WO2025037604A1 - Internal composition for lipolysis and/or weight loss, method for determining effectiveness, and index for quality of plant extract - Google Patents
Internal composition for lipolysis and/or weight loss, method for determining effectiveness, and index for quality of plant extract Download PDFInfo
- Publication number
- WO2025037604A1 WO2025037604A1 PCT/JP2024/028767 JP2024028767W WO2025037604A1 WO 2025037604 A1 WO2025037604 A1 WO 2025037604A1 JP 2024028767 W JP2024028767 W JP 2024028767W WO 2025037604 A1 WO2025037604 A1 WO 2025037604A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- fat
- gliasperin
- bmi
- reducing
- internal composition
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Images
Classifications
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23L—FOODS, FOODSTUFFS OR NON-ALCOHOLIC BEVERAGES, NOT OTHERWISE PROVIDED FOR; PREPARATION OR TREATMENT THEREOF
- A23L33/00—Modifying nutritive qualities of foods; Dietetic products; Preparation or treatment thereof
- A23L33/10—Modifying nutritive qualities of foods; Dietetic products; Preparation or treatment thereof using additives
- A23L33/105—Plant extracts, their artificial duplicates or their derivatives
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/335—Heterocyclic compounds having oxygen as the only ring hetero atom, e.g. fungichromin
- A61K31/35—Heterocyclic compounds having oxygen as the only ring hetero atom, e.g. fungichromin having six-membered rings with one oxygen as the only ring hetero atom
- A61K31/352—Heterocyclic compounds having oxygen as the only ring hetero atom, e.g. fungichromin having six-membered rings with one oxygen as the only ring hetero atom condensed with carbocyclic rings, e.g. methantheline
- A61K31/353—3,4-Dihydrobenzopyrans, e.g. chroman, catechin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K36/00—Medicinal preparations of undetermined constitution containing material from algae, lichens, fungi or plants, or derivatives thereof, e.g. traditional herbal medicines
- A61K36/18—Magnoliophyta (angiosperms)
- A61K36/185—Magnoliopsida (dicotyledons)
- A61K36/48—Fabaceae or Leguminosae (Pea or Legume family); Caesalpiniaceae; Mimosaceae; Papilionaceae
- A61K36/484—Glycyrrhiza (licorice)
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/04—Anorexiants; Antiobesity agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/06—Antihyperlipidemics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/08—Drugs for disorders of the metabolism for glucose homeostasis
- A61P3/10—Drugs for disorders of the metabolism for glucose homeostasis for hyperglycaemia, e.g. antidiabetics
Definitions
- the present invention relates to an internal composition for lipolysis and/or weight loss, a method for determining efficacy, and an indicator of the quality of a plant extract.
- Body fat occurs when energy intake is in excess of energy expenditure, and the excess accumulates as neutral fat in white fat cells. Excessive fat accumulation leads to obesity and weight gain, which not only causes various lifestyle-related diseases but also poses a major cosmetic problem. Obesity with a large accumulation of visceral fat has been linked to conditions such as insulin resistance and arteriosclerosis, while obesity with a large accumulation of subcutaneous fat is a major concern for both men and women from the perspective of cosmetic care.
- Patent Document 1 a mixture prepared by combining salmon milt extract, brewer's yeast extract, young barley leaf extract, and chicken collagen (chicken collagen) has an excellent lipolysis promoting effect
- Patent Document 2 that at least one of an extract of Betulaceae (Betulaceae) and an extract of Kumazasa (Poaceae) improves obesity and prevents the increase of adipose tissue by promoting the decomposition of fat in whole body or local adipose tissue
- Patent Document 2 certain plants such as Juniperus communis or their extracts are useful as medicines, foods, or cosmetics that promote the decomposition of neutral fats accumulated in adipose tissue and exert slimming effects such as suppressing, preventing, or improving obesity
- Patent Document 4 also discloses that a composition combining a licorice hydrophobic extract with an antioxidant component and the like has the effects of suppressing body fat accumulation, promoting body fat decomposition, and promoting energy production, but a sufficient lipolysis promoting effect is not obtained with licorice hydrophobic extract alone.
- the problem that the present invention aims to solve is to provide a new formulation that is excellent in promoting lipolysis, reducing visceral fat and body fat, reducing body weight, and improving BMI.
- Another object of the present invention is to provide a new formulation that is excellent in improving insulin resistance, reducing cholesterol levels, etc.
- gliasperin B a specific component in licorice extract, has an excellent effect of promoting lipolysis. They have also found that a preparation containing licorice extract containing gliasperin B has an excellent effect of promoting lipolysis, as well as an effect of reducing visceral fat and body fat, a weight loss effect, an effect of improving BMI, an effect of improving insulin resistance, and an effect of reducing cholesterol levels. They have also found that the content of gliasperin B in a plant extract is an indicator of the effectiveness of the plant extract for lipolysis, weight loss, etc.
- the gist of the present invention is as follows.
- An internal composition containing gliasperin B which is used for one or more of the following: decomposing fat, burning fat, assisting in fat consumption, improving BMI, assisting in lowering BMI, reducing abdominal fat, reducing visceral fat, reducing body fat, reducing waist circumference, improving insulin resistance, and reducing LDL cholesterol levels.
- the internal composition described in [1] or [2] which is used for adults with a BMI of 23 kg/ m2 or more and less than 30 kg/ m2 .
- [7] A method for determining the effectiveness of a plant extract for lipolysis and/or weight loss in a living body, using the content of gliasperin B as an indicator. [8] The method for determining the effectiveness of a compound according to [7] above, wherein the plant is licorice. [9] An indicator of the quality of a plant extract, the quality being related to effectiveness in lipolysis and/or weight loss in a living body, characterized in that the indicator consists of gliasperin B. [10] The indicator described in [9], wherein the plant is licorice.
- the internal composition of the present invention contains gliasperin B, preferably as an active ingredient, functional ingredient, indicator ingredient or contributing ingredient, and thus has an excellent lipolysis promoting effect and a remarkable weight loss effect.
- gliasperin B preferably as an active ingredient, functional ingredient, indicator ingredient or contributing ingredient
- the internal composition of the present invention can also improve insulin resistance and reduce sterol levels. Therefore, the internal composition of the present invention can prevent and improve obesity and lifestyle-related diseases in the long term, and can achieve the maintenance and improvement of health.
- the content of gliasperin B in a plant extract is an indicator of the effectiveness of the plant extract for lipolysis, weight loss, etc., it becomes possible to efficiently select a highly effective plant extract.
- FIG. 1 shows the lipolysis promoting effect of licorice extract and gliasperin B.
- FIG. 1 shows the effect of licorice extract on body weight, BMI and visceral fat level.
- the internal composition of the present invention contains gliasperin B. Although not limited thereto, it is preferable that gliasperin B is contained as an active ingredient, a functional ingredient, an indicator ingredient or a contributing ingredient. In addition to gliasperin B, which is an essential ingredient, the internal composition of the present invention may contain other ingredients within a range that does not impair the effects of the present invention.
- the internal composition of the present invention contains gliasperin B and other ingredients, and the form of the internal composition of the present invention, etc. will be described below.
- an active ingredient refers to a pharmacologically active ingredient or a physiologically active ingredient contained in a drug, quasi-drug, etc. that exerts a desired effect.
- functional ingredients refer to ingredients contained in internal compositions such as foods that contribute to a specific health purpose. Specifically, the mechanism of action related to functionality has been considered through in vitro tests, in vivo tests, or clinical trials (human tests), and the ingredients can be directly or indirectly qualitatively and quantitatively confirmed.
- Functional foods are provided to consumers with a display of functions based on the effects of the ingredients contained in the functional food, and when providing such foods to consumers as functional foods, the content of the functional ingredients and the method of quantifying them must be notified.
- index ingredient refers to an extract or essence that is considered to be a functional ingredient when a specific ingredient that can explain part of the scientific basis of functionality is known, but the functionality cannot be fully explained by the specific ingredient alone, and the mechanism of action related to the functionality to be displayed has been considered for at least one ingredient through in vitro tests, in vivo tests, or clinical tests (human tests).
- a contributing ingredient refers to a substance that, when incorporated, directly or indirectly affects the physiological functions of the body.
- the equivalent of a contributing ingredient could be, but is not limited to, a "contributing ingredient,” a "functional contributing ingredient,” or an “indicator ingredient.”
- the equivalent of a contributing ingredient in medicines and quasi-drugs is an "active ingredient.”
- this concept also includes ingredients that are implicitly linked to an effect and emphasized to consumers in the form of "Contains XX ingredient. For burning calories.”
- Glyasperin B (hereinafter also referred to as "GB") is an isoflavanone derivative represented by the following formula: It is also represented as 3-(2,4-dihydroxyphenyl)-5-hydroxy-2,3-dihydro-6-(3-methyl-2-butenyl)-7-methoxy-4H-1-benzopyran-4-one, 3-(2,4-dihydroxyphenyl)-5-hydroxy-6-(3-methyl-2-butenyl)-7-methoxy-3,4-dihydro-2H-1-benzopyran-4-one, or 3-(2,4-dihydroxyphenyl)-5-hydroxy-7-methoxy-6-(3-methylbut-2-en-1-yl)-3,4-dihydro-2H-1-benzopyran-4-one.
- Gliasperin B is a component that has been confirmed to be contained in licorice extract, and the gliasperin B in the present invention is preferably derived from licorice extract. It has been known that licorice extract has a lipolysis promoting effect, and there have been documents suggesting that glabridin, which is contained in large amounts in licorice extract, may have a lipolysis promoting effect (see Patent Document 4, etc.). However, it was not clear which component in licorice extract has the lipolysis promoting effect. The present inventors have discovered for the first time that, of the many components present in licorice extract, gliasperin B has a lipolysis promoting effect.
- the gliasperin B in the present invention may be derived from licorice or may be a chemically synthesized product. In this specification, when we say that it contains gliasperin B, this also includes the case where it contains a plant such as licorice that contains gliasperin B.
- the type of licorice is not particularly limited, but may be any plant of the genus Glycyrrhiza in the family Fabaceae, such as Glycyrrhiza uralensis (G. uralensis Fisch. et DC; ural licorice), Glycyrrhiza inflata (G. inflata BAT.; butterfly licorice), Glycyrrhiza glabra (G. glabra L.; licorice), Glycyrrhiza glabra L. ( ... These include G. glabra L. var glandu lifera Regel et Herder (Chinese tallow tree), G. aspera, G. echinata L. (Chinese tallow tree), and G. pallidiflora Maxim (Chinese tallow tree).
- Glycyrrhiza uralensis G. uralensis Fisch. et DC
- ural licorice
- Methods for obtaining gliasperin B derived from licorice include a method of purifying it from a licorice extract.
- the licorice extract is a licorice extract obtained by extracting the whole licorice plant or a crushed part of the plant (e.g., roots, stems, stolons, leaves, flowers, fruits, etc.) with a solvent, and the obtained licorice extract may be spray-dried or freeze-dried.
- the extraction solvent include water, alcohols such as methanol and ethanol, and mixed solvents of water and alcohols or ketones such as acetone. Of these, the extraction solvent is preferably water, alcohol, or aqueous alcohol, and more preferably hot water, ethanol, or aqueous ethanol.
- the alcohol concentration of the hydrous alcohol is 0.1% to 99.9% by mass, preferably 10% to 99.9% by mass, more preferably 30% to 70% by mass, even more preferably 40% to 60% by mass, and particularly preferably 50% by mass.
- it is 0.1% (v/v) to 99.9% (v/v), preferably 10% (v/v) to 99.9% (v/v), more preferably 30% (v/v) to 70% (v/v), even more preferably 40% (v/v) to 60% (v/v), and particularly preferably 50% (v/v).
- Distilled water is added to the dried licorice extract to dissolve it, and an aqueous licorice extract solution is prepared.
- An organic solvent such as ethyl acetate is added in an amount equal to half the volume of the aqueous solution, and the solution is mixed to separate into two layers, from which the organic solvent layer such as ethyl acetate is collected. This operation is repeated several times, preferably about three times.
- Anhydrous sodium sulfate is added to the obtained organic solvent layer, and the organic fraction can be obtained by filtering.
- the content of gliasperin B in the internal composition of the present invention is 0.00001% to 50% by mass, preferably 0.0001% to 20% by mass, more preferably 0.0005% to 10% by mass, and even more preferably 0.001% to 5% by mass.
- the internal composition of the present invention may contain, in addition to gliasperin B, a carrier, an excipient, a solvent, and other optional components within a range that does not impair the effects of the present invention.
- the internal composition of the present invention contains gliasperin B
- it may contain gliasperin B purified from licorice extract as described above or a chemically synthesized gliasperin B, or it may contain licorice extract so that the final content of gliasperin B in the internal composition is within the above numerical range.
- the internal composition of the present invention preferably contains gliasperin B within the above numerical range and does not contain any licorice-derived components other than gliasperin B, or, if it contains any licorice-derived components other than gliasperin B, the ratio of the gliasperin B content to the glycyrrhizic acid content (the value obtained by dividing the gliasperin B content by the glycyrrhizic acid content) is 0.001 or more, using glycyrrhizic acid, which is a quantitative indicator of licorice extract in the Japanese Pharmacopoeia, as a comparison standard.
- the internal composition of the present invention can be used as a food or drink, a functional food, a food for specified health uses, a nutrient-functional food, a cosmetic, a quasi-drug, a medicine, or a cosmetic agent, etc., to efficiently break down fat and reduce body fat, such as subcutaneous fat and visceral fat.
- the internal composition of the present invention when preparing the internal composition of the present invention as a food or beverage, in addition to gliasperin B, sweeteners, colorants, preservatives, thickeners, stabilizers, gelling agents, pasting agents, antioxidants, colorants, bleaching agents, mold inhibitors (mold inhibitors), yeast food, gum base, flavorings, acidulants, seasonings, emulsifiers, pH adjusters, kansui, leavening agents, nutritional enhancers, and other food and beverage ingredients can be mixed and prepared into the desired form.
- the internal composition of the present invention is made into a food or beverage form, there are no particular limitations on the form.
- Examples include supplement-type foods such as gels, granules, fine granules, capsules, tablets, powders, liquids, and semisolids; beverages such as carbonated drinks, soft drinks, milk drinks, alcoholic drinks, fruit juice drinks, teas, and nutritional drinks; powdered drinks such as powdered juice and powdered soup; confectioneries such as gum, tablets, candies, cookies, gummies, rice crackers, biscuits, and jellies; bread, noodles, cereals, jams, and seasonings.
- supplement-type foods such as gels, granules, fine granules, capsules, tablets, powders, liquids, and semisolids
- beverages such as carbonated drinks, soft drinks, milk drinks, alcoholic drinks, fruit juice drinks, teas, and nutritional drinks
- powdered drinks such as powdered juice and powdered soup
- confectioneries such as gum, tablets, candies, cookies, gummies, rice crackers, biscuits, and jellies
- These foods are used as foods and beverages to efficiently break down fat and reduce body fat such as subcutaneous fat and visceral fat, and can be used, for example, as nutraceuticals such as dietary supplements, functional foods, foods for specified health uses, and foods for the sick, in addition to general foods and beverages.
- the internal composition of the present invention is prepared as a medicine (including quasi-drugs), in addition to gliasperin B, other medicinal ingredients, pharma- ceutical acceptable carriers, additives, etc. may be optionally blended, if necessary.
- pharma- ceutical acceptable carriers and additives include binders, disintegrants, lubricants, wetting agents, buffers, preservatives, and flavorings.
- its form is not particularly limited. Examples include injections, external preparations, inhalants, suppositories, films, lozenges, liquids, powders, tablets, granules, capsules, syrups, eye drops, eyewashes, and nasal drops.
- forms suitable for oral administration i.e., internal medicines
- forms suitable for oral administration include lozenges, liquids, powders, tablets, granules, capsules, and syrups.
- These medicines are used as medicines to efficiently break down fat and reduce body fat, including subcutaneous fat and visceral fat.
- Gliasperin B contained in the internal composition of the present invention can also be used as a cosmetic (including functional cosmetic) or topical quasi-drug.
- a cosmetic or topical quasi-drug a pharma- ceutical or cosmetically acceptable carrier (water, oily components, etc.) is added to Gliasperin B and the cosmetic is prepared in the desired form.
- the cosmetic is not particularly limited in form as long as it can be applied to the skin. Examples include liquid, emulsion, powder, solid, suspension, cream, ointment, mousse, granule, tablet, gel, jelly, paste, gel, aerosol, spray, liniment, pack, etc. These cosmetic products are used as cosmetic products for efficiently breaking down fat and reducing subcutaneous fat, etc.
- the dosage and application amount of the internal composition of the present invention can be appropriately determined depending on the age, body weight, health condition, degree of obesity, disease state, etc. of the user, but is not limited to, and can be, in terms of the total amount of gliaperin B, 0.1 ⁇ g or more, 0.5 ⁇ g or more, 1 ⁇ g or more, 5 ⁇ g or more, 10 ⁇ g or more, 12 ⁇ g or more per day for an adult, and can be 5000 ⁇ g or less, 1000 ⁇ g or less, 500 ⁇ g or less, 100 ⁇ g or less, 60 ⁇ g or less, 20 ⁇ g or less, 15 ⁇ g or less, etc.
- the total amount of gliasperin B taken per day by an adult is 0.1 ⁇ g to 5000 ⁇ g, preferably 0.5 ⁇ g to 1000 ⁇ g, more preferably 1 ⁇ g to 500 ⁇ g, even more preferably 1 ⁇ g to 200 ⁇ g, even more preferably 1 ⁇ g to 100 ⁇ g, particularly preferably 5 ⁇ g to 100 ⁇ g, even more particularly preferably 5 ⁇ g to 60 ⁇ g, even more particularly preferably 10 ⁇ g to 50 ⁇ g, and most preferably 10 to 20 ⁇ g.
- an effective amount refers to the dosage at which the desired effect is observed, whether it is a direct or indirect action.
- the effective amount varies depending on age and body weight, but can be, for example, 1 ⁇ g or more, 3 ⁇ g or more, 5 ⁇ g or more, 7 ⁇ g or more, 10 ⁇ g or more, or 12 ⁇ g or more per day for an adult, calculated as the total amount of gliasperin B described above, or 1 mg or less, 500 ⁇ g or less, 200 ⁇ g or less, 100 ⁇ g or less, 60 ⁇ g or less, 50 ⁇ g or less, or 20 ⁇ g or less.
- the total amount of gliasperin B per day for an adult is 1 ⁇ g to 1 mg, preferably 1 ⁇ g to 500 ⁇ g, more preferably 1 ⁇ g to 200 ⁇ g, even more preferably 1 ⁇ g to 100 ⁇ g, even more preferably 5 ⁇ g to 100 ⁇ g, particularly preferably 5 ⁇ g to 60 ⁇ g, even more particularly preferably 10 ⁇ g to 50 ⁇ g, even more particularly preferably 10 ⁇ g to 20 ⁇ g, and most preferably 14 ⁇ g.
- the internal composition of the present invention is preferably used for adults with a BMI of 23 kg/m2 or more and less than 30 kg/ m2 , and more preferably for adults with a BMI of 23 kg/ m2 or more and less than 25 kg/ m2 .
- a BMI of 18.5 kg/ m2 or more and less than 25 kg/ m2 is considered to be normal body weight
- a BMI of 25 kg/ m2 or more and less than 30 kg/ m2 is considered to be obesity level 1.
- the internal composition of the present invention is preferably used for adults with a so-called normal body weight to slightly obese (obesity level 1), and is particularly preferably used for adults who are not classified as obese but are slightly obese from a normal body weight.
- BMI Body Mass Index
- BMI Body Mass Index
- the internal use composition of the present invention is preferably used for adults with a visceral fat area of 100 cm2 or more.
- the visceral fat area can be calculated in the usual manner by performing abdominal CT scan and taking multiple slice images at intervals of 1 cm or more centered on the navel.
- Another embodiment of the present invention is a weight loss agent, an agent for reducing visceral fat and body fat, an agent for improving BMI, an agent for improving insulin resistance, or an agent for reducing cholesterol levels, which contains gliasperin B as an active ingredient.
- the present invention also includes the above-mentioned weight loss agent, visceral fat/body fat reduction agent, BMI improvement agent, insulin resistance improvement agent, or cholesterol level reduction agent, which contain gliasperin B, and thus have excellent fat decomposition promoting effects and a remarkable weight loss effect.
- the weight loss agent, visceral fat/body fat reduction agent, BMI improvement agent, insulin resistance improvement agent, or cholesterol level reduction agent of the present invention fat in fat cells can be efficiently decomposed and body fat such as subcutaneous fat and visceral fat can be reduced, resulting in a remarkable weight loss effect and BMI improvement effect. Insulin resistance improvement and sterol level reduction effects can also be obtained.
