AGENTS FOR DELIVERY OF NUCLEIC ACIDS CROSS-REFERENCE This application claims priority to U.S. Application No.63/518,216, filed August 8, 2024 and U.S. Application No.63/679,895 file August 6, 2024. BACKGROUND [0001] Transfection is the process of introducing nucleic acids into eukaryotic cells by non-viral methods. Transfection methods allow the introduction of negatively charged molecules (e.g. phosphate backbones of DNA and RNA) into cells having a negatively charged membrane. Chemicals such as calcium phosphate and DEAE-dextran, or cationic lipid-based reagents coat the DNA, neutralizing or even creating an overall positive charge to the molecule. The DNA-transfection reagent complex easily crosses the cell membrane, especially for lipids that have a “fusogenic” component, which enhances fusion with the lipid bilayer of the cell. [0002] With the recent advances in nucleic-acid based therapeutics, and continued need for transfection reagents with low toxicity, there is a continued need for novel ionizable lipids that can be used in vitro and in vivo. SUMMARY [0003] Disclosed herein are compounds, compositions and methods that improve the efficiency of introducing macromolecules, such as nucleic acids, or small molecules (e.g, therapeutics), into cells. Compounds are provided, together with compositions containing these compounds and methods for using these new compounds and compositions for delivery of payloads, (e.g. a nucleic acid or small molecule), to cells. The compounds may be used alone for transfection, or they may be used in combination with additional reagents in transfection compositions. For example, the new compounds may be combined with one or more ionizable lipids and/or neutral lipids, with one or more cell surface ligands, with one or more fusion enhancing agents, and with one or more nuclear localization agents and one or more amphipathic peptides and
any combinations thereof. The resulting compositions may be complexed with one or more macromolecules (e.g,nucleic acids, such as DNA or RNA, proteins, ribonucleoproteins, and the like) and used to deliver these macromolecules into cells. [0004] Specifically there is provided a compound having the structure of Formula (I): O

where each R
1 is optionally substituted C
12-C
20 straight-chain or branched-chain alkyl, optionally substituted C
12-C
20 monounsaturated or polyunsaturated straight-chain or branched-chain alkenyl, -(CH2)(1-5)-O(CO)-(CH2)(10-18)-CH3, –(CO)C12-C20 alkyl, or –(CO)C
12-C
20 alkenyl; R
2 is optionally substituted C
1-C
20 straight-chain or branched-chain alkyl or optionally substituted C2-C20 monounsaturated straight-chain or branched-chain alkenyl; R
3 is optionally substituted C1-C6 straight-chain or branched-chain alkyl, optionally substituted C
2-C
6 monounsaturated or polyunsaturated straight-chain or branched-chain alkenyl, or -(CH2)0-3Het or AA; R
4 is H, optionally substituted C1-C6 straight-chain or branched-chain alkyl, or optionally substituted C
2-C
6 monounsaturated or polyunsaturated straight-chain or branched-chain alkenyl;
R
5 is H, an amine protecting group, –(CO)C1-C23 alkyl, –(CO)C2-C23 alkenyl or ; substituted C
1-C
20 straight-chain or branched-chain alkyl or
C20 monounsaturated straight-chain or branched-chain alkenyl; AA represents any natural or non-natural amino acid side chain; p is an integer from 1 to 50; and Het is an optionally substituted 5-7 membered monocyclic basic heterocycle, or an optionally substituted 8-11 membered bicyclic basic heterocyclyl. [0005] Also provided are compositions containing a compound as described above and at least one cationic lipid, and/or at least one neutral lipid. The cationic lipid may be LipofectAmine
™ 2000, LipofectAmine
™, Lipofectin®, DMRIE-C, CellFectin®(Invitrogen), Oligofectamine®(Invitrogen), LipofectAce® ( Invitrogen ), Fugene® (Roche, Basel, Switzerland), Fugene® HD ( Roche), Transfectam® (Transfectam, Promega, Madison, WI), Tfx-10® (Promega), Tfx-20® (Promega), Tfx- 50® ( Promega ), Transfectin
™ (BioRad, Hercules, CA), SilentFect
™(Bio-Rad), Effectene® (Qiagen, Valencia, CA), DC-chol ( Avanti Polar Lipids), GenePorter® (Gene Therapy Systems, San Diego, CA), DharmaFect 1® (Dharmacon, Lafayette, CO), DharmaFect 2® (Dharmacon), DharmaFect 3® (Dharmacon), DharmaFect 4® ( Dharmacon), Escort
™ III (Sigma, St. Louis, MO), Escort
™ IV (Sigma ), DOTMA, DOTAP, DMRIE, DC-Chol, DDAB, DOSPA, DOSPER, DOGS, TMTPS, TMTOS, TMTLS, TMTMS, TMDOS, N-1-dimethyl-N-1-(2,3-diaoleoyloxypropyl)-2- hydroxypropane-1,3-diamine, N-1-dimethyl-N-1-(2,3-diamyristyloxypropyl)-2- hydroxypropane-1,3-diamine, N-1-dimethyl-N-1-(2,3-diapalmityloxypropyl)-2- hydroxypropane-1,3-diamine, N-1-dimethyl-N-1-(2,3-diaoleoyloxypropyl)-2-(3-amino-2- hydroxypropyloxy)propane-1,3-diamine, N-1-dimethyl-N-1-(2,3-diamyristyloxypropyl)- 2-(3-amino-2-hydroxypropyloxy)propane-1,3-diamine, N-1-dimethyl-N-1-(2,3-
diapalmityloxypropyl)-2-(3-amino-2-hydroxypropyloxy)propane-1,3-diamine, L- spermine-5-carboxyl-3-(DL-1,2-dipalmitoyl-dimethylaminopropyl-ȕ-hydroxyethylamine, 3,5-(N,N-di-lysyl)-diaminobenzoyl-glycyl-3-(DL-1,2-dipalmitoyl-dimethylaminopropyl- ȕ-hydroxyethylamine), L-Lysine-bis(O,O'-oleoyl-ȕ-hydroxyethyl)amide dihydrochloride, L-Lysine-bis-(O,O'-palmitoyl-ȕ-hydroxyethyl)amide dihydrochloride, 1,4-bis[(3-(3- aminopropyl)-alkylamino)-2-hydroxypropyl)piperazine, L-Lysine-bis-(O,O'-myristoyl-ȕ- hydroxyethyl)amide dihydrochloride, L-Ornithine-bis-(O,O'-myristoyl-ȕ- hydroxyethyl)amide dihydrochloride, L-Ornithine-bis-(O,O'-oleoyl-ȕ- hydroxyethyl)amide dihydrochloride, 1,4-bis[(3-(3-aminopropyl)-oleylamino)-2- hydroxypropyl]piperazine, L-Ornithine-bis-(O,O'-palmitoyl-ȕ-hydroxyethyl)amide dihydrochloride, 1,4,-bis[(3-amino-2-hydroxypropyl)-oleylamino]-butane-2,3-diol, 1,4,- bis[(3-amino-2-hydroxypropyl)-palmitylamino]-butane-2,3-diol, 1,4,-bis[(3-amino-2- hydroxypropyl)-myristylamino]-butane-2,3-diol, 1,4-bis[(3- oleylamino)propyl]piperazine, L-Arginine-bis-(O,O'-oleoyl-ȕ-hydroxyethyl)amide dihydrochloride, bis[(3-(3-aminopropyl)-myristylamino)2-hydroxypropyl]piperazine, L- Arginine-bis-(O,O'-palmitoyl-ȕ-hydroxyethyl)amide dihydrochloride, L-Serine-bis-(O,O'- oleoyl-ȕ-hydroxyethyl)amide dihydrochloride, 1,4-bis[(3-(3-aminopropyl)- palmitylamino)-2-hydroxypropyl]piperazine, Glycine-bis-(O,O'-palmitoyl-ȕ- hydroxyethyl)amide dihydrochloride, Sarcosine-bis-(O,O'-palmitoyl-ȕ- hydroxyethyl)amide dihydrochloride, L-Histidine-bis-(O,O'-palmitoyl-ȕ- hydroxyethyl)amide dihydrochloride, cholesteryl-^ȕ-carboxyl- amidoethylenetrimethylammonium iodide, 1,4-bis[(3-myristylamino)propyl]piperazine, 1-dimethylamino-3-trimethylammonio-DL-2-propyl-cholesteryl carboxylate iodide, cholesteryl-^ȕ-carboxyamidoethyleneamine, cholesteryl-^ȕ- oxysuccinamidoethylenetrimethylammonium iodide, 1-dimethylamino-3- trimethylammonio-DL-2-propyl-cholesteryl-^ȕ-oxysuccinate iodide, 2-[(2- trimethylammonio)-ethylmethylamino] ethyl-cholesteryl-^ȕ-R[\VXFFLQDWH^ LRGLGH^^ ^ȕ>1- (N', N'-GLPHWK\ODPLQRHWKDQH^FDUEDPR\O@FKROHVWHURO^^ DQG^ ^ȕ-[N-(polyethyleneimine)- carbamoyl] cholesterol,1,4-bis[(3-palmitylamino)propyl]piperazine, L-Ornithylglycyl-N- (1-heptadecyloctadecyl)glycinamide, N
2,N
5 -Bis(3-aminopropyl)-L-ornithylglycyl-N- (1-
heptadecyloctadecyl)glycinamide, 1,4-bis[(3-(3-amino-2-hydroxypropyl)-alkylamino)-2- hydroxypropyl]piperazine N
2-[N
2,N
5 -Bis(3-aminopropyl)-L-ornithyl]-N,N-dioctadecyl- L-glutamine,N
2-[N
2,N
5 -Bis(aminopropyl)-L-ornithyl]-N-N-dioctadecyl-L-Į-glutamine, 1,4-bis[(3-(3-amino-2-hydroxypropyl)-oleylamino)2-hydroxypropyl]piperazine, N
2- [N
2,N
5 -Bis(aminopropyl)-L-ornithyl]-N-N-dioctadecyl-L-Į-asparagine, N-[N
2-[N
2,N
5- Bis[(1,1-dimethylethoxy)carbonyl]- N
2,N
5-bis[3-[(1,1- dimethylethoxy)carbonyl]aminopropyl]-L-ornithyl-N-N-dioctadecyl-L-glutaminyl]-L- glutamic acid, N
2-[N
2,N
5 -Bis(3-aminopropyl)-L-ornithyl]-N,N-diolyl-L-glutamine, N
2- [N
2,N
5 -Bis(aminopropyl)-L-ornithyl]-N-N-dioleyl-L-Į-glutamine,4-bis[(3-(3-amino-2- hydoxypropyl)-myristylamino)-2-hydroxypropyl]piperazine, N
2-[N
2,N
5 - Bis(aminopropyl)-L-ornithyl]-N-N-dioleyl-L-Į-asparagine, N-[N
2-[N
2,N
5-Bis[(1,1- dimethylethoxy)carbonyl]- N
2,N
5-bis[3-[(1,1-dimethylethoxy)carbonyl]aminopropyl]-L- ornithyl-N-N-dioleyl-L-glutaminyl]-L-glutamic acid, 1,4-bis[(3-(3-aminopropyl)- oleylamino)propyl]piperazine, N
2-[N
2,N
5 -Bis(3-aminopropyl)-L-ornithyl]-N,N- dipalmityl-L-glutamine,N
2-[N
2,N
5 -Bis(aminopropyl)-L-ornithyl]-N-N-dipalmityl-L-Į- glutamine, N
2-[N
2,N
5 -Bis(aminopropyl)-L-ornithyl]-N-N-dipalmityl-L-Į-asparagine, N- [N
2-[N
2,N
5-Bis[(1,1-dimethylethoxy)carbonyl]- N
2,N
5-bis[3-[(1,1- dimethylethoxy)carbonyl]aminopropyl]-L-ornithyl-N-N-dipalmityl-L-glutaminyl]-L- glutamic acid, N
2-[N
2,N
5 -Bis(3-aminopropyl)-L-ornithyl]-N,N-dimyristyl-L- glutamine, N
2-[N
2,N
5 -Bis(aminopropyl)-L-ornithyl]-N-N-dimyristyl-L-Į-glutamine, N
2- [N
2,N
5 -Bis(aminopropyl)-L-ornithyl]-N-N-dimyristyl-L-Į-asparagine, 1,4-bis[(3-(3- amino-2-hydroxypropyl)-palmitylamino)-2-hydroxypropyl]piperazine, N-[N
2-[N
2,N
5- Bis[(1,1-dimethylethoxy)carbonyl]- N
2,N
5-bis[3-[(1,1- dimethylethoxy)carbonyl]aminopropyl]-L-ornithyl-N-N-dimyristyl-L-glutaminyl]-L- glutamic acid, 1,4-bis[(3-(3-aminopropyl)-myristylamino)propyl]piperazine, N
2-[N
2,N
5 - Bis(3-aminopropyl)-L-ornithyl]-N,N-dilaureyl-L-glutamine, N
2-[N
2,N
5 - Bis(aminopropyl)-L-ornithyl]-N-N-dilaureyl-L-Į-glutamine, N
2-[N
2,N
5 - Bis(aminopropyl)-L-ornithyl]-N-N-dilaureyl-L-Į-asparagine, N-[N
2-[N
2,N
5-Bis[(1,1- dimethylethoxy)carbonyl]- N
2,N
5-bis[3-[(1,1-dimethylethoxy)carbonyl]aminopropyl]-L- ornithyl-N-N-dilaureyl-L-glutaminyl]-L-glutamic acid, 3-[N',N"-bis(2-
tertbutyloxycarbonylaminoethyl)guanidino]-N,N-dioctadec-9-enylpropionamide, 3- [N',N"-bis(2-tertbutyloxycarbonylaminoethyl)guanidino]-N,N-dipalmitylpropionamide, 3-[N',N"-bis(2-tertbutyloxycarbonylaminoethyl)guanidino]-N,N-dimyristylpropionamide, 1,4-bis[(3-(3-aminopropyl)-palmitylamino)propyl]piperazine, 1,4-bis[(3-(3-amino-2- hydroxypropyl)-oleylamino)propyl]piperazine, N,N-(2-hydroxy-3-aminopropyl)-N-2- hydroxypropyl-3-N,N-diolylaminopropane, N,N-(2-hydroxy-3-aminopropyl)-N-2- hydroxypropyl-3-N,N-dipalmitylaminopropane, N,N-(2-hydroxy-3-aminopropyl)-N-2- hydroxypropyl-3-N,N-dimyristylaminopropane, 1,4-bis[(3-(3-amino-2-hydoxypropyl)- myristylamino)propyl]piperazine, [(3-aminopropyl)-bis-(2-tetradecyloxyethyl)]methyl ammonium bromide, [(3-aminopropyl)-bis-(2-oleyloxyethyl)]methyl ammonium bromide, [(3-aminopropyl)-bis-(2-palmityloxyethyl)]methyl ammonium bromide, Oleoyl-2-hydroxy-3-N,N-dimethyamino propane, 2-didecanoyl-1-N,N- dimethylaminopropane, palmitoyl-2-hydroxy-3-N,N-dimethyamino propane, 1,2- dipalmitoyl-1-N,N-dimethylaminopropane, myristoyl-2-hydroxy-3-N,N-dimethyamino propane, 1,2-dimyristoyl-1-N,N-dimethylaminopropane, (3-Amino-propyl)-›4-(3-amino- propylamino)-4-tetradecylcarbamoyl-butylcarbamic acid cholesteryl ester, (3-Amino- propyl)-›4-(3-amino-propylamino-4-carbamoylbutylcarbamic acid cholesteryl ester, (3- Amino-propyl)-›4-(3-amino-propylamino)-4-(2-dimethylamino-ethylcarbamoy l)- butylcarbamic acid cholesteryl ester, Spermine-5-carboxyglycine (N'-stearyl-N'-oleyl) amide tetratrifluoroacetic acid salt, Spermine-5-carboxyglycine (N'-stearyl-N'-elaidyl) amide tetratrifluoroacetic acid salt, Agmatinyl carboxycholesterol acetic acid salt, Spermine-5-carboxy-ȕ-alanine cholesteryl ester tetratrifluoroacetic acid salt, 2,6- 'LDPLQRKH[DQRH\O^ ȕ-alanine cholesteryl ester bistrifluoroacetic acid salt, 2,4- 'LDPLQREXW\UR\O^ ȕ-alanine cholesteryl ester bistrifluoroacetic acid salt, N,N-Bis (3- aminopropyl)-3-DPLQRSURSLRQ\O^ ȕ-alanine cholesteryl ester tristrifluoroacetic acid salt., [N,N-Bis(2-hydroxyethyl)-2-aminoethyl]aminocarboxy cholesteryl ester, Stearyl carnitine ester, Palmityl carnitine ester, Myristyl carnitine ester, Stearyl stearoyl carnitine ester chloride salt, L-Stearyl Stearoyl Carnitine Ester, Stearyl oleoyl carnitine ester chloride, Palmityl palmitoyl carnitine ester chloride, Myristyl myristoyl carnitine ester chloride, L-Myristyl myristoyl carnitine ester chloride, 1,4-bis[(3-(3-amino-2-
hydroxypropyl)-palmitylamino)propyl]piperazine, N-(3-aminopropyl)-N,N'-bis- (dodecyloxyethyl)-piperazinium bromide, N-(3-aminopropyl)-N,N'-bis-(oleyloxyethyl)- piperazinium bromide, N-(3-aminopropyl)-N,N'-bis-(palmityloxyethyl)-piperazinium bromide, N-(3-aminopropyl)-N,N'-bis-(myristyloxyethyl)-piperazinium bromide, N-(3- aminopropyl)-N'-methyl-N,N'-(bis-2-dodecyloxyethyl)-piperazinium bromide, N-(3- aminopropyl)-N'-methyl-N,N'-(bis-2-oleyloxyethyl)-piperazinium bromide, N-(3- aminopropyl)-N'-methyl-N,N'-(bis-2-palmityloxyethyl)-piperazinium bromide, N-(3- aminopropyl)-N'-methyl-N,N'-(bis-2-myristyloxyethyl)-piperazinium bromide, 1,4-bis[(3- (3-aminopropyl)-oleylamino)-2-hydroxy-propyl]piperazine, 1,4-bis[(3-(3-aminopropyl)- myristylamino)-2-hydroxy-propyl]piperazine, or 1,4-bis[(3-(3-aminopropyl)- palmitylamino)-2-hydroxy-propyl]piperazine. [0006] When the composition contains a neutral lipid, that lipid may be, for example, DOPE, DPhPE, cholesterol, DOPC, Lyso-PE ( 1-acyl-2-hydroxy-sn-glycero-3- phosphoethanolamine), Lyso-PC ( 1-acyl-3-hydroxy-sn-glycero-3-phosphocholine), or 3- alkyloxy-2-hydroxy-1-acetamidopropane. The composition may contain more than one of these neutral lipids. [0007] Further provided are compositions containing a compound of Formula (I) as described above and a polyamine transfection agent. The polyamine transfection agent may be, for example, selected from the group consisting of dense star dendrimers, PAMAM dendrimers, NH3 core dendrimers, ethylenediamine core dendrimers, dendrimers of generation 5 or higher, dendrimers with substituted groups, dendrimers comprising one or more amino acids, grafted dendrimers, activated dendrimers, polyethylenimine, and polyethylenimine conjugates. [0008] Also provided are compositions containing a compound of Formula (I) as described above and a fusion agent. The fusion agent optionally may comprise a polycationic nucleic acid binding moiety. One or more cationic and/or neutral lipids may be present in these compositions. [0009] Further provided are compositions containing a compound of Formula (I) as described above and a cell surface ligand. The cell surface ligand optionally may
comprise a polycationic nucleic acid binding moiety. These compositions may also contain one or more cationic and/or neutral lipids and/or a fusion agent. [0010] In addition, compositions are provided containing a compound of Formula (I) as described above and a nuclear localization peptide or protein cell surface ligand. These compositions may also contain one or more cationic and/or neutral lipids and/or a fusion agent and/or a cell surface ligand. The cell surface ligand and/or the nuclear localization peptide optionally may comprise a polycationic nucleic acid binding moiety. [0011] The compositions may also contain an amphipathic peptide, which in some embodiments may also function as a fusion agent. Suitable amphipathic peptides include, but are not limited to, peptides comprising a sequence selected from the group consisting of FEAALAEALAEALA (SEQ ID No.: 10), Ac-LARLLPRLLARL-NHCH3 (SEQ ID No.: 11), GLLEELLELLEELWEELLEG (SEQ ID No.: 12), GWEGLIEGIEGGWEGLIEG(SEQ ID No.: 13), GLFEALAEFIEGGWEGLIEG (SEQ ID No.: 14), GLFEALLELLESLWELLLEA (SEQ ID No.: 15), GGYCLEKWMIVASELKCFGNTA (SEQ ID No.: 16), GGYCLTRWMLIEAELKCFGNTAV (SEQ ID No.: 17), and WEAALAEALAEALAEHLAEALAEALEALAA (SEQ ID No.: 18). The amphipathic peptide may optionally be linked to a polycationic nucleic acid binding moiety, for example via a covalent linkage. [0012] Also provided are compositions as described above further comprising a nucleic acid. The nucleic acid may be, for example, an RNA molecule, such as an RNAi molecule. In some embodiments, the RNA molecule is an mRNA. [0013] An aspect of the present application relates to a composition including one or more compounds described herein, and a payload. [0014] Another aspect of the present application relates to a composition including one or more compounds described herein, and one or more of a structural lipid, an ionizable lipid, and a stabilizing agent; and optionally, a payload. [0015] Methods are provided of introducing a nucleic acid into a eukaryotic cell, comprising contacting the cell with a composition as described above, thereby introducing the nucleic acid. The cell may be an animal cell, for example a human cell.
[0016] Also provided are kits containing a compound of Formula (I) and: a neutral lipid, a cationic lipid, a cell surface ligand, a fusion agent and/or a nuclear localization peptide or protein. [0017] Further provided are methods of expressing a protein in a cell, comprising contacting the cell with an expression vector encoding the protein and a compound of Formula (I) as described above or a composition as described above. [0018] Methods are provided for inhibiting expression of a protein in a cell, in which the cell is contacted with an RNAi molecule and a compound of Formula (I) described herein. [0019] In some embodiments at least two neutral lipids are present, which may, for example, be selected from the group consisting of DOPE, DPhPE, cholesterol, DOPC, Lyso-PE ( 1-acyl-2-hydroxy-sn-glycero-3-phosphoethanolamine), Lyso-PC ( 1-acyl-3- hydroxy-sn-glycero-3-phosphocholine), and 3-alkyloxy-2-hydroxy-1-acetamidopropane. [0020] These compositions also may contain one or more polyamine transfection agent, such as dense star dendrimers, PAMAM dendrimers, NH
3 core dendrimers, ethylenediamine core dendrimers, dendrimers of generation 5 or higher, dendrimers with substituted groups, dendrimers comprising one or more amino acids, grafted dendrimers, activated dendrimers, polyethylenimine, and/or polyethylenimine conjugates. [0021] The compositions may contain a fusion agent, a cell surface ligand, a nuclear localization peptide or protein, amphipathic peptide and/or a nuclear localization agent. A nucleic acid also may be present. The fusion agent, cell surface ligand, nuclear localization peptide and/or amphipathic peptide optionally may comprise a polycationic nucleic acid binding moiety. In some embodiments, the nuclear localization agent includes an amphipathic peptide. [0022] Also provided are methods of introducing a nucleic acid into a eukaryotic cell, comprising contacting the cell with a composition as described above, thereby introducing the nucleic acid, into the cell, for example a human cell or an animal cell. The nucleic acid may, for example, be an expression vector.
