WO2025035020A2 - Methods of using carfilzomib - Google Patents
Methods of using carfilzomib Download PDFInfo
- Publication number
- WO2025035020A2 WO2025035020A2 PCT/US2024/041550 US2024041550W WO2025035020A2 WO 2025035020 A2 WO2025035020 A2 WO 2025035020A2 US 2024041550 W US2024041550 W US 2024041550W WO 2025035020 A2 WO2025035020 A2 WO 2025035020A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- carfilzomib
- administered
- patient
- independently
- administering
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K38/00—Medicinal preparations containing peptides
- A61K38/04—Peptides having up to 20 amino acids in a fully defined sequence; Derivatives thereof
- A61K38/07—Tetrapeptides
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
Definitions
- Carfilzomib is a proteasomal inhibitor that is currently marketed as KYPROLIS® and is approved in many countries for the treatment of relapsed and/or refractory multiple myeloma (RRMM).
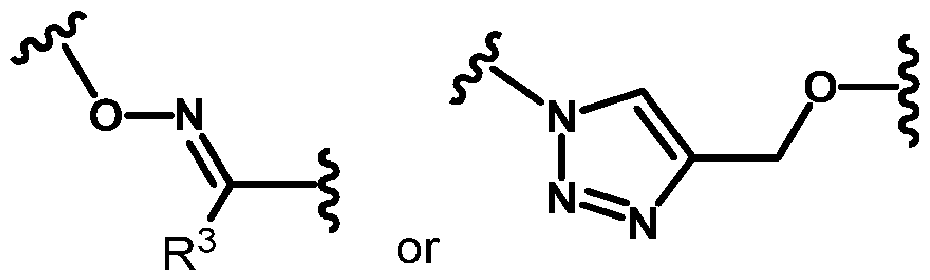
- RRMM refractory multiple myeloma
- carfilzomib is approved for use in combination with dexamethasone (Kd), with lenalidomide and dexamethasone (KRd), with daratumumab and dexamethasone (KdD), as well as with isatuximab and dexamethasone (Isa-Kd).
- Kd dexamethasone
- KRd lenalidomide and dexamethasone
- KdD daratumumab and dexamethasone
- Isa-Kd isatuximab and dexamethasone
- the current route of administration for KYPROLIS® is IV (intravenous) and dosing is based on the body surface area of the patient.
- the final concentration of reconstituted carfilzomib solution prior to addition into intravenous bags is 2 mg/mL.
- a twice weekly (twice-a-week) intravenous administration may be inconvenient for the patient and present challenges when contemplating administering carfilzomib in combination with other therapies for multiple myeloma.
- once weekly (once-a-week) carfilzomib dosing regimens and methods to lower the toxicity associated with carfilzomib would be useful.
- kits for treating multiple myeloma in a patient comprising administering to the patient a pharmaceutical composition comprising carfilzomib or a pharmaceutically acceptable salt thereof and an aqueous carrier, wherein the carfilzomib is administered at a dose volume of 2.5 mL/kg or higher.
- a disease or condition selected from the group consisting of cancer, autoimmune disease, graft or transplant-related condition, neurodegenerative disease, fibrotic-associated condition, ischemic-related conditions, infection (viral, parasitic or prokaryotic) and diseases associated with bone loss
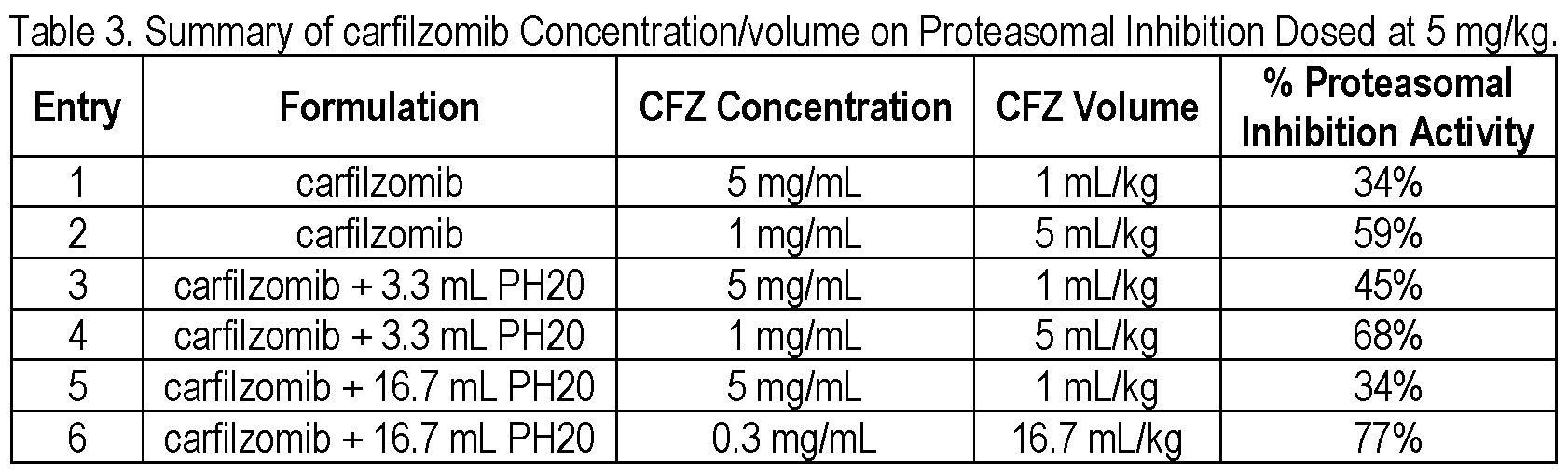
- Figure 1 shows the proteasomal inhibition activity displayed by various carfilzomib formulations when administered in mice.
- Figure 2 shows the effect of carfilzomib when administered subcutaneously dosed at 5 mg/kg at a dose.
- the red line is 5 mg/mL concentration (1 mL/kg) and the black line is 1 mg/mL concentration (5 mL/kg).
- Figure 3A shows the effect of two different concentrations of carfilzomib on proteasomal inhibition over the entire time course of the study.
- Figure 3B shows the effect of two different concentrations of carfilzomib on proteasomal inhibition over the first four hours.
- Figure 3C shows a comparison including data from subcutaneous administration studies.
- Figure 4A shows the effect of two different concentrations of pegylated carfilzomib (PEG-CFZ) on proteasomal inhibition over the entire time course of the study.
- Figure 4B shows the effect of two different concentrations of pegylated carfilzomib on proteasomal inhibition over the first six hours.
- Figure 5 shows the effect of two different concentrations and two different dose levels of pegylated carfilzomib (PEG-CFZ) on proteasomal inhibition over the entire time course of the study.
- PEG-CFZ pegylated carfilzomib
- Figure 6 shows the effect of two different concentrations of carfilzomib (CFZ) on proteasomal inhibition over the entire time course of the study.
- the present disclosure provides methods of treating a disease or condition comprising administering carfilzomib or a pharmaceutically acceptable salt thereof at a particular dose volume.
- Carfilzomib is a tetrapeptide epoxy ketone proteasome inhibitor that binds selectively and irreversibly to the constitutive proteasome and immunoproteosome. It has been approved to treat multiple myeloma and is marketed under the name KYPROLIS®. Carfilzomib, polymorphic forms, methods of making, formulations, its use and the like are described in, for example, U.S. Patent Application Publication Nos. 2005/0245435, 2014/0105921 and PCT Patent Publication Nos.
- Carfilzomib has the following chemical structure:
- the methods disclosed herein comprise administering carfilzomib or a pharmaceutically acceptable salt thereof.
- pharmaceutically acceptable salt embraces salts commonly used to form alkali metal salts and to form addition salts of free acids or free bases. The nature of the salt is not critical, provided that it is pharmaceutically -acceptable. Suitable pharmaceutically -acceptable acid addition salts of the compound may be prepared from an inorganic acid or from an organic acid. Examples of such inorganic acids include, without limitation, hydrochloric, hydrobromic, hydroiodic, nitric, carbonic, sulfuric and phosphoric acid.
- organic acids include, without limitation, aliphatic, cycloaliphatic, aromatic, arylaliphatic, heterocyclic, carboxylic and sulfonic classes of organic acids, examples of which are formic, acetic, adipic, butyric, propionic, succinic, glycolic, gluconic, lactic, malic, tartaric, citric, ascorbic, glucuronic, maleic, fumaric, pyruvic, aspartic, glutamic, benzoic, anthranilic, mesylic, 4-hydroxybenzoic, phenylacetic, mandelic, embonic (pamoic), methane sulfonic, ethanesulfonic, ethanedisulfonic, benzenesulfonic, pantothenic, 2- hydroxyethanesulfonic, toluenesulfonic, sulfanilic, cyclohexylaminosulfonic, camphoric, camphors,
- Suitable pharmaceutically acceptable base addition salts of carfilzomib include, without limitation, metallic salts such as salts made from aluminum, calcium, lithium, magnesium, potassium, sodium and zinc, or salts made from organic bases including primary, secondary, tertiary amines and substituted amines including cyclic amines such as caffeine, arginine, diethylamine, N-ethyl piperidine, histidine, glucamine, isopropylamine, lysine, morpholine, N-ethyl morpholine, piperazine, piperidine, triethylamine, trimethylamine. All of the salts contemplated herein may be prepared by conventional means from the corresponding compound by reacting, for example, the appropriate acid or base with the compound.
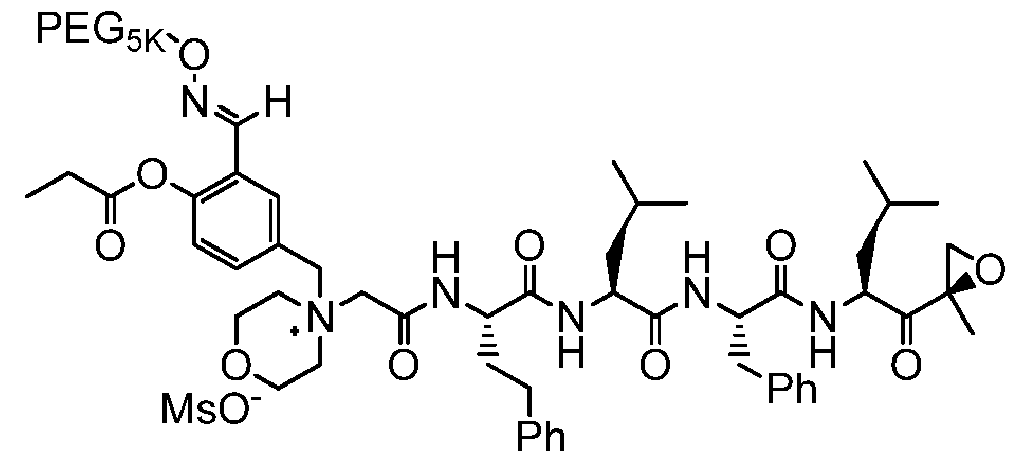
- the carfilzomib is pegylated.
- Pegylated carfilizomib is disclosed, for example, in PCT Publication No. WO 2017/205392A1 , the disclosure of which is incorporated herein by reference.
- the term "carfilzomib” also includes pegylated carfilzomib as described herein.
- the pegylated carfilzomib has a structure of Formula I:
- each R 1 is independently Ci- alkyl or Csjcycloalkyl
- each R 2 is independently Ci-ealkyl, -OCH3 or halogen
- each 0 is an integer selected from 0, 1, 2, and 3
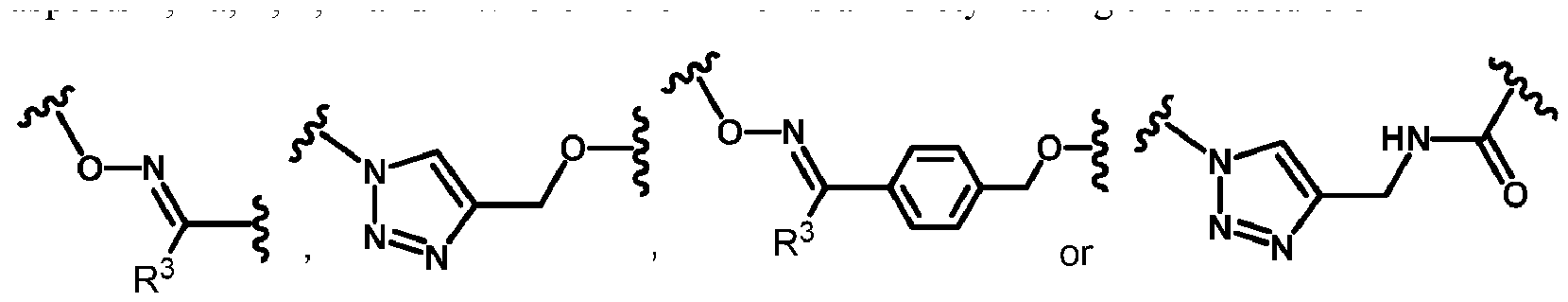
- each linker is independently a moiety independently having a structure of
- each R 3 is independently H or CH3; each n is an integer selected from 1, 2, 3, and 4; each p is independently an integer selected from 0, 1, 2, 3, and 4; each q is independently an integer selected from 1, 2, 3, 4, 5, 6, 7, 8, and 9; each r is independently an integer selected from 0, 1, 2, 3, 4, and 5; and PEG is a polyethylene glycol polymeric moiety having a molecular weight of about 500 to about 20,000 g/mol.
- each R 1 is independently Ci- alkyl or Csjcycloalkyl; each R 2 is independently Ci-ealkyl, -OCH3 or halogen; each 0 is independently an integer selected from 0, 1, 2, and 3; each linker is independently a moiety having the structure of:
- each each R 3 is independently H or CH 3 ; and each p is independently an integer selected from 0, 1, 2, 3, and 4; each n is independently an integer selected from 1, 2, 3, and 4; and PEG is a polyethylene glycol polymeric moiety having a molecular weight of about 500 to about 20,000 g/mol.
