate, 2.5 M NaCl, pH 7.4; 10 CV volume; 250 cm/hr flow rate) were performed, separated by a wash step of 25 mM tris, 100 mM sodium chloride (between the sodium deoxycholate and sodium caprylate wash steps).
- a wash step 25 mM Tris, 20 mM Sodium Deoxycholate, 100 mM NaCl, pH 8.2; 10 CV volume; 250 cm/hr flow rate
- a sodium caprylate wash step 25 mM Tris, 100 mM Sodium Caprylate, 2.5 M NaCl, pH 7.4; 10 CV volume; 250 cm/hr flow rate
- LC-UV-FD-MS liquid chromatography with ultraviolet, fluorescence, and mass spectrometric detection
- LC-UV-FD-MS liquid chromatography with ultraviolet, fluorescence, and mass spectrometric detection
- QE-HF Thermo Fisher Scientific Q-Exactive HF
- Orbitrap Eclipse high resolution mass spectrometer Samples were analyzed using a reversed phase column, Agilent Diphenyl RRHD (100x2.1mm, 1.8pm) at 60°C, using acetonitrile gradient at a flow rate of 0.3mL/min. Mobile phases contained 0.1% (v/v) formic acid.
- UV data was collected at wavelengths of 230nm, 280nm, 325nm, 365nm and 400nm. Fluorescence detector was set at an excitation wavelength of 365nm and an emission wavelength of 510nm with PMT gain 2.
- AGC automatic gain control
- HCD data-dependent higher-energy collision dissociation
- While Eclipse was set up to collect data with variable collision energy (20- 60%). For analysis, 35pg of protein or 50pL of wash fractions or dilute samples were typically analyzed.
- the 303 Da species was found on both samples taken from wash step and elution pool of the process in which deoxycholate and caprylate wash steps were not implemented.
- the analytical assay revealed that the 303 Da species was mainly contained in the deoxycholate wash step and not detected in the elution pool.
- the caprylate step also contained some 303 Da species, however due to the order of the steps the levels of 303 Da species in the caprylate step were very low compared to the level of 303 Da species found in the deoxycholate step. Therefore, it can be concluded that the detergent wash steps were very effective in clearing 303 Da species from elution pool during affinity capture step.
- CEX step is often leveraged as the main polishing step for many antibodies purification due to its ability to separate impurities based on charges.
- a modified CEX step has been implemented to reduce yellow color in antibody elution pool (e.g. drug substance intermediate).
- the separation power of CaptoSP ImpRes was combined with the ability of the AKTA to trace UV absorbance at 400 nm and 420 nm (wavelength absorbed by objects exhibiting yellow color), to show that yellow color impurity can be detected and fractionated using a CEX step of the present invention.
- CEX method leveraged the CaptoSP ImpRes resin from Cytiva.
- CEX was performed with steps of pre-equilibration, equilibration, loading, wash, elution, cleaning (non-denaturing and denaturing), and storage.
- a gradient elution was used, wherein the gradient started at 0% B and 100% A and ended at 65% B and 35% A, at a rate of 8 mM/CV.
- UV absorbance at wavelength 400 nm and 420 nm were tracked to monitor yellow color species.
- the elution pool was fractionated at 0.5 CV. Samples and controls were then submitted for HunterLab color assay and LC-MS assay for measurement of 303 Da species as described above.
- EXAMPLE 4 Developing sodium chloride salt wash during CEX polishing step [0076] Sodium chloride salt washes were further developed to provide better clearance of the yellow color species. The salt condition had to be adjusted to match run conditions, such as pH and loading levels.
- the buffer was 100 mM Acetate and either 65, 75, or 85 mM NaCl, pH 5.0; 3 CV; flow rate 140 cm/hr.
- CEX was performed with steps of pre-equilibration, equilibration, loading, wash (100 mM Sodium Acetate, pH 5.0), sodium chloride salt wash (100 mM Acetate and either 65, 75, or 85 mM NaCl, pH 5.0; 3 CV; flow rate 140 cm/hr), wash (100 mM Sodium Acetate, pH 5.0), elution, cleaning (non-denaturing and denaturing), and storage. Samples were taken after each step.
- Figure 4 shows the assay result from various samples taken during a CEX step without the sodium chloride salt wash, and a CEX step with the sodium chloride salt wash. Samples were taken after each step. The 303 Da species was present in the CEX elution pool without the sodium chloride salt wash, with a slightly higher concentration found during the first elution fraction. There was a higher concentration of 303 Da species found in the wash step samples in the CEX method using a sodium chloride salt wash step, and although it was still present in the elution pool of the proposed CEX method, the level was very low. Further processing through a third column (flowthrough MMC) cleared the 303 Da species to lower than assay detection level.
- flowthrough MMC cleared the 303 Da species to lower than assay detection level.
- Figure 5 and Table 3 demonstrate data on relative level of 303 Da species, obtained via integration of peak on LC-UV-FD-MS assay, in various Antibody 1 and Antibody 2 sodium chloride salt wash samples. Based on the data, the 75 mM sodium chloride salt wash is effective in reducing the level of 303 Da species in both Antibody 1 and Antibody 2 while retaining acceptable antibody yield. The data also shows that Antibody 2 has lower level of 303 Da species to begin with compared to Antibody 1.
Landscapes
- Chemical & Material Sciences (AREA)
- Analytical Chemistry (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Molecular Biology (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Life Sciences & Earth Sciences (AREA)
- Biophysics (AREA)
- Genetics & Genomics (AREA)
- Medicinal Chemistry (AREA)
- General Health & Medical Sciences (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Biochemistry (AREA)
- Immunology (AREA)
- Peptides Or Proteins (AREA)
Abstract
The present invention provides methods of reducing yellow color in an Fc-containing molecule sample, such as in an elution pool. Said methods are useful in manufacturing of therapeutic antibodies.
Description
METHODS FOR REDUCING YELLOW COLOR
FIELD OF THE INVENTION
[0001] The present invention relates to methods of reducing yellow color and/or impurities during purification of constructs containing an Fc. In particular, the methods of the present invention relate to purification of Fc-containing molecules, such as antibodies.
BACKGROUND OF THE INVENTION
[0002] Color is an important quality attribute for therapeutic drug substance. The color of a monoclonal antibody (mAb) drug substance can vary due to multiple reasons, such as association with cell culture media component or stress-induced chemical modification of amino acids in the mAb sequence (Ambrogelly, Antibodies 2021, 10(2), 21). The ability to control and/or reduce color-causing species, without losing potency, during downstream purification is important to manufacturing drug substance for patients.
SUMMARY OF THE INVENTION
[0003] The present invention provides a method of reducing yellow color in an Fc- containing molecule sample. In an embodiment, the method comprises capture affinity, wherein the capture affinity comprises a detergent wash that is sodium deoxycholate and/or sodium caprylate. In an embodiment, the capture affinity comprises sodium deoxycholate. In an embodiment, the capture affinity comprises sodium caprylate. In an embodiment, the capture affinity comprises a sodium deoxycholate step and a sodium caprylate step. In an embodiment, the deoxycholate wash comprises about 10 mM to about 30 mM sodium deoxycholate. In an embodiment, the deoxy cholate wash comprises about 15 mM to about 25 mM sodium deoxycholate. In an embodiment, the deoxycholate wash comprises about 10 mM, 11 mM, 12 mM, 13 mM, 14 mM, 15 mM, 16 mM, 17 mM, 18 mM, 19 mM, 20 mM, 21 mM, 22 mM, 23 mM, 24 mM, 25 mM, 26 mM, 27 mM, 28 mM, 29 mM, or 30 mM. In an embodiment, the deoxycholate wash comprises about 20 mM sodium deoxycholate. In an embodiment, the deoxycholate wash comprises 20 mM sodium deoxycholate. In an embodiment, the deoxycholate wash comprises about 15 to about 35 mM Tris, about 10 to about 30 mM sodium deoxy cholate, and about 90 to about 110 mM NaCl. In an embodiment, the deoxy cholate wash comprises about 25 mM Tris, about 20 mM sodium deoxycholate, and about 100 mM NaCl. In an embodiment, the deoxycholate wash comprises 25 mM Tris, 20 mM sodium deoxycholate,
and 100 mM NaCl. In an embodiment, the deoxycholate wash is at about pH 8.0-8.4. In an embodiment, the deoxycholate wash is at pH 8.2.
[0004] In an embodiment, the sodium caprylate wash comprises about 80 mM to about 120 mM sodium caprylate. In an embodiment, the sodium caprylate wash comprises about 90 mM to about 110 mM sodium caprylate. In an embodiment, the sodium caprylate wash comprises about 95 mM to about 105 mM sodium caprylate. In an embodiment, the sodium caprylate wash comprises about 100 mM sodium caprylate. In an embodiment, the sodium caprylate wash comprises 100 mM sodium caprylate. In an embodiment, the sodium caprylate wash comprises about 15 to about 35 mM Tris, about 90 to about 110 mM sodium caprylate and about 2 to about 3 M NaCl. In an embodiment, the sodium caprylate wash comprises about 25 mM Tris, about 100 mM sodium caprylate and about 2.5 M NaCl. In an embodiment, the sodium caprylate wash comprises 25 mM Tris, 100 mM sodium caprylate and 2.5 M NaCl. In a particular embodiment, the sodium caprylate wash is at about pH 7.2 to about 7.6. In a particular embodiment, the sodium caprylate wash is at pH 7.4. In an embodiment, the deoxycholate wash is performed prior to the caprylate wash. In an embodiment, the caprylate wash is performed prior to the deoxycholate wash.
[0005] The present invention provides a method of detecting yellow color in an Fc- containing molecule sample, wherein said method is a cation exchange (CEX) method comprising a strong cation exchange resin and detecting UV absorbance at 400 nm and 420 nm. In an embodiment, the CEX method comprises a gradient elution. In an embodiment, the gradient elution is from about 0% of 100 mM sodium acetate, pH 5.0, 500 mM sodium chloride, and about 100 % of 100 mM sodium acetate, pH 5.0; to about 65% of 100 mM sodium acetate, pH 5.0, 500 mM sodium chloride, and about 100 % of 100 mM sodium acetate, pH 5.0. In an embodiment, the gradient elution is from 0% of 100 mM sodium acetate, pH 5.0, 500 mM sodium chloride, and 100 % of 100 mM sodium acetate, pH 5.0; to 65% of 100 mM sodium acetate, pH 5.0, 500 mM sodium chloride, and 100 % of 100 mM sodium acetate, pH 5.0. In an embodiment, the gradient elution is performed at a rate of 8 mM/CV.
[0006] The present invention provides a method of reducing yellow color in an Fc- containing molecule sample, wherein said method is a CEX method comprising a strong cation exchange resin and a sodium chloride salt wash. In an embodiment, the sodium chloride salt wash is about 60 mM to about 90 mM sodium chloride. In an embodiment, the sodium chloride salt wash is about 65 mM to about 85 mM sodium chloride. In an embodiment, the sodium chloride salt wash is about 75 mM sodium chloride. In an embodiment, the sodium chloride salt wash is 75 mM sodium chloride. In an embodiment, the sodium chloride salt wash is 100 mM sodium acetate, pH 5.0, and 75 mM sodium chloride. In an embodiment, the method comprises a
gradient elution. In an embodiment, the gradient elution is from: 0% of 100 mM sodium acetate, pH 5.0, 500 mM sodium chloride, and 100 % of 100 mM sodium acetate, pH 5.0; to 65% of 100 mM sodium acetate, pH 5.0, 500 mM sodium chloride, and 100 % of 100 mM sodium acetate, pH 5.0.
[0007] The present invention provides a method of reducing yellow color in an Fc- containing molecule sample, comprising a combination of any of the methods described herein. In an embodiment, the method comprises a method comprising capture affinity, wherein the capture affinity comprises a detergent wash that is sodium deoxycholate and/or sodium caprylate, and a method comprising a CEX method comprising a strong cation exchange resin and a sodium chloride salt wash. In an embodiment, the capture affinity comprises sodium deoxycholate. In an embodiment, the capture affinity comprises sodium caprylate. In an embodiment, the capture affinity comprises a sodium deoxycholate step and a sodium caprylate step. In an embodiment, the deoxycholate wash comprises 20 mM sodium deoxycholate. In an embodiment, the deoxycholate wash comprises 25 mM Tris, 20 mM sodium deoxycholate, and 100 mM NaCl. In a particular embodiment, the deoxycholate wash is at about pH 8.2. In an embodiment, the sodium caprylate wash comprises 100 mM sodium caprylate. In an embodiment, the sodium caprylate wash comprises 25 mM Tris, 100 mM sodium caprylate and 2.5 M NaCl. In a particular embodiment, the sodium caprylate wash is at about pH 7.4. In an embodiment, the deoxycholate wash is performed prior to the caprylate wash. In an embodiment, the caprylate wash is performed prior to the deoxycholate wash. In an embodiment, the sodium chloride salt wash is 75 mM sodium chloride. In an embodiment, the sodium chloride salt wash is 100 mM sodium acetate, pH 5.0, and 75 mM sodium chloride. In an embodiment, the method comprises a gradient elution. In an embodiment, the gradient elution is from: 0% of 100 mM sodium acetate, pH 5.0, 500 mM sodium chloride, and 100 % of 100 mM sodium acetate, pH 5.0; to 65% of 100 mM sodium acetate, pH 5.0, 500 mM sodium chloride, and 100 % of 100 mM sodium acetate, pH 5.0.
[0008] The present invention also provides a method of reducing an impurity in an Fc- containing molecule sample, wherein said method comprises a method of the present invention. In an embodiment, the impurity is detected by liquid chromatography with ultraviolet, fluorescence, and mass spectrometric detection. In an embodiment, the impurity is a 303 Da species.
[0009] In an embodiment, the Fc-containing molecule sample is a drug substance intermediate.
[0010] In an embodiment, the method is performed during purification of the Fc- containing molecule.
[0011] In an embodiment, the Fc-containing molecule is an antibody. In an embodiment, the Fc-containing molecule is a heterodimeric antibody that has an Fc.