- the weight loss agent, visceral fat/body fat reduction agent, BMI improvement agent, insulin resistance improvement agent, or cholesterol level reduction agent of the present invention can prevent and improve obesity and lifestyle-related diseases in the long term, and can achieve the maintenance and promotion of health.
- the weight loss agent, visceral fat/body fat reducing agent, BMI improving agent, insulin resistance improving agent, or cholesterol level reducing agent of the present invention has the same configuration as the above-mentioned internal composition of the present invention containing gliasperin B as an active ingredient, the specific explanation can be applied as is to the explanation in the section on the internal composition of the present invention.
- the present invention also includes a method for determining the effectiveness of a plant extract in lipolysis and/or weight loss in a living body, using the content of gliasperin B as an index.
- a preparation containing gliasperin B as an active ingredient has an excellent lipolysis promoting effect and a remarkable weight loss effect.
- body fat such as subcutaneous fat and visceral fat
- BMI improvement effect it is possible to improve insulin resistance and reduce sterol levels.
- the above plant extract is not particularly limited as long as it contains gliasperin B, but licorice extract is preferable.
- the determination method of the present invention includes a step of measuring the content of gliasperin B in the plant extract (hereinafter also referred to as the "measurement step”). It is also preferable that the method includes a step of comparing the obtained amount of gliasperin B with a standard amount of gliasperin B (hereinafter also referred to as the “comparison step”).
- the method for measuring the content of gliasperin B in the plant extract is not particularly limited, and can be measured by a method known to those skilled in the art.
- Examples include Western blotting and ELISA methods.
- an anti-gliasperin B antibody that specifically binds to gliasperin B is used, and the content of gliasperin B can be measured with high accuracy.
- the intensity of the resulting gliasperin B-specific blot can be compared with the intensity of a standard specimen (positive control) of known concentration, thereby calculating the gliasperin B content in the sample.
- Gliasperin B used as a standard in the determination method of the present invention can be obtained, for example, by extraction from a natural source, by expression of a recombinant nucleic acid encoding gliasperin B, or by chemical synthesis.
- the comparison step involves comparing the content of gliasperin B in the extract to be evaluated with the content of the standard gliasperin B.
- the amount of gliasperin B to be used as the standard can be appropriately selected depending on the purpose and subject of the evaluation.
- the present invention also includes an invention relating to an index of the quality of a plant extract, the quality being related to the effectiveness of the plant extract in lipolysis and/or weight loss, and characterized in that the index is made of gliasperin B.
- a preparation containing gliasperin B as an active ingredient has an excellent lipolysis promoting effect and a remarkable weight loss effect.
- the preparation can efficiently decompose fat in adipocytes and reduce body fat such as subcutaneous fat and visceral fat, resulting in a remarkable weight loss effect and BMI improvement effect.
- the preparation can improve insulin resistance and reduce sterol levels.
- gliasperin B Since the above effects increase depending on the content of gliasperin B, it is possible to determine the effectiveness of the plant extract in lipolysis and/or weight loss in a living body by using gliasperin B in the plant extract as an index. Licorice is preferable as the plant in the plant extract.
- the indicator of the quality of the plant extract of the present invention corresponds to gliasperin B in the above-mentioned method for determining the effectiveness of the plant extract for lipolysis and/or weight loss.
- the specific explanation of the indicator of the present invention can be applied to the explanation of the above-mentioned determination method.
- Test food composition, packaging, etc.
- the test food contained 100 mg of licorice extract (manufactured by MG Pharma Co., Ltd.; derived from Ural licorice) per capsule, and was composed of other excipients.
- the control food did not contain licorice extract, and was composed of excipients colored with caramel coloring. All test foods were in the form of white hard capsules, packaged in aluminum pouches of 30 capsules each, and an identification code was displayed on the test foods. The indistinguishability of the test foods and their packaging was reviewed and approved by the IRB.
- the nutritional components and related components of the test foods are shown in Table 1 below.
- the amount of gliasperin B contained in the test foods was 14 ⁇ g per recommended daily intake.
- Subjects The target number of subjects in this study was calculated to be 34 per group, for a total of 68 subjects, assuming that the effect size of the test food is large (0.8) in a parallel group comparison study between two groups, the significance level is 0.05, and the detection power is 80-90%, so that 26-35 subjects per group are required. In addition, the number of entries was set at 80 (40 per group) in consideration of cases that may drop out or be discontinued during the study. Subjects were recruited from volunteers registered with the study contracting institution, and 81 subjects (50 men, 31 women) who met the following inclusion criteria, did not violate the exclusion criteria, and were deemed appropriate for participation in this study by the study principal physician were included as subjects.
- Selection Criteria For the screening test, participants were required to meet the following conditions: (1) Men and women aged 20 years or older and younger than 65 years, (2) individuals with a BMI of 23.0 kg/ m2 or more and less than 30.0 kg/ m2 , (3) individuals with a visceral fat area of 100 cm2 or more, (4) individuals who have been fully informed of the purpose and contents of this study, are capable of consent, have a full understanding of the study, voluntarily volunteer to participate, and can agree to participate in this study in writing.
- Exclusion Criteria (1) Persons suffering from or with a history of serious cardiovascular, hepatic, renal, respiratory, endocrine or metabolic disorders; (2) Persons receiving treatment for chronic diseases such as dyslipidemia, hypertension or diabetes; (3) Persons with mental illnesses such as depression, schizophrenia or bulimia nervosa; (4) Persons who regularly use medicines or quasi-drugs that may affect the evaluation of this study, such as body fat, triglycerides, body weight, cholesterol and energy metabolism; (5) Persons who regularly use supplements or health foods (specified health foods, functional foods, etc.) that may affect the evaluation of this study, such as body fat, triglycerides, body weight, cholesterol and energy metabolism; (6) Persons who may be allergic to the test foods.
- Study protocol This study was a randomized, placebo-controlled, double-blind, parallel-group comparative study. Tests were conducted at the start of intake of the test food, and after 4 weeks, 8 weeks, and 12 weeks of intake. A random number was generated for randomization by the allocation manager, who was not involved in the implementation of the study, and the subjects were assigned to the subjects who were enrolled by the study director. At this time, it was confirmed that there were no significant differences between the groups in the allocation factors of sex and age at the time of consent acquisition, and visceral fat area, BMI, hip circumference, and waist circumference at the time of screening examination.
- test food for the test food group test food for the test food group
- control food for the control food group control food for the control food group
- subjects were instructed to maintain normal eating and drinking habits as much as possible, and were prohibited from consuming health foods (foods for specified health uses, foods with functional claims, etc.) that may affect the study's evaluations of body fat, neutral fat, body weight, cholesterol, energy metabolism, etc., as well as grapefruit juice.
- subjects were instructed to maintain normal alcohol intake habits, and were prohibited from drinking amounts that deviated from normal intake.
- subjects were prohibited from consuming alcohol the day before the study, and were prohibited from consuming anything other than water after 9 p.m.
- subjects were prohibited from consuming anything other than water four hours before the abdominal CT scan.
- Abdominal CT scans were performed using a Discovery 710 or Discovery MIDR (GE Healthcare Japan, Ltd.), with three slices taken at 1 cm intervals centered on the navel, and the average values of visceral fat area, subcutaneous fat area, and total fat area were calculated. If the kidneys or ilium were included in the image taken at the navel position, a second image was taken centered on the fourth lumbar vertebra.
- Fatigue and sleep evaluation Fatigue was evaluated by VAS at the start of intake, and 4, 8, and 12 weeks after intake, when the subjects woke up at home.
- Sleep was evaluated by the OSA sleep questionnaire MA version at the start of intake, and 4, 8, and 12 weeks after intake, when the subjects woke up at home.
- the primary outcome was visceral fat area in abdominal CT scans. Secondary outcome measures were body weight, BMI, subcutaneous fat area in abdominal CT scans, total fat area, body fat percentage, body fat mass, muscle mass, body fat mass by region, basal metabolic rate, waist circumference, hip circumference, waist/hip ratio, VAS (feeling of fatigue), OSA Sleep Questionnaire MA version, ketone body fraction in blood tests, T-Cho, LDL-Cho, HDL-Cho, TG, blood glucose, HbA1c, insulin, and glycoalbumin.
- the average value and standard deviation of the values at each test time and the change amount from before the start of intake to each test time were tabulated by group, and an unpaired t-test was performed on the difference in the average value between the test food group and the control food group at each test time to compare the differences between the groups.
- Dunnett's multiple comparison test was performed on the difference in the average value between before the start of intake and each test time in each test food group to compare the differences due to the intake period of the test food.
- abdominal CT a paired t-test was performed to compare the differences due to the intake period of the test food. Note that no correction for multiplicity was performed for the paired t-test.
- the efficacy analysis set was set at 76 patients (38 in the test food group and 38 in the control food group).
- two patients in the control food group were excluded from the analysis for only those items due to issues with the reliability of the data for some evaluation items.
- hemolysis was observed in the blood sample, so LDH, K, and insulin in the blood test 4 weeks after intake were excluded from the analysis.
- food intake on the day of the test was suspected, so blood glucose and insulin in the blood test 8 weeks after intake were excluded from the analysis.
- the subject background of the subjects in the efficacy analysis is shown in Table 2.
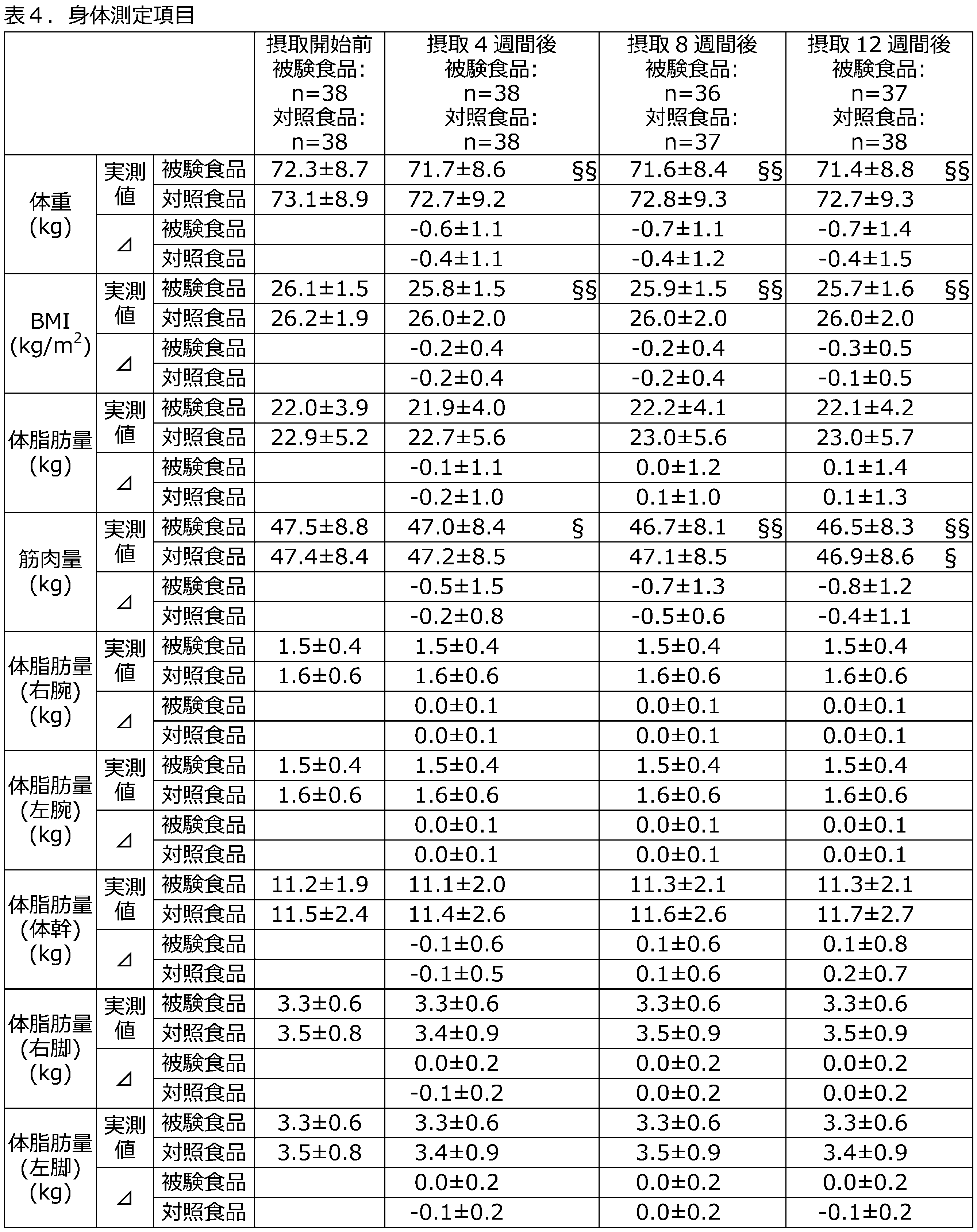
- Table 4 shows changes in body weight, BMI, body fat mass, muscle mass, body fat mass by region, waist circumference, and hip circumference in the PPS, as well as blood pressure and pulse rate in the FAS.
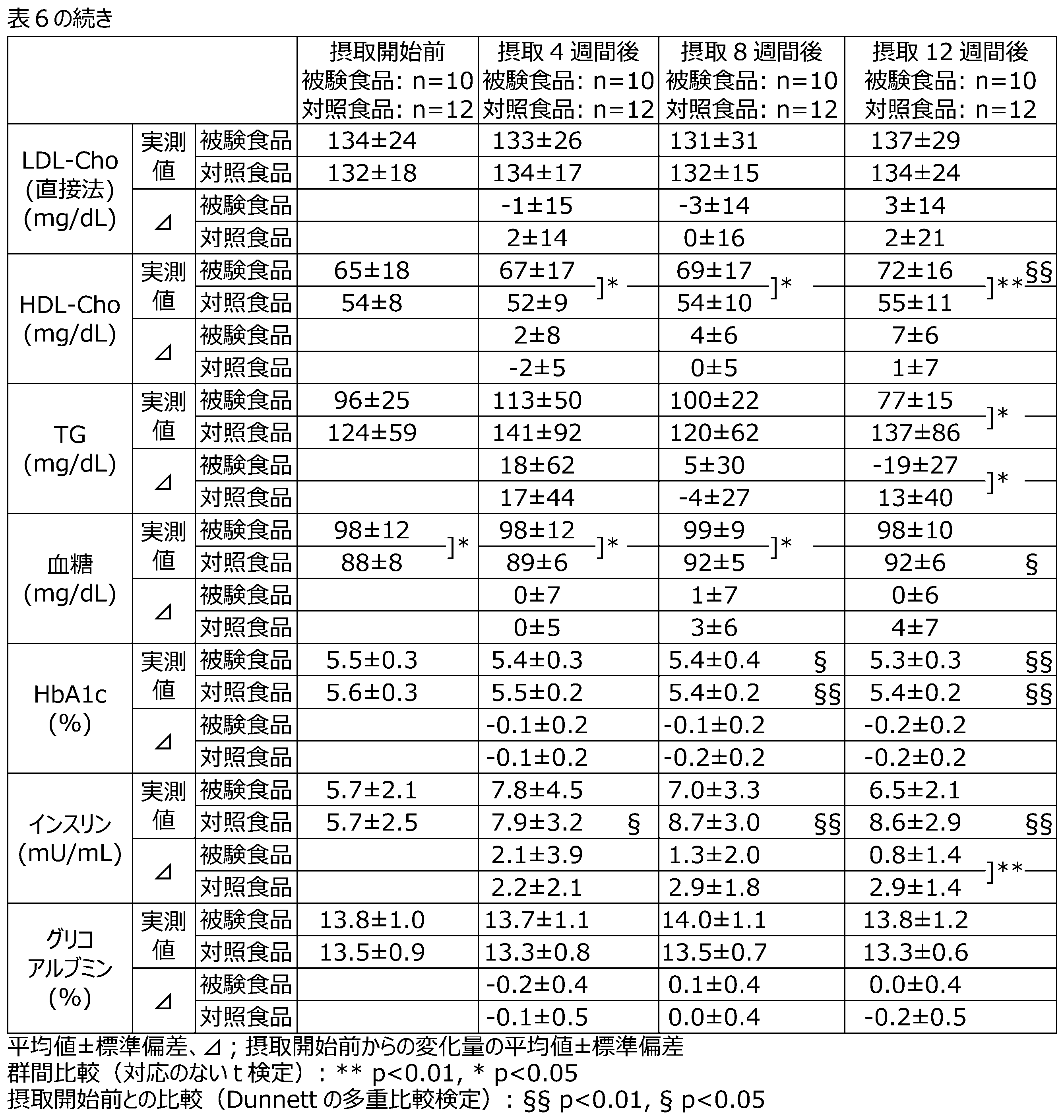
- Table 5 shows the changes in blood test items (secondary evaluation items) in the PPS.
- T-Cho the test food group showed a significant suppression of the change from before the start of intake compared to the control food group after 4 weeks of intake.
- blood glucose the test food group showed significantly higher values than the control food group before the start of intake, and showed significantly higher values even after 4 weeks of intake, but no difference was observed thereafter.
- insulin levels the test food group showed a significant decrease compared to the control food group.
- no clinically significant changes were observed in any of the subjects for other blood test items and urine test items in the FAS.
- the test food group showed a significant increase in the change from before the start of intake for total ketone bodies, acetoacetate, and 3-hydroxybutyrate after 12 weeks of intake compared to the control food group.
- a significant increase was observed in HDL-Cho after 4, 8, and 12 weeks of intake.
- TG a significant low was observed after 12 weeks of intake, and a significant decrease in the amount of change from before the start of intake was observed.
- blood glucose a significant high was observed before intake, and 4 and 8 weeks after intake.
- insulin a significant increase in the amount of change from before the start of intake was suppressed after 12 weeks of intake.
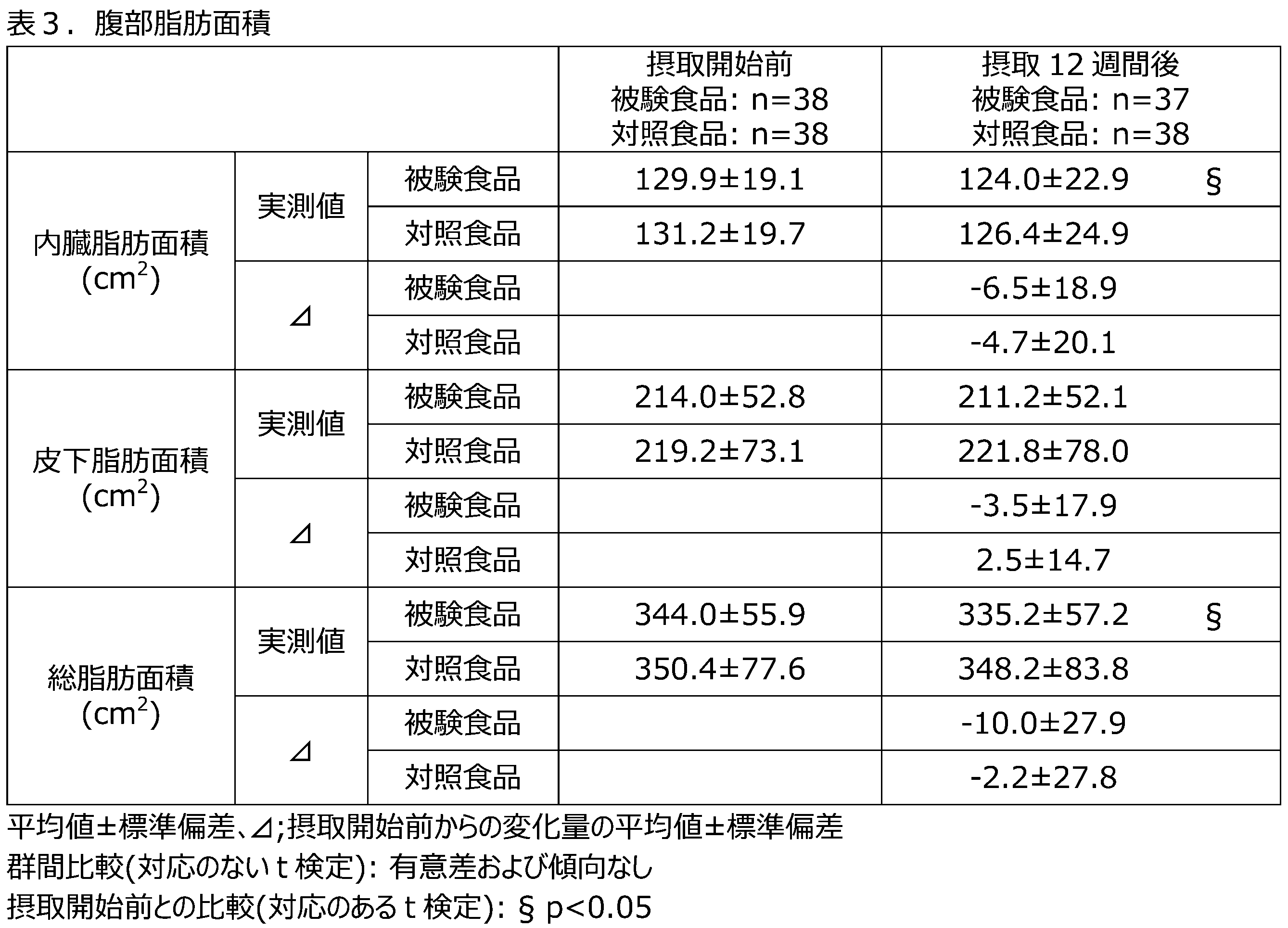
- the test food group showed significantly lower visceral fat area after 12 weeks of intake compared to the control food group, and the changes in subcutaneous fat area and total fat area from before the start of the study, as well as the changes in body weight and BMI from before the start of the study, were significantly reduced, indicating that licorice extract has a body fat reducing effect in humans as well, and associated decreases in body weight and BMI were observed.
