WO2025036643A1 - Biosurfactant for washing wool - Google Patents
Biosurfactant for washing wool Download PDFInfo
- Publication number
- WO2025036643A1 WO2025036643A1 PCT/EP2024/070359 EP2024070359W WO2025036643A1 WO 2025036643 A1 WO2025036643 A1 WO 2025036643A1 EP 2024070359 W EP2024070359 W EP 2024070359W WO 2025036643 A1 WO2025036643 A1 WO 2025036643A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- sbio
- aqueous mixture
- rhamnolipids
- biosurfactant
- preferred
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D1/00—Detergent compositions based essentially on surface-active compounds; Use of these compounds as a detergent
- C11D1/66—Non-ionic compounds
- C11D1/662—Carbohydrates or derivatives
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/60—Sugars; Derivatives thereof
- A61K8/602—Glycosides, e.g. rutin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61Q—SPECIFIC USE OF COSMETICS OR SIMILAR TOILETRY PREPARATIONS
- A61Q19/00—Preparations for care of the skin
- A61Q19/10—Washing or bathing preparations
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D2111/00—Cleaning compositions characterised by the objects to be cleaned; Cleaning compositions characterised by non-standard cleaning or washing processes
- C11D2111/10—Objects to be cleaned
- C11D2111/12—Soft surfaces, e.g. textile
Definitions
- the present invention relates to a method of washing a woolen article Aw-
- the method comprises a step in which Aw is contacted with an aqueous mixture L comprising at least one biosurfactant SBIO-
- the present invention furthermore relates to the use of at least one biosurfactant SBIO to reduce shrinking of at least one woolen article Aw during washing of Aw, wherein, during washing of Aw, Aw is contacted with an aqueous mixture L comprising at least one biosurfactant SBIO.
- the biosurfactant SBIO is selected from the group consisting of rhamnolipids, glucolipids, sophorolipids.
- the biosurfactant SBIO is selected from the group consisting of rhamnolipids, sophorolipids.
- the biosurfactant SBIO is selected from the group consisting of rhamnolipids.
- CN 102 660 392 A discloses a detergent for woolen articles comprising certain non-ionic surfactants such as tea saponins.
- CN 106 854 531 A discloses plant extracts from Angelica, Arnica, tea tree comprising tea saponines as surfactant in anti-shrinkage machine washing liquids for cashmere and woolen articles.
- US 2021/0171864 A1 discloses the use of biosurfactants such as rhamnolipids and sophorolipids for improving the color stability (“crockfastness”) of dyed fabrics such as wool.
- EP 0 439 019 A1 discloses the use of alkyl glycosides to prevent felting shrinkage during washing.
- WO 2022/162043 A1 discloses rhamnolipids as possible surfactants for washing a wide range of textiles, among those wool.
- the examples of WO 2022/162043 A1 disclose the washing of cotton swatches with a rhamnolipid, but not with wool.
- GB 1 515 792 A discusses specific problems in washing of wool, in particular with respect to felting and shrinking of wool during the wash.
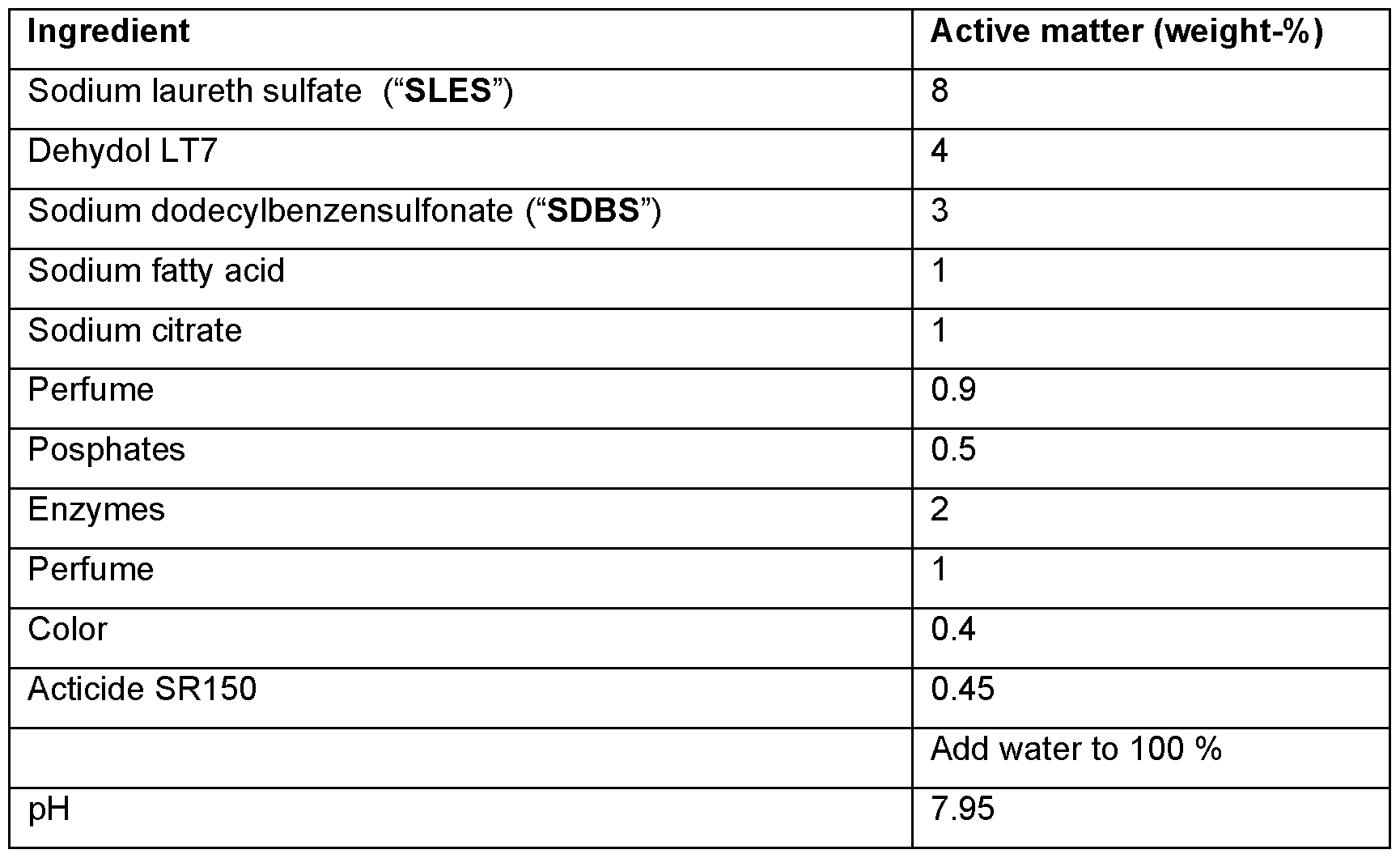
- a wool detergent comprising different surfactants, among these anionic surfactants such as sodium lauryl sulphate (“SLES”).
- SLES sodium lauryl sulphate
- biosurfactants such as tea saponins, alkyl glycosides and other plant extracts which have a mild effect on woolen garments and reduce shrinking
- these biosurfactants are also mild in their washing efficiency and cannot compete with well-established anionic surfactants such as sodium laureth sulfate (“SLES”), sodium dodecylbenzensulfonate (“SDBS”), or non-ionic surfactants such as ethoxylated alcohols.
- SLES sodium laureth sulfate
- SDBS sodium dodecylbenzensulfonate
- non-ionic surfactants such as ethoxylated alcohols.
- this wool detergent should have a similar washing efficiency as the well-established anionic and non-ionic surfactants.
- biosurfactants SBIO which are selected from the group consisting of rhamnolipids, glucolipids, sophorolipids, and preferably are selected from the group of rhamnolipids, reduce the shrinking of woolen articles during washing.
- the present invention hence relates to use of at least one biosurfactant SBIO, which is selected from the group consisting of rhamnolipids, glucolipids, sophorolipids, and in particular is selected from the group consisting of rhamnolipids, sophorolipids, and preferably is selected from the group consisting of rhamnolipids, for washing at least one woolen article Aw- During washing of Aw, Aw is contacted with an aqueous mixture L.
- This aqueous mixture L comprises the at least one biosurfactant SBIO-
- the present invention relates to a method of washing at least one woolen article Aw, comprising the steps of a. contacting the at least one woolen article Aw with an aqueous mixture L comprising at least one biosurfactant SBIO, wherein the at least one biosurfactant SBIO is selected from the group consisting of rhamnolipids, glucolipids, sophorolipids, and in particular is selected from the group consisting of rhamnolipids, sophorolipids, and preferably is selected from the group consisting of rhamnolipids, b. at least partially separating L from Aw, c. and optionally rinsing Aw with water.
- the temperature of the aqueous mixture L, for at least as long as the aqueous mixture L is contacted with the at least one woolen article Aw is in a range of from > 0 °C to 45 °C, in particular in a range of from 1 °C to 40 °C, preferably in a range of from 5 °C to 35 °C, more preferably in a range of from 10 °C to 30 °C, more preferably in a range of from 15 °C to 30 °C, even more preferably in a range of from 20 °C to 30 °C, even more preferably in a range of from 25 °C to 30 °C.
- the present invention relates to the use of at least one biosurfactant SBIO to reduce shrinking of at least one woolen article Aw during washing of the at least one a woolen article Aw- During washing of Aw, Aw is contacted with an agueous mixture L comprising at least one biosurfactant SBIO.
- the biosurfactant SBIO is selected from the group consisting of rhamnolipids, glucolipids, sophorolipids.
- the biosurfactant SBIO in particular, is selected from the group consisting of rhamnolipids, sophorolipids.
- the biosurfactant SBIO is preferably selected from rhamnolipids.
- the at least one biosurfactant SBIO is used in an agueous mixture L to reduce shrinking of the at least one woolen article Aw during washing of Aw in the agueous mixture L.
- the agueous mixture L hence comprises the at least one biosurfactant L and the at least one woolen article Aw-
- the article Aw essentially consists of wool.
- the woolen article Aw in particular is an article of clothing, preferably selected from pants, shirt, socks, hosiery, scarfs, gloves, shoes, sweaters, suits, hats, caps, bonnets. It may, in addition, also be selected from the group consisting of curtains, napkins, bed linen, tablecloth, blanket, bedspread, duvet cover.
- wool refers to the textile fiber of a mammal, in particular of livestock.
- it refers to the textile fiber of a mammal selected from the group consisting of sheep, goat (the goat being in particular selected from the group of cashmere goat, angora goat), rabbit (the rabbit being in particular an angora rabbit), camelide (the camelide being in particular selected from the group of llama, vikuna, alpaca, guanaco), yak, beaver, otter, guinea pig, horse, donkey, chinchilla.
- it refers to the textile fiber of a mammal selected from the group consisting of sheep, goat, most preferably of sheep.
- At least one biosurfactant SBIO reduces shrinking of the at least one woolen article Aw during washing in the aqueous mixture L comprising the at least one woolen article Aw and the at least one biosurfactant SBIO-
- Micronking or shrinkage of woolen fabrics is a natural phenomenon and occurs during washing.
- Fabric shrinkage is the reduction in the size or dimensions of a fabric or garment after being exposed to heat or undergoing washing. Shrinkage can occur in both lengthwise (warp) and widthwise (weft) directions.
- Woolen articles such as woolen fabrics are particularly prone to shrinkage during washing, in particular when elevated temperatures (above 20 °C) are applied and when the woolen article is moved in the aqueous mixture in which it is washed (i.e. the washing mixture) during washing, which is typically the case when agitation is applied during washing.
- Woolen articles are washed at low temperatures to not strain the woolen fibers.
- the temperature of the washing mixture does not exceed 45 °C.
- “Reduce shrinking of at least one woolen article Aw during washing of Aw” means that the extent of shrinking of Aw in an aqueous mixture L comprising the at least one biosurfactant SBIO is reduced during washing, compared to the case where Aw is washed under the same conditions (for example under the same temperature, washing program, pressure) in an aqueous mixture Lo, wherein Lo and L are identical, except that Lo comprises water instead of the at least one biosurfactant SBIO- Hence, from a formal point of view, Lo is obtained by substituting the mass of all biosurfactants SBIO comprised by L by the same mass of water.
- “Reduce shrinking of at least one woolen article Aw during washing of Aw” is therefore to be understood in the sense that Aw is at least partially protected against shrinking during washing when at least one biosurfactant SBIO is comprised by the aqueous mixture L which contacts, preferably comprises, the at least one woolen article Aw during washing.
- this reduction of shrinking (i.e. protection or resilience against shrinking) conferred by the at least one biosurfactant SBIO in the context of the present invention is higher than the reduction of shrinking conferred by water alone.
- this “reduction of shrinking” is improved not only compared to washing in pure water, but also in comparison with the shrinking during washing in aqueous mixtures comprising other surfactants.
- Ly is obtained from aqueous mixture L by substituting the mass of all biosurfactants SBIO comprised by L by the same mass of at least one surfactant Sy.
- Aqueous mixture LY comprises at least one surfactant Sy in the same amount (relative to the total weight of the aqueous mixture Ly) as the aqueous mixture L comprises the at least one biosurfactant SBIO (relative to the total weight of the aqueous mixture L).
- Aqueous mixture Ly does not comprise a biosurfactant SBIO- Hence, LY has the same mass mi.
- LY comprises XL wt.-% (relative to the total mass of mixture LY) of at least one surfactant SY.
- SY is different from SBIO and is in particular a surfactant with a comparable washing profile and efficiency.
- SY is selected from the group consisting of SLES, ethoxylated alcohols. More preferably SY is selected from the group consisting of SLES, a mixture of SLES and ethoxylated alcohols.
- Whether a certain woolen article Aw shrinks, as well as the extent of shrinking of a certain woolen article Aw during washing may be determined by methods that are known to the skilled person. Fabric shrinkage tests are typically performed according to recognized industry standards.
- AATCC TM135 Dimensional Changes in Automatic Home Laundering of Woven and Knit Fabrics.
- Assay X is the preferred test to determine the reduction of shrinking according to the present invention.
- Assay X is used, in particular, to determine the reduction of shrinking of the at least one woolen article Aw during washing in an aqueous mixture L, as well as the extent of shrinking of the at least one woolen article Aw during washing in an aqueous mixture L.
- Assay X is hence the preferred test to determine, when the at least one biosurfactant SBIO is used according to the first aspect of the present invention, the reduction of shrinking of the at least one woolen article Aw during washing, in particular in an aqueous mixture L, as well as the extent of shrinking of the at least one woolen article Aw during washing, in particular in an aqueous mixture L.
- additive X is preferably used according to the present invention to assess whether a given additive X reduces shrinking of a woolen article Aw-
- the additive to be tested is hereinafter abbreviated as “X”.
- Additive X may be a biosurfactant SBIO which may be employed in Assay X directly or as a mixture, in particular an aqueous mixture.
- Additive X may, alternatively, be a surfactant SY, wherein SY is different from the biosurfactant SBIO-
- additive X may be, in a further alternative, any other compound or mixture of compounds, which is to be investigated in terms of their efficiency to reduce shrinking of a woolen article.
- Additive X may be a pure compound or a mixture of compounds (such as an aqueous mixture).
- Assay X is divided into two parts, part 1 and part 2.
- a blank test is carried out, in which the shrinking of a model woolen fabric is tested.
- distilled water is used as “blank” detergent formulation.
- the shrinking of the model woolen fabric is measured.
- the water that is used has the same water hardness of 14 ° dGH or less.
- distilled water is used in part 1 and part 2.
- the additive X may be a pure compound or an aqueous mixture, preferably is an aqueous mixture, even more preferably is an aqueous mixture in which the additive X has a concentration of 1 weight-% to 50 weight-%, preferably 2 weight-% to 30 weight-%, more preferably 3 weight-% to 20 weight-%, even more preferably 4 weight-% to 15 weight-%, even more preferably 5 weight-% to 15 weight-%, even more preferably 6 weight-% to 15 weight-%, even more preferably 8 to 15 weight-%, even more preferably 10 to 15 weight-% even more preferably 12 to 15 weight-% (in each case relative to the total weight of the aqueous mixture).
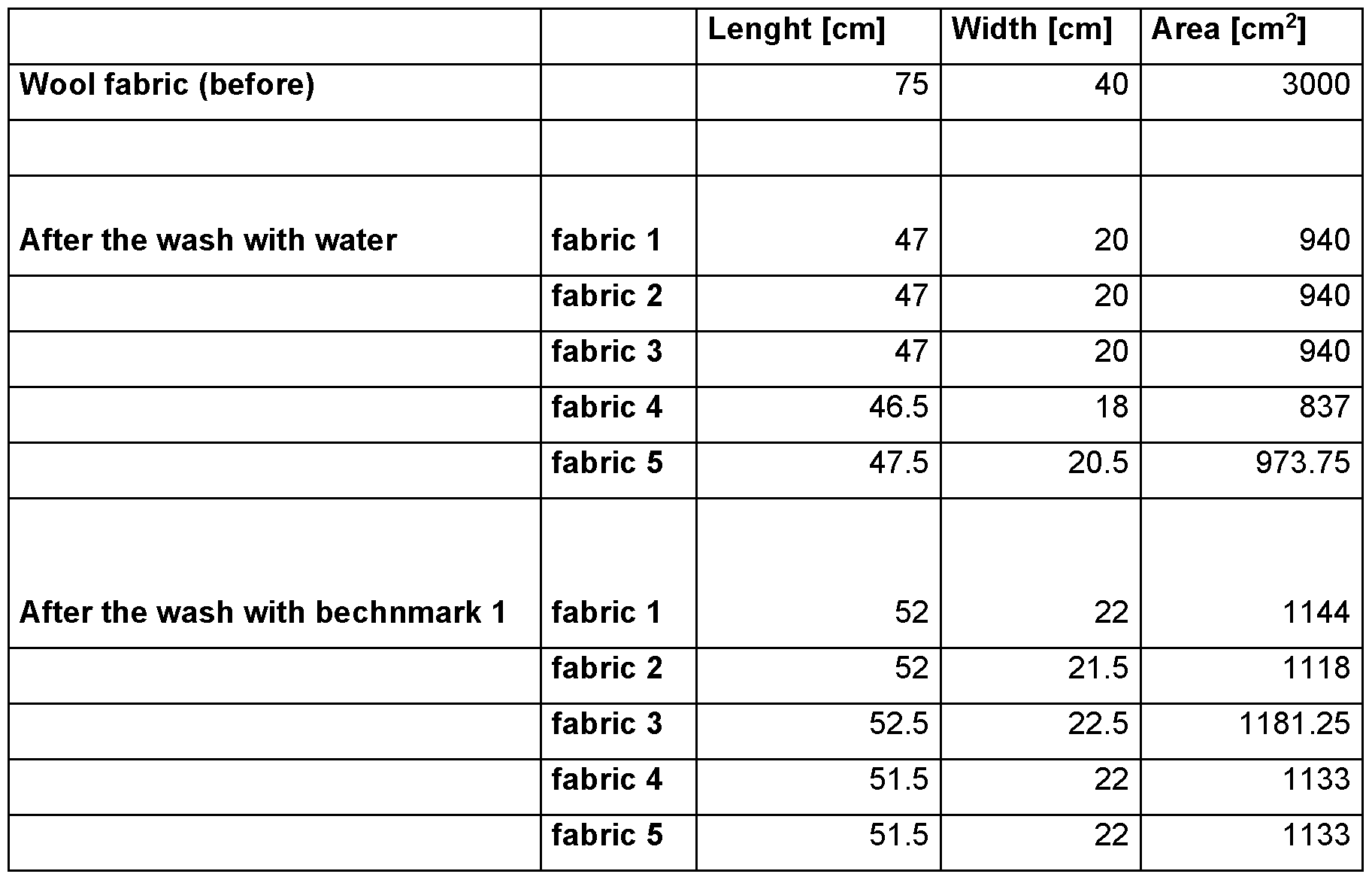
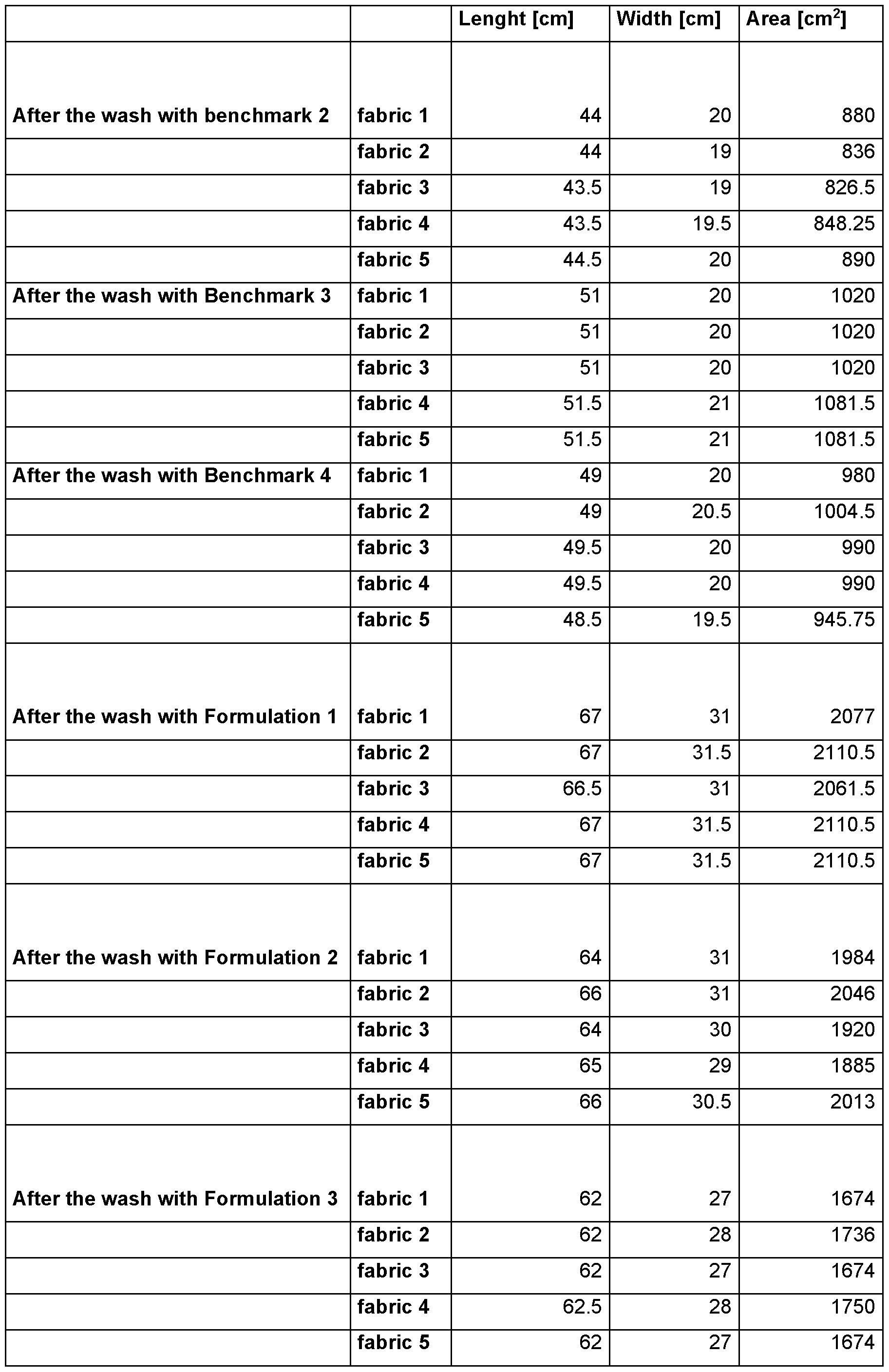
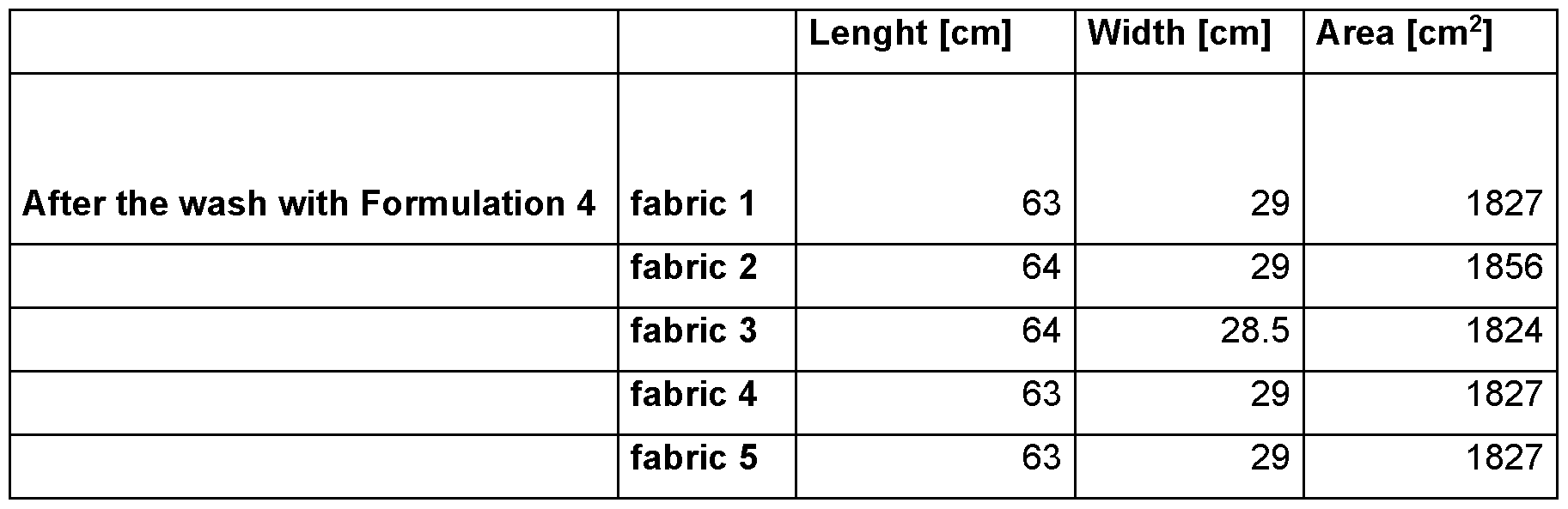
- the testing wool textiles are standard white unsoiled fabrics provided by wfk-Testgewebe GmbH (“WFK 60 A”, order code 60000; 100 % wool fabric).
- test swatches five of these standard wool textiles (abbreviated hereinafter as “test swatches”) are used and the area before (“Ao”) and the area after (in case of part 1 : “Asiank”; in case of part 2: “Ax”) the test run are measured to assess the extent of shrinking.
- Ao is the average area of the five standard wool textile swatches that is measured and calculated before the test procedure in part 1 or part 2. Since standard wool textiles swatches of the same size are used in part 1 and part 2, Ao in part 1 and Ao in part 2 are the same.
- Aeiank is the average area of the five standard wool textile swatches that is measured and calculated after the test procedure of part 1 .
- Ax is the average area of the five standard wool textile swatches that is measured and calculated after the test procedure of part 2.
- Ssiank (Ao - Asiank) I Ao.
- Sx (Ao - Ax) I Ao.
- Xi is deemed to be superior to X2 in reducing shrinking during washing if Sxi ⁇ Sx2-
- X2 is deemed to be superior to Xi in reducing shrinking during washing if Sx2 ⁇ Sxi.
- additive X is added into the dosing drawer.
- X may be added as the compound or as an aqueous mixture, for example, an aqueous mixture that contains the additive X in a concentration of 1 weight-% to 50 weight-%, preferably 2 weight-% to 30 weight-%, more preferably 3 weight-% to 20 weight-%, even more preferably 4 weight-% to 15 weight-%, even more preferably 5 weight-% to 15 weight-%, even more preferably 6 weight-% to 15 weight-%, even more preferably 8 to 15 weight-%, even more preferably 10 to 15 weight-% even more preferably 12 to 15 weight-% (in each case relative to the total weight of aqueous mixture).
- X is most preferably added as a 15 weight-% aqueous mixture of X, in particular where a biosurfactant SBIO is tested in Assay X.
- the “biosurfactant SBIO” is selected from the group consisting of glucolipids, rhamnolipids, sophorolipids, in particular selected from the group of sophorolipids, rhamnolipids, preferably from the group of rhamnolipids.
- Glucolipids, rhamnolipids, and sophorolipids are glycolipids. Such glycolipids are described in the art together with their syntheses, for example in EP 0499434 A1 (glucolipids are referred to as “glucose lipids” in this document), DE 196 48 439 A1 , DE 196 00 743 A1 .
- WO 03/006146 A1 , US 2008/0213194 A1 , JP H01 -304034 A1 , CN 1337439 A describe further methods for the synthesis of rhamnolipids.
- WO 03/002700 A1 , US 4,305,961 A, US 7,556,654 B1 describe further methods for the synthesis of sophorolipids.
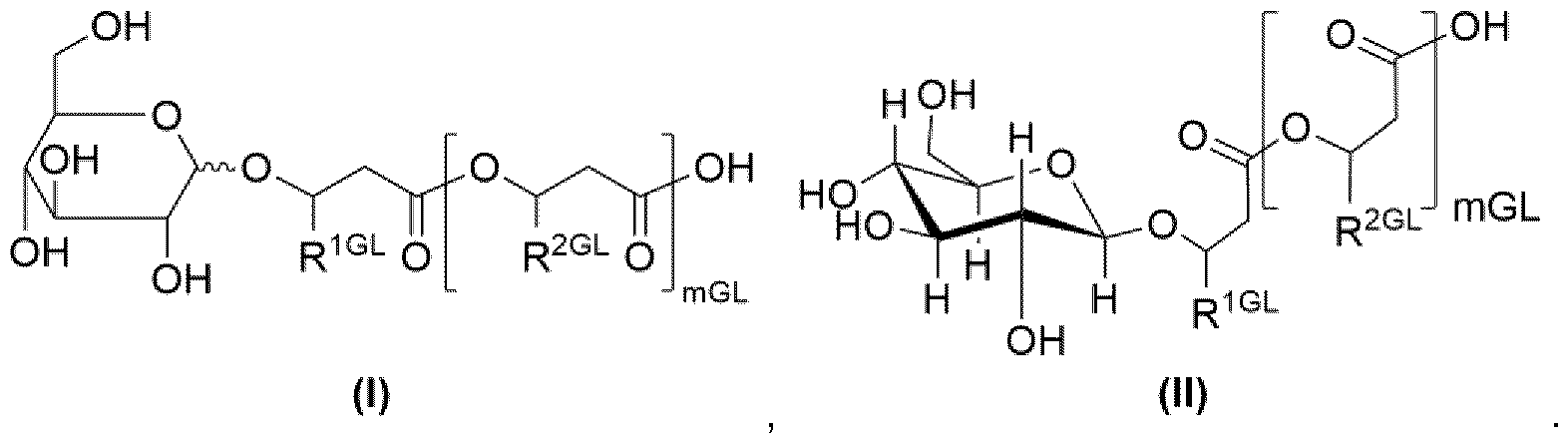
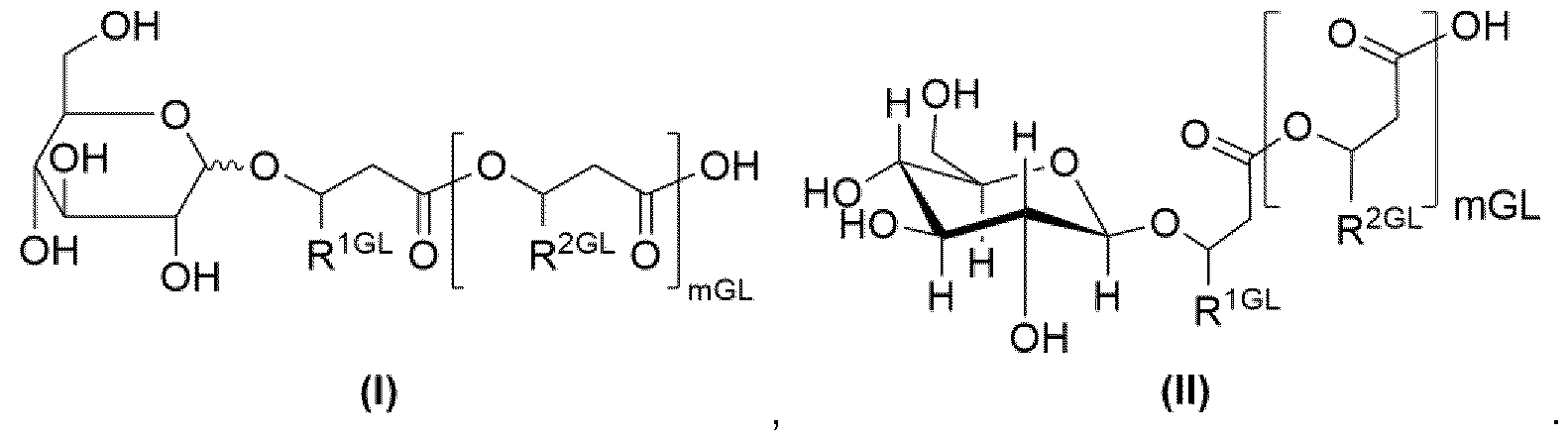
- glucolipid is, in particular, to be understood as referring to a structure according to formula (I), even more preferred a structure according to formula (II):
- mGL 3, 2, 1 or 0, preferably 1 or 0.
- Residues R 1GL and R 2GL are, independently of one another, an organic radical having 2 to 24 carbon atoms, preferably 5 to 20, more preferably 7 to 15, even more preferably 7, carbon atoms, wherein preferably R 1GL and R 2GL are independently of one another selected from the group consisting of optionally substituted alkyl radicals with 2 to 24 preferably 5 to 20, more preferably 7 to 15, even more preferably 7, carbon atoms, wherein hydroxy substituted alkyl radicals are preferred substituted alkyl radicals, optionally substituted alkenyl radicals with 2 to 24 carbon atoms, preferably 5 to 20, more preferably 7 to 15, even more preferably 7, carbon atoms, wherein hydroxy substituted alkenyl radicals are preferred substituted alkenyl radicals, wherein more preferably R 1GL and R 2GL are independently of one another selected from the group consisting of pentenyl, heptenyl, nonenyl, undecenyl, tridecenyl and -(CH2)oCH
- curvy bond in structure (I) implies that the respective substituent may be axial or equatorial, preferably is equatorial.
- Alkyl radicals may be branched or linear.
- Alkenyl radicals may be branched or linear and preferably contain between one to three double bonds.
- residues R 2GL may be identical or different from one another.
- glucolipids are used as biosurfactants SBIO according to the first aspect of the invention.
- glucolipids are used as biosurfactants SBIO according to the first aspect of the invention.
- 0.5 wt.-% to 20 wt.-%, preferably 3 wt.-% to 17 wt.-%, particularly preferably 5 wt.-% to 15 wt-%. of all glucolipids that are used as biosurfactants SBIO according to the first aspect of the invention are GL-C10C12:1 , where the wt.-% refers to the total weight of all the glucolipids that are used as biosurfactants SBIO according to the first aspect of the invention.
- glucolipids are used as biosurfactants SBIO according to the first aspect of the invention.
- 0.5 wt.-% to 20 wt.-%, preferably 2 wt.-% to 15 wt.-%, particularly preferably 3 wt.-% to 12 wt.-%, of all the glucolipids that are used as biosurfactants SBIO according to the first aspect of the invention are GL-C10C12, where the wt.-% refers to the total weight of all the glucolipids that are used as biosurfactants SBIO according to the first aspect of the invention.
- 1 wt.-% to 30 wt.-%, preferably 5 wt.-% to 25 wt.-%, particularly preferably 10 wt.-% to 20 wt.-%, of all the glucolipids that are used as biosurfactants SBIO according to the first aspect of the invention, are GL-C8C10,
- glucolipids that are used as biosurfactants SBIO according to the first aspect of the invention are GL-C10C12:1 ,
- 0.5 wt.-% to 20 wt.-%, preferably 2 wt.-% to 15 wt.-%, particularly preferably 3 wt.-% to 12 wt.-%, of all the glucolipids that are used as biosurfactants SBIO according to the first aspect of the invention, are GL-C10C12, where the wt.-% refers to the total weight of all the glucolipids that are used as biosurfactants SBIO according to the first aspect of the invention.
- glucolipids are used as biosurfactants SBIO according to the first aspect of the invention
- 10 wt.-% to 20 wt.-% of all the glucolipids that are used as biosurfactants SBIO according to the first aspect of the invention are GL-C8C10
- 5 wt.-% to 15 wt.-%, of all the glucolipids that are used as biosurfactants SBIO according to the first aspect of the invention are GL-C10C12:1 ,
- 3 wt.-% to 12 wt.-%, of all the glucolipids that are used as biosurfactants SBIO according to the first aspect of the invention are GL-C10C12, where the wt.-% refers to the total weight of all the glucolipids that are used as biosurfactants SBIO according to the first aspect of the invention.
- glucolipids are used as biosurfactants SBIO according to the first aspect of the invention.
- 0 wt.-% to 5 wt.-% preferably 0.01 wt.-% by weight to 4 wt.-%, particularly preferably 0.1 wt.-% to 3 wt.-%, of all the glucolipids that are used as biosurfactants SBIO according to the first aspect of the invention, are GL-C10, where the wt.-% refers to the total weight of all the glucolipids that are used as biosurfactants SBIO according to the first aspect of the invention.
- rhamnolipid is, in particular, to be understood as referring to compounds of the general formula (III) and salts thereof, preferably compounds according to the general structure (IV) and salts thereof,
- mRL 2, 1 or 0, preferably 1 or 0.
- nRL 1 or 0.
- the glycosidic bond between the two rhamnose units is preferably in the a-configuration.
- the optically active carbon atoms of the fatty acids are preferably present as R-enantiomers (e.g. (R)-3- ⁇ (R)-3-[2-O-(a-L-rhamnopyranosyl)-a-L-rhamnopyranosyl]oxydecanoyl ⁇ oxydecanoate).
- Rhamnolipids applicable in the context of the instant invention can also be produced by fermentation of Pseudomonas, especially Pseudomonas aeruginosa, which are preferably non genetically modified cells, a technology already disclosed in the eighties, as documented e.g. in EP 0282 942 A1 and DE 41 27 908 A1 .
- Rhamnolipids produced in Pseudomonas aeruginosa cells which have been improved for higher rhamnolipid titres by genetical modification can also be used in the context of the instant invention; such cells have for example been disclosed by Lei et al. in Biotechnol Lett. 2020 Jun;42(6):997-1002.
- Rhamnolipids produced by Pseudomonas aeruginosa are commercially available from Jeneil Biotech Inc., e.g. under the tradename Zonix, from Logos Technologies (technology acquired by Stepan), e.g. under the tradename NatSurFact, from Biotensidion GmbH, e.g. under the tradename Rhapynal, from AGAE technologies, e.g. under the name R90, R95, R95Md, R95Dd, from Locus Bio-Energy Solutions and from Shanghai Yusheng Industry Co. Ltd., e.g. under the tradename Bio- 201 Glycolipids.
- rhamnolipids are used as biosurfactants SBIO according to the first aspect of the invention
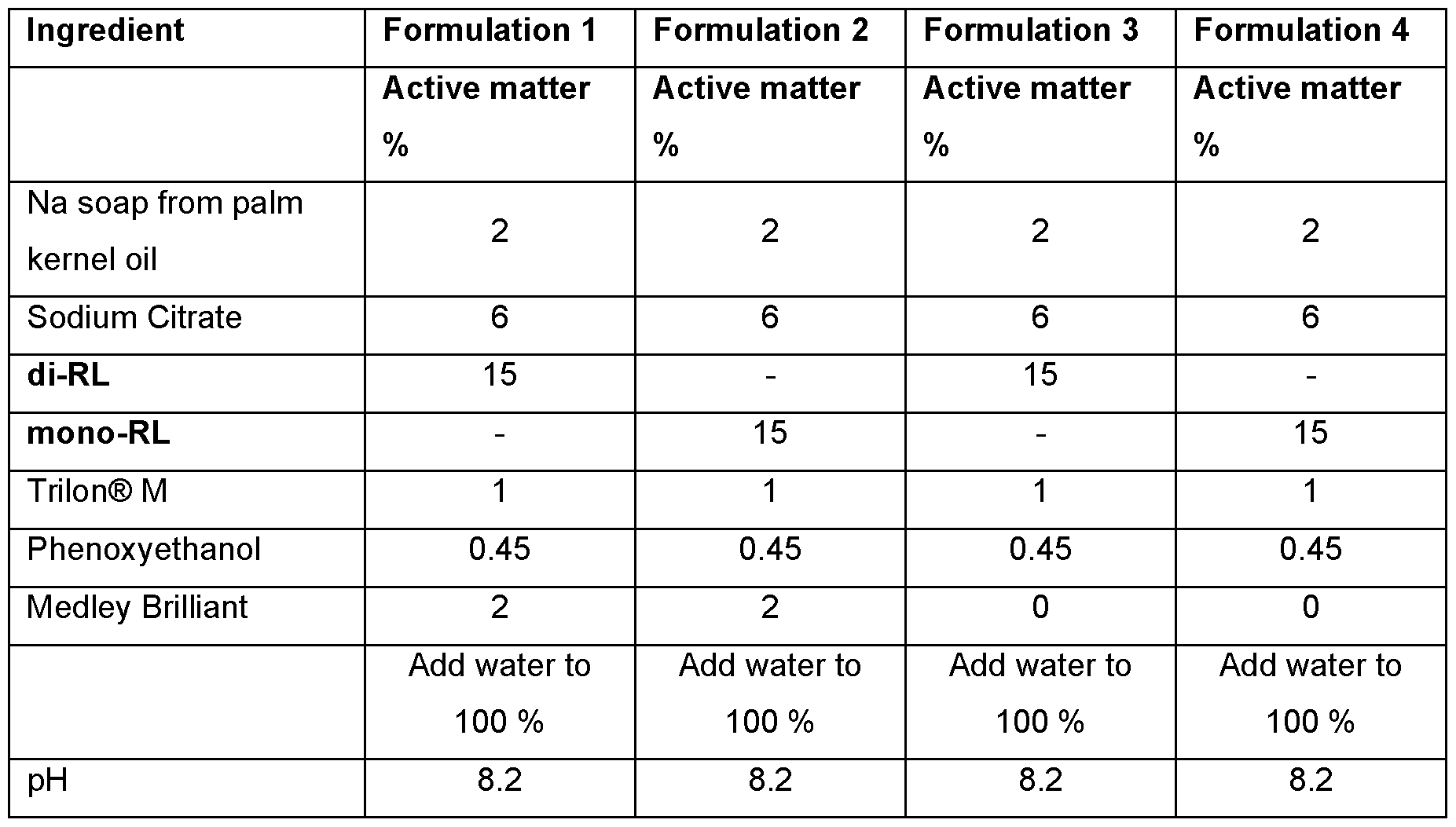
- mixtures of mono-rhamnolipids and dirhamnolipids wherein even more preferably the ratio of the weight of all mono-rhamnolipids used according to the first aspect of the invention to the weight of all di-rhamnolipids used according to the first aspect of the invention is in the range of from 9 : 1 to 1 : 9, preferably of from 8 : 2 to 2 : 8, more preferably of from 7 : 3 to 3 : 7, more preferred of from 6 : 4 to 4 : 6, most preferred is 1 : 1 .
- the rhamnolipids comprise 56 wt.-% to 95 wt.-%, preferably 60 wt.-% to 80 wt.-%, particularly preferably 66 wt.-% to 70 wt.-%, of diRL-C10C10, where the wt.-% refers to the total weight of all the rhamnolipids that are used as biosurfactants SBIO according to the first aspect of the invention.
- rhamnolipids are used as biosurfactants SBIO according to the first aspect of the invention.
- 0.5 wt.-% to 15 wt.-%, preferably 3 wt.-% to 12 wt.-%, particularly preferably 5 wt.-% to 10 wt.-%, of the rhamnolipids are diRL-C10C12:1 , where the wt.-% refers to the total weight of all the rhamnolipids that are used as biosurfactants SBIO according to the first aspect of the invention.
- rhamnolipids are used as biosurfactants SBIO according to the first aspect of the invention.