[0023] Also provided are methods of inhibiting expression of a protein in a cell, comprising contacting the cell with a double stranded RNAi molecule and a compound according to Formula (I), or a composition containing that compound. [0024] Also provided are methods of expressing a protein in a cell, comprising contacting the cell with an expression vector encoding the protein and a compound of Formula (I), or a composition containing that compound. [0025] Also provided is a method of increasing the transfection efficiency of a polycationic lipid containing N amine groups, comprising contacting said cationic lipid with an acylating reagent in an amount sufficient to acylate no more than N-1 of the amine groups. [0026] Also provided are compositions comprising an amphipathic peptide optionally comprising a polycationic nucleic acid binding moiety and a compound of Formula (I). [0027] These compositions also may include one or more reagents such as a cationic lipid, a cell surface ligand, a cell surface ligand comprising a polycationic nucleic acid binding moiety, a fusion agent, a fusion agent comprising a polycationic nucleic acid binding moiety, a nuclear localization peptide or protein, and a nuclear localization peptide or protein comprising a polycationic nucleic acid binding moiety. Advantageously, these compositions contain a compound of Formula (I), an amphipathic peptide optionally comprising a polycationic nucleic acid binding moiety, a cationic lipid and a cell surface ligand comprising a polycationic nucleic acid binding moiety. [0028] Other objects, features and advantages of the present invention will become apparent from the following detailed description. It should be understood, however, that the detailed description and the specific examples, while indicating preferred embodiments of the invention, are given by way of illustration only, since various changes and modifications within the spirit and scope of the invention will become apparent to those skilled in the art from this detailed description. BRIEF DESCRIPTION OF THE DRAWINGS
[0029] Figure 1 is a graph depicting size (d.nm) and polydispersity index of the lipid-mRNA formulations. [0030] Figure 2 is a graph depicting luciferase activity (in bioluminescence flux, photons/second (p/s)) in the lung of mice following intravenous administration of lipid-mRNA formulations. [0031] Figure 3 is a graph depicting luciferase activity (in bioluminescence flux, photons/second (p/s)) in the spleen of mice following intravenous administration of lipid-mRNA formulations. [0032] Figure 4 is a graph depicting the ratio of luciferase activity (in bioluminescence flux, photons/second (p/s)) in the lung by luciferase activity (in bioluminescence flux, photons/second (p/s)) in the spleen of mice following intravenous administration of lipid-mRNA formulations. DETAILED DESCRIPTION [0033] The disclosure relates to ionizable lipids and lipid compositions (e.g.,

and/or pharmaceutical agents, nutrients, a therapeutic, a prophylactic, or the like to a mammalian cell, specifically delivering a therapeutic and/or prophylactic to a mammalian or

in a wide variety of cells, and provide a high efficiency of transfection. Specifically, it has been found that molecules based on a core of N,N'-disubstituted 2,3,-dihydroxy-1,4-
butanediamine are useful for efficient delivery of macromolecules into cells. These molecules advantageously can be used with one or more neutral lipids and additional components such as fusogenic or fusion-enhancing molecules, additional cationic lipids, cell surface ligands, cell adhesion molecules, nuclear localization agents, and endosomal release agents, together with the payload (e.g, macromolecule or pharmaceutical agent, or nutrient, or the like). Such complexes are easily prepared and are stable and therefore are suitable for use in in vitro, ex vivo and in vivo applications, for example, delivery of therapeutic nucleic acids (e.g., siRNA therapeutics, mRNA vaccine preparations, and the like), in cell therapy applications (e.g., delivery of gene editing reagents), delivery of pharmaceutical agents, nutrients and the like to cells, e.g, in for cosmetic, nutraceutical, or therapeutic applications. The complex is easily prepared by straightforward methods and can be used on a wide variety of cells. [0034] Surprisingly, it also has been found that the nucleic acid transfection efficiency of cationic lipids in general, and the compounds described herein in particular, can be dramatically enhanced in many cases by reducing the net positive charge on the lipid by partial acylation of free primary and secondary amine functions on the lipid. Unexpectedly, this reduction in charge has been shown to greatly increase the ability of transfection complexes containing the modified lipid to efficiently transfect cells. Thus, for a lipid with N primary or secondary amines, it is possible to acylate up to N-1 of the amine groups. In most cases, the skilled artisan will recognize that the distribution of acyl groups in a lipid preparation with distinct amino groups will be statistical, because regiospecific acylation likely will not be possible unless the acylation is carried out as part of a more elaborate synthetic scheme. Thus, the distribution of acyl groups will be affected not only by the stoichiometry of the acylation reagent with respect to the lipid, but will also be affected by the reactivity of the amine groups, both initially (in the non-acylated amine) but also during the reaction, as acylation activity at a free amine is potentially affected by acylation at another amine elsewhere in the molecule. [0035] The enhancement of transfection is particularly marked for lipids containing 4 or more reactive amines, in addition to the possible presence of tertiary or quaternary amines but is not necessarily limited to these lipids. This observed result is
surprising in light of the prejudice in the art that a relatively high charge on a cationic lipid is desirable to enhance binding of negatively charged nucleic acids. [0036] Compounds are provided having the structure of Formula (I): O

where each R
1 is optionally substituted C
12-C
20 straight-chain or branched-chain alkyl, optionally substituted C12-C20 monounsaturated or polyunsaturated straight-chain or branched-chain alkenyl, -(CH2)(1-5)-O(CO)-(CH2) (10-18)-CH3, –(CO)C12-C20 alkyl, or –(CO)C
12-C
20 alkenyl; R
2 is optionally substituted C1-C20 straight-chain or branched-chain alkyl or optionally substituted C
2-C
20 monounsaturated straight-chain or branched-chain alkenyl; R
3 is optionally substituted C
1-C
6 straight-chain or branched-chain alkyl, optionally substituted C2-C6 monounsaturated or polyunsaturated straight-chain or branched-chain alkenyl, or -(CH
2)0-3Het or AA; R
4 is H, optionally substituted C
1-C
6 straight-chain or branched-chain alkyl, or optionally substituted C2-C6 monounsaturated or polyunsaturated straight-chain or branched-chain alkenyl;
R
5 is H, an amine protecting group, –(CO)C1-C23 alkyl, –(CO)C2-C23 alkenyl or AA ; substituted C
1-C
20 straight-chain or branched-chain alkyl or
C20 monounsaturated straight-chain or branched-chain alkenyl; AA represents any natural or non-natural amino acid side chain; p is an integer from 1 to 50; and Het is an optionally substituted 5-7 membered monocyclic basic heterocycle, or an optionally substituted 8-11 membered bicyclic basic heterocyclyl. [0037] In some embodiments of compounds of Formula (I), R
1 is selected from optionally substituted C12-C18 straight-chain or branched-chain alkyl, optionally substituted C
12-C
18 monounsaturated or polyunsaturated straight-chain or branched-chain alkenyl, –(CO)C
12-C
18 alkyl, and –(CO)C
12-C
18 alkenyl. [0038] In some embodiments of compound of Formula (I), R
1 may be optionally substituted C
14-C
20 straight-chain or branched-chain alkyl or optionally substituted C
14-C
20- monounsaturated or polyunsaturated straight-chain or branched-chain alkenyl. [0039] In some embodiments, R
1 is an optionally substituted C12-C18 straight- chain or branched-chain alkyl. In some embodiments, R
1 is an optionally substituted C
12 straight-chain or branched-chain alkyl. In some embodiments, R
1 is an optionally substituted C13 straight-chain or branched-chain alkyl. In some embodiments, R
1 is an optionally substituted C
14 straight-chain or branched-chain alkyl. In some embodiments, R
1 is an optionally substituted C
15 straight-chain or branched-chain alkyl. In some embodiments, R
1 is an optionally substituted C16 straight-chain or branched-chain alkyl. In some embodiments, R
1 is an optionally substituted C
17 straight-chain or branched-chain alkyl. In some embodiments, R
1 is an optionally substituted C
18 straight-chain or branched- chain alkyl.
[0040] In some embodiments, R
1 is an optionally substituted C12-C18 monounsaturated or polyunsaturated straight-chain or branched-chain alkenyl. In some embodiments, R
1 is an optionally substituted C12 monounsaturated or polyunsaturated straight-chain or branched-chain alkenyl. In some embodiments, R
1 is an optionally substituted C
13 monounsaturated or polyunsaturated straight-chain or branched-chain alkenyl. In some embodiments, R
1 is an optionally substituted C
14 monounsaturated or polyunsaturated straight-chain or branched-chain alkenyl. In some embodiments, R
1 is an optionally substituted C
15 monounsaturated or polyunsaturated straight-chain or branched- chain alkenyl. In some embodiments, R
1 is an optionally substituted C
16 monounsaturated or polyunsaturated straight-chain or branched-chain alkenyl. In some embodiments, R
1 is an optionally substituted C17 monounsaturated or polyunsaturated straight-chain or branched-chain alkenyl. In some embodiments, R
1 is an optionally substituted C
18 monounsaturated or polyunsaturated straight-chain or branched-chain alkenyl. In some embodiments, the alkenyl group has cis geometry. In some embodiments, the alkenyl group has trans geometry. [0041] In some embodiments, R
1 is a –(CO)C
12-C
18 alkyl. In some embodiments, R
1 is a –(CO)C12 alkyl. In some embodiments, R
1 is a –(CO)C13 alkyl. In some embodiments, R
1 is a –(CO)C
14 alkyl. In some embodiments, R
1 is a –(CO)C
15 alkyl. In some embodiments, R
1 is a –(CO)C
16 alkyl. In some embodiments, R
1 is a –(CO)C
17 alkyl. In some embodiments, R
1 is a –(CO)C18 alkyl. [0042] In some embodiments, R
1 is a –(CO)C12-C18 alkenyl. In some embodiments, R
1 is a –(CO)C
12 alkenyl. In some embodiments, R
1 is a –(CO)C
13 alkenyl. In some embodiments, R
1 is a –(CO)C14 alkenyl. In some embodiments, R
1 is a –(CO)C15 alkenyl. In some embodiments, R
1 is a –(CO)C16 alkenyl. In some embodiments, R
1 is a –(CO)C
17 alkenyl. In some embodiments, R
1 is a –(CO)C
18 alkenyl. In some embodiments, the alkenyl group has cis geometry. In some embodiments, the alkenyl group has trans geometry. [0043] In some embodiments of compounds of Formula (I), R
1 is selected from optionally substituted C
12 straight-chain or branched-chain alkyl, optionally substituted C
16 straight-chain or branched-chain alkyl, optionally substituted C18 straight-chain or
branched-chain alkyl, optionally substituted C12 monounsaturated straight-chain or branched-chain alkenyl, optionally substituted C
16 monounsaturated straight-chain or branched-chain alkenyl, optionally substituted C18 monounsaturated straight-chain or branched-chain alkenyl, –(CO)C12-C18 alkyl, and –(CO)C12-C18 alkenyl. [0044] In some embodiments of compounds of Formula (I), R
1 is selected from –(CO)C
12 alkyl, –(CO)C
15 alkyl, –(CO)C
18 alkyl, monounsaturated –(CO)C
16 alkenyl, and monounsaturated –(CO)C18 alkenyl. [0045] In some embodiments of compounds of Formula (I), R
1 is -(CH
2)
(1-5)-O(CO)-(CH
2)
(10-18)-CH
3. In some embodiments, R
1 is -(CH2)(1-3)-O(CO)-(CH2)(12-16)-CH3. In some embodiments, R
1 is -(CH2)3-O(CO)-(CH2)13-CH3. In some embodiments, R
1 is -(CH2)3-O(CO)-(CH2)14-CH3. In some embodiments, R
1 is -(CH
2)
3-O(CO)-(CH
2)
15-CH
3. In some embodiments, R
1 is -(CH2)3-O(CO)-(CH2)16-CH3. In some embodiments, R
1 is -(CH2)3-O(CO)-(CH2)17-CH3. In some embodiments, R
1 is -(CH2)3-O(CO)-(CH2)18-CH3. [0046] In any of these embodiments, each R
1 may be the same or different at each occurrence. [0047] In some embodiments of compounds of Formula (I), R
2 is C1-C6 alkyl. For example, R
2 may be methyl, ethyl, n-propyl, isopropyl, n-butyl, isobutyl, tert-butyl, pentyl, or hexyl. In some embodiments of Formula (I), R
2 is methyl. In some embodiments, R
2 is ethyl. In some embodiments, R
2 is n-propyl. In some embodiments, R
2 is isopropyl. In some embodiments, R
2 is n-butyl. In some embodiments, R
2 is isobutyl. In some embodiments, R
2 is tert-butyl. In some embodiments, R
2 is pentyl. In some embodiments, R
2 is hexyl. [0048] In some embodiments of compounds of Formula (I), R
2 is C2-C6 alkenyl. In some embodiments, R
2 is ethenyl. In some embodiments, R
2 is propenyl. In some embodiments, R
2 is butenyl. In some embodiments, R
2 is pentenyl. In some embodiments, R
2 is hexenyl. In some embodiments, the alkenyl group has cis geometry. In some embodiments, the alkenyl group has trans geometry. In some embodiments of compounds of Formula (I), R
2 is a neurotransmitter-based functional group selected from,
, ,
For example, R
3 may be methyl, ethyl, n-propyl, isopropyl, n-butyl, isobutyl, tert-butyl, pentyl, or hexyl. In some embodiments of Formula (I), R
3 is methyl. In some embodiments, R
3 is ethyl. In some embodiments, R
3 is n-propyl. In some embodiments, R
3 is isopropyl. In some embodiments, R
3 is n-butyl. In some embodiments, R
3 is isobutyl. In some embodiments, R
3 is tert-butyl. In some embodiments, R
3 is pentyl. In some embodiments, R
3 is hexyl. [0050] In some embodiments of compounds of Formula (I), R
3 is C2-C6 alkenyl. In some embodiments, R
3 is ethenyl. In some embodiments, R
3 is propenyl. In some embodiments, R
3 is butenyl. In some embodiments, R
3 is pentenyl. In some embodiments, R
3 is hexenyl. In some embodiments, the alkenyl group has cis geometry. In some embodiments, the alkenyl group has trans geometry. [0051] In some embodiments of the compounds of Formula (I), R
3 is -(CH2) 0-3-Het. In some embodiments, R
3 is –Het. In some embodiments R
3 is
TP386613WO1 -(CH2) 0-3-Het. In some embodiments, R
3 is -(CH2) 2-Het. In some embodiments, R
3 is -(CH
2)
3-Het. In some substituted 8-11 membered bicyclic basic heterocyclyl. In some
Het is an optionally substituted 5-7 membered monocyclic basic heterocycle. In some embodiments, Het is selected from N N N , , , ,
TP386613WO1 H , N , ,
,
is -(CH2)-Het, and Het is imidazole. [0053] In some embodiments of the compounds of Formula (I), R
4 is H or C
1-C
6 alkyl. For example, R
4 may be hydrogen, methyl, ethyl, n-propyl, isopropyl, n-butyl,
isobutyl, tert-butyl, pentyl, or hexyl. In some embodiments of the compounds of Formula (I), R
4 is hydrogen. In some embodiments of Formula (I), R
4 is methyl. In some embodiments, R
4 is ethyl. In some embodiments, R
4 is n-propyl. In some embodiments, R
4 is isopropyl. In some embodiments, R
4 is n-butyl. In some embodiments, R
4 is isobutyl. In some embodiments, R
4 is tert-butyl. In some embodiments, R
4 is pentyl. In some embodiments, R
4 is hexyl. [0054] In some embodiments of compounds of Formula (I), R
4 is C2-C6 alkenyl. In some embodiments, R
4 is ethenyl. In some embodiments, R
4 is propenyl. In some embodiments, R
4 is butenyl. In some embodiments, R
4 is pentenyl. In some embodiments, R
4 is hexenyl. In some embodiments, the alkenyl group has cis geometry. In some embodiments, the alkenyl group has trans geometry. [0055] In some embodiments of compounds of Formula (I), R
5 is hydrogen. In some embodiments of compounds of Formula (I), R
5 is an amine protecting group. In some embodiments of compounds of Formula (I), R
5 is –(CO)C1-C23 alkyl. In some embodiments of compounds of Formula (I), R
5 is –(CO)C
1-5 alkyl. In some embodiments of compounds of Formula (I), R
5 is –(CO)C
5-10 alkyl. In some embodiments of compounds of Formula (I), R
5 is –(CO)C10-20 alkyl. In some embodiments of compounds of Formula (I), R
5 is –(CO)C
10 alkyl. In some embodiments of compounds of Formula (I), R
5 is –(CO)C
11 alkyl. In some embodiments of compounds of Formula (I), R
5 is –(CO)C
12 alkyl. In some embodiments of compounds of Formula (I), R
5 is –(CO)C13 alkyl. In some embodiments of compounds of Formula (I), R
5 is –(CO)C14 alkyl. In some embodiments of compounds of Formula (I), R
5 is –(CO)C
15 alkyl. In some embodiments of compounds of Formula (I), R
5 is –(CO)C16 alkyl. In some embodiments of compounds of Formula (I), R
5 is –(CO)C17 alkyl. In some embodiments of compounds of Formula (I), R
5 is –(CO)C18 alkyl. In some embodiments of compounds of Formula (I), R
5 is –(CO)C
19 alkyl. In some embodiments of compounds of Formula (I), R
5 is –(CO)C20 alkyl. [0056] In some embodiments of compounds of Formula (I), R
5 is –(CO)C2-C23 alkenyl. In some embodiments of compounds of Formula (I), R
5 is –(CO)C
2-5 alkenyl. In some embodiments of compounds of Formula (I), R
5 is –(CO)C
5-10 alkenyl. In some embodiments of compounds of Formula (I), R
5 is –(CO)C10-20 alkenyl. In some
embodiments of compounds of Formula (I), R
5 is –(CO)C10 alkenyl. In some embodiments of compounds of Formula (I), R
5 is –(CO)C
11 alkenyl. In some embodiments of compounds of Formula (I), R
5 is –(CO)C12 alkenyl. In some embodiments of compounds of Formula (I), R
5 is –(CO)C13 alkenyl. In some embodiments of compounds of Formula (I), R
5 is –(CO)C
14 alkenyl. In some embodiments of compounds of Formula (I), R
5 is –(CO)C
15 alkenyl. In some embodiments of compounds of Formula (I), R
5 is –(CO)C16 alkenyl. In some embodiments of compounds of Formula (I), R
5 is –(CO)C
17 alkenyl. In some embodiments of compounds of Formula (I), R
5 is –(CO)C
18 alkenyl. In some embodiments of compounds of Formula (I), R
5 is –(CO)C19 alkenyl. In some embodiments of compounds of Formula (I), R
5 is –(CO)C20 alkenyl. [0057] In some embodiments of Formula (I), AA is glycine, histidine, serine, tryptophan, arginine, aspartic acid, or pyrrolysine, tyrosine. Preferably, the AA is histidine. [0058] In some embodiments of Formula (I), R
5 is a peptide chain that includes RGD or RYD tripeptide units. [0059] In some embodiments of Formula (I), R
5 is a peptide chain that includes histidine or an RGD or RYD tripeptide unit preceded by spacer such as a glycine or serine spacer (e.g., one or multiple glycine residues, one or more serine residues, or combination of glycine and serine residues). [0060] In some embodiments of Formula (I), R
5 is a peptide chain that includes repeat RGD or RYD tripeptide units preceded by spacer such as a glycine or serine spacer (e.g., one or multiple glycine residues, one or more serine residues, or combination of glycine and serine residues). [0061] In some embodiments of Formula (I), R
5 is a peptide chain that includes repeat GLF or WYG tripeptide units preceded by spacer such as a glycine or serine spacer (e.g., one or multiple glycine residues, one or more serine residues, or combination of glycine and serine residues). [0062] In some embodiments of Formula (I), R
5 is a peptide chain that includes repeat Poly-Arg or Poly-His units preceded by spacer such as a glycine or serine spacer
(e.g., one or multiple glycine residues, one or more serine residues, or combination of glycine and serine residues). [0063] In some embodiments of Formula (I), R
5 is a peptide chain selected from any of the peptide chains listed in Table 1. [0064] In some embodiments of compounds of Formula (I), R
6 is C
1-C
6 alkyl. For example, R
6 may be methyl, ethyl, n-propyl, isopropyl, n-butyl, isobutyl, tert-butyl, pentyl, or hexyl. In some embodiments of Formula (I), R
6 is methyl. In some embodiments, R
6 is ethyl. In some embodiments, R
6 is n-propyl. In some embodiments, R
6 is isopropyl. In some embodiments, R
6 is n-butyl. In some embodiments, R
6 is isobutyl. In some embodiments, R
6 is tert-butyl. In some embodiments, R