- the pegylated carfilzomib compound has a structure of Formula II , wherein
- each R 1 is independently Ci- alkyl or Csjcycloalkyl; each R 2 is independently Ci-ealkyl, -OCH3 or halogen; each linker is independently a moiety having the structure of
- the disclosure provides a compound of formula I, la, and II, wherein each R 1 is independently Ci- alkyl.
- each R 2 independently is H, CH3 or halogen (e.g., F, Br, and Cl).
- each linker is independently a moiety having the structure of wherein each R 3 is independently H or CH3; each q is independently an integer selected from 1 , 2, 3, 4, and 5; and each r is independently an integer selected from 0, 1 , 2, 3, and 4.
- each linker is independently a moiety having the structure of wherein each R 3 is independently H or CH3.
- each linker is independently a moiety having the structure of wherein R 3 is independently H or CH3; each q is independently 4 ; and each r is independently 2.
- each linker is independently a moiety having the structure of
- each R 3 is H.
- each R 1 is independently methyl, ethyl, propyl, isopropyl, butyl, t-butyl, pently, hexyl or heptyl.
- each R 1 is independently methyl, ethyl, propyl, isopropyl, butyl, t-butyl, pentyl, hexyl or heptyl; and the linker is
- each R 1 is methyl, ethyl, propyl, isopropyl, butyl, t-butyl, pentyl, hexyl or heptyl; and the linker is
- the disclosure provides a pegylated carfilzomib compound having the structure of
- the disclosure provides a compound of formula: wherein X is a halide, a sulfonate or an alkyl-sulfonate counterion salt.
- the disclosure provides a compound of formula:
- X is a halide, a sulfonate or an alky l-sulfonate counterion salt.
- the disclosure provides a compound of formula: wherein X is a halide, a sulfonate or an alky l-sulfonate counterion salt.
- the disclosure provides a compound of formula:
- each R 1 is independently methyl, ethyl, propyl, isopropyl, butyl, t-butyl, pently, hexyl or heptyl; each R 2 , independently, is CH3 or halogen; linker is a moiety having the structure of wherein each R 3 is independently H or CH3; and PEG is a polyethylene glycol polymeric moiety having a molecular weight of 2000, 3000, 5000 or 20,000 g/mol.
- each R is independently methyl, ethyl, propyl, isopropyl, butyl, t-butyl, pently, hexyl or heptyl; each R 2 , independently, is CH3; linker is a moiety having the structure of wherein each R 3 is independently H; and PEG is a polyethylene glycol polymeric moiety having a molecular weight of 3000, 5000 or 20,000 g/mol.
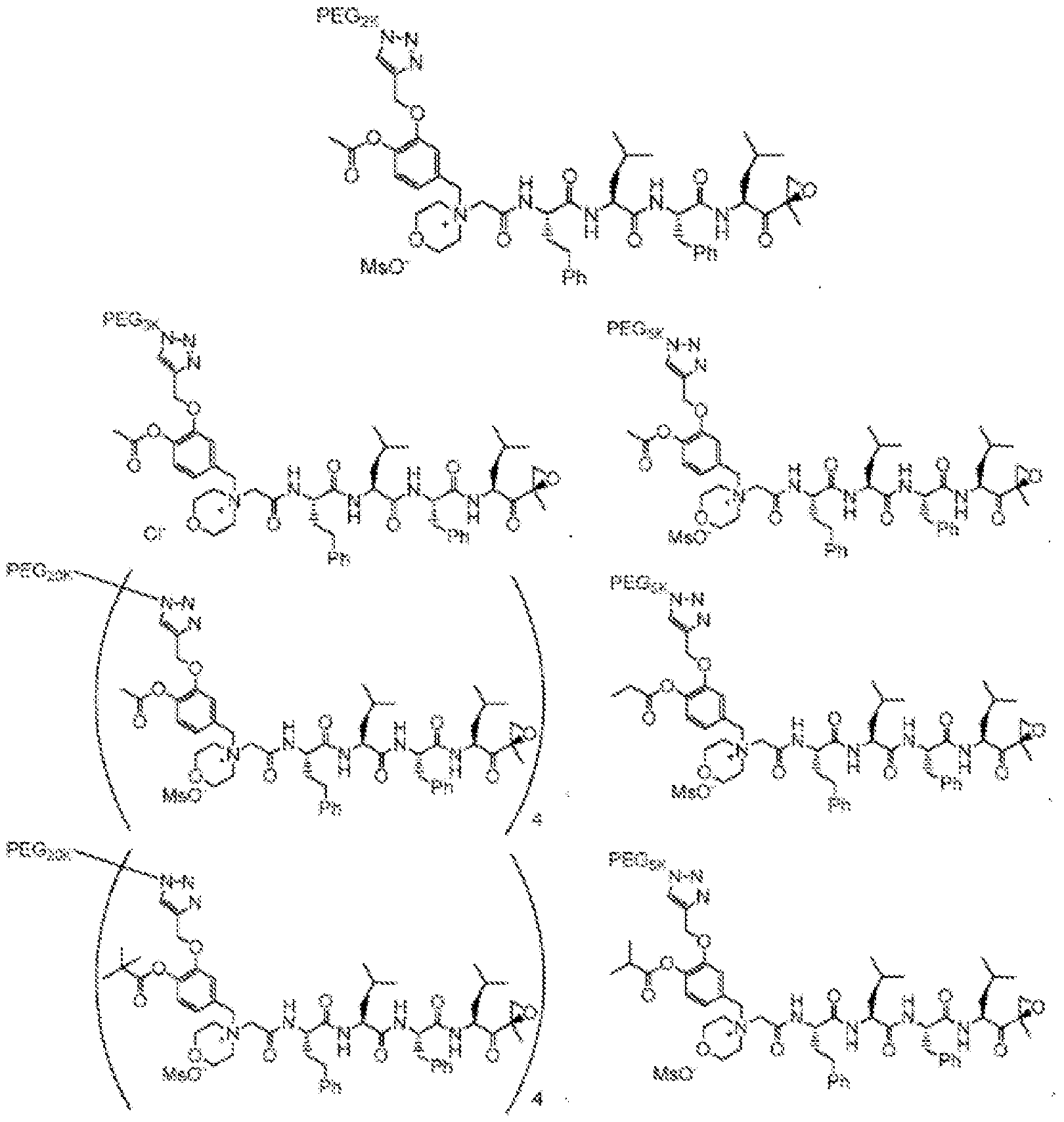
- the disclosure provides a compound selected from the following:
- the pegylated carfilizomib compound is N-(2-aminoethyl)-2-aminoethylated carfilizomib compound.
- the pegylated carfilizomib compound is N-(2-aminoethyl)-2-aminoethylated carfilizomib compound.
- the pegylated carfilizomib compound is N-(2-aminoethyl)-2-aminoethylated carfilizomib compound.
- the pegylated carfilizomib compound is N-(2-aminoethyl)-2-aminoethylated carfilizomib compound.
- the pegylated carfilizomib compound is N-(2-aminoethyl)-2-aminoethylated carfilizomib compound.
- the pegylated carfilizomib compound is N-(2-aminoethyl)-2-aminoethylated carfilizomib compound.
- the pegylated carfilizomib compound is N-(2-aminoethyl)-2-aminoethylated carfilizomib compound.
- the pegylated carfilizomib compound is N-(2-aminoethyl)-2-aminoethylated carfilizomib compound.
- the pegylated carfilizomib compound is N-(2-aminoethyl)-2-aminoethylated carfilizomib compound.
- the PEG has a weight ranging from about 2K to about 20K g/mol. In some embodiments, the PEG has a weight of 3K, 5K or 20K g/mol.
- the pharmaceutically acceptable salt of the pegylated carfilzomib is selected from a chloride anion, a bisulfate anion, a sulfate anion, a nitrate anion, a phosphate anion, an alky -sulfonate anion or an aryl-sulfonate anion.
- the pharmaceutically acceptable salt of the pegylated carfilzomib is a chloride anion or an alky -sulfonate anion.
- the pharmaceutically acceptable salt of the pegylated carfilzomib is a chloride anion or methane-sulfonate anion.
- the present disclosure provides methods for treating a disease or condition comprising administering to a patient carfilzomib or a pharmaceutically acceptable salt thereof.
- the terms “treat”, “treating”, and “treatment” as used herein refer to therapy, including without limitation, curative therapy, prophylactic therapy, and preventative therapy and generally include reversing, reducing, or arresting the symptoms, clinical signs, and underlying pathology of a condition in manner to improve or stabilize a patient's condition.
- Prophylactic treatment generally constitutes either preventing the onset of disorders altogether or delaying the onset of a pre-cl inically evident stage of disorders in individuals.
- the term "prophylactic or therapeutic" treatment is art- recognized and includes administration to the host of one or more of the subject compositions. If it is administered prior to clinical manifestation of the unwanted condition (e.g., disease or other unwanted state of the host animal), or after the condition has subsided, then the treatment is prophylactic (i.e., it protects the host against developing the unwanted condition), whereas if it is administered after manifestation of the unwanted condition, the treatment is therapeutic, (i.e., it is intended to diminish, ameliorate, or stabilize the existing unwanted condition or side effects thereof).
- the term "patient” as used herein refers to any mammal, including humans, and animals such as cows, horses, dogs and cats.
- the invention may be used in human patients as well as in veterinarian subjects and patients.
- carfilzomib or a pharmaceutically acceptable salt thereof is administered to a human patient.
- the phrase "therapeutical ly-effective" or “therapeutically effective amount” is intended to quantify the amount of a compound (e.g., carfilzomib or a pharmaceutically acceptable salt thereof), which when administered as part of a desired dosage regimen (to a patient, e.g., a human) alleviates a symptom, ameliorates a condition, or slows the onset of disease conditions according to clinically acceptable standards for the disorder or condition to be treated or the cosmetic purpose, for example, at a reasonable benefit/risk ratio applicable to any medical treatment.
- a compound e.g., carfilzomib or a pharmaceutically acceptable salt thereof
- it is the amount of a compound (e.g., carfilzomib or a pharmaceutically acceptable salt thereof) that can treat cancer, whether it is multiple myeloma or other hematologic malignancy or a solid tumor.
- the disclosure provides a method for treating a disease or condition selected from the group consisting of cancer, autoimmune disease, graft or transplant-related condition, neurodegenerative disease, fibrotic-associated condition, ischemic-related conditions, infection (viral, parasitic or prokaryotic) and diseases associated with bone loss, the method comprising administering to a patient a therapeutically effective amount of carfilzomib or a pharmaceutically acceptable salt thereof at a dose volume of 10 mL/kg or higher.
- a disease or condition selected from the group consisting of cancer, autoimmune disease, graft or transplant-related condition, neurodegenerative disease, fibrotic-associated condition, ischemic-related conditions, infection (viral, parasitic or prokaryotic) and diseases associated with bone loss
- the disclosure provides a method for treating a disease or condition selected from the group consisting of cancer, autoimmune disease, graft or transplant-related condition, neurodegenerative disease, fibrotic-associated condition, ischemic-related conditions, infection (viral, parasitic or prokaryotic) and diseases associated with bone loss, the method comprising administering to a patient a therapeutically effective amount of carfilzomi b or a pharmaceutically acceptable salt thereof at a concentration of less than 2 mg/mL.
- the disease or condition is cancer.
- cancer and “cancerous” when used herein refer to or describe the physiological condition in subjects that is typically characterized by unregulated cell growth.
- examples of cancer include, without limitation, hematologic malignancies or blood borne cancers such as multiple myeloma and leukemia, and other cancers such as carcinoma, lymphoma, sarcoma, and blastoma. More particular examples of such cancers include squamous cell carcinoma, lung cancer, pancreatic cancer, cervical cancer, bladder cancer, hepatoma, breast cancer, colon carcinoma, and head and neck cancer.
- the term “cancer” as used herein is not limited to any one specific form of the disease, it is believed that the methods of the invention will be particularly effective for cancers, in a subject, which have become resistant in some degree to treatment with anti-cancer agents, including without limitation chemotherapeutic agents, antimitotic agents, anthracyclines and the like, and for cancers which have relapsed post treatment with such anti-cancer agents.
- the cancer is multiple myeloma (e.g., relapsed and/or refractory multiple myeloma or newly diagnosed multiple myeloma).
- the multiple myeloma is relapsed and/or refractory multiple myeloma.
- the multiple myeloma is newly diagnosed multiple myeloma.
- the disclosure provides a method for treating multiple myeloma in a patient, comprising administering to the patient a pharmaceutical composition comprising carfilzomib or a pharmaceutically acceptable salt thereof and an aqueous carrier, wherein the carfilzomib is administered at a dose volume of 2.5 mL/kg or higher.
- the disclosure provides a method for treating multiple myeloma in a patient, comprising administering to the patient a pharmaceutical composition comprising carfilzomib or a pharmaceutically acceptable salt thereof and an aqueous carrier, wherein the carfilzomib is administered at a concentration of less than 2 mg/mL.
- the disclosed methods comprise administering carfilzomib or a pharmaceutically acceptable salt thereof, e.g., in a pharmaceutical composition, at a particular dose and/or dose volume.
- a greater proteasomal inhibition activity from carfilzomib is provided by increasing the volume (i.e., increasing the dose volume).
- a desired proteasomal inhibition response can be achieved by lowering the total dose amount of carfilzomib, for example, by decreasing the carfilzomib concentration and increasing its volume of administration.
- the disclosed methods provide several advantages over certain conventional administration, including but not limited to, lowering the toxicity profile of carfilzomib and providing once-weekly (once-per-week) intravenous dosing for carfilzomib.
- the dose of carfilzomib can be calculated using body surface area (BSA).
- BSA body surface area
- carfilzomib is administered at a dose selected from 10 to 100 mg/m 2 body surface area (e.g., 10, 15, 20, 25, 30, 35, 40, 45, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100 mg/m 2 body surface area).