- 0.5 wt.-% to 25 wt.-%, preferably 3 wt.-% to 15 wt.-%, particularly preferably 5 wt.-% to 12 wt.-%, of the rhamnolipids are diRL-C10C12, where the wt.-% refers to the total weight of all the rhamnolipids that are used as biosurfactants SBIO according to the first aspect of the invention.
- rhamnolipids are used as biosurfactants SBIO according to the first aspect of the invention.
- 0.1 wt.-% to 25 wt.-%, preferably 2 wt.-% to 10 wt.-%, particularly preferably 4 wt.-% to 8 wt.-%, of the rhamnolipids are diRL-C8C10, where the wt.-% refers to the total weight of all the rhamnolipids that are used as biosurfactants SBIO according to the first aspect of the invention.
- rhamnolipids are monoRL-C8C10 and/or, preferably and,
- rhamnolipids are monoRL-C10C10, where the wt.-% refers to the total weight of all the rhamnolipids that are used as biosurfactants SBIO according to the first aspect of the invention.
- rhamnolipids are used as biosurfactants SBIO according to the first aspect of the invention.
- 10 wt.-% to 30 wt.-%, preferably 20 wt.-% to 30 wt.-%, particularly preferably 25 wt.-% to 30 wt.-%, of the rhamnolipids are monoRL-C10C10, where the wt.-% refers to the total weight of all the rhamnolipids that are used as biosurfactants SBIO according to the first aspect of the invention.
- rhamnolipids are used as biosurfactants SBIO according to the first aspect of the invention.
- 10 wt.-% to 30wt.-%, preferably 12 wt.-% to 25 wt.-%, particularly preferably 15 wt.-% to 20 wt.-%, of the rhamnolipids are diRL-C10C10, where the wt.-% refers to the total weight of all the rhamnolipids that are used as biosurfactants SBIO according to the first aspect of the invention.
- rhamnolipids are used as biosurfactants SBIO according to the first aspect of the invention.
- 10 wt.-% to 30 wt.-%, preferably 12 wt.-% to 25 wt.-%, particularly preferably 15 wt.-% to 20 wt.-%, of the rhamnolipids are monoRL-C8C10, where the wt.-% refers to the total weight of all the rhamnolipids that are used as biosurfactants SBIO according to the first aspect of the invention.
- rhamnolipids are used as biosurfactants SBIO according to the first aspect of the invention.
- 3 wt.-% to 25 wt.-%, preferably 5 wt.-% to 20 wt.-%, particularly preferably 10 wt.-% to 15 wt.-%, of the rhamnolipids are monoRL-C10C12:1 , where the wt.-% refers to the total weight of all the rhamnolipids that are used as biosurfactants SBIO according to the first aspect of the invention.
- rhamnolipids are used as biosurfactants SBIO according to the first aspect of the invention.
- 1 wt.-% to 15 wt.-%, preferably 2 wt.-% to 10 wt.-%, particularly preferably 3 wt.-% to 8 wt.-%, of the rhamnolipids are diRL-C10C12, where the wt.-% refers to the total weight of all the rhamnolipids that are used as biosurfactants SBIO according to the first aspect of the invention.
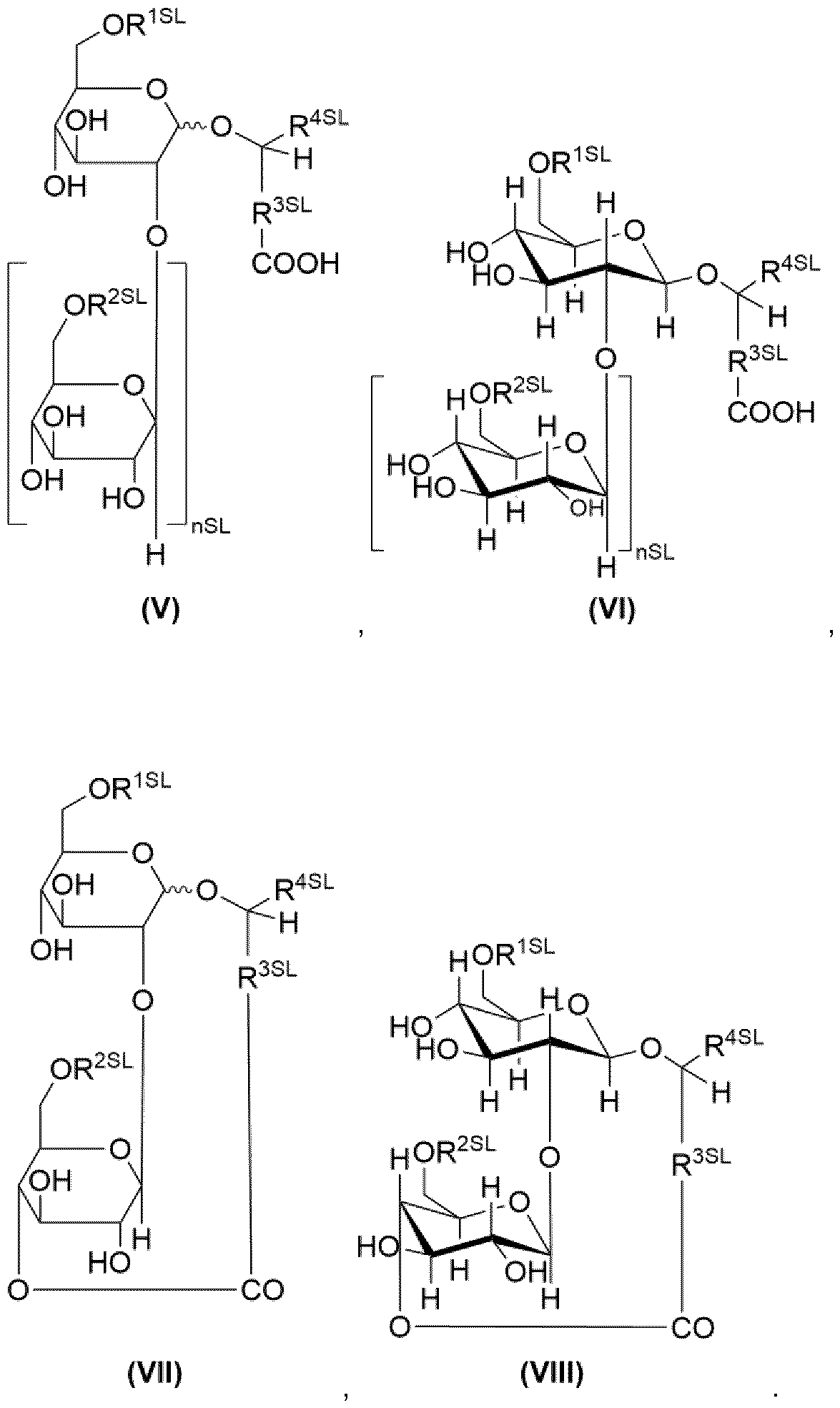
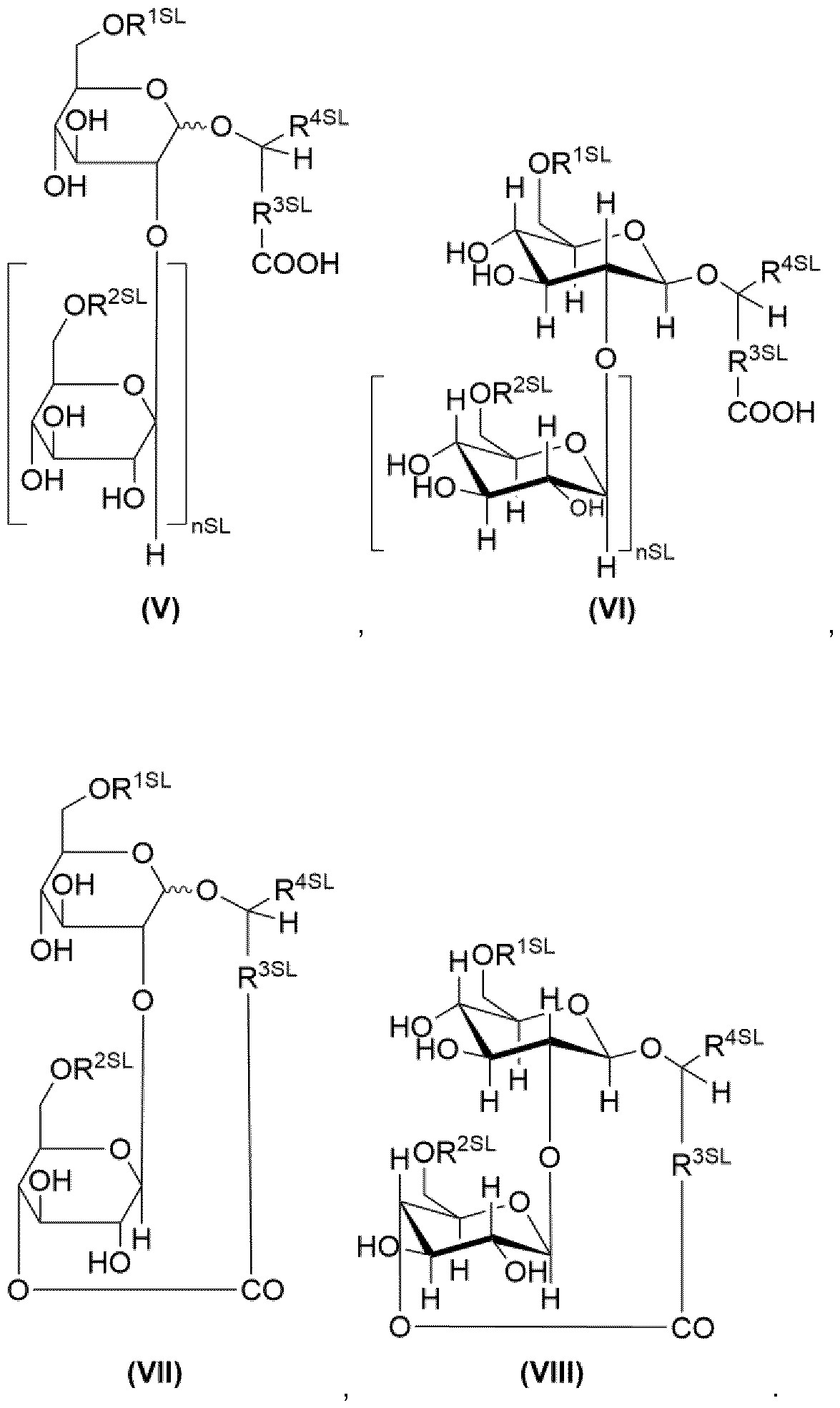
- the term “sophorolipids” is in particular to be understood as referring to compounds of the general formulae (V), (VII) and salts thereof, preferably compounds of the general formulae (VI), (VIII) and salts thereof: Formulae (V), (VII) represent the acid form, Formulae (VI), (VIII) represent the lactone form.
- nSL 1 or 0.
- R 1SL H or -CO-CH 3 ,
- R2SL H or -CO-CH 3 ,
- R 3SL a divalent organic moiety which comprises 6 to 32 carbon atoms, preferably 12 to 20, more preferably 14 to 16, most preferably 15.
- R 3 SI_ preferably is an optionally substituted, divalent hydrocarbon moiety comprising 6 to 32 carbon atoms, wherein hydroxy substituted hydrocarbon moieties are preferred as substituted hydrocarbon moi eties,
- R 3 SI_ more preferably is selected from the group consisting of
- optionally substituted alkylene radicals with 6 to 32, preferably 12 to 20, more preferably 14 to 16, most preferably 15 carbon atoms, wherein hydroxy-substituted alkylene radicals are preferred substituted alkylene radicals, and wherein it is preferred that the optionally substituted alkylene radicals are unbranched,
- optionally substituted alkenylene radicals with 6 to 32, preferably 12 to 20, more preferably 14 to 16, most preferably 15 carbon atoms, wherein hydroxy-substituted alkenylene radicals are preferred substituted alkenylene radicals, and wherein it is preferred that the optionally substituted alkenylene radicals are unbranched, and wherein it is preferred that the optionally substituted alkenylene radicals comprise one to three double or triple bonds.
- R 3SL even more preferably is selected from the group consisting of
- alkenylene radicals with 6 to 32, preferably 12 to 20, more preferably 14 to 16, most preferably 15 carbon atoms, wherein the alkenylene radicals preferably comprise one to three double or triple bonds, wherein the alkenylene radicals more preferably comprise one to three double bonds, even more preferably one double bond,
- alkenylene radicals with 6 to 32, preferably 12 to 20, more preferably 14 to 16, most preferably 15, carbon atoms carrying at least one hydroxy group, preferably one hydroxy group, wherein the alkenylene radicals preferably comprise one to three double or triple bonds, wherein the alkenylene radicals more preferably comprise one to three double bonds, even more preferably one double bond.
- R 4SL H, CH3 or a monovalent organic radical which comprises 2 to 10 carbon atoms.
- R 4SL is preferably selected from the group consisting of
- optionally substituted alkyl radicals with 2 to 10 carbon atoms, wherein hydroxy-substituted alkyl radicals are preferred substituted alkyl radicals, and wherein it is preferred that the optionally substituted alkyl radicals are unbranched,
- optionally substituted alkenyl radicals with 2 to 10 carbon atoms, wherein hydroxysubstituted alkenyl radicals are preferred substituted alkenyl radicals, and wherein it is preferred that the optionally substituted alkenyl radicals are unbranched, and wherein it is preferred that the optionally substituted alkenyl radicals comprise one to three double or triple bonds.
- R 4SL is more preferably selected from the group consisting of
- alkenyl radicals with 2 to 10 carbon atoms, wherein the alkenyl radicals preferably comprise one to three double or triple bonds, wherein the alkenyl radicals more preferably comprise one to three double bonds, even more preferably one double bond,
- alkenyl radicals with 2 to 10 carbon atoms carrying at least one hydroxy group, preferably one hydroxy group, wherein the alkenyl radicals preferably comprise one to three double or triple bonds, wherein the alkenyl radicals more preferably comprise one to three double bonds, even more preferably one double bond.
- R 4SL is most preferably selected from the group consisting of H, Methyl, Ethyl.
- the term “sophorolipids” is to be understood as referring to compounds of the general formulae (IX), (XI) and salts thereof, preferably compounds of the general formulae (X), (XII) and salts thereof:
- Formulae (IX), (X) represent the acid form
- Formulae (XI), (XII) represent the lactone form.
- nSL, R 1SL , R 2SL , R 4SL have the same meaning as described for formulae (V), (VII), (VI), (VIII).
- Sophorolipids may be used in accordance with the invention in their acid form or their lactone form.
- Preferred compositions in the context of the use according to the first aspect of the present invention comprise a sophorolipid in which the ratio by weight of lactone form to acid form is in the range of 20:80 to 80:20, especially preferably in the ranges of 30:70 to 40:60.
- Sophorolipids may be obtained as described in EP 1 411 111 A1 , paragraphs [0021], [0022].
- the at least on biosurfactant SBIO is comprised in an aqueous mixture L.
- Aw is contacted with the aqueous mixture L.
- the aqueous mixture L is hence, in particular, the washing solution in which the at least one article Aw is washed.
- the woolen article Aw is contacted with the aqueous mixture L comprising at least one biosurfactant SBIO” is to be understood that, in the context of the use according the first aspect of the invention, the woolen article Aw is brought in direct contact with the aqueous mixture L comprising at least one biosurfactant SBIO, “in direct contact” meaning “wetting” of the woolen article Aw with the aqueous mixture L.
- the article Aw is at least partially immersed, even more preferably fully immersed into the aqueous mixture L.
- the article Aw is moved in the aqueous mixture L during the use according to the first aspect of the invention.
- the mixture L is preferably brought in direct contact with at least a part of the surface of article Aw, onto which at least one impurity I is absorbed or adsorbed.
- a mixture L is obtained wherein in the water comprised by aqueous mixture L, at least one impurity I in dissolved or dispersed, preferably dispersed.
- the concentration of impurities I in the water comprised by aqueous mixture L therefore augments during the use according to the first aspect of the invention.
- the aqueous mixture L that is employed during the use according to the first aspect of the invention i.e. before it contacts the at least one woolen article Aw
- the mixture L that is obtained during the use according to the first aspect of the invention is a dispersion, in which the at least one impurity I is the dispersed phase in the water W comprised by aqueous mixture L.
- the impurity I may either be in the form of a liquid (e.g. droplet) or a particle, and preferably is a particle.
- the impurity I is at least one impurity which may be inorganic or organic, in particular inorganic, preferably clay, more preferably kaolin.
- Organic impurities are preferably selected from oils and fats, which may be of vegetable, animal or synthetic origin, food based particles, soil, pollen, sebum, body fluids, for example, blood, sperm, sweat, urine, feces, liquor.
- Inorganic impurities are preferably selected from the group consisting of carbon black, fine dust, plastic particles (e.g. microplastic particles), metal oxides (preferably iron oxide), silica (such as sand), clay, dyes (such as pigments), more preferably clay, in particular kaolin.
- the beneficial use according to the first aspect of the invention is the reduction of shrinking of the at least one article Aw during washing in the aqueous mixture L comprising the at least one biosurfactant SBIO- Since the tendency of woolen articles to shrink is especially pronounced at temperatures above 45 °C, the best effect of the present invention is obtained when the aqueous mixture L has a temperature at or below 45 °C for at least as long as the aqueous mixture L contacts the at least one woolen article Aw-
- the temperature of the aqueous mixture L, for at least as long as the aqueous mixture L is contacted with the at least one woolen article Aw is in a range of from > 0 °C to 45 °C, in particular in a range of from 1 °C to 40 °C, preferably in a range of from 5 °C to 35 °C, more preferably in a range of from 10 °C to 30 °C, more preferably in a range of from 15 °C to 30 °C, even more preferably in a range of from 20 °C to 30 °C, even more preferably in a range of from 25 °C to 30 °C.
- the aqueous mixture L comprises the at least one biosurfactant SBIO in an amount in the range of from 0.000001 wt.-% to 50 wt.-%, preferably in the range of from 0.00001 wt.-% to 30 wt.-%, more preferably in the range of from 0.00005 wt.-% to 20 wt.-%, more preferably in the range of from 0.00009 wt.-% ppm to 15 wt.-%, more preferably in the range of from 0.0001 wt.-% to 14 wt.-%, more preferably in the range of from 0.001 wt.-% to 13 wt.-%, more preferably in the range of from 0.002 wt.-% to 12 wt.-%, more preferably in the range of from 0.005 wt.-% to 11 wt.-%, more preferably in the range of from 0.01 wt.-% to 10 wt.-%, more
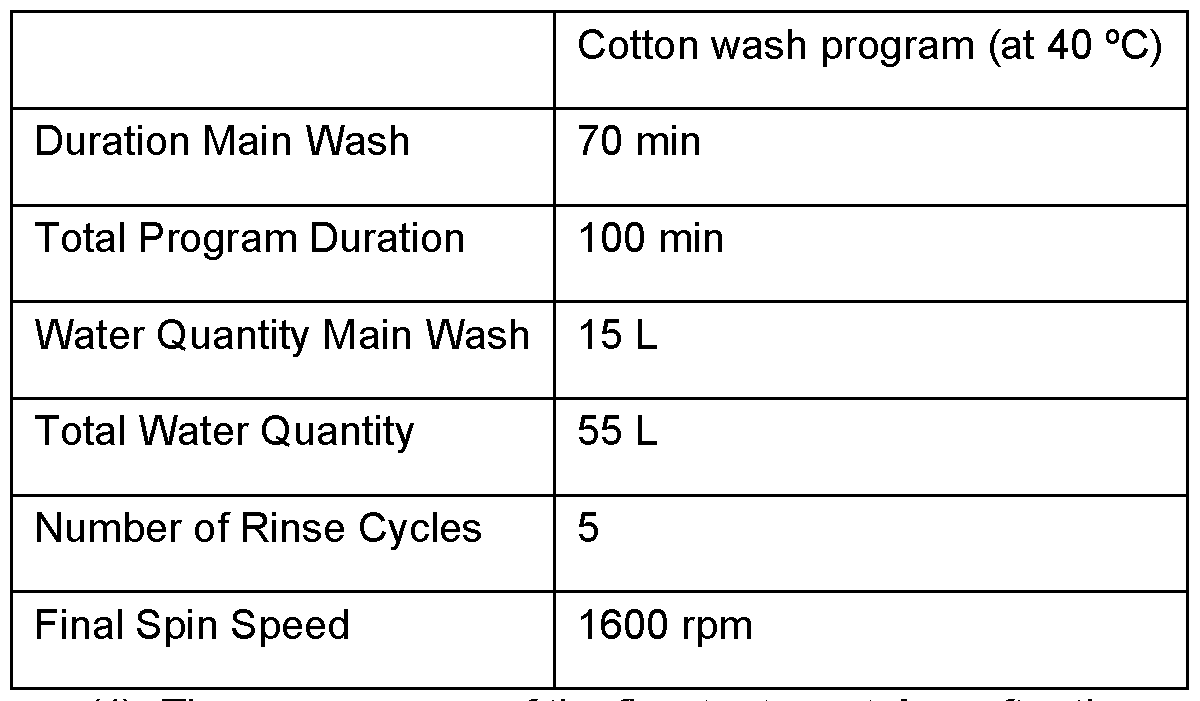
- Aw is contacted with the aqueous mixture L for at least 1 s, preferably at least 10 s, more preferably at least 1 min, even more preferably at least 5 min, preferably for 15 min to 240 min, even more preferably for 30 min to 180 min, even more preferably for 45 min to 120 min, even more preferably for 60 min to 120 min, even more preferably for 70 min to 100 min.
- the woolen article Aw is preferably moved in the aqueous mixture. This improves the washing efficiency.
- the washing in the context of the use according to the first aspect of the invention is preferably carried out in a washing machine.
- the at least one biosurfactant SBIO in the use according to the first aspect of the invention may be present at least partially as salts, in particular when it is comprised by the aqueous mixture L.
- the ratio of biosurfactants SBIO that are present as salts depends on the pH of the water in the aqueous mixture L.
- biosurfactants SBIO are salts in which the cation is selected from the group consisting of Li + , Na + , K + , Mg 2+ , Ca 2+ , Al 3+ , NH4 + , ammonium ions, wherein the ammonium ions may be primary, secondary, tertiary or quaternary.
- Particularly preferred cations are selected from the group comprising, preferably consisting of, Na + , K + , NH4 + and the triethanolammonium cation.
- ammonium ions are tetramethylammonium, tetraethylammonium, tetrapropylammonium, tetrabutylammonium, [(2-hydroxyethyl)trimethylammonium] (choline) and also the cations of 2-aminoethanol (ethanolamine, “MEA”), diethanolamine (“DEA”), 2,2',2"-nitrilotriethanol (triethanolamine, “TEA”), 1-aminopropan-2-ol (monoisopropanolamine), ethylenediamine, diethylenetriamine, triethylenetetramine, tetraethylenepentamine, 1 ,4-diethylenediamine (piperazine), aminoethylpiperazine and aminoethylethanolamine.
- mixtures of the abovementioned cations may also be present as cations of the salts of the at least one biosurfactant SBIO, if present as salts, according to the invention.
- aqueous mixture L may comprise further ingredients, preferably at least one ingredient selected from surfactants (wherein the surfactants are different from SBIO), enzymes, builders, solvents, preservatives, benefit agents, polymers.
- surfactants wherein the surfactants are different from SBIO
- enzymes preferably at least one ingredient selected from enzymes, builders, solvents, preservatives, benefit agents, polymers.
- aqueous mixture L in the context of the use according to the first aspect of the present invention, comprises, besides the at least one biosurfactant SBIO, at least one further surfactant SY, wherein the surfactant SY is different from SBIO-
- the at least one further surfactant SY may be selected from anionic surfactants, nonionic surfactants, cationic surfactants, amphoteric surfactants, and in particular is selected from anionic surfactants, nonionic surfactants.
- the aqueous mixture L in the context of the use according to the first aspect of the present invention comprises at least one further surfactant SY that is different from the biosurfactants SBIO
- the ratio of weights of all biosurfactants SBIO in the mixture L to the weight of all surfactants SY is in the range of from 99 : 1 to 1 : 99, preferably 90 : 10 to 10 : 90, more preferably 80 : 20 to 20 : 80, even more preferably 70 : 30 to 30 : 70, even more preferably 60 : 40 to 40 : 60, eve more preferably 50 : 50.
- Aqueous mixture L in the context of the use according to the first aspect of the present invention optionally comprises at least one nonionic surfactant.
- the nonionic surfactants are preferably selected from the group consisting of alkoxylated alcohols, alkoxylated fatty acid alkyl esters, alkyl glycosides, amine oxides, polyhdroxy fatty acid amides.
- the nonionic surfactants are more preferably selected from the group consisting of alkoxylated alcohols, alkoxylated fatty acid alkyl esters, alkyl glycosides.
- the at least one nonionic surfactant is selected from the group consisting of alcohol ethoxylates (“AE” or “AEO”), alcohol propoxylates, propoxylated fatty alcohols (“PFA”), alkoxylated fatty acid alkyl esters, such as ethoxylated and/or propoxylated fatty acid alkyl esters, alkylphenol ethoxylates (“APE”), nonylphenol ethoxylates (“NPE”), alkylpolyglycosides (“APG”), alkoxylated amines, fatty acid monoethanolamides (“FAM”), fatty acid diethanolamides (“FADA”), ethoxylated fatty acid monoethanolamides (“EFAM”), polyglycerol esters, glaycerol esters, propoxylated fatty acid monoethanolamides (“PFAM”), polyhydroxy alkyl fatty acid amides, or A/-acyl A/-alkyl derivatives of gluco
- Alkoxylated alcohols in the context of the present invention are preferably ethoxylated alcohols.
- alkoxylated alcohols are primary alcohols having preferably 8 to 18 carbon atoms and on average 1 to 12 mol of ethylene oxide (“EO”) per mol of alcohol, in which the alcohol radical can be linear or branched, preferably 2-position methyl-branched, or can contain linear and methyl- branched radicals in a mixture, as are customarily present in oxo alcohol radicals.
- EO ethylene oxide
- alcohol ethoxylates with linear radicals from alcohols of native origin having 12 to 18 carbon atoms, for example from coconut, palm, tallow fat or oleyl alcohol, and on average 2 to 8 EO per mol of alcohol are preferred.
- the preferred ethoxylated alcohols include, for example, Ci2-Ci4-alcohols with 3 EO, 4 EO or 7 EO, Cg-Cn-alcohol with 7 EO, Ci3-Ci5-alcohols with 3 EO, 5 EO, 7 EO or 8 EO, Ci2-Cia-alcohols with 3 EO, 5 EO or 7 EO and mixtures of these, such as mixtures of Ci2-Ci4-alcohol with 3 EO and Ci2-Cia-alcohol with 7 EO.
- the stated degrees of ethoxylation are statistical average values which can be an integer or a fraction for a specific product.
- Preferred alcohol ethoxylates have a narrow homolog distribution.
- Alkoxylated alcohols may also be selected from fatty alcohols with more than 12 EO. Examples thereof are tallow fatty alcohol with 14 EO, 25 EO, 30 EO or 40 EO. Nonionic surfactants which contain EO and propylene oxide (“PO”) groups together in the molecule can also be used. In this connection, it is possible to use block copolymers with EO-PO block units or PO-EO block units, but also EO-PO-EO copolymers or PO-EO-PO copolymers.
- alkyl glycosides can also be used as further nonionic surfactants.
- a further class of preferably used nonionic surfactants are alkoxylated, preferably ethoxylated or ethoxylated and propoxylated, fatty acid alkyl esters, preferably having 1 to 4 carbon atoms in the alkyl chain, in particular fatty acid methyl esters, as are described for example in JP S58-217598 A or which are preferably prepared by the process described in WO 90/13533 A1 .
- Nonionic surfactants of the amine oxide type for example A/-cocoalkyl-A/,A/-dimethylamine oxide and A/-tallowalkyl-A/,A/-dihydroxyethylamine oxide, and of the fatty acid alkanolamide type may also be suitable.
- the amount of these nonionic surfactants is preferably not more than that of the ethoxylated fatty alcohols, in particular not more than half thereof.
- polyhydroxy fatty acid amides are substances which can usually be obtained by reductive amination of a reducing sugar with ammonia, an alkylamine or an alkanolamine and subsequent acylation with a fatty acid, a fatty acid alkyl ester or a fatty acid chloride.
- Aqueous mixture L in the context of the use according to the first aspect of the present invention optionally comprises at least one anionic surfactant.
- the anionic surfactants are preferably selected from the group consisting of anionic surfactants of the sulfonate and anionic surfactants of the sulfate type.
- Preferred surfactants of the sulfonate type are selected from Cg-Cn-alkylbenzenesulfonates, olefinsulfonates, i.e. mixtures of alkene- and hydroxyalkanesulfonates, and also disulfonates, as are obtained, in particular, from Ci2-Ci8-monoolefins with a terminal or internal double bond by sulfonation with gaseous sulfur trioxide and subsequent alkaline or acidic hydrolysis of the sulfonation products.
- alkanesulfonates which are preferably obtained from Ci2-Cia-alkanes, for example by sulfochlorination or sulfoxidation with subsequent hydrolysis or neutralization.
- esters of a-sulfo fatty acids are also preferred, for example the a-sulfonated methyl esters of hydrogenated coconut, palm kernel or tallow fatty acids.
- Preferred surfactants of the sulfate type are selected from sulphated fatty acid glycerol esters.
- Fatty acid glycerol esters are to be understood as meaning the mono-, di- and triesters, and also mixtures thereof, as are obtained, in particular, in the preparation by esterification of a monoglycerol with 1 to 3 mol of fatty acid or in the transesterification of triglycerides with 0.3 to 2 mol of glycerol.
- Preferred sulfated fatty acid glycerol esters here are the sulfation products of saturated fatty acids having 6 to 22 carbon atoms, for example of caproic acid, caprylic acid, capric acid, myristic acid, lauric acid, palmitic acid, stearic acid or behenic acid.
- alkyl sulfates and alkenyl sulfates which are preferably the alkali metal, in particular the sodium, salts of the sulphuric acid half-esters of the Ci2-Cis-fatty alcohols, for example from coconut fatty alcohol, tallow fatty alcohol, lauryl, myristyl, cetyl or stearyl alcohol or the C10-C20-OXO alcohols and those half-esters of secondary alcohols of these chain lengths.
- alkyl sulfates and alkenyl sulfates of the specified chain length which contain a synthetic straight-chain alkyl radical prepared on a petrochemical basis, and which have an analogous degradation behaviour to the suitable compounds based on fatty chemical raw materials.
- the Ci2-Ci6-alkyl sulfates and Ci2-Cis-alkyl sulfates and also Ci4-Cis-alkyl sulfates are preferred.
- 2,3-Alky I sulfates which are prepared for example in accordance with the US 3,234,258 A or US 5,075,041 A and can be obtained as commercial products of the Shell Oil Company under the name DAN®, are also suitable anionic surfactants.
- EO ethylene oxide
- Ci2-Cis-fatty alcohols with 1 to 4 EO are also preferred.
- they are used in cleaning compositions only in relatively small amounts, for example in amounts of from 1 to 5 % by weight.
- alkylsulfosuccinic acid which are also referred to as sulfosuccinates or as sulfosuccinic acid esters and constitute the monoesters and/or diesters of sulfosuccinic acid with alcohols, preferably fatty alcohols and in particular ethoxylated fatty alcohols.
- Preferred sulfosuccinates contain Cs-Cis-fatty alcohol radicals or mixtures of these.
- Particularly preferred sulfosuccinates contain a fatty alcohol radical which is derived from ethoxylated fatty alcohols.
- sulfosuccinates whose fatty alcohol radicals are derived from ethoxylated fatty alcohols with a narrow homolog distribution are particularly preferred in turn. It is likewise also possible to use alkylsuccinic acid and alkenylsuccinic having preferably 8 to 18 carbon atoms in the alkyl chain/ alkenyl chain or salts thereof.
- Particularly preferred anionic surfactants are soaps. Also of suitability are saturated and unsaturated fatty acid soaps, such as the salts of lauric acid, myristic acid, palmitic acid, stearic acid, (hydrogenated) erucic acid and behenic acid, and also soap mixtures derived in particular from natural fatty acids, for example coconut, palm kernel, olive oil or tallow fatty acid.
- saturated and unsaturated fatty acid soaps such as the salts of lauric acid, myristic acid, palmitic acid, stearic acid, (hydrogenated) erucic acid and behenic acid, and also soap mixtures derived in particular from natural fatty acids, for example coconut, palm kernel, olive oil or tallow fatty acid.
- the anionic surfactants including the soaps can be in the form of their sodium, potassium or ammonium salts, as well as soluble salts of organic bases, such as mono-, di- or triethanolamine.
- the anionic surfactants are in the form of their sodium or potassium salts, in particular in the form of the sodium salts.
- Anionic surfactants preferably comprised by aqueous mixture L are selected from the group consisting of anionic surfactants of the sulfonate and anionic surfactants of the sulfate type, even more preferably selected from the group consisting of linear alkylbenzenesulfonates (“LAS”), isomers of LAS, branched alkylbenzenesulfonates (“BABS”), phenylalkanesulfonates, a-olefinsulfonates (“AOS”), olefin sulfonates, alkene sulfonates, alkane-2,3-diylbis(sulfates), hydroxyalkanesulfonates and disulfonates, alkyl sulfates (“AS”) such as sodium dodecyl sulfate (“SDS”), fatty alcohol sulfates (“FAS”), primary alcohol sulfates (“PAS”), alcohol ethersulfates (“A
- Aqueous mixture L in the context of the use according to the first aspect of the present invention optionally comprises at least one amphoteric surfactant, which preferably is a compound which carries at least one quaternary ammonium group and at least one -COO - or -SOs- group in the molecule, more preferably is selected from the group consisting of betaine, alkyldimethylbetaine, sulfobetaine.
- amphoteric surfactant which preferably is a compound which carries at least one quaternary ammonium group and at least one -COO - or -SOs- group in the molecule, more preferably is selected from the group consisting of betaine, alkyldimethylbetaine, sulfobetaine.
- betaine surfactants are alkyl- or alkylamidopropylbetaines.
- betaines such as the A/-alkyl-A/,A/-dimethylammonium glycinates, e.g. the cocoalkyldimethylammonium glycinate, A/-acylaminopropyl-A/,A/-dimethylammonium glycinates, e.g.
- cocoacylaminopropyldimethylammonium glycinate the Ci2-Ci8-alkyldimethylacetobetaine, the cocoamidopropyldimethylacetobetaine, 2-alkyl-3-carboxymethyl-3-hydroxyethylimidazolines and sulphobetaines having in each case 8 to 18 carbon atoms in the alkyl or acyl group, and also the cocoacylaminoethylhydroxyethylcarboxymethyl glycinate are preferred.
- a particularly preferred zwitterionic surfactant is the A/,A/-dimethyl-A/-(lauroylamidopropyl)ammoniumacetobetaine known under the INCI name Cocamidopropyl Betaine.
- Further suitable amphoteric surfactants are formed by the group of amphoacetates and amphodiacetates, in particular, for example, coco- or laurylamphoacetates or -diacetates, the group of amphopropionates and amphodipropionates, and the group of amino acid-based surfactants such as acyl glutamates, in particular disodium cocoyl glutamate and sodium cocoyl glutamate, acyl glycinates, in particular cocoyl glycinates, and acyl sarcosinates, in particular ammonium lauroyl sarcosinate and sodium cocoyl sarcosinate.
- Aqueous mixture L in the context of the use according to the first aspect of the present invention optionally comprises at least one cationic surfactant, which are preferably selected from the group consisting of alklydimethylethanolamine quat (“ADMEAQ”), cetyltrimethylammonium bromide (“CTAB”), dimethyldistearylammonium chloride (“DSDMAC”), and alkylbenzyldimethylammonium, alkyl quaternary ammonium compounds, alkoxylated quaternary ammonium (“AQA”) compounds.
- ADMEAQ alklydimethylethanolamine quat
- CTAB cetyltrimethylammonium bromide
- DMDMAC dimethyldistearylammonium chloride
- AQA alkylbenzyldimethylammonium
- AQA alkoxylated quaternary ammonium
- Non-limiting examples of non-ionic surfactants include alcohol ethoxylates (“AE” or “AEO”), alcohol propoxylates, propoxylated fatty alcohols (“PFA”), alkoxylated fatty acid alkyl esters, such as ethoxylated and/or propoxylated fatty acid alkyl esters, alkylphenol ethoxylates (“APE”), nonylphenol ethoxylates (“NPE”), alkylpolyglycosides (“APG”), alkoxylated amines, fatty acid monoethanolamides (“FAM”), fatty acid diethanolamides (“FADA”), ethoxylated fatty acid monoethanolamides (“EFAM”), polyglycerol esters, glaycerol esters, propoxylated fatty acid monoethanolamides (“PFAM”), polyhydroxy alkyl fatty acid amides, or A/-acyl A/-alkyl derivatives of glucosamine (glucamides, “GA”,
- the aqueous mixture L in the context of the use according to the first aspect of the invention may also comprise at least one enzyme E.
- the aqueous mixture L in the context of the use according to the first aspect of the invention comprises at least one enzyme E
- the enzyme E is selected from the group consisting of protease, amylase, cellulase, mannanase, lipase, cutinase, pectate lyase, peroxidase, oxidase, laccase, even more preferably selected from the group consisting of cellulase, protease, lipase, cutinase, amylase, peroxidase, oxidase, mannanase. It is even more preferred that the at least one enzyme E is selected from the group consisting of protease, amylase, lipase, mannanase. Most preferred, the at least one enzyme E is a lipase.
- the aqueous mixture L in the context of the use according to the first aspect of the invention comprises at least one enzyme E
- the amount of all enzymes E in the aqueous mixture L is in the range of 0.01 ppm to 1 wt.-%, preferably 0.01 ppm to 1000 ppm, more preferably 0.1 to 100 ppm, preferably 1 ppm to 10 ppm per the total weight of aqueous mixture L.
- Suitable cellulases include those of bacterial or fungal origin. Chemically modified or protein engineered mutants are included. Suitable cellulases include cellulases from the genera Bacillus, Pseudomonas, Humicola, Fusarium, Thielavia, Acremonium , e.g., the fungal cellulases produced from Humicola insolens, Myceliophthora thermophila and Fusarium oxysporum disclosed in US 4,435,307 A, US 5,648,263 A, US 5,691 ,178 A, US 5,776,757 A and WO 89/09259 A1 . Especially suitable cellulases are the alkaline or neutral cellulases having color care benefits.
- cellulases examples include cellulases described in EP 0 495 257 A1 , WO 91/17243 A1 , WO 96/11262 A1 , WO 96/29397 A1 , WO 98/08940 A1 .
- cellulase variants such as those described in WO 94/07998 A1 , WO 91/17244 A1 , US 5,457,046 A, US 5,686,593 A, US 5,763,254 A, WO 95/24471 A1 , WO 98/12307 A1 and WO 99/01544 A1.
- Example of cellulases exhibiting endo-beta-1 ,4-glucanase activity are those having described in WO 02/099091 A2.
- cellulases include CelluzymeTM, and CarezymeTM (Novozymes A/S), ClazinaseTM, and Puradax HATM (Genencor International Inc.), and KAC-500(B)TM (Kao Corporation).
- Suitable proteases include those of bacterial, fungal, plant, viral or animal origin e.g. vegetable or microbial origin. Microbial origin is preferred. Chemically modified or protein engineered mutants are included. It may be an alkaline protease, such as a serine protease or a metalloprotease. A serine protease may for example be of the S1 family, such as trypsin, or the S8 family such as subtilisin. A metalloproteases protease may for example be a thermolysin from e.g. family M4 or other metalloprotease such as those from M5, M7 or M8 families.
- subtilases refers to a sub-group of serine protease according to R.J. Siezen, W.M. de Vos, J.A.M. Leunissen, B.W. Dijkstra, Protein Engineering, Design and Selection 1991 , 4, 719-737 and R.J. Siezen & J.A. Leunissen, Protein Science 1997, 6, 501-523.
- Serine proteases are a subgroup of proteases characterized by having a serine in the active site, which forms a covalent adduct with the substrate.
- the subtilases may be divided into 6 sub-divisions, i.e. the Subtilisin family, the Thermitase family, the Proteinase K family, the Lantibiotic peptidase family, the Kexin family and the Pyrolysin family.
- subtilases are those derived from Bacillus such as Bacillus lentus, B. alkalophilus, B. subtilis, B. amyloliquefaciens, Bacillus pumilus and Bacillus gibsonii described in: US 7,262,042 B2 and WO 2009/021867 A2, and subtilisin lentus , subtilisin Novo, subtilisin Carlsberg, Bacillus licheniformis, subtilisin BPN', subtilisin 309, subtilisin 147 and subtilisin 168 described in
- Other useful proteases may be those described in WO 2019/105675 A1 , WO 01/016285 A2, and WO 02/016547 A2.
- trypsin-like proteases are trypsin (e.g. of porcine or bovine origin) and the Fusarium protease described in WO 89/06270 A1 , WO 94/25583 A1 and WO 2005/040372 A1 , and the chymotrypsin proteases derived from Cellumonas described in WO 2005/052161 A2 and WO 2005/052146 A2.
- a further preferred protease is the alkaline protease from Bacillus lentus DSM 5483, as described for example in WO 95/23221 A1 , and variants thereof which are described in WO 92/21760 A2, WO 95/23221 A1 , EP 1 921 147 A2 and EP 1 921 148 A2.
- metalloproteases are the neutral metalloprotease as described in
- WO 2007/044993 A2 (Genencor I nt.) such as those derived from Bacillus amyloliquefaciens.
- useful proteases are the variants described in: WO 92/19729 A1 , WO 96/34946 A1 , WO 98/20115 A1 , WO 98/20116 A1 , WO 99/11768 A1 , WO 01/44452 A1 , WO 03/006602 A2, WO 2004/03186 A2, WO 2004/041979 A2, WO 2007/006305 A1 , WO 2011/036263 A1 , WO 2011/036264 A1 .
- Suitable commercially available protease enzymes include those sold under the trade names Alcalase®, DuralaseTM, DurazymTM, Relase®, Relase® Ultra, Savinase®, Savinase® Ultra, Primase, Polarzyme®, Kannase®, Liquanase®, Liquanase® Ultra, Liquanase® 2.5 L, Ovozyme®, Coronase, Coronase® Ultra, Neutrase®, Everlase® and Esperase® (Novozymes A/S), those sold under the tradename Maxatase®, Maxacai®, Maxapem®, Purafect®, Purafect Prime®, Eraser®, Purafect MA®, Purafect Ox®, Purafect OxP®, Puramax®, Properase®, Ultimase®, FN2®, FN3®, FN4®, Excellase®, , Opticlean® and Optimase® (Danisco/DuPont),
- a protease preferably comprised in the composition according to the instant invention is Liquanase® 2.5 L.
- Suitable lipases and cutinases include those of bacterial or fungal origin. Chemically modified or protein engineered mutant enzymes are included. Examples include lipase from Thermomyces , e.g. from T. lanuginosus (previously named Humicola lanuginosa) as described in EP 0 258 068 A2 and EP 0 305 216 A1 , cutinase from Humicola, e.g. H. insolens (WO 96/13580 A1), lipase from strains of Pseudomonas (some of these now renamed to Burkholderia), e.g. P. alcaligenes or P.
- Thermomyces e.g. from T. lanuginosus (previously named Humicola lanuginosa) as described in EP 0 258 068 A2 and EP 0 305 216 A1
- cutinase from Humicola e.g. H. insol
- GDSL-type Streptomyces lipases (WO 2010/065455 A2), cutinase from Magnaporthe grisea (WO 2010/107560 A2), cutinase from Pseudomonas mendocina (US 5,389,536 A), lipase from Thermobifida fusca (WO 2011/084412 A1), Geobacillus stearothermophilus lipase (WO 2011/084417 A1), lipase from Bacillus subtilis (WO 2011/084599 A1), and lipase from Streptomyces griseus (WO 2011/150157 A2) and S. pristinaespiralis (WO 2012/137147 A1).
- lipase variants such as those described in EP 0 407 225 A1 , WO 92/05249 A1 , WO 94/01541 A1 , WO 94/25578 A1 , WO 95/14783 A1 , WO 95/30744 A2, WO 95/35381 A1 , WO 95/22615 A1 , WO 96/00292 A1 , WO 97/04079 A1 , WO 97/07202 A1 , WO 00/34450 A1 , WO 00/60063 A1 , WO 01/92502 A1 , WO 2007/87508 A2, ON 104031899 A and WO 2009/109500 A1.