6 is pentyl. In some embodiments, R
6 is hexyl. [0065] In some embodiments of compounds of Formula (I), R
6 is C
2-C
6 alkenyl. In some embodiments, R
6 is ethenyl. In some embodiments, R
6 is propenyl. In some embodiments, R
6 is butenyl. In some embodiments, R
6 is pentenyl. In some embodiments, R
6 is hexenyl. In some embodiments, the alkenyl group has cis geometry. In some embodiments, the alkenyl group has trans geometry. In some embodiments of compounds of Formula (I), R
6 is a neurotransmitter-based functional group selected from, but not limited to, ,
HO , of
Formula (Ia) OR
1 O where
11CH3, - (CH2)15CH3, -(CH2)17CH3, -1DC18, -(CO)(CH2)11CH3, -(CO)(CH2)15CH3,
3-O(CO)-(CH
2)
14- CH3, and -(CH2)3-O(CO)-(CH2)16-CH3. [0067] In some embodiments of compounds of formula (Ia), R
1 is -(CH2)11CH3. In some embodiments of compounds of formula (Ia), R
1 is -(CH
2)
15CH
3, -(CH
2)
17CH
3. In some embodiments of compounds of formula (Ia), R
1 is -1DC18. In some embodiments of compounds of formula (Ia), R
1 is -(CO)(CH2)11CH3. In some embodiments of compounds of formula (Ia), R
1 is -(CO)(CH
2)
15CH
3. In some embodiments of compounds of formula (Ia), R
1 is -(CO)(CH2)17CH3. In some embodiments of compounds of formula (Ia), R
1 is -(CO)-1DC18. In some embodiments of compounds of formula (Ia), R
1 is -(CH
2)
3-O(CO)-(CH
2)
12-CH
3. In some embodiments of compounds of formula (Ia), R
1 is
-(CH2)3-O(CO)-(CH2)14-CH3. In some embodiments of compounds of formula (Ia), R
1 is -(CH
2)
3-O(CO)-(CH
2)
16-CH
3. [0068] In some embodiments, the compounds of Formula (I) include: 2-acetamido-N-(2,3-bis(dodecyloxy)-4-(methylamino)butyl)-3-(1H-imidazol- 4-yl)-N-methylpropanamide; 2-acetamido-N-(2,3-bis(hexadecyloxy)-4-(methylamino)butyl)-3-(1H- imidazol-4-yl)-N-methylpropanamide; 2-acetamido-3-(1H-imidazol-4-yl)-N-methyl-N-(4-(methylamino)-2,3- bis(octadecyloxy)butyl)propanamide; 2-acetamido-3-(1H-imidazol-4-yl)-N-methyl-N-(4-(methylamino)-2,3- bis(((E)-octadec-9-en-1-yl)oxy)butyl)propanamide; 1-(2-acetamido-3-(1H-imidazol-4-yl)-N-methylpropanamido)-4- (methylamino)butane-2,3-diyl ditridecanoate; 1-(2-acetamido-3-(1H-imidazol-4-yl)-N-methylpropanamido)-4- (methylamino)butane-2,3-diyl diheptadecanoate; 1-(2-acetamido-3-(1H-imidazol-4-yl)-N-methylpropanamido)-4- (methylamino)butane-2,3-diyl dinonadecanoate; 1-(2-acetamido-3-(1H-imidazol-4-yl)-N-methylpropanamido)-4- (methylamino)butane-2,3-diyl (10E,10'E)-bis(nonadec-10-enoate); ((1-(2-acetamido-3-(1H-imidazol-4-yl)-N-methylpropanamido)-4- (methylamino)butane-2,3-diyl)bis(oxy))bis(propane-3,1-diyl) ditetradecanoate; ((1-(2-acetamido-3-(1H-imidazol-4-yl)-N-methylpropanamido)-4- (methylamino)butane-2,3-diyl)bis(oxy))bis(propane-3,1-diyl) dipalmitate; ((1-(2-acetamido-3-(1H-imidazol-4-yl)-N-methylpropanamido)-4- (methylamino)butane-2,3-diyl)bis(oxy))bis(propane-3,1-diyl) distearate, and stereoisomers thereof. [0069] The skilled artisan will recognize that, although the molecules of the invention are shown here for convenience in their neutral (unprotonated) forms, these molecules will exist in a partially or fully protonated form in solutions of appropriate pH,
and that the present invention encompasses the molecules in all their protonated, unprotonated, ionized and non-ionized forms without limitation, unless specifically indicated otherwise. Definitions: [0070] The term “alkyl” or “alkyl group” refers to a straight or branched hydrocarbon chain that is fully saturated (i.e., contains no double or triple bonds). The alkyl group may have 1 to 20 carbon atoms (whenever it appears herein, a numerical range such as “1 to 20” refers to each integer in the given range; e.g., “1 to 20 carbon atoms” means that the alkyl group may consist of 1 carbon atom, 2 carbon atoms, 3 carbon atoms, etc., up to and including 20 carbon atoms, although the present definition also covers the occurrence of the term “alkyl” where no numerical range is designated) which is optionally substituted. The alkyl group may also be a medium size alkyl having 1 to 9 carbon atoms. The alkyl group could also be a lower alkyl having 1 to 4 carbon atoms. The alkyl group of the compounds may be designated as “C1-4 alkyl”, “C1-20 alkyl”, “C4-30 alkyl” or similar designations. The notation “C
4-30 alkyl” means an optionally substituted linear or branched, saturated hydrocarbon including 4-30 carbon atoms. Unless otherwise specified, an alkyl group described herein refers to both unsubstituted and substituted alkyl groups. For example, the alkyl groups in the straight or branched hydrocarbon chains described above, may be substituted with hydroxyl groups. In another embodiment, the methylene groups are linked via disulfide bridges (—S—S—). Examples of alkyl radicals include, but are not limited to, methyl, ethyl, n-propyl, isopropyl, n-butyl, isobutyl, sec-butyl, tert-butyl, pentyl, isoamyl, n-hexyl, lauryl, palmityl, stearyl, and the like. [0071] The term “alkenyl”, alone or in combination with any other term, refers to a straight-chain or branched-chain monounsaturated or polyunsaturated aliphatic hydrocarbon radical containing the specified number of carbon atoms, or where no number is specified, in one embodiment from 2-30 carbon atoms (i.e. (C2-30)alkenyl), in one embodiment from 4-30 carbon atoms (i.e. (C4-30)alkenyl), and in another embodiment from 2-6 carbon atoms (i.e. (C
2-6)alkenyl). As used herein, the term “alkenyl” or “alkenyl group” means a linear or branched hydrocarbon including two or more carbon atoms (e.g., two, three, four, five, six, seven, eight, nine, ten, eleven, twelve, thirteen, fourteen, fifteen,
sixteen, seventeen, eighteen, nineteen, twenty, or more carbon atoms) and at least one double bond, which is optionally substituted. The notation “C
2-14 alkenyl” means an optionally substituted linear or branched hydrocarbon including 2-14 carbon atoms and at least one carbon-carbon double bond. An alkenyl group may include one, two, three, four, or more carbon-carbon double bonds. For example, Cis alkenyl may include one or more double bonds. A C
5 alkenyl group including two double bonds may be a linoleyl group. Unless otherwise specified, an alkenyl group described herein refers to both unsubstituted and substituted alkenyl groups. For example, the alkenyl groups in the straight or branched hydrocarbon chains described above, may be substituted with hydroxyl groups. In another embodiment, the alkenyl groups are linked via disulfide bridges (—S—S—). Examples of alkenyl radicals include, but are not limited to, ethenyl, E- and Z-propenyl, isopropenyl, E- and Z-butenyl, E- and Z-isobutenyl, E- and Z-pentenyl, E- and Z-hexenyl, E,E-, E,Z-, Z,E- and Z,Z-hexadienyl, oleoyl, palmitoleoyl, and the like. [0072] The term "alkynyl," alone or in combination with any other term, refers to a straight-chain or branched-chain hydrocarbon radical having one or more triple bonds containing the specified number of carbon atoms, or where no number is specified, in one embodiment from 2 to about 10 carbon atoms. Examples of alkynyl radicals include, but are not limited to, ethynyl, propynyl, propargyl, butynyl, pentynyl and the like. [0073] The term "alkoxy" refers to an alkyl ether radical, wherein the term "alkyl" is defined above. Examples of suitable alkyl ether radicals include, but are not limited to, methoxy, ethoxy, n-propoxy, isopropoxy, n-butoxy, isobutoxy, sec-butoxy, tert- butoxy and the like. [0074] The term "aryl," alone or in combination with any other term, refers to a carbocyclic aromatic radical (such as phenyl or naphthyl) containing the specified number of carbon atoms, in one embodiment from 6-15 carbon atoms (i.e. (C
6-15)aryl), and in another embodiment from 6-10 carbon atoms (i.e. (C6-10)aryl), optionally substituted with one or more substituents selected from alkyl, alkoxy, (for example methoxy), nitro, halogen, (for example chloro), amino, carboxylate and hydroxy. Examples of aryl radicals include, but are not limited to phenyl, p-tolyl, 4-hydroxyphenyl, 1-naphthyl, 2-naphthyl, indenyl, indanyl, azulenyl, fluorenyl, anthracenyl and the like.
[0075] The term "aralkyl", alone or in combination, means an alkyl radical as defined above in which one hydrogen atom is phenyl, benzyl, 2-phenylethyl and the like. [0076] The term "aralkoxycarbonyl", alone or in combination, means a radical of the formula -C(O)-O-aralkyl in which the term "aralkyl" has the significance given above. An example of an aralkoxycarbonyl radical is benzyloxycarbonyl. [0077] The term "aryloxy", alone or in combination, means a radical of the formula aryl-O- in which the term "aryl" has the significance given above. [0078] The term "alkanoyl", alone or in combination, means an acyl radical derived from an alkanecarboxylic acid, examples of which include acetyl, propionyl, butyryl, valeryl, 4-methylvaleryl, and the like. [0079] The term "aryloxyalkanoyl" means an acyl radical of the formula aryl- O-alkanoyl wherein aryl and alkanoyl have the significance given above. [0080] The term "aralkanoyl" means an acyl radical derived from an aryl- substituted alkanecarboxylic acid such as phenylacetyl, 3-phenylpropionyl (hydrocinnamoyl), 4-phenylbutyryl, (2-naphthyl)acetyl, 4-chlorohydrocinnamoyl, 4- aminohydrocinnamoyl, 4-phenylbutyryl, (1-naphthyl)acetyl, 4-chlorohydrocinnamoyl, 4- aminohydrocinnamoyl, 4-methoxyhydrocinnamoyl, and the like. [0081] The term "aroyl" means an acyl radical derived from an aromatic carboxylic acid. Examples of such radicals include aromatic carboxylic acids, an optionally substituted benzoic or naphthoic acid such as benzoyl, 4-chlorobenzoyl, 4- carboxybenzoyl, 4-benzyloxycarbonyl)benzoyl, 1-naphthoyl, 2-naphthoyl, 6-carboxy-2- naphthoyl, 6-(benzyloxycarbonyl)-2-naphthoyl, 3-benzyloxy-2-naphthoyl, 3-hydroxy-2- naphthoyl, 3-(benzyloxyformamido)-2-naphthoyl, and the like. [0082] The term "aminocarbonyl" alone or in combination, means an amino- substituted carbonyl (carbamoyl) group derived from an amino-substituted carboxylic acid wherein the amino group can be a primary, secondary or tertiary amino group continuing substituents selected from hydrogen, alkyl, aryl, aralkyl, cycloalkyl, cycloalkylalkyl radicals and the like. [0083] The term "aminoalkanoyl" means an acyl radical derived from an amino substituted alkanecarboxylic acid wherein the amino group can be a primary, secondary or
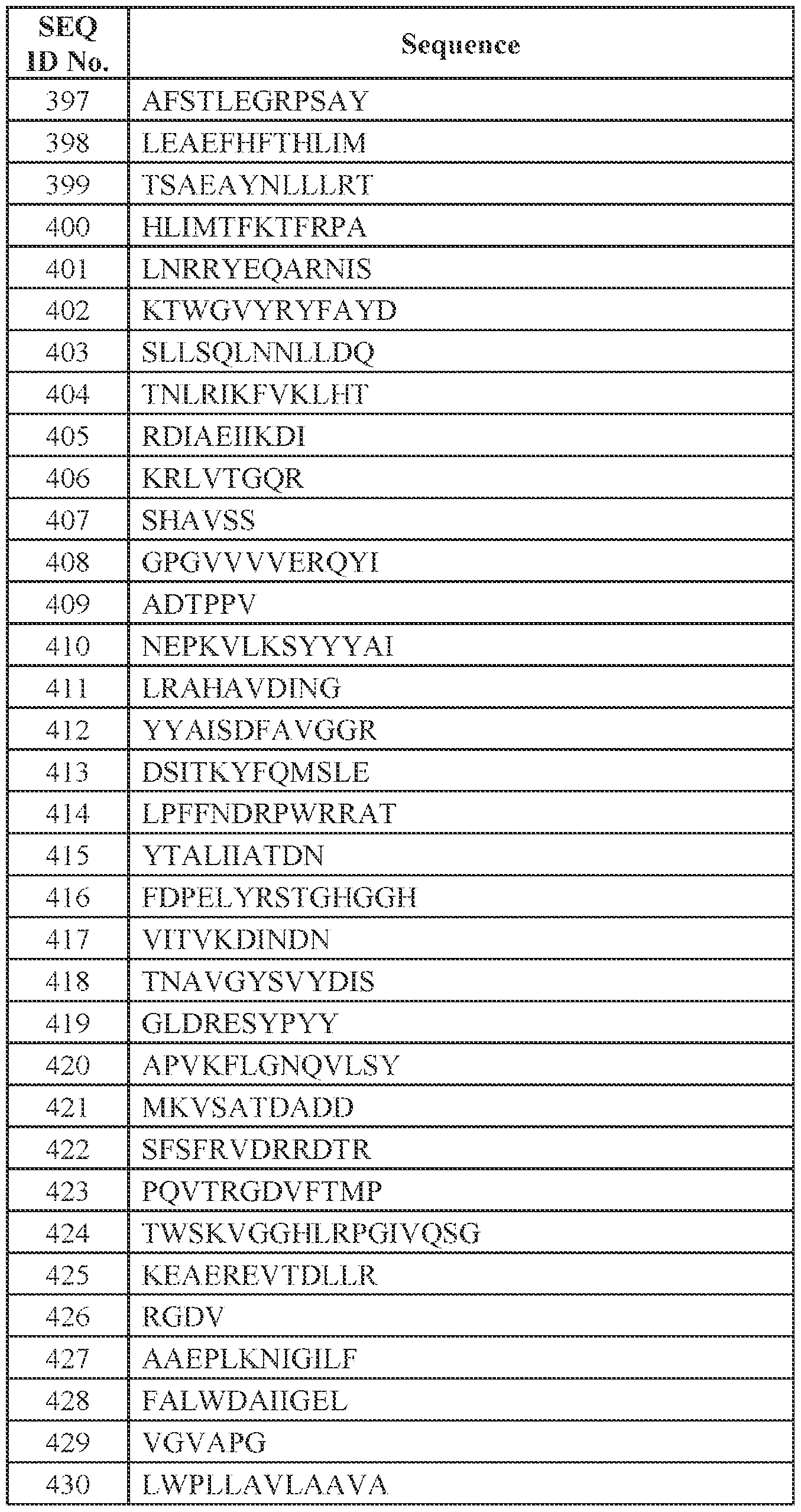
tertiary amino group containing substituents selected from the group consisting of hydrogen, cycloalkyl, cycloalkylalkyl radicals and the like, examples of which include N,N-dimethylaminoacetyl and N-benzylaminoacetyl. [0084] The term "carbocycle" refers to a non-aromatic stable 3- to 8-membered carbon ring which may be saturated, mono-unsaturated or poly-unsaturated. The carbocycle may be attached at any endocyclic carbon atom which results in a stable structure. Carbocycles in one embodiment have 5-7 carbons. [0085] The term "cycloalkyl", alone or in combination, means an alkyl radical which contains from about 3 to about 8 carbon atoms and is cyclic. Examples of such cycloalkyl radicals include cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl and the like. [0086] The term "cycloalkylalkyl" means an alkyl radical as defined above which is substituted by a cycloalkyl radical containing from about 3 to about 8, in one embodiment from about 3 to about 6, carbon atoms. [0087] The term "cycloalkylcarbonyl" means an acyl group derived from a monocyclic or bridged cycloalkanecarboxylic acid such as cyclopropanecarbonyl, cyclohexanecarbonyl, adamantanecarbonyl, and the like, or from a benz-fused monocyclic cycloalkanecarboxylic acid which is optionally substituted by, for example, alkanoylamino, such as 1,2,3,4-tetrahydro-2-naphthoyl, 2-acetamido-1,2,3,4-tetrahydro-2- naphthoyl. [0088] The term "cycloalkylalkoxycarbonyl" means an acyl group derived from a cycloalkylalkoxycarboxylic acid of the formula cycloalkylalkyl-O-COOH wherein cycloalkylalkyl has the significance given above. [0089] The term "basic heterocycle" refers to a stable optionally substituted 5- 7 membered monocyclic heterocyclic ring or optionally substituted 8-11 membered bicyclic heterocyclic ring which is either saturated or partially unsaturated, and which may be optionally benzofused if monocyclic and which is optionally substituted on one or more carbon atoms by halogen, alkyl, alkoxy, oxo, and the like, and/or on a secondary nitrogen atom (i.e., -NH-) by alkyl, aralkoxycarbonyl, alkanoyl, phenyl or phenylalkyl or on a tertiary nitrogen atom (i.e., +N-) by oxido and which is attached via a carbon atom. The term "heterocycle" or "heterocyclo" or "heterocyclyl" refers to a saturated (e.g.,
"heterocycloalkyl"), partially unsaturated (e.g., "heterocycloalkenyl" or "heterocycloalkynyl") or completely unsaturated (e.g., "heteroaryl") ring system where at least one of the ring atoms is a heteroatom (i.e., nitrogen, oxygen or sulfur), with the remaining ring atoms being independently selected from the group consisting of carbon, nitrogen, oxygen and sulfur. A heterocycle may be, without limitation, a single ring, two fused rings, or bridged or spiro rings. A heterocycle group can be linked to the parent molecular moiety via any substitutable carbon or nitrogen atom(s) in the group. Where a heterocycle group is a divalent moiety that links two other elements in a depicted chemical structure, the heterocycle group can be attached to the two other elements through any two substitutable ring atoms. Likewise, where a heterocycle group is a trivalent moiety that links three other elements in a depicted chemical structure, the heterocycle group can be attached to the three other elements through any three substitutable ring atoms, respectively. In the instant compounds, “Het” indicates a heterocycle containing 4-12 carbon atom, where at least one nitrogen atom is present in the ring(s). A heterocyclyl may be, without limitation, a monocycle which contains a single ring. Non-limiting examples of monocycles include furanyl, dihydrofuranyl, tetrahydrofuranyl, pyrrolyl, isopyrrolyl, pyrrolinyl, pyrrolidinyl, imidazolyl, isoimidazolyl, imidazolinyl, imidazolidinyl, pyrazolyl, pyrazolinyl, pyrazolidinyl, triazolyl, tetrazolyl, dithiolyl, oxathiolyl, oxazolyl, isoxazolyl, thiazolyl, isothiazolyl, thiazolinyl, isothiazolinyl, thiazolidinyl, isothiazolidinyl, thiodiazolyl, oxathiazolyl, oxadiazolyl (including 1,2,3-oxadiazolyl, 1,2,4-oxadiazolyl (also known as "azoximyl"), 1,2,5-oxadiazolyl (also known as "furazanyl"), and 1,3,4-oxadiazolyl), oxatriazolyl (including 1,2,3,4-oxatriazolyl and 1,2,3,5-oxatriazolyl), dioxazolyl (including 1,2,3-dioxazolyl, 1,2,4-dioxazolyl, 1,3,2- dioxazolyl, and 1,3,4-dioxazolyl), pyridinyl, piperidinyl, diazinyl (including pyridazinyl (also known as "1,2-diazinyl"), pyrimidinyl (also known as "1,3-diazinyl"), and pyrazinyl (also known as "1,4-diazinyl")), piperazinyl, triazinyl (including s-triazinyl (also known as "1,3,5-triazinyl"), as-triazinyl (also known 1,2,4-triazinyl), and v-triazinyl (also known as "1,2,3-triazinyl), oxazinyl (including 1,2,3-oxazinyl, 1,3,2-oxazinyl, 1,3,6-oxazinyl (also known as "pentoxazolyl"), 1,2,6-oxazinyl, and 1,4-oxazinyl), isoxazinyl (including o- isoxazinyl and p-isoxazinyl), oxazolidinyl, isoxazolidinyl, oxathiazinyl (including 1,2,5-
oxathiazinyl or 1,2,6-oxathiazinyl), oxadiazinyl (including 1,4,2-oxadiazinyl and 1,3,5,2- oxadiazinyl), morpholinyl, azepinyl, and diazepinyl. A heterocyclyl may also be, without limitation, a bicycle containing two fused rings, such as, for example, naphthyridinyl (including [1,8]naphthyridinyl, and [1,6]naphthyridinyl), thiazolpyrimidinyl, thienopyrimidinyl, pyrimidopyrimidinyl, pyridopyrimidinyl, pyrazolopyrimidinyl, indolizinyl, pyrindinyl, pyranopyrrolyl, 4H-quinolizinyl, purinyl, pyridopyridinyl (including pyrido[3,4-b]-pyridinyl, pyrido[3,2-b]-pyridinyl, and pyrido[4,3-b]-pyridinyl), pyridopyrimidine, and pteridinyl. Other non-limiting examples of fused-ring heterocycles include benzo-fused heterocyclyls, such as indolyl, isoindolyl, indoleninyl (also known as "pseudoindolyl"), isoindazolyl (also known as "benzpyrazolyl" or indazolyl), benzazinyl (including quinolinyl (also known as "1-benzazinyl") and isoquinolinyl (also known as "2- benzazinyl")), benzimidazolyl, phthalazinyl, quinoxalinyl, benzodiazinyl (including cinnolinyl (also known as "1,2-benzodiazinyl") and quinazolinyl (also known as "1,3- benzodiazinyl")), benzothiazolyl, 4,5,6,7-tetrahydrobenzo[d]thiazolyl, benzothiadiazolyl, benzimidazolyl, benzotriazolyl, benzoxazinyl (including 1,3,2-benzoxazinyl, 1,4,2- benzoxazinyl, 2,3,1-benzoxazinyl, and 3,1,4-benzoxazinyl), benzisoxazinyl (including 1,2-benzisoxazinyl and 1,4-benzisoxazinyl), and tetrahydroisoquinolinyl. A heterocyclyl may also be, without limitation, a spiro ring system, such as, for example, 1,4-dioxa-8- azaspiro[4.5]decanyl. A heterocyclyl may comprise one or more sulfur atoms as ring members; and in some cases, the sulfur atom(s) is oxidized to SO or SO2. The nitrogen heteroatom(s) in a heterocyclyl may or may not be quaternized and may or may not be oxidized to N-oxide. In addition, the nitrogen heteroatom(s) may or may not be N- protected. A heterocycle or carbocycle may be further substituted. Unless specified, the term "substituted" refers to substitution by independent replacement of one, two, or three or more of the hydrogen atoms with substituents including, but not limited to, -F, -Cl, -Br, -I, hydroxy, protected hydroxy, -NO2, -N3, -CN, -NH2, protected amino, oxo, thioxo, -NH- C2-C8-alkenyl, -NH-C2-C8-alkynyl, -NH-C3-C12-cycloalkyl, -NH-aryl, -NH-heteroaryl, - NH-heterocycloalkyl, -dialkylamino, -diarylamino, -diheteroarylamino, -O-C
1-C
12-alkyl, - O-C
2-C
8-alkenyl, alkynyl, -O-C
3-C
12-cycloalkyl, -O-aryl, -O-heteroaryl, -O- heterocycloalkyl, -C(O)-C1-C12-alkyl, -C(O)-C2-C8-alkenyl, -C(O)-C2-C8-alkynyl, -C(O)-
C3-C12-cycloalkyl, -C(O)-aryl, -C(O)-heteroaryl, -C(O)-heterocycloalkyl, -CONH2, - CONH-C
1-C
12-alkyl, -CONH-C
2-C
8-alkenyl, -CONH-C
2-C
8-alkynyl, -CONH-C
3-C
12- cycloalkyl, -CONH-aryl, -CONH-heteroaryl, -CONH-heterocycloalkyl, -OCO2-C1-C12- alkyl, -OCO2-C2-C8-alkenyl, -OCO2-C2-C8-alkynyl, -OCO2-C3-C12-cycloalkyl, -OCO2- aryl, -OCO
2-heteroaryl, -OCO
2-heterocycloalkyl, -OCONH
2, -OCONH-C
1-C
12-alkyl, - OCONH-C
2-C
8-alkenyl, -OCONH-C
2-C
8-alkynyl, -OCONH-C
3-C
12-cycloalkyl, - OCONH-aryl, -OCONH-heteroaryl, -OCONH-heterocycloalkyl, -NHC(O)-C1-C12-alkyl, - NHC(O)-C
2-C
8-alkenyl, -NHC(O)-C
2-C
8-alkynyl, -NHC(O)-C
3-C
12-cycloalkyl, - NHC(O)-aryl, -NHC(O)-heteroaryl, -NHC(O)-heterocycloalkyl, -NHCO
2-C
1-C
12-alkyl, - NHCO2-C2-C8-alkenyl, -NHCO2-C2-C8-alkynyl, -NHCO2-C3-C12-cycloalkyl, -NHCO2- aryl, -NHCO2-heteroaryl, -NHCO2-heterocycloalkyl, -NHC(O)NH2, -NHC(O)NH-C1-C12- alkyl, -NHC(O)NH-C
2-C
8-alkenyl, -NHC(O)NH-C
2-C
8-alkynyl, -NHC(O)NH-C
3-C
12- cycloalkyl, -NHC(O)NH-aryl, -NHC(O)NH-heteroaryl, -NHC(O)NH-heterocycloalkyl, - NHC(S)NH2, -NHC(S)NH-C1-C12-alkyl, -NHC(S)NH-C2-C8-alkenyl, -NHC(S)NH-C2- C
8-alkynyl, -NHC(S)NH-C
3-C
12-cycloalkyl, -NHC(S)NH-aryl, -NHC(S)NH-heteroaryl, - NHC(S)NH-heterocycloalkyl, -NHC(NH)NH
2, -NHC(NH)NH-C
1-C
12-alkyl, - NHC(NH)NH-C2-C8-alkenyl, NHC(NH)NH-C2-C8-alkynyl, -NHC(NH)NH-C3-C12- cycloalkyl, -NHC(NH)NH-aryl, -NHC(NH)NH-heteroaryl, -NHC(NH)NH- heterocycloalkyl, -NHC(NH)-C
1-C
12-
(NH)-C
2-C
8-alkenyl, -NHC(NH)-C
2-C
8- alkynyl, -NHC(NH)-C3-C12-cycloalkyl, -NHC(NH)-aryl, -NHC(NH)-heteroaryl, - NHC(NH)-heterocycloalkyl, -C(NH)NH-C1-C12-alkyl, -C(NH)NH-C2-C8-alkenyl, - C(NH)NH-C
2-C
8-alkynyl, -C(NH)NH-C
3-C
12-cycloalkyl, -C(NH)NH-aryl, -C(NH)NH- heteroaryl, -C(NH)NH-heterocycloalkyl, -S(O)-C1-C12-alkyl, -S(O)-C2-C8-alkenyl, -S(O)- C2-C8-alkynyl, -S(O)-C3-C12-cycloalkyl, -S(O)-aryl, -S(O)-heteroaryl, -S(O)- heterocycloalkyl, -SO
2NH
2, -SO
2NH-C
1-C
12-alkyl, -SO
2NH-C
2-C
8-alkenyl, -SO
2NH-C
2- C8-alkynyl, -SO2NH-C3-C12-cycloalkyl, -SO2NH-aryl, -SO2NH-heteroaryl, -SO2NH- heterocycloalkyl, -NHSO2-C1-C12-alkyl, -NHSO2-C2-C8-alkenyl, -NHSO2-C2-C8-alkynyl, -NHSO
2-C
3-C
