WO2025036848A1 - Anti-muc16 antibodies and uses thereof - Google Patents
Anti-muc16 antibodies and uses thereof Download PDFInfo
- Publication number
- WO2025036848A1 WO2025036848A1 PCT/EP2024/072621 EP2024072621W WO2025036848A1 WO 2025036848 A1 WO2025036848 A1 WO 2025036848A1 EP 2024072621 W EP2024072621 W EP 2024072621W WO 2025036848 A1 WO2025036848 A1 WO 2025036848A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- muc16
- antibody
- binding
- binding molecule
- cancer
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
- C07K16/18—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans
- C07K16/28—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants
- C07K16/30—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants from tumour cells
- C07K16/3076—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants from tumour cells against structure-related tumour-associated moieties
- C07K16/3092—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants from tumour cells against structure-related tumour-associated moieties against tumour-associated mucins
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
- C07K16/18—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans
- C07K16/28—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants
- C07K16/30—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants from tumour cells
- C07K16/3069—Reproductive system, e.g. ovaria, uterus, testes, prostate
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K2039/505—Medicinal preparations containing antigens or antibodies comprising antibodies
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/50—Immunoglobulins specific features characterized by immunoglobulin fragments
- C07K2317/56—Immunoglobulins specific features characterized by immunoglobulin fragments variable (Fv) region, i.e. VH and/or VL
- C07K2317/565—Complementarity determining region [CDR]
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/60—Immunoglobulins specific features characterized by non-natural combinations of immunoglobulin fragments
- C07K2317/62—Immunoglobulins specific features characterized by non-natural combinations of immunoglobulin fragments comprising only variable region components
- C07K2317/622—Single chain antibody (scFv)
Definitions
- This application generally relates to antibodies. More specifically, the application relates to monoclonal antibodies against MUC16, especially VHH-comprising antibodies, a method of preparing the same, and the use of the antibodies.
- Mucin 16 (MUC16, previously known as carcinoma antigen 125, CA125) was first identified by Bast et al. in 1981, and its cDNA sequence was later found corresponded to MUC16, a mucin protein [1-2], MUC16 is a heavily glycosylated single pass transmembrane protein with a molecular weight around 3000 - 5000 KD [2], It is the largest mucin protein, consisting of multiple domains including an extracellular N-terminal domain, a large tandem repeat domain which is interspersed with sea urchin sperm, enterokinase and agrin (SEA) domain, and a C-terminal domain that comprises the transmembrane region and a short cytoplasmic tail [3], MUC16 has 56 SEA domains, where the penultimate SEA (55 th ) domain has a conserved cleavage site [4], MUC16 could shed from the cell surface and release into the bloodstream to become soluble MUC16 (CA125) through
- MUC16 is only expressed at low levels in a few tissues including the respiratory tract and the female reproductive tract, particularly in glands and epithelial cells [5], The expression level of MUC16 is significantly higher in a spectrum of human cancers, including ovarian cancer, endometrial cancer, pancreatic cancer, than that of normal tissues.
- the shedding domain termed CAI 25 is a poor prognostic and diagnostic serum marker for ovarian cancer.
- CAI 25 is the most widely used ovarian tumor marker and often considered as the “gold standard’.
- Abnormal CAI 25 level was observed in 99 % of serous cancer patients rated from I to IV in the FIGO (International Federation of Gynecologists and Obstetricians) stages.
- Serum CA125 levels could increase up to 10 folds and exceed 2000 U/mL in many serous ovarian cancer patients at FIGO stage IV compared to stage I [8].
- elevated expression of CAI 25 is strongly correlated with poorer prognosis in multiple cancers [9]
- the limited expression of MUC16 on normal human tissues and its high expression in many common cancers makes it an attractive target for cancer therapy.
- MUC 16 -targeting therapeutic antibodies including oregovomab and abagovomab have been tested in clinical trials, only limited efficacy has been achieved in cancer patients [10, 11].
- a potential shortcoming of the several described antibody-based therapeutics is that they target the membrane distal region of MUC 16, and consequently, due to the high levels of circulating CAI 25 in cancer patients and the CAI 25 antigen sink effect, the target cell bound antibody is significantly reduced and the tumor killing effect would be therefore compromised [4]. Avoid binding to the soluble CAI 25 in the blood circulation might be critical in the development of therapeutic antibodies that target MUC 16-positive cancers.
- the present disclosure is directed to compounds, methods, compositions and articles of manufacture that provide MUC16-binding molecules with improved efficacy.
- the benefits provided by the present disclosure are broadly applicable in the field of antibody therapeutics and diagnostics and may be used in conjunction with other therapeutics such as antibodies that react with a variety of targets.
- MUC16-binding molecules such as monoclonal antibodies, that can specifically bind to human MUC 16 and are cross-reactive with cynomolgus monkey MUC 16.
- MUC16-binding molecules provide certain advantages compared to the agents, compositions and/or methods currently used and/or known in the art. These advantages include improved therapeutic and pharmacological properties, increased specificity, reduced immunogenicity, and other advantageous properties.
- MUC16-binding molecules such as monoclonal antibodies, against MUC 16 which can be used to treat MUC16-overexpressing tumor have been developed.
- the present disclosure provides MUC16-binding molecules, nucleic acid molecules encoding the same, expression vectors and host cells used for the expression of MUC16-binding molecules, and methods for using MUC16-binding molecules.
- MUC16-binding molecules of the present disclosure provide potent agents for the treatment of multiple cancers via modulating human immune function.
- the present disclosure provides a MUC16-binding molecule comprising at least one immunoglobulin single variable domain (e.g. VHH domain) that specifically binds to MUC16, such as human MUC16 and cyno MUC16.
- the single variable domain comprises CDR1, CDR2 and CDR3, and wherein: the CDR1 comprises an amino acid sequence as set forth in SEQ ID NO: 1; the CDR2 comprises an amino acid sequence as set forth in SEQ ID NO: 2; and the CDR3 comprises an amino acid sequence as set forth in SEQ ID NO: 3.
- the single variable domain as disclosed herein comprises: a CDR1 as set forth in SEQ ID NO: 1 ; a CDR2 as set forth in SEQ ID NO: 2; and a CDR3 as set forth in SEQ ID NO: 3.
- the single variable domain as disclosed herein comprises:
- (C) an amino acid sequence with addition, deletion and/or substitution of one or more (e.g. 1, 2 or 3) amino acids compared with the amino acid sequence as set forth in any one of SEQ ID NOs: 4-5 yet the specific binding affinity to MUC 16 is maintained (e.g., substantially maintained, for example, at least 50%, at least 60%, at least 70%, at least 80%, at least 90%, at least 95%).
- MUC 16-binding molecules as disclosed herein comprise one or more substitutions, additions, and/or deletions of amino acids in the framework regions, e.g. FRW1, FRW2, FRW3, and/or FRW4 of a single variable domain (e.g., VHH).
- FRW1 at the N terminal and/or FRW4 at the C terminal of the single variable domain is truncated, e.g. truncated by no more than 5, 4, 3, 2, or 1 amino acid(s).
- a single variable domain (e.g., VHH) comprises an amino acid sequence as set forth in any one of SEQ ID NOs: 4-5.
- a MUC 16-binding molecule as disclosed herein further comprises one or more human IgG constant domains, such as one or more human IgGl, IgG2, IgG3 or IgG4 constant domains.
- the IgG constant domain is a human IgGl constant domain or a variant thereof.
- a MUC 16-binding molecule comprises a variant of one or more human IgGl constant domains, e.g. an IgGl Fc with L234A/L235A substitutions, according to EU numbering.
- a MUC 16-binding molecule as disclosed herein has one or more of the following properties:
- a MUC 16-binding molecule as disclosed herein is a chimeric antibody, a humanized antibody or a folly human antibody. In some embodiments, the MUC 16-binding molecule is a dimer. In some embodiments, a MUC16-binding molecule as disclosed herein comprises an amino acid sequence as set forth in any one of SEQ ID NOs: 6-7.
- the present disclosure provides a nucleic acid molecule comprising a nucleic acid sequence encoding a MUC16-binding molecule as disclosed herein, such as a MUC16-binding molecule comprising a single variable domain (e.g., VHH).
- a MUC16-binding molecule comprising a single variable domain (e.g., VHH).
- the present disclosure provides a vector comprising a nucleic acid molecule as disclosed herein.
- the present disclosure provides a host cell comprising an expression vector or a nucleic acid molecule as disclosed herein.
- the present disclosure provides a pharmaceutical composition comprising a MUC16-binding molecule as disclosed herein and a pharmaceutically acceptable carrier.
- the present disclosure provides a method for preparing a MUC16- binding molecule which comprises expressing the MUC16-binding molecule in a host cell as disclosed herein and isolating the MUC16-binding molecule from the host cell.
- the present disclosure provides a method of modulating a MUC16- related immune response in a subject, comprising administering a MUC16-binding molecule as disclosed herein to the subject such that the MUC16-related immune response in the subject is modulated.
- the present disclosure provides a method for treating or preventing a MUC16 positive or MUC16 overexpressed cancer in a subject comprising administering an effective amount of a MUC16-binding molecule or a pharmaceutical composition as disclosed herein to the subject.
- the cancer includes but is not limited to, ovary cancer, lung cancer, pancreas cancer, breast cancer, uterine cancer, fallopian tube cancer, primary peritoneum cancer or cancer of any other tissue that expresses MUC16.
- the present disclosure provides use of a MUC16-binding molecule as disclosed herein in the manufacture of a medicament for diagnosing, treating or preventing a MUC16 positive cancer.
- the present disclosure provides a MUC16-binding molecule as disclosed herein for use in treating or preventing a MUC16 positive cancer.
- the present disclosure provides a MUC16-binding molecule as disclosed herein for use in diagnostic methods for identifying the presence of MUC16 in tissue and/or plasma samples.
- kits or devices and associated methods that employ a MUC16-binding molecule as disclosed herein, or pharmaceutical compositions as disclosed herein.
- Figure 1 shows purity analysis of W306106-Pl lR2-lB8-uIgG1: (A) SDS-PAGE analysis; (B) SEC-HPLC chromatogram.
- Figure 2 shows DSF profile of W306106-Pl lR2-lB8-uIgG1.
- Figures 3-4 show purity analysis of two batches of W306106-P1 lR2-lB8-z6-uIgG1 : (A) SDS-PAGE analysis; (B) SEC-HPLC chromatogram.
- Figure 5 shows SEC-HPLC profile of W306106-P1 !R2-lB8-z6-uIgG1 after 5 Freeze/Thaw cycles.
- Figure 6 shows SEC-HPLC profiles of W306106-P1 !R2-lB8-z6-uIgG1 after 14 days in 40 °C.
- Figure 7 shows DSF profile of W306106-P1 lR2-lB8-z6-uIgG1.
- Figure 8 shows DLS-kD profiles of W306106-P1 lR2-lB8-z6-uIgG1.
- Figures 9-10 show purity analysis of W306106-P11R2-1B8-Z6: (A) SDS-PAGE analysis; (B) SEC-HPLC chromatogram. Figure 10 is a concentrated form of the protein of Figure 9.
- Figure 11 shows DSF profiles of W306106-Pl lR2-lB8-z6.
- Figure 12 shows DLS-Radius size distribution profiles of W306106-P11R2-1B8-Z6.
- Figure 13 shows HIC-HPLC profiles of W306106-P11R2-1B8-Z6.
- Figure 14 shows DLS-kD profiles of W306106-P11R2-1B8-Z6.
- Figure 15 shows SEC-HPLC profiles of W306106-Pl lR2-lB8-z6 after 5 Freeze/Thaw cycles.
- Figure 16 shows SEC-HPLC profiles of W306106-P11R2-1B8-Z6 after 14 days in 40 °C.
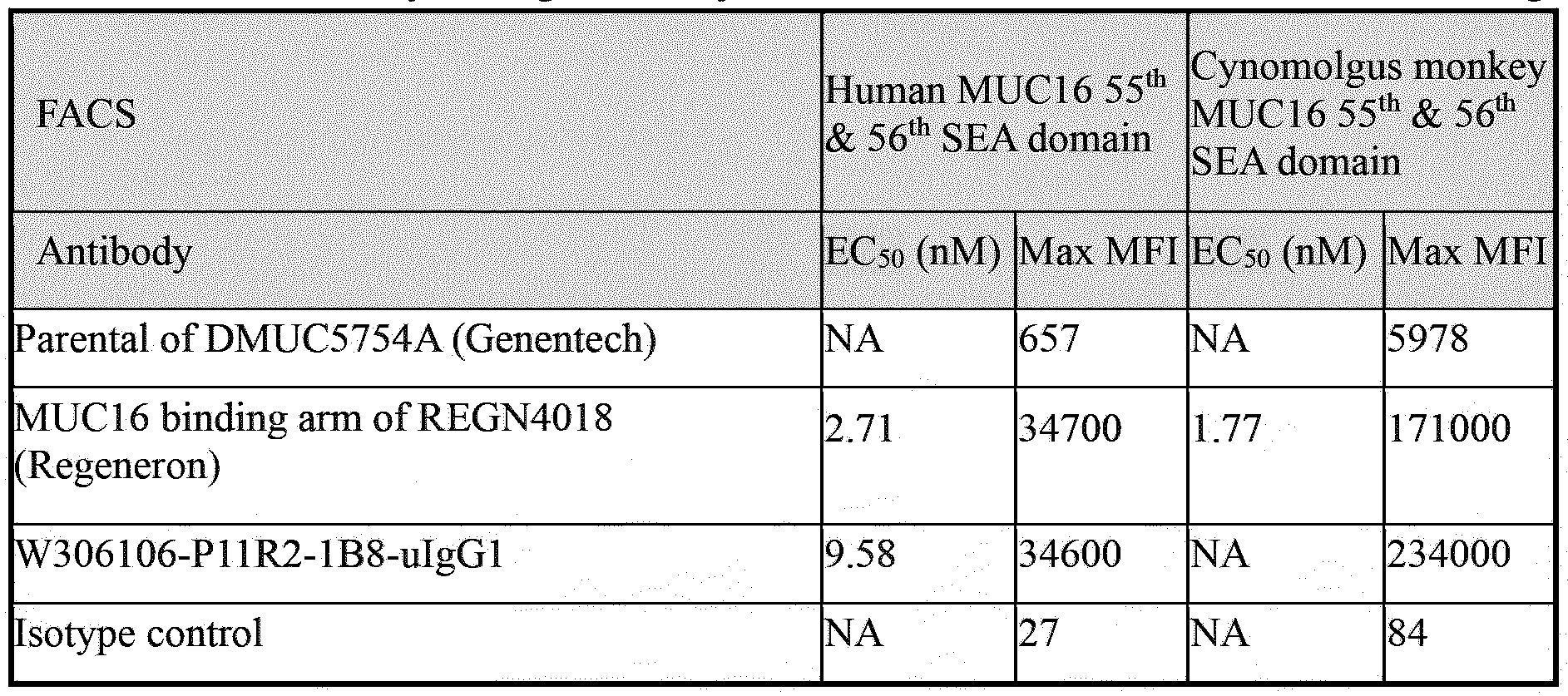
- Figure 17A-17B shows human (A) and cynomolgus monkey (B) MUC16 55 th & 56 th SEA domain FACS binding of W306106-P1 lR2-lB8-uIgG1.
- Figure 18A-18B shows human (A) and cynomolgus monkey (B) MUC16 55 th & 56 th SEA domain FACS binding of W306106-P1 lR2-lB8-z6-uIgG1.
- Figure 19A-19B shows human (A) and cynomolgus monkey (B) MUC16 55 th & 56 th SEA domain FACS binding of W306106-P11R2-1B8-Z6.
- Figure 20 shows human MUC16 binding of W306106-P1 lR2-lB8-uIgG1 on OVCAR-3 (MUC16 high ).
- Figure 21 A-2 ID shows human MUC 16 binding of W306106-P1 lR2-lB8-z6-uIgG1 on four human tumor cell lines.
- Figure 22A-22B shows human MUC16 binding of W306106-P11R2-1B8-Z6 on two human tumor cell lines.
- Figure 23A-23B shows ELISA binding of W306106-P1 lR2-lB8-uIgG1 to human MUC16 extracellular domain proteins.
- Figure 24A-24C shows ELISA binding of W306106-P1 !R2-lB8-z6-uIgG1 to human MUC16 extracellular domain proteins.
- Figure 25A-25C shows ELISA binding of W306106-P11R2-1B8-Z6 to human MUC16 extracellular domain proteins.
- Figure 26A-26B shows ELISA binding of W306106-P1 !R2-lB8-z6-uIgG1 to cynomolgus monkey (A) and mouse (B) MUC16 extracellular domain proteins.
- Figure 27A-27B shows ELISA binding of W306106-P 11R2- 1B8-Z6 to cynomolgus monkey (A) and mouse (B) MUC16 extracellular domain proteins.
- Figure 28 shows binding of W306106-P1 lR2-lB8-uIgG1 to W3XX106- hPro 1.ECD.AVI.His-P2.
- Figure 29 shows binding of W306106-P1 lR2-lB8-z6-uIgG1 to W3XX106- hProl.ECD.AVI.His-P2.
- Figure 30 shows the result of W306106-P1 lR2-lB8-uIgG1 in HCS internalization assay on OVCAR-3 cells.
- antibody e.g. anti-MUC16 antibody
- antigen-binding molecule e.g. MUC16-binding molecule
- antibody covers, but is not limited to, humanized antibodies, fully human antibodies, chimeric antibodies and single-domain antibodies (sdAbs, comprising just one chain, which is typically similar to a heavy chain), as well as fragments of any of the foregoing as long as they exhibit the desired antigen-binding activity, including, for example, an antibody comprising at least one VHH domain.
- a conventional antibody comprises a heavy chain(s) and a light chain(s).
- Heavy chains may be classified into p, 5, y, a and E, which define isotypes of an antibody as IgM, IgD, IgG, IgA and IgE, respectively.
- a heavy chain can comprise a heavy chain variable region (VH) and a heavy chain constant region (CH).
- a heavy chain can comprise one or more constant regions, for example, 3 constant regions (CHI, CH2 and CH3).
- a light chain can comprise a light chain variable region (VL) and a light chain constant region (CL).
- a VH and a VL region can further be divided into hypervariable regions (called complementary determining regions (CDRs)), which are interspaced by relatively conservative regions (called framework regions (FRW)).
- CDRs complementary determining regions
- FRW framework regions
- a VH and a VL can comprise 3 CDRs (Complementarity determining regions) and 4 FRs (Framework regions) in the following order: FRW1, CDR1, FRW2, CDR2, FRW3, CDR3, FRW4 from N-terminal to C-terminal.
- Antibodies can be of different antibody isotypes, for example, IgG (e.g., IgGl, IgG2, IgG3 or IgG4 subtype), IgAl, IgA2, IgD, IgE or IgM antibody.
- MUC16 or “MUC16 polypeptide” refers to mucin 16, which is a single transmembrane domain highly glycosylated integral membrane glycoprotein that is highly expressed in ovarian cancer.
- MUC16 is an extremely large glycoprotein (-22,152 amino acids) with approximately 12,000 amino acids of the heavily O-glycosylated N-terminal region, a tandem repeat region comprising approximately 60 repeats of 156 amino acids each, a transmembrane domain and a cytoplasmic tail of 32 amino acids.
- MUC16 harbors 56 SEA domains and each SEA domain constitutes a major portion (amino acids 1-128) of each tandem repeat.
- GenBankTM accession number NP 078966.2 provides an exemplary human MUC16 amino acid sequence.
- MUC16 is predicted to undergo cleavage in the penultimate and/or last SEA domain (i.e. the 55 th and 56 th SEA domain, which are closest to the transmembrane domain among the SEA domains) and phosphorylation event(s) in the cytoplasmic tail domain (CTD) is (are) believed to be critical determinants of its cleavage (Srustidhar Das et al., Understanding the Unique Attributes of MUC16 (CA125): Potential Implications in Targeted Therapy, Cancer Research, 2015).
- Fc region is used to define a C -terminal region of an immunoglobulin heavy chain, including, for example, native sequence Fc regions, recombinant Fc regions, and variant Fc regions. Although the boundaries of the Fc region of an immunoglobulin heavy chain might vary, the human IgG heavy chain Fc region is often defined to stretch from an amino acid residue at position Cys226 (according to the EU numbering system), or from Pro230 (according to the EU numbering system), to the carboxyl-terminus thereof.
- the C-terminal lysine (residue 447 according to the EU numbering system) of the Fc region may be removed, for example, during production or purification of the antibody, or by recombinantly engineering the nucleic acid encoding a heavy chain of the antibody.
- a “functional Fc region” possesses an “effector function” of a native sequence Fc region.
- effector functions include Clq binding; complement dependent cytotoxicity (CDC); Fc receptor binding; antibody-dependent cell-mediated cytotoxicity (ADCC); phagocytosis; down regulation of cell surface receptors (e.g., B cell receptor; BCR), etc.
- Such effector functions generally require the Fc region to be combined with a binding region or binding domain (e.g., an antibody variable region or domain, including a VHH domain) and can be assessed using various assays as disclosed.
- a “native sequence Fc region” comprises an amino acid sequence identical to the amino acid sequence of an Fc region found in nature, and not manipulated, modified, and/or changed (e.g., isolated, purified, selected, including or combining with other sequences such as variable region sequences) by a human.
- Native sequence human Fc regions include a native sequence human IgGl Fc region (non-A and A allotypes); native sequence human IgG2 Fc region; native sequence human IgG3 Fc region; and native sequence human IgG4 Fc region as well as naturally occurring variants thereof.
- a “variant Fc region” comprises an amino acid sequence which differs from that of a native sequence Fc region by virtue of at least one amino acid modification, (e.g., substituting, addition, or deletion) preferably one or more amino acid substitution(s).
- the variant Fc region has at least one amino acid substitution compared to a native sequence Fc region or to the Fc region of a parent polypeptide, for example, from about one to about ten amino acid substitutions, and preferably from about one to about five amino acid substitutions in a native sequence Fc region or in the Fc region of the parent polypeptide.
- a variant Fc region can possess at least about 80% homology with a native sequence Fc region and/or with an Fc region of a parent polypeptide, or at least about 90% homology therewith, for example, at least about 95% homology therewith.
- the variant Fc region herein described herein may have a loss of effector function (e.g., silent Fc).
- Antibodies described herein include, but are not limited to, synthetic antibodies, monoclonal antibodies, recombinantly produced antibodies, multispecific antibodies (e.g., including bispecific antibodies), human antibodies, humanized antibodies, chimeric antibodies, intrabodies, singlechain Fvs (scFv) (e.g., including monospecific, bispecific, etc.), camelized antibodies, Fab fragments, F(ab’) fragments, disulfide-linked Fvs (sdFv), anti-idiotypic (anti-Id) antibodies, and epitope-binding fragments of any of the above.
- synthetic antibodies e.g., monoclonal antibodies, recombinantly produced antibodies, multispecific antibodies (e.g., including bispecific antibodies), human antibodies, humanized antibodies, chimeric antibodies, intrabodies, singlechain Fvs (scFv) (e.g., including monospecific, bispecific, etc.), camelized antibodies, Fab fragments, F(ab’) fragments, disul
- VHH domain e.g. variable domain of a heavy chain antibody
- a VHH domain represents the smallest known antigen-binding unit generated by adaptive immune responses (Koch-Nolte F. et al., FASEB J. Nov; 21 (13):3490-8. Epub 2007 Jun 15 (2007)).
- a VHH domain may be a human domain, but also includes a single domain from other species such as rodent, nurse shark and Camelid VHH domains.
- Camelid VHH are immunoglobulin single variable domain polypeptides that are derived from species including camel, llama, alpaca, dromedary, and guanaco, which produce heavy chain antibodies naturally devoid of light chains.
- Such VHH domains may be humanized according to standard techniques available in the art and are considered as “single domain antibodies”.
- VHH includes camelid VHH domains and humanized VHH domains.
- humanized antibody is intended to refer to antibodies in which CDR sequences derived from the germline of another mammalian species, such as a mouse, llama or alpaca, have been grafted onto human framework sequences. Additional framework region modifications may be made within the human framework sequences.
- Ka is intended to refer to the association rate of a particular antibody-antigen interaction
- Kd is intended to refer to the dissociation rate of a particular antibody-antigen interaction
- Kd values for antibodies can be determined using methods well established in the art.
- KD is intended to refer to the dissociation constant of a particular antibody-antigen interaction, which is obtained from the ratio of Kd to Ka (e.g., Kd/Ka) and may be expressed as a molar concentration (M).
- a preferred method for determining the Ka, Kd and KD of an antibody is by using surface plasmon resonance, preferably using a biosensor system such as a Biacore® system.
- binding or “specifically binds” as used herein refers to a non-random binding reaction between two molecules, such as for example between an antibody and an antigen.
- high affinity refers to a MUC16 binding molecule such as an antibody having a KD of 1 x 10 -9 M or less, more preferably 5 x IO' 10 M or less, even more preferably IxlO' 10 or less, even more preferably 5 x 10 -11 or less for a target antigen.
- EC 50 which is also termed as “half maximal effective concentration” refers to the concentration of a drug, antibody or toxicant which induces a response halfway between the baseline and maximum after a specified exposure time. In the context of the present disclosure,EC 50 is expressed in the unit of “nM”.
- epitope refers to a portion of an antigen that an immunoglobulin or antibody specifically binds to. “Epitope” is also known as “antigenic determinant”. Epitope or antigenic determinant generally comprises chemically active surface groups of a molecule such as amino acids, carbohydrates or sugar side chains, and generally has a specific three-dimensional structure and a specific charge characteristic. For example, an epitope generally comprises at least 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14 or 15 consecutive or non-consecutive amino acids in a unique steric conformation, which may be “linear” or “conformational”. See, for example, Epitope Mapping Protocols in Methods in Molecular Biology, Vol. 66, G. E.
- isolated antibody is intended to refer to an antibody that is substantially free of other antibodies having different antigenic specificities (e.g., an isolated antibody that specifically binds a MUC16 protein is substantially free of antibodies that specifically bind antigens other than MUC 16 proteins).
- An isolated antibody that specifically binds a human MUC 16 protein may, however, have cross-reactivity to other antigens, such as MUC 16 proteins from other species.
- an isolated antibody can be substantially free of other cellular material and/or chemicals.
- vector refers to a nucleic acid vehicle which can have a polynucleotide inserted therein.
- the vector allows for the expression of the protein encoded by the polynucleotide inserted therein, the vector is called an expression vector.
- the vector can have carried genetic material elements expressed in a host cell by transformation, transduction, or transfection into the host cell.
- Vectors are well known by a person skilled in the art, including, but not limited to plasmids, phages, cosmids, artificial chromosome such as yeast artificial chromosome (YAC), bacterial artificial chromosome (BAC) or Pl -derived artificial chromosome (PAC); phage such as X phage or Ml 3 phage and animal virus.
- the animal viruses that can be used as vectors include, but are not limited to, retrovirus (including lentivirus), adenovirus, adeno-associated virus, herpes virus (such as herpes simplex virus), pox virus, baculovirus, papillomavirus, papova virus (such as SV40).
- a vector may comprise multiple elements for controlling expression, including, but not limited to, a promoter sequence, a transcription initiation sequence, an enhancer sequence, a selection element and a reporter gene.
- a vector may comprise an origin of replication.
- host cell refers to a cell into which a vector can be introduced, including, but not limited to, a prokaryotic cell such as E. coli or Bacillus subtilis, a fungal cell such as yeast cell or Aspergillus, an insect cell such as S2 Drosophila cell or Sf9, and an animal cell such as fibroblast, CHO cell, COS cell, NSO cell, HeLa cell, BHK cell, HEK 293 cell or human cell.
- a prokaryotic cell such as E. coli or Bacillus subtilis
- a fungal cell such as yeast cell or Aspergillus
- an insect cell such as S2 Drosophila cell or Sf9
- animal cell such as fibroblast, CHO cell, COS cell, NSO cell, HeLa cell, BHK cell, HEK 293 cell or human cell.
- identity refers to a relationship between the sequences of two or more polypeptide molecules or two or more nucleic acid molecules, as determined by aligning and comparing the sequences. “Percent identity” means the percent of identical residues between the amino acids or nucleotides in the compared molecules and is calculated based on the size of the smallest of the molecules being compared. For these calculations, gaps in alignments (if any) are preferably addressed by a particular mathematical model or computer program (e.g., an “algorithm”). Methods that can be used to calculate the identity of the aligned nucleic acids or polypeptides include those described in Computational Molecular Biology, (Lesk, A.
- immunogenicity refers to an ability to stimulate formation of specific antibodies or sensitized lymphocytes in organisms. It not only refers to a property of an antigen to stimulate a specific immunocyte to activate, proliferate and differentiate so as to finally generate immunologic effector substance such as antibody and sensitized lymphocyte, but also refers to a specific immune response that antibody or sensitized T lymphocyte can be formed in an immune system of an organism after stimulating the organism with an antigen. Immunogenicity is an important property of an antigen. Whether an antigen can successfully induce the generation of an immune response in a host depends on several factors, including properties of an antigen, reactivity of a host, and immunization means.
- transfection refers to a process by which nucleic acids are introduced into eukaryotic cells, particularly mammalian cells. Protocols and techniques for transfection include but not limited to lipid transfection and chemical and physical methods such as electroporation. A number of transfection techniques are well known in the art and are disclosed herein. See, e.g., Graham et al., 1973, Virology 52:456; Sambrook et al., 2001, Molecular Cloning: A Laboratory Manual, supra; Davis et al., 1986, Basic Methods in Molecular Biology, Elsevier; Chu et al, 1981, Gene 13: 197.
- SPR surface plasmon resonance
- FACS fluorescence-activated cell sorting
- subject includes any human or nonhuman animal, preferably humans.
- condition associated with MUC16 refers to any condition that is caused by, exacerbated by, or otherwise linked to increased or decreased (generally increased) expression or activities of MUC16 (e.g. a human MUC16).
- cancer refers to any tumor or any malignant cell growth or proliferation, primary or metastasis-mediated, including solid tumors and non-solid tumors such as leukemia.
- treatment refers generally to treatment or therapy, whether of a human or an animal, in which some desired therapeutic effect is achieved, for example, inhibition of the progress of a condition, and includes a reduction in the rate of progress, a halt in the rate of progress, regression of the condition, amelioration of the condition, and cure of the condition.
- Treatment as a prophylactic measure e.g., prophylaxis, prevention
- treating may refer to a dampening or slowing of a tumor or malignant cell growth, proliferation, or metastasis, or some combination thereof.
- treatment includes removal of all or part of a tumor, inhibiting or slowing tumor growth and metastasis, preventing or delaying the development of a tumor, or some combination thereof.
- a “therapeutically-effective amount,” as used herein, pertains to that amount of an active compound, or a material, composition or dosage from comprising an active compound, which is effective for producing some desired therapeutic effect, commensurate with a reasonable benefit/risk ratio, when administered in accordance with a desired treatment regimen.
- a “therapeutically-effective amount,” of a MUC16-binding molecule refers to an amount or concentration effective to treat a human MUC16-related disease or condition.
- host cell refers to a cell with the introduction of exogenous polynucleotides.
- pharmaceutically acceptable means that the vehicle, diluent, excipient and/or salts thereof, are chemically and/or physically compatible with other ingredients in the formulation, and physiologically compatible with the recipient.
- a pharmaceutically acceptable carrier and/or excipient refers to a carrier, stabilizer, and/or excipient pharmacologically and/or physiologically compatible with a subject and an active agent, which is well known in the art (see, e.g., Remington's Pharmaceutical Sciences. Edited by Gennaro AR, 19th ed. Pennsylvania: Mack Publishing Company, 1995), and includes, but is not limited to a pH adjuster, surfactant, adjuvant or an ionic strength enhancer.
- a pH adjuster includes, but is not limited to, phosphate buffer; a surfactant includes, but is not limited to, cationic, anionic, or non-ionic surfactant, e.g., Tween-80; an ionic strength enhancer includes, but is not limited to, sodium chloride.
- Carriers, excipients, or stabilizers are nontoxic to the cell or mammal being exposed thereto at the dosages and concentrations employed. Often the carrier is an aqueous pH buffered solution.
- carriers include buffers such as phosphate, citrate, and other organic acids; antioxidants including ascorbic acid; low molecular weight (e.g., less than about 10 amino acid residues) polypeptide; proteins, such as serum albumin, gelatin, or immunoglobulins; hydrophilic polymers such as polyvinylpyrrolidone; amino acids such as glycine, glutamine, asparagine, arginine or lysine; monosaccharides, disaccharides, and other carbohydrates including glucose, mannose, or dextrins; chelating agents such as EDTA; sugar alcohols such as mannitol or sorbitol; salt-forming counterions such as sodium; and/or nonionic surfactants such as TWEENTM, polyethylene glycol (PEG), and PLURONICSTM.
- buffers such as phosphate, citrate, and other organic acids
- antioxidants including ascorbic acid
- carrier can also refer to a diluent, adjuvant (e.g., Freund’s adjuvant (complete or incomplete)), excipient, or vehicle with which the therapeutic is administered.
- adjuvant e.g., Freund’s adjuvant (complete or incomplete)
- excipient or vehicle with which the therapeutic is administered.
- Such carriers can be sterile liquids, such as water and oils, including those of petroleum, animal, vegetable or synthetic origin, such as peanut oil, soybean oil, mineral oil, sesame oil and the like. Water is a exemplary carrier when a composition (e.g., a pharmaceutical composition) is administered intravenously.
- Saline solutions and aqueous dextrose and glycerol solutions can also be employed as liquid carriers, particularly for injectable solutions.
- Suitable excipients include starch, glucose, lactose, sucrose, gelatin, malt, rice, flour, chalk, silica gel, sodium stearate, glycerol monostearate, talc, sodium chloride, dried skim milk, glycerol, propylene glycol, water, ethanol and the like.
- the composition if desired, can also contain minor amounts of wetting or emulsifying agents, or pH buffering agents.
- Compositions can take the form of solutions, suspensions, emulsion, tablets, pills, capsules, powders, sustained-release formulations and the like.
- compositions can include standard carriers such as pharmaceutical grades of mannitol, lactose, starch, magnesium stearate, sodium saccharine, cellulose, magnesium carbonate, etc. Examples of suitable carriers are described in Remington’s Pharmaceutical Sciences (1990) Mack Publishing Co., Easton, PA.
- Compositions, including pharmaceutical compounds may contain a prophylactically or therapeutically effective amount of a MUC 16-binding agent (e.g., an anti-MUC 16 antibody), for example, in isolated or purified form, together with a suitable amount of carrier so as to provide the form for proper administration to the subject (e.g., patient).
- a MUC 16-binding agent e.g., an anti-MUC 16 antibody
- adjuvant refers to a non-specific immunopotentiator, which can enhance immune response to an antigen or change the type of immune response in an organism when it is delivered together with the antigen to the organism or is delivered to the organism in advance.
- adjuvants including, but not limited to, aluminium adjuvants (for example, aluminum hydroxide), Freund’s adjuvants (for example, Freund’s complete adjuvant and Freund’s incomplete adjuvant), coryne bacterium parvum, lipopolysaccharide, cytokines, and the like.
- Freund's adjuvant is the most commonly used adjuvant in animal experiments.
- Aluminum hydroxide adjuvant is more commonly used in clinical trials.
- MUC 16 comprises a large extracellular domain (CA-125), which is cleaved and released, and a retained domain (MUC-CD).
- MUC-CD comprises a non-repeating extracellular domain (MUC 16 ectodomain) proximal to a cleavage site, a transmembrane domain, and a cytoplasmic tail with potential phosphorylation sites.
- the released extracellular domain (CA-125) contains up to 60 tandem repeats of 156 amino acids, each with many potential glycosylation sites (O'Brien TJ, et al., Tumor Biol 22(6):348-66 (2001)).
- MUC 16 antigen is otherwise expressed only at low levels in normal tissues of the uterus, endometrium, fallopian tubes, ovaries, and serosa of the abdominal and thoracic cavities, MUC 16 is a potentially attractive target for immune-based therapies, including the targeting and treatment of cancer.
- MUC 16 Since a significant portion of the extracellular domain of MUC 16 is cleaved and secreted (i.e., CA-125), the utility of this portion of MUC16 to be used as a target antigen on ovarian carcinomas is limited. Many reported MUC 16 monoclonal antibodies bind to epitopes present on the large secreted CA-125 fraction of the glycoprotein, and not to the retained MUC 16 ectodomain. Thus, the generation of new antibodies to the region of MUC16 that is not shed are needed for diagnostic and therapeutic purposes. This strategy might allow better MUC 16-positive tumor cell targeting efficiency, and significantly improved pharmacokinetics and efficacy of the antibody.
- a MUC16-binding molecule in a general sense, may include any molecule that specifically binds to MUC 16.
- a “MUC16-binding molecule” may include a “MUC 16 antagonist” and an “anti- MUC16 antibody”.
- MUC16 antagonist refers to any chemical compound or biological molecule that blocks MUC 16 activities.
- Anti-MUC16 antibody includes, but not limited to, a chimeric antibody, a humanized antibody, a human antibody or a single-domain antibody.
- a MUC 16- binding molecule is not limited to a polypeptide or a protein and may comprise other components such as nucleotides, hybrids, glucans and a combination thereof.
- a MUC 16- binding molecule may be an MUC16-binding VHH, an anti-MUC16 antibody or anti-MUC16 fusion protein.
- MUC16-binding molecules as disclosed herein comprise at least one VHH that specifically binds to MUC 16.
- a MUC16-binding molecule may be a singledomain antibody and comprising one VHH.
- a single-domain antibody is able to bind selectively to a specific antigen (e.g., MUC 16).
- a MUC16-binding molecule comprises a VHH fused to an immunoglobulin Fc region, for example, an Fc region of IgG (e.g., IgG4 or IgGl).
- the Fc region is an Fc region of human IgGl.
- VHH By fusing a VHH to an Fc region, it may be more efficient to recruit effector functions. Also, fusion of a VHH to an Fc region may help a MUC16-binding molecule to form a dimer and may also help the extension of the half life of the MUC16-binding molecule in vivo.
- VHH molecules derived from Camelidae antibodies are among the smallest intact antigen-binding domains known (approximately 15 kDa, or 10 times smaller than a conventional IgG) and hence are well suited towards delivery to dense tissues and for accessing the limited space between macromolecules.
- VHHs as disclosed herein may be made by the skilled artisan according to methods known in the art or any future method.
- VHHs may be obtained using methods known in the art such as by immunizing a camel and obtaining hybridoma's therefrom, or by cloning a library of VHHs of the disclosure using molecular biology techniques known in the art and subsequent selection by using phage display.
- a VHH can be obtained by immunization of llamas or alpacas with the desired antigen and subsequent isolation of the mRNA coding for heavy-chain antibodies.
- a gene library of single-domain antibodies containing several million clones is produced. Screening techniques like phage display and ribosome display help to identify the clones binding the antigen.
- One technique is phage display in which a library of (e.g., human) antibodies is synthesized on phages, the library is screened with the antigen of interest or an antibody-binding portion thereof, and the phage that binds the antigen is isolated, from which one may obtain the immunoreactive fragments.
- kits for generating phage display libraries are commercially available (e.g., the Pharmacia Recombinant Phage Antibody System, catalog no. 27-9400-01; and the Stratagene SurfZAPTM phage display kit, catalog no. 240612).
- kits for generating phage display libraries are commercially available (e.g., the Pharmacia Recombinant Phage Antibody System, catalog no. 27-9400-01; and the Stratagene SurfZAPTM phage display kit, catalog no. 240612).
- There also are other methods and reagents that can be used in generating and screening antibody display libraries see, e.g., Barbas et al., Proc. Natl. Acad. Sci. USA 88:7978-7982 (1991)).
- potent clones When potent clones have been identified, their DNA sequence is optimized, for example, by affinity maturation or humanization. Humanization may prevent immunological reactions of the human organism against the antibody.
- the VHHs can be obtained (1) by isolating the VHH domain of a naturally occurring heavy chain antibody; (2) by expression of a nucleotide sequence encoding a naturally occurring VHH domain; (3) by “humanization” (as described below) of a naturally occurring VHH domain or by expression of a nucleic acid encoding a such humanized VHH domain; (4) by “camelization” of a naturally occurring VH domain from any animal species, in particular a species of mammal, such as from a human being, or by expression of a nucleic acid encoding such a camelized VH domain; (5) by “camelisation” of a “domain antibody” or “Dab” as described by Ward et al (supra), or by expression of a nucleic acid encoding such a camelized VH domain; (6) using synthetic or semi-synthetic techniques for preparing proteins, polypeptides or other amino acid sequences; (7) by preparing a nucleic acid encoding
- Single-domain antibodies are usually generated by PCR cloning of variable domain repertoire from blood, lymph node, or spleen cDNA obtained from immunized animals into a phage display vector.
- Antigen-specific single-domain antibodies are commonly selected by panning phase libraries on immobilized antigen, for example, antigen coated onto the plastic surface of a test tube, biotinylated antigens immobilized on Streptavidin beads, or membrane proteins expressed on the surface of cells.
- sdAbs can often been improved by mimicking this strategy in vitro, for example, by site directed mutagenesis of the CDR regions and further rounds of panning on immobilized antigen under conditions of increased stringency (higher temperature, high or low salt concentration, high or low pH, and low antigen concentrations) (Wesolowski et al., Single domain antibodies: promising experimental and therapeutic tools in infection and immunity. Med Microbiol Immunol (2009) 198: 157-174).
- a VHH may be truncated at the N-terminus or C-terminus such that it comprises only a partial FRW 1 and/or FRW4, or lacks one or both of those framework regions, so long as the VHH substantially maintains antigen binding and specificity (e.g., substantially maintained, for example, at least 50%, at least 60%, at least 70%, at least 80%, at least 90%, at least 95%).
- the present disclosure also encompasses MUC16-binding molecules with a masking moiety and/or cleavable moiety in which one or more of the MUC16-binding domains of the MUC16- binding molecules are masked (e.g., via a masking moiety) and/or activatable (e.g., via a cleavable moiety).
- a MUC16-binding molecule e.g., an antibody
- SAFE body masking technology see, e.g., US 2019/0241886)
- Probody masking technology see, e.g., US 2015/0079088,.
- Such technologies can be used to generate a MUC16-binding molecule (e.g., an antibody) that is masked and/or activatable.
- masked and/or activatable MUC16-binding molecules e.g., antibodies
- ADCs antibody-drug conjugates
- AADCs activatable antibody-drug conjugates
- MUC16-binding molecules of the present disclosure may be covalently bound by a synthetic linker to one or more agents such as drugs.
- a MUC16-binding molecule is linked or conjugated (directly or indirectly) to a moiety with effector function, such as cytotoxic activity (e.g., a chemotherapeutic moiety or a radioisotope) or immune recruitment activity.
- cytotoxic activity e.g., a chemotherapeutic moiety or a radioisotope
- immune recruitment activity e.g., a chemotherapeutic moiety or a radioisotope
- Moieties that are linked or conjugated (directly or indirectly) include drugs that are cytotoxic (e.g., toxins such as auristatins) or non-cytotoxic (e.g., signal transduction modulators such as kinases or masking moieties that mask one or more binding domains of a MUC16-binding molecule, or cleavable moieties that allow for activating a MUC16- binding molecule by cleaving of a cleavable moiety to unmask one or more binding domains of a MUC16-binding molecule in the tumor microenvironment, in the form of masked conjugates.