- Preferred commercial lipase products include LipolaseTM, LipexTM, LipolexTM, LipexTM 100 L Evity and LipocleanTM (Novozymes A/S), Lumafast (originally from Genencor) and Lipomax (originally from Gist-Brocades).
- a lipase preferably comprised in the composition according to the instant invention is LipexTM 100 L Evity.
- lipases sometimes referred to as acyltransferases or perhydrolases, e.g. acyltransferases with homology to Candida antarctica lipase A (WO 2010/111143 A2), acyltransferase from Mycobacterium smegmatis (WO 2005/56782 A2), perhydrolases from the CE 7 family (WO 2009/67279 A1), and variants of the Mycobacterium smegmatis perhydrolase in particular the S54V variant used in the commercial product Gentle Power Bleach from Huntsman Textile Effects Pte Ltd (WO 2010/100028 A2).
- Suitable amylases which can be used herein may be an a-amylase or a glucoamylase and may be of bacterial or fungal origin. Chemically modified or protein engineered mutants are included.
- Amylases include, for example, a-amylases obtained from Bacillus , e.g., a special strain of Bacillus licheniformis , described in more detail in GB 1 ,296,839.
- Suitable amylases include amylases having SEQ ID NO: 3 in WO 95/10603 A1 or variants having about 90 % sequence identity to SEQ ID NO: 3 thereof. Preferred variants are described in WO 94/02597 A1 , WO 94/18314 A1 , WO 97/43424 A1 and SEQ ID NO: 4 of WO 99/19467 A1. Different suitable amylases include amylases having SEQ ID NO: 6 in WO 02/10355 A2 or variants thereof having about 90% sequence identity thereto.
- amylases which are suitable are amylases having SEQ ID NO: 6 in WO 99/019467 A1 or variants thereof having about 90% sequence identity to SEQ ID NO: 6.
- Additional amylases which can be used are those having SEQ ID NO: 1 , SEQ ID NO: 3, SEQ ID NO: 2 or SEQ ID NO: 7 of WO 96/23873 A1 or variants thereof having 90% sequence identity to SEQ ID NO: 1 , SEQ ID NO: 2, SEQ ID NO: 3 or SEQ ID NO: 7.
- amylases which can be used are amylases having SEQ ID NO: 2 of WO 2008/153815 A1 , SEQ ID NO: 10 in WO 01/66712 A2 or variants thereof having about 90% sequence identity to SEQ ID NO: 2 of WO 2008/153815 A1 or about 90% sequence identity to SEQ ID NO: 10 in WO 01/66712 A2.
- amylases are the a-amylase having SEQ ID NO: 12 in WO 01/66712 A2 or a variant having at least about 90% sequence identity to SEQ ID NO: 12.
- amylase variants such as those described in WO 2011/098531 A1 , WO 2013/001078 A1 and WO 2013/001087 A2.
- amylases are AmplifyTM Prime 100 L, DuramylTM, TermamylTM, FungamylTM, StainzymeTM, Stainzyme PlusTM, NatalaseTM, Liquozyme X and BANTM (from Novozymes A/S), and RapidaseTM, PurastarTM/EffectenzTM, Powerase and Preferenz S100 (from Genencor International Inc./DuPont).
- Suitable peroxidases/oxidases include those of plant, bacterial or fungal origin. Chemically modified or protein engineered mutants are included. Examples of useful peroxidases include peroxidases from Coprinus , e.g., from C. cinereus , and variants thereof as those described in WO 93/24618 A1 , WO 95/10602 A1 , and WO 98/15257 A1 .
- the detergent enzyme(s) may be included in a detergent composition by adding separate additives containing one or more enzymes, or by adding a combined additive comprising all of these enzymes.
- a detergent additive as contemplated herein, i.e., a separate additive or a combined additive, can be formulated, for example, as a granulate, liquid, slurry, etc.
- Preferred detergent additive formulations are granulates, in particular non-dusting granulates, liquids, in particular stabilized liquids, or slurries.
- Mannanases which are particularly preferred according to the invention are mannanases which are sold, for example, under the trade names Mannaway® by the company Novozymes or Purabrite® by the company Genencor.
- a mannanase preferably comprised in the composition according to the instant invention is Mannaway® 4.0 L.
- the aqueous mixture L in the context of the use according to the first aspect of the invention preferably also comprise at least one builder Bz.
- aqueous mixture L in the context of the use according to the first aspect of the invention comprises at least one builder Bz
- the amount of all builders Bz in aqueous mixture L is preferably between 0.1 ppm to 1000 ppm, preferably 1 ppm to 100 ppm, more preferably 10 ppm to 50 ppm relative to the total weight of aqueous mixture L.
- the builder Bz may particularly be a chelating agent that forms water-soluble complexes with calcium and magnesium.
- builders Bz include zeolites, diphosphates (pyrophosphates), triphosphates such as sodium triphosphate (“STP” or “STPP”), carbonates such as sodium carbonate, soluble silicates such as sodium metasilicate, layered silicates (e.g., SKS-6 from Hoechst), ethanolamines such as 2-aminoethan-1-ol (“MEA”), diethanolamine (“DEA”, also known as iminodiethanol), triethanolamine (“TEA”, also known as 2,2',2"-nitrilotriethanol) , and carboxymethyl inulin (“CMI”), and combinations thereof.
- MEA 2-aminoethan-1-ol
- DEA diethanolamine
- TAA triethanolamine
- CMI carboxymethyl inulin
- Non-limiting examples of builders Bz include homopolymers of polyacrylates or copolymers thereof, such as poly(acrylic acid) (“PAA”) or copoly(acrylic acid/maleic acid) (“PAA/PMA”). Further nonlimiting examples of builders Bz include polyaspartic acids and polyglutamic acids and their salts, citrates, ascorbic acid chelators such as aminocarboxylates, aminopolycarboxylates, like A/,A/-dicarboxymethyl glutamic acid and methylglycine A/,A/-diacetic acid, and phosphonates, and alkyl- or alkenylsuccinic acid.
- PAA poly(acrylic acid)
- PAA/PMA copoly(acrylic acid/maleic acid)
- Further nonlimiting examples of builders Bz include polyaspartic acids and polyglutamic acids and their salts, citrates, ascorbic acid chelators such as aminocarboxylates, aminopolycarboxylates, like
- builders Bz include 2,2’,2”-nitrilotriacetic acid (“NTA”), ethylenediaminetetraacetic acid (“EDTA”), diethylenetriaminepentaacetic acid (“DTPA”), iminodisuccinic acid (“IDS”), ethylenediamine- A/,A/’-disuccinic acid (“EDDS”), glutamic acid-A/,A/-diacetic acid (“GLDA”), 1-hydroxyethane-1 ,1- diphosphonic acid (“HEDP”), ethylenediaminetetra-(methylenephosphonic acid) (“EDTMPA”), diethylenetriaminepentakis(methylenephosphonic acid) (“DTPMPA” or “DTMPA”), N-(2- hydroxyethyl)iminodiacetic acid (“EDG”), aspartic acid-AZ-monoacetic acid (“ASMA”), aspartic acid- A/,A/-diacetic acid (“ASDA”), aspartic acid-AZ-monoacetic
- Preferred builder Bz according to the instant invention are selected from the group of aminopolycarboxylates, like A/,A/-dicarboxymethyl glutamic acid and methylglycine A/,A/-diacetic acid, citrates, polyaspartic acids and polyglutamic acids and their salts.
- aqueous mixture L in the context of the use according to the first aspect of the invention may comprise at least one non-aqueous solvent.
- the ratio between the weight of the water comprised by aqueous mixture L to the weight of all non-aqueous solvents comprised by aqueous mixture L is preferably in the range of from 999 : 1 to 1 : 999, preferably in the range of from 99 : 1 to 1 : 99, more preferably in the range of from 9 : 1 to 1 : 9, more preferably in the range of from 8 : 2 to 2 : 8, more preferably in the range of from 7 : 3 to 3 : 7, more preferably in the range of from 6 : 4 to 4 : 6, more preferably at 1 : 1 .
- Suitable non-aqueous solvents include monohydric or polyhydric alcohols, alkanolamines or glycol ethers, provided they are miscible with water in the specified concentration range.
- the solvents are preferably selected from ethanol, n-propanol, /so-propanol, butanols, glycol, propanediol, butanediol, glycerine, diglycol, propyldiglycol, butyldiglycol, hexylene glycol, ethylene glycol methyl ether, ethylene glycol ethyl ether, ethylene glycol propyl ether, n-butyl glycol ether, ethylene glycol mono-glycol ether, diethylene glycol ethyl ether, propylene glycol methyl ether, propylene glycol ethyl ether, propylene glycol propyl ether, dipropylene glycol monomethyl ether, dipropylene glycol monoethy
- composition ZG is a surfactant preparation
- it preferably contains a polyol as the non-aqueous solvent.
- the polyol can comprise glycerol, 1 ,2-propanediol, 1 ,3-propanediol, ethylene glycol, diethylene glycol and / or dipropylene glycol.
- aqueous mixture L in the context of the use according to the first aspect of the invention may comprise at least one preservative.
- aqueous mixture L in the context of the use according to the first aspect of the invention comprises at least one preservative
- the amount of all preservatives in aqueous mixture L is preferably between 0.1 ppm to 1000 ppm, preferably 1 ppm to 100 ppm, more preferably 10 ppm to 50 ppm, relative to the total weight of aqueous mixture L.
- Preferred preservatives are selected from the group consisting of phenoxyethanol, sodium levulinate, sodium benzoate, p-anisic acid, potassium sorbate, benzoic acid, glyceryl caprylate, capryl glycol, penthylene glycol, methyl propane diol, bronopol, isothiazolinone (in particular selected from methylisothiazolinone, chloromethylisothiazolinone). 1 .4.6 Benefit
- aqueous mixture L in the context of the use according to the first aspect of the invention may comprise at least one benefit agent, preferably at least one encapsulate comprising a benefit agent, preferably a sensorial benefit agent.
- aqueous mixture L in the context of the use according to the first aspect of the invention comprises at least one benefit agent
- the total amount of benefit agent in aqueous mixture L is preferably between 0.1 ppm to 1000 ppm, preferably 1 ppm to 100 ppm, more preferably 10 ppm to 50 ppm relative to the total weight of aqueous mixture L.
- the preferred benefit agent is a perfume.
- a benefit agent may be provided in an encapsulate.
- Aqueous mixture L optionally also comprise an unconfined (also called non-encapsulated) benefit agent, for example a volatile benefit agent.
- the volatile benefit agent is a perfume
- the perfumes are suitable for use as the encapsulated volatile benefit agent and also as the unconfined perfume component.
- Preferred encapsulates in this context comprise shear/pressu re-sensitive action encapsulates, whereby the sensorial benefit agent is released in response to mechanical force (e.g., friction, pressure, shear stress) on the encapsulate.
- the encapsulate shell is preferably comprised of materials including but not limited to polyurethane, polyamide, polyolefin, polysaccharide, protein, silicone, lipid, modified cellulose, gums, polyacrylate, polyphosphate, polystyrene, polyesters or combinations of these materials.
- the benefit agent is a sensorial benefit agent, a skin benefit agent or an olfactory benefit agent and/or may be a volatile benefit agent.
- Sensorial benefit agents may also have benefits for hair and/or hard surfaces and/or fabrics.
- the sensorial benefit may have anti-foam properties, and as such it is advantageous for foaming purposes that it is encapsulated so as not interfering with the foam until release by rubbing.
- Suitable volatile benefit agents include but are not limited to perfumes, insect repellents, essential oils, sensates such as menthol and aromatherapy actives, preferably perfumes. Mixtures of volatile benefit agents may be used.
- aqueous mixture L in the context of the use according to the first aspect of the invention may comprise at least one polymer for use in detergents.
- the polymer may function as a co-builder as mentioned above, or may provide antiredeposition, fiber protection, soil release, dye transfer inhibition, viscosity modifiers, grease cleaning and/or anti-foaming properties.
- the amount of all polymers in aqueous mixture L is preferably between 0.1 ppm to 1000 ppm, preferably 1 ppm to 100 ppm, more preferably 10 ppm to 50 ppm relative to the total weight of aqueous mixture L.
- Exemplary polymers include starch, polyquaternium (in particular polyquaternium 10),(carboxymethyl)cellulose (“CMC”), poly(vinyl alcohol) (“PVA”), polyvinylpyrrolidone) (“PVP”), poly(ethyleneglycol) or poly(ethylene oxide) (“PEG”), ethoxylated poly(ethyleneimine), carboxymethyl inulin (“CMI”), and polycarboxylates such as PAA, PAA/PMA, poly-aspartic acid, and lauryl methacrylate/acrylic acid copolymers, hydrophobically modified CMC (“HM-CMC”) and silicones, copolymers of terephthalic acid and oligomeric glycols, copolymers of poly(ethylene terephthalate) and poly(oxyethene terephthalate) (“PET-POET”), PVP, poly(vinylimidazole) (“PVI”), poly(vinylpyridine-A/-oxide) (“PVPO”
- exemplary polymers include sulfonated polycarboxylates, polyethylene oxide and polypropylene oxide (“PEO-PPO”) and diquaternium ethoxy sulfate.
- PEO-PPO polypropylene oxide
- diquaternium ethoxy sulfate diquaternium ethoxy sulfate.
- Other exemplary polymers are disclosed in, e.g. WO 2006/130575 A2. Salts of the above-mentioned polymers are also contemplated.
- a composition ZG according to the instant invention is characterized in that it comprises at least one selected from anti-redeposition polymers and soil release polymers, with soil release polymers being preferred.
- the anti-redeposition polymer or soil release polymer is selected from the group consisting of modified cellulose, preferably carboxymethylcellulose, cellulose acetate and methylcellulose, modified starch, modified inulin, preferably carboxy methyl inulin, polyitaconic acid, polyvinylpyrrolidone, polyvinyl alcohol, and polyethylene glycol, with carboxymethylcellulose and methylcellulose being most preferred.
- Further preferred soil release polymers are water soluble polyesters as for example from the TexCare® range commercially available under the name TexCare SRN 260, TexCare SRN 170, TexCare SRN 260 Life and combinations thereof, as well, as the soil release polymers disclosed in WO 2016/075178 A1 , WO 2016/075179 A1 , EP 3 489 340 A1 and EP 3 489 338 A1 .
- Further preferred soil release polymers are selected from carboxy methyl inulins. A commercial example is CarboxylineOCMI.
- EP 1 746 109 A2 discloses hybrid polymers of amylose and acrylates, that can also advantageously used in the composition of the instant invention as soil release polymers.
- a commercial example for this type of soil release polymers is Alcoguard® H 5941.
- biopolymers including: starch, like e.g. corn starch, zea mays starch and tapioca starch, modified starch, like e.g. starch hydroxypropyltrimonium chloride and hydrolyzed corn starch, cellulose, bacterial cellulose, modified cellulose, like e.g. microcrystalline cellulose, hydroxypropyl methylcellulose and cetyl hydroxyethylcellulose, guar gum, pectin, inulin, carrageenan, alginate, galactoarabinan, polycitronellol, carboxymethyl inulin, carboxymethyl cellulose, polyitaconic acid and combinations and salts thereof.
- starch like e.g. corn starch
- zea mays starch and tapioca starch
- modified starch like e.g. starch hydroxypropyltrimonium chloride and hydrolyzed corn starch
- cellulose bacterial cellulose
- modified cellulose like e.g. microcrystalline cellulose,
- aqueous mixture L in the context of the use according to the first aspect of the invention comprises at least one polymer selected from the group consisting of carboxymethylcellulose, polyethylenglycol, starch, carboxymethyl inulin, poly-aspartic acid, polyglutamic acid, sulfonated polycarboxylates, polyethylene oxide, polypropylene oxide, polyquaternium. These polymers are preferred as they are ecologically favorable.
- Preferred polyquaternium is polyquaternium 10 (CAS-Nr.: 81859-24-7), which is quaternized hydroxyethyl cellulose.
- the aqueous mixture L in the context of the use according to the first aspect of the invention may comprise at least one further additive.
- the further additive is preferably selected from the group consisting of bleaching systems, antiredeposition aids, fiber protection agents, soil release agents, dye transfer inhibitors, fabric hueing agents, blueing dyes, enzyme stabilizing agents like boric acid, pH-regulators, emollients, emulsifiers, thickeners/ viscosity regulators /stabilizers, UV photoprotective filters, antioxidants, hydrotropes (or polyols), solids and fillers, film formers, pearlescent additives, deodorant and antiperspirant active ingredients, insect repellents, self-tanning agents, preservatives, conditioners, perfumes, dyes, odour absorbers, cosmetic active ingredients, care additives, superfatting agents, solvents, malodor removers.
- the at least one biosurfactant SBIO may be provided in pure form or as a composition ZG in mixture with further compounds, which are set forth below.
- Composition ZG preferably is a cleaning formulation which may be employed in the use according to the first aspect of the invention.
- composition ZG When subjected to the use according to the first aspect of the invention, the composition ZG may then be diluted with water and the thus obtained aqueous mixture L may then be contacted with the woolen article Aw to wash article Aw- Hence, typically, composition ZG comprises the same compounds (except water) as aqueous solution L, just in higher concentration.
- composition ZG comprises water W
- the pH of the water W in composition ZG at 25 °C is from 3.5 to 9, preferably from 5 to 9, preferably from 6 to 9, and particularly preferably from 7 to 8.
- Composition ZG may comprise further ingredients, depending on the specific area of application of composition ZG.
- the present invention is based on the use of the at least one biosurfactant SBIO to reduce shrinking of the woolen article Aw during washing.
- the combination of the at least one biosurfactant SBIO with further surfactants is therefore not necessary, since, in particular, certain surfactants such as SLES, an anionic surfactant, are inferior to SBIO in terms of reducing the shrinking of the woolen article Aw-
- certain surfactants such as SLES, an anionic surfactant
- composition ZG comprises, besides the at least one biosurfactant SBIO, at least one further surfactant which is different from the at least one biosurfactant SBIO, preferably at least two further surfactants which are both different from the at least one biosurfactant SBIO, more preferably at least three further surfactants which are different from the at least one biosurfactant SBIO-
- This at least one further surfactant may be selected from anionic surfactants, nonionic surfactants, cationic surfactants, amphoteric surfactants, and in particular is selected from anionic surfactants, nonionic surfactants.
- the amount of all biosurfactants, in particular the amount of all glucolipids, rhamnolipids, and sophorolipids, in the composition ZG is in the range of from 5 to 60 wt.-%, more preferably 15 to 40 wt.-%, based on the total mass of the composition ZG.
- Composition ZG optionally comprises at least one nonionic surfactants.
- Preferred nonionic surfactants are described under item 1.4.1 .
- Composition ZG optionally comprises at least one anionic surfactants.
- Preferred anionic surfactants are described under item 1 .4.2.
- Composition ZG optionally comprises at least one amphoteric surfactants.
- Preferred amphoteric surfactants are described under item 1 .4.3.
- Composition ZG optionally comprises at least one cationic surfactants.
- Preferred anionic surfactants are described under item 1 .4.4.
- Composition ZG optionally comprise at least one enzyme E.
- composition ZG comprises at least one enzyme E
- the enzyme E is selected from the group consisting of protease, amylase, cellulase, mannanase, lipase, cutinase, pectate lyase, peroxidase, oxidase, laccase, even more preferably selected from the group consisting of cellulase, protease, lipase, cutinase, amylase, peroxidase, oxidase, mannanase.
- the at least one enzyme E is selected from the group consisting of protease, amylase, lipase, mannanase. Most preferred, the at least one enzyme E is a lipase.
- composition ZG comprises at least one enzyme E
- the amount of all enzymes E in the composition ZG is in the range of 0.1 to 4 wt.-% per the total weight of composition ZG.
- the at least one enzyme E may be included in the composition ZG by adding separate additives (“detergent additives”) containing one or more enzymes, or by adding a combined additive comprising all enzymes E comprised by the composition ZG.
- a detergent additive as contemplated herein, i.e., a separate additive or a combined additive, can be formulated, for example, as a granulate, liquid, slurry, etc.
- Preferred detergent additive formulations are granulates, in particular non-dusting granulates, liquids, in particular stabilized liquids, or slurries.
- Non-dusting granulates may be produced, e.g., as disclosed in US 4,106,991 A and
- US 4,661 ,452 A may optionally be coated by methods known in the art.
- waxy coating materials are poly(ethylene oxide) products (polyethyleneglycol, “PEG”) with mean molar weights of from about 1000 to about 20000; ethoxylated nonylphenols having from 16 to 50 ethylene oxide units; ethoxylated fatty alcohols in which the alcohol contains from 12 to 20 carbon atoms and in which there are 15 to 80 ethylene oxide units; fatty alcohols; fatty acids; and mono- and di- and triglycerides of fatty acids.
- PEG poly(ethylene oxide) products
- PEG polyethyleneglycol
- Liquid enzyme preparations may, for instance, be stabilized by adding a polyol such as propylene glycol, a sugar or sugar alcohol, lactic acid or boric acid according to established methods.
- Protected enzymes may be prepared according to the method disclosed in EP 0 238216 A1 .
- Composition ZG optionally comprises at least one builder Bz.
- the amount of all builders Bz in composition ZG is preferably between 0.1 wt.-% to 10 wt.-%, preferably 1 to 7 wt.-%, relative to the total weight of composition ZG.
- Preferred builder Bz are described under item 1 .4.3
- composition ZG may comprise at least one solvent selected from water or non-aqueous solvent, preferably non-aqueous solvent.
- Suitable non-aqueous solvents are described under item 1.4.4.
- composition ZG comprises at least one non-aqueous solvent
- the amount of all non-aqueous solvent in composition ZG is preferably between 0.5 wt.-% to 15 wt.-%, preferably 1 wt.-% to 10 wt.-%, relative to the total weight of composition ZG.
- composition ZG optionally comprises at least one preservative.
- the amount of all preservatives in composition ZG is preferably between 0.001 wt.-% to 2 wt.-%, preferably 0.01 to 0.1 wt.-%, relative to the total weight of composition ZG.
- Suitable non-aqueous solvents are described under item 1.4.5.
- Composition ZG optionally comprise at least one encapsulate comprising a benefit agent, preferably a sensorial benefit agent.
- the preferred benefit agent is a perfume.
- a benefit agent may be provided in an encapsulate.
- Composition ZG optionally also comprises an unconfined (also called non-encapsulated) benefit agent, for example a volatile benefit agent. Where the volatile benefit agent is a perfume, the perfumes are suitable for use as the encapsulated volatile benefit agent and also as the unconfined perfume component.
- composition ZG comprises at least one benefit agent
- the total amount of benefit agent is preferably from 0.01 wt.-% to 10 wt.-%, more preferably from 0.05 wt.-% to
- Composition ZG optionally comprises at least one polymer for use in detergents that are different from the polymers described before.
- composition ZG comprises at least one polymer
- the amount of all polymers in composition ZG is preferably between 0.05 wt.-% to 8 wt.-%, preferably 0.1 to 5 wt.-%, relative to the total weight of composition ZG.
- Preferred polymers are described under item 1.4.7.
- Composition ZG comprise at least one further additives selected from the group consisting of bleaching systems, anti-redeposition aids, fiber protection agents, soil release agents, dye transfer inhibitors, fabric hueing agents, blueing dyes, enzyme stabilizing agents like boric acid, pH-regulators, emollients, emulsifiers, thickeners/ viscosity regulators /stabilizers, UV photoprotective filters, antioxidants, hydrotropes (or polyols), solids and fillers, film formers, pearlescent additives, deodorant and antiperspirant active ingredients, insect repellents, selftanning agents, preservatives, conditioners, perfumes, dyes, odour absorbers, cosmetic active ingredients, care additives, superfatting agents, solvents, malodor removers.
- further additives selected from the group consisting of bleaching systems, anti-redeposition aids, fiber protection agents, soil release agents, dye transfer inhibitors, fabric hueing agents, blueing dyes, enzyme stabilizing agents like boric acid,
- composition ZG is provided is not particularly limited and depends on the area of use.
- Composition ZG may be in any convenient form, e.g., a bar, a homogenous tablet, a tablet having two or more layers, a pouch having one or more compartments, a regular or compact powder, a granule, a paste, a gel, or a regular, compact or concentrated liquid.
- detergent formulation forms such as layers (same or different phases), pouches, as well as forms for machine dosing unit.
- Pouches can be configured as single or multicompartments. It can be of any form, shape and material which is suitable for hold the composition, e.g. without allowing the release of the composition from the pouch prior to water contact.
- the pouch is made from water soluble film which encloses an inner volume. Said inner volume can be divided into compartments of the pouch.
- Preferred films are polymeric materials preferably polymers which are formed into a film or sheet.
- Preferred polymers, copolymers or derivates thereof are selected polyacrylates, and water soluble acrylate copolymers, methyl cellulose, carboxy methyl cellulose, sodium dextrin, ethyl cellulose, hydroxyethyl cellulose, hydroxypropyl methyl cellulose, maltodextrin, poly methacrylates, most preferably polyvinyl alcohol copolymers and, hydroxypropyl methyl cellulose (HPMC).
- the level of polymer in the film for example PVA, is at least about 60%.
- Preferred average molecular weight will typically be from about 20,000 to about 150,000.
- Films can also be of blend compositions comprising hydrolytically degradable and water soluble polymer blends such as polyactide and polyvinyl alcohol (known under the Trade reference M8630 as sold by Chris Craft In. Prod. Of Gary, Ind., US) plus plasticisers like glycerol, ethylene glycerol, Propylene glycol, sorbitol and mixtures thereof.
- the pouches can comprise a solid laundry detergent composition or part components and/or a liquid cleaning composition or part components separated by the water- soluble film.
- the compartment for liquid components can be different in composition than compartments containing solids, see for example US 2009/0011970 A1 .
- Detergent ingredients can be separated physically from each other by compartments in water dissolvable pouches or in different layers of tablets. Thereby negative storage interaction between components can be avoided. Different dissolution profiles of each of the compartments can also give rise to delayed dissolution of selected components in the wash solution.
- Composition ZG can be in form of a laundry soap bar and used for hand washing laundry, fabrics and/or textiles.
- laundry soap bar includes laundry bars, soap bars, combo bars, syndet bars and detergent bars.
- the types of bar usually differ in the type of surfactant they contain, and the term laundry soap bar includes those containing soaps from fatty acids and/or synthetic soaps.
- the laundry soap bar has a physical form which is solid and not a liquid, gel or a powder at room temperature.
- the term solid is defined as a physical form which does not significantly change over time, i.e. if a solid object (e.g. laundry soap bar) is placed inside a container, the solid object does not change to fill the container it is placed in.
- the bar is a solid typically in bar form but can be in other solid shapes such as round or oval.
- the detergent composition according to the instant invention can be formulated as a granular detergent as described in WO 2009/092699 A1 , EP 1 705 241 A1 , EP 1 382 668 A1 , WO 2007/001262 A1 , US 6,472,364 B1 , WO 2004/074419 A2 or WO 2009/102854 A1 .
- Composition ZG may also be in the form of a liquid or gel detergent.
- a liquid or gel detergent may be aqueous, typically containing at least 20% by weight water, with the percentages referring to the total composition.
- Other types of liquids including without limitation, alkanols, amines, diols, ethers and polyols may be included in an aqueous liquid or gel.
- An aqueous liquid or gel detergent composition may contain from 0-30 wt.-% organic solvent, with the percentages referring to the total composition.
- a liquid or gel detergent may be non-aqueous.
- composition ZG according to the invention is characterized in that the pH of the composition at 25°C is from 3.0 to 9, preferably from 4.0 to 7 and particularly preferably from 5.0 to 6.6.
- the present invention relates to a method of washing at least one woolen article Aw, comprising the steps of a. contacting the at least one woolen article Aw with an aqueous mixture L comprising at least one biosurfactant SBIO, b. at least partially separating L from Aw, c.
- the temperature of the aqueous mixture L, for at least as long as the aqueous mixture L is contacted with the at least one woolen article Aw is in a range of from > 0 °C to 45 °C, in particular in a range of from 1 °C to 40 °C, preferably in a range of from 5 °C to 35 °C, more preferably in a range of from 10 °C to 30 °C, more preferably in a range of from 15 °C to 30 °C, even more preferably in a range of from 20 °C to 30 °C, even more preferably in a range of from 25 °C to 30 °C, wherein the biosurfactant SBIO is selected from the group consisting of rhamnolipids, glucolipids, sophorolipids, and wherein the biosurfactant SBIO, in particular, is selected from the group consisting of rhamnolipids, sophoro
- the article Aw essentially consists of wool.
- the woolen article Aw in particular is an article of clothing, preferably selected from pants, shirt, socks, hosiery, scarfs, gloves, shoes, sweaters, suits, hats, caps, bonnets. It may, in addition, also be selected from the group consisting of curtains, napkins, bed linen, tablecloth, blanket, bedspread, duvet cover.
- wool refers to the textile fiber of a mammal, in particular of livestock.
- it refers to the textile fiber of a mammal selected from the group consisting of sheep, goat (the goat being in particular selected from the group of cashmere goat, angora goat), rabbit (the rabbit being in particular an angora rabbit), camelide (the camelide being in particular selected from the group of llama, vikuna, alpaca, guanaco), yak, beaver, otter, guinea pig, horse, donkey, chinchilla.
- it refers to the textile fiber of a mammal selected from the group consisting of sheep, goat, most preferably of sheep.
- the shrinking of the at least one woolen article Aw is reduced during washing of Aw in the aqueous mixture L comprising the at least one woolen article Aw and the at least one biosurfactant SBIO-
- the shrinking as well as the extent of shrinking may be determined as described under chapter 1 .2 above.
- Assay X is used to determine whether a certain substance (abbreviated as “X”) reduces the shrinking of a woolen article Aw (as well as the extent of shrinking) during washing.
- Assay X is hence the preferred test to determine the reduction of shrinking of the at least one woolen article Aw during washing in the method according to the second aspect of the invention, in particular in an aqueous mixture L, as well as the extent of shrinking of the at least one woolen article Aw during washing, in particular in an aqueous mixture L.
- step a. of the method according to the second aspect of the invention the at least one woolen article Aw is contacted with an aqueous mixture L comprising at least one biosurfactant SBIO-
- the “biosurfactant Ssio” is selected from the group consisting of glucolipids, rhamnolipids, sophorolipids, in particular selected from the group of sophorolipids, rhamnolipids, preferably from the group of rhamnolipids.
- Glucolipids, rhamnolipids, and sophorolipids are glycolipids. Such glycolipids are described in the art together with their syntheses, for example in EP 0499434 A1 (glucolipids are referred to as “glucose lipids” in this document), DE 196 48 439 A1 , DE 196 00 743 A1 .
- WO 03/006146 A1 , US 2008/0213194 A1 , JP H01 -304034 A1 , CN 1337439 A describe further methods for the synthesis of rhamnolipids.
- WO 03/002700 A1 , US 4,305,961 A, US 7,556,654 B1 describe further methods for the synthesis of sophorolipids. 2.2. 1. 1 Glucolipids
- glucolipid is, in particular, to be understood as referring to a structure according to formula (I), even more preferred a structure according to formula (II):
- mGL 3, 2, 1 or 0, preferably 1 or 0.
- Residues R 1GL and R 2GL are, independently of one another, an organic radical having 2 to 24 carbon atoms, preferably 5 to 20, more preferably 7 to 15, even more preferably 7, carbon atoms, wherein preferably R 1GL and R 2GL are independently of one another selected from the group consisting of optionally substituted alkyl radicals with 2 to 24 preferably 5 to 20, more preferably 7 to 15, even more preferably 7, carbon atoms, wherein hydroxy substituted alkyl radicals are preferred substituted alkyl radicals, optionally substituted alkenyl radicals with 2 to 24 carbon atoms, preferably 5 to 20, more preferably 7 to 15, even more preferably 7, carbon atoms, wherein hydroxy substituted alkenyl radicals are preferred substituted alkenyl radicals, wherein more preferably R 1GL and R 2GL are independently of one another selected from the group consisting of pentenyl, heptenyl, nonenyl, undecenyl, tridecenyl and -(CH2)oCH
- curvy bond in structure (I) implies that the respective substituent may be axial or equatorial, preferably is equatorial.
- Alkyl radicals may be branched or linear.
- Alkenyl radicals may be branched or linear and preferably contain between one to three double bonds.
- residues R 2GL may be identical or different from one another.
- glucolipids are used as biosurfactants SBIO in the method according to the second aspect of the invention.
- glucolipids are used as biosurfactants SBIO in the method according to the second aspect of the invention.
- 0.5 wt.-% to 20 wt.-%, preferably 3 wt.-% to 17 wt.-%, particularly preferably 5 wt.-% to 15 wt-%. of all glucolipids that are used as biosurfactants SBIO in the method according to the second aspect of the invention are GL- C10C12:1 , where the wt.-% refers to the total weight of all the glucolipids that are used as biosurfactants SBIO in the method according to the second aspect of the invention.
- glucolipids are used as biosurfactants SBIO in the method according to the second aspect of the invention.
- 0.5 wt.-% to 20 wt.-%, preferably 2 wt.-% to 15 wt.-%, particularly preferably 3 wt.-% to 12 wt.-%, of all the glucolipids that are used as biosurfactants SBIO in the method according to the second aspect of the invention are GL-C10C12, where the wt.-% refers to the total weight of all the glucolipids that are used as biosurfactants SBIO in the method according to the second aspect of the invention.
- glucolipids are used as biosurfactants SBIO in the method according to the second aspect of the invention.
- 1 wt.-% to 30 wt.-% preferably 5 wt.-% to 25 wt.-%, particularly preferably 10 wt.-% to 20 wt.-%, of all the glucolipids that are used as biosurfactants SBIO in the method according to the second aspect of the invention, are GL-C8C10,
- glucolipids that are used as biosurfactants SBIO in the method according to the second aspect of the invention are GL-C10C12:1 ,
- 0.5 wt.-% to 20 wt.-%, preferably 2 wt.-% to 15 wt.-%, particularly preferably 3 wt.-% to 12 wt.-%, of all the glucolipids that are used as biosurfactants SBIO in the method according to the second aspect of the invention, are GL-C10C12, where the wt.-% refers to the total weight of all the glucolipids that are used as biosurfactants SBIO in the method according to the second aspect of the invention.
- glucolipids are used as biosurfactants SBIO in the method according to the second aspect of the invention.
- 10 wt.-% to 20 wt.-% of all the glucolipids that are used as biosurfactants SBIO in the method according to the second aspect of the invention are GL-C8C10,
- 3 wt.-% to 12 wt.-%, of all the glucolipids that are used as biosurfactants SBIO in the method according to the second aspect of the invention are GL-C10C12, where the wt.-% refers to the total weight of all the glucolipids that are used as biosurfactants SBIO in the method according to the second aspect of the invention.
- glucolipids are used as biosurfactants SBIO in the method according to the second aspect of the invention.
- 0 wt.-% to 5 wt.-% preferably 0.01 wt.-% by weight to 4 wt.-%, particularly preferably 0.1 wt.-% to 3 wt.-%, of all the glucolipids that are used as biosurfactants SBIO in the method according to the second aspect of the invention, are GL-C10, where the wt.-% refers to the total weight of all the glucolipids that are used as biosurfactants SBIO in the method according to the second aspect of the invention.
- rhamnolipid is, in particular, to be understood as referring to compounds of the general formula (III) and salts thereof, preferably compounds according to the general structure (IV) and salts thereof,
- mRL 2, 1 or 0, preferably 1 or 0.
- nRL 1 or 0.
- the glycosidic bond between the two rhamnose units is preferably in the a-configuration.
- optically active carbon atoms of the fatty acids are preferably present as R-enantiomers (e.g. (R)-3- ⁇ (R)-3-[2-O-(a-L-rhamnopyranosyl)-a-L-rhamnopyranosyl]oxydecanoyl ⁇ oxydecanoate).
- R-enantiomers e.g. (R)-3- ⁇ (R)-3-[2-O-(a-L-rhamnopyranosyl)-a-L-rhamnopyranosyl]oxydecanoyl ⁇ oxydecanoate.
- Rhamnolipids applicable in the context of the instant invention can also be produced by fermentation of Pseudomonas, especially Pseudomonas aeruginosa, which are preferably non genetically modified cells, a technology already disclosed in the eighties, as documented e.g. in EP 0 282 942 A1 and DE 41 27 908 A1 .
- Rhamnolipids produced in Pseudomonas aeruginosa cells which have been improved for higher rhamnolipid titres by genetical modification can also be used in the context of the instant invention; such cells have for example been disclosed by Lei et al. in Biotechnol Lett. 2020 Jun;42(6):997-1002.
- Rhamnolipids produced by Pseudomonas aeruginosa are commercially available from Jeneil Biotech Inc., e.g. under the tradename Zonix, from Logos Technologies (technology acquired by Stepan), e.g. under the tradename NatSurFact, from Biotensidion GmbH, e.g. under the tradename Rhapynal, from AGAE technologies, e.g. under the name R90, R95, R95Md, R95Dd, from Locus Bio-Energy Solutions and from Shanghai Yusheng Industry Co. Ltd., e.g. under the tradename Bio- 201 Glycolipids.
- rhamnolipids are used as biosurfactants SBIO in the method according to the second aspect of the invention
- mixtures of mono-rhamnolipids and di-rhamnolipids wherein even more preferably the ratio of the weight of all mono-rhamnolipids used in the method according to the second aspect of the invention to the weight of all di-rhamnolipids used in the method according to the second aspect of the invention is in the range of from 9 : 1 to 1 : 9, preferably of from 8 : 2 to 2 : 8, more preferably of from 7 : 3 to 3 : 7, more preferred of from 6 : 4 to 4 : 6, most preferred is 1 : 1 .
- the rhamnolipids comprise 56 wt.-% to 95 wt.-%, preferably 60 wt.-% to 80 wt.-%, particularly preferably 66 wt.-% to 70 wt.-%, of diRL-C10C10, where the wt.-% refers to the total weight of all the rhamnolipids that are used as biosurfactants SBIO in the method according to the second aspect of the invention.
- rhamnolipids are used as biosurfactants SBIO in the method according to the second aspect of the invention
- 0.5 wt.-% to 15 wt.-%, preferably 3 wt.-% to 12 wt.-%, particularly preferably 5 wt.-% to 10 wt.-%, of the rhamnolipids are diRL-C10C12:1 , where the wt.-% refers to the total weight of all the rhamnolipids that are used as biosurfactants SBIO in the method according to the second aspect of the invention.
- rhamnolipids are used as biosurfactants SBIO in the method according to the second aspect of the invention
- 0.5 wt.-% to 25 wt.-%, preferably 3 wt.-% to 15 wt.-%, particularly preferably 5 wt.-% to 12 wt.-%, of the rhamnolipids are diRL-C10C12, where the wt.-% refers to the total weight of all the rhamnolipids that are used as biosurfactants SBIO in the method according to the second aspect of the invention.
- rhamnolipids are used as biosurfactants SBIO in the method according to the second aspect of the invention
- 0.1 wt.-% to 25 wt.-%, preferably 2 wt.-% to 10 wt.-%, particularly preferably 4 wt.-% to 8 wt.-%, of the rhamnolipids are diRL-C8C10, where the wt.-% refers to the total weight of all the rhamnolipids that are used as biosurfactants SBIO in the method according to the second aspect of the invention.
- rhamnolipids are monoRL-C8C10 and/or, preferably and,
- rhamnolipids are monoRL-C10C10, where the wt.-% refers to the total weight of all the rhamnolipids that are used as biosurfactants SBIO in the method according to the second aspect of the invention.
- rhamnolipids are used as biosurfactants SBIO in the method according to the second aspect of the invention
- 10 wt.-% to 30 wt.-% preferably 20 wt.-% to 30 wt.-%, particularly preferably 25 wt.-% to 30 wt.-%, of the rhamnolipids are monoRL-C10C10, where the wt.-% refers to the total weight of all the rhamnolipids that are used as biosurfactants SBIO in the method according to the second aspect of the invention.
- rhamnolipids are used as biosurfactants SBIO in the method according to the second aspect of the invention
- 10 wt.-% to 30wt.-% preferably 12 wt.-% to 25 wt.-%, particularly preferably 15 wt.-% to 20 wt.-%, of the rhamnolipids are diRL-C10C10, where the wt.-% refers to the total weight of all the rhamnolipids that are used as biosurfactants SBIO in the method according to the second aspect of the invention.
- rhamnolipids are used as biosurfactants SBIO in the method according to the second aspect of the invention
- 10 wt.-% to 30 wt.-%, preferably 12 wt.-% to 25 wt.-%, particularly preferably 15 wt.-% to 20 wt.-%, of the rhamnolipids are monoRL-C8C10, where the wt.-% refers to the total weight of all the rhamnolipids that are used as biosurfactants SBIO in the method according to the second aspect of the invention.
- rhamnolipids are used as biosurfactants SBIO in the method according to the second aspect of the invention
- 3 wt.-% to 25 wt.-%, preferably 5 wt.-% to 20 wt.-%, particularly preferably 10 wt.-% to 15 wt.-%, of the rhamnolipids are monoRL-C10C12:1 , where the wt.-% refers to the total weight of all the rhamnolipids that are used as biosurfactants SBIO in the method according to the second aspect of the invention.
- rhamnolipids are used as biosurfactants SBIO in the method according to the second aspect of the invention
- 1 wt.-% to 15 wt.-%, preferably 2 wt.-% to 10 wt.-%, particularly preferably 3 wt.-% to 8 wt.-%, of the rhamnolipids are diRL-C10C12, where the wt.-% refers to the total weight of all the rhamnolipids that are used as biosurfactants SBIO in the method according to the second aspect of the invention.
- the term “sophorolipids” is in particular to be understood as referring to compounds of the general formulae (V), (VII) and salts thereof, preferably compounds of the general formulae (VI), (VIII) and salts thereof:
- Formulae (V), (VII) represent the acid form
- Formulae (VI), (VIII) represent the lactone form
- nSL 1 or 0.
- R 1SL H or -CO-CH 3 ,
- R 2SL H or -CO-CH 3 ,
- R3si_ a divalent organic moiety which comprises 6 to 32 carbon atoms, preferably 12 to 20, more preferably 14 to 16, most preferably 15.
- R 3SL preferably is an optionally substituted, divalent hydrocarbon moiety comprising 6 to 32 carbon atoms, wherein hydroxy substituted hydrocarbon moieties are preferred as substituted hydrocarbon moieties,
- R3SI_ m ore preferably is selected from the group consisting of
- optionally substituted alkylene radicals with 6 to 32, preferably 12 to 20, more preferably 14 to 16, most preferably 15 carbon atoms, wherein hydroxy-substituted alkylene radicals are preferred substituted alkylene radicals, and wherein it is preferred that the optionally substituted alkylene radicals are unbranched,
- optionally substituted alkenylene radicals with 6 to 32, preferably 12 to 20, more preferably 14 to 16, most preferably 15 carbon atoms, wherein hydroxy-substituted alkenylene radicals are preferred substituted alkenylene radicals, and wherein it is preferred that the optionally substituted alkenylene radicals are unbranched, and wherein it is preferred that the optionally substituted alkenylene radicals comprise one to three double or triple bonds.
- R 3SL even more preferably is selected from the group consisting of
- alkenylene radicals with 6 to 32, preferably 12 to 20, more preferably 14 to 16, most preferably 15 carbon atoms, wherein the alkenylene radicals preferably comprise one to three double or triple bonds, wherein the alkenylene radicals more preferably comprise one to three double bonds, even more preferably one double bond,
- alkenylene radicals with 6 to 32, preferably 12 to 20, more preferably 14 to 16, most preferably 15, carbon atoms carrying at least one hydroxy group, preferably one hydroxy group, wherein the alkenylene radicals preferably comprise one to three double or triple bonds, wherein the alkenylene radicals more preferably comprise one to three double bonds, even more preferably one double bond.
- R 4SL H, CH3 or a monovalent organic radical which comprises 2 to 10 carbon atoms.
- R 4SL is preferably selected from the group consisting of
- optionally substituted alkenyl radicals with 2 to 10 carbon atoms, wherein hydroxysubstituted alkenyl radicals are preferred substituted alkenyl radicals, and wherein it is preferred that the optionally substituted alkenyl radicals are unbranched, and wherein it is preferred that the optionally substituted alkenyl radicals comprise one to three double or triple bonds.