- gliasperin B in licorice extract contributes nearly 100% to TG decomposition in fat cells. Based on the above, it is believed that the body fat reducing effect of ingesting licorice extract is due to gliasperin B.
- Licorice extract-containing foods have been shown to have a body fat reducing effect in healthy adult men and women who are slightly obese (BMI 23 kg/ m2 or more and less than 25 kg/ m2 , abdominal visceral fat area 100 cm2 or more).
- BMI 23 kg/ m2 or more and less than 25 kg/ m2 , abdominal visceral fat area 100 cm2 or more part of the mechanism of action is the decomposition of TG in fat cells and the accompanying enhancement of fatty acid consumption ( ⁇ -oxidation).
- ⁇ Test 2> Five-week-old male C57BL/6J mice were divided into groups of six and fed MF (Oriental Yeast), high-fat, high-carbohydrate powdered feed (hereinafter abbreviated as HFS, D12079BM, Research Diet), or HFS mixed with licorice extract for 8 weeks. After 8 weeks of ingestion, the body weight and visceral fat weight (the sum of epididymal fat weight, perirenal fat weight, and mesenteric fat weight) were measured.
- MF Oriental Yeast
- HFS high-fat, high-carbohydrate powdered feed
- D12079BM high-carbohydrate powdered feed
- HFS high-carbohydrate powdered feed
- licorice extract HFS mixed with licorice extract
- the licorice extract was obtained by subjecting the powder obtained by crushing the root and stolon of licorice to a 50% (v/v) ethanol extraction process, drying the liquid by an evaporator and freeze-drying, and then crushing the liquid. These were each mixed with the feed at 0.3% and fed to the mice.
- the amount of gliasperin B contained in the licorice extract, and the body weight gain and visceral fat weight of the mice are shown in Table 7.
- the gliasperin B (GB) obtained here was used as a test substance in the following test using mouse preadipocytes 3T3-L1.
- the above licorice extract was obtained by the following method. That is, the stem, root, and stolons of licorice were crushed to obtain licorice powder. 50 g of licorice powder was extracted with 500 mL of 50% aqueous ethanol at 20°C, and the resulting extract was frozen and dried in an evaporator to obtain a 50% aqueous ethanol extract of licorice.
- gliasperin B The spectral data of the compound is shown below, and shows a very good agreement with the spectral data of gliasperin B, which has been reported in the literature as having been isolated from plants of the genus Glycyrrhiza (Zeng L. et al., Heterocycles 34:575-587 (1992)). Since there was no inconsistency with the structural information, the compound was identified as gliasperin B (GB).
- the amount of gliasperin B (GB) contained in the licorice extract was quantified using the high performance liquid chromatography method described in detail below. As a result, the amount of gliasperin B (GB) contained was 132.6 ⁇ g per 1 g of licorice extract.
- the amount of glycyrrhizic acid contained in the licorice extract was 123 mg per 1 g of licorice extract, and the value (ratio) of gliasperin B (GB) content/glycyrrhizic acid content was 0.00107.
- Mouse preadipocytes 3T3-L1 were cultured in differentiation induction medium (0.5 mM isobutyl-methylxanthine, 1 ⁇ M dexamethasone, 1 ⁇ g/mL insulin in 10% FBS/DMEM) for 2 days, and then the medium was replaced with 10% FBS/DMEM medium containing 1 ⁇ g/mL insulin and cultured for 7 days.
- the test substances (licorice extract and GB) were added on the 9th day after the start of differentiation induction, and the cells were then cultured for 19 hours.
- the licorice extract used in Test 1 was used as the licorice extract (final concentrations: 100 ⁇ g/mL, 200 ⁇ g/mL, 300 ⁇ g/mL), and the GB prepared in Test 1 was used as the GB (final concentrations: 13.5 ng/mL, 27 ng/mL, 40 ng/mL).
- Three subjects were tested for each group.
- Glycerol released into the medium upon decomposition of neutral fats accumulated in the cells was quantified using Labo Assay (TM) Triglyceride (Fujifilm Wako Pure Chemical). The results are shown in Figure 1.
- the amount of glycerol is shown as the mean value ⁇ standard deviation.
- the symbols in Figure 1 indicate the following: ** p ⁇ 0.01 vs. no treatment (Student's t-test).
- the amount of glycerol released increased in a GB concentration-dependent manner, indicating that intracellular lipolysis occurred in a GB concentration-dependent manner, and GB was found to promote lipolysis in adipocytes.
- licorice extract also released glycerol from adipocytes, but the amount of glycerol released was less when the licorice extract concentration was 300 ⁇ g/mL than when it was 100 ⁇ g/mL or 200 ⁇ g/mL.
- the concentrations of licorice extract 100 ⁇ g/mL, 200 ⁇ g/mL, and 300 ⁇ g/mL, respectively, are converted to the amount of GB contained in 13.5 ng/mL, 27 ng/mL, and 40 ng/mL.
- Licorice extract also has an excellent lipolysis effect, but the lipolysis effect decreased as the concentration increased, suggesting that licorice extract contains various components, some of which may inhibit lipolysis. From this, it can be said that in order to obtain a higher lipolysis effect, it is preferable to use a refined GB product rather than a licorice extract.
- a subject one male, aged 59 took 200 mg of the licorice extract used in Test 1 per day for 100 days.
- the subject was given a record sheet and asked to record the body weight, BMI, and visceral fat level measured with a Tanita body composition scale before the start of licorice extract intake (day 0), on the 30th day of intake, the 45th day of intake, the 72nd day of intake, the 83rd day of intake, the 92nd day of intake, and the day after the final intake (day 101).
- the changes in body weight, BMI, and visceral fat level from before the start of intake are shown in Figure 2.
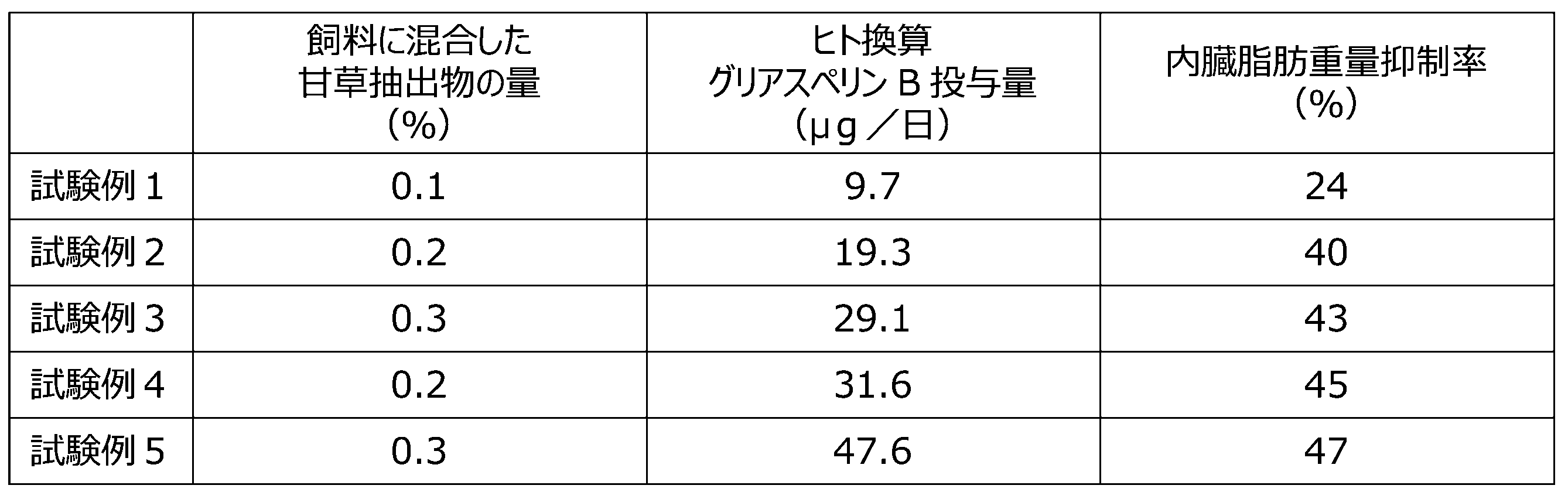
- ⁇ Test 6> Five-week-old male C57BL/6J mice were divided into groups of six and fed MF (Oriental Yeast), high-fat, high-carbohydrate powdered feed (hereinafter abbreviated as HFS, D12079BM, Research Diet), or HFS mixed with licorice extract for 8 weeks. After 8 weeks of ingestion, the visceral fat weight (the sum of the epididymal fat weight, perirenal fat weight, and mesenteric fat weight) was measured.
- the root and stolon of licorice were crushed into powder, and the solution obtained through a 50% (v/v) ethanol extraction process was dried and solidified using an evaporator and freeze-dried, and then crushed to obtain a licorice extract. These were mixed with the feed in the amounts (%) shown in Table 8 below and fed to the mice.
- the human-equivalent dose of gliasperin ( ⁇ g/day) and the visceral fat weight suppression rate (%) of the mice are also shown in Table 8.
- the internal composition of the present invention contains gliasperin B, which has an excellent lipolysis promoting effect and a significant weight loss effect.
- gliasperin B which has an excellent lipolysis promoting effect and a significant weight loss effect.
- fat in adipocytes can be efficiently broken down and body fat such as subcutaneous fat and visceral fat can be reduced, resulting in a significant weight loss effect and an improvement in BMI.
- the composition improves insulin resistance and reduces sterol levels. Therefore, the internal composition of the present invention can prevent and improve obesity and lifestyle-related diseases in the long term, thereby maintaining and improving health.
- the content of gliasperin B in a plant extract is an indicator of the effectiveness of the plant extract for lipolysis, weight loss, etc., making it possible to efficiently select a highly effective plant extract.
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Diabetes (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Engineering & Computer Science (AREA)
- Natural Medicines & Medicinal Plants (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Organic Chemistry (AREA)
- Hematology (AREA)
- Obesity (AREA)
- Bioinformatics & Cheminformatics (AREA)
- General Chemical & Material Sciences (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Botany (AREA)
- Epidemiology (AREA)
- Mycology (AREA)
- Child & Adolescent Psychology (AREA)
- Biotechnology (AREA)
- Medical Informatics (AREA)
- Microbiology (AREA)
- Alternative & Traditional Medicine (AREA)
- Emergency Medicine (AREA)
- Endocrinology (AREA)
- Nutrition Science (AREA)
- Food Science & Technology (AREA)
- Polymers & Plastics (AREA)
- Medicines Containing Plant Substances (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
Description
本発明は、脂肪分解及び/又は体重減少に対する内用組成物、有効性を判定する方法、並びに植物抽出物の品質の指標に関する。 The present invention relates to an internal composition for lipolysis and/or weight loss, a method for determining efficacy, and an indicator of the quality of a plant extract.
体脂肪は消費エネルギーに対し摂取エネルギーが過剰である場合に、その過剰分が、白色脂肪細胞の中性脂肪として蓄積して生じるものである。過剰な脂肪の蓄積によって肥満となって体重が増加し、その結果、この肥満は様々な生活習慣病の原因となるばかりでなく、美容面でも大きな問題となっている。体脂肪のうち内臓脂肪としての蓄積が大きい肥満は、インスリン抵抗性や動脈硬化等の病態との関係が指摘され、また、皮下脂肪として蓄積が大きい肥満は美容の観点から男女を問わず大きな関心事となっている。 Body fat occurs when energy intake is in excess of energy expenditure, and the excess accumulates as neutral fat in white fat cells. Excessive fat accumulation leads to obesity and weight gain, which not only causes various lifestyle-related diseases but also poses a major cosmetic problem. Obesity with a large accumulation of visceral fat has been linked to conditions such as insulin resistance and arteriosclerosis, while obesity with a large accumulation of subcutaneous fat is a major concern for both men and women from the perspective of cosmetic care.
従来から、肥満の予防と改善については、食事制限、消化管における糖質吸収阻害物質の探索等、摂取エネルギーを制限するような様々な対策が行われてきた。しかし、エネルギー摂取の制限は基礎代謝量を低下させる要因にもなり、肥満が改善されないことがある。また、エネルギー摂取の制限によって必要な栄養が不足し、健康を害することもある。そのため、肥満を解消するためには、蓄積された脂肪を積極的に分解して熱エネルギーとして発散し、その結果、理想的な体重を維持できることが理想的であると考えられるようになってきている。このような状況下、近年、食品素材中から脂肪分解促進作用を有する機能性成分の探索が活発に行われ、多くの飲食品が提案されている。 Traditionally, various measures to prevent and improve obesity have been taken, such as restricting energy intake, dietary restrictions, and the search for substances that inhibit carbohydrate absorption in the digestive tract. However, restricting energy intake can also cause a decrease in basal metabolic rate, and obesity may not improve. Restricting energy intake can also lead to a deficiency of necessary nutrients, which can be harmful to health. For this reason, it has come to be thought that the ideal way to eliminate obesity is to actively break down accumulated fat and release it as heat energy, thereby allowing one to maintain an ideal body weight. Under these circumstances, in recent years, there has been active research into functional ingredients that promote lipolysis among food ingredients, and many foods and beverages have been proposed.
例えば、サケ白子抽出物、ビール酵母抽出物、大麦若葉エキス、及びトリコラーゲン(チキンコラーゲン)を組み合わせて調製した混合物が、優れた脂肪分解促進作用を有すること(特許文献1)、カバノキ科シラカバの抽出物及びイネ科クマザサの抽出物の少なくともいずれかが、全身あるいは局所の脂肪組織における脂肪の分解の促進を通じて肥満体質の改善、及び脂肪組織の増大を防止すること(特許文献2)、セイヨウネズ等の特定の植物又はその抽出物が、脂肪組織に蓄積された中性脂肪の分解を促進し、肥満の抑制、防止又は改善等の痩身効果を発揮する医薬、食品又は化粧料として有用であること(特許文献3)等が知られている。しかし、これらの植物の抽出物等の脂肪分解効果、体重減少効果は十分とはいえないのが現状である。また特許文献4には、甘草疎水性抽出物と、抗酸化成分等を組み合わせた組成物が、体脂肪蓄積抑制、体脂肪分解促進及びエネルギー産生促進効果を奏することが開示されているが、甘草疎水性抽出物のみでは十分な脂肪分解促進効果は得られていない。 For example, it is known that a mixture prepared by combining salmon milt extract, brewer's yeast extract, young barley leaf extract, and chicken collagen (chicken collagen) has an excellent lipolysis promoting effect (Patent Document 1), that at least one of an extract of Betulaceae (Betulaceae) and an extract of Kumazasa (Poaceae) improves obesity and prevents the increase of adipose tissue by promoting the decomposition of fat in whole body or local adipose tissue (Patent Document 2), and that certain plants such as Juniperus communis or their extracts are useful as medicines, foods, or cosmetics that promote the decomposition of neutral fats accumulated in adipose tissue and exert slimming effects such as suppressing, preventing, or improving obesity (Patent Document 3). However, the lipolysis and weight loss effects of extracts of these plants are not sufficient at present. Patent Document 4 also discloses that a composition combining a licorice hydrophobic extract with an antioxidant component and the like has the effects of suppressing body fat accumulation, promoting body fat decomposition, and promoting energy production, but a sufficient lipolysis promoting effect is not obtained with licorice hydrophobic extract alone.
本発明が解決しようとする課題は、脂肪分解促進効果、内臓脂肪・体脂肪減少効果、体重減少効果、BMIの改善効果に優れる新規の製剤を提供することである。本発明はまた、インスリン抵抗性改善効果、コレステロール値減少効果等に優れる新規の製剤を提供することも目的とする。 The problem that the present invention aims to solve is to provide a new formulation that is excellent in promoting lipolysis, reducing visceral fat and body fat, reducing body weight, and improving BMI. Another object of the present invention is to provide a new formulation that is excellent in improving insulin resistance, reducing cholesterol levels, etc.
本発明者らは、鋭意研究を行った結果、甘草の抽出物中の特定の成分である「グリアスペリンB」が、優れた脂肪分解促進作用を有することを見出した。また、グリアスペリンBを含有する甘草の抽出物を含む製剤が、優れた脂肪分解促進効果に加えて、内臓脂肪・体脂肪減少効果、体重減少効果、BMIの改善効果、インスリン抵抗性改善効果、コレステロール値減少効果を奏することも見出した。さらに、植物抽出物におけるグリアスペリンBの含有量が、植物抽出物の脂肪分解、体重減少等に対する有効性の指標になることも見出した。本発明の要旨は以下のとおりである。 As a result of intensive research, the inventors have found that "gliasperin B," a specific component in licorice extract, has an excellent effect of promoting lipolysis. They have also found that a preparation containing licorice extract containing gliasperin B has an excellent effect of promoting lipolysis, as well as an effect of reducing visceral fat and body fat, a weight loss effect, an effect of improving BMI, an effect of improving insulin resistance, and an effect of reducing cholesterol levels. They have also found that the content of gliasperin B in a plant extract is an indicator of the effectiveness of the plant extract for lipolysis, weight loss, etc. The gist of the present invention is as follows.
[1]グリアスペリンBを含有する、脂肪の分解、脂肪の燃焼、脂肪の消費補助、BMIの改善、BMIの低下補助、腹部の脂肪減少、内臓脂肪減少、体脂肪の減少、ウエスト周囲径の減少、インスリン抵抗性の改善、LDLコレステロール値の減少のうちいずれか一以上のために用いられる内用組成物。
[2]グリアスペリンBを、有効量含有する、脂肪の分解、脂肪の燃焼、脂肪の消費補助、BMIの改善、BMIの低下補助、腹部の脂肪減少、内臓脂肪減少、体脂肪の減少、ウエスト周囲径の減少、インスリン抵抗性の改善、LDLコレステロール値の減少のうちいずれか一以上のために用いられる内用組成物。
[3]有効量が、成人1日あたり10μg以上である、[2]に記載の内用組成物。
[4]BMIが23kg/m2以上30kg/m2未満の成人に対して用いられる、[1]又は[2]に記載の内用組成物。
[5]内臓脂肪面積が100cm2以上の成人に対して用いられる、[1]又は[2]に記載の内用組成物。
[6]グリアスペリンBの総量換算で、5~100μg/日の用量で摂取される、[1]又は[2]に記載の内用組成物。
[7]植物抽出物について、グリアスペリンBの含有量を指標に、生体における脂肪分解及び/又は体重減少に対する有効性を判定する方法。
[8]上記植物が、甘草である、[7]に記載の脂肪分解及び/又は体重減少に対する有効性を判定する方法。
[9]植物抽出物の品質の指標であって、上記品質が、生体における脂肪分解及び/又は体重減少に対する有効性に関する品質であり、グリアスペリンBからなることを特徴とする指標。
[10]上記植物が、甘草である、[9]に記載の指標。
[1] An internal composition containing gliasperin B which is used for one or more of the following: decomposing fat, burning fat, assisting in fat consumption, improving BMI, assisting in lowering BMI, reducing abdominal fat, reducing visceral fat, reducing body fat, reducing waist circumference, improving insulin resistance, and reducing LDL cholesterol levels.
