WO2025035143A1 - Compositions and methods for treatment of spinal muscular atrophy - Google Patents
Compositions and methods for treatment of spinal muscular atrophy Download PDFInfo
- Publication number
- WO2025035143A1 WO2025035143A1 PCT/US2024/041840 US2024041840W WO2025035143A1 WO 2025035143 A1 WO2025035143 A1 WO 2025035143A1 US 2024041840 W US2024041840 W US 2024041840W WO 2025035143 A1 WO2025035143 A1 WO 2025035143A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- sequence
- aav
- seq
- capsid
- recombinant
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K14/00—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- C07K14/435—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- C07K14/46—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans from vertebrates
- C07K14/47—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans from vertebrates from mammals
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K48/00—Medicinal preparations containing genetic material which is inserted into cells of the living body to treat genetic diseases; Gene therapy
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K48/00—Medicinal preparations containing genetic material which is inserted into cells of the living body to treat genetic diseases; Gene therapy
- A61K48/005—Medicinal preparations containing genetic material which is inserted into cells of the living body to treat genetic diseases; Gene therapy characterised by an aspect of the 'active' part of the composition delivered, i.e. the nucleic acid delivered
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K48/00—Medicinal preparations containing genetic material which is inserted into cells of the living body to treat genetic diseases; Gene therapy
- A61K48/005—Medicinal preparations containing genetic material which is inserted into cells of the living body to treat genetic diseases; Gene therapy characterised by an aspect of the 'active' part of the composition delivered, i.e. the nucleic acid delivered
- A61K48/0058—Nucleic acids adapted for tissue specific expression, e.g. having tissue specific promoters as part of a contruct
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K48/00—Medicinal preparations containing genetic material which is inserted into cells of the living body to treat genetic diseases; Gene therapy
- A61K48/005—Medicinal preparations containing genetic material which is inserted into cells of the living body to treat genetic diseases; Gene therapy characterised by an aspect of the 'active' part of the composition delivered, i.e. the nucleic acid delivered
- A61K48/0066—Manipulation of the nucleic acid to modify its expression pattern, e.g. enhance its duration of expression, achieved by the presence of particular introns in the delivered nucleic acid
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K48/00—Medicinal preparations containing genetic material which is inserted into cells of the living body to treat genetic diseases; Gene therapy
- A61K48/0075—Medicinal preparations containing genetic material which is inserted into cells of the living body to treat genetic diseases; Gene therapy characterised by an aspect of the delivery route, e.g. oral, subcutaneous
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K14/00—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- C07K14/435—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- C07K14/46—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans from vertebrates
- C07K14/47—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans from vertebrates from mammals
- C07K14/4701—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans from vertebrates from mammals not used
- C07K14/4702—Regulators; Modulating activity
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K14/00—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- C07K14/435—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- C07K14/46—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans from vertebrates
- C07K14/47—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans from vertebrates from mammals
- C07K14/4701—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans from vertebrates from mammals not used
- C07K14/4707—Muscular dystrophy
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N15/00—Mutation or genetic engineering; DNA or RNA concerning genetic engineering, vectors, e.g. plasmids, or their isolation, preparation or purification; Use of hosts therefor
- C12N15/09—Recombinant DNA-technology
- C12N15/63—Introduction of foreign genetic material using vectors; Vectors; Use of hosts therefor; Regulation of expression
- C12N15/79—Vectors or expression systems specially adapted for eukaryotic hosts
- C12N15/85—Vectors or expression systems specially adapted for eukaryotic hosts for animal cells
- C12N15/86—Viral vectors
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K38/00—Medicinal preparations containing peptides
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2750/00—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA ssDNA viruses
- C12N2750/00011—Details
- C12N2750/14011—Parvoviridae
- C12N2750/14111—Dependovirus, e.g. adenoassociated viruses
- C12N2750/14141—Use of virus, viral particle or viral elements as a vector
- C12N2750/14143—Use of virus, viral particle or viral elements as a vector viral genome or elements thereof as genetic vector
Definitions
- SMA Spinal muscular atrophy
- SMA is an autosomal recessive disorder caused by mutations in the SMN1 gene resulting in reduced expression of survival motor neuron (SMN) protein. It is the most common genetic cause of infant mortality and the worldwide incidence of SMA is ⁇ 1 in 10,000 live births. SMN proteins have roles in spliceosomal assembly, protein translation, cytoskeletal dynamics, and mitochondrial function. Disruption of the SMA motor unit (motor neurons and the muscle fibers they innervate) underlies the most evident clinical manifestation of SMA (skeletal muscle weakness). Infants with SMA type 1 have severe manifestations, with profound hypotonia and progressive respiratory and swallowing difficulties. Patient management typically includes physiotherapy, orthopedic support, respiratory care, and management of bulbar impairment.
- nusinersen Spinraza
- Zolgensma onastage abeparvovec
- AAV adeno-associated virus
- risdiplam Evrysdi
- a recombinant adeno-associated virus comprising an AAV capsid containing a vector genome, wherein the vector genome comprises an expression cassette comprising: (a) a UbC promoter sequence, (b) a coding sequence for a functional human survival of motor neuron 1 (SMN1) protein, (c) at least four miR182 target sequences, and (d) a polyA sequence, wherein the coding sequence, the at least four miR182 target sequences, and the polyA sequence are operably linked to the UbC promoter sequence.
- the recombinant AAV of claim 1 wherein the SMN1 protein is an SMN1 isoform D protein.
- the coding sequence comprises the nucleotide sequence of SEQ ID NO: 7 or a nucleotide sequence at least 95% identical to SEQ ID NO: 7.
- the recombinant AAV is not a self- complementary AAV vector.
- composition comprising a stock of a recombinant AAV in an aqueous suspension media.
- the suspension is formulated for intrathecal delivery, optionally wherein the intrathecal delivery is intracerebroventricular (ICV) injection or intraci sternal magna (ICM) injection.
- ICV intracerebroventricular
- ICM intraci sternal magna
- compositions comprising a recombinant AAV and an aqueous formulation buffer.
- the composition is formulated for intrathecal delivery.
- the composition is formulated for intracerebroventricular (ICV) injection or intracistemal magna (ICM) injection.
- ICV intracerebroventricular
- ICM intracistemal magna
- a recombinant nucleic acid molecule comprising an expression cassette comprising: (a) a 5’ AAV ITR sequence, (b) a UbC promoter sequence, (c) a coding sequence for a functional human survival of motor neuron 1 (SMN1) protein, (d) at least four miR182 target sequences, (e) a polyA sequence, and (f) a 3’ AAV ITR sequence, wherein the coding sequence, the at least four miR182 target sequences, and the polyA sequence are operably linked to the UbC promoter sequence.
- the SMN1 protein is an SMN1 isoform D protein.
- the coding sequence comprises the nucleotide sequence of SEQ ID NO: 7 or a nucleotide sequence at least 95% identical to SEQ ID NO: 7.
- a plasmid comprising the nucleic acid is provided.
- a packaging host cell comprising the recombinant nucleic acid molecule or the plasmid.
- a recombinant AAV production system useful for producing a recombinant AAV.
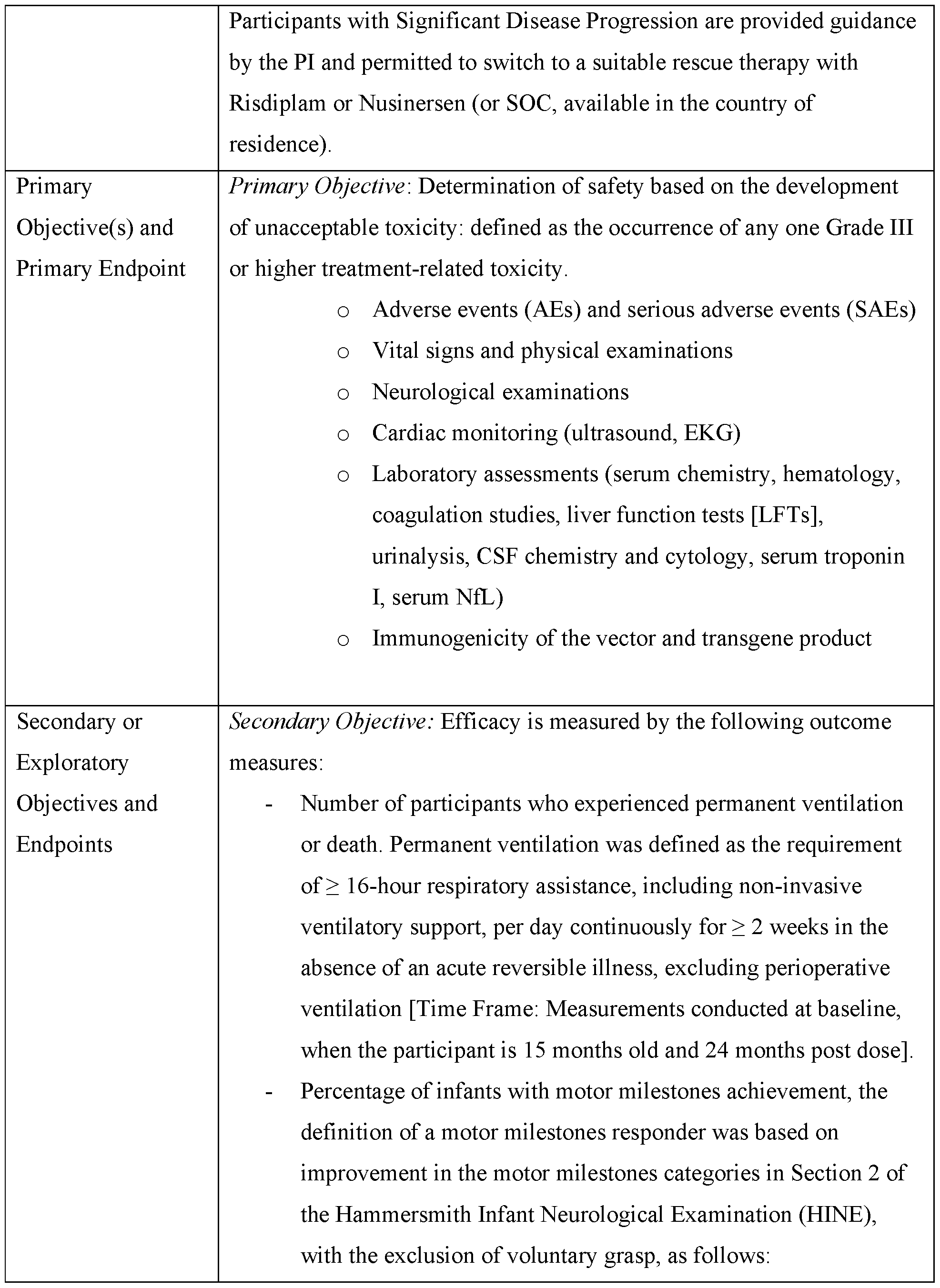
- a method for treating spinal muscular atrophy (SMA) in a subject in need thereof comprising administering to the subject an aqueous suspension comprising a recombinant AAV as described herein.
- the subject has type I (Werdnig-Hoffmann disease), type II (Dubowitz disease), type III (Kugelberg- Welander disease), or type IV (adult) SMA.
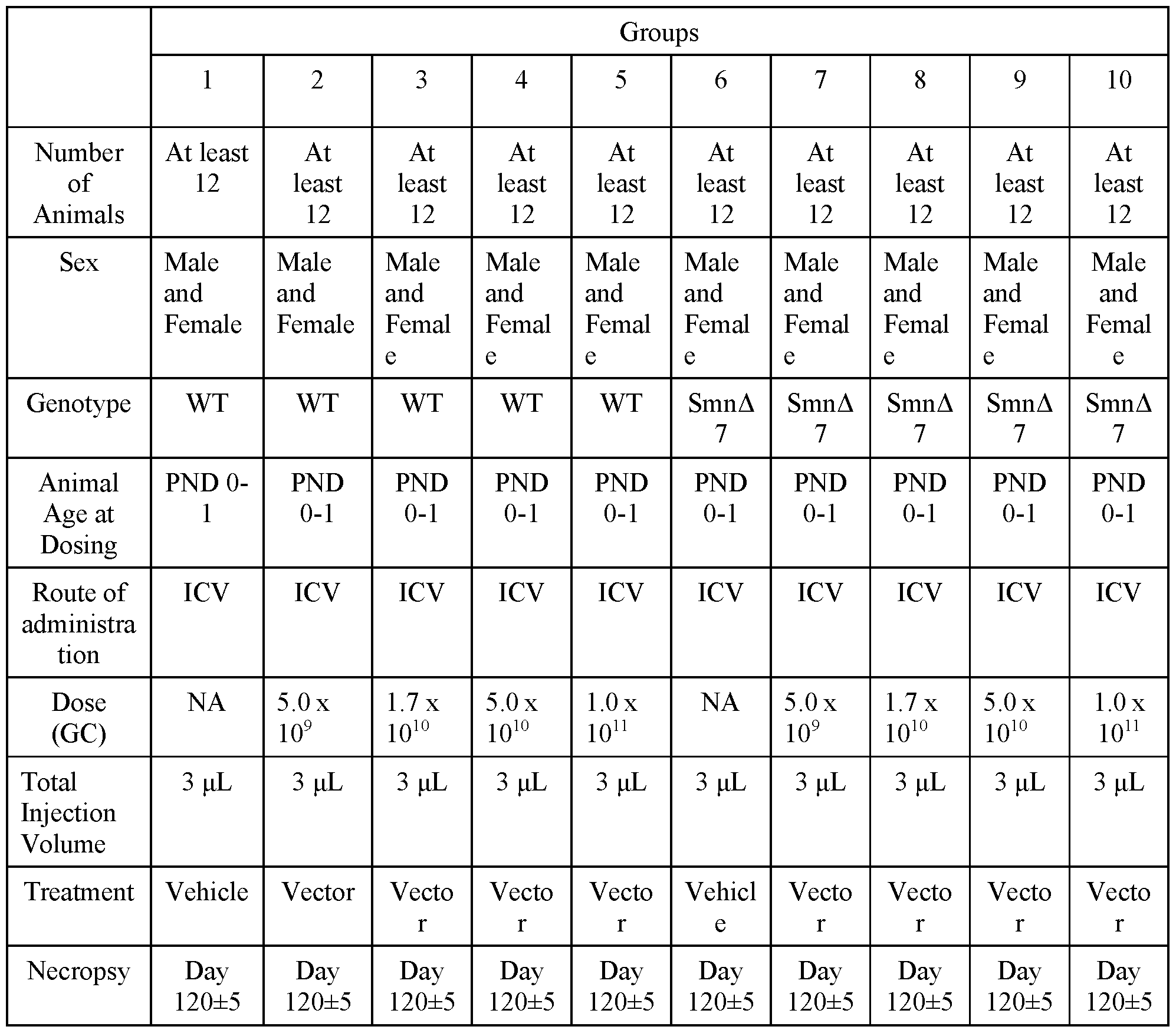
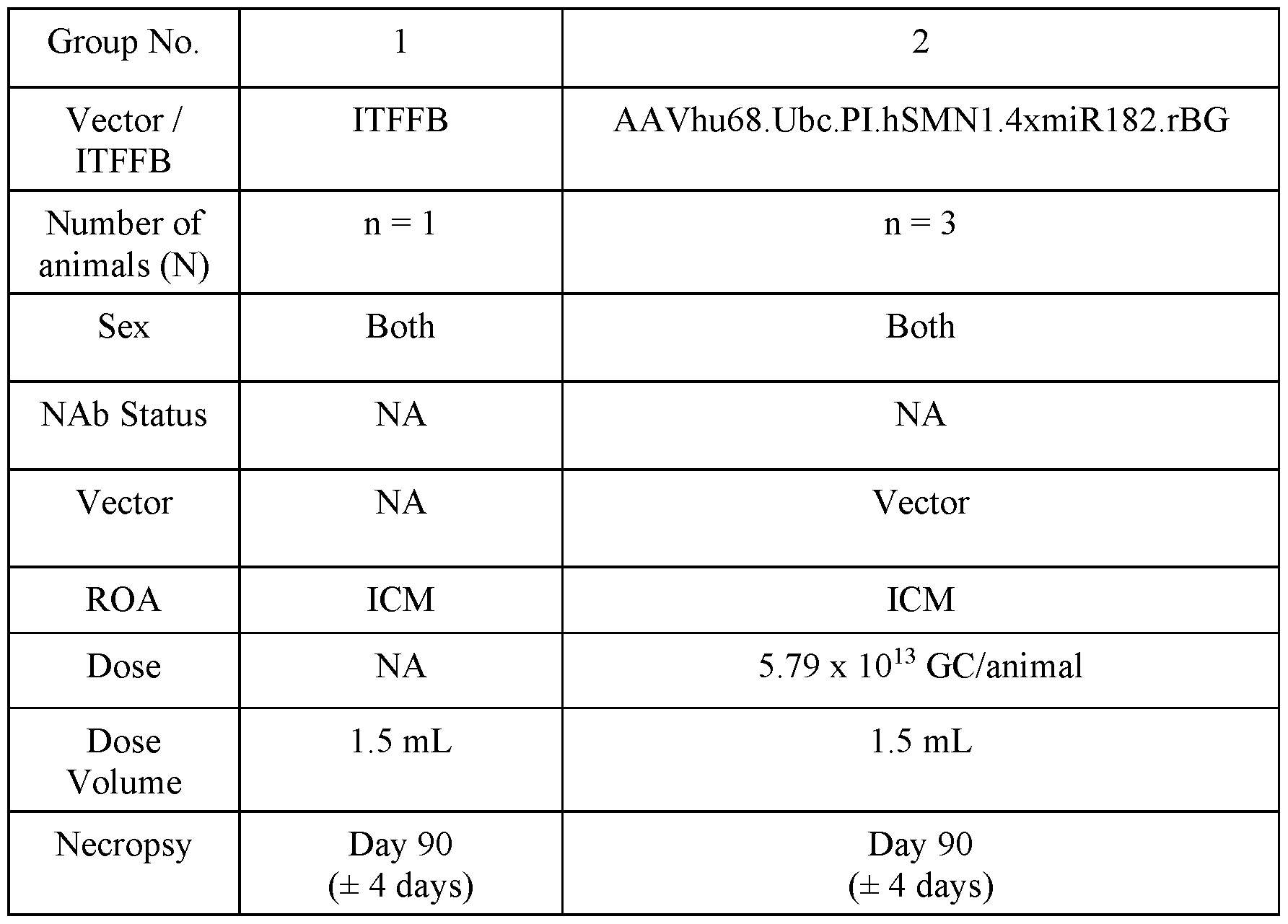
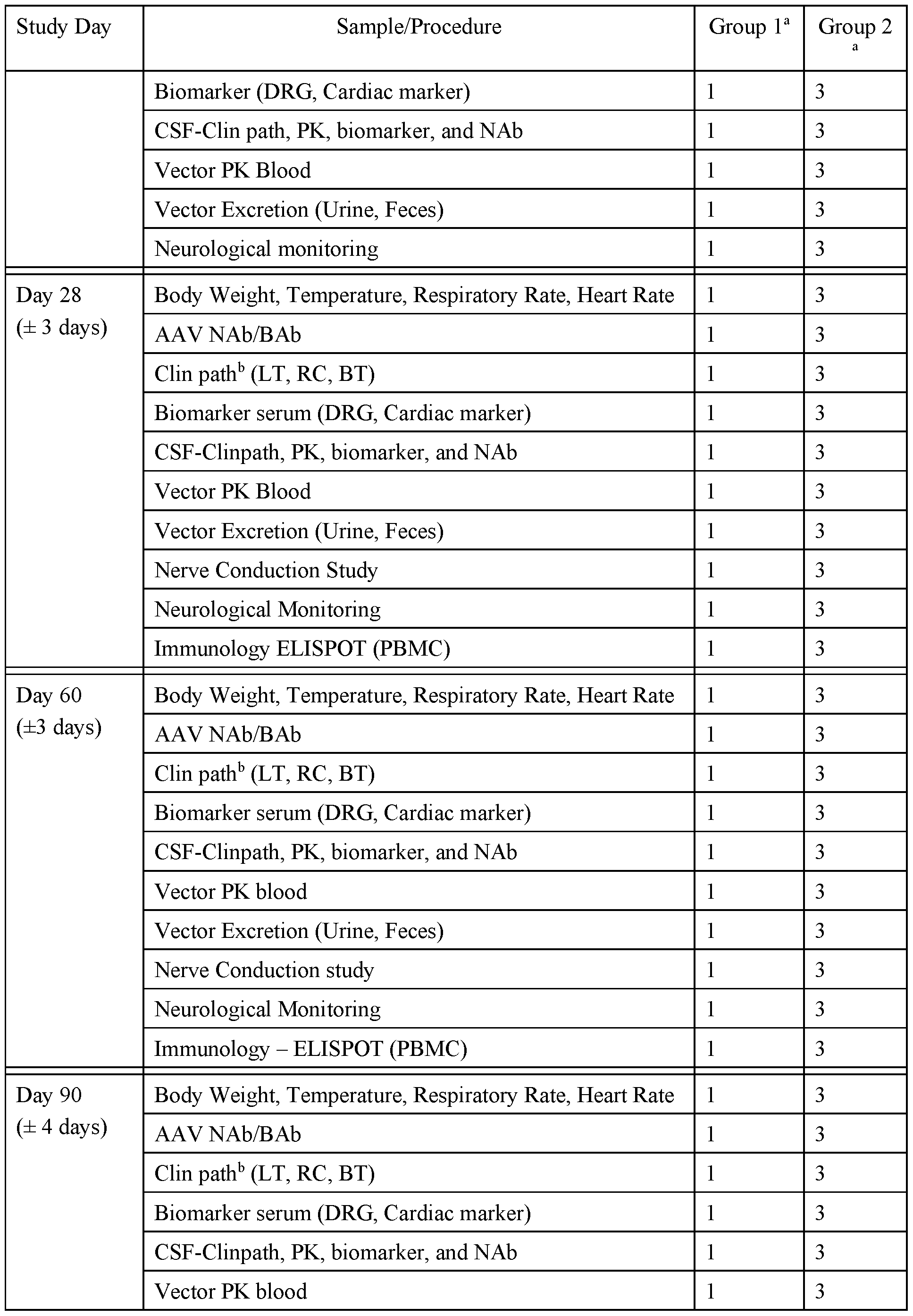
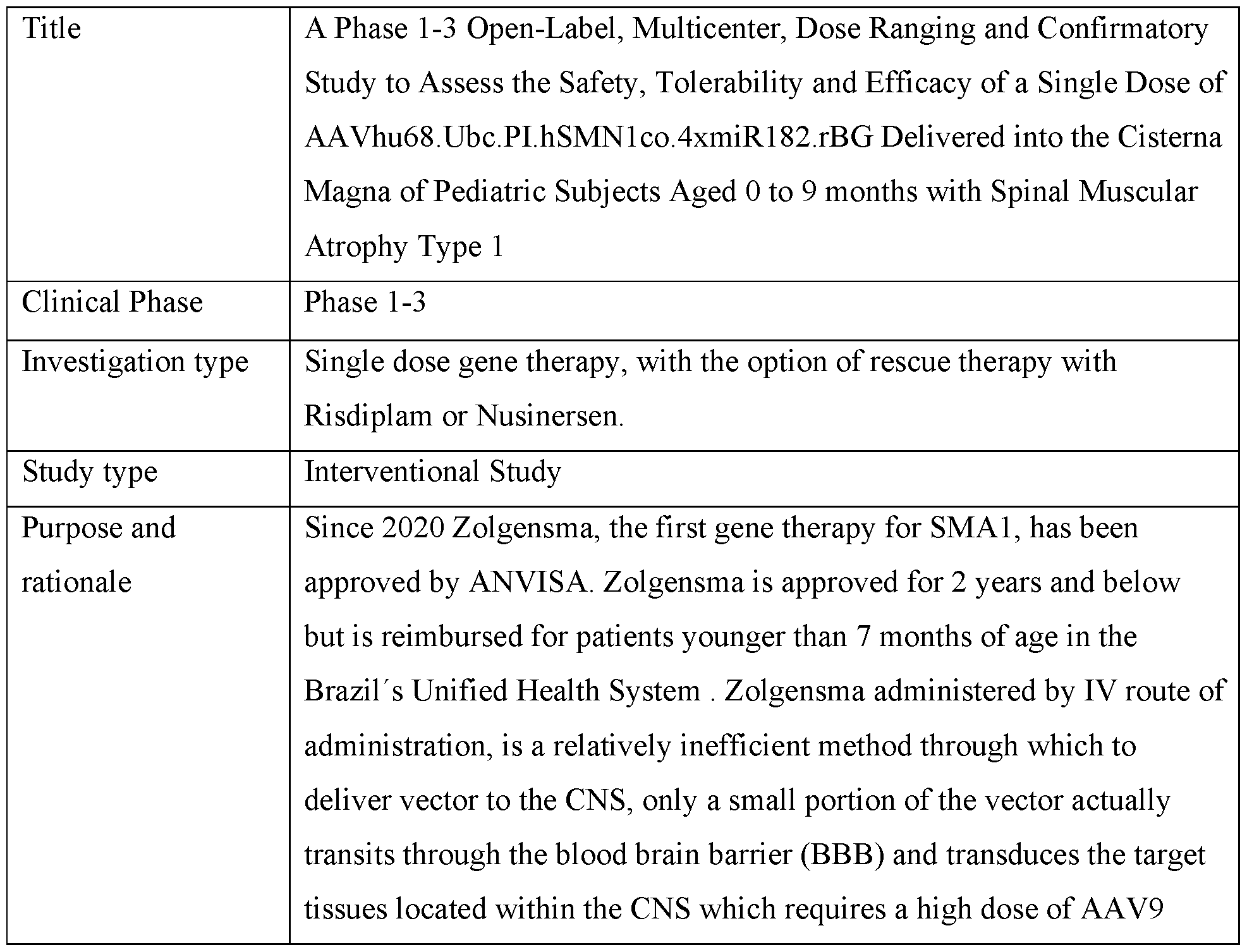
- FIG. 1 provides schematics of rAAV.hSMNl vector genomes.
- FIG. 2 shows ISH for detection of hSMNl expression in the liver of wildtype following IV administration of vectors with different promoters.
- FIG. 3 shows survival of SMNA7 mice following administration of AAVhu68. hSMNl vectors.
- FIG. 4 shows body weights of SMNA7 mice following administration of AAVhu68. hSMNl vectors.
- FIG. 5 shows righting reflex (left) and clasping (right) responses of SMNA7 mice following administration of AAVhu68. hSMNl vectors.
- FIG. 6 shows survival, body weights, and clasping response scores of C57BL/6J mice following ICV administration of AAVhu68. hSMNl vectors.
- FIG. 7A and FIG. 7B show levels of neuron transduction by measurement of transduced motor neurons by ISH (ChAT+hSMNl transgene+ cells) (FIG. 7A) and cardiac pathology scores following ICV administration of a AAVhu68.CB7.CI.hSMNlco.rBG or AAVhu68.Ubc.PI.hSMNlco.4xmiR182.rBG vector to SMNA7 mice.
- FIG. 8 A and FIG. 8B show levels of expression of hSMNl (ISH intensity) (FIG. 8 A) and quantification of SMN+ChAT+ cells (FIG. 9B) following ICM administration of AAVhu68.CB7.CI.hSMNlco.rBG (CB7), AAVhu68.CB7.CI.hSMNlco.4xmiR182.rBG (CB7.4xmiR182), and AAVhu68.UbC.PI.hSMNlco.4xmiR182.rBG (UBC.4xmiR182) vectors.
- FIG. 8 A and FIG. 8B show levels of expression of hSMNl (ISH intensity) (FIG. 8 A) and quantification of SMN+ChAT+ cells (FIG. 9B) following ICM administration of AAVhu68.CB7.CI.hSMNlco.rBG (CB7), AAVhu68.CB7.CI.
- CSF WBC counts left
- CSF RBC counts right
- results of clinical pathology studies including CSF WBC counts (left) and CSF RBC counts (right) following ICM administration of AAVhu68.CB7.CI.hSMNlco.rBG, AAVhu68.CB7.CI.hSMNlco.4xmiR182.rBG, and AAVhu68.UbC.PI.hSMNlco.4xmiR182.rBG vectors to NHPs.
- FIG. 10 shows results of nerve conduction studies following ICM administration of AAVhu68.CB7.CI.hSMNlco.rBG, AAVhu68.CB7.CI.hSMNlco.4xmiR182.rBG, and AAVhu68.UbC.PI.hSMNlco.4xmiR182.rBG vectors to NHPs.
- FIG. 11 shows results from assessments of DRG neuronal degeneration following ICM administration of AAVhu68.CB7.CI.hSMNlco.rBG, AAVhu68.CB7.CI.hSMNlco.4xmiR182.rBG, and AAVhu68.UbC.PI.hSMNlco.4xmiR182.rBG vectors to NHPs.
- FIG. 12 shows results from assessments of spinal cord axonopathy following ICM administration of AAVhu68.CB7.CI.hSMNlco.rBG, AAVhu68.CB7.CI.hSMNlco.4xmiR182.rBG, and AAVhu68.UbC.PI.hSMNlco.4xmiR182.rBG vectors to NHPs.
- FIG. 13 A and FIG. 13B show results from scoring of axonopathy severity (FIG. 13 A) and measurements of NfL in CSF on day 29 (FIG. 13B) following ICM administration of AAVhu68.CB7.CI.hSMNlco.rBG, AAVhu68.CB7.CI.hSMNlco.4xmiR182.rBG, and AAVhu68.UbC.PI.hSMNlco.4xmiR182.rBG vectors to NHPs.
- FIG. 14A and FIG. 14B show spinal cord axonopathy scores versus NfL levels in CSF (FIG. 14A) and serum (FIG. 14B) following ICM administration of AAVhu68.CB7.CI.hSMNlco.rBG, AAVhu68.CB7.CI.hSMNlco.4xmiR182.rBG, and AAVhu68.UbC.PI.hSMNlco.4xmiR182.rBG vectors to NHPs.
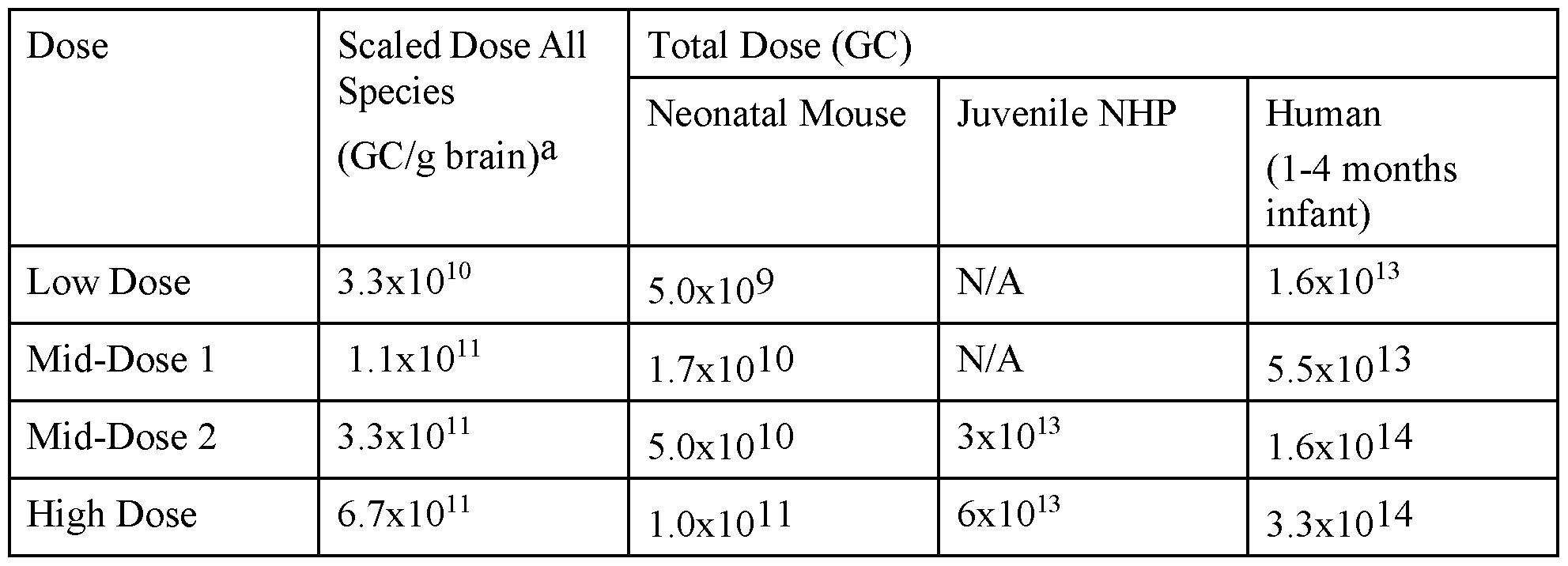
- FIG. 15 shows survival of SMNA7 mice following ICV administration of various doses of an AA Vhu68.Ubc.PI.hSMN1.4xmiRl 82. rBG vector.
- FIG. 16 shows survival of wildtype mice following ICV administration of various doses of an AAVhu68.Ubc.PI.hSMN1.4xmiRl 82. rBG vector.
- FIG. 17 shows body weights of SMNA7 mice following ICV administration of various doses of an AAVhu68.Ubc.PI.hSMN1.4xmiRl 82. rBG vector.
- FIG. 18 shows body weights of wildtype mice following ICV administration of various doses of an AAVhu68.Ubc.PI.hSMN1.4xmiRl 82. rBG vector.
- FIG. 19 shows results from evaluation of righting reflexes in SMNA7 mice following ICV administration of the indicated doses of an AAVhu68.Ubc.PI.hSMN1.4xmiR182.rBG vector.
- FIG. 20 shows results from evaluation of hindlimb clasping responses in wildtype mice following ICV administration of the indicated doses of an AAVhu68.Ubc.PI.hSMNl ,4xmiRl 82.rBG vector.
- FIG. 21 shows results from evaluation of hindlimb clasping responses in SMNA7 mice following ICV administration of the indicated of an AAVhu68.Ubc.PI.hSMNl ,4xmiRl 82.rBG vector.
- FIG. 22 shows results of nerve conduction studies (NCS) following ICM administration of AAVhu68.Ubc.PI.hSMN1.4xmiR182.rBG to NHPs.
- FIG. 23 shows results of clinical pathology studies, including CSF WBC counts (top left), CSF RBC counts (top right), CSF protein levels (bottom left), and CSF glucose levels (bottom right) following ICM administration of AAVhu68.Ubc.PI.hSMN1.4xmiR182.rBG to NHP.
- FIG. 24 shows results from evaluation of liver enzyme levels following ICM administration of AAVhu68.Ubc.PI.hSMN1.4xmiR182.rBG to NHPs.
- FIG. 25 A - FIG. 25C show results from evaluation of liver function by detection of total protein, albumin, globulin, and albumin/globulin ratios (A/G) (FIG. 25 A), measurement of creatine levels (FIG. 25B), and measurement of blood urea nitrogen (BUN) levels (FIG. 25C) following ICM administration of AAVhu68.Ubc.PI.hSMN1.4xmiR182.rBG to NHPs.
- FIG. 26 shows results from evaluation of blood clinical pathology following ICM administration of AAVhu68.Ubc.PI.hSMN1.4xmiR182.rBG to NHPs.
- AAV vectors having an AAV capsid containing a nucleic acid having a sequence encoding a human survival of motor neuron (SMN1) gene under control of regulatory sequences.
- AAV adeno-associated virus
- the vectors, as well as compositions containing these vectors, are useful for treatment of SMA in patients need thereof.
- SMA spinal muscular atrophy or proximal spinal muscular atrophy
- SMA is a genetic, neurodegenerative disorder characterized by the loss of spinal motor neurons.
- SMA is an autosomal recessive disease of early onset and is currently a leading cause of death among infants.
- the severity of SMA varies among patients and has thus been classified into different types depending on the age of onset and motor development milestones.
- SMA 0 designation has been proposed to reflect prenatal onset and severe joint contractures, facial diplegia, and respiratory failure.
- Three types of post-natal form of SMA have been designated.
- Type I SMA also called Werdnig-Hoffmann disease
- Type I SMA is the most severe form with onset at birth or within 6 months and typically results in death within 2 years.
- Type II SMA is the intermediate form with onset within the first 2 years. Children with Type II SMA are able to sit, but cannot stand or walk.
- Type III also called Kugelberg- Welander disease
- Type III SMA begins after 18 months to 2 years of age (Lefebvre et al., Hum. Mol. Genet., 1998, 7, 1531-1536) and usually has a chronic evolution. Children with Type III SMA can stand and walk unaided at least in infancy.
- Adult form (type IV) is the mildest form of SMA, with onset after 30 years of age, and few cases have been reported. Type III and type IV SMA are also known as later- onset SMA.
- SMA survival motor neuron gene 1
- SMA survival motor neuron gene 1
- SMN2 survival motor neuron gene 2
- SMA survival motor neuron gene 1
- SMN2 contains a translationally silent mutation at position +6 of exon 7, which results in inefficient inclusion of exon 7 in SMN2 transcripts.
- the predominant form of SMN2 is a truncated version, lacking exon 7, which is unstable and inactive (Cartegni and Krainer, Nat. Genet., 2002, 30, 377-384).
- Expression of the SMN2 gene results in approximately 10-20% of the SMN protein and 80-90% of the unstable/non-functional SMN delta 7 protein.
- SMN protein plays a well- established role in assembly of the spliceosome and may also mediate mRNA trafficking in the axon and nerve terminus of neurons.
- “Patient” or “subject” or “individual” as used herein refer to a mammalian animal, including a human, a veterinary or farm animal, a domestic animal or pet, and animals normally used for clinical research. In certain embodiments, the subject is a human.
- the words “comprise”, “comprises”, and “comprising” are to be interpreted inclusively rather than exclusively.
- the words “consist”, “consisting”, and its variants, are to be interpreted exclusively, rather than inclusively. While various embodiments in the specification are presented using “comprising” language, under other circumstances, a related embodiment is also intended to be included and described using “consisting of’ or “consisting essentially of’ language.
- the term “about” refers to a variant of ⁇ 10% from the reference integer and values therebetween, unless otherwise specified.
- “about” 500 pM includes ⁇ 50 (i.e., 450 - 550, which includes the integers therebetween).
- the term “about” is inclusive of all values within the range including both the integer and fractions.
- the term “about” when used to modify a numerical value means a variation of ⁇ 10%, ( ⁇ 10%, e.g., ⁇ 1, ⁇ 2, ⁇ 3, ⁇ 4, ⁇ 5, ⁇ 6, ⁇ 7, ⁇ 8, ⁇ 9, ⁇ 10, or values therebetween) from the reference given, unless otherwise specified.
- “up to” a number includes the number (for example, 50).
- the term “in the range” or “within a range” (and similar statements) includes the endpoints of the stated range.
- the recitations of numerical ranges by endpoints include all numbers subsumed within that range (e.g., 1 to 5 includes 1, 1.5, 2, 2.75, 3, 3.80, 4, 5, etc.).
- E ⁇ # or the term “e+#” is used to reference an exponent.
- 5E10 or “5el0” is 5 x 10 10 . These terms may be used interchangeably.
- engineered nucleic acid constructs that include hSMNl coding sequences.
- the nucleic acids can be used for generating viral vectors for therapeutic delivery of the transgene to target cells of a subject, and/or for delivery of the SMN1 coding sequence to a host cell, e.g., naked DNA or plasmid.
- a host cell e.g., naked DNA or plasmid.
- an expression cassette comprising the hSMNl sequence is provided.
- an “expression cassette” refers to a nucleic acid molecule which comprises a biologically useful nucleic acid sequence (e.g., a gene cDNA encoding a protein, enzyme or other useful gene product, mRNA, etc.) and regulatory sequences operably linked thereto which direct or modulate transcription, translation, and/or expression of the nucleic acid sequence and its gene product.
- a biologically useful nucleic acid sequence e.g., a gene cDNA encoding a protein, enzyme or other useful gene product, mRNA, etc.
- regulatory sequences operably linked thereto which direct or modulate transcription, translation, and/or expression of the nucleic acid sequence and its gene product.
- the term an “expression cassette” refers to a nucleic acid molecule that includes an SMN1 encoding sequence and regulatory sequences therefore (e.g., promoter, enhancer, poly A sequences), which cassette may be packaged into the capsid of a viral vector (e.g., a viral particle).
- such an expression cassette for generating a viral vector contains the hSMN sequences described herein flanked by packaging signals of the viral genome and other expression control sequences such as those described herein.
- the packaging signals are a 5’ inverted terminal repeat (ITR) sequence and a 3’ ITR sequence.
- ITR inverted terminal repeat
- the term “transgene” is used interchangeably with “expression cassette”. In other embodiments, the term “transgene” refers solely to the coding sequence for a selected gene, e.g., “hSMNl”.
- nucleic acid molecule that includes an expression cassette comprising: (a) a UbC promoter, (b) a coding sequence for a functional SMN1 protein, (c) at least four miR182 target sequences, and (d) a polyA sequence, optionally with spacer sequences therebetween.
- the expression cassette comprises (a) a 5’ AAV ITR, (b) a UbC promoter, (c) a coding sequence for a functional SMN1 protein, (d) at least four miR182 target sequences, (e) a polyA sequence, and (f) a 3’ AAV ITR, optionally with spacer sequences therebetween.
- the one or more of the UbC promoter, SMN1 coding sequence, miR182 target sequences, and polyA sequence are operably linked.
- the expression cassette includes additional regulatory elements (e.g., an intron sequence).
- SMSN1 or “hSMNl” includes any isoform of SMN1 that restores a desired function, reduces a symptom, or provides another desired physiologic result, when delivered a composition or method provided herein.
- the examples provided herein utilize the longest isoform, Isoform D, which is thought to be the predominant transcript produced by the gene is a patient unaffected by an SMN deficiency or defect.
- Isoform D provides a 294 amino acid protein [see, e.g., NCBI accession NM_000334/NP_000335; ENSEMBL ID ENST00000380707], the protein sequence is reproduced in SEQ ID NO: 5 and the coding sequence is reproduced in SEQ ID NO: 6.
- another isoform may be selected.
- Isoform B has an alternate inframe exon in the 3’ coding sequence, resulting in a protein which is shorter in length (262 amino acids) than Isoform D, but having the same N- and C-termini as that isoform. See, NCBI Accession No. NM_022874/NP_075012; ENSEMBL ID ENST00000503079.
- Isoform A lacks the penultimate exon, which results in an alternate translation stop codon as compared to Isoform D. Thus, Isoform A is shorter (282 amino acids) and has a distinct C- terminus compared to Isoform D. See, NCBI Accession No.
- NM_001297715/NP_001284644 ENSEMBL ID ENSTL00000506163.
- a “vector” as used herein is a biological or chemical moiety comprising a nucleic acid sequence which can be introduced into an appropriate target cell for replication or expression of said nucleic acid sequence.
- a vector include but not limited to a recombinant virus, a plasmid, Lipoplexes, a Polymersome, Polyplexes, a dendrimer, a cell penetrating peptide (CPP) conjugate, a magnetic particle, or a nanoparticle.
- a vector is a nucleic acid molecule into which an engineered nucleic acid encoding a functional hSMNl may be inserted, which can then be introduced into an appropriate target cell.
- Such vectors preferably have one or more origin of replication, and one or more site into which the recombinant DNA can be inserted.
- Vectors often have means by which cells with vectors can be selected from those without, e.g., they encode drug resistance genes.
- Common vectors include plasmids, viral genomes, and “artificial chromosomes”. Conventional methods of generation, production, characterization or quantification of the vectors are available to one of skill in the art.
- the vector is a nucleic acid comprising an expression cassette described herein (for example, “naked DNA”, “naked plasmid DNA”, RNA, and mRNA), which may be coupled with various compositions and nano particles, including, for examples, micelles, liposomes, cationic lipid - nucleic acid compositions, poly-glycan compositions and other polymers, lipid and/or cholesterol-based - nucleic acid conjugates) and other constructs such as are described herein. See, e.g., X. Su et al, Mol. Pharmaceutics, 2011, 8 (3), pp 774- 787; web publication: March 21, 2011; WO2013/182683, WO 2010/053572 and WO 2012/170930, all of which are incorporated herein by reference.
- an expression cassette described herein for example, “naked DNA”, “naked plasmid DNA”, RNA, and mRNA
- compositions and nano particles including, for examples, micelles, liposomes, cationic
- the vector described herein is a “replication-defective virus” or a “viral vector” which refers to a synthetic or artificial viral particle in which an expression cassette containing a nucleic acid sequence encoding hSMNl is packaged in a viral capsid or envelope, where any viral genomic sequences also packaged within the viral capsid or envelope are replication-deficient; i.e., they cannot generate progeny virions but retain the ability to infect target cells.
- a recombinant virus vector is an adeno-associated virus (AAV), an adenovirus, a bocavirus, a hybrid AAV/bocavirus, a herpes simplex virus, or a lentivirus.
- AAV adeno-associated virus
- adenovirus an adenovirus
- a bocavirus a bocavirus
- a hybrid AAV/bocavirus a hybrid AAV/bocavirus
- a herpes simplex virus or a lentivirus.
- a host cell having a nucleic acid including an SMN1 encoding sequence or an expression cassette that includes SMN1 encoding sequence is provided.
- the host cell contains a plasmid having an SMN1 encoding sequence or an expression cassette that includes SMN1 encoding sequence as described herein.
- the term “host cell” may refer to a packaging cell line in which a vector (e.g., a recombinant AAV) is produced.
- a host cell may be a prokaryotic or eukaryotic cell (e.g., human, insect, or yeast) that contains exogenous or heterologous DNA that has been introduced into the cell by any means, e.g., electroporation, calcium phosphate precipitation, microinjection, transformation, viral infection, transfection, liposome delivery, membrane fusion techniques, high velocity DNA-coated pellets, viral infection and protoplast fusion.
- host cells may include, but are not limited to an isolated cell, a cell culture, an Escherichia coli cell, a yeast cell, a human cell, a non-human cell, a mammalian cell, a non-mammalian cell, an insect cell, an HEK-293 cell, a liver cell, a kidney cell, a cell of the central nervous system, a neuron, a glial cell, or a stem cell.
- target cell refers to any cell in which expression of the functional hSMNl is desired.
- the term “target cell” is intended to reference a cell of a subject being treated for SMA that is intended to be transduced by an rAAV vector.
- Target cells include neuronal and non-neuronal cells.
- the target cell may be a central nervous system cell.
- the target cell is one or more of an excitatory neuron, an inhibitory neuron, a glial cell, a cortex cell, a frontal cortex cell, a cerebral cortex cell, a spinal cord cell.
- the target cell is in leptomeninges (LM) of the CNS.
- the target cell is in parenchyma of CNS.
- the target cells include one or more of muscle, liver, lung, airway epithelium, central nervous system, neurons, eye (ocular cells), or heart.
- compositions provided include an engineered human (h) survival of motor neuron (SMN) 1 encoding sequence.
- the native hSMNl encoding is provided in SEQ ID NO: 6, which encodes the amino acid sequence of SEQ ID NO: 5.
- the hSMN 1 coding sequences utilized can be generated in vitro or synthetically, or by any other suitable method using techniques well known in the art.
- the PCR- based accurate synthesis (PAS) of long DNA sequence method may be utilized, as described by Xiong et al, PCR-based accurate synthesis of long DNA sequences, Nature Protocols 1, 791 - 797 (2006).
- a method combining the dual asymmetrical PCR and overlap extension PCR methods is described by Young and Dong, Two-step total gene synthesis method, Nucleic Acids Res. 2004; 32(7): e59. See also, Gordeeva et al, J Microbiol Methods.
