WO2025031990A1 - Methods of controlling or preventing infestation of soybean plants by the phytopathogenic microorganism corynespora cassiicola - Google Patents
Methods of controlling or preventing infestation of soybean plants by the phytopathogenic microorganism corynespora cassiicola Download PDFInfo
- Publication number
- WO2025031990A1 WO2025031990A1 PCT/EP2024/072024 EP2024072024W WO2025031990A1 WO 2025031990 A1 WO2025031990 A1 WO 2025031990A1 EP 2024072024 W EP2024072024 W EP 2024072024W WO 2025031990 A1 WO2025031990 A1 WO 2025031990A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- methyl
- plants
- compound
- formula
- phytopathogen
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Classifications
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/48—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with two nitrogen atoms as the only ring hetero atoms
- A01N43/56—1,2-Diazoles; Hydrogenated 1,2-diazoles
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01P—BIOCIDAL, PEST REPELLANT, PEST ATTRACTANT OR PLANT GROWTH REGULATORY ACTIVITY OF CHEMICAL COMPOUNDS OR PREPARATIONS
- A01P3/00—Fungicides
Definitions
- the present invention relates to methods for controlling or preventing infestation of soybean plants by the phytopathogenic microorganism Corynespora cassiicola.
- Corynespora cassiicola infects over 530 species of plants in 53 families (Dixon, L. J., et al., Phytopathology 2009, 99(9), 1015-27). It is most common in the tropics and subtropics. It has also been isolated from nematodes and from human skin. Corynespora cassiicola is known as a pathogen of many agricultural crop plants, for example soybeans, corn, wheat, cotton, beans, potatoes, sweet potato, and tobacco. The disease caused by Corynespora cassiicola is called target leaf spot or target spot on several plants, for example target leaf spot of soybeans and target leaf spot on cotton.
- pyrazole derivatives as disclosed in WO 2020/056090 and WO 2021/183707, are highly effective at controlling or preventing the infestation of cotton by the phytopathogenic microorganisms of the family Corynesporascaceae, in particular Corynespora cassiicola. These highly effective compounds thus represent an important new solution for farmers to control or prevent infestation of cotton by the phytopathogenic microorganism Corynespora cassiicola.
- pesticidal compositions comprising particular 5-(trifluoro- methyl)-1 ,2,4-oxadiazole compounds and one or more formulation adjuvants, are highly effective at controlling or preventing the infestation of plants, in particular field-grown plants, by the phytopathogenic microorganisms of the family Corynesporascaceae, in particular Corynespora cassiicola.
- These highly effective pesticidal compositions thus represent an important new solution for farmers to control or prevent infestation of plants, in particular field-grown plants, by the phytopathogenic microorganism Corynespora cassiicola.
- Said pyrazole derivatives Said pyrazole derivatives, its pesticidal action and suitable methods for producing are disclosed in WO 2020/056090.
- Agrochemical compositions comprising said pyrazole derivatives, have been disclosed in WO 2021/18370.
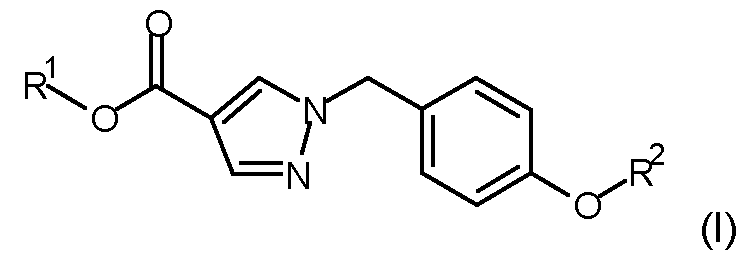
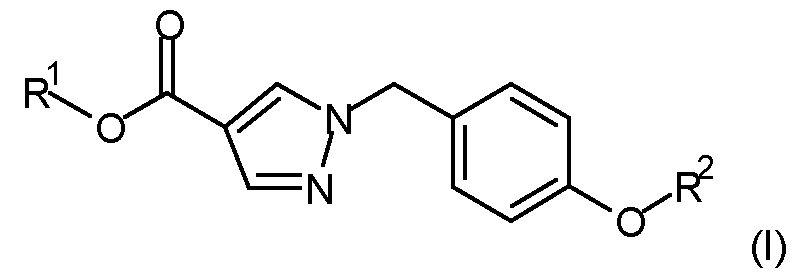
- a method of controlling or preventing infestation of plants by the phytopathogenic microorganism Corynespora cassiicola comprising applying to the phytopathogen, to the locus of the phytopathogen, or to a plant susceptible to attack by the phytopathogen, or to a propagation material thereof, a fungicidally effective amount of a compound according to formula (I) wherein
- R 1 is Ci-Csalkyl
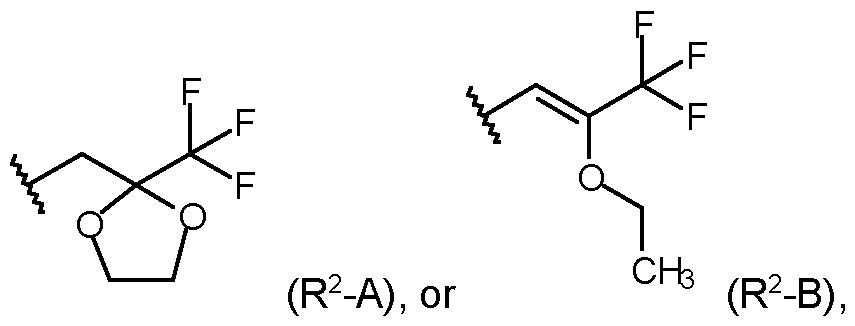
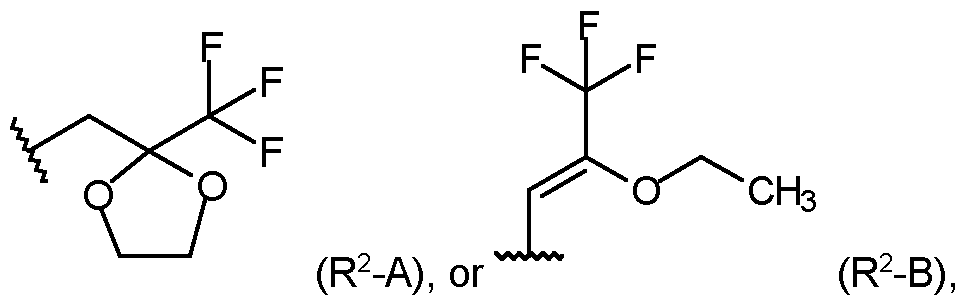
- R 2 is selected from the group consisting of: wherein the staggered li _ ne denotes the bond to the rest of the molecule; or an agrochemically acceptable salt, stereoisomer, enantiomer, tautomer or N-oxide thereof.
- a method of controlling or preventing infestation of plants by the phytopathogenic microorganism Corynespora cassiicola comprising applying to the phytopathogen, to the locus of the phytopathogen, or to a plant susceptible to attack by the phytopathogen, or to a propagation material thereof, a pesticidal composition comprising a compound of formula (I), and one or more formulation adjuvants.
- the compound according to formula (I), or the pesticidal composition comprising a compound of formula (I) and one or more formulation adjuvants is applied in the field.
- a method of controlling or preventing infestation of field-grown plants by the phytopathogenic microorganism Corynespora cassiicola comprising applying to the phytopathogen, to the locus of the phytopathogen, or to a plant susceptible to attack by the phytopathogen, or to a propagation material thereof, a compound of formula (I), or a pesticidal composition comprising a compound of formula (I) and one or more formulation adjuvants, formulation auxiliaries, diluents, and/or carriers.
- the method according to any one of embodiments above comprising the steps of providing a composition comprising a compound of formula (I); and applying the composition to a propagation material; and planting the propagation material.
- the method comprising the steps of providing a composition comprising a compound of formula (I); applying the composition to the phytopathogen, to the locus of the phytopathogen, or to a plant susceptible to attack by the phytopathogen.
- the pesticidal composition comprises at least and one or more formulation adjuvants, as well as and one or more formulation auxiliaries, diluents, and/or carriers.
- a compound of formula (I) for controlling or preventing infestation of plants by the phytopathogenic microorganism Corynespora cassiicola.
- a seventh aspect of the present invention there is provided a method or use according to any one of embodiments herein above, wherein the plant is selected from beans, cowpea, cucumber, papaya, soybean, potatoes, sweet potato, tomato, cotton, corn, wheat, tobacco, eggplant, basil, thyme, rubber tree, pawpaw tree, azalea and hydrangea.
- the plant is selected from soybeans, corn, wheat, cotton, beans, potatoes, sweet potato, and tobacco.
- the plants are genetically modified plants, preferably Bt plants.
- the present invention preferably also relates to a pesticidal composition suitable for control of diseases caused by phytopathogens comprising a component (A) and at least one or more compound(s) (B), wherein said component A is a compound of formula (I) wherein R 1 is Ci-Csalkyl; and R 2 is selected from the group consisting of: the molecule; and wherein said at least one or more compound(s) (B) is selected from other biologically active agents, such as bactericides, fungicides, nematicides, plant activators, acaricides, and insecticides, one or more adjuvant(s), a diluent or carrier, formulation auxiliary, or a combination thereof.
- the present invention preferably also relates to a pesticidal composition suitable for control of diseases caused by phytopathogens comprising a component (A) and at least one or more compound(s) (B), wherein said component A is a compound of formula (I) wherein R 1 is Ci-Csalkyl; and R 2 is selected from the group consisting of: wherein the staggered line denotes the bond to the rest of the molecule; and wherein said at least one or more compound(s) (B) is selected from pesticides.
- a pesticidal composition suitable for control of diseases caused by phytopathogens comprising a component (A) and at least one or more compound(s) (B), wherein said component A is a compound of formula (I) wherein R 1 is Ci-Csalkyl; and R 2 is selected from the group consisting of: wherein the staggered line denotes the bond to the rest of the molecule; and wherein said at least one or more compound(s) (B) is selected from pesticides.
- Compounds of formula (I) which have at least one basic centre can form, for example, acid addition salts, for example with strong inorganic acids such as mineral acids, for example perchloric acid, sulfuric acid, nitric acid, nitrous acid, a phosphorus acid or a hydrohalic acid, with strong organic carboxylic acids, such as Ci- C4alkanecarboxylic acids which are unsubstituted or substituted, for example by halogen, for example acetic acid, such as saturated or unsaturated dicarboxylic acids, for example oxalic acid, malonic acid, succinic acid, maleic acid, fumaric acid or phthalic acid, such as hydroxycarboxylic acids, for example ascorbic acid, lactic acid, malic acid, tartaric acid or citric acid, or such as benzoic acid, or with organic sulfonic acids, such as Ci- 04 alkane- or arylsulfonic acids which are unsubstituted or substituted, for example
- Compounds of formula (I) which have at least one acidic group can form, for example, salts with bases, for example mineral salts such as alkali metal or alkaline earth metal salts, for example sodium, potassium or magnesium salts, or salts with ammonia or an organic amine, such as morpholine, piperidine, pyrrolidine, a mono-, di- or tri-lower-alkylamine, for example ethyl-, diethyl-, triethyl- or dimethylpropylamine, or a mono-, di- ortrihydroxy-lower-alkylamine, for example mono-, di- ortriethanolamine.
- bases for example mineral salts such as alkali metal or alkaline earth metal salts, for example sodium, potassium or magnesium salts
- salts with ammonia or an organic amine such as morpholine, piperidine, pyrrolidine, a mono-, di- or tri-lower-alkylamine, for example ethyl-, die
- the compounds of formula (I) according to the invention are in free form, in oxidized form as a N-oxide or in salt form, e.g., an agronomically usable salt form.
- N-oxides are oxidized forms of tertiary amines or oxidized forms of nitrogen containing heteroaromatic compounds. They are described for instance in the book “Heterocyclic N-oxides” by A. Albini and S. Pietra, CRC Press, Boca Raton 1991 .
- the compounds of formula (I) according to the invention also include hydrates which may be formed during the salt formation.
- controlling refers to reducing the number of pests, eliminating pests and/or preventing further pest damage such that damage to a plant or to a plant derived product is reduced.
- pest refers to insects, and molluscs that are found in agriculture, horticulture, forestry, the storage of products of vegetable origin (such as fruit, grain, and timber); and those pests associated with the damage of man-made structures.
- the term pest encompasses all stages in the life cycle of the pest.
- effective amount refers to the amount of the compound, or a salt thereof, which, upon single or multiple applications provides the desired effect.
- an effective amount is readily determined by the skilled person in the art, using known techniques and by observing results obtained under analogous circumstances. In determining the effective amount, a number of factors are considered including, but not limited to the type of plant or derived product to be applied; the pest to be controlled and its lifecycle; the particular compound applied; the type of application; and other relevant circumstances.
- room temperature or “RT” or “rt” or “ambient temperature” refers to a temperature of about 15° C to about 35° C.
- rt can refer to a temperature of about 20° C to about 30° C.
- halogen refers to fluorine (fluoro), chlorine (chloro), bromine (bromo) or iodine (iodo), preferably fluorine, chlorine or bromine. This also applies, correspondingly, to halogen in combination with other meanings, such as haloalky I, haloalkenyl, haloalkynyl, haloalkoxy, and halocycloalkyl.
- Ci-C n -alkyl refers to a saturated straight-chain or branched hydrocarbon radical attached via any of the carbon atoms having 1 to n carbon atoms, for example, any one of the radicals methyl, ethyl, n-propyl, 1 -methylbutyl, 2-methylbutyl, 3-methylbutyl, 2, 2-dimethylpropyl, 1 -ethylpropyl, n-hexyl, n- pentyl, 1 ,1 -dimethylpropyl, 1 , 2-dimethylpropyl, 1 -methylpentyl, 2-methylpentyl, 3-methylpentyl, 4- methylpentyl, 1 ,1 -dimethylbutyl, 1 ,2-dimethylbutyl, 1 ,3-dimethylbutyl, 2,2-dimethylbutyl, 2,3-dimethylbutyl, 3,3- dimethylbutyl,
- Embodiments according to the invention are provided as set out below.
- R 1 is selected from methyl, or ethyl; and R 2 is selected from the group consisting of: wherein the staggered line denotes the bond to the rest of the molecule.
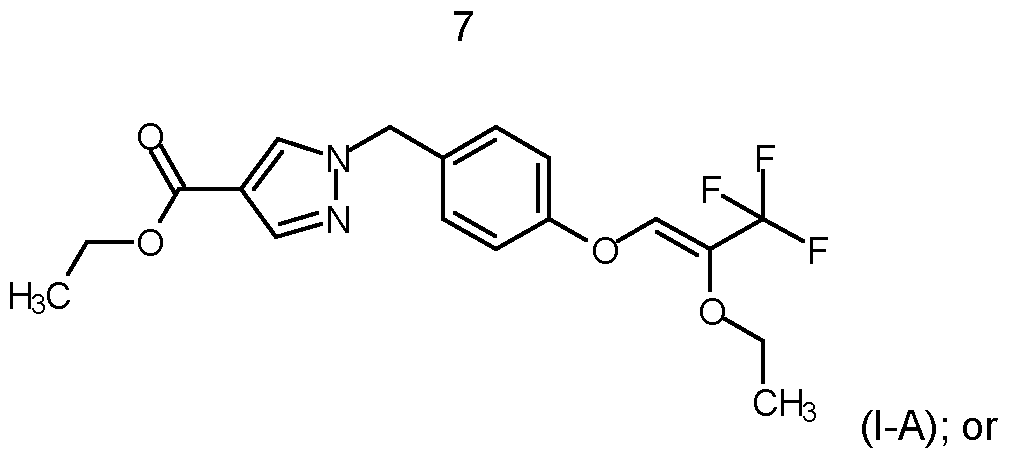
- compounds of formula (I) are selected from ethyl 1-[[4-[(Z)-2-ethoxy-3,3,3- trifluoro-prop-1-enoxy]phenyl]methyl]pyrazole-4-carboxylate according to structure (l-A) ethyl 1-[[4-[[2-(trifluoromethyl)-1 ,3-dioxolan-2-yl]methoxy]phenyl]methyl]pyrazole-4-carboxylate according to structure (l-B)
- compound of formula (I) is ethyl 1-[[4-[(Z)-2-ethoxy-3,3,3- trifluoro-prop-1-enoxy]phenyl]methyl]pyrazole-4-carboxylate according to structure l-A.
- compound of formula (I) is ethyl 1-[[4-[[2-(trifluoromethyl)- 1 ,3-dioxolan-2-yl]methoxy]phenyl]methyl]pyrazole-4-carboxylate according to structure l-B.
- the compound or compounds are generally applied as part of a pesticidal composition.
- composition comprising a compound of formula (I) for controlling or preventing infestation of plants by the phytopathogenic microorganism Corynespora cassiicola.
- Such pesticidal compositions typically comprise one or more adjuvants, formulation auxiliaries, diluents, and/or carriers as described in detail below, preferably at least one or more adjuvants.
- compositions comprising a compound of formula (I) and preferably one or more adjuvants, formulation auxiliaries, diluents, and/or carriers for controlling or preventing infestation of plants by the phytopathogenic microorganism Corynespora cassiicola comprising the steps of providing a composition comprising said compound of formula (I); and applying said composition to the phytopathogen, to the locus ofthe phytopathogen, orto a plant susceptible to attack by the phytopathogen.
- a fungicidal composition comprising a mixture of component (A) and a component (B) as active ingredients, wherein component (A) is selected from a compound of formula (I), (l-A), or (l-B), and component (B) is a compound selected from the group consisting of benzovindiflupyr, fluxapyroxad, pydiflumetofen, isopyrazam, fluopyram, penthiopyrad, sedaxane, bixafen, difenoconazole, cyproconazole, tebuconazole, hexaconazole, prothioconazole, propiconazole, epoxiconazole, metconazole, tetraconazole, fluoxytioconazole, 2-[2-chloro-4-(4-chlorophenoxy)phenyl]-2-hydroxy-3-(1 ,2,4- triazol-1-y
- component (A) is ethyl 1 -[[4-[(Z)-2-ethoxy- 3,3,3-trifluoro-prop-1-enoxy]phenyl]methyl]pyrazole-4-carboxylate (compound l-A), or a salt, enantiomer, tautomer or N-oxide thereof
- component (B) is selected from the group consisting of benzovindiflupyr, fluxapyroxad, pydiflumetofen, isopyrazam, fluopyram, penthiopyrad, sedaxane, bixafen, difenoconazole, cyproconazole, tebuconazole, hexaconazole, prothioconazole, propiconazole, epoxiconazole, metconazole, tetraconazole, fluoxytioconazole, 2-[2-chloro-4-(4
- component (A) is ethyl 1-[[4-[[2-(trifluoromethyl)-
- component (B) is selected from the group consisting of benzovindiflupyr, fluxapyroxad, pydiflumetofen, isopyrazam, fluopyram, penthiopyrad, sedaxane, bixafen, difenoconazole, cyproconazole, tebuconazole, hexaconazole, prothioconazole, propiconazole, epoxiconazole, metconazole, tetraconazole, fluoxytioconazole, 2-[2-chloro-4-(4-chlorophenoxy)phenyl]-2-hydroxy-3-(1 , 2 ,4-tri azo 1-1 - yl
- the weight ratio of component (A) to component (B) may be from 1000: 1 to 1 :1000, may be from 100:1 to 1 :100, preferably from 50:1 to 1 :50, more preferably from 20:1 to 1 :40, even more preferably from 15:1 to 1 :30, still more preferably from 12:1 to 1 :25, or from 10:1 to 1 :20, or from 10:1 to 1 :10, or from 5:1 and 1 :15, or from 5:1 to 1 :5, or from 4:1 to 1 :4, or from 3:1 to 1 :10, or from 3:1 to 1 :3, or from 2:1 to 1 :5, or 1 :1.
- the composition may comprise an additional active ingredient component (C), which is different to component (B), and is selected from the group consisting of metyltetrapole, azoxystrobin, trifloxystrobin, picoxystrobin, pyraclostrobin, metominostrobin, prothioconazole, cyproconazole, difenoconazole, tebuconazole, propiconazole, epoxiconazole, hexaconazole, mefentrifluconazole, metconazole, benzovindiflupyr, pydiflumetofen, fluxapyroxad, sedaxane, bixafen, isopyrazam, fluopyram, fluindapyr, isoflucypram, inpyrfluxam, quinofumelin, ipflufenoquin, aminopyrifen, fluazinam, fludioxonil, fen
- C additional active ingredient component
- the component (C) compounds are referred to herein and above by a so-called "ISO common name” or another "common name” being used in individual cases or a trademark name.
- the component (C) compounds are known and are commercially available and/or can be prepared using procedures known in the art.
- a fungicidal composition comprising a mixture of component (A) and a component (B) and a component (C) as active ingredients, wherein component (A) is selected from a compound of formula (I), (l-A), or (l-B), and component (B) and (C) are a compound selected from the group consisting of benzovindiflupyr, fluxapyroxad, pydiflumetofen, isopyrazam, fluopyram, penthiopyrad, sedaxane, bixafen, difenoconazole, cyproconazole, tebuconazole, hexaconazole, prothioconazole, propiconazole, epoxiconazole, metconazole, tetraconazole, fluoxytioconazole, 2-[2-chloro-4- (4-chlorophenoxy)phenyl]-2-hydroxy-3-(1
- component (A) is N-(2-fluorophenyl)-4-[5- (trifluoromethyl)-l ,2,4-oxadiazol-3-yl]benzamide (compound l-A), or a salt, enantiomer, tautomer or N-oxide thereof
- component (B) and (C) are a compound selected from the group consisting of benzovindiflupyr, fluxapyroxad, pydiflumetofen, isopyrazam, fluopyram, penthiopyrad, sedaxane, bixafen, difenoconazole, cyproconazole, tebuconazole, hexaconazole, prothioconazole, propiconazole, epoxiconazole, metconazole, tetraconazole, fluoxytioconazole, 2-[2-chloro-4-(4-chloroph)
- component (A) is N-(2-fluorophenyl)- 4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3-yl]benzamide (compound l-B), or a salt, enantiomer, tautomer or N- oxide thereof
- component (B) and (C) are a compound selected from the group consisting of benzovindiflupyr, fluxapyroxad, pydiflumetofen, isopyrazam, fluopyram, penthiopyrad, sedaxane, bixafen, difenoconazole, cyproconazole, tebuconazole, hexaconazole, prothioconazole, propiconazole, epoxiconazole, metconazole, tetraconazole, fluoxytioconazole, 2-[2-chloro-4-(4-chlorophenoxy)
- Components (B) and (C) in combination with component (A) may enhance the effectiveness of the latter against fungi, and vice versa. Additionally, the fungicidal compositions may be effective against a wider spectrum of fungal pathogens that can be combated with the individual active ingredients when used solely.
- the weight ratio of component (A) to the mixture of components (B) and (C) may be from 100:1 to 1 :100, or 50:1 to 1 :50, or 20:1 to 1 :20, or 10:1 to 1 :10, or 5:1 and 1 :5.
- the weight ratio of component (A) to the mixture of components (B) and (C) may be from 2:1 to 1 :2, or 4:1 to 2:1 , or 1 :1 , or 5:1 , or 5:2, or 5:3, or 5:4, or 4:1 , or 4:2, or 4:3, or 3:1 , or 3:2, or 2:1 , or 1 :5, or 2:5, or 3:5, or 4:5, or 1 :4, or 2:4, or 3:4, or 1 :3, or 2:3, or 1 :2, or 1 :600, or 1 :300, or 1 :150, or 1 :35, or 2:35, or 4:35, or 1 :75, or 2:75, or 4:75, or 1 :6000, or 1 :3000, or 1 :1500, or 1 :350, or 2:350, or 4:350, or 1 :750, or 2:750, or 4:750.
- Those mixing ratios are understood to include, on the one hand, ratios by weight and also,
- the weight ratio of component (A) to the sum of component (B) and component (C) may be from 100:1 to 1 :100, preferably from 50:1 to 1 :50, more preferably from 20:1 to 1 :40, even more preferably from 15:1 to 1 :30, still more preferably from 12:1 to 1 :25, or from 10:1 to 1 :20, or from 10:1 to 1 :10, or from 5:1 and 1 :15, or from 5:1 to 1 :5, or from 4:1 to 1 :4, or from 3:1 to 1 :10, or from 3:1 to 1 :3, or from 2:1 to 1 :5, or 1 :1 .
- the component (B) compounds are referred to herein and above by a so-called "ISO common name” or another "common name” being used in individual cases or a trademark name.
- the component (B) compounds are known and are commercially available and/or can be prepared using procedures known in the art and/or procedures reported in the literature such as, for instance, WO 2015/155075 and WO 2016/202742, WO 2017/005710, WO 2018/108977, WO 2018/153707, WO 2018/098216, WO 2019/093522, WO 2018/145921 , WO 2015/185485, WO 2017/076742, WO 2017/081311 , WO 2017/085100, WO 2017/093019, WO 2017/178245, and WO 2017/211649.
- the weight ratio of component (A) to the sum of first component (B) and second component (B) may be from 100:1 to 1 :100, preferably from 50:1 to 1 :50, more preferably from 20:1 to 1 :40, even more preferably from 15:1 to 1 :30, still more preferably from 12:1 to 1 :25, or from 10:1 to 1 :20, or from 10:1 to 1 :10, or from 5:1 and 1 :15, or from 5:1 to 1 :5, or from 4:1 to 1 :4, or from 3:1 to 1 :10, or from 3:1 to 1 :3, or from 2:1 to 1 :5, or 1 :1.
- composition stands for the various mixtures or combinations of component A, wherein said component A is selected from a compound of formula (I), (l-A), or (l-B), and components (B) (including the above-defined embodiments), for example in a single “ready-mix” form, in a combined spray mixture composed from separate formulations of the single active ingredient components, such as a “tank-mix”, and in a combined use of the single active ingredients when applied in a sequential manner, i.e. one after the other with a reasonably short period, such as a few hours or days.
- the order of applying the component A, wherein said component A is selected from a compound of formula (I), (l-A), or (I- B), and component (B) is not essential for working the present invention.
- a method for controlling harmful fungi wherein the pest, their habitat, breeding grounds, their locus or the plants to be protected against pest attack, the soil or plant propagation material (preferably seed) are treated with a pesticidally effective amount of a compound of formula (I) according to the present invention.
- a method for controlling harmful fungi wherein the pest, their habitat, breeding grounds, their locus or the plants to be protected against pest attack, the soil or plant propagation material (preferably seed) are treated with a composition comprising a pesticidally effective amount of a compound of formula (I) according to the present invention.
- the compounds of the present invention and compositions comprising said compound of formula (I) are suitable for controlling fungal plant diseases.
- the compounds of the present invention are also important for controlling a multitude of fungi on various cultivated plants, such as bananas, cotton, vegetable species, for example cucumbers, beans, and cucurbits; cereals such as maize, wheat, rye, barley, rice, oats; grass coffee, potatoes, sweet potatoes, corn, fruit species, soybean, tobacco, tomatoes, grapevines, ornamental plants, sugar cane and also on a large number of seeds.
- the compounds of formula (I), (l-A), or (l-B) according to the present invention are used in soybean, cotton, and corn.
- compounds of formula (I), (l-A), or (l-B) according to the present invention are used on genetically modified plants selected from soybean, cotton, and corn.
- said genetically modified plants selected from soybean, cotton, and corn are Bt plants, like Bt soybeans, Bt cotton and Bt corn.
- the compounds of formula (I), (l-A), or (l-B) according to the present invention are particularly suitable for controlling cotton diseases caused by Alternaria spp. (Alternaria leaf spot), Cercospora spp. (Cercospora leaf spots), Corynespora cassiicola (leaf spots); Ascochyta gossypii (ascochyta blight of cotton); Fusarium wilt in cotton (Gossypium hirsutum L.) caused by Fusarium oxysporum f. sp..
- the compounds of formula (I), (l-A), or (l-B) according to the present invention are particularly suitable for controlling cotton diseases caused by Corynespora cassiicola (leaf spots).
- the compounds of formula (I), (l-A), or (l-B) according to the present invention are suitable for controlling the following fungal diseases on soybeans: Altemaria spp. (Alternaria leaf spot); Cercospora spp. (Cercospora leaf spots), e.g., C. sojina or C. kikuchii Colletotrichum (teleomorph: Glomerella) spp. (anthracnose), e.g., C. truncatum or C.
- gloeosporioides Corynespora cassiicola (leaf spots); Dematophora (teleomorph: Rosellinia) necatrix (root and stem rot); Diaporthe spp., e.g., D. phaseolorum (damping off); Fusarium (teleomorph: Gibberella) spp. (wilt, root or stem rot), e.g. F. tucumaniae and F. brasiliense each causing sudden death syndrome on soybeans; Macrophomina phaseolina (syn. phaseoli) (root and stem rot); Microsphaera diffusa (powdery mildew); Peronospora spp.
- Phialophora spp. e.g., P. gregata. stem rot
- Phomopsis spp. e.g., stem rot: P. phaseoli (teleomorph: Diaporthe phaseolorum); Pythium spp. (damping-off); Phytophthora spp. (wilt, root, leaf, fruit and stem root), e.g., P. megasperma, syn. P. sojae); Rhizoctonia spp., e.g., R.
- solani root and stem rot
- Sclerotinia spp. stem rot or white mold
- Septoria spp. e.g., S. glycines (brown spot);
- S. rolfsii syn. Athelia rolfsii
- Thielaviopsis spp. black root rot.
- the compounds of formula (I), (l-A), or (l-B) according to the present invention are suitable for controlling the soybean diseases caused by Corynespora cassiicola (leaf spots).
- the compounds of the invention or fungicidal compositions according to the present invention comprising said compounds may be used in controlling or preventing phytopathogenic diseases, especially phytopathogenic fungi (such as Corynespora cassiicola) on soybean plants.
- phytopathogenic fungi such as Corynespora cassiicola
- said genetically modified plants are selected from soybean, cotton, and maize.
- said genetically modified plants are BT plants, selected from Bt soybean, Bt cotton and Bt maize.
- said genetically modified plants are BT plants, selected from Bt soybean, and Bt cotton.
- Examples of commercially available genetically modified soybean plants which can preferably be treated according to the invention, include, but not limited to, commercially available products such as plant seeds Intacta®, lntacta®2, Intacta® Roundup ReadyTM 2 Pro (lntacta®RR2 PRO), lntacta®2 XtendTM, Cultivance, Conkesta Soybean, Conkesta Enlist E3TM Soybean, EnlistTM Soybean, Enlist E3TM Soybean, Roundup ReadyTM Soybean, Roundup ReadyTM 2 XtendTM, Genuity® Roundup ReadyTM 2 XtendTM, Genuity® Roundup Ready 2 YieldTM, Herbicide-tolerant Soybean line, Optimum GATTM, Liberty LinkTM Soybean, Vistive GoldTM, Verdeca HB4 Soybean, TreusTM, PlenishTM.
- commercially available products such as plant seeds Intacta®, lntacta®
- said Bt soybean plants are selected from Intacta RR2 PRO®, or Conkesta Enlist E3®.
- a method for controlling Corynespora cassiicola on soybean plants wherein said soybean plants are genetically modified soybean plants, preferably Bt soybean plants, characterized by comprising the step of contacting the plant, parts thereof, propagation material thereof, the pests, their food source, habitat, or breeding ground with a compound selected from ethyl 1-[[4-[(Z)-2-ethoxy-3,3,3-trifluoro-prop-1-enoxy]phenyl]methyl]pyrazole-4-carboxylate according to structure (I-
- soybean plants are genetically modified soybean plants, preferably Bt soybean plants, characterized by comprising the step of contacting the plant, parts thereof, propagation material thereof, the pests, their food source, habitat, or breeding ground with a compound of formula (l-A).
- soybean plants are genetically modified soybean plants, preferably Bt soybean plants, characterized by comprising the step of contacting the plant, parts thereof, propagation material thereof, the pests, their food source, habitat, or breeding ground with a compound of formula (l-B).
- the compounds of the invention or fungicidal compositions according to the present invention comprising said compounds may be used in controlling or preventing phytopathogenic diseases, especially phytopathogenic fungi (such as Corynespora cassiicola) on cotton plants.
- phytopathogenic fungi such as Corynespora cassiicola
- Examples of commercially available genetically modified cotton plants which can preferably be treated according to the invention, include, but not limited to, commercially available products such as plant seeds Bollgard®, Bollgard II TM Cotton, LibertyLink®, Roundup ReadyTM, WideStrikeTM Cotton, Bollgard® III x Roundup ReadyTM FlexTM, GlyTolTM, GlyTolTM Liberty LinkTM, GlytolTM x TwinlinkTM, GlyTol LibertyLink TwinLink® Plus, FibermaxTM Liberty LinkTM, BollgardTM Cotton, IngardTM, Roundup ReadyTM BollgardTM Cotton, Roundup ReadyTM FlexTM Cotton, Roundup ReadyTM FlexTM Bollgard IITM Cotton, TwinLinkTM Cotton, BXNTM Cotton.
- commercially available products such as plant seeds Bollgard®, Bollgard II TM Cotton, LibertyLink®, Roundup ReadyTM, WideStrikeTM Cotton, Bollgard® III x Roundup ReadyTM FlexTM, GlyTolTM, GlyTolTM Liberty LinkTM, GlytolTM x TwinlinkTM, Gly
- said Bt cotton plants are selected from WideStrikeTM Cotton, Bollgard® III x Roundup ReadyTM FlexTM, GlyTolTM Liberty LinkTM, GlyTol LibertyLink TwinLink® Plus, Bollgard II TM Cotton, BollgardTM Cotton IngardTM, Roundup ReadyTM BollgardTM Cotton, Roundup ReadyTM FlexTM Cotton, and TwinLinkTM Cotton.
- a method for controlling Corynespora cassiicola on cotton plants wherein said cotton plants are genetically modified cotton plants, preferably Bt cotton plants, characterized by comprising the step of contacting the plant, parts thereof, propagation material thereof, the pests, their food source, habitat, or breeding ground with a compound selected from ethyl 1 -[[4-[(Z)-2-ethoxy- 3,3,3-trifluoro-prop-1-enoxy]phenyl]methyl]pyrazole-4-carboxylate according to structure (l-A) ethyl 1-[[4-[[2-(trifluoromethyl)-1 ,3-dioxolan-2-yl]methoxy]phenyl]methyl]pyrazole-4-carboxylate according to structure (l-B)
- a method for controlling Corynespora cassiicola on cotton plants wherein said cotton plants are genetically modified cotton plants, preferably Bt cotton plants, characterized by comprising the step of contacting the plant, parts thereof, propagation material thereof, the pests, their food source, habitat, or breeding ground with a compound of formula (l-A).
- a method for controlling Corynespora cassiicola on cotton plants wherein said cotton plants are genetically modified cotton plants, preferably Bt cotton plants, characterized by comprising the step of contacting the plant, parts thereof, propagation material thereof, the pests, their food source, habitat, or breeding ground with a compound of formula (l-B).