12-cycloalkyl, -NHSO
2-aryl, -NHSO
2-heteroaryl, -NHSO
2-heterocycloalkyl, -CH
2NH
2, -CH
2SO
2CH
3, -aryl, -arylalkyl, -heteroaryl, -heteroarylalkyl, -heterocycloalkyl, -C3-C12-cycloalkyl, polyalkoxyalkyl, polyalkoxy, -methoxymethoxy, -methoxyethoxy, -
SH, -S-C1-C12-alkyl, -S-C2-C8-alkenyl, -S-C2-C8-alkynyl, -S-C3-C12-cycloalkyl, -S-aryl, - heteroaryl, -S-heterocycloalkyl, or methylthiomethyl. It is understood that the aryls, heteroaryls, alkyls, and the like can be further substituted. [0090] The term "halogen" means fluorine, chlorine, bromine or iodine. [0091] The term “ionizable lipid” refers to any of a number of lipid species that carry a net positive charge at a selected pH, such as physiological pH 4 and a neutral charge at other pHs such as physiological pH 7. [0092] The term "N-protecting group" or "N-protected" refers to those groups capable of protecting an amino group against undesirable reactions. Commonly used N- protecting groups are described in Greene and Wuts, Protecting Groups in Chemical Synthesis (3
rd ed., John Wiley & Sons, NY (1999)). Non-limiting examples of N-protecting groups include acyl groups such as formyl, acetyl, propionyl, pivaloyl, t-butylacetyl, 2- chloroacetyl, 2-bromoacetyl, trifluoroacetyl, trichloroacetyl, phthalyl, o- nitrophenoxyacetyl, benzoyl, 4-chlorobenzoyl, 4-bromobenzoyl, or 4-nitrobenzoyl; sulfonyl groups such as benzenesulfonyl or p-toluenesulfonyl; sulfenyl groups such as phenylsulfenyl (phenyl-S-) or triphenylmethylsulfenyl (trityl-S-); sulfinyl groups such as p-methylphenylsulfinyl (p-methylphenyl-S(O)-) or t-butylsulfinyl (t-Bu-S(O)-); carbamate forming groups such as benzyloxycarbonyl, p-chlorobenzyloxycarbonyl, p- methoxybenzyloxycarbonyl, p-nitrobenzyloxycarbonyl, 2-nitrobenzyloxycarbonyl, p- bromobenzyloxycarbonyl, 3,4-dimethoxybenzyloxycarbonyl, 3,5- dimethoxybenzyloxycarbonyl, 2,4-dimethoxybenzyloxycarbonyl, 4- methoxybenzyloxycarbonyl, 2-nitro-4,5-dimethoxybenzyloxycarbonyl, 3,4,5- trimethoxybenzyloxycarbonyl, 1-(p-biphenylyl)-1-methylethoxy carbonyl, dimethyl-3,5- dimethoxybenzyloxycarbonyl, benzhydryloxycarbonyl, t-butyloxycarbonyl, diisopropylmethoxycarbonyl, isopropyloxy carbonyl, ethoxycarbonyl, methoxycarbonyl, allyloxycarbonyl, 2,2,2-trichloro-ethoxy-carbonyl, phenoxy carbonyl, 4-nitro-phenoxy carbonyl, cyclopentyloxycarbonyl, adamantyloxycarbonyl, cyclohexyloxycarbonyl, or phenylthiocarbonyl; alkyl groups such as benzyl, p-methoxybenzyl, triphenylmethyl, or benzyloxymethyl; p-methoxyphenyl; and silyl groups such as trimethylsilyl. Preferred N-
protecting groups include formyl, acetyl, benzoyl, pivaloyl, t-butylacetyl, phenylsulfonyl, benzyl, t-butyloxycarbonyl (Boc) and benzyloxycarbonyl (Cbz). [0093] The term “stabilizing agent” refers to a compound that mitigates the degradation of the lipid nanoparticles or a subpopulation of the lipid nanoparticles. [0094] The term "structural lipid” refers to sterols and lipids containing sterol like moieties. [0095] The term "surface ligand" or "cell surface ligand" refers to a chemical compound or structure which will bind to a surface receptor of a cell. The term "cell surface receptor" as used herein refers to a specific chemical grouping on the surface of a cell to which the ligand can attach. Cell surface receptors can be specific for a particular cell, i.e., found predominantly in one cell rather than in another type of cell (e.g., LDL and asialoglycoprotein receptors are specific for hepatocytes). The receptor facilitates the internalization of the ligand and attached molecules. A cell surface receptor includes but is not limited to a folate receptor, biotin receptor, lipoic acid receptor, low-density lipoprotein receptor, asialoglycoprotein receptor, insulin-like growth factor type II/cation-independent mannose-6-phosphate receptor, calcitonin gene-related peptide receptor, insulin-like growth factor I receptor, nicotinic acetylcholine receptor, hepatocyte growth factor receptor, endothelin receptor, bile acid receptor, bone morphogenetic protein receptor, cartilage induction factor receptor or glycosylphosphatidylinositol (GPI)-anchored proteins (e.g., .beta. adrenergic receptor, T-cell activating protein, Thy-1 protein, GPI- anchored 5' nucleotidase). These are nonlimiting examples. [0096] A receptor is a molecule to which a ligand binds specifically and with relatively high affinity. It is usually a protein or a glycoprotein, but may also be a glycolipid, a lipidpolysaccharide, a glycosaminoglycan or a glycocalyx. For purposes of this disclosure, epitopes to which an antibody or its fragments binds is construed as a receptor since the antigen:antibody complex undergoes endocytosis. Furthermore, surface ligand includes anything which is capable of entering the cell through cytosis (e.g. endocytosis, potocytosis, pinocytosis). [0097] As used herein, the term "ligand" refers to a chemical compound or structure which will bind to a receptor. This includes but is not limited to ligands such as
asialoorosomucoid, asialoglycoprotein, lipoic acid, biotin, apolipoprotein E sequence, insulin-like growth factor II, calcitonin gene-related peptide, thymopoietin, hepatocyte growth factor, endothelin-1, atrial natriuretic factor, RGD-containing cell adhesion peptides and the like. [0098] One skilled in the art will readily recognize that the ligand chosen will depend on which receptor is being bound. Since different types of cells have different receptors, this provides a method of targeting nucleic acid to specific cell types, depending on which cell surface ligand is used. Thus, the preferred cell surface ligand may depend on the targeted cell type. [0099] The term "nuclear localization agent," "nuclear localization signal," or "nuclear ligand" as used herein refers to a ligand, such as a peptide, which will cause an agent covalently or non-covalently linked to it to localize at the cell nucleus, typically by binding a nuclear receptor. The term "nuclear receptor" as used herein refers to a chemical grouping on the nuclear membrane which will bind a specific ligand and help transport the ligand, and accompanying linked moieties, through the nuclear membrane. Nuclear receptors can be but are not limited to those receptors which bind nuclear localization sequences. Nonlimiting examples of nuclear ligands include GYSTPPKKKRKVEDP (SEQ ID No.: 1), GYSTPPKTRRRP (SEQ ID No.: 2), GYSTPGRKKR (SEQ ID No.: 3), GYSTPRRNRRRRW (SEQ ID No.: 4), PDEVKRKKKPPTSYG (SEQ ID No.: 5), PRRRTKPPTSYG (SEQ ID No.: 6), RKKRGPTSYG (SEQ ID No.: 7), WRRRRNRRPTSYG (SEQ ID No.: 8), and GYGPPKKKRKVEAPYKA(K)20-40K (SEQ ID No.: 9), may be used to transport nucleic acid to the nucleus. [0100] The term “polycationic nucleic acid binding moiety” as used herein refers to a moiety containing multiple positive charges at physiological pH that allow the moiety to bind a negatively charged nucleic acid. A polycationic nucleic acid binding moiety may be linked to, for example, a cell surface ligand, a fusion agent, and/or a muclear localization peptide. The linkage may be covalent. Suitable polycationic nucleic acid binding moieties include polyamines and polybasic peptides containing, for example, multiple lysine, ornithine, or histidine residues.
[0101] The term "lysis agent" as used herein refers to a molecule, compound, protein or peptide which is capable of breaking down an endosomal membrane and freeing the DNA transporter into the cytoplasm of the cell. This term includes but is not limited to viruses, synthetic compounds, lytic peptides, or derivatives thereof. The term "lytic peptide" refers to a chemical grouping which penetrates a membrane such that the structural organization and integrity of the membrane is lost. As a result of the presence of the lysis agent, the membrane undergoes lysis, fusion or both. [0102] The term "nucleic acid," when not applied to a specific type of molecule such as unmodified DNA or RNA, refers to any type of nucleic acid that presently is known or that may be prepared or identified in the future, provided that the nucleic acid is sufficiently negatively charged to form a lipid aggregate, liposome, or liposome-like complex when admixed with any lipid of Formula (I) or (Ia). Nucleic acid, as used herein, refers to deoxyribonucleotides or ribonucleotides and mixtures and polymers thereof in single- or double-stranded form. The term encompasses nucleic acids containing known nucleotide analogs or modified backbone residues or linkages, which are synthetic, naturally occurring, and non-naturally occurring, which have similar binding properties as a reference nucleic acid, and which are metabolized in a manner similar to reference nucleotides. Examples of such analogs include, without limitation, phosphorothioates, phosphoramidates, methyl phosphonates, chiral-methyl phosphonates, 2-O-methyl ribonucleotides, peptide-nucleic acids (PNAs), 5 methyl cytosine, pseudouridine, N
1- methyl-pseudouridine, 5 methoxyuridine, and the like. In some embodiments, the payloads described herein can include mRNAs modified with one or more nucleotides selected from the group consisting of: SVHXGRXULGLQH^^DEEUHYLDWHG^E\^WKH^*UHHN^OHWWHU^³SVL´^RU^³^´^^^^- 2-

in the form of an antisense molecule, for example a “gap-mer” containing an RNA-DNA- RNA structure that activates RNAseH. The nucleic acid can be, for example, DNA or RNA, or RNA-DNA hybrid, and can be an oligonucleotide, plasmid, parts of a plasmid
DNA, pre-condensed DNA, product of a polymerase chain reaction (PCR), vectors, expression cassettes, chimeric sequences, chromosomal DNA, or derivatives of these groups or other form of nucleic acid molecule. The nucleic acid may be a double-stranded RNA molecule of the type used for inhibiting gene expression by RNA interference. The nucleic acid may be a short interfering double stranded RNA molecule (siRNA). The nucleic acid molecule can also be a Stealth¥RNAi molecule (Invitrogen Corporation/Life Technologies Corporation, Carlsbad, CA). [0103] The term “amphipathic peptide” refers to a peptide whose secondary structure places hydrophobic and hydrophilic amino acid residues on different faces of the peptide. The peptides often adopt a helical secondary structure. In some circumstances an amphipathic 15 peptide may also function as a fusion agent. Examples of amphipathic peptides suitable for use in the compositions described herein include, but are not limited to, peptides comprising a sequence selected from the group consisting of FEAALAEALAEALA (SEQ ID No.: 42), Ac- LARLLPRLLARL-NHCH3 (SEQ ID No.: 43), GLLEELLELLEELWEELLEG (SEQ ID No.: 44), GWEGLIEGIEGGWEGLIEG(SEQ ID No.: 45), GLFEALAEFIEGGWEGLIEG (SEQ ID No.: 46), GLFEALLELLESLWELLLEA (SEQ ID No.: 47), GGYCLEKWMIVASELKCFGNTA (SEQ ID No.: 48), GGYCLTRWMLIEAELKCFGNTAV (SEQ ID No.: 49), and WEAALAEALAEALAEHLAEALAEALEALAA (SEQ ID No.: 50). The amphipathic peptide may optionally be linked to a polycationic nucleic acid binding moiety, for example via a covalent linkage. Preparation of the lipids: [0104] The unsymmetrical compounds of general structure (I) may be synthesized using methods that are well known in the art, as shown, for example in Scheme 1. Dimethyl tartrate (a-1) can be treated with an alkylamine at elevated temperature (e.g., 70
oC) in a sealed pressure reactor to obtain compound a-2. This compound may be alkylated with an alkyl mesylate to obtain compound a-3, which is then reduced using lithium aluminum hydride to produce the bis-amine a-4. Compound a-4 may be acylated with one equivalent of a suitably protected amino acid a-5 using, for example, a
carbodiimide as a coupling agent to obtain the protected precursor of compound I. This precursor is then deprotected to produce the desired unsymmetric compound I. Specific examples of suitable amino acids for the acylation include Boc-protected carboxyspermine, histidine or lysine. [0105] Alternatively, the unsymmetric compounds of general structure (I) may be prepared using a tartaric acid monoester, readily prepared from diacetyl tartaric anhydride (see Organic Syntheses, Coll. Vol.4, p.242 (1963); Vol.35, p.49 (1955)) or from an erythronolactone such as a-6. See Scheme 2. Protection of the diol, followed by DIBAL reduction, reductive amination and amine protection produce compound a-7. Mild oxidation, reductive amination and protection produce compound a-8. Diol deprotection and alkylation produce compound a-9. Stepwise selective amine deprotection and coupling reactions then produces the unsymmetric compound I. Scheme 1 O O HO CO
2Me
Scheme 2
TP386613WO1 O O a O O HO
b
using methods that are well known in the art, as shown for Compound 1 shown below, in Scheme 3. SCHEME 3
O O HO HO NaH, 15-Crown-5 ether,
with methyl amine at elevated temperature under pressurized conditions to yield the bis-amide 1A. The bis-amide 1A is then treated with oleyl mesylate under basic conditions to yield the bis alkylated derivative 1B which is then subjected to reduction using LAH to yield the bis-amine 1E. Further, as sshown in Scheme 4 below, the derivative 1E is then coupled with the imidazole derivative 1C under basic conditions to obtain the adduct 1D. This was then subjected to BOC-removal using TFA to yield 4-(2-ammonio-3-(methyl(4- (methylammonio)-2,3-bis(((E)-octadec-9-en-1-yl)oxy)butyl)amino)-3-oxopropyl)-1H- imidazol-1-ium (Compound 1) sas a trifluoroacetate salt.. Scheme 4
O NHCH
3 NHCH
3

Formulations: [0108] The lipids described above may be formulated by various methods to be used in transfection. One of the simplest methods for formulation is reverse evaporation, as described in U.S. Pat. No.9,259,475, which is hereby incorporated by reference in its entirety. Other methods for formulation that can be used are sonication and microfluidization. Advantageously, the lipids are formulated as lipid nanoparticles using microfluidic mixing as described, for example, in Roces et al., Pharmaceutics, 12:1095 (2020). Suitable microfluidic mixing devices are commercially available from, for example, Precision Nanosystems (Vancouver, BC). Typically, microfluidic mixing combines two fluid streams, one containing the nucleic acid(s) and one containing the lipid
of Formula (I) or (Ia) and other components, such as the peptide, ligand and other lipid components as described below. [0109] For lipid nanoparticle compositions including an RNA, solutions of the RNA at concentrations of 0.1 mg/ml in deionized water are diluted in 50 mM sodium citrate buffer at a pH between 3 and 4 to form a stock solution. Nanoparticle compositions can be processed by dialysis to remove ethanol and achieve buffer exchange. Formulations may be dialyzed against phosphate buffered saline (PBS), pH 7.4, using a desired molecular weight cutoff, e.g.10 kD. The resulting nanoparticle suspension may be filtered through a ^^^^^P^VWHULOH^ILOWHUV^^6DUVWHGW^^1XPEUHFKW^^*HUPDQ\^^LQWR^JODVV^YLDOV^DQG^VHDOHG^ [0110] Methods of determining particle size in nanoparticles formulations are well-known in the art. For example, a Zetasizer Nano ZS (Malvern Instruments Ltd, Malvern, UK) can be used to determine the particle size, the polydispersity index (PDI) and the zeta potential of the nanoparticle compositions. UV-visible spectroscopy can be used to determine the concentration of payload such as nucleic acid (e.g., mRNA) in nanoparticle compositions. A quantity of the composition is diluted in a suitable solvent and the absorbance spectrum of the solution is recorded, for example, between 230 nm and 330 nm on a spectrophotometer. The concentration of therapeutic and/or prophylactic in the nanoparticle composition can be calculated based on the extinction coefficient of the therapeutic and/or prophylactic used in the composition and on the difference between the absorbance at a wavelength of, for example, 260 nm and the baseline value at a wavelength of, for example, 330 nm. [0111] For nanoparticle compositions including an RNA, a QUANT-IT™ RIBOGREEN® RNA assay (Invitrogen Corporation, Carlsbad, CA) can be used to evaluate the encapsulation of an RNA by the nanoparticle composition using methods provided by the manufacturer. The fluorescence intensity generated after addition of the RIBOGREEN reagent can be measured using a fluorescence plate reader at an excitation wavelength of, for example, about 480 nm and an emission wavelength of, for example, about 520 nm. The fluorescence values of the reagent blank are subtracted from that of each of the samples and the percentage of free RNA is determined by dividing the
fluorescence intensity of the intact sample (without addition of Triton X-100) by the fluorescence value of the disrupted sample (caused by the addition of Triton X-100). [0112] An aspect of the present application relates to a composition including one or more compounds described herein, and a payload. [0113] Another aspect of the present application relates to a composition including one or more compounds described herein, and one or more of a structural lipid, an ionizable lipid, and a stabilizing agent; and optionally, a payload. [0114] In some embodiments, the compositions include a compound described herein; one or more structural lipids; one or more stabilizing agents; and optionally, a payload. [0115] In some embodiments, the compositions include a compound described herein; one or more structural lipids; one or more stabilizing agents; one or more transfection enhancing agents; and optionally, a payload. [0116] In some embodiments, the compositions include 10 to 80 mol% of compounds described herein, excluding any payload, if present. Co-Lipids: [0117] Advantageously, the new lipids are formulated with one or more co- lipids, most advantageously neutral co-lipids, although the skilled artisan will recognize that other lipids, including cationic lipids, may be used. [0118] The new lipids may be formulated with one or more cationic lipids and/or one or more neutral lipids. The neutral lipid(s) may be, for example, DOPE, DPhPE, cholesterol, DOPC, Lyso-PE (1-acyl-2-hydroxy-sn-glycero-3-phosphoethanolamine), Lyso-PC (1-acyl-3-hydroxy-sn-glycero-3-phosphocholine), and/or 3-alkyloxy-2-hydroxy- 1-acetamidopropane. [0119] The cationic lipid may be selected from the group consisting of DOTMA, DOTAP, DMRIE, DC-Chol, DDAB, DOSPA, DOSPER, DOGS, TMTPS, TMTOS, TMTLS, TMTMS, TMDOS, N-1-dimethyl-N-1-(2,3-diaoleoyloxypropyl)-2- hydroxypropane-1,3-diamine, N-1-dimethyl-N-1-(2,3-diamyristyloxypropyl)-2- hydroxypropane-1,3-diamine, N-1-dimethyl-N-1-(2,3-diapalmityloxypropyl)-2- hydroxypropane-1,3-diamine, N-1-dimethyl-N-1-(2,3-diaoleoyloxypropyl)-2-(3-amino-2-
hydroxypropyloxy)propane-1,3-diamine, N-1-dimethyl-N-1-(2,3-diamyristyloxypropyl)- 2-(3-amino-2-hydroxypropyloxy)propane-1,3-diamine, N-1-dimethyl-N-1-(2,3- diapalmityloxypropyl)-2-(3-amino-2-hydroxypropyloxy)propane-1,3-diamine, L- spermine-5-carboxyl-3-(DL-1,2-dipalmitoyl-dimethylaminopropyl-ȕ-

3,5-(N,N-di-lysyl)-diaminobenzoyl-glycyl-3-(DL-1,2-dipalmitoyl-dimethylami ȕ-hydroxyethylamine), L-Lysine-bis(O,O'-oleoyl-ȕ-hydroxyethyl)amide dihydrochloride, L-Lysine-bis-(O,O'-palmitoyl-ȕ-hydroxyethyl)amide dihydrochloride, 1,4-bis[(3-(3- aminopropyl)-alkylamino)-2-hydroxypropyl)piperazine, L-Lysine-bis-(O,O'-myristoyl-ȕ- hydroxyethyl)amide dihydrochloride, L-Ornithine-bis-(O,O'-myristoyl-ȕ- hydroxyethyl)amide dihydrochloride, L-Ornithine-bis-(O,O'-oleoyl-ȕ- hydroxyethyl)amide dihydrochloride, 1,4-bis[(3-(3-aminopropyl)-oleylamino)-2- hydroxypropyl]piperazine, L-Ornithine-bis-(O,O'-palmitoyl-ȕ-hydroxyethyl)amide dihydrochloride, 1,4,-bis[(3-amino-2-hydroxypropyl)-oleylamino]-butane-2,3-diol, 1,4,- bis[(3-amino-2-hydroxypropyl)-palmitylamino]-butane-2,3-diol, 1,4,-bis[(3-amino-2- hydroxypropyl)-myristylamino]-butane-2,3-diol, 1,4-bis[(3- oleylamino)propyl]piperazine, L-Arginine-bis-(O,O'-oleoyl-ȕ-hydroxyethyl)amide dihydrochloride, bis[(3-(3-aminopropyl)-myristylamino)2-hydroxypropyl]piperazine, L- Arginine-bis-(O,O'-palmitoyl-ȕ-hydroxyethyl)amide dihydrochloride, L-Serine-bis-(O,O'- oleoyl-ȕ-hydroxyethyl)amide dihydrochloride, 1,4-bis[(3-(3-aminopropyl)- palmitylamino)-2-hydroxypropyl]piperazine, Glycine-bis-(O,O'-palmitoyl-ȕ- hydroxyethyl)amide dihydrochloride, Sarcosine-bis-(O,O'-palmitoyl-ȕ- hydroxyethyl)amide dihydrochloride, L-Histidine-bis-(O,O'-palmitoyl-ȕ- hydroxyethyl)amide dihydrochloride, cholesteryl-^ȕ-carboxyl- amidoethylenetrimethylammonium iodide, 1,4-bis[(3-myristylamino)propyl]piperazine, 1-dimethylamino-3-trimethylammonio-DL-2-propyl-cholesteryl carboxylate iodide, cholesteryl-^ȕ-carboxyamidoethyleneamine, cholesteryl-^ȕ- oxysuccinamidoethylenetrimethylammonium iodide, 1-dimethylamino-3- trimethylammonio-DL-2-propyl-cholesteryl-^ȕ-oxysuccinate iodide, 2-[(2- trimethylammonio)-ethylmethylamino] ethyl-cholesteryl-^ȕ-R[\VXFFLQDWH^ LRGLGH^^ ^ȕ>1- (N', N'-GLPHWK\ODPLQRHWKDQH^FDUEDPR\O@FKROHVWHURO^^ DQG^ ^ȕ-[N-(polyethyleneimine)-