[0012] In an embodiment, the yellow color or the impurity in a sample is reduced compared to yellow color or impurity in a sample that has been subjected to another wash or has not been subjected to any wash. In an embodiment, the yellow color or impurity is reduced by at least 3%. In an embodiment, the yellow color or impurity is reduced by at least 5%. In an embodiment, the yellow color or impurity is reduced by at least 10%. In an embodiment, the yellow color or impurity is reduced by at least 15%. In an embodiment, the yellow color or impurity is reduced by at least 20%. In an embodiment, the yellow color or impurity is reduced by at least 25%. In an embodiment, the yellow color or impurity is reduced by at least 30%. In an embodiment, the yellow color or impurity is reduced by at least 35%. In an embodiment, the yellow color or impurity is reduced by at least 40%. In an embodiment, the yellow color or impurity is reduced by at least 45%. In an embodiment, the yellow color or impurity is reduced by at least 50%. In an embodiment, the yellow color is reduced. In an embodiment, the impurity is reduced. In an embodiment, the yellow color is determined by the calculated b* value of the Fc-containing molecule sample. In an embodiment, the yellow color is determined by calculating the b* value of the Fc-containing molecule sample and comparing the b* value with a b* value of a sample of an Fc-containing molecule that has not been subjected to a wash (method) of the present invention. In an embodiment, the yellow color is determined by visual observation of the sample. In an embodiment, the yellow color is determined by visual observation of the Fc- containing molecule sample to obtain a visually observed yellow color, and comparing the visually observed yellow color with a visually observed yellow color of a sample of an Fc- containing molecule that has not been subjected to a wash (method) of the present invention.
[0013] In an embodiment, the sample is an elution pool.
BRIEF DESCRIPTION OF THE DRAWINGS
[0014] Figure 1. HunterLab Assay yellow color measurement on samples with and without detergent washes during affinity capture step. Figure 1 shows the result of HunterLab assay on up-concentrated neutralized ProA Pool samples or controls.
[0015] Figure 2. LC-UV-FD-MS measurement of 303 Da species on samples with and without detergent washes during affinity capture step. A. Control wash step (no detergent wash). B. Control elution pool (no detergent wash). C and D, detergent wash step and elution pool of
proposed method. Figure 2 shows measurement of 303 Da species on samples with and without detergent washes during affinity capture step.
[0016] Figure 3. UV absorbance at 280 nm, 400 nm, and 420 nm during CEX elution step. The dotted boxes denote fractions. The elution profile revealed higher A400 and A420 absorbances during the first two fractions, which correlates to visibly yellower fractions.
[0017] Figure 4. Hunterlab assay result from various salt wash samples as compared to yellow color standards (Y7-T4).
[0018] Figure 5. LC-UV-FD-MS results for detection of 303 Da species. A and B are 1st elution fraction and elution pool, respectively, using a CEX method without the sodium chloride salt wash step. C and D are wash sample and elution pool, respectively, of the CEX method with a sodium chloride salt wash step. E is the result from elution pool from an additional column step.
[0019] Figure 6. Level of 303 Da species in various sodium chloride salt wash samples of Antibody 1 and Antibody 2. The peak area was obtained by integration of 303 Da peak on the LC-UV-FD-MS chromatogram and reflects the relative level of 303 Da species in each sample. The sodium chloride salt washes are effective in reducing the level of 303 Da species in CEX pool samples. Antibody 2 has lower level of 303 Da species compared to Antibody 1.
DETAILED DESCRIPTION
[0020] During therapeutic monoclonal antibody (mAb) manufacturing process, Protein A affinity capture step is often the first critical step of purification, specifically designed to separate Fc-containing molecule from cell culture media components and other impurities present in the harvest material. The present invention describes the inclusion of a wash step using various salts, such as sodium caprylate and sodium deoxycholate, has been found to help with further removal of host cell protein (HCP) and DNA.
[0021] During the molecular assessment step of Antibody 1, it was discovered that the drug substance and intermediate pools were elevated in yellow color. Further investigation revealed that the yellow color can be reduced through modifications of two purification steps. The first step involved introducing detergent washes to capture affinity purification. The capture affinity purification is often the first critical step of therapeutic monoclonal antibody (mAb) purification process. It is designed to separate Fc-containing molecule from cell culture media components and other impurities present in the harvest material. The addition of detergent washes can disrupt non-covalent binding to further improve separation between Fc-containing
molecules and impurities. Two detergents, sodium caprylate (Hu et al., Protein Expr Purif. 2021 Oct;186: 105907) and sodium deoxycholate can remove host cell proteins including endogenous protease(s). The present invention discovered that the implementation of detergent wash(es) during Protein A step (capture affinity) resulted in a sample (e.g. elution pool) with a reduction in yellow color compared to a sample that has been subjected to a different wash or has not been subjected to any wash. The detergent washes also resulted in a reduction of an impurity present in cell culture.
[0022] Affinity chromatography (e.g. Protein A) is widely employed for the preparative purification of proteins possessing a heavy chain Fc region comprising the CH2 and CH3 domains of an immunoglobulin molecule. (Vola et al. Cell Biophys. 24-25: 27-36, 1994; Gagnon, Protein A affinity chromatography, In: Purification tools for monoclonal antibodies, 1996, Validated Biosystems, Tucson, Ariz., 1996; Aybay and Imir, J. Immunol. Methods 233(1-2): 77- 81, 2000; Ford et al., J. Chromatogr. B 754: 427-435, 2001; Fahrner et al, Biotechnology and Genetic Engineering News, 18: 301-327, 2001). Such Fc proteins include antibodies, particularly monoclonal antibodies, as well as Fc fusion proteins. Such proteins are typically produced by mammalian or bacterial cells engineered to express the desired recombinant protein, intracellularly or directly, into the culture medium or intracellularly. Purification of the expressed proteins typically begins with either collecting the cell culture medium to harvest extracellularly expressed protein or harvesting and lysing the host cells to release intracellularly expressed protein. This host cell “harvest medium” contains not only the protein of interest but also DNA, RNA, and protein contaminants from the host cell that must be separated from the desired protein. These host cell contaminants may be separated by various chromatographic methods based on their charge, size or hydrophobicity.
[0023] The affinity of immunoglobulin heavy chain Fc regions for the IgG binding domains of Protein A allows for the direct capture and purification of such Fc proteins from complex host cell harvest medium that may contain many different host cell contaminants. Despite the high specificity of the Fc region for Protein A, host cell protein contaminants may still be present at varying levels in the final column eluate, thereby reducing the purity of the of the final protein product. To be useful for human therapeutic purposes, protein products must be separated from all of the extraneous media components and cell by-products, creating a need for purification methods that can maintain product yield while reducing host cell contaminant levels. Traditionally, affinity chromatography includes the steps of binding the Fc-containing molecule to the immobilized ligand (e.g. Protein A), washing away non-bound components, and eluting the Fc-containing molecule by altering the buffer conditions so that Fc-containing molecule no longer binds the immobilized ligand.
[0024] Cation exchange chromatography is a form of ion exchange chromatography, which is used to separate molecules based on their net surface charge. Cation exchange chromatography, more specifically, uses a negatively charged ion exchange resin with an affinity for molecules having net positive surface charges.
[0025] Another way to reduce yellow color was by introducing a sodium chloride salt wash during cation exchange (CEX) polishing step. Since visible yellow color is associated with light absorbance around 400 nm, the AKTA UV detector was used to measure light absorbance at 400nm and 420 nm during a CEX run. The resulting chromatogram revealed that the yellow color species was asymmetrically distributed during the CEX elution step, with higher concentrations found at the beginning of the elution. Interestingly, the earlier fractions were also visibly more yellow. Based on this finding, a sodium chloride salt wash step was then developed to separate early fractions from the rest of the elution peak, which was shown to successfully reduce yellow color in the CEX elution pool. Up-concentration of fractions containing yellow color species resulted in a sample that had more intense yellow color than the concentrated control sample, while exclusion of these fractions resulted in a sample that was less yellow than the control sample. The dedicated yellow color fractions, being concentrated in the earlier more acidic portions of the CEX elution, are removed using a dedicated sodium chloride salt wash.
[0026] A 303 Da species was identified to be elevated in earlier CEX elution fractions, which are also yellow in color. The 303 Da species and yellow color appear to co-elute during the earlier fractions of the CEX elution. This species was also reduced with the addition of the salt wash step.
[0027] Techniques for Protein A chromatography are known in the art (e.g., Shukla et al., J Chromatogr A. 2007 Nov. 9; 1171 (1 -2):22-8; U.S. Pat. No. 8,263,750; Rodriguez et al., J Chromatogr B Analyt Technol Biomed Life Sci. 2020 Nov 10; 1157: 122332), and the process is routinely used to remove contaminants such as host cell protein, DNA, and viruses from a solution comprising antigen-binding protein molecules with an Fc region (Fc-containing molecules) based on the affinity of Protein A for the Fc and/or Fab region of immunoglobulins. In some embodiments, a neutral or basic loading buffer (such as pH 7 to 8) is used to bind the antigen-binding protein onto the Protein A resin. In some embodiments, low pH is used to elute the antigen-binding protein from the Protein A resin, such as a pH between 3 and 5, such as 3 to 4, or 4 to 5. It is envisioned that other resins or columns, such as Protein G or Protein A/G, are also encompassed in the present invention.
[0028] A chromatography column may be used for any of many different liquid chromatographic processes, including ion-exchange chromatography, size-exclusion chromatography, hydrophobic interaction chromatography, and affinity chromatography. In ion-
exchange chromatography, an Fc-containing molecule is separated from a complex solution or mixture based on electrostatic forces between charged functional groups of Fc-containing molecules and charged functional groups of the chromatography-column matrix. Cationexchange resins have negatively charged functional groups that attract positively charged functional groups of Fc-containing molecules, and anion-exchange resins have positively charged functional groups that attract negatively charged functional groups of Fc-containing molecules. Molecules bound through electrostatic forces to the matrix can be eluted by increasing the ionic strength of the buffer solution within the chromatography column over time. In size-exclusion chromatography, an Fc-containing molecule is separated from a complex solution or mixture based on the Fc-containing molecule's size-related exclusion from the interior regions of spherical beads that make up the matrix. Progress through the chromatography column of smaller molecules that are capable of diffusing into the beads is slowed with respect to the Fc-containing molecule. In hydrophobic interaction chromatography, an Fc-containing molecule is separated from a complex solution or mixture based on the hydrophobicity of the Fc-containing molecule. A complex solution containing the Fc-containing molecule is applied to a chromatography column equilibrated with a high salt buffer that facilitates binding of the Fc-containing molecule to the resin. A salt-gradient mobile phase with decreasing ionic strength is then introduced into the chromatography column to release bound Fc-containing molecules from the matrix. Alternatively, hydrophobic interaction chromatography may separate a monomeric Fc-containing molecule from a complex solution or mixture by binding hydrophobic impurities, including inactive dimers and aggregates of the Fc-containing molecule, while permitting monomeric Fc- containing molecules to flow through the chromatography column relatively unimpeded. In affinity chromatography, an Fc-containing molecule, such as an antibody, is separated from a complex solution based on the affinity of the Fc-containing molecule for a ligand or ligandbinding entity that is covalently bound to the matrix. Molecules in the complex solution or mixture with weak affinity, or lacking affinity, for the ligand or ligand-binding entity flow through the chromatography column unimpeded, leaving the Fc-containing molecule bound to the matrix. The Fc-containing molecule can then be eluted from the chromatography column by altering buffer conditions to decrease the affinity of the Fc-containing molecule for the ligand or ligand-binding entity.
[0029] Protein A is a ~41 kDa protein from Staphylococcus aureas that binds with high affinity (~10— 8 M-10-12 M to human IgG) to the CH2/CH3 domain of the Fc regions of antibodies and is therefore commonly immobilized within an affinity-chromatography matrix for purifying target antibodies. Due to the biochemical properties of protein A, including a lack of disulfide bond linkages, protein A is very stable and can be used with high salt conditions and/or
denaturants, such as 10 M urea, 6 M guanidine, and 80 mM dithiothreitol. Protein-A affinity chromatography is often used for purification of monoclonal antibodies and fusion proteins containing the antibody constant fragment Fc. About 98% of process impurities, including viral particles, can be removed by protein-A affinity column chromatography in a single step, with high product yields.
[0030] There are many commercially available protein-A affinity chromatography resins that may be used for antibody purification, including ProSep® controlled-pore glass resins produced by Millipore and Mab Select™, cross-linked agarose resin products produced by Amersham Biosciences, and other types of protein-A affinity chromatography resins, including gel-based resins and silica-based resins. The different types of protein-A affinity chromatography resins have different characteristics and properties, and chromatography columns prepared with a particular type of protein-A affinity chromatography resin may be more suitable than chromatography columns prepared with other types of protein-A affinity chromatography resins for purification of particular Fc-containing molecules under particular conditions.
[0031] Protein-A affinity column chromatography may be used as a first step, sometimes referred to as an initial purification step, or “capture step,” of a multi-step purification process. An example of a multi-step purification process includes a capture step and one or more polishing steps. The capture step may include adsorbing the protein from a complex solution or complex mixture to protein A immobilized on a solid phase, removing contaminants by washing the solid phase with a wash buffer, and recovering the Fc-containing molecule from the solid phase with an elution buffer. The elution buffer is designed to introduce conditions that interfere with protein- A/Fc binding, such as lowering the pH within the column matrix to a point at which Fc no longer binds to protein A. The capture step may concentrate the Fc-containing molecule. The effectiveness of the capture step may be assessed based on the speed of the chromatography process, the degree of separation of the Fc-containing molecule from undesired solutes and suspended entities, and the load capacity of the chromatography column. Chromatography resins with high load capacities and good flow properties may be particularly well suited for capture steps.
[0032] Suitable buffers for the equilibration and wash steps include tris and sodium chloride, pH 7.4. Acetate can be used for the elution step.
[0033] The term “about” as used herein is mean to include values within 10% of the stated value, both higher and lower than the stated value. For example, reference to “about 90 mM” includes values of 81 mM to 99 mM, inclusive.