- the compounds of formula (I), (l-A), or (l-B) according to the present invention, or fungicidal compositions comprising said compounds according to the present invention may be used in controlling or preventing phytopathogenic diseases, especially phytopathogenic fungi (such as Corynespora cassiicola) on soybean plants.
- phytopathogenic diseases especially phytopathogenic fungi (such as Corynespora cassiicola) on soybean plants.
- the compounds of formula (I), (l-A), or (l-B) according to the present invention, or fungicidal compositions comprising said compounds according to the present invention may be used in controlling or preventing phytopathogenic diseases, especially phytopathogenic fungi (such as Corynespora cassiicola) on genetically modified soybean plants, preferably Bt soybean plants.
- phytopathogenic diseases especially phytopathogenic fungi (such as Corynespora cassiicola) on genetically modified soybean plants, preferably Bt soybean plants.
- the compounds of formula (I), (l-A), or (l-B) according to the present invention, or fungicidal compositions comprising said compounds according to the present invention may be used in controlling or preventing phytopathogenic diseases, especially phytopathogenic fungi (such as Corynespora cassiicola) on cotton plants.
- phytopathogenic fungi such as Corynespora cassiicola
- the compounds of formula (I), (l-A), or (l-B) according to the present invention, or fungicidal compositions comprising said compounds according to the present invention may be used in controlling or preventing phytopathogenic diseases, especially phytopathogenic fungi (such as Corynespora cassiicola) on genetically modified cotton plants, preferably Bt cotton plants.
- phytopathogenic diseases especially phytopathogenic fungi (such as Corynespora cassiicola) on genetically modified cotton plants, preferably Bt cotton plants.
- fungicidal compositions according to the present invention comprising a compound of formula (I), (l-A), or (l-B) when used in controlling or preventing phytopathogenic diseases, especially phytopathogenic fungi (such as Corynespora cassiicola) on plants selected from soybean and cotton (in particular any of the transgenic plants as described above), may display a synergistic interaction between the active ingredients.
- phytopathogenic fungi such as Corynespora cassiicola
- Transgenic soybean plants expressing toxins for example insecticidal proteins such as delta-endotoxins, e.g., CrylAc (CrylAc Bt protein). Accordingly, this may include transgenic soybean plants comprising event MON87701 (disclosed in W02009/064652), event MON87701 x MON89788 (disclosed in WO2014/170327, e.g.
- event MON87751 (disclosed in WO2014/201235)
- event DAS-44406-6 e.g., commercially available as Enlist E3TM, DAS-44406-6, disclosed in WO2012/075426
- event DAS-81419-2 (described in WO2013/016527, e.g., commercially available as ConkestaTM soybean); event DAS-81419-2 x DAS-44406-6 (e.g., commercially available as ConkestaTM Enlist E3TM Soybean).
- transgenic events in transgenic soybean plants include event DAS-44406-6/pDAB8264.44.06.1 (soybean, herbicide-tolerance, disclosed in WO2012/075426); event DAS-81419-2 (described in WO2013/016527 (e.g., commercially available as aka ConkestaTM soybean, ConkestaTM Enlist E3TM soybean, DAS-81419-2 x DAS-44406-6); event DAS-14536-7/pDAB8291 .45.36.2 (soybean, herbicide-tolerance, disclosed in WO2012/075429); DAS-68416-4 (soybean, herbicide-tolerance, ATCC Accession No.
- PTA-10442 disclosed in WO2011/066384, WO2011/066360
- event DP-305423-1 (soybean, quality mark, disclosed in W02008/054747, e.g. commercially available as TreusTM, PlenishTM, Plenish® High Oleic Soybeans)
- event DP-356043-5 (soybean, herbicide-tolerance, deposited as ATCC PTA- 8287, disclosed in W02008/002872, e.g.
- event FG72 (soybean, herbicide-tolerance, disclosed in WO2011/063413); event LL27 (soybean, herbicide-tolerance, disclosed in W02006/108674); event LL55 (soybean, herbicide-tolerance, disclosed in WO 2006/108675); event EE- GM3/FG72 (soybean, herbicide-tolerance) optionally stacked with event EE-GM1/LL27 or event EE-GM2/LL55 (disclosed in WO2011/063413); event MON87701 (soybean, insect control, disclosed in W02009/064652, WO2014/170327); event MON87701 x MON89788 (disclosed in WO2014/170327, e.g.
- event MON87705 (soybean, improved fatty acid profile, herbicide-tolerance, disclosed in W02010/037016 or US20100080887A, e.g. commercially available as Vistive GoldTM); event MON87751 (lepidopteran-resistant, ATCC accession no. PTA-120166. disclosed in WO2014/201235); event MON87751xMON87701xMON89788xMON87708 (commercially available as Intacta2 Xtend®); event MON87708 (soybean, herbicide-tolerance, disclosed in WO2011/034704, e.g.
- event MON- 04032-6 event code: GTS 40-3-2, http://www.agbios.com/static/cropdb/LONG-GTS-40-3-2-printer.html, e.g. commercially available as Roundup Ready® soybean
- event HB4 event code IND-00410-5, US2022/009011 , e.g., commercially available as Verdeca HB4 Soybean.
- transgenic events in transgenic soybean plants which can preferably be treated according to the invention, include event A2704-12 (glufosinate tolerance, disclosed in W02006/108647, e.g., commercially available as Liberty LinkTM soybean), event A5547-127 (phosphinothricin tolerant, disclosed in W02006/108675, US8952142B2, e.g., commercially available as Liberty LinkTM soybean); A5547-35 (event code: ACS-GM008-6, gene: pat, e.g. commercially available as Liberty LinkTM soybean), event MON89788 (soybean, herbicide-tolerance, disclosed in W02006/130436, e.g.
- Genuity® Roundup Ready 2 YieldTM DP-305423-1 (soybean, quality mark, disclosed in W02008/054747, e.g., commercially available as TreusTM, PlenishTM, Plenish® High Oleic Soybeans); event MON87701 (soybean, insect resistant, disclosed in W02009/064652); event MON87701 x MON89788 (disclosed in WO2014/170327, e.g. commercially available as Intacta RR2 PRO® soybean); event MON87705 (soybean, improved fatty acid profile, herbicide-tolerance, disclosed in WO2010/037016 or US20100080887A, e.g.
- event FG72 (soybean, herbicide-tolerance, disclosed in WO2011/063413); evet FG72xA5547-127 (e.g. commercially available as LibertyLink® GT27TM); event SYHT0H2/SYN-000H2-5 (soybean, herbicide-tolerance, disclosed in WO2012/082548); event DAS-81419-2 (described in WO2013/016527, e.g., commercially available as ConkestaTM soybean); event DAS-44406-6 (disclosed in WO2012/075426, e.g., commercially available as Enlist E3TM), or event DAS-81419-2 (described in WO2013/016527, e.g., commercially available as ConkestaTM soybean); DAS81419-2xDAS4406 (e.g., commercially available as ConkestaTM Enlist E3TM soybean); event DP305423x GT
- event DP-356043-5 sibean, herbicide-tolerance, deposited as ATCC PTA-8287, disclosed in W02008/002872, e.g. commercially available as Optimum GATTM
- event MON-04032-6 event code: GTS 40-3-2, http://www.agbios.com/static/cropdb/LONG-GTS-4Q-3-2-printer.html, e.g. commercially available as Roundup Ready® soybean).
- transgenic events are provided by the United States Department of Agriculture's (USDA) Animal and Plant Health Inspection Service (APHIS) and can be found on their website on the World Wide Web at aphis.usda.gov.
- USDA United States Department of Agriculture's
- APIS Animal and Plant Health Inspection Service
- Transgenic soybean events comprising herbicide tolerance genes are for example, but not excluding others, GTS 40-3-2, MON87705, MON87708, MON87712, MON87769, MON89788, A2704-12, A2704-21 , A5547- 127, A5547-35, DP356043, DAS44406-6, DAS68416-4, DAS81419-2, GU262, SYHT0H2, W62, W98, FG72 and CV127.
- a compound of formula (I) (l-A), or (l-B) for controlling phytopathogenic fungi in genetically modified soybean plants, wherein said transgenic soybean events comprising herbicide tolerance genes are for example, but not excluding others, GTS 40-3-2, MON87705, MON87708, MON87712, MON87769, MON89788, A2704-12, A2704-21 , A5547-127, A5547-35, DP356043, DAS44406-6, DAS68416-4, DAS81419-2, GU262, SYHT0H2, W62, W98, FG72 and CV127.
- said transgenic soybean events comprising herbicide tolerance genes are for example, but not excluding others, GTS 40-3-2, MON87705, MON87708, MON87712, MON87769, MON89788, A2704-12, A2704-21 , A5547-127, A5547-35, DP356043, DAS44406-6, DAS6841
- Transgenic cotton events comprising herbicide tolerance genes are for example, but not excluding others, 19- 51 a, 31707, 42317, 81910, 281 -24-236, 3006-210-23, BXN1021 1 , BXN 10215, BXN10222, BXN 10224, MON1445, MON1698, MON88701 , MON88913, GHB1 19, GHB614, LLCotton25, T303-3 and T304-40.
- Transgenic cotton events comprising genes for insecticidal proteins are for example, but not excluding others, SGK321 , MON531 , MON757, MON1076, MON15985, 31707, 31803, 31807, 31808, 42317, BNLA-601 , Eventl, COT67B, COT102, T303-3, T304-40, GFM Cry1A, GK12, MLS 9124, 281 -24-236, 3006-210-23, GHB1 19 and SGK321. Increased yield has been created by increasing ear biomass using the transgene athb17, being present in corn event MON87403, or by enhancing photosynthesis using the transgene bbx32, being present in the soybean event MON87712.
- Transgenic cotton events comprising glufosinate tolerance genes are for example, but not excluding others, 3006-210-23 x 281-24-236 x MON 1445 (event code: DAS-21023-5 x DAS- 24236-5 x MON-01445-2, gene: bar, e.g., commercially available as WideStrikeTM Roundup ReadyTM Cotton), 3006-210-23 x 281-24-236 x MON88913 (event code: DAS-21023-5 x DAS-24236-5 x MON-88913-8, gene: bar, e.g., commercially available as WidestrikeTM Roundup Ready FlexTM Cotton), 3006-210-23 x 281-24-236 x MON88913 x COT102 (event code: DAS- 21023-5 x DAS-24236-5 x MON-88913-8 x SYN-IR102-7, gene: pat, e.g., commercially available as WidestrikeTM x Roundup Ready FlexTM x
- a method of controlling or preventing infestation of plants by the phytopathogenic microorganism Corynespora cassiicola comprising applying to the phytopathogen, to the locus of the phytopathogen, or to a plant susceptible to attack by the phytopathogen, or to a propagation material thereof, a compound according to formula (I) (l-A), or (l-B) according to the invention, wherein the compound according to formula (I) (l-A), or (l-B) is applied in the field.
- the terms “applied in the field”, “field-applied”, “field-grown” and “field cultivation” refer broadly to the application of the pesticidal compounds or compositions directly onto plants growing in an open field or traditional agricultural setting, as opposed to “liquid culture application” which typically involves the use of pesticidal compounds and compositions in a controlled, hydroponic, or soilless environment where plants are grown in nutrient-rich aqueous solutions, such as for the purpose of scientific research.
- the compounds and composition as defined herein have surprisingly proven to be particularly effective for field application as compared to liquid culture applications.
- the compound according to formula (I) (l-A), or (l-B) according to the invention is applied to a field-grown plant susceptible to attack by the phytopathogen by means of foliar application, soil application, in furrow application or drench application.
- the compound according to formula (I) (l-A), or (l-B) according to the invention is applied to the plant propagation material for the purpose of field cultivation of a plant susceptible to attack by the phytopathogen.
- the compound of formula (I), (l-A), or (l-B) according to the invention may be used in unmodified form or, preferably, together with the adjuvants conventionally employed in the art of formulation. To this end they may be conveniently formulated in known manner to emulsifiable concentrates, coatable pastes, directly sprayable or dilutable solutions or suspensions, dilute emulsions, wettable powders, soluble powders, dusts, granulates, and also encapsulations e.g., in polymeric substances. As with the type of the compositions, the methods of application, such as spraying, atomising, dusting, scattering, coating, or pouring, are chosen in accordance with the intended objectives and the prevailing circumstances. The compositions may also contain further adjuvants such as stabilizers, antifoams, viscosity regulators, binders or tackifiers as well as fertilizers, micronutrient donors or other formulations for obtaining special effects.
- adjuvants conventionally
- Suitable carriers and adjuvants can be solid or liquid and are substances useful in formulation technology, e.g., natural or regenerated mineral substances, solvents, dispersants, wetting agents, tackifiers, thickeners, binders, or fertilizers.
- Such carriers are for example described in WO 1997/33890.
- Suspension concentrates are aqueous formulations in which finely divided solid particles of the active compound are suspended. Such formulations include anti-settling agents and dispersing agents and may further include a wetting agent to enhance activity as well an anti-foam and a crystal growth inhibitor. In use, these concentrates are diluted in water and normally applied as a spray to the area to be treated. The amount of active ingredient may range from 0.5% to 95% of the concentrate.
- Wettable powders are in the form of finely divided particles which disperse readily in water or other liquid carriers.
- the particles contain the active ingredient retained in a solid matrix.
- Typical solid matrices include fuller’s earth, kaolin clays, silicas and other readily wet organic or inorganic solids. Wettable powders normally contain from 5% to 95% of the active ingredient plus a small amount of wetting, dispersing or emulsifying agent.
- Emulsifiable concentrates are homogeneous liquid compositions dispersible in water or other liquid and may consist entirely of the active compound with a liquid or solid emulsifying agent, or may also contain a liquid carrier, such as xylene, heavy aromatic naphthas, isophorone and other non-volatile organic solvents. In use, these concentrates are dispersed in water or other liquid and normally applied as a spray to the area to be treated. The amount of active ingredient may range from 0.5% to 95% of the concentrate.
- Granular formulations include both extrudates and relatively coarse particles and are usually applied without dilution to the area in which treatment is required.
- Typical carriers for granular formulations include sand, fuller’s earth, attapulgite clay, bentonite clays, montmorillonite clay, vermiculite, perlite, calcium carbonate, brick, pumice, pyrophyllite, kaolin, dolomite, plaster, wood flour, ground corn cobs, ground peanut hulls, sugars, sodium chloride, sodium sulphate, sodium silicate, sodium borate, magnesia, mica, iron oxide, zinc oxide, titanium oxide, antimony oxide, cryolite, gypsum, diatomaceous earth, calcium sulphate and other organic or inorganic materials which absorb or which can be coated with the active compound.
- Granular formulations normally contain 5% to 25% of active ingredients which may include surface-active agents such as heavy aromatic naphthas, kerosene and other petroleum fractions, or vegetable oils
- Dusts are free-flowing admixtures of the active ingredient with finely divided solids such as talc, clays, flours, and other organic and inorganic solids which act as dispersants and carriers.
- the active ingredients can also be contained in microcapsules.
- Microcapsules contain the active ingredients in a porous carrier. This enables the active ingredients to be released into the environment in controlled amounts (e.g., slow-release).
- Microcapsules usually have a diameter of from 0.1 to 500 microns. They contain active ingredients in an amount of about from 25 to 95 % by weight of the capsule weight.
- the active ingredients can be in the form of a monolithic solid, in the form of fine particles in solid or liquid dispersion or in the form of a suitable solution.
- the encapsulating membranes can comprise, for example, natural or synthetic rubbers, cellulose, styrene/butadiene copolymers, polyacrylonitrile, polyacrylate, polyesters, polyamides, polyureas, polyurethane, or chemically modified polymers, and starch xanthates, or other polymers that are known to the person skilled in the art.
- very fine microcapsules can be formed in which the active ingredient is contained in the form of finely divided particles in a solid matrix of base substance, but the microcapsules are not themselves encapsulated.
- Microcapsules are typically droplets or granules of the active ingredient enclosed in an inert porous shell which allows escape of the enclosed material to the surroundings at controlled rates.
- Encapsulated droplets are typically 1 to 50 microns in diameter.
- the enclosed liquid typically constitutes 50 to 95% of the weight of the capsule and may include solvent in addition to the active compound.
- Encapsulated granules are generally porous granules with porous membranes sealing the granule pore openings, retaining the active species in liquid form inside the granule pores.
- Granules typically range from 1 millimetre to 1 centimetre and preferably 1 to 2 millimetres in diameter. Granules are formed by extrusion, agglomeration or prilling, or are naturally occurring.
- Shell or membrane materials include natural and synthetic rubbers, cellulosic materials, styrene-butadiene copolymers, polyacrylonitriles, polyacrylates, polyesters, polyamides, polyureas, polyurethanes and starch xanthates.
- Other useful formulations for agrochemical applications include simple solutions of the active ingredient in a solvent in which it is completely soluble at the desired concentration, such as acetone, alkylated naphthalenes, xylene and other organic solvents. Pressurised sprayers, wherein the active ingredient is dispersed in finely divided form because of vaporisation of a low boiling dispersant solvent carrier, may also be used.
- Suitable agricultural adjuvants and carriers that are useful in formulating the compositions of the invention in the formulation types described above are well known to a person skilled in the art.
- Liquid carriers that can be employed include, for example, water, toluene, xylene, petroleum naphtha, crop oil, acetone, methyl ethyl ketone, cyclohexanone, acetic anhydride, acetonitrile, acetophenone, amyl acetate, 2- butanone, chlorobenzene, cyclohexane, cyclohexanol, alkyl acetates, diacetonalcohol, 1 ,2-dichloropropane, diethanolamine, p diethylbenzene, diethylene glycol, diethylene glycol abietate, diethylene glycol butyl ether, diethylene glycol ethyl ether, diethylene glycol methyl ether, N,N-dimethyl formamide, dimethyl sulfoxide, 1 ,4- dioxane, dipropylene glycol, dipropylene glycol methyl ether, dipropylene glycol dibenz
- Suitable solid carriers include, for example, talc, titanium dioxide, pyrophyllite clay, silica, attapulgite clay, kieselguhr, chalk, diatomaxeous earth, lime, calcium carbonate, bentonite clay, fuller’s earth, cotton seed hulls, wheat flour, soybean flour, pumice, wood flour, walnut shell flour and lignin.
- a broad range of surface-active agents are advantageously employed in both said liquid and solid compositions, especially those designed to be diluted with carrier before application.
- These agents when used, normally comprise from 0.1 % to 15% by weight of the formulation. They can be anionic, cationic, nonionic or polymeric in character and can be employed as emulsifying agents, wetting agents, suspending agents or for other purposes.
- Typical surface-active agents include salts of alkyl sulfates, such as diethanolammonium lauryl sulphate; alkylarylsulfonate salts, such as calcium dodecylbenzenesulfonate; alkylphenol-alkylene oxide addition products, such as nonylphenol-C.sub.
- alcohol-alkylene oxide addition products such as tridecyl alcohol-C.sub. 16 ethoxylate
- soaps such as sodium stearate
- alkylnaphthalenesulfonate salts such as sodium dibutylnaphthalenesulfonate
- dialkyl esters of sulfosuccinate salts such as sodium di(2- ethylhexyl) sulfosuccinate
- sorbitol esters such as sorbitol oleate
- quaternary amines such as lauryl trimethylammonium chloride
- polyethylene glycol esters of fatty acids such as polyethylene glycol stearate
- salts of mono and dialkyl phosphate esters such as mono and dialkyl phosphate esters.
- adjuvants commonly utilized in agricultural compositions include crystallisation inhibitors, viscosity modifiers, suspending agents, spray droplet modifiers, pigments, antioxidants, foaming agents, anti-foaming agents, light-blocking agents, compatibilizing agents, antifoam agents, sequestering agents, neutralising agents and buffers, corrosion inhibitors, dyes, odorants, spreading agents, penetration aids, micronutrients, emollients, lubricants and sticking agents.
- the compounds of formula (I), (l-A), or (l-B) according to the invention may be used in the form of a pesticidal composition further comprising one or more adjuvants selected from stabilizers, viscosity regulators, binders, tackifiers, fertilizers, crystallisation inhibitors, viscosity modifiers, suspending agents, spray droplet modifiers, pigments, antioxidants, foaming agents, anti-foaming agents, light-blocking agents, compatibilising agents, antifoam agents, sequestering agents, neutralising agents and buffers, corrosion inhibitors, dyes, odorants, spreading agents, penetration aids, micronutrients, emollients, lubricants, and sticking agents.
- adjuvants selected from stabilizers, viscosity regulators, binders, tackifiers, fertilizers, crystallisation inhibitors, viscosity modifiers, suspending agents, spray droplet modifiers, pigments, antioxidants, foaming agents, anti-foaming agents,
- the pesticidal composition may also comprise one or more agents that act as a carrier or as a diluent of the active ingredients, or both.
- the agent acting as a carrier or diluent, or both, hereinafter called “carrier or diluent”, may be liquid or solid.
- the compounds of formula (I), (l-A), or (l-B) according to the invention may be used in the form of a pesticidal composition further comprising one or more liquid carriers or diluents selected from xylene, heavy aromatic naphthas, isophorone, water, toluene, petroleum naphtha, crop oil, acetone, methyl ethyl ketone, cyclohexanone, acetic anhydride, acetonitrile, acetophenone, amyl acetate, 2-butanone, chlorobenzene, cyclohexane, cyclohexanol, alkyl acetates, diacetonalcohol, 1 ,2-dichloropropane, diethanolamine, p diethylbenzene, diethylene glycol, diethylene glycol abietate, diethylene glycol butyl ether, diethylene glycol ethyl ether, diethylene glycol methyl ether,
- the compounds of formula (I), (l-A), or (l-B) according to the invention may be used in the form of a pesticidal composition further comprising one or more solid diluents or carriers selected from talc, titanium dioxide, pyrophyllite clay, silica, attapulgite clay, kieselguhr, chalk, diatomaceous earth, lime, calcium carbonate, bentonite clay, Fuller's earth, cotton seed hulls, wheat flour, soybean flour, pumice, wood flour, walnut shell flour, and lignin.
- solid diluents or carriers selected from talc, titanium dioxide, pyrophyllite clay, silica, attapulgite clay, kieselguhr, chalk, diatomaceous earth, lime, calcium carbonate, bentonite clay, Fuller's earth, cotton seed hulls, wheat flour, soybean flour, pumice, wood flour, walnut shell flour, and lignin.
- the compounds of formula (I), (l-A), or (l-B) according to the invention may be used in the form of a pesticidal composition further comprising one or more formulation auxiliaries selected from unepoxidized or epoxidized vegetable oils, preservatives, inorganic compounds.
- biocidal active ingredients or compositions may be combined with the compounds or compositions of the invention and used in the methods of the invention and applied simultaneously or sequentially with the compounds or compositions of the invention. When applied simultaneously, these further active ingredients may be formulated together with the compounds or compositions of the invention or mixed in, for example, the spray tank. These further biocidal active ingredients may be fungicides, herbicides, insecticides, bactericides, acaricides, nematicides and/or plant growth regulators.
- Pesticidal agents are referred to herein using their common name are known, for example, from “The Pesticide Manual”, 15th Ed., British Crop Protection Council 2009.
- compositions of the invention may also be applied with one or more systemically acquired resistance inducers (“SAR” inducer).
- SAR inducers are known and described in, for example, United States Patent No. US 6,919,298 and include, for example, salicylates and the commercial SAR inducer acibenzolar- S-methyl.
- the compounds of formula (I), (l-A), or (l-B) according to the invention are normally used in the form of agrochemical compositions and can be applied to the crop area or plant to be treated, simultaneously or in succession with further compounds.
- further compounds can be e.g., fertilizers or micronutrient donors or other preparations, which influence the growth of plants. They can also be selective herbicides or non- selective herbicides as well as insecticides, fungicides, bactericides, nematicides, molluscicides or mixtures of several of these preparations, if desired together with further carriers, surfactants or application promoting adjuvants customarily employed in the art of formulation.
- the compounds of formula (I), (l-A), or (l-B) according to the invention may be used in the form of (fungicidal) compositions for controlling or protecting against phytopathogenic microorganisms, comprising as active ingredient a compound as defined herein, in free form or in agrochemical usable salt form, and at least one of the above-mentioned adjuvants, and preferably at least one formulation auxiliary, diluent and/or carrier.
- the invention therefore provides a composition, preferably a fungicidal composition, comprising a compound of formula (I), (l-A), or (l-B) according to the invention, an agriculturally acceptable carrier and optionally an adjuvant or an adjuvant and one or more auxiliaries, diluents and/or carriers.
- An agricultural acceptable carrier is for example a carrier that is suitable for agricultural use.
- Agricultural carriers are well known in the art.
- said composition may comprise at least one or more pesticidal-active compounds, for example an additional fungicidal active ingredient in addition to a compound of formula (I), (l-A), or (l-B).
- the compound of formula (I), (l-A), or (l-B) according to the invention may be the sole active ingredient of a composition or it may be admixed with one or more additional active ingredients such as a pesticide, including fungicides, herbicide, insecticide, synergist, or plant growth regulator where appropriate.
- additional active ingredient may, in some cases, result in unexpected synergistic activities.
- Suitable additional active ingredients include the following: acycloamino acid fungicides, aliphatic nitrogen fungicides, amide fungicides, anilide fungicides, antibiotic fungicides, aromatic fungicides, arsenical fungicides, aryl phenyl ketone fungicides, benzamide fungicides, benzanilide fungicides, benzimidazole fungicides, benzothiazole fungicides, botanical fungicides, bridged diphenyl fungicides, carbamate fungicides, carbanilate fungicides, conazole fungicides, copper fungicides, dicarboximide fungicides, dinitrophenol fungicides, dithiocarbamate fungicides, dithiolane fungicides, furamide fungicides, furanilide fungicides, hydrazide fungicides, imidazole fungicides, mercury fungicides, morpholine fung
- a further aspect of the present invention is a method of controlling diseases on useful plants or on propagation material thereof caused by phytopathogens, which comprises applying to the useful plants, the locus thereof or propagation material thereof a composition according to the invention.
- a method which comprises applying to the useful plants or to the locus thereof a composition according to the invention, more preferably to the useful plants.
- a method which comprises applying to the propagation material of the useful plants a composition according to the invention.
- composition refers to the various mixtures or combinations of components (A) and (B), for example in a single “ready-mix” form, in a combined spray mixture composed from separate formulations of the single active ingredient components, such as a “tank-mix”, and in a combined use of the single active ingredients when applied in a sequential manner, i.e. one after the other with a reasonably short period, such as a few hours or days.
- the order of applying the components (A) and (B) is not essential for working the present invention.
- compositions according to the invention are effective against harmful microorganisms, such as microorganisms, that cause phytopathogenic diseases, in particular against phytopathogenic fungi and bacteria.
- the formulation comprising a composition according to the invention may comprise of from 0.01 to 90% by weight of the composition comprising components (A) and (B), and of from 0 to 20% of an agriculturally acceptable surfactant.
- the formulation further comprises other active agents, in particular microbiocides and pesticides, more generally.
- the formulation further comprises of from 10 to 99.99% solid or liquid formulation inerts, conservatives and/or adjuvants.
- the present invention preferably also relates to a concentrated composition for dilution by the user, comprising a composition according or a formulation according to the invention, comprising of from 2 to 80% by weight, preferably between 5 and 70% by weight, of active agents comprising at least a composition comprising (A) and (B), and optionally, other active agents.
- the present invention preferably also relates to a seed dressing formulation for application to plant propagation materials, comprising a composition according to the invention, and further comprising a diluent.
- the seed dressing formulation is in the form of an aqueous suspension or in a dry powder form having good adherence to the plant propagation materials.
- the seed dressing formulation may comprise the active agents in an encapsulated form, preferably a slow-release capsules and/or microcapsules.
- the present invention preferably also relates to a method of combating and controlling phytopathogens, comprising applying a fungicidally effective amount of a composition according to the invention to a pest, a locus of pest, or to a plant susceptible to attack by a pest, with the exception of a method for treatment of the human or animal body by surgery or therapy and diagnostic methods practiced on the human or animal body.
- the methods, compositions and uses according to any one of embodiments according to the invention are preferably for controlling or preventing infestation of the crop by the phytopathogenic microorganisms Corynespora cassiicola, that are resistant to other fungicides.
- Corynespora cassiicola that are "resistant" to a particular fungicide refer e.g., to strains of Corynespora cassiicola fungi that are less sensitive to that fungicide compared to the expected sensitivity of the same species of Corynespora cassiicola fungi.
- the expected sensitivity can be measured using e.g., a strain that has not previously been exposed to the fungicide.
- an “effective” amount herein refers to an amount of the active ingredient that shows sufficient biocidal activity, e.g., at least 10 %, more preferably at least 20%, yet more preferably at least 50%, and again more preferably at least 70 % effectiveness, compared to the blind test.
- the composition according to the invention preferably comprise at least 0.01 ppm, more preferably at last 0.025 ppm of active ingredient, more preferably at least for example 6 ppm, 3 ppm, 2.2 ppm, 1 .5 ppm, 0.8 ppm, 0.74 ppm, 0.25 ppm, 0.2 ppm, or 0.082 ppm as applied.
- Application according to the methods or uses according to any one of embodiments according to the invention is preferably to a crop of plants, the locus thereof or propagation material thereof.
- Preferably application is to the phytopathogen, to the locus ofthe phytopathogen, orto a plant susceptible to attack by the phytopathogen, or to a propagation material thereof.
- Application of the compounds as defined in any one of embodiments 1 to 13 can be performed according to any of the usual modes of application, e.g., foliar, drench, soil, in furrow etc.
- the compounds or pesticidal compositions as defined herein are applied in the field to the foliage of the plant (foliar application), thus allowing to select frequency and rate of application to match the danger of infestation with the pest in question.
- the active ingredient can reach the plants via the root system (systemic action), by drenching the locus of the plants in the field with a liquid composition or by incorporating the active ingredient in solid form into the locus of the plants in the field, for example into the soil or furrow, such as in the form of granules. In the case of paddy rice crops, such granules can be metered into the flooded paddy-field.
- the compounds as defined in any one of embodiments according to the invention are suitable for use on any plant, including those that have been genetically modified to be resistant to active ingredients such as herbicides, or to produce biologically active compounds that control infestation by plant pests.
- a compound as defined in any one of embodiments according to the invention is used in the form of a composition (e.g., formulation) containing a carrier.
- a compound as defined in any one of embodiments according to the invention and compositions thereof can be used in various forms such as aerosol dispenser, capsule suspension, cold fogging concentrate, dustable powder, emulsifiable concentrate, emulsion oil in water, emulsion water in oil, encapsulated granule, fine granule, flowable concentrate for seed treatment, gas (under pressure), gas generating product, granule, hot fogging concentrate, macrogranule, microgranule, oil dispersible powder, oil miscible flowable concentrate, oil miscible liquid, paste, plant rodlet, powder for dry seed treatment, seed coated with a pesticide, soluble concentrate, soluble powder, solution for seed treatment, suspension concentrate (flowable concentrate), ultra-low volume (ulv) liquid, ultra-low volume (ulv) suspension, water dispersible granules or tablets, water dispers
- compositions of this invention can be mixed with one or more further pesticides including further fungicides, insecticides, nematicides, bactericides, acaricides, growth regulators, chemosterilants, semiochemicals, repellents, attractants, pheromones, feeding stimulants or other biologically active compounds to form a multi-component pesticide giving an even broader spectrum of agricultural protection.
- further pesticides including further fungicides, insecticides, nematicides, bactericides, acaricides, growth regulators, chemosterilants, semiochemicals, repellents, attractants, pheromones, feeding stimulants or other biologically active compounds to form a multi-component pesticide giving an even broader spectrum of agricultural protection.
- a formulation typically comprises a liquid or solid carrier and optionally one or more customary formulation auxiliaries, which may be solid or liquid auxiliaries, for example unepoxidized or epoxidized vegetable oils (for example epoxidized coconut oil, rapeseed oil or soya oil), antifoams, for example silicone oil, preservatives, clays, inorganic compounds, viscosity regulators, surfactant, binders and/or tackifiers.
- auxiliaries for example unepoxidized or epoxidized vegetable oils (for example epoxidized coconut oil, rapeseed oil or soya oil), antifoams, for example silicone oil, preservatives, clays, inorganic compounds, viscosity regulators, surfactant, binders and/or tackifiers.
- composition may also further comprise a fertilizer, a micronutrient donor or other preparations which influence the growth of plants as well as comprising a combination containing the compound of the invention with one or more other biologically active agents, such as bactericides, fungicides, nematicides, plant activators, acaricides, and insecticides.
- a fertilizer such as bactericides, fungicides, nematicides, plant activators, acaricides, and insecticides.
- compositions are prepared in a manner known perse, in the absence of auxiliaries for example by grinding, screening and/or compressing a solid compound of the present invention and in the presence of at least one auxiliary for example by intimately mixing and/or grinding the compound of the present invention with the auxiliary (auxiliaries).
- auxiliaries for example by grinding, screening and/or compressing a solid compound of the present invention
- at least one auxiliary for example by intimately mixing and/or grinding the compound of the present invention with the auxiliary (auxiliaries).
- the grinding/milling of the compounds is to ensure specific particle size.
- compositions for use in agriculture are emulsifiable concentrates, suspension concentrates, microemulsions, oil dispersibles, directly sprayable or dilutable solutions, spreadable pastes, dilute emulsions, soluble powders, dispersible powders, wettable powders, dusts, granules or encapsulations in polymeric substances, which comprise - at least - a compound of formula (I) as defined herein and the type of composition is to be selected to suit the intended aims and the prevailing circumstances.
- the compositions comprise 0.1 to 99 %, especially 0.1 to 95 %, of a compound (A), and optionally compound (B), as defined in any one of embodiments according to the invention and 1 to 99.9 %, especially 5 to 99.9 %, of at least one solid or liquid carrier, it being possible as a rule for 0 to 25 %, especially 0.1 to 20%, of the composition to be surfactants (% in each case meaning percent by weight).
- surfactants % in each case meaning percent by weight.
- the weight ratio of component (A) to component (B) is from 2000 : 1 to 1 : 1000.
- the weight ratio of component (A) to component (B) is preferably from 100 : 1 to 1 : 100; more preferably from 20 : 1 to 1 : 50, yet more preferably from 12 : 1 to 1 : 25; yet more preferably from 10 : 1 to 1 : 10, again more preferably from 5 : 1 to 1 : 15; and most preferably from 2 :1 to 1 : 5.
- compositions according to the invention can also have further surprising advantageous properties.
- advantageous properties are: more advantageous degradability; improved toxicological and/or ecotoxicological behaviour; or improved characteristics of the useful plants including: emergence, crop yields, more developed root system, tillering increase, increase in plant height, bigger leaf blade, less dead basal leaves, stronger tillers, greener leaf colour, less fertilizers needed, less seeds needed, more productive tillers, earlier flowering, early grain maturity, less plant verse (lodging), increased shoot growth, improved plant vigour, and early germination.
- Additional beneficial effects can be the suppression or reduction of development of resistance against a certain active ingredient, by combinations that may have one or more different modes of action may in particular be beneficial.