- R 4SL is more preferably selected from the group consisting of
- alkenyl radicals with 2 to 10 carbon atoms, wherein the alkenyl radicals preferably comprise one to three double or triple bonds, wherein the alkenyl radicals more preferably comprise one to three double bonds, even more preferably one double bond,
- alkenyl radicals with 2 to 10 carbon atoms carrying at least one hydroxy group, preferably one hydroxy group, wherein the alkenyl radicals preferably comprise one to three double or triple bonds, wherein the alkenyl radicals more preferably comprise one to three double bonds, even more preferably one double bond.
- R 4SL is most preferably selected from the group consisting of H, Methyl, Ethyl.
- the term “sophorolipids” is to be understood as referring to compounds of the general formulae (IX), (XI) and salts thereof, preferably compounds of the general formulae (X), (XII) and salts thereof:
- Formulae (IX), (X) represent the acid form
- Formulae (XI), (XII) represent the lactone form.
- nSL, R 1SL , R 2SL , R 4SL have the same meaning as described for formulae (V), (VII), (VI), (VIII).
- Sophorolipids may be used in accordance with the invention in their acid form or their lactone form.
- Preferred compositions in the context of the method according to the second aspect of the present invention comprise a sophorolipid in which the ratio by weight of lactone form to acid form is in the range of 20:80 to 80:20, especially preferably in the ranges of 30:70 to 40:60.
- Sophorolipids may be obtained as described in EP 1 411 111 A1 , paragraphs [0021], [0022]. 2.2.2 Aqueous mixture L
- step a. of the method according to the second aspect of the present invention the at least one woolen article Aw is contacted with an aqueous mixture L comprising at least one biosurfactant SBIO-
- the method according to the second aspect of the invention is a method washing of washing at least one woolen article Aw-
- the aqueous mixture L is hence, in particular, the washing solution in which the at least one article Aw is washed.
- the woolen article Aw is contacted with the aqueous mixture L comprising at least one biosurfactant SBIO” is to be understood that, in step a. of the method according the second aspect of the invention, the woolen article Aw is brought in direct contact with the aqueous mixture L comprising at least one biosurfactant SBIO, “in direct contact” meaning “wetting” of the woolen article Aw with the aqueous mixture L.
- the article Aw is at least partially immersed, even more preferably fully immersed into the aqueous mixture L.
- the article Aw is moved in the aqueous mixture L during step a. of the method according the second aspect of the invention.
- the mixture L is preferably brought in direct contact with at least a part of the surface of article Aw, onto which at least one impurity I is absorbed or adsorbed.
- a mixture L is obtained wherein in the water comprised by aqueous mixture L, at least one impurity I in dissolved or dispersed, preferably dispersed.
- the concentration of impurities I in the water comprised by aqueous mixture L therefore augments during step a. of the method according the second aspect of the invention.
- the aqueous mixture L that is employed during step a. of the method according the second aspect of the invention i.e. before it contacts the at least one woolen article Aw
- the mixture L that is obtained during the use according to step a. of the method according the second aspect of the invention is a dispersion, in which the at least one impurity I is the dispersed phase in the water W comprised by aqueous mixture L.
- the impurity I may either be in the form of a liquid (e.g. droplet) or a particle, and preferably is a particle.
- the impurity I is at least one impurity which may be inorganic or organic, in particular inorganic, preferably clay, more preferably kaolin.
- Organic impurities are preferably selected from oils and fats, which may be of vegetable, animal or synthetic origin, food based particles, soil, pollen, sebum, body fluids, for example, blood, sperm, sweat, urine, feces, liquor.
- Inorganic impurities are preferably selected from the group consisting of carbon black, fine dust, plastic particles (e.g. microplastic particles), metal oxides (preferably iron oxide), silica (such as sand), clay, dyes (such as pigments), more preferably clay, in particular kaolin.
- the beneficial effect of the method according to the second aspect of the invention is the reduction of shrinking of the at least one article Aw during washing in the aqueous mixture L comprising the at least one biosurfactant SBIO- Since the tendency of woolen articles to shrink is especially pronounced at temperatures above 45 °C, the best effect of the present invention is obtained when the aqueous mixture L has a temperature at or below 45 °C for at least as long as the aqueous mixture L contacts the at least one woolen article Aw-
- the temperature of the aqueous mixture L is in a range of from > 0 °C to 45 °C, in particular in a range of from 1 °C to 40 °C, preferably in a range of from 5 °C to 35 °C, more preferably in a range of from 10 °C to 30 °C, more preferably in a range of from 15 °C to 30 °C, even more preferably in a range of from 20 °C to 30 °C, even more preferably in a range of from 25 °C to 30 °C.
- the temperature of the aqueous mixture L is also in a range of from > 0 °C to 45 °C, in particular in a range of from 1 °C to 40 °C, preferably in a range of from 5 °C to 35 °C, more preferably in a range of from 10 °C to 30 °C, more preferably in a range of from 15 °C to 30 °C, even more preferably in a range of from 20 °C to 30 °C, even more preferably in a range of from 25 °C to 30 °C.
- the water which is used for rinsing Aw has a temperature, for at least as long as this water contacts the at least one woolen article Aw, in a range of from > 0 °C to 45 °C, in particular in a range of from 1 °C to 40 °C, preferably in a range of from 5 °C to 35 °C, more preferably in a range of from 10 °C to 30 °C, more preferably in a range of from 15 °C to 30 °C, even more preferably in a range of from 20 °C to 30 °C, even more preferably in a range of from 25 °C to 30 °C.
- the aqueous mixture L comprises the at least one biosurfactant SBIO in an amount in the range of from 0.000001 wt.-% to 50 wt.-%, preferably in the range of from 0.00001 wt.-% to 30 wt.-%, more preferably in the range of from 0.00005 wt.-% to 20 wt.-%, more preferably in the range of from 0.00009 wt.-% ppm to 15 wt.-%, more preferably in the range of from 0.0001 wt.-% to 14 wt.-%, more preferably in the range of from 0.001 wt.-% to 13 wt.-%, more preferably in the range of from 0.002 wt.-% to 12 wt.-%, more preferably in the range of from 0.005 wt.-% to
- 11 wt.-% more preferably in the range of from 0.01 wt.-% to 10 wt.-%, more preferably in the range of from 0.05 wt.-% to 9 wt.-%, more preferably in the range of from 0.1 wt.-% ppm to 8 wt.-%, more preferably in the range of from 0.5 wt.-% to 5 wt.-%, more preferably in the range of from 1 wt.-% to 5 wt.-%, more preferably in the range of from 1 wt.-% to 3 wt.-%, wherein the wt.-% in each case are the weight of all biosurfactants SBIO comprised by aqueous mixture L relative to the total weight of aqueous mixture L.
- Aw is contacted with the aqueous mixture L for at least 1 s, preferably at least 10 s, more preferably at least 1 min, even more preferably at least 5 min, preferably for 15 min to 240 min, even more preferably for 30 min to 180 min, even more preferably for 45 min to 120 min, even more preferably for 60 min to 120 min, even more preferably for 70 min to 100 min.
- the woolen article Aw is preferably moved in the aqueous mixture. This improves the washing efficiency.
- the washing in the context of the method according to the second aspect of the invention is preferably carried out in a washing machine.
- the at least one biosurfactant SBIO in the method according to the second aspect of the invention may be present at least partially as salts, in particular when it is comprised by the aqueous mixture L.
- the ratio of biosurfactants SBIO that are present as salts depends on the pH of the water in the aqueous mixture L.
- biosurfactants SBIO are salts in which the cation is selected from the group consisting of Li + , Na + , K + , Mg 2+ , Ca 2+ , AP + , NH4 + , ammonium ions, wherein the ammonium ions may be primary, secondary, tertiary or quaternary.
- Particularly preferred cations are selected from the group comprising, preferably consisting of, Na + , K + , NH4 + and the triethanolammonium cation.
- ammonium ions are tetramethylammonium, tetraethylammonium, tetrapropylammonium, tetrabutylammonium, [(2-hydroxyethyl)trimethylammonium] (choline) and also the cations of 2-aminoethanol (ethanolamine, “MEA”), diethanolamine (“DEA”), 2,2',2"-nitrilotriethanol (triethanolamine, “TEA”), 1-aminopropan-2-ol (monoisopropanolamine), ethylenediamine, diethylenetriamine, triethylenetetramine, tetraethylenepentamine, 1 ,4-diethylenediamine (piperazine), aminoethylpiperazine and aminoethylethanolamine.
- mixtures of the abovementioned cations may also be present as cations of the salts of the at least one biosurfactant SBIO, if present as salts, according to the invention.
- aqueous mixture L may comprise further ingredients, preferably at least one ingredient selected from surfactants (wherein the surfactants are different from SBIO), enzymes, builders, solvents, preservatives, benefit agents, polymers.
- aqueous mixture L in the context of the method according to the second aspect of the present invention, comprises, besides the at least one biosurfactant SBIO, at least one further surfactant SY, wherein the surfactant SY is different from SBIO.
- the at least one further surfactant SY may be selected from anionic surfactants, nonionic surfactants, cationic surfactants, amphoteric surfactants, and in particular is selected from anionic surfactants, nonionic surfactants.
- the aqueous mixture L in the context of the method according to the second aspect of the present invention comprises at least one further surfactant SY that is different from the biosurfactants SBIO
- the ratio of weights of all biosurfactants SBIO in the mixture L to the weight of all surfactants SY is in the range of from 99 : 1 to 1 : 99, preferably 90 : 10 to 10 : 90, more preferably 80 : 20 to 20 : 80, even more preferably 70 : 30 to 30 : 70, even more preferably 60 : 40 to 40 : 60, eve more preferably 50 : 50.
- Preferred nonionic surfactants are those described under item 1 .4.1 above.
- Preferred anionic surfactants are those described under item 1.4.2 above.
- Preferred amphoteric surfactants are those described under item 1 .4.3 above.
- Preferred cationic surfactants are those described under item 1.4.4 above.
- the aqueous mixture L in the context of the method according to the second aspect of the invention may also comprise at least one enzyme E.
- the enzyme E is selected from the group consisting of protease, amylase, cellulase, mannanase, lipase, cutinase, pectate lyase, peroxidase, oxidase, laccase, even more preferably selected from the group consisting of cellulase, protease, lipase, cutinase, amylase, peroxidase, oxidase, mannanase. It is even more preferred that the at least one enzyme E is selected from the group consisting of protease, amylase, lipase, mannanase. Most preferred, the at least one enzyme E is a lipase.
- the aqueous mixture L in the context of the method according to the second aspect of the invention comprises at least one enzyme E
- the amount of all enzymes E in the aqueous mixture L is in the range of 0.01 ppm to 1 wt.-%, preferably 0.01 ppm to 1000 ppm, more preferably 0.1 to 100 ppm, preferably 1 ppm to 10 ppm per the total weight of aqueous mixture L.
- Preferred cellulases are those described under point 1 .4.2.1 above.
- Preferred proteases are those described under point 1 .4.2.2 above.
- Preferred lipases and cutinases are those described under point 1 .4.2.3 above.
- Preferred amylases are those described under point 1 .4.2.4 above.
- Preferred oxidases and peroxidases are those described under point 1 .4.2.5 above.
- Preferred mannanases are those described under point 1.4.2.6 above.
- the aqueous mixture L in the context of the method according to the second aspect of the invention preferably also comprise at least one builder Bz.
- aqueous mixture L in the context of the method according to the second aspect of the invention comprises at least one builder Bz
- the amount of all builders Bz in aqueous mixture L is preferably between 0.1 ppm to 1000 ppm, preferably 1 ppm to 100 ppm, more preferably 10 ppm to 50 ppm relative to the total weight of aqueous mixture L.
- Preferred builders are those described under point 1 .4.3 above.
- aqueous mixture L in the context of the method according to the second aspect of the invention may comprise at least one non-aqueous solvent.
- the ratio between the weight of the water comprised by aqueous mixture L to the weight of all non-aqueous solvents comprised by aqueous mixture L is preferably in the range of from 999 : 1 to 1 : 999, preferably in the range of from 99 : 1 to 1 : 99, more preferably in the range of from 9 : 1 to 1 : 9, more preferably in the range of from 8 : 2 to 2 : 8, more preferably in the range of from 7 : 3 to 3 : 7, more preferably in the range of from 6 : 4 to 4 : 6, more preferably at 1 : 1 .
- Suitable non-aqueous solvents are those described under item 1 .4.4 above.
- aqueous mixture L in the context of the method according to the second aspect of the invention may comprise at least one preservative.
- aqueous mixture L in the context of the method according to the second aspect of the invention comprises at least one preservative
- the amount of all preservatives in aqueous mixture L is preferably between 0.1 ppm to 1000 ppm, preferably 1 ppm to 100 ppm, more preferably 10 ppm to 50 ppm, relative to the total weight of aqueous mixture L.
- Preferred preservatives are described under item 1 .4.5 above.
- aqueous mixture L in the context of the method according to the second aspect of the invention may comprise at least one benefit agent, preferably at least one encapsulate comprising a benefit agent, preferably a sensorial benefit agent.
- aqueous mixture L in the context of the method according to the second aspect of the invention comprises at least one benefit agent
- the total amount of benefit agent in aqueous mixture L is preferably between 0.1 ppm to 1000 ppm, preferably 1 ppm to 100 ppm, more preferably 10 ppm to 50 ppm relative to the total weight of aqueous mixture L.
- Preferred benefit agents are described under point 1 .4.6 above.
- aqueous mixture L in the context of the method according to the second aspect of the invention may comprise at least one polymer for use in detergents.
- the polymer may function as a co-builder as mentioned above, or may provide antiredeposition, fiber protection, soil release, dye transfer inhibition, viscosity modifiers, grease cleaning and/or antifoaming properties.
- the amount of all polymers in aqueous mixture L is preferably between 0.1 ppm to 1000 ppm, preferably 1 ppm to 100 ppm, more preferably 10 ppm to 50 ppm relative to the total weight of aqueous mixture L.
- aqueous mixture L in the context of the method according to the second aspect of the invention comprises at least one polymer selected from the group consisting of carboxymethylcellulose, polyethylenglycol, starch, carboxymethyl inulin, poly-aspartic acid, polyglutamic acid, sulfonated polycarboxylates, polyethylene oxide, polypropylene oxide, polyquaternium. These polymers are preferred as they are ecologically favorable.
- Preferred polyquaternium is polyquaternium 10, which is quaternized hydroxyethyl cellulose.
- the aqueous mixture L in the context of the method according to the second aspect of the invention may comprise at least one further additive.
- the further additive is preferably selected from the group set forth under item 1 .4.8 above.
- the at least one biosurfactant SBIO may be provided in the context of the method according to the second aspect of the invention in pure form or as a composition ZG, which may be a cleaning formulation.
- a preferred composition ZG is described under item 1 .5 above.
- step b. the aqueous mixture L is at least partially separated from the woolen article Aw-
- the separation is, in a preferred embodiment, carried out so that it is essentially complete.
- this step is carried out by withdrawing the article Aw from aqueous mixture L.
- the article Aw may be subjected to centrifugation.
- step c. of the method according to the second aspect of the invention the article Aw is rinsed with water.
- formulations 1 to 4 which are based on biosurfactants, were prepared and compared to benchmark formulations 1 to 4.
- Mono-rhamnolipid (“mono RL”) was prepared as described in example 2 of EP 3 061 442 A1 .
- the mono-rhamnolipid was employed as aqueous solution (75 wt.-%).
- Di-rhamnolipids (“diRL”) were prepared as described in example 1 of EP 3 061 442 A1.
- the di-rhamnolipid was employed as aqueous solution (50 wt.-%).
- composition of the benchmark formulations 1 to 4 were as follows:
- Benchmark 1 (Commercial product)
- Benchmark 2 (Commercial product)
- composition of the biosurfactant-based formulations 1 to 4 were as follows:
- the water used had a water hardness of 14 ° dGH, i.e. the concentration of earth alkali metal cations in the water was 2.5 mmol/l.
- a programmable Miele electronic household washing machine (“WCI 360 WTL; Power Wash 2.0”) was used. Fuzzy logic type control was disabled.
- the applied washing program is explained in the following table:
- the evaluation of the shrinking degree of the respective wool textiles was done by measuring the lengths and the width of the textile before and after the washing cycles.
- benchmark 3 and 4 differ from the formulations 1 to 4 only in the surfactants: benchmarks 3 and 4 comprise SLES or a mixture of SLES and a further surfactant, while formulations 1 to 4 are identical, except that they comprise rhamnolipids instead of the non- inventive surfactants.
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Epidemiology (AREA)
- Birds (AREA)
- Molecular Biology (AREA)
- Dermatology (AREA)
- Engineering & Computer Science (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Oil, Petroleum & Natural Gas (AREA)
- Wood Science & Technology (AREA)
- Organic Chemistry (AREA)
- Detergent Compositions (AREA)
Abstract
The present invention relates to a method of washing a woolen article AW. The method comprises a step in which AW is contacted with an aqueous mixture L comprising at least one biosurfactant SBio. The present invention furthermore relates to the use of at least one biosurfactant SBio to reduce shrinking of at least one woolen article AW during washing of AW, wherein, during washing of AW, AW is contacted with an aqueous mixture L comprising at least one biosurfactant SBio. The biosurfactant SBio is selected from the group consisting of rhamnolipids, glucolipids, sophorolipids. In particular, the biosurfactant SBio is selected from the group consisting of rhamnolipids, sophorolipids. Preferably, the biosurfactant SBio is selected from the group consisting of rhamnolipids.
Description
Biosurfactant for washing wool
The present invention relates to a method of washing a woolen article Aw- The method comprises a step in which Aw is contacted with an aqueous mixture L comprising at least one biosurfactant SBIO- The present invention furthermore relates to the use of at least one biosurfactant SBIO to reduce shrinking of at least one woolen article Aw during washing of Aw, wherein, during washing of Aw, Aw is contacted with an aqueous mixture L comprising at least one biosurfactant SBIO. The biosurfactant SBIO is selected from the group consisting of rhamnolipids, glucolipids, sophorolipids. In particular, the biosurfactant SBIO is selected from the group consisting of rhamnolipids, sophorolipids. Preferably, the biosurfactant SBIO is selected from the group consisting of rhamnolipids.
Background of the invention
Detergents and laundry compositions for washing wool or woolen articles are described in the art. CN 102 660 392 A discloses a detergent for woolen articles comprising certain non-ionic surfactants such as tea saponins.
CN 106 854 531 A discloses plant extracts from Angelica, Arnica, tea tree comprising tea saponines as surfactant in anti-shrinkage machine washing liquids for cashmere and woolen articles.
US 2021/0171864 A1 discloses the use of biosurfactants such as rhamnolipids and sophorolipids for improving the color stability (“crockfastness”) of dyed fabrics such as wool.
EP 0 439 019 A1 discloses the use of alkyl glycosides to prevent felting shrinkage during washing.
WO 2022/162043 A1 discloses rhamnolipids as possible surfactants for washing a wide range of textiles, among those wool. The examples of WO 2022/162043 A1 disclose the washing of cotton swatches with a rhamnolipid, but not with wool.
GB 1 515 792 A discusses specific problems in washing of wool, in particular with respect to felting and shrinking of wool during the wash. In order to solve this problem, it discloses a wool detergent comprising different surfactants, among these anionic surfactants such as sodium lauryl sulphate (“SLES”).
While the prior art discloses biosurfactants such as tea saponins, alkyl glycosides and other plant extracts which have a mild effect on woolen garments and reduce shrinking, these biosurfactants are also mild in their washing efficiency and cannot compete with well-established anionic
surfactants such as sodium laureth sulfate (“SLES”), sodium dodecylbenzensulfonate (“SDBS”), or non-ionic surfactants such as ethoxylated alcohols.
Hence, there still remains a need for detergents for wool washing which are superior in reducing the shrinking of woolen articles during the washing process. Simultaneously, this wool detergent should have a similar washing efficiency as the well-established anionic and non-ionic surfactants.
Short Summary of the Invention
It was now found that biosurfactants SBIO, which are selected from the group consisting of rhamnolipids, glucolipids, sophorolipids, and preferably are selected from the group of rhamnolipids, reduce the shrinking of woolen articles during washing.
In a first aspect, the present invention hence relates to use of at least one biosurfactant SBIO, which is selected from the group consisting of rhamnolipids, glucolipids, sophorolipids, and in particular is selected from the group consisting of rhamnolipids, sophorolipids, and preferably is selected from the group consisting of rhamnolipids, for washing at least one woolen article Aw- During washing of Aw, Aw is contacted with an aqueous mixture L. This aqueous mixture L comprises the at least one biosurfactant SBIO-
In a second aspect, the present invention relates to a method of washing at least one woolen article Aw, comprising the steps of a. contacting the at least one woolen article Aw with an aqueous mixture L comprising at least one biosurfactant SBIO, wherein the at least one biosurfactant SBIO is selected from the group consisting of rhamnolipids, glucolipids, sophorolipids, and in particular is selected from the group consisting of rhamnolipids, sophorolipids, and preferably is selected from the group consisting of rhamnolipids, b. at least partially separating L from Aw, c. and optionally rinsing Aw with water.
In the method according to the second aspect of the invention, the temperature of the aqueous mixture L, for at least as long as the aqueous mixture L is contacted with the at least one woolen article Aw, is in a range of from > 0 °C to 45 °C, in particular in a range of from 1 °C to 40 °C, preferably in a range of from 5 °C to 35 °C, more preferably in a range of from 10 °C to 30 °C, more preferably in a range of from 15 °C to 30 °C, even more preferably in a range of from 20 °C to 30 °C, even more preferably in a range of from 25 °C to 30 °C.
In a further aspect of the present invention, the use according to the first aspect of the invention is carried out in a method according to the second aspect of the present invention.
Detailed Description of the Invention
1 . Use of at least one biosurfactant SBIO to reduce shrinking of at least one woolen article Aw during washing of at least one woolen article Aw
In a first aspect, the present invention relates to the use of at least one biosurfactant SBIO to reduce shrinking of at least one woolen article Aw during washing of the at least one a woolen article Aw- During washing of Aw, Aw is contacted with an agueous mixture L comprising at least one biosurfactant SBIO.
The biosurfactant SBIO is selected from the group consisting of rhamnolipids, glucolipids, sophorolipids. The biosurfactant SBIO, in particular, is selected from the group consisting of rhamnolipids, sophorolipids. The biosurfactant SBIO is preferably selected from rhamnolipids.
In a preferred embodiment of the use according to the first aspect of the present invention, the at least one biosurfactant SBIO is used in an agueous mixture L to reduce shrinking of the at least one woolen article Aw during washing of Aw in the agueous mixture L. The agueous mixture L hence comprises the at least one biosurfactant L and the at least one woolen article Aw-
1.1 Woolen Article Aw
In the context of the use according to the first aspect of the invention, a “woolen article Aw” is to be understood as any article which comprises wool, preferably an article comprising at least 10 weight-% (= “wt.-%”) wool, more preferably at least 20 wt.-% wool, more preferably at least 30 wt.- % wool, more preferably at least 40 wt.-% wool, more preferably at least 50 wt.-% wool, more preferably at least 60 wt.-% wool, more preferably at least 70 wt.-% wool, more preferably at least 80 wt.-% wool, more preferably at least 90 wt.-% wool, more preferably at least 99 wt.-% wool, wherein the weight-% relate to the total mass of the woolen article. Most preferably, the article Aw essentially consists of wool.
The woolen article Aw in particular is an article of clothing, preferably selected from pants, shirt, socks, hosiery, scarfs, gloves, shoes, sweaters, suits, hats, caps, bonnets. It may, in addition, also be selected from the group consisting of curtains, napkins, bed linen, tablecloth, blanket, bedspread, duvet cover.
In the context of this invention, “wool” refers to the textile fiber of a mammal, in particular of livestock. In particular, it refers to the textile fiber of a mammal selected from the group consisting of sheep, goat (the goat being in particular selected from the group of cashmere goat, angora goat), rabbit (the rabbit being in particular an angora rabbit), camelide (the camelide being in
particular selected from the group of llama, vikuna, alpaca, guanaco), yak, beaver, otter, guinea pig, horse, donkey, chinchilla. Preferably, it refers to the textile fiber of a mammal selected from the group consisting of sheep, goat, most preferably of sheep.
It was now surprisingly found that the use of at least one biosurfactant SBIO according to the first aspect of the invention reduces shrinking of the at least one woolen article Aw during washing in the aqueous mixture L comprising the at least one woolen article Aw and the at least one biosurfactant SBIO-
1.2 Shrinking and Assay X
Shrinking or shrinkage of woolen fabrics is a natural phenomenon and occurs during washing. Fabric shrinkage is the reduction in the size or dimensions of a fabric or garment after being exposed to heat or undergoing washing. Shrinkage can occur in both lengthwise (warp) and widthwise (weft) directions. Woolen articles such as woolen fabrics are particularly prone to shrinkage during washing, in particular when elevated temperatures (above 20 °C) are applied and when the woolen article is moved in the aqueous mixture in which it is washed (i.e. the washing mixture) during washing, which is typically the case when agitation is applied during washing.
Woolen articles, in particular, are washed at low temperatures to not strain the woolen fibers. Hence, it is preferred that the temperature of the washing mixture does not exceed 45 °C.
“Reduce shrinking of at least one woolen article Aw during washing of Aw” means that the extent of shrinking of Aw in an aqueous mixture L comprising the at least one biosurfactant SBIO is reduced during washing, compared to the case where Aw is washed under the same conditions (for example under the same temperature, washing program, pressure) in an aqueous mixture Lo, wherein Lo and L are identical, except that Lo comprises water instead of the at least one biosurfactant SBIO- Hence, from a formal point of view, Lo is obtained by substituting the mass of all biosurfactants SBIO comprised by L by the same mass of water.
“Reduce shrinking of at least one woolen article Aw during washing of Aw” is therefore to be understood in the sense that Aw is at least partially protected against shrinking during washing when at least one biosurfactant SBIO is comprised by the aqueous mixture L which contacts, preferably comprises, the at least one woolen article Aw during washing. In other words, it means that at least a partial resilience against shrinking is conferred to the woolen article Aw during washing when at least one biosurfactant SBIO is comprised by the aqueous mixture L which contacts, preferably comprises, the at least one woolen article Aw during washing.
1.2. 1.1 ...compared with washing in water
It was surprisingly found that this reduction of shrinking (i.e. protection or resilience against shrinking) conferred by the at least one biosurfactant SBIO in the context of the present invention is higher than the reduction of shrinking conferred by water alone.
For example, it means that the extent of shrinking of the at least one woolen article Aw during washing of Aw in an aqueous mixture L with a given mass mi. (for example, mi. = 1 kg or mi. = 55 kg) comprising at least one biosurfactant SBIO, is reduced compared to the extent of shrinking of the at least one woolen article Aw during washing of Aw under the same conditions in pure water, wherein the water used for washing has the same mass mi. as the aqueous mixture L, but does not comprise any biosurfactant SBIO.
1.2. 1.2 ...compared with washing with other surfactants
Additionally, and even more surprisingly, this “reduction of shrinking” is improved not only compared to washing in pure water, but also in comparison with the shrinking during washing in aqueous mixtures comprising other surfactants.
In particular, it was found that the reduction of shrinking during washing with at least one biosurfactant SBIO in an aqueous mixture L is higher than the reduction of shrinking during washing with at least one surfactant SY in an aqueous mixture Ly. Formally, Ly is obtained from aqueous mixture L by substituting the mass of all biosurfactants SBIO comprised by L by the same mass of at least one surfactant Sy.
Hence, when one compares two cases (1) and (2), the shrinking of at least one woolen article Aw in case (1) is less than the shrinking of the same woolen article Aw in case (2):
(1) washing of Aw in an aqueous mixture L with a given mass mi. (for example, mi = 1 kg or mi. =55 kg) comprising at least one biosurfactant SBIO in an amount of XL wt.-% (relative to the total mass of mixture L);
(2) washing of Aw in an aqueous mixture Ly under the same conditions as in case (1), (in particular under the same temperature, pressure). Aqueous mixture LY comprises at least one surfactant Sy in the same amount (relative to the total weight of the aqueous mixture Ly) as the aqueous mixture L comprises the at least one biosurfactant SBIO (relative to the total weight of the aqueous mixture L). Aqueous mixture Ly does not comprise a biosurfactant SBIO-
Hence, LY has the same mass mi. as the aqueous mixture L and does not comprise any biosurfactant SBIO- Instead of biosurfactants SBIO, LY comprises XL wt.-% (relative to the total mass of mixture LY) of at least one surfactant SY.
SY is different from SBIO and is in particular a surfactant with a comparable washing profile and efficiency. Preferably, SY is selected from the group consisting of SLES, ethoxylated alcohols. More preferably SY is selected from the group consisting of SLES, a mixture of SLES and ethoxylated alcohols.
1 .2.2 Tests to determine the reduction of shrinking
Whether a certain woolen article Aw shrinks, as well as the extent of shrinking of a certain woolen article Aw during washing may be determined by methods that are known to the skilled person. Fabric shrinkage tests are typically performed according to recognized industry standards.
Some commonly used standards for fabric shrinkage testing that may be applied in the context of the present invention, including:
ASTM D3774: Standard Test Method for Width of Textile Fabric
ASTM D4970: Standard Test Method for Pilling Resistance and Other Related Surface Changes of Textile Fabrics
ISO 6330: Textiles - Domestic washing and drying procedures for textile testing
AATCC TM135: Dimensional Changes in Automatic Home Laundering of Woven and Knit Fabrics.
Further tests are described on the website https://www.testextextile.com/fabric-shrinkage-test-a- complete-guide/#What_is_the_Fabric_Shrinkage_Test_%E2%80%93_Definition (last retrieved 12 July 2024).
In the context of the present invention, the preferred test to determine whether a certain substance (abbreviated as “X”) reduces the shrinking of a woolen article Aw (as well as the extent of shrinking) during washing is Assay X as explained in the following.
Assay X is the preferred test to determine the reduction of shrinking according to the present invention.
Assay X is used, in particular, to determine the reduction of shrinking of the at least one woolen article Aw during washing in an aqueous mixture L, as well as the extent of shrinking of the at least one woolen article Aw during washing in an aqueous mixture L.
Assay X is hence the preferred test to determine, when the at least one biosurfactant SBIO is used according to the first aspect of the present invention, the reduction of shrinking of the at least one woolen article Aw during washing, in particular in an aqueous mixture L, as well as the extent of shrinking of the at least one woolen article Aw during washing, in particular in an aqueous mixture L.
1 .2.3 Assay X
Assay X is preferably used according to the present invention to assess whether a given additive X reduces shrinking of a woolen article Aw- The additive to be tested is hereinafter abbreviated as “X”. Additive X may be a biosurfactant SBIO which may be employed in Assay X directly or as a mixture, in particular an aqueous mixture. Additive X may, alternatively, be a surfactant SY, wherein SY is different from the biosurfactant SBIO- Likewise, additive X may be, in a further alternative, any other compound or mixture of compounds, which is to be investigated in terms of their efficiency to reduce shrinking of a woolen article.
Additive X may be a pure compound or a mixture of compounds (such as an aqueous mixture).
Assay X is divided into two parts, part 1 and part 2.
First, in part 1 , a blank test is carried out, in which the shrinking of a model woolen fabric is tested. For this blank test, distilled water is used as “blank” detergent formulation. After the model washing cycle, the shrinking of the model woolen fabric is measured. In the model washing cycle [step (3) in part 1 and part 2, respectively], the water that is used has the same water hardness of 14 ° dGH or less. Preferably, distilled water is used in part 1 and part 2.
Second, in part 2, the shrinking of the model woolen fabric is tested, wherein the same conditions of the model washing cycle as in part 1 are applied, but the additive X is tested. The additive X may be a pure compound or an aqueous mixture, preferably is an aqueous mixture, even more preferably is an aqueous mixture in which the additive X has a concentration of 1 weight-% to 50 weight-%, preferably 2 weight-% to 30 weight-%, more preferably 3 weight-% to 20 weight-%, even more preferably 4 weight-% to 15 weight-%, even more preferably 5 weight-% to 15 weight-%, even more preferably 6 weight-% to 15 weight-%, even more preferably 8 to 15 weight-%, even more preferably 10 to 15 weight-% even more preferably 12 to 15 weight-% (in each case relative to the total weight of the aqueous mixture).
In part 1 and part 2, the testing wool textiles are standard white unsoiled fabrics provided by wfk-Testgewebe GmbH (“WFK 60 A”, order code 60000; 100 % wool fabric). The test swatches have a length of 75 cm and a width of 40 cm. Their original area is hence Ao = (75 * 40) cm2 = 3000 cm2. In each test series, five of these standard wool textiles (abbreviated hereinafter as “test
swatches") are used and the area before (“Ao”) and the area after (in case of part 1 : “Asiank”; in case of part 2: “Ax”) the test run are measured to assess the extent of shrinking.
Ao is the average area of the five standard wool textile swatches that is measured and calculated before the test procedure in part 1 or part 2. Since standard wool textiles swatches of the same size are used in part 1 and part 2, Ao in part 1 and Ao in part 2 are the same.
Aeiank is the average area of the five standard wool textile swatches that is measured and calculated after the test procedure of part 1 .
Ax is the average area of the five standard wool textile swatches that is measured and calculated after the test procedure of part 2.
The extent of shrinking in the blank test (i.e. part 1), Ssiank, is then calculated as follows: SBIank = (Ao - Asiank) I Ao.
The extent of shrinking in the test of X (i.e. part 2), Sx, is then calculated as follows: Sx = (Ao - Ax) I Ao.
X is deemed to reduce shrinking during washing, if Sx < Ssiank-
Likewise, the reduction of shrinking obtained for two different additives Xi and X2 may also be compared as follows:
Assay X is carried out for Xi and X2.
The extent of shrinking in the test of Xi (i.e. part 2), Sxi, is then calculated as follows (Ax measured and calculated for X = Xi is abbreviated as “Axi”):
Sxi = (Ao - Axi) / Ao.
The extent of shrinking in the test of X2 (i.e. part 2), Sx2, is then calculated as follows (Ax measured and calculated for X = X2 is abbreviated as “Ax2”):
Sx2 = (Ao - AX2) / Ao.
Xi is deemed to be superior to X2 in reducing shrinking during washing if Sxi < Sx2-
X2 is deemed to be superior to Xi in reducing shrinking during washing if Sx2 < Sxi.
X2 and Xi are deemed to be equally effective in reducing shrinking during washing if Sxi = Sx2-
Part 1
Blank test
(1) Five test swatches with a given area (the average area of the five test swatches is measured and calculated as “Ao”) are added together with 2 kg of cotton ballast load to a Miele laundry machine (“WCI 360 WTL; Power Wash 2.0”). The cotton ballast load is prepared beforehand by washing it at 90 °C with ECE2 detergent without bleach.
(2) 50 g of distilled water are added into the dosing drawer.
(4) The average area of the five test swatches after the washing is measured and calculated aS Aeiank ■
(5) The extent of shrinking in the blank test (i.e. part 1), Ssiank, is then calculated as follows: SBIank = (Ao - Asiank) I Ao.
Part 2
(1) Five test swatches with a given area (the average area of the five test swatches is measured and calculated as “Ao”) are added together with 2 kg of cotton ballast load to a Miele laundry machine (“WCI 360 WTL; Power Wash 2.0”). The cotton ballast load is prepared beforehand by washing it at 90 °C with ECE2 detergent without bleach.
(2) 50 g of additive X is added into the dosing drawer. X may be added as the compound or as an aqueous mixture, for example, an aqueous mixture that contains the additive X in a concentration of 1 weight-% to 50 weight-%, preferably 2 weight-% to 30 weight-%, more preferably 3 weight-% to 20 weight-%, even more preferably 4 weight-% to 15 weight-%, even more preferably 5 weight-% to 15 weight-%, even more preferably 6 weight-% to 15 weight-%, even more preferably 8 to 15 weight-%, even more preferably 10 to 15 weight-% even more preferably 12 to 15 weight-% (in each case relative to the total weight of aqueous mixture). For comparison reasons, X is most preferably added as a 15 weight-% aqueous mixture of X, in particular where a biosurfactant SBIO is tested in Assay X.
(4) The average area of the five test swatches after the washing is measured and calculated as Ax .
(5) The extent of shrinking in the test of X (i.e. part 2), Sx, is then calculated as follows: Sx = (Ao - Ax) I Ao.
X is deemed to reduce shrinking during washing if Sx < Seiank-
1.3 Biosurfactant SBIO
In the context of the use according to the first aspect of the invention, the “biosurfactant SBIO” is selected from the group consisting of glucolipids, rhamnolipids, sophorolipids, in particular selected from the group of sophorolipids, rhamnolipids, preferably from the group of rhamnolipids.
Glucolipids, rhamnolipids, and sophorolipids are glycolipids. Such glycolipids are described in the art together with their syntheses, for example in EP 0499434 A1 (glucolipids are referred to as “glucose lipids” in this document), DE 196 48 439 A1 , DE 196 00 743 A1 . WO 03/006146 A1 , US 2008/0213194 A1 , JP H01 -304034 A1 , CN 1337439 A describe further methods for the synthesis of rhamnolipids. WO 03/002700 A1 , US 4,305,961 A, US 7,556,654 B1 describe further methods for the synthesis of sophorolipids.
1.3.1 Glucolipids
In the context of the use according to the first aspect of the present invention, the term “glucolipid” is, in particular, to be understood as referring to a structure according to formula (I), even more preferred a structure according to formula (II):
In formulae (I) and (II), mGL = 3, 2, 1 or 0, preferably 1 or 0.
Residues R1GL and R2GL are, independently of one another, an organic radical having 2 to 24 carbon atoms, preferably 5 to 20, more preferably 7 to 15, even more preferably 7, carbon atoms, wherein preferably R1GL and R2GL are independently of one another selected from the group consisting of optionally substituted alkyl radicals with 2 to 24 preferably 5 to 20, more preferably 7 to 15, even more preferably 7, carbon atoms, wherein hydroxy substituted alkyl radicals are preferred substituted alkyl radicals, optionally substituted alkenyl radicals with 2 to 24 carbon atoms, preferably 5 to 20, more preferably 7 to 15, even more preferably 7, carbon atoms, wherein hydroxy substituted alkenyl radicals are preferred substituted alkenyl radicals, wherein more preferably R1GL and R2GL are independently of one another selected from the group consisting of pentenyl, heptenyl, nonenyl, undecenyl, tridecenyl and -(CH2)oCH3 where o = 1 to 23, preferably 4 to 12. Most preferably, R1GL and R2GL are each n-heptyl.
Distinct glucolipids for mGL = 0 are abbreviated according to the following nomenclature: “GL-CX” is understood as meaning glucolipids of the general formulae (I) or (II) in which mGL = 0 and in which the radical R1GL = (CH2)o-CH3 where o = X-4.
Distinct glucolipids for mGL = 1 are abbreviated according to the following nomenclature: “GL-CXCY” is understood as meaning glucolipids of the general formulae (I) or (II) in which mGL = 1 and in which one of the radicals R1GL and R2GL = (CH2)o-CH3 where o = X-4 and the remaining radical R1GL or R2GL = (CH2)o-CH3 where o = Y-4.
The nomenclature used thus does not differentiate between “CXCY” and “CYCX”.
If one of the aforementioned indices X and/or Y is provided with “:Z”, then this means that the respective radical R1GL and/or R2GL = an unbranched, unsubstituted hydrocarbon radical with X-3 or Y-3 carbon atoms having Z double bonds.
The curvy bond in structure (I) [and also structures (III), (V), (VII), (IX), (XI) described hereinafter] implies that the respective substituent may be axial or equatorial, preferably is equatorial.
Alkyl radicals may be branched or linear.
Alkenyl radicals may be branched or linear and preferably contain between one to three double bonds.
In those cases where structures according to formulae (I) and (II) comprise more than one residue R2GL, these residues R2GL may be identical or different from one another.
In those embodiments in which glucolipids are used as biosurfactants SBIO according to the first aspect of the invention, it is preferred that 1 wt.-% to 30 wt.-%, preferably 5 wt.-% to 25 wt.-%, particularly preferably 10 wt.-% to 20 wt.-%, of all glucolipids that are used as biosurfactants SBIO according to the first aspect of the invention, are GL-C8C10, where the “wt.-%” (= weight-%; percentages by weight) refers to the total weight of all the glucolipids that are used as biosurfactants SBIO according to the first aspect of the invention.
In those embodiments in which glucolipids are used as biosurfactants SBIO according to the first aspect of the invention, it is alternatively preferred that 0.5 wt.-% to 20 wt.-%, preferably 3 wt.-% to 17 wt.-%, particularly preferably 5 wt.-% to 15 wt-%. of all glucolipids that are used as biosurfactants SBIO according to the first aspect of the invention, are GL-C10C12:1 , where the wt.-% refers to the total weight of all the glucolipids that are used as biosurfactants SBIO according to the first aspect of the invention.
In those embodiments in which glucolipids are used as biosurfactants SBIO according to the first aspect of the invention, it is alternatively preferred that 0.5 wt.-% to 20 wt.-%, preferably 2 wt.-% to 15 wt.-%, particularly preferably 3 wt.-% to 12 wt.-%, of all the glucolipids that are used as biosurfactants SBIO according to the first aspect of the invention, are GL-C10C12, where the wt.-% refers to the total weight of all the glucolipids that are used as biosurfactants SBIO according to the first aspect of the invention.
In those embodiments in which glucolipids are used as biosurfactants SBIO according to the first aspect of the invention, it is alternatively preferred that
1 wt.-% to 30 wt.-%, preferably 5 wt.-% to 25 wt.-%, particularly preferably 10 wt.-% to 20 wt.-%, of all the glucolipids that are used as biosurfactants SBIO according to the first aspect of the invention, are GL-C8C10,
0.5 wt.-% to 20 wt.-%, preferably 3 wt.-% to 17 wt.-%, particularly preferably 5 wt.-% to 15 wt.-%, of all the glucolipids that are used as biosurfactants SBIO according to the first aspect of the invention, are GL-C10C12:1 ,
0.5 wt.-% to 20 wt.-%, preferably 2 wt.-% to 15 wt.-%, particularly preferably 3 wt.-% to 12 wt.-%, of all the glucolipids that are used as biosurfactants SBIO according to the first aspect of the invention, are GL-C10C12,
where the wt.-% refers to the total weight of all the glucolipids that are used as biosurfactants SBIO according to the first aspect of the invention.
In those embodiments in which glucolipids are used as biosurfactants SBIO according to the first aspect of the invention, it is alternatively preferred that 10 wt.-% to 20 wt.-% of all the glucolipids that are used as biosurfactants SBIO according to the first aspect of the invention, are GL-C8C10, 5 wt.-% to 15 wt.-%, of all the glucolipids that are used as biosurfactants SBIO according to the first aspect of the invention, are GL-C10C12:1 ,
3 wt.-% to 12 wt.-%, of all the glucolipids that are used as biosurfactants SBIO according to the first aspect of the invention, are GL-C10C12, where the wt.-% refers to the total weight of all the glucolipids that are used as biosurfactants SBIO according to the first aspect of the invention.
Over and above this, in those embodiments in which glucolipids are used as biosurfactants SBIO according to the first aspect of the invention, it is alternatively preferred that 0 wt.-% to 5 wt.-%, preferably 0.01 wt.-% by weight to 4 wt.-%, particularly preferably 0.1 wt.-% to 3 wt.-%, of all the glucolipids that are used as biosurfactants SBIO according to the first aspect of the invention, are GL-C10, where the wt.-% refers to the total weight of all the glucolipids that are used as biosurfactants SBIO according to the first aspect of the invention.
1 .3.2 Rhamnolipids
In the context of the use according to the first aspect of the present invention, the term "rhamnolipid" is, in particular, to be understood as referring to compounds of the general formula (III) and salts thereof, preferably compounds according to the general structure (IV) and salts thereof,
In formulae (III) and (IV), mRL = 2, 1 or 0, preferably 1 or 0.
nRL = 1 or 0.