[0034] A range, such as, for example, a sodium caprylate wash comprising about 90 mM to about 110 mM sodium caprylate, is intended to include all values in between and including the lowest value (e.g. 90 mM) and the highest value (e.g. 110 mM).
[0035] The polishing steps may separate the Fc-containing molecule that has been concentrated and partially purified in the capture step from remaining impurities. The polishing steps may be useful for removing impurities that have relatively closely related chromatographic properties and that eluted together with the Fc-containing molecule during the capture step. Flow rates and load volumes of the polishing steps may be decreased in order to improve resolution. The polishing steps may be useful for removing product-related contaminants, such as aggregates, and trace amounts of process-related contaminants, such as viral particles, from the Fc-containing molecule-containing eluate, as well as for adjusting buffer conditions in preparation for subsequent processing steps.
[0036] Ion exchange, including cation exchange chromatography (CEX), is well known in the art (Cummins et al. (2017); in: Walls, D., Loughran, S. (eds) Protein Chromatography. Methods in Molecular Biology, vol 1485; Mohammed JS; A brief review on Ion Exchange Chromatography; PharmaTutor; 2017; 5(2); 30-38; Ullah, H. (2012); inTech. Doi: 10.5772/52537). Purification using ion exchange chromatography depends upon the reversible adsorption of charged solute molecules to immobilized ion exchange groups of opposite charge. Generally, an ion exchanger is brought to a starting state, in terms of pH and ionic strength, which allows the binding of the desired solute molecules. The exchanger groups are associated at this time with exchangeable counter-ions (usually simple anions or cations, such as chloride or sodium). The solute molecules carrying the appropriate charge then displace counter-ions and bind reversibly to the gel. Unbound substances can be washed out from the exchanger bed using starting buffer. Substances are removed from the column by changing to elution conditions unfavorable for ionic bonding of the solute molecules. This may involve increasing the ionic strength of the eluting buffer or changing its pH. Substances not eluted under the previous experimental conditions are removed from the column and are re-equilibrated at the starting conditions for the next purification. Separation is obtained since different substances have different degrees of interaction with the ion exchanger due to differences in their charges, charge densities and distribution of charge on their surfaces. These interactions can be controlled by varying conditions such as ionic strength and pH. Negatively charged exchangers have positively charged counter-ions (cations) and are termed cation exchangers.
[0037] Yellow color of a sample can be determined according to methods known in the art (see e.g. Ambrogelly A. The Different Colors of mAbs in Solution. Antibodies. 2021; 10(2):21).
[0038] The HunterLab UltraScan PRO spectrophotometer, which measures color expressed in the three dimensions of the human color response (ie, red, green and blue) rather than spectral colors (ie, wavelengths) by a physical detector in the visible wavelength range, can be used for the yellow color measurement in antibody samples. In an embodiment, the reduction in yellow color as part of the present invention is 1% through 100%. In an embodiment, the reduction in yellow color is 1%, 5%, 10%, 15%, 20%, 25%, 30%, 35%, 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 98%, 99%, or 100%. In an embodiment, the yellow color is determined by HunterLab assay.
[0039] In an embodiment, yellow color of a sample can be determined by visual observation of the sample (e.g. looking at the sample).
[0040] Up-concentrated refers to increasing the concentration of the ProA pool to ensure all of the samples are comparable to each other. This is important since color can intensify with increased concentrations.
[0041] A polishing step refers to column steps that are meant to clear out high molecular weights and clippings.
[0042] A detergent is class of molecules that have a hydrophobic chain. Examples include deoxycholate and sodium caprylate.
[0043] For an affinity capture step and/or a CEX step of the methods described herein, the sample can be taken based on absorbance, for example from 0.5 OD A280 to 0.5 OD A280. The sample can also be fractionated, which refers to cutting the peak into different fractions based on volume. For example, fractions may be collected every 100 mis.
[0044] UV absorbance at 400 nm (“A400”) and 420 nm (“A420”) measures yellow color. UV absorbance at 280 nm (“A280”) measures protein.
[0045] An elution pool refers to a solution containing the substance that is eluted from a column or resin. For example, the affinity column capture step and the CEX step both have an elution step in which the Fc-containing molecule (e.g. antibody) is removed from the column or resin in an elution buffer. Accordingly, an elution pool can comprise an elution buffer and an Fc- containing molecule. An elution pool is a sample, as used herein.
[0046] A gradient elution refers to eluting the Fc-containing molecule with a buffer gradient. For example, the gradient can start at 0% of 100 mM sodium acetate, pH 5.0, 500 mM sodium chloride, and 100 % of 100 mM sodium acetate, pH 5.0; and end at 65% of 100 mM sodium acetate, pH 5.0, 500 mM sodium chloride, and 100 % of 100 mM sodium acetate, pH 5.0. The rate is indicated as mM/CV.
[0047] An Fc-containing molecule is a construct (or polypeptide(s)) that has an Fc region that is capable of binding Protein A. Non-limiting examples of Fc-containing molecules are antibodies and heterodimeric antibodies.
[0048] In some embodiments, the Fc-containing molecule is an antibody. As used herein, the term “antibody” refers to a protein having a conventional immunoglobulin format, comprising heavy and light chains, and comprising variable and constant regions. For example, an antibody may be an IgG which is a “Y-shaped” structure of two identical pairs of polypeptide chains, each pair having one “light” (typically having a molecular weight of about 25 kDa) and one “heavy” chain (typically having a molecular weight of about 50-70 kDa). An antibody has a variable region and a constant region. In IgG formats, the variable region is generally about 100- 110 or more amino acids, comprises three complementarity determining regions (CDRs), is primarily responsible for antigen recognition, and substantially varies among other antibodies that bind to different antigens. The constant region allows the antibody to recruit cells and molecules of the immune system. The variable region is made of the N-terminal regions of each light chain and heavy chain, while the constant region is made of the C-terminal portions of each of the heavy and light chains. (Janeway et al., “Structure of the Antibody Molecule and the Immunoglobulin Genes”, Immunobiology: The Immune System in Health and Disease, 4th ed. Elsevier Science Ltd. /Garland Publishing, (1999)). The fragment crystallizable region (“Fc region”) is the end of the antibody that binds Fc receptors and proteins of the complement system. The IgG Fc region, for example, comprises two CH2 and two CH3 domains. The Fc region binds with high affinity to Protein A.
[0049] The general structure and properties of CDRs of antibodies have been described in the art. Briefly, in an antibody scaffold, the CDRs are embedded within a framework in the heavy and light chain variable region where they constitute the regions largely responsible for antigen binding and recognition. A variable region comprises at least three heavy or light chain CDRs (Kabat et al., 1991, Sequences of Proteins of Immunological Interest, Public Health Service N.I.H., Bethesda, Md.; see also Chothia and Lesk, 1987, J. Mol. Biol. 196:901-917; Chothia et al., 1989, Nature 342: 877-883), within a framework region (designated framework regions 1-4, FR1, FR2, FR3, and FR4, by Kabat et al., 1991; see also Chothia and Lesk, 1987, supra).
[0050] Human light chains are classified as kappa and lambda light chains. Heavy chains are classified as mu, delta, gamma, alpha, or epsilon, and define the antibody's isotype as IgM, IgD, IgG, IgA, and IgE, respectively. IgG has several subclasses, including, but not limited to IgGl, IgG2, IgG3, and IgG4. IgM has subclasses, including, but not limited to, IgMl and IgM2. Embodiments of the invention include all such classes or isotypes of antibodies. The light chain constant region can be, for example, a kappa- or lambda-type light chain constant region, e.g., a
human kappa- or lambda-type light chain constant region. The heavy chain constant region can be, for example, an alpha-, delta-, epsilon-, gamma-, or mu-type heavy chain constant regions, e.g., a human alpha-, delta-, epsilon-, gamma-, or mu-type heavy chain constant region. Accordingly, in exemplary embodiments, the antibody is an antibody of isotype IgA, IgD, IgE, IgG, or IgM, including any one of IgGl, IgG2, IgG3 or IgG4. IgGl antibodies are particularly susceptible to reduction of di-sulfide bonds and, as a result, represent one preferred embodiment of the disclosure.
[0051] The antibody may be a monoclonal antibody or a polyclonal antibody. In some embodiments, the antibody comprises a sequence that is substantially similar to a naturally- occurring antibody produced by a mammal, e.g., mouse, rabbit, goat, horse, chicken, hamster, human, and the like. In this regard, the antibody may be considered as a mammalian antibody, e.g., a mouse antibody, rabbit antibody, goat antibody, horse antibody, chicken antibody, hamster antibody, human antibody, and the like. In certain aspects, the monoclonal antibody is a human antibody. In certain aspects, the monoclonal antibody is a chimeric antibody or a humanized antibody. The term “chimeric antibody” is used herein to refer to an antibody containing constant domains from one species and the variable domains from a second, or more generally, containing stretches of amino acid sequence from at least two species. The term “humanized” when used in relation to antibodies refers to antibodies having at least CDR regions from a non-human source which are engineered to have a structure and immunological function more similar to true human antibodies than the original source antibodies. For example, humanizing can involve grafting CDR from a non-human antibody, such as a mouse antibody, into a human antibody.
Humanizing also can involve select amino acid substitutions to make a non-human sequence look more like a human sequence.
[0052] Advantageously, the methods are not limited to the antigen-specificity of the antibody. Accordingly, the antibody has any binding specificity for virtually any antigen. In exemplary aspects, the antibody binds to a hormone, growth factor, cytokine, a cell-surface receptor, or any ligand thereof.
[0053] In some embodiments, the Fc-containing molecule is a heterodimeric antibody, which refer to antibodies comprising two different light chains and two different heavy chains. In some embodiments, a hetero Ig comprises two Fabs and an Fc region. In some embodiments, the two Fabs are each N-terminal to the Fc region. In some embodiments, the two Fabs are each C-terminal to the Fc region.
[0054] The heterodimeric antibodies can comprise any immunoglobulin constant region. The term “constant region” as used herein refers to all domains of an antibody other than the variable region. The constant region is not involved directly in binding of an antigen, but exhibits
various effector functions. As described above, antibodies are divided into particular isotypes (IgA, IgD, IgE, IgG, and IgM) and subtypes (IgGl, IgG2, IgG3, IgG4, IgAl IgA2) depending on the amino acid sequence of the constant region of their heavy chains. The light chain constant region can be, for example, a kappa- or lambda-type light chain constant region, e.g., a human kappa- or lambda-type light chain constant region, which are found in all five antibody isotypes. [0055] The heavy chain constant region of the heterodimeric antibodies can be, for example, an alpha-, delta-, epsilon-, gamma-, or mu-type heavy chain constant region, e.g., a human alpha-, delta-, epsilon-, gamma-, or mu-type heavy chain constant region. In some embodiments, the heterodimeric antibodies comprise a heavy chain constant region from an IgGl, IgG2, IgG3, or IgG4 immunoglobulin.
[0056] An example of a heterodimeric antibody is a DuoBody®. DuoBodies can be made by the DuoBody® technology platform (Genmab A/S) as described, e.g., in International Publication Nos. WO 2008/119353, WO 2011/131746, WO 2011/147986, and WO 2013/060867, Labrijn A F et al., PNAS, 110(13): 5145-5150 (2013), Gramer et al., mAbs, 5(6): 962-973 (2013), and Labrijn et al., Nature Protocols, 9(10): 2450-2463 (2014). This technology can be used to combine one half of a first monospecific antibody containing two heavy and two light chains with one half of a second monospecific antibody containing two heavy and two light chains. The resultant heterodimer contains one heavy chain and one light chain from the first antibody paired with one heavy chain and one light chain from the second antibody. When both of the monospecific antibodies recognize different epitopes on different antigens, the resultant heterodimer is a multispecific antibody.
[0057] Another exemplary method of generating multispecific antibodies is by the knobs- into-holes technology (Ridgway et al., Protein Eng., 9:617-621 (1996); WO 2006/028936). The mispairing problem of Ig heavy chains that is a chief drawback for making multispecific antibodies is reduced in this technology by mutating selected amino acids forming the interface of the heavy chains in IgG. At positions within the heavy chain at which the two heavy chains interact directly, an amino acid with a small side chain (hole) is introduced into the sequence of one heavy chain and an amino acid with a large side chain (knob) into the counterpart interacting residue location on the other heavy chain. In some instances, antibodies of the disclosure have immunoglobulin chains in which the heavy chains have been modified by mutating selected amino acids that interact at the interface between two polypeptides so as to preferentially form a multispecific antibody. The multispecific antibodies can be composed of immunoglobulin chains of the same subclass or different subclasses.
[0058] Yet another method of generating multispecific antibodies is the CrossMab technology. CrossMab are chimeric antibodies constituted by the halves of two full-length
antibodies. For correct chain pairing, it combines two technologies: (i) the knob-into-hole which favors a correct pairing between the two heavy chains; and (ii) an exchange between the heavy and light chains of one of the two Fabs to introduce an asymmetry which avoids light-chain mispairing. See, Ridgway et al., Protein Eng., 9:617-621 (1996); Schaefer et al., PNAS, 108: 11187-11192 (2011). CrossMabs can combine two or more antigen-binding domains for targeting two or more targets or for introducing bivalency towards one target such as the 2: 1 format.
[0059] In some embodiments, the Fc-containing molecule is a T cell engager molecule that has an Fc. In the most general sense, a T cell engager (“TCE”) molecule comprises a polypeptide(s) that can bind to two different antigens, one of which is preferentially CD3. An example of a TCE molecule with an Fc is a BiTE® molecule with an Fc. Said Fc can be used to extend the half-life of the molecule.
[0060] In some embodiments, the antigen binding protein, antibody or therapeutic protein is one of those described in U.S. Pat. No. 7,947,809 and U.S. Patent Application Publication No.
20090041784 (glucagon receptor), U.S. Pat. Nos. 7,939,070, 7,833,527, 7,767,206, and 7,786,284 (IL-17 receptor A), U.S. Pat. Nos. 7,872,106 and 7,592,429 (Sclerostin), U.S. Pat. Nos. 7,871,611, 7,815,907, 7,037,498, 7,700,742, and U.S. Patent Application Publication No.