- drugs that are cytotoxic e.g., toxins such as auristatins
- non-cytotoxic e.g., signal transduction modulators such as kinases or masking moieties that mask one or more binding domains of a MUC16-binding molecule, or cleavable moieties that allow for activating a MUC
- Moieties that promote immune recruitment can include other antigen-binding agents, such as viral proteins that bind selectively to cells of the innate immune system.
- a MUC16-binding molecule is optionally linked or conjugated (directly or indirectly) to a moiety that facilitates isolation from a mixture (e.g., a tag) or a moiety with reporter activity (e.g., a detection label or reporter protein). It will be appreciated that the features of a MUC16-binding molecule described herein extend also to a polypeptide comprising a MUC16-binding molecule fragment.
- MUC16-binding molecules described herein may be linked or conjugated (directly or indirectly) to a polypeptide, which can result in the generation of an activatable antibody.
- a MUC16-binding molecule is linked or conjugated (directly or indirectly) to an agent.
- the agent is a drug, resulting in an ADC or an AADC when the antibody of the ADC comprises a masking moiety and a cleavable moiety.
- MUC16-binding molecules described herein are conjugated or recombinantly linked (directly or indirectly) to a therapeutic agent (e.g., a cytotoxic agent) or to a diagnostic or detectable agent.
- a therapeutic agent e.g., a cytotoxic agent
- the conjugated or recombinantly linked antibodies, including masked or activatable conjugates, can be useful, for example, for treating or preventing a disease, disorder or condition, such as a cancer or a tumor.
- Diagnosis and detection can be accomplished, for example, by coupling a MUC16-binding molecule to detectable substances including, for example: enzymes, including, but not limited to, horseradish peroxidase, alkaline phosphatase, beta-galactosidase, or acetylcholinesterase; prosthetic groups, including, but not limited to, streptavidin/biotin or avidin/biotin; fluorescent materials, including, but not limited to, umbelliferone, fluorescein, fluorescein isothiocynate, rhodamine, dichlorotriazinylamine fluorescein, dansyl chloride, or phycoerythrin; luminescent materials, including, but not limited to, luminol; bioluminescent materials, including, but not limited to, luciferase, luciferin, or aequorin; chemiluminescent material, including, but not limited to, an acridinium based
- Conjugates of an antibody and agent may be made using a variety of bifunctional protein coupling agents such as BMPS, EMCS, GMBS, HBVS, LC-SMCC, MBS, MPBH, SBAP, SIA, SIAB, SMCC, SMPB, SMPH, sulfo-EMCS, sulfo-GMBS, sulfo-KMUS, sulfo-MBS, sulfo-SIAB, sulfo-SMCC, sulfo- SMPB, and SVSB (succinimidyl-(4-vinylsulfone) benzoate).
- bifunctional protein coupling agents such as BMPS, EMCS, GMBS, HBVS, LC-SMCC, MBS, MPBH, SBAP, SIA, SIAB, SMCC, SMPB, SMPH, sulfo-EMCS, sulfo-GMBS, sulfo-KMUS, s
- conjugates of antibodies and agents including wherein the agent is a drug for the preparation of an ADC or AADC, may be prepared using any suitable methods as disclosed in the art (see, e.g., Bioconjugate Techniques (Hermanson ed., 2d ed. 2008)).
- thiomabs comprising cysteine substitutions at positions on the heavy and light chains that provide reactive thiol groups and do not disrupt immunoglobulin folding and assembly or alter antigen binding (see, e.g., Junutula et al., 2008, J. Immunol. Meth. 332: 41-52; and Junutula et al., 2008, Nature Biotechnol. 26:925-32).
- selenocysteine is cotranslationally inserted into an antibody sequence by recoding the stop codon UGA from termination to selenocysteine insertion, allowing site specific covalent conjugation at the nucleophilic selenol group of selenocysteine in the presence of the other natural amino acids (see, e.g., Hofer et al., 2008, Proc. Natl. Acad. Sci. USA 105:12451-56; and Hofer et al., 2009, Biochemistry 48(50): 12047-57).
- MUC16-binding molecules described herein may be monospecific, bispecific, trispecific or of greater multispecificity.
- Such agents may include antibodies.
- Multispecific antibodies, such as bispecific antibodies are monoclonal antibodies that have binding specificities for at least two different targets (e.g., antigens) or two different epitopes on the same target (e.g., a bispecific antibody directed to MUC16 with a first binding domain for a first epitope of MUC16, and a second binding domain for a second epitope of MUC16.
- the multispecific (e.g., bispecific) antibodies can be constructed based on the sequences of the antibodies described herein.
- the multispecific antibodies described herein are bispecific antibodies.
- bispecific antibodies are mouse, chimeric, human or humanized antibodies.
- one of the binding specificities of the multispecific antibody is for MUC 16 and the other is for any other target (e.g., antigen).
- a multispecific (e.g., bispecific) antibody can comprise more than one target (e.g., antigen) binding domain, in which different binding domains are specific for different targets (e.g., a first binding domain that binds to MUC 16 and a second binding domain that binds another target (e.g., antigen), such as an immune checkpoint regulator (e.g., a negative checkpoint regulator).
- an immune checkpoint regulator e.g., a negative checkpoint regulator
- multispecific (e.g., bispecific) antibody molecules can bind more than one (e.g., two or more) epitopes on the same target (e.g., antigen).
- one of the binding specificities is MUC 16 and the other is for one or more of Cytotoxic T-lymphocyte antigen-4 (CTLA-4), CD80, CD86, Programmed cell death 1 (PD-1), Programmed cell death ligand 1 (PD- Ll), Programmed cell death ligand 2 (PD-L2), Lymphocyte activation gene-3 (LAG-3; also known as CD223), Galectin-3, B and T lymphocyte attenuator (BTLA), T-cell membrane protein 3 (TIM3), Galectin-9 (GAL9), B7-H1, B7-H3, B7-H4, T-Cell immunoreceptor with Ig and ITIM domains (TIGIT/V stm3/WUCAM/VSIG9), V-domain Ig suppressor of T-Cell activation (CTLA-4), CD80,
- multispecific antibodies are known in the art, for example, by coexpression of two immunoglobulin heavy chain-light chain pairs, where the two heavy chains have different specificities (see, e.g., Milstein and Cuello, 1983, Nature 305:537-40).
- multispecific antibodies e.g., bispecific antibodies
- Bispecific Antibodies Kontermann ed., 2011.
- a humanized antibody can have one or more amino acid residues introduced into it from a source that is non-human. These non-human amino acid residues are often referred to as “import” residues, which are typically taken from an “import” variable domain.
- Humanized antibodies that bind MUC 16 may be produced using techniques known to those skilled in the art (e.g., Zhang et al., Molecular Immunology, 42(12): 1445-1451, 2005; Hwang et al., Methods, 36(1): 35-42, 2005; Dall’Acqua et al., Methods, 36(1): 43-60, 2005; Clark, Immunology Today, 21(8): 397-402, 2000, and U.S. Patent Nos. 6,180,370; 6,054,927; 5,869,619; 5,861,155; 5,712,120; and 4,816,567).
- a MUC16-binding molecule may be described as an anti-MUC16 antibody in the following sections.
- Antibodies of the disclosure including, for example, antibodies comprising at least one VHH domain, are characterized by particular functional features or properties of the antibodies.
- the antibodies have one or more of the following properties:
- An antibody of the disclosure binds to cell surface MUC 16 with high affinity.
- the binding of an antibody of the disclosure to MUC 16 can be assessed using one or more techniques well established in the art, for example, ELISA.
- the binding specificity of an antibody of the disclosure can also be determined by monitoring binding of the antibody to cells expressing a MUC 16 protein, e.g., by flow cytometry.
- an antibody can be tested by a flow cytometry assay (e.g., FACS) in which the antibody is reacted with a cell line that expresses human MUC 16, such as CHO cells and 293 cells that have been transfected to express MUC 16 on their cell surface.
- a flow cytometry assay e.g., FACS
- the binding of the antibody can be tested in BIAcore binding assays.
- suitable binding assays include ELISA assays, for example using a recombinant MUC 16 protein.
- an antibody of the disclosure binds to a cell surface MUC 16 (e.g., human MUC 16 55 th &56 th SEA domain) protein with a KD of 1 X 10 -7 M or less, 5 x 10 -8 M or less, 2 x 10 -8 M or less, 5 x 10 -9 M or less, 4 x 10 -9 M or less, 3 x 10 -9 M or less, 2 x 10 -9 M or less, 1 x 10 -9 M or less, 5 x IO' 10 M or less, or 1 x IO' 10 M or less.
- a cell surface MUC 16 e.g., human MUC 16 55 th &56 th SEA domain
- the antibodies of the disclosure bind to cynomolgus monkey MUC 16 (e.g., MUC 16 55 th &56 th SEA domain)at an EC 50 of no more than or about 10 nM, 9 nM, 8 nM, 7 nM, 6 nM, 5 nM, 4 nM, 3 nM, 2 nM, 1 nM, 0.9 nM, 0.8 nM, 0.7 nM, 0.6 nM, 0.5 nM, 0.4 nM, 0.3 nM, 0.2 nM, 0.1 nM, 0.09 nM, 0.08 nM, 0.07 nM, 0.06 nM, 0.05 nM, 0.04 nM, 0.03 nM, 0.02 nM, or 0.01 nM, as measured by FACS.
- cynomolgus monkey MUC 16 e.g., MUC 16 55 th &56 th SEA domain
- Anti-MUC16 antibodies comprising VHH CDRs
- an anti-MUC16 antibody as disclosed herein comprises at least one immunoglobulin single variable domain (e.g., VHH), wherein the VHH comprises CDR1, CDR2 and CDR3, and wherein CDR1 comprises the amino acid sequence as set forth in SEQ ID NO: 1 or an amino acid sequence that differs from SEQ ID NO: 1 by no more than 2 amino acid modifications (e.g. substitution, deletion and/or insertion); CDR2 comprises the amino acid sequence as set forth in SEQ ID NO: 2 or an amino acid sequence that differs from SEQ ID NO: 2 by no more than 2 amino acid modifications (e.g.
- VHH immunoglobulin single variable domain
- CDR1 comprises the amino acid sequence as set forth in SEQ ID NO: 1 or an amino acid sequence that differs from SEQ ID NO: 1 by no more than 2 amino acid modifications (e.g. substitution, deletion and/or insertion)
- CDR2 comprises the amino acid sequence as set forth in SEQ ID NO: 2 or an amino acid sequence that differs from SEQ ID NO: 2
- CDR3 comprises the amino acid sequence as set forth in SEQ ID NO: 3 or an amino acid sequence that differs from SEQ ID NO: 3 by no more than 2 amino acid modifications (e.g. substitution, deletion and/or insertion).
- the amino acid substitution is a conservative substitution.
- CDR1 comprises an amino acid sequence as set forth in SEQ ID NO: 1
- CDR2 comprises an amino acid sequence as set forth in SEQ ID NO: 2
- CDR3 comprises an amino acid sequence as set forth in SEQ ID NO: 3.
- the CDR numbering are according to Kabat + IMGT scheme.
- the extent of the framework region and CDRs can be precisely identified using methodology known in the art, for example, by the Kabat definition, the Chothia definition, the AbM definition, the contact definition, the IMGT definition (all of which are well known in the art) and any combinations thereof. See, e.g., Kabat, E.A., etal. (1991) Sequences of Proteins of Immunological Interest, Fifth Edition, U.S. Department of Health and Human Services, NIH Publication No. 91- 3242, Chothia et al., (1989) Nature 342:877; Chothia, C. et al. (1987) J. Mol. Biol. 196:901-917, Al-lazikani et al (1997) J. Molec.
- variable heavy sequence a variable light sequence and/or a VHH sequence
- disclosure of each variable region is a disclosure of the CDRs (e.g., CDR1, CDR2 and CDR3).
- two antibodies having the same VH, VL or VHH CDRs means that their CDRs are identical when determined by the same approach (e.g., the Kabat, AbM, Chothia, Contact, and IMGT numbering approaches as known in the art).
- Variable regions and CDRs in an antibody sequence can be identified according to general rules that have been developed in the art (for example, the Kabat, AbM, Chothia, Contact, and IMGT numbering system) or by aligning the sequences against a database of known variable regions. Methods for identifying these regions are described in Kontermann and Dubel, eds., Antibody Engineering, Springer, New York, NY, 2001 and Dinarello et al., Current Protocols in Immunology, John Wiley and Sons Inc., Hoboken, NJ, 2000. Exemplary databases of antibody sequences are described in, and can be accessed through, the “Abysis” website at www.bioinf.org.uk/abs (maintained by A.C.
- sequences are analyzed using the Abysis database, which integrates sequence data from Kabat, IMGT and the Protein Data Bank (PDB) with structural data from the PDB. See Dr. Andrew C. R. Martin's book chapter Protein Sequence and Structure Analysis of Antibody Variable Domains. In: Antibody Engineering Lab Manual (Ed.: Duebel, S.
- the Abysis database website further includes general rules that have been developed for identifying CDRs which can be used in accordance with the teachings herein.
- Figure 10 shows an alignment of exemplary immunoglobulin single variable domains and boundaries of CDRs are indicated by Kabat, AbM, Chothia, Contact, and IMGT numbering.
- a MUC16-binding molecule as disclosed herein comprises at least one immunoglobulin single variable domain (e.g., VHH), wherein the VHH comprises FRW1-CDR1- FRW2-CDR2-FRW3-CDR3-FRW4, and wherein CDR1 has an amino acid sequence as set forth in SEQ ID NO: 1, CDR2 has an amino acid sequence as set forth in SEQ ID NO: 2, and CDR3 has an amino acid sequence as set forth in SEQ ID NO: 3.
- VHH immunoglobulin single variable domain
- the FRW1 and FRW4 at the N and C terminal of the VHH comprised in a MUC16-binding molecule may be truncated such that it comprise only a partial FRW1 and/or FRW4, or the VHH lacks one or both of these framework regions, so long as the VHH substantially maintains antigen binding and specificity.
- an anti-MUC16 antibody (such as an anti-MUC16 single domain antibody) comprising one, two, or all three CDRs of the amino acid sequence as set forth in SEQ ID NO: 4.
- an anti-MUC16 antibody (such as an anti-MUC16 single domain antibody) comprising one, two, or all three CDRs of the amino acid sequence as set forth in SEQ ID NO: 5.
- the anti-MUC16 single domain antibody is camelid.
- the anti-MUC16 antibody (such as the anti-MUC16 single domain antibody) is humanized.
- the anti-MUC16 antibody (such as the anti-MUC16 single domain antibody) comprises an acceptor human framework, e.g., a human immunoglobulin framework or a human consensus framework.
- the anti-MUC16 antibody (such as the single domain antibody) comprises a CDR1 same as that of an amino acid sequence as set forth in SEQ ID NO: 4. In some embodiments, the anti-MUC16 antibody (such as the single domain antibody) comprises a CDR2 same as that of an amino acid sequence as set forth in SEQ ID NO: 4. In other embodiments, the anti-MUC16 antibody (such as the single domain antibody) comprises a CDR3 same as that of an amino acid sequence as set forth in SEQ ID NO: 4. In some embodiments, the anti-MUC16 antibody (such as the single domain antibody) comprises a CDR1 and a CDR2 same as those of an amino acid sequence as set forth in SEQ ID NO: 4.
- the anti-MUC16 antibody (such as the single domain antibody) comprises a CDR1 and a CDR3 same as those of an amino acid sequence as set forth in SEQ ID NO: 4. In some embodiments, the anti-MUC16 antibody (such as the single domain antibody) comprises a CDR2 and a CDR3 same as those of an amino acid sequence as set forth in SEQ ID NO: 4. In some embodiments, the anti-MUC16 antibody (such as the single domain antibody) comprises a CDR1, a CDR2, and a CDR3 same as those of an amino acid sequence as set forth in SEQ ID NO: 4. CDR sequences can be determined according to well-known numbering systems. In some embodiments, the CDRs are according to IMGT numbering.
- the CDRs are according to Kabat numbering. In some embodiments, the CDRs are according to a combination of Kabat and IMGT numbering. In other embodiments, the CDRs are according to Chothia numbering. In other embodiments, the CDRs are according to Contact numbering. In some embodiments, the CDRs are according to AbM numbering.
- the anti-MUC16 single domain antibody is camelid. In some embodiments, the anti-MUC16 antibody (such as the anti-MUC16 single domain antibody) is humanized. In some embodiments, the anti-MUC16 antibody (such as the anti-MUC16 single domain antibody) comprises an acceptor human framework, e.g., a human immunoglobulin framework or a human consensus framework.
- the anti-MUC16 antibody (such as the single domain antibody) comprises a CDR1 same as that of an amino acid sequence as set forth in SEQ ID NO: 5. In some embodiments, the anti-MUC16 antibody (such as the single domain antibody) comprises a CDR2 same as that of an amino acid sequence as set forth in SEQ ID NO: 5. In other embodiments, the anti-MUC16 antibody (such as the single domain antibody) comprises a CDR3 same as that of an amino acid sequence as set forth in SEQ ID NO: 5. In some embodiments, the anti-MUC16 antibody (such as the single domain antibody) comprises a CDR1 and a CDR2 same as those of an amino acid sequence as set forth in SEQ ID NO: 5.
- the anti-MUC16 antibody (such as the single domain antibody) comprises a CDR1 and a CDR3 same as those of an amino acid sequence as set forth in SEQ ID NO: 5. In some embodiments, the anti-MUC16 antibody (such as the single domain antibody) comprises a CDR2 and a CDR3 same as those of an amino acid sequence as set forth in SEQ ID NO: 5. In some embodiments, the anti-MUC16 antibody (such as the single domain antibody) comprises a CDR1, a CDR2, and a CDR3 same as those of an amino acid sequence as set forth in SEQ ID NO: 5. CDR sequences can be determined according to well-known numbering systems. In some embodiments, the CDRs are according to IMGT numbering.
- the CDRs are according to Kabat numbering. In some embodiments, the CDRs are according to a combination of Kabat and IMGT numbering. In other embodiments, the CDRs are according to Chothia numbering. In other embodiments, the CDRs are according to Contact numbering. In some embodiments, the CDRs are according to AbM numbering.
- the anti-MUC16 single domain antibody is camelid. In some embodiments, the anti-MUC16 antibody (such as the anti-MUC16 single domain antibody) is humanized. In some embodiments, the anti-MUC16 antibody (such as the anti-MUC16 single domain antibody) comprises an acceptor human framework, e.g., a human immunoglobulin framework or a human consensus framework.
- a single domain antibody that binds to MUC16 comprising the following structure: FR1-CDR1-FR2-CDR2-FR3-CDR3-FR4, wherein (i) the CDR1 comprises an amino acid sequence as set forth in SEQ ID NO: 1; (ii) the CDR2 comprises an amino acid sequence as set forth in SEQ ID NO: 2; and/or (iii) the CDR3 comprises an amino acid sequence as set forth in SEQ ID NO: 3.
- the anti-MUC16 single domain antibody is camelid.
- the anti-MUC16 single domain antibody is humanized.
- the anti-MUC16 single domain antibody comprises an acceptor human framework, e.g., a human immunoglobulin framework or a human consensus framework.
- the CDR1 comprises the exemplary amino acid sequence as set forth in SEQ ID NO: 1; the CDR2 comprises the exemplary amino acid sequence as set forth in SEQ ID NO: 2; and the CDR3 comprises the exemplary amino acid sequence as set forth in SEQ ID NO: 3.
- the CDR1 is according to Kabat+IMGT numbering, comprising the amino acid sequence as set forth in SEQ ID NO: 1; the CDR2 is according to Kabat+IMGT numbering, comprising the amino acid sequence as set forth in SEQ ID NO: 2; and the CDR3 is according to Kabat+IMGT numbering, comprising the amino acid sequence as set forth in SEQ ID NO: 3.
- the anti-MUC16 single domain antibody is camelid.
- the anti-MUC16 single domain antibody is humanized.
- the anti-MUC16 single domain antibody comprises an acceptor human framework, e.g., a human immunoglobulin framework or a human consensus framework.
- the single domain antibody further comprises one or more framework regions of W306106-P11R2-1B8 or W306106-P11R2-1B8-Z6.
- the single domain antibody comprises one or more framework(s) derived from a VHH domain comprising the sequence as set forth in SEQ ID NO: 4.
- the single domain antibody comprises one or more framework(s) derived from a VHH domain comprising the sequence as set forth in SEQ ID NO: 5.
- the single domain antibody provided herein is a humanized single domain antibody. Framework regions described herein are determined based upon the boundaries of the CDR numbering system.
- the framework regions are the amino acid residues surrounding the CDRs in the variable region in the format, from the N-terminus to C-terminus: FR1-CDR1-FR2-CDR2- FR3-CDR3-FR4.
- FR1 is defined as the amino acid residues N-terminal to the CDR1 amino acid residues as defined by, e.g., the IMGT numbering system, the Kabat numbering system, the Chothia numbering system, the Contact numbering system, the AbM numbering system or a combination thereof
- FR2 is defined as the amino acid residues between CDR1 and CDR2 amino acid residues as defined by, e.g., the IMGT numbering system, the Kabat numbering system, the Chothia numbering system, the Contact numbering system, the AbM numbering system or a combination thereof
- FR3 is defined as the amino acid residues between CDR2 and CDR3 amino acid residues as defined by, e.g., the IMGT numbering system, the Kabat numbering system, the Chothia numbering system, the Contact numbering system, the AbM numbering system or a combination thereof
- FR4 is defined as the amino acid residues C -terminal to the CDR3 amino acid residues as defined by
- an isolated anti-MUC16 single domain antibody comprising a VHH domain having the amino acid sequence as set forth in SEQ ID NO: 4. In some embodiments, there is provided a polypeptide comprising the amino acid sequence as set forth in SEQ ID NO: 4. In some embodiments, there is provided an isolated anti-MUC16 single domain antibody comprising a VHH domain having the amino acid sequence as set forth in SEQ ID NO: 5. In some embodiments, there is provided a polypeptide comprising the amino acid sequence as set forth in SEQ ID NO: 5.
- Anti-MUC16 antibodies comprising VHH sequences
- anti-MUC16 antibodies comprise at least one immunoglobulin single variable domain (e.g., VHH), wherein the VHH comprises or consists of:
- (C) an amino acid sequence with addition, deletion and/or substitution of one or more (for example, 1, 2, 3, 4, 5, 6, 7, 8, 9 or 10) amino acids compared with any one of SEQ ID NOs: 4 and 5.
- the percent identity between two amino acid sequences can be determined using the algorithm of E. Meyers and W. Miller (Comput. Appl. Biosci., 4:11-17 (1988)) which has been incorporated into the ALIGN program (version 2.0), using a PAM120 weight residue table, a gap length penalty of 12 and a gap penalty of 4.
- the percent identity between two amino acid sequences can be determined by the algorithm of Needleman and Wunsch (J. Mol. Biol.
- protein sequences of the present disclosure can further be used as a “query sequence” to perform a search against public databases to, for example, identify related sequences.
- Such searches can be performed using the XBLAST program (version 2.0) of Altschul, et al. (1990) J. Mol. Biol. 215:403-10.
- Gapped BLAST can be utilized as described in Altschul et al, (1997) Nucleic Acids Res. 25(17):3389-3402.
- the default parameters of the respective programs ⁇ e.g., XBLAST and NBLAST) can be used. See www.ncbi.nlm.nih.gov.
- the amino acid sequence of a VHH can be at least 85%, 86%, 87%, 88%, 89%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98% or 99% identical to any one of SEQ ID NOs: 4 and 5.
- the VHH may have same CDRs (CDR1, CDR2 and CDR3) as those of SEQ ID NO: 4 or 5, and at least 85%, 86%, 87%, 88%, 89%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98% or 99% identical in the framework region to those of SEQ ID NO: 4 or 5.
- anti-MUC16 antibodies may contain conservative substitution or modification of amino acids in the variable regions and/or constant regions. It is understood in the art that certain conservative sequence modification can be made which do not remove antigen binding. See, e.g., Brummell et al. (1993) Biochem 32:1180-8; de Wildt et al. (1997) Prot. Eng. 10:835-41; Komissarov et al. (1997) J. Biol. Chem. 272:26864- 26870; Hall et al. (1992) J. Immunol. 149:1605-12; Kelley and O’ Connell (1993) Biochem. 32:6862-35; Adib- Conquy et al. (1998) Int. Immunol. 10:341-6 and Beers et al. (2000) Clin. Can. Res. 6:2835-43.
- conservative substitution refers to an amino acid substitution which would not disadvantageously affect or change the essential properties of a protein/polypeptide comprising the amino acid sequence.
- a conservative substitution may be introduced by standard techniques known in the art such as site-directed mutagenesis and PCR-mediated mutagenesis.
- Conservative amino acid substitutions include substitutions wherein an amino acid residue is substituted with another amino acid residue having a similar side chain, for example, a residue physically or functionally similar (such as, having similar size, shape, charge, chemical property including the capability of forming covalent bond or hydrogen bond, etc.) to the corresponding amino acid residue.
- the families of amino acid residues having similar side chains have been defined in the art.
- amino acids having alkaline side chains for example, lysine, arginine and histidine
- amino acids having acidic side chains for example, aspartic acid and glutamic acid
- amino acids having uncharged polar side chains for example, glycine, asparagine, glutamine, serine, threonine, tyrosine, cysteine, tryptophan
- amino acids having nonpolar side chains for example, alanine, valine, leucine, isoleucine, proline, phenylalanine, methionine
- amino acids having P-branched side chains such as threonine, valine, isoleucine
- amino acids having aromatic side chains for example, tyrosine, phenylalanine, tryptophan, histidine.
- a corresponding amino acid residue is preferably substituted with another amino acid residue from the same side-chain family.
- Methods for identifying amino acid conservative substitutions are well known in the art (see, for example, Brummell et al., Biochem. 32: 1180-1187 (1993); Kobayashi et al., Protein Eng. 12(10): 879-884 (1999); and Burks et al., Proc. Natl. Acad. Sci. USA 94: 412- 417 (1997), which are incorporated herein by reference).
- the anti-MUC16 antibody comprises at least one VHH, and the VHH comprises an amino acid sequence as set forth in any one of SEQ ID NOs: 4-5. In some embodiments, the anti-MUC16 antibody comprises a VHH consists of the amino acid sequence as set forth in any one of SEQ ID NOs: 4-5.
- the anti-MUC16 antibody is a chimeric antibody, comprising a VHH fused to an Fc region of human IgGl or IgG4, wherein the VHH comprises an amino acid sequence as set forth in any one of SEQ ID NOs: 4-5.
- the anti-MUC16 antibody is a chimeric antibody comprising a VHH and an Fc region of human IgGl.
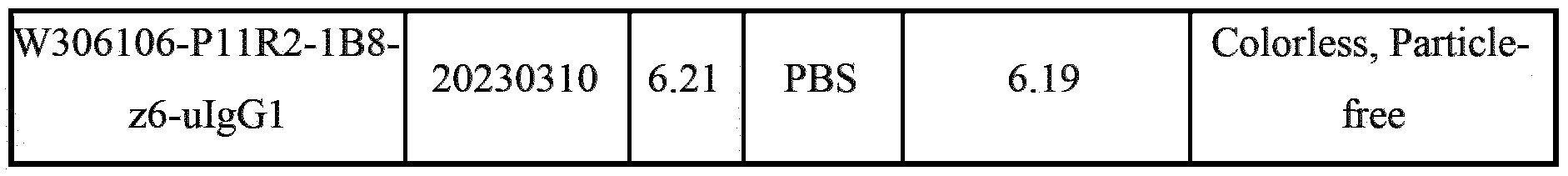
- Such antibodies are exemplified herein as “W306106-Pl lR2-lB8-uIgG1”.
- an anti- MUC16 antibody is a humanized antibody comprising a VHH and an Fc region of human IgGl.
- Such antibodies are exemplified herein as and “W306106-P1 lR2-lB8-z6-uIgG1”.
- the addition, deletion and/or substitution of at least one of the amino acids in the VHH region is not in any of the CDR sequences, but in the framework (FRW) sequences.
- an antibody or antigen-binding portion thereof as described above may comprise one or more substitutions of the amino acids in the framework sequences, e.g. FRW1, FRW2, FRW3, and/or FRW4 of the VHH region.
- an antibody or antigen-binding portion thereof as provided herein comprises any suitable framework region (FRW) sequences, as long as the antigen-binding domains can specifically bind to MUC16.
- FRW framework region
- an antibody or antigen-binding portion thereof may contain modification of one or more amino acids in the variable regions of the heavy chain and/or light chain, including wherein the modification is a conservative substitution. It is understood in the art that certain conservative sequence modifications can be made which do not remove antigen binding. See, e.g., Brummell et al. ( 1993) Biochem 32:1180-8; de Wildt et al. ( 1997) Prot. Eng. 10:835-41; Komissarov et al. (1997) J. Biol. Chem. 272:26864- 26870; Hall et al. (1992) J. Immunol. 149:1605-12; Kelley and O’ Connell (1993) Biochem. 32:6862-35; Adib-Conquy et al. (1998) Int. Immunol. 10:341-6 and Beers et al. (2000) Clin. Can. Res. 6:2835-43.
- an antibody or antigen-binding portion thereof comprises a VHH domain comprising an amino acid sequence as set forth in any one of SEQ ID NOs: 4-5, and a Fc region, optionally the antibody or antigen-binding portion thereof comprises an amino acid sequence as set forth in SEQ ID NO: 6 or 7.
- an antigen-binding domain of a MUC16-binding molecule is not limited to the VHH form and may adopt a variety of other formats, such as but not limited to, a Fab, a Fab', a F(ab')2, an Fv fragment, a single-chain antibody molecule (scFv).
- an antigen-binding domain is a Fv fragment with a VH region and a VL region in separate chains held together by tight, non-covalent interactions.
- Fc region comprising IgG constant domains
- Anti-MUC16 antibodies and antigen-binding fragments provided herein further comprise an Fc region comprising one or more human IgG constant domains.
- a human IgG constant domain may be a human IgGl, IgG2, IgG3 or IgG4 constant domain, preferably a human IgGl constant domain.
- the Fc region is a human IgGl Fc region, such as a wild-type Fc region or a Fc variant comprising one or more amino acid modifications (e.g. Leu234Ala/Leu235Ala or LALA) that alters the antibody-dependent cellular cytotoxicity (ADCC) or other effector fimctions.
- ADCC antibody-dependent cellular cytotoxicity
- the Fc modification comprises a LALA mutation, e.g. mutations of L234A and L235A, according to EU numbering as in Kabat et al..
- the Kabat numbering system is often used when referring to a residue in the variable domain (approximately residues 1-107 of the light chain and residues 1-113 of the heavy chain) (e.g., Kabat et al., Sequences of Immunological Interest. 5th Ed. Public Health Service, National Institutes of Health, Bethesda, Md. (1991)).
- EU numbering system or “EU index” is generally used when referring to a residue in an immunoglobulin heavy chain constant region (e.g., the EU index reported in Kabat et al., supra).
- EU numbering as in Kabat or “EU index as in Kabat” refers to the residue numbering of the human IgGl EU antibody. Unless stated otherwise herein, references to residue numbers in the constant domain of antibodies means residue numbering by the EU numbering system.
- the present disclosure provides a nucleic acid molecule comprising a nucleic acid sequence encoding a MUC16-binding molecule as disclosed herein, for example, encoding a single variable domain of a MUC16-binding molecule as disclosed herein.
- Nucleic acids of the disclosure can be obtained using standard molecular biology techniques.
- a nucleic acid encoding a VHH region can be converted to a full-length heavy chain gene by operatively linking the VHH-encoding nucleic acid to another nucleic acid encoding one or more heavy chain constant regions (e.g. CHI, CH2 and CH3).
- the sequences of human heavy chain constant region genes are known in the art (see e.g., Kabat et al. ( 1991 ), supra) and DNA fragments encompassing these regions can be obtained by standard PCR amplification.
- a heavy chain constant region can be an IgGl, IgG2, IgG3, IgG4, IgA, IgE, IgM or IgD constant region, such as an IgGl constant region.
- nucleic acids encoding VHH segments are obtained, these nucleic acids can be further manipulated by standard recombinant DNA techniques, for example to convert variable region genes to full-length antibody chain genes.
- a VHH-encoding nucleic acid is operatively linked to another nucleic acid encoding another protein, such as an antibody constant region or a flexible linker.
- the term “operatively linked”, as used in this context, is intended to mean that two or more nucleic acids are joined such that the amino acid sequences encoded by the two or more nucleic acids remain in-frame.
- the disclosure is directed to a nucleic acid molecule, comprising a nucleic acid sequence encoding a single variable domain (e.g., VHH) of a MUC16-binding molecule as disclosed herein.
- a single variable domain e.g., VHH
- the nucleic acid molecule comprises a nucleic acid sequence selected from the group consisting of:
- (C) a nucleic acid sequence that hybridizes under high stringency conditions to the complementary strand of the nucleic acid sequence of (A).
- nucleic acid molecule comprises a nucleic acid sequence encoding an anti-MUC16 single domain antibody comprising the amino acid sequence as set forth in SEQ ID NO: 4. In some embodiments, provided herein is a nucleic acid molecule comprises a nucleic acid sequence encoding an anti-MUC16 single domain antibody comprising the amino acid sequence as set forth in SEQ ID NO: 5. In some embodiments, the percent identity is derived from the degeneracy of the genetic code, and the encoded protein sequences remain unchanged.
- Exemplary high stringency conditions include hybridization at 45°C in 5X SSPE and 45% formamide, and a final wash at 65°C in 0.1 X SSC. It is understood in the art that conditions of equivalent stringency can be achieved through variation of temperature and buffer, or salt concentration as described Ausubel, et al. (Eds.), Protocols in Molecular Biology, John Wiley & Sons (1994), pp. 6.0.3 to 6.4.10. Modifications in hybridization conditions can be empirically determined or precisely calculated based on the length and the percentage of guanosine/ cytosine (GC) base pairing of the probe. The hybridization conditions can be calculated as described in Sambrook, et al, (Eds.), Molecular Cloning: A laboratory Manual. Cold Spring Harbor Laboratory Press: Cold Spring Harbor, New York (1989), pp. 9.47 to 9.51.
- Host cells as disclosed in the present disclosure may be any cell which is suitable for expressing the antibodies of the present disclosure, for example, yeast, bacterial, plant and mammalian cells.
- Mammalian host cells for expressing the antibodies of the present disclosure include Chinese Hamster Ovary (CHO cells) (including dhfr CHO cells, described in Urlaub and Chasin, (1980) Proc. Natl. Acad. ScL USA 77:4216-4220, used with a DHFR selectable marker, e.g., as described in R. J. Kaufman and P. A. Sharp (1982) J. Mol. Biol. 159:601-621), 293F cells, NSO myeloma cells, COS cells and SP2 cells.
- CHO cells Chinese Hamster Ovary (CHO cells) (including dhfr CHO cells, described in Urlaub and Chasin, (1980) Proc. Natl. Acad. ScL USA 77:4216-4220, used with a DHFR select
- GS gene expression system for use with NSO myeloma cells, another expression system is the GS gene expression system disclosed in WO 87/04462, WO 89/01036 and EP 338,841.
- monkey kidney CV1 line transformed by SV40 COS-7, ATCC CRL 1651
- human embryonic kidney line (293 or 293 cells subcloned for growth in suspension culture, Graham et al., J. Gen Virol. 36:59 (1977)
- baby hamster kidney cells BHK, ATCC CCL 10
- Chinese hamster ovary cells/-DHFR CHO, Urlaub et al., 1980, Proc. Natl. Acad. Sci. USA 77:4216
- mouse sertoli cells TM4, Mather, 1980, Biol.
- monkey kidney cells (CV1 ATCC CCL 70); African green monkey kidney cells (VERO-76, ATCC CRL- 1587); human cervical carcinoma cells (HELA, ATCC CCL 2); canine kidney cells (MDCK, ATCC CCL 34); buffalo rat liver cells (BRL 3A, ATCC CRL 1442); human lung cells (W138, ATCC CCL 75); human liver cells (Hep G2, HB 8065); mouse mammary tumor (MMT 060562, ATCC CCL51); TRI cells (Mather et al., 1982, Annals N.Y. Acad. Sci.
- MRC 5 cells MRC 5 cells; FS4 cells; mouse myeloma cells, such as NSO (e.g. RCB0213, 1992, Bio/Technology 10: 169) and SP2/0 cells (e.g. SP2/0-Agl4 cells, ATCC CRL 1581); rat myeloma cells, such as YB2/0 cells (e.g. YB2/3HL.P2.G11.16Ag.20 cells, ATCC CRL 1662); PER.C6 cells; and a human hepatoma line (Hep G2).
- NSO e.g. RCB0213, 1992, Bio/Technology 10: 169
- SP2/0 cells e.g. SP2/0-Agl4 cells, ATCC CRL 1581
- rat myeloma cells such as YB2/0 cells (e.g. YB2/3HL.P2.G11.16Ag.20 cells, ATCC CRL 1662); PER.C6 cells; and
- CHO cells are one of the cell lines that can be used herein, with CHO-K1, DUK- B11, CH0-DP12, CHO-DG44 (Somatic Cell and Molecular Genetics 12:555 (1986)), and Lecl3 being exemplary host cell lines.
- CHO-K1, DUK-B11, DG44 or CHO-DP12 host cells these may be altered such that they are deficient in their ability to fucosylate proteins expressed therein.
- the host cells herein are selected from CHO, CHO-S, HEK, HEK293, HEK-293F, Expi293F, PER.C6 or NSO cells or lymphocytic cells.
- Suitable prokaryotes for this purpose include eubacteria, such as Gram-negative or Grampositive organisms, for example, Enterobacteriaceae such as Escherichia, e.g., E. coli, Enterobacter, Erwinia, Klebsiella, Proteus, Salmonella, e.g., Salmonella typhimurium, Serratia, e.g., Serratia marcescans, and Shigella, as well as Bacilli such as B. subtilis and B. licheniformis, Pseudomonas such as P. aeruginosa, and Streptomyces.
- Enterobacteriaceae such as Escherichia, e.g., E. coli, Enterobacter, Erwinia, Klebsiella, Proteus
- Salmonella e.g., Salmonella typhimurium
- Serratia e.g., Serratia marcescans
- Shigella Salmonella
- eukaryotic microbes such as filamentous fungi or yeast are also suitable cloning or expression hosts for antibody-encoding vectors.
- Saccharomyces cerevisiae, or common baker’s yeast is the most commonly used among lower eukaryotic host microorganisms.
- a number of other genera, species, and strains are commonly available and useful herein, such as Schizosaccharomyces pombe; Kluyveromyces hosts such as, e.g., K. lactis, K. fragilis (ATCC 12,424), K. bulgaricus (ATCC 16,045), K. wickeramii (ATCC 24,178), K.
- waltii ATCC 56,500
- K. drosophilarum ATCC 36,906
- K. thermotolerans K. marxianus
- yarrowia EP 402,226
- Pichia pastoris EP 183,070
- Candida Trichoderma reesia
- Neurosporacrassa Schwanniomyces such as Schwanniomycesoccidentalis
- filamentous fungi such as, e.g., Neurospora, Penicillium, Tolypocladium, and Aspergillus hosts such as A. nidulans and A. niger.
- the antibody When recombinant expression vectors encoding an antibody are introduced into mammalian host cells, the antibody is produced by culturing the host cells for a period of time sufficient to allow for expression of the antibody in the host cells or, secretion of the antibody into the culture medium in which the host cells are grown. Antibodies can be recovered from the culture medium using standard protein purification methods.
- the present disclosure provides a pharmaceutical composition comprising a MUC16-binding molecule as disclosed herein, for example, comprising a single variable domain (e.g., VHH) of a MUC16-binding molecule as disclosed herein and a pharmaceutically acceptable carrier.
- the present disclosure provides a pharmaceutical composition comprising a nucleic acid encoding a MUC16-binding molecule as disclosed herein, for example, comprising a single variable domain (e.g., VHH) of a MUC16-binding molecule as disclosed herein and a pharmaceutically acceptable carrier.
- the present disclosure provides a pharmaceutical composition
- a pharmaceutical composition comprising a cell expressing a MUC16-binding molecule as disclosed herein, for example, comprising a single variable domain (e.g., VHH) of a MUC16-binding molecule as disclosed herein and a pharmaceutically acceptable carrier.
- a single variable domain e.g., VHH
- the pharmaceutical composition may optionally contain one or more additional components, including one or more pharmaceutically active ingredients, such as another antibody or a drug.
- additional components including one or more pharmaceutically active ingredients, such as another antibody or a drug.
- the pharmaceutical compositions of the disclosure also can be administered in a combination therapy with, for example, another immune-stimulatory agent, anti-cancer agent, an antiviral agent, or a vaccine, including wherein the anti-MUC16 antibody enhances the immune response.
- a pharmaceutically acceptable carrier can include, for example, a pharmaceutically acceptable liquid, gel or solid carriers, an aqueous medium, a non-aqueous medium, an anti-microbial agent, isotonic agents, buffers, antioxidants, anesthetics, suspending/dispersing agent, a chelating agent, a diluent, adjuvant, excipient or a nontoxic auxiliary substance, other known in the art various combinations of components or more.
- Suitable components of the pharmaceutical composition may include, for example, antioxidants, fillers, binders, disintegrating agents, buffers, preservatives, lubricants, flavorings, thickening agents, coloring agents, emulsifiers or stabilizers such as sugars and cyclodextrin.
- Suitable anti-oxidants may include, for example, methionine, ascorbic acid, EDTA, sodium thiosulfate, platinum, catalase, citric acid, cysteine, mercapto glycerol, thioglycolic acid, Mercapto sorbitol, butyl methyl anisole, butylated hydroxy toluene and/or propyl gallate.
- a composition may comprise an antibody or an antigen-binding fragment of the present disclosure and also comprise one or more anti-oxidants such as methionine, to prevent or reduce a decrease in binding affinity, thereby enhancing antibody stability and extended shelflife.
- the present disclosure provides a composition comprising one or more antibodies or antigen binding fragment thereof and one or more anti-oxidants such as methionine.
- the present disclosure further provides a variety of methods, wherein an antibody or antigen binding fragment thereof is mixed with one or more anti-oxidants, such as methionine, so that the antibody or antigen binding fragment thereof can be prevented from oxidation, to extend their shelflife and/or increased activity.
- pharmaceutical acceptable carriers may include, for example, aqueous vehicles such as sodium chloride injection, Ringer's injection, isotonic dextrose injection, sterile water injection, or dextrose and lactated Ringer's injection, nonaqueous vehicles such as fixed oils of vegetable origin, cottonseed oil, com oil, sesame oil, or peanut oil, antimicrobial agents at bacteriostatic or fungistatic concentrations, isotonic agents such as sodium chloride or dextrose, buffers such as phosphate or citrate buffers, antioxidants such as sodium bisulfate, local anesthetics such as procaine hydrochloride, suspending and dispersing agents such as sodium carboxymethylcelluose, hydroxypropyl methylcellulose, or polyvinylpyrrolidone, emulsifying agents such as Polysorbate 80 (TWEEN-80), sequestering or chelating agents such as EDTA (ethylenediaminetetraacetic acid) or EGTA (ethylene
- Antimicrobial agents utilized as carriers may be added to pharmaceutical compositions in multiple-dose containers that include phenols or cresols, mercurials, benzyl alcohol, chlorobutanol, methyl and propyl p-hydroxybenzoic acid esters, thimerosal, benzalkonium chloride and benzethonium chloride.
- Suitable excipients may include, for example, water, saline, dextrose, glycerol, or ethanol.
- Suitable non-toxic auxiliary substances may include, for example, wetting or emulsifying agents, pH buffering agents, stabilizers, solubility enhancers, or agents such as sodium acetate, sorbitan monolaurate, triethanolamine oleate, or cyclodextrin.