[2] An internal composition containing an effective amount of gliasperin B and used for one or more of the following: decomposing fat, burning fat, assisting in fat consumption, improving BMI, assisting in lowering BMI, reducing abdominal fat, reducing visceral fat, reducing body fat, reducing waist circumference, improving insulin resistance, and reducing LDL cholesterol levels.
[3] The oral composition described in [2], wherein the effective amount is 10 μg or more per day for an adult.
[4] The internal composition described in [1] or [2], which is used for adults with a BMI of 23 kg/ m2 or more and less than 30 kg/ m2 .
[5] The internal composition described in [1] or [2], which is used for adults with a visceral fat area of 100 cm2 or more.
[6] The internal composition according to [1] or [2], which is taken at a dose of 5 to 100 μg/day in terms of the total amount of gliaperin B.
[7] A method for determining the effectiveness of a plant extract for lipolysis and/or weight loss in a living body, using the content of gliasperin B as an indicator.
[8] The method for determining the effectiveness of a compound according to [7] above, wherein the plant is licorice.
[9] An indicator of the quality of a plant extract, the quality being related to effectiveness in lipolysis and/or weight loss in a living body, characterized in that the indicator consists of gliasperin B.
[10] The indicator described in [9], wherein the plant is licorice.
本発明の内用組成物は、グリアスペリンBを含有することにより、好ましくは有効成分、機能性関与成分、指標成分又は寄与成分として含有することにより、優れた脂肪分解促進作用を有すると共に、顕著な体重減少効果を奏する。本発明の内用組成物を用いることで、脂肪細胞中の脂肪を効率よく分解し、皮下脂肪、内臓脂肪等の体脂肪を減少させることができ、結果的に顕著な体重減少効果、BMIの改善効果を得ることができる。また、インスリン抵抗性改善、これステロール値低減効果も得られる。よって、本発明の内用組成物によると、肥満や生活習慣病を長期的に予防し、また改善することができ、健康の維持・増進を実現することができる。さらに、植物抽出物におけるグリアスペリンBの含有量が、植物抽出物の脂肪分解、体重減少等に対する有効性の指標になることから、有効性の高い植物抽出物を効率的に選定することが可能となる。 The internal composition of the present invention contains gliasperin B, preferably as an active ingredient, functional ingredient, indicator ingredient or contributing ingredient, and thus has an excellent lipolysis promoting effect and a remarkable weight loss effect. By using the internal composition of the present invention, fat in adipocytes can be efficiently broken down and body fat such as subcutaneous fat and visceral fat can be reduced, resulting in a remarkable weight loss effect and an improvement effect on BMI. In addition, the internal composition of the present invention can also improve insulin resistance and reduce sterol levels. Therefore, the internal composition of the present invention can prevent and improve obesity and lifestyle-related diseases in the long term, and can achieve the maintenance and improvement of health. Furthermore, since the content of gliasperin B in a plant extract is an indicator of the effectiveness of the plant extract for lipolysis, weight loss, etc., it becomes possible to efficiently select a highly effective plant extract.
以下に、本発明の内用組成物、脂肪分解及び/又は体重減少に対する有効性を判定する方法、並びに植物抽出物の品質の指標について詳細に説明する。 Below, the internal composition of the present invention, the method for determining the effectiveness of the composition on lipolysis and/or weight loss, and the quality indicators of the plant extract are described in detail.
<内用組成物>
本発明の内用組成物は、グリアスペリンBを含有する。限定はされないが、グリアスペリンBを有効成分、機能性関与成分、指標成分又は寄与成分として含有することが好ましい。本発明の内用組成物は、必須成分であるグリアスペリンBに加えて、本発明の効果を損なわない範囲でその他の成分を含有していてもよい。以下に、本発明の内用組成物が含むグリアスペリンB、及びその他の成分、本発明の内用組成物の形態等について説明する。
<Composition for internal use>
The internal composition of the present invention contains gliasperin B. Although not limited thereto, it is preferable that gliasperin B is contained as an active ingredient, a functional ingredient, an indicator ingredient or a contributing ingredient. In addition to gliasperin B, which is an essential ingredient, the internal composition of the present invention may contain other ingredients within a range that does not impair the effects of the present invention. The internal composition of the present invention contains gliasperin B and other ingredients, and the form of the internal composition of the present invention, etc. will be described below.
本明細書において、有効成分とは、医薬品、医薬部外品等に含まれる成分のうち、その目的の効果を表す薬理活性成分や生理活性成分をいう。 In this specification, an active ingredient refers to a pharmacologically active ingredient or a physiologically active ingredient contained in a drug, quasi-drug, etc. that exerts a desired effect.
本明細書において、機能性関与成分とは、食品等の内用組成物が含む成分のうち、特定の保健の目的に資する成分をいう。具体的には、機能性に係る作用機序について、in vitro試験及びin vivo試験又は臨床試験(ヒト試験)により考察されているものであり、直接的又は間接的な定性確認及び定量確認が可能な成分である。なお、機能性表示食品は、当該機能性表示食品に配合される成分が有する効果に基づく機能を表示して需要者に提供されるものであって、機能性表示食品として需要者に提供しようとする場合、上記機能性関与成分の含有量やその定量方法を届け出なければならないとされている。 In this specification, functional ingredients refer to ingredients contained in internal compositions such as foods that contribute to a specific health purpose. Specifically, the mechanism of action related to functionality has been considered through in vitro tests, in vivo tests, or clinical trials (human tests), and the ingredients can be directly or indirectly qualitatively and quantitatively confirmed. Functional foods are provided to consumers with a display of functions based on the effects of the ingredients contained in the functional food, and when providing such foods to consumers as functional foods, the content of the functional ingredients and the method of quantifying them must be notified.
本明細書において指標成分とは、機能性の科学的根拠の一部を説明できる特定の成分が判明しているものの、当該特定の成分のみでは機能性の全てを説明することができない抽出物、エキス等を機能性関与成分とする場合、表示しようとする機能性に係る作用機序について、少なくとも1つの成分について、in vitro試験及びin vivo試験又は臨床試験(ヒト試験)により考察されているものがある場合、このような成分のことをいう。 In this specification, the term "index ingredient" refers to an extract or essence that is considered to be a functional ingredient when a specific ingredient that can explain part of the scientific basis of functionality is known, but the functionality cannot be fully explained by the specific ingredient alone, and the mechanism of action related to the functionality to be displayed has been considered for at least one ingredient through in vitro tests, in vivo tests, or clinical tests (human tests).
本明細書において寄与成分とは、物質であって、配合することで、直接的又は間接的に、身体の生理学的機能に影響する成分をいう。特定保健用食品及び機能性表示食品において寄与成分に相当するものとして、「関与成分」、「機能性関与成分」、「指標成分」等が考えられるがこれらに限定されない。また、医薬品及び医薬部外品における寄与成分に相当するものは「有効成分」である。さらに、機能性表示食品以外の食品組成物の販売等において、「○○成分配合。燃焼サポートに」といった形で暗示的に消費者に効果と紐づけて強調している成分も含む概念である。 In this specification, a contributing ingredient refers to a substance that, when incorporated, directly or indirectly affects the physiological functions of the body. In foods for specified health uses and foods with functional claims, the equivalent of a contributing ingredient could be, but is not limited to, a "contributing ingredient," a "functional contributing ingredient," or an "indicator ingredient." In addition, the equivalent of a contributing ingredient in medicines and quasi-drugs is an "active ingredient." Furthermore, in the sale of food compositions other than foods with functional claims, this concept also includes ingredients that are implicitly linked to an effect and emphasized to consumers in the form of "Contains XX ingredient. For burning calories."
(グリアスペリンB)
グリアスペリンB(Glyasperin B、以下「GB」ともいう)は、下記式で表される、イソフラバノン誘導体である。3-(2,4-ジヒドロキシフェニル)-5-ヒドロキシ-2,3-ジヒドロ-6-(3-メチル-2-ブテニル)-7-メトキシ-4H-1-ベンゾピラン-4-オン、3-(2,4-ジヒドロキシフェニル)-5-ヒドロキシ-6-(3-メチル-2-ブテニル)-7-メトキシ-3,4-ジヒドロ-2H-1-ベンゾピラン-4-オン、又は3-(2,4-ジヒドロキシフェニル)-5-ヒドロキシ-7-メトキシ-6-(3-メチルブタ-2-エン-1-イル)-3,4-ジヒドロ-2H-1-ベンゾピラン-4-オンとも表される。
Glyasperin B (hereinafter also referred to as "GB") is an isoflavanone derivative represented by the following formula: It is also represented as 3-(2,4-dihydroxyphenyl)-5-hydroxy-2,3-dihydro-6-(3-methyl-2-butenyl)-7-methoxy-4H-1-benzopyran-4-one, 3-(2,4-dihydroxyphenyl)-5-hydroxy-6-(3-methyl-2-butenyl)-7-methoxy-3,4-dihydro-2H-1-benzopyran-4-one, or 3-(2,4-dihydroxyphenyl)-5-hydroxy-7-methoxy-6-(3-methylbut-2-en-1-yl)-3,4-dihydro-2H-1-benzopyran-4-one.
グリアスペリンBは、甘草抽出物中に含まれることが確認されている成分であり、本発明におけるグリアスペリンBとしても、甘草抽出物由来のものが好ましい。これまで、甘草抽出物が脂肪分解促進効果を奏することは知られており、甘草抽出物中に多く含まれるグラブリジンが脂肪分解促進効果を奏する可能性を示唆する文献は存在した(特許文献4等参照)。しかしながら、甘草抽出物中のどの成分が脂肪分解促進作用を奏するのかは明らかではなかった。本発明者らは、甘草抽出物中に多数存在する成分のうち、グリアスペリンBが脂肪分解促進効果を奏することを初めて見出した。そして、このグリアスペリンBを含有し、脂肪の分解、脂肪の燃焼、脂肪の消費補助、BMIの改善、BMIの低下補助、腹部の脂肪減少、内臓脂肪減少、体脂肪の減少、ウエスト周囲径の減少、インスリン抵抗性の改善、LDLコレステロール値の減少の効果を奏する、内用組成物の発明を完成させた。 Gliasperin B is a component that has been confirmed to be contained in licorice extract, and the gliasperin B in the present invention is preferably derived from licorice extract. It has been known that licorice extract has a lipolysis promoting effect, and there have been documents suggesting that glabridin, which is contained in large amounts in licorice extract, may have a lipolysis promoting effect (see Patent Document 4, etc.). However, it was not clear which component in licorice extract has the lipolysis promoting effect. The present inventors have discovered for the first time that, of the many components present in licorice extract, gliasperin B has a lipolysis promoting effect. They have then completed the invention of an internal composition that contains this gliasperin B and has the effects of decomposing fat, burning fat, assisting in fat consumption, improving BMI, assisting in lowering BMI, reducing abdominal fat, reducing visceral fat, reducing body fat, reducing waist circumference, improving insulin resistance, and reducing LDL cholesterol levels.
本発明におけるグリアスペリンBは、甘草に由来するものであってもよいし、化学合成品であってもよい。なお、本明細書でグリアスぺリンBを含有する、という場合、グリアスぺリンBを含有する甘草等の植物を含有する、場合を含む。 The gliasperin B in the present invention may be derived from licorice or may be a chemically synthesized product. In this specification, when we say that it contains gliasperin B, this also includes the case where it contains a plant such as licorice that contains gliasperin B.
上記甘草の種類は特に限定されないが、マメ科グリキルリーザ(Glycyrrhiza)属の植物、例えば、グリキルリーザ ウラレンシス(G.uralensis Fisch.et DC;ウラルカンゾウ)、グリキルリーザ インフラータ(G.inflata BAT.;チョウカカンゾウ)、グリキルリーザ グラブラ(G.glabra L.;ヨウカンゾウ)、グリキルリーザ グラブラ(G.glabra L.var glandu rifera Regel et Herder;ナンキンカンゾウ)、グリキルリーザ アスペラ(G.aspera)、グリキルリーザ エチナータ(G.echinata L.;シナカンゾウ)、グリキルリーザ パリディフローラ(G.pallidiflora Maxim;イヌカンゾウ)等が挙げられる。 The type of licorice is not particularly limited, but may be any plant of the genus Glycyrrhiza in the family Fabaceae, such as Glycyrrhiza uralensis (G. uralensis Fisch. et DC; ural licorice), Glycyrrhiza inflata (G. inflata BAT.; butterfly licorice), Glycyrrhiza glabra (G. glabra L.; licorice), Glycyrrhiza glabra L. ( ... These include G. glabra L. var glandu lifera Regel et Herder (Chinese tallow tree), G. aspera, G. echinata L. (Chinese tallow tree), and G. pallidiflora Maxim (Chinese tallow tree).
甘草に由来するグリアスペリンBを取得する方法としては、甘草抽出物から精製する方法が挙げられる。甘草抽出物とは、具体的には、甘草の全草、又は一部(例えば、根、茎、ストロン、葉、花、果実等)の粉砕物を溶媒で抽出した甘草抽出物であり、得られた甘草抽出物を噴霧乾燥、凍結乾燥したものでもよい。上記抽出溶媒としては、水、もしくはメタノール、エタノール等のアルコール類、又は水とアルコール類もしくはアセトン等のケトン類との混合溶媒等が挙げられる。これらのうち、抽出溶媒としては、水、アルコール、含水アルコールが好ましく、熱水、エタノール、含水エタノールがより好ましい。 Methods for obtaining gliasperin B derived from licorice include a method of purifying it from a licorice extract. Specifically, the licorice extract is a licorice extract obtained by extracting the whole licorice plant or a crushed part of the plant (e.g., roots, stems, stolons, leaves, flowers, fruits, etc.) with a solvent, and the obtained licorice extract may be spray-dried or freeze-dried. Examples of the extraction solvent include water, alcohols such as methanol and ethanol, and mixed solvents of water and alcohols or ketones such as acetone. Of these, the extraction solvent is preferably water, alcohol, or aqueous alcohol, and more preferably hot water, ethanol, or aqueous ethanol.
上記含水アルコールのアルコール濃度は、0.1質量%~99.9質量%であり、10質量%~99.9質量%であることが好ましく、30質量%~70質量%であることがより好ましく、40質量%~60質量%であることが更に好ましく、50質量%であることが特に好ましい。或いは、0.1%(v/v)~99.9%(v/v)であり、10%(v/v)~99.9%(v/v)であることが好ましく、30%(v/v)~70%(v/v)であることがより好ましく、40%(v/v)~60%(v/v)であることが更に好ましく、50%(v/v)であることが特に好ましい。 The alcohol concentration of the hydrous alcohol is 0.1% to 99.9% by mass, preferably 10% to 99.9% by mass, more preferably 30% to 70% by mass, even more preferably 40% to 60% by mass, and particularly preferably 50% by mass. Alternatively, it is 0.1% (v/v) to 99.9% (v/v), preferably 10% (v/v) to 99.9% (v/v), more preferably 30% (v/v) to 70% (v/v), even more preferably 40% (v/v) to 60% (v/v), and particularly preferably 50% (v/v).
上記甘草抽出物の乾燥品に蒸留水を加えて溶解し、甘草抽出物水溶液を調製する。この水溶液の容量に対して半量の酢酸エチル等の有機溶媒を加えて混和し二層に分離した状態とし、そこから酢酸エチル等の有機溶媒層を回収する操作を複数回、好ましくは3回程度繰り返す。得られた有機溶媒層に無水硫酸ナトリウムを加えた後、ろ過をすることで有機画分を得ることができる。有機画分をエバポレーターにて濃縮した後、シリカゲルカラムクロマトグラフにて、ヘキサン:酢酸エチル=1:1(v/v)等で溶出した画分を、ODSカラムを用いた高速液体クロマトグラフ(アセトニトリル:水=60:40,45:55(いずれもv/v))により精製を繰り返すことで、化合物(グリアスペリンB)を得ることができる。 Distilled water is added to the dried licorice extract to dissolve it, and an aqueous licorice extract solution is prepared. An organic solvent such as ethyl acetate is added in an amount equal to half the volume of the aqueous solution, and the solution is mixed to separate into two layers, from which the organic solvent layer such as ethyl acetate is collected. This operation is repeated several times, preferably about three times. Anhydrous sodium sulfate is added to the obtained organic solvent layer, and the organic fraction can be obtained by filtering. The organic fraction is concentrated using an evaporator, and the fraction eluted with hexane:ethyl acetate = 1:1 (v/v) or the like using a silica gel column chromatograph is repeatedly purified using high-performance liquid chromatography (acetonitrile:water = 60:40, 45:55 (both v/v)) using an ODS column to obtain the compound (gliasperin B).
本発明の内用組成物におけるグリアスペリンBの含有量は、0.00001質量%~50質量%であり、0.0001質量%~20質量%であることが好ましく、0.0005質量%~10質量%であることがより好ましく、0.001質量%~5質量%であることがさらに好ましい。 The content of gliasperin B in the internal composition of the present invention is 0.00001% to 50% by mass, preferably 0.0001% to 20% by mass, more preferably 0.0005% to 10% by mass, and even more preferably 0.001% to 5% by mass.
(その他の成分)
本発明の内用組成物は、グリアスペリンBに加えて、本発明の効果を損なわない範囲で、担体、賦形剤、溶媒、その他の任意成分を含有してもよい。
(Other ingredients)
The internal composition of the present invention may contain, in addition to gliasperin B, a carrier, an excipient, a solvent, and other optional components within a range that does not impair the effects of the present invention.
本発明の内用組成物は、グリアスペリンBを含有する際に、上述のような甘草抽出物から精製されたグリアスペリンBや、化学合成品であるグリアスペリンBを配合してもよいし、最終的に内用組成物におけるグリアスペリンBの含有量が上記数値範囲となるように、甘草抽出物を配合してもよい。また、本発明の内用組成物は、脂肪分解促進作用の観点から、グリアスペリンBの含有量が上記数値範囲内となると共に、グリアスペリンB以外の甘草由来成分を含有しないか、又はグリアスペリンB以外の甘草由来成分を含む場合には、日本薬局方における甘草エキスの定量指標であるグリチルリチン酸を比較基準として、グリアスペリンBの含有量とグリチルリチン酸の含有量の比(グリアスペリンBの含有量をグリチルリチン酸の含有量で除した値)が、0.001以上であることが好ましい。 When the internal composition of the present invention contains gliasperin B, it may contain gliasperin B purified from licorice extract as described above or a chemically synthesized gliasperin B, or it may contain licorice extract so that the final content of gliasperin B in the internal composition is within the above numerical range. From the viewpoint of promoting lipolysis, the internal composition of the present invention preferably contains gliasperin B within the above numerical range and does not contain any licorice-derived components other than gliasperin B, or, if it contains any licorice-derived components other than gliasperin B, the ratio of the gliasperin B content to the glycyrrhizic acid content (the value obtained by dividing the gliasperin B content by the glycyrrhizic acid content) is 0.001 or more, using glycyrrhizic acid, which is a quantitative indicator of licorice extract in the Japanese Pharmacopoeia, as a comparison standard.
本発明の内用組成物は、飲食品、機能性表示食品、特定保健用食品、栄養機能食品、化粧品、医薬部外品、医薬品又は化粧料等として、脂肪を効率よく分解し、皮下脂肪、内臓脂肪等の体脂肪を減少させるために用いられる。 The internal composition of the present invention can be used as a food or drink, a functional food, a food for specified health uses, a nutrient-functional food, a cosmetic, a quasi-drug, a medicine, or a cosmetic agent, etc., to efficiently break down fat and reduce body fat, such as subcutaneous fat and visceral fat.