20100255538 (IGF-1 receptor), U.S. Pat. No. 7,868,140 (B7RP1), U.S. Pat. No. 7,807,159 and U.S. Patent Application Publication No. 20110091455 (myostatin), U.S. Pat. Nos. 7,736,644, 7,628,986, 7,524,496, and U.S. Patent Application Publication No. 20100111979 (deletion mutants of epidermal growth factor receptor), U.S. Pat. No. 7,728,110 (SARS coronavirus), U.S. Pat. No. 7,718,776 and U.S. Patent Application Publication No. 20100209435 (OPGL), U.S. Pat. Nos. 7,658,924 and 7,521,053 (Angiopoietin-2), U.S. Pat. Nos. 7,601,818, 7,795,413, U.S. Patent Application Publication No. 20090155274, U.S. Patent Application Publication No.
20110040076 (NGF), U.S. Pat. No. 7,579,186 (TGF-P type II receptor), U.S. Pat. No. 7,541,438 (connective tissue growth factor), U.S. Pat. No. 7,438,910 (IL1-R1), U.S. Pat. No. 7,423,128 (properdin), U.S. Pat. Nos. 7,411,057, 7,824,679, 7,109,003, 6,682,736, 7,132,281, and
7,807,797 (CTLA-4), U.S. Pat. Nos. 7,084,257, 7,790,859, 7,335,743, 7,084,257, and U.S. Patent Application Publication No. 20110045537 (interferon-gamma), U.S. Pat. No. 7,932,372 (MAdCAM), U.S. Pat. No. 7,906,625, U.S. Patent Application Publication No. 20080292639, and U.S. Patent Application Publication No. 20110044986 (amyloid), U.S. Pat. Nos. 7,815,907 and 7,700,742 (insulin-like growth factor I), U.S. Pat. Nos. 7,566,772 and 7,964,193 (interleukin- 1P), U.S. Pat. Nos. 7,563,442, 7,288,251, 7,338,660, 7,626,012, 7,618,633, and U.S. Patent Application Publication No. 20100098694 (CD40), U.S. Pat. No. 7,498,420 (c-Met), U.S. Pat. Nos. 7,326,414, 7,592,430, and 7,728,113 (M-CSF), U.S. Pat. Nos. 6,924,360, 7,067,131, and
7,090,844 (MUC18), U.S. Pat. Nos. 6,235,883, 7,807,798, and U.S. Patent Application Publication No. 20100305307 (epidermal growth factor receptor), U.S. Pat. Nos. 6,716,587, 7,872,113, 7,465,450, 7,186,809, 7,317,090, and 7,638,606 (interleukin-4 receptor), U.S. Patent Application Publication No. 20110135657 (BETA-KLOTHO), U.S. Pat. Nos. 7,887,799 and 7,879,323 (fibroblast growth factor-like polypeptides), U.S. Pat. No. 7,867,494 (IgE), U.S. Patent Application Publication No. 20100254975 (ALPHA-4 BETA-7), U.S. Patent Application Publication No. 20100197005 and U.S. Pat. No. 7,537,762 (ACTIVIN RECEPTOR-LIKE KINASE-1), U.S. Pat. No. 7,585,500 and U.S. Patent Application Publication No. 20100047253 (IL-13), U.S. Patent Application Publication No. 20090263383 and U.S. Pat. No. 7,449,555 (CD148), U.S. Patent Application Publication No. 20090234106 (ACTIVIN A), U.S. Patent Application Publication No. 20090226447 (angiopoietin-1 and angiopoietin-2), U.S. Patent Application Publication No. 20090191212 (Angiopoietin-2), U.S. Patent Application Publication No. 20090155164 (C-FMS), U.S. Pat. No. 7,537,762 (activin receptor-like kinase-1), U.S. Pat. No. 7,371,381 (galanin), U.S. Patent Application Publication No. 20070196376 (INSULIN-LIKE GROWTH FACTORS), U.S. Pat. Nos. 7,267,960 and 7,741,115 (LDCAM), U.S. Pat. No. 7,265,212 (CD45RB), U.S. Pat. No. 7,709,611, U.S. Patent Application Publication No.
20060127393 and U.S. Patent Application Publication No. 20100040619 (DKK1), U.S. Pat. No. 7,807,795, U.S. Patent Application Publication No. 20030103978 and U.S. Pat. No. 7,923,008 (osteoprotegerin), U.S. Patent Application Publication No. 20090208489 (OV064), U.S. Patent Application Publication No. 20080286284 (PSMA), U.S. Pat. No. 7,888,482, U.S. Patent Application Publication No. 20110165171, and U.S. Patent Application Publication No. 20110059063 (PAR2), U.S. Patent Application Publication No. 20110150888 (HEPCIDIN), U.S. Pat. No. 7,939,640 (B7L-1), U.S. Pat. No. 7,915,391 (c-Kit), U.S. Pat. Nos. 7,807,796, 7,193,058, and U.S. Pat. No. 7,427,669 (ULBP), U.S. Pat. Nos. 7,786,271, 7,304,144, and U.S. Patent Application Publication No. 20090238823 (TSLP), U.S. Pat. No. 7,767,793 (SIGIRR), U.S. Pat. No. 7,705,130 (HER-3), U.S. Pat. No. 7,704,501 (ataxin-l-like polypeptide), U.S. Pat. Nos. 7,695,948 and 7,199,224 (TNF-a converting enzyme), U.S. Patent Application Publication No. 20090234106 (ACTIVIN A), U.S. Patent Application Publication No. 20090214559 and U.S. Pat. No. 7,438,910 (IL1-R1), U.S. Pat. No. 7,579,186 (TGF-P type II receptor), U.S. Pat. No. 7,569,387 (TNF receptor-like molecules), U.S. Pat. No. 7,541,438, (connective tissue growth factor), U.S. Pat. No. 7,521,048 (TRAIL receptor-2), U.S. Pat. Nos. 6,319,499, 7,081,523, and U.S. Patent Application Publication No. 20080182976 (erythropoietin receptor), U.S. Patent Application Publication No. 20080166352 and U.S. Pat. No. 7,435,796 (B7RP1), U.S. Pat. No. 7,423,128 (properdin), U.S. Pat. Nos. 7,422,742 and 7,141,653 (interleukin-5), U.S. Pat. Nos. 6,740,522 and 7,411,050 (RANKL), U.S. Pat. No. 7,378,091 (carbonic anhydrase IX (CA IX)
tumor antigen), U.S. Pat. Nos. 7,318,925 and 7,288,253 (parathyroid hormone), U.S. Pat. No. 7,285,269 (TNF), U.S. Pat. Nos. 6,692,740 and 7,270,817 (ACPL), U.S. Pat. No. 7,202,343 (monocyte chemo-attractant protein-1), U.S. Pat. No. 7,144,731 (SCF), U.S. Pat. Nos. 6,355,779 and 7,138,500 (4-1BB), U.S. Pat. No. 7,135,174 (PDGFD), U.S. Pat. Nos. 6,630,143 and 7,045,128 (Flt-3 ligand), U.S. Pat. No. 6,849,450 (metalloproteinase inhibitor), U.S. Pat. No. 6,596,852 (LERK-5), U.S. Pat. No. 6,232,447 (LERK-6), U.S. Pat. No. 6,500,429 (brain-derived neurotrophic factor), U.S. Pat. No. 6,184,359 (epithelium-derived T-cell factor), U.S. Pat. No. 6,143,874 (neurotrophic factor NNT-1), U.S. Patent Application Publication No. 20110027287 (PROPROTEIN CONVERTASE SUBTILISIN KEXIN TYPE 9 (PCSK9)), U.S. Patent Application Publication No. 20110014201 (IL- 18 RECEPTOR), and U.S. Patent Application Publication No. 20090155164 (C-FMS). The above patents and published patent applications are incorporated herein by reference in their entirety for purposes of their disclosure of variable domain polypeptides, variable domain encoding nucleic acids, host cells, vectors, methods of making polypeptides encoding said variable domains, pharmaceutical compositions, and methods of treating diseases associated with the respective target of the variable domain-containing antigen binding protein or antibody.
[0061] In some embodiments, the antibody is selected from the group consisting of Murom onab-CD3 (product marketed with the brand name Orthoclone Okt3®), Abciximab (product marketed with the brand name Reopro®.), Rituximab (product marketed with the brand name Mab Thera®, Rituxan®) (U.S. Pat. No. 5,843,439), Basiliximab (product marketed with the brand name Simulect®), Daclizumab (product marketed with the brand name Zenapax®), Palivizumab (product marketed with the brand name Synagis®), Infliximab (product marketed with the brand name Remicade®), Trastuzumab (product marketed with the brand name Herceptin®), Alemtuzumab (product marketed with the brand name MabCampath®, Campath- 1H®), Adalimumab (product marketed with the brand name Humira®), Tositumomab-1131 (product marketed with the brand name Bexxar®), Efalizumab (product marketed with the brand name Raptiva®), Cetuximab (product marketed with the brand name Erbitux®), Flbritumomab tiuxetan (product marketed with the brand name Zevalin®), I'Omalizumab (product marketed with the brand name Xolair®), Bevacizumab (product marketed with the brand name Avastin®), Natalizumab (product marketed with the brand name Tysabri®), Ranibizumab (product marketed with the brand name Lucentis®), Panitumumab (product marketed with the brand name Vectibix®), I'Eculizumab (product marketed with the brand name Soliris®), Certolizumab pegol (product marketed with the brand name Cimzia®), Golimumab (product marketed with the brand name Simponi®), Canakinumab (product marketed with the brand name Haris®), Catumaxomab (product marketed with the brand name Removab®), Ustekinumab (product marketed with the
brand name Stelara®), Tocilizumab (product marketed with the brand name RoActemra®, Actemra®), Ofatumumab (product marketed with the brand name Arzerra®), Denosumab (product marketed with the brand name Prolia®), Belimumab (product marketed with the brand name Benlysta®), Raxibacumab, Ipilimumab (product marketed with the brand name Yervoy®), and Pertuzumab (product marketed with the brand name Perjeta®). In exemplary embodiments, the antibody is one of anti-TNF alpha antibodies such as adalimumab, infliximab, etanercept, golimumab, and certolizumab pegol; anti-ILl.beta. antibodies such as canakinumab; anti-IL12/23 (p40) antibodies such as ustekinumab and briakinumab; and anti-IL2R antibodies, such as daclizumab. Examples of suitable anti-cancer antibodies include, but are not limited to, anti- CCR8 antibodies, anti-BAFF antibodies such as belimumab; anti-CD20 antibodies such as rituximab; anti-CD22 antibodies such as epratuzumab; anti-CD25 antibodies such as daclizumab; anti-CD30 antibodies such as iratumumab, anti-CD33 antibodies such as gemtuzumab, anti- CD52 antibodies such as alemtuzumab; anti-CD152 antibodies such as ipilimumab; anti-EGFR antibodies such as cetuximab; anti-HER2 antibodies such as trastuzumab and pertuzumab; anti- IL6 antibodies such as siltuximab; and anti-VEGF antibodies such as bevacizumab; anti-IL6 receptor antibodies such as tocilizumab.
[0062] A reduction in yellow color can be determined for a sample if the b* value or the amount of yellow color visually observed (by way of examples) in the sample is less using a wash of the present invention compared to a sample using another wash (e.g. a wash not of the present invention) or a sample which has not been subjected to any wash.
[0063] A reduction in the amount of impurity for a sample can be determined by comparing the amount of impurity in the sample using a wash of the present invention compared to the amount of impurity in a sample using another wash (e.g. a wash not of the present invention) or a sample which has not been subjected to any wash.
EXAMPLES
EXAMPLE 1. Using detergent wash during affinity capture step
[0064] Sodium deoxycholate or sodium caprylate were formulated into buffers to be used as a wash step during affinity capture purification of Antibody 1 harvested cell culture fluid (HCCF). Samples were generated using one of the detergent wash steps (deoxycholate or caprylate). The purification method utilized Cytiva MabSelect SuRe resin. Following the steps of equilibration, loading, and washing, a deoxycholate wash was step (25 mM Tris, 20 mM Sodium Deoxycholate, 100 mM NaCl, pH 8.2; 10 CV volume; 250 cm/hr flow rate) or a sodium caprylate step (25 mM Tris, 100 mM Sodium Caprylate, 2.5 M NaCl, pH 7.4; 10 CV volume;
250 cm/hr flow rate) was performed. After the deoxycholate or caprylate wash was performed, subsequent washes, elution, cleaning, and flush step(s) were performed prior to storage of the Fc- containing molecule. Control samples are generated from the same steps described below except there is no deoxycholate or caprylate wash step or the following wash step of 25 mM tris, lOOmM sodium chloride, pH 7.4.
[0065] During the elution step, the protein A pool is collected from 0.5 OD A280 to 0.5 OD A280.
[0066] For HunterLab color assay, neutralized (at pH 5.0) Protein A pools were up- concentrated to 70 mg/mL prior to submission. Photo-stressed samples generated through exposure to visible light (195 klux-hr visible light) were also submitted for HunterLab color assay.
[0067] The HunterLab UltraScan PRO spectrophotometer was used for the yellow color measurement in various Antibody 1 samples. The three color primaries measured across the visible wavelength range in a sample is mathematically converted by the instrument to the Opponent color scale values (CIELAB) of L*, a*, and b*. The L* is the degree of lightness to darkness, a* is the degree of redness to greenness, and b* is the degree of yellowness to blueness. The higher value of b* suggests that the sample is more yellow.
[0068] As shown in Figure 1, the addition of either detergent wash step reduces the yellow color. Y7-Y4 yellow color standards are included for reference. The results demonstrate that implementing either the deoxy cholate or the caprylate wash step reduces the b* value by ~ 35%.