- carfilzomib is administered at a dose of 15, 20, 27, 36, 45, 56, or 70 mg/m 2 body surface area.
- carfilzomib is administered at a dose of 27, 56, or 70 mg/m 2 body surface area.
- carfilzomib or a pharmaceutically acceptable salt thereof is administered at a dose volume of 2.5 mL/kg or higher, e.g., 2.5, 2.6, 2.7, 2.8, 2.9, 3.0, 3.1 , 3.2, 3.3, 3.4, 3.5, 3.6, 3.7, 3.8, 3.9, 4.0,
- carfilzomib or a pharmaceutically acceptable salt thereof can be administered at a dose volume bounded by, and including, any of the aforementioned values (e.g., 2.5 to 20, 2.6 to 19.9, 2.7 to 19.8, 2.8 to 19.7, 2.9 to 19.6, or 3.0 to 19.5 mL/kg).
- carfilzomib or a pharmaceutically acceptable salt thereof is administered at a dose volume of 2.0 to 3.0 mL/kg, 4.0 to 6.0 mL/kg, 8.0 to 12.0 mL/kg, or 14.0 to 18.0 mL/kg.
- carfilzomib is administered at a dose volume of 5 mL/kg or higher. In some embodiments, carfilzomib or a pharmaceutically acceptable salt thereof is administered at a dose volume of 10 mL/kg or higher. In some embodiments, carfilzomib is administered at a dose volume of 15 mL/kg or higher. In some embodiments, carfilzomib is administered at a dose volume of 16.7 mL/kg.
- carfilzomib or a pharmaceutically acceptable salt thereof is administered at a concentration of less than 2 mg/mL (e.g., 1.9, 1.8, 1.7, 1.6, 1.5, 1.4, 1.3, 1.2, 1.1 , 1.0, 0.9, 0.8, 0.7, 0.6, 0.5, 0.4, 0.3, 0.2, 0.1 or 0.05 mg/mL).
- carfilzomib or a pharmaceutically acceptable salt there can be administered at a concentration bounded by, and including, any of the aforementioned values (e.g., 0.05-1.9, 0.1 -1.8, 0.2-1.7, 0.3-1.6, 0.4-1.5, 0.5-1.4, 0.6-1.3, 0.7-1.2, 0.8-1.1, or 0.9-1.0 mg/mL).
- carfilzomib or a pharmaceutically acceptable salt thereof is administered at a concentration of 1 mg/mL.
- carfilzomib or a pharmaceutically acceptable salt thereof is administered at a concentration of 0.3 mg/mL.
- the carfilzomib or a pharmaceutically acceptable salt thereof is administered in a pharmaceutical composition.
- carfilzomib or a pharmaceutically acceptable salt thereof is administered in a pharmaceutical composition comprising carfilzomib or a pharmaceutically acceptable salt thereof and an aqueous carrier.
- the pharmaceutical composition further comprises a substituted cyclodextrin.
- substituted cyclodextrins include, for example, hydroxypropyl cyclodextrin (HPCD) and sulfobutylether cyclodextrin (SBECD).
- An exemplary SBECD is CAPTISOL® (commercially available from Ligand (San Diego, CA)).
- the substituted cyclodextrin when present, is present in a suitable amount.
- the substituted cyclodextrin can be present in an amount of 2.0 wt% or more of the total composition weight (e.g., 2.0, 2.5, 3.0, 3.5, 4.0, 4.5, 5.0, 5.5, 6.0, 6.5, 7.0, 7.5, 8.0, 8.5, 9.0, 9.5, 10.0, 10.5, 11.0, 11.5, 12.0, 12.5, 13.0, 13.5, 14.0, 14.5, or 15.0 wt% or more of the total composition weight).
- the substituted cyclodextrin can be present in an amount of 35 wt.% or less of the total composition weight (e.g., 35.0, 34.5, 34.0, 33.5, 33.0, 32.5, 32.0, 31.5, 31.0, 30.5, 30.0, 29.5, 29.0, 28.5, 28.0, 27.5, 27.0, 26.5, 26.0, 25.5, 25.0, 24.5, 24.0, 23.5, 23.0, 22.5, 22.0, 21.5, 21.0, 20.5, 20.0, 19.5, 19.0, 18.5, 18.0, 17.5, 17.0, 16.5, 16.0, or 15.5 wt% or less of the total composition weight).
- the total composition weight e.g., 35.0, 34.5, 34.0, 33.5, 33.0, 32.5, 32.0, 31.5, 31.0, 30.5, 30.0, 29.5, 29.0, 28.5, 28.0, 27.5, 27.0, 26.5, 26.0, 25.5, 25.0, 24.5, 24.0, 23.5, 23
- the substituted cyclodextrin can be present in any amount bounded by, and including, any of the aforementioned values (e.g., 2.0 to 35.0, 2.5 to 34.5, 3.0 to 34.0, 3.5 to 33.5, 4.0-33.0, 4.5 to 32.5, 5.0 to 32.0, 5.5 to 31.5, 6.0 to 31 .0, 6.5 to 30.5, 7.0 to 30.0, 7.5 to 29.5, 8.0 to 29.0, 8.5 to 28.5, 9.0 to 28.0, 9.5 to 27.5, 10.0 to 27.0, 10.5 to 26.5, 11.0 to 26.0, 11.5v25.5, 12.0 to 25.0, 12.5 to 24.5, 13.0 to 24.0, 13.5 to 23.5, 14.0 to 23.0, 14.5 to 22.5, 15.0 to 22.0, 15.5 to 21.5, 16.0 to 21.0, 16.5 to 20.5, 17.0 to 20.0, 17.5 to 19.5, or 18.0 to 19.0 wt% of the total composition weight).
- the aforementioned values
- the pharmaceutical composition comprises a buffer.
- the buffer is capable of buffering the pharmaceutical composition between a pH of 2.0 and 5.0.
- buffers include sodium phosphate, citric acid, acetic acid, tromethamine, histidine, gluconic acid, lactic acid, tartaric acid, aspartic acid, and glutamic acid.
- the buffer is a citrate buffer or acetate buffer.
- the buffer is a citrate buffer.
- the composition has a pH of 2.8 to 4.0 (e.g., 2.8, 2.9, 3.0, 3.1 , 3.2, 3.3, 3.4, 3.5, 3.6, 3.7, 3.8, 3.9, or 4.0).
- the composition comprises a suitable solvent (e.g., aqueous carrier).
- a suitable solvent e.g., aqueous carrier
- the solvent comprises water, e.g., water for injection (WFI).
- the pharmaceutical composition comprises (a) carfilzomib, or a pharmaceutically acceptable salt thereof; (b) sulfobutylether cyclodextrin (SBECD); (c) buffer; and (d) water at a pH of 2.8 to 4.0.
- SBECD sulfobutylether cyclodextrin
- Carfilzomib is administered via any suitable route. In some embodiments, is administered subcutaneously or intravenously. In some embodiments, the composition is administered subcutaneously. In some embodiments, the composition is administered by intravenous infusion.
- Carfilzomib is administered on a suitable schedule.
- the pharmaceutical composition is administered twice weekly (e.g., day 1 and 2 of each week).
- the pharmaceutical composition is administered once weekly (e.g., day 1 of each week).
- the method further comprising administering hyaluronidase.
- hyaluronidase is PH20 (EC 3.2.1.35; CAS 37326-33-3), a recombinant human enzyme that catalyzes the degradation of hyaluronic acid in the extracellular matrix to promote diffusion of extracellular substances.
- the hyaluronidase is administered at a suitable dose.
- hyaluronidase is administered at a dose of 50 to 300 units (e.g., 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 105, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, 200, 205, 210, 215, 220, 225, 230, 235, 240, 245, 250, 255, 260, 265, 270, 275, 280, 285, 290, 295, or 300 units).
- the disclosed methods comprise administering carfilzomib or a pharmaceutically acceptable salt thereof with additional active ingredients.
- the method further comprises administering to the patient dexamethasone.
- the method further comprises administering to the patient lenalidomide.
- the method further comprises administering to the patient daratumumab.
- the method further comprises administering to the patient daratumumab and hyaluronidase-fihj.
- the method further comprises administering to the patient isatuximab.
- a method for treating multiple myeloma in a patient comprising administering to the patient a pharmaceutical composition comprising carfilzomib or a pharmaceutically acceptable salt thereof and an aqueous carrier, wherein the carfilzomib is administered at a dose volume of 2.5 mL/kg or higher.
- a method for treating multiple myeloma in a patient comprising administering to the patient a pharmaceutical composition comprising carfilzomib or a pharmaceutically acceptable salt thereof and an aqueous carrier, wherein the carfilzomib is administered at a concentration of less than 2 mg/mL. 4. The method of embodiment 3, wherein the carfilzomib is administered at a concentration of
- a method for treating a patient suffering from a disease or condition selected from the group consisting of cancer, autoimmune disease, graft or transplant-related condition, neurodegenerative disease, fibrotic-associated condition, ischemic-related conditions, infection (viral, parasitic or prokaryotic) and diseases associated with bone loss comprising administering to the patient a therapeutically effective amount of carfilzomib or a pharmaceutically acceptable salt thereof at a dose volume of 10 mL/kg or higher.
- a method for treating a patient suffering from a disease or condition selected from the group consisting of cancer, autoimmune disease, graft or transplant-related condition, neurodegenerative disease, fibrotic-associated condition, ischemic-related conditions, infection (viral, parasitic or prokaryotic) and diseases associated with bone loss comprising administering to the patient a therapeutically effective amount of carfilzomib or a pharmaceutically acceptable salt thereof at a concentration of less than 2 mg/mL.
- substituted cyclodextrin comprises sulfobutylether cyclodextrin (SBECD).
- composition comprises (a) carfilzomib, or a pharmaceutically acceptable salt thereof; (b) sulfobutylether cyclodextrin (SBECD); (c) buffer; and (d) water.
- SBECD sulfobutylether cyclodextrin
- the buffer comprises a citrate buffer, a phosphate buffer, an acetate buffer, or a combination thereof.
- 0 is an integer selected from 0, 1, 2 and 3; each linker is a moiety independently having a structure of
- each R 3 is independently H or CH3 ; n is an integer selected from 1, 2, 3 and 4; each p is, independently, an integer selected from 0, 1, 2, 3 and 4; each q is, independently, an integer selected from 1, 2, 3, 4, 5, 6, 7, 8 and 9; each r is, independently, an integer selected from 0, 1, 2, 3, 4 and 5; and
- PEG is a polyethylene glycol polymeric moiety having a molecular weight of about 500 to about 20,000 g/mol.
- This example demonstrates embodiments of the disclosed methods comprising administering carfilzomib subcutaneously with and without hyaluronidase.
- Carfilzomib was weighed into appropriately sized reaction vessels (e.g., bottle or vial). 10 mM citrate, pH 3.1, 10% CAPTISOL® (Ligand; San Diego, CA) was added to the vessel with compound material to a final volume that resulted in a carfilzomib concentration of 0.3 mg/mL and 1 mg/mL. 10 mM citrate, pH 3.1, 25% CAPTISOL® was added to the vials with compound material to a final volume that resulted in a carfilzomib concentration of 5 mg/mL. The samples were stirred for 20 h at room temperature to allow for dissolution of the material. The samples were aseptically filled into vials, stoppered and capped. The samples were frozen at -70 °C for a period of 1 week prior to transport and dosing.
- CAPTISOL® Ligand; San Diego, CA
- Vehicle solutions were prepared using the same procedure and contained the following: 10 mM citrate, pH 3.1, 10% CAPTISOL® and 10mM citrate, pH 3.1, 25% CAPTISOL®.
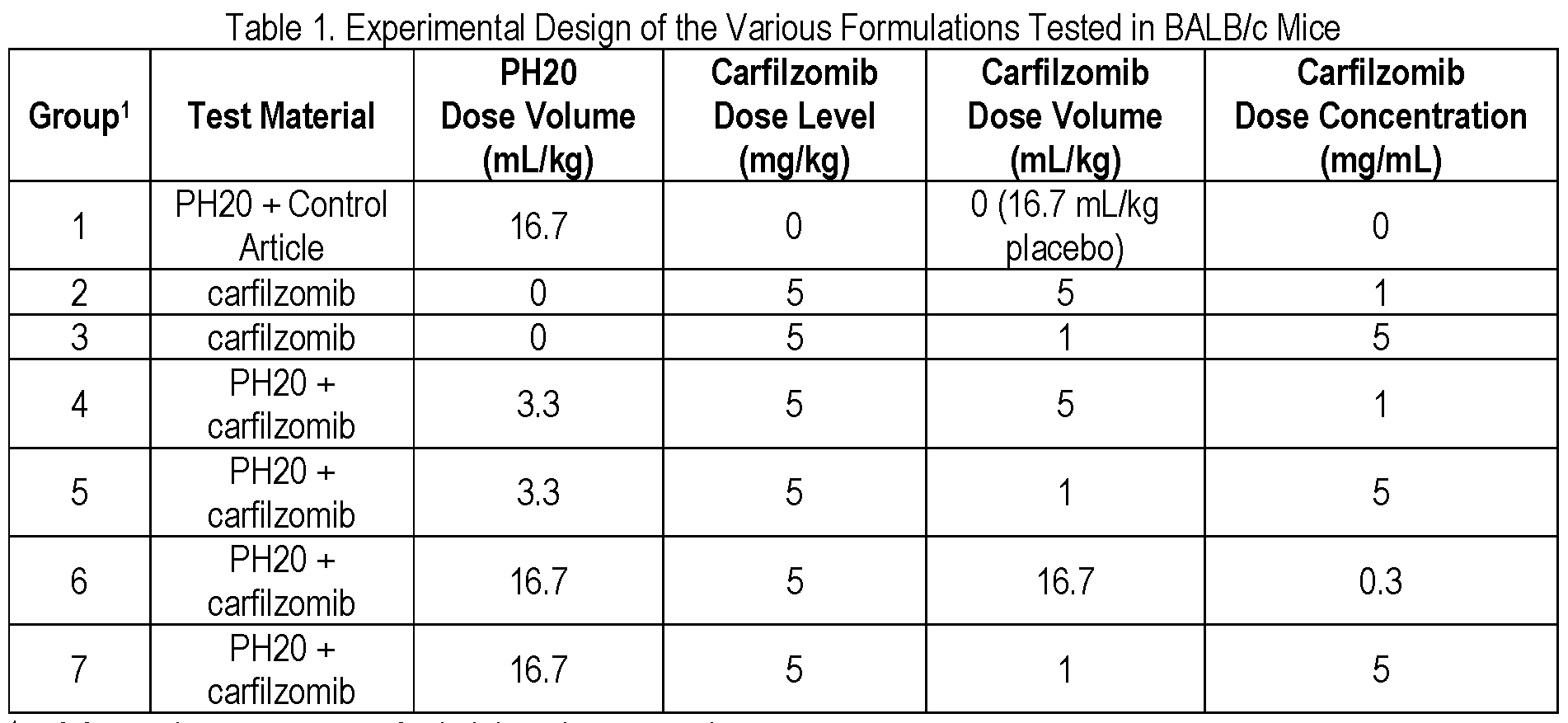
- mice Male BALB/c mice were distributed into seven groups as shown in Table 1 .