- a pharmaceutical composition of the disclosure may be administered to a subject in need thereof, by various routes, including, but not limited to, oral, intravenous, intra-arterial, subcutaneous, parenteral, intranasal, intramuscular, intracranial, intracardiac, intraventricular, intratracheal, buccal, rectal, intraperitoneal, intradermal, topical, transdermal, and intrathecal, or otherwise by implantation or inhalation.
- the subject compositions may be formulated into preparations in solid, semi-solid, liquid, or gaseous forms; including, but not limited to, tablets, capsules, powders, granules, ointments, solutions, suppositories, enemas, injections, inhalants, and aerosols.
- the appropriate formulation and route of administration may be selected according to the intended application and therapeutic regimen.
- Suitable formulations for enteral administration include hard or soft gelatin capsules, pills, tablets, including coated tablets, elixirs, suspensions, syrups or inhalations and controlled release forms thereof.
- Formulations suitable for parenteral administration include aqueous or non-aqueous, isotonic, pyrogen-free, sterile liquids (e.g., solutions, suspensions), in which the active ingredient is dissolved, suspended, or otherwise provided (e.g., in a liposome or other microparticulate).
- Such liquids may additional contain other pharmaceutically acceptable ingredients, such as anti-oxidants, buffers, preservatives, stabilisers, bacteriostats, suspending agents, thickening agents, and solutes which render the formulation isotonic with the blood (or other relevant bodily fluid) of the intended recipient.
- excipients include, for example, water, alcohols, polyols, glycerol, vegetable oils, and the like.
- suitable isotonic carriers for use in such formulations include Sodium Chloride Injection, Ringer's Solution, or Lactated Ringer's Injection.
- the particular dosage regimen, including dose, timing and repetition, will depend on the particular individual and that individual's medical history, as well as empirical considerations such as pharmacokinetics (e.g., half-life, clearance rate, etc.).
- Frequency of administration may be determined and adjusted over the course of therapy, and is based on reducing the number of proliferative or tumorigenic cells, maintaining the reduction of such neoplastic cells, reducing the proliferation of neoplastic cells, or delaying the development of metastasis.
- the dosage administered may be adjusted or attenuated to manage potential side effects and/or toxicity.
- sustained continuous release formulations of a subject therapeutic composition may be appropriate.
- appropriate dosages can vary from patient to patient. Determining the optimal dosage will generally involve the balancing of the level of therapeutic benefit against any risk or deleterious side effects.
- the selected dosage level will depend on a variety of factors including, but not limited to, the activity of the particular compound, the route of administration, the time of administration, the rate of excretion of the compound, the duration of the treatment, other drugs, compounds, and/or materials used in combination, the severity of the condition, and the species, sex, age, weight, condition, general health, and prior medical history of the patient.
- the amount of compound and route of administration will ultimately be at the discretion of the physician, veterinarian, or clinician, although generally the dosage will be selected to achieve local concentrations at the site of action that achieve the desired effect without causing substantial harmful or deleterious side-effects.
- a MUC16-binding molecule of the disclosure may be administered in various ranges. These include about 5 ⁇ g/kg body weight to about 100 mg/kg body weight per dose; about 50 ⁇ g/kg body weight to about 5 mg/kg body weight per dose; about 100 ⁇ g/kg body weight to about 10 mg/kg body weight per dose; and any values within the ranges. Other ranges include about 100 ⁇ g/kg body weight to about 20 mg/kg body weight per dose and about 0.5 mg/kg body weight to about 20 mg/kg body weight per dose.
- the dosage is at least about 100 ⁇ g/kg body weight, at least about 250 ⁇ g/kg body weight, at least about 750 ⁇ g/kg body weight, at least about 3 mg/kg body weight, at least about 5 mg/kg body weight, at least about 10 mg/kg body weight.
- an antibody or antigen binding portion thereof of the disclosure is preferably administered as needed to subjects in need thereof. Determination of the frequency of administration may be made by persons skilled in the art, such as an attending physician based on considerations of the condition being treated, age of the subject being treated, severity of the condition being treated, general state of health of the subject being treated and the like.
- the course of treatment involving a MUC16-binding molecule of the present disclosure will comprise multiple doses of the selected drug product over a period of weeks or months.
- a MUC16-binding molecule of the present disclosure may be administered once every day, every two days, every four days, every week, every ten days, every two weeks, every three weeks, every month, every six weeks, every two months, every ten weeks or every three months.
- the dosages may be altered or the interval may be adjusted based on patient response and clinical practices.
- Dosages and regimens may also be determined empirically for the disclosed therapeutic compositions in individuals who have been given one or more administration(s). For example, individuals may be given incremental dosages of a therapeutic composition produced as described herein. In some embodiments, the dosage may be gradually increased or reduced or attenuated based respectively on empirically determined or observed side effects or toxicity. To assess efficacy of the selected composition, a marker of the specific disease, disorder or condition can be followed as described previously.
- these include direct measurements of tumor size via palpation or visual observation, indirect measurement of tumor size by x-ray or other imaging techniques; an improvement as assessed by direct tumor biopsy and microscopic examination of the tumor sample; the measurement of an indirect tumor marker (e.g., PSA for prostate cancer) or a tumorigenic antigen, a decrease in pain or paralysis; improved speech, vision, breathing or other disability associated with the tumor; increased appetite; or an increase in quality of life as measured by accepted tests or prolongation of survival.
- an indirect tumor marker e.g., PSA for prostate cancer
- a tumorigenic antigen e.g., a tumorigenic antigen
- the dosage will vary depending on the individual, the type of neoplastic condition, the stage of neoplastic condition, whether the neoplastic condition has begun to metastasize to other location in the individual, and the past and concurrent treatments being used.
- Compatible formulations for parenteral administration may comprise a MUC16-binding molecule as disclosed herein in concentrations of from about 10 ⁇ g/ml to about 100 mg/ml.
- the concentrations of the MUC16-binding molecule e.g., antibody or the antigen binding portion thereof
- the concentrations of the MUC16-binding molecule will comprise 2 mg/ml, 3 mg/ml, 4 mg/ml, 5 mg/ml, 6 mg/ml, 8 mg/ml, 10 mg/ml, 12 mg/ml, 14 mg/ml, 16 mg/ml, 18 mg/ml, 20 mg/ml, 25 mg/ml, 30 mg/ml, 35 mg/ml, 40 mg/ml, 45 mg/ml, 50 mg/ml, 60 mg/ml, 70 mg/ml, 80 mg/ml, 90 mg/ml or 100 mg/ml.
- the MUC16-binding molecule e.g., antibody or the antigen binding portion thereof
- the antibodies, antibody compositions and methods of the present disclosure have numerous in vitro and in vivo utilities and uses including, for example, detection of MUC16 or enhancement of immune response.
- these molecules can be administered to cells in culture, in vitro or ex vivo, or to human subjects, e.g., in vivo, to enhance immunity in a variety of situations.
- the immune response can be modulated, for example, augmented, stimulated or up-regulated.
- the subjects include human patients in need of enhancement of an immune response.
- the methods are particularly suitable for treating human patients having a disorder that can be treated by augmenting an immune response (e.g., a T-cell mediated immune response). In some embodiments, the methods are particularly suitable for treatment of cancer in vivo.
- anti-MUC16 antibodies can be administered together with an antigen of interest or the antigen may already be present in the subject to be treated (e.g., a tumor-bearing or virus-bearing subject).
- the two can be administered in either order or simultaneously.
- the present disclosure further provides methods for detecting the presence of human MUC 16 antigen in a sample, or measuring the amount of human MUC 16 antigen, comprising contacting the sample, and a control sample, for example, with a human monoclonal antibody, or an antigen binding portion thereof, which specifically binds to human MUC 16, under conditions that allow for formation of a complex between the antibody or portion thereof and human MUC 16. The formation of a complex is then detected, wherein a difference complex formation between the sample compared to the control sample is indicative of the presence of human MUC16 antigen in the sample.
- anti-MUC16 antibodies of the disclosure can be used to purify human MUC 16 via immunoaffinity purification.
- the present disclosure provides a method of treating a disorder or a disease in a mammal, which comprises administering to the subject (for example, a human) in need of treatment a therapeutically effective amount of an anti-MUC16 antibody or antigen-binding portion thereof as disclosed herein.
- the present disclosure provides an anti- MUC16 antibody or antigen-binding portion thereof as disclosed herein for use in treating a disease or disorder.
- provided herein is a use of an anti-MUC16 antibody or antigen-binding portion thereof as disclosed herein for the manufacture of a medicament for the treatment of a disease or disorder.
- the disorder or disease may be a cancer.
- a variety of cancers where MUC 16 is implicated, whether malignant or benign and whether primary or secondary, may be treated or prevented with a method provided by the disclosure.
- the cancers may include, but not limited to, ovary cancer, lung cancer, pancreas cancer, breast cancer, uterine cancer, fallopian tube cancer, primary peritoneum cancer, adrenal cancer, liver cancer, kidney cancer, bladder cancer, gastric cancer, cervical cancer, esophageal cancer, colorectal cancer, prostate cancer, thyroid cancer, sarcomas, glioblastomas and head and neck cancer or cancer of any other tissue that expresses MUC16.
- Anti-MUC16 antibodies as disclosed herein can be used for treating lung cancers such as bronchogenic carcinoma, non-small cell lung cancer, squamous cell carcinoma, small cell carcinoma, large cell carcinoma, and adenocarcinoma, e.g. lung adenocarcinoma.
- the lung cancers may be refractory, relapsed or resistant to a platinum based agent (e.g., carboplatin, cisplatin, oxaliplatin, topotecan) and/or a taxane (e.g., docetaxel, paclitaxel, larotaxel or cabazitaxel).
- a platinum based agent e.g., carboplatin, cisplatin, oxaliplatin, topotecan
- a taxane e.g., docetaxel, paclitaxel, larotaxel or cabazitaxel.
- Cancers to be treated by anti-MUC16 antibodies as disclosed herein may also be large cell neuroendocrine carcinoma (LCNEC), medullary thyroid cancer, glioblastoma, neuroendocrine prostate cancer (NEPC), high-grade gastroenteropancreatic cancer (GEP) and malignant melanoma.
- Anti-MUC16 antibodies as disclosed herein may be used to treat neuroendocrine tumors (both NET and pNET) arising in the kidney, genitourinary tract (bladder, prostate, ovary, cervix, and endometrium), gastrointestinal tract (colon, stomach), thyroid (medullary thyroid cancer), and lung (small cell lung carcinoma and large cell neuroendocrine carcinoma).
- the disclosure also provides a method of enhancing (for example, stimulating) an immune response in a subject comprising administering to the subject a MUC16- binding molecule, for example, an anti-MUC16 antibody or an antigen binding portion thereof, of the disclosure such that an immune response in the subject is enhanced.
- the present disclosure provides an anti-MUC16 antibody or antigen-binding portion thereof as disclosed herein for use in enhancing (for example, stimulating) an immune response in a subject.
- provided herein is a use of an anti-MUC16 antibody or antigen-binding portion thereof as disclosed herein for the manufacture of a medicament for enhancing (for example, stimulating) an immune response in a subject.
- the subject is a mammal.
- the subject is a human.
- the term “enhancing an immune response” or its grammatical variations, means stimulating, evoking, increasing, improving, or augmenting any response of a mammal’s immune system.
- the immune response may be a cellular response (e.g. cell-mediated, such as cytotoxic T lymphocyte mediated) or a humoral response (e.g. antibody mediated response), and may be a primary or secondary immune response.
- Examples of enhancement of immune response include increased CD4 + helper T cell activity and generation of cytolytic T cells.
- the enhancement of immune response can be assessed using a number of in vitro or in vivo measurements known to those skilled in the art, including, but not limited to, cytotoxic T lymphocyte assays, release of cytokines (for example IL-2 production or IFN-y production), regression of tumors, survival of tumor bearing animals, antibody production, immune cell proliferation, expression of cell surface markers, and cytotoxicity.
- cytotoxic T lymphocyte assays release of cytokines (for example IL-2 production or IFN-y production), regression of tumors, survival of tumor bearing animals, antibody production, immune cell proliferation, expression of cell surface markers, and cytotoxicity.
- methods of the disclosure are useful to enhance the immune response by a mammal when compared to the immune response by an untreated mammal or a mammal not treated using the methods as disclosed herein.
- a MUC16-binding molecule may be used alone as a monotherapy, or may be used in combination with chemical therapies, radiotherapies, targeted therapies or cell immunotherapies etc.
- a MUC16-binding molecule (e.g., an anti-MUC16 antibody) may be used in combination with chemotherapies, including, for example, an anti-cancer agent, a cytotoxic agent or chemotherapeutic agent.
- anti-cancer agent or “anti-proliferative agent” means any agent that can be used to treat a cell proliferative disorder such as cancer, and includes, but is not limited to, cytotoxic agents, cytostatic agents, anti -angiogenic agents, debulking agents, chemotherapeutic agents, radiotherapy and radiotherapeutic agents, targeted anti-cancer agents, BRMs, therapeutic antibodies, cancer vaccines, cytokines, hormone therapies, radiation therapy and anti-metastatic agents and immunotherapeutic agents. It will be appreciated that, in some embodiments as discussed above, such anti-cancer agents may comprise conjugates and may be associated with the disclosed anti-MUC16 antibodies prior to administration.
- selected anti-cancer agents will be linked to the unpaired cysteines of the engineered antibodies to provide engineered conjugates (e.g., antibody-drug conjugates) as set forth herein. Accordingly, such engineered conjugates are expressly contemplated as being within the scope of the present disclosure.
- the disclosed anti-cancer agents will be given in combination with anti-MUC16 conjugates comprising a different therapeutic agent as set forth above.
- cytotoxic agent means a substance that is toxic to the cells and decreases or inhibits the function of cells and/or causes destruction of cells.
- the substance is a naturally occurring molecule derived from a living organism.
- cytotoxic agents include, but are not limited to, small molecule toxins or enzymatically active toxins of bacteria (e.g., Diptheria toxin, Pseudomonas endotoxin and exotoxin, Staphylococcal enterotoxin A), fungal (e.g., a-sarcin, restrictocin), plants (e.g., abrin, ricin, modeccin, viscumin, pokeweed anti-viral protein, saporin, gelonin, momoridin, trichosanthin, barley toxin, Aleurites fordii proteins, dianthin proteins, Phytolacca mericana proteins (PAPI, PAPII, and PAP-S), Momordica
- chemotherapeutic agent comprises a chemical compound that non-specifically decreases or inhibits the growth, proliferation, and/or survival of cancer cells (e.g., cytotoxic or cytostatic agents). Such chemical agents are often directed to intracellular processes necessary for cell growth or division, and are thus particularly effective against cancerous cells, which generally grow and divide rapidly. For example, vincristine depolymerizes microtubules, and thus inhibits cells from entering mitosis.
- chemotherapeutic agents can include any chemical agent that inhibits, or is designed to inhibit, a cancerous cell or a cell likely to become cancerous or generate tumorigenic progeny (e.g., TIC). Such agents are often administered, and are often most effective, in combination, e.g., in regimens such as CHOP or FOLFIRI.
- anti-cancer agents that may be used in combination with MUC16-binding molecules (e.g., anti-MUC16 antibodies) of the present disclosure (either as a component of a site specific conjugate or in an unconjugated state) include, but are not limited to, alkylating agents, alkyl sulfonates, aziridines, ethylenimines and methylamelamines, acetogenins, a camptothecin, bryostatin, callystatin, CC-1065, cryptophycins, dolastatin, duocarmycin, eleutherobin, pancratistatin, a sarcodictyin, spongistatin, nitrogen mustards, antibiotics, enediyne antibiotics, dynemicin, bisphosphonates, esperamicin, chromoprotein enediyne antiobiotic chromophores, aclacinomysins, actinomycin, authra
- anti-hormonal agents that act to regulate or inhibit hormone action on tumors
- anti-estrogens and selective estrogen receptor modulators aromatase inhibitors that inhibit the enzyme aromatase, which regulates estrogen production in the adrenal glands, and anti-androgens
- troxacitabine a 1,3- dioxolane nucleoside cytosine analog
- antisense oligonucleotides, ribozymes such as a VEGF expression inhibitor and a HER2 expression inhibitor
- vaccines PROLEUKIN® rIL-2; LURTOTECAN® topoisomerase 1 inhibitor; ABARELIX® rmRH; Vinorelbine and Esperamicins and pharmaceutically acceptable salts, acids or derivatives of any of the above.
- the present disclosure also provides for the combination of a MUC16-binding molecule with a radiotherapy (e.g., any mechanism for inducing DNA damage locally within tumor cells such as gamma-irradiation, X-rays, UV-irradiation, microwaves, electronic emissions and the like).
- a radiotherapy e.g., any mechanism for inducing DNA damage locally within tumor cells such as gamma-irradiation, X-rays, UV-irradiation, microwaves, electronic emissions and the like.
- Combination therapy using the directed delivery of radioisotopes to tumor cells is also contemplated, and the disclosed MUC16-binding molecules may be used in connection with a targeted anti-cancer agent or other targeting means.
- radiation therapy is administered in pulses over a period of time from about 1 to about 2 weeks.
- the radiation therapy may be administered to subjects having head and neck cancer for about 6 to 7 weeks.
- the radiation therapy may be administered as a single dose or as multiple, sequential doses.
- the disclosure provides in vitro and in vivo methods for detecting, diagnosing or monitoring proliferative disorders and methods of screening cells from a patient to identify tumor cells including tumorigenic cells.
- Such methods include identifying an individual having cancer for treatment or monitoring progression of a cancer, comprising contacting the patient or a sample obtained from a patient (either in vivo or in vitro) with an anti-MUC16 antibody as described herein and detecting presence or absence, or level of association, of the antibody to bound or free target molecules in the sample.
- the anti-MUC16 antibody will comprise a detectable label or reporter molecule as described herein.
- the association of an anti-MUC16 antibody with particular cells in the sample can denote that the sample may contain tumorigenic cells, thereby indicating that the individual having cancer may be effectively treated with an anti-MUC16 antibody as described herein.
- Samples can be analyzed by numerous assays, for example, radioimmunoassays, enzyme immunoassays (e.g. ELISA), competitive-binding assays, fluorescent immunoassays, immunoblot assays, Western Blot analysis and flow cytometry assays.
- Compatible in vivo theragnostic or diagnostic assays can comprise art recognized imaging or monitoring techniques, for example, magnetic resonance imaging, computerized tomography (e.g. CAT scan), positron tomography (e.g., PET scan), radiography, ultrasound, etc., as would be known by those skilled in the art.
- Pharmaceutical packs and kits can comprise art recognized imaging or monitoring techniques, for example, magnetic resonance imaging, computerized tomography (e.g. CAT scan), positron tomography (e.g., PET scan), radiography, ultrasound, etc., as would be known by those skilled in the art.
- Pharmaceutical packs and kits can comprise art recognized imaging or monitoring techniques, for example, magnetic resonance imaging, computerized tomography (e
- a unit dosage comprising one or more containers, comprising one or more doses of a MUC16-binding molecule are also provided.
- a unit dosage is provided wherein the unit dosage contains a predetermined amount of a composition comprising, for example, a MUC16-binding molecule, with or without one or more additional agents.
- such a unit dosage is supplied in single-use prefilled syringe for injection.
- the composition contained in the unit dosage may comprise saline, sucrose, or the like; a buffer, such as phosphate, or the like; and/or be formulated within a stable and effective pH range.
- a composition may be provided as a lyophilized powder that may be reconstituted upon addition of an appropriate liquid, for example, sterile water or saline solution.
- the composition comprises one or more substances that inhibit protein aggregation, including, but not limited to, sucrose and arginine. Any label on, or associated with, the container(s) indicates that the enclosed composition is used for treating the neoplastic disease condition of choice.
- kits for producing single-dose or multi-dose administration units of a MUC16-binding molecule and, optionally, one or more anti-cancer agents comprises a container and a label or package insert on or associated with the container.
- Suitable containers include, for example, bottles, vials, syringes, etc.
- the containers may be formed from a variety of materials such as glass or plastic and contain a pharmaceutically effective amount of the disclosed MUC16-binding molecules in a conjugated or unconjugated form.
- the container(s) comprise a sterile access port (for example the container may be an intravenous solution bag or a vial having a stopper pierceable by a hypodermic injection needle).
- kits will generally contain in a suitable container a pharmaceutically acceptable formulation of a MUC16-binding molecule in a conjugated or unconjugated form and, optionally, one or more anti-cancer agents in the same or different containers.
- the kits may also contain other pharmaceutically acceptable formulations, either for diagnosis or combined therapy.
- such kits may contain any one or more of a range of anti-cancer agents such as chemotherapeutic or radiotherapeutic drugs; anti-angiogenic agents; anti-metastatic agents; targeted anti-cancer agents; cytotoxic agents; and/or other anti-cancer agents.
- kits may have a single container that contains a MUC16-binding molecule, with or without additional components, or they may have distinct containers for each desired agent.
- a single solution may be pre-mixed, either in a molar equivalent combination, or with one component in excess of the other.
- the conjugates and any optional anti-cancer agent of the kit may be maintained separately within distinct containers prior to administration to a patient.
- the kits may also comprise a second/third container means for containing a sterile, pharmaceutically acceptable buffer or other diluents such as bacteriostatic water for injection (BWFI), phosphate-buffered saline (PBS), Ringer's solution and dextrose solution.
- BWFI bacteriostatic water for injection
- PBS phosphate-buffered saline
- Ringer's solution dextrose solution.
- the liquid solution is preferably an aqueous solution, for example, a sterile aqueous or saline solution.
- the components of the kit may be provided as dried powder(s).
- the powder can be reconstituted by the addition of a suitable solvent. It is envisioned that the solvent may also be provided in another container.
- kits may also contain a means by which to administer a MUC16-binding molecule and any optional components to a patient, e.g., one or more needles, I.V. bags or syringes or other such like apparatus, from which the formulation may be injected or introduced into the animal or applied to a diseased area of the body.
- the kits of the present disclosure will also typically include a means for containing the vials, or such like, and other component in close confinement for commercial sale, such as, e.g., injection or blow-molded plastic containers into which the desired vials and other apparatus are placed and retained.
- hProl includes human MUC16 55th & 56th SEA domain, and the juxtamembrane domain.
- cProl includes the cynomolgus counterpart of hProl.
- hPro2 includes human 56th SEA domain, and the juxtamembrane domain.
- cPro2 includes the cynomolgus counterpart of hPro2. The DNA segment was then sub-cloned into the pcDNA3.3 or pcDNA3.4 expression vector with 6x His or mFc (mIgG2a) at the C -terminal.
- the plasmids encoding the antigens were transfected into Expi293 cells at 100 mL, 200 mL, 300 mL and 500 mL scale. Cells were cultured for 5 days and the supernatant was collected for protein purification using HisTrap excel columns, Protein A column, and SEC column. The antigen concentration was detected by Nano Drop at 280 nm. The purity of the antigen was analyzed by SDS-PAGE and SEC-HPLC. The antigens were stored at -80 °C.
- W3XX106-cAb4 MUC16 binding monoclonal antibody prepared from the MUC16-binding arm of REGN4018
- W3XX106- BMK4 (9B11 antibody, WO2011119979 A2)
- W3XX106-BMK5 Parental antibody of DMUC5754A
- the amino acid sequences encoding the variable domains of W3XX106-cAb4 (WO2018/067331), W3XX106-BMK4 (WO2011/119979) and W3XX106-BMK5 (W02007/001851) were first codon optimized for mammalian expression and then synthesized by GENEWIZ (Su Zhou, CHINA).
- the DNA segments were then sub-cloned into pcDNA3.4 expression vectors with constant region of human IgGl .
- the plasmids containing VH and VL gene were co-transfected into Expi293 cells. Cells were cultured for 5 days and supernatant was collected for protein purification using Protein A column. The obtained antibodies were analyzed by SDS-PAGE and SEC-HPLC, and then stored at -80 °C.
- Human MUC16 56 th SEA domain expressing cell pool W3XX106-SK-OV-3.hPro2.pool was generated. Briefly, SK-OV-3 cells were transfected with pcDNA3.3 expression vector containing the DNA sequence of human 56 th SEA domain, transmembrane domain and intracellular domain, using Lipofectamine 2000 transfection kit according to manufacturer’s protocol. 48-72 hours post transfection, the transfected cells were cultured in medium containing 15 ⁇ g/mL Blasticidin for selection and tested for MUC16 56th SEA domain expression. The Cell pool was enriched by BD FACS MelodyTM cell sorter.
- Human MUC16 55th & 56th SEA domain expressing cell line W3XX106-SK-OV- 3.hProl.FL.A9 was generated. Briefly, SK-OV-3 cells were transfected with pcDNA3.3 expression vector containing the DNA sequence of human MUC16 55th & 56th SEA domain, transmembrane domain and intracellular domain, using Lipofectamine 2000 transfection kit according to manufacturer’s protocol. 48-72 hours post transfection, the transfected cells were cultured in medium containing 15 ⁇ g/mL Blasticidin for selection and tested for MUC16 55th & 56th SEA domain expression. The MUC16-expressing cell line was obtained by BD FACS MelodyTM cell sorter.
- Cynomolgus monkey MUC16 55th & 56th SEA domain expressing cell line W3XX106- SK-OV-3.cProl.FL.El was generated. Briefly, SK-OV-3 cells were transfected with pcDNA5 expression vector containing full-length of DNA sequence of cynomolgus monkey MUC16 55th & 56th SEA domain, transmembrane domain and intracellular domain, using Lipofectamine 2000 transfection kit according to manufacturer’s protocol, respectively. 72 hours post transfection, the transfected cells were cultured in medium containing 15 ⁇ g/mL Blasticidin for selection and tested for MUC16 expression. The MUC16-expressing cell line was obtained by BD FACSMelodyTM cell sorter and limiting dilution.
- Anti-MUC16 VHHs were generated by immunization of Camelidae animals and phage display technology. Briefly, Alpacas (Vicugna pacos) were subcutaneously immunized with W3XX106-hProl.ECD.mFc and W3XX106-hPro2.ECD.mFc. After immunization, peripheral blood was collected for construction of phage library displaying VHH fragments. After biopanning with corresponding target MUC16 proteins and W3XX106-SK-OV3-hProl cells, the positive VHH clones binding to MUC16 were selected. The animal information was showed in Table 2.
- the positive E. coli clones selected by target specific binding ELISA and FACS with E. coli supernatants were sent to Qingke (Shanghai, China) for nucleotide sequencing of VHH gene.
- W306106-P11R2-1B8 clone was selected based on the unique sequence and good binding data as shown in Table 3.
- the VH domain sequences of W306106-P11R2-1B8 are aligned to the human germline sequence repertoires of the VH at IMGT.
- the human germline sequence of the VH domain with the least number of amino acid differences in framework with respect to the VH domain sequence of W306106-P11R2-1B8 was selected as the humanization template of the VH domain.
- CDRs of W306106-P11R2-1B8 VHH domain were grafted into the framework of the humanization template to constitute the germlined VHH domain sequence.
- DNA sequence of humanization variants were subcloned into modified pcDNA3.4 expression vectors with constant region of human IgGl.
- the plasmids were transfected into Expi293 cells. Cells were cultured for 5 days and supernatant was collected for SPR ranking assay or protein purification. The purified antibodies were analyzed by SDS-PAGE and SEC, and then stored at -80 °C.
- the W306106-P11R2-1B8 was converted to VHH-Fc (hlgGl) fusion antibody. Briefly, the gene of W306106-P11R2-1B8 was PCR amplified from the pET-bac vectors using VHH-specific cloning primers containing appropriate restriction sites then cloned by fusion into a modified human hlgGl expression pcDNA3.3 vector to create corresponding clone of VHH-Fc (hlgGl) chimeric antibody. Expi293 cells were transiently transfected with the vector for antibody expression. The cell culture supernatant containing antibody was harvested and purified using Protein A chromatography. The generated antibody was named as “W306106-P11R2-1B8- ulgGl”. The obtained antibody was analyzed by SDS-PAGE and HPLC-SEC, and then stored at -80 °C.
- Tm melting temperature
- Tml Tm2
- Tm2 Tm2
- Data collection and Tm calculation were conducted automatically by the QuantStudio® Real Time PCR software (vl.3). The results were shown in Figure 2 and Table 5 below.
- AC-SINS Affinity-Capture Self-Interaction Nanoparticle Spectroscopy
- the mixture was filtered through a 0.22 pm PVDF membrane, and 1/10 of the starting volume of 50 mM PB, pH 7.0 buffer was used to elute the coated AuNP particles from the membrane to obtain lOx AuNP solution. All antibodies were diluted to 0.1 mg/mL before testing. 90 ⁇ L test antibody solution (0.1 mg/mL) was mixed with 20 ⁇ L 10 x AuNP and incubated at RT for 2 hours in a 96-well polypropylene plate. After incubation, 100 ⁇ L of the antibody- AuNP mixture was transferred into a 384-well polystyrene UV transparent plate. Absorbance data were collected from 510 to 570 nm at an increment of 1 nm. AXmax value was calculated by subtraction the max absorbance value of samples with that of PBS.
- BV particles were obtained by infecting Sf9 insect cells with a recombinant expressing green fluorescent protein (Bac-to-Bac, Thermofisher). Infected cultures were incubated for 40 hours at 27 °C under agitation (200 rpms), harvested and cells removed by centrifugation at 300 g for 5 minutes. The virus in the supernatant was pelleted by centrifugation at 300 g for 4 hours at 4 °C, resuspended in PBS buffer layered on a 4 mL 35 % (w/v) sucrose cushion in PBS and centrifuged at 25000 rpm for 4 hours at 4 °C. The supernatant with debris was discarded, the virus pellet gently rinsed once with PBS, resuspended in 1.2 mL of PBS with protease inhibitor cocktail (Roche) and stored at 4 °C for up to 4 months.
- Bac-to-Bac Thermofisher
- DNA sequence encoding the variable region of VHH antibody was subcloned into modified pcDNA3.4 expression vector with constant region of human IgGl.
- the plasmid was transfected into Expi293 cells. Cells were cultured for 5 days and supernatant was collected for protein purification using Protein A column. The obtained antibody was analyzed by SDS-PAGE and SEC, and then stored at -80 °C.
- the k off ranking is performed on a Biacore 8K surface plasmon resonance (SPR) equipment.
- SPR surface plasmon resonance
- CM5 sensor chips are first activated by 400 mM EDC and 100 mM NHS (GE) for 420 sec at the flow rate of 10 ⁇ L /minute.
- 30 ⁇ g/mL of anti-human Fc IgG (Jackson) in 10 mM NaAc (pH 4.5) is then injected to channel 1 to 8 at the flow rate of 10 ⁇ L/minute for 420 sec.
- the chips are then deactivated by 1 M ethanolamine-HCl (GE) at the flow rate of 10 ⁇ L/minute for 420 sec.
- the sample was centrifuged at 12000 rpm for 3 minutes at 4 °C, filtered by using a 0.1 pm filter, concentrated to 5-6 mg/mL and observed the appearance.
- the concentration of each sample was measured by Nanodrop2000 with 2 ⁇ L sample for 3 times and then diluted to 1 mg/mL by the sample storage buffer (PBS).
- Eight tubes of 300 ⁇ L antibody solution were prepared for each sample. Each tube was placed in its respective temperature conditions (either 4 °C or 40 °C or -80 °C) and stored for 14 days, or freezed (-80 °C)/thawed (25 °C) for 3 and 5 cycles. After the stress treatment, samples were centrifuged at 12,000 rpm for 3 minutes at 4 °C and observed visually. The protein concentration was measured by Nanodrop2000 and the data was recorded. Purity of each antibody was detected by Agilent 1260 Infinity II system with TSKgel G3000SWXL column.
- the freeze/thaw cycling test result summary of W306106-P1 !R2-lB8-z6-uIgG1 was shown in Table 11.
- the Accelerated thermal stability test result summary of W306106-P11R2-1B8-Z6- ulgGl was shown in Table 12.
- the SEC-HPLC profiles ofW306106-PHR2-lB8-z6-uIgG1 after 5 Freeze/Thaw cycles and W306106-P1 lR2-lB8-z6-uIgG1 after 14 days in 40 °C was showed in Figure 5 and Figure 6.
- T m melting temperature
- Tml value of W306106-P1 !R2-lB8-z6-uIgG1 is in the normal range.
- the kD measurement was investigated using DynaPro Plate Reader III (Wyatt Technology).
- sample preparation process the appearance of samples was observed at thawing, filtration and concentration by visual inspection. The purity of a dose point (15 mg/mL) will be tested after concentration.
- Samples were first filtered with 0.1 pm filter. Samples were then concentrated to over 20 mg/mL and diluted with corresponding buffer to a final concentration at 2.5, 5, 10, 15, and 20 mg/mL. 7.5 ⁇ L sample solution was then added to 1536 well microplate. The plate was sealed with the Clear Seal Film, and centrifuged at 3,000 rpm for 5 minutes at 4 °C to let the sample down to the bottom of the well. Each sample was tested in duplicate wells.
- the plate was put into the corresponding position and data collection was performed by the DYNAMICS operation software (v7.8.1.3). 5 acquisitions were collected for each protein sample while each acquisition time was 5 s. For each measurement, the diffusion coefficient was determined and plotted against protein concentration. kD values were calculated automatically by the software.
- AC-SINS Affinity-Capture Self-Interaction Nanoparticle Spectroscopy
- the mixture was filtered through a 0.22 pm PVDF membrane, and 1/ 10 of the starting volume of 50 mM PB, pH 7.0 buffer was used to elute the coated AuNP particles from the membrane to obtain 10 x AuNP solution.
- All antibodies were diluted to 0.1 mg/mL before testing.
- 90 ⁇ L of test antibody solution (0.1 mg/mL) was mixed with 20 ⁇ L 10 x AuNP and incubated at RT for 2 hours in a 96-well polypropylene plate. After incubation, 100 ⁇ L of the antibody- AuNP mixture was transferred into a 384-well polystyrene UV transparent plate.
- Absorbance data were collected from 510 to 570 nm at an increment of 1 nm. AXmax value was calculated by subtraction the max absorbance value of samples with that of PBS.
- Antibodies with low Almax value probably have low self-interaction propensity as showed in Table 15.
- ELISA assay 96-well high binding plates (Nunc-Immuno Plate, Thermo Scientific) were coated with 2 ⁇ g/mL HIS-tagged antigen and blocked with 2 % BSA-PBS. 100 ⁇ L antibodies (10 ⁇ g/mL) were added into the antigen-coated well and incubated at room temperature for 2 hours, and the binding of antibodies to the immobilized antigens on the plate was measured using Goat Anti-Human IgG-Fc-HPR labelled antibody. The HRP signal was detected by adding TMB peroxidase substrate, and the reaction was stopped after 12 minutes using 2 M HC1.
- FACS assay cells were transferred into 96-well U-bottom plates (BD) at a density of 1 x 10 5 cells/well and centrifuged before removing the supernatant. 100 ⁇ L antibodies (10 ⁇ g/mL) were used to re-suspend cells and incubated at 4 °C for 1 hour. The secondary antibody, PE-conjugated Goat Anti-Human IgG Fc fragment was diluted to 5 ⁇ g/mL, then added to the re-suspended cells and incubated at 4 °C for 30 minutes. Washing steps were performed twice followed by centrifugation at x 200 g at 4 °C. Finally, the cells were re-suspended in 1% BSA-1 x PBS and fluorescence intensity was measured by flow cytometry (BD Canto II) and analyzed by FlowJo.
- BVPs were incubated on ELISA plates by adding 100 mL of 1 % BVP stock (in 50 mM sodium carbonate, pH 9.6) per well and incubated at 4 °C for 24 hours. The next day, unbound BVPs were aspirated from the wells. All remaining steps were performed at room temperature. 100 ⁇ L of blocking buffer (PBS with 0.5 % BSA) was added to the 96-well and incubated for 1 hour prior to three washes with 300 mL of PBS. Next, 100 ⁇ L of 1 pM primary antibodies (i.e. test antibodies) in blocking buffer was added to the wells and incubated for 1 hour followed by six washes with 100 ⁇ L of PBS.
- blocking buffer PBS with 0.5 % BSA
- DNA sequence encoding the variable region of VHH antibodies were subcloned into modified pcDNA3.4 expression vectors.
- the plasmids were transfected into Expi293 cells. Cells were cultured for 5 days and supernatant was collected for protein purification using Protein A3 column. The obtained antibodies were analyzed by SDS-PAGE and SEC-HPLC, and then stored at -80 °C.
- Tm melting temperature
- Tml value was in the normal range as showed in Table 22.
- the DSF profile was showed in Figure 11.
- Hydrophobicity property of antibody was detected by HPLC 1260 Infinity II system (Agilent TechnologiesTM) with TSKgel butyl-NPR column. Each sample was diluted to 0.5 mg/mL with PBS buffer and 20 ⁇ L diluted sample was injected into the column, and seperated with a flow rate of 0.5 mL/minute for 61 minutes.
- the running buffer was 25 mM sodium phosphate, pH 7.0 (Buffer A) and 25 mM sodium phosphate, 1.5 M (NH4)2SO4, pH 7.0 (Buffer D).
- the running gradient was 100 % to 0 % Buffer D and 0 % to 100 % Buffer A from 3 to 53 minutes.
- the peak retention was detected with UV light of the wavelength at 280 nm and 230 nm.
- the retention time was analyzed with HIC-HPLC analysis method to integrate all peak areas from 20 minutes to 40 minutes.
- the operation and analysis software were the OpenLab CDS Workstation (v2.6.0.691).
- Retention time represents the hydrophobicity degree of the antibody, and long retention time indicates high potential hydrophobicity.
- Table 24 and Figure 13 indicated that the HIC- HPLC profile of W306106-P11R2-1B8-Z6 falls in the normal range.
- the concentration of each sample was measured by Nanodrop2000 with 2 ⁇ L sample for 3 times and then diluted to 1 mg/mL by the sample storage buffer (PBS).
- Eight tubes of 300 ⁇ L antibody solution was prepared for each sample. Placed each tube in its respective temperature conditions (either 4 °C or 40 °C or -80 °C) and stored for 14 days, or freezed (-80 °C)/thawed (25 °C) for 3 and 5 cycles. After the stress treatment, samples were centrifuged at 12,000 rpms for 3 minutes and observed visually. Measured the protein concentration by Nanodrop2000 and recorded the data. Purity of each antibody was detected by Agilent 1260 Infinity II system with TSKgel G3000SWXL column.
- Example 6 In vitro Characterization of W306106-PllR2-lB8-uIgGl (pre-humanization), W306106-PllR2-lB8-z6-uIgGl (after humanization) and W306106-PllR2-lB8-z6 (VHH after humanization)
- Binding of anti-MUC16 antibodies to MUC 16 expressing cells was determined by flow cytometry.
- W3XX106-SK-OV-3.hProl.FL.A9 cell expressing the human MUC 1655 th & 56 th SEA domain and a selected tumor cell line OVCAR-3 (1 x 10 5 cells/well) expressing high level of human MUC 16 were harvested by using or 0.25 % Trypsin-EDTA (1 x).
- the cells were incubated with serial diluted antibodies (for W3XX106-SK-OV-3.hProl.FL.A9, starting at 400 nM, 4-fold dilution to 0.00038 nM, for OVCAR-3, starting at 500 nM, 5-fold dilution to 0.0064 nM) in a volume of 100 ⁇ L for 1 hour at 4 °C.
- serial diluted antibodies for W3XX106-SK-OV-3.hProl.FL.A9, starting at 400 nM, 4-fold dilution to 0.00038 nM, for OVCAR-3, starting at 500 nM, 5-fold dilution to 0.0064 nM
- the parental of DMUC5754A (Genentech) and MUC16 binding arm of REGN-4018 (Regeneron) were used as positive controls, and human IgGl isotype control antibody W332-1.80.12.xAb.hIgGl was used as the negative control.
- W306106-P1 !R2-lB8-uIgG1 on W3XX106-SK-OV-3.hProl.FL.A9 and W3XX106-SK-OV-3.cProl.FL.El cells were shown in Figure 17A-17B, respectively.
- the EC50 and Max MFI were shown in Table 28.
- W306106-P 11R2- lB8-uIgG1 showed strong binding to the cell surface human and cynomolgus monkey MUC 16 55 th & 56 th SEA domain with EC50 of 2.71 nM and 1.77 nM respectively.
- the binding activity were comparable to MUC 16 binding arm of REGN4018 (Regeneron).
- the parental of DMUC5754A (Genentech) showed no binding to the cell surface human or cynomolgus monkey MUC 16 55 th & 56 th SEA domain.
- REGN4018 (Regeneron) with EC50 of 1.33 nM and 0.26 nM respectively.
- Binding of anti-MUC16 antibodies to MUC16 expressing cells was determined by flow cytometry.
- W3XX106-SK-OV-3.hProl.FL.A9 cell expressing the human MUC1655 th & 56 th SEA domain and four selected tumor cell lines including OVCAR-3, HDQ-P1, SK-OV-3 and A375 (1 x 10 5 cells/well) expressing different level of human MUC16 were harvested by using or 0.25 %
- OVCAR-3 and HDQ-P1 starting at 500 nM, 5-fold dilution to 0.00026 nM) in a volume of 100 ⁇ L for 1 hour at 4 °C.
- the parental of DMUC5754A (Genentech) was used as the positive control, and human IgGl isotype control antibody W332-1.80.12.xAb.hIgGl was used as the negative control.
- DMUC5754A (Genentech) showed no binding to the cell surface human or cynomolgus monkey MUC16 55 th & 56 th SEA domain.
- Table 30 Human and cynomolgus monkey MUC16 55 th & 56 th SEA domain FACS binding The binding results of W306106-P1 !R2-lB8-z6-uIgG1 on four selected human tumor cell lines expressing different level of human MUC16 were shown in Figure 21 including OVCAR-3 (MUC16 high ), HDQ-P1 (MUC16 medium ), SK-OV-3 (MUC16 low ) and A375 (MUC16 negative ).
- OVCAR-3 MUC16 high
- HDQ-P1 MUC16 medium
- SK-OV-3 MUC16 low
- A375 MUC16 negative
- W306106-Pl lR2-lB8-z6-uIgG1 showed specifically binding to the cell surface human MUC16 of OVCAR-3 (MUC16 high ), HDQ-P1 (MUC16 medium ) and SK-OV-3 (MUC16 low ).
- the binding activity of W306106-P11R2-1B8-Z6- ulgGl to OVCAR-3 (MUC16 hlgh ) was slightly weaker than the parental of DMUC5754A (Genentech), and the binding activity to HDQ-P1 (MUC16 medlum ) and SK-OV-3 (MUC16 low ) were comparable to the parental of DMUC5754A (Genentech).
- Both W306106-Pl lR2-lB8-z6-uIgG1 and the parental of DMUC5754A (Genentech) showed no binding to A375 (MUC16 negative ).
- Binding of anti-MUC16 antibodies to MUC16 expressing cells was determined by flow cytometry.
- W3XX106-SK-OV-3.hProl.FL.A9 cell expressing the human MUC1655 th & 56 th SEA domain and two selected tumor cell lines, OVCAR-3 and A375 (1 x 10 5 cells/well) expressing different level of human MUC16 were harvested by using or 0.25 % Trypsin-EDTA (1 x).
- the cells were incubated with serial diluted antibodies (for W3XX106-SK-OV-3.hProl.FL.A9 and A375, starting at 600 nM, 4-fold dilution to 0.0023 nM, for OVCAR-3, starting at 500 nM, 5-fold dilution to 0.0064 nM) in a volume of 100 ⁇ L for 1 hour at 4 °C.