EXAMPLE 2. Combination of the detergent wash steps was effective in clearing 303 Da species
[0069] Sodium deoxycholate and sodium caprylate were formulated into buffers to be used as a wash steps during affinity capture purification of Antibody 1 harvested cell culture fluid (HCCF). The purification method utilized Cytiva MabSelect SuRe resin. Protein A pools were collected and neutralized to pH 5.0, and samples were taken for analysis of 303 Da species. The procedure was performed essentially as described in Example 1, however both a deoxy cholate wash step and a caprylate wash step were used. Following the steps of equilibration, loading, and washing, a deoxycholate wash was step (25 mM Tris, 20 mM Sodium Deoxycholate, 100 mM NaCl, pH 8.2; 10 CV volume; 250 cm/hr flow rate) and a sodium caprylate wash step (25 mM Tris, 100 mM Sodium Caprylate, 2.5 M NaCl, pH 7.4; 10 CV volume; 250 cm/hr flow rate) were performed, separated by a wash step of 25 mM tris, 100 mM sodium chloride (between the sodium deoxycholate and sodium caprylate wash steps). After the
caprylate wash was performed, subsequent washes, elution, cleaning, and flush step(s) were performed prior to storage of the Fc-containing molecule.
[0070] For analysis of the 303 Da species, liquid chromatography with ultraviolet, fluorescence, and mass spectrometric detection (LC-UV-FD-MS) was performed using a Waters Acquity UPLC system with UV and fluorescence detector, directly connected to Thermo Fisher Scientific Q-Exactive HF (QE-HF) or Orbitrap Eclipse high resolution mass spectrometer. Samples were analyzed using a reversed phase column, Agilent Diphenyl RRHD (100x2.1mm, 1.8pm) at 60°C, using acetonitrile gradient at a flow rate of 0.3mL/min. Mobile phases contained 0.1% (v/v) formic acid. After initial hold at 1% B for 2 minutes, mobile phase B was increased linearly to 27%B in 13 minutes followed by 70%B in 7 minutes. Column was equilibrated with 1%B for 7 minutes. UV data was collected at wavelengths of 230nm, 280nm, 325nm, 365nm and 400nm. Fluorescence detector was set at an excitation wavelength of 365nm and an emission wavelength of 510nm with PMT gain 2. When mass spectrometer was used, QE- HF was set up to collect a high-resolution MS scan at resolution setting of 120,000 and automatic gain control (AGC) target of 1E6, followed by five data-dependent higher-energy collision dissociation (HCD) MS/MS (normalized collision energy = 27%), with dynamic exclusion, of the most abundant ions. While Eclipse was set up to collect data with variable collision energy (20- 60%). For analysis, 35pg of protein or 50pL of wash fractions or dilute samples were typically analyzed.
[0071] As shown in Figure 2, the 303 Da species was found on both samples taken from wash step and elution pool of the process in which deoxycholate and caprylate wash steps were not implemented. Conversely, in the method in which both deoxycholate and caprylate washes were implemented, the analytical assay revealed that the 303 Da species was mainly contained in the deoxycholate wash step and not detected in the elution pool. The caprylate step also contained some 303 Da species, however due to the order of the steps the levels of 303 Da species in the caprylate step were very low compared to the level of 303 Da species found in the deoxycholate step. Therefore, it can be concluded that the detergent wash steps were very effective in clearing 303 Da species from elution pool during affinity capture step.
EXAMPLE 3. Detection of Yellow Color Using CEX
[0072] CEX step is often leveraged as the main polishing step for many antibodies purification due to its ability to separate impurities based on charges. As described herein, a modified CEX step has been implemented to reduce yellow color in antibody elution pool (e.g. drug substance intermediate). In this experiment, the separation power of CaptoSP ImpRes (a strong cation exchange resin) was combined with the ability of the AKTA to trace UV
absorbance at 400 nm and 420 nm (wavelength absorbed by objects exhibiting yellow color), to show that yellow color impurity can be detected and fractionated using a CEX step of the present invention. CEX method leveraged the CaptoSP ImpRes resin from Cytiva.
[0073] CEX was performed with steps of pre-equilibration, equilibration, loading, wash, elution, cleaning (non-denaturing and denaturing), and storage. A gradient elution was used, wherein the gradient started at 0% B and 100% A and ended at 65% B and 35% A, at a rate of 8 mM/CV. During wash and elution steps, UV absorbance at wavelength 400 nm and 420 nm were tracked to monitor yellow color species. The elution pool was fractionated at 0.5 CV. Samples and controls were then submitted for HunterLab color assay and LC-MS assay for measurement of 303 Da species as described above.
[0074] As shown in Figure 3, the increase in UV absorbance at 400 nm and 420 nm correlate to visibly yellower fractions collected from the elution phase. Several pseudopools (combining proportionally equal parts of each of the fractions into one pool that is representative of all of the fractions) were also created to demonstrate that exclusion of the fractions with elevated 400 nm and 420 nm UV absorbance was able to reduce the level of yellow color in the resulting pools.
[0075] Pooled fractions were tested for quantification of yellow color and these results are shown below in Table 1. The first three fractions were visibly more yellow than the rest of the fractions and were not tested. However, subtracting these fractions from the pool resulted in a less yellow overall pool.
EXAMPLE 4. Developing sodium chloride salt wash during CEX polishing step [0076] Sodium chloride salt washes were further developed to provide better clearance of the yellow color species. The salt condition had to be adjusted to match run conditions, such as pH and loading levels. The buffer was 100 mM Acetate and either 65, 75, or 85 mM NaCl, pH 5.0; 3 CV; flow rate 140 cm/hr. CEX was performed with steps of pre-equilibration, equilibration, loading, wash (100 mM Sodium Acetate, pH 5.0), sodium chloride salt wash (100
mM Acetate and either 65, 75, or 85 mM NaCl, pH 5.0; 3 CV; flow rate 140 cm/hr), wash (100 mM Sodium Acetate, pH 5.0), elution, cleaning (non-denaturing and denaturing), and storage. Samples were taken after each step.
[0077] Higher sodium chloride salt wash level corresponds to lower yellow color as represented by b* value on HunterLAB assay, but also lead to lower yield. Based on robustness study data shown in Table 2, 75 mM salt was found to be the ideal sodium chloride salt wash condition to achieve the highest yellow color reduction while keeping acceptable yield. The 75 mM sodium chloride salt wash was also shown to be effective in reducing yellow color in Antibody 2. Step yield refers to how much antibody is loaded compared to how much antibody is in the elution pool.
Table 2, Results of robustness study data comparing reduction of yellow color at various sodium chloride salt wash levels.
[0078] These data demonstrate that the sodium chloride salt wash step during CEX polishing step can reduce yellow color at elution.
[0079] The reduction of 303 Da species was also determined. Figure 4 shows the assay result from various samples taken during a CEX step without the sodium chloride salt wash, and a CEX step with the sodium chloride salt wash. Samples were taken after each step. The 303 Da species was present in the CEX elution pool without the sodium chloride salt wash, with a slightly higher concentration found during the first elution fraction. There was a higher concentration of 303 Da species found in the wash step samples in the CEX method using a sodium chloride salt wash step, and although it was still present in the elution pool of the
proposed CEX method, the level was very low. Further processing through a third column (flowthrough MMC) cleared the 303 Da species to lower than assay detection level. Figure 5 and Table 3 demonstrate data on relative level of 303 Da species, obtained via integration of peak on LC-UV-FD-MS assay, in various Antibody 1 and Antibody 2 sodium chloride salt wash samples. Based on the data, the 75 mM sodium chloride salt wash is effective in reducing the level of 303 Da species in both Antibody 1 and Antibody 2 while retaining acceptable antibody yield. The data also shows that Antibody 2 has lower level of 303 Da species to begin with compared to Antibody 1.
Claims
1. A method of reducing yellow color in an Fc-containing molecule sample, wherein said method comprises capture affinity, and wherein the capture affinity comprises a detergent wash that is a sodium deoxycholate wash and/or a sodium caprylate wash.
2. The method of claim 1, wherein the detergent wash is a sodium deoxy cholate wash.
3. The method of claim 1, wherein the detergent wash is a sodium caprylate wash.
4. The method of claim 2, wherein the method further comprises an additional detergent wash wherein the additional detergent wash is sodium caprylate.
5. The method of any one of claims 1, 2, and 4, wherein the deoxy cholate wash comprises 20 mM sodium deoxycholate.
6. The method of claim 5, wherein the deoxycholate wash comprises 25 mM Tris, 20 mM sodium deoxy cholate, and 100 mM NaCl.
7. The method of claim 5 or claim 6, wherein the deoxycholate wash is at about pH 8.2.
8. The method of any one of claims 1-3, wherein the sodium caprylate wash comprises 100 mM sodium caprylate.
9. The method of claim 8, wherein the sodium caprylate wash comprises 25 mM Tris, 100 mM sodium caprylate and 2.5 M NaCl.
10. The method of claim 5 or claim 6, wherein the sodium caprylate wash is at about pH 7.4.
11. The method of any one of claims 4-10, wherein the deoxy cholate wash is performed prior to the caprylate detergent wash.
12. The method of any one of claims 4-10, wherein the caprylate wash is performed prior to the deoxycholate wash.
13. A method of detecting yellow color in an Fc-containing molecule sample, wherein said method is a cation exchange (CEX) method comprising a strong cation exchange resin and detecting UV absorbance at 400 nm and 420 nm.
14. The method of claim 13, wherein said method comprises a gradient elution.
15. A method of reducing yellow color in an Fc-containing molecule sample, wherein said method is a CEX method comprising a strong cation exchange resin and a sodium chloride salt wash.
16. The method of claim 15, wherein the sodium chloride salt wash is 75 mM sodium chloride.
17. The method of claim 15 or claim 16, wherein the sodium chloride salt wash is 100 mM sodium acetate, pH 5.0, and 75 mM sodium chloride.
18. The method of any one of claims 16-18, further comprising a gradient elution.
19. The method of claim 14 or claim 18, wherein the gradient elution is from: a. 0% of 100 mM sodium acetate, pH 5.0, 500 mM sodium chloride, and 100 % of 100 mM sodium acetate, pH 5.0; to b. 65% of 100 mM sodium acetate, pH 5.0, 500 mM sodium chloride, and 100 % of 100 mM sodium acetate, pH 5.0.
20. A method of reducing yellow color in an Fc-containing molecule sample, comprising the method of any one of claims 1-12, and further comprising the method of any one of claims 15-19.
21. A method of reducing an impurity in an Fc-containing molecule sample, comprising the method of any one of claims 1-12, and 15-19.
22. The method of claim 21, wherein the impurity is detected by liquid chromatography with ultraviolet, fluorescence, and mass spectrometric detection.
23. The method of claim 21 or claim 22, wherein the impurity is a 303 Da species.
24. The method of any one of claims 1-23, wherein the Fc-containing molecule sample is a drug substance intermediate.
25. The method of any one of claims 1-24, wherein the method is performed during purification of the Fc-containing molecule.
26. The method of any one of claims 1-25, wherein the Fc-containing molecule is an antibody.
27. The method of any one of claims 1-25, wherein the Fc-containing molecule is a heterodimeric antibody.
28. The method of any one of claims 1-27, wherein the yellow color or the impurity in the sample is reduced compared to a sample that has been subjected to another wash or has not been subjected to any wash.
29. The method of claim 28, wherein the yellow color is reduced by at least 5%.
30. The method of claim 28 or claim 29, wherein the yellow color is reduced by at least 10%.
31. The method of any one of claims 28-30, wherein the yellow color is reduced by at least 20%.
32. The method of any one of claims 28-31, wherein the yellow color is reduced by at least 35%.
33. The method of any one of claims 28-32, wherein the yellow color is determined by comparing b* value of the Fc-containing molecule sample with a sample of an Fc-containing molecule that has not been subjected to the method of any one of claims 1-20 and 24-32.