本発明の内用組成物を飲食品として調製する場合、グリアスペリンBの他に、甘味料、着色料、保存料、増粘剤、安定剤、ゲル化剤、糊剤、酸化防止剤、発色剤、漂白剤、防かび剤(防ばい剤)、イーストフード、ガムベース、香料、酸味料、調味料、乳化剤、pH調整剤、かんすい、膨脹剤、栄養強化剤、その他飲食品素材等を混合して、所望の形態に調製すればよい。本発明の内用組成物を飲食品形態にする場合、その形態については、特に制限されるものではない。一例として、ゲル状剤、顆粒、細粒、カプセル、錠剤、粉末、液剤、半固形剤等のサプリメントタイプの食品;炭酸飲料、清涼飲料、乳飲料、アルコール飲料、果汁飲料、茶類、栄養飲料等の飲料;粉末ジュース、粉末スープ等の粉末飲料;ガム、タブレット、キャンディー、クッキー、グミ、せんべい、ビスケット、ゼリー等の菓子類;パン、麺類、シリアル、ジャム、調味料等が例示される。これらの食品は、脂肪を効率よく分解し、皮下脂肪、内臓脂肪等の体脂肪を減少させるための飲食品として使用され、例えば、一般の飲食品の他、栄養補助食品、機能性表示食品、特定保健用食品、病者用食品等のニュートラシューティカルとしても使用できる。 When preparing the internal composition of the present invention as a food or beverage, in addition to gliasperin B, sweeteners, colorants, preservatives, thickeners, stabilizers, gelling agents, pasting agents, antioxidants, colorants, bleaching agents, mold inhibitors (mold inhibitors), yeast food, gum base, flavorings, acidulants, seasonings, emulsifiers, pH adjusters, kansui, leavening agents, nutritional enhancers, and other food and beverage ingredients can be mixed and prepared into the desired form. When the internal composition of the present invention is made into a food or beverage form, there are no particular limitations on the form. Examples include supplement-type foods such as gels, granules, fine granules, capsules, tablets, powders, liquids, and semisolids; beverages such as carbonated drinks, soft drinks, milk drinks, alcoholic drinks, fruit juice drinks, teas, and nutritional drinks; powdered drinks such as powdered juice and powdered soup; confectioneries such as gum, tablets, candies, cookies, gummies, rice crackers, biscuits, and jellies; bread, noodles, cereals, jams, and seasonings. These foods are used as foods and beverages to efficiently break down fat and reduce body fat such as subcutaneous fat and visceral fat, and can be used, for example, as nutraceuticals such as dietary supplements, functional foods, foods for specified health uses, and foods for the sick, in addition to general foods and beverages.
本発明の内用組成物を医薬品(医薬部外品を含む)として調製する場合、グリアスペリンBの他に、必要に応じて、他の薬効成分、薬学的に許容される担体や添加剤等を任意に配合してもよい。薬学的に許容される担体及び添加剤としては、具体的には、結合剤、崩壊剤、滑沢剤、湿潤化剤、緩衝剤、保存剤、香料等が例示される。本発明の内用組成物を医薬品として調製する場合、その形態については、特に制限されるものではない。一例として、注射剤、外用剤、吸入剤、座剤、フィルム剤、トローチ剤、液剤、散剤、錠剤、顆粒剤、カプセル剤、シロップ剤、点眼剤、洗眼剤、点鼻剤等を挙げることができる。これらの中でも、経口投与に適した形態(即ち、内服用医薬品)が好ましく、かかる形態として具体的にはトローチ剤、液剤、散剤、錠剤、顆粒剤、カプセル剤、シロップ剤等を挙げることができる。これらの医薬品(医薬部外品を含む)は、脂肪を効率よく分解し、皮下脂肪、内臓脂肪等の体脂肪を減少させるための医薬品として使用される。 When the internal composition of the present invention is prepared as a medicine (including quasi-drugs), in addition to gliasperin B, other medicinal ingredients, pharma- ceutical acceptable carriers, additives, etc. may be optionally blended, if necessary. Specific examples of pharma- ceutical acceptable carriers and additives include binders, disintegrants, lubricants, wetting agents, buffers, preservatives, and flavorings. When the internal composition of the present invention is prepared as a medicine, its form is not particularly limited. Examples include injections, external preparations, inhalants, suppositories, films, lozenges, liquids, powders, tablets, granules, capsules, syrups, eye drops, eyewashes, and nasal drops. Among these, forms suitable for oral administration (i.e., internal medicines) are preferred, and specific examples of such forms include lozenges, liquids, powders, tablets, granules, capsules, and syrups. These medicines (including quasi-drugs) are used as medicines to efficiently break down fat and reduce body fat, including subcutaneous fat and visceral fat.
本発明の内用組成物に含まれるグリアスペリンBは化粧料(機能性化粧料を含む)又は外用医薬部外品としても使用することができる。化粧料又は外用医薬部外品として調製するには、グリアスペリンBに加えて、薬学的又は化粧学的に許容される担体(水、油性成分等)を配合して、所望の形態に調製すればよい。上記化粧料は、皮膚に適用可能である限り、その形態については、特に制限されるものではない。一例として、液状、乳液状、粉末状、固形状、懸濁液状、クリーム状、軟膏状、ムース状、顆粒状、錠剤状、ゲル状、ゼリー状、ペースト状、ジェル状、エアゾール状、スプレー状、リニメント剤、パック剤等の形態を挙げることができる。これらの化粧料は、脂肪を効率よく分解し、皮下脂肪等を減少させるための化粧料として使用される。 Gliasperin B contained in the internal composition of the present invention can also be used as a cosmetic (including functional cosmetic) or topical quasi-drug. To prepare a cosmetic or topical quasi-drug, a pharma- ceutical or cosmetically acceptable carrier (water, oily components, etc.) is added to Gliasperin B and the cosmetic is prepared in the desired form. The cosmetic is not particularly limited in form as long as it can be applied to the skin. Examples include liquid, emulsion, powder, solid, suspension, cream, ointment, mousse, granule, tablet, gel, jelly, paste, gel, aerosol, spray, liniment, pack, etc. These cosmetic products are used as cosmetic products for efficiently breaking down fat and reducing subcutaneous fat, etc.
本発明の内用組成物の用量、適用量は、使用する人の年齢、体重、健康状態、肥満度、疾患の状態等によって適宜決定することができるが、限定はされないが、成人1日当たり、グリアスペリンBの総量換算で、0.1μg以上、0.5μg以上、1μg以上、5μg以上、10μg以上、12μg以上とすることができ、5000μg以下、1000μg以下、500μg以下、100μg以下、60μg以下、20μg以下、15μg以下、などとすることができる。例えば、成人1日当たり、グリアスペリンBの総量換算で、0.1μg~5000μg、好ましくは0.5μg~1000μg、より好ましくは1μg~500μg、さらに好ましくは1μg~200μg、さらにより好ましくは1μg~100μg、特に好ましくは5μg~100μg、より特に好ましくは5μg~60μg、さらに特に好ましくは10μg~50μg、最も好ましくは10~20μgの用量で摂取される。 The dosage and application amount of the internal composition of the present invention can be appropriately determined depending on the age, body weight, health condition, degree of obesity, disease state, etc. of the user, but is not limited to, and can be, in terms of the total amount of gliaperin B, 0.1 μg or more, 0.5 μg or more, 1 μg or more, 5 μg or more, 10 μg or more, 12 μg or more per day for an adult, and can be 5000 μg or less, 1000 μg or less, 500 μg or less, 100 μg or less, 60 μg or less, 20 μg or less, 15 μg or less, etc. For example, the total amount of gliasperin B taken per day by an adult is 0.1 μg to 5000 μg, preferably 0.5 μg to 1000 μg, more preferably 1 μg to 500 μg, even more preferably 1 μg to 200 μg, even more preferably 1 μg to 100 μg, particularly preferably 5 μg to 100 μg, even more particularly preferably 5 μg to 60 μg, even more particularly preferably 10 μg to 50 μg, and most preferably 10 to 20 μg.
本発明により提供される内用組成物をヒトに経口投与する場合、グリアスペリンBを有効量投与することが好ましい。本明細書にて、有効量とは、直接的な作用であれ、間接的な作用であれ、目的の効果が見られる場合の投与量を言う。有効量としては、年齢や体重によっても異なるが、例えば、成人1日当たり、上記に説明したグリアスペリンBの総量換算で1μg以上、3μg以上、5μg以上、7μg以上、10μg以上、12μg以上とすることができ、1mg以下、500μg以下、200μg以下、100μg以下、60μg以下、50μg以下、20μg以下などとすることができる。例えば、成人1日当たり、グリアスペリンBの総量換算で、1μg以上1mg以下、好ましくは1μg以上500μg以下、より好ましくは1μg以上200μg以下、さらに好ましくは1μg以上100μg以下、さらにより好ましくは5μg以上100μg以下、特に好ましくは5μg以上60μg以下、より特に好ましくは10μg以上50μg以下、さらに特に好ましくは10μg以上20μg以下、最も好ましくは14μgである。 When the internal composition provided by the present invention is orally administered to humans, it is preferable to administer an effective amount of gliasperin B. In this specification, an effective amount refers to the dosage at which the desired effect is observed, whether it is a direct or indirect action. The effective amount varies depending on age and body weight, but can be, for example, 1 μg or more, 3 μg or more, 5 μg or more, 7 μg or more, 10 μg or more, or 12 μg or more per day for an adult, calculated as the total amount of gliasperin B described above, or 1 mg or less, 500 μg or less, 200 μg or less, 100 μg or less, 60 μg or less, 50 μg or less, or 20 μg or less. For example, the total amount of gliasperin B per day for an adult is 1 μg to 1 mg, preferably 1 μg to 500 μg, more preferably 1 μg to 200 μg, even more preferably 1 μg to 100 μg, even more preferably 5 μg to 100 μg, particularly preferably 5 μg to 60 μg, even more particularly preferably 10 μg to 50 μg, even more particularly preferably 10 μg to 20 μg, and most preferably 14 μg.
本発明の内用組成物は、体重減少効果が得られ易いという観点から、BMIが23kg/m2以上30kg/m2未満の成人に対して用いられることが好ましく、BMIが23kg/m2以上25kg/m2未満の成人に対して用いられることがより好ましい。日本肥満学会の判定基準によると、BMIが18.5kg/m2以上25kg/m2未満が普通体重とされ、25kg/m2以上30kg/m2未満が肥満度1とされる。本発明の内用組成物は、いわゆる普通体重~肥満気味(肥満度1)程度の成人に対して好ましく用いられ、肥満には分類されないが、普通体重のやや肥満よりに位置する成人に対して特に好ましく用いられる。なお、BMI(Body Mass Index)とは、ボディマス指数とも呼ばれ、体重と身長から下記の計算式によって算出される、肥満度を表す体格指数である。成人においてはBMIが国際的な指標として用いられている。
計算式:BMI=体重kg÷(身長m)2
From the viewpoint of easily obtaining a weight loss effect, the internal composition of the present invention is preferably used for adults with a BMI of 23 kg/m2 or more and less than 30 kg/ m2 , and more preferably for adults with a BMI of 23 kg/ m2 or more and less than 25 kg/ m2 . According to the criteria of the Japan Society for the Study of Obesity, a BMI of 18.5 kg/ m2 or more and less than 25 kg/ m2 is considered to be normal body weight, and a BMI of 25 kg/ m2 or more and less than 30 kg/ m2 is considered to be obesity level 1. The internal composition of the present invention is preferably used for adults with a so-called normal body weight to slightly obese (obesity level 1), and is particularly preferably used for adults who are not classified as obese but are slightly obese from a normal body weight. BMI (Body Mass Index) is also called body mass index, and is a body mass index that indicates the degree of obesity, calculated from body weight and height using the following formula. BMI is used as an international index for adults.
Calculation formula: BMI = weight kg ÷ (height m) 2
本発明の内用組成物は、脂肪分解促進効果、体重減少効果が得られ易いという観点から、内臓脂肪面積が100cm2以上の成人に対して好ましく用いられる。内臓脂肪面積は、腹部CT撮影を行い、臍部を中心として1cm間隔等で複数のスライス画像の撮影を行い、常法に従って算出することができる。 From the viewpoint of easily obtaining the effects of promoting lipolysis and reducing body weight, the internal use composition of the present invention is preferably used for adults with a visceral fat area of 100 cm2 or more. The visceral fat area can be calculated in the usual manner by performing abdominal CT scan and taking multiple slice images at intervals of 1 cm or more centered on the navel.
本発明の他の実施形態は、グリアスペリンBを有効成分として含有する、体重減少剤、内臓脂肪・体脂肪減少剤、BMIの改善剤、インスリン抵抗性改善剤、又はコレステロール値低減剤である。 Another embodiment of the present invention is a weight loss agent, an agent for reducing visceral fat and body fat, an agent for improving BMI, an agent for improving insulin resistance, or an agent for reducing cholesterol levels, which contains gliasperin B as an active ingredient.
本発明は上記体重減少剤、内臓脂肪・体脂肪減少剤、BMIの改善剤、インスリン抵抗性改善剤、又はコレステロール値低減剤も含み、これらは、グリアスペリンBを含有することにより、優れた脂肪分解促進作用を有すると共に、顕著な体重減少効果を奏する。本発明の体重減少剤、内臓脂肪・体脂肪減少剤、BMIの改善剤、インスリン抵抗性改善剤、又はコレステロール値低減剤を用いることで、脂肪細胞中の脂肪を効率よく分解し、皮下脂肪、内臓脂肪等の体脂肪を減少させることができ、結果的に顕著な体重減少効果、BMIの改善効果を得ることができる。また、インスリン抵抗性改善、これステロール値低減効果も得られる。よって、本発明の体重減少剤、内臓脂肪・体脂肪減少剤、BMIの改善剤、インスリン抵抗性改善剤、又はコレステロール値低減剤によると、肥満や生活習慣病を長期的に予防し、また改善することができ、健康の維持・増進を実現することができる。なお、本発明の体重減少剤、内臓脂肪・体脂肪減少剤、BMIの改善剤、インスリン抵抗性改善剤、又はコレステロール値低減剤は、グリアスペリンBを有効成分として含有する上述の本発明の内用組成物と同様の構成を有するため、具体的な説明は、本発明の内用組成物の項の説明をそのまま適用できる。 The present invention also includes the above-mentioned weight loss agent, visceral fat/body fat reduction agent, BMI improvement agent, insulin resistance improvement agent, or cholesterol level reduction agent, which contain gliasperin B, and thus have excellent fat decomposition promoting effects and a remarkable weight loss effect. By using the weight loss agent, visceral fat/body fat reduction agent, BMI improvement agent, insulin resistance improvement agent, or cholesterol level reduction agent of the present invention, fat in fat cells can be efficiently decomposed and body fat such as subcutaneous fat and visceral fat can be reduced, resulting in a remarkable weight loss effect and BMI improvement effect. Insulin resistance improvement and sterol level reduction effects can also be obtained. Therefore, the weight loss agent, visceral fat/body fat reduction agent, BMI improvement agent, insulin resistance improvement agent, or cholesterol level reduction agent of the present invention can prevent and improve obesity and lifestyle-related diseases in the long term, and can achieve the maintenance and promotion of health. In addition, since the weight loss agent, visceral fat/body fat reducing agent, BMI improving agent, insulin resistance improving agent, or cholesterol level reducing agent of the present invention has the same configuration as the above-mentioned internal composition of the present invention containing gliasperin B as an active ingredient, the specific explanation can be applied as is to the explanation in the section on the internal composition of the present invention.
<植物抽出物の脂肪分解及び/又は体重減少に対する有効性を判定する方法>
本発明は、植物抽出物について、グリアスペリンBの含有量を指標に、生体における脂肪分解及び/又は体重減少に対する有効性を判定する方法の発明も含む。上述のとおり、グリアスペリンBを有効成分として含有する製剤は、優れた脂肪分解促進作用を有すると共に、顕著な体重減少効果を奏する。また、脂肪細胞中の脂肪を効率よく分解し、皮下脂肪、内臓脂肪等の体脂肪を減少させることができ、結果的に顕著な体重減少効果、BMIの改善効果を得ることができる。また、インスリン抵抗性改善、これステロール値低減効果も得られる。グリアスペリンBの含有量に応じて、上記効果も増大することから、植物抽出物中のグリアスペリンBの含有量を指標とすることで、その植物抽出物の生体における脂肪分解及び/又は体重減少に対する有効性を判定することが可能となる。上記植物抽出物としては、グリアスペリンBを含有するものであれば特に限定されないが、中でも甘草抽出物が好ましい。
Methods for determining the efficacy of plant extracts for lipolysis and/or weight loss
The present invention also includes a method for determining the effectiveness of a plant extract in lipolysis and/or weight loss in a living body, using the content of gliasperin B as an index. As described above, a preparation containing gliasperin B as an active ingredient has an excellent lipolysis promoting effect and a remarkable weight loss effect. In addition, it is possible to efficiently decompose fat in adipocytes and reduce body fat such as subcutaneous fat and visceral fat, resulting in a remarkable weight loss effect and BMI improvement effect. In addition, it is possible to improve insulin resistance and reduce sterol levels. Since the above effects increase depending on the content of gliasperin B, it is possible to determine the effectiveness of the plant extract in lipolysis and/or weight loss in a living body by using the content of gliasperin B in the plant extract as an index. The above plant extract is not particularly limited as long as it contains gliasperin B, but licorice extract is preferable.
以下に、本発明の判定方法を詳細に説明する。本発明の判定方法は、植物抽出物におけるグリアスペリンBの含有量を測定する工程(以下、「測定工程」ともいう)を含む。また、得られたグリアスペリンB量を、基準となるグリアスペリンB量と比較する工程(以下、「比較工程」ともいう)を含むことが好ましい。 The determination method of the present invention is described in detail below. The determination method of the present invention includes a step of measuring the content of gliasperin B in the plant extract (hereinafter also referred to as the "measurement step"). It is also preferable that the method includes a step of comparing the obtained amount of gliasperin B with a standard amount of gliasperin B (hereinafter also referred to as the "comparison step").
上記測定工程における、植物抽出物中のグリアスペリンBの含有量を測定方法は特に限定されず、当業者に公知の方法により測定することができる。例えば、ウェスタンブロッティングを行う方法、ELISAによる方法等が挙げられる。上記ウェスタンブロッティングを行う方法、ELISAによる方法のいずれにおいても、グリアスペリンBに特異的に結合する抗グリアスペリンB抗体を使用し、グリアスペリンBの含有量を精度よく測定することができる。なお、ウェスタンブロッティングを行う方法においては、得られるグリアスペリンB特異的なブロットの強度を、濃度のわかる標準標品(ポジティブコントロール)の強度と比較することで、サンプル中のグリアスペリンB含有量を算出することができる。 In the above measurement step, the method for measuring the content of gliasperin B in the plant extract is not particularly limited, and can be measured by a method known to those skilled in the art. Examples include Western blotting and ELISA methods. In both the above Western blotting and ELISA methods, an anti-gliasperin B antibody that specifically binds to gliasperin B is used, and the content of gliasperin B can be measured with high accuracy. In the Western blotting method, the intensity of the resulting gliasperin B-specific blot can be compared with the intensity of a standard specimen (positive control) of known concentration, thereby calculating the gliasperin B content in the sample.
本発明の判定方法において基準として用いるグリアスペリンBとしては、例えば、天然供給源からの抽出、グリアスペリンBをコードする組換え核酸の発現、又は化学合成によって得られ得る。 Gliasperin B used as a standard in the determination method of the present invention can be obtained, for example, by extraction from a natural source, by expression of a recombinant nucleic acid encoding gliasperin B, or by chemical synthesis.
本発明の判定方法においては、本比較工程により、評価対象の抽出物におけるグリアスペリンBの含有量と、基準とするグリアスペリンBの含有量とを比較する。評価の目的、対象に合わせて、基準とするグリアスペリンB量を適宜選択することができる。 In the assessment method of the present invention, the comparison step involves comparing the content of gliasperin B in the extract to be evaluated with the content of the standard gliasperin B. The amount of gliasperin B to be used as the standard can be appropriately selected depending on the purpose and subject of the evaluation.
<植物抽出物の品質の指標>
本発明は、植物抽出物の品質の指標であって、上記品質が、生体における脂肪分解及び/又は体重減少に対する有効性に関する品質であり、グリアスペリンBからなることを特徴とする指標に係る発明も含む。上述のとおり、グリアスペリンBを有効成分として含有する製剤は、優れた脂肪分解促進作用を有すると共に、顕著な体重減少効果を奏する。また、脂肪細胞中の脂肪を効率よく分解し、皮下脂肪、内臓脂肪等の体脂肪を減少させることができ、結果的に顕著な体重減少効果、BMIの改善効果を得ることができる。また、インスリン抵抗性改善、これステロール値低減効果も得られる。グリアスペリンBの含有量に応じて、上記効果も増大することから、植物抽出物中のグリアスペリンBを指標とすることで、その植物抽出物の生体における脂肪分解及び/又は体重減少に対する有効性を判定することが可能となる。上記植物抽出物における植物としては甘草が好ましい。
<Indicators of plant extract quality>
The present invention also includes an invention relating to an index of the quality of a plant extract, the quality being related to the effectiveness of the plant extract in lipolysis and/or weight loss, and characterized in that the index is made of gliasperin B. As described above, a preparation containing gliasperin B as an active ingredient has an excellent lipolysis promoting effect and a remarkable weight loss effect. In addition, the preparation can efficiently decompose fat in adipocytes and reduce body fat such as subcutaneous fat and visceral fat, resulting in a remarkable weight loss effect and BMI improvement effect. In addition, the preparation can improve insulin resistance and reduce sterol levels. Since the above effects increase depending on the content of gliasperin B, it is possible to determine the effectiveness of the plant extract in lipolysis and/or weight loss in a living body by using gliasperin B in the plant extract as an index. Licorice is preferable as the plant in the plant extract.