carbamoyl] cholesterol,1,4-bis[(3-palmitylamino)propyl]piperazine, L-Ornithylglycyl-N- (1-heptadecyloctadecyl)glycinamide, N
2,N
5 -Bis(3-aminopropyl)-L-ornithylglycyl-N- (1- heptadecyloctadecyl)glycinamide, 1,4-bis[(3-(3-amino-2-hydroxypropyl)-alkylamino)-2- hydroxypropyl]piperazine, N
2-[N
2,N
5 -Bis(3-aminopropyl)-L-ornithyl]-N,N-dioctadecyl- L-glutamine, N
2-[N
2,N
5 -Bis(aminopropyl)-L-ornithyl]-N-N-dioctadecyl-L-Į-glutamine, 1,4-bis[(3-(3-amino-2-hydroxypropyl)-oleylamino)2-hydroxypropyl]piperazine, N
2- [N
2,N
5 -Bis(aminopropyl)-L-ornithyl]-N-N-dioctadecyl-L-Į-asparagine, N-[N
2-[N
2,N
5- Bis[(1,1-dimethylethoxy)carbonyl]- N
2,N
5-bis[3-[(1,1- dimethylethoxy)carbonyl]aminopropyl]-L-ornithyl-N-N-dioctadecyl-L-glutaminyl]-L- glutamic acid, N
2-[N
2,N
5 -Bis(3-aminopropyl)-L-ornithyl]-N,N-diolyl-L-glutamine, N
2- [N
2,N
5 -Bis(aminopropyl)-L-ornithyl]-N-N-dioleyl-L-Į-glutamine,4-bis[(3-(3-amino-2- hydoxypropyl)-myristylamino)-2-hydroxypropyl]piperazine, N
2-[N
2,N
5 - Bis(aminopropyl)-L-ornithyl]-N-N-dioleyl-L-Į-asparagine, N-[N
2-[N
2,N
5-Bis[(1,1- dimethylethoxy)carbonyl]- N
2,N
5-bis[3-[(1,1-dimethylethoxy)carbonyl]aminopropyl]-L- ornithyl-N-N-dioleyl-L-glutaminyl]-L-glutamic acid, 1,4-bis[(3-(3-aminopropyl)- oleylamino)propyl]piperazine, N
2-[N
2,N
5 -Bis(3-aminopropyl)-L-ornithyl]-N,N- dipalmityl-L-glutamine,N
2-[N
2,N
5 -Bis(aminopropyl)-L-ornithyl]-N-N-dipalmityl-L-Į- glutamine, N
2-[N
2,N
5 -Bis(aminopropyl)-L-ornithyl]-N-N-dipalmityl-L-Į-asparagine, N- [N
2-[N
2,N
5-Bis[(1,1-dimethylethoxy)carbonyl]- N
2,N
5-bis[3-[(1,1- dimethylethoxy)carbonyl]aminopropyl]-L-ornithyl-N-N-dipalmityl-L-glutaminyl]-L- glutamic acid, N
2-[N
2,N
5 -Bis(3-aminopropyl)-L-ornithyl]-N,N-dimyristyl-L- glutamine, N
2-[N
2,N
5 -Bis(aminopropyl)-L-ornithyl]-N-N-dimyristyl-L-Į-glutamine, N
2- [N
2,N
5 -Bis(aminopropyl)-L-ornithyl]-N-N-dimyristyl-L-Į-asparagine, 1,4-bis[(3-(3- amino-2-hydroxypropyl)-palmitylamino)-2-hydroxypropyl]piperazine, N-[N
2-[N
2,N
5- Bis[(1,1-dimethylethoxy)carbonyl]- N
2,N
5-bis[3-[(1,1- dimethylethoxy)carbonyl]aminopropyl]-L-ornithyl-N-N-dimyristyl-L-glutaminyl]-L- glutamic acid, 1,4-bis[(3-(3-aminopropyl)-myristylamino)propyl]piperazine, N
2-[N
2,N
5 - Bis(3-aminopropyl)-L-ornithyl]-N,N-dilaureyl-L-glutamine, N
2-[N
2,N
5 - Bis(aminopropyl)-L-ornithyl]-N-N-dilaureyl-L-Į-glutamine, - Bis(aminopropyl)-L-ornithyl]-N-N-dilaureyl-L-Į-asparagine, Bis[(1,1-
dimethylethoxy)carbonyl]- N
2,N
5-bis[3-[(1,1-dimethylethoxy)carbonyl]aminopropyl]-L- ornithyl-N-N-dilaureyl-L-glutaminyl]-L-glutamic acid, 3-[N',N"-bis(2- tertbutyloxycarbonylaminoethyl)guanidino]-N,N-dioctadec-9-enylpropionamide, 3- [N',N"-bis(2-tertbutyloxycarbonylaminoethyl)guanidino]-N,N-dipalmitylpropionamide, 3-[N',N"-bis(2-tertbutyloxycarbonylaminoethyl)guanidino]-N,N-dimyristylpropionamide, 1,4-bis[(3-(3-aminopropyl)-palmitylamino)propyl]piperazine, 1,4-bis[(3-(3-amino-2- hydroxypropyl)-oleylamino)propyl]piperazine, N,N-(2-hydroxy-3-aminopropyl)-N-2- hydroxypropyl-3-N,N-diolylaminopropane, N,N-(2-hydroxy-3-aminopropyl)-N-2- hydroxypropyl-3-N,N-dipalmitylaminopropane, N,N-(2-hydroxy-3-aminopropyl)-N-2- hydroxypropyl-3-N,N-dimyristylaminopropane, 1,4-bis[(3-(3-amino-2-hydoxypropyl)- myristylamino)propyl]piperazine, [(3-aminopropyl)-bis-(2-tetradecyloxyethyl)]methyl ammonium bromide, [(3-aminopropyl)-bis-(2-oleyloxyethyl)]methyl ammonium bromide, [(3-aminopropyl)-bis-(2-palmityloxyethyl)]methyl ammonium bromide, Oleoyl-2-hydroxy-3-N,N-dimethyamino propane, 2-didecanoyl-1-N,N- dimethylaminopropane, palmitoyl-2-hydroxy-3-N,N-dimethyamino propane, 1,2- dipalmitoyl-1-N,N-dimethylaminopropane, myristoyl-2-hydroxy-3-N,N-dimethyamino propane, 1,2-dimyristoyl-1-N,N-dimethylaminopropane, (3-Amino-propyl)-›4-(3-amino- propylamino)-4-tetradecylcarbamoyl-butylcarbamic acid cholesteryl ester, (3-Amino- propyl)-›4-(3-amino-propylamino-4-carbamoylbutylcarbamic acid cholesteryl ester, (3- Amino-propyl)-›4-(3-amino-propylamino)-4-(2-dimethylamino-ethylcarbamoy l)- butylcarbamic acid cholesteryl ester, Spermine-5-carboxyglycine (N'-stearyl-N'-oleyl) amide tetratrifluoroacetic acid salt, Spermine-5-carboxyglycine (N'-stearyl-N'-elaidyl) amide tetratrifluoroacetic acid salt, Agmatinyl carboxycholesterol acetic acid salt, Spermine-5-carboxy-ȕ-alanine cholesteryl ester tetratrifluoroacetic acid salt, 2,6- 'LDPLQRKH[DQRH\O^ ȕ-alanine cholesteryl ester bistrifluoroacetic acid salt, 2,4- 'LDPLQREXW\UR\O^ ȕ-alanine cholesteryl ester bistrifluoroacetic acid salt, N,N-Bis (3- aminopropyl)-3-DPLQRSURSLRQ\O^ ȕ-alanine cholesteryl ester tristrifluoroacetic acid salt., [N,N-Bis(2-hydroxyethyl)-2-aminoethyl]aminocarboxy cholesteryl ester, Stearyl carnitine ester, Palmityl carnitine ester, Myristyl carnitine ester, Stearyl stearoyl carnitine ester chloride salt, L-Stearyl Stearoyl Carnitine Ester, Stearyl oleoyl carnitine ester
chloride, Palmityl palmitoyl carnitine ester chloride, Myristyl myristoyl carnitine ester chloride, L-Myristyl myristoyl carnitine ester chloride, 1,4-bis[(3-(3-amino-2- hydroxypropyl)-palmitylamino)propyl]piperazine, N-(3-aminopropyl)-N,N'-bis- (dodecyloxyethyl)-piperazinium bromide, N-(3-aminopropyl)-N,N'-bis-(oleyloxyethyl)- piperazinium bromide, N-(3-aminopropyl)-N,N'-bis-(palmityloxyethyl)-piperazinium bromide, N-(3-aminopropyl)-N,N'-bis-(myristyloxyethyl)-piperazinium bromide, N-(3- aminopropyl)-N'-methyl-N,N'-(bis-2-dodecyloxyethyl)-piperazinium bromide, N-(3- aminopropyl)-N'-methyl-N,N'-(bis-2-oleyloxyethyl)-piperazinium bromide, N-(3- aminopropyl)-N'-methyl-N,N'-(bis-2-palmityloxyethyl)-piperazinium bromide, N-(3- aminopropyl)-N'-methyl-N,N'-(bis-2-myristyloxyethyl)-piperazinium bromide, 1,4-bis[(3- (3-aminopropyl)-oleylamino)-2-hydroxy-propyl]piperazine, 1,4-bis[(3-(3-aminopropyl)- myristylamino)-2-hydroxy-propyl]piperazine, and 1,4-bis[(3-(3-aminopropyl)- palmitylamino)-2-hydroxy-propyl]piperazine. Polyamine Components: [0120] Other formulations may also include one or more polyamine transfection agents, such as dense star dendrimers, PAMAM dendrimers, NH
3 core dendrimers, ethylenediamine core dendrimers, dendrimers of generation 5 or higher, dendrimers with substituted groups, dendrimers comprising one or more amino acids, grafted dendrimers, activated dendrimers, polyethylenimine, and/or polyethylenimine conjugates. Transfection enhancing agents: [0121] The compositions described herein may include one or more transfection enhancing agents (e.g., a polycationic nucleic acid binding moiety). In some embodiments, the transfection enhancing agent selected from an endosomal release agent, a cell surface ligand, a nuclear localization agent, a cell-penetrating peptide, a fusogenic peptide, and any combination thereof. In some embodiments, the one or more transfection enhancing agents includes an amphipathic peptide. [0122] The compositions provided herein can advantageously include, e.g., one or more peptide sequences that enhance transfection efficiency, such as linkers, spacers,
or nuclear targeting sequences. The peptides provided herein can be included in the compositons herein independently (i.e., not covalently attached to another molecule) or alternatively, can be covalently linked to one or more molecules of the compositions provided herein (e.g., covalently linked to an ionizable or other lipid as described herein, covalently linked to another transfection enhancer as described herein, or covalently linked to a payload as described herein, or the like). In some embodiments, the covalent linkage can be via a spacer. The term "spacer," or "linker," which are used interchangeably herein, as used herein refers to a chemical structure that links two molecules to each other. In some embodiments, the spacer binds each molecule on a different part of the spacer molecule. In other embodiments, the spacer is a hydrophilic moiety and comprises about 6 to 30 carbon atoms. In other embodiments, the spacer comprises a ployether, for example -CH2-0-(CH2- CH
2- 0-)iCH
2-. In other embodiments, the spacer comprises a hydrophilic polymer, for example [(gly)i(ser)j]k (SEQ ID NO: 755). In these formulae i ranges from 1 to 6, j ranges from 1 to 6, and k ranges from 3 to 20. In some embodiments, the spacer is a peptide of sequence APYKAWK (SEQ ID NO:756). In other embodiments, the spacer is a sequence that is degraded in vivo by a peptidase. [0123] Still other formulations may include transfection enhancing agents such as a fusion agent, a cell surface ligand and/or a nuclear localization agent such as a nuclear receptor ligand peptide, Examples of transfection enhancing agents include, but are not limited to, reovirus-related fusogenic peptides (see WO07/130073, which is hereby incorporated by reference in its entirety), insulin, a transferrin, epidermal growth factor, fibroblast growth factor, a cell targeting antibody, a lactoferrin, a fibronectin, an adenovirus penton base, Knob, a hexon protein, a vesicular stomatitis virus glycoprotein, a Semliki Forest Virus core protein, a influenza hemagglutinin, a hepatitis B core protein, an HIV Tat protein, a herpes simplex virus VP22 protein, a histone protein, a arginine rich cell permeability protein, a high mobility group protein, and invasin protein, and internalin protein, an endotoxin, a diphtheria toxin, a shigella toxin, a melittin, a magainin, a gramicidin, a cecrophin, a defensin, a protegrin, a tachyplesin, a thionin, a indolicidin, a bactenecin, a drosomycin, an apidaecin, a cathelicidin, a bactericidal-permeability- increasing protein, a nisin, a buforin, and fragments thereof. In some embodiments, the
compositions include a cell-surface ligand (e.g., a RGD-containing peptide). Other cell penetrating peptides useful in the compositions provided herein include those provided in Table 1 below: Table 1 SEQ ID No. Sequence

SEQ ID No. Sequence H
SEQ ID No. Sequence
SEQ ID No. Sequence K
TP386613WO1 SEQ ID No. Sequence
SEQ ID No. Sequence
SEQ ID No. Sequence
TP386613WO1 SEQ ID No. Sequence
SEQ ID No. Sequence
SEQ ID No. Sequence
SEQ ID No. Sequence
SEQ ID No. Sequence
SEQ ID No. Sequence
SEQ ID No. Sequence
SEQ ID No. Sequence
SEQ ID No. Sequence P G P
TP386613WO1 SEQ ID No. Sequence G P K S I I T S W L E S
SEQ ID No. Sequence
TP386613WO1 SEQ ID No. Sequence L R
TP386613WO1 SEQ ID No. Sequence e
SEQ ID No. Sequence
SEQ ID No. Sequence
TP386613WO1 SEQ ID No. Sequence
SEQ ID No. Sequence
SEQ ID No. Sequence

Payload: [0124] An aspect of the present application relates to a composition including one or more compounds described herein, and a payload. [0125] According to the present disclosure, compositions as described herein can further comprise one or more bioactive molecules to be delivered to a cell (i.e., a payload). For example, bioactive molecules can include, e.g., nucleic acids, peptides, active pharmaceutical agents, nutrients, small molecules, or the like. Preferably, the payloads in the embodiments provided herein are biomolecules, e.g., either nucleic acids (RNA, DNA, etc., as described in more detail herein), peptides, or a combination thereof. In some embodiments, the payload can be a therapeutic and/or prophylactic agent. Alternatively, the payload can be used for cosmetic, or nutraceutical applications. The therapeutic and/or prophylactic agents are sometimes referred to as a “therapeutic payload” or “payload” in the present disclosure. In some embodiments, the payload can be administered in vivo or in vitro using the compositions provided herein as a delivery vehicle. In some embodiments, the compositions include lipid molecules functionalized with neurotransmitter-based functional groups allowing for the delivery of the payload to the brain via the blood-brain barrier (BBB). [0126] In some embodiments, the payload is a nucleic acid. In some embodiments, the compositions include a compound described herein with a charge N and a nucleic acid molecule with a charge P, where the combination of the compound and the nucleic acid contacting the cell comprises an N/P ratio from about 1 to 20. [0127] In some embodiments, the nucleic acid is an RNA. In some embodiments, the RNA is mRNA, siRNA, shRNA, self-replicating RNA (srRNA), an o- RNA, self-amplifying RNA, stRNA, trRNA, crRNA, sgRNA, RNAi molecule, an
asymmetrical interfering RNA (aiRNA), a microRNA (miRNA), a Dicer-substrate RNA (dsRNA), a small hairpin RNA (shRNA), or any combination thereof. In some embodiments, the RNA is an mRNA. The compositions can include two or more different mRNAs. [0128] In some embodiments, the RNA encodes an immunogen. In some embodiments, the RNA encodes a cancer antigen [0129] In some embodiments, the nucleic acid is a DNA. [0130] In some embodiments, the payload includes one or more peptides, and optionally a nucleic acid. In some embodiments, the peptide is covalently linked to a nucleic acid. [0131] In certain embodiments, a lower N/P ratio is preferred. Provided herein, are lipid compositions comprising at least one ionizable lipid having a charge (N), at least one peptide, and a nucleic acid molecule comprising a charge (P), wherein the composition comprises an N/P ratio of 0.01, or of 0.02, or of 0.04, or of 0.06, or of 0.08, or of 0.10, or of 0.12, or of 0.14, or of 0.16, or of 0.18, or of 0.20. In other examples, the N/P ratio is from 0.01 to 0.10. In other examples, the N/P ratio is from 0.01 to 0.20. In other examples, the N/P ratio is from 0.01 to 0.25. In other examples, the N/P ratio is from 0.01 to 0.33. In other examples, the N/P ratio is from 0.01 to 0.5. In other examples, the N/P ratio is from 0.01 to 1. In other examples, the N/P ratio is from 0.05 to 0.1. In other examples, the N/P ratio is from 0.05 to 0.125. In other examples, the N/P ratio is from 0.5 to 0.15. In other examples, the N/P ratio is from 0.05 to 0.167. In other examples, the N/P ratio is from 0.05 to 0.20. In other examples, the N/P ratio is from 0.05 to 0.25. In other examples, the N/P ratio is from 0.05 to 0.33. In other examples, the N/P ratio is from 0.05 to 0.5. In other examples, the N/P ratio is from 0.05 to 1.0. In some embodiments, the N/P ratio may be less than about 0.1. In some embodiments the N/P ratio is 0.1. In some embodiments, the N/P ratio is 0.025. In other embodiments, the N/P ratio is 0.01. In some embodiments the N/P ratio is 0.005. [0132] In certain embodiments, the one or more nucleic acids (e.g. mRNAs, siRNAs, sgRNAs), lipids, and amounts thereof may be selected to provide an N/P ratio from about 2.0 to about 8.0, such as 2, 3, 4, 5, 6, 7, and 8. In certain embodiments, the N/P
ratio may be from about 2.0 to about 5.0. In some embodiments, the N/P ratio may be about 4.0. In other embodiments, the N/P ratio is from about 5 to about 8. For example, the N/P ratio may be about 5.0, about 5.5, about 5.67, about 6.0, about 6.5, or about 7.0. [0133] In other embodiments, the one or more nucleic acids, lipids, and amounts thereof may be selected to provide an N/P ratio from about 5 to about 50, such as 5, 6, 7, 8, 9, 10, 12, 14, 16, 18, 20, 22, 24, 26, 28, 30, 32, 34, 36, 38, 40, 42, 44, 46, 48, or 50. In certain embodiments, the N/P ratio may be from about 5 to about 10. In other embodiments, the N/P ratio is from about 5 to about 20. In other embodiments, the N/P ratio may be from about 10 to about 20, about 10 to about 30, about 15 to about 30, about 15 to about 40, about 20 to about 30, about 20 to about 40, about 20 to about 50, about 30 to about 50, about 30 to about 40, or about 35 to about 50. [0134] Nucleic acids can be complexed on the interior of the lipid complex (e.g., liposomes, lipid nanoparticles). In some embodiments, the compositions have an encapsulation efficiency from about 75% to about 95%, or from about 85% to about 90%. In some examples, the encapsulation efficiency is from about 75% to about 100%. In some examples, the encapsulation efficiency is from about 75% to about 95%. In some examples, the encapsulation efficiency is from about 75% to about 90%. In some examples, the encapsulation efficiency is from about 75% to about 85%. In some examples, the encapsulation efficiency is from about 75% to about 80%. In some examples, the encapsulation efficiency is from about 80% to about 95%. In some examples, the encapsulation efficiency is from about 80% to about 90%. In some examples, the encapsulation efficiency is from about 80% to about 85%. [0135] The encapsulation efficiency (EE%) of nucleic acids such as RNA can be measured using a fluorescence plate-based assay employing the RiboGreen reagent. This assay measures the quantity of mRNA in samples with intact LNPs to determine the quantity of unencapsulated RNA as well as in LNP samples disrupted by triton X-100 to measure the total RNA. The % of encapsulation efficiency was calculated as the difference between the total RNA and the unencapsulated RNA divided by the total RNA. The efficiency of encapsulation of a therapeutic and/or prophylactic describes the amount of therapeutic and/or prophylactic that is encapsulated or otherwise associated with
a lipid composition after preparation, relative to the initial amount provided. In some embodiments, the encapsulation efficiency is desirably high (e.g., close to 100%). The encapsulation efficiency may be measured, for example, by comparing the amount of therapeutic and/or prophylactic in a solution containing the lipid complex composition before and after breaking up the lipid complex composition with one or more organic solvents or detergents. Fluorescence may be used to measure the amount of free therapeutic and/or prophylactic (e.g., RNA) in a solution. In some embodiments for the lipid compositions described herein, the encapsulation efficiency of a therapeutic and/or prophylactic may be at least 50%, for example 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or 100%. In some embodiments, the encapsulation efficiency may be at least 80%. In certain embodiments, the encapsulation efficiency may be at least 90%. Structural lipid: In some embodiments, the neutral lipid includes sterols, or lipids containing

moieties (“sterol derivatives”). As defined herein, "sterols" are a subgroup of steroids consisting of steroid alcohols. Exemplary sterols and lipids containing sterol moieties useful in the lipid composition formulations provided herein include, but are not limited to cholesterol, fecosterol, sitosterol, ergosterol, campesterol, stigmasterol, brassicasterol, tomatidine, tomatine, ursolic acid, alpha-tocopherol, hopanoids, phytosterols, steroids, and mixtures thereof. In some embodiments, the structural lipid is a sterol. Some lipid composition formulations provided herein include a sterol or sterol derivative. The sterols or sterol derivatives can be present at about 5-60 mol% of the overall lipid composition formulation. Advantageously, the sterol or sterol derivatives are present from about 15-50 mol%, e.g., 25-40 mol %. Preferably, the amount of the sterol (such as cholesterol) or sterol derivative in the lipid composition disclosed herein is at least about 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 38, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 51, 52, 53, 54, 55, 56, 57, 58, 59, or 60 mol % of the overall lipid formulation. Some lipid composition formulations provided herein do not include a sterol or sterol derivative.