Residue R1RL and R2RL are independently of one another, an organic radical having 2 to 24 carbon, preferably 5 to 13, more preferably 7 to 10, even more preferably 7, carbon atoms, wherein preferably R1RL and R2RL are independently of one another selected from the group consisting of optionally substituted alkyl radicals with 2 to 24, preferably 5 to 13, more preferably 7 to 10, even more preferably 7, carbon atoms, wherein hydroxy substituted alkyl radicals are preferred substituted alkyl radicals, optionally substituted alkenyl radicals with 2 to 24, preferably 5 to 13, more preferably 7 to 10, even more preferably 7, carbon atoms, wherein hydroxy substituted alkenyl radicals are preferred substituted alkenyl radicals, wherein more preferably R1 RL and R2RL are independently of one another selected from the group consisting of pentenyl, heptenyl, nonenyl, undecenyl, tridecenyl and -(CH2)o-CH3 where o = 1 to 23, preferably 4 to 12. Most preferably, R1RL and R2RL are each n-heptyl.
If nRL = 1 , the glycosidic bond between the two rhamnose units is preferably in the a-configuration. The optically active carbon atoms of the fatty acids are preferably present as R-enantiomers (e.g. (R)-3-{(R)-3-[2-O-(a-L-rhamnopyranosyl)-a-L-rhamnopyranosyl]oxydecanoyl}oxydecanoate).
The term "di-rhamnolipid" in the context of the present invention is understood to mean compounds of the general formulae (III) and (IV) or salts thereof, where nRL = 1 .
The term "mono-rhamnolipid" in the context of the present invention is understood to mean compounds of the general formulae (III) and (IV) or salts thereof, where nRL = 0.
Distinct rhamnolipids are abbreviated according to the following nomenclature:
"diRL-CXCY" are understood to mean di-rhamnolipids of the general formulae (III) and (IV), in which one of the residues R1RL and R2RL = -(CH2)o-CH3 where o = X-4 and the remaining residue R1 or R2 = -(CH2)O-CH3 where o = Y-4.
"monoRL-CXCY" are understood to mean mono-rhamnolipids of the general formulae (III) and (IV), in which one of the residues R1RL and R2RL = -(CH2)o-CH3 where o = X-4 and the remaining residue R1 RL o r R2RL = _(CH2)o-CH3 where o = Y-4.
The nomenclature used therefore does not distinguish between "CXCY" and "CYCX".
For rhamnolipids where mRL=0, the terms “monoRL-CX” or “diRL-CX” are used accordingly.
If one of the abovementioned indices X and/or Y is provided with ":Z", this signifies that the respective residue R1RL and/or R2RL is equal to an unbranched, unsubstituted hydrocarbon residue having X-3 or Y-3 carbon atoms having Z double bonds.
Methods for preparing the relevant rhamnolipids are disclosed, for example, in EP 2 786 743 A1 and EP 2 787 065 A1 . Rhamnolipids applicable in the context of the instant invention can also be produced by fermentation of Pseudomonas, especially Pseudomonas aeruginosa, which are
preferably non genetically modified cells, a technology already disclosed in the eighties, as documented e.g. in EP 0282 942 A1 and DE 41 27 908 A1 . Rhamnolipids produced in Pseudomonas aeruginosa cells which have been improved for higher rhamnolipid titres by genetical modification can also be used in the context of the instant invention; such cells have for example been disclosed by Lei et al. in Biotechnol Lett. 2020 Jun;42(6):997-1002.
Rhamnolipids produced by Pseudomonas aeruginosa are commercially available from Jeneil Biotech Inc., e.g. under the tradename Zonix, from Logos Technologies (technology acquired by Stepan), e.g. under the tradename NatSurFact, from Biotensidion GmbH, e.g. under the tradename Rhapynal, from AGAE technologies, e.g. under the name R90, R95, R95Md, R95Dd, from Locus Bio-Energy Solutions and from Shanghai Yusheng Industry Co. Ltd., e.g. under the tradename Bio- 201 Glycolipids.
In those embodiments in which rhamnolipids are used as biosurfactants SBIO according to the first aspect of the invention, it is alternatively preferred to use mixtures of mono-rhamnolipids and dirhamnolipids, wherein even more preferably the ratio of the weight of all mono-rhamnolipids used according to the first aspect of the invention to the weight of all di-rhamnolipids used according to the first aspect of the invention is in the range of from 9 : 1 to 1 : 9, preferably of from 8 : 2 to 2 : 8, more preferably of from 7 : 3 to 3 : 7, more preferred of from 6 : 4 to 4 : 6, most preferred is 1 : 1 .
Mono-rhamnolipids are compounds according to structures (III), preferably (IV), wherein nRL = 0. Di-rhamnolipids are compounds according to structures (III), preferably (IV), wherein nRL = 1 .
In those embodiments in which rhamnolipids are used as biosurfactants SBIO according to the first aspect of the invention, it is preferred that the rhamnolipids comprise 56 wt.-% to 95 wt.-%, preferably 60 wt.-% to 80 wt.-%, particularly preferably 66 wt.-% to 70 wt.-%, of diRL-C10C10, where the wt.-% refers to the total weight of all the rhamnolipids that are used as biosurfactants SBIO according to the first aspect of the invention.
In those embodiments in which rhamnolipids are used as biosurfactants SBIO according to the first aspect of the invention, it is alternatively preferred that 0.5 wt.-% to 15 wt.-%, preferably 3 wt.-% to 12 wt.-%, particularly preferably 5 wt.-% to 10 wt.-%, of the rhamnolipids are diRL-C10C12:1 , where the wt.-% refers to the total weight of all the rhamnolipids that are used as biosurfactants SBIO according to the first aspect of the invention.
In those embodiments in which rhamnolipids are used as biosurfactants SBIO according to the first aspect of the invention, it is alternatively preferred that 0.5 wt.-% to 25 wt.-%, preferably 3 wt.-% to 15 wt.-%, particularly preferably 5 wt.-% to 12 wt.-%, of the rhamnolipids are diRL-C10C12, where the wt.-% refers to the total weight of all the rhamnolipids that are used as biosurfactants SBIO according to the first aspect of the invention.
In those embodiments in which rhamnolipids are used as biosurfactants SBIO according to the first aspect of the invention, it is alternatively preferred that 0.1 wt.-% to 25 wt.-%, preferably 2 wt.-% to 10 wt.-%, particularly preferably 4 wt.-% to 8 wt.-%, of the rhamnolipids are diRL-C8C10, where the wt.-% refers to the total weight of all the rhamnolipids that are used as biosurfactants SBIO according to the first aspect of the invention.
In those embodiments in which rhamnolipids are used as biosurfactants SBIO according to the first aspect of the invention, it is even further preferred that
0.1 wt.-% to 5 wt.-%, preferably 0.5 wt.-% to 3 wt.-%, particularly preferably 0.5 wt.-% to 2 wt.-%, of the rhamnolipids are monoRL-C8C10 and/or, preferably and,
0.1 wt.-% to 5 wt.-%, preferably 0.5 wt.-% to 3 wt.-%, particularly preferably 0.5 wt.-% to 2 wt.-%, of the rhamnolipids are monoRL-C10C10, where the wt.-% refers to the total weight of all the rhamnolipids that are used as biosurfactants SBIO according to the first aspect of the invention.
In those embodiments in which rhamnolipids are used as biosurfactants SBIO according to the first aspect of the invention, it is alternatively preferred that 10 wt.-% to 30 wt.-%, preferably 20 wt.-% to 30 wt.-%, particularly preferably 25 wt.-% to 30 wt.-%, of the rhamnolipids are monoRL-C10C10, where the wt.-% refers to the total weight of all the rhamnolipids that are used as biosurfactants SBIO according to the first aspect of the invention.
In those embodiments in which rhamnolipids are used as biosurfactants SBIO according to the first aspect of the invention, it is alternatively preferred that 10 wt.-% to 30wt.-%, preferably 12 wt.-% to 25 wt.-%, particularly preferably 15 wt.-% to 20 wt.-%, of the rhamnolipids are diRL-C10C10, where the wt.-% refers to the total weight of all the rhamnolipids that are used as biosurfactants SBIO according to the first aspect of the invention.
In those embodiments in which rhamnolipids are used as biosurfactants SBIO according to the first aspect of the invention, it is alternatively preferred that 10 wt.-% to 30 wt.-%, preferably 12 wt.-% to 25 wt.-%, particularly preferably 15 wt.-% to 20 wt.-%, of the rhamnolipids are monoRL-C8C10, where the wt.-% refers to the total weight of all the rhamnolipids that are used as biosurfactants SBIO according to the first aspect of the invention.
In those embodiments in which rhamnolipids are used as biosurfactants SBIO according to the first aspect of the invention, it is alternatively preferred that 3 wt.-% to 25 wt.-%, preferably 5 wt.-% to 20 wt.-%, particularly preferably 10 wt.-% to 15 wt.-%, of the rhamnolipids are monoRL-C10C12:1 , where the wt.-% refers to the total weight of all the rhamnolipids that are used as biosurfactants SBIO according to the first aspect of the invention.
In those embodiments in which rhamnolipids are used as biosurfactants SBIO according to the first aspect of the invention, it is alternatively preferred that 1 wt.-% to 15 wt.-%, preferably 2 wt.-% to 10 wt.-%, particularly preferably 3 wt.-% to 8 wt.-%, of the rhamnolipids are diRL-C10C12, where the wt.-% refers to the total weight of all the rhamnolipids that are used as biosurfactants SBIO according to the first aspect of the invention.
1 .3.3 Sophorolipids
In the context of the use according to the first aspect of the present invention, the term “sophorolipids” is in particular to be understood as referring to compounds of the general formulae (V), (VII) and salts thereof, preferably compounds of the general formulae (VI), (VIII) and salts thereof:
Formulae (V), (VII) represent the acid form, Formulae (VI), (VIII) represent the lactone form.
In formulae (V) and (VI), nSL = 1 or 0.
In formulae (V), (VI), (VII), and (VIII),
R1SL = H or -CO-CH3,
R2SL = H or -CO-CH3,
R3SL = a divalent organic moiety which comprises 6 to 32 carbon atoms, preferably 12 to 20, more preferably 14 to 16, most preferably 15.
R3SI_ preferably is an optionally substituted, divalent hydrocarbon moiety comprising 6 to 32 carbon atoms, wherein hydroxy substituted hydrocarbon moieties are preferred as substituted hydrocarbon moi eties,
R3SI_ more preferably is selected from the group consisting of
• optionally substituted alkylene radicals with 6 to 32, preferably 12 to 20, more preferably 14 to 16, most preferably 15 carbon atoms, wherein hydroxy-substituted alkylene radicals are preferred substituted alkylene radicals, and wherein it is preferred that the optionally substituted alkylene radicals are unbranched,
• optionally substituted alkenylene radicals with 6 to 32, preferably 12 to 20, more preferably 14 to 16, most preferably 15 carbon atoms, wherein hydroxy-substituted alkenylene radicals are preferred substituted alkenylene radicals, and wherein it is preferred that the optionally substituted alkenylene radicals are unbranched, and wherein it is preferred that the optionally substituted alkenylene radicals comprise one to three double or triple bonds.
R3SL even more preferably is selected from the group consisting of
• unbranched or branched, preferably unbranched, alkylene radicals with 6 to 32, preferably 12 to 20, more preferably 14 to 16, most preferably 15, carbon atoms,
• unbranched or branched, preferably unbranched, alkylene radicals with 6 to 32 preferably 12 to 20, more preferably 14 to 16, most preferably 15, carbon atoms carrying at least one hydroxy group, preferably one hydroxy group,
• unbranched or branched, preferably unbranched, alkenylene radicals with 6 to 32, preferably 12 to 20, more preferably 14 to 16, most preferably 15 carbon atoms, wherein the alkenylene radicals preferably comprise one to three double or triple bonds, wherein the alkenylene radicals more preferably comprise one to three double bonds, even more preferably one double bond,
• unbranched or branched, preferably unbranched, alkenylene radicals with 6 to 32, preferably 12 to 20, more preferably 14 to 16, most preferably 15, carbon atoms carrying at least one hydroxy group, preferably one hydroxy group, wherein the
alkenylene radicals preferably comprise one to three double or triple bonds, wherein the alkenylene radicals more preferably comprise one to three double bonds, even more preferably one double bond.
R4SL = H, CH3 or a monovalent organic radical which comprises 2 to 10 carbon atoms.
R4SL is preferably selected from the group consisting of
• H,
• CH3,
• optionally substituted alkyl radicals with 2 to 10 carbon atoms, wherein hydroxy-substituted alkyl radicals are preferred substituted alkyl radicals, and wherein it is preferred that the optionally substituted alkyl radicals are unbranched,
• optionally substituted alkenyl radicals with 2 to 10 carbon atoms, wherein hydroxysubstituted alkenyl radicals are preferred substituted alkenyl radicals, and wherein it is preferred that the optionally substituted alkenyl radicals are unbranched, and wherein it is preferred that the optionally substituted alkenyl radicals comprise one to three double or triple bonds.
R4SL is more preferably selected from the group consisting of
• H,
• CH3,
• unbranched or branched, preferably unbranched, alkyl radicals with 2 to 10 carbon atoms,
• unbranched or branched, preferably unbranched, alkyl radicals with 2 to 10 carbon atoms carrying at least one hydroxy group, preferably one hydroxy group,
• unbranched or branched, preferably unbranched, alkenyl radicals with 2 to 10 carbon atoms, wherein the alkenyl radicals preferably comprise one to three double or triple bonds, wherein the alkenyl radicals more preferably comprise one to three double bonds, even more preferably one double bond,
• unbranched or branched, preferably unbranched, alkenyl radicals with 2 to 10 carbon atoms carrying at least one hydroxy group, preferably one hydroxy group, wherein the alkenyl radicals preferably comprise one to three double or triple bonds, wherein the alkenyl radicals more preferably comprise one to three double bonds, even more preferably one double bond.
R4SL is most preferably selected from the group consisting of H, Methyl, Ethyl.
In an even more preferred embodiment, the term “sophorolipids” is to be understood as referring to compounds of the general formulae (IX), (XI) and salts thereof, preferably compounds of the general formulae (X), (XII) and salts thereof:
Formulae (IX), (X) represent the acid form, Formulae (XI), (XII) represent the lactone form. nSL, R1SL, R2SL, R4SL have the same meaning as described for formulae (V), (VII), (VI), (VIII).
Sophorolipids may be used in accordance with the invention in their acid form or their lactone form. Preferred compositions in the context of the use according to the first aspect of the present invention comprise a sophorolipid in which the ratio by weight of lactone form to acid form is in the range of 20:80 to 80:20, especially preferably in the ranges of 30:70 to 40:60.
To determine the content of sophorolipids in the acid or lactone form in a formulation, refer to EP 1 411 111 B1 , page 8, paragraph [0053],
Sophorolipids may be obtained as described in EP 1 411 111 A1 , paragraphs [0021], [0022].
1.4 Aqueous Mixture L
In the use according to the first aspect of the invention, the at least on biosurfactant SBIO is comprised in an aqueous mixture L. During washing of Aw, Aw is contacted with the aqueous
mixture L. The aqueous mixture L is hence, in particular, the washing solution in which the at least one article Aw is washed.
“The woolen article Aw is contacted with the aqueous mixture L comprising at least one biosurfactant SBIO” is to be understood that, in the context of the use according the first aspect of the invention, the woolen article Aw is brought in direct contact with the aqueous mixture L comprising at least one biosurfactant SBIO, “in direct contact” meaning “wetting” of the woolen article Aw with the aqueous mixture L.
In a preferred embodiment of the use according to the first aspect of the invention, the article Aw is at least partially immersed, even more preferably fully immersed into the aqueous mixture L.
In an even more preferred embodiment, the article Aw is moved in the aqueous mixture L during the use according to the first aspect of the invention.
As Aw is contacted with the aqueous mixture L during washing of Aw, this means in particular that during washing of Aw in the aqueous mixture L during the use according to the first aspect of the invention, at least one impurity I which is absorbed or adsorbed on the article Aw is at least partially desorbed and thus removed from the article Aw and dispersed or dissolved, in particular dispersed, in aqueous mixture L during the use according to the first aspect of the invention. In other words: during the use according to the first aspect of the invention, the mixture L is preferably brought in direct contact with at least a part of the surface of article Aw, onto which at least one impurity I is absorbed or adsorbed.
Therefore, in particular, during the use according to the first aspect of the invention, a mixture L is obtained wherein in the water comprised by aqueous mixture L, at least one impurity I in dissolved or dispersed, preferably dispersed.
The concentration of impurities I in the water comprised by aqueous mixture L therefore augments during the use according to the first aspect of the invention. In a preferred embodiment, the aqueous mixture L that is employed during the use according to the first aspect of the invention (i.e. before it contacts the at least one woolen article Aw) is essentially free of impurity I.
Preferably, the mixture L that is obtained during the use according to the first aspect of the invention, is a dispersion, in which the at least one impurity I is the dispersed phase in the water W comprised by aqueous mixture L.
The impurity I may either be in the form of a liquid (e.g. droplet) or a particle, and preferably is a particle.
In a preferred embodiment, the impurity I is at least one impurity which may be inorganic or organic, in particular inorganic, preferably clay, more preferably kaolin.
Organic impurities are preferably selected from oils and fats, which may be of vegetable, animal or synthetic origin, food based particles, soil, pollen, sebum, body fluids, for example, blood, sperm, sweat, urine, feces, liquor.
Inorganic impurities are preferably selected from the group consisting of carbon black, fine dust, plastic particles (e.g. microplastic particles), metal oxides (preferably iron oxide), silica (such as sand), clay, dyes (such as pigments), more preferably clay, in particular kaolin.
The beneficial use according to the first aspect of the invention is the reduction of shrinking of the at least one article Aw during washing in the aqueous mixture L comprising the at least one biosurfactant SBIO- Since the tendency of woolen articles to shrink is especially pronounced at temperatures above 45 °C, the best effect of the present invention is obtained when the aqueous mixture L has a temperature at or below 45 °C for at least as long as the aqueous mixture L contacts the at least one woolen article Aw-
It is thus preferred in the context of the use according to the first aspect of the present invention, that the temperature of the aqueous mixture L, for at least as long as the aqueous mixture L is contacted with the at least one woolen article Aw, is in a range of from > 0 °C to 45 °C, in particular in a range of from 1 °C to 40 °C, preferably in a range of from 5 °C to 35 °C, more preferably in a range of from 10 °C to 30 °C, more preferably in a range of from 15 °C to 30 °C, even more preferably in a range of from 20 °C to 30 °C, even more preferably in a range of from 25 °C to 30 °C.
The aqueous mixture L, in particular, comprises the at least one biosurfactant SBIO in an amount in the range of from 0.000001 wt.-% to 50 wt.-%, preferably in the range of from 0.00001 wt.-% to 30 wt.-%, more preferably in the range of from 0.00005 wt.-% to 20 wt.-%, more preferably in the range of from 0.00009 wt.-% ppm to 15 wt.-%, more preferably in the range of from 0.0001 wt.-% to 14 wt.-%, more preferably in the range of from 0.001 wt.-% to 13 wt.-%, more preferably in the range of from 0.002 wt.-% to 12 wt.-%, more preferably in the range of from 0.005 wt.-% to 11 wt.-%, more preferably in the range of from 0.01 wt.-% to 10 wt.-%, more preferably in the range of from 0.05 wt.-% to 9 wt.-%, more preferably in the range of from 0.1 wt.-% ppm to 8 wt.-%, more preferably in the range of from 0.5 wt.-% to 5 wt.-%, more preferably in the range of from 1 wt.-% to 5 wt.-%, more preferably in the range of from 1 wt.-% to 3 wt.-%, wherein the wt.-% in each case are the weight of all biosurfactants SBIO comprised by aqueous mixture L relative to the total weight of aqueous mixture L.
During washing of Aw in the aqueous mixture L in the context of the use according to the first aspect of the invention, Aw is contacted with the aqueous mixture L for at least 1 s, preferably at least 10 s, more preferably at least 1 min, even more preferably at least 5 min, preferably for 15 min to 240 min, even more preferably for 30 min to 180 min, even more preferably for 45 min to 120 min, even more preferably for 60 min to 120 min, even more preferably for 70 min to 100 min.
During washing of Aw in the aqueous mixture L in the context of the use according to the first aspect of the invention, the woolen article Aw is preferably moved in the aqueous mixture. This improves the washing efficiency.
The washing in the context of the use according to the first aspect of the invention is preferably carried out in a washing machine.
The at least one biosurfactant SBIO in the use according to the first aspect of the invention may be present at least partially as salts, in particular when it is comprised by the aqueous mixture L.
In those embodiments where the at least one biosurfactant SBIO is at least partially present as salts, the ratio of biosurfactants SBIO that are present as salts depends on the pH of the water in the aqueous mixture L.
In those embodiments where at least one biosurfactant SBIO is present as salts, it is preferred that at least a part of the biosurfactants SBIO are salts in which the cation is selected from the group consisting of Li+, Na+, K+, Mg2+, Ca2+, Al3+, NH4+, ammonium ions, wherein the ammonium ions may be primary, secondary, tertiary or quaternary. Particularly preferred cations are selected from the group comprising, preferably consisting of, Na+, K+, NH4+ and the triethanolammonium cation.
Exemplary representatives of suitable ammonium ions are tetramethylammonium, tetraethylammonium, tetrapropylammonium, tetrabutylammonium, [(2-hydroxyethyl)trimethylammonium] (choline) and also the cations of 2-aminoethanol (ethanolamine, “MEA”), diethanolamine (“DEA”), 2,2',2"-nitrilotriethanol (triethanolamine, “TEA”), 1-aminopropan-2-ol (monoisopropanolamine), ethylenediamine, diethylenetriamine, triethylenetetramine, tetraethylenepentamine, 1 ,4-diethylenediamine (piperazine), aminoethylpiperazine and aminoethylethanolamine.
The mixtures of the abovementioned cations may also be present as cations of the salts of the at least one biosurfactant SBIO, if present as salts, according to the invention.
In the context of the use according to the first aspect of the present invention, aqueous mixture L may comprise further ingredients, preferably at least one ingredient selected from surfactants (wherein the surfactants are different from SBIO), enzymes, builders, solvents, preservatives, benefit agents, polymers.
1.4.1 Surfactants
Hence, in an optional embodiment, aqueous mixture L in the context of the use according to the first aspect of the present invention, comprises, besides the at least one biosurfactant SBIO, at least
one further surfactant SY, wherein the surfactant SY is different from SBIO- The at least one further surfactant SY may be selected from anionic surfactants, nonionic surfactants, cationic surfactants, amphoteric surfactants, and in particular is selected from anionic surfactants, nonionic surfactants.
In those cases where the aqueous mixture L in the context of the use according to the first aspect of the present invention comprises at least one further surfactant SY that is different from the biosurfactants SBIO, it is preferred the ratio of weights of all biosurfactants SBIO in the mixture L to the weight of all surfactants SY is in the range of from 99 : 1 to 1 : 99, preferably 90 : 10 to 10 : 90, more preferably 80 : 20 to 20 : 80, even more preferably 70 : 30 to 30 : 70, even more preferably 60 : 40 to 40 : 60, eve more preferably 50 : 50.
1.4. 1 Nonionic surfactants
Aqueous mixture L in the context of the use according to the first aspect of the present invention optionally comprises at least one nonionic surfactant.
The nonionic surfactants are preferably selected from the group consisting of alkoxylated alcohols, alkoxylated fatty acid alkyl esters, alkyl glycosides, amine oxides, polyhdroxy fatty acid amides. The nonionic surfactants are more preferably selected from the group consisting of alkoxylated alcohols, alkoxylated fatty acid alkyl esters, alkyl glycosides.
In a further embodiment, the at least one nonionic surfactant is selected from the group consisting of alcohol ethoxylates (“AE” or “AEO”), alcohol propoxylates, propoxylated fatty alcohols (“PFA”), alkoxylated fatty acid alkyl esters, such as ethoxylated and/or propoxylated fatty acid alkyl esters, alkylphenol ethoxylates (“APE”), nonylphenol ethoxylates (“NPE”), alkylpolyglycosides (“APG”), alkoxylated amines, fatty acid monoethanolamides (“FAM”), fatty acid diethanolamides (“FADA”), ethoxylated fatty acid monoethanolamides (“EFAM”), polyglycerol esters, glaycerol esters, propoxylated fatty acid monoethanolamides (“PFAM”), polyhydroxy alkyl fatty acid amides, or A/-acyl A/-alkyl derivatives of glucosamine (glucamides, “GA”, or fatty acid glucamide, “FAGA”), as well as products available under the trade names “SPAN” and “TWEEN”, and combinations thereof.
Alkoxylated alcohols in the context of the present invention are preferably ethoxylated alcohols. In particular, alkoxylated alcohols are primary alcohols having preferably 8 to 18 carbon atoms and on average 1 to 12 mol of ethylene oxide (“EO”) per mol of alcohol, in which the alcohol radical can be linear or branched, preferably 2-position methyl-branched, or can contain linear and methyl- branched radicals in a mixture, as are customarily present in oxo alcohol radicals. In particular, however, alcohol ethoxylates with linear radicals from alcohols of native origin having 12 to 18 carbon atoms, for example from coconut, palm, tallow fat or oleyl alcohol, and on average 2 to 8
EO per mol of alcohol are preferred. The preferred ethoxylated alcohols include, for example, Ci2-Ci4-alcohols with 3 EO, 4 EO or 7 EO, Cg-Cn-alcohol with 7 EO, Ci3-Ci5-alcohols with 3 EO, 5 EO, 7 EO or 8 EO, Ci2-Cia-alcohols with 3 EO, 5 EO or 7 EO and mixtures of these, such as mixtures of Ci2-Ci4-alcohol with 3 EO and Ci2-Cia-alcohol with 7 EO. The stated degrees of ethoxylation are statistical average values which can be an integer or a fraction for a specific product. Preferred alcohol ethoxylates have a narrow homolog distribution.
Alkoxylated alcohols may also be selected from fatty alcohols with more than 12 EO. Examples thereof are tallow fatty alcohol with 14 EO, 25 EO, 30 EO or 40 EO. Nonionic surfactants which contain EO and propylene oxide (“PO”) groups together in the molecule can also be used. In this connection, it is possible to use block copolymers with EO-PO block units or PO-EO block units, but also EO-PO-EO copolymers or PO-EO-PO copolymers.
It is of course also possible to use mixed alkoxylated nonionic surfactants in which EO and PO units are not distributed blockwise, but randomly. Such products are obtainable as a result of the simultaneous action of ethylene oxide and propylene oxide on fatty alcohols.
Furthermore, alkyl glycosides can also be used as further nonionic surfactants.
A further class of preferably used nonionic surfactants, are alkoxylated, preferably ethoxylated or ethoxylated and propoxylated, fatty acid alkyl esters, preferably having 1 to 4 carbon atoms in the alkyl chain, in particular fatty acid methyl esters, as are described for example in JP S58-217598 A or which are preferably prepared by the process described in WO 90/13533 A1 .
Nonionic surfactants of the amine oxide type, for example A/-cocoalkyl-A/,A/-dimethylamine oxide and A/-tallowalkyl-A/,A/-dihydroxyethylamine oxide, and of the fatty acid alkanolamide type may also be suitable. The amount of these nonionic surfactants is preferably not more than that of the ethoxylated fatty alcohols, in particular not more than half thereof.
Further suitable nonionic surfactants are polyhydroxy fatty acid amides; the polyhydroxy fatty acid amides are substances which can usually be obtained by reductive amination of a reducing sugar with ammonia, an alkylamine or an alkanolamine and subsequent acylation with a fatty acid, a fatty acid alkyl ester or a fatty acid chloride.
1.4.2 Anionic surfactants
Aqueous mixture L in the context of the use according to the first aspect of the present invention optionally comprises at least one anionic surfactant.
The anionic surfactants are preferably selected from the group consisting of anionic surfactants of the sulfonate and anionic surfactants of the sulfate type.
Preferred surfactants of the sulfonate type are selected from Cg-Cn-alkylbenzenesulfonates, olefinsulfonates, i.e. mixtures of alkene- and hydroxyalkanesulfonates, and also disulfonates, as are obtained, in particular, from Ci2-Ci8-monoolefins with a terminal or internal double bond by
sulfonation with gaseous sulfur trioxide and subsequent alkaline or acidic hydrolysis of the sulfonation products. Also preferred are alkanesulfonates which are preferably obtained from Ci2-Cia-alkanes, for example by sulfochlorination or sulfoxidation with subsequent hydrolysis or neutralization. Similarly, the esters of a-sulfo fatty acids (ester sulfonates) are also preferred, for example the a-sulfonated methyl esters of hydrogenated coconut, palm kernel or tallow fatty acids. Preferred surfactants of the sulfate type are selected from sulphated fatty acid glycerol esters. Fatty acid glycerol esters are to be understood as meaning the mono-, di- and triesters, and also mixtures thereof, as are obtained, in particular, in the preparation by esterification of a monoglycerol with 1 to 3 mol of fatty acid or in the transesterification of triglycerides with 0.3 to 2 mol of glycerol. Preferred sulfated fatty acid glycerol esters here are the sulfation products of saturated fatty acids having 6 to 22 carbon atoms, for example of caproic acid, caprylic acid, capric acid, myristic acid, lauric acid, palmitic acid, stearic acid or behenic acid.
Further preferred surfactants of the sulfate type are selected from alkyl sulfates and alkenyl sulfates, which are preferably the alkali metal, in particular the sodium, salts of the sulphuric acid half-esters of the Ci2-Cis-fatty alcohols, for example from coconut fatty alcohol, tallow fatty alcohol, lauryl, myristyl, cetyl or stearyl alcohol or the C10-C20-OXO alcohols and those half-esters of secondary alcohols of these chain lengths. Furthermore, preference is given to alkyl sulfates and alkenyl sulfates of the specified chain length which contain a synthetic straight-chain alkyl radical prepared on a petrochemical basis, and which have an analogous degradation behaviour to the suitable compounds based on fatty chemical raw materials. From the point of view of washing, the Ci2-Ci6-alkyl sulfates and Ci2-Cis-alkyl sulfates and also Ci4-Cis-alkyl sulfates are preferred. 2,3-Alky I sulfates, which are prepared for example in accordance with the US 3,234,258 A or US 5,075,041 A and can be obtained as commercial products of the Shell Oil Company under the name DAN®, are also suitable anionic surfactants.
The sulfuric acid monoesters of the straight-chain or branched C7-C2o-alcohols ethoxylated with 1 to 6 mol of ethylene oxide (“EO”), such as 2-methyl-branched Cg-Cn-alcohols having on average 3.5 mol of (“EO”) or Ci2-Cis-fatty alcohols with 1 to 4 EO, are also preferred. On account of their high foaming behaviour, they are used in cleaning compositions only in relatively small amounts, for example in amounts of from 1 to 5 % by weight.
Further preferred anionic surfactants are the salts of alkylsulfosuccinic acid, which are also referred to as sulfosuccinates or as sulfosuccinic acid esters and constitute the monoesters and/or diesters of sulfosuccinic acid with alcohols, preferably fatty alcohols and in particular ethoxylated fatty alcohols. Preferred sulfosuccinates contain Cs-Cis-fatty alcohol radicals or mixtures of these. Particularly preferred sulfosuccinates contain a fatty alcohol radical which is derived from ethoxylated fatty alcohols. In this connection, sulfosuccinates whose fatty alcohol radicals are derived from ethoxylated fatty alcohols with a narrow homolog distribution are particularly preferred in turn. It is likewise also possible to use alkylsuccinic acid and alkenylsuccinic having preferably 8 to 18 carbon atoms in the alkyl chain/ alkenyl chain or salts thereof.
Particularly preferred anionic surfactants are soaps. Also of suitability are saturated and unsaturated fatty acid soaps, such as the salts of lauric acid, myristic acid, palmitic acid, stearic
acid, (hydrogenated) erucic acid and behenic acid, and also soap mixtures derived in particular from natural fatty acids, for example coconut, palm kernel, olive oil or tallow fatty acid.
The anionic surfactants including the soaps can be in the form of their sodium, potassium or ammonium salts, as well as soluble salts of organic bases, such as mono-, di- or triethanolamine. Preferably, the anionic surfactants are in the form of their sodium or potassium salts, in particular in the form of the sodium salts.
Anionic surfactants preferably comprised by aqueous mixture L are selected from the group consisting of anionic surfactants of the sulfonate and anionic surfactants of the sulfate type, even more preferably selected from the group consisting of linear alkylbenzenesulfonates (“LAS”), isomers of LAS, branched alkylbenzenesulfonates (“BABS”), phenylalkanesulfonates, a-olefinsulfonates (“AOS”), olefin sulfonates, alkene sulfonates, alkane-2,3-diylbis(sulfates), hydroxyalkanesulfonates and disulfonates, alkyl sulfates (“AS”) such as sodium dodecyl sulfate (“SDS”), fatty alcohol sulfates (“FAS”), primary alcohol sulfates (“PAS”), alcohol ethersulfates (“AES” or “AEOS” or “FES”, also known as alcohol ethoxysulfates or fatty alcohol ether sulfates) such as sodium dodecylpoly(oxyethylene) sulphate (“SLES”), secondary alkanesulphonates (“SAS”), paraffin sulphonates (“PS”), ester sulphonates, sulphonated fatty acid glycerol esters, a-sulpho fatty acid methyl esters (“a-SFMe” or “SES”) including methyl ester sulphonate (“MES”), alkyl- or alkenylsuccinic acid, dodecenyl/tetradecenyl succinic acid (“DTSA”), fatty acid derivatives of amino acids, diesters and monoesters of sulfo-succinic acid or soap, and combinations thereof.
1.4.3 Amphoteric surfactants
Aqueous mixture L in the context of the use according to the first aspect of the present invention optionally comprises at least one amphoteric surfactant, which preferably is a compound which carries at least one quaternary ammonium group and at least one -COO - or -SOs- group in the molecule, more preferably is selected from the group consisting of betaine, alkyldimethylbetaine, sulfobetaine.
Preferred betaine surfactants are alkyl- or alkylamidopropylbetaines. In particular, betaines such as the A/-alkyl-A/,A/-dimethylammonium glycinates, e.g. the cocoalkyldimethylammonium glycinate, A/-acylaminopropyl-A/,A/-dimethylammonium glycinates, e.g. the cocoacylaminopropyldimethylammonium glycinate, the Ci2-Ci8-alkyldimethylacetobetaine, the cocoamidopropyldimethylacetobetaine, 2-alkyl-3-carboxymethyl-3-hydroxyethylimidazolines and sulphobetaines having in each case 8 to 18 carbon atoms in the alkyl or acyl group, and also the cocoacylaminoethylhydroxyethylcarboxymethyl glycinate are preferred. A particularly preferred zwitterionic surfactant is the A/,A/-dimethyl-A/-(lauroylamidopropyl)ammoniumacetobetaine known under the INCI name Cocamidopropyl Betaine.
Further suitable amphoteric surfactants are formed by the group of amphoacetates and amphodiacetates, in particular, for example, coco- or laurylamphoacetates or -diacetates, the group of amphopropionates and amphodipropionates, and the group of amino acid-based surfactants such as acyl glutamates, in particular disodium cocoyl glutamate and sodium cocoyl glutamate, acyl glycinates, in particular cocoyl glycinates, and acyl sarcosinates, in particular ammonium lauroyl sarcosinate and sodium cocoyl sarcosinate.
1.4.4 Cationic surfactants
Aqueous mixture L in the context of the use according to the first aspect of the present invention optionally comprises at least one cationic surfactant, which are preferably selected from the group consisting of alklydimethylethanolamine quat (“ADMEAQ”), cetyltrimethylammonium bromide (“CTAB”), dimethyldistearylammonium chloride (“DSDMAC”), and alkylbenzyldimethylammonium, alkyl quaternary ammonium compounds, alkoxylated quaternary ammonium (“AQA”) compounds. Non-limiting examples of non-ionic surfactants include alcohol ethoxylates (“AE” or “AEO”), alcohol propoxylates, propoxylated fatty alcohols (“PFA”), alkoxylated fatty acid alkyl esters, such as ethoxylated and/or propoxylated fatty acid alkyl esters, alkylphenol ethoxylates (“APE”), nonylphenol ethoxylates (“NPE”), alkylpolyglycosides (“APG”), alkoxylated amines, fatty acid monoethanolamides (“FAM”), fatty acid diethanolamides (“FADA”), ethoxylated fatty acid monoethanolamides (“EFAM”), polyglycerol esters, glaycerol esters, propoxylated fatty acid monoethanolamides (“PFAM”), polyhydroxy alkyl fatty acid amides, or A/-acyl A/-alkyl derivatives of glucosamine (glucamides, “GA”, or fatty acid glucamide, “FAGA”), as well as products available under the trade names “SPAN” and “TWEEN”, and combinations thereof.
1 .4.2 Enzymes
The aqueous mixture L in the context of the use according to the first aspect of the invention may also comprise at least one enzyme E.
In those embodiments where the aqueous mixture L in the context of the use according to the first aspect of the invention comprises at least one enzyme E, it is further preferred that the enzyme E is selected from the group consisting of protease, amylase, cellulase, mannanase, lipase, cutinase, pectate lyase, peroxidase, oxidase, laccase, even more preferably selected from the group consisting of cellulase, protease, lipase, cutinase, amylase, peroxidase, oxidase, mannanase. It is even more preferred that the at least one enzyme E is selected from the group consisting of protease, amylase, lipase, mannanase. Most preferred, the at least one enzyme E is a lipase.
In case the aqueous mixture L in the context of the use according to the first aspect of the invention comprises at least one enzyme E, it is preferred that the amount of all enzymes E in the aqueous
mixture L is in the range of 0.01 ppm to 1 wt.-%, preferably 0.01 ppm to 1000 ppm, more preferably 0.1 to 100 ppm, preferably 1 ppm to 10 ppm per the total weight of aqueous mixture L.
1.4.2. 1 Cellulases
Suitable cellulases include those of bacterial or fungal origin. Chemically modified or protein engineered mutants are included. Suitable cellulases include cellulases from the genera Bacillus, Pseudomonas, Humicola, Fusarium, Thielavia, Acremonium , e.g., the fungal cellulases produced from Humicola insolens, Myceliophthora thermophila and Fusarium oxysporum disclosed in US 4,435,307 A, US 5,648,263 A, US 5,691 ,178 A, US 5,776,757 A and WO 89/09259 A1 . Especially suitable cellulases are the alkaline or neutral cellulases having color care benefits. Examples of such cellulases are cellulases described in EP 0 495 257 A1 , WO 91/17243 A1 , WO 96/11262 A1 , WO 96/29397 A1 , WO 98/08940 A1 . Other examples are cellulase variants such as those described in WO 94/07998 A1 , WO 91/17244 A1 , US 5,457,046 A, US 5,686,593 A, US 5,763,254 A, WO 95/24471 A1 , WO 98/12307 A1 and WO 99/01544 A1.
Example of cellulases exhibiting endo-beta-1 ,4-glucanase activity (EC 3.2.1.4) are those having described in WO 02/099091 A2.
Commercially available cellulases include Celluzyme™, and Carezyme™ (Novozymes A/S), Clazinase™, and Puradax HA™ (Genencor International Inc.), and KAC-500(B)™ (Kao Corporation).
1.4.2.2 Proteases
Suitable proteases include those of bacterial, fungal, plant, viral or animal origin e.g. vegetable or microbial origin. Microbial origin is preferred. Chemically modified or protein engineered mutants are included. It may be an alkaline protease, such as a serine protease or a metalloprotease. A serine protease may for example be of the S1 family, such as trypsin, or the S8 family such as subtilisin. A metalloproteases protease may for example be a thermolysin from e.g. family M4 or other metalloprotease such as those from M5, M7 or M8 families.
The term “subtilases” refers to a sub-group of serine protease according to R.J. Siezen, W.M. de Vos, J.A.M. Leunissen, B.W. Dijkstra, Protein Engineering, Design and Selection 1991 , 4, 719-737 and R.J. Siezen & J.A. Leunissen, Protein Science 1997, 6, 501-523. Serine proteases are a subgroup of proteases characterized by having a serine in the active site, which forms a covalent adduct with the substrate. The subtilases may be divided into 6 sub-divisions, i.e. the Subtilisin family, the Thermitase family, the Proteinase K family, the Lantibiotic peptidase family, the Kexin family and the Pyrolysin family.
Examples of subtilases are those derived from Bacillus such as Bacillus lentus, B. alkalophilus, B. subtilis, B. amyloliquefaciens, Bacillus pumilus and Bacillus gibsonii described in: US 7,262,042 B2 and WO 2009/021867 A2, and subtilisin lentus , subtilisin Novo, subtilisin Carlsberg, Bacillus
licheniformis, subtilisin BPN', subtilisin 309, subtilisin 147 and subtilisin 168 described in
WO 89/06279 A1 and protease PD138 described in WO 93/18140 A1. Other useful proteases may be those described in WO 2019/105675 A1 , WO 01/016285 A2, and WO 02/016547 A2. Examples of trypsin-like proteases are trypsin (e.g. of porcine or bovine origin) and the Fusarium protease described in WO 89/06270 A1 , WO 94/25583 A1 and WO 2005/040372 A1 , and the chymotrypsin proteases derived from Cellumonas described in WO 2005/052161 A2 and WO 2005/052146 A2. A further preferred protease is the alkaline protease from Bacillus lentus DSM 5483, as described for example in WO 95/23221 A1 , and variants thereof which are described in WO 92/21760 A2, WO 95/23221 A1 , EP 1 921 147 A2 and EP 1 921 148 A2.
Examples of metalloproteases are the neutral metalloprotease as described in
WO 2007/044993 A2 (Genencor I nt.) such as those derived from Bacillus amyloliquefaciens. Examples of useful proteases are the variants described in: WO 92/19729 A1 , WO 96/34946 A1 , WO 98/20115 A1 , WO 98/20116 A1 , WO 99/11768 A1 , WO 01/44452 A1 , WO 03/006602 A2, WO 2004/03186 A2, WO 2004/041979 A2, WO 2007/006305 A1 , WO 2011/036263 A1 , WO 2011/036264 A1 .
Suitable commercially available protease enzymes include those sold under the trade names Alcalase®, Duralase™, Durazym™, Relase®, Relase® Ultra, Savinase®, Savinase® Ultra, Primase, Polarzyme®, Kannase®, Liquanase®, Liquanase® Ultra, Liquanase® 2.5 L, Ovozyme®, Coronase, Coronase® Ultra, Neutrase®, Everlase® and Esperase® (Novozymes A/S), those sold under the tradename Maxatase®, Maxacai®, Maxapem®, Purafect®, Purafect Prime®, Eraser®, Purafect MA®, Purafect Ox®, Purafect OxP®, Puramax®, Properase®, Ultimase®, FN2®, FN3®, FN4®, Excellase®, , Opticlean® and Optimase® (Danisco/DuPont), Axapem™ (Gist-Brocases N.V.), BLAP (sequence shown in Figure 29 of US 5,352,604 A) and variants hereof (Henkel AG) and KAP (Bacillus alkalophilus subtilisin) from Kao.
A protease preferably comprised in the composition according to the instant invention is Liquanase® 2.5 L.
1.4.2.3 Lipases and cutinases
Suitable lipases and cutinases include those of bacterial or fungal origin. Chemically modified or protein engineered mutant enzymes are included. Examples include lipase from Thermomyces , e.g. from T. lanuginosus (previously named Humicola lanuginosa) as described in EP 0 258 068 A2 and EP 0 305 216 A1 , cutinase from Humicola, e.g. H. insolens (WO 96/13580 A1), lipase from strains of Pseudomonas (some of these now renamed to Burkholderia), e.g. P. alcaligenes or P. pseudoalcaligenes (EP 0218272 A1), P. cepacia (EP 0 331 376 A2), P. sp. strain SD705 (US 5,827,718 A & US 5,942,431 A), P. wisconsinensis QNO 96/12012 A1),
GDSL-type Streptomyces lipases (WO 2010/065455 A2), cutinase from Magnaporthe grisea (WO 2010/107560 A2), cutinase from Pseudomonas mendocina (US 5,389,536 A), lipase from Thermobifida fusca (WO 2011/084412 A1), Geobacillus stearothermophilus lipase
(WO 2011/084417 A1), lipase from Bacillus subtilis (WO 2011/084599 A1), and lipase from Streptomyces griseus (WO 2011/150157 A2) and S. pristinaespiralis (WO 2012/137147 A1). Other examples are lipase variants such as those described in EP 0 407 225 A1 , WO 92/05249 A1 , WO 94/01541 A1 , WO 94/25578 A1 , WO 95/14783 A1 , WO 95/30744 A2, WO 95/35381 A1 , WO 95/22615 A1 , WO 96/00292 A1 , WO 97/04079 A1 , WO 97/07202 A1 , WO 00/34450 A1 , WO 00/60063 A1 , WO 01/92502 A1 , WO 2007/87508 A2, ON 104031899 A and WO 2009/109500 A1.