- Hyaluronidase (PH20) was administered first to the appropriate animals via subcutaneous injection into the lower flank (caudodorsal back) area once on Day 1 .
- the test and control articles were then administered to the appropriate animals via subcutaneous injection into the lower flank (caudodorsal back) area once on Day 1 .
- the second injection was made as close as possible (e.g., within 5 mm) of the site of the first injection.
- Samples from 6 male animals were collected from each group; 3 mice were bled at 1 h, 4 h and 24 h and 3 mice were bled at 2 h, 6 h, and 24 hours.
- Carfilzomib dose level (mg/kg) was maintained at the same 5 mg/kg among all the groups.
- Table 2 Sample Collection Schedule a Collections at 1, 2, 4, and 6 hours postdose were non-terminal. b Collections at 24 hours postdose were followed immediately by euthanasia. A time range of 24 to 26 hours after the last dose was considered acceptable. 1 .3 Proteasome Activity Assay
- Tissue samples were homogenized for 30 seconds on each side at 30 Hz in a Tissuelyser (Qiagen). Homogenized tissue samples and lysed whole blood samples were then spun at 14,000 rpm in a microcentrifuge at 4 °C for 15 minutes at maximum speed. 180 piL of lysates (supernatant) was then aliquoted into two 384-deep-well blocks. Plates were thawed a single time before analysis. After thawing on ice, lysates were diluted into lysis buffer, and protein concentration was determined via BCA protein assay (Pierce) using a BSA standard.
- BCA protein assay Pieris
- samples were adjusted with Assay Buffer (20 mM Tris, pH 8.0, 0.5 mM EDTA) to suitable 2X concentrations (2 mg/mL for whole blood and 0.7 mg/mL for tissues).
- Assay Buffer (20 mM Tris, pH 8.0, 0.5 mM EDTA)
- 2X concentrations (2 mg/mL for whole blood and 0.7 mg/mL for tissues).
- 2X Suc-LLVY-AMC substrate 120 piM in Assay Buffer containing 2% DMSO was added to the lysate and the fluorescence was monitored over time. Data are reported as the mean percent activity versus control article activity at respective time points.
- Equation 1 100 * (sample nM AMC/sec/pig protein)/ (vehicle AMC/sec/pig protein)
- This example demonstrates embodiments of the disclosed methods and shows the effect of varying the carfilzomib concentration on proteasomal activity for intravenous dosing.
- This example demonstrates embodiments of the disclosed methods and shows the effect of varying the carfilzomib concentration on proteasomal activity for subcutaneous (SC) dosing with or without hyaluronidase.
- This example demonstrates embodiments of the disclosed methods and shows the effect of varying the carfilzomib concentration on proteasomal activity for SC dosing with or without hyaluronidase.
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- Chemical & Material Sciences (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- Medicinal Chemistry (AREA)
- General Health & Medical Sciences (AREA)
- Pharmacology & Pharmacy (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Organic Chemistry (AREA)
- General Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Gastroenterology & Hepatology (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Immunology (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Epidemiology (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Acyclic And Carbocyclic Compounds In Medicinal Compositions (AREA)
Abstract
Provided herein are methods for treating a disease or condition selected from the group consisting of cancer, autoimmune disease, graft or transplant-related condition, neurodegenerative disease, fibrotic-associated condition, ischemic-related conditions, infection (viral, parasitic or prokaryotic) and diseases associated with bone loss, the method comprising administering to a patient a therapeutically effective amount of carfilzomib or a pharmaceutically acceptable salt thereof at a dose volume of 10 mL/kg or higher. Also provided herein are methods for treating multiple myeloma in a patient, comprising administering to the patient a pharmaceutical composition comprising carfilzomib or a pharmaceutically acceptable salt thereof and an aqueous carrier, wherein the carfilzomib is administered at a dose volume of 2.5 mL/kg or higher and/or at a concentration of less than 2 mg/mL.
Description
METHODS OF USING CARFILZOMIB
CROSS-REFERENCE TO RELATED APPLICATION
[0001] This application claims the benefit of priority to U.S. Provisional Patent Application No. 63/518,343, filed August 9, 2023, which is hereby incorporated by reference in its entirety.
BACKGROUND
[0002] Carfilzomib is a proteasomal inhibitor that is currently marketed as KYPROLIS® and is approved in many countries for the treatment of relapsed and/or refractory multiple myeloma (RRMM). In Europe and the US, carfilzomib is approved for use in combination with dexamethasone (Kd), with lenalidomide and dexamethasone (KRd), with daratumumab and dexamethasone (KdD), as well as with isatuximab and dexamethasone (Isa-Kd). Carfilzomib as a single agent is also indicated to treat patients with RRMM in the US. The current route of administration for KYPROLIS® is IV (intravenous) and dosing is based on the body surface area of the patient. The final concentration of reconstituted carfilzomib solution prior to addition into intravenous bags is 2 mg/mL.
SUMMARY
[0003] In some cases, a twice weekly (twice-a-week) intravenous administration may be inconvenient for the patient and present challenges when contemplating administering carfilzomib in combination with other therapies for multiple myeloma. Moreover, there is a known toxicity profile associated with carfilzomib that must be considered in the risk/benefit profile for multiple myeloma patients. Accordingly, once weekly (once-a-week) carfilzomib dosing regimens and methods to lower the toxicity associated with carfilzomib would be useful. In view of the foregoing, it may be useful to have safe and convenient alternative methods for treating a disease or condition (e.g., multiple myeloma) comprising administering carfilzomib or a pharmaceutically acceptable salt thereof.
[0004] Provided herein are methods for treating multiple myeloma in a patient, comprising administering to the patient a pharmaceutical composition comprising carfilzomib or a pharmaceutically acceptable salt thereof and an aqueous carrier, wherein the carfilzomib is administered at a dose volume of 2.5 mL/kg or higher.
[0005] Also provided herein are methods for treating multiple myeloma in a patient, comprising administering to the patient a pharmaceutical composition comprising carfilzomib or a pharmaceutically acceptable salt thereof and an aqueous carrier, wherein the carfilzomib is administered at a concentration of less than 2 mg/mL.
[0006] Also provided herein are methods for treating a disease or condition selected from the group consisting of cancer, autoimmune disease, graft or transplant-related condition, neurodegenerative disease, fibrotic- associated condition, ischemic-related conditions, infection (viral, parasitic or prokaryotic) and diseases associated with bone loss, the method comprising administering to a patient a therapeutically effective amount of carfilzomib or a pharmaceutically acceptable salt thereof at a dose volume of 10 mL/kg or higher.
[0007] Also provided herein are methods for treating a patient suffering from a disease or condition selected from the group consisting of cancer, autoimmune disease, graft or transplant-related condition,
neurodegenerative disease, fibrotic-associated condition, ischemic-related conditions, infection (viral, parasitic or prokaryotic) and diseases associated with bone loss, the method comprising administering to the patient a therapeutically effective amount of carfilzomi b or a pharmaceutically acceptable salt thereof at a concentration of less than 2 mg/mL.
BRIEF DESCRIPTION OF THE FIGURES
[0008] Figure 1 shows the proteasomal inhibition activity displayed by various carfilzomib formulations when administered in mice.
[0009] Figure 2 shows the effect of carfilzomib when administered subcutaneously dosed at 5 mg/kg at a dose. The red line is 5 mg/mL concentration (1 mL/kg) and the black line is 1 mg/mL concentration (5 mL/kg).
[0010] Figure 3A shows the effect of two different concentrations of carfilzomib on proteasomal inhibition over the entire time course of the study.
[0011] Figure 3B shows the effect of two different concentrations of carfilzomib on proteasomal inhibition over the first four hours.
[0012] Figure 3C shows a comparison including data from subcutaneous administration studies.
[0013] Figure 4A shows the effect of two different concentrations of pegylated carfilzomib (PEG-CFZ) on proteasomal inhibition over the entire time course of the study.
[0014] Figure 4B shows the effect of two different concentrations of pegylated carfilzomib on proteasomal inhibition over the first six hours.
[0015] Figure 5 shows the effect of two different concentrations and two different dose levels of pegylated carfilzomib (PEG-CFZ) on proteasomal inhibition over the entire time course of the study.
[0016] Figure 6 shows the effect of two different concentrations of carfilzomib (CFZ) on proteasomal inhibition over the entire time course of the study.
DETAILED DESCRIPTION
[0017] As described herein, the present disclosure provides methods of treating a disease or condition comprising administering carfilzomib or a pharmaceutically acceptable salt thereof at a particular dose volume.
Carfilzomib
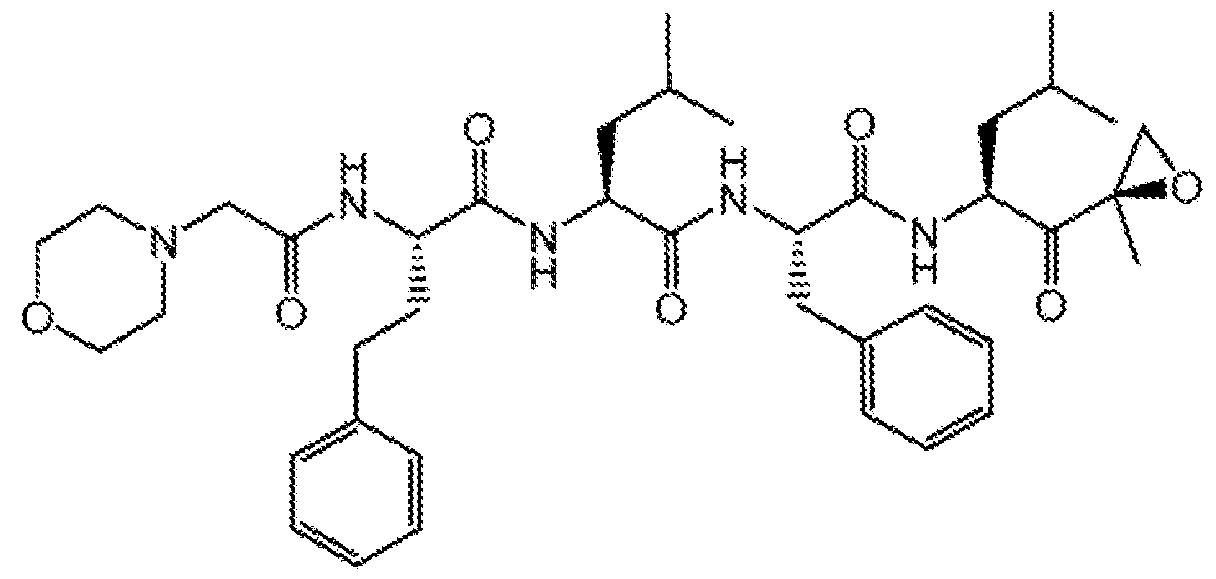
[0018] Carfilzomib is a tetrapeptide epoxy ketone proteasome inhibitor that binds selectively and irreversibly to the constitutive proteasome and immunoproteosome. It has been approved to treat multiple myeloma and is marketed under the name KYPROLIS®. Carfilzomib, polymorphic forms, methods of making, formulations, its use and the like are described in, for example, U.S. Patent Application Publication Nos. 2005/0245435, 2014/0105921 and PCT Patent Publication Nos. WC2006017842, WC2009045497, WC2014169897, WC2013169282, WC2014011695, WC2006063154, WC2014015016, and WC2010048298, each of which is hereby incorporated herein by reference in its entirety.
[0019] Carfilzomib has the following chemical structure:
[0020] The methods disclosed herein comprise administering carfilzomib or a pharmaceutically acceptable salt thereof. As used herein, the term "pharmaceutically acceptable salt" embraces salts commonly used to form alkali metal salts and to form addition salts of free acids or free bases. The nature of the salt is not critical, provided that it is pharmaceutically -acceptable. Suitable pharmaceutically -acceptable acid addition salts of the compound may be prepared from an inorganic acid or from an organic acid. Examples of such inorganic acids include, without limitation, hydrochloric, hydrobromic, hydroiodic, nitric, carbonic, sulfuric and phosphoric acid. Examples of organic acids include, without limitation, aliphatic, cycloaliphatic, aromatic, arylaliphatic, heterocyclic, carboxylic and sulfonic classes of organic acids, examples of which are formic, acetic, adipic, butyric, propionic, succinic, glycolic, gluconic, lactic, malic, tartaric, citric, ascorbic, glucuronic, maleic, fumaric, pyruvic, aspartic, glutamic, benzoic, anthranilic, mesylic, 4-hydroxybenzoic, phenylacetic, mandelic, embonic (pamoic), methane sulfonic, ethanesulfonic, ethanedisulfonic, benzenesulfonic, pantothenic, 2- hydroxyethanesulfonic, toluenesulfonic, sulfanilic, cyclohexylaminosulfonic, camphoric, camphorsulfonic, digluconic, cyclopentanepropionic, dodecylsulfonic, glucoheptanoic, glycerophosphonic, heptanoic, hexanoic, 2- hydroxy-ethanesulfonic, nicotinic, 2 -naphthalene sulfonic, oxalic, palmoic, pectinic, persulfuric, 2- phenylpropionic, picric, pivalic propionic, succinic, tartaric, thiocyanic, mesylic, undecanoic, stearic, algenic, p- hydroxybutyric, salicylic, galactaric and galacturonic acid.