- the antibody W306108-P24R2- IGl.His was used as the negative control.
- W306106-P11R2-1B8-Z6 on W3XX106-SK-OV-3.hProl.FL.A9 and W3XX106-SK-OV-3.cProl.FL.El cells were shown in Figure 19A-19B, respectively.
- the EC50 and Max MFI were shown in Table 32.
- W306106-Pl lR2-lB8-z6 showed strong binding to the cell surface human and cynomolgus monkey MUC16 55 th & 56 th SEA domain and the EC50 of binding to W3XX106-SK-OV-3.hProl.FL.A9 was 8.67 nM.
- W306106-P11R2-1B8-Z6 The binding results of W306106-P11R2-1B8-Z6 on two selected human tumor cell lines expressing different level of human MUC16 were shown in Figure 22 including OVCAR-3 (MUC16 high ) and A375 (MUC16 negative ).
- the EC50 and Max MFI were shown in Table 33.
- W306106-P11R2-1B8-Z6 showed specifically binding to the cell surface human MUC16 of OVCAR-3 (MUC16 higb ) with an EC50 of 3.65 nM and no binding to A375 (MUC16 negative ).
- W3XX106-SK-OV-3.cProl.FL.El cells expressing the cynomolgus monkey MUC16 55 th & 56 th SEA domain were harvested by using 0.25 % Trypsin-EDTA (1 x). Then the cells were incubated with serial diluted antibodies (starting at 400 nM, 4- fold dilution to 0.000095 nM) in a volume of 100 pL for 1 hour at 4 °C.
- the parental of DMUC5754A (Genentech) and MUC16 binding arm of REGN-4018 (Regeneron) were used as positive controls, and human IgGl isotype control antibody W332-1.80.12.xAb.hIgGl was used as the negative control.
- MFI mean fluorescence intensity
- W3XX106-SK-OV-3.cProl.FL.El cells expressing the cynomolgus monkey MUC16 55 th & 56 th SEA domain were harvested by using 0.25 % Trypsin-EDTA (1 x). Then the cells were incubated with serial diluted antibodies (starting at 500 nM, 5-fold dilution to 0.000010 nM) in a volume of 100 pL for 1 hour at 4 °C.
- the parental of DMUC5754A (Genentech) was used as the positive control, and human IgGl isotype control antibody W332-1.80.12.xAb.hIgGl was used as the negative control.
- W3XX106-SK-OV-3.cProl.FL.El cells expressing the cynomolgus monkey MUC16 55 th & 56 th SEA domain were harvested by using 0.25 % Trypsin-EDTA (1 x). Then the cells were incubated with serial diluted antibodies (starting at 600 nM, 4-fold dilution to 0.0023 nM) in a volume of 100 ⁇ L for 1 hour at 4 °C.
- the antibody W306108-P24R2-lGl.His was used as the negative control.
- ELISA plates were coated with human MUC 1656 th SEA domain or CA125 protein (2 ⁇ g/mL, 100 ⁇ L/well) in coating buffer and incubated at 4 °C overnight. The next day, the coating antigen buffer was removed and the ELISA plates were washed 1 time with washing buffer 1 x PBST (300 ⁇ L/wash). The ELISA plates were blocked with blocking buffer (100 % casein), 200 ⁇ L/well, and incubated at room temperature for 1 hour. Then, the ELISA plates were washed 3 times with washing buffer, 300 ⁇ L /well.
- the serial diluted antibodies in 50 % casein (100 ⁇ L/well, starting at 500 nM, 4-fold dilution to 0.00012 nM) were added and incubated at room temperature for 2 hours.
- the ELISA plates were washed 3 times with washing buffer, 300 ⁇ L/wash.
- the Goat anti-Human IgG-Fc Fragment Cross-Adsorbed Antibody (1 : 5000, HRP conjugated, Ab2) in 50 % casein was added, 100 ⁇ L /well and incubated at room temperature for 1 hour. After washing the ELISA plates for 6 times with washing buffer, 300 ⁇ L/well, TMB substrate was added with 100 ⁇ L/well, and the ELISA plates were incubated at room temperature for 10 minutes in the dark. The stop solution (2 M HC1) was added with 100 ⁇ L/well to stop further color developing. Finally, the ELISA plates were detected by microplate reader M5e at 450 nm and 540 nm.
- W306106-Pl lR2-lB8-uIgG1 showed good binding to recombinant human 56 th SEA domain MUC16 but showed no binding to CAI 25 (soluble MUC16) ( Figure 23).
- Parental of DMUC5754A bound to CA125 but not human 56th SEA domain MUC16.
- W306106-P11R2-lB8-uIgG1 could bind to human 56th SEA domain MUC16 and was not affected by CA125 (soluble MUC16).
- the EC50 and Max OD are shown in Table 34.
- ELISA plates were coated with human MUC16 55 th & 56 th protein, human MUC16 56 th protein or CAI 25 protein (2 ⁇ g/mL, 100 ⁇ L/well) in coating buffer and incubated at 4 °C overnight. The next day, the coating antigen buffer was removed and the ELISA plates were washed 1 time with washing buffer 1 x PBST (300 ⁇ L/wash). The ELISA plates were blocked with blocking buffer (100 % casein), 200 ⁇ L /well, and incubated at room temperature for 1 hour. Then, the ELISA plates were washed 3 times with washing buffer, 300 ⁇ L/well.
- serial diluted antibodies in 50 % casein (100 ⁇ L/well, starting at 400 nM, 4-fold dilution to 0.0015 nM or starting at 400 nM, 4-fold dilution to 0.00010 nM) were added and incubated at room temperature for 2 hours.
- the parental of DMUC5754A was used as the positive controls, and the W332-1.80.12.xAb.hIgGl isotype control antibody was used as negative control.
- the ELISA plates were washed 3 times with washing buffer, 300 ⁇ L/wash.
- the Goat anti-Human IgG-Fc Fragment Cross- Adsorbed Antibody (1:5000, HRP conjugated, Ab2) in 50 % casein was added, 100 ⁇ L /well and incubated at room temperature for 1 hour. After washing the ELISA plates for 6 times with washing buffer, 300 ⁇ L/well, TMB substrate was added with 100 ⁇ L/well, and the ELISA plates were incubated at room temperature for 10 minutes in the dark. The stop solution (2 M HC1) was added with 100 ⁇ L/well to stop further color developing. Finally, the ELISA plates were detected by microplate reader M5e at 450 nm and 540 nm.
- W306106-P1 lR2-lB8-z6-uIgG1 showed good binding to recombinant human 56th SEA domain MUC16 and human 55 th & 56 th SEA domain MUC16, but showed no binding to CA125 (soluble MUC16) ( Figure 24).
- Parental of DMUC5754A bound to CA125 but not human 56 th SEA domain MUC16 and human 55 th & 56 th SEA domain MUC16.
- W306106-P1 lR2-lB8-z6-uIgG1 could bind to human 56th SEA domain MUC16 and was not affected by CA125 (soluble MUC16).
- the EC50 and Max OD are shown in Table 35.
- ELISA plates were coated with human MUC16 55 th & 56 th SEA domain, human MUC16 56 th SEA domain or CA125 protein (2 ⁇ g/mL, 100 ⁇ L/well) in coating buffer and incubated at 4 °C overnight. The next day, the coating antigen buffer was removed and the ELISA plates were washed 1 time with washing buffer 1 x PBST (300 ⁇ L/wash). The ELISA plates were blocked with blocking buffer (100 % casein), 200 ⁇ L/well, and incubated at room temperature for 1 hour. Then, the ELISA plates were washed 3 times with washing buffer, 300 ⁇ L/well.
- the serial diluted antibodies in 50 % casein (100 ⁇ L/well, starting at 400 nM, 5-fold dilution to 0.00020 nM) were added and incubated at room temperature for 2 hours.
- the W306108-P24R2-lGl.His isotype control antibody was used as negative control.
- the ELISA plates were washed 3 times with washing buffer, 300 ⁇ L/wash.
- the MonoRabTM Rabbit Anti-Camelid VHH Cocktail Antibody (1:5000, HRP conjugated, Ab2) in 50 % casein was added, 100 ⁇ L/well and incubated at room temperature for 1 hour.
- W306106-P11R2-1B8-Z6 showed good binding to recombinant human 56 th SEA domain MUC16 and human 55 th & 56 th SEA domain MUC16, but showed no binding to CA125 (soluble MUC16) (Figure 25). This suggested that W306106-P11R2-1B8-Z6 could bind to human 56 th SEA domain MUC16 and was not affected by CA125 (soluble MUC16).
- the EC50 and Max OD are shown in Table 36.
- ELISA plates were coated with cynomolgus monkey and mouse MUC16 55 th & 56 th SEA domain (2 ⁇ g/mL, 100 ⁇ L/well) in coating buffer and incubated at 4 °C overnight. The next day, the coating antigen buffer was removed and the ELISA plates were washed 1 time with washing buffer 1 x PBST (300 ⁇ L/wash). The ELISA plates were blocked with blocking buffer (100 % casein), 200 ⁇ L/well, and incubated at room temperature for 1 hour. Then, the ELISA plates were washed 3 times with washing buffer, 300 pL/well. The serial diluted antibodies in 50 % casein
- Antibody (1:5000, HRP conjugated, Ab2) in 50 % casein was added, 100 pL/well and incubated at room temperature for 1 hour. After washing the ELISA plates for 6 times with washing buffer,
- TMB substrate was added with 100 pL/well, and the ELISA plates were incubated at room temperature for 10 minutes in the dark.
- the stop solution (2 M HC1) was added with 100 pL/well to stop further color developing.
- the ELISA plates were detected by microplate reader M5e at 450 nm and 540 nm.
- ELISA plates were coated with cynomolgus monkey and mouse MUC 16 55 th & 56 th SEA domain (2 pg/mL, 100 pL/well) in coating buffer and incubated at 4 °C overnight. The next day, the coating antigen buffer was removed and the ELISA plates were washed 1 time with washing buffer 1 x PBST (300 pL/wash). The ELISA plates were blocked with blocking buffer (100 % casein), 200 pL/well, and incubated at room temperature for 1 hour. Then, the ELISA plates were washed 3 times with washing buffer, 300 ⁇ L/well.
- the serial diluted antibodies in 50 % casein (100 ⁇ L/well, starting at 400 nM, 5-fold dilution to 0.00020 nM) were added and incubated at room temperature for 2 hours.
- the W306108-P24R2-lGl.His isotype control antibody was used as negative control.
- the ELISA plates were washed 3 times with washing buffer, 300 ⁇ L/wash.
- the MonoRabTM Rabbit Anti-Camelid VHH Cocktail Antibody (1:5000, HRP conjugated, Ab2) in 50 % casein was added, 100 ⁇ L /well and incubated at room temperature for 1 hour.
- W306106-P1 lR2-lB8-z6 showed good binding to recombinant cynomolgus monkey 55 th & 56 th SEA domain MUC16, but showed no binding to mouse 55 th & 56 th SEA domain MUC16
- the activator was prepared by mixing 400 mM EDC (l-Ethyl-3 -(3 -Dimethylaminopropyl) Carbodiimide) and 100 mM NHS (N-Hydroxy Succinimide) immediately prior to injection.
- the CM5 sensor chip was activated for 420 s at a flow rate of 10 ⁇ L/minute with the mixture.
- 30 ⁇ g/mL of anti-human IgG Fc antibody in 10 mM NaAc (pH 4.5) was then injected to channel 1-8 for 420 s at a flow rate of 10 ⁇ L/minute.
- the chip was deactivated by 1 M ethanolamine-HCl for 420 s at a flow rate of 10 ⁇ L/minute.
- 600-fold diluted NC and the same fold diluted W306106-Pl lR2-lB8-uIgG1 in running buffer lx HBS-EP+ (0.01 M HEPES, 0.15 M NaCl, 3 mM EDTA, 0.05 % surfactant P20, pH 7.4) were respectively injected to Fcl and Fc2 at a flow rate of 10 ⁇ L/min for 30 s.
- 500-fold diluted NC and the same fold diluted W306106-P1 lR3-lB8-uIgG1 in running buffer lx HBS-EP+ were respectively injected to Fcl andFc2 at a flow rate of 10 ⁇ L/minute for 30 s.
- k d labeled with * is approaching or outside the limits that can be measured by the instrument.
- the kd and KD are only for reference.
- the activator was prepared by mixing 400 mM EDC (l-Ethyl-3 -(3 -Dimethylaminopropyl) Carbodiimide) and 100 mM NHS (N-Hydroxy Succinimide) immediately prior to injection.
- the CM5 sensor chip was activated for 420 s at a flow rate of 10 ⁇ L /minute with the mixture.
- 30 ⁇ g/mL of anti-human IgG Fc antibody in 10 mM NaAc (pH 4.5) was then injected to channel 1-8 for 420 s at a flow rate of 10 ⁇ L/minute.
- the chip was deactivated by 1 M ethanolamine-HCl for 420 s at a flow rate of 10 ⁇ L /minute.
- kd labeled with * is approaching or outside the limits that can be measured by the instrument.
- the kd and KD are only for reference.
- the 96-well plate was coated with 2.5 ⁇ g/cm 2 Poly-D-Lysine (PDL) at 37 °C for 2 hours (1 : 1000), 100 ⁇ L/well.
- PDL Poly-D-Lysine
- Cells in the T75 flask were washed by PBS and detached by Versene. The cells were washed once with culture medium and resuspended in an appropriate volume of culture medium to the concentration of 3 x 10 5 cells/mL with pipettes. Then 100 ⁇ L cells suspension was aliquoted to each well of 96-well plate with multi-channel pipettes and incubated overnight. The second day, the medium was fetched and the cells were washed once with 1 % BSA.
- PDL Poly-D-Lysine
- the serial diluted Abs in 1 % BSA were added to the cell plates and incubated at 4 °C for 2 hours. After incubation, the cells were washed once by 1 % BSA, 200 ⁇ L /well. Goat anti-human IgG PE (1: 200 diluted in 1 % BSA, 100 ⁇ L/well) was added and incubated for 1 hour at 4 °C in the dark. After washing the cells with lx PBS / 1 % BSA, 1 % BSA was added to each well (100 ⁇ L/well) and the plate was incubated for 2 hours at 37 °C. And 1 % BSA was discarded and the cells were quenched at 4 °C for 4 minutes.
- the plate was washed once with PBS, 150 ⁇ L/well. Hoechst (1: 2000 dilution in PBS, 100 ⁇ L/well) was added and incubated for 20 minutes at room temperature. The plate was washed once with 1 x PBS and the cells were fixed with 4 % PFA for 15 minutes at room temperature and the plate was saved at 4 °C for further analysis. The plate was read by Oppretta.
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Life Sciences & Earth Sciences (AREA)
- Immunology (AREA)
- Medicinal Chemistry (AREA)
- General Health & Medical Sciences (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Molecular Biology (AREA)
- Biophysics (AREA)
- Biochemistry (AREA)
- Genetics & Genomics (AREA)
- Cell Biology (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Pregnancy & Childbirth (AREA)
- Reproductive Health (AREA)
- General Chemical & Material Sciences (AREA)
- Gynecology & Obstetrics (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Pharmacology & Pharmacy (AREA)
- Animal Behavior & Ethology (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Peptides Or Proteins (AREA)
Abstract
Provided in the present disclosure are MUC16-binding molecules, including anti-MUC16 antibodies, especially VHH-comprising antibodies, a method of preparing the same, and the use of the antibodies.
Description
ANTI-MUC16 ANTIBODIES AND USES THEREOF
SEQUENCE LISTING
The instant application contains a sequence listing which is hereby incorporated by reference in its entirety.
FIELD
This application generally relates to antibodies. More specifically, the application relates to monoclonal antibodies against MUC16, especially VHH-comprising antibodies, a method of preparing the same, and the use of the antibodies.
BACKGROUND
Mucin 16 (MUC16, previously known as carcinoma antigen 125, CA125) was first identified by Bast et al. in 1981, and its cDNA sequence was later found corresponded to MUC16, a mucin protein [1-2], MUC16 is a heavily glycosylated single pass transmembrane protein with a molecular weight around 3000 - 5000 KD [2], It is the largest mucin protein, consisting of multiple domains including an extracellular N-terminal domain, a large tandem repeat domain which is interspersed with sea urchin sperm, enterokinase and agrin (SEA) domain, and a C-terminal domain that comprises the transmembrane region and a short cytoplasmic tail [3], MUC16 has 56 SEA domains, where the penultimate SEA (55th) domain has a conserved cleavage site [4], MUC16 could shed from the cell surface and release into the bloodstream to become soluble MUC16 (CA125) through proteolytic cleavage, and a retained ectodomain between the cell membrane and the cleavage site remains on the cell surface [4],
Under normal physiological conditions, MUC16 is only expressed at low levels in a few tissues including the respiratory tract and the female reproductive tract, particularly in glands and epithelial cells [5], The expression level of MUC16 is significantly higher in a spectrum of human cancers, including ovarian cancer, endometrial cancer, pancreatic cancer, than that of normal tissues. The shedding domain termed CAI 25 is a poor prognostic and diagnostic serum marker for ovarian cancer. CAI 25 is the most widely used ovarian tumor marker and often considered as the “gold standard’. [6, 7], Abnormal CAI 25 level (> 35 U/mL) was observed in 99 % of serous cancer patients rated from I to IV in the FIGO (International Federation of Gynecologists and Obstetricians) stages. Serum CA125 levels could increase up to 10 folds and exceed 2000 U/mL in many serous ovarian cancer patients at FIGO stage IV compared to stage I [8], In addition to ovarian cancer, elevated expression of CAI 25 is strongly correlated with poorer prognosis in multiple cancers [9], The limited expression of MUC16 on normal human tissues and its high expression in many common cancers makes it an attractive target for cancer therapy.
While several MUC 16 -targeting therapeutic antibodies including oregovomab and abagovomab have been tested in clinical trials, only limited efficacy has been achieved in cancer patients [10, 11], A potential shortcoming of the several described antibody-based therapeutics is that they target the membrane distal region of MUC 16, and consequently, due to the high levels of circulating CAI 25 in cancer patients and the CAI 25 antigen sink effect, the target cell bound antibody is significantly reduced and the tumor killing effect would be therefore compromised [4]. Avoid binding to the soluble CAI 25 in the blood circulation might be critical in the development of therapeutic antibodies that target MUC 16-positive cancers.
There remains a need to develop novel anti-MUC16 antibodies, preferentially those binding to MUC16 ectodomain on the cell membrane over shedding CA125, to minimize the antigen sink effect from the high levels of soluble CA125 in cancer patients.
SUMMARY
The present disclosure is directed to compounds, methods, compositions and articles of manufacture that provide MUC16-binding molecules with improved efficacy. The benefits provided by the present disclosure are broadly applicable in the field of antibody therapeutics and diagnostics and may be used in conjunction with other therapeutics such as antibodies that react with a variety of targets.
The present disclosure provides MUC16-binding molecules, such as monoclonal antibodies, that can specifically bind to human MUC 16 and are cross-reactive with cynomolgus monkey MUC 16. Such MUC16-binding molecules provide certain advantages compared to the agents, compositions and/or methods currently used and/or known in the art. These advantages include improved therapeutic and pharmacological properties, increased specificity, reduced immunogenicity, and other advantageous properties.
In the present disclosure, MUC16-binding molecules, such as monoclonal antibodies, against MUC 16 which can be used to treat MUC16-overexpressing tumor have been developed. The present disclosure provides MUC16-binding molecules, nucleic acid molecules encoding the same, expression vectors and host cells used for the expression of MUC16-binding molecules, and methods for using MUC16-binding molecules. MUC16-binding molecules of the present disclosure provide potent agents for the treatment of multiple cancers via modulating human immune function.
In some embodiments, the present disclosure provides a MUC16-binding molecule comprising at least one immunoglobulin single variable domain (e.g. VHH domain) that specifically binds to MUC16, such as human MUC16 and cyno MUC16. In some embodiments, the single variable domain comprises CDR1, CDR2 and CDR3, and wherein: the CDR1 comprises an amino acid sequence as set forth in SEQ ID NO: 1; the CDR2 comprises an amino acid sequence as set forth in SEQ ID NO: 2; and
the CDR3 comprises an amino acid sequence as set forth in SEQ ID NO: 3.
In some embodiments, the single variable domain as disclosed herein comprises: a CDR1 as set forth in SEQ ID NO: 1 ; a CDR2 as set forth in SEQ ID NO: 2; and a CDR3 as set forth in SEQ ID NO: 3.
In some embodiments, the single variable domain as disclosed herein comprises:
(A) an amino acid sequence as set forth in any one of SEQ ID NOs: 4-5;
(B) an amino acid sequence at least 85%, 90%, or 95% identical to the amino acid sequence as set forth in any one of SEQ ID NOs: 4-5 yet the specific binding affinity to MUC16 is maintained (e.g., substantially maintained, for example, at least 50%, at least 60%, at least 70%, at least 80%, at least 90%, at least 95%); or
(C) an amino acid sequence with addition, deletion and/or substitution of one or more (e.g. 1, 2 or 3) amino acids compared with the amino acid sequence as set forth in any one of SEQ ID NOs: 4-5 yet the specific binding affinity to MUC 16 is maintained (e.g., substantially maintained, for example, at least 50%, at least 60%, at least 70%, at least 80%, at least 90%, at least 95%).
In some embodiments, MUC 16-binding molecules as disclosed herein comprise one or more substitutions, additions, and/or deletions of amino acids in the framework regions, e.g. FRW1, FRW2, FRW3, and/or FRW4 of a single variable domain (e.g., VHH). In some embodiments, FRW1 at the N terminal and/or FRW4 at the C terminal of the single variable domain is truncated, e.g. truncated by no more than 5, 4, 3, 2, or 1 amino acid(s).
In some embodiments, a single variable domain (e.g., VHH) comprises an amino acid sequence as set forth in any one of SEQ ID NOs: 4-5.
In some embodiments, a MUC 16-binding molecule as disclosed herein further comprises one or more human IgG constant domains, such as one or more human IgGl, IgG2, IgG3 or IgG4 constant domains. In some embodiments, the IgG constant domain is a human IgGl constant domain or a variant thereof. In some embodiments, a MUC 16-binding molecule comprises a variant of one or more human IgGl constant domains, e.g. an IgGl Fc with L234A/L235A substitutions, according to EU numbering.
In some embodiments, a MUC 16-binding molecule as disclosed herein has one or more of the following properties:
(a) specifically bind to human MUG 16 and cynomolgus monkey MUC16, especially to the 56th SEA domain of MUC16, but not to CA125 soluble protein, as measured by ELISA or FACS;
(b) have a low self-interaction propensity;
(c) have a good thermal stability; and
(d) have no non-specific binding.
In some embodiments, a MUC 16-binding molecule as disclosed herein is a chimeric antibody, a humanized antibody or a folly human antibody. In some embodiments, the MUC 16-binding molecule is a dimer.
In some embodiments, a MUC16-binding molecule as disclosed herein comprises an amino acid sequence as set forth in any one of SEQ ID NOs: 6-7.
In some embodiments, the present disclosure provides a nucleic acid molecule comprising a nucleic acid sequence encoding a MUC16-binding molecule as disclosed herein, such as a MUC16-binding molecule comprising a single variable domain (e.g., VHH).
In some embodiments, the present disclosure provides a vector comprising a nucleic acid molecule as disclosed herein.
In some embodiments, the present disclosure provides a host cell comprising an expression vector or a nucleic acid molecule as disclosed herein.
In some embodiments, the present disclosure provides a pharmaceutical composition comprising a MUC16-binding molecule as disclosed herein and a pharmaceutically acceptable carrier.
In some embodiments, the present disclosure provides a method for preparing a MUC16- binding molecule which comprises expressing the MUC16-binding molecule in a host cell as disclosed herein and isolating the MUC16-binding molecule from the host cell.
In some embodiments, the present disclosure provides a method of modulating a MUC16- related immune response in a subject, comprising administering a MUC16-binding molecule as disclosed herein to the subject such that the MUC16-related immune response in the subject is modulated.
In some embodiments, the present disclosure provides a method for treating or preventing a MUC16 positive or MUC16 overexpressed cancer in a subject comprising administering an effective amount of a MUC16-binding molecule or a pharmaceutical composition as disclosed herein to the subject. The cancer includes but is not limited to, ovary cancer, lung cancer, pancreas cancer, breast cancer, uterine cancer, fallopian tube cancer, primary peritoneum cancer or cancer of any other tissue that expresses MUC16.
In some embodiments, the present disclosure provides use of a MUC16-binding molecule as disclosed herein in the manufacture of a medicament for diagnosing, treating or preventing a MUC16 positive cancer.
In some embodiments, the present disclosure provides a MUC16-binding molecule as disclosed herein for use in treating or preventing a MUC16 positive cancer.
In some embodiments, the present disclosure provides a MUC16-binding molecule as disclosed herein for use in diagnostic methods for identifying the presence of MUC16 in tissue and/or plasma samples.
In some aspects, the present disclosure is directed to kits or devices and associated methods that employ a MUC16-binding molecule as disclosed herein, or pharmaceutical compositions as disclosed herein.
The foregoing is a summary and thus contains, by necessity, simplifications, generalizations, and omissions of detail; consequently, those skilled in the art will appreciate that the summary is illustrative only and is not intended to be in any way limiting.
BRIEF DESCRIPTION OF THE FIGURES
Figure 1 shows purity analysis of W306106-Pl lR2-lB8-uIgG1: (A) SDS-PAGE analysis; (B) SEC-HPLC chromatogram.
Figure 2 shows DSF profile of W306106-Pl lR2-lB8-uIgG1.
Figures 3-4 show purity analysis of two batches of W306106-P1 lR2-lB8-z6-uIgG1 : (A) SDS-PAGE analysis; (B) SEC-HPLC chromatogram.
Figure 5 shows SEC-HPLC profile of W306106-P1 !R2-lB8-z6-uIgG1 after 5 Freeze/Thaw cycles.
Figure 6 shows SEC-HPLC profiles of W306106-P1 !R2-lB8-z6-uIgG1 after 14 days in 40 °C.
Figure 7 shows DSF profile of W306106-P1 lR2-lB8-z6-uIgG1.
Figure 8 shows DLS-kD profiles of W306106-P1 lR2-lB8-z6-uIgG1.
Figures 9-10 show purity analysis of W306106-P11R2-1B8-Z6: (A) SDS-PAGE analysis; (B) SEC-HPLC chromatogram. Figure 10 is a concentrated form of the protein of Figure 9.
Figure 11 shows DSF profiles of W306106-Pl lR2-lB8-z6.
Figure 12 shows DLS-Radius size distribution profiles of W306106-P11R2-1B8-Z6.
Figure 13 shows HIC-HPLC profiles of W306106-P11R2-1B8-Z6.
Figure 14 shows DLS-kD profiles of W306106-P11R2-1B8-Z6.
Figure 15 shows SEC-HPLC profiles of W306106-Pl lR2-lB8-z6 after 5 Freeze/Thaw cycles.
Figure 16 shows SEC-HPLC profiles of W306106-P11R2-1B8-Z6 after 14 days in 40 °C.
Figure 17A-17B shows human (A) and cynomolgus monkey (B) MUC16 55th & 56th SEA domain FACS binding of W306106-P1 lR2-lB8-uIgG1.
Figure 18A-18B shows human (A) and cynomolgus monkey (B) MUC16 55th & 56th SEA domain FACS binding of W306106-P1 lR2-lB8-z6-uIgG1.
Figure 19A-19B shows human (A) and cynomolgus monkey (B) MUC16 55th & 56th SEA domain FACS binding of W306106-P11R2-1B8-Z6.
Figure 20 shows human MUC16 binding of W306106-P1 lR2-lB8-uIgG1 on OVCAR-3 (MUC16high).
Figure 21 A-2 ID shows human MUC 16 binding of W306106-P1 lR2-lB8-z6-uIgG1 on four human tumor cell lines.
Figure 22A-22B shows human MUC16 binding of W306106-P11R2-1B8-Z6 on two human tumor cell lines.
Figure 23A-23B shows ELISA binding of W306106-P1 lR2-lB8-uIgG1 to human MUC16 extracellular domain proteins.
Figure 24A-24C shows ELISA binding of W306106-P1 !R2-lB8-z6-uIgG1 to human MUC16 extracellular domain proteins.
Figure 25A-25C shows ELISA binding of W306106-P11R2-1B8-Z6 to human MUC16 extracellular domain proteins.
Figure 26A-26B shows ELISA binding of W306106-P1 !R2-lB8-z6-uIgG1 to cynomolgus monkey (A) and mouse (B) MUC16 extracellular domain proteins.
Figure 27A-27B shows ELISA binding of W306106-P 11R2- 1B8-Z6 to cynomolgus monkey (A) and mouse (B) MUC16 extracellular domain proteins.
Figure 28 shows binding of W306106-P1 lR2-lB8-uIgG1 to W3XX106- hPro 1.ECD.AVI.His-P2.
Figure 29 shows binding of W306106-P1 lR2-lB8-z6-uIgG1 to W3XX106- hProl.ECD.AVI.His-P2.
Figure 30 shows the result of W306106-P1 lR2-lB8-uIgG1 in HCS internalization assay on OVCAR-3 cells.
DETAILED DESCRIPTION
While the present disclosure may be embodied in many different forms, disclosed herein are specific illustrative embodiments thereof that exemplify the principles of the disclosure. It should be emphasized that the present disclosure is not limited to the specific embodiments illustrated. Moreover, any section headings used herein are for organizational purposes only and are not to be construed as limiting the subject matter described.
Unless otherwise defined herein, scientific and technical terms used in connection with the present disclosure shall have the meanings that are commonly understood by those of ordinary skill in the art. Further, unless otherwise required by context, singular terms shall include pluralities and plural terms shall include the singular. More specifically, as used in this specification and the appended claims, the singular forms “a,” “an” and “the” include plural referents unless the context clearly dictates otherwise. Thus, for example, reference to “a protein” includes a plurality of proteins; reference to “a cell” includes mixtures of cells, and the like. In this application, the use of “or” means “and/or” unless stated otherwise. Furthermore, the use of the term “comprising,” as well as other forms, such as “comprises" and “comprised”, is not limiting. In addition, ranges provided in the specification and appended claims include both end points and all points between the end points.
Generally, nomenclature used in connection with, and techniques of, cell and tissue culture, molecular biology, immunology, microbiology, genetics and protein and nucleic acid chemistry and hybridization described herein are well known and commonly used in the art. The methods
and techniques of the present disclosure are generally performed according to conventional methods well known in the art and as described in various general and more specific references that are cited and discussed throughout the present specification unless otherwise indicated. See, e.g., Abbas et al., Cellular and Molecular Immunology, 6th ed., W.B. Saunders Company (2010); Sambrook J. & Russell D. Molecular Cloning: A Laboratory Manual, 3rd ed., Cold Spring Harbor Laboratory Press, Cold Spring Harbor, N.Y. (2000); Ausubel et al., Short Protocols in Molecular Biology: A Compendium of Methods from Current Protocols in Molecular Biology, Wiley, John & Sons, Inc. (2002); Harlow and Lane Using Antibodies: A Laboratory Manual, Cold Spring Harbor Laboratory Press, Cold Spring Harbor, N.Y. (1998); and Coligan et al., Short Protocols in Protein Science, Wiley, John & Sons, Inc. (2003). The nomenclature used in connection with, and the laboratory procedures and techniques of, analytical chemistry, synthetic organic chemistry, and medicinal and pharmaceutical chemistry described herein are well known and commonly used in the art.
Definitions
In order to better understand the disclosure, the definitions and explanations of the relevant terms are provided as follows.
The term “antibody” (e.g. anti-MUC16 antibody) and “antigen-binding molecule” (e.g. MUC16-binding molecule) are used interchangeably in the broadest sense and encompass any form of antibody that exhibits the desired biological or binding activity. It covers, but is not limited to, humanized antibodies, fully human antibodies, chimeric antibodies and single-domain antibodies (sdAbs, comprising just one chain, which is typically similar to a heavy chain), as well as fragments of any of the foregoing as long as they exhibit the desired antigen-binding activity, including, for example, an antibody comprising at least one VHH domain. A conventional antibody comprises a heavy chain(s) and a light chain(s). Heavy chains may be classified into p, 5, y, a and E, which define isotypes of an antibody as IgM, IgD, IgG, IgA and IgE, respectively. A heavy chain can comprise a heavy chain variable region (VH) and a heavy chain constant region (CH). A heavy chain can comprise one or more constant regions, for example, 3 constant regions (CHI, CH2 and CH3). A light chain can comprise a light chain variable region (VL) and a light chain constant region (CL). A VH and a VL region can further be divided into hypervariable regions (called complementary determining regions (CDRs)), which are interspaced by relatively conservative regions (called framework regions (FRW)). A VH and a VL can comprise 3 CDRs (Complementarity determining regions) and 4 FRs (Framework regions) in the following order: FRW1, CDR1, FRW2, CDR2, FRW3, CDR3, FRW4 from N-terminal to C-terminal. Antibodies can be of different antibody isotypes, for example, IgG (e.g., IgGl, IgG2, IgG3 or IgG4 subtype), IgAl, IgA2, IgD, IgE or IgM antibody.
As used herein, the term “MUC16” or “MUC16 polypeptide” refers to mucin 16, which is a single transmembrane domain highly glycosylated integral membrane glycoprotein that is highly expressed in ovarian cancer. MUC16 is an extremely large glycoprotein (-22,152 amino acids) with approximately 12,000 amino acids of the heavily O-glycosylated N-terminal region, a tandem repeat region comprising approximately 60 repeats of 156 amino acids each, a transmembrane domain and a cytoplasmic tail of 32 amino acids. MUC16 harbors 56 SEA domains and each SEA domain constitutes a major portion (amino acids 1-128) of each tandem repeat. GenBank™ accession number NP 078966.2 provides an exemplary human MUC16 amino acid sequence. MUC16 is predicted to undergo cleavage in the penultimate and/or last SEA domain (i.e. the 55th and 56th SEA domain, which are closest to the transmembrane domain among the SEA domains) and phosphorylation event(s) in the cytoplasmic tail domain (CTD) is (are) believed to be critical determinants of its cleavage (Srustidhar Das et al., Understanding the Unique Attributes of MUC16 (CA125): Potential Implications in Targeted Therapy, Cancer Research, 2015).
The term “Fc region” is used to define a C -terminal region of an immunoglobulin heavy chain, including, for example, native sequence Fc regions, recombinant Fc regions, and variant Fc regions. Although the boundaries of the Fc region of an immunoglobulin heavy chain might vary, the human IgG heavy chain Fc region is often defined to stretch from an amino acid residue at position Cys226 (according to the EU numbering system), or from Pro230 (according to the EU numbering system), to the carboxyl-terminus thereof. The C-terminal lysine (residue 447 according to the EU numbering system) of the Fc region may be removed, for example, during production or purification of the antibody, or by recombinantly engineering the nucleic acid encoding a heavy chain of the antibody.
A “functional Fc region” possesses an “effector function” of a native sequence Fc region. Exemplary “effector functions” include Clq binding; complement dependent cytotoxicity (CDC); Fc receptor binding; antibody-dependent cell-mediated cytotoxicity (ADCC); phagocytosis; down regulation of cell surface receptors (e.g., B cell receptor; BCR), etc. Such effector functions generally require the Fc region to be combined with a binding region or binding domain (e.g., an antibody variable region or domain, including a VHH domain) and can be assessed using various assays as disclosed.
A “native sequence Fc region” comprises an amino acid sequence identical to the amino acid sequence of an Fc region found in nature, and not manipulated, modified, and/or changed (e.g., isolated, purified, selected, including or combining with other sequences such as variable region sequences) by a human. Native sequence human Fc regions include a native sequence human IgGl Fc region (non-A and A allotypes); native sequence human IgG2 Fc region; native sequence human IgG3 Fc region; and native sequence human IgG4 Fc region as well as naturally occurring variants thereof.
A “variant Fc region” comprises an amino acid sequence which differs from that of a native sequence Fc region by virtue of at least one amino acid modification, (e.g., substituting, addition, or deletion) preferably one or more amino acid substitution(s). In some embodiments, the variant Fc region has at least one amino acid substitution compared to a native sequence Fc region or to the Fc region of a parent polypeptide, for example, from about one to about ten amino acid substitutions, and preferably from about one to about five amino acid substitutions in a native sequence Fc region or in the Fc region of the parent polypeptide. A variant Fc region can possess at least about 80% homology with a native sequence Fc region and/or with an Fc region of a parent polypeptide, or at least about 90% homology therewith, for example, at least about 95% homology therewith. The variant Fc region herein described herein may have a loss of effector function (e.g., silent Fc).
Antibodies described herein include, but are not limited to, synthetic antibodies, monoclonal antibodies, recombinantly produced antibodies, multispecific antibodies (e.g., including bispecific antibodies), human antibodies, humanized antibodies, chimeric antibodies, intrabodies, singlechain Fvs (scFv) (e.g., including monospecific, bispecific, etc.), camelized antibodies, Fab fragments, F(ab’) fragments, disulfide-linked Fvs (sdFv), anti-idiotypic (anti-Id) antibodies, and epitope-binding fragments of any of the above.
The term “immunoglobulin single variable domain” or “single variable domain” or “VHH domain” or “VHH” or “heavy chain only antibody variable domain” may be used interchangeably herein and refers to a single chain antigen binding domain that is capable of binding to an antigen or epitope, independently of a different variable domain. A VHH domain (e.g. variable domain of a heavy chain antibody) represents the smallest known antigen-binding unit generated by adaptive immune responses (Koch-Nolte F. et al., FASEB J. Nov; 21 (13):3490-8. Epub 2007 Jun 15 (2007)). A VHH domain may be a human domain, but also includes a single domain from other species such as rodent, nurse shark and Camelid VHH domains. Camelid VHH are immunoglobulin single variable domain polypeptides that are derived from species including camel, llama, alpaca, dromedary, and guanaco, which produce heavy chain antibodies naturally devoid of light chains. Such VHH domains may be humanized according to standard techniques available in the art and are considered as “single domain antibodies”. As used herein, VHH includes camelid VHH domains and humanized VHH domains.
The term “humanized antibody” is intended to refer to antibodies in which CDR sequences derived from the germline of another mammalian species, such as a mouse, llama or alpaca, have been grafted onto human framework sequences. Additional framework region modifications may be made within the human framework sequences.
The term “Ka”, as used herein, is intended to refer to the association rate of a particular antibody-antigen interaction, whereas the term “Kd” as used herein, is intended to refer to the
dissociation rate of a particular antibody-antigen interaction. Kd values for antibodies can be determined using methods well established in the art. The term “KD” as used herein, is intended to refer to the dissociation constant of a particular antibody-antigen interaction, which is obtained from the ratio of Kd to Ka (e.g., Kd/Ka) and may be expressed as a molar concentration (M). A preferred method for determining the Ka, Kd and KD of an antibody is by using surface plasmon resonance, preferably using a biosensor system such as a Biacore® system.
The term “specific binding” or “specifically binds” as used herein refers to a non-random binding reaction between two molecules, such as for example between an antibody and an antigen.
The term “high affinity”, as used herein, refers to a MUC16 binding molecule such as an antibody having a KD of 1 x 10-9 M or less, more preferably 5 x IO'10 M or less, even more preferably IxlO'10 or less, even more preferably 5 x 10-11 or less for a target antigen.
The term “EC50”, as used herein, which is also termed as “half maximal effective concentration” refers to the concentration of a drug, antibody or toxicant which induces a response halfway between the baseline and maximum after a specified exposure time. In the context of the present disclosure,EC50 is expressed in the unit of “nM”.
The term “epitope”, as used herein, refers to a portion of an antigen that an immunoglobulin or antibody specifically binds to. “Epitope” is also known as “antigenic determinant”. Epitope or antigenic determinant generally comprises chemically active surface groups of a molecule such as amino acids, carbohydrates or sugar side chains, and generally has a specific three-dimensional structure and a specific charge characteristic. For example, an epitope generally comprises at least 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14 or 15 consecutive or non-consecutive amino acids in a unique steric conformation, which may be “linear” or “conformational”. See, for example, Epitope Mapping Protocols in Methods in Molecular Biology, Vol. 66, G. E. Morris, Ed. (1996). In a linear epitope, all the interaction sites between a protein and an interaction molecule (e.g., an antibody) are present linearly along the primary amino acid sequence of the protein. In a conformational epitope, the interaction sites span over amino acid residues that are separate from each other in a protein. Antibodies may be screened depending on competitiveness of binding to the same epitope by conventional techniques known by a person skilled in the art. For example, studies on competition or cross-competition may be conducted to obtain antibodies that compete or cross-compete with each other for binding to antigens. High-throughput methods for obtaining antibodies binding to the same epitope, which are based on their cross-competition, are described in an international patent application WO 03/48731.
The term “isolated antibody”, as used herein, is intended to refer to an antibody that is substantially free of other antibodies having different antigenic specificities (e.g., an isolated antibody that specifically binds a MUC16 protein is substantially free of antibodies that
specifically bind antigens other than MUC 16 proteins). An isolated antibody that specifically binds a human MUC 16 protein may, however, have cross-reactivity to other antigens, such as MUC 16 proteins from other species. Moreover, an isolated antibody can be substantially free of other cellular material and/or chemicals.
The term “vector”, as used herein, refers to a nucleic acid vehicle which can have a polynucleotide inserted therein. When the vector allows for the expression of the protein encoded by the polynucleotide inserted therein, the vector is called an expression vector. The vector can have carried genetic material elements expressed in a host cell by transformation, transduction, or transfection into the host cell. Vectors are well known by a person skilled in the art, including, but not limited to plasmids, phages, cosmids, artificial chromosome such as yeast artificial chromosome (YAC), bacterial artificial chromosome (BAC) or Pl -derived artificial chromosome (PAC); phage such as X phage or Ml 3 phage and animal virus. The animal viruses that can be used as vectors, include, but are not limited to, retrovirus (including lentivirus), adenovirus, adeno-associated virus, herpes virus (such as herpes simplex virus), pox virus, baculovirus, papillomavirus, papova virus (such as SV40). A vector may comprise multiple elements for controlling expression, including, but not limited to, a promoter sequence, a transcription initiation sequence, an enhancer sequence, a selection element and a reporter gene. In addition, a vector may comprise an origin of replication.
The term “host cell”, as used herein, refers to a cell into which a vector can be introduced, including, but not limited to, a prokaryotic cell such as E. coli or Bacillus subtilis, a fungal cell such as yeast cell or Aspergillus, an insect cell such as S2 Drosophila cell or Sf9, and an animal cell such as fibroblast, CHO cell, COS cell, NSO cell, HeLa cell, BHK cell, HEK 293 cell or human cell.
The term “identity”, as used herein, refers to a relationship between the sequences of two or more polypeptide molecules or two or more nucleic acid molecules, as determined by aligning and comparing the sequences. “Percent identity” means the percent of identical residues between the amino acids or nucleotides in the compared molecules and is calculated based on the size of the smallest of the molecules being compared. For these calculations, gaps in alignments (if any) are preferably addressed by a particular mathematical model or computer program (e.g., an “algorithm”). Methods that can be used to calculate the identity of the aligned nucleic acids or polypeptides include those described in Computational Molecular Biology, (Lesk, A. M., ed.), 1988, New York: Oxford University Press; Biocomputing Informatics and Genome Projects, (Smith, D. W., ed.), 1993, New York: Academic Press; Computer Analysis of Sequence Data, Part I, (Griffin, A. M., and Griffin, H. G., eds.), 1994, New Jersey: Humana Press; von Heinje, G., 1987, Sequence Analysis in
Molecular Biology, New York: Academic Press; Sequence Analysis Primer, (Gribskov, M. and Devereux, J., eds.), 1991, New York: M. Stockton Press; and Carillo et al, 1988, SIAMJ. Applied Math. 48: 1073.