- compositions according to the invention have a systemic action and can be used as foliar, soil and seed treatment fungicides.
- compositions according to the invention it is possible to inhibit or destroy the phytopathogenic microorganisms which occur in plants or in parts of plants (fruit, blossoms, leaves, stems, tubers, roots) in different useful plants, while at the same time the parts of plants which grow later are also protected from attack by phytopathogenic microorganisms.
- the compositions according to the invention can be applied to the phytopathogenic microorganisms, the useful plants, the locus thereof, the propagation material thereof, storage goods or technical materials threatened by microorganism attack.
- compositions according to the invention may be applied before or after infection of the useful plants, the propagation material thereof, storage goods or technical materials by the microorganisms.
- compositions according to the invention to be applied will depend on various factors, such as the compounds employed; the subject of the treatment, such as, for example plants, soil or seeds; the type of treatment, such as, for example spraying, dusting or seed dressing; the purpose of the treatment, such as, for example prophylactic or therapeutic; the type of fungi to be controlled or the application time.
- component (A) When applied to the useful plants component (A) is typically applied at a rate of 5 to 2000 g a.i./ha, particularly 10 to 1000 g a.i./ha, e.g., 50, 75, 100 or 200 g a.i./ha, preferably in association with 1 to 5000 g a.i./ha, particularly 2 to 2000 g a.i./ha, e.g., 100, 250, 500, 800, 1000, 1500 g a.i./ha of component (B).
- the compounds as defined in any one of embodiments according to the invention are preferably used for pest control at rates of 1 to 500 g/ha, preferably 50-300 g/ha.
- the compounds of formula (I) as defined in any one of embodiments according to the invention are used for pest control at rates of 1 to 500 g/ha, preferably 10 to 400 g/ha, more preferably 15 to 300 g/ha, most preferably 10 to 100 g/ha, such as 10, 15, 20, 25, 30, 35, 40, 45, 50, 55, 60, or 65 g a.i /ha.
- the compounds of formula (I) and pesticidal compositions comprising these compounds as defined in any one of embodiments according to the invention are applied at rates of 10 to 100 g a.i./ha, preferably 15 to 65 g a.i./ha, more preferably at rates of 20 to 60 g a.i./ha, such as 10, 15, 20, 30, 35, 40, or 50 g a.i /ha.
- the compound according to formula (I) is applied at a rate of at least 10 g/ha, preferably at least 10, 15, 20, 25, 30, 35, 40, 45, 50, 55, 60 g/ha.
- the compound according to formula (I) is applied at a rate of at most 100 g/ha, preferably at most 100, 95, 90, 85, 80, 75, 70, 65 g/ha.
- g a.i./ha refers to the application rate given in gram [g] of active ingredient [a.i.] per unit of surface [ha].
- the unit hectare symbol ha is the metric unit of area that equals a square with 100 m side (1 hm 2 ) or 10,000 square meters. Hectare is a commonly used unit of area in the metric system.
- compositions according to the invention depend on the type of effect desired, and typically range from 20 to 4000 g of total composition per hectare.
- compositions according to the invention are used for treating seed, rates of 0.001 to 50 g of a compound of component (A) per kg of seed, preferably from 0.01 to 10g per kg of seed, and preferably 0.001 to 50 g of a compound of component (B), per kg of seed, preferably from 0.01 to 10g per kg of seed, are generally sufficient.
- foliar formulation types for pre-mix compositions are:
- WP wettable powders
- WG water dispersible granules (powders)
- EW emulsions, oil in water
- CD oil-based suspension concentrate
- SE aqueous suspo-emulsion.
- seed treatment formulation types for pre-mix compositions are:
- WS wettable powders for seed treatment slurry
- WG water dispersible granules
- CS aqueous capsule suspension.
- formulation types suitable fortank-mix compositions are solutions, dilute emulsions, suspensions, or a mixture thereof, and dusts.
- the methods of application such as foliar, drench, spraying, atomizing, dusting, scattering, coating or pouring, are chosen in accordance with the intended objectives and the prevailing circumstances.
- the tank-mix compositions are generally prepared by diluting with a solvent (for example, water) the one or more pre-mix compositions containing different pesticides, and optionally further auxiliaries.
- a solvent for example, water
- Suitable carriers and adjuvants can be solid or liquid and are the substances ordinarily employed in formulation technology, e.g., natural or regenerated mineral substances, solvents, dispersants, wetting agents, tackifiers, thickeners, binders, or fertilizers.
- a tank-mix formulation for foliar or soil application comprises 0.1 to 20%, especially 0.1 to 15 %, of the desired ingredients, and 99.9 to 80 %, especially 99.9 to 85 %, of a solid or liquid auxiliaries (including, for example, a diluent or solvent such as water), where the auxiliaries can be a surfactant in an amount of 0 to 20 %, especially 0.1 to 15 %, based on the tank-mix formulation.
- auxiliaries including, for example, a diluent or solvent such as water
- a pre-mix formulation for foliar application comprises 0.1 to 99.9 %, especially 1 to 95 %, of the desired ingredients, and 99.9 to 0.1 %, especially 99 to 5 %, of a solid or liquid adjuvant (including, for example, a solvent such as water), where the auxiliaries can be a surfactant in an amount of 0 to 50 %, especially 0.5 to 40 %, based on the pre-mix formulation.
- a solid or liquid adjuvant including, for example, a solvent such as water
- a tank-mix formulation for seed treatment application comprises 0.25 to 80%, especially 1 to 75 %, of the desired ingredients, and 99.75 to 20 %, especially 99 to 25 %, of a solid or liquid auxiliaries (including, for example, a solvent such as water), where the auxiliaries can be a surfactant in an amount of 0 to 40 %, especially 0.5 to 30 %, based on the tank-mix formulation.
- auxiliaries including, for example, a solvent such as water
- a pre-mix formulation for seed treatment application comprises 0.5 to 99.9 %, especially 1 to 95 %, of the desired ingredients, and 99.5 to 0.1 %, especially 99 to 5 %, of a solid or liquid adjuvant (including, for example, a solvent such as water), where the auxiliaries can be a surfactant in an amount of 0 to 50 %, especially 0.5 to 40 %, based on the pre-mix formulation.
- a solid or liquid adjuvant including, for example, a solvent such as water
- Preferred seed treatment pre-mix formulations are aqueous suspension concentrates.
- the formulation can be applied to the seeds using conventional treating techniques and machines, such as fluidized bed techniques, the roller mill method, rotostatic seed treaters, and drum coaters. Other methods, such as spouted beds may also be useful.
- the seeds may be presized before coating. After coating, the seeds are typically dried and then transferred to a sizing machine for sizing. Such procedures are known in the art.
- the compounds of the present invention are particularly suited for use in soil and seed treatment applications.
- the pre-mix compositions of the invention contain 0.5 to 99.9 especially 1 to 95, advantageously 1 to 50 % by mass of the desired ingredients, and 99.5 to 0.1 , especially 99 to 5 % by mass of a solid or liquid adjuvant (including, for example, a solvent such as water), where the auxiliaries (or adjuvant) can be a surfactant in an amount of 0 to 50, especially 0.5 to 40 % by mass based on the mass of the pre-mix formulation.
- a solid or liquid adjuvant including, for example, a solvent such as water
- biocidally active ingredients or compositions may be combined with the compositions of the invention and used in the methods of the invention and applied simultaneously or sequentially with the compositions of the invention. When applied simultaneously, these further active ingredients may be formulated together with the compositions of the invention or mixed in, for example, the spray tank. These further biocidally active ingredients may be fungicides, herbicides, insecticides, bactericides, acaricides, nematicides and/or plant growth regulators.
- compositions of the invention may also be applied with one or more systemically acquired resistance inducers (“SAR” inducer).
- SAR inducers are known and described in, for example, United States Patent No. US6, 919,298 and include, for example, salicylates and the commercial SAR inducer acibenzolar- S-methyl.
- the compounds as defined in any one of embodiments according to the invention are normally used in the form of compositions and can be applied to the crop area or plant to be treated, simultaneously or in succession with further compounds.
- further compounds can be e.g., fertilizers or micronutrient donors or other preparations, which influence the growth of plants. They can also be selective herbicides or non-selective herbicides as well as insecticides, fungicides, bactericides, nematicides, molluscicides or mixtures of several of these preparations, if desired together with further carriers, surfactants or application promoting adjuvants customarily employed in the art of formulation.
- the compounds of formula (I) may be used in the form of (fungicidal) compositions for controlling or protecting against the phytopathogen(s) Corynespora cassiicola, comprising as active ingredient at least one compound as defined in any one of embodiments according to the invention, in free form or in agrochemically usable salt form, and at least one of the above-mentioned adjuvants.
- the plants and / or target crops in accordance with the invention include conventional as well as genetically enhanced or engineered varieties such as, for example, insect resistant (e.g., Bt. and VIP varieties) as well as disease resistant, herbicide tolerant (e.g., glyphosate- and glufosinate-resistant maize varieties commercially available under the trade names RoundupReady® and Li berty Lin k®) and nematode tolerant varieties.
- suitable genetically enhanced or engineered crop varieties include the Stoneville 5599BR cotton and Stoneville 4892BR cotton varieties.
- plants and/or “target crops” is to be understood as including also plants that have been rendered tolerant to herbicides like bromoxynil or classes of herbicides (such as, for example, HPPD inhibitors, ALS inhibitors, for example primisulfuron, prosulfuron and trifloxysulfuron, EPSPS (5-enol-pyrovyl-shikimate-3- phosphate-synthase) inhibitors, GS (glutamine synthetase) inhibitors or PPO (protoporphyrinogen-oxidase) inhibitors) as a result of conventional methods of breeding or genetic engineering.
- herbicides like bromoxynil or classes of herbicides
- EPSPS (5-enol-pyrovyl-shikimate-3- phosphate-synthase) inhibitors
- GS glutamine synthetase
- PPO protoporphyrinogen-oxidase
- An example of a crop that has been rendered tolerant to imidazolinones, e.g., imazamox, by conventional methods of breeding (mutagenesis) is Clearfield® summer rape (Canola).
- crops that have been rendered tolerant to herbicides or classes of herbicides by genetic engineering methods include glyphosate- and glufosinateresistant maize varieties commercially available under the trade names RoundupReady®, Herculex I® and Li berty Link®.
- plants and/or “target crops” is to be understood as including those which naturally are or have been rendered resistant to harmful insects. This includes plants transformed by the use of recombinant DNA techniques, for example, to be capable of synthesizing one or more selectively acting toxins, such as are known, for example, from toxin-producing bacteria. Examples of toxins which can be expressed include 8- endotoxins, vegetative insecticidal proteins (Vip), insecticidal proteins of bacteria colonizing nematodes, and toxins produced by scorpions, arachnids, wasps, and fungi.
- Vip vegetative insecticidal proteins
- insecticidal proteins of bacteria colonizing nematodes and toxins produced by scorpions, arachnids, wasps, and fungi.
- An example of a crop that has been modified to express the Bacillus thuringiensis toxin is the Bt maize KnockOut® (Syngenta Seeds).
- An example of a crop comprising more than one gene that codes for insecticidal resistance and thus expresses more than one toxin is VipCot® (Syngenta Seeds).
- Crops or seed material thereof can also be resistant to multiple types of pests (so-called stacked transgenic events when created by genetic modification).
- a plant can have the ability to express an insecticidal protein while at the same time being herbicide tolerant, for example Herculex I® (Dow AgroSciences, Pioneer Hi-Bred International).
- plants and/or “target crops” is to be understood as also including plants which have been so transformed by the use of recombinant DNA techniques that they are capable of synthesising antipathogenic substances having a selective action, such as, for example, the so-called "pathogenesis-related proteins" (PRPs, see e.g., EP0392225A).
- PRPs pathogenesis-related proteins
- Examples of such antipathogenic substances and transgenic plants capable of synthesising such antipathogenic substances are known, for example, from EP0392225A, WO95/33818, and EP0353191A.
- the methods of producing such transgenic plants are generally known to the person skilled in the art and are described, for example, in the publications mentioned above.
- Toxins that can be expressed by transgenic plants include, for example, insecticidal proteins from Bacillus cereus or Bacillus popilliae; or insecticidal proteins from Bacillus thuringiensis, such as 8-endotoxins, e.g. CrylAb, CrylAc, Cry1 F, Cry1 Fa2, Cry2Ab, Cry3A, Cry3Bb1 or Cry9C, or vegetative insecticidal proteins (Vip), e.g. Vip1 , Vip2, Vip3 or Vip3A; or insecticidal proteins of bacteria colonising nematodes, for example Photorhabdus spp.
- insecticidal proteins from Bacillus cereus or Bacillus popilliae such as 8-endotoxins, e.g. CrylAb, CrylAc, Cry1 F, Cry1 Fa2, Cry2Ab, Cry3A, Cry3Bb1 or Cry9C, or vegetative insecticidal proteins (
- Xenorhabdus spp. such as Photorhabdus luminescens, Xenorhabdus nematophilus
- toxins produced by animals such as scorpion toxins, arachnid toxins, wasp toxins and other insect-specific neurotoxins
- toxins produced by fungi such as Streptomycetes toxins, plant lectins, such as pea lectins, barley lectins or snowdrop lectins
- agglutinins proteinase inhibitors, such as trypsin inhibitors, serine protease inhibitors, patatin, cystatin, papain inhibitors
- steroid metabolism enzymes such as 3-hydroxysteroidoxidase, ecdysteroid- UDP-glycosyl-transferase, cholesterol oxidases, ecd
- 8-endotoxins for example CrylAb, CrylAc, Cry1 F, Cry1 Fa2, Cry2Ab, Cry3A, Cry3Bb1 or Cry9C, or vegetative insecticidal proteins (Vip), for example Vip1 , Vip2, Vip3 or Vip3A, expressly also hybrid toxins, truncated toxins and modified toxins.
- Hybrid toxins are produced recombinantly by a new combination of different domains of those proteins (see, for example, WO 02/15701).
- Truncated toxins for example a truncated CrylAb, are known.
- modified toxins one or more amino acids of the naturally occurring toxin are replaced.
- amino acid replacements preferably non-naturally present protease recognition sequences are inserted into the toxin, such as, for example, in the case of Cry3A055, a cathepsin-G-recognition sequence is inserted into a Cry3A toxin (see W003/018810).
- Cryl-type deoxyribonucleic acids and their preparation are known, for example, from WO 95/34656, EP-A-0 367 474, EP-A-0 401 979 and WO 90/13651.
- the toxin contained in the transgenic plants imparts to the plants tolerance to harmful insects.
- insects can occur in any taxonomic group of insects but are especially commonly found in the beetles (Coleoptera), two-winged insects (Diptera) and butterflies (Lepidoptera).
- Transgenic plants containing one or more genes that code for an insecticidal resistance and express one or more toxins are known and some of them are commercially available. Examples of such plants are: YieldGard ® (maize variety that expresses a CrylAb toxin); YieldGard Rootworm® (maize variety that expresses a Cry3Bb1 toxin); YieldGard Plus® (maize variety that expresses a CrylAb and a Cry3Bb1 toxin); Starlink® (maize variety that expresses a Cry9C toxin); Herculex I® (maize variety that expresses a Cry1 Fa2 toxin and the enzyme phosphinothricine N-acetyltransferase (PAT) to achieve tolerance to the herbicide glufosinate ammonium); NuCOTN 33B® (cotton variety that expresses a Cry1 Ac toxin); Bollgard I® (cotton variety that expresses
- transgenic crops are:
- MIR604 Maize from Syngenta Seeds SAS, Chemin de I'Hobit 27, F-31 790 St. Sauveur, France, registration number C/FR/96/05/10. Maize which has been rendered insect-resistant by transgenic expression of a modified Cry3A toxin. This toxin is Cry3A055 modified by insertion of a cathepsin-G-protease recognition sequence. The preparation of such transgenic maize plants is described in WO 03/018810.
- MON 863 Maize from Monsanto Europe S.A. 270-272 Avenue de Tervuren, B-1150 Brussels, Belgium, registration number C/DE/02/9. MON 863 expresses a Cry3Bb1 toxin and has resistance to certain Coleoptera insects. 5. IPC 531 Cotton from Monsanto Europe S.A. 270-272 Avenue de Tervuren, B-1150 Brussels, Belgium, registration number C/ES/96/02.
- NK603 x MON 810 Maize from Monsanto Europe S.A. 270-272 Avenue de Tervuren, B-1150 Brussels, Belgium, registration number C/GB/02/M3/03. Consists of conventionally bred hybrid maize varieties by crossing the genetically modified varieties NK603 and MON 810.
- NK603 x MON 810 Maize transgenically expresses the protein CP4 EPSPS, obtained from Agrobacterium sp. strain CP4, which imparts tolerance to the herbicide Roundup® (contains glyphosate), and also a CrylAb toxin obtained from Bacillus thuringiensis subsp. kurstaki which brings about tolerance to certain Lepidoptera, include the European corn borer.
- locus means fields in or on which plants are growing, or where seeds of cultivated plants are sown, or where seed will be placed into the soil. It includes soil, seeds, and seedlings, as well as established vegetation.
- plants refers to all physical parts of a plant, including seeds, seedlings, saplings, roots, tubers, stems, stalks, foliage, and fruits.
- plant propagation material is understood to denote generative parts of the plant, such as seeds, which can be used for the multiplication of the latter, and vegetative material, such as cuttings or tubers, for example potatoes. There may be mentioned for example seeds (in the strict sense), roots, fruits, tubers, bulbs, rhizomes and parts of plants. Germinated plants and young plants which are to be transplanted after germination or after emergence from the soil, may also be mentioned. These young plants may be protected before transplantation by a total or partial treatment by immersion. Preferably “plant propagation material” is understood to denote seeds.
- Pesticidal agents referred to herein using their common name are known, for example, from “The Pesticide Manual”, 15 th Ed., British Crop Protection Council 2009.
- the compounds as defined in any one of embodiments according to the invention for use in the inventive methods may be the sole active ingredient of a composition or it may be admixed with one or more additional active ingredients such as a pesticide, fungicide, synergist, herbicide, or plant growth regulator where appropriate.
- An additional active ingredient may, in some cases, result in unexpected synergistic activities.
- compositions according to the invention can also comprise further solid or liquid auxiliaries, such as stabilizers, for example unepoxidized or epoxidized vegetable oils (for example epoxidized coconut oil, rapeseed oil or soya oil), antifoams, for example silicone oil, preservatives, viscosity regulators, binders and/or tackifiers, fertilizers or other active ingredients for achieving specific effects, for example bactericides, fungicides, nematicides, plant activators, molluscicides or herbicides.
- auxiliaries such as stabilizers, for example unepoxidized or epoxidized vegetable oils (for example epoxidized coconut oil, rapeseed oil or soya oil), antifoams, for example silicone oil, preservatives, viscosity regulators, binders and/or tackifiers, fertilizers or other active ingredients for achieving specific effects, for example bactericides, fungicides, nematicides, plant
- the compounds, and compositions according to the invention are prepared in a manner known per se, in the absence of auxiliaries for example by grinding, screening and/or compressing a solid active ingredient and in the presence of at least one auxiliary for example by intimately mixing and/or grinding the active ingredient with the auxiliary (auxiliaries).
- auxiliaries for example by grinding, screening and/or compressing a solid active ingredient and in the presence of at least one auxiliary for example by intimately mixing and/or grinding the active ingredient with the auxiliary (auxiliaries).
- Another aspect of the invention is related to the use of a compound as defined in any one of embodiments according to the invention, of a composition comprising at least one compound as defined in, or of a fungicidal or insecticidal mixture comprising at least one compound as defined in any one of embodiments according to the invention, in admixture with other fungicides or insecticides as described above, for controlling or preventing infestation of plants, e.g. plants such as crop plants, propagation material thereof, e.g. seeds, harvested crops, e.g. harvested food crops, or non-living material by insects or by phytopathogenic microorganisms, preferably fungal organisms.
- plants e.g. plants such as crop plants, propagation material thereof, e.g. seeds, harvested crops, e.g. harvested food crops, or non-living material by insects or by phytopathogenic microorganisms, preferably fungal organisms.
- a further aspect of invention is related to a method of controlling or preventing an infestation of plants, e.g. plants such as crop plants, propagation material thereof, e.g. seeds, harvested crops, e.g. harvested food crops, or of non-living materials by phytopathogenic or spoilage microorganisms or organisms potentially harmful to man, especially fungal organisms, which comprises the application of a compound as defined in any one of embodiments according to the invention as active ingredient to the plants, to parts of the plants or to the locus thereof, to the propagation material thereof, or to any part of the non-living materials.
- plants e.g. plants such as crop plants, propagation material thereof, e.g. seeds, harvested crops, e.g. harvested food crops, or of non-living materials by phytopathogenic or spoilage microorganisms or organisms potentially harmful to man, especially fungal organisms
- a compound as defined in any one of embodiments according to the invention as active ingredient to the plants, to parts of the plants or to the locus thereof, to the propagation
- Controlling or preventing means reducing infestation by insects or by phytopathogenic or spoilage microorganisms or organisms potentially harmful to man, especially fungal organisms, to such a level that an improvement is demonstrated.
- a preferred method of controlling or preventing an infestation of crop plants by phytopathogenic microorganisms, especially fungal organisms, or insects which comprises the application of a compound as defined in any one of embodiments according to the invention, or an agrochemical composition which contains at least one of said compounds, is foliar application.
- the frequency of application and the rate of application will depend on the risk of infestation by the corresponding pathogen or insect.
- the compounds of formula (I), preferably in association with a compound (B), can also penetrate the plant through the roots via the soil (systemic action) by drenching the locus of the plant with a liquid formulation, or by applying the compounds in solid form to the soil, e.g., in granular form (soil application). In crops of water rice such granulates can be applied to the flooded rice field.
- the compounds of formula (I) may also be applied to seeds (coating) by impregnating the seeds or tubers either with a liquid formulation of the fungicide or coating them with a solid formulation.
- compositions comprising compounds of formula (I) are as described for compositions comprising compounds of formula (I) and additionally preferably at least one component (B) as described above.
- Their application can be both before and after the infection of the plants or parts thereof with the fungi. The treatment is preferably carried out prior to the infection.
- the application rates in the method according to the invention are as described above, e.g., typical are rates of 5 to 2000 g a.i./ha, particularly 10 to 1000 g a.i./ha, e.g., 50, 75, 100 or 200 g a.i./ha.
- Compounds of formula (I) can be applied to the plants once or more than once during a growing season.
- the compounds of formula (I) can be converted into the customary formulations described above, e.g., solutions, emulsions, suspensions, dusts, powders, pastes and granules.
- the use form will depend on the particular intended purpose; in each case, it should ensure a fine and even distribution of the compound of formula (I).
- plant as used herein includes seedlings, bushes and crops of fruits and vegetables.
- compositions comprising only a compound of formula (I) as the active ingredient.
- the compounds of the invention can be distinguished from known compounds by virtue of greater efficacy at low application rates, which can be verified by a person skilled in the art using the experimental procedures outlined in the Examples, using lower application rates, if necessary, for example 60 ppm, 20 ppm or 2 ppm.
- Compounds of formula (I) may possess any number of benefits including, inter alia, advantageous levels of biological activity for protecting plants against diseases that are caused by fungi or superior properties for use as agrochemical active ingredients (for example, greater biological activity, an advantageous spectrum of activity, an increased safety profile (including improved crop tolerance), improved physico-chemical properties, or increased biodegradability).
- Wettable powders a) b) c) active ingredients 25 % 50 % 75 % sodium lignosulfonate 5 % 5 % - sodium lauryl sulfate 3 % - 5 % sodium diisobutylnaphthalenesulfonate 6 % 10 % phenol polyethylene glycol ether (7-8 mol of ethylene oxide) 2 % highly dispersed silicic acid 5 % 10 % 10 %
- the combination is thoroughly mixed with the adjuvants and the mixture is thoroughly ground in a suitable mill, affording wettable powders that can be diluted with water to give suspensions of the desired concentration.
- Powders for dry seed treatment a) b) c) active ingredients 25 % 50 % 75 % light mineral oil 5 % 5 % 5 % highly dispersed silicic acid 5 % 5 %
- the combination is thoroughly mixed with the adjuvants and the mixture is thoroughly ground in a suitable mill, affording powders that can be used directly for seed treatment.
- Emulsifiable concentrate active ingredients 10 % octylphenol polyethylene glycol ether (4-5 mol of ethylene oxide) 3 % calcium dodecylbenzene sulfonate 3 % castor oil polyglycol ether (35 mol of ethylene oxide) 4 %
- Emulsions of any reguired dilution which can be used in plant protection, can be obtained from this concentrate by dilution with water.
- Ready-for-use dusts are obtained by mixing the combination with the carrier and grinding the mixture in a suitable mill. Such powders can also be used for dry dressings for seed.
- the combination is mixed and ground with the adjuvants, and the mixture is moistened with water.
- the mixture is extruded and then dried in a stream of air.
- the finely ground combination is uniformly applied, in a mixer, to the kaolin moistened with polyethylene glycol.
- Non-dusty coated granules are obtained in this manner.
- Suspension concentrate active ingredients 40 % propylene glycol 10 % nonylphenol polyethylene glycol ether (15 mol of ethylene oxide) 6 %
- the finely ground combination is intimately mixed with the adjuvants, giving a suspension concentrate from which suspensions of any desired dilution can be obtained by dilution with water.
- a suspension concentrate from which suspensions of any desired dilution can be obtained by dilution with water.
- living plants as well as plant propagation material can be treated and protected against infestation by microorganisms, by spraying, pouring or immersion.
- Flowable concentrate for seed treatment active ingredients 40 % propylene glycol 5 % copolymer butanol PO/EO 2 %
- Silicone oil (in the form of a 75 % emulsion in water) 0.2 %
- the finely ground combination is intimately mixed with the adjuvants, giving a suspension concentrate from which suspensions of any desired dilution can be obtained by dilution with water.
- a suspension concentrate from which suspensions of any desired dilution can be obtained by dilution with water.
- living plants as well as plant propagation material can be treated and protected against infestation by microorganisms, by spraying, pouring or immersion.
- 28 parts of the combination are mixed with 2 parts of an aromatic solvent and 7 parts of toluene diisocyanate/polymethylene-polyphenylisocyanate-mixture (8:1).
- This mixture is emulsified in a mixture of 1.2 parts of polyvinylalcohol, 0.05 parts of a defoamer and 51.6 parts of water until the desired particle size is achieved.
- a mixture of 2.8 parts 1 ,6-diaminohexane in 5.3 parts of water is added.
- the mixture is agitated until the polymerization reaction is completed.
- the obtained capsule suspension is stabilized by adding 0.25 parts of a thickener and 3 parts of a dispersing agent.
- the capsule suspension formulation contains 28% of the active ingredients.
- the medium capsule diameter is 8-15 microns.
- the resulting formulation is applied to seeds as an aqueous suspension in an apparatus suitable for that purpose.
- Formulation types include an emulsion concentrate (EC), a suspension concentrate (SC), a suspo-emulsion (SE), a capsule suspension (CS), a water dispersible granule (WG), an emulsifiable granule (EG), an emulsion, water in oil (EO), an emulsion, oil in water (EW), a micro-emulsion (ME), an oil dispersion (OD), an oil miscible flowable (OF), an oil miscible liquid (OL), a soluble concentrate (SL), an ultra-low volume suspension (SU), an ultra-low volume liquid (UL), a technical concentrate (TK), a dispersible concentrate (DC), a wettable powder (WP), a soluble granule (SG) or any technically feasible formulation in combination with agriculturally acceptable adjuvants.
- EC emulsion concentrate
- SC suspension concentrate
- SE suspo-emulsion
- CS capsule suspension
- WG water dispersible granule
- EG
- Example B1 Corynespora cassiicola (target leaf spot) - field trials 2023
- Improved yield is calculated by subtracting the yield of the untreated plants from the yield of the treated plants, then divide the result by the absolute value of the yield of the untreated plants and multiply the result by 100. The answer is the percent increase.
- Such compounds of formula (I) according to the present invention gave at least 50% control when compared to untreated control under the same conditions, which showed extensive disease development. Further compounds of formula (I) gave a significant yield increase when compared to untreated control under the same conditions, which showed extensive disease development.
- the activity of the compounds against Corynespora cassiicola in the field- providing overall control similar to that of the commercial triple mix - is surprising and unexpected given the lack of activity against the same fungus in liquid culture, especially when compared to the relative activity of the active ingredients from the commercial product in liquid culture.
- the compounds of formula (I), (l-A), or (l-B) may for example be distinguished from other compounds by virtue of greater efficacy at low application rates, which can be verified by the person skilled in the art using the experimental procedures outlined in the above biological test, using lower application rates, if necessary, for example 6 ppm, 3 ppm, 2.2 ppm, 1 .5 ppm, 0.8 ppm or 0.74 ppm.
Landscapes
- Life Sciences & Earth Sciences (AREA)
- Plant Pathology (AREA)
- Environmental Sciences (AREA)
- Pest Control & Pesticides (AREA)
- Engineering & Computer Science (AREA)
- Wood Science & Technology (AREA)
- Zoology (AREA)
- Mycology (AREA)
- General Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Microbiology (AREA)
- Chemical & Material Sciences (AREA)
- Agronomy & Crop Science (AREA)
- Health & Medical Sciences (AREA)
- Dentistry (AREA)
- General Health & Medical Sciences (AREA)
- Agricultural Chemicals And Associated Chemicals (AREA)
Abstract
The present invention relates to methods for controlling or preventing infestation of a plant by the phytopathogenic microorganism Corynespora cassiicola, comprising applying to the phytopathogen, to the locus of the phytopathogen, or to a plant susceptible to attack by the phytopathogen, or to a propagation material thereof, a fungicidally effective amount of a compound, N-oxide or agriculturally acceptable salts thereof as defined in claim 1.
Description
METHODS OF CONTROLLING OR PREVENTING INFESTATION OF PLANTS BY THE PHYTOPATHOGENIC MICROORGANISM CORYNESPORA CASSIICOLA
The present invention relates to methods for controlling or preventing infestation of soybean plants by the phytopathogenic microorganism Corynespora cassiicola.
Corynespora cassiicola infects over 530 species of plants in 53 families (Dixon, L. J., et al., Phytopathology 2009, 99(9), 1015-27). It is most common in the tropics and subtropics. It has also been isolated from nematodes and from human skin. Corynespora cassiicola is known as a pathogen of many agricultural crop plants, for example soybeans, corn, wheat, cotton, beans, potatoes, sweet potato, and tobacco. The disease caused by Corynespora cassiicola is called target leaf spot or target spot on several plants, for example target leaf spot of soybeans and target leaf spot on cotton.
The development of resistance of the phytopathogen(s) Corynespora cassiicola to many of the current commercial solutions restricts their utility and, whilst the development of new classes of agrochemical fungicides is on-going, many of these new classes of chemistry have limitations in their fungicidal spectrum and control only certain, specific phytopathogens. That is to say that, when a new class of chemistry is shown to control certain, specific phytopathogens on certain, specific crops, it cannot be expected that the same class of chemistry will prove useful in the control of the phytopathogen Corynespora cassiicola, for example on soybean or cotton.
There exists therefore a need for the development of new methods for controlling or preventing infestation by the phytopathogen Corynespora cassiicola.
It has now been surprisingly found that particular pyrazole derivatives as disclosed in WO 2020/056090 and WO 2021/183707, are highly effective at controlling or preventing the infestation of plants by the phytopathogenic microorganisms of the family Corynesporascaceae, in particular Corynespora cassiicola. These highly effective compounds thus represent an important new solution for farmers to control or prevent infestation of plants by the phytopathogenic microorganism Corynespora cassiicola.
Further it has now been surprisingly found that particular pyrazole derivatives as disclosed in WO 2020/056090 and WO 2021/183707, are highly effective at controlling or preventing the infestation of soybean plants by the phytopathogenic microorganisms of the family Corynesporascaceae, in particular Corynespora cassiicola. These highly effective compounds thus represent an important new solution for farmers to control or prevent infestation of soybean plants by the phytopathogenic microorganism Corynespora cassiicola.
Additionally, it has now been surprisingly found that particular pyrazole derivatives as disclosed in WO 2020/056090 and WO 2021/183707, are highly effective at controlling or preventing the infestation of genetically modified soybean plants by the phytopathogenic microorganisms of the family Corynesporascaceae, in particular Corynespora cassiicola. These highly effective compounds thus represent an important new solution for farmers to control or prevent infestation of genetically modified soybean plants, like Bt soybean plants, by the phytopathogenic microorganism Corynespora cassiicola.
Further it has now been surprisingly found that pyrazole derivatives as disclosed in WO 2020/056090 and WO 2021/183707, are highly effective at controlling or preventing the infestation of cotton by the phytopathogenic microorganisms of the family Corynesporascaceae, in particular Corynespora cassiicola. These highly effective compounds thus represent an important new solution for farmers to control or prevent infestation of cotton by the phytopathogenic microorganism Corynespora cassiicola.
Additionally, it has now been surprisingly found that particular pyrazole derivatives as disclosed in WO 2020/056090 and WO 2021/183707, are highly effective at controlling or preventing the infestation of genetically modified cotton by the phytopathogenic microorganisms of the family Corynesporascaceae, in particular Corynespora cassiicola. These highly effective compounds thus represent an important new solution for farmers to control or prevent infestation of genetically modified cotton, like Bt cotton, by the phytopathogenic microorganism Corynespora cassiicola.
Additionally, it has now been surprisingly found that particular 5-(trifluoro-methyl)-1 ,2,4-oxadiazole compounds, are highly effective at controlling or preventing the infestation of genetically modified cotton by the phytopathogenic microorganisms of the family Corynesporascaceae, in particular Corynespora cassiicola. These highly effective compounds thus represent an important new solution for farmers to control or prevent infestation of genetically modified cotton, like Bt cotton, by the phytopathogenic microorganism Corynespora cassiicola.
Additionally, it has now been surprisingly found that particular 5-(trifluoro-methyl)-1 ,2,4-oxadiazole compounds are highly effective at controlling or preventing the infestation of field-grown plants by the phytopathogenic microorganisms of the family Corynesporascaceae, in particular Corynespora cassiicola. These highly effective compounds thus represent an important new solution for farmers to control or prevent infestation of field-grown plants by the phytopathogenic microorganism Corynespora cassiicola.
Additionally, it has now been surprisingly found that pesticidal compositions comprising particular 5-(trifluoro- methyl)-1 ,2,4-oxadiazole compounds and one or more formulation adjuvants, are highly effective at controlling or preventing the infestation of plants, in particular field-grown plants, by the phytopathogenic microorganisms of the family Corynesporascaceae, in particular Corynespora cassiicola. These highly effective pesticidal compositions thus represent an important new solution for farmers to control or prevent infestation of plants, in particular field-grown plants, by the phytopathogenic microorganism Corynespora cassiicola.