[0021] Suitable pharmaceutically acceptable base addition salts of carfilzomib include, without limitation, metallic salts such as salts made from aluminum, calcium, lithium, magnesium, potassium, sodium and zinc, or salts made from organic bases including primary, secondary, tertiary amines and substituted amines including cyclic amines such as caffeine, arginine, diethylamine, N-ethyl piperidine, histidine, glucamine, isopropylamine, lysine, morpholine, N-ethyl morpholine, piperazine, piperidine, triethylamine, trimethylamine. All of the salts contemplated herein may be prepared by conventional means from the corresponding compound by reacting, for example, the appropriate acid or base with the compound.
[0022] In some embodiments, the carfilzomib is pegylated. Pegylated carfilizomib is disclosed, for example, in PCT Publication No. WO 2017/205392A1 , the disclosure of which is incorporated herein by reference. As used herein, the term "carfilzomib” also includes pegylated carfilzomib as described herein.
[0023] In some embodiments, the pegylated carfilzomib has a structure of Formula I:
(Formula I), or a pharmaceutically acceptable salt thereof, wherein each R1 is independently Ci- alkyl or Csjcycloalkyl; each R2 is independently Ci-ealkyl, -OCH3 or halogen; each 0 is an integer selected from 0, 1, 2, and 3; each linker is independently a moiety independently having a structure of
[0024] each R3 is independently H or CH3; each n is an integer selected from 1, 2, 3, and 4; each p is independently an integer selected from 0, 1, 2, 3, and 4; each q is independently an integer selected from 1, 2, 3, 4, 5, 6, 7, 8, and 9; each r is independently an integer selected from 0, 1, 2, 3, 4, and 5; and PEG is a polyethylene glycol polymeric moiety having a molecular weight of about 500 to about 20,000 g/mol.
[0025] In some embodiments, each R1 is independently Ci- alkyl or Csjcycloalkyl; each R2 is independently Ci-ealkyl, -OCH3 or halogen; each 0 is independently an integer selected from 0, 1, 2, and 3; each linker is independently a moiety having the structure of:
wherein each each R3 is independently H or CH3; and each p is independently an integer selected from 0, 1, 2, 3, and 4; each n is independently an integer selected from 1, 2, 3, and 4; and PEG is a polyethylene glycol polymeric moiety having a molecular weight of about 500 to about 20,000 g/mol.
[0026] In some embodiments, the pegylated carfilzomib compound has a structure of Formula II
, wherein
[0027] each R1 is independently Ci- alkyl or Csjcycloalkyl; each R2 is independently Ci-ealkyl, -OCH3 or halogen; each linker is independently a moiety having the structure of
[0028] wherein each R3 is independently H or CH3; each n is independently an integer selected from 1, 2, 3, and 4; each p is independently an integer selected from 0, 1, 2, 3, and 4; each q is independently an integer selected from 1, 2, 3, 4, 5, 6, 7, 8, and 9; each r is independently an integer selected from 0, 1, 2, 3, 4, and 5;
[0029] each X is independently a counter ion salt selected from a chloride, a bisulfate, a sulfate, a nitrate, a phosphate, an alky-sulfonate or an aryl-sulfonate; and PEG is a polyethylene glycol polymeric moiety having a molecular weight of about 2,000 to about 20,000 g/mol.
[0030] In some embodiments, the disclosure provides a compound of formula I, la, and II, wherein each R1 is independently Ci- alkyl.
[0031] In some embodiments, in conjunction with other above or below embodiments, each R2 independently is H, CH3 or halogen (e.g., F, Br, and Cl).
[0032] In some embodiments, in conjunction with other above or below embodiments, each linker is independently a moiety having the structure of
wherein each R3 is independently H or CH3; each q is independently an integer selected from 1 , 2, 3, 4, and 5; and each r is independently an integer selected from 0, 1 , 2, 3, and 4.
[0033] In some embodiments, in conjunction with other above or below embodiments, each linker is independently a moiety having the structure of
wherein each R3 is independently H or CH3.
[0034] In some embodiments, in conjunction with other above or below embodiments, each linker is independently a moiety having the structure of
wherein R3 is independently H or CH3; each q is independently 4 ; and each r is independently 2.
[0035] In some embodiments, in conjunction with other above or below embodiments, each linker is independently a moiety having the structure of
[0036] In some embodiments, in conjunction with other above or below embodiments, each R3 is H.
[0037] In some embodiments, in conjunction with other above or below embodiments, each R1 is independently methyl, ethyl, propyl, isopropyl, butyl, t-butyl, pently, hexyl or heptyl.
[0038] In some embodiments, in conjunction with other above or below embodiments, each R1 is independently methyl, ethyl, propyl, isopropyl, butyl, t-butyl, pentyl, hexyl or heptyl; and the linker is
[0039] In some embodiments, in conjunction with other above or below embodiments, each R1 is methyl, ethyl, propyl, isopropyl, butyl, t-butyl, pentyl, hexyl or heptyl; and the linker is
[0041] In some embodiments, in conjunction with other above or below embodiments,, the disclosure provides a pegylated carfilzomib compound having the structure of
[0042] wherein each R1 is independently Ci-ioalkyl; each R2 is independently Ci-ealkyl, -OCH3 or halogen; each R3 is independently H or CH3; each X" is a counter anion selected from chloride anion and a alkyl-sulfonate anion; n is 4; and PEG is a polyethylene glycol polymeric moiety having a molecular weight of about 2,000 to about 20,000 g/mol.
[0043] In some embodiments, in conjunction with other above or below embodiments,, the disclosure provides a compound of formula:
wherein X is a halide, a sulfonate or an alkyl-sulfonate counterion salt.
[0044] In some embodiments, in conjunction with other above or below embodiments, the disclosure provides a compound of formula:
[0045] In some embodiments, in conjunction with other above or below embodiments, the disclosure provides a compound of formula:
wherein X is a halide, a sulfonate or an alky l-sulfonate counterion salt.
[0046] In some embodiments, in conjunction with other above or below embodiments, the disclosure provides a compound of formula:
[0047] In some embodiments, in conjunction with other above or below embodiments, each R1 is independently methyl, ethyl, propyl, isopropyl, butyl, t-butyl, pently, hexyl or heptyl; each R2, independently, is CH3 or halogen; linker is a moiety having the structure of
wherein each R3 is independently H or CH3; and PEG is a polyethylene glycol polymeric moiety having a molecular weight of 2000, 3000, 5000 or 20,000 g/mol.
[0048] In some embodiments, in conjunction with other above or below embodiments, each R is independently methyl, ethyl, propyl, isopropyl, butyl, t-butyl, pently, hexyl or heptyl; each R2, independently, is CH3; linker is a moiety having the structure of
wherein each R3 is independently H; and PEG is a polyethylene glycol polymeric moiety having a molecular weight of 3000, 5000 or 20,000 g/mol.
[0059] In some embodiments, in conjunction with other above or below embodiments, the PEG has a weight ranging from about 2K to about 20K g/mol. In some embodiments, the PEG has a weight of 3K, 5K or 20K g/mol.
[0060] In some embodiments, in conjunction with other above or below embodiments, the pharmaceutically acceptable salt of the pegylated carfilzomib is selected from a chloride anion, a bisulfate anion, a sulfate anion, a nitrate anion, a phosphate anion, an alky -sulfonate anion or an aryl-sulfonate anion.
[0061] In some embodiments, the pharmaceutically acceptable salt of the pegylated carfilzomib is a chloride anion or an alky -sulfonate anion.
[0062] In some embodiments, the pharmaceutically acceptable salt of the pegylated carfilzomib is a chloride anion or methane-sulfonate anion.
Diseases and Conditions
[0063] The present disclosure provides methods for treating a disease or condition comprising administering to a patient carfilzomib or a pharmaceutically acceptable salt thereof. As used herein, the terms "treat", "treating", and "treatment" as used herein refer to therapy, including without limitation, curative therapy, prophylactic therapy, and preventative therapy and generally include reversing, reducing, or arresting the symptoms, clinical signs, and underlying pathology of a condition in manner to improve or stabilize a patient's condition.
Prophylactic treatment generally constitutes either preventing the onset of disorders altogether or delaying the onset of a pre-cl inically evident stage of disorders in individuals. The term "prophylactic or therapeutic" treatment is art- recognized and includes administration to the host of one or more of the subject compositions. If it is administered prior to clinical manifestation of the unwanted condition (e.g., disease or other unwanted state of the host animal), or after the condition has subsided, then the treatment is prophylactic (i.e., it protects the host against developing the unwanted condition), whereas if it is administered after manifestation of the unwanted condition, the treatment is therapeutic, (i.e., it is intended to diminish, ameliorate, or stabilize the existing unwanted condition or side effects thereof).
[0064] As used herein, the term "patient" as used herein refers to any mammal, including humans, and animals such as cows, horses, dogs and cats. Thus, the invention may be used in human patients as well as in veterinarian subjects and patients. In some embodiments, carfilzomib or a pharmaceutically acceptable salt thereof is administered to a human patient.
[0065] As used herein, the phrase "therapeutical ly-effective" or "therapeutically effective amount" is intended to quantify the amount of a compound (e.g., carfilzomib or a pharmaceutically acceptable salt thereof), which when administered as part of a desired dosage regimen (to a patient, e.g., a human) alleviates a symptom, ameliorates a condition, or slows the onset of disease conditions according to clinically acceptable standards for the disorder or condition to be treated or the cosmetic purpose, for example, at a reasonable benefit/risk ratio applicable to any medical treatment. Thus, it is the amount of a compound (e.g., carfilzomib or a pharmaceutically acceptable salt thereof) that can treat cancer, whether it is multiple myeloma or other hematologic malignancy or a solid tumor.
[0066] In some embodiments, the disclosure provides a method for treating a disease or condition selected from the group consisting of cancer, autoimmune disease, graft or transplant-related condition, neurodegenerative disease, fibrotic-associated condition, ischemic-related conditions, infection (viral, parasitic or prokaryotic) and diseases associated with bone loss, the method comprising administering to a patient a therapeutically effective amount of carfilzomib or a pharmaceutically acceptable salt thereof at a dose volume of 10 mL/kg or higher.
[0067] In some embodiments, the disclosure provides a method for treating a disease or condition selected from the group consisting of cancer, autoimmune disease, graft or transplant-related condition, neurodegenerative disease, fibrotic-associated condition, ischemic-related conditions, infection (viral, parasitic or prokaryotic) and diseases associated with bone loss, the method comprising administering to a patient a therapeutically effective amount of carfilzomi b or a pharmaceutically acceptable salt thereof at a concentration of less than 2 mg/mL.
[0068] In some embodiments, in conjunction with other embodiments, the disease or condition is cancer. As used herein, the terms "cancer" and "cancerous" when used herein refer to or describe the physiological condition in subjects that is typically characterized by unregulated cell growth. Examples of cancer include, without limitation, hematologic malignancies or blood borne cancers such as multiple myeloma and leukemia, and other cancers such as carcinoma, lymphoma, sarcoma, and blastoma. More particular examples of such cancers include squamous cell carcinoma, lung cancer, pancreatic cancer, cervical cancer, bladder cancer, hepatoma, breast cancer, colon carcinoma, and head and neck cancer. While the term "cancer" as used herein is not limited to any one specific form of the disease, it is believed that the methods of the invention will be particularly effective for cancers, in a subject, which have become resistant in some degree to treatment with anti-cancer agents, including without limitation chemotherapeutic agents, antimitotic agents, anthracyclines and the like, and for cancers which have relapsed post treatment with such anti-cancer agents. In some embodiments, the cancer is multiple myeloma (e.g., relapsed and/or refractory multiple myeloma or newly diagnosed multiple myeloma). In some embodiments, the multiple myeloma is relapsed and/or refractory multiple myeloma. In some embodiments, the multiple myeloma is newly diagnosed multiple myeloma.
[0069] In some embodiments, the disclosure provides a method for treating multiple myeloma in a patient, comprising administering to the patient a pharmaceutical composition comprising carfilzomib or a pharmaceutically acceptable salt thereof and an aqueous carrier, wherein the carfilzomib is administered at a dose volume of 2.5 mL/kg or higher.
[0070] In some embodiments, the disclosure provides a method for treating multiple myeloma in a patient, comprising administering to the patient a pharmaceutical composition comprising carfilzomib or a pharmaceutically acceptable salt thereof and an aqueous carrier, wherein the carfilzomib is administered at a concentration of less than 2 mg/mL.
Dosing
[0071] The disclosed methods comprise administering carfilzomib or a pharmaceutically acceptable salt thereof, e.g., in a pharmaceutical composition, at a particular dose and/or dose volume. Applicant has surprisingly discovered that for a given dose of carfilzomib, a greater proteasomal inhibition activity from carfilzomib is provided by increasing the volume (i.e., increasing the dose volume). Moreover, Applicant has surprisingly discovered that a desired proteasomal inhibition response can be achieved by lowering the total dose amount of carfilzomib, for example, by decreasing the carfilzomib concentration and increasing its volume of administration. The disclosed methods provide several advantages over certain conventional administration,
including but not limited to, lowering the toxicity profile of carfilzomib and providing once-weekly (once-per-week) intravenous dosing for carfilzomib.