34. The method of any one of claims 1-33, wherein the sample is an elution pool.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US202363532596P | 2023-08-14 | 2023-08-14 | |
| US63/532,596 | 2023-08-14 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2025038600A1 true WO2025038600A1 (en) | 2025-02-20 |
Family
ID=92583428
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2024/042037 Pending WO2025038600A1 (en) | 2023-08-14 | 2024-08-13 | Methods for reducing yellow color |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2025038600A1 (en) |
Citations (104)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1989006692A1 (en) * | 1988-01-12 | 1989-07-27 | Genentech, Inc. | Method of treating tumor cells by inhibiting growth factor receptor function |
| EP0388738A1 (en) * | 1989-03-14 | 1990-09-26 | F. Hoffmann-La Roche Ag | Plasmodium merozoite rhoptries antigen and derivatives |
| US5843439A (en) | 1992-11-13 | 1998-12-01 | Anderson; Darrell R. | Therapeutic application of chimeric and radiolabeled antibodies to human B lymphocyte restricted differentiation antigen for treatment of B cell lymphoma |
| US6143874A (en) | 1997-02-03 | 2000-11-07 | Amgen Inc | Antibodies to the neurotrophic factor NNT-1 |
| US6184359B1 (en) | 1993-03-08 | 2001-02-06 | Immunex Corporation | Antibodies to epithelium-derived T-cell factor |
| US6232447B1 (en) | 1994-10-05 | 2001-05-15 | Immunex Corporation | Antibody immunoreactive with a human cytokine designated LERK-6 |
| US6235883B1 (en) | 1997-05-05 | 2001-05-22 | Abgenix, Inc. | Human monoclonal antibodies to epidermal growth factor receptor |
| US6319499B1 (en) | 1994-07-26 | 2001-11-20 | Amgen Inc. | Methods for activating an erythropoietin receptor using antibodies |
| US6355779B1 (en) | 1993-05-07 | 2002-03-12 | Immunex Corporation | Cytokine designated 4-1BB ligand antibodies and human receptor that binds thereto |
| US6500429B2 (en) | 1996-07-19 | 2002-12-31 | Amgen Inc. | Antibodies against analogs of brain-derived neurotrophic factor |
| US20030103978A1 (en) | 2000-02-23 | 2003-06-05 | Amgen Inc. | Selective binding agents of osteoprotegerin binding protein |
| US6596852B2 (en) | 1994-07-08 | 2003-07-22 | Immunex Corporation | Antibodies that bind the cytokine designated LERK-5 |
| US6682736B1 (en) | 1998-12-23 | 2004-01-27 | Abgenix, Inc. | Human monoclonal antibodies to CTLA-4 |
| US6692740B2 (en) | 1998-01-23 | 2004-02-17 | Immunex Corporation | ACPL antibodies |
| US6716587B2 (en) | 1988-10-31 | 2004-04-06 | Immunex Corporation | Antibodies to interleukin-4 receptors and uses thereof |
| US6740522B2 (en) | 1996-12-23 | 2004-05-25 | Immunex Corporation | Antibodies against ligand for receptor activator of NF-kB |
| US6849450B2 (en) | 1989-05-19 | 2005-02-01 | Childrens Hospital Of Los Angeles | Antibodies to the metalloproteinase inhibitor |
| US6924360B2 (en) | 2001-12-28 | 2005-08-02 | Abgenix, Inc. | Antibodies against the MUC18 antigen |
| WO2006028936A2 (en) | 2004-09-02 | 2006-03-16 | Genentech, Inc. | Heteromultimeric molecules |
| US7037498B2 (en) | 2001-01-05 | 2006-05-02 | Abgenix, Inc. | Antibodies to insulin-like growth factor I receptor |
| US7045128B2 (en) | 1993-05-24 | 2006-05-16 | Immunex Corporation | Antibodies against flt3-ligand |
| US20060127393A1 (en) | 2004-08-04 | 2006-06-15 | Amgen Inc. | Antibodies to Dkk-1 |
| US7067131B2 (en) | 2001-12-28 | 2006-06-27 | Abgenix, Inc. | Methods for using anti-MUC18 antibodies |
| US7084257B2 (en) | 2001-10-05 | 2006-08-01 | Amgen Inc. | Fully human antibody Fab fragments with human interferon-gamma neutralizing activity |
| US7090844B2 (en) | 2001-12-28 | 2006-08-15 | Abgenix, Inc. | Use of antibodies against the MUC18 antigen |
| US7109003B2 (en) | 1998-12-23 | 2006-09-19 | Abgenix, Inc. | Methods for expressing and recovering human monoclonal antibodies to CTLA-4 |
| US7135174B2 (en) | 2002-01-07 | 2006-11-14 | Amgen Fremont, Inc. | Antibodies directed to PDGFD and uses thereof |
| US7138500B1 (en) | 1993-05-07 | 2006-11-21 | Immunex Corporation | Antibodies to human 4-1BB |
| US7141653B2 (en) | 2002-03-29 | 2006-11-28 | Schering Corporation | Human monoclonal antibodies to interleukin-5 |
| US7144731B2 (en) | 1989-10-16 | 2006-12-05 | Amgen Inc. | SCF antibody compositions and methods of using the same |
| US7186809B2 (en) | 2000-05-26 | 2007-03-06 | Immunex Corporation | Methods and compositions relating to anti-interleukin-4 receptor antibodies |
| US7193058B2 (en) | 1997-12-17 | 2007-03-20 | Immunex Corporation | ULBP antibodies |
| US7199224B2 (en) | 1995-06-08 | 2007-04-03 | Immunex Corporation | Antibodies that bind TNF-α converting enzyme |
| US7202343B2 (en) | 2002-08-19 | 2007-04-10 | Abgenix, Inc. | Antibodies directed to monocyte chemo-attractant protein-1 (MCP-1) and uses thereof |
| US20070196376A1 (en) | 2005-12-13 | 2007-08-23 | Amgen Fremont Inc. | Binding proteins specific for insulin-like growth factors and uses thereof |
| US7265212B2 (en) | 2001-12-03 | 2007-09-04 | Amgen Fremont Inc. | Anti-CD45RB antibodies |
| US7267960B2 (en) | 2003-07-25 | 2007-09-11 | Amgen Inc. | Antagonists and agonists of LDCAM and methods of use |
| US7285269B2 (en) | 2002-12-02 | 2007-10-23 | Amgen Fremont, Inc. | Antibodies directed to tumor necrosis factor |
| US7288253B2 (en) | 2003-08-08 | 2007-10-30 | Amgen Fremont, Inc. | Antibodies directed to parathyroid hormone (PTH) and uses thereof |
| US7288251B2 (en) | 2001-11-09 | 2007-10-30 | Abgenix, Inc. | Antibodies to CD40 |
| US7304144B2 (en) | 1998-11-13 | 2007-12-04 | Immunex Corporation | Antibodies binding to human TSLP Polypeptides |
| US7318925B2 (en) | 2003-08-08 | 2008-01-15 | Amgen Fremont, Inc. | Methods of use for antibodies against parathyroid hormone |
| US7326414B2 (en) | 2003-09-10 | 2008-02-05 | Warner-Lambert Company Llc | Antibodies to M-CSF |
| US7335743B2 (en) | 2002-10-16 | 2008-02-26 | Amgen Inc. | Human anti-IFN-γ neutralizing antibodies as selective IFN-γ pathway inhibitors |
| US7371381B2 (en) | 2003-12-12 | 2008-05-13 | Amgen Inc. | Anti-galanin antibodies and uses thereof |
| US7378091B2 (en) | 2001-12-03 | 2008-05-27 | Amgen Fremont Inc. | Antibodies against carbonic anhydrase IX (CA IX) tumor antigen |
| US20080166352A1 (en) | 2005-07-18 | 2008-07-10 | Amgen Inc. | Human anti-B7RP1 Neutralizing Antibodies |
| US7423128B2 (en) | 2004-11-03 | 2008-09-09 | Amgen Fremont Inc. | Anti-properdin antibodies, and methods for making and using same |
| WO2008119353A1 (en) | 2007-03-29 | 2008-10-09 | Genmab A/S | Bispecific antibodies and methods for production thereof |
| US7435796B1 (en) | 1999-02-03 | 2008-10-14 | Amgen Inc. | Antibodies which bind B7RP1 |
| US7438910B2 (en) | 2002-09-06 | 2008-10-21 | Amgen Inc. | Therapeutic human anti-IL1-R1 monoclonal antibody |
| US7449555B2 (en) | 2004-04-23 | 2008-11-11 | Amgen Inc. | Antibodies of angiogenesis inhibiting domains CD148 |
| US20080286284A1 (en) | 2001-10-23 | 2008-11-20 | Psma Development Company, Llc | Compositions of PSMA antibodies |
| US20080292639A1 (en) | 2005-01-24 | 2008-11-27 | Amgen Inc. | Humanized Anti-Amyloid Antibody |
| US20090041784A1 (en) | 2006-09-20 | 2009-02-12 | Amgen Inc. | Compositions and methods relating to glucagon receptor antibodies |
| US7498420B2 (en) | 2003-08-04 | 2009-03-03 | Amgen Fremont Inc. | Antibodies to c-Met |
| US7521053B2 (en) | 2001-10-11 | 2009-04-21 | Amgen Inc. | Angiopoietin-2 specific binding agents |
| US7521048B2 (en) | 2005-08-31 | 2009-04-21 | Amgen Inc. | TRAIL receptor-2 polypeptides and antibodies |
| US7537762B2 (en) | 2005-09-07 | 2009-05-26 | Amgen Fremont, Inc. | Human monoclonal antibodies to activin receptor-like kinase-1 |
| US7541438B2 (en) | 1997-12-25 | 2009-06-02 | Amgen, Inc. | Monoclonal antibody against connective tissue growth factor and medicinal uses thereof |
| US20090155274A1 (en) | 2003-07-15 | 2009-06-18 | Amgen Inc. | Human anti-ngf neutralizing antibodies as selective ngf pathway inhibitors |
| US20090155164A1 (en) | 2007-08-21 | 2009-06-18 | Amgen, Inc. | Human c-fms antigen binding proteins |
| US7566772B2 (en) | 2005-01-26 | 2009-07-28 | Amgen Fremont Inc. | Antibodies against interleukin-1β |
| US20090191212A1 (en) | 2002-10-10 | 2009-07-30 | Amgen, Inc. | Angiopoietin-2 Specific Binding Agents |
| US7569387B2 (en) | 2000-09-05 | 2009-08-04 | Amgen Inc. | Antibody to TNF receptor-like molecules |
| US20090208489A1 (en) | 2005-03-24 | 2009-08-20 | Millennium Pharmaceuticals, Inc. Intellectual Property Group | Antibodies That Bind OV064 and Methods of Use Therefor |
| US7579186B1 (en) | 1999-11-18 | 2009-08-25 | Amgen Fremont Inc. | Human monoclonal antibody against TGF-β type II receptor and medicinal use thereof |
| US7585500B2 (en) | 2004-11-17 | 2009-09-08 | Amgen Inc. | Fully human monoclonal antibodies to IL-13 |
| US20090226447A1 (en) | 2008-02-20 | 2009-09-10 | Amgen, Inc. | Antibodies directed to angiopoietin-1 and angiopoietin-2 and uses thereof |
| US20090234106A1 (en) | 2006-09-08 | 2009-09-17 | Amgen Inc. | Anti-activin a antibodies and uses thereof |
| US7592429B2 (en) | 2005-05-03 | 2009-09-22 | Ucb Sa | Sclerostin-binding antibody |
| US20090238823A1 (en) | 2007-09-10 | 2009-09-24 | Amgen Inc. | Antigen binding proteins capable of binding thymic stromal lymphopoietin |
| US20090263383A1 (en) | 2004-04-23 | 2009-10-22 | Amgen Inc. | Antibodies to angiogenesis inhibiting domains of CD148 |
| US7628986B2 (en) | 2003-06-27 | 2009-12-08 | Amgen Fremont Inc. | Antibodies directed to the deletion mutants of epidermal growth factor receptor and uses thereof |
| US7638606B2 (en) | 2003-11-07 | 2009-12-29 | Immunex Corporation | Antibodies that bind interleukin-4 receptor |
| US7704501B2 (en) | 2001-07-30 | 2010-04-27 | Immunex Corporation | Antibodies binding to human ataxin-1-like polypeptide |
| US7705130B2 (en) | 2005-12-30 | 2010-04-27 | U3 Pharma Gmbh | Antibodies directed to HER-3 and uses thereof |
| US7718776B2 (en) | 2002-04-05 | 2010-05-18 | Amgen Inc. | Human anti-OPGL neutralizing antibodies as selective OPGL pathway inhibitors |
| US7728110B2 (en) | 2006-05-19 | 2010-06-01 | Amgen, Inc. | Antibodies to SARS coronavirus |
| US7741115B2 (en) | 1998-08-07 | 2010-06-22 | Immunex Corporation | Antibodies that bind LDCAM |
| US7767206B2 (en) | 2006-10-02 | 2010-08-03 | Amgen Inc. | Neutralizing determinants of IL-17 Receptor A and antibodies that bind thereto |
| US7767793B2 (en) | 1997-12-23 | 2010-08-03 | Immunex Corporation | Antibodies to SIGIRR |
| US7807159B2 (en) | 2005-04-25 | 2010-10-05 | Amgen Fremont Inc. | Antibodies to myostatin |
| US7807795B2 (en) | 1997-04-16 | 2010-10-05 | Amgen Inc. | Antibodies to osteoprotegerin binding proteins |
| US7807798B2 (en) | 1997-05-05 | 2010-10-05 | Amgen Fremont Inc. | Human monoclonal antibodies to epidermal growth factor receptor |
| US20100254975A1 (en) | 2009-03-20 | 2010-10-07 | Amgen Inc. | Alpha-4 beta-7 heterodimer specific |
| US7867494B2 (en) | 2007-04-02 | 2011-01-11 | Amgen Fremont Inc. | Anti-IgE antibodies |
| US7871611B2 (en) | 2004-12-22 | 2011-01-18 | Amgen Inc. | Compositions and methods relating to anti IGF-1 receptor antibodies |
| US20110014201A1 (en) | 2007-07-24 | 2011-01-20 | Amgen Inc. | Il-18 receptor antigen binding proteins |
| US7879323B2 (en) | 1999-09-07 | 2011-02-01 | Amgen Inc. | Antibodies to fibroblast growth factor-like polypeptides |
| US20110027287A1 (en) | 2007-08-23 | 2011-02-03 | Amgen Inc. | Antigen binding proteins to proprotein convertase subtilisin kexin type 9 (pcsk9) |
| US7888482B2 (en) | 2006-02-10 | 2011-02-15 | Amgen Inc. | Antibodies that bind PAR-2 |
| US20110044986A1 (en) | 2007-12-21 | 2011-02-24 | Amgen Inc. | Anti-amyloid antibodies and uses thereof |