In particular, it has now been surprisingly found that particular 5-(trifluoro-methyl)-1 ,2,4-oxadiazole compounds and pesticidal compositions as defined herein are more effective at controlling or preventing the infestation of plants by the phytopathogenic microorganisms of the family Corynesporascaceae, in particular Corynespora cassiicola when applied in the field as compared to the activity against the same phytopathogen when applied in liquid culture, especially when compared to the relative activity of the active ingredients from the commercial product in liquid culture.
Said pyrazole derivatives, its pesticidal action and suitable methods for producing are disclosed in WO 2020/056090.
Agrochemical compositions comprising said pyrazole derivatives, have been disclosed in WO 2021/18370.
According to a first aspect of the present invention, there is provided a method of controlling or preventing infestation of plants by the phytopathogenic microorganism Corynespora cassiicola comprising applying to the phytopathogen, to the locus of the phytopathogen, or to a plant susceptible to attack by the phytopathogen, or to a propagation material thereof, a fungicidally effective amount of a compound according to formula (I)
wherein
R1 is Ci-Csalkyl; and
R2 is selected from the group consisting of: wherein the staggered li _
ne denotes the bond to the rest of the molecule; or an agrochemically acceptable salt, stereoisomer, enantiomer, tautomer or N-oxide thereof.
According to a second aspect of the present invention, there is provided a method of controlling or preventing infestation of plants by the phytopathogenic microorganism Corynespora cassiicola comprising applying to the phytopathogen, to the locus of the phytopathogen, or to a plant susceptible to attack by the phytopathogen, or to a propagation material thereof, a pesticidal composition comprising a compound of formula (I), and one or more formulation adjuvants.
In a preferred embodiment, the compound according to formula (I), or the pesticidal composition comprising a compound of formula (I) and one or more formulation adjuvants is applied in the field.
According to a third aspect of the present invention, there is provided a method of controlling or preventing infestation of field-grown plants by the phytopathogenic microorganism Corynespora cassiicola comprising applying to the phytopathogen, to the locus of the phytopathogen, or to a plant susceptible to attack by the phytopathogen, or to a propagation material thereof, a compound of formula (I), or a pesticidal composition comprising a compound of formula (I) and one or more formulation adjuvants, formulation auxiliaries, diluents, and/or carriers.
According to a fourth aspect of the present invention, there is provided the method according to any one of embodiments above comprising the steps of providing a composition comprising a compound of formula (I); and applying the composition to a propagation material; and planting the propagation material.
According to a fifth aspect of the present invention, there is provided the method according to any one of
embodiments above, comprising the steps of providing a composition comprising a compound of formula (I); applying the composition to the phytopathogen, to the locus of the phytopathogen, or to a plant susceptible to attack by the phytopathogen.
Preferably, in these aspects of the invention, the pesticidal composition comprises at least and one or more formulation adjuvants, as well as and one or more formulation auxiliaries, diluents, and/or carriers.
According to a sixth aspect of the present invention, there is provided the use of a compound of formula (I) for controlling or preventing infestation of plants by the phytopathogenic microorganism Corynespora cassiicola.
According to a seventh aspect of the present invention, there is provided a method or use according to any one of embodiments herein above, wherein the plant is selected from beans, cowpea, cucumber, papaya, soybean, potatoes, sweet potato, tomato, cotton, corn, wheat, tobacco, eggplant, basil, thyme, rubber tree, pawpaw tree, azalea and hydrangea.
According to an eighth aspect of the present invention, there is provided a method or use according to any one of embodiments herein above, wherein the plant is selected from soybeans, corn, wheat, cotton, beans, potatoes, sweet potato, and tobacco.
According to a ninth aspect of the present invention, there is provided a method or use according to any one of the previous embodiments, wherein the plants are genetically modified plants, preferably Bt plants.
According to a tenth aspect ofthe present invention, the present invention preferably also relates to a pesticidal composition suitable for control of diseases caused by phytopathogens comprising a component (A) and at least one or more compound(s) (B), wherein said component A is a compound of formula (I)
wherein R1 is Ci-Csalkyl; and R2 is selected from the group consisting of:
the molecule; and wherein said at least one or more compound(s) (B) is selected from other biologically active agents, such as bactericides, fungicides, nematicides, plant activators, acaricides, and insecticides, one or more adjuvant(s), a diluent or carrier, formulation auxiliary, or a combination thereof..
According to a tenth aspect ofthe present invention, the present invention preferably also relates to a pesticidal composition suitable for control of diseases caused by phytopathogens comprising a component (A) and at least one or more compound(s) (B), wherein said component A is a compound of formula (I)
wherein R1 is Ci-Csalkyl; and R2 is selected from the group consisting of:
wherein the staggered line denotes the bond to the rest of the molecule; and wherein said at least one or more compound(s) (B) is selected from pesticides.
Compounds of formula (I) which have at least one basic centre can form, for example, acid addition salts, for example with strong inorganic acids such as mineral acids, for example perchloric acid, sulfuric acid, nitric acid, nitrous acid, a phosphorus acid or a hydrohalic acid, with strong organic carboxylic acids, such as Ci- C4alkanecarboxylic acids which are unsubstituted or substituted, for example by halogen, for example acetic acid, such as saturated or unsaturated dicarboxylic acids, for example oxalic acid, malonic acid, succinic acid, maleic acid, fumaric acid or phthalic acid, such as hydroxycarboxylic acids, for example ascorbic acid, lactic acid, malic acid, tartaric acid or citric acid, or such as benzoic acid, or with organic sulfonic acids, such as Ci- 04 alkane- or arylsulfonic acids which are unsubstituted or substituted, for example by halogen, for example methane- or p-toluenesulfonic acid. Compounds of formula (I) which have at least one acidic group can form, for example, salts with bases, for example mineral salts such as alkali metal or alkaline earth metal salts, for example sodium, potassium or magnesium salts, or salts with ammonia or an organic amine, such as morpholine, piperidine, pyrrolidine, a mono-, di- or tri-lower-alkylamine, for example ethyl-, diethyl-, triethyl- or dimethylpropylamine, or a mono-, di- ortrihydroxy-lower-alkylamine, for example mono-, di- ortriethanolamine.
In each case, the compounds of formula (I) according to the invention are in free form, in oxidized form as a N-oxide or in salt form, e.g., an agronomically usable salt form. N-oxides are oxidized forms of tertiary amines or oxidized forms of nitrogen containing heteroaromatic compounds. They are described for instance in the book “Heterocyclic N-oxides” by A. Albini and S. Pietra, CRC Press, Boca Raton 1991 .
The compounds of formula (I) according to the invention also include hydrates which may be formed during the salt formation.
As used herein, the term "controlling" refers to reducing the number of pests, eliminating pests and/or preventing further pest damage such that damage to a plant or to a plant derived product is reduced.
As used herein, the term "pest" refers to insects, and molluscs that are found in agriculture, horticulture, forestry, the storage of products of vegetable origin (such as fruit, grain, and timber); and those pests associated with the damage of man-made structures. The term pest encompasses all stages in the life cycle of the pest.
As used herein, the term "effective amount" refers to the amount of the compound, or a salt thereof, which, upon single or multiple applications provides the desired effect.
An effective amount is readily determined by the skilled person in the art, using known techniques and by observing results obtained under analogous circumstances. In determining the effective amount, a number of factors are considered including, but not limited to the type of plant or derived product to be applied; the pest to be controlled and its lifecycle; the particular compound applied; the type of application; and other relevant circumstances.
As used herein, the term “room temperature” or “RT” or “rt” or “ambient temperature” refers to a temperature of about 15° C to about 35° C. For example, rt can refer to a temperature of about 20° C to about 30° C.
As used herein, the term "halogen" or “halo” refers to fluorine (fluoro), chlorine (chloro), bromine (bromo) or iodine (iodo), preferably fluorine, chlorine or bromine. This also applies, correspondingly, to halogen in combination with other meanings, such as haloalky I, haloalkenyl, haloalkynyl, haloalkoxy, and halocycloalkyl.
As used herein, the term "Ci-Cn-alkyl” refers to a saturated straight-chain or branched hydrocarbon radical attached via any of the carbon atoms having 1 to n carbon atoms, for example, any one of the radicals methyl, ethyl, n-propyl, 1 -methylbutyl, 2-methylbutyl, 3-methylbutyl, 2, 2-dimethylpropyl, 1 -ethylpropyl, n-hexyl, n- pentyl, 1 ,1 -dimethylpropyl, 1 , 2-dimethylpropyl, 1 -methylpentyl, 2-methylpentyl, 3-methylpentyl, 4- methylpentyl, 1 ,1 -dimethylbutyl, 1 ,2-dimethylbutyl, 1 ,3-dimethylbutyl, 2,2-dimethylbutyl, 2,3-dimethylbutyl, 3,3- dimethylbutyl, 1 -ethylbutyl, 2-ethylbutyl, 1 ,1 ,2-trimethylpropyl, 1 ,2,2-trimethylpropyl, 1-ethyl-1 -methylpropyl, or 1-ethyl-2-methylpropyl.
Embodiments according to the invention are provided as set out below.
R1 is selected from methyl, or ethyl; and R2 is selected from the group consisting of:
wherein the staggered line denotes the bond to the rest of the molecule.
In one embodiment ofthe invention, compounds of formula (I) are selected from ethyl 1-[[4-[(Z)-2-ethoxy-3,3,3- trifluoro-prop-1-enoxy]phenyl]methyl]pyrazole-4-carboxylate according to structure (l-A)
ethyl 1-[[4-[[2-(trifluoromethyl)-1 ,3-dioxolan-2-yl]methoxy]phenyl]methyl]pyrazole-4-carboxylate according to structure (l-B)
(l-B).
In one preferred embodiment of the invention, compound of formula (I) is ethyl 1-[[4-[(Z)-2-ethoxy-3,3,3- trifluoro-prop-1-enoxy]phenyl]methyl]pyrazole-4-carboxylate according to structure l-A.
In another preferred embodiment of the invention, compound of formula (I) is ethyl 1-[[4-[[2-(trifluoromethyl)- 1 ,3-dioxolan-2-yl]methoxy]phenyl]methyl]pyrazole-4-carboxylate according to structure l-B.
A skilled person is aware that according to the methods of the invention, the compound or compounds are generally applied as part of a pesticidal composition.
In one embodiment of the invention there is provided a composition comprising a compound of formula (I) for controlling or preventing infestation of plants by the phytopathogenic microorganism Corynespora cassiicola.
Such pesticidal compositions typically comprise one or more adjuvants, formulation auxiliaries, diluents, and/or carriers as described in detail below, preferably at least one or more adjuvants.
In another embodiment of the invention there is provided a composition comprising a compound of formula (I) and preferably one or more adjuvants, formulation auxiliaries, diluents, and/or carriers for controlling or preventing infestation of plants by the phytopathogenic microorganism Corynespora cassiicola comprising the steps of providing a composition comprising said compound of formula (I); and applying said composition to the phytopathogen, to the locus ofthe phytopathogen, orto a plant susceptible to attack by the phytopathogen.
In one embodiment of the present invention, there is provided a fungicidal composition comprising a mixture of component (A) and a component (B) as active ingredients, wherein component (A) is selected from a compound of formula (I), (l-A), or (l-B), and component (B) is a compound selected from the group consisting of benzovindiflupyr, fluxapyroxad, pydiflumetofen, isopyrazam, fluopyram, penthiopyrad, sedaxane, bixafen, difenoconazole, cyproconazole, tebuconazole, hexaconazole, prothioconazole, propiconazole, epoxiconazole, metconazole, tetraconazole, fluoxytioconazole, 2-[2-chloro-4-(4-chlorophenoxy)phenyl]-2-hydroxy-3-(1 ,2,4- triazol-1-yl)propanoate, methyl 3-[(4-chlorophenyl)methyl]-2-hydroxy-1-methyl-2-(1 ,2,4-triazol-1- ylmethyl)cyclopentanecarboxylate, flutriafol, mefentrifluconazole, ipconazole, paclobutrazol, azoxystrobin, trifloxystrobin, picoxystrobin, pyraclostrobin, metalaxyl-M, fenpropidin, fenpropimorph, cyprodinil, spiroxamine,
mancozeb, chlorothalonil, folpet, copper-oxychloride, copper-hydroxide, sulphur, oxathiapiprolin, ipflufenoquin, quinofumelin, mandipropamid, fluazinam, fludioxinil, fosetyl-aluminium, acibenzolar-S-methyl, enestrobin, inpyrfluxam, fluindapyr, isoflucypram, metyltetraprole, florylpicoxamid, metarylpicoxamide, flefunoxadiazam isofetamid, procymidone, carbendazim, fenhexamid, prochloraz, prohexadione-calcium, melaluca alternifolia oil (an extract of the tea tree plant Melaluca alternifolia (commercially available as Timorex Gold®, which is a broad-spectrum botanical biofungicide)), N'-[5-bromo-2-methyl-6-(1 -methyl-2-propoxy-ethoxy)-3-pyridyl]-N- ethyl-N-methylformamidine, cyclobutrifluram calcium phosphonate, cis-jasmone, trinexapac-ethyl, glyphosate, 2,4-D (2,4-dichlorophenoxyacetic acid); feneptamidoquin, methyl (Z)-3-methoxy-2-(2-methyl-5-phenyl- phenoxy)prop-2-enoate (this compound may be prepared from methods as described in JP2023078251), methyl (Z)-2-(5-cyclopentyl-2-methyl-phenoxy)-3-methoxy-prop-2-enoate; methyl (Z)-2-(5-cyclohexyl-2- methyl-phenoxy)-3-methoxy-prop-2-enoate (these compounds may be prepared from the methods described in W02020/193387); (5R)-3-[3-(3-chloro-2-fluoro-phenoxy)-6-methyl-pyridazin-4-yl]-5-[(2-chloro-4-methyl- phenyl)methyl]-5,6-dihydro-4H-1 ,2,4-oxadiazine; (5S)-3-[3-(3-chloro-2-fluoro-phenoxy)-6-methyl-pyridazin-4- yl]-5-[(2-chloro-4-methyl-phenyl) methyl]-5,6-dihydro-4H-1 ,2,4-oxadiazine; 3-[3-(3-chloro-2-fluoro-phenoxy)-6- methyl-pyridazin-4-yl]-5-[(2-chloro-4-methyl-phenyl)methyl]-5,6-dihydro-4H-1 ,2,4-oxadiazine (these compounds may be prepared from the methods described in WO 2020/127780, WO 2021/255070); 2-[cyano- (2,6-difluoro-4-pyridyl)amino]-5-methyl-N-spiro[3.4]octan-3-yl-thiazole-4-carboxamide; 2-[cyano-(2,6-difluoro- 4-pyridyl)amino]-N-(2,2-dimethylcyclobutyl)-5-methyl-thiazole-4-carboxamide (these compounds may be prepared from the methods described in WO2017207362A1 , WO2019105933A1 , W02020109509A1); 2-[(2,6- difluoro-4-pyridyl)-(2-methoxyacetyl)amino]-N-(2,2-dimethylcyclobutyl)-5-methyl-thiazole-4-carboxamide; 2- [(2,6-difluoro-4-pyridyl)-(tetrahydropyran-4-carbonyl)amino]-N-(2,2-dimethylcyclobutyl)-5-methyl-thiazole-4- carboxamide; 2-[(2,6-difluoro-4-pyridyl)-(oxetane-3-carbonyl)amino]-N-(2,2-dimethylcyclobutyl)-5-methyl- thiazole-4-carboxamide; 2-[acetyl-(2,6-difluoro-4-pyridyl)amino]-N-(2,2-dimethylcyclobutyl)-5-methyl-thiazole- 4-carboxamide; 2-[acetyl-(2,6-difluoro-4-pyridyl)amino]-5-methyl-N-spiro[3.4]octan-3-yl]-thiazole-4- carboxamide; 2-[(2,6-difluoro-4-pyridyl)-(2-methylpropanoyl)amino]-N-(2,2-dimethylcyclobutyl)-5-methyl- thiazole-4-carboxamide (these compounds may be prepared from the methods described in WO2017207362A1 , WO2019105933A1 , W02020109511A1 , WO2021244952A1); N-methoxy-N-[[4-[5- (trifluoromethyl)-l ,2,4-oxadiazol-3-yl]phenyl]methyl]cyclopropanecarboxamide; N,2-dimethoxy-N-[[4-[5- (trifluoromethyl)-l ,2,4-oxadiazol-3-yl]phenyl]methyl]propanamide, or the (/?) or (S) enantiomer or mixtures thereof (these compound may be prepared from the methods described in WO 2017/055473); or thiamethoxam, or a salt, enantiomer, tautomer or N-oxide thereof, for controlling or preventing infestation of plants by the phytopathogenic microorganism Corynespora cassiicola. Preferably said plants are selected from cotton, corn, or soybean. In one embodiment said plants are genetically modified plants, preferably Bt plants, more preferably Bt soybean plant, Bt cotton or Bt corn.
In a preferred composition according to this aspect of the invention, component (A) is ethyl 1 -[[4-[(Z)-2-ethoxy- 3,3,3-trifluoro-prop-1-enoxy]phenyl]methyl]pyrazole-4-carboxylate (compound l-A), or a salt, enantiomer, tautomer or N-oxide thereof, and component (B) is selected from the group consisting of benzovindiflupyr, fluxapyroxad, pydiflumetofen, isopyrazam, fluopyram, penthiopyrad, sedaxane, bixafen, difenoconazole,
cyproconazole, tebuconazole, hexaconazole, prothioconazole, propiconazole, epoxiconazole, metconazole, tetraconazole, fluoxytioconazole, 2-[2-chloro-4-(4-chlorophenoxy)phenyl]-2-hydroxy-3-(1 , 2 ,4-tri azo 1-1 - yl)propanoate, methyl 3-[(4-chlorophenyl)methyl]-2-hydroxy-1-methyl-2-(1 ,2,4-triazol-1 - ylmethyl)cyclopentanecarboxylate, flutriafol, mefentrifluconazole, ipconazole, paclobutrazol, azoxystrobin, trifloxystrobin, picoxystrobin, pyraclostrobin, metalaxyl-M, fenpropidin, fenpropimorph, cyprodinil, spiroxamine, mancozeb, chlorothalonil, folpet, copper-oxychloride, copper-hydroxide, sulphur, oxathiapiprolin, ipflufenoquin, quinofumelin, mandipropamid, fluazinam, fludioxinil, fosetyl-aluminium, acibenzolar-S-methyl, enestrobin, inpyrfluxam, fluindapyr, isoflucypram, metyltetraprole, florylpicoxamid, metarylpicoxamide, flefunoxadiazam isofetamid, procymidone, carbendazim, fenhexamid, prochloraz, prohexadione-calcium, melaluca alternifolia oil (an extract of the tea tree plant Melaluca alternifolia (commercially available as Timorex Gold®, which is a broad-spectrum botanical biofungicide)), N'-[5-bromo-2-methyl-6-(1 -methyl-2-propoxy-ethoxy)-3-pyridyl]-N- ethyl-N-methylformamidine, cyclobutrifluram calcium phosphonate, cis-jasmone, trinexapac-ethyl, glyphosate, 2,4-D (2,4-dichlorophenoxyacetic acid); feneptamidoquin, methyl (Z)-3-methoxy-2-(2-methyl-5-phenyl- phenoxy)prop-2-enoate (this compound may be prepared from methods as described in JP2023078251), methyl (Z)-2-(5-cyclopentyl-2-methyl-phenoxy)-3-methoxy-prop-2-enoate; methyl (Z)-2-(5-cyclohexyl-2- methyl-phenoxy)-3-methoxy-prop-2-enoate (these compounds may be prepared from the methods described in W02020/193387); (5R)-3-[3-(3-chloro-2-fluoro-phenoxy)-6-methyl-pyridazin-4-yl]-5-[(2-chloro-4-methyl- phenyl)methyl]-5,6-dihydro-4H-1 ,2,4-oxadiazine; (5S)-3-[3-(3-chloro-2-fluoro-phenoxy)-6-methyl-pyridazin-4- yl]-5-[(2-chloro-4-methyl-phenyl) methyl]-5,6-dihydro-4H-1 ,2,4-oxadiazine; 3-[3-(3-chloro-2-fluoro-phenoxy)-6- methyl-pyridazin-4-yl]-5-[(2-chloro-4-methyl-phenyl)methyl]-5,6-dihydro-4H-1 ,2,4-oxadiazine (these compounds may be prepared from the methods described in WO 2020/127780, WO 2021/255070); 2-[cyano- (2,6-difluoro-4-pyridyl)amino]-5-methyl-N-spiro[3.4]octan-3-yl-thiazole-4-carboxamide; 2-[cyano-(2,6-difluoro- 4-pyridyl)amino]-N-(2,2-dimethylcyclobutyl)-5-methyl-thiazole-4-carboxamide (these compounds may be prepared from the methods described in WO2017207362A1 , WO2019105933A1 , W02020109509A1); 2-[(2,6- difluoro-4-pyridyl)-(2-methoxyacetyl)amino]-N-(2,2-dimethylcyclobutyl)-5-methyl-thiazole-4-carboxamide; 2- [(2,6-difluoro-4-pyridyl)-(tetrahydropyran-4-carbonyl)amino]-N-(2,2-dimethylcyclobutyl)-5-methyl-thiazole-4- carboxamide; 2-[(2,6-difluoro-4-pyridyl)-(oxetane-3-carbonyl)amino]-N-(2,2-dimethylcyclobutyl)-5-methyl- thiazole-4-carboxamide; 2-[acetyl-(2,6-difluoro-4-pyridyl)amino]-N-(2,2-dimethylcyclobutyl)-5-methyl-thiazole- 4-carboxamide; 2-[acetyl-(2,6-difluoro-4-pyridyl)amino]-5-methyl-N-spiro[3.4]octan-3-yl]-thiazole-4- carboxamide; 2-[(2,6-difluoro-4-pyridyl)-(2-methylpropanoyl)amino]-N-(2,2-dimethylcyclobutyl)-5-methyl- thiazole-4-carboxamide (these compounds may be prepared from the methods described in WO2017207362A1 , WO2019105933A1 , W02020109511A1 , WO2021244952A1); N-methoxy-N-[[4-[5- (trifluoromethyl)-l ,2,4-oxadiazol-3-yl]phenyl]methyl]cyclopropanecarboxamide; N,2-dimethoxy-N-[[4-[5- (trifluoromethyl)-l ,2,4-oxadiazol-3-yl]phenyl]methyl]propanamide, or the (/?) or (S) enantiomer or mixtures thereof (these compound may be prepared from the methods described in WO 2017/055473); or thiamethoxam, or a salt, enantiomer, tautomer or N-oxide thereof, for controlling or preventing infestation of plants by the phytopathogenic microorganism Corynespora cassiicola. Preferably said plants are selected from
cotton, corn, or soybean. In one embodiment said plants are genetically modified plants, preferably Bt plants, more preferably Bt soybean plant, Bt cotton or Bt corn.
In another preferred composition according to the invention, component (A) is ethyl 1-[[4-[[2-(trifluoromethyl)-
1 .3-dioxolan-2-yl]methoxy]phenyl]methyl]pyrazole-4-carboxylate (compound l-B), or a salt, enantiomer, tautomer or N-oxide thereof, and component (B) is selected from the group consisting of benzovindiflupyr, fluxapyroxad, pydiflumetofen, isopyrazam, fluopyram, penthiopyrad, sedaxane, bixafen, difenoconazole, cyproconazole, tebuconazole, hexaconazole, prothioconazole, propiconazole, epoxiconazole, metconazole, tetraconazole, fluoxytioconazole, 2-[2-chloro-4-(4-chlorophenoxy)phenyl]-2-hydroxy-3-(1 , 2 ,4-tri azo 1-1 - yl)propanoate, methyl 3-[(4-chlorophenyl)methyl]-2-hydroxy-1-methyl-2-(1 ,2,4-triazol-1 - ylmethyl)cyclopentanecarboxylate, flutriafol, mefentrifluconazole, ipconazole, paclobutrazol, azoxystrobin, trifloxystrobin, picoxystrobin, pyraclostrobin, metalaxyl-M, fenpropidin, fenpropimorph, cyprodinil, spiroxamine, mancozeb, chlorothalonil, folpet, copper-oxychloride, copper-hydroxide, sulphur, oxathiapiprolin, ipflufenoquin, quinofumelin, mandipropamid, fluazinam, fludioxinil, fosetyl-aluminium, acibenzolar-S-methyl, enestrobin, inpyrfluxam, fluindapyr, isoflucypram, metyltetraprole, florylpicoxamid, metarylpicoxamide, flefunoxadiazam isofetamid, procymidone, carbendazim, fenhexamid, prochloraz, prohexadione-calcium, melaluca alternifolia oil (an extract of the tea tree plant Melaluca alternifolia (commercially available as Timorex Gold®, which is a broad-spectrum botanical biofungicide)), N'-[5-bromo-2-methyl-6-(1 -methyl-2-propoxy-ethoxy)-3-pyridyl]-N- ethyl-N-methylformamidine, cyclobutrifluram calcium phosphonate, cis-jasmone, trinexapac-ethyl, glyphosate,
2.4-D (2,4-dichlorophenoxyacetic acid) feneptamidoquin, methyl (Z)-3-methoxy-2-(2-methyl-5-phenyl- phenoxy)prop-2-enoate (this compound may be prepared from methods as described in JP2023078251), methyl (Z)-2-(5-cyclopentyl-2-methyl-phenoxy)-3-methoxy-prop-2-enoate; methyl (Z)-2-(5-cyclohexyl-2- methyl-phenoxy)-3-methoxy-prop-2-enoate (these compounds may be prepared from the methods described in W02020/193387); (5R)-3-[3-(3-chloro-2-fluoro-phenoxy)-6-methyl-pyridazin-4-yl]-5-[(2-chloro-4-methyl- phenyl)methyl]-5,6-dihydro-4H-1 ,2,4-oxadiazine; (5S)-3-[3-(3-chloro-2-fluoro-phenoxy)-6-methyl-pyridazin-4- yl]-5-[(2-chloro-4-methyl-phenyl) methyl]-5,6-dihydro-4H-1 ,2,4-oxadiazine; 3-[3-(3-chloro-2-fluoro-phenoxy)-6- methyl-pyridazin-4-yl]-5-[(2-chloro-4-methyl-phenyl)methyl]-5,6-dihydro-4H-1 ,2,4-oxadiazine (these compounds may be prepared from the methods described in WO 2020/127780, WO 2021/255070); 2-[cyano- (2,6-difluoro-4-pyridyl)amino]-5-methyl-N-spiro[3.4]octan-3-yl-thiazole-4-carboxamide; 2-[cyano-(2,6-difluoro- 4-pyridyl)amino]-N-(2,2-dimethylcyclobutyl)-5-methyl-thiazole-4-carboxamide (these compounds may be prepared from the methods described in WO2017207362A1 , WO2019105933A1 , W02020109509A1); 2-[(2,6- difluoro-4-pyridyl)-(2-methoxyacetyl)amino]-N-(2,2-dimethylcyclobutyl)-5-methyl-thiazole-4-carboxamide; 2- [(2,6-difluoro-4-pyridyl)-(tetrahydropyran-4-carbonyl)amino]-N-(2,2-dimethylcyclobutyl)-5-methyl-thiazole-4- carboxamide; 2-[(2,6-difluoro-4-pyridyl)-(oxetane-3-carbonyl)amino]-N-(2,2-dimethylcyclobutyl)-5-methyl- thiazole-4-carboxamide; 2-[acetyl-(2,6-difluoro-4-pyridyl)amino]-N-(2,2-dimethylcyclobutyl)-5-methyl-thiazole- 4-carboxamide; 2-[acetyl-(2,6-difluoro-4-pyridyl)amino]-5-methyl-N-spiro[3.4]octan-3-yl]-thiazole-4- carboxamide; 2-[(2,6-difluoro-4-pyridyl)-(2-methylpropanoyl)amino]-N-(2,2-dimethylcyclobutyl)-5-methyl- thiazole-4-carboxamide (these compounds may be prepared from the methods described in WO2017207362A1 , WO2019105933A1 , W02020109511A1 , WO2021244952A1); N-methoxy-N-[[4-[5-
(trifluoromethyl)-l ,2,4-oxadiazol-3-yl]phenyl]methyl]cyclopropanecarboxamide; N,2-dimethoxy-N-[[4-[5- (trifluoromethyl)-l ,2,4-oxadiazol-3-yl]phenyl]methyl]propanamide, or the (/?) or (S) enantiomer or mixtures thereof (these compound may be prepared from the methods described in WO 2017/055473); or thiamethoxam,or a salt, enantiomer, tautomer or N-oxide thereof, for controlling or preventing infestation of plants by the phytopathogenic microorganism Corynespora cassiicola. Preferably said plants are selected from cotton, corn, or soybean. In one embodiment said plants are genetically modified plants, preferably Bt plants, more preferably Bt soybean plant, Bt cotton or Bt corn.
In general, the weight ratio of component (A) to component (B) may be from 1000: 1 to 1 :1000, may be from 100:1 to 1 :100, preferably from 50:1 to 1 :50, more preferably from 20:1 to 1 :40, even more preferably from 15:1 to 1 :30, still more preferably from 12:1 to 1 :25, or from 10:1 to 1 :20, or from 10:1 to 1 :10, or from 5:1 and 1 :15, or from 5:1 to 1 :5, or from 4:1 to 1 :4, or from 3:1 to 1 :10, or from 3:1 to 1 :3, or from 2:1 to 1 :5, or 1 :1.
In any of the compositions according to the invention, the composition may comprise an additional active ingredient component (C), which is different to component (B), and is selected from the group consisting of metyltetrapole, azoxystrobin, trifloxystrobin, picoxystrobin, pyraclostrobin, metominostrobin, prothioconazole, cyproconazole, difenoconazole, tebuconazole, propiconazole, epoxiconazole, hexaconazole, mefentrifluconazole, metconazole, benzovindiflupyr, pydiflumetofen, fluxapyroxad, sedaxane, bixafen, isopyrazam, fluopyram, fluindapyr, isoflucypram, inpyrfluxam, quinofumelin, ipflufenoquin, aminopyrifen, fluazinam, fludioxonil, fenpicoxamid, florylpicoxamid, feneptamidoquin, fenpropidin, fenpropimorph, mancozeb, chlorothalonil, cyclobutrifluram, copper-oxychloride, copper-hydroxide, thiamethoxam, (Z,2E)-5-[1- (2,4-dichlorophenyl)pyrazol-3-yl]oxy-2-methoxyimino-N,3-dimethyl-pent-3-enamide, (Z,2E)-5-[1-(4- chlorophenyl)pyrazol-3-yl]oxy-2-methoxyimino-N,3-dimethyl-pent-3-enamide, 4-[[6-[2-(2,4-difluorophenyl)- 1 ,1-difluoro-2-hydroxy-3-(5-thioxo-4H-1 ,2,4-triazol-1-yl)propyl]-3-pyridyl]oxy]benzonitrile, methyl 3-[(4- chlorophenyl)methyl]-2-hydroxy-1-methyl-2-(1 ,2,4-triazol-1-ylmethyl)cyclopentanecarboxylate, 2-[6-(4- bromophenoxy)-2-(trifluoromethyl)-3-pyridyl]-1-(1 ,2,4-triazol-1-yl)propan-2-ol, 2-[6-(4-chlorophenoxy)-2- (trifluoromethyl)-3-pyridyl]-1-(1 ,2,4-triazol-1-yl)propan-2-ol, N-methyl-4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3- yl]benzenecarbothioamide, N-methyl-4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3-yl]benzamide, 2,2-difluoro-N- methyl-2-[4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3-yl]phenyl]acetamide, N-[N-methoxy-C-methyl- carbonimidoyl]-4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3-yl]benzamide, N-[(E)-methoxyiminomethyl]-4-[5- (trifluoromethyl)-l ,2,4-oxadiazol-3-yl]benzamide, N-[4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3-yl]phenyl] cyclopropanecarboxamide, N-(2-fluorophenyl)-4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3-yl]benzamide, , N- [(1 R)-1 -benzyl-1 ,3-dimethylbutyl]-8-fluoroquinoline-3-carboxamide, N-[(1 S)-1 -benzyl-1 ,3-dimethylbutyl]-8- fluoroquinoline-3-carboxamide (these compound may be prepared from the methods described in WO2017/153380), methyl (Z)-3-methoxy-2-(2-methyl-5-phenyl-phenoxy)prop-2-enoate (this compound may be prepared from methods as described in JP2023078251), methyl (Z)-2-(5-cyclopentyl-2-methyl-phenoxy)-3- methoxy-prop-2-enoate; methyl (Z)-2-(5-cyclohexyl-2-methyl-phenoxy)-3-methoxy-prop-2-enoate (these compounds may be prepared from the methods described in W02020/193387); (5R)-3-[3-(3-chloro-2-fluoro- phenoxy)-6-methyl-pyridazin-4-yl]-5-[(2-chloro-4-methyl-phenyl)methyl]-5,6-dihydro-4H-1 ,2,4-oxadiazine;
(5S)-3-[3-(3-chloro-2-fluoro-phenoxy)-6-methyl-pyridazin-4-yl]-5-[(2-chloro-4-methyl-phenyl) methyl]-5,6-
dihydro-4H-1 ,2,4-oxadiazine; 3-[3-(3-chloro-2-fluoro-phenoxy)-6-methyl-pyridazin-4-yl]-5-[(2-chloro-4-methyl- phenyl)methyl]-5,6-dihydro-4H-1 ,2,4-oxadiazine (these compounds may be prepared from the methods described in WO 2020/127780, WO 2021/255070); 2-[cyano-(2,6-difluoro-4-pyridyl)amino]-5-methyl-N- spiro[3.4]octan-3-yl-thiazole-4-carboxamide; 2-[cyano-(2,6-difluoro-4-pyridyl)amino]-N-(2,2- dimethylcyclobutyl)-5-methyl-thiazole-4-carboxamide (these compounds may be prepared from the methods described in WO2017207362A1 , WO2019105933A1 , W02020109509A1); 2-[(2,6-difluoro-4-pyridyl)-(2- methoxyacetyl)amino]-N-(2,2-dimethylcyclobutyl)-5-methyl-thiazole-4-carboxamide; 2-[(2,6-difluoro-4- pyridyl)-(tetrahydropyran-4-carbonyl)amino]-N-(2,2-dimethylcyclobutyl)-5-methyl-thiazole-4-carboxamide; 2- [(2,6-difluoro-4-pyridyl)-(oxetane-3-carbonyl)amino]-N-(2,2-dimethylcyclobutyl)-5-methyl-thiazole-4- carboxamide; 2-[acetyl-(2,6-difluoro-4-pyridyl)amino]-N-(2,2-dimethylcyclobutyl)-5-methyl-thiazole-4- carboxamide; 2-[acetyl-(2,6-difluoro-4-pyridyl)amino]-5-methyl-N-spiro[3.4]octan-3-yl]-thiazole-4- carboxamide; 2-[(2,6-difluoro-4-pyridyl)-(2-methylpropanoyl)amino]-N-(2,2-dimethylcyclobutyl)-5-methyl- thiazole-4-carboxamide (these compounds may be prepared from the methods described in WO2017207362A1 , WO2019105933A1 , W02020109511A1 , WO2021244952A1); N-methoxy-N-[[4-[5- (trifluoromethyl)-l ,2,4-oxadiazol-3-yl]phenyl]methyl]cyclopropanecarboxamide or the (/?) or (S) enantiomer or mixtures thereof; and N,2-dimethoxy-N-[[4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3- yl]phenyl]methyl]propanamide or the (/?) or (S) enantiomer or mixtures thereof (these compound may be prepared from the methods described in WO 2017/055473);, for controlling or preventing infestation of plants by the phytopathogenic microorganism Corynespora cassiicola. Preferably said plants are selected from cotton, corn, or soybean. In one embodiment said plants are genetically modified plants, preferably Bt plants, more preferably Bt soybean plant, Bt cotton or Bt corn.