Preferred commercial lipase products include Lipolase™, Lipex™, Lipolex™, Lipex™ 100 L Evity and Lipoclean™ (Novozymes A/S), Lumafast (originally from Genencor) and Lipomax (originally from Gist-Brocades).
A lipase preferably comprised in the composition according to the instant invention is Lipex™ 100 L Evity.
Still other examples are lipases sometimes referred to as acyltransferases or perhydrolases, e.g. acyltransferases with homology to Candida antarctica lipase A (WO 2010/111143 A2), acyltransferase from Mycobacterium smegmatis (WO 2005/56782 A2), perhydrolases from the CE 7 family (WO 2009/67279 A1), and variants of the Mycobacterium smegmatis perhydrolase in particular the S54V variant used in the commercial product Gentle Power Bleach from Huntsman Textile Effects Pte Ltd (WO 2010/100028 A2).
1.4.2.4 Amylases
Suitable amylases which can be used herein may be an a-amylase or a glucoamylase and may be of bacterial or fungal origin. Chemically modified or protein engineered mutants are included.
Amylases include, for example, a-amylases obtained from Bacillus , e.g., a special strain of Bacillus licheniformis , described in more detail in GB 1 ,296,839.
Suitable amylases include amylases having SEQ ID NO: 3 in WO 95/10603 A1 or variants having about 90 % sequence identity to SEQ ID NO: 3 thereof. Preferred variants are described in WO 94/02597 A1 , WO 94/18314 A1 , WO 97/43424 A1 and SEQ ID NO: 4 of WO 99/19467 A1. Different suitable amylases include amylases having SEQ ID NO: 6 in WO 02/10355 A2 or variants thereof having about 90% sequence identity thereto.
Further amylases which are suitable are amylases having SEQ ID NO: 6 in WO 99/019467 A1 or variants thereof having about 90% sequence identity to SEQ ID NO: 6.
Additional amylases which can be used are those having SEQ ID NO: 1 , SEQ ID NO: 3, SEQ ID NO: 2 or SEQ ID NO: 7 of WO 96/23873 A1 or variants thereof having 90% sequence identity to SEQ ID NO: 1 , SEQ ID NO: 2, SEQ ID NO: 3 or SEQ ID NO: 7.
Other amylases which can be used are amylases having SEQ ID NO: 2 of WO 2008/153815 A1 , SEQ ID NO: 10 in WO 01/66712 A2 or variants thereof having about 90% sequence identity to SEQ ID NO: 2 of WO 2008/153815 A1 or about 90% sequence identity to SEQ ID NO: 10 in WO 01/66712 A2.
Further suitable amylases are amylases having SEQ ID NO: 2 of WO 2009/061380 A2 or variants having about 90% sequence identity to SEQ ID NO: 2 thereof.
Other suitable amylases are the a-amylase having SEQ ID NO: 12 in WO 01/66712 A2 or a variant having at least about 90% sequence identity to SEQ ID NO: 12.
Other examples are amylase variants such as those described in WO 2011/098531 A1 , WO 2013/001078 A1 and WO 2013/001087 A2.
Commercially available amylases are Amplify™ Prime 100 L, Duramyl™, Termamyl™, Fungamyl™, Stainzyme™, Stainzyme Plus™, Natalase™, Liquozyme X and BAN™ (from Novozymes A/S), and Rapidase™, Purastar™/Effectenz™, Powerase and Preferenz S100 (from Genencor International Inc./DuPont).
1.4.2.5 Peroxidases/ oxidases
Suitable peroxidases/oxidases include those of plant, bacterial or fungal origin. Chemically modified or protein engineered mutants are included. Examples of useful peroxidases include peroxidases from Coprinus , e.g., from C. cinereus , and variants thereof as those described in WO 93/24618 A1 , WO 95/10602 A1 , and WO 98/15257 A1 .
Commercially available peroxidases include Guardzyme™ (Novozymes A/S). The detergent enzyme(s) may be included in a detergent composition by adding separate additives containing one or more enzymes, or by adding a combined additive comprising all of these enzymes. A detergent additive as contemplated herein, i.e., a separate additive or a combined additive, can be formulated, for example, as a granulate, liquid, slurry, etc. Preferred detergent additive formulations are granulates, in particular non-dusting granulates, liquids, in particular stabilized liquids, or slurries.
1.4.2.6 Mannanases
Mannanases which are particularly preferred according to the invention are mannanases which are sold, for example, under the trade names Mannaway® by the company Novozymes or Purabrite® by the company Genencor. A mannanase preferably comprised in the composition according to the instant invention is Mannaway® 4.0 L.
1 .4.3 Builders
The aqueous mixture L in the context of the use according to the first aspect of the invention preferably also comprise at least one builder Bz.
In those embodiments where aqueous mixture L in the context of the use according to the first aspect of the invention comprises at least one builder Bz, the amount of all builders Bz in aqueous
mixture L is preferably between 0.1 ppm to 1000 ppm, preferably 1 ppm to 100 ppm, more preferably 10 ppm to 50 ppm relative to the total weight of aqueous mixture L.
The builder Bz may particularly be a chelating agent that forms water-soluble complexes with calcium and magnesium. Non-limiting examples of builders Bz include zeolites, diphosphates (pyrophosphates), triphosphates such as sodium triphosphate (“STP” or “STPP”), carbonates such as sodium carbonate, soluble silicates such as sodium metasilicate, layered silicates (e.g., SKS-6 from Hoechst), ethanolamines such as 2-aminoethan-1-ol (“MEA”), diethanolamine (“DEA”, also known as iminodiethanol), triethanolamine (“TEA”, also known as 2,2',2"-nitrilotriethanol) , and carboxymethyl inulin (“CMI”), and combinations thereof.
Non-limiting examples of builders Bz include homopolymers of polyacrylates or copolymers thereof, such as poly(acrylic acid) (“PAA”) or copoly(acrylic acid/maleic acid) (“PAA/PMA”). Further nonlimiting examples of builders Bz include polyaspartic acids and polyglutamic acids and their salts, citrates, ascorbic acid chelators such as aminocarboxylates, aminopolycarboxylates, like A/,A/-dicarboxymethyl glutamic acid and methylglycine A/,A/-diacetic acid, and phosphonates, and alkyl- or alkenylsuccinic acid. Additional specific examples of builders Bz include 2,2’,2”-nitrilotriacetic acid (“NTA”), ethylenediaminetetraacetic acid (“EDTA”), diethylenetriaminepentaacetic acid (“DTPA”), iminodisuccinic acid (“IDS”), ethylenediamine- A/,A/’-disuccinic acid (“EDDS”), glutamic acid-A/,A/-diacetic acid (“GLDA”), 1-hydroxyethane-1 ,1- diphosphonic acid (“HEDP”), ethylenediaminetetra-(methylenephosphonic acid) (“EDTMPA”), diethylenetriaminepentakis(methylenephosphonic acid) (“DTPMPA” or “DTMPA”), N-(2- hydroxyethyl)iminodiacetic acid (“EDG”), aspartic acid-AZ-monoacetic acid (“ASMA”), aspartic acid- A/,A/-diacetic acid (“ASDA”), aspartic acid-AZ-monopropionic acid (“ASMP”), iminodisuccinic acid (“IDA”), A/-(2-sulfomethyl)-aspartic acid (“SMAS”), A/-(2-sulfoethyl)-aspartic acid (“SEAS”), A/-(2- sulfomethyl)-glutamic acid (“SMGL”), A/-(2-sulfoethyl)-glutamic acid (“SEGL”), AZ- methyliminodiacetic acid (“MIDA”), a-alanine-A/,A/-diacetic acid (“a-ALDA”), serine-A/,A/-diacetic acid (“SEDA”), isoserine-A/,A/-diacetic acid (“ISDA”), phenylalanine-A/,A/-diacetic acid (“PHDA”), anthranilic acid-A/,A/-diacetic acid (“ANDA”), sulfanilic acid-A/,A/-diacetic acid (“SLDA”), ta urine- AZ, AZ- diacetic acid (“TUDA”) and sulfomethyl-A/,A/-diacetic acid (“SMDA”), A/-(2-hydroxyethyl)- ethylidenediamine-A/,A/',A/'-triacetate (“HEDTA”), diethanolglycine (“DEG”), diethylenetriamine penta(methylenephosphonic acid) (“DTPMP”), aminotris(methylenephosphonic acid) (“ATMP”), and combinations and salts thereof.
Preferred builder Bz according to the instant invention are selected from the group of aminopolycarboxylates, like A/,A/-dicarboxymethyl glutamic acid and methylglycine A/,A/-diacetic acid, citrates, polyaspartic acids and polyglutamic acids and their salts.
1 .4.4 Solvents
In a preferred embodiment, aqueous mixture L in the context of the use according to the first aspect of the invention may comprise at least one non-aqueous solvent. In those embodiments
where aqueous mixture L in the context of the use according to the first aspect of the invention comprises at least one non-aqueous solvent, the ratio between the weight of the water comprised by aqueous mixture L to the weight of all non-aqueous solvents comprised by aqueous mixture L is preferably in the range of from 999 : 1 to 1 : 999, preferably in the range of from 99 : 1 to 1 : 99, more preferably in the range of from 9 : 1 to 1 : 9, more preferably in the range of from 8 : 2 to 2 : 8, more preferably in the range of from 7 : 3 to 3 : 7, more preferably in the range of from 6 : 4 to 4 : 6, more preferably at 1 : 1 .
Suitable non-aqueous solvents include monohydric or polyhydric alcohols, alkanolamines or glycol ethers, provided they are miscible with water in the specified concentration range. The solvents are preferably selected from ethanol, n-propanol, /so-propanol, butanols, glycol, propanediol, butanediol, glycerine, diglycol, propyldiglycol, butyldiglycol, hexylene glycol, ethylene glycol methyl ether, ethylene glycol ethyl ether, ethylene glycol propyl ether, n-butyl glycol ether, ethylene glycol mono-glycol ether, diethylene glycol ethyl ether, propylene glycol methyl ether, propylene glycol ethyl ether, propylene glycol propyl ether, dipropylene glycol monomethyl ether, dipropylene glycol monoethyl ether, diisopropylene glycol monomethyl ether, diisopropylene glycol monoethyl ether, methoxy triglycol, ethoxy triglycol, butoxy triglycol, 1-butoxyethoxy-2-propanol, n-butoxyethoxy-2- propanol, di-butoxyethoxy-2-propanol, 3-butyl-3-methoxyether solvents, and mixtures of these solvents, butoxyethoxy-2-propanol, 3-buty l-3-butyl ether, propyl glycol, di-octanol ether, di-butoxy- 2-propanol, 3-butyl-3-butylether, propanol, propylenglycol, di-butoxyethoxy-2- propanol, di- butoxyethoxy-2-propanol, 3-butoxy-3-methoxyether solvents, diisopropylene glycol monomethylether, diisopropylene glycol monomethylether, and mixtures of these solvents. In case composition ZG is a surfactant preparation, it preferably contains a polyol as the non-aqueous solvent. The polyol can comprise glycerol, 1 ,2-propanediol, 1 ,3-propanediol, ethylene glycol, diethylene glycol and / or dipropylene glycol.
1 .4.5 Preservatives
In a preferred embodiment, aqueous mixture L in the context of the use according to the first aspect of the invention may comprise at least one preservative.
In those embodiments where aqueous mixture L in the context of the use according to the first aspect of the invention comprises at least one preservative, the amount of all preservatives in aqueous mixture L is preferably between 0.1 ppm to 1000 ppm, preferably 1 ppm to 100 ppm, more preferably 10 ppm to 50 ppm, relative to the total weight of aqueous mixture L.
Preferred preservatives are selected from the group consisting of phenoxyethanol, sodium levulinate, sodium benzoate, p-anisic acid, potassium sorbate, benzoic acid, glyceryl caprylate, capryl glycol, penthylene glycol, methyl propane diol, bronopol, isothiazolinone (in particular selected from methylisothiazolinone, chloromethylisothiazolinone).
1 .4.6 Benefit
In a preferred embodiment, aqueous mixture L in the context of the use according to the first aspect of the invention may comprise at least one benefit agent, preferably at least one encapsulate comprising a benefit agent, preferably a sensorial benefit agent.
In those embodiments where aqueous mixture L in the context of the use according to the first aspect of the invention comprises at least one benefit agent, the total amount of benefit agent in aqueous mixture L is preferably between 0.1 ppm to 1000 ppm, preferably 1 ppm to 100 ppm, more preferably 10 ppm to 50 ppm relative to the total weight of aqueous mixture L.
The preferred benefit agent is a perfume. A benefit agent may be provided in an encapsulate. Aqueous mixture L optionally also comprise an unconfined (also called non-encapsulated) benefit agent, for example a volatile benefit agent. Where the volatile benefit agent is a perfume, the perfumes are suitable for use as the encapsulated volatile benefit agent and also as the unconfined perfume component.
Preferred encapsulates in this context comprise shear/pressu re-sensitive action encapsulates, whereby the sensorial benefit agent is released in response to mechanical force (e.g., friction, pressure, shear stress) on the encapsulate. The encapsulate shell is preferably comprised of materials including but not limited to polyurethane, polyamide, polyolefin, polysaccharide, protein, silicone, lipid, modified cellulose, gums, polyacrylate, polyphosphate, polystyrene, polyesters or combinations of these materials. Preferably the benefit agent is a sensorial benefit agent, a skin benefit agent or an olfactory benefit agent and/or may be a volatile benefit agent. Sensorial benefit agents may also have benefits for hair and/or hard surfaces and/or fabrics. The sensorial benefit may have anti-foam properties, and as such it is advantageous for foaming purposes that it is encapsulated so as not interfering with the foam until release by rubbing. Suitable volatile benefit agents include but are not limited to perfumes, insect repellents, essential oils, sensates such as menthol and aromatherapy actives, preferably perfumes. Mixtures of volatile benefit agents may be used.
In a preferred embodiment, aqueous mixture L in the context of the use according to the first aspect of the invention may comprise at least one polymer for use in detergents. The polymer may function as a co-builder as mentioned above, or may provide antiredeposition, fiber protection, soil release, dye transfer inhibition, viscosity modifiers, grease cleaning and/or anti-foaming properties. In those embodiments where aqueous mixture L in the context of the use according to the first aspect of the invention comprises at least one polymer, the amount of all polymers in aqueous mixture L is preferably between 0.1 ppm to 1000 ppm, preferably 1 ppm to 100 ppm, more preferably 10 ppm to 50 ppm relative to the total weight of aqueous mixture L.
Exemplary polymers include starch, polyquaternium (in particular polyquaternium 10),(carboxymethyl)cellulose (“CMC”), poly(vinyl alcohol) (“PVA”), polyvinylpyrrolidone) (“PVP”), poly(ethyleneglycol) or poly(ethylene oxide) (“PEG”), ethoxylated poly(ethyleneimine), carboxymethyl inulin (“CMI”), and polycarboxylates such as PAA, PAA/PMA, poly-aspartic acid, and lauryl methacrylate/acrylic acid copolymers, hydrophobically modified CMC (“HM-CMC”) and silicones, copolymers of terephthalic acid and oligomeric glycols, copolymers of poly(ethylene terephthalate) and poly(oxyethene terephthalate) (“PET-POET”), PVP, poly(vinylimidazole) (“PVI”), poly(vinylpyridine-A/-oxide) (“PVPO” or “PVPNO”) and polyvinylpyrrolidone-vinylimidazole (“PVPVI”). Further exemplary polymers include sulfonated polycarboxylates, polyethylene oxide and polypropylene oxide (“PEO-PPO”) and diquaternium ethoxy sulfate. Other exemplary polymers are disclosed in, e.g. WO 2006/130575 A2. Salts of the above-mentioned polymers are also contemplated.
Preferably a composition ZG according to the instant invention is characterized in that it comprises at least one selected from anti-redeposition polymers and soil release polymers, with soil release polymers being preferred. This has the technical effect, that the cleaning capabilities of the composition according to the instant invention is even more enhanced. It is preferred in the context of the instant invention, that the anti-redeposition polymer or soil release polymer is selected from the group consisting of modified cellulose, preferably carboxymethylcellulose, cellulose acetate and methylcellulose, modified starch, modified inulin, preferably carboxy methyl inulin, polyitaconic acid, polyvinylpyrrolidone, polyvinyl alcohol, and polyethylene glycol, with carboxymethylcellulose and methylcellulose being most preferred.
Further preferred soil release polymers are water soluble polyesters as for example from the TexCare® range commercially available under the name TexCare SRN 260, TexCare SRN 170, TexCare SRN 260 Life and combinations thereof, as well, as the soil release polymers disclosed in WO 2016/075178 A1 , WO 2016/075179 A1 , EP 3 489 340 A1 and EP 3 489 338 A1 . Further preferred soil release polymers are selected from carboxy methyl inulins. A commercial example is CarboxylineOCMI.
EP 1 746 109 A2 discloses hybrid polymers of amylose and acrylates, that can also advantageously used in the composition of the instant invention as soil release polymers. A commercial example for this type of soil release polymers is Alcoguard® H 5941.
Non-limiting examples of biopolymers including: starch, like e.g. corn starch, zea mays starch and tapioca starch, modified starch, like e.g. starch hydroxypropyltrimonium chloride and hydrolyzed corn starch, cellulose, bacterial cellulose, modified cellulose, like e.g. microcrystalline cellulose, hydroxypropyl methylcellulose and cetyl hydroxyethylcellulose, guar gum, pectin, inulin, carrageenan, alginate, galactoarabinan, polycitronellol, carboxymethyl inulin, carboxymethyl cellulose, polyitaconic acid and combinations and salts thereof.
In a preferred embodiment, aqueous mixture L in the context of the use according to the first aspect of the invention comprises at least one polymer selected from the group consisting of carboxymethylcellulose, polyethylenglycol, starch, carboxymethyl inulin, poly-aspartic acid, polyglutamic acid, sulfonated polycarboxylates, polyethylene oxide, polypropylene oxide, polyquaternium. These polymers are preferred as they are ecologically favorable. Preferred polyquaternium is polyquaternium 10 (CAS-Nr.: 81859-24-7), which is quaternized hydroxyethyl cellulose.
1 .4.8 Other Additives
In a preferred embodiment, the aqueous mixture L in the context of the use according to the first aspect of the invention may comprise at least one further additive.
The further additive is preferably selected from the group consisting of bleaching systems, antiredeposition aids, fiber protection agents, soil release agents, dye transfer inhibitors, fabric hueing agents, blueing dyes, enzyme stabilizing agents like boric acid, pH-regulators, emollients, emulsifiers, thickeners/ viscosity regulators /stabilizers, UV photoprotective filters, antioxidants, hydrotropes (or polyols), solids and fillers, film formers, pearlescent additives, deodorant and antiperspirant active ingredients, insect repellents, self-tanning agents, preservatives, conditioners, perfumes, dyes, odour absorbers, cosmetic active ingredients, care additives, superfatting agents, solvents, malodor removers.
Substances which can be used as exemplary representatives of the individual groups are known to the person skilled in the art and can be found for example in US 2011/0091399 A1 .
The amounts of the respective additives are dependent on the intended use.
1.5 Composition ZG
For carrying out the use according to the first aspect of the invention, the at least one biosurfactant SBIO may be provided in pure form or as a composition ZG in mixture with further compounds, which are set forth below.
Composition ZG preferably is a cleaning formulation which may be employed in the use according to the first aspect of the invention.
When subjected to the use according to the first aspect of the invention, the composition ZG may then be diluted with water and the thus obtained aqueous mixture L may then be contacted with the woolen article Aw to wash article Aw- Hence, typically, composition ZG comprises the same compounds (except water) as aqueous solution L, just in higher concentration.
Where composition ZG comprises water W, it is preferred that the pH of the water W in composition ZG at 25 °C is from 3.5 to 9, preferably from 5 to 9, preferably from 6 to 9, and particularly preferably from 7 to 8.
Composition ZG may comprise further ingredients, depending on the specific area of application of composition ZG.
The present invention is based on the use of the at least one biosurfactant SBIO to reduce shrinking of the woolen article Aw during washing. The combination of the at least one biosurfactant SBIO with further surfactants is therefore not necessary, since, in particular, certain surfactants such as SLES, an anionic surfactant, are inferior to SBIO in terms of reducing the shrinking of the woolen article Aw- On the other hand, a positive effect is already obtained if only a part of these prior art surfactants comprised by prior art cleaning formulations is substituted with at least one biosurfactant SBIO-
Hence, in an optional embodiment, composition ZG comprises, besides the at least one biosurfactant SBIO, at least one further surfactant which is different from the at least one biosurfactant SBIO, preferably at least two further surfactants which are both different from the at least one biosurfactant SBIO, more preferably at least three further surfactants which are different from the at least one biosurfactant SBIO- This at least one further surfactant may be selected from anionic surfactants, nonionic surfactants, cationic surfactants, amphoteric surfactants, and in particular is selected from anionic surfactants, nonionic surfactants.
It is preferred that the amount of all biosurfactants, in particular the amount of all glucolipids, rhamnolipids, and sophorolipids, in the composition ZG is in the range of from 5 to 60 wt.-%, more preferably 15 to 40 wt.-%, based on the total mass of the composition ZG.
In those cases where the composition ZG comprises at least one further surfactant SY that is different from the at least one biosurfactant SBIO, it is preferred that the amount of all surfactants SY different from biosurfactants SBIO in the composition ZG is in the range of from 5 to 60 weight-% (=”wt.-%”), more preferably 15 to 40 wt.-%, based on the total mass of the composition ZG.
1.5.1 Nonionic surfactants
Composition ZG optionally comprises at least one nonionic surfactants.
Preferred nonionic surfactants are described under item 1.4.1 .
1 .5.2 Anionic surfactants
Composition ZG optionally comprises at least one anionic surfactants.
Preferred anionic surfactants are described under item 1 .4.2.
1 .5.3 Amphoteric surfactants
Composition ZG optionally comprises at least one amphoteric surfactants.
Preferred amphoteric surfactants are described under item 1 .4.3.
1 .5.4 Cationic surfactants
Composition ZG optionally comprises at least one cationic surfactants.
Preferred anionic surfactants are described under item 1 .4.4.
1 .5.5 Enzymes
Composition ZG optionally comprise at least one enzyme E.
In those embodiments where composition ZG comprises at least one enzyme E, it is further preferred that the enzyme E is selected from the group consisting of protease, amylase, cellulase, mannanase, lipase, cutinase, pectate lyase, peroxidase, oxidase, laccase, even more preferably selected from the group consisting of cellulase, protease, lipase, cutinase, amylase, peroxidase, oxidase, mannanase.
Preferred examples of these enzymes are described above.
It is even more preferred that the at least one enzyme E is selected from the group consisting of protease, amylase, lipase, mannanase. Most preferred, the at least one enzyme E is a lipase.
In case the composition ZG comprises at least one enzyme E, it is preferred that the amount of all enzymes E in the composition ZG is in the range of 0.1 to 4 wt.-% per the total weight of composition ZG.
The at least one enzyme E may be included in the composition ZG by adding separate additives (“detergent additives”) containing one or more enzymes, or by adding a combined additive comprising all enzymes E comprised by the composition ZG. Such a detergent additive as contemplated herein, i.e., a separate additive or a combined additive, can be formulated, for example, as a granulate, liquid, slurry, etc. Preferred detergent additive formulations are granulates, in particular non-dusting granulates, liquids, in particular stabilized liquids, or slurries.
Non-dusting granulates may be produced, e.g., as disclosed in US 4,106,991 A and
US 4,661 ,452 A and may optionally be coated by methods known in the art. Examples of waxy coating materials are poly(ethylene oxide) products (polyethyleneglycol, “PEG”) with mean molar weights of from about 1000 to about 20000; ethoxylated nonylphenols having from 16 to 50 ethylene oxide units; ethoxylated fatty alcohols in which the alcohol contains from 12 to 20 carbon atoms and in which there are 15 to 80 ethylene oxide units; fatty alcohols; fatty acids; and mono- and di- and triglycerides of fatty acids. Examples of film-forming coating materials suitable for application by fluid bed techniques are given in GB 1 ,483,591 A. Liquid enzyme preparations may, for instance, be stabilized by adding a polyol such as propylene glycol, a sugar or sugar alcohol, lactic acid or boric acid according to established methods. Protected enzymes may be prepared according to the method disclosed in EP 0 238216 A1 .
1 .5.6 Builders
Composition ZG optionally comprises at least one builder Bz. In those embodiments where composition ZG comprises at least one builder Bz, the amount of all builders Bz in composition ZG is preferably between 0.1 wt.-% to 10 wt.-%, preferably 1 to 7 wt.-%, relative to the total weight of composition ZG.
Preferred builder Bz are described under item 1 .4.3
1 .5.7 Solvents
In a preferred embodiment, composition ZG may comprise at least one solvent selected from water or non-aqueous solvent, preferably non-aqueous solvent.
Suitable non-aqueous solvents are described under item 1.4.4.
In those embodiments where composition ZG comprises at least one non-aqueous solvent, the amount of all non-aqueous solvent in composition ZG is preferably between 0.5 wt.-% to 15 wt.-%, preferably 1 wt.-% to 10 wt.-%, relative to the total weight of composition ZG.
1 .5.8 Preservatives
Composition ZG optionally comprises at least one preservative.
In those embodiments where composition ZG comprises at least one preservative, the amount of all preservatives in composition ZG is preferably between 0.001 wt.-% to 2 wt.-%, preferably 0.01 to 0.1 wt.-%, relative to the total weight of composition ZG.
Suitable non-aqueous solvents are described under item 1.4.5.
1 .5.9 Benefit agent
Composition ZG optionally comprise at least one encapsulate comprising a benefit agent, preferably a sensorial benefit agent. The preferred benefit agent is a perfume. A benefit agent may be provided in an encapsulate. Composition ZG optionally also comprises an unconfined (also called non-encapsulated) benefit agent, for example a volatile benefit agent. Where the volatile benefit agent is a perfume, the perfumes are suitable for use as the encapsulated volatile benefit agent and also as the unconfined perfume component.
Preferred encapsulates in this context are described under item 1 .4.6
In those embodiments where composition ZG comprises at least one benefit agent, the total amount of benefit agent is preferably from 0.01 wt.-% to 10 wt.-%, more preferably from 0.05 wt.-% to
5 wt.-%, even more preferably from 0.1 wt.-% to 4.0 wt.-%, most preferably from 0.15 wt.-% to 4.0 wt.-%, based on the total weight of the composition ZG.
Composition ZG optionally comprises at least one polymer for use in detergents that are different from the polymers described before.
In those embodiments where composition ZG comprises at least one polymer, the amount of all polymers in composition ZG is preferably between 0.05 wt.-% to 8 wt.-%, preferably 0.1 to 5 wt.-%, relative to the total weight of composition ZG.
Preferred polymers are described under item 1.4.7.
1.5.11 Other Additives
Composition ZG comprise at least one further additives selected from the group consisting of bleaching systems, anti-redeposition aids, fiber protection agents, soil release agents, dye transfer inhibitors, fabric hueing agents, blueing dyes, enzyme stabilizing agents like boric acid,
pH-regulators, emollients, emulsifiers, thickeners/ viscosity regulators /stabilizers, UV photoprotective filters, antioxidants, hydrotropes (or polyols), solids and fillers, film formers, pearlescent additives, deodorant and antiperspirant active ingredients, insect repellents, selftanning agents, preservatives, conditioners, perfumes, dyes, odour absorbers, cosmetic active ingredients, care additives, superfatting agents, solvents, malodor removers.
Substances which can be used as exemplary representatives of the individual groups are known to the person skilled in the art and can be found for example in US 2011/0091399 A1 .
1.5.12 Form of composition ZG
The form in which composition ZG is provided is not particularly limited and depends on the area of use.
Composition ZG may be in any convenient form, e.g., a bar, a homogenous tablet, a tablet having two or more layers, a pouch having one or more compartments, a regular or compact powder, a granule, a paste, a gel, or a regular, compact or concentrated liquid. There are a number of detergent formulation forms such as layers (same or different phases), pouches, as well as forms for machine dosing unit.
Pouches can be configured as single or multicompartments. It can be of any form, shape and material which is suitable for hold the composition, e.g. without allowing the release of the composition from the pouch prior to water contact. The pouch is made from water soluble film which encloses an inner volume. Said inner volume can be divided into compartments of the pouch. Preferred films are polymeric materials preferably polymers which are formed into a film or sheet. Preferred polymers, copolymers or derivates thereof are selected polyacrylates, and water soluble acrylate copolymers, methyl cellulose, carboxy methyl cellulose, sodium dextrin, ethyl cellulose, hydroxyethyl cellulose, hydroxypropyl methyl cellulose, maltodextrin, poly methacrylates, most preferably polyvinyl alcohol copolymers and, hydroxypropyl methyl cellulose (HPMC). Preferably the level of polymer in the film, for example PVA, is at least about 60%. Preferred average molecular weight will typically be from about 20,000 to about 150,000. Films can also be of blend compositions comprising hydrolytically degradable and water soluble polymer blends such as polyactide and polyvinyl alcohol (known under the Trade reference M8630 as sold by Chris Craft In. Prod. Of Gary, Ind., US) plus plasticisers like glycerol, ethylene glycerol, Propylene glycol, sorbitol and mixtures thereof. The pouches can comprise a solid laundry detergent composition or part components and/or a liquid cleaning composition or part components separated by the water- soluble film. The compartment for liquid components can be different in composition than compartments containing solids, see for example US 2009/0011970 A1 .
Detergent ingredients can be separated physically from each other by compartments in water dissolvable pouches or in different layers of tablets. Thereby negative storage interaction between components can be avoided. Different dissolution profiles of each of the compartments can also
give rise to delayed dissolution of selected components in the wash solution.
Composition ZG can be in form of a laundry soap bar and used for hand washing laundry, fabrics and/or textiles. The term laundry soap bar includes laundry bars, soap bars, combo bars, syndet bars and detergent bars. The types of bar usually differ in the type of surfactant they contain, and the term laundry soap bar includes those containing soaps from fatty acids and/or synthetic soaps. The laundry soap bar has a physical form which is solid and not a liquid, gel or a powder at room temperature. The term solid is defined as a physical form which does not significantly change over time, i.e. if a solid object (e.g. laundry soap bar) is placed inside a container, the solid object does not change to fill the container it is placed in. The bar is a solid typically in bar form but can be in other solid shapes such as round or oval.
The detergent composition according to the instant invention can be formulated as a granular detergent as described in WO 2009/092699 A1 , EP 1 705 241 A1 , EP 1 382 668 A1 , WO 2007/001262 A1 , US 6,472,364 B1 , WO 2004/074419 A2 or WO 2009/102854 A1 .
Composition ZG may also be in the form of a liquid or gel detergent. This may be aqueous, typically containing at least 20% by weight water, with the percentages referring to the total composition. Other types of liquids, including without limitation, alkanols, amines, diols, ethers and polyols may be included in an aqueous liquid or gel. An aqueous liquid or gel detergent composition may contain from 0-30 wt.-% organic solvent, with the percentages referring to the total composition. A liquid or gel detergent may be non-aqueous.
In those embodiments, composition ZG according to the invention is characterized in that the pH of the composition at 25°C is from 3.0 to 9, preferably from 4.0 to 7 and particularly preferably from 5.0 to 6.6.
2. Method of washing a woolen article Aw
In a second aspect, the present invention relates to a method of washing at least one woolen article Aw, comprising the steps of a. contacting the at least one woolen article Aw with an aqueous mixture L comprising at least one biosurfactant SBIO, b. at least partially separating L from Aw, c. optionally rinsing Aw with water, wherein the temperature of the aqueous mixture L, for at least as long as the aqueous mixture L is contacted with the at least one woolen article Aw, is in a range of from > 0 °C to 45 °C, in particular in a range of from 1 °C to 40 °C, preferably in a range of from 5 °C to 35 °C, more preferably in a range of from 10 °C to 30 °C, more preferably in a range of from 15 °C to 30 °C, even more preferably in a range of from 20 °C to 30 °C, even more preferably in a range of from 25 °C to 30 °C, wherein the biosurfactant SBIO is selected from the group consisting of rhamnolipids, glucolipids, sophorolipids, and wherein the biosurfactant SBIO, in particular, is selected from the group consisting of rhamnolipids, sophorolipids, and wherein the biosurfactant SBIO is preferably selected from rhamnolipids.
2.1 Woolen Article Aw
In the context of the method according to the second aspect of the invention, a “woolen article Aw” is to be understood as any article which comprises wool, preferably an article comprising at least 10 weight-% (= “wt.-%”) wool, more preferably at least 20 wt.-% wool, more preferably at least 30 wt.-% wool, more preferably at least 40 wt.-% wool, more preferably at least 50 wt.-% wool, more preferably at least 60 wt.-% wool, more preferably at least 70 wt.-% wool, more preferably at least 80 wt.-% wool, more preferably at least 90 wt.-% wool, more preferably at least 99 wt.-% wool, wherein the weight-% relate to the total mass of the woolen article. Most preferably, the article Aw essentially consists of wool.
The woolen article Aw in particular is an article of clothing, preferably selected from pants, shirt, socks, hosiery, scarfs, gloves, shoes, sweaters, suits, hats, caps, bonnets. It may, in addition, also be selected from the group consisting of curtains, napkins, bed linen, tablecloth, blanket, bedspread, duvet cover.
In the context of this invention, “wool” refers to the textile fiber of a mammal, in particular of livestock. In particular, it refers to the textile fiber of a mammal selected from the group consisting of sheep, goat (the goat being in particular selected from the group of cashmere goat, angora
goat), rabbit (the rabbit being in particular an angora rabbit), camelide (the camelide being in particular selected from the group of llama, vikuna, alpaca, guanaco), yak, beaver, otter, guinea pig, horse, donkey, chinchilla. Preferably, it refers to the textile fiber of a mammal selected from the group consisting of sheep, goat, most preferably of sheep.
It was now surprisingly found that in the method according to the second aspect of the invention, the shrinking of the at least one woolen article Aw is reduced during washing of Aw in the aqueous mixture L comprising the at least one woolen article Aw and the at least one biosurfactant SBIO-
The shrinking as well as the extent of shrinking may be determined as described under chapter 1 .2 above. Preferably, Assay X is used to determine whether a certain substance (abbreviated as “X”) reduces the shrinking of a woolen article Aw (as well as the extent of shrinking) during washing. Assay X is hence the preferred test to determine the reduction of shrinking of the at least one woolen article Aw during washing in the method according to the second aspect of the invention, in particular in an aqueous mixture L, as well as the extent of shrinking of the at least one woolen article Aw during washing, in particular in an aqueous mixture L.
2.2 Step a.
In step a. of the method according to the second aspect of the invention, the at least one woolen article Aw is contacted with an aqueous mixture L comprising at least one biosurfactant SBIO-
2.2.1 Biosurfactant SBIO
In the context of the method according to the second aspect of the invention, the “biosurfactant Ssio” is selected from the group consisting of glucolipids, rhamnolipids, sophorolipids, in particular selected from the group of sophorolipids, rhamnolipids, preferably from the group of rhamnolipids.
Glucolipids, rhamnolipids, and sophorolipids are glycolipids. Such glycolipids are described in the art together with their syntheses, for example in EP 0499434 A1 (glucolipids are referred to as “glucose lipids” in this document), DE 196 48 439 A1 , DE 196 00 743 A1 . WO 03/006146 A1 , US 2008/0213194 A1 , JP H01 -304034 A1 , CN 1337439 A describe further methods for the synthesis of rhamnolipids. WO 03/002700 A1 , US 4,305,961 A, US 7,556,654 B1 describe further methods for the synthesis of sophorolipids.
2.2. 1. 1 Glucolipids
In the context of the method according to the second aspect of the present invention, the term “glucolipid” is, in particular, to be understood as referring to a structure according to formula (I), even more preferred a structure according to formula (II):
In formulae (I) and (II), mGL = 3, 2, 1 or 0, preferably 1 or 0.
Residues R1GL and R2GL are, independently of one another, an organic radical having 2 to 24 carbon atoms, preferably 5 to 20, more preferably 7 to 15, even more preferably 7, carbon atoms, wherein preferably R1GL and R2GL are independently of one another selected from the group consisting of optionally substituted alkyl radicals with 2 to 24 preferably 5 to 20, more preferably 7 to 15, even more preferably 7, carbon atoms, wherein hydroxy substituted alkyl radicals are preferred substituted alkyl radicals, optionally substituted alkenyl radicals with 2 to 24 carbon atoms, preferably 5 to 20, more preferably 7 to 15, even more preferably 7, carbon atoms, wherein hydroxy substituted alkenyl radicals are preferred substituted alkenyl radicals, wherein more preferably R1GL and R2GL are independently of one another selected from the group consisting of pentenyl, heptenyl, nonenyl, undecenyl, tridecenyl and -(CH2)oCH3 where o = 1 to 23, preferably 4 to 12. Most preferably, R1GL and R2GL are each n-heptyl.
Distinct glucolipids for mGL = 0 are abbreviated according to the following nomenclature:
“GL-CX” is understood as meaning glucolipids of the general formulae (I) or (II) in which mGL = 0 and in which the radical R1GL = (CH2)o-CH3 where o = X-4.
Distinct glucolipids for mGL = 1 are abbreviated according to the following nomenclature: “GL-CXCY” is understood as meaning glucolipids of the general formulae (I) or (II) in which mGL = 1 and in which one of the radicals R1GL and R2GL = (CH2)o-CH3 where o = X-4 and the remaining radical R1GL or R2GL = (CH2)o-CH3 where o = Y-4.
The nomenclature used thus does not differentiate between “CXCY” and “CYCX”.
If one of the aforementioned indices X and/or Y is provided with “:Z”, then this means that the respective radical R1GL and/or R2GL = an unbranched, unsubstituted hydrocarbon radical with X-3 or Y-3 carbon atoms having Z double bonds.
The curvy bond in structure (I) [and also structures (III), (V), (VII), (IX), (XI) described hereinafter] implies that the respective substituent may be axial or equatorial, preferably is equatorial.
Alkyl radicals may be branched or linear.
Alkenyl radicals may be branched or linear and preferably contain between one to three double bonds.
In those cases where structures according to formulae (I) and (II) comprise more than one residue R2GL, these residues R2GL may be identical or different from one another.
In those embodiments in which glucolipids are used as biosurfactants SBIO in the method according to the second aspect of the invention, it is preferred that 1 wt.-% to 30 wt.-%, preferably 5 wt.-% to 25 wt.-%, particularly preferably 10 wt.-% to 20 wt.-%, of all glucolipids that are used as biosurfactants SBIO in the method according to the second aspect of the invention, are GL-C8C10, where the “wt.-%” (= weight-%; percentages by weight) refers to the total weight of all the glucolipids that are used as biosurfactants SBIO in the method according to the second aspect of the invention.
In those embodiments in which glucolipids are used as biosurfactants SBIO in the method according to the second aspect of the invention, it is alternatively preferred that 0.5 wt.-% to 20 wt.-%, preferably 3 wt.-% to 17 wt.-%, particularly preferably 5 wt.-% to 15 wt-%. of all glucolipids that are used as biosurfactants SBIO in the method according to the second aspect of the invention, are GL- C10C12:1 , where the wt.-% refers to the total weight of all the glucolipids that are used as biosurfactants SBIO in the method according to the second aspect of the invention.
In those embodiments in which glucolipids are used as biosurfactants SBIO in the method according to the second aspect of the invention, it is alternatively preferred that 0.5 wt.-% to 20 wt.-%, preferably 2 wt.-% to 15 wt.-%, particularly preferably 3 wt.-% to 12 wt.-%, of all the glucolipids that are used as biosurfactants SBIO in the method according to the second aspect of the invention, are GL-C10C12, where the wt.-% refers to the total weight of all the glucolipids that are used as biosurfactants SBIO in the method according to the second aspect of the invention.
In those embodiments in which glucolipids are used as biosurfactants SBIO in the method according to the second aspect of the invention, it is alternatively preferred that
1 wt.-% to 30 wt.-%, preferably 5 wt.-% to 25 wt.-%, particularly preferably 10 wt.-% to 20 wt.-%, of all the glucolipids that are used as biosurfactants SBIO in the method according to the second aspect of the invention, are GL-C8C10,
0.5 wt.-% to 20 wt.-%, preferably 3 wt.-% to 17 wt.-%, particularly preferably 5 wt.-% to 15 wt.-%, of all the glucolipids that are used as biosurfactants SBIO in the method according to the second aspect of the invention, are GL-C10C12:1 ,
0.5 wt.-% to 20 wt.-%, preferably 2 wt.-% to 15 wt.-%, particularly preferably 3 wt.-% to 12 wt.-%, of all the glucolipids that are used as biosurfactants SBIO in the method according to the second aspect of the invention, are GL-C10C12, where the wt.-% refers to the total weight of all the glucolipids that are used as biosurfactants SBIO in the method according to the second aspect of the invention.
In those embodiments in which glucolipids are used as biosurfactants SBIO in the method according to the second aspect of the invention, it is alternatively preferred that 10 wt.-% to 20 wt.-% of all the glucolipids that are used as biosurfactants SBIO in the method according to the second aspect of the invention, are GL-C8C10,
5 wt.-% to 15 wt.-%, of all the glucolipids that are used as biosurfactants SBIO in the method according to the second aspect of the invention, are GL-C10C12:1 ,
3 wt.-% to 12 wt.-%, of all the glucolipids that are used as biosurfactants SBIO in the method according to the second aspect of the invention, are GL-C10C12, where the wt.-% refers to the total weight of all the glucolipids that are used as biosurfactants SBIO in the method according to the second aspect of the invention.
Over and above this, in those embodiments in which glucolipids are used as biosurfactants SBIO in the method according to the second aspect of the invention, it is alternatively preferred that 0 wt.-% to 5 wt.-%, preferably 0.01 wt.-% by weight to 4 wt.-%, particularly preferably 0.1 wt.-% to 3 wt.-%, of all the glucolipids that are used as biosurfactants SBIO in the method according to the second aspect of the invention, are GL-C10, where the wt.-% refers to the total weight of all the glucolipids that are used as biosurfactants SBIO in the method according to the second aspect of the invention.
2.2. 1.2 Rhamnolipids
In the context of the method according to the second aspect of the present invention, the term "rhamnolipid" is, in particular, to be understood as referring to compounds of the general formula (III) and salts thereof, preferably compounds according to the general structure (IV) and salts thereof,
In formulae (III) and (IV), mRL = 2, 1 or 0, preferably 1 or 0. nRL = 1 or 0.
Residue R1RL and R2RL are independently of one another, an organic radical having 2 to 24 carbon, preferably 5 to 13, more preferably 7 to 10, even more preferably 7, carbon atoms, wherein preferably R1RL and R2RL are independently of one another selected from the group consisting of optionally substituted alkyl radicals with 2 to 24, preferably 5 to 13, more preferably 7 to 10, even more preferably 7, carbon atoms, wherein hydroxy substituted alkyl radicals are preferred substituted alkyl radicals, optionally substituted alkenyl radicals with 2 to 24, preferably 5 to 13, more preferably 7 to 10, even more preferably 7, carbon atoms, wherein hydroxy substituted alkenyl radicals are preferred substituted alkenyl radicals, wherein more preferably R1 RL and R2RL are independently of one another selected from the group consisting of pentenyl, heptenyl, nonenyl, undecenyl, tridecenyl and -(CH2)o-CH3 where o = 1 to 23, preferably 4 to 12. Most preferably, R1RL and R2RL are each n-heptyl.
If nRL = 1 , the glycosidic bond between the two rhamnose units is preferably in the a-configuration.
The optically active carbon atoms of the fatty acids are preferably present as R-enantiomers (e.g. (R)-3-{(R)-3-[2-O-(a-L-rhamnopyranosyl)-a-L-rhamnopyranosyl]oxydecanoyl}oxydecanoate).
The term "di-rhamnolipid" in the context of the present invention is understood to mean compounds of the general formulae (III) and (IV) or salts thereof, where nRL = 1 .