| US20110059063A1 (en) | 2007-06-29 | 2011-03-10 | Amgen Inc. | Antigen binding proteins that bind PAR-2 |
| US7915391B2 (en) | 2006-04-24 | 2011-03-29 | Amgen Inc. | Humanized c-Kit antibody |
| US7923008B2 (en) | 1997-04-16 | 2011-04-12 | Amgen Inc. | Methods for decreasing osteoclast formation or bone resorption using an antibody to osteoprotegerin binding protein |
| US7932372B2 (en) | 2004-01-09 | 2011-04-26 | Amgen Fremont Inc. | Antibodies to MAdCAM |
| US7939640B2 (en) | 1998-08-07 | 2011-05-10 | Immunex Corporation | Antibodies that bind B7L-1 |
| US20110135657A1 (en) | 2009-12-07 | 2011-06-09 | Amgen Inc. | Human antigen binding proteins that bind beta-klotho, fgf receptors and complexes thereof |
| US20110150888A1 (en) | 2008-05-01 | 2011-06-23 | Amgen Inc. | Anti-hepcidin antibodies and methods of use |
| WO2011131746A2 (en) | 2010-04-20 | 2011-10-27 | Genmab A/S | Heterodimeric antibody fc-containing proteins and methods for production thereof |
| WO2011147986A1 (en) | 2010-05-27 | 2011-12-01 | Genmab A/S | Monoclonal antibodies against her2 |
| US8263750B2 (en) | 2006-03-16 | 2012-09-11 | Amgen Inc. | Method for purifying a protein using protein-A affinity chromatography using an intermediate wash step |
| WO2013060867A2 (en) | 2011-10-27 | 2013-05-02 | Genmab A/S | Production of heterodimeric proteins |
-
2024
- 2024-08-13 WO PCT/US2024/042037 patent/WO2025038600A1/en active Pending
Patent Citations (156)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1989006692A1 (en) * | 1988-01-12 | 1989-07-27 | Genentech, Inc. | Method of treating tumor cells by inhibiting growth factor receptor function |
| US6716587B2 (en) | 1988-10-31 | 2004-04-06 | Immunex Corporation | Antibodies to interleukin-4 receptors and uses thereof |
| US7317090B2 (en) | 1988-10-31 | 2008-01-08 | Immunex Corporation | Antibodies to Interleukin-4 receptors and uses thereof |
| EP0388738A1 (en) * | 1989-03-14 | 1990-09-26 | F. Hoffmann-La Roche Ag | Plasmodium merozoite rhoptries antigen and derivatives |
| US6849450B2 (en) | 1989-05-19 | 2005-02-01 | Childrens Hospital Of Los Angeles | Antibodies to the metalloproteinase inhibitor |
| US7144731B2 (en) | 1989-10-16 | 2006-12-05 | Amgen Inc. | SCF antibody compositions and methods of using the same |
| US5843439A (en) | 1992-11-13 | 1998-12-01 | Anderson; Darrell R. | Therapeutic application of chimeric and radiolabeled antibodies to human B lymphocyte restricted differentiation antigen for treatment of B cell lymphoma |
| US6184359B1 (en) | 1993-03-08 | 2001-02-06 | Immunex Corporation | Antibodies to epithelium-derived T-cell factor |
| US6355779B1 (en) | 1993-05-07 | 2002-03-12 | Immunex Corporation | Cytokine designated 4-1BB ligand antibodies and human receptor that binds thereto |
| US7138500B1 (en) | 1993-05-07 | 2006-11-21 | Immunex Corporation | Antibodies to human 4-1BB |
| US7045128B2 (en) | 1993-05-24 | 2006-05-16 | Immunex Corporation | Antibodies against flt3-ligand |
| US6596852B2 (en) | 1994-07-08 | 2003-07-22 | Immunex Corporation | Antibodies that bind the cytokine designated LERK-5 |
| US6319499B1 (en) | 1994-07-26 | 2001-11-20 | Amgen Inc. | Methods for activating an erythropoietin receptor using antibodies |
| US20080182976A1 (en) | 1994-07-26 | 2008-07-31 | Amgen Inc. | Antibodies which activate an erythropoietin receptor |
| US7081523B2 (en) | 1994-07-26 | 2006-07-25 | Amgen Inc. | Antibodies which activate an erythropoietin receptor |
| US6232447B1 (en) | 1994-10-05 | 2001-05-15 | Immunex Corporation | Antibody immunoreactive with a human cytokine designated LERK-6 |
| US7695948B2 (en) | 1995-06-08 | 2010-04-13 | Immunex Corporation | Antibodies that bind TNF-α converting enzyme |
| US7199224B2 (en) | 1995-06-08 | 2007-04-03 | Immunex Corporation | Antibodies that bind TNF-α converting enzyme |
| US6500429B2 (en) | 1996-07-19 | 2002-12-31 | Amgen Inc. | Antibodies against analogs of brain-derived neurotrophic factor |
| US6740522B2 (en) | 1996-12-23 | 2004-05-25 | Immunex Corporation | Antibodies against ligand for receptor activator of NF-kB |
| US7411050B2 (en) | 1996-12-23 | 2008-08-12 | Immunex Corporation | Monoclonal blocking antibody to human RANKL |
| US6143874A (en) | 1997-02-03 | 2000-11-07 | Amgen Inc | Antibodies to the neurotrophic factor NNT-1 |
| US7807795B2 (en) | 1997-04-16 | 2010-10-05 | Amgen Inc. | Antibodies to osteoprotegerin binding proteins |
| US7923008B2 (en) | 1997-04-16 | 2011-04-12 | Amgen Inc. | Methods for decreasing osteoclast formation or bone resorption using an antibody to osteoprotegerin binding protein |
| US6235883B1 (en) | 1997-05-05 | 2001-05-22 | Abgenix, Inc. | Human monoclonal antibodies to epidermal growth factor receptor |
| US7807798B2 (en) | 1997-05-05 | 2010-10-05 | Amgen Fremont Inc. | Human monoclonal antibodies to epidermal growth factor receptor |
| US20100305307A1 (en) | 1997-05-05 | 2010-12-02 | Amgen Fremont Inc. | Human monoclonal antibodies to epidermal growth factor receptor |
| US7427669B2 (en) | 1997-12-17 | 2008-09-23 | Immunex Corporation | ULBP antibodies |
| US7807796B2 (en) | 1997-12-17 | 2010-10-05 | Immunex Corporation | ULBP antibodies |
| US7193058B2 (en) | 1997-12-17 | 2007-03-20 | Immunex Corporation | ULBP antibodies |
| US7767793B2 (en) | 1997-12-23 | 2010-08-03 | Immunex Corporation | Antibodies to SIGIRR |
| US7541438B2 (en) | 1997-12-25 | 2009-06-02 | Amgen, Inc. | Monoclonal antibody against connective tissue growth factor and medicinal uses thereof |
| US7270817B2 (en) | 1998-01-23 | 2007-09-18 | Immunex Corporation | ACPL antibodies and methods of use thereof |
| US6692740B2 (en) | 1998-01-23 | 2004-02-17 | Immunex Corporation | ACPL antibodies |
| US7741115B2 (en) | 1998-08-07 | 2010-06-22 | Immunex Corporation | Antibodies that bind LDCAM |
| US7939640B2 (en) | 1998-08-07 | 2011-05-10 | Immunex Corporation | Antibodies that bind B7L-1 |
| US7786271B2 (en) | 1998-11-13 | 2010-08-31 | Immunex Corporation | Antibodies that inhibit TSLP activity |
| US7304144B2 (en) | 1998-11-13 | 2007-12-04 | Immunex Corporation | Antibodies binding to human TSLP Polypeptides |
| US7109003B2 (en) | 1998-12-23 | 2006-09-19 | Abgenix, Inc. | Methods for expressing and recovering human monoclonal antibodies to CTLA-4 |
| US6682736B1 (en) | 1998-12-23 | 2004-01-27 | Abgenix, Inc. | Human monoclonal antibodies to CTLA-4 |
| US7824679B2 (en) | 1998-12-23 | 2010-11-02 | Amgen Fremont Inc. | Human monoclonal antibodies to CTLA-4 |
| US7807797B2 (en) | 1998-12-23 | 2010-10-05 | Amgen Fremont Inc. | Human monoclonal antibodies to CTLA-4 |
| US7411057B2 (en) | 1998-12-23 | 2008-08-12 | Amgen Fremont Inc. | Nucleic acids encoding human monoclonal antibodies to CTLA-4 |
| US7132281B2 (en) | 1998-12-23 | 2006-11-07 | Amgen Fremont Inc. | Methods and host cells for producing human monoclonal antibodies to CTLA-4 |
| US7435796B1 (en) | 1999-02-03 | 2008-10-14 | Amgen Inc. | Antibodies which bind B7RP1 |
| US7879323B2 (en) | 1999-09-07 | 2011-02-01 | Amgen Inc. | Antibodies to fibroblast growth factor-like polypeptides |
| US7887799B2 (en) | 1999-09-07 | 2011-02-15 | Amgen Inc. | Antibodies to fibroblast growth factor-like polypeptides |
| US7579186B1 (en) | 1999-11-18 | 2009-08-25 | Amgen Fremont Inc. | Human monoclonal antibody against TGF-β type II receptor and medicinal use thereof |
| US20030103978A1 (en) | 2000-02-23 | 2003-06-05 | Amgen Inc. | Selective binding agents of osteoprotegerin binding protein |
| US7465450B2 (en) | 2000-05-26 | 2008-12-16 | Immunex Corporation | Methods and compositions relating to anti-interleukin-4 receptor antibodies |
| US7186809B2 (en) | 2000-05-26 | 2007-03-06 | Immunex Corporation | Methods and compositions relating to anti-interleukin-4 receptor antibodies |
| US7569387B2 (en) | 2000-09-05 | 2009-08-04 | Amgen Inc. | Antibody to TNF receptor-like molecules |
| US7815907B2 (en) | 2001-01-05 | 2010-10-19 | Amgen Fremont Inc. | Antibodies to insulin-like growth factor I receptor |
| US7700742B2 (en) | 2001-01-05 | 2010-04-20 | Amgen Fremont | Antibodies to insulin-like growth factor I receptor |
| US20100255538A1 (en) | 2001-01-05 | 2010-10-07 | Amgen Fremont Inc. | Antibodies to insulin-like growth factor i receptor |
| US7037498B2 (en) | 2001-01-05 | 2006-05-02 | Abgenix, Inc. | Antibodies to insulin-like growth factor I receptor |
| US7704501B2 (en) | 2001-07-30 | 2010-04-27 | Immunex Corporation | Antibodies binding to human ataxin-1-like polypeptide |
| US7084257B2 (en) | 2001-10-05 | 2006-08-01 | Amgen Inc. | Fully human antibody Fab fragments with human interferon-gamma neutralizing activity |
| US7521053B2 (en) | 2001-10-11 | 2009-04-21 | Amgen Inc. | Angiopoietin-2 specific binding agents |
| US7658924B2 (en) | 2001-10-11 | 2010-02-09 | Amgen Inc. | Angiopoietin-2 specific binding agents |
| US20080286284A1 (en) | 2001-10-23 | 2008-11-20 | Psma Development Company, Llc | Compositions of PSMA antibodies |
| US7338660B2 (en) | 2001-11-09 | 2008-03-04 | Abgenix, Inc. | Methods of treating cancer and enhancing immune responses with antibodies that bind CD40 |
| US7288251B2 (en) | 2001-11-09 | 2007-10-30 | Abgenix, Inc. | Antibodies to CD40 |
| US7618633B2 (en) | 2001-11-09 | 2009-11-17 | Amgen Fremont Inc. | Antibodies that bind CD40 and methods of treating cancer and enhancing immune responses |
| US7626012B2 (en) | 2001-11-09 | 2009-12-01 | Amgen Fremont Inc. | Nucleic acid molecules which encode antibodies that bind CD40 |
| US7563442B2 (en) | 2001-11-09 | 2009-07-21 | Abgenix, Inc. | Antibodies to CD40 and methods of treating cancer and enhancing immune responses |
| US20100098694A1 (en) | 2001-11-09 | 2010-04-22 | Amgen Fremont Inc. | Antibodies to cd40 |
| US7378091B2 (en) | 2001-12-03 | 2008-05-27 | Amgen Fremont Inc. | Antibodies against carbonic anhydrase IX (CA IX) tumor antigen |
| US7265212B2 (en) | 2001-12-03 | 2007-09-04 | Amgen Fremont Inc. | Anti-CD45RB antibodies |
| US7090844B2 (en) | 2001-12-28 | 2006-08-15 | Abgenix, Inc. | Use of antibodies against the MUC18 antigen |
| US6924360B2 (en) | 2001-12-28 | 2005-08-02 | Abgenix, Inc. | Antibodies against the MUC18 antigen |
| US7067131B2 (en) | 2001-12-28 | 2006-06-27 | Abgenix, Inc. | Methods for using anti-MUC18 antibodies |
| US7135174B2 (en) | 2002-01-07 | 2006-11-14 | Amgen Fremont, Inc. | Antibodies directed to PDGFD and uses thereof |
| US7141653B2 (en) | 2002-03-29 | 2006-11-28 | Schering Corporation | Human monoclonal antibodies to interleukin-5 |
| US7422742B2 (en) | 2002-03-29 | 2008-09-09 | Schering Corporation | Methods for using human monoclonal antibodies to interleukin-5 |
| US7718776B2 (en) | 2002-04-05 | 2010-05-18 | Amgen Inc. | Human anti-OPGL neutralizing antibodies as selective OPGL pathway inhibitors |
| US20100209435A1 (en) | 2002-04-05 | 2010-08-19 | Amgen Inc. | Human Anti-OPGL Neutralizing Antibodies As Selective OPGL Pathway Inhibitors |
| US7202343B2 (en) | 2002-08-19 | 2007-04-10 | Abgenix, Inc. | Antibodies directed to monocyte chemo-attractant protein-1 (MCP-1) and uses thereof |
| US20090214559A1 (en) | 2002-09-06 | 2009-08-27 | Amgen, Inc. | Therapeutic Human Anti-IL-1R1 Monoclonal Antibody |
| US7438910B2 (en) | 2002-09-06 | 2008-10-21 | Amgen Inc. | Therapeutic human anti-IL1-R1 monoclonal antibody |
| US20090191212A1 (en) | 2002-10-10 | 2009-07-30 | Amgen, Inc. | Angiopoietin-2 Specific Binding Agents |
| US7335743B2 (en) | 2002-10-16 | 2008-02-26 | Amgen Inc. | Human anti-IFN-γ neutralizing antibodies as selective IFN-γ pathway inhibitors |
| US7790859B2 (en) | 2002-10-16 | 2010-09-07 | Amgen Inc. | Human anti-IFN-γ neutralizing antibodies as selective IFN-γ pathway inhibitors |
| US20110045537A1 (en) | 2002-10-16 | 2011-02-24 | Amgen Inc. | Human Anti-IFN-gamma Neutralizing Antibodies as Selective IFN-gamma Pathway Inhibitors |