The term “immunogenicity”, as used herein, refers to an ability to stimulate formation of specific antibodies or sensitized lymphocytes in organisms. It not only refers to a property of an antigen to stimulate a specific immunocyte to activate, proliferate and differentiate so as to finally generate immunologic effector substance such as antibody and sensitized lymphocyte, but also refers to a specific immune response that antibody or sensitized T lymphocyte can be formed in an immune system of an organism after stimulating the organism with an antigen. Immunogenicity is an important property of an antigen. Whether an antigen can successfully induce the generation of an immune response in a host depends on several factors, including properties of an antigen, reactivity of a host, and immunization means.
The term “transfection” or “transfect”, as used herein, refers to a process by which nucleic acids are introduced into eukaryotic cells, particularly mammalian cells. Protocols and techniques for transfection include but not limited to lipid transfection and chemical and physical methods such as electroporation. A number of transfection techniques are well known in the art and are disclosed herein. See, e.g., Graham et al., 1973, Virology 52:456; Sambrook et al., 2001, Molecular Cloning: A Laboratory Manual, supra; Davis et al., 1986, Basic Methods in Molecular Biology, Elsevier; Chu et al, 1981, Gene 13: 197.
The term “SPR” or “surface plasmon resonance”, as used herein, refers to and includes an optical phenomenon that allows for an analysis of real-time biospecific interactions by detection of alterations in protein concentrations within a biosensor matrix, for example using the BIAcore system (Pharmacia Biosensor AB, Uppsala, Sweden and Piscataway, N.J.). For further descriptions, see Example and Jonsson, U., et al. ( 1993) Ann. Biol. Clin. 51:19-26; Jonsson, U., et al. (1991) Biotechniques 11:620-627; Johnsson, B., et al. (1995) J. Mol. Recognit. 8:125-131; and Johnnson, B., et al. (1991) Anal. Biochem. 198:268-277.
The term “fluorescence-activated cell sorting” or “FACS”, as used herein, refers to a specialized type of flow cytometry. It provides a method for sorting a heterogeneous mixture of biological cells into two or more containers, one cell at a time, based upon the specific light scattering and fluorescent characteristics of each cell (FlowMetric. “Sorting Out Fluorescence Activated Cell Sorting”. Retrieved 2017-11-09.). Instruments for carrying out FACS are known to those of skill in the art and are commercially available to the public. Examples of such instruments include FACS Star Plus, FACScan and FACSort instruments from Becton Dickinson (Foster City, Calif.) Epics C from Coulter Epics Division (Hialeah, Fla.) and MoFlo from Cytomation (Colorado Springs, Colo.).
The term “subject” includes any human or nonhuman animal, preferably humans.
The term “condition associated with MUC16” or “condition related to MUC16”, as used herein, refers to any condition that is caused by, exacerbated by, or otherwise linked to increased or decreased (generally increased) expression or activities of MUC16 (e.g. a human MUC16).
The term “cancer”, as used herein, refers to any tumor or any malignant cell growth or proliferation, primary or metastasis-mediated, including solid tumors and non-solid tumors such as leukemia.
The term “treatment”, “treating” or “treated”, as used herein in the context of treating a condition, pertains generally to treatment or therapy, whether of a human or an animal, in which some desired therapeutic effect is achieved, for example, inhibition of the progress of a condition, and includes a reduction in the rate of progress, a halt in the rate of progress, regression of the condition, amelioration of the condition, and cure of the condition. Treatment as a prophylactic measure (e.g., prophylaxis, prevention) is also included. For cancer, “treating” may refer to a dampening or slowing of a tumor or malignant cell growth, proliferation, or metastasis, or some combination thereof. For tumors, “treatment” includes removal of all or part of a tumor, inhibiting or slowing tumor growth and metastasis, preventing or delaying the development of a tumor, or some combination thereof.
The term “therapeutically-effective amount,” as used herein, pertains to that amount of an active compound, or a material, composition or dosage from comprising an active compound, which is effective for producing some desired therapeutic effect, commensurate with a reasonable benefit/risk ratio, when administered in accordance with a desired treatment regimen. For example, a “therapeutically-effective amount,” of a MUC16-binding molecule refers to an amount or concentration effective to treat a human MUC16-related disease or condition.
The term “host cell”, as used herein, refers to a cell with the introduction of exogenous polynucleotides.
The term “pharmaceutically acceptable”, as used herein, means that the vehicle, diluent, excipient and/or salts thereof, are chemically and/or physically compatible with other ingredients in the formulation, and physiologically compatible with the recipient.
As used herein, the term “a pharmaceutically acceptable carrier and/or excipient” refers to a carrier, stabilizer, and/or excipient pharmacologically and/or physiologically compatible with a subject and an active agent, which is well known in the art (see, e.g., Remington's Pharmaceutical Sciences. Edited by Gennaro AR, 19th ed. Pennsylvania: Mack Publishing Company, 1995), and includes, but is not limited to a pH adjuster, surfactant, adjuvant or an ionic strength enhancer. For example, a pH adjuster includes, but is not limited to, phosphate buffer; a surfactant includes, but is not limited to, cationic, anionic, or non-ionic surfactant, e.g., Tween-80; an ionic strength enhancer includes, but is not limited to, sodium chloride. Carriers, excipients, or stabilizers are
nontoxic to the cell or mammal being exposed thereto at the dosages and concentrations employed. Often the carrier is an aqueous pH buffered solution. Examples of carriers include buffers such as phosphate, citrate, and other organic acids; antioxidants including ascorbic acid; low molecular weight (e.g., less than about 10 amino acid residues) polypeptide; proteins, such as serum albumin, gelatin, or immunoglobulins; hydrophilic polymers such as polyvinylpyrrolidone; amino acids such as glycine, glutamine, asparagine, arginine or lysine; monosaccharides, disaccharides, and other carbohydrates including glucose, mannose, or dextrins; chelating agents such as EDTA; sugar alcohols such as mannitol or sorbitol; salt-forming counterions such as sodium; and/or nonionic surfactants such as TWEEN™, polyethylene glycol (PEG), and PLURONICS™. The term “carrier” can also refer to a diluent, adjuvant (e.g., Freund’s adjuvant (complete or incomplete)), excipient, or vehicle with which the therapeutic is administered. Such carriers can be sterile liquids, such as water and oils, including those of petroleum, animal, vegetable or synthetic origin, such as peanut oil, soybean oil, mineral oil, sesame oil and the like. Water is a exemplary carrier when a composition (e.g., a pharmaceutical composition) is administered intravenously. Saline solutions and aqueous dextrose and glycerol solutions can also be employed as liquid carriers, particularly for injectable solutions. Suitable excipients (e.g., pharmaceutical excipients) include starch, glucose, lactose, sucrose, gelatin, malt, rice, flour, chalk, silica gel, sodium stearate, glycerol monostearate, talc, sodium chloride, dried skim milk, glycerol, propylene glycol, water, ethanol and the like. The composition, if desired, can also contain minor amounts of wetting or emulsifying agents, or pH buffering agents. Compositions can take the form of solutions, suspensions, emulsion, tablets, pills, capsules, powders, sustained-release formulations and the like. Oral compositions, including formulations, can include standard carriers such as pharmaceutical grades of mannitol, lactose, starch, magnesium stearate, sodium saccharine, cellulose, magnesium carbonate, etc. Examples of suitable carriers are described in Remington’s Pharmaceutical Sciences (1990) Mack Publishing Co., Easton, PA. Compositions, including pharmaceutical compounds, may contain a prophylactically or therapeutically effective amount of a MUC 16-binding agent (e.g., an anti-MUC 16 antibody), for example, in isolated or purified form, together with a suitable amount of carrier so as to provide the form for proper administration to the subject (e.g., patient). The formulation should suit the mode of administration.
As used herein, the term “adjuvant” refers to a non-specific immunopotentiator, which can enhance immune response to an antigen or change the type of immune response in an organism when it is delivered together with the antigen to the organism or is delivered to the organism in advance. There are a variety of adjuvants, including, but not limited to, aluminium adjuvants (for example, aluminum hydroxide), Freund’s adjuvants (for example, Freund’s complete adjuvant and Freund’s incomplete adjuvant), coryne bacterium parvum, lipopolysaccharide, cytokines, and the
like. Freund's adjuvant is the most commonly used adjuvant in animal experiments. Aluminum hydroxide adjuvant is more commonly used in clinical trials.
MUC16-binding molecules
MUC 16 comprises a large extracellular domain (CA-125), which is cleaved and released, and a retained domain (MUC-CD). MUC-CD comprises a non-repeating extracellular domain (MUC 16 ectodomain) proximal to a cleavage site, a transmembrane domain, and a cytoplasmic tail with potential phosphorylation sites. Distal to the cleavage site, the released extracellular domain (CA-125) contains up to 60 tandem repeats of 156 amino acids, each with many potential glycosylation sites (O'Brien TJ, et al., Tumor Biol 22(6):348-66 (2001)). Since the MUC 16 antigen is otherwise expressed only at low levels in normal tissues of the uterus, endometrium, fallopian tubes, ovaries, and serosa of the abdominal and thoracic cavities, MUC 16 is a potentially attractive target for immune-based therapies, including the targeting and treatment of cancer.
Since a significant portion of the extracellular domain of MUC 16 is cleaved and secreted (i.e., CA-125), the utility of this portion of MUC16 to be used as a target antigen on ovarian carcinomas is limited. Many reported MUC 16 monoclonal antibodies bind to epitopes present on the large secreted CA-125 fraction of the glycoprotein, and not to the retained MUC 16 ectodomain. Thus, the generation of new antibodies to the region of MUC16 that is not shed are needed for diagnostic and therapeutic purposes. This strategy might allow better MUC 16-positive tumor cell targeting efficiency, and significantly improved pharmacokinetics and efficacy of the antibody.
In some aspects, the disclosure provides MUC16-binding molecules. A MUC16-binding molecule, in a general sense, may include any molecule that specifically binds to MUC 16. In some circumstances, a “MUC16-binding molecule” may include a “MUC 16 antagonist” and an “anti- MUC16 antibody”. “MUC16 antagonist” refers to any chemical compound or biological molecule that blocks MUC 16 activities. “Anti-MUC16 antibody” includes, but not limited to, a chimeric antibody, a humanized antibody, a human antibody or a single-domain antibody. A MUC 16- binding molecule is not limited to a polypeptide or a protein and may comprise other components such as nucleotides, hybrids, glucans and a combination thereof. As exemplified herein, a MUC 16- binding molecule may be an MUC16-binding VHH, an anti-MUC16 antibody or anti-MUC16 fusion protein.
In some embodiments, MUC16-binding molecules as disclosed herein comprise at least one VHH that specifically binds to MUC 16. Further, a MUC16-binding molecule may be a singledomain antibody and comprising one VHH. For example, a single-domain antibody is able to bind selectively to a specific antigen (e.g., MUC 16). In some embodiments, a MUC16-binding molecule comprises a VHH fused to an immunoglobulin Fc region, for example, an Fc region of IgG (e.g., IgG4 or IgGl). In some embodiments, the Fc region is an Fc region of human IgGl. By
fusing a VHH to an Fc region, it may be more efficient to recruit effector functions. Also, fusion of a VHH to an Fc region may help a MUC16-binding molecule to form a dimer and may also help the extension of the half life of the MUC16-binding molecule in vivo.
As known in the art, VHH molecules derived from Camelidae antibodies are among the smallest intact antigen-binding domains known (approximately 15 kDa, or 10 times smaller than a conventional IgG) and hence are well suited towards delivery to dense tissues and for accessing the limited space between macromolecules.
VHHs as disclosed herein may be made by the skilled artisan according to methods known in the art or any future method. For example, VHHs may be obtained using methods known in the art such as by immunizing a camel and obtaining hybridoma's therefrom, or by cloning a library of VHHs of the disclosure using molecular biology techniques known in the art and subsequent selection by using phage display.
For example, a VHH can be obtained by immunization of llamas or alpacas with the desired antigen and subsequent isolation of the mRNA coding for heavy-chain antibodies. By reverse transcription and polymerase chain reaction, a gene library of single-domain antibodies containing several million clones is produced. Screening techniques like phage display and ribosome display help to identify the clones binding the antigen. One technique is phage display in which a library of (e.g., human) antibodies is synthesized on phages, the library is screened with the antigen of interest or an antibody-binding portion thereof, and the phage that binds the antigen is isolated, from which one may obtain the immunoreactive fragments. Methods for preparing and screening such libraries are well known in the art and kits for generating phage display libraries are commercially available (e.g., the Pharmacia Recombinant Phage Antibody System, catalog no. 27-9400-01; and the Stratagene SurfZAP™ phage display kit, catalog no. 240612). There also are other methods and reagents that can be used in generating and screening antibody display libraries (see, e.g., Barbas et al., Proc. Natl. Acad. Sci. USA 88:7978-7982 (1991)).
When potent clones have been identified, their DNA sequence is optimized, for example, by affinity maturation or humanization. Humanization may prevent immunological reactions of the human organism against the antibody.
Accordingly, the VHHs can be obtained (1) by isolating the VHH domain of a naturally occurring heavy chain antibody; (2) by expression of a nucleotide sequence encoding a naturally occurring VHH domain; (3) by “humanization” (as described below) of a naturally occurring VHH domain or by expression of a nucleic acid encoding a such humanized VHH domain; (4) by “camelization” of a naturally occurring VH domain from any animal species, in particular a species of mammal, such as from a human being, or by expression of a nucleic acid encoding such a camelized VH domain; (5) by “camelisation” of a “domain antibody” or “Dab” as described by Ward et al (supra), or by expression of a nucleic acid encoding such a camelized VH domain; (6)
using synthetic or semi-synthetic techniques for preparing proteins, polypeptides or other amino acid sequences; (7) by preparing a nucleic acid encoding a VHH using techniques for nucleic acid synthesis, followed by expression of the nucleic acid thus obtained; (8) subjecting heavy chain antibodies or VHHs to affinity maturation, to mutagenesis (e.g. random mutagenesis or site- directed mutagenesis) and/or any other technique(s) in order to increase the affinity and/or specificity of the VHH; and/or (9) by any combination of the foregoing. Suitable methods and techniques for performing the foregoing will be clear to the skilled person based on the disclosure herein and, for example, include methods and techniques described in more detail herein.
Single-domain antibodies are usually generated by PCR cloning of variable domain repertoire from blood, lymph node, or spleen cDNA obtained from immunized animals into a phage display vector. Antigen-specific single-domain antibodies are commonly selected by panning phase libraries on immobilized antigen, for example, antigen coated onto the plastic surface of a test tube, biotinylated antigens immobilized on Streptavidin beads, or membrane proteins expressed on the surface of cells. The affinity of sdAbs can often been improved by mimicking this strategy in vitro, for example, by site directed mutagenesis of the CDR regions and further rounds of panning on immobilized antigen under conditions of increased stringency (higher temperature, high or low salt concentration, high or low pH, and low antigen concentrations) (Wesolowski et al., Single domain antibodies: promising experimental and therapeutic tools in infection and immunity. Med Microbiol Immunol (2009) 198: 157-174).
Methods for preparing a VHH specifically binding to an antigen or epitope was described in references, for example: R. van der Linden et al., Journal of Immunological Methods, 240(2000) 185-195; Li et al., J Biol Chem., 287(2012)13713-13721; Deffar et al., African Journal of Biotechnology Vol. 8(12), pp.2645, 17 June, 2009 and WO 94/04678.
In some embodiments, a VHH may be truncated at the N-terminus or C-terminus such that it comprises only a partial FRW 1 and/or FRW4, or lacks one or both of those framework regions, so long as the VHH substantially maintains antigen binding and specificity (e.g., substantially maintained, for example, at least 50%, at least 60%, at least 70%, at least 80%, at least 90%, at least 95%).
The present disclosure also encompasses MUC16-binding molecules with a masking moiety and/or cleavable moiety in which one or more of the MUC16-binding domains of the MUC16- binding molecules are masked (e.g., via a masking moiety) and/or activatable (e.g., via a cleavable moiety). Technologies for masking of a MUC16-binding molecule (e.g., an antibody) are well known in the art, including SAFE body masking technology (see, e.g., US 2019/0241886) and Probody masking technology (see, e.g., US 2015/0079088). Such technologies can be used to generate a MUC16-binding molecule (e.g., an antibody) that is masked and/or activatable. Such masked and/or activatable MUC16-binding molecules (e.g., antibodies) are useful for the
preparation of conjugates, including immunoconjugates, antibody-drug conjugates (ADCs), masked ADCs and activatable antibody-drug conjugates (AADCs), comprising any one of the MUC16-binding molecules (e.g., antibodies) of the present disclosure, including those directly or indirectly linked to another agent such as a drug. For example, MUC16-binding molecules of the present disclosure may be covalently bound by a synthetic linker to one or more agents such as drugs.
If desired, a MUC16-binding molecule is linked or conjugated (directly or indirectly) to a moiety with effector function, such as cytotoxic activity (e.g., a chemotherapeutic moiety or a radioisotope) or immune recruitment activity. Moieties that are linked or conjugated (directly or indirectly) include drugs that are cytotoxic (e.g., toxins such as auristatins) or non-cytotoxic (e.g., signal transduction modulators such as kinases or masking moieties that mask one or more binding domains of a MUC16-binding molecule, or cleavable moieties that allow for activating a MUC16- binding molecule by cleaving of a cleavable moiety to unmask one or more binding domains of a MUC16-binding molecule in the tumor microenvironment, in the form of masked conjugates. Moieties that promote immune recruitment can include other antigen-binding agents, such as viral proteins that bind selectively to cells of the innate immune system. Alternatively or in addition, a MUC16-binding molecule is optionally linked or conjugated (directly or indirectly) to a moiety that facilitates isolation from a mixture (e.g., a tag) or a moiety with reporter activity (e.g., a detection label or reporter protein). It will be appreciated that the features of a MUC16-binding molecule described herein extend also to a polypeptide comprising a MUC16-binding molecule fragment.
In some embodiments, MUC16-binding molecules described herein may be linked or conjugated (directly or indirectly) to a polypeptide, which can result in the generation of an activatable antibody. In some embodiments, a MUC16-binding molecule is linked or conjugated (directly or indirectly) to an agent. In some embodiments, the agent is a drug, resulting in an ADC or an AADC when the antibody of the ADC comprises a masking moiety and a cleavable moiety.
In some embodiments, MUC16-binding molecules described herein are conjugated or recombinantly linked (directly or indirectly) to a therapeutic agent (e.g., a cytotoxic agent) or to a diagnostic or detectable agent. The conjugated or recombinantly linked antibodies, including masked or activatable conjugates, can be useful, for example, for treating or preventing a disease, disorder or condition, such as a cancer or a tumor.
Diagnosis and detection can be accomplished, for example, by coupling a MUC16-binding molecule to detectable substances including, for example: enzymes, including, but not limited to, horseradish peroxidase, alkaline phosphatase, beta-galactosidase, or acetylcholinesterase; prosthetic groups, including, but not limited to, streptavidin/biotin or avidin/biotin; fluorescent materials, including, but not limited to, umbelliferone, fluorescein, fluorescein isothiocynate,
rhodamine, dichlorotriazinylamine fluorescein, dansyl chloride, or phycoerythrin; luminescent materials, including, but not limited to, luminol; bioluminescent materials, including, but not limited to, luciferase, luciferin, or aequorin; chemiluminescent material, including, but not limited to, an acridinium based compound or a HALOTAG; radioactive materials, including, but not limited to, iodine (1311, 1251, 1231, and 1211), carbon (14C), sulfur (35S), tritium (3H), indium (115In, 113In, 112In, and 11 1In), technetium (99Tc), thallium (201Ti), gallium (68Ga and 67Ga), palladium (103Pd), molybdenum (99Mo), xenon (133Xe), fluorine (18F), 153Sm, 177Lu, 159Gd, 149Pm, 140La, 175Yb, 166Ho, 90Y, 47Sc, 186Re, 188Re, 142Pr, 105Rh, 97Ru, 68Ge, 57Co, 65Zn, 85Sr, 32P, 153Gd, 169Yb, 51Cr, 54Mn, 75Se, 113Sn, or 117Sn; positron emitting metals using various positron emission tomographies; and non-radioactive paramagnetic metal ions.
Conjugates of an antibody and agent, including wherein the agent is a drug for the preparation of ADC or an AADC, may be made using a variety of bifunctional protein coupling agents such as BMPS, EMCS, GMBS, HBVS, LC-SMCC, MBS, MPBH, SBAP, SIA, SIAB, SMCC, SMPB, SMPH, sulfo-EMCS, sulfo-GMBS, sulfo-KMUS, sulfo-MBS, sulfo-SIAB, sulfo-SMCC, sulfo- SMPB, and SVSB (succinimidyl-(4-vinylsulfone) benzoate). The present disclosure further contemplates that conjugates of antibodies and agents, including wherein the agent is a drug for the preparation of an ADC or AADC, may be prepared using any suitable methods as disclosed in the art (see, e.g., Bioconjugate Techniques (Hermanson ed., 2d ed. 2008)).
Conventional conjugation strategies for antibodies and agents, including wherein the agent is a drug for the preparation of ADC or AADC, have been based on random conjugation chemistries involving the e-amino group of Lys residues or the thiol group of Cys residues, which results in heterogeneous conjugates. Recently developed techniques allow site-specific conjugation to antibodies, resulting in homogeneous loading and avoiding conjugate subpopulations with altered antigen-binding or pharmacokinetics. These include engineering of “thiomabs” comprising cysteine substitutions at positions on the heavy and light chains that provide reactive thiol groups and do not disrupt immunoglobulin folding and assembly or alter antigen binding (see, e.g., Junutula et al., 2008, J. Immunol. Meth. 332: 41-52; and Junutula et al., 2008, Nature Biotechnol. 26:925-32). In another method, selenocysteine is cotranslationally inserted into an antibody sequence by recoding the stop codon UGA from termination to selenocysteine insertion, allowing site specific covalent conjugation at the nucleophilic selenol group of selenocysteine in the presence of the other natural amino acids (see, e.g., Hofer et al., 2008, Proc. Natl. Acad. Sci. USA 105:12451-56; and Hofer et al., 2009, Biochemistry 48(50): 12047-57).
MUC16-binding molecules described herein may be monospecific, bispecific, trispecific or of greater multispecificity. Such agents may include antibodies. Multispecific antibodies, such as bispecific antibodies, are monoclonal antibodies that have binding specificities for at least two different targets (e.g., antigens) or two different epitopes on the same target (e.g., a bispecific
antibody directed to MUC16 with a first binding domain for a first epitope of MUC16, and a second binding domain for a second epitope of MUC16. In some embodiments, the multispecific (e.g., bispecific) antibodies can be constructed based on the sequences of the antibodies described herein. In some embodiments, the multispecific antibodies described herein are bispecific antibodies. In some embodiments, bispecific antibodies are mouse, chimeric, human or humanized antibodies. In some embodiments, one of the binding specificities of the multispecific antibody is for MUC 16 and the other is for any other target (e.g., antigen). In some embodiments, a multispecific (e.g., bispecific) antibody can comprise more than one target (e.g., antigen) binding domain, in which different binding domains are specific for different targets (e.g., a first binding domain that binds to MUC 16 and a second binding domain that binds another target (e.g., antigen), such as an immune checkpoint regulator (e.g., a negative checkpoint regulator). In some embodiments, multispecific (e.g., bispecific) antibody molecules can bind more than one (e.g., two or more) epitopes on the same target (e.g., antigen). In some embodiments, one of the binding specificities is MUC 16 and the other is for one or more of Cytotoxic T-lymphocyte antigen-4 (CTLA-4), CD80, CD86, Programmed cell death 1 (PD-1), Programmed cell death ligand 1 (PD- Ll), Programmed cell death ligand 2 (PD-L2), Lymphocyte activation gene-3 (LAG-3; also known as CD223), Galectin-3, B and T lymphocyte attenuator (BTLA), T-cell membrane protein 3 (TIM3), Galectin-9 (GAL9), B7-H1, B7-H3, B7-H4, T-Cell immunoreceptor with Ig and ITIM domains (TIGIT/V stm3/WUCAM/VSIG9), V-domain Ig suppressor of T-Cell activation (VISTA), Glucocorticoid-induced tumor necrosis factor receptor-related (GITR) protein, Herpes Virus Entry Mediator (HVEM), 0X40, CD27, CD28, CD137. CGEN-15001T, CGEN-15022, CGEN-15027, CGEN- 15049, CGEN- 15052, and CGEN- 15092.
Methods for making multispecific antibodies are known in the art, for example, by coexpression of two immunoglobulin heavy chain-light chain pairs, where the two heavy chains have different specificities (see, e.g., Milstein and Cuello, 1983, Nature 305:537-40). For further details of generating multispecific antibodies (e.g., bispecific antibodies), see, for example, Bispecific Antibodies (Kontermann ed., 2011).
The present disclosure provides humanized antibodies that bind MUC 16. Various methods for humanizing non-human antibodies are known in the art. For example, a humanized antibody can have one or more amino acid residues introduced into it from a source that is non-human. These non-human amino acid residues are often referred to as “import” residues, which are typically taken from an “import” variable domain. Humanized antibodies that bind MUC 16 may be produced using techniques known to those skilled in the art (e.g., Zhang et al., Molecular Immunology, 42(12): 1445-1451, 2005; Hwang et al., Methods, 36(1): 35-42, 2005; Dall’Acqua et al., Methods, 36(1): 43-60, 2005; Clark, Immunology Today, 21(8): 397-402, 2000, and U.S. Patent Nos. 6,180,370; 6,054,927; 5,869,619; 5,861,155; 5,712,120; and 4,816,567).
A MUC16-binding molecule may be described as an anti-MUC16 antibody in the following sections.
Anti-MUC16 antibodies with functional properties
Antibodies of the disclosure including, for example, antibodies comprising at least one VHH domain, are characterized by particular functional features or properties of the antibodies. In some embodiments, the antibodies have one or more of the following properties:
(a) bind to human MUC 16 and cyno MUC 16 with EC50 at nM grade, as measured by ELISA or FACS, bind to human MUC 16 with a KD no more than 0.1 nM, as measured by SPR;
(b) does not bind to CAI 25 soluble protein;
(c) have a low self-interaction propensity and good thermal stability; and
(d) have no non-specific binding.
An antibody of the disclosure binds to cell surface MUC 16 with high affinity. The binding of an antibody of the disclosure to MUC 16 can be assessed using one or more techniques well established in the art, for example, ELISA. The binding specificity of an antibody of the disclosure can also be determined by monitoring binding of the antibody to cells expressing a MUC 16 protein, e.g., by flow cytometry. For example, an antibody can be tested by a flow cytometry assay (e.g., FACS) in which the antibody is reacted with a cell line that expresses human MUC 16, such as CHO cells and 293 cells that have been transfected to express MUC 16 on their cell surface. Additionally or alternatively, the binding of the antibody, including the binding kinetics (e.g., Kd value) can be tested in BIAcore binding assays. Still other suitable binding assays include ELISA assays, for example using a recombinant MUC 16 protein. For example, an antibody of the disclosure binds to a cell surface MUC 16 (e.g., human MUC 16 55th &56th SEA domain) protein with a KD of 1 X 10-7 M or less, 5 x 10-8 M or less, 2 x 10-8 M or less, 5 x 10-9 M or less, 4 x 10-9 M or less, 3 x 10-9 M or less, 2 x 10-9 M or less, 1 x 10-9 M or less, 5 x IO'10 M or less, or 1 x IO'10 M or less.
In some embodiments, the antibodies of the disclosure bind to cynomolgus monkey MUC 16 (e.g., MUC 16 55th &56th SEA domain)at an EC50 of no more than or about 10 nM, 9 nM, 8 nM, 7 nM, 6 nM, 5 nM, 4 nM, 3 nM, 2 nM, 1 nM, 0.9 nM, 0.8 nM, 0.7 nM, 0.6 nM, 0.5 nM, 0.4 nM, 0.3 nM, 0.2 nM, 0.1 nM, 0.09 nM, 0.08 nM, 0.07 nM, 0.06 nM, 0.05 nM, 0.04 nM, 0.03 nM, 0.02 nM, or 0.01 nM, as measured by FACS.
Anti-MUC16 antibodies comprising VHH CDRs
In some embodiments, an anti-MUC16 antibody as disclosed herein comprises at least one immunoglobulin single variable domain (e.g., VHH), wherein the VHH comprises CDR1, CDR2 and CDR3, and wherein CDR1 comprises the amino acid sequence as set forth in SEQ ID NO: 1
or an amino acid sequence that differs from SEQ ID NO: 1 by no more than 2 amino acid modifications (e.g. substitution, deletion and/or insertion); CDR2 comprises the amino acid sequence as set forth in SEQ ID NO: 2 or an amino acid sequence that differs from SEQ ID NO: 2 by no more than 2 amino acid modifications (e.g. substitution, deletion and/or insertion); and CDR3 comprises the amino acid sequence as set forth in SEQ ID NO: 3 or an amino acid sequence that differs from SEQ ID NO: 3 by no more than 2 amino acid modifications (e.g. substitution, deletion and/or insertion). Preferably, the amino acid substitution is a conservative substitution.
In some specific embodiments, CDR1 comprises an amino acid sequence as set forth in SEQ ID NO: 1, CDR2 comprises an amino acid sequence as set forth in SEQ ID NO: 2, and CDR3 comprises an amino acid sequence as set forth in SEQ ID NO: 3. In some embodiments, the CDR numbering are according to Kabat + IMGT scheme.
The extent of the framework region and CDRs can be precisely identified using methodology known in the art, for example, by the Kabat definition, the Chothia definition, the AbM definition, the contact definition, the IMGT definition (all of which are well known in the art) and any combinations thereof. See, e.g., Kabat, E.A., etal. (1991) Sequences of Proteins of Immunological Interest, Fifth Edition, U.S. Department of Health and Human Services, NIH Publication No. 91- 3242, Chothia et al., (1989) Nature 342:877; Chothia, C. et al. (1987) J. Mol. Biol. 196:901-917, Al-lazikani et al (1997) J. Molec. Biol. 273:927-948; Edelman et al., Proc Natl Acad Sci U S A. 1969 May, 63(l):78-85; and Martin and Allen, m"Handbook of Therapeutic Antibodies”, chapter 5, 2007. See also hgmp.mrc.ac.uk and bioinf.org.uk/abs. Correspondence or alignments between numberings according to different definitions can for example be found at www.imgt.org/ (see also Giudicelli V et al. IMGT, the international ImMunoGeneTics database. Nucleic Acids Res. (1997) 25:206-11; and Lefranc MP et al., IMGT unique numbering for immunoglobulin and T cell receptor variable domains and Ig superfamily V-like domains. Dev Comp Immunol. (2003) 27:55-77).
As will be appreciated by those in the art, the exact numbering and placement of the CDRs can be different among different numbering systems. However, it should be understood that the disclosure of a variable heavy sequence, a variable light sequence and/or a VHH sequence includes the disclosure of the associated (inherent) CDRs. Accordingly, the disclosure of each variable region is a disclosure of the CDRs (e.g., CDR1, CDR2 and CDR3). Two antibodies having the same VH, VL or VHH CDRs means that their CDRs are identical when determined by the same approach (e.g., the Kabat, AbM, Chothia, Contact, and IMGT numbering approaches as known in the art).
Variable regions and CDRs in an antibody sequence can be identified according to general rules that have been developed in the art (for example, the Kabat, AbM, Chothia, Contact, and IMGT numbering system) or by aligning the sequences against a database of known variable
regions. Methods for identifying these regions are described in Kontermann and Dubel, eds., Antibody Engineering, Springer, New York, NY, 2001 and Dinarello et al., Current Protocols in Immunology, John Wiley and Sons Inc., Hoboken, NJ, 2000. Exemplary databases of antibody sequences are described in, and can be accessed through, the “Abysis" website at www.bioinf.org.uk/abs (maintained by A.C. Martin in the Department of Biochemistry & Molecular Biology University College London, London, England) and the VBASE2 website at www.vbase2.org, as described in Retter et al., Nucl. Acids Res., 33 (Database issue): D671 -D674 (2005). Preferably sequences are analyzed using the Abysis database, which integrates sequence data from Kabat, IMGT and the Protein Data Bank (PDB) with structural data from the PDB. See Dr. Andrew C. R. Martin's book chapter Protein Sequence and Structure Analysis of Antibody Variable Domains. In: Antibody Engineering Lab Manual (Ed.: Duebel, S. and Kontermann, R., Springer- Verlag, Heidelberg, ISBN-13: 978-3540413547, also available on the website bioinforg.uk/abs). The Abysis database website further includes general rules that have been developed for identifying CDRs which can be used in accordance with the teachings herein. Figure 10 shows an alignment of exemplary immunoglobulin single variable domains and boundaries of CDRs are indicated by Kabat, AbM, Chothia, Contact, and IMGT numbering.
In some embodiments, a MUC16-binding molecule as disclosed herein comprises at least one immunoglobulin single variable domain (e.g., VHH), wherein the VHH comprises FRW1-CDR1- FRW2-CDR2-FRW3-CDR3-FRW4, and wherein CDR1 has an amino acid sequence as set forth in SEQ ID NO: 1, CDR2 has an amino acid sequence as set forth in SEQ ID NO: 2, and CDR3 has an amino acid sequence as set forth in SEQ ID NO: 3. In some embodiments, the FRW1 and FRW4 at the N and C terminal of the VHH comprised in a MUC16-binding molecule may be truncated such that it comprise only a partial FRW1 and/or FRW4, or the VHH lacks one or both of these framework regions, so long as the VHH substantially maintains antigen binding and specificity.
In some embodiments, provided herein is an anti-MUC16 antibody (such as an anti-MUC16 single domain antibody) comprising one, two, or all three CDRs of the amino acid sequence as set forth in SEQ ID NO: 4. In some embodiments, there is provided an anti-MUC16 antibody (such as an anti-MUC16 single domain antibody) comprising one, two, or all three CDRs of the amino acid sequence as set forth in SEQ ID NO: 5. In some embodiments, the anti-MUC16 single domain antibody is camelid. In some embodiments, the anti-MUC16 antibody (such as the anti-MUC16 single domain antibody) is humanized. In some embodiments, the anti-MUC16 antibody (such as the anti-MUC16 single domain antibody) comprises an acceptor human framework, e.g., a human immunoglobulin framework or a human consensus framework.
In some embodiments, the anti-MUC16 antibody (such as the single domain antibody) comprises a CDR1 same as that of an amino acid sequence as set forth in SEQ ID NO: 4. In some
embodiments, the anti-MUC16 antibody (such as the single domain antibody) comprises a CDR2 same as that of an amino acid sequence as set forth in SEQ ID NO: 4. In other embodiments, the anti-MUC16 antibody (such as the single domain antibody) comprises a CDR3 same as that of an amino acid sequence as set forth in SEQ ID NO: 4. In some embodiments, the anti-MUC16 antibody (such as the single domain antibody) comprises a CDR1 and a CDR2 same as those of an amino acid sequence as set forth in SEQ ID NO: 4. In some embodiments, the anti-MUC16 antibody (such as the single domain antibody) comprises a CDR1 and a CDR3 same as those of an amino acid sequence as set forth in SEQ ID NO: 4. In some embodiments, the anti-MUC16 antibody (such as the single domain antibody) comprises a CDR2 and a CDR3 same as those of an amino acid sequence as set forth in SEQ ID NO: 4. In some embodiments, the anti-MUC16 antibody (such as the single domain antibody) comprises a CDR1, a CDR2, and a CDR3 same as those of an amino acid sequence as set forth in SEQ ID NO: 4. CDR sequences can be determined according to well-known numbering systems. In some embodiments, the CDRs are according to IMGT numbering. In some embodiments, the CDRs are according to Kabat numbering. In some embodiments, the CDRs are according to a combination of Kabat and IMGT numbering. In other embodiments, the CDRs are according to Chothia numbering. In other embodiments, the CDRs are according to Contact numbering. In some embodiments, the CDRs are according to AbM numbering. In some embodiments, the anti-MUC16 single domain antibody is camelid. In some embodiments, the anti-MUC16 antibody (such as the anti-MUC16 single domain antibody) is humanized. In some embodiments, the anti-MUC16 antibody (such as the anti-MUC16 single domain antibody) comprises an acceptor human framework, e.g., a human immunoglobulin framework or a human consensus framework.
In some embodiments, the anti-MUC16 antibody (such as the single domain antibody) comprises a CDR1 same as that of an amino acid sequence as set forth in SEQ ID NO: 5. In some embodiments, the anti-MUC16 antibody (such as the single domain antibody) comprises a CDR2 same as that of an amino acid sequence as set forth in SEQ ID NO: 5. In other embodiments, the anti-MUC16 antibody (such as the single domain antibody) comprises a CDR3 same as that of an amino acid sequence as set forth in SEQ ID NO: 5. In some embodiments, the anti-MUC16 antibody (such as the single domain antibody) comprises a CDR1 and a CDR2 same as those of an amino acid sequence as set forth in SEQ ID NO: 5. In some embodiments, the anti-MUC16 antibody (such as the single domain antibody) comprises a CDR1 and a CDR3 same as those of an amino acid sequence as set forth in SEQ ID NO: 5. In some embodiments, the anti-MUC16 antibody (such as the single domain antibody) comprises a CDR2 and a CDR3 same as those of an amino acid sequence as set forth in SEQ ID NO: 5. In some embodiments, the anti-MUC16 antibody (such as the single domain antibody) comprises a CDR1, a CDR2, and a CDR3 same as those of an amino acid sequence as set forth in SEQ ID NO: 5. CDR sequences can be determined
according to well-known numbering systems. In some embodiments, the CDRs are according to IMGT numbering. In some embodiments, the CDRs are according to Kabat numbering. In some embodiments, the CDRs are according to a combination of Kabat and IMGT numbering. In other embodiments, the CDRs are according to Chothia numbering. In other embodiments, the CDRs are according to Contact numbering. In some embodiments, the CDRs are according to AbM numbering. In some embodiments, the anti-MUC16 single domain antibody is camelid. In some embodiments, the anti-MUC16 antibody (such as the anti-MUC16 single domain antibody) is humanized. In some embodiments, the anti-MUC16 antibody (such as the anti-MUC16 single domain antibody) comprises an acceptor human framework, e.g., a human immunoglobulin framework or a human consensus framework.
In some embodiments, provided herein is a single domain antibody that binds to MUC16 comprising the following structure: FR1-CDR1-FR2-CDR2-FR3-CDR3-FR4, wherein (i) the CDR1 comprises an amino acid sequence as set forth in SEQ ID NO: 1; (ii) the CDR2 comprises an amino acid sequence as set forth in SEQ ID NO: 2; and/or (iii) the CDR3 comprises an amino acid sequence as set forth in SEQ ID NO: 3. In some embodiments, the anti-MUC16 single domain antibody is camelid. In some embodiments, the anti-MUC16 single domain antibody is humanized. In some embodiments, the anti-MUC16 single domain antibody comprises an acceptor human framework, e.g., a human immunoglobulin framework or a human consensus framework.
In some embodiments, the CDR1 comprises the exemplary amino acid sequence as set forth in SEQ ID NO: 1; the CDR2 comprises the exemplary amino acid sequence as set forth in SEQ ID NO: 2; and the CDR3 comprises the exemplary amino acid sequence as set forth in SEQ ID NO: 3. In some embodiments, the CDR1 is according to Kabat+IMGT numbering, comprising the amino acid sequence as set forth in SEQ ID NO: 1; the CDR2 is according to Kabat+IMGT numbering, comprising the amino acid sequence as set forth in SEQ ID NO: 2; and the CDR3 is according to Kabat+IMGT numbering, comprising the amino acid sequence as set forth in SEQ ID NO: 3. In some embodiments, the anti-MUC16 single domain antibody is camelid. In some embodiments, the anti-MUC16 single domain antibody is humanized. In some embodiments, the anti-MUC16 single domain antibody comprises an acceptor human framework, e.g., a human immunoglobulin framework or a human consensus framework.
In some embodiments, the single domain antibody further comprises one or more framework regions of W306106-P11R2-1B8 or W306106-P11R2-1B8-Z6. In some embodiments, the single domain antibody comprises one or more framework(s) derived from a VHH domain comprising the sequence as set forth in SEQ ID NO: 4. In some embodiments, the single domain antibody comprises one or more framework(s) derived from a VHH domain comprising the sequence as set forth in SEQ ID NO: 5. In some embodiments, the single domain antibody provided herein is a humanized single domain antibody.
Framework regions described herein are determined based upon the boundaries of the CDR numbering system. In other words, if the CDRs are determined by, e.g., IMGT, Kabat, Chothia, Contact, or AbM, then the framework regions are the amino acid residues surrounding the CDRs in the variable region in the format, from the N-terminus to C-terminus: FR1-CDR1-FR2-CDR2- FR3-CDR3-FR4. For example, FR1 is defined as the amino acid residues N-terminal to the CDR1 amino acid residues as defined by, e.g., the IMGT numbering system, the Kabat numbering system, the Chothia numbering system, the Contact numbering system, the AbM numbering system or a combination thereof, FR2 is defined as the amino acid residues between CDR1 and CDR2 amino acid residues as defined by, e.g., the IMGT numbering system, the Kabat numbering system, the Chothia numbering system, the Contact numbering system, the AbM numbering system or a combination thereof, FR3 is defined as the amino acid residues between CDR2 and CDR3 amino acid residues as defined by, e.g., the IMGT numbering system, the Kabat numbering system, the Chothia numbering system, the Contact numbering system, the AbM numbering system or a combination thereof, and FR4 is defined as the amino acid residues C -terminal to the CDR3 amino acid residues as defined by, e.g., the IMGT numbering system, the Kabat numbering system, the Chothia numbering system, the Contact numbering system, the AbM numbering system or a combination thereof.
In some embodiments, there is provided an isolated anti-MUC16 single domain antibody comprising a VHH domain having the amino acid sequence as set forth in SEQ ID NO: 4. In some embodiments, there is provided a polypeptide comprising the amino acid sequence as set forth in SEQ ID NO: 4. In some embodiments, there is provided an isolated anti-MUC16 single domain antibody comprising a VHH domain having the amino acid sequence as set forth in SEQ ID NO: 5. In some embodiments, there is provided a polypeptide comprising the amino acid sequence as set forth in SEQ ID NO: 5.
Anti-MUC16 antibodies comprising VHH sequences
In some embodiments, anti-MUC16 antibodies comprise at least one immunoglobulin single variable domain (e.g., VHH), wherein the VHH comprises or consists of:
(A) an amino acid sequence as set forth in any one of SEQ ID NOs: 4 and 5;
(B) an amino acid sequence which is at least 85%, at least 90%, or at least 95% identical to any one of SEQ ID NOs: 4 and 5; or
(C) an amino acid sequence with addition, deletion and/or substitution of one or more (for example, 1, 2, 3, 4, 5, 6, 7, 8, 9 or 10) amino acids compared with any one of SEQ ID NOs: 4 and 5.
The percent identity between two amino acid sequences can be determined using the algorithm of E. Meyers and W. Miller (Comput. Appl. Biosci., 4:11-17 (1988)) which has been
incorporated into the ALIGN program (version 2.0), using a PAM120 weight residue table, a gap length penalty of 12 and a gap penalty of 4. In addition, the percent identity between two amino acid sequences can be determined by the algorithm of Needleman and Wunsch (J. Mol. Biol. 48:444-453 (1970)) which has been incorporated into the GAP program in the GCG software package (available at http://www.gcg.com), using either a Blossum 62 matrix or a PAM250 matrix, and a gap weight of 16, 14, 12, 10, 8, 6, or 4 and a length weight of 1, 2, 3, 4, 5, or 6.