The term "mono-rhamnolipid" in the context of the present invention is understood to mean compounds of the general formulae (III) and (IV) or salts thereof, where nRL = 0.
Distinct rhamnolipids are abbreviated according to the following nomenclature:
"diRL-CXCY" are understood to mean di-rhamnolipids of the general formulae (III) and (IV), in which one of the residues R1RL and R2RL = -(CH2)o-CH3 where o = X-4 and the remaining residue R1 or R2 = -(CH2)O-CH3 where o = Y-4.
"monoRL-CXCY" are understood to mean mono-rhamnolipids of the general formulae (III) and (IV), in which one of the residues R1RL and R2RL = -(CH2)o-CH3 where o = X-4 and the remaining residue RIRL OR R2RL = _(CH2)O-CH3 where o = Y-4.
The nomenclature used therefore does not distinguish between "CXCY" and "CYCX". For rhamnolipids where mRL=0, the terms “monoRL-CX” or “diRL-CX” are used accordingly. If one of the abovementioned indices X and/or Y is provided with ":Z", this signifies that the respective residue R1RL and/or R2RL is equal to an unbranched, unsubstituted hydrocarbon residue having X-3 or Y-3 carbon atoms having Z double bonds.
Methods for preparing the relevant rhamnolipids are disclosed, for example, in EP 2 786 743 A1 and EP 2 787 065 A1 . Rhamnolipids applicable in the context of the instant invention can also be produced by fermentation of Pseudomonas, especially Pseudomonas aeruginosa, which are preferably non genetically modified cells, a technology already disclosed in the eighties, as documented e.g. in EP 0 282 942 A1 and DE 41 27 908 A1 . Rhamnolipids produced in Pseudomonas aeruginosa cells which have been improved for higher rhamnolipid titres by genetical modification can also be used in the context of the instant invention; such cells have for example been disclosed by Lei et al. in Biotechnol Lett. 2020 Jun;42(6):997-1002.
Rhamnolipids produced by Pseudomonas aeruginosa are commercially available from Jeneil Biotech Inc., e.g. under the tradename Zonix, from Logos Technologies (technology acquired by Stepan), e.g. under the tradename NatSurFact, from Biotensidion GmbH, e.g. under the tradename Rhapynal, from AGAE technologies, e.g. under the name R90, R95, R95Md, R95Dd, from Locus Bio-Energy Solutions and from Shanghai Yusheng Industry Co. Ltd., e.g. under the tradename Bio- 201 Glycolipids.
In those embodiments in which rhamnolipids are used as biosurfactants SBIO in the method according to the second aspect of the invention, it is alternatively preferred to use mixtures of mono-rhamnolipids and di-rhamnolipids, wherein even more preferably the ratio of the weight of all mono-rhamnolipids used in the method according to the second aspect of the invention to the weight of all di-rhamnolipids used in the method according to the second aspect of the invention is in the range of from 9 : 1 to 1 : 9, preferably of from 8 : 2 to 2 : 8, more preferably of from 7 : 3 to 3 : 7, more preferred of from 6 : 4 to 4 : 6, most preferred is 1 : 1 .
Mono-rhamnolipids are compounds according to structures (III), preferably (IV), wherein nRL = 0. Di-rhamnolipids are compounds according to structures (III), preferably (IV), wherein nRL = 1 .
In those embodiments in which rhamnolipids are used as biosurfactants SBIO in the method according to the second aspect of the invention, it is preferred that the rhamnolipids comprise
56 wt.-% to 95 wt.-%, preferably 60 wt.-% to 80 wt.-%, particularly preferably 66 wt.-% to 70 wt.-%, of diRL-C10C10, where the wt.-% refers to the total weight of all the rhamnolipids that are used as biosurfactants SBIO in the method according to the second aspect of the invention.
In those embodiments in which rhamnolipids are used as biosurfactants SBIO in the method according to the second aspect of the invention, it is alternatively preferred that 0.5 wt.-% to 15 wt.-%, preferably 3 wt.-% to 12 wt.-%, particularly preferably 5 wt.-% to 10 wt.-%, of the rhamnolipids are diRL-C10C12:1 , where the wt.-% refers to the total weight of all the rhamnolipids that are used as biosurfactants SBIO in the method according to the second aspect of the invention.
In those embodiments in which rhamnolipids are used as biosurfactants SBIO in the method according to the second aspect of the invention, it is alternatively preferred that 0.5 wt.-% to 25 wt.-%, preferably 3 wt.-% to 15 wt.-%, particularly preferably 5 wt.-% to 12 wt.-%, of the rhamnolipids are diRL-C10C12, where the wt.-% refers to the total weight of all the rhamnolipids that are used as biosurfactants SBIO in the method according to the second aspect of the invention.
In those embodiments in which rhamnolipids are used as biosurfactants SBIO in the method according to the second aspect of the invention, it is alternatively preferred that 0.1 wt.-% to 25 wt.-%, preferably 2 wt.-% to 10 wt.-%, particularly preferably 4 wt.-% to 8 wt.-%, of the rhamnolipids are diRL-C8C10, where the wt.-% refers to the total weight of all the rhamnolipids that are used as biosurfactants SBIO in the method according to the second aspect of the invention.
In those embodiments in which rhamnolipids are used as biosurfactants SBIO in the method according to the second aspect of the invention, it is even further preferred that
0.1 wt.-% to 5 wt.-%, preferably 0.5 wt.-% to 3 wt.-%, particularly preferably 0.5 wt.-% to 2 wt.-%, of the rhamnolipids are monoRL-C8C10 and/or, preferably and,
0.1 wt.-% to 5 wt.-%, preferably 0.5 wt.-% to 3 wt.-%, particularly preferably 0.5 wt.-% to 2 wt.-%, of the rhamnolipids are monoRL-C10C10, where the wt.-% refers to the total weight of all the rhamnolipids that are used as biosurfactants SBIO in the method according to the second aspect of the invention.
In those embodiments in which rhamnolipids are used as biosurfactants SBIO in the method according to the second aspect of the invention, it is alternatively preferred that 10 wt.-% to 30 wt.-%, preferably 20 wt.-% to 30 wt.-%, particularly preferably 25 wt.-% to 30 wt.-%, of the rhamnolipids are monoRL-C10C10, where the wt.-% refers to the total weight of all the rhamnolipids that are used as biosurfactants SBIO in the method according to the second aspect of the invention.
In those embodiments in which rhamnolipids are used as biosurfactants SBIO in the method according to the second aspect of the invention, it is alternatively preferred that 10 wt.-% to 30wt.-%, preferably 12 wt.-% to 25 wt.-%, particularly preferably 15 wt.-% to 20 wt.-%, of the rhamnolipids are diRL-C10C10, where the wt.-% refers to the total weight of all the rhamnolipids that are used as biosurfactants SBIO in the method according to the second aspect of the invention.
In those embodiments in which rhamnolipids are used as biosurfactants SBIO in the method according to the second aspect of the invention, it is alternatively preferred that 10 wt.-% to 30 wt.-%, preferably 12 wt.-% to 25 wt.-%, particularly preferably 15 wt.-% to 20 wt.-%, of the rhamnolipids are monoRL-C8C10, where the wt.-% refers to the total weight of all the rhamnolipids that are used as biosurfactants SBIO in the method according to the second aspect of the invention.
In those embodiments in which rhamnolipids are used as biosurfactants SBIO in the method according to the second aspect of the invention, it is alternatively preferred that 3 wt.-% to 25 wt.-%, preferably 5 wt.-% to 20 wt.-%, particularly preferably 10 wt.-% to 15 wt.-%, of the rhamnolipids are monoRL-C10C12:1 , where the wt.-% refers to the total weight of all the rhamnolipids that are used as biosurfactants SBIO in the method according to the second aspect of the invention.
In those embodiments in which rhamnolipids are used as biosurfactants SBIO in the method according to the second aspect of the invention, it is alternatively preferred that 1 wt.-% to 15 wt.-%, preferably 2 wt.-% to 10 wt.-%, particularly preferably 3 wt.-% to 8 wt.-%, of the rhamnolipids are diRL-C10C12, where the wt.-% refers to the total weight of all the rhamnolipids that are used as biosurfactants SBIO in the method according to the second aspect of the invention.
2.2. 1.3 Sophorolipids
In the context of the method according to the second aspect of the present invention, the term “sophorolipids” is in particular to be understood as referring to compounds of the general formulae (V), (VII) and salts thereof, preferably compounds of the general formulae (VI), (VIII) and salts thereof:
Formulae (V), (VII) represent the acid form, Formulae (VI), (VIII) represent the lactone form.
In formulae (V) and (VI), nSL = 1 or 0.
In formulae (V), (VI), (VII), and (VIII), R1SL = H or -CO-CH3,
R2SL = H or -CO-CH3,
R3si_ = a divalent organic moiety which comprises 6 to 32 carbon atoms, preferably 12 to 20, more preferably 14 to 16, most preferably 15.
R3SL preferably is an optionally substituted, divalent hydrocarbon moiety comprising 6 to 32 carbon atoms, wherein hydroxy substituted hydrocarbon moieties are preferred as substituted hydrocarbon moieties,
R3SI_ more preferably is selected from the group consisting of
• optionally substituted alkylene radicals with 6 to 32, preferably 12 to 20, more preferably 14 to 16, most preferably 15 carbon atoms, wherein hydroxy-substituted alkylene radicals are preferred substituted alkylene radicals, and wherein it is preferred that the optionally substituted alkylene radicals are unbranched,
• optionally substituted alkenylene radicals with 6 to 32, preferably 12 to 20, more preferably 14 to 16, most preferably 15 carbon atoms, wherein hydroxy-substituted alkenylene radicals are preferred substituted alkenylene radicals, and wherein it is preferred that the optionally substituted alkenylene radicals are unbranched, and wherein it is preferred that the optionally substituted alkenylene radicals comprise one to three double or triple bonds.
R3SL even more preferably is selected from the group consisting of
• unbranched or branched, preferably unbranched, alkylene radicals with 6 to 32, preferably 12 to 20, more preferably 14 to 16, most preferably 15, carbon atoms,
• unbranched or branched, preferably unbranched, alkylene radicals with 6 to 32 preferably 12 to 20, more preferably 14 to 16, most preferably 15, carbon atoms carrying at least one hydroxy group, preferably one hydroxy group,
• unbranched or branched, preferably unbranched, alkenylene radicals with 6 to 32, preferably 12 to 20, more preferably 14 to 16, most preferably 15 carbon atoms, wherein the alkenylene radicals preferably comprise one to three double or triple bonds, wherein the alkenylene radicals more preferably comprise one to three double bonds, even more preferably one double bond,
• unbranched or branched, preferably unbranched, alkenylene radicals with 6 to 32, preferably 12 to 20, more preferably 14 to 16, most preferably 15, carbon atoms carrying at least one hydroxy group, preferably one hydroxy group, wherein the alkenylene radicals preferably comprise one to three double or triple bonds, wherein the alkenylene radicals more preferably comprise one to three double bonds, even more preferably one double bond.
R4SL = H, CH3 or a monovalent organic radical which comprises 2 to 10 carbon atoms.
R4SL is preferably selected from the group consisting of
• H,
• CH3,
• optionally substituted alkyl radicals with 2 to 10 carbon atoms, wherein hydroxy-substituted alkyl radicals are preferred substituted alkyl radicals, and wherein it is preferred that the optionally substituted alkyl radicals are unbranched,
• optionally substituted alkenyl radicals with 2 to 10 carbon atoms, wherein hydroxysubstituted alkenyl radicals are preferred substituted alkenyl radicals, and wherein it is preferred that the optionally substituted alkenyl radicals are unbranched, and wherein it is preferred that the optionally substituted alkenyl radicals comprise one to three double or triple bonds.
R4SL is more preferably selected from the group consisting of
• H,
• CH3,
• unbranched or branched, preferably unbranched, alkyl radicals with 2 to 10 carbon atoms,
• unbranched or branched, preferably unbranched, alkyl radicals with 2 to 10 carbon atoms carrying at least one hydroxy group, preferably one hydroxy group,
• unbranched or branched, preferably unbranched, alkenyl radicals with 2 to 10 carbon atoms, wherein the alkenyl radicals preferably comprise one to three double or triple bonds, wherein the alkenyl radicals more preferably comprise one to three double bonds, even more preferably one double bond,
• unbranched or branched, preferably unbranched, alkenyl radicals with 2 to 10 carbon atoms carrying at least one hydroxy group, preferably one hydroxy group, wherein the alkenyl radicals preferably comprise one to three double or triple bonds, wherein the alkenyl radicals more preferably comprise one to three double bonds, even more preferably one double bond.
R4SL is most preferably selected from the group consisting of H, Methyl, Ethyl.
In an even more preferred embodiment, the term “sophorolipids” is to be understood as referring to compounds of the general formulae (IX), (XI) and salts thereof, preferably compounds of the general formulae (X), (XII) and salts thereof:
Formulae (IX), (X) represent the acid form, Formulae (XI), (XII) represent the lactone form. nSL, R1SL, R2SL, R4SL have the same meaning as described for formulae (V), (VII), (VI), (VIII).
Sophorolipids may be used in accordance with the invention in their acid form or their lactone form. Preferred compositions in the context of the method according to the second aspect of the present invention comprise a sophorolipid in which the ratio by weight of lactone form to acid form is in the range of 20:80 to 80:20, especially preferably in the ranges of 30:70 to 40:60.
To determine the content of sophorolipids in the acid or lactone form in a formulation, refer to EP 1 411 111 B1 , page 8, paragraph [0053],
Sophorolipids may be obtained as described in EP 1 411 111 A1 , paragraphs [0021], [0022]. 2.2.2 Aqueous mixture L
In step a. of the method according to the second aspect of the present invention, the at least one woolen article Aw is contacted with an aqueous mixture L comprising at least one biosurfactant SBIO- The method according to the second aspect of the invention is a method washing of washing
at least one woolen article Aw- The aqueous mixture L is hence, in particular, the washing solution in which the at least one article Aw is washed.
“The woolen article Aw is contacted with the aqueous mixture L comprising at least one biosurfactant SBIO” is to be understood that, in step a. of the method according the second aspect of the invention, the woolen article Aw is brought in direct contact with the aqueous mixture L comprising at least one biosurfactant SBIO, “in direct contact” meaning “wetting” of the woolen article Aw with the aqueous mixture L.
In a preferred embodiment of step a. of the method according the second aspect of the invention, the article Aw is at least partially immersed, even more preferably fully immersed into the aqueous mixture L.
In an even more preferred embodiment, the article Aw is moved in the aqueous mixture L during step a. of the method according the second aspect of the invention.
As Aw is contacted with the aqueous mixture L during washing of Aw, this means in particular that during washing of Aw in the aqueous mixture L during step a. of the method according the second aspect of the invention, at least one impurity I which is absorbed or adsorbed on the article Aw is at least partially desorbed and thus removed from the article Aw and dispersed or dissolved, in particular dispersed, in aqueous mixture L during step a. of the method according the second aspect of the invention. In other words: during step a. of the method according the second aspect of the invention, the mixture L is preferably brought in direct contact with at least a part of the surface of article Aw, onto which at least one impurity I is absorbed or adsorbed.
Therefore, in particular, during the use according to step a. of the method according the second aspect of the invention, a mixture L is obtained wherein in the water comprised by aqueous mixture L, at least one impurity I in dissolved or dispersed, preferably dispersed.
The concentration of impurities I in the water comprised by aqueous mixture L therefore augments during step a. of the method according the second aspect of the invention. In a preferred embodiment, the aqueous mixture L that is employed during step a. of the method according the second aspect of the invention (i.e. before it contacts the at least one woolen article Aw) is essentially free of impurity I.
Preferably, the mixture L that is obtained during the use according to step a. of the method according the second aspect of the invention, is a dispersion, in which the at least one impurity I is the dispersed phase in the water W comprised by aqueous mixture L.
The impurity I may either be in the form of a liquid (e.g. droplet) or a particle, and preferably is a particle.
In a preferred embodiment, the impurity I is at least one impurity which may be inorganic or organic, in particular inorganic, preferably clay, more preferably kaolin.
Organic impurities are preferably selected from oils and fats, which may be of vegetable, animal or synthetic origin, food based particles, soil, pollen, sebum, body fluids, for example, blood, sperm, sweat, urine, feces, liquor.
Inorganic impurities are preferably selected from the group consisting of carbon black, fine dust, plastic particles (e.g. microplastic particles), metal oxides (preferably iron oxide), silica (such as sand), clay, dyes (such as pigments), more preferably clay, in particular kaolin.
The beneficial effect of the method according to the second aspect of the invention is the reduction of shrinking of the at least one article Aw during washing in the aqueous mixture L comprising the at least one biosurfactant SBIO- Since the tendency of woolen articles to shrink is especially pronounced at temperatures above 45 °C, the best effect of the present invention is obtained when the aqueous mixture L has a temperature at or below 45 °C for at least as long as the aqueous mixture L contacts the at least one woolen article Aw-
Hence, the temperature of the aqueous mixture L, for at least as long as the aqueous mixture L is contacted with the at least one woolen article Aw, is in a range of from > 0 °C to 45 °C, in particular in a range of from 1 °C to 40 °C, preferably in a range of from 5 °C to 35 °C, more preferably in a range of from 10 °C to 30 °C, more preferably in a range of from 15 °C to 30 °C, even more preferably in a range of from 20 °C to 30 °C, even more preferably in a range of from 25 °C to 30 °C.
“For at least as long as the aqueous mixture L is contacted with the at least one woolen article Aw” in particular refers to the time during which Aw is contacted with the aqueous mixture L during step a. It is advantageous to avoid contacting Aw with any temperature above 45 °C throughout the method according to the second aspect of the invention. Thus, it is preferred that the temperature of the aqueous mixture L, for at least as long as it is at least partially separated from Aw in step b., is also in a range of from > 0 °C to 45 °C, in particular in a range of from 1 °C to 40 °C, preferably in a range of from 5 °C to 35 °C, more preferably in a range of from 10 °C to 30 °C, more preferably in a range of from 15 °C to 30 °C, even more preferably in a range of from 20 °C to 30 °C, even more preferably in a range of from 25 °C to 30 °C.
Likewise, it is even more preferred that during optional step c., the water which is used for rinsing Aw has a temperature, for at least as long as this water contacts the at least one woolen article Aw, in a range of from > 0 °C to 45 °C, in particular in a range of from 1 °C to 40 °C, preferably in a range of from 5 °C to 35 °C, more preferably in a range of from 10 °C to 30 °C, more preferably in a range of from 15 °C to 30 °C, even more preferably in a range of from 20 °C to 30 °C, even more preferably in a range of from 25 °C to 30 °C.
The aqueous mixture L, in particular, comprises the at least one biosurfactant SBIO in an amount in the range of from 0.000001 wt.-% to 50 wt.-%, preferably in the range of from 0.00001 wt.-% to
30 wt.-%, more preferably in the range of from 0.00005 wt.-% to 20 wt.-%, more preferably in the range of from 0.00009 wt.-% ppm to 15 wt.-%, more preferably in the range of from 0.0001 wt.-% to 14 wt.-%, more preferably in the range of from 0.001 wt.-% to 13 wt.-%, more preferably in the range of from 0.002 wt.-% to 12 wt.-%, more preferably in the range of from 0.005 wt.-% to
11 wt.-%, more preferably in the range of from 0.01 wt.-% to 10 wt.-%, more preferably in the range of from 0.05 wt.-% to 9 wt.-%, more preferably in the range of from 0.1 wt.-% ppm to 8 wt.-%, more preferably in the range of from 0.5 wt.-% to 5 wt.-%, more preferably in the range of from 1 wt.-% to 5 wt.-%, more preferably in the range of from 1 wt.-% to 3 wt.-%, wherein the wt.-% in each case are the weight of all biosurfactants SBIO comprised by aqueous mixture L relative to the total weight of aqueous mixture L.
During washing of Aw in the aqueous mixture L in the context of the method according to the second aspect of the invention, Aw is contacted with the aqueous mixture L for at least 1 s, preferably at least 10 s, more preferably at least 1 min, even more preferably at least 5 min, preferably for 15 min to 240 min, even more preferably for 30 min to 180 min, even more preferably for 45 min to 120 min, even more preferably for 60 min to 120 min, even more preferably for 70 min to 100 min.
During washing of Aw in the aqueous mixture L in the context of the method according to the second aspect of the invention, the woolen article Aw is preferably moved in the aqueous mixture. This improves the washing efficiency.
The washing in the context of the method according to the second aspect of the invention is preferably carried out in a washing machine.
The at least one biosurfactant SBIO in the method according to the second aspect of the invention may be present at least partially as salts, in particular when it is comprised by the aqueous mixture L.
In those embodiments where the at least one biosurfactant SBIO is at least partially present as salts, the ratio of biosurfactants SBIO that are present as salts depends on the pH of the water in the aqueous mixture L.
In those embodiments where at least one biosurfactant SBIO is present as salts, it is preferred that at least a part of the biosurfactants SBIO are salts in which the cation is selected from the group consisting of Li+, Na+, K+, Mg2+, Ca2+, AP+, NH4+, ammonium ions, wherein the ammonium ions may be primary, secondary, tertiary or quaternary. Particularly preferred cations are selected from the group comprising, preferably consisting of, Na+, K+, NH4+ and the triethanolammonium cation.
Exemplary representatives of suitable ammonium ions are tetramethylammonium, tetraethylammonium, tetrapropylammonium, tetrabutylammonium,
[(2-hydroxyethyl)trimethylammonium] (choline) and also the cations of 2-aminoethanol (ethanolamine, “MEA”), diethanolamine (“DEA”), 2,2',2"-nitrilotriethanol (triethanolamine, “TEA”), 1-aminopropan-2-ol (monoisopropanolamine), ethylenediamine, diethylenetriamine, triethylenetetramine, tetraethylenepentamine, 1 ,4-diethylenediamine (piperazine), aminoethylpiperazine and aminoethylethanolamine.
The mixtures of the abovementioned cations may also be present as cations of the salts of the at least one biosurfactant SBIO, if present as salts, according to the invention.
In the context of the method according to the second aspect of the present invention, aqueous mixture L may comprise further ingredients, preferably at least one ingredient selected from surfactants (wherein the surfactants are different from SBIO), enzymes, builders, solvents, preservatives, benefit agents, polymers.
2.2.3 Surfactants
Hence, in an optional embodiment, aqueous mixture L in the context of the method according to the second aspect of the present invention, comprises, besides the at least one biosurfactant SBIO, at least one further surfactant SY, wherein the surfactant SY is different from SBIO. The at least one further surfactant SY may be selected from anionic surfactants, nonionic surfactants, cationic surfactants, amphoteric surfactants, and in particular is selected from anionic surfactants, nonionic surfactants.
In those cases where the aqueous mixture L in the context of the method according to the second aspect of the present invention comprises at least one further surfactant SY that is different from the biosurfactants SBIO, it is preferred the ratio of weights of all biosurfactants SBIO in the mixture L to the weight of all surfactants SY is in the range of from 99 : 1 to 1 : 99, preferably 90 : 10 to 10 : 90, more preferably 80 : 20 to 20 : 80, even more preferably 70 : 30 to 30 : 70, even more preferably 60 : 40 to 40 : 60, eve more preferably 50 : 50.
Preferred nonionic surfactants are those described under item 1 .4.1 above.
Preferred anionic surfactants are those described under item 1.4.2 above.
Preferred amphoteric surfactants are those described under item 1 .4.3 above.
Preferred cationic surfactants are those described under item 1.4.4 above.
2.2.4 Enzymes
The aqueous mixture L in the context of the method according to the second aspect of the invention may also comprise at least one enzyme E.
In those embodiments where the aqueous mixture L in the context of the method according to the second aspect of the invention comprises at least one enzyme E, it is further preferred that the enzyme E is selected from the group consisting of protease, amylase, cellulase, mannanase, lipase, cutinase, pectate lyase, peroxidase, oxidase, laccase, even more preferably selected from the group consisting of cellulase, protease, lipase, cutinase, amylase, peroxidase, oxidase, mannanase. It is even more preferred that the at least one enzyme E is selected from the group consisting of protease, amylase, lipase, mannanase. Most preferred, the at least one enzyme E is a lipase.
In case the aqueous mixture L in the context of the method according to the second aspect of the invention comprises at least one enzyme E, it is preferred that the amount of all enzymes E in the aqueous mixture L is in the range of 0.01 ppm to 1 wt.-%, preferably 0.01 ppm to 1000 ppm, more preferably 0.1 to 100 ppm, preferably 1 ppm to 10 ppm per the total weight of aqueous mixture L.
Preferred cellulases are those described under point 1 .4.2.1 above.
Preferred proteases are those described under point 1 .4.2.2 above.
Preferred lipases and cutinases are those described under point 1 .4.2.3 above.
Preferred amylases are those described under point 1 .4.2.4 above.
Preferred oxidases and peroxidases are those described under point 1 .4.2.5 above. Preferred mannanases are those described under point 1.4.2.6 above.
2.2.5 Builders
The aqueous mixture L in the context of the method according to the second aspect of the invention preferably also comprise at least one builder Bz.
In those embodiments where aqueous mixture L in the context of the method according to the second aspect of the invention comprises at least one builder Bz, the amount of all builders Bz in aqueous mixture L is preferably between 0.1 ppm to 1000 ppm, preferably 1 ppm to 100 ppm, more preferably 10 ppm to 50 ppm relative to the total weight of aqueous mixture L.
Preferred builders are those described under point 1 .4.3 above.
2.2.6 Solvents
In a preferred embodiment, aqueous mixture L in the context of the method according to the second aspect of the invention may comprise at least one non-aqueous solvent. In those embodiments where aqueous mixture L in the context of the method according to the second aspect of the invention comprises at least one non-aqueous solvent, the ratio between the weight of the water comprised by aqueous mixture L to the weight of all non-aqueous solvents comprised by aqueous mixture L is preferably in the range of from 999 : 1 to 1 : 999, preferably in the range of
from 99 : 1 to 1 : 99, more preferably in the range of from 9 : 1 to 1 : 9, more preferably in the range of from 8 : 2 to 2 : 8, more preferably in the range of from 7 : 3 to 3 : 7, more preferably in the range of from 6 : 4 to 4 : 6, more preferably at 1 : 1 .
Suitable non-aqueous solvents are those described under item 1 .4.4 above.
2.2.7 Preservatives
In a preferred embodiment, aqueous mixture L in the context of the method according to the second aspect of the invention may comprise at least one preservative.
In those embodiments where aqueous mixture L in the context of the method according to the second aspect of the invention comprises at least one preservative, the amount of all preservatives in aqueous mixture L is preferably between 0.1 ppm to 1000 ppm, preferably 1 ppm to 100 ppm, more preferably 10 ppm to 50 ppm, relative to the total weight of aqueous mixture L.
Preferred preservatives are described under item 1 .4.5 above.
2.2.8 Benefit agent
In a preferred embodiment, aqueous mixture L in the context of the method according to the second aspect of the invention may comprise at least one benefit agent, preferably at least one encapsulate comprising a benefit agent, preferably a sensorial benefit agent.
In those embodiments where aqueous mixture L in the context of the method according to the second aspect of the invention comprises at least one benefit agent, the total amount of benefit agent in aqueous mixture L is preferably between 0.1 ppm to 1000 ppm, preferably 1 ppm to 100 ppm, more preferably 10 ppm to 50 ppm relative to the total weight of aqueous mixture L.
Preferred benefit agents are described under point 1 .4.6 above.
2.2.9 Polymers
In a preferred embodiment, aqueous mixture L in the context of the method according to the second aspect of the invention may comprise at least one polymer for use in detergents. The polymer may function as a co-builder as mentioned above, or may provide antiredeposition, fiber protection, soil release, dye transfer inhibition, viscosity modifiers, grease cleaning and/or antifoaming properties. In those embodiments where aqueous mixture L in the context of the method according to the second aspect of the invention comprises at least one polymer, the amount of all polymers in aqueous mixture L is preferably between 0.1 ppm to 1000 ppm, preferably 1 ppm to 100 ppm, more preferably 10 ppm to 50 ppm relative to the total weight of aqueous mixture L.
Exemplary polymers are those described under point 1 .4.7 above.
In a preferred embodiment, aqueous mixture L in the context of the method according to the second aspect of the invention comprises at least one polymer selected from the group consisting of carboxymethylcellulose, polyethylenglycol, starch, carboxymethyl inulin, poly-aspartic acid, polyglutamic acid, sulfonated polycarboxylates, polyethylene oxide, polypropylene oxide, polyquaternium. These polymers are preferred as they are ecologically favorable. Preferred polyquaternium is polyquaternium 10, which is quaternized hydroxyethyl cellulose.
2.2.10 Other Additives
In a preferred embodiment, the aqueous mixture L in the context of the method according to the second aspect of the invention may comprise at least one further additive.
The further additive is preferably selected from the group set forth under item 1 .4.8 above.
The amounts of the respective additives are dependent on the intended use.
2.2.11 Composition ZG
The at least one biosurfactant SBIO may be provided in the context of the method according to the second aspect of the invention in pure form or as a composition ZG, which may be a cleaning formulation.
A preferred composition ZG is described under item 1 .5 above.
2.3 Step b.
In step b., the aqueous mixture L is at least partially separated from the woolen article Aw-
The separation is, in a preferred embodiment, carried out so that it is essentially complete.
The skilled person is aware of how to carry out this separation. In general, this step is carried out by withdrawing the article Aw from aqueous mixture L. During step b., the article Aw may be subjected to centrifugation.
2.4 Optional step c.
In an optional step c. of the method according to the second aspect of the invention, the article Aw is rinsed with water.
Examples
1 . Formulations preparation
To show the surprising effects of the biosurfactants-based laundry detergent, formulations 1 to 4, which are based on biosurfactants, were prepared and compared to benchmark formulations 1 to 4.
The term "mono-rhamnolipid" in the context of the present invention is understood to mean compounds of the general formula (III), where nRL = 0.
Mono-rhamnolipid (“mono RL”) was prepared as described in example 2 of EP 3 061 442 A1 .
The mono-rhamnolipid was employed as aqueous solution (75 wt.-%). The term "di-rhamnolipid" in the context of the present invention is understood to mean compounds of the general formula (III), where nRL = 1 .
Di-rhamnolipids (“diRL”) were prepared as described in example 1 of EP 3 061 442 A1.
The di-rhamnolipid was employed as aqueous solution (50 wt.-%).
2. Content of each formulation The composition of the benchmark formulations 1 to 4 (not according to the invention) were as follows:
The composition of the biosurfactant-based formulations 1 to 4 (according to the invention) were as follows:
3. Test for anti-shrinking effects
For the evaluation of the benchmark compositions 1 to 4 and the compositions according to the invention (formulations 1 to 4), laundry tests with each composition and one blank test (only water, no addition of surfactant) were carried out in the washing machine for five cycles. Each test was performed at 40 °C. The tests were carried out according to the protocol of Assay X.
In each test the prepared five wool textiles from WFK 60A (order code: 60000 100 % wool fabric, length 75 cm and width 40 cm) were added together with 2 kg of ballast load to a laundry machine.
The water used had a water hardness of 14 ° dGH, i.e. the concentration of earth alkali metal cations in the water was 2.5 mmol/l.
50 g of each composition (benchmark 1 to 4, formulation 1 to 4, blank test) was used in the washing process.
A programmable Miele electronic household washing machine (“WCI 360 WTL; Power Wash 2.0”) was used. Fuzzy logic type control was disabled.
The applied washing program is explained in the following table:
Before the test was started, the 2 kg of standardized cotton load (ballast) was washed at 90 °C with ECE2 detergent without bleach.
The evaluation of the shrinking degree of the respective wool textiles was done by measuring the lengths and the width of the textile before and after the washing cycles.
4. Results
The length and width of each wool textile swatch before and after the wash are listed in the following table:
The comparison of the shrinking of the sample woolen cloths depending on different surfactants (formulations 1 to 4 viz benchmark formulations 1 to 4) surprisingly show that detergents based on biosurfactants such as rhamnolipids have a much lower shrinking effect on the wool fabric compared to washing in water and the washing with other surfactants.
In particular, the comparison between formulations 1 to 4 and the wash with water shows that the formulations 1 to 4 reduce the shrinking considerably.
In addition, the shrinking the comparison between formulations 1 to 4 and benchmarks 1 to 4 shows that the formulations 1 to 4 reduce the shrinking considerably also compared to the benchmarks. Of note, benchmark 3 and 4 differ from the formulations 1 to 4 only in the surfactants: benchmarks 3 and 4 comprise SLES or a mixture of SLES and a further surfactant, while formulations 1 to 4 are identical, except that they comprise rhamnolipids instead of the non- inventive surfactants.
Claims
1 . Use of at least one biosurfactant SBIO to reduce shrinking of at least one woolen article Aw during washing of Aw, wherein, during washing of Aw, Aw is contacted with an aqueous mixture L comprising at least one biosurfactant SBIO, wherein the biosurfactant SBIO is selected from the group consisting of rhamnolipids, glucolipids, sophorolipids, wherein the biosurfactant SBIO, in particular, is selected from the group consisting of rhamnolipids, sophorolipids, and wherein the biosurfactant SBIO is preferably selected from rhamnolipids.
2. Use according to claim 1 , wherein the temperature of the aqueous mixture L, for at least as long as the aqueous mixture L is contacted with the at least one woolen article Aw, is in a range of from > 0 °C to 45 °C, in particular in a range of from 1 °C to 40 °C, preferably in a range of from 5 °C to 35 °C, more preferably in a range of from 10 °C to 30 °C, more preferably in a range of from 15 °C to 30 °C, even more preferably in a range of from 20 °C to 30 °C, even more preferably in a range of from 25 °C to 30 °C.
3. Use according to claim 1 or 2, wherein Aw is contacted with the aqueous mixture L for at least 1 s, preferably at least 10 s, more preferably at least 1 min, even more preferably at least 5 min, even more preferably for 15 min to 240 min, even more preferably for 30 min to 180 min, even more preferably for 45 min to 120 min, even more preferably for 60 min to 120 min, even more preferably for 70 min to 100 min.
4. Use according to one of claims 1 to 3, wherein the at least one woolen article Aw is moved in the aqueous mixture L during washing.
5. Use according to one of claims 1 to 4, wherein the reduction of shrinking is determined by Assay X as set forth in the description.
6. Use according to one of claims 1 to 5, wherein the aqueous mixture L comprises at least one ingredient selected from the group of enzymes, builders, non-aqueous solvents, preservatives, benefit agents, polymers, surfactants, wherein the surfactants are different from SBIO.
7. Use according to claim 6, wherein the aqueous mixture L comprises at least one polymer selected from the group consisting of carboxymethylcellulose, polyethylenglycol, starch,
carboxymethyl inulin, poly-aspartic acid, poly-glutamic acid, sulfonated polycarboxylates, polyethylene oxide, polypropylene oxide, polyquaternium.
8. Use according to one of claims 1 to 7, wherein the woolen article Aw is an article of clothing.
9. Method of washing at least one woolen article Aw, comprising the steps of a. contacting the at least one woolen article Aw with an aqueous mixture L comprising at least one biosurfactant SBIO, b. at least partially separating L from Aw, c. optionally rinsing Aw with water, wherein the temperature of the aqueous mixture L, for at least as long as the aqueous mixture L is contacted with the at least one woolen article Aw, is in a range of from
> 0 °C to 45 °C, in particular in a range of from 1 °C to 40 °C, preferably in a range of from 5 °C to 35 °C, more preferably in a range of from 10 °C to 30 °C, more preferably in a range of from 15 °C to 30 °C, even more preferably in a range of from 20 °C to 30 °C, even more preferably in a range of from 25 °C to 30 °C, wherein the biosurfactant SBIO is selected from the group consisting of rhamnolipids, glucolipids, sophorolipids, and wherein the biosurfactant SBIO, in particular, is selected from the group consisting of rhamnolipids, sophorolipids, and wherein the biosurfactant SBIO is preferably selected from rhamnolipids.
10. Method according to claim 9, wherein the contacting in step a. is carried out for at least 1 s, preferably at least 10 s, more preferably at least 1 min, even more preferably at least
5 min, even more preferably for 15 min to 240 min, even more preferably for 30 min to 180 min, even more preferably for 45 min to 120 min, even more preferably for 60 min to 120 min, even more preferably for 70 min to 100 min.
11. Method according to claim 9 or 10, wherein the aqueous mixture L comprises at least one ingredient selected from the group of enzymes, builders, non-aqueous solvents, preservatives, benefit agents, polymers, surfactants, wherein the surfactants are different from SBIO.
12. Use according to claim 11 , wherein the aqueous mixture L comprises at least one polymer selected from the group consisting of carboxymethylcellulose, polyethylenglycol, starch, carboxymethyl inulin, poly-aspartic acid, poly-glutamic acid, sulfonated polycarboxylares, polyethylene oxide, polypropylene oxide, polyquaternium.
13. Method according to one of claims 9 to 12, wherein the woolen article Aw is moved in the aqueous mixture L during step a.