本発明の植物抽出物の品質の指標は、上述の植物抽出物の脂肪分解及び/又は体重減少に対する有効性を判定する方法におけるグリアスペリンBに該当する。本発明の指標の具体的な説明は、上述の判定方法における説明を適用できる。 The indicator of the quality of the plant extract of the present invention corresponds to gliasperin B in the above-mentioned method for determining the effectiveness of the plant extract for lipolysis and/or weight loss. The specific explanation of the indicator of the present invention can be applied to the explanation of the above-mentioned determination method.
以下に実施例を示して本発明を更に詳しく説明するが、本発明の範囲をこれらに限定するものではない。 The present invention will be explained in more detail below with reference to examples, but the scope of the present invention is not limited to these examples.
以下に示すとおり、被験者において、被験食品(甘草抽出物を配合したハードカプセルの製剤)を12週間摂取した場合における内臓脂肪に及ぼす影響について評価するため、対照食品(甘草抽出物を配合しないハードカプセル)を対照とした無作為化二重盲検並行群間比較試験を実施した。 As shown below, a randomized, double-blind, parallel-group comparative study was conducted in subjects to evaluate the effect on visceral fat of taking the test food (a hard capsule formulation containing licorice extract) for 12 weeks, using a control food (hard capsule without licorice extract) as a control.
<試験1>
1.試験食品(組成、包装等)
被験食品は1カプセルあたり、甘草抽出物(エムジーファーマ(株)製;ウラル甘草由来)を100mg含有し、その他賦形剤で構成された。対照食品は甘草抽出物を含まず、賦形剤をカラメル色素で着色したもので構成された。試験食品の形状はいずれも白色のハードカプセルであり、30粒ずつアルミパウチ袋に包装され、試験食品に識別コードを表示した。試験食品と包装の識別不能性についてIRBにて審査され承認された。試験食品の栄養成分及び関与成分を下記表1に示した。また、被験食品に含まれるグリアスペリンB量は1日摂取目安量当たり14μgであった。
<Test 1>
1. Test food (composition, packaging, etc.)
The test food contained 100 mg of licorice extract (manufactured by MG Pharma Co., Ltd.; derived from Ural licorice) per capsule, and was composed of other excipients. The control food did not contain licorice extract, and was composed of excipients colored with caramel coloring. All test foods were in the form of white hard capsules, packaged in aluminum pouches of 30 capsules each, and an identification code was displayed on the test foods. The indistinguishability of the test foods and their packaging was reviewed and approved by the IRB. The nutritional components and related components of the test foods are shown in Table 1 below. The amount of gliasperin B contained in the test foods was 14 μg per recommended daily intake.
2.被験者
本試験における目標症例者数は、2群間での並行群間比較試験において、試験食品の効果量を大(0.8)と仮定し、有意水準を0.05、検出力を80~90%とすると、1群26~35名の組み入れが必要であることから、目標症例者数を1群34名、計68名と算出した。また、試験中に脱落、試験中止例が発生することを考慮しエントリー数を80名(1群40名)と設定した。被験者は、試験受託機関に登録しているボランティアの中から公募し、以下の選択基準を満たし、かつ除外基準に抵触せず試験責任医師が本試験への参加を適当であると判断した81名(男性50名、女性31名)を被験者として組み入れた。
2. Subjects The target number of subjects in this study was calculated to be 34 per group, for a total of 68 subjects, assuming that the effect size of the test food is large (0.8) in a parallel group comparison study between two groups, the significance level is 0.05, and the detection power is 80-90%, so that 26-35 subjects per group are required. In addition, the number of entries was set at 80 (40 per group) in consideration of cases that may drop out or be discontinued during the study. Subjects were recruited from volunteers registered with the study contracting institution, and 81 subjects (50 men, 31 women) who met the following inclusion criteria, did not violate the exclusion criteria, and were deemed appropriate for participation in this study by the study principal physician were included as subjects.
(選択基準)
スクリーニング検査において、以下の条件を満たす者とした。
(1)20歳以上65歳未満の男女、(2)BMIが23.0kg/m2以上30.0kg/m2未満の者、(3)内臓脂肪面積が100cm2以上の者、(4)本試験の目的、内容について十分な説明を受け、同意能力があり、よく理解した上で自発的に参加を志願し、書面で本試験参加に同意できる者。
(除外基準)
(1)重篤な心血管障害、肝機能障害、腎機能障害、呼吸器障害、内分泌障害、代謝障害に罹患している者、又はこれらの既往歴がある者、(2)脂質異常症、高血圧、糖尿病等の慢性疾患の治療を受けている者、(3)うつ病、統合失調症、神経性大食症等の精神疾患の者、(4)体脂肪、中性脂肪、体重、コレステロール、エネルギー代謝等の本試験の評価に影響を及ぼす可能性がある医薬品、医薬部外品を常用している者、(5)体脂肪、中性脂肪、体重、コレステロール、エネルギー代謝等の本試験の評価に影響を及ぼす可能性があるサプリメント・健康食品(特定保健用食品、機能性表示食品等)を常用している者、(6)試験食品に対してアレルギーを引き起こす可能性がある者、(7)腹部CT撮影部位に金属が入っている者、(8)心臓ペースメーカー、植込み型除細動器等の体内植込み型医療機器が入っている者、(9)閉所恐怖症の者、(10)喫煙習慣がある者、(11)多量飲酒(純アルコール60g/日以上)する習慣がある者、(12)食生活が極端に不規則な者、(13)夜勤及び昼夜交代制勤務の者、(14)5日以上排便のない者、(15)更年期の症状がある者、(16)高血圧症、偽アルドステロン症、低カリウム血症、ミオパチーを有する者、(17)甘草が配合された医薬品、及び甘草を主として含有する食品を常用している者、(18)むくみ、脱力感が起きやすい者、(19)排尿障害がある者、(20)試験開始前1ヶ月以内に200 mL、又は3ヶ月以内に400mLを超える採血(献血等)をした者、(21)過去4ヶ月以内に他の臨床試験に参加した者、及び現在他の臨床試験に参加中の者、試験期間中に他の臨床試験に参加する予定のある者、(22)妊娠中もしくは授乳中の者、又は妊娠の予定のある者、(23)その他試験責任医師が不適当と判断した者。
(Selection Criteria)
For the screening test, participants were required to meet the following conditions:
(1) Men and women aged 20 years or older and younger than 65 years, (2) individuals with a BMI of 23.0 kg/ m2 or more and less than 30.0 kg/ m2 , (3) individuals with a visceral fat area of 100 cm2 or more, (4) individuals who have been fully informed of the purpose and contents of this study, are capable of consent, have a full understanding of the study, voluntarily volunteer to participate, and can agree to participate in this study in writing.
(Exclusion Criteria)
(1) Persons suffering from or with a history of serious cardiovascular, hepatic, renal, respiratory, endocrine or metabolic disorders; (2) Persons receiving treatment for chronic diseases such as dyslipidemia, hypertension or diabetes; (3) Persons with mental illnesses such as depression, schizophrenia or bulimia nervosa; (4) Persons who regularly use medicines or quasi-drugs that may affect the evaluation of this study, such as body fat, triglycerides, body weight, cholesterol and energy metabolism; (5) Persons who regularly use supplements or health foods (specified health foods, functional foods, etc.) that may affect the evaluation of this study, such as body fat, triglycerides, body weight, cholesterol and energy metabolism; (6) Persons who may be allergic to the test foods. (7) Persons with metal in the area where the abdominal CT scan will be performed; (8) Persons with implanted medical devices such as a cardiac pacemaker or an implantable cardioverter defibrillator; (9) Persons with claustrophobia; (10) Persons with a habit of smoking; (11) Persons with a habit of drinking large amounts of alcohol (60 g or more of pure alcohol per day); (12) Persons with extremely irregular eating habits; (13) Persons working night shifts or day-night shifts; (14) Persons who have not had a bowel movement for five days or more; (15) Persons with menopausal symptoms; (16) Persons with hypertension, pseudoaldosteronism, hypokalemia, or myopathy; (17) Persons who regularly use medicines containing licorice or foods that mainly contain licorice; (18) Persons who are prone to edema or weakness; (19) Persons with urinary problems; (20) Persons who have had 200 (21) Those who have participated in other clinical trials within the past four months, those currently participating in other clinical trials, and those who plan to participate in other clinical trials during the study period; (22) Those who are pregnant or breastfeeding, or those who plan to become pregnant; (23) Those who are otherwise deemed inappropriate by the principal investigator of the study.
3.試験プロトコール
本試験は無作為化プラセボ対照二重盲検並行群間比較試験とした。試験食品の摂取開始時、摂取4週間後、摂取8週間後、摂取12週間後に検査を実施した。試験責任者によって組み入れられた被験者に対し、試験の実施に関与しない割付責任者が、ランダム化のため乱数を発生させ被験者に割付けた。この際、割付因子である同意取得時の性別、年齢、スクリーニング検査時の内臓脂肪面積、BMI、ヒップ周囲径、ウエスト周囲径において、群間で有意な差がないことを確認した。
3. Study protocol This study was a randomized, placebo-controlled, double-blind, parallel-group comparative study. Tests were conducted at the start of intake of the test food, and after 4 weeks, 8 weeks, and 12 weeks of intake. A random number was generated for randomization by the allocation manager, who was not involved in the implementation of the study, and the subjects were assigned to the subjects who were enrolled by the study director. At this time, it was confirmed that there were no significant differences between the groups in the allocation factors of sex and age at the time of consent acquisition, and visceral fat area, BMI, hip circumference, and waist circumference at the time of screening examination.
試験期間中は、1日1回、1カプセルの試験食品(被験食品群には被験食品、対照食品群には対照食品)を夕食前に水又はぬるま湯とともに摂取させた。但し、夕食前に摂取ができなかった場合は可能なタイミングで1日1回、1カプセルを摂取させた。 During the study period, subjects were instructed to take one capsule of the test food (test food for the test food group, control food for the control food group) once a day with water or lukewarm water before dinner. However, if subjects were unable to take the test food before dinner, they were instructed to take one capsule once a day at a time that was possible.
試験期間中、摂取する飲食物については通常の習慣を極力維持することとし、体脂肪、中性脂肪、体重、コレステロール、エネルギー代謝等の本試験の評価に影響を及ぼす可能性がある健康食品(特定保健用食品、機能性表示食品等)、及びグレープフルーツジュースの摂取を禁止した。また、アルコール摂取量に関しては通常の摂取習慣を維持することとし、通常摂取する量を逸脱する飲み方を禁止した。なお、検査前日のアルコール摂取は禁止とし、21時以降は水以外の摂取を禁止した。また、腹部CT撮影の4時間前からは水以外の摂取を禁止した。試験期間中、通常の運動習慣(頻度、運動の種類等)を極力維持することとし、運動習慣を変えることを禁止した。試験期間中、緊急の場合を除き、新たな医薬品を使用する場合は試験責任医師の許可を得ることとした。検査日前2週間はワクチンの接種を禁止した。試験期間中の献血を禁止した。 During the study period, subjects were instructed to maintain normal eating and drinking habits as much as possible, and were prohibited from consuming health foods (foods for specified health uses, foods with functional claims, etc.) that may affect the study's evaluations of body fat, neutral fat, body weight, cholesterol, energy metabolism, etc., as well as grapefruit juice. In addition, subjects were instructed to maintain normal alcohol intake habits, and were prohibited from drinking amounts that deviated from normal intake. In addition, subjects were prohibited from consuming alcohol the day before the study, and were prohibited from consuming anything other than water after 9 p.m. In addition, subjects were prohibited from consuming anything other than water four hours before the abdominal CT scan. During the study period, subjects were instructed to maintain normal exercise habits (frequency, type of exercise, etc.), and were prohibited from changing their exercise habits. During the study period, except in emergency cases, permission from the study physician was required before using any new medications. Vaccinations were prohibited for two weeks prior to the study. Blood donation was prohibited during the study period.
本試験はヘルシンキ宣言(2013)-東京、ベニス、香港、サマーセットウェスト、エジンバラ修正、ワシントン、東京注釈追加、ソウル修正、フォルタレザ修正-の精神に則り、「人を対象とする生命科学・医学系研究に関する倫理指針(令和3年3月23日策定、令和3年6月30日施行、令和4年3月10日一部改正)」に従って実施した。実施するにあたり医療法人弘正会ふくだ内科クリニック倫理審査委員会(IRB)の承認を得た(承認番号:IRB-20220716-2)。また、大学病院医療情報ネットワーク臨床試験登録システム(UMIN)に臨床試験登録を行った(UMIN試験ID:UMIN000048430)。 This study was conducted in accordance with the spirit of the Declaration of Helsinki (2013) - Tokyo, Venice, Hong Kong, Somerset West, Edinburgh Amendment, Washington, Tokyo Annotation Added, Seoul Amendment, and Fortaleza Amendment - and in accordance with the "Ethical Guidelines for Life Science and Medical Research Involving Human Subjects (established March 23, 2021, enforced June 30, 2021, partially revised March 10, 2022)". Approval was obtained from the Institutional Review Board (IRB) of the Fukuda Naika Clinic, a medical corporation (approval number: IRB-20220716-2). In addition, the clinical trial was registered in the University Hospital Medical Information Network Clinical Trial Registration System (UMIN) (UMIN Study ID: UMIN000048430).
4.検査項目
1)身体検査及び腹部CT撮影
身体検査の項目は身長(スクリーニング検査時のみ)、InBody770((株)インボディ・ジャパン)を用いた体組成評価(体重、BMI、体脂肪率、筋肉量、部位別体脂肪量、基礎代謝、その他体組成成分分析等)、ウエスト周囲径、ヒップ周囲径、血圧及び脈拍の測定を実施し、選択基準に合致した者についてCT撮影を実施した。体重については、CT撮影時のみ、DST-210N(ムラテックKDS(株))を使用した。また、BMIは身長と体重から算出し、CT撮影時のBMIを摂取開始前の値とした。
4. Examination items 1) Physical examination and abdominal CT scan Physical examination items included height (only at the time of screening test), body composition evaluation using InBody770 (InBody Japan, Inc.) (weight, BMI, body fat percentage, muscle mass, body fat mass by region, basal metabolism, other body composition component analysis, etc.), waist circumference, hip circumference, blood pressure, and pulse rate, and CT scans were performed on those who met the selection criteria. For weight, DST-210N (Muratec KDS, Ltd.) was used only at the time of CT scan. BMI was calculated from height and weight, and the BMI at the time of CT scan was used as the value before starting intake.
腹部CT撮影はDiscovery710又はDiscovery MIDR(GEヘルスケア・ジャパン(株))を使用し、臍部を中心として1cm間隔で3スライスの画像の撮影を行い、内臓脂肪面積、皮下脂肪面積及び総脂肪面積について、その平均値を算出した。なお、臍部の位置撮影時の画像に腎臓、腸骨が含まれる場合には、第4腰椎を中心に再撮影を実施した。 Abdominal CT scans were performed using a Discovery 710 or Discovery MIDR (GE Healthcare Japan, Ltd.), with three slices taken at 1 cm intervals centered on the navel, and the average values of visceral fat area, subcutaneous fat area, and total fat area were calculated. If the kidneys or ilium were included in the image taken at the navel position, a second image was taken centered on the fourth lumbar vertebra.
2)食事記録及び活動記録
試験期間中、被験者にアルコール摂取量、医薬品の使用、食事内容等について生活記録日誌に記録させた。試験食品摂取期間中は試験食品の摂取について記録させた。また、検査日前3日間は食事内容に加えて量を記録させた。試験期間中の就寝時以外は被験者に活動量計(SAT-1、(株)メディシンク)を装着させ歩数、Active歩数、中強度活動時間を2週間毎に確認した。被験者の健康状態については、摂取4、8、12週間後に問診と医師診察を行い、自覚症状及び他覚症状の発生状況等を把握した。
2) Food and activity records During the test period, subjects were asked to record alcohol intake, drug use, dietary content, etc. in a daily diary. During the test food intake period, subjects were asked to record the intake of the test food. In addition, subjects were asked to record the amount of food consumed in addition to the content of the food consumed for three days prior to the test. During the test period, subjects were asked to wear an activity monitor (SAT-1, Medithink Co., Ltd.) except when sleeping, and the number of steps, active steps, and duration of moderate intensity activity were checked every two weeks. Regarding the health condition of the subjects, interviews and medical examinations were conducted by a doctor 4, 8, and 12 weeks after intake to ascertain the occurrence of subjective and objective symptoms, etc.
3)疲労感及び睡眠評価
疲労感について、摂取開始時、摂取4、8、12週間後に被験者の自宅にて起床時にVAS法による評価を実施した。睡眠の評価について、摂取開始時、摂取4、8、12週間後に被験者の自宅にて起床時にOSA睡眠調査票MA版を用いた評価を実施した。
3) Fatigue and sleep evaluation Fatigue was evaluated by VAS at the start of intake, and 4, 8, and 12 weeks after intake, when the subjects woke up at home. Sleep was evaluated by the OSA sleep questionnaire MA version at the start of intake, and 4, 8, and 12 weeks after intake, when the subjects woke up at home.
4)血液検査及び尿検査
血液検査は摂取前、摂取4、8、12週間後に採血を行い、WBC、RBC、Hb、Ht、MCV、MCH、MCHC、Plt、白血球分画、総蛋白、Alb、A/G比、AST、ALT、γ-GTP、クレアチニン、血糖、総コレステロール(T-Cho)、HDLコレステロール(HDL-Cho)、LDLコレステロール(LDL-Cho)、中性脂肪(TG)、CPK、尿酸(UA)、尿素窒素(UN)、ALP、LDH、Na、K、Cl、Ca、Mg、P、総ビリルビン(T-Bil)、HbA1c、グリコアルブミン、インスリン、ケトン体分画(総ケトン体、アセト酢酸、3-ヒドロキシ酪酸)について検査を行った。尿検査は摂取前及び摂取12週間後に糖、タンパク、潜血、ウロビリノーゲンについて検査を実施した。
4) Blood and urine tests Blood was taken before intake and 4, 8, and 12 weeks after intake for WBC, RBC, Hb, Ht, MCV, MCH, MCHC, Plt, white blood cell fraction, total protein, Alb, A/G ratio, AST, ALT, γ-GTP, creatinine, blood glucose, total cholesterol (T-Cho), HDL cholesterol (HDL-Cho), LDL cholesterol (LDL-Cho), triglyceride (TG), CPK, uric acid (UA), urea nitrogen (UN), ALP, LDH, Na, K, Cl, Ca, Mg, P, total bilirubin (T-Bil), HbA1c, glycoalbumin, insulin, and ketone body fraction (total ketone bodies, acetoacetic acid, 3-hydroxybutyric acid). Urine tests were performed for sugar, protein, occult blood, and urobilinogen before and 12 weeks after intake.
5.統計解析
主要評価項目は腹部CT撮影における内臓脂肪面積とした。副次評価項目は体重、BMI、腹部CT撮影における皮下脂肪面積、総脂肪面積、InBody 770における体脂肪率、体脂肪量、筋肉量、部位別体脂肪量、基礎代謝量、ウエスト周囲径、ヒップ周囲径、ウエスト/ヒップ比、VAS(疲労感)、OSA睡眠調査票MA版、血液検査におけるケトン体分画、T-Cho、LDL-Cho、HDL-Cho、TG、血糖、HbA1c、インスリン、グリコアルブミンとした。
5. Statistical analysis The primary outcome was visceral fat area in abdominal CT scans. Secondary outcome measures were body weight, BMI, subcutaneous fat area in abdominal CT scans, total fat area, body fat percentage, body fat mass, muscle mass, body fat mass by region, basal metabolic rate, waist circumference, hip circumference, waist/hip ratio, VAS (feeling of fatigue), OSA Sleep Questionnaire MA version, ketone body fraction in blood tests, T-Cho, LDL-Cho, HDL-Cho, TG, blood glucose, HbA1c, insulin, and glycoalbumin.