- DNA may also be generated from cells transfected with plasmids containing the hOTC sequences described herein. Kits and protocols are known and commercially available and include, without limitation, QIAGEN plasmid kits; Chargeswitch® Pro Filter Plasmid Kits (Invitrogen); and GenEluteTM Plasmid Kits (Sigma Aldrich). Other techniques useful herein include sequence- specific isothermal amplification methods that eliminate the need for thermocycling.
- DNA may also be generated from RNA molecules through amplification via the use of Reverse Transcriptases (RT), which are RNA-dependent DNA Polymerases. RTs polymerize a strand of DNA that is complimentary to the original RNA template and is referred to as cDNA. This cDNA can then be further amplified through PCR or isothermal methods as outlined above. Custom DNA can also be generated commercially from companies including, without limitation, GenScript; GENEWIZ®; GeneArt® (Life Technologies); and Integrated DNA Technologies.
- RT Reverse Transcriptases
- hSMNl is meant a gene which encodes an SMN protein which provides at least about 50%, at least about 75%, at least about 80%, at least about 90%, or about the same, or greater than 100% of the biological activity level of the native survival of motor neuron protein, or a natural variant or polymorph thereof which is not associated with disease. Additionally, SMN1 homologue- SMN2 also encodes the SMN protein, but processes the functional protein less efficiently. Based on the copy number of SMN2, subjects lacking a functional hSMNl gene demonstrate SMA to varying degrees. Thus, for some subjects, it may be desirable for the hSMNl protein to provide less than 100% of the biological activity of the native SMN1 protein. In certain embodiments, the terms “hSMNl”, “functional hSMNl”, and “SMN1” are used interchangeably.
- the standard of care may include nusinersen, which is an SMN2 pre-messenger ribonucleic acid (mRNA)-targeting antisense oligonucleotide (ASO) accepted by the FDA and EMA [SPINRAZATM, Biogen], See, e.g., US Patent Nos.
- nusinersen is an SMN2 pre-messenger ribonucleic acid (mRNA)-targeting antisense oligonucleotide (ASO) accepted by the FDA and EMA [SPINRAZATM, Biogen]
- SMN2- directed antisense oligonucleotide which is administered intrathecally.
- the recommended dosage is 12 mg (5mL per administration).
- Treatment is initiated with 4 loading doses; the first three loading doses being administered at 14-day intervals, the 4 th loading dose administered 30 days after the 3 rd does, and a maintenance dose administered once every 4 months thereafter.
- the amino acid sequence of the functional hSMNl is that of SEQ ID NO: 5.
- the amino acid sequence of the functional hSMNl is SEQ ID NO: 5 or a sequence sharing at least 95%, 96%, 97%, 98%, or 99% identity with SEQ ID NO: 5.
- a modified hSMN coding sequence is provided.
- the modified hSMN coding sequence has less than about 80% identity, preferably about 75% identity or less to the full-length native hSMN coding sequence (SEQ ID NO: 6).
- the modified hSMN coding sequence is characterized by improved translation rate as compared to native hSMN following AAV-mediated delivery.
- the modified hSMN coding sequence shares less than about 80%, 79%, 78%, 77%, 76%, 75%, 74%, 73%, 72%, 71%, 70%, 69%, 68%, 67%, 66%, 65%, 64%, 63%, 62%, or 61% identity to the full length native hSMNl coding sequence.
- the modified hSMNl coding sequence comprises SEQ ID NO: 7, or a sequence sharing at least 70%, 75%, 80%, 85%, 90%, 95% or greater identity with SEQ ID NO: 7.
- the modified hSMNl coding sequence is a sequence sharing at least 70%, 75%, 80%, 85%, 90%, 95% or greater identity with SEQ ID NO: 7 that encodes the amino acid sequence of SEQ ID NO: 5.
- the modified hSMNl coding sequence is a sequence having up to 1, up to 2, up to 3, up to 4, up to 5, up to 6, up to 7, up to 8, up to 9, or up to 10 basepair mismatches when aligned to the nucleotide sequence of SEQ ID NO: 7, and expresses a functional SMN1, In other embodiments, a different SMN1 coding sequence is selected.
- a recombinant nucleic acid molecule that includes an expression cassette, wherein the expression cassette has the nucleic acid sequence of SEQ ID NO: 16, or a nucleic acid sequence at least 99% identical to SEQ ID NO: 16.
- the expression cassette is flanked by a AAV 5’ ITR sequence and a AAV 3’ ITR sequence, optionally with additional sequences between the expression cassette and ITR sequences.
- a recombinant nucleic acid molecule that includes the nucleotide sequence of SEQ ID NO: 2 or a nucleotide sequence at least 99% identical to SEQ ID NO: 2.
- sequence identity refers to the residues in the two sequences which are the same when aligned for correspondence.
- the length of sequence identity comparison may be over the full-length of the genome, the full-length of a gene coding sequence, or a fragment of at least about 500 to 5000 nucleotides, is desired. However, identity among smaller fragments, e.g. of at least about nine nucleotides, usually at least about 20 to 24 nucleotides, at least about 28 to 32 nucleotides, at least about 36 or more nucleotides, may also be desired.
- Percent identity may be readily determined for amino acid sequences over the full- length of a protein, polypeptide, about 32 amino acids, about 330 amino acids, or a peptide fragment thereof or the corresponding nucleic acid sequence coding sequences.
- a suitable amino acid fragment may be at least about 8 amino acids in length, and may be up to about 700 amino acids.
- identity”, “homology”, or “similarity” is determined in reference to “aligned” sequences. “Aligned” sequences or “alignments” refer to multiple nucleic acid sequences or protein (amino acids) sequences, often containing corrections for missing or additional bases or amino acids as compared to a reference sequence.
- Identity may be determined by preparing an alignment of the sequences and through the use of a variety of algorithms and/or computer programs known in the art or commercially available (e.g., BLAST, ExPASy; Clustal Omega; FASTA; using, e.g., Needleman-Wunsch algorithm, Smith -Waterman algorithm). Alignments are performed using any of a variety of publicly or commercially available Multiple Sequence Alignment Programs. Sequence alignment programs are available for amino acid sequences, e.g., the “Clustal Omega”, “Clustal X”, “MUSCLE”, “MAP”, “PIMA”, “MSA”, “BLOCKMAKER”, “MEME”, and “Match-Box” programs.
- any of these programs are used at default settings, although one of skill in the art can alter these settings as needed.
- one of skill in the art can utilize another algorithm or computer program which provides at least the level of identity or alignment as that provided by the referenced algorithms and programs. See, e.g., J. D. Thompson et al, Nucl. Acids. Res., “A comprehensive comparison of multiple sequence alignments”, 27(13):2682-2690 (1999).
- nucleic acid sequences are also available for nucleic acid sequences. Examples of such programs include, “Clustal Omega”, “Clustal W”, “MUSCLE”, “CAP Sequence Assembly”, “BLAST”, “MAP”, and “MEME”, which are accessible through Web Servers on the internet. Other sources for such programs are known to those of skill in the art. Alternatively, Vector NTI utilities are also used. There are also a number of algorithms known in the art that can be used to measure nucleotide sequence identity, including those contained in the programs described above. As another example, polynucleotide sequences can be compared using FastaTM, a program in GCG Version 10.1. FastaTM provides alignments and percent sequence identity of the regions of the best overlap between the query and search sequences. For instance, percent sequence identity between nucleic acid sequences can be determined using FastaTM with its default parameters (a word size of 6 and the NOPAM factor for the scoring matrix) as provided in GCG Version 10.1, herein incorporated by reference.
- FastaTM provides alignments and percent
- operably linked sequences include both regulatory sequences that are contiguous or non-contiguous with the nucleic acid sequence and regulatory sequences that act in cis or trans with nucleic acid sequence.
- regulatory sequences typically include, e.g., one or more of a promoter, an enhancer, an intron, a Kozak sequence, a polyadenylation sequence, and a TATA signal.
- the expression cassette may contain regulatory sequences upstream (5’ to) of the gene sequence, e.g., one or more of a promoter, an enhancer, an intron, etc., and one or more of an enhancer, or regulatory sequences downstream (3’ to) a gene sequence, e.g., 3’ untranslated region (3’ UTR) comprising a polyadenylation site, among other elements.
- the regulatory sequences are operably linked to the nucleic acid sequence of a gene product, wherein the regulatory sequences are separated from the nucleic acid sequence encoding the gene product by intervening nucleic acid sequences, i.e., 5 ’-untranslated regions (5’ UTR).
- the expression cassette comprises nucleic acid sequence of one or more of gene products.
- the expression cassette can be a monocistronic or a bicistronic expression cassette.
- the term “transgene” refers to one or more DNA sequences from an exogenous source which are inserted into a target cell.
- such an expression cassette for generating a viral vector contains the coding sequence for the gene product described herein flanked by packaging signals of the viral genome and other expression control sequences such as those described herein.
- a vector genome may contain two or more expression cassettes.
- exogenous as used to describe a nucleic acid sequence or protein means that the nucleic acid or protein does not naturally occur in the position in which it exists in a chromosome, or host cell.
- An exogenous nucleic acid sequence also refers to a sequence derived from and inserted into the same host cell or subject, but which is present in a non- natural state, e.g., a different copy number, or under the control of different regulatory elements.
- heterologous as used to describe a nucleic acid sequence or protein means that the nucleic acid or protein was derived from a different organism or a different species of the same organism than the host cell or subject in which it is expressed.
- heterologous when used with reference to a protein or a nucleic acid in a plasmid, expression cassette, or vector, indicates that the protein or the nucleic acid is present with another sequence or subsequence which with which the protein or nucleic acid in question is not found in the same relationship to each other in nature.
- encoded amino acid sequence refers to the amino acid which is predicted based on the translation of a known DNA codon of a referenced nucleic acid sequence being translated to an amino acid.
- the following table illustrates DNA codons and twenty common amino acids, showing both the single letter code (SLC) and three letter code (3LC).
- the promoter is a chicken P-actin (also referred to as chicken beta-actin, CB or CBA) promoter.
- the promoter is a CB7 promoter (also referred to as hybrid CB7) comprising a cytomegalovirus immediate-early (CMV IE) enhancer and the chicken P-actin promoter, optionally with spacer sequence, optionally with a chimeric intron comprising chicken beta actin intron and further comprising a chicken betaactin splicing donor (including the exon sequence, chicken beta actin intron) and rabbit betaglobin splicing acceptor.
- CMV IE cytomegalovirus immediate-early
- CMV cytomegalovirus
- GenBank # K03104.1 Chicken beta-actin promoter (281 bp; CB; GenBank # X00182.1).
- the promoter is a ubiquitin C (UbC) promoter.
- the UbC promoter is a human UbC promoter. See, e.g., WO 2001/091800. See, e.g., GenBank accession numbers AF232305 (rat), D63791 (human), NCBI Reference Sequence: NG_027722.2, and Schorpp et al. Nucleic Acids Res. 1996 May 1 ;24(9): 1787-8.
- the UbC promoter comprises the nucleotide sequence of SEQ ID NO: 10.
- the UbC promoter comprises the nucleotide sequence of SEQ ID NO: 17.
- the UbC promoter comprises a nucleotide sequence having up to 1, up to 2, up to 3, up to 4, up to 5, up to 6, up to 7, up to 8, up to 9, or up to 10 basepair mismatches when aligned to the nucleotide sequence of either or both of SEQ ID NOs: 10 and 17, and, when operably linked to a transgene (i.e., SMN1), is capable of expressing the transgene in a target cell.
- a transgene i.e., SMN1
- a suitable promoter may include without limitation, an elongation factor 1 alpha (EFl alpha) promoter (see, e.g., Kim DW et al, Use of the human elongation factor 1 alpha promoter as a versatile and efficient expression system. Gene. 1990 Jul 16;91(2):217- 23), a human Synapsin 1 (hSyn) promoter (see, e.g., Kugler S et al, Human synapsin 1 gene promoter confers highly neuron-specific long-term transgene expression from an adenoviral vector in the adult rat brain depending on the transduced area. Gene Ther.
- EFl alpha elongation factor 1 alpha
- hSyn human Synapsin 1
- the promoter is a JeT promoter (Tornoe J et al. Gene. 2002 Sep 4;297(l-2):21-32). In still other embodiments, multiple enhancers and/or promoters may be included.
- an additional or alternative promoter sequence may be included as part of the expression control sequences (regulatory sequences).
- Constitutive promoters, regulatable promoters [see, e.g., WO 2011/126808 and WO 2013/04943], tissue specific promoters, or a promoter responsive to physiologic cues may be utilized in the vectors described herein.
- the promoter(s) can be selected from different sources, e.g., human cytomegalovirus immediate-early (CMV IE) enhancer/promoter, the SV40 early enhancer/promoter, the JC polymovirus promoter, myelin basic protein (MBP) or glial fibrillary acidic protein (GFAP) promoters, herpes simplex virus (HSV-1) latency associated promoter (LAP), rouse sarcoma virus (RSV) long terminal repeat (LTR) promoter, neuronspecific promoter (NSE), platelet derived growth factor (PDGF) promoter, melaninconcentrating hormone (MCH) promoter, CBA, or matrix metalloprotein promoter (MPP), and the chicken beta-actin promoter.
- CMV IE human cytomegalovirus immediate-early
- MBP myelin basic protein
- GFAP glial fibrillary acidic protein
- HSV-1 herpes simplex virus
- LAP rouse
- the expression cassette is designed for expression of the SMN1 in the central nervous system (CNS), including the cerebral spinal fluid and brain.
- the expression cassette is useful for expression in both the CNS and systemically.
- a vector may contain one or more other appropriate transcription initiation sequences, transcription termination sequences, enhancer sequences, efficient RNA processing signals such as splicing and polyadenylation (poly A) signals; sequences that stabilize cytoplasmic mRNA for example WPRE; sequences that enhance translation efficiency (i.e., Kozak consensus sequence); sequences that enhance protein stability; and when desired, sequences that enhance secretion of the encoded product.
- the regulatory sequences comprise an enhancer. In certain embodiments, the regulatory sequences comprise one enhancer. In another embodiment, the regulatory sequences contain two or more enhancers. These enhancers may be the same or may be different. For example, an enhancer may include an alpha mic/bik enhancer or a CMV IE enhancer. The enhancer may be present in two copies which are located adjacent to one another in an expression cassette. Alternatively, the dual copies of the enhancer may be separated by one or more sequences.
- the regulatory sequences comprise an intron.
- the intron is a chicken beta-actin intron.
- the intron is 875 bp (GenBank # X00182.1).
- the intron is 973 bp intron from the chicken beta actin gene (GenBank # X00182.1).
- the intron is a chimeric intron (CI) - a hybrid intron consisting of a human beta-globin splice donor and immunoglobulin G (IgG) splice acceptor elements.
- the intron is a chimeric intron comprising a chicken beta-actin splicing donor (including the exon sequence), chicken beta actin intron, and rabbit beta globin splicing acceptor.
- the intron includes the nucleic acid sequence of SEQ ID NO: 11.
- the intron comprises a nucleotide sequence having up to 1, up to 2, up to 3, up to 4, up to 5, up to 6, up to 7, up to 8, up to 9, or up to 10 basepair mismatches when aligned to the nucleotide sequence of SEQ ID NO: 11.
- Other suitable introns include those known in the art may by a human P-globulin intron, and/or a commercially available intron, and those described in WO 2011/126808.
- the regulatory sequences comprise a polyadenylation signal (poly A).
- suitable polyA sequences include, e.g., rabbit beta globin (RBG or rBG) poly A, SV40, SV50, bovine growth hormone (bGH), human growth hormone, and synthetic poly As.
- the polyA is a rabbit beta globin poly A (rabbit globin polyA or rBG). See, e.g., WO 2014/151341.
- the rBG polyA comprises SEQ ID NO: 13.
- a human growth hormone (hGH) polyadenylation sequence, an SV40 polyA, or a synthetic polyA may be included in an expression cassette.
- the SV40 polyA is selected.
- the expression cassette includes one or more miRNA (also referred to as miR or micro-RNA) target sequences in the untranslated region(s).
- the miRNA target sequences are designed to be specifically recognized by miRNA present in cells in which transgene expression is undesirable and/or reduced levels of transgene expression are desired.
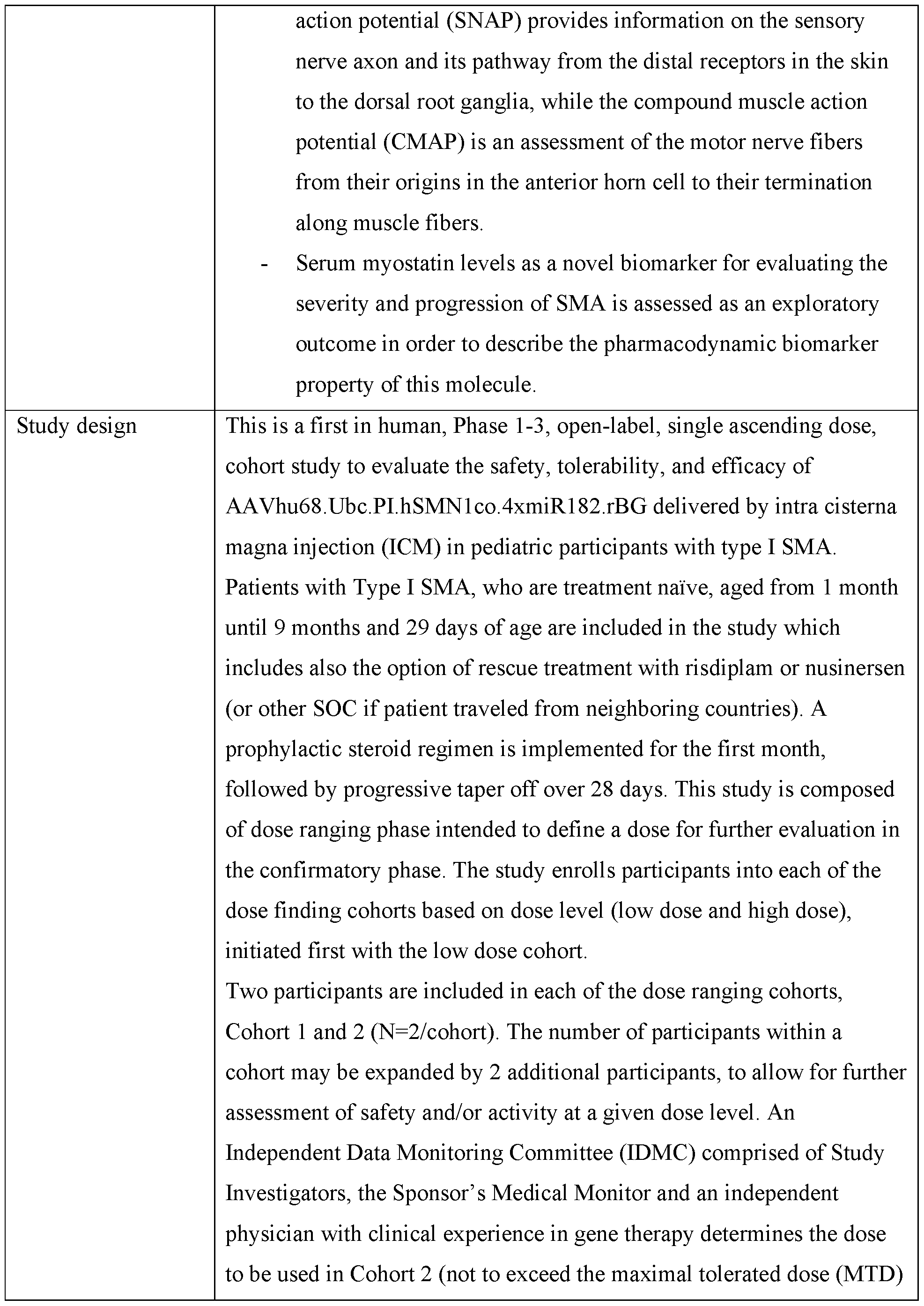
- the expression cassette includes miRNA target sequences that specifically reduce expression of SMN1 in dorsal root ganglion.
- the miRNA target sequences are located in the 3’ UTR, 5’ UTR, and/or in both 3’ and 5’ UTR of an expression cassette.
- the expression cassette comprises at least two, at least three, or at least four tandem repeats of dorsal root ganglion (DRG)-specific miRNA target sequences, wherein the tandem repeats comprise miRNA target sequences that may be the same or different.
- the start of the first of the DRG- specific miRNA tandem repeats is within 20 nucleotides from the 3’ end of the SMN1 encoding sequence.
- the start of the first of DRG-specific miRNA tandem repeats is at least 100 nucleotides from the 3’ end of the SMN1 encoding sequence.
- the miRNA tandem repeats comprise 200 to 1200 nucleotides in length.
- the inclusion of miR targets does not modify the expression or efficacy of the therapeutic transgene in one or more target tissues, relative to the expression cassette lacking the miR target sequences.
- the expression cassette contains at least one, two, three, or four miRNA target sequences that are miR-182 target sequences. In certain embodiments, the expression cassette contains miR-182 target sequences that include the nucleotide sequence AGTGTGAGTTCTACCATTGCCAAA (SEQ ID NO: 12). In certain embodiments, the expression cassette contains more than one copy (e.g., two, three, or four copies) of a sequence that is 100% complementary to the miR-182 seed sequence. In certain embodiments, a miR-182 target sequence is about 7 nucleotides to about 28 nucleotides in length and includes at least one region that is at least 100% complementary to the miR-182 seed sequence.
- a miR-182 target sequence contains a sequence with partial complementarity to SEQ ID NO: 12 and, thus, when aligned to SEQ ID NO: 12, there are one or more basepair mismatches.
- a miR-182 target sequence comprises a sequence having at least 1, 2, 3, 4, 5, 6, 7, 8, 9, or 10 basepair mismatches when aligned to SEQ ID NO: 12, where the basepair mismatches may be non-contiguous.
- a miR-182 target sequence includes a region of 100% complementarity which also comprises at least 30% of the length of the miR-182 target sequence.
- the region of 100% complementarity includes a sequence with 100% complementarity to the miR-182 seed sequence.
- the remainder of a miR-182 target sequence has at least about 80% to about 99% complementarity to miR-182.
- the expression cassette includes a miR-182 target sequence that comprises a truncated SEQ ID NO: 12, i.e., a sequence that lacks at least 1, 2, 3, 4, 5, 6, 7, 8, 9, or 10 nucleotides at either or both the 5’ or 3’ ends of SEQ ID NO: 12.
- tandem repeats is used herein to refer to the presence of two, three, four, or more consecutive miRNA target sequences. These miRNA target sequences may be continuous, i.e., located directly after one another such that the 3’ end of one is directly upstream of the 5’ end of the next with no intervening sequences, or vice versa. In another embodiments, two, three, four, or more of the miRNA target sequences are separated from each other by spacer sequence.
- the expression cassette includes one or more mirl82 target sequence located in a sequence that encodes a 5’ UTR and one or more mirl82 target sequence located in a sequence that encodes a 3’ UTR.
- the expression cassette provided includes at least four miR182 target sequences that are located 3’ of the SMN1 coding sequence and 5’ of the polyA sequence. In other embodiments, the expression cassette includes at least four miR182 target sequences, wherein at least one target sequence is located 5’ of the UbC promoter and at least one target sequence is located 3’ of the SMN1 coding sequence and 5’ of the polyA sequence.
- spacer is any selected nucleic acid sequence, e.g., of 1, 2, 3, 4, 5, 6, 7, 8, 9, or 10 nucleotides in length that is located between two or more consecutive miRNA target sequences.
- the spacer is 1 to 8 nucleotides in length, 2 to 7 nucleotides in length, 3 to 6 nucleotides in length, 4 nucleotides in length, 4 to 9 nucleotides, 3 to 7 nucleotides, or values which are longer.
- a spacer is a non-coding sequence.
- the spacer may be of 4 nucleotides.
- the spacer is GGAT.
- the spacer is six 6 nucleotides. In certain embodiments, the spacer is CACGTG or GCATGC. In certain embodiments, the tandem repeats contain two, three, four or more of the same miRNA target sequence. In certain embodiments, the tandem repeats contain at least two different miRNA target sequences, at least three different miRNA target sequences, or at least four different miRNA target sequences, etc. In certain embodiments, the tandem repeats may contain two or three of the same miRNA target sequence and a fourth miRNA target sequence which is different.
- a 3’ UTR may contain a tandem repeat immediately downstream of the transgene, UTR sequences, and two or more tandem repeats closer to the 3’ end of the UTR.
- the 5’ UTR may contain one, two or more miRNA target sequences.
- the 3’ UTR may contain tandem repeats and the 5’ UTR may contain at least one miRNA target sequence.
- the expression cassette contains two, three, four, or more tandem repeats that start within about 0 to 20 nucleotides of the stop codon for the SMN1 transgene. In other embodiments, the expression cassette contains the miRNA tandem repeats at least 100 to about 4000 nucleotides from the stop codon for the SMN1 transgene.
- rAAV Recombinant Adeno-Associated Virus
- rAAV recombinant adeno-associated virus
- rAAV vector having a capsid that contains an expression cassette that includes a nucleotide sequence encoding hSMNl.
- the rAAV vectors are useful for treating SMN1.
- the AAV capsid selected targets the cells to be treated.
- rAAV.hSMNl refers to an rAAV having a vector genome that includes an hSMNl coding sequence.
- a “rAAVhu68. hSMNl” refers to an rAAV having an AAVhu68 capsid and a vector genome that includes an hSMNl coding sequence.
- the vector genome comprises an AAV 5’ inverted terminal repeat (ITR) sequence, an expression cassette comprising an SMN1 coding sequence that is operably linked to regulatory sequences, and an AAV 3 ’ ITR.
- ITR inverted terminal repeat
- the term “vector genome” refers to a nucleic acid molecule which is packaged in a viral capsid, for example, an AAV capsid, and is capable of being delivered to a host cell or a cell in a patient.
- the vector genome comprises terminal repeat sequences (e.g., AAV inverted terminal repeat sequences (ITRs) necessary for packaging the vector genome into the capsid at the extreme 5’ and 3’ end and containing therebetween an expression cassette that includes an SMN1 encoding sequence as described herein operably linked to sequences which direct expression thereof.
- terminal repeat sequences e.g., AAV inverted terminal repeat sequences (ITRs) necessary for packaging the vector genome into the capsid at the extreme 5’ and 3’ end and containing therebetween an expression cassette that includes an SMN1 encoding sequence as described herein operably linked to sequences which direct expression thereof.
- AAV sequences present in a vector genome typically comprise the cis-acting 5’ and 3’ inverted terminal repeat (ITR) sequences (See, e.g., B. J. Carter, in “Handbook of Parvoviruses”, ed., P. Tijsser, CRC Press, pp. 155 168 (1990)).
- the ITR sequences are about 145 base pairs (bp) in length.
- substantially the entire sequences encoding the ITRs are used in the molecule, although some degree of minor modification of these sequences is permissible. The ability to modify these ITR sequences is within the skill of the art. (See, e.g., texts such as Sambrook et al, “Molecular Cloning.
- An example of such a molecule employed in the present invention is a “cis-acting” plasmid containing the transgene, in which the selected transgene sequence and associated regulatory elements are flanked by the 5’ and 3’ AAV ITR sequences.
- the ITRs are from an AAV different than that supplying a capsid.
- the ITR sequences are from AAV2.
- a shortened version of the 5’ ITR, termed AITR has been described in which the D-sequence and terminal resolution site (trs) are deleted.
- the vector genome (e.g., of a plasmid) includes a shortened AAV2 ITR of 130 base pairs, wherein the external A elements is deleted.
- the shortened ITR may revert back to the wild-type length of 145 base pairs during vector DNA amplification using the internal A element as a template and packaging into the capsid to form the viral particle.
- the full-length AAV 5’ and 3’ ITRs are used.
- ITRs from other AAV sources may be selected. Where the source of the ITRs is from AAV2 and the AAV capsid is from another AAV source, the resulting vector may be termed pseudotyped.
- other configurations of these elements may be suitable.
- AAV adeno-associated virus
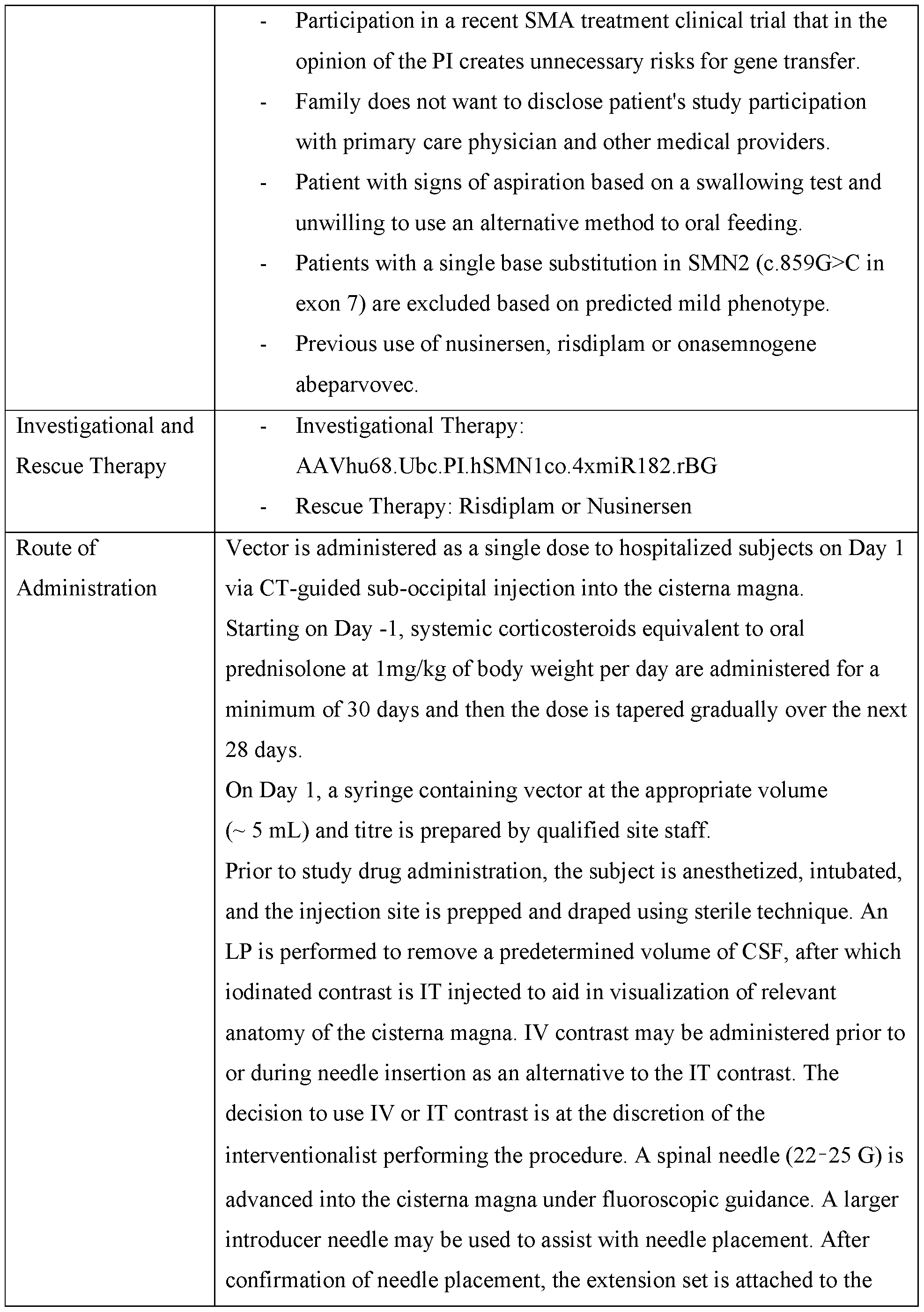
- An adeno-associated virus (AAV) viral vector is an AAV DNase-resistant particle having an AAV protein capsid into which is packaged expression cassette flanked by AAV inverted terminal repeat sequences (ITRs) for delivery to target cells.
- ITRs inverted terminal repeat sequences
- An AAV capsid is composed of 60 capsid (cap) protein subunits, VP1, VP2, and VP3, that are arranged in an icosahedral symmetry in a ratio of approximately 1 : 1 : 10 to 1 : 1 :20, depending upon the selected AAV.
- Various AAVs may be selected as sources for capsids of AAV viral vectors as identified above. See, e.g., US Published Patent Application No. 2007-0036760-Al; US Published Patent Application No. 2009-0197338-Al; EP 1310571.
- the AAV capsid, ITRs, and other selected AAV components described herein may be readily selected from among any AAV, including, without limitation, the AAVs commonly identified as AAV1, AAV2, AAV3, AAV4, AAV5, AAV6, AAV7, AAV8, AAV9, AAV8bp, AAV7M8 and AAVAnc80, AAVhu68, and variants of any of the known or mentioned AAVs or AAVs yet to be discovered or variants or mixtures thereof.
- An AAV9 capsid includes an rAAV having capsid proteins comprising an amino acid sequence which is 99% identical to AAS99264. See, also US7906111 and WO 2005/033321.
- capsids are provided in WO 2022/119871 and WO 2022/226263, which are incorporated herein by reference. See also PCT/US 19/19804 and PCT/US 19/19861, each entitled “Novel Adeno- Associated Virus (AAV) Vectors, AAV Vectors Having Reduced Capsid Deamidation and Uses Therefor” and filed Feb 27, 2019, PCT/US2024/036237, filed June 28, 2024, entitled “Mutant AAV with Central Nervous System Targeting Motifs and Compositions Containing Same,” and US 2024-0024507 Al, “Novel Compositions with Tissue-Specific Targeting Motifs And Compositions Containing Same,” which are incorporated by reference herein in their entireties.
- the capsid protein is designated by a number or a combination of numbers and letters following the term “AAV” in the name of the rAAV vector.
- a recombinant AAV having a vector genome that includes an expression cassette comprising: (a) a UbC promoter, (b) a coding sequence for a functional SMN1 protein, (c) at least four miR182 target sequences, and (d) a polyA sequence, optionally with spacer sequences therebetween.
- the coding sequence, the at least four miR182 target sequences, and the polyA sequence are operably linked to the UbC promoter.
- the expression includes additional regulatory elements (e.g., an intron).
- the polyA sequence is a rabbit beta-globin polyA sequence.
- the vector genome includes an expression cassette having the nucleic acid sequence of SEQ ID NO: 16, or a nucleic acid sequence at least 99% identical to SEQ ID NO: 16. In certain embodiments, the vector genome has the nucleotide sequence of SEQ ID NO: 2 or a nucleotide sequence at least 99% identical to SEQ ID NO: 2.
- the AAV capsid is a Clade F capsid, such as AAV9 capsid, AAVhu68 capsid, hu31 capsid, hu32 capsid, or a variation thereof. See, e.g., WO 2005/033321 published April 14, 2015, WO 2018/160582, and US 2015/0079038, each of which is incorporated herein by reference in its entirety.
- the AAV capsid is a non-clade F capsid, for example a Clade A, B, C, D, or E capsid.
- the non-Clade F capsid is an AAV1 or a variation thereof.
- the AAV capsid transduces a target cell other than the nervous system cells.
- the AAV capsid is a Clade A capsid (e.g., AAV1, AAV6, AAVrh91), a Clade B capsid (e.g., AAV 2), a Clade C capsid (e.g., hu53), a Clade D capsid (e.g., AAV7), or a Clade E capsid (e.g., rhlO).
- the Clade F AAV capsid is an AAVhu68 capsid [See, e.g., US2020/0056159; PCT/US21/55436; SEQ ID NO: 8 for nucleic acid sequence; SEQ ID NO: 9 for amino acid sequence], an AAVhu95 capsid [See, e.g., US Provisional Application No. 63/251,599, filed October 2, 2201, International Patent Application No. PCT/US2022/077315, filed September 30, 2022]; an AAVhu96 capsid [See, e.g., US Provisional Application No. 63/251,599, filed October 2, 2201, and International Patent Application No.
- the AAV capsid is a Clade A capsid, such as AAVrh91 capsid. See, PCT/US20/030266, filed April 29, 2020, now published WO2020/223231, and International Application No. PCT/US21/45945, filed August 13, 2021, which are incorporated herein by reference.
- the AAV capsid for the compositions and methods described herein is chosen based on the target cell.
- the AAV capsid transduces a CNS cell and/or a PNS cell.
- another AAV capsid may be chosen, the AAV capsid is selected from a cy02 capsid, a rh43 capsid, an AAV8 capsid, a rhOl capsid, an AAV9 capsid, an rh8 capsid, a rhlO capsid, a bbOl capsid, a hu37 capsid, a rh02 capsid, a rh20 capsid, a rh39 capsid, a rh64 capsid, an AAV6 capsid, an AAV1 capsid, a hu44 capsid, a hu48 capsid, a cy05 capsid a
- An AAV capsid is an assembly of a heterogeneous population of vpl, a heterogeneous population of vp2, and a heterogeneous population of vp3 proteins.
- the term “heterogeneous” or any grammatical variation thereof refers to a population consisting of elements that are not the same, for example, having vpl, vp2 or vp3 monomers (proteins) with different modified amino acid sequences.
- heterogeneous refers to a population consisting of elements that are not the same, for example, having vpl, vp2, or vp3 (also referred to as VP1, VP2, VP3, or Vpl, Vp2, Vp3) monomers (proteins) with different modified amino acid sequences.
- vpl, vp2 and vp3 proteins also referred to as VP1, VP2, VP3, or Vpl, Vp2, Vp3 monomers (proteins) with different modified amino acid sequences.
- heterogeneous population as used in connection with vpl, vp2 and vp3 proteins (alternatively termed isoforms), refers to differences in the amino acid sequence of the vpl, vp2 and vp3 proteins within a capsid.
- the AAV capsid contains subpopulations within the vpl proteins, within the vp2 proteins and within the vp3 proteins which have modifications from the predicted amino acid residues. These subpopulations include, at a minimum, certain deamidated asparagine (N or Asn) residues.
- certain subpopulations comprise at least one, two, three or four highly deamidated asparagines (N) positions in asparagine - glycine pairs and optionally further comprising other deamidated amino acids, wherein the deamidation results in an amino acid change and other optional modifications.
- AAV capsids are provided which have a heterogeneous population of AAV capsid isoforms (i.e., VP1, VP2, VP3) which contain multiple highly deamidated “NG” positions.
- the highly deamidated positions are in the locations identified below, with reference to the predicted full-length VP1 amino acid sequence.
- the capsid gene is modified such that the referenced “NG” is ablated and a mutant “NG” is engineered into another position.
- the rAAV provided herein is has an AAVhu68 capsid that includes AAVhu68 VP1 proteins, AAVhu68 VP2 proteins, and AAVhu68 VP3 proteins expressed from a nucleic acid sequence encoding SEQ ID NO: 9 (amino acids 1 to 736).
- the AAV hu68 capsid comprises a heterogenous population of AAVhu68 vpl proteins, a heterogenous population of AAVhu68 vp2 proteins; and a heterogenous population of AAVhu68 vp3 proteins comprising amino acid residues 1 to 736 (vpl), amino acid residues 138 to 736 (vp2), and amino acid residues 203 to 736 (vp3) of SEQ ID NO: 9, respectively, with amino acid modifications, wherein the heterogenous AAVhu68 vpl proteins, AAVhu68 vp2 proteins and AAVhu68 vp3 proteins contain amino acid modifications comprising 50% to 100% deamidation in at least two asparagines (N) in asparagine - glycine pairs in SEQ ID NO: 9 in two or more of asparagines (N) at positions N57, N329, N452, and/or N512 based on the numbering of the encoded AAVhu68 VP1
- the subpopulations of AAVhu68 VP1 proteins, AAVhu68 VP2 proteins and AAVhu68VP3 proteins further comprise one or more of: (a) at least 65% of asparagines (N) in position N57 of the vpl proteins are deamidated, based on the numbering of SEQ ID NO: 9; (b) at least 75% of N in position N329 of the vpl, v2 and vp3 proteins are deamidated, based on the residue numbering of the amino acid sequence of SEQ ID NO: 9, (c) at least 50% of N in position N452 of the vpl, v2 and vp3 proteins are deamidated, based on the residue numbering of the amino acid sequence of SEQ ID NO: 9; and/or (d) at least 75% of N in N512 of the vpl, v2 and vp3 proteins are deamidated, based on the residue numbering of the amino acid sequence of SEQ ID NO: 9, where
- the rAAVhu68 capsid comprises a subpopulation of vpl in which 75% to 100 % of the N at position 57 of the vpl proteins, based on the numbering of SEQ ID NO: 9, are deamidated as determined using mass spectrometry.
- the rAAVhu68 capsid comprises subpopulation of vpl proteins, vp2 proteins, and/or vp3 proteins in which 75% to 100% of the N at position 329, based on the numbering of SEQ ID NO: 9, are deamidated as determined using mass spectrometry.
- the rAAVhu68 capsid comprises subpopulation of vpl proteins, vp2 proteins, and/or vp3 proteins in which 75% to 100% of the N at position 452, based on the numbering of SEQ ID NO: 9, are deamidated as determined using mass spectrometry. In certain embodiments, the rAAVhu68 capsid comprises subpopulation of vpl proteins, vp2 proteins, and/or vp3 proteins in which 75% to 100% of the N at position 512, based on the numbering of SEQ ID NO: 9, are deamidated.
- the nucleic acid sequence encoding the proteins is SEQ ID NO: 9, or a sequence at least 80% to at least 99% identical to SEQ ID NO: 8 which encodes the amino acid sequence of SEQ ID NO: 9. In certain embodiments, the nucleic acid sequence is at least 80% to 97% identical to SEQ ID NO: 8.
- the rAAVhu68 capsid comprises subpopulations with 50% to 100% deamidation in each of N57, N329, N452, and N512, and optionally further comprise subpopulations of vpl, vp2 and/or vp3 proteins which further comprise 1% to about 40% deamidation in at least one or more of positions N94, N113, N252, N253, Q258, N270, N303, N304, N305, N319, N328, N336, N409, N410, N477, N515, N598, Q599, N628, N651, N663, N709, or combinations thereof, based on the amino acid numbering of SEQ ID NO: 9.
- the rAAVhu68 capsid vpl, vp2 and/or vp3 proteins further comprise one or more modifications selected from one or more modification in one or more of the following: acetylated lysine, phosphorylated serine and/or threonine, isomerized aspartic acid, oxidized tryptophan and/or methionine, or an amidated amino acid.
- the AAVhu68 capsid comprises: a heterogenous population of AAVhu68 vpl, AAVhu68vp2 and AAVhu68vp3 proteins produced by expression from a nucleic acid molecule having a nucleic acid sequence encoding the amino acid sequence of 1 to 736 of SEQ ID NO: 9, wherein the heterogenous population of AAVhu68 vpl proteins, AAVhu68 vp2 proteins, and AAVhu68 vp3 proteins comprise amino acids 1 to 736 (vpl), amino acids 138 to 736 (vp2), and amino acids 203 to 736 (vp3) of SEQ ID NO: 9, respectively, with amino acid modifications comprising 50% to 100% deamidation in at least two asparagines (N)-glycine pairs in two or more of N57, N329, N452, and/or N512 of SEQ ID NO: 9 as determined using mass spectrometry and optionally further comprising subpopulations comprising other deami
- the VP1 proteins the nucleic acid sequence is SEQ ID NO: 8 or a sequence at least 70% identical to SEQ ID NO: 8 which encodes the amino acid sequence of SEQ ID NO: 9.
- the nucleic acid sequence is SEQ ID NO: 8.
- the rAAVhu68 capsid comprises a subpopulation of AAVhu68 vpl in which 75% to 100 % of the N at position 57 of the vpl proteins are deamidated, as determined using mass spectrometry.
- the rAAVhu68 capsid comprises subpopulation of AAVhu68 vpl proteins, AAVhu68 vp2 proteins, and/or AAVhu68 vp3 proteins in which 75% to 100% of the N at position 329, based on the numbering of SEQ ID NO: 9 are deamidated as determined using mass spectrometry.
- the rAAVhu68 capsid comprises subpopulation of AAVhu68 vpl proteins, AAVhu68 vp2 proteins, and/or AAVhu68 vp3 proteins in which 75% to 100% of the N at position 452, based on the numbering of SEQ ID NO: 9, are deamidated as determined using mass spectrometry.
- the rAAVhu68 capsid comprises subpopulation of AAVhu68 vpl proteins, AAVhu68 vp2 proteins, and/or AAVhu68 vp3 proteins in which 75% to 100% of the N at position 512, based on the numbering of SEQ ID NO: 9, are deamidated.
- the rAAVhu68 capsid comprises subpopulations with 50% to 100% deamidation in each of N57, N329, N452, and N512, and optionally further comprise subpopulations of AAVhu68 vpl, AAVhu68 vp2 and/or AAVhu68 vp3 proteins which further comprise 1% to about 40% deamidation in at least one or more of positions N94, N113, N252, N253, Q258, N270, N303, N304, N305, N319, N328, N336, N409, N410, N477, N515, N598, Q599, N628, N651, N663, N709, or combinations thereof, based on the amino acid numbering of SEQ ID NO: 9.
- the AAVhu68 capsid comprises a heterogenous population of AAVhu68 vpl proteins, AAVhu68 vp2 proteins, and AAVhu68 vp3 proteins which comprise amino acids 1 to 736 (vpl), amino acids 138 to 736 (vp2), and amino acids 203 to 736 (vp3) of SEQ ID NO: 9, respectively, with amino acid modifications comprising 50% to 100% deamidation in at least two asparagines (N) in asparagine - glycine pairs in two or more of N57, N329, N452, and/or N512 of SEQ ID NO: 9 as determined using mass spectrometry and optionally further comprising subpopulations comprising other deamidated amino acids; wherein the AAVhu68 proteins contain amino acid modifications comprising 50% to 100% deamidation in two or more of asparagines (N) in position N57, N329, N452, and/or N512 based on the residue numbering of SEQ ID NO:
- the nucleic acid sequence is SEQ ID NO: 8 or a sequence at least 70% identical to SEQ ID NO: 8 which encodes the amino acid sequence of SEQ ID NO: 9.
- the nucleic acid sequence is SEQ ID NO: 8.
- the rAAVhu68 capsid comprises a subpopulation of AAVhu68 vpl in which 75% to 100 % of the N at position 57 of the vpl proteins are deamidated, as determined using mass spectrometry.
- the rAAVhu68 capsid comprises subpopulation of AAVhu68 vpl proteins, AAVhu68 vp2 proteins, and/or AAVhu68 vp3 proteins in which 75% to 100% of the N at position 329, based on the numbering of SEQ ID NO: 9, are deamidated as determined using mass spectrometry.
- the rAAVhu68 capsid comprises subpopulation of AAVhu68 vpl proteins, AAVhu68 vp2 proteins, and/or AAVhu68 vp3 proteins in which 75% to 100% of the N at position 452, based on the numbering of SEQ ID NO: 9, are deamidated as determined using mass spectrometry.
- the rAAVhu68 capsid comprises subpopulation of AAVhu68 vpl proteins, AAVhu68 vp2 proteins, and/or AAVhu68 vp3 proteins in which 75% to 100% of the N at position 512, based on the numbering of SEQ ID NO: 9, are deamidated as determined using mass spectrometry.
- the rAAVhu68 capsid comprises subpopulations with 50% to 100% deamidation in each of N57, N329, N452, and N512, and optionally further comprise 1% to about 40% deamidation in at least one or more of positions N94, N113, N252, N253, Q258, N270, N303, N304, N305, N319, N328, N336, N409, N410, N477, N515, N598, Q599, N628, N651, N663, N709, or combinations thereof, based on the amino acid numbering of SEQ ID NO: 9.