Additionally or alternatively, protein (e.g., antibody) sequences of the present disclosure can further be used as a “query sequence” to perform a search against public databases to, for example, identify related sequences. Such searches can be performed using the XBLAST program (version 2.0) of Altschul, et al. (1990) J. Mol. Biol. 215:403-10. BLAST protein searches can be performed with the XBLAST program, score = 50, wordlength = 3 to obtain amino acid sequences homologous to the antibody molecules of the disclosure. To obtain gapped alignments for comparison purposes, Gapped BLAST can be utilized as described in Altschul et al, (1997) Nucleic Acids Res. 25(17):3389-3402. When utilizing BLAST and Gapped BLAST programs, the default parameters of the respective programs {e.g., XBLAST and NBLAST) can be used. See www.ncbi.nlm.nih.gov.
In some embodiments, the amino acid sequence of a VHH can be at least 85%, 86%, 87%, 88%, 89%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98% or 99% identical to any one of SEQ ID NOs: 4 and 5. Preferably, the VHH may have same CDRs (CDR1, CDR2 and CDR3) as those of SEQ ID NO: 4 or 5, and at least 85%, 86%, 87%, 88%, 89%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98% or 99% identical in the framework region to those of SEQ ID NO: 4 or 5.
In some further embodiments, anti-MUC16 antibodies may contain conservative substitution or modification of amino acids in the variable regions and/or constant regions. It is understood in the art that certain conservative sequence modification can be made which do not remove antigen binding. See, e.g., Brummell et al. (1993) Biochem 32:1180-8; de Wildt et al. (1997) Prot. Eng. 10:835-41; Komissarov et al. (1997) J. Biol. Chem. 272:26864- 26870; Hall et al. (1992) J. Immunol. 149:1605-12; Kelley and O’ Connell (1993) Biochem. 32:6862-35; Adib- Conquy et al. (1998) Int. Immunol. 10:341-6 and Beers et al. (2000) Clin. Can. Res. 6:2835-43.
As described above, the term “conservative substitution”, as used herein, refers to an amino acid substitution which would not disadvantageously affect or change the essential properties of a protein/polypeptide comprising the amino acid sequence. For example, a conservative substitution may be introduced by standard techniques known in the art such as site-directed mutagenesis and PCR-mediated mutagenesis. Conservative amino acid substitutions include substitutions wherein an amino acid residue is substituted with another amino acid residue having a similar side chain, for example, a residue physically or functionally similar (such as, having similar size, shape, charge, chemical property including
the capability of forming covalent bond or hydrogen bond, etc.) to the corresponding amino acid residue. The families of amino acid residues having similar side chains have been defined in the art. These families include amino acids having alkaline side chains (for example, lysine, arginine and histidine), amino acids having acidic side chains (for example, aspartic acid and glutamic acid), amino acids having uncharged polar side chains (for example, glycine, asparagine, glutamine, serine, threonine, tyrosine, cysteine, tryptophan), amino acids having nonpolar side chains (for example, alanine, valine, leucine, isoleucine, proline, phenylalanine, methionine), amino acids having P-branched side chains (such as threonine, valine, isoleucine) and amino acids having aromatic side chains (for example, tyrosine, phenylalanine, tryptophan, histidine). Therefore, a corresponding amino acid residue is preferably substituted with another amino acid residue from the same side-chain family. Methods for identifying amino acid conservative substitutions are well known in the art (see, for example, Brummell et al., Biochem. 32: 1180-1187 (1993); Kobayashi et al., Protein Eng. 12(10): 879-884 (1999); and Burks et al., Proc. Natl. Acad. Sci. USA 94: 412- 417 (1997), which are incorporated herein by reference).
In some embodiments, the anti-MUC16 antibody comprises at least one VHH, and the VHH comprises an amino acid sequence as set forth in any one of SEQ ID NOs: 4-5. In some embodiments, the anti-MUC16 antibody comprises a VHH consists of the amino acid sequence as set forth in any one of SEQ ID NOs: 4-5.
In some embodiments, the anti-MUC16 antibody is a chimeric antibody, comprising a VHH fused to an Fc region of human IgGl or IgG4, wherein the VHH comprises an amino acid sequence as set forth in any one of SEQ ID NOs: 4-5. In some embodiments, the anti-MUC16 antibody is a chimeric antibody comprising a VHH and an Fc region of human IgGl. Such antibodies are exemplified herein as “W306106-Pl lR2-lB8-uIgG1”. In some further embodiments, an anti- MUC16 antibody is a humanized antibody comprising a VHH and an Fc region of human IgGl. Such antibodies are exemplified herein as and “W306106-P1 lR2-lB8-z6-uIgG1”.
In some embodiments, the addition, deletion and/or substitution of at least one of the amino acids in the VHH region is not in any of the CDR sequences, but in the framework (FRW) sequences. For example, an antibody or antigen-binding portion thereof as described above may comprise one or more substitutions of the amino acids in the framework sequences, e.g. FRW1, FRW2, FRW3, and/or FRW4 of the VHH region.
In some embodiments, an antibody or antigen-binding portion thereof as provided herein comprises any suitable framework region (FRW) sequences, as long as the antigen-binding domains can specifically bind to MUC16.
As described above, an antibody or antigen-binding portion thereof may contain modification of one or more amino acids in the variable regions of the heavy chain and/or
light chain, including wherein the modification is a conservative substitution. It is understood in the art that certain conservative sequence modifications can be made which do not remove antigen binding. See, e.g., Brummell et al. ( 1993) Biochem 32:1180-8; de Wildt et al. ( 1997) Prot. Eng. 10:835-41; Komissarov et al. (1997) J. Biol. Chem. 272:26864- 26870; Hall et al. (1992) J. Immunol. 149:1605-12; Kelley and O’ Connell (1993) Biochem. 32:6862-35; Adib-Conquy et al. (1998) Int. Immunol. 10:341-6 and Beers et al. (2000) Clin. Can. Res. 6:2835-43.
In some embodiments, an antibody or antigen-binding portion thereof comprises a VHH domain comprising an amino acid sequence as set forth in any one of SEQ ID NOs: 4-5, and a Fc region, optionally the antibody or antigen-binding portion thereof comprises an amino acid sequence as set forth in SEQ ID NO: 6 or 7.
An antigen-binding domain of a MUC16-binding molecule is not limited to the VHH form and may adopt a variety of other formats, such as but not limited to, a Fab, a Fab', a F(ab')2, an Fv fragment, a single-chain antibody molecule (scFv). In some embodiments, an antigen-binding domain is a Fv fragment with a VH region and a VL region in separate chains held together by tight, non-covalent interactions.
Fc region comprising IgG constant domains
Anti-MUC16 antibodies and antigen-binding fragments provided herein further comprise an Fc region comprising one or more human IgG constant domains. A human IgG constant domain may be a human IgGl, IgG2, IgG3 or IgG4 constant domain, preferably a human IgGl constant domain. In some embodiments, the Fc region is a human IgGl Fc region, such as a wild-type Fc region or a Fc variant comprising one or more amino acid modifications (e.g. Leu234Ala/Leu235Ala or LALA) that alters the antibody-dependent cellular cytotoxicity (ADCC) or other effector fimctions.
In some embodiments, the Fc modification comprises a LALA mutation, e.g. mutations of L234A and L235A, according to EU numbering as in Kabat et al.. The Kabat numbering system is often used when referring to a residue in the variable domain (approximately residues 1-107 of the light chain and residues 1-113 of the heavy chain) (e.g., Kabat et al., Sequences of Immunological Interest. 5th Ed. Public Health Service, National Institutes of Health, Bethesda, Md. (1991)). The “EU numbering system” or “EU index” is generally used when referring to a residue in an immunoglobulin heavy chain constant region (e.g., the EU index reported in Kabat et al., supra). The “EU numbering as in Kabat” or “EU index as in Kabat” refers to the residue numbering of the human IgGl EU antibody. Unless stated otherwise herein, references to residue numbers in the constant domain of antibodies means residue numbering by the EU numbering system.
Nucleic Acid Molecules Encoding Antibodies of the Disclosure
In some aspects, the present disclosure provides a nucleic acid molecule comprising a nucleic acid sequence encoding a MUC16-binding molecule as disclosed herein, for example, encoding a single variable domain of a MUC16-binding molecule as disclosed herein. Nucleic acids of the disclosure can be obtained using standard molecular biology techniques.
A nucleic acid encoding a VHH region can be converted to a full-length heavy chain gene by operatively linking the VHH-encoding nucleic acid to another nucleic acid encoding one or more heavy chain constant regions (e.g. CHI, CH2 and CH3). The sequences of human heavy chain constant region genes are known in the art (see e.g., Kabat et al. ( 1991 ), supra) and DNA fragments encompassing these regions can be obtained by standard PCR amplification. A heavy chain constant region can be an IgGl, IgG2, IgG3, IgG4, IgA, IgE, IgM or IgD constant region, such as an IgGl constant region.
Once nucleic acids encoding VHH segments are obtained, these nucleic acids can be further manipulated by standard recombinant DNA techniques, for example to convert variable region genes to full-length antibody chain genes. In these manipulations, a VHH-encoding nucleic acid is operatively linked to another nucleic acid encoding another protein, such as an antibody constant region or a flexible linker. The term “operatively linked”, as used in this context, is intended to mean that two or more nucleic acids are joined such that the amino acid sequences encoded by the two or more nucleic acids remain in-frame.
In some embodiments, the disclosure is directed to a nucleic acid molecule, comprising a nucleic acid sequence encoding a single variable domain (e.g., VHH) of a MUC16-binding molecule as disclosed herein.
In some embodiments, the nucleic acid molecule comprises a nucleic acid sequence selected from the group consisting of:
(A) a nucleic acid sequence that encodes a VHH region as set forth in any one of SEQ ID NOs: 4-5;
(B) a nucleic acid sequence with at least 80% (e.g. at least 85%, 86%, 87%, 88%, 89%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99%) sequence identity to the nucleic acid sequence of (A); and
(C) a nucleic acid sequence that hybridizes under high stringency conditions to the complementary strand of the nucleic acid sequence of (A).
In some embodiments, provided herein is a nucleic acid molecule comprises a nucleic acid sequence encoding an anti-MUC16 single domain antibody comprising the amino acid sequence as set forth in SEQ ID NO: 4. In some embodiments, provided herein is a nucleic acid molecule comprises a nucleic acid sequence encoding an anti-MUC16 single domain antibody comprising the amino acid sequence as set forth in SEQ ID NO: 5.
In some embodiments, the percent identity is derived from the degeneracy of the genetic code, and the encoded protein sequences remain unchanged.
Exemplary high stringency conditions include hybridization at 45°C in 5X SSPE and 45% formamide, and a final wash at 65°C in 0.1 X SSC. It is understood in the art that conditions of equivalent stringency can be achieved through variation of temperature and buffer, or salt concentration as described Ausubel, et al. (Eds.), Protocols in Molecular Biology, John Wiley & Sons (1994), pp. 6.0.3 to 6.4.10. Modifications in hybridization conditions can be empirically determined or precisely calculated based on the length and the percentage of guanosine/ cytosine (GC) base pairing of the probe. The hybridization conditions can be calculated as described in Sambrook, et al, (Eds.), Molecular Cloning: A laboratory Manual. Cold Spring Harbor Laboratory Press: Cold Spring Harbor, New York (1989), pp. 9.47 to 9.51.
Host Cells
Host cells as disclosed in the present disclosure may be any cell which is suitable for expressing the antibodies of the present disclosure, for example, yeast, bacterial, plant and mammalian cells. Mammalian host cells for expressing the antibodies of the present disclosure include Chinese Hamster Ovary (CHO cells) (including dhfr CHO cells, described in Urlaub and Chasin, (1980) Proc. Natl. Acad. ScL USA 77:4216-4220, used with a DHFR selectable marker, e.g., as described in R. J. Kaufman and P. A. Sharp (1982) J. Mol. Biol. 159:601-621), 293F cells, NSO myeloma cells, COS cells and SP2 cells. In particular, for use with NSO myeloma cells, another expression system is the GS gene expression system disclosed in WO 87/04462, WO 89/01036 and EP 338,841. Also included are monkey kidney CV1 line transformed by SV40 (COS-7, ATCC CRL 1651); human embryonic kidney line (293 or 293 cells subcloned for growth in suspension culture, Graham et al., J. Gen Virol. 36:59 (1977)); baby hamster kidney cells (BHK, ATCC CCL 10); Chinese hamster ovary cells/-DHFR (CHO, Urlaub et al., 1980, Proc. Natl. Acad. Sci. USA 77:4216); mouse sertoli cells (TM4, Mather, 1980, Biol. Reprod. 23:243-251); monkey kidney cells (CV1 ATCC CCL 70); African green monkey kidney cells (VERO-76, ATCC CRL- 1587); human cervical carcinoma cells (HELA, ATCC CCL 2); canine kidney cells (MDCK, ATCC CCL 34); buffalo rat liver cells (BRL 3A, ATCC CRL 1442); human lung cells (W138, ATCC CCL 75); human liver cells (Hep G2, HB 8065); mouse mammary tumor (MMT 060562, ATCC CCL51); TRI cells (Mather et al., 1982, Annals N.Y. Acad. Sci. 383:44-68); MRC 5 cells; FS4 cells; mouse myeloma cells, such as NSO (e.g. RCB0213, 1992, Bio/Technology 10: 169) and SP2/0 cells (e.g. SP2/0-Agl4 cells, ATCC CRL 1581); rat myeloma cells, such as YB2/0 cells (e.g. YB2/3HL.P2.G11.16Ag.20 cells, ATCC CRL 1662); PER.C6 cells; and a human hepatoma line (Hep G2). CHO cells are one of the cell lines that can be used herein, with CHO-K1, DUK-
B11, CH0-DP12, CHO-DG44 (Somatic Cell and Molecular Genetics 12:555 (1986)), and Lecl3 being exemplary host cell lines. In the case of CHO-K1, DUK-B11, DG44 or CHO-DP12 host cells, these may be altered such that they are deficient in their ability to fucosylate proteins expressed therein. In some embodiments, the host cells herein are selected from CHO, CHO-S, HEK, HEK293, HEK-293F, Expi293F, PER.C6 or NSO cells or lymphocytic cells.
Suitable prokaryotes for this purpose include eubacteria, such as Gram-negative or Grampositive organisms, for example, Enterobacteriaceae such as Escherichia, e.g., E. coli, Enterobacter, Erwinia, Klebsiella, Proteus, Salmonella, e.g., Salmonella typhimurium, Serratia, e.g., Serratia marcescans, and Shigella, as well as Bacilli such as B. subtilis and B. licheniformis, Pseudomonas such as P. aeruginosa, and Streptomyces.
In addition to prokaryotes, eukaryotic microbes such as filamentous fungi or yeast are also suitable cloning or expression hosts for antibody-encoding vectors. Saccharomyces cerevisiae, or common baker’s yeast, is the most commonly used among lower eukaryotic host microorganisms. However, a number of other genera, species, and strains are commonly available and useful herein, such as Schizosaccharomyces pombe; Kluyveromyces hosts such as, e.g., K. lactis, K. fragilis (ATCC 12,424), K. bulgaricus (ATCC 16,045), K. wickeramii (ATCC 24,178), K. waltii (ATCC 56,500), K. drosophilarum (ATCC 36,906), K. thermotolerans, and K. marxianus; yarrowia (EP 402,226); Pichia pastoris (EP 183,070); Candida; Trichoderma reesia (EP 244,234); Neurosporacrassa; Schwanniomyces such as Schwanniomycesoccidentalis; and filamentous fungi such as, e.g., Neurospora, Penicillium, Tolypocladium, and Aspergillus hosts such as A. nidulans and A. niger.
When recombinant expression vectors encoding an antibody are introduced into mammalian host cells, the antibody is produced by culturing the host cells for a period of time sufficient to allow for expression of the antibody in the host cells or, secretion of the antibody into the culture medium in which the host cells are grown. Antibodies can be recovered from the culture medium using standard protein purification methods.
Pharmaceutical Compositions
In some aspects, the present disclosure provides a pharmaceutical composition comprising a MUC16-binding molecule as disclosed herein, for example, comprising a single variable domain (e.g., VHH) of a MUC16-binding molecule as disclosed herein and a pharmaceutically acceptable carrier. In some aspects, the present disclosure provides a pharmaceutical composition comprising a nucleic acid encoding a MUC16-binding molecule as disclosed herein, for example, comprising a single variable domain (e.g., VHH) of a MUC16-binding molecule as disclosed herein and a pharmaceutically acceptable carrier. In some aspects, the present disclosure provides a
pharmaceutical composition comprising a cell expressing a MUC16-binding molecule as disclosed herein, for example, comprising a single variable domain (e.g., VHH) of a MUC16-binding molecule as disclosed herein and a pharmaceutically acceptable carrier.
Components of the compositions
The pharmaceutical composition may optionally contain one or more additional components, including one or more pharmaceutically active ingredients, such as another antibody or a drug. The pharmaceutical compositions of the disclosure also can be administered in a combination therapy with, for example, another immune-stimulatory agent, anti-cancer agent, an antiviral agent, or a vaccine, including wherein the anti-MUC16 antibody enhances the immune response. A pharmaceutically acceptable carrier can include, for example, a pharmaceutically acceptable liquid, gel or solid carriers, an aqueous medium, a non-aqueous medium, an anti-microbial agent, isotonic agents, buffers, antioxidants, anesthetics, suspending/dispersing agent, a chelating agent, a diluent, adjuvant, excipient or a nontoxic auxiliary substance, other known in the art various combinations of components or more.
Suitable components of the pharmaceutical composition may include, for example, antioxidants, fillers, binders, disintegrating agents, buffers, preservatives, lubricants, flavorings, thickening agents, coloring agents, emulsifiers or stabilizers such as sugars and cyclodextrin. Suitable anti-oxidants may include, for example, methionine, ascorbic acid, EDTA, sodium thiosulfate, platinum, catalase, citric acid, cysteine, mercapto glycerol, thioglycolic acid, Mercapto sorbitol, butyl methyl anisole, butylated hydroxy toluene and/or propyl gallate. As disclosed in the present disclosure, a composition may comprise an antibody or an antigen-binding fragment of the present disclosure and also comprise one or more anti-oxidants such as methionine, to prevent or reduce a decrease in binding affinity, thereby enhancing antibody stability and extended shelflife. Thus, in some embodiments, the present disclosure provides a composition comprising one or more antibodies or antigen binding fragment thereof and one or more anti-oxidants such as methionine. The present disclosure further provides a variety of methods, wherein an antibody or antigen binding fragment thereof is mixed with one or more anti-oxidants, such as methionine, so that the antibody or antigen binding fragment thereof can be prevented from oxidation, to extend their shelflife and/or increased activity.
To further illustrate, pharmaceutical acceptable carriers may include, for example, aqueous vehicles such as sodium chloride injection, Ringer's injection, isotonic dextrose injection, sterile water injection, or dextrose and lactated Ringer's injection, nonaqueous vehicles such as fixed oils of vegetable origin, cottonseed oil, com oil, sesame oil, or peanut oil, antimicrobial agents at bacteriostatic or fungistatic concentrations, isotonic agents such as sodium chloride or dextrose, buffers such as phosphate or citrate buffers, antioxidants such as sodium bisulfate, local anesthetics such as procaine hydrochloride, suspending and dispersing agents such as sodium
carboxymethylcelluose, hydroxypropyl methylcellulose, or polyvinylpyrrolidone, emulsifying agents such as Polysorbate 80 (TWEEN-80), sequestering or chelating agents such as EDTA (ethylenediaminetetraacetic acid) or EGTA (ethylene glycol tetraacetic acid), ethyl alcohol, polyethylene glycol, propylene glycol, sodium hydroxide, hydrochloric acid, citric acid, or lactic acid. Antimicrobial agents utilized as carriers may be added to pharmaceutical compositions in multiple-dose containers that include phenols or cresols, mercurials, benzyl alcohol, chlorobutanol, methyl and propyl p-hydroxybenzoic acid esters, thimerosal, benzalkonium chloride and benzethonium chloride. Suitable excipients may include, for example, water, saline, dextrose, glycerol, or ethanol. Suitable non-toxic auxiliary substances may include, for example, wetting or emulsifying agents, pH buffering agents, stabilizers, solubility enhancers, or agents such as sodium acetate, sorbitan monolaurate, triethanolamine oleate, or cyclodextrin.
Administration, Formulation and Dosage
A pharmaceutical composition of the disclosure may be administered to a subject in need thereof, by various routes, including, but not limited to, oral, intravenous, intra-arterial, subcutaneous, parenteral, intranasal, intramuscular, intracranial, intracardiac, intraventricular, intratracheal, buccal, rectal, intraperitoneal, intradermal, topical, transdermal, and intrathecal, or otherwise by implantation or inhalation. The subject compositions may be formulated into preparations in solid, semi-solid, liquid, or gaseous forms; including, but not limited to, tablets, capsules, powders, granules, ointments, solutions, suppositories, enemas, injections, inhalants, and aerosols. The appropriate formulation and route of administration may be selected according to the intended application and therapeutic regimen.
Suitable formulations for enteral administration include hard or soft gelatin capsules, pills, tablets, including coated tablets, elixirs, suspensions, syrups or inhalations and controlled release forms thereof.
Formulations suitable for parenteral administration (e.g., by injection), include aqueous or non-aqueous, isotonic, pyrogen-free, sterile liquids (e.g., solutions, suspensions), in which the active ingredient is dissolved, suspended, or otherwise provided (e.g., in a liposome or other microparticulate). Such liquids may additional contain other pharmaceutically acceptable ingredients, such as anti-oxidants, buffers, preservatives, stabilisers, bacteriostats, suspending agents, thickening agents, and solutes which render the formulation isotonic with the blood (or other relevant bodily fluid) of the intended recipient. Examples of excipients include, for example, water, alcohols, polyols, glycerol, vegetable oils, and the like. Examples of suitable isotonic carriers for use in such formulations include Sodium Chloride Injection, Ringer's Solution, or Lactated Ringer's Injection. Similarly, the particular dosage regimen, including dose, timing and repetition, will depend on the particular individual and that individual's medical history, as well as empirical considerations such as pharmacokinetics (e.g., half-life, clearance rate, etc.).
Frequency of administration may be determined and adjusted over the course of therapy, and is based on reducing the number of proliferative or tumorigenic cells, maintaining the reduction of such neoplastic cells, reducing the proliferation of neoplastic cells, or delaying the development of metastasis. In some embodiments, the dosage administered may be adjusted or attenuated to manage potential side effects and/or toxicity. Alternatively, sustained continuous release formulations of a subject therapeutic composition may be appropriate.
It will be appreciated by one of skill in the art that appropriate dosages can vary from patient to patient. Determining the optimal dosage will generally involve the balancing of the level of therapeutic benefit against any risk or deleterious side effects. The selected dosage level will depend on a variety of factors including, but not limited to, the activity of the particular compound, the route of administration, the time of administration, the rate of excretion of the compound, the duration of the treatment, other drugs, compounds, and/or materials used in combination, the severity of the condition, and the species, sex, age, weight, condition, general health, and prior medical history of the patient. The amount of compound and route of administration will ultimately be at the discretion of the physician, veterinarian, or clinician, although generally the dosage will be selected to achieve local concentrations at the site of action that achieve the desired effect without causing substantial harmful or deleterious side-effects.
In general, a MUC16-binding molecule of the disclosure may be administered in various ranges. These include about 5 μg/kg body weight to about 100 mg/kg body weight per dose; about 50 μg/kg body weight to about 5 mg/kg body weight per dose; about 100 μg/kg body weight to about 10 mg/kg body weight per dose; and any values within the ranges. Other ranges include about 100 μg/kg body weight to about 20 mg/kg body weight per dose and about 0.5 mg/kg body weight to about 20 mg/kg body weight per dose. In some embodiments, the dosage is at least about 100 μg/kg body weight, at least about 250 μg/kg body weight, at least about 750 μg/kg body weight, at least about 3 mg/kg body weight, at least about 5 mg/kg body weight, at least about 10 mg/kg body weight.
In any event, an antibody or antigen binding portion thereof of the disclosure is preferably administered as needed to subjects in need thereof. Determination of the frequency of administration may be made by persons skilled in the art, such as an attending physician based on considerations of the condition being treated, age of the subject being treated, severity of the condition being treated, general state of health of the subject being treated and the like.
In some embodiments, the course of treatment involving a MUC16-binding molecule of the present disclosure will comprise multiple doses of the selected drug product over a period of weeks or months. For example, a MUC16-binding molecule of the present disclosure may be administered once every day, every two days, every four days, every week, every ten days, every two weeks, every three weeks, every month, every six weeks, every two months, every ten weeks
or every three months. In this regard, it will be appreciated that the dosages may be altered or the interval may be adjusted based on patient response and clinical practices.
Dosages and regimens may also be determined empirically for the disclosed therapeutic compositions in individuals who have been given one or more administration(s). For example, individuals may be given incremental dosages of a therapeutic composition produced as described herein. In some embodiments, the dosage may be gradually increased or reduced or attenuated based respectively on empirically determined or observed side effects or toxicity. To assess efficacy of the selected composition, a marker of the specific disease, disorder or condition can be followed as described previously. For cancer, these include direct measurements of tumor size via palpation or visual observation, indirect measurement of tumor size by x-ray or other imaging techniques; an improvement as assessed by direct tumor biopsy and microscopic examination of the tumor sample; the measurement of an indirect tumor marker (e.g., PSA for prostate cancer) or a tumorigenic antigen, a decrease in pain or paralysis; improved speech, vision, breathing or other disability associated with the tumor; increased appetite; or an increase in quality of life as measured by accepted tests or prolongation of survival. It will be apparent to one of skill in the art that the dosage will vary depending on the individual, the type of neoplastic condition, the stage of neoplastic condition, whether the neoplastic condition has begun to metastasize to other location in the individual, and the past and concurrent treatments being used.
Compatible formulations for parenteral administration (e.g., intravenous injection) may comprise a MUC16-binding molecule as disclosed herein in concentrations of from about 10 μg/ml to about 100 mg/ml. In some embodiments, the concentrations of the MUC16-binding molecule (e.g., antibody or the antigen binding portion thereof) will comprise 20 μg/ml, 40 μg/ml, 60 μg/ml, 80 μg/ml, 100 μg/ml, 200 μg/ml, 300, μg/ml, 400 μg/ml, 500 μg/ml, 600 μg/ml, 700 μg/ml, 800 μg/ml, 900 μg/ml or 1 mg/ml. In some embodiments, the concentrations of the MUC16-binding molecule (e.g., antibody or the antigen binding portion thereof) will comprise 2 mg/ml, 3 mg/ml, 4 mg/ml, 5 mg/ml, 6 mg/ml, 8 mg/ml, 10 mg/ml, 12 mg/ml, 14 mg/ml, 16 mg/ml, 18 mg/ml, 20 mg/ml, 25 mg/ml, 30 mg/ml, 35 mg/ml, 40 mg/ml, 45 mg/ml, 50 mg/ml, 60 mg/ml, 70 mg/ml, 80 mg/ml, 90 mg/ml or 100 mg/ml.
Applications of the Disclosure
The antibodies, antibody compositions and methods of the present disclosure have numerous in vitro and in vivo utilities and uses including, for example, detection of MUC16 or enhancement of immune response. For example, these molecules can be administered to cells in culture, in vitro or ex vivo, or to human subjects, e.g., in vivo, to enhance immunity in a variety of situations. The immune response can be modulated, for example, augmented, stimulated or up-regulated.
For example, the subjects include human patients in need of enhancement of an immune response. The methods are particularly suitable for treating human patients having a disorder that can be treated by augmenting an immune response (e.g., a T-cell mediated immune response). In some embodiments, the methods are particularly suitable for treatment of cancer in vivo. To achieve antigen-specific enhancement of immunity, anti-MUC16 antibodies can be administered together with an antigen of interest or the antigen may already be present in the subject to be treated (e.g., a tumor-bearing or virus-bearing subject). When antibodies to MUC 16 are administered together with another agent, the two can be administered in either order or simultaneously.
The present disclosure further provides methods for detecting the presence of human MUC 16 antigen in a sample, or measuring the amount of human MUC 16 antigen, comprising contacting the sample, and a control sample, for example, with a human monoclonal antibody, or an antigen binding portion thereof, which specifically binds to human MUC 16, under conditions that allow for formation of a complex between the antibody or portion thereof and human MUC 16. The formation of a complex is then detected, wherein a difference complex formation between the sample compared to the control sample is indicative of the presence of human MUC16 antigen in the sample. Moreover, anti-MUC16 antibodies of the disclosure can be used to purify human MUC 16 via immunoaffinity purification.
Treatment of disorders including cancers
In some aspects, the present disclosure provides a method of treating a disorder or a disease in a mammal, which comprises administering to the subject (for example, a human) in need of treatment a therapeutically effective amount of an anti-MUC16 antibody or antigen-binding portion thereof as disclosed herein. In some aspects, the present disclosure provides an anti- MUC16 antibody or antigen-binding portion thereof as disclosed herein for use in treating a disease or disorder. In some aspects, provided herein is a use of an anti-MUC16 antibody or antigen-binding portion thereof as disclosed herein for the manufacture of a medicament for the treatment of a disease or disorder. The disorder or disease may be a cancer.
A variety of cancers where MUC 16 is implicated, whether malignant or benign and whether primary or secondary, may be treated or prevented with a method provided by the disclosure. The cancers may include, but not limited to, ovary cancer, lung cancer, pancreas cancer, breast cancer, uterine cancer, fallopian tube cancer, primary peritoneum cancer, adrenal cancer, liver cancer, kidney cancer, bladder cancer, gastric cancer, cervical cancer, esophageal cancer, colorectal cancer, prostate cancer, thyroid cancer, sarcomas, glioblastomas and head and neck cancer or cancer of any other tissue that expresses MUC16.
Anti-MUC16 antibodies as disclosed herein can be used for treating lung cancers such as bronchogenic carcinoma, non-small cell lung cancer, squamous cell carcinoma, small cell carcinoma, large cell carcinoma, and adenocarcinoma, e.g. lung adenocarcinoma. The lung cancers
may be refractory, relapsed or resistant to a platinum based agent (e.g., carboplatin, cisplatin, oxaliplatin, topotecan) and/or a taxane (e.g., docetaxel, paclitaxel, larotaxel or cabazitaxel).
Cancers to be treated by anti-MUC16 antibodies as disclosed herein may also be large cell neuroendocrine carcinoma (LCNEC), medullary thyroid cancer, glioblastoma, neuroendocrine prostate cancer (NEPC), high-grade gastroenteropancreatic cancer (GEP) and malignant melanoma. Anti-MUC16 antibodies as disclosed herein may be used to treat neuroendocrine tumors (both NET and pNET) arising in the kidney, genitourinary tract (bladder, prostate, ovary, cervix, and endometrium), gastrointestinal tract (colon, stomach), thyroid (medullary thyroid cancer), and lung (small cell lung carcinoma and large cell neuroendocrine carcinoma).
Stimulation of an immune response
In some aspects, the disclosure also provides a method of enhancing (for example, stimulating) an immune response in a subject comprising administering to the subject a MUC16- binding molecule, for example, an anti-MUC16 antibody or an antigen binding portion thereof, of the disclosure such that an immune response in the subject is enhanced. In some aspects, the present disclosure provides an anti-MUC16 antibody or antigen-binding portion thereof as disclosed herein for use in enhancing (for example, stimulating) an immune response in a subject. In some aspects, provided herein is a use of an anti-MUC16 antibody or antigen-binding portion thereof as disclosed herein for the manufacture of a medicament for enhancing (for example, stimulating) an immune response in a subject. For example, in some embodiments, the subject is a mammal. In some embodiments, the subject is a human.
The term “enhancing an immune response” or its grammatical variations, means stimulating, evoking, increasing, improving, or augmenting any response of a mammal’s immune system. The immune response may be a cellular response (e.g. cell-mediated, such as cytotoxic T lymphocyte mediated) or a humoral response (e.g. antibody mediated response), and may be a primary or secondary immune response. Examples of enhancement of immune response include increased CD4+ helper T cell activity and generation of cytolytic T cells. The enhancement of immune response can be assessed using a number of in vitro or in vivo measurements known to those skilled in the art, including, but not limited to, cytotoxic T lymphocyte assays, release of cytokines (for example IL-2 production or IFN-y production), regression of tumors, survival of tumor bearing animals, antibody production, immune cell proliferation, expression of cell surface markers, and cytotoxicity. For example, methods of the disclosure are useful to enhance the immune response by a mammal when compared to the immune response by an untreated mammal or a mammal not treated using the methods as disclosed herein.
A MUC16-binding molecule may be used alone as a monotherapy, or may be used in combination with chemical therapies, radiotherapies, targeted therapies or cell immunotherapies etc.
Combined use with chemotherapies
A MUC16-binding molecule (e.g., an anti-MUC16 antibody) may be used in combination with chemotherapies, including, for example, an anti-cancer agent, a cytotoxic agent or chemotherapeutic agent.
The term “anti-cancer agent” or “anti-proliferative agent” means any agent that can be used to treat a cell proliferative disorder such as cancer, and includes, but is not limited to, cytotoxic agents, cytostatic agents, anti -angiogenic agents, debulking agents, chemotherapeutic agents, radiotherapy and radiotherapeutic agents, targeted anti-cancer agents, BRMs, therapeutic antibodies, cancer vaccines, cytokines, hormone therapies, radiation therapy and anti-metastatic agents and immunotherapeutic agents. It will be appreciated that, in some embodiments as discussed above, such anti-cancer agents may comprise conjugates and may be associated with the disclosed anti-MUC16 antibodies prior to administration. For example, in some embodiments selected anti-cancer agents will be linked to the unpaired cysteines of the engineered antibodies to provide engineered conjugates (e.g., antibody-drug conjugates) as set forth herein. Accordingly, such engineered conjugates are expressly contemplated as being within the scope of the present disclosure. In some embodiments, the disclosed anti-cancer agents will be given in combination with anti-MUC16 conjugates comprising a different therapeutic agent as set forth above.
As used herein the term “cytotoxic agent” means a substance that is toxic to the cells and decreases or inhibits the function of cells and/or causes destruction of cells. In some embodiments, the substance is a naturally occurring molecule derived from a living organism. Examples of cytotoxic agents include, but are not limited to, small molecule toxins or enzymatically active toxins of bacteria (e.g., Diptheria toxin, Pseudomonas endotoxin and exotoxin, Staphylococcal enterotoxin A), fungal (e.g., a-sarcin, restrictocin), plants (e.g., abrin, ricin, modeccin, viscumin, pokeweed anti-viral protein, saporin, gelonin, momoridin, trichosanthin, barley toxin, Aleurites fordii proteins, dianthin proteins, Phytolacca mericana proteins (PAPI, PAPII, and PAP-S), Momordica charantia inhibitor, curcin, crotin, saponaria officinalis inhibitor, gelonin, mitegellin, restrictocin, phenomycin, neomycin, and the tricothecenes) or animals, (e.g., cytotoxic RNases, such as extracellular pancreatic RNases; DNase I, including fragments and/or variants thereof).
For the purposes of the present disclosure a “chemotherapeutic agent” comprises a chemical compound that non-specifically decreases or inhibits the growth, proliferation, and/or survival of cancer cells (e.g., cytotoxic or cytostatic agents). Such chemical agents are often directed to intracellular processes necessary for cell growth or division, and are thus particularly effective against cancerous cells, which generally grow and divide rapidly. For example, vincristine
depolymerizes microtubules, and thus inhibits cells from entering mitosis. In general, chemotherapeutic agents can include any chemical agent that inhibits, or is designed to inhibit, a cancerous cell or a cell likely to become cancerous or generate tumorigenic progeny (e.g., TIC). Such agents are often administered, and are often most effective, in combination, e.g., in regimens such as CHOP or FOLFIRI.
Examples of anti-cancer agents that may be used in combination with MUC16-binding molecules (e.g., anti-MUC16 antibodies) of the present disclosure (either as a component of a site specific conjugate or in an unconjugated state) include, but are not limited to, alkylating agents, alkyl sulfonates, aziridines, ethylenimines and methylamelamines, acetogenins, a camptothecin, bryostatin, callystatin, CC-1065, cryptophycins, dolastatin, duocarmycin, eleutherobin, pancratistatin, a sarcodictyin, spongistatin, nitrogen mustards, antibiotics, enediyne antibiotics, dynemicin, bisphosphonates, esperamicin, chromoprotein enediyne antiobiotic chromophores, aclacinomysins, actinomycin, authramycin, azaserine, bleomycins, cactinomycin, carabicin, carminomycin, carzinophilin, chromomycinis, dactinomycin, daunorubicin, detorubicin, 6-diazo- 5-oxo-L-norleucine, ADRIAMYCIN® doxorubicin, epirubicin, esorubicin, idarubicin, marcellomycin, mitomycins, mycophenolic acid, nogalamycin, olivomycins, peplomycin, potfiromycin, puromycin, quelamycin, rodorubicin, streptonigrin, streptozocin, tubercidin, ubenimex, zinostatin, zorubicin; anti-metabolites, erlotinib, vemurafenib, crizotinib, sorafenib, ibrutinib, enzalutamide, folic acid analogues, purine analogs, androgens, anti-adrenals, folic acid replenisher such as frolinic acid, aceglatone, aldophosphamide glycoside, aminolevulinic acid, eniluracil, amsacrine, bestrabucil, bisantrene, edatraxate, defofamine, demecolcine, diaziquone, elfomithine, elliptinium acetate, an epothilone, etoglucid, gallium nitrate, hydroxyurea, lentinan, lonidainine, maytansinoids, mitoguazone, mitoxantrone, mopidanmol, nitraerine, pentostatin, phenamet, pirarubicin, losoxantrone, podophyllinic acid, 2- ethylhydrazide, procarbazine, PSK® polysaccharide complex (JHS Natural Products, Eugene, OR), razoxane; rhizoxin; sizofiran; spirogermanium; tenuazonic acid; triaziquone; 2,2',2"-trichlorotriethylamine; trichothecenes (especially T-2 toxin, verracurin A, roridin A and anguidine); urethan; vindesine; dacarbazine; mannomustine; mitobronitol; mitolactol; pipobroman; gacytosine; arabinoside (“Ara-C”); cyclophosphamide; thiotepa; taxoids, chloranbucil; GEMZAR® gemcitabine; 6-thioguanine; mercaptopurine; methotrexate; platinum analogs, vinblastine; platinum; etoposide (VP- 16); ifosfamide; mitoxantrone; vincristine; NAVELBINE® vinorelbine; novantrone; teniposide; edatrexate; daunomycin; aminopterin; xeloda; ibandronate; irinotecan (Camptosar, CPT-11), topoisomerase inhibitor RFS 2000; difluorometlhylomithine; retinoids; capecitabine; combretastatin; leucovorin; oxaliplatin; inhibitors of PKC -alpha, Raf, H-Ras, EGFR and VEGF- A that reduce cell proliferation and pharmaceutically acceptable salts, acids or derivatives of any of the above. Also included in this definition are anti-hormonal agents that act to regulate or inhibit
hormone action on tumors such as anti-estrogens and selective estrogen receptor modulators, aromatase inhibitors that inhibit the enzyme aromatase, which regulates estrogen production in the adrenal glands, and anti-androgens; as well as troxacitabine (a 1,3- dioxolane nucleoside cytosine analog); antisense oligonucleotides, ribozymes such as a VEGF expression inhibitor and a HER2 expression inhibitor; vaccines, PROLEUKIN® rIL-2; LURTOTECAN® topoisomerase 1 inhibitor; ABARELIX® rmRH; Vinorelbine and Esperamicins and pharmaceutically acceptable salts, acids or derivatives of any of the above.
Combined use with radiotherapies
The present disclosure also provides for the combination of a MUC16-binding molecule with a radiotherapy (e.g., any mechanism for inducing DNA damage locally within tumor cells such as gamma-irradiation, X-rays, UV-irradiation, microwaves, electronic emissions and the like). Combination therapy using the directed delivery of radioisotopes to tumor cells is also contemplated, and the disclosed MUC16-binding molecules may be used in connection with a targeted anti-cancer agent or other targeting means. Typically, radiation therapy is administered in pulses over a period of time from about 1 to about 2 weeks. The radiation therapy may be administered to subjects having head and neck cancer for about 6 to 7 weeks. Optionally, the radiation therapy may be administered as a single dose or as multiple, sequential doses.
Diagnosis
The disclosure provides in vitro and in vivo methods for detecting, diagnosing or monitoring proliferative disorders and methods of screening cells from a patient to identify tumor cells including tumorigenic cells. Such methods include identifying an individual having cancer for treatment or monitoring progression of a cancer, comprising contacting the patient or a sample obtained from a patient (either in vivo or in vitro) with an anti-MUC16 antibody as described herein and detecting presence or absence, or level of association, of the antibody to bound or free target molecules in the sample. In some embodiments, the anti-MUC16 antibody will comprise a detectable label or reporter molecule as described herein.
In some embodiments, the association of an anti-MUC16 antibody with particular cells in the sample can denote that the sample may contain tumorigenic cells, thereby indicating that the individual having cancer may be effectively treated with an anti-MUC16 antibody as described herein.
Samples can be analyzed by numerous assays, for example, radioimmunoassays, enzyme immunoassays (e.g. ELISA), competitive-binding assays, fluorescent immunoassays, immunoblot assays, Western Blot analysis and flow cytometry assays. Compatible in vivo theragnostic or diagnostic assays can comprise art recognized imaging or monitoring techniques, for example, magnetic resonance imaging, computerized tomography (e.g. CAT scan), positron tomography (e.g., PET scan), radiography, ultrasound, etc., as would be known by those skilled in the art.
Pharmaceutical packs and kits
Pharmaceutical packs and kits comprising one or more containers, comprising one or more doses of a MUC16-binding molecule are also provided. In some embodiments, a unit dosage is provided wherein the unit dosage contains a predetermined amount of a composition comprising, for example, a MUC16-binding molecule, with or without one or more additional agents. In some embodiments, such a unit dosage is supplied in single-use prefilled syringe for injection. In some embodiments, the composition contained in the unit dosage may comprise saline, sucrose, or the like; a buffer, such as phosphate, or the like; and/or be formulated within a stable and effective pH range. Alternatively, in some embodiments, a composition may be provided as a lyophilized powder that may be reconstituted upon addition of an appropriate liquid, for example, sterile water or saline solution. In some embodiments, the composition comprises one or more substances that inhibit protein aggregation, including, but not limited to, sucrose and arginine. Any label on, or associated with, the container(s) indicates that the enclosed composition is used for treating the neoplastic disease condition of choice.
The present disclosure also provides kits for producing single-dose or multi-dose administration units of a MUC16-binding molecule and, optionally, one or more anti-cancer agents. The kit comprises a container and a label or package insert on or associated with the container. Suitable containers include, for example, bottles, vials, syringes, etc. The containers may be formed from a variety of materials such as glass or plastic and contain a pharmaceutically effective amount of the disclosed MUC16-binding molecules in a conjugated or unconjugated form. In some embodiments, the container(s) comprise a sterile access port (for example the container may be an intravenous solution bag or a vial having a stopper pierceable by a hypodermic injection needle). Such kits will generally contain in a suitable container a pharmaceutically acceptable formulation of a MUC16-binding molecule in a conjugated or unconjugated form and, optionally, one or more anti-cancer agents in the same or different containers. The kits may also contain other pharmaceutically acceptable formulations, either for diagnosis or combined therapy. For example, in addition to a MUC16-binding molecule of the disclosure such kits may contain any one or more of a range of anti-cancer agents such as chemotherapeutic or radiotherapeutic drugs; anti-angiogenic agents; anti-metastatic agents; targeted anti-cancer agents; cytotoxic agents; and/or other anti-cancer agents.