14. Method according to one of claims 9 to 13, which is carried out in a washing machine.
15. Use according to one of claims 1 to 8 in a method according to one of claims 9 to 14.
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP23191487 | 2023-08-15 | ||
| EP23191487.0 | 2023-08-15 | ||
| EP23191489.6 | 2023-08-15 | ||
| EP23191489 | 2023-08-15 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2025036643A1 true WO2025036643A1 (en) | 2025-02-20 |
Family
ID=91966254
Family Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2024/070359 Pending WO2025036643A1 (en) | 2023-08-15 | 2024-07-18 | Biosurfactant for washing wool |
| PCT/EP2024/070356 Pending WO2025036642A1 (en) | 2023-08-15 | 2024-07-18 | Improved method for cleaning |
Family Applications After (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2024/070356 Pending WO2025036642A1 (en) | 2023-08-15 | 2024-07-18 | Improved method for cleaning |
Country Status (1)
| Country | Link |
|---|---|
| WO (2) | WO2025036643A1 (en) |
Citations (128)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3234258A (en) | 1963-06-20 | 1966-02-08 | Procter & Gamble | Sulfation of alpha olefins |
| GB1296839A (en) | 1969-05-29 | 1972-11-22 | ||
| GB1483591A (en) | 1973-07-23 | 1977-08-24 | Novo Industri As | Process for coating water soluble or water dispersible particles by means of the fluid bed technique |
| GB1515792A (en) | 1975-07-23 | 1978-06-28 | Hoechst Ag | Detergent composition for wool |
| US4106991A (en) | 1976-07-07 | 1978-08-15 | Novo Industri A/S | Enzyme granulate composition and process for forming enzyme granulates |
| US4305961A (en) | 1978-09-28 | 1981-12-15 | Kao Soap Co., Ltd. | Cosmetic composition |
| JPS58217598A (en) | 1982-06-10 | 1983-12-17 | 日本油脂株式会社 | Detergent composition |
| US4435307A (en) | 1980-04-30 | 1984-03-06 | Novo Industri A/S | Detergent cellulase |
| EP0218272A1 (en) | 1985-08-09 | 1987-04-15 | Gist-Brocades N.V. | Novel lipolytic enzymes and their use in detergent compositions |
| US4661452A (en) | 1984-05-29 | 1987-04-28 | Novo Industri A/S | Enzyme containing granulates useful as detergent additives |
| EP0238216A1 (en) | 1986-02-20 | 1987-09-23 | Albright & Wilson Limited | Protected enzyme systems |
| EP0258068A2 (en) | 1986-08-29 | 1988-03-02 | Novo Nordisk A/S | Enzymatic detergent additive |
| EP0282942A2 (en) | 1987-03-17 | 1988-09-21 | University Of Iowa Research Foundation | Method for producing rhamnose |
| EP0305216A1 (en) | 1987-08-28 | 1989-03-01 | Novo Nordisk A/S | Recombinant Humicola lipase and process for the production of recombinant humicola lipases |
| WO1989006279A1 (en) | 1988-01-07 | 1989-07-13 | Novo-Nordisk A/S | Mutated subtilisin genes |
| WO1989006270A1 (en) | 1988-01-07 | 1989-07-13 | Novo-Nordisk A/S | Enzymatic detergent |
| EP0331376A2 (en) | 1988-02-28 | 1989-09-06 | Amano Pharmaceutical Co., Ltd. | Recombinant DNA, bacterium of the genus pseudomonas containing it, and process for preparing lipase by using it |
| WO1989009259A1 (en) | 1988-03-24 | 1989-10-05 | Novo-Nordisk A/S | A cellulase preparation |
| JPH01304034A (en) | 1988-05-30 | 1989-12-07 | Agency Of Ind Science & Technol | Metal soap |
| WO1990013533A1 (en) | 1989-04-28 | 1990-11-15 | Henkel Kommanditgesellschaft Auf Aktien | The use of calcined hydrotalcites as catalysts for ethoxylating or propoxylating fatty acid esters |
| EP0407225A1 (en) | 1989-07-07 | 1991-01-09 | Unilever Plc | Enzymes and enzymatic detergent compositions |
| EP0439019A1 (en) | 1990-01-10 | 1991-07-31 | Kao Corporation | liquid detergent composition for clothes |
| WO1991017243A1 (en) | 1990-05-09 | 1991-11-14 | Novo Nordisk A/S | A cellulase preparation comprising an endoglucanase enzyme |
| WO1991017244A1 (en) | 1990-05-09 | 1991-11-14 | Novo Nordisk A/S | An enzyme capable of degrading cellulose or hemicellulose |
| US5075041A (en) | 1990-06-28 | 1991-12-24 | Shell Oil Company | Process for the preparation of secondary alcohol sulfate-containing surfactant compositions |
| WO1992005249A1 (en) | 1990-09-13 | 1992-04-02 | Novo Nordisk A/S | Lipase variants |
| EP0495257A1 (en) | 1991-01-16 | 1992-07-22 | The Procter & Gamble Company | Compact detergent compositions with high activity cellulase |
| EP0499434A1 (en) | 1991-02-12 | 1992-08-19 | Unilever Plc | Detergent compositions |
| WO1992019729A1 (en) | 1991-05-01 | 1992-11-12 | Novo Nordisk A/S | Stabilized enzymes and detergent compositions |
| WO1992021760A1 (en) | 1991-05-29 | 1992-12-10 | Cognis, Inc. | Mutant proteolytic enzymes from bacillus |
| DE4127908A1 (en) | 1991-08-23 | 1993-02-25 | Hoechst Ag | Inducing rhamno:lipid synthesis in Pseudomonas aeruginosa - with glyceric acid ether lipid, to form intermediate for rhamnose which is used in synthesis of chiral cpds. |
| WO1993018140A1 (en) | 1992-03-04 | 1993-09-16 | Novo Nordisk A/S | Novel proteases |
| WO1993024618A1 (en) | 1992-06-01 | 1993-12-09 | Novo Nordisk A/S | Peroxidase variants with improved hydrogen peroxide stability |
| WO1994001541A1 (en) | 1992-07-06 | 1994-01-20 | Novo Nordisk A/S | C. antarctica lipase and lipase variants |
| WO1994002597A1 (en) | 1992-07-23 | 1994-02-03 | Novo Nordisk A/S | MUTANT α-AMYLASE, DETERGENT, DISH WASHING AGENT, AND LIQUEFACTION AGENT |
| WO1994007998A1 (en) | 1992-10-06 | 1994-04-14 | Novo Nordisk A/S | Cellulase variants |
| WO1994018314A1 (en) | 1993-02-11 | 1994-08-18 | Genencor International, Inc. | Oxidatively stable alpha-amylase |
| US5352604A (en) | 1989-08-25 | 1994-10-04 | Henkel Research Corporation | Alkaline proteolytic enzyme and method of production |
| WO1994025583A1 (en) | 1993-05-05 | 1994-11-10 | Novo Nordisk A/S | A recombinant trypsin-like protease |
| WO1994025578A1 (en) | 1993-04-27 | 1994-11-10 | Gist-Brocades N.V. | New lipase variants for use in detergent applications |
| US5389536A (en) | 1986-11-19 | 1995-02-14 | Genencor, Inc. | Lipase from Pseudomonas mendocina having cutinase activity |
| WO1995010602A1 (en) | 1993-10-13 | 1995-04-20 | Novo Nordisk A/S | H2o2-stable peroxidase variants |
| WO1995010603A1 (en) | 1993-10-08 | 1995-04-20 | Novo Nordisk A/S | Amylase variants |
| WO1995014783A1 (en) | 1993-11-24 | 1995-06-01 | Showa Denko K.K. | Lipase gene and variant lipase |
| WO1995023221A1 (en) | 1994-02-24 | 1995-08-31 | Cognis, Inc. | Improved enzymes and detergents containing them |
| WO1995024471A1 (en) | 1994-03-08 | 1995-09-14 | Novo Nordisk A/S | Novel alkaline cellulases |
| WO1995030744A2 (en) | 1994-05-04 | 1995-11-16 | Genencor International Inc. | Lipases with improved surfactant resistance |
| WO1995035381A1 (en) | 1994-06-20 | 1995-12-28 | Unilever N.V. | Modified pseudomonas lipases and their use |
| WO1996011262A1 (en) | 1994-10-06 | 1996-04-18 | Novo Nordisk A/S | An enzyme and enzyme preparation with endoglucanase activity |
| WO1996012012A1 (en) | 1994-10-14 | 1996-04-25 | Solvay S.A. | Lipase, microorganism producing same, method for preparing said lipase and uses thereof |
| WO1996013580A1 (en) | 1994-10-26 | 1996-05-09 | Novo Nordisk A/S | An enzyme with lipolytic activity |
| WO1996023873A1 (en) | 1995-02-03 | 1996-08-08 | Novo Nordisk A/S | Amylase variants |
| WO1996029397A1 (en) | 1995-03-17 | 1996-09-26 | Novo Nordisk A/S | Novel endoglucanases |
| WO1996034946A1 (en) | 1995-05-05 | 1996-11-07 | Novo Nordisk A/S | Protease variants and compositions |
| US5648263A (en) | 1988-03-24 | 1997-07-15 | Novo Nordisk A/S | Methods for reducing the harshness of a cotton-containing fabric |
| DE19600743A1 (en) | 1996-01-11 | 1997-07-24 | Henkel Kgaa | Use of mixture of glyco-lipid and surfactant in hand dish-washing detergent |
| WO1997043424A1 (en) | 1996-05-14 | 1997-11-20 | Genencor International, Inc. | MODIFIED α-AMYLASES HAVING ALTERED CALCIUM BINDING PROPERTIES |
| WO1998008940A1 (en) | 1996-08-26 | 1998-03-05 | Novo Nordisk A/S | A novel endoglucanase |
| WO1998012307A1 (en) | 1996-09-17 | 1998-03-26 | Novo Nordisk A/S | Cellulase variants |
| WO1998015257A1 (en) | 1996-10-08 | 1998-04-16 | Novo Nordisk A/S | Diaminobenzoic acid derivatives as dye precursors |
| WO1998020115A1 (en) | 1996-11-04 | 1998-05-14 | Novo Nordisk A/S | Subtilase variants and compositions |
| WO1998020116A1 (en) | 1996-11-04 | 1998-05-14 | Novo Nordisk A/S | Subtilase variants and compositions |
| DE19648439A1 (en) | 1996-11-22 | 1998-05-28 | Henkel Kgaa | Skin-friendly washing=up liquid with good cleaning performance |
| US5827718A (en) | 1993-08-30 | 1998-10-27 | Novo Nordisk A/S | Lipase, microorganisms producing the lipase, method of producing the lipase and use of the lipase |
| WO1999001544A1 (en) | 1997-07-04 | 1999-01-14 | Novo Nordisk A/S | FAMILY 6 ENDO-1,4-β-GLUCANASE VARIANTS AND CLEANING COMPOSIT IONS CONTAINING THEM |
| WO1999011768A1 (en) | 1997-08-29 | 1999-03-11 | Novo Nordisk A/S | Protease variants and compositions |
| WO1999019467A1 (en) | 1997-10-13 | 1999-04-22 | Novo Nordisk A/S | α-AMYLASE MUTANTS |
| US5942431A (en) | 1995-02-27 | 1999-08-24 | Novo Nordisk A/S | DNA sequences encoding lipases and method for producing lipases |
| WO2000060063A1 (en) | 1999-03-31 | 2000-10-12 | Novozymes A/S | Lipase variant |
| WO2001016285A2 (en) | 1999-08-31 | 2001-03-08 | Novozymes A/S | Novel proteases and variants thereof |
| WO2001044452A1 (en) | 1999-12-15 | 2001-06-21 | Novozymes A/S | Subtilase variants having an improved wash performance on egg stains |
| WO2001066712A2 (en) | 2000-03-08 | 2001-09-13 | Novozymes A/S | Variants with altered properties |
| WO2001092502A1 (en) | 2000-06-02 | 2001-12-06 | Novozymes A/S | Cutinase variants |
| WO2002010355A2 (en) | 2000-08-01 | 2002-02-07 | Novozymes A/S | Alpha-amylase mutants with altered stability |
| CN1337439A (en) | 2000-08-09 | 2002-02-27 | 大庆油田有限责任公司勘探开发研究院 | Oil-displacing agent and its application |
| WO2002016547A2 (en) | 2000-08-21 | 2002-02-28 | Novozymes A/S | Subtilase enzymes |
| US6472364B1 (en) | 1998-10-13 | 2002-10-29 | The Procter & Gamble Company | Detergent compositions or components |
| WO2002099091A2 (en) | 2001-06-06 | 2002-12-12 | Novozymes A/S | Endo-beta-1,4-glucanase from bacillus |
| WO2003002700A1 (en) | 2001-06-27 | 2003-01-09 | Saraya Co., Ltd. | Low-foaming detergent compositions |
| WO2003006146A1 (en) | 2001-07-13 | 2003-01-23 | Cognis France S.A. | Aqueous preparations |
| WO2003006602A2 (en) | 2001-07-12 | 2003-01-23 | Novozymes A/S | Subtilase variants |
| EP1382668A1 (en) | 2002-06-11 | 2004-01-21 | Unilever N.V. | Detergent tablets |
| WO2004074419A2 (en) | 2003-02-18 | 2004-09-02 | Novozymes A/S | Detergent compositions |
| WO2005040372A1 (en) | 2003-10-23 | 2005-05-06 | Novozymes A/S | Protease with improved stability in detergents |
| WO2005052146A2 (en) | 2003-11-19 | 2005-06-09 | Genencor International, Inc. | Serine proteases, nucleic acids encoding serine enzymes and vectors and host cells incorporating same |
| WO2005056782A2 (en) | 2003-12-03 | 2005-06-23 | Genencor International, Inc. | Perhydrolase |
| EP1705241A1 (en) | 2005-03-23 | 2006-09-27 | Unilever N.V. | Detergent compositions in tablet form |
| WO2006130575A2 (en) | 2005-05-31 | 2006-12-07 | The Procter & Gamble Company | Polymer-containing detergent compositions and their use |
| WO2007001262A1 (en) | 2005-06-17 | 2007-01-04 | The Procter & Gamble Company | Organic catalyst with enhanced enzyme compatibility |
| EP1746109A2 (en) | 2005-07-21 | 2007-01-24 | National Starch and Chemical Investment Holding Corporation | Hybrid copolymers |
| WO2007044993A2 (en) | 2005-10-12 | 2007-04-19 | Genencor International, Inc. | Use and production of storage-stable neutral metalloprotease |
| WO2007087508A2 (en) | 2006-01-23 | 2007-08-02 | Novozymes A/S | Lipase variants |
| US7262042B2 (en) | 2001-12-20 | 2007-08-28 | Henkel Kommanditgesellschaft Auf Aktien (Henkel Kgaa) | Alkaline protease from Bacillus gibsonii (DSM 14393) and washing and cleaning products comprising said alkaline protease |
| US20080213194A1 (en) | 2006-07-27 | 2008-09-04 | Desanto Keith | Rhamnolipid-based formulations |
| WO2008153815A2 (en) | 2007-05-30 | 2008-12-18 | Danisco Us, Inc., Genencor Division | Variants of an alpha-amylase with improved production levels in fermentation processes |
| US20090011970A1 (en) | 2007-07-02 | 2009-01-08 | Marc Francois Theophile Evers | Laundry multi-compartment pouch composition |
| WO2009021867A2 (en) | 2007-08-10 | 2009-02-19 | Henkel Ag & Co. Kgaa | Agents containing proteases |
| WO2009061380A2 (en) | 2007-11-05 | 2009-05-14 | Danisco Us Inc., Genencor Division | VARIANTS OF BACILLUS sp. TS-23 ALPHA-AMYLASE WITH ALTERED PROPERTIES |
| WO2009067279A1 (en) | 2007-11-21 | 2009-05-28 | E.I. Du Pont De Nemours And Company | Production of peracids using an enzyme having perhydrolysis activity |
| US7556654B1 (en) | 2004-10-15 | 2009-07-07 | Naturell | Methods for cleaning materials |
| WO2009092699A1 (en) | 2008-01-24 | 2009-07-30 | Unilever Nv | Machine dishwash detergent compositions |
| WO2009102854A1 (en) | 2008-02-15 | 2009-08-20 | The Procter & Gamble Company | Cleaning compositions |
| WO2010065455A2 (en) | 2008-12-01 | 2010-06-10 | Danisco Us Inc. | Enzymes with lipase activity |
| WO2010100028A2 (en) | 2009-03-06 | 2010-09-10 | Huntsman Advanced Materials (Switzerland) Gmbh | Enzymatic textile bleach-whitening methods |
| WO2010107560A2 (en) | 2009-03-18 | 2010-09-23 | Danisco Us Inc. | Fungal cutinase from magnaporthe grisea |
| WO2010111143A2 (en) | 2009-03-23 | 2010-09-30 | Danisco Us Inc. | Cal a-related acyltransferases and methods of use, thereof |
| US20110091399A1 (en) | 2008-05-15 | 2011-04-21 | Evonik Goldschmidt Gmbh | Use of organo-modified siloxane block copolymers for producing cosmetic or pharmaceutical compositions |
| WO2011084412A1 (en) | 2009-12-21 | 2011-07-14 | Danisco Us Inc. | Detergent compositions containing thermobifida fusca lipase and methods of use thereof |
| WO2011084417A1 (en) | 2009-12-21 | 2011-07-14 | Danisco Us Inc. | Detergent compositions containing geobacillus stearothermophilus lipase and methods of use thereof |
| WO2011084599A1 (en) | 2009-12-21 | 2011-07-14 | Danisco Us Inc. | Detergent compositions containing bacillus subtilis lipase and methods of use thereof |
| WO2011098531A1 (en) | 2010-02-10 | 2011-08-18 | Novozymes A/S | Variants and compositions comprising variants with high stability in presence of a chelating agent |
| WO2011150157A2 (en) | 2010-05-28 | 2011-12-01 | Danisco Us Inc. | Detergent compositions containing streptomyces griseus lipase and methods of use thereof |
| CN102660392A (en) | 2012-05-29 | 2012-09-12 | 西北民族大学 | Detergent for wool |
| WO2012137147A1 (en) | 2011-04-08 | 2012-10-11 | Danisco Us, Inc. | Compositions |
| WO2013001087A2 (en) | 2011-06-30 | 2013-01-03 | Novozymes A/S | Method for screening alpha-amylases |
| WO2013001078A1 (en) | 2011-06-30 | 2013-01-03 | Novozymes A/S | Alpha-amylase variants |
| CN104031899A (en) | 2013-03-05 | 2014-09-10 | 中国科学院天津工业生物技术研究所 | Lipase and application thereof |
| EP2787065A1 (en) | 2013-04-02 | 2014-10-08 | Evonik Industries AG | Detergent composition for textiles comprising rhamnolipids having a predominant share of di-rhamnolipids |
| EP2786743A1 (en) | 2013-04-02 | 2014-10-08 | Evonik Industries AG | Mixture composition containing rhamnolipids |
| WO2016075179A1 (en) | 2014-11-11 | 2016-05-19 | Clariant International Ltd | Laundry detergents containing soil release polymers |
| WO2016075178A1 (en) | 2014-11-11 | 2016-05-19 | Clariant International Ltd | Laundry detergents containing soil release polymers |
| EP3061442A1 (en) | 2015-02-27 | 2016-08-31 | Evonik Degussa GmbH | Composition comprising rhamnolipid and siloxane |
| CN106854531A (en) | 2017-01-04 | 2017-06-16 | 长沙协浩吉生物工程有限公司 | A kind of compound method of woollen and cashmere anti-shrinkage machine washing liquid |
| EP3489338A1 (en) | 2017-11-28 | 2019-05-29 | Clariant International Ltd | Detergent compositions containing renewably sourced soil release polyesters |
| EP3489340A1 (en) | 2017-11-28 | 2019-05-29 | Clariant International Ltd | Renewably sourced soil release polyesters |
| WO2019105675A1 (en) | 2017-11-30 | 2019-06-06 | Unilever Plc | Detergent composition comprising protease |
| US20210171864A1 (en) | 2019-09-29 | 2021-06-10 | Tianjin Zhongtian Jingke Technology Co., Ltd. | Soaping Agent And Preparation Method And Application Thereof |
| WO2022162043A1 (en) | 2021-01-28 | 2022-08-04 | Novozymes A/S | Lipase with low malodor generation |
Family Cites Families (42)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| FR2740779B1 (en) | 1995-11-08 | 1997-12-05 | Rhone Poulenc Chimie | COMPOSITION BASED ON ENZYME AND SOPHOROLIPIDE IN LACTONE FORM AND USE THEREOF IN DETERGENT FORMULATIONS FOR THE WASHING OF LAUNDRY |
| PT1445302E (en) | 2003-01-28 | 2006-10-31 | Ecover N V | DETERGENT COMPOSITIONS |
| FR2855752B1 (en) | 2003-06-03 | 2005-08-26 | Lvmh Rech | COSMETIC USE OF SOPHOROLIPIDS AS REGULATORY AGENTS OF SUB-CUTANEOUS ADIPOSE MASS AND APPLICATION TO SLURRY |
| US7994138B2 (en) | 2004-06-01 | 2011-08-09 | Agscitech Inc. | Microbial biosurfactants as agents for controlling pests |
| JP4548827B2 (en) | 2004-09-06 | 2010-09-22 | サラヤ株式会社 | Biodegradable liquid detergent composition |
| JP2006083238A (en) | 2004-09-14 | 2006-03-30 | Saraya Kk | Cleanser composition |
| JP2006274233A (en) | 2005-03-29 | 2006-10-12 | Saraya Kk | Bleaching agent composition |
| JP4858946B2 (en) | 2006-01-10 | 2012-01-18 | 独立行政法人産業技術総合研究所 | Emulsifier or solubilizer |
| JP5294226B2 (en) | 2006-09-07 | 2013-09-18 | 独立行政法人産業技術総合研究所 | W / O type microemulsion |
| DE102009045077A1 (en) | 2009-09-29 | 2011-03-31 | Evonik Goldschmidt Gmbh | Use of sophorolipids and their derivatives in combination with pesticides as adjuvant / additive for crop protection and industrial non-crop sector |
| DE102010014680A1 (en) | 2009-11-18 | 2011-08-18 | Evonik Degussa GmbH, 45128 | Cells, nucleic acids, enzymes and their use, as well as methods for producing sophorolipids |
| DE102010032484A1 (en) | 2010-07-28 | 2012-02-02 | Evonik Goldschmidt Gmbh | Cells and methods for producing rhamnolipids |
| DE102011090030B4 (en) | 2011-12-28 | 2025-11-06 | Evonik Operations Gmbh | Aqueous hair and skin cleansing compositions containing biosurfactants |
| DE102012201360A1 (en) | 2012-01-31 | 2013-08-01 | Evonik Industries Ag | New cell capable of forming rhamnolipid compound, which is useful for the production of cosmetic, dermatological or pharmaceutical formulations, of plant protection formulations and of care and cleaning agents and surfactant concentrates |
| DE102012221519A1 (en) | 2012-11-26 | 2014-05-28 | Evonik Industries Ag | Process for the isolation of rhamnolipids |
| EP2786742A1 (en) | 2013-04-02 | 2014-10-08 | Evonik Industries AG | Cosmetics containing rhamnolipids |
| DE102013206314A1 (en) | 2013-04-10 | 2014-10-16 | Evonik Industries Ag | Cosmetic formulation containing copolymer as well as sulfosuccinate and / or biosurfactant |
| DE102014209346A1 (en) | 2014-05-16 | 2015-11-19 | Evonik Degussa Gmbh | Yield increase by sophorolipids |
| EP2949214A1 (en) | 2014-05-26 | 2015-12-02 | Evonik Degussa GmbH | Methods of producing rhamnolipids |
| EP3002328A1 (en) | 2014-09-30 | 2016-04-06 | Evonik Degussa GmbH | Formulation containing biotensides |
| PL3023431T3 (en) | 2014-11-19 | 2017-07-31 | Evonik Degussa Gmbh | Concentrated, low viscosity rhamnolipid compounds |
| US20180066297A1 (en) | 2015-02-19 | 2018-03-08 | Evonik Degussa Gmbh | Rhamnolipid synthesis |
| EP3070155A1 (en) | 2015-03-18 | 2016-09-21 | Evonik Degussa GmbH | Composition comprising peptidase and biosurfactant |
| US10941173B2 (en) | 2016-02-22 | 2021-03-09 | Evonik Operations Gmbh | Rhamnolipid amides for hair scent retention |
| EP3419985B1 (en) | 2016-02-22 | 2025-04-09 | Evonik Operations GmbH | Rhamnolipid esters as non-ionic surfactants for cosmetic use |
| JP2019508564A (en) | 2016-03-18 | 2019-03-28 | エボニック デグサ ゲーエムベーハーEvonik Degussa GmbH | Granules comprising an inorganic solid support on which at least one biosurfactant is contained |
| WO2018065314A1 (en) | 2016-10-07 | 2018-04-12 | Evonik Degussa Gmbh | Composition containing glycolipids and preservatives |
| BR112019008320A2 (en) | 2016-10-24 | 2020-01-28 | Evonik Degussa Gmbh | cells and method to produce ramnolipids using alternative glucose transporters |
| WO2018077700A1 (en) | 2016-10-24 | 2018-05-03 | Evonik Degussa Gmbh | Rhamnolipid-producing cell having reduced glucose dehydrogenase activity |
| US11464717B2 (en) | 2017-02-10 | 2022-10-11 | Evonik Operations Gmbh | Oral care composition containing at least one biosurfactant and fluoride |
| EP3615646B1 (en) | 2017-04-27 | 2024-01-24 | Evonik Operations GmbH | Biodegradable cleaning composition |
| US11696885B2 (en) | 2017-08-24 | 2023-07-11 | Evonik Operations Gmbh | Rhamnolipid derivatives as emulsifiers and dispersing aids |
| US11236372B2 (en) | 2018-02-09 | 2022-02-01 | Evonik Operations Gmbh | Lipid production |
| JP7502321B2 (en) | 2019-03-07 | 2024-06-18 | エボニック オペレーションズ ゲーエムベーハー | Rhamnolipids as adhesion promoters. |
| US11918670B2 (en) | 2020-03-11 | 2024-03-05 | Evonik Operations Gmbh | Mixture composition comprising glycolipids and triethyl citrate |
| JP2023520661A (en) | 2020-03-24 | 2023-05-18 | エボニック オペレーションズ ゲーエムベーハー | Composition containing rhamnolipid, alkyl polyglycoside and acyl lactylate |
| US12497426B2 (en) | 2020-07-22 | 2025-12-16 | Evonik Operations Gmbh | Rhamnolipid oligo-esters |
| EP4015588A1 (en) | 2020-12-15 | 2022-06-22 | Evonik Operations GmbH | Use of rhamnolipids and / or sophorolipids in coating compositions for maintaining a homogeneous colour image of the dry coating film and / or for increasing work safety in the production of coating compositions |
| EP4015583A1 (en) | 2020-12-15 | 2022-06-22 | Evonik Operations GmbH | Use of rhamnolipids and / or sophorolipids for increasing the yield and / or for maintaining the application properties upon storage of coating compositions |
| US20250073149A1 (en) | 2021-05-05 | 2025-03-06 | Evonik Operations Gmbh | Compositions containing at least one biosurfactant and at least one sulfonic or sulfinic acid derivative |
| WO2023099346A1 (en) | 2021-12-02 | 2023-06-08 | Evonik Operations Gmbh | Composition comprising glucolipids |
| EP4234671A1 (en) | 2022-02-24 | 2023-08-30 | Evonik Operations GmbH | Compositions containing biosurfactants and a lipase from stachybotrys chlorohalonata |
-
2024
- 2024-07-18 WO PCT/EP2024/070359 patent/WO2025036643A1/en active Pending
- 2024-07-18 WO PCT/EP2024/070356 patent/WO2025036642A1/en active Pending
Patent Citations (138)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3234258A (en) | 1963-06-20 | 1966-02-08 | Procter & Gamble | Sulfation of alpha olefins |
| GB1296839A (en) | 1969-05-29 | 1972-11-22 | ||
| GB1483591A (en) | 1973-07-23 | 1977-08-24 | Novo Industri As | Process for coating water soluble or water dispersible particles by means of the fluid bed technique |
| GB1515792A (en) | 1975-07-23 | 1978-06-28 | Hoechst Ag | Detergent composition for wool |
| US4106991A (en) | 1976-07-07 | 1978-08-15 | Novo Industri A/S | Enzyme granulate composition and process for forming enzyme granulates |
| US4305961A (en) | 1978-09-28 | 1981-12-15 | Kao Soap Co., Ltd. | Cosmetic composition |
| US4435307A (en) | 1980-04-30 | 1984-03-06 | Novo Industri A/S | Detergent cellulase |
| JPS58217598A (en) | 1982-06-10 | 1983-12-17 | 日本油脂株式会社 | Detergent composition |
| US4661452A (en) | 1984-05-29 | 1987-04-28 | Novo Industri A/S | Enzyme containing granulates useful as detergent additives |
| EP0218272A1 (en) | 1985-08-09 | 1987-04-15 | Gist-Brocades N.V. | Novel lipolytic enzymes and their use in detergent compositions |
| EP0238216A1 (en) | 1986-02-20 | 1987-09-23 | Albright & Wilson Limited | Protected enzyme systems |
| EP0258068A2 (en) | 1986-08-29 | 1988-03-02 | Novo Nordisk A/S | Enzymatic detergent additive |
| US5389536A (en) | 1986-11-19 | 1995-02-14 | Genencor, Inc. | Lipase from Pseudomonas mendocina having cutinase activity |
| EP0282942A2 (en) | 1987-03-17 | 1988-09-21 | University Of Iowa Research Foundation | Method for producing rhamnose |
| EP0305216A1 (en) | 1987-08-28 | 1989-03-01 | Novo Nordisk A/S | Recombinant Humicola lipase and process for the production of recombinant humicola lipases |
| WO1989006270A1 (en) | 1988-01-07 | 1989-07-13 | Novo-Nordisk A/S | Enzymatic detergent |
| WO1989006279A1 (en) | 1988-01-07 | 1989-07-13 | Novo-Nordisk A/S | Mutated subtilisin genes |
| EP0331376A2 (en) | 1988-02-28 | 1989-09-06 | Amano Pharmaceutical Co., Ltd. | Recombinant DNA, bacterium of the genus pseudomonas containing it, and process for preparing lipase by using it |
| US5691178A (en) | 1988-03-22 | 1997-11-25 | Novo Nordisk A/S | Fungal cellulase composition containing alkaline CMC-endoglucanase and essentially no cellobiohydrolase |
| WO1989009259A1 (en) | 1988-03-24 | 1989-10-05 | Novo-Nordisk A/S | A cellulase preparation |
| US5648263A (en) | 1988-03-24 | 1997-07-15 | Novo Nordisk A/S | Methods for reducing the harshness of a cotton-containing fabric |
| US5776757A (en) | 1988-03-24 | 1998-07-07 | Novo Nordisk A/S | Fungal cellulase composition containing alkaline CMC-endoglucanase and essentially no cellobiohydrolase and method of making thereof |
| JPH01304034A (en) | 1988-05-30 | 1989-12-07 | Agency Of Ind Science & Technol | Metal soap |
| WO1990013533A1 (en) | 1989-04-28 | 1990-11-15 | Henkel Kommanditgesellschaft Auf Aktien | The use of calcined hydrotalcites as catalysts for ethoxylating or propoxylating fatty acid esters |
| EP0407225A1 (en) | 1989-07-07 | 1991-01-09 | Unilever Plc | Enzymes and enzymatic detergent compositions |
| US5352604A (en) | 1989-08-25 | 1994-10-04 | Henkel Research Corporation | Alkaline proteolytic enzyme and method of production |
| EP0439019A1 (en) | 1990-01-10 | 1991-07-31 | Kao Corporation | liquid detergent composition for clothes |
| WO1991017243A1 (en) | 1990-05-09 | 1991-11-14 | Novo Nordisk A/S | A cellulase preparation comprising an endoglucanase enzyme |
| US5457046A (en) | 1990-05-09 | 1995-10-10 | Novo Nordisk A/S | Enzyme capable of degrading cellullose or hemicellulose |
| US5686593A (en) | 1990-05-09 | 1997-11-11 | Novo Nordisk A/S | Enzyme capable of degrading cellulose or hemicellulose |
| WO1991017244A1 (en) | 1990-05-09 | 1991-11-14 | Novo Nordisk A/S | An enzyme capable of degrading cellulose or hemicellulose |
| US5763254A (en) | 1990-05-09 | 1998-06-09 | Novo Nordisk A/S | Enzyme capable of degrading cellulose or hemicellulose |
| US5075041A (en) | 1990-06-28 | 1991-12-24 | Shell Oil Company | Process for the preparation of secondary alcohol sulfate-containing surfactant compositions |
| WO1992005249A1 (en) | 1990-09-13 | 1992-04-02 | Novo Nordisk A/S | Lipase variants |
| EP0495257A1 (en) | 1991-01-16 | 1992-07-22 | The Procter & Gamble Company | Compact detergent compositions with high activity cellulase |
| EP0499434A1 (en) | 1991-02-12 | 1992-08-19 | Unilever Plc | Detergent compositions |
| WO1992019729A1 (en) | 1991-05-01 | 1992-11-12 | Novo Nordisk A/S | Stabilized enzymes and detergent compositions |
| WO1992021760A1 (en) | 1991-05-29 | 1992-12-10 | Cognis, Inc. | Mutant proteolytic enzymes from bacillus |
| DE4127908A1 (en) | 1991-08-23 | 1993-02-25 | Hoechst Ag | Inducing rhamno:lipid synthesis in Pseudomonas aeruginosa - with glyceric acid ether lipid, to form intermediate for rhamnose which is used in synthesis of chiral cpds. |
| WO1993018140A1 (en) | 1992-03-04 | 1993-09-16 | Novo Nordisk A/S | Novel proteases |
| WO1993024618A1 (en) | 1992-06-01 | 1993-12-09 | Novo Nordisk A/S | Peroxidase variants with improved hydrogen peroxide stability |
| WO1994001541A1 (en) | 1992-07-06 | 1994-01-20 | Novo Nordisk A/S | C. antarctica lipase and lipase variants |
| WO1994002597A1 (en) | 1992-07-23 | 1994-02-03 | Novo Nordisk A/S | MUTANT α-AMYLASE, DETERGENT, DISH WASHING AGENT, AND LIQUEFACTION AGENT |
| WO1994007998A1 (en) | 1992-10-06 | 1994-04-14 | Novo Nordisk A/S | Cellulase variants |
| WO1994018314A1 (en) | 1993-02-11 | 1994-08-18 | Genencor International, Inc. | Oxidatively stable alpha-amylase |
| WO1994025578A1 (en) | 1993-04-27 | 1994-11-10 | Gist-Brocades N.V. | New lipase variants for use in detergent applications |
| WO1994025583A1 (en) | 1993-05-05 | 1994-11-10 | Novo Nordisk A/S | A recombinant trypsin-like protease |
| US5827718A (en) | 1993-08-30 | 1998-10-27 | Novo Nordisk A/S | Lipase, microorganisms producing the lipase, method of producing the lipase and use of the lipase |
| WO1995010603A1 (en) | 1993-10-08 | 1995-04-20 | Novo Nordisk A/S | Amylase variants |
| WO1995010602A1 (en) | 1993-10-13 | 1995-04-20 | Novo Nordisk A/S | H2o2-stable peroxidase variants |
| WO1995014783A1 (en) | 1993-11-24 | 1995-06-01 | Showa Denko K.K. | Lipase gene and variant lipase |
| EP1921147A2 (en) | 1994-02-24 | 2008-05-14 | Henkel Kommanditgesellschaft auf Aktien | Improved enzymes and detergents containing them |
| EP1921148A2 (en) | 1994-02-24 | 2008-05-14 | Henkel Kommanditgesellschaft auf Aktien | Improved enzymes and detergents containing them |
| WO1995023221A1 (en) | 1994-02-24 | 1995-08-31 | Cognis, Inc. | Improved enzymes and detergents containing them |
| WO1995024471A1 (en) | 1994-03-08 | 1995-09-14 | Novo Nordisk A/S | Novel alkaline cellulases |
| WO1995030744A2 (en) | 1994-05-04 | 1995-11-16 | Genencor International Inc. | Lipases with improved surfactant resistance |
| WO1995035381A1 (en) | 1994-06-20 | 1995-12-28 | Unilever N.V. | Modified pseudomonas lipases and their use |
| WO1996011262A1 (en) | 1994-10-06 | 1996-04-18 | Novo Nordisk A/S | An enzyme and enzyme preparation with endoglucanase activity |
| WO1996012012A1 (en) | 1994-10-14 | 1996-04-25 | Solvay S.A. | Lipase, microorganism producing same, method for preparing said lipase and uses thereof |
| WO1996013580A1 (en) | 1994-10-26 | 1996-05-09 | Novo Nordisk A/S | An enzyme with lipolytic activity |
| WO1996023873A1 (en) | 1995-02-03 | 1996-08-08 | Novo Nordisk A/S | Amylase variants |
| US5942431A (en) | 1995-02-27 | 1999-08-24 | Novo Nordisk A/S | DNA sequences encoding lipases and method for producing lipases |
| WO1996029397A1 (en) | 1995-03-17 | 1996-09-26 | Novo Nordisk A/S | Novel endoglucanases |
| WO1996034946A1 (en) | 1995-05-05 | 1996-11-07 | Novo Nordisk A/S | Protease variants and compositions |
| DE19600743A1 (en) | 1996-01-11 | 1997-07-24 | Henkel Kgaa | Use of mixture of glyco-lipid and surfactant in hand dish-washing detergent |
| WO1997043424A1 (en) | 1996-05-14 | 1997-11-20 | Genencor International, Inc. | MODIFIED α-AMYLASES HAVING ALTERED CALCIUM BINDING PROPERTIES |
| WO1998008940A1 (en) | 1996-08-26 | 1998-03-05 | Novo Nordisk A/S | A novel endoglucanase |
| WO1998012307A1 (en) | 1996-09-17 | 1998-03-26 | Novo Nordisk A/S | Cellulase variants |
| WO1998015257A1 (en) | 1996-10-08 | 1998-04-16 | Novo Nordisk A/S | Diaminobenzoic acid derivatives as dye precursors |
| WO1998020116A1 (en) | 1996-11-04 | 1998-05-14 | Novo Nordisk A/S | Subtilase variants and compositions |
| WO1998020115A1 (en) | 1996-11-04 | 1998-05-14 | Novo Nordisk A/S | Subtilase variants and compositions |
| DE19648439A1 (en) | 1996-11-22 | 1998-05-28 | Henkel Kgaa | Skin-friendly washing=up liquid with good cleaning performance |
| WO1999001544A1 (en) | 1997-07-04 | 1999-01-14 | Novo Nordisk A/S | FAMILY 6 ENDO-1,4-β-GLUCANASE VARIANTS AND CLEANING COMPOSIT IONS CONTAINING THEM |
| WO1999011768A1 (en) | 1997-08-29 | 1999-03-11 | Novo Nordisk A/S | Protease variants and compositions |
| WO1999019467A1 (en) | 1997-10-13 | 1999-04-22 | Novo Nordisk A/S | α-AMYLASE MUTANTS |
| US6472364B1 (en) | 1998-10-13 | 2002-10-29 | The Procter & Gamble Company | Detergent compositions or components |
| WO2000060063A1 (en) | 1999-03-31 | 2000-10-12 | Novozymes A/S | Lipase variant |
| WO2001016285A2 (en) | 1999-08-31 | 2001-03-08 | Novozymes A/S | Novel proteases and variants thereof |
| WO2001044452A1 (en) | 1999-12-15 | 2001-06-21 | Novozymes A/S | Subtilase variants having an improved wash performance on egg stains |
| WO2001066712A2 (en) | 2000-03-08 | 2001-09-13 | Novozymes A/S | Variants with altered properties |
| WO2001092502A1 (en) | 2000-06-02 | 2001-12-06 | Novozymes A/S | Cutinase variants |
| WO2002010355A2 (en) | 2000-08-01 | 2002-02-07 | Novozymes A/S | Alpha-amylase mutants with altered stability |
| CN1337439A (en) | 2000-08-09 | 2002-02-27 | 大庆油田有限责任公司勘探开发研究院 | Oil-displacing agent and its application |
| WO2002016547A2 (en) | 2000-08-21 | 2002-02-28 | Novozymes A/S | Subtilase enzymes |
| WO2002099091A2 (en) | 2001-06-06 | 2002-12-12 | Novozymes A/S | Endo-beta-1,4-glucanase from bacillus |
| EP1411111A1 (en) | 2001-06-27 | 2004-04-21 | Saraya Co., Ltd. | Low foaming detergent compositions |
| WO2003002700A1 (en) | 2001-06-27 | 2003-01-09 | Saraya Co., Ltd. | Low-foaming detergent compositions |
| EP1411111B1 (en) | 2001-06-27 | 2008-09-10 | Saraya Co., Ltd. | Low foaming detergent compositions |
| WO2003006602A2 (en) | 2001-07-12 | 2003-01-23 | Novozymes A/S | Subtilase variants |
| WO2003006146A1 (en) | 2001-07-13 | 2003-01-23 | Cognis France S.A. | Aqueous preparations |
| US7262042B2 (en) | 2001-12-20 | 2007-08-28 | Henkel Kommanditgesellschaft Auf Aktien (Henkel Kgaa) | Alkaline protease from Bacillus gibsonii (DSM 14393) and washing and cleaning products comprising said alkaline protease |
| EP1382668A1 (en) | 2002-06-11 | 2004-01-21 | Unilever N.V. | Detergent tablets |
| WO2004074419A2 (en) | 2003-02-18 | 2004-09-02 | Novozymes A/S | Detergent compositions |
| WO2005040372A1 (en) | 2003-10-23 | 2005-05-06 | Novozymes A/S | Protease with improved stability in detergents |
| WO2005052161A2 (en) | 2003-11-19 | 2005-06-09 | Genencor International, Inc. | Serine proteases, nucleic acids encoding serine enzymes and vectors and host cells incorporating same |
| WO2005052146A2 (en) | 2003-11-19 | 2005-06-09 | Genencor International, Inc. | Serine proteases, nucleic acids encoding serine enzymes and vectors and host cells incorporating same |
| WO2005056782A2 (en) | 2003-12-03 | 2005-06-23 | Genencor International, Inc. | Perhydrolase |
| US7556654B1 (en) | 2004-10-15 | 2009-07-07 | Naturell | Methods for cleaning materials |
| EP1705241A1 (en) | 2005-03-23 | 2006-09-27 | Unilever N.V. | Detergent compositions in tablet form |
| WO2006130575A2 (en) | 2005-05-31 | 2006-12-07 | The Procter & Gamble Company | Polymer-containing detergent compositions and their use |
| WO2007001262A1 (en) | 2005-06-17 | 2007-01-04 | The Procter & Gamble Company | Organic catalyst with enhanced enzyme compatibility |
| EP1746109A2 (en) | 2005-07-21 | 2007-01-24 | National Starch and Chemical Investment Holding Corporation | Hybrid copolymers |
| WO2007044993A2 (en) | 2005-10-12 | 2007-04-19 | Genencor International, Inc. | Use and production of storage-stable neutral metalloprotease |
| WO2007087508A2 (en) | 2006-01-23 | 2007-08-02 | Novozymes A/S | Lipase variants |
| US20080213194A1 (en) | 2006-07-27 | 2008-09-04 | Desanto Keith | Rhamnolipid-based formulations |
| WO2008153815A2 (en) | 2007-05-30 | 2008-12-18 | Danisco Us, Inc., Genencor Division | Variants of an alpha-amylase with improved production levels in fermentation processes |
| US20090011970A1 (en) | 2007-07-02 | 2009-01-08 | Marc Francois Theophile Evers | Laundry multi-compartment pouch composition |
| WO2009021867A2 (en) | 2007-08-10 | 2009-02-19 | Henkel Ag & Co. Kgaa | Agents containing proteases |
| WO2009061380A2 (en) | 2007-11-05 | 2009-05-14 | Danisco Us Inc., Genencor Division | VARIANTS OF BACILLUS sp. TS-23 ALPHA-AMYLASE WITH ALTERED PROPERTIES |
| WO2009067279A1 (en) | 2007-11-21 | 2009-05-28 | E.I. Du Pont De Nemours And Company | Production of peracids using an enzyme having perhydrolysis activity |
| WO2009092699A1 (en) | 2008-01-24 | 2009-07-30 | Unilever Nv | Machine dishwash detergent compositions |
| WO2009102854A1 (en) | 2008-02-15 | 2009-08-20 | The Procter & Gamble Company | Cleaning compositions |
| US20110091399A1 (en) | 2008-05-15 | 2011-04-21 | Evonik Goldschmidt Gmbh | Use of organo-modified siloxane block copolymers for producing cosmetic or pharmaceutical compositions |
| WO2010065455A2 (en) | 2008-12-01 | 2010-06-10 | Danisco Us Inc. | Enzymes with lipase activity |
| WO2010100028A2 (en) | 2009-03-06 | 2010-09-10 | Huntsman Advanced Materials (Switzerland) Gmbh | Enzymatic textile bleach-whitening methods |
| WO2010107560A2 (en) | 2009-03-18 | 2010-09-23 | Danisco Us Inc. | Fungal cutinase from magnaporthe grisea |
| WO2010111143A2 (en) | 2009-03-23 | 2010-09-30 | Danisco Us Inc. | Cal a-related acyltransferases and methods of use, thereof |
| WO2011084412A1 (en) | 2009-12-21 | 2011-07-14 | Danisco Us Inc. | Detergent compositions containing thermobifida fusca lipase and methods of use thereof |
| WO2011084417A1 (en) | 2009-12-21 | 2011-07-14 | Danisco Us Inc. | Detergent compositions containing geobacillus stearothermophilus lipase and methods of use thereof |
| WO2011084599A1 (en) | 2009-12-21 | 2011-07-14 | Danisco Us Inc. | Detergent compositions containing bacillus subtilis lipase and methods of use thereof |
| WO2011098531A1 (en) | 2010-02-10 | 2011-08-18 | Novozymes A/S | Variants and compositions comprising variants with high stability in presence of a chelating agent |
| WO2011150157A2 (en) | 2010-05-28 | 2011-12-01 | Danisco Us Inc. | Detergent compositions containing streptomyces griseus lipase and methods of use thereof |
| WO2012137147A1 (en) | 2011-04-08 | 2012-10-11 | Danisco Us, Inc. | Compositions |
| WO2013001087A2 (en) | 2011-06-30 | 2013-01-03 | Novozymes A/S | Method for screening alpha-amylases |
| WO2013001078A1 (en) | 2011-06-30 | 2013-01-03 | Novozymes A/S | Alpha-amylase variants |
| CN102660392A (en) | 2012-05-29 | 2012-09-12 | 西北民族大学 | Detergent for wool |
| CN104031899A (en) | 2013-03-05 | 2014-09-10 | 中国科学院天津工业生物技术研究所 | Lipase and application thereof |
| EP2786743A1 (en) | 2013-04-02 | 2014-10-08 | Evonik Industries AG | Mixture composition containing rhamnolipids |
| EP2787065A1 (en) | 2013-04-02 | 2014-10-08 | Evonik Industries AG | Detergent composition for textiles comprising rhamnolipids having a predominant share of di-rhamnolipids |
| WO2016075179A1 (en) | 2014-11-11 | 2016-05-19 | Clariant International Ltd | Laundry detergents containing soil release polymers |
| WO2016075178A1 (en) | 2014-11-11 | 2016-05-19 | Clariant International Ltd | Laundry detergents containing soil release polymers |
| EP3061442A1 (en) | 2015-02-27 | 2016-08-31 | Evonik Degussa GmbH | Composition comprising rhamnolipid and siloxane |
| CN106854531A (en) | 2017-01-04 | 2017-06-16 | 长沙协浩吉生物工程有限公司 | A kind of compound method of woollen and cashmere anti-shrinkage machine washing liquid |
| EP3489338A1 (en) | 2017-11-28 | 2019-05-29 | Clariant International Ltd | Detergent compositions containing renewably sourced soil release polyesters |
| EP3489340A1 (en) | 2017-11-28 | 2019-05-29 | Clariant International Ltd | Renewably sourced soil release polyesters |
| WO2019105675A1 (en) | 2017-11-30 | 2019-06-06 | Unilever Plc | Detergent composition comprising protease |
| US20210171864A1 (en) | 2019-09-29 | 2021-06-10 | Tianjin Zhongtian Jingke Technology Co., Ltd. | Soaping Agent And Preparation Method And Application Thereof |
| WO2022162043A1 (en) | 2021-01-28 | 2022-08-04 | Novozymes A/S | Lipase with low malodor generation |
Non-Patent Citations (3)
| Title |
|---|
| LEI ET AL., BIOTECHNOL LETT., vol. 42, no. 6, June 2020 (2020-06-01), pages 997 - 1002 |
| R.J. SIEZENJ.A. LEUNISSEN, PROTEIN SCIENCE, vol. 6, 1997, pages 501 - 523 |
| R.J. SIEZENW.M. DE VOSJ.A.M. LEUNISSENB.W. DIJKSTRA, PROTEIN ENGINEERING, DESIGN AND SELECTION, vol. 4, 1991, pages 719 - 737 |
Also Published As
| Publication number | Publication date |
|---|---|
| WO2025036642A1 (en) | 2025-02-20 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP3786272A1 (en) | Unit dose liquid laundry detergent composition | |
| CN110088261B (en) | Cleaning compositions containing enzymes | |
| CN103517975B (en) | Water-based concentrates laundry detergent composition | |
| JP2019059943A (en) | Preventing adhesion of bacteria | |
| JP6957727B2 (en) | Functionalized siloxane polymer and composition containing it | |
| CN109072133A (en) | It is used to handle the purposes of fabric with the active polypeptide of DNA enzymatic | |
| JP2013503950A (en) | Condensable liquid laundry detergent composition | |
| EP3686265A1 (en) | Detergent composition with sophorolipids | |
| JP7000554B2 (en) | Functionalized siloxane polymer and composition containing it | |
| CA3043443A1 (en) | Stabilization of enzymes in compositions | |
| KR20170107073A (en) | Acidic liquid compression cleaners containing hydroxycarboxylic acids, non-ionic surfactants and enzymes | |
| EP4045625A1 (en) | Storage-stable hydrolase containing liquids | |
| WO2025036643A1 (en) | Biosurfactant for washing wool | |
| US12448588B2 (en) | Composition comprising biosurfactant and persicomycin (3-(3′hydroxy)hydroxy fatty acid) | |
| US20250171719A1 (en) | Bio based composition | |
| DE102015215591A1 (en) | Low-water, two-phase liquid detergent with acidic pH | |
| EP4479501A1 (en) | Concentrated free-flowing detergent composition having improved properties | |
| EP2918664A2 (en) | Washing or cleaning agent comprising hydrolytically active enzyme and steroid acids | |
| WO2026032788A1 (en) | Improved method for reducing malodor | |
| WO2026032783A1 (en) | Bacillus velezensis strains in cleaning and animal feeding | |
| WO2026032787A1 (en) | Bacillus subtilis strains in cleaning solutions and animal feeding | |
| WO2026032784A1 (en) | Bacillus velezensis strains in cleaning and animal feeding | |
| WO2026032785A1 (en) | Bacillus licheniformis strains in cleaning and animal feeding | |
| EP4692292A1 (en) | Improved method for germinating bacterial spores | |
| EP4692293A1 (en) | Composition |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 24746266 Country of ref document: EP Kind code of ref document: A1 |