- the AAVhu68 capsid comprises AAVhu68 vpl proteins, AAVhu68 vp2 proteins, and AAVhu68 vp3 proteins produced from a nucleic acid sequence encoding the amino acid sequence of SEQ ID NO: 9, wherein the AAVhu68vpl proteins comprise a glutamic acid at position 67 and a valine at position 157 and the AAVhu68vp2 proteins comprise a valine at position 157 based on the numbering of SEQ ID NO: 9.
- AAVhu68 capsid comprises subpopulations of AAVhu68 vpl, AAVhu68 vp2 and AAVhu68 vp3 proteins, wherein the subpopulations of the AAVhu68 vpl, AAVhu68 vp2 and AAV hu68 vp3 proteins comprise at least 50% to 100% deamidated asparagines (N) in asparagine - glycine pairs at each of positions 57, 329, 452, 512, relative to the amino acids in SEQ ID NO: 9, wherein the deamidated asparagines are deamidated to aspartic acid, isoaspartic acid, an interconverting aspartic acid/isoaspartic acid pair, or combinations thereof, as determined using mass spectrometry.
- N deamidated asparagines
- the nucleic acid sequence encoding the vpl proteins is SEQ ID NO: 9, or a sequence at least 80% to at least 99% identical to SEQ ID NO: 8 which encodes the amino acid sequence of SEQ ID NO: 9; optionally wherein the nucleic acid sequence is at least 80% to 97% identical to SEQ ID NO: 8.
- AAVhu68 capsid comprises heterogenous populations of AAVhu68 vpl, AAVhu68 vp2 and AAVhu68 vp3 proteins, wherein the AAVhu68 vpl proteins are amino acids 1 to 736 of SEQ ID NO: 9 (vpl) which comprise a glutamic acid at position 67 and a valine at position 157 and further comprise subpopulations of vpl proteins comprising modified amino acids based on the amino acids positions in SEQ ID NO: 9, wherein the AAVhu68 vp2 proteins are amino acids 138 to 736 of SEQ ID NO: 9 (vp2) which comprise a valine at position 157 and further comprise subpopulations of vp2 proteins comprising modified amino acids based on the amino acid positions in SEQ ID NO: 9, and wherein the AAVhu68 vp3 proteins are amino acids 203 to 736 of SEQ ID NO: 9 (vp3), which comprise subpopulations of vp3 proteins
- the subpopulations of AAVhu68 vpl, vp2 and vp3 proteins further comprise: (i) one or more modifications selected from: acetylated lysine, phosphorylated serine and/or threonine, isomerized aspartic acid, deamidated glutamines, oxidated tryptophan and/or methionine, or an amidated amino acid as determined using mass spectrometry; and/or (ii) 1% to 40% deamidation of asparagines at one or more of positions N94, N113, N252, N253, Q259, N270, N303, N304, N305, N319, N328, N336, N409, N410, N477, N515, N598, Q599, N628, N651, N663, N709, or combinations thereof, based on the numbering of SEQ ID NO: 9, as determined using mass spectrometry.
- the AAVhu68 capsid subpopulations of AAVhu68 vpl, vp2 and vp3 capsid proteins further comprise (a) at least 65% of asparagines (N) in asparagine - glycine pairs located at positions 57 of the vpl proteins are deamidated, as determined using mass spectrometry, based on the numbering of SEQ ID NO: 9; and/or (b) at least 75% of N in asparagine - glycine pairs in position 329 of the vpl, vp2 and vp3 proteins are deamidated, as determined using mass spectrometry, based on the residue numbering of the amino acid sequence of SEQ ID NO: 9; and/or (c) at least 50% of N in asparagine - glycine pairs in position 452 of the vpl, vp2 and vp3 proteins are deamidated, as determined using mass spectrometry, based on the residue numbering
- the nucleic acid sequence encoding the vpl proteins is SEQ ID NO: 8, or a sequence at least 80% to at least 99% identical to SEQ ID NO: 8 which encodes the amino acid sequence of SEQ ID NO: 9; optionally wherein the nucleic acid sequence is at least 80% to 97% identical to SEQ ID NO: 8.
- an rAAV production system useful for producing a rAAV as described herein.
- the production system comprises a cell culture comprising (a) a nucleic acid sequence encoding an AAV capsid protein; (b) the vector genome; and (c) sufficient AAV rep functions and helper functions to permit packaging of the vector genome into the AAV capsid.
- the vector genome comprises SEQ ID NO: 2.
- the vector genome comprises SEQ ID NO: 16.
- the cell culture is bacterial cell culture.
- the cell culture is mammalian cell culture.
- the cell culture is a human embryonic kidney 293 (HEK293) cell culture.
- the cell culture is a suspension cell culture.
- the AAV rep is from a different AAV.
- the AAV rep is from AAV2.
- the AAV rep coding sequence and cap genes are on the same nucleic acid molecule, wherein there is optionally a spacer between the rep sequence and cap gene.
- the vector genomes can be carried on any suitable vector, e.g., a plasmid, which is delivered to a packaging host cell.
- a suitable vector e.g., a plasmid
- the plasmids useful in this invention may be engineered such that they are suitable for replication and packaging in vitro in prokaryotic cells, insect cells, mammalian cells, among others. Suitable transfection techniques and packaging host cells are known and/or can be readily designed by one of skill in the art.
- a gene therapy vector refers to a rAAV as described herein, which is suitable for use in treating a patient.
- the ITRs are the only AAV components required in cis in the same construct as the nucleic acid molecule containing the gene.
- the cap and rep genes can be supplied in trans.
- potency e.g., biological activity
- the expression cassettes described herein are engineered into a genetic element (e.g., a shuttle plasmid) which transfers the sequences carried thereon into a packaging host cell for production of a viral vector.
- the selected genetic element may be delivered to an AAV packaging cell by any suitable method, including transfection, electroporation, liposome delivery, membrane fusion techniques, high velocity DNA-coated pellets, viral infection and protoplast fusion.
- Stable AAV packaging cells can also be made. The methods used to make such constructs are known to those with skill in nucleic acid manipulation and include genetic engineering, recombinant engineering, and synthetic techniques. See, e.g., Molecular Cloning: A Laboratory Manual, ed.
- AAV intermediate or “AAV vector intermediate” refers to an assembled rAAV capsid which lacks the desired genomic sequences packaged therein. These may also be termed an “empty” capsid. Such a capsid may contain no detectable genomic sequences of an expression cassette, or only partially packaged genomic sequences which are insufficient to achieve expression of the gene product. These empty capsids are non-functional to transfer the gene of interest to a host cell.
- the recombinant adeno-associated virus (AAV) described herein may be generated using techniques which are known. See, e.g., WO 2003/042397; WO 2005/033321, WO 2006/110689; US 7588772 B2.
- Such a method involves culturing a host cell which contains a nucleic acid sequence encoding an AAV capsid protein; a functional rep gene; an expression cassette composed of, at a minimum, AAV inverted terminal repeats (ITRs) and a transgene; and sufficient helper functions to permit packaging of the expression cassette into the AAV capsid protein.
- ITRs AAV inverted terminal repeats
- a production cell culture useful for producing a recombinant AAV having a capsid selected from AAVhu68, AAVrh91, AAVhu95 or AAVhu96 is provided.
- Such a cell culture contains a nucleic acid which expresses the AAV capsid protein in the host cell; a nucleic acid molecule suitable for packaging into the AAV capsid, e.g., a vector genome that contains AAV ITRs and a non- AAV nucleic acid sequence encoding a transgene (hSMNl) operably linked to regulatory sequences that direct expression of the transgene in a host cell; and sufficient AAV rep functions and adenovirus helper functions to permit packaging of the vector genome into the AAV capsid.
- a nucleic acid which expresses the AAV capsid protein in the host cell e.g., a vector genome that contains AAV ITRs and a non- AAV nucleic acid sequence encoding a transgene
- the cell culture is composed of mammalian cells (e.g., human embryonic kidney 293 cells, among others) or insect cells (e.g., Spodoptera frugiperda (Sf9) cells).
- baculovirus provides the helper functions necessary for packaging the vector genome into the recombinant AAV capsid.
- the rep functions are provided by an AAV other than AAV2, selected to complement the source of the ITRs.
- Methods for manufacturing the gene therapy vectors described herein include methods well known in the art such as generation of plasmid DNA used for production of the gene therapy vectors, generation of the vectors, and purification of the vectors.
- the gene therapy vector is an AAV vector and the plasmids generated are an AAV cis-plasmid encoding the AAV vector genome and the gene of interest, an AAV transplasmid containing AAV rep and cap genes, and an adenovirus helper plasmid.
- the vector generation process can include method steps such as initiation of cell culture, passage of cells, seeding of cells, transfection of cells with the plasmid DNA, post-transfection medium exchange to serum free medium, and the harvest of vector-containing cells and culture media.
- the harvested vector-containing cells and culture media are referred to herein as crude cell harvest.
- the gene therapy vectors are introduced into insect cells by infection with baculovirus-based vectors.
- Zhang et al., 2009 Adenovirus-adeno-associated virus hybrid for large-scale recombinant adeno-associated virus production, Human Gene Therapy 20:922-929, the contents of each of which is incorporated herein by reference in its entirety.
- Methods of making and using these and other AAV production systems are also described in the following US patents, the contents of each of which is incorporated herein by reference in its entirety: US Patent Nos.
- the crude cell harvest may thereafter be subject method steps such as concentration of the vector harvest, diafiltration of the vector harvest, microfluidization of the vector harvest, nuclease digestion of the vector harvest, filtration of microfluidized intermediate, crude purification by chromatography, crude purification by ultracentrifugation, buffer exchange by tangential flow filtration, and/or formulation and filtration to prepare bulk vector.
- An affinity chromatography purification followed anion exchange resin chromatography are used to purify the vector drug product and to remove empty capsids.
- GC genome copies
- the number of particles (pt) per 20 pL loaded is then multiplied by 50 to give particles (pt) /mL.
- Pt/mL divided by GC/mL gives the ratio of particles to genome copies (pt/GC).
- Pt/mL-GC/mL gives empty pt/mL.
- Empty pt/mL divided by pt/mL and x 100 gives the percentage of empty particles.
- the methods include subjecting the treated AAV stock to SDS-polyacrylamide gel electrophoresis, consisting of any gel capable of separating the three capsid proteins, for example, a gradient gel containing 3-8% Tris-acetate in the buffer, then running the gel until sample material is separated, and blotting the gel onto nylon or nitrocellulose membranes, preferably nylon.
- Anti-AAV capsid antibodies are then used as the primary antibodies that bind to denatured capsid proteins, preferably an anti-AAV capsid monoclonal antibody, most preferably the Bl anti-AAV-2 monoclonal antibody (Wobus et al., J. Virol. (2000) 74:9281- 9293).
- a secondary antibody is then used, one that binds to the primary antibody and contains a means for detecting binding with the primary antibody, more preferably an anti- IgG antibody containing a detection molecule covalently bound to it, most preferably a sheep anti-mouse IgG antibody covalently linked to horseradish peroxidase.
- a method for detecting binding is used to semi-quantitatively determine binding between the primary and secondary antibodies, preferably a detection method capable of detecting radioactive isotope emissions, electromagnetic radiation, or colorimetric changes, most preferably a chemiluminescence detection kit.
- a detection method capable of detecting radioactive isotope emissions, electromagnetic radiation, or colorimetric changes, most preferably a chemiluminescence detection kit.
- samples from column fractions can be taken and heated in SDS-PAGE loading buffer containing reducing agent (e.g., DTT), and capsid proteins were resolved on pre-cast gradient polyacrylamide gels (e.g., Novex).
- Silver staining may be performed using SilverXpress (Invitrogen, CA) according to the manufacturer's instructions or other suitable staining method, i.e., SYPRO ruby or coomassie stains.
- the concentration of AAV vector genomes (vg) in column fractions can be measured by quantitative real time PCR (Q-PCR).
- Samples are diluted and digested with DNase I (or another suitable nuclease) to remove exogenous DNA. After inactivation of the nuclease, the samples are further diluted and amplified using primers and a TaqManTM fluorogenic probe specific for the DNA sequence between the primers. The number of cycles required to reach a defined level of fluorescence (threshold cycle, Ct) is measured for each sample on an Applied Biosystems Prism 7700 Sequence Detection System. Plasmid DNA containing identical sequences to that contained in the AAV vector is employed to generate a standard curve in the Q-PCR reaction. The cycle threshold (Ct) values obtained from the samples are used to determine vector genome titer by normalizing it to the Ct value of the plasmid standard curve. End-point assays based on the digital PCR can also be used.
- DNase I or another
- an optimized q-PCR method which utilizes a broadspectrum serine protease, e.g., proteinase K (such as is commercially available from Qiagen). More particularly, the optimized qPCR genome titer assay is similar to a standard assay, except that after the DNase I digestion, samples are diluted with proteinase K buffer and treated with proteinase K followed by heat inactivation. Suitably samples are diluted with proteinase K buffer in an amount equal to the sample size.
- the proteinase K buffer may be concentrated to 2 -old or higher. Typically, proteinase K treatment is about 0.2 mg/mL, but may be varied from 0.1 mg/mL to about 1 mg/mL.
- the treatment step is generally conducted at about 55 °C for about 15 minutes, but may be performed at a lower temperature (e.g., about 37 °C to about 50 °C) over a longer time period (e.g., about 20 minutes to about 30 minutes), or a higher temperature (e.g., up to about 60 °C) for a shorter time period (e.g., about 5 to 10 minutes).
- heat inactivation is generally at about 95 °C for about 15 minutes, but the temperature may be lowered (e.g., about 70 to about 90 °C) and the time extended (e.g., about 20 minutes to about 30 minutes). Samples are then diluted (e.g., 1000- fold) and subjected to TaqMan analysis as described in the standard assay.
- droplet digital PCR may be used.
- ddPCR droplet digital PCR
- methods for determining single-stranded and self-complementary AAV vector genome titers by ddPCR have been described. See, e.g., M. Lock et al, Hu Gene Therapy Methods, Hum Gene Ther Methods. 2014 Apr;25(2): 115-25. doi: 10.1089/hgtb.2013.131. Epub 2014 Feb 14.
- the manufacturing process for rAAV as described herein involves method as described in US Provisional Patent Application No. 63/371,597, filed August 16, 2022, and US Provisional Patent Application No. 63/371,592, filed August 16, 2022, which are incorporated herein by reference in their entirety.
- the method for separating rAAVhu68 (or AAVrh91, AAVhu95 or AAVhu96) particles having packaged genomic sequences from genome-deficient AAVhu68 (or AAVrh91 or AAVhu95 or AAVhu96) intermediates involves subjecting a suspension comprising recombinant AAVhu68 (or AAVrh91) viral particles and AAVhu68 (or AAVrh91 or AVhu95 or AAVhu96) capsid intermediates to fast performance liquid chromatography, wherein the AAVhu68 (or AAVrh91 or AAVhu95 or AAVhu96) viral particles and AAVhu68 intermediates are bound to a strong anion exchange resin equilibrated at a pH of about 10.2 (or about 9.8 for AAVrh91), and subjected to a salt gradient while monitoring eluate for ultraviolet absorbance at about 260 nanometers (nm) and about 280 nm.
- the pH may be in the range of about 10 to 10.4.
- the AAV full capsids are collected from a fraction which is eluted when the ratio of A260/A280 reaches an inflection point.
- the diafiltered product may be applied to an affinity resin (Life Technologies) that efficiently captures the AAV serotype. Under these ionic conditions, a significant percentage of residual cellular DNA and proteins flow through the column, while AAV particles are efficiently captured.
- the term “clade” as it relates to groups of AAV refers to a group of AAV which are phylogenetically related to one another as determined using a Neighbor- Joining algorithm by a bootstrap value of at least 75% (of at least 1000 replicates) and a Poisson correction distance measurement of no more than 0.05, based on alignment of the AAV vpl amino acid sequence.
- the Neighbor- Joining algorithm has been described in the literature. See, e.g., M. Nei and S. Kumar, Molecular Evolution and Phylogenetics (Oxford University Press, New York (2000). Computer programs are available that can be used to implement this algorithm. For example, the MEGA v2.1 program implements the modified Nei-Gojobori method.
- AAV vpl capsid protein Using these techniques and computer programs, and the sequence of an AAV vpl capsid protein, one of skill in the art can readily determine whether a selected AAV is contained in one of the clades identified herein, in another clade, or is outside these clades. See, e.g., G Gao, et al, J Virol, 2004 Jun; 78(10): 6381-6388, which identifies Clades A, B, C, D, E and F, and provides nucleic acid sequences of novel AAV, GenBank Accession Numbers AY530553 to AY530629. See, also, WO 2005/033321.
- sc refers to self-complementary.
- Self-complementary AAV refers a construct in which a coding region carried by a recombinant AAV nucleic acid sequence has been designed to form an intra-molecular double-stranded DNA template.
- dsDNA double stranded DNA
- scAAV Self-complementary recombinant adeno-associated virus
- Self-complementary AAVs are described in, e.g., U.S. Patent Nos. 6,596,535; 7,125,717; and 7,456,683, each of which is incorporated herein by reference in its entirety.
- the rAAV provided herein is a scAAV vector.
- the rAAV is not a scAAV vector and includes a s single-stranded DNA genome.
- a “replication-defective virus” or “viral vector” refers to a synthetic or artificial viral particle in which an expression cassette containing a gene of interest is packaged in a viral capsid or envelope, where any viral genomic sequences also packaged within the viral capsid or envelope are replication-deficient; i.e., they cannot generate progeny virions but retain the ability to infect target cells.
- the genome of the viral vector does not include genes encoding the enzymes required to replicate (the genome can be engineered to be "gutless" - containing only the gene of interest flanked by the signals required for amplification and packaging of the artificial genome), but these genes may be supplied during production. Therefore, it is deemed safe for use in gene therapy since replication and infection by progeny virions cannot occur except in the presence of the viral enzyme required for replication.
- the capsid protein is a non-naturally occurring capsid.
- Such an artificial capsid may be generated by any suitable technique, using a selected AAV sequence (e.g., a fragment of a vpl capsid protein) in combination with heterologous sequences which may be obtained from a different selected AAV, non-contiguous portions of the same AAV, from a non- AAV viral source, or from a non-viral source.
- An artificial AAV may be, without limitation, a pseudotyped AAV, a chimeric AAV capsid, a recombinant AAV capsid, or a “humanized” AAV capsid.
- Pseudotyped vectors, wherein the capsid of one AAV is replaced with a heterologous capsid protein, are useful in the invention.
- AAV2/5 and AAV2/8 are exemplary pseudotyped vectors.
- the selected genetic element may be delivered by any suitable method, including transfection, electroporation, liposome delivery, membrane fusion techniques, high velocity DNA-coated pellets, viral infection and protoplast fusion.
- the methods used to make such constructs are known to those with skill in nucleic acid manipulation and include genetic engineering, recombinant engineering, and synthetic techniques. See, e.g., Green and Sambrook, Molecular Cloning: A Laboratory Manual, Cold Spring Harbor Press, Cold Spring Harbor, NY (2012).
- rAAV particles are referred to as DNase resistant.
- DNase endonuclease
- other endo- and exo- nucleases may also be used in the purification steps described herein, to remove contaminating nucleic acids.
- Such nucleases may be selected to degrade single stranded DNA and/or double-stranded DNA, and RNA.
- Such steps may contain a single nuclease, or mixtures of nucleases directed to different targets, and may be endonucleases or exonucleases.
- nuclease-resistant indicates that the AAV capsid has fully assembled around the expression cassette which is designed to deliver a gene to a host cell and protects these packaged genomic sequences from degradation (digestion) during nuclease incubation steps designed to remove contaminating nucleic acids which may be present from the production process.
- heterogeneous refers to a population consisting of elements that are not the same, for example, having vpl, vp2 or vp3 monomers (proteins) with different modified amino acid sequences.
- heterogeneous refers to differences in the amino acid sequence of the vpl, vp2 and vp3 proteins within a capsid.
- the AAV capsid contains subpopulations within the vpl proteins, within the vp2 proteins and within the vp3 proteins which have modifications from the predicted amino acid residues. These subpopulations include, at a minimum, certain deamidated asparagine (N or Asn) residues.
- certain subpopulations comprise at least one, two, three or four highly deamidated asparagines (N) positions in asparagine - glycine pairs and optionally further comprising other deamidated amino acids, wherein the deamidation results in an amino acid change and other optional modifications.
- N highly deamidated asparagines
- a “subpopulation” of vp proteins refers to a group of vp proteins which has at least one defined characteristic in common and which consists of at least one group member to less than all members of the reference group, unless otherwise specified.
- a “subpopulation” of vpl proteins is at least one (1) vpl protein and less than all vpl proteins in an assembled AAV capsid, unless otherwise specified.
- a “subpopulation” of vp3 proteins may be one (1) vp3 protein to less than all vp3 proteins in an assembled AAV capsid, unless otherwise specified.
- vpl proteins may be a subpopulation of vp proteins; vp2 proteins may be a separate subpopulation of vp proteins, and vp3 are yet a further subpopulation of vp proteins in an assembled AAV capsid.
- vpl, vp2 and vp3 proteins may contain subpopulations having different modifications, e.g., at least one, two, three or four highly deamidated asparagines, e.g., at asparagine - glycine pairs.
- nucleic acid can be RNA, DNA, or a modification thereof, and can be single or double stranded, and can be selected, for example, from a group including nucleic acid encoding a protein of interest, oligonucleotides, nucleic acid analogues, for example peptide-nucleic acid (PNA), pseudocomplementary PNA (pc-PNA), locked nucleic acid (LNA) etc.
- PNA peptide-nucleic acid
- pc-PNA pseudocomplementary PNA
- LNA locked nucleic acid
- a nucleotide refers to a ribonucleotide, deoxynucleotide or a modified form of either type of nucleotide (e.g., a peptide nucleic acid oligomer).
- nucleic acid molecules are also intended to be a part of the present invention.
- Functional variants are nucleic acid sequences that can be directly translated, using the standard genetic code, to provide an amino acid sequence identical to that translated from the parental nucleic acid molecules.
- nucleic acid sequences include, for example.
- the recombinant nucleic acid molecules encoding a functional SMN1 protein are useful in generating expression cassettes and vector genomes and may be engineered for expression in yeast cells, insect cells, or mammalian cells, such as human cells. Methods are known and have been described previously (e.g., WO 96/09378). A sequence is considered engineered if at least one non-preferred codon as compared to a wild type (WT) sequence is replaced by a codon that is more preferred.
- WT wild type
- a non-preferred codon is a codon that is used less frequently in an organism than another codon coding for the same amino acid
- a codon that is more preferred is a codon that is used more frequently in an organism than a non-preferred codon.
- the frequency of codon usage for a specific organism can be found in codon frequency tables, such as in kazusa.jp/codon.
- more than one non-preferred codon, preferably most or all non-preferred codons are replaced by codons that are more preferred.
- the most frequently used codons in an organism are used in an engineered sequence. Replacement by preferred codons generally leads to higher expression.
- nucleic acid sequence encoding an amino acid sequence includes all nucleotide sequences that are degenerate versions of each other and that encode the same amino acid sequence.
- Nucleic acid sequences can be cloned using routine molecular biology techniques, or generated de novo by DNA synthesis, which can be performed using routine procedures by service companies having business in the field of DNA synthesis and/or molecular cloning (e.g., GeneArt, GenScript, Life Technologies, Eurofins).
- a nucleic acid molecule that contains a sequence encoding an SMN1 gene described herein in any suitable genetic element, e.g., naked DNA, phage, transposon, cosmid, episome, etc., which transfers the SMN1 gene to a host cell, e.g., for generating non-viral delivery systems (e.g., RNA-based systems, naked DNA, or the like), or for generating viral vectors in a packaging host cell, and/or for delivery to a target cells in a subject.
- the genetic element is a vector.
- the genetic element is a plasmid.
- engineered constructs are known to those with skill in nucleic acid manipulation and include genetic engineering, recombinant engineering, and synthetic techniques. See, e.g., Green and Sambrook, Molecular Cloning: A Laboratory Manual, Cold Spring Harbor Press, Cold Spring Harbor, NY (2012).
- compositions comprising an rAAV or a stock of rAAV as described herein in a formulation buffer.
- the pharmaceutical compositions described herein are designed for delivery to subjects in need thereof by any suitable route, or a combination of different routes.
- the pharmaceutical composition is formulated at about 1 x 10 9 genome copies (GC)/mL to about 1 x 10 14 GC/mL of the rAAV. In other embodiments, the pharmaceutical composition is formulated at about 3 x 10 9 GC/mL to about 3 x 10 13 GC/mL of the rAAV. In yet other embodiments, the pharmaceutical composition is formulated at about 1 x 10 9 GC/mL to about 1 x 10 13 GC/mL of the rAAV. In certain embodiments, the pharmaceutical composition is formulated at least 1 x 10 11 GC/mL of the rAAV.
- GC genome copies
- composition comprising an rAAV as described herein and an aqueous suspension media.
- the suspension is formulated for intravenous delivery, intrathecal administration, or intracerebroventricular administration.
- the composition contains at least one rAAV stock and an optional carrier, excipient and/or preservative.
- a “stock” of rAAV refers to a population of rAAV. Despite heterogeneity in their capsid proteins due to deamidation, rAAV in a stock are expected to share an identical vector genome.
- a stock can include rAAV having capsids with, for example, heterogeneous deamidation patterns characteristic of the selected AAV capsid proteins and a selected production system. The stock may be produced from a single production system or pooled from multiple runs of the production system. A variety of production systems, including but not limited to those described herein, may be selected.
- carrier includes any and all solvents, dispersion media, vehicles, coatings, diluents, antibacterial and antifungal agents, isotonic and absorption delaying agents, buffers, carrier solutions, suspensions, colloids, and the like.
- carrier includes any and all solvents, dispersion media, vehicles, coatings, diluents, antibacterial and antifungal agents, isotonic and absorption delaying agents, buffers, carrier solutions, suspensions, colloids, and the like.
- Supplementary active ingredients can also be incorporated into the compositions.
- pharmaceutically-acceptable refers to molecular entities and compositions that do not produce an allergic or similar untoward reaction when administered to a host.
- Delivery vehicles such as liposomes, nanocapsules, nanoparticles, lipid nanoparticle (LNP), microparticles, microspheres, lipid particles, vesicles, and the like, may be used for the introduction of the compositions of the present invention into suitable host cells.
- the rAAV vector delivered vector genomes may be formulated for delivery either encapsulated in a lipid particle, a liposome, a vesicle, a nanosphere, or a nanoparticle or the like.
- the composition includes a final formulation suitable for delivery to a subject, e.g., is an aqueous liquid suspension buffered to a physiologically compatible pH and salt concentration.
- a final formulation suitable for delivery to a subject e.g., is an aqueous liquid suspension buffered to a physiologically compatible pH and salt concentration.
- one or more surfactants are present in the formulation.
- the composition may be transported as a concentrate which is diluted for administration to a subject.
- the composition may be lyophilized and reconstituted at the time of administration.
- a suitable surfactant, or combination of surfactants may be selected from among non-ionic surfactants that are nontoxic.
- the composition includes a carrier, diluent, excipient and/or adjuvant.
- Suitable carriers may be readily selected by one of skill in the art in view of the indication for which the transfer virus is directed.
- one suitable carrier includes saline, which may be formulated with a variety of buffering solutions (e.g., phosphate buffered saline).
- Other exemplary carriers include sterile saline, lactose, sucrose, calcium phosphate, gelatin, dextran, agar, pectin, peanut oil, sesame oil, and water.
- the buffer/carrier should include a component that prevents the rAAV, from sticking to the infusion tubing but does not interfere with the rAAV binding activity in vivo.
- a suitable surfactant, or combination of surfactants may be selected from among non-ionic surfactants that are nontoxic.
- a difunctional block copolymer surfactant terminating in primary hydroxyl groups is selected, e.g., such as Pluronic® F68 [BASF], Lutrol® F68, Synperonic® F68, Kolliphor® Pl 88) which has a neutral pH, has an average molecular weight of 8400.
- Poloxamers may be selected, i.e., nonionic triblock copolymers composed of a central hydrophobic chain of polyoxypropylene (polypropylene oxide)) flanked by two hydrophilic chains of polyoxyethylene (poly(ethylene oxide)), SOLUTOL HS 15 (Macrogol- 15 Hydroxystearate), LABRASOL (Poly oxy capryllic glyceride), poly oxy -oleyl ether, TWEEN (polyoxyethylene sorbitan fatty acid esters), ethanol and polyethylene glycol.
- the formulation contains a poloxamer.
- copolymers are commonly named with the letter "P" (for poloxamer) followed by three digits: the first two digits x 100 give the approximate molecular mass of the poly oxypropylene core, and the last digit x 10 gives the percentage polyoxyethylene content.
- Poloxamer 188 is selected.
- the surfactant may be present in an amount up to about 0.0005 % to about 0.001% of the suspension.
- the composition containing the rAAV is delivered at a pH in the range of 6 to 8, or 7.2 to 7.8, or 7.5 to 8.
- a pH above 7.5 may be desired, e.g., 7.5 to 8, or 7.8.
- a pH of about 6.8 to about 7.2 may be desired.
- the formulation may contain a buffered saline aqueous solution not comprising sodium bicarbonate.
- a buffered saline aqueous solution comprising one or more of sodium phosphate, sodium chloride, potassium chloride, calcium chloride, magnesium chloride and mixtures thereof, in water, such as a Harvard’s buffer.
- the buffer is PBS.
- the buffer is an artificial cerebrospinal fluid (aCSF), e.g., Eliott’s formulation buffer; or Harvard apparatus perfusion fluid (an artificial CSF with final Ion Concentrations (in mM): Na 150; K 3.0; Ca 1.4; Mg 0.8; P 1.0; Cl 155).
- the aqueous solution may further contain Kolliphor® Pl 88, a pol oxamer which is commercially available from BASF which was formerly sold under the trade name Lutrol® F68.
- the aqueous solution may have a pH of 7.2.
- the formulation may contain a buffered saline aqueous solution comprising 1 mM Sodium Phosphate (NasPCh), 150 mM sodium chloride (NaCl), 3mM potassium chloride (KC1), 1.4 mM calcium chloride (CaC12), 0.8 mM magnesium chloride (MgCh), and 0.001% poloxamer (e.g., Kolliphor®) 188, pH 7.2. See, e.g., harvardapparatus.com/harvard-apparatus-perfusion-fluid.html.
- Harvard’s buffer is preferred due to better pH stability observed with Harvard’s buffer.
- the formulation buffer is artificial CSF with Pluronic F68.
- the formulation may contain one or more permeation enhancers.
- suitable permeation enhancers may include, e.g., mannitol, sodium glycocholate, sodium taurocholate, sodium deoxycholate, sodium salicylate, sodium caprylate, sodium caprate, sodium lauryl sulfate, polyoxyethylene-9-laurel ether, or EDTA.
- compositions of the invention may contain, in addition to the rAAV and carrier(s), other conventional pharmaceutical ingredients, such as preservatives, or chemical stabilizers.
- suitable exemplary preservatives include chlorobutanol, potassium sorbate, sorbic acid, sulfur dioxide, propyl gallate, the parabens, ethyl vanillin, glycerin, phenol, and parachlorophenol.
- Suitable chemical stabilizers include gelatin and albumin.
- compositions according to the present invention may comprise a pharmaceutically acceptable carrier, such as defined above.
- the compositions described herein comprise an effective amount of one or more AAV suspended in a pharmaceutically suitable carrier and/or admixed with suitable excipients designed for delivery to the subject via injection, osmotic pump, intrathecal catheter, or for delivery by another device or route.
- suitable excipients designed for delivery to the subject via injection, osmotic pump, intrathecal catheter, or for delivery by another device or route.
- an om maya reservoir is used for delivery.
- the composition is formulated for intrathecal delivery.
- the composition is formulated for intravenous (iv) delivery.
- a therapeutically effective amount of a vector is included in the pharmaceutical composition.
- the selection of the carrier is not a limitation of the present invention.
- Other conventional pharmaceutically acceptable carrier such as preservatives, or chemical stabilizers.
- Suitable exemplary preservatives include chlorobutanol, potassium sorbate, sorbic acid, sulfur dioxide, propyl gallate, the parabens, ethyl vanillin, glycerin, phenol, and parachlorophenol.
- Suitable chemical stabilizers include gelatin and albumin.
- the term “dosage” or “amount” can refer to the total dosage or amount delivered to the subject in the course of treatment, or the dosage or amount delivered in a single unit (or multiple unit or split dosage) administration.
- compositions can be formulated in dosage units to contain an amount of rAAV that is in the range of about 1 x 10 9 genome copies (GC) per gram (g) of brain mass to about 1 x 10 13 GC per g of brain mass, including all integers or fractional amounts within the range and the endpoints.
- the dosage is 1 x IO 10 GC per gram of brain mass to about 1 x 10 13 GC per gram of brain mass.
- the dose of the rAAV administered to a patient is at least about 1.0 x 10 9 GC/g, about 1.5 x 10 9 GC/g, about 2.0 x 10 9 GC/g, about 2.5 x 10 9 GC/g, about 3.0 x 10 9 GC/g, about 3.5 x 10 9 GC/g, about 4.0 x 10 9 GC/g, about 4.5 x 10 9 GC/g, about 5.0 x 10 9 GC/g, about 5.5 x 10 9 GC/g, about 6.0 x 10 9 GC/g, about 6.5 x 10 9 GC/g, about 7.0 x 10 9 GC/g, about 7.5 x 10 9 GC/g, about 8.0 x 10 9 GC/g, about 8.5 x 10 9 GC/g, about 9.0 x 10 9 GC/g, about 9.5 x 10 9 GC/g, about 1.0 x 10 10 GC/g, about 1.5 x 10 10 GC/g, about 2.0
- compositions can be formulated in dosage units to contain an amount of replication-defective virus that is in the range of about 1.0 x 10 9 GC to about 1.0 x 10 16 GC of the rAAV (to treat an average subject of 70 kg in body weight) including all integers or fractional amounts within the range, and preferably 1.0 x 10 12 GC to 1.0 x 10 14 GC for a human patient.
- the compositions are formulated to contain at least IxlO 9 , 2xl0 9 , 3xl0 9 , 4xl0 9 , 5xl0 9 , 6xl0 9 , 7xl0 9 , 8xl0 9 , or 9xl0 9 GC per dose including all integers or fractional amounts within the range.
- the compositions are formulated to contain at least IxlO 10 , 2xlO 10 , 3xl0 10 , 4xlO 10 , 5xl0 10 , 6xlO 10 , 7xlO 10 , 8xl0 10 , or 9xlO 10 GC per dose including all integers or fractional amounts within the range.
- compositions are formulated to contain at least IxlO 11 , 2xlO n , 3xl0 n , 4xlO n , 5xl0 n , 6xlO n , 7xlO n , 8xl0 n , or 9xlO n GC per dose including all integers or fractional amounts within the range.
- the compositions are formulated to contain at least IxlO 12 , 2xl0 12 , 3xl0 12 , 4xl0 12 , 5xl0 12 , 6xl0 12 , 7xl0 12 , 8xl0 12 , or 9xl0 12 GC per dose including all integers or fractional amounts within the range.
- compositions are formulated to contain at least IxlO 13 , 2xl0 13 , 3xl0 13 , 4xl0 13 , 5xl0 13 , 6xl0 13 , 7xl0 13 , 8xl0 13 , or 9xl0 13 GC per dose including all integers or fractional amounts within the range.
- the compositions are formulated to contain at least IxlO 14 , 2xl0 14 , 3xl0 14 , 4x1014, 5xl0 14 , 6xl0 14 , 7xl0 14 , 8xl0 14 , or 9xl0 14 GC per dose including all integers or fractional amounts within the range.
- compositions are formulated to contain at least IxlO 15 , 2xl0 15 , 3xl0 15 , 4xl0 15 , 5xl0 15 , 6xl0 15 , 7xl0 15 , 8xl0 15 , or 9xl0 15 GC per dose including all integers or fractional amounts within the range.
- the dose can range from IxlO 10 to about IxlO 12 GC per dose including all integers or fractional amounts within the range.
- the composition is administered intravenously. In certain embodiments, the composition is administered intrathecally, optionally via intra-ci sterna magna (ICM) injection. In certain embodiments, the composition is administered via intraparenchymal administration. In certain embodiments, the composition is administered via an Ommaya Reservoir delivery system. In certain embodiments, the composition is administered via more than one route, for example intravenously and intrathecally.
- ICM intra-ci sterna magna
- the composition is administered via intraparenchymal administration. In certain embodiments, the composition is administered via an Ommaya Reservoir delivery system. In certain embodiments, the composition is administered via more than one route, for example intravenously and intrathecally.
- the pharmaceutical composition comprising the rAAV as described herein is administrable at a dose of about I x lO 9 GC per gram of brain mass to about I x lO 13 GC per gram of brain mass. In certain embodiments, the pharmaceutical composition comprising the rAAV as described herein is administrable at a dose of about 1 x 10 11 GC per gram of brain mass to about 6 x 10 11 GC per gram of brain mass. In certain embodiments, the pharmaceutical composition comprising the rAAV as described herein is administrable at a dose of about I x lO 11 GC per gram of brain mass.
- the pharmaceutical composition comprising the rAAV as described herein is administrable at a dose of about 3.3 x 10 11 GC per gram of brain mass. In certain embodiments, the pharmaceutical composition comprising the rAAV as described herein is administrable at a dose of about 6.7 x 10 11 GC per gram of brain mass.
- provided herein are methods of treating SMA in a subject in need thereof.
- the methods include administering an rAAV vector provided herein, or a composition comprising the rAAV in an aqueous suspension.
- the methods include treating a subject having one or more symptoms associated with SMA (e.g., atrophy of the limb muscles, difficulty or inability walking, difficulty breathing, or other symptoms of SMA).
- the subject has reduced levels of the survival motor neuron (SMN) protein as a result of loss or mutation of the SMN1 gene.
- the subject has been identified as having type I (Werdnig-Hoffmann disease), type II (Dubowitz disease), type III (Kugelberg- Welander disease), or type IV (adult) SMA.
- the subject having SMA has two mutant alleles of the genomic SMN1 gene.
- the subject has a deletion or mutation (e.g., loss of function point mutation) in each SMN1 allele.
- the subject is homozygous for a SMN1 gene mutation.
- the subject is heterozygous for two different SMN1 gene mutations.
- the subject has at least 1, at least 2, at least 3, at least 4 copies of a functional SMN2 gene.
- the subject has fewer 1, 2, 3, or 4 copies of a functional SMN2 gene.
- the subject is human and selected from the pediatric and adult population. In certain embodiments, the subject is greater than or equal to 18 years of age (e.g., 18 years of age or older).
- the subject is younger than 18 years of age, younger than 10 years of age, or younger than 6 years of age. In certain embodiments, the subject is around 2 weeks, 1 month, 3 months, 6 months, 1 year, 2 years, 3 years, 4 years, or 5 years of age. In certain embodiments, the subject is less than 1 month old. In certain embodiments, the subject is less than 6 months old. In certain embodiments, the subject is less than 1 year old. In certain embodiments, the subject is 1 month to 10 months old. In certain embodiments, the subject is post-natal and less than 2 years old with spinal muscular atrophy (SMA) with bi-allelic mutations in the survival motor neuron 1 (SMN1) gene. In certain embodiments, the subject is not age limited.
- SMA spinal muscular atrophy
- SMA survival motor neuron 1
- treatment includes alleviating symptoms of a patient such that the severity of disease is reduced (e.g., from SMA1 (also, Werdnig-Hoffmann disease, 0- 6 months) to SMA2 (intermediate - 6-18 months, Dubowitz disease), SMA3 (>12 months, Kugelberg- Welander disease), or SMA4 (adult onset).
- SMA1 also, Werdnig-Hoffmann disease, 0- 6 months
- SMA2 intermediate - 6-18 months, Dubowitz disease
- SMA3 >12 months
- Kugelberg- Welander disease Kugelberg- Welander disease
- SMA4 adult onset
- the methods include treating a subject following onset, or prior to onset of one or more disease symptoms (including, for example, one or more of areflexia, particularly in extremities; overall muscle weakness, poor muscle tone, limpness or a tendency to flop; difficulty achieving developmental milestones, difficulty sitting/standing/walking; In small children: adopting of a frog-leg position when sitting (hips abducted and knees flexed); loss of strength of the respiratory muscles: weak cough, weak cry (infants), accumulation of secretions in the lungs or throat, respiratory distress; bellshaped torso (caused by using only abdominal muscles for respiration) in severe SMA type; fasciculations (twitching) of the tongue; and difficulty sucking or swallowing, poor feeding).
- one or more disease symptoms including, for example, one or more of areflexia, particularly in extremities; overall muscle weakness, poor muscle tone, limpness or a tendency to flop; difficulty achieving developmental milestones, difficulty sitting/standing/walking; In small children: adopting of a frog
- the method comprises administering to a subject a suspension of an rAAV as described herein in a formulation buffer at a dose of about 1 x 10 11 to about 1 x 10 14 GC/kg body weight.
- the method comprises intravenous administration at a dose ranging from about 1 x 10 12 genome copies (GC)/kg of rAAV to about 1 x 10 14 GC of rAAV per kg.
- a dose is about 1 x 10 13 GC/kg to about 1 x 10 14 GC of rAAV per patient, or about 3 x 10 13 GC/kg.
- delivery via intravenous administration is contemplated with a dose of about 3 x 10 12 GC/kg to about 1 x 10 14 GC/kg, further including doses of about 3.0 x 10 13 GC/kg and about 1.0 x 10 13 GC/kg.
- the method comprises administering to a subject a suspension of an rAAV as described herein in a formulation buffer at a dose of about 1 x 10 9 GC per gram of brain mass to about 1 x 10 13 GC per gram of brain mass. In certain embodiments, the method comprises administration of an rAAV as described herein at a dose of about 1 x 10 11 GC per gram of brain mass to about 6 x 10 11 GC per gram of brain mass. In certain embodiments, the method comprises administration of an rAAV as described herein at a dose of about 1 x 10 11 GC per gram of brain mass.
- the method comprises administration of an rAAV as described herein at a dose of about 3.3 x 10 11 GC per gram of brain mass. In certain embodiments, the method comprises administration of an rAAV as described herein at a dose of about 6.7 x 10 11 GC per gram of brain mass.
- the subject is delivered a therapeutically effective amount of an rAAV described herein.
- a “therapeutically effective amount” refers to the amount of the composition comprising the nucleic acid sequence encoding SMN1 that delivers and expresses in the target cells an amount of the gene product sufficient to achieve efficacy.
- the dosage is adjusted to balance the therapeutic benefit against any side effects and such dosages may vary depending upon the therapeutic application for which the recombinant vector is employed.
- the levels of expression of the transgene product can be monitored to determine the frequency of dosage resulting in viral vectors, preferably AAV vectors containing the transgene.
- dosage regimens similar to those described for therapeutic purposes may be utilized for immunization using the compositions described herein.
- Suitable, conventional, and pharmaceutically acceptable routes of administration include, but are not limited to, direct delivery to a desired organ (e.g., brain, CSF, heart), intranasal, intrathecal, intratracheal, intraarterial, intraocular, intravenous, intramuscular, subcutaneous, intradermal, intraparenchymal, intracerebroventricular, intrathecal, ICM, lumbar puncture and other parenteral routes of administration. Routes of administration may be combined, if desired.
- the term “administration” or any grammatical variations thereof refers to delivery of a composition described herein to a subject.
- Suitable volumes for delivery of these doses and concentrations may be determined by one of skill in the art. For example, volumes of about 1 pL to 150 mL may be selected for CNS delivery, with the higher volumes being selected for adults. Typically, for newborn infants a suitable volume is about 0.5 mL to about 10 mL, for older infants, about 0.5 mL to about 15 mL may be selected. For toddlers, a volume of about 0.5 mL to about 20 mL may be selected. For children, volumes of up to about 30 mL may be selected. For pre-teens and teens, volumes up to about 50 mL may be selected. In still other embodiments, a patient may receive an intrathecal administration in a volume of about 5 mL to about 15 mL are selected, or about 7.5 mL to about 10 mL.
- the method comprises administering a rAAV as described herein to a subject.
- the rAAV preferably suspended in a physiologically compatible carrier, may be administered to a subject, human or non-human mammalian patient.
- the rAAV is suitably suspended in an aqueous solution containing saline, a surfactant, and a physiologically compatible salt or mixture of salts.
- the formulation is adjusted to a physiologically acceptable pH, e.g., in the range of pH 6 to 9, or pH 6.5 to 7.5, pH 7.0 to 7.7, or pH 7.2 to 7.8.
- the pharmaceutical compositions, as described herein, and the uses thereof comprise delivering to the subject via injection, osmotic pump, intrathecal catheter, or for delivery by another device or route.
- the composition is formulated for intrathecal delivery.
- Intrathecal delivery refers to a route of administration via an injection into the spinal canal, more specifically into the subarachnoid space so that it reaches the cerebrospinal fluid (CSF).
- Intrathecal delivery may include lumbar puncture, intraventricular (including intracerebroventricular (ICV)), suboccipital/intracisternal, and/or Cl-2 puncture.
- material may be introduced for diffusion throughout the subarachnoid space by means of lumbar puncture.
- injection may be into the cistema magna.
- a rAAV vector, or composition as described herein is administrated to a subject in need via the intrathecal administration.
- the intrathecal administration is performed as described in US Patent Publication No. 2018/0339065 Al, published November 29, 2019, which is incorporated herein by reference in its entirety.
- the CNS administration is performed using Ommaya Reservoir (also referred to as Ommaya device or Ommaya system).
- intra sternal delivery or “intracisternal administration” refer to a route of administration for drugs directly into the cerebrospinal fluid of the cisterna magna cerebellomedularis, more specifically via a suboccipital puncture or by direct injection into the cisterna magna or via permanently positioned tube.
- administration of a rAAV vector provided herein is associated with reduced toxicity, which can include, for example, one or more of reduced neuronal degeneration, reduced spinal cord axonopathy, reduced dorsal root ganglia (DRG) toxicity (including, e.g., lesions characterized by neuronal degeneration/necrosis, nerve fiber degeneration, and/or mononuclear cell infiltration), reduced NfL levels in serum and/or CSF, reduced liver toxicity or risk of liver injury or failure.
- reduced toxicity can include, for example, one or more of reduced neuronal degeneration, reduced spinal cord axonopathy, reduced dorsal root ganglia (DRG) toxicity (including, e.g., lesions characterized by neuronal degeneration/necrosis, nerve fiber degeneration, and/or mononuclear cell infiltration), reduced NfL levels in serum and/or CSF, reduced liver toxicity or risk of liver injury or failure.
- DDG dorsal root
- the toxicity is reduced relative to another gene therapy for delivery of SMN1, for example a gene therapy that requires an alternative AVV capsid, is an scAAV, utilizes an alternative promoter that is not a UbC promoter, has a vector genome that does not include miRNA target sequences, and/or is delivered by an alternative route of administration, in particular intravenous administration.
- the reduced toxicity allows for a lower dose of the rAAV provided herein to be delivered, relative to another gene therapy for delivery of SMN1.
- the methods provided include obtaining one or more measurements of an NfL level in a biological sample obtained from the subject.
- administration of an rAAV provided herein results in a reduced or undetectable increase in NfL levels in a biological sample (e.g., serum, plasma, and/or CSF) as compared to an alternative gene therapy for delivery of SMN1.
- a biological sample e.g., serum, plasma, and/or CSF
- the methods provided include obtaining one or more measurements of serum myostatin levels in a biological sample obtained from the subject.