For example, the kits may have a single container that contains a MUC16-binding molecule, with or without additional components, or they may have distinct containers for each desired agent. Where combined therapeutics are provided for conjugation, a single solution may be pre-mixed, either in a molar equivalent combination, or with one component in excess of the other. Alternatively, the conjugates and any optional anti-cancer agent of the kit may be maintained
separately within distinct containers prior to administration to a patient. The kits may also comprise a second/third container means for containing a sterile, pharmaceutically acceptable buffer or other diluents such as bacteriostatic water for injection (BWFI), phosphate-buffered saline (PBS), Ringer's solution and dextrose solution.
When the components of the kit are provided in one or more liquid solutions, the liquid solution is preferably an aqueous solution, for example, a sterile aqueous or saline solution. However, the components of the kit may be provided as dried powder(s). When reagents or components are provided as a dry powder, the powder can be reconstituted by the addition of a suitable solvent. It is envisioned that the solvent may also be provided in another container.
As indicated briefly above the kits may also contain a means by which to administer a MUC16-binding molecule and any optional components to a patient, e.g., one or more needles, I.V. bags or syringes or other such like apparatus, from which the formulation may be injected or introduced into the animal or applied to a diseased area of the body. The kits of the present disclosure will also typically include a means for containing the vials, or such like, and other component in close confinement for commercial sale, such as, e.g., injection or blow-molded plastic containers into which the desired vials and other apparatus are placed and retained.
Sequence Listing Summary
Appended to the instant application is a sequence listing comprising a number of amino acid sequences. The following Tables A-F provide a summary of the included sequences. The exemplary antibodies may be referred to as “WuXi Bio Lead” antibodies in the present disclosure.
EXAMPLES
The present disclosure, thus generally described, will be understood more readily by reference to the following Examples, which are provided by way of illustration and are not intended to be limiting of the present disclosure. The Examples are not intended to represent that the experiments below are all or the only experiments performed.
Example 1: Preparation of Antigens, Benchmark Antibodies and Cell Lines
1.1 Generation of antigens
The amino acid sequence encoding the extracellular domain of human MUC16 (hProl) (UniProt: Q8WXI7, residues 14192 to 14451), human MUC16 (hPro2) (UniProt: Q8WXI7, residues 14307 to 14451), mouse MUC16 (mProl) (UniProt: A0A140LJ72, residues 8158 to 8422), mouse MUC16 (mPro2) (UniProt: A0A140LJ72, residues 8273 to 8422), cynomolgus monkey MUC16 (cProl) (NCBI: XP 015296314.1, residues 14029 to 14292), cynomolgus monkey MUC16 (cPro2) (NCBI: XP O 15296314.1, residues 14145 to 14292), were first codon optimized for mammalian expression, and then synthesized by GENEWIZ (Su Zhou, CHINA). hProl includes human MUC16 55th & 56th SEA domain, and the juxtamembrane domain. cProl includes the cynomolgus counterpart of hProl. hPro2 includes human 56th SEA domain, and the juxtamembrane domain. cPro2 includes the cynomolgus counterpart of hPro2. The DNA segment was then sub-cloned into the pcDNA3.3 or pcDNA3.4 expression vector with 6x His or mFc (mIgG2a) at the C -terminal.
The plasmids encoding the antigens were transfected into Expi293 cells at 100 mL, 200 mL, 300 mL and 500 mL scale. Cells were cultured for 5 days and the supernatant was collected for protein purification using HisTrap excel columns, Protein A column, and SEC column. The antigen concentration was detected by Nano Drop at 280 nm. The purity of the antigen was analyzed by SDS-PAGE and SEC-HPLC. The antigens were stored at -80 °C.
1.2 Preparation of benchmark antibodies (BMKs)
Three anti-MUC16 antibodies were used as controls, namely, W3XX106-cAb4 (MUC16 binding monoclonal antibody prepared from the MUC16-binding arm of REGN4018), W3XX106- BMK4 (9B11 antibody, WO2011119979 A2) and W3XX106-BMK5 (Parental antibody of DMUC5754A). The amino acid sequences encoding the variable domains of W3XX106-cAb4 (WO2018/067331), W3XX106-BMK4 (WO2011/119979) and W3XX106-BMK5 (W02007/001851) were first codon optimized for mammalian expression and then synthesized by GENEWIZ (Su Zhou, CHINA). The DNA segments were then sub-cloned into pcDNA3.4 expression vectors with constant region of human IgGl . The plasmids containing VH and VL gene were co-transfected into Expi293 cells. Cells were cultured for 5 days and supernatant was collected for protein purification using Protein A column. The obtained antibodies were analyzed by SDS-PAGE and SEC-HPLC, and then stored at -80 °C.
1.3 Cell Pool/Line Generation
Human MUC16 56th SEA domain expressing cell pool W3XX106-SK-OV-3.hPro2.pool was generated. Briefly, SK-OV-3 cells were transfected with pcDNA3.3 expression vector containing the DNA sequence of human 56th SEA domain, transmembrane domain and intracellular domain, using Lipofectamine 2000 transfection kit according to manufacturer’s protocol. 48-72 hours post transfection, the transfected cells were cultured in medium containing 15 μg/mL Blasticidin for selection and tested for MUC16 56th SEA domain expression. The Cell pool was enriched by BD FACS Melody™ cell sorter.
Human MUC16 55th & 56th SEA domain expressing cell line W3XX106-SK-OV- 3.hProl.FL.A9 was generated. Briefly, SK-OV-3 cells were transfected with pcDNA3.3 expression vector containing the DNA sequence of human MUC16 55th & 56th SEA domain, transmembrane domain and intracellular domain, using Lipofectamine 2000 transfection kit according to manufacturer’s protocol. 48-72 hours post transfection, the transfected cells were cultured in medium containing 15 μg/mL Blasticidin for selection and tested for MUC16 55th & 56th SEA domain expression. The MUC16-expressing cell line was obtained by BD FACS Melody™ cell sorter.
Cynomolgus monkey MUC16 55th & 56th SEA domain expressing cell line W3XX106- SK-OV-3.cProl.FL.El was generated. Briefly, SK-OV-3 cells were transfected with pcDNA5 expression vector containing full-length of DNA sequence of cynomolgus monkey MUC16 55th & 56th SEA domain, transmembrane domain and intracellular domain, using Lipofectamine 2000 transfection kit according to manufacturer’s protocol, respectively. 72 hours post transfection, the transfected cells were cultured in medium containing 15 μg/mL Blasticidin for selection and tested for MUC16 expression. The MUC16-expressing cell line was obtained by BD FACSMelody™ cell sorter and limiting dilution.
Example 2: Generation and humanization of VHHs
2,1 Alpaca immunity and anti-MUC16 VHH generation
Anti-MUC16 VHHs were generated by immunization of Camelidae animals and phage display technology. Briefly, Alpacas (Vicugna pacos) were subcutaneously immunized with W3XX106-hProl.ECD.mFc and W3XX106-hPro2.ECD.mFc. After immunization, peripheral blood was collected for construction of phage library displaying VHH fragments. After biopanning with corresponding target MUC16 proteins and W3XX106-SK-OV3-hProl cells, the positive VHH clones binding to MUC16 were selected. The animal information was showed in Table 2.
The positive E. coli clones selected by target specific binding ELISA and FACS with E. coli supernatants were sent to Qingke (Shanghai, China) for nucleotide sequencing of VHH gene. The sequencing results were analyzed using Vector NTI (Thermo Fisher Scientific, USA). After a series of panning and screening, W306106-P11R2-1B8 clone was selected based on the unique sequence and good binding data as shown in Table 3.
2,3 Design of humanization and PTM removal variants
The VH domain sequences of W306106-P11R2-1B8 are aligned to the human germline sequence repertoires of the VH at IMGT. The human germline sequence of the VH domain with the least number of amino acid differences in framework with respect to the VH domain sequence of W306106-P11R2-1B8 was selected as the humanization template of the VH domain. CDRs of W306106-P11R2-1B8 VHH domain were grafted into the framework of the humanization template to constitute the germlined VHH domain sequence.
To restore the antigen binding capacity which is usually lost upon CDR grafting, several “back mutation” positions in the framework are empirically selected to convert the amino acids in the germlined sequence to their counterpart amino acids in the original murine sequence. A set of humanization variants are empirically designed to explore different combinations of these selected back mutation sites. One humanized VHH, designated as W306106-P11R2-1B8-Z6, was selected.
2,4 Humanization variants production
DNA sequence of humanization variants were subcloned into modified pcDNA3.4 expression vectors with constant region of human IgGl. The plasmids were transfected into Expi293 cells. Cells were cultured for 5 days and supernatant was collected for SPR ranking assay
or protein purification. The purified antibodies were analyzed by SDS-PAGE and SEC, and then stored at -80 °C.
Example 3: Generation and characterization of W306106-PllR2-lB8-uIgGl (prehumanization antibody)
3.1 Construction of chimeric VHH-Fc
The W306106-P11R2-1B8 was converted to VHH-Fc (hlgGl) fusion antibody. Briefly, the gene of W306106-P11R2-1B8 was PCR amplified from the pET-bac vectors using VHH-specific cloning primers containing appropriate restriction sites then cloned by fusion into a modified human hlgGl expression pcDNA3.3 vector to create corresponding clone of VHH-Fc (hlgGl) chimeric antibody. Expi293 cells were transiently transfected with the vector for antibody expression. The cell culture supernatant containing antibody was harvested and purified using Protein A chromatography. The generated antibody was named as “W306106-P11R2-1B8- ulgGl”. The obtained antibody was analyzed by SDS-PAGE and HPLC-SEC, and then stored at -80 °C.
When transiently transfected to Expi293 cells and purified by Protein A column, the yield of W306106-P1 !R2-lB8-uIgG1 was 574 mg/L and the purity was 99.7 % by SEC-HPLC (Figure 1).
3,2 Thermal Stability Test by DSF
Tm (melting temperature) of each antibody was investigated using QuantStudio® 7 Flex Real-Time PCR system. 19 μL antibody solution was mixed with 1 μL 80 x SYPRO Orange solution (Invitrogen) in the 96 wells plate. Each sample was tested in duplicate wells. The plate was sealed with the Optical Adhesive Film and centrifuged at 3,000 rpms for 5 minutes to remove any air bubbles. The plate was heated from 26 °C to 95 °C at a rate of 0.9 °C/minute, and the resulting fluorescence data was collected. The negative derivatives of the fluorescence changes with respect to different temperatures were calculated, and the maximal value was defined as
melting temperature Tm. If a protein has multiple unfolding transitions, the first two Tms were reported, named as Tml and Tm2. Data collection and Tm calculation were conducted automatically by the QuantStudio® Real Time PCR software software (vl.3). The results were shown in Figure 2 and Table 5 below.
3.3 Affinity-Capture Self-Interaction Nanoparticle Spectroscopy (AC-SINS)
Self-Interaction of antibodies was investigated using AC-SINS method. Goat anti-human IgG Fc antibodies (capture) and ChromPure Goat IgG antibodies (non-capture) were buffer exchanged into 20 mM NaAc (pH 4.3), and then the concentration was normalized to 0.4 mg/mL, respectively. Capture and non-capture IgG solution were mixed with a volume ratio of 4: 1. Then the IgG mixture was mixed with gold nanoparticle (AuNP) solution at a volume ratio of 1 : 9, and incubated at room temperature (RT) overnight. To block the empty sites on the AuNP, thiolated PEG was added into the mixture to a final concentration 0.1 pM and incubated at RT for 1 hour. The mixture was filtered through a 0.22 pm PVDF membrane, and 1/10 of the starting volume of 50 mM PB, pH 7.0 buffer was used to elute the coated AuNP particles from the membrane to obtain lOx AuNP solution. All antibodies were diluted to 0.1 mg/mL before testing. 90 μL test antibody solution (0.1 mg/mL) was mixed with 20 μL 10 x AuNP and incubated at RT for 2 hours in a 96-well polypropylene plate. After incubation, 100 μL of the antibody- AuNP mixture was transferred into a 384-well polystyrene UV transparent plate. Absorbance data were collected from 510 to 570 nm at an increment of 1 nm. AXmax value was calculated by subtraction the max absorbance value of samples with that of PBS.
BV particles were obtained by infecting Sf9 insect cells with a recombinant expressing green fluorescent protein (Bac-to-Bac, Thermofisher). Infected cultures were incubated for 40 hours at 27 °C under agitation (200 rpms), harvested and cells removed by centrifugation at 300 g for 5 minutes. The virus in the supernatant was pelleted by centrifugation at 300 g for 4 hours at 4 °C, resuspended in PBS buffer layered on a 4 mL 35 % (w/v) sucrose cushion in PBS and centrifuged at 25000 rpm for 4 hours at 4 °C. The supernatant with debris was discarded, the virus pellet gently rinsed once with PBS, resuspended in 1.2 mL of PBS with protease inhibitor cocktail (Roche) and stored at 4 °C for up to 4 months.
Example 4: Generation and characterization of W306106-PllR2-lB8-z6-uIgG1 (after humanization)
4,1 Construction of W306106-P1 !R2-lB8-z6-uIgGl
DNA sequence encoding the variable region of VHH antibody was subcloned into modified pcDNA3.4 expression vector with constant region of human IgGl. The plasmid was transfected into Expi293 cells. Cells were cultured for 5 days and supernatant was collected for protein purification using Protein A column. The obtained antibody was analyzed by SDS-PAGE and SEC, and then stored at -80 °C.
When transiently transfected to Expi293 cells and purified by Protein A column, the yield of W306106-P11R2-lB8-z6-uIgG1 was 636.67 mg/L and the purity was 96.64 % by SEC-HPLC (Figure 3). Different batches of W306106-P 11R2- lB8-z6-uIgG1 were also used for developability test. The SDS-PAGE & SEC-HPLC results of a different batch of W306106-P11R2-1B8-Z6- ulgGl were shown in Figure 4. The purification summary of humanized VHH-Fc antibodies was listed in Table 8.
4,2 Affinity assay of humanization variants
The koff ranking is performed on a Biacore 8K surface plasmon resonance (SPR) equipment.
CM5 sensor chips are first activated by 400 mM EDC and 100 mM NHS (GE) for 420 sec at the flow rate of 10 μL /minute. 30 μg/mL of anti-human Fc IgG (Jackson) in 10 mM NaAc (pH 4.5) is then injected to channel 1 to 8 at the flow rate of 10 μL/minute for 420 sec. The chips are then deactivated by 1 M ethanolamine-HCl (GE) at the flow rate of 10 μL/minute for 420 sec.
The SPR ranking of humanization variants was showed in Table 9.
Table 9. SPR ranking result of humanization variants supernatant
All variants have the same human IgGl isotype. koff labeled with * is approaching or outside the limits that can be measured by the instrument.
4,3 Appearance test
The sample was centrifuged at 12000 rpm for 3 minutes at 4 °C, filtered by using a 0.1 pm filter, concentrated to 5-6 mg/mL and observed the appearance.
The data indicated the Ab showed colorless and particle free as shown in Table 10.
4.4 Stress test
The concentration of each sample was measured by Nanodrop2000 with 2 μL sample for 3 times and then diluted to 1 mg/mL by the sample storage buffer (PBS). Eight tubes of 300 μL antibody solution were prepared for each sample. Each tube was placed in its respective temperature conditions (either 4 °C or 40 °C or -80 °C) and stored for 14 days, or freezed (-80 °C)/thawed (25 °C) for 3 and 5 cycles. After the stress treatment, samples were centrifuged at 12,000 rpm for 3 minutes at 4 °C and observed visually. The protein concentration was measured by Nanodrop2000 and the data was recorded. Purity of each antibody was detected by Agilent 1260 Infinity II system with TSKgel G3000SWXL column. 25 μL sample was injected into the column, and seperated with a flow rate of 1 mL/minutes for 20 minutes. The running buffer was 50 mM sodium phosphate, 150 mM NaCl, pH 7.0. The peak retention was detected with UV light of the wavelength at 280 nm. The purity of each antibody was analyzed with SEC-HPLC analysis method to integrate all peak areas from 4.5 minutes to 10.5 minutes. The OpenLab CDS Workstation was used as the operation and analysis software (v2.3.0.443).
The freeze/thaw cycling test result summary of W306106-P1 !R2-lB8-z6-uIgG1 was shown in Table 11. The Accelerated thermal stability test result summary of W306106-P11R2-1B8-Z6- ulgGl was shown in Table 12. The SEC-HPLC profiles ofW306106-PHR2-lB8-z6-uIgG1 after 5 Freeze/Thaw cycles and W306106-P1 lR2-lB8-z6-uIgG1 after 14 days in 40 °C was showed in Figure 5 and Figure 6.
Note: Colorless (CL), slight-opalescent (SO), particle-free (PF), particle-observed (PO).
Table 12. Accelerated thermal stability test result summary
4.5 Thermal stability test by DSF
Tm (melting temperature) of each antibody was investigated using QuantStudio® 7 Flex Real-Time PCR system. 19 μL of antibody solution was mixed with 1 μL of 80 x SYPRO Orange solution (Invitrogen) in the 96 well plate. The plate was sealed with the Optical Adhesive Film and centrifuged at 3,000 rpm for 5 minutes at 4 °C to remove any air bubbles. The plate was heated from 26 °C to 95 °C at a rate of 0.9 °C/minutes, and the resulting fluorescence data was collected. The negative derivatives of the fluorescence changes with respect to different temperatures were calculated, and the maximal value was defined as melting temperature Tm. If a protein has multiple unfolding transitions, the first two Tm were reported, named as Tm1 and Tm2. Data collection and Tm calculation were conducted automatically by the QuantStudio® Real Time PCR software software (vl.3).
Tml value of W306106-P1 !R2-lB8-z6-uIgG1 is in the normal range. The DSF profile and
DSF test summary was showed in Figure 7 and Table 13.
4,6 Determination of diffusion interaction parameter (kD) by PLS
The kD measurement was investigated using DynaPro Plate Reader III (Wyatt Technology).
During the sample preparation process, the appearance of samples was observed at thawing,
filtration and concentration by visual inspection. The purity of a dose point (15 mg/mL) will be tested after concentration. Samples were first filtered with 0.1 pm filter. Samples were then concentrated to over 20 mg/mL and diluted with corresponding buffer to a final concentration at 2.5, 5, 10, 15, and 20 mg/mL. 7.5 μL sample solution was then added to 1536 well microplate. The plate was sealed with the Clear Seal Film, and centrifuged at 3,000 rpm for 5 minutes at 4 °C to let the sample down to the bottom of the well. Each sample was tested in duplicate wells. The plate was put into the corresponding position and data collection was performed by the DYNAMICS operation software (v7.8.1.3). 5 acquisitions were collected for each protein sample while each acquisition time was 5 s. For each measurement, the diffusion coefficient was determined and plotted against protein concentration. kD values were calculated automatically by the software.
The DLS-kD test result summary of humanized VHH-Fc antibody was shown in Figure 8 and Table 14.
4.7 Affinity-Capture Self-Interaction Nanoparticle Spectroscopy (AC-SINS)
Self-Interaction of antibodies was investigated using AC-SINS method. Goat anti-human IgG Fc antibodies (capture) and ChromPure Goat IgG antibodies (non-capture) were buffer exchanged into 20 mM NaAc (pH 4.3), and then the concentration was normalized to 0.4 mg/mL, respectively. Capture and non-capture IgG solution were mixed with a volume ratio of 4: 1. Then the IgG mixture was mixed with gold nanoparticle (AuNP) solution at a volume ratio of 1:9, and incubated at room temperature (RT) overnight. To block the empty sites on the AuNP, thiolated PEG was added into the mixture to a final concentration 0.1 pM and incubated at RT for 1 hour. The mixture was filtered through a 0.22 pm PVDF membrane, and 1/ 10 of the starting volume of 50 mM PB, pH 7.0 buffer was used to elute the coated AuNP particles from the membrane to obtain 10 x AuNP solution. All antibodies were diluted to 0.1 mg/mL before testing. 90 μL of test antibody solution (0.1 mg/mL) was mixed with 20 μL 10 x AuNP and incubated at RT for 2 hours in a 96-well polypropylene
plate. After incubation, 100 μL of the antibody- AuNP mixture was transferred into a 384-well polystyrene UV transparent plate. Absorbance data were collected from 510 to 570 nm at an increment of 1 nm. AXmax value was calculated by subtraction the max absorbance value of samples with that of PBS.
Antibodies with low Almax value probably have low self-interaction propensity as showed in Table 15.
4,8 Non-specific binding
4,8,1 Full panel non-specific binding
ELISA assay: 96-well high binding plates (Nunc-Immuno Plate, Thermo Scientific) were coated with 2 μg/mL HIS-tagged antigen and blocked with 2 % BSA-PBS. 100 μL antibodies (10 μg/mL) were added into the antigen-coated well and incubated at room temperature for 2 hours, and the binding of antibodies to the immobilized antigens on the plate was measured using Goat Anti-Human IgG-Fc-HPR labelled antibody. The HRP signal was detected by adding TMB peroxidase substrate, and the reaction was stopped after 12 minutes using 2 M HC1.
FACS assay: cells were transferred into 96-well U-bottom plates (BD) at a density of 1 x 105 cells/well and centrifuged before removing the supernatant. 100 μL antibodies (10 μg/mL) were used to re-suspend cells and incubated at 4 °C for 1 hour. The secondary antibody, PE-conjugated Goat Anti-Human IgG Fc fragment was diluted to 5 μg/mL, then added to the re-suspended cells and incubated at 4 °C for 30 minutes. Washing steps were performed twice followed by centrifugation at x 200 g at 4 °C. Finally, the cells were re-suspended in 1% BSA-1 x PBS and fluorescence intensity was measured by flow cytometry (BD Canto II) and analyzed by FlowJo.
The ELISA and FACS panel screening data and BVP data in Tables 16-18 showed that W306106-P1 !R2-lB8-z6-uIgG1 has no non-specific binding.
4,8,2 BVP non-specific binding
BVPs were incubated on ELISA plates by adding 100 mL of 1 % BVP stock (in 50 mM sodium carbonate, pH 9.6) per well and incubated at 4 °C for 24 hours. The next day, unbound BVPs were aspirated from the wells. All remaining steps were performed at room temperature. 100 μL of blocking buffer (PBS with 0.5 % BSA) was added to the 96-well and incubated for 1 hour prior to three washes with 300 mL of PBS. Next, 100 μL of 1 pM primary antibodies (i.e. test antibodies) in blocking buffer was added to the wells and incubated for 1 hour followed by six washes with 100 μL of PBS. 100 μL of anti-Human-IgG-HRP was added to the wells and incubated for 1 hour followed by 6 washes as before. Finally, 100 μL of TMB substrate was added to each well and incubated for 15 minutes. The reactions were stopped by adding 100 μL of 2 M
sulfuric acid to each well. The absorbance was read at 450 nm and BVP score were determined by normalizing absorbance by control wells with no test antibody.
Example 5: Generation and characterization of W306106-PllR2-lB8-z6 (humanized VHH)
5.1 Construction of humanized VHHs
DNA sequence encoding the variable region of VHH antibodies were subcloned into modified pcDNA3.4 expression vectors. The plasmids were transfected into Expi293 cells. Cells were cultured for 5 days and supernatant was collected for protein purification using Protein A3 column. The obtained antibodies were analyzed by SDS-PAGE and SEC-HPLC, and then stored at -80 °C.
When transiently transfected to Expi293 cells and purified by Protein A3 column, the yield of W306106-Pl lR2-lB8-z6 was 230 mg/L and the purity was 99.81 % by SEC-HPLC (Figure 9 and Table 19).
W306106-P11R2-1B8-Z6 was further concentrated by ultrafiltration (Table 20). The purity of the VHHs were determined by SDS-PAGE & SEC-HPLC (Figure 10).
5.2 Appearance test
The appearance of samples was observed at thawing, filtration and concentration by visual inspection. The samples were centrifuge with the speed of 12000 rpms for 3 minutes. Filter samples and buffer using a 0.1 pm filter. The samples were concentrated to 5-6 mg/mL and observed for the appearance of the samples.
Colorless (CL), slight-opalescent (SO), particle-free (PF), particle-observed (PO)
5.3 Thermal Stability Test by DSF
Tm (melting temperature) of each antibody was investigated using QuantStudio® 7 Flex Real-Time PCR system. 19 μL antibody solution was mixed with 1 μL 80 x SYPRO Orange solution (Invitrogen) in the 96 wells plate. Each sample was tested in duplicate wells. The plate was sealed with the Optical Adhesive Film and centrifuged at 3,000 rpms for 5 minutes to remove any air bubbles. The plate was heated from 26 °C to 95 °C at a rate of 0.9 °C/minute, and the resulting fluorescence data was collected. The negative derivatives of the fluorescence changes with respect to different temperatures were calculated, and the maximal value was defined as melting temperature Tm. If a protein has multiple unfolding transitions, the first two Tms were reported, named as Tml and Tm2. Data collection and Tm calculation were conducted automatically by the QuantStudio® Real Time PCR software software (vl.3).
Tml value was in the normal range as showed in Table 22. The DSF profile was showed in Figure 11.
5,4 Radius (nm) measurement by DLS
Radius measurement was investigated using DynaPro Plate Reader III (Wyatt Technology). Samples were first filtered with 0.2 pm filter and diluted to 1 mg/mL. Then samples were centrifuged at 12000 rpms for 3 minutes and 7.5 μL sample solution was then added to 1536 wells
microplate. The plate was sealed with the Clear Seal Film, and centrifuged at 3,000 rpms for 5 minutes to let the sample down to the bottom of the well. Each sample was tested in duplicate wells. Put the plate into the corresponding position and data collection was performed by the DYNAMICS operation software (v7.8.1.3). 5 acquisitions were collected for each protein sample while each acquisition time was 5 s. Radius were calculated automatically by the operation software.
The data in Table 23 and Figure 12 indicated the polydispersity was in the normal range.
5.5 Hydrophobicity interaction chromatography HPLC (HIC-HPLC)
Hydrophobicity property of antibody was detected by HPLC 1260 Infinity II system (Agilent Technologies™) with TSKgel butyl-NPR column. Each sample was diluted to 0.5 mg/mL with PBS buffer and 20 μL diluted sample was injected into the column, and seperated with a flow rate of 0.5 mL/minute for 61 minutes. The running buffer was 25 mM sodium phosphate, pH 7.0 (Buffer A) and 25 mM sodium phosphate, 1.5 M (NH4)2SO4, pH 7.0 (Buffer D). The running gradient was 100 % to 0 % Buffer D and 0 % to 100 % Buffer A from 3 to 53 minutes. The peak retention was detected with UV light of the wavelength at 280 nm and 230 nm. The retention time was analyzed with HIC-HPLC analysis method to integrate all peak areas from 20 minutes to 40 minutes. The operation and analysis software were the OpenLab CDS Workstation (v2.6.0.691).
Retention time represents the hydrophobicity degree of the antibody, and long retention time indicates high potential hydrophobicity. The data in Table 24 and Figure 13 indicated that the HIC- HPLC profile of W306106-P11R2-1B8-Z6 falls in the normal range.
5,6 Determination of diffusion interaction parameter (kD) by PLS kD measurement was investigated using PynaPro Plate Reader III (Wyatt Technology). During the sample preparation process, the appearance of samples was observed at thawing, filtration and concentration by visual inspection. The purity of a dose point (15 mg/mL) would be tested after concentration. Samples were first filtered with 0.1 pm filter. Samples were then concentrated to over 20 mg/mL and diluted with corresponding buffer to a final concentration at 2.5, 5, 10, 15, and 20 mg/mL. 7.5 μL sample solution was then added to 1536 wells microplate. The plate was sealed with the ClearSeal Film, and centrifuged at 3,000 rpms for 5 minutes to let the sample down to the bottom of the well. Each sample was tested in duplicate wells. Put the plate into the corresponding position and data collection was performed by the DYNAMICS operation software (v7.8.1.3). 5 acquisitions were collected for each protein sample while each acquisition time was 5 seconds. For each measurement, the diffusion coefficient was determined and plotted against protein concentration. kD values were calculated automatically by the software.
Lead with high kD and monodisperse size most likely has low aggregation propensity. The data in Table 25 and Figure 14 indicated that W306106-P11R2-1B8-Z6 showed good property.
5,7 Stress Test
The concentration of each sample was measured by Nanodrop2000 with 2 μL sample for 3 times and then diluted to 1 mg/mL by the sample storage buffer (PBS). Eight tubes of 300 μL antibody solution was prepared for each sample. Placed each tube in its respective temperature conditions (either 4 °C or 40 °C or -80 °C) and stored for 14 days, or freezed (-80 °C)/thawed (25 °C) for 3 and 5 cycles. After the stress treatment, samples were centrifuged at 12,000 rpms for 3 minutes and observed visually. Measured the protein concentration by Nanodrop2000 and recorded the data. Purity of each antibody was detected by Agilent 1260 Infinity II system with
TSKgel G3000SWXL column. 25 μL sample was injected into the column, and seperated with a flow rate of 1 mL/minute for 20 minutes. The running buffer was 50 mM sodium phosphate, 150 mM NaCl, pH7.0. The peak retention was detected with UV light of the wavelength at 280 run. The purity of each antibody was analyzed with SEC-HPLC analysis method to integrate all peak areas from 4.5 minutes to 10.5 minutes. The operation and analysis software were the OpenLab CDS Workstation (v2.3.0.443).
The data in Tables 26-27 and Figures 15-16 indicated that the stress test profiles of W306106-P11R2-1B8-Z6 fall in the normal range.
Table 26. Freeze/thaw cycling test result summary
Note: Colorless (CL), slight-opalescent (SO), particle- free (PF), particle-observed (PO)
Note: Colorless (CL), slight-opalescent (SO), particle-free (PF), particle-observed (PO)
Example 6: In vitro Characterization of W306106-PllR2-lB8-uIgGl (pre-humanization), W306106-PllR2-lB8-z6-uIgGl (after humanization) and W306106-PllR2-lB8-z6 (VHH after humanization)
6.1 FACS binding of antibodies to cell surface human MUC16 proteins
6.1.1 FACS binding of antibody to cell surface human MUC 16 proteins (pre-humanization)
Binding of anti-MUC16 antibodies to MUC 16 expressing cells was determined by flow
cytometry. W3XX106-SK-OV-3.hProl.FL.A9 cell expressing the human MUC 1655th & 56th SEA domain and a selected tumor cell line OVCAR-3 (1 x 105 cells/well) expressing high level of human MUC 16 were harvested by using or 0.25 % Trypsin-EDTA (1 x). Then the cells were incubated with serial diluted antibodies (for W3XX106-SK-OV-3.hProl.FL.A9, starting at 400 nM, 4-fold dilution to 0.00038 nM, for OVCAR-3, starting at 500 nM, 5-fold dilution to 0.0064 nM) in a volume of 100 μL for 1 hour at 4 °C. The parental of DMUC5754A (Genentech) and MUC16 binding arm of REGN-4018 (Regeneron) were used as positive controls, and human IgGl isotype control antibody W332-1.80.12.xAb.hIgGl was used as the negative control. After washing the cells twice with 1 % BSA, Alexa Fluor® 647 AffiniPure Goat Anti-Human IgG, Fey fragment specific (1: 500 dilution in 1 % BSA) was added and incubated for 30 minutes at 4 °C in the dark. After washing the cells twice with 1 % BSA, the cells were re-suspended in 1 % BSA. The mean fluorescence intensity (MFI) of the cells was measured by a flow cytometer and analyzed by FlowJo. The EC50 values were calculated by four-parameter non-linear regression analysis using GraphPad Prism software.
The binding results of W306106-P1 !R2-lB8-uIgG1 on W3XX106-SK-OV-3.hProl.FL.A9 and W3XX106-SK-OV-3.cProl.FL.El cells were shown in Figure 17A-17B, respectively. The EC50 and Max MFI were shown in Table 28. W306106-P 11R2- lB8-uIgG1 showed strong binding to the cell surface human and cynomolgus monkey MUC 16 55th & 56th SEA domain with EC50 of 2.71 nM and 1.77 nM respectively. The binding activity were comparable to MUC 16 binding arm of REGN4018 (Regeneron). The parental of DMUC5754A (Genentech) showed no binding to the cell surface human or cynomolgus monkey MUC 16 55th & 56th SEA domain.
Table 28. Human and cynomolgus monkey MUC 16 55th & 56th SEA domain FACS binding
The binding results of W306106-P1 !R2-lB8-uIgGl on OVCAR-3 (MUC16hlgh) was shown in Figure 20. The EC50 and Max MFI were shown in Table 29. W306106-P1 !R2-lB8-uIgGl showed strongly binding to the cell surface human MUC16 with an EC50 of 3.35 nM. The binding activity was comparable to the parental of DMUC5754A (Genentech) and MUC16 binding arm of
6,1,2 FACS binding of antibody to cell surface human MUC 16 proteins (after humanization)
Binding of anti-MUC16 antibodies to MUC16 expressing cells was determined by flow cytometry. W3XX106-SK-OV-3.hProl.FL.A9 cell expressing the human MUC1655th & 56th SEA domain and four selected tumor cell lines including OVCAR-3, HDQ-P1, SK-OV-3 and A375 (1 x 105 cells/well) expressing different level of human MUC16 were harvested by using or 0.25 %
Trypsin-EDTA (1 x). Then the cells were incubated with serial diluted antibodies (for W3XX106-
SK-OV-3. hProl. FL. A9, SK-OV-3 and A375, starting at 400 nM, 4-fold dilution to 0.0015 nM, for
OVCAR-3 and HDQ-P1, starting at 500 nM, 5-fold dilution to 0.00026 nM) in a volume of 100 μL for 1 hour at 4 °C. The parental of DMUC5754A (Genentech) was used as the positive control, and human IgGl isotype control antibody W332-1.80.12.xAb.hIgGl was used as the negative control. After washing the cells twice with 1 % BSA, Alexa Fluor® 647 AffiniPure Goat Anti¬
Human IgG, Fcγ fragment specific (1: 500 dilution in 1 % BSA) was added and incubated for 30 minutes at 4 °C in the dark. After washing the cells twice with 1 % BSA, the cells were resuspended in 1 % BSA. The mean fluorescence intensity (MFI) of the cells was measured by a flow cytometer and analyzed by Flow Jo. The EC50 values were calculated by four-parameter non- linear regression analysis using GraphPad Prism software.
The binding results of W306106-P1 !R2-lB8-z6-uIgGl on W3XX106-SK-OV-
3.hProl.FL.A9 and W3XX106-SK-OV-3.cProl.FL.El cells were shown in Figure 18A-18B, respectively. The EC50 and Max MFI were shown in Table 30. W306106-P1 !R2-lB8-z6-uIgGl
showed strong binding to the cell surface human and cynomolgus monkey MUC16 55th & 56th
SEA domain, while the parental of DMUC5754A (Genentech) showed no binding to the cell surface human or cynomolgus monkey MUC16 55th & 56th SEA domain.
Table 30. Human and cynomolgus monkey MUC16 55th & 56th SEA domain FACS binding
The binding results of W306106-P1 !R2-lB8-z6-uIgG1 on four selected human tumor cell lines expressing different level of human MUC16 were shown in Figure 21 including OVCAR-3 (MUC16high), HDQ-P1 (MUC16medium), SK-OV-3 (MUC16low) and A375 (MUC16negative). The
EC50 and Max MFI were shown in Table 31. W306106-Pl lR2-lB8-z6-uIgG1 showed specifically binding to the cell surface human MUC16 of OVCAR-3 (MUC16high ), HDQ-P1 (MUC16medium) and SK-OV-3 (MUC16low). The binding activity of W306106-P11R2-1B8-Z6- ulgGl to OVCAR-3 (MUC16hlgh) was slightly weaker than the parental of DMUC5754A (Genentech), and the binding activity to HDQ-P1 (MUC16medlum) and SK-OV-3 (MUC16low) were comparable to the parental of DMUC5754A (Genentech). Both W306106-Pl lR2-lB8-z6-uIgG1 and the parental of DMUC5754A (Genentech) showed no binding to A375 (MUC16negative).
Table 31. Human MUC16 FACS binding on four human tumor cell lines
6,1.3 FACS binding of antibody to cell surface human MUC16 proteins (humanized VHH)
Binding of anti-MUC16 antibodies to MUC16 expressing cells was determined by flow cytometry. W3XX106-SK-OV-3.hProl.FL.A9 cell expressing the human MUC1655th & 56th SEA domain and two selected tumor cell lines, OVCAR-3 and A375 (1 x 105 cells/well) expressing different level of human MUC16 were harvested by using or 0.25 % Trypsin-EDTA (1 x). Then the cells were incubated with serial diluted antibodies (for W3XX106-SK-OV-3.hProl.FL.A9 and A375, starting at 600 nM, 4-fold dilution to 0.0023 nM, for OVCAR-3, starting at 500 nM, 5-fold dilution to 0.0064 nM) in a volume of 100 μL for 1 hour at 4 °C. The antibody W306108-P24R2- IGl.His was used as the negative control. After washing the cells twice with 1 % BSA, MonoRab™ Rabbit Anti Camelid VHH Cocktail [iFluor 647] (1:500 dilution in 1 % BSA) was added and incubated for 30 minutes at 4 °C in the dark. After washing the cells twice with 1 % BSA, the cells were re-suspended in 1 % BSA. The mean fluorescence intensity (MFI) of the cells was measured by a flow cytometer and analyzed by FlowJo. The EC50 values were calculated by four-parameter non-linear regression analysis using GraphPad Prism software.
The binding results of W306106-P11R2-1B8-Z6 on W3XX106-SK-OV-3.hProl.FL.A9 and W3XX106-SK-OV-3.cProl.FL.El cells were shown in Figure 19A-19B, respectively. The EC50 and Max MFI were shown in Table 32. W306106-Pl lR2-lB8-z6 showed strong binding to the cell surface human and cynomolgus monkey MUC16 55th & 56th SEA domain and the EC50 of binding to W3XX106-SK-OV-3.hProl.FL.A9 was 8.67 nM.
The binding results of W306106-P11R2-1B8-Z6 on two selected human tumor cell lines expressing different level of human MUC16 were shown in Figure 22 including OVCAR-3 (MUC16high) and A375 (MUC16negative). The EC50 and Max MFI were shown in Table 33.
W306106-P11R2-1B8-Z6 showed specifically binding to the cell surface human MUC16 of OVCAR-3 (MUC16higb) with an EC50 of 3.65 nM and no binding to A375 (MUC16negative).
6.2
6,2.1 FACS binding of antibody to cell surface cynomolgus monkey MUC16 proteins (pre- humanization)
W3XX106-SK-OV-3.cProl.FL.El cells expressing the cynomolgus monkey MUC16 55th & 56th SEA domain were harvested by using 0.25 % Trypsin-EDTA (1 x). Then the cells were incubated with serial diluted antibodies (starting at 400 nM, 4- fold dilution to 0.000095 nM) in a volume of 100 pL for 1 hour at 4 °C. The parental of DMUC5754A (Genentech) and MUC16 binding arm of REGN-4018 (Regeneron) were used as positive controls, and human IgGl isotype control antibody W332-1.80.12.xAb.hIgGl was used as the negative control. After washing the cells with 1 % BSA, Alexa Fluor® 647 AffiniPure Goat Anti-Human IgG, Fey fragment specific
(1:500 dilution in 1 % BSA) was added and incubated for 30 minutes at 4 °C in the dark. After washing the cells twice with 1 % BSA, the cells were re-suspended in 1 % BSA. The mean fluorescence intensity (MFI) of the cells was measured by a flow cytometer and analyzed by
FlowJo. The EC50 values were calculated by four-parameter non-linear regression analysis using
GraphPad Prism software. The results were shown in Table 28.
6,2,2 FACS binding of antibody to cell surface cynomolgus monkey MUC16 proteins (after humanization)
W3XX106-SK-OV-3.cProl.FL.El cells expressing the cynomolgus monkey MUC16 55th & 56th SEA domain were harvested by using 0.25 % Trypsin-EDTA (1 x). Then the cells were incubated with serial diluted antibodies (starting at 500 nM, 5-fold dilution to 0.000010 nM) in a volume of 100 pL for 1 hour at 4 °C. The parental of DMUC5754A (Genentech) was used as the positive control, and human IgGl isotype control antibody W332-1.80.12.xAb.hIgGl was used as the negative control. After washing the cells with 1 % BSA, Alexa Fluor® 647 AffiniPure Goat
Anti-Human IgG, Fey fragment specific (1 :500 dilution in 1 % BSA) was added and incubated for 30 minutes at 4 °C in the dark. After washing the cells twice with 1 % BSA, the cells were resuspended in 1 % BSA. The mean fluorescence intensity (MFI) of the cells was measured by a flow cytometer and analyzed by FlowJo. The EC50 values were calculated by four-parameter nonlinear regression analysis using GraphPad Prism software. The results were shown in Table 30.
6.2.3 FACS binding of antibody to cell surface cynomolgus monkey MUC16 proteins (VHH after humanization)
W3XX106-SK-OV-3.cProl.FL.El cells expressing the cynomolgus monkey MUC16 55th & 56th SEA domain were harvested by using 0.25 % Trypsin-EDTA (1 x). Then the cells were incubated with serial diluted antibodies (starting at 600 nM, 4-fold dilution to 0.0023 nM) in a volume of 100 μL for 1 hour at 4 °C. The antibody W306108-P24R2-lGl.His was used as the negative control. After washing the cells twice with 1 % BSA, MonoRab™Rabbit Anti Camelid VHH Cocktail [iFluor 647] (1 : 500 dilution in 1 % BSA) was added and incubated for 30 minutes at 4 °C in the dark. After washing the cells twice with 1 % BSA, the cells were re-suspended in 1 % BSA. The mean fluorescence intensity (MFI) of the cells was measured by a flow cytometer and analyzed by FlowJo. The EC50 values were calculated by four-parameter non-linear regression analysis using GraphPad Prism software. The results were shown in Table 32.
6.3 Human MUC16 extracellular domain determination binding (ELISA)
6,3.1 Human MUC16 extracellular domain determination binding (pre-humanization)
ELISA plates were coated with human MUC 1656th SEA domain or CA125 protein (2 μg/mL, 100 μL/well) in coating buffer and incubated at 4 °C overnight. The next day, the coating antigen buffer was removed and the ELISA plates were washed 1 time with washing buffer 1 x PBST (300 μL/wash). The ELISA plates were blocked with blocking buffer (100 % casein), 200 μL/well, and incubated at room temperature for 1 hour. Then, the ELISA plates were washed 3 times with washing buffer, 300 μL /well. The serial diluted antibodies in 50 % casein (100 μL/well, starting at 500 nM, 4-fold dilution to 0.00012 nM) were added and incubated at room temperature for 2 hours. The MUC16 binding arm of REGN4018 and the parental of DMUC5754A, were used as the positive controls, and the W332- 1.80.12.xAb.hIgGl isotype control antibody was used as negative control. Subsequently, the ELISA plates were washed 3 times with washing buffer, 300 μL/wash. The Goat anti-Human IgG-Fc Fragment Cross-Adsorbed Antibody (1 : 5000, HRP conjugated, Ab2)
in 50 % casein was added, 100 μL /well and incubated at room temperature for 1 hour. After washing the ELISA plates for 6 times with washing buffer, 300 μL/well, TMB substrate was added with 100 μL/well, and the ELISA plates were incubated at room temperature for 10 minutes in the dark. The stop solution (2 M HC1) was added with 100 μL/well to stop further color developing. Finally, the ELISA plates were detected by microplate reader M5e at 450 nm and 540 nm.