The component (C) compounds are referred to herein and above by a so-called "ISO common name" or another "common name" being used in individual cases or a trademark name. The component (C) compounds are known and are commercially available and/or can be prepared using procedures known in the art.
In another embodiment of the present invention, there is provided a fungicidal composition comprising a mixture of component (A) and a component (B) and a component (C) as active ingredients, wherein component (A) is selected from a compound of formula (I), (l-A), or (l-B), and component (B) and (C) are a compound selected from the group consisting of benzovindiflupyr, fluxapyroxad, pydiflumetofen, isopyrazam, fluopyram, penthiopyrad, sedaxane, bixafen, difenoconazole, cyproconazole, tebuconazole, hexaconazole, prothioconazole, propiconazole, epoxiconazole, metconazole, tetraconazole, fluoxytioconazole, 2-[2-chloro-4- (4-chlorophenoxy)phenyl]-2-hydroxy-3-(1 , 2 ,4-tri azo 1-1 -yl)propanoate, methyl 3-[(4-chlorophenyl)methyl]-2- hydroxy-1-methyl-2-(1 ,2,4-triazol-1-ylmethyl)cyclopentanecarboxylate, flutriafol, mefentrifluconazole, ipconazole, paclobutrazol, azoxystrobin, trifloxystrobin, picoxystrobin, pyraclostrobin, metalaxyl-M, fenpropidin, fenpropimorph, cyprodinil, spiroxamine, mancozeb, chlorothalonil, folpet, copper-oxychloride, copper-hydroxide, sulphur, oxathiapiprolin, ipflufenoquin, quinofumelin, mandipropamid, fluazinam, fludioxinil, fosetyl-aluminium, acibenzolar-S-methyl, enestrobin, inpyrfluxam, fluindapyr, isoflucypram, metyltetraprole, florylpicoxamid, metarylpicoxamide, flefunoxadiazam isofetamid, procymidone, carbendazim, fenhexamid, prochloraz, prohexadione-calcium, melaluca alternifolia oil (an extract of the tea tree plant Melaluca alternifolia
(commercially available as Timorex Gold®, which is a broad-spectrum botanical biofungicide)), N'-[5-bromo- 2-methyl-6-(1-methyl-2-propoxy-ethoxy)-3-pyridyl]-N-ethyl-N-methylformamidine, cyclobutrifluram calcium phosphonate, cis-jasmone, trinexapac-ethyl, glyphosate, 2,4-D (2,4-dichlorophenoxyacetic acid); N-[(1 R)-1- benzyl-1 ,3-dimethylbutyl]-8-fluoroquinoline-3-carboxamide, N-[(1 S)-1 -benzyl-1 ,3-dimethylbutyl]-8-fluoro- quinoline-3-carboxamide (these compound may be prepared from the methods described in WO2017/153380), methyl (Z)-3-methoxy-2-(2-methyl-5-phenyl-phenoxy)prop-2-enoate (this compound may be prepared from methods as described in JP2023078251), methyl (Z)-3-methoxy-2-(2-methyl-5-phenyl-phenoxy)prop-2-enoate (this compound may be prepared from methods as described in JP2023078251), methyl (Z)-2-(5-cyclopentyl- 2-methyl-phenoxy)-3-methoxy-prop-2-enoate; methyl (Z)-2-(5-cyclohexyl-2-methyl-phenoxy)-3-methoxy-prop- 2-enoate (these compounds may be prepared from the methods described in W02020/193387); (5R)-3-[3-(3- chloro-2-fluoro-phenoxy)-6-methyl-pyridazin-4-yl]-5-[(2-chloro-4-methyl-phenyl)methyl]-5,6-dihydro-4H-1 ,2,4- oxadiazine; (5S)-3-[3-(3-chloro-2-fluoro-phenoxy)-6-methyl-pyridazin-4-yl]-5-[(2-chloro-4-methyl-phenyl) methyl]-5,6-dihydro-4H-1 ,2,4-oxadiazine; 3-[3-(3-chloro-2-fluoro-phenoxy)-6-methyl-pyridazin-4-yl]-5-[(2- chloro-4-methyl-phenyl)methyl]-5,6-dihydro-4H-1 ,2,4-oxadiazine (these compounds may be prepared from the methods described in WO 2020/127780, WO 2021/255070); 2-[cyano-(2,6-difluoro-4-pyridyl)amino]-5-methyl- N-spiro[3.4]octan-3-yl-thiazole-4-carboxamide; 2-[cyano-(2,6-difluoro-4-pyridyl)amino]-N-(2,2- dimethylcyclobutyl)-5-methyl-thiazole-4-carboxamide (these compounds may be prepared from the methods described in WO2017207362A1 , WO2019105933A1 , W02020109509A1); 2-[(2,6-difluoro-4-pyridyl)-(2- methoxyacetyl)amino]-N-(2,2-dimethylcyclobutyl)-5-methyl-thiazole-4-carboxamide; 2-[(2,6-difluoro-4- pyridyl)-(tetrahydropyran-4-carbonyl)amino]-N-(2,2-dimethylcyclobutyl)-5-methyl-thiazole-4-carboxamide; 2- [(2,6-difluoro-4-pyridyl)-(oxetane-3-carbonyl)amino]-N-(2,2-dimethylcyclobutyl)-5-methyl-thiazole-4- carboxamide; 2-[acetyl-(2,6-difluoro-4-pyridyl)amino]-N-(2,2-dimethylcyclobutyl)-5-methyl-thiazole-4-carbox- amide; 2-[acetyl-(2,6-difluoro-4-pyridyl)amino]-5-methyl-N-spiro[3.4]octan-3-yl]-thiazole-4-carboxamide; 2- [(2,6-difluoro-4-pyridyl)-(2-methylpropanoyl)amino]-N-(2,2-dimethylcyclobutyl)-5-methyl-thiazole-4-carbox- amide (these compounds may be prepared from the methods described in WO2017207362A1 , WO2019105933A1 , W02020109511A1 , WO2021244952A1); N-methoxy-N-[[4-[5-(trifluoromethyl)-1 ,2,4- oxadiazol-3-yl]phenyl]methyl]cyclopropanecarboxamide or the (R) or (S) enantiomer or mixtures thereof; and N,2-dimethoxy-N-[[4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3-yl]phenyl]methyl]propanamide or the (R) or (S) enantiomer or mixtures thereof (these compound may be prepared from the methods described in WO 2017/055473) or thiamethoxam, or a salt, enantiomer, tautomer or N-oxide thereof, wherein component (B) and (C) are not the same compound, for controlling or preventing infestation of plants by the phytopathogenic microorganism Corynespora cassiicola. Preferably said plants are selected from cotton, corn, or soybean. In one embodiment said plants are genetically modified plants, preferably Bt plants, more preferably Bt soybean plant, Bt cotton or Bt corn.
In a preferred composition according to this aspect of the invention, component (A) is N-(2-fluorophenyl)-4-[5- (trifluoromethyl)-l ,2,4-oxadiazol-3-yl]benzamide (compound l-A), or a salt, enantiomer, tautomer or N-oxide thereof, and component (B) and (C) are a compound selected from the group consisting of benzovindiflupyr, fluxapyroxad, pydiflumetofen, isopyrazam, fluopyram, penthiopyrad, sedaxane, bixafen, difenoconazole,
cyproconazole, tebuconazole, hexaconazole, prothioconazole, propiconazole, epoxiconazole, metconazole, tetraconazole, fluoxytioconazole, 2-[2-chloro-4-(4-chlorophenoxy)phenyl]-2-hydroxy-3-(1 , 2 ,4-tri azo 1-1 - yl)propanoate, methyl 3-[(4-chlorophenyl)methyl]-2-hydroxy-1-methyl-2-(1 ,2,4-triazol-1-ylmethyl) cyclopentane carboxylate, flutriafol, mefentrifluconazole, ipconazole, paclobutrazol, azoxystrobin, trifloxystrobin, picoxystrobin, pyraclostrobin, metalaxyl-M, fenpropidin, fenpropimorph, cyprodinil, spiroxamine, mancozeb, chlorothalonil, folpet, copper-oxychloride, copper-hydroxide, sulphur, oxathiapiprolin, ipflufenoquin, quinofumelin, mandipropamid, fluazinam, fludioxinil, fosetyl-aluminium, acibenzolar-S-methyl, enestrobin, inpyrfluxam, fluindapyr, isoflucypram, metyltetraprole, florylpicoxamid, metarylpicoxamide, flefunoxadiazam isofetamid, procymidone, carbendazim, fenhexamid, prochloraz, prohexadione-calcium, melaluca alternifolia oil (an extract of the tea tree plant Melaluca alternifolia (commercially available as Timorex Gold®, which is a broad-spectrum botanical biofungicide)), N'-[5-bromo-2-methyl-6-(1 -methyl-2-propoxy-ethoxy)-3-pyridyl]-N- ethyl-N-methylformamidine, cyclobutrifluram calcium phosphonate, cis-jasmone, trinexapac-ethyl, glyphosate, 2,4-D (2,4-dichlorophenoxyacetic acid); N-[(1 R)-1-benzyl-1 ,3-dimethylbutyl]-8-fluoroquinoline-3-carboxamide, N-[(1 S)-1-benzyl-1 ,3-dimethylbutyl]-8-fluoroquinoline-3-carboxamide (these compound may be prepared from the methods described in WO2017/153380), methyl (Z)-3-methoxy-2-(2-methyl-5-phenyl-phenoxy)prop-2- enoate (this compound may be prepared from methods as described in JP2023078251), methyl (Z)-3-methoxy- 2-(2-methyl-5-phenyl-phenoxy)prop-2-enoate (this compound may be prepared from methods as described in JP2023078251), methyl (Z)-2-(5-cyclopentyl-2-methyl-phenoxy)-3-methoxy-prop-2-enoate; methyl (Z)-2-(5- cyclohexyl-2-methyl-phenoxy)-3-methoxy-prop-2-enoate (these compounds may be prepared from the methods described in W02020/193387); (5R)-3-[3-(3-chloro-2-fluoro-phenoxy)-6-methyl-pyridazin-4-yl]-5-[(2- chloro-4-methyl-phenyl)methyl]-5,6-dihydro-4H-1 ,2,4-oxadiazine; (5S)-3-[3-(3-chloro-2-fluoro-phenoxy)-6- methyl-pyridazin-4-yl]-5-[(2-chloro-4-methyl-phenyl) methyl]-5,6-dihydro-4H-1 ,2,4-oxadiazine; 3-[3-(3-chloro- 2-fluoro-phenoxy)-6-methyl-pyridazin-4-yl]-5-[(2-chloro-4-methyl-phenyl)methyl]-5,6-dihydro-4H-1 ,2,4- oxadiazine (these compounds may be prepared from the methods described in WO 2020/127780, WO 2021/255070); 2-[cyano-(2,6-difluoro-4-pyridyl)amino]-5-methyl-N-spiro[3.4]octan-3-yl-thiazole-4-carbox- amide; 2-[cyano-(2,6-difluoro-4-pyridyl)amino]-N-(2,2-dimethylcyclobutyl)-5-methyl-thiazole-4-carboxamide (these compounds may be prepared from the methods described in WO2017207362A1 , WO2019105933A1 , W02020109509A1); 2-[(2,6-difluoro-4-pyridyl)-(2-methoxyacetyl)amino]-N-(2,2-dimethylcyclobutyl)-5-methyl- thiazole-4-carboxamide; 2-[(2,6-difluoro-4-pyridyl)-(tetrahydropyran-4-carbonyl)amino]-N-(2,2-dimethyl- cyclobutyl)-5-methyl-thiazole-4-carboxamide; 2-[(2,6-difluoro-4-pyridyl)-(oxetane-3-carbonyl)amino]-N-(2,2- dimethylcyclobutyl)-5-methyl-thiazole-4-carboxamide; 2-[acetyl-(2,6-difluoro-4-pyridyl)amino]-N-(2,2-dimethyl- cyclobutyl)-5-methyl-thiazole-4-carboxamide; 2-[acetyl-(2,6-difluoro-4-pyridyl)amino]-5-methyl-N- spiro[3.4]octan-3-yl]-thiazole-4-carboxamide; 2-[(2,6-difluoro-4-pyridyl)-(2-methylpropanoyl)amino]-N-(2,2- dimethylcyclobutyl)-5-methyl-thiazole-4-carboxamide (these compounds may be prepared from the methods described in WO2017207362A1 , WO2019105933A1 , W02020109511A1 , WO2021244952A1); N-methoxy-N- [[4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3-yl]phenyl]methyl]cyclopropanecarboxamide or the (R) or (S) enantiomer or mixtures thereof; and N,2-dimethoxy-N-[[4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3- yl]phenyl]methyl]propanamide or the (R) or (S) enantiomer or mixtures thereof (these compound may be
prepared from the methods described in WO 2017/055473)or thiamethoxam, or a salt, enantiomer, tautomer or N-oxide thereof, wherein component (B) and (C) are not the same compound, for controlling or preventing infestation of plants by the phytopathogenic microorganism Corynespora cassiicola. Preferably said plants are selected from cotton, corn, or soybean. In one embodiment said plants are genetically modified plants, preferably Bt plants, more preferably Bt soybean plant, Bt cotton or Bt corn.
In another preferred composition according to this aspect of the invention, component (A) is N-(2-fluorophenyl)- 4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3-yl]benzamide (compound l-B), or a salt, enantiomer, tautomer or N- oxide thereof, and component (B) and (C) are a compound selected from the group consisting of benzovindiflupyr, fluxapyroxad, pydiflumetofen, isopyrazam, fluopyram, penthiopyrad, sedaxane, bixafen, difenoconazole, cyproconazole, tebuconazole, hexaconazole, prothioconazole, propiconazole, epoxiconazole, metconazole, tetraconazole, fluoxytioconazole, 2-[2-chloro-4-(4-chlorophenoxy)phenyl]-2-hydroxy-3-(1 ,2,4- triazol-1-yl)propanoate, methyl 3-[(4-chlorophenyl)methyl]-2-hydroxy-1-methyl-2-(1 ,2,4-triazol-1- ylmethyl)cyclopentanecarboxylate, flutriafol, mefentrifluconazole, ipconazole, paclobutrazol, azoxystrobin, trifloxystrobin, picoxystrobin, pyraclostrobin, metalaxyl-M, fenpropidin, fenpropimorph, cyprodinil, spiroxamine, mancozeb, chlorothalonil, folpet, copper-oxychloride, copper-hydroxide, sulphur, oxathiapiprolin, ipflufenoquin, quinofumelin, mandipropamid, fluazinam, fludioxinil, fosetyl-aluminium, acibenzolar-S-methyl, enestrobin, inpyrfluxam, fluindapyr, isoflucypram, metyltetraprole, florylpicoxamid, metarylpicoxamide, flefunoxadiazam isofetamid, procymidone, carbendazim, fenhexamid, prochloraz, prohexadione-calcium, melaluca alternifolia oil (an extract of the tea tree plant Melaluca alternifolia (commercially available as Timorex Gold®, which is a broad-spectrum botanical biofungicide)), N'-[5-bromo-2-methyl-6-(1 -methyl-2-propoxy-ethoxy)-3-pyridyl]-N- ethyl-N-methylformamidine, cyclobutrifluram calcium phosphonate, cis-jasmone, trinexapac-ethyl, glyphosate, 2,4-D (2,4-dichlorophenoxyacetic acid); N-[(1 R)-1-benzyl-1 ,3-dimethylbutyl]-8-fluoroquinoline-3-carboxamide, N-[(1 S)-1-benzyl-1 ,3-dimethylbutyl]-8-fluoroquinoline-3-carboxamide (these compound may be prepared from the methods described in WO2017/153380), methyl (Z)-3-methoxy-2-(2-methyl-5-phenyl-phenoxy)prop-2- enoate (this compound may be prepared from methods as described in JP2023078251), methyl (Z)-3-methoxy- 2-(2-methyl-5-phenyl-phenoxy)prop-2-enoate (this compound may be prepared from methods as described in JP2023078251), methyl (Z)-2-(5-cyclopentyl-2-methyl-phenoxy)-3-methoxy-prop-2-enoate; methyl (Z)-2-(5- cyclohexyl-2-methyl-phenoxy)-3-methoxy-prop-2-enoate (these compounds may be prepared from the methods described in W02020/193387); (5R)-3-[3-(3-chloro-2-fluoro-phenoxy)-6-methyl-pyridazin-4-yl]-5-[(2- chloro-4-methyl-phenyl)methyl]-5,6-dihydro-4H-1 ,2,4-oxadiazine; (5S)-3-[3-(3-chloro-2-fluoro-phenoxy)-6- methyl-pyridazin-4-yl]-5-[(2-chloro-4-methyl-phenyl) methyl]-5,6-dihydro-4H-1 ,2,4-oxadiazine; 3-[3-(3-chloro- 2-fluoro-phenoxy)-6-methyl-pyridazin-4-yl]-5-[(2-chloro-4-methyl-phenyl)methyl]-5,6-dihydro-4H-1 ,2,4- oxadiazine (these compounds may be prepared from the methods described in WO 2020/127780, WO 2021/255070); 2-[cyano-(2,6-difluoro-4-pyridyl)amino]-5-methyl-N-spiro[3.4]octan-3-yl-thiazole-4- carboxamide; 2-[cyano-(2,6-difluoro-4-pyridyl)amino]-N-(2,2-dimethylcyclobutyl)-5-methyl-thiazole-4-carbox- amide (these compounds may be prepared from the methods described in WO2017207362A1 , WO2019105933A1 , W02020109509A1); 2-[(2,6-difluoro-4-pyridyl)-(2-methoxyacetyl)amino]-N-(2,2- dimethylcyclobutyl)-5-methyl-thiazole-4-carboxamide; 2-[(2,6-difluoro-4-pyridyl)-(tetrahydropyran-4-
carbonyl)amino]-N-(2,2-dimethylcyclobutyl)-5-methyl-thiazole-4-carboxamide; 2-[(2,6-difluoro-4-pyridyl)- (oxetane-3-carbonyl)amino]-N-(2,2-dimethylcyclobutyl)-5-methyl-thiazole-4-carboxamide; 2-[acetyl-(2,6- difluoro-4-pyridyl)amino]-N-(2,2-dimethylcyclobutyl)-5-methyl-thiazole-4-carboxamide; 2-[acetyl-(2,6-difluoro- 4-pyridyl)amino]-5-methyl-N-spiro[3.4]octan-3-yl]-thiazole-4-carboxamide; 2-[(2,6-difluoro-4-pyridyl)-(2- methylpropanoyl)amino]-N-(2,2-dimethylcyclobutyl)-5-methyl-thiazole-4-carboxamide (these compounds may be prepared from the methods described in WO2017207362A1 , WO2019105933A1 , W02020109511A1 , WO2021244952A1); N-methoxy-N-[[4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3-yl]phenyl]methyl]cyclopropane- carboxamide or the (/?) or (S) enantiomer or mixtures thereof; and N,2-dimethoxy-N-[[4-[5-(trifluoromethyl)- 1 ,2,4-oxadiazol-3-yl]phenyl]methyl]propanamide or the (/?) or (S) enantiomer or mixtures thereof (these compound may be prepared from the methods described in WO 2017/055473)or thiamethoxam, or a salt, enantiomer, tautomer or N-oxide thereof, wherein component (B) and (C) are not the same compound, for controlling or preventing infestation of plants by the phytopathogenic microorganism Corynespora cassiicola. Preferably said plants are selected from cotton, corn, or soybean. In one embodiment said plants are genetically modified plants, preferably Bt plants, more preferably Bt soybean plant, Bt cotton or Bt corn.
Components (B) and (C) in combination with component (A) may enhance the effectiveness of the latter against fungi, and vice versa. Additionally, the fungicidal compositions may be effective against a wider spectrum of fungal pathogens that can be combated with the individual active ingredients when used solely. Generally, the weight ratio of component (A) to the mixture of components (B) and (C) may be from 100:1 to 1 :100, or 50:1 to 1 :50, or 20:1 to 1 :20, or 10:1 to 1 :10, or 5:1 and 1 :5. Otherwise, the weight ratio of component (A) to the mixture of components (B) and (C) may be from 2:1 to 1 :2, or 4:1 to 2:1 , or 1 :1 , or 5:1 , or 5:2, or 5:3, or 5:4, or 4:1 , or 4:2, or 4:3, or 3:1 , or 3:2, or 2:1 , or 1 :5, or 2:5, or 3:5, or 4:5, or 1 :4, or 2:4, or 3:4, or 1 :3, or 2:3, or 1 :2, or 1 :600, or 1 :300, or 1 :150, or 1 :35, or 2:35, or 4:35, or 1 :75, or 2:75, or 4:75, or 1 :6000, or 1 :3000, or 1 :1500, or 1 :350, or 2:350, or 4:350, or 1 :750, or 2:750, or 4:750. Those mixing ratios are understood to include, on the one hand, ratios by weight and also, on other hand, molar ratios.
In embodiments of the invention where the composition comprise a component (A), a component (B) and a component (C), the weight ratio of component (A) to the sum of component (B) and component (C) may be from 100:1 to 1 :100, preferably from 50:1 to 1 :50, more preferably from 20:1 to 1 :40, even more preferably from 15:1 to 1 :30, still more preferably from 12:1 to 1 :25, or from 10:1 to 1 :20, or from 10:1 to 1 :10, or from 5:1 and 1 :15, or from 5:1 to 1 :5, or from 4:1 to 1 :4, or from 3:1 to 1 :10, or from 3:1 to 1 :3, or from 2:1 to 1 :5, or 1 :1 .
The component (B) compounds are referred to herein and above by a so-called "ISO common name" or another "common name" being used in individual cases or a trademark name. The component (B) compounds are known and are commercially available and/or can be prepared using procedures known in the art and/or procedures reported in the literature such as, for instance, WO 2015/155075 and WO 2016/202742, WO 2017/005710, WO 2018/108977, WO 2018/153707, WO 2018/098216, WO 2019/093522, WO 2018/145921 , WO 2015/185485, WO 2017/076742, WO 2017/081311 , WO 2017/085100, WO 2017/093019, WO 2017/178245, and WO 2017/211649.
In embodiments of the invention where the composition comprise a component A, wherein said component A is selected from a compound of formula (I), (l-A), or (l-B), a first component (B) and a second component (B), wherein said second component (B) is different than said first component (B), the weight ratio of component (A) to the sum of first component (B) and second component (B) may be from 100:1 to 1 :100, preferably from 50:1 to 1 :50, more preferably from 20:1 to 1 :40, even more preferably from 15:1 to 1 :30, still more preferably from 12:1 to 1 :25, or from 10:1 to 1 :20, or from 10:1 to 1 :10, or from 5:1 and 1 :15, or from 5:1 to 1 :5, or from 4:1 to 1 :4, or from 3:1 to 1 :10, or from 3:1 to 1 :3, or from 2:1 to 1 :5, or 1 :1.
Throughout this document the expression “composition” stands for the various mixtures or combinations of component A, wherein said component A is selected from a compound of formula (I), (l-A), or (l-B), and components (B) (including the above-defined embodiments), for example in a single “ready-mix” form, in a combined spray mixture composed from separate formulations of the single active ingredient components, such as a “tank-mix”, and in a combined use of the single active ingredients when applied in a sequential manner, i.e. one after the other with a reasonably short period, such as a few hours or days. The order of applying the component A, wherein said component A is selected from a compound of formula (I), (l-A), or (I- B), and component (B) is not essential for working the present invention.
In one embodiment of the present invention, there is provided a method for controlling harmful fungi, wherein the pest, their habitat, breeding grounds, their locus or the plants to be protected against pest attack, the soil or plant propagation material (preferably seed) are treated with a pesticidally effective amount of a compound of formula (I) according to the present invention.
In one embodiment of the present invention, there is provided a method for controlling harmful fungi, wherein the pest, their habitat, breeding grounds, their locus or the plants to be protected against pest attack, the soil or plant propagation material (preferably seed) are treated with a composition comprising a pesticidally effective amount of a compound of formula (I) according to the present invention.
Preferably the compounds of the present invention and compositions comprising said compound of formula (I) are suitable for controlling fungal plant diseases.
The compounds of the present invention are also important for controlling a multitude of fungi on various cultivated plants, such as bananas, cotton, vegetable species, for example cucumbers, beans, and cucurbits; cereals such as maize, wheat, rye, barley, rice, oats; grass coffee, potatoes, sweet potatoes, corn, fruit species, soybean, tobacco, tomatoes, grapevines, ornamental plants, sugar cane and also on a large number of seeds. In a preferred embodiment, the compounds of formula (I), (l-A), or (l-B) according to the present invention, are used in soybean, cotton, and corn. In a preferred embodiment, compounds of formula (I), (l-A), or (l-B) according to the present invention are used on genetically modified plants selected from soybean, cotton, and corn. Preferably said genetically modified plants selected from soybean, cotton, and corn are Bt plants, like Bt soybeans, Bt cotton and Bt corn.
In one embodiment, the compounds of formula (I), (l-A), or (l-B) according to the present invention, are particularly suitable for controlling cotton diseases caused by Alternaria spp. (Alternaria leaf spot), Cercospora
spp. (Cercospora leaf spots), Corynespora cassiicola (leaf spots); Ascochyta gossypii (ascochyta blight of cotton); Fusarium wilt in cotton (Gossypium hirsutum L.) caused by Fusarium oxysporum f. sp..
Preferably, the compounds of formula (I), (l-A), or (l-B) according to the present invention, are particularly suitable for controlling cotton diseases caused by Corynespora cassiicola (leaf spots).
In one embodiment, the compounds of formula (I), (l-A), or (l-B) according to the present invention, are suitable for controlling the following fungal diseases on soybeans: Altemaria spp. (Alternaria leaf spot); Cercospora spp. (Cercospora leaf spots), e.g., C. sojina or C. kikuchii Colletotrichum (teleomorph: Glomerella) spp. (anthracnose), e.g., C. truncatum or C. gloeosporioides); Corynespora cassiicola (leaf spots); Dematophora (teleomorph: Rosellinia) necatrix (root and stem rot); Diaporthe spp., e.g., D. phaseolorum (damping off); Fusarium (teleomorph: Gibberella) spp. (wilt, root or stem rot), e.g. F. tucumaniae and F. brasiliense each causing sudden death syndrome on soybeans; Macrophomina phaseolina (syn. phaseoli) (root and stem rot); Microsphaera diffusa (powdery mildew); Peronospora spp. (downy mildew), e.g., P. manshurica; Phakopsora pachyrhizi and P. meibomiae (soybean rust); Phialophora spp., e.g., P. gregata. stem rot; Phomopsis spp., e.g., stem rot: P. phaseoli (teleomorph: Diaporthe phaseolorum); Pythium spp. (damping-off); Phytophthora spp. (wilt, root, leaf, fruit and stem root), e.g., P. megasperma, syn. P. sojae); Rhizoctonia spp., e.g., R. solani (root and stem rot); Sclerotinia spp. (stem rot or white mold); Septoria spp., e.g., S. glycines (brown spot); S. rolfsii (syn. Athelia rolfsii); or Thielaviopsis spp. (black root rot).
Preferably, the compounds of formula (I), (l-A), or (l-B) according to the present invention, are suitable for controlling the soybean diseases caused by Corynespora cassiicola (leaf spots).
Preferably, the compounds of the invention or fungicidal compositions according to the present invention comprising said compounds may be used in controlling or preventing phytopathogenic diseases, especially phytopathogenic fungi (such as Corynespora cassiicola) on soybean plants.
In another embodiment of the invention, there is provided a method of controlling or preventing infestation of genetically modified plants by especially phytopathogenic fungi, especially such as Corynespora cassiicola, wherein a compound of formula (I) according to the present invention is applied to said plants. Preferably said genetically modified plants are selected from soybean, cotton, and maize. In another preferred embodiment said genetically modified plants are BT plants, selected from Bt soybean, Bt cotton and Bt maize. In another preferred embodiment said genetically modified plants are BT plants, selected from Bt soybean, and Bt cotton.
Examples of commercially available genetically modified soybean plants, which can preferably be treated according to the invention, include, but not limited to, commercially available products such as plant seeds Intacta®, lntacta®2, Intacta® Roundup Ready™ 2 Pro (lntacta®RR2 PRO), lntacta®2 Xtend™, Cultivance, Conkesta Soybean, Conkesta Enlist E3™ Soybean, Enlist™ Soybean, Enlist E3™ Soybean, Roundup Ready™ Soybean, Roundup Ready™ 2 Xtend™, Genuity® Roundup Ready™ 2 Xtend™, Genuity® Roundup Ready 2 Yield™, Herbicide-tolerant Soybean line, Optimum GAT™, Liberty Link™ Soybean, Vistive Gold™, Verdeca HB4 Soybean, Treus™, Plenish™.
In one embodiment of the invention, there is provided a method of controlling or preventing infestation of genetically modified soybean plants by especially phytopathogenic fungi, especially such as Corynespora cassiicola, wherein a compound of formula (I) according to the present invention is applied to said soybean plants, and wherein said soybean plants are BT soybeans. Preferably said Bt soybean plants are selected from Intacta RR2 PRO®, or Conkesta Enlist E3®.
In a preferred embodiment of the invention, there is provided a method for controlling Corynespora cassiicola on soybean plants, wherein said soybean plants are genetically modified soybean plants, preferably Bt soybean plants, characterized by comprising the step of contacting the plant, parts thereof, propagation material thereof, the pests, their food source, habitat, or breeding ground with a compound selected from ethyl 1-[[4-[(Z)-2-ethoxy-3,3,3-trifluoro-prop-1-enoxy]phenyl]methyl]pyrazole-4-carboxylate according to structure (I-
A)
ethyl 1-[[4-[[2-(trifluoromethyl)-1 ,3-dioxolan-2-yl]methoxy]phenyl]methyl]pyrazole-4-carboxylate according to structure (l-B)
In a more preferred embodiment the invention, there is provided a method for controlling Corynespora cassiicola on soybean plants, wherein said soybean plants are genetically modified soybean plants, preferably Bt soybean plants, characterized by comprising the step of contacting the plant, parts thereof, propagation material thereof, the pests, their food source, habitat, or breeding ground with a compound of formula (l-A).
In another more preferred embodiment of the invention, there is provided a method for controlling Corynespora cassiicola on soybean plants, wherein said soybean plants are genetically modified soybean plants, preferably Bt soybean plants, characterized by comprising the step of contacting the plant, parts thereof, propagation material thereof, the pests, their food source, habitat, or breeding ground with a compound of formula (l-B).
Preferably, the compounds of the invention or fungicidal compositions according to the present invention comprising said compounds may be used in controlling or preventing phytopathogenic diseases, especially phytopathogenic fungi (such as Corynespora cassiicola) on cotton plants.
Examples of commercially available genetically modified cotton plants, which can preferably be treated according to the invention, include, but not limited to, commercially available products such as plant seeds Bollgard®, Bollgard II ™ Cotton, LibertyLink®, Roundup Ready™, WideStrike™ Cotton, Bollgard® III x Roundup Ready™ Flex™, GlyTol™, GlyTol™ Liberty Link™, Glytol™ x Twinlink™, GlyTol LibertyLink TwinLink® Plus, Fibermax™ Liberty Link™, Bollgard™ Cotton, Ingard™, Roundup Ready™ Bollgard™ Cotton, Roundup Ready™ Flex™ Cotton, Roundup Ready™ Flex™ Bollgard II™ Cotton, TwinLink™ Cotton, BXN™ Cotton.
In one embodiment of the invention, there is provided a method of controlling or preventing infestation of genetically modified cotton plants by especially phytopathogenic fungi, especially such as Corynespora cassiicola, wherein a compound of formula (I) according to the present invention is applied to said cotton plants, and wherein said cotton plants are BT cotton. Preferably said Bt cotton plants are selected from WideStrike™ Cotton, Bollgard® III x Roundup Ready™ Flex™, GlyTol™ Liberty Link™, GlyTol LibertyLink TwinLink® Plus, Bollgard II ™ Cotton, Bollgard™ Cotton Ingard™, Roundup Ready™ Bollgard™ Cotton, Roundup Ready™ Flex™ Cotton, and TwinLink™ Cotton.
In a preferred embodiment of the invention, there is provided a method for controlling Corynespora cassiicola on cotton plants, wherein said cotton plants are genetically modified cotton plants, preferably Bt cotton plants, characterized by comprising the step of contacting the plant, parts thereof, propagation material thereof, the pests, their food source, habitat, or breeding ground with a compound selected from ethyl 1 -[[4-[(Z)-2-ethoxy- 3,3,3-trifluoro-prop-1-enoxy]phenyl]methyl]pyrazole-4-carboxylate according to structure (l-A)
ethyl 1-[[4-[[2-(trifluoromethyl)-1 ,3-dioxolan-2-yl]methoxy]phenyl]methyl]pyrazole-4-carboxylate according to structure (l-B)
In a more preferred embodiment the invention, there is provided a method for controlling Corynespora cassiicola on cotton plants, wherein said cotton plants are genetically modified cotton plants, preferably Bt cotton plants, characterized by comprising the step of contacting the plant, parts thereof, propagation material thereof, the pests, their food source, habitat, or breeding ground with a compound of formula (l-A).
In another more preferred embodiment of the invention, there is provided a method for controlling Corynespora cassiicola on cotton plants, wherein said cotton plants are genetically modified cotton plants, preferably Bt cotton plants, characterized by comprising the step of contacting the plant, parts thereof, propagation material thereof, the pests, their food source, habitat, or breeding ground with a compound of formula (l-B).
The compounds of formula (I), (l-A), or (l-B) according to the present invention, or fungicidal compositions comprising said compounds according to the present invention, may be used in controlling or preventing phytopathogenic diseases, especially phytopathogenic fungi (such as Corynespora cassiicola) on soybean plants.
In one embodiment, the compounds of formula (I), (l-A), or (l-B) according to the present invention, or fungicidal compositions comprising said compounds according to the present invention, may be used in controlling or preventing phytopathogenic diseases, especially phytopathogenic fungi (such as Corynespora cassiicola) on genetically modified soybean plants, preferably Bt soybean plants.
The compounds of formula (I), (l-A), or (l-B) according to the present invention, or fungicidal compositions comprising said compounds according to the present invention, may be used in controlling or preventing phytopathogenic diseases, especially phytopathogenic fungi (such as Corynespora cassiicola) on cotton plants.