- administration of an rAAV provided herein results in lower levels of serum myostatin levels in a biological sample (e.g., serum, plasma, and/or CSF) as compared to an alternative gene therapy for delivery of SMN1.
- Biological samples may be obtained prior to in addition to following administration of the rAAV vector to evaluate relative changes.
- compositions of the invention are designed so that rAAV vectors carry the nucleic acid expression cassettes including an SMN1 encoding sequence and regulatory sequences that direct expression of the SMN1 in a target cell.
- rAAV vectors carry the nucleic acid expression cassettes including an SMN1 encoding sequence and regulatory sequences that direct expression of the SMN1 in a target cell.
- the method includes administering a co-therapy that is an immunosuppressant to the subject.
- Immunosuppressants for such co-therapy include, but are not limited to, a glucocorticoid, steroids or corticosteroids, antimetabolites, T-cell inhibitors, a macrolide (e.g., a rapamycin or rapalog), and cytostatic agents including an alkylating agent, an anti-metabolite, a cytotoxic antibiotic, an antibody, or an agent active on immunophilin.
- the immune suppressant may include a nitrogen mustard, nitrosourea, platinum compound, methotrexate, azathioprine, mycophenolate mofetil , methotrexate, leflunomide (Arava), cyclophosphamide, chlorambucil (Leukeran), a chloroquine (e.g., hydroxychloroquine), quinine sulfate, mefloquine, a combination of atovaquone and proguanil, sulfasalazine, mercaptopurine, fluorouracil, dactinomycin, an anthracycline, mitomycin C, bleomycin, mithramycin, IL-2 receptor- (CD25-) or CD3-directed antibodies, anti-IL-2 antibodies, ciclosporinabatacept (Orencia), adalimumab (Humira), anakinra (Kineret), certolizumab (Cimzia),
- the immunosuppressive therapy may be started 0, 1, 2, 7, or more days prior to administration of the rAAV vector.
- Such therapy may involve coadministration of two or more drugs, the (e.g., prednisone, micophenolate mofetil (MMF) and/or sirolimus (i.e., rapamycin)) on the same day.
- drugs e.g., prednisone, micophenolate mofetil (MMF) and/or sirolimus (i.e., rapamycin)
- MMF micophenolate mofetil
- sirolimus i.e., rapamycin
- Such therapy may be for about 1 week (7 days), about 60 days, or longer, as needed.
- a tacrolimus-free regimen is selected.
- the requirement for treatment with an immunosuppressant as required for another SMA gene therapy, in terms of dose and/or duration is reduced or eliminated.
- the methods provided include treatment with an immunosuppressant for no longer than about one week, about two weeks, about three weeks, or about one month following administration of the rAAV vector.
- the method comprises a combined treatment that includes administering a rAAV vector as described herein that includes an SMN1 coding sequence in combination with an antisense oligonucleotide (ASO) that increases full-length SMN2 mRNA in a subject (e.g., an ASO that promotes the inclusion of exon 7 in SMN2 mRNA) (See US 2021/0308281 Al, which is incorporated herein by reference).
- ASO antisense oligonucleotide
- the SMN2 ASO is SPINRAZA® (nusinersen).
- the rAAV and ASO may be combined in a single formulation or administered separately.
- the method comprises a combined treatment that includes administering a rAAV vector as described herein that includes an SMN1 coding sequence in combination with one or more additional agent that are olesoxime, AVX-101, CK-2127107, RG7916, RG7800, R07034067, LMI070, SRK-015, a p38aDMAPK inhibitor, a DcpS inhibitor, or a INK inhibitor.
- the method comprises a combined treatment that includes an agent that is a neuroprotective compound such as olesoxime, thyrotropin-releasing hormone, riulzole, certain beta-lactam antibiotics such as ceflt4riaxone, or follistatin.
- the method comprises a combined treatment that includes an agent that promotes muscle restoration, such a reldesemtiv (skeletal troponin activator), apitegromab (Mab that blocks myostatin), GYM329 (blocks myostatin activation).
- the rAAV and the additional agent(s) may be combined in a single formulation or administered separately.
- the method comprises a combined treatment that includes administering an rAAV vector as described herein that includes an SMN1 coding sequence in combination with a small molecule that promotes SMN function (See US 2022/0280548 Al, which is incorporated herein by reference).
- the rAAV and the small molecule may be combined in a single formulation or administered separately.
- the small molecule is a splice modulator, an HD AC inhibitor, or a molecule that modulates the activity of an mRNA decapping enzyme.
- the small molecule is an SMN2 splice modulator.
- the splice modulator is a 7-Disubstituted- phenyl tetracycline. In certain embodiments, the splice modulator is a substituted isoindolinone. In certain embodiments, the splice modulator is a substituted carbazole derivative. In certain embodiments, the SMN2 splice modulators is a substituted 1, 4- diazepane. In certain embodiments, the SMN2 splice modulators is a substituted pyridazine. In certain embodiments, the SMN2 splice modulator is Risdiplam. In certain embodiments, the SMN2 splice modulator is Branaplam.
- the method includes administering anti-AAV neutralizing antibodies (NAb) to reduce peripheral transduction, and to mitigate the potential risk of AAV-induced toxicity.
- the method includes detecting the presence of systemic AAV NAb prior to treating with anti-AAV NAb, wherein patients with levels of anti-AAV NAb in excess of a predetermined level against the rAAV capsid (or a sero- crossreactive capsid) do not require pretreatment.
- levels may be, e.g., in excess of about 1 : 10, about 1 :20, about 1 :50, about 1 : 100, about 1 :250, or higher or lower levels.
- the method further comprises intravenously administering human anti-AAV polyclonal antibodies (e.g., plasma-derived, pooled human immunoglobulin (IVIG)), an anti- AAV monoclonal antibody, or a cocktail of anti-AAV antibodies, to a patient about 1 day to about 2 hours before treatment with the rAAV.
- human anti-AAV polyclonal antibodies e.g., plasma-derived, pooled human immunoglobulin (IVIG)
- IVIG human immunoglobulin
- an anti- AAV monoclonal antibody e.g., anti- AAV monoclonal antibody
- cocktail of anti-AAV antibodies e.g., anti-AAV monoclonal antibody
- the methods of treatment provided herein are not restricted by the presence of anti-AAV NAb detected in a subject, such that efficacy of the treatment is not limited and/or there is no requirement to exclude or delay treatment of a subject that is found to have detectable levels of anti-AAV NAb.
- a combination regimen for preventing off-target delivery rAAV, the regimen comprising (a) pretreating the patient by systemically administering a composition comprising anti-AAV capsid neutralizing antibodies directed against an AAV capsid in a recombinant AAV vector, and (b) administering the rAAV as described herein.
- a composition comprising anti-AAV capsid neutralizing antibodies directed against an AAV capsid in a recombinant AAV vector, and (b) administering the rAAV as described herein.
- a “neutralizing antibody” or “NAb” binds specifically to a viral capsid or envelope and interferes with the infectivity of the virus or a recombinant viral vector having the viral capsid or envelope, thus preventing the recombinant viral vector from delivering effective amounts of a gene product encoded by an expression cassette in its vector genome.
- Various methods for assessing neutralizing antibodies in a patient’s sera may be utilized. The term method and assay may be used interchangeably.
- the term “neutralization assay” and “serum virus neutralization assay” refers to a serological test to detect the presence of systemic antibodies that may prevent infectivity of a virus.
- Immunological assays may include enzyme immunoassay (EIA), radioimmunoassay (RIA), which uses radioactive isotopes, fluoroimmunoassay (FIA) which uses fluorescent materials, chemiluminescent immunoassay (CLIA) which uses chemiluminescent materials and counting immunoassay (CIA) which employs particle-counting techniques, other modified assays such as western blot, immunohistochemistry (IHC) and agglutination.
- EIA enzyme immunoassay
- RIA radioimmunoassay
- FFA fluoroimmunoassay
- FIA fluoroimmunoassay
- CLIA chemiluminescent immunoassay
- CIA counting immunoassay
- other modified assays such as western blot, immunohistochemistry (IHC) and agglutination.
- ELISA enzyme-linked immunosorbent assay
- Example of suitable methods include those described, e.g., R Calcedo, et al, Journal Infectious Diseases, 2009, 199:381-290; GUO, et al., “Rapid AAV_Neutralizing Antibody Determination with a Cell-Binding Assay”, Molecular Therapy: Methods & Clinical Development Vol. 13 June 2019, T. Ito et al, “A convenient enzyme-linked immunosorbent assay for rapid screening of anti-adeno-associated virus neutralizing antibodies”, Ann Clin Biochem 2009; 46: 508-510; US 2018/0356394A2 (Voyager Therapeutics).
- the neutralization ability of an antibody is usually measured via the expression of a reporter gene such as luciferase or GFP.
- a reporter gene such as luciferase or GFP.
- the antibody tested should display a neutralizing activity of 50% or more in one of the neutralization assays described herein.
- neutralizing capacity is determined by measuring the activity of a reporter gene product (e.g., luciferase, GFP).
- the neutralizing capacity of an antibody to a specific viral vector may be at least 50%, e.g., at least 55%, 60%, 65%, 66%, 67%, 68%, 69%, 70%, 71%, 72%, 73%, 74%, 75%, 76%, 77%, 78%, 79%, 80%, 81%, 82%, 83%, 84%, 85%, 86%, 87%, 88%, 89%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or at least 99%.
- NAb titer a measurement of how much neutralizing antibody (e.g., anti-AAV Nab) is produced which neutralizes the physiologic effect of its targeted epitope (e.g., an AAV).
- Anti-AAV NAb titers may be measured as described in, e.g., Calcedo, R., et al., Worldwide Epidemiology of Neutralizing Antibodies to Adeno- Associated Viruses. Journal of Infectious Diseases, 2009. 199(3): p. 381-390, which is incorporated by reference herein.
- Still other co-therapeutics may include, e.g., anti-IgG enzymes, which have been described as being useful for depleting anti-AAV antibodies (and thus may permit administration to patients testing above a threshold level of antibody for the selected AAV capsid), and/or delivery of anti-FcRN antibodies and/or one or more of a) a steroid or combination of steroids and/or (b) an IgG-cleaving enzyme, (c) an inhibitor of Fc-IgE binding; (d) an inhibitor of Fc-IgM binding; (e) an inhibitor of Fc-IgA binding; and/or (f) gamma interferon.
- anti-IgG enzymes which have been described as being useful for depleting anti-AAV antibodies (and thus may permit administration to patients testing above a threshold level of antibody for the selected AAV capsid
- delivery of anti-FcRN antibodies and/or one or more of a) a steroid or combination of steroids and/or (b) an Ig
- anti-FcRN antibodies include, e.g., rozanolixizumab (UCB7665) (UCB SA); IMVT-1401, RVT-1401 (HL 161), HBM9161 (all form HanAll BioPhrma Co. Ltd), Nipocalimab (M281) (Momenta Pharmaceuticals Inc), ARGX-113 (efgartigimod) (Argenx S.E.), orilanolimab (ALXN 1830, SYNT001, Al exion Pharmaceuticals Inc), SYNT002, ABY-039 (Affibody AB), or DX-2507 (Takeda Pharmaceutical Co. Ltd).
- a combination of anti-FcRN antibodies is administered.
- an anti-FcRN antibody is administered in combination with a suitable anti- FcRn ligand (i.e., a peptide or protein construct binding human FcRn so as to inhibit IgG binding).
- an anti-FcRn ligand(s) is administered to a patient having neutralizing antibodies prior to and, optionally, concurrently with a selected viral vector.
- continued expression of an anti-FcRn ligand post administration of the gene therapy vector may desired on a short-term (transient basis), e.g., until such time as the viral vector clears from the patient.
- persistent expression of an anti-FcRn ligand may be desired.
- the ligand may be delivered via a viral vector, including, e.g., in the viral vector expressing the therapeutic transgene.
- the therapeutic gene being delivered is an antibody or antibody construct or another construct comprising an IgG chain.
- the anti-FcRn ligand is delivered or dosed transiently so that the amount of anti-FcRn ligand in the circulation is cleared from the sera before effective levels of vector-mediated transgene product are expressed.
- the FcRn ligand is delivered one to seven days prior to administration of the vector (e.g., rAAV). In certain embodiments, the FcRn ligand is delivered daily. In certain embodiments, the FcRn ligand (e.g., immunoglobulin construct(s)) is delivered on the same day as the vector is administered. In certain embodiments, the FcRn ligand (e.g., immunoglobulin construct(s)) is delivered at least one day to four weeks post- rAAV administration. In certain embodiments, the ligand is delivered for four weeks to six months post-rAAV administration. In certain embodiments, the ligand is dosed via a different route of administration than the rAAV.
- the ligand is dosed orally, intravenously, or intraperitoneally. See also, International Patent Application No. PCT/US2021/037575, filed June 16, 2021, and now published WO 2021/257668 Al, which is incorporated herein by reference in its entirety.
- the method includes measuring a neurofilament level (e.g., pNF-H level).
- the method involves providing a biological sample obtained from a human subject having mutations in both copies of the SMN1 gene that lead to functional SMN protein deficiency; and measuring a neurofilament level (e.g., pNF-H level) in the biological sample.
- the method involves: measuring a neurofilament level (e.g., pNF-H level) in a first biological sample obtained from the human subject before administering a rAAV as described herein that delivers an SMN1 coding sequence; administering the rAAV (e.g., a therapeutically effective amount of the rAAV) to the human subject; and measuring a neurofilament level (e.g., pNF-H level) in a second biological sample obtained from the human subject after administration of the rAAV.
- the neurofilament is a neurofilament heavy chain (e.g., phosphorylated NF-H).
- the neurofilament is a neurofilament medium/intermediate chain. In certain embodiments, the neurofilament is a neurofilament light chain. In certain embodiments, the neurofilament is intemexin. In certain embodiments, the neurofilament is peripherin. See US 2023/0107651 Al, which is incorporated herein by reference.
- biological sample refers to any cell, biological fluid or tissue.
- suitable samples for use in this invention may include, without limitation, whole blood, leukocytes, fibroblasts, serum, urine, plasma, saliva, bone marrow, cerebrospinal fluid, amniotic fluid, and skin cells.
- samples may further be diluted with saline, buffer or a physiologically acceptable diluent. Alternatively, such samples are concentrated by conventional means.
- a kit which includes a concentrated vector suspended in a formulation (optionally frozen), optional dilution buffer, and devices and components required for intrathecal, intracerebroventricular or intracistemal administration.
- the kit may additional or alternatively include components for intravenous delivery.
- the kit provides sufficient buffer to allow for injection. Such buffer may allow for about a 1 :1 to a 1 :5 dilution of the concentrated vector, or more.
- higher or lower amounts of buffer or sterile water are included to allow for dose titration and other adjustments by the treating clinician.
- one or more components of the device are included in the kit.
- Suitable dilution buffer is available, such as, a saline, a phosphate buffered saline (PBS) or a glycerol/PBS.
- the vectors, rAAV, or compositions thereof provided herein may be administered intrathecally via the method and/or the device provided in this section and described in WO 2017/136500 and WO 2018/160582, which are incorporated by reference herein. Alternatively, other devices and methods may be selected.
- the method comprises the steps of CT-guided sub-occipital injection via spinal needle into the cistema magna of a patient.
- CT Computed Tomography
- the term Computed Tomography (CT) refers to radiography in which a three-dimensional image of a body structure is constructed by computer from a series of plane cross-sectional images made along an axis.
- CT Computed Tomography
- the apparatus is described in US Patent Publication No. 2018-0339065 Al, published November 29, 2019, which is incorporated herein by reference in its entirety.
- the vectors, rAAV or compositions thereof provided herein may be administered using an Ommaya Reservoir.
- Example 1 Development of improved AAV vectors for delivery of an hSMNl transgene A series of recombinant vector genomes were engineered and packaged in a
- AAVhu68 capsid The constructs included that same SMN1 coding sequence but different regulatory sequences, including combinations of one of four promoters CB7, UbC, Jet, and hSyn, intron sequences, and miR182 target sequences.
- the vector genomes and their components are depicted in FIG 1.
- ITR Inverted terminal repeats
- AAV ITRs are sequences that are identical on both ends, but in opposite orientation.
- the AAV2 ITR sequences function as both the origin of vector DNA replication and the packaging signal of the vector genome, when AAV and adenovirus helper functions are provided in trans. As such, the ITR sequences represent the only cis sequences required for vector genome replication and packaging. 2.
- CB7 promoter a cytomegalovirus immediate-early (CMV IE) enhancer and the chicken P-actin promoter
- CMV IE cytomegalovirus immediate-early
- UbC ubiquitin C
- hSYN human synapsin 1
- JeT promoter synthetic promoter
- Introns (i) chimeric chicken P-actin intron (CI); and (ii): intron (PI) (SEQ ID NO: 11) (see also GenBank: U47121.2).
- hSMNl coding sequence engineered SMN1 coding sequence (SEQ ID NO: 7).
- miR182 target sequences a tandem repeat of four target sequences (each having the nucleotide sequence set forth in SEQ ID NO: 12) that are capable of being bound by miR182 to promote a DRG-specific reduction of transgene expression and toxicity (as described, for example, in WO 2020/132455 Al and WO 2021/231579 Al).
- Polyadenylation Signal The rabbit P-globin polyadenylation signal (rBG) provides cis sequences for efficient polyadenylation of the antibody mRNA. This element functions as a signal for transcriptional termination, a specific cleavage event at the 3’ end of the nascent transcript and addition of a long polyadenyl tail. (SEQ ID NO: 13).
- trans plasmid pAAV2/hu68n.KanR helper plasmid: pAdDeltaF6(Kan)
- mice were euthanized and liver tissue was harvested for detection of hSMNl transcript by ISH. IV administered WT mice showed that the CB7 promoter generated the highest expression of hSMNl, followed by the Ubc promoter and the Jet promoter.
- mice FVB.Cg- Grm7Tg.(SMN2)89Ahmb SmnltmlMsdTg(SMN2*delta7)4299Ahmb/J., also referred as SMN A 7 mice.
- This triple mutant mouse harbors two transgenic alleles and a single targeted null mutant (the mice lack murine Smn and express an intact human SMN2 gene plus SMN2A7 cDNA).
- the mice exhibit low birth weight and at week-1 display an abnormal gait, shakiness in the hind limbs, and a tendency to fall over. The mean survival of these mice is approximately 17 days.
- mice were administered IxlO 11 GC (ICV) of one of the following vectors:
- hSMNl transgene
- SMNA7 mice treated with AAVhu68.Ubc.PI.hSMN1.4xmiR182.rBG also demonstrated a gradual improvement in body weights that was better than with the other vectors (FIG. 4).
- mice In addition to best survival, AAVhu68.Ubc.PI.hSMN1.4xmiR182.rBG treated mice also showed normalized righting reflex and improved clasping scores, a measure of compromised information about limb and joint position, altered reflexes, or improper integration of sensory and motor information including dysfunction of proprioceptive neurons (FIG. 5).
- Example 3 NHP study for expression and safety following administration of SMN1 AAV vectors
- a safety and toxicity study in NHP was conducted with the following three test articles ICM administered at a dose of 3.0 x 10 13 GC (3.3 x 10 11 GC/g brain) to adult rhesus macaques (6-7 years old), three animals per group, both genders: AAVhu68.CB7.hSMNl.rBG
- hSMNl expressed in motor neurons following ICM administration was verified by hSMNl and ChAT ISH. Overall, the number of hSMNl positive cells was comparable in all three groups, as expected given the use of the same capsid, although the signal intensity was weaker with the UbC promoter, as expected given the relative strengths of the CB7 and UbC promoters (FIG. 8A and FIG. 8B). Based on the improved survival and rescue of phenotype in rodent studies with 52% of motor neuron transduction, the proportion of hSMNl+ cells detected in UbC. hSMNl.4xmiRl 82 administered animals (45%) is likely to lead to benefit in patients.
- the clinical pathology assessment revealed a mild asymptomatic pleocytosis in the CSF of animals treated with the vector containing the Ubc promoter (AAVhu68.UbC.PI.hSMN1.4xmiR182.rBG) (FIG. 9).
- Nerve conduction velocity and sensory nerve action potential amplitude were not affected by administration of any of the vectors (FIG. 10) and neuronal degeneration was minimal to mild and similar across the three different promoters (FIG. 11).
- AAVhu68.UbC. hSMNl.4XmiRl 82 led to the least levels of spinal cord dorsal axonopathy, and the lowest CSF NfL elevation at Day 28, consistent with a favorable DRG toxicity profile (FIG. 12; FIG. 13A and FIG. 13B).
- the lower NfL fold change in AAVhu68.UbC. hSMNl.4XmiRl 82 treated animals was consistent with lower axonopathy scores (FIG. 14A and FIG. 14B).
- Blood chemistry was unremarkable in treated macaques over the 35-day study period. No animal presented with a clinically remarkable condition or neurological concern from cage side assessments. Blood clotting was normal, with the exception of small, transient increases in clotting factor fibrinogen. Biomarkers of liver and kidney damage and circulating ions and metabolites were all within normal ranges in treated monkeys. Treatment with the three vectors showed no evidence of toxicity.
- ELISPOT testing identified moderate immune responses against AAVhu68 peptide pool C in PBMC and spleen. Overall, immune responses against AAVhu68 were milder in the Ubc.4XmiR182 and CB7.4XmiR182 promoter groups. Immune responses to AAVhu68 did not lead to any adverse events in the vector treated animals. ELISPOT testing identified moderate immune responses against hSMNl peptides in PBMCs, liver and spleen.
- Anti -transgene immunogenicity can confound results of NHP studies due to non-selfresponses but is not a concern in the trial as patient are expected to be tolerant to self SMN coming from SMN2 transcription.
- the MED was determined based on analysis of improvement in survival, behavioral phenotypes, along with efficacy of transgene product expression in the target tissues of AAVhu68.Ubc.PI.hSMN1.4xmiR182.rBG -treated SMNA7 mice compared to vehicle-treated WT control mice.
- AAVhu68.Ubc.PI.hSMN1.4xmiR182.rBG -treated wildtype mice did not exhibit evidence of over expression toxicity until study day 120 (FIG. 16).
- Administration of AAVhu68.Ubc.PI.hSMN1.4xmiR182.rBG to SMNA7 mice at two doses resulted in improvement of body weight (FIG. 17), while administration of the vector to wildtype mice did not impact body weight gain (FIG. 18).
- Example 5 Toxicology Study - AAVhu68.Ubc.PI.hSMN1.4xmiR182.rBG administered via intra-ci sterna magna (ICM) injection to juvenile rhesus macaques
- a GLP-compliant toxicology study was performed using juvenile (2-3 years old) NHPs. This age range allows for modeling the size and anatomy of the cisterna magna of the youngest intended patient population to the greatest extent feasible.
- the dimensions of the CNS in the NHP act as a representative model of our target clinical population and allow for the administration of AAVhu68.Ubc.PI.hSMN1.4xmiR182.rBG using the intended clinical route of administration.
- the toxicological and immune responses of the NHP closely resemble that of humans. This study provided critical data on route of administration- related safety and pharmacokinetics of the test article after administration to rhesus macaques.
- Both sexes were selected to model the intended patient population in the planned clinical trial (male and female SMA patients).
- the GLP-compliant toxicology study in juvenile rhesus macaques used a dose of 5.8 x 10 13 GC corresponding to the highest dose (1 x 10 11 GC/animal) used in the MED mouse study, which equates to approximately 6.7 x 10 11 GC/g of brain for mice (assuming brain weight in newborn mice is 0.150g) and 6.0 x 10 13 GC/g for juvenile rhesus (assuming brain weight of 90 grams).
- the dose also represents a close to technical maximal feasible dose based on CSF safe maximal injection volume (1.5 mL) in juvenile rhesus macaques and vector titer constraints.
- the GLP-compliant toxicology study in juvenile rhesus macaques was 90 days long.
- the 90-day duration of this study allowed for the confirmation of the non-progressive nature of the DRG pathology and to obtain chronic safety and immunology data.
- the ICM route was selected for the GLP-compliant toxicology study in juvenile rhesus macaques to mirror the intended clinical route and enable the use of a clinical administration system comparable to the one utilized in the planned clinical trial.
- BAb binding antibodies
- BT Blue Top: Sodium Citrate
- CSF cerebrospinal fluid
- DRG dorsal root ganglion
- ELISPOT enzyme-linked immunospot
- GT Green Top: Heparin
- ITFFB intrathecal final formulation buffer
- kg kilogram
- LT Lavender Top: K2EDTA; mg, milligram
- NA Not Applicable
- NAb neutralizing antibodies
- PMBC peripheral blood mononuclear cell
- PK pharmacokinetics
- RC Red Top: No anti-coagulant.
- a Number of animals assessed. b Includes complete blood counts and differentials (hematology), clinical chemistries, and coagulation panel.
- mice were anesthetized and bled from a peripheral vein for coagulation panel, CBC, serum chemistry, neutralizing (NAb) and binding (BAb) antibodies to the hu68 capsid, peripheral blood mononuclear cells (PBMC) isolation, vector PK, and biomarker analysis,
- ICM administration of AAVhu68.Ubc.PI.hSMN1.4xmiR182.rBG to juvenile NHPs did not impact sensory nerve action potential (SNAP) or conduction velocity in the treated animals (FIG. 22).
- No CSF related abnormalities were identified following ICM administration of AA Vhu68.Ubc.PI.hSMN1.4xmiRl 82. rBG to juvenile NHPs (FIG. 23).
- Administration of the vector did not result in elevation of liver enzymes (FIG. 24) or impact liver functions (FIG. 23 A - FIG. 23 C).
- ICM Administration of AAVhu68.Ubc.PI.hSMN1.4xmiR182.rBG to juvenile NHPs did not impact coagulation parameters (FIG. 26).
- ICM administration of AAVhu68.Ubc.PI.hSMN1.4xmiR182.rBG to rhesus monkeys was safe and well-tolerated.
- Example 6 Method for Scaling from Nonclinical Doses to Clinical Doses
- ICM vector administration results in immediate vector distribution within the CSF compartment, it is anticipated that both efficacy and toxicity are related to CNS vector exposure. Doses are therefore scaled by brain mass, which provides an approximation of the size of the CSF compartment. Dose conversions between animals and patients in the FIH trial are based on a brain mass of 0.15 g for a neonatal mouse, 90 g for a juvenile NHP, 488 g for a 1-4 months old -month infant and 960 g for a >12- month child. Estimated brain weights for each age range for human infants are derived from the male and female brain weights presented in the table below.
- Example dose scaling from neonatal mice, juvenile NHPs, and equivalent human doses are presented using doses used in the NHP toxicology and murine MED studies.
- the administration volume is also scaled from NHPs to humans based on the estimated volumes for cerebral CSF and spinal CSF so that less than 10% of the total CSF volume is being administered, consistent with NHP safety studies.
- Example dose scaling from animals to humans based on brain mass a Doses are scaled based on a brain mass of 0.15 g for a neonatal mouse, 90 g for a juvenile rhesus macaque, and 488 g for a 1-4-month-old human.
- the selected clinical doses are greater than the mouse MED, to support the prospect of direct benefit in the phase I trial and is lower than or equal to the highest tolerated dose in the GLP compliant NHP safety study.
- use of the optimal route of administration in rodents to demonstrate target engagement while the NHP study informs on the safety of the maximal feasible dose (MFD) with intrathecal dosing constraints (limited volume and vector titer). If the NHP study demonstrates safety, then patients are dosed close to the MFD, which is typically several folds over the MED obtained in mice.
- MFD maximal feasible dose
- DRG pathology presents as mostly minimal to moderate asymptomatic DRG pathology characterized by mononuclear cell infiltrates, neuronal degeneration, and secondary axonopathy of central and peripheral axons.
- DRG pathology is similar from 1-5 months post injection and becomes less severe after 6 months. Higher AAV doses correlate with increased severity. Younger NHPs (infants and juveniles) appear to exhibit less severe pathology compared to adult NHPs. Animal sex and vector purification method have no impact.
- DRG sensory neuron degeneration occurs in the absence of toxicity to other heavily transduced cells (e.g., hepatocytes, cortical neurons, and spinal motor neurons), indicating that DRG sensory neurons are selectively vulnerable to AAV-mediated toxicity.
- DRG sensory neurons are selectively vulnerable to AAV-mediated toxicity.
- only a subset of DRG sensory neurons is affected, degenerating within 14-30 days after gene transfer. Following this acute insult, no further degeneration of transduced cells is detectable after 35 days, although slow degeneration of the associated axons can continue until 90 days. Findings are thereafter non progressive between 90 and 180 days with either similar or improved pathology scores.
- the proposed clinical trial is designed to further improve on the safety profile of previous AAV clinical trials by using an ICM ROA that requires lower doses of vector than those typically administered systemically to reduce the risk of sensory neuron toxicity.
- the planned clinical trial also employs detailed monitoring for sensory changes with a longitudinal monitoring of serum NfL levels to detect even subclinical DRG toxicity.
- AAVhu68.Ubc.PI.hSMNlco.4xmiR182.rBG incorporates miR182 target sequences into the 3’ UTR of the human SMN1 sequence. These sequences were incorporated to downregulate transgene product expression in DRG in an effort to minimize the potential for AAV-associated sensory neuron toxicity, which is believed to be primarily caused by transgene product overexpression.
- MicroRNAs post-transcriptionally downregulate expression of target messenger ribonucleic acids (mRNAs) in multicellular organisms by affecting both mRNA stability and translation.
- mRNAs target messenger ribonucleic acids
- miR182 expression is largely restricted to the dorsal root ganglia (DRG)
- DRG dorsal root ganglia
- the miR182 target sequences enable DRG-specific downregulation of the human SMN1 transgene product.
- This detargeting approach aims to minimize the potential for AAV-associated DRG sensory neuron toxicity.
- the current clinical trial is designed to further improve on the safety profile of previous AAV clinical trials by using an ICM route that requires lower doses of vector than those typically administered systemically to reduce the risk of sensory neuron toxicity.
- the planned clinical trial therefore employs detailed monitoring for sensory changes as well as nerve conduction studies to detect even subclinical DRG toxicity.
- the benefit/risk profile for ICM administration of AAVhu68.Ubc.PI.hSMNlco.4xmiR182.rBG is expected to remain favorable.
- Example 7 First-in-human (FH4) trial
- the FIH trial is an open-label, multi-center, dose escalation study of
- ICM intra-cistema magna
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Genetics & Genomics (AREA)
- General Health & Medical Sciences (AREA)
- Engineering & Computer Science (AREA)
- Molecular Biology (AREA)
- Medicinal Chemistry (AREA)
- Organic Chemistry (AREA)
- Biotechnology (AREA)
- Biochemistry (AREA)
- Veterinary Medicine (AREA)
- Pharmacology & Pharmacy (AREA)
- Public Health (AREA)
- Epidemiology (AREA)
- Animal Behavior & Ethology (AREA)
- Zoology (AREA)
- Biophysics (AREA)
- Gastroenterology & Hepatology (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Toxicology (AREA)
- Biomedical Technology (AREA)
- Bioinformatics & Cheminformatics (AREA)
- General Engineering & Computer Science (AREA)
- Wood Science & Technology (AREA)
- Physics & Mathematics (AREA)
- Plant Pathology (AREA)
- Virology (AREA)
- Microbiology (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
- Medicines Containing Material From Animals Or Micro-Organisms (AREA)
Abstract
Expression cassettes and recombinant AAV vectors comprising the same for delivery of a nucleotide sequence encoding hSMN1 are provided. Compositions provided are useful in methods for the treatment of spinal muscular atrophy (SMA) in a subject in need thereof.
Description
COMPOSITIONS AND METHODS FOR TREATMENT OF SPINAL MUSCULAR ATROPHY
REFERENCE TO AN ELECTRONIC SEQUENCE LISTING
The electronic sequence listing filed herewith named “21-10509. PCT. xml” (53,945 bytes, created on August 8, 2024) is incorporated herein by reference in its entirety.
BACKGROUND OF THE INVENTION
Spinal muscular atrophy (SMA) is an autosomal recessive disorder caused by mutations in the SMN1 gene resulting in reduced expression of survival motor neuron (SMN) protein. It is the most common genetic cause of infant mortality and the worldwide incidence of SMA is ~1 in 10,000 live births. SMN proteins have roles in spliceosomal assembly, protein translation, cytoskeletal dynamics, and mitochondrial function. Disruption of the SMA motor unit (motor neurons and the muscle fibers they innervate) underlies the most evident clinical manifestation of SMA (skeletal muscle weakness). Infants with SMA type 1 have severe manifestations, with profound hypotonia and progressive respiratory and swallowing difficulties. Patient management typically includes physiotherapy, orthopedic support, respiratory care, and management of bulbar impairment.
Several therapies for SMA have been developed, including nusinersen (Spinraza), an antisense oligonucleotide that alters splicing of SMN2 protein, onasemnogene abeparvovec (Zolgensma), and adeno-associated virus (AAV)-mediated gene therapy, and risdiplam (Evrysdi), a modifier of splicing of SMN2 protein. While significant advances in the treatment of SMA, these treatments are costly and can require repeated administration (in the case of nusinersen and risdiplam). While AAV-mediated gene therapy is effective with only a single administration, the current FDA approved product (Zolgensma) for treatment of SMA type I is costly and has been associated with several incidents of severe toxicity, including acute liver failure. Further, its safety profile currently prohibits extension of use of Zolgensma to less severe, older SMA II/III patients.
What is needed are improved treatments for SMA that are effective to ameliorate symptoms of the disorder while having reduced potential toxic side-effects.
SUMMARY OF THE INVENTION
In one aspect, provided herein is a recombinant adeno-associated virus (AAV) comprising an AAV capsid containing a vector genome, wherein the vector genome comprises an expression cassette comprising: (a) a UbC promoter sequence, (b) a coding sequence for a functional human survival of motor neuron 1 (SMN1) protein, (c) at least four miR182 target sequences, and (d) a polyA sequence, wherein the coding sequence, the at least four miR182 target sequences, and the polyA sequence are operably linked to the UbC promoter sequence. In certain embodiments, the recombinant AAV of claim 1, wherein the SMN1 protein is an SMN1 isoform D protein. In certain embodiments, the coding sequence comprises the nucleotide sequence of SEQ ID NO: 7 or a nucleotide sequence at least 95% identical to SEQ ID NO: 7. In certain embodiments, the recombinant AAV is not a self- complementary AAV vector.
In another aspect, provided herein is a composition comprising a stock of a recombinant AAV in an aqueous suspension media. In certain embodiments, the suspension is formulated for intrathecal delivery, optionally wherein the intrathecal delivery is intracerebroventricular (ICV) injection or intraci sternal magna (ICM) injection.
In another aspect, provided herein is a pharmaceutical composition comprising a recombinant AAV and an aqueous formulation buffer. In certain embodiments, the composition is formulated for intrathecal delivery. In certain embodiments, the composition is formulated for intracerebroventricular (ICV) injection or intracistemal magna (ICM) injection.
In yet another aspect, provided herein is a recombinant nucleic acid molecule comprising an expression cassette comprising: (a) a 5’ AAV ITR sequence, (b) a UbC promoter sequence, (c) a coding sequence for a functional human survival of motor neuron 1 (SMN1) protein, (d) at least four miR182 target sequences, (e) a polyA sequence, and (f) a 3’ AAV ITR sequence, wherein the coding sequence, the at least four miR182 target sequences, and the polyA sequence are operably linked to the UbC promoter sequence. In certain embodiments, the SMN1 protein is an SMN1 isoform D protein. In certain embodiments, the coding sequence comprises the nucleotide sequence of SEQ ID NO: 7 or a nucleotide sequence at least 95% identical to SEQ ID NO: 7. In certain embodiments, a plasmid comprising the nucleic acid is provided. In yet embodiments, provided herein is a packaging host cell comprising the recombinant nucleic acid molecule or the plasmid.
In another aspect, provided herein is a recombinant AAV production system useful for producing a recombinant AAV.
In yet another aspect, provided herein is a method for treating spinal muscular atrophy (SMA) in a subject in need thereof, the method comprising administering to the subject an aqueous suspension comprising a recombinant AAV as described herein. In certain embodiments, the subject has type I (Werdnig-Hoffmann disease), type II (Dubowitz disease), type III (Kugelberg- Welander disease), or type IV (adult) SMA.
These and other aspects of the invention are apparent from the following detailed description of the invention.
BRIEF DESCRIPTION OF THE DRAWINGS
FIG. 1 provides schematics of rAAV.hSMNl vector genomes.
FIG. 2 shows ISH for detection of hSMNl expression in the liver of wildtype following IV administration of vectors with different promoters.
FIG. 3 shows survival of SMNA7 mice following administration of AAVhu68. hSMNl vectors.
FIG. 4 shows body weights of SMNA7 mice following administration of AAVhu68. hSMNl vectors.
FIG. 5 shows righting reflex (left) and clasping (right) responses of SMNA7 mice following administration of AAVhu68. hSMNl vectors.
FIG. 6 shows survival, body weights, and clasping response scores of C57BL/6J mice following ICV administration of AAVhu68. hSMNl vectors.
FIG. 7A and FIG. 7B show levels of neuron transduction by measurement of transduced motor neurons by ISH (ChAT+hSMNl transgene+ cells) (FIG. 7A) and cardiac pathology scores following ICV administration of a AAVhu68.CB7.CI.hSMNlco.rBG or AAVhu68.Ubc.PI.hSMNlco.4xmiR182.rBG vector to SMNA7 mice.
FIG. 8 A and FIG. 8B show levels of expression of hSMNl (ISH intensity) (FIG. 8 A) and quantification of SMN+ChAT+ cells (FIG. 9B) following ICM administration of AAVhu68.CB7.CI.hSMNlco.rBG (CB7), AAVhu68.CB7.CI.hSMNlco.4xmiR182.rBG (CB7.4xmiR182), and AAVhu68.UbC.PI.hSMNlco.4xmiR182.rBG (UBC.4xmiR182) vectors.
FIG. 9 shows results of clinical pathology studies, including CSF WBC counts (left) and CSF RBC counts (right) following ICM administration of AAVhu68.CB7.CI.hSMNlco.rBG, AAVhu68.CB7.CI.hSMNlco.4xmiR182.rBG, and AAVhu68.UbC.PI.hSMNlco.4xmiR182.rBG vectors to NHPs.
FIG. 10 shows results of nerve conduction studies following ICM administration of AAVhu68.CB7.CI.hSMNlco.rBG, AAVhu68.CB7.CI.hSMNlco.4xmiR182.rBG, and AAVhu68.UbC.PI.hSMNlco.4xmiR182.rBG vectors to NHPs.
FIG. 11 shows results from assessments of DRG neuronal degeneration following ICM administration of AAVhu68.CB7.CI.hSMNlco.rBG, AAVhu68.CB7.CI.hSMNlco.4xmiR182.rBG, and AAVhu68.UbC.PI.hSMNlco.4xmiR182.rBG vectors to NHPs.
FIG. 12 shows results from assessments of spinal cord axonopathy following ICM administration of AAVhu68.CB7.CI.hSMNlco.rBG, AAVhu68.CB7.CI.hSMNlco.4xmiR182.rBG, and AAVhu68.UbC.PI.hSMNlco.4xmiR182.rBG vectors to NHPs.
FIG. 13 A and FIG. 13B show results from scoring of axonopathy severity (FIG. 13 A) and measurements of NfL in CSF on day 29 (FIG. 13B) following ICM administration of AAVhu68.CB7.CI.hSMNlco.rBG, AAVhu68.CB7.CI.hSMNlco.4xmiR182.rBG, and AAVhu68.UbC.PI.hSMNlco.4xmiR182.rBG vectors to NHPs.
FIG. 14A and FIG. 14B show spinal cord axonopathy scores versus NfL levels in CSF (FIG. 14A) and serum (FIG. 14B) following ICM administration of AAVhu68.CB7.CI.hSMNlco.rBG, AAVhu68.CB7.CI.hSMNlco.4xmiR182.rBG, and AAVhu68.UbC.PI.hSMNlco.4xmiR182.rBG vectors to NHPs.
FIG. 15 shows survival of SMNA7 mice following ICV administration of various doses of an AA Vhu68.Ubc.PI.hSMN1.4xmiRl 82. rBG vector.
FIG. 16 shows survival of wildtype mice following ICV administration of various doses of an AAVhu68.Ubc.PI.hSMN1.4xmiRl 82. rBG vector.
FIG. 17 shows body weights of SMNA7 mice following ICV administration of various doses of an AAVhu68.Ubc.PI.hSMN1.4xmiRl 82. rBG vector.
FIG. 18 shows body weights of wildtype mice following ICV administration of various doses of an AAVhu68.Ubc.PI.hSMN1.4xmiRl 82. rBG vector.
FIG. 19 shows results from evaluation of righting reflexes in SMNA7 mice following ICV administration of the indicated doses of an AAVhu68.Ubc.PI.hSMN1.4xmiR182.rBG vector.
FIG. 20 shows results from evaluation of hindlimb clasping responses in wildtype mice following ICV administration of the indicated doses of an AAVhu68.Ubc.PI.hSMNl ,4xmiRl 82.rBG vector.
FIG. 21 shows results from evaluation of hindlimb clasping responses in SMNA7 mice following ICV administration of the indicated of an AAVhu68.Ubc.PI.hSMNl ,4xmiRl 82.rBG vector.
FIG. 22 shows results of nerve conduction studies (NCS) following ICM administration of AAVhu68.Ubc.PI.hSMN1.4xmiR182.rBG to NHPs.
FIG. 23 shows results of clinical pathology studies, including CSF WBC counts (top left), CSF RBC counts (top right), CSF protein levels (bottom left), and CSF glucose levels (bottom right) following ICM administration of AAVhu68.Ubc.PI.hSMN1.4xmiR182.rBG to NHP.
FIG. 24 shows results from evaluation of liver enzyme levels following ICM administration of AAVhu68.Ubc.PI.hSMN1.4xmiR182.rBG to NHPs.
FIG. 25 A - FIG. 25C show results from evaluation of liver function by detection of total protein, albumin, globulin, and albumin/globulin ratios (A/G) (FIG. 25 A), measurement of creatine levels (FIG. 25B), and measurement of blood urea nitrogen (BUN) levels (FIG. 25C) following ICM administration of AAVhu68.Ubc.PI.hSMN1.4xmiR182.rBG to NHPs.
FIG. 26 shows results from evaluation of blood clinical pathology following ICM administration of AAVhu68.Ubc.PI.hSMN1.4xmiR182.rBG to NHPs.
DETAILED DESCRIPTION OF THE INVENTION
Provided herein are recombinant adeno-associated virus (AAV) vectors having an AAV capsid containing a nucleic acid having a sequence encoding a human survival of motor neuron (SMN1) gene under control of regulatory sequences. The vectors, as well as compositions containing these vectors, are useful for treatment of SMA in patients need thereof.
Spinal muscular atrophy or proximal spinal muscular atrophy (SMA) is a genetic, neurodegenerative disorder characterized by the loss of spinal motor neurons. SMA is an
autosomal recessive disease of early onset and is currently a leading cause of death among infants. The severity of SMA varies among patients and has thus been classified into different types depending on the age of onset and motor development milestones. SMA 0 designation has been proposed to reflect prenatal onset and severe joint contractures, facial diplegia, and respiratory failure. Three types of post-natal form of SMA have been designated. Type I SMA (also called Werdnig-Hoffmann disease) is the most severe form with onset at birth or within 6 months and typically results in death within 2 years. Children with type I SMA are unable to sit or walk and have serious respiratory dysfunction. Type II SMA is the intermediate form with onset within the first 2 years. Children with Type II SMA are able to sit, but cannot stand or walk. Type III (also called Kugelberg- Welander disease) begins after 18 months to 2 years of age (Lefebvre et al., Hum. Mol. Genet., 1998, 7, 1531-1536) and usually has a chronic evolution. Children with Type III SMA can stand and walk unaided at least in infancy. Adult form (type IV) is the mildest form of SMA, with onset after 30 years of age, and few cases have been reported. Type III and type IV SMA are also known as later- onset SMA.
The molecular basis of SMA results from the loss of both copies of survival motor neuron gene 1 (SMN1), which may also be known as SMN Telomeric, a protein that is part of a multi-protein complex thought to be involved in snRNP biogenesis and recycling. A nearly identical gene, SMN2, which may also be known as SMN Centromeric, exists in a duplicated region on chromosome 5ql3 and modulates disease severity. Expression of the normal SMN1 gene results solely in expression of survival motor neuron (SMN) protein. Although SMN1 and SMN2 have the potential to code for the same protein, SMN2 contains a translationally silent mutation at position +6 of exon 7, which results in inefficient inclusion of exon 7 in SMN2 transcripts. Thus, the predominant form of SMN2 is a truncated version, lacking exon 7, which is unstable and inactive (Cartegni and Krainer, Nat. Genet., 2002, 30, 377-384). Expression of the SMN2 gene results in approximately 10-20% of the SMN protein and 80-90% of the unstable/non-functional SMN delta 7 protein. SMN protein plays a well- established role in assembly of the spliceosome and may also mediate mRNA trafficking in the axon and nerve terminus of neurons.
“Patient” or “subject” or “individual” as used herein refer to a mammalian animal, including a human, a veterinary or farm animal, a domestic animal or pet, and animals normally used for clinical research. In certain embodiments, the subject is a human.
The words “comprise”, “comprises”, and “comprising” are to be interpreted inclusively rather than exclusively. The words “consist”, “consisting”, and its variants, are to be interpreted exclusively, rather than inclusively. While various embodiments in the specification are presented using “comprising” language, under other circumstances, a related embodiment is also intended to be included and described using “consisting of’ or “consisting essentially of’ language. As used throughout this specification and the claims, the terms “comprising”, “containing”, “including”, and its variants are inclusive of other components, elements, integers, steps and the like. Conversely, the term “consisting” and its variants are exclusive of other components, elements, integers, steps and the like.
It is to be noted that the term “a” or “an” refers to one or more. As such, the terms “a” (or “an”), “one or more,” and “at least one” are used interchangeably herein.
As used herein, the term “about”
refers to a variant of ±10% from the reference integer and values therebetween, unless otherwise specified. For example, “about” 500 pM includes ±50 (i.e., 450 - 550, which includes the integers therebetween). For other values, particularly when reference is to a percentage (e.g., 90% of taste), the term “about” is inclusive of all values within the range including both the integer and fractions.
As described above, the term “about” when used to modify a numerical value means a variation of ±10%, (±10%, e.g., ±1, ±2, ±3, ±4, ±5, ±6, ±7, ±8, ±9, ±10, or values therebetween) from the reference given, unless otherwise specified. Herein, “up to” a number (for example, up to 50) includes the number (for example, 50). The term “in the range” or “within a range” (and similar statements) includes the endpoints of the stated range. Also herein, the recitations of numerical ranges by endpoints include all numbers subsumed within that range (e.g., 1 to 5 includes 1, 1.5, 2, 2.75, 3, 3.80, 4, 5, etc.).
In certain instances, the term “E±#” or the term “e+#” is used to reference an exponent. For example, “5E10” or “5el0” is 5 x 1010. These terms may be used interchangeably.
With regard to the description of various embodiments herein, it is intended that each of the compositions herein described, is useful, in another embodiment, in the methods of the invention. In addition, it is also intended that each of the compositions herein described as useful in the methods, is, in another embodiment, itself an embodiment of the invention.
Unless defined otherwise in this specification, technical and scientific terms used herein have the same meaning as commonly understood by one of ordinary skill in the art to
which this invention belongs and by reference to published texts, which provide one skilled in the art with a general guide to many of the terms used in the present application.