[0137] In some embodiments, the compositions include a structural lipid in a concentration of 14-50 mol% of the composition, excluding any payload, if present. In some embodiments, the structural lipid is selected from the group consisting of: cholesterol, fecosterol, sitosterol, ergosterol, campesterol, stigmasterol, brassicasterol, tomatidine, tomatine, ursolic acid, alpha-tocopherol, hopanoids, phytosterols, steroids, and any combination thereof. Stabilizing agent: [0138] The lipid compositions provided herein can include a stabilizing agent, such as a stabilizing lipid. Stabilizing lipids can be neutral lipids, or they can be charged. Stabilizing lipids that can advantageously be used in the formulations provided herein include, but are not limited to, polyethylene glycol (PEG)-modified lipids. Non-limiting examples of PEG-lipids include PEG-modified phosphatidylethanolamine and phosphatidic acid, PEG-ceramide conjugates (e.g., PEG-CerC14 or PEG-CerC20), PEG- modified dialkylamines and PEG-modified 1,2-diacyloxypropan-3-amines. Such lipids are also referred to as PEGylated lipids. For example, a PEG lipid can be PEG-c-DOMG, PEG- DMG, PEG-DLPE, PEG-DMPE, PEG-DPPC, or a PEG-DSPE lipid. Other exemplarily PEGylated lipids include DSPE-PEG2000 (distearoyl-poly(ethylene glycol)), PEG2000- DSPE-PEG
500, 1,2-distearyloxypropyl-3-amine-PEG
2000, hexadecylcarbamoylmethyl hexadecanoate-PEG
2000, cholesteryl hemisuccinate-PEG
2000, photocleavable cholesteryl- PEG2000, cholesterol-hyperbranched polyglycerol, poly(2-methyl-2-oxazoline) (PMOZ)- or poly(2-ethyl-2-oxazoline) (PEOZ)-DSPE, poly(hydroxyethyl-l-asparagine)- succinyldioctadecylamine, DSPE-poly(2-tert-butoxy-N-(2-(methacryloyloxy)ethyl)-N,N- dimethyl-2-oxoethanamonium). In some embodiments, the lipid compositions do not include a PEGylated lipid. [0139] In some embodiments, the compositions include a stabilizing agent in a concentration of 0.1-10 mol% of the composition, excluding any payload, if present. In some embodiments, the stabilizing agent comprises one or more phospholipids selected from the group consisting of: 1,2-dilinoleoyl-sn-glycero-3-phosphocholine (DLPC), 1,2- dimyristoyl-sn-glycero-phosphocholine (DMPC), 1,2-dioleoyl-sn-glycero-3- phosphocholine (DOPC), 1,2-dipalmitoyl-sn-glycero-3-phosphocholine (DPPC), 1,2-
distearoyl-sn-glycero-3-phosphocholine (DSPC), 1,2-diundecanoyl-sn-glycero- phosphocholine (DUPC), 1-palmitoyl-2-oleoyl-sn-glycero-3-phosphocholine (POPC), 1,2-di-O-octadecenyl-sn-glycero-3-phosphocholine (18:0 Diether PC), 1-oleoyl-2- cholesterylhemisuccinoyl-sn-glycero-3-phosphocholine (OChemsPC), 1-hexadecyl-sn- glycero-3-phosphocholine (C16 Lyso PC), 1,2-dilinolenoyl-sn-glycero-3-phosphocholine, 1,2-diarachidonoyl-sn-glycero-3-phosphocholine, 1,2-didocosahexaenoyl-sn-glycero-3- phosphocholine, 1,2-dioleoyl-sn-glycero-3-phosphoethanola mine (DOPE), 1,2- diphytanoyl-sn-glycero-3-phosphoethanolamine (ME 16.0 PE), 1,2-distearoyl-sn-glycero- 3-phosphoethanolamine, 1,2-dilinoleoyl-sn-glycero-3-phosphoethanolamine, 1,2- dilinolenoyl-sn-glycero-3-phosphoethanolamine, 1,2-diarachidonoyl-sn-glycero-3- phosphoethanolamine, 1,2-didocosahexaenoyl-sn-glycero-3-phosphoethanolamine, 1,2- dioleoyl-sn-glycero-3-phospho-rac-(1-glycerol) sodium salt (DOPG), and sphingomyelin. In some embodiments, the formulations may also include one or more lipids derived from viral capsids, e.g, from enveloped viruses. [0140] Other stabilizing agents useful in the compositions disclosed herein include, e.g., polyglycol lipids, yoxyethylene alkyl ethers, diblock polyoxyethylene ether co-polymers, triblock polyoxyethylene alkyl ethers co-polymers, and amphiphilic branched polymers. In embodiments, stabilizing agent can be polyoxyethylene (20) oleyl ether, polyoxyethylene (23) lauryl ether, polyoxyethylene (40) stearate ("Myrj52"), poly(propylene glycol)11-block-poly(ethylene glycol)16-block-poly(propylene glycol)11, poly(propylene glycol)12-block-poly(ethylene glycol)28-block-poly(propylene glycol)12, polysorbate 80 (also known as Tween 80, IUPAC name 2-[2-[3,4-bis(2- hydroxyethoxy)oxolan-2-yl]-2-(2-hydroxyethoxy)ethoxy]ethyl octadec-9-enoate), Myrj52 (Polyoxyethylene (40) stearate), Brij™ S10 (Polyoxyethylene (10) stearyl ether), BRIJ™ L4 = Polyoxyethylene (4) lauryl ether; BRIJ™ S20= Polyoxyethylene (20) stearyl ether; BRIJ™ S35= Polyoxyethylene (23) lauryl ether; TPGS 1000 =D-Į-Tocopherol polyethylene glycol 1000 succinate; Tween 20/Polysorbate 80/ Tridecyl-D-maltoside in equal ratios, and combinations thereof. [0141] In some embodiments, stabilizing agent is selected from the group consisting of: a surfactant, a neutral lipid, a polymer-conjugated lipid, polyethylene glycol,
a phospholipid, and any combination thereof. In some embodiments, the stabilizing agent is a PEG-modified lipid. Exemplary PEG-modified lipids include, but are not limited to, a PEG-modified phosphatidylethanolamine, a PEG-modified phosphatidic acid, a PEG- ceramide conjugate, a PEG-modified dialkylamine, a PEG-modified 1,2-diacyloxypropan- 3-amine, and any combination thereof. In some embodiments, the PEG-modified lipid is selected from PEG-c-DOMG, PEG-DMG, PEG-DLPE, PEG-DMPE, PEG-DPPC, PEG- DSPE, and any combination thereof. [0142] In certain compositions, the stabilizing agent (e.g., PEGylated lipid or other stabilizing agent), is present at about 0.1 - 5 mol% of the lipid composition. For example, in some compositions, the stabilizing agent is present at about 0.5 mol%, 1 mol%, 1.5 mol%, 2 mol%, 2.5 mol %, 3 mol%, 3.5 mol %, 4 mol %, 4.5 mol%, 5 mol%, or any value in between, of the lipid composition. In other examples, the stabilizing agent is present at about 0.5 mol% to about 5 mol% of the lipid composition. In other examples, the stabilizing agent is present at about 0.5 mol% to about 4 mol% of the lipid composition. In other examples, the stabilizing agent is present at about 0.5 mol% to about 3 mol% of the lipid composition. In other examples, the stabilizing agent is present at about 0.5 mol% to about 2 mol% of the lipid composition. In other examples, the stabilizing agent is present at about 0.5 mol% to about 1 mol% of the lipid composition. In other examples, the stabilizing agent is present at about 1 mol% to about 5 mol% of the lipid composition. In other examples, the stabilizing agent is present at about 1 mol% to about 4 mol% of the lipid composition. In other examples, the stabilizing agent is present at about 1 mol% to about 3 mol% of the lipid composition. In other examples, the stabilizing agent is present at about 1 mol% to about 2 mol% of the lipid composition. Exosomes, exosome lipids: [0143] The lipid compositions provided herein can also be combined with one or more exosomes, or biological materials (e.g., lipids, proteins, nucleic acids, or the like) derived or purified from exosomes. [0144] The term “exosome” refers to the small membrane vesicles secreted by most cells that contain cell specific payloads of proteins, lipids and, genetic material and other biomolecules that are transported to other cells in different location of the tissue.
Exosomes can be considered liposomal particles. Exosomes or lipid mixtures obtained therefrom, can be used in combination with other transfection agents or helper lipid mixtures. Exosomes are also referred to as microvesicles, epididimosomes, argosomes, exosome-like vesicles, microparticles, promininosomes, prostasomes, dexosomes, texosomes, archeosomes and oncosomes. tExosomes useful in the compositions and methods described herein also include synthetic exosomes. Non-limiting examples of synthetic exosomes useful in the embodiments described herein are described, e.g., in Li, YJ., Wu, JY., Liu, J. et al. Artificial exosomes for translational nanomedicine. J Nanobiotechnol 19, 242 (2021), US Patent No. 11938219, US Patent Application Publication No. US2023/0181466, and the like, each of which is herein incorporated by reference in its entirety [0145] Examples of lipid constituents isolated from exosomes useful in the compositions provided herein include, but are not limited to, Lyso-PC (non-limiting examples of which C-18, C-16, C-14 and mixture), Lyso-bisphospahtidic acid (non- limiting example of which is C-18, C-16 and C-14), sphingomyelin, ceramides (non- limiting examples C-8- C-24), disaturated PC (non-limiting examples DSPC, DPPC, DMPC and others where Cn (n= 8 – 25)), diunsaturated PC-MIX (non-limiting examples of which are DOPC, DP(db)PC) phosphatidyl serine (PS), phosphatidyl inositol (PI)), disaturated PE non-limiting example, DSPE, DPPE, DMPE), di-unsaturated PE-MIX (non- limiting example DOPE DP(db)PE), phosphatidyl glycerol (PG) (non-limiting examples of which are C-18 – C-22), cholesterol, and diglycerides, such as cardiolipin. Ionizable Lipids: [0146] The compositions described herein can include an ionizable lipid. Suitable ionizable lipids include, but are not limited to, cationic lipids such as GeneIn
TM, LipofectAmine
TM 2000, LipofectAmine
TM, Lipofectin®, DMRIE-C, CellFectin® (Invitrogen), Oligofectamine® (Invitrogen), LipofectAce® (Invitrogen), Fugene® (Roche, Basel, Switzerland), Fugene® HD (Roche), Transfectam® (Tranfectam, Promega, Madison, WI), Tfx-10® (Promega), Tfx-20® (Promega), Tfx-50® (Promega), Transfectin
TM (BioRad, Hercules, CA), SilentFect
TM (Bio-Rad), Effectene® (Qiagen, Valencia, CA), DC-chol (Avanti Polar Lipids), GenePorter® (Gene Therapy Systems, San
Diego, CA), DharmaFect 1® (Dharmacon, Lafayette, CO), DharmaFect 2® (Dharmacon), DharmaFect 3® (Dharmacon), DharmaFect 4® (Dharmacon), Escort
TM III (Sigma, St. Louis, MO), Escort
TM IV (Sigma), DOTMA, DOTAP, DMRIE, DC-Chol, DDAB, DOSPA, DOSPER, DOGS, TMTPS, TMTOS, TMTLS, TMTMS, TMDOS, N-1- dimethyl-N-1-(2,3-diaoleoyloxypropyl)-2-hydroxypropane-1,3-diamine, N-1-dimethyl-N- 1-(2,3-diamyristyloxypropyl)-2-hydroxypropane-1,3-diamine, N-1-dimethyl-N-1-(2,3- diapalmityloxy-propyl)-2-hydroxypropane-1,3-diamine, N-1-dimethyl-N-1-(2,3- diaoleoyl-oxypropyl)-2-(3-amino-2-hydroxypropyloxy)propane-1,3-diamine, N-1- dimethyl-N-1-(2,3-diamyristyloxy-propyl)-2-(3-amino-2-hydroxypropyloxy)propane-1,3- diamine, N-1-dimethyl-N-1-(2,3-diapalm-ityloxypropyl)-2-(3-amino-2- hydroxypropyloxy)propane-1,3-diamine, L-spermine-5-carboxyl-3-(DL-1,2-dipalmitoyl- dimethylaminopropyl-ȕ-hydroxyethylamine, 3,5-(N,N-di-lysyl)-diamino-benzoyl-glycyl- 3-(DL-1,2-dipalmitoyl-dimethylaminopropyl-ȕ-hydroxyethylamine), L-Lysine-bis(O,O’- oleoyl-ȕ-hydroxy-ethyl)amidedihydrochloride, L-Lysine-bis-(O,O’-palmitoyl-ȕ- hydroxyethyl)amidedihydrochloride, 1,4-bis[(3-(3-aminopropyl)-alkylamino)-2- hydroxypropyl)-piperazine, L-Lysine-bis-(O,O’-myristoyl-ȕ-hydroxyethyl)amide dihydrochloride, L-Ornithine-bis-(O,O’-myristoyl-ȕ-hydroxyethyl)amide dihydrochloride, L-Ornithine-bis-(O,O’-oleoyl-ȕ-hydroxyethyl)amide dihydrochloride, 1,4-bis[(3-(3-aminopropyl)-oleylamino)-2-hydroxy-propyl]piperazine, L-Ornithine-bis- (O,O’-palmitoyl-ȕ-hydroxyethyl)amide dihydrochloride, 1,4,-bis[(3-amino-2- hydroxypropyl)-oleylamino]-butane-2,3-diol, 1,4,-bis[(3-amino-2-hydroxy-propyl)- palmitylamino]-butane-2,3-diol, 1,4,-bis[(3-amino-2-hydroxypropyl)-myristylamino]- butane-2,3-diol, 1,4-bis[(3-oleylamino)propyl]-piperazine, L-Arginine-bis-(O,O’-oleoyl- ȕ-hydroxyethyl)amide dihydrochloride, bis[(3-(3-aminopropyl)-myristylamino)2- hydroxy-propyl]piperazine, L-Arginine-bis-(O,O’-palmitoyl-ȕ-hydroxyethyl)amide dihydrochloride, L-Serine-bis-(O,O’-oleoyl-ȕ-hydroxyethyl)amide dihydrochloride, 1,4- bis[(3-(3-aminopropyl)-palmitylamino)-2-hydroxypropyl]piperazine, Glycine-bis-(O,O’- palmitoyl-ȕ-hydroxy-ethyl)amide dihydrochloride, Sarcosine-bis-(O,O’-palmitoyl-ȕ- hydroxyethyl)amide dihydrochloride, L-Histidine-bis-(O,O’-palmitoyl-ȕ- hydroxyethyl)amide dihydrochloride, cholesteryl-^ȕ-carboxyl-
amidoethylenetrimethylammonium iodide, 1,4-bis[(3-myristyl-amino)-propyl]- piperazine, 1-dimethylamino-3-trimethylammonio-DL-2-propyl-cholesteryl carboxylate iodide, cholesteryl-^ȕ-carboxyamidoethyleneamine, cholesteryl-^ȕ-oxysuccinamido- ethylene-trimethylammonium iodide, 1-dimethylamino-3-trimethylammonio-DL-2- propyl-cholesteryl-^ȕ-oxysuccinate iodide, 2-[(2-trimethylammonio)-ethylmethyl- amino]ethyl-cholesteryl-^ȕ-R[\VXFFLQDWH^ LRGLGH^^ ^ȕ>1-(N’, N’-dimethylamino-ethane)- FDUEDPR\O@FKROHVWHURO^^DQG^^ȕ-[N-(polyethyleneimine)-carbamoyl] cholesterol,1,4-bis[(3- palmitylamino)propyl]piperazine, L-Ornithylglycyl-N-(1-heptadecyloctadecyl)- glycinamide, N
2,N
5-Bis(3-aminopropyl)-L-ornithyl-glycyl-N-(1-heptadecyloctadecyl)- glycinamide, 1,4-bis[(3-(3-amino-2-hydroxypropyl)-alkyl-amino)-2-hydroxypropyl]- piperazine, N2-[N2,N5-Bis(3-aminopropyl)-L-ornithyl]-N,N-diocta-decyl-L- glutamine,N2-[N2,N5-Bis(aminopropyl)-L-ornithyl]-N-N-dioctadecyl-L-Į-glutamine, 1,4-bis[(3-(3-amino-2-hydroxypropyl)-oleylamino)2-hydroxypropyl]piperazine, N
2- [N
2,N
5-Bis(amino-propyl)-L-ornithyl]-N-N-dioctadecyl-L-Į-asparagine, N-[N
2-[N
2,N
5- Bis[(1,1-dimethyl-ethoxy)carbonyl]-N
2,N
5-bis[3-[(1,1- dimethylethoxy)carbonyl]aminopropyl]-L-ornithyl-N-N-dioctadecyl-L-glutaminyl]-L- glutamic acid, N
2-[N
2,N
5-Bis(3-aminopropyl)-L-ornithyl]-N,N-diolyl-L-glutamine, N
2- [N
2,N
5-Bis(aminopropyl)-L-ornithyl]-N-N-dioleyl-L-Į-glutamine,4-bis[(3-(3-amino-2- hydoxypropyl)-myristylamino)-2-hydroxy-propyl]piperazine, N
2-[N
2,N
5- Bis(aminopropyl)-L-ornithyl]-N-N-dioleyl-L-Į-asparagine, N-[N
2-[N
2,N
5-Bis[(1,1- dimethylethoxy)carbonyl]-N
2,N
5-bis[3-[(1,1-dimethylethoxy)carbonyl]aminopropyl]-L- ornithyl-N-N-dioleyl-L-glutaminyl]-L-glutamic acid, 1,4-bis[(3-(3-aminopropyl)-oleyl- amino)-propyl]piperazine, N
2-[N
2,N
5-Bis(3-aminopropyl)-L-ornithyl]-N,N-dipalmityl-L- glutamine,N
2-[N
2,N
5-Bis(amino-propyl)-L-ornithyl]-N-N-dipalmityl-L-Į-glutamine, N
2- [N
2,N
5-Bis(amino-propyl)-L-ornithyl]-N-N-dipalmityl-L-Į-asparagine, N-[N
2-[N
2,N
5- Bis[(1,1-dimethylethoxy)-carbonyl]-N
2,N
5-bis[3-[(1,1- dimethylethoxy)carbonyl]aminopropyl]-L-ornithyl-N-N-dipalmityl-L-glutaminyl]-L- glutamic acid, N
2-[N
2,N
5-Bis(3-aminopropyl)-L-ornithyl]-N,N-dimyristyl-L-glutamine, N
2-[N
2,N
5-Bis(aminopropyl)-L-ornithyl]-N-N-dimyristyl-L-Į-glutamine, N
2-[N
2,N
5- Bis(aminopropyl)-L-ornithyl]-N-N-dimyristyl-L-Į-asparagine, 1,4-bis[(3-(3-amino-2-
hydroxy-propyl)-palmitylamino)-2-hydroxypropyl]-piperazine, N-[N
2-[N
2,N
5-Bis[(1,1- dimethyl-ethoxy)carbonyl]-N
2,N
5-bis[3-[(1,1-dimethylethoxy)carbonyl]aminopropyl]-L- ornithyl-N-N-dimyristyl-L-glutaminyl]-L-glutamic acid, 1,4-bis[(3-(3-aminopropyl)- myristyl-amino)-propyl]piperazine, N
2-[N
2,N
5-Bis(3-aminopropyl)-L-ornithyl]-N,N- dilaureyl-L-glutamine, N
2-[N
2,N
5-Bis(amino-propyl)-L-ornithyl]-N-N-dilaureyl-L-Į- glutamine, N
2-[N
2,N
5–Bis-(amino-propyl)-L-ornithyl]-N-N-dilaureyl-L-Į-asparagine, N- [N
2-[N
2,N
5-Bis[(1,1-dimethylethoxy)-carbonyl]-N
2,N
5-bis[3-[(1,1- dimethylethoxy)carbonyl]aminopropyl]-L-ornithyl-N-N-dilaureyl-L-glutaminyl]-L- glutamic acid, 3-[N’,N”-bis(2-tertbutyloxycarbonyl-amino-ethyl)guanidino]-N,N- dioctadec-9-enylpropionamide, 3-[N’,N”-bis(2-tertbutyloxy-carbonylamino- ethyl)guanidino]-N,N-dipalmitylpropionamide, 3-[N’,N”-bis(2-tertbutyl-oxycarbonyl- aminoethyl)guanidino]-N,N-dimyristylpropionamide, 1,4-bis[(3-(3-amino-propyl)- palmityl-amino)propyl]piperazine, 1,4-bis[(3-(3-amino-2-hydroxypropyl)-oleyl- amino)propyl]piperazine, N,N-(2-hydroxy-3-aminopropyl)-N-2-hydroxypropyl-3-N,N- diolylaminopropane, N,N-(2-hydroxy-3-aminopropyl)-N-2-hydroxypropyl-3-N,N- dipalmitylaminopropane, N,N-(2-hydroxy-3-aminopropyl)-N-2-hydroxypropyl-3-N,N- dimyristylaminopropane, 1,4-bis[(3-(3-amino-2-hydoxypropyl)-myristylamino)- propyl]piperazine, [(3-aminopropyl)-bis-(2-tetradecyl-oxyethyl)]methyl ammonium bromide, [(3-aminopropyl)-bis-(2-oleyloxyethyl)]methyl ammonium bromide, [(3- aminopropyl)-bis-(2-palmityloxyethyl)]methyl ammonium bromide, oleoyl-2-hydroxy-3- N,N-dimethyamino propane, 2-didecanoyl-1-N,N-dimethylaminopropane, palmitoyl-2- hydroxy-3-N,N-dimethyamino propane, 1,2-dipalmitoyl-1-N,N-dimethylamino-propane, myristoyl-2-hydroxy-3-N,N-dimethyamino propane, 1,2-dimyristoyl-1-N,N- dimethylaminopropane, (3-amino-propyl)-›4-(3-amino-propylamino)-4-tetradecyl- carbamoyl-butylcarbamic acid cholestryl ester, (3-Amino-propyl)-›4-(3-amino- propylamino-4-carbamoylbutylcarbamic acid cholestryl ester, (3-Amino-propyl)-›4-(3- amino-propylamino)-4-(2-dimethylamino-ethylcarbamoy-l)-butylcarbamic acid cholesteryl ester, Spermine-5-carboxyglycine (N’-stearyl-N’-oleyl) amide tetratrifluoro- acetic acid salt, Spermine-5-carboxyglycine (N’-stearyl-N’-elaidyl) amide tetratri- fluoroacetic acid salt, Agmatinyl carboxycholesterol acetic acid salt, Spermine-5-carboxy-
ȕ-alanine cholesteryl ester tetratrifluoroacetic acid salt, 2,6-'LDPLQRKH[DQRH\O^ȕ-alanine cholesteryl ester bistrifluoroacetic acid salt, 2,4-'LDPLQREXW\UR\O^ ȕ-alanine cholesteryl ester bistrifluoroacetic acid salt, N,N-Bis (3-aminopropyl)-3-aminopropionyO^ ȕ-alanine cholesteryl ester tristrifluoroacetic acid salt, [N,N-Bis(2-hydroxyethyl)-2- aminoethyl]aminocarboxy cholesteryl ester, Stearyl carnitine ester, Palmityl carnitine ester, Myristyl carnitine ester, Stearyl stearoyl carnitine ester chloride salt, L-Stearyl Stearoyl Carnitine Ester, Stearyl oleoyl carnitine ester chloride, Palmityl palmitoyl carnitine ester chloride, Myristyl myristoyl carnitine ester chloride, L-Myristyl myristoyl carnitine ester chloride, 1,4-bis[(3-(3-amino-2-hydroxypropyl)-palmityl-amino)propyl]- piperazine, N-(3-aminopropyl)-N,N’-bis-(dodecyloxyethyl)-piperazinium bromide, N-(3- aminopropyl)-N,N’-bis-(oleyloxyethyl)-piperazinium bromide, N-(3-aminopropyl)-N,N’- bis-(palmityloxyethyl)-piperazinium bromide, N-(3-aminopropyl)-N,N’-bis- (myristyloxyethyl)-piperazinium bromide, N-(3-aminopropyl)-N’-methyl-N,N’-(bis-2- dodecyloxyethyl)-piperazinium bromide, N-(3-aminopropyl)-N’-methyl-N,N’-(bis-2- oleyloxyethyl)-piperazinium bromide, N-(3-aminopropyl)-N’-methyl-N,N’-(bis-2- palmityloxyethyl)-piperazinium bromide, N-(3-aminopropyl)-N’-methyl-N,N’-(bis-2- myristyloxyethyl)-piperazinium bromide, 1,4-bis[(3-(3-aminopropyl)-oleylamino)-2- hydroxy-propyl]piperazine, 1,4-bis[(3-(3-aminopropyl)-myristylamino)-2-hydroxy- propyl]piperazine, or 1,4-bis[(3-(3-aminopropyl)-palmitylamino)-2-hydroxypropyl]- piperazine, 2,3-dioleyloxy-1,4-N,N’-dimethyl-N,N’-di(2-hydroxy-3-aminopropyl)- diaminobutane, 2,3-dipalmitoleoyloxy-1,4-N,N’-dimethyl-N,N’-di(2-hydroxy-3-amino- propyl)-diaminobutane, 2,3-dimyristoleoyloxy-1,4-N,N’-dimethyl-N,N’-di(2-hydroxy-3- aminopropyl)-diaminobutane, 2,3-dioleyloxy-1,4-N,N’-dimethyl-N,N’-di(3-amino- propyl)-diaminobutane, 2,3-dipalmitoleoyloxy-1,4-N,N’-dimethyl-N,N’-di(3-amino- propyl)-diamino-butane, 2,3-dimyristoleoyloxy-1,4-N,N’-dimethyl-N,N’-di(3-amino- propyl)-diaminobutane, 2,3-dioleyloxy-1,4-N,N’-dimethyl-N,N’-di(5-carboxamido- spermine)-diaminobutane, 2,3-dipalm-itoleoyloxy-1,4-N,N’-dimethyl-N,N’-di(5- carboxamidospermine)-diaminobutane, 2,3-dimyrist-oleoyloxy-1,4-N,N’-dimethyl-N,N’- di(5-caqrboxamidospermine)-diaminobutane, 2,3-dioleyloxy-1,4-N,N’-dimethyl-N,N’- di(lysyl)-diaminobutane, 2,3-dipalmitoleoyloxy-1,4-N,N’-dimethyl-N,N’-di(lysyl)-