The compounds of formula (I), (l-A), or (l-B) according to the present invention, or fungicidal compositions comprising said compounds according to the present invention, may be used in controlling or preventing phytopathogenic diseases, especially phytopathogenic fungi (such as Corynespora cassiicola) on genetically modified cotton plants, preferably Bt cotton plants.
Under certain circumstances, fungicidal compositions according to the present invention comprising a compound of formula (I), (l-A), or (l-B) when used in controlling or preventing phytopathogenic diseases, especially phytopathogenic fungi (such as Corynespora cassiicola) on plants selected from soybean and cotton (in particular any of the transgenic plants as described above), may display a synergistic interaction between the active ingredients.
Transgenic soybean plants expressing toxins, for example insecticidal proteins such as delta-endotoxins, e.g., CrylAc (CrylAc Bt protein). Accordingly, this may include transgenic soybean plants comprising event MON87701 (disclosed in W02009/064652), event MON87701 x MON89788 (disclosed in WO2014/170327, e.g. commercially available as Intacta RR2 PRO® soybean), event MON87751 (disclosed in WO2014/201235), event DAS-44406-6 (e.g., commercially available as Enlist E3™, DAS-44406-6, disclosed in WO2012/075426), or event DAS-81419-2 (described in WO2013/016527, e.g., commercially available as Conkesta™ soybean); event DAS-81419-2 x DAS-44406-6 (e.g., commercially available as Conkesta™ Enlist E3™ Soybean).
Useful transgenic events in transgenic soybean plants, which can be treated according to the invention, include event DAS-44406-6/pDAB8264.44.06.1 (soybean, herbicide-tolerance, disclosed in WO2012/075426); event DAS-81419-2 (described in WO2013/016527 (e.g., commercially available as aka Conkesta™ soybean,
Conkesta™ Enlist E3™ soybean, DAS-81419-2 x DAS-44406-6); event DAS-14536-7/pDAB8291 .45.36.2 (soybean, herbicide-tolerance, disclosed in WO2012/075429); DAS-68416-4 (soybean, herbicide-tolerance, ATCC Accession No. PTA-10442, disclosed in WO2011/066384, WO2011/066360); event DP-305423-1 (soybean, quality mark, disclosed in W02008/054747, e.g. commercially available as Treus™, Plenish™, Plenish® High Oleic Soybeans); event DP-356043-5 (soybean, herbicide-tolerance, deposited as ATCC PTA- 8287, disclosed in W02008/002872, e.g. commercially available as Optimum GAT™); event FG72 (soybean, herbicide-tolerance, disclosed in WO2011/063413); event LL27 (soybean, herbicide-tolerance, disclosed in W02006/108674); event LL55 (soybean, herbicide-tolerance, disclosed in WO 2006/108675); event EE- GM3/FG72 (soybean, herbicide-tolerance) optionally stacked with event EE-GM1/LL27 or event EE-GM2/LL55 (disclosed in WO2011/063413); event MON87701 (soybean, insect control, disclosed in W02009/064652, WO2014/170327); event MON87701 x MON89788 (disclosed in WO2014/170327, e.g. commercially available as Intacta RR2 PRO® soybean); event MON87705 (soybean, improved fatty acid profile, herbicide-tolerance, disclosed in W02010/037016 or US20100080887A, e.g. commercially available as Vistive Gold™); event MON87751 (lepidopteran-resistant, ATCC accession no. PTA-120166. disclosed in WO2014/201235); event MON87751xMON87701xMON89788xMON87708 (commercially available as Intacta2 Xtend®); event MON87708 (soybean, herbicide-tolerance, disclosed in WO2011/034704, e.g. commercially available as Genuity® Roundup Ready™ 2 Xtend™); event MON87708xMON89788 (soybean, e.g. commercially available as Roundup Ready™ 2 Xtend™); event MON87712 (soybean, yield, disclosed in WO2012/051199); event MON87754 (soybean, quality feature, disclosed in WO2010/024976); event MON87769 (soybean, quality attribute, disclosed in W02009/102873); event MON89788 (soybean, herbicide-tolerance, disclosed in W02006/130436, e.g. commercially available as Genuity® Roundup Ready 2 Yield™); event SYHT0H2/SYN- 000H2-5 (soybean, herbicide-tolerance, disclosed in WO2012/082548); event DAS-21606-3 (soybean, herbicide-tolerance, disclosed in WO2012/033794); event 8264.44.06.1 (soybean, stacked herbicidetolerance, disclosed in W02022/012075426); event pDAB8291.45.36.2 (soybean, stacked herbicidetolerance, disclosed in WO2012/075429); event pDAB8264.42.32.1 (soybean, stacked herbicide-tolerance, disclosed in WO2013/010094); event A2704-12 (glufosinate tolerance, disclosed in W02006/108647); event A5547-127 (phosphinothricin tolerant, disclosed in W02006/108675); event BPS-CV127- 91 (herbicide tolerance, disclosed in WO 2010/080829); event GU262 (phosphinothricin tolerant, described in APHIS regulatory reference US 98-238-01 p); event MON 87708 x MON 89788 x A5547-127; G72xA5547-127 (event code: MST-FG072-3xACS-GM006-4, e.g. commercially available as Liberty Link™ soybean), event MON- 04032-6 (event code: GTS 40-3-2, http://www.agbios.com/static/cropdb/LONG-GTS-40-3-2-printer.html, e.g. commercially available as Roundup Ready® soybean), event HB4 (event code IND-00410-5, US2022/009011 , e.g., commercially available as Verdeca HB4 Soybean).
Particularly useful transgenic events in transgenic soybean plants, which can preferably be treated according to the invention, include event A2704-12 (glufosinate tolerance, disclosed in W02006/108647, e.g., commercially available as Liberty Link™ soybean), event A5547-127 (phosphinothricin tolerant, disclosed in W02006/108675, US8952142B2, e.g., commercially available as Liberty Link™ soybean); A5547-35 (event code: ACS-GM008-6, gene: pat, e.g. commercially available as Liberty Link™ soybean), event MON89788
(soybean, herbicide-tolerance, disclosed in W02006/130436, e.g. commercially available as Genuity® Roundup Ready 2 Yield™); DP-305423-1 (soybean, quality mark, disclosed in W02008/054747, e.g., commercially available as Treus™, Plenish™, Plenish® High Oleic Soybeans); event MON87701 (soybean, insect resistant, disclosed in W02009/064652); event MON87701 x MON89788 (disclosed in WO2014/170327, e.g. commercially available as Intacta RR2 PRO® soybean); event MON87705 (soybean, improved fatty acid profile, herbicide-tolerance, disclosed in WO2010/037016 or US20100080887A, e.g. commercially available as Vistive Gold™); event FG72 (soybean, herbicide-tolerance, disclosed in WO2011/063413); evet FG72xA5547-127 (e.g. commercially available as LibertyLink® GT27™); event SYHT0H2/SYN-000H2-5 (soybean, herbicide-tolerance, disclosed in WO2012/082548); event DAS-81419-2 (described in WO2013/016527, e.g., commercially available as Conkesta™ soybean); event DAS-44406-6 (disclosed in WO2012/075426, e.g., commercially available as Enlist E3™), or event DAS-81419-2 (described in WO2013/016527, e.g., commercially available as Conkesta™ soybean); DAS81419-2xDAS4406 (e.g., commercially available as Conkesta™ Enlist E3™ soybean); event DP305423x GTS 40-3-2 (soybean, quality mark, disclosed in W02008/054747, e.g. commercially available as Plenish® High Oleic Soybeans); event DP-356043-5 (soybean, herbicide-tolerance, deposited as ATCC PTA-8287, disclosed in W02008/002872, e.g. commercially available as Optimum GAT™), event MON-04032-6 (event code: GTS 40-3-2, http://www.agbios.com/static/cropdb/LONG-GTS-4Q-3-2-printer.html, e.g. commercially available as Roundup Ready® soybean).
Furthermore, such a list of transgenic events is provided by the United States Department of Agriculture's (USDA) Animal and Plant Health Inspection Service (APHIS) and can be found on their website on the World Wide Web at aphis.usda.gov.
Transgenic soybean events comprising herbicide tolerance genes are for example, but not excluding others, GTS 40-3-2, MON87705, MON87708, MON87712, MON87769, MON89788, A2704-12, A2704-21 , A5547- 127, A5547-35, DP356043, DAS44406-6, DAS68416-4, DAS81419-2, GU262, SYHT0H2, W62, W98, FG72 and CV127.
According to one embodiment of the invention, there is provided the use of a compound of formula (I) (l-A), or (l-B) for controlling phytopathogenic fungi in genetically modified soybean plants, wherein said transgenic soybean events comprising herbicide tolerance genes are for example, but not excluding others, GTS 40-3-2, MON87705, MON87708, MON87712, MON87769, MON89788, A2704-12, A2704-21 , A5547-127, A5547-35, DP356043, DAS44406-6, DAS68416-4, DAS81419-2, GU262, SYHT0H2, W62, W98, FG72 and CV127.
Transgenic cotton events comprising herbicide tolerance genes are for example, but not excluding others, 19- 51 a, 31707, 42317, 81910, 281 -24-236, 3006-210-23, BXN1021 1 , BXN 10215, BXN10222, BXN 10224, MON1445, MON1698, MON88701 , MON88913, GHB1 19, GHB614, LLCotton25, T303-3 and T304-40.
Transgenic cotton events comprising genes for insecticidal proteins are for example, but not excluding others, SGK321 , MON531 , MON757, MON1076, MON15985, 31707, 31803, 31807, 31808, 42317, BNLA-601 , Eventl, COT67B, COT102, T303-3, T304-40, GFM Cry1A, GK12, MLS 9124, 281 -24-236, 3006-210-23, GHB1
19 and SGK321. Increased yield has been created by increasing ear biomass using the transgene athb17, being present in corn event MON87403, or by enhancing photosynthesis using the transgene bbx32, being present in the soybean event MON87712.
Transgenic cotton events comprising glufosinate tolerance genes are for example, but not excluding others, 3006-210-23 x 281-24-236 x MON 1445 (event code: DAS-21023-5 x DAS- 24236-5 x MON-01445-2, gene: bar, e.g., commercially available as WideStrike™ Roundup Ready™ Cotton), 3006-210-23 x 281-24-236 x MON88913 (event code: DAS-21023-5 x DAS-24236-5 x MON-88913-8, gene: bar, e.g., commercially available as Widestrike™ Roundup Ready Flex™ Cotton), 3006-210-23 x 281-24-236 x MON88913 x COT102 (event code: DAS- 21023-5 x DAS-24236-5 x MON-88913-8 x SYN-IR102-7, gene: pat, e.g., commercially available as Widestrike™ x Roundup Ready Flex™ x VIPCOT™ Cotton), GHB614 x LLCotton25 (event code: BCS-GH002-5 x ACS-GH001-3, gene: bar, e.g., commercially available as GlyTol™ Liberty Link™), GHB614 x T304-40 x GHB119 (event code: BCS-GH002-5 x BCS-GH004-7 x BCS-GH005-8, gene: bar, e.g., commercially available as Glytol™ x Twinlink™), LLCotton25 (event code: ACS-GH001-3, gene: bar, e.g., commercially available as ACS- GH001-3), GHB614 x T304-40 x GHB119 x COT102 (event code: BCS- GH002-5 x BCS-GH004-7 x BCS-GH005-8 x SYN-IR102-7, gene: bar, e.g., commercially available as Glytol™ x Twinlink™ x VIPCOT™ Cotton), LLCotton25 x MON 15985 (event code: ACS-GH001-3 x MON-15985-7, gene: bar, e.g., commercially available as Fibermax™ Liberty Link™ Bollgard II ™), T304-40 x GHB1 19 (event code: BCS-GH004-7 x BCS-GH005-8, gene: bar, e.g., commercially available as TwinLink™ Cotton), GHB614 x T304-40 x GHB1 19 x COT102 (event code: BCS-GH002-5 x BCS-GH004-7 x BCS-GH005-8 x SYN-IR102- 7, gene: bar, e.g., commercially available as Glytol™ x Twinlink™ x VIPCOT™ Cotton), GHB1 19 (event code: BCS-GH005-8, gene: bar), GHB614 x LLCotton25 x MON15985 (event code: CS-GH002-5 x ACS-GH001-3 x MON-15985-7, gene: bar), MON 88701-3 (event code: MON88701 , gene: bar), T303-3 (event code: BCS- GH003-6, gene: bar), T304-40 (event code: BCS-GH003-6, gene: bar), (event code: BCS-GH004-7, gene: bar), 81910 (event code: DAS-81910-7, gene: pat), MON8870 (event code: MON 88701-3, gene: bar), MON88701 x MON88913 (event code: MON 88701-3 x MON-88913-8, gene: bar), MON88701 x MON88913 x MON15985 (event code: MON 88701-3 x MON-88913-8 x MON-15985-7, gene: bar), 281-24-236 x 3006- 210-23 x COT102 x 81910 (event code: DAS-24236-5 x DAS-21023-5 x SYN-IR102-7 x DAS-81910-7, gene: pat), COT102 x MON15985 x MON88913 x MON88701 (event code: SYN-IR102-7 x MON-15985-7 x MON- 88913-8 x MON 88701-3, gene: bar) and 3006-210-23 x 281-24-236 x MON88913 x COT102 x 81910 (event code: DAS-21023-5 x DAS-24236-5 x MON-88913-8 x SYN-IR102-7 x DAS-81910-7, gene: pat).
In a preferred aspect of the invention there is provided a method of controlling or preventing infestation of plants by the phytopathogenic microorganism Corynespora cassiicola, comprising applying to the phytopathogen, to the locus of the phytopathogen, or to a plant susceptible to attack by the phytopathogen, or to a propagation material thereof, a compound according to formula (I) (l-A), or (l-B) according to the invention, wherein the compound according to formula (I) (l-A), or (l-B) is applied in the field.
As used herein, the terms “applied in the field”, “field-applied”, “field-grown” and “field cultivation” refer broadly to the application of the pesticidal compounds or compositions directly onto plants growing in an open field or
traditional agricultural setting, as opposed to “liquid culture application” which typically involves the use of pesticidal compounds and compositions in a controlled, hydroponic, or soilless environment where plants are grown in nutrient-rich aqueous solutions, such as for the purpose of scientific research. As previously indicated, the compounds and composition as defined herein have surprisingly proven to be particularly effective for field application as compared to liquid culture applications.
In a preferred embodiment, the compound according to formula (I) (l-A), or (l-B) according to the invention is applied to a field-grown plant susceptible to attack by the phytopathogen by means of foliar application, soil application, in furrow application or drench application.
In another embodiment, the compound according to formula (I) (l-A), or (l-B) according to the invention is applied to the plant propagation material for the purpose of field cultivation of a plant susceptible to attack by the phytopathogen.
The compound of formula (I), (l-A), or (l-B) according to the invention may be used in unmodified form or, preferably, together with the adjuvants conventionally employed in the art of formulation. To this end they may be conveniently formulated in known manner to emulsifiable concentrates, coatable pastes, directly sprayable or dilutable solutions or suspensions, dilute emulsions, wettable powders, soluble powders, dusts, granulates, and also encapsulations e.g., in polymeric substances. As with the type of the compositions, the methods of application, such as spraying, atomising, dusting, scattering, coating, or pouring, are chosen in accordance with the intended objectives and the prevailing circumstances. The compositions may also contain further adjuvants such as stabilizers, antifoams, viscosity regulators, binders or tackifiers as well as fertilizers, micronutrient donors or other formulations for obtaining special effects.
Suitable carriers and adjuvants, e.g., for agricultural use, can be solid or liquid and are substances useful in formulation technology, e.g., natural or regenerated mineral substances, solvents, dispersants, wetting agents, tackifiers, thickeners, binders, or fertilizers. Such carriers are for example described in WO 1997/33890.
Suspension concentrates are aqueous formulations in which finely divided solid particles of the active compound are suspended. Such formulations include anti-settling agents and dispersing agents and may further include a wetting agent to enhance activity as well an anti-foam and a crystal growth inhibitor. In use, these concentrates are diluted in water and normally applied as a spray to the area to be treated. The amount of active ingredient may range from 0.5% to 95% of the concentrate.
Wettable powders are in the form of finely divided particles which disperse readily in water or other liquid carriers. The particles contain the active ingredient retained in a solid matrix. Typical solid matrices include fuller’s earth, kaolin clays, silicas and other readily wet organic or inorganic solids. Wettable powders normally contain from 5% to 95% of the active ingredient plus a small amount of wetting, dispersing or emulsifying agent.
Emulsifiable concentrates are homogeneous liquid compositions dispersible in water or other liquid and may consist entirely of the active compound with a liquid or solid emulsifying agent, or may also contain a liquid carrier, such as xylene, heavy aromatic naphthas, isophorone and other non-volatile organic solvents. In use,
these concentrates are dispersed in water or other liquid and normally applied as a spray to the area to be treated. The amount of active ingredient may range from 0.5% to 95% of the concentrate.
Granular formulations include both extrudates and relatively coarse particles and are usually applied without dilution to the area in which treatment is required. Typical carriers for granular formulations include sand, fuller’s earth, attapulgite clay, bentonite clays, montmorillonite clay, vermiculite, perlite, calcium carbonate, brick, pumice, pyrophyllite, kaolin, dolomite, plaster, wood flour, ground corn cobs, ground peanut hulls, sugars, sodium chloride, sodium sulphate, sodium silicate, sodium borate, magnesia, mica, iron oxide, zinc oxide, titanium oxide, antimony oxide, cryolite, gypsum, diatomaceous earth, calcium sulphate and other organic or inorganic materials which absorb or which can be coated with the active compound. Granular formulations normally contain 5% to 25% of active ingredients which may include surface-active agents such as heavy aromatic naphthas, kerosene and other petroleum fractions, or vegetable oils; and/or stickers such as dextrins, glue or synthetic resins.
Dusts are free-flowing admixtures of the active ingredient with finely divided solids such as talc, clays, flours, and other organic and inorganic solids which act as dispersants and carriers.
The active ingredients (compounds of formula (l-A) or (l-B) and mixtures thereof with component (B) can also be contained in microcapsules. Microcapsules contain the active ingredients in a porous carrier. This enables the active ingredients to be released into the environment in controlled amounts (e.g., slow-release). Microcapsules usually have a diameter of from 0.1 to 500 microns. They contain active ingredients in an amount of about from 25 to 95 % by weight of the capsule weight. The active ingredients can be in the form of a monolithic solid, in the form of fine particles in solid or liquid dispersion or in the form of a suitable solution. The encapsulating membranes can comprise, for example, natural or synthetic rubbers, cellulose, styrene/butadiene copolymers, polyacrylonitrile, polyacrylate, polyesters, polyamides, polyureas, polyurethane, or chemically modified polymers, and starch xanthates, or other polymers that are known to the person skilled in the art. Alternatively, very fine microcapsules can be formed in which the active ingredient is contained in the form of finely divided particles in a solid matrix of base substance, but the microcapsules are not themselves encapsulated.
Microcapsules are typically droplets or granules of the active ingredient enclosed in an inert porous shell which allows escape of the enclosed material to the surroundings at controlled rates. Encapsulated droplets are typically 1 to 50 microns in diameter. The enclosed liquid typically constitutes 50 to 95% of the weight of the capsule and may include solvent in addition to the active compound. Encapsulated granules are generally porous granules with porous membranes sealing the granule pore openings, retaining the active species in liquid form inside the granule pores. Granules typically range from 1 millimetre to 1 centimetre and preferably 1 to 2 millimetres in diameter. Granules are formed by extrusion, agglomeration or prilling, or are naturally occurring. Examples of such materials are vermiculite, sintered clay, kaolin, attapulgite clay, sawdust, and granular carbon. Shell or membrane materials include natural and synthetic rubbers, cellulosic materials, styrene-butadiene copolymers, polyacrylonitriles, polyacrylates, polyesters, polyamides, polyureas, polyurethanes and starch xanthates.
Other useful formulations for agrochemical applications include simple solutions of the active ingredient in a solvent in which it is completely soluble at the desired concentration, such as acetone, alkylated naphthalenes, xylene and other organic solvents. Pressurised sprayers, wherein the active ingredient is dispersed in finely divided form because of vaporisation of a low boiling dispersant solvent carrier, may also be used.
Suitable agricultural adjuvants and carriers that are useful in formulating the compositions of the invention in the formulation types described above are well known to a person skilled in the art.
Liquid carriers that can be employed include, for example, water, toluene, xylene, petroleum naphtha, crop oil, acetone, methyl ethyl ketone, cyclohexanone, acetic anhydride, acetonitrile, acetophenone, amyl acetate, 2- butanone, chlorobenzene, cyclohexane, cyclohexanol, alkyl acetates, diacetonalcohol, 1 ,2-dichloropropane, diethanolamine, p diethylbenzene, diethylene glycol, diethylene glycol abietate, diethylene glycol butyl ether, diethylene glycol ethyl ether, diethylene glycol methyl ether, N,N-dimethyl formamide, dimethyl sulfoxide, 1 ,4- dioxane, dipropylene glycol, dipropylene glycol methyl ether, dipropylene glycol dibenzoate, diproxitol, alkyl pyrrolidinone, ethyl acetate, 2-ethyl hexanol, ethylene carbonate, 1 ,1 ,1 -trichloroethane, 2-heptanone, alpha pinene, d-limonene, ethylene glycol, ethylene glycol butyl ether, ethylene glycol methyl ether, gammabutyrolactone, glycerol, glycerol diacetate, glycerol monoacetate, glycerol triacetate, hexadecane, hexylene glycol, isoamyl acetate, isobornyl acetate, isooctane, isophorone, isopropyl benzene, isopropyl myristate, lactic acid, laurylamine, mesityl oxide, methoxy-propanol, methyl isoamyl ketone, methyl isobutyl ketone, methyl laurate, methyl octanoate, methyl oleate, methylene chloride, m-xylene, n-hexane, n-octylamine, octadecanoic acid, octyl amine acetate, oleic acid, oleylamine, o-xylene, phenol, polyethylene glycol (PEG400), propionic acid, propylene glycol, propylene glycol monomethyl ether, p-xylene, toluene, triethyl phosphate, triethylene glycol, xylene sulfonic acid, paraffin, mineral oil, trichloroethylene, perchloroethylene, ethyl acetate, amyl acetate, butyl acetate, methanol, ethanol, isopropanol, and higher molecular weight alcohols such as amyl alcohol, tetrahydrofurfuryl alcohol, hexanol, octanol, etc., ethylene glycol, propylene glycol, glycerine and N- methyl-2-pyrrolidinone. Water is generally the carrier of choice for the dilution of concentrates.
Suitable solid carriers include, for example, talc, titanium dioxide, pyrophyllite clay, silica, attapulgite clay, kieselguhr, chalk, diatomaxeous earth, lime, calcium carbonate, bentonite clay, fuller’s earth, cotton seed hulls, wheat flour, soybean flour, pumice, wood flour, walnut shell flour and lignin.
A broad range of surface-active agents are advantageously employed in both said liquid and solid compositions, especially those designed to be diluted with carrier before application. These agents, when used, normally comprise from 0.1 % to 15% by weight of the formulation. They can be anionic, cationic, nonionic or polymeric in character and can be employed as emulsifying agents, wetting agents, suspending agents or for other purposes. Typical surface-active agents include salts of alkyl sulfates, such as diethanolammonium lauryl sulphate; alkylarylsulfonate salts, such as calcium dodecylbenzenesulfonate; alkylphenol-alkylene oxide addition products, such as nonylphenol-C.sub. 18 ethoxylate; alcohol-alkylene oxide addition products, such as tridecyl alcohol-C.sub. 16 ethoxylate; soaps, such as sodium stearate; alkylnaphthalenesulfonate salts, such as sodium dibutylnaphthalenesulfonate; dialkyl esters of sulfosuccinate salts, such as sodium di(2- ethylhexyl) sulfosuccinate; sorbitol esters, such as sorbitol oleate; quaternary amines, such as lauryl
trimethylammonium chloride; polyethylene glycol esters of fatty acids, such as polyethylene glycol stearate; block copolymers of ethylene oxide and propylene oxide; and salts of mono and dialkyl phosphate esters.
Other adjuvants commonly utilized in agricultural compositions include crystallisation inhibitors, viscosity modifiers, suspending agents, spray droplet modifiers, pigments, antioxidants, foaming agents, anti-foaming agents, light-blocking agents, compatibilizing agents, antifoam agents, sequestering agents, neutralising agents and buffers, corrosion inhibitors, dyes, odorants, spreading agents, penetration aids, micronutrients, emollients, lubricants and sticking agents.
Thus, in an embodiment the compounds of formula (I), (l-A), or (l-B) according to the invention may be used in the form of a pesticidal composition further comprising one or more adjuvants selected from stabilizers, viscosity regulators, binders, tackifiers, fertilizers, crystallisation inhibitors, viscosity modifiers, suspending agents, spray droplet modifiers, pigments, antioxidants, foaming agents, anti-foaming agents, light-blocking agents, compatibilising agents, antifoam agents, sequestering agents, neutralising agents and buffers, corrosion inhibitors, dyes, odorants, spreading agents, penetration aids, micronutrients, emollients, lubricants, and sticking agents.
The pesticidal composition may also comprise one or more agents that act as a carrier or as a diluent of the active ingredients, or both. The agent acting as a carrier or diluent, or both, hereinafter called “carrier or diluent”, may be liquid or solid.
In an embodiment the compounds of formula (I), (l-A), or (l-B) according to the invention may be used in the form of a pesticidal composition further comprising one or more liquid carriers or diluents selected from xylene, heavy aromatic naphthas, isophorone, water, toluene, petroleum naphtha, crop oil, acetone, methyl ethyl ketone, cyclohexanone, acetic anhydride, acetonitrile, acetophenone, amyl acetate, 2-butanone, chlorobenzene, cyclohexane, cyclohexanol, alkyl acetates, diacetonalcohol, 1 ,2-dichloropropane, diethanolamine, p diethylbenzene, diethylene glycol, diethylene glycol abietate, diethylene glycol butyl ether, diethylene glycol ethyl ether, diethylene glycol methyl ether, n,n-dimethyl formamide, dimethyl sulfoxide, 1 ,4- dioxane, dipropylene glycol, dipropylene glycol methyl ether, dipropylene glycol dibenzoate, diproxitol, alkyl pyrrolidinone, ethyl acetate, 2-ethyl hexanol, ethylene carbonate, 1 ,1 ,1 -trichloroethane, 2-heptanone, alpha pinene, d-limonene, ethylene glycol, ethylene glycol butyl ether, ethylene glycol methyl ether, gammabutyrolactone, glycerol, glycerol diacetate, glycerol monoacetate, glycerol triacetate, hexadecane, hexylene glycol, isoamyl acetate, isobornyl acetate, isooctane, isopropyl benzene, isopropyl myristate, lactic acid, laurylamine, mesityl oxide, methoxy-propanol, methyl isoamyl ketone, methyl isobutyl ketone, methyl laurate, methyl octanoate, methyl oleate, methylene chloride, m-xylene, n-hexane, n-octylamine, octadecanoic acid, octyl amine acetate, oleic acid, oleylamine, o-xylene, phenol, polyethylene glycol (PEG400), propionic acid, propylene glycol, propylene glycol monomethyl ether, p-xylene, triethyl phosphate, triethylene glycol, xylene sulfonic acid, paraffin, mineral oil, trichloroethylene, perchloroethylene, ethyl acetate, amyl acetate, butyl acetate, methanol, ethanol, isopropanol, higher molecular weight alcohols (amyl alcohol, tetrahydrofurfuryl alcohol, hexanol, octanol, etc.), and water (for dilution of concentrates).
In an embodiment the compounds of formula (I), (l-A), or (l-B) according to the invention may be used in the form of a pesticidal composition further comprising one or more solid diluents or carriers selected from talc, titanium dioxide, pyrophyllite clay, silica, attapulgite clay, kieselguhr, chalk, diatomaceous earth, lime, calcium carbonate, bentonite clay, Fuller's earth, cotton seed hulls, wheat flour, soybean flour, pumice, wood flour, walnut shell flour, and lignin.
In an embodiment the compounds of formula (I), (l-A), or (l-B) according to the invention may be used in the form of a pesticidal composition further comprising one or more formulation auxiliaries selected from unepoxidized or epoxidized vegetable oils, preservatives, inorganic compounds.
In addition, further, other biocidal active ingredients or compositions may be combined with the compounds or compositions of the invention and used in the methods of the invention and applied simultaneously or sequentially with the compounds or compositions of the invention. When applied simultaneously, these further active ingredients may be formulated together with the compounds or compositions of the invention or mixed in, for example, the spray tank. These further biocidal active ingredients may be fungicides, herbicides, insecticides, bactericides, acaricides, nematicides and/or plant growth regulators.
Pesticidal agents are referred to herein using their common name are known, for example, from "The Pesticide Manual", 15th Ed., British Crop Protection Council 2009.
In addition, the compositions of the invention may also be applied with one or more systemically acquired resistance inducers (“SAR” inducer). SAR inducers are known and described in, for example, United States Patent No. US 6,919,298 and include, for example, salicylates and the commercial SAR inducer acibenzolar- S-methyl.
The compounds of formula (I), (l-A), or (l-B) according to the invention are normally used in the form of agrochemical compositions and can be applied to the crop area or plant to be treated, simultaneously or in succession with further compounds. These further compounds can be e.g., fertilizers or micronutrient donors or other preparations, which influence the growth of plants. They can also be selective herbicides or non- selective herbicides as well as insecticides, fungicides, bactericides, nematicides, molluscicides or mixtures of several of these preparations, if desired together with further carriers, surfactants or application promoting adjuvants customarily employed in the art of formulation.
The compounds of formula (I), (l-A), or (l-B) according to the invention may be used in the form of (fungicidal) compositions for controlling or protecting against phytopathogenic microorganisms, comprising as active ingredient a compound as defined herein, in free form or in agrochemical usable salt form, and at least one of the above-mentioned adjuvants, and preferably at least one formulation auxiliary, diluent and/or carrier.
The invention therefore provides a composition, preferably a fungicidal composition, comprising a compound of formula (I), (l-A), or (l-B) according to the invention, an agriculturally acceptable carrier and optionally an adjuvant or an adjuvant and one or more auxiliaries, diluents and/or carriers. An agricultural acceptable carrier is for example a carrier that is suitable for agricultural use. Agricultural carriers are well known in the art.
Preferably, said composition may comprise at least one or more pesticidal-active compounds, for example an additional fungicidal active ingredient in addition to a compound of formula (I), (l-A), or (l-B).
The compound of formula (I), (l-A), or (l-B) according to the invention may be the sole active ingredient of a composition or it may be admixed with one or more additional active ingredients such as a pesticide, including fungicides, herbicide, insecticide, synergist, or plant growth regulator where appropriate. An additional active ingredient may, in some cases, result in unexpected synergistic activities.
Examples of suitable additional active ingredients include the following: acycloamino acid fungicides, aliphatic nitrogen fungicides, amide fungicides, anilide fungicides, antibiotic fungicides, aromatic fungicides, arsenical fungicides, aryl phenyl ketone fungicides, benzamide fungicides, benzanilide fungicides, benzimidazole fungicides, benzothiazole fungicides, botanical fungicides, bridged diphenyl fungicides, carbamate fungicides, carbanilate fungicides, conazole fungicides, copper fungicides, dicarboximide fungicides, dinitrophenol fungicides, dithiocarbamate fungicides, dithiolane fungicides, furamide fungicides, furanilide fungicides, hydrazide fungicides, imidazole fungicides, mercury fungicides, morpholine fungicides, organophosphorous fungicides, organotin fungicides, oxathiin fungicides, oxazole fungicides, phenylsulfamide fungicides, polysulfide fungicides, pyrazole fungicides, pyridine fungicides, pyrimidine fungicides, pyrrole fungicides, quaternary ammonium fungicides, quinoline fungicides, quinone fungicides, quinoxaline fungicides, strobilurin fungicides, sulfonanilide fungicides, thiadiazole fungicides, thiazole fungicides, thiazolidine fungicides, thiocarbamate fungicides, thiophene fungicides, triazine fungicides, triazole fungicides, triazolopyrimidine fungicides, urea fungicides, valinamide fungicides, and zinc fungicides.
A further aspect of the present invention is a method of controlling diseases on useful plants or on propagation material thereof caused by phytopathogens, which comprises applying to the useful plants, the locus thereof or propagation material thereof a composition according to the invention. Preferred is a method, which comprises applying to the useful plants or to the locus thereof a composition according to the invention, more preferably to the useful plants. Further preferred is a method, which comprises applying to the propagation material of the useful plants a composition according to the invention.
Throughout this document the expression “composition” refers to the various mixtures or combinations of components (A) and (B), for example in a single “ready-mix” form, in a combined spray mixture composed from separate formulations of the single active ingredient components, such as a “tank-mix”, and in a combined use of the single active ingredients when applied in a sequential manner, i.e. one after the other with a reasonably short period, such as a few hours or days. The order of applying the components (A) and (B) is not essential for working the present invention.
The compositions according to the invention are effective against harmful microorganisms, such as microorganisms, that cause phytopathogenic diseases, in particular against phytopathogenic fungi and bacteria.
Preferably, the formulation comprising a composition according to the invention may comprise of from 0.01 to 90% by weight of the composition comprising components (A) and (B), and of from 0 to 20% of an agriculturally
acceptable surfactant. Preferably, the formulation further comprises other active agents, in particular microbiocides and pesticides, more generally.
Advantageously, the formulation further comprises of from 10 to 99.99% solid or liquid formulation inerts, conservatives and/or adjuvants.
The present invention preferably also relates to a concentrated composition for dilution by the user, comprising a composition according or a formulation according to the invention, comprising of from 2 to 80% by weight, preferably between 5 and 70% by weight, of active agents comprising at least a composition comprising (A) and (B), and optionally, other active agents.
The present invention preferably also relates to a seed dressing formulation for application to plant propagation materials, comprising a composition according to the invention, and further comprising a diluent. Preferably, the seed dressing formulation is in the form of an aqueous suspension or in a dry powder form having good adherence to the plant propagation materials. Preferably, the seed dressing formulation may comprise the active agents in an encapsulated form, preferably a slow-release capsules and/or microcapsules.
The present invention preferably also relates to a method of combating and controlling phytopathogens, comprising applying a fungicidally effective amount of a composition according to the invention to a pest, a locus of pest, or to a plant susceptible to attack by a pest, with the exception of a method for treatment of the human or animal body by surgery or therapy and diagnostic methods practiced on the human or animal body.