Expression Cassette
In one aspect, provided herein are engineered nucleic acid constructs that include hSMNl coding sequences. The nucleic acids can be used for generating viral vectors for therapeutic delivery of the transgene to target cells of a subject, and/or for delivery of the SMN1 coding sequence to a host cell, e.g., naked DNA or plasmid. In certain embodiments, an expression cassette comprising the hSMNl sequence is provided.
As used herein, an “expression cassette” refers to a nucleic acid molecule which comprises a biologically useful nucleic acid sequence (e.g., a gene cDNA encoding a protein, enzyme or other useful gene product, mRNA, etc.) and regulatory sequences operably linked thereto which direct or modulate transcription, translation, and/or expression of the nucleic acid sequence and its gene product. In certain embodiments, the term an “expression cassette” refers to a nucleic acid molecule that includes an SMN1 encoding sequence and regulatory sequences therefore (e.g., promoter, enhancer, poly A sequences), which cassette may be packaged into the capsid of a viral vector (e.g., a viral particle). Typically, such an expression cassette for generating a viral vector contains the hSMN sequences described herein flanked by packaging signals of the viral genome and other expression control sequences such as those described herein. For example, for an AAV viral vector, the packaging signals are a 5’ inverted terminal repeat (ITR) sequence and a 3’ ITR sequence. In certain embodiments, the term “transgene” is used interchangeably with “expression cassette”. In other embodiments, the term “transgene” refers solely to the coding sequence for a selected gene, e.g., “hSMNl”.
In certain embodiments, provided herein is recombinant nucleic acid molecule that includes an expression cassette comprising: (a) a UbC promoter, (b) a coding sequence for a functional SMN1 protein, (c) at least four miR182 target sequences, and (d) a polyA sequence, optionally with spacer sequences therebetween. In certain embodiments, the expression cassette comprises (a) a 5’ AAV ITR, (b) a UbC promoter, (c) a coding sequence for a functional SMN1 protein, (d) at least four miR182 target sequences, (e) a polyA sequence, and (f) a 3’ AAV ITR, optionally with spacer sequences therebetween. In certain embodiments, the one or more of the UbC promoter, SMN1 coding sequence, miR182 target
sequences, and polyA sequence are operably linked. In certain embodiments, the expression cassette includes additional regulatory elements (e.g., an intron sequence).
As used herein, the term “SMN1” or “hSMNl” includes any isoform of SMN1 that restores a desired function, reduces a symptom, or provides another desired physiologic result, when delivered a composition or method provided herein. The examples provided herein utilize the longest isoform, Isoform D, which is thought to be the predominant transcript produced by the gene is a patient unaffected by an SMN deficiency or defect. Isoform D provides a 294 amino acid protein [see, e.g., NCBI accession NM_000334/NP_000335; ENSEMBL ID ENST00000380707], the protein sequence is reproduced in SEQ ID NO: 5 and the coding sequence is reproduced in SEQ ID NO: 6. However, another isoform may be selected. For example, Isoform B has an alternate inframe exon in the 3’ coding sequence, resulting in a protein which is shorter in length (262 amino acids) than Isoform D, but having the same N- and C-termini as that isoform. See, NCBI Accession No. NM_022874/NP_075012; ENSEMBL ID ENST00000503079. Isoform A lacks the penultimate exon, which results in an alternate translation stop codon as compared to Isoform D. Thus, Isoform A is shorter (282 amino acids) and has a distinct C- terminus compared to Isoform D. See, NCBI Accession No.
NM_001297715/NP_001284644; ENSEMBL ID ENSTL00000506163.
A “vector” as used herein is a biological or chemical moiety comprising a nucleic acid sequence which can be introduced into an appropriate target cell for replication or expression of said nucleic acid sequence. Examples of a vector include but not limited to a recombinant virus, a plasmid, Lipoplexes, a Polymersome, Polyplexes, a dendrimer, a cell penetrating peptide (CPP) conjugate, a magnetic particle, or a nanoparticle. In certain embodiments, a vector is a nucleic acid molecule into which an engineered nucleic acid encoding a functional hSMNl may be inserted, which can then be introduced into an appropriate target cell. Such vectors preferably have one or more origin of replication, and one or more site into which the recombinant DNA can be inserted. Vectors often have means by which cells with vectors can be selected from those without, e.g., they encode drug resistance genes. Common vectors include plasmids, viral genomes, and “artificial chromosomes”. Conventional methods of generation, production, characterization or quantification of the vectors are available to one of skill in the art.
In certain embodiments, the vector is a nucleic acid comprising an expression cassette described herein (for example, “naked DNA”, “naked plasmid DNA”, RNA, and mRNA), which may be coupled with various compositions and nano particles, including, for examples, micelles, liposomes, cationic lipid - nucleic acid compositions, poly-glycan compositions and other polymers, lipid and/or cholesterol-based - nucleic acid conjugates) and other constructs such as are described herein. See, e.g., X. Su et al, Mol. Pharmaceutics, 2011, 8 (3), pp 774- 787; web publication: March 21, 2011; WO2013/182683, WO 2010/053572 and WO 2012/170930, all of which are incorporated herein by reference.
In certain embodiments, the vector described herein is a “replication-defective virus” or a “viral vector” which refers to a synthetic or artificial viral particle in which an expression cassette containing a nucleic acid sequence encoding hSMNl is packaged in a viral capsid or envelope, where any viral genomic sequences also packaged within the viral capsid or envelope are replication-deficient; i.e., they cannot generate progeny virions but retain the ability to infect target cells.
As used herein, a recombinant virus vector is an adeno-associated virus (AAV), an adenovirus, a bocavirus, a hybrid AAV/bocavirus, a herpes simplex virus, or a lentivirus.
In certain embodiments, a host cell having a nucleic acid including an SMN1 encoding sequence or an expression cassette that includes SMN1 encoding sequence is provided. In certain embodiments, the host cell contains a plasmid having an SMN1 encoding sequence or an expression cassette that includes SMN1 encoding sequence as described herein.
As used herein, the term “host cell” may refer to a packaging cell line in which a vector (e.g., a recombinant AAV) is produced. A host cell may be a prokaryotic or eukaryotic cell (e.g., human, insect, or yeast) that contains exogenous or heterologous DNA that has been introduced into the cell by any means, e.g., electroporation, calcium phosphate precipitation, microinjection, transformation, viral infection, transfection, liposome delivery, membrane fusion techniques, high velocity DNA-coated pellets, viral infection and protoplast fusion. Examples of host cells may include, but are not limited to an isolated cell, a cell culture, an Escherichia coli cell, a yeast cell, a human cell, a non-human cell, a mammalian cell, a non-mammalian cell, an insect cell, an HEK-293 cell, a liver cell, a kidney cell, a cell of the central nervous system, a neuron, a glial cell, or a stem cell.
As used herein, the term “target cell” refers to any cell in which expression of the functional hSMNl is desired. In certain embodiments, the term “target cell” is intended to reference a cell of a subject being treated for SMA that is intended to be transduced by an rAAV vector. Target cells include neuronal and non-neuronal cells. In certain embodiment, the target cell may be a central nervous system cell. In certain embodiments, the target cell is one or more of an excitatory neuron, an inhibitory neuron, a glial cell, a cortex cell, a frontal cortex cell, a cerebral cortex cell, a spinal cord cell. In certain embodiments, the target cell is in leptomeninges (LM) of the CNS. In certain embodiments, the target cell is in parenchyma of CNS. In certain embodiments, the target cells include one or more of muscle, liver, lung, airway epithelium, central nervous system, neurons, eye (ocular cells), or heart.
In certain embodiments, the compositions provided include an engineered human (h) survival of motor neuron (SMN) 1 encoding sequence. The native hSMNl encoding is provided in SEQ ID NO: 6, which encodes the amino acid sequence of SEQ ID NO: 5.
The hSMN 1 coding sequences utilized can be generated in vitro or synthetically, or by any other suitable method using techniques well known in the art. For example, the PCR- based accurate synthesis (PAS) of long DNA sequence method may be utilized, as described by Xiong et al, PCR-based accurate synthesis of long DNA sequences, Nature Protocols 1, 791 - 797 (2006). A method combining the dual asymmetrical PCR and overlap extension PCR methods is described by Young and Dong, Two-step total gene synthesis method, Nucleic Acids Res. 2004; 32(7): e59. See also, Gordeeva et al, J Microbiol Methods. Improved PCR-based gene synthesis method and its application to the Citrobacter freundii phytase gene codon modification. 2010 May;81(2): 147-52. Epub 2010 Mar 10; see, also, the following patents on oligonucleotide synthesis and gene synthesis, Gene Seq. 2012 Apr;6(l): 10-21; US 8008005; and US 7985565. Each of these documents is incorporated herein by reference. In addition, kits and protocols for generating DNA via PCR are available commercially. These include the use of polymerases including, without limitation, Taq polymerase; OneTaq® (New England Biolabs); Q5® High-Fidelity DNA Polymerase (New England Biolabs); and GoTaq® G2 Polymerase (Promega). DNA may also be generated from cells transfected with plasmids containing the hOTC sequences described herein. Kits and protocols are known and commercially available and include, without limitation, QIAGEN plasmid kits; Chargeswitch® Pro Filter Plasmid Kits (Invitrogen); and GenElute™ Plasmid Kits (Sigma Aldrich). Other techniques useful herein include sequence-
specific isothermal amplification methods that eliminate the need for thermocycling. Instead of heat, these methods typically employ a strand-displacing DNA polymerase, like Bst DNA Polymerase, Large Fragment (New England Biolabs), to separate duplex DNA. DNA may also be generated from RNA molecules through amplification via the use of Reverse Transcriptases (RT), which are RNA-dependent DNA Polymerases. RTs polymerize a strand of DNA that is complimentary to the original RNA template and is referred to as cDNA. This cDNA can then be further amplified through PCR or isothermal methods as outlined above. Custom DNA can also be generated commercially from companies including, without limitation, GenScript; GENEWIZ®; GeneArt® (Life Technologies); and Integrated DNA Technologies.
By “functional hSMNl”, is meant a gene which encodes an SMN protein which provides at least about 50%, at least about 75%, at least about 80%, at least about 90%, or about the same, or greater than 100% of the biological activity level of the native survival of motor neuron protein, or a natural variant or polymorph thereof which is not associated with disease. Additionally, SMN1 homologue- SMN2 also encodes the SMN protein, but processes the functional protein less efficiently. Based on the copy number of SMN2, subjects lacking a functional hSMNl gene demonstrate SMA to varying degrees. Thus, for some subjects, it may be desirable for the hSMNl protein to provide less than 100% of the biological activity of the native SMN1 protein. In certain embodiments, the terms “hSMNl”, “functional hSMNl”, and “SMN1” are used interchangeably.
A variety of assays exist for measuring SMN expression and activity levels in vitro. See, e.g., Tanguy et al, 2015, cited above. The methods described herein can also be combined with any other therapy for treatment of SMA or the symptoms thereof. In certain embodiments, the standard of care may include nusinersen, which is an SMN2 pre-messenger ribonucleic acid (mRNA)-targeting antisense oligonucleotide (ASO) accepted by the FDA and EMA [SPINRAZA™, Biogen], See, e.g., US Patent Nos. 6,166,197, US 6,210,892, US 7,101,993; US 7,838,657; US 8,110,560; US 8,361,977; US 8,980,853. This is an SMN2- directed antisense oligonucleotide which is administered intrathecally. The recommended dosage is 12 mg (5mL per administration). Treatment is initiated with 4 loading doses; the first three loading doses being administered at 14-day intervals, the 4th loading dose administered 30 days after the 3rd does, and a maintenance dose administered once every 4 months thereafter.
In certain embodiments, the amino acid sequence of the functional hSMNl is that of SEQ ID NO: 5. In certain embodiments the amino acid sequence of the functional hSMNl is SEQ ID NO: 5 or a sequence sharing at least 95%, 96%, 97%, 98%, or 99% identity with SEQ ID NO: 5. In certain embodiments, a modified hSMN coding sequence is provided. Preferably, the modified hSMN coding sequence has less than about 80% identity, preferably about 75% identity or less to the full-length native hSMN coding sequence (SEQ ID NO: 6). In certain embodiments, the modified hSMN coding sequence is characterized by improved translation rate as compared to native hSMN following AAV-mediated delivery. In certain embodiments, the modified hSMN coding sequence shares less than about 80%, 79%, 78%, 77%, 76%, 75%, 74%, 73%, 72%, 71%, 70%, 69%, 68%, 67%, 66%, 65%, 64%, 63%, 62%, or 61% identity to the full length native hSMNl coding sequence. In certain embodiment, the modified hSMNl coding sequence comprises SEQ ID NO: 7, or a sequence sharing at least 70%, 75%, 80%, 85%, 90%, 95% or greater identity with SEQ ID NO: 7. In certain embodiment, the modified hSMNl coding sequence is a sequence sharing at least 70%, 75%, 80%, 85%, 90%, 95% or greater identity with SEQ ID NO: 7 that encodes the amino acid sequence of SEQ ID NO: 5. In certain embodiments, the modified hSMNl coding sequence is a sequence having up to 1, up to 2, up to 3, up to 4, up to 5, up to 6, up to 7, up to 8, up to 9, or up to 10 basepair mismatches when aligned to the nucleotide sequence of SEQ ID NO: 7, and expresses a functional SMN1, In other embodiments, a different SMN1 coding sequence is selected.
In certain embodiments, provided herein is a recombinant nucleic acid molecule that includes an expression cassette, wherein the expression cassette has the nucleic acid sequence of SEQ ID NO: 16, or a nucleic acid sequence at least 99% identical to SEQ ID NO: 16. In certain embodiments, the expression cassette is flanked by a AAV 5’ ITR sequence and a AAV 3’ ITR sequence, optionally with additional sequences between the expression cassette and ITR sequences. In certain embodiments, provided herein is a recombinant nucleic acid molecule that includes the nucleotide sequence of SEQ ID NO: 2 or a nucleotide sequence at least 99% identical to SEQ ID NO: 2.
The term “percent (%) identity”, “sequence identity”, “percent sequence identity”, or “percent identical” in the context of nucleic acid sequences refers to the residues in the two sequences which are the same when aligned for correspondence. The length of sequence identity comparison may be over the full-length of the genome, the full-length of a gene
coding sequence, or a fragment of at least about 500 to 5000 nucleotides, is desired. However, identity among smaller fragments, e.g. of at least about nine nucleotides, usually at least about 20 to 24 nucleotides, at least about 28 to 32 nucleotides, at least about 36 or more nucleotides, may also be desired.
Percent identity may be readily determined for amino acid sequences over the full- length of a protein, polypeptide, about 32 amino acids, about 330 amino acids, or a peptide fragment thereof or the corresponding nucleic acid sequence coding sequences. A suitable amino acid fragment may be at least about 8 amino acids in length, and may be up to about 700 amino acids. Generally, when referring to “identity”, “homology”, or “similarity” between two different sequences, “identity”, “homology” or “similarity” is determined in reference to “aligned” sequences. “Aligned” sequences or “alignments” refer to multiple nucleic acid sequences or protein (amino acids) sequences, often containing corrections for missing or additional bases or amino acids as compared to a reference sequence.
Identity may be determined by preparing an alignment of the sequences and through the use of a variety of algorithms and/or computer programs known in the art or commercially available (e.g., BLAST, ExPASy; Clustal Omega; FASTA; using, e.g., Needleman-Wunsch algorithm, Smith -Waterman algorithm). Alignments are performed using any of a variety of publicly or commercially available Multiple Sequence Alignment Programs. Sequence alignment programs are available for amino acid sequences, e.g., the “Clustal Omega”, “Clustal X”, “MUSCLE”, “MAP”, “PIMA”, “MSA”, “BLOCKMAKER”, “MEME”, and “Match-Box” programs. Generally, any of these programs are used at default settings, although one of skill in the art can alter these settings as needed. Alternatively, one of skill in the art can utilize another algorithm or computer program which provides at least the level of identity or alignment as that provided by the referenced algorithms and programs. See, e.g., J. D. Thompson et al, Nucl. Acids. Res., “A comprehensive comparison of multiple sequence alignments”, 27(13):2682-2690 (1999).
Multiple sequence alignment programs are also available for nucleic acid sequences. Examples of such programs include, “Clustal Omega”, “Clustal W”, “MUSCLE”, “CAP Sequence Assembly”, “BLAST”, “MAP”, and “MEME”, which are accessible through Web Servers on the internet. Other sources for such programs are known to those of skill in the art. Alternatively, Vector NTI utilities are also used. There are also a number of algorithms known in the art that can be used to measure nucleotide sequence identity, including those
contained in the programs described above. As another example, polynucleotide sequences can be compared using Fasta™, a program in GCG Version 10.1. Fasta™ provides alignments and percent sequence identity of the regions of the best overlap between the query and search sequences. For instance, percent sequence identity between nucleic acid sequences can be determined using Fasta™ with its default parameters (a word size of 6 and the NOPAM factor for the scoring matrix) as provided in GCG Version 10.1, herein incorporated by reference.
As used herein, “operably linked” sequences include both regulatory sequences that are contiguous or non-contiguous with the nucleic acid sequence and regulatory sequences that act in cis or trans with nucleic acid sequence. Such regulatory sequences typically include, e.g., one or more of a promoter, an enhancer, an intron, a Kozak sequence, a polyadenylation sequence, and a TATA signal. The expression cassette may contain regulatory sequences upstream (5’ to) of the gene sequence, e.g., one or more of a promoter, an enhancer, an intron, etc., and one or more of an enhancer, or regulatory sequences downstream (3’ to) a gene sequence, e.g., 3’ untranslated region (3’ UTR) comprising a polyadenylation site, among other elements. In certain embodiments, the regulatory sequences are operably linked to the nucleic acid sequence of a gene product, wherein the regulatory sequences are separated from the nucleic acid sequence encoding the gene product by intervening nucleic acid sequences, i.e., 5 ’-untranslated regions (5’ UTR). In certain embodiments, the expression cassette comprises nucleic acid sequence of one or more of gene products. In some embodiments, the expression cassette can be a monocistronic or a bicistronic expression cassette. In other embodiments, the term “transgene” refers to one or more DNA sequences from an exogenous source which are inserted into a target cell. Typically, such an expression cassette for generating a viral vector contains the coding sequence for the gene product described herein flanked by packaging signals of the viral genome and other expression control sequences such as those described herein. In certain embodiments, a vector genome may contain two or more expression cassettes.
The term “exogenous” as used to describe a nucleic acid sequence or protein means that the nucleic acid or protein does not naturally occur in the position in which it exists in a chromosome, or host cell. An exogenous nucleic acid sequence also refers to a sequence derived from and inserted into the same host cell or subject, but which is present in a non-
natural state, e.g., a different copy number, or under the control of different regulatory elements.
The term “heterologous” as used to describe a nucleic acid sequence or protein means that the nucleic acid or protein was derived from a different organism or a different species of the same organism than the host cell or subject in which it is expressed. The term “heterologous” when used with reference to a protein or a nucleic acid in a plasmid, expression cassette, or vector, indicates that the protein or the nucleic acid is present with another sequence or subsequence which with which the protein or nucleic acid in question is not found in the same relationship to each other in nature.
As used herein, “encoded amino acid sequence” refers to the amino acid which is predicted based on the translation of a known DNA codon of a referenced nucleic acid sequence being translated to an amino acid. The following table illustrates DNA codons and twenty common amino acids, showing both the single letter code (SLC) and three letter code (3LC).
In certain embodiments, the promoter is a chicken P-actin (also referred to as chicken beta-actin, CB or CBA) promoter. In further embodiments, the promoter is a CB7 promoter (also referred to as hybrid CB7) comprising a cytomegalovirus immediate-early (CMV IE) enhancer and the chicken P-actin promoter, optionally with spacer sequence, optionally with a chimeric intron comprising chicken beta actin intron and further comprising a chicken betaactin splicing donor (including the exon sequence, chicken beta actin intron) and rabbit betaglobin splicing acceptor. See, e.g., cytomegalovirus (CMV) immediate early enhancer (260 bp, C4; GenBank # K03104.1). Chicken beta-actin promoter (281 bp; CB; GenBank # X00182.1).
In other embodiments, the promoter is a ubiquitin C (UbC) promoter. In certain embodiments, the UbC promoter is a human UbC promoter. See, e.g., WO 2001/091800. See, e.g., GenBank accession numbers AF232305 (rat), D63791 (human), NCBI Reference Sequence: NG_027722.2, and Schorpp et al. Nucleic Acids Res. 1996 May 1 ;24(9): 1787-8. In certain embodiments, the UbC promoter comprises the nucleotide sequence of SEQ ID NO: 10. In certain embodiments, the UbC promoter comprises the nucleotide sequence of SEQ ID NO: 17. In certain embodiments, the UbC promoter comprises a nucleotide sequence having up to 1, up to 2, up to 3, up to 4, up to 5, up to 6, up to 7, up to 8, up to 9, or up to 10 basepair mismatches when aligned to the nucleotide sequence of either or both of SEQ ID
NOs: 10 and 17, and, when operably linked to a transgene (i.e., SMN1), is capable of expressing the transgene in a target cell.
In certain embodiments, still other promoters and/or enhancers may be selected. In another embodiment, a suitable promoter may include without limitation, an elongation factor 1 alpha (EFl alpha) promoter (see, e.g., Kim DW et al, Use of the human elongation factor 1 alpha promoter as a versatile and efficient expression system. Gene. 1990 Jul 16;91(2):217- 23), a human Synapsin 1 (hSyn) promoter (see, e.g., Kugler S et al, Human synapsin 1 gene promoter confers highly neuron-specific long-term transgene expression from an adenoviral vector in the adult rat brain depending on the transduced area. Gene Ther. 2003 Feb;10(4):337-47), a neuron-specific enolase (NSE) promoter (see, e.g., Kim J et al, Involvement of cholesterol-rich lipid rafts in interleukin-6-induced neuroendocrine differentiation of LNCaP prostate cancer cells. Endocrinology. 2004 Feb;145(2):613-9. Epub 2003 Oct 16), or a CB6 promoter (see, e.g., Large-Scale Production of Adeno- Associated Viral Vector Serotype-9 Carrying the Human Survival Motor Neuron Gene, Mol Biotechnol. 2016 Jan;58(l):30-6. doi: 10.1007/sl2033-015-9899-5). In certain embodiments, the promoter is a JeT promoter (Tornoe J et al. Gene. 2002 Sep 4;297(l-2):21-32). In still other embodiments, multiple enhancers and/or promoters may be included.
In certain embodiments, an additional or alternative promoter sequence may be included as part of the expression control sequences (regulatory sequences). Constitutive promoters, regulatable promoters [see, e.g., WO 2011/126808 and WO 2013/04943], tissue specific promoters, or a promoter responsive to physiologic cues may be utilized in the vectors described herein. The promoter(s) can be selected from different sources, e.g., human cytomegalovirus immediate-early (CMV IE) enhancer/promoter, the SV40 early enhancer/promoter, the JC polymovirus promoter, myelin basic protein (MBP) or glial fibrillary acidic protein (GFAP) promoters, herpes simplex virus (HSV-1) latency associated promoter (LAP), rouse sarcoma virus (RSV) long terminal repeat (LTR) promoter, neuronspecific promoter (NSE), platelet derived growth factor (PDGF) promoter, melaninconcentrating hormone (MCH) promoter, CBA, or matrix metalloprotein promoter (MPP), and the chicken beta-actin promoter. In certain embodiments, the expression cassette is designed for expression of the SMN1 in the central nervous system (CNS), including the cerebral spinal fluid and brain. In a further embodiment, the expression cassette is useful for expression in both the CNS and systemically.
In addition to a promoter, a vector may contain one or more other appropriate transcription initiation sequences, transcription termination sequences, enhancer sequences, efficient RNA processing signals such as splicing and polyadenylation (poly A) signals; sequences that stabilize cytoplasmic mRNA for example WPRE; sequences that enhance translation efficiency (i.e., Kozak consensus sequence); sequences that enhance protein stability; and when desired, sequences that enhance secretion of the encoded product.
In certain embodiments, the regulatory sequences comprise an enhancer. In certain embodiments, the regulatory sequences comprise one enhancer. In another embodiment, the regulatory sequences contain two or more enhancers. These enhancers may be the same or may be different. For example, an enhancer may include an alpha mic/bik enhancer or a CMV IE enhancer. The enhancer may be present in two copies which are located adjacent to one another in an expression cassette. Alternatively, the dual copies of the enhancer may be separated by one or more sequences.
In certain embodiments, the regulatory sequences comprise an intron. In certain embodiments, the intron is a chicken beta-actin intron. In one embodiment, the intron is 875 bp (GenBank # X00182.1). In certain embodiments, the intron is 973 bp intron from the chicken beta actin gene (GenBank # X00182.1). In certain embodiments, the intron is a chimeric intron (CI) - a hybrid intron consisting of a human beta-globin splice donor and immunoglobulin G (IgG) splice acceptor elements. In certain embodiments, the intron is a chimeric intron comprising a chicken beta-actin splicing donor (including the exon sequence), chicken beta actin intron, and rabbit beta globin splicing acceptor. In certain embodiments, the intron includes the nucleic acid sequence of SEQ ID NO: 11. In certain embodiments, the intron comprises a nucleotide sequence having up to 1, up to 2, up to 3, up to 4, up to 5, up to 6, up to 7, up to 8, up to 9, or up to 10 basepair mismatches when aligned to the nucleotide sequence of SEQ ID NO: 11. Other suitable introns include those known in the art may by a human P-globulin intron, and/or a commercially available intron, and those described in WO 2011/126808.
In certain embodiments, the regulatory sequences comprise a polyadenylation signal (poly A). Examples of suitable polyA sequences include, e.g., rabbit beta globin (RBG or rBG) poly A, SV40, SV50, bovine growth hormone (bGH), human growth hormone, and synthetic poly As. Optionally, one or more sequences may be selected to stabilize mRNA. In certain embodiments, the polyA is a rabbit beta globin poly A (rabbit globin polyA or rBG).
See, e.g., WO 2014/151341. In certain embodiments, the rBG polyA comprises SEQ ID NO: 13. In certain embodiments, a human growth hormone (hGH) polyadenylation sequence, an SV40 polyA, or a synthetic polyA may be included in an expression cassette. In certain embodiments, the SV40 polyA is selected.
In certain embodiments, the expression cassette includes one or more miRNA (also referred to as miR or micro-RNA) target sequences in the untranslated region(s). The miRNA target sequences are designed to be specifically recognized by miRNA present in cells in which transgene expression is undesirable and/or reduced levels of transgene expression are desired. In certain embodiments, the expression cassette includes miRNA target sequences that specifically reduce expression of SMN1 in dorsal root ganglion. In certain embodiments, the miRNA target sequences are located in the 3’ UTR, 5’ UTR, and/or in both 3’ and 5’ UTR of an expression cassette. In certain embodiments, the expression cassette comprises at least two, at least three, or at least four tandem repeats of dorsal root ganglion (DRG)-specific miRNA target sequences, wherein the tandem repeats comprise miRNA target sequences that may be the same or different. In certain embodiments, the start of the first of the DRG- specific miRNA tandem repeats is within 20 nucleotides from the 3’ end of the SMN1 encoding sequence. In certain embodiments, the start of the first of DRG-specific miRNA tandem repeats is at least 100 nucleotides from the 3’ end of the SMN1 encoding sequence. In certain embodiments, the miRNA tandem repeats comprise 200 to 1200 nucleotides in length. In certain embodiment, the inclusion of miR targets does not modify the expression or efficacy of the therapeutic transgene in one or more target tissues, relative to the expression cassette lacking the miR target sequences.
In certain embodiments, the expression cassette contains at least one, two, three, or four miRNA target sequences that are miR-182 target sequences. In certain embodiments, the expression cassette contains miR-182 target sequences that include the nucleotide sequence AGTGTGAGTTCTACCATTGCCAAA (SEQ ID NO: 12). In certain embodiments, the expression cassette contains more than one copy (e.g., two, three, or four copies) of a sequence that is 100% complementary to the miR-182 seed sequence. In certain embodiments, a miR-182 target sequence is about 7 nucleotides to about 28 nucleotides in length and includes at least one region that is at least 100% complementary to the miR-182 seed sequence. In certain embodiments, a miR-182 target sequence contains a sequence with partial complementarity to SEQ ID NO: 12 and, thus, when aligned to SEQ ID NO: 12, there
are one or more basepair mismatches. In certain embodiments, a miR-182 target sequence comprises a sequence having at least 1, 2, 3, 4, 5, 6, 7, 8, 9, or 10 basepair mismatches when aligned to SEQ ID NO: 12, where the basepair mismatches may be non-contiguous. In certain embodiments, a miR-182 target sequence includes a region of 100% complementarity which also comprises at least 30% of the length of the miR-182 target sequence. In certain embodiments, the region of 100% complementarity includes a sequence with 100% complementarity to the miR-182 seed sequence. In certain embodiments, the remainder of a miR-182 target sequence has at least about 80% to about 99% complementarity to miR-182. In certain embodiments, the expression cassette includes a miR-182 target sequence that comprises a truncated SEQ ID NO: 12, i.e., a sequence that lacks at least 1, 2, 3, 4, 5, 6, 7, 8, 9, or 10 nucleotides at either or both the 5’ or 3’ ends of SEQ ID NO: 12.
The term “tandem repeats” is used herein to refer to the presence of two, three, four, or more consecutive miRNA target sequences. These miRNA target sequences may be continuous, i.e., located directly after one another such that the 3’ end of one is directly upstream of the 5’ end of the next with no intervening sequences, or vice versa. In another embodiments, two, three, four, or more of the miRNA target sequences are separated from each other by spacer sequence. In certain embodiments, the expression cassette includes one or more mirl82 target sequence located in a sequence that encodes a 5’ UTR and one or more mirl82 target sequence located in a sequence that encodes a 3’ UTR. In certain embodiments, the expression cassette provided includes at least four miR182 target sequences that are located 3’ of the SMN1 coding sequence and 5’ of the polyA sequence. In other embodiments, the expression cassette includes at least four miR182 target sequences, wherein at least one target sequence is located 5’ of the UbC promoter and at least one target sequence is located 3’ of the SMN1 coding sequence and 5’ of the polyA sequence.
As used herein, as “spacer” is any selected nucleic acid sequence, e.g., of 1, 2, 3, 4, 5, 6, 7, 8, 9, or 10 nucleotides in length that is located between two or more consecutive miRNA target sequences. In certain embodiments, the spacer is 1 to 8 nucleotides in length, 2 to 7 nucleotides in length, 3 to 6 nucleotides in length, 4 nucleotides in length, 4 to 9 nucleotides, 3 to 7 nucleotides, or values which are longer. Suitably, a spacer is a non-coding sequence. In certain embodiments, the spacer may be of 4 nucleotides. In certain embodiments, the spacer is GGAT. In certain embodiments, the spacer is six 6 nucleotides. In certain embodiments, the spacer is CACGTG or GCATGC.
In certain embodiments, the tandem repeats contain two, three, four or more of the same miRNA target sequence. In certain embodiments, the tandem repeats contain at least two different miRNA target sequences, at least three different miRNA target sequences, or at least four different miRNA target sequences, etc. In certain embodiments, the tandem repeats may contain two or three of the same miRNA target sequence and a fourth miRNA target sequence which is different.
In certain embodiments, there may be at least two different sets of tandem repeats in the expression cassette. For example, a 3’ UTR may contain a tandem repeat immediately downstream of the transgene, UTR sequences, and two or more tandem repeats closer to the 3’ end of the UTR. In another example, the 5’ UTR may contain one, two or more miRNA target sequences. In another example the 3’ UTR may contain tandem repeats and the 5’ UTR may contain at least one miRNA target sequence.
In certain embodiments, the expression cassette contains two, three, four, or more tandem repeats that start within about 0 to 20 nucleotides of the stop codon for the SMN1 transgene. In other embodiments, the expression cassette contains the miRNA tandem repeats at least 100 to about 4000 nucleotides from the stop codon for the SMN1 transgene.
See also WO 2020/132455 Al and WO 2021/231579 Al, which are incorporated by reference herein in their entireties.
Recombinant Adeno-Associated Virus (rAAV)
Provided herein are recombinant adeno-associated virus (rAAV) vector having a capsid that contains an expression cassette that includes a nucleotide sequence encoding hSMNl. The rAAV vectors are useful for treating SMN1. Suitably, the AAV capsid selected targets the cells to be treated.
As used herein, “rAAV.hSMNl” refers to an rAAV having a vector genome that includes an hSMNl coding sequence. A “rAAVhu68. hSMNl” refers to an rAAV having an AAVhu68 capsid and a vector genome that includes an hSMNl coding sequence. The vector genome comprises an AAV 5’ inverted terminal repeat (ITR) sequence, an expression cassette comprising an SMN1 coding sequence that is operably linked to regulatory sequences, and an AAV 3 ’ ITR.
As used herein, the term “vector genome” refers to a nucleic acid molecule which is packaged in a viral capsid, for example, an AAV capsid, and is capable of being delivered to
a host cell or a cell in a patient. In certain embodiments, the vector genome comprises terminal repeat sequences (e.g., AAV inverted terminal repeat sequences (ITRs) necessary for packaging the vector genome into the capsid at the extreme 5’ and 3’ end and containing therebetween an expression cassette that includes an SMN1 encoding sequence as described herein operably linked to sequences which direct expression thereof.
AAV sequences present in a vector genome typically comprise the cis-acting 5’ and 3’ inverted terminal repeat (ITR) sequences (See, e.g., B. J. Carter, in “Handbook of Parvoviruses”, ed., P. Tijsser, CRC Press, pp. 155 168 (1990)). The ITR sequences are about 145 base pairs (bp) in length. Preferably, substantially the entire sequences encoding the ITRs are used in the molecule, although some degree of minor modification of these sequences is permissible. The ability to modify these ITR sequences is within the skill of the art. (See, e.g., texts such as Sambrook et al, “Molecular Cloning. A Laboratory Manual”, 2d ed., Cold Spring Harbor Laboratory, New York (1989); and K. Fisher et al., J. Virol., 70:520 532 (1996)). An example of such a molecule employed in the present invention is a “cis-acting” plasmid containing the transgene, in which the selected transgene sequence and associated regulatory elements are flanked by the 5’ and 3’ AAV ITR sequences. In one embodiment, the ITRs are from an AAV different than that supplying a capsid. In one embodiment, the ITR sequences are from AAV2. A shortened version of the 5’ ITR, termed AITR, has been described in which the D-sequence and terminal resolution site (trs) are deleted. In certain embodiments, the vector genome (e.g., of a plasmid) includes a shortened AAV2 ITR of 130 base pairs, wherein the external A elements is deleted. The shortened ITR may revert back to the wild-type length of 145 base pairs during vector DNA amplification using the internal A element as a template and packaging into the capsid to form the viral particle. In other embodiments, the full-length AAV 5’ and 3’ ITRs are used. However, ITRs from other AAV sources may be selected. Where the source of the ITRs is from AAV2 and the AAV capsid is from another AAV source, the resulting vector may be termed pseudotyped. However, other configurations of these elements may be suitable.
The term “AAV” as used herein refers to naturally occurring adeno-associated viruses, adeno-associated viruses available to one of skill in the art and/or in light of the composition(s) and method(s) described herein, as well as artificial AAVs. An adeno- associated virus (AAV) viral vector is an AAV DNase-resistant particle having an AAV protein capsid into which is packaged expression cassette flanked by AAV inverted terminal
repeat sequences (ITRs) for delivery to target cells. An AAV capsid is composed of 60 capsid (cap) protein subunits, VP1, VP2, and VP3, that are arranged in an icosahedral symmetry in a ratio of approximately 1 : 1 : 10 to 1 : 1 :20, depending upon the selected AAV. Various AAVs may be selected as sources for capsids of AAV viral vectors as identified above. See, e.g., US Published Patent Application No. 2007-0036760-Al; US Published Patent Application No. 2009-0197338-Al; EP 1310571. See also, WO 2003/042397 (AAV7 and other simian AAV), US Patent 7790449 and US Patent 7282199 (AAV8), WO 2005/033321 and US 7,906,111 (AAV9), and WO 2006/110689, and WO 2003/042397 (rh.10). These documents also describe other AAV which may be selected for generating AAV and are incorporated by reference. Among the AAVs isolated or engineered from human or non-human primates (NHP) and well characterized, human AAV2 is the first AAV that was developed as a gene transfer vector; it has been widely used for efficient gene transfer experiments in different target tissues and animal models. Unless otherwise specified, the AAV capsid, ITRs, and other selected AAV components described herein, may be readily selected from among any AAV, including, without limitation, the AAVs commonly identified as AAV1, AAV2, AAV3, AAV4, AAV5, AAV6, AAV7, AAV8, AAV9, AAV8bp, AAV7M8 and AAVAnc80, AAVhu68, and variants of any of the known or mentioned AAVs or AAVs yet to be discovered or variants or mixtures thereof. An AAV9 capsid includes an rAAV having capsid proteins comprising an amino acid sequence which is 99% identical to AAS99264. See, also US7906111 and WO 2005/033321. Additional capsids are provided in WO 2022/119871 and WO 2022/226263, which are incorporated herein by reference. See also PCT/US 19/19804 and PCT/US 19/19861, each entitled “Novel Adeno- Associated Virus (AAV) Vectors, AAV Vectors Having Reduced Capsid Deamidation and Uses Therefor” and filed Feb 27, 2019, PCT/US2024/036237, filed June 28, 2024, entitled “Mutant AAV with Central Nervous System Targeting Motifs and Compositions Containing Same,” and US 2024-0024507 Al, “Novel Compositions with Tissue-Specific Targeting Motifs And Compositions Containing Same,” which are incorporated by reference herein in their entireties. In certain embodiments, the capsid protein is designated by a number or a combination of numbers and letters following the term “AAV” in the name of the rAAV vector.
In certain embodiments, a recombinant AAV is provided having a vector genome that includes an expression cassette comprising: (a) a UbC promoter, (b) a coding sequence for a
functional SMN1 protein, (c) at least four miR182 target sequences, and (d) a polyA sequence, optionally with spacer sequences therebetween. The coding sequence, the at least four miR182 target sequences, and the polyA sequence are operably linked to the UbC promoter. In certain embodiments, the expression includes additional regulatory elements (e.g., an intron). In certain embodiments, the polyA sequence is a rabbit beta-globin polyA sequence. In certain embodiments, the vector genome includes an expression cassette having the nucleic acid sequence of SEQ ID NO: 16, or a nucleic acid sequence at least 99% identical to SEQ ID NO: 16. In certain embodiments, the vector genome has the nucleotide sequence of SEQ ID NO: 2 or a nucleotide sequence at least 99% identical to SEQ ID NO: 2.
In certain embodiments, the AAV capsid is a Clade F capsid, such as AAV9 capsid, AAVhu68 capsid, hu31 capsid, hu32 capsid, or a variation thereof. See, e.g., WO 2005/033321 published April 14, 2015, WO 2018/160582, and US 2015/0079038, each of which is incorporated herein by reference in its entirety. In certain embodiments, the AAV capsid is a non-clade F capsid, for example a Clade A, B, C, D, or E capsid. In certain embodiment, the non-Clade F capsid is an AAV1 or a variation thereof. In certain embodiment, the AAV capsid transduces a target cell other than the nervous system cells. In certain embodiments, the AAV capsid is a Clade A capsid (e.g., AAV1, AAV6, AAVrh91), a Clade B capsid (e.g., AAV 2), a Clade C capsid (e.g., hu53), a Clade D capsid (e.g., AAV7), or a Clade E capsid (e.g., rhlO).
In certain embodiments, the Clade F AAV capsid is an AAVhu68 capsid [See, e.g., US2020/0056159; PCT/US21/55436; SEQ ID NO: 8 for nucleic acid sequence; SEQ ID NO: 9 for amino acid sequence], an AAVhu95 capsid [See, e.g., US Provisional Application No. 63/251,599, filed October 2, 2201, International Patent Application No. PCT/US2022/077315, filed September 30, 2022]; an AAVhu96 capsid [See, e.g., US Provisional Application No. 63/251,599, filed October 2, 2201, and International Patent Application No. PCT/US2022/077315, filed September 30, 2022]; or an AAV9 capsid. In certain embodiments, the AAV capsid is a Clade A capsid, such as AAVrh91 capsid. See, PCT/US20/030266, filed April 29, 2020, now published WO2020/223231, and International Application No. PCT/US21/45945, filed August 13, 2021, which are incorporated herein by reference.
In certain embodiments, the AAV capsid for the compositions and methods described herein is chosen based on the target cell. In certain embodiment, the AAV capsid transduces a
CNS cell and/or a PNS cell. In certain embodiments, another AAV capsid may be chosen, the AAV capsid is selected from a cy02 capsid, a rh43 capsid, an AAV8 capsid, a rhOl capsid, an AAV9 capsid, an rh8 capsid, a rhlO capsid, a bbOl capsid, a hu37 capsid, a rh02 capsid, a rh20 capsid, a rh39 capsid, a rh64 capsid, an AAV6 capsid, an AAV1 capsid, a hu44 capsid, a hu48 capsid, a cy05 capsid a hul 1 capsid, a hu32 capsid, a pi2 capsid, or a variation thereof.
An AAV capsid is an assembly of a heterogeneous population of vpl, a heterogeneous population of vp2, and a heterogeneous population of vp3 proteins. As used herein when used to refer to vp capsid proteins, the term “heterogeneous” or any grammatical variation thereof, refers to a population consisting of elements that are not the same, for example, having vpl, vp2 or vp3 monomers (proteins) with different modified amino acid sequences.
As used herein when used to refer to vp capsid proteins, the term “heterogeneous” or any grammatical variation thereof, refers to a population consisting of elements that are not the same, for example, having vpl, vp2, or vp3 (also referred to as VP1, VP2, VP3, or Vpl, Vp2, Vp3) monomers (proteins) with different modified amino acid sequences. The term “heterogeneous population” as used in connection with vpl, vp2 and vp3 proteins (alternatively termed isoforms), refers to differences in the amino acid sequence of the vpl, vp2 and vp3 proteins within a capsid. The AAV capsid contains subpopulations within the vpl proteins, within the vp2 proteins and within the vp3 proteins which have modifications from the predicted amino acid residues. These subpopulations include, at a minimum, certain deamidated asparagine (N or Asn) residues. For example, certain subpopulations comprise at least one, two, three or four highly deamidated asparagines (N) positions in asparagine - glycine pairs and optionally further comprising other deamidated amino acids, wherein the deamidation results in an amino acid change and other optional modifications.
In certain embodiments, AAV capsids are provided which have a heterogeneous population of AAV capsid isoforms (i.e., VP1, VP2, VP3) which contain multiple highly deamidated “NG” positions. In certain embodiments, the highly deamidated positions are in the locations identified below, with reference to the predicted full-length VP1 amino acid sequence. In other embodiments, the capsid gene is modified such that the referenced “NG” is ablated and a mutant “NG” is engineered into another position.
In certain embodiments, the rAAV provided herein is has an AAVhu68 capsid that includes AAVhu68 VP1 proteins, AAVhu68 VP2 proteins, and AAVhu68 VP3 proteins expressed from a nucleic acid sequence encoding SEQ ID NO: 9 (amino acids 1 to 736).
In certain embodiments, the AAV hu68 capsid comprises a heterogenous population of AAVhu68 vpl proteins, a heterogenous population of AAVhu68 vp2 proteins; and a heterogenous population of AAVhu68 vp3 proteins comprising amino acid residues 1 to 736 (vpl), amino acid residues 138 to 736 (vp2), and amino acid residues 203 to 736 (vp3) of SEQ ID NO: 9, respectively, with amino acid modifications, wherein the heterogenous AAVhu68 vpl proteins, AAVhu68 vp2 proteins and AAVhu68 vp3 proteins contain amino acid modifications comprising 50% to 100% deamidation in at least two asparagines (N) in asparagine - glycine pairs in SEQ ID NO: 9 in two or more of asparagines (N) at positions N57, N329, N452, and/or N512 based on the numbering of the encoded AAVhu68 VP1 amino acid sequence (SEQ ID NO: 9) as determined using mass spectrometry and optionally further comprising subpopulations comprising other deamidated amino acids, wherein the deamidation results in an amino acid change, wherein the deamidated asparagines are deamidated to aspartic acid, isoaspartic acid, an interconverting aspartic acid/isoaspartic acid pair, or combinations thereof. In certain embodiments, the subpopulations of AAVhu68 VP1 proteins, AAVhu68 VP2 proteins and AAVhu68VP3 proteins further comprise one or more of: (a) at least 65% of asparagines (N) in position N57 of the vpl proteins are deamidated, based on the numbering of SEQ ID NO: 9; (b) at least 75% of N in position N329 of the vpl, v2 and vp3 proteins are deamidated, based on the residue numbering of the amino acid sequence of SEQ ID NO: 9, (c) at least 50% of N in position N452 of the vpl, v2 and vp3 proteins are deamidated, based on the residue numbering of the amino acid sequence of SEQ ID NO: 9; and/or (d) at least 75% of N in N512 of the vpl, v2 and vp3 proteins are deamidated, based on the residue numbering of the amino acid sequence of SEQ ID NO: 9, wherein the heterogenous population of AAVhu68 vpl proteins comprise glutamic acid at position 67 based on the numbering of SEQ ID NO: 9 and the heterogenous population of AAVhu68vpl and AAVhu68 vp2 proteins comprising valine at position 157, based on the numbering of the vpl capsid of SEQ ID NO: 9. In certain embodiments, the rAAVhu68 capsid comprises a subpopulation of vpl in which 75% to 100 % of the N at position 57 of the vpl proteins, based on the numbering of SEQ ID NO: 9, are deamidated as determined using mass spectrometry. In certain embodiments, the rAAVhu68 capsid comprises
subpopulation of vpl proteins, vp2 proteins, and/or vp3 proteins in which 75% to 100% of the N at position 329, based on the numbering of SEQ ID NO: 9, are deamidated as determined using mass spectrometry. In certain embodiments, the rAAVhu68 capsid comprises subpopulation of vpl proteins, vp2 proteins, and/or vp3 proteins in which 75% to 100% of the N at position 452, based on the numbering of SEQ ID NO: 9, are deamidated as determined using mass spectrometry. In certain embodiments, the rAAVhu68 capsid comprises subpopulation of vpl proteins, vp2 proteins, and/or vp3 proteins in which 75% to 100% of the N at position 512, based on the numbering of SEQ ID NO: 9, are deamidated. In certain embodiments, the nucleic acid sequence encoding the proteins is SEQ ID NO: 9, or a sequence at least 80% to at least 99% identical to SEQ ID NO: 8 which encodes the amino acid sequence of SEQ ID NO: 9. In certain embodiments, the nucleic acid sequence is at least 80% to 97% identical to SEQ ID NO: 8. In certain embodiments, the rAAVhu68 capsid comprises subpopulations with 50% to 100% deamidation in each of N57, N329, N452, and N512, and optionally further comprise subpopulations of vpl, vp2 and/or vp3 proteins which further comprise 1% to about 40% deamidation in at least one or more of positions N94, N113, N252, N253, Q258, N270, N303, N304, N305, N319, N328, N336, N409, N410, N477, N515, N598, Q599, N628, N651, N663, N709, or combinations thereof, based on the amino acid numbering of SEQ ID NO: 9. In certain embodiments, the rAAVhu68 capsid vpl, vp2 and/or vp3 proteins further comprise one or more modifications selected from one or more modification in one or more of the following: acetylated lysine, phosphorylated serine and/or threonine, isomerized aspartic acid, oxidized tryptophan and/or methionine, or an amidated amino acid.