[0072] The dose of carfilzomib can be calculated using body surface area (BSA). The BSA can be calculated using any suitable method. In some embodiments, carfilzomib is administered at a dose selected from 10 to 100 mg/m2 body surface area (e.g., 10, 15, 20, 25, 30, 35, 40, 45, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100 mg/m2 body surface area). In some embodiments, in conjunction with other embodiments, carfilzomib is administered at a dose of 15, 20, 27, 36, 45, 56, or 70 mg/m2 body surface area. In some embodiments, in conjunction with other embodiments, carfilzomib is administered at a dose of 27, 56, or 70 mg/m2 body surface area.
Dose Volume
[0073] In some embodiments, carfilzomib or a pharmaceutically acceptable salt thereof is administered at a dose volume of 2.5 mL/kg or higher, e.g., 2.5, 2.6, 2.7, 2.8, 2.9, 3.0, 3.1 , 3.2, 3.3, 3.4, 3.5, 3.6, 3.7, 3.8, 3.9, 4.0,
4.1 , 4.2, 4.3, 4.4, 4.5, 4.6, 4.7, 4.8, 4.9, 5.0, 5.1 , 5.2, 5.3, 5.4, 5.5, 5.6, 5.7, 5.8. 5.9. 6.0, 6.1 , 6.2, 6.3, 6.4, 6.5,
6.6, 6.7, 6.8, 6.9. 7.0, 7.1 , 7.2, 7.3, 7.4, 7.5, 7.6, 7.7, 7.8. 7.9, 8.0, 8.1 , 8.2, 8.3, 8.4, 8.5, 8.6, 8.7, 8.8, 8.9, 9.0,
9.1 , 9.2, 9.3, 9.4, 9.5, 9.6, 9.7, 9.8, 9.9, 10.0, 10.1 , 10.2, 10.3, 10.4, 10.5, 10.6, 10.7, 10.8, 10.9, 11.0, 11.1 , 11.2,
11.3, 11.4, 11.5, 11.6, 11.7, 11.8, 11.9, 12.0, 12.1 , 12.2, 12.3, 12.4, 12.5, 12.6, 12.7. 12.8. 12.9, 13.0, 13.1 , 13.2,
13.3, 13.4, 13.5, 13.6, 13.7, 13.8, 13.9, 14.0, 14.1 , 14.2, 14.3, 14.4, 14.5, 14.6, 14.7, 14.8, 14.9, 15.0, 15.1 , 15.2,
15.3, 15.4, 15.5, 15.6, 15.7, 15.8, 15.9, 16.0, 16.1 , 16.2, 16.3, 16.4, 16.5, 16.6, 16.7, 16.8, 16.9, 17.0, 18.1 , 18.2,
18.3, 18.4, 18.5, 18.6, 18.7, 18.8, 18.9, 19.0, 19.1 , 19.2, 19.3, 19.4, 19.5, 19.6, 19.7, 19.8, 19.9, or 20.0 mL/kg or higher. Thus, carfilzomib or a pharmaceutically acceptable salt thereof can be administered at a dose volume bounded by, and including, any of the aforementioned values (e.g., 2.5 to 20, 2.6 to 19.9, 2.7 to 19.8, 2.8 to 19.7, 2.9 to 19.6, or 3.0 to 19.5 mL/kg). In some embodiments, in conjunction with other embodiments, carfilzomib or a pharmaceutically acceptable salt thereof is administered at a dose volume of 2.0 to 3.0 mL/kg, 4.0 to 6.0 mL/kg, 8.0 to 12.0 mL/kg, or 14.0 to 18.0 mL/kg.
[0074] In some embodiments, carfilzomib is administered at a dose volume of 5 mL/kg or higher. In some embodiments, carfilzomib or a pharmaceutically acceptable salt thereof is administered at a dose volume of 10 mL/kg or higher. In some embodiments, carfilzomib is administered at a dose volume of 15 mL/kg or higher. In some embodiments, carfilzomib is administered at a dose volume of 16.7 mL/kg.
Dose Concentration
[0075] In some embodiments, carfilzomib or a pharmaceutically acceptable salt thereof is administered at a concentration of less than 2 mg/mL (e.g., 1.9, 1.8, 1.7, 1.6, 1.5, 1.4, 1.3, 1.2, 1.1 , 1.0, 0.9, 0.8, 0.7, 0.6, 0.5, 0.4, 0.3, 0.2, 0.1 or 0.05 mg/mL). Thus, carfilzomib or a pharmaceutically acceptable salt there can be administered at a concentration bounded by, and including, any of the aforementioned values (e.g., 0.05-1.9, 0.1 -1.8, 0.2-1.7, 0.3-1.6, 0.4-1.5, 0.5-1.4, 0.6-1.3, 0.7-1.2, 0.8-1.1, or 0.9-1.0 mg/mL). In some embodiments, carfilzomib or a pharmaceutically acceptable salt thereof is administered at a concentration of 1 mg/mL. In some embodiments, carfilzomib or a pharmaceutically acceptable salt thereof is administered at a concentration of 0.3 mg/mL.
Compositions
[0076] In some embodiments, the carfilzomib or a pharmaceutically acceptable salt thereof is administered in a pharmaceutical composition. In some embodiments, carfilzomib or a pharmaceutically acceptable salt thereof is administered in a pharmaceutical composition comprising carfilzomib or a pharmaceutically acceptable salt thereof and an aqueous carrier. In some embodiments, in conjunction with other embodiments, the pharmaceutical composition further comprises a substituted cyclodextrin. -Illustrative nonlimiting substituted cyclodextrins include, for example, hydroxypropyl cyclodextrin (HPCD) and sulfobutylether cyclodextrin (SBECD). An exemplary SBECD is CAPTISOL® (commercially available from Ligand (San Diego, CA)).
[0077] The substituted cyclodextrin (e.g., SBECD), when present, is present in a suitable amount. In some embodiments, the substituted cyclodextrin can be present in an amount of 2.0 wt% or more of the total composition weight (e.g., 2.0, 2.5, 3.0, 3.5, 4.0, 4.5, 5.0, 5.5, 6.0, 6.5, 7.0, 7.5, 8.0, 8.5, 9.0, 9.5, 10.0, 10.5, 11.0, 11.5, 12.0, 12.5, 13.0, 13.5, 14.0, 14.5, or 15.0 wt% or more of the total composition weight). Alternatively, or in addition, the substituted cyclodextrin can be present in an amount of 35 wt.% or less of the total composition weight (e.g., 35.0, 34.5, 34.0, 33.5, 33.0, 32.5, 32.0, 31.5, 31.0, 30.5, 30.0, 29.5, 29.0, 28.5, 28.0, 27.5, 27.0, 26.5, 26.0, 25.5, 25.0, 24.5, 24.0, 23.5, 23.0, 22.5, 22.0, 21.5, 21.0, 20.5, 20.0, 19.5, 19.0, 18.5, 18.0, 17.5, 17.0, 16.5, 16.0, or 15.5 wt% or less of the total composition weight). Thus, the substituted cyclodextrin can be present in any amount bounded by, and including, any of the aforementioned values (e.g., 2.0 to 35.0, 2.5 to 34.5, 3.0 to 34.0, 3.5 to 33.5, 4.0-33.0, 4.5 to 32.5, 5.0 to 32.0, 5.5 to 31.5, 6.0 to 31 .0, 6.5 to 30.5, 7.0 to 30.0, 7.5 to 29.5, 8.0 to 29.0, 8.5 to 28.5, 9.0 to 28.0, 9.5 to 27.5, 10.0 to 27.0, 10.5 to 26.5, 11.0 to 26.0, 11.5v25.5, 12.0 to 25.0, 12.5 to 24.5, 13.0 to 24.0, 13.5 to 23.5, 14.0 to 23.0, 14.5 to 22.5, 15.0 to 22.0, 15.5 to 21.5, 16.0 to 21.0, 16.5 to 20.5, 17.0 to 20.0, 17.5 to 19.5, or 18.0 to 19.0 wt% of the total composition weight). In some embodiments, the SBECD is present in an amount of 2.5 to 30 wt% of the total composition.
[0078] In some embodiments, in conjunction with other embodiments, the pharmaceutical composition comprises a buffer. The buffer is capable of buffering the pharmaceutical composition between a pH of 2.0 and 5.0. Nonlimiting examples of buffers include sodium phosphate, citric acid, acetic acid, tromethamine, histidine, gluconic acid, lactic acid, tartaric acid, aspartic acid, and glutamic acid. In some embodiments, the buffer is a citrate buffer or acetate buffer. In some embodiments, the buffer is a citrate buffer. In some embodiments, the composition has a pH of 2.8 to 4.0 (e.g., 2.8, 2.9, 3.0, 3.1 , 3.2, 3.3, 3.4, 3.5, 3.6, 3.7, 3.8, 3.9, or 4.0).
[0079] The composition comprises a suitable solvent (e.g., aqueous carrier). In some embodiments, the solvent comprises water, e.g., water for injection (WFI).
[0080] In some embodiments, the pharmaceutical composition comprises (a) carfilzomib, or a pharmaceutically acceptable salt thereof; (b) sulfobutylether cyclodextrin (SBECD); (c) buffer; and (d) water at a pH of 2.8 to 4.0.
Route of Administration & Schedule
[0081] Carfilzomib is administered via any suitable route. In some embodiments, is administered subcutaneously or intravenously. In some embodiments, the composition is administered subcutaneously. In some embodiments, the composition is administered by intravenous infusion.
[0082] Carfilzomib is administered on a suitable schedule. In some embodiments, the pharmaceutical composition is administered twice weekly (e.g., day 1 and 2 of each week). In some embodiments, the pharmaceutical composition is administered once weekly (e.g., day 1 of each week).
Additional Agents
[0083] In some embodiments, the method further comprising administering hyaluronidase. An illustrative suitable hyaluronidase is PH20 (EC 3.2.1.35; CAS 37326-33-3), a recombinant human enzyme that catalyzes the degradation of hyaluronic acid in the extracellular matrix to promote diffusion of extracellular substances.
[0084] The hyaluronidase is administered at a suitable dose. In some embodiments, hyaluronidase is administered at a dose of 50 to 300 units (e.g., 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 105, 110, 115, 120, 125, 130, 135, 140, 145, 150, 155, 160, 165, 170, 175, 180, 185, 190, 195, 200, 205, 210, 215, 220, 225, 230, 235, 240, 245, 250, 255, 260, 265, 270, 275, 280, 285, 290, 295, or 300 units).
Combination Therapy
[0085] In some embodiments, the disclosed methods comprise administering carfilzomib or a pharmaceutically acceptable salt thereof with additional active ingredients. In some embodiments, in conjunction with other embodiments the method further comprises administering to the patient dexamethasone. In some embodiments, in conjunction with other embodiments the method further comprises administering to the patient lenalidomide. In some embodiments, in conjunction with other embodiments the method further comprises administering to the patient daratumumab. In some embodiments, in conjunction with other embodiments the method further comprises administering to the patient daratumumab and hyaluronidase-fihj. In some embodiments, in conjunction with other embodiments the method further comprises administering to the patient isatuximab.
EMBODIMENTS
1 . A method for treating multiple myeloma in a patient, comprising administering to the patient a pharmaceutical composition comprising carfilzomib or a pharmaceutically acceptable salt thereof and an aqueous carrier, wherein the carfilzomib is administered at a dose volume of 2.5 mL/kg or higher.
2. The method of embodiment 1 , wherein the carfilzomib is administered at a dose volume of 5 mL/kg or higher.
3. A method for treating multiple myeloma in a patient, comprising administering to the patient a pharmaceutical composition comprising carfilzomib or a pharmaceutically acceptable salt thereof and an aqueous carrier, wherein the carfilzomib is administered at a concentration of less than 2 mg/mL.
4. The method of embodiment 3, wherein the carfilzomib is administered at a concentration of
1 mg/mL.
5. The method of embodiment 3, wherein the carfilzomib is administered at a concentration of 0.3 mg/mL.
6. A method for treating a patient suffering from a disease or condition selected from the group consisting of cancer, autoimmune disease, graft or transplant-related condition, neurodegenerative disease, fibrotic-associated condition, ischemic-related conditions, infection (viral, parasitic or prokaryotic) and diseases associated with bone loss, the method comprising administering to the patient a therapeutically effective amount of carfilzomib or a pharmaceutically acceptable salt thereof at a dose volume of 10 mL/kg or higher.
7. The method of embodiment 6, wherein the disease or condition is cancer.
8. The method of embodiment 7, wherein the cancer is multiple myeloma.
9. The method of any one of embodiments 6-8, wherein the dose volume is 15 mL/kg or higher.
10. The method of embodiment 9, wherein the dose volume is 16.7 mL/kg.
11. A method for treating a patient suffering from a disease or condition selected from the group consisting of cancer, autoimmune disease, graft or transplant-related condition, neurodegenerative disease, fibrotic-associated condition, ischemic-related conditions, infection (viral, parasitic or prokaryotic) and diseases associated with bone loss, the method comprising administering to the patient a therapeutically effective amount of carfilzomib or a pharmaceutically acceptable salt thereof at a concentration of less than 2 mg/mL.
12. The method of embodiment 11, wherein the disease or condition is cancer.
13. The method of embodiment 12, wherein the cancer is multiple myeloma.
14. The method of any one of embodiments 11-13, wherein the concentration is 1 mg/mL or less.
15. The method of embodiment 14, wherein the concentration is 0.3 mg/mL.
16. The method of any one of embodiments 1-15, wherein the carfilzomib is administered at a dose of 27 mg/m2, 56 mg/m2, or 70 mg/m2 body surface area.
17. The method of any one of embodiments 1-16, wherein the carfilzomib is administered in a pharmaceutical composition comprising a substituted cyclodextrin.
18. The method of embodiment 17, wherein the substituted cyclodextrin comprises sulfobutylether cyclodextrin (SBECD).
19. The method of embodiment 17 or 18, wherein the pharmaceutical composition comprises (a) carfilzomib, or a pharmaceutically acceptable salt thereof; (b) sulfobutylether cyclodextrin (SBECD); (c) buffer; and (d) water.