diaminobutane, 2,3-dimyristoleoyloxy-1,4-N,N’-dimethyl-N,N’-di(lysyl)-diamino- butane, 2,3-dioleyloxy-1,4-N,N’-dimethyl-N,N’-di(histidyl)-diaminobutane, 2,3- dipalmitoleoyl-oxy-1,4-N,N’-dimethyl-N,N’-di(histidyl)-diaminobutane, 2,3- dimyristoleoyloxy-1,4-N,N’-dimethyl-N,N’-di(histidyl)-diaminobutane, 2,3-dioleyloxy- N,N’-dimethyl-1,4 -diaminobutane, 2,3-dipalmitoleoyloxy-N,N’-dimethyl-1,4- diaminobutane, 2,3-dimyrist-oleoyloxy-N,N’-dimethyl-1,4-diaminobutane; PAMAM dendrimers, NH3 core dendrimers, ethylenediamine core dendrimers, polyethylenimine, and polyethylenimine conjugates. In some embodiments, the compositions include one or more cationic lipids. Polymers: [0147] The formulations described herein may also include one or more polymer. In some embodiments, the polymer is selected from a dense star dendrimer, a PAMAM dendrimer, an NH3 core dendrimer, an ethylenediamine core dendrimer, a dendrimer of generation 5 or higher, a dendrimer with a substituted group, a dendrimer comprising one or more amino acids, a grafted dendrimer, an activated dendrimer, polyethylenimine, polyethylenimine conjugates, polylysine, polyarginine, polyornithine, histone, and any combination thereof. In some embodiments, the polymer is a linear or branched PEI. Methods of Use: [0148] Use of these compositions in transfection can be carried out by methods that are known in the art. See for example, WO07/130073, at pages 54-60 which describes "before" and "after" protocols for transfection where the components of a transfection complex are mixed in differing orders prior to addition to a cell culture. Typically, a liposomal preparation of the lipid, with or without colipid is prepared, and is then mixed with a macromolecule, such as a DNA molecule or RNAi molecule to form a transfection complex. The complex is then added to a cell culture and transfection is monitored using well known methods. Additional components such as cell surface ligands, fusion agents, nuclear localization agents and the like may be added to the nucleic acid prior to admixture with the liposome, or may be added to the liposome prior to addition of nucleic acid.
[0149] An aspect of the present application relates to a method of delivering a payload to a cell, including: providing a composition described herein; providing a cell; and contacting the cell with the composition. [0150] A further aspect of the present application relates to method for delivering a composition to a subject, including: administering the composition described herein to the subject. In some embodiments, the administration is selected from the group consisting of: subcutaneous administration, intramuscular administration, intranasal administration, intra-tumoral administration, administration to the brain, administration to the spinal cord, administration to the eye, administration to the lymph node of a subject, and any combination thereof. In some embodiments, the administration is systemic. [0151] Cells which can be transfected according to these methods include, but are not limited to, virtually any eukaryotic cell including primary cells, cells in culture, a passaged cell culture or a cell line, and cells in cultured tissue. Suitable cells include human cell lines and animal cell lines. The cell may be a fibroblast. The cells can be attached cells or cells in suspension (suspension cells). In certain illustrative aspects, the cells are suspension CHO-S cells and suspension 293-F cells. Other cells that may be used include, without limitation, 293, 293-S, CHO, Cos, 3T3, Hela, primary fibroblasts, A549, Be2C, SW480, CHOK1, Griptite 293, HepG2, Jurkat, LNCap, MCF-7, NIH-3T3, PC12, C6, Caco-2, COS-7, HL60, HT-1080, IMR-90, K-562, SK-BR3, PHP1, HUVEC, MJ90, NHFF, NDFF and primary neurons. In some embodiments, the eukaryotic cell is a mammalian cell. [0152] In some embodiments, the contacting of the cells is in vitro. In some embodiments, the contacting of the cells is ex vivo. In some embodiments, the contacting of the cells is in vivo. [0153] In another embodiment is a method for producing a protein which includes contacting a cell with a lipid-nucleic acid complex as described above, where the nucleic acid encodes the protein. The cells are incubated to produce the protein and the protein is collected. Cells which can be used for protein production are described above. In addition, any composition which includes a lipid of Formula (I) or (Ia) can be used for transfection of cells. Such compositions are further discussed herein, and include, but are
not limited to compositions comprising lipids of Formula (I) or (Ia), a co-lipid and an optional transfection enhancing agent such as a fusogenic peptide or protein. For example, the methods provided herein include methods of producing therapeutic or prophylactic proteins, either in vivo or in vitro. By way of example, the compositions provided herein can be used to deliver mRNA encoding immunogens (e.g, for vaccines), mRNA encoding therapeutic proteins (e.g, growth factors, enzymes, cytokines, or the like), or the like. [0154] In another embodiment is a method for inhibiting production of a protein in a cell, comprising contacting the cell with a lipid-nucleic acid complex as described above, where the nucleic acid is a double stranded RNA molecule, such as an RNAi or siRNA molecule designed to inhibit expression of the protein. Methods of designing such RNA molecules are well known in the art. Lipids of Formula (I) and (Ia) are particularly suitable for deliver of RNAi molecules in this fashion. The cells are incubated and the phenotypic consequence of inhibiting production of the selected protein is observed. The lipids formulated in this manner were used in transfection. Reagent kits: [0155] Components of the transfection compositions described above can be provided in a reagent kit. The kits contain the lipid of Formula (I) or (Ia) above, together with additional components, such as a neutral lipid, a cationic lipid, cell surface ligands, fusion agents, and/or nuclear localization agents and the like. The kit components may be separate or may be premixed in any manner. For example, the lipid of formula (I) or (Ia) may be admixed with one or more neutral lipid. Additional components may also be present in the same container or may be present in one or more separate containers. The kits typically include vessels, such as vials and/or tubes, which are packaged together, for example in a cardboard box. The kits can be shipped from a supplier to a customer. For example, in one example provided herein is a kit that includes a vial that includes a liposomal formulation as described above and, optionally, a transfection agent and a transfection enhancing peptide. The kit can also include, for example, a separate vessel that includes a transfection enhancing agent, such as a transfection enhancing peptide, for example Plus Reagent™ (Invitrogen Corp., Carlsbad, CA). The kit can also include in separate containers, cells, cell culture medium, and a reporter nucleic acid sequence, such
as a plasmid that expresses a reporter gene. In certain examples, the culture medium can be reduced-serum medium and/or protein expression medium. [0156] Also provided are kits containing a compound of Formula (I) or (Ia) and additional reagents such as a cationic lipid, a neutral lipid, an amphipathic peptide, an amphipathic peptide comprising a polycationic nucleic acid binding moiety, a cell surface ligand, a cell surface ligand comprising a polycationic nucleic acid binding moiety, a fusion agent, a fusion agent comprising a polycationic nucleic acid binding moiety, a nuclear localization peptide or protein, and a nuclear localization peptide or protein comprising a polycationic nucleic acid binding moiety. The kits may contain one, some, or all of these additional reagents, in any possible combination. Advantageously, the additional reagents include a cationic lipid, an amphipathic peptide and a cell surface ligand that contains a polycationic nucleic acid binding moiety. When the cell surface ligand is a peptide or protein, the polycationic nucleic acid binding moiety is a polybasic amino acid sequence. [0157] In one embodiment, a kit comprises individual portions of, or a mixture of, cationic lipid, such as a lipid of Formula (I) or (Ia) and peptide, protein or fragment thereof or modified peptide, protein or fragment thereof. In another embodiment, a kit comprises individual portions of, or a mixture of, polycationic polymers and peptide, protein or fragments thereof or modified peptide, protein or fragments thereof. Cationic lipid transfection kits can optionally include neutral lipid as well as other transfection- enhancing agents or other additives, and the relative amounts of components in the kit may be adjusted to facilitate preparation of transfection compositions. Kit components can include appropriate medium or solvents for other kit components. [0158] Payloads that can be delivered by the methods of this invention include nucleic acids, proteins, ribonucloeproteins, and the like, including DNA and RNA (including RNAi/siRNA) of any size from any source comprising natural bases or non- natural bases, and include those encoding and capable of expressing therapeutic or otherwise useful proteins in cells, those which inhibit undesired expression of nucleic acids in cells, those which inhibit undesired enzymatic activity or activate desired enzymes, those which catalyze reactions (ribozymes), and those which function in diagnostic assays (e.g.,
diagnostic nucleic acids). Therapeutic nucleic acids include those nucleic acids that encode or can express therapeutically useful proteins, peptides or polypeptides in cells, those which inhibit undesired expression of nucleic acids in cells, and those which inhibit undesired enzymatic activity or activate desired enzymes in cells. In certain embodiments, the payload comprises an RNA molecule. The compositions can be used to deliver RNA payloads such as mRNA, siRNA, shRNA, miRNA, self-replicating RNA (srRNA), self- amplifying RNA, stRNA, sgRNA, or combinations thereof. In some embodiments, the RNA molecule comprises more than one RNA molecule, e.g., more than one mRNA. In some embodiments, the payload includes a nucleic acid that encodes a therapeutic protein, such as an antibody, growth factor, cytokine, enzyme, or the like. The compositions and methods provided herein can also be readily adapted in view of the disclosure herein to introduce biologically active macromolecules other than nucleic acids including, among others, polyamines, polyamine acids, polypeptides and proteins into eukaryotic cells. Other materials useful, for example as therapeutic agents, diagnostic materials, research reagents, which can be bound to the peptides and modified peptides and introduced into eukaryotic cells by the methods of this invention. Yet other payloads include small molecules, nutrients, and the like. [0159] In some embodiments, the payloads described herein can include ribonucleoprotein complexes (e.g., Cas9/guide RNA) which are delivered into a cell at high efficiencies. A “ribonucleoprotein complex,” or “ribonucleoprotein particle” as provided herein refers to a complex or particle including a nucleoprotein and a ribonucleic acid. A “guide RNA” or “gRNA” as provided herein refers to a ribonucleotide sequence capable of binding a nucleoprotein, thereby forming ribonucleoprotein complex. In embodiments, the guide RNA includes one or more RNA molecules. A “nucleoprotein” as provided herein refers to a protein capable of binding a nucleic acid (e.g., RNA, DNA). Where the nucleoprotein binds a ribonucleic acid it is referred to as “ribonucleoprotein.” The interaction between the ribonucleoprotein and the ribonucleic acid may be direct, e.g., by covalent bond, or indirect, e.g., by non-covalent bond (e.g. electrostatic interactions (e.g. ionic bond, hydrogen bond, halogen bond), van der Waals interactions (e.g. dipole-dipole, dipole-induced dipole, London dispersion), ring stacking (pi effects), hydrophobic
interactions and the like). Non-limiting examples of ribonucleoproteins include ribosomes, telomerase, RNAseP, hnRNP, CRISPR associated protein 9 (Cas9) and small nuclear RNPs (snRNPs). The ribonucleoprotein may be an enzyme. In embodiments, the ribonucleoprotein is an endonuclease. Thus, in embodiments, the ribonucleoprotein complex includes an endonuclease and a ribonucleic acid. In embodiments, the endonuclease is a CRISPR associated protein 9 [0160] The compositions provided herein can be delivered to cells via in vivo administration. For in vivo administration, the pharmaceutical compositions are preferably administered parenterally (e.g., intraarticularly, intravenously, intraperitoneally, subcutaneously, intrathecally, intradermally, intratracheally, intraosseous, intramuscularly or intratumorally). In particular embodiments, the pharmaceutical compositions are administered intravenously, intrathecally, or intraperitoneally by a bolus injection. Other routes of administration include topical (skin, eyes, mucus membranes), oral, pulmonary, intranasal, sublingual, rectal, and vaginal administration. [0161] Typical applications include using well known procedures to provide intracellular delivery of siRNA to knock down or silence specific cellular targets in vitro and in vivo. Alternatively, applications include delivery of DNA or mRNA sequences that code for therapeutically useful polypeptides. In this manner, therapy is provided for genetic diseases by supplying deficient or absent gene products. Methods of the present invention may be practiced in vitro, ex vivo, or in vivo. For example, the compositions of the present invention can also be used for delivery of payloads to cells in vivo, using methods which are known to those of skill in the art. In another example, the compositions of the invention can be used for delivery of a payload to a sample of patient cells that are ex vivo, then are returned to the patient. [0162] For in vivo administration, the pharmaceutical compositions are preferably administered parenterally (e.g., intraarticularly, intravenously, intraperitoneally, subcutaneously, intrathecally, intradermally, intratracheally, intraosseous, intramuscularly or intratumorally). In particular embodiments, the compositions provided herein are administered intravenously, intrathecally, or intraperitoneally by a bolus injection. Other
routes of administration include topical (skin, eyes, mucus membranes), oral, pulmonary, intranasal, sublingual, rectal, and vaginal. [0163] For ex vivo applications, the compositions provided herein are preferably administered to biological samples that have been removed from the organism, then the cells are washed and restored to the organism. The organism may be a mammal, and in particular may be a mammal (e.g., a primate), such as a human. This process is used for cell reprogramming, genetic restoration, immunotherapy, for example. [0164] In one embodiment, the present invention provides a method of modulating the expression of a target polynucleotide or polypeptide. These methods generally include contacting a cell with a composition of the present invention that is associated with a payload (e.g., a nucleic acid) that is capable of modulating the expression of a target polynucleotide or polypeptide. As used herein, the term "modulating" refers to altering the expression of a target polynucleotide or polypeptide. Modulating can mean increasing or enhancing, or it can mean decreasing or reducing. [0165] In related embodiments, provided herein are methods of treating a disease or disorder characterized by overexpression of a polypeptide in a subject, including providing to the subject a composition provided herein, wherein the composition includes a payload that is a therapeutic agent selected from an siRNA, a microRNA, an antisense oligonucleotide, and a plasmid capable of expressing an siRNA, a microRNA, or an antisense oligonucleotide, and wherein the siRNA, microRNA, or antisense RNA includes a polynucleotide that specifically binds to a polynucleotide that encodes the polypeptide, or a complement thereof. [0166] In related embodiments, provided herein are methods of treating a disease or disorder characterized by under-expression of a polypeptide in a subject. These methods can include providing to the subject a composition as provided herein, wherein the composition includes a payload that is a therapeutic agent selected from an mRNA, a self-amplifying RNA (SAM), a self-replicating DNA, or a plasmid, comprises a nucleic acid therapeutic that specifically encodes or expresses the under-expressed polypeptide, or a complement thereof.