| US7285269B2 (en) | 2002-12-02 | 2007-10-23 | Amgen Fremont, Inc. | Antibodies directed to tumor necrosis factor |
| US20100111979A1 (en) | 2003-06-27 | 2010-05-06 | Amgen Fremont Inc. | Antibodies directed to the deletion mutants of epidermal growth factor receptor and uses thereof |
| US7628986B2 (en) | 2003-06-27 | 2009-12-08 | Amgen Fremont Inc. | Antibodies directed to the deletion mutants of epidermal growth factor receptor and uses thereof |
| US7736644B2 (en) | 2003-06-27 | 2010-06-15 | Amgen Fremont Inc. | Antibodies directed to the deletion mutants of epidermal growth factor receptor and uses thereof |
| US7795413B2 (en) | 2003-07-15 | 2010-09-14 | Amgen, Inc. | Nucleic acids encoding human anti-NGF neutralizing antibodies as selective NGF pathway inhibitors |
| US7601818B2 (en) | 2003-07-15 | 2009-10-13 | Amgen, Inc. | Human anti-NGF neutralizing antibodies as selective NGF pathway inhibitors |
| US20110040076A1 (en) | 2003-07-15 | 2011-02-17 | Amgen Inc. | Human Anti-NGF Neutralizing Antibodies as Selective NGF Pathway Inhibitors |
| US20090155274A1 (en) | 2003-07-15 | 2009-06-18 | Amgen Inc. | Human anti-ngf neutralizing antibodies as selective ngf pathway inhibitors |
| US7267960B2 (en) | 2003-07-25 | 2007-09-11 | Amgen Inc. | Antagonists and agonists of LDCAM and methods of use |
| US7498420B2 (en) | 2003-08-04 | 2009-03-03 | Amgen Fremont Inc. | Antibodies to c-Met |
| US7288253B2 (en) | 2003-08-08 | 2007-10-30 | Amgen Fremont, Inc. | Antibodies directed to parathyroid hormone (PTH) and uses thereof |
| US7318925B2 (en) | 2003-08-08 | 2008-01-15 | Amgen Fremont, Inc. | Methods of use for antibodies against parathyroid hormone |
| US7728113B2 (en) | 2003-09-10 | 2010-06-01 | Amgen Fremont Inc. | Methods of treating arthritic conditions with antibodies to M-CSF |
| US7592430B2 (en) | 2003-09-10 | 2009-09-22 | Amgen Fremont | Antibodies to M-CSF |
| US7326414B2 (en) | 2003-09-10 | 2008-02-05 | Warner-Lambert Company Llc | Antibodies to M-CSF |
| US7872113B2 (en) | 2003-11-07 | 2011-01-18 | Immunex Corporation | Nucleic acids encoding antibodies that bind interleukin-4 receptor |
| US7638606B2 (en) | 2003-11-07 | 2009-12-29 | Immunex Corporation | Antibodies that bind interleukin-4 receptor |
| US7371381B2 (en) | 2003-12-12 | 2008-05-13 | Amgen Inc. | Anti-galanin antibodies and uses thereof |
| US7932372B2 (en) | 2004-01-09 | 2011-04-26 | Amgen Fremont Inc. | Antibodies to MAdCAM |
| US20090263383A1 (en) | 2004-04-23 | 2009-10-22 | Amgen Inc. | Antibodies to angiogenesis inhibiting domains of CD148 |
| US7449555B2 (en) | 2004-04-23 | 2008-11-11 | Amgen Inc. | Antibodies of angiogenesis inhibiting domains CD148 |
| US7709611B2 (en) | 2004-08-04 | 2010-05-04 | Amgen Inc. | Antibodies to Dkk-1 |
| US20100040619A1 (en) | 2004-08-04 | 2010-02-18 | Amgen Inc. | Treatment methods using dkk-1 antibodies |
| US20060127393A1 (en) | 2004-08-04 | 2006-06-15 | Amgen Inc. | Antibodies to Dkk-1 |
| WO2006028936A2 (en) | 2004-09-02 | 2006-03-16 | Genentech, Inc. | Heteromultimeric molecules |
| US7423128B2 (en) | 2004-11-03 | 2008-09-09 | Amgen Fremont Inc. | Anti-properdin antibodies, and methods for making and using same |
| US20100047253A1 (en) | 2004-11-17 | 2010-02-25 | Amgen Inc. | Fully human monoclonal antibodies to il-13 |
| US7585500B2 (en) | 2004-11-17 | 2009-09-08 | Amgen Inc. | Fully human monoclonal antibodies to IL-13 |
| US7871611B2 (en) | 2004-12-22 | 2011-01-18 | Amgen Inc. | Compositions and methods relating to anti IGF-1 receptor antibodies |
| US20080292639A1 (en) | 2005-01-24 | 2008-11-27 | Amgen Inc. | Humanized Anti-Amyloid Antibody |
| US7906625B2 (en) | 2005-01-24 | 2011-03-15 | Amgen Inc. | Humanized anti-amyloid antibody |
| US7964193B2 (en) | 2005-01-26 | 2011-06-21 | Amgen Fremont Inc. | Antibodies against interleukin-1 β |
| US7566772B2 (en) | 2005-01-26 | 2009-07-28 | Amgen Fremont Inc. | Antibodies against interleukin-1β |
| US20090208489A1 (en) | 2005-03-24 | 2009-08-20 | Millennium Pharmaceuticals, Inc. Intellectual Property Group | Antibodies That Bind OV064 and Methods of Use Therefor |
| US7807159B2 (en) | 2005-04-25 | 2010-10-05 | Amgen Fremont Inc. | Antibodies to myostatin |
| US20110091455A1 (en) | 2005-04-25 | 2011-04-21 | Amgen Fremont Inc. | Antibodies to myostatin |
| US7592429B2 (en) | 2005-05-03 | 2009-09-22 | Ucb Sa | Sclerostin-binding antibody |
| US7872106B2 (en) | 2005-05-03 | 2011-01-18 | Amgen Inc. | Sclerostin-binding antibodies |
| US20080166352A1 (en) | 2005-07-18 | 2008-07-10 | Amgen Inc. | Human anti-B7RP1 Neutralizing Antibodies |
| US7868140B2 (en) | 2005-07-18 | 2011-01-11 | Amgen Inc. | Human anti-B7RP1 neutralizing antibodies |
| US7521048B2 (en) | 2005-08-31 | 2009-04-21 | Amgen Inc. | TRAIL receptor-2 polypeptides and antibodies |
| US20100197005A1 (en) | 2005-09-07 | 2010-08-05 | Amgen Fremont Inc. | Human monoclonal antibodies to activin receptor-like kinase-1 |
| US7537762B2 (en) | 2005-09-07 | 2009-05-26 | Amgen Fremont, Inc. | Human monoclonal antibodies to activin receptor-like kinase-1 |
| US20070196376A1 (en) | 2005-12-13 | 2007-08-23 | Amgen Fremont Inc. | Binding proteins specific for insulin-like growth factors and uses thereof |
| US7705130B2 (en) | 2005-12-30 | 2010-04-27 | U3 Pharma Gmbh | Antibodies directed to HER-3 and uses thereof |
| US20110165171A1 (en) | 2006-02-10 | 2011-07-07 | Amgen Inc. | Antibodies that bind par-2 |
| US7888482B2 (en) | 2006-02-10 | 2011-02-15 | Amgen Inc. | Antibodies that bind PAR-2 |
| US8263750B2 (en) | 2006-03-16 | 2012-09-11 | Amgen Inc. | Method for purifying a protein using protein-A affinity chromatography using an intermediate wash step |
| US7915391B2 (en) | 2006-04-24 | 2011-03-29 | Amgen Inc. | Humanized c-Kit antibody |
| US7728110B2 (en) | 2006-05-19 | 2010-06-01 | Amgen, Inc. | Antibodies to SARS coronavirus |
| US20090234106A1 (en) | 2006-09-08 | 2009-09-17 | Amgen Inc. | Anti-activin a antibodies and uses thereof |
| US20090041784A1 (en) | 2006-09-20 | 2009-02-12 | Amgen Inc. | Compositions and methods relating to glucagon receptor antibodies |
| US7947809B2 (en) | 2006-09-20 | 2011-05-24 | Amgen Inc. | Compositions and methods relating to glucagon receptor antibodies |
| US7767206B2 (en) | 2006-10-02 | 2010-08-03 | Amgen Inc. | Neutralizing determinants of IL-17 Receptor A and antibodies that bind thereto |
| US7939070B2 (en) | 2006-10-02 | 2011-05-10 | Amgen Inc. | IL-17 receptor A antigen binding proteins |
| US7833527B2 (en) | 2006-10-02 | 2010-11-16 | Amgen Inc. | Methods of treating psoriasis using IL-17 Receptor A antibodies |
| US7786284B2 (en) | 2006-10-02 | 2010-08-31 | Amgen Inc. | Polynucleotides encoding IL-17 receptor A antigen binding proteins |
| WO2008119353A1 (en) | 2007-03-29 | 2008-10-09 | Genmab A/S | Bispecific antibodies and methods for production thereof |
| US7867494B2 (en) | 2007-04-02 | 2011-01-11 | Amgen Fremont Inc. | Anti-IgE antibodies |
| US20110059063A1 (en) | 2007-06-29 | 2011-03-10 | Amgen Inc. | Antigen binding proteins that bind PAR-2 |
| US20110014201A1 (en) | 2007-07-24 | 2011-01-20 | Amgen Inc. | Il-18 receptor antigen binding proteins |
| US20090155164A1 (en) | 2007-08-21 | 2009-06-18 | Amgen, Inc. | Human c-fms antigen binding proteins |
| US20110027287A1 (en) | 2007-08-23 | 2011-02-03 | Amgen Inc. | Antigen binding proteins to proprotein convertase subtilisin kexin type 9 (pcsk9) |
| US20090238823A1 (en) | 2007-09-10 | 2009-09-24 | Amgen Inc. | Antigen binding proteins capable of binding thymic stromal lymphopoietin |
| US20110044986A1 (en) | 2007-12-21 | 2011-02-24 | Amgen Inc. | Anti-amyloid antibodies and uses thereof |
| US20090226447A1 (en) | 2008-02-20 | 2009-09-10 | Amgen, Inc. | Antibodies directed to angiopoietin-1 and angiopoietin-2 and uses thereof |
| US20110150888A1 (en) | 2008-05-01 | 2011-06-23 | Amgen Inc. | Anti-hepcidin antibodies and methods of use |
| US20100254975A1 (en) | 2009-03-20 | 2010-10-07 | Amgen Inc. | Alpha-4 beta-7 heterodimer specific |
| US20110135657A1 (en) | 2009-12-07 | 2011-06-09 | Amgen Inc. | Human antigen binding proteins that bind beta-klotho, fgf receptors and complexes thereof |
| WO2011131746A2 (en) | 2010-04-20 | 2011-10-27 | Genmab A/S | Heterodimeric antibody fc-containing proteins and methods for production thereof |
| WO2011147986A1 (en) | 2010-05-27 | 2011-12-01 | Genmab A/S | Monoclonal antibodies against her2 |
| WO2013060867A2 (en) | 2011-10-27 | 2013-05-02 | Genmab A/S | Production of heterodimeric proteins |
Non-Patent Citations (21)
| Title |
|---|
| AYBAYIMIR, J. IMMUNOL. METHODS, vol. 233, no. 1-2, 2000, pages 77 - 81 |
| CHOTHIA ET AL., NATURE, vol. 342, 1989, pages 877 - 883 |
| CHOTHIALESK, J. MOL. BIOL., vol. 196, 1987, pages 901 - 917 |
| FAHRNER ET AL., BIOTECHNOLOGY AND GENETIC ENGINEERING NEWS, vol. 18, 2001, pages 301 - 327 |
| FORD ET AL., J. CHROMATOGR. B, vol. 754, 2001, pages 427 - 435 |
| GAGNONPROTEIN A AFFINITY CHROMATOGRAPHY, PURIFICATION TOOLS FOR MONOCLONAL ANTIBODIES, 1996 |
| GRAMER ET AL., MABS, vol. 5, no. 6, 2013, pages 962 - 973 |
| HU ET AL., PROTEIN EXPR PURIF, vol. 186, October 2021 (2021-10-01), pages 105907 |
| JANEWAY ET AL.: "Immunobiology: The Immune System in Health and Disease", 1999, ELSEVIER SCIENCE LTD, article "Structure of the Antibody Molecule and the Immunoglobulin Genes" |
| KABAT ET AL.: "Sequences of Proteins of Immunological Interest", 1991, PUBLIC HEALTH SERVICE N.I.H |
| LABRIJN A F ET AL., PNAS, vol. 110, no. 13, 2013, pages 5145 - 5150 |
| LABRIJN ET AL., NATURE PROTOCOLS, vol. 9, no. 10, 2014, pages 2450 - 2463 |
| MOHAMMED JS: "A brief review on Ion Exchange Chromatography", PHARMATUTOR, vol. 1485, no. 2, 2017, pages 30 - 38 |
| RIDGWAY ET AL., PROTEIN ENG, vol. 9, 1996, pages 617 - 621 |
| RIDGWAY ET AL., PROTEIN ENG., vol. 9, 1996, pages 617 - 621 |
| RODRIGUEZ ET AL., J CHROMATOGR B ANALYT TECHNOL BIOMED LIFE SCI, vol. 1157, 10 November 2020 (2020-11-10), pages 122332 |
| SCHAEFER ET AL., PNAS, vol. 108, 2011, pages 11187 - 11192 |
| SHUKLA ET AL., J CHROMATOGR A, vol. 1171, no. 1-2, 9 November 2007 (2007-11-09), pages 22 - 8 |
| ULLAH, H, INTECH, 2012 |
| VALIDATED BIOSYSTEMS, 1996 |
| VOLA ET AL., CELL BIOPHYS., vol. 24-25, 1994, pages 27 - 36 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP7610638B2 (en) | Method for reducing host cell proteins in affinity chromatography - Patents.com | |
| JP7084301B2 (en) | Affinity chromatography purification method using low conductivity wash buffer | |
| JP7668351B2 (en) | Method for reducing host cell protein content in a protein purification process - Patents.com | |
| US12435106B2 (en) | Process of purification of protein | |
| KR20220069043A (en) | Methods and compositions comprising an anti-CTLA4 monoclonal antibody with reduced host cell protein and increased polysorbate-80 stability | |
| US20220119499A1 (en) | Method for the reduction of host cell proteins in affinity chromatography | |
| WO2025038600A1 (en) | Methods for reducing yellow color | |
| CN114729003A (en) | Method for increasing antibody yield in ion exchange chromatography process | |
| CN113444142A (en) | Application of arginine in ion exchange chromatography purification of hydrophobic protein | |
| HK40108969A (en) | Antibody variant with reduced biological activity | |
| CN118291432A (en) | A method for purifying protein using non-affinity chromatography capture technology | |
| HK1251584B (en) | Method for the reduction of host cell proteins in affinity chromatography | |
| HK1249122B (en) | Affinity chromatography purification with low conductivity wash buffer |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 24762532 Country of ref document: EP Kind code of ref document: A1 |