20. The method of embodiment 19, wherein the SBECD is present at 2.5 wt% to 30 wt%.
21. The method of embodiment 19 or 20, wherein the buffer comprises a citrate buffer, a phosphate buffer, an acetate buffer, or a combination thereof.
22. The method of embodiment 21 , wherein the buffer is a citrate buffer.
23. The method of any one of embodiments 1-22, wherein the pharmaceutical composition has a pH of 2.8 to 4.0.
24. The method of any one of embodiments 1-23, wherein the pharmaceutical composition is administered subcutaneously or intravenously.
25. The method of embodiment 24, wherein the pharmaceutical composition is administered subcutaneously.
26. The method of embodiment 24, wherein the pharmaceutical composition is administered by intravenous infusion.
27. The method of any one of embodiments 1-26, wherein the pharmaceutical composition is administered once weekly.
28. The method any one of embodiments 1-27, further comprising administering to the patient hyaluronidase.
29. The method of embodiment 28, wherein the hyaluronidase is PH20.
30. The method of embodiment 28 or 29, wherein hyaluronidase is administered at a dose of 50 to 300 units.
31. The method of any one of embodiments 1-30, further comprising administering to the patient dexamethasone.
32. The method of any one of embodiments 1-31, further comprising administering to the patient lenalidomide.
33. The method of any one of embodiments 1-32, further comprising administering to the patient daratumumab.
34. The method of any one of embodiments 1-33, wherein the carfilzomib is pegylated.
(Formula I), or a pharmaceutically acceptable salt thereof, wherein each R1, independently, is Ci-ioalkyl or Csjcycloalkyl; each R2, independently, is Ci-ealkyl, -OCH3 or halogen;
0 is an integer selected from 0, 1, 2 and 3; each linker is a moiety independently having a structure of
each R3 is independently H or CH3 ; n is an integer selected from 1, 2, 3 and 4; each p is, independently, an integer selected from 0, 1, 2, 3 and 4; each q is, independently, an integer selected from 1, 2, 3, 4, 5, 6, 7, 8 and 9; each r is, independently, an integer selected from 0, 1, 2, 3, 4 and 5; and
PEG is a polyethylene glycol polymeric moiety having a molecular weight of about 500 to about 20,000 g/mol.
EXAMPLES
[0086] The following examples further illustrate the disclosed methods, but of course, should not be construed as in any way limiting its scope.
EXAMPLE 1
[0087] This example demonstrates embodiments of the disclosed methods comprising administering carfilzomib subcutaneously with and without hyaluronidase.
1.1 Carfilzomib Formulation
[0088] Carfilzomib was weighed into appropriately sized reaction vessels (e.g., bottle or vial). 10 mM citrate, pH 3.1, 10% CAPTISOL® (Ligand; San Diego, CA) was added to the vessel with compound material to a final volume that resulted in a carfilzomib concentration of 0.3 mg/mL and 1 mg/mL. 10 mM citrate, pH 3.1, 25% CAPTISOL® was added to the vials with compound material to a final volume that resulted in a carfilzomib concentration of 5 mg/mL. The samples were stirred for 20 h at room temperature to allow for dissolution of the material. The samples were aseptically filled into vials, stoppered and capped. The samples were frozen at -70 °C for a period of 1 week prior to transport and dosing.
[0089] Vehicle solutions were prepared using the same procedure and contained the following: 10 mM citrate, pH 3.1, 10% CAPTISOL® and 10mM citrate, pH 3.1, 25% CAPTISOL®.
1.2 Animal Dosing Study Design
[0090] Male BALB/c mice were distributed into seven groups as shown in Table 1 . Hyaluronidase (PH20) was administered first to the appropriate animals via subcutaneous injection into the lower flank (caudodorsal back) area once on Day 1 . The test and control articles were then administered to the appropriate animals via subcutaneous injection into the lower flank (caudodorsal back) area once on Day 1 . The second injection was made as close as possible (e.g., within 5 mm) of the site of the first injection. Samples from 6 male animals were collected from each group; 3 mice were bled at 1 h, 4 h and 24 h and 3 mice were bled at 2 h, 6 h, and 24 hours. Carfilzomib dose level (mg/kg) was maintained at the same 5 mg/kg among all the groups.
1n=6 for each group; route of administration was subcutaneous
[0091] Blood samples were taken on Day 1 after dosing and tissues (heart, liver, kidney, brain and adrenal glands) were collected for all groups at 24 h post-dose. Pharmacodynamics testing was conducted on the blood pellets from samples collected at 1, 2, 4, 6, and 24 h post-dose (Table 2). The dosing, monitoring, sample collection and processing to blood lysates were performed. Tissues were snap frozen on dry ice after collection. Samples were then shipped to a lab for pharmacodynamic testing. The pharmacodynamic response was determined by measurement of proteasome activity in whole blood (primarily erythrocytes) and tissues using a fluorogenic substrate of the chymotrypsin-like activity of the proteasome.
Table 2: Sample Collection Schedule
a Collections at 1, 2, 4, and 6 hours postdose were non-terminal. b Collections at 24 hours postdose were followed immediately by euthanasia. A time range of 24 to 26 hours after the last dose was considered acceptable.
1 .3 Proteasome Activity Assay
[0092] Proteolysis of a fluorescent tetrapeptide by the chymotrypsin-like (CT-L) activity of the proteasome was used to measure proteasome activity. Whole blood was lysed in 2 volumes of cold (4 °C) lysis buffer (20 mM Tris, pH 8.0, 5 mM EDTA) and snap frozen. Tissues were collected and snap frozen on dry ice. Frozen tissue samples and lysates of whole blood were stored at -80 °C until analyzed. All tissue samples were thawed on ice for 30 min, and then resuspended in cold lysis buffer. 300 piL of lysis buffer was used for approximately 0.1 g samples. Tissue samples were homogenized for 30 seconds on each side at 30 Hz in a Tissuelyser (Qiagen). Homogenized tissue samples and lysed whole blood samples were then spun at 14,000 rpm in a microcentrifuge at 4 °C for 15 minutes at maximum speed. 180 piL of lysates (supernatant) was then aliquoted into two 384-deep-well blocks. Plates were thawed a single time before analysis. After thawing on ice, lysates were diluted into lysis buffer, and protein concentration was determined via BCA protein assay (Pierce) using a BSA standard. Depending on protein concentrations, samples were adjusted with Assay Buffer (20 mM Tris, pH 8.0, 0.5 mM EDTA) to suitable 2X concentrations (2 mg/mL for whole blood and 0.7 mg/mL for tissues). Finally, 2X Suc-LLVY-AMC substrate (120 piM in Assay Buffer containing 2% DMSO) was added to the lysate and the fluorescence was monitored over time. Data are reported as the mean percent activity versus control article activity at respective time points. This number is a simple percent calculation obtained through equation 1 below, where the nM AMC is the concentration of the fluorescent product released in the CT-L proteasome reaction: Equation 1 : 100 * (sample nM AMC/sec/pig protein)/ (vehicle AMC/sec/pig protein)
1 .4 Carfi Izomi b Sample Analysis
[0093] All carfilzomib formulations were uniform, no visible particulates were noted during appearance testing. Endotoxin testing resulted in readings of less than 1.0 EU/mL for all formulations prepared. Aliquots of all pre and post dose formulations were taken and analyzed by HPLC using a 48 -minute reversed phase gradient method with UV detection. All formulations showed main peak carfilzomib percentages in the 97.8-98.5% range. Pre and post dose analysis of carfilzomib showed that active drug was at concentration of within 1 % of target. Carfilzomib samples were stable for the duration of the dosing. No notable decrease of main peaks were observed for any of the samples tested.
1.5 Proteasomal Inhibition Activity of the Various Formulations
[0094] The proteasomal inhibition activity of the various formulations described in Table 1 are shown in Figure 1 and summarized in Table 3. All formulations were dosed at a fixed dose of 5 mg/kg. There were two arms of this study without hyaluronidase.
[0095] As demonstrated by the results shown in Figure 1 and Table 3, the lowest proteasomal inhibition response was observed from the arm with the highest concentration and lowest dose volume (Entry 1; FIG. 1 red line, 5 mg/mL and 1 mL/kg of animal), which had a proteasomal inhibition of 34%. In contrast, the arm dosed at 1 mg/mL and 5 mL/kg, exhibited 59% proteasomal inhibition (Entry 2). In addition, in the arm with 16.7 mL PH20 (hyaluronidase), the 5 mg/mL carfilzomib concentration (1 mL/kg of carfilzomib) exhibited 45% proteasomal
inhibition (Entry 3), wherein the arm at 0.3 mg/mL carfilzomib concentration (16.7 mL/kg of carfilzomib) exhibited more proteasomal inhibition at 77% (Entry 6).
EXAMPLE 2
[0096] This example demonstrates embodiments of the disclosed methods and shows the effect of carfilzomib concentration and dose volume on proteasomal inhibition for a given dose.
[0097] Sample preparation, conduct of animal study, and proteasomal inhibition assays were conducted as described for Example 1. The data is shown in Figure 2 and summarized in Table 4.
[0098] As demonstrated by the results shown in Figure 2 and Table 4, animals dosed at a concentration of 5 mg/mL and a dose volume of 1 mL/kg exhibited 15-50% proteasomal inhibition (Entry 1 ; FIG. 2 red line) (approximately 15% over most time points), whereas animals dosed at a concentration of 1 mg/mL and a dose volume of 5 mL/kg exhibited 50-70% inhibition (Entry 2) (approximately 70% over most time points).
EXAMPLE 3
[0099] This example demonstrates embodiments of the disclosed methods and shows the effect of varying the carfilzomib concentration on proteasomal activity for intravenous dosing.
[0100] Sample preparation, conduct of animal study, and proteasomal inhibition assays were conducted as described for Example 1 with the following exceptions: a) lyophilized, not-for-human use carfilzomib (NHU) was used and b) animals were dosed intravenously instead of subcutaneously. The data are shown in Figures 3A and 3B and summarized in Table 5. Figure 3A shows the results over the entire time course of the study and Figure 3B shows the results over timepoints 0-4 hours.
[0101] As demonstrated by the results shown in Figure 3 and Table 5, animals dosed at a carfilzomib concentration of 5 mg/mL and a dose volume of 1 mL/kg (Entry 1) exhibited approximately 60-75% proteasomal inhibition activity and animals dosed at a carfilzomib concentration of 2 mg/mL and a dose volume of 2.5 mL/kg (Entry 2) exhibited approximately 80-90% proteasomal inhibition (maximum inhibition of 93%; FIG 3B). Figure 3C shows an overlay of this data along with subQ administration at 2 mg/mL and 2.5 mL/kg.
EXAMPLE 4
[0102] This example demonstrates embodiments of the disclosed methods and shows the effect of varying the carfilzomib concentration on proteasomal activity for subcutaneous (SC) dosing with or without hyaluronidase.
[0103] Sample preparation, conduct of animal study, and proteasomal inhibition assays were conducted as described for Example 1 with the following exceptions: a) a dose of 20 mg/kg of pegylated carfilzomib was used and b) combination with hyaluronidase was also tested. The data are shown in Figures 4A and 4B and summarized in Table 6. Figure 4A shows the results over the entire time course of the study and Figure 4B shows the results over timepoints 0-6 hours.
[0104] As demonstrated by the results shown in Figure 4 and Table 6, animals dosed at a carfilzomib concentration of 20 mg/mL and a dose volume of 1 mL/kg (Entry 1) exhibited 100% proteasomal inhibition activity and animals dosed at a carfilzomib concentration of 4 mg/mL and a dose volume of 5 mL/kg (Entry 2) exhibited 98.4% maximum proteasomal inhibition.
EXAMPLE 5
[0105] This example demonstrates embodiments of the disclosed methods and shows the effect of varying the carfilzomib concentration on proteasomal activity for SC dosing with or without hyaluronidase.
[0106] Sample preparation, conduct of animal study, and proteasomal inhibition assays were conducted as described for Example 1 with the following exceptions: a) a dose of 20 and 50 mg/kg of pegylated carfilzomib was used and b) combination with hyaluronidase was tested. The data are shown in Figure 5 and summarized in Table 7. Figure 5 shows the results over the entire time course of the study.
Table 7. Summary of SC Administration of pegylated carfilzomib with or without hyaluronidase.
[0107] As demonstrated by the results shown in Figure 5 and Table 7, animals dosed at a carfilzomib concentration of 4 mg/mL and a dose volume of 1 mL/kg (Entry 1) exhibited 100% proteasomal inhibition activity at 20 mg/kg, and animals dosed at a carfilzomib concentration of 50 mg/mL and a dose volume of 1 mL/kg (Entry 2) dosed at 50 mg/kg exhibited 54.5% maximum proteasomal inhibition.
EXAMPLE 6
[0108] This example demonstrates embodiments of the disclosed methods and shows the effect of varying the carfilzomib concentration on proteasomal activity for IV dosing.
[0109] Sample preparation, conduct of animal study, and proteasomal inhibition assays were conducted as described for Example 1 with the following exceptions: a) lyophilized, NHU carfilzomib was used and b) animals were dosed IV instead of SC. The data are shown in Figure 6 and summarized in Table 8. Figure 6 shows the results over the entire time course of the studies.
[0110] As demonstrated by the results shown in Figure 6 and Table 8, animals dosed at a carfilzomib concentration of 5 mg/mL and a dose volume of 1 mL/kg (Entry 1, Figure 6's top graph) exhibited 70.4% proteasomal inhibition activity, and animals dosed at a carfilzomib concentration of 2 mg/mL and a dose volume of 2.5 mL/kg (Entry 2, Figure 6's bottom graph) exhibited 95% maximum proteasomal inhibition.
[0111] All references, including publications, patent applications, and patents, cited herein are hereby incorporated by reference to the same extent as if each reference were individually and specifically indicated to be incorporated by reference and were set forth in its entirety herein.