In certain embodiments, the AAVhu68 capsid comprises: a heterogenous population of AAVhu68 vpl, AAVhu68vp2 and AAVhu68vp3 proteins produced by expression from a nucleic acid molecule having a nucleic acid sequence encoding the amino acid sequence of 1 to 736 of SEQ ID NO: 9, wherein the heterogenous population of AAVhu68 vpl proteins, AAVhu68 vp2 proteins, and AAVhu68 vp3 proteins comprise amino acids 1 to 736 (vpl), amino acids 138 to 736 (vp2), and amino acids 203 to 736 (vp3) of SEQ ID NO: 9, respectively, with amino acid modifications comprising 50% to 100% deamidation in at least two asparagines (N)-glycine pairs in two or more of N57, N329, N452, and/or N512 of SEQ ID NO: 9 as determined using mass spectrometry and optionally further comprising subpopulations comprising other deamidated amino acids; wherein the heterogenous
population of AAVhu68 vpl proteins comprise glutamic acid at position 67 based on the numbering of SEQ ID NO: 9 and the heterogenous population of AAVhu68vpl and AAVhu68 vp2 proteins comprising valine at position 157, based on the numbering of the vpl capsid of SEQ ID NO: 9. In certain embodiments, the VP1 proteins the nucleic acid sequence is SEQ ID NO: 8 or a sequence at least 70% identical to SEQ ID NO: 8 which encodes the amino acid sequence of SEQ ID NO: 9. In certain embodiments, the nucleic acid sequence is SEQ ID NO: 8. In certain embodiments, the rAAVhu68 capsid comprises a subpopulation of AAVhu68 vpl in which 75% to 100 % of the N at position 57 of the vpl proteins are deamidated, as determined using mass spectrometry. In certain embodiments, the rAAVhu68 capsid comprises subpopulation of AAVhu68 vpl proteins, AAVhu68 vp2 proteins, and/or AAVhu68 vp3 proteins in which 75% to 100% of the N at position 329, based on the numbering of SEQ ID NO: 9 are deamidated as determined using mass spectrometry. In certain embodiments, the rAAVhu68 capsid comprises subpopulation of AAVhu68 vpl proteins, AAVhu68 vp2 proteins, and/or AAVhu68 vp3 proteins in which 75% to 100% of the N at position 452, based on the numbering of SEQ ID NO: 9, are deamidated as determined using mass spectrometry. In certain embodiments, the rAAVhu68 capsid comprises subpopulation of AAVhu68 vpl proteins, AAVhu68 vp2 proteins, and/or AAVhu68 vp3 proteins in which 75% to 100% of the N at position 512, based on the numbering of SEQ ID NO: 9, are deamidated. In certain embodiments, the rAAVhu68 capsid comprises subpopulations with 50% to 100% deamidation in each of N57, N329, N452, and N512, and optionally further comprise subpopulations of AAVhu68 vpl, AAVhu68 vp2 and/or AAVhu68 vp3 proteins which further comprise 1% to about 40% deamidation in at least one or more of positions N94, N113, N252, N253, Q258, N270, N303, N304, N305, N319, N328, N336, N409, N410, N477, N515, N598, Q599, N628, N651, N663, N709, or combinations thereof, based on the amino acid numbering of SEQ ID NO: 9.
In certain embodiments, the AAVhu68 capsid comprises a heterogenous population of AAVhu68 vpl proteins, AAVhu68 vp2 proteins, and AAVhu68 vp3 proteins which comprise amino acids 1 to 736 (vpl), amino acids 138 to 736 (vp2), and amino acids 203 to 736 (vp3) of SEQ ID NO: 9, respectively, with amino acid modifications comprising 50% to 100% deamidation in at least two asparagines (N) in asparagine - glycine pairs in two or more of N57, N329, N452, and/or N512 of SEQ ID NO: 9 as determined using mass spectrometry and optionally further comprising subpopulations comprising other deamidated amino acids;
wherein the AAVhu68 proteins contain amino acid modifications comprising 50% to 100% deamidation in two or more of asparagines (N) in position N57, N329, N452, and/or N512 based on the residue numbering of SEQ ID NO: 9 as determined using mass spectrometry and optionally further comprising subpopulations comprising other deamidated amino acids; and wherein the heterogenous population of AAVhu68 vpl proteins comprise at least a subpopulation having glutamic acid at position 67 based on the numbering of SEQ ID NO: 9, and the heterogenous population of AAVhu68vpl and AAVhu68 vp2 proteins comprising at least subpopulations comprising valine at position 157 based on the numbering of SEQ ID NO: 9. In certain embodiments, the nucleic acid sequence is SEQ ID NO: 8 or a sequence at least 70% identical to SEQ ID NO: 8 which encodes the amino acid sequence of SEQ ID NO: 9. In certain embodiments, the nucleic acid sequence is SEQ ID NO: 8. In certain embodiments, the rAAVhu68 capsid comprises a subpopulation of AAVhu68 vpl in which 75% to 100 % of the N at position 57 of the vpl proteins are deamidated, as determined using mass spectrometry. In certain embodiments, the rAAVhu68 capsid comprises subpopulation of AAVhu68 vpl proteins, AAVhu68 vp2 proteins, and/or AAVhu68 vp3 proteins in which 75% to 100% of the N at position 329, based on the numbering of SEQ ID NO: 9, are deamidated as determined using mass spectrometry. In certain embodiments, the rAAVhu68 capsid comprises subpopulation of AAVhu68 vpl proteins, AAVhu68 vp2 proteins, and/or AAVhu68 vp3 proteins in which 75% to 100% of the N at position 452, based on the numbering of SEQ ID NO: 9, are deamidated as determined using mass spectrometry. In certain embodiments, the rAAVhu68 capsid comprises subpopulation of AAVhu68 vpl proteins, AAVhu68 vp2 proteins, and/or AAVhu68 vp3 proteins in which 75% to 100% of the N at position 512, based on the numbering of SEQ ID NO: 9, are deamidated as determined using mass spectrometry. In certain embodiments, the rAAVhu68 capsid comprises subpopulations with 50% to 100% deamidation in each of N57, N329, N452, and N512, and optionally further comprise 1% to about 40% deamidation in at least one or more of positions N94, N113, N252, N253, Q258, N270, N303, N304, N305, N319, N328, N336, N409, N410, N477, N515, N598, Q599, N628, N651, N663, N709, or combinations thereof, based on the amino acid numbering of SEQ ID NO: 9.
In certain embodiments, the AAVhu68 capsid comprises AAVhu68 vpl proteins, AAVhu68 vp2 proteins, and AAVhu68 vp3 proteins produced from a nucleic acid sequence encoding the amino acid sequence of SEQ ID NO: 9, wherein the AAVhu68vpl proteins
comprise a glutamic acid at position 67 and a valine at position 157 and the AAVhu68vp2 proteins comprise a valine at position 157 based on the numbering of SEQ ID NO: 9. In certain embodiments, AAVhu68 capsid comprises subpopulations of AAVhu68 vpl, AAVhu68 vp2 and AAVhu68 vp3 proteins, wherein the subpopulations of the AAVhu68 vpl, AAVhu68 vp2 and AAV hu68 vp3 proteins comprise at least 50% to 100% deamidated asparagines (N) in asparagine - glycine pairs at each of positions 57, 329, 452, 512, relative to the amino acids in SEQ ID NO: 9, wherein the deamidated asparagines are deamidated to aspartic acid, isoaspartic acid, an interconverting aspartic acid/isoaspartic acid pair, or combinations thereof, as determined using mass spectrometry. In certain embodiments, the nucleic acid sequence encoding the vpl proteins is SEQ ID NO: 9, or a sequence at least 80% to at least 99% identical to SEQ ID NO: 8 which encodes the amino acid sequence of SEQ ID NO: 9; optionally wherein the nucleic acid sequence is at least 80% to 97% identical to SEQ ID NO: 8.
In certain embodiments, AAVhu68 capsid comprises heterogenous populations of AAVhu68 vpl, AAVhu68 vp2 and AAVhu68 vp3 proteins, wherein the AAVhu68 vpl proteins are amino acids 1 to 736 of SEQ ID NO: 9 (vpl) which comprise a glutamic acid at position 67 and a valine at position 157 and further comprise subpopulations of vpl proteins comprising modified amino acids based on the amino acids positions in SEQ ID NO: 9, wherein the AAVhu68 vp2 proteins are amino acids 138 to 736 of SEQ ID NO: 9 (vp2) which comprise a valine at position 157 and further comprise subpopulations of vp2 proteins comprising modified amino acids based on the amino acid positions in SEQ ID NO: 9, and wherein the AAVhu68 vp3 proteins are amino acids 203 to 736 of SEQ ID NO: 9 (vp3), which comprise subpopulations of vp3 proteins comprising modified amino acids based on the amino acid positions in SEQ ID NO: 9, wherein the subpopulations of the AAVhu68 vpl, AAVhu68 vp2 and AAV hu68 vp3 proteins comprise at least 50% to 100% deamidated asparagines (N) in asparagine - glycine pairs at each of positions 57, 329, 452, 512, relative to the amino acids in SEQ ID NO: 9, wherein the deamidated asparagines are deamidated to aspartic acid, isoaspartic acid, an interconverting aspartic acid/isoaspartic acid pair, or combinations thereof, as determined using mass spectrometry. In certain embodiments, the subpopulations of AAVhu68 vpl, vp2 and vp3 proteins further comprise: (i) one or more modifications selected from: acetylated lysine, phosphorylated serine and/or threonine, isomerized aspartic acid, deamidated glutamines, oxidated tryptophan and/or methionine, or
an amidated amino acid as determined using mass spectrometry; and/or (ii) 1% to 40% deamidation of asparagines at one or more of positions N94, N113, N252, N253, Q259, N270, N303, N304, N305, N319, N328, N336, N409, N410, N477, N515, N598, Q599, N628, N651, N663, N709, or combinations thereof, based on the numbering of SEQ ID NO: 9, as determined using mass spectrometry. In certain embodiments, the AAVhu68 capsid subpopulations of AAVhu68 vpl, vp2 and vp3 capsid proteins further comprise (a) at least 65% of asparagines (N) in asparagine - glycine pairs located at positions 57 of the vpl proteins are deamidated, as determined using mass spectrometry, based on the numbering of SEQ ID NO: 9; and/or (b) at least 75% of N in asparagine - glycine pairs in position 329 of the vpl, vp2 and vp3 proteins are deamidated, as determined using mass spectrometry, based on the residue numbering of the amino acid sequence of SEQ ID NO: 9; and/or (c) at least 50% of N in asparagine - glycine pairs in position 452 of the vpl, vp2 and vp3 proteins are deamidated, as determined using mass spectrometry, based on the residue numbering of the amino acid sequence of SEQ ID NO: 9; and/or (d) at least 75% of N in asparagine - glycine pairs in position 512 of the vpl, vp2 and vp3 proteins are deamidated, as determined using mass spectrometry, based on the residue numbering of the amino acid sequence of SEQ ID NO: 9. In certain embodiments, the nucleic acid sequence encoding the vpl proteins is SEQ ID NO: 8, or a sequence at least 80% to at least 99% identical to SEQ ID NO: 8 which encodes the amino acid sequence of SEQ ID NO: 9; optionally wherein the nucleic acid sequence is at least 80% to 97% identical to SEQ ID NO: 8.
See also WO 2018/160582, which is incorporated herein by reference.
Additionally, provided herein, is an rAAV production system useful for producing a rAAV as described herein. The production system comprises a cell culture comprising (a) a nucleic acid sequence encoding an AAV capsid protein; (b) the vector genome; and (c) sufficient AAV rep functions and helper functions to permit packaging of the vector genome into the AAV capsid. In certain embodiments, the vector genome comprises SEQ ID NO: 2. In certain embodiments, the vector genome comprises SEQ ID NO: 16. In certain embodiments, the cell culture is bacterial cell culture. In certain embodiments, the cell culture is mammalian cell culture. In certain embodiments, the cell culture is a human embryonic kidney 293 (HEK293) cell culture. In certain embodiments, the cell culture is a suspension cell culture. In certain embodiments, the AAV rep is from a different AAV. In certain embodiments, wherein the AAV rep is from AAV2. In certain embodiments, the AAV rep
coding sequence and cap genes are on the same nucleic acid molecule, wherein there is optionally a spacer between the rep sequence and cap gene.
For use in producing an AAV viral vector (e.g., a recombinant AAV), the vector genomes can be carried on any suitable vector, e.g., a plasmid, which is delivered to a packaging host cell. The plasmids useful in this invention may be engineered such that they are suitable for replication and packaging in vitro in prokaryotic cells, insect cells, mammalian cells, among others. Suitable transfection techniques and packaging host cells are known and/or can be readily designed by one of skill in the art.
Methods for generating and isolating AAVs suitable for use as vectors are known in the art. See generally, e.g., Grieger & Samulski, 2005, Adeno-associated virus as a gene therapy vector: Vector development, production and clinical applications, Adv. Biochem. Engin/Biotechnol. 99: 119-145; Buning et al., 2008, Recent developments in adeno- associated virus vector technology, J. Gene Med. 10:717-733; and the references cited below, each of which is incorporated herein by reference in its entirety. As used herein, a gene therapy vector refers to a rAAV as described herein, which is suitable for use in treating a patient. For packaging a gene into virions, the ITRs are the only AAV components required in cis in the same construct as the nucleic acid molecule containing the gene. The cap and rep genes can be supplied in trans.
Methods for determining potency (e.g., biological activity) of SMN polypeptides, in particular those expressed by recombinant viral vectors used for the treatment of SMA, are known in the art. See US 2022/0267798 Al, which is incorporated herein by reference.
In certain embodiments, the expression cassettes described herein are engineered into a genetic element (e.g., a shuttle plasmid) which transfers the sequences carried thereon into a packaging host cell for production of a viral vector. In one embodiment, the selected genetic element may be delivered to an AAV packaging cell by any suitable method, including transfection, electroporation, liposome delivery, membrane fusion techniques, high velocity DNA-coated pellets, viral infection and protoplast fusion. Stable AAV packaging cells can also be made. The methods used to make such constructs are known to those with skill in nucleic acid manipulation and include genetic engineering, recombinant engineering, and synthetic techniques. See, e.g., Molecular Cloning: A Laboratory Manual, ed. Green and Sambrook, Cold Spring Harbor Press, Cold Spring Harbor, NY (2012).
The term “AAV intermediate” or “AAV vector intermediate” refers to an assembled rAAV capsid which lacks the desired genomic sequences packaged therein. These may also be termed an “empty” capsid. Such a capsid may contain no detectable genomic sequences of an expression cassette, or only partially packaged genomic sequences which are insufficient to achieve expression of the gene product. These empty capsids are non-functional to transfer the gene of interest to a host cell.
The recombinant adeno-associated virus (AAV) described herein may be generated using techniques which are known. See, e.g., WO 2003/042397; WO 2005/033321, WO 2006/110689; US 7588772 B2. Such a method involves culturing a host cell which contains a nucleic acid sequence encoding an AAV capsid protein; a functional rep gene; an expression cassette composed of, at a minimum, AAV inverted terminal repeats (ITRs) and a transgene; and sufficient helper functions to permit packaging of the expression cassette into the AAV capsid protein. Methods of generating the capsid, coding sequences therefor, and methods for production of rAAV viral vectors have been described. See, e.g., Gao, et al, Proc. Natl. Acad. Sci. U.S.A. 100 (10), 6081-6086 (2003) and US 2013/0045186A1.
In certain embodiments, a production cell culture useful for producing a recombinant AAV having a capsid selected from AAVhu68, AAVrh91, AAVhu95 or AAVhu96 is provided. Such a cell culture contains a nucleic acid which expresses the AAV capsid protein in the host cell; a nucleic acid molecule suitable for packaging into the AAV capsid, e.g., a vector genome that contains AAV ITRs and a non- AAV nucleic acid sequence encoding a transgene (hSMNl) operably linked to regulatory sequences that direct expression of the transgene in a host cell; and sufficient AAV rep functions and adenovirus helper functions to permit packaging of the vector genome into the AAV capsid. In certain embodiments, the cell culture is composed of mammalian cells (e.g., human embryonic kidney 293 cells, among others) or insect cells (e.g., Spodoptera frugiperda (Sf9) cells). In certain embodiments, baculovirus provides the helper functions necessary for packaging the vector genome into the recombinant AAV capsid. Optionally the rep functions are provided by an AAV other than AAV2, selected to complement the source of the ITRs.
Methods for manufacturing the gene therapy vectors described herein include methods well known in the art such as generation of plasmid DNA used for production of the gene therapy vectors, generation of the vectors, and purification of the vectors. In some embodiments, the gene therapy vector is an AAV vector and the plasmids generated are an
AAV cis-plasmid encoding the AAV vector genome and the gene of interest, an AAV transplasmid containing AAV rep and cap genes, and an adenovirus helper plasmid. The vector generation process can include method steps such as initiation of cell culture, passage of cells, seeding of cells, transfection of cells with the plasmid DNA, post-transfection medium exchange to serum free medium, and the harvest of vector-containing cells and culture media. The harvested vector-containing cells and culture media are referred to herein as crude cell harvest. In yet another system, the gene therapy vectors are introduced into insect cells by infection with baculovirus-based vectors. For reviews on these production systems, see generally, e.g., Zhang et al., 2009, Adenovirus-adeno-associated virus hybrid for large-scale recombinant adeno-associated virus production, Human Gene Therapy 20:922-929, the contents of each of which is incorporated herein by reference in its entirety. Methods of making and using these and other AAV production systems are also described in the following US patents, the contents of each of which is incorporated herein by reference in its entirety: US Patent Nos. 5,139,941; 5,741,683; 6,057,152; 6,204,059; 6,268,213; 6,491,907; 6,660,514; 6,951,753; 7,094,604; 7,172,893; 7,201,898; 7,229,823; and 7,439,065.
The crude cell harvest may thereafter be subject method steps such as concentration of the vector harvest, diafiltration of the vector harvest, microfluidization of the vector harvest, nuclease digestion of the vector harvest, filtration of microfluidized intermediate, crude purification by chromatography, crude purification by ultracentrifugation, buffer exchange by tangential flow filtration, and/or formulation and filtration to prepare bulk vector. An affinity chromatography purification followed anion exchange resin chromatography are used to purify the vector drug product and to remove empty capsids. These methods are described in more detail in W02017/160360, filed December 9, 2016, entitled “Scalable Purification Method for AAV9”, which is incorporated by reference. Purification methods for AAV8, W02017/100676, filed December 9, 2016, and rhlO, W02017/100704, filed December 9, 2016, entitled “Scalable Purification Method for AAVrhlO”, also filed December 11, 2015, and for AAV1, W02017/100674, filed December 9, 2016 for “Scalable Purification Method for AAV1”, filed December 11, 2015, are all incorporated by reference herein. Other suitable methods may be selected.
To calculate empty and full particle content, VP3 band volumes for a selected sample (e.g., in examples herein an iodixanol gradient-purified preparation where # of genome copies (GC) = # of particles) are plotted against GC particles loaded. The resulting linear
equation (y = mx+c) is used to calculate the number of particles in the band volumes of the test article peaks. The number of particles (pt) per 20 pL loaded is then multiplied by 50 to give particles (pt) /mL. Pt/mL divided by GC/mL gives the ratio of particles to genome copies (pt/GC). Pt/mL-GC/mL gives empty pt/mL. Empty pt/mL divided by pt/mL and x 100 gives the percentage of empty particles.
Generally, methods for assaying for empty capsids and AAV vector particles with packaged genomes have been known in the art. See, e.g., Grimm et al., Gene Therapy (1999) 6: 1322-1330; Sommer et al., Molec. Ther. (2003) 7: 122-128. To test for denatured capsid, the methods include subjecting the treated AAV stock to SDS-polyacrylamide gel electrophoresis, consisting of any gel capable of separating the three capsid proteins, for example, a gradient gel containing 3-8% Tris-acetate in the buffer, then running the gel until sample material is separated, and blotting the gel onto nylon or nitrocellulose membranes, preferably nylon. Anti-AAV capsid antibodies are then used as the primary antibodies that bind to denatured capsid proteins, preferably an anti-AAV capsid monoclonal antibody, most preferably the Bl anti-AAV-2 monoclonal antibody (Wobus et al., J. Virol. (2000) 74:9281- 9293). A secondary antibody is then used, one that binds to the primary antibody and contains a means for detecting binding with the primary antibody, more preferably an anti- IgG antibody containing a detection molecule covalently bound to it, most preferably a sheep anti-mouse IgG antibody covalently linked to horseradish peroxidase. A method for detecting binding is used to semi-quantitatively determine binding between the primary and secondary antibodies, preferably a detection method capable of detecting radioactive isotope emissions, electromagnetic radiation, or colorimetric changes, most preferably a chemiluminescence detection kit. For example, for SDS-PAGE, samples from column fractions can be taken and heated in SDS-PAGE loading buffer containing reducing agent (e.g., DTT), and capsid proteins were resolved on pre-cast gradient polyacrylamide gels (e.g., Novex). Silver staining may be performed using SilverXpress (Invitrogen, CA) according to the manufacturer's instructions or other suitable staining method, i.e., SYPRO ruby or coomassie stains. In one embodiment, the concentration of AAV vector genomes (vg) in column fractions can be measured by quantitative real time PCR (Q-PCR). Samples are diluted and digested with DNase I (or another suitable nuclease) to remove exogenous DNA. After inactivation of the nuclease, the samples are further diluted and amplified using primers and a TaqMan™ fluorogenic probe specific for the DNA sequence between the primers. The number of cycles
required to reach a defined level of fluorescence (threshold cycle, Ct) is measured for each sample on an Applied Biosystems Prism 7700 Sequence Detection System. Plasmid DNA containing identical sequences to that contained in the AAV vector is employed to generate a standard curve in the Q-PCR reaction. The cycle threshold (Ct) values obtained from the samples are used to determine vector genome titer by normalizing it to the Ct value of the plasmid standard curve. End-point assays based on the digital PCR can also be used.
In certain embodiments, an optimized q-PCR method is used which utilizes a broadspectrum serine protease, e.g., proteinase K (such as is commercially available from Qiagen). More particularly, the optimized qPCR genome titer assay is similar to a standard assay, except that after the DNase I digestion, samples are diluted with proteinase K buffer and treated with proteinase K followed by heat inactivation. Suitably samples are diluted with proteinase K buffer in an amount equal to the sample size. The proteinase K buffer may be concentrated to 2 -old or higher. Typically, proteinase K treatment is about 0.2 mg/mL, but may be varied from 0.1 mg/mL to about 1 mg/mL. The treatment step is generally conducted at about 55 °C for about 15 minutes, but may be performed at a lower temperature (e.g., about 37 °C to about 50 °C) over a longer time period (e.g., about 20 minutes to about 30 minutes), or a higher temperature (e.g., up to about 60 °C) for a shorter time period (e.g., about 5 to 10 minutes). Similarly, heat inactivation is generally at about 95 °C for about 15 minutes, but the temperature may be lowered (e.g., about 70 to about 90 °C) and the time extended (e.g., about 20 minutes to about 30 minutes). Samples are then diluted (e.g., 1000- fold) and subjected to TaqMan analysis as described in the standard assay.
Additionally, or alternatively, droplet digital PCR (ddPCR) may be used. For example, methods for determining single-stranded and self-complementary AAV vector genome titers by ddPCR have been described. See, e.g., M. Lock et al, Hu Gene Therapy Methods, Hum Gene Ther Methods. 2014 Apr;25(2): 115-25. doi: 10.1089/hgtb.2013.131. Epub 2014 Feb 14.
In certain embodiments, the manufacturing process for rAAV as described herein involves method as described in US Provisional Patent Application No. 63/371,597, filed August 16, 2022, and US Provisional Patent Application No. 63/371,592, filed August 16, 2022, which are incorporated herein by reference in their entirety.
In brief, the method for separating rAAVhu68 (or AAVrh91, AAVhu95 or AAVhu96) particles having packaged genomic sequences from genome-deficient AAVhu68
(or AAVrh91 or AAVhu95 or AAVhu96) intermediates involves subjecting a suspension comprising recombinant AAVhu68 (or AAVrh91) viral particles and AAVhu68 (or AAVrh91 or AVhu95 or AAVhu96) capsid intermediates to fast performance liquid chromatography, wherein the AAVhu68 (or AAVrh91 or AAVhu95 or AAVhu96) viral particles and AAVhu68 intermediates are bound to a strong anion exchange resin equilibrated at a pH of about 10.2 (or about 9.8 for AAVrh91), and subjected to a salt gradient while monitoring eluate for ultraviolet absorbance at about 260 nanometers (nm) and about 280 nm. Although less optimal for rAAVhu68 and AAVrh91, the pH may be in the range of about 10 to 10.4. In this method, the AAV full capsids are collected from a fraction which is eluted when the ratio of A260/A280 reaches an inflection point. In one example, for the Affinity Chromatography step, the diafiltered product may be applied to an affinity resin (Life Technologies) that efficiently captures the AAV serotype. Under these ionic conditions, a significant percentage of residual cellular DNA and proteins flow through the column, while AAV particles are efficiently captured.
As used herein, the term “clade” as it relates to groups of AAV refers to a group of AAV which are phylogenetically related to one another as determined using a Neighbor- Joining algorithm by a bootstrap value of at least 75% (of at least 1000 replicates) and a Poisson correction distance measurement of no more than 0.05, based on alignment of the AAV vpl amino acid sequence. The Neighbor- Joining algorithm has been described in the literature. See, e.g., M. Nei and S. Kumar, Molecular Evolution and Phylogenetics (Oxford University Press, New York (2000). Computer programs are available that can be used to implement this algorithm. For example, the MEGA v2.1 program implements the modified Nei-Gojobori method. Using these techniques and computer programs, and the sequence of an AAV vpl capsid protein, one of skill in the art can readily determine whether a selected AAV is contained in one of the clades identified herein, in another clade, or is outside these clades. See, e.g., G Gao, et al, J Virol, 2004 Jun; 78(10): 6381-6388, which identifies Clades A, B, C, D, E and F, and provides nucleic acid sequences of novel AAV, GenBank Accession Numbers AY530553 to AY530629. See, also, WO 2005/033321.
The abbreviation “sc” refers to self-complementary. “Self-complementary AAV” refers a construct in which a coding region carried by a recombinant AAV nucleic acid sequence has been designed to form an intra-molecular double-stranded DNA template. Upon infection, rather than waiting for cell mediated synthesis of the second strand, the two
complementary halves of scAAV will associate to form one double stranded DNA (dsDNA) unit that is ready for immediate replication and transcription. See, e.g., D M McCarty et al, “Self-complementary recombinant adeno-associated virus (scAAV) vectors promote efficient transduction independently of DNA synthesis”, Gene Therapy, (August 2001), Vol 8, Number 16, Pages 1248-1254. Self-complementary AAVs are described in, e.g., U.S. Patent Nos. 6,596,535; 7,125,717; and 7,456,683, each of which is incorporated herein by reference in its entirety. In certain embodiments, the rAAV provided herein is a scAAV vector. In certain embodiments, the rAAV is not a scAAV vector and includes a s single-stranded DNA genome.
A “replication-defective virus" or “viral vector” refers to a synthetic or artificial viral particle in which an expression cassette containing a gene of interest is packaged in a viral capsid or envelope, where any viral genomic sequences also packaged within the viral capsid or envelope are replication-deficient; i.e., they cannot generate progeny virions but retain the ability to infect target cells. In one embodiment, the genome of the viral vector does not include genes encoding the enzymes required to replicate (the genome can be engineered to be "gutless" - containing only the gene of interest flanked by the signals required for amplification and packaging of the artificial genome), but these genes may be supplied during production. Therefore, it is deemed safe for use in gene therapy since replication and infection by progeny virions cannot occur except in the presence of the viral enzyme required for replication.
As used herein, the terms “recombinant AAV”, “rAAV” and “artificial AAV” used interchangeably, mean, without limitation, a AAV comprising a capsid protein and a vector genome packaged therein, wherein the vector genome comprising a nucleic acid heterologous to the AAV. In one embodiment, the capsid protein is a non-naturally occurring capsid. Such an artificial capsid may be generated by any suitable technique, using a selected AAV sequence (e.g., a fragment of a vpl capsid protein) in combination with heterologous sequences which may be obtained from a different selected AAV, non-contiguous portions of the same AAV, from a non- AAV viral source, or from a non-viral source. An artificial AAV may be, without limitation, a pseudotyped AAV, a chimeric AAV capsid, a recombinant AAV capsid, or a “humanized” AAV capsid. Pseudotyped vectors, wherein the capsid of one AAV is replaced with a heterologous capsid protein, are useful in the invention. In one embodiment, AAV2/5 and AAV2/8 are exemplary pseudotyped vectors. The selected genetic
element may be delivered by any suitable method, including transfection, electroporation, liposome delivery, membrane fusion techniques, high velocity DNA-coated pellets, viral infection and protoplast fusion. The methods used to make such constructs are known to those with skill in nucleic acid manipulation and include genetic engineering, recombinant engineering, and synthetic techniques. See, e.g., Green and Sambrook, Molecular Cloning: A Laboratory Manual, Cold Spring Harbor Press, Cold Spring Harbor, NY (2012).
In many instances, rAAV particles are referred to as DNase resistant. However, in addition to this endonuclease (DNase), other endo- and exo- nucleases may also be used in the purification steps described herein, to remove contaminating nucleic acids. Such nucleases may be selected to degrade single stranded DNA and/or double-stranded DNA, and RNA. Such steps may contain a single nuclease, or mixtures of nucleases directed to different targets, and may be endonucleases or exonucleases.
The term “nuclease-resistant” indicates that the AAV capsid has fully assembled around the expression cassette which is designed to deliver a gene to a host cell and protects these packaged genomic sequences from degradation (digestion) during nuclease incubation steps designed to remove contaminating nucleic acids which may be present from the production process.
As used herein when used to refer to vp capsid proteins, the term “heterogeneous” or any grammatical variation thereof, refers to a population consisting of elements that are not the same, for example, having vpl, vp2 or vp3 monomers (proteins) with different modified amino acid sequences.
The term “heterogeneous” as used in connection with vpl, vp2 and vp3 proteins (alternatively termed isoforms), refers to differences in the amino acid sequence of the vpl, vp2 and vp3 proteins within a capsid. The AAV capsid contains subpopulations within the vpl proteins, within the vp2 proteins and within the vp3 proteins which have modifications from the predicted amino acid residues. These subpopulations include, at a minimum, certain deamidated asparagine (N or Asn) residues. For example, certain subpopulations comprise at least one, two, three or four highly deamidated asparagines (N) positions in asparagine - glycine pairs and optionally further comprising other deamidated amino acids, wherein the deamidation results in an amino acid change and other optional modifications.
As used herein, a “subpopulation” of vp proteins refers to a group of vp proteins which has at least one defined characteristic in common and which consists of at least one
group member to less than all members of the reference group, unless otherwise specified. For example, a “subpopulation” of vpl proteins is at least one (1) vpl protein and less than all vpl proteins in an assembled AAV capsid, unless otherwise specified. A “subpopulation” of vp3 proteins may be one (1) vp3 protein to less than all vp3 proteins in an assembled AAV capsid, unless otherwise specified. For example, vpl proteins may be a subpopulation of vp proteins; vp2 proteins may be a separate subpopulation of vp proteins, and vp3 are yet a further subpopulation of vp proteins in an assembled AAV capsid. In another example, vpl, vp2 and vp3 proteins may contain subpopulations having different modifications, e.g., at least one, two, three or four highly deamidated asparagines, e.g., at asparagine - glycine pairs.
Nucleic Acid Molecules
The term “nucleic acid” as used herein can be RNA, DNA, or a modification thereof, and can be single or double stranded, and can be selected, for example, from a group including nucleic acid encoding a protein of interest, oligonucleotides, nucleic acid analogues, for example peptide-nucleic acid (PNA), pseudocomplementary PNA (pc-PNA), locked nucleic acid (LNA) etc. A nucleotide refers to a ribonucleotide, deoxynucleotide or a modified form of either type of nucleotide (e.g., a peptide nucleic acid oligomer). One of ordinary skill will appreciate that functional variants of these nucleic acid molecules are also intended to be a part of the present invention. Functional variants are nucleic acid sequences that can be directly translated, using the standard genetic code, to provide an amino acid sequence identical to that translated from the parental nucleic acid molecules. Such nucleic acid sequences include, for example.
In certain embodiments, the recombinant nucleic acid molecules encoding a functional SMN1 protein, and other constructs encompassed by the present disclosure, are useful in generating expression cassettes and vector genomes and may be engineered for expression in yeast cells, insect cells, or mammalian cells, such as human cells. Methods are known and have been described previously (e.g., WO 96/09378). A sequence is considered engineered if at least one non-preferred codon as compared to a wild type (WT) sequence is replaced by a codon that is more preferred. Herein, a non-preferred codon is a codon that is used less frequently in an organism than another codon coding for the same amino acid, and a codon that is more preferred is a codon that is used more frequently in an organism than a non-preferred codon. The frequency of codon usage for a specific organism can be found in
codon frequency tables, such as in kazusa.jp/codon. Preferably more than one non-preferred codon, preferably most or all non-preferred codons, are replaced by codons that are more preferred. Preferably the most frequently used codons in an organism are used in an engineered sequence. Replacement by preferred codons generally leads to higher expression. It will also be understood by a skilled person that numerous different nucleic acid molecules can encode the same polypeptide as a result of the degeneracy of the genetic code. It is also understood that skilled persons may, using routine techniques, make nucleotide substitutions that do not affect the amino acid sequence encoded by the nucleic acid molecules to reflect the codon usage of any particular host organism in which the polypeptides are to be expressed. Therefore, unless otherwise specified, a “nucleic acid sequence encoding an amino acid sequence” includes all nucleotide sequences that are degenerate versions of each other and that encode the same amino acid sequence. Nucleic acid sequences can be cloned using routine molecular biology techniques, or generated de novo by DNA synthesis, which can be performed using routine procedures by service companies having business in the field of DNA synthesis and/or molecular cloning (e.g., GeneArt, GenScript, Life Technologies, Eurofins).
By “engineered” is meant that a nucleic acid molecule that contains a sequence encoding an SMN1 gene described herein in any suitable genetic element, e.g., naked DNA, phage, transposon, cosmid, episome, etc., which transfers the SMN1 gene to a host cell, e.g., for generating non-viral delivery systems (e.g., RNA-based systems, naked DNA, or the like), or for generating viral vectors in a packaging host cell, and/or for delivery to a target cells in a subject. In certain embodiments, the genetic element is a vector. In certain embodiments, the genetic element is a plasmid. The methods used to make such engineered constructs are known to those with skill in nucleic acid manipulation and include genetic engineering, recombinant engineering, and synthetic techniques. See, e.g., Green and Sambrook, Molecular Cloning: A Laboratory Manual, Cold Spring Harbor Press, Cold Spring Harbor, NY (2012).
Pharmaceutical Composition
In certain embodiments, provided herein are pharmaceutical compositions comprising an rAAV or a stock of rAAV as described herein in a formulation buffer. The pharmaceutical
compositions described herein are designed for delivery to subjects in need thereof by any suitable route, or a combination of different routes.
In certain embodiments, the pharmaceutical composition is formulated at about 1 x 109 genome copies (GC)/mL to about 1 x 1014 GC/mL of the rAAV. In other embodiments, the pharmaceutical composition is formulated at about 3 x 109 GC/mL to about 3 x 1013 GC/mL of the rAAV. In yet other embodiments, the pharmaceutical composition is formulated at about 1 x 109 GC/mL to about 1 x 1013 GC/mL of the rAAV. In certain embodiments, the pharmaceutical composition is formulated at least 1 x 1011 GC/mL of the rAAV.
Provided herein also is a composition comprising an rAAV as described herein and an aqueous suspension media. In certain embodiments, the suspension is formulated for intravenous delivery, intrathecal administration, or intracerebroventricular administration. In one aspect, the composition contains at least one rAAV stock and an optional carrier, excipient and/or preservative.
As used herein, a “stock” of rAAV refers to a population of rAAV. Despite heterogeneity in their capsid proteins due to deamidation, rAAV in a stock are expected to share an identical vector genome. A stock can include rAAV having capsids with, for example, heterogeneous deamidation patterns characteristic of the selected AAV capsid proteins and a selected production system. The stock may be produced from a single production system or pooled from multiple runs of the production system. A variety of production systems, including but not limited to those described herein, may be selected.
As used herein, “carrier” includes any and all solvents, dispersion media, vehicles, coatings, diluents, antibacterial and antifungal agents, isotonic and absorption delaying agents, buffers, carrier solutions, suspensions, colloids, and the like. The use of such media and agents for pharmaceutical active substances is well known in the art. Supplementary active ingredients can also be incorporated into the compositions. The phrase “pharmaceutically-acceptable” refers to molecular entities and compositions that do not produce an allergic or similar untoward reaction when administered to a host. Delivery vehicles such as liposomes, nanocapsules, nanoparticles, lipid nanoparticle (LNP), microparticles, microspheres, lipid particles, vesicles, and the like, may be used for the introduction of the compositions of the present invention into suitable host cells. In particular, the rAAV vector delivered vector genomes may be formulated for delivery either
encapsulated in a lipid particle, a liposome, a vesicle, a nanosphere, or a nanoparticle or the like.
In certain embodiments, the composition includes a final formulation suitable for delivery to a subject, e.g., is an aqueous liquid suspension buffered to a physiologically compatible pH and salt concentration. Optionally, one or more surfactants are present in the formulation. In another embodiment, the composition may be transported as a concentrate which is diluted for administration to a subject. In other embodiments, the composition may be lyophilized and reconstituted at the time of administration.
A suitable surfactant, or combination of surfactants, may be selected from among non-ionic surfactants that are nontoxic.
In certain embodiments, the composition includes a carrier, diluent, excipient and/or adjuvant. Suitable carriers may be readily selected by one of skill in the art in view of the indication for which the transfer virus is directed. For example, one suitable carrier includes saline, which may be formulated with a variety of buffering solutions (e.g., phosphate buffered saline). Other exemplary carriers include sterile saline, lactose, sucrose, calcium phosphate, gelatin, dextran, agar, pectin, peanut oil, sesame oil, and water. The buffer/carrier should include a component that prevents the rAAV, from sticking to the infusion tubing but does not interfere with the rAAV binding activity in vivo. A suitable surfactant, or combination of surfactants, may be selected from among non-ionic surfactants that are nontoxic. In one embodiment, a difunctional block copolymer surfactant terminating in primary hydroxyl groups is selected, e.g., such as Pol oxamer 188 (also known under the commercial names Pluronic® F68 [BASF], Lutrol® F68, Synperonic® F68, Kolliphor® Pl 88) which has a neutral pH, has an average molecular weight of 8400. Other surfactants and other Poloxamers may be selected, i.e., nonionic triblock copolymers composed of a central hydrophobic chain of polyoxypropylene (polypropylene oxide)) flanked by two hydrophilic chains of polyoxyethylene (poly(ethylene oxide)), SOLUTOL HS 15 (Macrogol- 15 Hydroxystearate), LABRASOL (Poly oxy capryllic glyceride), poly oxy -oleyl ether, TWEEN (polyoxyethylene sorbitan fatty acid esters), ethanol and polyethylene glycol. In one embodiment, the formulation contains a poloxamer. These copolymers are commonly named with the letter "P" (for poloxamer) followed by three digits: the first two digits x 100 give the approximate molecular mass of the poly oxypropylene core, and the last digit x 10 gives the percentage polyoxyethylene content. In one embodiment Poloxamer 188 is selected.
The surfactant may be present in an amount up to about 0.0005 % to about 0.001% of the suspension.
In certain embodiments, the composition containing the rAAV is delivered at a pH in the range of 6 to 8, or 7.2 to 7.8, or 7.5 to 8. For intrathecal delivery, a pH above 7.5 may be desired, e.g., 7.5 to 8, or 7.8. For intravenous delivery, a pH of about 6.8 to about 7.2 may be desired.
In certain embodiments, the formulation may contain a buffered saline aqueous solution not comprising sodium bicarbonate. Such a formulation may contain a buffered saline aqueous solution comprising one or more of sodium phosphate, sodium chloride, potassium chloride, calcium chloride, magnesium chloride and mixtures thereof, in water, such as a Harvard’s buffer. In one embodiment, the buffer is PBS. In another embodiment, the buffer is an artificial cerebrospinal fluid (aCSF), e.g., Eliott’s formulation buffer; or Harvard apparatus perfusion fluid (an artificial CSF with final Ion Concentrations (in mM): Na 150; K 3.0; Ca 1.4; Mg 0.8; P 1.0; Cl 155). The aqueous solution may further contain Kolliphor® Pl 88, a pol oxamer which is commercially available from BASF which was formerly sold under the trade name Lutrol® F68. The aqueous solution may have a pH of 7.2.
In another embodiment, the formulation may contain a buffered saline aqueous solution comprising 1 mM Sodium Phosphate (NasPCh), 150 mM sodium chloride (NaCl), 3mM potassium chloride (KC1), 1.4 mM calcium chloride (CaC12), 0.8 mM magnesium chloride (MgCh), and 0.001% poloxamer (e.g., Kolliphor®) 188, pH 7.2. See, e.g., harvardapparatus.com/harvard-apparatus-perfusion-fluid.html. In certain embodiments, Harvard’s buffer is preferred due to better pH stability observed with Harvard’s buffer.
In certain embodiments, the formulation buffer is artificial CSF with Pluronic F68. In other embodiments, the formulation may contain one or more permeation enhancers. Examples of suitable permeation enhancers may include, e.g., mannitol, sodium glycocholate, sodium taurocholate, sodium deoxycholate, sodium salicylate, sodium caprylate, sodium caprate, sodium lauryl sulfate, polyoxyethylene-9-laurel ether, or EDTA.
Optionally, the compositions of the invention may contain, in addition to the rAAV and carrier(s), other conventional pharmaceutical ingredients, such as preservatives, or chemical stabilizers. Suitable exemplary preservatives include chlorobutanol, potassium
sorbate, sorbic acid, sulfur dioxide, propyl gallate, the parabens, ethyl vanillin, glycerin, phenol, and parachlorophenol. Suitable chemical stabilizers include gelatin and albumin.
The compositions according to the present invention may comprise a pharmaceutically acceptable carrier, such as defined above. Suitably, the compositions described herein comprise an effective amount of one or more AAV suspended in a pharmaceutically suitable carrier and/or admixed with suitable excipients designed for delivery to the subject via injection, osmotic pump, intrathecal catheter, or for delivery by another device or route. In certain embodiments, an ommaya reservoir is used for delivery. In one example, the composition is formulated for intrathecal delivery. In one example, the composition is formulated for intravenous (iv) delivery.
In certain embodiments, a therapeutically effective amount of a vector is included in the pharmaceutical composition. The selection of the carrier is not a limitation of the present invention. Other conventional pharmaceutically acceptable carrier, such as preservatives, or chemical stabilizers. Suitable exemplary preservatives include chlorobutanol, potassium sorbate, sorbic acid, sulfur dioxide, propyl gallate, the parabens, ethyl vanillin, glycerin, phenol, and parachlorophenol. Suitable chemical stabilizers include gelatin and albumin.
As used herein, the term “dosage” or “amount” can refer to the total dosage or amount delivered to the subject in the course of treatment, or the dosage or amount delivered in a single unit (or multiple unit or split dosage) administration.
In certain embodiments, the compositions can be formulated in dosage units to contain an amount of rAAV that is in the range of about 1 x 109 genome copies (GC) per gram (g) of brain mass to about 1 x 1013 GC per g of brain mass, including all integers or fractional amounts within the range and the endpoints. In other embodiments, the dosage is 1 x IO10 GC per gram of brain mass to about 1 x 1013 GC per gram of brain mass. In specific embodiments, the dose of the rAAV administered to a patient is at least about 1.0 x 109 GC/g, about 1.5 x 109 GC/g, about 2.0 x 109 GC/g, about 2.5 x 109 GC/g, about 3.0 x 109 GC/g, about 3.5 x 109 GC/g, about 4.0 x 109 GC/g, about 4.5 x 109 GC/g, about 5.0 x 109 GC/g, about 5.5 x 109 GC/g, about 6.0 x 109 GC/g, about 6.5 x 109 GC/g, about 7.0 x 109 GC/g, about 7.5 x 109 GC/g, about 8.0 x 109 GC/g, about 8.5 x 109 GC/g, about 9.0 x 109 GC/g, about 9.5 x 109 GC/g, about 1.0 x 1010 GC/g, about 1.5 x 1010 GC/g, about 2.0 x 1010 GC/g, about 2.5 x 1010 GC/g, about 3.0 x 1010 GC/g, about 3.5 x 1010 GC/g, about 4.0 x 1010 GC/g, about 4.5 x 1010 GC/g, about 5.0 x 1010 GC/g, about 5.5 x 1010 GC/g, about 6.0 x 1010 GC/g,
about 6.5 x IO10 GC/g, about 7.0 x IO10 GC/g, about 7.5 x IO10 GC/g, about 8.0 x IO10 GC/g, about 8.5 x IO10 GC/g, about 9.0 x IO10 GC/g, about 9.5 x IO10 GC/g, about 1.0 x 1011 GC/g, about 1.5 x 1011 GC/g, about 2.0 x 1011 GC/g, about 2.5 x 1011 GC/g, about 3.0 x 1011 GC/g, about 3.5 x 1011 GC/g, about 4.0 x 1011 GC/g, about 4.5 x 1011 GC/g, about 5.0 x 1011 GC/g, about 5.5 x 1011 GC/g, about 6.0 x 1011 GC/g, about 6.5 x 1011 GC/g, about 7.0 x 1011 GC/g, about 7.5 x 1011 GC/g, about 8.0 x 1011 GC/g, about 8.5 x 1011 GC/g, about 9.0 x 1011 GC/g, about 9.5 x 1011 GC/g, about 1.0 x 1012 GC/g, about 1.5 x 1012 GC/g, about 2.0 x 1012 GC/g, about 2.5 x 1012 GC/g, about 3.0 x 1012 GC/g, about 3.5 x 1012 GC/g, about 4.0 x 1012 GC/g, about 4.5 x 1012 GC/g, about 5.0 x 1012 GC/g, about 5.5 x 1012 GC/g, about 6.0 x 1012 GC/g, about 6.5 x 1012 GC/g, about 7.0 x 1012 GC/g, about 7.5 x 1012 GC/g, about 8.0 x 1012 GC/g, about 8.5 x 1012 GC/g, about 9.0 x 1012 GC/g, about 9.5 x 1012 GC/g, about 1.0 x 1013 GC/g, about 1.5 x 1013 GC/g, about 2.0 x 1013 GC/g, about 2.5 x 1013 GC/g, about 3.0 x 1013 GC/g, about 3.5 x 1013 GC/g, about 4.0 x 1013 GC/g, about 4.5 x 1013 GC/g, about 5.0 x 1013 GC/g, about 5.5 x 1013 GC/g, about 6.0 x 1013 GC/g, about 6.5 x 1013 GC/g, about 7.0 x 1013 GC/g, about 7.5 x 1013 GC/g, about 8.0 x 1013 GC/g, about 8.5 x 1013 GC/g, about 9.0 x 1013 GC/g, about 9.5 x 1013 GC/g, or about 1.0 x 1014 GC/g brain mass.