The methods, compositions and uses according to any one of embodiments according to the invention are preferably for controlling or preventing infestation of the crop by the phytopathogenic microorganisms Corynespora cassiicola, that are resistant to other fungicides. Corynespora cassiicola that are "resistant" to a particular fungicide refer e.g., to strains of Corynespora cassiicola fungi that are less sensitive to that fungicide compared to the expected sensitivity of the same species of Corynespora cassiicola fungi. The expected sensitivity can be measured using e.g., a strain that has not previously been exposed to the fungicide. An “effective” amount herein refers to an amount of the active ingredient that shows sufficient biocidal activity, e.g., at least 10 %, more preferably at least 20%, yet more preferably at least 50%, and again more preferably at least 70 % effectiveness, compared to the blind test. In the present case, the composition according to the invention preferably comprise at least 0.01 ppm, more preferably at last 0.025 ppm of active ingredient, more preferably at least for example 6 ppm, 3 ppm, 2.2 ppm, 1 .5 ppm, 0.8 ppm, 0.74 ppm, 0.25 ppm, 0.2 ppm, or 0.082 ppm as applied.
Application according to the methods or uses according to any one of embodiments according to the invention is preferably to a crop of plants, the locus thereof or propagation material thereof. Preferably application is to the phytopathogen, to the locus ofthe phytopathogen, orto a plant susceptible to attack by the phytopathogen, or to a propagation material thereof. Application of the compounds as defined in any one of embodiments 1 to 13 can be performed according to any of the usual modes of application, e.g., foliar, drench, soil, in furrow etc.
In preferred embodiments ofthe present invention, the compounds or pesticidal compositions as defined herein are applied in the field to the foliage of the plant (foliar application), thus allowing to select frequency and rate
of application to match the danger of infestation with the pest in question. Alternatively, the active ingredient can reach the plants via the root system (systemic action), by drenching the locus of the plants in the field with a liquid composition or by incorporating the active ingredient in solid form into the locus of the plants in the field, for example into the soil or furrow, such as in the form of granules. In the case of paddy rice crops, such granules can be metered into the flooded paddy-field.
The compounds as defined in any one of embodiments according to the invention are suitable for use on any plant, including those that have been genetically modified to be resistant to active ingredients such as herbicides, or to produce biologically active compounds that control infestation by plant pests.
Generally, a compound as defined in any one of embodiments according to the invention is used in the form of a composition (e.g., formulation) containing a carrier. A compound as defined in any one of embodiments according to the invention and compositions thereof can be used in various forms such as aerosol dispenser, capsule suspension, cold fogging concentrate, dustable powder, emulsifiable concentrate, emulsion oil in water, emulsion water in oil, encapsulated granule, fine granule, flowable concentrate for seed treatment, gas (under pressure), gas generating product, granule, hot fogging concentrate, macrogranule, microgranule, oil dispersible powder, oil miscible flowable concentrate, oil miscible liquid, paste, plant rodlet, powder for dry seed treatment, seed coated with a pesticide, soluble concentrate, soluble powder, solution for seed treatment, suspension concentrate (flowable concentrate), ultra-low volume (ulv) liquid, ultra-low volume (ulv) suspension, water dispersible granules or tablets, water dispersible powder for slurry treatment, water soluble granules or tablets, water soluble powder for seed treatment and wettable powder.
Compositions of this invention, including all of the above disclosed embodiments and preferred examples thereof, can be mixed with one or more further pesticides including further fungicides, insecticides, nematicides, bactericides, acaricides, growth regulators, chemosterilants, semiochemicals, repellents, attractants, pheromones, feeding stimulants or other biologically active compounds to form a multi-component pesticide giving an even broader spectrum of agricultural protection.
A formulation typically comprises a liquid or solid carrier and optionally one or more customary formulation auxiliaries, which may be solid or liquid auxiliaries, for example unepoxidized or epoxidized vegetable oils (for example epoxidized coconut oil, rapeseed oil or soya oil), antifoams, for example silicone oil, preservatives, clays, inorganic compounds, viscosity regulators, surfactant, binders and/or tackifiers. The composition may also further comprise a fertilizer, a micronutrient donor or other preparations which influence the growth of plants as well as comprising a combination containing the compound of the invention with one or more other biologically active agents, such as bactericides, fungicides, nematicides, plant activators, acaricides, and insecticides.
The compositions are prepared in a manner known perse, in the absence of auxiliaries for example by grinding, screening and/or compressing a solid compound of the present invention and in the presence of at least one auxiliary for example by intimately mixing and/or grinding the compound of the present invention with the
auxiliary (auxiliaries). In the case of solid compounds of the invention, the grinding/milling of the compounds is to ensure specific particle size.
Examples of compositions for use in agriculture are emulsifiable concentrates, suspension concentrates, microemulsions, oil dispersibles, directly sprayable or dilutable solutions, spreadable pastes, dilute emulsions, soluble powders, dispersible powders, wettable powders, dusts, granules or encapsulations in polymeric substances, which comprise - at least - a compound of formula (I) as defined herein and the type of composition is to be selected to suit the intended aims and the prevailing circumstances.
As a rule, the compositions comprise 0.1 to 99 %, especially 0.1 to 95 %, of a compound (A), and optionally compound (B), as defined in any one of embodiments according to the invention and 1 to 99.9 %, especially 5 to 99.9 %, of at least one solid or liquid carrier, it being possible as a rule for 0 to 25 %, especially 0.1 to 20%, of the composition to be surfactants (% in each case meaning percent by weight). Whereas concentrated compositions tend to be preferred for commercial goods, the end consumer as a rule uses dilute compositions which have substantially lower concentrations of active ingredient.
With respect to compositions comprising component (A) and component (B); in general, the weight ratio of component (A) to component (B) is from 2000 : 1 to 1 : 1000. The weight ratio of component (A) to component (B) is preferably from 100 : 1 to 1 : 100; more preferably from 20 : 1 to 1 : 50, yet more preferably from 12 : 1 to 1 : 25; yet more preferably from 10 : 1 to 1 : 10, again more preferably from 5 : 1 to 1 : 15; and most preferably from 2 :1 to 1 : 5.
Besides a potential synergistic action with respect to fungicidal activity, the compositions according to the invention can also have further surprising advantageous properties. Examples of such advantageous properties that may be mentioned are: more advantageous degradability; improved toxicological and/or ecotoxicological behaviour; or improved characteristics of the useful plants including: emergence, crop yields, more developed root system, tillering increase, increase in plant height, bigger leaf blade, less dead basal leaves, stronger tillers, greener leaf colour, less fertilizers needed, less seeds needed, more productive tillers, earlier flowering, early grain maturity, less plant verse (lodging), increased shoot growth, improved plant vigour, and early germination.
Additional beneficial effects can be the suppression or reduction of development of resistance against a certain active ingredient, by combinations that may have one or more different modes of action may in particular be beneficial.
Some compositions according to the invention have a systemic action and can be used as foliar, soil and seed treatment fungicides.
With the compositions according to the invention it is possible to inhibit or destroy the phytopathogenic microorganisms which occur in plants or in parts of plants (fruit, blossoms, leaves, stems, tubers, roots) in different useful plants, while at the same time the parts of plants which grow later are also protected from attack by phytopathogenic microorganisms.
The compositions according to the invention can be applied to the phytopathogenic microorganisms, the useful plants, the locus thereof, the propagation material thereof, storage goods or technical materials threatened by microorganism attack.
The compositions according to the invention may be applied before or after infection of the useful plants, the propagation material thereof, storage goods or technical materials by the microorganisms.
The amount of a composition according to the invention to be applied, will depend on various factors, such as the compounds employed; the subject of the treatment, such as, for example plants, soil or seeds; the type of treatment, such as, for example spraying, dusting or seed dressing; the purpose of the treatment, such as, for example prophylactic or therapeutic; the type of fungi to be controlled or the application time.
When applied to the useful plants component (A) is typically applied at a rate of 5 to 2000 g a.i./ha, particularly 10 to 1000 g a.i./ha, e.g., 50, 75, 100 or 200 g a.i./ha, preferably in association with 1 to 5000 g a.i./ha, particularly 2 to 2000 g a.i./ha, e.g., 100, 250, 500, 800, 1000, 1500 g a.i./ha of component (B).
The compounds as defined in any one of embodiments according to the invention are preferably used for pest control at rates of 1 to 500 g/ha, preferably 50-300 g/ha.
Preferably the compounds of formula (I) as defined in any one of embodiments according to the invention are used for pest control at rates of 1 to 500 g/ha, preferably 10 to 400 g/ha, more preferably 15 to 300 g/ha, most preferably 10 to 100 g/ha, such as 10, 15, 20, 25, 30, 35, 40, 45, 50, 55, 60, or 65 g a.i /ha.
In certain embodiments of the present invention, the compounds of formula (I) and pesticidal compositions comprising these compounds as defined in any one of embodiments according to the invention are applied at rates of 10 to 100 g a.i./ha, preferably 15 to 65 g a.i./ha, more preferably at rates of 20 to 60 g a.i./ha, such as 10, 15, 20, 30, 35, 40, or 50 g a.i /ha.
In an embodiment of the method according to the invention, the compound according to formula (I) is applied at a rate of at least 10 g/ha, preferably at least 10, 15, 20, 25, 30, 35, 40, 45, 50, 55, 60 g/ha.
In an embodiment of the method according to the invention, the compound according to formula (I) is applied at a rate of at most 100 g/ha, preferably at most 100, 95, 90, 85, 80, 75, 70, 65 g/ha.
The term “g a.i./ha” as used herein refer to the application rate given in gram [g] of active ingredient [a.i.] per unit of surface [ha]. The unit hectare (symbol ha) is the metric unit of area that equals a square with 100 m side (1 hm2) or 10,000 square meters. Hectare is a commonly used unit of area in the metric system.
In agricultural practice the application rates of the compositions according to the invention depend on the type of effect desired, and typically range from 20 to 4000 g of total composition per hectare.
When the compositions according to the invention are used for treating seed, rates of 0.001 to 50 g of a compound of component (A) per kg of seed, preferably from 0.01 to 10g per kg of seed, and preferably 0.001 to 50 g of a compound of component (B), per kg of seed, preferably from 0.01 to 10g per kg of seed, are generally sufficient.
Examples of foliar formulation types for pre-mix compositions are:
GR: Granules
WP: wettable powders
WG: water dispersible granules (powders)
SG: water soluble granules
SL: soluble concentrates
EC: emulsifiable concentrate
DC: dispersible concentrate
EW: emulsions, oil in water
ME: micro-emulsion
SC: aqueous suspension concentrate
CS: aqueous capsule suspension
CD: oil-based suspension concentrate, and
SE: aqueous suspo-emulsion.
Whereas examples of seed treatment formulation types for pre-mix compositions are:
WS: wettable powders for seed treatment slurry
LS: solution for seed treatment
ES: emulsions for seed treatment
FS: suspension concentrate for seed treatment
WG: water dispersible granules, and
CS: aqueous capsule suspension.
Examples of formulation types suitable fortank-mix compositions are solutions, dilute emulsions, suspensions, or a mixture thereof, and dusts.
As with the nature of the formulations, the methods of application, such as foliar, drench, spraying, atomizing, dusting, scattering, coating or pouring, are chosen in accordance with the intended objectives and the prevailing circumstances.
The tank-mix compositions are generally prepared by diluting with a solvent (for example, water) the one or more pre-mix compositions containing different pesticides, and optionally further auxiliaries.
Suitable carriers and adjuvants can be solid or liquid and are the substances ordinarily employed in formulation technology, e.g., natural or regenerated mineral substances, solvents, dispersants, wetting agents, tackifiers, thickeners, binders, or fertilizers.
Generally, a tank-mix formulation for foliar or soil application comprises 0.1 to 20%, especially 0.1 to 15 %, of the desired ingredients, and 99.9 to 80 %, especially 99.9 to 85 %, of a solid or liquid auxiliaries (including, for example, a diluent or solvent such as water), where the auxiliaries can be a surfactant in an amount of 0 to 20 %, especially 0.1 to 15 %, based on the tank-mix formulation.
Typically, a pre-mix formulation for foliar application comprises 0.1 to 99.9 %, especially 1 to 95 %, of the desired ingredients, and 99.9 to 0.1 %, especially 99 to 5 %, of a solid or liquid adjuvant (including, for example, a solvent such as water), where the auxiliaries can be a surfactant in an amount of 0 to 50 %, especially 0.5 to 40 %, based on the pre-mix formulation.
Normally, a tank-mix formulation for seed treatment application comprises 0.25 to 80%, especially 1 to 75 %, of the desired ingredients, and 99.75 to 20 %, especially 99 to 25 %, of a solid or liquid auxiliaries (including, for example, a solvent such as water), where the auxiliaries can be a surfactant in an amount of 0 to 40 %, especially 0.5 to 30 %, based on the tank-mix formulation.
Typically, a pre-mix formulation for seed treatment application comprises 0.5 to 99.9 %, especially 1 to 95 %, of the desired ingredients, and 99.5 to 0.1 %, especially 99 to 5 %, of a solid or liquid adjuvant (including, for example, a solvent such as water), where the auxiliaries can be a surfactant in an amount of 0 to 50 %, especially 0.5 to 40 %, based on the pre-mix formulation.
Whereas commercial products will preferably be formulated as concentrates (e.g., pre-mix composition (formulation)), the end user will normally employ dilute formulations (e.g., tank mix composition).
Preferred seed treatment pre-mix formulations are aqueous suspension concentrates. The formulation can be applied to the seeds using conventional treating techniques and machines, such as fluidized bed techniques, the roller mill method, rotostatic seed treaters, and drum coaters. Other methods, such as spouted beds may also be useful. The seeds may be presized before coating. After coating, the seeds are typically dried and then transferred to a sizing machine for sizing. Such procedures are known in the art. The compounds of the present invention are particularly suited for use in soil and seed treatment applications. In general, the pre-mix compositions of the invention contain 0.5 to 99.9 especially 1 to 95, advantageously 1 to 50 % by mass of the desired ingredients, and 99.5 to 0.1 , especially 99 to 5 % by mass of a solid or liquid adjuvant (including, for example, a solvent such as water), where the auxiliaries (or adjuvant) can be a surfactant in an amount of 0 to 50, especially 0.5 to 40 % by mass based on the mass of the pre-mix formulation.
In addition, further, other biocidally active ingredients or compositions may be combined with the compositions of the invention and used in the methods of the invention and applied simultaneously or sequentially with the compositions of the invention. When applied simultaneously, these further active ingredients may be formulated together with the compositions of the invention or mixed in, for example, the spray tank. These further biocidally active ingredients may be fungicides, herbicides, insecticides, bactericides, acaricides, nematicides and/or plant growth regulators.
In addition, the compositions of the invention may also be applied with one or more systemically acquired resistance inducers (“SAR” inducer). SAR inducers are known and described in, for example, United States Patent No. US6, 919,298 and include, for example, salicylates and the commercial SAR inducer acibenzolar- S-methyl.
The compounds as defined in any one of embodiments according to the invention are normally used in the form of compositions and can be applied to the crop area or plant to be treated, simultaneously or in succession
with further compounds. These further compounds can be e.g., fertilizers or micronutrient donors or other preparations, which influence the growth of plants. They can also be selective herbicides or non-selective herbicides as well as insecticides, fungicides, bactericides, nematicides, molluscicides or mixtures of several of these preparations, if desired together with further carriers, surfactants or application promoting adjuvants customarily employed in the art of formulation.
The compounds of formula (I) may be used in the form of (fungicidal) compositions for controlling or protecting against the phytopathogen(s) Corynespora cassiicola, comprising as active ingredient at least one compound as defined in any one of embodiments according to the invention, in free form or in agrochemically usable salt form, and at least one of the above-mentioned adjuvants.
The plants and / or target crops in accordance with the invention include conventional as well as genetically enhanced or engineered varieties such as, for example, insect resistant (e.g., Bt. and VIP varieties) as well as disease resistant, herbicide tolerant (e.g., glyphosate- and glufosinate-resistant maize varieties commercially available under the trade names RoundupReady® and Li berty Lin k®) and nematode tolerant varieties. By way of example, suitable genetically enhanced or engineered crop varieties include the Stoneville 5599BR cotton and Stoneville 4892BR cotton varieties.
The term "plants" and/or “target crops” is to be understood as including also plants that have been rendered tolerant to herbicides like bromoxynil or classes of herbicides (such as, for example, HPPD inhibitors, ALS inhibitors, for example primisulfuron, prosulfuron and trifloxysulfuron, EPSPS (5-enol-pyrovyl-shikimate-3- phosphate-synthase) inhibitors, GS (glutamine synthetase) inhibitors or PPO (protoporphyrinogen-oxidase) inhibitors) as a result of conventional methods of breeding or genetic engineering. An example of a crop that has been rendered tolerant to imidazolinones, e.g., imazamox, by conventional methods of breeding (mutagenesis) is Clearfield® summer rape (Canola). Examples of crops that have been rendered tolerant to herbicides or classes of herbicides by genetic engineering methods include glyphosate- and glufosinateresistant maize varieties commercially available under the trade names RoundupReady®, Herculex I® and Li berty Link®.
The term "plants" and/or “target crops” is to be understood as including those which naturally are or have been rendered resistant to harmful insects. This includes plants transformed by the use of recombinant DNA techniques, for example, to be capable of synthesizing one or more selectively acting toxins, such as are known, for example, from toxin-producing bacteria. Examples of toxins which can be expressed include 8- endotoxins, vegetative insecticidal proteins (Vip), insecticidal proteins of bacteria colonizing nematodes, and toxins produced by scorpions, arachnids, wasps, and fungi. An example of a crop that has been modified to express the Bacillus thuringiensis toxin is the Bt maize KnockOut® (Syngenta Seeds). An example of a crop comprising more than one gene that codes for insecticidal resistance and thus expresses more than one toxin is VipCot® (Syngenta Seeds). Crops or seed material thereof can also be resistant to multiple types of pests (so-called stacked transgenic events when created by genetic modification). For example, a plant can have
the ability to express an insecticidal protein while at the same time being herbicide tolerant, for example Herculex I® (Dow AgroSciences, Pioneer Hi-Bred International).
The term "plants" and/or “target crops” is to be understood as also including plants which have been so transformed by the use of recombinant DNA techniques that they are capable of synthesising antipathogenic substances having a selective action, such as, for example, the so-called "pathogenesis-related proteins" (PRPs, see e.g., EP0392225A). Examples of such antipathogenic substances and transgenic plants capable of synthesising such antipathogenic substances are known, for example, from EP0392225A, WO95/33818, and EP0353191A. The methods of producing such transgenic plants are generally known to the person skilled in the art and are described, for example, in the publications mentioned above.
Toxins that can be expressed by transgenic plants include, for example, insecticidal proteins from Bacillus cereus or Bacillus popilliae; or insecticidal proteins from Bacillus thuringiensis, such as 8-endotoxins, e.g. CrylAb, CrylAc, Cry1 F, Cry1 Fa2, Cry2Ab, Cry3A, Cry3Bb1 or Cry9C, or vegetative insecticidal proteins (Vip), e.g. Vip1 , Vip2, Vip3 or Vip3A; or insecticidal proteins of bacteria colonising nematodes, for example Photorhabdus spp. or Xenorhabdus spp., such as Photorhabdus luminescens, Xenorhabdus nematophilus; toxins produced by animals, such as scorpion toxins, arachnid toxins, wasp toxins and other insect-specific neurotoxins; toxins produced by fungi, such as Streptomycetes toxins, plant lectins, such as pea lectins, barley lectins or snowdrop lectins; agglutinins; proteinase inhibitors, such as trypsin inhibitors, serine protease inhibitors, patatin, cystatin, papain inhibitors; ribosome-inactivating proteins (RIP), such as ricin, maize-RIP, abrin, luffin, saporin or bryodin; steroid metabolism enzymes, such as 3-hydroxysteroidoxidase, ecdysteroid- UDP-glycosyl-transferase, cholesterol oxidases, ecdysone inhibitors, HMG-COA-reductase, ion channel blockers, such as blockers of sodium or calcium channels, juvenile hormone esterase, diuretic hormone receptors, stilbene synthase, bibenzyl synthase, chitinases and glucanases.
Further, in the context of the present invention there are to be understood by 8-endotoxins, for example CrylAb, CrylAc, Cry1 F, Cry1 Fa2, Cry2Ab, Cry3A, Cry3Bb1 or Cry9C, or vegetative insecticidal proteins (Vip), for example Vip1 , Vip2, Vip3 or Vip3A, expressly also hybrid toxins, truncated toxins and modified toxins. Hybrid toxins are produced recombinantly by a new combination of different domains of those proteins (see, for example, WO 02/15701). Truncated toxins, for example a truncated CrylAb, are known. In the case of modified toxins, one or more amino acids of the naturally occurring toxin are replaced. In such amino acid replacements, preferably non-naturally present protease recognition sequences are inserted into the toxin, such as, for example, in the case of Cry3A055, a cathepsin-G-recognition sequence is inserted into a Cry3A toxin (see W003/018810).
More examples of such toxins or transgenic plants capable of synthesising such toxins are disclosed, for example, in EP-A-0 374 753, WO93/07278, WO95/34656, EP-A-0 427 529, EP-A-451 878 and WG03/052073.
The processes for the preparation of such transgenic plants are generally known to the person skilled in the art and are described, for example, in the publications mentioned above. Cryl-type deoxyribonucleic acids and
their preparation are known, for example, from WO 95/34656, EP-A-0 367 474, EP-A-0 401 979 and WO 90/13651.
The toxin contained in the transgenic plants imparts to the plants tolerance to harmful insects. Such insects can occur in any taxonomic group of insects but are especially commonly found in the beetles (Coleoptera), two-winged insects (Diptera) and butterflies (Lepidoptera).
Transgenic plants containing one or more genes that code for an insecticidal resistance and express one or more toxins are known and some of them are commercially available. Examples of such plants are: YieldGard ® (maize variety that expresses a CrylAb toxin); YieldGard Rootworm® (maize variety that expresses a Cry3Bb1 toxin); YieldGard Plus® (maize variety that expresses a CrylAb and a Cry3Bb1 toxin); Starlink® (maize variety that expresses a Cry9C toxin); Herculex I® (maize variety that expresses a Cry1 Fa2 toxin and the enzyme phosphinothricine N-acetyltransferase (PAT) to achieve tolerance to the herbicide glufosinate ammonium); NuCOTN 33B® (cotton variety that expresses a Cry1 Ac toxin); Bollgard I® (cotton variety that expresses a Cry1 Ac toxin); Bollgard II® (cotton variety that expresses a CrylAc and a Cry2Ab toxin); VipCot ® (cotton variety that expresses a Vip3A and a CrylAb toxin); NewLeaf® (potato variety that expresses a Cry3A toxin); NatureGard®, Agrisure® GT Advantage (GA21 glyphosate-tolerant trait), Agrisure® CB Advantage (Bt11 corn borer (CB) trait) and Protecta®.
Further examples of such transgenic crops are:
1. Bt11 Maize from Syngenta Seeds SAS, Chemin de I'Hobit 27, F-31 790 St. Sauveur, France, registration number C/FR/96/05/10. Genetically modified Zea mays which has been rendered resistant to attack by the European corn borer (Ostrinia nubilalis and Sesamia nonagrioides) by transgenic expression of a truncated CrylAb toxin. Bt11 maize also transgenically expresses the enzyme PAT to achieve tolerance to the herbicide glufosinate ammonium.
2. Bt176 Maize from Syngenta Seeds SAS, Chemin de I'Hobit 27, F-31 790 St. Sauveur, France, registration number C/FR/96/05/10. Genetically modified Zea mays which has been rendered resistant to attack by the European corn borer (Ostrinia nubilalis and Sesamia nonagrioides) by transgenic expression of a CrylAb toxin. Bt176 maize also transgenically expresses the enzyme PAT to achieve tolerance to the herbicide glufosinate ammonium.
3. MIR604 Maize from Syngenta Seeds SAS, Chemin de I'Hobit 27, F-31 790 St. Sauveur, France, registration number C/FR/96/05/10. Maize which has been rendered insect-resistant by transgenic expression of a modified Cry3A toxin. This toxin is Cry3A055 modified by insertion of a cathepsin-G-protease recognition sequence. The preparation of such transgenic maize plants is described in WO 03/018810.
4. MON 863 Maize from Monsanto Europe S.A. 270-272 Avenue de Tervuren, B-1150 Brussels, Belgium, registration number C/DE/02/9. MON 863 expresses a Cry3Bb1 toxin and has resistance to certain Coleoptera insects.
5. IPC 531 Cotton from Monsanto Europe S.A. 270-272 Avenue de Tervuren, B-1150 Brussels, Belgium, registration number C/ES/96/02.
6. 1507 Maize from Pioneer Overseas Corporation, Avenue Tedesco, 7 B-1160 Brussels, Belgium, registration number C/NL/00/10. Genetically modified maize for the expression of the protein Cry1 F for achieving resistance to certain Lepidoptera insects and of the PAT protein for achieving tolerance to the herbicide glufosinate ammonium.
7. NK603 x MON 810 Maize from Monsanto Europe S.A. 270-272 Avenue de Tervuren, B-1150 Brussels, Belgium, registration number C/GB/02/M3/03. Consists of conventionally bred hybrid maize varieties by crossing the genetically modified varieties NK603 and MON 810. NK603 x MON 810 Maize transgenically expresses the protein CP4 EPSPS, obtained from Agrobacterium sp. strain CP4, which imparts tolerance to the herbicide Roundup® (contains glyphosate), and also a CrylAb toxin obtained from Bacillus thuringiensis subsp. kurstaki which brings about tolerance to certain Lepidoptera, include the European corn borer.
The term “locus” as used herein means fields in or on which plants are growing, or where seeds of cultivated plants are sown, or where seed will be placed into the soil. It includes soil, seeds, and seedlings, as well as established vegetation.
The term “plants” refers to all physical parts of a plant, including seeds, seedlings, saplings, roots, tubers, stems, stalks, foliage, and fruits.
The term “plant propagation material” is understood to denote generative parts of the plant, such as seeds, which can be used for the multiplication of the latter, and vegetative material, such as cuttings or tubers, for example potatoes. There may be mentioned for example seeds (in the strict sense), roots, fruits, tubers, bulbs, rhizomes and parts of plants. Germinated plants and young plants which are to be transplanted after germination or after emergence from the soil, may also be mentioned. These young plants may be protected before transplantation by a total or partial treatment by immersion. Preferably “plant propagation material” is understood to denote seeds.
Pesticidal agents referred to herein using their common name are known, for example, from "The Pesticide Manual", 15th Ed., British Crop Protection Council 2009.
The compounds as defined in any one of embodiments according to the invention for use in the inventive methods may be the sole active ingredient of a composition or it may be admixed with one or more additional active ingredients such as a pesticide, fungicide, synergist, herbicide, or plant growth regulator where appropriate. An additional active ingredient may, in some cases, result in unexpected synergistic activities.
The compositions according to the invention can also comprise further solid or liquid auxiliaries, such as stabilizers, for example unepoxidized or epoxidized vegetable oils (for example epoxidized coconut oil, rapeseed oil or soya oil), antifoams, for example silicone oil, preservatives, viscosity regulators, binders and/or tackifiers, fertilizers or other active ingredients for achieving specific effects, for example bactericides, fungicides, nematicides, plant activators, molluscicides or herbicides.
The compounds, and compositions according to the invention are prepared in a manner known per se, in the absence of auxiliaries for example by grinding, screening and/or compressing a solid active ingredient and in the presence of at least one auxiliary for example by intimately mixing and/or grinding the active ingredient with the auxiliary (auxiliaries). These processes for the preparation of the compositions and the use of the compounds (I) for the preparation of these compositions are also a subject of the invention.
Another aspect of the invention is related to the use of a compound as defined in any one of embodiments according to the invention, of a composition comprising at least one compound as defined in, or of a fungicidal or insecticidal mixture comprising at least one compound as defined in any one of embodiments according to the invention, in admixture with other fungicides or insecticides as described above, for controlling or preventing infestation of plants, e.g. plants such as crop plants, propagation material thereof, e.g. seeds, harvested crops, e.g. harvested food crops, or non-living material by insects or by phytopathogenic microorganisms, preferably fungal organisms.
A further aspect of invention is related to a method of controlling or preventing an infestation of plants, e.g. plants such as crop plants, propagation material thereof, e.g. seeds, harvested crops, e.g. harvested food crops, or of non-living materials by phytopathogenic or spoilage microorganisms or organisms potentially harmful to man, especially fungal organisms, which comprises the application of a compound as defined in any one of embodiments according to the invention as active ingredient to the plants, to parts of the plants or to the locus thereof, to the propagation material thereof, or to any part of the non-living materials.
Controlling or preventing means reducing infestation by insects or by phytopathogenic or spoilage microorganisms or organisms potentially harmful to man, especially fungal organisms, to such a level that an improvement is demonstrated.
A preferred method of controlling or preventing an infestation of crop plants by phytopathogenic microorganisms, especially fungal organisms, or insects which comprises the application of a compound as defined in any one of embodiments according to the invention, or an agrochemical composition which contains at least one of said compounds, is foliar application. The frequency of application and the rate of application will depend on the risk of infestation by the corresponding pathogen or insect.
However, the compounds of formula (I), preferably in association with a compound (B), can also penetrate the plant through the roots via the soil (systemic action) by drenching the locus of the plant with a liquid formulation, or by applying the compounds in solid form to the soil, e.g., in granular form (soil application). In crops of water rice such granulates can be applied to the flooded rice field. The compounds of formula (I) may also be applied to seeds (coating) by impregnating the seeds or tubers either with a liquid formulation of the fungicide or coating them with a solid formulation.
Further characteristics of preferred binary compositions comprising compounds of formula (I), their application methods to cereals and their use rates are as described for compositions comprising compounds of formula (I) and additionally preferably at least one component (B) as described above.
Their application can be both before and after the infection of the plants or parts thereof with the fungi. The treatment is preferably carried out prior to the infection. When a compound of formula (I) is used on its own, the application rates in the method according to the invention are as described above, e.g., typical are rates of 5 to 2000 g a.i./ha, particularly 10 to 1000 g a.i./ha, e.g., 50, 75, 100 or 200 g a.i./ha. Compounds of formula (I) can be applied to the plants once or more than once during a growing season.
For use in the method according to the invention, the compounds of formula (I) can be converted into the customary formulations described above, e.g., solutions, emulsions, suspensions, dusts, powders, pastes and granules. The use form will depend on the particular intended purpose; in each case, it should ensure a fine and even distribution of the compound of formula (I).
The term "plant" as used herein includes seedlings, bushes and crops of fruits and vegetables.
EXAMPLES
The Examples which follow serve to illustrate the invention and are not meant in any way to limit the invention.
The same formulations can be used for compositions comprising only a compound of formula (I) as the active ingredient.
The compounds of the invention can be distinguished from known compounds by virtue of greater efficacy at low application rates, which can be verified by a person skilled in the art using the experimental procedures outlined in the Examples, using lower application rates, if necessary, for example 60 ppm, 20 ppm or 2 ppm.
Compounds of formula (I) may possess any number of benefits including, inter alia, advantageous levels of biological activity for protecting plants against diseases that are caused by fungi or superior properties for use as agrochemical active ingredients (for example, greater biological activity, an advantageous spectrum of activity, an increased safety profile (including improved crop tolerance), improved physico-chemical properties, or increased biodegradability).
FORMULATION EXAMPLES
Wettable powders a) b) c) active ingredients 25 % 50 % 75 % sodium lignosulfonate 5 % 5 % - sodium lauryl sulfate 3 % - 5 % sodium diisobutylnaphthalenesulfonate 6 % 10 % phenol polyethylene glycol ether (7-8 mol of ethylene oxide) 2 % highly dispersed silicic acid 5 % 10 % 10 %
Kaolin 62 % 27 % -
The combination is thoroughly mixed with the adjuvants and the mixture is thoroughly ground in a suitable mill, affording wettable powders that can be diluted with water to give suspensions of the desired concentration.
Powders for dry seed treatment a) b) c) active ingredients 25 % 50 % 75 %
light mineral oil 5 % 5 % 5 % highly dispersed silicic acid 5 % 5 %
Kaolin 65 % 40 % -
Talcum 20 %
The combination is thoroughly mixed with the adjuvants and the mixture is thoroughly ground in a suitable mill, affording powders that can be used directly for seed treatment.
Emulsifiable concentrate active ingredients 10 % octylphenol polyethylene glycol ether (4-5 mol of ethylene oxide) 3 % calcium dodecylbenzene sulfonate 3 % castor oil polyglycol ether (35 mol of ethylene oxide) 4 %
Cyclohexanone 30 % xylene mixture 50 %
Emulsions of any reguired dilution, which can be used in plant protection, can be obtained from this concentrate by dilution with water.
Dusts a) b) c)
Active ingredients 5 % 6 % 4 %
Talcum 95 % -
Kaolin 94 % - mineral filler 96 %
Ready-for-use dusts are obtained by mixing the combination with the carrier and grinding the mixture in a suitable mill. Such powders can also be used for dry dressings for seed.
Extruder granules
Active ingredients 15 % sodium lignosulfonate 2 % carboxymethylcellulose 1 %
Kaolin 82 %
The combination is mixed and ground with the adjuvants, and the mixture is moistened with water. The mixture is extruded and then dried in a stream of air.
Coated granules
Active ingredients 8% polyethylene glycol (mol. wt. 200) 3 %
Kaolin 89 %
The finely ground combination is uniformly applied, in a mixer, to the kaolin moistened with polyethylene glycol. Non-dusty coated granules are obtained in this manner.
Suspension concentrate active ingredients 40 % propylene glycol 10 % nonylphenol polyethylene glycol ether (15 mol of ethylene oxide) 6 %
Sodium lignosulfonate 10 % carboxymethylcellulose 1 % silicone oil (in the form of a 75 % emulsion in water) 1 %
Water 32 %
The finely ground combination is intimately mixed with the adjuvants, giving a suspension concentrate from which suspensions of any desired dilution can be obtained by dilution with water. Using such dilutions, living plants as well as plant propagation material can be treated and protected against infestation by microorganisms, by spraying, pouring or immersion.
Flowable concentrate for seed treatment active ingredients 40 % propylene glycol 5 % copolymer butanol PO/EO 2 %
Tristyrenephenole with 10-20 moles EO 2 %
1 ,2-benzisothiazolin-3-one (in the form of a 20% solution in water) 0.5 % monoazo-pigment calcium salt 5 %
Silicone oil (in the form of a 75 % emulsion in water) 0.2 %
Water 45.3 %
The finely ground combination is intimately mixed with the adjuvants, giving a suspension concentrate from which suspensions of any desired dilution can be obtained by dilution with water. Using such dilutions, living plants as well as plant propagation material can be treated and protected against infestation by microorganisms, by spraying, pouring or immersion.
Slow-Release Capsule Suspension
28 parts of the combination are mixed with 2 parts of an aromatic solvent and 7 parts of toluene diisocyanate/polymethylene-polyphenylisocyanate-mixture (8:1). This mixture is emulsified in a mixture of 1.2 parts of polyvinylalcohol, 0.05 parts of a defoamer and 51.6 parts of water until the desired particle size is achieved. To this emulsion a mixture of 2.8 parts 1 ,6-diaminohexane in 5.3 parts of water is added. The mixture is agitated until the polymerization reaction is completed. The obtained capsule suspension is stabilized by adding 0.25 parts of a thickener and 3 parts of a dispersing agent. The capsule suspension formulation contains 28% of the active ingredients. The medium capsule diameter is 8-15 microns. The resulting formulation is applied to seeds as an aqueous suspension in an apparatus suitable for that purpose.