[0112] The use of the terms “a” and “an” and “the” and “at least one” and similar referents in the context of describing the invention (especially in the context of the following claims) are to be construed to cover both the singular and the plural, unless otherwise indicated herein or clearly contradicted by context. The use of the term “at least one” followed by a list of one or more items (for example, “at least one of A and B”) is to be construed to mean one item selected from the listed items (A or B) or any combination of two or more of the listed items (A and B), unless otherwise indicated herein or clearly contradicted by context. The terms “comprising,” “having,” “including,” and “containing” are to be construed as open-ended terms (i.e., meaning “including, but not limited to,”) unless otherwise noted. Recitation of ranges of values herein are merely intended to serve as a shorthand method of referring individually to each separate value falling within the range, unless otherwise indicated herein, and each separate value is incorporated into the specification as if it were individually recited herein. All methods described herein can be performed in any suitable order unless otherwise indicated herein or otherwise clearly contradicted by context. The use of any and all examples, or exemplary language (for example, “such as”) provided herein, is intended merely to better
illuminate the invention and does not pose a limitation on the scope of the invention unless otherwise claimed. No language in the specification should be construed as indicating any non-claimed element as essential to the practice of the invention.
Claims
1 . A method for treating multiple myeloma in a patient, comprising administering to the patient a pharmaceutical composition comprising carfilzomib or a pharmaceutically acceptable salt thereof and an aqueous carrier, wherein the carfilzomib is administered at a dose volume of 2.5 mL/kg or higher.
2. The method of claim 1, wherein the carfilzomib is administered at a dose volume of 5 mL/kg or higher.
3. A method for treating multiple myeloma in a patient, comprising administering to the patient a pharmaceutical composition comprising carfilzomib or a pharmaceutically acceptable salt thereof and an aqueous carrier, wherein the carfilzomib is administered at a concentration of less than 2 mg/mL.
4. The method of claim 3, wherein the carfilzomib is administered at a concentration of 1 mg/mL.
5. The method of claim 3, wherein the carfilzomib is administered at a concentration of 0.3 mg/mL.
6. A method for treating a patient suffering from a disease or condition selected from the group consisting of cancer, autoimmune disease, graft or transplant-related condition, neurodegenerative disease, fibrotic-associated condition, ischemic-related conditions, infection (viral, parasitic or prokaryotic) and diseases associated with bone loss, the method comprising administering to the patient a therapeutically effective amount of carfilzomib or a pharmaceutically acceptable salt thereof at a dose volume of 10 mL/kg or higher.
7. The method of claim 6, wherein the disease or condition is cancer.
8. The method of claim 7, wherein the cancer is multiple myeloma.
9. The method of any one of claims 6-8, wherein the dose volume is 15 mL/kg or higher.
10. The method of claim 9, wherein the dose volume is 16.7 mL/kg.
11. A method for treating a patient suffering from a disease or condition selected from the group consisting of cancer, autoimmune disease, graft or transplant-related condition, neurodegenerative disease, fibrotic-associated condition, ischemic-related conditions, infection (viral, parasitic or prokaryotic) and diseases associated with bone loss, the method comprising administering to the patient a therapeutically effective amount of carfilzomib or a pharmaceutically acceptable salt thereof at a concentration of less than 2 mg/mL.
12. The method of claim 11, wherein the disease or condition is cancer.
13. The method of claim 12, wherein the cancer is multiple myeloma.
14. The method of any one of claims 11-13, wherein the concentration is 1 mg/mL or less.
15. The method of claim 14, wherein the concentration is 0.3 mg/mL.
16. The method of any one of claims 1-15, wherein the carfilzomib is administered at a dose of 27 mg/m2, 56 mg/m2, or 70 mg/m2 body surface area.
17. The method of any one of claims 1-16, wherein the carfilzomib is administered in a pharmaceutical composition comprising a substituted cyclodextrin.
18. The method of claim 17, wherein the substituted cyclodextrin comprises sulfobutylether cyclodextrin (SBECD).
19. The method of claim 17 or 18, wherein the pharmaceutical composition comprises (a) carfilzomib, or a pharmaceutically acceptable salt thereof; (b) sulfobutylether cyclodextrin (SBECD); (c) buffer; and (d) water.
20. The method of claim 19, wherein the SBECD is present at 2.5 wt% to 30 wt%.
21. The method of claim 19 or 20, wherein the buffer comprises a citrate buffer, a phosphate buffer, an acetate buffer, or a combination thereof.
22. The method of claim 21, wherein the buffer is a citrate buffer.
23. The method of any one of claims 1-22, wherein the pharmaceutical composition has a pH of
2.8 to 4.0.
24. The method of any one of claims 1-23, wherein the pharmaceutical composition is administered subcutaneously or intravenously.
25. The method of claim 24, wherein the pharmaceutical composition is administered subcutaneously.
26. The method of claim 24, wherein the pharmaceutical composition is administered by intravenous infusion.
27. The method of any one of claims 1-26, wherein the pharmaceutical composition is administered once-weekly.
28. The method any one of claims 1-27, further comprising administering to the patient hyaluronidase.
29. The method of claim 28, wherein the hyaluronidase is PH20.
30. The method of claim 28 or 29, wherein hyaluronidase is administered at a dose of 50 to 300 units.
31. The method of any one of claims 1-30, further comprising administering to the patient dexamethasone.
32. The method of any one of claims 1-31, further comprising administering to the patient lenalidomide.
33. The method of any one of claims 1-32, further comprising administering to the patient daratumumab.
34. The method of any one of claims 1-33, wherein the carfilzomib is pegylated.
35. The method of claim 34, wherein the pegylated carfilzomib has a structure of Formula I:
(Formula I), or a pharmaceutically acceptable salt thereof, wherein each R1, independently, is Ci-ioalkyl or Csjcycloalkyl; each R2, independently, is Ci-ealkyl, -OCH3 or halogen;
0 is an integer selected from 0, 1, 2 and 3; each linker is a moiety independently having a structure of
each R3 is independently H or CH3 ; n is an integer selected from 1, 2, 3 and 4; each p is, independently, an integer selected from 0, 1, 2, 3 and 4; each q is, independently, an integer selected from 1, 2, 3, 4, 5, 6, 7, 8 and 9; each r is, independently, an integer selected from 0, 1, 2, 3, 4 and 5; and
PEG is a polyethylene glycol polymeric moiety having a molecular weight of about 500 to about 20,000 g/mol.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| AU2024321532A AU2024321532A1 (en) | 2023-08-09 | 2024-08-08 | Diluted carfilzomib for use in treating multiple myeloma |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US202363518343P | 2023-08-09 | 2023-08-09 | |
| US63/518,343 | 2023-08-09 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO2025035020A2 true WO2025035020A2 (en) | 2025-02-13 |
| WO2025035020A3 WO2025035020A3 (en) | 2025-03-20 |
Family
ID=92582818
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2024/041550 Pending WO2025035020A2 (en) | 2023-08-09 | 2024-08-08 | Methods of using carfilzomib |
Country Status (2)
| Country | Link |
|---|---|
| AU (1) | AU2024321532A1 (en) |
| WO (1) | WO2025035020A2 (en) |
Citations (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20050245435A1 (en) | 2004-04-15 | 2005-11-03 | Proteolix, Inc. | Compounds for enzyme inhibition |
| WO2006063154A1 (en) | 2004-12-07 | 2006-06-15 | Proteolix, Inc. | Composition for proteasome inhibition |
| WO2009045497A1 (en) | 2007-10-04 | 2009-04-09 | Proteolix, Inc. | Crystalline peptide epoxy ketone protease inhibitors and the synthesis of amino acid keto-epoxides |
| WO2010048298A1 (en) | 2008-10-21 | 2010-04-29 | Proteolix, Inc. | Combination therapy with peptide epoxyketones |
| WO2013169282A1 (en) | 2012-05-08 | 2013-11-14 | Onyx Therapeutics, Inc. | Cylodextrin complexation methods for formulating peptide proteasome inhibitors |
| WO2014011695A2 (en) | 2012-07-09 | 2014-01-16 | Onyx Therapeutics, Inc. | Prodrugs of peptide epoxy ketone protease inhibitors |
| WO2014015016A1 (en) | 2012-07-18 | 2014-01-23 | Onyx Therapeutics, Inc. | Liposomal compositions of epoxyketone-based proteasome inhibitors |
| WO2014169897A1 (en) | 2013-04-19 | 2014-10-23 | Glatt Systemtechnik Gmbh | Device for introducing a defined quantity of a second powder into a process vessel |
| WO2017205392A1 (en) | 2016-05-24 | 2017-11-30 | Amgen Inc. | Pegylated carfilzomib compounds |
Family Cites Families (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2018138557A1 (en) * | 2017-01-24 | 2018-08-02 | Orbicular Pharmaceutical Technologies Pvt. Ltd. | Ready-to-use carfilzomib compositions |
-
2024
- 2024-08-08 AU AU2024321532A patent/AU2024321532A1/en active Pending
- 2024-08-08 WO PCT/US2024/041550 patent/WO2025035020A2/en active Pending
Patent Citations (11)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20050245435A1 (en) | 2004-04-15 | 2005-11-03 | Proteolix, Inc. | Compounds for enzyme inhibition |
| WO2006017842A1 (en) | 2004-08-06 | 2006-02-16 | Proteolix, Inc. | Compounds for proteasome enzyme inhibition |
| WO2006063154A1 (en) | 2004-12-07 | 2006-06-15 | Proteolix, Inc. | Composition for proteasome inhibition |
| WO2009045497A1 (en) | 2007-10-04 | 2009-04-09 | Proteolix, Inc. | Crystalline peptide epoxy ketone protease inhibitors and the synthesis of amino acid keto-epoxides |
| WO2010048298A1 (en) | 2008-10-21 | 2010-04-29 | Proteolix, Inc. | Combination therapy with peptide epoxyketones |
| WO2013169282A1 (en) | 2012-05-08 | 2013-11-14 | Onyx Therapeutics, Inc. | Cylodextrin complexation methods for formulating peptide proteasome inhibitors |
| WO2014011695A2 (en) | 2012-07-09 | 2014-01-16 | Onyx Therapeutics, Inc. | Prodrugs of peptide epoxy ketone protease inhibitors |
| US20140105921A1 (en) | 2012-07-09 | 2014-04-17 | Onyx Therapeutics, Inc. | Prodrugs of Peptide Epoxy Ketone Protease Inhibitors |
| WO2014015016A1 (en) | 2012-07-18 | 2014-01-23 | Onyx Therapeutics, Inc. | Liposomal compositions of epoxyketone-based proteasome inhibitors |
| WO2014169897A1 (en) | 2013-04-19 | 2014-10-23 | Glatt Systemtechnik Gmbh | Device for introducing a defined quantity of a second powder into a process vessel |
| WO2017205392A1 (en) | 2016-05-24 | 2017-11-30 | Amgen Inc. | Pegylated carfilzomib compounds |
Non-Patent Citations (1)
| Title |
|---|
| no. 37326-33-3 |
Also Published As
| Publication number | Publication date |
|---|---|
| WO2025035020A3 (en) | 2025-03-20 |
| AU2024321532A1 (en) | 2026-01-29 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| ES2527870T3 (en) | Pharmaceutical formulations containing methylnaltrexone | |
| Chen et al. | A stapled peptide antagonist of MDM2 carried by polymeric micelles sensitizes glioblastoma to temozolomide treatment through p53 activation | |
| TWI489984B (en) | Formulations for parenteral delivery of compounds and uses thereof | |
| US20100178328A1 (en) | Combination therapy for ovarian cancer | |
| CN101244255B (en) | Thymus gland pentapeptide injection and uses thereof | |
| US20140140925A1 (en) | Folate-targeted diagnostics and treatment | |
| JP6106228B2 (en) | Aqueous solutions containing 3-quinuclidinone for the treatment of hyperproliferative diseases, autoimmune diseases, and heart diseases | |
| US20230100247A1 (en) | Taxol conjugate compounds, pharmaceutical compositions comprising the same, and methods for their use | |
| KR20200052326A (en) | Formulation of Copanlis | |
| WO2025035020A2 (en) | Methods of using carfilzomib | |
| PL209147B1 (en) | Compositions comprising epothilones and their use for the treatment of carcinoid syndrome | |
| WO2022193898A1 (en) | Polypeptide coupled boron carrying agent, preparation method therefor, and pharmaceutical formulation | |
| US20210322519A1 (en) | Pharmaceutical compositions of ghrh analogs and uses thereof | |
| AU688305B2 (en) | Process to prepare pharmaceutical compositions containing vecuronium bromide | |
| AU2019289886B2 (en) | Composition containing a somatostatin analogue for radiopharmaceutical use | |
| CN116963732A (en) | Pharmaceutical compositions and preparation methods thereof | |
| CN114344333A (en) | Use of animal non-pathogenic cell-related components and pharmaceutical compositions comprising the same | |
| WO2024175055A1 (en) | Taxol conjugate compounds, compositions and methods for their use | |
| CA3037757A1 (en) | Pharmaceutical compositions of ghrh analogs and uses thereof | |
| WO2023013755A1 (en) | Cancer peptide vaccine cocktail formulation amd method for producing same | |
| WO2024178236A1 (en) | Taxol conjugate compounds and pharmaceutical compositions thereof | |
| EP2240194A1 (en) | Treatment of melanoma with alpha thymosin peptides in combination with a kinase inhibitor | |
| CA3183299A1 (en) | Low-dose pharmaceutical compositions of ghrh analogs and uses thereof | |
| EP2328411A1 (en) | Pharmaceutical composition of a radioiodinated benzamide derivative and methods of making the same | |
| US20140350273A1 (en) | Pediatric uses of cabazitaxel |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 24762493 Country of ref document: EP Kind code of ref document: A2 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: AU2024321532 Country of ref document: AU |
|
| ENP | Entry into the national phase |
Ref document number: 2024321532 Country of ref document: AU Date of ref document: 20240808 Kind code of ref document: A |