Further, the compositions can be formulated in dosage units to contain an amount of replication-defective virus that is in the range of about 1.0 x 109 GC to about 1.0 x 1016 GC of the rAAV (to treat an average subject of 70 kg in body weight) including all integers or fractional amounts within the range, and preferably 1.0 x 1012 GC to 1.0 x 1014 GC for a human patient. In certain embodiments, the compositions are formulated to contain at least IxlO9, 2xl09, 3xl09, 4xl09, 5xl09, 6xl09, 7xl09, 8xl09, or 9xl09 GC per dose including all integers or fractional amounts within the range. In another embodiment, the compositions are formulated to contain at least IxlO10, 2xlO10, 3xl010, 4xlO10, 5xl010, 6xlO10, 7xlO10, 8xl010, or 9xlO10 GC per dose including all integers or fractional amounts within the range. In another embodiment, the compositions are formulated to contain at least IxlO11, 2xlOn, 3xl0n, 4xlOn, 5xl0n, 6xlOn, 7xlOn, 8xl0n, or 9xlOn GC per dose including all integers or fractional amounts within the range. In another embodiment, the compositions are formulated to contain at least IxlO12, 2xl012, 3xl012, 4xl012, 5xl012, 6xl012, 7xl012, 8xl012, or 9xl012 GC per dose including all integers or fractional amounts within the range. In another embodiment, the compositions are formulated to contain at least IxlO13, 2xl013, 3xl013, 4xl013, 5xl013, 6xl013, 7xl013, 8xl013, or 9xl013 GC per dose including all integers
or fractional amounts within the range. In another embodiment, the compositions are formulated to contain at least IxlO14, 2xl014, 3xl014, 4x1014, 5xl014, 6xl014, 7xl014, 8xl014, or 9xl014 GC per dose including all integers or fractional amounts within the range. In another embodiment, the compositions are formulated to contain at least IxlO15, 2xl015, 3xl015, 4xl015, 5xl015, 6xl015, 7xl015, 8xl015, or 9xl015 GC per dose including all integers or fractional amounts within the range. In certain embodiments, for human application the dose can range from IxlO10 to about IxlO12 GC per dose including all integers or fractional amounts within the range.
In certain embodiments, the composition is administered intravenously. In certain embodiments, the composition is administered intrathecally, optionally via intra-ci sterna magna (ICM) injection. In certain embodiments, the composition is administered via intraparenchymal administration. In certain embodiments, the composition is administered via an Ommaya Reservoir delivery system. In certain embodiments, the composition is administered via more than one route, for example intravenously and intrathecally.
In certain embodiments, the pharmaceutical composition comprising the rAAV as described herein is administrable at a dose of about I x lO9 GC per gram of brain mass to about I x lO13 GC per gram of brain mass. In certain embodiments, the pharmaceutical composition comprising the rAAV as described herein is administrable at a dose of about 1 x 1011 GC per gram of brain mass to about 6 x 1011 GC per gram of brain mass. In certain embodiments, the pharmaceutical composition comprising the rAAV as described herein is administrable at a dose of about I x lO11 GC per gram of brain mass. In certain embodiments, the pharmaceutical composition comprising the rAAV as described herein is administrable at a dose of about 3.3 x 1011 GC per gram of brain mass. In certain embodiments, the pharmaceutical composition comprising the rAAV as described herein is administrable at a dose of about 6.7 x 1011 GC per gram of brain mass.
Methods
In certain embodiments, provided herein are methods of treating SMA in a subject in need thereof. The methods include administering an rAAV vector provided herein, or a composition comprising the rAAV in an aqueous suspension.
In certain embodiments, the methods include treating a subject having one or more symptoms associated with SMA (e.g., atrophy of the limb muscles, difficulty or inability
walking, difficulty breathing, or other symptoms of SMA). In certain embodiments, the subject has reduced levels of the survival motor neuron (SMN) protein as a result of loss or mutation of the SMN1 gene. In certain embodiments, the subject has been identified as having type I (Werdnig-Hoffmann disease), type II (Dubowitz disease), type III (Kugelberg- Welander disease), or type IV (adult) SMA. In certain embodiments, the subject having SMA has two mutant alleles of the genomic SMN1 gene. In certain embodiments, the subject has a deletion or mutation (e.g., loss of function point mutation) in each SMN1 allele. In certain embodiments, the subject is homozygous for a SMN1 gene mutation. In certain embodiments, the subject is heterozygous for two different SMN1 gene mutations. In certain embodiments, the subject has at least 1, at least 2, at least 3, at least 4 copies of a functional SMN2 gene. In certain embodiments, the subject has fewer 1, 2, 3, or 4 copies of a functional SMN2 gene. In certain embodiments, the subject is human and selected from the pediatric and adult population. In certain embodiments, the subject is greater than or equal to 18 years of age (e.g., 18 years of age or older). In certain embodiments, the subject is younger than 18 years of age, younger than 10 years of age, or younger than 6 years of age. In certain embodiments, the subject is around 2 weeks, 1 month, 3 months, 6 months, 1 year, 2 years, 3 years, 4 years, or 5 years of age. In certain embodiments, the subject is less than 1 month old. In certain embodiments, the subject is less than 6 months old. In certain embodiments, the subject is less than 1 year old. In certain embodiments, the subject is 1 month to 10 months old. In certain embodiments, the subject is post-natal and less than 2 years old with spinal muscular atrophy (SMA) with bi-allelic mutations in the survival motor neuron 1 (SMN1) gene. In certain embodiments, the subject is not age limited.
In certain embodiments, treatment includes alleviating symptoms of a patient such that the severity of disease is reduced (e.g., from SMA1 (also, Werdnig-Hoffmann disease, 0- 6 months) to SMA2 (intermediate - 6-18 months, Dubowitz disease), SMA3 (>12 months, Kugelberg- Welander disease), or SMA4 (adult onset).
In certain embodiments, the methods include treating a subject following onset, or prior to onset of one or more disease symptoms (including, for example, one or more of areflexia, particularly in extremities; overall muscle weakness, poor muscle tone, limpness or a tendency to flop; difficulty achieving developmental milestones, difficulty sitting/standing/walking; In small children: adopting of a frog-leg position when sitting (hips abducted and knees flexed); loss of strength of the respiratory muscles: weak cough, weak
cry (infants), accumulation of secretions in the lungs or throat, respiratory distress; bellshaped torso (caused by using only abdominal muscles for respiration) in severe SMA type; fasciculations (twitching) of the tongue; and difficulty sucking or swallowing, poor feeding).
In some embodiments, the method comprises administering to a subject a suspension of an rAAV as described herein in a formulation buffer at a dose of about 1 x 1011 to about 1 x 1014 GC/kg body weight. In certain embodiments, the method comprises intravenous administration at a dose ranging from about 1 x 1012 genome copies (GC)/kg of rAAV to about 1 x 1014 GC of rAAV per kg. In certain embodiments, a dose is about 1 x 1013 GC/kg to about 1 x 1014 GC of rAAV per patient, or about 3 x 1013 GC/kg. In certain embodiments, delivery via intravenous administration is contemplated with a dose of about 3 x 1012 GC/kg to about 1 x 1014 GC/kg, further including doses of about 3.0 x 1013 GC/kg and about 1.0 x 1013 GC/kg.
In certain embodiments, the method comprises administering to a subject a suspension of an rAAV as described herein in a formulation buffer at a dose of about 1 x 109 GC per gram of brain mass to about 1 x 1013 GC per gram of brain mass. In certain embodiments, the method comprises administration of an rAAV as described herein at a dose of about 1 x 1011 GC per gram of brain mass to about 6 x 1011 GC per gram of brain mass. In certain embodiments, the method comprises administration of an rAAV as described herein at a dose of about 1 x 1011 GC per gram of brain mass. In certain embodiments, the method comprises administration of an rAAV as described herein at a dose of about 3.3 x 1011 GC per gram of brain mass. In certain embodiments, the method comprises administration of an rAAV as described herein at a dose of about 6.7 x 1011 GC per gram of brain mass.
In certain embodiments, the subject is delivered a therapeutically effective amount of an rAAV described herein. As used herein, a “therapeutically effective amount” refers to the amount of the composition comprising the nucleic acid sequence encoding SMN1 that delivers and expresses in the target cells an amount of the gene product sufficient to achieve efficacy. The dosage is adjusted to balance the therapeutic benefit against any side effects and such dosages may vary depending upon the therapeutic application for which the recombinant vector is employed. The levels of expression of the transgene product can be monitored to determine the frequency of dosage resulting in viral vectors, preferably AAV vectors containing the transgene. Optionally, dosage regimens similar to those described for
therapeutic purposes may be utilized for immunization using the compositions described herein.
Suitable, conventional, and pharmaceutically acceptable routes of administration include, but are not limited to, direct delivery to a desired organ (e.g., brain, CSF, heart), intranasal, intrathecal, intratracheal, intraarterial, intraocular, intravenous, intramuscular, subcutaneous, intradermal, intraparenchymal, intracerebroventricular, intrathecal, ICM, lumbar puncture and other parenteral routes of administration. Routes of administration may be combined, if desired.
As used herein, the term “administration” or any grammatical variations thereof refers to delivery of a composition described herein to a subject.
Suitable volumes for delivery of these doses and concentrations may be determined by one of skill in the art. For example, volumes of about 1 pL to 150 mL may be selected for CNS delivery, with the higher volumes being selected for adults. Typically, for newborn infants a suitable volume is about 0.5 mL to about 10 mL, for older infants, about 0.5 mL to about 15 mL may be selected. For toddlers, a volume of about 0.5 mL to about 20 mL may be selected. For children, volumes of up to about 30 mL may be selected. For pre-teens and teens, volumes up to about 50 mL may be selected. In still other embodiments, a patient may receive an intrathecal administration in a volume of about 5 mL to about 15 mL are selected, or about 7.5 mL to about 10 mL.
In certain embodiments, the method comprises administering a rAAV as described herein to a subject. The rAAV, preferably suspended in a physiologically compatible carrier, may be administered to a subject, human or non-human mammalian patient. In certain embodiments, for administration to a human patient, the rAAV is suitably suspended in an aqueous solution containing saline, a surfactant, and a physiologically compatible salt or mixture of salts. Suitably, the formulation is adjusted to a physiologically acceptable pH, e.g., in the range of pH 6 to 9, or pH 6.5 to 7.5, pH 7.0 to 7.7, or pH 7.2 to 7.8. As the pH of the cerebrospinal fluid is about 7.28 to about 7.32, for intrathecal delivery, a pH within this range may be desired; whereas for intravenous delivery, a pH of about 6.8 to about 7.2 may be desired. For intravenous delivery, a pH of about 6.8 to about 7.2 may be desired. However, other pHs within the broadest ranges and these subranges may be selected for other route of delivery.
Suitably, the pharmaceutical compositions, as described herein, and the uses thereof comprise delivering to the subject via injection, osmotic pump, intrathecal catheter, or for delivery by another device or route. In one example, the composition is formulated for intrathecal delivery.
As used herein, the terms “intrathecal delivery” or “intrathecal administration” refer to a route of administration via an injection into the spinal canal, more specifically into the subarachnoid space so that it reaches the cerebrospinal fluid (CSF). Intrathecal delivery may include lumbar puncture, intraventricular (including intracerebroventricular (ICV)), suboccipital/intracisternal, and/or Cl-2 puncture. For example, material may be introduced for diffusion throughout the subarachnoid space by means of lumbar puncture. In another example, injection may be into the cistema magna. In certain embodiment, a rAAV vector, or composition as described herein is administrated to a subject in need via the intrathecal administration. In certain embodiments, the intrathecal administration is performed as described in US Patent Publication No. 2018/0339065 Al, published November 29, 2019, which is incorporated herein by reference in its entirety. In certain embodiments, the CNS administration is performed using Ommaya Reservoir (also referred to as Ommaya device or Ommaya system).
As used herein, the terms “intraci sternal delivery” or “intracisternal administration” refer to a route of administration for drugs directly into the cerebrospinal fluid of the cisterna magna cerebellomedularis, more specifically via a suboccipital puncture or by direct injection into the cisterna magna or via permanently positioned tube.
In certain embodiments, administration of a rAAV vector provided herein is associated with reduced toxicity, which can include, for example, one or more of reduced neuronal degeneration, reduced spinal cord axonopathy, reduced dorsal root ganglia (DRG) toxicity (including, e.g., lesions characterized by neuronal degeneration/necrosis, nerve fiber degeneration, and/or mononuclear cell infiltration), reduced NfL levels in serum and/or CSF, reduced liver toxicity or risk of liver injury or failure. In certain embodiments, the toxicity is reduced relative to another gene therapy for delivery of SMN1, for example a gene therapy that requires an alternative AVV capsid, is an scAAV, utilizes an alternative promoter that is not a UbC promoter, has a vector genome that does not include miRNA target sequences, and/or is delivered by an alternative route of administration, in particular intravenous
administration. In certain embodiments, the reduced toxicity allows for a lower dose of the rAAV provided herein to be delivered, relative to another gene therapy for delivery of SMN1.
Increased levels of NfL have been detected following administration of gene therapies (Alves CRR, et al. Implications of circulating neurofilaments for spinal muscular atrophy treatment early in life: A case series. Mol Ther Methods Clin Dev. 2021 Oct 30;23:524-538). In certain embodiments, the methods provided include obtaining one or more measurements of an NfL level in a biological sample obtained from the subject. In certain embodiments, administration of an rAAV provided herein results in a reduced or undetectable increase in NfL levels in a biological sample (e.g., serum, plasma, and/or CSF) as compared to an alternative gene therapy for delivery of SMN1. In certain embodiments, the methods provided include obtaining one or more measurements of serum myostatin levels in a biological sample obtained from the subject. In certain embodiments, administration of an rAAV provided herein results in lower levels of serum myostatin levels in a biological sample (e.g., serum, plasma, and/or CSF) as compared to an alternative gene therapy for delivery of SMN1. Biological samples may be obtained prior to in addition to following administration of the rAAV vector to evaluate relative changes.
In a further aspect, suitably, the compositions of the invention are designed so that rAAV vectors carry the nucleic acid expression cassettes including an SMN1 encoding sequence and regulatory sequences that direct expression of the SMN1 in a target cell. The use of compositions described herein in methods to treat SMA are described, as are uses of these compositions in regimens, which may include delivery of one or more other cotherapies.
In certain embodiments, the method includes administering a co-therapy that is an immunosuppressant to the subject. Immunosuppressants for such co-therapy include, but are not limited to, a glucocorticoid, steroids or corticosteroids, antimetabolites, T-cell inhibitors, a macrolide (e.g., a rapamycin or rapalog), and cytostatic agents including an alkylating agent, an anti-metabolite, a cytotoxic antibiotic, an antibody, or an agent active on immunophilin. The immune suppressant may include a nitrogen mustard, nitrosourea, platinum compound, methotrexate, azathioprine, mycophenolate mofetil , methotrexate, leflunomide (Arava), cyclophosphamide, chlorambucil (Leukeran), a chloroquine (e.g., hydroxychloroquine), quinine sulfate, mefloquine, a combination of atovaquone and proguanil, sulfasalazine, mercaptopurine, fluorouracil, dactinomycin, an anthracycline,
mitomycin C, bleomycin, mithramycin, IL-2 receptor- (CD25-) or CD3-directed antibodies, anti-IL-2 antibodies, ciclosporinabatacept (Orencia), adalimumab (Humira), anakinra (Kineret), certolizumab (Cimzia), etanercept (Enbrel), golimumab (Simponi), infliximab (Remicade), rituximab (Rituxan), tocilizumab (Actemra) and tofacitinib (Xeljanz), cyclosporine, tacrolimus, sirolimus, IFN-P, IFN-y, an opioid, or TNF-a (tumor necrosis factor-alpha) binding agent, and combinations of these drugs .
In certain embodiments, the immunosuppressive therapy may be started 0, 1, 2, 7, or more days prior to administration of the rAAV vector. Such therapy may involve coadministration of two or more drugs, the (e.g., prednisone, micophenolate mofetil (MMF) and/or sirolimus (i.e., rapamycin)) on the same day. One or more of these drugs may be continued after gene therapy administration, at the same dose or an adjusted dose. Such therapy may be for about 1 week (7 days), about 60 days, or longer, as needed. In certain embodiments, a tacrolimus-free regimen is selected.
In certain embodiments, the requirement for treatment with an immunosuppressant as required for another SMA gene therapy, in terms of dose and/or duration is reduced or eliminated. In certain embodiments, the methods provided include treatment with an immunosuppressant for no longer than about one week, about two weeks, about three weeks, or about one month following administration of the rAAV vector.
In certain embodiments, the method comprises a combined treatment that includes administering a rAAV vector as described herein that includes an SMN1 coding sequence in combination with an antisense oligonucleotide (ASO) that increases full-length SMN2 mRNA in a subject (e.g., an ASO that promotes the inclusion of exon 7 in SMN2 mRNA) (See US 2021/0308281 Al, which is incorporated herein by reference). In certain embodiments, the SMN2 ASO is SPINRAZA® (nusinersen). The rAAV and ASO may be combined in a single formulation or administered separately.
In certain embodiments, the method comprises a combined treatment that includes administering a rAAV vector as described herein that includes an SMN1 coding sequence in combination with one or more additional agent that are olesoxime, AVX-101, CK-2127107, RG7916, RG7800, R07034067, LMI070, SRK-015, a p38aDMAPK inhibitor, a DcpS inhibitor, or a INK inhibitor. In certain embodiments, the method comprises a combined treatment that includes an agent that is a neuroprotective compound such as olesoxime, thyrotropin-releasing hormone, riulzole, certain beta-lactam antibiotics such as ceflt4riaxone,
or follistatin. In certain embodiments, the method comprises a combined treatment that includes an agent that promotes muscle restoration, such a reldesemtiv (skeletal troponin activator), apitegromab (Mab that blocks myostatin), GYM329 (blocks myostatin activation). The rAAV and the additional agent(s) may be combined in a single formulation or administered separately.
In certain embodiments, the method comprises a combined treatment that includes administering an rAAV vector as described herein that includes an SMN1 coding sequence in combination with a small molecule that promotes SMN function (See US 2022/0280548 Al, which is incorporated herein by reference). The rAAV and the small molecule may be combined in a single formulation or administered separately. In certain embodiments, the small molecule is a splice modulator, an HD AC inhibitor, or a molecule that modulates the activity of an mRNA decapping enzyme. In certain embodiments, the small molecule is an SMN2 splice modulator. In certain embodiments, the splice modulator is a 7-Disubstituted- phenyl tetracycline. In certain embodiments, the splice modulator is a substituted isoindolinone. In certain embodiments, the splice modulator is a substituted carbazole derivative. In certain embodiments, the SMN2 splice modulators is a substituted 1, 4- diazepane. In certain embodiments, the SMN2 splice modulators is a substituted pyridazine. In certain embodiments, the SMN2 splice modulator is Risdiplam. In certain embodiments, the SMN2 splice modulator is Branaplam.
In certain embodiments, the method includes administering anti-AAV neutralizing antibodies (NAb) to reduce peripheral transduction, and to mitigate the potential risk of AAV-induced toxicity. In certain embodiments, the method includes detecting the presence of systemic AAV NAb prior to treating with anti-AAV NAb, wherein patients with levels of anti-AAV NAb in excess of a predetermined level against the rAAV capsid (or a sero- crossreactive capsid) do not require pretreatment. Such levels may be, e.g., in excess of about 1 : 10, about 1 :20, about 1 :50, about 1 : 100, about 1 :250, or higher or lower levels. In certain embodiments, the method further comprises intravenously administering human anti-AAV polyclonal antibodies (e.g., plasma-derived, pooled human immunoglobulin (IVIG)), an anti- AAV monoclonal antibody, or a cocktail of anti-AAV antibodies, to a patient about 1 day to about 2 hours before treatment with the rAAV. In certain embodiments, the methods of treatment provided herein are not restricted by the presence of anti-AAV NAb detected in a subject, such that efficacy of the treatment is not limited and/or there is no requirement to
exclude or delay treatment of a subject that is found to have detectable levels of anti-AAV NAb.
In certain embodiments, a combination regimen is provided for preventing off-target delivery rAAV, the regimen comprising (a) pretreating the patient by systemically administering a composition comprising anti-AAV capsid neutralizing antibodies directed against an AAV capsid in a recombinant AAV vector, and (b) administering the rAAV as described herein. See also, US Provisional Patent Application No. 63/328,227, filed April 6, 2022, which is incorporated herein by reference in its entirety.
As used herein, a “neutralizing antibody” or “NAb” binds specifically to a viral capsid or envelope and interferes with the infectivity of the virus or a recombinant viral vector having the viral capsid or envelope, thus preventing the recombinant viral vector from delivering effective amounts of a gene product encoded by an expression cassette in its vector genome. Various methods for assessing neutralizing antibodies in a patient’s sera may be utilized. The term method and assay may be used interchangeably. As used herein, the term “neutralization assay” and “serum virus neutralization assay” refers to a serological test to detect the presence of systemic antibodies that may prevent infectivity of a virus. Such assays may also qualitatively or quantitatively discern the binding capacity (e.g., magnitude) or efficiency of the antibodies to neutralize a target. Immunological assays may include enzyme immunoassay (EIA), radioimmunoassay (RIA), which uses radioactive isotopes, fluoroimmunoassay (FIA) which uses fluorescent materials, chemiluminescent immunoassay (CLIA) which uses chemiluminescent materials and counting immunoassay (CIA) which employs particle-counting techniques, other modified assays such as western blot, immunohistochemistry (IHC) and agglutination. One of the most common enzyme immunoassays is enzyme-linked immunosorbent assay (ELISA).
Example of suitable methods include those described, e.g., R Calcedo, et al, Journal Infectious Diseases, 2009, 199:381-290; GUO, et al., “Rapid AAV_Neutralizing Antibody Determination with a Cell-Binding Assay”, Molecular Therapy: Methods & Clinical Development Vol. 13 June 2019, T. Ito et al, “A convenient enzyme-linked immunosorbent assay for rapid screening of anti-adeno-associated virus neutralizing antibodies”, Ann Clin Biochem 2009; 46: 508-510; US 2018/0356394A2 (Voyager Therapeutics). Additionally, commercial kits exist (see, e.g., Athena Diagnostics, Invitrogen, ThermoFisher.com; Covance).
The neutralization ability of an antibody is usually measured via the expression of a reporter gene such as luciferase or GFP. In order to determine and compare the activity of a neutralizing antibody, the antibody tested should display a neutralizing activity of 50% or more in one of the neutralization assays described herein. In some examples, neutralizing capacity is determined by measuring the activity of a reporter gene product (e.g., luciferase, GFP). The neutralizing capacity of an antibody to a specific viral vector may be at least 50%, e.g., at least 55%, 60%, 65%, 66%, 67%, 68%, 69%, 70%, 71%, 72%, 73%, 74%, 75%, 76%, 77%, 78%, 79%, 80%, 81%, 82%, 83%, 84%, 85%, 86%, 87%, 88%, 89%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or at least 99%.
As used herein, the term “NAb titer” a measurement of how much neutralizing antibody (e.g., anti-AAV Nab) is produced which neutralizes the physiologic effect of its targeted epitope (e.g., an AAV). Anti-AAV NAb titers may be measured as described in, e.g., Calcedo, R., et al., Worldwide Epidemiology of Neutralizing Antibodies to Adeno- Associated Viruses. Journal of Infectious Diseases, 2009. 199(3): p. 381-390, which is incorporated by reference herein.
Still other co-therapeutics may include, e.g., anti-IgG enzymes, which have been described as being useful for depleting anti-AAV antibodies (and thus may permit administration to patients testing above a threshold level of antibody for the selected AAV capsid), and/or delivery of anti-FcRN antibodies and/or one or more of a) a steroid or combination of steroids and/or (b) an IgG-cleaving enzyme, (c) an inhibitor of Fc-IgE binding; (d) an inhibitor of Fc-IgM binding; (e) an inhibitor of Fc-IgA binding; and/or (f) gamma interferon. anti-FcRN antibodies include, e.g., rozanolixizumab (UCB7665) (UCB SA); IMVT-1401, RVT-1401 (HL 161), HBM9161 (all form HanAll BioPhrma Co. Ltd), Nipocalimab (M281) (Momenta Pharmaceuticals Inc), ARGX-113 (efgartigimod) (Argenx S.E.), orilanolimab (ALXN 1830, SYNT001, Al exion Pharmaceuticals Inc), SYNT002, ABY-039 (Affibody AB), or DX-2507 (Takeda Pharmaceutical Co. Ltd). In certain embodiments, a combination of anti-FcRN antibodies is administered. In certain embodiments, an anti-FcRN antibody is administered in combination with a suitable anti- FcRn ligand (i.e., a peptide or protein construct binding human FcRn so as to inhibit IgG binding).
In certain embodiments, an anti-FcRn ligand(s) (e.g., antibodies) is administered to a patient having neutralizing antibodies prior to and, optionally, concurrently with a selected
viral vector. In certain embodiments, continued expression of an anti-FcRn ligand post administration of the gene therapy vector may desired on a short-term (transient basis), e.g., until such time as the viral vector clears from the patient. In certain embodiments, persistent expression of an anti-FcRn ligand may be desired. Optionally, in this embodiment, the ligand may be delivered via a viral vector, including, e.g., in the viral vector expressing the therapeutic transgene. However, this embodiment is not desirable where the therapeutic gene being delivered is an antibody or antibody construct or another construct comprising an IgG chain. In such embodiments, where an antibody construct having an IgG chain is being delivered via a viral vector to a patient having pre-existing immunity, the anti-FcRn ligand is delivered or dosed transiently so that the amount of anti-FcRn ligand in the circulation is cleared from the sera before effective levels of vector-mediated transgene product are expressed.
In certain embodiments, the FcRn ligand is delivered one to seven days prior to administration of the vector (e.g., rAAV). In certain embodiments, the FcRn ligand is delivered daily. In certain embodiments, the FcRn ligand (e.g., immunoglobulin construct(s)) is delivered on the same day as the vector is administered. In certain embodiments, the FcRn ligand (e.g., immunoglobulin construct(s)) is delivered at least one day to four weeks post- rAAV administration. In certain embodiments, the ligand is delivered for four weeks to six months post-rAAV administration. In certain embodiments, the ligand is dosed via a different route of administration than the rAAV. In certain embodiments, the ligand is dosed orally, intravenously, or intraperitoneally. See also, International Patent Application No. PCT/US2021/037575, filed June 16, 2021, and now published WO 2021/257668 Al, which is incorporated herein by reference in its entirety.
In certain embodiments, the method includes measuring a neurofilament level (e.g., pNF-H level). The method involves providing a biological sample obtained from a human subject having mutations in both copies of the SMN1 gene that lead to functional SMN protein deficiency; and measuring a neurofilament level (e.g., pNF-H level) in the biological sample. In certain embodiments, the method involves: measuring a neurofilament level (e.g., pNF-H level) in a first biological sample obtained from the human subject before administering a rAAV as described herein that delivers an SMN1 coding sequence; administering the rAAV (e.g., a therapeutically effective amount of the rAAV) to the human subject; and measuring a neurofilament level (e.g., pNF-H level) in a second biological
sample obtained from the human subject after administration of the rAAV. In certain embodiments, the neurofilament is a neurofilament heavy chain (e.g., phosphorylated NF-H). In certain embodiments, the neurofilament is a neurofilament medium/intermediate chain. In certain embodiments, the neurofilament is a neurofilament light chain. In certain embodiments, the neurofilament is intemexin. In certain embodiments, the neurofilament is peripherin. See US 2023/0107651 Al, which is incorporated herein by reference.
As used herein, the term “biological sample” refers to any cell, biological fluid or tissue. Suitable samples for use in this invention may include, without limitation, whole blood, leukocytes, fibroblasts, serum, urine, plasma, saliva, bone marrow, cerebrospinal fluid, amniotic fluid, and skin cells. Such samples may further be diluted with saline, buffer or a physiologically acceptable diluent. Alternatively, such samples are concentrated by conventional means.
Kits
In certain embodiments, a kit is provided which includes a concentrated vector suspended in a formulation (optionally frozen), optional dilution buffer, and devices and components required for intrathecal, intracerebroventricular or intracistemal administration. In another embodiment, the kit may additional or alternatively include components for intravenous delivery. In one embodiment, the kit provides sufficient buffer to allow for injection. Such buffer may allow for about a 1 :1 to a 1 :5 dilution of the concentrated vector, or more. In other embodiments, higher or lower amounts of buffer or sterile water are included to allow for dose titration and other adjustments by the treating clinician. In still other embodiments, one or more components of the device are included in the kit. Suitable dilution buffer is available, such as, a saline, a phosphate buffered saline (PBS) or a glycerol/PBS.
Apparatus and Method for Delivery of a Pharmaceutical Composition
In one aspect, the vectors, rAAV, or compositions thereof provided herein may be administered intrathecally via the method and/or the device provided in this section and described in WO 2017/136500 and WO 2018/160582, which are incorporated by reference herein. Alternatively, other devices and methods may be selected. In certain embodiments, the method comprises the steps of CT-guided sub-occipital injection via spinal needle into the
cistema magna of a patient. As used herein, the term Computed Tomography (CT) refers to radiography in which a three-dimensional image of a body structure is constructed by computer from a series of plane cross-sectional images made along an axis. In certain embodiments, the apparatus is described in US Patent Publication No. 2018-0339065 Al, published November 29, 2019, which is incorporated herein by reference in its entirety. In certain embodiments, the vectors, rAAV or compositions thereof provided herein may be administered using an Ommaya Reservoir.
The following examples are provided to illustrate certain aspects of the claimed invention. The invention is not limited to these examples.
EXAMPLES
Example 1 : Development of improved AAV vectors for delivery of an hSMNl transgene A series of recombinant vector genomes were engineered and packaged in a
AAVhu68 capsid. The constructs included that same SMN1 coding sequence but different regulatory sequences, including combinations of one of four promoters CB7, UbC, Jet, and hSyn, intron sequences, and miR182 target sequences. The vector genomes and their components are depicted in FIG 1.
UbC. PI. hSMNl co. rBG
UbC.PI.hSMNlco.4xmiR182.rBG (SEQ ID NO: 2) CB7.CI.hSMNlco.4xmiR182.rBG (SEQ ID NO: 1) hSYN.hSMNlco.4xmiR182.rBG (SEQ ID NO: 3) Jet.hSMNlco.4xmiR182.rBG (SEQ ID NO: 4)
Description of the Vector Elements:
1. Inverted terminal repeats (ITR): AAV ITRs are sequences that are identical on both ends, but in opposite orientation. The AAV2 ITR sequences function as both the origin of vector DNA replication and the packaging signal of the vector genome, when AAV and adenovirus helper functions are provided in trans. As such, the ITR sequences represent the only cis sequences required for vector genome replication and packaging.
2. Promoters: (i) CB7 promoter (a cytomegalovirus immediate-early (CMV IE) enhancer and the chicken P-actin promoter); (ii) ubiquitin C (UbC) promoter (SEQ ID NO: 10); (iii) human synapsin 1 (hSYN) promoter; and (iv) JeT promoter (synthetic promoter).
3. Introns: (i) chimeric chicken P-actin intron (CI); and (ii): intron (PI) (SEQ ID NO: 11) (see also GenBank: U47121.2).
4. hSMNl coding sequence: engineered SMN1 coding sequence (SEQ ID NO: 7).
5. miR182 target sequences: a tandem repeat of four target sequences (each having the nucleotide sequence set forth in SEQ ID NO: 12) that are capable of being bound by miR182 to promote a DRG-specific reduction of transgene expression and toxicity (as described, for example, in WO 2020/132455 Al and WO 2021/231579 Al).
6. Polyadenylation Signal (poly A): The rabbit P-globin polyadenylation signal (rBG) provides cis sequences for efficient polyadenylation of the antibody mRNA. This element functions as a signal for transcriptional termination, a specific cleavage event at the 3’ end of the nascent transcript and addition of a long polyadenyl tail. (SEQ ID NO: 13).
AAVhu68 vectors were prepared using conventional triple transfection techniques in 293 cells as described [Mizukami, Hiroaki, et al. A Protocol for AAV vector production and purification. Diss. Division of Genetic Therapeutics, Center for Molecular Medicine, 1998; Lock, M., et al, Hum Gene Ther, 21 : 1259-1271 (2010)]. trans plasmid: pAAV2/hu68n.KanR helper plasmid: pAdDeltaF6(Kan)
Example 2: AAVhu68. hSMNl Vector Potency Study in Mice
A study was conducted to evaluate the expression of hSMNl following delivery of AAVhu68 vectors. Groups of C57BL/6J mice were injected IV with IxlO12 GC of one of the following vectors:
AAVhu68.CB7.hSMNl.rBG
AAVhu68.CB7.hSMNl.4XmiR182.rBG
AAVhu68. Jet.hSMNl.4XmiR182.rBG
AAVhu68.Ubc.hSMNl.4XmiR182.rBG
On day 7, mice were euthanized and liver tissue was harvested for detection of hSMNl transcript by ISH. IV administered WT mice showed that the CB7 promoter generated the highest expression of hSMNl, followed by the Ubc promoter and the Jet promoter. The presence of four sequence targets for miR182 into the vector genome within the 3' untranslated region of the corresponding transgene messenger RNA, which are added to the vectors to prevent transgene overexpression dependent DRG toxicity, did not impact the level of hSMNl expression generated by the CB7 promoter (FIG. 2).
This study demonstrated the superiority of the CB7 promoter over other promoters in generating high levels of hSMNl expression in the liver following IV administration of the different vectors tested. For safety reasons, weaker expression in liver such as those achieved with Ubc and Jet promoters is favorable.
An additional study was conducted using an SMA mouse model, FVB.Cg- Grm7Tg.(SMN2)89Ahmb SmnltmlMsdTg(SMN2*delta7)4299Ahmb/J., also referred as SMN A 7 mice. This triple mutant mouse harbors two transgenic alleles and a single targeted null mutant (the mice lack murine Smn and express an intact human SMN2 gene plus SMN2A7 cDNA). The mice exhibit low birth weight and at week-1 display an abnormal gait, shakiness in the hind limbs, and a tendency to fall over. The mean survival of these mice is approximately 17 days.
Neonate (PO) SMN A 7 mice were administered IxlO11 GC (ICV) of one of the following vectors:
AAVhu68.CB7.hSMNl.rBG, n=15
AAVhu68.CB7.hSMNl.4XmiR182.rBG, n=13
AAVhu68.Jet.hSMNl.4XmiR182.rBG, n=10
AAVhu68.Syn.hSMNl.4XmiR182.rBG, n=9 AAVhu68.Ubc.hSMNl.4XmiR182.rBG, n=9 In life read outs included monitoring survival, body weight, right reflex, and clasping. Mice were euthanized on day 120 and tissues were harvested for determination of transgene (hSMNl) expression by ISH and potential toxicity, particularly in spinal cord and dorsal root ganglia (DRG).
WT (C57BL6/J) and SMNA7 mice were administered PBS as controls. Median survival of SMNA7 mice was 12 days when administered PBS only.
In contrast, the median survival of SMNA7 mice was massively increased when animals were ICV administered AAVhu68.UbcTI.hSMNl.4xmiR182.rBG. Because 55% of animals treated with AAVhu68.Ubc.PI.hSMNlco.4xmiR182.rBG survived until the study end point at Day 120, median survival could not be defined, but was at least 10-fold the median survival with PBS only (FIG. 3).
Median survival was also increased to 39 days with AAVhu68.CB7.hSMNl.rBG and 26 days with AAVhu68.CB7.hSMNl.4xmiR182.rBG.
However, ICV administration of AAVhu68.Jet.hSMNl.4xmiR182.rBG and AAVhu68.hSyn.hSMNlco.4xmiR182.rBG barely changed median survival of SMNA7 mice, with values of 14 and 15 days, respectively.
SMNA7 mice treated with AAVhu68.Ubc.PI.hSMN1.4xmiR182.rBG also demonstrated a gradual improvement in body weights that was better than with the other vectors (FIG. 4).
In addition to best survival, AAVhu68.Ubc.PI.hSMN1.4xmiR182.rBG treated mice also showed normalized righting reflex and improved clasping scores, a measure of compromised information about limb and joint position, altered reflexes, or improper integration of sensory and motor information including dysfunction of proprioceptive neurons (FIG. 5).
Importantly, AAVhu68.Ubc.PI.hSMN1.4xmiR182.rBG, and other vectors, did not cause toxicity in healthy littermates (smn+/+) in terms of survival, body weight gain, and clasping reflex, the latter demonstrating that SMN overexpression was not associated with motor and sensory circuit toxicity (FIG. 6).
Survival rescue was also associated with higher average motor neuron transduction (52 % of ChAT+ cells express hSMNl by ISH) (FIG. 7A) and a better cardiac pathology rescue in SMNA7 mice treated with AAVhu68.Ubc.PI.hSMNlco.4xmiR182.rBG.KanR (p7097) compared to vehicle and other test articles treated mice (FIG. 7B).
Example 3: NHP study for expression and safety following administration of SMN1 AAV vectors
A safety and toxicity study in NHP was conducted with the following three test articles ICM administered at a dose of 3.0 x 1013 GC (3.3 x 1011 GC/g brain) to adult rhesus macaques (6-7 years old), three animals per group, both genders:
AAVhu68.CB7.hSMNl.rBG
AAVhu68.CB7.hSMNl.4XmiR182.rBG
AAVhu68.Ubc.hSMNl.4XmiR182.rBG
In life evaluations included behavioral observations, clinical pathology studies, CSF collections, and nerve conduction studies. Animals were euthanized on day 35 and tissues were harvested for detection of hSMNl expression by ISH and H&E staining for pathology. Immune responses to the transgene and vector were also measured.
Expression of hSMNl in motor neurons following ICM administration was verified by hSMNl and ChAT ISH. Overall, the number of hSMNl positive cells was comparable in all three groups, as expected given the use of the same capsid, although the signal intensity was weaker with the UbC promoter, as expected given the relative strengths of the CB7 and UbC promoters (FIG. 8A and FIG. 8B). Based on the improved survival and rescue of phenotype in rodent studies with 52% of motor neuron transduction, the proportion of hSMNl+ cells detected in UbC. hSMNl.4xmiRl 82 administered animals (45%) is likely to lead to benefit in patients.
The clinical pathology assessment revealed a mild asymptomatic pleocytosis in the CSF of animals treated with the vector containing the Ubc promoter (AAVhu68.UbC.PI.hSMN1.4xmiR182.rBG) (FIG. 9).
Nerve conduction velocity and sensory nerve action potential amplitude (SNAP) were not affected by administration of any of the vectors (FIG. 10) and neuronal degeneration was minimal to mild and similar across the three different promoters (FIG. 11).
When aggregating data from all the histology sections scored within groups, AAVhu68.UbC. hSMNl.4XmiRl 82 led to the least levels of spinal cord dorsal axonopathy, and the lowest CSF NfL elevation at Day 28, consistent with a favorable DRG toxicity profile (FIG. 12; FIG. 13A and FIG. 13B). The lower NfL fold change in AAVhu68.UbC. hSMNl.4XmiRl 82 treated animals was consistent with lower axonopathy scores (FIG. 14A and FIG. 14B).
Blood chemistry was unremarkable in treated macaques over the 35-day study period. No animal presented with a clinically remarkable condition or neurological concern from cage side assessments. Blood clotting was normal, with the exception of small, transient increases in clotting factor fibrinogen. Biomarkers of liver and kidney damage and circulating
ions and metabolites were all within normal ranges in treated monkeys. Treatment with the three vectors showed no evidence of toxicity.
An increase in neutralizing and binding antibody titers was observed, with the highest levels being detected on Day 35 in all treated animals. The increase in antibody titers was not associated with any clinical complications. ELISPOT testing identified moderate immune responses against AAVhu68 peptide pool C in PBMC and spleen. Overall, immune responses against AAVhu68 were milder in the Ubc.4XmiR182 and CB7.4XmiR182 promoter groups. Immune responses to AAVhu68 did not lead to any adverse events in the vector treated animals. ELISPOT testing identified moderate immune responses against hSMNl peptides in PBMCs, liver and spleen. Overall, immune responses against hSMNl were milder in the Ubc.4XmiR182 and CB7.4XmiR182 promoter groups. Immune responses to AAVhu68 did not lead to any adverse events in the vector treated animals.
Anti -transgene immunogenicity can confound results of NHP studies due to non-selfresponses but is not a concern in the trial as patient are expected to be tolerant to self SMN coming from SMN2 transcription.
Example 4: Pharmacology and Safety Study of AAVhu68.Ubc.PI.hSMN1.4xmiR182.rBG to Determine the Minimum Effective Dose (MED) Following ICV Administration in SMNA7 Mice
A study was performed to evaluate efficacy and safety, and to determine the MED of ICV-administered AAVhu68.Ubc.PI.hSMN1.4xmiR182.rBG.
As summarized in the table below, the study evaluated N=120 neonatal (PND 0-1) AAVhu68.Ubc.PI. hSMNl.4xmiRl 82. rBG -treated SMNA7 mice (N=60) and wild type controls (N=60). The study included one necropsy time point (120 days post treatment).
Four dose levels of AAVhu68.Ubc.PI.hSMN1.4xmiR182.rBG were evaluated following ICV administration. The dose levels evaluated bracket the anticipated clinical doses.
MED Study Design
In-life assessments included viability checks performed daily, body weight measurements, and behavioral assessments (surface righting reflex and hindlimb clasping tests). Transgene product expression (by ISH and IF) was evaluated in the disease-relevant target tissues and highly transduced peripheral tissues.
The MED was determined based on analysis of improvement in survival, behavioral phenotypes, along with efficacy of transgene product expression in the target tissues of AAVhu68.Ubc.PI.hSMN1.4xmiR182.rBG -treated SMNA7 mice compared to vehicle-treated WT control mice. Administration of AAVhu68.Ubc.PI.hSMN1.4xmiR182.rBG at a higher dose (1.0 x 1011 GC) increased the survival of SMNA7 mice (FIG. 15).
AAVhu68.Ubc.PI.hSMN1.4xmiR182.rBG -treated wildtype mice did not exhibit evidence of
over expression toxicity until study day 120 (FIG. 16). Administration of AAVhu68.Ubc.PI.hSMN1.4xmiR182.rBG to SMNA7 mice at two doses (1.0 x 1011 GC and 5.0 x 109 GC) resulted in improvement of body weight (FIG. 17), while administration of the vector to wildtype mice did not impact body weight gain (FIG. 18).
Administration of AAVhu68.Ubc.PI.hSMN1.4xmiR182.rBG to SMNA7 mice at three doses resulted in improvements in righting reflex compared to untreated mice beginning at PND7 (FIG. 19). AAVhu68.Ubc.PI.hSMN1.4xmiR182.rBG -treated SMNA7 mice at two doses (1.0 x 1011 GC and 5.0 x 109 GC) exhibited improvement in clasping phenotype (FIG. 21). AAVhu68.Ubc.PI.hSMN1.4xmiR182.rBG -treated wildtype mice did not exhibit clasping phenotype, indicating that there was no overexpression toxicity (FIG. 20).
Example 5: Toxicology Study - AAVhu68.Ubc.PI.hSMN1.4xmiR182.rBG administered via intra-ci sterna magna (ICM) injection to juvenile rhesus macaques
Animal Model and Age of Animals
A GLP-compliant toxicology study was performed using juvenile (2-3 years old) NHPs. This age range allows for modeling the size and anatomy of the cisterna magna of the youngest intended patient population to the greatest extent feasible. The dimensions of the CNS in the NHP act as a representative model of our target clinical population and allow for the administration of AAVhu68.Ubc.PI.hSMN1.4xmiR182.rBG using the intended clinical route of administration. Furthermore, the toxicological and immune responses of the NHP closely resemble that of humans. This study provided critical data on route of administration- related safety and pharmacokinetics of the test article after administration to rhesus macaques.
Both sexes were selected to model the intended patient population in the planned clinical trial (male and female SMA patients).
Dose selection
The GLP-compliant toxicology study in juvenile rhesus macaques used a dose of 5.8 x 1013 GC corresponding to the highest dose (1 x 1011 GC/animal) used in the MED mouse study, which equates to approximately 6.7 x 1011 GC/g of brain for mice (assuming brain weight in newborn mice is 0.150g) and 6.0 x 1013 GC/g for juvenile rhesus (assuming brain weight of 90 grams). The dose also represents a close to technical maximal feasible
dose based on CSF safe maximal injection volume (1.5 mL) in juvenile rhesus macaques and vector titer constraints.
Study Duration
The GLP-compliant toxicology study in juvenile rhesus macaques was 90 days long. The 90-day duration of this study allowed for the confirmation of the non-progressive nature of the DRG pathology and to obtain chronic safety and immunology data.
Route of Administration
The ICM route was selected for the GLP-compliant toxicology study in juvenile rhesus macaques to mirror the intended clinical route and enable the use of a clinical administration system comparable to the one utilized in the planned clinical trial.
Abbreviations: BAb, binding antibodies; BT, Blue Top: Sodium Citrate; CSF, cerebrospinal fluid;
DRG, dorsal root ganglion; ELISPOT, enzyme-linked immunospot; GT, Green Top: Heparin; ITFFB, intrathecal final formulation buffer; kg, kilogram; LT, Lavender Top: K2EDTA; mg, milligram; NA, Not Applicable; NAb, neutralizing antibodies; PMBC, peripheral blood mononuclear cell; PK, pharmacokinetics; RC, Red Top: No anti-coagulant. a Number of animals assessed. b Includes complete blood counts and differentials (hematology), clinical chemistries, and coagulation panel.
Animals were visually observed daily for general appearance and signs of toxicity, which may include, but are not limited to, neurologic signs or lethargy, distress, and changes in behavior.
Throughout the study, at timepoints defined in the table above:
• animals underwent neurological monitoring,
• nerve conduction study test results were recorded from the left and right median nerves,
• animals were anesthetized and bled from a peripheral vein for coagulation panel, CBC, serum chemistry, neutralizing (NAb) and binding (BAb) antibodies to the hu68 capsid, peripheral blood mononuclear cells (PBMC) isolation, vector PK, and biomarker analysis,
• animals were anesthetized, transferred from the animal holding to the procedure room and placed in the lateral decubitus position with the head flexed forward for CSF collection for CSF clinical pathology, CSF biomarker, CSF NAb and/or CSF pharmacokinetics analysis.
After necropsy, a comprehensive list of tissues (CNS, PNS and peripheral organs) was evaluated by a board-certified Veterinary Pathologist, with peer review conducted by an independent board-certified pathologist.
Results
ICM administration of AAVhu68.Ubc.PI.hSMN1.4xmiR182.rBG to juvenile NHPs did not impact sensory nerve action potential (SNAP) or conduction velocity in the treated animals (FIG. 22). No CSF related abnormalities were identified following ICM administration of AA Vhu68.Ubc.PI.hSMN1.4xmiRl 82. rBG to juvenile NHPs (FIG. 23). Administration of the vector did not result in elevation of liver enzymes (FIG. 24) or impact liver functions (FIG. 23 A - FIG. 23 C). ICM Administration of AAVhu68.Ubc.PI.hSMN1.4xmiR182.rBG to juvenile NHPs did not impact coagulation parameters (FIG. 26). Overall, ICM administration of AAVhu68.Ubc.PI.hSMN1.4xmiR182.rBG to rhesus monkeys was safe and well-tolerated.
Example 6: Method for Scaling from Nonclinical Doses to Clinical Doses
Because ICM vector administration results in immediate vector distribution within the CSF compartment, it is anticipated that both efficacy and toxicity are related to CNS vector exposure. Doses are therefore scaled by brain mass, which provides an approximation of the size of the CSF compartment. Dose conversions between animals and patients in the FIH trial are based on a brain mass of 0.15 g for a neonatal mouse, 90 g for a juvenile NHP, 488 g for a 1-4 months old -month infant and 960 g for a >12- month child. Estimated brain weights for each age range for human infants are derived from the male and female brain weights presented in the table below. An example of dose scaling from neonatal mice, juvenile NHPs, and equivalent human doses are presented using doses used in the NHP toxicology and murine MED studies. The administration volume is also scaled from NHPs to humans based on the estimated volumes for cerebral CSF and spinal CSF so that less than 10% of the total CSF volume is being administered, consistent with NHP safety studies.