Formulation types include an emulsion concentrate (EC), a suspension concentrate (SC), a suspo-emulsion (SE), a capsule suspension (CS), a water dispersible granule (WG), an emulsifiable granule (EG), an emulsion,
water in oil (EO), an emulsion, oil in water (EW), a micro-emulsion (ME), an oil dispersion (OD), an oil miscible flowable (OF), an oil miscible liquid (OL), a soluble concentrate (SL), an ultra-low volume suspension (SU), an ultra-low volume liquid (UL), a technical concentrate (TK), a dispersible concentrate (DC), a wettable powder (WP), a soluble granule (SG) or any technically feasible formulation in combination with agriculturally acceptable adjuvants.
BIOLOGICAL EXAMPLES
Example B1 : Corynespora cassiicola (target leaf spot) - field trials 2023
Various compounds were formulated and applied to soybean plants in a foliar broadcast spray using a handheld horizontal boom sprayer. Three applications were made over the course of the growing season, starting approximately 45 days after crop emergence, with following applications at approximately 14-day intervals. Trials were conducted across different locations in Brazil.
Assessments of severity of infection from Corynespora cassiicola were made at approximately weekly intervals, based on the percentage of leaf area covered by disease lesions. Values are given for the reduction of disease in the treated plots relative to the untreated plots ( % of control based on AUDPC for each location), except for untreated that the values mean % severity (Table 1 and Table 2).
Improved yield is calculated by subtracting the yield of the untreated plants from the yield of the treated plants, then divide the result by the absolute value of the yield of the untreated plants and multiply the result by 100. The answer is the percent increase.
Such compounds of formula (I) according to the present invention gave at least 50% control when compared to untreated control under the same conditions, which showed extensive disease development. Further compounds of formula (I) gave a significant yield increase when compared to untreated control under the same conditions, which showed extensive disease development.
The activity of the compounds against Corynespora cassiicola in the field- providing overall control similar to that of the commercial triple mix - is surprising and unexpected given the lack of activity against the same fungus in liquid culture, especially when compared to the relative activity of the active ingredients from the commercial product in liquid culture.
Accordingly, the compounds of formula (I), (l-A), or (l-B) may for example be distinguished from other compounds by virtue of greater efficacy at low application rates, which can be verified by the person skilled in the art using the experimental procedures outlined in the above biological test, using lower application rates, if necessary, for example 6 ppm, 3 ppm, 2.2 ppm, 1 .5 ppm, 0.8 ppm or 0.74 ppm.
Claims
1. A method of controlling or preventing infestation of plants by the phytopathogenic microorganism Corynespora cassiicola, comprising applying to the phytopathogen, to the locus of the phytopathogen, or to a plant susceptible to attack by the phytopathogen, or to a propagation material thereof, a compound according to formula (I)
wherein
R1 is Ci-Csalkyl; and
2. A method of controlling or preventing infestation of plants by the phytopathogenic microorganism Corynespora cassiicola comprising applying to the phytopathogen, to the locus of the phytopathogen, or to a plant susceptible to attack by the phytopathogen, or to a propagation material thereof, a pesticidal composition comprising a compound of formula (I) as defined in claim 1 , and one or more adjuvants.
3. The method according to claim 1 or 2, wherein the compound or composition is applied in the field.
4. The method according to any one of claims 1 to 3, wherein the compound or composition is applied by means of foliar application, soil application, in furrow application or drench application.
5. The method according to any one of claims 1 to 4, wherein the compound according to formula (I) is applied at a rate of at least 10 g/ha, preferably at least 10, 15, 20, 25, 30, 35, 40, 45, 50, 55, 60 g/ha.
6. The method according to any one of claims 1 to 5, wherein the compound according to formula (I) is applied at a rate of at most 100 g/ha, preferably at most 100, 95, 90, 85, 80, 75, 70, 65 g/ha.
7. The method according to any of claims 1 to 6, wherein the compound according to formula (I) is selected from ethyl 1-[[4-[(Z)-2-ethoxy-3,3,3-trifluoro-prop-1-enoxy]phenyl]methyl]pyrazole-4- carboxylate according to structure (l-A)
ethyl 1-[[4-[[2-(trifluoromethyl)-1 ,3-dioxolan-2-yl]methoxy]phenyl]methyl]pyrazole-4-carboxylate according to structure (l-B)
8. The method according to any of claims 1 to 7, wherein the compound according to formula (I) is ethyl 1-[[4-[(Z)-2-ethoxy-3, 3, 3-trifluoro-prop-1-enoxy] phenyl] methyl] pyrazole-4-carboxylate.
9. The method according to any of claims 1 to 7, wherein the compound according to formula (I) is ethyl 1-[[4-[[2-(trifluoromethyl)-1,3-dioxolan-2-yl] methoxy] phenyl] methyl] pyrazole-4-carboxylate
10. A method for controlling or preventing infestation of plants by the phytopathogenic microorganism Corynespora cassiicola, comprising applying to the phytopathogen, to the locus of the phytopathogen, or to a plant susceptible to attack by the phytopathogen, or to a propagation material thereof, a compound of formula (I) as defined in any of claims 1 to 9, and one or more pesticide as mixing partner, or a pesticidal composition comprising a compound of formula (I) as defined in any of claims 1 , 7, 8 or 9, one or more pesticides as mixing partner, and one or more adjuvants..
11 . The method according to any one of claims 1 to 10 wherein the plant is selected from soybeans, corn, wheat, cotton, beans, potatoes, sweet potato, or tobacco.
12. The method according to any one of claims 1 to 10, wherein the plants are genetically modified plants selected from soybeans, corn, wheat, cotton, beans, potatoes, sweet potato, or tobacco.
13. The method according to any one of claims 1 to 12, wherein the plants are Bt plants.
14. The method according to according to any one of claims 1 to 13, wherein the plants are selected from soybean, or cotton, preferably soybean plants.
15. The method according to any one of claims 1 to 14, comprising the steps of providing a composition comprising a compound of formula (I) in a biocidally effective amount; and applying the composition to a propagation material; and planting the propagation material, preferably in the field.
16. The method according to any one of claims 1 to 15, comprising the steps of providing a composition comprising a compound of formula (I) in a biocidally effective amount; and applying the composition to the phytopathogen, to the locus of the phytopathogen, or to a plant susceptible to attack by the phytopathogen.
17. The use of compounds of formula (I) as defined in any of claims 1 , 7, 8 or 9 for controlling Corynespora cassiicola in genetically modified soybean, cotton, or corn according to the method of any of claim 1 to 16.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP23189877.6 | 2023-08-04 | ||
| EP23189877 | 2023-08-04 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2025031990A1 true WO2025031990A1 (en) | 2025-02-13 |
Family
ID=87557979
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2024/072024 Pending WO2025031990A1 (en) | 2023-08-04 | 2024-08-02 | Methods of controlling or preventing infestation of soybean plants by the phytopathogenic microorganism corynespora cassiicola |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2025031990A1 (en) |
Citations (73)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0353191A2 (en) | 1988-07-29 | 1990-01-31 | Ciba-Geigy Ag | DNA sequences encoding polypeptides having beta-1,3-glucanase activity |
| EP0367474A1 (en) | 1988-11-01 | 1990-05-09 | Mycogen Corporation | Novel bacillus thuringiensis isolate denoted b.t. ps81gg, active against lepidopteran pests, and a gene encoding a lepidopteran-active toxin |
| EP0374753A2 (en) | 1988-12-19 | 1990-06-27 | American Cyanamid Company | Insecticidal toxines, genes coding therefor, antibodies binding them, transgenic plant cells and plants expressing these toxines |
| EP0392225A2 (en) | 1989-03-24 | 1990-10-17 | Ciba-Geigy Ag | Disease-resistant transgenic plants |
| WO1990013651A1 (en) | 1989-05-09 | 1990-11-15 | Imperial Chemical Industries Plc | Bacterial genes |
| EP0401979A2 (en) | 1989-05-18 | 1990-12-12 | Mycogen Corporation | Novel bacillus thuringiensis isolates active against lepidopteran pests, and genes encoding novel lepidopteran-active toxins |
| EP0427529A1 (en) | 1989-11-07 | 1991-05-15 | Pioneer Hi-Bred International, Inc. | Larvicidal lectins and plant insect resistance based thereon |
| EP0451878A1 (en) | 1985-01-18 | 1991-10-16 | Plant Genetic Systems, N.V. | Modifying plants by genetic engineering to combat or control insects |
| WO1993007278A1 (en) | 1991-10-04 | 1993-04-15 | Ciba-Geigy Ag | Synthetic dna sequence having enhanced insecticidal activity in maize |
| WO1995033818A2 (en) | 1994-06-08 | 1995-12-14 | Ciba-Geigy Ag | Genes for the synthesis of antipathogenic substances |
| WO1995034656A1 (en) | 1994-06-10 | 1995-12-21 | Ciba-Geigy Ag | Novel bacillus thuringiensis genes coding toxins active against lepidopteran pests |
| WO1997033890A1 (en) | 1996-03-11 | 1997-09-18 | Novartis Ag | Pyrimidin-4-one derivatives as pesticide |
| WO2002015701A2 (en) | 2000-08-25 | 2002-02-28 | Syngenta Participations Ag | Bacillus thuringiensis crystal protein hybrids |
| WO2003018810A2 (en) | 2001-08-31 | 2003-03-06 | Syngenta Participations Ag | Modified cry3a toxins and nucleic acid sequences coding therefor |
| WO2003052073A2 (en) | 2001-12-17 | 2003-06-26 | Syngenta Participations Ag | Novel corn event |
| US6919298B2 (en) | 2002-04-04 | 2005-07-19 | Valent Biosciences Corporation | Enhanced herbicide composition |
| WO2006108647A1 (en) | 2005-04-12 | 2006-10-19 | Vekoerrer Franz | Method for producing dental molded parts |
| WO2006108675A2 (en) | 2005-04-11 | 2006-10-19 | Bayer Bioscience N.V. | Elite event a5547-127 and methods and kits for identifying such event in biological samples |
| WO2006108674A2 (en) | 2005-04-08 | 2006-10-19 | Bayer Bioscience N.V. | Elite event a2704-12 and methods and kits for identifying such event in biological samples |
| WO2006130436A2 (en) | 2005-05-27 | 2006-12-07 | Monsanto Technology Llc | Soybean event mon89788 and methods for detection thereof |
| WO2008002872A2 (en) | 2006-06-28 | 2008-01-03 | Pioneer Hi-Bred International, Inc. | Soybean event 3560.4.3.5 and compositions and methods for the identification and/or detection thereof |
| WO2008054747A2 (en) | 2006-10-31 | 2008-05-08 | E. I. Du Pont De Nemours And Company | Soybean event dp-305423-1 and compositions and methods for the identification and/or detection thereof |
| WO2009064652A1 (en) | 2007-11-15 | 2009-05-22 | Monsanto Technology Llc | Soybean plant and seed corresponding to transgenic event mon87701 and methods for detection thereof |
| WO2009102873A1 (en) | 2008-02-15 | 2009-08-20 | Monsanto Technology Llc | Soybean plant and seed corresponding to transgenic event mon87769 and methods for detection thereof |
| WO2010024976A1 (en) | 2008-08-29 | 2010-03-04 | Monsanto Technology Llc | Soybean plant and seed corresponding to transgenic event mon87754 and methods for detection thereof |
| WO2010037016A1 (en) | 2008-09-29 | 2010-04-01 | Monsanto Technology Llc | Soybean transgenic event mon87705 and methods for detection thereof |
| WO2010080829A1 (en) | 2009-01-07 | 2010-07-15 | Basf Agrochemical Products B.V. | Soybean event 127 and methods related thereto |
| WO2011034704A1 (en) | 2009-09-17 | 2011-03-24 | Monsanto Technology Llc | Soybean transgenic event mon 87708 and methods of use thereof |
| WO2011063413A2 (en) | 2009-11-23 | 2011-05-26 | Bayer Bioscience N.V. | Herbicide tolerant soybean plants and methods for identifying same |
| WO2011066360A1 (en) | 2009-11-24 | 2011-06-03 | Dow Agrosciences Llc | Detection of aad-12 soybean event 416 |
| WO2011066384A1 (en) | 2009-11-24 | 2011-06-03 | Dow Agrosciences Llc | Aad-12 event 416, related transgenic soybean lines, and event-specific identification thereof |
| WO2012033794A2 (en) | 2010-09-08 | 2012-03-15 | Dow Agrosciences Llc | Aad-12 event 1606 and related transgenic soybean lines |
| WO2012051199A2 (en) | 2010-10-12 | 2012-04-19 | Monsanto Technology Llc | Soybean plant and seed corresponding to transgenic event mon87712 and methods for detection thereof |
| WO2012075429A1 (en) | 2010-12-03 | 2012-06-07 | Dow Agrosciences Llc | Stacked herbicide tolerance event 8291.45.36.2, related transgenic soybean lines, and detection thereof |
| WO2012075426A1 (en) | 2010-12-03 | 2012-06-07 | Dow Agrosciences Llc | Stacked herbicide tolerance event 8264.44.06.1, related transgenic soybean lines, and detection thereof |
| WO2012082548A2 (en) | 2010-12-15 | 2012-06-21 | Syngenta Participations Ag | Soybean event syht0h2 and compositions and methods for detection thereof |
| WO2013010094A1 (en) | 2011-07-13 | 2013-01-17 | Dow Agrosciences Llc | Stacked herbicide tolerance event 8264.42.32.1, related transgenic soybean lines, and detection thereof |
| WO2013016527A1 (en) | 2011-07-26 | 2013-01-31 | Dow Agrosciences Llc | Insect resistant and herbicide tolerant soybean event 9582.814.19.1 |
| WO2014170327A1 (en) | 2013-04-19 | 2014-10-23 | Bayer Cropscience Ag | Method for combating pests |
| WO2014201235A2 (en) | 2013-06-14 | 2014-12-18 | Monsanto Technology Llc | Soybean transgenic event mon87751 and methods for detection and use thereof |
| GB2521255A (en) * | 2013-10-22 | 2015-06-17 | Syngenta Participations Ag | Method of combating corynespora |
| WO2015155075A1 (en) | 2014-04-11 | 2015-10-15 | Syngenta Participations Ag | Fungicidal n'-[2-methyl-6-[2-alkoxy-ethoxy]-3-pyridyl]-n-alkyl-formamidine derivatives for use in agriculture |
| WO2015185485A1 (en) | 2014-06-06 | 2015-12-10 | Basf Se | Use of substituted oxadiazoles for combating phytopathogenic fungi |
| WO2016202742A1 (en) | 2015-06-15 | 2016-12-22 | Bayer Cropscience Aktiengesellschaft | Halogen-substituted phenoxyphenylamidines and the use thereof as fungicides |
| WO2017005710A1 (en) | 2015-07-08 | 2017-01-12 | Bayer Cropscience Aktiengesellschaft | Phenoxyhalogenphenylamidines and the use thereof as fungicides |
| WO2017055473A1 (en) | 2015-10-02 | 2017-04-06 | Syngenta Participations Ag | Microbiocidal oxadiazole derivatives |
| WO2017076742A1 (en) | 2015-11-05 | 2017-05-11 | Basf Se | Substituted oxadiazoles for combating phytopathogenic fungi |
| WO2017081311A1 (en) | 2015-11-13 | 2017-05-18 | Basf Se | Substituted oxadiazoles for combating phytopathogenic fungi |
| WO2017085100A1 (en) | 2015-11-19 | 2017-05-26 | Basf Se | Substituted oxadiazoles for combating phytopathogenic fungi |
| WO2017093019A1 (en) | 2015-12-03 | 2017-06-08 | Basf Se | Substituted oxadiazoles for combating phytopathogenic fungi |
| WO2017153380A1 (en) | 2016-03-10 | 2017-09-14 | Syngenta Participations Ag | Microbiocidal quinoline (thio)carboxamide derivatives |
| WO2017178245A1 (en) | 2016-04-11 | 2017-10-19 | Basf Se | Substituted oxadiazoles for combating phytopathogenic fungi |
| US9823801B2 (en) | 2010-10-04 | 2017-11-21 | Au Optronics Corporation | Touch panel and repairing method thereof |
| WO2017207362A1 (en) | 2016-05-30 | 2017-12-07 | Syngenta Participations Ag | Microbiocidal thiazole derivatives |
| WO2017211649A1 (en) | 2016-06-09 | 2017-12-14 | Basf Se | Substituted oxadiazoles for combating phytopathogenic fungi |
| WO2018098216A1 (en) | 2016-11-22 | 2018-05-31 | Vps-3, Inc. | Use of a difluoro-(2-hydroxypropyl)pyridine compound as a fungicide for control of phytopathogenic fungi of barley |
| WO2018108977A1 (en) | 2016-12-14 | 2018-06-21 | Bayer Cropscience Aktiengesellschaft | Active compound combinations |
| WO2018145921A1 (en) | 2017-02-10 | 2018-08-16 | Bayer Aktiengesellschaft | Composition for controlling harmful microorganisms comprising 1 -(phenoxy-pyridinyl)-2-(1,2,4-triazol-1 -yl)-ethanol derivatives |
| WO2018153707A1 (en) | 2017-02-22 | 2018-08-30 | Basf Se | Crystalline forms of a strobilurin type compound for combating phytopathogenic fungi |
| WO2019093522A1 (en) | 2017-11-13 | 2019-05-16 | 株式会社クレハ | Azole derivative, intermediate compound, method for producing azole derivative, agent for agricultural and horticultural use, and material protection agent for industrial use |
| WO2019105933A1 (en) | 2017-11-29 | 2019-06-06 | Syngenta Participations Ag | Microbiocidal thiazole derivatives |
| WO2020056090A1 (en) | 2018-09-14 | 2020-03-19 | Fmc Corporation | Fungicidal halomethyl ketones and hydrates |
| WO2020109511A1 (en) | 2018-11-30 | 2020-06-04 | Syngenta Crop Protection Ag | Microbiocidal 2-acylamino-thiazole-4-carboxamide derivatives |
| WO2020109509A1 (en) | 2018-11-30 | 2020-06-04 | Syngenta Participations Ag | Microbiocidal thiazole derivatives |
| WO2020127780A1 (en) | 2018-12-20 | 2020-06-25 | Bayer Aktiengesellschaft | Heterocyclyl pyridazine as fungicidal compounds |
| WO2020193387A1 (en) | 2019-03-22 | 2020-10-01 | Syngenta Crop Protection Ag | Fungicidal compounds |
| WO2021018370A1 (en) | 2019-07-26 | 2021-02-04 | Telefonaktiebolaget Lm Ericsson (Publ) | Methods for data model sharing for a radio access network and related infrastructure |
| WO2021183707A2 (en) | 2020-03-11 | 2021-09-16 | Fmc Corporation | Fungicidal halomethyl ketones and hydrates and their mixtures |
| WO2021244952A1 (en) | 2020-06-03 | 2021-12-09 | Syngenta Crop Protection Ag | Microbiocidal derivatives |
| WO2021255070A1 (en) | 2020-06-18 | 2021-12-23 | Bayer Aktiengesellschaft | Active compound combinations |
| US20220009011A1 (en) | 2020-07-09 | 2022-01-13 | Nanjing Chervon Industry Co., Ltd. | Reciprocating saw |
| WO2023046861A1 (en) * | 2021-09-22 | 2023-03-30 | Syngenta Crop Protection Ag | Fungicidal compositions |
| JP2023078251A (en) | 2023-03-10 | 2023-06-06 | 住友化学株式会社 | Method for producing acrylic acid derivative |
-
2024
- 2024-08-02 WO PCT/EP2024/072024 patent/WO2025031990A1/en active Pending
Patent Citations (76)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0451878A1 (en) | 1985-01-18 | 1991-10-16 | Plant Genetic Systems, N.V. | Modifying plants by genetic engineering to combat or control insects |
| EP0353191A2 (en) | 1988-07-29 | 1990-01-31 | Ciba-Geigy Ag | DNA sequences encoding polypeptides having beta-1,3-glucanase activity |
| EP0367474A1 (en) | 1988-11-01 | 1990-05-09 | Mycogen Corporation | Novel bacillus thuringiensis isolate denoted b.t. ps81gg, active against lepidopteran pests, and a gene encoding a lepidopteran-active toxin |
| EP0374753A2 (en) | 1988-12-19 | 1990-06-27 | American Cyanamid Company | Insecticidal toxines, genes coding therefor, antibodies binding them, transgenic plant cells and plants expressing these toxines |
| EP0392225A2 (en) | 1989-03-24 | 1990-10-17 | Ciba-Geigy Ag | Disease-resistant transgenic plants |
| WO1990013651A1 (en) | 1989-05-09 | 1990-11-15 | Imperial Chemical Industries Plc | Bacterial genes |
| EP0401979A2 (en) | 1989-05-18 | 1990-12-12 | Mycogen Corporation | Novel bacillus thuringiensis isolates active against lepidopteran pests, and genes encoding novel lepidopteran-active toxins |
| EP0427529A1 (en) | 1989-11-07 | 1991-05-15 | Pioneer Hi-Bred International, Inc. | Larvicidal lectins and plant insect resistance based thereon |
| WO1993007278A1 (en) | 1991-10-04 | 1993-04-15 | Ciba-Geigy Ag | Synthetic dna sequence having enhanced insecticidal activity in maize |
| WO1995033818A2 (en) | 1994-06-08 | 1995-12-14 | Ciba-Geigy Ag | Genes for the synthesis of antipathogenic substances |
| WO1995034656A1 (en) | 1994-06-10 | 1995-12-21 | Ciba-Geigy Ag | Novel bacillus thuringiensis genes coding toxins active against lepidopteran pests |
| WO1997033890A1 (en) | 1996-03-11 | 1997-09-18 | Novartis Ag | Pyrimidin-4-one derivatives as pesticide |
| WO2002015701A2 (en) | 2000-08-25 | 2002-02-28 | Syngenta Participations Ag | Bacillus thuringiensis crystal protein hybrids |
| WO2003018810A2 (en) | 2001-08-31 | 2003-03-06 | Syngenta Participations Ag | Modified cry3a toxins and nucleic acid sequences coding therefor |
| WO2003052073A2 (en) | 2001-12-17 | 2003-06-26 | Syngenta Participations Ag | Novel corn event |
| US6919298B2 (en) | 2002-04-04 | 2005-07-19 | Valent Biosciences Corporation | Enhanced herbicide composition |
| WO2006108674A2 (en) | 2005-04-08 | 2006-10-19 | Bayer Bioscience N.V. | Elite event a2704-12 and methods and kits for identifying such event in biological samples |
| WO2006108675A2 (en) | 2005-04-11 | 2006-10-19 | Bayer Bioscience N.V. | Elite event a5547-127 and methods and kits for identifying such event in biological samples |
| US8952142B2 (en) | 2005-04-11 | 2015-02-10 | Bayer Cropscience N.V. | Elite event A5547-127 and methods and kits for identifying such event in biological samples |
| WO2006108647A1 (en) | 2005-04-12 | 2006-10-19 | Vekoerrer Franz | Method for producing dental molded parts |
| WO2006130436A2 (en) | 2005-05-27 | 2006-12-07 | Monsanto Technology Llc | Soybean event mon89788 and methods for detection thereof |
| WO2008002872A2 (en) | 2006-06-28 | 2008-01-03 | Pioneer Hi-Bred International, Inc. | Soybean event 3560.4.3.5 and compositions and methods for the identification and/or detection thereof |
| WO2008054747A2 (en) | 2006-10-31 | 2008-05-08 | E. I. Du Pont De Nemours And Company | Soybean event dp-305423-1 and compositions and methods for the identification and/or detection thereof |
| WO2009064652A1 (en) | 2007-11-15 | 2009-05-22 | Monsanto Technology Llc | Soybean plant and seed corresponding to transgenic event mon87701 and methods for detection thereof |
| WO2009102873A1 (en) | 2008-02-15 | 2009-08-20 | Monsanto Technology Llc | Soybean plant and seed corresponding to transgenic event mon87769 and methods for detection thereof |
| WO2010024976A1 (en) | 2008-08-29 | 2010-03-04 | Monsanto Technology Llc | Soybean plant and seed corresponding to transgenic event mon87754 and methods for detection thereof |
| WO2010037016A1 (en) | 2008-09-29 | 2010-04-01 | Monsanto Technology Llc | Soybean transgenic event mon87705 and methods for detection thereof |
| US20100080887A1 (en) | 2008-09-29 | 2010-04-01 | Monsanto Technology Llc | Soybean Transgenic Event MON87705 and Methods for Detection Thereof |
| WO2010080829A1 (en) | 2009-01-07 | 2010-07-15 | Basf Agrochemical Products B.V. | Soybean event 127 and methods related thereto |
| WO2011034704A1 (en) | 2009-09-17 | 2011-03-24 | Monsanto Technology Llc | Soybean transgenic event mon 87708 and methods of use thereof |
| WO2011063413A2 (en) | 2009-11-23 | 2011-05-26 | Bayer Bioscience N.V. | Herbicide tolerant soybean plants and methods for identifying same |
| WO2011066360A1 (en) | 2009-11-24 | 2011-06-03 | Dow Agrosciences Llc | Detection of aad-12 soybean event 416 |
| WO2011066384A1 (en) | 2009-11-24 | 2011-06-03 | Dow Agrosciences Llc | Aad-12 event 416, related transgenic soybean lines, and event-specific identification thereof |
| WO2012033794A2 (en) | 2010-09-08 | 2012-03-15 | Dow Agrosciences Llc | Aad-12 event 1606 and related transgenic soybean lines |
| US9823801B2 (en) | 2010-10-04 | 2017-11-21 | Au Optronics Corporation | Touch panel and repairing method thereof |
| WO2012051199A2 (en) | 2010-10-12 | 2012-04-19 | Monsanto Technology Llc | Soybean plant and seed corresponding to transgenic event mon87712 and methods for detection thereof |
| WO2012075429A1 (en) | 2010-12-03 | 2012-06-07 | Dow Agrosciences Llc | Stacked herbicide tolerance event 8291.45.36.2, related transgenic soybean lines, and detection thereof |
| WO2012075426A1 (en) | 2010-12-03 | 2012-06-07 | Dow Agrosciences Llc | Stacked herbicide tolerance event 8264.44.06.1, related transgenic soybean lines, and detection thereof |
| WO2012082548A2 (en) | 2010-12-15 | 2012-06-21 | Syngenta Participations Ag | Soybean event syht0h2 and compositions and methods for detection thereof |
| WO2013010094A1 (en) | 2011-07-13 | 2013-01-17 | Dow Agrosciences Llc | Stacked herbicide tolerance event 8264.42.32.1, related transgenic soybean lines, and detection thereof |
| WO2013016527A1 (en) | 2011-07-26 | 2013-01-31 | Dow Agrosciences Llc | Insect resistant and herbicide tolerant soybean event 9582.814.19.1 |
| WO2014170327A1 (en) | 2013-04-19 | 2014-10-23 | Bayer Cropscience Ag | Method for combating pests |
| WO2014201235A2 (en) | 2013-06-14 | 2014-12-18 | Monsanto Technology Llc | Soybean transgenic event mon87751 and methods for detection and use thereof |
| GB2521255A (en) * | 2013-10-22 | 2015-06-17 | Syngenta Participations Ag | Method of combating corynespora |
| WO2015155075A1 (en) | 2014-04-11 | 2015-10-15 | Syngenta Participations Ag | Fungicidal n'-[2-methyl-6-[2-alkoxy-ethoxy]-3-pyridyl]-n-alkyl-formamidine derivatives for use in agriculture |
| WO2015185485A1 (en) | 2014-06-06 | 2015-12-10 | Basf Se | Use of substituted oxadiazoles for combating phytopathogenic fungi |
| WO2016202742A1 (en) | 2015-06-15 | 2016-12-22 | Bayer Cropscience Aktiengesellschaft | Halogen-substituted phenoxyphenylamidines and the use thereof as fungicides |
| WO2017005710A1 (en) | 2015-07-08 | 2017-01-12 | Bayer Cropscience Aktiengesellschaft | Phenoxyhalogenphenylamidines and the use thereof as fungicides |
| WO2017055473A1 (en) | 2015-10-02 | 2017-04-06 | Syngenta Participations Ag | Microbiocidal oxadiazole derivatives |
| WO2017076742A1 (en) | 2015-11-05 | 2017-05-11 | Basf Se | Substituted oxadiazoles for combating phytopathogenic fungi |
| WO2017081311A1 (en) | 2015-11-13 | 2017-05-18 | Basf Se | Substituted oxadiazoles for combating phytopathogenic fungi |
| WO2017085100A1 (en) | 2015-11-19 | 2017-05-26 | Basf Se | Substituted oxadiazoles for combating phytopathogenic fungi |
| WO2017093019A1 (en) | 2015-12-03 | 2017-06-08 | Basf Se | Substituted oxadiazoles for combating phytopathogenic fungi |
| WO2017153380A1 (en) | 2016-03-10 | 2017-09-14 | Syngenta Participations Ag | Microbiocidal quinoline (thio)carboxamide derivatives |
| WO2017178245A1 (en) | 2016-04-11 | 2017-10-19 | Basf Se | Substituted oxadiazoles for combating phytopathogenic fungi |
| WO2017207362A1 (en) | 2016-05-30 | 2017-12-07 | Syngenta Participations Ag | Microbiocidal thiazole derivatives |
| WO2017211649A1 (en) | 2016-06-09 | 2017-12-14 | Basf Se | Substituted oxadiazoles for combating phytopathogenic fungi |
| WO2018098216A1 (en) | 2016-11-22 | 2018-05-31 | Vps-3, Inc. | Use of a difluoro-(2-hydroxypropyl)pyridine compound as a fungicide for control of phytopathogenic fungi of barley |
| WO2018108977A1 (en) | 2016-12-14 | 2018-06-21 | Bayer Cropscience Aktiengesellschaft | Active compound combinations |
| WO2018145921A1 (en) | 2017-02-10 | 2018-08-16 | Bayer Aktiengesellschaft | Composition for controlling harmful microorganisms comprising 1 -(phenoxy-pyridinyl)-2-(1,2,4-triazol-1 -yl)-ethanol derivatives |
| WO2018153707A1 (en) | 2017-02-22 | 2018-08-30 | Basf Se | Crystalline forms of a strobilurin type compound for combating phytopathogenic fungi |
| WO2019093522A1 (en) | 2017-11-13 | 2019-05-16 | 株式会社クレハ | Azole derivative, intermediate compound, method for producing azole derivative, agent for agricultural and horticultural use, and material protection agent for industrial use |
| WO2019105933A1 (en) | 2017-11-29 | 2019-06-06 | Syngenta Participations Ag | Microbiocidal thiazole derivatives |
| US20220030868A1 (en) * | 2018-09-14 | 2022-02-03 | Fmc Corporation | Fungicidal halomethyl ketones and hydrates |
| WO2020056090A1 (en) | 2018-09-14 | 2020-03-19 | Fmc Corporation | Fungicidal halomethyl ketones and hydrates |
| WO2020109511A1 (en) | 2018-11-30 | 2020-06-04 | Syngenta Crop Protection Ag | Microbiocidal 2-acylamino-thiazole-4-carboxamide derivatives |
| WO2020109509A1 (en) | 2018-11-30 | 2020-06-04 | Syngenta Participations Ag | Microbiocidal thiazole derivatives |
| WO2020127780A1 (en) | 2018-12-20 | 2020-06-25 | Bayer Aktiengesellschaft | Heterocyclyl pyridazine as fungicidal compounds |
| WO2020193387A1 (en) | 2019-03-22 | 2020-10-01 | Syngenta Crop Protection Ag | Fungicidal compounds |
| WO2021018370A1 (en) | 2019-07-26 | 2021-02-04 | Telefonaktiebolaget Lm Ericsson (Publ) | Methods for data model sharing for a radio access network and related infrastructure |
| WO2021183707A2 (en) | 2020-03-11 | 2021-09-16 | Fmc Corporation | Fungicidal halomethyl ketones and hydrates and their mixtures |
| WO2021244952A1 (en) | 2020-06-03 | 2021-12-09 | Syngenta Crop Protection Ag | Microbiocidal derivatives |
| WO2021255070A1 (en) | 2020-06-18 | 2021-12-23 | Bayer Aktiengesellschaft | Active compound combinations |
| US20220009011A1 (en) | 2020-07-09 | 2022-01-13 | Nanjing Chervon Industry Co., Ltd. | Reciprocating saw |
| WO2023046861A1 (en) * | 2021-09-22 | 2023-03-30 | Syngenta Crop Protection Ag | Fungicidal compositions |
| JP2023078251A (en) | 2023-03-10 | 2023-06-06 | 住友化学株式会社 | Method for producing acrylic acid derivative |
Non-Patent Citations (2)
| Title |
|---|
| "The Pesticide Manual", 2009, BRITISH CROP PROTECTION COUNCIL |
| DIXON, L. J ET AL., PHYTOPATHOLOGY, vol. 99, no. 9, 2009, pages 1015 - 27 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN106614620B (en) | fungicidal composition | |
| US10111433B2 (en) | Fungicidal compositions | |
| JP2021505586A (en) | Microbial killing phenylamidine derivative | |
| US20090054233A1 (en) | Methods for protecting useful plants or plant propagation material | |
| JP2023527893A (en) | Bactericidal composition | |
| CN115136961A (en) | Fungicidal compositions | |
| CN115334885B (en) | Method for controlling or preventing plants from being infected by plant pathogenic microorganisms such as Corynepora polytomata, soybean gray spot pathogen and/or soybean purple spot pathogen | |
| CN115297727B (en) | Method for controlling or preventing plants from being infected by plant pathogenic microorganism Corynespora polytomata | |
| WO2025031990A1 (en) | Methods of controlling or preventing infestation of soybean plants by the phytopathogenic microorganism corynespora cassiicola | |
| WO2025031989A1 (en) | Methods of controlling or preventing infestation of plants by the phytopathogenic microorganism corynespora cassiicola | |
| EP4117433A1 (en) | Methods of controlling or preventing infestation of plants by the phytopathogenic microorganism corynespora cassiicola | |
| WO2023145769A1 (en) | γ-LACTAM COMPOUND AND COMPOSITION CONTAINING SAME | |
| WO2025031913A1 (en) | Methods of controlling or preventing infestation of plants by the phytopathogenic microorganism corynespora cassiicola | |
| WO2025125639A1 (en) | Method of pathogen control in soybean | |
| WO2024223858A1 (en) | Methods of controlling or preventing infestation of plants by the phytopathogenic microorganism corynespora cassiicola | |
| JP2019094335A (en) | Method of controlling harmful arthropod using heterocyclic compound | |
| AU2023379602A1 (en) | Mixtures of succinate dehydrogenase inhibitors and picolinamides | |
| CN117241668A (en) | Microbicidal quinoline/quinoxaline benzothiazine derivatives |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 24752050 Country of ref document: EP Kind code of ref document: A1 |