統計解析は統計処理ソフトSPSS Ver.26(日本アイ・ビー・エム(株))を使用し、有意水準は両側検定で5%とした。また、被験者属性による傾向を確認するため、摂取開始前のBMIが23kg/m2以上25kg/m2未満の者、BMIが25kg/m2以上30kg/m2未満の者及び性別によるサブグループ解析を実施した。各検査時期の値及び摂取開始前から各検査時期の変化量について平均値及び標準偏差を群別に集計し、各検査時期における被験食品群と対照食品群の平均値の差について対応のないt検定を実施し、群間差を比較した。また、各試験食品群における摂取開始前と各検査時期の平均値の差についてDunnettの多重比較検定を実施し、試験食品の摂取期間による差を比較した。腹部CT撮影については、対応のあるt検定を実施し、試験食品の摂取期間による差を比較した。なお、対応のあるt検定については多重性の補正を行わなかった。 Statistical analysis was performed using statistical processing software SPSS Ver. 26 (IBM Japan, Ltd.), and the significance level was set at 5% in a two-sided test. In addition, in order to confirm the tendency according to the subject attributes, subgroup analysis was performed according to the subjects with a BMI of 23 kg/ m2 or more and less than 25 kg/ m2 before the start of intake, the subjects with a BMI of 25 kg/ m2 or more and less than 30 kg/ m2 , and the gender. The average value and standard deviation of the values at each test time and the change amount from before the start of intake to each test time were tabulated by group, and an unpaired t-test was performed on the difference in the average value between the test food group and the control food group at each test time to compare the differences between the groups. In addition, Dunnett's multiple comparison test was performed on the difference in the average value between before the start of intake and each test time in each test food group to compare the differences due to the intake period of the test food. For abdominal CT, a paired t-test was performed to compare the differences due to the intake period of the test food. Note that no correction for multiplicity was performed for the paired t-test.
(結果)
1.解析対象者
本試験で組み入れられた被験者は81名(被験食品群41名、対照食品群40名)であったが、ランダム化後に被験食品群において1名が除外基準(2)に抵触する治療を開始するため試験中止症例となり80名で試験を開始した。試験食品摂取開始後に、被験食品群において除外基準(15)に抵触していることが試験開始後に判明した1名、対照食品群において試験食品を一度も摂取しておらず自己都合により試験を中止した1名を除く78名(被験食品群39名、対照食品群39名)を安全性解析集団(FAS)とした。また、試験食品摂取期間中に被験食品群において自己免疫性膵炎の発症による試験中止が1名、対照食品群で自己都合による試験中止が1名発生したため、有効性解析集団(PPS)は76名(被験食品群38名、対照食品群38名)とした、また、対照食品群において一部評価項目のデータの信頼性に課題のあるため当該項目についてのみ解析から除外した、部分解析除外が2例発生した。1例は血液サンプルに溶血が認められたため、摂取4週後の血液検査におけるLDH、K、インスリンを解析から除外した。1例は検査当日の食事摂取(管理事項の不遵守)が疑われたため、8週間後の血液検査における血糖、インスリンを解析から除外した。有効性解析対象者の被験者背景を表2に示した。
(result)
1. Subjects analyzed Eighty-one subjects were enrolled in this study (41 in the test food group, 40 in the control food group), but after randomization, one subject in the test food group discontinued the study due to initiating treatment that violated the exclusion criterion (2), and the study began with 80 subjects. One subject in the test food group who was found to violate the exclusion criterion (15) after the start of the study after starting to take the test food, and one subject in the control food group who had never taken the test food and discontinued the study for personal reasons, were excluded, leaving 78 subjects (39 in the test food group, 39 in the control food group) as the safety analysis set (FAS). In addition, one patient in the test food group discontinued the study due to the onset of autoimmune pancreatitis during the test food intake period, and one patient in the control food group discontinued the study for personal reasons, so the efficacy analysis set (PPS) was set at 76 patients (38 in the test food group and 38 in the control food group). In addition, two patients in the control food group were excluded from the analysis for only those items due to issues with the reliability of the data for some evaluation items. In one patient, hemolysis was observed in the blood sample, so LDH, K, and insulin in the blood test 4 weeks after intake were excluded from the analysis. In one patient, food intake on the day of the test (non-compliance with management items) was suspected, so blood glucose and insulin in the blood test 8 weeks after intake were excluded from the analysis. The subject background of the subjects in the efficacy analysis is shown in Table 2.
FAS、PPS、BMIによるサブグループ、性別によるサブグループ、いずれの集団においても割付因子である同意取得時の性別、年齢、スクリーニング時の内臓脂肪面積、BMI、ウエスト周囲径、ヒップ周囲径において、被験食品群と対照食品群の間に有意差はみられなかった。 In none of the groups (FAS, PPS, subgroups by BMI, or subgroups by gender) were there any significant differences between the test food group and the control food group in the allocation factors of gender at the time of consent, age, visceral fat area at screening, BMI, waist circumference, or hip circumference.
2.解析結果
(腹部脂肪面積)
有効性解析集団(PPS)における内臓脂肪面積、皮下脂肪面積及び総脂肪面積の変化を表3に示す。主要評価項目である内臓脂肪面積について、被験食品群と対照食品群との間に有意な差はみられなかった。被験食品群内の経時変化において、内臓脂肪面積と総脂肪面積について、摂取12週間後に摂取開始前と比較して有意な減少がみられた。
2. Analysis results (abdominal fat area)
The changes in visceral fat area, subcutaneous fat area, and total fat area in the efficacy analysis set (PPS) are shown in Table 3. No significant difference was observed between the test food group and the control food group in visceral fat area, the primary endpoint. In the test food group, a significant decrease was observed in visceral fat area and total fat area after 12 weeks of intake compared to before intake began.
(身体計測項目)
PPSにおける体重、BMI、体脂肪量、筋肉量、部位別体脂肪量、ウエスト周囲径及びヒップ周囲径、またFASにおける血圧及び脈拍数の変化を表4に示す。血圧及び脈拍数について、摂取4週間後に被験食品群は対照食品群と比較して、収縮期血圧、拡張期血圧に有意な高値がみられ、脈拍数の変化量に有意な高値がみられたが、それ以降有意な変動は認められなかった。その他の各評価項目について、被験食品群と対照食品群との間に有意な差はみられなかった。
(Physical measurement items)
Table 4 shows changes in body weight, BMI, body fat mass, muscle mass, body fat mass by region, waist circumference, and hip circumference in the PPS, as well as blood pressure and pulse rate in the FAS. Four weeks after ingestion, the test food group showed significantly higher systolic and diastolic blood pressure values and significantly higher changes in pulse rate compared to the control food group, but no significant changes were observed thereafter. No significant differences were observed between the test food group and the control food group in any of the other evaluation items.
(血液検査及び尿検査)
PPSにおける血液検査項目(副次評価項目)の変化を表5に示す。T-Choについて摂取4週間後に被験食品群は対照食品群と比較して摂取開始前からの変化量に有意な低下抑制がみられた。血糖について被験食品群は対照食品群と比較して摂取開始前から優位に高値を示し、摂取4週間後においても有意に高値を示したが、それ以降差はみられなかった。インスリン値について被験食品群は対照食品群と比較して有意な低下がみられた。また、FASにおける他の血液検査項目及び尿検査項目について、いずれの被験者も臨床上問題となる変化は認められなかった。
(Blood and urine tests)
Table 5 shows the changes in blood test items (secondary evaluation items) in the PPS. For T-Cho, the test food group showed a significant suppression of the change from before the start of intake compared to the control food group after 4 weeks of intake. For blood glucose, the test food group showed significantly higher values than the control food group before the start of intake, and showed significantly higher values even after 4 weeks of intake, but no difference was observed thereafter. For insulin levels, the test food group showed a significant decrease compared to the control food group. In addition, no clinically significant changes were observed in any of the subjects for other blood test items and urine test items in the FAS.
(活動量計)
試験期間中の活動量について、歩数、Active歩数、中強度活動時間を2週間毎に確認したが、被験食品と対照食品群との間に有意な差はみられなかった。
(Activity monitor)
During the study period, activity levels were checked every two weeks in terms of number of steps, active steps, and duration of moderate intensity activity, but no significant differences were found between the test food and control food groups.
(サブグループ解析)
摂取開始前のBMIが23kg/m2以上25kg/m2未満のサブグループ解析の結果を表6に示す。摂取開始前のBMIが23kg/m2以上25kg/m2未満のサブグループ解析における、腹部脂肪面積の評価項目において、内臓脂肪面積について被験食品群は対照食品群と比較して、摂取12週間後に有意な低値がみられた。また、皮下脂肪面積及び総脂肪面積の試験開始前からの変化量について摂取12週間後に有意な減少がみられた。また身体計測項目において、被験食品群は対照食品群と比較して、体重及びBMIについて摂取開始前からの変化量について摂取12週間後に有意な減少がみられた。さらに血液検査の評価項目において、被験食品群は対照食品群と比較して、総ケトン体、アセト酢酸及び3-ヒドロキシ酪酸について摂取12週間後に、摂取開始前からの変化量の有意な上昇がみられた。HDL-Choについて摂取4、8、12週間後に有意な上昇がみられた。TGについて摂取12週間後に有意な低値、摂取開始前からの変化量の有意な低下がみられた。血糖値について摂取前、摂取4、8週間後に有意な高値がみられた。インスリンについて摂取12週間後に、摂取開始前からの変化量の有意な上昇抑制がみられた。
(Subgroup analysis)
The results of the subgroup analysis of BMI before the start of intake from 23 kg/ m2 to less than 25 kg/ m2 are shown in Table 6. In the subgroup analysis of BMI before the start of intake from 23 kg/ m2 to less than 25 kg/ m2 , the test food group showed a significantly lower visceral fat area after 12 weeks of intake compared to the control food group in the evaluation item of abdominal fat area. In addition, a significant decrease was observed in the change in subcutaneous fat area and total fat area from before the start of the study after 12 weeks of intake. In addition, in the physical measurement items, the test food group showed a significant decrease in the change in weight and BMI from before the start of intake after 12 weeks of intake compared to the control food group. Furthermore, in the evaluation items of blood tests, the test food group showed a significant increase in the change from before the start of intake for total ketone bodies, acetoacetate, and 3-hydroxybutyrate after 12 weeks of intake compared to the control food group. A significant increase was observed in HDL-Cho after 4, 8, and 12 weeks of intake. For TG, a significant low was observed after 12 weeks of intake, and a significant decrease in the amount of change from before the start of intake was observed. For blood glucose, a significant high was observed before intake, and 4 and 8 weeks after intake. For insulin, a significant increase in the amount of change from before the start of intake was suppressed after 12 weeks of intake.
摂取開始前のBMIが25kg/m2以上30kg/m2未満のサブグループ解析、性別によるサブグループ解析においては、いずれの評価項目においても被験食品群と対照食品群の間に有意な差は認められなかった。 In the subgroup analysis of those with a BMI of 25 kg/ m2 or greater but less than 30 kg/ m2 before intake and in the subgroup analysis by gender, no significant differences were observed between the test food group and the control food group in any of the evaluation items.
(考察)
本試験では、甘草抽出物含有食品の体脂肪低減作用についてBMIが23kg/m2以上30kg/m2未満、腹部内臓脂肪面積が100cm2以上の健康成人男女を対象に甘草抽出物を100mg含有するカプセル(被験食品)又は甘草抽出物を含有しないカプセル(対照食品)を1日1回1カプセル、12週間摂取させる無作為化プラセボ対照二重盲検並行群間比較試験にて検討した。結果、BMIが23kg/m2以上25kg/m2未満のサブグループにおいて、被験食品群は対照食品群と比較して、摂取12週間後の内臓脂肪面積について有意な低値がみられ、皮下脂肪面積及び総脂肪面積の試験開始前からの変化量、また体重及びBMIの試験開始前からの変化量について、有意な減少がみられたことから甘草抽出物はヒトにおいても体脂肪低減作用を有することが示され、それに伴う体重及びBMI低下がみられた。
(Discussion)
In this study, the body fat reducing effect of licorice extract-containing food was examined in healthy adult men and women with a BMI of 23 kg/ m2 or more but less than 30 kg/ m2 and an abdominal visceral fat area of 100 cm2 or more in a randomized placebo-controlled double-blind parallel group comparative study in which a capsule containing 100 mg of licorice extract (test food) or a capsule not containing licorice extract (control food) was taken once a day for 12 weeks. As a result, in the subgroup with a BMI of 23 kg/ m2 or more but less than 25 kg/ m2 , the test food group showed significantly lower visceral fat area after 12 weeks of intake compared to the control food group, and the changes in subcutaneous fat area and total fat area from before the start of the study, as well as the changes in body weight and BMI from before the start of the study, were significantly reduced, indicating that licorice extract has a body fat reducing effect in humans as well, and associated decreases in body weight and BMI were observed.
甘草抽出物の体脂肪低減作用の機序については、細胞試験において脂肪細胞中のTGの分解を促進することが後述する試験により確認された。動物試験においても、体脂肪蓄積抑制や脂肪細胞のサイズ減少が確認されており、細胞試験の結果を支持する結果が報告されている。本試験においても体脂肪低減作用が認められたが、その際に総ケトン体、アセト酢酸、3-ヒドロキシ酪酸について、被験食品群は対照食品群と比較して有意な上昇がみられた。血中ケトン体の上昇はβ酸化による脂肪酸の消費の指標でありインスリン抵抗性を示す際にもみられるが、血糖値の上昇やインスリン値の上昇も認められておらず、インスリン抵抗性の増悪によるものでは無く、他の機能性食品等でも報告のあるエネルギー消費の亢進によるものと考えられた。 Regarding the mechanism of licorice extract's body fat reducing effect, the following test confirmed that it promotes the breakdown of TG in fat cells in cell tests. In animal tests, inhibition of body fat accumulation and reduction in fat cell size were also confirmed, and results that support the results of the cell tests have been reported. A body fat reducing effect was also observed in this test, and significant increases were observed in total ketone bodies, acetoacetic acid, and 3-hydroxybutyric acid in the test food group compared to the control food group. An increase in blood ketone bodies is an indicator of fatty acid consumption through β-oxidation and is also seen in cases of insulin resistance, but no increase in blood glucose or insulin levels was observed, and it was thought that this was not due to an increase in insulin resistance, but rather due to increased energy consumption, as has been reported in other functional foods.
これらの結果から、甘草抽出物摂取により脂肪細胞でTGが分解された際に、遊離脂肪酸が放出され、速やかにβ酸化によりエネルギー源として消費された結果として体脂肪低減作用をもたらしたことが示唆された。 These results suggest that when TGs are broken down in fat cells following ingestion of licorice extract, free fatty acids are released and quickly consumed as an energy source through β-oxidation, resulting in a reduction in body fat.
後述する試験により脂肪細胞中のTG分解については、甘草抽出物中のグリアスペリンBがほぼ100%の寄与率を示すことが確認されている。以上のことから、甘草抽出物摂取による体脂肪低減作用はグリアスペリンBによるものであると考えられた。 The tests described below have confirmed that gliasperin B in licorice extract contributes nearly 100% to TG decomposition in fat cells. Based on the above, it is believed that the body fat reducing effect of ingesting licorice extract is due to gliasperin B.
本試験では、甘草抽出物の安全性についても確認した。その結果、臨床検査値の異常を含め、甘草抽出物摂取に起因する有害事象は認められておらず、甘草抽出物の長期摂取における安全性に問題が無いと考えられた。 The safety of licorice extract was also confirmed in this study. As a result, no adverse events attributable to the intake of licorice extract, including abnormalities in clinical test values, were observed, and it was considered that there were no issues with the safety of long-term intake of licorice extract.
(結論)
甘草抽出物含有食品は、肥満気味(BMIが23kg/m2以上、25kg/m2未満。腹部内臓脂肪面積が100cm2以上。)の健康成人男女において、体脂肪低減作用を有することが示された。また、その作用機序の一端として、脂肪細胞中のTGの分解作用、それに伴う脂肪酸消費(β酸化)の亢進が考えられた。
(Conclusion)
Licorice extract-containing foods have been shown to have a body fat reducing effect in healthy adult men and women who are slightly obese (BMI 23 kg/ m2 or more and less than 25 kg/ m2 , abdominal visceral fat area 100 cm2 or more). In addition, it is thought that part of the mechanism of action is the decomposition of TG in fat cells and the accompanying enhancement of fatty acid consumption (β-oxidation).
<試験2>
5週齢の雄性C57BL/6Jマウスを6匹ずつに分け、MF(オリエンタル酵母)、高脂肪高糖質食粉末飼料(以下HFSと略す、D12079BM、Research Diet)又は甘草抽出物を混合したHFSを与えて8週間飼育した。摂取8週間後に体重および内臓脂肪重量(副睾丸周囲脂肪重量、腎周囲脂肪重量及び腸間膜脂肪重量の合算)を測定した。なお、甘草抽出物として、甘草の根及びストロンの部分を粉砕して得た粉末から、50%(v/v)エタノール抽出工程を経て得られた液を、エバポレーター及び凍結乾燥にて乾固した後粉砕し、甘草抽出物を得た。これらを、それぞれ飼料に0.3%混合して与えた。甘草抽出物に含まれるグリアスペリンB量及び、マウスの体重増加量と内臓脂肪重量を表7に示す。
<Test 2>
Five-week-old male C57BL/6J mice were divided into groups of six and fed MF (Oriental Yeast), high-fat, high-carbohydrate powdered feed (hereinafter abbreviated as HFS, D12079BM, Research Diet), or HFS mixed with licorice extract for 8 weeks. After 8 weeks of ingestion, the body weight and visceral fat weight (the sum of epididymal fat weight, perirenal fat weight, and mesenteric fat weight) were measured. The licorice extract was obtained by subjecting the powder obtained by crushing the root and stolon of licorice to a 50% (v/v) ethanol extraction process, drying the liquid by an evaporator and freeze-drying, and then crushing the liquid. These were each mixed with the feed at 0.3% and fed to the mice. The amount of gliasperin B contained in the licorice extract, and the body weight gain and visceral fat weight of the mice are shown in Table 7.
(結論)
甘草抽出物の中でも、グリアスペリンBの配合量が多いものを摂取したマウスほど、体重の増加が緩やかであり、内臓脂肪重量も軽かった。このことから、甘草抽出物の体脂肪・内臓脂肪低減作用にはグリアスペリンBが大きく関与しており、ひいては試験例1の結果にも関与していることが示唆された。
(Conclusion)
Mice that ingested licorice extract containing a higher amount of gliasperin B experienced a slower weight gain and a lower visceral fat weight. This suggests that gliasperin B is closely involved in the body fat/visceral fat reducing effect of licorice extract, and is also involved in the results of Test Example 1.
<試験3>グリアスペリンBの調製
カンゾウ抽出物1gあたり10mlの蒸留水を加えて溶解させカンゾウ抽出物水溶液を調製した。この水溶液の容量に対して半量の酢酸エチルを加えて混和し二層に分離した状態とし、そこから酢酸エチル層を回収する操作を3度繰り返した。得られた酢酸エチル層に無水硫酸ナトリウムを加えた後、ろ過をすることで有機画分を得た。有機画分をエバポレーターにて濃縮した後、シリカゲルカラムクロマトグラフにて、ヘキサン:酢酸エチル=1:1(v/v)で溶出した画分を、ODSカラムを用いた高速液体クロマトグラフ(アセトニトリル:水=60:40,45:55(いずれもv/v))により精製を繰り返すことで、化合物(グリアスペリンB)を得た。ここで得られたグリアスペリンB(GB)を以下のマウス前駆脂肪細胞3T3-L1を使用した試験において被験物質として用いた。
<Test 3> Preparation of
なお、上記カンゾウ抽出物は、以下の方法によって得られたものである。即ち、甘草の茎、根及びストロンの部分を粉砕し、甘草粉末を得た。甘草粉末50gを20℃で含水50%エタノール500mLを用いて抽出して得られた抽出物を凍結及びエバポレーターにて乾燥し、甘草の含水50%エタノール抽出物を得た。 The above licorice extract was obtained by the following method. That is, the stem, root, and stolons of licorice were crushed to obtain licorice powder. 50 g of licorice powder was extracted with 500 mL of 50% aqueous ethanol at 20°C, and the resulting extract was frozen and dried in an evaporator to obtain a 50% aqueous ethanol extract of licorice.
化合物のスペクトルデータは以下に示す通りであり、文献(Zeng L. et al., Heterocycles 34:575-587(1992))においてGlycyrrhiza属植物より単離報告がなされているグリアスペリンBのスペクトルデータと非常によい一致を示しており、構造情報との矛盾が認められないことから、化合物がグリアスペリンB(GB)であると同定した。 The spectral data of the compound is shown below, and shows a very good agreement with the spectral data of gliasperin B, which has been reported in the literature as having been isolated from plants of the genus Glycyrrhiza (Zeng L. et al., Heterocycles 34:575-587 (1992)). Since there was no inconsistency with the structural information, the compound was identified as gliasperin B (GB).