[0167] In one embodiment, the compounds, compositions, and methods and uses of the described herein are for delivering a biologically active agent to liver cells (e.g. hepatocytes). In one embodiment, the compounds, compositions, and methods and uses of the invention are for delivering a biologically active agent to a tumor or to tumor cells (e.g. a primary tumor or metastatic cancer cells). In another embodiment, the compounds, compositions, and methods and uses are for delivering a biologically active agent to the skin adipose, muscle and lymph nodes (subcutaneous dosing). [0168] For delivery of a biologically active agent to the liver or liver cells, in one embodiment a composition of the invention is contacted with the liver or liver cells of the via parenteral administration (e.g. intravenous, intramuscular, subcutaneous administration) or local administration (e.g. direct injection, portal vein injection, catheterization, stenting), to facilitate delivery. For delivery of a biologically active agent to the kidney or kidney cells, in one embodiment a composition of the invention is contacted with the kidney or kidney cells of the patient via parenteral administration (e.g. intravenous, intramuscular, subcutaneous administration) or local administration (e.g. direct injection, catheterization, stenting), to facilitate delivery. For delivery of a biologically active agent to a tumor or tumor cells, in one embodiment, a composition of the invention is contacted with the tumor or tumor cells of the patient via parenteral administration (e.g. intravenous, intramuscular, subcutaneous administration) or local administration (e.g. direct injection, catheterization, stenting), to facilitate delivery. [0169] For delivery of a payload to the CNS or CNS cells), in one embodiment compositions described herein can be contacted with the CNS or CNS cells (e.g. brain cells and/or spinal cord cells) of the patient via parenteral administration (e.g. intravenous, intramuscular, subcutaneous administration) or local administration (e.g. direct injection, catheterization, stenting, osmotic pump administration (e.g. intrathecal or ventricular)), to facilitate delivery. For delivery of a payload to the Peripheral Nervous System (PNS) or PNS cells, compositions described herein can be contacted with the PNS or PNS cells of the patient via parenteral administration (e.g. intravenous, intramuscular, subcutaneous administration) or local administration (e.g. direct injection), to facilitate delivery. For delivery of a payload to a lung or lung cells, compositions provided herein can be contacted
with the lung or lung cells of the patient via parenteral administration (e.g. intravenous, intramuscular, subcutaneous administration) or local administration (e.g. pulmonary administration directly to lung tissues and cells), to facilitate delivery. [0170] For delivery of a payload to the vasculature or vascular cells, compositions provided herein can be contacted with the vasculature or vascular cells of the patient via parenteral administration (e.g. intravenous, intramuscular, subcutaneous administration) or local administration (e.g. clamping, catheterization, stenting), to facilitate delivery. [0171] For delivery of a payload to the skin or skin cells (e.g. dermis cells and/or follicular cells), compositions described herein can be contacted with the skin or skin cells (e.g. dermis cells and/or follicular cells) of the patient via parenteral administration (e.g. intravenous, intramuscular, subcutaneous administration) or local administration (e.g. direct dermal application, iontophoresis), to facilitate delivery. For delivery of a payload to an eye or ocular cells (e.g. macula, fovea, cornea, retina), in one embodiment a composition of the invention is contacted with the eye or ocular cells (e.g. macula, fovea, cornea, retina) of the patient via parenteral administration (e.g. intravenous, intramuscular, subcutaneous administration) or local administration (e.g. direct injection, intraocular injection, periocular injection, subretinal, iontophoresis, use of eyedrops, implants), to facilitate delivery. For delivery of a payload to an ear or cells of the ear (e.g. cells of the inner ear, middle ear and/or outer ear), compositions provided herein can be contacted with the ear or cells of the ear (e.g. cells of the inner ear, middle ear and/or outer ear) of the patient as is generally known in the art, such as via parenteral administration (e.g. intravenous, intramuscular, subcutaneous administration) or local administration (e.g. direct injection), to facilitate delivery. For delivery of a payload (e.g. RNA encoding an immunogen) to cells of the immune system (e.g. antigen-presenting cells, including professional antigen presenting cells), compositions provided herein can be delivered intramuscularly, after which immune cells can infiltrate the delivery site and process delivered RNA and/or process encoded antigen produced by non-immune cells, such as muscle cells. Such immune cells can include macrophages (e.g. bone marrow derived macrophages), dendritic cells (e.g. bone marrow derived plasmacytoid dendritic cells
and/or bone marrow derived myeloid dendritic cells), monocytes (e.g. human peripheral blood monocytes), etc. (for example, see WO2012/006372 by Geall, Andy et al.). [0172] Immunization. For immunization purposes, compositions provided herein can be prepared as an injectable, a pulmonary or nasal aerosol, or in a delivery device (e.g. syringe, nebulizer, sprayer, inhaler, dermal patch, etc.). This delivery device can be used to administer a pharmaceutical composition to a subject, e.g. to a human, for immunization. [0173] For immunization purposes, in some embodiments, the embodiments provided herein include delivering one or more RNAs that encode(s) an immunogen. The immunogen can elicit an immune response which recognizes the immunogen, to provide immunity against a pathogen, or against an allergen, or against a tumor antigen. Immunizing against disease and/or infection caused by a pathogen is preferred. [0174] In some embodiments, the kit includes one or more compounds of Formula (I) or (Ia), and one or more of a structural lipid, an ionizable lipid, and a stabilizing agent. [0175] In some embodiments, the kit includes one or more compounds of Formula (I) or (Ia), one or more structural lipids, one or more stabilizing agents; and optionally, a payload. [0176] In some embodiments, the kit includes one or more compounds of Formula (I) or (Ia), one or more structural lipids, one or more stabilizing agents; one or more fusion agent; and optionally, a payload. [0177] Nucleic acids that can be transfected by the methods of this invention include DNA and RNA (including RNAi/siRNA) of any size from any source comprising natural bases or non-natural bases, and include those encoding and capable of expressing therapeutic or otherwise useful proteins in cells, those which inhibit undesired expression of nucleic acids in cells, those which inhibit undesired enzymatic activity or activate desired enzymes, those which catalyze reactions (ribozymes), and those which function in diagnostic assays (e.g., diagnostic nucleic acids). Therapeutic nucleic acids include those nucleic acids that encode or can express therapeutically useful proteins, peptides or
polypeptides in cells, those which inhibit undesired expression of nucleic acids in cells, and those which inhibit undesired enzymatic activity or activate desired enzymes in cells. [0178] The compositions and methods provided herein can also be readily adapted in view of the disclosure herein to introduce biologically-active macromolecules other than nucleic acids including, among others, polyamines, polyamine acids, polypeptides and proteins into eukaryotic cells. Other materials useful, for example as therapeutic agents, diagnostic materials, research reagents, which can be bound to the peptides and modified peptides and introduced into eukaryotic cells by the methods of this invention. EXAMPLES [0179] Unless otherwise defined, scientific and technical terms used in connection with the disclosure shall have the meanings that are commonly understood by those of ordinary skill in the art. Further, unless otherwise required by context, singular terms shall include pluralities and plural terms shall include the singular. Generally, nomenclatures utilized in connection with, and techniques of, cell and tissue culture, molecular biology, and protein and oligo- or polynucleotide chemistry and hybridization described herein are those well known and commonly used in the art. Standard techniques are used for recombinant DNA, oligonucleotide synthesis, and tissue culture and transformation (e.g., electroporation, lipofection). Enzymatic reactions and purification techniques are performed according to manufacturer’s specifications or as commonly accomplished in the art or as described herein. The nomenclatures utilized in connection with, and the laboratory procedures and techniques of, analytical chemistry, synthetic organic chemistry, and medicinal and pharmaceutical chemistry described herein are those well-known and commonly used in the art. Standard techniques are used for chemical syntheses, chemical analyses, pharmaceutical preparation, formulation, and delivery, and treatment of patients. Preparation of the Compounds of Formula I: [0180] The compounds disclosed herein may be synthesized by methods described below, or by modification of these methods. Ways of modifying the methodology include, among others, temperature, solvent, reagents etc., known to those
skilled in the art. In general, during any of the processes for preparation of the compounds disclosed herein, it may be necessary and/or desirable to protect sensitive or reactive groups on any of the molecules concerned. This may be achieved by means of conventional protecting groups, such as those described in Protective Groups in Organic Chemistry (ed. J.F.W. McOmie, Plenum Press, 1973); and T.W. Green, P.G.M. Wuts, Protecting Groups in Organic Synthesis (3rd ed.) Wiley, New York (1999), which are both hereby incorporated herein by reference in their entirety. The protecting groups may be removed at a convenient subsequent stage using methods known from the art. Synthetic chemistry transformations useful in synthesizing applicable compounds are known in the art and include e.g. those described in R. Larock, Comprehensive Organic Transformations, VCH Publishers, 1989, or L. Paquette, ed., Encyclopedia of Reagents for Organic Synthesis, John Wiley and Sons, 1995, which

both hereby incorporated herein by reference in their entirety. The routes shown and described herein are illustrative only and are not intended, nor are they to be construed, to limit the scope of the claims in any manner whatsoever. Those skilled in the art will be able to recognize modifications of the disclosed syntheses and to devise alternate routes based on the disclosures herein; all such modifications and alternate routes are within the scope of the claims. [0181] In the following schemes, protecting groups for oxygen atoms are selected for their compatibility with the requisite synthetic steps as well as compatibility of the introduction and deprotection steps with the overall synthetic schemes (T.W. Green, P.G.M. Wuts, Protecting Groups in Organic Synthesis (3rd ed.) Wiley, New York (1999)). [0182] If the compounds of the present technology contain one or more chiral centers, such compounds can be prepared or isolated as pure stereoisomers, i.e., as individual enantiomers or d(l) stereoisomers, or as stereoisomer-enriched mixtures. All such stereoisomers (and enriched mixtures) are included within the scope of the present technology, unless otherwise indicated. Pure stereoisomers (or enriched mixtures) may be prepared using, for example, optically active starting materials or stereoselective reagents well-known in the art. Alternatively, racemic mixtures of such compounds can be separated using, for example, chiral column chromatography, chiral resolving agents and the like.
[0183] The starting materials for the following reactions are generally known compounds or can be prepared by known procedures or obvious modifications thereof. For example, many of the starting materials are available from commercial suppliers such as Aldrich Chemical Co. (Milwaukee, Wisconsin, USA), Bachem (Torrance, California , USA), Emka-Chemce or Sigma (St. Louis, Missouri, USA). Others may be prepared by procedures, or obvious modifications thereof, described in standard reference texts such as Fieser and Fieser's Reagents for Organic Synthesis, Volumes 1-15 (John Wiley, and Sons, 1991), Rodd's Chemistry of Carbon Compounds, Volumes 1-5, and Supplementals (Elsevier Science Publishers, 1989), Organic Reactions, Volumes 1-40 (John Wiley, and Sons, 1991), March's Advanced Organic Chemistry, (John Wiley, and Sons, 5th Edition, 2001), and Larock's Comprehensive Organic Transformations (VCH Publishers Inc., 1989). EXAMPLES General procedures [0184] It will be apparent to the skilled artisan that methods for preparing precursors and functionality related to the compounds claimed herein are generally described in the literature. In these reactions, it is also possible to make use of variants which are themselves known to those of ordinary skill in this art, but are not mentioned in greater detail. The skilled artisan given the literature and this disclosure is well equipped to prepare any of the compounds. [0185] The present invention, thus generally described, will be understood more readily by reference to the following examples, which are provided by way of illustration and are not intended to be limiting of the present invention. [0186] It is recognized that the skilled artisan in the art of organic chemistry can readily carry out manipulations without further direction, that is, it is well within the scope and practice of the skilled artisan to carry out these manipulations. These include reduction of carbonyl compounds to their corresponding alcohols, oxidations, acylations, aromatic substitutions, both electrophilic and nucleophilic, etherifications, esterification and saponification and the like. These manipulations are discussed in standard texts such as March Advanced Organic Chemistry (Wiley), Carey and Sundberg, Advanced Organic
Chemistry (incorporated herein by reference in their entirety) and the like. All the intermediate compounds of the present invention were used without further purification unless otherwise specified. [0187] The skilled artisan will readily appreciate that certain reactions are best carried out when other functionality is masked or protected in the molecule, thus avoiding any undesirable side reactions and/or increasing the yield of the reaction. Often the skilled artisan utilizes protecting groups to accomplish such increased yields or to avoid the undesired reactions. These reactions are found in the literature and are also well within the scope of the skilled artisan. Examples of many of these manipulations can be found for example in T. W. Greene and P.G.M. Wuts Protecting Groups in Organic Synthesis, 4th Ed., John Wiley & Sons (2007), incorporated herein by reference in its entirety. [0188] The following example schemes are provided for the guidance of the reader, and represent preferred methods for making the compounds exemplified herein. These methods are not limiting, and it will be apparent that other routes may be employed to prepare these compounds. Such methods specifically include solid phase based chemistries, including combinatorial chemistry. The skilled artisan is thoroughly equipped to prepare these compounds by those methods given the literature and this disclosure. The compound numberings used in the synthetic schemes depicted below are meant for those specific schemes only, and should not be construed as or confused with same numberings in other sections of the application. [0189] Trademarks used herein are examples only and reflect illustrative materials used at the time of the invention. The skilled artisan will recognize that variations in lot, manufacturing processes, and the like, are expected. Hence the examples, and the trademarks used in them are non-limiting, and they are not intended to be limiting, but are merely an illustration of how a skilled artisan may choose to perform one or more of the embodiments of the invention. [0190] The following abbreviations have the indicated meanings: (BOC)
2O = di-tert-butyl dicarbonate DCM = dichloromethane
DIEA = N,N-Diisopropylethylamine DIPEA = N,N-Diisopropylethylamine DMF = N,N-dimethylformamide DMP = Dess Martin Periodinane DNs = dinitrosulfonyl ESBL = extended-VSHFWUXP^ȕ-lactamase EtOAc = ethyl acetate EA = ethyl acetate FCC = Flash Column Chromatography HATU = 2-(7-aza-1H-benzotriazole-1-yl)-1,1,3,3- tetramethyluronium hexafluorophosphate MeCN = acetonitrile NMR = nuclear magnetic resonance PE = Petroleum Ether Prep = preparatory Py = pyridine RT or rt = room temperature Sat. = saturated aqueous TBDMSCl = tert-butyldimethylsilyl chloride TBS = tert-butyldimethylsilyl TEA = triethylamine TFA = trifluoroacetic acid THF = tetrahydrofuran TLC = thin layer chromatography [0191] The following example schemes are provided for the guidance of the reader, and collectively represent an example method for making the compounds provided herein. Furthermore, other methods for preparing compounds described herein will be readily apparent to the person of ordinary skill in the art in light of the following
reaction schemes and examples. Unless otherwise indicated, all variables are as defined above. Example 1 Synthesis of 4-(2-ammonio-3-(methyl(4-(methylammonio)-2,3-bis(((E)-octadec-9- en-1-yl)oxy)butyl)amino)-3-oxopropyl)-1H-imidazol-1-ium (Compound 1) [0192] A heavy-walled glass pressure reactor charged with (-)-Dimethyl L- tartrate a-1 (7.12 g, 40 mmoles) and 2M methylamine in tetrahydrofuran (50 mL, 100 mmoles, 2.5 equiv.) was added and the reactor was sealed and heated to 90 °C and stirred for a 16-hour period resulting in a mixture that contained a white precipitate. After the overnight period, the reaction was allowed to cool to room temperature. The white crystalline material was collected by filtration and dried under reduced pressure for an overnight period. The recovered crystals were analyzed and yielded intermediate 1A. (6.4g, 36.1mmolrd, 91.1% yield). [0193] A heavy-walled glass flask equipped with an addition funnel, and condenser was charged with sodium hydride (1.8 g of 60% dispersion in silicon oil, 1.08 g net, 45 mmoles, 2.64 equiv.) and the sodium hydride was washed with hexane (3 x 50 mL) under inert conditions. The flask containing the sodium hydride was charged with 740 mL of tetrahydrofuran, the intermediate 1A (3.0 g, 17 mmoles) and 15-crown-5 ether (1.68 mL, 8 mmoles, 0.5 equiv.), sealed, stirred, and heated to reflux (78 °C) for a 16-hour period. After the 16-hour period, oleyl methanesulfonate (21.7 g, 62.7 mmoles, 3.68 equiv.) was added to the flask using the addition funnel, sealed, stirred, and heated to reflux (78 °C) for a 16-hour period. After the 16-hour reaction period the reaction was allowed to cool to room temperature and the entire reaction mixture was transferred to an extraction funnel and extracted with 1 liter of water. The upper layer was separated, collected, and dried down resulting in 19.91 g of a dark brown/orange oil. The resulting oil was purified on a normal phase column (0-10%) dichloromethane/methanol gradient to produce 2.89g (4.27mmoles, 25% yield) of the desired compound 1B. [0194] Charged a heavy-walled glass flask equipped with an addition funnel, stirring bar and condenser with tetrahydrofuran (500 mL) and the C18:1 diamide 1B (2.89 g, 4.27 mmoles). Slowly added lithium aluminum hydride (1M LAH in tetrahydrofuran,
57 mL, 57 mmoles) utilizing the addition funnel. After addition was complete, refluxed (78 °C) the reaction mixture for a 16-hour period. After the 16-hour period, allowed the reaction mix to cool to room temperature then neutralized/quenched the LAH with 2.16 mL of water, 2.16 mL of 15% sodium hydroxide and 6.48 mL of water. The resulting mixture was vacuum filtered with the flow-through collected and dried down under reduced pressure. The resulting material was purified utilizing a reverse phase column (100% methanol) to produce 0.76 g (1.17 mmoles, 27.4% yield) of desired material, intermediate 1E. [0195] Under inert conditions, charged a heavy-walled glass flask with tert- butyl 4-(2-((tert-butoxycarbonyl)amino)-3-((2,5-dioxopyrrolidin-1-yl)oxy)-3-oxopropyl)- 1H-imidazole-1-carboxylate 1C (0.209 g, 0.45 mmoles), 12.5 mL of dichloromethane the C18:1 diamine 1E (0.5 g, 0.77 mmoles). Stirred reaction mix for an overnight period at room temperature. After the overnight period, diluted the reaction mixture with 100 mL of dichloromethane and extracted with 100 mL of water and with 100 mL of saturated sodium bicarbonate. Collected the organic layer and dried down under reduced pressure for an overnight period. Purified the crude by normal phase chromatography (0-15%, methanol/chloroform gradient) to produce 0.258 g (0.26 mmoles, 58% yield) of desired product tert-butyl 4-(2-((tert-butoxycarbonyl)amino)-3-(methyl(4-(methylamino)-2,3- bis(((E)-octadec-9-en-1-yl)oxy)butyl)amino)-3-oxopropyl)-1H-imidazole-1-carboxylate 1D. Desired product identity confirmed by mass pec. Analysis (M + H
+ = 986.813. [0196] Under inert conditions, charged a heavy-walled glass flask with 0.233 g (0.24 mmoles) of tert-butyl 4-(2-((tert-butoxycarbonyl)amino)-3-(methyl(4- (methylamino)-2,3-bis(((E)-octadec-9-en-1-yl)oxy)butyl)amino)-3-oxopropyl)-1H- imidazole-1-carboxylate 1D, 10 mL of dichloromethane and trifluoroacetic acid (1.08 mL, 14mmoles). Equipped the flask with a stirring bar and mixed for a 1.5-hour period at room temperature. After the 1.5-hour period, dried down the reaction mixture under reduced pressure for an overnight period. Purified the resulting residue by reverse-phase chromatography (0-100%, 70% methanol in water/methanol gradient) to produce 0.102 g (0.09 mmoles, 38% yield) of
TP386613WO1 [0197] 4-(2-ammonio-3-(methyl(4-(methylammonio)-2,3-bis(((E)-octadec-9- en-1-yl)oxy)butyl)amino)-3-oxopropyl)-1H-imidazol-1-ium trifluoroacetate salt (Compound 1) product. TLC analysis of the purified material confirmed a single product. Desired product identity confirmed by mass spectral analysis (M + H
+ = 786.706) (free base). Example 2 [0198] LNP formulations were screened and assessed by in vivo functional testing using the RNA payload of the complex. Performance and transfection efficiency analyses included payload delivery, biodistribution, and expression of the payload-encoded protein Compositions including Compound 1 and helper lipids were made and complexed with mRNA. As shown in Table 1, the formulations examined varied in molar ratios of Compound 1 and helper lipids. Table 1. Exemplary LNP formulations Formulation Mono- Peptide Histidine DOPE DSPC Cholesterol DMG- N/P SEQ ID 7 ml

[0199] All the LNP formulations contained the Compound 1 and DOPE, Cholesterol, and DMG-PEG. Some formulations included peptide SEQ ID NO: 47. All the lipids were weighed and solubilized in ethanol at the desired molar ratio. This lipid mix and firefly luciferase (fLuc) mRNA were complexed into LNPs using a microfluidic device. The LNPs were dialyzed in phosphate buffer and particle size and homogeneity were measured using dynamic light scattering. The LNPs were injected in mice the next day.
[0200] Female BALB/c mice aged 6-10 weeks old were purchased from The Jackson Laboratory and were acclimatized for 7 days before the study. Mice were injected with LNPs equivalent to 10ௗµg fLuc mRNA using intravenous tail vein injection in a total volume of 200µl. $W^^ௗK^SRVW-injection, mice were anesthetized with isofluorane anesthesia DQG^LPDJHG^^^ௗPLQ^DIWHU^LQWUDSHULWRQHDO^LQMHFWLRQ^RI^10^ௗ^/^Rediject D-Luciferin (Perkin Elmer). Bioluminescence imaging was quantified in vivo (whole body) and ex vivo (organ) using an IVIS Lumina III imaging system (Perkin Elmer) and analyzed using Living Image software. [0201] All the LNP formulations with Compound 1 had particle size >300nm, polydispersity index < 0.3, (Figure 1). Intravenous administration of the LNP formulations with Compound 1 resulted in mRNA delivery and luciferase expression distributed between the lung and the spleen (Figures 2, 3 and 4) of the injected mice. OTHER EMBODIMENTS: [0202] While the invention has been described in conjunction with the detailed description thereof, the foregoing description is intended to illustrate and not limit the scope of the invention, which is defined by the scope of the appended claims. Other aspects, advantages, and modifications are within the scope of the following claims. [0203] The patent and scientific literature referred to herein establishes the knowledge that is available to those with skill in the art. All references, e.g., U.S. patents, U.S. patent application publications, PCT patent applications designating the U.S., published foreign patents and patent applications cited herein are incorporated herein by reference in their entireties. GenBank and NCBI submissions indicated by accession number cited herein are incorporated herein by reference. All other published references, documents, manuscripts and scientific literature cited herein are incorporated herein by reference. In the case of conflict, the present specification, including definitions, will control. In addition, the materials, methods, and examples are illustrative only and not intended to be limiting. [0204] While this invention has been particularly shown and described with references to preferred embodiments thereof, it will be understood by those skilled in the
art that various changes in form and details may be made therein without departing from the scope of the invention encompassed by the appended claims. [0205] Although the disclosure has been described with reference to the above examples, it will be understood that modifications and variations are encompassed within the spirit and scope of the disclosure. Accordingly, the disclosure is limited only by the following claims.