W306106-Pl lR2-lB8-uIgG1 showed good binding to recombinant human 56th SEA domain MUC16 but showed no binding to CAI 25 (soluble MUC16) (Figure 23). However, Parental of DMUC5754A bound to CA125 but not human 56th SEA domain MUC16. This suggested that W306106-P11R2-lB8-uIgG1 could bind to human 56th SEA domain MUC16 and was not affected by CA125 (soluble MUC16). The EC50 and Max OD are shown in Table 34.
Table 34. The ELISA binding E aCn5d0 Max OD of anti-MUC16 Abs to human MUC16 extracellular domain proteins
6,3,2 Human MUC16 extracellular domain determination binding (after humanization)
ELISA plates were coated with human MUC16 55th & 56th protein, human MUC16 56th protein or CAI 25 protein (2 μg/mL, 100 μL/well) in coating buffer and incubated at 4 °C overnight. The next day, the coating antigen buffer was removed and the ELISA plates were washed 1 time with washing buffer 1 x PBST (300 μL/wash). The ELISA plates were blocked with blocking buffer (100 % casein), 200 μL /well, and incubated at room temperature for 1 hour. Then, the ELISA plates were washed 3 times with washing buffer, 300 μL/well. The serial diluted antibodies in 50 % casein (100 μL/well, starting at 400 nM, 4-fold dilution to 0.0015 nM or starting at 400 nM, 4-fold dilution to 0.00010 nM) were added and incubated at room temperature for 2 hours. The parental of DMUC5754A was used as the positive controls, and the W332-1.80.12.xAb.hIgGl isotype control antibody was used as negative control. Subsequently, the ELISA plates were
washed 3 times with washing buffer, 300 μL/wash. The Goat anti-Human IgG-Fc Fragment Cross- Adsorbed Antibody (1:5000, HRP conjugated, Ab2) in 50 % casein was added, 100 μL /well and incubated at room temperature for 1 hour. After washing the ELISA plates for 6 times with washing buffer, 300 μL/well, TMB substrate was added with 100 μL/well, and the ELISA plates were incubated at room temperature for 10 minutes in the dark. The stop solution (2 M HC1) was added with 100 μL/well to stop further color developing. Finally, the ELISA plates were detected by microplate reader M5e at 450 nm and 540 nm.
W306106-P1 lR2-lB8-z6-uIgG1 showed good binding to recombinant human 56th SEA domain MUC16 and human 55th & 56th SEA domain MUC16, but showed no binding to CA125 (soluble MUC16) (Figure 24). However, Parental of DMUC5754A bound to CA125 but not human 56th SEA domain MUC16 and human 55th & 56th SEA domain MUC16. This suggested that W306106-P1 lR2-lB8-z6-uIgG1 could bind to human 56th SEA domain MUC16 and was not affected by CA125 (soluble MUC16). The EC50 and Max OD are shown in Table 35.
Table 35. The ELISA binding E aCnd50 Max OD of anti-MUC16 Abs to human MUC16 extracellular domain proteins
6.3,3 Human MUC16 extracellular domain determination binding (humanized VHH)
ELISA plates were coated with human MUC16 55th & 56th SEA domain, human MUC16 56th SEA domain or CA125 protein (2 μg/mL, 100 μL/well) in coating buffer and incubated at 4 °C overnight. The next day, the coating antigen buffer was removed and the ELISA plates were washed 1 time with washing buffer 1 x PBST (300 μL/wash). The ELISA plates were blocked with blocking buffer (100 % casein), 200 μL/well, and incubated at room temperature for 1 hour. Then, the ELISA plates were washed 3 times with washing buffer, 300 μL/well. The serial diluted antibodies in 50 % casein (100 μL/well, starting at 400 nM, 5-fold dilution to 0.00020 nM) were
added and incubated at room temperature for 2 hours. The W306108-P24R2-lGl.His isotype control antibody was used as negative control. Subsequently, the ELISA plates were washed 3 times with washing buffer, 300 μL/wash. The MonoRab™ Rabbit Anti-Camelid VHH Cocktail Antibody (1:5000, HRP conjugated, Ab2) in 50 % casein was added, 100 μL/well and incubated at room temperature for 1 hour. After washing the ELISA plates for 6 times with washing buffer, 300 μL /well, TMB substrate was added with 100 μL/well, and the ELISA plates were incubated at room temperature for 10 minutes in the dark. The stop solution (2 M HC1) was added with 100 μL/well to stop further color developing. Finally, the ELISA plates were detected by microplate reader M5e at 450 nm and 540 nm.
W306106-P11R2-1B8-Z6 showed good binding to recombinant human 56th SEA domain MUC16 and human 55th & 56th SEA domain MUC16, but showed no binding to CA125 (soluble MUC16) (Figure 25). This suggested that W306106-P11R2-1B8-Z6 could bind to human 56th SEA domain MUC16 and was not affected by CA125 (soluble MUC16). The EC50 and Max OD are shown in Table 36.
Table 36. The ELISA binding EC50 and Max OD of anti-MUC16 Abs to human MUC16 extracellular domain proteins
6,4 ELISA binding of antibodies to cynomolgus monkey and mouse MUC16 extracellular domain (ELISA)
6.4.1 ELISA binding of antibodies to cynomolgus monkey and mouse MUC16 extracellular domain (after humanization)
ELISA plates were coated with cynomolgus monkey and mouse MUC16 55th & 56th SEA domain (2 μg/mL, 100 μL/well) in coating buffer and incubated at 4 °C overnight. The next day, the coating antigen buffer was removed and the ELISA plates were washed 1 time with washing buffer 1 x PBST (300 μL/wash). The ELISA plates were blocked with blocking buffer (100 % casein), 200 μL/well, and incubated at room temperature for 1 hour. Then, the ELISA plates were
washed 3 times with washing buffer, 300 pL/well. The serial diluted antibodies in 50 % casein
(100 pL/well, starting at 400 nM, 4-fold dilution to 0.0015 nM or starting at 400 nM, 4-fold dilution to 0.00010 nM) were added and incubated at room temperature for 2 hours. The parental of DMUC5754A was used as the positive controls, and the W332-1.80.12.xAb.hIgGl isotype control antibody was used as negative control. Subsequently, the ELISA plates were washed 3 times with washing buffer, 300 pL/wash. The Goat anti-Human IgG-Fc Fragment Cross- Adsorbed
Antibody (1:5000, HRP conjugated, Ab2) in 50 % casein was added, 100 pL/well and incubated at room temperature for 1 hour. After washing the ELISA plates for 6 times with washing buffer,
300 pL/well, TMB substrate was added with 100 pL/well, and the ELISA plates were incubated at room temperature for 10 minutes in the dark. The stop solution (2 M HC1) was added with 100 pL/well to stop further color developing. Finally, the ELISA plates were detected by microplate reader M5e at 450 nm and 540 nm.
W3 06106-P 11 R2- 1 B 8-z6-uIgG 1 showed good binding to recombinant cynomolgus monkey
55th & 56th SEA domain MUC 16, but showed no binding to mouse 55th & 56th SEA domain
MUC 16 (Figure 26). The EC50 and Max OD are shown in Table 37.
6,4,2 ELISA binding of antibodies to cynomolgus monkey and mouse MUC 16 extracellular domain (humanized VHH )
ELISA plates were coated with cynomolgus monkey and mouse MUC 16 55th & 56th SEA domain (2 pg/mL, 100 pL/well) in coating buffer and incubated at 4 °C overnight. The next day, the coating antigen buffer was removed and the ELISA plates were washed 1 time with washing buffer 1 x PBST (300 pL/wash). The ELISA plates were blocked with blocking buffer (100 % casein), 200 pL/well, and incubated at room temperature for 1 hour. Then, the ELISA plates were
washed 3 times with washing buffer, 300 μL/well. The serial diluted antibodies in 50 % casein (100 μL/well, starting at 400 nM, 5-fold dilution to 0.00020 nM) were added and incubated at room temperature for 2 hours. The W306108-P24R2-lGl.His isotype control antibody was used as negative control. Subsequently, the ELISA plates were washed 3 times with washing buffer, 300 μL/wash. The MonoRab™ Rabbit Anti-Camelid VHH Cocktail Antibody (1:5000, HRP conjugated, Ab2) in 50 % casein was added, 100 μL /well and incubated at room temperature for 1 hour. After washing the ELISA plates for 6 times with washing buffer, 300 μL/well, TMB substrate was added with 100 μL/well, and the ELISA plates were incubated at room temperature for 10 minutes in the dark. The stop solution (2 M HC1) was added with 100 μL/well to stop further color developing. Finally, the ELISA plates were detected by microplate reader M5e at 450 nm and 540 nm.
W306106-P1 lR2-lB8-z6showed good binding to recombinant cynomolgus monkey 55th & 56th SEA domain MUC16, but showed no binding to mouse 55th & 56th SEA domain MUC16
(Figure 27). The EC50 and Max OD are shown in Table 38.
Table 38. The ELISA binding E aCnd50 Max OD of anti-MUC16 Abs to cynomolgus monkey and mouse MUC16 extracellular domain proteins
6.5 Affinity to human 55th+56th SEA domain (SPR)
The affinity data of anti-MUC16 Abs to human 55th + 56th SEA domain of MUC16 were tested for SPR analysis.
(1) For pre-humanization Ab:
The activator was prepared by mixing 400 mM EDC (l-Ethyl-3 -(3 -Dimethylaminopropyl) Carbodiimide) and 100 mM NHS (N-Hydroxy Succinimide) immediately prior to injection. The CM5 sensor chip was activated for 420 s at a flow rate of 10 μL/minute with the mixture. 30 μg/mL of anti-human IgG Fc antibody in 10 mM NaAc (pH 4.5) was then injected to channel 1-8 for 420
s at a flow rate of 10 μL/minute. The chip was deactivated by 1 M ethanolamine-HCl for 420 s at a flow rate of 10 μL/minute.
600-fold diluted NC and the same fold diluted W306106-Pl lR2-lB8-uIgG1 in running buffer lx HBS-EP+ (0.01 M HEPES, 0.15 M NaCl, 3 mM EDTA, 0.05 % surfactant P20, pH 7.4) were respectively injected to Fcl and Fc2 at a flow rate of 10 μL/min for 30 s. 500-fold diluted NC and the same fold diluted W306106-P1 lR3-lB8-uIgG1 in running buffer lx HBS-EP+ were respectively injected to Fcl andFc2 at a flow rate of 10 μL/minute for 30 s. 3 concentrations (12.5, 50 and 200 nM) of antigen W3XX106-hProl.ECD.AVI.His-P2 and running buffer were injected orderly to Fcl-Fc2 at a flow rate of 30 μL/minute for an association phase of 180 s, followed by 3600 s dissociation. 10 mM glycine (pH 1.5) as regeneration buffer was injected to flow cells following every dissociation phase.
The affinity of W306106-P1 !R2-lB8-uIgG1 and W306106-P1 !R2-lB8-z6-uIgGl (prehumanization and humanization Ab) is shown in Figures 28-29 and Table 39 and Table 40.
Note: kd labeled with * is approaching or outside the limits that can be measured by the instrument. The kd and KD are only for reference.
(2) For humanization Ab:
The activator was prepared by mixing 400 mM EDC (l-Ethyl-3 -(3 -Dimethylaminopropyl) Carbodiimide) and 100 mM NHS (N-Hydroxy Succinimide) immediately prior to injection. The CM5 sensor chip was activated for 420 s at a flow rate of 10 μL /minute with the mixture. 30 μg/mL of anti-human IgG Fc antibody in 10 mM NaAc (pH 4.5) was then injected to channel 1-8 for 420 s at a flow rate of 10 μL/minute. The chip was deactivated by 1 M ethanolamine-HCl for 420 s at a flow rate of 10 μL /minute.
0.25 μg/mL of W306106-P11R2-lB8-z6-uIgG1 in running buffer lx HBS-EP+ was injected to Fc2 at a flow rate of 10 μL /minute for 30 s. 8 concentrations (0.391, 0.781, 1.563, 3.125, 6.25, 12.5, 25 and 50 nM) of antigen W3XX106-hProl.ECD.AVI.His-P2 and running buffer were
injected orderly to Fcl-Fc2 at a flow rate of 30 μL/minute for an association phase of 180 s, followed by 3600 s dissociation. 10 mM glycine (pH 1.5) as regeneration buffer was injected to flow cells following every dissociation phase.
Note: kd labeled with * is approaching or outside the limits that can be measured by the instrument. The kd and KD are only for reference.
6,6 HCS internalization assay by Operetta
The 96-well plate was coated with 2.5 μg/cm2 Poly-D-Lysine (PDL) at 37 °C for 2 hours (1 : 1000), 100 μL/well. Cells in the T75 flask were washed by PBS and detached by Versene. The cells were washed once with culture medium and resuspended in an appropriate volume of culture medium to the concentration of 3 x 105 cells/mL with pipettes. Then 100 μL cells suspension was aliquoted to each well of 96-well plate with multi-channel pipettes and incubated overnight. The second day, the medium was fetched and the cells were washed once with 1 % BSA. The serial diluted Abs in 1 % BSA (100 μL/well) were added to the cell plates and incubated at 4 °C for 2 hours. After incubation, the cells were washed once by 1 % BSA, 200 μL /well. Goat anti-human IgG PE (1: 200 diluted in 1 % BSA, 100 μL/well) was added and incubated for 1 hour at 4 °C in the dark. After washing the cells with lx PBS / 1 % BSA, 1 % BSA was added to each well (100 μL/well) and the plate was incubated for 2 hours at 37 °C. And 1 % BSA was discarded and the cells were quenched at 4 °C for 4 minutes. Then the plate was washed once with PBS, 150 μL/well. Hoechst (1: 2000 dilution in PBS, 100 μL/well) was added and incubated for 20 minutes at room temperature. The plate was washed once with 1 x PBS and the cells were fixed with 4 % PFA for 15 minutes at room temperature and the plate was saved at 4 °C for further analysis. The plate was read by Oppretta.
W306106-P1 !R2-lB8-uIgG1 mediated good target internalization in OVCAR-3 cells tested by HCS internalization assay, which was comparable to MUC16 binding arm of REGN4018 (Regeneron) (Figure 30). The EC50 and Max MFI were shown in Table 41.
Table 41. The EC a50nd Max MFI of HCS internalization of anti-MUC16 mAbs
Those skilled in the art will further appreciate that the present disclosure may be embodied in other specific forms without departing from the spirit or central attributes thereof. In that the foregoing description of the present disclosure discloses only exemplary embodiments thereof, it is to be understood that other variations are contemplated as being within the scope of the present disclosure. Accordingly, the present invention is not limited to the particular embodiments that have been described in detail herein. Rather, reference should be made to the appended claims as indicative of the scope and content of the invention.
References
[1] Bast R C, Feeney M, Lazarus H, et al. Reactivity of a monoclonal antibody with human ovarian carcinomafJ]. The Journal of clinical investigation, 1981, 68(5): 1331-1337.
[2] Yin B W T, Dnistrian A, Lloyd K O. Ovarian cancer antigen CA125 is encoded by the MUC16 mucin gene[J]. International journal of cancer, 2002, 98(5): 737-740.
[3] Aithal A, Rauth S, Kshirsagar P, et al. MUC16 as a novel target for cancer therapy [J]. Expert opinion on therapeutic targets, 2018, 22(8): 675-686.
[4] Das S, Majhi P D, Al-Mugotir M H, et al. Membrane proximal ectodomain cleavage of MUC16 occurs in the acidifying Golgi/post-Golgi compartments [J]. Scientific reports, 2015, 5(1): 9759.
[5] Haridas D, Ponnusamy M P, Chugh S, et al. MUC16: molecular analysis and its functional implications in benign and malignant conditions [J]. The FASEB Journal, 2014, 28(10): 4183- 4199.
[6] Liede A, Karlan B Y, Baldwin R L, et al. Cancer incidence in a population of Jewish women at risk of ovarian cancerfj], Journal of clinical oncology, 2002, 20(6): 1570-1577.
[7] Kim Y W, Bae S M, Kim I W, et al. Multiplexed bead-based immunoassay of four serum biomarkers for diagnosis of ovarian cancerfj]. Oncology reports, 2012, 28(2): 585-591.
[8] Morales-Vasquez F, Pedemera E, Reynaga-Obregon J, et al. High levels of pretreatment CAI 25 are associated to improved survival in high grade serous ovarian carcinomafJ]. Journal of Ovarian Research, 2016, 9(1): 1-6.
[9] Streppel M M, Vincent A, Mukherjee R, et al. Mucin 16 (cancer antigen 125) expression in human tissues and cell lines and correlation with clinical outcome in adenocarcinomas of the
pancreas, esophagus, stomach, and colon[j]. Human pathology, 2012, 43(10): 1755-1763.
[10] Berek J S, Taylor P T, Gordon A, et al. Randomized, placebo-controlled study of oregovomab for consolidation of clinical remission in patients with advanced ovarian cancer [J], Journal of Clinical Oncology, 2004, 22(17): 3507-3516. [11] Sabbatini P, Harter P, Scambia G, et al. Abagovomab as maintenance therapy in patients with epithelial ovarian cancer: a phase III trial of the AGO OVAR, COGI, GINECO, and GEICO — the MIMOSA study [J]. Journal of clinical oncology, 2013, 31(12): 1554.
Claims
1. A MUC16-binding molecule comprising an immunoglobulin single variable domain, wherein the single variable domain comprises a CDR1, CDR2, and CDR3 of a VHH as set forth in SEQ ID NO: 4 or 5.
2. The MUC16-binding molecule of claim 1, wherein the CDR1 comprises an amino acid sequence as set forth in SEQ ID NO: 1, the CDR2 comprises an amino acid sequence as set forth in SEQ ID NO: 2, and the CDR3 comprises an amino acid sequence as set forth in SEQ ID NO: 3.
3. The MUC16-binding molecule of any of claims 1 to 2, wherein the single variable domain comprises:
(A) the amino acid sequence as set forth in any one of SEQ ID NOs: 4-5;
(B) an amino acid sequence at least 85%, 90%, or 95% identical to the amino acid sequence as set forth in any one of SEQ ID NOs: 4-5 yet retaining the specific binding affinity to MUC16.
4. The MUC16-binding molecule of any of claims 1 to 3, comprising one or more substitutions, additions and/or deletions of amino acids in the framework regions, e.g. FRW1, FRW2, FRW3, and/or FRW4 of the single variable domain.
5. The MUC16-binding molecule of any of claims 1 to 6, wherein the single variable domain comprises the amino acid sequence as set forth in SEQ ID NO: 4 or 5.
6. The MUC16-binding molecule of any of claims 1 to 5, wherein the MUC16-binding molecule further comprises a human IgG constant domain.
7. The MUC16-binding molecule of claim 6, wherein the human IgG constant domain is a human IgGl, IgG2, IgG3 or IgG4 constant domain, such as a human IgGl constant domain or a variant thereof.
8. The MUC16-binding molecule of any of claims 1 to 7, which has one or more of the following properties:
(a) specifically bind to human MUC16 and cyno MUC16 but not to CA125 soluble protein;
(b) have a low self-interaction propensity;
(c) have a good thermal stability; and
(d) have no non-specific binding.
9. The MUC16-binding molecule of any of claims 1 to 8, wherein the MUC16-binding molecule is a chimeric antibody or a humanized antibody.
10. The MUC16-binding molecule of any of claims 1 to 9, which comprises an amino acid sequence as set forth in any one of SEQ ID NOs: 6-7.
11. The MUC16-binding molecule of any of claims 1 to 10, which is a dimer.
12. A fusion protein comprising the MUC16-binding molecule as defined in any of claims 1 to 11 fused to a heterologous peptide, such as an antigen-binding domain targeting a different antigen.
13. A nucleic acid molecule comprising a nucleic acid sequence encoding the single variable domain of the MUC16-binding molecule as defined in any of claims 1 to 12.
14. A vector comprising the nucleic acid molecule of claim 13.
15. A host cell comprising the vector of claim 16 or the nucleic acid molecule of claim 13.
16. A pharmaceutical composition comprising at least one MUC16-binding molecule as defined in any of claims 1 to 11 and a pharmaceutically acceptable carrier.
17. A method for producing the MUC16-binding molecule comprising the steps of:
- culturing the host cell of claim 15 under suitable conditions to express the MUC16-binding molecule; and
- isolating the MUC16-binding molecule from the culture supernatant.
18. A method of modulating a MUC16 related immune response in a subject, comprising administering to the subject the MUC16-binding molecule as defined in any of claims 1 to 11 or the pharmaceutical composition of claim 16 such that an immune response is modulated in the subject.
19. A method for treating or preventing cancer in a subject, comprising administering an effective amount of the MUC16-binding molecule as defined in any of claims 1 to 11 or the pharmaceutical composition of claim 16 to the subject, wherein the cancer is MUC16 positive or overexpressed.
20. The method of claim 19, wherein the cancer is selected from ovary cancer, lung cancer, pancreas cancer, breast cancer, uterine cancer, fallopian tube cancer, primary peritoneum cancer or cancer of any other tissue that expresses MUC16.
21. Use of the MUC 16-binding molecule as defined in any of claims 1 to 11 in the manufacture of a medicament for diagnosing, preventing or treating MUC 16 positive cancer.
22. MUC16-binding molecule as defined in any of claims 1 to 11 for use in treating or preventing MUC16 positive cancer.
23. A kit for treating or diagnosing cancer, comprising a container comprising the MUC 16-binding molecule as defined in any of claims 1 to 11.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CNPCT/CN2023/112575 | 2023-08-11 | ||
| CN2023112575 | 2023-08-11 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2025036848A1 true WO2025036848A1 (en) | 2025-02-20 |
Family
ID=92408903
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2024/072621 Pending WO2025036848A1 (en) | 2023-08-11 | 2024-08-09 | Anti-muc16 antibodies and uses thereof |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2025036848A1 (en) |
Citations (19)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0183070A2 (en) | 1984-10-30 | 1986-06-04 | Phillips Petroleum Company | Transformation of yeasts of the genus pichia |
| WO1987004462A1 (en) | 1986-01-23 | 1987-07-30 | Celltech Limited | Recombinant dna sequences, vectors containing them and method for the use thereof |
| EP0244234A2 (en) | 1986-04-30 | 1987-11-04 | Alko Group Ltd. | Transformation of trichoderma |
| WO1989001036A1 (en) | 1987-07-23 | 1989-02-09 | Celltech Limited | Recombinant dna expression vectors |
| US4816567A (en) | 1983-04-08 | 1989-03-28 | Genentech, Inc. | Recombinant immunoglobin preparations |
| EP0338841A1 (en) | 1988-04-18 | 1989-10-25 | Celltech Limited | Recombinant DNA methods, vectors and host cells |
| EP0402226A1 (en) | 1989-06-06 | 1990-12-12 | Institut National De La Recherche Agronomique | Transformation vectors for yeast yarrowia |
| WO1994004678A1 (en) | 1992-08-21 | 1994-03-03 | Casterman Cecile | Immunoglobulins devoid of light chains |
| US5712120A (en) | 1994-06-30 | 1998-01-27 | Centro De Immunologia Molecular | Method for obtaining modified immunoglobulins with reduced immunogenicity of murine antibody variable domains, compositions containing them |
| US5861155A (en) | 1993-12-08 | 1999-01-19 | Astra Ab | Humanized antibodies and uses thereof |
| US5869619A (en) | 1991-12-13 | 1999-02-09 | Xoma Corporation | Modified antibody variable domains |
| US6054927A (en) | 1999-09-13 | 2000-04-25 | Eaton Corporation | Apparatus and method for sensing an object within a monitored zone |
| US6180370B1 (en) | 1988-12-28 | 2001-01-30 | Protein Design Labs, Inc. | Humanized immunoglobulins and methods of making the same |
| WO2003048731A2 (en) | 2001-12-03 | 2003-06-12 | Abgenix, Inc. | Antibody categorization based on binding characteristics |
| WO2007001851A2 (en) | 2005-06-20 | 2007-01-04 | Genentech, Inc. | Compositions and methods for the diagnosis and treatment of tumor |
| WO2011119979A2 (en) | 2010-03-26 | 2011-09-29 | Memorial Sloan-Kettering Cancer Center | Antibodies to muc16 and methods of use thereof |
| US20150079088A1 (en) | 2013-07-25 | 2015-03-19 | Cytomx Therapeutics, Inc. | Multispecific antibodies, multispecific activatable antibodies and methods of using the same |
| WO2018067331A1 (en) | 2016-09-23 | 2018-04-12 | Regeneron Pharmaceuticals, Inc. | Bi specific anti-muc16-cd3 antibodies and nti-muc16 drug conjugates |
| US20190241886A1 (en) | 2018-02-02 | 2019-08-08 | Adagene Inc. | Precision/context-dependent activatable antibodies, and methods of making and using the same |
-
2024
- 2024-08-09 WO PCT/EP2024/072621 patent/WO2025036848A1/en active Pending
Patent Citations (19)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4816567A (en) | 1983-04-08 | 1989-03-28 | Genentech, Inc. | Recombinant immunoglobin preparations |
| EP0183070A2 (en) | 1984-10-30 | 1986-06-04 | Phillips Petroleum Company | Transformation of yeasts of the genus pichia |
| WO1987004462A1 (en) | 1986-01-23 | 1987-07-30 | Celltech Limited | Recombinant dna sequences, vectors containing them and method for the use thereof |
| EP0244234A2 (en) | 1986-04-30 | 1987-11-04 | Alko Group Ltd. | Transformation of trichoderma |
| WO1989001036A1 (en) | 1987-07-23 | 1989-02-09 | Celltech Limited | Recombinant dna expression vectors |
| EP0338841A1 (en) | 1988-04-18 | 1989-10-25 | Celltech Limited | Recombinant DNA methods, vectors and host cells |
| US6180370B1 (en) | 1988-12-28 | 2001-01-30 | Protein Design Labs, Inc. | Humanized immunoglobulins and methods of making the same |
| EP0402226A1 (en) | 1989-06-06 | 1990-12-12 | Institut National De La Recherche Agronomique | Transformation vectors for yeast yarrowia |
| US5869619A (en) | 1991-12-13 | 1999-02-09 | Xoma Corporation | Modified antibody variable domains |
| WO1994004678A1 (en) | 1992-08-21 | 1994-03-03 | Casterman Cecile | Immunoglobulins devoid of light chains |
| US5861155A (en) | 1993-12-08 | 1999-01-19 | Astra Ab | Humanized antibodies and uses thereof |
| US5712120A (en) | 1994-06-30 | 1998-01-27 | Centro De Immunologia Molecular | Method for obtaining modified immunoglobulins with reduced immunogenicity of murine antibody variable domains, compositions containing them |
| US6054927A (en) | 1999-09-13 | 2000-04-25 | Eaton Corporation | Apparatus and method for sensing an object within a monitored zone |
| WO2003048731A2 (en) | 2001-12-03 | 2003-06-12 | Abgenix, Inc. | Antibody categorization based on binding characteristics |
| WO2007001851A2 (en) | 2005-06-20 | 2007-01-04 | Genentech, Inc. | Compositions and methods for the diagnosis and treatment of tumor |
| WO2011119979A2 (en) | 2010-03-26 | 2011-09-29 | Memorial Sloan-Kettering Cancer Center | Antibodies to muc16 and methods of use thereof |
| US20150079088A1 (en) | 2013-07-25 | 2015-03-19 | Cytomx Therapeutics, Inc. | Multispecific antibodies, multispecific activatable antibodies and methods of using the same |
| WO2018067331A1 (en) | 2016-09-23 | 2018-04-12 | Regeneron Pharmaceuticals, Inc. | Bi specific anti-muc16-cd3 antibodies and nti-muc16 drug conjugates |
| US20190241886A1 (en) | 2018-02-02 | 2019-08-08 | Adagene Inc. | Precision/context-dependent activatable antibodies, and methods of making and using the same |
Non-Patent Citations (81)
| Title |
|---|
| "Antibody Engineering", 2001, SPRINGER |
| "Bispecific Antibodies", 2011 |
| "Computer Analysis of Sequence Data, Part I", 1994, JOHN WILEY & SONS, pages: 603 - 6410 |
| "GenBank", Database accession no. NP 078966.2 |
| "Molecular Cloning: A laboratory Manual", 1989, COLD SPRING HARBOR LABORATORY PRESS, pages: 47 - 51 |
| "NCBI", Database accession no. XP_015296314.1 |
| "UniProt", Database accession no. AOA140LJ72 |
| ABBAS ET AL.: "Cellular and Molecular Immunology", 2010, W.B. SAUNDERS COMPANY |
| ADIB-CONQUY ET AL., INT. IMMUNOL., vol. 10, 1998, pages 341 - 6 |
| AITHAL ARAUTH SKSHIRSAGAR P ET AL.: "MUC16 as a novel target for cancer therapy [J", EXPERT OPINION ON THERAPEUTIC TARGETS, vol. 22, no. 8, 2018, pages 675 - 686, XP093121913, DOI: 10.1080/14728222.2018.1498845 |
| AL-LAZIKANI ET AL., J. MOLEC. BIOL., vol. 273, 1997, pages 927 - 948 |
| ALTSCHUL ET AL., J. MOL. BIOL., vol. 215, 1990, pages 403 - 10 |
| ALTSCHUL ET AL., NUCLEIC ACIDS RES., vol. 25, no. 17, 1997, pages 3389 - 3402 |
| BARBAS ET AL., PROC. NATL. ACAD. SCI. USA, vol. 88, 1991, pages 7978 - 7982 |
| BAST R CFEENEY MLAZARUS H ET AL.: "Reactivity of a monoclonal antibody with human ovarian carcinoma[J", THE JOURNAL OF CLINICAL INVESTIGATION, vol. 68, no. 5, 1981, pages 1331 - 1337, XP003002433, DOI: 10.1172/JCI110380 |
| BEERS ET AL., CLIN. CAN. RES., vol. 6, 2000, pages 2835 - 43 |
| BEREK J STAYLOR P TGORDON A ET AL.: "Randomized, placebo-controlled study of oregovomab for consolidation of clinical remission in patients with advanced ovarian cancer[J", JOURNAL OF CLINICAL ONCOLOGY, vol. 22, no. 17, 2004, pages 3507 - 3516, XP055532136, DOI: 10.1200/JCO.2004.09.016 |
| BIO/TECHNOLOGY, vol. 10, 1992, pages 169 |
| BRUMMELL ET AL., BIOCHEM, vol. 32, 1993, pages 1180 - 8 |
| BURKS ET AL., PROC. NATL. ACAD. SCI. USA, vol. 94, 1997, pages 412 - 417 |
| CARILLO ET AL., SIAMJ. APPLIED MATH., vol. 48, 1988, pages 1073 |
| CHOTHIA ET AL., NATURE, vol. 342, 1989, pages 877 |
| CHOTHIA, C. ET AL., J. MOL. BIOL., vol. 196, 1987, pages 901 - 917 |
| CHU ET AL., GENE, vol. 13, 1981, pages 197 |
| CLARK, IMMUNOLOGY TODAY, vol. 21, no. 8, 2000, pages 397 - 402 |
| DALL' ACQUA ET AL., METHODS, vol. 36, no. 1, 2005, pages 43 - 60 |
| DAS SMAJHI P DAL-MUGOTIR MH ET AL.: "Membrane proximal ectodomain cleavage of MUC16 occurs in the acidifying Golgi/post-Golgi compartments [J", SCIENTIFIC REPORTS, vol. 5, no. 1, 2015, pages 9759 |
| DAS SRUSTIDHAR ET AL: "Membrane proximal ectodomain cleavage of MUC16 occurs in the acidifyingGolgi/post-Golgi compartments", SCIENTIFIC REPORTS, vol. 5, no. 1, 5 June 2015 (2015-06-05), US, XP093214182, ISSN: 2045-2322, Retrieved from the Internet <URL:https://www.nature.com/articles/srep09759> DOI: 10.1038/srep09759 * |
| DAS SRUSTIDHAR ET AL: "Understanding the Unique Attributes of MUC16 (CA125): Potential Implications in Targeted Therapy", CANCER RESEARCH, vol. 75, no. 22, 15 November 2015 (2015-11-15), San Diego, CA . Philadelphia (PA, pages 4669 - 4674, XP093057260, ISSN: 0008-5472, Retrieved from the Internet <URL:https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4651718/pdf/nihms-713157.pdf> DOI: 10.1158/0008-5472.CAN-15-1050 * |
| DE WILDT ET AL., PROT. ENG., vol. 10, 1997, pages 835 - 41 |
| DEFFAR ET AL., AFRICAN JOURNAL OF BIOTECHNOLOGY, vol. 8, no. 12, 17 June 2009 (2009-06-17), pages 2645 |
| DHARMA RAO ET AL: "Novel Monoclonal Antibodies against the Proximal (Carboxy-Terminal) Portions of MUC16", 1 October 2010 (2010-10-01), XP093213978, Retrieved from the Internet <URL:https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4388147/pdf/nihms-416843.pdf> DOI: 10.1097/PAI * |
| DR. ANDREW C. R.: "Antibody Engineering Lab Manual", SPRINGER-VERLAG, article "Protein Sequence and Structure Analysis of Antibody Variable Domains" |
| E. MEYERSW. MILLER, COMPUT. APPL. BIOSCI., vol. 4, 1988, pages 11 - 17 |
| EDELMAN ET AL., PROC NATL ACAD SCI U S A., vol. 3, no. 1, 6 May 1969 (1969-05-06), pages 78 - 85 |
| EPITOPE MAPPING PROTOCOLS IN METHODS IN MOLECULAR BIOLOGY, vol. 66, 1996 |
| FLOWMETRIC, SORTING OUT FLUORESCENCE ACTIVATED CELL SORTING, 9 November 2017 (2017-11-09) |
| GIUDICELLI V ET AL.: "IMGT, the international ImMunoGeneTics database", NUCLEIC ACIDS RES., vol. 25, 1997, pages 206 - 11 |
| GRAHAM ET AL., J. GEN VIROL., vol. 36, 1977, pages 59 |
| GRAHAM ET AL., VIROLOGY, vol. 52, 1973, pages 456 |
| HALL ET AL., J. IMMUNOL., vol. 149, 1992, pages 1605 - 12 |
| HARIDAS DPONNUSAMY M PCHUGH S ET AL.: "MUC16: molecular analysis and its functional implications in benign and malignant conditions [J", THE FASEB JOURNAL, vol. 28, no. 10, 2014, pages 4183 - 4199, XP002762382 |
| HOFER ET AL., BIOCHEMISTRY, vol. 48, no. 50, 2009, pages 12047 - 57 |
| HOFER ET AL., PROC. NATL. ACAD. SCI. USA, vol. 105, 2008, pages 12451 - 56 |
| JOHNNSON, B. ET AL., ANAL. BIOCHEM., vol. 198, 1991, pages 268 - 277 |
| JOHNSSON, B. ET AL., J. MOL. RECOGNIT., vol. 8, 1995, pages 125 - 131 |
| JÖNSSON, U. ET AL., ANN. BIOL. CLIN., vol. 51, 1993, pages 19 - 26 |
| JÖNSSON, U. ET AL., BIOTECHNIQUES, vol. 11, 1991, pages 620 - 627 |
| JUNUTULA ET AL., J. IMMUNOL. METH., vol. 332, 2008, pages 41 - 52 |
| JUNUTULA ET AL., NATURE BIOTECHNOL., vol. 26, 2008, pages 925 - 32 |
| KABAT, E.A. ET AL.: "Sequences of Proteins of Immunological Interest", 1991, DEPARTMENT OF HEALTH AND HUMAN SERVICES |
| KELLEYO' CONNELL, BIOCHEM., vol. 32, 1993, pages 1180 - 1187 |
| KIM Y WBAE S MKIM I W ET AL.: "Multiplexed bead-based immunoassay of four serum biomarkers for diagnosis of ovarian cancer[J", ONCOLOGY REPORTS, vol. 28, no. 2, 2012, pages 585 - 591 |
| KOBAYASHI ET AL., PROTEIN ENG., vol. 12, no. 10, 1999, pages 879 - 884 |
| KOCH-NOLTE F. ET AL., FASEB J., no. 13, 15 June 2007 (2007-06-15), pages 3490 - 8 |
| KOMISSAROV ET AL., J. BIOL. CHEM., vol. 272, 1997, pages 26864 - 26870 |
| LEFRANC MP ET AL.: "IMGT unique numbering for immunoglobulin and T cell receptor variable domains and Ig superfamily V-like domains", DEV COMP IMMUNOL., vol. 27, 2003, pages 55 - 77, XP055585227, DOI: 10.1016/S0145-305X(02)00039-3 |
| LI ET AL., J BIOL CHEM., vol. 287, 2012, pages 13713 - 13721 |
| LIEDE AKARLAN B YBALDWIN R L ET AL.: "Cancer incidence in a population of Jewish women at risk of ovarian cancer[J", JOURNAL OF CLINICAL ONCOLOGY, vol. 20, no. 6, 2002, pages 1570 - 1577 |
| MARTINALLEN, HANDBOOK OF THERAPEUTIC ANTIBODIES, vol. 5, 2007 |
| MATHER ET AL., ANNALS N.Y. ACAD. SCI., vol. 383, 1982, pages 44 - 68 |
| MATHER, BIOL. REPROD., vol. 23, 1980, pages 243 - 251 |
| MILSTEINCUELLO, NATURE, vol. 305, 1983, pages 537 - 40 |
| MORALES-VASQUEZ FPEDERNERA EREYNAGA-OBREGON J ET AL.: "High levels of pretreatment CA125 are associated to improved survival in high grade serous ovarian carcinoma[J", JOURNAL OF OVARIAN RESEARCH, vol. 9, no. 1, 2016, pages 1 - 6 |
| NEEDLEMANWUNSCH, J. MOL. BIOL., vol. 48, 1970, pages 444 - 453 |
| O'BRIEN TJ ET AL., TUMOR BIOL, vol. 22, no. 6, 2001, pages 348 - 66 |
| R. J. KAUFMANP. A. SHARP, J. MOL. BIOL., vol. 159, 1982, pages 601 - 621 |
| R. VAN DER LINDEN ET AL., JOURNAL OF IMMUNOLOGICAL METHODS, vol. 240, 2000, pages 185 - 195 |
| RETTER ET AL., NUCL. ACIDS RES., vol. 33, 2005, pages D671 - D674 |
| SABBATINI PHARTER PSCAMBIA G ET AL.: "Abagovomab as maintenance therapy in patients with epithelial ovarian cancer: a phase III trial of the AGO OVAR, COGI, GINECO, and GEICO-the MIMOSA study [J", JOURNAL OF CLINICAL ONCOLOGY, vol. 31, no. 12, 2013, pages 1554 |
| SOMATIC CELL AND MOLECULAR GENETICS, vol. 12, 1986, pages 555 |
| SRUSTIDHAR DAS ET AL.: "Understanding the Unique Attributes of MUC16 (CA125): Potential Implications in Targeted Therapy", CANCER RESEARCH, 2015 |
| STREPPEL M MVINCENT AMUKHERJEE R ET AL.: "Mucin 16 (cancer antigen 125) expression in human tissues and cell lines and correlation with clinical outcome in adenocarcinomas of the pancreas, esophagus, stomach, and colon[J", HUMAN PATHOLOGY, vol. 43, no. 10, 2012, pages 1755 - 1763, XP028939577, DOI: 10.1016/j.humpath.2012.01.005 |
| URLAUB ET AL., PROC. NATL. ACAD. SCI. USA, vol. 77, 1980, pages 4216 |
| URLAUBCHASIN, PROC. NATL. ACAD. SCL USA, vol. 77, 1980, pages 4216 - 4220 |
| WESOLOWSKI ET AL.: "Single domain antibodies: promising experimental and therapeutic tools in infection and immunity", MED MICROBIOL IMMUNOL, vol. 198, 2009, pages 157 - 174 |
| WHITE BRANDY ET AL: "Crystal structure of a human MUC16 SEA domain reveals insight into the nature of the CA125 tumor marker", PROTEINS: STRUCTURE, FUNCTION, AND BIOINFORMATICS, vol. 90, no. 5, 25 January 2022 (2022-01-25), US, pages 1210 - 1218, XP093214187, ISSN: 0887-3585, Retrieved from the Internet <URL:https://onlinelibrary.wiley.com/doi/full-xml/10.1002/prot.26303> DOI: 10.1002/prot.26303 * |
| YIN B W T ET AL: "Molecular cloning of the CA125 ovarian cancer antigen", JOURNAL OF BIOLOGICAL CHEMISTRY, AMERICAN SOCIETY FOR BIOCHEMISTRY AND MOLECULAR BIOLOGY, US, vol. 276, no. 29, 20 July 2001 (2001-07-20), pages 27371 - 27375, XP002953227, ISSN: 0021-9258, DOI: 10.1074/JBC.M103554200 * |
| YIN B W TDNISTRIAN ALLOYD K O: "Ovarian cancer antigen CA125 is encoded by the MUC16 mucin gene[J", INTERNATIONAL JOURNAL OF CANCER, vol. 98, no. 5, 2002, pages 737 - 740, XP071280850, DOI: 10.1002/ijc.10250 |
| YIN BEATRICE W T ET AL: "Ovarian cancer antigen CA125 is encoded by the MUC16 mucin gene", INTERNATIONAL JOURNAL OF CANCER, JOHN WILEY & SONS, INC, US, vol. 98, no. 5, 10 April 2002 (2002-04-10), pages 737 - 740, XP009143084, ISSN: 0020-7136, DOI: 10.1002/IJC.10250 * |
| ZHANG ET AL., MOLECULAR IMMUNOLOGY, vol. 42, no. 12, 2005, pages 1445 - 1451 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US20240301067A1 (en) | Novel anti-pd-1 antibodies | |
| US12371493B2 (en) | Monoclonal antibody against human LAG-3, method for preparing the same, and use thereof | |
| WO2019022187A1 (en) | ANTIBODY ANTI CD147 | |
| KR102866812B1 (en) | Novel anti-PD-L1 antibodies | |
| WO2023040940A1 (en) | Use of pvrig/tigit binding protein in combination with immune checkpoint inhibitor in treatment of cancers | |
| WO2023222017A1 (en) | Anti-b7h3 antibody and uses thereof | |
| WO2021057930A1 (en) | A novel anti-pd-l1/anti-lag-3 bispecific antibody and uses thereof | |
| WO2023041041A1 (en) | D3-binding molecules and uses thereof | |
| WO2025036848A1 (en) | Anti-muc16 antibodies and uses thereof | |
| TWI847270B (en) | D3 binding molecules and their uses | |
| WO2024146553A1 (en) | Antibodies against cd47, method for preparing the same, and use thereof | |
| WO2025104253A1 (en) | Anti-muc16 antibodies and uses thereof | |
| WO2025026333A1 (en) | Antibody against fgfr2b, method for preparing the same, and use thereof | |
| WO2024251140A1 (en) | Anti-tigit antibodies and uses thereof | |
| WO2025061165A1 (en) | Antibodies against pvrig or tigit, bispecific antibodies constructed therefrom, methods for their preparation and uses thereof | |
| WO2024146539A1 (en) | Bispecific antibodies against pd-l1 and cd47, method for preparing the same, and use thereof | |
| KR20230119179A (en) | Antibodies to P-cadherin and uses thereof |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 24755244 Country of ref document: EP Kind code of ref document: A1 |