1H-NMR(重アセトン)ppm:1.63(3H, s), 1.74(3H, s), 3.24(2H, d, J=7.0 Hz), 3.90(3H, s), 4.27(1H, dd, J=5.5及び10.5 Hz), 4.48(1H, dd, J=5.0及び11.5 Hz), 4.62(1H, t, J=10.5 Hz), 5.17(1H, t, J=7.0 Hz), 6.13(1H, s), 6.33(1H, dd, J=2.0及び8.5 Hz), 6.45(1H, d, J=2.0 Hz), 6.94(1H, d, J=8.5Hz), 8.30(1H, bs), 8.63(1H, bs), 12.48(1H, s)
13C-NMR(重アセトン)ppm:17.6, 21.4, 25.7, 47.2, 56.2, 71.0, 91.3, 103.5, 103.7, 107.6, 109.7, 113.5, 123.3, 131.1,131.5, 156.8, 158.7, 161.1, 162.7, 165.8, 198.7
ESI+-MS m/z:393.13059[M+Na]+(計算値、C21H22O6Na:393.13086)
1H-NMR (acetone-d) ppm: 1.63 (3H, s), 1.74 (3H, s), 3.24 (2H, d, J = 7.0 Hz), 3.90 (3H, s), 4.27 (1H, dd, J = 5.5 and 10.5 Hz), 4.48 (1H, dd, J = 5.0 and 11.5 Hz), 4.62 (1H, t, J = 10.5 Hz), 5.17 (1H, t, J = 7.0 Hz), 6.13 (1H, s), 6.33 (1H, dd, J = 2.0 and 8.5 Hz), 6.45 (1H, d, J = 2.0 Hz), 6.94 (1H, d, J=8.5Hz), 8.30 (1H, bs), 8.63 (1H, bs), 12.48 (1H, s)
13C-NMR (acetone-d) ppm: 17.6, 21.4, 25.7, 47.2, 56.2, 71.0, 91.3, 103.5, 103.7, 107.6, 109.7, 113.5, 123.3, 131.1, 131.5, 156.8, 158.7, 161.1, 162.7, 165.8, 198.7
ESI+-MS m/z: 393.13059 [M+Na]+ (calculated value, C21H22O6Na: 393.13086)
カンゾウ抽出物に含まれるグリアスペリンB(GB)は、以下に詳細を記載する高速液体クロマトグラフ法により定量した。その結果、グリアスペリンB(GB)の含有量は、甘草抽出物1gあたり132.6μgであった。また、上記カンゾウ抽出物に含まれるグリチルリチン酸は、甘草抽出物1gあたり123mgであり、グリアスペリンB(GB)含有量/グリチルリチン酸含有量の値(比)は、0.00107であった。 The amount of gliasperin B (GB) contained in the licorice extract was quantified using the high performance liquid chromatography method described in detail below. As a result, the amount of gliasperin B (GB) contained was 132.6 μg per 1 g of licorice extract. The amount of glycyrrhizic acid contained in the licorice extract was 123 mg per 1 g of licorice extract, and the value (ratio) of gliasperin B (GB) content/glycyrrhizic acid content was 0.00107.
(グリアスペリンB(GB)の分析条件)
カラム;TSKgel ODS-100V 5μm(東ソー)4.6mm(内径)×150mm(長さ)
カラム温度;40℃、移動相;アセトニトリル:水=45:55(v/v)
流速;1ml/min
検出波長;290nm
GBの保持時間;33.2分
(Conditions for analysis of gliasperin B (GB))
Column: TSKgel ODS-100V 5 μm (Tosoh) 4.6 mm (inner diameter) × 150 mm (length)
Column temperature: 40° C., mobile phase: acetonitrile:water=45:55 (v/v)
Flow rate: 1ml/min
Detection wavelength: 290 nm
GB retention time: 33.2 min
<試験4>カンゾウ抽出物及びグリアスペリンB(GB)の脂肪分解効果の検討
マウス前駆脂肪細胞3T3-L1を、分化誘導培地(0.5mM Isobutyl-methylxanthine、1μM Dexamethasone、1μg/mL Insulin in 10%FBS/DMEM)にて2日間培養した後、1μg/mLのInsulinを含有する10%FBS/DMEM培地に交換して7日間培養した。被験物質(カンゾウ抽出物及びGB)は分化誘導開始から9日目において添加し、その後19時間培養した。カンゾウ抽出物としては、試験1で用いたカンゾウ抽出物を用い(最終濃度100μg/mL、200μg/mL、300μg/mL)、GBとしては、試験1で調製したGBを用いた(最終濃度13.5ng/mL、27ng/mL、40ng/mL)。各群3例として試験を行った。細胞中に蓄積している中性脂肪が分解して培地中に放出されるグリセロールを、ラボアッセイ(TM)トリグリセライド(富士フィルム和光純薬)にて定量した。結果を図1に示す。グリセロール量は、平均値±標準偏差で示した。図1中の各符号は以下を示す。** p<0.01 vs.無処置(Student’s t検定)。
<Test 4> Study of lipolytic effect of licorice extract and gliasperin B (GB) Mouse preadipocytes 3T3-L1 were cultured in differentiation induction medium (0.5 mM isobutyl-methylxanthine, 1 μM dexamethasone, 1 μg/mL insulin in 10% FBS/DMEM) for 2 days, and then the medium was replaced with 10% FBS/DMEM medium containing 1 μg/mL insulin and cultured for 7 days. The test substances (licorice extract and GB) were added on the 9th day after the start of differentiation induction, and the cells were then cultured for 19 hours. The licorice extract used in Test 1 was used as the licorice extract (final concentrations: 100 μg/mL, 200 μg/mL, 300 μg/mL), and the GB prepared in Test 1 was used as the GB (final concentrations: 13.5 ng/mL, 27 ng/mL, 40 ng/mL). Three subjects were tested for each group. Glycerol released into the medium upon decomposition of neutral fats accumulated in the cells was quantified using Labo Assay (TM) Triglyceride (Fujifilm Wako Pure Chemical). The results are shown in Figure 1. The amount of glycerol is shown as the mean value ± standard deviation. The symbols in Figure 1 indicate the following: ** p<0.01 vs. no treatment (Student's t-test).
図1に示すとおり、GBの濃度依存的に放出されるグリセロール量が増加しており、細胞内の脂肪分解がGBの濃度依存的に起こることが示され、GBは脂肪細胞の脂肪分解促進作用を有することがわかった。一方、カンゾウ抽出物によっても脂肪細胞からグリセロールが放出されたが、カンゾウ抽出物の濃度が300μg/mLの場合には、100μg/mLや200μg/mLの場合よりも放出されるグリセロール量が減少した。なお、カンゾウ抽出物のそれぞれの濃度100μg/mL、200μg/mL、300μg/mLは、含まれるGB量に換算するとそれぞれ13.5ng/mL、27ng/mL、40ng/mLとなる。カンゾウ抽出物でも優れた脂肪分解効果を奏するが、その濃度が高くなると脂肪分解効果が低減することから、カンゾウ抽出物中には、種々の成分が含まれており、その中には脂肪分解を阻害する成分が含まれている可能性が示唆された。このことから、より高い脂肪分解効果を得るためには、カンゾウ抽出物よりもGBの精製品を用いる方が好ましいといえる。 As shown in Figure 1, the amount of glycerol released increased in a GB concentration-dependent manner, indicating that intracellular lipolysis occurred in a GB concentration-dependent manner, and GB was found to promote lipolysis in adipocytes. On the other hand, licorice extract also released glycerol from adipocytes, but the amount of glycerol released was less when the licorice extract concentration was 300 μg/mL than when it was 100 μg/mL or 200 μg/mL. The concentrations of licorice extract, 100 μg/mL, 200 μg/mL, and 300 μg/mL, respectively, are converted to the amount of GB contained in 13.5 ng/mL, 27 ng/mL, and 40 ng/mL. Licorice extract also has an excellent lipolysis effect, but the lipolysis effect decreased as the concentration increased, suggesting that licorice extract contains various components, some of which may inhibit lipolysis. From this, it can be said that in order to obtain a higher lipolysis effect, it is preferable to use a refined GB product rather than a licorice extract.
<試験5>カンゾウ抽出物の体重、BMI及び内臓脂肪レベル(内臓脂肪蓄積の指標値)への効果
被験者(59歳の男性1名)は、試験1で用いたカンゾウ抽出物を一日あたり200mg、100日間摂取した。被験者には記録用紙を渡し、タニタの体組成計にて測定した体重、BMI及び内臓脂肪レベルをカンゾウ抽出物の摂取開始前(0日目)、摂取30日目、摂取45日目、摂取72日目、摂取83日目、摂取92日目及び最終摂取日翌日(101日目)に記録させた。摂取開始前からの体重、BMI及び内臓脂肪レベルの変化量を図2に示す。
<Test 5> Effects of licorice extract on body weight, BMI, and visceral fat level (index value of visceral fat accumulation) A subject (one male, aged 59) took 200 mg of the licorice extract used in Test 1 per day for 100 days. The subject was given a record sheet and asked to record the body weight, BMI, and visceral fat level measured with a Tanita body composition scale before the start of licorice extract intake (day 0), on the 30th day of intake, the 45th day of intake, the 72nd day of intake, the 83rd day of intake, the 92nd day of intake, and the day after the final intake (day 101). The changes in body weight, BMI, and visceral fat level from before the start of intake are shown in Figure 2.
図2に示すとおり、GBを含むカンゾウ抽出物を摂取することにより、先行して内臓脂肪レベルが低下し、追って体重及びBMIが低下することが示された。上記試験3の結果と考え合わせると、カンゾウ抽出物中のGBが脂肪細胞中の脂肪を効率よく分解し、内臓脂肪等の体脂肪を減少させたものと考えられた。 As shown in Figure 2, it was shown that the intake of licorice extract containing GB first reduced visceral fat levels, followed by reductions in body weight and BMI. Considering this together with the results of Test 3 above, it is believed that the GB in the licorice extract efficiently decomposed fat in adipocytes, reducing body fat such as visceral fat.
<試験6>
5週齢の雄性C57BL/6Jマウスを6匹ずつに分け、MF(オリエンタル酵母)、高脂肪高糖質食粉末飼料(以下HFSと略す、D12079BM、Research Diet)又は甘草抽出物を混合したHFSを与えて8週間飼育した。摂取8週間後に内臓脂肪重量(副睾丸周囲脂肪重量、腎周囲脂肪重量及び腸間膜脂肪重量の合算)を測定した。なお、甘草抽出物(試験例1~5)として、甘草の根及びストロンの部分を粉砕して得た粉末から、50%(v/v)エタノール抽出工程を経て得られた液を、エバポレーター及び凍結乾燥にて乾固した後粉砕し、甘草抽出物を得た。これらを、それぞれ飼料に下記表8に示す量(%)混合して与えた。ヒト換算のグリアスペリン投与量(μg/日)と、マウスの内臓脂肪重抑制率(%)を表8に合わせて示す。
<Test 6>
Five-week-old male C57BL/6J mice were divided into groups of six and fed MF (Oriental Yeast), high-fat, high-carbohydrate powdered feed (hereinafter abbreviated as HFS, D12079BM, Research Diet), or HFS mixed with licorice extract for 8 weeks. After 8 weeks of ingestion, the visceral fat weight (the sum of the epididymal fat weight, perirenal fat weight, and mesenteric fat weight) was measured. For the licorice extract (Test Examples 1 to 5), the root and stolon of licorice were crushed into powder, and the solution obtained through a 50% (v/v) ethanol extraction process was dried and solidified using an evaporator and freeze-dried, and then crushed to obtain a licorice extract. These were mixed with the feed in the amounts (%) shown in Table 8 below and fed to the mice. The human-equivalent dose of gliasperin (μg/day) and the visceral fat weight suppression rate (%) of the mice are also shown in Table 8.
(結論)
上記表に示すとおり、マウスに対するグリアスペリンの投与量(摂取量)がヒト換算で9.7μg/日~47.6μg/日の条件で、顕著な内臓脂肪重量抑制効果が見られた。
(Conclusion)
As shown in the above table, a significant visceral fat weight suppressing effect was observed when the administration (ingestion) amount of gliasperin to mice was 9.7 μg/day to 47.6 μg/day calculated in human terms.
本発明の内用組成物は、グリアスペリンBを含有することにより、優れた脂肪分解促進作用を有すると共に、顕著な体重減少効果を奏する。本発明の内用組成物を用いることで、脂肪細胞中の脂肪を効率よく分解し、皮下脂肪、内臓脂肪等の体脂肪を減少させることができ、結果的に顕著な体重減少効果、BMIの改善効果を得ることができる。また、インスリン抵抗性改善、これステロール値低減効果も得られる。よって、本発明の内用組成物によると、肥満や生活習慣病を長期的に予防し、また改善することができ、健康の維持・増進を実現することができる。さらに、植物抽出物におけるグリアスペリンBの含有量が、植物抽出物の脂肪分解、体重減少等に対する有効性の指標になることから、有効性の高い植物抽出物を効率的に選定することが可能となる。 The internal composition of the present invention contains gliasperin B, which has an excellent lipolysis promoting effect and a significant weight loss effect. By using the internal composition of the present invention, fat in adipocytes can be efficiently broken down and body fat such as subcutaneous fat and visceral fat can be reduced, resulting in a significant weight loss effect and an improvement in BMI. In addition, the composition improves insulin resistance and reduces sterol levels. Therefore, the internal composition of the present invention can prevent and improve obesity and lifestyle-related diseases in the long term, thereby maintaining and improving health. Furthermore, the content of gliasperin B in a plant extract is an indicator of the effectiveness of the plant extract for lipolysis, weight loss, etc., making it possible to efficiently select a highly effective plant extract.
Claims (10)
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2023132207 | 2023-08-15 | ||
| JP2023-132207 | 2023-08-15 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2025037604A1 true WO2025037604A1 (en) | 2025-02-20 |
Family
ID=94632295
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2024/028767 Pending WO2025037604A1 (en) | 2023-08-15 | 2024-08-09 | Internal composition for lipolysis and/or weight loss, method for determining effectiveness, and index for quality of plant extract |
Country Status (2)
| Country | Link |
|---|---|
| TW (1) | TW202515530A (en) |
| WO (1) | WO2025037604A1 (en) |
Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2002047699A1 (en) * | 2000-12-12 | 2002-06-20 | Kaneka Corporation | Compositions for preventing or ameliorating multiple risk factor syndromes |
| WO2003037316A1 (en) * | 2001-10-11 | 2003-05-08 | Kaneka Corporation | Peroxisome proliferator activated receptor ligands and process for producing the same |
| JP2003252784A (en) * | 2002-02-27 | 2003-09-10 | Kanegafuchi Chem Ind Co Ltd | α-glucosidase inhibitor |
| WO2017094892A1 (en) * | 2015-12-03 | 2017-06-08 | エムジーファーマ株式会社 | Healthy obesity maintenance agent |
| JP2023120103A (en) * | 2022-02-17 | 2023-08-29 | エムジーファーマ株式会社 | lipolysis accelerator |
-
2024
- 2024-08-09 WO PCT/JP2024/028767 patent/WO2025037604A1/en active Pending
- 2024-08-13 TW TW113130335A patent/TW202515530A/en unknown
Patent Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2002047699A1 (en) * | 2000-12-12 | 2002-06-20 | Kaneka Corporation | Compositions for preventing or ameliorating multiple risk factor syndromes |
| WO2003037316A1 (en) * | 2001-10-11 | 2003-05-08 | Kaneka Corporation | Peroxisome proliferator activated receptor ligands and process for producing the same |
| JP2003252784A (en) * | 2002-02-27 | 2003-09-10 | Kanegafuchi Chem Ind Co Ltd | α-glucosidase inhibitor |
| WO2017094892A1 (en) * | 2015-12-03 | 2017-06-08 | エムジーファーマ株式会社 | Healthy obesity maintenance agent |
| JP2023120103A (en) * | 2022-02-17 | 2023-08-29 | エムジーファーマ株式会社 | lipolysis accelerator |
Non-Patent Citations (1)
| Title |
|---|
| KURODA MINPEI, YOSHIHIRO MIMAKI, YUTAKA SASHIDA, TATSUMASA MAE, TOZO NISHIYAMA, MISUZU TSUKAGAWA, EISAKU KONISHI, KAKU NAKAGAWA, M: "50(P-15) Preventive and Ameliorative Effects of Glycyrrhizae Radix on Type-2 Diabetes and Abdominal Obesity, and Identification of the Active Ingredients Using a PPAR-γ Ligand-binding Screening Method", SYMPOSIUM ON THE CHEMISTRY OF NATURAL PRODUCTS, SYMPOSIUM PAPERS, 1 January 2003 (2003-01-01), pages 293 - 298, XP093279336, Retrieved from the Internet <URL:https://www.jstage.jst.go.jp/article/tennenyuki/45/0/45_293/_article/-char/en> DOI: 10.24496/tennenyuki.45.0_293 * |
Also Published As
| Publication number | Publication date |
|---|---|
| TW202515530A (en) | 2025-04-16 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| Zemestani et al. | Chamomile tea improves glycemic indices and antioxidants status in patients with type 2 diabetes mellitus | |
| Hitoe et al. | Seaweed fucoxanthin supplementation improves obesity parameters in mild obese Japanese subjects | |
| TWI329516B (en) | Composition for preventing or ameliorating multiple risk factor syndromes and visceral fat-type obesity | |
| Vieira Senger et al. | Effect of green tea (Camellia sinensis) consumption on the components of metabolic syndrome in elderly | |
| Yoshino et al. | Polymethoxyflavone purified from Kaempferia parviflora reduces visceral fat in Japanese overweight individuals: a randomised, double-blind, placebo-controlled study | |
| Mišan et al. | Buckwheat–enriched instant porridge improves lipid profile and reduces inflammation in participants with mild to moderate hypercholesterolemia | |
| Ferguson et al. | Effect of a polyphenol-rich dietary supplement containing Pinus massoniana bark extract on blood pressure in healthy adults: A parallel, randomized placebo-controlled trial | |
| RU2327463C2 (en) | Medicinal agent and food substance or beverage for improved hyperglycemia | |
| Jung et al. | The effect of maté extract on body weight and fat reduction in obese women: A randomized placebo-controlled clinical trial | |
| JP6977053B2 (en) | A composition for the prevention or treatment of obesity or a metabolic syndrome caused by obesity containing formic acid or a pharmaceutically acceptable salt thereof as an active ingredient. | |
| Masuzaki et al. | Safety and efficacy of nanoparticulated brown rice germ extract on reduction of body fat mass and improvement of fuel metabolism in both pre-obese and mild obese subjects without excess of visceral fat accumulation. | |
| WO2025037604A1 (en) | Internal composition for lipolysis and/or weight loss, method for determining effectiveness, and index for quality of plant extract | |
| Kim et al. | Sunflower seed extract supplementation reduces body fat in adults with obesity: A double-blind, randomized, placebo-controlled trial | |
| Nishimura et al. | Effects of daily intake of Harudori-kombu: A randomized, double-blind, placebo-controlled, parallel-group study | |
| JP7445689B2 (en) | lipolysis accelerator | |
| Hanoman et al. | Bitter melon in combination with diet modification and regular exercise can prevent and treat obesity and hypertension cost-effectively | |
| Hosseini et al. | Impact of short–term intake of Cinnamon on serum glucose and lipid profile in patients with type 2 diabetes mellitus | |
| Khan et al. | To study the effect of sprouted fenugreek seeds as nutraceutical as an add-on therapy in patients of diabetes mellitus, obesity and metabolic syndrome | |
| Tominaga et al. | Effect of licorice flavonoid oil on visceral fat in obese subjects in the United States: a randomized, double-blind, placebo-controlled study | |
| JP6990539B2 (en) | Composition for prevention / improvement of decrease in vascular flexibility and prevention / improvement of arteriosclerosis | |
| Georgoulis et al. | The effectiveness of a natural aqueous extract of Chios mastic in the management of irritable bowel syndrome: the MASTIQUA randomized controlled clinical trial | |
| Gomes et al. | Dietary calcium from dairy, body composition and glycaemic control in patients with type 2 diabetes pursuing an energy restricted diet: A parallel group randomised clinical trial | |
| Fahey | Health benefits of apple cider vinegar and other common vinegars: A review | |
| Kim et al. | A 8-week, randomized, double-blind, placebo-controlled human trial to evaluate the efficacy and safety of Saccharum officinarum wax alcohols (Policosanol) on improvement of blood cholesterol | |
| CN100502922C (en) | A capsule for regulating blood pressure and regulating blood sugar |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 24854193 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2025540689 Country of ref document: JP Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2025540689 Country of ref document: JP |