Example dose scaling from animals to humans based on brain mass
a Doses are scaled based on a brain mass of 0.15 g for a neonatal mouse, 90 g for a juvenile rhesus macaque, and 488 g for a 1-4-month-old human.
Based on scaling across species, the selected clinical doses are greater than the mouse MED, to support the prospect of direct benefit in the phase I trial and is lower than or equal to the highest tolerated dose in the GLP compliant NHP safety study. In other words, use of the optimal route of administration in rodents to demonstrate target engagement while the NHP study informs on the safety of the maximal feasible dose (MFD) with intrathecal dosing constraints (limited volume and vector titer). If the NHP study demonstrates safety, then patients are dosed close to the MFD, which is typically several folds over the MED obtained in mice.
Sensory Neuron Toxicity in Nonclinical Studies
Published data have shown that AAV vectors administered via the ICM route led to histopathological evidence of damage to peripheral sensory neuron and their associated axons. A meta-analysis of 33 nonclinical studies in 256 NHPs has shown that immunosuppression does not prevent DRG degeneration in NHPs, and T cell responses detected by ELISpot do not predict DRG degeneration. The transgene, time post injection, and dose appear to have the greatest impact on the severity of the sensory neuron pathology, suggesting that transgene overexpression drives the early events leading to neuronal degeneration. DRG pathology presents as mostly minimal to moderate asymptomatic DRG pathology characterized by mononuclear cell infiltrates, neuronal degeneration, and secondary axonopathy of central and peripheral axons. DRG pathology is similar from 1-5 months post injection and becomes less severe after 6 months. Higher AAV doses correlate
with increased severity. Younger NHPs (infants and juveniles) appear to exhibit less severe pathology compared to adult NHPs. Animal sex and vector purification method have no impact. In most studies, DRG sensory neuron degeneration occurs in the absence of toxicity to other heavily transduced cells (e.g., hepatocytes, cortical neurons, and spinal motor neurons), indicating that DRG sensory neurons are selectively vulnerable to AAV-mediated toxicity. In all cases, only a subset of DRG sensory neurons is affected, degenerating within 14-30 days after gene transfer. Following this acute insult, no further degeneration of transduced cells is detectable after 35 days, although slow degeneration of the associated axons can continue until 90 days. Findings are thereafter non progressive between 90 and 180 days with either similar or improved pathology scores.
Similar asymptomatic DRG microscopic findings (mononuclear cell inflammation sometimes associated with neuronal satellitosis or neuronal necrosis), with onset within weeks of dose administration but no progressive course, were observed 2 weeks after intrathecal administration to cynomolgus NHPs in nonclinical studies conducted in support of onasemnogene abeparvovec (Zolgensma) for the treatment of SMA. increased serum and CSF NfL levels were observed on day 22, with trends toward pre-dose and vehicle control concentrations by day 163 and resolution by day 365, and a correlation between CSF and serum NfL concentrations was observed (R2 = 0.9044).
Two recent studies have demonstrated that the biomarker NfL correlates with DRG sensory neuron degeneration. Fader and colleagues evaluated dose- and time-dependent NfL level changes following AAV gene therapy, comparing protein profiles with DRG pathology. Serum/plasma NfL levels were strongly associated with the severity of neuronal degeneration and axonal loss, with elevations commencing from day 8 in rodents and day 14 in monkeys.
Similarly, in studies conducted with the AAVhu68 capsid, administered via the ICM ROA in healthy NHPs, Hordeaux and colleagues observed that animals with histopathological findings of DRG pathology 2-to-3-months post-AAV administration exhibited elevated NfL levels in both CSF and serum three to four weeks post dosing that returned to low baseline values subsequently. The extent of the NfL spike observed at 1 month correlated remarkably well with the severity of spinal cord dorsal axonopathy seen at necropsy several weeks later, thereby offering a unique opportunity to monitor the extent of DRG pathology during the in-life phase. The observation of a NfL spike 3-to-4-weeks post
dosing, at peak transgene expression, followed by subsequent resolution closely matches the non-progressive nature of the DRG pathology.
In humans, however, clinical trials utilizing high doses of IV-administered AAV5, AAV8, or AAV9 vectors have not identified signs of sensory neuron toxicity. To date, there is only a single report of a patient who developed sensory symptoms approximately 3 weeks following IT administration of an AAVrhlO vector expressing an anti-SODl miRNA at a dose of 4.2xl014 GC (equivalent to 3.1xlOn GC/g brain). This patient, affected by ALS, exhibited reduced sensory nerve action potential amplitudes and had radiographic and eventually pathologic evidence of DRG degeneration. However, sensory symptoms are frequently reported in patients with ALS, and the link between the IT administration of the AAV vector and the development of sensory neuropathy was uncertain. Therefore, to date, no clear causation has ever been confirmed in humans between CNS administration of a recombinant AAV vector and sensory deficits.
Based on the existing nonclinical and clinical data for other AAV programs, the true risk of sensory neuron toxicity in humans is unknown but expected to be low. The proposed clinical trial is designed to further improve on the safety profile of previous AAV clinical trials by using an ICM ROA that requires lower doses of vector than those typically administered systemically to reduce the risk of sensory neuron toxicity. The planned clinical trial also employs detailed monitoring for sensory changes with a longitudinal monitoring of serum NfL levels to detect even subclinical DRG toxicity.
AAVhu68.Ubc.PI.hSMNlco.4xmiR182.rBG incorporates miR182 target sequences into the 3’ UTR of the human SMN1 sequence. These sequences were incorporated to downregulate transgene product expression in DRG in an effort to minimize the potential for AAV-associated sensory neuron toxicity, which is believed to be primarily caused by transgene product overexpression. MicroRNAs post-transcriptionally downregulate expression of target messenger ribonucleic acids (mRNAs) in multicellular organisms by affecting both mRNA stability and translation. Since miR182 expression is largely restricted to the dorsal root ganglia (DRG), the miR182 target sequences enable DRG-specific downregulation of the human SMN1 transgene product. This detargeting approach aims to minimize the potential for AAV-associated DRG sensory neuron toxicity.
The current clinical trial is designed to further improve on the safety profile of previous AAV clinical trials by using an ICM route that requires lower doses of vector than those typically administered systemically to reduce the risk of sensory neuron toxicity.
The planned clinical trial therefore employs detailed monitoring for sensory changes as well as nerve conduction studies to detect even subclinical DRG toxicity. In light of the severity of SMA, the benefit/risk profile for ICM administration of AAVhu68.Ubc.PI.hSMNlco.4xmiR182.rBG is expected to remain favorable.
Example 7: First-in-human (FH4) trial The FIH trial is an open-label, multi-center, dose escalation study of
AAVhu68.Ubc.PI.hSMNlco.4xmiR182.rBG administered via an intra-cistema magna (ICM) injection to evaluate safety, tolerability, and exploratory efficacy endpoints in pediatric patients with Type 1 SMA.
All documents cited in this specification are incorporated herein by reference. US Provisional Patent Application No. 63/518,796, filed August 10, 2023, and US Provisional Patent Application No. 63/662,509, filed June 21, 2024, are incorporated herein by reference. While the invention has been described with reference to particular embodiments, it will be appreciated that modifications can be made without departing from the spirit of the invention. Such modifications are intended to fall within the scope of the appended claims.
Claims
1. A recombinant adeno-associated virus (AAV) comprising an AAV capsid containing a vector genome, wherein the vector genome comprises an expression cassette comprising:
(a) a UbC promoter sequence,
(b) a coding sequence for a functional human survival of motor neuron 1
(SMN1) protein,
(c) at least four miR182 target sequences, and
(d) a polyA sequence, wherein the coding sequence, the at least four miR182 target sequences, and the polyA sequence are operably linked to the UbC promoter sequence.
2. The recombinant AAV of claim 1, wherein the SMN1 protein is an SMN1 isoform D protein.
3. The recombinant AAV of claim 1 or 2, wherein the coding sequence comprises the nucleotide sequence of SEQ ID NO: 7 or a nucleotide sequence at least 95% identical to SEQ ID NO: 7.
4. The recombinant AAV of any one of claims 1 to 3, wherein the AAV capsid is a clade F AAV capsid.
5. The recombinant AAV of any one of claims 1 to 4, wherein the AAV capsid is an AAVhu68 capsid.
6. The recombinant AAV of claim 5, wherein the AAVhu68 capsid comprises VP1, VP2, and VP3 proteins expressed from a nucleic acid sequence encoding the amino acid sequence of SEQ ID NO: 9 (amino acids 1 to 736).
7. The recombinant AAV of any one of claims 1 to 3, wherein the AAV capsid is an AAVhu95 capsid, an AAVhu96 capsid, or an AAV9 capsid.
8. The recombinant AAV of any one of claims 1 to 7, wherein the at least four miR182 target sequences are located 3’ of the SMN1 coding sequence and 5’ of the poly A sequence.
9. The recombinant AAV of any one of claims 1 to 7, wherein the at least four miR182 target sequences include at least one target sequence that is located 5’ of the UbC promoter and at least one target sequence that is located 3’ of the SMN1 coding sequence and 5’ of the poly A sequence.
10. The recombinant AAV of any one of claims 1 to 9, wherein the each of the miR182 target sequences comprises the nucleotide sequence of SEQ ID NO: 12.
11. The recombinant AAV of any one of claims 1 to 10, wherein the UbC promoter comprises the nucleotides sequence of SEQ ID NO: 10.
12. The recombinant AAV of any one of claims 1 to 11, wherein the polyA sequence is a rabbit beta-globin (rBG) polyA sequence.
13. The recombinant AAV of claim 12, wherein the rBG polyA sequence comprises the nucleotide sequence of SEQ ID NO: 13.
14. The recombinant AAV of any one of claims 1 to 13, wherein the expression cassette further comprises an intron.
15. The recombinant AAV of any one of claims 1 to 14, wherein the intron comprises the nucleotide sequence of SEQ ID NO: 11.
16. The recombinant AAV of any one of claims 1 to 15, wherein the vector genome further comprises an AAV 5’ ITR sequence and an AAV 3’ ITR sequence, optionally wherein the AAV 5’ ITR sequence and the AAV 3’ ITR sequence are from AAV2.
17. The recombinant AAV according to any one of claims 1 to 16, wherein the expression cassette comprises the nucleic acid sequence of SEQ ID NO: 16, or a nucleic acid sequence at least 99% identical to SEQ ID NO: 16.
18. The recombinant AAV of any one of claims 1 to 17, wherein the vector genome comprises nucleic acid sequence of SEQ ID NO: 2 or a sequence at least 99% identical to SEQ ID NO: 2.
19. The recombinant AAV of any one of claims 1 to 18, wherein the recombinant AAV is formulated in an aqueous suspension for systemic delivery and/or delivery to the central nervous system.
20. A composition comprising a stock of the recombinant AAV of any one of claims 1 to 18 and an aqueous suspension media.
21. The composition of claim 20, wherein the suspension is formulated for intrathecal delivery, optionally wherein the intrathecal delivery is intracerebroventricular (ICV) injection or intracistemal magna (ICM) injection.
22. A pharmaceutical composition comprising the recombinant AAV of any one of claims 1 to 19 and an aqueous formulation buffer.
23. The pharmaceutical composition of claim 22, which is formulated for intrathecal delivery.
24. The pharmaceutical composition of claim 22, which is formulated for intracerebroventricular (ICV) injection or intracistemal magna (ICM) injection.
25. A recombinant nucleic acid molecule comprising an expression cassette comprising:
(a) a 5’ AAV ITR sequence,
(b) a UbC promoter sequence,
(c) a coding sequence for a functional human survival of motor neuron 1
(SMN1) protein,
(d) at least four miR182 target sequences,
(e) a polyA sequence, and
(f) a 3 ’ AAV ITR sequence, wherein the coding sequence, the at least four miR182 target sequences, and the polyA sequence are operably linked to the UbC promoter sequence.
26. The recombinant nucleic acid molecule of claim 25, wherein the SMN1 protein is an SMN1 isoform D protein.
27. The recombinant nucleic acid molecule of claim 25 or 26, wherein the coding sequence comprises the nucleotide sequence of SEQ ID NO: 7 or a nucleotide sequence at least 95% identical to SEQ ID NO: 7.
28. The recombinant nucleic acid molecule of any one of claims 25 to 27, wherein the at least at least four miR182 target sequences are located 3’ of the SMN1 coding sequence and 5’ of the polyA sequence.
29. The recombinant nucleic acid molecule of any one of claims 25 to 27, wherein the at least four miR182 target sequences include at least one target sequence that is located 5’ of the UbC promoter and at least one target sequence that is located 3’ of the SMN1 coding sequence and 5’ of the polyA sequence.
30. The recombinant nucleic acid molecule of any one of claims 25 to 29, wherein the UbC promoter comprises the nucleotides sequence of SEQ ID NO: 10.
31. The recombinant nucleic acid molecule of any one of claims 25 to 30, wherein each of the miR182 target sequences comprises the nucleotide sequence of SEQ ID NO: 12.
32. The recombinant nucleic acid molecule of any one of claims 25 to 31, wherein the polyA sequence is a rabbit beta-globin (rBG) polyA sequence.
33. The recombinant nucleic acid molecule of claim 32, wherein the rBG polyA sequence comprises the nucleotide sequence of SEQ ID NO: 13.
34. The recombinant nucleic acid molecule of any one of claims 25 to 33, wherein the expression cassette further comprises an intron.
35. The recombinant nucleic acid molecule of claim 34, wherein the intron comprises the nucleotide sequence of SEQ ID NO: 11.
36. The recombinant nucleic acid molecule of any one of claims 25 to 34, wherein the AAV 5’ ITR sequence and the AAV 3’ ITR sequence are from AAV2, optionally wherein the AAV 5’ ITR sequence comprises the nucleotide sequence of SEQ ID NO: 14 and/or the AAV 3’ ITR sequence comprises the nucleotide sequence of SEQ ID NO: 15.
37. The recombinant nucleic acid molecule of any one of claims 25 to 35, wherein the expression cassette comprises the nucleic acid sequence of SEQ ID NO: 16, or a nucleic acid sequence at least 99% identical to SEQ ID NO: 16.
38. The recombinant nucleic acid molecule of any one of claims 25 to 37, which comprises the nucleotide sequence of SEQ ID NO: 2 or a nucleotide sequence at least 99% identical to SEQ ID NO: 2.
39. A plasmid comprising the nucleic acid molecule of any one of claims 25 to 38.
40. A packaging host cell comprising the recombinant nucleic acid molecule of any one of claims 25 to 38 or the plasmid of claim 39.
41. The packaging host cell of claim 40, which further comprises AAV rep coding sequences operably linked to sequences that express rep protein in the packaging host cell, an
AAV capsid coding sequence operably linked to sequences the express AAV capsid proteins in the packaging host cell, and helper virus functions necessary to permit packaging of the expression cassette and the AAV 5’ ITR sequence and the AAV 3’ ITR sequence into the AAV capsid.
42. The packaging host cell of claim 40 or 41, wherein the AAV capsid is an AAVhu68 capsid.
43. The packaging host cell of claim 40 or 41, wherein the AAV capsid is an AAV9, AAVrh91, AAVhu95, or AAVhu96 capsid.
44. A recombinant AAV production system for producing the recombinant AAV of any one of claims 1 to 19, wherein the production system comprises a cell culture comprising the packaging host cell of any of claims 28 to 31.
45. The recombinant AAV production system of claim 44, wherein the AAV capsid is an AAVhu68 capsid.
46. The recombinant AAV production system of claim 44, wherein the AAV capsid is an AAV9, AAVrh91, AAVhu95, or AAVhu96 capsid.
47. The recombinant AAV production system of any one of claims 44 to 46, wherein the vector genome comprises the nucleotide sequence of SEQ ID NO: 16.
48. The recombinant AAV production system of any one of claims 44 to 47, wherein the vector genome comprises nucleic acid sequence of SEQ ID NO: 2.
49. A method for treating spinal muscular atrophy (SMA) in a subject in need thereof, the method comprising administering to the subject an aqueous suspension comprising the recombinant AAV of any one of claims 1 to 19.
50. The method of claim 49, wherein the subject has type I (Werdnig-Hoffmann disease), type II (Dubowitz disease), type III (Kugelberg-Welander disease), or type IV (adult) SMA.
51. The method of claim 49 or 50, wherein the suspension is administered intrathecally, intravenously, or both intrathecally and intravenously.
52. The method of claim 49 or 50, wherein the suspension is administered intrathecally and the dose of the recombinant AAV is about 1 x 1011 GC per gram of brain mass to about 6 x 1011 GC per gram of brain mass, optionally wherein the intrathecal administration is intracerebroventricular (ICV) injection or intraci sternal magna (ICM) injection.
53. The recombinant AAV of any one of claims 1 to 19 for use in preparing a medicament for treatment of SMA.
54. The recombinant AAV of any one of claims 1 to 19 for use in the treatment of SMA.
55. Use of the recombinant AAV of any one of claims 1 to 19, the composition of claim 20 or 21, the pharmaceutical composition of any one of claims 22 to 24, the recombinant nucleic acid molecule of any one of claims 25 to 38, the plasmid of claim 39, the packaging host cell of any one of claims 40 to 43, or the recombinant AAV production system of any one of claims 44 to 48 in the preparation of a medicament for treating SMA and/or a symptoms associated with functional SMN protein deficiency.
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US202363518796P | 2023-08-10 | 2023-08-10 | |
| US63/518,796 | 2023-08-10 | ||
| US202463662509P | 2024-06-21 | 2024-06-21 | |
| US63/662,509 | 2024-06-21 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2025035143A1 true WO2025035143A1 (en) | 2025-02-13 |
Family
ID=92543299
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2024/041840 Pending WO2025035143A1 (en) | 2023-08-10 | 2024-08-10 | Compositions and methods for treatment of spinal muscular atrophy |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2025035143A1 (en) |
Citations (56)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5139941A (en) | 1985-10-31 | 1992-08-18 | University Of Florida Research Foundation, Inc. | AAV transduction vectors |
| WO1996009378A1 (en) | 1994-09-19 | 1996-03-28 | The General Hospital Corporation | Overexpression of mammalian and viral proteins |
| US5741683A (en) | 1995-06-07 | 1998-04-21 | The Research Foundation Of State University Of New York | In vitro packaging of adeno-associated virus DNA |
| US6057152A (en) | 1992-12-04 | 2000-05-02 | University Of Pittsburgh | Recombinant viral vector system |
| US6166197A (en) | 1995-03-06 | 2000-12-26 | Isis Pharmaceuticals, Inc. | Oligomeric compounds having pyrimidine nucleotide (S) with 2'and 5 substitutions |
| US6204059B1 (en) | 1994-06-30 | 2001-03-20 | University Of Pittsburgh | AAV capsid vehicles for molecular transfer |
| US6210892B1 (en) | 1998-10-07 | 2001-04-03 | Isis Pharmaceuticals, Inc. | Alteration of cellular behavior by antisense modulation of mRNA processing |
| US6268213B1 (en) | 1992-06-03 | 2001-07-31 | Richard Jude Samulski | Adeno-associated virus vector and cis-acting regulatory and promoter elements capable of expressing at least one gene and method of using same for gene therapy |
| WO2001091800A1 (en) | 2000-05-30 | 2001-12-06 | Isis Innovation Limited | Ubiquitin promoter in vectors for gene therapy in respiratory tract |
| US6491907B1 (en) | 1998-11-10 | 2002-12-10 | The University Of North Carolina At Chapel Hill | Recombinant parvovirus vectors and method of making |
| EP1310571A2 (en) | 2001-11-13 | 2003-05-14 | The Trustees of The University of Pennsylvania | A Method of detecting and/or identifying adeno-associated virus (AVV) sequences and isolating novel sequences identified thereby |
| US6596535B1 (en) | 1999-08-09 | 2003-07-22 | Targeted Genetics Corporation | Metabolically activated recombinant viral vectors and methods for the preparation and use |
| US6660514B1 (en) | 1998-05-27 | 2003-12-09 | University Of Florida Research Foundation | Method of preparing recombinant adeno-associated virus compositions |
| WO2005033321A2 (en) | 2003-09-30 | 2005-04-14 | The Trustees Of The University Of Pennsylvania | Adeno-associated virus (aav) clades, sequences, vectors containing same, and uses therefor |
| US6951753B2 (en) | 1989-09-07 | 2005-10-04 | The 501 Trustees Of Princeton University | Helper-free stocks of recombinant adeno-associated virus vectors |
| US7094604B2 (en) | 2002-06-05 | 2006-08-22 | University Of Florida Research Foundation, Inc. | Production of pseudotyped recombinant AAV virions |
| US7101993B1 (en) | 1990-01-11 | 2006-09-05 | Isis Pharmaceuticals, Inc. | Oligonucleotides containing 2′-O-modified purines |
| WO2006110689A2 (en) | 2005-04-07 | 2006-10-19 | The Trustees Of The University Of Pennsylvania | Method of increasing the function of an aav vector |
| US7201898B2 (en) | 2000-06-01 | 2007-04-10 | The University Of North Carolina At Chapel Hill | Methods and compounds for controlled release of recombinant parvovirus vectors |
| US7229823B2 (en) | 1997-04-14 | 2007-06-12 | Richard Jude Samulski | Methods for increasing the efficiency of recombinant AAV product |
| US7282199B2 (en) | 2001-12-17 | 2007-10-16 | The Trustees Of The University Of Pennsylvania | Adeno-associated virus (AAV) serotype 8 sequences, vectors containing same, and uses therefor |
| US7439065B2 (en) | 1995-06-07 | 2008-10-21 | The University Of North Carolina At Chapel Hill | Helper virus-free AAV production |
| US7456683B2 (en) | 2005-06-09 | 2008-11-25 | Panasonic Corporation | Amplitude error compensating device and quadrature skew error compensating device |
| US7588772B2 (en) | 2006-03-30 | 2009-09-15 | Board Of Trustees Of The Leland Stamford Junior University | AAV capsid library and AAV capsid proteins |
| WO2010053572A2 (en) | 2008-11-07 | 2010-05-14 | Massachusetts Institute Of Technology | Aminoalcohol lipidoids and uses thereof |
| US7838657B2 (en) | 2004-12-03 | 2010-11-23 | University Of Massachusetts | Spinal muscular atrophy (SMA) treatment via targeting of SMN2 splice site inhibitory sequences |
| US7985565B2 (en) | 1997-04-01 | 2011-07-26 | Illumina, Inc. | Method of nucleic acid amplification |
| US8008005B2 (en) | 2001-05-18 | 2011-08-30 | Wisconsin Alumni Research Foundation | Method for the synthesis of DNA sequences |
| WO2011126808A2 (en) | 2010-03-29 | 2011-10-13 | The Trustees Of The University Of Pennsylvania | Pharmacologically induced transgene ablation system |
| WO2012170930A1 (en) | 2011-06-08 | 2012-12-13 | Shire Human Genetic Therapies, Inc | Lipid nanoparticle compositions and methods for mrna delivery |
| WO2013004943A1 (en) | 2011-07-06 | 2013-01-10 | Gaztransport Et Technigaz | Thermally-insulating sealed tank built into a load-bearing structure |
| US8361977B2 (en) | 2005-06-23 | 2013-01-29 | Isis Pharmaceuticals, Inc. | Compositions and methods for modulation of SMN2 splicing |
| WO2013182683A1 (en) | 2012-06-08 | 2013-12-12 | Ethris Gmbh | Pulmonary delivery of messenger rna |
| WO2014151341A1 (en) | 2013-03-15 | 2014-09-25 | The Trustees Of The University Of Pennsylvania | Compositions and methods for treating mpsi |
| US8980853B2 (en) | 2009-06-17 | 2015-03-17 | Isis Pharmaceuticals, Inc. | Compositions and methods for modulation of SMN2 splicing in a subject |
| US20150079038A1 (en) | 2013-09-13 | 2015-03-19 | California Institute Of Technology | Selective recovery |
| WO2017100674A1 (en) | 2015-12-11 | 2017-06-15 | The Trustees Of The University Of Pennsylvania | Scalable purification method for aav1 |
| WO2017100676A1 (en) | 2015-12-11 | 2017-06-15 | The Trustees Of The University Of Pennsylvania | Scalable purification method for aav8 |
| WO2017100704A1 (en) | 2015-12-11 | 2017-06-15 | The Trustees Of The University Of Pennsylvania | Scalable purification method for aavrh10 |
| WO2017136500A1 (en) | 2016-02-03 | 2017-08-10 | The Trustees Of The University Of Pennsylvania | Gene therapy for treating mucopolysaccharidosis type i |
| WO2017160360A2 (en) | 2015-12-11 | 2017-09-21 | The Trustees Of The University Of Pennsylvania | Scalable purification method for aav9 |
| WO2018160582A1 (en) | 2017-02-28 | 2018-09-07 | The Trustees Of The University Of Pennsylvania | Adeno-associated virus (aav) clade f vector and uses therefor |
| US20180339065A1 (en) | 2015-10-28 | 2018-11-29 | The Trustees Of The University Of Pennsylvania | Intrathecal administration of adeno-associated-viral vectors for gene therapy |
| US20180356394A1 (en) | 2015-12-02 | 2018-12-13 | Voyager Therapeutics, Inc. | Assays for the detection of aav neutralizing antibodies |
| WO2020132455A1 (en) | 2018-12-21 | 2020-06-25 | The Trustees Of The University Of Pennsylvania | Compositions for drg-specific reduction of transgene expression |
| WO2020223231A1 (en) | 2019-04-29 | 2020-11-05 | The Trustees Of The University Of Pennsylvania | Novel aav capsids and compositions containing same |
| US20210308281A1 (en) | 2018-08-15 | 2021-10-07 | Biogen Ma Inc. | Combination therapy for spinal muscular atrophy |
| WO2021231579A1 (en) | 2020-05-12 | 2021-11-18 | The Trustees Of The University Of Pennsylvania | Compositions for drg-specific reduction of transgene expression |
| WO2021257668A1 (en) | 2020-06-17 | 2021-12-23 | The Trustees Of The University Of Pennsylvania | Compositions and methods for treatment of gene therapy patients |
| WO2022094078A1 (en) * | 2020-10-28 | 2022-05-05 | The Trustees Of The University Of Pennsylvania | Compositions useful in treatment of rett syndrome |
| WO2022119871A2 (en) | 2020-12-01 | 2022-06-09 | The Trustees Of The University Of Pennsylvania | Novel compositions with tissue-specific targeting motifs and compositions containing same |
| US20220267798A1 (en) | 2019-08-08 | 2022-08-25 | Biogen Ma Inc. | Potency assays for viral vector production |
| US20220280548A1 (en) | 2019-08-15 | 2022-09-08 | Biogen Ma Inc. | Combination therapy for spinal muscular atrophy |
| WO2022226263A1 (en) | 2021-04-23 | 2022-10-27 | The Trustees Of The University Of Pennsylvania | Novel compositions with brain-specific targeting motifs and compositions containing same |
| WO2023056399A1 (en) * | 2021-10-02 | 2023-04-06 | The Trustees Of The University Of Pennsylvania | Novel aav capsids and compositions containing same |
| US20230107651A1 (en) | 2018-01-25 | 2023-04-06 | Biogen Ma Inc. | Methods of treating spinal muscular atrophy |
-
2024
- 2024-08-10 WO PCT/US2024/041840 patent/WO2025035143A1/en active Pending
Patent Citations (67)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5139941A (en) | 1985-10-31 | 1992-08-18 | University Of Florida Research Foundation, Inc. | AAV transduction vectors |
| US6951753B2 (en) | 1989-09-07 | 2005-10-04 | The 501 Trustees Of Princeton University | Helper-free stocks of recombinant adeno-associated virus vectors |
| US7101993B1 (en) | 1990-01-11 | 2006-09-05 | Isis Pharmaceuticals, Inc. | Oligonucleotides containing 2′-O-modified purines |
| US6268213B1 (en) | 1992-06-03 | 2001-07-31 | Richard Jude Samulski | Adeno-associated virus vector and cis-acting regulatory and promoter elements capable of expressing at least one gene and method of using same for gene therapy |
| US6057152A (en) | 1992-12-04 | 2000-05-02 | University Of Pittsburgh | Recombinant viral vector system |
| US6204059B1 (en) | 1994-06-30 | 2001-03-20 | University Of Pittsburgh | AAV capsid vehicles for molecular transfer |
| WO1996009378A1 (en) | 1994-09-19 | 1996-03-28 | The General Hospital Corporation | Overexpression of mammalian and viral proteins |
| US6166197A (en) | 1995-03-06 | 2000-12-26 | Isis Pharmaceuticals, Inc. | Oligomeric compounds having pyrimidine nucleotide (S) with 2'and 5 substitutions |
| US7439065B2 (en) | 1995-06-07 | 2008-10-21 | The University Of North Carolina At Chapel Hill | Helper virus-free AAV production |
| US5741683A (en) | 1995-06-07 | 1998-04-21 | The Research Foundation Of State University Of New York | In vitro packaging of adeno-associated virus DNA |
| US7985565B2 (en) | 1997-04-01 | 2011-07-26 | Illumina, Inc. | Method of nucleic acid amplification |
| US7229823B2 (en) | 1997-04-14 | 2007-06-12 | Richard Jude Samulski | Methods for increasing the efficiency of recombinant AAV product |
| US6660514B1 (en) | 1998-05-27 | 2003-12-09 | University Of Florida Research Foundation | Method of preparing recombinant adeno-associated virus compositions |
| US6210892B1 (en) | 1998-10-07 | 2001-04-03 | Isis Pharmaceuticals, Inc. | Alteration of cellular behavior by antisense modulation of mRNA processing |
| US6491907B1 (en) | 1998-11-10 | 2002-12-10 | The University Of North Carolina At Chapel Hill | Recombinant parvovirus vectors and method of making |
| US7172893B2 (en) | 1998-11-10 | 2007-02-06 | University Of North Carolina At Chapel Hill | Virus vectors and methods of making and administering the same |
| US6596535B1 (en) | 1999-08-09 | 2003-07-22 | Targeted Genetics Corporation | Metabolically activated recombinant viral vectors and methods for the preparation and use |
| US7125717B2 (en) | 1999-08-09 | 2006-10-24 | Targeted Genetics Corporation | Metabolically activated recombinant viral vectors and methods for their preparation and use |
| WO2001091800A1 (en) | 2000-05-30 | 2001-12-06 | Isis Innovation Limited | Ubiquitin promoter in vectors for gene therapy in respiratory tract |
| US7201898B2 (en) | 2000-06-01 | 2007-04-10 | The University Of North Carolina At Chapel Hill | Methods and compounds for controlled release of recombinant parvovirus vectors |
| US8008005B2 (en) | 2001-05-18 | 2011-08-30 | Wisconsin Alumni Research Foundation | Method for the synthesis of DNA sequences |
| US20130045186A1 (en) | 2001-11-13 | 2013-02-21 | The Trustees Of The University Of Pennsylvania | Method of Detecting and/or Identifying Adeno-Associated Virus (AAV) Sequences and Isolating Novel Sequences Identified Thereby |
| WO2003042397A2 (en) | 2001-11-13 | 2003-05-22 | The Trustees Of The University Of Pennsylvania | A method of detecting and/or identifying adeno-associated virus (aav) sequences and isolating novel sequences identified thereby |
| EP1310571A2 (en) | 2001-11-13 | 2003-05-14 | The Trustees of The University of Pennsylvania | A Method of detecting and/or identifying adeno-associated virus (AVV) sequences and isolating novel sequences identified thereby |
| US7790449B2 (en) | 2001-12-17 | 2010-09-07 | The Trustees Of The University Of Pennsylvania | Adeno-associated virus (AAV) serotype 8 sequences, vectors containing the same, and uses therefor |
| US7282199B2 (en) | 2001-12-17 | 2007-10-16 | The Trustees Of The University Of Pennsylvania | Adeno-associated virus (AAV) serotype 8 sequences, vectors containing same, and uses therefor |
| US7094604B2 (en) | 2002-06-05 | 2006-08-22 | University Of Florida Research Foundation, Inc. | Production of pseudotyped recombinant AAV virions |
| US7906111B2 (en) | 2003-09-30 | 2011-03-15 | The Trustees Of The University Of Pennsylvania | Adeno-associated virus (AAV) clades, sequences, vectors containing same, and uses therefor |
| US20070036760A1 (en) | 2003-09-30 | 2007-02-15 | The Trutees Of The University Of Pennsylvania | Adeno-associated virus (aav) clades, sequences, vectors containing same, and uses therefor |
| WO2005033321A2 (en) | 2003-09-30 | 2005-04-14 | The Trustees Of The University Of Pennsylvania | Adeno-associated virus (aav) clades, sequences, vectors containing same, and uses therefor |
| US8110560B2 (en) | 2004-12-03 | 2012-02-07 | University Of Massachusetts | Spinal muscular atrophy (SMA) treatment via targeting of SMN2 splice site inhibitory sequences |
| US7838657B2 (en) | 2004-12-03 | 2010-11-23 | University Of Massachusetts | Spinal muscular atrophy (SMA) treatment via targeting of SMN2 splice site inhibitory sequences |
| US20090197338A1 (en) | 2005-04-07 | 2009-08-06 | The Trustees Of Teh University Of Pennsylvania | Method of Increasing the Function of an AAV Vector |
| WO2006110689A2 (en) | 2005-04-07 | 2006-10-19 | The Trustees Of The University Of Pennsylvania | Method of increasing the function of an aav vector |
| US7456683B2 (en) | 2005-06-09 | 2008-11-25 | Panasonic Corporation | Amplitude error compensating device and quadrature skew error compensating device |
| US8361977B2 (en) | 2005-06-23 | 2013-01-29 | Isis Pharmaceuticals, Inc. | Compositions and methods for modulation of SMN2 splicing |
| US7588772B2 (en) | 2006-03-30 | 2009-09-15 | Board Of Trustees Of The Leland Stamford Junior University | AAV capsid library and AAV capsid proteins |
| WO2010053572A2 (en) | 2008-11-07 | 2010-05-14 | Massachusetts Institute Of Technology | Aminoalcohol lipidoids and uses thereof |
| US8980853B2 (en) | 2009-06-17 | 2015-03-17 | Isis Pharmaceuticals, Inc. | Compositions and methods for modulation of SMN2 splicing in a subject |
| WO2011126808A2 (en) | 2010-03-29 | 2011-10-13 | The Trustees Of The University Of Pennsylvania | Pharmacologically induced transgene ablation system |
| WO2012170930A1 (en) | 2011-06-08 | 2012-12-13 | Shire Human Genetic Therapies, Inc | Lipid nanoparticle compositions and methods for mrna delivery |
| WO2013004943A1 (en) | 2011-07-06 | 2013-01-10 | Gaztransport Et Technigaz | Thermally-insulating sealed tank built into a load-bearing structure |
| WO2013182683A1 (en) | 2012-06-08 | 2013-12-12 | Ethris Gmbh | Pulmonary delivery of messenger rna |
| WO2014151341A1 (en) | 2013-03-15 | 2014-09-25 | The Trustees Of The University Of Pennsylvania | Compositions and methods for treating mpsi |
| US20150079038A1 (en) | 2013-09-13 | 2015-03-19 | California Institute Of Technology | Selective recovery |
| US20180339065A1 (en) | 2015-10-28 | 2018-11-29 | The Trustees Of The University Of Pennsylvania | Intrathecal administration of adeno-associated-viral vectors for gene therapy |
| US20180356394A1 (en) | 2015-12-02 | 2018-12-13 | Voyager Therapeutics, Inc. | Assays for the detection of aav neutralizing antibodies |
| WO2017100676A1 (en) | 2015-12-11 | 2017-06-15 | The Trustees Of The University Of Pennsylvania | Scalable purification method for aav8 |
| WO2017160360A2 (en) | 2015-12-11 | 2017-09-21 | The Trustees Of The University Of Pennsylvania | Scalable purification method for aav9 |
| WO2017100704A1 (en) | 2015-12-11 | 2017-06-15 | The Trustees Of The University Of Pennsylvania | Scalable purification method for aavrh10 |
| WO2017100674A1 (en) | 2015-12-11 | 2017-06-15 | The Trustees Of The University Of Pennsylvania | Scalable purification method for aav1 |
| WO2017136500A1 (en) | 2016-02-03 | 2017-08-10 | The Trustees Of The University Of Pennsylvania | Gene therapy for treating mucopolysaccharidosis type i |
| WO2018160582A1 (en) | 2017-02-28 | 2018-09-07 | The Trustees Of The University Of Pennsylvania | Adeno-associated virus (aav) clade f vector and uses therefor |
| US20200056159A1 (en) | 2017-02-28 | 2020-02-20 | The Trustees Of The University Of Pennsylvania | Novel adeno-associated virus (aav) clade f vector and uses therefor |
| US20230107651A1 (en) | 2018-01-25 | 2023-04-06 | Biogen Ma Inc. | Methods of treating spinal muscular atrophy |
| US20210308281A1 (en) | 2018-08-15 | 2021-10-07 | Biogen Ma Inc. | Combination therapy for spinal muscular atrophy |
| WO2020132455A1 (en) | 2018-12-21 | 2020-06-25 | The Trustees Of The University Of Pennsylvania | Compositions for drg-specific reduction of transgene expression |
| WO2020223231A1 (en) | 2019-04-29 | 2020-11-05 | The Trustees Of The University Of Pennsylvania | Novel aav capsids and compositions containing same |
| US20220267798A1 (en) | 2019-08-08 | 2022-08-25 | Biogen Ma Inc. | Potency assays for viral vector production |
| US20220280548A1 (en) | 2019-08-15 | 2022-09-08 | Biogen Ma Inc. | Combination therapy for spinal muscular atrophy |
| WO2021231579A1 (en) | 2020-05-12 | 2021-11-18 | The Trustees Of The University Of Pennsylvania | Compositions for drg-specific reduction of transgene expression |
| WO2021257668A1 (en) | 2020-06-17 | 2021-12-23 | The Trustees Of The University Of Pennsylvania | Compositions and methods for treatment of gene therapy patients |
| WO2022094078A1 (en) * | 2020-10-28 | 2022-05-05 | The Trustees Of The University Of Pennsylvania | Compositions useful in treatment of rett syndrome |
| WO2022119871A2 (en) | 2020-12-01 | 2022-06-09 | The Trustees Of The University Of Pennsylvania | Novel compositions with tissue-specific targeting motifs and compositions containing same |
| US20240024507A1 (en) | 2020-12-01 | 2024-01-25 | The Trustees Of The University Of Pennsylvania | Novel compositions with tissue-specific targeting motifs and compositions containing same |
| WO2022226263A1 (en) | 2021-04-23 | 2022-10-27 | The Trustees Of The University Of Pennsylvania | Novel compositions with brain-specific targeting motifs and compositions containing same |
| WO2023056399A1 (en) * | 2021-10-02 | 2023-04-06 | The Trustees Of The University Of Pennsylvania | Novel aav capsids and compositions containing same |
Non-Patent Citations (40)
| Title |
|---|
| "GenBank Accession", Database accession no. AY530629 |
| "GenBank", Database accession no. AF232305 |
| "Large-Scale Production of Adeno-Associated Viral Vector Serotype-9 Carrying the Human Survival Motor Neuron Gene", MOL BIOTECHNOL., vol. 58, no. 1, January 2016 (2016-01-01), pages 30 - 6 |
| "NCBI", Database accession no. NM 022874/NP 075012 |
| ALVES CRR ET AL.: "Implications of circulating neurofilaments for spinal muscular atrophy treatment early in life: A case series", MOL THER METHODS CLIN DEV., vol. 23, 30 October 2021 (2021-10-30), pages 524 - 538 |
| B. J. CARTER: "Handbook of Parvoviruses", vol. 168, 1990, CRC PRESS, pages: 155 |
| BUNING ET AL.: "Recent developments in adeno-associated virus vector technology", J. GENE MED., vol. 10, 2008, pages 717 - 733 |
| CALCEDO, R. ET AL.: "Worldwide Epidemiology of Neutralizing Antibodies to Adeno-Associated Viruses", JOURNAL OF INFECTIOUS DISEASES, vol. 199, no. 3, 2009, pages 381 - 390 |
| CARTEGNIKRAINER, NAT. GENET., vol. 30, 2002, pages 377 - 384 |
| D M MCCARTY ET AL.: "Self-complementary recombinant adeno-associated virus (scAAV) vectors promote efficient transduction independently of DNA synthesis", GENE THERAPY, vol. 8, no. 16, August 2001 (2001-08-01), pages 1248 - 1254, XP037773369, DOI: 10.1038/sj.gt.3301514 |
| G GAO ET AL., J VIROL, vol. 78, no. 10, June 2004 (2004-06-01), pages 6381 - 6388 |
| GAO ET AL., PROC. NATL. ACAD. SCI. U.S.A., vol. 100, no. 10, 2003, pages 6081 - 6086 |
| GENE SEQ., vol. 6, no. 1, April 2012 (2012-04-01), pages 10 - 21 |
| GORDEEVA ET AL.: "Improved PCR-based gene synthesis method and its application to the Citrobacter freundii phytase gene codon modification", J MICROBIOL METHODS, vol. 81, no. 2, 10 March 2010 (2010-03-10), pages 147 - 52, XP027012609 |
| GREENSAMBROOK: "Molecular Cloning: A Laboratory Manual", 2012, COLD SPRING HARBOR PRESS |
| GRIEGERSAMULSKI: "Adeno-associated virus as a gene therapy vector: Vector development, production and clinical applications", ADV. BIOCHEM. ENGIN/BIOTECHNOL., vol. 99, 2005, pages 119 - 145, XP009125595 |
| GRIMM ET AL., GENE THERAPY, vol. 6, 1999, pages 1322 - 1330 |
| GUO ET AL.: "Rapid AAV Neutralizing Antibody Determination with a Cell-Binding Assay", MOLECULAR THERAPY: METHODS & CLINICAL DEVELOPMENT, 13 June 2019 (2019-06-13) |
| J. D. THOMPSON ET AL.: "A comprehensive comparison of multiple sequence alignments", NUCL. ACIDS. RES., vol. 27, no. 13, 1999, pages 2682 - 2690, XP002191669, DOI: 10.1093/nar/27.13.2682 |
| K. FISHER ET AL., J. VIROL., vol. 70, no. 520, 1996, pages 532 |
| KIM DW ET AL.: "Use of the human elongation factor 1 alpha promoter as a versatile and efficient expression system.", GENE, vol. 91, no. 2, 16 July 1990 (1990-07-16), pages 217 - 23 |
| KIM J ET AL.: "Involvement of cholesterol-rich lipid rafts in interleukin-6-induced neuroendocrine differentiation of LNCaP prostate cancer cells", ENDOCRINOLOGY, vol. 145, no. 2, 16 October 2003 (2003-10-16), pages 613 - 9 |
| KÜGLER S ET AL.: "Human synapsin 1 gene promoter confers highly neuron-specific long-term transgene expression from an adenoviral vector in the adult rat brain depending on the transduced area", GENE THER., vol. 10, no. 4, February 2003 (2003-02-01), pages 337 - 47, XP037772810, DOI: 10.1038/sj.gt.3301905 |
| LEFEBVRE ET AL., HUM. MOL. GENET., vol. 7, no. 15, 1998, pages 31 - 1536 |
| LOCK, M. ET AL., HUM GENE THER, vol. 21, 2010, pages 1259 - 1271 |
| M. LOCK ET AL., HU GENE THERAPY METHODS, HUM GENE THER METHODS., vol. 25, no. 2, 14 February 2014 (2014-02-14), pages 115 - 25 |
| M. NEIS. KUMAR: "Molecular Evolution and Phylogenetics", 2000, OXFORD UNIVERSITY PRESS |
| MIZUKAMIHIROAKI ET AL.: "A Protocol for AAV vector production and purification", DISS. DIVISION OF GENETIC THERAPEUTICS, CENTER FOR MOLECULAR MEDICINE, 1998 |
| NOVEL ADENO-ASSOCIATED VIRUS (AAV) VECTORS, AAV VECTORS HAVING REDUCED CAPSID DEAMIDATION AND USES THEREFOR, 27 February 2019 (2019-02-27) |
| NUCLEIC ACIDS RES., vol. 32, no. 7, 2004, pages 59 |
| R CALCEDO ET AL., JOURNAL INFECTIOUS DISEASES, vol. 199, 2009, pages 381 - 290 |
| SAMBROOK ET AL.: "Molecular Cloning. A Laboratory Manual", 1989, COLD SPRING HARBOR LABORATORY |
| SCHORPP ET AL., NUCLEIC ACIDS RES., vol. 24, no. 9, 1 May 1996 (1996-05-01), pages 1787 - 8 |
| SOMMER ET AL., MOLEC. THER., vol. 7, 2003, pages 122 - 128 |
| T. ITO ET AL.: "A convenient enzyme-linked immunosorbent assay for rapid screening of anti-adeno-associated virus neutralizing antibodies", ANN CLIN BIOCHEM, vol. 46, 2009, pages 508 - 510, XP055328294, DOI: 10.1258/acb.2009.009077 |
| TORNØE J ET AL., GENE, vol. 297, no. 1-2, 4 September 2002 (2002-09-04), pages 21 - 32 |
| WOBUS ET AL., J. VIROL., vol. 74, 2000, pages 9281 - 9293 |
| X. SU ET AL., MOL. PHARMACEUTICS, vol. 8, no. 3, 21 March 2011 (2011-03-21), pages 774 - 787 |
| XIONG ET AL.: "PCR-based accurate synthesis of long DNA sequences", NATURE PROTOCOLS, vol. 1, 2006, pages 791 - 797, XP001539584, DOI: 10.1038/nprot.2006.103 |
| ZHANG ET AL.: "Adenovirus-adeno-associated virus hybrid for large-scale recombinant adeno-associated virus production", HUMAN GENE THERAPY, vol. 20, 2009, pages 922 - 929, XP055873883 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP3717652A1 (en) | Gene therapy for mucopolysaccharidosis iiib | |
| JP7534290B2 (en) | Compositions useful for treating gm1 gangliosidosis | |
| EP3717653A1 (en) | Gene therapy for mucopolysaccharidosis iiia | |
| US20230167455A1 (en) | Compositions useful in treatment of cdkl5 deficiency disorder (cdd) | |
| US20220370638A1 (en) | Compositions and methods for treatment of maple syrup urine disease | |
| AU2021273273A1 (en) | Compositions useful for treatment of pompe disease | |
| EP3931337A1 (en) | Compositions useful in treatment of krabbe disease | |
| US20230414785A1 (en) | Compositions and uses thereof for treatment of angelman syndrome | |
| WO2023077143A1 (en) | Compositions useful in treatment of cdkl5 deficiency disorder (cdd) | |
| WO2025035143A1 (en) | Compositions and methods for treatment of spinal muscular atrophy | |
| AU2024320694A1 (en) | Compositions and methods for treatment of spinal muscular atrophy | |
| US20240115733A1 (en) | Compositions and methods for treatment of niemann pick type a disease | |
| US20250041445A1 (en) | Gene therapy for treatment of mucopolysaccharidosis iiia | |
| WO2024258961A1 (en) | Aav gene therapy for mucopolysaccharidosis iiib | |
| WO2025129157A1 (en) | Gene therapy for treatment of canavan disease |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 24762191 Country of ref document: EP Kind code of ref document: A1 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 829658 Country of ref document: NZ Ref document number: AU2024320694 Country of ref document: AU |
|
| WWP | Wipo information: published in national office |
Ref document number: 829658 Country of ref document: NZ |